Attached files
UNITED
STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
|
(Mark
One)
|
||
|
R
|
ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT
OF 1934.
|
For
the fiscal year ended December 31, 2009
Or
|
£
|
TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT
OF 1934.
|
For
the transition period from ________ to __________
Commission
file number 033-10893
China
Baicaotang Medicine Limited
(Exact
Name of Registrant as Specified in Its Charter)
|
Delaware
|
20-8067060
|
|
|
(State
or Other Jurisdiction
of
Incorporation or Organization)
|
(I.R.S.
Employer
Identification
No.)
|
No.
102, Chengzhan Road
Liuzhou
City, Guangxi Province, P.R.C. 545007
(Address
of Principal Executive Offices) (Zip Code)
Registrant’s
telephone number, including area code: Tel.: (86) 772-363-8318
Securities
registered pursuant to Section 12(b) of the
Act: None
Securities
registered pursuant to Section 12(g) of the Act: Common Stock,
par value $0.001 per share
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act. Yes £
No R
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Act. Yes R
No £
Indicate
by check mark whether the registrant: (1) has filed all reports required to
be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934
during the preceding 12 months (or for such shorter period that the
registrant was required to file such reports), and (2) has been subject to
such filing requirements for the past 90 days. Yes R
No £
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K (§ 229.405 of this chapter) is not contained herein,
and will not be contained, to the best of registrant’s knowledge, in definitive
proxy or information statements incorporated by reference in Part III of
this Form 10-K or any amendment to this Form 10-K. R
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting company. See
the definitions of “large accelerated filer,” “accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act. (Check
one):
|
Large
accelerated filer £
|
Accelerated
filer £
|
Non-accelerated
filer £
|
Smaller
reporting company R
|
Indicate
by check mark whether the registrant is a shell company (as defined in Exchange
Act Rule 12b-2). Yes £
No R
The
aggregate market value of the registrant’s common stock, $0.001 par value
per share, held by non-affiliates of the registrant on June 30, 2009, was
approximately $0 (based on the fact that prior to March 2010 there has been no
trading of the registrant’s common stock on the Over the Counter Bulletin
Board). Shares of the registrant’s common stock held by each officer and
director and each person known to the registrant to own 10% or more of the
outstanding voting power of the registrant have been excluded in that such
persons may be deemed to be affiliates. This determination of affiliate status
is not a determination for other purposes.
As of
March 30, 2009 there were 38,154,340 shares of common stock
outstanding.
DOCUMENTS
INCORPORATED BY REFERENCE: None
CHINA
BAICAOTANG MEDICINE LIMITED
ANNUAL
REPORT ON FORM 10-K
FOR
THE FISCAL YEAR ENDED DECEMBER 31, 2009
|
PART
I
|
1
|
|
|
Business
|
1
|
|
|
Risk
Factors
|
23
|
|
|
Unresolved
Staff Comments
|
37
|
|
|
Properties
|
37
|
|
|
Legal
Proceedings
|
38
|
|
|
Submission
of Matters to a Vote of Security Holders
|
39
|
|
|
PART
II
|
40
|
|
|
Market
for Common Equity, Related Stockholder Matters and Issuer
Purchases of Equity Securities
|
40
|
|
|
Selected
Consolidated Financial Data
|
40
|
|
|
Management’s
Discussion and Analysis of Financial Condition and Results of
Operations
|
42
|
|
|
Quantitative
and Qualitative Disclosures about Market Risk
|
46
|
|
|
Financial
Statements and Supplementary Data
|
46
|
|
|
Changes
in and Disagreements with Accountants on Accounting and Financial
Disclosure
|
47
|
|
|
Controls
and Procedures
|
47
|
|
|
Other
Information
|
48
|
|
|
PART
III
|
49
|
|
|
Directors
and Executive Officers and Corporate Governance
|
49
|
|
|
Executive
Compensation
|
51
|
|
|
Security
Ownership of Certain Beneficial Owners and Management and Related
Stockholder Matters
|
53
|
|
|
Certain
Relationships and Related Transactions, and Director
Independence
|
54
|
|
|
Principal
Accounting Fees and Services
|
55
|
|
|
PART
IV
|
56
|
|
|
Exhibits,
Financial Statement Schedules
|
56
|
|
CAUTIONARY
STATEMENT
This
prospectus contains forward-looking statements. Forward-looking statements give
our current expectations or forecasts of future events. You can identify these
statements by the fact that they do not relate strictly to historical or current
facts. Forward-looking statements involve risks and uncertainties.
Forward-looking statements include statements regarding, among other things, (a)
our projected sales, profitability and cash flows, (b) our growth strategies,
(c) anticipated trends in our industries, (d) our future financing plans and (e)
our anticipated needs for working capital. They are generally identifiable by
use of the words “may,” “will,” “should,” “anticipate,” “estimate,” “plan,”
“potential,” “projects,” “continuing,” “ongoing,” “expects,” “management
believes,” “we believe,” “we intend” or the negative of these words or other
variations on these words or comparable terminology. In particular, these
include statements relating to future actions, future performance, sales
efforts, expenses, the outcome of contingencies such as legal proceedings, and
financial results.
Any or
all of our forward-looking statements in this prospectus may turn out to be
inaccurate. They can be affected by inaccurate assumptions we might make or by
known or unknown risks or uncertainties. Consequently, no forward-looking
statement can be guaranteed. Actual future results may vary materially as a
result of various factors, including, without limitation, the risks outlined
under “Risk Factors” and matters described in this prospectus generally. In
light of these risks and uncertainties, there can be no assurance that the
forward-looking statements contained in this filing will in fact occur and you
should not place undue reliance on these forward-looking
statements.
PART
I
References
in this prospectus to the “PRC” or “China” are to the People’s Republic of
China. Except as otherwise specifically stated or unless the context otherwise
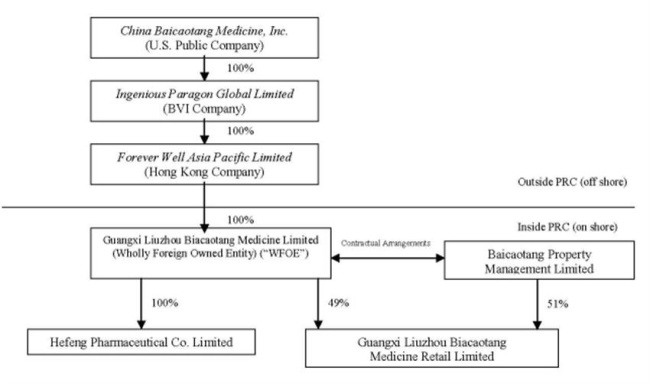
requires, the terms “Company,” “we,” “us” and “our” refer to collectively (i)
China Baicaotang Medicine Limited, a corporation incorporated in the State of
Delaware; (ii) Ingenious Paragon Global Limited (“Ingenious”), a British Virgin
Islands company which is a wholly-owned subsidiary; (iii) Forever Well Asia
Pacific Limited (“Forever Well”), a Hong Kong company which is a wholly-owned
subsidiary of Ingenious; (iv) Guangxi Liuzhou Baicaotang Medicine Limited
(“Liuzhou BCT”), a PRC wholly foreign-owned enterprise
(“WFOE”) which is a wholly-owned subsidiary of Forever Well; (v)
Hefeng Pharmaceutical Co. Limited (“Hefeng Pharmaceutical”), a PRC company which
is a wholly-owned subsidiary of Liuzhou BCT; and (vi) Guangxi Liuzhou Baicaotang
Medicine Retail Limited (“BCT Retail”), a PRC company of which 49% of its
registered capital was contributed by Liuzhou BCT and 51% of its registered
capital was contributed by Baicaotang Property Management Limited (“Property
Management”), an affiliated company.
References
to BCT Retail’s “registered capital” are to the equity of BCT Retail, which
under PRC law is measured not in terms of shares owned but in terms of the
amount of capital that has been or will be contributed to a company by a
particular shareholder or all shareholders. The portion of a limited liability
company’s total capital contributed by a particular shareholder represents that
shareholder’s ownership of the company and the total amount of capital
contributed by all shareholders is the company’s total equity. Capital
contributions are made to a company by deposits into a dedicated account in the
company’s name, which the company may access in order to meet its financial
needs. When a company’s accountant certifies to PRC authorities that a capital
contribution has been made and the company has received the necessary government
permission to increase its contributed capital, the capital contribution is
registered with regulatory authorities and becomes a part of the company’s
“registered capital”.
Summary
We are
engaged in pharmaceutical distribution, retail pharmacy and manufacture of
pharmaceuticals and medical-related products through our three subsidiaries
Liuzhou BCT, Hefeng Pharmaceutical, and BCT Retail, each of which is located in
Guangxi Province, China.
We have
integrated operations in the following business segments:
|
·
|
Pharmaceutical
distribution.
|
Pharmaceutical
distribution is our principal business. We conduct our wholesale business
through Liuzhou BCT by purchasing from pharmaceutical product suppliers and then
distributing the products to our wholesale customers, including hospitals,
retail drug stores, other pharmaceutical wholesalers, clinics, medical centers,
and individuals. Our pharmaceutical distribution business is focused on the
market of Guangxi province, which includes major cities such as Nanning, Liuzhou
and Guilin, and which has approximately 50 million people. According
to a Report from the Guanxi
Tax Bureau there is an annual market demand for pharmaceutical
distributors in Guangxi province of approximately $4 billion per
year. We believe that we operate one of the largest regional
wholesale networks in Guangxi province supported by strategically placed
warehouse facilities.
We
distribute over 8,000 products from nearly 4,000 suppliers through our wholesale
distribution in compliance with applicable PRC
regulations. Hefeng Pharmaceutical, which is one of our wholly
owned subsidiaries, is also one of our suppliers.
PRC rules
and regulations require most public hospitals and healthcare institutions to
purchase medicines from pharmaceutical distributors through a centralized
tendering process, which includes the implementation of government-mandated
price controls. The manufacturers of provincial catalog medicines that are on
the hospitals’ formularies are invited to bid and participate in the centralized
tendering process, which they must do so directly. The bidding process covers
multiple categories of medicines used by the hospitals. A duly organized
committee of pharmaceutical and clinical medical experts is responsible for bid
evaluations. Selection is based on a number of factors, including bid price,
quality, clinical effectiveness, and manufacturer’s reputation and service. The
supply of a particular type of medicine is generally made on a non-exclusive
basis by multiple manufacturers and distributors. We typically advise and assist
pharmaceutical manufacturers in the hospital tendering process and distribute
products of pharmaceutical manufacturers upon purchase orders being made by the
hospitals after the bidding process.
The
Guangxi centralized-online tendering system was started in 2006, and in
2009 the tendering started to be applied also under the New Rural Co-operation
Health Insurance Plan. At the first tendering in 2009 we were awarded
distribution rights for six cities and towns under the New Rural Co-operation
Health Insurance Plan, including Liuzhou, Yizhou, Lipu, Gongchen, Luzhai Laibin
and Heshan, and were selected as one of two exclusive distributors for these six
cities.
|
·
|
Retail
pharmacy.
|
Established
in 2001, BCT Retail operates what we believe to be the largest regional retail
network in Guangxi province, consisting of 66 directly owned retail stores in
Guangxi province under the registered name “Baicaotang 百草堂.” Our retail
stores provide convenient, high quality and professional pharmaceutical services
and supply a wide variety of medicines, including western medicine, traditional
Chinese medicine (“TCM”), dried Chinese herbal medicine, roughly processed
Chinese herbal medicine, family planning products, and seasonal medicine. Among
the 66 stores, there are 16 stores that are medi-care qualified stores, where
customers are able to make their purchases either by cash or by using their
medi-care insurance card for payment.
|
·
|
Hefeng
Pharmaceutical
|
Hefeng
Pharmaceutical has a manufacturing facility on approximately 40,000 square
meters of land and manufactures four products units:
|
·
|
A
Chinese herbal medicine abstraction unit for raw material and medicine
paste with 100 tons of annual abstraction
capacity;
|
|
·
|
A
granular formulation unit with an annual production capacity of 2 billion
packages;
|
|
·
|
A
pill formulation unit with an annual production capacity of 3 billion
pills, and
|
|
·
|
A
liquid formulation unit with an annual production capacity of 0.1 billion
injections.
|
Hefeng
Pharmaceutical produces and sells pharmaceutical products under the registered
name “Asio (亚太)” including:
traditional anti-inflammatory and antibacterial drugs, cancer treatment drugs,
cardio-vascular disease drugs and hepatitis drugs. Hefeng Pharmaceutical’s
best-selling products include:
|
·
|
Tabellae
Sarcandrae, a TCM drug that has similar anti-inflammatory and
antibacterial effects as anti-biotics in Western
medicine;
|
|
·
|
Corydalis
Saxicola Bunting (Yanhuanglian), an important component in various
prescriptions in TCM;
|
|
·
|
Hydroxycamptotbecine
Injection, which is used to treat cancers such as carcinoma ventricui,
carcinoma hepatitis and colon
cancer;
|
|
·
|
Ethacridine
Lactate Injection which is used for second trimester pregnancy termination
from week 12-26; and
|
|
·
|
Levodopa,
a TCM drug that is used to treat stiffness, tremors, spasms and poor
muscle control related to Parkinson’s
disease.
|
In
addition, Hefeng Pharmaceutical collaborates with several renowned medical
research universities in China to continuously improve its raw material
abstraction efficiency and production process, and to develop alternative
formulas for existing drugs.
Corporate
Structure and History
We were
originally incorporated in the State of Delaware on November 30, 2006 under the
name Purden Lake Resource Corp. to engage in the acquisition, exploration and
development of natural resource properties. Prior to December 23, 2009 we were a
blank check company with nominal assets. We changed our name to China
Baicaotang Medicine Limited on December 24, 2009.
Corporate
Structure
Our
wholly-owned subsidiary, Ingenious is a British Virgin Islands corporation that
owns 100% of the issued and outstanding capital stock of Forever Well, a Hong
Kong company. Forever Well is the sole shareholder of Liuzhou BCT, a
PRC wholly foreign-owned enterprise. Liuzhou BCT contributed 100% of
the registered capital of Hefeng Pharmaceutical and 49% of the registered
capital of BCT Retail. The remaining 51% of the registered capital of BCT Retail
was contributed by Property Management, an affiliate of Liuzhou
BCT.
