Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Impax Laboratories, LLC | ipxl-01x11x2017x8k.htm |

1
35th Annual J.P. Morgan
Healthcare Conference
January 11, 2017

2
Impax Cautionary Statement Regarding Forward
Looking Statements
"Safe Harbor" statement under the Private Securities Litigation Reform Act of 1995:
To the extent any statements made in this news release contain information that is not historical; these statements are forward-looking in nature
and express the beliefs and expectations of management. Such statements are based on current expectations and involve a number of known
and unknown risks and uncertainties that could cause the Company’s future results, performance, or achievements to differ significantly from
the results, performance, or achievements expressed or implied by such forward-looking statements. Such risks and uncertainties include, but
are not limited to: fluctuations in revenues and operating income; the Company’s ability to successfully develop and commercialize
pharmaceutical products in a timely manner; reductions or loss of business with any significant customer; the substantial portion of the
Company’s total revenues derived from sales of a limited number of products; the impact of consolidation of the Company’s customer base; the
impact of competition; the Company’s ability to sustain profitability and positive cash flows; any delays or unanticipated expenses in connection
with the operation of the Company’s manufacturing facilities; the effect of foreign economic, political, legal, and other risks on the Company’s
operations abroad; the uncertainty of patent litigation and other legal proceedings; the increased government scrutiny on the Company’s
agreements with brand pharmaceutical companies; product development risks and the difficulty of predicting FDA filings and approvals;
consumer acceptance and demand for new pharmaceutical products; the impact of market perceptions of the Company and the safety and
quality of the Company’s products; the Company’s determinations to discontinue the manufacture and distribution of certain products; the
Company’s ability to achieve returns on its investments in research and development activities; changes to FDA approval requirements; the
Company’s ability to successfully conduct clinical trials; the Company’s reliance on third parties to conduct clinical trials and testing; the
Company’s lack of a license partner for commercialization of NUMIENTTM (IPX066) outside of the United States; impact of illegal distribution
and sale by third parties of counterfeits or stolen products; the availability of raw materials and impact of interruptions in the Company’s supply
chain; the Company’s policies regarding returns, allowances and chargebacks; the use of controlled substances in the Company’s products; the
effect of current economic conditions on the Company’s industry, business, results of operations and financial condition; disruptions or failures
in the Company’s information technology systems and network infrastructure caused by third party breaches or other events; the Company’s
reliance on alliance and collaboration agreements; the Company’s reliance on licenses to proprietary technologies; the Company’s dependence
on certain employees; the Company’s ability to comply with legal and regulatory requirements governing the healthcare industry; the regulatory
environment; the effect of certain provisions in the Company’s government contracts; the Company’s ability to protect its intellectual property;
exposure to product liability claims; risks relating to goodwill and intangibles; changes in tax regulations; the Company’s ability to manage
growth, including through potential acquisitions and investments; the risks related to the Company’s acquisitions of or investments in
technologies, products or businesses; the restrictions imposed by the Company’s credit facility and indenture; the Company’s level of
indebtedness and liabilities and the potential impact on cash flow available for operations; uncertainties involved in the preparation of the
Company’s financial statements; the Company’s ability to maintain an effective system of internal control over financial reporting; the effect of
terrorist attacks on the Company’s business; the location of the Company’s manufacturing and research and development facilities near
earthquake fault lines; expansion of social media platforms and other risks described in the Company’s periodic reports filed with the Securities
and Exchange Commission. Forward-looking statements speak only as to the date on which they are made, and the Company undertakes no
obligation to update publicly or revise any forward-looking statement, regardless of whether new information becomes available, future
developments occur or otherwise.
Trademarks referenced herein are the property of their respective owners.
©2017 Impax Laboratories, Inc. All Rights Reserved.

