Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - Impax Laboratories, LLC | ex9918k.htm |
| 8-K - 8-K - Impax Laboratories, LLC | ipxl-11x09x2017x8k.htm |

1
Third Quarter 2017 Results
and Business Update
November 9, 2017

2
Impax Cautionary Statement Regarding
Forward Looking Statements
"Safe Harbor" statement under the Private Securities Litigation Reform Act of 1995:
To the extent any statements made in this presentation contain information that is not historical; these statements are forward-looking in nature
and express the beliefs and expectations of management. Such statements are based on current expectations and involve a number of known
and unknown risks and uncertainties that could cause the Company’s future results, performance, or achievements to differ significantly from
the results, performance, or achievements expressed or implied by such forward-looking statements. Such risks and uncertainties include, but
are not limited to, fluctuations in the Company’s operating results and financial condition, the volatility of the market price of the Company’s
common stock, the Company’s ability to successfully develop and commercialize pharmaceutical products in a timely manner, the impact of
competition, the effect of any manufacturing or quality control problems, the Company’s ability to manage its growth, risks related to
acquisitions of or investments in technologies, products or businesses, the risks related to the sale or closure of the Company’s Taiwan
manufacturing facility, effects from fluctuations in currency exchange rates between the U.S. dollar and the Taiwan dollar, risks relating to
goodwill and intangibles, the reduction or loss of business with any significant customer, the substantial portion of the Company’s total revenues
derived from sales of a limited number of products, the impact of consolidation of the Company’s customer base, the Company’s ability to
sustain profitability and positive cash flows, the impact of any valuation allowance on the Company’s deferred tax assets, the restrictions
imposed by the Company’s credit facility and indenture, the Company’s level of indebtedness and liabilities and the potential impact on cash
flow available for operations, the availability of additional funds in the future, any delays or unanticipated expenses in connection with the
operation of the Company’s manufacturing facilities, the effect of foreign economic, political, legal and other risks on the Company’s operations
abroad, the uncertainty of patent litigation and other legal proceedings, the increased government scrutiny on the Company’s agreements to
settle patent litigations, product development risks and the difficulty of predicting FDA filings and approvals, consumer acceptance and demand
for new pharmaceutical products, the impact of market perceptions of the Company and the safety and quality of the Company’s products, the
Company’s determinations to discontinue the manufacture and distribution of certain products, the Company’s ability to achieve returns on its
investments in research and development activities, changes to FDA approval requirements, the Company’s ability to successfully conduct
clinical trials, the Company’s reliance on third parties to conduct clinical trials and testing, the Company’s lack of a license partner for
commercialization of Numient® (IPX066) outside of the United States, impact of illegal distribution and sale by third parties of counterfeits or
stolen products, the availability of raw materials and impact of interruptions in the Company’s supply chain, the Company’s policies regarding
returns, rebates, allowances and chargebacks, the use of controlled substances in the Company’s products, the effect of current economic
conditions on the Company’s industry, business, results of operations and financial condition, disruptions or failures in the Company’s
information technology systems and network infrastructure caused by third party breaches or other events, the Company’s reliance on alliance
and collaboration agreements, the Company’s reliance on licenses to proprietary technologies, the Company’s dependence on certain
employees, the Company’s ability to comply with legal and regulatory requirements governing the healthcare industry, the regulatory
environment, the effect of certain provisions in the Company’s government contracts, the Company’s ability to protect its intellectual property,
exposure to product liability claims, changes in tax regulations, uncertainties involved in the preparation of the Company’s financial statements,
the Company’s ability to maintain an effective system of internal control over financial reporting, the effect of terrorist attacks on the Company’s
business, the location of the Company’s manufacturing and research and development facilities near earthquake fault lines, expansion of social
media platforms and other risks described in the Company’s periodic reports filed with the Securities and Exchange Commission. Forward-
looking statements speak only as to the date on which they are made, and the Company undertakes no obligation to update publicly or revise
any forward-looking statement, regardless of whether new information becomes available, future developments occur or otherwise.
Trademarks referenced herein are the property of their respective owners.
©2017 Impax Laboratories, Inc. All Rights Reserved.

