Attached files
| file | filename |
|---|---|
| 8-K - PDL BIOPHARMA, INC. | v213740_8k.htm |
31st Annual Cowen Health Care Conference March 8, 2011

Forward Looking Statements This presentation contains forward -looking statements, including PDL’s expectations with respect to its future royalty revenues, expenses, net income, and cash provided by operating activities. Each of these forward -looking statements involves risks and uncertainties. Actual results may differ materially from those, express or implied, in these forward -looking statements. Factors that may cause differences between current expectations and actual results include, but are not limited to, the following: •The expected rate of growth in royalty-bearing product sales by PDL’s existing licensees; • The relative mix of royalty-bearing Genentech products manufactured and sold outside the U.S. versus manufactured or sold in the U.S.; • The ability of our licensees to receive regulatory approvals to market and launch new royalty-bearing
products and whether such products, if launched, will be commercially successful; • Changes in any of the other assumptions on which PDL’s projected royalty revenues are based; • Changes in foreign currency rates; • Positive or negative results in PDL’s attempt to acquire royalty-related assets; • The outcome of pending litigation or disputes, including our current dispute with Genentech related to ex-U.S. sales of Genentech licensed products; and • The failure of licensees to comply with existing license agreements, including any failure to pay royalties due. Other factors that may cause PDL’s actual results to differ materially from those expressed or implied in the forward -looking statements in this presentation are discussed in PDL’s filings with the SEC, including the "Risk Factors" sections of its annual and quarterly reports filed with the SEC. Copies of PDL’s filings with the SEC may be obtained at the
"Investors" section of PDL’s website at www.pdl.com. PDL expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any forward -looking statements contained herein to reflect any change in PDL’s expectations with regard thereto or any change in events, conditions or circumstances on which any such statements are based for any reason, except as required by law, even as new information becomes available or other events occur in the future. All forward -looking statements in this presentation are qualified in their entirety by this cautionary statement.

Key Information Company: PDL BioPharma, Inc. Ticker: PDLI (NASDAQ) Location: Incline Village, Nevada Employees: Less than 10 2010 Revenues: $345 million 2011-Q1 Revenue Guidance: $83 million 2011 Regular Dividends: $0.60 /share paid quarterly in $0.15 increments on March 15, June 15, September 15 & December 15 EOY-2010 Cash Position 1: $248 million Shares O/S2: ~ 140 million Average Daily Volume: ~ 2.9 million shares 1. As of December 31, 2010; 2. Not fully diluted

Overview of PDL BioPharma

Company Overview • PDL pioneered the humanization of monoclonal antibodies which enabled the discovery of a new generation of targeted treatments for cancer and immunologic diseases • PDL’s primary assets are its antibody humanization patents and royalty assets which consist of its Queen et al. patents and license agreements • Licensees consist of large biotechnology and pharmaceutical companies including Roche/Genentech/ Novartis, Elan/BiogenIdec, Pfizer/Wyeth/J&J and Chugai

Antibody Humanization Technology • Antibodies are naturally produced by humans to fight foreign substances, such as bacteria and viruses • In the 1980’s, scientists began creating antibodies in non-human immune systems, such as those of mice, that could target specific sites on cells to fight various human diseases • However, mouse derived antibodies are recognized by the human body as foreign substances and may be rejected by the human immune system • PDL’s technology allows for the “humanization” of mouse derived antibodies by moving the important binding regions from the mouse antibody onto a human framework • PDL’s humanization technology is important because the humanized antibodies retain the binding and activity levels from the
original mouse antibody • PDL’s technology has been incorporated into antibodies to treat cancer, eye diseases, arthritis, multiple sclerosis and other health conditions with aggregate annual sales of almost $20 billion

Mission • Manage patent portfolio • Manage license agreements • Optimize return for shareholders

Corporate Governance Management Board of Directors • John McLaughlin • Fred Frank President & CEO Lead Director • Christine Larson • Jody Lindell VP & CFO • John McLaughlin • Christopher Stone VP, General Counsel & • Paul Sandman Secretary • Harold Selick • Caroline Krumel VP of Finance

