Attached files
| file | filename |
|---|---|
| EX-12 - EXHIBIT 12 - Philip Morris International Inc. | pm-ex12q4201610xkversion.htm |
| EX-32.2 - EXHIBIT 32.2 - Philip Morris International Inc. | pm-ex322_123116xq4.htm |
| EX-32.1 - EXHIBIT 32.1 - Philip Morris International Inc. | pm-ex321_123116xq4.htm |
| EX-31.2 - EXHIBIT 31.2 - Philip Morris International Inc. | pm-ex312_123116xq4.htm |
| EX-31.1 - EXHIBIT 31.1 - Philip Morris International Inc. | pm-ex311_123116xq4.htm |
| EX-24 - EXHIBIT 24 - Philip Morris International Inc. | pm-ex24_123116xq4.htm |
| EX-23 - EXHIBIT 23 - Philip Morris International Inc. | pm-ex23_123116xq4.htm |
| EX-21 - EXHIBIT 21 - Philip Morris International Inc. | pm-ex21_123116xq4.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
ý | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2016
OR
¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number: 001-33708
PHILIP MORRIS INTERNATIONAL INC.
(Exact name of registrant as specified in its charter)
Virginia | 13-3435103 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
120 Park Avenue, New York, New York | 10017 | |
(Address of principal executive offices) | (Zip Code) | |
917-663-2000
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Title of each class | Name of each exchange on which registered | |
Common Stock, no par value | New York Stock Exchange | |
1.625% Notes due 2017 | New York Stock Exchange | |
1.250% Notes due 2017 | New York Stock Exchange | |
1.125% Notes due 2017 | New York Stock Exchange | |
1.250% Notes due 2017 | New York Stock Exchange | |
5.650% Notes due 2018 | New York Stock Exchange | |
1.875% Notes due 2019 | New York Stock Exchange | |
1.375% Notes due 2019 | New York Stock Exchange | |
2.125% Notes due 2019 | New York Stock Exchange | |
1.750% Notes due 2020 | New York Stock Exchange | |
4.500% Notes due 2020 | New York Stock Exchange | |
1.875% Notes due 2021 | New York Stock Exchange | |
1.875% Notes due 2021 | New York Stock Exchange | |
4.125% Notes due 2021 | New York Stock Exchange | |
2.900% Notes due 2021 | New York Stock Exchange | |
2.500% Notes due 2022 | New York Stock Exchange | |
2.625% Notes due 2023 | New York Stock Exchange | |
2.125% Notes due 2023 | New York Stock Exchange | |
3.600% Notes due 2023 | New York Stock Exchange | |
2.875% Notes due 2024 | New York Stock Exchange | |
3.250% Notes due 2024 | New York Stock Exchange | |
2.750% Notes due 2025 | New York Stock Exchange | |
Title of each class | Name of each exchange on which registered | |
3.375% Notes due 2025 | New York Stock Exchange | |
2.750% Notes due 2026 | New York Stock Exchange | |
2.875% Notes due 2026 | New York Stock Exchange | |
2.875% Notes due 2029 | New York Stock Exchange | |
3.125% Notes due 2033 | New York Stock Exchange | |
2.000% Notes due 2036 | New York Stock Exchange | |
6.375% Notes due 2038 | New York Stock Exchange | |
4.375% Notes due 2041 | New York Stock Exchange | |
4.500% Notes due 2042 | New York Stock Exchange | |
3.875% Notes due 2042 | New York Stock Exchange | |
4.125% Notes due 2043 | New York Stock Exchange | |
4.875% Notes due 2043 | New York Stock Exchange | |
4.250% Notes due 2044 | New York Stock Exchange | |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes þ No ¨
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No þ
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes þ No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes þ No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer þ | Accelerated filer ¨ | Non-accelerated filer ¨ | Smaller reporting company ¨ |
(Do not check if a smaller reporting company) | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨ No þ
As of June 30, 2016, the aggregate market value of the registrant’s common stock held by non-affiliates of the registrant was approximately $158 billion based on the closing sale price of the common stock as reported on the New York Stock Exchange.
Class | Outstanding at | January 31, 2017 | ||
Common Stock, no par value | 1,551,385,884 | shares | ||
DOCUMENTS INCORPORATED BY REFERENCE
Document | Parts Into Which Incorporated |
Portions of the registrant’s definitive proxy statement for use in connection with its annual meeting of shareholders to be held on May 3, 2017, to be filed with the Securities and Exchange Commission (“SEC”) on or about March 23, 2017. | Part III |
TABLE OF CONTENTS
Page | |||
Item 1. | |||
Item 1A. | |||
Item 1B. | |||
Item 2. | |||
Item 3. | |||
Item 4. | |||
Item 5. | |||
Item 6. | |||
Item 7. | |||
Item 7A. | |||
Item 8. | |||
Item 9. | |||
Item 9A. | |||
Item 9B. | |||
Item 10. | |||
Item 11. | |||
Item 12. | |||
Item 13. | |||
Item 14. | |||
Item 15. | |||
In this report, “PMI,” “we,” “us” and “our” refers to Philip Morris International Inc. and its subsidiaries.
PART I
Item 1. | Business. |
(a) General Development of Business
General
Philip Morris International Inc. is a Virginia holding company incorporated in 1987. Our subsidiaries and affiliates and their licensees are engaged in the manufacture and sale of cigarettes, other tobacco products and other nicotine-containing products in markets outside of the United States of America. Our products are sold in more than 180 markets, and in many of these markets they hold the number one or number two market share position. We have a wide range of premium, mid-price and low-price brands. Our portfolio comprises both international and local brands.
Our portfolio of international and local brands is led by Marlboro, the world’s best-selling international cigarette, which accounted for approximately 35% of our total 2016 shipment volume. Marlboro is complemented in the premium-price category by Parliament and Virginia S. Our leading mid-price brands are L&M, Lark, Merit, Muratti and Philip Morris. Other leading international brands include Bond Street, Chesterfield, Next and Red & White.
We also own a number of important local cigarette brands, such as Dji Sam Soe, Sampoerna and U Mild in Indonesia; Champion, Fortune and Jackpot in the Philippines; Apollo-Soyuz and Optima in Russia; Morven Gold in Pakistan; Boston in Colombia; Belmont, Canadian Classics and Number 7 in Canada; f6 in Germany; Delicados in Mexico; Assos in Greece, and Petra in the Czech Republic and Slovakia. While there are a number of markets where local brands remain important, international brands are expanding their share in numerous markets. With international brands contributing approximately 77% of our shipment volume in 2016, we are well positioned to continue to benefit from this trend.
In addition to the manufacture and sale of cigarettes and other tobacco products, we are engaged in the development and commercialization of Reduced-Risk Products ("RRPs"). RRPs is the term we use to refer to products that present, are likely to present, or have the potential to present less risk of harm to smokers who switch to these products versus continued smoking. We have a range of RRPs in various stages of development, scientific assessment and commercialization. Because our RRPs do not burn tobacco, they produce far lower quantities of harmful and potentially harmful compounds than found in cigarette smoke. Further details on our RRPs are described in the Research and Development section below and Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations of this Annual Report on Form 10-K ("Item 7"), Business Environment — Reduced-Risk Products.
Separation from Altria Group, Inc.
We were a wholly owned subsidiary of Altria Group, Inc. ("Altria") until the distribution of all of our shares owned by Altria (the “Spin-off”) was made on March 28, 2008 (the "Distribution Date").
Acquisitions and Other Business Arrangements
Details about our acquisitions and other business arrangements are described in Item 8. Financial Statements and Supplementary Data of this Annual Report on Form 10-K ("Item 8") in Note 6. Acquisitions and Other Business Arrangements to our consolidated financial statements.
Source of Funds — Dividends
We are a legal entity separate and distinct from our direct and indirect subsidiaries. Accordingly, our right, and thus the right of our creditors and stockholders, to participate in any distribution of the assets or earnings of any subsidiary is subject to the prior rights of creditors of such subsidiary, except to the extent that claims of our company itself as a creditor may be recognized. As a holding company, our principal sources of funds, including funds to make payment on our debt securities, are from the receipt of dividends and repayment of debt from our subsidiaries. Our principal wholly owned and majority-owned subsidiaries currently are not limited by long-term debt or other agreements in their ability to pay cash dividends or to make other distributions with respect to their common stock.
(b) Financial Information About Segments
We divide our markets into four geographic regions, which constitute our segments for financial reporting purposes:
1
• | The European Union (“EU”) Region is headquartered in Lausanne, Switzerland, and covers all the EU countries and also comprises Switzerland, Norway and Iceland, which are linked to the EU through trade agreements; |
• | The Eastern Europe, Middle East & Africa (“EEMA”) Region is also headquartered in Lausanne and includes Eastern Europe, certain Balkan countries, Turkey, the Middle East and Africa and our international duty free business; |
• | The Asia Region is headquartered in Hong Kong and covers all other Asian markets as well as Australia, New Zealand and the Pacific Islands; and |
• | The Latin America & Canada Region is headquartered in New York and covers the South American continent, Central America, Mexico, the Caribbean and Canada. |
Net revenues and operating companies income* (together with a reconciliation to operating income) attributable to each segment for each of the last three years are set forth in Note 12. Segment Reporting to the consolidated financial statements in Item 8. See Item 7 of this Annual Report on Form 10-K for a discussion of our operating results by business segment.
The relative percentages of operating companies income attributable to each reportable segment were as follows:
2016 | 2015 | 2014 | ||||||
European Union | 35.8 | % | 32.6 | % | 31.6 | % | ||
Eastern Europe, Middle East & Africa | 27.1 | 31.2 | 33.5 | |||||
Asia | 28.7 | 26.3 | 26.4 | |||||
Latin America & Canada | 8.4 | 9.9 | 8.5 | |||||
100.0 | % | 100.0 | % | 100.0 | % | |||
______________________________
* | Our management evaluates segment performance and allocates resources based on operating companies income, which we define as operating income, excluding general corporate expenses and amortization of intangibles, plus equity (income)/loss in unconsolidated subsidiaries, net. The accounting policies of the segments are the same as those described in Note 2. Summary of Significant Accounting Policies to the consolidated financial statements in Item 8. |
We use the term net revenues to refer to our operating revenues from the sale of our products, net of sales and promotion incentives. Our net revenues and operating income are affected by various factors, including the volume of products we sell, the price of our products, changes in currency exchange rates and the mix of products we sell. Mix is a term used to refer to the proportionate value of premium-price brands to mid-price or low-price brands in any given market (product mix). Mix can also refer to the proportion of shipment volume in more profitable markets versus shipment volume in less profitable markets (geographic mix). We often collect excise taxes from our customers and then remit them to local governments, and, in those circumstances, we include excise taxes in our net revenues and excise taxes on products. Our cost of sales consists principally of tobacco leaf, non-tobacco raw materials, labor and manufacturing costs.
Our marketing, administration and research costs include the costs of marketing and selling our products, other costs generally not related to the manufacture of our products (including general corporate expenses), and costs incurred to develop new products. The most significant components of our marketing, administration and research costs are marketing and sales expenses and general and administrative expenses.
(c) Narrative Description of Business
Our subsidiaries and affiliates and their licensees are engaged in the manufacture, market and sale of cigarettes, other tobacco products and other nicotine-containing products in markets outside the United States of America.
Our total cigarette shipments decreased by 4.1% in 2016 to 812.9 billion units. We estimate that international cigarette market shipments were approximately 5.3 trillion units in 2016, a 3.0% decrease over 2015. We estimate that our reported share of the international cigarette market (which is defined as worldwide cigarette volume, excluding the United States of America) was approximately 15.3% in 2016, 15.5% in 2015 and 15.4% in 2014. Excluding the People’s Republic of China (“PRC”), we estimate that our reported share of the international cigarette market was approximately 27.9%, 28.5%, and 28.3% in 2016, 2015 and 2014, respectively.
Shipments of our principal cigarette brand, Marlboro, decreased by 1.4% in 2016 and represented approximately 9.6% of the international cigarette market, excluding the PRC, in 2016, 9.6% in 2015 and 9.4% in 2014.
We have a cigarette market share of at least 15% and, in a number of instances, substantially more than 15%, in approximately 100 markets, including Algeria, Argentina, Australia, Austria, Belgium, Canada, the Czech Republic, France, Germany, Hong Kong,
2
Indonesia, Israel, Italy, Japan, Korea, Kuwait, Mexico, the Netherlands, Norway, the Philippines, Poland, Portugal, Russia, Saudi Arabia, Spain, Switzerland, Thailand, Turkey and United Arab Emirates.
The term IQOS Consumables refers to the consumable used in our IQOS device. Total shipment volume of IQOS Consumables reached 7.4 billion units, up from 396 million units during the year ended December 31, 2015.
Our other tobacco products ("OTP") primarily include tobacco for roll-your-own and make-your-own cigarettes, pipe tobacco, cigars and cigarillos, and do not include Reduced-Risk Products. Total shipment volume of OTP, in cigarette equivalent units, decreased by 4.5% to 32.6 billion units.
References to total international cigarette market, defined as worldwide cigarette volume excluding the United States, total cigarette market, total market and market shares in this Form 10-K are our tax-paid estimates based on the latest available data from a number of internal and external sources.
Consumer Focused Marketing & Sales
In 2016, we continued to deploy our new strategic framework that combines our marketing and sales expertise with our in-depth knowledge of various sales territories. This framework allows us not only to engage more effectively with our adult smokers but also to enhance the success of our direct and indirect trade partners. The main benefits are:
• | Improved effectiveness of direct adult smoker engagement activities; |
• | More effective communication with our retailers about our brands; |
• | Increased speed, efficiency and widespread availability of our products; and |
• | Distribution and Sales Strategies and Trade Engagement Programs tailored to the individual characteristics of each market. |
The four main types of distribution that we use globally, often simultaneously in a given market, are:
• | Direct Sales and Distribution, where we have set up our own distribution selling directly to the retailers; |
• | Distribution through Independent Distributors who also are distributing other fast-moving consumer goods and are responsible for distribution in a single market; |
• | Exclusive Zonified Distribution, where the distributors are dedicated to us in tobacco products distribution and assigned to exclusive territories within a market, enabling them to get an appropriate return on their investment; and |
• | Distribution through national or regional wholesalers that then supply the retail trade. |
In many markets we also directly supply key accounts, including gas stations, retail chains and supermarkets.
Our distribution and sales systems are supported by sales forces that total approximately 19,800 employees worldwide. Our sales forces are well trained and recognized by trade surveys for their professionalism.
Our products are marketed and promoted through various media and channels, including, where permitted by law, point of sale communications, brand events, access-restricted websites and printed and direct communication to verified adult smokers. Our direct communication with verified adult smokers utilizes mail, e-mail and other electronic communication tools. Promotional activities include, where permitted by law, competitions, invitations to events, interactive programs, consumer premiums and price promotions. To support advertising and promotional activities in the markets, we have a dedicated consumer engagement group that develops innovative engagement tools for adult smokers based on the latest technologies and adult smoker trends. In addition, we developed digital and non-digital engagement programs for retailers.
Our IQOS distribution strategy is tailored to the individual characteristic of each market. We have adopted a scalable strategy for the IQOS commercialization focused on retail experience, guided consumer trials and customer care. We are also developing several ways to connect with adult smokers through digital communication tools such as mobile apps in several markets.
3
Competition
We are subject to highly competitive conditions in all aspects of our business. We compete primarily on the basis of product quality, brand recognition, brand loyalty, taste, innovation, packaging, service, marketing, advertising and retail price. Our competitors include three large international tobacco companies and several regional and local tobacco companies and, in some instances, state-owned tobacco enterprises, principally in Algeria, Egypt, the PRC, Taiwan, Thailand and Vietnam. Industry consolidation and privatizations of state-owned enterprises have led to an overall increase in competitive pressures. Some competitors have different profit and volume objectives, and some international competitors are susceptible to changes in different currency exchange rates. We predominantly sell American blend cigarette brands, such as Marlboro, L&M, Parliament and Chesterfield, which are the most popular across many of our markets. We seek to compete in all profitable retail price categories, although our brand portfolio is weighted towards the premium-price category.
Procurement and Raw Materials
We purchase tobacco leaf of various types, grades and styles throughout the world, mostly through independent tobacco suppliers. We also contract directly with farmers in several countries, including Argentina, Brazil, Colombia, Ecuador, Italy, Kazakhstan, Pakistan, the Philippines and Poland. Direct sourcing from farmers represents approximately 17% of PMI’s global leaf requirements. The largest supplies of tobacco leaf are sourced from Brazil, Indonesia (mostly for domestic use in kretek products), China, Malawi, India, the United States, Mozambique, Argentina, Philippines and Tanzania.
We believe that there is an adequate supply of tobacco leaf in the world markets to satisfy our current and anticipated production requirements.
In addition to tobacco leaf, we purchase a wide variety of direct materials from a total of approximately 450 suppliers. Our top ten suppliers of direct materials combined represent approximately 50% of our total direct materials purchases. The three most significant direct materials that we purchase are printed paper board used in packaging, acetate tow used in filter making and fine paper used in cigarette manufacturing. In addition, the adequate supply and procurement of cloves are of particular importance to our Indonesian business.
Business Environment
Information called for by this Item is hereby incorporated by reference to the paragraphs in Item 7, Business Environment.
Other Matters
Customers
None of our business segments is dependent upon a single customer or a few customers, the loss of which would have a material adverse effect on our consolidated results of operations.
Employees
At December 31, 2016, we employed approximately 79,500 people worldwide, including employees under temporary contracts and hourly paid part-time staff. Our businesses are subject to a number of laws and regulations relating to our relationship with our employees. Generally, these laws and regulations are specific to the location of each business. In addition, in accordance with European Union requirements, we have established a European Works Council composed of management and elected members of our workforce. We believe that our relations with our employees and their representative organizations are excellent.
Executive Officers of the Registrant
The disclosure regarding executive officers is set forth under the heading “Executive Officers as of February 14, 2017” in Item 10. Directors, Executive Officers and Corporate Governance of this Annual Report on Form 10-K ("Item 10").
Research and Development
Our product development is based on the elimination of combustion via tobacco heating and other innovative systems for aerosol generation, which we believe is the most promising path to providing a better choice for those who would otherwise continue to smoke. We recognize that no single product will appeal to all adult smokers. Therefore, we are developing a portfolio of products intended to
4
appeal to a variety of distinct tastes. Four RRP platforms are in various stages of development and commercialization readiness. We describe each of them in more detail in Item 7, Business Environment—Reduced-Risk Products.
The research and development expense for the years ended December 31, 2016, 2015 and 2014, is set forth in Item 8, Note 14. Additional Information to the consolidated financial statements.
Intellectual Property
Our trademarks are valuable assets, and their protection and reputation are essential to us. We own the trademark rights to all of our principal brands, including Marlboro, or have the right to use them in all countries where we use them.
In addition, we have more than 5,900 granted patents worldwide and approximately 6,400 pending patent applications. Our patent portfolio, as a whole, is material to our business. However, no one patent, or group of related patents, is material to us. We also have registered industrial designs and proprietary secrets, technology, know-how, processes and other intellectual property rights that are not registered.
Effective January 1, 2008, PMI entered into an Intellectual Property Agreement with Philip Morris USA Inc. (“PM USA”). The Intellectual Property Agreement governs the ownership of intellectual property between PMI and PM USA. Ownership of the jointly funded intellectual property has been allocated as follows:
• | PMI owns all rights to the jointly funded intellectual property outside the United States, its territories and possessions; and |
• | PM USA owns all rights to the jointly funded intellectual property in the United States, its territories and possessions. |
Ownership of intellectual property related to patent applications and resulting patents based solely on the jointly funded intellectual property, regardless of when filed or issued, will be exclusive to PM USA in the United States, its territories and possessions and exclusive to PMI everywhere else.
The Intellectual Property Agreement contains provisions concerning intellectual property that is independently developed by us or PM USA following the Distribution Date. For ten years following the Distribution Date, independently developed intellectual property may be subject to rights under certain circumstances that would allow either us or PM USA a priority position to obtain the rights to the new intellectual property from the other party, with the price and other commercial terms to be negotiated.
In the event of a dispute between us and PM USA under the Intellectual Property Agreement, we have agreed with PM USA to submit the dispute first to negotiation between our and PM USA’s senior executives and then to binding arbitration.
Seasonality
Our business segments are not significantly affected by seasonality, although in certain markets cigarette consumption trends rise during the summer months due to longer daylight time and tourism.
Environmental Regulation
We are subject to international, national and local environmental laws and regulations in the countries in which we do business. We have specific programs across our business units designed to meet applicable environmental compliance requirements and reduce our carbon footprint and wastage as well as water and energy consumption. We report externally about our climate change mitigation strategy, together with associated targets and results in reducing our carbon footprint, through CDP (formerly, the Carbon Disclosure Project), the leading international non-governmental organization assessing the work of thousands of companies worldwide in the area of climate change. We have developed and implemented a consistent environmental and occupational health, safety and security management system ("EHSS"), which involves policies, standard practices and procedures at all our manufacturing centers. We also conduct regular safety assessments at our offices, warehouses and car fleet organizations. Furthermore, we have engaged an external certification body to validate the effectiveness of our EHSS management system at our manufacturing centers around the world, in accordance with internationally recognized standards for safety and environmental management. The environmental performance data we report externally is also verified by a qualified third party. Our subsidiaries expect to continue to make investments in order to drive improved performance and maintain compliance with environmental laws and regulations. We assess and report the compliance status of all our legal entities on a regular basis. Based on the management and controls we have in place and our review of climate change risks (both physical and regulatory), environmental expenditures have not had, and are not expected to have, a material adverse effect on our consolidated results of operations, capital expenditures, financial position, earnings or competitive position.
5
(d) Financial Information About Geographic Areas
The amounts of net revenues and long-lived assets attributable to each of our geographic segments for each of the last three fiscal years are set forth in Item 8, Note 12. Segment Reporting to the consolidated financial statements.
(e) Available Information
We are required to file with the SEC annual, quarterly and current reports, proxy statements and other information required by the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Investors may read and copy any document that we file, including this Annual Report on Form 10-K, at the SEC’s Public Reference Room at 100 F Street, NE, Washington, D.C. 20549. Investors may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. In addition, the SEC maintains an Internet website at http://www.sec.gov that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC, from which investors can electronically access our SEC filings.
We make available free of charge on, or through, our website at www.pmi.com our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. Investors can access our filings with the SEC by visiting www.pmi.com.
The information on our website is not, and shall not be deemed to be, a part of this report or incorporated into any other filings we make with the SEC.
Item 1A. Risk Factors.
The following risk factors should be read carefully in connection with evaluating our business and the forward-looking statements contained in this Annual Report on Form 10-K. Any of the following risks could materially adversely affect our business, our operating results, our financial condition and the actual outcome of matters as to which forward-looking statements are made in this Annual Report on Form 10-K.
Forward-Looking and Cautionary Statements
We may from time to time make written or oral forward-looking statements, including statements contained in this Annual Report on Form 10-K and other filings with the SEC, in reports to stockholders and in press releases and investor webcasts. You can identify these forward-looking statements by use of words such as "strategy," "expects," "continues," "plans," "anticipates," "believes," "will," "estimates," "intends," "projects," "goals," "targets" and other words of similar meaning. You can also identify them by the fact that they do not relate strictly to historical or current facts.
We cannot guarantee that any forward-looking statement will be realized, although we believe we have been prudent in our plans and assumptions. Achievement of future results is subject to risks, uncertainties and inaccurate assumptions. Should known or unknown risks or uncertainties materialize, or should underlying assumptions prove inaccurate, actual results could vary materially from those anticipated, estimated or projected. Investors should bear this in mind as they consider forward-looking statements and whether to invest in or remain invested in our securities. In connection with the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, we are identifying important factors that, individually or in the aggregate, could cause actual results and outcomes to differ materially from those contained in any forward-looking statements made by us; any such statement is qualified by reference to the following cautionary statements. We elaborate on these and other risks we face throughout this document, particularly in Item 7, Business Environment. You should understand that it is not possible to predict or identify all risk factors. Consequently, you should not consider the following to be a complete discussion of all potential risks or uncertainties. We do not undertake to update any forward-looking statement that we may make from time to time, except in the normal course of our public disclosure obligations.
6
Risks Related to Our Business and Industry
• | Consumption of tax-paid cigarettes continues to decline in many of our markets. |
This decline is due to multiple factors, including increased taxes and pricing, governmental actions, the diminishing social acceptance of smoking, continuing economic and geopolitical uncertainty, and the continuing prevalence of illicit products. These factors and their potential consequences are discussed more fully below and in Item 7, Business Environment.
• | Cigarettes are subject to substantial taxes. Significant increases in cigarette-related taxes have been proposed or enacted and are likely to continue to be proposed or enacted in numerous jurisdictions. These tax increases may disproportionately affect our profitability and make us less competitive versus certain of our competitors. |
Tax regimes, including excise taxes, sales taxes and import duties, can disproportionately affect the retail price of cigarettes versus other tobacco products, or disproportionately affect the relative retail price of our cigarette brands versus cigarette brands manufactured by certain of our competitors. Because our portfolio is weighted toward the premium-price cigarette category, tax regimes based on sales price can place us at a competitive disadvantage in certain markets. As a result, our volume and profitability may be adversely affected in these markets.
Increases in cigarette taxes are expected to continue to have an adverse impact on our sales of cigarettes, due to resulting lower consumption levels, a shift in sales from manufactured cigarettes to other tobacco products and from the premium-price to the mid-price or low-price cigarette categories, where we may be under-represented, from local sales to legal cross-border purchases of lower price products, or to illicit products such as contraband, counterfeit and "illicit whites."
• | Our business faces significant governmental action aimed at increasing regulatory requirements with the goal of reducing or preventing the use of tobacco products. |
Governmental actions, combined with the diminishing social acceptance of smoking and private actions to restrict smoking, have resulted in reduced industry volume in many of our markets, and we expect that such factors will continue to reduce consumption levels and will increase down-trading and the risk of counterfeiting, contraband, "illicit whites" and legal cross-border purchases. Significant regulatory developments will take place over the next few years in most of our markets, driven principally by the World Health Organization's Framework Convention on Tobacco Control (“FCTC”). The FCTC is the first international public health treaty on tobacco, and its objective is to establish a global agenda for tobacco regulation. The FCTC has led to increased efforts by tobacco control advocates and public health organizations to promote increasingly restrictive regulatory measures on the marketing and sale of tobacco products to adult smokers. Regulatory initiatives that have been proposed, introduced or enacted include:
• | restrictions on or licensing of outlets permitted to sell cigarettes; |
• | the levying of substantial and increasing tax and duty charges; |
• | restrictions or bans on advertising, marketing and sponsorship; |
• | the display of larger health warnings, graphic health warnings and other labeling requirements; |
• | restrictions on packaging design, including the use of colors, and plain packaging; |
• | restrictions on packaging and cigarette formats and dimensions; |
• | restrictions or bans on the display of tobacco product packaging at the point of sale and restrictions or bans on cigarette vending machines; |
• | requirements regarding testing, disclosure and performance standards for tar, nicotine, carbon monoxide and other smoke constituents; |
• | disclosure, restrictions, or bans of tobacco product ingredients; |
• | increased restrictions on smoking in public and work places and, in some instances, in private places and outdoors; |
• | restrictions on the sale of novel tobacco or nicotine-containing products; |
• | elimination of duty free sales and duty free allowances for travelers; and |
• | encouraging litigation against tobacco companies. |
Our operating income could be significantly affected by regulatory initiatives resulting in a significant decrease in demand for our brands, in particular requirements that lead to a commoditization of tobacco products, as well as any significant increase in the cost of complying with new regulatory requirements.
7
• | Litigation related to tobacco use and exposure to environmental tobacco smoke could substantially reduce our profitability and could severely impair our liquidity. |
There is litigation related to tobacco products pending in certain jurisdictions. Damages claimed in some tobacco-related litigation are significant and, in certain cases in Brazil, Canada and Nigeria, range into the billions of U.S. dollars. We anticipate that new cases will continue to be filed. The FCTC encourages litigation against tobacco product manufacturers. It is possible that our consolidated results of operations, cash flows or financial position could be materially affected in a particular fiscal quarter or fiscal year by an unfavorable outcome or settlement of certain pending litigation. See Item 3. Legal Proceedings and Item 8, Financial Statements and Supplementary Data, Note 19. Contingencies for a discussion of pending litigation.
• | We face intense competition, and our failure to compete effectively could have a material adverse effect on our profitability and results of operations. |
We compete primarily on the basis of product quality, brand recognition, brand loyalty, taste, innovation, packaging, service, marketing, advertising and price. We are subject to highly competitive conditions in all aspects of our business. The competitive environment and our competitive position can be significantly influenced by weak economic conditions, erosion of consumer confidence, competitors' introduction of lower-price products or innovative products, higher tobacco product taxes, higher absolute prices and larger gaps between retail price categories, and product regulation that diminishes the ability to differentiate tobacco products. Competitors include three large international tobacco companies and several regional and local tobacco companies and, in some instances, state-owned tobacco enterprises, principally in Algeria, the PRC, Egypt, Taiwan, Thailand and Vietnam. Industry consolidation and privatizations of state-owned enterprises have led to an overall increase in competitive pressures. Some competitors have different profit and volume objectives, and some international competitors are susceptible to changes in different currency exchange rates.
• | Because we have operations in numerous countries, our results may be influenced by economic, regulatory and political developments, natural disasters or conflicts. |
Some of the countries in which we operate face the threat of civil unrest and can be subject to regime changes. In others, nationalization, terrorism, conflict and the threat of war may have a significant impact on the business environment. Economic, political, regulatory or other developments or natural disasters could disrupt our supply chain, manufacturing capabilities or distribution capabilities. In addition, such developments could lead to loss of property or equipment that are critical to our business in certain markets and difficulty in staffing and managing our operations, which could reduce our volumes, revenues and net earnings.
In certain markets, we are dependent on governmental approvals of various actions such as price changes, and failure to obtain such approvals could impair growth in our profitability.
In addition, despite our high ethical standards and rigorous control and compliance procedures aimed at preventing and detecting unlawful conduct, given the breadth and scope of our international operations, we may not be able to detect all potential improper or unlawful conduct by our employees and international partners.
• | We may be unable to anticipate changes in consumer preferences or to respond to consumer behavior influenced by economic downturns. |
Our tobacco business is subject to changes in consumer preferences, which may be influenced by local economic conditions. To be successful, we must:
• | promote brand equity successfully; |
• | anticipate and respond to new consumer trends; |
• | develop new products and markets and broaden brand portfolios; |
• | improve productivity; |
• | ensure adequate production capacity to meet demand for our products; and |
• | be able to protect or enhance margins through price increases. |
In periods of economic uncertainty, consumers may tend to purchase lower-price brands, and the volume of our premium-price and mid-price brands and our profitability could suffer accordingly. Such down-trading trends may be reinforced by regulation that limits branding, communication and product differentiation.
8
• | We lose revenues as a result of counterfeiting, contraband, cross-border purchases and non-tax-paid volume produced by local manufacturers. |
Large quantities of counterfeit cigarettes are sold in the international market. We believe that Marlboro is the most heavily counterfeited international cigarette brand, although we cannot quantify the revenues we lose as a result of this activity. In addition, our revenues are reduced by contraband, legal cross-border purchases and non-tax-paid volume produced by local manufacturers.
• | From time to time, we are subject to governmental investigations on a range of matters. |
Investigations include allegations of contraband shipments of cigarettes, allegations of unlawful pricing activities within certain markets, allegations of underpayment of customs duties and/or excise taxes, allegations of false and misleading usage of descriptors and allegations of unlawful advertising. We cannot predict the outcome of those investigations or whether additional investigations may be commenced, and it is possible that our business could be materially affected by an unfavorable outcome of pending or future investigations. See Item 3, Legal Proceedings-Other Litigation and Item 7, Business Environment-Governmental Investigations for a description of certain governmental investigations to which we are subject.
• | We may be unsuccessful in our attempts to introduce Reduced-Risk Products, and regulators may not permit the commercialization of these products or health-related claims. |
Our key strategic priorities are: to develop and commercialize products that present less risk of harm to adult smokers who switch to those products versus continued smoking; and to convince current adult smokers who would otherwise continue to smoke to switch to those RRPs. For our efforts to be successful, we must: develop RRPs that such adult smokers find acceptable alternatives to smoking; conduct rigorous scientific studies to substantiate that they reduce exposure to harmful and potentially harmful constituents in smoke and, ultimately, that these products present, are likely to present, or have the potential to present less risk of harm to adult smokers who switch to them versus continued smoking; and effectively advocate for the development of science-based regulatory frameworks for the development and commercialization of RRPs, including communication of scientifically substantiated information to enable adult consumers to make better health choices. We might not succeed in our efforts. If we do not succeed, but others do, we may be at a competitive disadvantage. Furthermore, we cannot predict whether regulators will permit the sale and/or marketing of RRPs with health-related claims. Such restrictions could limit the success of our RRPs.
• | Our reported results could be adversely affected by unfavorable currency exchange rates, and currency devaluations could impair our competitiveness. |
We conduct our business primarily in local currency and, for purposes of financial reporting the local currency results are translated into U.S. dollars based on average exchange rates prevailing during a reporting period. During times of a strengthening U.S. dollar, our reported net revenues and operating income will be reduced because the local currency translates into fewer U.S. dollars. During periods of local economic crises, foreign currencies may be devalued significantly against the U.S. dollar, reducing our margins. Actions to recover margins may result in lower volume and a weaker competitive position.
• | The repatriation of our foreign earnings, changes in the earnings mix, and changes in tax laws may increase our effective tax rate. Our ability to receive payments from foreign subsidiaries or to repatriate royalties and dividends could be restricted by local country currency exchange controls. |
Because we are a U.S. holding company, our most significant source of funds is distributions from our non-U.S. subsidiaries. Under current U.S. tax law, in general we do not pay U.S. taxes on our foreign earnings until they are repatriated to the U.S. as distributions from our non-U.S. subsidiaries. These distributions may result in a residual U.S. tax cost. It may be advantageous to us in certain circumstances to significantly increase the amount of such distributions, which could result in a material increase in our overall effective tax rate under current U.S. tax law. Furthermore, our effective tax rate could be increased due to changes in the earnings mix or applicable tax laws. In addition, certain countries in which we operate have adopted or could institute currency exchange controls that limit or prohibit our local subsidiaries' ability to convert local currency into U.S. dollars or to make payments outside the country. This could subject us to the risks of local currency devaluation and business disruption.
• | Our ability to grow profitability may be limited by our inability to introduce new products, enter new markets or improve our margins through higher pricing and improvements in our brand and geographic mix. |
Our profit growth may suffer if we are unable to introduce new products or enter new markets successfully, to raise prices or to improve the proportion of our sales of higher margin products and in higher margin geographies.
9
• | We may be unable to expand our brand portfolio through successful acquisitions or the development of strategic business relationships. |
One element of our growth strategy is to strengthen our brand portfolio and market positions through selective acquisitions and the development of strategic business relationships. Acquisition and strategic business development opportunities are limited and present risks of failing to achieve efficient and effective integration, strategic objectives and anticipated revenue improvements and cost savings. There is no assurance that we will be able to acquire attractive businesses on favorable terms, or that future acquisitions or strategic business developments will be accretive to earnings.
• | Government mandated prices, production control programs, shifts in crops driven by economic conditions and the impact of climate change may increase the cost or reduce the quality of the tobacco and other agricultural products used to manufacture our products. |
As with other agricultural commodities, the price of tobacco leaf and cloves can be influenced by imbalances in supply and demand, and crop quality can be influenced by variations in weather patterns, including those caused by climate change. Tobacco production in certain countries is subject to a variety of controls, including government mandated prices and production control programs. Changes in the patterns of demand for agricultural products could cause farmers to plant less tobacco. Any significant change in tobacco leaf and clove prices, quality and quantity could affect our profitability and our business.
• | Our ability to implement our strategy of attracting and retaining the best global talent may be impaired by the decreasing social acceptance of cigarette smoking. |
The tobacco industry competes for talent with consumer products and other companies that enjoy greater societal acceptance. As a result, we may be unable to attract and retain the best global talent.
• | The failure of our information systems to function as intended or their penetration by outside parties with the intent to corrupt them could result in business disruption, litigation and regulatory action, and loss of revenue, assets or personal or other sensitive data. |
We use information systems to help manage business processes, collect and interpret business data and communicate internally and externally with employees, suppliers, customers and others. Some of these information systems are managed by third-party service providers. We have backup systems and business continuity plans in place, and we take care to protect our systems and data from unauthorized access. Nevertheless, failure of our systems to function as intended, or penetration of our systems by outside parties intent on extracting or corrupting information or otherwise disrupting business processes, could place us at a competitive disadvantage, result in a loss of revenue, assets or personal or other sensitive data, litigation and regulatory action, cause damage to our reputation and that of our brands and result in significant remediation and other costs to us.
• | We may be required to replace third-party contract manufacturers or service providers with our own resources. |
In certain instances, we contract with third parties to manufacture some of our products or product parts or to provide other services. We may be unable to renew these agreements on satisfactory terms for numerous reasons, including government regulations. Accordingly, our costs may increase significantly if we must replace such third parties with our own resources.
Item 1B. | Unresolved Staff Comments. |
None.
10
Item 2. Properties.
At December 31, 2016, we operated and owned 48 manufacturing facilities and maintained contract manufacturing relationships with 22 third-party manufacturers across 21 markets. In addition, we work with 38 third-party operators in Indonesia who manufacture our hand-rolled cigarettes.
PMI-Owned Manufacturing Facilities
EU (1) | EEMA | Asia | Latin America & Canada | TOTAL | ||||||||||
Fully integrated | 9 | 8 | 9 | 8 | 34 | |||||||||
Make-pack | — | — | 1 | 2 | 3 | |||||||||
Other | 4 | 1 | 3 | 3 | 11 | |||||||||
Total | 13 | 9 | 13 | 13 | 48 | |||||||||
(1) Includes facilities that produced IQOS Consumables in 2016.
In 2016, 27 of our facilities each manufactured over 10 billion cigarettes, of which eight facilities each produced over 30 billion units. Our largest factories are in St. Petersburg and Krasnodar (Russia), Sukorejo and Karawang (Indonesia), Izmir (Turkey), Marikina and Batangas (Philippines), Krakow (Poland), Berlin (Germany), Kharkiv (Ukraine), and Klaipeda (Lithuania). Our smallest factories are mostly in Latin America and Asia, where due to tariff and other constraints we have established small manufacturing units in individual markets. We will continue to optimize our manufacturing base, taking into consideration the evolution of trade blocks.
The plants and properties owned or leased and operated by our subsidiaries are maintained in good condition and are believed to be suitable and adequate for our present needs.
We are integrating IQOS Consumables production into our existing manufacturing facilities in Europe and progressing with our plans to build manufacturing capacity for our other RRP platforms. For further details on our IQOS Consumables capacity, see Item 7, Business Environment-Reduced-Risk Products.
Item 3. | Legal Proceedings. |
Tobacco-Related Litigation
Legal proceedings covering a wide range of matters are pending or threatened against us, and/or our subsidiaries, and/or our indemnitees in various jurisdictions. Our indemnitees include distributors, licensees and others that have been named as parties in certain cases and that we have agreed to defend, as well as to pay costs and some or all of judgments, if any, that may be entered against them. Pursuant to the terms of the Distribution Agreement between Altria Group, Inc. ("Altria") and PMI, PMI will indemnify Altria and Philip Morris USA Inc. ("PM USA"), a U.S. tobacco subsidiary of Altria, for tobacco product claims based in substantial part on products manufactured by PMI or contract manufactured for PMI by PM USA, and PM USA will indemnify PMI for tobacco product claims based in substantial part on products manufactured by PM USA, excluding tobacco products contract manufactured for PMI.
It is possible that there could be adverse developments in pending cases against us and our subsidiaries. An unfavorable outcome or settlement of pending tobacco-related litigation could encourage the commencement of additional litigation.
Damages claimed in some of the tobacco-related litigation are significant and, in certain cases in Brazil, Canada and Nigeria, range into the billions of U.S. dollars. The variability in pleadings in multiple jurisdictions, together with the actual experience of management in litigating claims, demonstrate that the monetary relief that may be specified in a lawsuit bears little relevance to the ultimate outcome. Much of the tobacco-related litigation is in its early stages, and litigation is subject to uncertainty. However, as discussed below, we have to date been largely successful in defending tobacco-related litigation.
We and our subsidiaries record provisions in the consolidated financial statements for pending litigation when we determine that an unfavorable outcome is probable and the amount of the loss can be reasonably estimated. At the present time, while it is reasonably possible that an unfavorable outcome in a case may occur, after assessing the information available to it (i) management has not concluded that it is probable that a loss has been incurred in any of the pending tobacco-related cases; (ii) management is unable to estimate the
11
possible loss or range of loss for any of the pending tobacco-related cases; and (iii) accordingly, no estimated loss has been accrued in the consolidated financial statements for unfavorable outcomes in these cases, if any. Legal defense costs are expensed as incurred.
It is possible that our consolidated results of operations, cash flows or financial position could be materially affected in a particular fiscal quarter or fiscal year by an unfavorable outcome or settlement of certain pending litigation. Nevertheless, although litigation is subject to uncertainty, we and each of our subsidiaries named as a defendant believe, and each has been so advised by counsel handling the respective cases, that we have valid defenses to the litigation pending against us, as well as valid bases for appeal of adverse verdicts. All such cases are, and will continue to be, vigorously defended. However, we and our subsidiaries may enter into settlement discussions in particular cases if we believe it is in our best interests to do so.
To date, no tobacco-related case has been finally resolved in favor of a plaintiff against us, our subsidiaries or indemnitees.
The table below lists the number of tobacco-related cases pending against us and/or our subsidiaries or indemnitees as of February 10, 2017, December 31, 2015 and December 31, 2014:
Type of Case | Number of Cases Pending as of February 10, 2017 | Number of Cases Pending as of December 31, 2015 | Number of Cases Pending as of December 31, 2014 | ||||||
Individual Smoking and Health Cases | 63 | 68 | 63 | ||||||
Smoking and Health Class Actions | 11 | 11 | 11 | ||||||
Health Care Cost Recovery Actions | 16 | 16 | 15 | ||||||
Lights Class Actions | — | — | — | ||||||
Individual Lights Cases | 2 | 3 | 2 | ||||||
Public Civil Actions | 2 | 3 | 2 | ||||||
Since 1995, when the first tobacco-related litigation was filed against a PMI entity, 456 Smoking and Health, Lights, Health Care Cost Recovery, and Public Civil Actions in which we and/or one of our subsidiaries and/or indemnitees were a defendant have been terminated in our favor. Thirteen cases have had decisions in favor of plaintiffs. Nine of these cases have subsequently reached final resolution in our favor and four remain on appeal.
12
The table below lists the verdict and significant post-trial developments in the four pending cases where a verdict was returned in favor of the plaintiff:
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
February 2004 | Brazil/The Smoker Health Defense Association | Class Action | The Civil Court of São Paulo found defendants liable without hearing evidence. In April 2004, the court awarded “moral damages” of R$1,000 (approximately $321) per smoker per full year of smoking plus interest at the rate of 1% per month, as of the date of the ruling. The court did not assess actual damages, which were to be assessed in a second phase of the case. The size of the class was not defined in the ruling. | Defendants appealed to the São Paulo Court of Appeals, which annulled the ruling in November 2008, finding that the trial court had inappropriately ruled without hearing evidence and returned the case to the trial court for further proceedings. In May 2011, the trial court dismissed the claim. Plaintiff appealed the decision. In February 2015, the appellate court unanimously dismissed plaintiff's appeal. In September 2015, plaintiff appealed to the Superior Court of Justice. In addition, the defendants filed a constitutional appeal to the Federal Supreme Tribunal on the basis that plaintiff did not have standing to bring the lawsuit. This appeal is still pending. | ||||
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
May 27, 2015 | Canada/Cecilia Létourneau | Class Action | On May 27, 2015, the Superior Court of the District of Montreal, Province of Quebec ruled in favor of the Létourneau class on liability and awarded a total of CAD 131 million (approximately $100 million) in punitive damages, allocating CAD 46 million (approximately $35 million) to our subsidiary. The trial court ordered defendants to pay the full punitive damage award into a trust within 60 days. The court did not order the payment of compensatory damages. | In June 2015, our subsidiary commenced the appellate process with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make the payment into a trust. In August 2015, plaintiffs filed a motion for security with the Court of Appeal covering both the Létourneau case and the Blais case described below. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling CAD 226 million (approximately $173 million) to cover both the Létourneau and Blais cases. The hearing for the merits appeal took place in November 2016. (See below for further detail.) | ||||
13
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
May 27, 2015 | Canada/Conseil Québécois Sur Le Tabac Et La Santé and Jean-Yves Blais | Class Action | On May 27, 2015, the Superior Court of the District of Montreal, Province of Quebec ruled in favor of the Blais class on liability and found the class members’ compensatory damages totaled approximately CAD 15.5 billion (approximately $11.8 billion), including pre-judgment interest. The trial court awarded compensatory damages on a joint and several liability basis, allocating 20% to our subsidiary (approximately CAD 3.1 billion including pre-judgment interest (approximately $2.4 billion)). The trial court awarded CAD 90,000 (approximately $68,800) in punitive damages, allocating CAD 30,000 (approximately $22,900) to our subsidiary. The trial court ordered defendants to pay CAD 1 billion (approximately $764 million) of the compensatory damage award, CAD 200 million (approximately $153 million) of which is our subsidiary’s portion, into a trust within 60 days. | In June 2015, our subsidiary commenced the appellate process with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make the payment into a trust. In August 2015, plaintiffs filed a motion for security with the Court of Appeal. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling, together with the Létourneau case, CAD 226 million (approximately $173 million). The hearing for the merits appeal took place in November 2016. (See below for further detail.) | ||||
14
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
August 5, 2016 | Argentina/Hugo Lespada | Individual Action | On August 5, 2016, the Civil Court No. 14 - Mar del Plata, issued a verdict in favor of plaintiff, an individual smoker, and awarded him ARS 110,000 (approximately $7,100), plus interest, in compensatory and moral damages. The Court found that our subsidiary failed to warn plaintiff of the risk of becoming addicted to cigarettes. | On August 23, 2016, our subsidiary filed its notice of appeal. | ||||
Pending claims related to tobacco products generally fall within the following categories:
Smoking and Health Litigation: These cases primarily allege personal injury and are brought by individual plaintiffs or on behalf of a class or purported class of individual plaintiffs. Plaintiffs' allegations of liability in these cases are based on various theories of recovery, including negligence, gross negligence, strict liability, fraud, misrepresentation, design defect, failure to warn, breach of express and implied warranties, violations of deceptive trade practice laws and consumer protection statutes. Plaintiffs in these cases seek various forms of relief, including compensatory and other damages, and injunctive and equitable relief. Defenses raised in these cases include licit activity, failure to state a claim, lack of defect, lack of proximate cause, assumption of the risk, contributory negligence, and statute of limitations.
As of February 10, 2017, there were a number of smoking and health cases pending against us, our subsidiaries or indemnitees, as follows:
• | 63 cases brought by individual plaintiffs in Argentina (35), Brazil (16), Canada (2), Chile (5), Costa Rica (2), Italy (1), the Philippines (1) and Scotland (1), compared with 68 such cases on December 31, 2015, and 63 cases on December 31, 2014; and |
• | 11 cases brought on behalf of classes of individual plaintiffs in Brazil (2) and Canada (9), compared with 11 such cases on December 31, 2015 and December 31, 2014. |
In the first class action pending in Brazil, The Smoker Health Defense Association (ADESF) v. Souza Cruz, S.A. and Philip Morris Marketing, S.A., Nineteenth Lower Civil Court of the Central Courts of the Judiciary District of São Paulo, Brazil, filed July 25, 1995, our subsidiary and another member of the industry are defendants. The plaintiff, a consumer organization, is seeking damages for all addicted smokers and former smokers, and injunctive relief. In 2004, the trial court found defendants liable without hearing evidence and awarded “moral damages” of R$1,000 (approximately $321) per smoker per full year of smoking plus interest at the rate of 1% per month, as of the date of the ruling. The court did not award actual damages, which were to be assessed in the second phase of the case. The size of the class was not estimated. Defendants appealed to the São Paulo Court of Appeals, which annulled the ruling in November 2008, finding that the trial court had inappropriately ruled without hearing evidence and returned the case to the trial court for further proceedings. In May 2011, the trial court dismissed the claim. In February 2015, the appellate court unanimously dismissed plaintiff's appeal. In September 2015, plaintiff appealed to the Superior Court of Justice. In addition, the defendants filed a constitutional appeal to the Federal Supreme Tribunal on the basis that plaintiff did not have standing to bring the lawsuit. Both appeals are still pending.
In the second class action pending in Brazil, Public Prosecutor of São Paulo v. Philip Morris Brasil Industria e Comercio Ltda., Civil Court of the City of São Paulo, Brazil, filed August 6, 2007, our subsidiary is a defendant. The plaintiff, the Public Prosecutor of the State of São Paulo, is seeking (i) damages on behalf of all smokers nationwide, former smokers, and their relatives; (ii) damages on behalf of people exposed to environmental tobacco smoke nationwide, and their relatives; and (iii) reimbursement of the health care costs allegedly incurred for the treatment of tobacco-related diseases by all Brazilian States and Municipalities, and the Federal District. In an interim ruling issued in December 2007, the trial court limited the scope of this claim to the State of São Paulo only. In December 2008, the Seventh Civil Court of São Paulo issued a decision declaring that it lacked jurisdiction because the case involved issues similar to the ADESF case discussed above and should be transferred to the Nineteenth Lower Civil Court in São Paulo where the ADESF case is
15
pending. The court further stated that these cases should be consolidated for the purposes of judgment. In April 2010, the São Paulo Court of Appeals reversed the Seventh Civil Court's decision that consolidated the cases, finding that they are based on different legal claims and are progressing at different stages of proceedings. This case was returned to the Seventh Civil Court of São Paulo, and our subsidiary filed its closing arguments in December 2010. In March 2012, the trial court dismissed the case on the merits. In January 2014, the São Paulo Court of Appeals rejected plaintiff’s appeal and affirmed the trial court decision. In July 2014, plaintiff appealed to the Superior Court of Justice.