3
Paul Bisaro
President & CEO

4
Focused on Improving Profitability and Earnings
$228
$202 $206
3Q16 2Q17 3Q17
$59
$39
$46
3Q16 2Q17 3Q17
$0.37
$0.18
$0.23
3Q16 2Q17 3Q17
Revenues
$ millions
Adjusted EBITDA
$ millions
Adjusted EPS
Refer to the GAAP to non-GAAP reconciliation tables in the appendix for a reconciliation of non-GAAP results.
Third Quarter 2017
Year over year performance impacted by generic industry dynamics
Sequential improvement in profitability and earnings over 2Q17
driven by acceleration of our cost savings initiatives
Adjusted EBITDA up 16%
Adjusted EPS up 28%

5
Third Quarter 2017 Business Update
Quarter Results
3Q17 vs. 3Q16 – Revenue down 14%
Driven by lower pricing (-18%) and partially
offset by increased volumes
Excluding epinephrine auto-injector and
gAdderall XR®, 3Q17 pricing across generic
products was down ~7% to 9%
3Q17 vs. 2Q17 – Revenue and pricing flat
Strong epinephrine auto-injector and diclofenac
sodium gel 3% sales partially offset by lower
sales of budesonide, fenofibrate and authorized
generic Oxycontin ER®
Post Third Quarter Events
Generic Renvela® approval and launch
Generic Concerta® launch timing delayed
Quarter Results
3Q17 vs. 3Q16 – Revenue up 5%
Driven by higher sales of Rytary® and Emverm®
3Q17 vs. 2Q17 – Revenue up 8%
Driven by higher sales of Albenza® and Zomig®
Resolved prior quarter Albenza supply issue
Generic Division Specialty Pharma Division
6
0
10
20
30
40
50
60
70
80
0.5 h 1 h 1.5 h 2 hP
ro
p
ort
io
n
o
f
S
u
b
je
c
ts
A
c
h
ie
v
in
g
De
fi
n
e
d
Red
u
c
ti
o
n
i
n
O
ff
T
im
e
Reduction in Off Time from Baseline
IPX203
IR CD-LD
0
10
20
30
40
50
60
7-point 13-point
P
ro
p
ort
io
n
o
f
S
u
b
je
c
ts
A
c
h
ie
v
in
g
S
p
e
c
if
ie
d
I
m
pro
v
e
m
e
n
t
o
n
MD
S
-
U
P
DRS
P
art
III
P
re
d
o
s
e
o
n
Da
y
1
5
IPX203
IR CD-LD
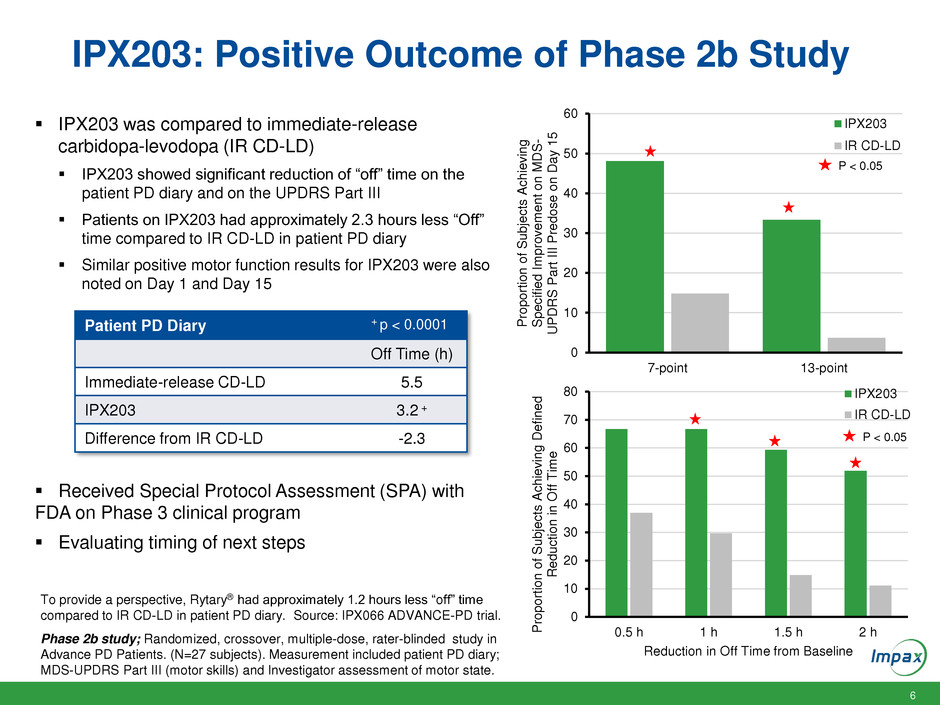
IPX203: Positive Outcome of Phase 2b Study
IPX203 was compared to immediate-release
carbidopa-levodopa (IR CD-LD)
IPX203 showed significant reduction of “off” time on the
patient PD diary and on the UPDRS Part III
Patients on IPX203 had approximately 2.3 hours less “Off”
time compared to IR CD-LD in patient PD diary
Similar positive motor function results for IPX203 were also
noted on Day 1 and Day 15
Received Special Protocol Assessment (SPA) with
FDA on Phase 3 clinical program
Evaluating timing of next steps
Patient PD Diary
Off Time (h)
Immediate-release CD-LD 5.5
IPX203 3.2 +
Difference from IR CD-LD -2.3
+ p < 0.0001
P < 0.05
P < 0.05
To provide a perspective, Rytary® had approximately 1.2 hours less “off” time
compared to IR CD-LD in patient PD diary. Source: IPX066 ADVANCE-PD trial.
Phase 2b study; Randomized, crossover, multiple-dose, rater-blinded study in
Advance PD Patients. (N=27 subjects). Measurement included patient PD diary;
MDS-UPDRS Part III (motor skills) and Investigator assessment of motor state.