Royalty Revenue

Royalty Revenue & Licensed Products Royalties by Product ($ in millions) $400 $350 $300 $250 $200 $150 $100 $50 $0 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 Herceptin Avastin Lucentis Xolair Synagis Tysabri Other
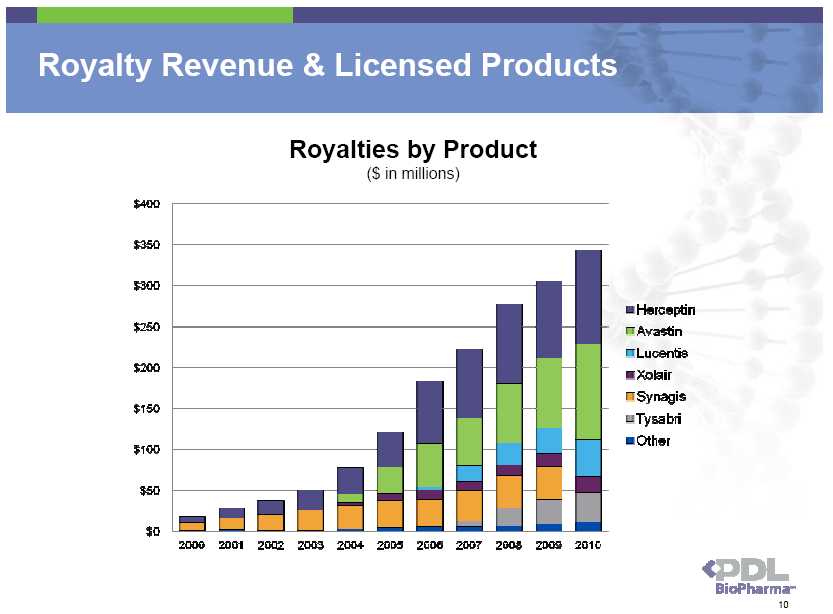
Royalties: When Licensed Product is Made or Sold • PDL’s revenues consist of royalties generated on sales of licensed products • Sold before the expiration of the Queen et al. patents in 2013/14 or • Made prior to the expiration of the Queen et al. patents and sold anytime thereafter Example of Antibody Bulk Manufacturing Schedule Purification to Concentrated Bulk/Frozen Cell Quality Release Culture Bulk Frozen Storage Testing 3 mos 1mo 1mo 2-18 months 1 mo 3 mos 5 mos 10 mos 15 mos 20 mos 27 mos Example of Antibody Formulation, Fill and Finish Schedule Thaw, Formulation Quality Packaging & Vial Filling Release & Quality Inventory ½ month 1 month ½ month 2-3 months
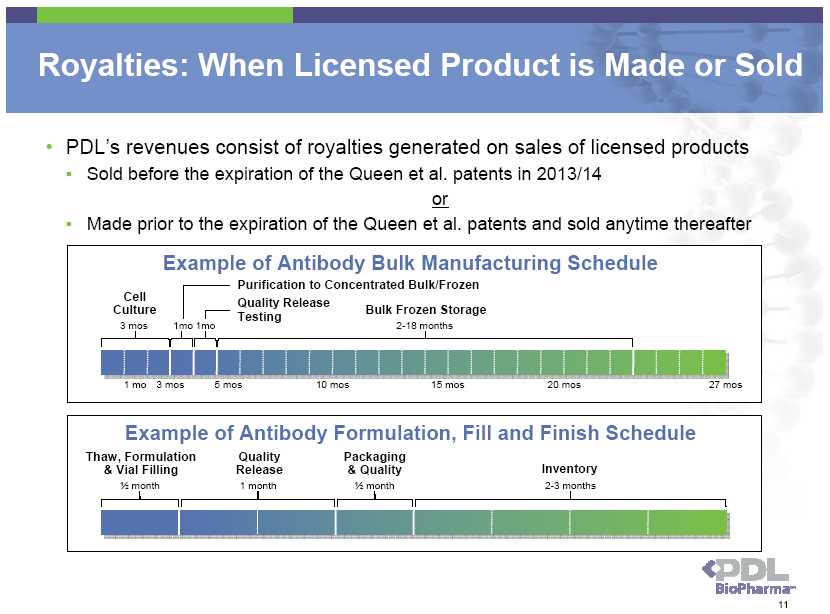
Genentech/Roche Royalties * Product Made in U.S. Net Sales up to $1.5 Billion 3.0% Net Sales Between $1.5 Billion and $2.5 Billion 2.5% Net Sales Between $2.5 Billion and $4.0 Billion 2.0% Net Sales Over $4.0 Billion 1.0% Product Made and Sold Ex-U.S. All Sales 3.0% * Excludes royalties for Actemra / RoActemra • Genentech/Roche commercialized products include Avastin, Herceptin, Lucentis and Xolair • In 2009, only 12% of Genentech/Roche sales were ex-U.S. manufactured and sold products • In 2010, 26% of Genentech/Roche sales were ex-U.S. manufactured and sold products • Average royalty rate on all Genentech/Roche products under Genentech license in 2010 was 1.9% versus 1.7% in 2009 • U.S. only effective rate was 1.5% in both years
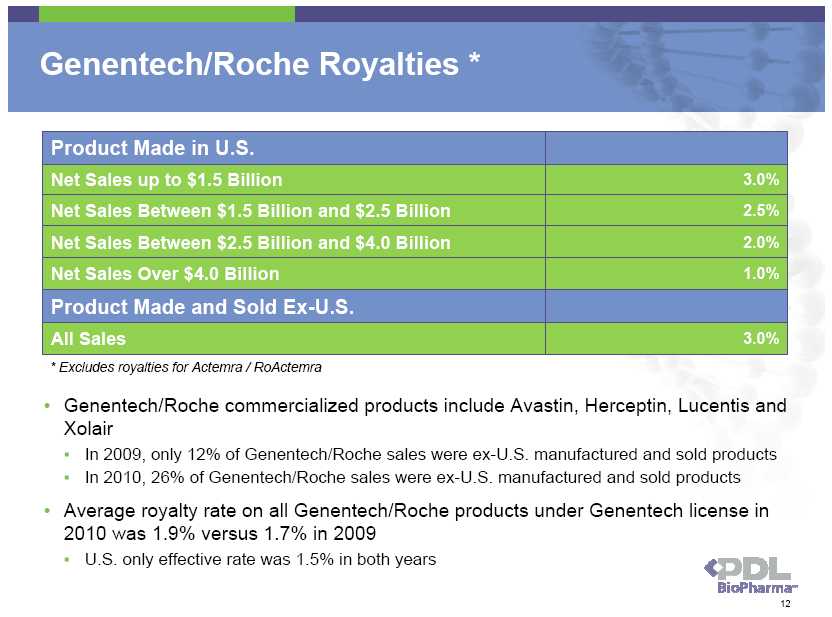
Genentech/Roche – Future Manufacturing • Roche has begun to move some manufacturing ex-U.S. • Current production at Penzburg (Herceptin) and Basel (Avastin) plants • Two new plants in Singapore (CHO = antibody and e. coli = antibody fragment) - E. coli (Lucentis) and CHO (Avastin) plants are approved for commercial supply to the US - E. coli and CHO plants are expected to be approved for commercial supply to the EU in 2011 - Currently, all Lucentis is made in the U.S.
Genentech/Roche - Future Royalty Products • In December 2008, Genentech exercised options for 4 additional antigens and extended other options paying fees of $1.8 million • Genentech can convert the exercised options into license agreements by identifying the target antigen if certain other conditions are met • Genentech/Roche have a number of humanized antibodies in Phase 2/3 • Pertuzumab: HER2+ breast cancer • Ocrelizumab: Relapsing remitting multiple sclerosis • Lebrikizumab: Asthma