In the first class action pending in Canada, Cecilia Létourneau v. Imperial Tobacco Ltd., Rothmans, Benson & Hedges Inc. and JTI Macdonald Corp., Quebec Superior Court, Canada, filed in September 1998, our subsidiary and other Canadian manufacturers (Imperial Tobacco Canada Ltd. and JTI-MacDonald Corp.) are defendants. The plaintiff, an individual smoker, sought compensatory and punitive damages for each member of the class who is deemed addicted to smoking. The class was certified in 2005. Trial began in March 2012 and concluded in December 2014. The trial court issued its judgment on May 27, 2015. The trial court found our subsidiary and two other Canadian manufacturers liable and awarded a total of CAD 131 million (approximately $100 million) in punitive damages, allocating CAD 46 million (approximately $35 million) to our subsidiary. The trial court found that defendants violated the Civil Code of Quebec, the Quebec Charter of Human Rights and Freedoms, and the Quebec Consumer Protection Act by failing to warn adequately of the dangers of smoking. The trial court also found that defendants conspired to prevent consumers from learning the dangers of smoking. The trial court further held that these civil faults were a cause of the class members’ addiction. The trial court rejected other grounds of fault advanced by the class, holding that: (i) the evidence was insufficient to show that defendants marketed to youth, (ii) defendants’ advertising did not convey false information about the characteristics of cigarettes, and (iii) defendants did not commit a fault by using the descriptors light or mild for cigarettes with a lower tar delivery. The trial court estimated the size of the addiction class at 918,000 members but declined to award compensatory damages to the addiction class because the evidence did not establish the claims with sufficient accuracy. The trial court ordered defendants to pay the full punitive damage award into a trust within 60 days and found that a claims process to allocate the awarded damages to individual class members would be too expensive and difficult to administer. The trial court ordered a briefing on the proposed process for the distribution of sums remaining from the punitive damage award after payment of attorneys’ fees and legal costs. In June 2015, our subsidiary commenced the appellate process by filing its inscription of appeal of the trial court’s judgment with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust within 60 days notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make the payment into a trust within 60 days. In August 2015, plaintiffs filed a motion with the Court of Appeal seeking security in both the Létourneau case and the Blais case described below. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling CAD 226 million (approximately $173 million), in the form of cash into a court trust or letters of credit, in six equal consecutive quarterly installments of approximately CAD 37.6 million (approximately $28.7 million) beginning in December 2015 through March 2017. See the Blais description for further detail concerning the security order. The Court of Appeal heard oral arguments on the merits appeal in November 2016. Our subsidiary and PMI believe that the findings of liability and damages were incorrect and should ultimately be set aside on any one of many grounds, including the following: (i) holding that defendants violated Quebec law by failing to warn class members of the risks of smoking even after the court found that class members knew, or should have known, of the risks, (ii) finding that plaintiffs were not required to prove that defendants’ alleged misconduct caused injury to each class member in direct contravention of binding precedent, (iii) creating a factual presumption, without any evidence from class members or otherwise, that defendants’ alleged misconduct caused all smoking by all class members, (iv) holding that the addiction class members’ claims for punitive damages were not time-barred even though the case was filed more than three years after a prominent addiction warning appeared on all packages, and (v) awarding punitive damages to punish defendants without proper consideration as to whether punitive damages were necessary to deter future misconduct.
In the second class action pending in Canada, Conseil Québécois Sur Le Tabac Et La Santé and Jean-Yves Blais v. Imperial Tobacco Ltd., Rothmans, Benson & Hedges Inc. and JTI Macdonald Corp., Quebec Superior Court, Canada, filed in November 1998, our subsidiary and other Canadian manufacturers (Imperial Tobacco Canada Ltd. and JTI-MacDonald Corp.) are defendants. The plaintiffs, an anti-smoking organization and an individual smoker, sought compensatory and punitive damages for each member of the class who allegedly suffers from certain smoking-related diseases. The class was certified in 2005. Trial began in March 2012 and concluded in December 2014. The trial court issued its judgment on May 27, 2015. The trial court found our subsidiary and two other Canadian manufacturers liable and found that the class members’ compensatory damages totaled approximately CAD 15.5 billion, including pre-judgment interest (approximately $11.8 billion). The trial court awarded compensatory damages on a joint and several liability basis, allocating 20% to our subsidiary (approximately CAD 3.1 billion, including pre-judgment interest (approximately $2.4 billion)). In addition, the trial court awarded CAD 90,000 (approximately $68,800) in punitive damages, allocating CAD 30,000 (approximately $22,900) to our subsidiary and found that defendants violated the Civil Code of Quebec, the Quebec Charter of Human Rights and Freedoms, and the Quebec Consumer Protection Act by failing to warn adequately of the dangers of smoking. The trial court also found that defendants conspired to prevent consumers from learning the dangers of smoking. The trial court further held that these civil faults were a cause of the class members’ diseases. The trial court rejected other grounds of fault advanced by the class, holding that: (i) the evidence was insufficient to show that defendants marketed to youth, (ii) defendants’ advertising did not convey false information about the characteristics of cigarettes, and (iii) defendants did not commit a fault by using the descriptors light or mild for cigarettes with a lower tar delivery. The
16
trial court estimated the disease class at 99,957 members. The trial court ordered defendants to pay CAD 1 billion (approximately $764 million) of the compensatory damage award into a trust within 60 days, CAD 200 million (approximately $153 million) of which is our subsidiary’s portion and ordered briefing on a proposed claims process for the distribution of damages to individual class members and for payment of attorneys’ fees and legal costs. In June 2015, our subsidiary commenced the appellate process by filing its inscription of appeal of the trial court’s judgment with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust within 60 days notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make an initial payment within 60 days. In August 2015, plaintiffs filed a motion with the Court of Appeal seeking an order that defendants place irrevocable letters of credit totaling CAD 5 billion (approximately $3.8 billion) into trust, to secure the judgments in both the Létourneau and Blais cases. Plaintiffs subsequently withdrew their motion for security against JTI-MacDonald Corp. and proceeded only against our subsidiary and Imperial Tobacco Canada Ltd. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling CAD 226 million (approximately $173 million) to cover both the Létourneau and Blais cases. Such security may take the form of cash into a court trust or letters of credit, in six equal consecutive quarterly installments of approximately CAD 37.6 million (approximately $28.7 million) beginning in December 2015 through March 2017. The Court of Appeal ordered Imperial Tobacco Canada Ltd. to furnish security totaling CAD 758 million (approximately $579 million) in seven equal consecutive quarterly installments of approximately CAD 108 million (approximately $83 million) beginning in December 2015 through June 2017. In December 2016, our subsidiary made its fifth quarterly installment of security for approximately CAD 37.6 million (approximately $28.7 million) into a court trust. This payment is included in other assets on the consolidated balance sheets and in cash used in operating activities in the consolidated statements of cash flows. The Court of Appeal ordered that the security is payable upon a final judgment of the Court of Appeal affirming the trial court’s judgment or upon further order of the Court of Appeal. The Court of Appeal heard oral arguments on the merits appeal in November 2016. Our subsidiary and PMI believe that the findings of liability and damages were incorrect and should ultimately be set aside on any one of many grounds, including the following: (i) holding that defendants violated Quebec law by failing to warn class members of the risks of smoking even after the court found that class members knew, or should have known, of the risks, (ii) finding that plaintiffs were not required to prove that defendants’ alleged misconduct caused injury to each class member in direct contravention of binding precedent, (iii) creating a factual presumption, without any evidence from class members or otherwise, that defendants’ alleged misconduct caused all smoking by all class members, (iv) relying on epidemiological evidence that did not meet recognized scientific standards, and (v) awarding punitive damages to punish defendants without proper consideration as to whether punitive damages were necessary to deter future misconduct.
In the third class action pending in Canada, Kunta v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Winnipeg, Canada, filed June 12, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and chronic obstructive pulmonary disease (“COPD”), severe asthma, and mild reversible lung disease resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, as well as restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. In September 2009, plaintiff's counsel informed defendants that he did not anticipate taking any action in this case while he pursues the class action filed in Saskatchewan (see description of Adams, below).
In the fourth class action pending in Canada, Adams v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Saskatchewan, Canada, filed July 10, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and COPD resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who have smoked a minimum of 25,000 cigarettes and have allegedly suffered, or suffer, from COPD, emphysema, heart disease, or cancer, as well as restitution of profits. Preliminary motions are pending.
In the fifth class action pending in Canada, Semple v. Canadian Tobacco Manufacturers' Council, et al., The Supreme Court (trial court), Nova Scotia, Canada, filed June 18, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges his own addiction to tobacco products and COPD resulting from the use of tobacco products. He is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, as well as restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. No activity in this case is anticipated while plaintiff's counsel pursues the class action filed in Saskatchewan (see description of Adams, above).
In the sixth class action pending in Canada, Dorion v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Alberta, Canada, filed June 15, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and chronic bronchitis and severe sinus infections resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. To date, we, our subsidiaries, and our indemnitees have not been properly served with
17
the complaint. No activity in this case is anticipated while plaintiff's counsel pursues the class action filed in Saskatchewan (see description of Adams, above).
In the seventh class action pending in Canada, McDermid v. Imperial Tobacco Canada Limited, et al., Supreme Court, British Columbia, Canada, filed June 25, 2010, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges his own addiction to tobacco products and heart disease resulting from the use of tobacco products. He is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who were alive on June 12, 2007, and who suffered from heart disease allegedly caused by smoking, their estates, dependents and family members, plus disgorgement of revenues earned by the defendants from January 1, 1954, to the date the claim was filed.
In the eighth class action pending in Canada, Bourassa v. Imperial Tobacco Canada Limited, et al., Supreme Court, British Columbia, Canada, filed June 25, 2010, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, the heir to a deceased smoker, alleges that the decedent was addicted to tobacco products and suffered from emphysema resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who were alive on June 12, 2007, and who suffered from chronic respiratory diseases allegedly caused by smoking, their estates, dependents and family members, plus disgorgement of revenues earned by the defendants from January 1, 1954, to the date the claim was filed. In December 2014, plaintiff filed an amended statement of claim.
In the ninth class action pending in Canada, Suzanne Jacklin v. Canadian Tobacco Manufacturers' Council, et al., Ontario Superior Court of Justice, filed June 20, 2012, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and COPD resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who have smoked a minimum of 25,000 cigarettes and have allegedly suffered, or suffer, from COPD, heart disease, or cancer, as well as restitution of profits. Plaintiff's counsel has indicated that he does not intend to take any action in this case in the near future.
Health Care Cost Recovery Litigation: These cases, brought by governmental and non-governmental plaintiffs, seek reimbursement of health care cost expenditures allegedly caused by tobacco products. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including unjust enrichment, negligence, negligent design, strict liability, breach of express and implied warranties, violation of a voluntary undertaking or special duty, fraud, negligent misrepresentation, conspiracy, public nuisance, defective product, failure to warn, sale of cigarettes to minors, and claims under statutes governing competition and deceptive trade practices. Plaintiffs in these cases seek various forms of relief including compensatory and other damages, and injunctive and equitable relief. Defenses raised in these cases include lack of proximate cause, remoteness of injury, failure to state a claim, adequate remedy at law, “unclean hands” (namely, that plaintiffs cannot obtain equitable relief because they participated in, and benefited from, the sale of cigarettes), and statute of limitations.
As of February 10, 2017, there were 16 health care cost recovery cases pending against us, our subsidiaries or indemnitees in Canada (10), Korea (1) and Nigeria (5), compared with 16 such cases on December 31, 2015 and 15 such cases on December 31, 2014.
In the first health care cost recovery case pending in Canada, Her Majesty the Queen in Right of British Columbia v. Imperial Tobacco Limited, et al., Supreme Court, British Columbia, Vancouver Registry, Canada, filed January 24, 2001, we, our subsidiaries, our indemnitee (PM USA), and other members of the industry are defendants. The plaintiff, the government of the province of British Columbia, brought a claim based upon legislation enacted by the province authorizing the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, resulting from a “tobacco related wrong.” The Supreme Court of Canada has held that the statute is constitutional. We and certain other non-Canadian defendants challenged the jurisdiction of the court. The court rejected the jurisdictional challenge. Pre-trial discovery is ongoing.
In the second health care cost recovery case filed in Canada, Her Majesty the Queen in Right of New Brunswick v. Rothmans Inc., et al., Court of Queen's Bench of New Brunswick, Trial Court, New Brunswick, Fredericton, Canada, filed March 13, 2008, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of New Brunswick based on legislation enacted in the province. This legislation is similar to the law introduced in British Columbia that authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing. In September 2016, the trial court entered a consent order establishing a discovery timetable that contemplates the province of New Brunswick applying by September 2017 for a trial date.
In the third health care cost recovery case filed in Canada, Her Majesty the Queen in Right of Ontario v. Rothmans Inc., et al., Ontario Superior Court of Justice, Toronto, Canada, filed September 29, 2009, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Ontario based on legislation
18
enacted in the province. This legislation is similar to the laws introduced in British Columbia and New Brunswick that authorize the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the fourth health care cost recovery case filed in Canada, Attorney General of Newfoundland and Labrador v. Rothmans Inc., et al., Supreme Court of Newfoundland and Labrador, St. Johns, Canada, filed February 8, 2011, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Newfoundland and Labrador based on legislation enacted in the province that is similar to the laws introduced in British Columbia, New Brunswick and Ontario. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the fifth health care cost recovery case filed in Canada, Attorney General of Quebec v. Imperial Tobacco Limited, et al., Superior Court of Quebec, Canada, filed June 8, 2012, we, our subsidiary, our indemnitee (PM USA), and other members of the industry are defendants. The claim was filed by the government of the province of Quebec based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the sixth health care cost recovery case filed in Canada, Her Majesty in Right of Alberta v. Altria Group, Inc., et al., Supreme Court of Queen's Bench Alberta, Canada, filed June 8, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Alberta based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the seventh health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Manitoba v. Rothmans, Benson & Hedges, Inc., et al., The Queen's Bench, Winnipeg Judicial Centre, Canada, filed May 31, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Manitoba based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in September 2014. Pre-trial discovery is ongoing.
In the eighth health care cost recovery case filed in Canada, The Government of Saskatchewan v. Rothmans, Benson & Hedges Inc., et al., Queen's Bench, Judicial Centre of Saskatchewan, Canada, filed June 8, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Saskatchewan based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in February 2015. Discovery is scheduled to begin in 2017.
In the ninth health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Prince Edward Island v. Rothmans, Benson & Hedges Inc., et al., Supreme Court of Prince Edward Island (General Section), Canada, filed September 10, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Prince Edward Island based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in February 2015. Discovery is scheduled to begin in 2017.
In the tenth health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Nova Scotia v. Rothmans, Benson & Hedges Inc., et al., Supreme Court of Nova Scotia, Canada, filed January 2, 2015, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Nova Scotia based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in July 2015. Discovery is scheduled to begin in 2017.
In the first health care cost recovery case in Nigeria, The Attorney General of Lagos State v. British American Tobacco (Nigeria) Limited, et al., High Court of Lagos State, Lagos, Nigeria, filed March 13, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We are in the process
19
of making challenges to service and the court's jurisdiction. Currently, the case is stayed in the trial court pending the appeals of certain co-defendants relating to service objections.
In the second health care cost recovery case in Nigeria, The Attorney General of Kano State v. British American Tobacco (Nigeria) Limited, et al., High Court of Kano State, Kano, Nigeria, filed May 9, 2007, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We are in the process of making challenges to service and the court's jurisdiction. Currently, the case is stayed in the trial court pending the appeals of certain co-defendants relating to service objections.
In the third health care cost recovery case in Nigeria, The Attorney General of Gombe State v. British American Tobacco (Nigeria) Limited, et al., High Court of Gombe State, Gombe, Nigeria, filed October 17, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. In February 2011, the court ruled that the plaintiff had not complied with the procedural steps necessary to serve us. As a result of this ruling, plaintiff must re-serve its claim. We have not yet been re-served.
In the fourth health care cost recovery case in Nigeria, The Attorney General of Oyo State, et al., v. British American Tobacco (Nigeria) Limited, et al., High Court of Oyo State, Ibadan, Nigeria, filed May 25, 2007, we and other members of the industry are defendants. Plaintiffs seek reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We challenged service as improper. In June 2010, the court ruled that plaintiffs did not have leave to serve the writ of summons on the defendants and that they must re-serve the writ. We have not yet been re-served.
In the fifth health care cost recovery case in Nigeria, The Attorney General of Ogun State v. British American Tobacco (Nigeria) Limited, et al., High Court of Ogun State, Abeokuta, Nigeria, filed February 26, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. In May 2010, the trial court rejected our service objections. We have appealed.
In the health care cost recovery case in Korea, the National Health Insurance Service v. KT&G, et. al., filed April 14, 2014, our subsidiary and other Korean manufacturers are defendants. Plaintiff alleges that defendants concealed the health hazards of smoking, marketed to youth, added ingredients to make their products more harmful and addictive, and misled consumers into believing that Lights cigarettes are safer than regular cigarettes. The National Health Insurance Service seeks to recover approximately $53.7 million allegedly incurred in treating 3,484 patients with small cell lung cancer, squamous cell lung cancer, and squamous cell laryngeal cancer from 2003 to 2012. The case is now in the evidentiary phase.
Lights Cases: These cases, brought by individual plaintiffs, allege that the use of the term “lights” constitutes fraudulent and misleading conduct. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including misrepresentation, deception, and breach of consumer protection laws. Plaintiffs seek various forms of relief including restitution, injunctive relief, and compensatory and other damages. Defenses raised include lack of causation, lack of reliance, assumption of the risk, and statute of limitations.
As of February 10, 2017, there were 2 lights cases brought by individual plaintiffs pending against our subsidiaries or indemnitees in Chile (1) and Italy (1), compared with 3 such cases on December 31, 2015, and 2 such cases on December 31, 2014.
Public Civil Actions: Claims have been filed either by an individual, or a public or private entity, seeking to protect collective or individual rights, such as the right to health, the right to information or the right to safety. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including product defect, concealment, and misrepresentation. Plaintiffs in these cases seek various forms of relief including injunctive relief such as banning cigarettes, descriptors, smoking in certain places and advertising, as well as implementing communication campaigns and reimbursement of medical expenses incurred by public or private institutions.
As of February 10, 2017, there were 2 public civil actions pending against our subsidiaries in Argentina (1) and Venezuela (1), compared with 3 such cases on December 31, 2015, and 2 such cases on December 31, 2014.
In the public civil action in Argentina, Asociación Argentina de Derecho de Danos v. Massalin Particulares S.A., et al., Civil Court of Buenos Aires, Argentina, filed February 26, 2007, our subsidiary and another member of the industry are defendants. The plaintiff, a consumer association, seeks the establishment of a relief fund for reimbursement of medical costs associated with diseases allegedly caused by smoking. Our subsidiary filed its answer in September 2007. In March 2010, the case file was transferred to the Federal Court
20
on Administrative Matters after the Civil Court granted plaintiff's request to add the national government as a co-plaintiff in the case. The case is currently in the evidentiary stage.
In the public civil action in Venezuela, Federation of Consumers and Users Associations (“FEVACU”), et al. v. National Assembly of Venezuela and the Venezuelan Ministry of Health, Constitutional Chamber of the Venezuelan Supreme Court, filed April 29, 2008, we were not named as a defendant, but the plaintiffs published a notice pursuant to court order, notifying all interested parties to appear in the case. In January 2009, our subsidiary appeared in the case in response to this notice. The plaintiffs purport to represent the right to health of the citizens of Venezuela and claim that the government failed to protect adequately its citizens' right to health. The claim asks the court to order the government to enact stricter regulations on the manufacture and sale of tobacco products. In addition, the plaintiffs ask the court to order companies involved in the tobacco industry to allocate a percentage of their “sales or benefits” to establish a fund to pay for the health care costs of treating smoking-related diseases. In October 2008, the court ruled that plaintiffs have standing to file the claim and that the claim meets the threshold admissibility requirements. In December 2012, the court admitted our subsidiary and BAT's subsidiary as interested third parties. In February 2013, our subsidiary answered the complaint.
Other Litigation
The Department of Special Investigations of the government of Thailand ("DSI") conducted an investigation into alleged underpayment by our subsidiary, Philip Morris (Thailand) Limited ("PM Thailand"), of customs duties and excise taxes relating to imports from the Philippines covering the period 2003-2007. On January 18, 2016, the Public Prosecutor filed charges against our subsidiary and seven former and current employees in the Bangkok Criminal Court alleging that PM Thailand and the individual defendants jointly and with the intention to defraud the Thai government, underdeclared import prices of cigarettes to avoid full payment of taxes and duties in connection with 272 import entries of cigarettes from the Philippines during the period of July 2003 to June 2006. The government is seeking a fine of approximately THB 80.8 billion (approximately $2.29 billion). The case is in the pre-trial evidentiary phase. Trials are scheduled to begin during the last quarter of 2017. PM Thailand believes that its declared import prices are in compliance with the Customs Valuation Agreement of the World Trade Organization and Thai law and that the allegations of the Public Prosecutor are inconsistent with several decisions already taken by Thai Customs and other Thai governmental agencies.
The DSI also conducted an investigation into alleged underpayment by PM Thailand of customs duties and excise taxes relating to imports from Indonesia covering the period 2000-2003. On January 26, 2017, the Public Prosecutor filed charges against PM Thailand and its Thai ex-employee in the Bangkok Criminal Court alleging that PM Thailand and its Thai ex-employee jointly and with the intention to defraud the Thai government underdeclared import prices of cigarettes to avoid full payment of taxes and duties in connection with 780 import entries during the period from January 2002 to July 2003. The government is seeking a fine of approximately THB 19.8 billion (approximately $562 million). The first hearing, which will focus on preliminary procedural matters, is scheduled for April 2017. PM Thailand disagrees with the allegations and believes that its declared import prices are in compliance with the Customs Valuation Agreement of the WTO and Thai law.
The South Korean Board of Audit and Inspection (“BAI”) conducted an audit of certain Korean government agencies and the tobacco industry into whether inventory movements ahead of the January 1, 2015 increase of cigarette-related taxes by tobacco companies, including Philip Morris Korea Inc. ("PM Korea"), our South Korean affiliate, were in compliance with South Korean tax laws. In November 2016, the tax authorities completed their audit and assessed allegedly underpaid taxes and penalties. In order to avoid nonpayment financial costs, PM Korea paid the total amount that the tax authorities demanded, namely, approximately KRW 100 billion (approximately $85 million) in December 2016 and approximately KRW 118 billion (approximately $100 million) in January 2017. The total demanded amount is included in other assets, and the January 2017 payment is included in accrued liabilities, in the consolidated balance sheets for the year ended 2016. The December 2016 payment is reflected in cash used in operating activities in the consolidated statements of cash flows for the year ended 2016. In the beginning of 2017, PM Korea received demand notices from other government authorities for other amounts of approximately KRW 54 billion (approximately $46 million) in the aggregate. PM Korea will appeal the assessments. The tax authorities have also referred the matter to the Public Prosecutor, who will further investigate and decide whether to file criminal charges against PM Korea and/or other alleged co-offenders. If the Public Prosecutor decides to prosecute, it may seek up to three times the underpaid tax for company criminal penalties and up to five times the underpaid tax for individual criminal penalties. PM Korea believes that it has paid cigarette-related taxes in compliance with the South Korean tax laws. In addition, the South Korean Ministry of Strategy and Finance (“MOSF”) filed a criminal complaint with the Public Prosecutor against PM Korea and its managing director. In its criminal complaint, the MOSF alleged that PM Korea exceeded the monthly product withdrawal limits that the MOSF had set in its notice. The Public Prosecutor will investigate the MOSF’s criminal complaint and decide whether to prosecute. PM Korea disagrees with the MOSF’s allegations.
21
We are also involved in additional litigation arising in the ordinary course of our business. While the outcomes of these proceedings are uncertain, management does not expect that the ultimate outcomes of other litigation, including any reasonably possible losses in excess of current accruals, will have a material adverse effect on our consolidated results of operations, cash flows or financial position.
Item 4. | Mine Safety Disclosures. |
Not applicable.
PART II
Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities. |
The principal stock exchange on which our common stock (no par value) is listed is the New York Stock Exchange. At January 31, 2017, there were approximately 61,000 holders of record of our common stock.
Our common stock is also listed on the NYSE Euronext in Paris and the SIX Swiss Exchange.
22
Performance Graph
The graph below compares the cumulative total shareholder return on PMI's common stock with the cumulative total return for the same period of PMI's Peer Groups (Old and New) and the S&P 500 Index. The graph assumes the investment of $100 as of December 31, 2011, in PMI common stock (at prices quoted on the New York Stock Exchange) and each of the indices as of the market close and reinvestment of dividends on a quarterly basis.
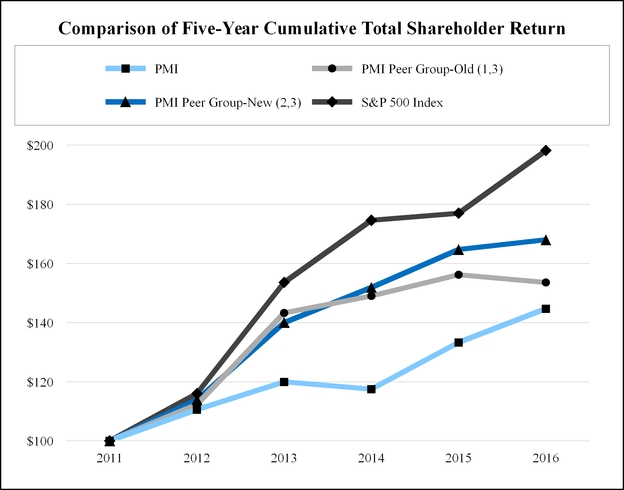
Date | PMI | PMI Peer Group-Old (1,3) | PMI Peer Group-New (2,3) | S&P 500 Index | ||||
December 31, 2011 | $100.00 | $100.00 | $100.00 | $100.00 | ||||
December 31, 2012 | $110.60 | $112.30 | $113.90 | $116.00 | ||||
December 31, 2013 | $120.00 | $143.30 | $140.00 | $153.60 | ||||
December 31, 2014 | $117.50 | $149.00 | $151.90 | $174.60 | ||||
December 31, 2015 | $133.30 | $156.20 | $164.70 | $177.00 | ||||
December 31, 2016 | $144.70 | $153.60 | $168.00 | $198.20 | ||||
(1) The PMI Peer Group-Old (formerly, the PMI Compensation Survey Group) consists of the following companies with substantial global sales that are direct competitors; or have similar market capitalization; or are primarily focused on consumer products (excluding high technology and financial services); and are companies for which comparative executive compensation data are readily available: Bayer AG, British American Tobacco p.l.c., The Coca-Cola Company, Diageo plc, GlaxoSmithKline, Heineken N.V., Imperial Brands PLC (formerly, Imperial Tobacco Group PLC), Johnson & Johnson, McDonald's Corp., Mondelēz International, Inc., Nestlé S.A., Novartis AG, PepsiCo, Inc., Pfizer Inc., Roche Holding AG, Unilever NV and PLC and Vodafone Group Plc.
(2) In 2016, PMI determined that it should use a single customized peer group both to benchmark its compensation programs and to compare its Total Shareholder Return when calculating its Performance Share Unit performance factor. The new Peer Group was established based on a review of four characteristics: global presence; a focus on consumer products; and net revenues and a market capitalization of a similar size to those of PMI. The review also considered the primary international tobacco companies. As a result of this review, Altria Group, Inc., Anheuser-Busch InBev SA/NV, Colgate-Palmolive Co., Japan Tobacco Inc., Kimberly-Clark Corporation, The Kraft-Heinz Company, The Procter & Gamble Company and Reynolds American Inc. were added to the new Peer Group and Bayer AG, GlaxoSmithKline, Novartis AG, Pfizer Inc. and Vodafone Group Plc. were removed.
23
(3) On October 1, 2012, Mondelēz International, Inc., formerly Kraft Foods Inc., announced that it had completed the spin-off of its North American grocery business, Kraft Foods Group, Inc. Mondelēz International, Inc. was retained in the PMI Peer Group-Old (formerly, PMI Compensation Survey Group) and in the PMI Peer Group-New indices because of its global footprint. The PMI Peer Group-Old and in the PMI Peer Group-New indices total cumulative return calculation weight Mondelēz International, Inc.'s total shareholder return at 65% of historical Kraft Foods Inc.'s market capitalization on December 31, 2011, based on Mondelēz International, Inc.'s initial market capitalization relative to the combined market capitalization of Mondelēz International, Inc. and Kraft Foods Group, Inc. in October 2012. Although included in the PMI Peer Group-New, Kraft Heinz Company is not included in the total cumulative return calculation, having become publicly traded only in October 2012.
Note: Figures are rounded to the nearest $0.10.
Issuer Purchases of Equity Securities During the Quarter Ended December 31, 2016
Our share repurchase activity for each of the three months in the quarter ended December 31, 2016, was as follows:
Period | Total Number of Shares Repurchased | Average Price Paid per Share | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs (2) | Approximate Dollar Value of Shares that May Yet be Purchased Under the Plans or Programs | ||||||||||
October 1, 2016 – October 31, 2016 (1) | — | $ | — | — | $ | — | ||||||||
November 1, 2016 – November 30, 2016 (1) | — | $ | — | — | $ | — | ||||||||
December 1, 2016 – December 31, 2016 (1) | — | $ | — | — | $ | — | ||||||||
Pursuant to Publicly Announced Plans or Programs | — | $ | — | |||||||||||
October 1, 2016 – October 31, 2016 (3) | 424 | $ | 97.91 | |||||||||||
November 1, 2016 – November 30, 2016 (3) | 1,144 | $ | 96.18 | |||||||||||
December 1, 2016 – December 31, 2016 (3) | 20,088 | $ | 91.70 | |||||||||||
For the Quarter Ended December 31, 2016 | 21,656 | $ | 92.06 | |||||||||||
(1) | Our authorized three-year share repurchase program of $18 billion expired in August 2015. During this reporting period, we did not have an authorized share repurchase program. |
(2) | Aggregate number of shares repurchased under the above-mentioned share repurchase program as of the end of the period presented. |
(3) | Shares repurchased represent shares tendered to us by employees who vested in restricted share unit awards and used shares to pay all, or a portion of, the related taxes. |
The other information called for by this Item is included in Item 8, Note 23. Quarterly Financial Data (Unaudited) to the consolidated financial statements.
24
Item 6. Selected Financial Data
(in millions of dollars, except per share data)
2016 | 2015 | 2014 | 2013 | 2012 | |||||||||||||||
Summary of Operations: | |||||||||||||||||||
Net revenues | $ | 74,953 | $ | 73,908 | $ | 80,106 | $ | 80,029 | $ | 77,393 | |||||||||
Cost of sales | 9,391 | 9,365 | 10,436 | 10,410 | 10,373 | ||||||||||||||
Excise taxes on products | 48,268 | 47,114 | 50,339 | 48,812 | 46,016 | ||||||||||||||
Gross profit | 17,294 | 17,429 | 19,331 | 20,807 | 21,004 | ||||||||||||||
Operating income | 10,815 | 10,623 | 11,702 | 13,515 | 13,863 | ||||||||||||||
Interest expense, net | 891 | 1,008 | 1,052 | 973 | 859 | ||||||||||||||
Earnings before income taxes | 9,924 | 9,615 | 10,650 | 12,542 | 13,004 | ||||||||||||||
Pre-tax profit margin | 13.2 | % | 13.0 | % | 13.3 | % | 15.7 | % | 16.8 | % | |||||||||
Provision for income taxes | 2,768 | 2,688 | 3,097 | 3,670 | 3,833 | ||||||||||||||
Net earnings | 7,250 | 7,032 | 7,658 | 8,850 | 9,154 | ||||||||||||||
Net earnings attributable to noncontrolling interests | 283 | 159 | 165 | 274 | 354 | ||||||||||||||
Net earnings attributable to PMI | 6,967 | 6,873 | 7,493 | 8,576 | 8,800 | ||||||||||||||
Basic earnings per share | 4.48 | 4.42 | 4.76 | 5.26 | 5.17 | ||||||||||||||
Diluted earnings per share | 4.48 | 4.42 | 4.76 | 5.26 | 5.17 | ||||||||||||||
Dividends declared per share | 4.12 | 4.04 | 3.88 | 3.58 | 3.24 | ||||||||||||||
Capital expenditures | 1,172 | 960 | 1,153 | 1,200 | 1,056 | ||||||||||||||
Depreciation and amortization | 743 | 754 | 889 | 882 | 898 | ||||||||||||||
Property, plant and equipment, net | 6,064 | 5,721 | 6,071 | 6,755 | 6,645 | ||||||||||||||
Inventories | 9,017 | 8,473 | 8,592 | 9,846 | 8,949 | ||||||||||||||
Total assets | 36,851 | 33,956 | 35,187 | 38,168 | 37,670 | ||||||||||||||
Long-term debt | 25,851 | 25,250 | 26,929 | 24,023 | 17,639 | ||||||||||||||
Total debt | 29,067 | 28,480 | 29,455 | 27,678 | 22,839 | ||||||||||||||
Stockholders' deficit | (10,900 | ) | (11,476 | ) | (11,203 | ) | (6,274 | ) | (3,154 | ) | |||||||||
Common dividends declared as a % of Diluted EPS | 92.0 | % | 91.4 | % | 81.5 | % | 68.1 | % | 62.7 | % | |||||||||
Market price per common share — high/low | 104.20-84.46 | 90.27-75.27 | 91.63-75.28 | 96.73-82.86 | 94.13-72.85 | ||||||||||||||
Closing price of common share at year end | 91.49 | 87.91 | 81.45 | 87.13 | 83.64 | ||||||||||||||
Price/earnings ratio at year end — Diluted | 20 | 20 | 17 | 17 | 16 | ||||||||||||||
Number of common shares outstanding at year end (millions) | 1,551 | 1,549 | 1,547 | 1,589 | 1,654 | ||||||||||||||
Number of employees | 79,500 | 80,200 | 82,500 | 91,100 | 87,100 | ||||||||||||||
This Selected Financial Data should be read in conjunction with Item 7 and Item 8.
25
Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations. |
The following discussion should be read in conjunction with the other sections of this Annual Report on Form 10-K, including the consolidated financial statements and related notes contained in Item 8, and the discussion of risks and cautionary factors that may affect future results in Item 1A. Risk Factors.
Description of Our Company
We are a holding company whose subsidiaries and affiliates, and their licensees, are engaged in the manufacture and sale of cigarettes, other tobacco products and other nicotine-containing products in markets outside the United States of America. We manage our business in four segments:
• | European Union; |
• | Eastern Europe, Middle East & Africa (“EEMA”); |
• | Asia; and |
• | Latin America & Canada. |
Our products are sold in more than 180 markets, and in many of these markets they hold the number one or number two market share position. We have a wide range of premium, mid-price and low-price brands. Our portfolio comprises both international and local brands. In addition to the manufacture and sale of cigarettes and other tobacco products, we are engaged in the development and commercialization of Reduced-Risk Products ("RRPs"). RRPs is the term we use to refer to products that present, are likely to present, or have the potential to present less risk of harm to smokers who switch to these products versus continued smoking. We have a range of RRPs in various stages of development, scientific assessment and commercialization. Because our RRPs do not burn tobacco, they produce far lower quantities of harmful and potentially harmful compounds than found in cigarette smoke.
We use the term net revenues to refer to our operating revenues from the sale of our products, net of sales and promotion incentives. Our net revenues and operating income are affected by various factors, including the volume of products we sell, the price of our products, changes in currency exchange rates and the mix of products we sell. Mix is a term used to refer to the proportionate value of premium-price brands to mid-price or low-price brands in any given market (product mix). Mix can also refer to the proportion of shipment volume in more profitable markets versus shipment volume in less profitable markets (geographic mix). We often collect excise taxes from our customers and then remit them to governments, and, in those circumstances, we include the excise taxes in our net revenues and in excise taxes on products. Our cost of sales consists principally of tobacco leaf, non-tobacco raw materials, labor and manufacturing costs.
Our marketing, administration and research costs include the costs of marketing and selling our products, other costs generally not related to the manufacture of our products (including general corporate expenses), and costs incurred to develop new products. The most significant components of our marketing, administration and research costs are marketing and sales expenses and general and administrative expenses.
Philip Morris International Inc. is a legal entity separate and distinct from our direct and indirect subsidiaries. Accordingly, our right, and thus the right of our creditors and stockholders, to participate in any distribution of the assets or earnings of any subsidiary is subject to the prior rights of creditors of such subsidiary, except to the extent that claims of our company itself as a creditor may be recognized. As a holding company, our principal sources of funds, including funds to make payment on our debt securities, are from the receipt of dividends and repayment of debt from our subsidiaries. Our principal wholly owned and majority-owned subsidiaries currently are not limited by long-term debt or other agreements in their ability to pay cash dividends or to make other distributions with respect to their common stock.
26
Executive Summary
The following executive summary provides significant highlights from the Discussion and Analysis that follows.
• | Consolidated Operating Results – The changes in our reported diluted earnings per share (“diluted EPS”) for the year ended December 31, 2016, from the comparable 2015 amounts, were as follows: |
Diluted EPS | % Growth | ||||
For the year ended December 31, 2015 | $ | 4.42 | |||
2015 Asset impairment and exit costs | 0.03 | ||||
2015 Tax items | (0.03 | ) | |||
Subtotal of 2015 items | — | ||||
2016 Asset impairment and exit costs | — | ||||
2016 Tax items | — | ||||
Subtotal of 2016 items | — | ||||
Currency | (0.46 | ) | |||
Interest | 0.05 | ||||
Change in tax rate | 0.03 | ||||
Operations | 0.44 | ||||
For the year ended December 31, 2016 | $ | 4.48 | 1.4 | % | |
See the discussion of events affecting the comparability of statement of earnings amounts in the Consolidated Operating Results section of the following Discussion and Analysis.
• | Asset Impairment and Exit Costs – During 2015, we recorded pre-tax asset impairment and exit costs of $68 million ($52 million after tax or $0.03 per share) related to severance costs for the organizational restructuring in the European Union segment. During 2016, we did not have an EPS impact related to asset impairment and exit costs. |
• | Income Taxes – Our effective income tax rate for 2016 decreased by 0.1 percentage point to 27.9%. The 2015 tax items that increased our diluted EPS by $0.03 per share in 2015 in the table above represents a reduction in unrecognized tax benefits of $41 million following the conclusion of the IRS examinations of Altria Group, Inc.'s ("Altria") consolidated tax returns for the years 2007 and 2008 and PMI's consolidated tax returns for the years 2009 through 2011. Prior to March 28, 2008, PMI was a wholly owned subsidiary of Altria. The change in tax rate that increased our diluted EPS by $0.03 per share in 2016 in the table above was primarily due to earnings mix by taxing jurisdiction and repatriation cost differences. |
• | Currency – The unfavorable currency impact during 2016 results from the strengthening of the U.S. dollar, especially against the Egyptian pound, Indonesian rupiah, Kazakh tenge, Mexican peso, Russian ruble and Turkish lira, partially offset by the Japanese yen. This unfavorable currency movement has impacted our profitability across our primary revenue markets and local currency cost bases. |
• | Interest – The favorable impact of interest was due primarily to lower effective interest rates on debt and higher interest income. |
• | Operations – The increase in diluted EPS of $0.44 from our operations in the table above was due primarily to the following segments: |
• | EEMA: Higher pricing and lower marketing, administration and research costs, partially offset by unfavorable volume/mix; |
• | European Union: Higher pricing, lower manufacturing costs and lower marketing, administration and research costs, partially offset by unfavorable volume/mix; |
• | Asia: Higher pricing and lower marketing, administration and research costs, partially offset by unfavorable volume/mix; and |
• | Latin America & Canada: Higher pricing, partially offset by unfavorable volume/mix, higher manufacturing costs and higher |
27
marketing, administration and research costs.
For further details, see the Consolidated Operating Results and Operating Results by Business Segment sections of the following Discussion and Analysis.
• 2017 Forecasted Results – On February 2, 2017, we announced our forecast for 2017 full-year reported diluted EPS to be in a range of $4.70 to $4.85, at prevailing exchange rates at that time, versus $4.48 in 2016. Excluding an unfavorable currency impact at then-prevailing exchange rates of approximately $0.18 per share for the full-year 2017, the reported diluted earnings per share range represents an increase of approximately 9% to 12% versus adjusted diluted earnings per share of $4.48 in 2016. This forecast reflects net revenue growth, excluding excise taxes, in excess of our current annual growth target range of 4%-6%, excluding currency and acquisitions. This forecast does not include any share repurchases in 2017.
Our 2016 reported and adjusted diluted EPS was $4.48. During 2016, we did not have an EPS impact related to asset impairment and exit costs, and discrete tax items.
Adjusted diluted EPS is not a measure under accounting principles generally accepted in the United States of America ("U.S. GAAP"). We define adjusted diluted EPS as reported diluted EPS adjusted for asset impairment and exit costs, discrete tax items and unusual items. We believe it is appropriate to disclose this measure as it represents core earnings, improves comparability and helps investors analyze business performance and trends. Adjusted diluted EPS should be considered neither in isolation nor as a substitute for reported diluted EPS prepared in accordance with U.S. GAAP.
This 2017 guidance excludes the impact of any future acquisitions, unanticipated asset impairment and exit cost charges, future changes in currency exchange rates and any unusual events. The factors described in Item 1A. Risk Factors represent continuing risks to this forecast.
Discussion and Analysis
Critical Accounting Estimates
Item 8, Note 2. Summary of Significant Accounting Policies to our consolidated financial statements includes a summary of the significant accounting policies and methods used in the preparation of our consolidated financial statements. In most instances, we must use a particular accounting policy or method because it is the only one that is permitted under U.S. GAAP.
The preparation of financial statements requires that we use estimates and assumptions that affect the reported amounts of our assets, liabilities, net revenues and expenses, as well as our disclosure of contingencies. If actual amounts differ from previous estimates, we include the revisions in our consolidated results of operations in the period during which we know the actual amounts. Historically, aggregate differences, if any, between our estimates and actual amounts in any year have not had a significant impact on our consolidated financial statements.
The selection and disclosure of our critical accounting estimates have been discussed with our Audit Committee. The following is a discussion of the more significant assumptions, estimates, accounting policies and methods used in the preparation of our consolidated financial statements:
• Revenue Recognition - We recognize revenue when persuasive evidence of an arrangement exists, delivery of product has occurred, the sales price is fixed or determinable and collectability is reasonably assured. For our company, this means that revenue is recognized when title and risk of loss is transferred to our customers. Title transfers to our customers upon shipment or upon receipt at the customer's location as determined by the sales terms for each transaction. The company estimates the cost of sales returns based on historical experience, and these estimates are immaterial.
• Goodwill and Non-Amortizable Intangible Assets Valuation - We test goodwill and non-amortizable intangible assets for impairment annually or more frequently if events occur that would warrant such review. During the second quarter of 2016, we changed the date of our annual goodwill impairment test from the first quarter to the second quarter. The change was made to more closely align the impairment testing date with our long-range planning and forecasting process. We have determined that this change in accounting principle is preferable under the circumstances and believe that the change in the annual impairment testing date did not delay, accelerate, or avoid an impairment charge. While the company has the option to perform a qualitative assessment for both goodwill and non-amortizable intangible assets to determine if it is more likely than not that an impairment exists, the company elects to perform the quantitative assessment for our annual impairment analysis. The impairment analysis involves comparing the fair value of each reporting
28
unit or non-amortizable intangible asset to the carrying value. If the carrying value exceeds the fair value, goodwill or a non-amortizable intangible asset is considered impaired. To determine the fair value of goodwill, we primarily use a discounted cash flow model, supported by the market approach using earnings multiples of comparable global and local companies within the tobacco industry. At December 31, 2016, the carrying value of our goodwill was $7.3 billion, which is related to ten reporting units, each of which consists of a group of markets with similar economic characteristics. The estimated fair value of each of our ten reporting units exceeded the carrying value as of December 31, 2016. To determine the fair value of non-amortizable intangible assets, we primarily use a discounted cash flow model applying the relief-from-royalty method. We concluded that the fair value of our non-amortizable intangible assets exceeded the carrying value. These discounted cash flow models include management assumptions relevant for forecasting operating cash flows, which are subject to changes in business conditions, such as volumes and prices, costs to produce, discount rates and estimated capital needs. Management considers historical experience and all available information at the time the fair values are estimated, and we believe these assumptions are consistent with the assumptions a hypothetical marketplace participant would use. Since the March 28, 2008, spin-off from Altria, we have not recorded a charge to earnings for an impairment of goodwill or non-amortizable intangible assets.
• Marketing and Advertising Costs - We incur certain costs to support our products through programs that include advertising, marketing, consumer engagement and trade promotions. The costs of our advertising and marketing programs are expensed in accordance with U.S. GAAP. Recognition of the cost related to our consumer engagement and trade promotion programs contain uncertainties due to the judgment required in estimating the potential performance and compliance for each program. For volume-based incentives provided to customers, management continually assesses and estimates, by customer, the likelihood of the customer's achieving the specified targets, and records the reduction of revenue as the sales are made. For other trade promotions, management relies on estimated utilization rates that have been developed from historical experience. Changes in the assumptions used in estimating the cost of any individual marketing program would not result in a material change in our financial position, results of operations or operating cash flows. We have not made any material changes in the accounting methodology used to estimate our marketing programs during the past three years.
• Employee Benefit Plans - As discussed in Item 8, Note 13. Benefit Plans to our consolidated financial statements, we provide a range of benefits to our employees and retired employees, including pensions, postretirement health care and postemployment benefits (primarily severance). We record annual amounts relating to these plans based on calculations specified by U.S. GAAP. These calculations include various actuarial assumptions, such as discount rates, assumed rates of return on plan assets, compensation increases, mortality, turnover rates and health care cost trend rates. We review actuarial assumptions on an annual basis and make modifications to the assumptions based on current rates and trends when it is deemed appropriate to do so. As permitted by U.S. GAAP, any effect of the modifications is generally amortized over future periods. We believe that the assumptions utilized in calculating our obligations under these plans are reasonable based upon our historical experience and advice from our actuaries.
Weighted-average discount rate assumptions for pensions and postretirement plans are as follows:
2016 | 2015 | |
U.S. pension plans | 4.07% | 4.30% |
Non-U.S. pension plans | 1.39% | 1.68% |
Postretirement plans | 3.68% | 4.45% |
We anticipate that assumption changes will decrease 2017 pre-tax U.S. and non-U.S. pension and postretirement expense to approximately $199 million as compared with approximately $215 million in 2016, excluding amounts related to early retirement programs. The anticipated decrease is primarily due to lower interest cost, partially offset by lower expected return on assets.
Weighted-average expected rate of return and discount rate assumptions have a significant effect on the amount of expense reported for the employee benefit plans. A fifty-basis-point decrease in our discount rate would increase our 2017 pension and postretirement expense by approximately $43 million, and a fifty-basis-point increase in our discount rate would decrease our 2017 pension and postretirement expense by approximately $40 million. Similarly, a fifty-basis-point decrease (increase) in the expected return on plan assets would increase (decrease) our 2017 pension expense by approximately $33 million. See Item 8, Note 13. Benefit Plans to our consolidated financial statements for a sensitivity discussion of the assumed health care cost trend rates.
Beginning January 1, 2017, we changed the method used to calculate the service and interest cost components for the net periodic pension benefit costs. Specifically, service costs will be determined based on duration-specific spot rates applied to service cost cash flows and interest costs will be determined by applying duration specific spot rates to the year-by-year projected benefit. The change will not have a material impact to our consolidated results of operations, financial position or cash flows and will be accounted for prospectively as a change in accounting estimate.
• Income Taxes - Income tax provisions for jurisdictions outside the United States, as well as state and local income tax provisions, are determined on a separate company basis, and the related assets and liabilities are recorded in our consolidated balance sheets.
29
The extent of our operations involves dealing with uncertainties and judgments in the application of complex tax regulations in a multitude of jurisdictions. The final taxes paid are dependent upon many factors, including negotiations with taxing authorities in various jurisdictions and resolution of disputes arising from federal, state, and international tax audits. In accordance with the authoritative guidance for income taxes, we evaluate potential tax exposures and record tax liabilities for anticipated tax audit issues based on our estimate of whether, and the extent to which, additional taxes will be due. We adjust these reserves in light of changing facts and circumstances; however, due to the complexity of some of these uncertainties, the ultimate resolution may result in a payment that is materially different from our current estimate of the tax liabilities. If our estimate of tax liabilities proves to be less than the ultimate assessment, an additional charge to expense would result. If payment of these amounts ultimately proves to be less than the recorded amounts, the reversal of the liabilities would result in tax benefits being recognized in the period when we determine the liabilities are no longer necessary.
The effective tax rates used for interim reporting are based on our full-year geographic earnings mix projections and cash repatriation plans. Changes in currency exchange rates, earnings mix by taxing jurisdiction or in cash repatriation plans could have an impact on the effective tax rates, which we monitor each quarter. Significant judgment is required in determining income tax provisions and in evaluating tax positions.
