Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - NANOSPHERE INC | d88768d8k.htm |
Exhibit 99.1
Issuer Free Writing Prospectus
Filed pursuant to Rule 433
Registration No. 333-207460
December 10, 2015
NANOSPHERE
COMPANY OVERVIEW
Nanosphere, Inc. (the “Company”) has filed a registration statement (including a preliminary prospectus) with the U.S. Securities and Exchange Commission (the “SEC”) for the offering to which this presentation relates. Before you invest, you should read the prospectus in that registration statement and other documents the Company has filed with the SEC for more completion information about the Company and this offering. You may obtain these documents free of charge by visiting the SEC’s online database (EDGAR) on the SEC’s website at www.sec.gov. The Company’s registration statement on Form S-1 containing the preliminary prospectus, dated December 10, 2015, is available at the following link:
http://www.sec.gov/Archives/edgar/data/1105184/000119312515399534/0001193125-15-399534-index.htm
Alternatively, you may obtain a copy of the preliminary prospectus and any other publicly available documents from the Company’s website at http://www.nanosphere.us/investors/sec-filings or from H.C. Wainwright & Co. at placements@hcwco.com or by calling 212-356-0500.
|
|
NANOSPHERE COMPANY OVERVIEW
FORWARD-LOOKING STATEMENTS
historical This presentation facts, included contains in forward-looking this presentation statements regarding our about strategy, us and future our industry operations, that involve future financial substantial position, risks and future uncertainties. net sales, projected All statements, expenses, other products’ than statements placements, of in performance particular and and the acceptance, relative performance prospects and of other plans and market management’s participants objectives, are forward-looking as well as statements. the growth of These the overall statements market involve for our known products and unknown in general risks, and certain uncertainties products and other looking factors statements. that may cause our actual results, levels of activity, performance or achievement to be materially different from those expressed or implied by the forward-“would,” In some cases, “should,” you can “could,” identify “can,” forward-looking “predict,” “potential,” statements “continue,” by terms “objective,” such as “anticipate,” or the negative “believe,” of these “estimate,” terms, “expect,” and similar “intend,” expressions “may,” intended “might,” to “plan,” identify “project,” forward-looking “will,” are statements. based on However, assumptions not and all forward-looking subject to risks statements and uncertainties. contain Given these these identifying uncertainties, words. These you should forward-looking not place undue statements reliance reflect on these our current forward-looking views about statements. future events Actual and events or results could differ materially from those expressed or implied by these forward-looking statements as a result of various factors.