7
2017 Initiatives Original Timing Updated Timing
Consolidation of all generic R&D to Hayward, CA Completed mid-2017 Completed on-time
Closure of Middlesex packaging site Completion by end of 1Q18 Completion by end of 2017
Rationalizing generic portfolio to eliminate low-value
products
Completion by 1Q18 On target
Strategic alternatives for Taiwan manufacturing site As late as end of 2019 1Q18 (1)
Reorganizing certain functions including quality,
engineering and supply chain operations
As late as end of 2019 1Q18 (1)
Accelerated Progress on Consolidation and
Improvement Plan
~$85M Run-Rate Savings Expected by Year-End 2018 (1)
2017
~$10 million
End of 2018 (1)
Full run-rate savings of ~$85M
(1) Assuming a close by end of 1Q18, total run-rate savings achieved by year-end 2018. Potential sale of Taiwan facility currently in late-stage negotiations with potential buyer.

8
A Strategic Combination for Long-Term Growth
(1) Per Last Twelve Months IMS Gross Revenues as of June 2017.
(2) Includes expected Year 1 run-rate synergies.
(3) In addition to the previously announced Impax standalone cost savings initiatives.
Expanded Portfolio to Drive Growth
Creates 5th largest U.S. generics
company(1)
Increases scale and diversification across
currently marketed product families and
R&D pipeline
High-margin specialty franchise is
expected to provide stable cash flow and
a long-term growth platform
Significant Financial Benefits
Annual double-digit revenue, adjusted
EBITDA and adjusted EPS growth over
next 3 years driven by already filed new
product launches
Significant projected cash flow generation
enables de-leveraging and future
investment in high-growth specialty and
other adjacencies
Accretive to Impax’s adjusted EPS in the
first 12 months after close(2)
$200 million in expected annual synergies
within 3 years(3)
Initiated pre-close integration planning

9
Bryan Reasons
Chief Financial Officer

10
Generic Division 3Q 2017 Results
Refer to the GAAP to non-GAAP reconciliation tables in the appendix for a reconciliation of non-GAAP results.
$ millions 3Q 2017 2Q 2017
Change
3Q/2Q
3Q 2016
Change
3Q/3Q
GENERIC DIVISION
Total Revenues $151.1 $150.9 0% $175.3 (14%)
GAAP Gross Margin (2%) 28% -- (112%) --
Adjusted Gross Margin 35% 43% -- 44% --
GAAP (Loss) Operating Income ($21.5) $12.6 (271%) ($233.3) 91%
Adjusted Operating Income $34.8 $39.5 (12%) $56.0 (38%)