We do not
have direct 100% ownership interest in BCT Retail due to the restriction of
foreign investment in pharmacy chains with more than 30
drugstores. We have entered into contractual arrangements with
Property Management pursuant to which we loaned money equal to 51% of the
registered capital of BCT Retail to the shareholders of Property Management and
such Property Management pledged their 51% equity interests in BCT
Retail.
The chart
below illustrates the current structure of the Company:
Reorganization
In 2008,
the shareholders of Liuzhou BCT (the “Liuzhou BCT Shareholders”) and Xiaoyan
Zhang, our CFO, developed a restructuring plan (the
“Restructuring”). At that time, Ms. Zhang, who is a citizen of Hong
Kong, was the sole shareholder of Ingenious, which was the parent company of
Forever Well. The first step was for Forever Well to acquire 100% of
the equity interests of Liuzhou BCT and its subsidiaries (the “PRC Operating
Companies”). Liuzhou BCT was owned at that time by certain former and
current employees and directors of Liuzhou BCT. After the acquisition
of the PRC Operating Companies by Forever Well was consummated, the second step
was for Ingenious to enter into and complete a transaction with a U.S. public
reporting company whereby that company would acquire Ingenious.
The
acquisition of Liuzhou BCT was structured to comply with the PRC M&A
Laws. Under the PRC M&A Laws, the acquisition of PRC companies by
foreign companies that are controlled by PRC citizens who are affiliated with
the PRC companies is strictly regulated and requires approval from the Ministry
of Commerce, which approval is burdensome to obtain. Such
restrictions however, do not apply to foreign entities which are controlled by
foreign persons. So as not to violate the PRC M&A Laws, in August
2008 Forever Well acquired 100% of the equity interest of Liuzhou BCT from the
Liuzhou BCT Shareholders. Since PRC M&A Laws would have
prohibited Liuzhou BCT Shareholders who were PRC citizens from immediately
receiving a controlling interest in Ingenious in a share exchange as
consideration for the sale of their interest in Liuzhou BCT, Liuzhou BCT
Shareholders holding a majority of the equity interest in Liuzhou BCT and Ms.
Zhang instead agreed that they would enter into an Earn-In Agreement to grant
those Liuzhou BCT Shareholders a call right to acquire Ms. Zhang’s interest in
Ingenious (or its public parent company as the case may be) after the
acquisition of Liuzhou BCT was consummated in compliance with PRC
law. The Earn-In Agreement was entered into by Ms. Zhang and certain
Liuzhou BCT Shareholders on December 30, 2009 and enables those Liuzhou BCT
Shareholders to purchase shares of Ingenious (or its public parent company) from
Ms. Zhang for a nominal amount per share provided that we meet certain
performance targets for fiscal 2010 and 2011. The number of shares
which can be purchased by the Liuzhou BCT Shareholders from Ms. Zhang is in
proportion to their former relative ownership interest in Liuzhou
BCT.
On
December 30, 2009, the goal of the Restructuring was realized when we entered
into a share exchange agreement with Ingenious, pursuant to which we acquired
100% of the equity of Ingenious in exchange for the issuance of an aggregate of
32,000,000 shares of our common stock to Ms. Zhang and to certain former Liuzhou
BCT Shareholders. As of the date of this prospectus, Ms. Zhang owns
58.9% of our common stock. As a result of this transaction, we are a
holding company which, through our direct and indirect ownership of Ingenious,
Forever Well, Liuzhou BCT, Hefeng Pharmaceutical, and BCT Retail, now has
operations based in the PRC.
Private
Placement
On
October 23, 2009, we entered into a subscription agreement (the “Subscription
Agreement”) with certain investors (the “Investors”) for the sale of up to an
aggregate of 1,147 units (the “Units”) in a private placement (the “Private
Placement”). Each Unit consists of (i) 3,937 shares of our common
stock, and (ii) a warrant to purchase 1,968 shares of our common stock at an
exercise price of $3.81 per share (the “Investor
Warrant”). Simultaneously with the closing of the Share Exchange, we
completed the initial closing of the Private Placement of $6,323,002
or 632.3 Units, (the “Initial Closing”). Upon the Initial
Closing, we issued an aggregate of 2,489,370 shares of our common stock and
warrants exercisable for up to 1,244,368 shares of our common stock at an
exercise price of $3.81 per share. In addition, in connection with the initial
closing of the Private Placement, we issued Agent Warrants to the placement
agents (the “Co-Placement Agents”) that are exercisable for 248,937 shares of
our common stock at an exercise price of $3.05 per share.
On
February 1, 2010, we completed the second closing of the Private Placement of
$2,616,108 (the “Second Closing”) of 261.61 Units, consisting of an aggregate of
1,029,970 shares of our common stock and warrants exercisable for 514,933 shares
of our common stock at an exercise price of $3.81. In connection with
Second Closing, we issued Placement Agent Warrants to the Co-Placement Agents
that are exercisable for 102,997 shares of our common stock at an exercise price
of $3.05 per share on a cash or cashless basis.
Where
You Can Find Us
Our
principal executive office is located at No. 102 Chengzhan Road, Liuzhou City,
Guangxi province, China and our telephone number is (86) 772-363-8318. Our corporate website is
www.china-bct.com.
Information contained on, or accessed through our website is not intended to
constitute and shall not be deemed to constitute part of this
prospectus.
Overview
We are
engaged in pharmaceutical distribution, retail pharmacy and manufacture of
pharmaceuticals and medical-related products through our two wholly-owned
subsidiaries Liuzhou BCT and Hefeng Pharmaceutical, and BCT Retail, a retail
company that we control through a series of contractual arrangements, each of
which is located in Guangxi province, China.
|
·
|
Pharmaceutical
distribution
|
Pharmaceutical
distribution is our principal business. We conduct our wholesale
business through Liuzhou BCT by purchasing pharmaceutical products from
suppliers and then distributing them to our wholesale customers, including
hospitals, retail drug stores, other pharmaceutical wholesalers, clinics,
medical centers, and individuals. Our pharmaceutical distribution business is
focused on the market of Guangxi province, which includes major cities such as
Nanning, Liuzhou and Guilin and which has a population of approximately 50
million people. According to a Report from the Guanxi Tax
Bureau there is an annual market demand for pharmaceutical distributors
in Guangxi province of approximately $4 billion per year. We believe
that we operate one of the largest regional wholesale networks in Guanxi
Province supported by strategically placed warehouse facilities.
We
distribute over 8,000 products from nearly 4,000 suppliers through our wholesale
distribution in compliance with PRC regulations. Hefeng
Pharmaceutical which is one of our wholly-owned subsidiaries is also one of our
suppliers. Liuzhou BCT’s GSP Certificate, which is a certification that
drugstores in China are required to obtain was issued on September 18, 2009 and
will expire on September 17, 2013, subject to renewal of an additional five-year
term.
|
·
|
Retail
Business
|
Established
in 2001, BCT Retail operates the largest regional retail network in Guangxi
province, consisting of 66 directly owned retail stores in Guangxi province
under the registered name “Baicaotang 百草堂.” BCT
Retail’s GSP Certificate was issued on February 18, 2009 and will expire on
February 17, 2010, subject to renewal of additional five-year term. Our retail
stores provide convenient, high quality and professional pharmaceutical
services, and supply a wide variety of medicines, including western medicine,
TCM, dried Chinese herbal medicine, roughly processed Chinese herbal medicine,
family planning products, and seasonal medicine. Among the 66 stores,
there are 16 stores that are medi-care qualified stores, where customers are
able to make their purchase either by cash or by using their medi-care insurance
card for payment.
|
·
|
Hefeng
Pharmaceutical
|
Hefeng
Pharmaceutical has a manufacturing facility located on approximately 40,000
square meters of land, and manufactures four products units:
|
·
|
A
Chinese herbal medicine abstraction unit for raw material and medicine
paste with 100 tons of annual abstraction
capacity;
|
|
·
|
A
granular formulation unit with an annual production capacity of 2 billion
packages;
|
|
·
|
A
pill formulation unit with an annual production capacity of 3 billion
pills; and
|
|
·
|
A
liquid formulation unit with an annual production capacity of 0.1 billion
injections.
|
Hefeng
Pharmaceutical produces and sells pharmaceutical products under the registered
name “Asio (亚太)” including:
traditional anti-inflammatory and antibacterial drugs, cancer treatment drugs,
cardio-vascular disease drugs and hepatitis drugs.
Hefeng
Pharmaceutical’s best-selling products include:
|
·
|
Tabellae
Sarcandrae, a TCM drug that has similar anti-inflammatory and
antibacterial effects as anti-biotics in Western
medicine;
|
|
·
|
Corydalis
Saxicola Bunting (Yanhuanglian), an important component in various
prescriptions in TCM;
|
|
·
|
Hydroxycamptotbecine
Injection; which is used to treat cancers such as esophagus cancer,
carcinoma ventriculi, carcinoma hepatis and colon
cancer.
|
|
·
|
Yinge
Tongmai Tea, which is made of gingko biloba, kudzu rot and Chinese tea and
is used to clear up blood vessels and treat cardio-vascular diseases.;
and
|
|
·
|
Levodopa,
a TCM drug that is used to treat stiffness, tremors, spasms and poor
muscle control related to Parkinson’s
disease.
|
In
addition, Hefeng Pharmaceutical operates a raw material plantation base,
collaborating with several renowned medical research universities in China to
continuously improve its raw material abstraction efficiency and production
process, and to develop alternative formula for existing drugs.
Hefeng
Pharmaceutical’s GMP Certificate was issued on July 14, 2009 and will expire on
July 13, 2010, and its GMP Certificate for Small Volume Parental Solution was
issued on July 10, 2006 and will expire on July 9, 2011, both of which will be
subject to renewal of additional five-year term.
The table
below sets forth a breakdown of our external segment revenue after elimination
of inter-segment sales, and each segment revenue item as a percentage of our
total revenue, as well as our inter-segment sales for the year ended December
31, 2009 and December 31, 2008. For the year ended December 31, 2009,
we had approximately $22.5 million of inter-segment revenue, which includes
approximately $21.7 million in sales from our pharmaceutical distribution
segment to our retail pharmacy segment, and approximately $762,000 in sales from
our manufacturing pharmacy segment to our retail pharmacy
segment External segment revenue refers to segment revenue
after inter-segment elimination.
|
December
31
|
||||||||
|
2009
|
2008
|
|||||||
|
External
Segment revenue
|
’000
|
%
|
’000
|
%
|
||||
|
Pharmaceutical
distribution
|
$
|
97,137
|
71.4
|
$
|
72,807
|
66.8
|
||
|
Retail
pharmacy
|
31,223
|
22.9
|
28,593
|
26.2
|
||||
|
Manufacturing
pharmacy
|
7,727
|
5.7
|
7,592
|
7
|
||||
|
136,087
|
100.0
|
108,992
|
100.0
|
|||||
|
Inter-segment
revenue eliminated
|
22,492
|
N/A
|
21,656
|
N/A
|
||||
Our
Products
Products
Offered by Hefeng Pharmaceutical
Manufacturing
both Chinese medicine and Western medicine, Hefeng Pharmaceutical maintains
valid production licenses for 76 drugs. Below is the description of the five (5)
best-selling drugs.
|
·
|
Corydalis
Saxicola Bunting (Yanhuanglian)
|
|
Corydalis
Saxicola Bunting is an important component in various prescriptions in
TCM. Yanhuanglian has been demonstrated to possess many pharmacological
activities, including antibacterial, antiviral and anticancer activities.
The active fractions are dehydrocavidine, coptisine, dehydroapocavidine
and tetradehydroscoulerine. Systemic clearance of the four active
alkaloids in plasma was over 93% of hepatic blood flow, indicating they
may be quickly eliminated via hepatic clearance. Less than 10% of the drug
was excreted via urine following intravenous and oral administration,
suggesting that these four alkaloids may undergo significant metabolism in
the body or the drug may be excreted via other routes other than urine.
Intravenous administration of Yanhuanglian is the most common clinical
practice, because it can improve absorption of the four active alkaloids
into systemic circulation.
|
 |
We are
the sole licensed producer for this drug in China, and the market demand for
this drug has been extremely strong due to the effectiveness of the drug to
treat hepatic diseases.
|
·
|
Tabellae
Sarcandrae
|
|
Tabellae
Sarcandrae, a TCM protected drug, has similar anti-inflammatory and
antibacterial effects as anti-biotics in Western medicine. Tabellae
Sarcandrae possessed marked inhibition effect to auricular inflammation in
mice caused by croton oil, footpad inflammation in rats caused by
carragheenin and granuloma in mice by cotton ball. It could also relieve
obviously abdominal pain caused by acetic acid and inhibit bacterial
growth. The TCM protection is valid from December 19,
2006 to August 1, 2012 and is renwable.
|
 |
|
·
|
Hydroxycamptotbecine
Injection
|
|
Hydroxycamptotbecine
Injection is used to treat cancers such as esophagus cancer, carcinoma
ventriculi, carcinoma hepatis and colon cancer. We are one of three
licensed producers of this drug in China.
|
 |
|
·
|
Levodopa
|
|
Levodopa
is used to treat the stiffness, tremors, spasms, and poor muscle control
of Parkinson’s disease. Levodopa is also used to treat these same muscular
conditions when they are caused by drugs such as chlorpromazine
(Thorazine), fluphenazine (Prolixin), perphenazine (Trilafon), and others.
As traditional Chinese medicine has become more and more popular
worldwide, especially in South-east Asian countries whose cultures are
similar to that of the Chinese, we have exported roughly processed
Levodopa raw material to Japan and Thailand.
|
 |
|
·
|
Ethacridine
Lactate Injection
|
|
This
family planning drug very popular in China; it’s used for
second trimester pregnancy termination from week 12-26 at
hospitals. We are one of three licensed producers in
China.
|
 |
Products
and services offered by Retail Chain
Retail
Chain provides our customers with high-quality, professional and convenient
pharmaceutical services and supplies of a wide variety of medicines, including
Western medicine, TCM, raw materials of dried herbal products, roughly processed
herbal medicine, family planning products, as well as convenient seasonal and
promotional items. A typical retail drug store of Retail Chain
carries approximately 2,800 to 3,200 different products. Management regularly
reviews and refines the product selection in order to respond to change in
demographics, lifestyles, shopping habits and product preferences of our
customers.