These update forward-looking any of these forward-looking statements represent statements our to estimates reflect circumstances and assumptions or events only as that of the occur date after hereof. the statement Unless required is made by or U. to S. federal conform securities these statements laws, we do to not actual intend results. to subsequent The following quarterly presentation reports should on Form be viewed 10-Q. Our in conjunction actual results with may the differ consolidated materially financial from those statements anticipated and notes in these thereto forward-looking appearing in statements our Annual as Report a result on of Form various 10-K factors, and including but not limited to:
| • |
|
or inaccurate failure of estimates the market of for the these potential products market to size grow for as our anticipated, products (including the hospital lab market in general and the blood stream infection (BSI) market in particular) the predictive past performance of our future of results other companies in the manner (such we as believe Cepheid) them which to be, we believe to have been in a market position analogous to where we believe we are now may not be our meningitis analysis MDx of who product our competitors markets) and have our been, predictions who they of relevant are now future and who performance they will be may in the be inaccurate, future (particularly in the BSI, enteric, extended tuberculosis and comparisons of actual financial results for another company to what we predict will be our future financial results may be inapposite, |
| • |
|
predictions of when “breakeven customer base” is achieved and its relationship to our cash flow position, needs and “burn” may prove to be inaccurate, |
| • |
|
entrance of other competitors or other factors causing us to lose competitive advantage in the sample-to-result MDx market, |
| • |
|
a lack of commercial acceptance of the Verigene System, its array of tests, and the development of additional tests, which could negatively affect our financial results, |
| • |
|
failure ability to of sell third-party our products, payors to reimburse our customers for the use of our clinical diagnostic products or reduction of reimbursement levels, which could harm our which failure could of our cause products lost revenue, to perform delayed as expected or reduced or to obtain market certain acceptance approvals of our or products, the questioning increased of the costs reliability and damage of the to technology our reputation, on which our products are based, our inability to manage our anticipated growth, constraints or inefficiencies caused by unanticipated acceleration and deceleration of customer demand, and |
| 2 |
|
| • |
|