11
Specialty Pharma Division 3Q 2017 Results
Refer to the GAAP to non-GAAP reconciliation tables in the appendix for a reconciliation of non-GAAP results.
$ millions 3Q 2017 2Q 2017
Change
3Q/2Q
3Q 2016
Change
3Q/3Q
SPECIALTY PHARMA DIVISION
Total Revenues $55.3 $51.2 8% $52.6 5%
GAAP Gross Margin 68% 59% -- 58% --
Adjusted Gross Margin 85% 71% -- 73% --
GAAP Operating Income $16.4 $6.9 145% ($6.7) 345%
Adjusted Operating Income $25.6 $12.7 102% $13.9 84%

12
Consolidated 3Q 2017 Results
Refer to the GAAP to non-GAAP reconciliation tables in the appendix for a reconciliation of non-GAAP results.
$ millions, except per share amounts 3Q 2017 2Q 2017
Change
3Q/2Q
3Q 2016
Change
3Q/3Q
EBITDA ($15.4) $16.6 (193%) ($247.9) 94%
Adjusted EBITDA $45.6 $39.2 16% $116.7 (61%)
GAAP Loss Per Share ($0.69) ($0.28) (146%) ($2.51) 72%
Adjusted Diluted EPS $0.23 $0.18 28% $0.37 (38%)
GAAP Tax Rate 6% 3% -- 37% --
Adjusted Tax Rate 35% 31% -- 34% --

13
Paul Bisaro
President & CEO

14
Updated 4Q17 Guidance Assumptions
Factors Adversely Impacting 4Q
• Delay in launch of generic Concerta® and generic Welchol®
• Late approval of generic Renvela® and launch into a more
competitive market
• Inability of partner to supply generic Aggrenox® impacting royalty
income
• In addition, lower sequential revenues of epinephrine auto-injector
due to seasonality
Factors Positively Impacting 4Q
• Acceleration of cost improvement program (CIP)
• Reduction in R&D spending due to generic site consolidation
Expect 2018 to benefit from delayed and new generic launch
revenue and accelerated contribution from CIP

15
Updated 2017 Financial Guidance
* Excludes new cost savings initiatives as outlined on slide 7.
The Company’s full year 2017 estimates are based on management’s current expectations, including with respect to prescription trends,
pricing levels, inventory levels, and the anticipated timing of future product launches and events. These statements are forward-looking, and
actual results could differ materially depending on market conditions and the factors set forth under our “Safe Harbor” statement above.
Previous Guidance
August 9*
Updated Guidance
November 9
Adjusted Gross Margin as a % of
Revenues
~ 47% to 49% ~ 47%
Adjusted R&D & Patent Litigation
Expense
~ $93M to $97M ~ $84M to $88M
Adjusted Selling, General &
Administrative Expense
~ $190M to $195M No Change
Adjusted Interest Expense ~ $28M No Change
Adjusted EPS ~ $0.55 to $0.70 ~ $0.60 to $0.65
Tax Rate ~ 33% to 34% ~33%
Capital Expenditures ~ $25M to $30M No Change

16
Path Forward
• Generics: Continuing internal R&D investment and external R&D license efforts
• Specialty: Continuing focus on Movement Disorders pipeline and opportunistically
in-license external opportunities
Invest in
Organic
Growth
• Maintain high level of Quality and Compliance
• Achieve superior service levels
• Deliver differentiated products to our customers
Maintain
Customer
Focus
• Achieve consolidation targets without business disruption
• Continue to explore additional cost savings opportunities
Achieve
“CIP” Target
• Strengthen Generic and Specialty franchises
Pursue
Creative
Business
Development
Position Impax for Sustainable Long-Term Growth

