Attached files
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C.20549
Form 10-K
|
(Mark One)
|
|
|
[X] ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
|
|
For the fiscal year ended December 31, 2016
|
|
|
[ ] TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
|
|
|
For the transition period from __________ to __________
|
|
|
000-53089
|
|
|
Commission File Number
|
|
|
HCi Viocare
|
|
|
(Exact name of registrant as specified in its charter)
|
|
|
Nevada
|
30-0428006
|
|
(State or other jurisdiction of incorporation or organization)
|
(I.R.S. Employer Identification No.)
|
|
Kintyre House, 209 Govan Road, Glasgow Scotland
|
G51 1HJ
|
|
(Address of principal executive offices)
|
(Zip Code)
|
|
+44 141 370 0321
|
|
|
(Registrant’s telephone number, including area code)
|
|
|
Securities registered pursuant to Section 12(b) of the Exchange Act:
|
|
|
Title of each class
|
Name of each exchange on which registered
|
|
n/a
|
n/a
|
|
Securities registered pursuant to Section 12(g) of the Exchange Act:
|
|
Common Stock
|
|
Title of class
|
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
|
Yes
|
[ ]
|
No
|
[X]
|
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act.
|
Yes
|
[X]
|
No
|
[ ]
|
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
|
Yes
|
[X]
|
No
|
[ ]
|
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files).
|
Yes
|
[X]
|
No
|
[ ]
|
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K.
|
Yes
|
[ ]
|
No
|
[X]
|
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer
|
[ ]
|
Accelerated filer
|
[ ]
|
|
Non-accelerated filer
|
[ ]
|
Smaller reporting company
|
[X]
|
|
(Do not check if a smaller reporting company)
|
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
|
Yes
|
[ ]
|
No
|
[X]
|
|
The aggregate market value of voting and non-voting common stock held by non-affiliates of the registrant as at June 29, 2016 (the last business day of the registrant’s most recently completed second quarter), was approximately $33,729,754 based on the $1.00 per share which was the last selling price of the Company’s common stock, assuming solely for the purpose of this calculation that all directors, officers and greater than 10% stockholders of the registrant are affiliates. The determination of affiliate status for this purpose is not necessarily conclusive for any other purpose.
|
APPLICABLE ONLY TO CORPORATE REGISTRANTS
Indicate the number of shares outstanding of each of the registrant’s classes of common stock, as of the latest practicable date.
|
195,064,844 shares of common stock issued and outstanding as of April 14, 2017
|
DOCUMENTS INCORPORATED BY REFERENCE
List hereunder the following documents if incorporated by reference and the Part of the Form 10-K (e.g. Part I, Part II, etc.) into which the document is incorporated: (1) Any annual report to security holders; (2) Any proxy or information statement; and (3) Any prospectus filed pursuant to Rule 424(b) or (c) of the Securities Act of 1933. The listed documents should be clearly described for identification purposes.
|
None
|
2
TABLE OF CONTENTS
|
Page
|
||
|
PART I
|
||
|
Item 1
|
Business
|
5
|
|
Item 1A
|
Risk Factors
|
29
|
|
Item 1B
|
Unresolve Unresolved Staff Comments
|
38
|
|
Item 2
|
Properties
|
38
|
|
Item 3
|
Legal Proceedings
|
39
|
|
Item 4
|
Mine Safety Disclosure’s
|
39
|
|
PART II
|
||
|
Item 5
|
Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
|
40
|
|
Item 6
|
Selected Financial Data
|
42
|
|
Item 7
|
Management’s Discussion and Analysis of Financial Condition and Results of Operations
|
42
|
|
Item 7A
|
Quantitative and Qualitative Disclosures About Market Risk
|
48
|
|
Item 8
|
Financial Statements and Supplementary Data
|
F-1
|
|
Item 9
|
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
|
49
|
|
Item 9A
|
Controls and Procedures
|
49
|
|
Item 9B
|
Other Information
|
50
|
|
PART III
|
||
|
Item 10
|
Directors, Executive Officers and Corporate Governance
|
51
|
|
Item 11
|
Executive Compensation
|
54
|
|
Item 12
|
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
|
57
|
|
Item 13
|
Certain Relationships and Related Transactions, and Director Independence
|
58
|
|
Item 14
|
Principal Accounting Fees and Services
|
61
|
|
PART IV
|
||
|
Item 15
|
Exhibits, Financial Statement Schedules
|
62
|
|
SIGNATURES
|
65
|
3
Forward Looking Statements
This Annual Report on Form 10-K (“Annual Report”) contains forward-looking statements. These statements relate to future events or our future financial performance. A number of important factors could cause our actual results to differ materially from those expressed in any forward-looking statements made by us in this Form 10-K. In some cases, you can identify forward-looking statements by terminology such as "may," "should," "expects," "plans," "anticipates," "believes," "estimates," "predicts," "potential," or "continue" or the negative of these terms or other comparable terminology. These statements are only predictions and involve known and unknown risks, uncertainties and other factors, including the risks in the section entitled "Risk Factors" and the risks set out below, any of which may cause our or our industry’s actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements.
Our business is subject to a number of risks of which you should be aware before making an investment decision. These risks are discussed more fully in the "Risk Factors" section of this annual report. These risks include, by way of example and not in limitation:
|
·
|
we have a limited operating history, have only recently commenced generating revenues in our first Prosthetics and Orthotics clinic and by way of licensing fees relative to development of our technology, and we currently have no commercial products in the market;
|
|
·
|
we currently have several technologies and product prototypes which, while nearing commercialization, require further development to market the specific application.. Our ability to generate product revenues, which may not occur for several years, if ever, will depend on the successful development and commercialization of these technologies including the “Sensor” technology, the “Socketfit” technology and product applications derived from our “Smart Insole” product prototypes as well as our recently developed portable motion capture and gait analysis technology.
|
|
·
|
our current and any future collaborations with third parties for the development and commercialization of our current product or any newly acquired products or businesses may not be successful;
|
|
·
|
we intend to acquire and operate additional clinics related to our business, however, there can be no assurance that we will successful in doing so or if that if we do acquire such clinics that they will be profitable;
|
|
·
|
risks related to the failure to successfully manage or achieve growth of our business if we are successful in the development of our technology or the acquisition of clinics; and
|
|
·
|
other risks and uncertainties related to our business strategy.
|
This list is not an exhaustive list of the factors that may affect any of our forward-looking statements. These and other factors should be considered carefully and readers should not place undue reliance on our forward-looking statements.
Forward looking statements are made based on management’s beliefs, estimates and opinions on the date the statements are made and we undertake no obligation to update forward-looking statements if these beliefs, estimates and opinions or other circumstances should change. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by applicable law, including the securities laws of the United States, we do not intend to update any of the forward-looking statements to conform these statements to actual results.
The safe harbors of forward-looking statements provided by Section 21E of the Exchange Act are unavailable to issuers of penny stock. As we issued securities at a price below $5.00 per share, our shares are considered penny stock and such safe harbors set forth under the Private Securities Litigation Reform Act of 1995 are unavailable to us.
Our financial statements are stated in United States dollars and are prepared in accordance with United States generally accepted accounting principles.
In this annual report, unless otherwise specified, all dollar amounts are expressed in United States dollars and all references to "common stock" refer to the common shares in our capital stock.
As used in this Annual Report, the terms "we," "us," "company," "our" and "Viocare" mean HCi Viocare, and our wholly-owned Scottish subsidiaries: HCi Viocare Technologies Limited and HCi Viocare Clinics UK Limited, unless otherwise indicated.
4
PART I
ITEM 1. BUSINESS
1.1 BACKGROUND
The address of our principal executive office is Kintyre House, 209 Govan Road, Glasgow G511HJ, Scotland, United Kingdom. Our telephone number is +44 141 370 0321. The Company’s web-site is http://www.hciviocare.com.
Our common stock is quoted on the OTC Markets Inc. owned and operated Inter-dealer Quotation System (“OTCQB”) under the symbol "VICA".
We were incorporated in the State of Nevada on March 26, 2007 as a company intending to sell medical devices in the northern regions of China. Our intent was to seek strategic relationships with medical device manufacturers both in China and North America with the aim to be their sales and distribution agent in northern China. We also intended to assist Chinese medical device manufacturers on the development of the North American market. The Company did not find suitable relationships with which to progress its business until it undertook a change in management in September 2013. With the change in management, the Company determined that it would initially concentrate on the development and marketing of medical devices in Europe, more particularly initially in Scotland, where management had identified several opportunities for entry into the markets for prosthetics and orthotics. We are currently engaged in healthcare innovation in the fields of prosthetics and orthotics (P&O), and we intend to be engaged in the operation of P&O total rehabilitation clinics. We have two wholly-owned Scottish subsidiaries: HCi Viocare Technologies Limited and HCi Viocare Clinics UK Limited.
On December 27, 2013, pursuant to approval of the Board of Directors and the majority stock holder on November 28, 2013 and November 29, 2013 respectively, the Company filed a Certificate of Amendment to the Articles of Incorporation of the Company for the purpose of clearly providing the directors of the Company with the authority to issue both common and preferred stock without approval by the stockholders and to grant the authority of the directors of the Company to issue such shares of common and/or preferred stock in one or more series, with such voting power, designation, preferences and rights or qualifications, limitations or restrictions as they may determine by resolution of the Board of Directors.
On January 13, 2014, the Company filed a Certificate of Designation with the Secretary of State of the State of Nevada. The Certificate of Designation sets forth the rights, preferences and privileges of a class of the Company’s preferred stock. Such class shall be designated as the “Series A Preferred Stock” and the number of shares constituting such series shall be 5,000,000 shares. The holders of Series A Preferred Stock will be entitled to a preference over all of the shares of the Company’s common stock. The holders of common stock and the holders of Series A preferred stock vote together as a single class with the holders of the Series A preferred stock having 50 votes per share of Series A preferred stock and the holders of common stock having 1 vote per share of common stock. Holders of Series A Preferred Stock are entitled to notice of any stockholders’ meeting. No shares of Series A preferred stock have been issued as of the filing date of this report.
On the 15th of January, 2014, we formed our wholly-owned subsidiary, HCi Viocare Technologies Limited (“Viocare Technologies”), a corporation incorporated pursuant to the laws of Scotland, under registration number SC467480. Viocare Technologies undertakes research, development and commercialization of state of the art, new, innovative medical devices, methods and products in the fields of prosthetics, orthotics, rehabilitation, bioengineering, mobility, diabetes, diabetic foot, tissue mechanics, ultrasonics, medical signal processing and analysis, medical technology, orthopedics and robotic surgery. Viocare Technologies also plans to expand its research and development reach, to create a stable pipeline of new products and upgrades in the fields of prosthetics, orthotics, and diabetes.
On the 12th of February, 2014 we acquired intellectual property rights ("IPR”) over an innovative medical technology entitled ‘SocketFit’ from our Board Member Dr. Christos Kapatos, as well as over any additional technologies under development or that may be developed in the future. Consideration for the acquisition of the exclusive, perpetual, revocable, transferable, royalty free worldwide ownership of the IPR, and know how to develop and exploit the IPR was the issuance of 3,500,000 shares of the common stock of the Company to Kapatos. SocketFit is a system that will help overcome technical and resource hurdles endemic to the prosthetic sector. The system has been designed with the aim of offering optimally fitted prosthetic sockets that will reduce the number of prostheses made for, resulting in a reduced number of visits by the patient to the prosthetic, and also assisting in the rehabilitation of amputees.
On March 21, 2014, the Company’s name changed from China Northern Medical Device, Inc. to HCi Viocare in regard to actions taken on November 28, 2013, when the Board of Directors of the Company approved, and recommended to the majority stockholder that they approve the name change. On November 29, 2013, the majority stockholder approved the name change by written consent in lieu of a meeting, in accordance with Nevada State Law. FINRA approved the name change with an effective date of March 21, 2014. Our trading symbol is “VICA”. We filed an amendment to our Articles of Incorporation with the Secretary of State of Nevada changing our name to HCi Viocare to be effective on March 21, 2014.
On April 17, 2014, the Company, through our Scottish subsidiary, HCi Viocare Technologies Limited, entered into an acquisition agreement with Dr. Christos Kapatos, a director of both the Company and HCi Viocare Technologies (“Kapatos”), to acquire all rights and interest in and to the background IPR for a developing technology now known as “Flexisense”. Kapatos has conceived and been working on a Smart Insole System which is believed to be a state-of-the-art, pressure and shear force sensing insole with an incorporated real-time global display application and a fully featured support web-space that suggests to the user and his podiatrist/physiotherapist when inappropriate or dangerous conditions are developing on the feet. The product is being designed to help mitigate diabetic foot complications, such as ulceration, infection and amputation
5
The competing insoles that were commercially available at this time can only monitor vertical pressure on the foot and not shear and have a significantly higher cost, and are intended for use in clinical or research environments.
As consideration for the acquisition:
|
·
|
HCi Viocare Technologies shall cause the parent Company to issue to Kapatos a total of 3,500,000 shares of the common stock of the Company on execution of the agreement and 3,500,000 shares of the common stock of the Company for each and every new version of the technology (new version to mean any update or improvement to the initial commercial product to be developed from the technology, after the commercial development of the first Market Ready Insole from the technology acquired, should the new version be developed to commercial market ready stage);
|
|
·
|
a cash bonus in the amount of $10,000,000USD should the technology be sold at any stage of development for an amount equal or greater than five hundred million ($500,000,000) cash or the equivalent thereof by way of any other consideration.
|
On May 8, 2015, the Company through its wholly owned subsidiary company HCi Viocare Technologies Limited entered into an agreement with Dr. Christos Kapatos, to amend the acquisition agreement (“Amendment of Acquisition Agreement”) dated April 17, 2014. Under the Amendment of Acquisition Agreement, the parties agreed, among certain other terms and conditions, to the issuance of 7,000,000 restricted shares of the common stock of the Company as additional consideration for all other applications of the Smart Insole technology. Furthermore, under the terms of the Amendment of Acquisition Agreement no cash bonus shall be provided to Kapatos.
On April 16, 2014, the Company entered into a Consulting Agreement with Kapatos whereby he will provide his services as Chief Technical Officer for the Company and the Company’s wholly-owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be forty thousand Euros (€40,000) (USD$43,625) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Kapatos shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million US dollars ($20,000,000 USD).
Mr. Kapatos abstained from voting on the acquisition of the IPR and the Consulting Agreement. Kapatos is an officer of the Company, and formerly an officer of the Company’s subsidiaries.
On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Dr. Christos Kapatos. During fiscal 2016 this agreement was extended for a further year.
On April 15, 2014, the Company established a scientific advisory board whereby the Company intends to appoint certain advisors that can contribute to the Company’s overall business strategy and future direction.
Concurrently, the Company appointed Professor Martha Lucia Zequera, Professor William Sandham, Professor Stephan Solomonidis and Doctor Christos Kapatos to the Advisory Board of the Company and entered into advisory board agreements with each member. Compensation for the members of the Advisory Board shall be the grant of a total of 5,000 stock options entitling the advisory board member the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five years from the date of vesting. The advisory board appointments are for a term of one year and the options granted under the agreements will vest on completion of the term of the appointment. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
On May 1, 2015, the Company approved a one-year extension of the advisory agreements entered into with Professor William Sandham, Professor Stephan Solomonidis and Doctor Christos Kapatos which thereafter shall expire on April 15, 2016.
6
Further, on April 16, 2014, the Company entered into Consulting Agreements with the members of its Scientific Advisory Board as follows:
|
-
|
Professor Stephan Solomonidis (“Solomonidis”) will provide services as Head of Research for the Company and the Company’s wholly owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be twenty thousand pounds (£20,000 GBP) (USD$29,650) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Solomonidis’ fee will be readjusted to the amount of forty thousand pounds (£40,000 GBP) (USD$59,300) per annum, and he shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million dollars ($20,000,000 USD).
On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Professor Stephan Solomonidis. When the contract came up for renewal in April, 2016,it was not renewed for a further term.
|
|
|
-
|
Professor Martha Lucia Zequera (“Zequera”) will provide services as Director of Diabetic Foot Related Products and Services for the Company and the Company’s wholly owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be twenty thousand dollars ($20,000 USD) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Zequera’s fee will be readjusted to the amount of forty thousand dollars ($40,000 USD) per annum, and she shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million dollars ($20,000,000 USD).
The consulting agreement entered into with Professor Martha Lucia Zequera was not renewed upon its expiration.
|
|
-
|
Professor William Sandham (“Sandham”) will provide services as Director of Diabetes Technology Research for the Company and the Company’s wholly owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be Twenty Thousand pounds (£20,000 GBP) (USD$29,650) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Sandham’s fee will be readjusted to the amount of forty thousand pounds (£40,000 GBP) (USD$59,300) per annum, and he shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million dollars ($20,000,000 USD).
On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Professor William Sandham. When the contract came up for renewal in April, 2016,it was not renewed for a further term.
|
|
-
|
On June 9, 2014, the Company appointed Mr. William Spence to the Advisory Board of the Company and entered into an advisory board agreement. Compensation shall be the grant of a total of 5,000 stock options entitling Mr. William Spence the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share. The advisory board appointment is for a term of one year and the options granted under the agreement will vest on completion of the term of the appointment. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
The consulting agreement entered into with Mr. William Spence was not renewed upon its expiration.
|
On June 9, 2014, the Company, through our Scottish subsidiary, HCi Viocare Clinics, entered into a Share Purchase Agreement with Mr. William Donald Spence, Miss Catriona Ann Spence, and Mrs. Eilidh Isabel Malcolm (together the “vendors”), to acquire 100% of the issued capital of W D Spence Prosthetics Limited (the “Clinic”). The Clinic is incorporated in Scotland under company number SC307652, having its registered office at 8 Tomcroy Terrace, Pitlochry, Perthshire, PH16 5JA, UK. The Clinic is a private company limited by shares, with no registered charges, and the total issued share capital amounts to 1,000 ordinary shares par value of £1 each. The vendors to the Share Purchase Agreement are the beneficial owners and registered holders of all (100%) the shares of the Clinic, with Mr. William Donald Spence holding 520 ordinary shares, and Miss Catriona Ann Spence and Mrs. Eilidh Isabel Malcolm each holding 240 ordinary shares. Mr. Spence is also the director of the Clinic, and Miss Malcolm is the secretary. The Clinic has no employees or subsidiaries, and is not a subsidiary of another company.
The Clinic is located in Glasgow, Scotland, and is a fully operational prosthetics clinic. The acquisition of the Clinic is the first step to the Company’s and HCi Viocare Clinics’ intention to develop the first chain of prosthetics and orthotics (P&O) and diabetes clinics in the European market, covering Southern Europe, the Middle East and North Africa.
7
As consideration for the acquisition, HCi Viocare Clinics shall pay to the vendors in cash the amount of one hundred thousand GBP (£100,000) (USD$168,121) on execution of the agreement.
Further, on June 10, 2014, HCi Viocare Clinics entered into a Taxation Undertaking with the vendors pursuant to the Share Purchase Agreement. Concurrently, on June 10, 2014, the Company made the required payment under the agreement to the vendors, thus completing the acquisition of the Clinic.
Also on June 9, 2014, HCi Viocare Clinics entered into an agreement with Mr. William Donald Spence (the “Employment Agreement”) whereby he will provide his services as Director of Clinical Operations of HCi Viocare Clinics and its wholly-owned subsidiary clinics. The contract has a term of one year, starting on June 10, 2014, and ending on June 9, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation shall be sixty thousand GBP (£60,000) (USD$88,950) per year payable monthly in arrears on the last Friday of every month by credit transfer. HCi Viocare Clinics shall also pay any fees in connection with the membership of Mr. Spence to any professional, regulatory or other such body of which Mr. Spence’s membership is required to carry out his role with HCi Viocare Clinics, as well as all insurance payments. During the period ended June 30, 2015 Mr. Spence resigned as the Director of Clinical Operations and his annual contract was not renewed.
Further, on June 9, 2014, the Company appointed Mr. William Spence to the Advisory Board of the Company and entered into an advisory board agreement. Compensation shall be the grant of a total of 5,000 stock options entitling Mr. William Spence the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from the date of vesting. The advisory board appointment is for a term of one year and the options granted under the agreement will vest on completion of the term of the appointment. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
The advisory board agreement entered into with William Spence was not renewed upon its expiration date.
On November 7, 2014, the Company’s subsidiary, HCi Viocare Clinics entered into a service agreement with Mrs. Heleen Francoise Kist whereby she will provide her services as Chief Operating Officer (“COO”) of the Company. The contract has a term of one year, being effective as of October 1, 2014, and ending on September 30, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation was thirty thousand GBP (£30,000) (USD$44,475) per year payable monthly in arrears on the last Friday of every month by credit transfer. On October 15, 2015, the Company’s COO, Heleen Kist and wholly owned subsidiary, HCI Viocare Clinics UK, signed an Addendum to an employment contract to extend the term to September 30, 2016, with a remuneration increase to forty five thousand GBP (£45,000) (USD$ 66,712) per year. Subsequently, on August 29, 2016 the Company’s subsidiary, HCi Viocare Clinics UK Ltd. approved a termination agreement with Heleen Kist which became effective at the close of her existing contract. Mrs. Kist resigned her positions as an officer and under the terms of the termination agreement the Company has undertaken to repurchase a total of seven hundred thousand (700,000) shares of the Company’s common stock at a purchase price of US$0.02 per share for total of fourteen thousand (US$14,000) USD for return to treasury and cancellation.
On January 1, 2015, the Company entered into a consulting agreement with an individual residing in the Czech Republic, for the provision of Investor Relations Services for the Company. Under the terms of the agreement, Mikulas Dylowicz shall be an independent contractor and shall provide services including responding to shareholder and investor email and telephone inquiries, dissemination of the Company’s public press releases and working with the Company to continue to raise operating capital. Mikulas Dylowicz shall receive monthly consideration of Euros €2,000 per month and shall be reimbursed for all approved out-of-pocket expenses including travel and office expense. During fiscal 2016 the consulting agreement with Mr. Dylowicz was terminated.
On March 2, 2015, the Company announced the conclusion of a lease agreement for a modern, stand-alone 5,300 square foot facility in Glasgow, Scotland with an entry date of 1 March, 2015. The building hosts the Company’s first Viocare centre, a full service Prosthetic and Orthotic (“P&O”) practice with a superior standard of personalised care. This new facility serves as the centre of reference and training for the intended chain of Viocare P&O and diabetic foot centres across Europe, ensuring their adherence to British and International standards. The Company’s Technologies subsidiary occupies the first floor of the building to continue to develop its portfolio of bioengineering innovations.
8
On March 31, 2015, the Company’s wholly owned subsidiary HCi Viocare Clinics and its subsidiary W D Spence Prosthetics Limited completed a merger with the resulting combined entity having the name HCi Viocare Clinics UK Limited.
On May 7, 2015, the Company entered into an agreement with HELLENIC AMERICAN SECURITIES S.A. (“HAS S.A.”) for a term of one year. According to the respective agreement HAS S.A. shall provide its services and advice to the Company regarding the capital structure, industry strategy and similar matters, as well mergers and acquisitions under the Greek Law, an introduction of the Company’s securities to Greek and Foreign Investors, both Private and Institutional ones, as well as preparation of analyses for the Company which will be circulated to various websites and media groups.
HAS S.A. is a Public Limited Stock Brokerage Company, member of the Athens Stock Exchange and Athens Derivative Exchange, incorporated in Greece, with registered office in Athens.
HAS S.A. is entitled to the following consideration under the Agreement:
1. $3,000 per month or its current equivalent in Euro, plus applicable VAT, payable monthly on the first day of the month, for each quarter;
2. Issuance of 168,000 restricted shares of the Company’s common stock quarterly (the “Shares”), payable in monthly installments as to 56,000 shares per month, to be issued and delivered within 10 days after the 1st day of each month.
The contract expired and on July 6, 2016 at which time the Company and HAS S.A. entered into a Mutual Release and Waiver of Compensation Agreement where under HAS S.A. has agreed to waive all further compensation payable under the terms of the original Agreement in the total amount of $33,000 and 560,000 shares of common stock as at the expiry date. The Company recorded a gain of $469,640 against consulting fees due to this debt extinguishment.
On June 11, 2015, the Company entered into a Loan Agreement with the Company’s CEO, President and Director, Sotirios Leontaritis, (the “Lender”), whereby the Lender, in order to provide general working capital and to allow the Company to effectuate its business plan, agreed to provide the Company a loan facility of EUR150,000 with an annual interest of 5%, payable on maturity, and the Company agreed to repay the loan within 5 years from the date of the loan agreement. This loan was settled in full during fiscal 2016.
On July 8, 2015, the Company incorporated HCi Viocare Clinics (Hellas) S.A. in order to carry out operations for the P&O clinic under development in Athens, Greece.
On August 8, 2015 FINRA approved a seven (7) new for one (1) old forward split of our authorized and issued and outstanding shares of common. A Certificate of Change for the stock split was filed and became effective with the Nevada Secretary of State on August 7, 2015. Consequently, our authorized share capital increased from 100,000,000 to 700,000,000 shares of common stock and our issued and outstanding common stock increased accordingly, all with a par value of $0.0001. Our preferred stock remained unchanged. All share and per share figures contained herein reflect the impact of the forward split.
On July 18, 2015, the Company appointed Mr. Yiannis Levantis as a member of the Board of Directors.
On September 1, 2015, the Board of Directors approved a consulting agreement with Sergios Katsaros and appointed Mr. Katsaros Vice President of HCi Viocare.
Under the terms of the consulting agreement, Mr. Katsaros will work directly with the Company’s President and CEO in order to create and implement the Company’s strategic plan and assist in securing additional financing to meet the needs of the Company’s business plan and corporate objectives. The initial term of the contract is six months and Mr. Katsaros will receive compensation of EUR2,000 per month. Further, on March 1, 2016 the Company approved a one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros, originally entered into on September 1, 2015. On March 1, 2017, the Company approved another one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros. The revision to the Agreement ("Addendum No. 2") has a term of one year, being effective as of March 1, 2017, and ending on February 28, 2018. All other terms and conditions remain valid and in force.
On September 1, 2015, the Company appointed Ms. Evaggelia Moschidou as Marketing Director of the Company. Subsequently during fiscal 2016 the contract with Ms. Moshidou was not renewed and the parties agreed to services on a month to month basis.
9
On September 25, 2015, the official opening of the Company’s new Research and Development (R&D) center together with its first Prosthetics and Orthotics (P&O) clinic in the UK took place in Glasgow, Scotland with an official ribbon cutting by Scotland’s First Minister, Nicola Sturgeon MSP. Additional information and updates on the Company’s Clinic and R&D center can be found at the new Glasgow clinic website: www.hci-viocare.co.uk and on Twitter: @HCiVioClinic.
On February 1, 2016, the Company entered into a consulting agreement with Sitimo Ltd. for the development and expansion of its business and establishment of Prosthetics and Orthotics clinics in the region of South Eastern Europe as well as the Middle East. The Consultant undertakes to introduce the Company to the healthcare market of South Eastern Europe and Middle East.
The Agreement has a term of twelve (12) months and the Consultant is entitled to remuneration for the provision of services in the form of 100,000 shares of the common stock to be issued within thirty days from the enforcement of the Agreement.
On February 2, 2016, the Company entered into an Agreement with LXM Finance LLP (“LXM”), a company with a registered office in London, UK in order to be assisted with fund raising. LXM is member of LXM Group is a joint venture between the company’s management and the owners of the Libra Group, an international business group with operations spanning five continents and 30 locations worldwide. The group has a multi-billion dollar diversified investment portfolio with interests principally in shipping, aviation, hospitality, real estate and renewable energy.
On February 10, 2016, the Company entered into a consulting agreement with an individual for the introduction of the Company to the healthcare market of Spain in view of the Company's plans to expand its clinics business and establish a Prosthetic and Orthotic Clinic in Spain. The Agreement has a six-month term ending on August 2, 2016, and the Consultant shall be remunerated with 50,000 shares of the Company's common stock.
On February 18, 2016, the Company entered into a consulting agreement with an individual for the introduction of the Company to the healthcare market of Middle East and specifically of Saudi Arabia in view of the Company project to expand its business and establish a Prosthetic and Orthotic Clinic in the Middle East. The Agreement has a six-month term ending on August 18, 2016 and the Consultant shall be remunerated with 200,000 shares of the common stock.
On February 19, 2016, the Company entered into a consulting agreement with an individual for the introduction of the Company to the healthcare market of Turkey in view of the Company project to expand its business and establish a Prosthetic and Orthotic Clinic in Turkey. The Agreement has a six-month term ending on August 19, 2016 and the Consultant shall be remunerated with 200,000 shares of the common stock.
On April 11, 2016 the Company announced the branding of its unique smart insole technology as “FlexisenseTM”.
On May 18, 2016, the Company entered into a Professional Relations and Consulting Agreement with Acorn Management Partners, LLC (“Acorn”), a Georgia limited liability company, for a term of 12 months. Under the terms of the agreement, Acorn will create a market awareness program and introduce financial professionals to the Company targeting the US marketplace. The program shall be designed to build long-term relationships between the Company and licensed financial professionals. In consideration of the Services, the Company agrees to pay to Acorn the following fees:
|
·
|
First 6 Month period
|
-Cash: $17,500 per month, monthly payment made upon the execution of the contract
-Stock: $250,000 dollars’ worth of restricted 144 common stock of the Company (“VICA”), due upon the execution of contract. The number of the shares to be determined by dividing $250,000 by the closing price on the last trading day’s closing price on the day before the execution day.
|
·
|
Second 6 Month period
|
-Cash: $17,500 per month, monthly payment made upon the execution of the contract
-Stock: $250,000 dollars’ worth of restricted 144 common stock of the Company (“VICA”), due upon the execution of contract. The number of the shares to be determined by dividing $250,000 by the closing price on the last trading day’s closing price on the day before the execution day. The Company terminated its agreement with Acorn prior to the close of the initial six-month period.
10
On September 15, 2016, the Company appointed Mr. Nikolaos Kardaras as Director of its subsidiary HCi Viocare Clinics UK Ltd. and HCi Viocare Technologies, Inc. subsequent to the resignation of Dr. Christos Kapatos from both corporations effective June 27, 2016.
On September 23, 2016 the Company entered into a licensing agreement with respect to certain applications of its Flexisense™ Technology with Carilex Medical Inc. a leading medical mattresses manufacturer. Under the terms of the agreement the Company will receive a Technology Access Fee, as well as certain staggered success fee payments as each commercialized product under the scope of the agreement is completed. In addition, there are product based royalty fees payable to the Company on a sliding scale based on production levels for all commercial products sold into the marketplace.
On November 3, 2016, the Company entered into a services agreement with a company under the name KCN Ltd. for the introduction of the Company to potential investors in the Middle East and specifically in Saudi Arabia in view of the Company project to expand its business and establish three (3) Prosthetic and Orthotic Clinics in the region. The services agreement has a six-month term ending on May 2, 2017, and the Consultant shall be remunerated with a success fee of 2% of the investment amount.
On December 20, 2016, the Company appointed Mr. Brian Maguire as Director of its subsidiary HCi Viocare Clinics UK Ltd.
On January 1, 2017 the Company approved a three-year extension to the consulting agreement between the Company and its President & CEO and controlling shareholder, Sotirios Leontaritis, originally entered into on January 1, 2014.
The revision to the Agreement ("Addendum No. 1") has a term of three years, being effective as of January 1, 2017, and ending on December 31, 2019, renewable for such further term as may be mutually agreed between the parties. As per Addendum No. 1 the Company shall pay to Leontaritis US$120,000 per annum payable in monthly payments of US$10,000 a month for the term of the contract. Further, under the terms of the Addendum No. 1, Leontaritis is entitled to acquire at his discretion 3,000,000 shares of the common stock at a price of $0.30 per share for a term of five (5) years.
Additional information on the Company’s activities, technologies and management team can be found at the Company’s website: http://www.hciviocare.com
Other than as set out herein, we have not been involved in any bankruptcy, receivership or similar proceedings, nor have we been a party to any material reclassification, merger, consolidation or purchase or sale of a significant amount of assets not in the ordinary course of our business.
11
Our Current Business – Research & Development and commercialization of technology for healthcare innovation including various athletic applications, and the operation of Prosthetics and Orthotics clinics.
The Company intends to exploit the growing need for high quality care and prevention in the growing population of the obese, diabetic, amputated and/or movement impaired through development of:
|
-
|
Chain of Clinics: Prosthetic, Orthotic and (diabetic foot) rehabilitation centres in poorly served markets, such as Mediterranean Europe & the Middle East. Differentiation through British standards in clinical practice, qualifications and training.
|
|
-
|
Technologies: Suite of patented solutions to help prevent, monitor & alleviate complications across different patient and athletic populations. Collaboration with and licensing to established industry participants globally to facilitate rapid development, distribution and scale.
|
We commenced modest revenue generating operations in fiscal 2014 and are primarily engaged in the research and development, commercialization and marketing of innovative medical technologies including wearable devices and on body applications that have use in insoles, in shoes, in cushions, seats, mattresses, saddles and any other wearable devices such as smart apparel and sports equipment. Further we are in development for more far reaching applications including use in automotive tires and other targeted industries where sensor technology may have valuable application. Further we continue to operate and expand our brand of prosthetics and orthotics (“P&O”) and diabetic foot total rehabilitation clinics. During fiscal 2016 our flagship prosthetics clinic in Glasgow Scotland completed its first full year of operations after relocation to a larger state of the art facility which allowed for a substantive increase in our year over year revenues. Presently our revenue stream is derived primarily from the Company’s flagship P&O clinic located in Glasgow, Scotland, and with some initial commercial income from prospective licensing partners for the Technologies businesses.