For further details, see Item 8, Note 11. Income Taxes to our consolidated financial statements.
• Hedging - As discussed below in “Market Risk,” we use derivative financial instruments principally to reduce exposures to market risks resulting from fluctuations in foreign currency exchange and interest rates by creating offsetting exposures. For derivatives to which we have elected to apply hedge accounting, gains and losses on these derivatives are initially deferred in accumulated other comprehensive losses on the consolidated balance sheet and recognized in the consolidated statement of earnings in the periods when the related hedged transactions are also recognized in operating results. If we had elected not to use the hedge accounting provisions, gains (losses) deferred in stockholders’ (deficit) equity would have been recorded in our net earnings for these derivatives.
• Contingencies - As discussed in Item 8, Note 19. Contingencies to our consolidated financial statements, legal proceedings covering a wide range of matters are pending or threatened against us, and/or our subsidiaries, and/or our indemnitees in various jurisdictions. We and our subsidiaries record provisions in the consolidated financial statements for pending litigation when we determine that an unfavorable outcome is probable and the amount of the loss can be reasonably estimated. The variability in pleadings in multiple jurisdictions, together with the actual experience of management in litigating claims, demonstrate that the monetary relief that may be specified in a lawsuit bears little relevance to the ultimate outcome. Much of the tobacco-related litigation is in its early stages, and litigation is subject to uncertainty. At the present time, while it is reasonably possible that an unfavorable outcome in a case may occur, after assessing the information available to it: (i) management has not concluded that it is probable that a loss has been incurred in any of the pending tobacco-related cases; (ii) management is unable to estimate the possible loss or range of loss for any of the pending tobacco-related cases; and (iii) accordingly, no estimated loss has been accrued in the consolidated financial statements for unfavorable outcomes in these cases, if any. Legal defense costs are expensed as incurred.
30
Consolidated Operating Results
Our cigarette volume, net revenues, excise taxes on products and operating companies income by segment were as follows:
(in millions) | 2016 | 2015 | 2014 | ||||||||
Cigarette Volume | |||||||||||
European Union | 193,586 | 194,589 | 194,746 | ||||||||
Eastern Europe, Middle East & Africa | 271,393 | 279,411 | 278,374 | ||||||||
Asia | 260,029 | 281,350 | 288,128 | ||||||||
Latin America & Canada | 87,938 | 91,920 | 94,706 | ||||||||
Total cigarette volume | 812,946 | 847,270 | 855,954 | ||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Net Revenues | |||||||||||
European Union | $ | 27,129 | $ | 26,563 | $ | 30,517 | |||||
Eastern Europe, Middle East & Africa | 18,286 | 18,328 | 20,469 | ||||||||
Asia | 20,531 | 19,469 | 19,255 | ||||||||
Latin America & Canada | 9,007 | 9,548 | 9,865 | ||||||||
Net revenues | $ | 74,953 | $ | 73,908 | $ | 80,106 | |||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Excise Taxes on Products | |||||||||||
European Union | $ | 18,967 | $ | 18,495 | $ | 21,370 | |||||
Eastern Europe, Middle East & Africa | 11,286 | 10,964 | 11,855 | ||||||||
Asia | 11,850 | 11,266 | 10,527 | ||||||||
Latin America & Canada | 6,165 | 6,389 | 6,587 | ||||||||
Excise taxes on products | $ | 48,268 | $ | 47,114 | $ | 50,339 | |||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Operating Income | |||||||||||
Operating companies income: | |||||||||||
European Union | $ | 3,994 | $ | 3,576 | $ | 3,815 | |||||
Eastern Europe, Middle East & Africa | 3,016 | 3,425 | 4,033 | ||||||||
Asia | 3,196 | 2,886 | 3,187 | ||||||||
Latin America & Canada | 938 | 1,085 | 1,030 | ||||||||
Amortization of intangibles | (74 | ) | (82 | ) | (93 | ) | |||||
General corporate expenses | (161 | ) | (162 | ) | (165 | ) | |||||
Less: | |||||||||||
Equity (income)/loss in unconsolidated subsidiaries, net | (94 | ) | (105 | ) | (105 | ) | |||||
Operating income | $ | 10,815 | $ | 10,623 | $ | 11,702 | |||||
As discussed in Item 8, Note 12. Segment Reporting to our consolidated financial statements, we evaluate segment performance and allocate resources based on operating companies income, which we define as operating income, excluding general corporate expenses and amortization of intangibles, plus equity (income)/loss in unconsolidated subsidiaries, net. We believe it is appropriate to disclose this measure to help investors analyze the business performance and trends of our various business segments.
31
References to total international cigarette market, defined as worldwide cigarette volume excluding the United States, total cigarette market, total market and market shares throughout this Discussion and Analysis are our tax-paid estimates based on the latest available data from a number of internal and external sources.
The following events that occurred during 2016, 2015 and 2014 affected the comparability of our statement of earnings amounts:
• Asset Impairment and Exit Costs – For the year ended December 31, 2016, we did not incur asset impairment and exit costs. For the years ended December 31, 2015 and 2014, pre-tax asset impairment and exit costs by segment were as follows:
(in millions) | 2015 | 2014 | ||||||
Separation programs: | ||||||||
European Union | $ | 68 | $ | 351 | ||||
Eastern Europe, Middle East & Africa | — | 2 | ||||||
Asia | — | 35 | ||||||
Latin America & Canada | — | 3 | ||||||
Total separation programs | 68 | 391 | ||||||
Asset impairment charges: | ||||||||
European Union | — | 139 | ||||||
Latin America & Canada | — | 5 | ||||||
Total asset impairment charges | — | 144 | ||||||
Asset impairment and exit costs | $ | 68 | $ | 535 | ||||
For further details, see Item 8, Note 5. Asset Impairment and Exit Costs to our consolidated financial statements.
• Acquisitions and Other Business Arrangements – For further details, see Item 8, Note 6. Acquisitions and Other Business Arrangements to our consolidated financial statements.
2016 compared with 2015
The following discussion compares our consolidated operating results for the year ended December 31, 2016, with the year ended December 31, 2015.
Our cigarette shipment volume decreased by 4.1%, or by 4.7% excluding net estimated inventory movements, due to:
• | European Union, principally Italy, Germany and Greece, partly offset by Poland and Spain; |
• | EEMA, mainly North Africa, primarily Algeria, and Russia, partly offset by Saudi Arabia and Ukraine; |
• | Asia, principally Indonesia, Pakistan, the Philippines and Thailand, partly offset by Korea; and |
• | Latin America & Canada, predominantly Argentina, partly offset by Mexico. |
Our cigarette market share increased in a number of markets, including Brazil, Canada, Colombia, the Czech Republic, France, Kuwait, Mexico, the Netherlands, Norway, Poland, Saudi Arabia, Spain, Switzerland, Turkey and the United Arab Emirates.
32
Our cigarette shipment volume by brand is shown in the table below:
PMI Cigarette Shipment Volume by Brand (Million Units) | |||||||
Full-Year | |||||||
2016 | 2015 | Change | |||||
Marlboro | 281,720 | 285,583 | (1.4 | )% | |||
L&M | 96,770 | 97,884 | (1.1 | )% | |||
Parliament | 45,671 | 44,879 | 1.8 | % | |||
Bond Street | 44,567 | 43,608 | 2.2 | % | |||
Chesterfield | 46,291 | 41,397 | 11.8 | % | |||
Philip Morris | 35,914 | 35,815 | 0.3 | % | |||
Lark | 27,571 | 28,828 | (4.4 | )% | |||
Others | 234,442 | 269,276 | (12.9 | )% | |||
Total PMI | 812,946 | 847,270 | (4.1 | )% | |||
Cigarette shipment volume of Marlboro decreased, driven by Algeria, Argentina, Egypt and Vietnam, as well as in-switching from that brand to IQOS Consumables, partly offset by Korea, Mexico, the Philippines, Saudi Arabia and Spain.
Cigarette shipment volume of L&M decreased, notably in Russia, Thailand and Turkey, partly offset by Algeria, Kazakhstan and Ukraine. Cigarette shipment volume of Parliament increased, mainly driven by Korea, Turkey and Ukraine, partly offset by Japan and Russia. Cigarette shipment volume of Bond Street increased, mainly driven by Ukraine, partly offset by Kazakhstan. Cigarette shipment volume of Chesterfield increased, mainly driven by Argentina, the Czech Republic, reflecting the morphing of Red & White, Turkey and the United Kingdom, partly offset by Russia. Cigarette shipment volume of Philip Morris increased, driven mainly by Italy and Russia, partly offset by Argentina. Cigarette shipment volume of Lark decreased, principally due to Japan and Turkey. Cigarette shipment volume of "Others" decreased, mainly due to local, largely low-margin brands in Pakistan, the Philippines, Russia and Ukraine.
Our other tobacco products ("OTP") primarily include tobacco for roll-your-own and make-your-own cigarettes, pipe tobacco, cigars and cigarillos, and do not include Reduced-Risk Products. Total shipment volume of OTP, in cigarette equivalent units, decreased by 4.5% to 32.6 billion units.
Total shipment volume for cigarettes and OTP, in cigarette equivalent units, decreased by 4.1%.
Total shipment volume of IQOS Consumables reached 7.4 billion units, up from 396 million units in 2015.
Our net revenues and excise taxes on products were as follows:
For the Years Ended December 31, | |||||||||||||||
(in millions) | 2016 | 2015 | Variance | % | |||||||||||
Net revenues | $ | 74,953 | $ | 73,908 | $ | 1,045 | 1.4 | % | |||||||
Excise taxes on products | 48,268 | 47,114 | 1,154 | 2.4 | % | ||||||||||
Net revenues, excluding excise taxes on products | $ | 26,685 | $ | 26,794 | $ | (109 | ) | (0.4 | )% | ||||||
Net revenues, which include excise taxes billed to customers, increased by $1.0 billion. Excluding excise taxes, net revenues decreased by $109 million, due to:
• | unfavorable currency ($1.3 billion) and |
• | unfavorable volume/mix ($450 million), partly offset by |
• | price increases ($1.6 billion). |
33
The unfavorable currency was due primarily to the Argentine peso, Canadian dollar, Egyptian pound, Euro, Kazakh tenge, Mexican peso, Philippine peso, Russian ruble and Turkish lira, partially offset by the Japanese yen.
Net revenues include $739 million in 2016 related to sale of RRPs, mainly driven by Japan. This amount includes excise taxes billed to customers. Excluding excise taxes, net revenues for RRPs were $733 million in 2016. In some jurisdictions, including Japan, we are not responsible for collecting excise taxes. Approximately 22% of our $733 million in 2016 RRP net revenues, excluding excise taxes, were from IQOS devices.
Net revenues related to RRPs represent the sale of heat-not-burn consumables, including IQOS Consumables, IQOS devices and related accessories, and other nicotine-containing products, primarily e-vapor products, net of sales and promotion incentives.
Net revenues include $1.8 billion in 2016 and 2015 related to sales of OTP. These net revenue amounts include excise taxes billed to customers. Excluding excises taxes, net revenues for OTP were $644 million in 2016 and $673 million in 2015.
Excise taxes on products increased by $1.2 billion, due to:
• | higher excise taxes resulting from changes in retail prices and tax rates ($5.3 billion), partly offset by |
• | favorable currency ($3.9 billion) and |
• | lower excise taxes resulting from volume/mix ($236 million). |
Governments have consistently increased excise taxes in most of the markets in which we operate. As discussed in Business Environment, we expect excise taxes to continue to increase.
Our cost of sales; marketing, administration and research costs; and operating income were as follows:
For the Years Ended December 31, | |||||||||||||||
(in millions) | 2016 | 2015 | Variance | % | |||||||||||
Cost of sales | $ | 9,391 | $ | 9,365 | $ | 26 | 0.3 | % | |||||||
Marketing, administration and research costs | 6,405 | 6,656 | (251 | ) | (3.8 | )% | |||||||||
Operating income | 10,815 | 10,623 | 192 | 1.8 | % | ||||||||||
Cost of sales increased by $26 million, due to:
• | higher cost of sales resulting from volume/mix ($242 million), partly offset by |
• | favorable currency ($216 million). |
Marketing, administration and research costs decreased by $251 million, due to:
• | lower expenses ($210 million, driven by a favorable comparison to 2015, notably related to cigarette brand building and business optimization initiatives, partly offset by increased support behind Reduced-Risk Products) and |
• | favorable currency ($41 million). |
Operating income increased by $192 million, due primarily to:
• | price increases ($1.6 billion) and |
• | lower marketing, administration and research costs ($210 million) and |
• | the non-recurrence of the 2015 pre-tax charges for asset impairment and exit costs ($68 million), partly offset by |
• | unfavorable currency ($1.0 billion) and |
• | unfavorable volume/mix ($692 million). |
34
Interest expense, net, of $891 million decreased by $117 million, due primarily to lower effective interest rates on debt and higher interest income.
Our effective tax rate decreased by 0.1 percentage point to 27.9%. The 2015 effective tax rate was unfavorably impacted by changes to repatriation assertions on certain foreign subsidiary historical earnings ($58 million), partially offset by a reduction in unrecognized tax benefits of $41 million following the conclusion of the IRS examinations of Altria's consolidated tax returns for the years 2007 and 2008 and PMI's consolidated tax returns for the years 2009 through 2011. Prior to March 28, 2008, PMI was a wholly owned subsidiary of Altria. Changes in currency exchange rates, earnings mix by taxing jurisdiction or in our cash repatriation plans could have an impact on the effective tax rate, which we monitor each quarter. Significant judgment is required in determining income tax provisions and in evaluating tax positions. Based upon tax regulations in existence at December 31, 2016, and our cash repatriation plans, we estimate that our 2017 effective tax rate will be approximately 28%.
We are regularly examined by tax authorities around the world, and we are currently under examination in a number of jurisdictions. It is reasonably possible that within the next 12 months certain tax examinations will close, which could result in a change in unrecognized tax benefits along with related interest and penalties. An estimate of any possible charge cannot be made at this time.
Net earnings attributable to PMI of $7.0 billion increased by $94 million (1.4%). This increase was due primarily to higher operating income as discussed above, and lower interest expense, net. Diluted and basic EPS of $4.48 increased by 1.4%. Excluding an unfavorable currency impact of $0.46, diluted EPS increased by 11.8%.
2015 compared with 2014
The following discussion compares our consolidated operating results for the year ended December 31, 2015, with the year ended December 31, 2014.
Our cigarette shipment volume was down by 1.0%, excluding acquisitions, reflected declines in:
• | Asia, mainly due to Korea, Pakistan and the Philippines; and |
• | Latin America & Canada, mainly due to Argentina, Brazil, Ecuador and Mexico; |
partially offset by growth in:
• | EEMA, notably Egypt, Saudi Arabia and Turkey, partially offset by Kazakhstan and Ukraine. |
Total cigarette volume in the European Union was essentially flat, with declines in Greece, Italy and the United Kingdom largely offset by growth in France, Germany and Spain.
For the year ended December 31, 2015, estimated inventory movements were favorable, driven principally by a favorable comparison in Japan as a result of the 2014 correction of distributor inventory movements partly related to the VAT increase of April 2014. Excluding these estimated inventory movements, our total cigarette shipment volume decreased by 1.6%, excluding acquisitions.
Our cigarette market share increased in a number of key markets, including Argentina, Austria, Belgium, Egypt, France, Germany, Korea, the Netherlands, the Philippines, Poland, Russia, Saudi Arabia, Spain and Switzerland.
35
Our cigarette shipment volume by brand is shown in the table below:
PMI Cigarette Shipment Volume by Brand (Million Units) | |||||||
Full-Year | |||||||
2015 | 2014 | Change | |||||
Marlboro | 285,583 | 282,997 | 0.9 | % | |||
L&M | 97,884 | 94,168 | 3.9 | % | |||
Parliament | 44,879 | 47,199 | (4.9 | )% | |||
Bond Street | 43,608 | 43,585 | 0.1 | % | |||
Chesterfield | 41,397 | 42,144 | (1.8 | )% | |||
Philip Morris | 35,815 | 31,948 | 12.1 | % | |||
Lark | 28,828 | 28,473 | 1.2 | % | |||
Others | 269,276 | 285,440 | (5.7 | )% | |||
Total PMI | 847,270 | 855,954 | (1.0 | )% | |||
The increase in cigarette shipment volume of Marlboro reflected growth in: the European Union, notably France, Germany and Spain, partly offset by Italy and the United Kingdom; EEMA, notably Saudi Arabia and Turkey, partly offset by North Africa and Ukraine; and Asia, notably the Philippines and Vietnam, partly offset by Japan and Korea. Cigarette shipment volume of Marlboro decreased in Latin America & Canada, mainly due to Argentina, Brazil and Mexico, partly offset by Colombia.
The increase in cigarette shipment volume of L&M was predominantly driven by growth in EEMA, notably Egypt, Turkey and Ukraine, partly offset by Russia. The decrease in cigarette shipment volume of Parliament was primarily due to Kazakhstan, Korea, Russia and Ukraine, partly offset by Japan and Turkey. Cigarette shipment volume of Bond Street was essentially flat, with growth, notably driven by Australia, Russia and Serbia, largely offset by declines in the European Union, Kazakhstan and Ukraine. The decrease in cigarette shipment volume of Chesterfield was due to EEMA, mainly Russia, Turkey and Ukraine, partly offset by the European Union, mainly the Czech Republic, Italy and Poland, and by Latin America & Canada, mainly Mexico. The increase in cigarette shipment volume of Philip Morris primarily reflects the morphing of Diana in Italy. The increase in cigarette shipment volume of Lark was principally driven by Japan, partly offset by Korea.
Total shipment volume of OTP, in cigarette equivalent units, increased by 1.0% to 34.1 billion units.
Total shipment volume for cigarettes and OTP, in cigarette equivalent units, decreased by 1.0%, excluding acquisitions.
Our net revenues and excise taxes on products were as follows:
For the Years Ended December 31, | |||||||||||||||
(in millions) | 2015 | 2014 | Variance | % | |||||||||||
Net revenues | $ | 73,908 | $ | 80,106 | $ | (6,198 | ) | (7.7 | )% | ||||||
Excise taxes on products | 47,114 | 50,339 | (3,225 | ) | (6.4 | )% | |||||||||
Net revenues, excluding excise taxes on products | $ | 26,794 | $ | 29,767 | $ | (2,973 | ) | (10.0 | )% | ||||||
36
Net revenues, which include excise taxes billed to customers, decreased by $6.2 billion. Excluding excise taxes, net revenues decreased by $3.0 billion due primarily to:
• | unfavorable currency ($4.7 billion) and |
• | unfavorable volume/mix ($325 million), partly offset by |
• | price increases ($2.1 billion). |
Currency movements decreased net revenues by $13.5 billion and net revenues, excluding excise taxes on products, by $4.7 billion. The $4.7 billion decrease was due primarily to the Argentine peso, Australian dollar, Canadian dollar, Euro, Indonesian rupiah, Japanese yen, Mexican peso, Russian ruble, Turkish lira and the Ukrainian hryvnia.
Net revenues include $1.8 billion in 2015 and $2.0 billion in 2014 related to sales of OTP. These net revenue amounts include excise taxes billed to customers. Excluding excises taxes, net revenues for OTP were $673 million in 2015 and $753 million in 2014.
Excise taxes on products decreased by $3.2 billion, due primarily to:
• | favorable currency ($8.8 billion), partly offset by |
• | higher excise taxes resulting from changes in retail prices and tax rates ($5.4 billion) and |
• | higher excise taxes resulting from volume/mix ($142 million). |
Our cost of sales; marketing, administration and research costs; and operating income were as follows:
For the Years Ended December 31, | |||||||||||||||
(in millions) | 2015 | 2014 | Variance | % | |||||||||||
Cost of sales | $ | 9,365 | $ | 10,436 | $ | (1,071 | ) | (10.3 | )% | ||||||
Marketing, administration and research costs | 6,656 | 7,001 | (345 | ) | (4.9 | )% | |||||||||
Operating income | 10,623 | 11,702 | (1,079 | ) | (9.2 | )% | |||||||||
Cost of sales decreased by $1.1 billion, due primarily to:
• | favorable currency ($1.4 billion), partly offset by |
• | higher manufacturing costs ($166 million) and |
• | higher cost of sales resulting from volume/mix ($148 million). |
Marketing, administration and research costs decreased by $345 million, due primarily to:
• | favorable currency ($979 million), partly offset by |
• | higher expenses ($628 million, primarily higher marketing and selling expenses). |
Operating income decreased by $1.1 billion, due primarily to:
• | unfavorable currency ($2.3 billion), |
• | higher marketing, administration and research costs ($628 million), |
• | unfavorable volume/mix ($473 million) and |
• | higher manufacturing costs ($166 million), partly offset by |
• | price increases ($2.1 billion) and |
• | lower pre-tax charges for asset impairment and exit costs ($467 million). |
37
Our effective tax rate decreased by 1.1 percentage points to 28.0%. The 2015 effective tax rate was unfavorably impacted by changes to repatriation assertions on certain foreign subsidiary historical earnings ($58 million), partially offset by a reduction in unrecognized tax benefits of $41 million following the conclusion of the IRS examinations of Altria's consolidated tax returns for the years 2007 and 2008 and PMI's consolidated tax returns for the years 2009 through 2011. The 2014 effective tax rate was unfavorably impacted by the asset impairment and exit costs related to the factory closures. The effective tax rate is based on our full-year earnings mix by taxing jurisdiction and cash repatriation plans.
Net earnings attributable to PMI of $6.9 billion decreased by $620 million (8.3%). This decrease was due primarily to lower operating income as discussed above, partially offset by a lower effective tax rate. Diluted and basic EPS of $4.42 decreased by 7.1%. Excluding an unfavorable currency impact of $1.20, diluted EPS increased by 18.1%.
Operating Results by Business Segment
Business Environment
Taxes, Legislation, Regulation and Other Matters Regarding the Manufacture, Marketing, Sale and Use of Tobacco Products
The tobacco industry and our business face a number of challenges that may adversely affect our business, volume, results of operations, cash flows and financial position. These challenges, which are discussed below and in “Cautionary Factors That May Affect Future Results,” include:
• | regulatory restrictions on our portfolio of products, including restrictions on the packaging, marketing, and sale of tobacco or other nicotine-containing products that could reduce our competitiveness, eliminate our ability to communicate with adult smokers, or even ban certain of our products; |
• | fiscal challenges, such as excise tax increases and discriminatory tax structures; |
• | illicit trade in cigarettes and other tobacco products, including counterfeit, contraband and so-called “illicit whites”; |
• | intense competition, including from non-tax paid volume by certain local manufacturers; |
• | pending and threatened litigation as discussed in Item 8, Note 19. Contingencies; and |
• | governmental investigations. |
Regulatory Restrictions: The tobacco industry operates in a highly regulated environment. The well-known risks of smoking have led regulators to impose significant restrictions and high excise taxes on cigarettes.
We support a comprehensive regulatory framework for tobacco products based on the principle of harm reduction, including mandated health warnings, minimum age laws, restrictions on advertising, and public place smoking restrictions. We also support regulatory measures that help reduce illicit trade.
Much of the regulation that shapes the business environment in which we operate is driven by the World Health Organization's (“WHO”) Framework Convention on Tobacco Control (“FCTC”), which entered into force in 2005. The FCTC is the first international public health treaty and has as its main objective to establish a global agenda for tobacco regulation, with the purpose of reducing tobacco use. To date, 179 countries and the European Union are Parties to the FCTC. The treaty requires Parties to have in place various tobacco control measures and recommends others. The FCTC governing body, the Conference of the Parties (“CoP”), has also adopted non-binding guidelines and policy recommendations related to certain articles of the FCTC that go beyond the text of the treaty.
We have opposed certain measures and continue to engage in a dialogue with regulators with respect to those measures that we do not believe would protect public health and, if implemented, could disrupt competition, severely limit our ability to market and sell our products to adult smokers, or increase illicit trade. Certain measures are discussed in more detail below. It is not possible to predict whether or to what extent measures recommended in the FCTC guidelines will be implemented.
38
Fiscal Challenges: Excessive and disruptive excise, sales and other tax increases and discriminatory tax structures are expected to continue to have an adverse impact on our profitability, due to lower consumption and consumer down-trading to non-premium, discount, other low-price or low-taxed tobacco products, such as fine cut tobacco and illicit products. In addition, in certain jurisdictions, our combustible products, mainly, cigarettes, are subject to tax structures that discriminate against premium-price products and manufactured cigarettes. We believe that such tax policies undermine public health by encouraging consumers to turn to the illicit trade for cheaper tobacco products and ultimately undercut government revenue objectives, disrupt the competitive environment, and encourage criminal activity. Other jurisdictions have imposed, or are seeking to impose, levies or other taxes specifically on tobacco companies, such as taxes on revenues and/or profits.
EU Tobacco Products Directive: In April 2014, the EU adopted the text of a significantly revised EU Tobacco Products Directive (TPD), which entered into force in May 2016. To date, 23 Member States and Norway have adopted laws transposing the TPD, while the remaining ones are concluding the national transposition. The TPD sets forth a comprehensive set of regulatory requirements for tobacco products, including:
• | health warnings covering 65% of the front and back panels of cigarette packs, with an option for Member States to further standardize tobacco packaging, including the introduction of plain packaging; |
• | a ban on characterizing flavors in some tobacco products, with a transition period for menthol expiring in May 2020; |
• | security features and tracking and tracing measures some of which could increase operational expenses depending on the scope of the implementing regulation; and |
• | a framework for the regulation of novel tobacco products and e-cigarettes, including requirements for health warnings and information leaflets, a prohibition on product packaging text related to reduced risk, and the introduction of notification requirements or authorization procedures in advance of commercialization. |
Plain Packaging and Other Packaging Restrictions: Plain packaging legislation bans the use of branding, logos and colors on packaging other than the brand name and variant that may be printed only in specified locations and in a uniform font. To date, Australia, France, Hungary, Ireland, New Zealand, Norway and the U.K., have adopted plain packaging legislation. In Australia, plain packaging has been implemented since December 2012. In France, plain packaging has been fully implemented since January 2017, and in the U.K., full compliance is required as of May 2017. In Hungary, full compliance is required immediately for new product launches and no later than May 2019 for other products. No implementation date has yet been set in New Zealand, Norway, or Ireland.
Several countries have initiated World Trade Organization ("WTO") dispute settlement proceedings against Australia related to Australia's plain packaging legislation. The matter is still pending before the WTO panel. In May 2015, three of our subsidiaries filed papers in the English High Court seeking judicial review of the U.K.’s plain packaging legislation. In May 2016, the Court issued a judgment rejecting our claims as well as those from other tobacco companies. We chose not to appeal the decision. In December 2016, the Conseil d’Etat in France upheld the decree that implements France’s plain packaging legislation.
Other countries are also considering adopting plain packaging legislation, including, but not limited to, Canada, Turkey and Singapore.
Some countries have adopted, or are considering adopting, packaging restrictions that could have an impact similar to plain packaging. Examples of such restrictions include standardizing the shape and size of packages, prohibiting certain colors or the use of certain descriptive phrases on packaging, and requiring very large graphic health warnings that leave little space for branding.
Restrictions and Bans on the Use of Ingredients: The WHO and others in the public health community have recommended restrictions or total bans on the use of some or all ingredients in tobacco products, including menthol. Broad restrictions and ingredient bans would require us to reformulate our American blend tobacco products and could reduce our ability to differentiate these products in the market in the long term. Menthol bans would eliminate the entire category of mentholated tobacco products. The European Union has banned flavored tobacco products, subject to an exemption until May 2020 for menthol. Other countries may follow the EU’s approach. For instance, Turkey has banned menthol as of May 2020. More sweeping ingredient bans have been adopted by Canada and Brazil. While the Canadian ingredient ban currently exempts menthol at the national level, the majority of Canadian provinces have adopted or are in the process of adopting menthol bans. The Canadian federal government also recently published draft regulations that would prohibit menthol in cigarettes. The Brazil ingredients ban, which would prohibit the use of virtually all ingredients with flavoring or aromatic properties, is not in force due to a legal challenge by a tobacco industry union, of which our Brazilian subsidiary is a member. Other lawsuits are also pending against the Brazil ingredients ban. It is not possible to predict the outcome of these legal proceedings.
39
Bans on Display of Tobacco Products at Retail: In a number of our markets, including, but not limited to, Australia, Canada, Norway, Russia, and Singapore, governments have banned the display of tobacco products at the point of sale. Other countries are also considering similar bans.
Bans and Restrictions on Advertising, Marketing, Promotions and Sponsorships: For many years, the FCTC has called for, and countries have imposed, partial or total bans on tobacco advertising, marketing, promotions and sponsorships, including bans and restrictions on advertising on radio and television, in print and on the Internet. The FCTC guidelines recommend that governments prohibit all forms of communication with adult smokers.
Restrictions on Product Design: Some members of the public health community are calling for the further standardization of tobacco products by requiring, for example, that cigarettes have a certain minimum diameter, which would amount to a ban on slim cigarettes, or requiring the use of standardized filter and cigarette paper designs. In addition, at its meeting in November 2016, the CoP adopted non-binding guidelines recommending that countries regulate product design features that increase the attractiveness of tobacco products, such as the diameter of cigarettes and the use of flavor capsules. Reduced cigarette ignition propensity (“RCIP”) standards previously recommended by the FCTC guidelines have been adopted in several of our markets (e.g., Australia, Canada, South Africa, South Korea, and the EU), and are being considered in others.
Restrictions on Public Smoking: The pace and scope of public smoking restrictions have increased significantly in most of our markets. Many countries around the world have adopted, or are likely to adopt, regulations that restrict or ban smoking in public and/or work places, restaurants, bars and nightclubs. Some public health groups have called for, and some countries, regional governments and municipalities have adopted or proposed, bans on smoking in outdoor places, as well as bans on smoking in cars (typically, when minors are present) and private homes.
Other Regulatory Issues: Some regulators are considering, or in some cases have adopted, regulatory measures designed to reduce the supply of tobacco products. These include regulations intended to reduce the number of retailers selling tobacco by, for example, reducing the overall number of tobacco retail licenses available or banning the sale of tobacco within arbitrary distances of certain public facilities.
In a limited number of markets, most notably Japan, we are dependent on governmental approvals that may limit our pricing flexibility.
Illicit Trade: The illicit tobacco trade creates a cheap and unregulated supply of tobacco products, undermines efforts to reduce smoking, especially among youth, damages legitimate businesses, stimulates organized crime, increases corruption and reduces government tax revenue. Illicit trade may account for as much as 10% of global cigarette consumption; this includes counterfeit, contraband and the growing problem of “illicit whites,” which are cigarettes legally produced in one jurisdiction for the sole purpose of being exported and illegally sold in another jurisdiction where they have no legitimate market. We estimate that illicit trade in the European Union accounted for slightly less than 10% of total cigarette consumption in 2016.
A number of jurisdictions are considering actions to prevent illicit trade. In November 2012, the FCTC adopted the Protocol to Eliminate Illicit Trade in Tobacco Products (the “Protocol”), which includes supply chain control measures, such as licensing of manufacturers and distributors, enforcement in free trade zones, controls on duty free and Internet sales and the implementation of tracking and tracing technologies. To date, 54 countries have signed the Protocol, and 24 countries and the European Union have ratified it. The Protocol will come into force once the fortieth country ratifies it, after which countries must implement its measures via national legislation. We expect, and welcome, that other countries will ratify the Protocol.
We and our subsidiaries have entered into cooperation agreements with governments and authorities to support their anti-illicit trade efforts. In 2004, we entered into a cooperation agreement with the EU and its member states that provided for cooperation with European law enforcement agencies on anti-contraband and on anti-counterfeit efforts. Under the terms of this agreement we made financial contributions of approximately $75 million per year to support these efforts. The EU Commission decided not to renew the agreement, which expired in July 2016.
In 2009, our Colombian subsidiaries entered into an Investment and Cooperation Agreement with the national and regional governments of Colombia to promote investment in, and cooperation on, anti-contraband and anti-counterfeit efforts. The agreement provides $200 million in funding over a 20-year period to address issues such as combating the illegal cigarette trade and increasing the quality and quantity of locally grown tobacco.
In May 2016, PMI launched PMI IMPACT, a new global initiative that supports third-party projects dedicated to fighting illegal trade and related crimes such as corruption, organized criminal networks and money laundering. The centerpiece of PMI IMPACT is a council of external independent experts with impeccable credentials in the fields of law, anti-corruption and law enforcement. The experts are
40
responsible for evaluating and approving funding proposals for PMI IMPACT grants. PMI has pledged $100 million to fund projects within PMI IMPACT during a three-year period. We expect that the first round of grants will be awarded in the second quarter of 2017.
In November 2016, PMI signed a joint Declaration of Intent to Prevent the Maritime Transportation of Counterfeit Goods together with eight other global brand owners and five of the world’s largest shipping companies. This commitment was a result of a dialogue with the International Chamber of Commerce’s Business Action to Stop Counterfeiting and Piracy. The signatories aim to tackle the infiltration of shipping services by criminal networks that exploit vessels to transport counterfeit goods, including “illicit whites,” across the oceans.
Reduced-Risk Products
Our Approach to RRPs: We recognize that smoking cigarettes causes serious diseases and that the best way to avoid the harms of smoking is never to start or to quit. Nevertheless, it is predicted that over the next decade the number of smokers will remain largely unchanged from the current estimate of 1.1 billion, despite the considerable efforts to discourage smoking.
Cigarettes burn tobacco, which produces smoke. As a result of the combustion process, the smoker inhales various toxic substances. In contrast, Reduced-Risk Products do not burn tobacco and produce significantly lower levels of harmful or potentially harmful compounds than found in smoke.
For smokers who would otherwise continue to smoke, we believe that RRPs offer a much better choice. Accordingly, our key strategic priorities are: to develop and commercialize products that present less risk of harm to adult smokers who switch to those products versus continued smoking; and to convince current adult smokers who would otherwise continue to smoke to switch to those Reduced-Risk Products.
We recognize that this transformation from cigarettes to RRPs will take time and that the speed of transformation will depend in part upon factors beyond our control, such as the willingness of governments, regulators and other policy groups to embrace RRPs as a desired solution to the smoking problem. We also recognize that the transformation must be funded from our existing cigarette business. For so long as a significant number continues to smoke, it is critical that the industry be led by responsible and ethical manufacturers. Therefore, during the transformation, we intend to remain the leading international cigarette manufacturer.
We have a range of RRPs in various stages of development, scientific assessment and commercialization. We conduct rigorous scientific assessment of our RRP platforms to establish that they reduce exposure to harmful and potentially harmful constituents in smoke and, ultimately, that these products present, are likely to present, or have the potential to present less risk of harm to adult smokers who switch to them versus continued smoking. We draw upon a team of expert scientists and engineers from a broad spectrum of scientific disciplines and our extensive learnings of consumer preferences to develop and assess our RRPs. Our efforts are guided by the following key objectives:
• | to develop RRPs that adult smokers who would otherwise continue to smoke find to be satisfying alternatives to smoking; |
• | for those adult smokers, our goal is to offer RRPs with a scientifically substantiated risk reduction profile that approaches as closely as possible that associated with smoking cessation; |
• | to substantiate the reduction of risk for the individual adult smoker and the reduction of harm to the population as a whole, based on scientific evidence of the highest standard that is made available for scrutiny and review by external independent scientists and relevant regulatory bodies; and |
• | to advocate for the development of science-based regulatory frameworks for the development and commercialization of RRPs, including the communication of scientifically substantiated information to enable adult consumers to make better health choices. |
Our RRP Platforms: Our product development is based on the elimination of combustion via tobacco heating and other innovative systems for aerosol generation, which we believe is the most promising path to providing a better choice for those who would otherwise continue to smoke. We recognize that no single product will appeal to all adult smokers. Therefore, we are developing a portfolio of products intended to appeal to a variety of distinct tastes.
Four RRP platforms are in various stages of development and commercialization readiness:
Platform 1 uses a precisely controlled heating device that we are commercializing under the IQOS brand name, into which a specially designed and proprietary consumable tobacco product (“IQOS Consumables”) is inserted and heated to generate an aerosol. Eight clinical studies have been completed (including two with the duration of three months). The study results show a substantial reduction in relevant biomarkers of exposure to harmful or potentially harmful constituents (“HPHCs”) in those adult smokers who switched to IQOS compared to those who continued to smoke cigarettes for the duration of the study. The reductions measured in those
41
who switched to IQOS approached those that were observed in study participants who quit smoking for the duration of the study. While these reduced exposure clinical studies were primarily designed to focus on biomarkers of exposure, in our three-month studies, we also measured six clinical risk markers. These clinical risk markers are associated with disease mechanisms known to be affected by smoking and to reverse upon cessation. The results are generally consistent with the expected direction of change and indicate that switching completely to IQOS led to an overall improvement of clinical risk markers affected by smoking after only three months. We also initiated a 6+6 month exposure response study in December 2014 and expect the results regarding the first six-month term in the third quarter of 2017. We have developed a new version of IQOS to further improve the consumer experience and plan to introduce this new version in the second quarter of 2017.
Platform 2 uses a pressed carbon heat source, which when ignited, generates an aerosol by heating tobacco. A pharmacokinetic study and a five-day reduced-exposure study with Platform 2 have now been completed. In the pharmacokinetic study, we observed that the nicotine pharmacokinetic profile and the subjective user satisfaction are comparable to smoking cigarettes, indicating that this platform could be an acceptable substitute for adult smokers who want to continue to use tobacco products but seek an alternative to cigarettes. The results of the reduced exposure study show a substantial reduction in relevant biomarkers of exposure to HPHCs in those who switched to Platform 2 compared to those who continued to smoke cigarettes over a five-day period. The clinical phase of a three-month reduced-exposure study has also been completed; we expect the results to be reported in 2017.
Platform 3 creates an aerosol of nicotine salt formed by the chemical reaction of nicotine with a weak organic acid. We are exploring two routes for this platform, one with electronics and one without. We have begun pre-clinical and clinical testing of this platform, including a pharmacokinetic study in New Zealand for the electronic version. The study assessed this product's nicotine pharmacokinetic profile in comparison to a nicotine inhalator. The study showed that nicotine absorption with this product reached peak levels six times faster than with the inhalator, in line with the pharmacokinetic profile of a cigarette, suggesting a significantly higher potential for acceptance by smokers. We have also completed the clinical phase of a pharmacodynamics study in the U.S. and expect final results for this study in the first half of 2017. We expect to commence a safety and efficacy study in the first half of 2017.
Platform 4 covers e-vapor products, which are battery-powered devices that produce an aerosol by vaporizing a liquid nicotine solution. Our e-vapor products comprise devices using current generation technology, and we are well advanced in the development and commercialization of our next generation of e-vapor technology, MESH, which addresses certain challenges presented by some e-vapor products currently on the market. Our MESH products are designed to ensure the consistency and quality of the generated aerosol. We have commenced non-clinical studies, and our planned clinical assessment includes a pharmacokinetic study scheduled to start in 2017 and a reduced-exposure study that we expect to start in 2018.
We are also developing other potential product platforms.
Commercialization of RRPs: In 2014, we introduced the IQOS system in pilot city launches in Nagoya, Japan, and in Milan, Italy. Since then, we have expanded our commercialization activities to include all of Japan, as well as multiple cities in Italy. We also launched IQOS in various cities in Switzerland, Russia, Romania, Ukraine, Portugal, Germany, New Zealand, Canada and other markets. By the end of December 2016, IQOS was available for sale nationwide in Japan and in key cities in 20 markets, and approximately 1.4 million adult smokers had converted to it. We expect to expand IQOS nationally in many of these markets in 2017 as IQOS Consumables capacity increases. We are also targeting launches of IQOS in key cities in an additional ten to fifteen markets by the end of 2017 as capacity permits.
On the basis of our experience in Japan and Italy, we estimate that only a very small percentage of adult smokers who fully convert to IQOS switch back to cigarettes.
In the first quarter of 2016, we started broad commercial production of IQOS Consumables. To date, we have experienced supply shortages resulting from stronger-than-anticipated demand, primarily in Japan, and expect capacity limitations to ease as 2017 unfolds. Our 2016 year-end installed capacity was approximately 15 billion IQOS Consumables, and we expect that our 2017 year-end installed IQOS Consumables capacity will reach approximately 50 billion units. This installed capacity is expected to allow us to produce approximately 32 billion units in 2017, up from 7 billion in 2016. We estimate that as of 2018, we can increase installed annual capacity, as needed, by approximately four billion units per month, with a lead time of 12 months.
As a result of the supply shortages of IQOS Consumables, in June 2016, we began to limit the sale of our IQOS devices in Japan. While we currently have sufficient capacity with respect to IQOS devices, we plan to expand our supplier base. Our IQOS devices are subject to product warranties for a period of 12 months from the date of purchase or such other periods as required by law. Warranty claims for these devices have not had a material impact on our consolidated financial position, results of operations or cash flows in any of the periods presented, although they may become more significant as the number of units sold increases.
42
We currently market our e-vapor products in several markets, including the United Kingdom, Spain, and Israel. In November 2016, we began the city test of MESH, our Platform 4 product, in Birmingham, United Kingdom. We also expect to conduct city tests of our Platform 2 and Platform 3 products in 2017.
RRP Regulation and Taxation: RRPs contain nicotine and are not risk-free. We therefore support science-based regulation and taxation of RRPs. Regulation and taxation should differentiate between cigarettes and products that present, are likely to present, or have the potential to present less risk of harm to adult smokers who switch to these products versus continued smoking. Regulation should provide minimum standards for RRPs and specific rules for, among other things, ingredients, labelling and consumer communication, and should ensure that the public is informed about the health risks of all tobacco and nicotine-containing products. Regulation, as well as tobacco industry activities, should reflect the fact that youth should not consume nicotine in any form.
Some governments have banned or are seeking to ban or severely restrict emerging tobacco and nicotine-containing products such as our RRPs. These regulations might foreclose or unreasonably restrict consumer access even to products that might be shown to be a better health choice than continuing to smoke. We oppose such blanket bans and unreasonable restrictions of products that have the potential to present less risk of harm compared to continued smoking. By contrast, we support regulation that sets clear standards and propels innovation to benefit adult smokers.
In the United States, an established regulatory framework for assessing “Modified Risk Tobacco Products” and “New Tobacco Products” exists under the jurisdiction of the Food and Drug Administration (“FDA”). Future FDA actions may influence the regulatory approach of other interested governments. In December 2016, we submitted a Modified Risk Tobacco Product Application for Platform 1 to the FDA. We plan to submit a Premarket Tobacco Application in the first quarter of 2017 for Platform 1.
In the EU, a majority of Member States have transposed the EU Tobacco Products Directive, including the provisions on Novel Tobacco Products, such as IQOS Consumables, and e-cigarettes. Most of the EU Member States require a notification submitted six months before the intended placing on the market of a Novel Tobacco Product, while some require pre-market authorizations for the introduction of such products. To date, pursuant to these requirements, we have filed a comprehensive dossier summarizing our scientific assessment of Platform 1 in 14 Member States.
There can be no assurance that we will succeed in our efforts to replace cigarettes with RRPs or that regulation will allow us to commercialize RRPs in all markets or treat RRPs differently from combustible tobacco products such as cigarettes.
Our RRP Business Development Initiatives: In December 2013, we established a strategic framework with Altria Group, Inc. (“Altria”) under which Altria will make available its e-vapor products exclusively to us for commercialization outside the United States, and we will make available two of our RRPs exclusively to Altria for commercialization in the United States. In March 2015, we launched Solaris, a Platform 4 e-vapor product licensed from Altria, in Spain. In December 2015, we introduced Solaris in Israel.
In July 2015, we extended the strategic framework with Altria to include a Joint Research, Development and Technology Sharing Agreement. The additional agreement provides the framework under which PMI and Altria will collaborate to develop the next generation of e-vapor products for commercialization in the United States by Altria and in markets outside the United States by PMI. The collaboration between PMI and Altria in this endeavor is enabled by exclusive technology cross licenses and technical information sharing. The agreements also provide for cooperation on the scientific assessment of, and for the sharing of improvements to, the existing generation of licensed products.
In June 2014, we acquired 100% of Nicocigs Limited, a leading U.K.-based e-vapor company whose principal brand is Nicolites. This acquisition provided PMI with immediate access to, and a significant presence in, the U.K. e-vapor market.
Governmental Investigations
From time to time, we are subject to governmental investigations on a range of matters. We describe certain matters pending in Thailand and South Korea in Item 3. Legal Proceedings-Other Litigation.
In November 2010, a WTO panel issued its decision in a dispute relating to facts that arose from August 2006 between the Philippines and Thailand concerning a series of Thai customs and tax measures affecting cigarettes imported by PM Thailand into Thailand from the Philippines (see Item 3. Legal Proceedings-Other Litigation for additional information). The WTO panel decision, which was upheld by the WTO Appellate Body, concluded that Thailand had no basis to find that PM Thailand's declared customs values and taxes paid were too low, as alleged by the DSI in 2009. The decision also created obligations for Thailand to revise its laws, regulations, or practices affecting the customs valuation and tax treatment of future cigarette imports. Thailand agreed in September 2011 to fully comply with the decision by October 2012. The Philippines contends that to date Thailand has not fully complied and commenced formal proceedings
43
at the WTO to address the outstanding issues. The Philippines has repeatedly expressed concerns with ongoing investigations by Thailand of PM Thailand, including those that led to the criminal charges described in Item 3. Legal Proceedings-Other Litigation, noting that these investigations appear to be based on grounds not supported by WTO customs valuation rules and inconsistent with several decisions already taken by Thai Customs and other Thai governmental agencies.
Acquisitions and Other Business Arrangements
We discuss our acquisitions and other business arrangements in Item 8, Note 6. Acquisitions and Other Business Arrangements to our consolidated financial statements.
Investments in Unconsolidated Subsidiaries
We discuss our investments in unconsolidated subsidiaries in Item 8, Note 4. Investments in Unconsolidated Subsidiaries to our consolidated financial statements.
Asset Impairment and Exit Costs
In November 2015, we commenced the implementation of a restructuring program within our European Union segment. The program is expected to be completed by the end of 2017. In total, we have incurred a total pre-tax charge of approximately $93 million for the program. During 2015, we recorded pre-tax exit costs of $68 million related to employee separation costs. In addition, as part of the total program, up to $25 million of pre-tax implementation costs, primarily related to costs for the project team and notice period payments, have been reflected in cost of sales and marketing, administration and research costs in our consolidated statement of earnings.
On April 4, 2014, we announced the initiation by our affiliate Philip Morris Holland B.V. ("PMH") of consultations with employee representatives on a proposal to discontinue cigarette production at its factory located in Bergen op Zoom, the Netherlands. PMH reached an agreement with the trade unions and their members on a social plan, and ceased cigarette production on September 1, 2014. In total, we have incurred a total pre-tax charge of approximately $549 million for the program. During 2014, we recorded pre-tax asset impairment and exit costs of $489 million. This amount included employee separation costs of $343 million, asset impairment costs of $139 million and other separation costs of $7 million. In addition, as part of the total program, approximately $60 million of pre-tax implementation costs, primarily related to notice period payments, have been reflected in cost of sales and marketing, administration and research costs in our consolidated statement of earnings, of which $50 million were recognized during 2014. Excluding asset impairment costs, substantially all of these charges have resulted in cash expenditures. The program was substantially completed as of December 31, 2015.
Trade Policy
We are subject to various trade restrictions imposed by the United States of America and countries in which we do business (“Trade Sanctions”), including the trade and economic sanctions administered by the U.S. Department of the Treasury's Office of Foreign Assets Control and the U.S. Department of State. It is our policy to comply fully with these Trade Sanctions.
Tobacco products are agricultural products under U.S. law and are not technological or strategic in nature. From time to time we make sales in countries subject to Trade Sanctions, either where such sanctions do not apply to our business or pursuant to exemptions or licenses.
To our knowledge, none of our commercial arrangements results in the governments of any country identified by the U.S. government as a state sponsor of terrorism, nor entities controlled by those governments, receiving cash or acting as intermediaries in violation of U.S. laws.
We do not sell products in Iran, Sudan, North Korea, Cuba and Syria. We may from time to time explore opportunities to sell our products in one or more of these countries, as permitted by law.
Certain states within the U.S. have enacted legislation permitting state pension funds to divest or abstain from future investment in stocks of companies that do business with certain countries that are sanctioned by the U.S. We do not believe such legislation has had a material effect on the price of our shares.
44
2016 compared with 2015
The following discussion compares operating results within each of our reportable segments for 2016 with 2015.
European Union:
European Union | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2016 | 2015 | Variance | % | |||||||||||
Net revenues | $ | 27,129 | $ | 26,563 | $ | 566 | 2.1 | % | |||||||
Excise taxes on products | 18,967 | 18,495 | 472 | 2.6 | % | ||||||||||
Net revenues, excluding excise taxes on products | 8,162 | 8,068 | 94 | 1.2 | % | ||||||||||
Operating companies income | 3,994 | 3,576 | 418 | 11.7 | % | ||||||||||
Net revenues, which include excise taxes billed to customers, increased by $566 million. Excluding excise taxes, net revenues increased by $94 million, due to:
• | price increases ($390 million), party offset by |
• | unfavorable volume/mix ($149 million) and |
• | unfavorable currency ($147 million). |
The net revenues of the European Union segment include $1.4 billion in 2016 and $1.5 billion in 2015 related to sales of OTP. Excluding excise taxes, OTP net revenues for the European Union segment were $485 million in 2016 and $509 million in 2015.