Our
product selection is designed to offer choices and convenience to our customers
and to achieve high gross margins for us. We offer our customers a broader range
of choices in two respects. First, we offer a wide range of complementary
products in each therapeutic category so that customers have more choices to
suit their needs. For example, a customer looking for a cough remedy will be
able to find a wide variety of choices including different OTC drugs,
nutritional supplements and herbal products. Second, for products with the same
therapeutic purpose, we offer choices in each of the high, medium and low price
ranges to suit the needs of customers with different spending
power.
|
·
|
Packaged Western and
TCM. We offer approximately 2,750 packaged drugs including
prescription and OTC drugs. We accept prescriptions only from licensed
healthcare providers and do not prescribe medications or otherwise
practice medicine. Our in-store pharmacists verify the validity, accuracy
and completeness of all prescription drug orders. We ask all prescription
drug customers to provide us with information regarding drug allergies,
current medical conditions and current
medications.
|
|
·
|
Chinese Herbal
Medicine. We
offer approximately 450 types of various drinkable herbal remedies and
packages of assorted herbs for making soup, which are used by consumers as
health supplements. Herbal products typically have higher margins than
prescription and OTC drugs.
|
|
·
|
Family Planning
Products. We offer approximately 40 family planning products, which
include family care products such as portable medical devices for family
use, birth control and early pregnancy test products and convenience
products. Our family planning products also include seasonal and
promotional items tailored to local consumer demand for convenience and
quality. We believe offering these products increases customer visits by
increasing the shopping convenience for our
customers.
|
Products
offered by our Pharmaceutical Distribution Segment
Our
pharmaceutical distribution segment provides Retail Chain with the majority of
the pharmaceutical products sold in retail drugstores. Approximately 95% of the
packaged Western medicine and TCM, 100% of the Chinese Herbal Medicine and 100%
of the family planning products sold by Retail Chain are supplied by our
pharmaceutical distribtion segment.
Besides
providing procurement to our retail business, the majority of the sales revenue
of our pharmaceutical distribution segment arises from supplying pharmaceutical
products to hospitals, clinics and healthcare centers at provincial, city,
county and district levels. Further, our pharmaceutical distribution
segment also distributes our products to other retail networks.
Increasing
coverage of social medical insurance in China
The
National Medical Insurance Program (“National Program”), which was introduced in
1999, is the largest medical insurance program in China. The National Program is
funded with varying levels of contributions from the PRC Government, individual
program participants and their employers. In 1999, the National Program was
originally launched as the Urban Worker Basic Medical Insurance Program (“Urban
Worker Program”), a mandatory scheme covering urban workers and their minor
children. In 2007, a voluntary component called the Urban Resident Basic Medical
Insurance Program (“Urban Resident Program”) was further implemented as part of
the National Program, to cover the rest of the urban residents that are not
covered by the Urban Workers Program. The National Program provides guidance on
which prescription and over-the-counter medicines are included in the program
and to what extent the purchases of these medicines are
reimbursable.
We
believe that only a small percentage of the Chinese population can afford
commercial insurance plans. Therefore, the National Program coverage is expected
to expand in the future. According to the PRC National Bureau of Statistics, the
percentage of PRC urban residents grew from approximately 37.7% of the total
population to 44.9% from 2001 to 2007. The number of people covered by the
National Program increased from approximately 37.9 million in 2000 to 180.2
million in 2007, representing an CAGR of 25%. This trend is anticipated to
continue as the Eleventh Five-Year Plan projects that the PRC urban population
will increase from 45% to 47% of China’s total population between 2007 and 2010.
Furthermore, the provincial and municipal authorities who are responsible for
administering social medical insurance funds to cover such reimbursements have
been gradually increasing funding in recent years. According to the PRC Ministry
of Labor and Social Security, total funding under the national insurance program
reached RMB225.7 billion, or $28.9 billion, in 2008, representing an increase of
29.2% from 2007. The availability of funding is expected to increase
significantly in the near future, primarily as a result of increased financial
and policy support from various levels of the PRC Government.
Increasing
access to healthcare in rural areas
At the
fifth meeting of the tenth National People’s Congress held in March 2007, the
PRC Government announced its goal to accelerate the reform and development of
healthcare services in the PRC and focus on building a basic healthcare system
that covers both rural and urban areas. The PRC Government’s plans include
providing expanded healthcare services for its rural citizens and establishing
comprehensive community healthcare service centers that would provide basic
medical treatment and pharmaceutical services, as well as upgrading existing
class-two hospitals and state owned medical facilities. The public health
service centers would be allocated based on demand and population. In addition,
the PRC Government has actively promoted the implementation of the New Rural
Cooperative Medical Insurance Scheme (“New Rural Insurance Scheme”), which seeks
to provide healthcare services to the vast rural areas of China. The program
extends to cover approximately 2,729 counties in the PRC, which account for
95.4% of the total number of counties in the PRC. In addition, the program
covers approximately 814 million rural residents, which accounts for
approximately 91.5% of the total population engaged in the agricultural industry
in China as of December 31, 2008.
PRC
Healthcare Reform Plan
In
September 2008, the State Council published a draft plan to ease the
difficulties and minimize the costs for PRC citizens to obtain proper healthcare
treatment. On 17 March 2009, the PRC Government issued the Opinion on Deepening
the Healthcare System Reform (the “Opinion”). The State Council subsequently
released the Notice on Important Implementing Plans for the Healthcare System
Reform 2009-2011 (the “Implementing Plan”). The goal of the healthcare reform
plan is to establish a basic, universal healthcare framework to provide Chinese
citizens with safe, efficient, convenient and affordable healthcare. The Opinion
calls for healthcare reform to be carried out in two steps:
|
·
|
Step
One, which will be completed by 2011, aims to increase the accessibility
while reducing the cost of healthcare. During this phase, the PRC
Government will build up a network of basic healthcare facilities, expand
coverage of the public medical insurance system to cover 90% or more of
the population, and reform the drug supply and public hospital
system.
|
|
·
|
Step
Two, which will take place between 2011 and 2020, envisions the
establishment of a universal healthcare system. The entire population
should be covered by public medical insurance; drugs and medical services
should be accessible and affordable to citizens in all public healthcare
facilities.
|
While the
PRC Government has neither provided a concrete timetable nor steps to implement
certain tasks, such as the public hospital reform, it has released execution
guidance for other tasks. Most notably, the PRC Government has announced it will
spend an additional approximately RMB 850 billion, or $125 billion from 2009 to
2011 on the healthcare industry. A significant portion will be expended to
establish a basic healthcare medical insurance regime, which aims to cover over
90% of the national population by 2011, mainly through the Urban Worker Program,
Urban Resident Program and the New Rural Insurance Scheme. The PRC Government
further announced that the annual subsidy for each participant will be increased
from approximately RMB 40, or $5.90 to approximately RMB120, or
$17.60 for Urban Resident Program participants, and from approximately RMB 80,
or $11.76 to approximately RMB120 RMB, or $17.60 for New Rural Insurance Scheme
participants, starting from 2010. The reform plan will also raise the cap on
claim payments from four times the local average annual income to six times such
income. Another significant part of the spending plan focuses on healthcare
facilities. The PRC Government plans to build 29,000 rural clinics in 2009. In
the next three years, the PRC government plans to build an additional 5,000
rural clinics, 2,000 county-level hospitals and 2,400 urban community clinics in
under-developed areas. This substantial increase in healthcare spending is
expected to expedite the growth of the healthcare industry in
China.
Under the
healthcare reform plan, the additional funding for the healthcare industry will
primarily target four fundamental healthcare systems in China:
|
·
|
The
public health services system. This system focuses on preventing disease
and promoting health as a complementary alternative to medical treatment.
The public health services system will provide services such as
immunizations, regular physical check-ups (for senior citizens over 65
years of age and children under three years of age), pre-natal and
post-natal check-ups for women, prevention of infectious or chronic
diseases and other preventative and fitness
activities.
|
|
·
|
The
public medical insurance system. This system covers drugs and medical
treatments for the majority of the population. The healthcare reform plan
will retain the framework of the current public medical insurance schemes
under the National Program, but will expand them to cover more of the
population and increase the scope of treatments, raise the cap on claim
payments and cover more claims at higher
percentages.
|
|
·
|
The
public healthcare delivery system. One of the primary goals of the
Implementing Plan is to build more healthcare facilities and to improve
the training of healthcare professionals. Beyond additional public
wellness centers, the reform plan aims to place a medical clinic in every
village and a hospital in every county by 2011. In addition, the PRC
Government will encourage private investors to establish public non-profit
hospitals.
|
|
·
|
The
drug supply system. This system regulates pricing and how drugs will be
procured prescribed and dispensed in healthcare facilities. The healthcare
reform plan will focus on pricing, procurement, prescription and
dispensing of essential drugs.
|
The
Opinion and the Implementing Plan direct relevant governmental authorities,
including the Ministry of Health, SFDA and the National Development Reform
Commission, or NDRC, to adopt implementing regulations for the reforms outlined
in the healthcare reform plan. Although the healthcare reform plan is expected
to benefit our pharmaceutical distribution, retail pharmacy and other business
operations and improve our competitive position, the full effect of the
healthcare reform plan on our operations is as yet unclear.
Industry
Overview
We
operate in the large and growing pharmaceutical wholesale and retail industry in
China, which we believe offers compelling industry fundamentals and benefits
from favorable demographics. With approximately one-fifth of the world’s
population and one of the world’s fastest growing economies, China presents
significant potential for the retail drugstore industry. According to the PRC
State Information Center and Bejing Duhuida Consulting’s Research Report, total
expenditure on pharmaceutical products in China increased from RMB175.6 billion,
or $23.1 billion in 2002 to RMB360.3 billion, or $47.3 billion in 2006,
representing a compound annual growth rate of 19.7% in that period. Beijing
Duhuida Consulting, an independent market research and consulting firm, expects
expenditure on pharmaceutical products in China to grow at 23.5% annually
between 2007 and 2011, and to reach RMB965.2 billion, or $126.8 billion, in
2011. The following chart sets forth the historical and projected expenditure on
pharmaceutical products in China by category of pharmaceutical products for the
periods indicated:
|
(In
RMB billions)
|
||||||||||||||||||||
|
2007
|
2008
|
2009 | E | 2010 | E | 2011 | E | |||||||||||||
|
OTC
drugs
|
||||||||||||||||||||
|
Western
medicine
|
10.6 | 12.1 | 13.5 | 14.9 | 16 | |||||||||||||||
|
Traditional
Chinese medicine
|
42.3 | 48.5 | 53.8 | 59.7 | 64.2 | |||||||||||||||
|
Subtotal
|
52.9 | 60.6 | 67.3 | 74.6 | 80.2 | |||||||||||||||
|
Prescription
drugs
|
||||||||||||||||||||
|
Western
medicine
|
230.7 | 293 | 365.2 | 479 | 626.7 | |||||||||||||||
|
Traditional
Chinese medicine
|
57.7 | 73.3 | 91.3 | 119.7 | 156.7 | |||||||||||||||
|
Subtotal
|
288.4 | 366.3 | 456.5 | 598.7 | 783.4 | |||||||||||||||
|
Nutritional
supplements
|
52.4 | 54.8 | 59 | 63.4 | 68.9 | |||||||||||||||
|
Herbal
products
|
21.1 | 23.4 | 26.8 | 30.8 | 32.8 | |||||||||||||||
|
Total expenditure on
pharmaceutical products
|
414.8 | 505.1 | 609.6 | 767.5 | 965.3 | |||||||||||||||
(Source:
PRC State Information Center and Beijing Duhuida Consulting’s Research
Report)
The
growth potential of the pharmaceutical wholesale and drugstore industry in China
is further demonstrated by the rapid growing expenditure on pharmaceutical
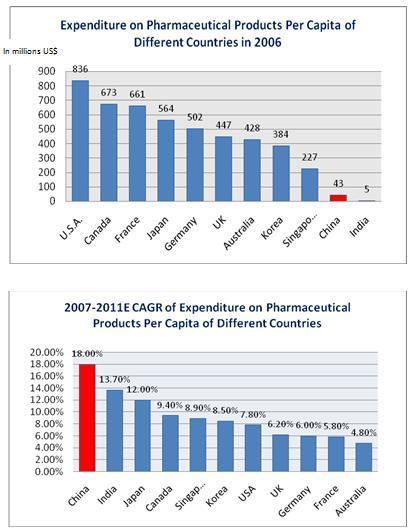
products per capita. According to the PRC National Bureau of Statistics,
expenditure on pharmaceutical products per capita in China was $42.6 in
2006, which lags far behind many developed countries such as the
United States, Germany, France, Australia and Japan. However, it is projected
that expenditure on pharmaceutical products per capita in China is to grow at
18.0% annually between 2007 and 2011 and reach $99.2 by 2011. This growth rate
is significantly higher than in many developed countries such as the United
States, Germany, France, Australia and Japan, where the growth rate of the
expenditure on pharmaceutical products is projected to be between 4.8% and 13.7%
from 2007 to 2011. The following charts set forth expenditure on pharmaceutical
products per capita of various countries in 20061 and the projected growth rate
of expenditure on pharmaceutical products per capita of various countries from
2007 to 2011.
