Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - PROGENICS PHARMACEUTICALS INC | form8_k12oct2015.htm |
 |
Progenics Pharmaceuticals, Inc.
777 Old Saw Mill River Road
Tarrytown, New York 10591
Fax: (914) 789-2817
(914) 789-2800
www.progenics.com
|
Data from Phase 2 Study of Progenics' 1404 Imaging Agent Presented
at EANM Congress 2015
- Uptake Associated with Gleason Score Upgrade/Downgrade Following Radical Prostatectomy -
Tarrytown, N.Y., October 12, 2015 – Progenics Pharmaceuticals, Inc., (Nasdaq: PGNX) today announced the presentation of additional data from a Phase 2 trial of its PSMA-targeted imaging agent candidate, 1404, at the 28th Annual European Association of Nuclear Medicine (EANM) Congress being held in Hamburg, Germany. 1404 is a prostate specific membrane antigen (PSMA) targeted small molecule labeled with technetium-99m designed to "visualize" prostate cancer.
Results from the Phase 2 trial in high-risk patients undergoing radical prostatectomy were presented by Professor Karolien Goffin, M.D., Ph.D., University Hospitals Leuven, and included previously reported sensitivity and accuracy data for 1404 and data related to the uptake of 1404 in the lobes of the prostate gland. New data presented at the meeting demonstrate that 1404 uptake in the prostate gland is significantly higher in prostate cancer patients with upgraded Gleason scores at radical prostatectomy from those who are downgraded at radical prostatectomy.
"The data presented demonstrate the potential of 1404 to better assess the stage and extent of a patient's prostate cancer versus biopsy, as confirmed by comparison with histopathology, the truth standard, obtained from radical prostatectomy," stated Dr. Goffin.
In the Phase 2 study, 31% (25/81) of evaluable patients had their biopsy Gleason score upgraded to >7 or downgraded to 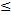 7 by histopathologic assessment of the prostatectomy specimen. Of patients upgraded to a Gleason score >7, 79% (11/14) demonstrated high lesion uptake of 1404 with a mean tumor to background ratio (TBR) of 47:1. Of patients downgraded to Gleason score
7 by histopathologic assessment of the prostatectomy specimen. Of patients upgraded to a Gleason score >7, 79% (11/14) demonstrated high lesion uptake of 1404 with a mean tumor to background ratio (TBR) of 47:1. Of patients downgraded to Gleason score 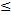 7, 91% (10/11) had significantly (p<0.001) lower lesion uptake of 1404 with a mean TBR of 16:1.
7, 91% (10/11) had significantly (p<0.001) lower lesion uptake of 1404 with a mean TBR of 16:1.
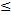 7 by histopathologic assessment of the prostatectomy specimen. Of patients upgraded to a Gleason score >7, 79% (11/14) demonstrated high lesion uptake of 1404 with a mean tumor to background ratio (TBR) of 47:1. Of patients downgraded to Gleason score
7 by histopathologic assessment of the prostatectomy specimen. Of patients upgraded to a Gleason score >7, 79% (11/14) demonstrated high lesion uptake of 1404 with a mean tumor to background ratio (TBR) of 47:1. Of patients downgraded to Gleason score 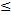 7, 91% (10/11) had significantly (p<0.001) lower lesion uptake of 1404 with a mean TBR of 16:1.
7, 91% (10/11) had significantly (p<0.001) lower lesion uptake of 1404 with a mean TBR of 16:1. About 1404, an Imaging Compound Targeting Prostate Specific Membrane Antigen
Progenics' molecular imaging radiopharmaceutical product candidate 1404 targets the extracellular domain of prostate specific membrane antigen (PSMA), a protein amplified on the surface of > 95% of prostate cancer cells and a validated target for the detection of primary and metastatic prostate cancer. 1404 is labeled with technetium-99m, a gamma-emitting isotope that is widely available, is easy to prepare, and is attractive for nuclear medicine imaging applications. The image created provides the opportunity to visualize cancer, potentially allowing for improved detection and staging, more precise biopsies, and a targeted treatment plan including active surveillance as a disease management tool.
About Prostate Cancer
Prostate cancer is the second most common form of cancer affecting men in the United States: an estimated one in seven men will be diagnosed with prostate cancer in his lifetime. The American Cancer Society estimates that each year approximately 220,800 new cases of prostate cancer will be diagnosed and about 27,540 men will die of the disease, and that approximately 2.9 million men in the U.S. currently count themselves among prostate cancer survivors.
Page 2
About Progenics
Progenics Pharmaceuticals, Inc. is developing innovative medicines for oncology, with a pipeline that includes several product candidates in later-stage clinical development. Progenics' first-in-class PSMA-targeted technology platform for prostate cancer includes a small molecule imaging agent that has completed a phase 2 trial and an antibody drug conjugate therapeutic which has also completed a two-cohort phase 2 clinical trial. Among other assets in its pipeline of targeted radiotherapy and molecular imaging compounds is AZEDRA™, an ultra-orphan radiotherapy candidate currently in a phase 2 study under an SPA. Progenics' first commercial product, RELISTOR® (methylnaltrexone bromide) for opioid-induced constipation, is partnered with and marketed by Valeant Pharmaceuticals International, Inc. For additional information, please visit www.progenics.com.
This press release may contain projections and other "forward-looking statements" regarding future events. Statements contained in this communication that refer to Progenics' estimated or anticipated future results or other non-historical facts are forward-looking statements that reflect Progenics' current perspective of existing trends and information as of the date of this communication. Forward looking statements generally will be accompanied by words such as "anticipate," "believe," "plan," "could," "should," "estimate," "expect," "forecast," "outlook," "guidance," "intend," "may," "might," "will," "possible," "potential," "predict," "project," or other similar words, phrases or expressions. Such statements are predictions only, and are subject to risks and uncertainties that could cause actual events or results to differ materially. These risks and uncertainties include, among others, the cost, timing and unpredictability of results of clinical trials and other development activities and collaborations, such as the Phase 3 clinical program for 1404; the unpredictability of the duration and results of regulatory review of New Drug Applications and Investigational NDAs; market acceptance for approved products; the sales of products by our partners and the royalty revenue generated thereby; generic and other competition; the possible impairment of, inability to obtain and costs of obtaining intellectual property rights; possible product safety or efficacy concerns, general business, financial and accounting matters, litigation and other risks. More information concerning Progenics and such risks and uncertainties is available on its website, and in its press releases and reports it files with the U.S. Securities and Exchange Commission. Progenics is providing the information in this press release as of its date and, except as expressly required by law, Progenics' disclaims any intent or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or circumstances or otherwise.
Additional information concerning Progenics and its business may be available in press releases or other public announcements and public filings made after this release. For more information, please visit www.progenics.com. Please follow us on LinkedIn®.
Information on or accessed through our website or social media sites is not included in the company's SEC filings.
(PGNX-F)