Genentech / Roche – US & EU Filings Avastin BC Adjuvant HER2+ Avastin Avastin BC Adj Triple Negative HER 2- BC adj Avastin Avastin Avastin mCRC TML Glioblastoma 1st Line NSCLC adj Avastin & Herceptin Herceptin HER2+ mBC 1st Line BC HER 2+ Adj 2 Year Avastin Herceptin Ovarian Cancer 1st Line US Subcutaneous Formulation Avastin Actemra Relapsed Ovarian Cancer Ankylosing Spondylitis Avastin + Herceptin Actemra mBC HER+ 2nd Line SC Formulation (EU) Lucentis Actemra Diabetic Macular Edema (US) RA DMARD H2H (EU) Pertuzumab 1 Afutuzumab (GA101) mBC HER2+ 1st Line Chronic
Lymphocytic Leukemia 2011 2012 2013 Post 2013 1. Not a licensed product

Royalty Products – Approved

Royalty Products – Avastin Licensee Product Status Indications Roche (Genentech) Avastin Approved Colorectal Cancer NSCLC Metastatic Renal Cell Glioblastoma Metastatic Breast HER2- 1st Line sBLA Metastatic Breast HER2- 2nd Line Phase 3 Ovarian Cancer Gastric Prostate Cancer Adjuvant settings On December 16, 2010, FDA notified Roche/Genentech of its intention to withdraw Avastin’s approval as first line treatment for HER2- breast cancer inHER2+ Stomach and Gastro- combination Esophageal with cancers paclitaxel . In response to request from Genentech, FDA scheduled a hearing on June 28-29, 2011 to allow Genentech to present why Avastin Phase should3 remain DME FDA-approved for HER2- breast cancer. On December 16,
2010, EMA narrowed, but did not withdraw, Avastin’s approval for first line treatment of HER2- breast cancer to use in combination with paclitaxel only. Roche lowered its estimate of peak annual sales from of Avastin from CHF8 – CHF9 billion to CHF7 billion. Based on our internal model, we project Avastin for treatment of metastatic HER2- breast cancer represents slightly more than 2% of total PDL royalty revenue.

Royalty Products – Avastin Licensee Product Status Indications Roche (Genentech) Avastin Approved Colorectal Cancer NSCLC Metastatic Renal Cell Glioblastoma Metastatic Breast HER2- 1st Line sBLA Metastatic Breast HER2- 2nd Line Phase 3 Ovarian Cancer Gastric On February 7, 2011, Genentech reported that Phase 3 trial in women with previously treated (recurrent), platinum-sensitive ovarian cancer showed an improvement in progression free survival in those patients treated with Avastin in combination with chemotherapy (carboplatin and gemcitabine) followed by continued use of Avastin alone compared to those treated with chemotherapy alone. Two previous Phase 3 studies in women with newly diagnosed ovarian cancer demonstrated that
front-line Avastin in combination with standard chemotherapy (carboplatin and paclitaxel), followed by the continued use of Avastin alone, significantly increased progression free survival compared to treatment with chemotherapy alone. Roche has submitted an application for approval for front line treatment in EU and expects a decision later in 2011 Genentech expects to file an application for approval in US in 2011.