The Company’s technologies and R&D portfolio presently consists of:
|
•
|
Filing of 3 patents in the UK: pressure sensor system; pressure & shear; pressure & positioning (Dec 2014) (all of which patents were abandoned upon the filing of a new International (PCT) patent application in December 2015 which covers over 140 countries around the world, and includes further advances made to the sensor systems, and a wider range of applications in products that touch people’s everyday lives;
|
|
•
|
2 further patents drafted and pending filing during fiscal 2017;
|
|
•
|
In excess of 4 prototypes: pressure & shear insole; smart cushion, smart mattress, with several of these prototypes in the commercialization stage;
|
|
•
|
1 proof of concept device: robotic surgical assistive device;
|
|
•
|
High level diagrams or complex documentation and software for other innovations in the portfolio.
|
|
•
|
Various customized prototypes in testing and development with commercial partners.
|
1.2 OUR TECHNOLOGIES
Research and Development
As at December 31, 2016 and 2015, the date of this Annual Report, we had expended a total of $340,782 and $5,317,942 respectively on research and development, including costs related to shares issued for acquisition of our Insole and SocketFit technologies and their derivative applications.
12
|
A.
|
The Sensor Technology
|
As part of our ongoing development efforts stemming from R&D related to our base technologies acquired in fiscal 2014 from Dr. Christos Kapatos, notably the Smart Insole technology, HCi Viocare has developed a sensor system that can be incorporated into any thin, flexible, bendable material and is capable of measuring direct force applied vertically and shear forces applied horizontally. Each sensor can connect with other sensors, creating a network, in order to monitor an entire area.
Due to the nature of the technology used, which is currently being kept confidential, the sensor is unaffected by bending, temperature, humidity and excess pressure, load, making it suable for medical applications, wearable devices and on-body applications. The sensor system has been designed to be used in insoles, in shoes, in cushions, seats, mattresses, saddles and any other wearable devices such as smart apparel and sports equipment. A key feature of the sensor system is its low cost, with the components for each one sensing point measuring pressure and shear forces at a cost of US$1.10, which is an order of magnitude lower than conventional pressure-only sensing solutions such as capacitance, resistance or strain gauges.
The sensor system has been integrated into a prototype of an insole which is an autonomous, non-invasive, stand-alone unit, with its own rechargeable power supply, microcontroller and wireless communication unit. The insole can connect with smartphones, tablet pcs and pcs, through a mobile application, in order to transmit the data collected to the end user. End users could be a diabetic patient, to monitor pressure and shear forces for the prevention of diabetic foot ulcers, athletes interested in the impact on their knees of their running style or wanting to monitor the totality of their golf swing, or clinicians such as physiotherapists, orthotists and prosthetists needing to evaluate a person’s gait.
The Company believes that there are applications for its force sensing technology in many fields within healthcare and sports, as well as more industrial fields due to its relative low cost vs conventional pressure sensing, its ability to measure shear forces in addition to pressure, its bendability and its reliability in a wide range of environments.
During fiscal 2016 the Company branded its unique area of sensor based technologies, “FlexisenseTM”. Please visit http://flexisense.hciviocare.com.
Flexisense is a thin, flexible, bendable, sensor drone micro-electro-mechanical system capable of measuring direct force applied vertically on both its sides, shear force applied on its surface and 3D position and velocity of the object attached to it. The sensor is an autonomous, non-invasive, stand-alone unit, with its own rechargeable power supply, micro-controller and wireless communication unit. The drone can wirelessly connect with other drones, creating a network, in order to monitor an entire area. It can also connect with smartphones, tablet and computers, through an application which has also been developed, in order to transmit the data collected to the end user.
Flexisense is unaffected by bending, temperature, humidity and excess pressure, load and shear, and completely insulated, making it suitable for medical applications, wearable devices and on-body applications. The drone can be made in a plethora of shapes and sizes, to fit any application/situation. The drone has been designed to be used in insoles, in shoes, in cushions, seats, mattresses, saddles and any other wearable devices such as smart apparel and sports equipment.
Flexisense has application in every operation that a “fusion” force/pressure, shear and 3D position and velocity sensor is required. However, it was designed and developed specifically as a non-invasive medical sensor to be used in wearable devices, smart garments, training apparatus and for medical applications such as on-body, non-invasive, real time force, pressure, shear and 3D position and velocity measurements. For example, such a sensor can be invaluable for use in the monitoring of a person’s gait providing data about the pressures applied on the foot, the interaction between shoe and foot and the forces applied in the system and the angles of the joints and posture position.
The Company concentrated its R&D efforts during early fiscal 2016 on the expansion of the commercial development of Flexisense and the second half of 2016 on seeking licensing partners for Flexisense and mattress application of the technologies, with conversations taking place with various industry partners. During fiscal 2016, the Company entered into an agreement with Carilex Medical Inc. a leading medical mattresses manufacturer. Under the terms of the agreement the Company received a technology access fee, and will receive certain staggered success fee payments as each commercialized product under the scope of the agreement is completed. In addition, there are product based royalty fees payable to the Company on a sliding scale based on production levels for all commercial products sold into the marketplace.
In addition, we have entered into several cooperative agreements with industry partners across several other areas of application for our technologies and we have customized prototypes under review with various commercial partners for commercialization in fiscal 2017.
Flexisense Application: Diabetic Insole
The Company has developed a prototype for a smart insole measuring pressure and shear forces that can communicate wirelessly to smartphones and could be worn by diabetic patients to alert them to risky behavior and provide them and their clinicians with information to help prevent the diabetic foot ulcers that can ultimately result in amputation.
Many complications can be associated with diabetes. Diabetes disrupts the vascular system, affecting many areas of the body such as the eyes, kidneys, legs, and feet. People with diabetes are more prone to infection. They can also develop neuropathy (damaged nerves) or peripheral vascular disease (blocked arteries) of the legs. This can cause diminished feeling in the feet leading to unnoticed cuts, scratches, and tissue breakdown. This may result in ulceration and infection. Infection and foot ulceration, alone or in combination, often lead to amputation.
13
In terms of market size of the opportunity: foot complaints are the leading cause of hospitalisation of people with diabetes and is estimated that 30% of all diabetics will develop a serious foot complaint at some time (http://www.who.int/topics/diabetes_mellitus/en/). In the USA alone the value of the diabetic foot ulcers therapeutics market in 2011 was $1.53 billion, showing continuous growth.
(http://www.companiesandmarkets.com/News/Healthcare-and-Medical/Diabetic-foot-ulcers-therapeutics-market-expected-to-maintain-a-moderate-growth/NI3941
It is widely understood that pressure and shear forces on the feet cause ulcerations and due to diabetic patients having neuropathy (resulting in loss of sensation in the feet), feet are not offloaded or taken care of appropriately.
It is very important for diabetics to take the necessary precautions to prevent all foot related injuries. Due to the consequences of neuropathy, daily observation of the feet is critical. Theoretically, if diabetes is well controlled and monitored it should be possible to avoid these foot problems. However, whilst clinical advice to diabetics is to ‘mind their feet’ and monitor them daily, they are not provided with a usable tool to measure the stresses and strains on their feet where there is some loss of sensation. This is what the insole intends to address.
The resulting product(s) will be a Class 1 medical device, depending on claims and disclaimers. While default alert settings would be provided, based on existing academic and clinical research, users would be encouraged to get their physician to set parameters.
The Company is not alone in recognizing the importance of measuring pressure on the feet of diabetics. Orpyx in Canada has launched a consumer product priced at $2,400, which requires an additional on-shoe component and a dedicated watch vs. HCi Viocare’s fully integrated insole which could be marketed at <$500. A few other start-up companies (FeetMe, Sensoria) and research groups are working on diabetic foot monitoring insoles, but all only measure pressure, ignoring shear forces which can greatly impact ulceration, and use conversional pressure sensing (capacitive or resistive technology) which the Company feels may not be as reliable in the hot and humid environment of a shoe. Because HCi Viocare’s technology measures all three forces (pressure and 2 horizontal shear forces) we are able to model with reasonable accuracy the calorific expenditure of the wearer, thereby potentially contributing to the diabetic patient’s glucose management.
The Company is presently seeking licensing partners in respect of the diabetic insole application. The license partner could be any company marketing products to diabetics (e.g. blood glucose monitors and lancets) or wearable technologies company wanting to extend its healthcare reach (iHealth, Samsung) or foot specialist already catering to diabetic foot market (eg Dr. Scholl).
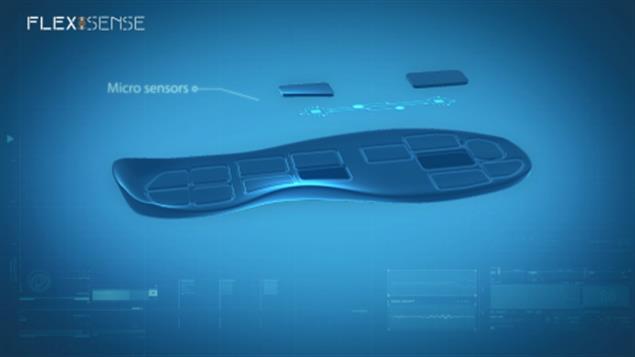
Figure: Flexisense application to smart insole
14
Flexisense Application: Medical gait analysis insoles
Orthotists, podiatrists, physiotherapists and prosthetists all require a good understanding of a patient’s gait in order to diagnose biomechanical issues, provide appropriate rehabilitation and ensure appropriate alignment of the body when using orthoses (such as braces) or prosthetic limbs. Equally, researchers in these fields and biomedical engineering more generally are interested in the forces involved in gait and their impact on the body.
Academic and commercial researchers may have force plates at their disposal: metal plates built into the floor, which can capture the forces of a person walking or running over them. However, there are prohibitively priced (>$40,000) for most clinicians. There are therefore different systems on the market addressing clinicians and researchers:
|
·
|
optical gait analysis: whilst these can show movement very well, they do not yield any information on forces
|
|
·
|
pressure mats: these measure pressure but have a small surface meaning that one can only capture a snapshot of the gait
|
|
·
|
pressure sensing insoles: which can measure pressure more realistically over a larger distance
|
Presently we are aware of 3 devices that compete directly with the insole intended for clinical use only, each priced at in excess of $4,000: from Algeos (www.algeos.co.uk) which attaches sensors directly to the foot; and insoles from Tekscan (www.tekscan.com) , from Novel, (www.novel.de) and Moticon (www.moticon.de).
Management believes its medical insole would be unique in its ability to measure shear forces, and to operate for longer periods of time reliability and accurately due to limitations of the competing technologies (eg in humidity, when bent). Moreover, unlike the HCi Viocare prototype, existing insoles are not stand-alone, requiring ancillary componentry to communicate with the data capture unit and therefore making their use restricted to in-clinic. A cheaper, stand-alone insole could be given to patients to take away and record gait over time and over different surfaces.
The medical insole can also be expanded to become a combined 3D motion capture and gait analysis system for use by physiotherapists, orthotists, athletes and possibly in film and gaming.
Flexisense Application: Athletic insoles
Sports and fitness wearable devices have been in the market for more than 30 years. However, in the last 3 years the application of micro-electronics, wireless technology and the explosion of smartphones and mobile applications has revolutionised the market, with many established, as well as new, companies offering products applications for a wide range of sports.
Wearable wireless devices in the Sports, Fitness and Wellness market measure sports performance, daily activity, sleep patterns, and other related physical parameters. Another key factor that distinguishes this market is its consumer focus and that, unlike devices in the other healthcare markets, these devices usually do not require regulatory approval as a medical device.
The consumer market for wearable devices is exploding, and people appear to have an insatiable appetite for data to analyse their fitness and athletic performance. Nearly 50 million sports performance monitors were sold in 2014 and Worldwide revenue is forecast to reach $2.3 billion in 2017. http://cdn2.hubspot.net/hub/396065/file-2568104498-pdf/Blog_Resources/IHS-Wearable-Technology.pdf?t=1427903372862
Companies like Adidas-Salomon Ag, Nike Inc., Garmin, New Balance and Suunto invest millions of dollars every year for the development of wearable devices and material, and the market is booming as demand is ever increasing. The focus for most of these companies seems to be in the running shoes, apparel and equipment sector, as this sector is the fastest growing sports and fitness sector world-wide with billions of dollars of revenue every year. Under Armour has just launched its first smart shoe, monitoring only cadence.
15
The Company’s sensor technology can be used in an insole or built into shoes to provide information to the wearer. It is the only pressure sensing technology that can work accurately in a warm and moist environment and with significant bending, like that experienced during running.
In keeping with the attractiveness of the market, there are a number of relatively new companies intending to launch insoles to give information to runners, together with coaching ‘Apps’. Some of these measure only contact or movement (e.g., www.ambiorun.com) and others do measure pressure, usually not fully wireless/stand-alone (Medhab www.medhab.com; Arion www.ato-gear.com). Another competitor is Sensoria which markets a smart textile sock that measures pressure and pace. It also has a ‘running coach’ App. It requires an anklet to be worn, to house the battery and communication modules, and the socks require washing, potentially reducing their life span.
Note that the resistive or capacitive technologies used by competition struggle with bending and hot/humid environments and are therefore considered to be less reliable. Also, due to the cost associated with pressure sensing, competitors usually only have a small number of sensing points, limiting the value of the information. The Company’s more cost effective sensing technology would permit a much greater number of points.
A further important differentiator from the competition is that the Company’s sensor technology measures forces in three directions, giving not just plantar pressure information but also making the system capable of using industry accepted biomechanical modelling capabilities to project the impact on the knee of the wearer’s running style. And the knee is an area of great focus for runners, who frequently experience injuries.
The athletic insoles could be coupled with other devices to give wearable performance information across a number of sports, including the high value tennis and golf markets. Golfers, for example would be able to link their swing (from an on-club sensor) to their weight displacement to optimise their performance. Currently on-racket or on-club sensors exist but fail to address this critical weight placement / foot force element for the athletes.
The Company is presently seeking licensing partners in respect of the athletic insole applications, with a number of discussions with market leaders in their fields ongoing and additional commercial prototypes under review.
Smart Mattress
Ulceration and skin (soft tissue) breakage is very common to people with tetraplegia or paraplegia and to most patients with limited movability that spend time in a bed, or in a wheelchair. It is caused by low continued pressure over time and/or high (peak) pressure points. The most commonly used method in a clinical environment, to avoid ulceration in such patients, is for a nurse to “turn” them, in better words; to change the position in which they lay in bed, frequently. This practice is supported by specialist therapeutic support surfaces (mattresses and overlays) to attempt to reduce pressure points. The more modern surfaces are powered air mattresses. There are different types of powered air mattresses, which can alternate pressure in their air cells on a pre-defined timetable.
In terms of market opportunity, hospitals are under tremendous pressure to address decubitus, particularly in the US where payers are moving away from covering “never events”. Pressure ulcers affect a million patients in the USA alone each year and it costs the US healthcare system alone an est. $12 billion in direct treatment costs, not counting associated costs such as medical malpractice lawsuits;
16
Existing market solutions:
|
•
|
Adoption of pressure relieving overlays is increasing; these are generally not ‘active’ and merely made of a suitable material to reduce pressure, resulting in a lower cost; Whilst there is no direct clinical evidence for their effectiveness, there is a widespread belief and acceptance that they can reduce pressure ulcers;
|
|
•
|
Winncare's Axensor system is an example of an inflatable mattress with sections which goes through a standard cycle of in-/deflation to relieve pressure; these are increasingly common for high risk patients and are supplied by all leading therapeutic support surface manufacturers.
|
|
•
|
Israeli start-up Wellsense has the first pressure sensing mattress cover and their clinical evidence shows that the monitoring of pressure alone is already effective; however, it has low adoption because it is not breathable (which could aggravate ulcer formation) and it is costs >$3,000.
|
With the Company’s sensor technology incorporated into mattresses and overlays, for which a prototype exists, technology licensees could develop a range of products with the following: lower production costs, sensors not affecting the properties of the mattresses, ability to measure shear in addition to pressure (shear forces being a recognized key contributor to ulceration).
Furthermore, with the sensor integrated into an inflatable mattress itself, we have developed a smart mattress that can respond intelligently to the pressure and shear data and inflate and deflate the mattress compartments automatically to reduce the risk of ulceration for patients, in between nursing interventions. This dynamic smart mattress adapts to the specific need of every patient and provides relief and adjustment only where and when is needed, maximising the effectiveness of the treatment. This would be classed as a Class 1 medical device, and the license partner would need to pursue such registration prior to launch.
As noted above the Company has entered into an agreement with Carilex Medical Inc. a leading medical mattresses manufacturer where under the Company receives technology access fees and additional fees for each commercialized product under the scope of the agreement when completed. Further there are product based royalty fees payable to the Company on a sliding scale based on production levels for all commercial products sold into the marketplace.
Wheelchairs and medical chairs
The same technology as the Mattress can be used to provide pressure and shear monitoring integrated into wheelchair cushions and medical day-chairs to help prevent ulceration. This may be licensed to the same license partner as the mattress solutions.
Automotive
By incorporating the HCi Viocare sensor technology into automotive seating, professional drivers could be monitored automatically for posture, projected blood circulation, pressure and shear. Posture is important as back pain is a leading cause of absenteeism in the workplace, but in a driving environment, positioning can be an indicator of fatigue, a leading cause of accidents. Adoption of such technology could be driven by health and safety considerations and/or the insurance industry.
The Company is seeking partners in the automotive industry to discuss the potential of incorporating the technology into their vehicles.
|
B.
|
SocketFit
|
The Company will build on the work done by Dr. Kapatos in developing a scanning device to determine the optimal shape of an amputee’s socket, capturing the internal geometry and the biomechanical properties of the tissues in the residual limb. This SocketFit technology was acquired in 2014. The intention is to partner with a leading scanning provider or manufacturer in the P&O market, through a development and license agreement, to produce a socket system for use by prosthetists worldwide.
17
Market opportunity
The number of amputees world-wide is estimated to be 20 million, and for most amputees finding a well-fitted prosthesis is far from easy. Traditional methods of design, manufacture and fitting of a prosthetic socket are typically carried out by “artisan” techniques but unfortunately often result in ill-fitting devices that make wearing a prosthesis almost intolerable for a large number of amputees.
A prosthetic socket is a custom-made “cone” that connects the rest of the prosthesis (foot, shank and knee) to an amputee’s residual limb. Sockets generally need to be replaced every other year, though more frequently in the initial stages after amputation when the residual limb can still change shape significantly. While many significant technological advances have been made with the design and manufacture of prosthetic components, such as electronic knee assemblies and feet, socket design has not kept up. Over the past 20 years a great deal of research has been undertaken to automate the process of socket design and manufacturing, but it has been met with limited success. Most sockets continue to be created with hand-sculpted plaster moulds made by the Prosthetist or a technician hours or days after examining the amputee. The result is that, typically, one in four sockets are discarded because of their poor fit.
Many prosthetists, (if not all), are frustrated by the lack of an objective method that would ensure an optimum fit of the socket. This ‘need’ for assistance instigated the advent of industrial sector technologies to be taken up in the field, even though these systems, in reality, lack the necessary ‘ingredient’ to allow the optimum socket to be produced. They merely attempt to replicate current practices in a digital manner (CAD-CAM systems), without taking into account the biomechanical or anatomical characteristics of the residual limb.
The key element of the project is work done by Dr. Christos Kapatos to improve the nature of the data used in socket modeling software. Finite element analysis (FEA) has been used widely in a variety of applications, including prosthetics. But its prosthetics applications have suffered from the fact that only external boundary data and limited information on the nature of the internal tissue was provided. The results were not promising. By including far more data on the nature of the internal anatomy as well as, for the first time ever (to our knowledge) data on the bio-mechanical properties of the tissue to the FEA, a system can be created that enables Prosthetists to build a socket that evenly distributes weight, provides enhanced comfort, and raises the bar across the industry on socket creation.
SocketFit is a digital system for assessing an amputee’s residual limb and for the production of truly functional and comfortable prosthetic sockets. It takes account of the external and internal geometry of the amputee’s stump, the biomechanical properties of each individual soft tissue layer i.e. skin, fat, muscle and bone, and the boundary and loading conditions of a complete prosthesis to generate a virtual 3D model of the residual limb. It is then possible to produce an accurate, functional and comfortable prosthetic socket.
By minimising the time and cost of socket production and by reducing the number of faulty sockets (it has been reported that a quarter of all prostheses are currently rejected due to poor fit), there will be a reduction in costs incurred by health services and insurance companies worldwide as well as great benefits to the amputee.
The technology
The intended Socket-Fit device consists of three modules:
|
1.
|
The Ring - a device to scan the residual limb and export data, which uses ultrasound technology.
The external geometry of the medium under examination, is acquired by the position data from the stepper instruments and the use of the rotary position sensors; as the ring moves vertically, spinning at the same time, the spring-supported arms comes in contact with the entire residual limb, mapping every detail and transmitting it to a PC.
The internal geometry is acquired with the use of the on-board ultrasound transducers. Information on the mechanical properties of the tissues has also been captured.
|
|
2.
|
Data Tools – software to analyse and collate the data into intelligible information.
All the data are sent to the control box for onward transmission to the PC. Software generates a 3D image of the stump’s external and internal geometry.
|
|
3.
|
Socket Modelling – FEA simulation
FEA and optimisation software generate the optimum design for the socket in order to obtain the best pressure distribution, and therefore the most comfortable prosthesis for the amputee.
|
18
The process to getting to an optimized socket shape is shown in figure 2 and a schematic of the system in figure 3.
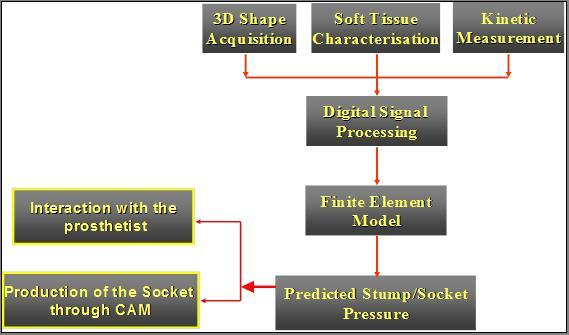
Figure 2: Process map of system

Figure 3: Schematic of the system
19
The greatest value provided by the acquired SocketFit technology rests in the algorithms that transform the data captured by the ultrasound transducers on the Ring to sanitized, usable data about the biomechanical properties of the tissues scanned. Specifically, the algorithms use data derived from the scans to generate stress-strain curves for the tissue layers within the limb.
This data is then used by the FEA software to simulate pressures onto the residual limb, similar to those experienced when walking, for different shapes of sockets, until the ultimate shape is found – meaning, the shape that provides uniform pressure where needed and avoids known pain areas (eg bone) to create a socket designed to have superior comfort in use.
Current Status of SocketFit
We plan to first find a commercialization partner, to help fund and/or conduct the required research and development and to launch the product within two years of commencement of the proposed research and development. This is to minimise risk but also because the technology could be designed to (retro-)fit into the partners’ existing product suite, minimizing effort. While the Company focuses attention on its immediately marketable Sensor Technology and its varying applications, any additional development work for SocketFit is currently on hold.
|
C.
|
Other technology opportunities
|
Blood flow visualization:
The Company has developed a proof of concept device that permits the visualization of peripheral blood flow. This could be incorporated into a smart ‘mat’ for the diagnosis and progression monitoring of diabetic foot conditions. Diabetic patients can suffer from reduced peripheral blood flow in their feet. This contributes to neuropathy but also prevents the healing of diabetic foot ulcers, which can lead to amputation. Such a mat could shift the monitoring of diabetic foot care to primary care, away from podiatry specialists, but also provide essential health information to podiatrists which they would otherwise not have. With a relatively low manufacturing cost, patients could even monitor their own condition at home. It is hoped the product would be prescribed/reimbursed.
Sleep apnea device (BiPAP)
|
•
|
The company has developed an early prototype of a small, silent, portable bidirectional airway device to assist sufferers of sleep apnea
|
Robotic surgical assist
|
•
|
Working (miniature) prototype of robotic device to assist in orthopaedic surgery, reducing number of staff in OR; uses proprietary sensor technology also used in Digital Health products
|
Adaptive socket
|
•
|
The ‘Holy Grail’ of prosthetic sockets, this technology would utilise our proprietary pressure and shear sensors to signal a smart material inside the socket to adapt its shape to the wearer’s gait and daily fluctuations in the size and shape of the stump.
|
Photo acoustic imaging
|
•
|
The team is exploring a theory that would remove the barriers currently preventing the human adoption of photo acoustic imaging, producing a relatively low-cost non-contact, non-invasive methodology. This would be a substantial project but with the potential to transform medical imaging.
|
20
|
D.
|
Intellectual property
|
We have acquired exclusive ownership of the innovative medical device ‘SocketFit’. Dr. Kapatos in conjunction with another development company had prior filed a patent application which has been abandoned. Significant development of the technology has been undertaken since the filing of the abandoned patent and, after corresponding with our patent agents, it was deemed in the best interests of the Company not to revive the old patent, but to apply for new – multiple patents on the technology, and use the old patent as background technology, as new innovations have sprang out of the initial patent and additional fields of application have been identified.
We have acquired all rights and interest in and to the background IPR for a developing technology now known as “Flexisense”. Dr. Kapatos has conceived and been working on various applications of Flexisense including a Smart Insole System which is believed to be a state-of-the-art, pressure and shear pressure sensing insole with an incorporated real-time global display application and a fully featured support web-space that tells the user and his podiatrist/physiotherapist when inappropriate or dangerous conditions are developing on the feet. The product is being designed to mitigate diabetic foot complications, such as ulceration, infection and amputation, as well as improve the total health of a person with diabetes, by incorporating the readings from the insole system to a specialized web-space that will help the user monitor not just the health of his feet but also his diabetes by looking after his diet and exercise. The Company has also identified various other applications. Presently we have filed three patents in the UK in respect of this IPR:
British Patent Application No. 1421952.1, HCI VIOCARE TECHNOLOGIES LTD. (abandoned December 2015)
- The first patent based on development of the “Insole” IP relates to the Company’s pressure sensing system technology, which supports the development of very low cost, flexible and scalable pressure sensing devices in a range of sectors including wearable devices, Prosthetics & Orthotics, medical devices (e.g. for the diabetic foot), and automotive health and safety.
British Patent Application No. 1421950.5, HCI VIOCARE TECHNOLOGIES LTD. (abandoned December 2015)
- The second patent, also based on additional development of the “Insole” IP relates to the Company’s ground-breaking shear sensing system technology, which is supports the development of low cost, flexible and scalable sensor systems in a range of sectors including wearable devices, medical devices (e.g. diabetic foot), Prosthetics & Orthotics and automotive health and safety
British Patent Application No. 1421953.9, HCI VIOCARE TECHNOLOGIES LTD. (abandoned December 2015)
- The third patent application also follows additional in-house development of the “Insole” intellectual property (IP) and relates to pioneering low-cost, portable motion capture and gait analysis technology, which has applications in a range of sectors, including athletic wearable devices, physiotherapy, orthotics and potentially also gaming and film.
International Patent Application No. PCT/GB2015/053785, HCI VIOCARE TECHNOLOGIES LTD.
On December 10, 2015 the Company through its subsidiary HCi Viocare Technologies Ltd. filed an international application according to the Patent Cooperation Treaty (PCT) which received a PCT application number PCT/GB2015/053785. The filed international patent covers over 140 countries around the world. The new patent builds on the company’s three UK patent applications filed in December 2014 (as set out above), with further advances made to the sensor systems, and a wider range of applications in products that touch people’s everyday lives.
The Company has drafted and performed preliminary patent searches on two new innovations during fiscal 2016, with intent to progress into full patent applications in 2017.
21
|
E.
|
Regulatory Requirements
|
The Company’s business model of licensing technology to commercialization partners means that it is the partner, who will be the manufacturer of the eventual products and therefore subjected to the regulations of manufacturing and marketing devices. However, as the Company will support its licensing partner in the development of the final products it has ensured that in understands the eventual requirements, and has identified suitable consultants to provide support, where required.
The products are expected to be marketed worldwide, with an initial commercialization in Europe and the USA, depending on the partner. Their regulatory systems are highlighted below, together with the anticipated implications for the products that may result from our technologies.
Europe
Products that will be marketed in Europe will be required to comply with the EU Directives regarding medical devices, which apply to all EU member states. The basic relevant EU Directive which applies to our case is Directive 93/42/EEC (as amended by 2007/47/EC) regulating medical devices, as well as Directive 98/68/EEC that requires the manufacturers to place CE marking on their products to demonstrate compliance with the above regulations.
The Directive on medical devices also lays down rules in Annex IX for the classification of devices based essentially on the potential risks involved, with class III devices having the highest potential risk and class I device the lowest.
With reference to the stated Rules to determine a product’s classification, the anticipated classification of the products that could arise from the Company’s technology are listed below. It is important to note that classification is determined only at the stage of a final product and with reference to its features and the claims made by the manufacturer. Hence, there classifications are dependent on the decisions of the license partner and may be subject to change:
|
·
|
Diabetic and Medical Insoles: Class I
|
|
·
|
SocketFit: Class IIa – because of the use of ultrasound, which applies energy to the body. This is the usual classification for imaging devices.
|
|
·
|
Photoacoustic imaging: Class IIa – because energy is applied to the body
|
|
·
|
Blood Flow visualization: Class IIs – because energy is applied to the body
|
|
·
|
BiPAP: Class IIb – as it is relied upon for breathing
|
The Notified Body of the country in which the products are marketed will need to notified by the manufacturer, and their specific processes for registration and audit will need to be adhered to.
USA Regulations
Medical devices are subject to the general controls of the Federal Food Drug & Cosmetic (FD&C) Act which are contained in the final procedural regulations in Title 21 Code of Federal Regulations Part 800-1200 (21 CFR Parts 800 - 1299). These controls are the baseline requirements that apply to all medical devices necessary for marketing, proper labeling and monitoring its performance once the device is on the market.
Much like in Europe, the license partner will be required to determine how FDA may classify our devices - which one of the three classes the device may fall into. Unless exempt, FDA will classify our devices. Classification identifies the level of regulatory control that is necessary to assure the safety and effectiveness of a medical device. Most importantly, the classification of the device will identify, unless exempt, the marketing process (either premarket notification [510(k)] or premarket approval (PMA)) the manufacturer must complete in order to obtain FDA clearance/approval for marketing.
The Company will need to review the development of data and/or information necessary to submit a marketing application, and to obtain FDA clearance to market. For some [510(k)] submissions and most PMA applications, clinical performance data is required to obtain clearance to market. In these cases, conduct of the trial must be done in accord with FDA's Investigational Device Exemption (IDE) regulation, in addition to marketing clearance.
22
1.3 OUR CLINICS
|
A.
|
Market and Industry
|
It is estimated that the global health market is growing by 6-8% per annum, which suggests it will double in the next 10 years. The global Orthopedic Prosthetics market seems to be following the same pattern, as it is growing rapidly, and is projected to exceed US$ 4.5 billion by 2017, according to the 2011 Global Data’s report “Orthotics and Prosthetics - Global Pipeline Analysis, Competitive Landscape and Market Forecasts to 2017”.
Demand for orthopedic prosthetics products is driven by a multitude of factors, including:
|
·
|
growing elderly population: The percentage of people aged 65 and older will increase at a rate three times that of younger generations. Subsequently, the frequency of vascular diseases and diabetes – diseases that often lead to amputation – will increase considerably;
|
|
·
|
causes of amputation: the most common causes of amputation are prosperity diseases related to diseases, such as diabetes and vascular diseases. Accidents account for only 26% of all amputations in developed countries. Indicatively, it is estimated that the number of people with diabetes worldwide (382 million as of November 2013) is expected to reach 592 million by 2035. As a result of the general increase in diabetes and vascular diseases, the number of amputations in the western world is also likely to continue to increase;
|
|
·
|
rising incidence of degenerative joint diseases, such as osteoporosis and arthritis, and increasing numbers of sports injuries;
|
|
·
|
social demands: the growing desire of more and more disabled individuals – especially young people – to become independent, lead active lifestyles, and go about their daily routines without outside help;
|
|
·
|
renewal: the lifetime of traditional prosthetic devices used by previous generations is three to five years. Also prosthetic sockets require replacement after three to six months;
|
|
·
|
market niche: reorganization of healthcare services has led to increased pressure to lower costs within the health sector, which in turn increases the demand for the efficiency of the solutions chosen; and
|
|
·
|
the growing demand for orthopedic prosthetics products is also driving the development of innovative medical devices forward. Advancements in biological science and stem cells technology, as well as rapid technological advances in medical science and development of durable and improved materials and implants have led to large number of people requiring physical rehabilitation, thus contributing to the market’s growth.
|
Overall, the market is set to grow significantly as the incidence of amputation increases. The number of amputees living in developing countries is estimated by the World Health Organization to have reached 300 million with up to 95% lacking access to prosthetic devices, thereby creating an increasing consumer base in need for affordable and reliable orthopedic prosthetics products and services that will improve the amputees’ long-term health and well-being.
Current solutions are inadequate, however, as state-of-the-art solutions from the US and Europe are cost-prohibitive, while low-cost devices run the risk of poor quality and/or unreliable performance. Especially in low-income areas and in developing countries, where the P&O industry is highly fragmented and poorly organized, with significant disparities in terms of clinical infrastructure, provision of medical rehabilitation and assistive devices, demographics (such as gender and/or socioeconomic inequalities), and funding.
23
|
B.
|
Clinics:
|
On January 15, 2014, we formed our other wholly-owned Scottish subsidiary, HCi Viocare Clinics UK Limited with registration number SC467486 (“Viocare Clinics”), with the intent to create the first state-of-the-art chain of Prosthetic and Orthotic (P&O) and diabetic foot clinics with global reach, while capitalizing on the aforementioned internally developed technologies. Viocare Clinics will establish and/or acquire movement rehabilitation clinics, limb fitting centers and diabetic foot centers, in the UK, Europe and the Middle East, initially. These centers will offer standardized and specialized rehabilitation and consultation services for the physically impaired, as well as for their families, friends, their workplaces and companies that employ or work with them. Some of the sections that will be covered by the centers are:
|
·
|
Prosthetic rehabilitation;
|
|
·
|
Orthotic rehabilitation;
|
|
·
|
Diabetic foot;
|
|
·
|
Foot congenital disorder and musculoskeletal defects;
|
|
·
|
Movement assisting devices;
|
|
·
|
Gait analysis;
|
|
·
|
Pediatric movement analysis and consultation;
|
|
·
|
Living and working spaces design and consultation for people with disabilities;
|
|
·
|
Walking training and body strengthening following amputation, stroke or accident;
|
|
·
|
Phantomlimb syndrome consultation;
|
|
·
|
Workshops and educational and training programs; and
|
|
·
|
Expert opinion mediation with insurance companies and organizations.
|
24
The primary customers will be the physically impaired:
|
·
|
Amputees;
|
|
·
|
People with spasticity;
|
|
·
|
People with walking difficulties;
|
|
·
|
People with genetic abnormalities;
|
|
·
|
People with deformities;
|
|
·
|
People with musculoskeletal pain;
|
|
·
|
People with diabetes;
|
|
·
|
People with flat foot problems or just pain caused by long working hours or hard labor; and
|
|
·
|
Companies or organizations that employ or work with people with disabilities.
|
However, the market is not limited to these people; our customers will be families with children with disabilities and businesses that employ disabled persons.
