Attached files
| file | filename |
|---|---|
| 8-K - FORM 8K - WEST PHARMACEUTICAL SERVICES INC | form8k.htm |

WEST PHARMACEUTICAL SERVICES UPDATE
Solutions for Injectable Drug Delivery NYSE:WST www.westpharma.com
CJS Securities’
11th Annual “New Ideas for the New Year”
Investor Conference
New York, NY January 12, 2011

This presentation contains “forward-looking statements”, as that term is defined in the Private
Securities Litigation Reform Act of 1995, that are based on management’s beliefs and
assumptions, current expectations, estimates and forecasts. Statements that are not
historical facts, including statements that are preceded by, followed by, or that include, words
such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms
of similar meaning are forward-looking statements. West’s estimated or anticipated future
results, product performance or other non-historical facts are forward-looking and reflect our
current perspective on existing trends and information.
Securities Litigation Reform Act of 1995, that are based on management’s beliefs and
assumptions, current expectations, estimates and forecasts. Statements that are not
historical facts, including statements that are preceded by, followed by, or that include, words
such as “estimate,” “expect,” “intend,” “believe,” “plan,” “anticipate” and other words and terms
of similar meaning are forward-looking statements. West’s estimated or anticipated future
results, product performance or other non-historical facts are forward-looking and reflect our
current perspective on existing trends and information.
Many of the factors that will determine the Company’s future results are beyond the ability of
the Company to control or predict. These statements are subject to known or unknown risks
or uncertainties, and therefore, actual results could differ materially from past results and
those expressed or implied in any forward-looking statement. You should bear this in mind as
you consider forward-looking statements. A non-exclusive list of important factors that may
affect future results may be found in West’s filings with the Securities and Exchange
Commission, including our annual report on Form 10-K and our periodic reports on Form 10-
Q and Form 8-K. You should evaluate any statement in light of these important factors.
the Company to control or predict. These statements are subject to known or unknown risks
or uncertainties, and therefore, actual results could differ materially from past results and
those expressed or implied in any forward-looking statement. You should bear this in mind as
you consider forward-looking statements. A non-exclusive list of important factors that may
affect future results may be found in West’s filings with the Securities and Exchange
Commission, including our annual report on Form 10-K and our periodic reports on Form 10-
Q and Form 8-K. You should evaluate any statement in light of these important factors.
Safe Harbor Statement
2

West is a globally diversified manufacturer of products consumed in the healthcare and
consumer markets.
consumer markets.
Every day over 80 million West and Daikyo products are used to enhance the quality of
healthcare worldwide.
healthcare worldwide.
3

A Diverse, Stable Customer Base
Pharmaceutical/Biotech
Generic
Medical Device
4

West’s Structure
Packaging Systems
• 9-month 2010 sales: $591 MM
• Established leadership
• High market share
• Stable growth rate
Delivery Systems
• 9-month 2010 sales: $240 MM
• Proprietary devices
• Contract manufacturing
• High projected growth rate
Development
Primary Package
Administration
5

2010 Retrospective
• Operating Challenges:
– Currency volatility
– Increased R&D spending on new product programs
– Manufacturing footprint: Phased reductions of operations at St. Austell,
UK and Montgomery, PA plants announced in Q4
UK and Montgomery, PA plants announced in Q4
• Nine-Months ended September 30, 2010
– Sales up 8.6% (9.4% excluding currency effects)
– Operating Profit up 11.9%
– Earnings per Diluted Share $1.70 vs $1.53
– Capex $49.8 million
• Q4, Full-year 2010 results to be announced February 17, 2011
• Second-half 2009 results yield difficult 2010 comparisons:
– $22 million of 2009 H1N1 Sales, of which $12 million were in Q4 ‘09
– Average $/Euro exchange of $1.45 in second half 2009, $1.48 in Q4 ’09
6

2011 Outlook
• Asymmetrical global economic recovery
– Europe, US, Japan remain sluggish
– China, India, Brazil stronger
– Continuing currencies, commodity price volatility
• New product approvals delayed
– Bydureon
– Benlysta
– Expansion of indications for approved drugs difficult (Avastin)
• Broader trends in Pharma/Device markets continue:
– Generic shift to India
– Pharma M&A - shift to large molecules
– Rising competitive and pricing pressures
• More demanding regulatory environment
• Anticipate revenue growth of 3-5%, excluding currency effects.
7