Operating companies income increased by $418 million during 2016. This increase was due primarily to:
• | price increases ($390 million), |
• | the non-recurrence of the 2015 pre-tax charges for asset impairment and exit costs ($68 million), |
• | lower manufacturing costs ($49 million), |
• | lower marketing, administration and research costs ($47 million) and |
• | favorable currency ($34 million), partly offset by |
• | unfavorable volume/mix ($168 million). |
European Union - Industry Volume
The estimated total cigarette market decreased by 1.6% to 501.0 billion units. The moderate decline of the estimated total cigarette market reflected improved macroeconomics, a lower prevalence of illicit trade and, in certain geographies, the estimated positive impact of immigration, which was concentrated in the first half of 2016.
The estimated total OTP market decreased by 2.3% to 152.5 billion cigarette equivalent units, reflecting a lower total fine cut market, down by 2.4% to 141.7 billion cigarette equivalent units.
45
European Union - PMI Shipment Volume and Market Share
Cigarette shipment volume and market share performance by brand are shown in the tables below:
European Union Cigarette Shipment Volume by Brand (Million Units) | ||||||||
Full-Year | ||||||||
2016 | 2015 | Change | ||||||
Marlboro | 96,245 | 95,588 | 0.7 | % | ||||
L&M | 34,691 | 35,010 | (0.9 | )% | ||||
Chesterfield | 30,140 | 28,278 | 6.6 | % | ||||
Philip Morris | 16,290 | 14,205 | 14.7 | % | ||||
Others | 16,220 | 21,508 | (24.6 | )% | ||||
Total EU | 193,586 | 194,589 | (0.5 | )% | ||||
European Union Cigarette Market Shares by Brand | ||||||||
Full-Year | ||||||||
Change | ||||||||
2016 | 2015 | p.p. | ||||||
Marlboro | 19.0 | % | 18.8 | % | 0.2 | |||
L&M | 6.9 | % | 6.9 | % | — | |||
Chesterfield | 5.9 | % | 5.6 | % | 0.3 | |||
Philip Morris | 3.2 | % | 3.2 | % | — | |||
Others | 3.3 | % | 3.8 | % | (0.5 | ) | ||
Total EU | 38.3 | % | 38.3 | % | — | |||
Our cigarette shipment volume decreased by 0.5% to 193.6 billion units, mainly due to Italy, Germany and Greece, partly offset by Poland and Spain. Cigarette shipment volume of Marlboro increased by 0.7%, mainly driven by Spain, partly offset by Greece. Our total cigarette market share was flat at 38.3%, with gains, notably in the Czech Republic, France, Poland and Spain, offset by declines, mainly in Greece and Italy. Cigarette shipment volume of "Others" decreased, mainly due the morphing of various trademarks in the Czech Republic and Italy into international brands.
Our shipments of OTP decreased by 5.3% to 22.2 billion cigarette equivalent units. Our total OTP market share decreased by 0.4 points to 14.6%.
46
European Union - Key Market Commentaries
In France, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
France Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 44.9 | 45.5 | (1.2 | )% | |||||
PMI Shipments (million units) | 19,243 | 18,943 | 1.6 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 26.4 | % | 25.9 | % | 0.5 | ||||
Philip Morris | 10.2 | % | 9.5 | % | 0.7 | ||||
Chesterfield | 3.1 | % | 3.3 | % | (0.2 | ) | |||
Others | 2.7 | % | 2.9 | % | (0.2 | ) | |||
Total | 42.4 | % | 41.6 | % | 0.8 | ||||
The estimated total cigarette market decreased moderately by 1.2%, partly reflecting a lower prevalence of illicit trade and e-vapor products. The increase in our cigarette shipment volume mainly reflected market share growth, driven by Marlboro, as well as the launch of certain Philip Morris variants in January 2016. The estimated total industry fine cut category of 14.7 billion cigarette equivalent units increased by 1.2%. Our market share of the category increased by 0.6 points to 25.6%.
In Germany, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Germany Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 78.1 | 80.0 | (2.4 | )% | |||||
PMI Shipments (million units) | 28,950 | 29,778 | (2.8 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 22.5 | % | 22.1 | % | 0.4 | ||||
L&M | 11.6 | % | 11.9 | % | (0.3 | ) | |||
Chesterfield | 1.6 | % | 1.7 | % | (0.1 | ) | |||
Others | 1.4 | % | 1.5 | % | (0.1 | ) | |||
Total | 37.1 | % | 37.2 | % | (0.1 | ) | |||
The estimated total cigarette market decreased by 2.4%, primarily reflecting the impact of price increases. The decrease in our cigarette shipment volume primarily reflected the lower total market. The estimated total industry fine cut category of 40.6 billion cigarette equivalent units decreased by 0.8%. Our market share of the category decreased by 1.4 points to 11.3%.
47
In Italy, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Italy Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 72.1 | 73.8 | (2.4 | )% | |||||
PMI Shipments (million units) | 38,624 | 39,717 | (2.8 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 24.2 | % | 24.6 | % | (0.4 | ) | |||
Chesterfield | 11.5 | % | 11.0 | % | 0.5 | ||||
Philip Morris | 8.5 | % | 9.2 | % | (0.7 | ) | |||
Others | 8.2 | % | 8.9 | % | (0.7 | ) | |||
Total | 52.4 | % | 53.7 | % | (1.3 | ) | |||
The estimated total cigarette market decreased by 2.4%, primarily reflecting the impact of price increases. The decline of our cigarette shipments, down by 4.8% excluding the net impact of distributor inventory movements, reflected the lower total market, and lower cigarette market share, notably due to Marlboro as a result of its price increase in the second quarter of 2016, and low-price Philip Morris, impacted by the growth of the super-low price segment, partly offset by super-low price Chesterfield. The estimated total industry fine cut category of 6.6 billion cigarette equivalent units increased by 3.5%. Our market share of the category decreased by 1.9 points to 39.2%.
In Poland, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Poland Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 41.3 | 41.1 | 0.5 | % | |||||
PMI Shipments (million units) | 17,485 | 16,763 | 4.3 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 11.6 | % | 11.4 | % | 0.2 | ||||
L&M | 18.5 | % | 18.1 | % | 0.4 | ||||
Chesterfield | 9.1 | % | 8.6 | % | 0.5 | ||||
Others | 3.1 | % | 2.7 | % | 0.4 | ||||
Total | 42.3 | % | 40.8 | % | 1.5 | ||||
The estimated total cigarette market increased by 0.5%, primarily reflecting a lower prevalence of non-duty paid products. The increase in our cigarette shipment volume was mainly driven by higher cigarette market share, principally L&M, reflecting the positive impact of brand support, Chesterfield, benefiting from its 100s and super-slims variants, and RGD in "Others," up by 0.4 points to 2.6%. The estimated total industry fine cut category of 4.1 billion cigarette equivalent units increased by 3.3%. Our market share of the category decreased by 4.7 points to 26.7%, mainly due to increased price competition at the bottom of the market.
48
In Spain, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Spain Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 46.7 | 46.7 | (0.1 | )% | |||||
PMI Shipments (million units) | 16,365 | 15,435 | 6.0 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 18.0 | % | 17.0 | % | 1.0 | ||||
Chesterfield | 8.6 | % | 9.1 | % | (0.5 | ) | |||
L&M | 5.4 | % | 5.8 | % | (0.4 | ) | |||
Others | 1.9 | % | 1.5 | % | 0.4 | ||||
Total | 33.9 | % | 33.4 | % | 0.5 | ||||
The estimated total cigarette market decreased by 0.1%, reflecting an improved economy and the favorable estimated impact of in-switching from other tobacco products. Excluding the net impact of distributor inventory movements, our cigarette shipment volume increased by 1.6%, driven by higher market share reflecting the strong performance of Marlboro, benefiting from its round price point in the vending channel and the new Architecture 2.0. The estimated total industry fine cut category of 9.3 billion cigarette equivalent units decreased by 2.7%. Our market share of the fine cut category decreased by 1.9 points to 11.6%.
Eastern Europe, Middle East & Africa:
Eastern Europe, Middle East & Africa | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2016 | 2015 | Variance | % | |||||||||||
Net revenues | $ | 18,286 | $ | 18,328 | $ | (42 | ) | (0.2 | )% | ||||||
Excise taxes on products | 11,286 | 10,964 | 322 | 2.9 | % | ||||||||||
Net revenues, excluding excise taxes on products | 7,000 | 7,364 | (364 | ) | (4.9 | )% | |||||||||
Operating companies income | 3,016 | 3,425 | (409 | ) | (11.9 | )% | |||||||||
Net revenues, which include excise taxes billed to customers, decreased by $42 million. Excluding excise taxes, net revenues decreased by $364 million, due to:
• | unfavorable currency ($600 million) and |
• | unfavorable volume/mix ($348 million), partly offset by |
• | price increases ($584 million). |
49
Operating companies income decreased by $409 million during 2016. This decrease was due primarily to:
• | unfavorable currency ($839 million) and |
• | unfavorable volume/mix ($333 million), partly offset by |
• | price increases ($584 million) and |
• | lower marketing, administration and research costs ($170 million). |
Eastern Europe, Middle East & Africa - PMI Cigarette Shipment Volume
Our cigarette shipment volume decreased by 2.9% to 271.4 billion units, mainly due to North Africa, primarily Algeria, and Russia, partially offset by Saudi Arabia and Ukraine. Cigarette shipment volume of Marlboro decreased by 8.5% to 73.8 billion units, principally due to Algeria and Egypt, partly offset by Saudi Arabia. Cigarette shipment volume of Parliament increased by 1.0% to 33.9 billion units, driven by Saudi Arabia, Turkey and Ukraine, partly offset by Russia. Cigarette shipment volume of L&M increased by 1.9% to 52.2 billion units, driven notably by Algeria, Kazakhstan and Ukraine, partly offset by Russia and Turkey.
Eastern Europe, Middle East & Africa - Key Market Commentaries
In North Africa (defined as Algeria, Egypt, Libya, Morocco and Tunisia), estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
North Africa Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 139.0 | 138.5 | 0.4 | % | |||||
PMI Shipments (million units) | 34,035 | 38,111 | (10.7 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 8.5 | % | 13.7 | % | (5.2 | ) | |||
L&M | 12.5 | % | 11.9 | % | 0.6 | ||||
Others | 2.7 | % | 2.3 | % | 0.4 | ||||
Total | 23.7 | % | 27.9 | % | (4.2 | ) | |||
The estimated total cigarette market increased by 0.4%, driven by Egypt, Morocco and Tunisia, partly offset by Algeria. The decrease in our cigarette shipment volume reflected lower market share, mainly due to Marlboro in Algeria, principally resulting from the impact of excise tax-driven price increases, as well as lower-than-anticipated acceptance of Architecture 2.0 for Marlboro Round Taste.
50
In Russia, estimated industry size, our cigarette shipment volume and market share performance, as measured by Nielsen, are shown in the table below.
Russia Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 280.7 | 294.1 | (4.6 | )% | |||||
PMI Shipments (million units) | 79,651 | 84,422 | (5.7 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 1.4 | % | 1.4 | % | — | ||||
Parliament | 3.8 | % | 3.9 | % | (0.1 | ) | |||
Bond Street | 8.4 | % | 8.4 | % | — | ||||
Others | 13.6 | % | 14.7 | % | (1.1 | ) | |||
Total | 27.2 | % | 28.4 | % | (1.2 | ) | |||
The estimated total cigarette market decreased by 4.6%, mainly due to the impact of excise tax-driven price increases. The decrease in our cigarette shipment volume, down by 8.3% excluding the impact of estimated distributor inventory movements, mainly reflected the lower total market, and lower cigarette market share primarily due to a decline in "Others" of mid-price L&M and Chesterfield and super-low Optima, resulting from the timing of retail price increases compared to competition.
In Turkey, estimated industry size, our cigarette shipment volume and market share performance, as measured by Nielsen, are shown in the table below.
Turkey Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 105.5 | 103.2 | 2.2 | % | |||||
PMI Shipments (million units) | 49,624 | 49,014 | 1.2 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 10.2 | % | 9.5 | % | 0.7 | ||||
Parliament | 11.7 | % | 11.6 | % | 0.1 | ||||
Lark | 7.4 | % | 7.6 | % | (0.2 | ) | |||
Others | 15.0 | % | 15.1 | % | (0.1 | ) | |||
Total | 44.3 | % | 43.8 | % | 0.5 | ||||
The estimated total cigarette market increased by 2.2%, primarily reflecting a lower prevalence of illicit trade. The increase in our cigarette shipment volume was mainly driven by the higher total market. Our higher market share, led by Marlboro, primarily reflecting the growth of its slimmer Touch variant, and Chesterfield, partly offset by L&M in "Others."
51
In Ukraine, estimated industry size, our cigarette shipment volume and market share performance, as measured by Nielsen, are shown in the table below.
Ukraine Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 72.7 | 70.6 | 2.9 | % | |||||
PMI Shipments (million units) | 22,014 | 19,195 | 14.7 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 3.2 | % | 3.8 | % | (0.6 | ) | |||
Parliament | 3.0 | % | 2.9 | % | 0.1 | ||||
Bond Street | 10.1 | % | 8.3 | % | 1.8 | ||||
Others | 13.6 | % | 15.0 | % | (1.4 | ) | |||
Total | 29.9 | % | 30.0 | % | (0.1 | ) | |||
The estimated total cigarette market increased by 2.9%, mainly driven by a lower prevalence of illicit trade. The increase in our cigarette shipment volume reflected the higher total cigarette market. The decrease in our market share was primarily due to Marlboro, reflecting the impact of widened price gaps, and mid-price Chesterfield and super-low President in "Others," mainly resulting from competitive price pressure in the low price segment, partly offset by Bond Street and L&M in "Others."
Asia:
Asia | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2016 | 2015 | Variance | % | |||||||||||
Net revenues | $ | 20,531 | $ | 19,469 | $ | 1,062 | 5.5 | % | |||||||
Excise taxes on products | 11,850 | 11,266 | 584 | 5.2 | % | ||||||||||
Net revenues, excluding excise taxes on products | 8,681 | 8,203 | 478 | 5.8 | % | ||||||||||
Operating companies income | 3,196 | 2,886 | 310 | 10.7 | % | ||||||||||
Net revenues, which include excise taxes billed to customers, increased by $1.1 billion. Excluding excise taxes, net revenues increased by $478 million, due primarily to:
• | price increases ($335 million) and |
• | favorable volume/mix ($151 million). |
Net revenues include $666 million in 2016 related to sale of RRPs, mainly driven by Japan. Excluding excise taxes, net revenues for RRPs were $666 million in 2016. In some jurisdictions, including Japan, we are not responsible for collecting excise taxes.
52
Operating companies income increased by $310 million during 2016. This increase was due primarily to:
• | price increases ($335 million), |
• | favorable currency ($52 million) and |
• | lower marketing, administration and research costs ($28 million), partly offset by |
• | unfavorable volume/mix ($106 million). |
Asia - PMI Cigarette Shipment Volume
Our cigarette shipment volume decreased by 7.6% to 260.0 billion units, mainly due to: Indonesia; Pakistan, reflecting a lower total estimated cigarette market resulting from excise tax-driven price increases and the growth of illicit trade; the Philippines; and Thailand, primarily reflecting the impact of excise tax-driven price increases in the first quarter of 2016, as well as lower market share; and in-switching from our cigarette brands to IQOS Consumables; partly offset by Korea, reflecting a normalization of the total estimated cigarette market following the disruptive excise tax increase in January 2015.
Cigarette shipment volume of Marlboro increased by 4.0% to 76.5 billion units, mainly driven by Korea and the Philippines, partly offset by Vietnam, as well as in-switching from that brand to IQOS Consumables. Cigarette shipment volume of Parliament increased by 7.5% to 10.1 billion units, driven by Korea. Cigarette shipment volume of Lark decreased by 3.8% to 17.6 billion units, principally due to Japan.
Asia - Key Market Commentaries
In Indonesia, estimated industry size, our cigarette shipment volume, market share and segmentation performance are shown in the tables below.
Indonesia Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 315.6 | 320.0 | (1.4 | )% | |||||
PMI Shipments (million units) | 105,524 | 109,840 | (3.9 | )% | |||||
PMI Cigarette Market Share | |||||||||
Sampoerna A | 14.0 | % | 14.6 | % | (0.6 | ) | |||
Dji Sam Soe | 6.5 | % | 6.9 | % | (0.4 | ) | |||
U Mild | 4.2 | % | 4.7 | % | (0.5 | ) | |||
Others | 8.7 | % | 8.1 | % | 0.6 | ||||
Total | 33.4 | % | 34.3 | % | (0.9 | ) | |||
53
Indonesia Segmentation Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | p.p. | |||||||
Segment % of Total Market | |||||||||
Hand-Rolled Kretek (SKT) | 18.2 | % | 19.1 | % | (0.9 | ) | |||
Machine-Made Kretek (SKM) | 75.8 | % | 74.7 | % | 1.1 | ||||
Whites (SPM) | 6.0 | % | 6.2 | % | (0.2 | ) | |||
Total | 100.0 | % | 100.0 | % | — | ||||
PMI % Share of Segment | |||||||||
Hand-Rolled Kretek (SKT) | 37.3 | % | 37.7 | % | (0.4 | ) | |||
Machine-Made Kretek (SKM) | 28.9 | % | 29.7 | % | (0.8 | ) | |||
Whites (SPM) | 79.5 | % | 80.3 | % | (0.8 | ) | |||
The estimated total cigarette market decreased by 1.4%, mainly reflecting a soft economic environment and the impact of excise tax-driven price increases. The decrease in our cigarette shipments was mainly due to lower market share, reflecting the soft performance of our SKM portfolio, due to competitors' discounted product offerings, and our SKT portfolio, broadly in line with industry trends, as well as a lower estimated total market.
In Japan, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Japan Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 173.8 | 182.3 | (4.6 | )% | |||||
PMI Shipments (million units) | 43,915 | 45,690 | (3.9 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 10.9 | % | 11.3 | % | (0.4 | ) | |||
Parliament | 2.4 | % | 2.3 | % | 0.1 | ||||
Lark | 9.9 | % | 9.9 | % | — | ||||
Others | 1.7 | % | 1.8 | % | (0.1 | ) | |||
Total | 24.9 | % | 25.3 | % | (0.4 | ) | |||
The estimated total cigarette market decreased by 4.6%, reflecting the continued underlying cigarette consumption decline, the growth of Reduced-Risk Products, and the impact of the April price increases of certain brands of our key competitor. Excluding the net impact of distributor inventory movements, our cigarette shipment volume decreased by 6.5%. The decline was mainly due to a lower total cigarette market, as well as lower cigarette market share, reflecting the impact of competitors' retail pricing, competitors' differentiated menthol taste product offerings and in-switching from our cigarette brands to IQOS Consumables.
The estimated national market share of IQOS Consumables was 2.9%, bringing our total combined national market share to 27.1%, up by 1.7 points. We calculate national market share for IQOS Consumables in Japan as the total sales volume for IQOS Consumables as a percentage of the total estimated sales volume for cigarettes and IQOS Consumables.
54
In Korea, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Korea Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 73.6 | 67.3 | 9.4 | % | |||||
PMI Shipments (million units) | 15,490 | 14,201 | 9.1 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 9.6 | % | 9.6 | % | — | ||||
Parliament | 8.5 | % | 8.4 | % | 0.1 | ||||
Virginia S. | 2.4 | % | 2.6 | % | (0.2 | ) | |||
Others | 0.5 | % | 0.6 | % | (0.1 | ) | |||
Total | 21.0 | % | 21.2 | % | (0.2 | ) | |||
Excluding a favorable comparison with the prior year driven by estimated trade inventory movements, the estimated total cigarette market increased by 4.3%, reflecting the normalization of the market following the disruptive excise tax increase of 120% in January 2015. The growth in our cigarette shipment volume primarily reflected the higher estimated total market.
In the Philippines, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Philippines Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 79.3 | 90.2 | (12.0 | )% | |||||
PMI Shipments (million units) | 56,611 | 66,236 | (14.5 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 28.4 | % | 20.0 | % | 8.4 | ||||
Fortune | 23.4 | % | 29.2 | % | (5.8 | ) | |||
Jackpot | 7.9 | % | 12.4 | % | (4.5 | ) | |||
Others | 11.6 | % | 11.8 | % | (0.2 | ) | |||
Total | 71.3 | % | 73.4 | % | (2.1 | ) | |||
The estimated total cigarette market decreased by 12.0%, mainly due to the impact of excise tax-driven price increases. The decline in our cigarette shipment volume reflected the lower total market, as well as the impact of these price increases on market share, particularly on our low and super-low price brands, Fortune and Jackpot, partly offset by an increase in market share of Marlboro, benefiting from its narrowed price gap with lower-priced brands as a result of the price increases.
55
Latin America & Canada:
Latin America & Canada | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2016 | 2015 | Variance | % | |||||||||||
Net revenues | $ | 9,007 | $ | 9,548 | $ | (541 | ) | (5.7 | )% | ||||||
Excise taxes on products | 6,165 | 6,389 | (224 | ) | (3.5 | )% | |||||||||
Net revenues, excluding excise taxes on products | 2,842 | 3,159 | (317 | ) | (10.0 | )% | |||||||||
Operating companies income | 938 | 1,085 | (147 | ) | (13.5 | )% | |||||||||
Net revenues, which include excise taxes billed to customers, decreased by $541 million. Excluding excise taxes, net revenues decreased by $317 million, due to:
• | unfavorable currency ($525 million) and |
• | unfavorable volume/mix ($104 million), partly offset by |
• | price increases ($312 million). |
Operating companies income decreased by $147 million during 2016. This decrease was due to:
• | unfavorable currency ($282 million), |
• | unfavorable volume/mix ($85 million), |
• | higher manufacturing costs ($57 million) and |
• | higher marketing, administration and research costs ($35 million), partly offset by |
• | price increases ($312 million). |
Latin America & Canada - PMI Cigarette Shipment Volume
Our cigarette shipment volume decreased by 4.3% to 87.9 billion units, mainly due to Argentina, partly offset by Mexico. While cigarette shipment volume of Marlboro decreased by 1.8% to 35.2 billion units, its market share increased by 0.6 points to an estimated 15.8%, primarily driven by Brazil, up by 0.6 points to 10.3%, Colombia, up by 0.3 points to 9.3%, and Mexico, up by 1.3 points to 48.9%, partly offset by Argentina, down by 1.9 points to 22.4%. Cigarette shipment volume of Philip Morris decreased by 15.3% to 16.5 billion units, mainly due to Argentina.
56
Latin America & Canada - Key Market Commentaries
In Argentina, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Argentina Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 36.1 | 40.8 | (11.6 | )% | |||||
PMI Shipments (million units) | 27,512 | 31,910 | (13.8 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 22.4 | % | 24.3 | % | (1.9 | ) | |||
Parliament | 1.9 | % | 2.1 | % | (0.2 | ) | |||
Philip Morris | 41.6 | % | 44.7 | % | (3.1 | ) | |||
Others | 10.4 | % | 7.1 | % | 3.3 | ||||
Total | 76.3 | % | 78.2 | % | (1.9 | ) | |||
The decline of the estimated total cigarette market of 11.6% mainly reflected a soft economic environment and the impact of the May 2016 excise tax increase that drove a more than 50% increase in average industry retail prices. The decrease in our cigarette shipment volume was principally due to the lower total market. Our lower cigarette market share primarily reflected growth in competitors' super-low priced products benefiting from down-trading, partly offset by low-price Chesterfield in "Others." The capsule segment was up by 1.0 point to 17.4% of the total market; our share of the segment increased by 0.4 points to 73.9%.
In Canada, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Canada Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 26.3 | 26.7 | (1.6 | )% | |||||
PMI Shipments (million units) | 10,049 | 9,926 | 1.2 | % | |||||
PMI Cigarette Market Share | |||||||||
Belmont | 3.7 | % | 3.3 | % | 0.4 | ||||
Canadian Classics | 10.2 | % | 10.3 | % | (0.1 | ) | |||
Next | 11.3 | % | 10.6 | % | 0.7 | ||||
Others | 13.2 | % | 13.1 | % | 0.1 | ||||
Total | 38.4 | % | 37.3 | % | 1.1 | ||||
The estimated total cigarette market decreased by 1.6%. The increase in our cigarette shipment volume was principally driven by higher cigarette market share, favorably impacted by estimated trade inventory movements, partly offset by a lower total market.
57
In Mexico, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Mexico Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2016 | 2015 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 36.2 | 33.9 | 6.7 | % | |||||
PMI Shipments (million units) | 25,080 | 23,246 | 7.9 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 48.9 | % | 47.6 | % | 1.3 | ||||
Delicados | 9.7 | % | 10.6 | % | (0.9 | ) | |||
Benson & Hedges | 4.7 | % | 4.5 | % | 0.2 | ||||
Others | 6.0 | % | 5.8 | % | 0.2 | ||||
Total | 69.3 | % | 68.5 | % | 0.8 | ||||
The estimated total cigarette market increased by 6.7%, or by 1.9% excluding the net impact of estimated trade inventory movements, primarily reflecting improved market conditions and a lower prevalence of illicit trade. The increase in our cigarette shipment volume reflected the higher total market. Our cigarette market share, benefiting from the impact of estimated inventory movements, was up by 0.8 points, with growth of Marlboro and Benson & Hedges, reflecting the impact of new product launches, partly offset by low-price Delicados. Our share of the premium segment, representing 56.8% of the total market, increased by 1.1 points to 93.5%.
2015 compared with 2014
The following discussion compares operating results within each of our reportable segments for 2015 with 2014.
European Union:
European Union | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2015 | 2014 | Variance | % | |||||||||||
Net revenues | $ | 26,563 | $ | 30,517 | $ | (3,954 | ) | (13.0 | )% | ||||||
Excise taxes on products | 18,495 | 21,370 | (2,875 | ) | (13.5 | )% | |||||||||
Net revenues, excluding excise taxes on products | 8,068 | 9,147 | (1,079 | ) | (11.8 | )% | |||||||||
Operating companies income | 3,576 | 3,815 | (239 | ) | (6.3 | )% | |||||||||
Net revenues, which include excise taxes billed to customers, decreased by $4.0 billion. Excluding excise taxes, net revenues decreased by $1.1 billion, due primarily to:
• | unfavorable currency ($1.5 billion) and |
• | unfavorable volume/mix ($29 million), partly offset by |
• | price increases ($442 million). |
The net revenues of the European Union segment include $1.5 billion in 2015 and $1.7 billion in 2014 related to sales of OTP. Excluding excise taxes, OTP net revenues for the European Union segment were $509 million in 2015 and $574 million in 2014.
58
Operating companies income decreased by $239 million during 2015. This decrease was due primarily to:
• | unfavorable currency ($857 million), |
• | higher marketing, administration and research costs ($242 million) and |
• | unfavorable volume/mix ($47 million), partly offset by |
• | price increases ($442 million), |
• | lower pre-tax charges for asset impairment and exit costs ($422 million, primarily due to the non-recurrence of the 2014 pre-tax charge related to the decision to discontinue cigarette production in the Netherlands) and |
• | lower manufacturing costs ($46 million). |
European Union - Industry Volume
The estimated total cigarette market in the European Union of 508.9 billion units decreased by 0.7%. The net impact of estimated trade inventory movements was neutral. The moderate decline of the estimated total cigarette market reflected, in certain key geographies, improving economies, a decrease in the prevalence of illicit trade, lower out-switching to the fine cut category and a lower prevalence of e-vapor products.
The estimated total OTP market in the European Union of 156.1 billion cigarette equivalent units increased by 0.3%. The total fine cut market was up by 0.8% to 145.1 billion cigarette equivalent units.
European Union - PMI Shipment Volume and Market Share
Cigarette shipment volume and market share performance by brand are shown in the tables below:
European Union Cigarette Shipment Volume by Brand (Million Units) | ||||||||
Full-Year | ||||||||
2015 | 2014 | Change | ||||||
Marlboro | 95,588 | 94,537 | 1.1 | % | ||||
L&M | 35,010 | 34,943 | 0.2 | % | ||||
Chesterfield | 28,278 | 27,100 | 4.3 | % | ||||
Philip Morris | 14,205 | 10,224 | 38.9 | % | ||||
Others | 21,508 | 27,942 | (23.0 | )% | ||||
Total EU | 194,589 | 194,746 | (0.1 | )% | ||||
European Union Cigarette Market Shares by Brand | ||||||||
Full-Year | ||||||||
Change | ||||||||
2015 | 2014 | p.p. | ||||||
Marlboro | 18.8 | % | 18.7 | % | 0.1 | |||
L&M | 6.9 | % | 6.8 | % | 0.1 | |||
Chesterfield | 5.6 | % | 5.3 | % | 0.3 | |||
Philip Morris | 3.2 | % | 3.2 | % | — | |||
Others | 3.8 | % | 4.2 | % | (0.4 | ) | ||
Total EU | 38.3 | % | 38.2 | % | 0.1 | |||
Our cigarette shipment volume of 194.6 billion units decreased by 0.1%, or by 0.4% excluding favorable net trade inventory movements, mainly in Italy. Market share increased by 0.1 point to 38.3%, with gains notably in France, Germany, Poland and Spain largely offset by the Czech Republic, Greece, Italy and Portugal.
59
Our shipments of OTP of 23.4 billion cigarette equivalent units increased by 2.2%. Our total OTP market share increased by 0.1 point to 15.0%.
European Union - Key Market Commentaries
In France, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
France Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 45.5 | 45.0 | 1.0 | % | |||||
PMI Shipments (million units) | 18,943 | 18,563 | 2.0 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 25.9 | % | 25.1 | % | 0.8 | ||||
Philip Morris | 9.5 | % | 9.4 | % | 0.1 | ||||
Chesterfield | 3.3 | % | 3.4 | % | (0.1 | ) | |||
Others | 2.9 | % | 3.1 | % | (0.2 | ) | |||
Total | 41.6 | % | 41.0 | % | 0.6 | ||||
The increase in the estimated total cigarette market reflected its general recovery since the second half of 2014 and a lower prevalence of e-vapor products and illicit trade. The increase in our cigarette shipment volume mainly reflected market share growth, notably of premium brands Marlboro, benefiting from a round retail price point of €7.00 per pack and the launch of Marlboro 25s in the first quarter of 2015, and Philip Morris. The estimated total industry fine cut category of 14.5 billion cigarette equivalent units increased by 6.9%. Our market share of the category decreased by 1.2 share points to 25.0%.
In Germany, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Germany Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 80.0 | 80.4 | (0.4 | )% | |||||
PMI Shipments (million units) | 29,778 | 29,411 | 1.2 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 22.1 | % | 21.7 | % | 0.4 | ||||
L&M | 11.9 | % | 11.8 | % | 0.1 | ||||
Chesterfield | 1.7 | % | 1.7 | % | — | ||||
Others | 1.5 | % | 1.4 | % | 0.1 | ||||
Total | 37.2 | % | 36.6 | % | 0.6 | ||||
The decline of the estimated total cigarette market was partly due to the impact of price increases, partially offset by a lower prevalence of illicit trade. The increase in our cigarette shipment volume principally reflected market share growth, driven by Marlboro, mainly reflecting the positive impact of the new Architecture 2.0, and L&M, benefiting from a rounded retail price point of €5.00 per pack of 19s. The estimated total industry fine cut category of 41.0 billion cigarette equivalent units decreased by 0.6%. Our market share of the category decreased by 0.2 points to 12.7%.
60
In Italy, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Italy Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 73.8 | 74.4 | (0.8 | )% | |||||
PMI Shipments (million units) | 39,717 | 40,439 | (1.8 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 24.6 | % | 25.7 | % | (1.1 | ) | |||
Chesterfield | 11.0 | % | 9.2 | % | 1.8 | ||||
Philip Morris | 9.2 | % | 10.4 | % | (1.2 | ) | |||
Others | 8.9 | % | 9.6 | % | (0.7 | ) | |||
Total | 53.7 | % | 54.9 | % | (1.2 | ) | |||
The moderate decrease in the estimated total cigarette industry was driven by an improved macro-economic environment and a lower prevalence of illicit trade and e-vapor products. Excluding the favorable net impact of estimated trade inventory movements, our cigarette shipment volume declined by 2.9%, mainly reflecting market share loss, notably of: Marlboro, largely due to its price increase in the first quarter of 2015 to €5.20 per pack from its round retail price point of €5.00 per pack; and Philip Morris, including the morphed Diana that had been impacted by the growth of the super-low price segment; partly offset by super-low price Chesterfield. The estimated total industry fine cut category of 6.4 billion cigarette equivalent units increased by 5.1%. Our market share of the category decreased by 0.4 points to 41.1%.
In Poland, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Poland Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 41.1 | 42.1 | (2.3 | )% | |||||
PMI Shipments (million units) | 16,763 | 16,630 | 0.8 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 11.4 | % | 11.2 | % | 0.2 | ||||
L&M | 18.1 | % | 18.2 | % | (0.1 | ) | |||
Chesterfield | 8.6 | % | 7.6 | % | 1.0 | ||||
Others | 2.7 | % | 3.1 | % | (0.4 | ) | |||
Total | 40.8 | % | 40.1 | % | 0.7 | ||||
The decrease in the estimated total cigarette market reflected the impact of price increases and an increase in the prevalence of illicit products, partly offset by a lower prevalence of e-vapor products. The increase in our cigarette shipment volume reflected higher market share, driven by Marlboro, partly reflecting the positive impact of the new Architecture 2.0, and Chesterfield, benefiting from its super-slims variants, partly offset by declines from super-low price brands. The estimated total industry fine cut category of 4.0 billion cigarette equivalent units increased by 11.0%, mainly reflecting the retail price impact of excise tax restructuring on the cigar and cigarillo categories that drove higher in-switching to the fine cut category, as well as a lower prevalence of illicit OTP. Our market share of the category decreased by 3.3 points to 31.4%, mainly due to increased price competition at the bottom end of the market.
61
In Spain, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Spain Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 46.7 | 47.0 | (0.6 | )% | |||||
PMI Shipments (million units) | 15,435 | 14,879 | 3.7 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 17.0 | % | 15.9 | % | 1.1 | ||||
Chesterfield | 9.1 | % | 9.2 | % | (0.1 | ) | |||
L&M | 5.8 | % | 6.1 | % | (0.3 | ) | |||
Others | 1.5 | % | 0.9 | % | 0.6 | ||||
Total | 33.4 | % | 32.1 | % | 1.3 | ||||
The decrease in the estimated total cigarette market mainly reflected the impact of price increases, partly offset by an improving economy, and a lower prevalence of illicit trade and e-vapor products. The increase in our cigarette shipment volume principally reflected higher market share, driven mainly by Marlboro, benefiting from a round price point in the vending channel, the new Architecture 2.0, and an improving economy. The estimated total industry fine cut category of 9.5 billion cigarette equivalent units decreased by 2.1%. Our market share of the fine cut category decreased by 1.3 points to 13.5%.
Eastern Europe, Middle East & Africa:
Eastern Europe, Middle East & Africa | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2015 | 2014 | Variance | % | |||||||||||
Net revenues | $ | 18,328 | $ | 20,469 | $ | (2,141 | ) | (10.5 | )% | ||||||
Excise taxes on products | 10,964 | 11,855 | (891 | ) | (7.5 | )% | |||||||||
Net revenues, excluding excise taxes on products | 7,364 | 8,614 | (1,250 | ) | (14.5 | )% | |||||||||
Operating companies income | 3,425 | 4,033 | (608 | ) | (15.1 | )% | |||||||||
Net revenues, which include excise taxes billed to customers, decreased by $2.1 billion. Excluding excise taxes, net revenues decreased by $1.3 billion, due primarily to:
• | unfavorable currency ($1.8 billion) and |
• | unfavorable volume/mix ($53 million), partly offset by |
• | price increases ($637 million). |
62
Operating companies income decreased by $608 million during 2015. This decrease was due primarily to:
• | unfavorable currency ($938 million), |
• | higher marketing, administration and research costs ($175 million), |
• | unfavorable volume/mix ($123 million) and |
• | higher manufacturing costs ($54 million), partially offset by |
• | price increases ($637 million) and |
• | higher equity income from unconsolidated subsidiaries ($44 million). |
Eastern Europe, Middle East & Africa - PMI Cigarette Shipment Volume
Our cigarette shipment volume of 279.4 billion units increased by 0.4%, driven notably by Egypt, Saudi Arabia and Turkey, partially offset by Kazakhstan and Ukraine. Excluding favorable net estimated trade inventory movements, our cigarette shipment volume was essentially flat. Our cigarette shipment volume of premium brands decreased by 0.6%, mainly due to Parliament, down by 3.2% to 33.6 billion units, mainly due to Kazakhstan, Russia and Ukraine, partly offset by Turkey, partly offset by growth from Marlboro, up by 0.7% to 80.7 billion units, driven by Saudi Arabia and Turkey, partly offset by North Africa and Ukraine. Our cigarette shipment volume of L&M increased by 8.4% to 51.2 billion units, driven notably by Egypt, Turkey and Ukraine, partly offset by Russia.
Eastern Europe, Middle East & Africa - Key Market Commentaries
In North Africa, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
North Africa Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 138.5 | 143.3 | (3.4 | )% | |||||
PMI Shipments (million units) | 38,111 | 37,782 | 0.9 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 13.7 | % | 15.3 | % | (1.6 | ) | |||
L&M | 11.9 | % | 8.9 | % | 3.0 | ||||
Others | 2.3 | % | 1.9 | % | 0.4 | ||||
Total | 27.9 | % | 26.1 | % | 1.8 | ||||
The decline of the estimated total market was principally due to Egypt, reflecting the impact of excise tax-driven price increases. The increase in our cigarette shipment volume was primarily driven by Egypt, reflecting higher market share, mainly of L&M, resulting from improved territorial coverage and brand building activities, partly offset by Algeria and Tunisia.
63
In Russia, estimated industry size, our cigarette shipment volume and market share performance, as measured by Nielsen, are shown in the table below.
Russia Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 294.1 | 314.1 | (6.4 | )% | |||||
PMI Shipments (million units) | 84,422 | 84,948 | (0.6 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 1.4 | % | 1.6 | % | (0.2 | ) | |||
Parliament | 3.9 | % | 3.7 | % | 0.2 | ||||
Bond Street | 8.4 | % | 7.7 | % | 0.7 | ||||
Others | 14.7 | % | 14.5 | % | 0.2 | ||||
Total | 28.4 | % | 27.5 | % | 0.9 | ||||
The decline of the estimated total cigarette market was mainly due to the unfavorable impact of excise tax-driven price increases and lower consumer purchasing power as a result of a weak economy. The decrease in our cigarette shipment volume mainly reflected the lower total market, largely offset by market share gains, primarily by premium Parliament, low-price Bond Street, notably its Compact 7.0 variant, and super-low price Next in "Others."
In Turkey, estimated industry size, our cigarette shipment volume and market share performance, as measured by Nielsen, are shown in the table below.
Turkey Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 103.2 | 94.7 | 9.0 | % | |||||
PMI Shipments (million units) | 49,014 | 46,309 | 5.8 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 9.5 | % | 8.6 | % | 0.9 | ||||
Parliament | 11.6 | % | 11.2 | % | 0.4 | ||||
Lark | 7.6 | % | 9.0 | % | (1.4 | ) | |||
Others | 15.1 | % | 15.2 | % | (0.1 | ) | |||
Total | 43.8 | % | 44.0 | % | (0.2 | ) | |||
The increase in the estimated total cigarette market mainly reflected a significantly lower prevalence of illicit trade. The increase in our cigarette shipment volume was driven by a higher total market. The decline in our market share was mainly due to low-price Lark, reflecting the impact of price repositioning by our principal competitor in May 2014, partly offset by Marlboro, notably its Touch 7.0 variants, and Parliament, benefiting from the growth of Parliament Night Blue KS, the leading SKU sold on the market, and from up-trading from the mid-price segment.
64
In Ukraine, estimated industry size, our cigarette shipment volume and market share performance, as measured by Nielsen, are shown in the table below.
Ukraine Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 70.6 | 69.7 | 1.3 | % | |||||
PMI Shipments (million units) | 19,195 | 23,273 | (17.5 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 3.8 | % | 4.9 | % | (1.1 | ) | |||
Parliament | 2.9 | % | 3.1 | % | (0.2 | ) | |||
Bond Street | 8.3 | % | 8.9 | % | (0.6 | ) | |||
Others | 15.0 | % | 15.8 | % | (0.8 | ) | |||
Total | 30.0 | % | 32.7 | % | (2.7 | ) | |||
The increase in the estimated total market was mainly driven by a lower prevalence of illicit trade. The decrease in our cigarette shipment volume largely reflected lower market share, primarily due to Marlboro, reflecting the impact of widened price gaps, and Bond Street, mainly resulting from competitive price pressure in the low-price segment.
Asia:
Asia | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2015 | 2014 | Variance | % | |||||||||||
Net revenues | $ | 19,469 | $ | 19,255 | $ | 214 | 1.1 | % | |||||||
Excise taxes on products | 11,266 | 10,527 | 739 | 7.0 | % | ||||||||||
Net revenues, excluding excise taxes on products | 8,203 | 8,728 | (525 | ) | (6.0 | )% | |||||||||
Operating companies income | 2,886 | 3,187 | (301 | ) | (9.4 | )% | |||||||||
Net revenues, which include excise taxes billed to customers, increased by $214 million. Excluding excise taxes, net revenues decreased by $525 million, due to:
• | unfavorable currency ($875 million) and |
• | unfavorable volume/mix ($100 million), partly offset by |
• | price increases ($450 million). |
65
Operating companies income decreased by $301 million during 2015. This decrease was due primarily to:
• | unfavorable currency ($388 million), |
• | higher marketing, administration and research costs ($165 million), |
• | unfavorable volume/mix ($162 million) and |
• | higher manufacturing costs ($70 million), partly offset by |
• | price increases ($450 million) and |
• | the non-recurrence of the 2014 pre-tax charges for asset impairment and exit costs ($35 million) due to the factory closure in Australia. |
Asia - PMI Cigarette Shipment Volume
Our cigarette shipment volume of 281.4 billion units decreased by 2.4%, mainly due to: Korea; Pakistan, reflecting a lower total estimated market resulting from the June and December 2015 excise tax-driven price increases, coupled with an increase in the prevalence of illicit trade and lower market share; and the Philippines. Excluding distributor inventory movements in Japan, reflecting a favorable comparison in 2015 resulting from the correction in 2014 of distributor inventory movements related to the VAT increase of April 2014, our cigarette shipment volume decreased by 3.1%.
Our cigarette shipment volume of Marlboro of 73.5 billion units increased by 3.0%, mainly driven by the Philippines and Vietnam, partly offset by Japan and Korea. Cigarette shipment volume of Parliament of 9.4 billion units decreased by 11.5%, primarily due to Korea, partly offset by Japan. Cigarette shipment volume of Lark of 18.3 billion units increased by 3.3%, principally driven by Japan, partly offset by Korea.
Asia - Key Market Commentaries
In Indonesia, estimated industry size, our cigarette shipment volume, market share and segmentation performance are shown in the tables below.
Indonesia Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 320.0 | 319.0 | 0.3 | % | |||||
PMI Shipments (million units) | 109,840 | 109,694 | 0.1 | % | |||||
PMI Cigarette Market Share | |||||||||
Sampoerna A | 14.6 | % | 14.2 | % | 0.4 | ||||
Dji Sam Soe | 6.9 | % | 6.2 | % | 0.7 | ||||
U Mild | 4.7 | % | 5.3 | % | (0.6 | ) | |||
Others | 8.1 | % | 8.7 | % | (0.6 | ) | |||
Total | 34.3 | % | 34.4 | % | (0.1 | ) | |||
66
Indonesia Segmentation Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | p.p. | |||||||
Segment % of Total Market | |||||||||
Hand-Rolled Kretek (SKT) | 19.1 | % | 20.6 | % | (1.5 | ) | |||
Machine-Made Kretek (SKM) | 74.7 | % | 73.0 | % | 1.7 | ||||
Whites (SPM) | 6.2 | % | 6.4 | % | (0.2 | ) | |||
Total | 100.0 | % | 100.0 | % | — | ||||
PMI % Share of Segment | |||||||||
Hand-Rolled Kretek (SKT) | 37.7 | % | 37.5 | % | 0.2 | ||||
Machine-Made Kretek (SKM) | 29.7 | % | 29.6 | % | 0.1 | ||||
Whites (SPM) | 80.3 | % | 79.1 | % | 1.2 | ||||
The estimated total cigarette market increased slightly, reflecting a soft economic environment. The slight change in our market share reflected a strong performance from our machine-made kretek brands, notably Sampoerna A, Dji Sam Soe Magnum and Dji Sam Soe Magnum Blue, offset by U Mild, and a decline in our hand-rolled kretek portfolio, notably due to Sampoerna Hijau in "Others," down by 0.4 share points to 2.9%.
In Japan, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Japan Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 182.3 | 186.2 | (2.1 | )% | |||||
PMI Shipments (million units) | 45,690 | 45,556 | 0.3 | % | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 11.3 | % | 11.6 | % | (0.3 | ) | |||
Parliament | 2.3 | % | 2.2 | % | 0.1 | ||||
Lark | 9.9 | % | 10.0 | % | (0.1 | ) | |||
Others | 1.8 | % | 2.1 | % | (0.3 | ) | |||
Total | 25.3 | % | 25.9 | % | (0.6 | ) | |||
The decrease of the estimated total cigarette market moderated to 2.1%. Excluding estimated inventory movements, driven principally by a favorable comparison as a result of the 2014 correction of distributor inventory movements partly related to the VAT increase of April 2014, our cigarette shipment volume decreased by 4.3%. The decline was mainly due to a lower total market, and lower market share principally reflecting the impact of competitive retail price and new menthol taste product offerings.
67
In Korea, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Korea Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 67.3 | 88.1 | (23.6 | )% | |||||
PMI Shipments (million units) | 14,201 | 17,346 | (18.1 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 9.6 | % | 7.8 | % | 1.8 | ||||
Parliament | 8.4 | % | 8.6 | % | (0.2 | ) | |||
Virginia S. | 2.6 | % | 2.6 | % | — | ||||
Others | 0.6 | % | 0.7 | % | (0.1 | ) | |||
Total | 21.2 | % | 19.7 | % | 1.5 | ||||
The decline of the estimated total cigarette market reflected the impact of the January 2015 excise tax increase and related retail price increases. Excluding the impact of estimated inventory movements associated with the timing of the excise tax increase, the total cigarette market declined by approximately 17.3%. The decline in our cigarette shipment volume reflected the lower estimated total market, partly offset by share growth, driven by Marlboro, benefiting from the positive impact of pricing for our principal domestic competitor's main brands.
In the Philippines, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Philippines Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 90.2 | 94.9 | (4.9 | )% | |||||
PMI Shipments (million units) | 66,236 | 68,358 | (3.1 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 20.0 | % | 16.0 | % | 4.0 | ||||
Fortune | 29.2 | % | 28.9 | % | 0.3 | ||||
Jackpot | 12.4 | % | 15.0 | % | (2.6 | ) | |||
Others | 11.8 | % | 12.1 | % | (0.3 | ) | |||
Total | 73.4 | % | 72.0 | % | 1.4 | ||||
The estimated total cigarette market decreased by 4.9%, mainly due to the impact of price increases. The decline in our cigarette shipment volume reflected the lower total market combined with lower consumption of our low and super-low price brands, following price increases in late 2014 and early 2015, partly offset by higher market share, driven by adult smoker uptrading to Marlboro, combined with market share growth of Fortune, reflecting the narrowing of retail price gaps with brands at the bottom end of the market.
68
Latin America & Canada:
Latin America & Canada | For the Years Ended December 31, | ||||||||||||||
(in millions) | 2015 | 2014 | Variance | % | |||||||||||
Net revenues | $ | 9,548 | $ | 9,865 | $ | (317 | ) | (3.2 | )% | ||||||
Excise taxes on products | 6,389 | 6,587 | (198 | ) | (3.0 | )% | |||||||||
Net revenues, excluding excise taxes on products | 3,159 | 3,278 | (119 | ) | (3.6 | )% | |||||||||
Operating companies income | 1,085 | 1,030 | 55 | 5.3 | % | ||||||||||
Net revenues, which include excise taxes billed to customers, decreased by $317 million. Excluding excise taxes, net revenues decreased by $119 million, due primarily to:
• | unfavorable currency ($505 million) and |
• | unfavorable volume/mix ($143 million), partly offset by |
• | price increases ($525 million). |
Operating companies income increased by $55 million during 2015. This increase was due primarily to:
• | price increases ($525 million), partly offset by |
• | unfavorable currency ($210 million), |
• | unfavorable volume/mix ($141 million), |
• | higher manufacturing costs ($88 million) and |
• | higher marketing, administration and research costs ($42 million). |
Latin America & Canada - PMI Cigarette Shipment Volume and Market Share
Our cigarette shipment volume of 91.9 billion units decreased by 2.9%, mainly due to Argentina, Brazil, Canada and Mexico. Although shipment volume of Marlboro of 35.8 billion units decreased by 3.2%, our Regional market share increased by 0.2 points to an estimated 15.2%. Market share of Marlboro increased notably in Brazil and Colombia, by 0.4 and 1.1 points to 9.7% and 9.0%, respectively. Shipment volume of Philip Morris of 19.4 billion units increased by 1.7%, driven mainly by Canada.
69
Latin America & Canada - Key Market Commentaries
In Argentina, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Argentina Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 40.8 | 41.9 | (2.5 | )% | |||||
PMI Shipments (million units) | 31,910 | 32,323 | (1.3 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 24.3 | % | 24.3 | % | — | ||||
Parliament | 2.1 | % | 2.2 | % | (0.1 | ) | |||
Philip Morris | 44.7 | % | 43.4 | % | 1.3 | ||||
Others | 7.1 | % | 7.3 | % | (0.2 | ) | |||
Total | 78.2 | % | 77.2 | % | 1.0 | ||||
The decline of the estimated total cigarette market was mainly due to the impact of price increases and a challenging economic environment. The decrease in our shipment volume was mainly due to a lower estimated total market, partly offset by market share growth, driven primarily by Philip Morris, reflecting the positive impact of the brand's capsule variants. Our share of the growing capsule segment, representing 16.4% of the total market, grew by 4.4 points to 73.5%.