Viocare Clinics operates in accordance with the standard mandated by the British Association of Prosthetist Orthotists and the International Society of Prosthetics and Orthotics.
In the next 3-4 years we target to establish a chain of 5 prosthetic, orthotic and diabetic foot rehabilitation centres in Europe and 2 centres in the Middle East through JVs, under the Viocare brand.
|
•
|
Glasgow: Existing Prosthetic practice acquired and being expanded to include Orthotics; serves as the centre of reference and training ground for our other clinics.
|
|
|
•
|
Mediterranean: The Mediterranean is prime location for medical and wellbeing tourism. It has also been used in governmental aid, e.g. Libya’s amputees, which we would seek to re-start. Targeted locations: Athens, Spain, Portugal.
|
25
Glasgow Clinic
|
·
|
The private practice of William Spence, WD Spence Prosthetics Ltd was acquired in June 2014, a profitable venture that was established in 2006, operating part-time with borrowed premises;
|
|
·
|
New premises of 5300 ft2 were secured in Spring 2015, to increase the capacity of the clinic, provide an on-site workshop to improve turnaround times and also to house our R&D staff, thereby facilitating the exchange of ideas;
|
|
·
|
The clinic was refurbished and reopened in September 2015 and is currently being actively marketed, with patient lead coming via:
|
|
-
|
Personal injury solicitors (road traffic and workplace accidents)
|
|
-
|
Word of mouth or internet searches
|
|
-
|
Social media marketing
|
|
-
|
Print advertisement
|
|
·
|
Due to the high quality care of the National Health Service, the market for private P&O provision is small, however, the clinic is the only private P&O clinic in the whole of Scotland;
|
|
·
|
It attracts patients from England also, where the market leader is Dorset Orthopedics (owned by Otto Bock) with a number of full-service and satellite clinics. However, provision in the North continues to be very limited.
|
The Glasgow clinic has recently completed its first full year of operations since relocation to a larger state of the art faculty. As a result the Clinic has experienced substantive year over year revenue growth as we begin to establish a presence in the UK market. The Company anticipated continued growth in the future as the clinic’s level of service and facility benefits become more well known in the UK market.
Competition
Hanger (www.hanger.com) dominates the prosthetic and orthotic clinic market in the USA, with over 1300 clinicians across over 740 clinics. However, in the rest of the world, provision of P&O services is extremely fragmented. Otto Bock, the market leading manufacturer of prosthetic components and vertically integrated to also provide clinical services at their headquarters and through clinics that have been acquired over time, focusing on Northern, Western Europe. Ossur, the Icelandic company which has roughly 20% of the P&O componentry market, has also expanded into clinic in Northern Europe.
The Company’s focus is on areas that are under-served both in terms of capacity and in the quality of service. Competition varies location by location ranging from the very fragmented to the very concentrated.
The Viocare clinics will seek to differentiate through high quality care, staff trained to British and International standards, modern systems and processes and a wide range of products from low- to high end. They will also provide a more holistic suite of services, to include physiotherapy, mental health and lifestyle rehabilitation. Also, our scientific team will lead the technological diversification of the clinics, thereby setting them apart from the competition. We will capitalize on our Scientific Board’s vast network of connections to attract customers from neighboring nations and will seek cooperation with global Medical Tourism operators.
26
|
C.
|
Trademarks
|
National Trademark (Greece)
On March 18, 2015 the Company entered into a Trademark Assignment and Conveyance Agreement (“Trademark Assignment”) with its CEO, President and Director, Mr. Sotirios Leontaritis in respect of the national trademark “viocare” No. 223133 registered for protection in classes 10, 12 and 44 in Greece.
The aforementioned trademark was granted to Mr. Leontaritis on October 11, 2013 and is valid for a period of 10 years from the date of grant. On June 5, 2015, the Trademark Assignment was filed with the Greek Trademarks office for processing. The assignment of the trademark became effective August 4, 2015. t. Mr. Leontaritis received no compensation for the Trademark Assignment.
National Trademark (USA)
On April 8, 2016 the Company filed two applications with the US Trademark Office which received the filing nos. 86969716 and 86969718 for the registration of trademark FLEXI SENSE and FLEXISENSE, respectively, in classes 9, 10, 12 and 25. The registration of said trademarks is pending due to the Office Actions filed on July 26, 2016 against the registration of both the above trademarks in class 9. The Company is in correspondence with the Office Examiner and arguments are underway.
Further to the above, the Company received an opposition letter from the company “Grupo Flexi de Leon S.A.” against the registration of Company’s trademarks FLEXI SENSE and FLEXISENSE in class 25 as well as the registration or any use of mark “FLEXI” by the Company. The Company is in correspondence with Grupo Flexi de Leon S.A. for an amicable settlement.
On May 13, 2014 the Company filed an application with the Office for Harmonization in the Internal Market (OHIM) which received the filing no. 012870754 for the registration of the individual figurative trademark “viocare” for protection in classes 10,12 and 44 within the European Union. The CTM is valid until its expiration/renewal on 13.05.2024.
|
D.
|
Regulations for P&O Clinics
|
The operation of our clinics will be subject to, and must comply with, the respective country’s government regulations. Given that we presently only have one operting clinic, located in Glasgow, Scotland, UK, we are currently most interested in the respective applicable regulations in that jurisdiction. As we determine to establish clinics in other jurisdictions we will be required to understand and be compliant with the laws in those respective jurisdictions in which we intend to operate.
Scotland
|
1.
|
The Health and Social Work Professions Order 2001, made under section 60 of the Health Act 1999, established the UK Health & Care Professions Council (HCPC) that regulates, among others, prosthetists/orthotists in the UK:
|
|
|
·
|
according to the interpretation set out in Schedule 3, “relevant professions means arts therapists; chiropodists; clinical scientists; dietitians; medical laboratory technicians; occupational therapists; orthoptists; paramedics; physiotherapists; prosthetists and orthotists; radiographers; and speech and language therapists”;
|
|
|
·
|
according to article 3, par. 1, the Council is responsible for setting out “the standards of education, training, conduct and performance for members of the relevant professions and to ensure the maintenance of those standards”;
|
|
·
|
according to article 5, “the Council shall establish and maintain a register of members of the relevant professions”;
|
|
·
|
the HCPC sets out for all registrants standards of conduct, performance and ethics, and standards for CPD, as well as standards of proficiency for prosthetists/orthotists specifically
|
|
2.
|
The Health and Care Professions Council (Constitution) Order 2009 which sets out the composition of the Council (as amended by the Health and Care Professions Council (Constitution) Order 2014);
|
|
3.
|
The Regulation of Care (Scotland) Act of 2001 established the Scottish Commission for the Regulation of Care (‘the Care Commission’), and set out the care services that it will regulate, i.e. all adult, child and independent healthcare services in Scotland, one of which independent healthcare services is ‘independent clinics’. As of April 1st 2011, the work of the Care Commission passed to a new body, the Care Inspectorate. Regulation of independent healthcare, including regulation of independent clinics, has passed to Healthcare Improvement Scotland. Healthcare Improvement Scotland is currently responsible for regulating independent hospitals, voluntary hospices, and private psychiatric hospitals. Regulation of independent clinics, independent medical agencies and independent ambulance services has not yet commenced. The Scottish Government consulted on the future arrangements for regulation of independent healthcare services, including the definition and scope of the services that should be regulated last year and are currently considering the way forward.
|
27
1.4 EMPLOYEES
The Company presently has a total of 7 employees and 5 consultants working in its administrative offices in Greece and its UK Clinic. We also retain financial, legal and management consultants as required to meet our financial reporting and SEC reporting compliance requirements on an annual basis. Details of certain of these consulting agreements with respect to our officers, directors and key advisors are provided below.
We have a consulting agreement with our Chief Executive Officer, Sotirios Leontaritis which was entered into on January 15, 2014, whereby Mr. Leontaritis shall provide services to the Company as the Company’s President and Chief Executive Officer in regards to the Company’s management and operations for the period from January 1, 2014 to December 31, 2017. Under the terms of the agreement, the Company agreed to pay to Mr. Leontaritis US$60,000 per annum payable in monthly payments of US$5,000 a month for the term of the contract. Further, under the terms of the contract, Mr. Leontaritis will receive cash compensation of US$5,000 and a stock award of 7,000,000 shares of common stock of the Company in consideration of his services as an officer and director for the period from September 10, 2013 to December 31, 2013. On January 22, 2014, the Company issued the 7,000,000 shares to Mr. Leontaritis pursuant to the agreement. On January 1, 2017 the Company approved a three-year extension to the consulting agreement between the Company and its President & CEO and controlling shareholder, Sotirios Leontaritis, originally entered into on January 1, 2014.
The revision to the Agreement has a term of three years, being effective as of January 1, 2017, and ending on December 31, 2019, renewable for such further term as may be mutually agreed between the parties. As per Addendum No. 1 the Company shall pay to Leontaritis US$120,000 per annum payable in monthly payments of US$10,000 a month for the term of the contract. Further, under the terms of the Addendum No. 1, Leontaritis is entitled to acquire at his discretion 3,000,000 shares of the common stock at a price of $0.30 per share for a term of five (5) years.
On April 16, 2014, the Company entered into a Consulting Agreement with Kapatos whereby he will provide his services as Chief Technical Officer for the Company and the Company’s wholly-owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be forty thousand Euros (€40,000) per year payable in equal monthly installments beginning on May 1, 2014. On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Dr. Christos Kapatos. During fiscal 2016, this agreement was extended for a further one year period.
On November 7, 2014, the Company’s subsidiary, HCi Viocare Clinics entered into a service agreement with Mrs. Heleen Francoise Kist whereby she will provide her services as Chief Operating Officer (“COO”) of the Company. The contract has a term of one year, being effective as of October 1, 2014, and ending on September 30, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation was thirty thousand GBP (£30,000) (USD$44,475) per year payable monthly in arrears on the last Friday of every month by credit transfer. On October 15, 2015, the Company’s COO, Heleen Kist and wholly owned subsidiary, HCI Viocare Clinics UK, signed an Addendum to an employment contract to extend the term to September 30, 2016, with a remuneration increase to forty five thousand GBP (£45,000) (USD$ 66,712) per year. Subsequently, on August 29, 2016 the Company’s subsidiary, HCi Viocare Clinics UK Ltd. approved a termination agreement with Heleen Kist which became effective at the close of her existing contract. Mrs. Kist resigned her positions as an officer and under the terms of the termination agreement the Company has undertaken to repurchase a total of seven hundred thousand (700,000) shares of the Company’s common stock at a purchase price of US$0.02 per share for total of fourteen thousand (US$14,000) USD for return to treasury and cancellation.
On January 1, 2015, the Company entered into a consulting agreement with an individual residing in the Czech Republic, for the provision of Investor Relations Services for the Company. Under the terms of the agreement, Mikulas Dylowicz shall be an independent contractor and shall provide services including responding to shareholder and investor email and telephone inquiries, dissemination of the Company’s public press releases and working with the Company to continue to raise operating capital. Mikulas Dylowicz shall receive monthly consideration of Euros €2,000 per month and shall be reimbursed for all approved out-of-pocket expenses including travel and office expense. During fiscal 2016 the consulting agreement with Mr. Dylowicz was terminated.
On September 1, 2015, the Board of Directors approved a consulting agreement with Sergios Katsaros and appointed Mr. Katsaros Vice President of HCi Viocare.
Under the terms of the consulting agreement, Mr. Katsaros will work directly with the Company’s President and CEO in order to create and implement the Company’s strategic plan and assist in securing additional financing to meet the needs of the Company’s business plan and corporate objectives. The initial term of the contract is six months and Mr. Katsaros will receive compensation of EUR2,000 per month. Further, on March 1, 2016 the Company approved a one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros, originally entered into on September 1, 2015. On March 1, 2017, the Company approved another one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros. The revision to the Agreement ("Addendum No. 2") has a term of one year, being effective as of March 1, 2017, and ending on February 28, 2018. All other terms and conditions remain valid and in force.
On September 1, 2015, the Company appointed Ms. Evaggelia Moschidou as Marketing Director of the Company. Subsequently during fiscal 2016 the contract with Ms. Moshidou was not renewed and the parties agreed to services on a month to month basis.
We presently do not have pension, health, annuity, insurance, stock options, profit sharing or similar benefit plans; however, we may adopt such plans in the future. From time to time the Company grants stock awards or options to purchase shares to its officers, directors, employees and consultants.
1.5 AVAILABLE INFORMATION
Our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and all amendments to those reports that we file with the Securities and Exchange Commission, or SEC, are available at the SEC's public reference room at 100 F Street, N.E., Washington, D.C. 20549. The public may obtain information on the operation of the public reference room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a website at www.sec.gov that contains reports, proxy and information statements and other information regarding reporting companies.
28
An investment in our securities should be considered highly speculative due to various factors, including the nature of our business and the present stage of our development. An investment in our securities should only be undertaken by persons who have sufficient financial resources to afford the total loss of their investment. In addition to the usual risks associated with investment in a business, the following is a general description of significant risk factors which should be considered. You should carefully consider the following material risk factors and all other information contained in this Annual Report before deciding to invest in our common stock. If any of the following risks occur, our business, financial condition and results of operations could be materially and adversely affected. Additional risks and uncertainties we do not presently know or that we currently deem immaterial may also impair our business, financial condition or operating results.
Risks Associated with our Business
Implications of being an emerging growth company
As a company with less than $1 billion in revenue during our last fiscal year, we qualify as an "emerging growth company" as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and we may remain an emerging growth company until December 31, 2018, subject to satisfaction of certain conditions. For so long as we remain an emerging growth company, we are permitted and intend to rely on exemptions from certain disclosure and other requirements that are applicable to other public companies that are not emerging growth companies. In particular, in this annual report, we have provided only two years of audited financial statements and have not included all of the executive compensation related information that would be required if we were not an emerging growth company. Accordingly, the information contained herein may be different than the information you receive from other public companies in which you hold stock.
We have a limited business history and have losses that we expect to continue into the future. If the losses continue we will have to suspend operations or cease functioning.
We were incorporated on March 26, 2007, and have commenced our proposed business operations after a change in business focus in fiscal 2013. To date we have earned minimal revenues, and gross profit of $365,735 in fiscal 2016 which has not been sufficient to cover our operatinal overheads. Our net loss since inception is $31,749,207, While we have existing operations and have commenced revenue generating operations, we do not have revenues sufficient to meet our ongoing overheads and and have no significant assets other than fixed assets and cash on hand. We have yet to generate positive earnings and there can be no assurance that we will ever operate profitably. We have a limited operating history, upon which an evaluation of our future success or failure can be made. Prior to completion of the development stage, we anticipate that we will incur increased operating expenses without realizing any revenues. We therefore expect to incur significant losses into the foreseeable future. If our business plan is not successful and we are not able to operate profitably, then our stock may become worthless and investors may lose all of their investment in our company.
Our ability to achieve and maintain profitability and positive cash flow is dependent upon:
|
•
|
our ability to find profitable markets in which to operate our P&O total rehabilitation clinics and launch products derived from our technologies;
|
|
•
|
our ability to generate revenues; and
|
|
•
|
our ability to manage research, development and costs of operations.
|
While we believe that our business plan has merit, there can be no assurance that we will be able to achieve any of the above and be successful in our future planned operations, and if our losses continue we will have to suspend operations or cease functioning.
Our limited operating history may make it difficult for you to evaluate the success of our business to date and to assess our future viability.
While we were incorporated in 2007 we commenced operations in our chosen field of business focus in fiscal 2013. We have commenced revenue generating operations in our two targered field of operations, however, neither revenue stream is presently sufficient to meet our operational overheads. We will require further capital and additional expansion of our areas of business focus in order to achieve profitable operations. While we are operating a successful P&O clinic in Glasgow, Scotland we have not yet demonstrated our ability to successfully franchise our "P&O Clinics concept" or open additional clinics following our flagship location. In addition, we have not yet successfully commercialized any of our developed technolgies. We are currently working with industry partners towards the presenetation of products based on our technologies to the marker. Consequently, any predictions you make about our future success or viability may not be as accurate as they could be if we had a longer operating history demonstrating successful operations across all our areas of business focus.
In addition, as a new business, we may encounter unforeseen expenses, difficulties, complications, delays and other known and unknown factors. We will need to transition from a company with a research and development focus to a company capable of supporting commercial activities. We may not be successful in such a transition.
We have not yet found commercial partners for our saleable products and may never have saleable products.
We have only recently acquired technologies and developed products ready for commercialization. We have not yet found any commercial partners for our products and many of our products require additional research and development. We will also need to develop a market for our products if our commercial partners do not have an established marketing channel. There can be no assurance that we will successfully find commercial partners, market our products or complete our research and development of new products.
29
If healthcare professionals do not adopt our products in sufficient numbers or as rapidly as we anticipate, our operating results will be harmed.
The success of any of our products which we may develop will depend heavily on acceptance by healthcare professionals who will prescribe and recommend our products, and our failure to maintain a high level of confidence by key healthcare professionals in our products could adversely affect our business.
Consumers may not use our services, and thus we may never become profitable.
Our products represent a significant change from traditional diabetes treatment, and patients may be reluctant to accept them, or may not find them preferable to conventional treatment. In addition, patients may not comply with recommended treatment guidelines which could compromise the effectiveness of their treatment. Our success will depend upon the rapid acceptance of our products by a large number of potential patients to whom we intend to actively market. Market acceptance will depend in part upon the recommendations of prosthetists and orthotists, as well as other factors including effectiveness, safety, reliability, improved treatment aesthetics and greater comfort and hygiene compared to conventional prosthetic-orthotic products. Furthermore, consumers may not respond to our direct marketing campaigns or we may be unsuccessful in reaching our target audience. If consumers prove unwilling to adopt our products as rapidly or in the numbers that we anticipate, our operating results will be harmed.
If the manufacturers of our products encounter problems or delays in the manufacturing process, our ability to generate revenue will be limited.
We intend to rely on third parties for our manufacturing process if we determine to manufacture and market our products directly. Our rapid growth may exceed the capacity of these manufacturers to produce our products in sufficient quantities to support our growth. In the event of delivery delays or shortages of our products, our business and growth prospects may be harmed.
The manufacturers of our products may encounter difficulties in scaling up production to meet demand, including:
|
·
|
problems involving production yields;
|
|
·
|
shortages of key manufacturing equipment;
|
|
·
|
shortages of qualified personnel; and
|
|
·
|
failure to develop new software processes.
|
If demand for our products exceeds manufacturing capacity, we could develop a substantial backlog of customer orders. If our manufacturers are unable to establish and maintain larger-scale manufacturing capabilities, our ability to generate revenue will be limited and our reputation in the marketplace would be damaged.
If we do not enhance our product offerings, either through the acquisition of or investment in new and complementary technologies, products or businesses, or our research and development efforts, we may be unable to compete effectively.
Our success depends in part on our ability to continually enhance and broaden our product offerings in response to changing customer demands, competitive pressures and technologies, and our ability to increase our market share. Accordingly, we expect to consider opportunities to acquire or make investments in other technologies, products and businesses that could enhance our capabilities, complement our current technology, or expand the breadth of our markets or customer base. Acquisitions and strategic investments involve numerous risks, including:
|
·
|
problems assimilating the purchased technologies, products or business operations;
|
|
·
|
issues maintaining uniform standards, procedures, controls and policies;
|
|
·
|
unanticipated costs associated with the acquisition or strategic investment;
|
|
·
|
diversion of management's attention from our core business;
|
|
·
|
adverse effects on existing business relationships with third-party distributers, suppliers and customers;
|
|
·
|
risks associated with entering new markets in which we have limited experience;
|
|
·
|
our ability to obtain regulatory approvals to market new products; and
|
|
·
|
potential loss of key employees of acquired businesses.
|
30
While we are in negotiations for further acquisitions, currently our sole commitment is for the development of our Flexisense and SocketFit technologies. We do not know if we will be able to successfully complete any further acquisitions, or whether we will be able to successfully integrate any acquired business, product or technology into our business or retain any key personnel, suppliers or distributors. Our ability to successfully grow through acquisitions depends upon our ability to identify, negotiate, complete and integrate suitable acquisitions and to obtain any necessary financing. If we are unable to integrate any acquired technology, products or business effectively, our business, financial condition and results of operations could be materially adversely affected.
Furthermore, research and development efforts may require a substantial investment of time and resources before we are adequately able to determine the commercial viability of a new product, technology, material or other innovation. In addition, even if we are able to successfully develop enhancements or new generations of our products, these enhancements or new generations of products may not produce net sales in excess of the costs of development and they may be quickly rendered obsolete by changing customer preferences or the introduction by our competitors of products embodying newer technologies or features.
Issues related to the quality and safety of our products, packaging or sterilization could cause a product recall resulting in harm to our reputation and negatively impact our operating results.
Issues related to quality and safety of products, packaging or sterilization could jeopardize our image and reputation. Negative publicity related to these types of concerns, whether valid or not, might negatively impact demand for our products, or cause production and delivery disruptions. We may need to recall products if they become unfit for use.
Our business exposes us to risks of product liability claims, and we may incur substantial expenses if we are sued for product liability.
Medical devices involve an inherent risk of product liability claims. We may be held liable if any product we develop or any product that uses or incorporates any of our technologies causes injury or is otherwise found unsuitable. Although we intend to continue to maintain product liability insurance, adequate insurance may not be available on acceptable terms and may not provide adequate coverage against potential liabilities. A product liability claim, regardless of its merit or eventual outcome, could result in significant legal defense costs. Costs associated with these potential claims would increase our expenses, and could have a material adverse effect on our business, financial condition and results of operations.
Software defects may be discovered in our products which would damage our ability to sell our products, our results of operations, financial condition and cash flows.
Our systems will incorporate sophisticated computer software. Complex software frequently contains errors, especially when first introduced. Because our products are designed to be used to perform complex interventional procedures, we expect that physicians and hospitals will have an increased sensitivity to the potential for software and other defects. We cannot assure you that our software will not experience errors or performance problems in the future. If we experience software errors or performance problems, we would likely also experience:
|
•
|
loss of revenue;
|
|
|
•
|
increase in reportable adverse events to applicable authorities;
|
|
|
•
|
delay in market acceptance of our products;
|
|
|
•
|
damage to our reputation;
|
|
|
•
|
additional regulatory filings;
|
|
|
•
|
product recalls;
|
|
|
•
|
increased service or warranty costs; and/or
|
|
|
•
|
product liability claims relating to the software defects.
|
31
We may fail to receive positive clinical results for our products in development that require clinical trials, and even if we receive positive clinical results, we may still fail to receive the necessary clearance or approvals to market our products.
In the development of new products or new indications for, or modifications to, existing products, we may conduct or sponsor clinical trials. Clinical trials are expensive and require significant investment of time and resources. Clinical trials are subject to regulations for clinical studies in each respective country and, failure to comply with such regulations, including, but not limited to, failure to obtain adequate consent of subjects, failure to adequately disclose financial conflicts, or failure to report data or adverse events accurately, could result in fines, penalties, and/or suspension of trials.
Our business may be harmed by technological and therapeutic changes, or the demand for our services could be diminished by the development of alternative treatments. Future innovations could make our inventions obsolete.
Our success will depend, in part, on continued demand for products that will incorporate our inventions. Changes in technology or customer requirements could render these inventions obsolete or unmarketable.
We will rely on information technology systems for accounting and finance, inventory management, engineering, distribution and other functions, and to maintain our research and development data. If such adopted information technology systems fail to adequately perform these functions, or if we experience an interruption in their operation, our business, financial condition and results of operations could be adversely affected.
The efficient operation of our business will be dependent on our information technology systems which we intend to develop. We will rely on our information technology systems to effectively manage:
|
·
|
sales and marketing, accounting and financial functions;
|
|
·
|
order entry, order fulfillment and inventory replenishment processes;
|
|
·
|
engineering tasks; and
|
|
·
|
our research and development data.
|
The failure of our intended information technology systems to perform as we anticipate, or our failure to effectively implement new systems, could disrupt our business and product development and could result in decreased sales, increased overhead costs, excess inventory and product shortages, all of which could have a material adverse effect on our business, financial condition and results of operations. In addition, our information technology systems are vulnerable to damage or interruption from:
|
·
|
tornado, earthquake, fire, flood and other natural disasters;
|
|
·
|
terrorist attacks and attacks by computer viruses or hackers;
|
|
·
|
power loss; and
|
|
·
|
network failure of computer systems, Internet, telecommunications or data.
|
Any such interruption could have material adverse effect on our business, financial condition and results of operations.
Our success depends in part on our proprietary technology and if we are unable to successfully enforce our intellectual property rights, our competitive position may be harmed.
Our success will depend in part on our ability to maintain existing intellectual property and to obtain and maintain further intellectual property protection for our products, in the U.K., Europe, the U.S.A., and in other countries. Our inability to do so could harm our competitive position. We do not currently have any granted patents related to our technology and our current technology could be at risk if we successfully obtain patent protection. While we have applied for patents for various products based on our IPR, there is no assurance we will be successful in obtaining these patents. We will rely on our IP portfolio of future patent applications to protect a large part of our intellectual property and our competitive position. However, our future patent filings may not issue as patents. Additionally, any patents issued to us may be challenged, invalidated, held unenforceable, circumvented, or may not be sufficiently broad to prevent third parties from producing competing products similar in design to our products. In addition, protection afforded by foreign patents may be more limited than that provided under U.S. patents and intellectual property laws.
32
We rely on confidentiality agreements that could be breached and may be difficult to enforce which could have a material adverse effect on our business and competitive position.
Our policy is to enter into agreements relating to the non-disclosure of confidential information with third parties, including our contractors, consultants, advisors and research collaborators, as well as into agreements that purport to require the disclosure and assignment to us of the rights to the ideas, developments, discoveries and inventions of our employees and consultants while we employ them. However, these agreements can be difficult and costly to enforce. Moreover, to the extent that our contractors, consultants, advisors and research collaborators apply or independently develop intellectual property in connection with any of our projects, disputes may arise as to the proprietary rights to this type of information. If a dispute arises, a court may determine that the right belongs to a third party, and enforcement of our rights can be costly and unpredictable. In addition, we rely on trade secrets and proprietary know-how that we will seek to protect in part by confidentiality agreements with our employees, contractors, consultants, advisors or others. Despite the protective measures we employ, we still face the risk that:
|
•
|
these agreements may be breached;
|
|
|
•
|
these agreements may not provide adequate remedies for the applicable type of breach; or
|
|
|
•
|
our trade secrets or proprietary know-how will otherwise become known.
|
Any breach of our confidentiality agreements or our failure to effectively enforce such agreements would have a material adverse effect on our business and competitive position.
The medical device industry is characterized by extensive litigation over patents and other intellectual property rights and we could become subject to litigation that could be costly, result in the diversion of management's time and efforts, require us to pay damages, and/or prevent us from marketing our existing or future products.
We may be the subject of patent or other litigation in the future. An adverse determination in a patent suit in any litigation or interference proceeding to which we may become a party could subject us to significant liabilities. An adverse determination of this nature could also put our patents, when granted, at risk of being invalidated or interpreted narrowly or require us to seek licenses from third parties. Licenses may not be available on commercially reasonable terms or at all, in which event our business would be materially adversely affected. We may also receive letters from third parties drawing our attention to their patent rights. While we do not believe that we infringe any valid and enforceable rights which have been brought to our attention, there may be other more pertinent rights of which we are presently unaware. If we infringe the patents or proprietary rights of other parties, our ability to grow our business will be severely limited. The defense and prosecution of intellectual property suits, interference proceedings and related legal and administrative proceedings could result in substantial expense to us and significant diversion of effort by our technical and management personnel.
Extensive and changing government regulation of the healthcare industry may be expensive to comply with and exposes us to the risk of substantial government penalties.
We face risks related to our international operations, including the need to obtain necessary foreign regulatory clearance or approvals. If we fail to obtain regulatory clearances in countries in which we intend to operate for products under development, we will not be able to commercialize these products in those countries. Sales of our products will be subject to regulatory requirements that vary widely from country to country. We may be unable to obtain regulatory approvals in the countries in which we intend to operate or to market our products. We may also incur significant costs in attempting to obtain and in maintaining regulatory approvals. If we experience delays in receipt of approvals to market our products or if we fail to receive these approvals, we may be unable to market our products or enhancements in our intended markets in a timely manner, if at all.
|
·
|
Noncompliance with applicable regulatory requirements can result in enforcement action which may include recalling products, ceasing product marketing, and paying significant fines and penalties, which could limit product sales, delay product shipment and adversely affect our profitability.
|
Our results of operations will suffer if we are unable to manage our operations effectively.
The sale and shipment of our products across international borders will subject us to extensive foreign governmental trade, import and export, and custom regulations and laws. Compliance with these regulations is costly and exposes us to penalties for non-compliance. Other laws and regulations that can significantly impact us include various anti-bribery laws and anti-boycott laws. Any failure to comply with applicable legal and regulatory obligations could impact us in a variety of ways that include, but are not limited to, significant criminal, civil and administrative penalties, including imprisonment of individuals, fines and penalties, denial of export privileges, seizure of shipments, restrictions on certain business activities and exclusion or debarment from government contracting. Also, the failure to comply with applicable legal and regulatory obligations could result in the disruption of our distribution and sales activities. In addition, some of the countries in which we may sell our products may be subject to political, economic or social instability. Our intended international operations expose us and our distributors to risks inherent in operating in foreign jurisdictions. These risks include:
|
·
|
adverse changes in tariffs and trade restrictions;
|
|
·
|
political, social and economic instability and increased security concerns;
|
|
·
|
longer collection periods and difficulties in collecting receivables from foreign entities;
|
|
·
|
exposure to different legal standards;
|
33
|
·
|
transportation delays and difficulties of managing international distribution channels;
|
|
·
|
lack of stringent protection of intellectual property;
|
|
·
|
potentially adverse tax consequences and the complexities of foreign value-added tax systems;
|
|
·
|
difficulties in staffing and managing foreign operations;
|
|
·
|
obstacles to obtaining domestic and foreign export, import and other governmental approvals, permits and licenses and compliance with foreign laws;
|
|
·
|
limitations on the repatriation of earnings; and
|
|
·
|
increased financing costs.
|
These risks may limit or disrupt our operations, restrict the movement of funds or result in the deprivation of contractual rights or the taking of property by nationalization or expropriation without fair compensation. Our continued success as a global company depends, in part, on our ability to develop and implement policies and strategies that are effective in anticipating and managing these and other risks in the countries in which we do business. Failure to manage these and other risks may have a material adverse effect on our operations in any particular country and on our business as a whole.
If we cannot successfully implement our business strategy, our business, results of operations and potential for growth will be adversely affected.
Our business strategy includes the development and marketing of our products and the obtaining of regulatory approval to sell our products in certain key markets and the acquisition or direct development of P&O clinics in various jurisdictions. Our ability to achieve our business strategy is subject to a variety of factors, many of which are beyond our control. In addition, the implementation of our business strategy may not improve our operating results. We may decide to alter or discontinue aspects of our business strategy and may adopt alternative or additional strategies due to business or competitive factors not currently foreseen, such as the introduction of new products or technologies by our competitors or the lack of clients for our clinics. Any failure to successfully implement our business strategy would have a material adverse effect on our business, financial condition and results of operations.
If we lose our key personnel or are unable to attract and retain key personnel, we may be unable to pursue business opportunities or develop our products.
We are highly dependent upon the experience and ability of certain key employees in our management and clinical bioengineering teams, including Sotirios Leontaritis, Director, President, Treasurer, Chief Financial Officer and Chief Executive Officer; Nikolaos Kardaras, Director and Secretary, and Dr. Christos Kapatos, Director and CTO, who heads the development of our technology. The loss of the services of any one of those individuals could have an adverse effect on our business, and may significantly delay or prevent the achievement of our product development and other business objectives. Our future success will also depend on our ability to identify, recruit, train and retain additional qualified personnel. There is currently a shortage of skilled clinical, bioengineering and management personnel and intense competition for these personnel. Thus, we may not be successful in retaining our key personnel or their services.
34
Risks Associated with our Common Stock
We are an "emerging growth company," and the reduced disclosure requirements applicable to emerging growth companies may make our common stock less attractive to investors.
We are an "emerging growth company," as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, and may remain an emerging growth company until December 31, 2018, provided that, if the market value of our common stock that is held by non-affiliates exceeds $700 million as of any June 30 before that time or if we have annual gross revenues of $1 billion or more in any fiscal year, we would cease to be an emerging growth company as of December 31 of the applicable year. We also would cease to be an emerging growth company if we issue more than $1 billion of non-convertible debt over a three-year period. For so long as we remain an emerging growth company, we are permitted and intend to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include:
|
·
|
providing only three years of audited financial statements, in addition to any required unaudited interim financial statements, with correspondingly reduced "Management's discussion and analysis of financial condition and results of operations" disclosure;
|
|
·
|
not being required to comply with the auditor attestation requirements in the assessment of our internal control over financial reporting;
|
|
·
|
not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor's report providing additional information about the audit and the financial statements;
|
|
·
|
reduced disclosure obligations regarding executive compensation; and
|
|
·
|
exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved.
|
We may choose to take advantage of some, but not all, of the available exemptions. We have taken advantage of reduced reporting burdens in this filing. In particular, in this filing, we have provided only two years of audited financial statements and have not included all of the executive compensation related information that would be required if we were not an emerging growth company. We cannot predict whether investors will find our common stock less attractive if we rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
In addition, the JOBS Act also provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to delay such adoption of new or revised accounting standards, and as a result, we may not comply with new or revised accounting standards on the relevant dates, on which adoption of such standards is required for public companies that are not emerging growth companies. As a result of such election, our financial statements may not be comparable to the financial statements of other public companies.