• Packaging Systems Segment
– Demographics and increasing prevalence of chronic disease
– Biologics
– Growth in Emerging Markets
– Increased access to Healthcare
– Escalating quality expectations: “Zero Defects”
• Delivery Systems Segment
– Glass compatibility/breakage issues
– Demand for combination products that promote safety, dosing accuracy,
ease of use, and deliver cost savings
ease of use, and deliver cost savings
Growth Drivers
8

• Packaging Systems (Pharm Systems) Segment
– The Global Quality Initiative
• Grow incremental value per unit - e.g WESTAR RU Launch
– Geographic expansion - capacity investments in Asia
– Improved operating efficiency: lean operations
– Strategic acquisitions and partnerships
• Delivery Systems (Tech Group) Segment
– Expand proprietary offerings
– Capitalize on emerging CZ opportunities
– Develop new platform opportunities - combination products
Growth Strategy
9

|
Category
|
Key Customers
|
Projected
Growth |
|
Diabetes
|
|
> 10 %
|
|
Oncology
|
|
> 10 %
|
|
Vaccines
|
|
> 10 %
|
|
Autoimmune
|
|
> 8%
|
IMS April 2010 Report; Business Insights 2009; GBI Research 2009
10
Therapeutic Category Growth Drivers

Traditional Pharmaceutical Delivery
West MixJect®
and Vial2Bag®
and Vial2Bag®
Prefilled Glass
Cartridge and
Pen
Cartridge and
Pen
Glass
Vials
and
Syringes
Vials
and
Syringes
11

High-Value Products Drive Growth
Westar® RS
State of the art
ready-to-sterilize
elastomer closures
Westar® RU
Sterile ready-to-
use syringe
plungers and
closures
use syringe
plungers and
closures
Envision™
Technically advanced,
automated vision
inspection system
NovaPure™
Unrivaled
Quality…by
design
12

Value Proposition
Proprietary
Products
Products
Revenue and
Margin
Opportunity
Margin
Opportunity
Disposable Device
Components
Components
Westar® RS
Mix2Vial®
NovaGuard™
éris™
Westar® RU
Standard
Components
Components
Consumer
Products
Products
Packaging
Delivery
13

Faster Growth of High-Value Products
Pharmaceutical Packaging Systems
Pharmaceutical Packaging Systems
14

Future Pharmaceutical Delivery
West
ConfiDose®
Auto-injector
System
ConfiDose®
Auto-injector
System
West
Electronic
Patch-injector
Electronic
Patch-injector
15

Integrated Solutions for Container, Administration
and Injection Systems
and Injection Systems
Daikyo Crystal Zenith®
Bulk Drug
Containers
Vials &
Plastic Caps
Plastic Caps
Insert Needle
& Luer Lock
Syringes
& Luer Lock
Syringes
Cartridges
Novel Devices
Administration Systems
Reconstitution
& Transfer
Systems
& Transfer
Systems
ConfiDose® and Eris™ are registered trademarks and trademarks of West Pharmaceutical Services, Inc. in the United States and other jurisdictions.
Daikyo Crystal Zenith® is a registered trademark of Daikyo Seiko, Ltd.
16
Electronic
Patch Injector
Patch Injector
Injection Systems
NovaGuard™
Needle Safety
Needle Safety
ConfiDose®
Auto-Injector
System
Auto-Injector
System
Eris™
Needle Safety
Needle Safety
Prototype
Intradermal
Adapter
Intradermal
Adapter

Concerns With Glass Syringes
• Interaction with sensitive biologics
• Protein aggregation (silicone oil)
• Residual chemicals (tungsten, glue)
• Glass flakes
• Breakage
• In process
• Within auto-injector systems
• Recent FDA recalls
• Dimensional variation
• Breakage in auto-injector systems
• Designed for manual injection
• Variable silicone distribution
• Amount of silicone coverage
• Age of barrel
• Inconsistent break force and
sustaining-force
sustaining-force
• Quality
• Cosmetic defects
• High levels of inspection necessary
• High “cost of quality”
Siliconized Glass Syringe
Crystal Zenith Syringe
17