In Canada, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Canada Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 26.7 | 27.3 | (2.3 | )% | |||||
PMI Shipments (million units) | 9,926 | 10,275 | (3.4 | )% | |||||
PMI Cigarette Market Share | |||||||||
Belmont | 3.3 | % | 3.0 | % | 0.3 | ||||
Canadian Classics | 10.3 | % | 10.4 | % | (0.1 | ) | |||
Next | 10.6 | % | 10.6 | % | — | ||||
Others | 13.1 | % | 13.6 | % | (0.5 | ) | |||
Total | 37.3 | % | 37.6 | % | (0.3 | ) | |||
The estimated total cigarette market decreased by 2.3%. Excluding the favorable impact of estimated competitors' trade inventory movements, the total market declined by 4.6%, mainly due to the impact of tax-driven price increases. The decrease in our cigarette shipment volume was principally due to a lower estimated total market. Our market share was also negatively impacted by the above-mentioned estimated competitors' trade inventory movements.
70
In Mexico, estimated industry size, our cigarette shipment volume and market share performance are shown in the table below.
Mexico Key Market Data | |||||||||
Full-Year | |||||||||
Change | |||||||||
2015 | 2014 | % / p.p. | |||||||
Total Cigarette Market (billion units) | 33.9 | 33.5 | 1.4 | % | |||||
PMI Shipments (million units) | 23,246 | 23,861 | (2.6 | )% | |||||
PMI Cigarette Market Share | |||||||||
Marlboro | 47.6 | % | 49.7 | % | (2.1 | ) | |||
Delicados | 10.6 | % | 11.1 | % | (0.5 | ) | |||
Benson & Hedges | 4.5 | % | 5.2 | % | (0.7 | ) | |||
Others | 5.8 | % | 5.3 | % | 0.5 | ||||
Total | 68.5 | % | 71.3 | % | (2.8 | ) | |||
The estimated total cigarette market increased by 1.4%. Excluding the unfavorable impact of estimated trade inventory movements, the total market increased by 3.8%, primarily reflecting a lower prevalence of illicit trade. The decrease in our cigarette shipment volume was mainly driven by: lower market share, mainly due to Marlboro, reflecting adult smoker down-trading; and the timing of price increases by our principal competitor in the first quarter of 2015; partly offset by gains for certain low-price local trademark brands.
Financial Review
Ÿ Net Cash Provided by Operating Activities
Net cash provided by operating activities of $8.1 billion for the year ended December 31, 2016, increased by $212 million from the comparable 2015 period. Excluding unfavorable currency movements of $409 million, the change was due primarily to net earnings growth and lower cash payments related to exit costs, partly offset by higher working capital requirements and 2016 installment payments of security into a court trust pertaining to the Létourneau and Blais cases as well as a 2016 payment to the South Korean tax authorities (see Item 8, Note 19. Contingencies for additional information).
Excluding currency, the unfavorable variance in working capital was due primarily to the following:
• | more cash used for accounts receivable, primarily due to the timing of sales and cash collections (including unfavorable comparisons to the cash flows provided for accounts receivable in 2015 following the expansion of arrangements to sell accounts receivable to unaffiliated financial institutions as disclosed in Item 8, Note 21. Sale of Accounts Receivable), partly offset by |
• | more cash provided by accrued liabilities and other current assets, primarily due to the timing of payments for excise taxes. |
Net cash provided by operating activities of $7.9 billion for the year ended December 31, 2015, increased by $126 million from the comparable 2014 period. The change was due primarily to net earnings growth (excluding unfavorable currency of $1.9 billion) and working capital initiatives.
Excluding currency, the favorable variance in working capital was due primarily to the following:
• | more cash provided by accounts receivable, primarily due to the timing of sales and cash collections (including the sale of accounts receivable in 2015 to unaffiliated financial institutions as disclosed in Item 8, Note 21. Sale of Accounts Receivable); and |
• | less cash used for accrued liabilities and other current assets, primarily due to the timing of payments for excise taxes; partially offset by |
• | more cash used for inventories, primarily related to higher finished goods inventories. |
71
Ÿ Net Cash Used in Investing Activities
Net cash used in investing activities of $968 million for the year ended December 31, 2016, increased by $260 million from the comparable 2015 period, due primarily to higher capital expenditures.
Net cash used in investing activities of $708 million for the year ended December 31, 2015, decreased by $288 million from the comparable 2014 period, due primarily to lower capital expenditures and purchases of businesses in 2014.
In June 2014, we acquired 100% of Nicocigs Limited, a leading U.K.-based e-vapor company, for the final purchase price of $103 million, net of cash acquired. For further details, see Item 8, Note 6. Acquisitions and Other Business Arrangements to our consolidated financial statements.
Our capital expenditures were $1.2 billion in 2016, $1.0 billion in 2015 and $1.2 billion in 2014. The 2016 expenditures were primarily related to investments in RRPs, productivity-enhancing programs, and equipment for new products. We expect total capital expenditures in 2017 of approximately $1.5 billion (including additional capital expenditures related to our ongoing investment in RRPs to support capacity expansion, notably for IQOS Consumables), to be funded by operating cash flows.
Ÿ Net Cash Used in Financing Activities
During 2016, net cash used in financing activities was $5.4 billion, compared with net cash used in financing activities of $4.7 billion during 2015 and $6.8 billion in 2014.
The 2016 change was due primarily to lower net proceeds received from the sale of subsidiary shares to noncontrolling interests, partially offset by higher proceeds from long-term debt issuances.
The 2015 change was due primarily to the cash used in 2014 to repurchase our common stock pursuant to our share repurchase program, as well as the 2015 net proceeds received from the sale of subsidiary shares to noncontrolling interests, partially offset by lower net cash proceeds in 2015 from long-term debt.
On January 30, 2014, the Indonesian Stock Exchange (“IDX”) adopted a regulation requiring all listed public companies to have at least a 7.5% public shareholding by January 30, 2016. In order to comply with this requirement, our subsidiary PT HM Sampoerna Tbk. (“Sampoerna”), of which we held a 98.18% interest, conducted a rights issue. In connection with the rights issue, PT Philip Morris Indonesia (“PMID”), a fully consolidated subsidiary of PMI, sold 264,209,711 of the rights to third-party investors. Delivery of the rights sold took place on October 26, 2015. The total net proceeds from the rights issue were $1.5 billion at prevailing exchange rates on the closing date. For further details, see Item 8, Note 6. Acquisitions and Other Business Arrangements to our consolidated financial statements.
During 2014, we used a total of $13.2 billion to repurchase our common stock, pay dividends and repay debt. These uses were partially offset by proceeds from our debt offerings and short-term borrowings in 2014 of $6.6 billion.
Dividends paid in 2016, 2015 and 2014 were $6.4 billion, $6.3 billion and $6.0 billion, respectively.
Ÿ Debt and Liquidity
We define cash and cash equivalents as short-term, highly liquid investments, readily convertible to known amounts of cash that mature within a maximum of three months and have an insignificant risk of change in value due to interest rate or credit risk changes. As a policy, we do not hold any investments in structured or equity-linked products. Our cash and cash equivalents are predominantly held in demand deposits with institutions that have investment-grade long-term credit rating.
72
Credit Ratings – The cost and terms of our financing arrangements as well as our access to commercial paper markets may be affected by applicable credit ratings. On March 21, 2016, Standard & Poor's affirmed our long-term credit rating at "A" and short-term at "A-1," but revised our outlook to "Negative" from "Stable." On August 1, 2016, Fitch affirmed our long-term credit rating at "A" and short-term at "F1," as well as our "Negative" outlook. We do not expect the Standard & Poor's and Fitch negative outlooks to impact our borrowing costs. On August 10, 2016, Moody's affirmed our long-term credit rating at "A2" and short-term at "P-1," as well as our "Stable" outlook. At February 13, 2017, our credit ratings and outlook by major credit rating agencies were as follows:
Short-term | Long-term | Outlook | |
Moody’s | P-1 | A2 | Stable |
Standard & Poor’s | A-1 | A | Negative |
Fitch | F1 | A | Negative |
Credit Facilities – On January 27, 2016, we entered into an agreement to extend the term of our existing $2.5 billion multi-year revolving credit facility from February 28, 2020, to February 28, 2021. On January 27, 2017, we entered into an agreement to extend the term of our $2.0 billion 364-day revolving credit facility from February 7, 2017, to February 6, 2018.
At February 13, 2017, our committed credit facilities were as follows:
(in billions) | ||||
Type | Committed Credit Facilities | |||
364-day revolving credit, expiring February 6, 2018 | $ | 2.0 | ||
Multi-year revolving credit, expiring February 28, 2021 | 2.5 | |||
Multi-year revolving credit, expiring October 1, 2020 (1) | 3.5 | |||
Total facilities | $ | 8.0 | ||
(1) On August 30, 2016, we entered into an agreement, effective October 1, 2016, to extend the term of our multi-year revolving credit facility, for an additional year covering the period October 1, 2020 to October 1, 2021 in the amount of $3.35 billion.
At February 13, 2017, there were no borrowings under the committed credit facilities, and the entire committed amounts were available for borrowing.
All banks participating in our committed credit facilities have an investment-grade long-term credit rating from the credit rating agencies. We continuously monitor the credit quality of our banking group, and at this time we are not aware of any potential non-performing credit provider.
Each of these facilities requires us to maintain a ratio of consolidated earnings before interest, taxes, depreciation and amortization (“consolidated EBITDA”) to consolidated interest expense of not less than 3.5 to 1.0 on a rolling four-quarter basis. At December 31, 2016, our ratio calculated in accordance with the agreements was 10.6 to 1.0. These facilities do not include any credit rating triggers, material adverse change clauses or any provisions that could require us to post collateral. We expect to continue to meet our covenants. The terms “consolidated EBITDA” and “consolidated interest expense,” both of which include certain adjustments, are defined in the facility agreements previously filed with the U.S. Securities and Exchange Commission.
In addition to the committed credit facilities discussed above, certain of our subsidiaries maintain short-term credit arrangements to meet their respective working capital needs. These credit arrangements, which amounted to approximately $2.9 billion at December 31, 2016, and December 31, 2015, are for the sole use of our subsidiaries. Borrowings under these arrangements amounted to $643 million at December 31, 2016, and $825 million at December 31, 2015.
Commercial Paper Program – We have commercial paper programs in place in the U.S. and in Europe. Our commercial paper programs in place in the U.S. and in Europe currently have an aggregate issuance capacity of $8.0 billion. At December 31, 2016 and December 31, 2015, we had no commercial paper outstanding.
73
We expect that the existence of the commercial paper program and the committed credit facilities, coupled with our operating cash flows, will enable us to meet our liquidity requirements.
Sale of Accounts Receivable – To mitigate credit risk and enhance cash and liquidity management we sell trade receivables to unaffiliated financial institutions. These arrangements allow us to sell, on an ongoing basis, certain trade receivables without recourse. The trade receivables sold are generally short-term in nature and are removed from the consolidated balance sheets. We sell trade receivables under two types of arrangements, servicing and non-servicing.
PMI’s operating cash flows were positively impacted by the amount of the trade receivables sold and derecognized from the consolidated balance sheets, which remained outstanding with the unaffiliated financial institutions. The trade receivables sold that remained outstanding under these arrangements as of December 31, 2016, 2015 and 2014, were $729 million, $888 million and $120 million, respectively. The net proceeds received are included in cash provided by operating activities in the consolidated statements of cash flows.
For further details, see Item 8, Note 21. Sale of Accounts Receivable to our consolidated financial statements.
Debt – Our total debt was $29.1 billion at December 31, 2016, and $28.5 billion at December 31, 2015. Our total debt is primarily fixed rate in nature. For further details, see Item 8, Note 7. Indebtedness. The weighted-average all-in financing cost of our total debt was 2.8% in 2016, compared to 3.0% in 2015. See Item 8, Note 16. Fair Value Measurements to our consolidated financial statements for a discussion of our disclosures related to the fair value of debt. The amount of debt that we can issue is subject to approval by our Board of Directors.
On February 21, 2014, we filed a shelf registration statement with the U.S. Securities and Exchange Commission, under which we may from time to time sell debt securities and/or warrants to purchase debt securities over a three-year period. During February 2017, we plan to file a new shelf registration statement with the U.S. Securities and Exchange Commission.
Our debt issuances in 2016 were as follows:
(in millions) | |||||||||
Type | Face Value | Interest Rate | Issuance | Maturity | |||||
U.S. dollar notes | (a) | $500 | 1.375 | % | February 2016 | February 2019 | |||
U.S. dollar notes | (a) | $750 | 1.875 | % | February 2016 | February 2021 | |||
U.S. dollar notes | (a) | $750 | 2.750 | % | February 2016 | February 2026 | |||
U.S. dollar notes | (b) | $500 | 2.125 | % | May 2016 | May 2023 | |||
U.S. dollar notes | (b) | $500 | 4.250 | % | May 2016 | (d) | November 2044 | ||
EURO notes | (c) | €500 (approximately $578) | 2.000 | % | May 2016 | May 2036 | |||
(a) Interest on these notes is payable semi-annually in arrears beginning in August 2016.
(b) Interest on these notes is payable semi-annually in arrears beginning in November 2016.
(c) Interest on these notes is payable annually in arrears beginning in May 2017.
(d) These notes are a further issuance of the 4.25% notes issued by PMI in November 2014.
The net proceeds from the sale of the securities listed in the table above were used for general corporate purposes.
The weighted-average time to maturity of our long-term debt was 10.6 years at the end of 2016 and 10.5 years at the end of 2015.
• | Off-Balance Sheet Arrangements and Aggregate Contractual Obligations |
We have no off-balance sheet arrangements, including special purpose entities, other than guarantees and contractual obligations discussed below.
Guarantees – At December 31, 2016, we were contingently liable for $0.8 billion of guarantees of our own performance, which were primarily related to excise taxes on the shipment of our products. There is no liability in the consolidated financial statements associated with these guarantees. At December 31, 2016, our third-party guarantees were insignificant.
74
Aggregate Contractual Obligations – The following table summarizes our contractual obligations at December 31, 2016:
Payments Due | |||||||||||||||
Total | 2017 | 2018-2019 | 2020-2021 | 2022 and Thereafter | |||||||||||
(in millions) | |||||||||||||||
Long-term debt (1) | $28,674 | $2,573 | $5,006 | $5,549 | $15,546 | ||||||||||
Interest on borrowings (2) | 10,969 | 911 | 1,529 | 1,261 | 7,268 | ||||||||||
Operating leases (3) | 662 | 182 | 208 | 108 | 164 | ||||||||||
Purchase obligations (4): | |||||||||||||||
Inventory and production costs | 3,871 | 1,571 | 1,010 | 525 | 765 | ||||||||||
Other | 1,474 | 1,122 | 298 | 46 | 8 | ||||||||||
5,345 | 2,693 | 1,308 | 571 | 773 | |||||||||||
Other long-term liabilities (5) | 462 | 62 | 75 | 38 | 287 | ||||||||||
$46,112 | $6,421 | $8,126 | $7,527 | $24,038 | |||||||||||
(1) Amounts represent the expected cash payments of our long-term debt and capital lease obligations.
(2) Amounts represent the expected cash payments of our interest expense on our long-term debt, including the current portion of long-term debt. Interest on our fixed-rate debt is presented using the stated interest rate. Amounts exclude the amortization of debt discounts, the amortization of loan fees and fees for lines of credit that would be included in interest expense in the consolidated statements of earnings.
(3) Amounts represent the minimum rental commitments under non-cancelable operating leases.
(4) Purchase obligations for inventory and production costs (such as raw materials, indirect materials and supplies, packaging, co-manufacturing arrangements, storage and distribution) are commitments for projected needs to be utilized in the normal course of business. Other purchase obligations include commitments for marketing, advertising, capital expenditures, information technology and professional services. Arrangements are considered purchase obligations if a contract specifies all significant terms, including fixed or minimum quantities to be purchased, a pricing structure and approximate timing of the transaction. Amounts represent the minimum commitments under non-cancelable contracts. Any amounts reflected on the consolidated balance sheet as accounts payable and accrued liabilities are excluded from the table above.
(5) Other long-term liabilities consist primarily of postretirement health care costs and accruals established for employment costs. The following long-term liabilities included on the consolidated balance sheet are excluded from the table above: accrued pension and postemployment costs, tax contingencies, insurance accruals and other accruals. We are unable to estimate the timing of payments (or contributions in the case of accrued pension costs) for these items. Currently, we anticipate making pension contributions of approximately $70 million in 2017, based on current tax and benefit laws (as discussed in Item 8, Note 13. Benefit Plans to our consolidated financial statements).
Ÿ Equity and Dividends
We discuss our stock awards as of December 31, 2016, in Item 8, Note 9. Stock Plans to our consolidated financial statements.
On August 1, 2012, we began repurchasing shares under a three-year $18.0 billion share repurchase program that was authorized by our Board of Directors in June 2012. From August 1, 2012, through December 31, 2014, we repurchased 144.6 million shares of our common stock at a cost of $12.7 billion under this repurchase program. Our authorized three-year share repurchase program of $18.0 billion expired in August 2015. During 2016 and 2015, we did not repurchase any shares of our common stock and we do not presently intend to repurchase shares of our common stock in 2017.
Dividends paid in 2016 were $6.4 billion. During the third quarter of 2016, our Board of Directors approved a 2.0% increase in the quarterly dividend to $1.04 per common share. As a result, the present annualized dividend rate is $4.16 per common share.
Market Risk
Ÿ Counterparty Risk - We predominantly work with financial institutions with strong short- and long-term credit ratings as assigned by Standard & Poor’s and Moody’s. These banks are also part of a defined group of relationship banks. Non-investment grade institutions are only used in certain emerging markets to the extent required by local business needs. We have a conservative approach when it comes to choosing financial counterparties and financial instruments. As such we do not invest or hold investments in any structured or equity-linked products. The majority of our cash and cash equivalents is currently invested in demand deposits maturing within less than 30 days.
We continuously monitor and assess the credit worthiness of all our counterparties.
75
Ÿ Derivative Financial Instruments - We operate in markets outside of the U.S., with manufacturing and sales facilities in various locations throughout the world. Consequently, we use certain financial instruments to manage our foreign currency and interest rate exposure. We use derivative financial instruments principally to reduce our exposure to market risks resulting from fluctuations in foreign exchange rates by creating offsetting exposures. We are not a party to leveraged derivatives and, by policy, do not use derivative financial instruments for speculative purposes.
See Item 8, Note 15. Financial Instruments, Item 8, Note 16. Fair Value Measurements and Item 8, Note 20. Balance Sheet Offsetting to our consolidated financial statements for further details on our derivative financial instruments and the related collateral arrangements.
Ÿ Value at Risk - We use a value at risk computation to estimate the potential one-day loss in the fair value of our interest-rate-sensitive financial instruments and to estimate the potential one-day loss in pre-tax earnings of our foreign currency price-sensitive derivative financial instruments. This computation includes our debt, short-term investments, and foreign currency forwards, swaps and options. Anticipated transactions, foreign currency trade payables and receivables, and net investments in foreign subsidiaries, which the foregoing instruments are intended to hedge, were excluded from the computation.
The computation estimates were made assuming normal market conditions, using a 95% confidence interval. We use a “variance/co-variance” model to determine the observed interrelationships between movements in interest rates and various currencies. These interrelationships were determined by observing interest rate and forward currency rate movements over the preceding quarter for determining value at risk at December 31, 2016 and 2015, and over each of the four preceding quarters for the calculation of average value at risk amounts during each year. The values of foreign currency options do not change on a one-to-one basis with the underlying currency and were valued accordingly in the computation.
The estimated potential one-day loss in fair value of our interest-rate-sensitive instruments, primarily debt, under normal market conditions and the estimated potential one-day loss in pre-tax earnings from foreign currency instruments under normal market conditions, as calculated in the value at risk model, were as follows:
Pre-Tax Earnings Impact | |||||||
(in millions) | At 12/31/16 | Average | High | Low | |||
Instruments sensitive to: | |||||||
Foreign currency rates | $63 | $58 | $87 | $34 | |||
Fair Value Impact | |||||||
(in millions) | At 12/31/16 | Average | High | Low | |||
Instruments sensitive to: | |||||||
Interest rates | $143 | $147 | $217 | $112 | |||
Pre-Tax Earnings Impact | |||||||
(in millions) | At 12/31/15 | Average | High | Low | |||
Instruments sensitive to: | |||||||
Foreign currency rates | $65 | $74 | $96 | $62 | |||
Fair Value Impact | |||||||
(in millions) | At 12/31/15 | Average | High | Low | |||
Instruments sensitive to: | |||||||
Interest rates | $102 | $120 | $147 | $102 | |||
The value at risk computation is a risk analysis tool designed to statistically estimate the maximum probable daily loss from adverse movements in interest and foreign currency rates under normal market conditions. The computation does not purport to represent actual losses in fair value or earnings to be incurred by us, nor does it consider the effect of favorable changes in market rates. We cannot predict
76
actual future movements in such market rates and do not present these results to be indicative of future movements in market rates or to be representative of any actual impact that future changes in market rates may have on our future results of operations or financial position.
Contingencies
See Item 3 and Item 8, Note 19. Contingencies to our consolidated financial statements for a discussion of contingencies.
Cautionary Factors That May Affect Future Results
Forward-Looking and Cautionary Statements
We may from time to time make written or oral forward-looking statements, including statements contained in filings with the SEC, in reports to stockholders and in press releases and investor webcasts. You can identify these forward-looking statements by use of words such as "strategy," "expects," "continues," "plans," "anticipates," "believes," "will," "estimates," "intends," "projects," "goals," "targets" and other words of similar meaning. You can also identify them by the fact that they do not relate strictly to historical or current facts.
We cannot guarantee that any forward-looking statement will be realized, although we believe we have been prudent in our plans and assumptions. Achievement of future results is subject to risks, uncertainties and inaccurate assumptions. Should known or unknown risks or uncertainties materialize, or should underlying assumptions prove inaccurate, actual results could vary materially from those anticipated, estimated or projected. Investors should bear this in mind as they consider forward-looking statements and whether to invest in or remain invested in our securities. In connection with the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, we are identifying important factors that, individually or in the aggregate, could cause actual results and outcomes to differ materially from those contained in any forward-looking statements made by us; any such statement is qualified by reference to the following cautionary statements. We elaborate on these and other risks we face throughout this document, particularly in Item 1A. Risk Factors, and Business Environment of this section. You should understand that it is not possible to predict or identify all risk factors. Consequently, you should not consider the following to be a complete discussion of all potential risks or uncertainties. We do not undertake to update any forward-looking statement that we may make from time to time, except in the normal course of our public disclosure obligations.
Item 7A. | Quantitative and Qualitative Disclosures About Market Risk. |
The information called for by this Item is included in Item 7, Market Risk.
77
Item 8. | Financial Statements and Supplementary Data. |
Consolidated Balance Sheets
(in millions of dollars, except share data)
at December 31, | 2016 | 2015 | |||||
Assets | |||||||
Cash and cash equivalents | $ | 4,239 | $ | 3,417 | |||
Receivables (less allowances of $42 in 2016 and $58 in 2015) | 3,499 | 2,778 | |||||
Inventories: | |||||||
Leaf tobacco | 2,498 | 2,640 | |||||
Other raw materials | 1,569 | 1,613 | |||||
Finished product | 4,950 | 4,220 | |||||
9,017 | 8,473 | ||||||
Deferred income taxes | — | 488 | |||||
Other current assets | 853 | 648 | |||||
Total current assets | 17,608 | 15,804 | |||||
Property, plant and equipment, at cost: | |||||||
Land and land improvements | 590 | 583 | |||||
Buildings and building equipment | 3,474 | 3,361 | |||||
Machinery and equipment | 7,366 | 6,978 | |||||
Construction in progress | 930 | 845 | |||||
12,360 | 11,767 | ||||||
Less: accumulated depreciation | 6,296 | 6,046 | |||||
6,064 | 5,721 | ||||||
Goodwill (Note 3) | 7,324 | 7,415 | |||||
Other intangible assets, net (Note 3) | 2,470 | 2,623 | |||||
Investments in unconsolidated subsidiaries (Note 4) | 1,011 | 890 | |||||
Deferred income taxes | 859 | 360 | |||||
Other assets | 1,515 | 1,143 | |||||
Total Assets | $ | 36,851 | $ | 33,956 | |||
See notes to consolidated financial statements.
78
at December 31, | 2016 | 2015 | |||||
Liabilities | |||||||
Short-term borrowings (Note 7) | $ | 643 | $ | 825 | |||
Current portion of long-term debt (Note 7) | 2,573 | 2,405 | |||||
Accounts payable | 1,666 | 1,289 | |||||
Accrued liabilities: | |||||||
Marketing and selling | 575 | 640 | |||||
Taxes, except income taxes | 6,204 | 5,121 | |||||
Employment costs | 800 | 903 | |||||
Dividends payable | 1,621 | 1,589 | |||||
Other | 1,553 | 1,438 | |||||
Income taxes | 832 | 970 | |||||
Deferred income taxes | — | 206 | |||||
Total current liabilities | 16,467 | 15,386 | |||||
Long-term debt (Note 7) | 25,851 | 25,250 | |||||
Deferred income taxes | 1,897 | 1,543 | |||||
Employment costs | 2,800 | 2,566 | |||||
Other liabilities | 736 | 687 | |||||
Total liabilities | 47,751 | 45,432 | |||||
Contingencies (Note 19) | |||||||
Stockholders’ (Deficit) Equity | |||||||
Common stock, no par value (2,109,316,331 shares issued in 2016 and 2015) | — | — | |||||
Additional paid-in capital | 1,964 | 1,929 | |||||
Earnings reinvested in the business | 30,397 | 29,842 | |||||
Accumulated other comprehensive losses | (9,559 | ) | (9,402 | ) | |||
22,802 | 22,369 | ||||||
Less: cost of repurchased stock (557,930,784 and 559,972,262 shares in 2016 and 2015, respectively) | 35,490 | 35,613 | |||||
Total PMI stockholders’ deficit | (12,688 | ) | (13,244 | ) | |||
Noncontrolling interests | 1,788 | 1,768 | |||||
Total stockholders’ deficit | (10,900 | ) | (11,476 | ) | |||
Total Liabilities and Stockholders’ (Deficit) Equity | $ | 36,851 | $ | 33,956 | |||
See notes to consolidated financial statements.
79
Consolidated Statements of Earnings
(in millions of dollars, except per share data)
for the years ended December 31, | 2016 | 2015 | 2014 | ||||||||
Net revenues | $ | 74,953 | $ | 73,908 | $ | 80,106 | |||||
Cost of sales | 9,391 | 9,365 | 10,436 | ||||||||
Excise taxes on products | 48,268 | 47,114 | 50,339 | ||||||||
Gross profit | 17,294 | 17,429 | 19,331 | ||||||||
Marketing, administration and research costs | 6,405 | 6,656 | 7,001 | ||||||||
Asset impairment and exit costs (Note 5) | — | 68 | 535 | ||||||||
Amortization of intangibles | 74 | 82 | 93 | ||||||||
Operating income | 10,815 | 10,623 | 11,702 | ||||||||
Interest expense, net (Note 14) | 891 | 1,008 | 1,052 | ||||||||
Earnings before income taxes | 9,924 | 9,615 | 10,650 | ||||||||
Provision for income taxes | 2,768 | 2,688 | 3,097 | ||||||||
Equity (income)/loss in unconsolidated subsidiaries, net | (94 | ) | (105 | ) | (105 | ) | |||||
Net earnings | 7,250 | 7,032 | 7,658 | ||||||||
Net earnings attributable to noncontrolling interests | 283 | 159 | 165 | ||||||||
Net earnings attributable to PMI | $ | 6,967 | $ | 6,873 | $ | 7,493 | |||||
Per share data (Note 10): | |||||||||||
Basic earnings per share | $ | 4.48 | $ | 4.42 | $ | 4.76 | |||||
Diluted earnings per share | $ | 4.48 | $ | 4.42 | $ | 4.76 | |||||
See notes to consolidated financial statements.
80
Consolidated Statements of Comprehensive Earnings
(in millions of dollars)
for the years ended December 31, | 2016 | 2015 | 2014 | ||||||||
Net earnings | $ | 7,250 | $ | 7,032 | $ | 7,658 | |||||
Other comprehensive earnings (losses), net of income taxes: | |||||||||||
Change in currency translation adjustments: | |||||||||||
Unrealized gains (losses), net of income taxes of ($101) in 2016, ($143) in 2015 and ($161) in 2014 | (14 | ) | (2,248 | ) | (1,746 | ) | |||||
(Gains)/losses transferred to earnings, net of income taxes of $- in 2016, 2015 and 2014 | 5 | (1 | ) | (5 | ) | ||||||
Change in net loss and prior service cost: | |||||||||||
Net losses and prior service costs, net of income taxes of $78 in 2016, $17 in 2015 and $167 in 2014 | (460 | ) | (536 | ) | (1,148 | ) | |||||
Amortization of net losses, prior service costs and net transition costs, net of income taxes of ($43) in 2016, ($48) in 2015 and ($42) in 2014 | 224 | 227 | 173 | ||||||||
Change in fair value of derivatives accounted for as hedges: | |||||||||||
Gains (losses) recognized, net of income taxes of ($4) in 2016, ($5) in 2015 and ($13) in 2014 | 8 | 38 | 98 | ||||||||
(Gains) losses transferred to earnings, net of income taxes of ($3) in 2016, $14 in 2015 and $10 in 2014 | 30 | (102 | ) | (38 | ) | ||||||
Total other comprehensive losses | (207 | ) | (2,622 | ) | (2,666 | ) | |||||
Total comprehensive earnings | 7,043 | 4,410 | 4,992 | ||||||||
Less comprehensive earnings attributable to: | |||||||||||
Noncontrolling interests | 233 | 113 | 135 | ||||||||
Comprehensive earnings attributable to PMI | $ | 6,810 | $ | 4,297 | $ | 4,857 | |||||
See notes to consolidated financial statements.
81
Consolidated Statements of Stockholders' (Deficit) Equity
(in millions of dollars, except per share data)
PMI Stockholders’ (Deficit) Equity | |||||||||||||||||||||||||||
Common Stock | Additional Paid-in Capital | Earnings Reinvested in the Business | Accumulated Other Comprehensive Losses | Cost of Repurchased Stock | Noncontrolling Interests | Total | |||||||||||||||||||||
Balances, January 1, 2014 | $ | — | $ | 723 | $ | 27,843 | $ | (4,190 | ) | $ | (32,142 | ) | $ | 1,492 | $ | (6,274 | ) | ||||||||||
Net earnings | 7,493 | 165 | 7,658 | ||||||||||||||||||||||||
Other comprehensive earnings (losses), net of income taxes | (2,636 | ) | (30 | ) | (2,666 | ) | |||||||||||||||||||||
Issuance of stock awards and exercise of stock options | (13 | ) | 180 | 167 | |||||||||||||||||||||||
Dividends declared ($3.88 per share) | (6,087 | ) | (6,087 | ) | |||||||||||||||||||||||
Payments to noncontrolling interests | (207 | ) | (207 | ) | |||||||||||||||||||||||
Common stock repurchased | (3,800 | ) | (3,800 | ) | |||||||||||||||||||||||
Other | 6 | 6 | |||||||||||||||||||||||||
Balances, December 31, 2014 | — | 710 | 29,249 | (6,826 | ) | (35,762 | ) | 1,426 | (11,203 | ) | |||||||||||||||||
Net earnings | 6,873 | 159 | 7,032 | ||||||||||||||||||||||||
Other comprehensive earnings (losses), net of income taxes | (2,576 | ) | (46 | ) | (2,622 | ) | |||||||||||||||||||||
Issuance of stock awards | (3 | ) | 149 | 146 | |||||||||||||||||||||||
Dividends declared ($4.04 per share) | (6,280 | ) | (6,280 | ) | |||||||||||||||||||||||
Payments to noncontrolling interests | (171 | ) | (171 | ) | |||||||||||||||||||||||
Sale (purchase) of subsidiary shares to/(from) noncontrolling interests (Note 6) | 1,222 | 400 | 1,622 | ||||||||||||||||||||||||
Balances, December 31, 2015 | — | 1,929 | 29,842 | (9,402 | ) | (35,613 | ) | 1,768 | (11,476 | ) | |||||||||||||||||
Net earnings | 6,967 | 283 | 7,250 | ||||||||||||||||||||||||
Other comprehensive earnings (losses), net of income taxes | (157 | ) | (50 | ) | (207 | ) | |||||||||||||||||||||
Issuance of stock awards | 37 | 123 | 160 | ||||||||||||||||||||||||
Dividends declared ($4.12 per share) | (6,412 | ) | (6,412 | ) | |||||||||||||||||||||||
Payments to noncontrolling interests | (219 | ) | (219 | ) | |||||||||||||||||||||||
Other | (2 | ) | 6 | 4 | |||||||||||||||||||||||
Balances, December 31, 2016 | $ | — | $ | 1,964 | $ | 30,397 | $ | (9,559 | ) | $ | (35,490 | ) | $ | 1,788 | $ | (10,900 | ) | ||||||||||
See notes to consolidated financial statements.
82
Consolidated Statements of Cash Flows
(in millions of dollars)
for the years ended December 31, | 2016 | 2015 | 2014 | ||||||||
CASH PROVIDED BY (USED IN) OPERATING ACTIVITIES | |||||||||||
Net earnings | $ | 7,250 | $ | 7,032 | $ | 7,658 | |||||
Adjustments to reconcile net earnings to operating cash flows: | |||||||||||
Depreciation and amortization | 743 | 754 | 889 | ||||||||
Deferred income tax (benefit) provision | 182 | (18 | ) | (62 | ) | ||||||
Asset impairment and exit costs, net of cash paid | (31 | ) | (164 | ) | 175 | ||||||
Cash effects of changes, net of the effects from acquired companies: | |||||||||||
Receivables, net | (1,009 | ) | 647 | (463 | ) | ||||||
Inventories | (695 | ) | (841 | ) | 105 | ||||||
Accounts payable | 373 | 310 | 177 | ||||||||
Income taxes | (209 | ) | (42 | ) | (230 | ) | |||||
Accrued liabilities and other current assets | 1,477 | (8 | ) | (507 | ) | ||||||
Pension plan contributions | (191 | ) | (154 | ) | (191 | ) | |||||
Other | 187 | 349 | 188 | ||||||||
Net cash provided by operating activities | 8,077 | 7,865 | 7,739 | ||||||||
CASH PROVIDED BY (USED IN) INVESTING ACTIVITIES | |||||||||||
Capital expenditures | (1,172 | ) | (960 | ) | (1,153 | ) | |||||
Investments in unconsolidated subsidiaries | (41 | ) | (55 | ) | (29 | ) | |||||
Purchase of businesses, net of acquired cash | — | — | (110 | ) | |||||||
Other | 245 | 307 | 296 | ||||||||
Net cash used in investing activities | (968 | ) | (708 | ) | (996 | ) | |||||
See notes to consolidated financial statements.
83
for the years ended December 31, | 2016 | 2015 | 2014 | ||||||||
CASH PROVIDED BY (USED IN) FINANCING ACTIVITIES | |||||||||||
Short-term borrowing activity by original maturity: | |||||||||||
Net repayments - maturities of 90 days or less | $ | (12 | ) | $ | (266 | ) | $ | (516 | ) | ||
Issuances - maturities longer than 90 days | — | — | 1,007 | ||||||||
Repayments - maturities longer than 90 days | — | — | (1,571 | ) | |||||||
Long-term debt proceeds | 3,536 | 1,539 | 5,591 | ||||||||
Long-term debt repaid | (2,393 | ) | (1,229 | ) | (1,240 | ) | |||||
Repurchases of common stock | — | (48 | ) | (3,833 | ) | ||||||
Dividends paid | (6,378 | ) | (6,250 | ) | (6,035 | ) | |||||
Sale (purchase) of subsidiary shares to/(from) noncontrolling interests (Note 6) | 7 | 1,622 | — | ||||||||
Other | (173 | ) | (104 | ) | (242 | ) | |||||
Net cash used in financing activities | (5,413 | ) | (4,736 | ) | (6,839 | ) | |||||
Effect of exchange rate changes on cash and cash equivalents | (874 | ) | (686 | ) | (376 | ) | |||||
Cash and cash equivalents: | |||||||||||
Increase (Decrease) | 822 | 1,735 | (472 | ) | |||||||
Balance at beginning of year | 3,417 | 1,682 | 2,154 | ||||||||
Balance at end of year | $ | 4,239 | $ | 3,417 | $ | 1,682 | |||||
Cash Paid: | |||||||||||
Interest | $ | 1,052 | $ | 1,045 | $ | 1,068 | |||||
Income taxes | $ | 2,829 | $ | 2,771 | $ | 3,577 | |||||
See notes to consolidated financial statements.
84
Notes to Consolidated Financial Statements
Note 1. | ||||
Background and Basis of Presentation:
Background
Philip Morris International Inc. is a holding company incorporated in Virginia, U.S.A., whose subsidiaries and affiliates and their licensees are engaged in the manufacture and sale of cigarettes, other tobacco products and other nicotine-containing products in markets outside of the United States of America. Throughout these financial statements, the term "PMI" refers to Philip Morris International Inc. and its subsidiaries.
Basis of presentation
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America ("U.S. GAAP") requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent liabilities at the dates of the financial statements and the reported amounts of net revenues and expenses during the reporting periods. Significant estimates and assumptions include, among other things: pension and benefit plan assumptions; useful lives and valuation assumptions of goodwill and other intangible assets; marketing programs, and income taxes. Actual results could differ from those estimates.
The consolidated financial statements include PMI, as well as its wholly owned and majority-owned subsidiaries. Investments in which PMI exercises significant influence (generally 20%-50% ownership interest) are accounted for under the equity method of accounting. Investments in which PMI has an ownership interest of less than 20%, or does not exercise significant influence, are accounted for under the cost method of accounting. All intercompany transactions and balances have been eliminated.
Certain prior years' amounts have been reclassified to conform with the current year's presentation, due primarily to new accounting guidance related to deferred income taxes and fair value of certain assets that are to be measured at net asset value. For further details, see Note 13. Benefit Plans, Note 16. Fair Value Measurements and Note 22. New Accounting Standards. The reclassifications did not have an impact on PMI's consolidated financial position, results of operations or cash flows in any of the periods presented.
Note 2. | |
Summary of Significant Accounting Policies:
Cash and cash equivalents
Cash equivalents include demand deposits with banks and all highly liquid investments with original maturities of three months or less.
Depreciation
Property, plant and equipment are stated at historical cost and depreciated by the straight-line method over the estimated useful lives of the assets. Machinery and equipment are depreciated over periods ranging from 3 to 15 years, and buildings and building improvements over periods up to 40 years.
Employee benefit plans
PMI provides a range of benefits to its employees and retired employees, including pensions, postretirement health care and postemployment benefits (primarily severance). PMI records annual amounts relating to these plans based on calculations specified under U.S. GAAP. PMI recognizes the funded status of its defined pension and postretirement plans on the consolidated balance sheets. The funded status is measured as the difference between the fair value of the plans assets and the benefit obligation. PMI measures the plan assets and liabilities at the end of the fiscal year. For defined benefit pension plans, the benefit obligation is the projected benefit obligation. For the postretirement health care plans, the benefit obligation is the accumulated postretirement benefit obligation. Any
85
plan with an overfunded status is recognized as an asset, and any plan with an underfunded status is recognized as a liability. Any gains or losses and prior service costs or credits that have not been recognized as a component of net periodic benefit costs are recorded as a component of other comprehensive earnings (losses), net of deferred taxes. PMI elects to recognize actuarial gains/(losses) using the corridor approach.
Foreign currency translation
PMI translates the results of operations of its subsidiaries and affiliates using average exchange rates during each period, whereas balance sheet accounts are translated using exchange rates at the end of each period. Currency translation adjustments are recorded as a component of stockholders’ (deficit) equity. In addition, some of PMI’s subsidiaries have assets and liabilities denominated in currencies other than their functional currencies, and to the extent those are not designated as net investment hedges, these assets and liabilities generate transaction gains and losses when translated into their respective functional currencies.
Goodwill and non-amortizable intangible assets valuation
PMI tests goodwill and non-amortizable intangible assets for impairment annually or more frequently if events occur that would warrant such review. During the second quarter of 2016, PMI changed the date of its annual goodwill impairment test from the first quarter to the second quarter. The change was made to more closely align the impairment testing date with PMI’s long-range planning and forecasting process. PMI has determined that this change in accounting principle is preferable under the circumstances and believes that the change in the annual impairment testing date did not delay, accelerate, or avoid an impairment charge. The impairment analysis involves comparing the fair value of each reporting unit or non-amortizable intangible asset to the carrying value. If the carrying value exceeds the fair value, goodwill or a non-amortizable intangible asset is considered impaired.
Hedging instruments
Derivative financial instruments are recorded at fair value on the consolidated balance sheets as either assets or liabilities. Changes in the fair value of derivatives are recorded each period either in accumulated other comprehensive losses on the consolidated balance sheet, or in earnings, depending on whether a derivative is designated and effective as part of a hedge transaction and, if it is, the type of hedge transaction. Gains and losses on derivative instruments reported in accumulated other comprehensive losses are reclassified to the consolidated statements of earnings in the periods in which operating results are affected by the hedged item. Cash flows from hedging instruments are classified in the same manner as the affected hedged item in the consolidated statements of cash flows.
Impairment of long-lived assets
PMI reviews long-lived assets, including amortizable intangible assets, for impairment whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable. PMI performs undiscounted operating cash flow analyses to determine if an impairment exists. For purposes of recognition and measurement of an impairment for assets held for use, PMI groups assets and liabilities at the lowest level for which cash flows are separately identifiable. If an impairment is determined to exist, any related impairment loss is calculated based on fair value. Impairment losses on assets to be disposed of, if any, are based on the estimated proceeds to be received, less costs of disposal.
Impairment of investments in unconsolidated subsidiaries
Investments in unconsolidated subsidiaries are evaluated for impairment whenever events or changes in circumstances indicate that the carrying amount of the investments may not be recoverable. An impairment loss would be recorded whenever a decline in value of an equity investment below its carrying amount is determined to be other than temporary. PMI determines whether a loss is other than temporary by considering the length of time and extent to which the fair value of the equity investment has been less than the carrying amount, the financial condition of the equity investment, and the intent to retain the investment for a period of time is sufficient to allow for any anticipated recovery in market value.
Income taxes
Income tax provisions for jurisdictions outside the United States, as well as state and local income tax provisions, are determined on a separate company basis, and the related assets and liabilities are recorded in PMI’s consolidated balance sheets. Significant judgment is required in determining income tax provisions and in evaluating tax positions. PMI recognizes accrued interest and penalties associated with uncertain tax positions as part of the provision for income taxes on the consolidated statements of earnings.
86
Inventories
Inventories are stated at the lower of cost or market. The first-in, first-out and average cost methods are used to cost substantially all inventories. It is a generally recognized industry practice to classify leaf tobacco inventory as a current asset, although part of such inventory, because of the duration of the aging process, ordinarily would not be utilized within one year.
Marketing costs
PMI supports its products with advertising, adult consumer engagement and trade promotions. Such programs include, but are not limited to, discounts, rebates, in-store display incentives, e-commerce, mobile and other digital platforms, adult consumer activation and promotion activities, as well as costs associated with adult consumer experience outlets and other adult consumer touchpoints and volume-based incentives. Advertising, as well as certain consumer engagement and trade activities costs, are expensed as incurred. Trade promotions are recorded as a reduction of revenues based on amounts estimated as being due to customers at the end of a period, based principally on historical utilization. For interim reporting purposes, advertising and certain consumer engagement expenses are charged to earnings based on estimated sales and related expenses for the full year.
Revenue recognition
PMI recognizes revenues, net of sales incentives and including shipping and handling charges billed to customers, either upon shipment or delivery of goods when title and risk of loss pass to customers. Excise taxes billed by PMI to customers are reported in net revenues. Shipping and handling costs are classified as part of cost of sales.
On May 28, 2014, the Financial Accounting Standards Board issued Accounting Standards Update ASU 2014-09, "Revenue from Contracts with Customers." For further details, see Note 22. New Accounting Standards.
Stock-based compensation
PMI measures compensation cost for all stock-based awards at fair value on date of grant and recognizes the compensation costs over the service periods for awards expected to vest. For further details, see Note 9. Stock Plans.
Note 3. | ||
Goodwill and Other Intangible Assets, net:
Goodwill and other intangible assets, net, by segment were as follows:
Goodwill | Other Intangible Assets, net | ||||||||||||||
(in millions) | December 31, 2016 | December 31, 2015 | December 31, 2016 | December 31, 2015 | |||||||||||
European Union | $ | 1,238 | $ | 1,310 | $ | 479 | $ | 516 | |||||||
Eastern Europe, Middle East & Africa | 372 | 374 | 200 | 201 | |||||||||||
Asia | 3,596 | 3,581 | 1,074 | 1,087 | |||||||||||
Latin America & Canada | 2,118 | 2,150 | 717 | 819 | |||||||||||
Total | $ | 7,324 | $ | 7,415 | $ | 2,470 | $ | 2,623 | |||||||
87
Goodwill primarily reflects PMI’s acquisitions in Canada, Colombia, Greece, Indonesia, Mexico, Pakistan and Serbia, as well as the business combination in the Philippines.
The movements in goodwill were as follows:
(in millions) | European Union | Eastern Europe, Middle East & Africa | Asia | Latin America & Canada | Total | |||||||||||||||
Balance at January 1, 2015 | $ | 1,439 | $ | 476 | $ | 3,904 | $ | 2,569 | $ | 8,388 | ||||||||||
Changes due to: | ||||||||||||||||||||
Currency | (129 | ) | (102 | ) | (323 | ) | (419 | ) | (973 | ) | ||||||||||
Balance at December 31, 2015 | 1,310 | 374 | 3,581 | 2,150 | 7,415 | |||||||||||||||
Changes due to: | ||||||||||||||||||||
Currency | (72 | ) | (2 | ) | 15 | (32 | ) | (91 | ) | |||||||||||
Balance at December 31, 2016 | $ | 1,238 | $ | 372 | $ | 3,596 | $ | 2,118 | $ | 7,324 | ||||||||||
Additional details of other intangible assets were as follows:
December 31, 2016 | December 31, 2015 | ||||||||||||||
(in millions) | Gross Carrying Amount | Accumulated Amortization | Gross Carrying Amount | Accumulated Amortization | |||||||||||
Non-amortizable intangible assets | $ | 1,455 | $ | 1,527 | |||||||||||
Amortizable intangible assets | 1,598 | $ | 583 | 1,609 | $ | 513 | |||||||||
Total other intangible assets | $ | 3,053 | $ | 583 | $ | 3,136 | $ | 513 | |||||||
Non-amortizable intangible assets substantially consist of trademarks from PMI’s acquisitions in Indonesia in 2005 and Mexico in 2007. Amortizable intangible assets primarily consist of certain trademarks and distribution networks associated with business combinations. The gross carrying amount, the range of useful lives as well as the weighted-average remaining useful life of amortizable intangible assets at December 31, 2016, were as follows:
Description (dollars in millions) | Gross Carrying Amount | Initial Estimated Useful Lives | Weighted-Average Remaining Useful Life | |||
Trademarks | $ | 1,364 | 2 - 40 years | 20 years | ||
Distribution networks | 147 | 5 - 30 years | 10 years | |||
Other (including farmer contracts and intellectual property rights) | 87 | 4 - 17 years | 10 years | |||
$ | 1,598 | |||||
Pre-tax amortization expense for intangible assets during the years ended December 31, 2016, 2015 and 2014, was $74 million, $82 million and $93 million, respectively. Amortization expense for each of the next five years is estimated to be $87 million or less, assuming no additional transactions occur that require the amortization of intangible assets.
The decrease in the gross carrying amount of other intangible assets from December 31, 2015, was due to currency movements.
88
Note 4. | ||||
Investments in Unconsolidated Subsidiaries:
At December 31, 2016 and 2015, PMI had total investments in unconsolidated subsidiaries of $1,011 million and $890 million, respectively, which were accounted for under the equity method of accounting. Equity method investments are initially recorded at cost. Under the equity method of accounting, the investment is adjusted for PMI's proportionate share of earnings or losses and movements in currency translation adjustments. The carrying value of our equity method investments at December 31, 2016 and 2015 exceeded our share of the unconsolidated subsidiaries' book value by $867 million and $806 million, respectively. The difference between the investment carrying value and the amount of underlying equity in net assets, excluding $810 million and $744 million attributable to goodwill as of December 31, 2016 and 2015, respectively, is being amortized on a straight-line basis over the underlying assets' estimated useful lives as of the date of the acquisition of 3 to 20 years. At December 31, 2016 and 2015, PMI received year-to-date dividends from unconsolidated subsidiaries of $117 million and $127 million, respectively.
PMI holds a 49% equity interest in United Arab Emirates-based Emirati Investors-TA (FZC) (“EITA”), formerly Arab Investors-TA (FZC). As a result of this transaction, PMI holds an approximate 25% economic interest in Société des Tabacs Algéro-Emiratie (“STAEM”), an Algerian joint venture that is 51% owned by EITA and 49% by the Algerian state-owned enterprise Société Nationale des Tabacs et Allumettes SpA. STAEM manufactures and distributes under license some of PMI’s brands. The initial investment in EITA was recorded at cost and is included in investments in unconsolidated subsidiaries on the consolidated balance sheets.