3
Impax at a Glance
Generic Division
Targeting high-value solid oral and alternative
dosage form ANDAs that are difficult to develop
• Diversified commercial portfolio of ~72 products
• 25 ANDAs pending approval at FDA
• 26 ANDAs under development
Specialty Pharma Division
Focused on developing branded Central Nervous
System disorder and other specialty products
• 5 commercialized products
• 1 under development
Pipeline data as of Dec. 31, 2016
A specialty pharmaceutical company applying its formulation expertise and
drug delivery technology to the development, manufacturing and marketing of
specialty generics and branded products

4
Leveraging Our Strengths and Focusing on
Opportunities for Growth
• Select the right products for internal and partnership development
• Continue focus on sustainable quality and compliance programs
• Develop, file and launch on time and capitalize on market disruptions
• Ensure effective and compliant marketing and sales execution of brand
and generic products
Leveraging
Our Strengths
• Increase productivity from internal R&D programs
• Diversify via select partnering and business development activities in both
brand and generic businesses
• Improve cost of goods sold and supply chain flexibility
Focusing on
Opportunities
for Growth
• Increased rate of ANDA approvals by FDA – though also an opportunity
• Aggressive competition and pricing activities
• Consolidation of payers and customers
Current
Challenges

5
Aligning Organization and Resources to Support
Growth Initiatives
Completed/In-Process
Estimated Annual Savings
~$41MM-$47MM*
Operational and R&D
restructuring (2015)
Closed central packaging
operation/transferred distribution
to UPS (2015)
Closing Middlesex, NJ
manufacturing and packaging
facility (2017)
Evaluating Additional
Efficiency Opportunities to
Free Up Resources to Invest
in Growth Initiatives and
Reduce Long-Term Debt
*Based on estimates as of Dec. 31, 2016. Currently expect to realize full annual savings beginning in 2018.

6
Diversified Generic Portfolio of Solid Oral and
Alternative Dose Form Products
13
50
9
Commercialized Portfolio of
72 Products
Controlled Release SOD
Other SOD
Alternative Dose
SOD – Solid Oral Dose; Data as of Dec. 31, 2016
Product Portfolio by Number of Competing
Generic Products
Sole player in market 10%
One 13%
Two 21%
Three 18%
Four or more 38%
62% of Impax’s commercialized portfolio
have three or fewer competing products

7
Growing Epinephrine Auto-Injector
0%
1%
2%
3%
4%
5%
6%
7%
8%
9%
10%
2
0
1
6
/0
1
/0
1
2
0
1
6
/0
1
/0
8
2
0
1
6
/0
1
/1
5
2
0
1
6
/0
1
/2
2
2
0
1
6
/0
1
/2
9
2
0
1
6
/0
2
/0
5
2
0
1
6
/0
2
/1
2
2
0
1
6
/0
2
/1
9
2
0
1
6
/0
2
/2
6
2
0
1
6
/0
3
/0
4
2
0
1
6
/0
3
/1
1
2
0
1
6
/0
3
/1
8
2
0
1
6
/0
3
/2
5
2
0
1
6
/0
4
/0
1
2
0
1
6
/0
4
/0
8
2
0
1
6
/0
4
/1
5
2
0
1
6
/0
4
/2
2
2
0
1
6
/0
4
/2
9
2
0
1
6
/0
5
/0
6
2
0
1
6
/0
5
/1
3
2
0
1
6
/0
5
/2
0
2
0
1
6
/0
5
/2
7
2
0
1
6
/0
6
/0
3
2
0
1
6
/0
6
/1
0
2
0
1
6
/0
6
/1
7
2
0
1
6
/0
6
/2
4
2
0
1
6
/0
7
/0
1
2
0
1
6
/0
7
/0
8
2
0
1
6
/0
7
/1
5
2
0
1
6
/0
7
/2
2
2
0
1
6
/0
7
/2
9
2
0
1
6
/0
8
/0
5
2
0
1
6
/0
8
/1
2
2
0
1
6
/0
8
/1
9
2
0
1
6
/0
8
/2
6
2
0
1
6
/0
9
/0
2
2
0
1
6
/0
9
/0
9
2
0
1
6
/0
9
/1
6
2
0
1
6
/0
9
/2
3
2
0
1
6
/0
9
/3
0
2
0
1
6
/1
0
/0
7
2
0
1
6
/1
0
/1
4
2
0
1
6
/1
0
/2
1
2
0
1
6
/1
0
/2
8
2
0
1
6
/1
1
/0
4
2
0
1
6
/1
1
/1
1
2
0
1
6
/1
1
/1
8
2
0
1
6
/1
1
/2
5
2
0
1
6
/1
2
/0
2
2
0
1
6
/1
2
/0
9
2
0
1
6
/1
2
/1
6
2
0
1
6
/1
2
/2
3
Share Growth in 2016
4%
9%
• Continuing to expand production/supply
• Strong performance during peak seasonal demand
• Exceeding 2016 goal of 1% share growth per quarter
• Awareness campaign “The GO-TO-CHOICE”
› Media, website, physicians, pharmacies
IMS NPA Weekly Dec. 23, 2016