17
Third Quarter 2017 Results
and Business Update
Q&A Session
November 9, 2017
18
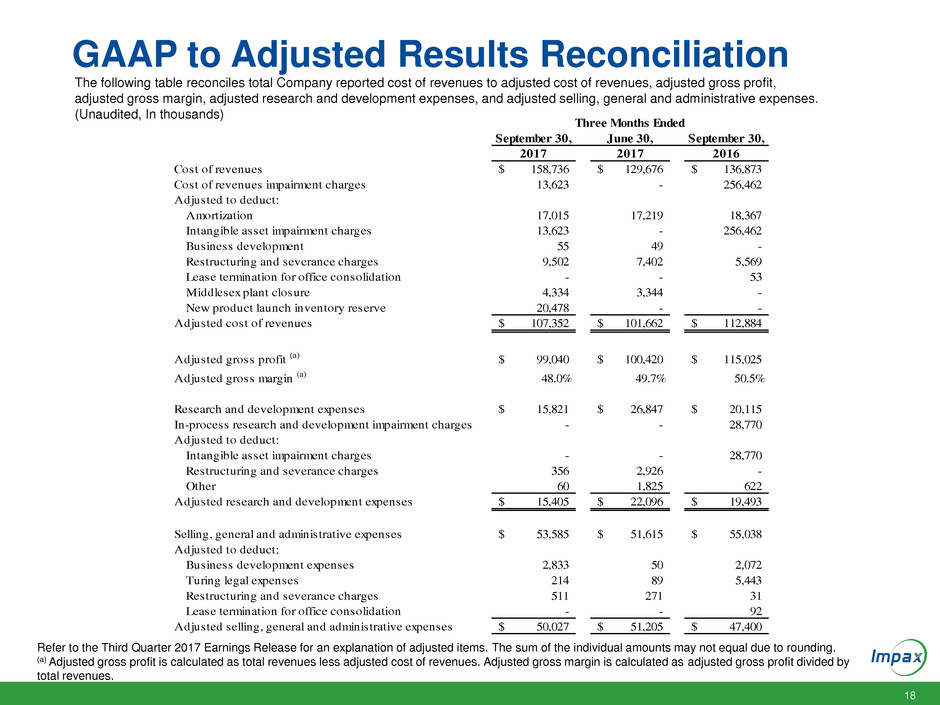
GAAP to Adjusted Results Reconciliation
The following table reconciles total Company reported cost of revenues to adjusted cost of revenues, adjusted gross profit,
adjusted gross margin, adjusted research and development expenses, and adjusted selling, general and administrative expenses.
(Unaudited, In thousands)
Refer to the Third Quarter 2017 Earnings Release for an explanation of adjusted items. The sum of the individual amounts may not equal due to rounding.
(a) Adjusted gross profit is calculated as total revenues less adjusted cost of revenues. Adjusted gross margin is calculated as adjusted gross profit divided by
total revenues.
September 30, June 30, September 30,
2017 2017 2016
Cost of revenues 158,736$ 129,676$ 136,873$
Cost of revenues impairment charges 13,623 - 256,462
Adjusted to deduct:
Amortization 17,015 17,219 18,367
Intangible asset impairment charges 13,623 - 256,462
Business development 55 49 -
Restructuring and severance charges 9,502 7,402 5,569
Lease termination for office consolidation - - 53
Middlesex plant closure 4,334 3,344 -
New product launch inventory reserve 20,478 - -
Adjusted cost of revenues 107,352$ 101,662$ 112,884$
Adjusted gross profit (a) 99,040$ 100,420$ 115,025$
Adjusted gross margin (a) 48.0% 49.7% 50.5%
Research and development expenses 15,821$ 26,847$ 20,115$
In-process research and development impairment charges - - 28,770
Adjusted to deduct:
Intangible asset impairment charges - - 28,770
Restructuring and severance charges 356 2,926 -
Other 60 1,825 622
Adjusted research and development expenses 15,405$ 22,096$ 19,493$
Selling, general and administrative expenses 53,585$ 51,615$ 55,038$
Adjusted to deduct:
Business development expenses 2,833 50 2,072
Turing legal expenses 214 89 5,443
Restructuring and severance charges 511 271 31
Lease termination for office consolidation - - 92
Adjusted selling, general and administrative expenses 50,027$ 51,205$ 47,400$
Three Months Ended