(Source:
PRC National Bureau of Statistics and Beijing Duhuida Consulting’s Research
Report)
Market
Overview
In China,
retail pharmaceutical and other healthcare related products could be purchased
at either hospital pharmacies or non-hospital drugstores, including independent
drugstores and drugstore chains. Historically, sales by hospital pharmacies
accounted for a larger percentage of retail sales of pharmaceutical products in
China. This is because out-patients typically purchase their prescription drugs
at hospital pharmacies in accordance with doctors’ prescriptions. However, if a
medical condition can be treated with OTC drugs, many Chinese people typically
choose to purchase OTC drugs from non-hospital drugstores instead of consulting
a doctor in a hospital for prescription medicines.
|
(in
RMB billions)
|
2007
|
2008
|
2009 | E | 2010 | E | 2011 | E | ||||||||||||
|
Hospital
pharmacies
|
254.1 | 278.9 | 298.2 | 318.8 | 325.9 | |||||||||||||||
|
Non-hospital
drugstores
|
107.3 | 125.8 | 152.4 | 184.5 | 226.4 | |||||||||||||||
|
Total
retail drug sales
|
361.4 | 404.7 | 450.6 | 503.3 | 552.3 | |||||||||||||||
|
Non-hospital
drugstore
|
||||||||||||||||||||
(Source:
Beijing DHD’s research report)
In recent
years, the PRC government has promulgated a number of measures to regulate the
pharmaceutical industry, directly or indirectly, which are expected to favor
non-hospital drugstores more than hospital pharmacies. According to 2007-2008
Research on Financing and M&A Strategy of China Pharmaceutical Chain Store
for Beijing Duhuida Consulting Limited sales from non-hospital drugstores are
expected to constitute 41.0% of total drug sales in 2011, compared to 28.4% in
2006. The above table sets forth historical and projected retail drug sales by
hospital pharmacies and non-hospital drugstores, which include independent
drugstores and drugstore chains, for the periods indicated:
Fragmentation
of the Pharmaceutical Chain Store Industry and the Trend for
Consolidation
The
drugstore industry in China is highly fragmented. Retail pharmacies in China
include chain drugstores, individual stores, and OTC counters in retail chain
stores and supermarkets. While pharmacy chain stores and retail chain stores
with OTC counters are expanding quickly, neither format has developed a
nationwide presence in China. The NDRC reported that as of December 31, 2004,
7,445 pharmaceutical product wholesalers, 1,410 pharmacy chain stores and 58,065
individual pharmaceutical product retailers have obtained Good Supply Practices
certification. According to Status Quo of Drug Supervision in China, there were
more than 340,000 retail pharmaceutical stores in China in 2007. Given the level
of fragmentation and increased regulatory requirements, the Company believes
retailers with an effective nationwide presence and a strong reputation are most
likely to thrive.
Non-Pharmaceutical
Sales Opportunity at Retail Pharmacies
We
believe drugstore non-pharmaceutical merchandise, combined with prescription and
non-prescription drugs, provides customers with a complete wellness solution.
Non-pharmaceutical merchandise includes nutrition supplements, beauty, cosmetics
and fragrance products, personal care products, as well as consumable, seasonal,
promotional and other non-prescription products.
Challenges
for the Drugstore Industry in China and Increased Competition
While the
Chinese economy in general and the drugstore industry in particular have grown
significantly in the past decade, such growth may not continue in the future.
The drugstore industry in China faces a number of challenges,
including:
|
·
|
Competition
in the retail drugstore market in China may also
intensify;
|
|
·
|
Industry
reforms aimed to meet China’s commitments under WTO may foster increased
competition from multinational pharmacy chains at the expense of
China-based pharmacy chains; and
|
|
·
|
Current
PRC laws and regulations limit any foreign investor’s ownership of
drugstores to 49.0% if the foreign investor owns interests in more than 30
drugstores in China that sell a variety of branded pharmaceutical products
sourced from different suppliers. If this restriction is relaxed or
eliminated, there may be increasing competition from large foreign
drugstore chains which intend to enter into the drugstore industry in
China.
|
Production
Facility
Our
production plant maintains Good Manufacturing Practice (“GMP”) certification
authorized by the national accreditation bodies of the PRC. A
GMP-certified facility operates under the GMP parameters prescribed by the
institution granting such certification. GMP parameters are operating standards
that are formed to ensure product quality, by regulating the manufacturing
space, the storage warehouse for raw materials and finished products, and
laboratory areas of the production facility. Hefeng Pharmaceutical operates our
production line and holds a general GMP Certificate that was issued on July 14,
2009 and will expire on July 13, 2010, and also holds a GMP Certificate for
Small Volume Parental Solution that was issued on July 10, 2006 and will expire
on July 9, 2011. Both GMP Certificates will be subject to renewal of additional
five-year term.
Target
Market
Our
business operations are located in Guangxi province which hosts many second- and
third- tier cities with less competition in the market of manufacture and
distribution of pharmaceutical products. Through our experience in operating in
such a business environment, we have accumulated extensive business operating
experience in developing a market in second- and three-tier cities and rural
areas, and have built a strong reputation and brand name awareness in Guangxi
province. Moreover, we have not only gained valuable experience in
operational management, but also built up a strong sales network in Guangxi
provision. With the brand name and leading position we have established in
Guangxi province, we will continue building and expanding our retail and
wholesale business in the second- and third-tier cities and the rural areas in
Guangxi province through our current retail stores and the new stores that we
may acquire in the future. Based upon on continued forecasted growth
in Guangxi province, we may apply the business model we have establish in
Guangxi province to our business expansion in the second- and third-tier cities
and the rural areas of our contiguous provinces, such as Yunnan or Huainan
provinces.
Competition
Retail
and Wholesale
The
pharmaceutical industry in China is intensely competitive, rapidly evolving and
highly fragmented. In many large cities in China, we need to not only compete
with other retail drugstores, but also face increasing competition pressure from
discount stores, convenience stores and supermarkets. In order to maintain our
competitive position in the market, we have increasingly diversified products
and services by offering some non-drug products that are provided in regular
convenience stores. In addition, we also increased our competitiveness through
careful selection of store location, merchandise, and services.
With the
continuous consolidation of the pharmaceutical industry and opening of new
drugstore chains in large cities, we will face more competition in the industry.
However, in many of our targeted second- and third- tier cities and rural areas,
we are facing less competition because major drugstore chains have not entered
into the market. We are in a good position to establish our standing and
reputation in these targeted markets. In addition, the pharmaceutical industry
has entrance barriers for new entrants due to the requirements for capital,
brand name, management expertise, etc. Further, PRC laws and regulations limit a
foreign investor’s ownership in retail drugstores to the maximum of 49.0% if
such investor holds ownership interest in more than 30 drug stores that sell a
variety of branded drugs sourced from different suppliers. This limitation,
together with the complexity of the Chinese market, creates a barrier for
foreign retail drugstore chain operators to enter into the PRC market. As a
result, currently we do not face notable competition from foreign owned
drugstore chains.
Because
our network covers many cities and areas, and many of drugstores are regional,
our competitors vary from region to region. Each region can have its own, among
others, distinct demographics, local regulations and shopping style. We do not
consider any individual regional drugstore as our major competitor, but we
compete with them on an aggregate basis. Our main competitors in Guangxi
province are Sinopharma Liuzhou Branch and Sinopharma Nanning Branch, Liuzhou
Medical and Pharmaceutical Limited on wholesale side; Shenzhen Accordance Pharm.
Chain Store Inc., and Hunan Laobaixing Pharmacy Chain on retail
side.
Manufacturing
With
respect to competition in our drug production business, we compete with other
manufacturers in each specific drug category. For instance, although we are the
sole authorized producer of Corydalis Saxicola Bunting (Yanhuanglian), which is
the preferred drug treating chronic hepatitis A, B and C, there are drugs that
have a similar medical effect for treating hepatitis.
Government
Regulation
We are
subject to various Chinese laws and regulations pertaining to the pharmaceutical
industry. We have attained certificates, permits, and licenses required for the
operation of a pharmaceutical enterprise and the manufacturing of pharmaceutical
products in China.
In 1998,
the PRC State Food and Drug Administration (“SFDA”) introduced the GMP
Certificate in order to promote quality and safety of pharmaceutical production
Good Manufacturing Practices have been revised in July and October, 2004. We are
required to meet GMP standards in order to continue manufacturing pharmaceutical
products and health foods. For each new product, we prepare documentation of
pharmacological, toxicity, pharmacokinetics and drug metabolism studies in
addition to providing samples of the drug. The documentation and samples are
then submitted to the provincial food and drug administration. This process
typically takes approximately three months. After the documentation and samples
have been approved by the provincial food and drug administration, the
provincial administration submits the approved documentation and samples to the
SFDA. The SFDA examines the documentation and tests the samples and presents the
findings to the New Drug Examination Committee for approval. If the application
is approved by the SFDA, the SFDA will issue a clinical trial license to the
applicant for clinical trials. This clinical trial license approval typically
takes one year, followed by approximately two years of trials, depending on the
category and class of the new drug. The SFDA then examines the documentation
from the trial and, if approved, issues the new drug license to the applicant.
This process usually takes eight months. The entire process takes anywhere from
three to four years.
The GMP
certificate is valid for a term of five years, the pharmaceutical products
production permits are subject to renewal every five years, and the health food
production permits are valid for three-year terms, and each must be renewed
before its expiration, if applicable. If our GMP certificate expires without
renewal, we will not be able to continue manufacturing pharmaceutical products,
which will cause our production operations to be terminated.
In
addition, a distributor of pharmaceutical products in China must obtain a
pharmaceutical distribution permit from the relevant provincial or local SFDA
branches. The distribution permit is granted if the relevant SFDA provincial
branch receives satisfactory inspection results of the distributor’s facilities,
warehouse, hygiene environment, quality control systems, personnel and
equipment. A pharmaceutical distribution permit is valid for five
years.
The SFDA
applies Good Supply Practice (“GSP”) standards to all pharmaceutical wholesale
distributors as well as our retail to ensure the quality of distribution in
China. The currently applicable GSP standards require pharmaceutical
distributors to implement controls on the distribution of medicine, including
standards regarding staff qualifications, distribution premises, warehouses,
inspection equipment and facilities, management and quality control. A
certificate for GSP standards, or GSP certificate, is valid for five years,
except for a newly established pharmaceutical distribution company, for which
the GSP certificate is valid for only one year. If our GSP certificate expires
without renewal, we will not be able to continue distributing pharmaceutical
products, which will cause our wholesale and retail distribution to be
terminated.
Competitive
Advantage
As
the leading pharmaceutical distributor in the region, we are well-positioned to
benefit from the strong growth, consolidation, and regulatory reform in the PRC
pharmaceutical and healthcare industry.
The PRC
healthcare market is one of the fastest-growing healthcare markets in the world,
driven by China’s rapidly growing economy, rising living standards, increased
health consciousness, large aging population and proactive government policies.
Furthermore, the PRC Government recently announced a reform plan to spend RMB850
billion on healthcare in addition to the regular healthcare budget from 2009 to
2011, in order to increase the availability of healthcare, basic medicines and
health insurance coverage for people in China. As a comparison, in 2007, the
total healthcare expenditure in China was approximately RMB1.1 trillion, of
which approximately RMB230 billion was government spending, according to the
Ministry of Health. The healthcare reform plan is expected to accelerate growth
in the PRC pharmaceutical industry not only by the increased government
spending, but also by the expected increases in private healthcare spending
stimulated by larger government subsidies to PRC residents, as per capita
healthcare spending remains much lower than in developed countries. We are
well-positioned to capture business opportunities resulting from this fast
growing market.
Building
up a modernized logistic center in 2010 and 2011, streamlined supply chain and
increased capital entrance barrier for smaller competitors to further strengthen
our leading position and differentiate in the region
In
addition, China’s pharmaceutical distribution market is highly fragmented and is
characterized by inefficient supply chains. The highly fragmented pharmaceutical
distribution industry has recently commenced a process of consolidation, which
has led to an increase in market share of the pharmaceutical distributors in the
market, we expect the PRC pharmaceutical distribution market to continue to
consolidate into one with larger and more efficient distributors. In addition,
we expect the healthcare reform plan to promote further consolidation, as it
calls for reducing the number of layers between manufacturers and
consumers
of medicines. We believe that we have the scale, industry standing, brand and
financial strength to compete effectively during this process of consolidation.
The PRC Government has also adopted measures to raise the operating standards of
pharmaceutical companies and promote the quality of distribution of
pharmaceutical products in China, in order to ensure a stable supply of safe,
effective medicines at reasonable prices. We believe we will benefit from future
regulatory reforms, which require pharmaceutical manufacturers and distributors
to implement more stringent standards on the manufacturing and distribution of
pharmaceutical products. Unlike smaller distributors, we have a large-scale
distribution network, high quality equipment and facilities, leading management
and qualified personnel, which are required to satisfy the higher standards.
These attributes also provide us with a competitive edge over our
competitors.
Leading
Product and Brand Name in Diversified Area
Over the
years, we have developed and introduced a number of pharmaceutical products
under the brand name Asio (亚太), which we
believe are the leading products in their respective market segments, including
Levodopa, which treats the stiffness, tremors, spasms, and poor muscle control
of Parkinson’s disease; Hydroxycamptotbecine Injection, which we believe is one
of the best choices to fight cancers of various kinds with minimal side effects;
Tabellae Sarcandrae, which is called Chinese anti-biotic and has similar
anti-inflammatory and antibacterial effects as anti-biotics used in Western
medicine; Corydalis Saxicola Bunting (Yanhuanglian), which treats hepatitis,
liver cirrhosis, liver ascites and liver cancer; and Yinge Tongmai Tea, which
improves cardio-vascular condition and blood circulation, and can be taken as a
normal healthy drink on a daily basis. As a result, “Asio (亚太)” has become a
widely recognized brand name for our self-developed drugs in China. Among our
five best-selling drugs, we are the sole producer of Corydalis Saxicola Bunting
(Yanhuanglian) and Hydroxycamptotbecine Injection.
In
addition, our sales and marketing teams are specialized in promoting products in
different therapeutic categories. The teams have strong relationships with
healthcare executives, doctors and pharmacies in their respective target markets
and possess extensive sales and marketing experience in promoting prescription
and non-prescription pharmaceutical products.
Strong
Research and Development Capability
Our
research and development department relies upon a research institute jointly
formed by us and several renowned Chinese medicine universities, including China
Medicine University located in Nanjing China, which focuses on
utilizing Guangxi’s natural herbal medicine resources to produce drugs that meet
the demand of the Chinese domestic market.