Royalty Products – Lucentis On January 7, Novartis announced that Lucentis has been approved in the EU for the treatment visual impairment due to diabetic macular edema (DME). DME is a leading cause of blindness in the working-age population in most developed countries. On February 11, 2011, Genentech announced that one of two Phase 3 studies evaluating in patients with DME showed that a significantly higher percentage of patients receiving monthly dosing of Lucentis achieved an improvement in vision of at least 15 letters on the eye chart at 24 months compared to those in a control group, who received a placebo injection. Lucentis Approved AMD Approved RVO Phase 3 (US) DME Xolair Approved Moderate -Severe Asthma sBLA Pediatric Asthma Elan Tysabri Approved Multiple Sclerosis Roche (Chugai)
Actemra Approved Rheumatoid Arthritis
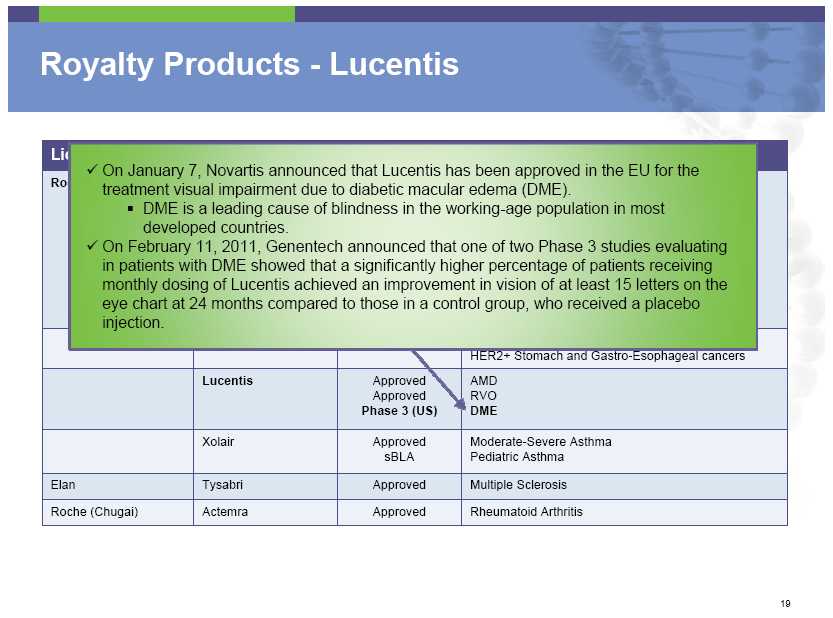
Royalty Products – Lucentis On November 22, 2010, Regeneron and its partner, Bayer reported top line data from two Phase 3 trials investigating its VEGF trap in age-related macular degeneration (AMD) patients which suggest that it may be injected into the eye every other month with safety and efficacy comparable to that of monthly dosing of Lucentis. On December 20, 2010 Regeneron has also reported positive Phase 3 data in the treatment of retinal vein occlusion (RVO) for which Lucentis is approved. Unlike the AMD trial, monthly administration was used in the RVO trial, which does not afford a dosing advantage with respect to Lucentis. On February, 22, 2011, Regeneron and its partner, Bayer, had filed an application for approval of its VEGF trap for treatment of AMD. Lucentis Approved AMD
Approved RVO Phase 3 DME Xolair Approved Moderate -Severe Asthma sBLA Pediatric Asthma Elan Tysabri Approved Multiple Sclerosis Roche (Chugai) Actemra Approved Rheumatoid Arthritis