those set forth under “Risk Factors” in our Annual Report on Form 10-K. |
|
|
NANOSPHERE COMPANY OVERVIEW
OUR MISSION
Enhance Medicine Through Targeted & Flexible Diagnostics
| • |
|
Improve Patient Care |
o Save lives o Decrease morbidity o Reduce spread of antibiotic resistance
| • |
|
Lower Costs to the Healthcare System |
o Shorter length of stay o Antibiotic savings o Demonstrated economic outcomes o Minimize unnecessary testing
| • |
|
Deliver Exceptional Customer Service |
o Value to lab / clinician / administration o Customer-based development
|
|
NANOSPHERE COMPANY OVERVIEW
WHY WE BELIEVE WE WILL SUCCEED
Installed Base
o Over 300 U.S. microbiology customer placements and growing o Leverage through expanding menu
Reliable and Versatile Technology
o Clinical evidence validates exceptional performance o Detection of organism and drug resistance, proteins o Flexible reporting enables lab to respond to clinician needs
Financial Strengths
o Flex Pricing to address customer demand o Competitive cost advantage
|
|
NANOSPHERE COMPANY OVERVIEW
TWO MAIN DRIVERS OF MDX MARKETS
Need for more timely Need to address Need and cost-effective costly and complex results disease states
High Count Sample-To-
Product Feature Multi-
Result
Plexing
Conversion of traditional culture-based methods to molecular will be driven by the need for rapid and clinically actionable results that will either improve outcomes or improve economics
ACCOUNTABLE CARE
|
|
NANOSPHERE COMPANY OVERVIEW
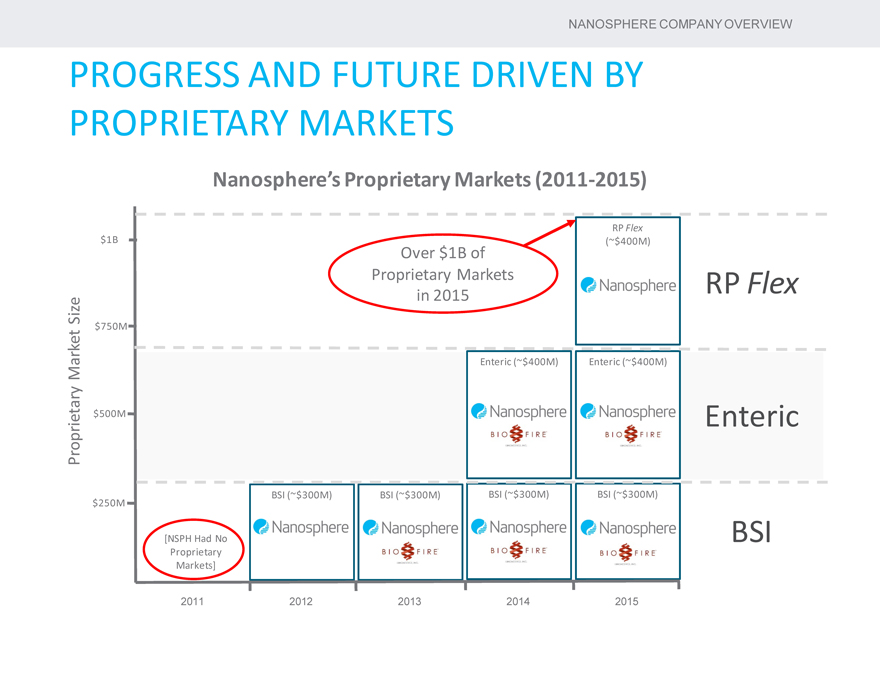
PROGRESS AND FUTURE DRIVEN BY PROPRIETARY MARKETS
Nanosphere’s Proprietary Markets (2011-2015)
RP Flex
$1B (~$400M)
Over $1B of
Proprietary Markets RP Flex Size in 2015
$750M
Market Enteric (~$400M) Enteric (~$400M)
Proprietary $500M Enteric
BSI (~$300M) BSI (~$300M) BSI (~$300M) BSI (~$300M) $250M
[NSPH Had No BSI Proprietary Markets]
2011 2012 2013 2014 2015
|
|
NANOSPHERE COMPANY OVERVIEW
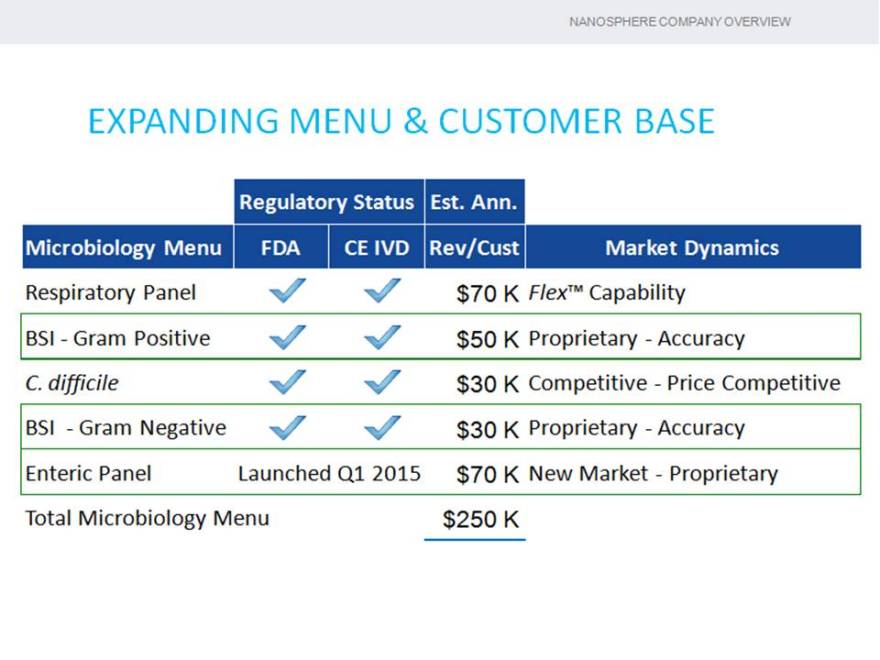
EXPANDING MENU & CUSTOMER BASE
Regulatory Status Est. Ann.
Microbiology Menu FDA CE IVD Rev/Cust Market Dynamics
Respiratory Panel $70 K Flex™ Capability
BSI—Gram Positive $50 K Proprietary—Accuracy
C. difficile $30 K Competitive—Price Competitive
BSI—Gram Negative $30 K Proprietary—Accuracy
Enteric Panel Launched Q1 2015 $70 K New Market—Proprietary
Total Microbiology Menu $250 K
|
|
NANOSPHERE COMPANY OVERVIEW
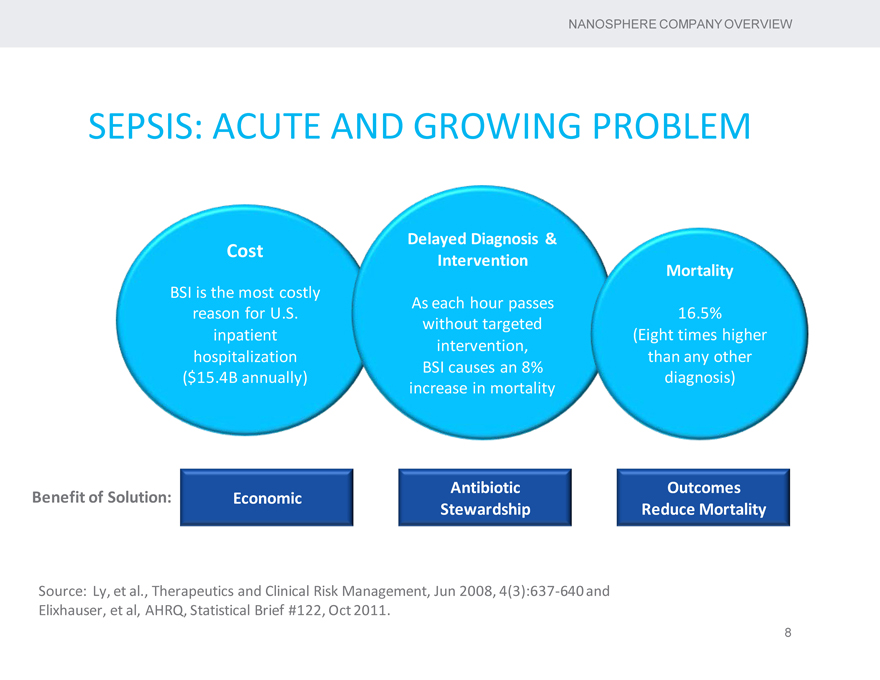
SEPSIS: ACUTE AND GROWING PROBLEM
Delayed Diagnosis &
Cost
Intervention
Mortality
BSI is the most costly As each hour passes reason for U.S. 16.5% without targeted inpatient (Eight times higher intervention, hospitalization than any other BSI causes an 8%
($15.4B annually) diagnosis) increase in mortality
Antibiotic Outcomes Benefit of Solution: Economic Stewardship Reduce Mortality
Source: Ly, et al., Therapeutics and Clinical Risk Management, Jun 2008, 4(3):637-640 and Elixhauser, et al, AHRQ, Statistical Brief #122, Oct 2011.