8
Continuing to Expand Generic Pipeline
Source of sales data: IMS NPA Nov. 2016; *U.S. Brand/Generic market sales; Pipeline data as of Dec. 31, 2016
Portfolio of 51 Products Targeting
Current U.S. Brand/Generic Market of $24B
Solid Oral
Dose
Products
Alternative
Dose
Products
Total
Products
Potential # of
Products
First-to-File or
First-to-Market
$
Value*
Pending at FDA 17 8 25 10 $14B
Under
Development
19 7 26 21 $10B
Total Pipeline 36 15 51 31 $24B

9
ANDA Pipeline Includes Several Potential High-Value
First-to-Market Opportunities
Source of sales data: IMS NPA Nov. 2016; Pipeline data as of Dec. 31, 2016; TA = tentative approval
1 Launched authorized generic in April 2016
2 Assuming final FDA approval, earliest potential launch date/timing based on settlement or patent expiration date
Disclosed Pending ANDAs and Tentative Approval Products
Greneric Product Name Brand
IMS
Sales
Potential Launch
Timing
FTM
Opportunity
Oxycodone ER tablet (new
formulation) 1
OxyContin® $2.3B Settled, not disclosed
Sevelamer Carbonate IR tablet Renvela® $1.9B Approval
Methylphenidate HCI ER tablet Concerta® $1.8B Approval
Teriflunomide IR tablet Aubagio® $1.1B Pending litigation
Ezetimibe/Simvastatin IR tablet – TA 2 Vytorin® $691M April 2017
Colesevelam IR tablet Welchol® $637M Approval
Oxymorphone ER tablet (new
formulation)
Opana ER® $308M Pending litigation
Aspirin/Dipyridamole ER capsule - TA Aggrenox® $243M Approval
Fentanyl Buccal IR tablet Fentora® $148M Settled, not disclosed
Dutasteride/Tamsulosin IR capsule Jalyn® $50M Approval
Risedronate Sodium DR tablet Atelvia® $34M Approval

10
Product Therapeutic Area
Parkinson’s Disease
Migraine
Anthelmintic
Anthelmintic
Specialty Pharma Portfolio

11
Actions Taken to Deliver Growth
0.0%
1.0%
2.0%
3.0%
0
2,000
4,000
6,000
8,000
10,000
12,000
14,000
CD
L
D
S
h
a
re
T
R
x
Monthly TRx and Share of National CD-LD TRx Since Launch
Rytary TRx CD-LD Share
• Redefined clinical messaging related to formulation
• Simplified dose conversion guidance for general neurologists
• Launched myRytary Patient Support Program www.myrytary.com
Source: IMS NPA Monthly Dec. 2016