19
GAAP to Adjusted Net Income Reconciliation
The following table reconciles reported net loss to adjusted net income.
(Unaudited, In thousands, except per share and per share data)
Refer to the Third Quarter 2017 Earnings Release for an explanation of adjusted items. The sum of the individual amounts may not equal due to rounding.
September 30, June 30, September 30,
2017 2017 2016
Net loss (49,369)$ (20,417)$ (179,337)$
Adjusted to add (deduct):
Amortization 17,015 17,219 18,367
Non-cash interest expense 6,547 6,430 5,890
Business development expenses 2,888 99 2,072
Intangible asset impairment charges 13,623 - 285,232
Reserve for Turing receivable - 2,353 -
Turing legal expenses 214 89 5,443
Restructuring and severance charges 10,937 10,599 5,600
Fixed asset impairment charges - 1,894 -
Lease termination for office consolidation - - 144
Gain on sale of intangible assets - (11,850) -
Gain on disposal of property, plant and equipment (4,379) (350) -
Loss on debt extinguishment - - -
Middlesex plant closure 4,334 3,344 -
New product launch inventory reserve 20,478 - -
Legal settlements - 7,900 -
Other 6,393 2,286 622
Income tax effect (11,998) (6,456) (117,884)
Adjusted net income 16,683$ 13,140$ 26,149$
Adjusted net income per diluted share 0.23$ 0.18$ 0.37$
Net loss per diluted share (0.69)$ (0.28)$ (2.51)$
Diluted weighted-average common shares outstanding 72,172 71,804 71,542
Three Months Ended

20
GAAP to Adjusted EBITDA Reconciliation
Refer to the Third Quarter 2017 Earnings Release for an explanation of adjusted items. The sum of the individual amounts may not equal due to rounding.
The following table reconciles reported net loss to adjusted EBITDA.
(Unaudited, In thousands)
September 30, June 30, September 30,
2017 2017 2016
Net loss (49,369)$ (20,417)$ (179,337)$
Adjusted to add (deduct):
Interest expense 13,636 13,369 11,089
Interest income (336) (155) (222)
Income taxes (3,045) (520) (104,531)
Depreciation and amortization 23,708 24,355 25,059
EBITDA (15,406) 16,632 (247,942)
Adjusted to add (deduct):
Share-based compensation expense 6,490 6,225 7,713
Business development expenses 2,888 99 2,072
Intangible asset impairment charges 13,623 - 285,232
Reserve for Turing receivable - 2,353 -
Turing legal expenses 214 89 5,443
Restructuring and severance charges 10,937 10,599 5,600
Fixed asset impairment charges - 1,894 -
Lease termination for office consolidation - - 144
Gain on sale of intangible assets - (11,850) -
Gain on disposal of property, plant and equipment (4,379) (350) -
Loss on debt extinguishment - - -
Middlesex plant closure 4,334 3,344 -
New product launch inventory reserve 20,478 - -
Legal settlements - 7,900 -
Other 6,393 2,286 622
Adjusted EBITDA 45,572$ 39,221$ 58,884$
Three Months Ended