We do not intend to pay dividends on any investment in the shares of our stock.
We have never paid any cash dividends and currently do not intend to pay any dividends for the foreseeable future. To the extent that we require additional funding currently not provided for in our financing plan, our funding sources may prohibit the payment of a dividend. Because we do not intend to declare dividends, any gain on an investment in our company will need to come through an increase in the stock’s price. This may never happen and investors may lose all of their investment in us.
Because we can issue additional shares of common stock, purchasers of our common stock may incur immediate dilution and may experience further dilution.
We are authorized to issue up to 700,000,000 shares of common stock, of which 195,064,844shares are issued and outstanding. Our board of directors has the authority to cause us to issue additional shares of common stock, and to determine the rights, preferences and privileges of such shares, without consent of any of our stockholders. Consequently, the stockholders may experience more dilution in their ownership of our stock in the future.
Because we can issue shares of preferred stock which will have certain voting rights, which, if voted collectively, could result in a control of any matters put before the Company’s stockholders, the shareholders of our common stock will suffer substantial dilution if issued or converted.
We have the right to issue a total of 5,000,000 shares of Series A Preferred Stock. Except with respect to matters which adversely affect the holders of Series A Preferred Stock, as required by law, or as required by the Articles of Incorporation, the holders of Series A Preferred and the holders of Common Stock shall be entitled to notice of any stockholders' meeting and to vote as a single class upon any matter submitted to the stockholders for a vote, on the following basis:
|
(a)
|
holders of Common Stock shall have one vote per share of Common Stock held by them; and
|
|
(b)
|
holders of Series A Preferred Stock shall have 50 votes per share of Series A Preferred Stock held by them.
|
35
Holders of the Series A Preferred Stock have voting rights with respect to matters that generally require the approval of voting stockholders, in addition to voting rights as specifically required by Nevada law. In the event the holders of Series A Preferred Stock vote collectively as to any matter put before a vote, such holders of Series A Preferred Stock could control the vote. While the holders of Series A Preferred Stock have not advised that they intend to vote collectively on any matter, nor are we aware of any voting agreements currently in place, we cannot know with certainty that such holders of Series A Preferred Stock will not vote collectively in the future or enter into any voting agreements.
Further, the holders of any Series A Preferred Stock will have the right to convert to common stock at their election on the basis of 20 shares of common stock for each one share of Series A Preferred Stock issued, thus the holders of our common stock would suffer substantial dilution upon conversion.
A decline in the price of our common stock could affect our ability to raise further working capital, it may adversely impact our ability to continue operations, and we may go out of business.
A prolonged decline in the price of our common stock could result in a reduction in the liquidity of our common stock and a reduction in our ability to raise capital. Because we may attempt to acquire a significant portion of the funds we need in order to conduct our planned operations through the sale of equity securities, a decline in the price of our common stock could be detrimental to our liquidity and our operations because the decline may cause investors to not choose to invest in our stock. If we are unable to raise the funds we require for all our planned operations, we may be forced to reallocate funds from other planned uses and may suffer a significant negative effect on our business plan and operations, including our ability to develop new products and continue our current operations. As a result, our business may suffer, and not be successful and we may go out of business. We also might not be able to meet our financial obligations if we cannot raise enough funds through the sale of our common stock and we may be forced to go out of business.
Our stock is a penny stock. Trading of our stock may be restricted by the Securities and Exchange Commission’s penny stock regulations which may limit a stockholder’s ability to buy and sell our stock.
Our stock is a penny stock. The Securities and Exchange Commission has adopted Rule 15g-9 which generally defines “penny stock” to be any equity security that has a market price (as defined) less than $5.00 per share or an exercise price of less than $5.00 per share, subject to certain exceptions. Our securities are covered by the penny stock rules, which impose additional sales practice requirements on broker-dealers who sell to persons other than established customers and “accredited investors”. The term “accredited investor” refers generally to institutions with assets in excess of $5,000,000 or individuals with a net worth in excess of $1,000,000 or annual income exceeding $200,000 or $300,000 jointly with their spouse. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document in a form prepared by the SEC which provides information about penny stocks and the nature and level of risks in the penny stock market. The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction and monthly account statements showing the market value of each penny stock held in the customer’s account. The bid and offer quotations, and the broker-dealer and salesperson compensation information, must be given to the customer orally or in writing prior to effecting the transaction and must be given to the customer in writing before or with the customer’s confirmation. In addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from these rules, the broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written agreement to the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for the stock that is subject to these penny stock rules. Consequently, these penny stock rules may affect the ability of broker-dealers to trade our securities. We believe that the penny stock rules discourage investor interest in and limit the marketability of our common stock.
FINRA sales practice requirements may also limit a stockholder's ability to buy and sell our stock.
In addition to the “penny stock” rules promulgated by the Securities and Exchange Commission, the Financial Industry Regulatory Authority (FINRA) has adopted rules that require that in recommending an investment to a customer, a broker-dealer must have reasonable grounds for believing that the investment is suitable for that customer. Prior to recommending speculative low priced securities to their non-institutional customers, broker-dealers must make reasonable efforts to obtain information about the customer’s financial status, tax status, investment objectives and other information. Under interpretations of these rules, the FINRA believes that there is a high probability that speculative low priced securities will not be suitable for at least some customers. The FINRA requirements make it more difficult for broker-dealers to recommend that their customers buy our common stock, which may limit your ability to buy and sell our stock.
Technology company stock prices are especially volatile, and this volatility may depress the price of our common stock.
The stock market has experienced significant price and volume fluctuations, and the market prices of technology companies have been highly volatile. We believe that various factors may cause the market price of our common stock to fluctuate, perhaps substantially, including, among others, the following:
|
•
|
announcements of developments in our patent enforcement actions, should patents be granted to us;
|
|
•
|
developments or disputes concerning our inventions or patents, if granted;
|
|
•
|
our competitors' technological innovations;
|
|
•
|
developments in relationships with licensees;
|
36
|
•
|
variations in our quarterly operating results;
|
|
•
|
our failure to meet or exceed securities analysts' expectations of our financial results;
|
|
•
|
a change in financial estimates or securities analysts' recommendations;
|
|
•
|
changes in management's or securities analysts' estimates of our financial performance;
|
|
•
|
changes in market valuations of similar companies;
|
|
•
|
the current sovereign debt crises affecting several countries in the European Union and concerns about sovereign debt of the United States;
|
|
•
|
announcements by us or our competitors of significant contracts, acquisitions, strategic partnerships, joint ventures, capital commitments, new technologies, or patents, if granted; and
|
|
•
|
failure to complete significant transactions.
|
The financial crisis affecting the banking system and financial markets and the uncertainty in global economic conditions, which began in late 2007 and has continued up until today, have resulted in a tightening in the credit markets, a low level of liquidity in many financial markets, and extreme volatility in the credit, equity and fixed income markets. If investors have concerns that our business, operating results and financial condition will be negatively impacted by global economic conditions, our stock price could fluctuate significantly in future periods.
In addition, we believe that fluctuations in our stock price during applicable periods can also be impacted by court rulings and/or other developments in our patent licensing and enforcement actions. Court rulings in patent enforcement actions are often difficult to understand, even when favorable or neutral to the value of the patents we intend to apply for and our overall business, and we believe that investors in the market may overreact, causing fluctuations in our stock prices that may not accurately reflect the impact of court rulings on our business operations and assets.
In the past, companies that have experienced volatility in the market price of their stock have been the objects of securities class action litigation. If our common stock was the object of securities class action litigation, it could result in substantial costs and a diversion of management's attention and resources, which could materially harm our business and financial results.
Risks Related to our Financial Results and Need for Additional Financing
We may be unable to raise additional capital if it should be necessary, which could harm our ability to compete.
We expect to incur significant expenditures in order to establish an international brand, to develop both product and process technology, and to operate P&O clinics around the world. We do not currently have the funds available to effectuate our business plan. We have to date relied on loans from our chief executive officer and certain private placements of our common stock to fund our ongoing operations, as well as revenues generated from our Glasgow Clinic and initial technology access fees, however, we cannot be assured that these opportunities will provide funding for our ongoing operations and we may require substantial funding that may not be available when required. We will be required to seek substantial funding in the equity markets or by loans for operations. We may not be able to raise funds when needed, or on acceptable terms.
We may not generate sufficient cash flow to service our debt, pay our contractual obligations and operate our business.
Our ability to make payments on our indebtedness and contractual obligations, and to fund our operations depends on our future performance and financial results, which, to a certain extent, are subject to general economic, financial, competitive, regulatory and other factors, including the interest rate environment, that are beyond our control. Until such time as we can establish operations to generate sufficient liquidity to meet our obligations as they come due you are at risk of our business failing and the loss of your investment.
There can be no assurance that our business will generate sufficient cash flow, if any, from operations in the future to service our debt, pay our contractual obligations and operate our business as currently planned.
37
Our financial results may vary significantly from quarter-to-quarter due to a number of factors, which may lead to volatility in our stock price.
Our quarterly revenue and results of operations may vary significantly from quarter-to-quarter if, and when, we (i) expand our chain of P&O Clinics to increase our revenue in this segment, and (ii) successfully commercialize any one of our technologies so that we are actively receiving both licensing fees and royalties from the ongoing sales of our products. While we are generating revenue at our flagship P&O clinic and we are earning revenue from licensing fees for access to our technology by industry partners, we are still working on expanding our revenue base so that we can cover operations and so that we can make accurate preductions about our annual operting costs and funding requirements. This variability may lead to volatility in our stock price as research analysts and investors respond to these quarterly fluctuations. These fluctuations are due to numerous factors, including: fluctuations in consumer demand for our products; our ability to design and deliver products to our consumers in a timely and cost-effective manner; quality control problems in the manufacturing operations; our ability to timely obtain adequate quantities of the components used in our products; new product introductions and enhancements by us and our competitors; unanticipated increases in costs or expenses; and fluctuations in foreign currency exchange rates. The foregoing factors are difficult to forecast, and these, as well as other factors, could materially and adversely affect our quarterly and annual results of operations.
Prolonged negative economic conditions in domestic and global markets may adversely affect our suppliers and consumers, which could harm our financial position.
Global credit and financial markets have experienced extreme disruptions over the past few years, including severely diminished liquidity and credit availability, declines in consumer confidence, declines in economic growth, increases in unemployment rates, and uncertainty about economic stability. Although these markets have recently shown signs of rebounding, there can be no assurance that further deterioration in credit and financial markets and confidence in economic conditions will not occur. Our general business strategy may be adversely affected by the recent economic downturn, volatile business environments and continued unpredictable and unstable market conditions. If the current equity and credit markets deteriorate further, or do not improve, it may make debt or equity financings, if needed, more difficult, more costly and more dilutive. Failure to secure financing, if needed, in a timely manner, on favorable terms or at all could have a material adverse effect on our growth strategy, financial performance and stock price, and could require us to delay or abandon certain aspects of our business strategy. These negative changes in domestic and global economic conditions or additional disruptions of either or both of the financial and credit markets may have a material adverse effect on our business, financial condition and results of operations.
As a “smaller reporting company”, we are not required to provide the information required by this Item.
The Company operates from a branch office at 2A Kolokotroni Street, 17563 Paleo Faliro, Greece. The address of our current corporate office in Glasgow is Kintyre House, 209 Govan Road, Glasgow Scotland G51 1HJ United Kingdom. Our telephone number is +44 141 370 0321.
Executive Office in Greece
On February 3, 2014, the Company leased office space in Paleo Faliro, Greece on a three-year lease, commencing on March 1, 2014 and ending on February 28, 2017. The monthly rental fee is $1,700 (EUR € 1,554) including applicable taxes in the first year. Thereafter, for every further lease year and for the whole duration of the lease agreement, as well as in case of compulsory statutory extension or extension by tacit agreement, the monthly rent shall be annually adjusted by a percentage equal to the Consumer Price Index of the adjustment month with respect to the respective month of the year before (simple annual rate of change), as this is calculated by the Hellenic Statistical Authority (ELSTAT), plus two (2) percentage points (+2%). The Company and the lessor are currently negotiating on terms for a renewal of the lease for fiscal 2017.
Under the term of the lease agreement, the Company paid a total of $25,010 (EUR €18, 540) including $4,047 (EUR €3,000) for deposit and $20,963 (EUR €15,540) for ten months’ rental fee upon executing the agreement. As of December 31, 2016, $3,161 (EUR €3,000) is shown as deposit in the prepaid account.
Proposed Clinic in Greece
On December 29, 2014, the Company leased office space at Peania Region of Attica in Greece on a ten-year lease, commencing on January 1, 2015 and ending on October 5, 2024. The monthly rental fee is $6,996 (EUR €6,233) plus applicable taxes and the monthly operating cost estimate is $1,120 (EUR €997).
Under the term of the lease agreement, the Company paid a total of $13,991 (EUR €12,465) as deposit in the prepaid account.
On October 9, 2015, the terms of the lease were amended to include the following provisions:
|
-
|
Effective September 1, 2015, the monthly rental fee shall be reduced to $3,500 (EUR €3,116) for a period of 12 months ending August 31, 2016.
|
|
-
|
In the event that the Lessee commences leasehold improvements prior to August 31, 2016, it shall inform the Lessor respectively and pay retroactively the remaining balance of the total amount of monthly rental in full from the period commencing September 1, 2015, as if they have not been reduced;
|
|
-
|
In the event that the Company does not proceed with leasehold improvements in the 12-month period, the lease agreement will terminate and the Lessor with retain the deposit.
|
|
-
|
The Lessor until such time as the leasehold improvements commence, may at any time and without indemnity terminate the lease agreement with a 30-day prior written notice to the Lessee.
|
As of December 31, 2016, the Company and the Lessor concluded the Termination of Lease Agreement dated August 1, 2016.
38
Glasgow, Scotland P&O Clinic and research facility
On March 2, 2015, the Company entered into a lease agreement for a modern, stand-alone 5,300 square foot facility in Glasgow, Scotland with an entry date of 1 March, 2015. The building will host the Company’s first Viocare center, a full service Prosthetic and Orthotic (“P&O”) practice with a superior standard of personalized care. The renovations to the clinic were completed in June, and the facility was opened on August 1, 2015 with an official ribbon cutting on 25 September 2015. During the first year from the date of entry, the lease fees are USD$26,000 (GBP £20,000) per annum (exclusive of VAT). On the second anniversary of the date of entry, the lease will increase to USD$52,000 (GBP £40,000) per annum (exclusive of VAT). In the third anniversary of the date of entry, the lease will be USD$52,000 (GBP £40,000) per annum (exclusive of VAT). In the fourth anniversary of the date of entry, the lease will decrease to USD$39,000(GBP £30,000) per annum (exclusive of VAT). In the fifth anniversary of the date of entry, the lease will be USD$52,000 (GBP £40,000) per annum (exclusive of VAT).
Under the term of the lease agreement, the Company paid a total of $28,738 (GBP £20,000) as a deposit.
Viocare, Inc. a New Jersey corporation (the «Plaintiff») filed a complaint on October 24, 2016 in the United States District Court, District of Nevada objecting to the use of the name «viocare» by HCi Viocare, of Nevada (the «Defendant»). The Company and Viocare Inc. have agreed to terms of settlement, the details of which shall be published once fully executed by both parties and filed with the courts.
We know of no other material, active or pending legal proceedings against our Company, nor of any proceedings that a governmental authority is contemplating against us.
Not Applicable
39
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Price of and Dividends on the Registrant’s Common Equity and Related Stockholder Matters
Market Information
Our common stock is quoted on the OTC Markets Inc. owned and operated Inter-dealer Quotation System (“OTCQB”) under the symbol "VICA". Information provided below represents the two years ended December 31, 2016, and the impact of a 7 for 1 forward split effective August 7, 2015:
|
Quarter
|
High ($)
|
Low ($)
|
||||||
|
4th Quarter ended 12/31/2016
|
1.53
|
0.40
|
||||||
|
3rd Quarter ended 9/31/2016
|
1.10
|
.832
|
||||||
|
2nd Quarter ended 6/31/2016
|
0.85
|
0.61
|
||||||
|
1st Quarter ended 3/31/2016
|
0.86
|
0.54
|
||||||
|
4th Quarter ended 12/31/2015
|
0.7751
|
0.56
|
||||||
|
8/7/2015 through 9/30/2015
|
0.67
|
0.055
|
||||||
|
Forward Split 8/7/2015 on basis of 7 for 1
|
||||||||
|
7/1/2015 through 8/6/2015 (presplit)
|
4.80
|
3.50
|
||||||
|
2nd Quarter ended 6/30/2015 (presplit)
|
4.85
|
3.51
|
||||||
|
1st Quarter ended 3/31/2015 (presplit)
|
4.00
|
1.90
|
||||||
The above information was taken from information on the OTC Markets website at www.otcmarkets.com. The quotations provided may reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not represent actual transactions. The pre-split pricing is not adjusted for the split which occurred on August 7, 2015.
Our common shares are issued in registered form. Our registrar and transfer agent is Action Stock Transfer Corporation, 2469 E. Fort Union Blvd, Suite 214, Salt Lake City, UT 84121. Telephone: 801-274-1088
Holders
As of April 14, 2017, we had outstanding 195,064,844shares of common stock, which were held by 63 record holders of the Company’s common stock (which number does not include the number of stockholders whose shares are held by a brokerage house or clearing agency, but does include such brokerage houses or clearing agencies as one record holder.
40
Dividends
We have never declared or paid dividends on our common stock. We intend to retain earnings, if any, to support the development of our business and therefore do not anticipate paying cash dividends for the foreseeable future. Payment of future dividends, if any, will be at the discretion of our board of directors after taking into account various factors, including current financial condition, operating results and current and anticipated cash needs.
Repurchase of Equity Securities
On August 30, 2016, the Board of Directors of the Company approved the repurchase from Mrs Heleen Kist of a total of seven hundred thousand (700,000) shares of the Company at a purchase price of US$0.02 per share for total of fourteen thousand (US$14,000) USD for return to treasury and cancellation. The $14,000 was recorded as accounts payable on the balance sheet as of December 31, 2016.
Recent Sales of Unregistered Securities; Use of Proceeds from Registered Securities
Recent Sales of Unregistered Securities
During the three months ended December 31, 2016 and the subsequent period to the date of the filing of this annual report, the Company issued the following shares:
On October 11, 2016, the Company entered into Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 40,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$20,000. The shares are subject to applicable resale restrictions
On October 24, 2016, the Company granted a total of 25,000 stock awards to various employees of its wholly owned UK subsidiaries with a market value of $1 per share. The shares are subject to applicable resale restrictions
On November 4, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 85,000 shares of the Company’s common stock at a purchase price of US$0.60 per share for total cash proceeds of US$51,000. The shares are subject to applicable resale restrictions
On November 18, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 66,300 shares of the Company’s common stock at a purchase price of US$0.80 per share for total cash proceeds of US$53,040. The shares are subject to applicable resale restrictions
On December 7, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 21440 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$10,720. The shares are subject to applicable resale restrictions
On January 1, 2017, the Company approved a three-year extension to the consulting agreement between the Company and its President & CEO and controlling shareholder, Sotirios Leontaritis, originally entered into on January 1, 2014.
Under the terms of the revised agreement, Mr. Leontaritis is entitled to acquire at his discretion 3,000,000 shares of the common stock at a price of $0.30 per share for a term of five (5) years.
On March 3, 2017, the Company approved the issuance of 1,000,000 common shares for the services provided by Mr. Nikolaos Kardaras, Director of the Company, in the form of stock award which shall vest as of the date of grant. The shares were valued at the fair market value of $0.172 on grant date. Concurrrently the final 100,000 shares issuable under a stock award originally granted on March 1, 2016 to Mr. Sergios Katsaros, Vice President, had vested in full and was issued.
On March 6, 2017, the Company approved the issuance of 50,000 common shares for the services provided by Mr. Ioannis Kardaras, consultant, in the form of stock award which shall vest as of the date of grant. The shares were valued at the fair market value of $0.172 on grant date.
On March 7, 2017, the Company approved the issuance of 50,000 common shares for the services provided by Mr. Apostolos Digbassanis, consultant, in the form of stock award which shall vest as of the date of grant. The shares were valued at the fair market value of $0.172 on grant date.
On March 17, 2017, the Company approved the issuance of 1,000,000 common shares for the services provided by Mr. Sergios Katsaros, Vice-president, officer of the Company, in the form of stock award which shall vest as of the date of grant. The shares were valued at the fair market value of $0.17 on grant date.
41
On March 22, 2017, the Company approved the issuance of 50,000 common shares for the services provided by Mr. Brian Maguire, Director of HCi Viocare Clinics UK Ltd., subsidiary of the Company, in the form of stock award which shall vest as of the date of grant. The shares were valued at the fair market value of $0.25 on grant date.
The shares of Common Stock referenced above were issued in reliance upon the exemption from securities registration afforded by the provisions of Regulation S of the Securities Act of 1933, as amended, (“Securities Act”), as promulgated by the U.S. Securities and Exchange Commission under the Securities Act. Our reliance upon the exemption under Rule 903 of Regulation S of the Securities Act was based on the fact that the sales of the securities were completed in an "offshore transaction", as defined in Rule 902(h) of Regulation S. We did not engage in any directed selling efforts, as defined in Regulation S, in the United States in connection with the sale of the securities. The investor was not a US person, as defined in Regulation S, and was not acquiring the securities for the account or benefit of a US person.
Securities Authorized for Issuance Under Equity Compensation Plans
The Company does not currently have any equity compensation plans and thus no securities have been issued.
As a “smaller reporting company”, we are not required to provide the information required by this Item.
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATION
The following discussion should be read in conjunction with our audited financial statements and the related notes that appear elsewhere in this Annual Report. The following discussion contains forward-looking statements that reflect our plans, estimates and beliefs. Our actual results could differ materially from those discussed in such forward looking statements. Factors that could cause or contribute to such differences include those discussed below and elsewhere in this Annual Report.
Our financial statements are stated in United States dollars and are prepared in accordance with United States generally accepted accounting principles.
The Company has only recently commenced generated revenues from planned principal operations and continues to develop its intellectual property to expand its revenue base through joint ventures and commercialization arrangements with third parties.
Results of Operations for the Year ended December 31, 2016 Compared to the Year ended December 31, 2015
Revenue, Cost of Revenue, and Gross Profit - We generated $687,448 in revenues from operations and $321,713 in cost of goods sold for the year ended December 31, 2016, compared to $60,121 in revenue and $31,787 costs of goods sold for the year ended December 31, 2015. The substantive increase to revenues is due primarily to increased revenues at our Glasgow P&O Clinic which carried out a full year of operations in fiscal 2016 after being fully upgraded and renovated during fiscal 2015. In addition our presence in the Glasgow market for services had been securely established during fiscal 2016 allowing a steady increase to monthly patient intake. Further in fiscal 2016 the Company received its first revenues from technology licensing agreements including technology access agreements to vet and commercialize certain applications of our Flexisense technology. There were no similar revenues in fiscal 2015.
Operating Expenses – Operating expenses decreased by $5,314,415 to $1,971,308 in 2016 compared to $7,285,723 in operating expenses in 2015, a decrease of approximately 73%. The substantial decrease in research and development by $4,977,160 from $5,317,942 (2015) to $340,782 (2016) is predominantly related to the issuance of 7,000,000 shares of common stock to Kapatos valued at fair market value on issuance totaling $4,550,000 during the year ended December 31, 2015, with no similar expense in fiscal 2016. However, consultancy fees increased substantially from $246,178 (2015) to $542,456 in the current year, predominantly related to the issuance of 800,000 shares as a result of various consulting agreement with third parties valued at $689,000, with no comparative expenses in the prior period ended December 31, 2015. The agreements relate to our ongoing business development efforts. The increase to consultancy fees in the current year is net $469,640 in accruals which were reversed in the current period as a certain services agreement was terminated and fees prior accrued waived in full. Further, there was a decrease in both professional fees and office expenses during the year ended December 31, 2016 due to the fact that certain stock awards issued in the year ended December 31, 2015 to in-house legal counsel and an administrative support assistant had no recurring charges during the year ended December 31, 2016. However, we did record expense relative to stock awards in fiscal 2016 for our Vice President totaling $169,000.
Loss from Operations - Due to the substantive increase in revenue and decrease in operating expenses, including a dramatic reduction to fees allocated to Research and Development, as well as the cancelation of fees payable under a terminated consulting contract we recorded a loss from operations of $1,605,573 during the year ended December 31, 2016 as compared to $7,257,389 in the prior comparative twelve-month period.
Other expenses - during year ended December 31, 2016, a substantive decrease to interest from $379,743 (2015) to $3,982 (2016) as a result of the settlmeent of a substantial amount of convertible notes payable during fiscal 2015 which greatly reduced our debt load. During the year ended December 31, 2015, a loss on debt settlement of $1,804,704 was recorded in respect of the settlement of this debt due to the difference in the market price of the shares issued on settlement, and the conversion price agreed with the noteholder. There was no comparative amount in the same period in 2016. During fiscal 2016 we managed to repay a substantive portion of outstanding loans from related parties prior to the accrual of substantive interest expense.
Net Loss - As a result of the substantial decrease in operating expenses during the year ended December 31, 2016 and the substantive increase to gross profit, the Company recorded a net loss $1,596,159 in the year ended December 31, 2016 compared to a net loss of $9,436,259 in the year ended December 31, 2015.
42
CAPITAL RESOURCES AND LIQUIDITY
At December 31, 2016, we had $2,492 cash on hand and liabilities of $1,020,951 in accounts payable and accrued expenses (including amounts due to related parties) and $383,164 in advances from related parties. Presently we rely on our President and director, as well as private placements from qualified investors to fund our general operating expenses. The Company has secured operating loans and advances from the Company’s Executive Officers of $511,850 in 2016 and $455,364 in 2015, and has closed private placements in the amount of $844,287 during the year ended December 31, 2016. These amounts contribute to our ongoing operating expenses and obligations until such time as the Company can conclude a larger equity based financing, anticipated during the early part of the second quarter of fiscal 2017.
The Company expects it will need to raise a total of $5,000,000 to fund its proposed operations for the next twelve months. It intends to fund operations by a combination of related party loans and equity placements. The Company intends to undertake a registration statement or bond offering to raise further funds up to a total of up to $30,000,000 which it hopes to file prior during fiscal 2017. In the interim, the Company is seeking equity private placements with which to fund operations until such time as it can file and clear the proposed registration statements. Our President and CEO, Mr. Sotirios Leontaritis continues to personally fund any short fall in our ongoing operations by way of loans and advances.
There can be no assurance that continued funding will be available on satisfactory terms.
GOING CONCERN
The Company incurred net losses of $1,596,159 and $9,436,259 for the year ended December 31, 2016 and 2015, respectively and has a retained deficit of $31,749,207. In addition, the Company had a working capital deficiency of $1,298,956 and a stockholders' deficit of $1,084,825 at December 31, 2016. These factors raise substantial doubt about the Company's ability to continue as a going concern.
There can be no assurance that sufficient funds required during the next year or thereafter will be generated from operations or that funds will be available from external sources such as debt or equity financings or other potential sources. The lack of additional capital resulting from the inability to generate cash flow from operations or to raise capital from external sources would force the Company to substantially curtail or cease operations and would, therefore, have a material adverse effect on its business. Furthermore, there can be no assurance that any such required funds, if available, will be available on attractive terms or that they will not have a significant dilutive effect on the Company's existing stockholders.
The accompanying financial statements do not include any adjustments related to the recoverability or classification of asset-carrying amounts or the amounts and classification of liabilities that may result should the Company be unable to continue as a going concern.
Need for Additional Financing
Costs associated with being a public company are much higher than those of a private company. There are present legal and accounting expenses, reporting requirements to the SEC, and investor relation costs that must be borne by a public company but not by a private company. These costs can be a burdensome expense which could adversely affect our financial survival. The ongoing regulatory costs, reporting requirements, and management details, which must be met when maintaining a public company, may make the economic viability of our Company very doubtful. In addition, the Company has projected it will need to raise up to $5,000,000 to meet its planned business objectives for 2017, including the acquisition of a new P&O clinic and continuation of its ongoing technology licensing and development efforts and there can be no assurance those funds will be available if, and when needed.
In the past we have relied on advances from our president to cover our operating costs, as well as equity private placements from qualified investors. There can be no assurance that the Company will be successful in finding joint venture partners or commercial partners to market and develop its technologies, or that funds for the development of the Company’s additional in-house technology development will be available if and when needed, or that funding will be available for marketing our products, if and when required. We were unable to open our planned clinic in Athens during fiscal 2016 as a result of a shortfall of available funds. There can be no assurance that the Company will be able to obtain funding as needed in order to meet our objectives on our proposed timeline, or at all.
We have no significant off-balance sheet arrangements that have or are reasonably likely to have a current or future effect on our financial condition, changes in our financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that are material to our stockholders.
43
Contractual Obligations
The following table outlines payments due under our significant contractual obligations over the periods shown, exclusive of interest, and based on the assumption that one year employment contracts terminate at the close of their one year term, without renewal. Further for any office locations where there is no underlying long term lease we have included amounts we estimate payable on a month to month basis for a one year term:
|
Payments due by Period
|
||||||||||||||||||||
|
Less than
|
One to
|
Three to
|
More Than
|
|||||||||||||||||
|
Contractual Obligations At December 31, 2016
|
One Year
|
Three Years
|
Five Years
|
Five Years
|
Total
|
|||||||||||||||
| Salaries and Wages | $ | 470,310 | $ | 240,000 | $ | - | $ | - | $ | 710,310 | ||||||||||
| Office lease and rentals | 72,560 | 98,583 | 171,143 | |||||||||||||||||
|
Related Parties Debt
|
|
383,164
|
|
-
|
|
-
|
|
-
|
|
383,164
|
||||||||||
|
Convertible Debt
|
$
|
- |
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
||||||||||
The above table outlines our obligations as of December 31, 2016 and does not reflect any changes in our obligations that have occurred after that date.
Critical Accounting Policies
Principals of Consolidation
The consolidated financial statements include the accounts of HCi Viocare and its wholly-owned subsidiaries, HCi Viocare Technologies Limited, HCi Viocare Clinics UK Limited and HCi Viocare Clinics (Hellas) S.A. All significant intercompany balances and transactions have been eliminated.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with generally accepted accounting principles in the United States of America ("US GAAP") and are presented in U.S. dollars.
Use of Estimates
The preparation of financial statements in conformity with generally accepted accounting principles requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenue and expenses during the reporting period. Actual results when ultimately realized could differ from these estimates.
Cash and Cash Equivalents
Cash and cash equivalents include cash on hand, deposits in banks with maturities of three months or less, and all highly liquid investments which are unrestricted as to withdrawal or use, and which have original maturities of three months or less.
Fair Value of Financial Instruments
The carrying value of financial instruments including cash and cash equivalents, receivables, prepaid expenses, accounts payable and accrued expenses, approximates their fair value due to the relatively short-term nature of these instruments.
Foreign Currencies
Items included in the consolidated financial statements of each of the Company and its subsidiaries are measured using the currency of the primary economic environment in which the entity operates (the ‘functional currency’). The Company's reporting currency is the U.S. dollar. The functional currency of subsidiaries based in the UK is pound sterling and the functional currency of the Company's subsidiary based in the Greece is the Euro. All transactions initiated in Pounds and Euro are translated into U.S. dollars in accordance with Accounting Standards Codification (“ASC”) 830-30, "Translation of Financial Statements," as follows:
i) assets and liabilities are translated at the closing rate at the date of the balance sheet or 1USD=0.9490EUR, 1USD=0.8127GBP;
ii) income and expenses are translated at average exchange rates, or 1USD=0.9636EUR, 1USD=0.73996GBP;
iii) all resulting exchange differences are recognized as other comprehensive income, a separate component of equity.
Adjustments arising from such translations are included in accumulated other comprehensive income (loss) in stockholders’ equity.
44
Revenue Recognition
The Company recognizes revenue when the earnings process is complete and persuasive evidence of an arrangement exists. This generally occurs when prosthetic products are fitted to customers, both title and the risks and rewards of ownership are transferred or services have been rendered and accepted, the selling price is fixed or determinable, and collectability is reasonably assured.
Inventory
Inventories, which consist principally of raw materials and parts, are stated at the lower of cost or market. Cost is determined using the first-in, first-out method and are adjusted to actual cost quarterly based on a physical count. Net realizable value is the estimated selling price in the ordinary course of business, less applicable variable selling expenses. Work-in-process inventory consists of materials, labor and a predetermined fixed rate of overhead which is valued based on established standards for the stage of completion of each custom order. We do not carry finished goods on hand. Material, labor and overhead costs are determined at the individual clinic level. Presently we only maintain very limited parts and raw materials inventory.
Warranty
We do not record warranty liabilities at the time of sale for the estimated costs that may be incurred under the terms of the applicable limited warranty as all component parts are covered by our respective industry suppliers.
Advertising Costs
The Company expenses advertising costs as incurred or the first time the advertising takes place, whichever is earlier, in accordance with ASC 720-35. Advertising costs were immaterial for the year ended December 31, 2016 and the year ended December 31, 2015, respectively.
Research and Development Costs
The Company charges research and development costs to expense when incurred in accordance with FASB ASC 730, “Research and Development”. Research and development costs were $340,782 and $5,317,942 for the year ended December 31, 2016 and 2015, respectively.
Related parties
For the purposes of these financial statements, parties are considered to be related if one party has the ability, directly or indirectly, to control the party or exercise significant influence over the party in making financial and operating decisions, or vice versa, or where the Company and the party are subject to common control or common significant influence. Related parties may be individuals or other entities.
Stock-based compensation
For stock-based compensation the Company follows the guidance codified in the Compensation – Stock Compensation Topic of FASB ASC (“ASC 718”). The Company determines the value of stock issued at the date of grant. It also determines at the date of grant, the value of stock at fair market value or the value of services rendered (based on contract or otherwise) whichever is more readily determinable.