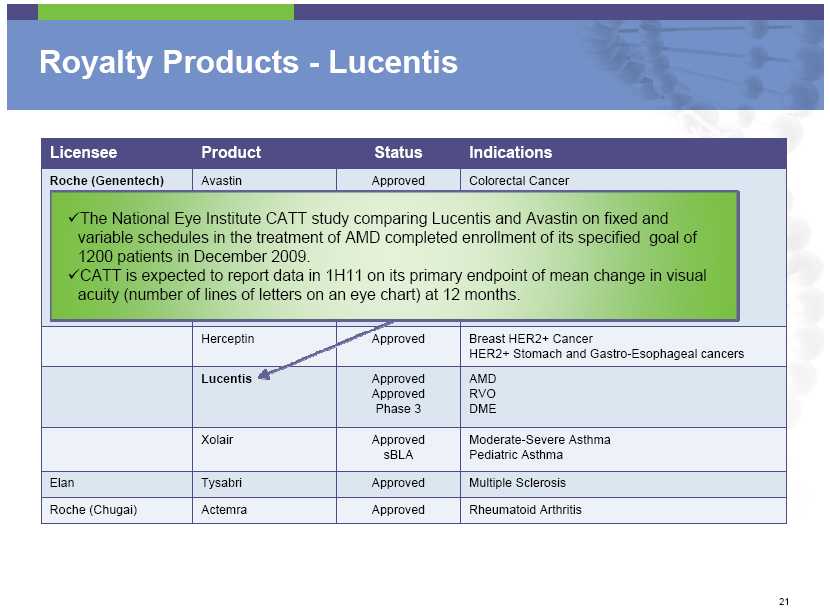
Royalty Products – Lucentis Licensee Product Status Indications Roche (Genentech) Avastin Approved Colorectal Cancer The National Eye Institute CATT study comparing Lucentis and Avastin on fixed and variable schedules in the treatment of AMD completed enrollment of its specified goal of 1200 patients in December 2009. CATT is expected to report data in 1H11 on its primary endpoint of mean change in visual acuity (number of lines of letters on an eye chart) at 12 months . Herceptin Approved Breast HER2+ Cancer HER2+ Stomach and Gastro-Esophageal cancers Lucentis Approved AMD Approved RVO Phase 3 DME Xolair Approved Moderate
-Severe Asthma sBLA Pediatric Asthma Elan Tysabri Approved Multiple Sclerosis Roche (Chugai) Actemra Approved Rheumatoid Arthritis
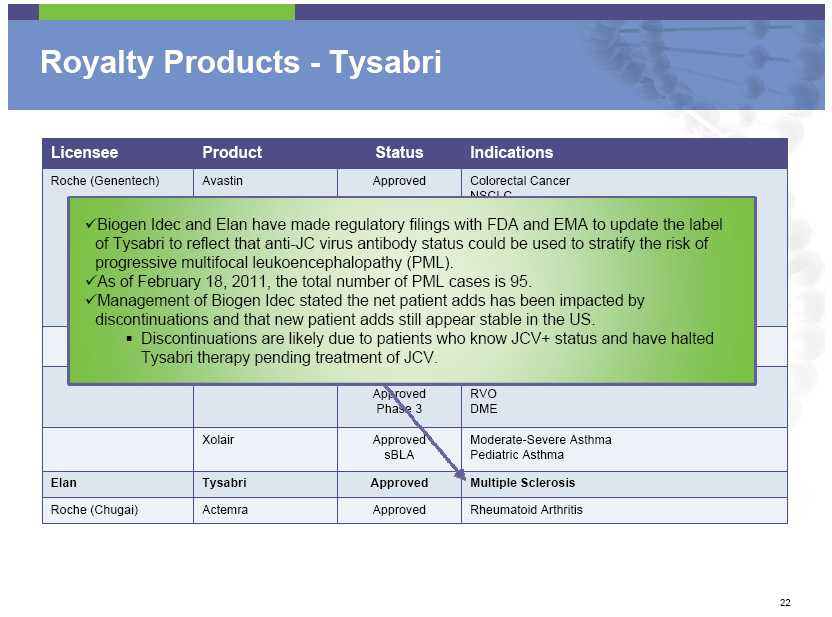
Royalty Products – Tysabri Licensee Product Status Indications Roche (Genentech) Avastin Approved Colorectal Cancer NSCLC Biogen Idec and Elan have made regulatory filings with FDA and EMA to update the label of Tysabri to reflect that anti-JC virus antibody status could be used to stratify the risk of progressive multifocal leukoencephalopathy (PML). As of February 18, 2011, the total number of PML cases is 95. Management of Biogen Idec stated the net patient adds has been impacted by discontinuations and that new patient adds still appear stable in the US. Discontinuations are likely due to Approved patients who know JCV+ status and have halted Tysabri therapy pending treatment of JCV. Approved RVO Phase 3 DME Xolair Approved Moderate -Severe Asthma sBLA Pediatric Asthma Elan Tysabri Approved
Multiple Sclerosis Roche (Chugai) Actemra Approved Rheumatoid Arthritis
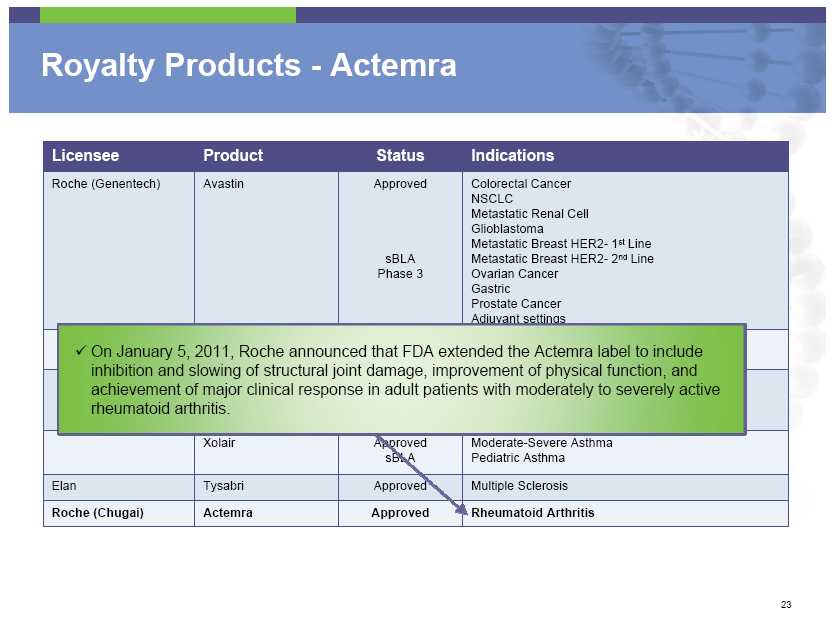
Royalty Products – Actemra Licensee Product Status Indications Roche (Genentech) Avastin Approved Colorectal Cancer NSCLC Metastatic Renal Cell Glioblastoma Metastatic Breast HER2- 1st Line sBLA Metastatic Breast HER2- 2nd Line Phase 3 Ovarian Cancer Gastric Prostate Cancer Adjuvant settings On January 5, 2011, Roche announced that FDA extended the Actemra label to include inhibition and slowing of structural joint damage, improvement of physical function, and achievement of major clinical response in adult patients with moderately to severely active rheumatoid arthritis. Xolair Approved Moderate -Severe Asthma sBLA Pediatric Asthma Elan
Tysabri Approved Multiple Sclerosis Roche (Chugai) Actemra Approved Rheumatoid Arthritis
Future Royalty Products – Development Stage

