Attached files
| file | filename |
|---|---|
| EX-32.2 - EXHIBIT 32.2 CERTIFICATION - PROGENICS PHARMACEUTICALS INC | ex32_212312009.htm |
| EX-32.1 - EXHIBIT 32.1 CERTIFICATION - PROGENICS PHARMACEUTICALS INC | ex32_112312009.htm |
| EX-23.1 - EXHIBIT 23.1 CONSENT OF PRICEWATERHOUSECOOPERS - PROGENICS PHARMACEUTICALS INC | ex23_112312009.htm |
| EX-31.1 - EXHIBIT 31.1 CERTIFICATION - PROGENICS PHARMACEUTICALS INC | ex31_112312009.htm |
| EX-31.2 - EXHIBIT 31.2 CERTIFICATION - PROGENICS PHARMACEUTICALS INC | ex31_212312009.htm |
| EX-10.33 - EXHIBIT 10.33 TERMINATION AND TRANSITION AGREEMENT - PROGENICS PHARMACEUTICALS INC | ex10_3312312009.htm |
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON,
D.C. 20549
_____________
FORM
10-K
(Mark
One)
|
x
|
ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
|
|
For
the fiscal year ended December 31, 2009
|
|
|
Or
|
|
|
¨
|
TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
|
|
For
the transition period from ___________ to
___________
|
Commission
File No. 000-23143
_____________
PROGENICS
PHARMACEUTICALS, INC.
(Exact
name of registrant as specified in its charter)
|
Delaware
|
13-3379479
|
|
(State
or other jurisdiction of
incorporation
or organization)
|
(I.R.S.
Employer Identification Number)
|
_____________
777
Old Saw Mill
River Road
Tarrytown,
NY 10591
(Address
of principal executive offices, including zip code)
Registrant’s
telephone number, including area code: (914) 789-2800
Securities
registered pursuant to Section 12(b) of the Act:
Title of each
class Name of each exchange on which
registered
Common Stock, par value $0.0013 per
share The NASDAQ Stock Market
LLC
|
Securities
registered pursuant to Section 12(g) of the Act:
|
None
|
_____________
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act. Yes o No
x
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Act.Yes o No
x
Indicate
by check mark whether the registrant (1) has filed all reports required to be
filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the
preceding 12 months (or for such shorter period that the registrant was required
to file such reports) and (2) has been subject to such filing requirements for
the past 90 days.
Yes x No
¨
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K is not contained herein, and will not be contained, to the best
of registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this
Form 10-K.
¨
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer or a smaller reporting company. See
definition of “large accelerated filer,” “accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Act:
|
Large
Accelerated Filer ¨
|
Accelerated
Filer x
|
|
Non-accelerated
Filer ¨ (Do not check if
a smaller reporting company)
|
Smaller
Reporting Company ¨
|
Indicate
by check mark whether the registrant is a shell company (as defined in Rule
12b-2 of the Exchange
Act). Yes
o No
x
The
aggregate market value of the voting and non-voting stock held by non-affiliates
of the registrant on June 30, 2009, based upon the closing price of the Common
Stock on The NASDAQ Stock Market LLC on that date of $5.15 per share, was
$69,591,796 (1).
|
(1)
|
Calculated
by excluding all shares that may be deemed to be beneficially owned by
executive officers, directors and five percent stockholders of the
Registrant, without conceding that any such person is an “affiliate” of
the Registrant for purposes of the Federal securities
laws.
|
As of
March 5, 2010, 32,229,627 shares of Common Stock, par value $.0013 per share,
were outstanding.
DOCUMENTS
INCORPORATED BY REFERENCE
Specified
portions of the Registrant’s definitive proxy statement to be filed in
connection with solicitation of proxies for its 2010 Annual Meeting of
Shareholders are hereby incorporated by reference into Part III of this
Form 10-K where such portions are referenced.
TABLE OF CONTENTS
|
PART
I
|
||
|
Item
1.
|
2
|
|
|
Item
1A.
|
13
|
|
|
Item
1B.
|
24
|
|
|
Item
2.
|
24
|
|
|
Item
3.
|
24
|
|
|
Item
4.
|
24
|
|
|
PART
II
|
||
|
Item
5.
|
25
|
|
|
Item
6.
|
26
|
|
|
Item
7.
|
27
|
|
|
Item
7A.
|
41
|
|
|
Item
8.
|
41
|
|
|
Item
9.
|
41
|
|
|
Item
9A.
|
42
|
|
|
Item
9B.
|
42
|
|
|
PART
III
|
||
|
Item
10.
|
43
|
|
|
Item
11.
|
43
|
|
|
Item
12.
|
43
|
|
|
Item
13.
|
43
|
|
|
Item
14.
|
43
|
|
|
PART
IV
|
||
|
Item
15.
|
44
|
|
|
F-1
|
||
|
S-1
|
||
|
E-1
|
||
PART
I
This document contains statements
that do not relate strictly to historical fact, any of which may be forward-looking statements within
the meaning of the U.S. Private Securities Litigation Reform Act of 1995.
When we use the words
“anticipates,” “plans,” “expects” and similar expressions, we are identifying
forward-looking statements. Forward-looking statements involve known and unknown
risks and uncertainties which may cause our actual results, performance or
achievements to be materially different from those expressed or implied by
forward-looking statements. While it is impossible to identify
or predict all such matters, these differences may result from, among other
things, the inherent uncertainty of the timing and success of, and expense
associated with, research, development, regulatory approval and
commercialization of our products and product candidates, including the risks
that clinical trials will not commence or proceed as planned; products appearing
promising in early trials will not demonstrate efficacy or safety in
larger-scale trials; clinical trial data on our products and product candidates
will be unfavorable; our products will not receive marketing approval from
regulators or, if approved, do not gain sufficient market acceptance to justify
development and commercialization costs; competing products currently on the
market or in development might reduce the commercial potential of our products;
we, our collaborators or others might identify side effects after the product is
on the market; or efficacy or safety concerns regarding marketed products,
whether or not originating from subsequent testing or other activities by us,
governmental regulators, other entities or organizations or otherwise, and
whether or not scientifically justified, may lead to product recalls,
withdrawals of marketing approval, reformulation of the product, additional
pre-clinical testing or clinical trials, changes in labeling of the product, the
need for additional marketing applications, declining sales or other adverse
events.
We
are also subject to risks and uncertainties associated with the actions of our
corporate, academic and other collaborators and government regulatory agencies,
including risks from market forces and trends; potential product liability;
intellectual property, litigation, environmental and other risks; the risk that
we may not be able to enter into a favorable collaboration or other
relationships or that existing or future relationships may not proceed as
planned; the risk that current and pending patent protection for our products
may be invalid, unenforceable or challenged, or fail to provide adequate market
exclusivity, or that our rights to in-licensed intellectual property may be
terminated for our failure to satisfy performance milestones; the risk of
difficulties in, and regulatory compliance relating to, manufacturing products;
and the uncertainty of our future profitability.
Risks
and uncertainties also include general economic conditions, including interest-
and currency exchange-rate fluctuations and the availability of capital; changes
in generally accepted accounting principles; the impact of legislation and
regulatory compliance; the highly regulated nature of our business, including
government cost-containment initiatives and restrictions on third-party payments
for our products; trade buying patterns; the competitive climate of our
industry; and other factors set forth in this document and other reports filed
with the U.S. Securities and Exchange Commission (SEC). In particular, we cannot
assure you that RELISTOR® will
be commercially successful or be approved in the future in other formulations,
indications or jurisdictions, or that any of our other programs will result in a
commercial product.
We
do not have a policy of updating or revising forward-looking statements, and we
assume no obligation to update any statements as a result of new information or
future events or developments. It should not be assumed that our silence over
time means that actual events are bearing out as expressed or implied in
forward-looking statements.
Available
Information
We file
annual, quarterly and current reports, proxy statements and other documents with
the SEC under the Securities Exchange Act of 1934. The SEC maintains an Internet
website that contains reports, proxy and information statements and other
information regarding issuers, including Progenics, that file electronically
with the SEC. You may obtain documents that we file with the SEC at http://www.sec.gov,
and read and copy them at the SEC’s Public Reference Room at 100 F Street NE,
Washington, DC 20549. You may obtain information on operation of the Public
Reference Room by calling the SEC at 1-800-SEC-0330. We also make available our
annual, quarterly and current reports and proxy materials on
http://www.progenics.com.
Progenics
Pharmaceuticals, Inc. is a biopharmaceutical company focusing on the development
and commercialization of innovative therapeutic products to treat the unmet
medical needs of patients with debilitating conditions and life-threatening
diseases. Our principal programs are directed toward supportive care, oncology
and virology. We commenced principal operations in 1988, became publicly traded
in 1997 and throughout have been engaged primarily in research and development
efforts, developing manufacturing capabilities, establishing corporate
collaborations and raising capital. All of our operations are conducted at our
facilities in Tarrytown, New York.
In supportive care, our first
commercial product is RELISTOR (methylnaltrexone
bromide) subcutaneous injection, a first-in-class therapy for opioid-induced
constipation approved for sale in over 40 countries worldwide, including the
United States, the European Union, Canada, Australia and Brazil. Marketing
applications are pending elsewhere throughout the world.
In
October 2009, we and Wyeth Pharmaceuticals terminated our 2005 RELISTOR
collaboration, as a result of which we regained all worldwide rights to
RELISTOR. Under our Transition Agreement, Wyeth is continuing to market and sell
RELISTOR for a U.S. Sales Period ending September 30, 2010 and an ex-U.S. Sales
Period ending December 31, 2010. After this transition period, we will assume
full control of and responsibility for future development and commercialization
of RELISTOR. Shortly after the termination, Pfizer Inc. completed its
previously-announced acquisition of Wyeth, which is now a wholly owned
subsidiary of Pfizer.
We are
pursuing a range of strategic alternatives for RELISTOR, including licensing,
collaboration and/or strategic alliances with world-wide or regional partners,
U.S. commercialization of the currently-approved product on our own or with
pharmaceutical detailing and sales organizations and/or co-promotion of the
franchise with a partner using our own sales force. Our October 2008 out-license
to Ono Pharmaceutical of the rights to subcutaneous RELISTOR in Japan is
unaffected by termination of the Wyeth collaboration.
With the
reacquisition of our rights to RELISTOR, we will be required, to the extent such
tasks are not undertaken by one or more partners, to address new technological,
clinical and commercial challenges, including, if we choose to sell and support
or co-promote RELISTOR, hiring a sales force, developing a commercial regulatory
compliance program and otherwise building a commercial infrastructure. This will
be particularly the case with the oral formulation of the drug, for which
we have taken over development responsibilities as part of the
reacquisition. We expect to continue to incur operating losses which could
increase significantly as we expand clinical trial and other product development
efforts that we choose or are obligated to undertake. See Supportive Care; Licenses; and Risk Factors.
In oncology, we are conducting a
phase 1 clinical trial of a fully human monoclonal antibody-drug conjugate (ADC)
directed against prostate specific membrane antigen (PSMA), a protein found at
high levels on the surface of prostate cancer cells and also on the
neovasculature of a number of other types of solid tumors. We are also
developing therapeutic vaccines designed to stimulate an immune response to PSMA
as an immunotherapy for prostate cancer. We are engaged in research to
identify possible candidates that may be effective in blocking certain signaling
pathways that are critical in the growth of aggressive cancers. See Oncology.
In virology, we are developing a
viral-entry inhibitor -- a humanized monoclonal antibody, PRO 140 -- for human
immunodeficiency virus (HIV), the virus that causes acquired immunodeficiency
syndrome, or AIDS. We are developing the subcutaneous form of PRO 140 for
treatment of HIV infection, which has the potential for weekly
self-administration. In our hepatitis C efforts, we are evaluating
second-generation HCV-entry inhibitors as possible development candidates. We
are also engaged in research regarding a prophylactic vaccine against HIV
infection and researching possible candidates that are HCV-entry inhibitors. See
Virology.
Licensing and Collaboration.
We seek out promising new products and technologies around which to build
new development programs or enhance existing programs. We own the worldwide
commercialization rights to each of our product candidates currently in
development, and we are seeking collaboration partners to accelerate
development of these candidates.
Following
is a summary of our principal programs and product candidates:
|
Lead
commercial product
|
Approved
indication
|
Status
|
||
|
Supportive
Care
|
||||
|
RELISTOR-Subcutaneous
injection
|
Approved
U.S. label: Treatment of opioid-induced constipation (OIC) in
advanced-illness patients receiving palliative care when laxative therapy
has not been sufficient (1)
|
Marketed
in the U.S., E.U., Canada, Australia, Brazil and elsewhere.
|
||
|
Program/product
candidates
|
Proposed
therapeutic area
|
Status (2)
|
||
|
Supportive
Care
|
||||
|
RELISTOR-Subcutaneous
injection
|
Treatment
of OIC in patients with chronic pain not related to cancer
|
Phase
3
|
||
|
RELISTOR-Oral
|
Treatment
of OIC
|
Expected
to commence Phase 2b/3 trial in second half of 2010
|
||
|
Oncology
|
||||
|
Prostate
cancer
|
||||
|
PSMA
ADC
|
Treatment
of prostate cancer
|
Phase
1
|
||
|
Recombinant
protein vaccine (rsPSMA)
|
Immunotherapy
for prostate cancer
|
Phase
1
|
||
|
Viral-vector
vaccine (PSMA VRP)
|
Immunotherapy
for prostate cancer
|
Phase
1
|
||
|
Evaluating
multiplex phosphoinositide 3-kinase (PI3K) inhibitor development
candidates
|
Treatment
of cancer
|
Research
|
||
|
Virology
|
||||
|
Human
Immunodeficiency Virus (HIV)
|
||||
|
PRO
140
|
Treatment
of HIV infection
|
Phase
2 (3)
|
||
|
ProVax
|
Treatment
and prevention of HIV infection
|
Research
|
||
|
Hepatitis
C Virus (HCV)
Evaluating
HCV-entry development candidates
|
Treatment
of HCV infection
|
Research
|
||
________________
|
(1)
|
RELISTOR
is a Wyeth trademark which Progenics will acquire upon completion of the
transition period following termination of our 2005 collaboration. The use
of RELISTOR beyond four months has not been studied. Full U.S. prescribing
information is available at www.RELISTOR.com. In this document,
“RELISTOR”
may refer to methylnaltrexone as it has been and is being developed and
commercialized by or in collaboration with Progenics. Subcutaneous
RELISTOR has received regulatory marketing approval for specific
indications, and references to RELISTOR do not imply that any other form
or possible use of the drug has received such
approval.
|
|||||
|
(2)
|
Research
means initial research related to specific molecular targets, synthesis of
new chemical entities, assay development or screening for
identification of lead compounds.
|
|||||
|
Pre-clinical
means a lead compound undergoing toxicology, formulation and other testing
in preparation for clinical trials.
|
||||||
|
Phase
1-3 clinical trials are safety and efficacy tests in
humans:
|
||||||
|
Phase
1: Initial evaluation of safety in humans; study method of action and
metabolization.
|
||||||
|
Phase
2: Evaluation of safety, dosing and activity or efficacy; continue safety
evaluation.
|
||||||
|
Phase
3: Larger scale evaluation of safety, efficacy and dosage.
|
||||||
|
(3)
|
Advancement
of this program is subject to our obtaining pivotal clinical-trial
funding, for which we have applied to government agencies.
|
|||||
Supportive
Care
It is
estimated that in 2008 nearly 250 million prescriptions for opioid-based
medications such as morphine and codeine were written in the U.S. to control
moderate-to-severe pain in patients receiving palliative care, undergoing
surgery, experiencing chronic pain or with other medical conditions. Opioids
relieve pain by interacting with receptors located in the brain and spinal cord,
but also activate receptors in the gut, often resulting in
constipation, referred to as opioid-induced constipation or OIC. As a
result of OIC, many patients may stop or reduce their opioid therapy, opting to
endure pain in order to obtain relief from their OIC and its associated side
effects.
RELISTOR, the first approved
treatment for OIC that addresses the underlying mechanism of this condition, is
a selective, peripherally acting, mu-opioid-receptor antagonist that decreases
the constipating side effects induced by opioid pain medications in the
gastrointestinal tract without diminishing the ability of these medications to
relieve pain. Relief of OIC is an important need that is not adequately met by
any other approved drug or intervention. Because of its chemical composition,
RELISTOR has restricted access to the blood-brain barrier to enter the central
nervous system, where pain is perceived. Outside the central nervous system,
RELISTOR competes with opioid pain medications for binding sites on opioid
receptors, displacing the pain medications only in the periphery and selectively
“turning off” the constipating effects of those medications on the
gastrointestinal tract without affecting pain relief occurring in the central
nervous system.
Subcutaneous
RELISTOR. In the second quarter of 2008 we began earning royalties on
Wyeth’s sales of RELISTOR. RELISTOR net sales and related royalties earned
through the end of 2009 are set forth below. Our recognition of royalty revenue
for financial reporting purposes is explained in Management’s Discussion and Analysis
of Financial Condition and Results of Operations (MD&A) and our
financial statements elsewhere in this document.
|
2009
|
|||||||||
|
First
Quarter
|
Second
Quarter
|
Third
Quarter
|
Fourth
Quarter
|
Full
Year
|
|||||
|
(in
thousands)
|
|||||||||
|
Net
Sales By Wyeth
|
$1,900
|
$3,200
|
$3,300
|
$3,900
|
$12,300
|
||||
|
Royalties
Earned
|
280
|
487
|
497
|
589
|
1,853
|
||||
|
2008
|
|||||||
|
Second
Quarter
|
Third
Quarter
|
Fourth
Quarter
|
Full
Year
|
||||
|
(in
thousands)
|
|||||||
|
Net
Sales By Wyeth
|
$2,100
|
$800
|
$1,500
|
$4,400
|
|||
|
Royalties
Earned
|
321
|
117
|
227
|
665
|
|||
Under the
Transition Agreement, Wyeth has agreed to pay us the sum of $10.0 million in six
quarterly installments and is continuing certain ongoing development efforts for
subcutaneous RELISTOR, at its expense, through September 30, 2010. Wyeth’s
international sales and marketing obligations during the ex-U.S. Sales Period
are subject to certain extension and early transition options available to us.
Wyeth will continue to pay royalties on worldwide sales as provided in the 2005
collaboration agreement except that no royalties will be payable in respect of
ex-U.S. sales during (i) the fourth quarter of 2010 to the extent certain
financial targets are not met or (ii) an extended ex-U.S. Sales Period in the
subject country. Principal responsibility for regulatory submissions and
interactions for all other formulations and presentations of RELISTOR will be
transferred during and as part of the transition. Wyeth is also providing
financial resources, aggregating up to approximately $14.5 million, and/or other
assistance with respect to agreed-upon regulatory, manufacturing and supply
matters. We have agreed to purchase Wyeth’s remaining inventory of subcutaneous
RELISTOR at the end of the Sales Periods on agreed-upon terms and
conditions.
Under its
License Agreement from us, in June 2009, Ono began clinical testing in Japan of
RELISTOR subcutaneous injection and is continuing development of the drug there.
In August 2009, we and Wyeth announced submission to U.S. and E.U. regulators of
applications for subcutaneous RELISTOR in pre-filled syringes, which are
designed to ease preparation and administration for patients and
caregivers.
We are
also developing subcutaneous RELISTOR for treatment of OIC outside the
advanced-illness setting, in individuals with chronic pain not related to
cancer. We and Wyeth recently completed enrollment in a one-year, open-label
safety study, results from which, together with results from a previous phase 3
efficacy trial, will support planned regulatory filings in early 2011 in the
U.S., Europe and elsewhere for approval of RELISTOR to treat OIC in the
chronic-pain setting.
Oral
RELISTOR. As
part of our reacquisition of RELISTOR, we have taken over development
responsibilities for an oral formulation of RELISTOR for the treatment of OIC in
patients with chronic, non-cancer pain. In March 2010, we announced that
we plan to advance oral methylnaltrexone for the treatment of OIC into late
stage clinical development and will commence a phase 2b/3 clinical trial of
a methylnaltrexone tablet in chronic-pain patients in the second half of
2010.
* * *
Our
oncology and virology product candidates are not as advanced in development as
RELISTOR, and we do not expect any recurring revenues from sales or otherwise
with respect to these product candidates in the near term.
Oncology
Prostate
cancer is a common cancer affecting men in the U.S. and a leading cause
of cancer deaths in men each year. The National Cancer Institute estimates that,
based on rates from 2002-2004, one in six men will be diagnosed with this
disease. The American Cancer Society estimated that 192,280 new cases of
prostate cancer would be diagnosed and that 27,360 men would die from the
disease in 2009 in the U.S. Conventional therapies, including radical
prostatectomy, radiation, hormone therapies and chemotherapy, may have or
increase the risk of side effects, including impotence, incontinence, high
cholesterol levels and increased blood-clot risk, and some are generally not
intended to be curative and are not actively used to treat localized,
early-stage prostate cancer.
Through
our PSMA Development Company, we conduct research and development programs
relating to antibody and vaccine therapies directed against prostate specific
membrane antigen, or PSMA, a protein that is
abundantly expressed on the surface of prostate cancer cells as well as cells in
the newly formed blood vessels of many other solid tumors. Our fully human
monoclonal ADC is designed to deliver a chemotherapeutic agent to cancer cells
by targeting the three-dimensional structure of the PSMA protein on these cells
and binding to and internalizing within the cell. We believe a PSMA-directed
therapy may have application in prostate cancer and solid tumors of other types
of cancer. We are conducting a phase 1 dose-escalation clinical study, commenced
in 2008, to assess PSMA ADC’s safety, tolerability and initial clinical activity
in patients with progressive, castrate-resistant prostate cancer. We are
pursuing strategic collaborations with biopharmaceutical companies to support
development of PSMA ADC.
We have
also initiated clinical study of a therapeutic vaccine utilizing viral vectors
designed to deliver the PSMA gene to certain immune system cells in order to
generate potent and specific immune responses to prostate cancer cells. In
pre-clinical studies, this vaccine generated a potent dual response against
PSMA, by both antibodies and killer T-cells, the two principal mechanisms used
by the immune system to eliminate abnormal cells. In addition, we are developing
a vaccine combining the PSMA cancer antigen (recombinant soluble PSMA) with an
immune stimulant to induce an immune response against prostate cancer
cells.
We
recently presented data from preclinical studies of novel multiplex
phosphoinositide 3-kinase (PI3K) inhibitors -- synthetic, small-molecule
compounds identified by us that, in laboratory studies, blocked both PI3K, a key
regulator of one molecular signaling pathway, and MNK, an oncogenic kinase in
the Ras pathway. We believe simultaneously blocking these interlinked cellular
pathways may provide a strategy to combat some of the most aggressive forms of
cancer.
Virology
HIV. An
estimated 33 million people worldwide are infected with HIV, which causes slowly
progressing immune system deterioration resulting in AIDS. Although most
prevalent in sub-Saharan Africa, the current commercial market is generally
limited to the U.S. and Europe, where it is estimated over two million people
are infected. HIV infects and kills cells that have the CD4 receptor on their
surface, including critical immune system components such as T-lymphocytes,
monocytes, macrophages and dendritic cells. The current standard of HIV care,
known as combination therapy, slows progression of disease but is not a cure.
HIV’s rapid mutation rate results in resistance to existing drugs, while dosing
inconsistencies and toxic side effects further dilute their impact.
Viral
entry inhibitors, such as our drug candidate PRO 140, represent the newest
class of drugs for HIV patients. Our program is based on blocking the binding of
HIV to CCR5, a co-receptor to CD4. CCR5 inhibitors such as PRO 140 do not block
the entry of virus that uses CXCR4, a less ubiquitous co-receptor to CD4 that is
used by some strains of HIV either exclusively or in addition to CCR5. At
therapeutic concentrations tested to date, PRO 140 does not appear to interfere
with CCR5’s normal function in the body’s inflammatory response. PRO 140 has
been granted “Fast Track” status from the FDA, which facilitates development and
expedites regulatory review of drugs intended to address unmet medical needs for
serious or life-threatening conditions. We are developing the subcutaneous form
of PRO 140 for treatment of HIV infection, which has the potential for weekly
self-administration. Advancement of this program is subject to our obtaining
pivotal clinical-trial funding, for which we have applied to government
agencies.
ProVax is our vaccine product candidate, under development for the
prevention of HIV infection or as a therapeutic treatment for HIV-positive
individuals, which is designed to stimulate the production of specific HIV
neutralizing antibodies, a process that to date has been difficult to induce
with vaccines currently in development. We have previously funded this project
via a National Institutes of Health contract, and in June 2009, were awarded a
five-year NIH grant totaling up to $14.5 million to continue this work, subject
to annual funding approvals and customary compliance obligations.
HCV. We are evaluating second-generation HCV-entry inhibitors as
possible development candidates to treat the most common blood-borne infection
in the U.S. and a major cause of chronic liver disease. According to the U.S.
Centers for Disease Control and Prevention, an estimated 4.1 million Americans
have been infected with HCV, of whom 3.2 million are chronically infected, with
approximately 17,000 new infections in 2007; more than 170 million people
worldwide are estimated to be infected. Chronic hepatitis C, an under-recognized
disease which can lead to cirrhosis and ultimately liver failure, is now the
most common reason for liver transplantation and the leading cause of liver
cancer in the U.S.
Licenses
Progenics and PSMA LLC are parties to license agreements under which we have in-
and/or out-licensed rights to use certain technologies and materials. These
licenses provide for various royalty, milestone and other payment,
commercialization, sublicensing, patent prosecution and enforcement, insurance,
indemnification and other obligations and rights, and are subject to certain
reservations of rights. Our costs in defending patent rights, both our own and
those we license, have historically not been material. Set forth below is a
summary of the more significant of these licenses.
RELISTOR
· The
October 2009 Transition Agreement with Wyeth provides for the termination of our
2005 collaboration agreement and the transition to Progenics of responsibility
for the development and commercialization of RELISTOR. Under it, Wyeth’s license
of Progenics technology is terminated except as necessary for performance of
Wyeth’s obligations during the transition period, and Wyeth has returned the
rights to RELISTOR that we granted under the 2005 agreement.
Wyeth is
continuing to market and sell RELISTOR worldwide in accordance with an
agreed-upon commercialization plan. Wyeth will continue to market and sell
RELISTOR through September 30, 2010 in the United States and through December
31, 2010 in the rest of the world other than Japan, subject to ex-U.S.
country-by-country extensions, at Progenics’ option in certain circumstances,
and ex-U.S. country-by-country early transition, at Progenics’ option. Wyeth
shall continue to pay royalties as provided in the 2005 Agreement except that no
royalties shall be payable in respect of ex-U.S. sales made during (i) the
fourth quarter of 2010 to the extent that certain financial targets for such
quarter are not met or (ii) an extended international sale period in the subject
country. Wyeth has also agreed to pay us the sum of $10.0 million in
installments through January, 2011, and, at its expense, is continuing certain
ongoing development efforts for subcutaneous RELISTOR through September 30,
2010, including conducting specified clinical studies, and is providing support
for developing specified new delivery mechanisms, in accordance with an
agreed-upon development plan.
Progenics
has assumed responsibility for development of oral RELISTOR, including conduct
of clinical trials, and the parties are transitioning to Progenics
responsibility for regulatory submissions and interactions for all other
formulations and presentations of RELISTOR. Wyeth is providing financial
resources and/or other assistance with respect to additional agreed-upon
regulatory, manufacturing and supply matters, and Progenics has agreed to
purchase Wyeth’s remaining inventory of subcutaneous RELISTOR at the end of its
Sales Periods on agreed-upon terms and conditions. Wyeth has granted us a
fully-paid up, irrevocable, exclusive (except as to Wyeth and its affiliates)
license to relevant technology generated by Wyeth alone or in conjunction with
Progenics during the course of our 2005 collaboration and during Wyeth’s
performance of its Termination Agreement obligations, assigned or agreed to
assign to us specified RELISTOR-related commercial intellectual property,
including trademarks, Internet domain names and copyrights, and agreed not to
assert against us or our affiliates, licensees or other third-party business
partners any patent rights owned by Wyeth or its affiliates that would be
infringed by unlicensed development or commercialization of RELISTOR. Progenics
now controls filing, prosecution, abandonment and enforcement against third
party infringers of the patent rights related to its technology and those
licensed to it by Wyeth. The Termination Agreement also provides for transfer of
development, manufacturing and commercialization records and other materials,
mutual releases between the parties and indemnification, dispute resolution,
non-disparagement and other customary provisions.
The 2005 collaboration agreement was in effect until October 2009, which
includes periods covered by this report. Under that agreement, we granted to
Wyeth an exclusive, worldwide license to develop and commercialize RELISTOR and
assigned certain agreements to it. We were responsible for developing the
subcutaneous and intravenous formulations in the U.S. until they received
regulatory approval, while Wyeth was responsible for these formulations outside
the U.S. (other than Japan after execution of the Ono license) and for
developing the oral formulation worldwide. From January 2006 to October 2009,
development costs for RELISTOR were paid by Wyeth. We were reimbursed for
out-of-pocket costs and received reimbursement for our efforts based on our
employees devoted to them, subject to Wyeth’s audit rights and possible
reconciliation. Commercialization decisions were made by Wyeth, which also was
obligated to pay all commercialization costs, including manufacturing costs, and
retained all proceeds from product sales, subject to royalties and other amounts
payable to us. The collaboration was overseen by committees, which coordinated
or monitored the companies’ key strategic, development and commercialization
activities.
· We have
exclusive rights to develop and commercialize methylnaltrexone, the active
ingredient of RELISTOR, under license from the University of Chicago. We are
obligated to make milestone and royalty payments to the University that in
general extend to the expiration of the last-to-expire relevant
patent.
· We have
licensed to Ono
Pharmaceutical the rights to subcutaneous RELISTOR in Japan, where Ono is
responsible for developing and commercializing subcutaneous RELISTOR, including
conducting clinical development to support regulatory marketing approval. Ono
will own the subcutaneous filings and approvals relating to RELISTOR in Japan.
We have received a $15.0 million upfront payment from Ono, and are entitled to
receive up to an additional $20.0 million, payable upon achievement of
development milestones. Ono is also obligated to pay us royalties and
commercialization milestones on sales of subcutaneous RELISTOR in Japan. Ono has
the option to acquire the rights to develop and commercialize other formulations
of RELISTOR in Japan, on terms to be negotiated separately. Supervision of and
consultation with respect to Ono’s development and commercialization
responsibilities are carried out by joint committees. The Ono License contains,
among other terms, provisions which permit termination by either party upon the
occurrence of certain events.
PSMA
· PSMA
Development Company LLC has a collaboration agreement with Seattle Genetics, Inc., under
which SGI has granted it an exclusive worldwide license to SGI’s proprietary ADC
technology. PSMA LLC has the right to use this technology, which is based in
part on technology licensed by SGI from third parties, to link chemotherapeutic
agents to PSMA LLC’s monoclonal antibodies that target prostate specific
membrane antigen. PSMA LLC is responsible for research, product development,
manufacturing and commercialization of all products, and may sublicense the ADC
technology to a third party manufacturer. PSMA LLC is obligated to make
maintenance payments, milestone payments aggregating up to $14.0 million and to
pay royalties to SGI and its licensors, as applicable, on a percentage of net
sales. The SGI agreement terminates at the latest of (i) the tenth anniversary
of the first commercial sale of each licensed product in each country or (ii)
the latest date of expiration of patents underlying the licensed products. PSMA
LLC may terminate the agreement upon advance written notice, and SGI may
terminate if PSMA LLC fails to cure a breach of an SGI in-license within a
specified time period after written notice. In addition, either party may
terminate the agreement after written notice upon an uncured breach or in the
event of bankruptcy of the other party. As of December 31, 2009, PSMA LLC has
paid approximately $3.6 million under this agreement, including $1.0 million in
milestone payments.
· PSMA LLC
also has a worldwide exclusive licensing agreement with Abgenix (now Amgen Fremont, Inc.) to use
its XenoMouse®
technology for generating fully human antibodies to PSMA LLC’s PSMA antigen.
PSMA LLC is obligated to make development and commercialization milestone
payments with respect to products incorporating an antibody generated utilizing
the XenoMouse technology. As of December 31, 2009, PSMA LLC has paid $850,000
under this agreement and is obligated to pay up to an additional $6.25 million
if certain milestones are met, along with royalties based upon net sales of
antibody products, if any. This agreement may be terminated, after an
opportunity to cure, by Abgenix for cause upon 30 days prior written notice;
PSMA LLC has the right to terminate upon 30 days prior written notice. The
agreement continues until the later of the expiration of the XenoMouse
technology patents that may result from pending patent applications or seven
years from the first commercial sale of the products.
· PSMA LLC
has a worldwide exclusive license agreement with AlphaVax Human Vaccines to use
its Replicon Vector system to create a therapeutic prostate cancer vaccine
incorporating PSMA LLC’s proprietary PSMA antigen. PSMA LLC is obligated to make
development and commercialization milestone payments with respect to products
incorporating AlphaVax’s system. As of December 31, 2009, PSMA LLC has paid $2.0
million under this agreement and is obligated to pay up to an additional $5.3
million if certain milestones are met along with annual maintenance fees and
royalties based upon net sales of any products developed using AlphaVax’ system.
This agreement may be terminated, after an opportunity to cure, by AlphaVax
under specified circumstances, including PSMA LLC’s failure to achieve
milestones; the consent of AlphaVax to revisions to the milestones due dates may
not, however, be unreasonably withheld. PSMA LLC has the right to terminate upon
30 days prior written notice. The agreement continues until the later of the
expiration of the patents relating to AlphaVax’s system or seven years from the
first commercial sale of the products developed using that system. Pending U.S.
and international patent applications and patent-term extensions may extend the
period of our license rights when and if they are allowed, issued or
granted.
PRO 140; Virology
· Protein Design Labs (now Facet Biotech Corporation)
humanized a murine monoclonal antibody developed by us (humanized PRO 140) and
granted us related licenses under patents and patent applications, in addition
to know-how. In general, these licenses are fully paid after the latest of (i)
the tenth anniversary of the first commercial sale of a product developed
thereunder, (ii) expiration of the last-to-expire relevant patent or (iii) the
tenth anniversary of the latest filed pending patent application. Pending U.S.
and international patent applications and patent-term extensions may extend the
period of our license rights when and if they are allowed, issued or granted. We
may terminate the license on 60 days prior written notice, and either party may
terminate on 30 days prior written notice for an uncured material breach (ten
days for payment default). As of December 31, 2009, we have paid Facet’s
predecessors $5.4 million, and if all milestones are achieved, we will be
obligated to pay an additional approximately $2.0 million. We are also required
to pay annual maintenance fees of $150,000 and royalties on sales of products
developed under the license.