45
Income Taxes
The Company accounts for income taxes in accordance with FASB ASC 740, "Income Taxes", which requires the asset and liability approach for financial accounting and reporting for income taxes. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance related to deferred tax assets is recorded when it is more likely than not that some portion or all of the deferred tax assets will not be realized.
The Company applies the prevailing taxation rates for operating subsidiaries in the United Kingdom and Greece. In each of these jurisdictions deferred tax arises as a result of including items of income and expenditure in taxation computations in periods different from those in which they are included in the company's financial statements. Deferred tax is provided in full on timing differences which result in an obligation to pay more tax, or a right to pay less tax, at a future date at the average tax rates that are expected to apply when the timing differences reverse, based on current tax rates and laws. In Greece, after the Company's first year of recorded net income over EUR$1, the Comapny is required to make advance tax payments monthly based on the calculated income tax payable on the the prior year's operations.
The Company has retained deficit from operations with respect to all its operating entities. Because there is no certainty that we will realize taxable income in the future, the Company did not record any deferred tax benefit as a result of these losses.
Income tax years for 2013, 2014 and 2015 are open to examination by the taxing authorities.
Earnings (Loss) Per Share
The Company reports earnings per share in accordance with FASB ASC 260, “Earnings Per Share.” FASB ASC 260 requires presentation of basic and diluted earnings per share in conjunction with the disclosure of the methodology used in computing such earnings per share. Basic earnings (loss) per share is computed by dividing income (loss) available to common shareholders by the weighted-average number of common shares outstanding during the period. Diluted earnings per share is computed similar to basic earnings per share except that the denominator is increased to include the number of additional common shares that would have been outstanding if the potential common shares had been issued and if the additional common shares were dilutive. The Company had potentially dilutive securities outstanding (convertible debt and liabilities) for the years ended December 31, 2016 and 2015, respectively, however, since the Company reflected a net loss in the years ended December 31, 2016 and 2015, the effect of considering any common stock equivalents, if outstanding, would have been anti-dilutive. A separate computation of diluted earnings (loss) per share is not presented.
|
December 31, 2016
|
||||
|
Common stock issuable upon conversion of 25,000 Series A Preferred Stock Options
|
500,000 | |||
Comprehensive Income
FASB ASC 220, “Comprehensive Income”, establishes standards for reporting and display of comprehensive income, its components and accumulated balances. Comprehensive income, as defined, includes all changes in equity during a period, exclusive of shareholder transactions. Accordingly, comprehensive income (loss) may include certain changes in shareholders’ equity (deficit) that are excluded from net income (loss).
Segment Reporting
FASB ASC 820 “Segments Reporting” establishes standards for reporting information about operating segments on a basis consistent with the Company’s internal organization structure as well as information about geographical areas, business segments and major customers in financial statements. Our proposed business segments are expected to span more than one geographical area. Specifically, the Company intends to operate prosthetic and orthotic rehabilitation clinics with various European based locations, as well as a corporate development and technology center which will undertake ongoing research and marketing activities.
46
Fair Value of Measurements
Accounting principles generally accepted in the United States define fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants at the measurement date. Additionally, the inputs used to measure fair value are prioritized based on a three-level hierarchy. This hierarchy requires entities to maximize the use of observable inputs and minimize the use of unobservable inputs. The three levels of inputs used to measure fair value are as follows:
Level 1: Unadjusted quoted prices in active markets for identical assets or liabilities.
Level 2: Input other than quoted market prices that are observable, either directly or indirectly, and reasonably available. Observable inputs reflect the assumptions market participants would use in pricing the asset or liability and are developed based on market data obtained from sources independent of the Company.
Level 3: Unobservable inputs. Unobservable inputs reflect the assumptions that the Company develops based on available information about what market participants would use in valuing the asset or liability.
An asset or liability’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement. Availability of observable inputs can vary and is affected by a variety of factors. The Company uses judgment in determining fair value of assets and liabilities and Level 3 assets and liabilities involve greater judgment than Level 1 and Level 2 assets or liabilities.
Recent Accounting Pronouncements
In January 2016, the FASB issued ASU 2016-01, Financial Instruments - Overall: Recognition and Measurement of Financial Assets and Financial Liabilities. This new standard provides guidance on how entities measure certain equity investments and present changes in the fair value. This standard requires that entities measure certain equity investments that do not result in consolidation and are not accounted for under the equity method at fair value and recognize any changes in fair value in net income. ASU 2016-01 is effective for fiscal years beginning after December 31, 2017. The Company is currently evaluating the provisions of this guidance and assessing its impact on the Company's financial statements and disclosures.
In February 2016, the FASB issued ASU No. 2016-02, Leases. The new standard requires that all lessees recognize the assets and liabilities that arise from leases on the balance sheet and disclose qualitative and quantitative information about its leasing arrangements. The new standard will be effective for us on January 1, 2019. The Company is currently evaluating the provisions of this guidance and assessing its impact on the Company's financial statements and disclosures.
In March 2016, the FASB issued ASU No. 2016-09, Compensation - Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting. The new standard involves several aspects of the accounting for share-based payment transactions, including the income tax consequences, classification of awards as either equity or liabilities and classification on the statement of cash flows. The new standard will be effective for us on January 1, 2017. This standard is currently under evaluation by management, however, this standard is not expected to have a material impact on our financial position, results of operations or statement of cash flows upon adoption.
In June 2016, the Financial Accounting Standards Board ("FASB") issued Accounting Standards Update ("ASU") No. 2016-13, Financial Instruments—Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments. The new standard requires financial assets measured at amortized cost be presented at the net amount expected to be collected, through an allowance for credit losses that is deducted from the amortized cost basis. The standard will be effective for the Company beginning January 1, 2020, with early application permitted. This standard is not expected to have a material impact on our financial position, results of operations or statement of cash flows upon adoption.
47
In August 2016, the Financial Accounting Standards Board ("FASB") issued Accounting Standards Update ("ASU") No. 2016-15, Statement of Cash Flows (Topic 230): Classification of Certain Cash Receipts and Cash Payments. The new standard will make eight targeted changes to how cash receipts and cash payments are presented and classified in the statement of cash flows. The standard will be effective for the Company beginning January 1, 2018, with early application permitted. The standard will require adoption on a retrospective basis unless it is impracticable to apply, in which case we would be required to apply the amendments prospectively as of the earliest date practicable. The Company is currently evaluating the provisions of this guidance and assessing its impact on the Company's financial statements and disclosures.
Other accounting standards that have been issued or proposed by the FASB or other standards-setting bodies that do not require adoption until a future date are not expected to have a material impact on our financial statements upon adoption.
As a “smaller reporting company”, we are not required to provide the information required by this Item.
48
ITEM 8. FINANCIAL STATEMENTSAND SUPPLEMENTARY DATA
HCi Viocare
FINANCIAL STATEMENTS
|
PAGE
|
|||
|
Reports of Independent Registered Public Accounting Firms
|
F-2
|
||
|
Consolidated Balance Sheets
|
F-3
|
||
|
Consolidated Statements of Operations and Comprehensive Loss
|
F-4
|
||
|
Consolidated Statement of Changes in Shareholders’ Equity (Deficit)
|
F-5
|
||
|
Consolidated Statements of Cash Flows
|
F-6
|
||
|
Notes to the Audited Consolidated Financial Statements
|
F-7 to F-28
|
F-1
Heaton & Company, PLLC
240 N. East Promontory
Suite 200
Farmington, Utah 84025
(T) 801.218.3523
heatoncpas.com
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To The Board of Directors and Stockholders of
HCi Viocare
We have audited the accompanying consolidated balance sheets of HCi Viocare (the Company) as of December 31, 2016 and 2015, and the related statements of operations, stockholders’ equity (deficit) and cash flows for the years then ended. These consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States of America). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the consolidated financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of HCi Viocare as of December 31, 2016 and 2015, and the results of its operations and its cash flows for the years then ended, in conformity with accounting principles generally accepted in the United States of America.
The accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern. As discussed in Note 2 to the consolidated financial statements, the Company has negative working capital and has not generated revenues to cover operating expenses. These factors, among others, raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to this matter are also described in Note 2. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
/s/Heaton & Company, PLLC
Farmington, Utah
April 18, 2017
F-2
HCi Viocare
CONSOLIDATED BALANCE SHEETS
|
December 31,
2016
|
December 31,
2015
|
|||||||
|
ASSETS
|
||||||||
|
Current Assets:
|
||||||||
|
Cash and cash equivalents
|
$
|
2,492
|
$
|
9,579
|
||||
|
Accounts receivable
|
15,212
|
3,075
|
||||||
|
Other receivable
|
39,371
|
39,590
|
||||||
|
Inventory
|
4,524
|
6,346
|
||||||
|
Prepaid expenses (Note 5)
|
43,560
|
78,207
|
||||||
|
Total Current Assets
|
105,159
|
136,797
|
||||||
|
Long-term Assets
|
||||||||
|
Property, plant and equipment, net (Note 6)
|
214,131
|
310,612
|
||||||
|
Total Assets
|
$
|
319,290
|
$
|
447,409
|
||||
|
LIABILITIES AND STOCKHOLDERS' DEFICIT
|
||||||||
|
Current Liabilities:
|
||||||||
|
Accounts payable and accrued expenses
|
$
|
511,652
|
$
|
355,417
|
||||
|
Accounts payable and accrued expenses-related party (Note 10)
|
509,299
|
294,972
|
||||||
|
Advances from a related party (Note 10)
|
383,164
|
649,420
|
||||||
|
Liability for unissued shares
|
-
|
249,040
|
||||||
|
Corporate taxes
|
-
|
12,074
|
||||||
|
Total Current Liabilities
|
1,404,115
|
1,560,923
|
||||||
|
Loan from a related party (Note 8)
|
-
|
163,595
|
||||||
|
Deferred taxes
|
-
|
192
|
||||||
|
Total Liabilities
|
1,404,115
|
1,724,710
|
||||||
|
Commitments and Contingencies
|
||||||||
|
Stockholders' Equity (Deficit):
|
||||||||
|
Preferred stock, par value $0.0001, 5,000,000 shares authorized; none issued and outstanding as of September 30, 2016 and December 31, 2015
|
-
|
-
|
||||||
|
Common stock, par value $0.0001, 700,000,000 shares authorized; 192,814,844 shares issued and outstanding as of December 31, 2016 and 190,743,961 shares issued and outstanding as of December 31, 2015
|
19,281
|
19,074
|
||||||
|
Additional paid-in capital
|
30,561,930
|
28,767,850
|
||||||
|
Retained Deficit
|
(31,749,207
|
)
|
(30,153,048
|
)
|
||||
|
Accumulated other comprehensive income
|
83,171
|
88,823
|
||||||
|
Stockholders' equity (deficiency)
|
(1,084,825
|
)
|
(1,227,301
|
)
|
||||
|
Total Liabilities and Stockholders' Equity (Deficiency)
|
$
|
319,290
|
$
|
447,409
|
||||
See Notes to Consolidated Financial Statements
F-3
HCi Viocare
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
|
For the Year Ended
|
||||||||
|
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Revenues
|
||||||||
|
Sales
|
$
|
687,448
|
$
|
60,121
|
||||
|
Cost of goods sold
|
321,713
|
31,787
|
||||||
|
Gross Profit
|
365,735
|
28,334
|
||||||
|
Operating Expenses
|
||||||||
|
Depreciation
|
67,647
|
47,564
|
||||||
|
Office rent
|
106,530
|
122,169
|
||||||
|
Office expenses
|
197,885
|
373,149
|
||||||
|
Consultancy Fees
|
542,456
|
246,178
|
||||||
|
Professional fees
|
610,671
|
1,011,668
|
||||||
|
Research and development
|
340,782
|
5,317,942
|
||||||
|
Travel and entertainment
|
105,337
|
167,053
|
||||||
|
Total Operating Expenses
|
1,971,308
|
7,285,723
|
||||||
|
Income (loss) from Operations
|
(1,605,573
|
)
|
(7,257,389
|
)
|
||||
|
Other Income (Expenses)
|
||||||||
|
Gain (loss) on debt settlement
|
-
|
(1,804,704
|
)
|
|||||
|
Gain (loss) on foreign currency transaction
|
2,196
|
6,483
|
||||||
|
Interest Expenses due to related party
|
(3,982
|
)
|
(379,743
|
)
|
||||
|
Total Other Income (Expenses)
|
(1,786
|
)
|
(2,177,964
|
)
|
||||
|
Income (Loss) before Provision for Income Tax
|
(1,607,359
|
)
|
(9,435,353
|
)
|
||||
|
Provision for Income Tax
|
11,200
|
(906
|
)
|
|||||
|
Net Income (Loss)
|
$
|
(1,596,159
|
)
|
$
|
(9,436,259
|
)
|
||
|
Basic and fully diluted loss per share
|
$
|
(0.01
|
)
|
$
|
(0.06
|
)
|
||
|
Weighted average shares outstanding
|
192,272,521
|
155,174,460
|
||||||
|
Comprehensive Income (Loss) :
|
||||||||
|
Net loss
|
$
|
(1,596,159
|
)
|
$
|
(9,436,259
|
)
|
||
|
Effect of foreign currency translation
|
(5,652
|
)
|
35,232
|
|||||
|
Comprehensive Loss
|
$
|
(1,601,811
|
)
|
$
|
(9,401,027
|
)
|
||
See Notes to Consolidated Financial Statements
F-4
| Common stock |
Additional
Paid-in
Capital
|
Accumulated
other
comprehensive
income (loss)
|
||||||||||||||||||||||
| Shares | Amount |
Retained
deficit
|
Total
equity
|
|||||||||||||||||||||
|
Balances, December 31, 2014
|
102,458,881
|
$
|
10,246
|
$
|
20,632,647
|
$
|
53,591
|
$
|
(20,716,789
|
)
|
$
|
(20,305
|
)
|
|||||||||||
|
Proceeds received from issuance of common stock in private placement
|
1,319,180
|
132
|
550,315
|
-
|
-
|
550,447
|
||||||||||||||||||
|
Shares issuance for stock award
|
816,669
|
81
|
533,254
|
-
|
-
|
533,335
|
||||||||||||||||||
|
Issuance of common stock in connection with the acquisition of patented technologies
|
7,000,000
|
700
|
4,549,300
|
-
|
-
|
4,550,000
|
||||||||||||||||||
|
Conversion of convertible note and debt settlement
|
79,037,231
|
7,904
|
2,135,535
|
-
|
-
|
2,143,439
|
||||||||||||||||||
|
Shares issuance for services provided
|
112,000
|
11
|
75,589
|
-
|
-
|
75,600
|
||||||||||||||||||
|
Vesting of stock options
|
-
|
-
|
-291,210
|
-
|
-
|
291,210
|
||||||||||||||||||
|
Net income (loss)
|
-
|
-
|
-
|
-
|
(9,436,259
|
)
|
(9,436,259
|
)
|
||||||||||||||||
|
Foreign currency translation adjustments
|
-
|
-
|
-
|
35,232
|
-
|
35,232
|
||||||||||||||||||
|
Balances, December 31, 2015
|
190,743,961
|
19,074
|
28,767,850
|
88,823
|
(30,153,048
|
)
|
(1,277,301
|
)
|
||||||||||||||||
|
Proceeds received from issuance of common stock in private placement
|
1,633,383
|
163
|
844,124
|
- | - |
844,287
|
||||||||||||||||||
|
Shares issuance for stock award
|
275,000
|
28
|
248,722
|
- | - |
248,750
|
||||||||||||||||||
|
Shares issuance for services provided
|
862,500
|
86
|
715,164
|
- | - |
715,250
|
||||||||||||||||||
|
Share cancellation
|
(700,000
|
)
|
(70)
|
(13,930
|
)
|
- | - |
(14,000
|
)
|
|||||||||||||||
|
Net income (loss)
|
- | - | - | - |
(1,596,159
|
)
|
(1,596,159
|
)
|
||||||||||||||||
|
Foreign currency translation adjustments
|
- | - | - |
(5,652
|
)
|
- |
(5,652
|
)
|
||||||||||||||||
|
Balances, December 31, 2016
|
192,814,844
|
$
|
19,281
|
$
|
30,561,930
|
$
|
83,171
|
$
|
(31,749,207
|
)
|
$
|
(1,084,825
|
)
|
|||||||||||
See Notes to Consolidated Financial Statements
F-5
HCi Viocare
CONSOLIDATED STATEMENTS OF CASH FLOWS
|
For the Year Ended
|
||||||||
|
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Operating Activities
|
||||||||
|
Net loss
|
$
|
(1,596,159
|
)
|
$
|
(9,436,259
|
)
|
||
|
Adjustments to reconcile net loss to net cash used by operating activities:
|
||||||||
|
Stock options vested
|
-
|
291,210
|
||||||
|
Shares issued for acquisition of patented technologies
|
-
|
4,550,000
|
||||||
|
Shares issued for stock award
|
248,750
|
533,335
|
||||||
|
Shares issued for services provided
|
715,250
|
75,600
|
||||||
|
Amortization of beneficial conversion feature
|
-
|
368,273
|
||||||
|
Gain (loss) on debt settlement
|
-
|
1,804,704
|
||||||
|
Gain on foreign currency transactions
|
-
|
(27,411
|
)
|
|||||
|
Depreciation
|
67,647
|
47,564
|
||||||
|
Changes in operating assets and liabilities:
|
||||||||
|
Decrease (Increase) in accounts receivable
|
(11,244
|
)
|
(2,201
|
)
|
||||
|
Decrease (Increase) in prepaid expense
|
25,654
|
(39,646
|
)
|
|||||
|
Decrease (Increase) in other receivable
|
(12,032
|
)
|
(41,289
|
)
|
||||
|
Decrease (Increase) in inventory
|
752
|
85
|
||||||
|
Increase (Decrease) in accounts payable and accrued expenses
|
172,528
|
291,186
|
||||||
|
Increase (Decrease) in accounts payable and accrued expenses, related party
|
220,659
|
218,830
|
||||||
|
Increase (Decrease) in corporate taxes
|
(10,037
|
)
|
12,074
|
|||||
|
Increase (Decrease) in liability of unissued shares
|
(249,040
|
)
|
249,040
|
|||||
|
Net cash used by operating activities
|
(427,272
|
)
|
(1,104,905
|
)
|
||||
|
Investing Activities
|
||||||||
|
Additions to Property, plant and equipment
|
(17,016
|
)
|
(274,470
|
)
|
||||
|
Net cash (used) by investing activities
|
(17,016
|
)
|
(274,470
|
)
|
||||
|
Financing Activities
|
||||||||
|
Advances and Loans from related parties
|
511,850
|
455,364
|
||||||
|
Repayments, advances and loans from related party
|
(905,681
|
)
|
-
|
|||||
|
Proceeds from private placement
|
844,287
|
550,447
|
||||||
|
Net cash provided by financing activities
|
450,456
|
1,005,811
|
||||||
|
Increase (decrease) in cash
|
6,168
|
(373,564
|
)
|
|||||
|
Cash at beginning of period
|
9,579
|
408,252
|
||||||
|
Effects of exchange rates on cash
|
(13,255
|
)
|
(25,109
|
)
|
||||
|
Cash at end of period
|
$
|
2,492
|
$
|
9,579
|
||||
|
Supplemental Disclosures of Cash Flow Information:
|
||||||||
|
Cash paid during the year for:
|
||||||||
|
Interest
|
$
|
8,750
|
$
|
-
|
||||
|
Income taxes
|
$
|
-
|
$
|
-
|
||||
|
Non-Cash Investing and Financing Activities:
|
||||||||
|
Convertible notes converted to shares
|
$
|
- |
|
$
|
326,619
|
|||
|
Interest payable converted to shares
|
$
|
- |
|
$
|
12,116
|
|||
See Notes to Consolidated Financial Statements
F-6
Note 1 - ORGANIZATION AND BUSINESS BACKGROUND
HCi Viocare ("VICA" or the "Company") was incorporated on March 26, 2007 under the laws of the State of Nevada. The Company has selected December 31 as its fiscal year end.
While the Company has generated revenues from a portion of its planned principal operations, we are not yet able to meet operational overheads and are not yet profitable. We are considered an emerging growth enterprise. The Company was originally formed to sell medical devices with an emphasis on portable medical devices designed for home treatments with the initial focus in the northern regions of China. The Company’s intent was to seek strategic relationships with medical device manufacturers both in China and North America with the aim to be their sales and distribution agent in Northern China and to assist Chinese medical device manufacturers on the development of the North American market.
On September 10, 2013, the controlling shareholder of the Company sold his controlling interest in the shares of the Company and there was a change in the Board of Directors of the Company, effecting a change in control of the Company. The business of the Company remains in the field of medical devices and other opportunities related to their uses. We are currently engaged in the technology development and licensing of bioengineering innovations for the health, sports and wellness sectors, and we intend to be engaged in the operation of prosthetic and orthotic (P&O) total rehabilitation clinics.
On January 15, 2014, the Company incorporated two wholly-owned subsidiaries in Scotland, U.K., HCi Viocare Technologies Limited and HCi Viocare Clinics UK Limited. The Company intends to operate in Scotland under these two subsidiaries, one of which will undertake the development and marketing of technologies and the other which is a P&O clinic, which will serve as the centre of reference and training for the company’s further clinics.
On February 12, 2014, through our wholly owned subsidiary, HCi Viocare Technologies Limited, the Company acquired an interest in a patented technology known as “Socket-Fit”. SocketFit is a system that will help overcome technical and resource hurdles endemic to the prosthetic sector. The system has been designed with the aim of offering optimally fitted prosthetic sockets that will reduce the number of prostheses made for patients, resulting in a reduced number of visits by the patient to the prosthetic, and also assisting in the rehabilitation of amputees. Socket-Fit is a digital system for assessing an amputee’s residual limb and for the production of truly functional and comfortable prosthetic sockets. The technology takes account of the external and internal geometry of the amputee’s stump, the biomechanical properties of each individual soft tissue layer and the boundary and loading conditions of a complete prosthesis to generate a virtual 3D model of the residual limb making it possible to produce an accurate, functional and comfortable prosthetic socket. By minimizing the time and cost of socket production and reducing the number of faulty sockets there will be a reduction in costs incurred by health services and insurance companies worldwide as well as benefits to the amputee. The Company intends to undertake and fund, through its U.K. subsidiary, a project to improve the nature of the data used in socket modeling software with a view to creating a system that will enable prosthetists to build a socket that evenly distributes weight, provides enhanced comfort, and can be marketed and used across the industry for improved socket creation.
On February 19, 2014, the Company filed a Certificate of Amendment with the Secretary of State of Nevada to change the name of the Company to HCi Viocare effective March 21, 2014. Effective March 21, 2014, in accordance with approval from FINRA, we changed our name from China Northern Medical Device, Inc. to HCi Viocare. Concurrently we commenced trading on the Over-the-Counter Bulletin Board under the symbol “VICA”.
On April 16, 2014, through our wholly owned subsidiary HCi Viocare Technologies Limited, we acquired all rights and interest in and to the background Intellectual Property Rights (“IPR”) for a developing technology known as “Smart Insole”. The Smart Insole system is believed to be a state-of-the-art, pressure and shear (friction)-sensing insole that can wirelessly communicate with connected devices. The insole has a number of applications, including the mitigation of diabetic foot complications, such as ulceration, infection and amputation, in clinical gait analysis and in sports, as a wearable device to help athletes optimize their performance and prevent injury. The sensing system is very low cost, compared to traditional pressure sensing technologies, in our opinion making such applications affordable to the consumer for the first time.
F-7
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 1 - ORGANIZATION AND BUSINESS BACKGROUND (continued)
On June 9, 2014, the Company, through our wholly owned subsidiary, HCi Viocare Clinics, acquired W D Spence Prosthetics Limited (the “Clinic”). The Clinic is located in Glasgow, Scotland, and is a fully operational prosthetics and orthotics clinic. The acquisition of the Clinic is the first step to the Company’s and HCi Viocare Clinics’ intention to develop the first chain of prosthetics and orthotics (P&O) and diabetic foot rehabilitation clinics in the European market, covering Southern Europe, the Middle East and North Africa.
On March 31, 2015, the Company’s wholly owned subsidiary HCi Viocare Clinics and its subsidiary W D Spence Prosthetics Limited completed a merger with the resulting combined entity having the name HCi Viocare Clinics UK Limited.
On July 8, 2015, the Company incorporated HCi Viocare Clinics (Hellas) S A in order to carry out operations for the P&O clinic under development in Athens, Greece.
On August 8, 2015 FINRA approved a seven (7) new for one (1) old forward split of our authorized and issued and outstanding shares of common. A Certificate of Change for the stock split was filed and became effective with the Nevada Secretary of State on August 7, 2015. Consequently, our authorized share capital increased from 100,000,000 to 700,000,000 shares of common stock and our issued and outstanding common stock increased accordingly, all with a par value of $0.0001. Our preferred stock remained unchanged. All share and per share figures contained herein reflect the impact of the forward split.
Note 2 - GOING CONCERN
The Company incurred net losses of $1,596,159 and $9,436,259 for the year ended December 31, 2016 and 2015, respectively and has a retained deficit of $31,749,207. In addition, the Company had a working capital deficiency of $1,298,956 and a stockholders' deficit of $1,084,825 at December 31, 2016. These factors raise substantial doubt about the Company's ability to continue as a going concern.
There can be no assurance that sufficient funds required during the next year or thereafter will be generated from operations or that funds will be available from external sources such as debt or equity financings or other potential sources. The lack of additional capital resulting from the inability to generate cash flow from operations or to raise capital from external sources would force the Company to substantially curtail or cease operations and would, therefore, have a material adverse effect on its business. Furthermore, there can be no assurance that any such required funds, if available, will be available on attractive terms or that they will not have a significant dilutive effect on the Company's existing stockholders.
The accompanying financial statements do not include any adjustments related to the recoverability or classification of asset-carrying amounts or the amounts and classification of liabilities that may result should the Company be unable to continue as a going concern.
The Company has relied heavily for its financing needs on its Chief Executive Officer and President as more fully disclosed in Note 10.
Note 3 - CONTROL BY PRINCIPAL STOCKHOLDER/OFFICER
Our Chief Executive Officer owns beneficially and in the aggregate, the majority of the voting power of the Company. Accordingly, the Chief Executive Officer has the ability to control the approval of most corporate actions, including approving significant expenses, increasing the authorized capital stock and the dissolution, merger or sale of the Company's assets.
F-8
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 4 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Principals of Consolidation
The consolidated financial statements include the accounts of HCi Viocare and its wholly-owned subsidiaries, HCi Viocare Technologies Limited, HCi Viocare Clinics UK Limited and HCi Viocare Clinics (Hellas) S.A. All significant intercompany balances and transactions have been eliminated.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with generally accepted accounting principles in the United States of America ("US GAAP") and are presented in U.S. dollars.
Use of Estimates
The preparation of financial statements in conformity with generally accepted accounting principles requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenue and expenses during the reporting period. Actual results when ultimately realized could differ from these estimates.
Cash and Cash Equivalents
Cash and cash equivalents include cash on hand, deposits in banks with maturities of three months or less, and all highly liquid investments which are unrestricted as to withdrawal or use, and which have original maturities of three months or less.
Fair Value of Financial Instruments
The carrying value of financial instruments including cash and cash equivalents, receivables, prepaid expenses, accounts payable and accrued expenses, approximates their fair value due to the relatively short-term nature of these instruments.
Foreign Currencies
Items included in the consolidated financial statements of each of the Company and its subsidiaries are measured using the currency of the primary economic environment in which the entity operates (the ‘functional currency’). The Company's reporting currency is the U.S. dollar. The functional currency of subsidiaries based in the UK is pound sterling and the functional currency of the Company's subsidiary based in the Greece is the Euro. All transactions initiated in Pounds and Euro are translated into U.S. dollars in accordance with Accounting Standards Codification (“ASC”) 830-30, "Translation of Financial Statements," as follows:
i) assets and liabilities are translated at the closing rate at the date of the balance sheet or 1USD=0.9490EUR, 1USD=0.8127GBP;
ii) income and expenses are translated at average exchange rates, or 1USD=0.9636EUR, 1USD=0.73996GBP;
iii) all resulting exchange differences are recognized as other comprehensive income, a separate component of equity.
Adjustments arising from such translations are included in accumulated other comprehensive income (loss) in stockholders’ equity.
F-9
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 4 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Revenue Recognition
The Company recognizes revenue when the earnings process is complete and persuasive evidence of an arrangement exists. This generally occurs when prosthetic products are fitted to customers, both title and the risks and rewards of ownership are transferred or services have been rendered and accepted, the selling price is fixed or determinable, and collectability is reasonably assured.
Inventory
Inventories, which consist principally of raw materials and parts, are stated at the lower of cost or market. Cost is determined using the first-in, first-out method and are adjusted to actual cost quarterly based on a physical count. Net realizable value is the estimated selling price in the ordinary course of business, less applicable variable selling expenses. Work-in-process inventory consists of materials, labor and a predetermined fixed rate of overhead which is valued based on established standards for the stage of completion of each custom order. We do not carry finished goods on hand. Material, labor and overhead costs are determined at the individual clinic level. Presently we only maintain very limited parts and raw materials inventory.
Warranty
We do not record warranty liabilities at the time of sale for the estimated costs that may be incurred under the terms of the applicable limited warranty as all component parts are covered by our respective industry suppliers.
Advertising Costs
The Company expenses advertising costs as incurred or the first time the advertising takes place, whichever is earlier, in accordance with ASC 720-35. Advertising costs were immaterial for the year ended December 31, 2016 and the year ended December 31, 2015, respectively.
Research and Development Costs
The Company charges research and development costs to expense when incurred in accordance with FASB ASC 730, “Research and Development”. Research and development costs were $340,782 and $5,317,942 for the year ended December 31, 2016 and 2015, respectively.
Related parties
For the purposes of these financial statements, parties are considered to be related if one party has the ability, directly or indirectly, to control the party or exercise significant influence over the party in making financial and operating decisions, or vice versa, or where the Company and the party are subject to common control or common significant influence. Related parties may be individuals or other entities.
Stock-based compensation
For stock-based compensation the Company follows the guidance codified in the Compensation – Stock Compensation Topic of FASB ASC (“ASC 718”). The Company determines the value of stock issued at the date of grant. It also determines at the date of grant, the value of stock at fair market value or the value of services rendered (based on contract or otherwise) whichever is more readily determinable.
F-10
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 4 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Income Taxes
The Company accounts for income taxes in accordance with FASB ASC 740, "Income Taxes", which requires the asset and liability approach for financial accounting and reporting for income taxes. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance related to deferred tax assets is recorded when it is more likely than not that some portion or all of the deferred tax assets will not be realized.
The Company applies the prevailing taxation rates for operating subsidiaries in the United Kingdom and Greece. In each of these jurisdictions deferred tax arises as a result of including items of income and expenditure in taxation computations in periods different from those in which they are included in the company's financial statements. Deferred tax is provided in full on timing differences which result in an obligation to pay more tax, or a right to pay less tax, at a future date at the average tax rates that are expected to apply when the timing differences reverse, based on current tax rates and laws. In Greece, after the Company's first year of recorded net income over EUR$1, the Comapny is required to make advance tax payments monthly based on the calculated income tax payable on the the prior year's operations.
The Company has retained deficit from operations with respect to all its operating entities. Because there is no certainty that we will realize taxable income in the future, the Company did not record any deferred tax benefit as a result of these losses.
Income tax years for 2013, 2014 and 2015 are open to examination by the taxing authorities.
Earnings (Loss) Per Share
The Company reports earnings per share in accordance with FASB ASC 260, “Earnings Per Share.” FASB ASC 260 requires presentation of basic and diluted earnings per share in conjunction with the disclosure of the methodology used in computing such earnings per share. Basic earnings (loss) per share is computed by dividing income (loss) available to common shareholders by the weighted-average number of common shares outstanding during the period. Diluted earnings per share is computed similar to basic earnings per share except that the denominator is increased to include the number of additional common shares that would have been outstanding if the potential common shares had been issued and if the additional common shares were dilutive. The Company had potentially dilutive securities outstanding (convertible debt and liabilities) for the years ended December 31, 2016 and 2015, respectively, however, since the Company reflected a net loss in the years ended December 31, 2016 and 2015, the effect of considering any common stock equivalents, if outstanding, would have been anti-dilutive. A separate computation of diluted earnings (loss) per share is not presented.
|
December 31, 2016
|
||||
|
Common stock issuable upon conversion of 25,000 Series A Preferred Stock Options
|
500,000 | |||
Comprehensive Income
FASB ASC 220, “Comprehensive Income”, establishes standards for reporting and display of comprehensive income, its components and accumulated balances. Comprehensive income, as defined, includes all changes in equity during a period, exclusive of shareholder transactions. Accordingly, comprehensive income (loss) may include certain changes in shareholders’ equity (deficit) that are excluded from net income (loss).
Segment Reporting
FASB ASC 820 “Segments Reporting” establishes standards for reporting information about operating segments on a basis consistent with the Company’s internal organization structure as well as information about geographical areas, business segments and major customers in financial statements. Our proposed business segments are expected to span more than one geographical area. Specifically, the Company intends to operate prosthetic and orthotic rehabilitation clinics with various European based locations, as well as a corporate development and technology center which will undertake ongoing research and marketing activities.
F-11
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 4 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Fair Value of Measurements
Accounting principles generally accepted in the United States define fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants at the measurement date. Additionally, the inputs used to measure fair value are prioritized based on a three-level hierarchy. This hierarchy requires entities to maximize the use of observable inputs and minimize the use of unobservable inputs. The three levels of inputs used to measure fair value are as follows:
Level 1: Unadjusted quoted prices in active markets for identical assets or liabilities.
Level 2: Input other than quoted market prices that are observable, either directly or indirectly, and reasonably available. Observable inputs reflect the assumptions market participants would use in pricing the asset or liability and are developed based on market data obtained from sources independent of the Company.
Level 3: Unobservable inputs. Unobservable inputs reflect the assumptions that the Company develops based on available information about what market participants would use in valuing the asset or liability.