In 2013, PMI acquired from Megapolis Investment BV a 20% equity interest in Megapolis Distribution BV, the holding company of CJSC TK Megapolis ("Megapolis"), PMI's distributor in Russia, for a purchase price of $760 million. An additional payment of up to $100 million, which was contingent on Megapolis's operational performance over the four fiscal years following the closing of the transaction, was also to be made by PMI if the performance criteria were satisfied. PMI had also agreed to provide Megapolis Investment BV with a $100 million interest-bearing loan. PMI and Megapolis Investment BV had agreed to set off any future contingent payments owed by PMI against the future repayments due under the loan agreement. Any loan repayments in excess of the contingent consideration earned by the performance of Megapolis were to be repaid, in cash, to PMI on March 31, 2017. PMI had initially recorded a $100 million asset related to the loan receivable and a discounted liability of $86 million related to the contingent consideration. The initial investment in Megapolis was recorded at cost and is included in investments in unconsolidated subsidiaries on the consolidated balance sheets. As of December 31, 2016, Megapolis had satisfied the performance criteria, which resulted in total contingent consideration of $100 million. As required under the terms of the agreement, the amount of the contingent consideration was fully offset against the future repayments due under the loan agreement.
PMI’s earnings activity from unconsolidated subsidiaries was as follows:
For the Years Ended December 31, | |||||||
(in millions) | 2016 | 2015 | |||||
Net revenues | $ | 3,985 | $ | 4,172 | |||
PMI’s balance sheet activity related to unconsolidated subsidiaries was as follows:
At December 31, | |||||||
(in millions) | 2016 | 2015 | |||||
Receivables | $ | 289 | $ | 64 | |||
Notes receivable | $ | — | $ | 100 | |||
Other liabilities | $ | — | $ | 100 | |||
The activity primarily related to agreements with PMI’s unconsolidated subsidiaries within the Eastern Europe, Middle East & Africa segment. These agreements, which are in the ordinary course of business, are primarily for distribution, contract manufacturing and licenses. PMI eliminated its respective share of all significant intercompany transactions with the equity method investees.
89
Note 5. | ||||
Asset Impairment and Exit Costs:
During 2016, PMI did not incur asset impairment and exit costs. During 2015 and 2014, pre-tax asset impairment and exit costs consisted of the following:
(in millions) | 2015 | 2014 | |||||
Separation programs: | |||||||
European Union | $ | 68 | $ | 351 | |||
Eastern Europe, Middle East & Africa | — | 2 | |||||
Asia | — | 35 | |||||
Latin America & Canada | — | 3 | |||||
Total separation programs | 68 | 391 | |||||
Asset impairment charges: | |||||||
European Union | — | 139 | |||||
Latin America & Canada | — | 5 | |||||
Total asset impairment charges | — | 144 | |||||
Asset impairment and exit costs | $ | 68 | $ | 535 | |||
Movement in Exit Cost Liabilities
The movement in exit cost liabilities for PMI was as follows:
(in millions) | |||
Liability balance, January 1, 2015 | $ | 270 | |
Charges, net | 68 | ||
Cash spent | (232 | ) | |
Currency/other | (52 | ) | |
Liability balance, December 31, 2015 | $ | 54 | |
Charges, net | — | ||
Cash spent | (31 | ) | |
Currency/other | (4 | ) | |
Liability balance, December 31, 2016 | $ | 19 | |
Cash payments related to exit costs at PMI were $31 million, $232 million and $360 million for the years ended December 31, 2016, 2015 and 2014, respectively. Future cash payments for exit costs incurred to date are expected to be approximately $19 million and will be substantially paid by the end of 2017.
The pre-tax asset impairment and exit costs shown above are primarily a result of the following:
The Netherlands
On April 4, 2014, PMI announced the initiation by its affiliate, Philip Morris Holland B.V. (“PMH”), of consultations with employee representatives on a proposal to discontinue cigarette production at its factory located in Bergen op Zoom, the Netherlands. PMH reached an agreement with the trade unions and their members on a social plan and ceased cigarette production on September 1, 2014. During 2014, total pre-tax asset impairment and exit costs of $489 million were recorded for this program in the European Union segment. This amount includes employee separation costs of $343 million, asset impairment costs of $139 million and other separation costs of $7 million.
90
Other
Separation Program Charges
PMI recorded other pre-tax separation program charges of $68 million and $41 million for the years ended December 31, 2015 and 2014, respectively. The 2015 other pre-tax separation program charges primarily related to severance costs for the organizational restructuring in the European Union segment. The 2014 other pre-tax separation program charges were primarily related to severance costs for factory closures in Australia and Canada and the restructuring of the U.S. leaf purchasing model.
Asset Impairment Charges
During 2014, PMI recorded other pre-tax asset impairment charges of $5 million related to a factory closure in Canada.
Note 6. | ||||
Acquisitions and Other Business Arrangements:
As announced in June 2015, PMI’s subsidiary PT HM Sampoerna Tbk. (“Sampoerna”), of which PMI held a 98.18% interest, was required to comply with the January 30, 2014, Indonesian Stock Exchange (“IDX”) regulation requiring all listed public companies to have at least a 7.5% public shareholding by January 30, 2016. In order to comply with this requirement, Sampoerna conducted a rights issue (the “Rights Issue”). The exercise price for the rights was set at Rp. 77,000 per share, a 1.349% premium to the closing price on the IDX as of September 30, 2015. In connection with the Rights Issue, PT Philip Morris Indonesia (“PMID”), a fully consolidated subsidiary of PMI, sold 264,209,711 of the rights to third-party investors. Delivery of the rights sold took place on October 26, 2015. The total net proceeds from the Rights Issue were $1.5 billion at prevailing exchange rates on the closing date. The sale of the rights resulted in an increase to PMI's additional paid-in capital of $1.1 billion.
In June 2014, PMI acquired 100% of Nicocigs Limited, a leading U.K.-based e-vapor company, for the final purchase price of $103 million, net of cash acquired, with additional contingent payments of up to $77 million, primarily relating to performance targets over a three-year period. As of December 31, 2016, the performance targets over the three-year period were not met. The effect of this acquisition was not material to PMI's consolidated financial position, results of operations or cash flows in any of the periods presented.
In September 2013, Grupo Carso, S.A.B. de C.V. ("Grupo Carso") sold to PMI its remaining 20% interest in PMI's Mexican tobacco business for $703 million. As a result, PMI now owns 100% of its Mexican tobacco business. A former director of PMI, whose term expired at the Annual Meeting of Shareholders in May 2015, had an affiliation with Grupo Carso. The final purchase price was subject to an adjustment based on the actual performance of the Mexican tobacco business over the three-year period ending two fiscal years after the closing of the purchase. In May 2015, PMI received a payment of $113 million from Grupo Carso as the final purchase price adjustment. This resulted in a total net purchase price of $590 million. In addition, PMI agreed to pay a dividend of approximately $38 million to Grupo Carso related to the earnings of the Mexican tobacco business for the nine months ended September 30, 2013. In March 2014, the dividend was declared and paid. The purchase of the remaining 20% interest resulted in a net decrease to PMI's additional paid-in capital of $559 million.
91
Note 7. | ||||
Indebtedness:
Short-Term Borrowings
At December 31, 2016 and 2015, PMI’s short-term borrowings and related average interest rates consisted of the following:
December 31, 2016 | December 31, 2015 | ||||||||||||
(in millions) | Amount Outstanding | Average Year-End Rate | Amount Outstanding | Average Year-End Rate | |||||||||
Commercial paper | $ | — | — | % | $ | — | — | % | |||||
Bank loans | 643 | 5.0 | 825 | 6.1 | |||||||||
$ | 643 | $ | 825 | ||||||||||
Given the mix of subsidiaries and their respective local economic environments, the average interest rate for bank loans above can vary significantly from day to day and country to country.
The fair values of PMI’s short-term borrowings at December 31, 2016 and 2015, based upon current market interest rates, approximate the amounts disclosed above.
Long-Term Debt
At December 31, 2016 and 2015, PMI’s long-term debt consisted of the following:
December 31, | |||||||
(in millions) | 2016 | 2015 | |||||
U.S. dollar notes, 1.125% to 6.375% (average interest rate 3.662%), due through 2044 | $ | 19,857 | $ | 18,091 | |||
Foreign currency obligations: | |||||||
Euro notes, 1.750% to 3.125% (average interest rate 2.400%), due through 2036 | 6,828 | 7,423 | |||||
Swiss franc notes, 0.750% to 2.000% (average interest rate 1.269%), due through 2024 | 1,312 | 1,690 | |||||
Other (average interest rate 3.162%), due through 2024 | 427 | 451 | |||||
28,424 | 27,655 | ||||||
Less current portion of long-term debt | 2,573 | 2,405 | |||||
$ | 25,851 | $ | 25,250 | ||||
Other debt:
Other foreign currency debt above includes mortgage debt in Switzerland, capital lease obligations and a bank loan in the Philippines.
92
Debt Issuances Outstanding:
PMI’s debt issuances outstanding at December 31, 2016, were as follows:
(in millions) | ||||||||
Type | Face Value | Interest Rate | Issuance | Maturity | ||||
U.S. dollar notes | $550 | 1.625% | March 2012 | March 2017 | ||||
U.S. dollar notes | $750 | 1.125% | August 2012 | August 2017 | ||||
U.S. dollar notes | $500 | 1.250% | August 2015 | August 2017 | ||||
U.S. dollar notes | $500 | 1.250% | November 2014 | November 2017 | ||||
U.S. dollar notes | $2,500 | 5.650% | May 2008 | May 2018 | ||||
U.S. dollar notes | $750 | 1.875% | November 2013 | January 2019 | ||||
U.S. dollar notes | $500 | 1.375% | February 2016 | February 2019 | ||||
U.S. dollar notes | $1,000 | 4.500% | March 2010 | March 2020 | ||||
U.S. dollar notes | $750 | 1.875% | February 2016 | February 2021 | ||||
U.S. dollar notes | $350 | 4.125% | May 2011 | May 2021 | ||||
U.S. dollar notes | $750 | 2.900% | November 2011 | November 2021 | ||||
U.S. dollar notes | $750 | 2.500% | August 2012 | August 2022 | ||||
U.S. dollar notes | $600 | 2.625% | March 2013 | March 2023 | ||||
U.S. dollar notes | $500 | 2.125% | May 2016 | May 2023 | ||||
U.S. dollar notes | $500 | 3.600% | November 2013 | November 2023 | ||||
U.S. dollar notes | $750 | 3.250% | November 2014 | November 2024 | ||||
U.S. dollar notes | $750 | 3.375% | August 2015 | August 2025 | ||||
U.S. dollar notes | $750 | 2.750% | February 2016 | February 2026 | ||||
U.S. dollar notes | $1,500 | 6.375% | May 2008 | May 2038 | ||||
U.S. dollar notes | $750 | 4.375% | November 2011 | November 2041 | ||||
U.S. dollar notes | $700 | 4.500% | March 2012 | March 2042 | ||||
U.S. dollar notes | $750 | 3.875% | August 2012 | August 2042 | ||||
U.S. dollar notes | $850 | 4.125% | March 2013 | March 2043 | ||||
U.S. dollar notes | $750 | 4.875% | November 2013 | November 2043 | ||||
U.S. dollar notes | $750 | 4.250% | November 2014 | November 2044 | ||||
U.S. dollar notes | $500 | 4.250% | May 2016(a) | November 2044 | ||||
EURO notes | (b) | €750 (approximately $951) | 2.125% | May 2012 | May 2019 | |||
EURO notes | (b) | €1,250 (approximately $1,621) | 1.750% | March 2013 | March 2020 | |||
EURO notes | (b) | €750 (approximately $1,029) | 1.875% | March 2014 | March 2021 | |||
EURO notes | (b) | €600 (approximately $761) | 2.875% | May 2012 | May 2024 | |||
EURO notes | (b) | €750 (approximately $972) | 2.750% | March 2013 | March 2025 | |||
EURO notes | (b) | €1,000 (approximately $1,372) | 2.875% | March 2014 | March 2026 | |||
EURO notes | (b) | €500 (approximately $697) | 2.875% | May 2014 | May 2029 | |||
EURO notes | (b) | €500 (approximately $648) | 3.125% | June 2013 | June 2033 | |||
EURO notes | (b) | €500 (approximately $578) | 2.000% | May 2016 | May 2036 | |||
Swiss franc notes | (b) | CHF200 (approximately $217) | 0.875% | March 2013 | March 2019 | |||
Swiss franc notes | (b) | CHF275 (approximately $311) | 0.750% | May 2014 | December 2019 | |||
Swiss franc notes | (b) | CHF325 (approximately $334) | 1.000% | September 2012 | September 2020 | |||
Swiss franc notes | (b) | CHF300 (approximately $335) | 2.000% | December 2011 | December 2021 | |||
Swiss franc notes | (b) | CHF250 (approximately $283) | 1.625% | May 2014 | May 2024 | |||
(a) These notes are a further issuance of the 4.250% notes issued by PMI in November 2014.
(b) USD equivalents for foreign currency notes were calculated based on exchange rates on the date of issuance.
93
The net proceeds from the sale of the securities listed in the table above were used for general corporate purposes, including working capital requirements and repurchase of PMI's common stock.
Aggregate maturities:
Aggregate maturities of long-term debt are as follows:
(in millions) | |||
2017 | $ | 2,573 | |
2018 | 2,506 | ||
2019 | 2,500 | ||
2020 | 2,623 | ||
2021 | 2,926 | ||
2022-2026 | 7,430 | ||
2027-2031 | 522 | ||
Thereafter | 7,594 | ||
28,674 | |||
Debt discounts | (250 | ) | |
Total long-term debt | $ | 28,424 | |
See Note 16. Fair Value Measurements for additional disclosures related to the fair value of PMI’s debt.
Credit Facilities
On January 27, 2016, PMI entered into an agreement to amend and extend its existing $2.0 billion 364-day revolving credit facility from February 9, 2016, to February 7, 2017. On January 27, 2016, PMI also entered into an agreement to extend the term of its existing $2.5 billion multi-year revolving credit facility from February 28, 2020, to February 28, 2021.
At December 31, 2016, PMI’s total committed credit facilities and commercial paper outstanding were as follows:
Type (in billions of dollars) | Committed Credit Facilities | Commercial Paper | |||||
364-day revolving credit, expiring February 7, 2017 | $ | 2.0 | |||||
Multi-year revolving credit, expiring February 28, 2021 | 2.5 | ||||||
Multi-year revolving credit, expiring October 1, 2020 (1) | 3.5 | ||||||
Total facilities | $ | 8.0 | |||||
Commercial paper outstanding | $ | — | |||||
(1) On August 30, 2016, PMI entered into an agreement, effective October 1, 2016, to extend the term of its multi-year revolving credit facility, for an additional year covering the period October 1, 2020, to October 1, 2021, in the amount of $3.35 billion.
At December 31, 2016, there were no borrowings under these committed credit facilities, and the entire committed amounts were available for borrowing.
On January 27, 2017, PMI entered into an agreement to extend the term of its $2.0 billion 364-day revolving credit facility from February 7, 2017, to February 6, 2018.
94
Each of these facilities requires PMI to maintain a ratio of consolidated earnings before interest, taxes, depreciation and amortization (“consolidated EBITDA”) to consolidated interest expense of not less than 3.5 to 1.0 on a rolling four-quarter basis. At December 31, 2016, PMI’s ratio calculated in accordance with the agreements was 10.6 to 1.0. These facilities do not include any credit rating triggers, material adverse change clauses or any provisions that could require PMI to post collateral. The terms “consolidated EBITDA” and “consolidated interest expense,” both of which include certain adjustments, are defined in the facility agreements previously filed with the Securities and Exchange Commission.
In addition to the committed credit facilities discussed above, certain subsidiaries maintain short-term credit arrangements to meet their respective working capital needs. These credit arrangements, which amounted to approximately $2.9 billion at December 31, 2016 and December 31, 2015, respectively, are for the sole use of the subsidiaries. Borrowings under these arrangements amounted to $643 million at December 31, 2016, and $825 million at December 31, 2015.
Note 8. | ||||
Capital Stock:
Shares of authorized common stock are 6.0 billion; issued, repurchased and outstanding shares were as follows:
Shares Issued | Shares Repurchased | Shares Outstanding | ||||||
Balances, January 1, 2014 | 2,109,316,331 | (520,313,919 | ) | 1,589,002,412 | ||||
Repurchase of shares | (45,206,473 | ) | (45,206,473 | ) | ||||
Issuance of stock awards and exercise of stock options | 3,103,757 | 3,103,757 | ||||||
Balances, December 31, 2014 | 2,109,316,331 | (562,416,635 | ) | 1,546,899,696 | ||||
Issuance of stock awards | 2,444,373 | 2,444,373 | ||||||
Balances, December 31, 2015 | 2,109,316,331 | (559,972,262 | ) | 1,549,344,069 | ||||
Issuance of stock awards | 2,041,478 | 2,041,478 | ||||||
Balances, December 31, 2016 | 2,109,316,331 | (557,930,784 | ) | 1,551,385,547 | ||||
On August 1, 2012, PMI commenced a three-year $18 billion share repurchase program that was authorized by PMI's Board of Directors in June 2012. From August 1, 2012, through December 31, 2014, PMI repurchased 144.6 million shares of its common stock at a cost of $12.7 billion, or $87.48 per share, under this repurchase program. During 2016 and 2015, PMI did not repurchase any shares of its common stock. During 2014, PMI repurchased $3.8 billion of its common stock under the repurchase program.
At December 31, 2016, 27,213,699 shares of common stock were reserved for stock awards under PMI’s stock plans, and 250 million shares of preferred stock, without par value, were authorized but unissued. PMI currently has no plans to issue any shares of preferred stock.
Note 9. | ||||
Stock Plans:
In May 2012, PMI's shareholders approved the Philip Morris International Inc. 2012 Performance Incentive Plan (the "2012 Plan"). Under the 2012 Plan, PMI may grant to eligible employees restricted stock, restricted stock units and deferred stock units (collectively referred to as restricted share units), performance-based cash incentive awards and performance-based equity awards. Up to 30 million shares of PMI’s common stock may be issued under the 2012 Plan. At December 31, 2016, shares available for grant under the 2012 Plan were 21,180,030.
95
In 2008, PMI adopted the Philip Morris International Inc. 2008 Stock Compensation Plan for Non-Employee Directors (the “Non-Employee Directors Plan”). A non-employee director is defined as a member of the PMI Board of Directors who is not a full-time employee of PMI or of any corporation in which PMI owns, directly or indirectly, stock possessing at least 50% of the total combined voting power of all classes of stock entitled to vote in the election of directors in such corporation. Up to 1 million shares of PMI common stock may be awarded under the Non-Employee Directors Plan. As of December 31, 2016, shares available for grant under the plan were 677,539.
Restricted share unit (RSU) awards
PMI may grant RSU awards to eligible employees; recipients may not sell, assign, pledge or otherwise encumber such awards. Such awards are subject to forfeiture if certain employment conditions are not met. RSU awards generally vest on the third anniversary of the grant date. RSU awards do not carry voting rights, although they do earn dividend equivalents.
During 2016, the activity for RSU awards was as follows:
Number of Shares | Weighted- Average Grant Date Fair Value Per Share | |||||
Balance at January 1, 2016 | 5,702,000 | $ | 82.92 | |||
Granted | 1,212,600 | 89.03 | ||||
Vested | (2,302,525 | ) | 87.81 | |||
Forfeited | (111,085 | ) | 82.38 | |||
Balance at December 31, 2016 | 4,500,990 | $ | 82.08 | |||
During the years ended December 31, 2016, 2015 and 2014, the weighted-average grant date fair value of the RSU awards granted to PMI employees and the recorded compensation expense related to RSU awards were as follows:
(in millions, except per RSU award granted) | Total Weighted-Average Grant Date Fair Value of RSU Awards Granted | Weighted-Average Grant Date Fair Value Per RSU Award Granted | Compensation Expense related to RSU Awards | |||||||
2016 | $ | 108 | $ | 89.03 | $ | 126 | ||||
2015 | $ | 126 | $ | 82.28 | $ | 166 | ||||
2014 | $ | 189 | $ | 77.79 | $ | 210 | ||||
The fair value of the RSU awards at the date of grant is amortized to expense over the restriction period, typically three years after the date of the award, or upon death, disability or reaching the age of 58. As of December 31, 2016, PMI had $105 million of total unrecognized compensation costs related to non-vested RSU awards. These costs are expected to be recognized over a weighted-average period of two years, or upon death, disability or reaching the age of 58.
During the years ended December 31, 2016, 2015 and 2014, share and fair value information for PMI RSU awards that vested were as follows:
(dollars in millions) | Shares of RSU Awards that Vested | Grant Date Fair Value of Vested Shares of RSU Awards | Total Fair Value of RSU Awards that Vested | ||||||
2016 | 2,302,525 | $ | 202 | $ | 210 | ||||
2015 | 2,711,974 | $ | 217 | $ | 224 | ||||
2014 | 3,974,560 | $ | 255 | $ | 320 | ||||
96
Performance share unit (PSU) awards
PMI may grant PSU awards to certain executives; recipients may not sell, assign, pledge or otherwise encumber such awards. The PSU awards require the achievement of certain performance factors, which are predetermined at the time of grant, over a three-year performance cycle. PMI’s performance metrics consist of PMI’s Total Shareholder Return (TSR) relative to a predetermined peer group, PMI’s currency-neutral compound annual adjusted operating companies income growth rate, excluding acquisitions, and PMI’s performance against specific measures of innovation. The aggregate of the weighted performance factors for the three metrics determines the percentage of PSUs that will vest at the end of the three-year performance cycle. Each vested PSU entitles the participant to one share of common stock. An aggregate weighted PSU performance factor of 100 will result in the targeted number of PSUs being vested. The minimum percentage of PSUs that can vest is zero, with a maximum percentage of 200. At the end of the performance cycle, participants are entitled to an amount equivalent to the accumulated dividends paid on common stock during the performance cycle for the number of shares earned. PSU awards do not carry voting rights.
During 2016, the activity for PSU awards was as follows:
Number of Shares | Grant Date Fair Value Subject to TSR Performance Factor Per Share | Grant Date Fair Value Subject to Other Performance Factors Per Share | |||||||
Balance at January 1, 2016 | — | $ | — | $ | — | ||||
Granted | 428,400 | 104.60 | 89.02 | ||||||
Vested | — | — | — | ||||||
Forfeited | (830 | ) | 104.60 | 89.02 | |||||
Balance at December 31, 2016 | 427,570 | $ | 104.60 | $ | 89.02 | ||||
The grant date fair value of the PSU awards granted to employees during the year ended December 31, 2016, was $22 million or $104.60 per PSU for market based awards subject to the TSR performance factor, which was determined by using the Monte Carlo simulation model, and $19 million or $89.02 per PSU for awards subject to the other performance factors, which was determined by using the average of the high and low market price of PMI’s stock at the date of grant. PMI recorded compensation expense related to PSU awards of $27 million for the year ended December 31, 2016. The fair value of the PSU award at the date of grant is amortized to expense over the performance period, which is typically three years after the date of the award, or upon death, disability or reaching the age of 58. As of December 31, 2016, PMI had $25 million of total unrecognized compensation cost related to non-vested PSU awards. This cost is recognized over a weighted-average performance cycle period of two years, or upon death, disability or reaching the age of 58. PMI did not grant any PSU awards during 2015 and 2014.
Note 10. | ||||
Earnings per Share:
Unvested share-based payment awards that contain non-forfeitable rights to dividends or dividend equivalents are participating securities and therefore are included in PMI’s earnings per share calculation pursuant to the two-class method.
Basic and diluted earnings per share (“EPS”) were calculated using the following:
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Net earnings attributable to PMI | $ | 6,967 | $ | 6,873 | $ | 7,493 | |||||
Less distributed and undistributed earnings attributable to share-based payment awards | 19 | 24 | 34 | ||||||||
Net earnings for basic and diluted EPS | $ | 6,948 | $ | 6,849 | $ | 7,459 | |||||
Weighted-average shares for basic and diluted EPS | 1,551 | 1,549 | 1,566 | ||||||||
For the 2016, 2015 and 2014 computations, there were no antidilutive stock options.
97
Note 11. | ||||
Income Taxes:
Earnings before income taxes and provision for income taxes consisted of the following for the years ended December 31, 2016, 2015 and 2014:
(in millions) | 2016 | 2015 | 2014 | ||||||||
Earnings before income taxes | $ | 9,924 | $ | 9,615 | $ | 10,650 | |||||
Provision for income taxes: | |||||||||||
United States federal and state: | |||||||||||
Current | $ | (39 | ) | $ | (56 | ) | $ | (56 | ) | ||
Deferred | 293 | 117 | 162 | ||||||||
Total United States | 254 | 61 | 106 | ||||||||
Outside United States: | |||||||||||
Current | 2,625 | 2,762 | 3,215 | ||||||||
Deferred | (111 | ) | (135 | ) | (224 | ) | |||||
Total outside United States | 2,514 | 2,627 | 2,991 | ||||||||
Total provision for income taxes | $ | 2,768 | $ | 2,688 | $ | 3,097 | |||||
United States income tax is primarily attributable to repatriation costs.
At December 31, 2016, applicable United States federal income taxes and foreign withholding taxes have not been provided on approximately $23 billion of accumulated earnings of foreign subsidiaries that are expected to be permanently reinvested. These earnings have been or will be invested to support the growth of PMI's international business. Further, PMI does not foresee a need to repatriate these earnings to the U.S. since its U.S. cash requirements are supported by distributions from foreign entities of earnings that have not been designated as permanently reinvested and existing credit facilities. Repatriation of earnings from foreign subsidiaries for which PMI has asserted that the earnings are permanently reinvested would result in additional U.S. income and foreign withholding taxes. The determination of the amount of deferred tax related to these earnings is not practicable due to the complexity of the U.S. foreign tax credit regime, as well as differences between earnings determined for book and tax purposes mainly resulting from intercompany transactions, purchase accounting and currency fluctuations.
A reconciliation of the beginning and ending amount of unrecognized tax benefits is as follows:
(in millions) | 2016 | 2015 | 2014 | ||||||||
Balance at January 1, | $ | 88 | $ | 123 | $ | 114 | |||||
Additions based on tax positions related to the current year | 13 | 17 | 20 | ||||||||
Additions for tax positions of previous years | 1 | 6 | 11 | ||||||||
Reductions for tax positions of prior years | (7 | ) | (42 | ) | (3 | ) | |||||
Reductions due to lapse of statute of limitations | (14 | ) | (7 | ) | (8 | ) | |||||
Settlements | (2 | ) | (1 | ) | (3 | ) | |||||
Other | — | (8 | ) | (8 | ) | ||||||
Balance at December 31, | $ | 79 | $ | 88 | $ | 123 | |||||
98
Unrecognized tax benefits and PMI’s liability for contingent income taxes, interest and penalties were as follows:
(in millions) | December 31, 2016 | December 31, 2015 | December 31, 2014 | ||||||||
Unrecognized tax benefits | $ | 79 | $ | 88 | $ | 123 | |||||
Accrued interest and penalties | 15 | 28 | 40 | ||||||||
Tax credits and other indirect benefits | (31 | ) | (40 | ) | (54 | ) | |||||
Liability for tax contingencies | $ | 63 | $ | 76 | $ | 109 | |||||
The amount of unrecognized tax benefits that, if recognized, would impact the effective tax rate was $47 million at December 31, 2016. The remainder, if recognized, would principally affect deferred taxes.
For the years ended December 31, 2016, 2015 and 2014, PMI recognized income (expense) in its consolidated statements of earnings of $13 million, $3 million and $(19) million, respectively, related to interest and penalties.
PMI is regularly examined by tax authorities around the world and is currently under examination in a number of jurisdictions. The U.S. federal statute of limitations remains open for the years 2013 and onward. Foreign and U.S. state jurisdictions have statutes of limitations generally ranging from three to five years. Years still open to examination by foreign tax authorities in major jurisdictions include Germany (2011 onward), Indonesia (2012 onward), Russia (2015 onward) and Switzerland (2016 onward).
It is reasonably possible that within the next 12 months certain tax examinations will close, which could result in a change in unrecognized tax benefits, along with related interest and penalties. An estimate of any possible change cannot be made at this time.
The effective income tax rate on pre-tax earnings differed from the U.S. federal statutory rate for the following reasons for the years ended December 31, 2016, 2015 and 2014:
2016 | 2015 | 2014 | ||||||
U.S. federal statutory rate | 35.0 | % | 35.0 | % | 35.0 | % | ||
Increase (decrease) resulting from: | ||||||||
Foreign rate differences | (12.6 | ) | (12.3 | ) | (11.2 | ) | ||
Dividend repatriation cost | 5.8 | 5.7 | 5.0 | |||||
Other | (0.3 | ) | (0.4 | ) | 0.3 | |||
Effective tax rate | 27.9 | % | 28.0 | % | 29.1 | % | ||
The 2016 effective tax rate decreased 0.1 percentage point to 27.9%. The change in the effective tax rate for 2016, as compared to 2015, was primarily due to earnings mix by taxing jurisdiction and repatriation cost differences.
The 2015 effective tax rate decreased 1.1 percentage points to 28.0%. The effective tax rate for 2015 was unfavorably impacted by changes to repatriation assertions on certain foreign subsidiary historical earnings ($58 million), partially offset by the recognition of tax benefits of $41 million following the conclusion of the IRS examinations of Altria's consolidated tax returns for the years 2007 and 2008 and PMI's consolidated tax returns for the years 2009 through 2011. Prior to March 28, 2008, PMI was a wholly owned subsidiary of Altria. Excluding the effect of these items, the change in the effective tax rate for 2015, as compared to 2014, was primarily due to earnings mix by taxing jurisdiction and repatriation cost differences.
The 2014 effective tax rate decreased 0.2 percentage points to 29.1%. Excluding 2013 special tax items associated with the American Taxpayer Relief Act of 2012 that was enacted in January 2013 ($17 million) and the enactment of tax law changes in Mexico during 2013 ($14 million), the change in the effective tax rate for the year ended December 31, 2014, was primarily due to earnings mix by taxing jurisdiction and repatriation cost differences.
99
The tax effects of temporary differences that gave rise to deferred income tax assets and liabilities consisted of the following:
At December 31, | |||||||
(in millions) | 2016 | 2015 | |||||
Deferred income tax assets: | |||||||
Accrued postretirement and postemployment benefits | $ | 287 | $ | 275 | |||
Accrued pension costs | 256 | 230 | |||||
Inventory | 241 | 174 | |||||
Accrued liabilities | 137 | 153 | |||||
Other | 173 | 164 | |||||
Total deferred income tax assets | 1,094 | 996 | |||||
Deferred income tax liabilities: | |||||||
Trade names | (554 | ) | (593 | ) | |||
Property, plant and equipment | (217 | ) | (218 | ) | |||
Unremitted earnings | (636 | ) | (554 | ) | |||
Foreign exchange | (725 | ) | (532 | ) | |||
Total deferred income tax liabilities | (2,132 | ) | (1,897 | ) | |||
Net deferred income tax liabilities | $ | (1,038 | ) | $ | (901 | ) | |
Note 12. | ||||
Segment Reporting:
PMI’s subsidiaries and affiliates are engaged in the manufacture and sale of cigarettes, other tobacco products and other nicotine-containing products in markets outside of the United States of America. Reportable segments for PMI are organized and managed by geographic region. PMI’s reportable segments are the European Union; Eastern Europe, Middle East & Africa; Asia; and Latin America & Canada. PMI records net revenues and operating companies income to its segments based upon the geographic area in which the customer resides.
PMI’s management evaluates segment performance and allocates resources based on operating companies income, which PMI defines as operating income, excluding general corporate expenses and amortization of intangibles, plus equity (income)/loss in unconsolidated subsidiaries, net. Interest expense, net, and provision for income taxes are centrally managed and, accordingly, such items are not presented by segment since they are excluded from the measure of segment profitability reviewed by management. Information about total assets by segment is not disclosed because such information is not reported to or used by PMI’s chief operating decision maker. Segment goodwill and other intangible assets, net, are disclosed in Note 3. Goodwill and Other Intangible Assets, net. The accounting policies of the segments are the same as those described in Note 2. Summary of Significant Accounting Policies.
Segment data were as follows:
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Net revenues: | |||||||||||
European Union | $ | 27,129 | $ | 26,563 | $ | 30,517 | |||||
Eastern Europe, Middle East & Africa | 18,286 | 18,328 | 20,469 | ||||||||
Asia | 20,531 | 19,469 | 19,255 | ||||||||
Latin America & Canada | 9,007 | 9,548 | 9,865 | ||||||||
Net revenues(1) | $ | 74,953 | $ | 73,908 | $ | 80,106 | |||||
(1) Total net revenues attributable to customers located in Indonesia, PMI’s largest market in terms of net revenues, were $7.7 billion, $7.1 billion and $7.2 billion for the years ended December 31, 2016, 2015 and 2014, respectively. Total net revenues attributable to customers located in Germany were $7.1 billion, $7.2 billion and $8.3 billion for the years ended December 31, 2016, 2015 and 2014, respectively.
100
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Earnings before income taxes: | |||||||||||
Operating companies income: | |||||||||||
European Union | $ | 3,994 | $ | 3,576 | $ | 3,815 | |||||
Eastern Europe, Middle East & Africa | 3,016 | 3,425 | 4,033 | ||||||||
Asia | 3,196 | 2,886 | 3,187 | ||||||||
Latin America & Canada | 938 | 1,085 | 1,030 | ||||||||
Amortization of intangibles | (74 | ) | (82 | ) | (93 | ) | |||||
General corporate expenses | (161 | ) | (162 | ) | (165 | ) | |||||
Less: | |||||||||||
Equity (income)/loss in unconsolidated subsidiaries, net | (94 | ) | (105 | ) | (105 | ) | |||||
Operating income | 10,815 | 10,623 | 11,702 | ||||||||
Interest expense, net | (891 | ) | (1,008 | ) | (1,052 | ) | |||||
Earnings before income taxes | $ | 9,924 | $ | 9,615 | $ | 10,650 | |||||
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Depreciation expense: | |||||||||||
European Union | $ | 247 | $ | 230 | $ | 206 | |||||
Eastern Europe, Middle East & Africa | 150 | 163 | 212 | ||||||||
Asia | 184 | 184 | 278 | ||||||||
Latin America & Canada | 79 | 85 | 90 | ||||||||
660 | 662 | 786 | |||||||||
Other | 9 | 10 | 10 | ||||||||
Total depreciation expense | $ | 669 | $ | 672 | $ | 796 | |||||
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Capital expenditures: | |||||||||||
European Union | $ | 665 | $ | 497 | $ | 537 | |||||
Eastern Europe, Middle East & Africa | 223 | 147 | 216 | ||||||||
Asia | 180 | 185 | 272 | ||||||||
Latin America & Canada | 103 | 130 | 125 | ||||||||
1,171 | 959 | 1,150 | |||||||||
Other | 1 | 1 | 3 | ||||||||
Total capital expenditures | $ | 1,172 | $ | 960 | $ | 1,153 | |||||
101
At December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Long-lived assets: | |||||||||||
European Union | $ | 3,282 | $ | 3,129 | $ | 3,242 | |||||
Eastern Europe, Middle East & Africa | 866 | 743 | 836 | ||||||||
Asia | 1,916 | 1,743 | 1,838 | ||||||||
Latin America & Canada | 765 | 605 | 704 | ||||||||
Total long-lived assets | 6,829 | 6,220 | 6,620 | ||||||||
Other | 750 | 644 | 269 | ||||||||
Total property, plant and equipment, net and Other assets | $ | 7,579 | $ | 6,864 | $ | 6,889 | |||||
Long-lived assets consist of non-current assets other than goodwill; other intangible assets, net; deferred tax assets, investments in unconsolidated subsidiaries, and financial instruments. PMI's largest markets in terms of long-lived assets are Switzerland, Indonesia and Italy. Total long-lived assets located in Switzerland, which is reflected in the European Union segment above, were $0.9 billion, $0.9 billion and $1.0 billion at December 31, 2016, 2015 and 2014, respectively. Total long-lived assets located in Indonesia, which is reflected in the Asia segment above, were $0.8 billion, $0.7 billion and $0.7 billion at December 31, 2016, 2015 and 2014, respectively. Total long-lived assets located in Italy, which is reflected in the European Union segment above, were $0.7 billion, $0.4 billion and $0.3 billion at December 31, 2016, 2015 and 2014, respectively.
Items affecting the comparability of results from operations were as follows:
• | Asset Impairment and Exit Costs - See Note 5. Asset Impairment and Exit Costs for a breakdown of asset impairment and exit costs by segment. |
• | Acquisitions and Other Business Arrangements - For further details, see Note 6. Acquisitions and Other Business Arrangements. |
Note 13. | ||||
Benefit Plans:
Pension coverage for employees of PMI’s subsidiaries is provided, to the extent deemed appropriate, through separate plans, many of which are governed by local statutory requirements. In addition, PMI provides health care and other benefits to substantially all U.S. retired employees and certain non-U.S. retired employees. In general, health care benefits for non-U.S. retired employees are covered through local government plans.
102
Pension and Postretirement Benefit Plans
Obligations and Funded Status
The postretirement health care plans are not funded. The projected benefit obligations, plan assets and funded status of PMI’s pension plans, and the accumulated benefit obligation and net amount accrued for PMI's postretirement health care plans, at December 31, 2016 and 2015, were as follows:
Pension | |||||||||||||||||||||||
U.S. Plans | Non-U.S. Plans | Postretirement | |||||||||||||||||||||
(in millions) | 2016 | 2015 | 2016 | 2015 | 2016 | 2015 | |||||||||||||||||
Benefit obligation at January 1, | $ | 389 | $ | 438 | $ | 7,697 | $ | 7,638 | $ | 211 | $ | 238 | |||||||||||
Service cost | 5 | 5 | 202 | 200 | 3 | 4 | |||||||||||||||||
Interest cost | 17 | 17 | 129 | 139 | 9 | 9 | |||||||||||||||||
Benefits paid | (18 | ) | (51 | ) | (222 | ) | (225 | ) | (10 | ) | (11 | ) | |||||||||||
Settlement and curtailment | — | — | (1 | ) | (16 | ) | — | — | |||||||||||||||
Actuarial losses (gains) | 12 | (20 | ) | 415 | 261 | 15 | (12 | ) | |||||||||||||||
Currency | — | — | (329 | ) | (365 | ) | (2 | ) | (17 | ) | |||||||||||||
Other | — | — | 91 | 65 | 1 | — | |||||||||||||||||
Benefit obligation at December 31, | 405 | 389 | 7,982 | 7,697 | 227 | 211 | |||||||||||||||||
Fair value of plan assets at January 1, | 298 | 312 | 6,106 | 6,410 | |||||||||||||||||||
Actual return on plan assets | 13 | — | 309 | 56 | |||||||||||||||||||
Employer contributions | 4 | 37 | 187 | 117 | |||||||||||||||||||
Employee contributions | — | — | 39 | 37 | |||||||||||||||||||
Benefits paid | (18 | ) | (51 | ) | (222 | ) | (225 | ) | |||||||||||||||
Settlement and curtailment | — | — | — | (14 | ) | ||||||||||||||||||
Currency | — | — | (259 | ) | (275 | ) | |||||||||||||||||
Fair value of plan assets at December 31, | 297 | 298 | 6,160 | 6,106 | |||||||||||||||||||
Net pension and postretirement liability recognized at December 31, | $ | (108 | ) | $ | (91 | ) | $ | (1,822 | ) | $ | (1,591 | ) | $ | (227 | ) | $ | (211 | ) | |||||
At December 31, 2016 and 2015, the Swiss pension plan represented 60% and 61% of the non-U.S. benefit obligation, respectively, and approximately 60% of the non-U.S. fair value of plan assets for each of the years.
At December 31, 2016 and 2015, the amounts recognized on PMI's consolidated balance sheets for the combined U.S. and non-U.S. pension plans, and postretirement plans were as follows:
Pension | Postretirement | ||||||||||||||
(in millions) | 2016 | 2015 | 2016 | 2015 | |||||||||||
Other assets | $ | 33 | $ | 47 | |||||||||||
Accrued liabilities — employment costs | (23 | ) | (23 | ) | $ | (10 | ) | $ | (9 | ) | |||||
Long-term employment costs | (1,940 | ) | (1,706 | ) | (217 | ) | (202 | ) | |||||||
$ | (1,930 | ) | $ | (1,682 | ) | $ | (227 | ) | $ | (211 | ) | ||||
The accumulated benefit obligation, which represents benefits earned to date, for the U.S. pension plans was $376 million and $360 million at December 31, 2016 and 2015, respectively. The accumulated benefit obligation for non-U.S. pension plans was $7,555 million and $7,157 million at December 31, 2016 and 2015, respectively.
For U.S. pension plans with accumulated benefit obligations in excess of plan assets, the projected benefit obligation, accumulated benefit obligation and fair value of plan assets were $405 million, $376 million and $297 million, respectively, as of December 31, 2016. The projected benefit obligation, accumulated benefit obligation and fair value of plan assets were $389 million, $360 million and $298
103
million, respectively, as of December 31, 2015. The underfunding relates to plans for salaried employees that cannot be funded under IRS regulations. For non-U.S. plans with accumulated benefit obligations in excess of plan assets, the projected benefit obligation, accumulated benefit obligation and fair value of plan assets were $6,529 million, $6,246 million, and $4,712 million, respectively, as of December 31, 2016, and $6,355 million, $5,961 million, and $4,766 million, respectively, as of December 31, 2015.
The following weighted-average assumptions were used to determine PMI’s pension and postretirement benefit obligations at December 31:
Pension | |||||||||||||||||
U.S. Plans | Non-U.S. Plans | Postretirement | |||||||||||||||
2016 | 2015 | 2016 | 2015 | 2016 | 2015 | ||||||||||||
Discount rate | 4.07 | % | 4.30 | % | 1.39 | % | 1.68 | % | 3.68 | % | 4.45 | % | |||||
Rate of compensation increase | 3.00 | 3.00 | 1.61 | 1.98 | |||||||||||||
Health care cost trend rate assumed for next year | 7.15 | 6.23 | |||||||||||||||
Ultimate trend rate | 5.08 | 4.75 | |||||||||||||||
Year that rate reaches the ultimate trend rate | 2029 | 2029 | |||||||||||||||
The discount rate for the largest U.S. and non-U.S. pension plans is based on a yield curve constructed from a portfolio of high quality corporate bonds that produces a cash flow pattern equivalent to each plan’s expected benefit payments. The discount rate for the remaining non-U.S. plans is developed from local bond indices that match local benefit obligations as closely as possible.
Components of Net Periodic Benefit Cost
Net periodic pension and postretirement health care costs consisted of the following for the years ended December 31, 2016, 2015 and 2014:
Pension | |||||||||||||||||||||||||||||||||||
U.S. Plans | Non-U.S. Plans | Postretirement | |||||||||||||||||||||||||||||||||
(in millions) | 2016 | 2015 | 2014 | 2016 | 2015 | 2014 | 2016 | 2015 | 2014 | ||||||||||||||||||||||||||
Service cost | $ | 5 | $ | 5 | $ | 5 | $ | 202 | $ | 200 | $ | 211 | $ | 3 | $ | 4 | $ | 4 | |||||||||||||||||
Interest cost | 17 | 17 | 17 | 129 | 139 | 205 | 9 | 9 | 10 | ||||||||||||||||||||||||||
Expected return on plan assets | (14 | ) | (15 | ) | (16 | ) | (332 | ) | (325 | ) | (357 | ) | — | — | — | ||||||||||||||||||||
Amortization: | |||||||||||||||||||||||||||||||||||
Net losses | 9 | 14 | 6 | 177 | 180 | 115 | 2 | 4 | 2 | ||||||||||||||||||||||||||
Prior service cost | — | — | 1 | 4 | 4 | 5 | — | — | (1 | ) | |||||||||||||||||||||||||
Settlement and curtailment | — | 1 | 5 | 4 | 2 | 1 | — | — | — | ||||||||||||||||||||||||||
Net periodic pension and postretirement costs | $ | 17 | $ | 22 | $ | 18 | $ | 184 | $ | 200 | $ | 180 | $ | 14 | $ | 17 | $ | 15 | |||||||||||||||||
As of December 31, 2016, PMI elected to change the method used to calculate the service and interest cost components of the net periodic pension benefit costs. Historically, these costs were determined utilizing a single weighted-average discount rate based on a yield curve used to measure the benefit obligation at the beginning of the period. As of January 1, 2017, PMI will utilize a full yield curve approach in the estimation of the service and interest costs by applying the specific spot rates along the yield curve to the relevant projected cash flows. Specifically, service costs will be determined based on duration-specific spot rates applied to service cost cash flows, and interest costs will be determined by applying duration specific spot rates to the year-by-year projected benefit payments. PMI has changed to the new method to provide a more precise measurement of service and interest costs by improving the correlation between the projected benefit cash flows to the corresponding spot rates along the yield curve. PMI will account for this change as a change in accounting estimate on a prospective basis. This change does not affect the measurement of PMI’s pension plan obligations and will not have a material impact on PMI’s consolidated results of operations, financial position or cash flows.
Settlement and curtailment charges were due primarily to early retirement programs.
104
For the combined U.S. and non-U.S. pension plans, the estimated net loss and prior service cost that are expected to be amortized from accumulated other comprehensive earnings into net periodic benefit cost during 2017 are $177 million and $13 million, respectively.
The following weighted-average assumptions were used to determine PMI’s net pension and postretirement health care costs:
Pension | ||||||||||||||||||||||||||
U.S. Plans | Non-U.S. Plans | Postretirement | ||||||||||||||||||||||||
2016 | 2015 | 2014 | 2016 | 2015 | 2014 | 2016 | 2015 | 2014 | ||||||||||||||||||
Discount rate | 4.30 | % | 3.95 | % | 4.80 | % | 1.68 | % | 1.92 | % | 3.09 | % | 4.45 | % | 4.20 | % | 5.01 | % | ||||||||
Expected rate of return on plan assets | 4.60 | 5.10 | 5.70 | 5.39 | 5.38 | 5.63 | ||||||||||||||||||||
Rate of compensation increase | 3.00 | 3.00 | 3.00 | 1.98 | 2.06 | 2.34 | ||||||||||||||||||||
Health care cost trend rate | 6.23 | 6.62 | 6.60 | |||||||||||||||||||||||
PMI’s expected rate of return on pension plan assets is determined by the plan assets’ historical long-term investment performance, current asset allocation and estimates of future long-term returns by asset class.
PMI and certain of its subsidiaries sponsor defined contribution plans. Amounts charged to expense for defined contribution plans totaled $56 million, $52 million and $62 million for the years ended December 31, 2016, 2015 and 2014, respectively.
Plan Assets
PMI’s investment strategy for U.S. and non-U.S. pension plans is based on an expectation that equity securities will outperform debt securities over the long term. Accordingly, the target allocation of PMI’s plan assets is broadly characterized as approximately a 60%/40% split between equity and debt securities. The strategy primarily utilizes indexed U.S. equity securities, international equity securities and investment-grade debt securities. PMI’s plans have no investments in hedge funds, private equity or derivatives. PMI attempts to mitigate investment risk by rebalancing between equity and debt asset classes once a year or as PMI’s contributions and benefit payments are made.
The fair value of PMI’s pension plan assets at December 31, 2016 and 2015, by asset category was as follows:
Asset Category (in millions) | At December 31, 2016 | Quoted Prices In Active Markets for Identical Assets/Liabilities (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | |||||||||||
Cash and cash equivalents | $ | 8 | $ | 8 | |||||||||||
Equity securities: | |||||||||||||||
U.S. securities | 131 | 131 | |||||||||||||
International securities | 432 | 432 | |||||||||||||
Investment funds(a) | 5,270 | 3,530 | $ | 1,740 | |||||||||||
International government bonds | 309 | 309 | |||||||||||||
Other | 10 | 10 | |||||||||||||
Total assets in the fair value hierarchy | $ | 6,160 | $ | 4,420 | $ | 1,740 | $ | — | |||||||
Investment funds measured at net asset value(b) | 297 | ||||||||||||||
Total assets | $ | 6,457 | |||||||||||||
(a) Investment funds whose objective seeks to replicate the returns and characteristics of specified market indices (primarily MSCI — Europe, Switzerland, North America, Asia Pacific, Japan; Russell 3000; S&P 500 for equities, and Citigroup EMU and Barclays Capital U.S. for bonds), primarily consist of mutual funds, common trust funds and commingled funds. Of these funds, 60% are invested in U.S. and international equities; 19% are invested in U.S. and international government bonds; 11% are invested in real estate and other money markets, and 10% are invested in corporate bonds.
105
(b) In accordance with FASB ASC Subtopic 820-10, certain investments measured at fair value using the net asset value per share practical expedient have not been classified in the fair value hierarchy. The fair value amounts presented in this table are intended to permit reconciliation of the fair value hierarchy to the amounts presented in the statement of financial position.
Asset Category (in millions) | At December 31, 2015 | Quoted Prices In Active Markets for Identical Assets/Liabilities (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | |||||||||||
Cash and cash equivalents | $ | 225 | $ | 225 | |||||||||||
Equity securities: | |||||||||||||||
U.S. securities | 120 | 120 | |||||||||||||
International securities | 409 | 409 | |||||||||||||
Investment funds(a) | 5,039 | 3,446 | 1,593 | ||||||||||||
International government bonds | 289 | 289 | |||||||||||||
Other | 24 | 24 | |||||||||||||
Total assets in the fair value hierarchy | $ | 6,106 | $ | 4,513 | $ | 1,593 | $ | — | |||||||
Investment funds measured at net asset value(b) | 298 | ||||||||||||||
Total assets | $ | 6,404 | |||||||||||||
(a) Investment funds whose objective seeks to replicate the returns and characteristics of specified market indices (primarily MSCI — Europe, Switzerland, North America, Asia Pacific, Japan; Russell 3000; S&P 500 for equities, and Citigroup EMU and Barclays Capital U.S. for bonds), primarily consist of mutual funds, common trust funds and commingled funds. Of these funds, 61% were invested in U.S. and international equities; 18% were invested in U.S. and international government bonds; 11% were invested in real estate and other money markets, and 10% were invested in corporate bonds.