| 8 |
|
|
|
NANOSPHERE COMPANY OVERVIEW
MICROBIOLOGY TEST REQUIREMENTS
| • |
|
Targeted AND Multiplexed (Flexible) |
o Multiple infectious target identification & resistance o Clinically actionable result
| • |
|
Accurate |
o >95% sensitivity o >95% specificity
| • |
|
Fast |
o Rapid turnaround time
| • |
|
Easy to Use |
o Sample-to-result
| • |
|
On Demand |
o Random access
| • |
|
Moderate Complexity |
9
|
|
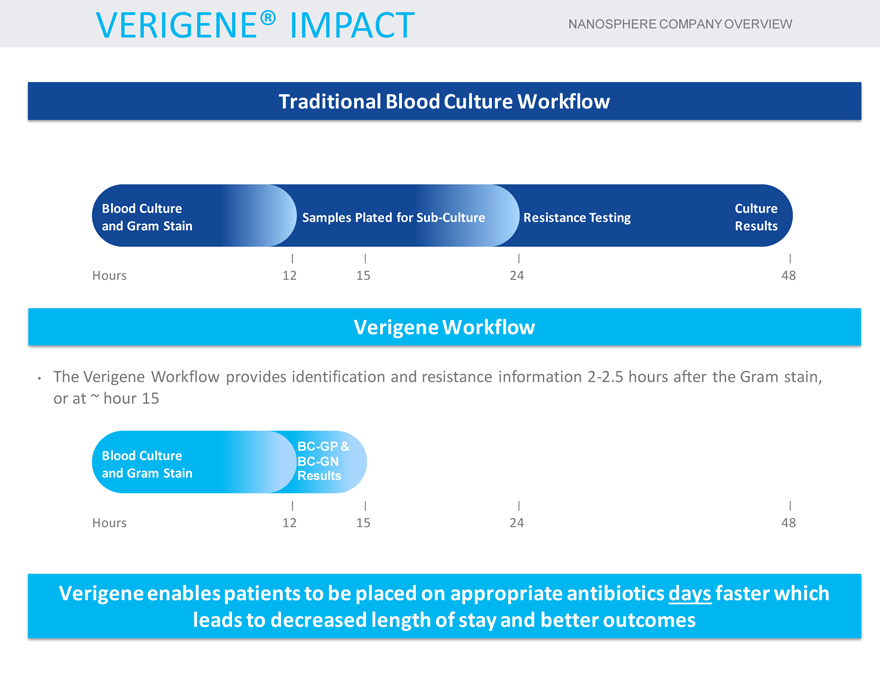
VERIGENE® IMPACT NANOSPHERE COMPANY OVERVIEW
Traditional Blood Culture Workflow
Blood Culture Culture Samples Plated for Sub-Culture Resistance Testing and Gram Stain Results
Hours 12 15 24 48
Verigene Workflow
| • |
|
The Verigene Workflow provides identification and resistance information 2-2.5 hours after the Gram stain, or at ~ hour 15 |
BC-GP &
Blood Culture BC-GN and Gram Stain Results
Hours 12 15 24 48
Verigene enables patients to be placed on appropriate antibiotics days faster which leads to decreased length of stay and better outcomes
|
|
NANOSPHERE COMPANY OVERVIEW
VERIGENE CHANGES THE GAME
Saves Last Line
Saves Money Therapies
Saves Lives
Recent study by UF Shands New Hanover Regional
Medical Center: 80% reduction in
| 21.7 |
|
day reduction in 60 hour reduction to I.C.U. mortality |
Length of stay/patient discontinuation of from 47.8%
unnecessary therapy to 9.5%
$61,000 reduction in and $115K annual
cost/patient pharmacy savings
Benefit of Solution: Economic Antibiotic Outcomes
Stewardship Reduced Mortality
Sources: Sango, et al. A stewardship approach to optimize antimicrobial therapy through use of a rapid microarray assay on blood cultures positive with
Enterococcus species. J. Clinical Microbiology, 2013 September 23, 01951-13.