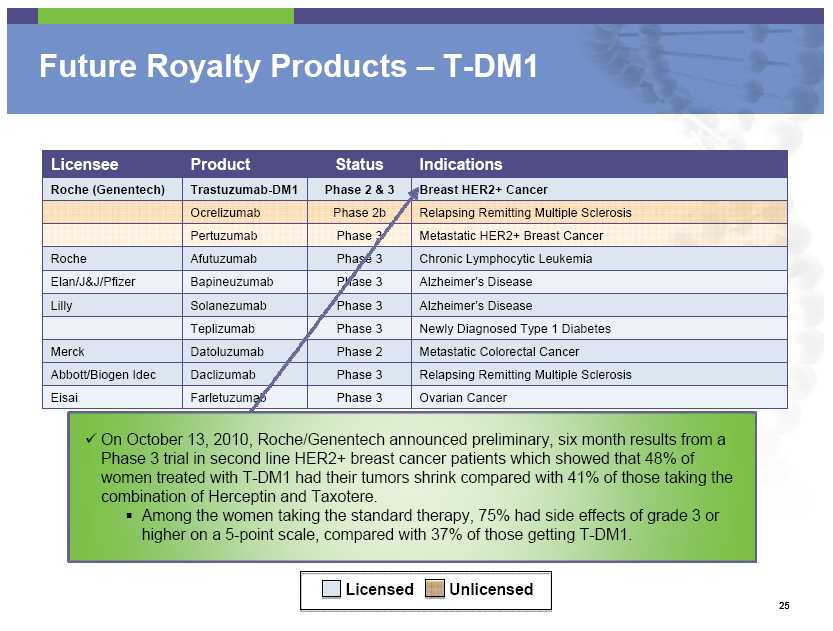
Future Royalty Products – T-DM1 Licensee Product Status Indications Roche (Genentech) Trastuzumab -DM1 Phase 2 & 3 Breast HER2+ Cancer Ocrelizumab Phase 2b Relapsing Remitting Multiple Sclerosis Pertuzumab Phase 3 Metastatic HER2+ Breast Cancer Roche Afutuzumab Phase 3 Chronic Lymphocytic Leukemia Elan/J&J/Pfizer Bapineuzumab Phase 3 Alzheimer’s Disease Lilly Solanezumab Phase 3 Alzheimer’s Disease Teplizumab Phase 3 Newly Diagnosed Type 1 Diabetes Merck Datoluzumab Phase 2 Metastatic Colorectal Cancer Abbott/Biogen Idec Daclizumab Phase 3 Relapsing Remitting Multiple Sclerosis Eisai Farletuzumab Phase 3 Ovarian Cancer On October 13, 2010, Roche/Genentech announced preliminary, six month results from a Phase 3 trial in second line HER2+ breast cancer patients which showed
that 48% of women treated with T-DM1 had their tumors shrink compared with 41% of those taking the combination of Herceptin and Taxotere. Among the women taking the standard therapy, 75% had side effects of grade 3 or higher on a 5-point scale, compared with 37% of those getting T-DM1. Licensed Unlicensed
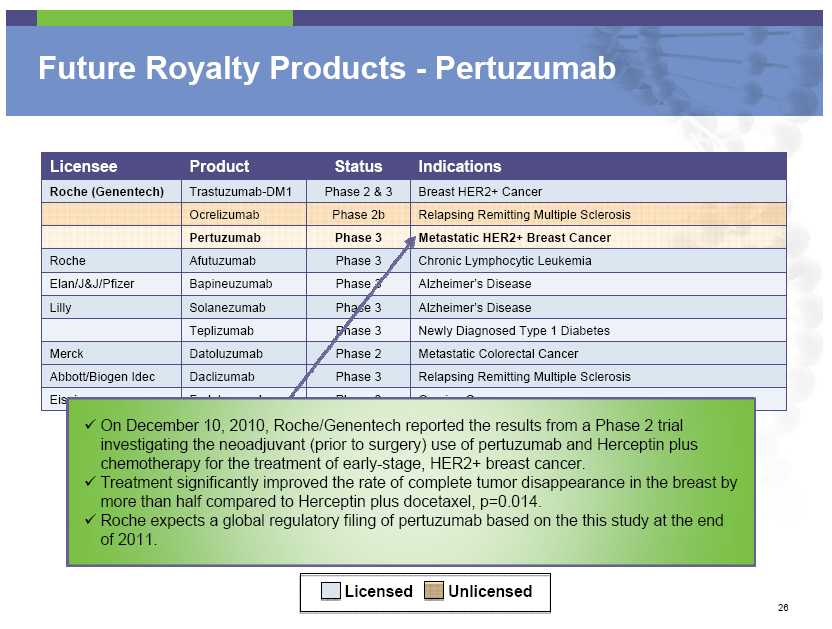
Future Royalty Products – Pertuzumab Licensee Product Status Indications Roche (Genentech) Trastuzumab -DM1 Phase 2 & 3 Breast HER2+ Cancer Ocrelizumab Phase 2b Relapsing Remitting Multiple Sclerosis Pertuzumab Phase 3 Metastatic HER2+ Breast Cancer Roche Afutuzumab Phase 3 Chronic Lymphocytic Leukemia Elan/J&J/Pfizer Bapineuzumab Phase 3 Alzheimer’s Disease Lilly Solanezumab Phase 3 Alzheimer’s Disease Teplizumab Phase 3 Newly Diagnosed Type 1 Diabetes Merck Datoluzumab Phase 2 Metastatic Colorectal Cancer Abbott/Biogen Idec Daclizumab Phase 3 Relapsing Remitting Multiple Sclerosis On December 10, 2010, Roche/Genentech reported the results from a Phase 2 trial investigating the neoadjuvant (prior to surgery) use of pertuzumab and Herceptin plus chemotherapy for
the treatment of early-stage, HER2+ breast cancer. Treatment significantly improved the rate of complete tumor disappearance in the breast by more than half compared to Herceptin plus docetaxel, p=0.014. Roche expects a global regulatory filing of pertuzumab based on the this study at the end of 2011. Licensed Unlicensed
Debt