Guangxi
is a province with a wealth of natural resources for developing traditional
Chinese medicine. In addition, they also used great efforts to discover advanced
production process of Levodopa and Corydalis Saxicola Bunting (Yanhuanglian), to
develop better understanding of the composition of Hydroxycamptotbecine and its
derivatives, to develop key technology of figure print quality control, and to
research the production technology of herbal medicine in limestone mountainous
areas.
Extensive
Wholesale and Retail Distribution Network and medi-care qualified
stores
We
operate wholesale and retail distribution networks covering major cities,
townships, and counties of Guangxi province. Our wholesale network is the
largest regional wholesale network in China, which offers more than 10,000
pharmaceutical products to our clients. Further, BCT Retail offers more than
3,200 pharmaceutical products in more than 66 retail stores, holding the leading
market position in many major cities of Guangxi province. Among the 66 stores,
there are 16 stores are covered by medi-care insurance plan for citizens and
employees. We’ve also signed an MOU with local authorities to be authorized to
open 30 retail stores under the New Rural Co-operation Health Issuance
Plan.
Economic
of Scope and Integration Ability
We
believe we have the potential to expand our scale by acquiring more wholesale
and retail networks. By acquisition of a production facility in 2008, we have
established a full-integrated operation. Established in the early
1970s, our production facility has accumulated a large reserve of production
licenses, established an experienced management team, formed a strong research
and development team, and developed solid relationships with local raw material
providers. This integration enables us to enjoy economies of scale
and also provides us with an ability to increase our operation efficiency in
order to sustain strong profit margins and favorable growth rates.
Experienced
Management Team with Proven Track Record
Over the
past decade, Mr. Hui Tian Tang, Chairman of our board of directors and other
members of our senior management team have taken a successful leadership in our
operations and increased our revenue and profit. Many members of our senior
management team have worked with us since our inception or otherwise have broad
experience in the retail industry, and have developed extensive expertise in
operating a national chain of drugstores, which is important to our future
development. The Chief Operating Officer of Liuzhou BCT, Jing Hua Li, has
extensive experience in chain store retailing, gained from his four years
service with Meidong Bip-technology (AOB), a publicly held multinational
pharmaceutical corporation. In addition, the majority of our
mid-level managers and managers of our regional operations and stores have been
with us for many years. These managers have obtained extensive experience
through our internal management training system and real practice in managing
retail stores and distribution centers.
Well-established
Good Supply Practices (GSP) control system
Our
logistics system is established in line with GSP. We also have a full-time GSP
supervision body comprised of professionals with professional pharmacist
qualification or pharmacist-in-charge titles. They are familiar with national
laws and regulations on medicine quality, and have extensive knowledge and
practical experience in the implementation of GSP in the
enterprise.
Professional
storage, advanced transportation equipment, comprehensive management system and
detailed flow records are integrated with hardware and software of the
pharmaceutical business. The medicine warehouse occupies an area of 4,800 square
meters, including warehouses of room temperature (0-30°C), cool (<20°C) and
cold (2-8°C), stock control and subsidiary operation area, chemical laboratory
and has advanced instruments and facilities.
GSP,
which can be called a total quality control system for the pharmaceutical
industry, is strictly observed from purchase to sale to storage, forming a
secure tunnel for the medicine transportation and guaranteeing medicine
quality.
Growth
Strategy
Expanding
of Our Wholesale and Retail Networks targeting the Second and Third Tier Cities
and Rural Markets
We
believe that maintaining a large number of retail stores in desirable geographic
markets is essential to our competitiveness and our ability to increase our
profitability. We are attempting to significantly expand our market presence in
our contiguous provinces in China by effectively leveraging our existing
operating infrastructure.
Maintain
and Improve Customer Loyalty with Effective Marketing and Promotional
Programs
We
believe that a strong brand name is critical to obtaining customers’ trust in
our business, as well as building customer loyalty and increasing customer
visits to our stores. As a result, we intend to continue promoting aggressively
and effectively both our brand name and our private label products.
Specifically, we will continue to deploy the following marketing and promotional
initiatives:
1. adopting
advanced category management by focusing on seasonal and cross-merchandising,
and offering a wider selection of products;
2. offering
services that are carefully tailored to our customers’ healthcare needs,
including integrated health programs focused on health supplements, weight
management, diabetes, infant care and birth control;
3. enhancing
our customer loyalty by organizing community-based activities and targeted
promotion programs;
4. using
data mining techniques to tailor relevant promotional offers to our target
customers, especially our loyalty members;
5. enlarging
the number of our promotional partners and developing additional promotional
campaigns with these partners; and
6. advertising
our brand and private label products in selected newspapers that service our
targeted cities.
Selectively
Pursue Complementary Acquisitions of Chain Stores and Wholesale
Networks
We
currently plan to selectively acquire drugstore chains or independently operated
drugstores that complement our existing store network or help us to establish a
presence in new markets. In particular, we plan to grow through first acquiring
similar businesses in the cities in Guangxi province and then acquiring business
targets outside of the Guangxi province. We target retail chains or individual
stores in prime locations and with good brand names, well-developed facilities
and customer bases that are complementary to ours, and which are commercially
attractive. We believe that our relationship with many industry participants and
our knowledge of, and operational expertise in, the drugstore market in China
will assist us in making acquisitions. We also believe that we can rapidly and
successfully integrate newly acquired stores into our current distribution
network and quickly realize operating and financial benefits.
Invest
in a more advanced logistics and information management systems to improve cost
and operating efficiencies.
We intend
to invest in an advanced logistic system and taken significant steps to
reengineer and integrate our logistics process and reorganize our logistic
network to ensure our competitiveness as efficient logistics and information
management systems are critical to our business and networks. In Liuzhou city
first, we plan to establish regional and provincial logistics facilities
equipped with leading technology and information systems. These improvements
have provided a direct and high-speed information exchange channel that connects
us to our customers and suppliers throughout China, covering each stage of our
pharmaceutical distribution operations, including electronic order entry,
invoice preparation, purchasing, inventory tracking, GSP-certified warehousing
and logistics and delivery arrangements. As a result, we have shortened delivery
lead-times, increased responsiveness to customer demands and reduced
distribution and selling expenses.
Continue
to build upon our integrated business platform in order to enhance the synergies
between our pharmaceutical distribution, retail pharmacy and other
businesses.
We will
continue to build upon our integrated business platform in order to enhance the
synergies that arise from our pharmaceutical operations spanning the
distribution, retail and manufacturing of pharmaceutical and healthcare products
and expand our reach to end-customers. We plan to leverage our existing
businesses to capitalize on particular opportunities that may arise and create
efficiencies and cost savings in our business operations. We also plan to
utilize our extensive distribution network to provide reliable supply channels
for our retail drug stores which, in turn, will sell the pharmaceutical and
healthcare products supplied by our distribution network. Further, we intend to
utilize our pharmaceutical manufacturing operations to produce private-label
products for our retail pharmacy operations. We expect to maintain the
flexibility to reallocate manufacturing capacity of our products in response to
potential changes in supply and demand, as well as to control inventory in a way
that enables us to meet expected demand for our products. With operations in
multiple segments of the pharmaceutical industry, we are able to maintain
control over the quality, supply chain management and marketing of our products.
We believe that our integrated business platform maximizes the synergies between
our businesses by optimizing our operational efficiency while reducing the costs
of our customers, allowing us to further solidify our leadership in the PRC
pharmaceutical distribution industry.
Intellectual
Property
We
believe our rights to our trade names and trademarks are the most important
factors in implementing our marketing strategy. Our company’s name, Baicaotang
(百草堂), means
“Place for all kind of Chinese Herbal Medicines” in Chinese. We registered
“Baicaotang (百草堂)” as the
trademark for our business group, and Asio (亚太) as the
trademark for the medicines that our production plant produces.
Under the
PRC law, we have the exclusive right to use a trademark for products and
services for which the trademark has been registered with the State
Administration for Industry and Commerce. Certain things cannot be
registered as trademarks including but not limited to the marks set forth in
Article 10 and 11 of the Trademark Law, single color, sound, activity, odor,
three-dimension trademarks only indicating the shape of the product, copy,
imitation or translation of well-known trademarks not registered in China to be
used on the same or similar products, copy, imitation or translation of
well-known trademarks registered in China to be used on any products and
geographic marks not indicating the resource of the products. Trademark registration
is valid for 10 years, starting from the day
the registration is approved. For renewal of registration, an
application shall be made within 6 months before the expiration of such 10
years. If no application is made within that period, an extension of 6 months
may be granted.
The
registration of a trademark shall undergo two steps: preliminary examination and
public announcement. If the registration application is revoked in
the preliminary examination process, or is challenged by any party in the public
announcement process and such challenge is accepted by the Trademark Bureau, the
applicant may within 15 days apply to the Trademark Review and Adjudication
Board for a re-examination and, if still not being satisfied with the result, it
may also within 30 days file a lawsuit to the court against the Trademark Review
and Adjudication Board. Within 3 months after the trademark to be
registered is publicly announced, if no challenge or opposition is received, the
registration shall be approved with a trademark registration certificate to the
applicant.
If we
believe that a third party has infringed upon our exclusive rights with respect
to any of our registered or licensed trademarks, we may, through appropriate
administrative and/or civil procedures, institute proceedings to request the
relevant authority for an injunction or to resolve the infringement through
negotiation or litigation with the infringer. The relevant authority also has
power to impose fines, confiscate or destroy the infringing products or
equipment used to manufacture the infringing products. As our brand names and
trademarks become more recognized in the pharmaceutical market in China, we are
devoting additional resources to increasing and enforcing our trademark rights,
which is critical to our overall branding strategy and reputation.
We also
rely on trade secrets to protect our know-how and other proprietary information.
Like other retailers, we generate proprietary information in connection with our
operations, such as pricing, purchasing, promotional strategies, customer lists
and supplier lists. We believe this proprietary information is essential to the
operations of our business and the success of our competition strategies.
Therefore, we strive to protect such information. For example, the key members
of our management team have signed a confidentiality agreement with us pursuant
to which they have committed not to disclose the confidential information
acquired during their employment with us and not to compete with us for three
years after their employment terminates. If we believe that any
operator has infringed upon our trade secrets, we may, through appropriate
administrative and/or civil procedures, institute proceedings to request the
relevant authority for an injunction or to resolve the infringement through
negotiation or litigation with the infringer. The relevant authority also has
power to impose certain amount of fine upon the infringer.
Employees
As of
December 31, 2009, we had 816 full time employees.
OUR
HISTORY AND CORPORATE STRUCTURE
We were
originally incorporated in the State of Delaware on November 30, 2006 under the
name Purden Lake Resource Corp. to engage in the acquisition, exploration and
development of natural resource properties. We ceased our
operations because we had not commenced any exploration activities, and as a
result prior to December 30, 2009 we were a “blank check company with nominal
assets. On December 24, 2009 we changed our name to China Baicaotang Medicine
Limited.
Our
wholly-owned subsidiary, Ingenious, was incorporated under the laws of the
British Virgin Islands on May 29, 2008. Ingenious owns 100% of
the issued and outstanding capital stock of Forever Well, a Hong Kong company
incorporated on January 10, 2008. Forever Well is the sole
shareholder of Liuzhou BCT a PRC limited company established on April 3,
1986. Liuzhou BCT contributed 100% of the registered capital Hefeng
Pharmaceutical, a PRC company established on September 18, 2000 and 49% of the
registered capital of BCT Retail, a PRC company established on October 30, 2001.
The remaining 51% of the registered capital of BCT Retail was contributed by
Property Management, an affiliate of Liuzhou BCT.
The chart
below illustrates the current structure of the Company:

The
Reorganization
In 2008,
the shareholders of Liuzhou BCT (the “Liuzhou BCT Shareholders”) and Xiaoyan
Zhang, our CFO, developed a restructuring plan (the “Restructuring”). At that
time, Ms. Zhang, who is a citizen of Hong Kong, was the sole shareholder of
Ingenious, which was the parent company of Forever Well. The first step was for
Forever Well to acquire 100% of the equity interests of Liuzhou BCT and its
subsidiaries (the “PRC Operating Companies”). Liuzhou BCT was owned at that time
by certain former and current employees and directors of Liuzhou BCT. After the
acquisition of the PRC Operating Companies by Forever Well was consummated, the
second step was for Ingenious to enter into and complete a transaction with a
U.S. public reporting company whereby that company would acquire
Ingenious.
The
acquisition of Liuzhou BCT was structured to comply with the PRC M&A
Laws. Under the PRC M&A Laws, the acquisition of PRC companies by
foreign companies that are controlled by PRC citizens who are affiliated with
the PRC companies is strictly regulated and requires approval from the Ministry
of Commerce, which approval is burdensome to obtain. Such
restrictions however, do not apply to foreign entities which are controlled by
foreign persons. These restrictions apply only at a “snapshot in
time” that occurs at the time PRC companies are acquired by a foreign
entity. In our case this was in August 2008 when Forever Well
acquired 100% of the equity interest of Liuzhou BCT from the Liuzhou BCT
Shareholders for aggregate consideration of RMB10,000,000 (approximately
$1,470,588) which was the registered and fully paid up capital of Liuzhou
BCT. At that time Forever Well was owned by Ingenious, and a majority
of Ingenious’ equity was owned by Ms. Zhang, a Hong Kong citizen.
Since PRC
M&A Laws would have prohibited Liuzhou BCT Shareholders who were PRC
citizens from immediately receiving a controlling interest in Ingenious in a
share exchange as consideration for the sale of their interest in Liuzhou BCT,
Liuzhou BCT Shareholders holding a majority of the equity interest in Liuzhou
BCT and Ms. Zhang instead agreed that they would enter into an Earn-In Agreement
to grant those Liuzhou BCT Shareholders a call right to acquire up to all of Ms.
Zhang’s interest in Ingenious (or a public parent company of Ingenious, as the
case may be) after the acquisition of Liuzhou BCT was consummated in compliance
with PRC law. The Earn-In Agreement was entered into by Ms. Zhang and
certain Liuzhou BCT Shareholders on December 30, 2009 and enables those Liuzhou
BCT Shareholders to purchase shares of Ingenious (or its public parent company)
from Ms. Zhang for a nominal amount per share provided that the Company meets
certain performance targets for fiscal 2011 and 2012. For the 2011
and 2012 fiscal years the performance targets for the Company are $26 million
and $28 million after tax audited net income, respectively. If the
2011 performance target is met, the Liuzhou BCT Shareholders have the right to
acquire 50% of shares held by Ms. Zhang over which they have a call
right. If the 2012 performance target is met, the Liuzhou BCT
Shareholders have the right to acquire the other 50% of the shares held by Ms.