McNabb et al. Results of Rapid Identification of Gram-Positive Bacteria Combined with Rapid Pharmacy Notification and Treatment in a 11
Community Hospital Healthcare System. Poster presented at 2015 Making A Difference in Infectious Diseases (MAD-ID) Conference in
Orlando, Florida.
|
|
NANOSPHERE COMPANY OVERVIEW
VERIGENE IMPACT IN COMMUNITY HOSPITALS
Scripps Memorial Hospital1
| • |
|
25.3 hour reduction in time to optimal therapy |
| • |
|
5.1 day length of stay reduction |
| • |
|
$10,547 reduction in hospital costs per patient |
Blount Memorial Hospital2
| • |
|
21.0 hour reduction in time to optimal therapy |
| • |
|
3.5-day ICU length of stay reduction |
| • |
|
$600,000 cost savings per year |
1IDWeek 2014 Poster 2ICAAC 2014 Poster
|
|
COMPETITIVE ADVANTAGES NANOSPHERE COMPANY OVERVIEW
Rapid Blood Culture Testing Benefits Versus BioFire
Nanosphere direct detection (no PCR) provides for ?Reduce Mortality: Each hour that appropriate accuracy in both sensitivity and specificity required for antimicrobial treatment is delayed, a sepsis patient’s critical therapeutic change mortality rate increases by 7.6% (1)
| • |
|
BioFire’s PCR-based approach results in higher risk of ?Reduce Hospitalization Costs: Implementing rapid results false positives as advised to their customers in the reporting for S. aureus blood cultures can lead to an below Field Safety Corrective Action average 6.2-day reduction in length of stay and a $21,387 ? Clinically relevant design: assay designed to be based on reduction in costs per S. aureus-infected patient (2) gold standard Gram stain Stewardship: Rapid BioFire charges for results not indicated by gold ?Antimicrobial mecA reporting for standard results in an additional cost of ~ $50/test patients with S. aureus bacteremia results in a 25.4-hour reduction in the time to optimal antimicrobial therapy (3) |
Reduce the Impact of Contaminants: Patients with false-positive blood culture results triggered by contaminants such as S. epidermidis have hospitalization costs $8,750 higher than true negative blood culture patients (4)
(1) Kumar et al. 2006. Crit Care Med, 34:1589-96. (2) Bauer et al. 2010. Clin Infect Dis, 51:1074-80. (3) Carver et al. 2008. J Clin Microbiol, 46:2381-83. (4) Zwang and Albert 2006. J Hosp Med, 1:272-76.