An asset or liability’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement. Availability of observable inputs can vary and is affected by a variety of factors. The Company uses judgment in determining fair value of assets and liabilities and Level 3 assets and liabilities involve greater judgment than Level 1 and Level 2 assets or liabilities.
Recent Accounting Pronouncements
In January 2016, the FASB issued ASU 2016-01, Financial Instruments - Overall: Recognition and Measurement of Financial Assets and Financial Liabilities. This new standard provides guidance on how entities measure certain equity investments and present changes in the fair value. This standard requires that entities measure certain equity investments that do not result in consolidation and are not accounted for under the equity method at fair value and recognize any changes in fair value in net income. ASU 2016-01 is effective for fiscal years beginning after December 31, 2017. The Company is currently evaluating the provisions of this guidance and assessing its impact on the Company's financial statements and disclosures.
In February 2016, the FASB issued ASU No. 2016-02, Leases. The new standard requires that all lessees recognize the assets and liabilities that arise from leases on the balance sheet and disclose qualitative and quantitative information about its leasing arrangements. The new standard will be effective for us on January 1, 2019. The Company is currently evaluating the provisions of this guidance and assessing its impact on the Company's financial statements and disclosures.
In March 2016, the FASB issued ASU No. 2016-09, Compensation - Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting. The new standard involves several aspects of the accounting for share-based payment transactions, including the income tax consequences, classification of awards as either equity or liabilities and classification on the statement of cash flows. The new standard will be effective for us on January 1, 2017. This standard is currently under evaluation by management, however, this standard is not expected to have a material impact on our financial position, results of operations or statement of cash flows upon adoption.
In June 2016, the Financial Accounting Standards Board ("FASB") issued Accounting Standards Update ("ASU") No. 2016-13, Financial Instruments—Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments. The new standard requires financial assets measured at amortized cost be presented at the net amount expected to be collected, through an allowance for credit losses that is deducted from the amortized cost basis. The standard will be effective for the Company beginning January 1, 2020, with early application permitted. This standard is not expected to have a material impact on our financial position, results of operations or statement of cash flows upon adoption.
F-12
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 4 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (continued)
Recent Accounting Pronouncements (continued)
In August 2016, the Financial Accounting Standards Board ("FASB") issued Accounting Standards Update ("ASU") No. 2016-15, Statement of Cash Flows (Topic 230): Classification of Certain Cash Receipts and Cash Payments. The new standard will make eight targeted changes to how cash receipts and cash payments are presented and classified in the statement of cash flows. The standard will be effective for the Company beginning January 1, 2018, with early application permitted. The standard will require adoption on a retrospective basis unless it is impracticable to apply, in which case we would be required to apply the amendments prospectively as of the earliest date practicable. The Company is currently evaluating the provisions of this guidance and assessing its impact on the Company's financial statements and disclosures.
Other accounting standards that have been issued or proposed by the FASB or other standards-setting bodies that do not require adoption until a future date are not expected to have a material impact on our financial statements upon adoption.
Note 5 - PREPAID EXPENSES
Prepaid expenses consist of the following:
|
December 31,
2016
|
December 31,
2015
|
|||||||
|
Office lease, including security deposits
|
$
|
35,977
|
$
|
51,429
|
||||
|
Professional fees
|
-
|
13,779
|
||||||
|
Travel advances and other expenses
|
7,583
|
12,999
|
||||||
|
Total prepaid expense
|
$
|
43,560
|
$
|
78,207
|
||||
Note 6 - PROPERTY AND EQUIPMENT
|
|
Leasehold Improvement
|
Office Furniture
|
Computer
& Equipment
|
Vehicles
|
Plant
&
Machine
|
Lab Equipment
|
Total
|
|||||||||||||||||||||
|
Cost
|
||||||||||||||||||||||||||||
|
At December 31, 2014
|
30,221
|
6,353
|
12,523
|
60,782
|
-
|
-
|
109,879
|
|||||||||||||||||||||
|
Additions - purchase
|
188,559
|
25,841
|
16,860
|
-
|
1,343
|
41,867
|
274,470
|
|||||||||||||||||||||
|
Foreign exchange
|
(8,117
|
)
|
1,144
|
(1,954
|
)
|
(6,250
|
)
|
(57
|
)
|
(4,437
|
)
|
(19,671
|
)
|
|||||||||||||||
|
At December 31, 2015
|
210,663
|
33,338
|
27,429
|
54,532
|
1,286
|
37,430
|
364,678
|
|||||||||||||||||||||
|
Additions - purchase
|
3,413
|
416
|
5,343
|
-
|
7,844
|
-
|
17,016
|
|||||||||||||||||||||
|
Foreign exchange
|
(29,649
|
)
|
(4,230
|
)
|
(3,078
|
)
|
(1,846
|
)
|
(734
|
)
|
(6,313
|
)
|
(45,850
|
)
|
||||||||||||||
|
At December 31, 2016
|
184,427
|
29,524
|
29,694
|
52,686
|
8,396
|
31,117
|
335,844
|
|||||||||||||||||||||
|
Amortization
|
||||||||||||||||||||||||||||
|
At December 31, 2014
|
3,299
|
1,332
|
1,340
|
531
|
-
|
-
|
6,502
|
|||||||||||||||||||||
|
Charge for the period
|
21,317
|
5,557
|
5,363
|
11,248
|
134
|
3,945
|
47,564
|
|||||||||||||||||||||
|
At December 31, 2015
|
$
|
24,616
|
$
|
6,889
|
$
|
6,703
|
$
|
11,779
|
$
|
134
|
$
|
3,945
|
$
|
54,066
|
||||||||||||||
|
Charge for the period
|
33,612
|
8,024
|
7,330
|
10,560
|
1,008
|
7,113
|
67,647
|
|||||||||||||||||||||
|
At December 31, 2016
|
58,228
|
14,913
|
14,033
|
22,339
|
1,142
|
11,058
|
121,713
|
|||||||||||||||||||||
|
Carrying Amounts
|
||||||||||||||||||||||||||||
|
At December 31, 2015
|
$
|
186,047
|
$
|
26,449
|
$
|
20,726
|
$
|
42,753
|
$
|
1,152
|
$
|
33,485
|
$
|
310,612
|
||||||||||||||
|
At December 31, 2016
|
$
|
140,224
|
$
|
17,368
|
$
|
18,044
|
$
|
35,240
|
$
|
8,222
|
$
|
23,261
|
$
|
214,131
|
||||||||||||||
F-13
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 6 - PROPERTY AND EQUIPMENT (continued)
Leasehold improvements are amortized over the term of the lease: three to ten years.
Furniture is amortized over three to five years and computer and equipment is amortized over three years.
Vehicles are amortized over five years.
Plant and machine equipment and lab equipment is amortized over 4 years.
Note 7 - OFFICE LEASE
Office in Greece
On February 3, 2014, the Company leased office space in Paleo Faliro, Greece on a three-year lease, commencing on March 1, 2014 and ending on February 28, 2017. The monthly rental fee is $1,700 (EUR € 1,554) including applicable taxes in the first year. Thereafter, for every further lease year and for the whole duration of the lease agreement, as well as in case of compulsory statutory extension or extension by tacit agreement, the monthly rent shall be annually adjusted by a percentage equal to the Consumer Price Index of the adjustment month with respect to the respective month of the year before (simple annual rate of change), as this is calculated by the Hellenic Statistical Authority (ELSTAT), plus two (2) percentage points (+2%).
Under the term of the lease agreement, the Company paid a total of $25,010 (EUR €18, 540) including $4,047 (EUR €3,000) for deposit and $20,963 (EUR €15,540) for ten months’ rental fee upon executing the agreement. As of December 31, 2016, $3,161 (EUR €3,000) is shown as deposit in the prepaid account. The lease expired subsequent to fiscal year end and is currently under negotiation for rewnewal on similar terms.
Clinic in Greece
On December 29, 2014, the Company leased office space at Peania Region of Attica in Greece on a ten-year lease, commencing on January 1, 2015 and ending on October 5, 2024. The monthly rental fee is $6,996 (EUR €6,233) plus applicable taxes and the monthly operating cost estimate is $1,120 (EUR €997).
Under the term of the lease agreement, the Company paid a total of $13,991 (EUR €12,465) as deposit in the prepaid account.
On October 9, 2015, the terms of the lease were amended to include the following provisions:
|
-
|
Effective September 1, 2015, the monthly rental fee shall be reduced to $3,500 (EUR €3,116) for a period of 12 months ending August 31, 2016.
|
|
-
|
In the event that the Lessee commences leasehold improvements prior to August 31, 2016, it shall inform the Lessor respectively and pay retroactively the remaining balance of the total amount of monthly rental in full from the period commencing September 1, 2015, as if they have not been reduced;
|
|
-
|
In the event that the Company does not proceed with leasehold improvements in the 12-month period, the lease agreement will terminate and the Lessor with retain the deposit.
|
|
-
|
The Lessor until such time as the leasehold improvements commence, may at any time and without indemnity terminate the lease agreement with a 30-day prior written notice to the Lessee.
|
As of December 31, 2016, the Company and the Lessor concluded the Termination of Lease Agreement dated August 1, 2016, and the Lessor waived all maintenance charges and utility charges for fiscal 2016. The following table is the summary of balance owing to Lessor as at December 31, 2016:
|
December 31, 2016
|
||||||||
|
Amount owed for rent
|
€
|
12,913
|
$
|
14,463
|
||||
|
Deduct: security deposit
|
(12,465
|
)
|
(13,991
|
)
|
||||
|
Total amount owed for rent
|
448
|
472
|
||||||
|
Total amount for maintenance charges (before 2016)
|
14,617
|
15,459
|
||||||
|
Total amount for utility charges (before 2016)
|
12,865
|
13,606
|
||||||
|
Total
|
€
|
27,930
|
$
|
29,537
|
||||
F-14
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 7 - OFFICE LEASE (continued)
Lease in UK
On March 2, 2015 the Company entered into a lease agreement for a modern, stand-alone 5,300 square foot facility in Glasgow, Scotland with an entry date of 1 March, 2015. The building will host the Company’s first Viocare center, a full service Prosthetic and Orthotic (“P&O”) practice with a superior standard of personalized care. The renovations to the clinic were completed in June, and the facility was opened on August 1, 2015 with an official ribbon cutting on 25 September 2015. During the first year from the date of entry, the lease fees are USD$26,000 (GBP £20,000) per annum (exclusive of VAT). On the second anniversary of the date of entry, the lease will increase to USD$52,000 (GBP £40,000) per annum (exclusive of VAT). In the third anniversary of the date of entry, the lease will be USD$52,000 (GBP £40,000) per annum (exclusive of VAT). In the fourth anniversary of the date of entry, the lease will decrease to USD$39,000(GBP £30,000) per annum (exclusive of VAT). In the fifth anniversary of the date of entry, the lease will be USD$52,000 (GBP £40,000) per annum (exclusive of VAT).
Under the term of the lease agreement, the Company paid a total of $28,738 (GBP £20,000) as deposit in the prepaid account.
|
Location
|
Greece
|
United Kingdom
|
||||||
|
As of December 31, 2015
|
$
|
16,867
|
$
|
34,562
|
||||
|
Prepaid rental fee
|
(13,991
|
)
|
3,248
|
|||||
|
Foreign exchange on the deposits
|
285
|
(4,994
|
)
|
|||||
|
As of December 31, 2016
|
$
|
3,161
|
$
|
32,816
|
||||
As of December 31, 2016, the approximate future aggregate minimum lease payments in respect of our current obligations were as follows:
|
Location
|
Greece
|
United Kingdom
|
||||||
|
2017(1)
|
$ | 20,560 | $ | 52,000 | ||||
|
2018
|
- | 40,083 | ||||||
|
2019
|
- | 49,834 | ||||||
|
2020
|
- | 8,666 | ||||||
| $ | 20,560 | $ | 150,583 | |||||
(1)The Company’s lease for its Athens corporate office expired February 2017 and is currently under negotiation for extension. The Company has estimated lease fees payable for the 2017 fiscal year based on its existing rental rate on a month to month basis.
Note 8 - LOAN
On June 11, 2015 and February 10, 2016, the Company entered into loan agreements with Mr. Leontaritis, the President of the Company, pursuant to which the Company received $163,595 (EUR €150,000) and $44,925 (EUR€40,000 as advances (collectively the “Notes”). The Notes earn simple interest accruing at five percent (5%) per annum and are due and payable within five years from the date of the agreement (“Maturity Date”).
During the year ended December 31, 2016 we accrued interest totaling $3,598 in respect of these loans.
During the year ended December 31, 2016, the Company repaid USD $222,822 (EUR €197,770) in cash in full settlement of the above notes, including accrued interest expense of USD $8,750 (EUR €7,770).
Note 9 - COMMITMENTS
|
(1)
|
Consulting Agreement with Kapatos
|
On April 16, 2014, the Company entered into a Consulting Agreement with Kapatos whereby he will provide his services as Chief Technical Officer for the Company and the Company’s wholly-owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be forty thousand Euros (€40,000) (USD$42,150) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Kapatos shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million US dollars ($20,000,000 USD).
On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Dr. Christos Kapatos. Further on August 2, 2016 the Company approved a further one-year extension so that the agreement will expire April 16, 2017.
F-15
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 9 - COMMITMENTS(continued)
|
(2)
|
Advisors
|
On April 15, 2014, the Company established a scientific advisory board whereby the Company intends to appoint certain advisors that can contribute to the Company’s overall business strategy and future direction. On April 16, 2014 the Company entered into Consulting Agreements with the members of its Scientific Advisory Board as follows:
|
-
|
Professor Stephan Solomonidis (“Solomonidis”) will provide services as Head of Research for the Company and the Company’s wholly owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be twenty thousand pounds (£20,000 GBP) (USD$24,610) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Solomonidis’ fee will be readjusted to the amount of forty thousand pounds (£40,000 GBP) (USD$49,220) per annum, and he shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million dollars ($20,000,000 USD).
On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Professor Stephan Solomonidis. The consulting agreement entered into with Professor Stephan Solomonidis was not renewed upon its expiration.
|
|
-
|
Professor William Sandham (“Sandham”) will provide services as Director of Diabetes Technology Research for the Company and the Company’s wholly owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be Twenty Thousand pounds (£20,000 GBP) (USD$24,610) per year payable in equal monthly installments beginning on May 1, 2014. In the case that a research and development project is initiated and completed during the term of the agreement, Sandham’s fee will be readjusted to the amount of forty thousand pounds (£40,000 GBP) (USD$49,220) per annum, and he shall receive one million four hundred thousand (1,400,000) shares of the Company’s common stock for each research project completed with a valuation less than twenty million dollars ($20,000,000 USD), three million five hundred thousand (3,500,000) shares of the Company’s common stock for each research project completed with a valuation equal or greater than twenty million dollars ($20,000,000 USD).
On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Professor William Sandham. The consulting agreement entered into with Professor William Sandham was not renewed upon its expiration.
|
|
3)
|
Employment Agreement with Heleen Kist
|
On November 7, 2014, the Company’s subsidiary, HCi Viocare Clinics entered into a service agreement with Mrs. Heleen Francoise Kist (the “Agreement”) whereby she will provide her services as Chief Operating Officer (“COO”) of the Company. The contract has a term of one year, being effective as of October 1, 2014, and ending on September 30, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation was thirty thousand GBP (£30,000) (USD$36,910) per year payable monthly in arrears on the last Friday of every month by credit transfer. On October 15, 2015, the Company’s COO, Heleen Kist and wholly owned subsidiary, HCI Viocare Clinics UK, signed an Addendum to an employment contract to extend the term to September 30, 2016, with a remuneration increase to forty-five thousand GBP (£45,000) (USD$ 55,370) per year.
On August 30, 2016 the Board of Directors of the Company approved the termination of the services agreement of Mrs. Heleen Francoise Kist, Chief Operating Officer of the Company following her resignation from any and all positions with the Company and its subsidiaries.
On August 30, 2016 the Board of Directors of the Company approved the repurchase from Mrs Heleen Kist of a total of seven hundred thousand (700,000) shares of the Company at a purchase price of US$0.02 per share for total of fourteen thousand (US$14,000) USD for return to treasury and cancellation. The $14,000 was recorded as accounts payable on the balance sheet as of December 31, 2016.
F-16
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 9 - COMMITMENTS(continued)
|
(4)
|
Consulting Agreement with Hellenic American Securities S.A.
|
On May 7, 2015, the Company entered into an Agreement with HELLENIC AMERICAN SECURITIES S.A. (“HAS S.A.”) for the provision of services to firms regarding their capital structure, industry strategy and similar matters (the “Agreement”) HAS S.A. is a Public Limited Stock Brokerage Company, member of the Athens Stock Exchange and Athens Derivative Exchange incorporated in Greece with registered office in Athens.
According to the Agreement which carries a term of one year, HAS S.A. shall provide advice to the Company regarding its capital structure, industry strategy and similar matters, as well as advice and services regarding mergers and acquisitions under the Greek Law, an introduction of the Company’s securities to Greek and Foreign Investors, both Private and Institutional ones as well as preparation of analyses for the Company which will be circulated to various websites and media groups.
HAS S.A. is entitled to the following consideration under the Agreement:
|
1.
|
$3,000 per month or its current equivalent in Euro, plus applicable VAT, payable monthly on the first day of the month, for each quarter;
|
|
2.
|
Issuance of 168,000 restricted shares of the Company’s common stock quarterly (the “Shares”), payable in monthly installments as to 56,000 shares per month, to be issued and delivered within 10 days after the 1st of each month.
|
As at August 1, 2016 a total of 560,000 shares had not yet been issued (December 31, 2015 – 336,000 shares) and are reflected on the Company’s balance sheet in current liabilities as Liability for Unissued Shares, in the amount of $436,640 (December 31, 2015 - $249,040). In addition, a total of $33,000 accrued for monthly service fees are included in accounts payable (December 31, 2015 - $24,000). The contract expired and on July 6, 2016 at which time the Company and HAS S.A. entered into a Mutual Release and Waiver of Compensation Agreement where under HAS S.A. has agreed to waive all remaining compensation payable under the terms of the original Agreement in the total amount of $33,000 including 560,000 shares of common stock which remained unissued. The Company recorded a gain of $469,640 against consulting fees due to this debt extinguishment.
|
(5)
|
Consulting Agreement with Sergios Katsaros
|
On September 1, 2015, the Board of Directors of the Company approved a consulting agreement with Sergios Katsaros and appointed Mr. Katsaros Vice President of HCi Viocare. Under the terms of the consulting agreement, Mr. Katsaros will work directly with the Company’s President and CEO in order to create and implement the Company’s strategic plan and assist in securing additional financing to meet the needs of the Company’s business plan and corporate objectives. The initial term of the contract is six months and Mr. Katsaros will receive compensation of EUR€2,000 (USD$2,100) per month. On March 1, 2016, the Company approved a one-year extension to the consulting agreement (the “Agreement”) between the Company and its Vice - President, Sergios Katsaros, originally entered into on September 1, 2015.
|
(6)
|
Financing agreement with LXM Finance LLP
|
On February 2, 2016, the Company entered into a three month Agreement with LXM Finance LLP (“LXM”), a company having a registered office in London, UK. Under the terms of the Agreement, LXM shall assist the Company with its proposed raising of funds (in the amount of up to fifty million Euros (50,000,000 €), through introducing potential investors to the Company in order for an increase in the Company’s share capital, debt financing or by a combination thereof to be effected.
LXM is entitled to the following consideration under the Agreement:
F-17
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 9 - COMMITMENTS(continued)
|
(6)
|
Financing agreement with LXM Finance LLP (continued)
|
|
(a)
|
a Retainer Fee of 12,500 Euros per month, payable in arrears; and,
|
|
(b)
|
a Success Fee of 7% of the Investment Amount(s) accepted and closed on by the Company by any Introduced Investor; the Success Fee shall be payable to LXM Finance in cleared funds (to an account notified in writing by LXM Finance to the Company for such purpose) within 10 Business days after the date on which such funds are accepted and closed on by the Company by such Introduced Investor and irrespective of whether or not the Engagement Letter has been terminated as at such date of investment or commitment
|
Notwithstanding the above, LXM Finance shall be entitled (in its sole discretion), to opt (by way of written notice to the Company) to receive up to 100% of the amount of any Success Fee in the form of securities in the Company in lieu of any cash payment subject to the Company’s agreement in writing, in compliance with all applicable securities rules and regulations, without prejudice to the Company’s right to offer cash form of payment.
On August 1, 2016, the Company entered into an Extension and Amendment of Engagement Letter originally entered into on February 2, 2016 with LXM. As per the amended terms of the Extension and Amendment of Engagement Letter, the requirement for a monthly retainer fee is deleted and all prior fees accrued to the date of the amendment remain due and payable; LXM shall provide services in order to assist the Company with fund raising for a term of one (1) year as of August 1, 2016, which may be renewed subject to a written agreement of the parties. LXM shall also provide services for the possible sale of Company assets and/or parts of its business, in which case it shall receive a sale fee of 7% of the purchase amount. All other terms and conditions of the Engagement Letter of February 8, 2016, remain valid and in force.
As of December 31, 2016, $13,170 (12,500 euros) have been paid, and a further $26,340 (25,000 euros) has been accrued in respect of this agreement.
|
(7)
|
Agreement with Renell Wertpapierhandelsbank AG (“Renell”)
|
On April 22, 2016, the Company entered into an agreement with Renell Wertpapierhandelsbank AG (“Renell”) with respect to the inclusion and trading of all Company shares in the open market at the Berlin Stock Exchange in Germany. As consideration for the services rendered Renell shall receive a fee of EUR10,000 and EUR3,500 annually thereafter as listing fees. The agreement was terminated during fiscal 2016 when the Company was unable to successfully list on the Berlin Stock Exchange. No further fees are payable under this agreement.
|
(8)
|
Professional Relations and Consulting Agreement with Acorn Management Partners, LLC (“Acorn”)
|
On May 18, 2016, the Company entered into a Professional Relations and Consulting Agreement with Acorn Management Partners, LLC (“Acorn”), a Georgia limited liability company, for a term of 12 months. Under the terms of the agreement, Acorn will create a market awareness program and introduce financial professionals to the Company targeting the US marketplace. The program shall be designed to build long-term relationships between the Company and licensed financial professionals. In consideration of the Services, the Company agrees to pay to Acorn the following fees:
|
·
|
First 6 Month period
|
|
-
|
Cash: $17,500 per month, monthly payment made upon the execution of the contract
|
|
-
|
Stock: $250,000 dollars’ worth of restricted 144 common stock of the Company (“VICA”), due upon the execution of contract. 312,500 shares were issued on May 20, 2016 and was determined by dividing $250,000 by the closing price on the last trading day’s closing price on the day before the execution day.
|
|
·
|
Second 6 Month period
|
|
-
|
Cash: $17,500 per month, monthly payment made upon the execution of the contract
|
|
-
|
Stock: $250,000 dollars’ worth of restricted 144 common stock of the Company (“VICA”), due upon the execution of contract. The number of the shares to be determined by dividing $250,000 by the closing price on the last trading day’s closing price on the day before the execution day.
|
As of December 31, 2016, $30,500 has been paid, and a further $74,500 has been accrued in respect of this agreement relative to the first six month term. Prior to conclusion of the first six months the contract was terminated by mutual agreement of both parties and as at December 31, 2016 the Company had entered negotiations to settle remaining amounts payable.
|
(9)
|
Services Agreement with KCN Ltd.
|
On November 3, 2016, the Company entered into a services agreement with a company under the name KCN Ltd. for the introduction of the Company to potential investors in the Middle East and specifically in Saudi Arabia in view of the Company project to expand its business and establish three (3) Prosthetic and Orthotic Clinics in the region. The services agreement has a six-month term ending on May 2, 2017, and the Consultant shall be remunerated with a success fee of 2% of the investment amount.
F-18
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 10 - RELATED PARTY TRANSACTIONS
The following table provides details of the Company’s related party transactions during the year ended December 31, 2016 and 2015:
(a) Services provided from related parties:
|
Year Ended
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Consulting fees from CEO and President (i)
|
$
|
60,000
|
$
|
60,000
|
||||
|
Consulting fees from a Director (ii)
|
44,266
|
44,380
|
||||||
|
Professional fees from Director (iii)
|
13,280
|
13,380
|
||||||
|
Consulting fees for VP (iv)
|
26,560
|
8,876
|
||||||
|
Consulting fees for COO (v)
|
36,529
|
51,582
|
||||||
|
Stock-based compensation (VP) (iv)
|
169,000
|
-
|
||||||
|
Stock award granted to Director (iii)
|
-
|
450,000
|
||||||
|
$
|
349,635
|
$
|
628,218
|
|||||
|
(i)
|
On September 10, 2013 Mr. Leontaritis was appointed President. On January 15, 2014, the Board of Directors of the Company approved the execution of a consulting agreement between the Company and Sotirios Leontaritis (“Leontaritis”), whereby Leontaritis shall provide services to the Company as the Company’s President and Chief Executive Officer in regards to the Company’s management and operations for the period from January 1, 2014 to December 31, 2017. Under the terms of the agreement, the Company agreed to pay to Leontaritis US$60,000 per annum payable in monthly payments of US$5,000 a month for the term of the contract.
|
|
|
(ii)
|
On September 30, 2013, the Board of Directors of the Company appointed Dr. Christos Kapatos as a director of the Company. During fiscal 2016 and fiscal 2015 Dr. Kapatos invoiced the Company GBP$40,000 for services rendered in each fiscal year respectively.
|
|
|
(iii)
|
On September 10, 2013, the Board of Directors of the Company elected Nikolaos Kardaras as Secretary and a director of the Company. On May 28, 2015, the Company approved the issuance of 700,000 common shares as compensation for prior services provided by director Nikolaos Kardaras in the form of fully vested stock awards. During fiscal 2016 and fiscal 2015 Mr. Kardaras invoiced the Company EUR $12,000 for services rendered in his capactiy as a director in each fiscal year respectively.
|
|
(iv)
|
On September 1, 2015, the Board of Directors of the Company approved a consulting agreement with Sergios Katsaros and appointed Mr. Katsaros Vice President of HCi Viocare. Under the terms of the consulting agreement, the initial term of the contract is six months and Mr. Katsaros will receive compensation of EUR€2,000 (USD$2,180) per month.
On March 1, 2016, the Company approved a one-year extension to the consulting agreement. Concurrently, the Company approved the grant of a stock award of 300,000 common shares as compensation for the services provided by Vice President, Sergios Katsaros. The award vests in three equal instalments of 100,000 shares as of the date of grant, the six-month anniversary of the date of grant and the 12-month anniversary of date of grant. During fiscal 2016 a total of 200,000 awards were fully vested with a fair market value of $169,000.
|
|
(v)
|
On November 7, 2014, the Company’s subsidiary, HCi Viocare Clinics (formerly W.D. Spence Prosthetics Limited) entered into a service agreement with Mrs. Heleen Francoise Kist (the “Agreement”) whereby she will provide her services as Chief Operating Officer (“COO”) of the Company. The contract has a term of one year, being effective as of October 1, 2014, and ending on September 30, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation shall be thirty thousand GBP (£30,000) (USD$43,180) per year payable monthly in arrears on the last Friday of every month by credit transfer.
On October 15, 2015, the Company’s COO, Heleen Kist and wholly owned subsidiary, HCI Viocare Clinics UK, signed an Addendum to an employment contract to extend the term to September 30, 2016, with a remuneration increase to forty-five thousand GBP (£45,000) (USD$ 64,800) per year.
On August 30, 2016, the Board of Directors of the Company approved the termination of the services agreement of Mrs. Heleen Francoise Kist, Chief Operation Officer of the Company following her resignation from any and all positions with the Company and its subsidiaries.
|
F-19
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 10 - RELATED PARTY TRANSACTIONS (continued)
(b) Accounts payable and accrued liabilities from related parties:
|
Balance,
December 31,
2015
($)
|
Services provided
during the period
($)
|
Reimbursement on Company’s expenses
and interest on loan
($)
|
Payments
($)
|
Foreign exchange
($)
|
Balance,
December 31,
2016
($)
|
||||||||||||||||
|
Consulting fees from CEO and President (i)
|
293,881
|
60,000
|
131,025
|
(8,750
|
)
|
(10,058
|
)
|
466,098
|
|||||||||||||
|
Consulting fees from a Director (ii)
|
-
|
44,266
|
-
|
(2,117
|
)
|
42,149
|
|||||||||||||||
|
Professional fees from Director (iii)
|
1,091
|
13,280
|
-
|
(13,319
|
)
|
-
|
1,052
|
||||||||||||||
|
Consulting fees for VP (iv)
|
-
|
26,560
|
-
|
(26,560
|
)
|
-
|
-
|
||||||||||||||
|
Consulting fees for COO (v)
|
-
|
36,529
|
-
|
(36,529
|
)
|
-
|
-
|
||||||||||||||
|
294,972
|
180,635
|
131,025
|
(85,158
|
)
|
(12,175
|
)
|
509,299
|
||||||||||||||
(c) Advances from related parties:
|
Balance, December 31, 2015
($)
|
Addition
($)
|
Assignment
& Assumption
($)
|
Payment
($)
|
Foreign exchange
($)
|
Balance, December 31, 2016
($)
|
|||||||||||||||||||
|
CEO and President (i)
|
615,898 | 466,925 | (109,079 | ) | (619,187 | ) | (38,480 | ) | 316,077 | |||||||||||||||
|
Director (ii)
|
- | 109,079 | (40,076 | ) | (1,916 | ) | 67,087 | |||||||||||||||||
|
COO (v)
|
33,522 | (32,346 | ) | (1,176 | ) | - | ||||||||||||||||||
| 649,420 | 466,925 | - | (691,609 | ) | (41,572 | ) | 383,164 | |||||||||||||||||
(ii) During January 2016, Christos Kapatos, director and CTO, entered into a private agreement for the assignment and assumption of certain advances payable with Sotirios Leontaritis, President and CEO, in respect of an amount totalling EUR$100,000 (US$109,079). During the year ended December 2016 amounts totaling $40,076 were repaid to Dr. Kapatos leaving a balance of $67,087 due and payable as at December 31, 2016.
(d) Loan Agreement from related parties (ref Note 8 - Loan)
|
Balance,
December 31, 2015
($)
|
Addition
($)
|
Payment
($)
|
Foreign Exchange on the Note
($)
|
Balance,
December 31, 2016
($)
|
||||||||||||||||
|
CEO and President (i)
|
163,595
|
44,925
|
(214,072
|
)
|
5,552
|
-
|
||||||||||||||
Note 11 - CAPITAL STOCK
The Articles of Incorporation authorize the Company to issue 5,000,000 shares of Series A preferred stock with a par value of $0.0001, and 700,000,000 shares of common stock with a par value of $0.0001. No shares of preferred stock have been issued, however, the Company has granted 25,000 fully vested stock options for the purchase of 25,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from the date of vesting. (ref: Note 14 below).
On August 8, 2015 FINRA approved a seven (7) new for one (1) old forward split of our authorized and issued and outstanding shares of common. A Certificate of Change for the stock split was filed and became effective with the Nevada Secretary of State on August 7, 2015. Consequently, our authorized share capital increases from 100,000,000 to 700,000,000 shares of common stock and our issued and outstanding common stock increases accordingly, all with a par value of $0.0001. Our preferred stock remains unchanged.
F-20
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 11 - CAPITAL STOCK (continued)
Share issuances during the year ended December 31, 2016:
On January 7, 2016, the Company entered into two Private Placement Subscription Agreements with two natural persons. Under the terms of the Agreements the Individuals subscribed for a cumulative total of 70,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$35,000. The shares are subject to applicable resale restrictions.
On February 1, 2016, the Company entered into a consulting agreement with Sitimo Ltd. Under the terms of the Agreement, Consultant shall provide consulting services to the Company for the development and expansion of its business and establishment of Prosthetics and Orthotics clinics in the region of South Eastern Europe as well as the Middle East. The Consultant undertakes to introduce the Company to the healthcare market of South Eastern Europe and Middle East. The Agreement has a term of twelve (12) months and the Consultant is entitled to remuneration for the provision of services in the form of 100,000 shares of the common stock to be issued within thirty days from the enforcement of the Agreement. As at March 31, 2016 100,000 shares have been issued in accordance with the terms and valued at $85,000 the fair market value on agreement date, which amount has been expensed as consulting fees.
On February 10, 2016, the Company entered into a consulting agreement with an individual. Under the terms and conditions of the Agreement the Consultant shall provide consulting services and introduce the Company to the healthcare market of Spain in view of the Company's plans to expand its clinics business and establish a Prosthetic and Orthotic Clinic in Spain. The Agreement has a six-month term ending on August 2, 2016, and the Consultant shall be remunerated with 50,000 shares of the Company's common stock. As at March 31, 2016 50,000 shares have been issued in accordance with the terms and valued at $42,250 the fair market value on agreement date, which amount has been expensed as consulting fees.
On February 17, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreement the Individual subscribed for a total of 3,625 shares of the Company’s common stock at a purchase price of US$0.40 per share for total cash proceeds of US$1,450. The shares are subject to applicable resale restrictions.
On February 18, 2016, the Company entered into a consulting agreement with an individual. Under the terms and conditions of the Agreement the Consultant shall provide consulting services and introduce the Company to the healthcare market of Middle East and specifically of Saudi Arabia in view of the Company project to expand its business and establish a Prosthetic and Orthotic Clinic in the Middle East. The Agreement has a six-month term ending on August 18, 2016 and the Consultant shall be remunerated with 200,000 shares of the common stock. As at March 31, 2016 200,000 shares have been issued in accordance with the terms and valued at $169,000 the fair market value on agreement date, which amount has been expensed as consulting fees.
On February 19, 2016, the Company entered into a consulting agreement with an individual. Under the terms and conditions of the Agreement the Consultant shall provide consulting services and introduce the Company to the healthcare market of Turkey in view of the Company project to expand its business and establish a Prosthetic and Orthotic Clinic in Turkey. The Agreement has a six-month term ending on August 19, 2016 and the Consultant shall be remunerated with 200,000 shares of the common stock. As at March 31, 2016 200,000 shares have been issued in accordance with the terms and valued at $169,000 the fair market value on agreement date, which amount has been expensed as consulting fees.