2010 Glass-related Recalls
18

Electronic Patch Injector System
• Controlled, subcutaneous, micro-
infusion delivery of high volumes
and high viscosity drugs
infusion delivery of high volumes
and high viscosity drugs
• Prefilled cartridge, no need for
user filling
user filling
• Based on Daikyo CZ cartridge
• Compact
• Hidden needle for safety
• Single push-button operation
• Fully programmable
19
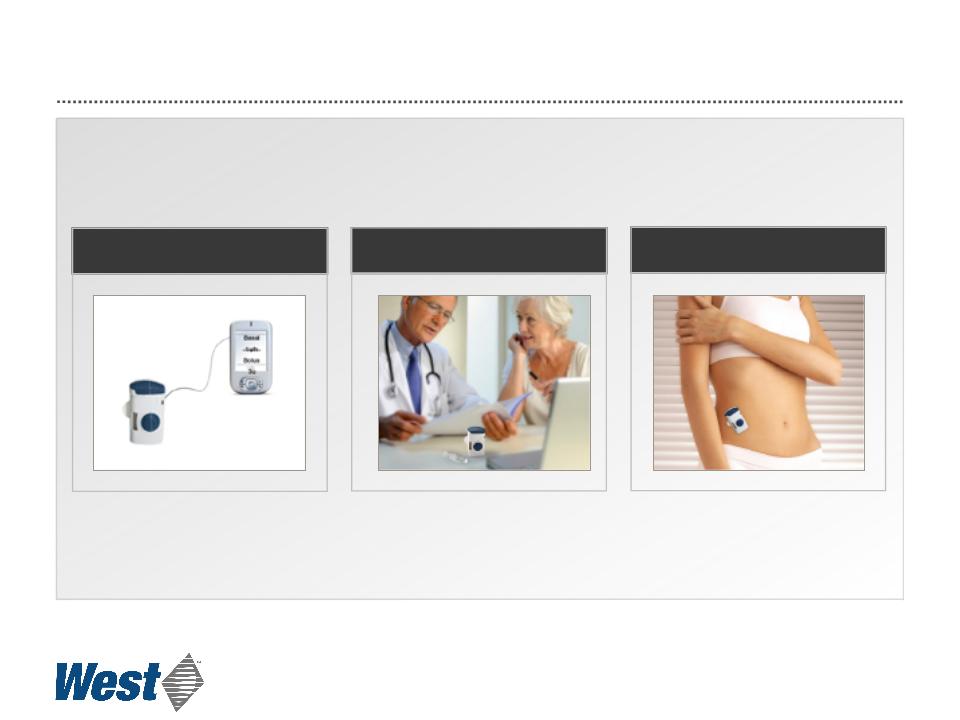
Prototype Electronic Patch Injector Operation
Programmed by PDA or PC
Dose may be customized
Attached and activated by patient
20

Our Long-Term Focus
• Pharmaceutical Packaging Systems
– Organic growth (on average) of 3-5% per year
– Margin expansion through improved operating efficiency, product mix
– Capital investments targeted at enhanced quality and value
• Pharmaceutical Delivery Systems
– Deliver the potential of Daikyo CZ products
– Increase healthcare-consumable contract manufacturing revenue
– Grow proprietary safety and delivery system businesses
• Financial discipline
– Operating cash flow: Discretionary SG&A, R&D and capital spending that
are supported by revenue growth.
are supported by revenue growth.
– Deliver returns on invested capital (“ROIC”) that regularly exceed weighted
average cost of capital (“WACC”).
average cost of capital (“WACC”).
– Align incentives with financial performance and value creation
21

Long-Term Growth Opportunity
Strategic Planning Goals:
• Projected 2014 sales of $0.6 billion
• Projected 2014 Operating margin: > 20%
$1.5 billion market for components with unit
growth 0% to 8% per year, depending on
product and therapeutic segment
growth 0% to 8% per year, depending on
product and therapeutic segment
Strategic Planning Goals:
• Projected 2014 sales of $1.0 billion
• Projected 2014 Operating margin: > 20%
|
Pharmaceutical
Packaging Systems
|
|
Primary Container Solutions
|
|
Pharmaceutical Delivery
Systems |
|
Administration Systems
|
Consolidated 2014 Planning Objectives
• 2014 Sales: $1.6 billion
• 2014 Operating Margin: 19%
• 2014 Consolidated ROIC: 17%
22

Summary
• Well positioned
– Substantial market share
– Proprietary technology
– Diversified customer base
– Global footprint
• Significant growth potential
– Strength in new product pipeline
– Preferred products for biologics
• The financial strength to invest
– Reliable operating cash flow
– Balance sheet strength
• 2010 Year-end call
– February 17, 2011 - 9:00AM
Injectable Container Solutions
Advanced
Injection
Systems
Injection
Systems
Prefillable Syringe Systems
Safety and Administration
Systems
Systems
23

WEST PHARMACEUTICAL SERVICES UPDATE
Solutions for Injectable Drug Delivery NYSE:WST www.westpharma.com
24