Zhang over which they have a call right. The number of shares which
can be acquired by the Liuzhou BCT Shareholders under the Earn-In Agreement is
in proportion to their former relative ownership interest in Liuzhou
BCT.
On
December 30, 2009, the goal of the Restructuring was realized when we entered
into a share exchange agreement with Ingenious, pursuant to which we acquired
100% of the equity of Ingenious in exchange for the issuance of an aggregate of
32,000,000 shares of our common stock to Ms. Zhang and to certain Liuzhou BCT
Shareholders. As of the date of this prospectus, Ms. Zhang owns 58.9%
of our common stock. As a result of this transaction, we are a
holding company which, through our direct and indirect ownership of Ingenious,
Forever Well, Liuzhou BCT, Hefeng Pharmaceutical and BCT Retail, now has
operations based in the PRC.
Private
Placement
Simultaneously
with the closing of the Share Exchange, we completed the initial closing the
Private Placement of $6,323,002 or 632.3 units. Each unit consists of (i) 3,937
shares of common stock, and (ii) a warrant (the “Warrant”) to purchase 1,968
shares of common stock at an exercise price of $3.81 per share. Upon
the initial closing of the Private Placement, we issued an aggregate of
2,489,370 shares of our common stock and warrants exercisable for 1,224,368
shares of our common stock at an exercise price of $3.81 per share. In
addition, in connection with the initial closing of the Private
Placement, we issued Agent Warrants to the Co-Placement Agents that are
exercisable for 248,937 shares of common stock at an exercise price of $3.05 per
share, on a cash or cashless basis.
On
February 1, 2010, we completed the Second Closing of the Private Placement of
$2,616,108 or 261.61 units with the issuance of a total of 1,029,970 shares of
our common stock and Investor Warrants exercisable for 514,933 shares of our
common stock at an exercise price of $3.81. In connection with the
Second Closing of the Private Placement, we issued Agent Warrants to the
Co-Placement Agents that are exercisable for 102,997 shares of common stock at
an exercise price of $3.05 per share, on a cash or cashless basis.
Contractual
Arrangements with Property Management
We do not
100% ownership interest in BCT Retail due to the restriction of foreign
investment in pharmacy chains with 30 or more drugstores. We have
entered into contractual arrangements with Property Management pursuant to which
the shareholders of Property Management pledged to us their equity interests in
BCT Retail and provide us with the ability to effectively control BCT
Retail.
The
contractual agreements entered into by us and the shareholders of Property
Management include:
Share Transfer
Agreement. Under this agreement dated April 1, 2008 by and between
Liuzhou BCT, which held 100% equity interests of BCT Retail, and Property
Management, Property Management acquired from Liuzhou BCT 51% equity interests
in BCT Retail. The total amount of transfer price was
RMB153,000. Within twenty (20) business days from the date when
Property Management paid the transfer price, all the parties were to amend the
articles of association of BCT Retail and register such equity transfer with the
competent Authority of Industry and Commerce.
Shares Pledge
Agreement. Under this agreement dated May 3, 2008 among Liuzhou BCT and
the shareholders of Property Management, the shareholders of Property Management
pledged all of their equity interests in BCT Retail to Liuzhou BCT to guarantee
their obligations to repay a loan in the amount of RMB153,000 made from Liuzhou
BCT to them.
Share Repurchase
Agreement. Under this agreement dated July 31, 2008 by and between
Liuzhou BCT and Property Management, Liuzhou BCT was granted a preemption right
to repurchase the 51% equity interests in BCT Retail held by Property
Management.
Shares Pledge
Agreement. Under this agreement dated March 31, 2009 among Liuzhou BCT
and the shareholders of Property Management, the shareholders of Property
Management pledged all of their equity interests in BCT Retail to Liuzhou BCT to
guarantee their obligations under a loan in the amount of RMB 1.377 million made
from Liuzhou BCT to them.
Agreements
relating to the Private Placement
Registration
Rights
Pursuant
to the Subscription Agreement, on or prior to March 3, 2010 (the “Registration
Statement Filing Date”), we shall file with the Securities and Exchange
Commission (the “SEC”) a registration statement (the “Registration Statement”)
under the Securities Act of 1933, as amended (the “Securities Act”), (i)
registering for resale by the Investors (a) the shares of common stock issued to
the Investors in the Private Placement and (b) the shares of common stock
underlying the Investor Warrants; and (ii) registering for resale for the
Co-Placement Agents of the Private Placement and other agents, the shares of
common stock underlying the Agent Warrants (all of the foregoing securities
being collectively referred herein as the “Registrable Securities”). We shall
use our best efforts to have the Registration Statement declared effective prior
to the 150th day following the Second Closing, provided, however, that in
the event of a “full review” by the SEC, we shall be afforded an additional 30
days and shall have the Registration Statement declared effective prior to the
180th day following the Closing (the “Registration Effective
Date”).
In the
event that (i) the Registration Statement has not been (x) filed on or prior to
the Registration Filing Date or (y) declared effective by the SEC on or before
the Registration Effective Date; and (ii) the Registrable Securities included in
such Registration Statement are not saleable under Rule 144, we shall pay to
each Investor as liquidated damages, a cash payment equal to 1% of the
aggregated amount invested by such Investor in the Private Placement for the
first 30 days and 1% of the aggregated amount vested by such Investors in the
Private Placement for every 30-day period thereafter until the Registration
Statement has been filed and/or declared effective, or such proportionate
percentage for any period less than 30 days.
Make
Good Escrow
In
addition, pursuant to the Subscription Agreement, at the closing of the Private
Placement, Xiaoyan Zhang, our CFO placed 4,000,000 shares of our common stock
owned by her (the “Make Good Shares”) in an escrow account administrated by an
escrow (the “Make Good Escrow Agent”). In the event we fail to achieve a
performance target of $26,000,000 recurring operating net income under the U.S.
GAAP before any extra-ordinary gain and excluding any non-cash expenses for our
fiscal year ending December 31, 2010 (the “Performance Target”), the Make Good
Escrow Agent shall distribute 1,000,000 Make Good Shares to all Investors on a
pro rata basis for every $1,000,000 shortfall under the Performance
Target. If the Performance Target is met, the Make Good Shares shall
be returned to Ms. Zhang.
|
Risk
Factors
|
An
investment in our common stock involves a high degree of risk. You should
carefully consider the risks described below and the other information contained
in this report before deciding to invest in our common stock.
Risks
Relating to Our Business
OUR
OPERATING HISTORY MAY NOT SERVE AS AN ADEQUATE BASIS TO JUDGE OUR FUTURE
PROSPECTS AND RESULTS OF OPERATIONS.
Our
operating history may not provide a meaningful basis on which to evaluate our
business. We cannot assure you that we will maintain our profitability or that
we will not incur net losses in the future. We expect that our operating
expenses will increase as we expand. Any significant failure to realize
anticipated revenue growth could result in significant operating
losses.
WE
MAY BE UNABLE TO COMPETE SUCCESSFULLY AGAIN NEW AND EXISTING
COMPETITORS.
The major
market of our products and business operations is the cities, villages and towns
of Guangxi province. We may face increasing competition in the future as we
expand our business in Guangxi province and our adjacent provinces. As we expand
our operations in wholesale and retail distribution and manufacture of
pharmaceutical products, we will encounter competition from other companies
existing in our target markets and may face future completion from new foreign
and domestic competitors entering the pharmaceutical promotion and distribution
market in China. Our current and future competitors may be able to respond more
quickly to new or changing opportunities and customer requirements and may be
able to undertake more extensive promotional and distribution activities, offer
more attractive terms to customers, and adopt more aggressive pricing policies.
Competition could reduce our market share or force us to lower our prices to
unprofitable levels.
WE
MAY BE UNABLE TO MANAGE OUR GROWTH EFFECTIVELY.
Our
business strategy is based on the assumption that we will acquire additional
retail and wholesale distribution channels in the future and that the number of
our customers and the extent of our operations will grow. Our ability to compete
effectively and to manage our future growth, if any, requires us
to:
|
·
|
continue
to improve our financial and management controls and reporting systems and
procedures to support the proposed expansion of our business operations of
the acquisition of any additional retail and wholesale distribution
channels in the future; and
|
|
·
|
locate
or hire, at reasonable compensation rates, qualified personnel and other
employees necessary to expand our capacity in order to accommodate the
proposed expansion of our business
operations.
|
If we are
unable to accomplish any of these objectives, we will be unsuccessful in
effectively managing our growth, which could harm our business, operating
results, and financial condition.
OUR
CURRENT BUSINESS OPEARTIONS RELY HEAVILY UPON MR. HUI TIAN TANG, OUR CHIEF
EXECUTIVE OFFICER AND CHAIRMAN.
We have
been heavily dependent upon the expertise and management of Mr. Hui Tian Tang,
our Chairman and Chief Executive Officer, and his continued services. The loss
of Mr. Tang’s’ services could seriously interrupt our business operations.
Although we have entered into an employment contract with Mr. Tang, pursuant to
which Mr. Tang agrees to serve as our full time Chief Executive Officer, and Mr.
Tang has not indicated any intention of leaving us, the loss of his service for
any reason could have a very negative impact on our ability to fulfill our
business plan.
THE
LIMITED PUBLIC COMPANY EXPERIENCE OF OUR MANAGEMENT TEAM COULD ADVERSELY IMPACT
OUR ABILITY TO COMPLY WITH THE REPORTING REQUIREMENTS OF U.S. SECURITIES
LAWS.
Our management team has
limited public company experience, which could impair our ability to comply with
legal and regulatory requirements such as those imposed by Sarbanes-Oxley Act of
2002. Our senior management has never had sole responsibility for managing a
publicly traded company. Such responsibilities include complying with federal
securities laws and making required disclosures on a timely basis. Our senior
management may not be able to implement programs and policies in an effective
and timely manner that adequately respond to such increased legal, regulatory
compliance and reporting requirements, including the establishing and
maintaining internal controls over financial reporting. Any such
deficiencies, weaknesses or lack of compliance could have a materially adverse
effect on our ability to comply with the reporting requirements of the Exchange
Act, which is necessary to maintain our public company status. If we were to
fail to fulfill those obligations, our ability to continue as a U.S. public
company would be in jeopardy in which event you could lose your entire
investment in our company.
WE
MAY BE UNSUCCESSFUL IN ATTRACTING OR RETAINING KEY SALES, MARKETING AND OTHER
PERSONNEL.
The
success of our business is dependent on our ability to attract and retain highly
skilled managers and sales and marketing personnel. Our sales personnel carry
out our critical promotional and sales activities. There is intense competition
for qualified sales and marketing personnel, and we may be unable to attract,
assimilate or retain additional qualified sales and marketing personnel on a
timely basis. Our inability to retain key personnel or the failure to attract
additional qualified personnel could harm our development and results of
operations. In addition, as we plan to expand in China, we will need to attract
additional qualified managerial staff and other personnel. We may have
difficulty in hiring and retaining a sufficient number of qualified personnel to
work in China. This may impede the development of our distribution business and
the expansion of our business in China.
IF
WE CANNOT SUCCEED IN IMPLEMENTING OUR GROWTH STRATEGY, OUR GROWTH, FINANCIAL
CONDITION, RESULTS OF OPERATIONS AND CASH FLOW MAY BE ADVERSELY.
We plan
to grow in Guangxi province through increasing the distribution and sales of our
products by penetrating the existing markets and then expand our business
operations to enter into new geographic markets in our neighbor provinces.
However, many obstacles to entering such new markets exist, including, but not
limited to, shipping and delivery costs and costs associated with marketing
efforts. We cannot, therefore, assure you that we will be able to successfully
overcome such obstacles and establish our products in any additional markets.
Our inability to implement this organic growth strategy successfully may have a
negative impact on our growth, future financial condition, results of operations
or cash flows.
IF
WE NEED ADDITIONAL CAPITAL TO FUND OUR GROWING OPERATIONS, WE MAY NOT BE ABLE TO
OBTAIN SUFFICIENT CAPITAL AND MAY BE FORCED TO LIMIT THE SCOPE OF OUR
OPERATIONS.
In
connection with our growth strategies, we may experience increased capital needs
and accordingly, we may not have sufficient capital to fund our future
operations without additional capital investments. Our capital needs will depend
on numerous factors, including (i) our profitability; (ii) the release of
competitive products by our competition; (iii) the level of our investment in
research and development; and (iv) the amount of our capital expenditures,
including acquisitions. We cannot assure you that we will be able to obtain
capital in the future to meet our needs.
If we
cannot obtain additional funding, we may be required to: (i) limit our
investments in research and development; (ii) limit our marketing efforts; and
(iii) decrease or eliminate capital expenditures. Such reductions could
materially adversely affect our business and our ability to
compete.
Even if
we do find a source of additional capital, we may not be able to negotiate terms
and conditions for receiving the additional capital that are acceptable to us.
Any future capital investments could dilute or otherwise materially and
adversely affect the holdings or rights of our existing shareholders. In
addition, new equity or convertible debt securities issued by us to obtain
financing could have rights, preferences and privileges senior to our common
stock. We cannot give you any assurance that any additional financing will be
available to us, or if available, will be on terms favorable to us.
IF
WE FAIL TO INCREASE OUR BRAND RECOGNITION, WE MAY FACE DIFFICULTY IN OBTAINING
NEW CUSTOMERS AND BUSINESS PARTNERS.
We
believe that establishing, maintaining and enhancing our brand in a
cost-effective manner is critical to achieving widespread acceptance of our
current and future services and is an important element in our effort to
increase our customer base and obtain new business partners. We believe that the
importance of brand recognition will increase as competition in our market
develops. Some of our potential competitors already have well-established brands
in the pharmaceutical promotion and distribution industry. Successful promotion
of our brand will depend largely on our ability to maintain a sizeable and
active customer base, our marketing efforts and our ability to provide reliable
and useful services at competitive prices. Brand promotion activities may not
yield increased revenue, and even if they do, any increased revenue may not
offset the expenses we incur in building our brand. If we fail to successfully
promote and maintain our brand, or if we incur substantial expenses in an
unsuccessful attempt to promote and maintain our brand, we may fail to attract
enough new customers or retain our existing customers to the extent necessary to
realize a sufficient return on our brand-building efforts, in which case our
business, operating results and financial condition would be materially
adversely affected.