On March 1, 2016, the Company approved the grant of a stock award of 300,000 common shares as compensation for the services provided by Vice President, Sergios Katsaros. The award vests in three equal instalments of 100,000 shares as of the date of grant, the six-month anniversary of the date of grant and the 12-month anniversary of date of grant. On March 1, 2016 100,000 shares have been issued in accordance with the terms of the award valued at $84,500, the fair market value on grant date, which amount has been expensed as stock based compensation. On October 1, 2016 100,000 shares have been issued in accordance with the terms of the award valued at $97,500, the fair market value on grant date, which amount has been expensed as stock based compensation.
F-21
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 11 - CAPITAL STOCK (continued)
Share issuances during the year ended December 31, 2016: (continued)
On March 2, 2016, the Company entered into Private Placement Subscription Agreements with two individuals. Under the terms of the Agreements the Individuals each subscribed for a total of 50,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cumulative cash proceeds of US$50,000. The shares are subject to applicable resale restrictions.
On March 8, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreement the Individual subscribed for a total of 240,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$120,000. The shares are subject to applicable resale restrictions.
On March 9, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreement the Individual subscribed each for a total of 54,875 shares of the Company’s common stock at a purchase price of US$0.40 per share for cash proceeds of US$21,950. The shares are subject to applicable resale restrictions.
On March 22, 2016, the Company approved the issuance of 50,000 common shares for services provided by Mr. Mikulas Dylowicz, consultant, in the form of stock awards which shall vest as of the date of grant. The shares were valued at the fair market value on the date of grant totaling $46,480, which amount has been expensed as stock based compensation.
On April 4, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreement the Individual subscribed for a total of 400,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$200,000. The shares are subject to applicable resale restrictions.
On May 20, 2016, the Company entered into Private Placement Subscription Agreements with two individuals. Under the terms of the Agreements the Individuals subscribed for a total of 35,355 shares of the Company’s common stock at a purchase price of US$0.60 per share for total cash proceeds of US$21,213. The shares are subject to applicable resale restrictions.
On May 20, 2016, the Company issued 312,500 common shares for services provided by Acorn Management Partners LLC under the professional relations and consulting agreement. The number of the shares to be determined by dividing $250,000 by the closing price on the last trading day’s closing price on the day before the execution day.
On July 7, 2016, the Company entered into a Private Placement Subscription Agreement (the “Agreement”) with an individual (the “Individual”). Under the terms of the Agreement the Individual subscribed for a total of 31,320 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$15,660. The shares are subject to applicable resale restrictions.
On July 7, 2016, the Company entered into a Private Placement Subscription Agreement (the “Agreement”) with an individual (the “Individual”). Under the terms of the Agreement the Individual subscribed each for a total of 24,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for cash proceeds of US$12,000. The shares are subject to applicable resale restrictions.
F-22
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 11 - CAPITAL STOCK (continued)
Share issuances during the year ended December 31, 2016: (continued)
On July 14, 2016, the Company entered into a Private Placement Subscription Agreement (the “Agreement”) with an individual (the “Individual”). Under the terms of the Agreement the Individual subscribed for a total of 33,468 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$16,734. The shares are subject to applicable resale restrictions.
On July 22, 2016, the Company entered into a Private Placement Subscription Agreement (the “Agreement”) with an individual (the “Individual”). Under the terms of the Agreement the Individual subscribed for a total of 8,000 shares of the Company’s common stock at a purchase price of US$0.69 per share for total cash proceeds of US$5,500. The shares are subject to applicable resale restrictions.
On August 30, the Board of Directors of the Company approved the repurchase from Mrs Heleen Kist of a total of seven hundred thousand (700,000) shares of the Company at a purchase price of US$0.02 per share for total of fourteen thousand (US$14,000) USD for return to treasury and cancellation.
On September 9, 2016, the Company entered into five Private Placement Subscription Agreements (the “Agreement”) with five individuals (the “Individual”). Under the terms of the Agreements the Individuals subscribed for a total of 420,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$210,000. The shares are subject to applicable resale restrictions.
On October 11, 2016, the Company entered into Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 40,000 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$20,000. The shares are subject to applicable resale restrictions
On October 24, 2016, the Company granted a total of 25,000 stock awards to various employees of its wholly owned UK subsidiaries with a market value of $1 per share. The shares are subject to applicable resale restrictions
On November 4, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 85,000 shares of the Company’s common stock at a purchase price of US$0.60 per share for total cash proceeds of US$51,000. The shares are subject to applicable resale restrictions
On November 18, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 66,300 shares of the Company’s common stock at a purchase price of US$0.80 per share for total cash proceeds of US$53,040. The shares are subject to applicable resale restrictions
On December 7, 2016, the Company entered into a Private Placement Subscription Agreement with an individual. Under the terms of the Agreements the Individual subscribed for a total of 21440 shares of the Company’s common stock at a purchase price of US$0.50 per share for total cash proceeds of US$10,720. The shares are subject to applicable resale restrictions
As at December 31, 2016 and December 31, 2015 the Company had a total of 192,814,844 and 190,743,961 shares issued and outstanding, respectively.
F-23
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 11 - CAPITAL STOCK (continued)
Liability for Unissued Shares
Under the terms of the contract with the Company, Hellenic American Securities S.A shall receive payments of 56,000 shares of Company’s common stock on each month from July 2015 to April 2016 respectively (ref Note 11(5)). As at June 30, 2016 a total of 560,000 shares (December 31, 2015 - 336,000 shares) had not yet been issued in the amount of $436,640 (December 31, 2015 - $249,040).
On August 1, 2016, the Company and HAS S.A. entered into a Mutual Release and Waiver of Compensation Agreement where under HAS S.A. has agreed to waive all further compensation payable under the terms of the original Agreement in the total amount of $436,640 liability for the unissued 560,000 shares of common stock.
Designation of Series A Preferred Stock
On January 13, 2014, the Company filed a Certificate of Designation with the Secretary of State of the State of Nevada. The Certificate of Designation sets forth the rights, preferences and privileges of a class of the Company’s preferred stock. Such class shall be designated as the “Series A Preferred Stock” and the number of shares constituting such series shall be 5,000,000 shares. The holders of Series A Preferred Stock will be entitled to a preference over all of the shares of the Company’s common stock. Holders of Series A Preferred Stock shall have 50 votes per share of Series A Preferred Stock held by them and shall be entitled to notice of any stockholders’ meeting and to vote as a single class upon any matter submitted to the stockholders for a vote. Each share of Series A Preferred Stock is convertible into 20 shares of our common stock at any time at the holder’s option. Shares of Series A Preferred Stock shall not be entitled to any dividends. The preferred stock shall be entitled to a preference over all of the shares of common stock of the Company with respect to the distribution of assets in the event of the liquidation, dissolution or winding up of the Company, whether voluntary or involuntary, or any other distribution of the assets of the Company among its shareholders for the purpose of winding up its affairs.
Note 12 - STOCK OPTIONS
On April 15, 2014, the Company appointed Professor Martha Lucia Zequera, Professor William Sandham, Professor Stephan Solomonidis and Doctor Christos Kapatos to the Advisory Board of the Company and entered into advisory board agreements with respect to services to be provided. Compensation for the members of the Advisory Board shall be the grant of a total of 5,000 stock options entitling the advisory board member the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from the date of vesting. The advisory board appointments are for a term of one year and the options granted under the agreements will vest on completion of the term of the appointment. All of the aforementioned stock options vested in April 2015. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
On June 9, 2014, the Company appointed Mr. William Spence to the Advisory Board of the Company and entered into an advisory board agreement with Mr. Spence. Compensation shall be the grant of a total of 5,000 stock options entitling Mr. Spence the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from the date of vesting. The advisory board appointment is for a term of one year and the options granted under the agreement will vest on completion of the term of the appointment. The aforementioned options vested upon the one-year anniversary of the date of appointment. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
The following table summarizes information concerning stock options outstanding as of December 31, 2016 and December 31, 2015:
F-24
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 12 - STOCK OPTIONS
|
December 31, 2016
|
December 31, 2015
|
|||||||||||||||
|
Series A Preferred stock
|
Weighted Average Exercise Price
$
|
Series A Preferred stock
|
Weighted Average Exercise Price
$
|
|||||||||||||
|
Outstanding at beginning of the year
|
25,000
|
0.04
|
25,000
|
0.04
|
||||||||||||
|
Granted
|
-
|
-
|
-
|
-
|
||||||||||||
|
Exercised
|
-
|
-
|
-
|
-
|
||||||||||||
|
Expired or cancelled
|
-
|
-
|
-
|
-
|
||||||||||||
|
Outstanding at the period
|
25,000
|
0.04
|
25,000
|
0.04
|
||||||||||||
|
Stock
Options
|
Weighted Average
Grant Date
Fair Value
|
|||||||
|
Unvested, end of December 31, 2014
|
25,000
|
$
|
0.04
|
|||||
|
Granted
|
-
|
|||||||
|
Vested
|
(25,000
|
)
|
-
|
|||||
|
Forfeited
|
-
|
-
|
||||||
|
Unvested, end of December 31, 2015
|
-
|
$
|
-
|
|||||
|
Unvested, end of September 30, 2016
|
-
|
$
|
-
|
|||||
Valuation Assumptions
The Company accounts for share-based payments pursuant to ASC 718, “Stock Compensation” and, accordingly, the Company records compensation expense for share-based awards based upon an assessment of the grant date fair value for stock options using the Black-Scholes option pricing model. The fair value of stock options under the Black-Scholes model requires management to make assumptions regarding projected employee stock option exercise behaviors, risk-free interest rates, volatility of the Company’s stock price and expected dividends.
Stock compensation expense for stock options is recognized over the vesting period of the award.
The following table presents the range of the weighted average fair value of options granted and the related assumptions used in the Black-Scholes model for stock option grants:
|
Options Granted
September 30, 2014
|
||||
|
Fair value of options granted
|
1.40 ~ 2.00
|
|||
|
Assumptions used:
|
||||
|
Expected life (years) (a)
|
1.00
|
|||
|
Risk free interest rate (b)
|
0.11
|
%
|
||
|
Volatility (c)
|
117.09 ~ 119.83
|
% | ||
|
Dividend yield (d)
|
0.00
|
%
|
||
|
a)
|
Expected life: The expected term of options granted is determined using the “shortcut” method allowed by SAB No.107. Under this approach, the expected term is presumed to be immediately after the vesting date at the end of the contractual term of one (1) year.
|
|
|
b)
|
Risk-free interest rate: The rate is based on the U.S. Treasury zero-coupon yield curve on the grant date for a maturity similar to the expected life of the options.
|
|
|
c)
|
Volatility: The expected volatility of the Company’s common stock is calculated by using the historical daily volatility of the Company’s stock price calculated over a period of time representative of the expected life of the options.
|
|
|
d)
|
Dividend yield: The dividend yield rate is not considered in the model, as the Company has not established a dividend policy for the stock.
|
F-25
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 13– PROVISION FOR INCOME TAXES:
The Company's operations in Greece have no tax free income threshold and are subject to taxation at a rate of 29% applied to all net income earned after $1 Euro. Further, after its first year of profitable operations, the Company is required to remit estimated income taxes prorated monthly based on the prior year's calculatd income taxes payable.
The Company's operations in the United Kingdom are subject to Corporation Tax at a single taxation rate of 20% calculated on net income. The Company's operations are subject to certain releif from taxation with respect to R&D credit claims and may be subject to "Patent Box" relief which provides for a lower rate of income tax payable on all licensing income generated from the corporate owned patents. There are numerous critera required to be met to qualify for such relief.
The Company has experienced losses since inception. As a result, it has incurred no Federal income tax. The Internal Revenue Code allows net operating losses (NOL's) to be carried forward and applied against future profits for a period of twenty years. The Greece Tax Code permits such carryforwards for a period of five years. The United Kingdom Tax Code permits such carryforwards indefinitely. The total of these NOL's at December 31, 2016 was $4,619,000 in the U.S., $1,244,000 in Greece and $306,000 in the U.K and at December 31, 2015 was $3,843,000 in the U.S., $800,840 in Greece and $453,700 in the U.K.
The provision for income taxes in US consists of the following:
|
Year ended
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Current operations
|
$
|
348,400
|
$
|
534,200
|
||||
|
Timing differences, Stock based compensation
|
(84,575
|
)
|
-
|
|||||
|
Less, Change in valuation allowance
|
(263,825
|
)
|
(534,200
|
)
|
||||
|
Net refundable amount
|
$
|
-
|
$
|
-
|
||||
The provision for income taxes in Greece consists of the following:
|
Year ended
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Current operations
|
$
|
128,670
|
$
|
92,000
|
||||
|
Less, Change in valuation allowance
|
(128,670
|
)
|
(92,000
|
)
|
||||
|
Net refundable amount
|
$
|
-
|
$
|
-
|
||||
The provision for income taxes in UK consists of the following:
|
Year ended
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Current operations
|
$
|
27,780
|
$
|
1,012,000
|
||||
|
Research and development
|
(57,180
|
)
|
(980,600
|
)
|
||||
|
Less, Change in valuation allowance
|
29,400
|
(31,400
|
)
|
|||||
|
Net refundable amount
|
$
|
-
|
$
|
-
|
||||
The cumulative tax effect at the expected rates in U.S., in Greece and in U.K. for significant items comprising our net deferred tax amounts as of December 31, 2016 and 2015 are as follows:
|
December 31, 2016
|
December 31, 2015
|
|||||||||||||||||||||||
|
US Expected rate 34%
|
Greece Expected rate 29%
|
UK Expected rate 20%
|
US Expected rate 34%
|
Greece Expected rate 26%
|
UK Expected rate 20%
|
|||||||||||||||||||
|
Deferred tax asset attributable to:
|
||||||||||||||||||||||||
|
Net operating loss carryover
|
$
|
4,619,000
|
$
|
1,244,000
|
$
|
306,000
|
$
|
2,403,548
|
$
|
231,800
|
$
|
226,800
|
||||||||||||
|
Less, Valuation allowance
|
(4,619,000
|
)
|
(1,244,000
|
)
|
(306,000
|
)
|
(2,403,548
|
)
|
(231,800
|
)
|
(226,800
|
)
|
||||||||||||
|
Net deferred tax asset
|
$
|
-
|
$
|
$
|
-
|
$
|
-
|
$
|
-
|
$
|
-
|
|||||||||||||
F-26
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 13 – PROVISION FOR INCOME TAXES: (continued)
The Company has no tax position at December 31, 2016 and 2015 for which the ultimate deductibility is highly certain but for which there is uncertainty about the timing of such deductibility. The Company recognizes interest accrued related to unrecognized tax benefits in interest expense and penalties in operating expenses. No such interest or penalties were recognized during the period presented. The Company had no accruals for interest and penalties at December 31, 2016 and 2015. The Company’s utilization of any net operating loss carry forward may be unlikely as a result of its intended activities.
The Company recognized deferred tax assets and liabilities for both the expected impact of differences between the financial statements and the tax basis of assets and liabilities, and for the expected future tax benefit to be derived from tax losses and tax credit carryforwards. The Company will establish a valuation allowance to reflect the likelihood of realization of deferred tax assets.
Note 14 - SEGMENT REPORTING
The Company’s operations are classified into two reportable segments that provide different products or services. Separate management of each segment is required because each business unit is subject to different marketing, operations, and growth and technology development strategies.
The Clinics segment derives its revenue from provision of services at our P&O Clinic located in Glasgow, Scotland. The Technology segment derives its income from the licensing of its proprietary technologies and ultimately recurring royalty income as well as technology access fees. Prsently we are only deriving income from our UK-based operations. We expect this to be the case until such time as we are able to expand our chain of P&O Clinics.
There are no inter-segment sales however, the Company’s two primary operating segments do share cost on certain operational overheads including facility rent and staff salaries.
|
Clinics
(UK)
|
Technology
(UK)
|
All Other
(Greece)
|
Total
|
|||||||||||||
|
Revenue
|
$ | 575,001 | $ | 112,447 | $ | - | $ | 687,448 | ||||||||
|
Depreciation & amortization
|
6,061 | 35,730 | 25,856 | 67,647 | ||||||||||||
|
Loss from operations
|
(23,735 | ) | (115,151 | ) | $ | (1,466,686 | ) | $ | (1,605,572 | ) | ||||||
|
Interest expenses
|
- | - | $ | (3,982 | ) | $ | (3,982 | ) | ||||||||
|
Income tax expense
|
11,200 | - | $ | - | $ | 11,200 | ||||||||||
|
Assets
|
45,034 | 173,927 | $ | 100,329 | $ | 319,290 | ||||||||||
|
Expenditure on long-lived assets
|
$ | - | $ | - | $ | - | $ | - | ||||||||
F-27
HCi Viocare
Notes to Audited Consolidated Financial Statements
Note 15 - SUBSEQUENT EVENTS
On January 1, 2017, the Company approved a three-year extension to the consulting agreement between the Company and its President & CEO and controlling shareholder, Sotirios Leontaritis, originally entered into on January 1, 2014.
The revision to the Agreement ("Addendum No. 1") has a term of three years, being effective as of January 1, 2017, and ending on December 31, 2019, renewable for such further term as may be mutually agreed between the parties. As per Addendum No. 1 the Company shall pay to Leontaritis US$120,000 per annum payable in monthly payments of US$10,000 a month for the term of the contract. Further, under the terms of the Addendum No. 1, Leontaritis is entitled to acquire at his discretion 3,000,000 shares of the common stock at a price of $0.30 per share for a term of five (5) years.
On March 1, 2017, the Company approved a one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros, originally entered into on September 1, 2015. The revision to the Agreement ("Addendum No. 2") has a term of one year, being effective as of March 1, 2017, and ending on February 28, 2018, renewable for such further term as may be mutually agreed between the parties. All other terms and conditions remain valid and in force. Concurrrently the final 100,000 shares issuable under a stock award originally granted on March 1, 2016 was issued.
On March 3, 2017, the Company approved the issuance of 1,000,000 common shares for the services provided by Mr. Nikolaos Kardaras, Director of the Company, in the form of stock award which shall vest as of the date of grant.
On March 6, 2017, the Company approved the issuance of 50,000 common shares for the services provided by a consultant in the form of stock award which shall vest as of the date of grant.
On March 7, 2017, the Company approved the issuance of 50,000 common shares for the services provided a consultant in the form of stock award which shall vest as of the date of grant.
On March 17, 2017, the Company approved the issuance of 1,000,000 common shares for the services provided by Mr. Sergios Katsaros, Vice-president, officer of the Company, in the form of stock award which shall vest as of the date of grant.
On March 22, 2017, the Company approved the issuance of 50,000 common shares for the services provided by Mr. Brian Maguire, Director of HCi Viocare Clinics UK Ltd., subsidiary of the Company, in the form of stock award which shall vest as of the date of grant.
F-28
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
Evaluation of Disclosure Controls and Procedures
Our management, under supervision and with the participation of the Company’s Principal Executive Officer and Principal Financial Officer, evaluated the effectiveness of our disclosure controls and procedures, as defined under Exchange Act Rule 13a-15(e). Based upon this evaluation, the Principal Executive Officer and Principal Financial Officer concluded that, as of December 31, 2016, because of the material weakness in our internal control over financial reporting (“ICFR”) described below, our disclosure controls and procedures were not effective.
Disclosure controls and procedures are controls and other procedures that are designed to ensure that required information to be disclosed in our reports filed or submitted under the Securities Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that required information to be disclosed in our reports filed under the Exchange Act is accumulated and communicated to our management, including our principal executive officer and our principal financial officer, as appropriate, to allow timely decisions regarding required disclosure.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined under Exchange Act Rules 13a-15(f) and 14d-14(f). Our internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles.
All internal control systems, no matter how well designed, have inherent limitations and may not prevent or detect misstatements. Therefore, even those systems determined to be effective can only provide reasonable assurance with respect to financial reporting reliability and financial statement preparation and presentation. In addition, projections of any evaluation of effectiveness to future periods are subject to risk that controls become inadequate because of changes in conditions and that the degree of compliance with the policies or procedures may deteriorate.
Management assessed the effectiveness of our internal control over financial reporting as of December 31, 2016. In making the assessment, management used the criteria issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework 2013. Based on its assessment, management concluded that, as of December 31, 2016, our internal control over financial reporting was not effective and that material weaknesses in ICFR existed as more fully described below.
49
As defined by Auditing Standard No. 5, “An Audit of Internal Control Over Financial Reporting that is Integrated with an Audit of Financial Statements” established by the Public Company Accounting Oversight Board (“PCAOB”), a material weakness is a deficiency or combination of deficiencies that results in more than a remote likelihood that a material misstatement of annual or interim financial statements will not be prevented or detected. In connection with the assessment described above, management identified the following control deficiencies that represent material weaknesses as of December 31, 2016:
|
1)
|
Lack of an independent audit committee or audit committee financial expert, and no independent directors. We do not have any members of the Board who are independent directors and we do not have an audit committee. These factors may be counter to corporate governance practices as defined by the various stock exchanges and may lead to less supervision over management;
|
|
2)
|
Insufficient written policies and procedures for accounting and financial reporting with respect to the requirements and application of US GAAP and SEC disclosure requirements;
|
Management's Remediation Initiatives
As of December 31, 2016, management assessed the effectiveness of our internal control over financial reporting. Based on that evaluation, it was concluded that during the period covered by this report, the internal controls and procedures were not effective due to deficiencies that existed in the design or operation of our internal controls over financial reporting. However, management believes these weaknesses did not have an effect on our financial results. During the course of our evaluation, we did not discover any fraud involving management or any other personnel who play a significant role in our disclosure controls and procedures or internal controls over financial reporting.
Due to a lack of financial and personnel resources, we are not able to, and do not intend to, immediately take any action to remediate these material weaknesses. We will not be able to do so until, if ever, we acquire sufficient financing and staff to do so. We will implement further controls as circumstances, cash flow, and working capital permits. Notwithstanding the assessment that our ICFR was not effective and that there were material weaknesses as identified in this report, we believe that our financial statements contained in our Annual Report on Form 10-K for the period ended December 31, 2016, fairly presents our financial position, results of operations, and cash flows for the periods covered, as identified, in all material respects.
Management believes that the material weaknesses set forth above were the result of the scale of our operations and intrinsic to our small size. Management also believes that these weaknesses did not have an effect on our financial results.
This Annual Report does not include an attestation report of the Company’s registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by the Company’s registered public accounting firm pursuant to the rules of the Securities and Exchange Commission that permit the Company to provide only management’s report in this annual report.
Changes in Internal Control over Financial Reporting
During the period covered by this report, there were no changes (including corrective actions with regard to significant deficiencies or material weaknesses) in our internal controls over financial reporting that occurred that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
None.
50
PART III
Executive Officer and Directors
The following individuals serve as the directors and executive officers of our Company as of the date of this annual report.
|
Name
|
Position Held with Our Company
|
Age
|
Date First Elected or Appointed
|
|
Sotirios Leontaritis
|
Chief Executive Officer, Chief Financial Officer, President, Treasurer, and Director
|
56
|
September 10, 2013
|
|
Nikolaos Kardaras
|
Secretary and Director
|
66
|
September 10, 2013
|
|
Christos Kapatos
|
Director, Chief Technical Officer
|
39
|
September 30, 2013
|
|
Yiannis Levantis
|
Director
|
41
|
July 18, 2015
|
|
Sergios Katsaros
|
Vice President
|
41
|
September 1, 2015
|
Our Board of Directors consists of only one class. All of the directors will serve until the next annual meeting of stockholders; until their successors are elected and qualified; or until their earlier death, retirement, resignation or removal. There are no family relationships among directors and executive officers. There are no arrangements or understandings between any director or executive officer and any other person(s) pursuant to which a director or executive officer was selected. Our executive officers are elected by our Board of Directors and hold office until their death, resignation or removal from office.
Our Board of Directors believes that the Board and our executive officer possesses a range of talent, skill, and experience sufficient to provide sound and prudent guidance with respect to our operations and interests. The information below with respect to our sole director and officer includes the individual’s experience, qualifications, attributes, and skills that led our Board of Directors to the conclusion that he or she should serve as a director and/or executive officer.
We know of no pending proceedings to which any director, member of senior management, or affiliate is either a party adverse to us, or our subsidiaries, or has a material interest adverse to us or our subsidiaries.
None of our executive officers or directors have been involved in any bankruptcy proceedings within the last five years, been convicted in or has pending any criminal proceedings, been subject to any order, judgment or decree enjoining, barring, suspending or otherwise limiting involvement in any type of business, securities or banking activity or been found to have violated any federal or state securities or commodities law.
51
Business Experience
The following is a brief account of the education and business experience during at least the past five years of each director, executive officer and key employee of our company, indicating the person’s principal occupation during that period, and the name and principal business of the organization in which such occupation and employment were carried out.
Mr. Leontaritis has been a self-employed private businessman residing in Athens, Greece and has been for the past five years. He has been involved in the public markets for over 25 years in numerous capacities. In 2004, he was the co-founder of a public bio-tech startup company in Athens, Greece. In 2007 he co-founded a solar energy company also in Greece. For the past two years, Mr. Leontaritis has been working on a project for rehabilitation centers, diabetic foot care and research and development of health care technologies with a Cyprus corporation, H.C.I. Viocare Ltd., of which he is currently the sole shareholder.
He is not an officer and director of any other reporting issuers.
Nikolaos Kardaras – Secretary and Director
Mr. Nikolaos Kardaras is member of the Piraeus Bar, having practiced law since 1976. He graduated from the Law School of Athens, Greece in 1974. His current practice encompasses the disciplines of maritime, commercial, civil, initiatives and investments law, acquisition and mergers, and international shipping and commercial transactions.
Mr. Kardaras is not an officer or director of any other reporting issuers.
Dr. Christos Kapatos – Director, Chief Technical Officer
Dr. Kapatos operates a bioengineering technology consulting business, HCinnovations, Glasgow, where he has been the consultant and the managing director since 2009. He specializes in the fields of bioengineering, education, medical devices, healthcare automation, advanced prosthetics, orthotics and diabetic technologies, and medical technology startups. He has published a number of papers and has filed patents for prosthetic technologies. From 2008 to 2011 he was the innovation and commercialization director for Diolab Ltd, an international medical equipment company. From 2008 to present, he has worked with Scotsig, a company with small to medium equipment technology in the field of medical signal processing, as a medical devices, innovation and commercialization consultant. Dr. Kapatos received a BEng degree from Northumbria University in 1998, an Msc degree in bioengineering from the University of Strathclyde in 1999, a Msc degree related to new venture creation from Caledonian University Business School in 2007, and a Phd in Bioengineering from the University of Strathclyde in 2006. He has worked with Mr. Leontaritis on projects for H.C.I. Viocare Ltd.
Dr. Kapatos is not an officer and director of any other reporting issuers.
Sergios Katsaros, Vice President
Mr. Katsaros received a MEng Honours in Electrical and Electronic Engineering from the University of Bristol in 1998 and a MBA in International Business from the University of Bristol in association with École nationale des ponts et chausses (ENPC), France, in 2000. Mr. Katsaros has worked for various large international conglomerates including Deutsche Börse AG, Mercedes – Benz Hellas, Alpha Private Bank and ASC Energy. Mr. Katsaros has held various key positions in these business entities including the position of Key Account Manager. Over the recent years Mr. Katsaros has worked in the financial sector as a private banker handling relationship management and sales and also as an asset manager in cooperation with various Swiss entities. Mr. Katsaros also has significant experience having served as a team leader in the energy sector in the areas of business development and oil arbitrage.
Mr. Katsaros is not an officer or director of any other reporting issuers.
52
Yiannis Levantis, Director
Mr. Levantis received a BEng, Honors in Mechanical Engineering from the University of Manchester Institute of Science and Technology (UMIST) in 1997, an MSc in Computational and Experimental Stress Analysis from UMIST in 1998 and an MSc in Financial Management and Control from Aston Business School in 1999. Mr. Levantis has worked for various large international conglomerates including Unilever, Johnson & Johnson, GlaxoSmithKline and Rolls Royce Plc. and has extensive experience in definition and execution of strategy and roadmaps to support business strategy and drive operational efficiencies. Over the most recent years Mr. Levantis has been employed as the Global IT director (Rolls Royce PLC – Aerospace, Defence & Power Systems July 2011 to present and GlaxoSmithKline, Pharmaceutical January 2008 to June 2011) undertaking such rolls as global SAP-ERP design and delivery, delivering IT strategy and IT enabled business transformation programs supporting various business functions including supply chain purchasing, finance, quality and HR and operating with annual budgets in excess of £20 million as well as other key management and financial planning solutions.
Mr. Levantis is not an officer or director of any other reporting issuers.
Code of Ethics
The Company has adopted a Code of Ethics applicable to its Chief Executive Officer and Chief Financial Officer. This Code of Ethics was filed as an exhibit to the Form 10-K for the year ending December 31, 2008 filed on April 6, 2009.
We will provide a copy of the Code of Ethics to any person without charge, upon request. Requests can be sent to our company at the address on the cover of this annual report.
Nominating Committee
There have been no material changes to the procedures by which security holders may recommend nominees to the Company's board of directors.
Audit Committee
At this time, the Company is not required to have an audit committee. Further, since there no independent members of the Board it is not feasible at this time to have an audit committee. The Board of Directors performs the same functions as an audit committee. The Board of Directors in performing its functions as an audit committee has determined that it does not have an audit committee financial expert.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Securities Exchange Act of 1934, as amended, requires our directors, executive officers, and stockholders holding more than 10% of our outstanding common stock, to file with the Securities and Exchange Commission initial reports of ownership and reports of changes in beneficial ownership of our common stock. Executive officers, directors and greater-than-10% stockholders are required by SEC regulations to furnish us with copies of all Section 16(a) reports they file. Based solely on our review of forms 3, 4 and 5 and amendments thereto furnished to the Company during its most recent fiscal year, the following directors, officers and/or person beneficially owning greater than 10% of the Company’s equity securities failed to timely file reports required by Section 16(a) of the Exchange Act during the most recent fiscal year or prior fiscal years:
Sergios Katsaros failed to timely file his Form 4’s in connection with the grant of stock awards previously disclosed in financial and annual filings.
The number of Forms 3, 4 and 5 and the number of transactions that were not filed timely are as follows: Sergios Katsaros (2 forms, 2 transactions). We note in a review of the filings with the Securities and Exchange Commission that Mr. Jinzhao Wu has not filed a Form 4 or Form 5 reporting the disposition of his shares of the Company during fiscal 2013.
53
|
·
|
Our principal executive officers for the fiscal years ended December 31, 2016 and 2015
|
|
SUMMARY COMPENSATION TABLE
|
|||||||||
|
Name
and Principal
Position
|
Year
|
Salary
($)
|
Bonus
($)
|
Stock
Awards
($)
|
Option
Awards
($)
|
Non-Equity
Incentive Plan
Compensation
($)
|
Change in
Pension
Value and
Nonqualified
Deferred
Compensation
Earnings
($)
|
All
Other
Compensation
($)
|
Total
($)
|
|
Sotirios Leontaritis(1)
Chief Executive Officer, Chief Financial Officer, President, Treasurer, and Director
|
2016
|
60,000
|
Nil
|
Nil
|
Nil
|
Nil
|
Nil
|
Nil
|
60,000
|
|
2015
|
60,000
|
Nil
|
Nil
|
Nil
|
Nil
|
Nil
|
Nil
|
60,000
|
|
|
Heleen Kist(3)
Chief Operating Officer (resigned August 30, 2016)
|
2016
|
36,529
|
Nil
|
Nil
|
Nil |
Nil
|
Nil
|
Nil
|
36,529
|
|
2015
|
51,582
|
Nil
|
Nil
|
Nil |
Nil
|
Nil
|
Nil
|
51,582
|
|
|
Christos Kapatos (4)
Chief Technical Officer
|
2016
|
44,266
|
Nil
|
Nil
|
Nil |
Nil
|
Nil
|
Nil
|
44,266
|
|
2015
|
44,380
|
Nil
|
Nil
|
Nil |
Nil
|
Nil
|
Nil
|
44,380
|
|
|
Sergios Katsaros(2)
Vice President
|
2016
|
26,560
|
Nil
|
169,000
|
Nil
|
Nil
|
Nil
|
Nil
|
195,560
|
|
2015
|
8,876
|
Nil
|
Nil
|
Nil
|
Nil
|
Nil
|
Nil
|
8,876
|
|
(1) Mr. Sotirios Leontaritis was appointed as Chief Executive Officer, President, Treasurer, and Director on September 9, 2013 and Chief Financial Officer on June 9, 2014. During fiscal 2015 and 2016 Mr. Leontaritis accrued total compensation of $60,000 per annum for total accruals of $120,000 which remained unpaid at December 31, 2016.
(2)Sergios Katsaros was appointed Vice President on September 1, 2015. During fiscal 2016 a total of 200,000 stock awards granted on his original appointment date fully vested and were valued at the market price on the date of the award totaling $169,000.
(3) Ms. Kist was appointed Chief Operating Officer on November 7, 2014.
(4) Dr. Christos Kapatos was appointed to the Company’s Board of Directors on September 30, 2013 and agreed to act as the Company’s Chief Technical Officer on April 16, 2014. During fiscal 2016 Mr. Kapatos accrued his total compensation of $44,266 which was unpaid as at December 31, 2016.
54
Outstanding Equity Awards at Fiscal Year-End
None.