· We have a
letter agreement with the Aaron
Diamond AIDS Research Center pursuant to which we have the exclusive
right to pursue the commercial development, directly or with a partner, of
products related to HIV based on patents jointly owned by ADARC and
us.
Patents
and Proprietary Technology
Our policy is to protect our proprietary technology, and we consider the
protection of our rights to be important to our business. In addition to seeking
U.S. patent protection for many of our inventions, we generally file patent
applications in Canada, Japan, European countries that are party to the European
Patent Convention and additional foreign countries on a selective basis in order
to protect the inventions that we consider to be important to the development of
our foreign business. Generally, patents issued in the U.S. are
effective:
· if the patent application was filed prior to
June 8, 1995, for the longer of 17 years from the date of issue or 20 years from
the earliest asserted filing date; or
· if the application was filed on or after June 8,
1995, for 20 years from the earliest asserted filing date.
In addition, in certain instances, the patent term can be extended up to a
maximum of five years to recapture a portion of the term during which the FDA
regulatory review was being conducted. The duration of foreign patents varies in
accordance with the provisions of applicable local law, although most countries
provide for patent terms of 20 years from the earliest asserted filing date and
allow patent extensions similar to those permitted in the U.S.
We also rely on trade secrets, proprietary know-how and continuing technological
innovation to develop and maintain a competitive position in our product areas.
We generally require our employees, consultants and corporate partners who have
access to our proprietary information to sign confidentiality
agreements.
Our patent portfolio relating to our proprietary technologies in the supportive
care, oncology and virology areas is currently comprised, on a worldwide basis,
of 154 patents that have been issued and 372 pending patent applications, which
we either own directly or of which we are the exclusive licensee. Our issued
patents expire on dates ranging from 2010 through 2026. Patent-term extensions
and pending patent applications may extend the period of patent protection
afforded our products in development.
We are aware of intellectual property rights held by third parties that relate
to products or technologies we are developing. For example, we are aware of
others investigating methylnaltrexone and other peripheral opioid antagonists,
PSMA or related compounds, CCR5 monoclonal antibodies and HCV viral entry
inhibitors, and of patents and applications held or filed by others in those
areas. While the validity of issued patents, patentability of claimed inventions
in pending applications and applicability of any of them to our programs are
uncertain, patent rights asserted against us could adversely affect our ability
to commercialize or collaborate with others regarding our products.
The research, development and commercialization of a biopharmaceutical product
often present alternative development and optimization routes at various stages
in the development process. Preferred routes cannot be identified with certainty
at the outset because they will depend upon subsequent discoveries and test
results. There are numerous third-party patents in our field, and it is possible
that to pursue the preferred development route of one or more of our product
candidates we will need to obtain a license under a patent, which could decrease
the ultimate profitability of the applicable product. If we cannot negotiate a
license, pursuit of a less desirable development route or termination of the
entire program may be necessary.
Government
Regulation
Progenics and its product candidates are subject to comprehensive regulation by
the FDA and comparable authorities in other countries. Pharmaceutical regulation
currently is a topic of substantial interest in lawmaking and regulatory bodies
in the U.S. and internationally, and numerous proposals exist for changes in FDA
and non-U.S. regulation of pre-clinical and clinical testing, safety,
effectiveness, approval, manufacture, labeling, marketing, export, storage,
recordkeeping, advertising, promotion and other aspects of biologics, small
molecule drugs and medical devices, many of which, if adopted, could
significantly alter our business and the current regulatory structure described
below.
FDA Regulation. FDA approval of our product candidates, including a
review of the manufacturing processes and facilities used to produce them, are
required before they may be marketed in the U.S. This process is costly, time
consuming and subject to unanticipated delays, and a drug candidate may fail to
progress at any point.
None of our product candidates other than RELISTOR has received marketing
approval from the FDA or any other regulatory authority. The process required by
the FDA before product candidates may be approved for marketing in the U.S.
generally involves:
· pre-clinical laboratory and animal
tests;
· submission to the FDA and effectiveness of an
IND before clinical trials may begin;
· adequate and well-controlled human clinical
trials to establish the safety and efficacy of the product for its intended
indication (animal and other nonclinical studies also
are typically conducted during each phase of human clinical
trials);
· submission to the FDA of a marketing
application; and
· FDA review of the marketing application in order
to determine, among other things, whether the product is safe and effective for
its intended uses.
Pre-clinical
tests include laboratory evaluation of product chemistry and animal
studies to gain preliminary information about a product’s pharmacology and
toxicology and to identify safety problems that would preclude testing in
humans. Products must generally be manufactured according to current Good
Manufacturing Practices, and pre-clinical safety tests must be conducted by
laboratories that comply with FDA good laboratory practices
regulations.
Results of pre-clinical tests are submitted to the FDA as part of an IND (Investigational New Drug)
application, which must become effective before clinical trials may commence.
The IND submission must include, among other things, a description of the
sponsor’s investigational plan; protocols for each planned study; chemistry,
manufacturing and control information; pharmacology and toxicology information
and a summary of previous human experience with the investigational
drug.
Unless the FDA objects to, makes comments or raises questions concerning an IND,
it becomes effective 30 days following submission, and initial clinical studies
may begin. Companies often obtain affirmative FDA approval, however, before
beginning such studies. We cannot assure you that an IND submission by us will
result in FDA authorization to commence clinical trials.
Clinical
trials involve the administration of the investigational new drug to
healthy volunteers or to individuals under the supervision of a qualified
principal investigator. Clinical trials must be conducted in accordance with the
FDA’s Good Clinical Practice requirements under protocols submitted to the FDA
that detail, among other things, the objectives of the study, parameters used to
monitor safety and effectiveness criteria to be evaluated. Each clinical study
must be conducted under the auspices of an Institutional Review Board, which
considers, among other things, ethical factors, safety of human subjects,
possible liability of the institution and informed consent disclosure which must
be made to participants in the trial.
Clinical trials are typically conducted in three sequential phases, which may
overlap. During phase 1, when the drug is initially administered to human
subjects, the product is tested for safety, dosage tolerance, absorption,
metabolism, distribution and excretion. Phase 2 involves studies in a limited
population to evaluate preliminarily the efficacy of the product for specific,
targeted indications, determine dosage tolerance and optimal dosage and identify
possible adverse effects and safety risks.
When a product candidate is found in phase 2 evaluation to have an effect and an
acceptable safety profile, phase 3 trials are undertaken in order to further
evaluate clinical efficacy and test for safety within an expanded population.
Safety studies are conducted in accordance with the FDA’s International
Conference on Harmonization (ICH) Guidelines. Phase 2 results do not guarantee a
similar outcome in phase 3 trials. The FDA may suspend clinical trials at any
point in this process if it concludes that clinical subjects are being exposed
to an unacceptable health risk.
A New Drug Application,
or NDA, is an
application to the FDA to market a new drug. A Biologic License Application,
or BLA, is an
application to market a biological product. The new drug or biological product
may not be marketed in the U.S. until the FDA has approved the NDA or issued a
biologic license. The NDA must contain, among other things, information on
chemistry, manufacturing and controls; non-clinical pharmacology and toxicology;
human pharmacokinetics and bioavailability; and clinical data. The BLA must
contain, among other things, data derived from nonclinical laboratory and
clinical studies which demonstrate that the product meets prescribed standards
of safety, purity and potency, and a full description of manufacturing methods.
Supplemental NDAs (sNDAs) are submitted to obtain regulatory approval for
additional indications for a previously approved drug.
The results of the pre-clinical studies and clinical studies, the chemistry and
manufacturing data, and the proposed labeling, among other things, are submitted
to the FDA in the form of an NDA or BLA. The FDA may refuse to accept the
application for filing if certain administrative and content criteria are not
satisfied, and even after accepting the application for review, the FDA may
require additional testing or information before approval of the application.
Our analysis of the results of our clinical studies is subject to review and
interpretation by the FDA, which may differ from our analysis. We cannot assure
you that our data or our interpretation of data will be accepted by the FDA. In
any event, the FDA must deny an NDA or BLA if applicable regulatory requirements
are not ultimately satisfied. In addition, we may encounter delays or rejections
based upon changes in applicable law or FDA policy during the period of product
development and FDA regulatory review. If regulatory approval of a product is
granted, such approval may be made subject to various conditions, including
post-marketing testing and surveillance to monitor the safety of the product, or
may entail limitations on the indicated uses for which it may be marketed.
Finally, product approvals may be withdrawn if compliance with regulatory
standards is not maintained or if problems occur following initial
marketing.
Both before and after approval is obtained, a product, its manufacturer and the
sponsor of the marketing application for the product are subject to
comprehensive regulatory oversight. Violations of regulatory requirements at any
stage, including the pre-clinical and clinical testing process, the approval
process, or thereafter, may result in various adverse consequences, including
FDA delay in approving or refusal to approve a product, withdrawal of an
approved product from the market or the imposition of criminal penalties against
the manufacturer or sponsor. Later discovery of previously unknown problems may
result in restrictions on the product, manufacturer or sponsor, including
withdrawal of the product from the market. New government requirements may be
established that could delay or prevent regulatory approval of our products
under development.
Regulation Outside the U.S. Whether or not FDA approval has been
obtained, approval of a pharmaceutical product by comparable government
regulatory authorities in foreign countries must be obtained prior to marketing
the product there. The approval procedure varies from country to country, and
the time required may be longer or shorter than that required for FDA approval.
The requirements we must satisfy to obtain regulatory approval by governmental
agencies in other countries prior to commercialization of our products there can
be rigorous, costly and uncertain, and there can be no assurance that approvals
will be granted on a timely basis or at all. We do not currently have any
facilities or personnel outside of the U.S.
In the European Union, Canada, Australia and Japan, regulatory requirements and
approval processes are similar in principle to those in the United States.
Regulatory approval in Japan requires that clinical trials of new drugs be
conducted in Japanese patients. Depending on the type of drug for which approval
is sought, there are currently two potential tracks for marketing approval in
the E.U. countries: mutual recognition and the centralized procedure. These
review mechanisms may ultimately lead to approval in all E.U. countries, but
each method grants all participating countries some decision-making authority in
product approval. The centralized procedure, which is mandatory for
biotechnology derived products, results in a recommendation in all member
states, while the E.U. mutual recognition process involves country-by-country
approval.
In other countries, regulatory requirements may require additional pre-clinical
or clinical testing regardless of whether FDA approval has been obtained. This
is the case in Japan, where Ono is responsible for developing and
commercializing the subcutaneous form of RELISTOR and where trials are required
to involve patient populations which we and Wyeth have not examined in detail.
If the particular product is manufactured in the U.S., we must also comply with
FDA and other U.S. export provisions.
In most countries outside the U.S., coverage, pricing and reimbursement
approvals are also required. There can be no assurance that the resulting
pricing of our products would be sufficient to generate an acceptable return to
us.
Other Regulation. In addition to regulations enforced by the FDA, we are
also subject to regulation under the Occupational Safety and Health Act, the
Environmental Protection Act, the Toxic Substances Control Act, the Resource
Conservation and Recovery Act and various other current and potential future
federal, state or local regulations. Our research and development involves the
controlled use of hazardous materials, chemicals, viruses and various
radioactive compounds. Although we believe that our safety procedures for
storing, handling, using and disposing of such materials comply with the
standards prescribed by applicable regulations, we cannot completely eliminate
the risk of accidental contaminations or injury from these materials. In the
event of such an accident, we could be held liable for any legal and regulatory
violations as well as damages that result. Any such liability could have a
material adverse effect on Progenics.
Manufacturing
Under the Transition Agreement with Wyeth, we will be responsible for obtaining
supplies of RELISTOR after the end of Wyeth’s transition obligations, which
include supplying us and our collaboration partner(s) with finished subcutaneous
drug product and active pharmaceutical ingredient (API) remaining in Wyeth’s
inventories. Going forward, we expect we or our collaboration partner(s) or
licensee(s) will extend current relationships or contract with one or more other
contract manufacturing organizations (CMOs) for supply of RELISTOR API and
subcutaneous and oral finished drug product. See Risk Factors.
We currently manufacture clinical trial supplies of our PSMA monoclonal antibody
in our biologics pilot production facilities in Tarrytown, New York, and have
engaged third-party CMOs for other portions of the PSMA ADC manufacturing
process. We expect our manufacturing capacity will not be sufficient for all of
our late-stage clinical trials or commercial-scale requirements. If we are
unable to arrange for satisfactory CMO services, or otherwise determine to
acquire additional manufacturing capacity, we will need to expand our
manufacturing staff and facilities or obtain new facilities. In order to
establish a full-scale commercial manufacturing facility for any of our product
candidates, we would need to spend substantial additional funds, hire and train
significant numbers of employees and comply with the extensive FDA regulations
applicable to such a facility.
Sales
and Marketing
We are pursuing a range of strategic alternatives for RELISTOR, including
licensing, collaboration, strategic alliances with worldwide or regional
partners, U.S. commercialization of the currently approved product on our own or
with third-party detailing and sales organizations and/or co-promotion of the
franchise with our own sales force. We also continue to seek strategic
collaborations and other funding support for product candidates in our pipeline.
We plan to market other products for which we obtain regulatory approval through
co-marketing, co-promotion, licensing and distribution arrangements with
third-party collaborators, and may also consider contracting with professional
detailing and sales organizations to perform promotional and/or
medical-scientific support functions for all our products. See Risk Factors.
Competition
Competition in the biopharmaceutical industry is intense and characterized by
ongoing research and development and technological change. We face competition
from many for-profit companies
and major universities and research institutions in the U.S. and abroad.
We will face competition from companies marketing existing products or
developing new products for diseases targeted by our technologies. Many of our
competitors have substantially greater resources, experience in conducting
pre-clinical studies and clinical trials and obtaining regulatory approvals for
their products, operating experience, research and development and marketing
capabilities and production capabilities than we do. Our products under
development may not compete successfully with existing products or products
under development by other companies, universities and other institutions. Our
competitors may succeed in obtaining FDA marketing approval for products more
rapidly than we do. Drug manufacturers that are first in the market with a
therapeutic for a specific indication generally obtain and maintain a
significant competitive advantage over later entrants. Accordingly, we believe
that the speed with which we develop products, complete the clinical trials and
approval processes and ultimately supply commercial quantities of the products
to the market will be an important competitive factor.
RELISTOR is
the first FDA-approved product for any indication involving OIC. We are,
however, aware of products in pre-clinical or clinical development that target
the side effects of opioid pain therapy. Adolor Corporation markets ENTEREG®
(alvimopan) for the treatment of postoperative ileus, and is re-evaluating an
entry-stage compound for OIC in chronic-pain patients. Sucampo Pharmaceuticals,
Inc., in collaboration with Takeda Pharmaceutical Company Limited, markets
AMITIZA®
(lubiprostone) for chronic idiopathic constipation and recently completed two
phase 3 pivotal clinical trials of this drug for opioid-induced bowel
dysfunction. In Europe Mundipharma International Limited markets TARGIN®
(oxycodone/naloxone), a combination of an opioid and a systemic opioid
antagonist. A Nektar Therapeutics-AstraZeneca collaboration has announced phase
2 results of an oral peripheral mu-opioid receptor antagonist in patients with
OIC and is developing a related combination product. Alkermes, Inc. recently
completed phase 1 clinical testing on an oral peripherally-restricted opioid
antagonist, and has a combination product in preclinical testing. Theravance,
Inc. recently completed phase 1 clinical testing of an oral peripheral mu-opioid
antagonist.
Radiation and surgery are two principal traditional forms of treatment for prostate cancer, to which our
PSMA-based development efforts are directed. If the disease spreads, hormone
(androgen) suppression therapy is often used to slow the cancer’s progression.
This form of treatment, however, can eventually become ineffective. We are aware
of several competitors who are developing alternative treatments for
castrate-resistant prostate cancer, some of which are directed against
PSMA.
Currently approved drugs for the treatment of HIV infection and AIDS have shown efficacy
alone and in conjunction with other agents, the latter of which we have not
demonstrated for PRO 140. We are aware of two approved drugs, Trimeris’
FUZEON®
and Pfizer’s SELZENTRY®,
designed to treat HIV infection by blocking viral entry.
HCV
infection is most commonly treated by a combination of interferon and
ribavirin. Seroconversion and/or sustained response to such therapies ranges
from 30-50%. Tolerability and route of administration for this therapy may
compromise a patient’s ability to persist with treatment for the 48-72 months
sometimes required. We are aware of several competitors who are developing small
molecule HCV antivirals, including viral-entry inhibition-based
treatments.
A significant amount of research in the biopharmaceutical field is also being
carried out at academic and
government institutions. An element of our research and development
strategy is to in-license technology and product candidates from academic and
government institutions. These institutions are sensitive to the commercial
value of their findings and pursue patent protection and negotiate
licensing arrangements to collect royalties for use of
technology they develop. These institutions may also market
competitive commercial products on their own or in collaboration with
competitors and compete with us in recruiting highly qualified scientific
personnel. Any resulting increase in the cost or decrease in the availability of
technology or product candidates from these institutions may adversely affect
our business strategy.
Competition with respect to our technologies and products is based on, among
other things, (i) product efficacy, safety, reliability, method of
administration, availability, price and clinical benefit relative to cost; (ii)
timing and scope of regulatory approval; (iii) sales, marketing and
manufacturing capabilities; (iv) collaborator capabilities; (v) insurance and
other reimbursement coverage; and (vi) patent protection.
Our competitive position will also depend on our ability to attract and retain
qualified personnel, obtain patent protection or otherwise develop proprietary
products or processes, and secure sufficient capital resources for the typically
substantial period between technological conception and commercial
sales.
Product
Liability
The testing, manufacturing and marketing of our product candidates and products
involves an inherent risk of product liability attributable to unwanted and
potentially serious health effects. To the extent we elect to test, manufacture
or market product candidates and products independently, we will bear the risk
of product liability directly. We have obtained product liability insurance
coverage in the amount of $10.0 million per occurrence, subject to a deductible
and a $10.0 million aggregate limitation. In addition, where the local statutory
requirements exceed the limits of our existing insurance or local policies of
insurance are required, we maintain additional clinical trial liability
insurance to meet these requirements. This insurance is subject to deductibles
and coverage limitations. We may not be able to continue to maintain insurance
at a reasonable cost, or in adequate amounts.
Human
Resources
At December 31, 2009, we had 204 full-time employees, 32 of whom hold Ph.D.
degrees, 8 of whom hold M.D. degrees and two of whom, including Dr. Paul J.
Maddon, our Chief Executive Officer and Chief Science Officer, hold both Ph.D.
and M.D. degrees. At that date, 158 employees were engaged in research and
development, medical, regulatory affairs and manufacturing activities and 46
were engaged in finance, legal, administration and business development. In
early 2010, we implemented a reduction in headcount, as a result of which we
have 178 full-time employees at March 1, 2010. We consider our relations with
our employees to be good. None of our employees is covered by a collective
bargaining agreement.
Item
1A. Risk Factors
Our business and operations entail a variety of serious risks and uncertainties,
including those described below.
Our
business is inherently risky.
We are subject to the risks of failure inherent in the development of product
candidates based on new technologies. We must complete successfully clinical
trials and obtain regulatory approvals for our product candidates as well as
additional formulations of and indications for RELISTOR. In the Japanese market,
we must rely on Ono to conduct successful clinical trials and obtain regulatory
approvals. Our other research and development programs, including those related
to PSMA and PRO 140, involve novel approaches to human therapeutics. There is
little precedent for the successful commercialization of products based on our
technologies, and there are a number of technological challenges that we must
overcome to complete most of our development efforts. We may not be able
successfully to develop further any of our products.
In addition to the risks we face in our research and development activities, and
our business as a publicly held commercial enterprise devoted to developing and
commercializing high-technology consumer products, the reacquisition of our
rights to RELISTOR presents us with substantial new risks, including the
following:
The
return of our rights to RELISTOR presents us with substantial new risks. For
example, if we are unable to establish satisfactory relationships with one or
more partners to develop and commercialize RELISTOR worldwide, we would need to
make significant investment to establish a sales and marketing infrastructure
and related staff, and in the meantime may be dependent on third parties for
their expertise in this area.
As a result of the termination of the Wyeth collaboration, we continue to face
all of the risks to which we are currently subject as well as the additional
risks attendant to establishing new collaborative or other relationships with
one or more partners to develop and commercialize RELISTOR. If we are unable to
do so, we will also face substantial risk in having sole responsibility for
developing and commercializing a regulated pharmaceutical product. Even with a
partner, significant investment, time and managerial resources may be required
to build a commercial infrastructure to co-promote, co-market or otherwise
market, sell and support a pharmaceutical product. To the extent not funded from
outside sources -- and in any case prior to securing any such funding -- these
efforts will increase our cash requirements and result in a higher cash burn
rate, which will have a material adverse effect on our financial resources
and operational results.
If, after the end of Wyeth’s involvement with the product, we undertake
development and commercialization of RELISTOR without one or more partners, the
financial and managerial resources necessary to transition to a commercial
organization would require us to divert resources from our development efforts,
including those for oral RELISTOR and our product candidates, to commercial
ones. Even if we establish relationships with one or more partners for these
tasks, we may have to divert resources from these programs to the extent we do
not fund commercial activities in full from those external sources.
Should we choose to commercialize RELISTOR directly, even in part of the world,
we may not be successful in developing an effective sales, marketing and
distribution infrastructure or in achieving sufficient market acceptance.
Alternatively, we may also consider contracting with a third party professional
pharmaceutical detailing and sales organization to perform all or certain
marketing functions. To the extent we enter into distribution, co-marketing,
co-promotion, detailing or licensing arrangements for our products, any revenues
we receive will depend in substantial part or primarily on the efforts of third
parties. We may not be able to control the amount and timing of marketing
resources these third parties devote to our products.
We
are dependent on Wyeth, Ono and other business partners to develop and
commercialize RELISTOR in their respective areas, exposing us to significant
risks. As a result of termination of our collaboration, Wyeth has a limited
ongoing commercial interest in RELISTOR during the transition.
We are and will be dependent upon Wyeth (during the transition of its
responsibilities to us pursuant to the Transition Agreement), Ono and any other
business partner(s) with which we may collaborate in the future to perform and
fund development, including clinical testing of RELISTOR, make related
regulatory filings and manufacture and market products in their respective
territories. Revenues from the sale of RELISTOR currently depend almost entirely
upon the efforts of Wyeth, which during the transition has significant
discretion in determining the efforts and resources it applies to sales of
RELISTOR. Ono will have similar discretion with respect to sales in Japan.
Neither may be effective in marketing such products, and these considerations
may apply to other business partners. Our business relationships with Wyeth, Ono
and other partners may not be scientifically, clinically or commercially
successful.
As a result of termination of
our
relationship with Wyeth, we are seeking alternative arrangements with one or
more other parties to develop and commercialize RELISTOR. We might also
seek alternative arrangements if our relationship with Ono were to
terminate. We may not be able to enter into such arrangements with other
suitable companies on acceptable terms or at all, in which event we would have
to develop sales and marketing organization and a distribution infrastructure,
neither of which we currently have, in order to continue to develop and
commercialize RELISTOR on our own. Developing these resources would be an
expensive and lengthy process and will have a material adverse effect on our
financial resources and profitability. Termination of our relationship with
Wyeth may also seriously compromise the development program for RELISTOR and
possibly our other product candidates, as we may experience significant delays
and may have to assume full funding and other responsibility for development and
commercialization. Any of these outcomes would result in delays in our ability
to distribute RELISTOR and would increase our expenses.
Wyeth’s development and commercialization obligations during the transition
period are limited by the Transition Agreement and are less extensive than its
obligations under the 2005 collaboration. The acquisition of Wyeth by Pfizer has
resulted in management and personnel changes, and by returning our RELISTOR
rights, Wyeth has a limited ongoing commercial interest in the RELISTOR
franchise. We cannot guarantee that Wyeth’s efforts during the transition will
achieve any particular level of success in marketing and sale, regulatory
approval or clinical development of RELISTOR.
We have had and may have future disagreements with Wyeth and Ono concerning
product development, marketing strategies, manufacturing and supply issues, and
rights relating to intellectual property. Both of them have significantly
greater financial and managerial resources than we do, which either could draw
upon in the event of a dispute. Disagreements between either of them and us
could lead to lengthy and expensive litigation or other dispute-resolution
proceedings as well as to extensive financial and operational consequences to
us, and have a material adverse effect on our business, results of operations
and financial condition. These considerations may apply to other business
partners with which we may collaborate in the future.
We
are subject to extensive regulation, which can be costly and time consuming and
can subject us to unanticipated fines and delays.
We and our products are subject to comprehensive regulation by the FDA and
comparable authorities in other countries. These agencies and other entities
regulate the pre-clinical and clinical testing, safety, effectiveness, approval,
manufacture, labeling, marketing, export, storage, recordkeeping, advertising,
promotion and other aspects of our products. If we violate regulatory
requirements at any stage, whether before or after marketing approval is
obtained, we may be subject to forced removal of a product from the market,
product seizure, civil and criminal penalties and other adverse consequences. We
cannot guarantee that approvals of proposed products, processes or facilities
will be granted on a timely basis, or at all. If we experience delays or
failures in obtaining approvals, commercialization of our product candidates
will be slowed or stopped. Even if we obtain regulatory approval, the approval
may include significant limitations on indicated uses for which the product
could be marketed or other significant marketing restrictions.
As a result of termination of the Wyeth collaboration, we will be responsible
for complying with these regulations and will be required to develop an
appropriate infrastructure to do so, to the extent we are unable to have such
tasks performed by one or more business partners.
If
we are unable to negotiate collaborative agreements, or if such arrangements are
delayed or are relatively unfavorable to us, our cash burn rate will increase
and our rate of product development could decrease.
We are pursuing significant collaborations, strategic partnerships or other
arrangements to continue worldwide development and commercialization of RELISTOR
following expiration of the transition period. Our overall business strategy
includes entering into collaborations with pharmaceutical and biotechnology
companies to develop and commercialize product candidates and technologies. We
may not be successful in negotiating such arrangements at all, in optimal time
frames or on favorable terms. In particular, if we do not enter into new
collaborative arrangements for RELISTOR on satisfactory terms and within the
timeframes established by the Transition Agreement -- and in any case prior to
entering into any such arrangements -- we will have to devote more of our
resources to clinical product development and product-launch activities, and our
cash burn rate will increase. In that event, we will need to take steps to
reduce our rate of product development for other programs, and may also have to
seek additional sources of capital. If we are not successful in negotiating
satisfactory new arrangements with respect to the RELISTOR franchise, these
challenges will be substantial. We cannot assure you that any
currently-contemplated or future collaboration or other initiatives for funding
our product candidate programs will be successfully concluded.
We
lack sales and marketing infrastructure and related staff, which will require
significant investment to establish and in the meantime may make us dependent on
third parties for their expertise in this area.
We have no established sales, marketing or distribution infrastructure.
Significant investment, time and managerial resources are required to build the
commercial infrastructure required to market, sell and support a pharmaceutical
product. Should we choose to commercialize RELISTOR or another product directly,
we may not be successful in developing an effective commercial infrastructure or
in achieving sufficient market acceptance. Alternatively, we may choose to
market and sell our products through distribution, co-marketing, co-promotion or
licensing arrangements with third parties. We may also consider contracting with
third party professional pharmaceutical detailing and sales organizations to
perform the marketing function for our products. To the extent that we enter
into such arrangements, any revenues we receive will depend on the efforts of
third parties. We may not control the amount and timing of marketing resources
these third parties devote to our products.
Our
products may face regulatory, legal or commercial challenges even after
approval.
Even if our products receive regulatory approval:
· They might not obtain labeling claims necessary
to make the product commercially viable (in general, labeling claims define the
medical conditions for which a drug
product
may be marketed, and are therefore very important to the commercial success of a
product), or may be required to carry “black box” or other warnings that
adversely
affect their commercial success.
· Approval may be limited to uses of the product
for treatment or prevention of diseases or conditions that are relatively less
financially advantageous to us than approval
of greater or different scope, or subject to an FDA-imposed Risk Evaluation and
Mitigation Strategy (REMS) that limits the sources from and conditions under
which they
may be dispensed.
· We or our collaborators might be required to
undertake post-marketing trials to verify the product’s efficacy or
safety.
· We, our collaborators or others might identify
side effects after the product is on the market.
· Efficacy or safety concerns regarding marketed
products may lead to product recalls, withdrawals of marketing approval,
reformulation of the product, additional pre-
clinical testing or clinical trials, changes in labeling of the product, the
need for additional marketing applications, declining sales or other adverse
events. For example, in
early 2009 the FDA identified a potential RELISTOR safety issue involving
gastrointestinal perforation. The agency is continuing to evaluate to determine
the need for any
regulatory action;
the appearance of a drug on this FDA list does not mean that the agency has
concluded that the drug has such a risk. These potential consequences may
occur whether or not the concerns originate from subsequent testing or other
activities by us, governmental regulators, other entities or organizations or
otherwise, and
whether or not they are scientifically justified.
· We or our collaborators might experience
manufacturing problems, which could have the same, similar or other
consequences.
· We and our collaborators will be subject to
ongoing FDA obligations and continuous regulatory review.
· If products lose previously received marketing
and other approvals, our financial results would be adversely
affected.
Competing
products in development may adversely affect acceptance of our
products.
We are aware of the following competition and potential competition to
RELISTOR:
· An Adolor Corporation-GlaxoSmithKline PLC
collaboration received FDA approval in 2008 for ENTEREG®
(alvimopan), an oral opioid antagonist for postoperative ileus
indicated “to accelerate the time to upper and lower gastrointestinal recovery
following partial large or small bowel resection surgery with primary
anastomosis.” Adolor is
also re-evaluating an entry-stage compound for OIC in chronic-pain
patients.
· A Sucampo Pharmaceuticals, Inc.-Takeda
Pharmaceutical Company Limited collaboration, markets AMITIZA®
(lubiprostone) for chronic idiopathic constipation and
recently completed two phase 3 pivotal clinical trials of this drug for
opioid-induced bowel dysfunction.
· In Europe, Mundipharma International Limited
markets TARGIN®
(oxycodone/naloxone), a combination of an opioid and a systemic opioid
antagonist.
· A Nektar Therapeutics-AstraZeneca PLC
collaboration recently announced phase 2 results of an oral peripheral
mu-opioid receptor antagonist in patients with
OIC,
and is developing a related combination product. AstraZeneca is a leader in
gastrointestinal medicine, and their collaboration may have a time-to-market
advantage over
us with respect to an oral therapy for OIC in chronic-pain
patients.
· Alkermes, Inc. recently completed phase 1
clinical testing of an oral peripherally-restricted opioid antagonist, and has a
combination product in preclinical testing.
· Theravance, Inc. recently completed phase 1
clinical testing of and oral peripheral mu-opioid antagonist.
Any of these approved products or product candidates may achieve a significant
competitive advantage relative to our product. In any event, the existing or
future marketing and sales capabilities of these competitors may impair our
ability to compete effectively in the market.
Radiation and surgery are two principal traditional forms of treatment for
prostate cancer, to which our PSMA-based development efforts are directed. If
the disease spreads, hormone (androgen) suppression therapy is often used to
slow the cancer’s progression. This form of treatment, however, can eventually
become ineffective. We are aware of several competitors who are developing
alternative treatments for castrate-resistant prostate cancer, some of which are
directed against PSMA.
In the case of PRO 140, currently-approved drugs for the treatment of HIV
infection and AIDS have shown efficacy alone and in conjunction with other
agents, the latter of which we have not demonstrated for PRO 140. We are aware
of two approved drugs, Trimeris’ FUZEON® and
Pfizer’s SELZENTRY®,
designed to treat HIV infection by blocking viral entry. We are also aware of
various HCV drugs in pre-clinical or clinical development.
Both
our immediate need to transition RELISTOR and our long-term strategy with
respect to RELISTOR and our other product candidates may require us to obtain
additional financing. Our access to capital funding is uncertain.
As of December 31, 2009, we had cash, cash equivalents and marketable
securities, including non-current portion, totaling $96.2 million. During the
year ended December 31, 2009, we had a net loss of $30.6 million and cash used
in operating activities was $49.8 million. Additional expenses we incur in
future development and commercialization of RELISTOR will result in accelerating
diminution of our cash and growth of our losses to the extent those expenses are
not funded from outside sources. By reacquiring the rights to RELISTOR from
Wyeth, we have become responsible for the future development and
commercialization of the currently-marketed drug after the transition and for
new formulations. We do not have committed external sources of funding for these
responsibilities, and are seeking additional external funding through
collaborative, license or other agreements with one or more pharmaceutical
companies. To the extent we are not successful in these efforts, we will have to
fund them from cash on hand.
With regard to both RELISTOR and our other product candidates, we expect to
continue to incur significant development expenditures. We cannot predict when
we will need additional funds, how much we will need, the form any financing may
take (such as securities issuance or royalty or other financing), or whether
additional funds will be available at all, especially in light of current
conditions in global credit and financial markets. Our need for future funding
will depend on numerous factors, such as the availability of new product
development projects or other opportunities which we cannot predict, and many of
which are outside our control. In particular, we cannot assure you that any
currently-contemplated or future initiatives for funding our product candidate
programs will be successful.
Our access to capital funding is always uncertain. Stresses in international
markets are still affecting access to capital. We may not be able at the
necessary time to obtain additional funding on acceptable terms, or at all. Our
inability to raise additional capital on terms reasonably acceptable to us would
seriously jeopardize our business.
If we raise funds by issuing and selling securities, it may be on terms that are
not favorable to existing stockholders. If we raise funds by selling equity
securities, current stockholders will be diluted, and new investors could have
rights superior to existing stockholders. Raising funds by selling debt
securities often entails significant restrictive covenants and repayment
obligations.
If
testing does not yield successful results, our products will not be
approved.