21
Generic Division GAAP to Adjusted
Results Reconciliation
The following tables reconcile the Impax Generics Division reported cost of revenues and income (loss) from operations to adjusted cost
of revenues, adjusted gross profit, adjusted gross margin and adjusted operating income.
(Unaudited, In thousands)
Refer to the Third Quarter 2017 Earnings Release for an explanation of adjusted items. The sum of the individual amounts may not equal due to rounding.
(a) Adjusted gross profit is calculated as total revenues less adjusted cost of revenues. Adjusted gross margin is calculated as adjusted gross profit divided by
total revenues.
September 30, June 30, September 30,
2017 2017 2016
Cost of revenues 141,133$ 108,901$ 115,020$
Cost of revenues impairment charges 13,623 - 256,462
Adjusted to deduct:
Amortization 13,181 13,385 10,951
Intangible asset impairment charges 13,623 - 256,462
Restructuring and severance charges 4,245 5,396 5,569
Lease termination for office consolidation - - 53
Middle x plant closure 4,334 3,344 -
New product launch inventory reserve 20,478 - -
Adjusted cost of revenues 98,895$ 86,776$ 98,447$
Adjusted gross profit (a) 52,203$ 64,113$ 76,873$
Adjusted gross margin (a) 34.5% 42.5% 43.8%
Three Months Ended
GAAP (loss) income from operations (21,497)$ 12,640$ (233,330)$
Adjusted to add (deduct):
Amortization 13,181 13,385 10,951
Intangible asset impairment charges 13,623 - 272,005
Restructuring and severance charges 4,601 8,32 5,569
Lease termination for office consolidation - - 144
Payments for licensing agreements 60 1,825 622
Middlesex plant closure 4,334 3,344 -
New product launch inventory reserve 20,478 - -
Adjusted income from operations 34,780$ 39,516$ 55,961$
22
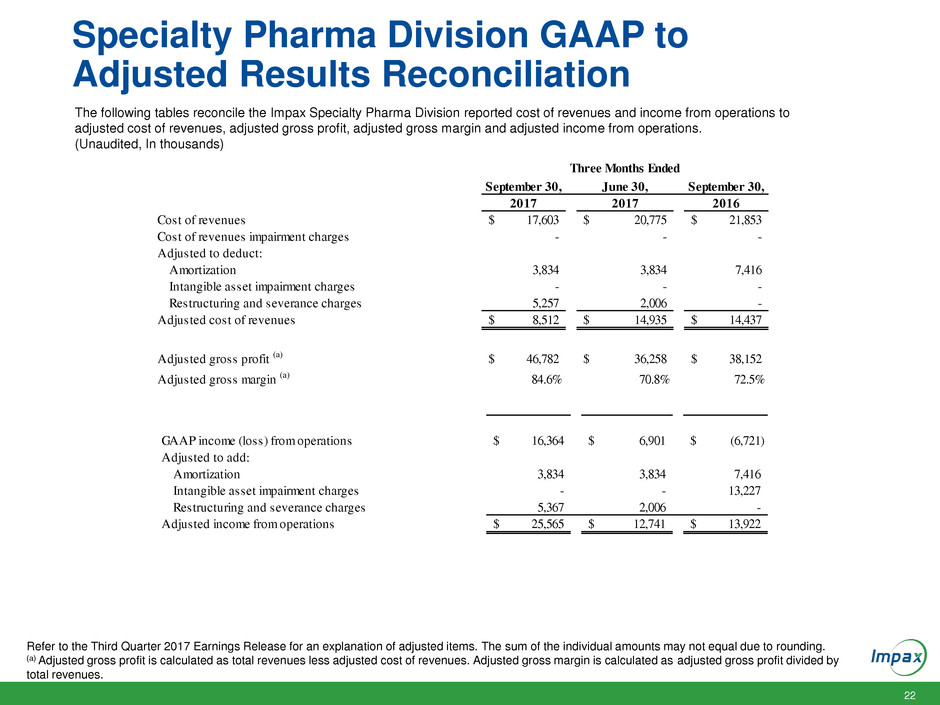
Specialty Pharma Division GAAP to
Adjusted Results Reconciliation
The following tables reconcile the Impax Specialty Pharma Division reported cost of revenues and income from operations to
adjusted cost of revenues, adjusted gross profit, adjusted gross margin and adjusted income from operations.
(Unaudited, In thousands)
Refer to the Third Quarter 2017 Earnings Release for an explanation of adjusted items. The sum of the individual amounts may not equal due to rounding.
(a) Adjusted gross profit is calculated as total revenues less adjusted cost of revenues. Adjusted gross margin is calculated as adjusted gross profit divided by
total revenues.
September 30, June 30, September 30,
2017 2017 2016
Cost of revenues 17,603$ 20,775$ 21,853$
Cost of revenues impairment charges - - -
Adjusted to deduct:
Amortizatio 3,834 3,834 7,416
Intangible asset impairment charges - - -
Restructuring and severance charges 5,257 2,006 -
Adjusted cost of revenues 8,512$ 14,935$ 14,437$
Adjusted gross profit (a) 46,782$ 36,258$ 38,152$
Adjusted gross margin (a) 84.6% 70.8% 72.5%
Three Months Ended
GAAP income (loss) from operations 1 ,364 6,901 (6,721)$
djusted to add:
Amortization 3,834 3,834 7,416
Intangible asset impairment charges - - 13,227
Restructuring and severance charges 5,367 2,006 -
Adjusted income from operations 25,565$ 12,741$ 13,922$