|
|
NANOSPHERE COMPANY OVERVIEW
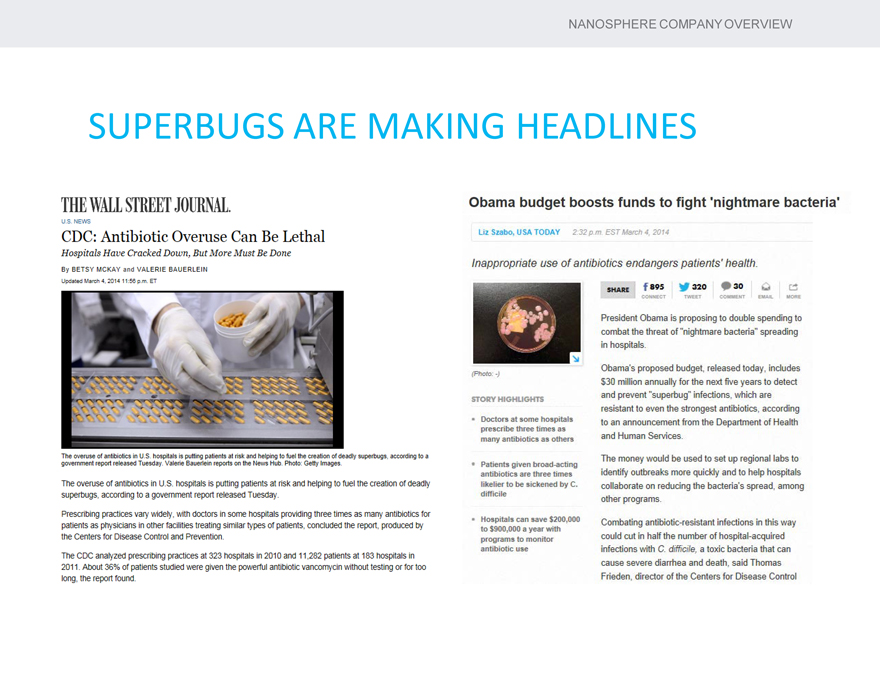
SUPERBUGS ARE MAKING HEADLINES
|
|
NANOSPHERE COMPANY OVERVIEW
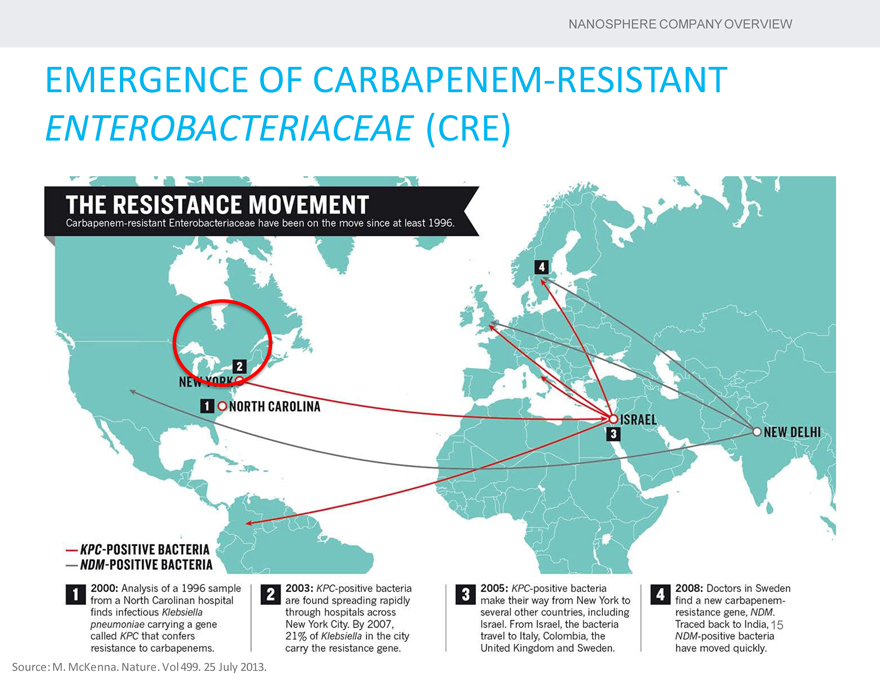
EMERGENCE OF CARBAPENEM-RESISTANT
ENTEROBACTERIACEAE (CRE)
15
Source: M. McKenna. Nature. Vol 499. 25 July 2013.