$520 Million Total Debt • $250 million 2.00% convertible senior notes due February 2012; current balance $134 million a2010 Corporate goal to extend repayment of a portion of this debt without significant increase in coupon rate was accomplished in November 2010 Accomplished through repurchases and exchange of $92 million for new 2015 Notes Conversion rate is 140.571 shares / $1,000 face amount ($7.11/share) • $180 million 2.875% convertible senior notes due February 2015 placed November 1, 2010 In addition to exchanging 2012 Notes, placed an additional $88 million of 2015 Notes Proceeds may be used to buy back shares, repurchase 2012 Notes and/or acquire new royalty assets Conversion rate is 140.571 shares / $1,000 face amount ($7.11/share) • $300 million 10.25% note; current
balance $204 million Approximately 40% of Genentech royalties dedicated to quarterly principal and interest payments; principal repayment fluctuates in relation to royalties received Anticipated final maturity is September 2012; legal maturity is March 2015 After final maturity, securitized Genentech royalties will be retained by PDL Distributed $200 million of proceeds as special dividend of $1.67/share in December 2009

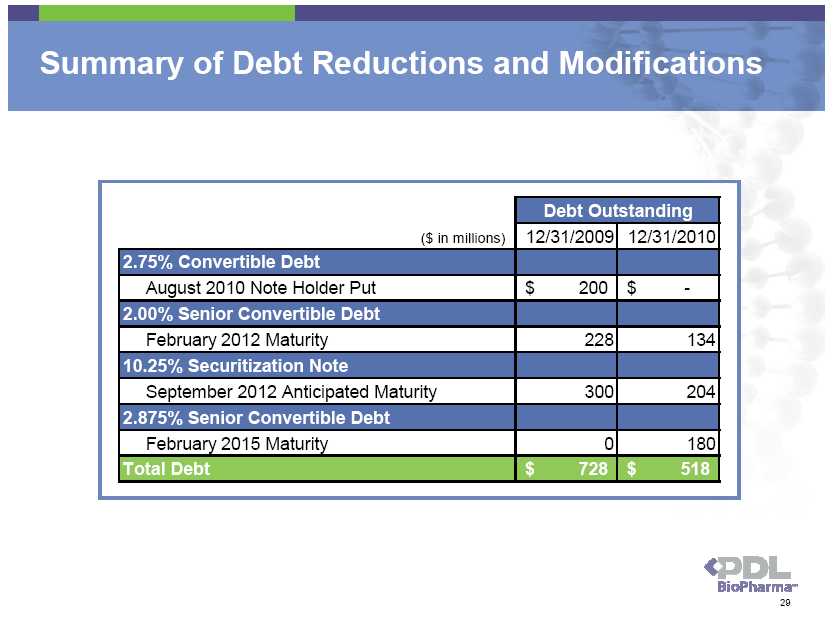
Summary of Debt Reductions and Modifications Debt Outstanding ($ in millions) 12/31/2009 12/31/2010 2.75% Convertible Debt August 2010 Note Holder Put $ 200 $ - 2.00% Senior Convertible Debt February 2012 Maturity 228 134 10.25% Securitization Note September 2012 Anticipated Maturity 300 204 2.875% Senior Convertible Debt February 2015 Maturity 0 180 Total Debt $ 728 $ 518
Legal Matters

Settlements of Disputes • UCB PDL received $10 million from UCB in return for PDL agreement not to sue UCB for any royalties related to Cimzia UCB terminated patent interference proceedings before the U.S. Patent and Trademark office ending all administrative challenges to the Queen et al. patents UCB also withdrew its opposition appeal to our European patent in the European Patent Office (EPO) • MedImmune PDL paid MedImmune $65 million on February 15, 2011 and will pay them an additional $27.5 million by February 2012 No further payments will be owed by either party MedImmune also ceased all support of any party involved in the EPO opposition of our European patent