Regulatory approvals are necessary before product candidates can be marketed. To
obtain them, we or our collaborators must demonstrate a product’s safety and
efficacy through extensive pre-clinical and clinical testing. Numerous adverse
events may arise during, or as a result of, the testing process, such
as:
|
|
· results
of pre-clinical studies being inconclusive or not indicative of results in
human clinical trials;
|
|
|
· potential
products not having the desired efficacy or having undesirable side
effects or other characteristics that preclude marketing approval or limit
their commercial use if approved;
|
|
|
· after
reviewing test results, we or our collaborators may abandon projects which
we previously believed to be promising;
and
|
|
|
· we,
our collaborators or regulators may suspend or terminate clinical trials
if we or they believe that the participating subjects are being exposed to
unacceptable health risks.
|
Clinical testing is very expensive and can take many years. Results attained in
early human clinical trials may not be indicative of results in later clinical
trials. In addition, many of our investigational or experimental drugs, such as
PRO 140 and the PSMA product candidates, are at an early stage of development,
and successful commercialization of early stage product candidates requires
significant research, development, testing and approvals by regulators, and
additional investment. Our products in the research or pre-clinical development
stage may not yield results that would permit or justify clinical testing. Our
failure to demonstrate adequately the safety and efficacy of a product under
development would delay or prevent marketing approval, which could adversely
affect our operating results and credibility.
A
setback in clinical development programs could adversely affect us.
We and Wyeth continue to conduct clinical trials of RELISTOR. If the results of
these or future trials are not satisfactory, we encounter problems enrolling
subjects, clinical trial supply issues or other difficulties arise, or we
experience setbacks in developing drug formulations, including raw
material-supply, manufacturing or stability difficulties, our entire RELISTOR
development program could be adversely affected, resulting in delays in trials
or regulatory filings for further marketing approval. Conducting additional
clinical trials or making significant revisions to our clinical development plan
would lead to delays in regulatory filings. If clinical trials indicate a
serious problem with the safety or efficacy of a RELISTOR product, we, Wyeth,
Ono or another partner may stop development or commercialization of affected
products. Since RELISTOR is our only approved product, any setback of these
types could have a material adverse effect on our business, results of
operations and financial condition.
Ono is conducting required clinical trials with Japanese patients to obtain
regulatory approval of RELISTOR in Japan. There can be no assurance that these
clinical trials will yield results adequate for that regulatory
approval.
We are conducting clinical trials of PRO 140, PSMA ADC and prostate cancer
vaccine candidates. If the results of these or future clinical studies
of our candidates are not satisfactory, we would need to reconfigure our
clinical trial programs to conduct additional trials or abandon the program
involved. Because our vaccine product candidates may be deemed to involve gene
therapy, a relatively new technology that has not been extensively tested in
humans, regulatory requirements applicable to them may be unclear, or subject to
substantial regulatory review that delays the development and approval process
generally.
Our
clinical trials could take longer than we expect.
Projections that we publicly announce of commencement and duration of clinical
trials may not be certain. For example, we have experienced clinical trial
delays in the past as a result of slower than anticipated enrollment. These
delays may recur. Delays can be caused by, among other things:
|
|
· deaths
or other adverse medical events involving subjects in our clinical
trials;
|
|
|
· regulatory
or patent issues;
|
|
|
· interim
or final results of ongoing clinical
trials;
|
|
|
· failure
to enroll clinical sites as
expected;
|
|
|
· competition
for enrollment from clinical trials conducted by others in similar
indications;
|
|
|
· scheduling
conflicts with participating clinicians and clinical
institutions;
|
|
|
· disagreements,
disputes or other matters arising from
collaborations;
|
|
|
· our
inability to obtain additional funding when needed;
and
|
|
|
· manufacturing
problems.
|
We have limited experience in conducting clinical trials, and we rely on others
to conduct, supervise or monitor some or all aspects of some of our clinical
trials. In addition, certain clinical trials for our product candidates may be
conducted by government-sponsored agencies, and consequently will be dependent
on governmental participation and funding. Under our 2005 collaboration
agreement with Wyeth, Wyeth had the responsibility to conduct some of the
clinical trials, including all trials outside of the United States other than
Japan, where Ono has the responsibility for clinical trials. Upon completion of
the transition, we will have responsibility for these trials, or will contract
with other parties to conduct them. We have less control over the timing and
other aspects of these clinical trials than if we conducted them entirely on our
own.
Our
product candidates may not obtain regulatory approvals needed for
marketing.
None of our product candidates other than RELISTOR has been approved by
applicable regulatory authorities for marketing. The process of obtaining FDA
and foreign regulatory approvals often takes many years and can vary
substantially based upon the type, complexity and novelty of the products
involved. We have had only limited experience in filing and pursuing
applications and other submissions necessary to gain marketing approvals.
Products under development may never obtain marketing approval from the FDA
or other regulatory authorities necessary for
commercialization.
The commercial success of our products will depend upon their acceptance by the
medical community and third party payors as clinically useful, cost effective
and safe. If health care providers believe that patients can be managed
adequately with alternative, currently available therapies, they may not
prescribe our products, especially if the alternative therapies are viewed as
more effective, as having a better safety or tolerability profile, as being more
convenient to the patient or health care providers or as being less expensive.
For pharmaceuticals administered in an institutional setting, the ability of the
institution to be adequately reimbursed could also play a significant role in
demand for our products. Even if our products obtain marketing approval, they
may not achieve market acceptance. If any of our products do not achieve market
acceptance, we will likely lose our entire investment in that
product.
Marketplace
acceptance will depend in part on competition in our industry, which is
intense.
The extent to which any of our products achieves market acceptance will depend
on competitive factors. Competition in our industry is intense, and it is
accentuated by the rapid pace of technological development. There are currently
marketed products that will compete with the product candidates that we are
developing, including AIDS drugs and chemotherapy drugs for treating cancer.
There are also product candidates in pre-clinical or clinical development that
target the side effects of opioid pain therapy, and a marketed product for the
treatment of post-operative ileus, could compete with
RELISTOR. Many of our competitors have substantially greater research and
development capabilities and experience and greater manufacturing, marketing,
financial and managerial resources than we do. These competitors may develop
products that are superior to those we are developing and render our products or
technologies non-competitive or obsolete. If our product candidates receive
marketing approval but cannot compete effectively in the marketplace, our
operating results and financial position would suffer.
Competition with respect to our
technologies and products is based on, among other things, (i) product efficacy,
safety, reliability, method of administration, availability, price and clinical
benefit relative to cost; (ii) timing and scope of regulatory approval; (iii)
sales, marketing and manufacturing capabilities; (iv) collaborator capabilities;
(v) insurance and other reimbursement coverage; and (vi) patent protection.
Competitive disadvantages in any of these factors could materially harm our
business and financial condition.
If
we are unable to obtain sufficient quantities of the raw and bulk materials
needed to make our products, our product development and commercialization could
be slowed or stopped.
Wyeth, during the transition, and thereafter we and/or any partner(s) may not be
able to fulfill manufacturing obligations for RELISTOR, either on our own or
through third-party suppliers. Our existing arrangements with suppliers for our
other product candidates may not result in the supply of sufficient quantities
of our product candidates needed to accomplish our clinical development
programs, and we may not have the right or capability to manufacture sufficient
quantities of these products to meet our needs if our suppliers are unable or
unwilling to do so. We currently obtain supplies of critical raw materials used
in production of our product candidates from single sources. We do not have
long-term contracts with any of these suppliers. Any delay or disruption in the
availability of raw materials would slow or stop product development and
commercialization of the relevant product. A delay or disruption of supplies of
RELISTOR would have a material adverse effect on the RELISTOR franchise, and
therefore on our business as a whole.
We
have limited manufacturing capabilities, which could adversely affect our
ability to commercialize products.
Under the Transition Agreement with Wyeth, we will be responsible for obtaining
supplies of RELISTOR after the end of Wyeth’s transition obligations, which
include supplying us and our collaboration partner(s) with finished subcutaneous
drug product and API remaining in Wyeth’s inventories. Going forward, we expect
we or collaboration partner(s) or licensee(s) will extend current relationships
or contract with one or more other CMOs for supply of RELISTOR API and
subcutaneous and oral finished drug product. These arrangements may not be on
optimally-advantageous terms, and will subject us to risks that the
counterparties may not perform optimally in terms of quality or
reliability.
With respect to our other product candidates, our limited manufacturing
capabilities may result in increased costs of production or delay product
development or commercialization. In order to commercialize our product
candidates successfully, we or our collaborators would need to be able to
manufacture products in commercial quantities, in compliance with regulatory
requirements, at acceptable costs and in a timely manner. Manufacture of our
product candidates can be complex, difficult to accomplish even in small
quantities, difficult to scale-up for large-scale production and subject to
delays, inefficiencies and low yields of quality products. The cost of
manufacturing some of our products may make them prohibitively expensive. If
adequate supplies of any of our product candidates or related materials are not
available to us on a timely basis or at all, our clinical trials could be
seriously delayed, since these materials are time consuming to manufacture and
cannot be readily obtained from third-party sources.
We operate pilot-scale manufacturing facilities for the production of vaccines
and recombinant proteins. These facilities will not be sufficient for late-stage
clinical trials for these types of product candidates or commercial-scale
manufacturing. We may be required to expand further our manufacturing staff and
facilities, obtain new facilities or contract with corporate collaborators or
other third parties to assist with production.
In the event that we decide to establish a commercial-scale manufacturing
facility for products that may be approved in the future, we will require
substantial additional funds and will be required to hire and train significant
numbers of employees and comply with applicable regulations, which are
extensive. We may not be able to build a manufacturing facility that both meets
regulatory requirements and is sufficient for our clinical trials or commercial
scale manufacturing.
We have entered into arrangements with third parties for the manufacture of some
of our product candidates and, in some cases, new means of administration for
these product candidates. Our third-party sourcing strategy may not result in a
cost-effective means for manufacturing products. In employing third-party
manufacturers, we do not control many aspects of the manufacturing process,
including compliance with the FDA’s current Good Manufacturing Practices and
other regulatory requirements. We may not be able to obtain adequate supplies
from third-party manufacturers in a timely fashion for development or
commercialization purposes, and commercial quantities of products may not be
available from contract manufacturers at acceptable costs.
We
are dependent on our patents and other intellectual property rights. The
validity, enforceability and commercial value of these rights are highly
uncertain.
Our success is dependent in part upon obtaining, maintaining and enforcing
patent and other intellectual property rights. The patent position of
biotechnology and pharmaceutical firms is highly uncertain and involves many
complex legal and technical issues. There is no clear policy involving the
breadth of claims allowed, or the degree of protection afforded, under patents
in this area. Accordingly, patent applications owned by or licensed to us may
not result in patents being issued. We are aware of others who have patent
applications or patents containing claims similar to or overlapping those in our
patents and patent applications. We do not expect to know for several years the
relative strength or scope of our patent position. Patents that we own or
license may not enable us to preclude competitors from commercializing drugs,
and consequently may not provide us with any meaningful competitive
advantage.
We own or have licenses to several issued patents. The issuance of a patent,
however, is not conclusive as to its validity or enforceability, which can be
challenged in litigation. Our patents may be successfully challenged. We may
incur substantial costs in litigation seeking to uphold the validity of patents
or to prevent infringement. If the outcome of litigation is adverse to us, third
parties may be able to use our patented invention without payment to us. Third
parties may also avoid our patents through design innovation.
Most of our product candidates, including RELISTOR, PRO 140 and our PSMA and HCV
program candidates, incorporate to some degree intellectual property licensed
from third parties. We can lose the right to patents and other intellectual
property licensed to us if the related license agreement is terminated due to a
breach by us or otherwise. Our ability, and that of our collaboration partners,
to commercialize products incorporating licensed intellectual property would be
impaired if the related license agreements were terminated.
The license agreements from which we derive or out-license intellectual property
provide for various royalty, milestone and other payment, commercialization,
sublicensing, patent prosecution and enforcement, insurance, indemnification and
other obligations and rights, and are subject to certain reservations of rights.
While we generally have the right to defend and enforce patents licensed by us,
either in the first instance or if the licensor chooses not to do so, we must
usually bear the cost of doing so. As a result of the Transition Agreement, we
will be required to defend and enforce our RELISTOR patents, an obligation that
Wyeth undertook in the 2005 collaboration agreement. With respect to Japan, Ono
has certain limited rights to prosecute, maintain and enforce relevant
intellectual property. With most of our in-licenses, the licensor bears the cost
of engaging in all of these activities, although we may share in those costs
under specified circumstances.
We also rely on unpatented technology, trade secrets and confidential
information. Third parties may independently develop substantially equivalent
information and techniques or otherwise gain access to our technology or
disclose our technology, and we may be unable to effectively protect our rights
in unpatented technology, trade secrets and confidential information. We require
each of our employees, consultants and advisors to execute a confidentiality
agreement at the commencement of an employment or consulting relationship with
us. These agreements may, however, not provide effective protection in the event
of unauthorized use or disclosure of confidential information.
If
we do not achieve milestones or satisfy conditions regarding some of our product
candidates, we may not maintain our rights under related licenses.
We are required to make substantial cash payments, achieve milestones and
satisfy other conditions, including filing for and obtaining marketing approvals
and introducing products, to maintain rights under our intellectual property
licenses. Due to the nature of these agreements and the uncertainties of
research and development, we may not be able to achieve milestones or satisfy
conditions to which we have contractually committed, and as a result may be
unable to maintain our rights under these licenses. If we do not comply with our
license agreements, the licensors may terminate them, which could result in our
losing our rights to, and therefore being unable to commercialize, related
products.
If
we infringe third-party patent or other intellectual property rights, we may
need to alter or terminate a product development program.
There may be patent or other intellectual property rights belonging to others
that require us to alter our products, pay licensing fees or cease certain
activities. If our products infringe patent or other intellectual property
rights of others, the owners of those rights could bring legal actions against
us claiming damages and seeking to enjoin manufacturing and marketing of the
affected products. If these legal actions are successful, in addition to any
potential liability for damages, we could be required to obtain a license in
order to continue to manufacture or market the affected products. We may not
prevail in any action brought against us, and any license required under any
rights that we infringe may not be available on acceptable terms or at all. We
are aware of intellectual property rights held by third parties that relate to
products or technologies we are developing. For example, we are aware of other
groups investigating methylnaltrexone and other peripheral opioid antagonists,
PSMA or related compounds and CCR5 monoclonal antibodies and of patents held,
and patent applications filed, by these groups in those areas. While the
validity of these issued patents, patentability of these pending patent
applications and applicability of any of them to our programs are uncertain, if
asserted against us, any related patent or other intellectual property rights
could adversely affect our ability to commercialize our products.
The research, development and commercialization of a biopharmaceutical often
involve alternative development and optimization routes, which are presented at
various stages in the development process. The preferred routes cannot be
predicted at the outset of a research and development program because they will
depend on subsequent discoveries and test results. There are numerous
third-party patents in our field, and we may need to obtain a license under a
patent in order to pursue the preferred development route of one or more of our
products. The need to obtain a license would decrease the ultimate profitability
of the applicable product. If we cannot negotiate a license, we might have to
pursue a less desirable development route or terminate the program
altogether.
We
are dependent upon third parties for a variety of functions. These arrangements
may not provide us with the benefits we expect.
We rely in part on third parties to perform a variety of functions. We are party
to numerous agreements which place substantial responsibility on clinical
research organizations, consultants and other service providers for the
development of our products. We also rely on medical and academic institutions
to perform aspects of our clinical trials of product candidates. In addition, an
element of our research and development strategy is to in-license technology and
product candidates from academic and government institutions in order to
minimize investments in early research. We have entered into agreements under
which we have depended on Wyeth and Ono, respectively, for the commercialization
and development of RELISTOR. We may not be able to replace the benefits to us of
the Wyeth agreement on attractive terms. We may not be able to maintain our
relationships with Ono or new partners, or establish new ones on beneficial
terms. We may not be able to enter new arrangements without undue delays or
expenditures, and these arrangements may not allow us to compete
successfully.
We
are exposed to product liability claims, and in the future may not be able to
obtain insurance against claims at a reasonable cost or at all. This exposure
may increase with our reacquisition of the rights to RELISTOR to the extent we
are not indemnified by a new partner.
Our business exposes us to product liability risks, which are inherent in the
testing, manufacturing, marketing and sale of pharmaceutical products. We may
not be able to avoid product liability exposure. If a product liability claim is
successfully brought against us, our financial position may be adversely
affected. Pursuant to the Transition Agreement, we will assume responsibility
for product liability risks arising from marketing and sales of RELISTOR, which
Wyeth had borne under our 2005 collaboration.
Product liability insurance for the biopharmaceutical industry is generally
expensive, when available at all. We have obtained product liability insurance
in the amount of $10.0 million per occurrence, subject to a deductible and a
$10.0 million annual aggregate limitation. Where local statutory requirements
exceed the limits of our existing insurance or where local policies of insurance
are required, we maintain additional clinical trial liability insurance to meet
these requirements. Our current insurance coverage may not be adequate to cover
claims brought against us. Some of our license and other agreements require us
to obtain product liability insurance. Adequate insurance coverage may not be
available to us at a reasonable cost in the future.
We
handle hazardous materials and must comply with environmental laws and
regulations, which can be expensive and restrict how we do business. If we are
involved in a hazardous waste spill or other accident, we could be liable for
damages, penalties or other forms of censure.
Our research and development work and manufacturing processes involve the use of
hazardous, controlled and radioactive materials. We are subject to federal,
state and local laws and regulations governing the use, manufacture, storage,
handling and disposal of these materials. Despite procedures that we implement
for handling and disposing of these materials, we cannot eliminate the risk of
accidental contamination or injury. In the event of a hazardous waste spill or
other accident, we could be liable for damages, penalties or other forms of
censure. We may be required to incur significant costs to comply with
environmental laws and regulations in the future.
If
we lose key management and scientific personnel on whom we depend, our business
could suffer.
We are dependent upon our key management and scientific personnel. In
particular, the loss of Dr. Maddon could cause our management and operations to
suffer. Our employment agreement with Dr. Maddon is effective on a year-to-year
basis, subject to automatic renewal unless either party terminates. Employment
agreements do not assure the continued employment of an employee. We maintain
key-man life insurance on Dr. Maddon which is not necessarily related to his
value to the Company at any given time.
Competition for qualified employees among companies in the biopharmaceutical
industry is intense. Our future success depends upon our ability to attract,
retain and motivate highly skilled employees. In order to commercialize our
products successfully, we may be required to expand substantially our personnel,
particularly in the areas of manufacturing, clinical trials management,
regulatory affairs, business development and marketing. We may not be successful
in hiring or retaining qualified personnel.
If
health care reform measures are enacted, our operating results and our ability
to commercialize products could be adversely affected.
In recent years, there have been numerous proposals to change the health care
system in the U.S. and in other jurisdictions. Some of these proposals have
included measures that would change the nature of and regulatory requirements
relating to drug discovery, clinical testing and regulatory approvals, limit or
eliminate payments for medical procedures and treatments, or subject the pricing
of pharmaceuticals to government control. Outside the U.S., and
particularly in the E.U., the pricing of prescription pharmaceuticals is
subject to governmental control. In addition, as a result of the trend towards
managed health care in the U.S., as well as legislative proposals to reduce
government insurance programs, third-party payors are increasingly attempting to
contain health care costs by limiting both coverage and the level of
reimbursement of new drug products. Consequently, significant uncertainty exists
as to the reimbursement status of newly approved health care
products.
If we or any of our collaborators succeed in bringing one or more of our
products to market, third party payors may establish and maintain price levels
insufficient for us to realize an appropriate return on our investment in
product development. Significant changes in the health care system in the U.S.
or elsewhere, including changes resulting from adverse trends in third-party
reimbursement programs, could have a material adverse effect on our operating
results and our ability to raise capital and commercialize
products.
A
substantial portion of our funding has come from federal government grants and
research contracts. We cannot rely on these grants or contracts as a continuing
source of funds.
A substantial portion of our revenues to date, albeit decreasing since 2006, has
been derived from federal government grants and research contracts. During the
last three years, we generated revenues from awards made to us by the NIH
between 2003 and 2009, to partially fund some of our programs. We cannot rely on
grants or additional contracts as a continuing source of funds. Funds available
under these grants and contracts must be applied by us toward the research and
development programs specified by the government rather than for all our
programs generally. The government’s obligation to make payments under these
grants and contracts is subject to appropriation by the U.S. Congress for
funding in each year. It is possible that Congress or the government agencies
that administer these government research programs will decide to scale back
these programs or terminate them due to their own budgetary constraints.
Additionally, these grants and research contracts are subject to adjustment
based upon the results of periodic audits performed on behalf of the granting
authority. Consequently, the government may not award grants or research
contracts to us in the future, and any amounts that we derive from existing
awards may be less than those received to date. In those circumstances, we would
need to provide funding on our own, obtain other funding, or scale back the
affected program. In particular, we cannot assure you that any
currently-contemplated or future efforts to obtain funding for our product
candidate programs through government grants or contracts will be successful, or
that any such arrangements which we do conclude will supply us with sufficient
funds to complete our development programs without providing additional funding
on our own or obtaining other funding.
We
have a history of operating losses, and we may never be profitable.
We have incurred substantial losses since our inception. As of December 31,
2009, we had an accumulated deficit of $329.3 million. We have derived no
significant revenues from product sales or royalties. We may not achieve
significant product sales or royalty revenue for a number of years, if ever. We
expect to continue to incur operating losses in the future, which could increase
significantly if we attempt to develop and commercialize RELISTOR without
adequate collaboration and/or financial arrangements and, at the same time,
expand our clinical trial programs and other product development
efforts.
Our ability to achieve and sustain profitability is dependent in part on
obtaining regulatory approval for and then commercializing our products, either
alone or with others. We may not be able to develop and commercialize products
beyond subcutaneous RELISTOR. Our operations may not be profitable even if any
of our other products under development are commercialized. Additional expenses
we incur in future development and commercialization of RELISTOR may cause our
losses to grow and to accelerate.
Our
stock price has a history of volatility. You should consider an investment in
our stock as risky and invest only if you can withstand a significant
loss.
Our stock price has a history of significant volatility. Between January 1, 2007
and December 31, 2009, our stock price has ranged from $30.31 to $3.53 per
share. Between January 1, 2010 and March 5, 2010, it has ranged from $4.99 to
$4.16 per share. Historically, our stock price has fluctuated through an even
greater range. At times, our stock price has been volatile even in the absence
of significant news or developments relating to us. The stock prices of
biotechnology companies and the stock market generally have been subject to
dramatic price swings in recent years, and financial and market conditions
in the past two years have resulted in widespread pressures on securities
of issuers throughout the world economy. Factors that may have a significant
impact on the market price of our common stock include:
· the results of clinical trials and pre-clinical
studies involving our products or those of our competitors;
· changes in the status of any of our drug
development programs, including delays in clinical trials or program
terminations;
· developments regarding our efforts to achieve
marketing approval for our products;
· developments in our relationships with Wyeth,
Ono and any other business partner(s) with which we may collaborate in the
future regarding the development and
commercialization of RELISTOR;
· developments in current or future relationships
with other collaborative partners with respect to other products and
candidates;
· announcements of technological innovations or
new commercial products by us, our collaborators or our
competitors;
· developments in patent or other proprietary
rights;
· governmental regulation;
· changes in reimbursement policies or health care
legislation;
· public concern as to the safety and efficacy of
products developed by us, our collaborators or our competitors;
· our ability to fund ongoing
operations;
· fluctuations in our operating results;
and
· general market conditions.
Purchases of our common shares pursuant to our share repurchase program may,
depending on their timing, volume and form, result in our stock price to be
higher than it would be in the absence of such purchases. If purchases under the
program are discontinued, our stock price may fall.
Our
principal stockholders are able to exert significant influence over matters
submitted to stockholders for approval.
At December 31, 2009, our directors and executive officers and stockholders
affiliated with Tudor Investment Corporation together beneficially owned or
controlled approximately one-fifth of our outstanding shares of common stock. At
that date, our five largest stockholders, excluding our directors and executive
officers and stockholders affiliated with Tudor, beneficially owned or
controlled in the aggregate approximately 45% of our outstanding shares. Our
directors and executive officers and Tudor-related stockholders, should they
choose to act together, could exert significant influence in determining the
outcome of corporate actions requiring stockholder approval and otherwise
control our business. This control could have the effect of delaying or
preventing a change in control of us and, consequently, could adversely affect
the market price of our common stock. Other significant but unrelated
stockholders could also exert influence in such matters.
Anti-takeover
provisions may make the removal of our Board of Directors and/or management more
difficult, and consequently, may discourage hostile bids for control of our
company that may be beneficial to our stockholders.
Our Board of Directors is authorized, without further stockholder action, to
issue from time to time shares of preferred stock in one or more designated
series or classes. The issuance of preferred stock, as well as provisions in
certain of our stock options that provide for acceleration of exercisability
upon a change of control, and Section 203 and other provisions of the Delaware
General Corporation Law could:
|
|
· make
the takeover of Progenics or the removal of our Board of Directors or
management more difficult;
|
|
|
· discourage
hostile bids for control of Progenics in which stockholders may receive a
premium for their shares of common stock;
and
|
|
|
· otherwise
dilute the rights of holders of our common stock and depress the market
price of our common stock.
|
If
there are substantial sales of our common stock, the market price of our common
stock could decline.
Sales of substantial
numbers of shares of common stock could cause a decline in the market price of
our stock. We require substantial external funding to finance our research and
development programs and may seek such funding through the issuance and sale of
our common stock. In addition, some of our other stockholders are entitled to
require us to register their shares of common stock for offer or sale to the
public. We have filed Form S-8 registration statements registering shares
issuable pursuant to our equity compensation plans and periodically seek to
increase the amount of securities available under these plans. Any sales by
existing stockholders or holders of options, or other rights, may have an
adverse effect on our ability to raise capital and may adversely affect the
market price of our common stock.
Item
1B.
Unresolved Staff
Comments
There were no unresolved SEC staff comments regarding our periodic or current
reports under the Exchange Act as of December 31, 2009.
Item
2. Properties
As of December 31, 2009, we occupy in total approximately 131,500 square feet of
laboratory, manufacturing and office space on a single campus in Tarrytown, New
York, under five lease agreements, the majority of which expired December 31,
2009, while the remaining leases were to expire in either 2012 or 2014. In
October 2009, we renewed to December 31, 2020, our lease agreement for a total
of 149,300 square feet of office and laboratory space in the same
campus.
In addition to rents due under these arrangements, we are obligated to pay
additional facilities charges, including utilities, taxes and operating
expenses.
Item
3. Legal Proceedings
We are not a party to any material legal proceedings.
Item 4. (Removed and Reserved under recent SEC
rules)
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder
Matters and Issuer Purchases of Equity Securities
Price
Range of Common Stock
Our common stock is quoted on The NASDAQ Stock Market LLC under the symbol
“PGNX.” The following table sets forth, for the periods indicated, the high and
low sales price per share of the common stock, as reported on The NASDAQ Stock
Market LLC. Such prices reflect inter-dealer prices, without retail mark-up,
markdown or commission and may not represent actual transactions.
|
High
|
Low
|
|||||
|
2009:
|
Fourth
quarter
|
$
|
5.48
|
$
|
3.53
|
|
|
Third
quarter
|
6.14
|
4.92
|
||||
|
Second
quarter
|
7.05
|
4.50
|
||||
|
First
quarter
|
10.81
|
5.08
|
||||
|
2008:
|
Fourth
quarter
|
14.10
|
6.77
|
|||
|
Third
quarter
|
17.50
|
11.88
|
||||
|
Second
quarter
|
17.94
|
6.66
|
||||
|
First
quarter
|
19.25
|
4.33
|
On March 5, 2010, the last sale price for our common stock, as reported by The
NASDAQ Stock Market LLC, was $4.65. There were approximately 103 holders of record of our
common stock as of March 5, 2010.
Comparative
Stock Performance Graph
The graph below compares, for the past five years, the cumulative stockholder
return on our common stock with the cumulative stockholder return of (i) the
Nasdaq Stock Market (U.S.) Index and (ii) the Nasdaq Pharmaceutical Index,
assuming the investment in each equaled $100 on December 31, 2004.
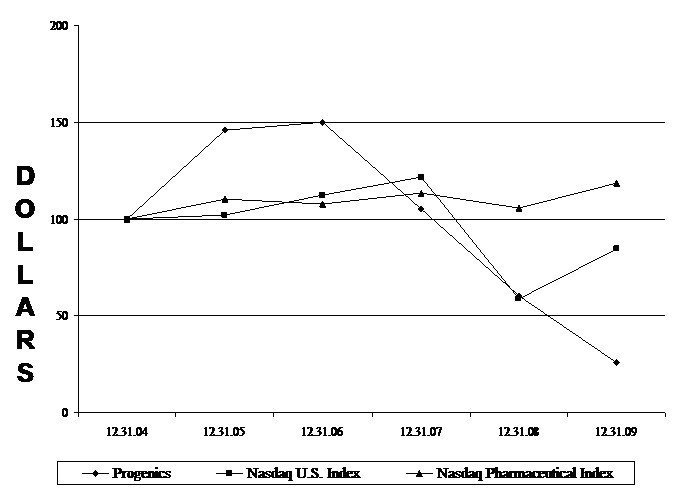
Dividends
We have not paid any dividends since our inception and currently anticipate that
all earnings, if any, will be retained for development of our business and that
no dividends on our common stock will be declared in the foreseeable
future.
Item
6. Selected Financial Data
The selected financial data presented below as of December 31, 2009 and 2008 and
for each of the three years in the period ended December 31, 2009 are derived
from our audited financial statements, included elsewhere herein. The selected
financial data presented below with respect to the balance sheet data as of
December 31, 2007, 2006 and 2005 and for each of the two years in the period
ended December 31, 2006 are derived from our audited financial statements not
included herein. The data set forth below should be read in conjunction with
Management’s Discussion and Analysis of Financial Condition and Results of
Operations and the Financial Statements and related Notes included elsewhere
herein.
|
Years
Ended December 31,
|
||||||||||||||||||||
|
2009
|
2008
|
2007
|
2006
|
2005
|
||||||||||||||||
|
(in
thousands, except per share data)
|
||||||||||||||||||||
|
Statement
of Operations Data:
|
||||||||||||||||||||
|
Revenues:
|
||||||||||||||||||||
|
Research
and development
|
$ | 44,351 | $ | 59,885 | $ | 65,455 | $ | 58,415 | $ | - | ||||||||||
|
Royalty
income
|
2,372 | 146 | - | - | - | |||||||||||||||
|
Research
and development from joint venture
|
- | - | - | - | 988 | |||||||||||||||
|
Research
grants and contract
|
1,968 | 7,460 | 10,075 | 11,418 | 8,432 | |||||||||||||||
|
Other
revenues
|
256 | 180 | 116 | 73 | 66 | |||||||||||||||
|
Total
revenues
|
48,947 | 67,671 | 75,646 | 69,906 | 9,486 | |||||||||||||||
|
Expenses:
|
||||||||||||||||||||
|
Research
and development
|
49,798 | 82,290 | 95,234 | 61,711 | 43,419 | |||||||||||||||
|
In-process
research and development
|
- | - | - | 13,209 | - | |||||||||||||||
|
License
fees – research and development
|
1,058 | 2,830 | 942 | 390 | 20,418 | |||||||||||||||
|
General
and administrative
|
25,106 | 28,834 | 27,901 | 22,259 | 13,565 | |||||||||||||||
|
Royalty
expense
|
237 | 15 | - | - | - | |||||||||||||||
|
Loss
in joint venture
|
- | - | - | 121 | 1,863 | |||||||||||||||
|
Depreciation
and amortization
|
5,078 | 4,609 | 3,027 | 1,535 | 1,748 | |||||||||||||||
|
Total
expenses
|
81,277 | 118,578 | 127,104 | 99,225 | 81,013 | |||||||||||||||
|
Operating
loss
|
(32,330 | ) | (50,907 | ) | (51,458 | ) | (29,319 | ) | (71,527 | ) | ||||||||||
|
Other
income:
|
||||||||||||||||||||
|
Interest
income
|
1,481 | 6,235 | 7,770 | 7,701 | 2,299 | |||||||||||||||
|
Gain
on sale of marketable securities
|
237 | - | - | - | - | |||||||||||||||
|
Total
other income
|
1,718 | 6,235 | 7,770 | 7,701 | 2,299 | |||||||||||||||
|
Net
loss before income taxes
|
(30,612 | ) | (44,672 | ) | (43,688 | ) | (21,618 | ) | (69,228 | ) | ||||||||||
| Income taxes | - | - | - | - | (201 | ) | ||||||||||||||
|
Net
loss
|
$ | (30,612 | ) | $ | (44,672 | ) | $ | (43,688 | ) | $ | (21,618 | ) | $ | (69,429 | ) | |||||
|
Per
share amounts on net loss:
|
||||||||||||||||||||
|
Basic
and diluted
|
$ | (0.98 | ) | $ | (1.48 | ) | $ | (1.59 | ) | $ | (0.84 | ) | $ | (3.32 | ) | |||||
|
December
31,
|
||||||||||||||||||||
|
2009
|
2008
|
2007
|
2006
|
2005
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Balance
Sheet Data:
|
||||||||||||||||||||
|
Cash,
cash equivalents and
marketable
securities
|
$ | 96,196 | $ | 141,374 | $ | 170,370 | $ | 149,100 | $ | 173,090 | ||||||||||
|
Working
capital
|
95,388 | 85,983 | 102,979 | 91,827 | 137,101 | |||||||||||||||
|
Total
assets
|
113,613 | 157,833 | 189,539 | 165,911 | 184,003 | |||||||||||||||
|
Deferred
revenue, long-term
|
- | - | 9,131 | 16,101 | - | |||||||||||||||
|
Other
liabilities, long-term
|
- | 266 | 359 | 123 | 49 | |||||||||||||||
|
Total
stockholders’ equity
|
107,607 | 119,369 | 147,499 | 110,846 | 112,732 | |||||||||||||||
Item
7. Management’s Discussion and Analysis of Financial Condition
and Results of Operations (MD&A)
Overview
General. We are a biopharmaceutical company focusing on the development
and commercialization of innovative therapeutic products to treat the unmet
medical needs of patients with debilitating conditions and life-threatening
diseases. Our principal programs are directed toward supportive care, oncology
and virology. We commenced principal operations in 1988, became publicly traded
in 1997 and throughout have been engaged primarily in research and development
efforts, developing manufacturing capabilities, establishing corporate
collaborations and raising capital.
In June 2008, we began recognizing royalty income from net sales by Wyeth of
subcutaneous RELISTOR. With the reacquisition of our rights to RELISTOR in
October 2009, we will be required, to the extent such tasks are not undertaken
by one or more partners, to address new technological, clinical and commercial
challenges, including, if we choose to sell and support or co-promote RELISTOR,
hiring a sales force, developing a commercial regulatory compliance program and
otherwise building a commercial infrastructure. We continue to monitor our
program expenditures, including headcount levels, in conjunction with program
and program candidates that we choose or are obligated to undertake. We expect
to continue to incur operating losses during the near term.