|
|
NANOSPHERE COMPANY OVERVIEW
CDC LANDMARK RECOMMENDATIONS
| • |
|
Burden and Threat Posed by Antibiotic-Resistant Infections |
o Drug resistant infections are a threat to human and economic health o Overuse of antibiotics is the single most important factor o > 10 million deaths per year by 2050 due to antimicrobial resistance1 o Urgent action is needed NOW by everyone who uses antibiotics
| • |
|
Four Core Actions Must Be Taken |
o Avoid infections o Track infections o Improve antibiotic stewardship and use o Development and implementation of drugs and DIAGNOSTIC TESTS
| • |
|
Nanosphere |
o Right place (point-of-need) o Right time (faster than conventional methods) o Right results (ONLY test on market to detect top 5 genes associated with CRE)
| (1) |
|
The Review on Antimicrobial Resistance: Commissioned Report by the UK Government and the Wellcome Trust |
|
|
NANOSPHERE COMPANY OVERVIEW
SOLVING THE ENTERIC PROBLEM
| • |
|
Large Market Opportunity |
o 3.7 million U.S. emergency department visits in 2010 for unknown gastrointestinal symptoms of bacterial or viral pathogens o 1.3 million resulted in hospitalizations
| • |
|
Costly to the Healthcare System |
o U.S. patients suffering from gastrointestinal infections cost the healthcare system over $6.0 billion o $1.8 billion in inpatient stays in 2010
| • |
|
Verigene Sensitivity and Specificity |
o Allow for more efficient and timely rule-in and rule-out o Potential to eliminate significant number of hospital admissions and unnecessary testing
Source: Healthcare Cost and Utilization Project. Statistical Brief #150. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb150.jsp. Accessed March 14, 2014.
|
|
NANOSPHERE COMPANY OVERVIEW
VERIGENE FLEX SYSTEM
| • |
|
One Instrument |
| • |
|
One Consumable |
| • |
|
Room Temperature |
| • |
|
Optimal Footprint |
| • |
|
Reduced Assay Time |
| • |
|
Easy Workflow |
|
|
NANOSPHERE COMPANY OVERVIEW
VERIGENE FLEX SYSTEM
| • |
|
Compact |
o Each instrument is equivalent to 1 Reader and 6 SPs
| • |
|
Hands-off Sample-to-Result |
o All processing and analysis occurs in the same box
| • |
|
Single Consumable Design |
|
|
VERIGENE FLEX SYSTEM