AS
A DISTRIBUTOR AND MANUFACTURER OF PHARMACEUTICAL PRODUCTS, WE ARE EXPOSED TO
INHERENT RISKS RELATING TO PRODUCT LIABILITY AND PERSONAL INJURY
CLAIMS.
Pharmacies
are exposed to risks inherent in the manufacturing and distribution of
pharmaceutical and other healthcare products, such as with respect to improper
filling of prescriptions, labeling of prescriptions, adequacy of warnings,
unintentional distribution of counterfeit drugs. In addition, product liability
claims may be asserted against us with respect to any of the products we sell
and as a retailer, we are required to pay for damages for any successful product
liability claim against us, although we may have the right under applicable PRC
laws, rules and regulations to recover from the relevant manufacturer for
compensation we paid to our customers in connection with a product liability
claim. We may also be obligated to recall affected products. If we are found
liable for product liability claims, we could be required to pay substantial
monetary damages. Furthermore, even if we successfully defend ourselves against
this type of claim, we could be required to spend significant management,
financial and other resources, which could disrupt our business, and our
reputation as well as our brand name may also suffer. We, like many other
similar companies in China, do not carry product liability insurance. As a
result, any imposition of product liability could materially harm our business,
financial condition and results of operations. In addition, we do not have any
business interruption insurance due to the limited coverage of any business
interruption insurance in China, and as a result, any business disruption or
natural disaster could severely disrupt our business and operations and
significantly decrease our revenue and profitability.
THE
RETAIL PRICES OF SOME OF OUR PRODUCTS ARE SUBJECT TO CONTROL, INCLUDING PERIODIC
DOWNWARD ADJUSTMENT, BY PRC GOVERNMENTAL AUTHORITIES.
A number
of our pharmaceutical products, primarily those included in the national and
provincial medical insurance catalogs, are subject to price controls in the form
of fixed retail prices or retail price ceilings. In addition, the retail prices
of these products are also subject to periodic downward adjustments as the PRC
governmental authorities seek to make pharmaceutical products more affordable to
the general public. Since May 1998, the relevant PRC governmental authorities
have ordered price reductions of thousands of pharmaceutical products. The
latest price reduction occurred in October 2008 and affected 1,357 different
pharmaceutical products. Any future price controls or government mandated price
reductions may have a material adverse affect on our financial condition and
results of operations, including significantly reducing our revenue and
profitability.
OUR
OPERATING RESULTS MAY FLUCTUATE AS A RESULT OF FACTORS BEYOND OUR
CONTROL.
Our
operating results may fluctuate significantly in the future as a result of a
variety of factors, many of which are beyond our control. These factors
include:
|
·
|
the
costs of pharmaceutical products and
development;
|
|
·
|
the
relative speed and success with which we can obtain and maintain
customers, merchants and vendors for our services and manufacturers and
suppliers of products to market to our
customers;
|
|
·
|
capital
expenditures for equipment;
|
|
·
|
marketing
and promotional activities and other
costs;
|
|
·
|
changes
in our pricing policies, suppliers and
competitors;
|
|
·
|
the
ability of our suppliers to provide products in a timely manner to our
customers;
|
|
·
|
changes
in operating expenses;
|
|
·
|
increased
competition in our markets; and
|
|
·
|
other
general economic and seasonal
factors.
|
WE
MAY BE SUBJECT TO FINES AND PENALTIES IF WE FAIL TO COMPLY WITH THE APPLICABLE
PRC LAWS AND REGULATIONS GOVERNING SALES OF MEDICINES UNDER THE PRC NATIONAL
MEDICAL INSURANCE PROGRAM.
Eligible
participants in the PRC national medical insurance program, mainly consisting of
urban residents in China, are entitled to buy medicines using their medical
insurance cards in an authorized pharmacy, provided that the medicines they
purchase have been included in the national or provincial medical insurance
catalogs. The pharmacy, in turn, obtains reimbursement from the relevant
government social security bureaus. Moreover, the applicable PRC laws, rules and
regulations prohibit pharmacies from selling goods other than pre-approved
medicines when purchases are made with medical insurance cards, as well as
providing cash to the customer for the medical insurance card. We have
established procedures to prohibit our drugstores from selling unauthorized
goods to customers who make purchases with medical insurance cards. However, we
cannot assure you that those procedures will be strictly followed by all of our
employees in all of our stores. If we fail to observe the above laws,
rules and regulations with respect to purchases made with medical insurance
cards, we may be fined and our qualification for selling medicines included in
the national or provincial medical insurance catalogs may be withdrawn by
competent authorities.
OUR
RETAIL OPERATIONS REQUIRE A NUMBER OF PERMITS AND LICENSES IN ORDER TO CARRY ON
THEIR BUSINESS.
Drugstores
in China are required to obtain certain permits and licenses from various PRC
governmental authorities, including GSP certification. We are also required to
obtain food hygiene certificates for the distribution of nutritional supplements
and food products other than medicine. We cannot assure you that we can maintain
all required licenses, permits and certifications to carry on our business at
all times, and from time to time we may have not been in compliance with all
such required licenses, permits and certifications. Moreover, these licenses,
permits and certifications are subject to periodic renewal and/or reassessment
by the relevant PRC governmental authorities and the standards of such renewal
or reassessment may change from time to time. We intend to apply for the renewal
of these licenses, permits and certifications when required by then applicable
laws and regulations. Any failure by us to obtain and maintain all licenses,
permits and certifications necessary to carry on our business at any time could
have a material adverse effect on our business, financial condition and results
of operations. In addition, any inability to renew these licenses, permits and
certifications could severely disrupt our business, and prevent us from
continuing to carry on our business. Any changes in the standards used by
governmental authorities in considering whether to renew or reassess our
business licenses, permits and certifications, as well as any enactment of new
regulations that may restrict the conduct of our business, may also decrease our
revenue and/or increase our costs and materially reduce our profitability and
prospects. Furthermore, if the interpretation or implementation of existing laws
and regulations changes or if new regulations come into effect requiring us to
obtain any additional licenses, permits or certifications that were previously
not required to operate our existing businesses, we cannot assure you that we
may successfully obtain such licenses, permits or certifications.
WE
MAY HAVE DIFFICULTY DEFENDING OUR INTELLECTUAL PROPERTY RIGHTS FROM INFRINGEMENT
RESULTING IN LAWSUITS REQUIRING US TO DEVOTE FINANCIAL AND MANAGEMENT RESOURCES
THAT WOULD HAVE A NEGATIVE IMPACT ON OUR OPERATING RESULTS.
We regard
our trademarks and other similar intellectual properties as critical to our
success. We rely on trademark and other similar intellectual properties, as well
as confidentiality and license agreements with certain of our employees,
customers and others to protect our proprietary rights. We have received
trademark registration for certain of our products in the PRC. No assurance can
be given that our licenses will not be challenged, invalidated, infringed or
circumvented, or that our intellectual property rights will provide competitive
advantages to us. There can be no assurance that we will be able to obtain a
license from a third-party technology that we may need to conduct our business
or that such technology can be licensed at a reasonable cost.
Presently,
we sell our products mainly in PRC. To date, no trademark or patent filings have
been made other than in PRC. To the extent that we market our products in other
countries, we may have to take additional action to protect our intellectual
property. The measures we take to protect our proprietary rights may be
inadequate and we cannot give you any assurance that our competitors will not
independently develop formulations and processes that are substantially
equivalent or superior to our own or copy our products.
WE
MAY BE ADVERSELY AFFECTED BY COMPLEXITY, UNCERTAINTIES AND CHANGES IN CHINESE
REGULATION OF DRUGSTORES AND THE PRACTICE OF MEDICINE.
The
Chinese government regulates drugstores, including foreign ownership, and the
licensing and permit requirements. These laws and regulations are relatively new
and evolving, and their interpretation and enforcement involve significant
uncertainty. As a result, in certain circumstances it may be difficult to
determine what actions or omissions may be deemed to be a violation of
applicable laws and regulations. Issues, risks and uncertainties relating to
Chinese government regulation of the industry include the
following:
|
·
|
We
only have 49% ownership interest in BCT Retail. We are not able to own
100% interest in BCT Retail due to the restriction of foreign investment
in pharmacy chains with 30 or more drugstores and foreign ownership of
medical practices; and
|
|
·
|
uncertainties
relating to the regulation of drugstores, including evolving licensing
practices, means that permits, licenses or operations at our company may
be subject to challenge. This may disrupt our business, or subject us to
sanctions, requirements to increase capital or other conditions or
enforcement, or compromise enforceability of related contractual
arrangements, or have other harmful effects on
us.
|
The
interpretation and application of existing Chinese laws, regulations and
policies and possible new laws, regulations or policies have created substantial
uncertainties regarding the legality of existing and future foreign investments
in, and the businesses and activities of, pharmaceutical businesses in China,
including our business.
THERE
IS NO ASSURANCE THAT WE WILL PAY DIVIDENDS TO SHAREHOLDERS IN THE
FUTURE.
Although
Liuzhou BCT, our wholly owned subsidiary, declared and paid to its then existing
shareholders cash dividends in the amount of $6,940,000 and $2,044,056 for the
years ended December 31, 2008 and 2007, respectively, our board of directors
does not intend to distribute dividends in the near future. The declaration,
payment and amount of any future dividends will be made at the discretion of the
board of directors, and will depend upon, among other things, the results of our
operations, cash flows and financial condition, operating and capital
requirements, and other factors as the board of directors considers relevant.
There is no assurance that future dividends will be paid, and, if dividends are
paid, there is no assurance with respect to the amount of any such
dividend.
WE
MAY INCUR SIGNIFICANT COSTS TO ENSURE COMPLIANCE WITH UNITED STATES CORPORATE
GOVERNANCE AND ACCOUNTING REQUIREMENTS.
We may
incur significant costs associated with our public company reporting
requirements, costs associated with newly applicable corporate governance
requirements, including requirements under the Sarbanes-Oxley Act of 2002 and
other rules implemented by the Securities and Exchange Commission. We expect all
of these applicable rules and regulations to significantly increase our legal
and financial compliance costs and to make some activities more time consuming
and costly. We also expect that these applicable rules and regulations may make
it more difficult and more expensive for us to obtain director and officer
liability insurance and we may be required to accept reduced policy limits and
coverage or incur substantially higher costs to obtain the same or similar
coverage. As a result, it may be more difficult for us to attract and retain
qualified individuals to serve on our board of directors or as executive
officers. We are currently evaluating and monitoring developments with respect
to these newly applicable rules, and we cannot predict or estimate the amount of
additional costs we may incur or the timing of such costs.
Risks
Relating to the People’s Republic of China
THE
SALES PRICES OF SOME MEDICINES ARE CURRENTLY CONTROLLED BY THE CHINESE
GOVERNMENT AND THAT MAY ADVERSELY AFFECT OUR BUSINESS.
Prices
paid by end consumers for many of our medicines are regulated by PRC’s State
Development and Reform Commission. PRC justifies its need to control the drug
prices on the basis that, at present, only employees at state or private
companies have health insurance. About 900 million rural Chinese people and 35
million urban unemployed Chinese people lack insurance coverage and cannot
afford expensive drugs. Our future profitability might suffer if a significant
portion of our revenues were to be derived from products whose final selling
prices were state-controlled and if those prices were held at levels close to or
below our cost of sales.
SALES
OF OUR PRODUCTS COULD BE HARMED BY THE WIDESPREAD PRESENCE OF COUNTERFEIT
MEDICATION IN PRC NEGATIVELY IMPACTING OUR PROFITABILITY.
Chinese
counterfeiting of pharmaceuticals and other products affecting public health has
grown in tandem with counterfeiting and piracy of goods such as brand-name
clothing, compact discs and computer software. This situation negatively affects
China Baicaotang and other major domestic and foreign drug manufacturers in PRC,
especially for products marketed through the over the counter rather
than hospital channel. With the expansion of our business and increased
recognition of our brand name, such risks may increase. Currently,
active pharmaceutical ingredients are governed only by chemical regulations. Our
ability to increase sales as rapidly as we would like, and our profitability,
could be affected if this problem persists or worsens.
SUBSTANTIALLY
ALL OF OUR OPERATING ASSETS ARE LOCATED IN CHINA AND SUBSTANTIALLY ALL OF OUR
REVENUE WILL BE DERIVED FROM OUR OPERATIONS IN CHINA SO OUR BUSINESS, RESULTS OF
OPERATIONS AND PROSPECTS ARE SUBJECT TO THE ECONOMIC, POLITICAL AND LEGAL
POLICIES, DEVELOPMENTS AND CONDITIONS IN CHINA.
The PRC’s
economic, political and social conditions, as well as government policies, could
impair our business. The PRC economy differs from the economies of
most developed countries in many respects. China’s GDP has grown
consistently since 1978 (National Bureau of Statistics of
China). However, we cannot assure you that such growth will be
sustained in the future. If, in the future, China’s economy experiences a
downturn or grows at a slower rate than expected, there may be less demand for
spending in certain industries. A decrease in demand for spending in certain
industries could impair our ability to remain profitable. The PRC’s
economic growth has been uneven, both geographically and among various sectors
of the economy. The PRC government has implemented various measures to encourage
economic growth and guide the allocation of resources. Some of these measures
benefit the overall PRC economy, but may have a negative effect on
us. For example, our financial condition and results of operations
may be hindered by PRC government control over capital investments or changes in
tax regulations.
The PRC
economy has been transitioning from a planned economy to a more market-oriented
economy. Although in recent years the PRC government has implemented measures
emphasizing the use of market forces for economic reform, the reduction of state
ownership of productive assets and the establishment of sound corporate
governance in business enterprises, a substantial portion of productive assets
in China is still owned by the PRC government. In addition, the PRC government
continues to play a significant role in regulating industry development by
imposing industrial policies. It also exercises significant control over PRC
economic growth through the allocation of resources, controlling payment of
foreign currency-denominated obligations, setting monetary policy and providing
preferential treatment to particular industries or companies.