Our sources of revenues for the three years ended December 31, 2009 have been
payments under our collaboration agreements and funds from research grants and
contracts from the NIH related to our oncology and virology programs. From
January 2006 to October 2009, we recognized revenues from Wyeth for (i)
reimbursement of our development expenses for RELISTOR as incurred, (ii) the
$60.0 million upfront payment we received from Wyeth over the period of our
development obligations, (iii) milestones achieved during our
collaboration with Wyeth and (iv) royalties earned based on net sales of
RELISTOR. To date, our product sales have consisted solely of limited revenues
from the sale of research reagents and we expect that future sales will not
significantly increase over current levels.
A majority of our expenditures to date have been for research and development
activities. During 2009, expenses for our RELISTOR and HIV research program
decreased to $7.8 million and $11.8 million, respectively, compared to (i) $25.4
million and $39.4 million, respectively in 2008 and (ii) $41.5 million and $29.0
million, respectively in 2007. Expenses for our cancer research programs
increased to $20.1 million during 2009 compared to (i) $10.8 million in 2008 and
(ii) $16.1 million in 2007. We expect our expenses for RELISTOR will increase in
2010 and thereafter compared to 2009, as a result of our reacquisition of our
rights to RELISTOR. We also expect that our non-RELISTOR development expenses
will be affected by our RELISTOR expenditures. A portion of our non-RELISTOR
expenses are reimbursed through government funding.
At December 31, 2009, we had cash, cash equivalents and marketable securities
totaling $96.2 million. We expect that cash, cash equivalents and marketable
securities on hand at December 31, 2009 will be sufficient to fund operations at
current levels beyond one year. Cash used in operating
activities for the year ended December 31, 2009 was $49.8 million and our net
loss for 2009 was $30.6 million. We have had recurring losses since inception.
Other than potential revenues from the RELISTOR franchise, we do not anticipate
generating significant recurring revenues, from royalties, product sales or
otherwise, in the near term. Consequently, we may require significant additional
external funding to continue our operations at their current levels in the
future. Such funding may be derived from additional collaboration or licensing
agreements with pharmaceutical or other companies or from the sale of our common
stock or other securities to investors or government funding, but may also not
be available to us on acceptable terms or at all.
Supportive Care. Our first commercial product is RELISTOR®
(methylnaltrexone bromide) subcutaneous injection, a first-in-class
therapy for opioid-induced constipation approved for sale in over 40 countries
worldwide, including the U.S., E.U., Canada, Australia and Brazil. Marketing
applications are pending elsewhere throughout the world.
In October 2009, we and Wyeth Pharmaceuticals terminated our 2005 RELISTOR
collaboration, as a result of which we regained all worldwide rights to
RELISTOR. Under our Transition Agreement, Wyeth is continuing to market and sell
RELISTOR for a U.S. Sales Period ending September 30, 2010 and an ex-U.S. Sales
Period ending December 31, 2010. After this transition period, we will assume
full control of and responsibility for future development and commercialization
of RELISTOR. Shortly after the termination, Pfizer Inc. completed its
previously-announced acquisition of Wyeth, which is now a wholly owned
subsidiary of Pfizer.
We are pursuing a range of strategic alternatives for RELISTOR, including
licensing, collaboration and/or strategic alliances with world-wide or regional
partners, U.S. commercialization of the currently-approved product on our own or
with pharmaceutical detailing and sales organizations and/or co-promotion of the
franchise with a partner using our own sales force.
Under the Transition Agreement, Wyeth has agreed to pay us the sum of $10.0
million in six quarterly installments and is continuing certain ongoing
development efforts for subcutaneous RELISTOR, at its expense, through September
30, 2010. Wyeth’s international sales and marketing obligations during the
ex-U.S. Sales Period are subject to certain extension and early transition
options available to us. Wyeth will continue to pay royalties on worldwide sales
as provided in the 2005 collaboration agreement except that no royalties will be
payable in respect of ex-U.S. sales during (i) the fourth quarter of 2010 to the
extent certain financial targets are not met or (ii) an extended ex-U.S. Sales
Period in the subject country. We have agreed to purchase Wyeth’s remaining
inventory of subcutaneous RELISTOR at the end of the Sales Periods on
agreed-upon terms and conditions. Principal responsibility for regulatory
submissions and interactions for all other formulations and presentations of
RELISTOR will be transferred during and as part of the transition. Wyeth is also
providing financial resources, aggregating up to approximately $14.5 million,
and/or other assistance with respect to agreed-upon regulatory, manufacturing
and supply matters.
Our October 2008 out-license to Ono Pharmaceutical of the rights to subcutaneous
RELISTOR in Japan is unaffected by termination of the Wyeth collaboration. In
June 2009, Ono began clinical testing in Japan of RELISTOR subcutaneous
injection. In August 2009, we and Wyeth announced submission to U.S. and E.U.
regulators of applications for subcutaneous RELISTOR in pre-filled
syringes, which are designed to ease preparation and administration for patients
and caregivers.
We are also developing subcutaneous RELISTOR for treatment of OIC outside the
advanced-illness setting, in individuals with chronic pain not related to
cancer. We and Wyeth recently completed enrollment in a one-year, open-label
safety study, results from which, together with results from a previous phase 3
efficacy trial will support planned regulatory filings in early 2011 in the
U.S., Europe and elsewhere for approval of RELISTOR to treat OIC in the
chronic-pain setting.
As part of our reacquisition of RELISTOR, we have taken over development
responsibilities for an oral formulation of RELISTOR for the treatment of OIC in
patients with chronic, non-cancer pain. In March 2010, we announced that we
plan to advance oral methylnaltrexone for the treatment of OIC into late stage
clinical development and will commence a phase 2b/3 clinical trial of a
methylnaltrexone tablet in chronic-pain patients in the second half of
2010.
Our 2005 collaboration agreement with Wyeth was terminated by the October 2009
Transition Agreement, but the 2005 agreement remained in effect for the time
periods prior to termination covered by this report. Prior to the Transition
Agreement, we received upfront, milestone and royalty payments from Wyeth, and
were reimbursed for expenses we incurred in connection with the development of
RELISTOR; manufacturing and commercialization expenses for RELISTOR were funded
by Wyeth. At inception of the Wyeth collaboration, Wyeth paid to us a $60.0
million non-refundable upfront payment. Wyeth made $39.0 million in milestone
payments thereafter. Costs for the development of RELISTOR incurred by Wyeth or
us starting January 1, 2006 through termination of the 2005 collaboration
agreement were paid by Wyeth. We were reimbursed by Wyeth for our development
costs based on the number of our full-time equivalent employees (FTEs) devoted
to the development project, all subject to Wyeth’s audit rights and possible
reconciliation. During the applicable royalty periods, Wyeth was obligated to
pay us royalties on net sales, as defined (which included specified sales
deductions), of RELISTOR by Wyeth throughout the world other than Japan, where
we licensed rights to subcutaneous RELISTOR to Ono. Wyeth’s royalty obligations
continue during the transition period as provided in the Transition
Agreement.
Under our License Agreement with Ono, in October 2008 we out-licensed rights to
subcutaneous RELISTOR in Japan in return for an upfront payment of $15.0 million
and the right to receive potential milestones, upon achievement of development
responsibilities by Ono, of up to $20.0 million, commercial milestones and
royalties on sales by Ono of subcutaneous RELISTOR in Japan. Ono also has the
option to acquire from us the rights to develop and commercialize in Japan other
formulations of RELISTOR on terms to be negotiated separately. Ono may request
us to perform activities related to its development and commercialization
responsibilities beyond our participation in joint committees and specified
technology transfer related tasks which will be at its expense, and reimbursable
at the time we perform these services.
Future royalty and milestone payments will depend on success in development and
commercialization of RELISTOR, which will be dependent on many factors, such as
the actions of Wyeth during the transition, Ono’s efforts and those of any other
business partner(s) with which we may collaborate, decisions by the FDA and
other regulatory bodies, the outcome of clinical and other testing of RELISTOR,
and our own efforts. Many of these matters are outside our control. In
particular, we cannot guarantee that we will be able to successfully partner the
RELISTOR franchise. We also cannot guarantee, in light of Wyeth’s limited
obligations under the Transition Agreement, its acquisition by Pfizer and its
limited ongoing commercial interest in the RELISTOR franchise, that Wyeth’s
efforts during the transition will achieve any particular level of success in
marketing and sales, regulatory approval or clinical development of subcutaneous
RELISTOR.
Oncology. In the area of prostate cancer, we are conducting a phase 1
clinical trial of a fully human monoclonal ADC directed against PSMA, a protein
found at high levels on the surface of prostate cancer cells and also on the
neovasculature of a number of other types of solid tumors. We are also
developing therapeutic vaccines designed to stimulate an immune response to
PSMA. Our PSMA programs are conducted through our wholly owned subsidiary, PSMA
Development Company LLC.
We recently presented data from preclinical studies of novel multiplex
phosphoinositide 3-kinase (PI3K) inhibitors -- synthetic, small-molecule
compounds identified by us that, in laboratory studies, blocked both PI3K, a key
regulator of one molecular signaling pathway, and MNK, an oncogenic kinase in
the Ras pathway. We believe simultaneously blocking these interlinked cellular
pathways may provide a strategy to combat some of the most aggressive forms of
cancer.
Virology. In the area of virology, we are developing a viral-entry
inhibitor -- a humanized monoclonal antibody, PRO 140 -- for HIV, the virus that
causes AIDS. We are developing the subcutaneous form of PRO 140 for treatment of
HIV infection, which has the potential for weekly self-administration.
Advancement of this program is subject to our obtaining pivotal clinical-trial
funding, for which we have applied to government agencies. In our hepatitis C
virus infection efforts, we are evaluating second-generation HCV-entry
inhibitors as possible development candidates. We are also engaged in research
regarding a prophylactic vaccine against HIV infection.
Our oncology and virology product candidates are not as advanced in development
as RELISTOR, and we do not expect any recurring revenues from sales or otherwise
with respect to these product candidates in the near term.
Results
of Operations (amounts in thousands)
Revenues:
Our sources of revenue during the years ended December 31, 2009, 2008 and 2007,
included our 2005 collaboration with Wyeth, which was effective from January
2006 to October 2009, our License Agreement with Ono, our research grants and
contract from the NIH and, to a small extent, our sale of research reagents. In
June 2008, we began recognizing royalty income from net sales by Wyeth of
subcutaneous RELISTOR.
|
Sources
of Revenue
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
Change
|
||||||||||
|
Research
and development
|
$44,351
|
$59,885
|
$65,455
|
(26%)
|
(9%)
|
|||||
|
Royalty
income
|
2,372
|
146
|
-
|
1525%
|
N/A
|
|||||
|
Research
grants and contract
|
1,968
|
7,460
|
10,075
|
(74%)
|
(26%)
|
|||||
|
Other
revenues
|
256
|
180
|
116
|
42%
|
55%
|
|||||
|
$48,947
|
$67,671
|
$75,646
|
(28%)
|
(11%)
|
||||||
2009 vs. 2008
Research and development revenue:
Wyeth Collaboration. For the three years ended December 31, 2009, we
recognized as revenue: (i) in May 2007, $9,000, representing two milestone
payments, related to the acceptance for review of applications submitted for
marketing approval of a subcutaneous formulation of RELISTOR in the U.S. and
European Union, (ii) in April 2008, $15,000 milestone payment related to the FDA
approval of subcutaneous RELISTOR and (iii) in July 2008, $10,000 milestone
payment related to the European approval of subcutaneous formulation of
RELISTOR. We have analyzed the facts and circumstances of these milestones and
believe that they met those criteria for revenue recognition upon achievement of
the respective milestones. See Critical Accounting Policies –
Revenue Recognition.
During the years ended December 31, 2009 and 2008, we recognized $29,298 and
$59,885, respectively, of revenue from Wyeth, consisting of (i) $14,562 and
$10,228, respectively, of the $60,000 upfront payment we received upon entering
into our 2005 collaboration, (ii) $4,736 and $24,657, respectively, as
reimbursement of our development expenses, and (iii) $10,000 and $25,000,
respectively, under the Transition Agreement and non-refundable milestone
payments.
From the inception of the Wyeth Collaboration through December 31, 2009, we
recognized revenue for the entire $60,000 upfront payment, $104,054 as
reimbursement for our development costs, and a total of $39,000 for
non-refundable milestone payments. We do not expect to receive additional
reimbursement revenue under the 2005 collaboration due to its termination.
Wyeth, at its expense, is continuing certain ongoing development efforts for
subcutaneous RELISTOR through September 30, 2010.
We recognize a portion of the upfront payment in a reporting period in
accordance with the proportionate performance method, which is based on the
percentage of actual effort performed on our development obligations in that
period relative to total effort expected for all of our performance obligations
under the arrangement, as reflected in the most recent development plan and
budget approved by Wyeth and us. During the third quarter of 2007, a revised
budget was approved, which extended our performance period to the end of 2009
and, thereby, decreased the amount of revenue we are recognizing in each
reporting period. The Transition Agreement shortened the obligation period from
the end of 2009 to October 2009 and we recognized the remaining $5.2 million of
unamortized upfront payment as revenue during the fourth quarter of
2009.
Ono License Agreement. In October 2008, we
entered into a License Agreement with Ono and in November 2008, received
an upfront payment of $15,000. We are entitled to receive potential milestones
and royalty payments. During the year ended December 31, 2009, we recognized the
$15,000 of the upfront payment as revenue, due to satisfying our performance
obligations.
Royalty income.
We began earning royalties from net sales by Wyeth of subcutaneous
RELISTOR in June 2008. During the years ended
December 31, 2009 and 2008, we earned royalties of $1,853 and $665,
respectively, based on the net sales of RELISTOR and we recognized $2,372
and $146, respectively, of royalty income. The remaining deferred royalty
revenue balance of $807, as of September 30, 2009, was recognized as royalty
income during the fourth quarter of 2009, the period in which our development
obligations under the 2005 collaboration agreement terminated. Our royalties from net
sales by Wyeth of RELISTOR, as defined, are based on royalty rates under our
2005 collaboration. These rates can range up to 30% of U.S. and 25% of foreign
net sales at the highest sales levels.
Global net sales of RELISTOR were $12.3 million for the year ended December 31,
2009, comprised of $7.1 million of U.S. net sales and $5.2 million of ex-U.S.
net sales. Global net sales of RELISTOR were $4.4 million for the year ended
December 31, 2008, with U.S. and ex-U.S. net sales constituting $2.8 million and
$1.6 million, respectively.
Research grants and contract. In 2003, we were awarded a contract (NIH
Contract) by the NIH to develop a prophylactic vaccine (ProVax) designed to
prevent HIV from becoming established in uninfected individuals exposed to the
virus. Funding under the NIH Contract provided for pre-clinical research,
development and early clinical testing. These funds were used principally in
connection with our ProVax HIV vaccine program. The NIH Contract expired in
December 2008 and through that date, we recognized revenue of $15,509, including
$180 for the achievement of two milestones, and in June 2009, were awarded a
five-year NIH grant totaling up to $14.5 million to continue this work, subject
to annual funding approvals and customary compliance obligations.
Revenues from research grants and contract from the NIH decreased to $1,968 for
the year ended December 31, 2009 from $7,460 for the year ended December 31,
2008; $1,968 and $5,251 from grants and $0 and $2,209 from the NIH Contract for
the years ended December 31, 2009 and 2008, respectively. The decrease in grant
and contract revenue resulted from fewer active grants and reimbursable expenses
in 2009 than in 2008, and the expiration of the NIH Contract in December
2008.
Other revenues, primarily from orders for research reagents, increased to
$256 for the year ended December 31, 2009 from $180 for the year ended December
31, 2008.
2008 vs. 2007
Research revenues
from collaborator. During the years ended December 31, 2008 and 2007, we
recognized $59,885 and $65,455, respectively, of revenue from Wyeth, consisting
of (i) $10,228 and $16,378, respectively, of the $60,000 upfront payment we
received upon entering into our Collaboration in December 2005, (ii) $24,657 and
$40,077, respectively, as reimbursement of our development expenses, and (iii)
$25,000 and $9,000, respectively, of non-refundable milestone payments.
Ono License
Agreement. As of December 31, 2008, relative to the $15.0 million upfront
payment from Ono, we recorded $15.0 million as deferred revenue – current, which
was recognized as revenue during the first quarter of 2009, due to satisfying
our performance obligations.
Royalty income.
During the
year ended December 31, 2008, we earned royalties of $665, based on the net
sales of RELISTOR and we recognized $146, of royalty income. As of
December 31, 2008, we have recorded a cumulative total of $519 as deferred
revenue – current. The $519 of deferred royalty revenue was to be recognized as
royalty income over the period of our development obligations relating to
RELISTOR, which was shortened by the Transition Agreement from the end of 2009
to October 2009.
Global net sales of RELISTOR, which began in June 2008, were $4.4 million in the
year ended December 31, 2008. U.S. RELISTOR net sales totaled $2.8 million in
2008 and ex-U.S. RELISTOR net sales totaled $1.6 million in 2008.
Research grants and contract. Revenues from research grants and contract
from the NIH decreased to $7,460 for the year ended December 31, 2008 from
$10,075 for the year ended December 31, 2007; $5,251 and $6,185 from grants and
$2,209 and $3,890 from the NIH Contract for the years ended December 31, 2008
and 2007, respectively. The decrease in grant revenue resulted from fewer
reimbursable expenses in 2008 than in 2007 on new and continuing grant related
projects, and decreased activity under the NIH Contract.
Other revenues, primarily from higher orders for research reagents,
increased to $180 for the year ended December 31, 2008 from $116 for the year
ended December 31, 2007.
Expenses:
Research and
Development Expenses include scientific labor, supplies, facility costs,
clinical trial costs, product manufacturing costs, royalty payments and license
fees. Research and development expenses, including license fees and royalty
expense, decreased to $51,093 for the year ended December 31, 2009 from $85,135
for the year ended December 31, 2008, and from $96,176 in the year ended
December 31, 2007. During 2009, the decrease in research and development
expenses over those in 2008 and 2007 was primarily due to reduced RELISTOR
development expenses following completion of clinical trials and reduced
manufacturing activities for PRO 140 clinical trials, partially offset by
increases in PSMA-related activities. See Liquidity and Capital Resources –
Uses of Cash, for details of the changes in these expenses by project.
From 2006 through October 2009, Wyeth reimbursed us for development expenses we
incurred related to RELISTOR under the development plan agreed to between Wyeth
and us. Portions of our expenses related to our HIV, HCV and PSMA programs are
funded through grants and a contract from the NIH (see Revenues- Research Grants and
Contract). The changes in research and development expense, by category
of expense, are as follows:
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Salaries
and benefits (cash)
|
$21,576
|
$24,383
|
$24,061
|
(12%)
|
1%
|
||||
2009 vs.
2008 Salaries and benefits (cash) decreased due to a
decline in average headcount to 175 from 196 for the years ended December 31,
2009 and 2008, respectively, in the research and development, manufacturing and
clinical departments as part of our efforts to manage costs.
2008 vs.
2007 Salaries and benefits (cash) increased due to an
increase in average headcount to 196 from 190 for the years ended December 31,
2008 and 2007, respectively, in the research and development, manufacturing and
clinical departments.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
||||||
|
Percent
change
|
||||||||||
|
Share-based
compensation (non-cash)
|
$7,225
|
$7,241
|
$7,104
|
0%
|
2%
|
|||||
2009 vs.
2008 Share-based compensation (non-cash) decreased for
the year ended December 31, 2009 compared to the year ended December 31, 2008
due to lower employee stock purchase plan expense, partially offset by higher
restricted stock and stock option plan expenses. See Critical Accounting Policies −
Share-Based Payment Arrangements.
2008 vs.
2007 Share-based compensation (non-cash) increased due
to increase in employee stock purchase plan expenses, partially offset by lower
stock options and restricted stock expenses for the year ended December 31, 2008
compared to the year ended December 31, 2007. See Critical Accounting Policies −
Share-Based Payment Arrangements.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Clinical
trial costs
|
$2,198
|
$14,127
|
$19,225
|
(84%)
|
(27%)
|
||||
2009 vs.
2008 Clinical trial costs decreased primarily due to
lower expenses for (i) RELISTOR ($9,768), from reduced clinical trial
activities, and (ii) HIV ($2,821), due to decreased PRO 140 clinical trial
activities and Other ($5), partially offset by an increase in expenses for
Cancer ($665), all for the year ended December 31, 2009 compared to the year
ended December 31, 2008.
2008 vs.
2007 Clinical trial costs decreased primarily due to
lower expenses for (i) RELISTOR ($6,686), from reduced clinical trial
activities, and (ii) Cancer ($1,534), due to termination of a development
program in 2007, partially offset by an increase in HIV ($3,122), due to
increased PRO 140 clinical trial activities, all for the year ended December 31,
2008 compared to the year ended December 31, 2007.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Laboratory
supplies
|
$3,011
|
$3,944
|
$5,196
|
(24%)
|
(24%)
|
||||
2009 vs.
2008 Laboratory supplies decreased due to lower expenses
for HIV ($1,841), resulting from a decline in the purchases of drug supplies,
partially offset by an increase in (i) Cancer ($842), due to higher expenses for
PSMA ADC, (ii) Other projects ($62) and (iii) RELISTOR ($4), all for the year
ended December 31, 2009 compared to the year ended December 31,
2008.
2008 vs. 2007
Laboratory supplies decreased due to lower expenses for (i) HIV ($808),
resulting from a decline in the purchases of drug supplies, (ii) Cancer ($235),
due to fewer expenses for PSMA vaccines and a terminated program, and (iii)
Other projects ($209), all for the year ended December 31, 2008 compared to the
year ended December 31, 2007.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Contract
manufacturing and subcontractors
|
$8,040
|
$21,681
|
$26,051
|
(63%)
|
(17%)
|
||||
2009 vs.
2008 Contract manufacturing and subcontractors decreased
due to lower (i) HIV expenses ($13,514), resulting from a decline in
manufacturing expenses for PRO 140 and (ii) RELISTOR expenses ($1,439),
partially offset by increases in both Cancer ($956), due to higher contract
manufacturing expenses for PSMA ADC, and Other ($356), all for the year ended
December 31, 2009 compared to the year ended December 31, 2008. These expenses
are related to the conduct of clinical trials, including manufacture by third
parties of drug materials, testing, analysis, formulation and toxicology
services, and vary as the timing and level of such services are
required.
2008 vs.
2007 Contract manufacturing and subcontractors decreased
due to lower (i) Cancer expenses ($5,401), due to lower contract manufacturing
expenses for PSMA ADC, and (ii) RELISTOR expenses ($2,301), partially offset by
increases in both HIV ($3,052), due to higher contract manufacturing for PRO 140
and Other ($280), all for the year ended December 31, 2008 compared to the year
ended December 31, 2007. These expenses are related to the conduct of clinical
trials, including manufacture by third parties of drug materials, testing,
analysis, formulation and toxicology services, and vary as the timing and level
of such services are required.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Consultants
|
$1,006
|
$3,514
|
$4,722
|
(71%)
|
(26%)
|
||||
2009
vs. 2008 Consultants expenses
decreased due to lower expenses for (i) RELISTOR ($1,493), (ii) Cancer
($305), (iii) HIV ($525) and (iv) Other projects ($185), all for the year ended
December 31, 2009 compared to the year ended December 31, 2008. These expenses
are related to the monitoring of clinical trials as well as the analysis of data
from completed clinical trials and vary as the timing and level of such services
are required.
2008
vs. 2007 Consultants expenses
decreased due to lower expenses for (i) RELISTOR ($1,579), and (ii) Other
projects ($174), partially offset by increases in HIV ($294) and Cancer ($251),
all for the year ended December 31, 2008 compared to the year ended December 31,
2007. These expenses are related to the monitoring of clinical trials as well as
the analysis of data from completed clinical trials and vary as the timing and
level of such services are required.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
License
fees
|
$1,058
|
$2,830
|
$942
|
(63%)
|
200%
|
||||
2009 vs.
2008 License fees decreased primarily due to a decline
in expenses for HIV ($774), Cancer ($516) and RELISTOR ($482), all for the year
ended December 31, 2009 compared to the year ended December 31,
2008.
2008 vs.
2007 License fees increased primarily due to an increase
in expenses for HIV ($1,100), RELISTOR ($522) and Cancer ($266), all for the
year ended December 31, 2008 compared to the year ended December 31,
2007.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Royalty
expense
|
$237
|
$15
|
$-
|
1480%
|
N/A
|
||||
2009 vs.
2008 We incurred $185 and $67, respectively, of royalty
costs and recognized $237 and $15, respectively, of royalty expenses during the
years ended December 31, 2009 and 2008. The remaining deferred royalty charges
balance of $81, as of September 30, 2009, was recognized as royalty expense
during the fourth quarter 2009, the period in which our development obligations
relating to RELISTOR terminated.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Other
operating expenses
|
$6,742
|
$7,400
|
$8,875
|
(9%)
|
(17%)
|
||||
2009 vs.
2008 Other operating expenses decreased for the year
ended December 31, 2009 compared to the year ended December 31, 2008, primarily
due to a decrease in rent ($503), travel ($195), insurance ($82), facilities
($23) and other operating expenses ($223), partially offset by an increase in
computer expenses ($368).
2008 vs.
2007 Other operating expenses decreased for the year
ended December 31, 2008 compared to the year ended December 31, 2007, primarily
due to a decrease in computer expenses ($1,760), insurance ($294), facilities
($186) and travel ($102), partially offset by an increase in other operating
expenses ($21) and rent ($846).
General and
Administrative Expenses decreased to $25,106 in the year ended December
31, 2009 from $28,834 in the year ended December 31, 2008 and from $27,901 in
the year ended December 31, 2007, as follows:
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Salaries
and benefits (cash)
|
$8,257
|
$8,610
|
$7,243
|
(4%)
|
19%
|
||||
2009
vs. 2008 Salaries and benefits (cash) decreased due to
lower bonus expense for the year ended December 31, 2009 compared to the same
period in 2008, and a decrease in average headcount to 49 from 52, in the
general and administrative departments as part of our efforts to manage
costs.
2008
vs. 2007 Salaries and benefits (cash) increased due to
compensation increases and an increase in average headcount to 52 from 43 in the
general and administrative departments for the years ended December 31, 2008 and
2007, respectively.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Share-based
compensation (non-cash)
|
$5,761
|
$6,892
|
$8,202
|
(16%)
|
(16%)
|
||||
2009 vs.
2008 Share-based compensation (non-cash) decreased due
to decrease in stock option and employee stock purchase plans expenses,
partially offset by an increase in restricted stock expenses for the year ended
December 31, 2009 compared to the year ended December 31, 2008. See Critical Accounting Policies
−Share-Based Payment Arrangements.
2008 vs.
2007 Share-based compensation (non-cash) decreased due
to decrease in stock options and restricted stock expenses, partially offset by
an increase in employee stock purchase plans expenses for the year ended
December 31, 2008 compared to the year ended December 31, 2007. See Critical Accounting Policies
−Share-Based Payment Arrangements.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Consulting
and professional fees
|
$6,696
|
$7,915
|
$6,552
|
(15%)
|
21%
|
||||
2009
vs. 2008 Consulting and professional fees decreased due
to a decrease in consultant fees ($790), patent fees ($540) and public relations
($156), which were partially offset by an increase in audit and compliance fees
($185), legal fees ($64) and other ($18), all for the year ended December 31,
2009 compared to the year ended December 31, 2008.
2008
vs. 2007 Consulting and professional fees increased due
to increases in consultant fees ($1,135), legal and patent fees ($132) and other
miscellaneous costs ($164), which were partially offset by a decrease in audit
and tax fees ($68), all for the year ended December 31, 2008 compared to the
year ended December 31, 2007.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Other
operating expenses
|
$4,392
|
$5,417
|
$5,904
|
(19%)
|
(8%)
|
||||
2009
vs. 2008 Other operating expenses decreased due to lower
spending on recruiting ($234), computer software ($280), travel ($140), taxes
($12), rent ($167), conferences and seminars ($54) and other operating expenses
($195), partially offset by an increase in investor relations ($57), all for the
year ended December 31, 2009 compared to the year ended December 31, 2008.
2008
vs. 2007 Other operating expenses decreased due to lower
spending on recruiting ($452), facilities ($142), investor relations ($91),
taxes ($27) and other operating expenses ($44), partially offset by increases in
rent ($269), all for the year ended December 31, 2008 compared to the year ended
December 31, 2007.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Depreciation
and amortization
|
$5,078
|
$4,609
|
$3,027
|
10%
|
52%
|
||||
2009
vs. 2008 Depreciation and amortization expense increased
to $5,078 for the year ended December 31, 2009 from $4,609 for the year ended
December 31, 2008, due to fixed asset purchases in 2008 and leasehold
improvements placed in service during 2007.
2008
vs. 2007 Depreciation and amortization expense increased
to $4,609 for the year ended December 31, 2008 from $3,027 for the year ended
December 31, 2007, due to increased amortization of leasehold improvements.
Approximately $3.8 million of leasehold improvements was placed in service
during 2007, which is being amortized through the end of the lease term of
December 31, 2009.
|
2009
|
2008
|
2007
|
2009
vs. 2008
|
2008
vs. 2007
|
|||||
|
Percent
change
|
|||||||||
|
Other
income
|
$1,718
|
$6,235
|
$7,770
|
(72%)
|
(20%)
|
||||
2009
vs. 2008 Interest income decreased to $1,481 for the
year ended December 31, 2009 from $6,235 for the year ended December 31, 2008.
Interest income, as reported, is primarily the result of investment income from
our marketable securities, decreased by the amortization of premiums we paid or
increased by the amortization of discounts we received for those marketable
securities. For the years ended December 31, 2009 and 2008, investment income
decreased to $2,075 from $7,195, respectively, due to a decrease in interest
rates, lower average balances of cash equivalents and corporate debt securities
and higher average balances of money market funds in 2009 than in 2008.
Amortization of premiums, net of discounts, was ($594) and ($960) for years
ended December 31, 2009 and 2008, respectively. In addition, other income for
the year ended December 31, 2009 includes $237 of gains from sale of marketable
securities.
2008
vs. 2007 Interest income decreased to $6,235 for the
year ended December 31, 2008 from $7,770 for the year ended December 31, 2007.
Interest income, as reported, is primarily the result of investment income from
our marketable securities, decreased by the amortization of premiums we paid or
increased by the amortization of discounts we received for those marketable
securities. For the years ended December 31, 2008 and 2007, investment income
decreased to $7,195 from $7,325, respectively, due to a decrease in interest
rates and lower average balance of cash equivalents and marketable securities in
2008 than in 2007. Amortization of premiums, net of discounts, was ($960) and
$445 for the years ended December 31, 2008 and 2007, respectively.
Income
Taxes:
For the years ended December 31, 2009, 2008 and 2007, we had losses both for
book and tax purposes.
Net
Loss:
Our net loss was $30,612 for the year ended December 31, 2009, $44,672 for the
year ended December 31, 2008 and $43,688 for the year ended December 31,
2007.
Liquidity
and Capital Resources
We have to date generated only modest amounts of product and royalty revenue,
and consequently have relied principally on external funding and our 2005
collaboration to finance our operations. We have funded our operations since
inception primarily through private placements of equity securities, public
offerings of common stock, payments received under collaboration agreements,
funding under government research grants and contracts, interest on investments,
proceeds from the exercise of outstanding options and warrants, and the sale of
our common stock under our two employee stock purchase plans (Purchase Plans).
Advancement of the PRO 140 program is subject to our obtaining
pivotal-clinical-trial funding, for which we have applied to government
agencies. We are also pursuing strategic collaborations with biopharmaceutical
companies for PSMA ADC.
With the reacquisition of our rights to RELISTOR, we will be required, to the
extent such tasks are not undertaken by one or more partners, to address new
technological, clinical and commercial challenges, including, if we choose to
sell and support or co-promote RELISTOR, hiring a sales force, developing a
commercial regulatory compliance program and otherwise building a commercial
infrastructure. We continue to monitor our program expenditures, including
headcount levels, in conjunction with program and program candidates that we
choose or are obligated to undertake. We expect to continue to incur operating
losses during the near term. We cannot forecast with any degree of certainty,
however, which products or indications, if any, will be subject to future
arrangements, or how they would affect our capital requirements. The
consummation of other agreements would further allow us to advance other
projects with current funds. We have agreed to purchase Wyeth’s remaining
inventory of subcutaneous RELISTOR at the end of its sales periods on
agreed-upon terms and conditions. If we were to undertake development and
commercialization of RELISTOR or any other product candidate on our own without
a partner, however, we would be required to establish manufacturing and
marketing capabilities and fund a sales force, which we currently do not
have.
At December 31, 2009, we had cash, cash equivalents and marketable securities,
including non-current portion, totaling $96.2 million compared with $141.4
million at December 31, 2008. We expect that our existing cash, cash equivalents
and marketable securities at December 31, 2009 are sufficient to fund current
operations beyond one year. Our cash flow from operating activities was negative
for the years ended December 31, 2009, 2008 and 2007 due primarily to the excess
of expenditures on our research and development programs and general and
administrative costs related to those programs over cash received from
collaborators and government grants and contracts to fund such programs, as
described below. See Risk
Factors.
Sources
of Cash
Operating Activities. Our collaboration with Wyeth provided us with a
$60.0 million upfront payment in December 2005. We also received $34.0 million
in milestone payments during the three years ended December 31, 2009. In
addition, from January 2006 to October 2009, Wyeth reimbursed us for development
expenses we incurred related to RELISTOR under the development plan agreed to
between us. For the years ended December 31, 2009, 2008 and 2007, we received
$4.7 million, $24.7 million and $40.1 million, respectively, of such
reimbursement. These reimbursements have ceased as a result of termination of
the 2005 Wyeth collaboration.
Under the Transition Agreement, Wyeth has agreed pay us the sum of $10.0 million
in six quarterly installments, and its sales and marketing obligations during
the transition will continue in the United States through a U.S. Sales Period
and worldwide excluding Japan through the ex-U.S. Sales Period, in accordance
with an agreed-upon commercialization plan. The ex-U.S. Sales Period is subject
to certain extension and early termination options available to us. Wyeth will
continue to pay royalties as provided in the 2005 collaboration agreement except
that no royalties will be payable in respect of ex-U.S. sales during (i) the
fourth quarter of 2010 to the extent certain financial targets for that quarter
are not met or (ii) an extended international sale period in the subject
country. Wyeth is also continuing certain ongoing development efforts for
RELISTOR, at its expense, through September 30, 2010, and is providing financial
resources and/or other assistance, aggregating up to approximately $14.5
million, with respect to agreed-upon regulatory, manufacturing and supply
matters.