Current Verigene System Next Generation Verigene System
Competitive Best-in-Class
High multiplexed ? One step sample-to-result ? Accurate ? Flexibility / Cost per test ? Flexible ? Consolidates consumables ? Easy to use ? Room temperature stability ? On demand ? Optimal footprint Moderate complexity ? Best-in-class interface
|
|
NANOSPHERE COMPANY OVERVIEW
GLOBAL COMMERCIAL STRATEGY
| • |
|
U.S. Direct Sales Force |
o Productivity now Driving Execution
| • |
|
Placement penetration |
| • |
|
Contract time to live implementation |
| • |
|
Accelerate contract conversion Purchase/Rental |
| • |
|
Drive menu through current customer base |
o Gram Negative o Enteric o Respiratory Flex
| • |
|
Europe Distribution |
| • |
|
Asia / Pacific |
o Hitachi High Technologies
22
|
|
NANOSPHERE COMPANY OVERVIEW
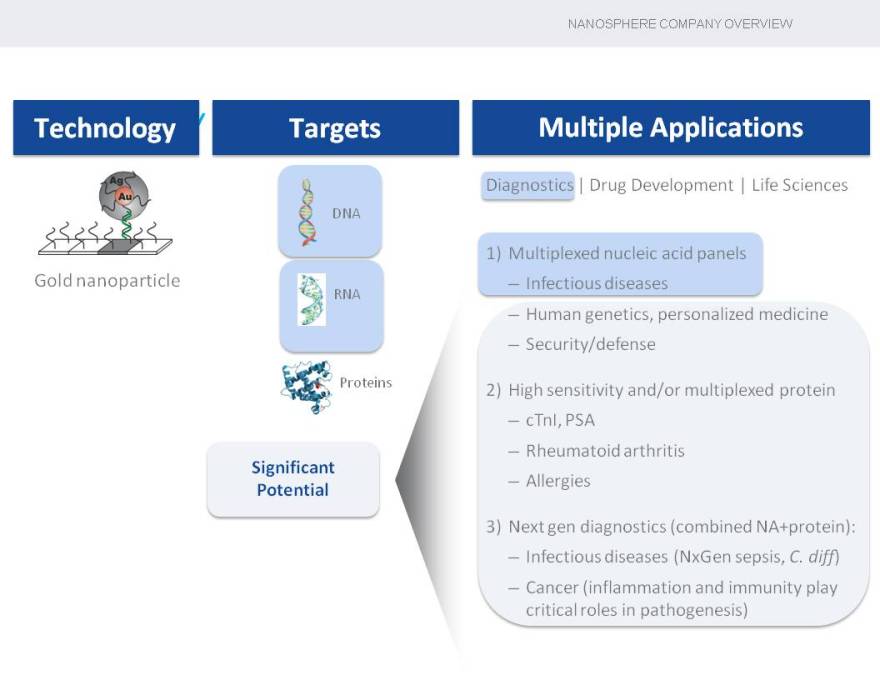
Technology Targets Multiple Applications
Diag es
DNA
1) Multiplexed nucleic acid panels Gold nanoparticle • Infectious diseases
RNA
| • |
|
Human genetics, personalized medicine |
| • |
|
Security/defense |
Proteins 2) High sensitivity and/or multiplexed protein • cTnI, PSA
| • |
|
Rheumatoid arthritis |
Significant
Potential • Allergies
3) Next gen diagnostics (combined NA+protein):
| • |
|
Infectious diseases (NxGen sepsis, C. diff) |
| • |
|
Cancer (inflammation and immunity play critical roles in pathogenesis) |
|
|
NANOSPHERE COMPANY OVERVIEW
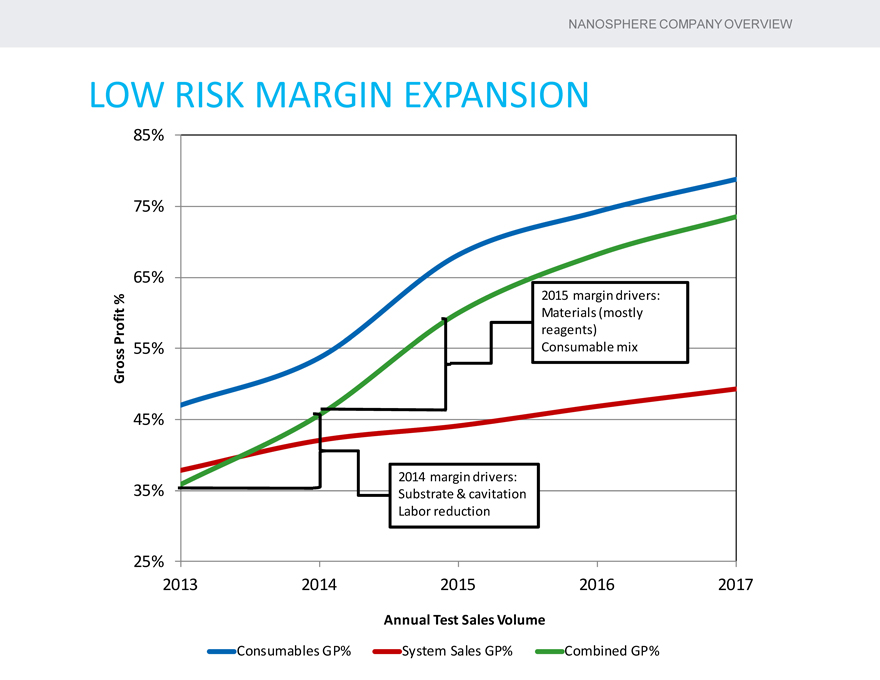
LOW RISK MARGIN EXPANSION
85%
75%
65%
% 2015 margin drivers: Materials (mostly Profit reagents) Gross 55% Consumable mix
45%
2014 margin drivers: 35% Substrate & cavitation Labor reduction
25%
2013 2014 2015 2016 2017
Annual Test Sales Volume
Consumables GP% System Sales GP% Combined GP%
|
|
NANOSPHERE COMPANY OVERVIEW
EVIDENCE OF OUR BELIEF
| • |
|
Leverage in Customer Base |
| • |
|
Expanding Menu |
| • |
|
Best-in-Class Performance |
| • |
|
Novel Flex™ tests designed to better compliment how healthcare is practiced |
| • |
|
Differentiating Value in Protein |
| • |
|
De-Risked for Technology/Regulatory/Reimbursement |
| • |
|
Straightforward Cost Reduction Leverage |
| • |
|
Focus on Execution and Delivering Results! |