(b) In accordance with FASB ASC Subtopic 820-10, certain investments measured at fair value using the net asset value per share practical expedient have not been classified in the fair value hierarchy. The fair value amounts presented in this table are intended to permit reconciliation of the fair value hierarchy to the amounts presented in the statement of financial position.
See Note 16. Fair Value Measurements for a discussion of the fair value of pension plan assets.
PMI makes, and plans to make, contributions to the extent that they are tax deductible and to meet specific funding requirements of its funded U.S. and non-U.S. pension plans. Currently, PMI anticipates making contributions of approximately $70 million in 2017 to its pension plans, based on current tax and benefit laws. However, this estimate is subject to change as a result of changes in tax and other benefit laws, as well as asset performance significantly above or below the assumed long-term rate of return on pension assets, or changes in interest and currency rates.
The estimated future benefit payments from PMI pension plans at December 31, 2016, are as follows:
(in millions) | U.S. Plans | Non-U.S. Plans | |||||
2017 | $ | 18 | $ | 247 | |||
2018 | 18 | 272 | |||||
2019 | 24 | 265 | |||||
2020 | 22 | 280 | |||||
2021 | 21 | 300 | |||||
2022 - 2026 | 124 | 1,675 | |||||
PMI's expected future annual benefit payments for its postretirement health care plans are estimated to be not material through 2026.
106
Assumed health care cost trend rates have a significant effect on the amounts reported for the health care plans. A one-percentage-point change in assumed health care trend rates would have the following effects as of December 31, 2016:
One-Percentage-Point Increase | One-Percentage-Point Decrease | ||||
Effect on total service and interest cost | 23.9 | % | (18.1 | )% | |
Effect on postretirement benefit obligation | 19.4 | (15.3 | ) | ||
Postemployment Benefit Plans
PMI and certain of its subsidiaries sponsor postemployment benefit plans covering substantially all salaried and certain hourly employees. The cost of these plans is charged to expense over the working life of the covered employees. Net postemployment costs were $166 million, $187 million and $167 million for the years ended December 31, 2016, 2015 and 2014, respectively.
The estimated net loss for the postemployment benefit plans that will be amortized from accumulated other comprehensive losses into net postemployment costs during 2017 is approximately $67 million.
The amounts recognized in accrued postemployment costs on PMI's consolidated balance sheets at December 31, 2016 and 2015, were $727 million and $745 million, respectively.
During 2016, 2015 and 2014, certain salaried employees left PMI under separation programs. These programs resulted in incremental postemployment costs and benefit obligations. For further details see Note 5. Asset Impairment and Exit Costs.
The accrued postemployment costs were determined using a weighted-average discount rate of 2.8% and 3.5% in 2016 and 2015, respectively; an assumed ultimate annual weighted-average turnover rate of 2.8% and 2.7% in 2016 and 2015, respectively; assumed compensation cost increases of 2.6% in 2016 and 2.2% in 2015, and assumed benefits as defined in the respective plans. In accordance with local regulations, certain postemployment plans are funded. As a result, the accrued postemployment costs disclosed above are presented net of the related assets of $25 million and $26 million at December 31, 2016 and 2015, respectively. Postemployment costs arising from actions that offer employees benefits in excess of those specified in the respective plans are charged to expense when incurred.
Comprehensive Earnings (Losses)
The amounts recorded in accumulated other comprehensive losses at December 31, 2016, consisted of the following:
(in millions) | Pension | Post- retirement | Post- employment | Total | |||||||||||
Net losses | $ | (3,314 | ) | $ | (73 | ) | $ | (713 | ) | $ | (4,100 | ) | |||
Prior service cost | (53 | ) | 4 | — | (49 | ) | |||||||||
Net transition obligation | (5 | ) | — | — | (5 | ) | |||||||||
Deferred income taxes | 350 | 24 | 215 | 589 | |||||||||||
Losses to be amortized | $ | (3,022 | ) | $ | (45 | ) | $ | (498 | ) | $ | (3,565 | ) | |||
107
The amounts recorded in accumulated other comprehensive losses at December 31, 2015, consisted of the following:
(in millions) | Pension | Post- retirement | Post- employment | Total | |||||||||||
Net losses | $ | (3,074 | ) | $ | (61 | ) | $ | (710 | ) | $ | (3,845 | ) | |||
Prior service cost | (40 | ) | 5 | — | (35 | ) | |||||||||
Net transition obligation | (5 | ) | — | — | (5 | ) | |||||||||
Deferred income taxes | 320 | 20 | 213 | 553 | |||||||||||
Losses to be amortized | $ | (2,799 | ) | $ | (36 | ) | $ | (497 | ) | $ | (3,332 | ) | |||
The amounts recorded in accumulated other comprehensive losses at December 31, 2014, consisted of the following:
(in millions) | Pension | Post- retirement | Post- employment | Total | |||||||||||
Net losses | $ | (2,760 | ) | $ | (77 | ) | $ | (721 | ) | $ | (3,558 | ) | |||
Prior service cost | (45 | ) | 6 | — | (39 | ) | |||||||||
Net transition obligation | (6 | ) | — | — | (6 | ) | |||||||||
Deferred income taxes | 342 | 25 | 216 | 583 | |||||||||||
Losses to be amortized | $ | (2,469 | ) | $ | (46 | ) | $ | (505 | ) | $ | (3,020 | ) | |||
The movements in other comprehensive earnings (losses) during the year ended December 31, 2016, were as follows:
(in millions) | Pension | Post- retirement | Post- employment | Total | |||||||||||
Amounts transferred to earnings as components of net periodic benefit cost: | |||||||||||||||
Amortization: | |||||||||||||||
Net losses | $ | 193 | $ | 2 | $ | 62 | $ | 257 | |||||||
Prior service cost | 6 | — | — | 6 | |||||||||||
Other income/expense: | |||||||||||||||
Net losses | 4 | — | — | 4 | |||||||||||
Prior service cost | — | — | — | — | |||||||||||
Deferred income taxes | (26 | ) | — | (17 | ) | (43 | ) | ||||||||
177 | 2 | 45 | 224 | ||||||||||||
Other movements during the year: | |||||||||||||||
Net losses | (437 | ) | (15 | ) | (65 | ) | (517 | ) | |||||||
Prior service cost | (18 | ) | — | — | (18 | ) | |||||||||
Deferred income taxes | 55 | 4 | 19 | 78 | |||||||||||
(400 | ) | (11 | ) | (46 | ) | (457 | ) | ||||||||
Total movements in other comprehensive earnings (losses) | $ | (223 | ) | $ | (9 | ) | $ | (1 | ) | $ | (233 | ) | |||
108
The movements in other comprehensive earnings (losses) during the year ended December 31, 2015, were as follows:
(in millions) | Pension | Post- retirement | Post- employment | Total | |||||||||||
Amounts transferred to earnings as components of net periodic benefit cost: | |||||||||||||||
Amortization: | |||||||||||||||
Net losses | $ | 194 | $ | 4 | $ | 69 | $ | 267 | |||||||
Prior service cost | 4 | — | — | 4 | |||||||||||
Other income/expense: | |||||||||||||||
Net losses | 3 | — | — | 3 | |||||||||||
Prior service cost | 1 | — | — | 1 | |||||||||||
Deferred income taxes | (26 | ) | (2 | ) | (20 | ) | (48 | ) | |||||||
176 | 2 | 49 | 227 | ||||||||||||
Other movements during the year: | |||||||||||||||
Net losses | (510 | ) | 12 | (58 | ) | (556 | ) | ||||||||
Deferred income taxes | 4 | (4 | ) | 17 | 17 | ||||||||||
(506 | ) | 8 | (41 | ) | (539 | ) | |||||||||
Total movements in other comprehensive earnings (losses) | $ | (330 | ) | $ | 10 | $ | 8 | $ | (312 | ) | |||||
The movements in other comprehensive earnings (losses) during the year ended December 31, 2014, were as follows:
(in millions) | Pension | Post- retirement | Post- employment | Total | |||||||||||
Amounts transferred to earnings as components of net periodic benefit cost: | |||||||||||||||
Amortization: | |||||||||||||||
Net losses | $ | 121 | $ | 2 | $ | 66 | $ | 189 | |||||||
Prior service cost | 6 | (1 | ) | — | 5 | ||||||||||
Other income/expense: | |||||||||||||||
Net losses | 14 | 2 | — | 16 | |||||||||||
Prior service cost | 5 | — | — | 5 | |||||||||||
Deferred income taxes | (21 | ) | (1 | ) | (20 | ) | (42 | ) | |||||||
125 | 2 | 46 | 173 | ||||||||||||
Other movements during the year: | |||||||||||||||
Net losses | (1,149 | ) | (34 | ) | (126 | ) | (1,309 | ) | |||||||
Prior service cost | (5 | ) | — | — | (5 | ) | |||||||||
Deferred income taxes | 118 | 12 | 37 | 167 | |||||||||||
(1,036 | ) | (22 | ) | (89 | ) | (1,147 | ) | ||||||||
Total movements in other comprehensive earnings (losses) | $ | (911 | ) | $ | (20 | ) | $ | (43 | ) | $ | (974 | ) | |||
109
Note 14. | ||||
Additional Information:
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Research and development expense | $ | 429 | $ | 423 | $ | 433 | |||||
Advertising expense | $ | 405 | $ | 448 | $ | 439 | |||||
Foreign currency net transaction losses | $ | 272 | $ | 102 | $ | 174 | |||||
Interest expense | $ | 1,069 | $ | 1,132 | $ | 1,170 | |||||
Interest income | (178 | ) | (124 | ) | (118 | ) | |||||
Interest expense, net | $ | 891 | $ | 1,008 | $ | 1,052 | |||||
Rent expense | $ | 284 | $ | 286 | $ | 336 | |||||
Minimum rental commitments under non-cancelable operating leases in effect at December 31, 2016, were as follows:
(in millions) | |||
2017 | $ | 182 | |
2018 | 119 | ||
2019 | 89 | ||
2020 | 62 | ||
2021 | 46 | ||
Thereafter | 164 | ||
$ | 662 | ||
Note 15. | ||||
Financial Instruments:
Overview
PMI operates in markets outside of the United States of America, with manufacturing and sales facilities in various locations around the world. PMI utilizes certain financial instruments to manage foreign currency and interest rate exposure. Derivative financial instruments are used by PMI principally to reduce exposures to market risks resulting from fluctuations in foreign currency exchange and interest rates by creating offsetting exposures. PMI is not a party to leveraged derivatives and, by policy, does not use derivative financial instruments for speculative purposes. Financial instruments qualifying for hedge accounting must maintain a specified level of effectiveness between the hedging instrument and the item being hedged, both at inception and throughout the hedged period. PMI formally documents the nature and relationships between the hedging instruments and hedged items, as well as its risk-management objectives, strategies for undertaking the various hedge transactions and method of assessing hedge effectiveness. Additionally, for hedges of forecasted transactions, the significant characteristics and expected terms of the forecasted transaction must be specifically identified, and it must be probable that each forecasted transaction will occur. If it were deemed probable that the forecasted transaction would not occur, the gain or loss would be recognized in earnings. PMI reports its net transaction gains or losses in marketing, administration and research costs on the consolidated statements of earnings.
PMI uses deliverable and non-deliverable forward foreign exchange contracts, foreign currency swaps and foreign currency options, collectively referred to as foreign exchange contracts ("foreign exchange contracts"), and interest rate contracts to mitigate its exposure to changes in exchange and interest rates from third-party and intercompany actual and forecasted transactions. The primary currencies
110
to which PMI is exposed include the Australian dollar, Euro, Indonesian rupiah, Japanese yen, Mexican peso, Russian ruble, Swiss franc and Turkish lira. At December 31, 2016 and 2015, PMI had contracts with aggregate notional amounts of $29.2 billion and $24.9 billion, respectively. Of the $29.2 billion aggregate notional amount at December 31, 2016, $5.0 billion related to cash flow hedges, $10.6 billion related to hedges of net investments in foreign operations and $13.6 billion related to other derivatives that primarily offset currency exposures on intercompany financing. Of the $24.9 billion aggregate notional amount at December 31, 2015, $3.2 billion related to cash flow hedges, $6.4 billion related to hedges of net investments in foreign operations and $15.3 billion related to other derivatives that primarily offset currency exposures on intercompany financing.
The fair value of PMI’s foreign exchange contracts included in the consolidated balance sheet as of December 31, 2016 and 2015, were as follows:
Asset Derivatives | Liability Derivatives | ||||||||||||||||||
Fair Value | Fair Value | ||||||||||||||||||
(in millions) | Balance Sheet Classification | 2016 | 2015 | Balance Sheet Classification | 2016 | 2015 | |||||||||||||
Foreign exchange contracts designated as hedging instruments | Other current assets | $ | 207 | $ | 301 | Other accrued liabilities | $ | 66 | $ | 26 | |||||||||
Other assets | 436 | 181 | Other liabilities | 36 | 117 | ||||||||||||||
Foreign exchange contracts not designated as hedging instruments | Other current assets | 161 | 7 | Other accrued liabilities | 61 | 29 | |||||||||||||
Other assets | 9 | 85 | Other liabilities | — | — | ||||||||||||||
Total derivatives | $ | 813 | $ | 574 | $ | 163 | $ | 172 | |||||||||||
For the years ended December 31, 2016, 2015 and 2014, PMI's cash flow and net investment hedging instruments impacted the consolidated statements of earnings and comprehensive earnings as follows:
(pre-tax, millions) | For the Year Ended December 31, | ||||||||||||||||||||||||
Amount of Gain/(Loss) Recognized in Other Comprehensive Earnings/(Losses) on Derivatives | Statement of Earnings Classification of Gain/(Loss) Reclassified from Other Comprehensive Earnings/(Losses) into Earnings | Amount of Gain/(Loss) Reclassified from Other Comprehensive Earnings/(Losses) into Earnings | |||||||||||||||||||||||
2016 | 2015 | 2014 | 2016 | 2015 | 2014 | ||||||||||||||||||||
Derivatives in Cash Flow Hedging Relationship | |||||||||||||||||||||||||
Foreign exchange contracts | $ | 12 | $ | 43 | $ | 111 | |||||||||||||||||||
Net revenues | $ | (38 | ) | $ | 149 | $ | 115 | ||||||||||||||||||
Cost of sales | 46 | (3 | ) | — | |||||||||||||||||||||
Marketing, administration and research costs | (11 | ) | 1 | (28 | ) | ||||||||||||||||||||
Interest expense, net | (30 | ) | (31 | ) | (39 | ) | |||||||||||||||||||
Derivatives in Net Investment Hedging Relationship | |||||||||||||||||||||||||
Foreign exchange contracts | 296 | 253 | 269 | ||||||||||||||||||||||
Total | $ | 308 | $ | 296 | $ | 380 | $ | (33 | ) | $ | 116 | $ | 48 | ||||||||||||
Cash Flow Hedges
PMI has entered into foreign exchange contracts to hedge the foreign currency exchange and interest rate risks related to certain forecasted transactions. The effective portion of gains and losses associated with qualifying cash flow hedge contracts is deferred as a component of accumulated other comprehensive losses until the underlying hedged transactions are reported in PMI’s consolidated statements of earnings. During the years ended December 31, 2016, 2015 and 2014, ineffectiveness related to cash flow hedges was not material. As
111
of December 31, 2016, PMI has hedged forecasted transactions for periods not exceeding the next eighteen months, with the exception of one foreign exchange contract that expires in May 2024. The impact of these hedges is primarily included in operating cash flows on PMI’s consolidated statements of cash flows.
Hedges of Net Investments in Foreign Operations
PMI designates certain foreign currency denominated debt and foreign exchange contracts as net investment hedges of its foreign operations. For the years ended December 31, 2016, 2015 and 2014, these hedges of net investments resulted in gains, net of income taxes, of $430 million, $761 million and $952 million, respectively. These gains were reported as a component of accumulated other comprehensive losses within currency translation adjustments. For the years ended December 31, 2016, 2015 and 2014, ineffectiveness related to net investment hedges was not material. Other investing cash flows on PMI’s consolidated statements of cash flows include the premiums paid for, and settlements of, net investment hedges.
Other Derivatives
PMI has entered into foreign exchange contracts to hedge the foreign currency exchange and interest rate risks related to intercompany loans between certain subsidiaries, and third-party loans. While effective as economic hedges, no hedge accounting is applied for these contracts; therefore, the unrealized gains (losses) relating to these contracts are reported in PMI’s consolidated statements of earnings. For the years ended December 31, 2016, 2015 and 2014, the losses from contracts for which PMI did not apply hedge accounting were $85 million, $587 million and $481 million, respectively. The losses from these contracts substantially offset the gains generated by the underlying intercompany and third-party loans being hedged.
For the years ended December 31, 2016, 2015 and 2014, the net impact of these contracts on the consolidated statements of earnings was not material.
Qualifying Hedging Activities Reported in Accumulated Other Comprehensive Losses
Derivative gains or losses reported in accumulated other comprehensive losses are a result of qualifying hedging activity. Transfers of these gains or losses to earnings are offset by the corresponding gains or losses on the underlying hedged item. Hedging activity affected accumulated other comprehensive losses, net of income taxes, as follows:
For the Years Ended December 31, | |||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Gain as of January 1, | $ | 59 | $ | 123 | $ | 63 | |||||
Derivative (gains)/losses transferred to earnings | 30 | (102 | ) | (38 | ) | ||||||
Change in fair value | 8 | 38 | 98 | ||||||||
Gain as of December 31, | $ | 97 | $ | 59 | $ | 123 | |||||
At December 31, 2016, PMI expects $60 million of derivative gains that are included in accumulated other comprehensive losses to be reclassified to the consolidated statement of earnings within the next 12 months. These gains are expected to be substantially offset by the statement of earnings impact of the respective hedged transactions.
Contingent Features
PMI’s derivative instruments do not contain contingent features.
Credit Exposure and Credit Risk
PMI is exposed to credit loss in the event of non-performance by counterparties. While PMI does not anticipate non-performance, its risk is limited to the fair value of the financial instruments less any cash collateral received or pledged. PMI actively monitors its exposure to credit risk through the use of credit approvals and credit limits and by selecting and continuously monitoring a diverse group of major international banks and financial institutions as counterparties.
112
Fair Value
See Note 16. Fair Value Measurements and Note 20. Balance Sheet Offsetting for additional discussion of derivative financial instruments.
Note 16. | ||||
Fair Value Measurements:
The authoritative guidance defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. The guidance also establishes a fair value hierarchy, which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The guidance describes three levels of input that may be used to measure fair value, which are as follows:
Level 1 | — | Quoted prices in active markets for identical assets or liabilities; |
Level 2 | — | Observable inputs other than Level 1 prices, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities; and |
Level 3 | — | Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. |
PMI's policy is to reflect transfers between hierarchy levels at the end of the reporting period.
Derivative Financial Instruments
PMI assesses the fair value of its foreign exchange contracts and interest rate contracts using standard valuation models that use, as their basis, readily observable market inputs. The fair value of PMI’s foreign exchange forward contracts is determined by using the prevailing foreign exchange spot rates and interest rate differentials and the respective maturity dates of the instruments. The fair value of PMI’s currency options is determined by using a Black-Scholes methodology based on foreign exchange spot rates and interest rate differentials, currency volatilities and maturity dates. PMI’s derivative financial instruments have been classified within Level 2 at December 31, 2016 and 2015. See Note 15. Financial Instruments for additional discussion of derivative financial instruments.
Pension Plan Assets
The fair value of pension plan assets determined by using readily available quoted market prices in active markets has been classified within Level 1 of the fair value hierarchy at December 31, 2016 and 2015. The fair value of pension plan assets determined by using quoted prices in markets that are not active has been classified within Level 2 at December 31, 2016 and 2015. See Note 13. Benefit Plans for additional discussion of pension plan assets.
Debt
The fair value of PMI’s outstanding debt, which is utilized solely for disclosure purposes, is determined using quotes and market interest rates currently available to PMI for issuances of debt with similar terms and remaining maturities. The aggregate carrying value of PMI’s debt, excluding short-term borrowings and $13 million of capital lease obligations, was $28,411 million at December 31, 2016. The aggregate carrying value of PMI’s debt, excluding short-term borrowings and $13 million of capital lease obligations, was $27,642 million at December 31, 2015. The fair value of PMI's outstanding debt, excluding the aforementioned short-term borrowings and capital lease obligations, was classified within Level 1 and Level 2 at December 31, 2016 and 2015.
113
The aggregate fair values of PMI’s derivative financial instruments, pension plan assets and debt as of December 31, 2016 and 2015, were as follows:
(in millions) | Fair Value At December 31, 2016 | Quoted Prices in Active Markets for Identical Assets/Liabilities (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | |||||||||||
Assets: | |||||||||||||||
Foreign exchange contracts | $ | 813 | $ | — | $ | 813 | $ | — | |||||||
Pension plan assets | 6,160 | 4,420 | 1,740 | — | |||||||||||
Total assets in fair value hierarchy | $ | 6,973 | $ | 4,420 | $ | 2,553 | $ | — | |||||||
Pension plan assets measured at net asset value(a) | 297 | ||||||||||||||
Total assets | $ | 7,270 | |||||||||||||
Liabilities: | |||||||||||||||
Debt | $ | 30,192 | $ | 29,756 | $ | 436 | $ | — | |||||||
Foreign exchange contracts | 163 | — | 163 | — | |||||||||||
Total liabilities | $ | 30,355 | $ | 29,756 | $ | 599 | $ | — | |||||||
(in millions) | Fair Value At December 31, 2015 | Quoted Prices in Active Markets for Identical Assets/Liabilities (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | |||||||||||
Assets: | |||||||||||||||
Foreign exchange contracts | $ | 574 | $ | — | $ | 574 | $ | — | |||||||
Pension plan assets | 6,106 | 4,513 | 1,593 | — | |||||||||||
Total assets in fair value hierarchy | $ | 6,680 | $ | 4,513 | $ | 2,167 | $ | — | |||||||
Pension plan assets measured at net asset value(a) | 298 | ||||||||||||||
Total assets | $ | 6,978 | |||||||||||||
Liabilities: | |||||||||||||||
Debt | $ | 29,287 | $ | 28,822 | $ | 465 | $ | — | |||||||
Foreign exchange contracts | 172 | — | 172 | — | |||||||||||
Total liabilities | $ | 29,459 | $ | 28,822 | $ | 637 | $ | — | |||||||
(a) In accordance with FASB ASC Subtopic 820-10, certain investments measured at fair value using the net asset value per share practical expedient have not been classified in the fair value hierarchy. The fair value amounts presented in these tables are intended to permit reconciliation of the fair value hierarchy to the amounts presented in the statement of financial position.
114
Note 17. | ||||
Accumulated Other Comprehensive Losses:
PMI's accumulated other comprehensive losses, net of taxes, consisted of the following:
(Losses) Earnings | At December 31, | ||||||||||
(in millions) | 2016 | 2015 | 2014 | ||||||||
Currency translation adjustments | $ | (6,091 | ) | $ | (6,129 | ) | $ | (3,929 | ) | ||
Pension and other benefits | (3,565 | ) | (3,332 | ) | (3,020 | ) | |||||
Derivatives accounted for as hedges | 97 | 59 | 123 | ||||||||
Total accumulated other comprehensive losses | $ | (9,559 | ) | $ | (9,402 | ) | $ | (6,826 | ) | ||
Reclassifications from Other Comprehensive Earnings
The movements in accumulated other comprehensive losses and the related tax impact, for each of the components above, that are due to current period activity and reclassifications to the income statement are shown on the consolidated statements of comprehensive earnings for the years ended December 31, 2016, 2015, and 2014. For the years ended December 31, 2016, 2015, and 2014, $(5) million, $1 million and $5 million of net currency translation adjustment gains/(losses) were transferred from other comprehensive earnings to marketing, administration and research costs in the consolidated statements of earnings, respectively, upon liquidation of subsidiaries. For additional information, see Note 13. Benefit Plans and Note 15. Financial Instruments for disclosures related to PMI's pension and other benefits and derivative financial instruments.
Note 18. | ||||
E.C. Agreement:
In 2004, PMI entered into an agreement with the European Commission (“E.C.”) and 10 Member States of the European Union that provides for broad cooperation with European law enforcement agencies on anti-contraband and anti-counterfeit efforts. This agreement was signed by all 27 Member States. The agreement resolved all disputes between the parties relating to these issues. Under the terms of the agreement, PMI made 13 payments over 12 years, including an initial payment of $250 million, which was recorded as a pre-tax charge against its earnings in 2004. The agreement called for additional payments of approximately $150 million on the first anniversary of the agreement (this payment was made in July 2005), approximately $100 million on the second anniversary (this payment was made in July 2006) and approximately $75 million each year thereafter for 10 years, each of which was adjusted based on certain variables, including PMI’s market share in the European Union in the year preceding payment. Because the additional payments were subject to these variables, PMI recorded charges for them as an expense in cost of sales when product was shipped. In addition, PMI was also responsible to pay the excise taxes, VAT and customs duties on qualifying product seizures of up to 90 million cigarettes and was subject to payments of five times the applicable taxes and duties if qualifying product seizures exceeded 90 million cigarettes in a given year. In October 2014, this agreement was amended, and the threshold was increased to 450 million cigarettes in a given year. This modification was effective as of July 2012. PMI’s annual payments related to product seizures have been immaterial. Total charges related to the E.C. Agreement of $34 million, $79 million and $71 million were recorded in cost of sales in 2016, 2015 and 2014, respectively. The E.C. decided not to renew the agreement, which expired in July 2016.
115
Note 19. | ||||
Contingencies:
Tobacco-Related Litigation
Legal proceedings covering a wide range of matters are pending or threatened against us, and/or our subsidiaries, and/or our indemnitees in various jurisdictions. Our indemnitees include distributors, licensees and others that have been named as parties in certain cases and that we have agreed to defend, as well as to pay costs and some or all of judgments, if any, that may be entered against them. Pursuant to the terms of the Distribution Agreement between Altria Group, Inc. ("Altria") and PMI, PMI will indemnify Altria and Philip Morris USA Inc. ("PM USA"), a U.S. tobacco subsidiary of Altria, for tobacco product claims based in substantial part on products manufactured by PMI or contract manufactured for PMI by PM USA, and PM USA will indemnify PMI for tobacco product claims based in substantial part on products manufactured by PM USA, excluding tobacco products contract manufactured for PMI.
It is possible that there could be adverse developments in pending cases against us and our subsidiaries. An unfavorable outcome or settlement of pending tobacco-related litigation could encourage the commencement of additional litigation.
Damages claimed in some of the tobacco-related litigation are significant and, in certain cases in Brazil, Canada and Nigeria, range into the billions of U.S. dollars. The variability in pleadings in multiple jurisdictions, together with the actual experience of management in litigating claims, demonstrate that the monetary relief that may be specified in a lawsuit bears little relevance to the ultimate outcome. Much of the tobacco-related litigation is in its early stages, and litigation is subject to uncertainty. However, as discussed below, we have to date been largely successful in defending tobacco-related litigation.
We and our subsidiaries record provisions in the consolidated financial statements for pending litigation when we determine that an unfavorable outcome is probable and the amount of the loss can be reasonably estimated. At the present time, while it is reasonably possible that an unfavorable outcome in a case may occur, after assessing the information available to it (i) management has not concluded that it is probable that a loss has been incurred in any of the pending tobacco-related cases; (ii) management is unable to estimate the possible loss or range of loss for any of the pending tobacco-related cases; and (iii) accordingly, no estimated loss has been accrued in the consolidated financial statements for unfavorable outcomes in these cases, if any. Legal defense costs are expensed as incurred.
It is possible that our consolidated results of operations, cash flows or financial position could be materially affected in a particular fiscal quarter or fiscal year by an unfavorable outcome or settlement of certain pending litigation. Nevertheless, although litigation is subject to uncertainty, we and each of our subsidiaries named as a defendant believe, and each has been so advised by counsel handling the respective cases, that we have valid defenses to the litigation pending against us, as well as valid bases for appeal of adverse verdicts. All such cases are, and will continue to be, vigorously defended. However, we and our subsidiaries may enter into settlement discussions in particular cases if we believe it is in our best interests to do so.
To date, no tobacco-related case has been finally resolved in favor of a plaintiff against us, our subsidiaries or indemnitees.
The table below lists the number of tobacco-related cases pending against us and/or our subsidiaries or indemnitees as of December 31, 2016, December 31, 2015 and December 31, 2014:
Type of Case | Number of Cases Pending as of December 31, 2016 | Number of Cases Pending as of December 31, 2015 | Number of Cases Pending as of December 31, 2014 | ||||||
Individual Smoking and Health Cases | 64 | 68 | 63 | ||||||
Smoking and Health Class Actions | 11 | 11 | 11 | ||||||
Health Care Cost Recovery Actions | 16 | 16 | 15 | ||||||
Lights Class Actions | — | — | — | ||||||
Individual Lights Cases | 3 | 3 | 2 | ||||||
Public Civil Actions | 2 | 3 | 2 | ||||||
Since 1995, when the first tobacco-related litigation was filed against a PMI entity, 454 Smoking and Health, Lights, Health Care Cost Recovery, and Public Civil Actions in which we and/or one of our subsidiaries and/or indemnitees were a defendant have been terminated in our favor. Thirteen cases have had decisions in favor of plaintiffs. Nine of these cases have subsequently reached final resolution in our favor and four remain on appeal.
116
The table below lists the verdict and significant post-trial developments in the four pending cases where a verdict was returned in favor of the plaintiff:
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
February 2004 | Brazil/The Smoker Health Defense Association | Class Action | The Civil Court of São Paulo found defendants liable without hearing evidence. In April 2004, the court awarded “moral damages” of R$1,000 (approximately $319) per smoker per full year of smoking plus interest at the rate of 1% per month, as of the date of the ruling. The court did not assess actual damages, which were to be assessed in a second phase of the case. The size of the class was not defined in the ruling. | Defendants appealed to the São Paulo Court of Appeals, which annulled the ruling in November 2008, finding that the trial court had inappropriately ruled without hearing evidence and returned the case to the trial court for further proceedings. In May 2011, the trial court dismissed the claim. Plaintiff appealed the decision. In February 2015, the appellate court unanimously dismissed plaintiff's appeal. In September 2015, plaintiff appealed to the Superior Court of Justice. In addition, the defendants filed a constitutional appeal to the Federal Supreme Tribunal on the basis that plaintiff did not have standing to bring the lawsuit. This appeal is still pending. | ||||
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
May 27, 2015 | Canada/Cecilia Létourneau | Class Action | On May 27, 2015, the Superior Court of the District of Montreal, Province of Quebec ruled in favor of the Létourneau class on liability and awarded a total of CAD 131 million (approximately $100 million) in punitive damages, allocating CAD 46 million (approximately $35 million) to our subsidiary. The trial court ordered defendants to pay the full punitive damage award into a trust within 60 days. The court did not order the payment of compensatory damages. | In June 2015, our subsidiary commenced the appellate process with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make the payment into a trust. In August 2015, plaintiffs filed a motion for security with the Court of Appeal covering both the Létourneau case and the Blais case described below. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling CAD 226 million (approximately $172 million) to cover both the Létourneau and Blais cases. The hearing for the merits appeal took place in November 2016. (See below for further detail.) | ||||
117
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
May 27, 2015 | Canada/Conseil Québécois Sur Le Tabac Et La Santé and Jean-Yves Blais | Class Action | On May 27, 2015, the Superior Court of the District of Montreal, Province of Quebec ruled in favor of the Blais class on liability and found the class members’ compensatory damages totaled approximately CAD 15.5 billion (approximately $11.8 billion), including pre-judgment interest. The trial court awarded compensatory damages on a joint and several liability basis, allocating 20% to our subsidiary (approximately CAD 3.1 billion including pre-judgment interest (approximately $2.4 billion)). The trial court awarded CAD 90,000 (approximately $68,500) in punitive damages, allocating CAD 30,000 (approximately $22,800) to our subsidiary. The trial court ordered defendants to pay CAD 1 billion (approximately $761 million) of the compensatory damage award, CAD 200 million (approximately $152 million) of which is our subsidiary’s portion, into a trust within 60 days. | In June 2015, our subsidiary commenced the appellate process with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make the payment into a trust. In August 2015, plaintiffs filed a motion for security with the Court of Appeal. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling, together with the Létourneau case, CAD 226 million (approximately $172 million). The hearing for the merits appeal took place in November 2016. (See below for further detail.) | ||||
Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||
August 5, 2016 | Argentina/Hugo Lespada | Individual Action | On August 5, 2016, the Civil Court No. 14 - Mar del Plata, issued a verdict in favor of plaintiff, an individual smoker, and awarded him ARS 110,000 (approximately $6,900), plus interest, in compensatory and moral damages. The Court found that our subsidiary failed to warn plaintiff of the risk of becoming addicted to cigarettes. | On August 23, 2016, our subsidiary filed its notice of appeal. | ||||
118
Pending claims related to tobacco products generally fall within the following categories:
Smoking and Health Litigation: These cases primarily allege personal injury and are brought by individual plaintiffs or on behalf of a class or purported class of individual plaintiffs. Plaintiffs' allegations of liability in these cases are based on various theories of recovery, including negligence, gross negligence, strict liability, fraud, misrepresentation, design defect, failure to warn, breach of express and implied warranties, violations of deceptive trade practice laws and consumer protection statutes. Plaintiffs in these cases seek various forms of relief, including compensatory and other damages, and injunctive and equitable relief. Defenses raised in these cases include licit activity, failure to state a claim, lack of defect, lack of proximate cause, assumption of the risk, contributory negligence, and statute of limitations.
As of December 31, 2016, there were a number of smoking and health cases pending against us, our subsidiaries or indemnitees, as follows:
• | 64 cases brought by individual plaintiffs in Argentina (35), Brazil (16), Canada (2), Chile (6), Costa Rica (2), Italy (1), the Philippines (1) and Scotland (1), compared with 68 such cases on December 31, 2015, and 63 cases on December 31, 2014; and |
• | 11 cases brought on behalf of classes of individual plaintiffs in Brazil (2) and Canada (9), compared with 11 such cases on December 31, 2015 and December 31, 2014. |
In the first class action pending in Brazil, The Smoker Health Defense Association (ADESF) v. Souza Cruz, S.A. and Philip Morris Marketing, S.A., Nineteenth Lower Civil Court of the Central Courts of the Judiciary District of São Paulo, Brazil, filed July 25, 1995, our subsidiary and another member of the industry are defendants. The plaintiff, a consumer organization, is seeking damages for all addicted smokers and former smokers, and injunctive relief. In 2004, the trial court found defendants liable without hearing evidence and awarded “moral damages” of R$1,000 (approximately $319) per smoker per full year of smoking plus interest at the rate of 1% per month, as of the date of the ruling. The court did not award actual damages, which were to be assessed in the second phase of the case. The size of the class was not estimated. Defendants appealed to the São Paulo Court of Appeals, which annulled the ruling in November 2008, finding that the trial court had inappropriately ruled without hearing evidence and returned the case to the trial court for further proceedings. In May 2011, the trial court dismissed the claim. In February 2015, the appellate court unanimously dismissed plaintiff's appeal. In September 2015, plaintiff appealed to the Superior Court of Justice. In addition, the defendants filed a constitutional appeal to the Federal Supreme Tribunal on the basis that plaintiff did not have standing to bring the lawsuit. Both appeals are still pending.
In the second class action pending in Brazil, Public Prosecutor of São Paulo v. Philip Morris Brasil Industria e Comercio Ltda., Civil Court of the City of São Paulo, Brazil, filed August 6, 2007, our subsidiary is a defendant. The plaintiff, the Public Prosecutor of the State of São Paulo, is seeking (i) damages on behalf of all smokers nationwide, former smokers, and their relatives; (ii) damages on behalf of people exposed to environmental tobacco smoke nationwide, and their relatives; and (iii) reimbursement of the health care costs allegedly incurred for the treatment of tobacco-related diseases by all Brazilian States and Municipalities, and the Federal District. In an interim ruling issued in December 2007, the trial court limited the scope of this claim to the State of São Paulo only. In December 2008, the Seventh Civil Court of São Paulo issued a decision declaring that it lacked jurisdiction because the case involved issues similar to the ADESF case discussed above and should be transferred to the Nineteenth Lower Civil Court in São Paulo where the ADESF case is pending. The court further stated that these cases should be consolidated for the purposes of judgment. In April 2010, the São Paulo Court of Appeals reversed the Seventh Civil Court's decision that consolidated the cases, finding that they are based on different legal claims and are progressing at different stages of proceedings. This case was returned to the Seventh Civil Court of São Paulo, and our subsidiary filed its closing arguments in December 2010. In March 2012, the trial court dismissed the case on the merits. In January 2014, the São Paulo Court of Appeals rejected plaintiff’s appeal and affirmed the trial court decision. In July 2014, plaintiff appealed to the Superior Court of Justice.
In the first class action pending in Canada, Cecilia Létourneau v. Imperial Tobacco Ltd., Rothmans, Benson & Hedges Inc. and JTI Macdonald Corp., Quebec Superior Court, Canada, filed in September 1998, our subsidiary and other Canadian manufacturers (Imperial Tobacco Canada Ltd. and JTI-MacDonald Corp.) are defendants. The plaintiff, an individual smoker, sought compensatory and punitive damages for each member of the class who is deemed addicted to smoking. The class was certified in 2005. Trial began in March 2012 and concluded in December 2014. The trial court issued its judgment on May 27, 2015. The trial court found our subsidiary and two other Canadian manufacturers liable and awarded a total of CAD 131 million (approximately $100 million) in punitive damages, allocating CAD 46 million (approximately $35 million) to our subsidiary. The trial court found that defendants violated the Civil Code of Quebec, the Quebec Charter of Human Rights and Freedoms, and the Quebec Consumer Protection Act by failing to warn adequately of the dangers of smoking. The trial court also found that defendants conspired to prevent consumers from learning the dangers of smoking. The trial court further held that these civil faults were a cause of the class members’ addiction. The trial court rejected other grounds of fault advanced by the class, holding that: (i) the evidence was insufficient to show that defendants marketed to youth, (ii) defendants’ advertising did not convey false information about the characteristics of cigarettes, and (iii) defendants did not commit a fault by using
119
the descriptors light or mild for cigarettes with a lower tar delivery. The trial court estimated the size of the addiction class at 918,000 members but declined to award compensatory damages to the addiction class because the evidence did not establish the claims with sufficient accuracy. The trial court ordered defendants to pay the full punitive damage award into a trust within 60 days and found that a claims process to allocate the awarded damages to individual class members would be too expensive and difficult to administer. The trial court ordered a briefing on the proposed process for the distribution of sums remaining from the punitive damage award after payment of attorneys’ fees and legal costs. In June 2015, our subsidiary commenced the appellate process by filing its inscription of appeal of the trial court’s judgment with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust within 60 days notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make the payment into a trust within 60 days. In August 2015, plaintiffs filed a motion with the Court of Appeal seeking security in both the Létourneau case and the Blais case described below. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling CAD 226 million (approximately $172 million), in the form of cash into a court trust or letters of credit, in six equal consecutive quarterly installments of approximately CAD 37.6 million (approximately $28.6 million) beginning in December 2015 through March 2017. See the Blais description for further detail concerning the security order. The Court of Appeal heard oral arguments on the merits appeal in November 2016. Our subsidiary and PMI believe that the findings of liability and damages were incorrect and should ultimately be set aside on any one of many grounds, including the following: (i) holding that defendants violated Quebec law by failing to warn class members of the risks of smoking even after the court found that class members knew, or should have known, of the risks, (ii) finding that plaintiffs were not required to prove that defendants’ alleged misconduct caused injury to each class member in direct contravention of binding precedent, (iii) creating a factual presumption, without any evidence from class members or otherwise, that defendants’ alleged misconduct caused all smoking by all class members, (iv) holding that the addiction class members’ claims for punitive damages were not time-barred even though the case was filed more than three years after a prominent addiction warning appeared on all packages, and (v) awarding punitive damages to punish defendants without proper consideration as to whether punitive damages were necessary to deter future misconduct.
In the second class action pending in Canada, Conseil Québécois Sur Le Tabac Et La Santé and Jean-Yves Blais v. Imperial Tobacco Ltd., Rothmans, Benson & Hedges Inc. and JTI Macdonald Corp., Quebec Superior Court, Canada, filed in November 1998, our subsidiary and other Canadian manufacturers (Imperial Tobacco Canada Ltd. and JTI-MacDonald Corp.) are defendants. The plaintiffs, an anti-smoking organization and an individual smoker, sought compensatory and punitive damages for each member of the class who allegedly suffers from certain smoking-related diseases. The class was certified in 2005. Trial began in March 2012 and concluded in December 2014. The trial court issued its judgment on May 27, 2015. The trial court found our subsidiary and two other Canadian manufacturers liable and found that the class members’ compensatory damages totaled approximately CAD 15.5 billion, including pre-judgment interest (approximately $11.8 billion). The trial court awarded compensatory damages on a joint and several liability basis, allocating 20% to our subsidiary (approximately CAD 3.1 billion, including pre-judgment interest (approximately $2.4 billion)). In addition, the trial court awarded CAD 90,000 (approximately $68,500) in punitive damages, allocating CAD 30,000 (approximately $22,800) to our subsidiary and found that defendants violated the Civil Code of Quebec, the Quebec Charter of Human Rights and Freedoms, and the Quebec Consumer Protection Act by failing to warn adequately of the dangers of smoking. The trial court also found that defendants conspired to prevent consumers from learning the dangers of smoking. The trial court further held that these civil faults were a cause of the class members’ diseases. The trial court rejected other grounds of fault advanced by the class, holding that: (i) the evidence was insufficient to show that defendants marketed to youth, (ii) defendants’ advertising did not convey false information about the characteristics of cigarettes, and (iii) defendants did not commit a fault by using the descriptors light or mild for cigarettes with a lower tar delivery. The trial court estimated the disease class at 99,957 members. The trial court ordered defendants to pay CAD 1 billion (approximately $761 million) of the compensatory damage award into a trust within 60 days, CAD 200 million (approximately $152 million) of which is our subsidiary’s portion and ordered briefing on a proposed claims process for the distribution of damages to individual class members and for payment of attorneys’ fees and legal costs. In June 2015, our subsidiary commenced the appellate process by filing its inscription of appeal of the trial court’s judgment with the Court of Appeal of Quebec. Our subsidiary also filed a motion to cancel the trial court’s order for payment into a trust within 60 days notwithstanding appeal. In July 2015, the Court of Appeal granted the motion to cancel and overturned the trial court’s ruling that our subsidiary make an initial payment within 60 days. In August 2015, plaintiffs filed a motion with the Court of Appeal seeking an order that defendants place irrevocable letters of credit totaling CAD 5 billion (approximately $3.8 billion) into trust, to secure the judgments in both the Létourneau and Blais cases. Plaintiffs subsequently withdrew their motion for security against JTI-MacDonald Corp. and proceeded only against our subsidiary and Imperial Tobacco Canada Ltd. In October 2015, the Court of Appeal granted the motion and ordered our subsidiary to furnish security totaling CAD 226 million (approximately $172 million) to cover both the Létourneau and Blais cases. Such security may take the form of cash into a court trust or letters of credit, in six equal consecutive quarterly installments of approximately CAD 37.6 million (approximately $28.6 million) beginning in December 2015 through March 2017. The Court of Appeal ordered Imperial Tobacco Canada Ltd. to furnish security totaling CAD 758 million (approximately $577 million) in seven equal consecutive quarterly installments of approximately CAD 108 million (approximately $82 million) beginning in December 2015 through June 2017. In December 2016, our subsidiary made its fifth quarterly installment of security for approximately CAD 37.6 million (approximately $28.6 million) into a court trust. This payment is included in other assets on the consolidated balance sheets and in cash used in operating activities in the consolidated statements of cash flows. The Court of Appeal ordered that the security is payable upon a final judgment of the Court of Appeal affirming the trial court’s judgment or upon further order of the Court of Appeal. The Court of Appeal heard oral arguments on the merits appeal in November 2016. Our subsidiary and PMI believe that the findings of liability and damages were incorrect and should ultimately be set aside on any one of many grounds,
120
including the following: (i) holding that defendants violated Quebec law by failing to warn class members of the risks of smoking even after the court found that class members knew, or should have known, of the risks, (ii) finding that plaintiffs were not required to prove that defendants’ alleged misconduct caused injury to each class member in direct contravention of binding precedent, (iii) creating a factual presumption, without any evidence from class members or otherwise, that defendants’ alleged misconduct caused all smoking by all class members, (iv) relying on epidemiological evidence that did not meet recognized scientific standards, and (v) awarding punitive damages to punish defendants without proper consideration as to whether punitive damages were necessary to deter future misconduct.
In the third class action pending in Canada, Kunta v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Winnipeg, Canada, filed June 12, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and chronic obstructive pulmonary disease (“COPD”), severe asthma, and mild reversible lung disease resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, as well as restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. In September 2009, plaintiff's counsel informed defendants that he did not anticipate taking any action in this case while he pursues the class action filed in Saskatchewan (see description of Adams, below).
In the fourth class action pending in Canada, Adams v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Saskatchewan, Canada, filed July 10, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and COPD resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who have smoked a minimum of 25,000 cigarettes and have allegedly suffered, or suffer, from COPD, emphysema, heart disease, or cancer, as well as restitution of profits. Preliminary motions are pending.
In the fifth class action pending in Canada, Semple v. Canadian Tobacco Manufacturers' Council, et al., The Supreme Court (trial court), Nova Scotia, Canada, filed June 18, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges his own addiction to tobacco products and COPD resulting from the use of tobacco products. He is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, as well as restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. No activity in this case is anticipated while plaintiff's counsel pursues the class action filed in Saskatchewan (see description of Adams, above).
In the sixth class action pending in Canada, Dorion v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Alberta, Canada, filed June 15, 2009, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and chronic bronchitis and severe sinus infections resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. To date, we, our subsidiaries, and our indemnitees have not been properly served with the complaint. No activity in this case is anticipated while plaintiff's counsel pursues the class action filed in Saskatchewan (see description of Adams, above).
In the seventh class action pending in Canada, McDermid v. Imperial Tobacco Canada Limited, et al., Supreme Court, British Columbia, Canada, filed June 25, 2010, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges his own addiction to tobacco products and heart disease resulting from the use of tobacco products. He is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who were alive on June 12, 2007, and who suffered from heart disease allegedly caused by smoking, their estates, dependents and family members, plus disgorgement of revenues earned by the defendants from January 1, 1954, to the date the claim was filed.
In the eighth class action pending in Canada, Bourassa v. Imperial Tobacco Canada Limited, et al., Supreme Court, British Columbia, Canada, filed June 25, 2010, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, the heir to a deceased smoker, alleges that the decedent was addicted to tobacco products and suffered from emphysema resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who were alive on June 12, 2007, and who suffered from chronic respiratory diseases allegedly caused by smoking, their estates, dependents and family members, plus disgorgement of revenues earned by the defendants from January 1, 1954, to the date the claim was filed. In December 2014, plaintiff filed an amended statement of claim.
In the ninth class action pending in Canada, Suzanne Jacklin v. Canadian Tobacco Manufacturers' Council, et al., Ontario Superior Court of Justice, filed June 20, 2012, we, our subsidiaries, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and COPD resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who have
121
smoked a minimum of 25,000 cigarettes and have allegedly suffered, or suffer, from COPD, heart disease, or cancer, as well as restitution of profits. Plaintiff's counsel has indicated that he does not intend to take any action in this case in the near future.
Health Care Cost Recovery Litigation: These cases, brought by governmental and non-governmental plaintiffs, seek reimbursement of health care cost expenditures allegedly caused by tobacco products. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including unjust enrichment, negligence, negligent design, strict liability, breach of express and implied warranties, violation of a voluntary undertaking or special duty, fraud, negligent misrepresentation, conspiracy, public nuisance, defective product, failure to warn, sale of cigarettes to minors, and claims under statutes governing competition and deceptive trade practices. Plaintiffs in these cases seek various forms of relief including compensatory and other damages, and injunctive and equitable relief. Defenses raised in these cases include lack of proximate cause, remoteness of injury, failure to state a claim, adequate remedy at law, “unclean hands” (namely, that plaintiffs cannot obtain equitable relief because they participated in, and benefited from, the sale of cigarettes), and statute of limitations.
As of December 31, 2016, there were 16 health care cost recovery cases pending against us, our subsidiaries or indemnitees in Canada (10), Korea (1) and Nigeria (5), compared with 16 such cases on December 31, 2015 and 15 such cases on December 31, 2014.
In the first health care cost recovery case pending in Canada, Her Majesty the Queen in Right of British Columbia v. Imperial Tobacco Limited, et al., Supreme Court, British Columbia, Vancouver Registry, Canada, filed January 24, 2001, we, our subsidiaries, our indemnitee (PM USA), and other members of the industry are defendants. The plaintiff, the government of the province of British Columbia, brought a claim based upon legislation enacted by the province authorizing the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, resulting from a “tobacco related wrong.” The Supreme Court of Canada has held that the statute is constitutional. We and certain other non-Canadian defendants challenged the jurisdiction of the court. The court rejected the jurisdictional challenge. Pre-trial discovery is ongoing.