Under our License Agreement with Ono, we received from Ono, in November 2008, an
upfront payment of $15.0 million, which was recognized as revenue during the
first quarter of 2009, upon satisfaction of our performance obligations, and are
entitled to receive potential milestone payments, upon achievement of
development milestones by Ono, of up to $20.0 million, commercial milestones and
royalties on sales of subcutaneous RELISTOR in Japan. Ono is also responsible
for development and commercialization costs for subcutaneous RELISTOR in
Japan.
We have funded our ProVax HIV vaccine program through contracts with the NIH
providing for pre-clinical research, development and early clinical testing
support. Through December 2008, we recognized revenue of $15.5 million from the
NIH, including $0.2 million for the achievement of two milestones, and in June
2009 were awarded a new five-year NIH grant totaling up to $14.5 million to
continue this work, subject to annual funding approvals and customary compliance
obligations.
A substantial portion of our revenues to date has been derived from federal
government grants. During the years ended December 31, 2009, 2008 and 2007, we
recognized as revenue awards made to us by the NIH between 2004 and 2009, to
partially fund some of our programs. For the years ended December 31, 2009, 2008
and 2007, we recognized $2.0 million, $5.3 million and $6.2 million,
respectively, of revenue from all of our NIH grants.
Changes in Accounts receivable and Accounts payable for the years ended December
31, 2009, 2008 and 2007 resulted from the timing of receipts from the NIH and
Wyeth, and payments made to trade vendors in the normal course of
business.
Other than amounts to be received from Wyeth, Ono and from currently approved
grants, we have no committed external sources of capital. Other than revenues
from RELISTOR, we expect no significant product revenues for a number of years,
as it will take at least that much time, if ever, to bring our product
candidates to the commercial marketing stage.
Investing Activities. We purchase and sell marketable securities in order
to provide funding for operations. Our marketable securities, which include
corporate debt securities, securities of government-sponsored entities and
auction rate securities, are classified as available-for-sale.
A substantial portion of our cash and cash equivalents ($90.9 million) are
guaranteed by the U.S. Treasury or Federal Deposit Insurance Corporation’s
guarantee program. Our marketable securities ($5.3 million), which include
corporate debt securities and auction rate securities, are classified as
available for sale and include $2.9 million of securities collateralized by
student loan obligations subsidized by the U.S. government. These investments,
while rated investment grade by the Standard & Poor’s and Moody’s rating
agencies and predominantly having scheduled maturities greater than ten years,
are heavily concentrated in the U.S. financial sector, which continues to be
under substantial stress.
As a result of changes in general market conditions during 2008, we determined
to reduce the principal amount of auction rate securities in our portfolio as
they came up for auction and invest the proceeds in other securities in
accordance with our investment guidelines. Beginning in the first quarter of
2008, auctions failed for certain of our auction rate securities because sell
orders exceeded buy orders. As a result, at December 31, 2009, we continue to
hold approximately $3.8 million of auction rate securities which, in the event
of auction failure, are reset according to the contractual terms in the
governing instruments. To date, we have received all scheduled interest payments
on these securities. We will not realize cash in respect of the principal amount
of these securities until a successful auction occurs, the issuer calls or
restructures the underlying security, the underlying security matures and is
paid, or a buyer outside the auction process emerges.
We monitor markets for our investments, but cannot guarantee that additional
losses will not be required to be recorded. Valuation of securities is subject
to uncertainties that are difficult to predict, such as changes to credit
ratings of the securities and/or the underlying assets supporting them, default
rates applicable to the underlying assets, underlying collateral value, discount
rates, counterparty risk, ongoing strength and quality of market credit and
liquidity and general economic and market conditions. We do not believe the
carrying values of our investments are other than temporarily impaired and
therefore expect the positions will eventually be liquidated without significant
loss.
Our marketable securities are purchased and, in the case of auction rate
securities, sold by third-party brokers in accordance with our investment policy
guidelines. Our brokerage account requires that all marketable securities be
held to maturity unless authorization is obtained from us to sell earlier. In
fact, we had a history of holding all marketable securities to maturity prior to
the second quarter of 2009, when we decided to sell a portion of our marketable
securities which had scheduled maturities between the fourth quarter of 2009 and
the third quarter of 2010. The proceeds from these sales were $24.8 million,
resulting in a gain of $0.2 million.
We expect to recover the amortized cost of all of our investments at maturity.
Because we do not anticipate having to sell these securities in order to operate
our business and believe it is not more likely than not that we will be required
to sell these securities before recovery of principal, we do not consider these
marketable securities to be other than temporarily impaired at December 31,
2009.
Financing Activities. During the years ended December 31, 2009, 2008 and
2007, we received cash of $4.9 million, $6.5 million and $7.8 million,
respectively, from the exercise of stock options by employees, directors and
non-employee consultants, from the sale of our common stock under our Purchase
Plans and from sale of common stock in public offering in 2007. The amount of
cash we receive from these sources fluctuates commensurate with headcount levels
and changes in the price of our common stock on the grant date for options
exercised, and on the sale date for shares sold under the Purchase
Plans.
In 2007, we completed a public offering of 2.6 million shares of our common
stock, pursuant to a shelf registration statement that had been filed with the
SEC in 2006, which had registered 4.0 million shares of our common stock; that
registration statement has now expired. We received proceeds of $57.3 million,
or $22.04 per share, which was net of underwriting discounts and commissions of
approximately $2.9 million, and paid approximately $0.2 million in other
offering expenses.
In 2008, we obtained approvals for RELISTOR from the FDA, as well as European
Union, Canadian, Australian, Venezuelan and other regulatory authorities. Under
the Transition Agreement, Wyeth, at its expense, is continuing certain ongoing
development efforts for subcutaneous RELISTOR through September 30, 2010, and
its sales and marketing obligations during the transition will continue in the
United States through a U.S. Sales Period and worldwide excluding Japan through
the ex-U.S. Sales Period, in accordance with an agreed-upon commercialization
plan. The ex-U.S. Sales Period is subject to certain extension and early
termination options available to us. Wyeth will continue to pay royalties as
provided in the 2005 collaboration agreement except that no royalties will be
payable in respect of ex-U.S. sales during (i) the fourth quarter of 2010 to the
extent certain financial targets for that quarter are not met or (ii) an
extended international sale period in the subject country.
Unless we obtain regulatory approval from the FDA for additional product
candidates and/or enter into agreements with corporate collaborators with
respect to the development of RELISTOR and our additional technologies, we will
be required to fund our operations in the future through offerings of equity or
debt securities, royalty or other financing agreements, U.S. commercialization
or co-promotion with our own sales force, and/or grants and government
contracts. Adequate additional funding may not be available to us on acceptable
terms or at all. Our inability to raise additional capital on terms reasonably
acceptable to us may seriously jeopardize the future success of our
business.
Uses
of Cash
Operating Activities. The majority of our cash has been used to advance
our research and development programs. We currently have major research and
development programs in supportive care, oncology and virology, and are
conducting several smaller research projects in those latter areas. Our total
expenses for research and development from inception through December 31, 2009
have been approximately $529.6 million. For various reasons, including the early
stage of certain of our programs, the timing and results of our clinical trials,
our dependence in certain instances on third parties and the uncertainty of the
specific nature of RELISTOR-related future arrangements and relationships
following termination of the Wyeth collaboration, many of which are outside of
our control, we cannot estimate the total remaining costs to be incurred and
timing to complete all our research and development programs.
For the years ended December 31, 2009, 2008 and 2007, research and development
costs incurred, by project, were as follows:
|
For
the Year Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
(in
millions)
|
||||||||||||
|
RELISTOR
|
$ | 7.8 | $ | 25.4 | $ | 41.5 | ||||||
|
HIV
|
11.8 | 39.4 | 29.0 | |||||||||
|
Cancer
|
20.1 | 10.8 | 16.1 | |||||||||
|
Other
programs
|
11.4 | 9.5 | 9.6 | |||||||||
|
Total
|
$ | 51.1 | $ | 85.1 | $ | 96.2 | ||||||
We will require additional funding to continue our research and product
development programs, conduct pre-clinical studies and clinical trials, fund
operating expenses, pursue regulatory approvals for our product candidates, file
and prosecute patent applications and enforce or defend patent claims, if any,
and fund product in-licensing and any possible acquisitions.
Investing Activities. During the years ended December 31, 2009, 2008 and
2007, we have spent $0.9 million, $2.2 million and $5.2 million, respectively,
on capital expenditures. These reductions have resulted from our continuing
efforts to manage costs in light of available resources and prospects. These
expenditures have been primarily related to the purchase of laboratory equipment
for our research and development projects, for the manufacture of research and
clinical products.
Contractual
Obligations
Our funding requirements, both for the next 12 months and beyond, will include
required payments under operating leases and licensing and collaboration
agreements. The following table summarizes our contractual obligations as of
December 31, 2009 for future payments under these agreements:
|
Payments
due by December 31,
|
||||||||||||||||||||
|
Total
|
2010
|
2011-2012 | 2013-2014 |
Thereafter
|
||||||||||||||||
|
(in
millions)
|
||||||||||||||||||||
|
Operating
leases
|
$ | 46.0 | $ | 3.7 | $ | 7.7 | $ | 8.0 | $ | 26.6 | ||||||||||
|
License
and collaboration agreements (1)
|
81.9 | 0.8 | 1.8 | 4.3 | 75.0 | |||||||||||||||
|
Total
|
$ | 127.9 | $ | 4.5 | $ | 9.5 | $ | 12.3 | $ | 101.6 | ||||||||||
_______________
|
(1)
|
Assumes
attainment of milestones covered under each agreement, including those to
which PSMA LLC is a party. The timing of the achievement of the related
milestones is highly uncertain, and accordingly the actual timing of
payments, if any, is likely to vary, perhaps significantly, relative to
the timing contemplated by this
table.
|
We periodically assess the scientific progress and merits of each of our
programs to determine if continued research and development is commercially and
economically viable. Certain of our programs have been terminated due to the
lack of scientific progress and prospects for ultimate commercialization.
Because of the uncertainties associated with research and development in these
programs, the duration and completion costs of our research and development
projects are difficult to estimate and are subject to considerable variation.
Our inability to complete research and development projects in a timely manner
or failure to enter into collaborative agreements could significantly increase
capital requirements and adversely affect our liquidity.
Our cash requirements may vary materially from those now planned because of
results of research and development and product testing, changes in existing
relationships or new relationships with licensees, licensors or other
collaborators, changes in the focus and direction of our research and
development programs, competitive and technological advances, the cost of
filing, prosecuting, defending and enforcing patent claims, the regulatory
approval process, manufacturing and marketing and other costs associated with
the commercialization of products following receipt of regulatory approvals and
other factors.
The above discussion contains forward-looking statements based on our current
operating plan and the assumptions on which it relies. There could be deviations
from that plan that would consume our assets earlier than planned.
Off-Balance
Sheet Arrangements and Guarantees
We have no obligations under off-balance sheet arrangements and do not guarantee
the obligations of any other unconsolidated entity.
Critical
Accounting Policies
We prepare our financial statements in conformity with accounting principles
generally accepted in the United States of America. Our significant accounting
policies are disclosed in Note 2 to our financial statements included in this
Annual Report on Form 10-K for the year ended December 31, 2009. The selection
and application of these accounting principles and methods requires us to make
estimates and assumptions that affect the reported amounts of assets,
liabilities, revenues and expenses, as well as certain financial statement
disclosures. On an ongoing basis, we evaluate our estimates. We base our
estimates on historical experience and on various other assumptions that are
believed to be reasonable under the circumstances. The results of our evaluation
form the basis for making judgments about the carrying values of assets and
liabilities that are not otherwise readily apparent. While we believe that the
estimates and assumptions we use in preparing the financial statements are
appropriate, these estimates and assumptions are subject to a number of factors
and uncertainties regarding their ultimate outcome and, therefore, actual
results could differ from these estimates.
We have identified our critical accounting policies and estimates below. These
are policies and estimates that we believe are the most important in portraying
our financial condition and results of operations, and that require our most
difficult, subjective or complex judgments, often as a result of the need to
make estimates about the effect of matters that are inherently uncertain. We
have discussed the development, selection and disclosure of these critical
accounting policies and estimates with the Audit Committee of our Board of
Directors.
Revenue Recognition. We recognize revenue from all sources based on the
provisions of the SEC’s Staff Accounting Bulletin (SAB) No. 104 (SAB 104) and
ASC 605 Revenue Recognition.
Collaborations may contain substantive milestone payments to which we apply the
substantive milestone method (Substantive Milestone Method). Substantive
milestone payments are considered to be performance payments that are recognized
upon achievement of the milestone only if all of the following conditions are
met: (i) the milestone payment is non-refundable, (ii) achievement of the
milestone involves a degree of risk and was not reasonably assured at the
inception of the arrangement, (iii) substantive effort is involved in achieving
the milestone, (iv) the amount of the milestone payment is reasonable in
relation to the effort expended or the risk associated with achievement of the
milestone, and (v) a reasonable amount of time passes between the upfront
license payment and the first milestone payment as well as between each
subsequent milestone payment.
Determination as to whether a milestone meets the aforementioned conditions
involves management’s judgment. If any of these conditions are not met, the
resulting payment would not be considered a substantive milestone and,
therefore, the resulting payment would be part of the consideration and be
recognized as revenue as such performance obligations are
performed.
Royalty revenue is recognized based upon net sales of related licensed products.
Royalty revenue is recognized in the period the sales occur, provided that the
royalty amounts are fixed or determinable, collection of the related receivable
is reasonably assured and we have no remaining performance obligations under the
arrangement providing for the royalty. If royalties are received when we have
remaining performance obligations, they would be attributed to the services
being provided under the arrangement and, therefore, recognized as such
obligations are performed under the proportionate performance
method.
We recognize upfront license payments as revenue upon delivery of the license
only if the license had standalone value and the fair value of the undelivered
performance obligations, typically including research or steering or other
committee services, could be determined. If the fair value of the undelivered
performance obligations could be determined, such obligations would then be
accounted for separately as performed. If the license is considered to either
(i) not have standalone value, or (ii) have standalone value but the fair value
of any of the undelivered performance obligations could not be determined, the
upfront license payments would be recognized as revenue over the estimated
period of when our performance obligations are performed.
Under the proportionate performance method we recognize revenue provided that we
can reasonably estimate the level of effort required to complete our performance
obligations under an arrangement and such performance obligations are provided
on a best-efforts basis. Direct labor hours or full-time equivalents will
typically be used as the measure of performance. Revenue is recognized in any
period as the percent of actual effort expended in that period relative to total
effort for all of our performance obligations under the arrangement. Significant
judgment and estimates are required in determining the nature and assignment of
tasks to be accomplished by each of the parties and the level of effort required
for us to complete our performance obligations under the arrangement. The nature
and assignment of tasks to be performed by each party involves the preparation,
discussion and approval by the parties of a development plan and
budget.
Amounts not expected to be recognized within one year of the balance sheet date
are classified as long-term deferred revenue. The estimate of the classification
of deferred revenue as short-term or long-term is based upon management’s
current operating budget with the collaborator for the total effort required to
complete our performance obligations under the arrangement.
As the development programs progress over time, the development budgets,
including the amount of FTEs, may be revised, resulting in a change to the
development period or costs. Changes in the development estimates are likely to
affect the amount of revenue recognized in the period of change and each year in
the future as compared to prior periods. Under the Wyeth collaboration, we
recognized $6.2 million less revenue from the $60.0 million upfront payment
during the year ended December 31, 2008 compared to the amounts recognized in
2007 due to an extension of the development budget from December 31, 2008 to
December 31, 2009. Conversely, we recognized $4.3 million more revenue during
2009 compared to amounts recognized in 2008 due to the increase in the percent
of actual effort expended in 2009 relative to the total remaining effort to
complete development.
Share-Based Payment Arrangements. Our share-based compensation to
employees includes non-qualified stock options, restricted stock and shares
issued under our Purchase Plans, which are compensatory under ASC 718
Compensation – Stock Compensation. We account for share-based compensation to
non-employees, including non-qualified stock options and restricted stock, in
accordance with ASC 505 Equity.
The fair value of each non-qualified stock option award is estimated on the date
of grant using the Black-Scholes option pricing model.
The model requires input assumptions with respect to (i) expected volatility of
our common stock, which is based upon the daily quoted market prices on The
NASDAQ Stock Market LLC over a period equal to the expected term, (ii) the
period of time over which employees, officers, our chief executive officer,
directors and non-employee consultants are expected to hold their options prior
to exercise, (iii) zero expected dividend yield due to never having paid
dividends and not expecting to pay dividends in the future, and (iv) risk-free
interest rates for periods within the expected term of the options, which are
based on the U.S. Treasury yield curve in effect at the time of
grant.
Historical volatilities are based upon daily quoted market prices of our common
stock on The NASDAQ Stock Market LLC over a period equal to the expected term of
the related equity instruments. We rely only on historical volatility since it
provides the most reliable indication of future volatility. Future volatility is
expected to be consistent with historical; historical volatility is calculated
using a simple average calculation; historical data is available for the length
of the option’s expected term and a sufficient number of price observations are
used consistently. Since our stock options are not traded on a public market, we
do not use implied volatility.
The expected term of options granted represents the period of time that options
granted are expected to be outstanding based upon historical data related to
exercise and post-termination cancellation activity. The expected term of stock
options granted to our Chief Executive Officer and non-employee directors and
consultants are calculated separately from stock options granted to employees
and officers.
We apply a forfeiture rate to the number of unvested awards in each reporting
period in order to estimate the number of awards that are expected to vest.
Estimated forfeiture rates are based upon historical data on vesting behavior of
employees. We adjust the total amount of compensation cost recognized for each
award, in the period in which each award vests, to reflect the actual
forfeitures related to that award. Changes in our estimated forfeiture rate will
result in changes in the rate at which compensation cost for an award is
recognized over its vesting period.
Changes
in the assumptions used to compute the fair value of the option awards are
likely to affect the fair value of the non-qualified stock option awards and the
amount of compensation expense recognized in future periods. A higher
volatility, shorter expected term and higher risk-free rate increases the
resulting compensation expense recognized in future periods as compared to prior
periods. Conversely, a lower volatility, longer expected term and lower
risk-free rate decreases the resulting compensation expense recognized in future
periods as compared to prior periods.
For performance-based stock option awards vesting of a defined portion of each
award will occur earlier if a defined performance condition is achieved; more
than one condition may be achieved in any period. We estimate the probability of
achievement of each performance condition and use those probabilities to
determine the requisite service period of each award. The requisite service
period for the award is the shortest of the explicit or implied service periods.
In the case of the executive’s options, the explicit service period is nine
years and eleven months from the respective grant dates. The implied service
periods related to the performance conditions are the estimated times for each
performance condition to be achieved. Thus, compensation expense will be
recognized over the shortest estimated time for the achievement of performance
conditions for that award (assuming that the performance conditions will be
achieved before the cliff vesting occurs). For performance and market-based
stock option awards (consisting of options and restricted stock in 2008 and
options in 2009) to our Chief Executive Officer vesting occurs on the basis of
the achievement of specified performance or market-based milestones. The options
have an exercise price equal to the closing price on our common stock on the
date of grant. The awards to our Chief Executive Officer are valued using a
Monte Carlo simulation and the expense related to these grants will be
recognized over the shortest estimated time for the achievement of the
performance or market conditions. The awards will not vest unless one of the
milestones is achieved or the market condition is met. Changes in the estimate
of probability of achievement of any performance or market condition will be
reflected in compensation expense of the period of change and future periods
affected by the change.
Research and Development Expenses Including Clinical Trial Expenses.
Clinical trial expenses, which are included in research and development
expenses, represent obligations resulting from our contracts with various
clinical investigators and clinical research organizations in connection with
conducting clinical trials for our product candidates. Such costs are expensed
as incurred, and are based on the expected total number of patients in the
trial, the rate at which the patients enter the trial and the period over which
the clinical investigators and clinical research organizations are expected to
provide services. We believe that this method best approximates the efforts
expended on a clinical trial with the expenses we record. We adjust our rate of
clinical expense recognition if actual results differ from our estimates. In
addition to clinical trial expenses, we estimate the amounts of other research
and development expenses, for which invoices have not been received at the end
of a period, based upon communication with third parties that have provided
services or goods during the period. Such estimates are subject to change as
additional information becomes available.
Fair Value Measurements. Our available-for-sale investment portfolio
consists of money market funds, corporate debt securities and auction rate
securities, and is recorded at fair value in the accompanying consolidated
balance sheets in accordance with ASC 320 Investments – Debt and Equity
Securities. The change in the fair value of these investments is recorded as a
component of other comprehensive loss.
We continue to monitor markets for our investments and consider the impact, if
any, of market conditions on the fair market value of our
investments.
We expect to recover the amortized cost of all of our investments at maturity.
Currently, we do not anticipate having to sell these securities in order to
operate our business and we believe that it is not more likely than not that we
will be required to sell these securities before recovery of principal. We do
not believe the carrying values of our investments are other than temporarily
impaired and therefore expect the positions will eventually be liquidated
without significant loss.
Valuation of securities is subject to uncertainties that are difficult to
predict, such as changes to credit ratings of the securities and/or the
underlying assets supporting them, default rates applicable to the underlying
assets, underlying collateral value, discount rates, counterparty risk, ongoing
strength and quality of market credit and liquidity and general economic and
market conditions. The valuation of the auction rate securities we hold is based
on an internal analysis of timing of expected future successful auctions,
collateralization of underlying assets of the security and credit quality of the
security.
Impact
of Recently Issued Accounting Standards
In October 2009, the FASB issued ASU 2009-13 to address the accounting for
multiple-deliverable arrangements. In an arrangement with multiple deliverables,
the delivered items shall be considered a separate unit of accounting if both
(i) the delivered items have value to a collaborator on a stand-alone basis, in
that, the collaborator could resell the delivered items on a stand-alone basis,
and (ii) the arrangement includes a general right of return relative to the
delivered item, delivery or performance of the undelivered item or items is
considered probable and substantially in our control. This ASU will be effective
prospectively for revenue arrangements entered into or materially modified in
fiscal years beginning on or after June 15, 2010. Early adoption is permitted.
We are currently evaluating the impact this ASU will have on our financial
statements.
Item
7A. Quantitative and Qualitative Disclosures about Market
Risk
Our primary investment objective is to preserve principal while maximizing yield
without significantly increasing our risk. Our investments consist of money
market funds, taxable corporate debt securities and auction rate securities. Our
investments totaled $89.5 million at December 31, 2009, of which $1.5 million
(2%) had fixed interest rates and $88.0 million (98%) had interest rates that
were variable. As a result, we do not believe that we have a material exposure
to interest-rate risk. Our marketable securities are classified as
available-for-sale.
As a result of changes in general market conditions during 2008, we determined
to reduce the principal amount of auction rate securities in our portfolio as
they came up for auction and invest the proceeds in other securities in
accordance with our investment guidelines. At December 31, 2009, we continue to
hold approximately $3.8 million (4.2% of assets measured at fair value) of
auction rate securities, in respect of which we have received all scheduled
interest payments and which, in the event of auction failure, have interest
rates reset according to the contractual terms in the governing instruments. The
principal amount of these remaining auction rate securities will not be
accessible until a successful auction occurs, the issuer calls or restructures
the underlying security, the underlying security matures and is paid or a buyer
outside the auction process emerges.
We continue to monitor the market for auction rate securities and consider the
impact, if any, of market conditions on the fair market value of our
investments. We believe that the failed auctions experienced to date are not a
result of the deterioration of the underlying credit quality of these
securities, although valuation of them is subject to uncertainties that are
difficult to predict, such as changes to credit ratings of the securities and/or
the underlying assets supporting them, default rates applicable to the
underlying assets, underlying collateral value, discount rates, counterparty
risk, ongoing strength and quality of market credit and liquidity, and general
economic and market conditions. We do not believe the carrying values of these
auction rate securities are other than temporarily impaired and therefore expect
the positions will eventually be liquidated without significant
loss.
The valuation of the auction rate securities we hold is based on an internal
analysis of timing of expected future successful auctions, collateralization of
underlying assets of the security and credit quality of the security. We
re-evaluated the valuation of these securities as of December 31, 2009 and the
temporary impairment amount decreased $8.0 thousand from $316.0 thousand at
December 31, 2008 to $308.0 thousand. A 100 basis point increase to our internal
analysis would result in an insignificant increase in the temporary impairment
of these securities as of the year ended December 31, 2009.
Item
8. Financial Statements and Supplementary
Data
See page F-1, Index to
Consolidated Financial Statements.
Item
9. Changes in and Disagreements With Accountants on
Accounting and Financial Disclosure
None.
Item
9A. Controls and Procedures
Evaluation
of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that
information required to be disclosed in our Exchange Act reports is recorded,
processed, summarized and reported within the timelines specified in the SEC’s
rules and forms, and that such information is accumulated and communicated to
our management, including our Chief Executive Officer and Chief Financial
Officer, as appropriate, to allow timely decisions regarding required
disclosure. In designing and evaluating the disclosure controls and procedures,
management recognized that any controls and procedures, no matter how well
designed and operated, can only provide reasonable assurance of achieving the
desired control objectives, and in reaching a reasonable level of assurance,
management necessarily was required to apply its judgment in evaluating the
cost-benefit relationship of possible controls and procedures. We have a
Disclosure Committee consisting of members of our senior management which
monitors and implements our policy of disclosing material information concerning
the Company in accordance with applicable law.
As required by SEC Rule 13a-15(e), we carried out an evaluation, under the
supervision and with the participation of our management, including our Chief
Executive Officer and our Chief Financial Officer, of the effectiveness of the
design and operation of our disclosure controls and procedures as of the end of
the period covered by this report. Based on the foregoing, our Chief Executive
Officer and Chief Financial Officer concluded that our current disclosure
controls and procedures, as designed and implemented, were effective at the
reasonable assurance level.
Changes
in Internal Control over Financial Reporting
There have been no changes in our internal control over financial reporting, as
such term is defined in the Exchange Act Rules 13a-15(f) and 15d-15(f) during
our fiscal quarter ended December 31, 2009 that have materially affected, or are
reasonably likely to materially affect, our internal control over financial
reporting.
Management’s
Report on Internal Control Over Financial Reporting
Internal control over financial reporting is a process designed by, or under the
supervision of, our principal executive and principal financial officers and
effected by our Board, management and other personnel to provide reasonable
assurance regarding the reliability of financial reporting and the preparation
of financial statements for external purposes in accordance with generally
accepted accounting principles. It includes policies and procedures
that:
· Pertain to the maintenance of records that in
reasonable detail accurately and fairly reflect the transactions and
dispositions of our assets;
· Provide reasonable assurance that transactions
are recorded as necessary to permit preparation of financial statements in
accordance with generally accepted accounting
principles,
and that our receipts and expenditures are being made only in accordance with
authorization of our management and directors; and
· Provide reasonable assurance regarding
prevention or timely detection of unauthorized acquisition, use or disposition
of our assets that could have a material effect on
the financial statements.
Because of its inherent limitations, internal control over financial reporting
may not prevent or detect misstatements. Projections of any evaluation of
effectiveness to future periods are subject to the risk that controls may become
inadequate because of changes in conditions, or that the degree of compliance
with the policies or procedures may deteriorate.
Management has used the framework set forth in the report entitled Internal Control –
Integrated
Framework issued by the Committee of Sponsoring Organizations of the
Treadway Commission, known as COSO, to evaluate the effectiveness of our
internal control over financial reporting. Management has concluded that our
internal control over financial reporting was effective as of December 31, 2009.
The effectiveness of our internal control over financial reporting as of
December 31, 2009 has been audited by PricewaterhouseCoopers LLP, an independent
registered public accounting firm, as stated in their report which appears
herein.
Item
9B. Other Information
None.
PART
III
The
information required by the Form 10-K Items listed in the following table will
be included under the respective headings specified for such Items in our
definitive proxy statement for our 2010 Annual Meeting of Stockholders to be
filed with the SEC:
|
Item of Form 10-K
|
Location in 2010 Proxy
Statement
|
|
Item
10. Directors, Executive Officers and Corporate
Governance
|
Election
of Directors.
Board
and Committee Meetings.
Executive
Officers of the Company.
Section
16(a) Beneficial Ownership Reporting and Compliance.
Code
of Business Ethics and Conduct.*
*The full text of our code of
business ethics and conduct is available on our website
(http://www.progenics.com/documents.cfm).
|
|
Item
11. Executive Compensation
|
Executive
Compensation.
Compensation
Committee Report.
Compensation
Committee Interlocks and Insider Participation.
|
|
Item
12.Security Ownership of Certain Beneficial Owners and Management and
Related Stockholder Matters
|
Equity
Compensation Plan Information.
Security Ownership of Certain
Beneficial Owners and Management.
|
|
Item
13. Certain Relationships and Related Transactions, and Director
Independence
|
Certain
Relationships and Related Transactions.
Affirmative Determinations
Regarding Director Independence and Other
Matters.
|
|
Item
14. Principal Accounting Fees and Services
|
Fees
Billed for Services Rendered by our Independent Registered Public
Accounting Firm.
Pre-approval
of Audit and Non-Audit Services by the Audit
Committee.
|
PART
IV
Item
15. Exhibits, Financial Statement Schedules
The following documents or the portions thereof indicated are filed as a part of
this Report.
|
|
(a)
|
Documents
filed as part of this Report:
|
|
|
Consolidated
Financial Statements of Progenics Pharmaceuticals,
Inc.:
|
Report of Independent Registered Public Accounting Firm
Consolidated Balance Sheets at December 31, 2009 and 2008
Consolidated Statements of Operations for the years ended December 31, 2009,
2008 and 2007
Consolidated Statements of Stockholders’ Equity and Comprehensive Loss for the
years ended December 31, 2009, 2008 and 2007
Consolidated
Statements of Cash Flows for the years ended December 31, 2009, 2008 and
2007
Notes to Consolidated Financial Statements
|
|
(b)
|
Financial
Statement Schedules
|
All financial statement schedules referred to in Item 12-01 of
Regulation S-X are inapplicable and therefore have been omitted.
|
|
(c)
|
Item
601 Exhibits
|
Those exhibits required to be filed by Item 601 of Regulation S-K are listed in
the Exhibit Index immediately following the signature page hereof and preceding
the
exhibits filed herewith, and such listing is incorporated herein by
reference.
PROGENICS
PHARMACEUTICALS, INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
|
Page
|
|
|
Report
of Independent Registered Public Accounting Firm
|
F-2
|
|
Financial
Statements:
|
|
|
Consolidated
Balance Sheets at December 31, 2009 and 2008
|
F-3
|
|
Consolidated
Statements of Operations for the years ended December 31, 2009, 2008 and
2007
|
F-4
|
|
Consolidated
Statements of Stockholders’ Equity and Comprehensive Loss
for
the years ended December 31, 2009, 2008 and 2007
|
F-5
|
|
Consolidated
Statements of Cash Flows for the years ended December 31, 2009, 2008 and
2007
|
F-6
|
|
Notes
to Consolidated Financial Statements
|
F-7
|
Report
of Independent Registered Public Accounting Firm
To
the Board of
Directors and Stockholders of
Progenics
Pharmaceuticals, Inc.:
In our
opinion, the consolidated financial statements listed in the index appearing
under Item 15(a)(1) present fairly, in all material respects, the financial
position of Progenics Pharmaceuticals, Inc. and its subsidiaries at December 31,
2009 and 2008, and the results of their operations and their cash flows for each
of the three years in the period ended December 31, 2009 in conformity with
accounting principles generally accepted in the United States of
America. Also in our opinion, the Company maintained, in all material
respects, effective internal control over financial reporting as of December 31,
2009, based on criteria established in Internal Control - Integrated
Framework issued by the Committee of Sponsoring Organizations of the
Treadway Commission (COSO). The Company's management is responsible for these
financial statements and for maintaining effective internal control over
financial reporting and for its assessment of the effectiveness of internal
control over financial reporting, included in Management's Report on Internal
Control Over Financial Reporting appearing under Item 9A. Our responsibility is
to express opinions on these financial statements, and on the Company's internal
control over financial reporting based on our integrated audits. We conducted
our audits in accordance with the standards of the Public Company Accounting
Oversight Board (United States). Those standards require that we plan and
perform the audits to obtain reasonable assurance about whether the financial
statements are free of material misstatement and whether effective internal
control over financial reporting was maintained in all material respects. Our
audits of the financial statements included examining, on a test basis, evidence
supporting the amounts and disclosures in the financial statements, assessing
the accounting principles used and significant estimates made by management, and
evaluating the overall financial statement presentation. Our audit of internal
control over financial reporting included obtaining an understanding of internal
control over financial reporting, assessing the risk that a material weakness
exists, and testing and evaluating the design and operating effectiveness of
internal control based on the assessed risk. Our audits also included performing
such other procedures as we considered necessary in the circumstances. We
believe that our audits provide a reasonable basis for our
opinions.
As
discussed in Note 2 to the consolidated financial statements, the Company
changed the manner in which it accounts for uncertainties in income taxes in
2007 and the manner in which it accounts for fair value measurements for its
financial assets and financial liabilities in 2008.
A
company’s internal control over financial reporting is a process designed to
provide reasonable assurance regarding the reliability of financial reporting
and the preparation of financial statements for external purposes in accordance
with generally accepted accounting principles. A company’s internal control over
financial reporting includes those policies and procedures that (i) pertain to
the maintenance of records that, in reasonable detail, accurately and fairly
reflect the transactions and dispositions of the assets of the company; (ii)
provide reasonable assurance that transactions are recorded as necessary to
permit preparation of financial statements in accordance with generally accepted
accounting principles, and that receipts and expenditures of the company are
being made only in accordance with authorizations of management and directors of
the company; and (iii) provide reasonable assurance regarding prevention or
timely detection of unauthorized acquisition, use, or disposition of the
company’s assets that could have a material effect on the financial
statements.
Because
of its inherent limitations, internal control over financial reporting may not
prevent or detect misstatements. Also, projections of any evaluation of
effectiveness to future periods are subject to the risk that controls may become
inadequate because of changes in conditions, or that the degree of compliance
with the policies or procedures may deteriorate.