12
Patient Support Program Introduced
• Third-party managed
• Patient specific case manager
• Prior authorization status updates
• Appeals support
• Commercial copay assistance support
• Trial product vouchers
• Alternative funding referrals
• Patient assistance program application
support
• Educational resources
www.myrytary.com
13
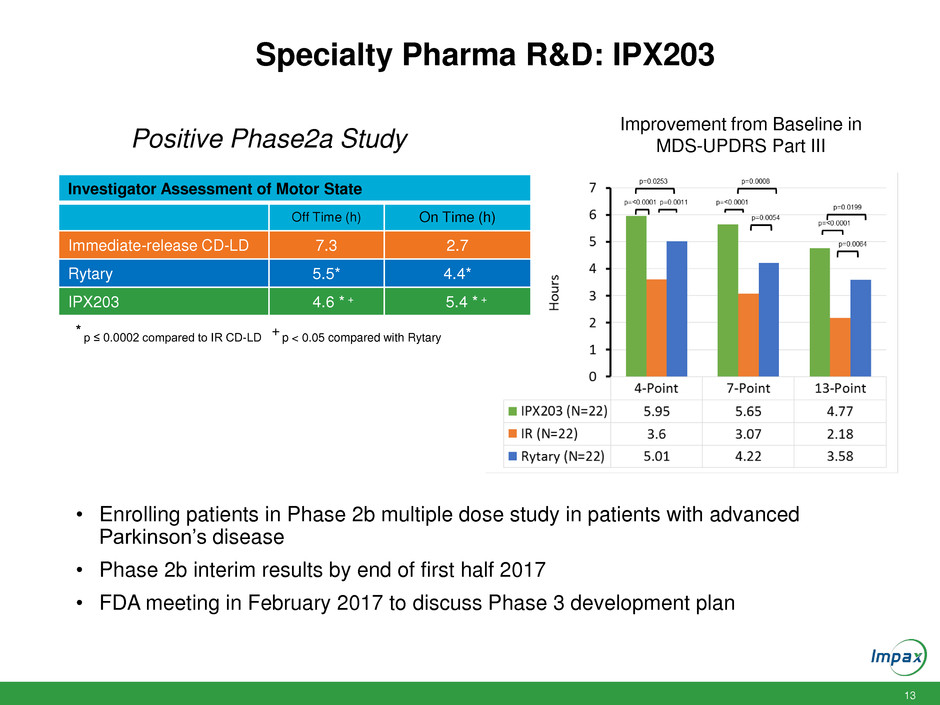
Specialty Pharma R&D: IPX203
Investigator Assessment of Motor State
Off Time (h) On Time (h)
Immediate-release CD-LD 7.3 2.7
Rytary 5.5* 4.4*
IPX203 4.6 * + 5.4 * +
* p ≤ 0.0002 compared to IR CD-LD + p < 0.05 compared with Rytary
Improvement from Baseline in
MDS-UPDRS Part III
• Enrolling patients in Phase 2b multiple dose study in patients with advanced
Parkinson’s disease
• Phase 2b interim results by end of first half 2017
• FDA meeting in February 2017 to discuss Phase 3 development plan
Positive Phase2a Study