Settlement of Disputes • Novartis PDL dismissed its claims against Novartis in its Nevada lawsuit Novartis withdrew its opposition appeal to our European patent in EPO PDL will pay Novartis an amount based on Novartis’ net ex-U.S. sales of Lucentis during calendar year 2011 and beyond, which amount is less than the royalty amount Genentech pays PDL on the same sales of Lucentis and it is not currently material Novartis settlement does not affect PDL’s claims against Genentech and Roche in the Nevada state court action BioTransplant PDL acquired BioTransplant, a bankrupt company, because it was one of the appellants in the appeal opposition to our European patent before the EPO PDL instructed BioTransplant to withdraw its opposition appeal in the EPO PDL believes that BioTransplant’s activities before EPO were financially supported by MedImmune before PDL’s
acquisition

Effect of Settlements • U.S. Patent Interferences All patent interferences before the U.S. Patent and Trademark Office relating to the Queen at al. patents have been resolved in PDL’s favor • Appeal Opposition to PDL’s European Patent With the withdrawals from EPO proceeding of UCB, MedImmune and Novartis by virtue of settlements, and BioTransplant by virtue of its acquisition by PDL: - EPO cancelled its appeal hearing, - Appeal proceeding was terminated, and - 2007 decision upholding the claims of our European patent will become the final decision of EPO In 2010, 35% of PDL’s revenues resulted from sales of products that were made in Europe and sold outside of the United States aBased on Roche announcements, PDL anticipates that this percentage of
revenues will increase in the future • MedImmune, UCB and Novartis cannot challenge the Queen et al. patents in the future nor assist others in doing so

Genentech Communication • On August 11, 2010, PDL received a fax from Genentech on behalf of Roche and Novartis asserting that Avastin, Herceptin, Lucentis and Xolair (Genentech Products) do not infringe PDL’s supplementary protection certificates (SPC’s) and seeking a response from PDL SPC’s are extensions of patent term in Europe that are issued on a country -by-country and product -by-product basis An SPC is granted to a specific product designated by generic name (e.g. trastuzumab for Herceptin) • PDL responded on August 31, 2010 that Genentech’s assertions are without merit, that we disagree with their assertions of non-infringement and, further, cautioned that Genentech had waived its rights to challenge our patents, including SPC’s There have been
discussions among the parties

Nevada Litigation • PDL filed suit against Genentech, Roche and Novartis in Nevada state court Lawsuit against Novartis has been dismissed under PDL’s settlement with Novartis • Lawsuit states that August 11th fax sent at the behest of Roche and Novartis damaged PDL and constitutes a breach of Genentech’s obligations under its 2003 Settlement Agreement with PDL • Complaint seeks compensatory damages, including liquidated damages and other monetary remedies set forth in the 2003 Settlement Agreement, punitive damages and attorney’s fees

Genentech and Roche Response • In November 2010, Genentech and Roche filed a motion to dismiss our complaint because they contend that 2003 Settlement Agreement applies only to PDL’s U.S. patent rights PDL believes that the 2003 Settlement Agreement is not limited to PDL’s U.S. patent rights but also includes PDL’s European patent rights To prevail on their motion to dismiss, Genentech and Roche must establish that PDL can prove no set of facts which, if accepted by the court, would entitle PDL to the relief requested in our complaint • In addition, Roche has asserted that the Nevada court lacks personal jurisdiction over them To prevail on their motions to dismiss for lack of jurisdiction, Roche must establish that its conduct does not permit a Nevada court to
adjudicate the claims asserted in the complaint without violating due process PDL disagrees with these arguments and intends to oppose both motions • The Nevada court has not yet fixed a date on which it would hear and decide Genentech and Roche’s motions

2003 Settlement Agreement • The 2003 Settlement Agreement was entered into as part of a definitive agreement resolving intellectual property disputes between the two companies at that time • The agreement limits Genentech’s ability to challenge infringement of our patent rights, including SPC’s, and waives Genentech’s right to challenge the validity of our patent rights • Breaches of 2003 Settlement Agreement PDL may also be entitled to either terminate our license agreements with Genentech or be paid a flat royalty of 3.75% on past and future U.S.-based Sales of the Genentech Products - Retroactive royalty rate of 3.75% on past sales of the Genentech Products made in the U.S. and sold anywhere plus interest is up to $1 billion - PDL has not
projected value of 3.75% prospective royalty on future sales of Genentech Products made in the U.S. and sold anywhere - Liquidated and other damages

Optimizing Stockholder Return

Optimizing Stockholder Return • Continuously evaluating alternatives: Dividends Convertible note buyback / capital restructure Share repurchase Company sale Purchase of commercial stage, royalty generating assets Do not expect to securitize any more assets

Investment Rationale • Strong revenue growth from approved products • Potential for additional indications from existing products, new product approvals and purchase of new royalty assets • Significantly reduced expenses with no R&D burn • Liquidity - volume averages 2.9 million shares/day • Return to stockholders In 2011, $0.60/share to be paid in quarterly regular dividends of $0.15/share on March 15, June 15, September 15 and December 15