/s/
PricewaterhouseCoopers LLP
New York,
New York
March 10,
2010
CONSOLIDATED
BALANCE SHEETS
(in
thousands, except for par value and share amounts)
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Assets
|
||||||||
|
Current
assets:
|
||||||||
|
Cash
and cash equivalents
|
$ | 90,903 | $ | 56,186 | ||||
|
Marketable
securities
|
1,501 | 63,127 | ||||||
|
Accounts
receivable
|
7,522 | 1,337 | ||||||
|
Other
current assets
|
1,468 | 3,531 | ||||||
|
Total
current assets
|
101,394 | 124,181 | ||||||
|
Marketable
securities
|
3,792 | 22,061 | ||||||
|
Fixed
assets, at cost, net of accumulated depreciation and
amortization
|
6,560 | 11,071 | ||||||
|
Other
assets
|
1,867 | 520 | ||||||
|
Total
assets
|
$ | 113,613 | $ | 157,833 | ||||
|
Liabilities and
Stockholders’
Equity
|
||||||||
|
Current
liabilities:
|
||||||||
|
Accounts
payable and accrued expenses
|
$ | 5,836 | $ | 6,496 | ||||
|
Deferred
revenue - current
|
- | 31,645 | ||||||
|
Other
current liabilities
|
170 | 57 | ||||||
|
Total
current liabilities
|
6,006 | 38,198 | ||||||
|
Other
liabilities
|
- | 266 | ||||||
|
Total
liabilities
|
6,006 | 38,464 | ||||||
|
Commitments
and contingencies (Note 8)
|
||||||||
|
Stockholders’
equity:
|
||||||||
|
Preferred
stock, $.001 par value; 20,000,000 shares authorized; issued and
outstanding - none
|
- | - | ||||||
|
Common
stock, $.0013 par value; 40,000,000 shares authorized; issued - 32,142,062
in 2009 and 30,807,387 in 2008
|
42 | 40 | ||||||
|
Additional
paid-in capital
|
439,943 | 422,085 | ||||||
|
Accumulated
deficit
|
(329,330 | ) | (298,718 | ) | ||||
|
Accumulated
other comprehensive loss
|
(307 | ) | (1,297 | ) | ||||
|
Treasury
stock, at cost (200,000 shares in 2009 and 2008)
|
(2,741 | ) | (2,741 | ) | ||||
|
Total
stockholders’ equity
|
107,607 | 119,369 | ||||||
|
Total
liabilities and stockholders’ equity
|
$ | 113,613 | $ | 157,833 | ||||
The
accompanying notes are an integral part of the financial
statements.
CONSOLIDATED
STATEMENTS OF OPERATIONS
(in
thousands, except for loss per share data)
|
Years
Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Revenues:
|
||||||||||||
|
Research
and development
|
$ | 44,351 | $ | 59,885 | $ | 65,455 | ||||||
|
Royalty
income
|
2,372 | 146 | - | |||||||||
|
Research
grants and contract
|
1,968 | 7,460 | 10,075 | |||||||||
|
Other
revenues
|
256 | 180 | 116 | |||||||||
|
Total
revenues
|
48,947 | 67,671 | 75,646 | |||||||||
|
Expenses:
|
||||||||||||
|
Research
and development
|
49,798 | 82,290 | 95,234 | |||||||||
|
License
fees - research and development
|
1,058 | 2,830 | 942 | |||||||||
|
General
and administrative
|
25,106 | 28,834 | 27,901 | |||||||||
|
Royalty
expense
|
237 | 15 | - | |||||||||
|
Depreciation
and amortization
|
5,078 | 4,609 | 3,027 | |||||||||
|
Total
expenses
|
81,277 | 118,578 | 127,104 | |||||||||
|
Operating
loss
|
(32,330 | ) | (50,907 | ) | (51,458 | ) | ||||||
|
Other
income:
|
||||||||||||
|
Interest
income
|
1,481 | 6,235 | 7,770 | |||||||||
|
Gain
on sale of marketable securities
|
237 | - | - | |||||||||
|
Total
other income
|
1,718 | 6,235 | 7,770 | |||||||||
|
Net
loss
|
$ | (30,612 | ) | $ | (44,672 | ) | $ | (43,688 | ) | |||
|
Net
loss per share - basic and diluted
|
$ | (0.98 | ) | $ | (1.48 | ) | $ | (1.59 | ) | |||
|
Weighted-average
shares - basic and diluted
|
31,219 | 30,142 | 27,447 | |||||||||
The
accompanying notes are an integral part of the financial
statements.
CONSOLIDATED
STATEMENTS OF STOCKHOLDERS’ EQUITY AND COMPREHENSIVE LOSS
For
the Years Ended December 31, 2009, 2008 and 2007
(in thousands)
|
Common
Stock
|
Additional
|
Accumulated
Other
|
Treasury
Stock
|
|||||||||||||||||||||||||||||
|
Shares
|
Amount
|
Paid-In
Capital
|
Accumulated
Deficit
|
Comprehensive
(Loss) Income
|
Shares
|
Amount
|
Total
|
|||||||||||||||||||||||||
|
Balance
at December 31, 2006
|
26,199 | $ | 34 | $ | 321,315 | $ | (210,358 | ) | $ | (145 | ) | - | $ | - | $ | 110,846 | ||||||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||||||
|
Net
loss
|
- | - | - | (43,688 | ) | - | - | - | (43,688 | ) | ||||||||||||||||||||||
|
Net
change in unrealized gain on marketable securities
|
- | - | - | - | 151 | - | - | 151 | ||||||||||||||||||||||||
|
Total
comprehensive loss:
|
(43,537 | ) | ||||||||||||||||||||||||||||||
|
Compensation
expenses for share-based payment arrangements
|
- | - | 15,306 | - | - | - | - | 15,306 | ||||||||||||||||||||||||
|
Issuance
of restricted stock, net of forfeitures
|
267 | - | - | - | - | - | - | - | ||||||||||||||||||||||||
|
Sale
of common stock in a public offering ($23.15 per share, net of
underwriting discounts and commissions and other offering expenses of
$3,112) (see Note 7)
|
2,600 | 3 | 57,075 | - | - | - | - | 57,078 | ||||||||||||||||||||||||
|
Sale
of common stock under employee stock purchase plans and exercise of stock
options
|
688 | 2 | 7,823 | - | - | - | - | 7,825 | ||||||||||||||||||||||||
|
Repurchase
of restricted stock
|
- | - | (19 | ) | - | - | - | - | (19 | ) | ||||||||||||||||||||||
|
Balance
at December 31, 2007
|
29,754 | 39 | 401,500 | (254,046 | ) | 6 | - | - | 147,499 | |||||||||||||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||||||
|
Net
loss
|
- | - | - | (44,672 | ) | - | - | - | (44,672 | ) | ||||||||||||||||||||||
|
Net
change in unrealized gain on marketable securities
|
- | - | - | - | (1,303 | ) | - | - | (1,303 | ) | ||||||||||||||||||||||
|
Total
comprehensive loss:
|
(45,975 | ) | ||||||||||||||||||||||||||||||
|
Compensation
expenses for share-based payment arrangements
|
- | - | 14,133 | - | - | - | - | 14,133 | ||||||||||||||||||||||||
|
Issuance
of restricted stock, net of forfeitures
|
216 | - | - | - | - | - | - | - | ||||||||||||||||||||||||
|
Sale
of common stock under employee stock purchase plans and exercise of stock
options
|
837 | 1 | 6,452 | - | - | - | - | 6,453 | ||||||||||||||||||||||||
|
Treasury
shares acquired under repurchase program
|
- | - | - | - | - | (200 | ) | (2,741 | ) | (2,741 | ) | |||||||||||||||||||||
|
Balance
at December 31, 2008
|
30,807 | 40 | 422,085 | (298,718 | ) | (1,297 | ) | (200 | ) | (2,741 | ) | 119,369 | ||||||||||||||||||||
|
Comprehensive
loss:
|
||||||||||||||||||||||||||||||||
|
Net
loss
|
- | - | - | (30,612 | ) | - | - | - | (30,612 | ) | ||||||||||||||||||||||
|
Net
change in unrealized loss on marketable securities
|
- | - | - | - | 990 | - | - | 990 | ||||||||||||||||||||||||
|
Total
comprehensive loss:
|
(29,622 | ) | ||||||||||||||||||||||||||||||
|
Compensation
expenses for share-based payment arrangements
|
- | - | 12,986 | - | - | - | - | 12,986 | ||||||||||||||||||||||||
|
Issuance
of restricted stock, net of forfeitures
|
266 | - | - | - | - | - | - | - | ||||||||||||||||||||||||
|
Sale
of common stock under employee stock purchase plans and exercise of stock
options
|
1,069 | 2 | 4,872 | - | - | - | - | 4,874 | ||||||||||||||||||||||||
|
Balance
at December 31, 2009
|
32,142 | $ | 42 | $ | 439,943 | $ | (329,330 | ) | $ | (307 | ) | (200 | ) | $ | (2,741 | ) | $ | 107,607 | ||||||||||||||
The
accompanying notes are an integral part of the financial
statements.
CONSOLIDATED
STATEMENTS OF CASH FLOWS
(in
thousands)
|
Years
Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Cash
flows from operating activities:
|
||||||||||||
|
Net
loss
|
$ | (30,612 | ) | $ | (44,672 | ) | $ | (43,688 | ) | |||
|
Adjustments
to reconcile net loss to net cash used in operating
activities:
|
||||||||||||
|
Depreciation
and amortization
|
5,078 | 4,609 | 3,027 | |||||||||
|
Write-off
of fixed assets
|
334 | 3 | - | |||||||||
|
Amortization
of discounts, net of premiums, on marketable securities
|
889 | 960 | (445 | ) | ||||||||
|
Expenses
for share-based compensation awards
|
12,986 | 14,133 | 15,306 | |||||||||
|
Gain
on sale of marketable securities
|
(237 | ) | - | - | ||||||||
|
Changes
in assets and liabilities:
|
||||||||||||
|
(Increase)
decrease in accounts receivable
|
(6,185 | ) | 658 | (296 | ) | |||||||
|
Decrease
(increase) in other current assets
|
2,063 | (420 | ) | 70 | ||||||||
|
Increase
in other assets
|
(1,667 | ) | - | - | ||||||||
|
(Decrease)
increase in accounts payable and accrued expenses
|
(660 | ) | (8,269 | ) | 2,913 | |||||||
|
(Decrease)
increase in deferred revenue
|
(31,645 | ) | 4,786 | (16,231 | ) | |||||||
|
Increase
in other current liabilities
|
113 | - | 57 | |||||||||
|
(Decrease)
increase in other liabilities
|
(266 | ) | (93 | ) | 236 | |||||||
|
Net
cash used in operating activities
|
(49,809 | ) | (28,305 | ) | (39,051 | ) | ||||||
|
Cash
flows from investing activities:
|
||||||||||||
|
Capital
expenditures
|
(901 | ) | (2,172 | ) | (5,151 | ) | ||||||
|
Sales/maturities
of marketable securities
|
80,233 | 128,705 | 252,850 | |||||||||
|
Purchase
of marketable securities
|
- | (56,209 | ) | (275,048 | ) | |||||||
|
Decrease
(increase) in restricted cash
|
320 | 32 | (8 | ) | ||||||||
|
Net
cash provided by (used in) investing activities
|
79,652 | 70,356 | (27,357 | ) | ||||||||
|
Cash
flows from financing activities:
|
||||||||||||
|
Proceeds
from sale of common stock in public offering
|
- | - | 60,190 | |||||||||
|
Expenses
related to the sale of common stock in public offering
|
- | - | (3,112 | ) | ||||||||
|
Purchase
of treasury stock
|
- | (2,741 | ) | - | ||||||||
|
Proceeds
from the exercise of stock options and sale of common stock under the
Employee Stock Purchase Plan
|
4,874 | 6,453 | 7,825 | |||||||||
|
Repurchase
of restricted stock
|
- | - | (19 | ) | ||||||||
|
Net
cash provided by financing activities
|
4,874 | 3,712 | 64,884 | |||||||||
|
Net
increase (decrease) in cash and cash equivalents
|
34,717 | 45,763 | (1,524 | ) | ||||||||
|
Cash
and cash equivalents at beginning of period
|
56,186 | 10,423 | 11,947 | |||||||||
|
Cash
and cash equivalents at end of period
|
$ | 90,903 | $ | 56,186 | $ | 10,423 | ||||||
The
accompanying notes are an integral part of the financial
statements.
F-6
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
(amounts
in thousands, except per share amounts or unless otherwise noted)
1.
Organization and Business
Progenics
Pharmaceuticals, Inc. (“Progenics,” “we” or “us”) is a biopharmaceutical company
focusing on the development and commercialization of innovative therapeutic
products to treat the unmet medical needs of patients with debilitating
conditions and life-threatening diseases. Our principal programs are directed
toward supportive care, oncology and virology.
Progenics
commenced principal operations in 1988, became publicly traded in 1997 and
throughout has been engaged primarily in research and development efforts,
developing manufacturing capabilities, establishing corporate collaborations and
raising capital. Certain of our intellectual property rights are held by wholly
owned subsidiaries. None of our subsidiaries other than PSMA Development Company
LLC (“PSMA LLC”) had operations during the years ended December 31, 2009, 2008
or 2007. All of our operations are conducted at our facilities in Tarrytown, New
York.
Supportive
Care
Our first
commercial product is RELISTOR®
(methylnaltrexone bromide) subcutaneous injection, a first-in-class therapy for
opioid-induced constipation approved for sale in over 40 countries worldwide,
including the United States, European Union member states, Canada, Australia and
Brazil. Marketing applications are pending elsewhere throughout the
world.
In
October 2009, we and Wyeth Pharmaceuticals terminated our 2005 RELISTOR
collaboration, as a result of which we regained all worldwide rights to
RELISTOR. Under our Transition Agreement, Wyeth is continuing to market and sell
RELISTOR for a U.S. Sales Period ending September 30, 2010 and an ex-U.S. Sales
Period ending December 31, 2010. After this transition period, we will assume
full control of and responsibility for future development and commercialization
of RELISTOR. Shortly after the termination, Pfizer Inc. completed its
previously-announced acquisition of Wyeth, which is now a wholly owned
subsidiary of Pfizer.
We are
pursuing a range of strategic alternatives for RELISTOR, including licensing,
collaboration and/or strategic alliances with world-wide or regional partners,
U.S. commercialization of the currently-approved product on our own or with
pharmaceutical detailing and sales organizations and/or co-promotion of the
franchise with a partner using our own sales force.
Under the
Transition Agreement, Wyeth has agreed to pay to us the sum of $10.0 million in
six quarterly installments and is continuing certain ongoing development efforts
for subcutaneous RELISTOR, at its expense, through September 30, 2010. Wyeth’s
international sales and marketing obligations during the ex-U.S. Sales Period
are subject to certain extension and early transition options available to us.
We have no further obligations to Wyeth under the 2005 collaboration agreement.
Wyeth will continue to pay royalties on worldwide sales as provided in the 2005
collaboration agreement except that no royalties will be payable in respect of
ex-U.S. sales during (i) the fourth quarter of 2010 to the extent certain
financial targets are not met or (ii) an extended ex-U.S. Sales Period in the
subject country. Principal responsibility for regulatory submissions and
interactions for all other formulations and presentations of RELISTOR will be
transferred during and as part of the transition. Wyeth is also providing
financial resources, aggregating up to approximately $14.5 million, and/or other
assistance with respect to agreed-upon regulatory, manufacturing and supply
matters. We have agreed to purchase Wyeth’s remaining inventory of subcutaneous
RELISTOR at the end of the Sales Periods on agreed-upon terms and
conditions.
Prior to
the Transition Agreement (including periods covered by this report), we received
upfront, milestone, and royalty payments from Wyeth, and were reimbursed for
expenses we incurred in connection with the development of RELISTOR;
manufacturing and commercialization expenses for RELISTOR were funded by
Wyeth.
Our October 2008 out-license to Ono
Pharmaceutical of the rights to subcutaneous RELISTOR in Japan is
unaffected by termination of the Wyeth collaboration. Under the License
Agreement, we received an upfront payment of $15.0 million, and are entitled to
receive potential milestones, upon achievement of development milestones by Ono,
of up to $20.0 million, commercial milestones and royalties on sales by Ono of
subcutaneous RELISTOR in Japan. Ono also has the option to acquire from us the
rights to develop and commercialize in Japan other formulations of RELISTOR,
including intravenous and oral forms, on terms to be negotiated
separately.
F-7
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Oncology
In the
area of prostate cancer, we are conducting a phase 1 clinical trial of a fully
human monoclonal antibody-drug conjugate (“ADC”) directed against prostate
specific membrane antigen (“PSMA”), a protein found at high levels on the
surface of prostate cancer cells and also on the neovasculature of a number of
other types of solid tumors. We are also developing therapeutic vaccines
designed to stimulate an immune response to PSMA. Our PSMA programs are
conducted through our wholly owned subsidiary, PSMA Development Company
LLC.
Virology
In the
area of virology, we are developing a viral-entry inhibitor -- a humanized
monoclonal antibody, PRO 140 -- for infection due to human immunodeficiency
virus (“HIV”), the virus that causes AIDS. We are developing the subcutaneous
form of PRO 140 for treatment of HIV infection, which has the potential for
weekly self-administration. Advancement of this program is subject to our
obtaining pivotal clinical-trial funding, for which we have applied to
government agencies. In our hepatitis C virus infection efforts, we are
evaluating second-generation HCV-entry inhibitors as possible development
candidates. We are also engaged in research regarding a prophylactic vaccine
against HIV infection.
Our
oncology and virology product candidates are not as advanced in development as
RELISTOR, and we do not expect any recurring revenues from sales or otherwise
with respect to these product candidates in the near term.
Funding
and Financial Matters
We will
require additional funding to continue our current programs to completion, which
may involve collaboration agreement, license or sale transactions or royalty
sales or financings with respect to our products and product candidates. We may
also seek to raise additional capital through sales of common stock or other
securities, and expect to continue funding some programs in part through
government awards.
Progenics
has had recurring losses since inception. At December 31, 2009, we had cash,
cash equivalents and marketable securities, including non-current portion,
totaling $96.2 million which we expect will be sufficient to fund current
operations beyond one year. During the year ended December 31, 2009, we had a
net loss of $30.6 million and used $49.8 million of cash in operating
activities. At December 31, 2009, we had an accumulated deficit of $329.3
million.
Pending
use in our business, our revenues and proceeds of financing activities are held
in cash, cash equivalents and marketable securities. Our marketable securities,
which include corporate debt securities and auction rate securities, are
classified as available-for-sale.
In April,
2008, our Board of Directors approved a share repurchase program to acquire up
to $15.0 million of our outstanding common shares, under which we have $12.3
million remaining available. Purchases may be discontinued at any time. We did
not repurchase any common shares during the year ended December 31,
2009.
2.
Summary of Significant Accounting Policies
Basis
of Presentation
The
consolidated financial statements have been prepared on the basis of accounting
principles generally accepted in the United States of America (“GAAP”). The
preparation of financial statements in conformity with GAAP requires management
to make estimates and assumptions that affect the amounts reported in the
financial statements and the accompanying notes. As additional information
becomes available or actual amounts become determinable, the recorded estimates
are revised and reflected in the operating results. Actual results could differ
from those estimates.
Consolidation
The
consolidated financial statements include the accounts of Progenics and PSMA
LLC, as of and for the years ended December 31, 2009, 2008 and 2007.
Inter-company transactions have been eliminated in
consolidation.
F-8
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Revenue
Recognition
We
recognize revenue from all sources based on the provisions of the SEC’s Staff
Accounting Bulletin (“SAB”) No. 104 (“SAB 104”) and ASC 605 Revenue
Recognition.
Collaborations
may contain substantive milestone payments to which we apply the substantive
milestone method (“Substantive Milestone Method”). Substantive milestone
payments are considered to be performance payments that are recognized upon
achievement of the milestone only if all of the following conditions are met:
(i) the milestone payment is non-refundable, (ii) achievement of the milestone
involves a degree of risk and was not reasonably assured at the inception of the
arrangement, (iii) substantive effort is involved in achieving the milestone,
(iv) the amount of the milestone payment is reasonable in relation to the effort
expended or the risk associated with achievement of the milestone, and (v) a
reasonable amount of time passes between the upfront license payment and the
first milestone payment as well as between each subsequent milestone
payment.
Determination
as to whether a milestone meets the aforementioned conditions involves
management’s judgment. If any of these conditions are not met, the resulting
payment would not be considered a substantive milestone and, therefore, the
resulting payment would be part of the consideration and be recognized as
revenue as such performance obligations are performed.
Non-refundable
upfront license fees are recognized as revenue when we have a contractual right
to receive such payment, the contract price is fixed or determinable, the
collection of the resulting receivable is reasonably assured and we have no
further performance obligations. Multiple element arrangements, such as license
and development arrangements are analyzed to determine whether the deliverables,
which often include a license and performance obligations, such as research and
steering or other committee services, can be separated in accordance with ASC
605 Revenue Recognition. We would recognize upfront license payments as revenue
upon delivery of the license only if the license had standalone value and the
fair value of the undelivered performance obligations could be determined. If
the fair value of the undelivered performance obligations could be determined,
such obligations would then be accounted for separately as performed. If the
license is considered to either (i) not have standalone value, or (ii) have
standalone value but the fair value of any of the undelivered performance
obligations could not be determined, the upfront license payments would be
recognized as revenue over the estimated period of when our performance
obligations are performed. Any unamortized remainder of the upfront payment is
recognized upon termination of collaborations.
We must
determine the period over which our performance obligations are performed and
revenue related to upfront license payments are recognized. Revenue is
recognized using either a proportionate performance or straight-line method. We
recognize revenue using the proportionate performance method provided that we
can reasonably estimate the level of effort required to complete our performance
obligations under an arrangement and such performance obligations are provided
on a best-efforts basis. Direct labor hours or full-time equivalents will
typically be used as the measure of performance. Under the proportionate
performance method, revenue related to upfront license payments is recognized in
any period as the percent of actual effort expended in that period relative to
total effort for all of our performance obligations under the
arrangement.
During
the course of a collaboration agreement that involves a development plan and
budget, the amount of the upfront license payment that is recognized as revenue
in any period increases or decreases as the percentage of actual effort
increases or decreases. When a new budget is approved, the remaining
unrecognized amount of the upfront license fee is recognized prospectively, by
applying the changes in the total estimated effort or period of development that
is specified in the revised approved budget.
If we
cannot reasonably estimate the level of effort required to complete our
performance obligations under an arrangement and the performance obligations are
provided on a best-efforts basis, then the total upfront license payments would
be recognized as revenue on a straight-line basis over the period we expect to
complete our performance obligations.
If we are
involved in a steering or other committee as part of a multiple element
arrangement, we assess whether our involvement constitutes a performance
obligation or a right to participate. For those committees that are deemed
obligations, we will evaluate our participation along with other obligations in
the arrangement and will attribute revenue to our participation through the
period of our committee responsibilities.
F-9
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
We
recognize revenue for payments that are contingent upon performance solely by
our collaborator immediately upon the achievement of the defined event if we
have no related performance obligations.
Reimbursement
of costs is recognized as revenue provided the provisions of ASC 605 Revenue
Recognition are met, the amounts are determinable and collection of the related
receivable is reasonably assured.
Amounts
received prior to satisfying the above revenue recognition criteria are recorded
as deferred revenue in the accompanying consolidated balance sheets. Amounts not
expected to be recognized within one year of the balance sheet date are
classified as long-term deferred revenue. The estimate of the classification of
deferred revenue as short-term or long-term is based upon management’s current
operating budget with the collaborator for the total effort required to complete
our performance obligations under the arrangement.
Royalty
revenue is recognized in the period the sales occur, provided that the royalty
amounts are fixed or determinable, collection of the related receivable is
reasonably assured and we have no remaining performance obligations under the
arrangement providing for the royalty. If royalties are received when we have
remaining performance obligations, they would be attributed to the services
being provided under the arrangement and, therefore, recognized as such
obligations are performed under either the proportionate performance or
straight-line methods, as applicable.
During
the years ended December 31, 2009, 2008 and 2007, we also recognized revenue
from government research grants (and contract in the 2008 and 2007 periods),
which are used to subsidize a portion of certain of our research projects
(“Projects”), exclusively from the National Institutes of Health (“NIH”). We
also recognized revenue from the sale of research reagents during those
periods.
NIH grant
and contract revenue is recognized as efforts are expended and as related
subsidized project costs are incurred. We perform work under the NIH grants and
contract on a best-effort basis. The NIH reimburses us for costs associated with
projects in the fields of virology and cancer, including pre-clinical research,
development and early clinical testing of a prophylactic vaccine designed to
prevent HIV from becoming established in uninfected individuals exposed to the
virus, as requested by the NIH.
Transition
Agreement with Wyeth – October 2009
The
Transition Agreement provides for the termination of the 2005 Wyeth
collaboration agreement and the transition to Progenics of the rights to develop
and commercialize RELISTOR. Under it, Wyeth’s license of Progenics technology is
terminated except as necessary for performance of Wyeth’s obligations during the
transition period and Wyeth has returned the rights to RELISTOR that we had
previously granted under the 2005 collaboration agreement. During the
transition, Wyeth is obligated to pay all costs of commercialization of
subcutaneous RELISTOR, including manufacturing costs, and retains all proceeds
from its sale of the products, subject to royalties due to us. Decisions with
respect to commercialization of the product during the transition period are to
be made solely by Wyeth. We have no further obligations to Wyeth under the 2005
collaboration agreement.
Wyeth
Collaboration Agreement – December 2005 to October 2009
The Wyeth
collaboration agreement was in effect until October 2009 which includes periods
covered by this report. The Wyeth Collaboration Agreement involved three
formulations of RELISTOR: (i) a subcutaneous formulation to be used in patients
with opioid-induced constipation (“OIC”), (ii) an intravenous formulation to be
used in patients with post-operative ileus (“POI”) and (iii) an oral formulation
to be used in patients with OIC.
The Wyeth
Collaboration Agreement established a Joint Steering Committee (“JSC”) and a
Joint Development Committee (“JDC”), each with an equal number of
representatives from both Wyeth and us. The JSC coordinated the companies’ key
activities, while the JDC coordinated the development of RELISTOR by Wyeth and
us. A Joint Commercialization Committee (“JCC”), composed of company
representatives in number and function according to our respective
responsibilities, facilitated open communication between Wyeth and us on
commercialization matters.
F-10
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
The
agreement included a non-refundable upfront license fee, reimbursement of
development costs, research and development payments based upon our achievement
of clinical development milestones, contingent payments based upon the
achievement by Wyeth of defined events and royalties on product sales. We
recognized research revenue from Wyeth from January 1, 2006 to October 2009. We
recognized revenue related to the upfront license payment we received from Wyeth
using the proportionate performance method since we reasonably estimated the
level of effort required to complete our performance obligations based upon the
most current budget approved by both Wyeth and us. Such performance obligations
were provided by us on a best-efforts basis. Full-time equivalents were used as
the measure of performance. Significant judgment is required in determining the
nature and assignment of tasks to be accomplished by each of the parties and the
level of effort required for us to complete our performance obligations under
the arrangement. The nature and assignment of tasks performed by each party
involved the preparation, discussion and approval by the parties of a
development plan and budget.
The
amount of the Wyeth upfront license payment that we recognized as revenue for
each fiscal quarter prior to the third quarter of 2007 was based upon several
revised and approved budgets, although the revisions to those budgets did not
materially affect the amount of revenue recognized in those periods. During the
third quarter of 2007, the estimate of our total remaining effort to complete
our development obligations was increased significantly based upon a revised
development budget approved by both us and Wyeth. As a result, the period over
which our obligations were to be performed, and over which the upfront payment
was to be amortized, was extended from the end of 2008 to the end of 2009. The
Transition Agreement between Wyeth and us shortened the obligation period from
the end of 2009 to October 2009 and resulted in the recognition, during the
fourth quarter of 2009, of the remaining $5.2 million unamortized upfront
payment balance at September 30, 2009.
In
relation to the Wyeth collaboration, we assessed the nature of our involvement
with the JSC, JDC and JCC. Our involvement in the first two such committees was
one of several obligations to develop the subcutaneous and intravenous
formulations of RELISTOR through regulatory approval in the U.S. We combined the
committee obligations with the other development obligations and accounted for
these obligations during the development phase as a single unit of accounting.
After the period during which we have developmental responsibilities, however,
we assessed the nature of our involvement with the committees as a right, rather
than an obligation. Our assessment was based upon the fact that we negotiated to
be on these committees as an accommodation for our granting of the license for
RELISTOR to Wyeth. Further, Wyeth had been granted by us an exclusive worldwide
license, even as to us, to develop and commercialize RELISTOR and we had
assigned the agreements for the manufacture of RELISTOR by third parties to
Wyeth. We were responsible for developing the subcutaneous and intravenous
formulations in the U.S. until they receive regulatory approval, while Wyeth was
responsible for these formulations outside the U.S. other than Japan. Wyeth was
also responsible for the development of the oral formulation worldwide excluding
Japan. We transferred to Wyeth all existing supply agreements with third parties
for RELISTOR and sublicensed intellectual property rights to permit Wyeth to
manufacture RELISTOR, during the development and commercialization phases of the
Wyeth Collaboration Agreement, in both bulk and finished form for all products
worldwide. We had no further manufacturing obligations under the 2005
Collaboration. We transferred to Wyeth all know-how, as defined, related to
RELISTOR. Based upon our research and development programs, such period will
cease upon completion of our development obligations under the 2005 Wyeth
collaboration agreement.
Following
regulatory approval of the subcutaneous and intravenous formulations of
RELISTOR, Wyeth was required to continue to develop the oral formulation and to
commercialize all formulations as provided in the Wyeth Collaboration Agreement,
for which it was capable and responsible. We expected at the beginning of the
agreement, that the activities of these committees for the period were to be
focused on Wyeth’s development and commercialization obligations. As discussed
in Note 1, we and Wyeth terminated our collaboration in October 2009, as a
result of which we regained all worldwide rights to RELISTOR and our out-license
to Ono, with respect to Japan, is unaffected by the termination of the Wyeth
collaboration.
From
January 2006 to October 2009, costs for the development of RELISTOR incurred by
Wyeth or us were paid by Wyeth. Wyeth had the right once annually to engage an
independent public accounting firm to audit expenses for which we had been
reimbursed during the prior three years. If the accounting firm concluded that
any such expenses had been understated or overstated, a reconciliation was
performed. From January 2006 to October 2009, we recognized as research and
development revenue from collaborator, amounts received from Wyeth for
reimbursement of our development expenses for RELISTOR as incurred under the
development plan agreed to between us and Wyeth. In addition to the upfront
payment and reimbursement of our development costs, Wyeth made milestone
payments to us upon achievement of specific milestones (development related
milestones for clinical and regulatory events). Upon achievement of defined
substantive development milestones by us for the subcutaneous and intravenous
formulations, the milestone payments were recognized as
revenue.
F-11
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
During
May 2007, April 2008 and July 2008, we earned $9.0 million (two milestone
payments), $15.0 million and $10.0 million, respectively, upon achievement of
non-refundable milestones anticipated in the Wyeth collaboration; the first for
the submission and acceptance for review of an NDA for a subcutaneous
formulation of RELISTOR with the FDA and a comparable submission in the European
Union, the second for the FDA approval of subcutaneous RELISTOR and the third
for the European approval of subcutaneous RELISTOR. We considered those
milestones to be substantive based on the significant degree of risk at the
inception of the collaboration related to the conduct and successful completion
of clinical trials and, therefore, of not achieving the milestones; the amount
of the payment received relative to the significant costs incurred since
inception of the Wyeth collaboration and amount of effort expended or the risk
associated with the achievement of these milestones; and the passage of 17, 28
and 31 months, respectively, from inception of the collaboration to the
achievement of those milestones. Therefore, we recognized the milestone payments
as revenue in the respective periods in which the milestones were earned. As of
December 31, 2008, relative to the $60.0 million upfront license payment
received from Wyeth, we recorded $14.6 million as deferred revenue – current,
which is recognized as revenue over the period of our development obligations
relating to RELISTOR. In addition, at December 31, 2008, we recorded $1.6
million as deferred revenue - current, related to reimbursements from Wyeth for
development costs.
In
addition, during years ended December 31, 2009 and 2008, we earned royalties of
$1,853 and $665, respectively, based on the net sales of subcutaneous RELISTOR,
and we recognized $2,372 and $146, respectively, of royalty income. As of
December 31, 2008, we recorded a cumulative total of $519 as deferred revenue –
current, which was being recognized as royalty income over the period of our
development obligations relating to RELISTOR. The remaining deferred royalty
revenue balance of $807, as of September 30, 2009, was recognized as royalty
income during the fourth quarter of 2009, the period in which our development
obligations under the Wyeth Collaboration Agreement terminated. We incurred $185
and $67, respectively, of royalty costs and recognized $237 and $15,
respectively, of royalty expenses during the years ended December 31, 2009 and
2008. As of December 31, 2008, we recorded a cumulative total of $52 of deferred
royalty costs from the royalty costs incurred during the last three quarters of
2008, which were being recognized as royalty expense over the period of our
development obligations relating to RELISTOR. The remaining deferred royalty
charges balance of $81, as of September 30, 2009, was recognized as royalty
expense during the fourth quarter 2009, the period in which our development
obligations relating to RELISTOR terminated.
Ono
Agreement – October 2008
Ono is
responsible for developing and commercializing subcutaneous RELISTOR in Japan,
including conducting the clinical development necessary to support regulatory
marketing approval. Ono will own the subcutaneous filings and approvals relating
to RELISTOR in Japan. In addition to the $15.0 million upfront payment from Ono,
we are entitled to receive up to an additional $20.0 million, payable upon
achievement by Ono of its development milestones. Ono is also obligated to pay
to us royalties and commercialization milestones on sales by Ono of subcutaneous
RELISTOR in Japan. Ono has the option to acquire from us the rights to develop
and commercialize in Japan other formulations of RELISTOR, including intravenous
and oral forms, on terms to be negotiated separately. Ono may request us to
perform activities related to its development and commercialization
responsibilities beyond our participation in joint committees and specified
technology transfer-related tasks which will be at its expense, and payable to
us for the services it requests, at the time we perform services for them.
Revenue earned from activities we perform for Ono is recorded in research and
development revenue.
We
recognized the upfront payment of $15.0 million, which we received from Ono in
November 2008, as research and development revenue during the first quarter of
2009, upon satisfaction of our performance obligations.