14
Positive Performance Trend
0%
10%
20%
30%
40%
50%
2,000
4,000
6,000
8,000
10,000
12,000
14,000
A
pr
-1
2
J
u
n
-1
2
A
u
g
-1
2
O
c
t-
1
2
D
e
c
-1
2
F
e
b
-1
3
A
pr
-1
3
J
u
n
-1
3
A
u
g
-1
3
O
c
t-
1
3
D
e
c
-1
3
F
e
b
-1
4
A
pr
-1
4
J
u
n
-1
4
A
u
g
-1
4
O
c
t-
1
4
D
e
c
-1
4
F
e
b
-1
5
A
pr
-1
5
J
u
n
-1
5
A
u
g
-1
5
O
c
t-
1
5
D
e
c
-1
5
F
e
b
-1
6
A
pr
-1
6
J
u
n
-1
6
A
u
g
-1
6
O
c
t-
1
6
D
e
c
-1
6
Nas
a
l
T
rip
ta
n
S
h
a
re
T
R
x
Monthly TRx and Share of Nasal Triptan TRx Since Assuming Product Promotion
TRx Nasal Triptan Share
• Emphasis on second position promotion
• Increased non-personal promotion
• Solid growth from general neurologists, headache specialists and pediatricians
Source: IMS NPA Monthly Dec 2016; Nasal Segment Imitrex, Sumatriptan, Onzetra Xsail (launched May 2016) and ZOMIG Nasal Spray
15
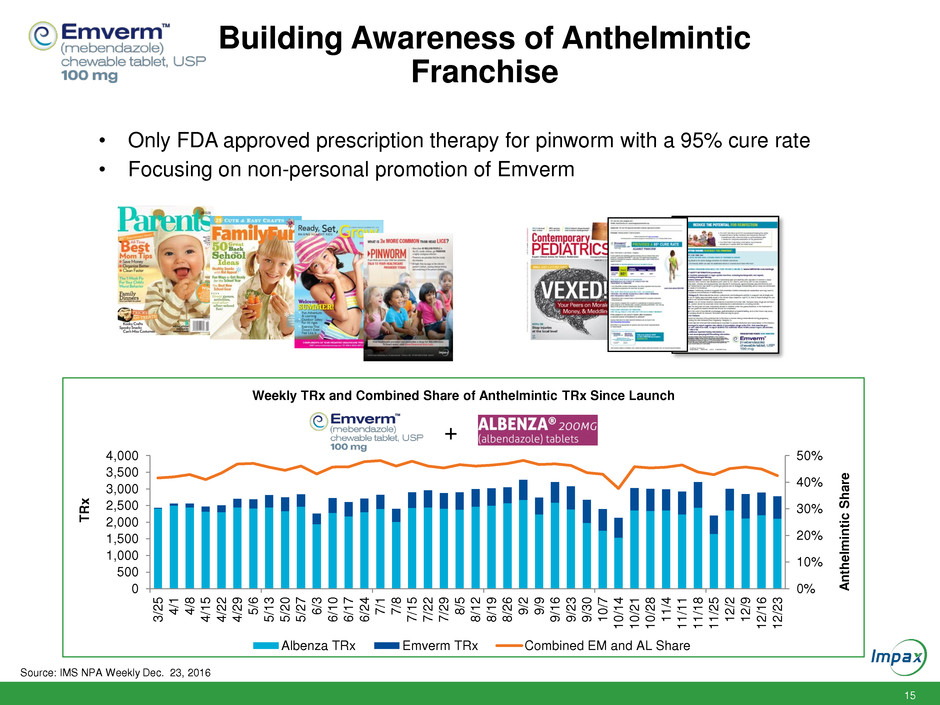
Building Awareness of Anthelmintic
Franchise
0%
10%
20%
30%
40%
50%
0
500
1,000
1,500
2,000
2,500
3,000
3,500
4,000
3
/2
5
4
/1
4
/8
4
/1
5
4
/2
2
4
/2
9
5
/6
5
/1
3
5
/2
0
5
/2
7
6
/3
6
/1
0
6
/1
7
6
/2
4
7
/1
7
/8
7
/1
5
7
/2
2
7
/2
9
8
/5
8
/1
2
8
/1
9
8
/2
6
9
/2
9
/9
9
/1
6
9
/2
3
9
/3
0
1
0
/7
1
0
/1
4
1
0
/2
1
1
0
/2
8
1
1
/4
1
1
/1
1
1
1
/1
8
1
1
/2
5
1
2
/2
1
2
/9
1
2
/1
6
1
2
/2
3
A
n
th
elmin
tic
S
h
a
re
T
R
x
Weekly TRx and Combined Share of Anthelmintic TRx Since Launch
Albenza TRx Emverm TRx Combined EM and AL Share
• Only FDA approved prescription therapy for pinworm with a 95% cure rate
• Focusing on non-personal promotion of Emverm
Source: IMS NPA Weekly Dec. 23, 2016
+

16
Current Priorities
Focus on quality and operations
Maximize profitability within generic portfolio
Effectively execute on Rytary® and Emverm® growth initiatives
Capitalize on new generic product launches
Continue to build diversification within our portfolio
Explore additional expense savings and efficiency opportunities

17
Investment Thesis
Pipeline data as of Dec. 31, 2016 Source of sales data: IMS Nov. 2016; *U.S. Brand/Generic market sales;
• Generic pipeline of 51 products targeting $24B U.S. sales*
• Specialty pipeline focused on next generation opportunities
• Solid platform on which to build long-term growth
Targeting
Sustainable
Generic and
Specialty
Pharma
Markets
• Generic and Specialty Pharma commercialization success
• Track record of complex formulation and development
• Hatch-Waxman expertise and Paragraph IV successes
Established
Core
Competencies
• Track record of profitability and balance sheet to support growth
• Diversifying and building scale organically and through business development
• Demonstrated ability to integrate product and company acquisitions
Flexible
Financial
Profile