Option Grants and Exercises
On April 15, 2014, the Company appointed Professor Martha Lucia Zequera, Professor William Sandham, Professor Stephan Solomonidis and Doctor Christos Kapatos to the Advisory Board of the Company and entered into advisory board agreements with respect to services to be provided. Compensation for the members of the Advisory Board shall be the grant of a total of 5,000 stock options entitling the advisory board member the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from the date of vesting. The advisory board appointments are for a term of one year and the options granted under the agreements will vest on completion of the term of the appointment. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
On June 9, 2014, the Company appointed Mr. William Spence to the Advisory Board of the Company and entered into an advisory board agreement with Mr. Spence. Compensation shall be the grant of a total of 5,000 stock options entitling Mr. Spence the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from the date of vesting. The advisory board appointment is for a term of one year and the options granted under the agreement will vest on completion of the term of the appointment. The Series A Preferred shares when vested will be convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
The following table summarizes information concerning stock options outstanding as of December 31, 2016:
|
December 31, 2016
|
December 31, 2015
|
|||||||||
|
Series A Preferred stock
|
Weighted Average Exercise Price
$
|
Series A Preferred stock
|
Weighted Average Exercise Price
$
|
|||||||
|
Outstanding at beginning of the year
|
-
|
-
|
-
|
-
|
||||||
|
Granted
|
25,000
|
0.04
|
25,000
|
0.04
|
||||||
|
Exercised
|
-
|
-
|
-
|
-
|
||||||
|
Expired or cancelled
|
-
|
-
|
-
|
-
|
||||||
|
Outstanding at the period
|
25,000
|
0.04
|
25,000
|
0.04
|
||||||
Employment Agreements, Officers and Directors
We have a consulting agreement with our Chief Executive Officer, Sotirios Leontaritis which was entered into on January 15, 2014, whereby Mr. Leontaritis shall provide services to the Company as the Company’s President and Chief Executive Officer in regards to the Company’s management and operations for the period from January 1, 2014 to December 31, 2017. Under the terms of the agreement, the Company agreed to pay to Mr. Leontaritis US$60,000 per annum payable in monthly payments of US$5,000 a month for the term of the contract. Further, under the terms of the contract, Mr. Leontaritis will receive cash compensation of US$5,000 and a stock award of 7,000,000 shares of common stock of the Company in consideration of his services as an officer and director for the period from September 10, 2013 to December 31, 2013. On January 22, 2014, the Company issued the 7,000,000 shares to Mr. Leontaritis pursuant to the agreement. On January 1, 2017, the Company approved a three-year extension to the consulting agreement between the Company and its President & CEO and controlling shareholder, Sotirios Leontaritis, originally entered into on January 1, 2014.
55
The revision to the Agreement has a term of three years, being effective as of January 1, 2017, and ending on December 31, 2019, renewable for such further term as may be mutually agreed between the parties. As per Addendum No. 1 the Company shall pay to Leontaritis US$120,000 per annum payable in monthly payments of US$10,000 a month for the term of the contract. Further, under the terms of the Addendum No. 1, Leontaritis is entitled to acquire at his discretion 3,000,000 shares of the common stock at a price of $0.30 per share for a term of five (5) years.
On April 16, 2014, the Company entered into a Consulting Agreement with Kapatos whereby he will provide his services as Chief Technical Officer for the Company and the Company’s wholly-owned subsidiaries. The contract has a term of one year, renewable for such further term as may be mutually agreed between the parties. Compensation shall be forty thousand Euros (€40,000) per year payable in equal monthly installments beginning on May 1, 2014. On May 1, 2015, the Company approved a one-year extension of the consulting agreement entered into with Dr. Christos Kapatos. During fiscal 2016, this agreement was extended for a further one year period.
On November 7, 2014, the Company’s subsidiary, HCi Viocare Clinics entered into a service agreement with Mrs. Heleen Francoise Kist whereby she will provide her services as Chief Operating Officer (“COO”) of the Company. The contract has a term of one year, being effective as of October 1, 2014, and ending on September 30, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation was thirty thousand GBP (£30,000) (USD$44,475) per year payable monthly in arrears on the last Friday of every month by credit transfer. On October 15, 2015, the Company’s COO, Heleen Kist and wholly owned subsidiary, HCI Viocare Clinics UK, signed an Addendum to an employment contract to extend the term to September 30, 2016, with a remuneration increase to forty five thousand GBP (£45,000) (USD$ 66,712) per year. Subsequently, on August 29, 2016 the Company’s subsidiary, HCi Viocare Clinics UK Ltd. approved a termination agreement with Heleen Kist which became effective at the close of her existing contract. Mrs. Kist resigned her positions as an officer and under the terms of the termination agreement the Company has undertaken to repurchase a total of seven hundred thousand (700,000) shares of the Company’s common stock at a purchase price of US$0.02 per share for total of fourteen thousand (US$14,000) USD for return to treasury and cancellation.
On September 1, 2015, the Board of Directors approved a consulting agreement with Sergios Katsaros and appointed Mr. Katsaros Vice President of HCi Viocare. Under the terms of the consulting agreement, Mr. Katsaros will work directly with the Company’s President and CEO in order to create and implement the Company’s strategic plan and assist in securing additional financing to meet the needs of the Company’s business plan and corporate objectives. The initial term of the contract is six months and Mr. Katsaros will receive compensation of EUR2,000 per month. Further, on March 1, 2016 the Company approved a one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros, originally entered into on September 1, 2015. On March 1, 2017, the Company approved another one-year extension to the consulting agreement between the Company and its Vice - President, Sergios Katsaros. The revision to the Agreement ("Addendum No. 2") has a term of one year, being effective as of March 1, 2017, and ending on February 28, 2018. All other terms and conditions remain valid and in force.
Compensation of Directors
During the most recent fiscal years ended 2016 and 2015, only Mr. Nikolaos Kardaras has received compensation for his services as a director of the Company. Mr. Kardaras received $13,280 in the fiscal year ended December 31, 2016 and $13,380 in the fiscal year ended December 2015. In addition, during fiscal 2015 Mr. Kardaras was issued a fully vested stock award of 700,000 shares of the Company’s common stock for prior services provided, valued at $450,000, which was the market price on the date of issue.
The Company has made no arrangements for the cash remuneration of its directors, except that they will be entitled to receive reimbursement for actual, demonstrable out-of-pocket expenses, including travel expenses, if any, made on the Company’s behalf.
Compensation Committee
We do not currently have a compensation committee. The Chief Executive Officer’s executive compensation has been approved by our Board of Directors. All of our other compensation contracts have been negotiated by our Chief Executive Officer and approved by our Board of Directors.
56
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS.
Security Ownership of Certain Beneficial Owners
Based on 195,064,844shares of common stock issued and outstanding as of April 14, 2017, the beneficial owners holding more than 5% of the outstanding common stock, taking into account beneficial ownership of common stock if all outstanding options, warrants, rights and conversion privileges (to which the applicable 5% stockholders have the right to exercise in the next 60 days) are exercised and additional shares of common stock are issued, are disclosed below:
|
Title of Class
|
Name and address of Beneficial Owner
|
Amount and Nature
of Beneficial Ownership
|
Percentage of Class (1)
|
|
Common Stock
|
Sotirios Leontaritis
|
145,710,562 shares held directly (3)
|
73.57%
|
|
Common Stock
|
Christos Kapatos
|
14,100,000 shares held directly(2)
|
7.23%
|
(1)Beneficial ownership is determined in accordance with SEC rules and includes voting or investment power with respect to securities. All shares of common stock subject to options or warrants exercisable within 60 days of April 14, 2017 are deemed to be outstanding and beneficially owned by the persons holding those options or warrants for the purpose of computing the number of shares beneficially owned and the percentage ownership of that person. They are not, however, deemed to be outstanding beneficially owned for the purpose of computing the percentage ownership of any other person. Subject to the paragraph above, the percentage ownership of outstanding shares is based on 195,064,844shares of common stock issued and outstanding as of April 14, 2017.
(2)This includes a total of 5,000 stock options entitling Mr. Kapatos the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from April 16, 2014. The Series A Preferred shares are convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
(3)Both the amount of ownership and the percentage of class calculation includes a total of 3,000,000 common shares which Mr. Leontaritis is entitled to acquire at his discretion effective January 1, 2017 at a price of $0.30 per share for a term of five (5) years.
Security Ownership of Management
The following table shows, as of April 14, 2017, the shares of the Company’s common stock beneficially owned by each director (including each nominee), by each of the executive officers and by all directors and executive officers as a group. Information is also provided regarding beneficial ownership of common stock if all outstanding options, warrants, rights and conversion privileges (to which the applicable officers and directors have the right to exercise in the next 60 days) are exercised and additional shares of common stock are issued.
|
Title of Class
|
Name of Beneficial Owner
|
Amount and Nature of Beneficial Ownership
|
Percentage of Class(1)
|
|
Common Stock
|
Sotirios Leontaritis, CEO and President
|
145,710,562 shares held directly (3)
|
73.57%
|
|
Common Stock
|
Christos Kapatos, Director and CTO
|
14,100,000 shares held directly(2)
|
7.23%
|
|
Common Stock
|
Nikolaos Kardaras, Director and Secretary
|
1,700,000 shares held directly
|
0.87%
|
|
Common Stock
|
Sergios Katsaros, Vice President
|
1,300,000 shares held directly
|
0.67%
|
|
All Officers and Directors as a Group
|
162,810,562
|
82.33%
|
(1)Beneficial ownership is determined in accordance with SEC rules and includes voting or investment power with respect to securities. All shares of common stock subject to options or warrants exercisable within 60 days of April 14, 2017 are deemed to be outstanding and beneficially owned by the persons holding those options or warrants for the purpose of computing the number of shares beneficially owned and the percentage ownership of that person. They are not, however, deemed to be outstanding beneficially owned for the purpose of computing the percentage ownership of any other person. Subject to the paragraph above, the percentage ownership of outstanding shares is based on 195,064,844shares of common stock outstanding as of April 14, 2017.
(2)This includes a total of 5,000 stock options entitling Mr. Kapatos the right to purchase a total of 5,000 shares of Series A Preferred Stock of the Company at a price of $0.04 per share for a period of five (5) years from April 16, 2014. The Series A Preferred shares are convertible on the basis of 20 shares of common stock for each one share held and have voting rights of 50 votes per share of Series A Preferred stock held at any meetings of the stockholders.
(3)Both the amount of ownership and the percentage of class calculation includes a total of 3,000,000 common shares which Mr. Leontaritis is entitled to acquire at his discretion effective January 1, 2017 at a price of $0.30 per share for a term of five (5) years.
57
Changes in Control
None
Securities authorized for issuance under equity compensation plans.
None.
The following table provides details of the Company’s related party transactions during the year ended December 31, 2016 and 2015:
(a) Services provided from related parties:
|
Year Ended
December 31,
|
||||||||
|
2016
|
2015
|
|||||||
|
Consulting fees from CEO and President (i)
|
$
|
60,000
|
$
|
60,000
|
||||
|
Consulting fees from a Director (ii)
|
44,266
|
44,380
|
||||||
|
Professional fees from Director (iii)
|
13,280
|
13,380
|
||||||
|
Consulting fees for VP (iv)
|
26,560
|
8,876
|
||||||
|
Consulting fees for COO (v)
|
36,529
|
51,582
|
||||||
|
Stock-based compensation (VP) (iv)
|
169,000
|
-
|
||||||
|
Stock award granted to Director (iii)
|
-
|
450,000
|
||||||
|
$
|
349,635
|
$
|
628,218
|
|||||
|
(i)
|
On September 10, 2013 Mr. Leontaritis was appointed President. On January 15, 2014, the Board of Directors of the Company approved the execution of a consulting agreement between the Company and Sotirios Leontaritis (“Leontaritis”), whereby Leontaritis shall provide services to the Company as the Company’s President and Chief Executive Officer in regards to the Company’s management and operations for the period from January 1, 2014 to December 31, 2017. Under the terms of the agreement, the Company agreed to pay to Leontaritis US$60,000 per annum payable in monthly payments of US$5,000 a month for the term of the contract.
|
|
|
(ii)
|
On September 30, 2013, the Board of Directors of the Company appointed Dr. Christos Kapatos as a director of the Company. During fiscal 2016 and fiscal 2015 Dr. Kapatos invoiced the Company GBP$40,000 for services rendered in each fiscal year respectively.
|
|
|
(iii)
|
On September 10, 2013, the Board of Directors of the Company elected Nikolaos Kardaras as Secretary and a director of the Company. On May 28, 2015, the Company approved the issuance of 700,000 common shares as compensation for prior services provided by director Nikolaos Kardaras in the form of fully vested stock awards. During fiscal 2016 and fiscal 2015 Mr. Kardaras invoiced the Company EUR $12,000 for services rendered in his capactiy as a director in each fiscal year respectively.
|
|
(iv)
|
On September 1, 2015, the Board of Directors of the Company approved a consulting agreement with Sergios Katsaros and appointed Mr. Katsaros Vice President of HCi Viocare. Under the terms of the consulting agreement, the initial term of the contract is six months and Mr. Katsaros will receive compensation of EUR€2,000 (USD$2,180) per month.
On March 1, 2016, the Company approved a one-year extension to the consulting agreement. Concurrently, the Company approved the grant of a stock award of 300,000 common shares as compensation for the services provided by Vice President, Sergios Katsaros. The award vests in three equal instalments of 100,000 shares as of the date of grant, the six-month anniversary of the date of grant and the 12-month anniversary of date of grant. During fiscal 2016 a total of 200,000 awards were fully vested with a fair market value of $169,000.
|
|
(v)
|
On November 7, 2014, the Company’s subsidiary, HCi Viocare Clinics (formerly W.D. Spence Prosthetics Limited) entered into a service agreement with Mrs. Heleen Francoise Kist (the “Agreement”) whereby she will provide her services as Chief Operating Officer (“COO”) of the Company. The contract has a term of one year, being effective as of October 1, 2014, and ending on September 30, 2015, renewable for such further term as may be mutually agreed between the parties. Compensation shall be thirty thousand GBP (£30,000) (USD$43,180) per year payable monthly in arrears on the last Friday of every month by credit transfer.
On October 15, 2015, the Company’s COO, Heleen Kist and wholly owned subsidiary, HCI Viocare Clinics UK, signed an Addendum to an employment contract to extend the term to September 30, 2016, with a remuneration increase to forty-five thousand GBP (£45,000) (USD$ 64,800) per year.
On August 30, 2016, the Board of Directors of the Company approved the termination of the services agreement of Mrs. Heleen Francoise Kist, Chief Operation Officer of the Company following her resignation from any and all positions with the Company and its subsidiaries.
|
58
(b) Accounts payable and accrued liabilities from related parties:
|
Balance,
December 31,
2015
($)
|
Services provided
during the period
($)
|
Reimbursement on Company’s expenses
and interest on loan
($)
|
Payments
($)
|
Foreign exchange
($)
|
Balance,
December 31,
2016
($)
|
||||||||||||||||
|
Consulting fees from CEO and President (i)
|
293,881
|
60,000
|
131,025
|
(8,750
|
)
|
(10,058
|
)
|
466,098
|
|||||||||||||
|
Consulting fees from a Director (ii)
|
-
|
44,266
|
-
|
(2,117
|
)
|
42,149
|
|||||||||||||||
|
Professional fees from Director (iii)
|
1,091
|
13,280
|
-
|
(13,319
|
)
|
-
|
1,052
|
||||||||||||||
|
Consulting fees for VP (iv)
|
-
|
26,560
|
-
|
(26,560
|
)
|
-
|
-
|
||||||||||||||
|
Consulting fees for COO (v)
|
-
|
36,529
|
-
|
(36,529
|
)
|
-
|
-
|
||||||||||||||
|
294,972
|
180,635
|
131,025
|
(85,158
|
)
|
(12,175
|
)
|
509,299
|
||||||||||||||
(c) Advances from related parties:
|
Balance, December 31, 2015
($)
|
Addition
($)
|
Assignment
& Assumption
($)
|
Payment
($)
|
Foreign exchange
($)
|
Balance, December 31, 2016
($)
|
|||||||||||||||||||
|
CEO and President (i)
|
615,898 | 466,925 | (109,079 | ) | (619,187 | ) | (38,480 | ) | 316,077 | |||||||||||||||
|
Director (ii)
|
- | 109,079 | (40,076 | ) | (1,916 | ) | 67,087 | |||||||||||||||||
|
COO (v)
|
33,522 | (32,346 | ) | (1,176 | ) | - | ||||||||||||||||||
| 649,420 | 466,925 | - | (691,609 | ) | (41,572 | ) | 383,164 | |||||||||||||||||
(ii) During January 2016, Christos Kapatos, director and CTO, entered into a private agreement for the assignment and assumption of certain advances payable with Sotirios Leontaritis, President and CEO, in respect of an amount totalling EUR$100,000 (US$109,079). During the year ended December 2016 amounts totaling $40,076 were repaid to Dr. Kapatos leaving a balance of $67,087 due and payable as at December 31, 2016.
(d) Loan Agreement from related parties (ref Note 8 - Loan)
|
Balance,
December 31, 2015
($)
|
Addition
($)
|
Payment
($)
|
Foreign Exchange on the Note
($)
|
Balance,
December 31, 2016
($)
|
||||||||||||||||
|
CEO and President (i)
|
163,595
|
44,925
|
(214,072
|
)
|
5,552
|
-
|
||||||||||||||
On June 11, 2015 and February 10, 2016, the Company entered into loan agreements with Mr. Leontaritis, the President of the Company, pursuant to which the Company received $163,595 (EUR €150,000) and $44,925 (EUR€40,000 as advances (collectively the “Notes”). The Notes earn simple interest accruing at five percent (5%) per annum and are due and payable within five years from the date of the agreement (“Maturity Date”).
During the year ended December 31, 2016 we accrued interest totaling $3,598 in respect of these loans.
During the year ended December 31, 2016, the Company repaid USD $222,822 (EUR €197,770) in cash in full settlement of the above notes, including accrued interest expense of USD $8,750 (EUR €7,770).
59
|
Review, Approval or Ratification of Transactions with Related Persons
|
We do not currently have any written policies and procedures for the review, approval or ratification of any transactions with related persons.
|
Promoters and Certain Control Persons
|
Our Chief Executive Officer owns beneficially and in the aggregate, the majority of the voting power of the Company. Accordingly, the Chief Executive Officer has the ability to control the approval of most corporate actions, including approving significant expenses, increasing the authorized capital stock and the dissolution, merger or sale of the Company's assets.
|
Parents
|
None.
|
Director Independence
|
As of the date of this Annual Report, we have one independent director. Mr. Yiannis Levantis.
We have developed the following categorical standards for determining the materiality of relationships that the directors may have with the Company. This information is not available on our Web site because such site is currently under development.
A director shall not be deemed to have a material relationship with the Company that impairs the director's independence as a result of any of the following relationships:
|
1.
|
the director is an officer or other person holding a salaried position of an entity (other than a principal, equity partner or member of such entity) that provides professional services to the Company and the amount of all payments from the Company to such entity during the most recently completed fiscal year was less than two percent of such entity’s consolidated gross revenues;
|
|
|
2.
|
the director is the beneficial owner of less than five (5%) per cent of the outstanding equity interests of an entity that does business with the Company;
|
|
|
3.
|
the director is an executive officer of a civic, charitable or cultural institution that received less than the greater of one million ($1,000,000) dollars or two (2%) per cent of its consolidated gross revenues, as such term is construed by the New York Stock Exchange for purposes of Section 303A.02(b)(v) of the Corporate Governance Standards, from the Company or any of its subsidiaries for each of the last three (3) fiscal years;
|
|
|
4.
|
the director is an officer of an entity that is indebted to the Company, or to which the Company is indebted, and the total amount of either the Company's or the business entity's indebtedness is less than three (3%) per cent of the total consolidated assets of such entity as of the end of the previous fiscal year; and
|
|
|
5.
|
the director obtained products or services from the Company on terms generally available to customers of the Company for such products or services. The Board retains the sole right to interpret and apply the foregoing standards in determining the materiality of any relationship.
|
The Board shall undertake an annual review of the independence of all non-management directors. To enable the Board to evaluate each non-management director, in advance of the meeting at which the review occurs, each non-management director shall provide the Board with full information regarding the director’s business and other relationships with the Company, its affiliates and senior management.
Directors must inform the Board whenever there are any material changes in their circumstances or relationships that could affect their independence, including all business relationships between a director and the Company, its affiliates, or members of senior management, whether or not such business relationships would be deemed not to be material under any of the categorical standards set forth above. Following the receipt of such information, the Board shall re-evaluate the director's independence.
60
The aggregate fees billed for the most recently completed fiscal year ended December 31, 2016 and for the fiscal year ended December 31, 2015 for professional services rendered by the principal accountant for the audit of our annual financial statements and review of the financial statements included in our quarterly reports on Form 10-Q and services that are normally provided by the accountant in connection with statutory and regulatory filings or engagements for these fiscal periods were as follows:
|
Year Ended
|
||||||||
|
December 31, 2016
$
|
December 31, 2015
$
|
|||||||
|
Audit Fees
|
27,182
|
21,000
|
||||||
|
Audit Related Fees
|
-0-
|
-0-
|
||||||
|
Tax Fees
|
-0-
|
7,050
|
||||||
|
All Other Fees
|
-0-
|
-0-
|
||||||
|
Total
|
27,182
|
28,050
|
||||||
Audit Fees
During the fiscal year ended December 31, 2016, we incurred approximately $27,182 in fees to our principal independent accountants for professional services rendered in connection with the audit and reviews of our financial statements for fiscal year ended December 31, 2016.
During the fiscal year ended December 31, 2015, we incurred approximately $21,000 in fees to our principal independent accountants for professional services rendered in connection with the audit and reviews of our financial statements for fiscal year ended December 31, 2015.
Audit-Related Fees
The aggregate fees billed during the fiscal years ended December 31, 2016 and 2015 for assurance and related services by our principal independent accountants that are reasonably related to the performance of the audit or review of our financial statements (and are not reported under Item 9(e)(1) of Schedule 14A was $0 and $0, respectively.
Tax Fees
The aggregate fees billed during the fiscal years ended December 31, 2016 and 2015 for professional services rendered by our principal accountant for tax compliance, tax advice and tax planning were $Nil and $7,050, respectively.
All Other Fees
The aggregate fees billed during the fiscal year ended December 31, 2016 and 2015 for products and services provided by our principal independent accountants (other than the services reported in Items 9(e)(1) through 9(e)(3) of Schedule 14A was $0.
Our board of directors pre-approves all services provided by our independent auditors. All of the above services and fees were reviewed and approved by the board of directors either before or after the respective services were rendered.
Our board of directors has considered the nature and amount of fees billed by our independent auditors and believes that the provision of services for activities unrelated to the audit is compatible with maintaining our independent auditors’ independence.
61
PART IV
|
Exhibits:
|
Description
|
|
|
3.1.1
|
Articles of Incorporation
|
Incorporated by reference to our Registration Statement filed with the SEC on Form SB-2 dated June 29, 2007.
|
|
3.1.2
|
Certificate of Amendment to the Articles of Incorporation
|
Incorporated by reference to our Definitive 14C filed with the SEC on December 27, 2013
|
|
3.1.3
|
Certificate of Amendment to the Articles of Incorporation
|
Incorporated by reference to our Definitive 14C filed with the SEC on March 3, 2014
|
|
3.1.4
|
Certificate of Change to the Articles of Incorporation
|
Incorporated by reference to our Form 8-K filed with the SEC on August 7, 2015
|
|
3.2
|
Bylaws
|
Incorporated by reference to our Registration Statement filed with the SEC on Form SB-2/A dated October 11, 2007.
|
|
4.1
|
Certificate of Designation
|
Incorporated by reference to our Definitive 14C filed with the SEC on December 27, 2013
|
|
10.1
|
Contract between the Company and Sotirios Leontaritis dated effective January 1, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on January 21, 2014
|
|
10.2
|
Acquisition Agreement between HCi VioCare Technologies Limited and Christos Kapatos dated February 2, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on February 14, 2014.
|
|
10.3
|
Acquisition Agreement between HCi VioCare Technologies Limited and Dr. Christos Kapatos dated April 16, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on May 6, 2014
|
|
10.4
|
Consulting Agreement between the Company and Dr. Christos Kapatos dated April 16, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on May 6, 2014
|
|
10.5
|
Consulting Agreement between the Company and Professor Stephan Solomonidis dated April 16, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on May 6, 2014
|
|
10.6
|
Consulting Agreement between the Company and Professor William Sandham dated April 16, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on May 6, 2014
|
|
10.7
|
Form of Advisory Board Agreement
|
Incorporated by reference to our Form 8-K filed with the SEC on May 6, 2014
|
|
10.8
|
Letter from Zhen dated June 19, 2014 regarding change in certified accountant.
|
Incorporated by reference to our Form 8-K filed with the SEC on June 25, 2014
|
|
10.9
|
Agreement between HCi Viocare Clinics UK Limited (Formerly W.D. Spence Prosthetics Limited) and Heleen Francoise Kist, dated November 7, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on November 12, 2014
|
|
10.10
|
Share purchase agreement between Sotirios Leontaritis and HCi Viocare Dated November 28, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on December 2, 2014
|
|
10.11
|
Private placement subscription agreement dated December 24, 2014
|
Incorporated by reference to our Form 8-K filed with the SEC on January 2, 2015
|
|
10.12
|
Consulting Agreement between the Company and Mikulas Dylowicz dated January 1, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on January 2, 2015
|
|
10.13
|
Form of Private Placement Subscription Agreement between the Company and a private investor dated February 2, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on February 9, 2015
|
|
10.14
|
Form of Private Placement Subscription Agreement between the Company and a private investor dated March 23, 2015
|
Incorporated by reference to our Form 10-K filed with the SEC on April 16, 2015
|
|
10.15
|
The Agreement with HELLENIC AMERICAN SECURITIES S.A. dated May 7, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on May 13, 2015
|
|
10.16
|
The Amendment of Acquisition Agreement between HCi VioCare Technologies Limited and Dr. Christos Kapatos dated May 8, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on May 13, 2015
|
|
10.17
|
The Addendums to the Consulting Agreements of April 16, 2014 with Dr. Christos Kapatos, Professor William Sandham and Professor Stephan Solomonidis dated May 1, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on May 13, 2015
|
|
10.18
|
The amendments to the scientific advisory agreements, dated May 8, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on May 13, 2015
|
|
10.19
|
The debt settlement agreement with Leontaritis dated May 28, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on June 3, 2015
|
62
|
10.20
|
The grant notice for stock awards issued May 28, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on June 3, 2015
|
|
10.21
|
Loan agreement dated June 11, 2015 between the Company and its President, Sotirios Leontaritis
|
Incorporated by reference to our Form 8-K filed with the SEC on June 18, 2015
|
|
10.22
|
Trademark assignment dated March 18, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on June 18, 2015
|
|
10.23
|
The grant notice for stock award dated July 24, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on July 29, 2015
|
|
10.24
|
Consulting agreement between HCi Viocare and Sergios Katsaros, dated September 1, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on September 10, 2015
|
|
10.25
|
Form of Private Placement Subscription Agreement between the Company and a private investor
|
Incorporated by reference to our Form 8-K filed with the SEC on October 8, 2015
|
|
10.26
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on October 14, 2015
|
|
10.27
|
Addendum No. 1 to the services agreement between HCi Viocare Clinics UK Limited and Heleen Francoise Kist, dated October 15, 2015
|
Incorporated by reference to our Form 8-K filed with the SEC on October 16, 2015
|
|
10.28
|
Form of Private Placement Subscription Agreement between the Company and the Individuals
|
Incorporated by reference to our Form 10-Q filed with the SEC on November 16, 2015
|
|
10.29
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 10-Q filed with the SEC on November 16, 2015
|
|
10.30
|
Form of Private Placement Subscription Agreement between the Company and the Individuals
|
Incorporated by reference to our Form 8-K filed with the SEC on January 8, 2016
|
|
10.31
|
The Agreement with LXM Finance LLP
|
Incorporated by reference to our Form 8-K filed with the SEC on February 11, 2016
|
|
10.32
|
The Agreement with Consultant dated February 1, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on February 11, 2016
|
|
10.33
|
The Agreement with Consultant dated February 18, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on February 11, 2016
|
|
10.34
|
The Agreement with Consultant dated February 19, 2016.
|
Incorporated by reference to our Form 8-K filed with the SEC on February 11, 2016
|
|
10.35
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on March 3, 2016
|
|
10.36
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on March 11, 2016
|
|
10.37
|
Addendum No. 1 to the consulting agreement between HCi Viocare and Sergios Katsaros, dated September 1, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on March 11, 2016
|
|
10.38
|
The grant notice for stock awards issued March 1, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on March 11, 2016
|
|
10.39
|
The grant notice for stock awards issued March 22, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on March 23, 2016
|
|
10.40
|
Form of Private Placement Subscription Agreement between the Company and the Individuals
|
Incorporated by reference to our Form 8-K filed with the SEC on April 18, 2016
|
|
10.41
|
The Agreement with Renell Wertpapierhandelsbank AG dated April 22, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on April 24, 2016
|
|
10.42
|
Form of Private Placement Subscription Agreement between the Company and the Individuals
|
Incorporated by reference to our Form 10-Q filed with the SEC on May 23, 2016
|
|
10.43
|
Professional Relations and Consulting Agreement between the Company and Acorn Management Partners, LLC
|
Incorporated by reference to our Form 10-Q filed with the SEC on May 23, 2016
|
|
10.44
|
The Agreement with HAS S.A. dated July 6, 2016
|
Incorporated by reference to our Form 10-Q filed with the SEC on August 22, 2016
|
|
10.45
|
Form of Private Placement Subscription Agreement between the Company and the Individuals
|
Incorporated by reference to our Form 8-K filed with the SEC on July 13, 2016
|
|
10.46
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on July 15, 2016
|
|
10.47
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on July 27, 2016
|
|
10.48
|
The Extension and Amendment of Engagement Letter with LXM Finance LLP
|
Incorporated by reference to our Form 10-Q filed with the SEC on August 22, 2016
|
63
|
10.49
|
The Addendum No. 2 to the Consulting Agreement of April 16, 2014 with Dr. Christos Kapatos, dated August 2, 2016
|
Incorporated by reference to our Form 10-Q filed with the SEC on August 22, 2016
|
|
10.50
|
The Addendum No. 2 to the Scientific Advisory Board Agreement of April 15, 2014 with Dr. Christos Kapatos, dated August 2, 2016
|
Incorporated by reference to our Form 10-Q filed with the SEC on August 22, 2016
|
|
10.51
|
Form of termination relating to services agreement between HCi Viocare Clinics UK Limited and Heleen Francoise Kist, dated August 29, 2016
|
Incorporated by reference to our Form 8-K filed with the SEC on September 22, 2016
|
|
10.52
|
Form of Private Placement Subscription Agreements between the Company and the Individual subscribers
|
Incorporated by reference to our Form 8-K filed with the SEC on September 22, 2016
|
|
10.53
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 10-Q filed with the SEC on November 14, 2016
|
|
10.54
|
Form of grant notice for stock awards issued October 24, 2016
|
Incorporated by reference to our Form 10-Q filed with the SEC on November 14, 2016
|
|
10.55
|
The agreement with KCN Ltd.
|
Incorporated by reference to our Form 10-Q filed with the SEC on November 14, 2016
|
|
10.56
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 10-Q filed with the SEC on November 14, 2016
|
|
10.57
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on November 22, 2016
|
|
10.58
|
Form of Private Placement Subscription Agreement between the Company and the Individual
|
Incorporated by reference to our Form 8-K filed with the SEC on December 7, 2016
|
|
10.59
|
Addendum No. 2 to the consulting agreement between HCi Viocare and Sergios Katsaros, dated March 1, 2017
|
Incorporated by reference to our Form 8-K filed with the SEC on March 10, 2017
|
|
10.60
|
Addendum No. 1 to the consulting agreement
between HCi Viocare and Sotirios Leontaritis
dated January 1, 2017
|
Filed herewith
|
|
10.61
|
The grant notice for stock awards issued March 3, 2017
|
Filed herewith
|
|
10.62
|
The grant notice for stock awards issued March 6, 2017
|
Filed herewith
|
|
10.63
|
The grant notice for stock awards issued March 7, 2017
|
Filed herewith
|
|
10.64
|
The grant notice for stock awards issued March 17, 2017
|
Filed herewith
|
|
10.65
|
The grant notice for stock awards issued March 22, 2017
|
Filed herewith
|
|
14.1
|
Code of Ethics
|
Incorporated by reference to the registration statement filed with the SEC on Form 10-K dated April 6, 2009.
|
|
21
|
List of subsidiaries
|
- HCi Viocare Clinics UK Limited
- HCi Viocare Technologies UK Limited
- HCi Viocare Clinics (Hellas) S A
|
|
31.1
|
Certification of Principal Executive Officer and Principal Financial Officer filed pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
|
Filed herewith
|
|
32.1
|
Certification of Principal Executive Officer and Principal Financial Officer pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
|
Filed herewith
|
|
101.CAL
|
XBRL Taxonomy Extension Calculation Linkbase
|
Filed herewith
|
|
101.DEF
|
XBRL Taxonomy Extension Definition Linkbase
|
Filed herewith
|
|
101.INS
|
XBRL Instance Document
|
Filed herewith
|
|
101.LAB
|
XBRL Taxonomy Extension Label Linkbase
|
Filed herewith
|
|
101.PRE
|
XBRL Taxonomy Extension Presentation Linkbase
|
Filed herewith
|
|
101.SCH
|
XBRL Taxonomy Extension Schema
|
Filed herewith
|
64
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
HCi Viocare
|
|||
|
Date:
|
April 19, 2017
|
By:
|
/s/ Sotirios Leontaritis
|
|
Name:
|
Sotirios Leontaritis
|
||
|
Title:
|
Chief Executive Officer and President (Principal Executive Officer) Chief Financial Officer (Principal Financial Officer and Principal Accounting Officer)
|
||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
|
Date:
|
April 19, 2017
|
By:
|
/s/ Sotirios Leontaritis
|
|
Name:
|
Sotirios Leontaritis
|
||
|
Title:
|
President, Chief Executive Officer, Chief Financial Officer, Treasurer, and Director (Principal Executive Officer, Principal Financial Officer and Principal Accounting Officer)
|
||
|
Date:
|
April 19, 2017
|
By:
|
/s/ Nikolaos Kardaras
|
|
Name:
|
Nikolaos Kardaras
|
||
|
Title:
|
Secretary and Director
|
||
|
Date:
|
April 19, 2017
|
By:
|
/s/ Christos Kapatos
|
|
Name:
|
Christos Kapatos
|
||
|
Title:
|
Director
|
||
|
Date:
|
April 19, 2017
|
By:
|
/s/ Yiannis Levantis
|
|
Name:
|
Yiannis Levantis
|
||
|
Title:
|
Director
|
65