Research
and Development Expenses
Research
and development expenses include costs directly attributable to the conduct of
research and development programs, including the cost of salaries, payroll
taxes, employee benefits, materials, supplies, maintenance of research
equipment, costs related to research collaboration and licensing agreements, the
purchase of in-process research and development, the cost of services provided
by outside contractors, including services related to the our clinical trials,
clinical trial expenses, the full cost of manufacturing drug for use in
research, pre-clinical development and clinical trials. All costs associated
with research and development are expensed as incurred.
F-12
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Clinical
trial expenses, which are included in research and development expenses,
represent obligations resulting from our contracts with various clinical
investigators and clinical research organizations in connection with conducting
clinical trials for our product candidates. Such costs are expensed as incurred,
and are based on the expected total number of patients in the trial, the rate at
which the patients enter the trial and the period over which clinical
investigators or clinical research organizations are expected to provide
services. At each period end, we evaluate the accrued expense balance related to
these activities based upon information received from the suppliers and
estimated progress towards completion of the research or development objectives
to ensure that the balance is reasonably stated. Such estimates are subject to
change as additional information becomes available.
Use
of Estimates
Significant
estimates include useful lives of fixed assets, the periods over which certain
revenues and expenses will be recognized, including research and development
revenue recognized from non-refundable up-front licensing payments and expense
recognition of certain clinical trial costs which are included in research and
development expenses, the amount of non-cash compensation costs related to
share-based payments to employees and non-employees and the periods over which
those costs are expensed and the likelihood of realization of deferred tax
assets.
Patents
As a
result of research and development efforts conducted by us, we have applied, or
are applying, for a number of patents to protect proprietary inventions. All
costs associated with patents are expensed as incurred.
Net
Loss Per Share
We
prepare our earnings per share (“EPS”) data in accordance with ASC 260 Earnings
Per Share. Basic net loss per share amounts have been computed by dividing net
loss by the weighted-average number of shares of common shares outstanding
during the period. In June 2008, the FASB updated ASC 260 Earnings Per Share by
requiring entities, when calculating EPS, to allocate earnings to unvested and
contingently issuable share-based payment awards that have non-forfeitable
rights to dividends or dividend equivalents when calculating EPS and also
present both basic EPS and diluted EPS pursuant to the two-class method. The
update to ASC 260 Earnings Per Share was effective January 1, 2009 and required
retrospective application. We adopted this update on January 1, 2009 and the
adoption had no material impact on basic and diluted earnings per share for the
years ended December 31, 2009, 2008 and 2007. Potential common shares, amounts
of unrecognized compensation expense and windfall tax benefits have been
excluded from diluted net loss per share since they would be
anti-dilutive.
Concentrations
of Credit Risk
Financial
instruments that potentially subject Progenics to concentrations of credit risk
consist of cash, cash equivalents, marketable securities and receivables from
Wyeth, Ono or the NIH. We invest our excess cash in money market funds,
corporate debt securities and federal agency issues. We have established
guidelines that relate to credit quality, diversification and maturity and that
limit exposure to any one issue of securities. We hold no collateral for these
financial instruments.
Cash
and Cash Equivalents
We
consider all highly liquid investments which have maturities of three months or
less, when acquired, to be cash equivalents. The carrying amount reported in the
balance sheet for cash and cash equivalents approximates its fair value. Cash
and cash equivalents subject us to concentrations of credit risk. At December
31, 2009 and 2008, we have invested approximately $84,169 and $43,859,
respectively, in cash equivalents in the form of money market funds with two
major investment companies and held approximately $6,734 and $12,327,
respectively, in a single commercial bank. Restricted cash of $200 and $520 at
December 31, 2009 and 2008, respectively, consists of collateral for a letter of
credit securing lease obligations and credit card balances and the carrying
value of restricted cash approximates its fair value.
F-13
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Marketable
Securities
In
accordance with ASC 320 Investments - Debt and Equity Securities, investments
are classified as available-for-sale. Available-for-sale securities are carried
at fair value, with the unrealized gains and losses reported in comprehensive
income (loss). The amortized cost of debt securities is adjusted for
amortization of premiums and accretion of discounts to maturity. Such
amortization is included in interest income or expense. Realized gains and
losses and declines in value judged to be other-than-temporary, if any, on
available-for-sale securities are included in other income or expense. In
computing realized gains and losses, we compute the cost of its investments on a
specific identification basis. Such cost includes the direct costs to acquire
the securities, adjusted for the amortization of any discount or premium. The
fair value of marketable securities has been estimated based on a three-level
hierarchy for fair value measurements. Interest and dividends on securities
classified as available-for-sale are included in interest income (see Note
3).
At
December 31, 2009 and 2008, our investment in marketable securities in the
current and long term assets sections of the consolidated balance sheets
included $3.8 million and $4.1 million, respectively, of auction rate
securities. Valuation of securities is subject to uncertainties that are
difficult to predict, such as changes to credit ratings of the securities and/or
the underlying assets supporting them, default rates applicable to the
underlying assets, underlying collateral value, discount rates, counterparty
risk, ongoing strength and quality of market credit and liquidity and general
economic and market conditions. The valuation of the auction rate securities we
hold is based on an internal analysis of timing of expected future successful
auctions, collateralization of underlying assets of the security and credit
quality of the security. As a result of the estimated fair value, we
re-evaluated the valuation of these securities as of December 31, 2009 and the
temporary impairment amount decreased $8.0 thousand from $316.0 thousand at
December 31, 2008 to $308.0 thousand. All income generated from these current
investments was recorded as interest income (see Note 3).
Fair
Value Measurements
We
adopted ASC 820 Fair Value Measurements and Disclosures effective January 1,
2008 and 2009, for financial assets and financial liabilities and nonfinancial
assets and nonfinancial liabilities, respectively. Fair value measurements and
disclosures define fair value as the price that would be received in selling an
asset or paid in transferring a liability (i.e., the “exit price”) in an
orderly transaction between market participants at the measurement date, and
establish a framework to make the measurement of fair value more consistent and
comparable. The adoption of fair value measurements and disclosures did not have
a material impact on our financial position or results of
operations.
We use a
three-level hierarchy for fair value measurements that distinguishes between
market participant assumptions developed from market data obtained from sources
independent of the reporting entity (“observable inputs”) and the reporting
entity’s own assumptions about market participant assumptions developed from the
best information available in the circumstances (“unobservable inputs”). The
hierarchy level assigned to each security in our available-for-sale portfolio is
based on our assessment of the transparency and reliability of the inputs used
in the valuation of such instrument at the measurement date. The three hierarchy
levels are defined as follows:
|
|
· Level
1 - Valuations based on unadjusted quoted market prices in active markets
for identical securities.
|
|
|
· Level
2 - Valuations based on observable inputs other than Level 1 prices, such
as quoted prices for similar assets at the measurement date, quoted prices
in markets that are not active or other inputs that are observable, either
directly or indirectly.
|
|
|
· Level
3 - Valuations based on inputs that are unobservable and significant to
the overall fair value measurement, and involve management
judgment.
|
Other
current assets are comprised of financial instruments and non-financial
instruments. The financial instruments primarily consist of interest and other
receivables of $77 and $2,036 at December 31, 2009 and 2008, respectively, and
we believe that carrying value is a reasonable estimate of fair value as the
receivables are expected to be settled for cash within one year. Other assets of
$1,667 at December 31, 2009 represent the long term portion of amounts due to
Progenics from collaborator. We believe that carrying value approximates fair
value.
F-14
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Fixed
Assets
Leasehold
improvements, furniture and fixtures, and equipment are stated at cost.
Furniture, fixtures and equipment are depreciated on a straight-line basis over
their estimated useful lives. Leasehold improvements are amortized on a
straight-line basis over the
life of the lease or of the improvement, whichever is shorter. Costs of
construction of long-lived assets are capitalized but are not depreciated until
the assets are placed in service.
Expenditures
for maintenance and repairs which do not materially extend the useful lives of
the assets are charged to expense as incurred. The cost and accumulated
depreciation of assets retired or sold are removed from the respective accounts
and any gain or loss is recognized in operations. The estimated useful lives of
fixed assets are as follows:
|
Computer
equipment
|
3
years
|
|
Machinery
and equipment
|
5-7
years
|
|
Furniture
and fixtures
|
5
years
|
|
Leasehold
improvements
|
Earlier
of life of improvement or lease
|
Impairment
of Long-Lived Assets
We
periodically assess the recoverability of fixed assets and evaluate such assets
for impairment whenever events or changes in circumstances indicate that the
carrying amount of an asset may not be recoverable. In accordance with ASC 360
Property, Plant, and Equipment - Impairment or Disposal of Long-Lived Assets, if
indicators of impairment exist, we assess the recoverability of the affected
long-lived assets by determining whether the carrying value of such assets can
be recovered through undiscounted future operating cash flows. If the carrying
amount is not recoverable, we measure the amount of any impairment by comparing
the carrying value of the asset to the present value of the expected future cash
flows associated with the use of the asset. No impairments occurred as of
December 31, 2009, 2008 or 2007.
Income
Taxes
We
account for income taxes in accordance with the provisions of ASC 740 Income
Taxes, which requires that we recognize deferred tax liabilities and assets for
the expected future tax consequences of events that have been included in the
financial statements or tax returns. Under this method, deferred tax assets and
liabilities are determined on the basis of the difference between the tax basis
of assets and liabilities and their respective financial reporting amounts
(“temporary differences”) at enacted tax rates in effect for the years in which
the temporary differences are expected to reverse. A valuation allowance is
established for deferred tax assets for which realization is
uncertain.
In
accordance with ASC 718 Compensation – Stock Compensation and ASC 505 Equity, we
have made a policy decision related to intra-period tax allocation, to account
for utilization of windfall tax benefits based on provisions in the tax law that
identify the sequence in which amounts of tax benefits are used for tax purposes
(i.e., tax law
ordering).
Uncertain
tax positions are accounted for in accordance with ASC 740 Income Taxes, which
prescribes a comprehensive model for the manner in which a company should
recognize, measure, present and disclose in its financial statements all
material uncertain tax positions that we have taken or expect to take on a tax
return. ASC 740 applies to income taxes and is not intended to be applied by
analogy to other taxes, such as sales taxes, value-add taxes, or property taxes.
We review our nexus in various tax jurisdictions and our tax positions related
to all open tax years for events that could change the status of our ASC 740
liability, if any, or require an additional liability to be recorded. Such
events may be the resolution of issues raised by a taxing authority, expiration
of the statute of limitations for a prior open tax year or new transactions for
which a tax position may be deemed to be uncertain. Those positions, for which
management’s assessment is that there is more than a 50 percent probability of
sustaining the position upon challenge by a taxing authority based upon its
technical merits, are subjected to the measurement criteria of ASC 740. We
record the largest amount of tax benefit that is greater than 50 percent likely
of being realized upon ultimate settlement with a taxing authority having full
knowledge of all relevant information. Any ASC 740 liabilities for which we
expect to make cash payments within the next twelve months are classified as
“short term.” In the event that we conclude that we are subject to interest
and/or penalties arising from uncertain tax positions, we will record interest
and penalties as a component of income taxes (see Note 11).
F-15
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Risks
and Uncertainties
We have
to date generated only modest amounts of product and royalty revenue, and except
for RELISTOR have no products approved by the FDA for marketing. There can be no
assurance that our research and development will be successfully completed, that
any products developed will obtain necessary marketing approval by regulatory
authorities or that any approved products will be commercially viable. In
addition, we operate in an environment of rapid change in technology, and we are
dependent upon establishing satisfactory relationships with one or more
partner(s) and the continued services of our current employees, consultants and
subcontractors. We are also dependent upon Wyeth and/or Ono fulfilling their
manufacturing obligations, either on their own or through third-party suppliers.
For the years ended December 31, 2009, 2008 and 2007, the primary sources of our
revenues were Wyeth, Ono and research grant and contract revenues from the NIH.
There can be no assurance that revenues from Wyeth, Ono or from research awards
will continue. Substantially all of our accounts receivable at December 31, 2009
and 2008 were from the above-named sources.
Comprehensive
Loss
Comprehensive
loss represents the change in net assets of a business enterprise during a
period from transactions and other events and circumstances from non-owner
sources. Our comprehensive loss includes net loss adjusted for the change in net
unrealized gain or loss on marketable securities. The disclosures required by
ASC 220 Comprehensive Income for the years ended December 31, 2009, 2008 and
2007 have been included in the Consolidated Statements of Stockholders’ Equity
and Comprehensive Loss. There was no income tax expense/benefit allocated to any
component of Other Comprehensive Loss (see Note 11).
Impact
of Recently Issued Accounting Standards
In
October 2009, the FASB issued ASU 2009-13 to address the accounting for
multiple-deliverable arrangements. In an arrangement with multiple deliverables,
the delivered items shall be considered a separate unit of accounting if both
(i) the delivered items have value to a collaborator on a stand-alone basis, in
that, the collaborator could resell the delivered items on a stand-alone basis,
and (ii) the arrangement includes a general right of return relative to the
delivered item, delivery or performance of the undelivered item or items is
considered probable and substantially in our control. This ASU will be effective
prospectively for revenue arrangements entered into or materially modified in
fiscal years beginning on or after June 15, 2010. Early adoption is permitted.
We are currently evaluating the impact this ASU will have on our financial
statements.
3.
Fair Value Measurements and Marketable Securities
Our
available-for-sale investment portfolio consists of marketable securities,
including corporate debt securities, securities of government-sponsored entities
and auction rate securities, and is recorded at fair value in the accompanying
Consolidated Balance Sheets in accordance with ASC 320 Investments – Debt and
Equity Securities. The change in the fair value of these marketable securities
is recorded as a component of other comprehensive loss (see Note
2).
Marketable
securities consisted of the following:
|
December
31,
2009
|
December
31,
2008
|
|||||||
|
Short-term
|
||||||||
|
Corporate
debt securities
|
$ | 1,501 | $ | 63,127 | ||||
|
Total
short-term marketable securities
|
1,501 | 63,127 | ||||||
|
Long-term
|
||||||||
|
Corporate
debt securities and securities of government-sponsored
entities
|
- | 18,002 | ||||||
|
Auction
rate securities
|
3,792 | 4,059 | ||||||
|
Total
long-term marketable securities
|
3,792 | 22,061 | ||||||
|
Total
marketable securities
|
$ | 5,293 | $ | 85,188 | ||||
The
following table presents our available-for-sale investments measured at fair
value on a recurring basis as of December 31, 2009 and 2008, classified by
valuation hierarchy (as previously discussed):
|
Fair
Value Measurements at Reporting Date Using
|
||||||||||||||||
|
Description
|
Balance
at
December
31, 2009
|
Quoted
Prices
in
Active
Markets
for Identical Assets
(Level
1)
|
Significant
Other
Observable
Inputs
(Level
2)
|
Significant
Unobservable Inputs
(Level
3)
|
||||||||||||
|
Money
market funds
|
$ | 84,169 | $ | 84,169 | $ | - | $ | - | ||||||||
|
Corporate
debt securities
|
1,501 | - | 1,501 | - | ||||||||||||
|
Auction
rate securities
|
3,792 | - | - | 3,792 | ||||||||||||
|
Total
|
$ | 89,462 | $ | 84,169 | $ | 1,501 | $ | 3,792 | ||||||||
|
Fair
Value Measurements at Reporting Date Using
|
||||||||||||
|
Description
|
Balance
at
December
31,
2008
|
Quoted
Prices
in
Active
Markets
for Identical Assets
(Level
1)
|
Significant
Other
Observable
Inputs
(Level
2)
|
Significant
Unobservable
Inputs
(Level
3)
|
||||||||
|
Money
market funds
|
$
|
43,859
|
$
|
43,859
|
$
|
-
|
$
|
-
|
||||
|
Corporate
debt securities and securities of government-sponsored
entities
|
81,129
|
-
|
81,129
|
-
|
||||||||
|
Auction
rate securities
|
4,059
|
-
|
-
|
4,059
|
||||||||
|
Total
|
$
|
129,047
|
$
|
43,859
|
$
|
81,129
|
$
|
4,059
|
||||
At
December 31, 2009, we hold $3.8 million (4.2% of total assets measured at fair
value) in auction rate securities which are classified as Level 3. The fair
value of these securities includes $2.9 million of securities collateralized by
student loan obligations subsidized by the U.S. government and $0.9 million of
investment company perpetual preferred stock. Auction rate securities are
collateralized long-term instruments that were intended to provide liquidity
through a Dutch auction process that resets the applicable interest rate at
pre-determined intervals, typically every 7 to 35 days. Beginning in the first
quarter of 2008, auctions failed for certain of our auction rate securities
because sell orders exceeded buy orders, and we were unable to dispose of those
securities at auction. We will not realize cash in respect of the principal
amount of these securities until a successful auction occurs, the issuer calls
or restructures the security, the security reaches any scheduled maturity and is
paid (which is inapplicable to the perpetual preferred mentioned above) or a
buyer outside the auction process emerges. As of December 31, 2009, we have
received all scheduled interest payments on these securities, which, in the
event of auction failure, are reset according to the contractual terms in the
governing instruments.
F-16
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
The
valuation of auction rate securities we hold is based on Level 3 unobservable
inputs which consist of our internal analysis of (i) timing of expected future
successful auctions, (ii) collateralization of underlying assets of the security
and (iii) credit quality of the security. We re-evaluated the valuation of these
securities as of December 31, 2009 and the temporary impairment amount decreased
$8.0 from $316.0 at December 31, 2008, to $308.0, which is reflected as a part
of other comprehensive loss on our accompanying Consolidated Balance Sheets.
These securities are held “available-for-sale” and the unrealized loss is
included in other comprehensive loss. Due to the uncertainty related to the
liquidity in the auction rate security market and therefore when individual
positions may be liquidated, we have classified these auction rate securities as
long-term assets on our accompanying Consolidated Balance Sheets.
We
continue to monitor markets for our investments and consider the impact, if any,
of market conditions on the fair market value of our investments. We do not
believe the carrying values of our investments are other than temporarily
impaired and therefore expect the positions will eventually be liquidated
without significant loss.
For those
of our financial instruments with significant Level 3 inputs (all of which are
auction rate securities), the following tables summarize the activities for the
years ended December 31, 2009 and 2008:
|
Fair
Value Measurements Using Significant
Unobservable
Inputs
(Level
3)
|
||||||||
|
Description
|
For
the Year Ended
December
31, 2009
|
For
the Year Ended
December
31, 2008
|
||||||
|
Balance
at beginning of period
|
$ | 4,059 | $ | - | ||||
|
Transfers
into Level 3
|
- | 8,150 | ||||||
|
Total
realized/unrealized gains (losses) (1)
|
||||||||
|
Included
in net loss
|
- | - | ||||||
|
Included
in comprehensive income (loss)
|
8 | (316 | ) | |||||
|
Settlements
|
(275 | ) | (3,775 | ) | ||||
|
Balance
at end of period
|
$ | 3,792 | $ | 4,059 | ||||
|
(1) Total
amount of unrealized gains (losses) for the period included in other
comprehensive loss attributable to the change in fair market value of
related assets still held at the reporting date
|
$ | - | $ | (316 | ) | |||
The
following tables summarize the amortized cost basis, the aggregate fair value
and gross unrealized holding gains and losses at December 31, 2009 and
2008:
|
Amortized
|
Fair
|
Unrealized
Holding
|
||||||||||||||||||
|
Cost
Basis
|
Value
|
Gains
|
(Losses)
|
Net
|
||||||||||||||||
|
2009:
|
||||||||||||||||||||
|
Maturities
less than one year:
|
||||||||||||||||||||
|
Corporate
debt securities
|
$ | 1,501 | $ | 1,502 | $ | 1 | $ | - | $ | 1 | ||||||||||
|
Maturities
greater than ten years:
|
||||||||||||||||||||
|
Auction
rate securities
|
1,000 | 940 | - | (60 | ) | (60 | ) | |||||||||||||
|
Investments
without stated maturity dates:
|
||||||||||||||||||||
|
Auction rate securities
|
3,100 | 2,852 | - | (248 | ) | (248 | ) | |||||||||||||
| $ | 5,601 | $ | 5,294 | $ | 1 | $ | (308 | ) | $ | (307 | ) | |||||||||
|
Amortized
|
Fair
|
Unrealized
Holding
|
||||||||||||||||||
|
Cost
Basis
|
Value
|
Gains
|
(Losses)
|
Net
|
||||||||||||||||
|
2008:
|
||||||||||||||||||||
|
Maturities
less than one year:
|
||||||||||||||||||||
|
Corporate debt securities
|
$ | 63,982 | $ | 63,127 | $ | 114 | $ | (969 | ) | $ | (855 | ) | ||||||||
|
Maturities
between one and five years:
|
||||||||||||||||||||
|
Corporate debt securities
|
17,129 | 16,995 | 71 | (205 | ) | (134 | ) | |||||||||||||
|
Government-sponsored entities
|
999 | 1,007 | 8 | - | 8 | |||||||||||||||
|
Maturities
greater than ten years:
|
||||||||||||||||||||
|
Auction rate securities
|
3,200 | 2,944 | - | (256 | ) | (256 | ) | |||||||||||||
|
Investments
without stated maturity dates:
|
||||||||||||||||||||
|
Auction rate securities
|
1,175 | 1,115 | - | (60 | ) | (60 | ) | |||||||||||||
| $ | 86,485 | $ | 85,188 | $ | 193 | $ | (1,490 | ) | $ | (1,297 | ) | |||||||||
Progenics’
computes the cost of its investments on a specific identification basis. Such
cost includes the direct costs to acquire the securities, adjusted for the
amortization of any discount or premium.
F-17
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
The
following table shows the gross unrealized losses and fair value of our
marketable securities with unrealized losses that are not deemed to be
other-than-temporarily impaired, aggregated by investment category and length of
time that individual securities have been in a continuous unrealized loss
position, at December 31, 2009 and 2008.
At
December 31, 2009:
|
Less
than 12 Months
|
12
Months or Greater
|
Total
|
||||||||||||||||||||||
|
Description of Securities
|
Fair
Value
|
Unrealized
Losses
|
Fair
Value
|
Unrealized
Losses
|
Fair
Value
|
Unrealized
Losses
|
||||||||||||||||||
|
Auction
rate securities
|
$ | - | $ | - | $ | 3,792 | $ | (308 | ) | $ | 3,792 | $ | (308 | ) | ||||||||||
|
Total
|
$ | - | $ | - | $ | 3,792 | $ | (308 | ) | $ | 3,792 | $ | (308 | ) | ||||||||||
At
December 31, 2008:
|
Less
than 12 Months
|
12
Months or Greater
|
Total
|
||||||||||||||||||||||
|
Description of Securities
|
Fair
Value
|
Unrealized
Losses
|
Fair
Value
|
Unrealized
Losses
|
Fair
Value
|
Unrealized
Losses
|
||||||||||||||||||
|
Corporate
debt securities
|
$ | 57,567 | $ | (1,174 | ) | $ | - | $ | - | $ | 57,567 | $ | (1,174 | ) | ||||||||||
|
Auction
rate securities
|
4,059 | (316 | ) | 4,059 | (316 | ) | ||||||||||||||||||
|
Total
|
$ | 61,626 | $ | (1,490 | ) | $ | - | $ | - | $ | 61,626 | $ | (1,490 | ) | ||||||||||
Other-than-temporary impairment
analysis on corporate debt securities. At December 31, 2008, we owned 34
securities maturing in less than one year, with a gross unrealized loss position
of $969 ($46,028 of the total fair value) and there were 9 securities in the
portfolio maturing between one and two years, with a gross unrealized loss
position of $205 ($11,539 of the total fair value). The severity of the
unrealized losses for the securities in an unrealized loss position at December
31, 2008 was between less than one percent and 17.67 percent below amortized
cost, and the weighted average duration of the unrealized losses of these
securities was 6.98 months.
We have
evaluated our individual corporate debt securities holdings for
other-than-temporary impairment and determined that the unrealized losses as of
December 31, 2008 are attributable to our purchase of corporate debt securities
which traded at a premium in early 2008, and declined in market value during
2008. Because we do not intend to sell these securities, and believe it is not
more likely than not that we would be required to sell these securities before
recovery of principal, we do not consider these securities to be
other-than-temporarily impaired at December 31, 2008.
Other-than-temporary impairment
analysis on auction rate securities. The unrealized losses in our auction
rate securities investments were the result of an internal analysis of timing of
expected future successful auctions, collateralization of underlying assets of
the security and credit quality of the security. At December 31, 2009 and 2008,
there were two and three securities with a gross unrealized loss position of
$308 and $316 ($3,792 and $4,059 of the total fair value),
respectively.
The
severity of the unrealized losses for auction rate securities at December 31,
2009 and 2008 was between 6 percent and 8 percent below amortized cost, and the
weighted average duration of the unrealized losses for these securities was 22
and 9.25 months, respectively.
We have
evaluated our individual auction rate securities holdings for
other-than-temporary impairment and determined that the unrealized losses as of
December 31, 2009 and 2008 are attributable to uncertainty in the liquidity of
the auction rate security market. Because we do not intend to sell these
securities, and believe it is not more likely than not that we would be required
to sell these securities before recovery of principal, we do not consider these
securities to be other-than-temporarily impaired at December 31, 2009 and
2008.
F-18
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
4.
Accounts Receivable
Our
accounts receivable represent amounts due to Progenics from research from
collaborator, royalties, research grants and contract and the sales of research
reagents. These amounts are considered to be short-term as they are expected to
be collected within one year and we believe carrying value approximates fair
value.
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
National
Institutes of Health
|
$ | 210 | $ | 1,107 | ||||
|
Royalties
|
589 | 229 | ||||||
|
Research
and development from collaborator
|
6,667 | - | ||||||
|
Other
|
56 | 1 | ||||||
|
Total
|
$ | 7,522 | $ | 1,337 | ||||
5.
Fixed Assets
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Computer
equipment
|
$ | 2,443 | $ | 2,335 | ||||
|
Machinery
and equipment
|
13,237 | 13,161 | ||||||
|
Furniture
and fixtures
|
740 | 750 | ||||||
|
Leasehold
improvements
|
10,662 | 10,546 | ||||||
|
Construction
in progress
|
831 | 907 | ||||||
| 27,913 | 27,699 | |||||||
|
Less,
accumulated depreciation and amortization
|
(21,353 | ) | (16,628 | ) | ||||
|
Total
|
$ | 6,560 | $ | 11,071 | ||||
At
December 31, 2009, $5.9 million and $1.6 million of leasehold improvements were
being amortized over periods of 0.3 – 5.8 years and 2.0 – 4.0 years,
respectively, under leases with terms through December 31, 2009 and June 30,
2010, respectively. At December 31, 2008, $5.8 million, $0.9 million and $0.7
million of leasehold improvements were being amortized over periods of 1.0 – 5.8
years, 4.0 – 4.7 years and 8.5 years, respectively, under leases with terms
through December 31, 2009, June 29, 2012 and December 31, 2014,
respectively.
6.
Accounts Payable and Accrued Expenses
The
carrying value of our accounts payable and accrued expenses approximates fair
value, as it represents amounts due to vendors and employees, which will be
satisfied within one year.
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Accounts
payable
|
$ | 596 | $ | 899 | ||||
|
Accrued
consulting and clinical trial costs
|
2,663 | 3,556 | ||||||
|
Accrued
payroll and related costs
|
1,321 | 1,093 | ||||||
|
Legal
and professional fees
|
1,070 | 925 | ||||||
|
Other
|
186 | 23 | ||||||
|
Total
|
$ | 5,836 | $ | 6,496 | ||||
7.
Stockholders’ Equity
We are
authorized to issue 40,000 shares of common stock, par value $.0013 (“Common
Stock”), and 20,000 shares of preferred stock, par value $.001. The Board of
Directors has the authority to issue common and preferred shares, in series,
with rights and privileges as determined by the Board of
Directors.
F-19
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
On
September 25, 2007, we completed a public offering of 2.6 million shares of our
Common Stock, pursuant to a shelf registration statement that had been filed
with the SEC in 2006, which had registered 4.0 million shares of our Common
Stock. We received proceeds of $57.3 million, or $22.04 per share, which was net
of underwriting discounts and commissions of approximately $2.9 million, and
paid approximately $0.2 million in other offering expenses.
On April
24, 2008, our Board of Directors approved a share repurchase program to acquire
up to $15.0 million of our outstanding common shares. During the year ended
December 31, 2008, we have repurchased 200,000 of our outstanding common shares
for a total of $2.7 million. Purchases may be discontinued at any time. We did
not repurchase any common shares during the year ended December 31, 2009. We
have $12.3 million remaining available for purchases under the
program.
8.
Commitments and Contingencies
a.
Operating Leases
As of
December 31, 2009, we leased a total of 131,500 square feet of office and
laboratory space, under five lease agreements, the majority of which expired
December 31, 2009, while the remaining leases were to expire in either 2012 or
2014. In October 2009, we renewed to December 31, 2020, our lease agreement to
lease a total of 149,300 square feet of office and laboratory space in six
locations in Tarrytown, New York.
Rental
payments are recognized as rent expense on a straight-line basis over the term
of the lease. In addition to rents due under these agreements, we are obligated
to pay additional facilities charges, including utilities, taxes and operating
expenses. We also lease certain office equipment under non-cancelable operating
leases, which expire at various times through August 2010.
As of
December 31, 2009, future minimum annual payments under all operating lease
agreements are as follows:
|
Years
ending December 31,
|
Minimum
Annual
Payments
|
|||
|
2010
|
$ | 3,682 | ||
|
2011
|
3,777 | |||
|
2012
|
3,872 | |||
|
2013
|
3,968 | |||
|
2014
|
4,067 | |||
|
Thereafter
|
26,632 | |||
|
Total
|
$ | 45,998 | ||
Rental
expense totaled approximately $2,773, $2,971 and $2,415 for the years ended
December 31, 2009, 2008 and 2007, respectively. For the years ended December 31,
2009 and 2008, amounts paid exceeded rent expense by $154 and $93, respectively,
due to the recognition of escalation clauses and lease incentives. For the year
ended December 31, 2007, we recognized rent expense in excess of amounts paid of
$17, due to the recognition of escalation clauses and lease incentives.
Additional facility charges, including utilities, taxes and operating expenses,
for the years ended December 31, 2009, 2008 and 2007 were approximately $3,060,
$3,533 and $2,974, respectively.
b.
Licensing, Service and Supply Agreements
Progenics
has entered into intellectual property-based license and service agreements in
connection with their product development programs. Progenics has recognized
milestone, license and sublicense fees and supply costs, which are included in
research and development expenses, totaling approximately $788, $2,422 and $988
for the years ended December 31, 2009, 2008 and 2007,
respectively.
F-20
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
|
Agreement
|
Paid
from
inception
to December 31,
2009
|
Future
Commitments
|
Terms
|
|||
|
Progenics’ agreements
with:
|
||||||
|
Facet
Biotech Corporation (formerly Protein Design Labs, Inc.)
|
$
5,350
|
$
2,600 (1)
|
Annual
maintenance, milestone and royalty payments for the humanized murine
monoclonal antibody developed by us.
|
|||
|
University
of Chicago
|
790
|
410
|
Milestone
and royalty payments for rights to develop and commercialize
methylnaltrexone.
|
|||
|
Sloan-Kettering
Institute for Cancer Research
|
1,000
|
-
|
Terminated
in 2008.
|
|||
|
Aquila
Biopharmaceuticals, Inc.
|
769
|
-
|
Terminated
in 2008.
|
|||
|
Columbia
University
|
$1,197
|
-
(1)
|
Terminated
and amended in 2009, under which we retain rights to certain technology
for sales of reagents and other purposes.
|
|||
|
PSMA LLC’s agreements
with:
|
||||||
|
Amgen
Fremont, Inc. (formerly Abgenix)
|
850
|
6,250
(1)
|
Annual
maintenance and milestone payments to use XenoMouse® technology for
generating fully human antibodies to PSMA LLC’s PSMA
antigen.
|
|||
|
AlphaVax
Human Vaccines
|
2,036
|
5,300
(1)
|
Annual
maintenance and milestone payments to use AlphaVax Replicon Vector system
to create a therapeutic cancer vaccine incorporating PSMA LLC’s
proprietary PSMA antigen.
|
|||
|
Seattle
Genetics, Inc.
|
3,600
|
14,000
(1)
|
Milestone
and periodic maintenance payments to use ADC technology to link
chemotherapeutic agents to monoclonal antibodies that target prostate
specific membrane antigen. ADC technology is based in part on technology
licensed by SGI from third parties.
|
|||
|
Cornell
Research Foundation
|
110
|
915
(1)
|
Annual
minimum royalty and milestone payments.
|
|||
|
Former
member of PSMA LLC
|
128
|
52,197
|
Milestone
and royalty payments to use technology related to
PSMA.
|
|||
(1) Royalties are estimable
upon sales of products under the license.
As part
of our research and development efforts, we enter into consulting agreements
with external scientific specialists (“Scientists”). These agreements contain
various terms and provisions, including fees to be paid by us and royalties, in
the event of future sales, and milestone payments, upon achievement of defined
events, payable by us. Certain Scientists are members of the Progenics’
Scientific Advisory Board (the “SAB Members”), including Stephen P. Goff, Ph.D.
and David A. Scheinberg, M.D., Ph.D., both of whom are also members of our Board
of Directors. Some Scientists have purchased our Common Stock or received stock
options which are subject to vesting provisions. We have recognized expenses
with regard to the consulting agreements of the SAB Members totaling
approximately $220, $358 and $1,092 for the years ended December 31, 2009, 2008
and 2007, respectively. Those expenses include the fair value of stock options
granted during 2009, 2008 and 2007, which were fully vested at grant date, of
approximately $83, $217 and $691, respectively. For the year ended December 31,
2007, those expenses include a portion of restricted stock, granted in 2007,
that vested in 2007, of approximately $127. Such amounts of fair value are
included in research and development compensation expense for each year
presented (see Note 9).
F-21
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
9.
Share-Based Payment Arrangements
Our
share-based compensation to employees includes non-qualified stock options,
restricted stock and shares issued under our Purchase Plans, which are
compensatory under ASC 718 Compensation – Stock Compensation. We account for
share-based compensation to non-employees, including non-qualified stock options
and restricted stock, in accordance with ASC 505 Equity.
Compensation
cost for share-based awards will be recognized in our financial statements over
the related requisite service periods; usually the vesting periods for awards
with a service condition. We have made an accounting policy decision to use the
straight-line method of attribution of compensation expense, under which the
grant date fair value of share-based awards will be recognized on a
straight-line basis over the total requisite service period for the total
award.