In the second health care cost recovery case filed in Canada, Her Majesty the Queen in Right of New Brunswick v. Rothmans Inc., et al., Court of Queen's Bench of New Brunswick, Trial Court, New Brunswick, Fredericton, Canada, filed March 13, 2008, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of New Brunswick based on legislation enacted in the province. This legislation is similar to the law introduced in British Columbia that authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing. In September 2016, the trial court entered a consent order establishing a discovery timetable that contemplates the province of New Brunswick applying by September 2017 for a trial date.
In the third health care cost recovery case filed in Canada, Her Majesty the Queen in Right of Ontario v. Rothmans Inc., et al., Ontario Superior Court of Justice, Toronto, Canada, filed September 29, 2009, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Ontario based on legislation enacted in the province. This legislation is similar to the laws introduced in British Columbia and New Brunswick that authorize the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the fourth health care cost recovery case filed in Canada, Attorney General of Newfoundland and Labrador v. Rothmans Inc., et al., Supreme Court of Newfoundland and Labrador, St. Johns, Canada, filed February 8, 2011, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Newfoundland and Labrador based on legislation enacted in the province that is similar to the laws introduced in British Columbia, New Brunswick and Ontario. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the fifth health care cost recovery case filed in Canada, Attorney General of Quebec v. Imperial Tobacco Limited, et al., Superior Court of Quebec, Canada, filed June 8, 2012, we, our subsidiary, our indemnitee (PM USA), and other members of the industry are defendants. The claim was filed by the government of the province of Quebec based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
In the sixth health care cost recovery case filed in Canada, Her Majesty in Right of Alberta v. Altria Group, Inc., et al., Supreme Court of Queen's Bench Alberta, Canada, filed June 8, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Alberta based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Pre-trial discovery is ongoing.
122
In the seventh health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Manitoba v. Rothmans, Benson & Hedges, Inc., et al., The Queen's Bench, Winnipeg Judicial Centre, Canada, filed May 31, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Manitoba based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in September 2014. Pre-trial discovery is ongoing.
In the eighth health care cost recovery case filed in Canada, The Government of Saskatchewan v. Rothmans, Benson & Hedges Inc., et al., Queen's Bench, Judicial Centre of Saskatchewan, Canada, filed June 8, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Saskatchewan based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in February 2015. Discovery is scheduled to begin in 2017.
In the ninth health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Prince Edward Island v. Rothmans, Benson & Hedges Inc., et al., Supreme Court of Prince Edward Island (General Section), Canada, filed September 10, 2012, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Prince Edward Island based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in February 2015. Discovery is scheduled to begin in 2017.
In the tenth health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Nova Scotia v. Rothmans, Benson & Hedges Inc., et al., Supreme Court of Nova Scotia, Canada, filed January 2, 2015, we, our subsidiaries, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Nova Scotia based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.” Defendants filed their defenses in July 2015. Discovery is scheduled to begin in 2017.
In the first health care cost recovery case in Nigeria, The Attorney General of Lagos State v. British American Tobacco (Nigeria) Limited, et al., High Court of Lagos State, Lagos, Nigeria, filed March 13, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We are in the process of making challenges to service and the court's jurisdiction. Currently, the case is stayed in the trial court pending the appeals of certain co-defendants relating to service objections.
In the second health care cost recovery case in Nigeria, The Attorney General of Kano State v. British American Tobacco (Nigeria) Limited, et al., High Court of Kano State, Kano, Nigeria, filed May 9, 2007, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We are in the process of making challenges to service and the court's jurisdiction. Currently, the case is stayed in the trial court pending the appeals of certain co-defendants relating to service objections.
In the third health care cost recovery case in Nigeria, The Attorney General of Gombe State v. British American Tobacco (Nigeria) Limited, et al., High Court of Gombe State, Gombe, Nigeria, filed October 17, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. In February 2011, the court ruled that the plaintiff had not complied with the procedural steps necessary to serve us. As a result of this ruling, plaintiff must re-serve its claim. We have not yet been re-served.
In the fourth health care cost recovery case in Nigeria, The Attorney General of Oyo State, et al., v. British American Tobacco (Nigeria) Limited, et al., High Court of Oyo State, Ibadan, Nigeria, filed May 25, 2007, we and other members of the industry are defendants. Plaintiffs seek reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We challenged service as improper. In June 2010, the court ruled that plaintiffs did not have leave to serve the writ of summons on the defendants and that they must re-serve the writ. We have not yet been re-served.
In the fifth health care cost recovery case in Nigeria, The Attorney General of Ogun State v. British American Tobacco (Nigeria) Limited, et al., High Court of Ogun State, Abeokuta, Nigeria, filed February 26, 2008, we and other members of the industry are defendants.
123
Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. In May 2010, the trial court rejected our service objections. We have appealed.
In the health care cost recovery case in Korea, the National Health Insurance Service v. KT&G, et. al., filed April 14, 2014, our subsidiary and other Korean manufacturers are defendants. Plaintiff alleges that defendants concealed the health hazards of smoking, marketed to youth, added ingredients to make their products more harmful and addictive, and misled consumers into believing that Lights cigarettes are safer than regular cigarettes. The National Health Insurance Service seeks to recover approximately $53.7 million allegedly incurred in treating 3,484 patients with small cell lung cancer, squamous cell lung cancer, and squamous cell laryngeal cancer from 2003 to 2012. The case is now in the evidentiary phase.
Lights Cases: These cases, brought by individual plaintiffs, allege that the use of the term “lights” constitutes fraudulent and misleading conduct. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including misrepresentation, deception, and breach of consumer protection laws. Plaintiffs seek various forms of relief including restitution, injunctive relief, and compensatory and other damages. Defenses raised include lack of causation, lack of reliance, assumption of the risk, and statute of limitations.
As of December 31, 2016, there were 3 lights cases brought by individual plaintiffs pending against our subsidiaries or indemnitees in Chile (2) and Italy (1), compared with 3 such cases on December 31, 2015, and 2 such cases on December 31, 2014.
Public Civil Actions: Claims have been filed either by an individual, or a public or private entity, seeking to protect collective or individual rights, such as the right to health, the right to information or the right to safety. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including product defect, concealment, and misrepresentation. Plaintiffs in these cases seek various forms of relief including injunctive relief such as banning cigarettes, descriptors, smoking in certain places and advertising, as well as implementing communication campaigns and reimbursement of medical expenses incurred by public or private institutions.
As of December 31, 2016, there were 2 public civil actions pending against our subsidiaries in Argentina (1) and Venezuela (1), compared with 3 such cases on December 31, 2015, and 2 such cases on December 31, 2014.
In the public civil action in Argentina, Asociación Argentina de Derecho de Danos v. Massalin Particulares S.A., et al., Civil Court of Buenos Aires, Argentina, filed February 26, 2007, our subsidiary and another member of the industry are defendants. The plaintiff, a consumer association, seeks the establishment of a relief fund for reimbursement of medical costs associated with diseases allegedly caused by smoking. Our subsidiary filed its answer in September 2007. In March 2010, the case file was transferred to the Federal Court on Administrative Matters after the Civil Court granted plaintiff's request to add the national government as a co-plaintiff in the case. The case is currently in the evidentiary stage.
In the public civil action in Venezuela, Federation of Consumers and Users Associations (“FEVACU”), et al. v. National Assembly of Venezuela and the Venezuelan Ministry of Health, Constitutional Chamber of the Venezuelan Supreme Court, filed April 29, 2008, we were not named as a defendant, but the plaintiffs published a notice pursuant to court order, notifying all interested parties to appear in the case. In January 2009, our subsidiary appeared in the case in response to this notice. The plaintiffs purport to represent the right to health of the citizens of Venezuela and claim that the government failed to protect adequately its citizens' right to health. The claim asks the court to order the government to enact stricter regulations on the manufacture and sale of tobacco products. In addition, the plaintiffs ask the court to order companies involved in the tobacco industry to allocate a percentage of their “sales or benefits” to establish a fund to pay for the health care costs of treating smoking-related diseases. In October 2008, the court ruled that plaintiffs have standing to file the claim and that the claim meets the threshold admissibility requirements. In December 2012, the court admitted our subsidiary and BAT's subsidiary as interested third parties. In February 2013, our subsidiary answered the complaint.
Other Litigation
The Department of Special Investigations of the government of Thailand ("DSI") conducted an investigation into alleged underpayment by our subsidiary, Philip Morris (Thailand) Limited ("PM Thailand"), of customs duties and excise taxes relating to imports from the Philippines covering the period 2003-2007. On January 18, 2016, the Public Prosecutor filed charges against our subsidiary and seven former and current employees in the Bangkok Criminal Court alleging that PM Thailand and the individual defendants jointly and with the intention to defraud the Thai government, underdeclared import prices of cigarettes to avoid full payment of taxes and duties in connection with 272 import entries of cigarettes from the Philippines during the period of July 2003 to June 2006. The government is seeking a fine of approximately THB 80.8 billion (approximately $2.29 billion). The case is in the pre-trial evidentiary phase. Trials are scheduled to begin during the last quarter of 2017. PM Thailand believes that its declared import prices are in compliance with the Customs Valuation Agreement of the World Trade Organization and Thai law and that the allegations of the Public Prosecutor are inconsistent with several decisions already taken by Thai Customs and other Thai governmental agencies.
124
The DSI also conducted an investigation into alleged underpayment by PM Thailand of customs duties and excise taxes relating to imports from Indonesia covering the period 2000-2003. On January 26, 2017, the Public Prosecutor filed charges against PM Thailand and its Thai ex-employee in the Bangkok Criminal Court alleging that PM Thailand and its Thai ex-employee jointly and with the intention to defraud the Thai government underdeclared import prices of cigarettes to avoid full payment of taxes and duties in connection with 780 import entries during the period from January 2002 to July 2003. The government is seeking a fine of approximately THB 19.8 billion (approximately $561 million). The first hearing, which will focus on preliminary procedural matters, is scheduled for April 2017. PM Thailand disagrees with the allegations and believes that its declared import prices are in compliance with the Customs Valuation Agreement of the WTO and Thai law.
The South Korean Board of Audit and Inspection (“BAI”) conducted an audit of certain Korean government agencies and the tobacco industry into whether inventory movements ahead of the January 1, 2015 increase of cigarette-related taxes by tobacco companies, including Philip Morris Korea Inc. ("PM Korea"), our South Korean affiliate, were in compliance with South Korean tax laws. In November 2016, the tax authorities completed their audit and assessed allegedly underpaid taxes and penalties. In order to avoid nonpayment charges, PM Korea paid the total amount that the tax authorities demanded, namely, approximately KRW 100 billion (approximately $85 million) in December 2016 and KRW 118 billion (approximately $101 million) in January 2017. The total demanded amount is included in other assets, and the January 2017 payment is included in accrued liabilities, in the consolidated balance sheets for the year ended 2016. The December 2016 payment is reflected in cash used in operating activities in the consolidated statements of cash flows for the year ended 2016. In the beginning of 2017, PM Korea received demand notices from other government authorities for other amounts of approximately KRW 54 billion (approximately $47 million) in the aggregate. PM Korea will appeal the assessments. The tax authorities have also referred the matter to the Public Prosecutor, who will further investigate and decide whether to file criminal charges against PM Korea and/or other alleged co-offenders. If the Public Prosecutor decides to prosecute, it may seek up to three times the underpaid tax for company criminal penalties and up to five times the underpaid tax for individual criminal penalties. PM Korea believes that it has paid cigarette-related taxes in compliance with the South Korean tax laws. In addition, the South Korean Ministry of Strategy and Finance (“MOSF”) filed a criminal complaint with the Public Prosecutor against PM Korea and its managing director. In its criminal complaint, the MOSF alleged that PM Korea exceeded the monthly product withdrawal limits that the MOSF had set in its notice. The Public Prosecutor will investigate the MOSF’s criminal complaint and decide whether to prosecute. PM Korea disagrees with the MOSF’s allegations.
We are also involved in additional litigation arising in the ordinary course of our business. While the outcomes of these proceedings are uncertain, management does not expect that the ultimate outcomes of other litigation, including any reasonably possible losses in excess of current accruals, will have a material adverse effect on our consolidated results of operations, cash flows or financial position.
Note 20. | |||||
Balance Sheet Offsetting:
Derivative Financial Instruments
PMI uses foreign exchange contracts and interest rate contracts to mitigate its exposure to changes in exchange and interest rates from third-party and intercompany actual and forecasted transactions. Substantially all of PMI's derivative financial instruments are subject to master netting arrangements, whereby the right to offset occurs in the event of default by a participating party. While these contracts contain the enforceable right to offset through close-out netting rights, PMI elects to present them on a gross basis in the consolidated balance sheets. Collateral associated with these arrangements is in the form of cash and is unrestricted. See Note 15. Financial Instruments for disclosures related to PMI's derivative financial instruments.
125
The effects of these derivative financial instrument assets and liabilities on PMI's consolidated balance sheets were as follows:
(in millions) | Gross Amounts Recognized | Gross Amount Offset in the Consolidated Balance Sheet | Net Amounts Presented in the Consolidated Balance Sheet | Gross Amounts Not Offset in the Consolidated Balance Sheet | Net Amount | ||||||||||||||
Financial Instruments | Cash Collateral Received/Pledged | ||||||||||||||||||
At December 31, 2016 | |||||||||||||||||||
Assets | |||||||||||||||||||
Foreign exchange contracts | $ | 813 | $ | — | $ | 813 | $ | (126 | ) | $ | (607 | ) | $ | 80 | |||||
Liabilities | |||||||||||||||||||
Foreign exchange contracts | $ | 163 | $ | — | $ | 163 | $ | (126 | ) | $ | (31 | ) | $ | 6 | |||||
At December 31, 2015 | |||||||||||||||||||
Assets | |||||||||||||||||||
Foreign exchange contracts | $ | 574 | $ | — | $ | 574 | $ | (131 | ) | $ | (432 | ) | $ | 11 | |||||
Liabilities | |||||||||||||||||||
Foreign exchange contracts | $ | 172 | $ | — | $ | 172 | $ | (131 | ) | $ | (30 | ) | $ | 11 | |||||
Note 21. | |||||
Sale of Accounts Receivable:
To mitigate risk and enhance cash and liquidity management PMI sells trade receivables to unaffiliated financial institutions. These arrangements allow PMI to sell, on an ongoing basis, certain trade receivables without recourse. The trade receivables sold are generally short-term in nature and are removed from the consolidated balance sheets. PMI sells trade receivables under two types of arrangements, servicing and non-servicing. For servicing arrangements, PMI continues to service the sold trade receivables on an administrative basis and does not act on behalf of the unaffiliated financial institutions. When applicable, a servicing liability is recorded for the estimated fair value of the servicing. The amounts associated with the servicing liability were not material for the years ended December 31, 2016 and 2015. Under the non-servicing arrangements, PMI does not provide any administrative support or servicing after the trade receivables have been sold to the unaffiliated financial institutions.
Cumulative trade receivables sold, including excise taxes, for the years ended December 31, 2016 and 2015, were $9,447 million and $3,299 million, respectively. PMI’s operating cash flows were positively impacted by the amount of the trade receivables sold and derecognized from the consolidated balance sheets, which remained outstanding with the unaffiliated financial institutions. The trade receivables sold that remained outstanding under these arrangements as of December 31, 2016, 2015 and 2014, were $729 million, $888 million and $120 million, respectively. The net proceeds received are included in cash provided by operating activities in the consolidated statements of cash flows. The difference between the carrying amount of the trade receivables sold and the sum of the cash received is recorded as a loss on sale of trade receivables within marketing, administration and research costs in the consolidated statements of earnings. For the years ended December 31, 2016, 2015 and 2014 the loss on sale of trade receivables was immaterial.
Note 22. | |||||
New Accounting Standards:
On February 25, 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update ASU 2016-02, “Leases” (“ASU 2016-02”). ASU 2016-02 requires organizations that lease assets to recognize on the balance sheet the assets and liabilities for the rights and obligations created by those leases. Additionally, ASU 2016-02 modifies current guidance for lessors' accounting. ASU 2016-02 is effective for interim and annual reporting periods beginning on or after January 1, 2019, with early adoption permitted. PMI is currently assessing the impact that the adoption of ASU 2016-02 will have on its financial position or results of operations.
126
On January 5, 2016, the FASB issued Accounting Standard Update ASU 2016-01, “Financial Instruments - Overall (Subtopic 825-10): Recognition and Measurement of Financial Assets and Financial Liabilities” (“ASU 2016-01”). ASU 2016-01 will require equity investments (except those accounted for under the equity method of accounting, or those that result in consolidation of the investee) to be measured at fair value with changes in fair value recognized in net income. Additionally, ASU 2016-01 also changes certain disclosure requirements and other aspects of current U.S. GAAP. ASU 2016-01 is effective for interim and annual reporting periods beginning on or after January 1, 2018. PMI is currently assessing its cost method investments and the impact that the adoption of ASU 2016-01 will have on its financial position or results of operations.
On November 20, 2015, the FASB issued Accounting Standard Update ASU 2015-17, “Balance Sheet Classification of Deferred Taxes” (“ASU 2015-17”). ASU 2015-17 requires that all deferred tax assets and liabilities, along with any related valuation allowance, be classified as noncurrent on the balance sheet. ASU 2015-17 is effective for interim and annual reporting periods beginning on or after January 1, 2017, with early adoption permitted. PMI elected to adopt ASU 2015-17 prospectively as of October 1, 2016. Prior periods have not been retrospectively adjusted. Had ASU 2015-17 been adopted retrospectively, the amounts in the consolidated balance sheets as of December 31, 2015, would have been as follows:
At December 31, 2015 | |||||||
(in millions) | As Reported | Retrospective Adoption | |||||
Assets | |||||||
Current deferred income taxes | $ | 488 | $ | — | |||
Noncurrent deferred income taxes | $ | 360 | $ | 769 | |||
Other assets | $ | 1,143 | $ | 1,143 | |||
Total assets | $ | 33,956 | $ | 33,877 | |||
Liabilities | |||||||
Current deferred income taxes | $ | 206 | $ | — | |||
Noncurrent deferred income taxes | $ | 1,543 | $ | 1,670 | |||
Total liabilities | $ | 45,432 | $ | 45,353 | |||
The adoption of ASU 2015-17 did not have a material impact on PMI’s consolidated results of operations, financial position or cash flows.
On May 28, 2014, the FASB issued Accounting Standards Update ASU 2014-09, “Revenue from Contracts with Customers” (“ASU 2014-09”). ASU 2014-09 contains principles that an entity will need to apply to determine the measurement of revenue and timing of when it is recognized. The underlying principle is that an entity will recognize revenue to depict the transfer of promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services.
Entities can apply the final standard using one of the following two methods:
1. | retrospectively to each prior period presented; or |
2. | retrospectively, with the cumulative effect of initially applying ASU 2014-09 recognized at the date of initial application, with additional disclosures in reporting periods that include the date of initial application. |
ASU 2014-09 is effective for interim and annual reporting periods beginning on or after January 1, 2017. In July 2015, the FASB approved a proposal which allows for a deferral of the implementation until January 1, 2018, and permits early application, but not before the original effective date of January 1, 2017. PMI plans to adopt ASU 2014-09 on January 1, 2018, and does not expect the adoption to have a material impact on its consolidated financial position or results of operations. Following the release of ASU 2016-12 in May 2016, which provided some practical expedients to the original standard, PMI is continuing to assess the standard’s policy election for gross versus net presentation of excise taxes and, related to this assessment, the method of adoption.
127
Note 23. | |||||||||
Quarterly Financial Data (Unaudited):
2016 Quarters | |||||||||||||||
(in millions, except per share data) | 1st | 2nd | 3rd | 4th | |||||||||||
Net revenues | $ | 16,788 | $ | 19,041 | $ | 19,935 | $ | 19,189 | |||||||
Gross profit | $ | 3,987 | $ | 4,285 | $ | 4,550 | $ | 4,472 | |||||||
Net earnings attributable to PMI | $ | 1,530 | $ | 1,788 | $ | 1,938 | $ | 1,711 | |||||||
Per share data: | |||||||||||||||
Basic EPS | $ | 0.98 | $ | 1.15 | $ | 1.25 | $ | 1.10 | |||||||
Diluted EPS | $ | 0.98 | $ | 1.15 | $ | 1.25 | $ | 1.10 | |||||||
Dividends declared | $ | 1.02 | $ | 1.02 | $ | 1.04 | $ | 1.04 | |||||||
Market price: | |||||||||||||||
— High | $ | 99.53 | $ | 102.55 | $ | 104.20 | $ | 98.21 | |||||||
— Low | $ | 84.46 | $ | 95.91 | $ | 96.95 | $ | 86.78 | |||||||
2015 Quarters | |||||||||||||||
(in millions, except per share data) | 1st | 2nd | 3rd | 4th | |||||||||||
Net revenues | $ | 17,352 | $ | 18,763 | $ | 19,422 | $ | 18,371 | |||||||
Gross profit | $ | 4,387 | $ | 4,481 | $ | 4,544 | $ | 4,017 | |||||||
Net earnings attributable to PMI | $ | 1,795 | $ | 1,887 | $ | 1,942 | $ | 1,249 | |||||||
Per share data: | |||||||||||||||
Basic EPS | $ | 1.16 | $ | 1.21 | $ | 1.25 | $ | 0.80 | |||||||
Diluted EPS | $ | 1.16 | $ | 1.21 | $ | 1.25 | $ | 0.80 | |||||||
Dividends declared | $ | 1.00 | $ | 1.00 | $ | 1.02 | $ | 1.02 | |||||||
Market price: | |||||||||||||||
— High | $ | 85.29 | $ | 86.91 | $ | 86.51 | $ | 90.27 | |||||||
— Low | $ | 75.30 | $ | 75.27 | $ | 76.54 | $ | 78.41 | |||||||
Basic and diluted EPS are computed independently for each of the periods presented. Accordingly, the sum of the quarterly EPS amounts may not agree to the total for the year.
128
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of
Philip Morris International Inc. and Subsidiaries:
In our opinion, the accompanying consolidated balance sheets and the related consolidated statements of earnings, comprehensive earnings, stockholders’ (deficit) equity, and cash flows, present fairly, in all material respects, the financial position of Philip Morris International Inc. and its subsidiaries (“PMI”) at December 31, 2016 and 2015, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2016 in conformity with accounting principles generally accepted in the United States of America. Also in our opinion, PMI maintained, in all material respects, effective internal control over financial reporting as of December 31, 2016, based on criteria established in Internal Control — Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). PMI’s management is responsible for these financial statements, for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Report of Management on Internal Control over Financial Reporting. Our responsibility is to express opinions on these financial statements and on PMI’s internal control over financial reporting based on our integrated audits. We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement and whether effective internal control over financial reporting was maintained in all material respects. Our audits of the financial statements included examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
PricewaterhouseCoopers SA
/S/ BARRY J. MISTHAL | /S/ DR. MICHAEL ABRESCH | |
Barry J. Misthal | Dr. Michael Abresch | |
Lausanne, Switzerland | ||
February 2, 2017 | ||
129
Report of Management on Internal Control Over Financial Reporting
Management of Philip Morris International Inc. (“PMI”) is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Securities Exchange Act of 1934, as amended. PMI’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America. Internal control over financial reporting includes those written policies and procedures that:
• | pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of PMI; |
• | provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with accounting principles generally accepted in the United States of America; |
• | provide reasonable assurance that receipts and expenditures of PMI are being made only in accordance with the authorization of management and directors of PMI; and |
• | provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of assets that could have a material effect on the consolidated financial statements. |
Internal control over financial reporting includes the controls themselves, monitoring and internal auditing practices and actions taken to correct deficiencies as identified.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Management assessed the effectiveness of PMI’s internal control over financial reporting as of December 31, 2016. Management based this assessment on criteria for effective internal control over financial reporting described in Internal Control — Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission. Management’s assessment included an evaluation of the design of PMI’s internal control over financial reporting and testing of the operational effectiveness of its internal control over financial reporting. Management reviewed the results of its assessment with the Audit Committee of our Board of Directors.
Based on this assessment, management determined that, as of December 31, 2016, PMI maintained effective internal control over financial reporting.
PricewaterhouseCoopers SA, an independent registered public accounting firm, who audited and reported on the consolidated financial statements of PMI included in this report, has audited the effectiveness of PMI’s internal control over financial reporting as of December 31, 2016, as stated in their report herein.
February 2, 2017
130
Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure. |
None.
Item 9A. | Controls and Procedures. |
PMI carried out an evaluation, with the participation of PMI’s management, including PMI’s Chief Executive Officer and Chief Financial Officer, of the effectiveness of PMI’s disclosure controls and procedures (as defined in Rule 13a-15(e) under the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this report. Based upon that evaluation, PMI’s Chief Executive Officer and Chief Financial Officer concluded that PMI’s disclosure controls and procedures are effective. There have been no changes in PMI’s internal control over financial reporting during the most recent fiscal quarter that have materially affected, or are reasonably likely to materially affect, PMI’s internal control over financial reporting.
The Report of Management on Internal Control over Financial Reporting and the Report of Independent Registered Public Accounting Firm are included in Item 8.
Item 9B. | Other Information. |
None.
PART III
Except for the information relating to the executive officers set forth in Item 10 and the information relating to equity compensation plans set forth in Item 12, the information called for by Items 10-14 is hereby incorporated by reference to PMI’s definitive proxy statement for use in connection with its annual meeting of stockholders to be held on May 3, 2017, that will be filed with the SEC on or about March 23, 2017 (the “proxy statement”), and, except as indicated therein, made a part hereof.
Item 10. | Directors, Executive Officers and Corporate Governance. |
Executive Officers as of February 14, 2017:
Name | Office | Age | ||||
André Calantzopoulos | Chief Executive Officer | 59 | ||||
Drago Azinovic | President, Eastern Europe, Middle East & Africa Region & PMI Duty Free | 54 | ||||
Werner Barth | Senior Vice President, Marketing & Sales | 52 | ||||
Charles Bendotti | Senior Vice President, Human Resources | 44 | ||||
Patrick Brunel | Senior Vice President and Chief Information Officer | 51 | ||||
Frank de Rooij | Vice President, Treasury and Corporate Finance | 51 | ||||
Frederic de Wilde | President, European Union Region | 49 | ||||
Marc S. Firestone | Senior Vice President and General Counsel | 57 | ||||
Paul Janelle | Vice President, Corporate Planning and Business Development | 51 | ||||
Martin King | President, Asia Region | 52 | ||||
Andreas Kurali | Vice President and Controller | 51 | ||||
Marco Mariotti | Senior Vice President, Corporate Affairs | 52 | ||||
Antonio Marques | Senior Vice President, Operations | 61 | ||||
Jacek Olczak | Chief Financial Officer | 52 | ||||
Jeanne Pollès | President, Latin America & Canada Region | 52 | ||||
Jerry E. Whitson | Deputy General Counsel and Corporate Secretary | 61 | ||||
Miroslaw Zielinski | President, Reduced-Risk Products | 55 | ||||
All of the above-mentioned officers, except for Mr. Firestone, have been employed by us in various capacities during the past five years.
131
Before joining Philip Morris International Inc. in April 2012, Mr. Firestone was Executive Vice President, Corporate and Legal Affairs and General Counsel of Kraft Foods Inc., where he served since 2003. From 1988 to 2003, Mr. Firestone held numerous positions in the law departments of Philip Morris Companies Inc. and Philip Morris International Inc., lastly as Senior Vice President & General Counsel of PMI.
Codes of Conduct and Corporate Governance
We have adopted the Philip Morris International Code of Conduct, which complies with requirements set forth in Item 406 of Regulation S-K. This Code of Conduct applies to all of our employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, and persons performing similar functions. We have also adopted a code of business conduct and ethics that applies to the members of our Board of Directors. These documents are available free of charge on our website at www.pmi.com.
In addition, we have adopted corporate governance guidelines and charters for our Audit, Finance, Compensation and Leadership Development, Product Innovation and Regulatory Affairs and Nominating and Corporate Governance committees of the Board of Directors. All of these documents are available free of charge on our website at www.pmi.com. Any waiver granted by Philip Morris International Inc. to its principal executive officer, principal financial officer or controller or any person performing similar functions under the Code of Conduct, or certain amendments to the Code of Conduct, will be disclosed on our website at www.pmi.com.
The information on our website is not, and shall not be deemed to be, a part of this Report or incorporated into any other filings made with the SEC.
Also refer to Board Operations and Governance - Committees of the Board, Election of Directors - Process for Nominating Directors and Election of Directors - Director Nominees and Section 16(a) Beneficial Ownership Reporting Compliance sections of the proxy statement.
Item 11. | Executive Compensation. |
Refer to Compensation Discussion and Analysis and Compensation of Directors sections of the proxy statement.
Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters. |
The number of shares to be issued upon exercise or vesting and the number of shares remaining available for future issuance under PMI’s equity compensation plans at December 31, 2016, were as follows:
Number of Securities to be Issued upon Exercise of Outstanding Options and Vesting of RSUs and PSUs (a) | Weighted Average Exercise Price of Outstanding Options and Vesting of RSUs and PSUs (b) | Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (excluding Securities reflected in column (a)) (c) | |||||||
Equity compensation plans approved by stockholders(1) | 5,356,130(2) | $ | — | 21,857,569 | |||||
(1) The 2008 Stock Compensation Plan for Non-Employee Directors was approved by our former parent company in connection with the Spin-off.
(2) Represents 4,500,990 shares of common stock that may be issued upon vesting of the restricted share units granted under the Philip Morris International Inc. 2012 Performance Incentive Plan (the "2012 Plan") and 855,140 shares that may be issued upon vesting of the performance share units granted under the 2012 Plan if maximum performance targets are achieved for each performance cycle. PMI has not granted options since the Spin-off.
Refer to Stock Ownership Information section of the proxy statement.
132
Item 13. | Certain Relationships and Related Transactions, and Director Independence. |
Refer to Related Person Transactions and Code of Conduct and Election of Directors - Independence of Nominees sections of the proxy statement.
Item 14. | Principal Accounting Fees and Services. |
Refer to Audit Committee Matters section of the proxy statement.
PART IV
Item 15. | Exhibits and Financial Statement Schedules. |
(a) Index to Consolidated Financial Statements and Schedules
Page | |
Consolidated Balance Sheets at December 31, 2016 and 2015 | 78 - 79 |
Consolidated Statements of Earnings for the years ended December 31, 2016, 2015 and 2014 | 80 |
Consolidated Statements of Comprehensive Earnings for the years ended December 31, 2016, 2015 and 2014 | 81 |
Consolidated Statements of Stockholders’ (Deficit) Equity for the years ended December 31, 2016, 2015 and 2014 | 82 |
Consolidated Statements of Cash Flows for the years ended December 31, 2016, 2015 and 2014 | 83 - 84 |
Notes to Consolidated Financial Statements | 85 - 128 |
Report of Independent Registered Public Accounting Firm | 129 |
Report of Management on Internal Control Over Financial Reporting | 130 |
Schedules have been omitted either because such schedules are not required or are not applicable.
(b) The following exhibits are filed as part of this Report:
133
2.1 | — | Distribution Agreement between Altria Group, Inc. and Philip Morris International Inc. dated January 30, 2008 (incorporated by reference to Exhibit 2.1 to the Registration Statement on Form 10 filed February 7, 2008). | ||
3.1 | — | Amended and Restated Articles of Incorporation of Philip Morris International Inc. (incorporated by reference to Exhibit 3.1 to the Registration Statement on Form 10 filed February 7, 2008). | ||
3.2 | — | Amended and Restated By-laws of Philip Morris International Inc. (incorporated by reference to Exhibit 3.1 to the Current Report on Form 8-K filed December 9, 2016). | ||
4.1 | — | Specimen Stock Certificate of Philip Morris International Inc. (incorporated by reference to Exhibit 4.1 to the Registration Statement on Form 10 filed February 7, 2008). | ||
4.2 | — | Indenture dated as of April 25, 2008, between Philip Morris International Inc. and HSBC Bank USA, National Association, as Trustee (incorporated by reference to Exhibit 4.3 to the Registration Statement on Form S-3, dated April 25, 2008). | ||
4.3 | — | Issue and Paying Agency Agreement, dated March 13, 2009, by and among Philip Morris International Inc., HSBC Private Bank (C.I.) Limited, Jersey Branch, as registrar, HSBC Bank PLC, as principal paying agent and HSBC Corporate Trustee Company (UK) Limited, as trustee (incorporated by reference to Exhibit 4.1 to the Current Report on Form 8-K filed March 19, 2009). | ||
4.4 | — | Trust Deed relating to Euro Medium Term Note Programme, dated March 13, 2009, between Philip Morris International Inc., as issuer, and HSBC Corporate Trustee Company (UK) Limited, as trustee (incorporated by reference to Exhibit 4.2 to the Current Report on Form 8-K filed March 19, 2009). | ||
4.5 | — | Deed of Termination and Release, dated April 28, 2016, of the Euro Medium Term Note Programme, between Philip Morris International Inc., as issuer, HSBC Private Bank (C.I.) Limited, as registrar, HSBC Bank PLC, as principal paying agent, HSBC Bank PLC, as the transfer agent, and HSBC Corporate Trustee Company (UK) Limited, as trustee (incorporated by reference to Exhibit 4.1 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2016). | ||
4.6 | — | The Registrant agrees to furnish copies of any instruments defining the rights of holders of long-term debt of the Registrant and its consolidated subsidiaries that does not exceed 10 percent of the total assets of the Registrant and its consolidated subsidiaries to the Commission upon request. | ||
10.1 | — | Employee Matters Agreement between Altria Group, Inc. and Philip Morris International Inc., dated as of March 28, 2008 (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed March 31, 2008). | ||
10.2 | — | Intellectual Property Agreement between Philip Morris International Inc. and Philip Morris USA Inc., dated as of January 1, 2008 (incorporated by reference to Exhibit 10.4 to the Registration Statement on Form 10 filed March 5, 2008). | ||
10.3 | — | Credit Agreement relating to a US$3,500,000,000 Revolving Credit Facility (including a US$800,000,000 swingline option) dated as of October 25, 2011, among Philip Morris International Inc. and the Initial Lenders named therein and Citibank International plc, as Facility Agent, and Citibank, N.A., as Swingline Agent, and Citigroup Global Markets Limited, Barclays Capital, BNP Paribas, Credit Suisse AG, Cayman Islands Branch, Deutsche Bank Securities Inc., Goldman Sachs International, HSBC Bank PLC, J.P. Morgan Limited, RBS Securities Inc. and Société Générale as Mandated Lead Arrangers and Bookrunners (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed October 26, 2011). | ||
10.4 | __ | Amendment No. 1, dated as of August 31, 2012, to the Credit Agreement, dated as of October 25, 2011, among Philip Morris International Inc., the lenders named therein and Citibank International plc, as Facility Agent (incorporated by reference to Exhibit 10.6 to the Quarterly Report on Form 10-Q for the quarter ended September 30, 2012). | ||
10.5 | __ | Credit Agreement, dated as of February 12, 2013, among Philip Morris International Inc., the lenders named therein and Citibank Europe PLC, UK Branch (formerly, The Royal Bank of Scotland plc), as Administrative Agent (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed February 15, 2013). | ||
134
10.6 | __ | Extension Agreement, effective January 31, 2014, to Credit Agreement, dated as of February 12, 2013, among Philip Morris International Inc., the lenders party thereto and Citibank Europe PLC, UK Branch (formerly, The Royal Bank of Scotland plc), as Administrative Agent (incorporated by reference to Exhibit 10.3 to the Quarterly Report on Form 10-Q for the quarter ended March 31, 2014). | ||
10.7 | __ | Credit Agreement, dated as of February 28, 2014, among Philip Morris International Inc., the lenders named therein, J.P. Morgan Europe Limited, as Facility Agent, and JPMorgan Chase Bank, N.A., as Swingline Agent (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed March 3, 2014). | ||
10.8 | __ | Extension Agreement, effective as of February 10, 2015, to Credit Agreement dated as of February 12, 2013, among Philip Morris International Inc., the lenders named therein and Citibank Europe PLC, UK Branch (formerly, The Royal Bank of Scotland plc), as Administrative Agent (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed January 29, 2015). | ||
10.9 | __ | Extension Agreement, effective as of February 28, 2015, to the Credit Agreement, dated as of February 28, 2014, among Philip Morris International Inc., the lenders named therein, J.P. Morgan Europe Limited, as Facility Agent, and JPMorgan Chase Bank, N.A. as Swingline Agent (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed January 29, 2015). | ||
10.10 | __ | Amendment No. 1, dated as of July 20, 2015, to the Credit Agreement, dated as of February 12, 2013, among Philip Morris International Inc., the lenders named therein, The Royal Bank of Scotland plc, as resigning administrative agent, and Citibank Europe PLC, UK Branch (formerly, Citibank International Limited), as successor administrative agent (incorporated by reference to Exhibit 10.52 to the Annual Report on Form 10-K for the year ended December 31, 2015). | ||
10.11 | — | Credit Agreement, dated as of October 1, 2015, among Philip Morris International Inc., the lenders named therein, Citibank Europe PLC, UK Branch (formerly, Citibank International Limited), as Facility Agent, and Citibank, N.A., as Swingline Agent (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed October 5, 2015). | ||
10.12 | — | Amendment No. 2, effective as of February 9, 2016, to the Credit Agreement dated as of February 12, 2013, with the lenders named therein and Citibank Europe PLC, UK Branch (formerly, Citibank International Limited), as administrative agent (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed January 28, 2016). | ||
10.13 | — | Extension Agreement, effective February 28, 2016, to the Credit Agreement, dated as of February 28, 2014, among Philip Morris International Inc., each lender named therein, J.P. Morgan Europe Limited, as facility agent, and JPMorgan Chase Bank, N.A., as swingline agent (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed January 28, 2016). | ||
10.14 | — | Extension Agreement, effective as of October 1, 2016, to the Credit Agreement dated as of October 1, 2015, among Philip Morris International Inc., lenders named therein, Citibank Europe PLC, UK Branch (formerly, Citibank International Limited), as facility agent, and Citibank, N.A., as swingline agent (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed August 31, 2016). | ||
10.15 | — | Anti-Contraband and Anti-Counterfeit Agreement and General Release, dated as of July 9, 2004, and Appendices (Portions of this exhibit have been omitted pursuant to a request for confidential treatment filed with the Securities and Exchange Commission) (incorporated by reference to Exhibit 10.7 to the Registration Statement on Form 10 filed February 7, 2008). | ||
10.16 | — | Modification Agreement, dated as of October 14, 2014, to the Anti-Contraband and Anti-Counterfeit Agreement and General Release, dated as of July 9, 2004 (incorporated by reference to Exhibit 10.2 to the Quarterly Report on Form 10-Q for the quarter ended September 30, 2014). | ||
10.17 | — | Philip Morris International Inc. Automobile Policy (incorporated by reference to Exhibit 10.8 to the Registration Statement on Form 10 filed February 7, 2008).* | ||
135
10.18 | — | Philip Morris International Benefit Equalization Plan, as amended and in effect on August 6, 2012 (incorporated by reference to Exhibit 10.2 to the Quarterly Report on Form 10-Q for the quarter ended September 30, 2012).* | ||
10.19 | — | Philip Morris International Inc. 2012 Performance Incentive Plan, effective May 9, 2012 (incorporated by reference to Exhibit A to the Definitive Proxy Statement filed on March 30, 2012).* | ||
10.20 | — | Pension Fund of Philip Morris in Switzerland (IC) (incorporated by reference to Exhibit 10.2 to the Quarterly Report on Form 10-Q for the quarter ended March 31, 2015).* | ||
10.21 | — | Summary of Supplemental Pension Plan of Philip Morris in Switzerland (incorporated by reference to Exhibit 10.1 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2015).* | ||
10.22 | — | Form of Restated Employee Grantor Trust Enrollment Agreement (Executive Trust Arrangement) (incorporated by reference to Exhibit 10.18 to the Registration Statement on Form 10 filed February 7, 2008).* | ||
10.23 | — | Form of Restated Employee Grantor Trust Enrollment Agreement (Secular Trust Arrangement) (incorporated by reference to Exhibit 10.19 to the Registration Statement on Form 10 filed February 7, 2008).* | ||
10.24 | — | Philip Morris International Inc. 2008 Stock Compensation Plan for Non-Employee Directors (amended and restated as of January 1, 2015) (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed December 15, 2014).* | ||
10.25 | — | Philip Morris International Inc. 2008 Deferred Fee Plan for Non-Employee Directors (incorporated by reference to Exhibit 10.21 to the Registration Statement on Form 10 filed February 7, 2008).* | ||
10.26 | — | Supplemental Letter to the Employment Agreement (as amended) with André Calantzopoulos (incorporated by reference to Exhibit 10.25 to the Annual Report on Form 10-K for the year ended December 31, 2014). The Employment Agreement was previously filed as Exhibit 10.22 to the Registration Statement on Form 10 filed February 7, 2008 and is incorporated by reference to this Exhibit 10.26. The Amendment to the Employment Agreement was previously filed as Exhibit 10.1 to the Current Report on Form 8-K/A filed June 13, 2013, and is incorporated by reference to this Exhibit 10.26.* | ||
10.27 | — | Amendment to Employment Agreement with Marc S. Firestone (incorporated by reference to Exhibit 10.25 to the Annual Report on Form 10-K for the year ended December 31, 2013). The Employment Agreement was previously filed as Exhibit 10.1 to the Quarterly Report on Form 10-Q for the quarter ended March 31, 2013, and is incorporated by reference to this Exhibit 10.27.* | ||
10.28 | — | Amendment to Employment Agreement with Matteo Pellegrini (incorporated by reference to Exhibit 10.26 to the Annual Report on Form 10-K for the year ended December 31, 2013). The Employment Agreement was previously filed as Exhibit 10.4 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2011, and is incorporated by reference to this Exhibit 10.28.* | ||
10.29 | — | Agreement with Louis C. Camilleri (incorporated by reference to Exhibit 10.25 to the Registration Statement on Form 10 filed February 7, 2008).* | ||
10.30 | — | Amendment to Employment Agreement with Martin King (incorporated by reference to Exhibit 10.2 to the Quarterly Report on Form 10-Q for the quarter ended September 30, 2015). The Employment Agreement was previously filed as Exhibit 10.1 to the Quarterly Report on Form 10-Q for the quarter ended March 31, 2015, and is incorporated by reference to this Exhibit 10.30.* | ||
10.31 | — | Supplemental Letter to the Employment Agreement (as amended) with Miroslaw Zielinski (incorporated by reference to Exhibit 10.3 to the Quarterly Report on Form 10-Q for the quarter ended September 30, 2015). The Employment Agreement was previously filed as Exhibit 10.2 to the Quarterly Report on Form 10-Q for the quarter ended March 31, 2013 and is incorporated by reference to this Exhibit 10.31. The Amendment to the Employment Agreement was previously filed as Exhibit 10.28 to the Annual Report on Form 10-K for the year ended December 31, 2013, and is incorporated by reference to this Exhibit 10.31.* | ||
10.32 | — | Early Retirement and Release Agreement with Matteo Pellegrini (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed July 27, 2015).* | ||
136
10.33 | — | Time Sharing Agreement between PMI Global Services Inc. and Louis C. Camilleri dated August 18, 2010 (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed August 19, 2010).* | ||
10.34 | — | Amendment No. 1 to the Time Sharing Agreement between PMI Global Services Inc. and Louis C. Camilleri, dated August 22, 2012 (incorporated by reference to Exhibit 10.4 to the Quarterly Report on Form 10-Q for the quarter ended September 30, 2012).* | ||
10.35 | — | Amendment No. 2 to the Time Sharing Agreement between PMI Global Services Inc. and Louis C. Camilleri, dated October 23, 2012 (incorporated by reference to Exhibit 10.27 to the Annual Report on Form 10-K for the year ended December 31, 2012).* | ||
10.36 | — | Amendment No. 3 to the Time Sharing Agreement between PMI Global Services Inc. and Louis C. Camilleri, dated December 31, 2014 (incorporated by reference to Exhibit 10.34 to the Annual Report on Form 10-K for the year ended December 31, 2014).* | ||
10.37 | — | Time Sharing Agreement between PMI Global Services Inc. and André Calantzopoulos, dated May 8, 2013 (incorporated by reference to Exhibit 10.1 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2013).* | ||
10.38 | — | Amendment No. 1 to the Time Sharing Agreement between PMI Global Services Inc. and André Calantzopoulos, dated December 23, 2014 (incorporated by reference to Exhibit 10.36 to the Annual Report on Form 10-K for the year ended December 31, 2014).* | ||
10.39 | — | Amendment to the Employment Agreement with Jacek Olczak (incorporated by reference to Exhibit 10.33 to the Annual Report on Form 10-K for the year ended December 31, 2013). The Employment Agreement was previously filed as Exhibit 10.4 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2012, and is incorporated by reference to this Exhibit 10.39.* | ||
10.40 | — | Amended and Restated Supplemental Management Employees’ Retirement Plan (incorporated by reference to Exhibit 10.27 to the Annual Report on Form 10-K for the year ended December 31, 2008).* | ||
10.41 | — | Supplemental Equalization Plan, amended and restated as of June 29, 2015 (incorporated by reference to Exhibit 10.2 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2015).* | ||
10.42 | — | Form of Supplemental Equalization Plan Employee Grantor Trust Enrollment Agreement (Secular Trust) (incorporated by reference to Exhibit 10.31 to the Annual Report on Form 10-K for the year ended December 31, 2008).* | ||
10.43 | — | Form of Supplemental Equalization Plan Employee Grantor Trust Enrollment Agreement (Executive Trust) (incorporated by reference to Exhibit 10.32 to the Annual Report on Form 10-K for the year ended December 31, 2008).* | ||
10.44 | — | Philip Morris International Inc. Form of Indemnification Agreement with Directors and Executive Officers (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed September 18, 2009).* | ||
10.45 | — | Form of Deferred Stock Agreement (2013 Grants) (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed February 12, 2013).* | ||
10.46 | — | Form of Deferred Stock Agreement (2014 Grants) (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed February 7, 2014).* | ||
10.47 | — | Form of Deferred Stock Agreement (2015 Grants) (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed February 10, 2015).* | ||
10.48 | — | Philip Morris International Inc. Tax Return Preparation Services Policy (incorporated by reference to Exhibit 10.51 to the Annual Report on Form 10-K for the year ended December 31, 2014).* | ||
10.49 | — | Form of Restricted Stock Unit Agreement (2016 Grants) (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed February 9, 2016).* | ||
10.50 | — | Form of Performance Share Unit Agreement (2016 Grants) (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed February 9, 2016).* | ||
137
10.51 | — | Form of Restricted Stock Unit Agreement (2017 Grants) (incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K filed February 7, 2017).* | ||
10.52 | — | Form of Performance Share Unit Agreement (2017 Grants) (incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K filed February 7, 2017).* | ||
12 | — | Statement regarding computation of ratios of earnings to fixed charges. | ||
18 | — | Letter from PricewaterhouseCoopers SA, Independent Registered Accounting Firm of the Registrant, Regarding Change in Accounting Principle (incorporated by reference to Exhibit 18 to the Quarterly Report on Form 10-Q for the quarter ended June 30, 2016). | ||
21 | — | Subsidiaries of Philip Morris International Inc. | ||
23 | — | Consent of independent registered public accounting firm. | ||
24 | — | Powers of attorney. | ||
31.1 | — | Certification of the Registrant’s Chief Executive Officer pursuant to Rule 13a-14(a)/15d-14(a) of the Securities Exchange Act of 1934, as amended, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | ||
31.2 | — | Certification of the Registrant’s Chief Financial Officer pursuant to Rule 13a-14(a)/15d-14(a) of the Securities Exchange Act of 1934, as amended, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | ||
32.1 | — | Certification of the Registrant’s Chief Executive Officer pursuant to 18 U.S.C. 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | ||
32.2 | — | Certification of the Registrant’s Chief Financial Officer pursuant to 18 U.S.C. 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | ||
101.INS | — | XBRL Instance Document. | ||
101.SCH | — | XBRL Taxonomy Extension Schema. | ||
101.CAL | — | XBRL Taxonomy Extension Calculation Linkbase. | ||
101.DEF | — | XBRL Taxonomy Extension Definition Linkbase. | ||
101.LAB | — | XBRL Taxonomy Extension Label Linkbase. | ||
101.PRE | — | XBRL Taxonomy Extension Presentation Linkbase. | ||
* | Denotes management contract or compensatory plan or arrangement in which directors or executive officers are eligible to participate. |
138
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
PHILIP MORRIS INTERNATIONAL INC. | |
By: | /s/ ANDRÉ CALANTZOPOULOS |
(André Calantzopoulos Chief Executive Officer) | |
Date: February 14, 2017
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the date indicated:
Signature | Title | Date |
/s/ ANDRÉ CALANTZOPOULOS | Chief Executive Officer | February 14, 2017 |
(André Calantzopoulos) | ||
/s/ JACEK OLCZAK | Chief Financial Officer | February 14, 2017 |
(Jacek Olczak) | ||
/s/ ANDREAS KURALI | Vice President and Controller | February 14, 2017 |
(Andreas Kurali) | ||
*HAROLD BROWN, LOUIS C. CAMILLERI, MASSIMO FERRAGAMO, WERNER GEISSLER, JENNIFER LI, JUN MAKIHARA, SERGIO MARCHIONNE, KALPANA MORPARIA, LUCIO A. NOTO, ROBERT B. POLET, STEPHEN M. WOLF | Directors | |
*By: | /s/ ANDRÉ CALANTZOPOULOS | February 14, 2017 | |
(André Calantzopoulos Attorney-in-fact) | |||
139