We have
adopted three stock incentive plans, the 1989 Non-Qualified Stock Option Plan,
the 1996 Amended Stock Incentive Plan and the 2005 Stock Incentive Plan (the
“Plans”). Under each of these Plans as amended, up to 375, 5,000 and 5,450
shares of common stock, respectively, have been reserved for the issuance of
awards to employees, consultants, directors and other individuals who render
services to Progenics (collectively, “Awardees”). The Plans contain certain
anti-dilution provisions in the event of a stock split, stock dividend or other
capital adjustment as defined. The 1989 Plan provides for the Board, or the
Compensation Committee (“Committee”) of the Board, to grant stock options to
Awardees and to determine the exercise price, vesting term and expiration date.
The 1996 Plan and the 2005 Plan provide for the Board or Committee to grant to
Awardees stock options, stock appreciation rights, restricted stock, performance
awards or phantom stock, as defined (collectively, “Awards”). The Committee is
also authorized to determine the term and vesting of each Award and the
Committee may in its discretion accelerate the vesting of an Award at any time.
Stock options granted under the Plans generally vest pro rata over four to ten
years and have terms of ten to twenty years. Restricted stock issued under the
1996 Plan or 2005 Plan usually vests annually over a four year period, unless
specified otherwise by the Committee. The exercise price of outstanding
non-qualified stock options is usually equal to the fair value of our common
stock on the date of grant. The exercise price of non-qualified stock options
granted from the 2005 Plan and incentive stock options (“ISO”) granted from the
Plans may not be lower than the fair value of our common stock on the dates of
grant. At December 31, 2009, 2008 and 2007, all outstanding stock options were
non-qualified options. The 1989 and 1996 Plans terminated in April 1994 and
October 2006, respectively, and the 2005 Plan will terminate in April 2015;
options granted before termination of the Plans will continue under the
respective Plans until exercised, cancelled or expired.
We apply
a forfeiture rate to the number of unvested awards in each reporting period in
order to estimate the number of awards that are expected to vest. Estimated
forfeiture rates are based upon historical data on vesting behavior of
employees. We adjust the total amount of compensation cost recognized for each
award, in the period in which each award vests, to reflect the actual
forfeitures related to that award. Changes in our estimated forfeiture rate will
result in changes in the rate at which compensation cost for an award is
recognized over its vesting period.
Under ASC
718 Compensation – Stock Compensation, the fair value of each non-qualified
stock option award is estimated on the date of grant using the Black-Scholes
option pricing model, which requires input assumptions noted in the following
table. Ranges of assumptions for inputs are disclosed where the value of such
assumptions varied during the related period. Historical volatilities are based
upon daily quoted market prices of our common stock on The NASDAQ Stock Market
LLC over a period equal to the expected term of the related equity instruments.
We rely only on historical volatility since it provides the most reliable
indication of future volatility. Future volatility is expected to be consistent
with historical; historical volatility is calculated using a simple average
calculation; historical data is available for the length of the option’s
expected term and a sufficient number of price observations are used
consistently. Since our stock options are not traded on a public market, we do
not use implied volatility. For the years ended December 31, 2009, 2008 and
2007, our expected term was calculated based upon historical data related to
exercise and post-termination cancellation activity. Accordingly, for grants
made to employees and officers (excluding our Chief Executive Officer) and
directors, we are using expected terms of 5.3 and 7.3 years, 5.33 and 7.30
years, and 5.25 and 7.5 years, respectively. The expected term of stock options
granted to our Chief Executive Officer and non-employee directors and
consultants are calculated separately from stock options granted to employees
and officers and the expected term was 8 years and 7.5 years or the years ended
December 31, 2009 and 2008. Expected term for options granted to non-employee
consultants was ten years, which is the contractual term of those options. We
have never paid dividends and do not expect to pay dividends in the future.
Therefore, our dividend rate is zero. The risk-free rate for periods within the
expected term of the options is based on the U.S. Treasury yield curve in effect
at the time of grant.
F-22
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
|
For
the Years Ended
December
31,
|
||||||
|
2009
|
2008
|
2007
|
||||
|
Expected
volatility
|
70%
- 91%
|
66%
- 91%
|
50%
- 89%
|
|||
|
Expected
dividends
|
zero
|
zero
|
zero
|
|||
|
Expected
term (years)
|
5.3
– 10
|
5.33
- 10
|
5.25
- 10
|
|||
|
Weighted
average expected term (years)
|
7.10
|
6.78
|
6.90
|
|||
|
Risk-free
rate
|
1.78%
- 3.22%
|
1.69%
- 3.79%
|
3.88%
- 4.93%
|
|||
A summary
of option activity under the Plans as of December 31, 2009 and changes during
the year then ended is presented below:
|
Options
|
Shares
|
Weighted
Average Exercise Price
|
Weighted
Average Remaining Contractual Term (Yr.)
|
Aggregate
Intrinsic Value
|
||||
|
Outstanding
at January 1, 2009
|
4,451
|
$18.78
|
||||||
|
Granted
|
838
|
5.43
|
||||||
|
Exercised
|
(8)
|
1.33
|
||||||
|
Forfeited
or expired
|
(372)
|
18.13
|
||||||
|
Outstanding
at December 31, 2009
|
4,909
|
$16.58
|
5.66
|
$242,653
|
||||
|
Exercisable
at December 31, 2009
|
3,455
|
$18.16
|
4.55
|
$242,653
|
The
weighted average grant-date fair value of options granted under the Plans during
the years ended December 31, 2009, 2008 and 2007 was $3.39, $10.09 and $16.18,
respectively. The total intrinsic value of options exercised during the years
ended December 31, 2009, 2008 and 2007 was $41, $969 and $3,766,
respectively.
The
options granted under the Plans, described above, include 33, 113, 38, 75, 145
and 113 non-qualified stock options granted to our Chief Executive Officer on
July 1, 2002, 2003, 2004 and 2005, on July 3, 2006 and on July 2, 2007,
respectively, which cliff vest after nine years and eleven months from the
respective grant date. The July 1, 2002, 2003 and 2005 awards have fully vested.
Vesting of a defined portion of each award will occur earlier if a defined
performance condition is achieved; more than one condition may be achieved in
any period. In accordance with ASC 718 Compensation – Stock Compensation, at the
end of each reporting period, we will estimate the probability of achievement of
each performance condition and will use those probabilities to determine the
requisite service period of each award. The requisite service period for the
award is the shortest of the explicit or implied service periods. In the case of
the executive’s options, the explicit service period is nine years and eleven
months from the respective grant dates. The implied service periods related to
the performance conditions are the estimated times for each performance
condition to be achieved. Thus, compensation expense will be recognized over the
shortest estimated time for the achievement of performance conditions for that
award (assuming that the performance conditions will be achieved before the
cliff vesting occurs). On July 1, 2008 and 2009, we granted awards (consisting
of options and restricted stock in 2008 and options in 2009) to our Chief
Executive Officer which vest on the basis of the achievement of specified
performance or market-based milestones. The options have an exercise price equal
to the closing price on our common stock on the date of grant. The awards to our
Chief Executive Officer are valued using a Monte Carlo simulation and the
expense related to these grants will be recognized over the shortest estimated
time for the achievement of the performance or market conditions. The awards
will not vest unless one of the milestones is achieved or the market condition
is met. Changes in the estimate of probability of achievement of any performance
or market condition will be reflected in compensation expense of the period of
change and future periods affected by the change.
At
December 31, 2009, the estimated requisite service periods for the 2004, 2006
and 2009 awards, described above, were 1.5, 6.25 and 2.0 years, respectively.
For the years ended December 31, 2009, 2008 and 2007, the total compensation
expense recognized for the performance-based options was $0.5 million, $1.3
million and $1.5 million, respectively.
F-23
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
A summary
of the status of our restricted stock awarded under the Plans which has not yet
vested as of December 31, 2009 and changes during the year then ended is
presented below:
|
Restricted
Stock Awards
|
Shares
|
Weighted
Average Grant-Date
Fair
Value
|
||
|
Nonvested
at January 1, 2009
|
566
|
$18.81
|
||
|
Granted
|
325
|
5.01
|
||
|
Vested
|
(306)
|
15.17
|
||
|
Forfeited
|
(37)
|
16.26
|
||
|
Nonvested
at December 31, 2009
|
548
|
$12.82
|
Our two
employee stock purchase plans (the “Purchase Plans”), the 1998 Employee Stock
Purchase Plan (the “Qualified Plan”) and the 1998 Non-Qualified Employee
Purchase Plan (the “Non-Qualified Plan”), as amended, provide for the issuance
of up to 3,400 and 1,100 shares of common stock, respectively. The Purchase
Plans provide for the grant to all employees of options to use an amount equal
to up to 25% of their quarterly compensation, as such percentage is determined
by the Board of Directors prior to the date of grant, to purchase shares of our
common stock at a price per share equal to the lesser of the fair market value
of the common stock on the date of grant or 85% of the fair market value on the
date of exercise. Options are granted automatically on the first day of each
fiscal quarter and expire six months after the date of grant. The Qualified Plan
is not available to employees owning more than five percent of the common stock
and imposes certain other quarterly limitations on the option grants. Options
under the Non-Qualified Plan are granted to the extent that option grants are
restricted under the Qualified Plan.
The fair
value of shares purchased under the Purchase Plans was estimated on the date of
grant in accordance with ASC 718 Compensation – Stock Compensation, via the same
option valuation model used for options granted under the Plans, but with the
following assumptions:
|
For
the Years Ended
December
31,
|
||||||
|
2009
|
2008
|
2007
|
||||
|
Expected
volatility
|
46%
- 100%
|
83%
- 170%
|
40%
- 46%
|
|||
|
Expected
dividends
|
zero
|
zero
|
zero
|
|||
|
Expected
term
|
6
months
|
6
months
|
6
months
|
|||
|
Risk-free
rate
|
0.00%
- 0.38%
|
0.14%
- 2.74%
|
3.91%
- 5.10%
|
|||
Purchases
of common stock under the Purchase Plans during the years ended December 31,
2009, 2008 and 2007 are summarized as follows:
|
Qualified
Plan
|
Non-Qualified
Plan
|
||||||||||
|
Shares
Purchased
|
Price
Range
|
Weighted
Average
Grant-Date Fair Value
|
Shares
Purchased
|
Price
Range
|
Weighted
Average
Grant-Date Fair Value
|
||||||
|
2009
|
872
|
$3.37
- $9.13
|
$1.49
|
189
|
$3.98
- $9.13
|
$1.58
|
|||||
|
2008
|
538
|
$4.26
- $15.32
|
$4.44
|
127
|
$6.07
- $15.32
|
$4.83
|
|||||
|
2007
|
179
|
$16.27
- $23.46
|
$3.41
|
45
|
$17.80
- $23.46
|
$3.43
|
|||||
F-24
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
The total
compensation expense of shares, granted to both employees and non-employees,
under all of our share-based payment arrangements that was recognized in
operations during the years ended December 31, 2009, 2008 and 2007
was:
|
Years
Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Recognized
as:
|
||||||||||||
|
Research
and Development
|
$ | 7,225 | $ | 7,241 | $ | 7,104 | ||||||
|
General
and Administrative
|
5,761 | 6,892 | 8,202 | |||||||||
|
Total
|
$ | 12,986 | $ | 14,133 | $ | 15,306 | ||||||
No tax
benefit was recognized related to such compensation cost because we had net
losses for the periods presented and the related deferred tax assets were fully
offset by valuation allowance. Accordingly, no amounts related to windfall tax
benefits have been reported in cash flows from operations or cash flows from
financing activities for the periods presented.
As of
December 31, 2009, there was $9.9 million, $5.1 million and $0.03 million of
total unrecognized compensation cost related to non-vested stock options under
the Plans, the non-vested shares and the Purchase Plans, respectively. Those
costs are expected to be recognized over weighted average periods of 2.31 years,
1.56 years and 0.04 years, respectively. Cash received from exercises under all
share-based payment arrangements for the year ended December 31, 2009 was $4.9
million. We issue new shares of our common stock upon share option exercise and
share purchase.
In
applying the treasury stock method for the calculation of diluted EPS, amounts
of unrecognized compensation expense and windfall tax benefits are required to
be included in the assumed proceeds in the denominator of the diluted EPS
calculation unless they are anti-dilutive. We incurred net losses for the years
ended December 31, 2009, 2008 and 2007 and, therefore, such amounts have not
been included in the calculations for those periods since they would be
anti-dilutive. As a result, basic and diluted EPS are the same for each period.
We have made an accounting policy decision to calculate windfall tax
benefits/shortfalls, for purposes of diluted EPS calculation, excluding the
impact of pro forma deferred tax assets. This policy decision will apply when we
have net income.
10.
Employee Savings Plan
The terms
of the amended and restated Progenics Pharmaceuticals 401(k) Plan (the “Amended
Plan”), among other things, allow eligible employees to participate in the
Amended Plan by electing to contribute to the Amended Plan a percentage of their
compensation to be set aside to pay their future retirement benefits. During
2009, 2008 and 2007, we matched 50%, 100% and 100%, respectively, of those
employee contributions that are equal to 5%-8% of compensation and are made by
eligible employees to the Amended Plan (the “Matching Contribution”). In
addition, we may also make a discretionary contribution each year on behalf of
all participants who are non-highly compensated employees. We made Matching
Contributions of approximately $718, $1,727 and $1,538 to the Amended Plan for
the years ended December 31, 2009, 2008 and 2007, respectively. No discretionary
contributions were made during those years.
11.
Income Taxes
We
account for income taxes using the liability method in accordance with ASC 740
Income Taxes. Deferred income taxes reflect the net tax effects of temporary
differences between the carrying amounts of assets and liabilities for financial
reporting purposes and the amounts used for income tax purposes.
There is no provision or benefit for federal or state income taxes for the years
ended December 31, 2009, 2008 or 2007. We have completed a calculation, under
Internal Revenue Code Section 382, the results of which indicate that past
ownership changes will limit utilization of net operating loss carry-forwards
(“NOL’s”) in the future. Future ownership changes may further limit the future
utilization of net operating loss and tax credit carry-forwards as defined by
the federal and state tax codes.
F-25
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Deferred
tax assets consist of the following:
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Depreciation
and amortization
|
$ | 6,831 | $ | 6,231 | ||||
|
R&E
tax credit carry-forwards
|
10,363 | 9,139 | ||||||
|
NYS
investment tax credit carry-forwards
|
1,168 | - | ||||||
|
AMT
credit carry-forwards
|
306 | 306 | ||||||
|
Net
operating loss carry-forwards
|
83,546 | 87,672 | ||||||
|
Deferred
revenue
|
- | 12,396 | ||||||
|
Capitalized
research and development expenditures
|
23,492 | - | ||||||
|
Stock
compensation
|
13,142 | 10,923 | ||||||
|
Other
items
|
2,585 | 2,402 | ||||||
| 141,433 | 129,069 | |||||||
|
Valuation
allowance
|
(141,433 | ) | (129,069 | ) | ||||
| $ | — | $ | — | |||||
We do not
recognize deferred tax assets considering our history of taxable losses and the
uncertainty regarding our ability to generate sufficient taxable income in the
future to utilize these deferred tax assets.
For the
years ended December 31, 2009 and 2008, we incurred net losses for tax purposes
and recognized a full tax valuation against deferred taxes. During 2009, we
implemented a tax planning strategy to elect and capitalize, for tax purposes
only, a total of $70.0 million of 2008 and 2009 research and development
(“R&D”) costs and amortize these costs over ten years for tax purposes. This
strategy had the effect of decreasing deferred taxes for net operating losses
and increasing deferred taxes for capitalized R&D.
The
following is a reconciliation of income taxes computed at the Federal statutory
income tax rate to the actual effective income tax provision:
|
Year
Ended December 31,
|
||||||
|
2009
|
2008
|
2007
|
||||
|
U.S.
Federal statutory rate
|
(34.0)%
|
(34.0)%
|
(34.0)%
|
|||
|
State
income taxes, net of Federal benefit
|
(4.6)
|
(5.4)
|
(5.6)
|
|||
|
Research
and experimental tax credit
|
(4.0)
|
(4.3)
|
(4.2)
|
|||
|
Change
in valuation allowance
|
40.4
|
43.3
|
40.8
|
|||
|
Equity
compensation
|
6.3
|
-
|
-
|
|||
|
Investment
tax credit
|
(3.8)
|
-
|
-
|
|||
|
Other
|
(0.3)
|
0.4
|
3.0
|
|||
|
Income
tax provision
|
0.0%
|
0.0%
|
0.0%
|
|||
As of
December 31, 2009, we had available, for tax return purposes, unused NOL’s of
approximately $229.8 million, which will expire in various years from 2018 to
2029, $18.2 million of which were generated from deductions that, when realized,
will reduce taxes payable and will increase paid-in-capital.
We have
reviewed our nexus in various tax jurisdictions and our tax positions related to
all open tax years for events that could change the status of our ASC 740 Income
Taxes liability, if any, or require an additional liability to be recorded. Such
events may be the resolution of issues raised by a taxing authority, expiration
of the statute of limitations for a prior open tax year or new transactions for
which a tax position may be deemed to be uncertain. During the years ended
December 31, 2009, 2008 and 2007, we had no unrecognized tax benefits resulting
from tax positions during a prior or current period, settlements with taxing
authorities or the expiration of the applicable statute of limitations. At
December 31, 2009, there were no amounts of unrecognized tax benefits that, if
recognized, would affect the effective tax rate and there were no tax positions
for which it is reasonably possible that the total amounts of unrecognized tax
benefits will significantly increase or decrease within twelve months from the
respective date. As of December 31, 2009, we are subject to federal and state
income tax in the United States. Open tax years relate to years in which unused
net operating losses were generated or, if used, for which the statute of
limitation for examination by taxing authorities has not expired. Our open tax
years extend back to 1995, with the exception of 1997, during which we reported
net income. No amounts of interest or penalties were recognized in our
Consolidated Statements of Operations or Consolidated Balance Sheets upon
adoption of ASC 740 Income Taxes as of and for the years ended December 31,
2009, 2008 and 2007.
F-26
PROGENICS PHARMACEUTICALS,
INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS ¾
continued
(amounts
in thousands, except per share amounts or unless otherwise noted)
Our
research and experimental (“R&E”) tax credit carry-forwards of approximately
$10.4 million at December 31, 2009 expire in various years from 2010 to 2029.
During the year ended December 31, 2009, research and experimental tax credit
carry-forwards of approximately $144 expired.
12.
Net Loss Per Share
Our basic
net loss per share amounts have been computed by dividing net loss by the
weighted-average number of common shares outstanding during the period. For the
years ended December 31, 2009, 2008 and 2007, we reported a net loss and,
therefore, potential common shares were not included since such inclusion would
have been anti-dilutive. The calculations of net loss per share, basic and
diluted, are as follows:
|
Net
Loss
(Numerator)
|
Weighted
Average
Common
Shares
(Denominator)
|
Per
Share
Amount
|
||||||||||
|
2009:
|
||||||||||||
|
Basic
and diluted
|
$ | (30,612 | ) | 31,219 | $ | (0.98 | ) | |||||
|
2008:
|
||||||||||||
|
Basic
and diluted
|
$ | (44,672 | ) | 30,142 | $ | (1.48 | ) | |||||
|
2007:
|
||||||||||||
|
Basic
and diluted
|
$ | (43,688 | ) | 27,447 | $ | (1.59 | ) | |||||
For the
years ended December 31, 2009, 2008 and 2007, potential common shares which have
been excluded from diluted per share amounts because their effect would have
been anti-dilutive include the following:
|
Years Ended
December 31,
|
||||||||||||||||||||||||
|
2009
|
2008
|
2007
|
||||||||||||||||||||||
|
Weighted
Average
Number
|
Weighted
Average
Exercise
Price
|
Weighted
Average
Number
|
Weighted
Average
Exercise
Price
|
Weighted
Average
Number
|
Weighted
Average
Exercise
Price
|
|||||||||||||||||||
|
Options
and warrants
|
4,705 | $ | 17.48 | 4,854 | $ | 18.01 | 4,703 | $ | 17.56 | |||||||||||||||
|
Restricted
stock
|
516 | 522 | 454 | |||||||||||||||||||||
|
Total
|
5,221 | 5,376 | 5,157 | |||||||||||||||||||||
13.
Unaudited Quarterly Results
Summarized
quarterly financial data for the years ended December 31, 2009 and 2008 are as
follows:
|
Quarter
Ended
March
31,
2009
(unaudited)
|
Quarter
Ended
June
30,
2009
(unaudited)
|
Quarter
Ended
September
30,
2009
(unaudited)
|
Quarter
Ended
December
31,
2009
(unaudited)
|
|||||||||||||
|
Revenue
|
$ | 20,904 | $ | 5,469 | $ | 5,419 | $ | 17,155 | ||||||||
|
Net
loss
|
(1,788 | ) | (15,171 | ) | (13,014 | ) | (639 | ) | ||||||||
|
Net
loss per share (basic and diluted)
|
(0.06 | ) | (0.49 | ) | (0.41 | ) | (0.02 | ) | ||||||||
|
Quarter
Ended
March
31,
2008
(unaudited)
|
Quarter
Ended
June
30,
2008
(unaudited)
|
Quarter
Ended
September
30,
2008
(unaudited)
|
Quarter
Ended
December
31,
2008
(unaudited)
|
|||||||||||||
|
Revenue
|
$ | 14,762 | $ | 28,584 | $ | 17,497 | $ | 6,828 | ||||||||
|
Net
loss
|
(15,485 | ) | (2,369 | ) | (12,220 | ) | (14,598 | ) | ||||||||
|
Net
loss per share (basic and diluted)
|
(0.52 | ) | (0.08 | ) | (0.40 | ) | (0.48 | ) | ||||||||
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the Registrant has duly caused this report to be signed on its behalf by
the undersigned, hereunto duly authorized.
|
PROGENICS
PHARMACEUTICALS, INC.
|
||
|
By:
|
/s/
PAUL J. MADDON
|
|
|
Paul
J. Maddon, M.D., Ph.D.
(Duly
authorized officer of the
Registrant
and Chief Executive Officer, Chief Science Officer and
Director)
|
||
Date:
March 15, 2010
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the registrant in the
capacities and on the dates indicated.
|
Capacity
|
Date
|
||
|
/s/
KURT W. BRINER
|
Chairman
|
March
15, 2010
|
|
|
Kurt
W. Briner
|
|||
|
/s/
PAUL J. MADDON
|
Chief
Executive Officer, Chief Science
|
March
15, 2010
|
|
|
Paul
J. Maddon, M.D., Ph.D.
|
Officer
and Director (Principal Executive Officer)
|
||
|
/s/
MARK R. BAKER
|
President
and Director
|
March
15, 2010
|
|
|
Mark
R. Baker, J.D.
|
|||
|
/s/
CHARLES A. BAKER
|
Director
|
March
15, 2010
|
|
|
Charles
A. Baker
|
|||
|
/s/
PETER J. CROWLEY
|
Director
|
March
15, 2010
|
|
|
Peter
J. Crowley
|
|||
|
/s/
MARK F. DALTON
|
Director
|
March
15, 2010
|
|
|
Mark
F. Dalton
|
|||
|
|
Director
|
March
__, 2010
|
|
|
Stephen
P. Goff, Ph.D.
|
|||
|
/s/
DAVID A. SCHEINBERG
|
Director
|
March
15, 2010
|
|
|
David
A. Scheinberg, M.D., Ph.D.
|
|||
|
/s/
NICOLE S. WILLIAMS
|
Director
|
March
15, 2010
|
|
|
Nicole
S. Williams
|
|||
|
/s/
ROBERT A. MCKINNEY
|
Chief
Financial Officer, Senior Vice President,
|
March
15, 2010
|
|
|
Robert
A. McKinney, CPA
|
Finance
& Operations and Treasurer
|
||
|
(Principal
Financial and Accounting Officer)
|
EXHIBIT INDEX
|
Exhibit
|
||
|
Number
*
|
Description
|
|
|
3.1(14)
|
Restated
Certificate of Incorporation of the Registrant.
|
|
|
3.2(14)
|
Amended
and Restated By-laws of the Registrant.
|
|
|
4.1(1)
|
Specimen
Certificate for Common Stock, $0.0013 par value per share, of the
Registrant.
|
|
|
10.1(1)
|
Form
of Registration Rights Agreement.
|
|
|
10.2(1)
|
1989
Non-Qualified Stock Option Plan‡
|
|
|
10.3(1)
|
1993
Stock Option Plan, as amended‡
|
|
|
10.4(1)
|
1993
Executive Stock Option Plan‡
|
|
|
10.5(3)
|
Amended
and Restated 1996 Stock Incentive Plan‡
|
|
|
10.6(14)
|
2005
Stock Incentive Plan‡
|
|
|
10.6.1(10)
|
Form
of Non-Qualified Stock Option Award Agreement‡
|
|
|
10.6.2(10)
|
Form
of Restricted Stock Award Agreement‡
|
|
|
10.6.3(16)
|
Amended
2005 Stock Incentive Plan ‡
|
|
|
10.6.4(18)
|
Form
of Non-Qualified Stock Option Award Agreement ‡
|
|
|
10.6.5(18)
|
Form of Restricted Stock Award
Agreement ‡
|
|
|
10.7(15)
|
Form
of Indemnification Agreement‡
|
|
|
10.8(19)
|
Employment
Agreement, dated December 31, 2007, between the Registrant and
Dr. Paul J. Maddon‡
|
|
|
10.9(1)
|
Letter
dated August 25, 1994 between the Registrant and Dr. Robert J.
Israel‡
|
|
|
10.10(8)
|
Amended
1998 Employee Stock Purchase Plan‡
|
|
|
10.11(8)
|
Amended
1998 Non-qualified Employee Stock Purchase Plan‡
|
|
|
10.15(5)
|
Amended
and Restated Sublease, dated June 6, 2000, between the Registrant and
Crompton Corporation.
|
|
|
10.16(2)†
|
Development
and License Agreements, dated April 30, 1999, between Protein Design
Labs, Inc. and the Registrant.
|
|
|
10.16.1(11)
|
Letter
Agreement, dated November 24, 2003, relating to the Development and
License Agreement between Protein Design Labs, Inc. and the
Registrant.
|
|
|
10.18(4)
|
Director
Stock Option Plan‡
|
|
|
10.19(6)†
|
Exclusive
Sublicense Agreement, dated September 21, 2001, between the
Registrant and UR Labs, Inc.
|
|
|
10.19.1(9)
|
Amendment
to Exclusive Sublicense Agreement, dated September 21, 2001, between
the Registrant and UR Labs, Inc.
|
|
|
10.20(7)
|
Research
and Development Contract, dated September 26, 2003, between the
National Institutes of Health and the Registrant.
|
|
|
10.21(7)
|
Agreement
of Lease, dated September 30, 2003, between Eastview Holdings LLC and
the Registrant.
|
|
|
10.22(7)
|
Letter
Agreement, dated October 23, 2003, amending Agreement of Lease
between Eastview Holdings LLC and the Registrant.
|
|
|
10.23(11)
|
Summary
of Non-Employee Director Compensation‡
|
|
|
10.24(12)
†
|
License
and Co-Development Agreement, dated December 23, 2005, by and among
Wyeth, acting through Wyeth Pharmaceuticals Division, Wyeth-Whitehall
Pharmaceuticals, Inc. and Wyeth-Ayerst Lederle, Inc. and the Registrant
and Progenics Pharmaceuticals Nevada, Inc.
|
|
|
10.25(12)
†
|
Option
and License Agreement, dated May 8, 1985, by and between the
University of Chicago and UR Labs, Inc., as amended by (i) Amendment to
Option and License Agreement, dated September 17, 1987, by and
between the University of Chicago and UR Labs, Inc., (ii) Second Amendment
to Option and License Agreement, dated March 3, 1989, by and among
the University of Chicago, ARCH Development Corporation and UR Labs, Inc.,
and (iii) Letter Agreement Related to Progenics’ RELISTOR In-License
dated, December 22, 2005, by and among the University of Chicago,
acting on behalf of itself and ARCH Development Corporation, the
Registrant, Progenics Pharmaceuticals Nevada, Inc. and Wyeth, acting
through its Wyeth Pharmaceuticals Division.
|
|
|
10.26(13)
|
Membership
Interest Purchase Agreement, dated April 20, 2006, between the
Registrant Inc. and Cytogen Corporation.
|
|
|
10.27(13)
†
|
Amended
and Restated PSMA/PSMP License Agreement, dated April 20, 2006, by
and among the Registrant, Cytogen Corporation and PSMA Development Company
LLC.
|
|
|
10.28(17)
|
Consulting
Agreement, dated May 1, 1995, between Active Biotherapies, Inc. and
Dr. David A. Scheinberg, M.D., Ph.D., as amended on June 13,
1995, as assigned to the Registrant, and as amended on January 1,
2001‡
|
|
|
10.29(20)
†
|
License
Agreement, dated as of October 16, 2008, by and among Ono Pharmaceutical
Co., Ltd. and the Registrant.
|
|
10.30(20) †
|
Partial
Termination and License Agreement, dated October 16, 2008, by and
among Wyeth, acting through Wyeth Pharmaceuticals Division,
Wyeth-Whitehall Pharmaceuticals, Inc. and Wyeth-Ayerst Lederle, Inc. and
the Registrant and Progenics Pharmaceuticals Nevada,
Inc.
|
|
|
10.31(20) †
|
Consent,
Acknowledgment and Agreement, dated as of October 16, 2008, by and among
Wyeth, acting through Wyeth Pharmaceuticals Division, Wyeth-Whitehall
Pharmaceuticals, Inc. and Wyeth-Ayerst Lederle, Inc., the Registrant and
Ono Pharmaceutical Co., Ltd.
|
|
|
10.32(20) †
|
2008
Agreement Related to Progenics’ MNTX In-License, dated October 16,
2008, by and among the University of Chicago, acting on behalf of itself
and ARCH Development Corporation, the Registrant, Progenics
Pharmaceuticals Nevada, Inc. and Ono Pharmaceutical Co.,
Ltd.
|
|
|
10.33
††
|
Termination
and Transition Agreement, effective as of October 1, 2009, by and among
Wyeth, acting through Wyeth Pharmaceuticals Division, Wyeth-Whitehall
Pharmaceuticals, Inc., Wyeth-Ayerst Lederle, Inc., and AHP Manufacturing
B.V., and the Registrant, Progenics Pharmaceuticals Nevada, Inc. and
Excelsior Life Sciences Ireland Limited.
|
|
|
21.1(19)
|
Subsidiaries
of the Registrant.
|
|
|
23.1
|
Consent
of PricewaterhouseCoopers LLP.
|
|
|
31.1
|
Certification
of Paul J. Maddon, M.D., Ph.D., Chief Executive Officer of the Registrant
pursuant to 13a-14(a) and Rule 15d-14(a) under the Securities
Exchange Act of 1934, as amended.
|
|
|
31.2
|
Certification
of Robert A. McKinney, Chief Financial Officer, Senior Vice President,
Finance and Operations and Treasurer of the Registrant pursuant to
13a-14(a) and Rule 15d-14(a) under the Securities Exchange Act of 1934, as
amended.
|
|
|
32.1
|
Certification
of Paul J. Maddon, M.D., Ph.D., Chief Executive Officer of the Registrant
pursuant to 18 U.S.C. Section 1350, as adopted pursuant to
Section 906 of the Sarbanes-Oxley Act of 2002.
|
|
|
32.2
|
Certification
of Robert A. McKinney, Chief Financial Officer, Senior Vice President,
Finance and Operations and Treasurer of the Registrant pursuant to 18
U.S.C. Section 1350, as adopted pursuant to Section 906 of the
Sarbanes-Oxley Act of 2002.
|
____________
|
*
|
Exhibits
footnoted as previously filed have been filed as an exhibit to the
document of the Registrant referenced in the footnote below, and are
incorporated by reference herein.
|
|
|
(1)
|
Previously
filed in Registration Statement on Form S-1, Commission File
No. 333-13627.
|
|
|
(2)
|
Previously
filed in Quarterly Report on Form 10-Q for the quarterly period ended
June 30, 1999.
|
|
|
(3)
|
Previously
filed in Registration Statement on Form S-8, Commission File
No. 333-120508.
|
|
|
(4)
|
Previously
filed in Annual Report on Form 10-K for the year ended
December 31, 1999.
|
|
|
(5)
|
Previously
filed in Quarterly Report on Form 10-Q for the quarterly period ended
June 30, 2000.
|
|
|
(6)
|
Previously
filed in Annual Report on Form 10-K for the year ended
December 31, 2002.
|
|
|
(7)
|
Previously
filed in Quarterly Report on Form 10-Q for the quarterly period
ending September 30, 2003.
|
|
|
(8)
|
Previously
filed in Registration Statement on Form S-8, Commission File
No. 333-143671.
|
|
|
(9)
|
Previously
filed in Current Report on Form 8-K filed on September 20,
2004.
|
|
|
(10)
|
Previously
filed in Current Report on Form 8-K filed on July 8,
2008.
|
|
|
(11)
|
Previously
filed in Annual Report on Form 10-K for the year ended
December 31, 2004.
|
|
|
(12)
|
Previously
filed in Annual Report on Form 10-K for the year ended
December 31, 2005.
|
|
|
(13)
|
Previously
filed in Quarterly Report on Form 10-Q for the quarterly period
ending June 30, 2006.
|
|
|
(14)
|
Previously
filed in Current Report on Form 8-K filed on May 13,
2005.
|
|
|
(15)
|
Previously
filed in Quarterly Report on Form 10-Q for the quarterly period
ending March 31, 2007.
|
|
|
(16)
|
Previously
filed in Registration Statement on Form S-8, Commission File
No. 333-143670.
|
|
|
(17)
|
Previously
filed in Annual Report on Form 10-K/A for the year ended
December 31, 2006.
|
|
|
(18)
|
Previously
filed in Current Report on Form 8-K filed on July 8,
2008.
|
|
|
(19)
|
Previously
filed in Annual Report on Form 10-K for the year ended December 31,
2007.
|
|
|
(20)
|
Previously
filed in Annual Report on Form 10-K for the year ended December 31,
2008.
|
|
|
†
|
Confidential
treatment granted as to certain portions omitted and filed separately with
the Commission.
|
|
|
††
|
Confidential
treatment requested as to certain portions omitted and filed separately
with the Commission.
|
|
|
‡
|
Management
contract or compensatory plan or
arrangement.
|
|