IF
THE MINISTRY OF COMMERCE, OR MOFCOM, CHINA SECURITIES REGULATORY COMMISSION, OR
CSRC, OR ANOTHER PRC REGULATORY AGENCY, DETERMINES THAT MOFCOM AND CSRC APPROVAL
OF OUR MERGER WAS REQUIRED OR IF OTHER REGULATORY OBLIGATIONS ARE IMPOSED UPON
US, WE MAY INCUR SANCTIONS, PENALTIES OR ADDITIONAL COSTS WHICH WOULD DAMAGE OUR
BUSINESS.
On August
8, 2006, six PRC regulatory agencies, including the MOFCOM and the CSRC,
promulgated the Rules on Acquisition of Domestic Enterprises by Foreign
Investors, or the M&A Rules, a new regulation with respect to the mergers
and acquisitions of domestic enterprises by foreign investors that became
effective on September 8, 2006. Article 11 of the New M&A Rules
requires when a domestic company, enterprise or natural person uses an offshore
company legally established or controlled by the domestic company, enterprise or
natural person to engage in the merger and acquisition of a related domestic
company, the application must be submitted to MOFCOM for
approval. Article 40 of the New M&A Rules requires that an
offshore special purpose vehicle formed for overseas listing purposes and
controlled directly or indirectly by PRC companies or individuals should obtain
the approval of the CSRC prior to the listing and trading of such offshore
special purpose vehicle’s securities on an overseas stock exchange, especially
in the event that the offshore special purpose vehicle acquires shares of or
equity interests in the PRC companies in exchange for the shares of offshore
companies. Article 39 of the New M&A Rules defines an offshore
special purpose vehicle as an offshore company directly or indirectly controlled
by a PRC domestic company or natural person for the purpose of the offshore
listing of their equity interests in the domestic company. On
September 21, 2006, the CSRC published on its official website procedures and
filing requirements for offshore special purpose vehicles seeking CSRC approval
of their overseas listings.
We
believe, based on the opinion of our PRC legal counsel, Broad & Bright Law
Firm, that MOFCOM and CSRC approvals were not required for our merger
transaction or for the listing and trading of our securities on a trading market
because we are not an offshore special purpose vehicle that is directly or
indirectly controlled by PRC companies or individuals. Although the
merger and acquisition regulations provide specific requirements and procedures,
there are still many ambiguities in the meaning of many
provisions. Further regulations are anticipated in the future, but
until there has been clarification either by pronouncements, regulation or
practice, there is some uncertainty in the scope of the regulations and the
regulators have wide latitude in the enforcement of the regulations and approval
of transactions. If the MOFCOM, CSRC or another PRC regulatory agency
subsequently determines that the MOFCOM and CSRC approvals were required, we may
face sanctions by the MOFCOM, CSRC or another PRC regulatory
agency. If this happens, these regulatory agencies may impose fines
and penalties on our operations in China, limit our operating privileges in
China, delay or restrict the repatriation of the proceeds from this offering
into China, restrict or prohibit payment or remittance of dividends paid by
Liuzhou BCT, or take other actions that could damage our business, financial
condition, results of operations, reputation and prospects, as well as the
trading price of our securities.
THE
NEW M&A REGULATIONS ESTABLISH MORE COMPLEX PROCEDURES FOR SOME ACQUISITIONS
OF CHINESE COMPANIES BY FOREIGN INVESTORS, WHICH COULD MAKE IT MORE DIFFICULT
FOR US TO PURSUE GROWTH THROUGH ACQUISITION IN CHINA.
The New
M&A Rules establish additional procedures and requirements that could make
some acquisitions of PRC companies by foreign investors, such as ours, more
time-consuming and complex, including requirements in some instances that the
approval of the Ministry of Commerce shall be required for transactions
involving the shares of an offshore listed company being used as the acquisition
consideration by foreign investors. In the future, we may grow our
business in part by acquiring complementary businesses. Complying
with the requirements of the New M&A Rules to complete such transactions
could be time-consuming, and any required approval processes, including
obtaining approval from the Ministry of Commerce, may delay or inhibit our
ability to complete such transactions, which could affect our ability to expand
our business or maintain our market share.
UNDER
THE NEW EIT LAW, WE MAY BE CLASSIFIED AS A “RESIDENT ENTERPRISE” OF CHINA. SUCH
CLASSIFICATION WILL LIKELY RESULT IN UNFAVORABLE TAX CONSEQUENCES TO US AND OUR
NON-PRC STOCKHOLDERS.
On March
16, 2007, the National People’s Congress of China passed the New EIT Law, and on
November 28, 2007, the State Council of China passed the New EIT Law
Implementing Rules which took effect on January 1, 2008. Under the New EIT Law,
an enterprise established outside of China with “de facto management bodies”
within China is considered a “resident enterprise,” meaning that it can be
treated in a manner similar to a Chinese enterprise for enterprise income tax
purposes. The implementing rules of the New EIT Law define de facto
management as “substantial and overall management and control over the
production and operations, personnel, accounting, and properties” of the
enterprise.
On April
22, 2009, the State Administration of Taxation issued the Notice Concerning
Relevant Issues Regarding Cognizance of Chinese Investment Controlled
Enterprises Incorporated Offshore as Resident Enterprises pursuant to Criteria
of de facto Management Bodies, or the Notice, further interpreting the
application of the New EIT Law and its implementation non-Chinese enterprise or
group controlled offshore entities. Pursuant to the Notice, an
enterprise incorporated in an offshore jurisdiction and controlled by a Chinese
enterprise or group will be classified as a “non-domestically incorporated
resident enterprise” if (i) its senior management in charge of daily operations
reside or perform their duties mainly in China; (ii) its financial or personnel
decisions are made or approved by bodies or persons in China; (iii) its
substantial assets and properties, accounting books, corporate chops, board and
shareholder minutes are kept in China; and (iv) at least half of its directors
with voting rights or senior management often resident in China. A
resident enterprise would be subject to an enterprise income tax rate of 25% on
its worldwide income and must pay a withholding tax at a rate of 10% when paying
dividends to its non-PRC shareholders. However, it remains unclear as
to whether the Notice is applicable to an offshore enterprise incorporated by a
Chinese natural person. Nor are detailed measures on imposition of
tax from non-domestically incorporated resident enterprises are
available. Therefore, it is unclear how tax authorities will determine tax
residency based on the facts of each case.
We may be
deemed to be a resident enterprise by Chinese tax authorities. If the
PRC tax authorities determine that we are a “resident enterprise” for PRC
enterprise income tax purposes, a number of unfavorable PRC tax consequences
could follow. First, we may be subject to the enterprise income tax at a rate of
25% on our worldwide taxable income as well as PRC enterprise income tax
reporting obligations. In our case, this would mean that income such as interest
on financing proceeds and non-China source income would be subject to PRC
enterprise income tax at a rate of 25%. Second, although under the New EIT Law
and its implementing rules dividends paid to us from our PRC subsidiaries would
qualify as “tax-exempt income,” we cannot guarantee that such dividends will not
be subject to a 10% withholding tax, as the PRC foreign exchange control
authorities, which enforce the withholding tax, have not yet issued guidance
with respect to the processing of outbound remittances to entities that are
treated as resident enterprises for PRC enterprise income tax
purposes. Finally, it is possible that future guidance issued with
respect to the new “resident enterprise” classification could result in a
situation in which a 10% withholding tax is imposed on dividends we pay to our
non-PRC shareholders and with respect to gains derived by our non-PRC
shareholders from transferring our shares. We are actively monitoring
the possibility of “resident enterprise” treatment for the 2010 tax year and are
evaluating appropriate organizational changes to avoid this treatment, to the
extent possible.
If we
were treated as a “resident enterprise” by PRC tax authorities, we would be
subject to taxation in both the U.S. and China, and our PRC tax may not be
creditable against our U.S. tax.
IF
THE PRC IMPOSES RESTRICTIONS DESIGNED TO REDUCE INFLATION, FUTURE ECONOMIC
GROWTH IN THE PRC COULD BE SEVERELY CURTAILED WHICH COULD HURT OUR BUSINESS AND
PROFITABILITY.
While the
economy of the PRC has experienced rapid growth, this growth has been uneven
among various sectors of the economy and in different geographical areas of the
country. Rapid economic growth often can lead to growth in the supply
of money and rising inflation. In order to control inflation in the
past, the PRC has imposed controls on bank credits, limits on loans for fixed
assets and restrictions on state bank lending. Imposition of similar
restrictions may lead to a slowing of economic growth, a decrease in demand for
our steel products and generally damage our business and
profitability.
FLUCTUATIONS
IN EXCHANGE RATES COULD HARM OUR BUSINESS AND THE VALUE OF OUR
SECURITIES.
The value
of our ordinary shares will be indirectly affected by the foreign exchange rate
between U.S. dollars and RMB and between those currencies and other currencies
in which our sales may be denominated. Because substantially all of our earnings
and cash assets are denominated in RMB and the net proceeds from this offering
will be denominated and our financial results are reported in U.S. dollars,
fluctuations in the exchange rate between the U.S. dollar and the RMB will
affect the relative purchasing power of these proceeds, our balance sheet and
our earnings per share in U.S. dollars following this offering. In
addition, appreciation or depreciation in the value of the RMB relative to the
U.S. dollar would affect our financial results reported in U.S. dollar terms
without giving effect to any underlying change in our business or results of
operations. Fluctuations in the exchange rate will also affect the
relative value of any dividend we issue that will be exchanged into U.S. dollars
as well as earnings from, and the value of, any U.S. dollar-denominated
investments we make in the future. Since July 2005, the RMB has not
been pegged to the U.S. dollar. Although the People’s Bank of China regularly
intervenes in the foreign exchange market to prevent significant short-term
fluctuations in the exchange rate, the RMB may appreciate or depreciate
significantly in value against the U.S. dollar in the medium to long term.
Moreover, it is possible that in the future PRC authorities may lift
restrictions on fluctuations in the RMB exchange rate and lessen intervention in
the foreign exchange market.
Very
limited hedging transactions are available in China to reduce our exposure to
exchange rate fluctuations. To date, we have not entered into any hedging
transactions. While we may enter into hedging transactions in the future, the
availability and effectiveness of these transactions may be limited, and we may
not be able to successfully hedge our exposure at all. In addition, our foreign
currency exchange losses may be magnified by PRC exchange control regulations
that restrict our ability to convert RMB into foreign currencies.
EXCHANGE
CONTROLS THAT EXIST IN THE PRC MAY LIMIT OUR ABILITY TO UTILIZE OUR CASH FLOW
EFFECTIVELY.
We are
subject to the PRC’s rules and regulations on currency conversion. In
the PRC, the State Administration for Foreign Exchange, or SAFE, regulates the
conversion between Renminbi and foreign currencies. Currently, foreign
investment enterprises, or FIEs, are required to apply to the SAFE for “Foreign
Exchange Registration Certificates for FIEs.” As a result of our ownership of
Liuzhou BCT, Liuzhou BCT is a FIE. With such registration
certificates, which need to be renewed annually, FIEs are allowed to open
foreign currency accounts including a “current account” and “capital account.”
Currency conversion within the scope of the “current account,” such as
remittance of foreign currencies for payment of dividends, can be effected
without requiring the approval of the SAFE. However, conversion of
currency in the “capital account,” including capital items such as direct
foreign investment, loans and securities, still require approval of the SAFE.
Further, any capital contributions to Liuzhou BCT by its offshore shareholder
must be approved by the Ministry of Commerce in China or its local counterpart.
We cannot assure you that the PRC regulatory authorities will not impose further
restrictions on the convertibility of the Renminbi. Any future restrictions on
currency exchanges may limit our ability to use our cash flow for the
distribution of dividends to our shareholders or to fund operations it may have
outside of the PRC.
In August
2008, SAFE promulgated Circular 142, a notice regulating the conversion by FIEs
of foreign currency into Renminbi by restricting how the converted Renminbi may
be used. Circular 142 requires that Renminbi converted from the
foreign currency-dominated capital of a FIE may only be used for purposes within
the business scope approved by the applicable government authority and may not
be used for equity investments within the PRC unless specifically provided for
otherwise. In addition, SAFE strengthened its oversight over the flow
and use of Renminbi funds converted from the foreign currency-dominated capital
of a FIE. The use of such Renminbi may not be changed without
approval from SAFE, and may not be used to repay Renminbi loans if the proceeds
of such loans have not yet been used. Violations of Circular 142 may
result in severe penalties, including substantial fines as set forth in the SAFE
rules.
PRC
REGULATIONS RELATING TO THE ESTABLISHMENT OF OFFSHORE SPECIAL PURPOSE COMPANIES
BY PRC RESIDENTS MAY SUBJECT OUR PRC RESIDENT SHAREHOLDERS TO PERSONAL LIABILITY
AND LIMIT OUR ABILITY TO INJECT CAPITAL INTO OUR PRC SUBSIDIARIES, LIMIT OUR PRC
SUBSIDIARIES’ ABILITY TO DISTRIBUTE PROFITS TO US, OR OTHERWISE ADVERSELY AFFECT
US.
SAFE
issued a public notice in October 2005, or the SAFE notice, requiring PRC
residents to register with the local SAFE branch before establishing or
controlling any company outside of China for the purpose of capital financing
with assets or equities of PRC companies, referred to in the notice as an
“offshore special purpose company.” PRC residents that are shareholders of
offshore special purpose companies established before November 1, 2005 were
required to register with the local SAFE branch before March 31, 2006.The
failure of our beneficial owners to timely amend their SAFE registrations
pursuant to the SAFE notice or the failure of future beneficial owners of our
company who are PRC residents to comply with the registration procedures set
forth in the SAFE notice may subject such beneficial owners to fines and legal
sanctions and may also limit our ability to contribute additional capital into
our PRC subsidiaries, limit our PRC subsidiaries’ ability to distribute
dividends to our company or otherwise adversely affect our
business.
BECAUSE
CHINESE LAW GOVERNS MANY OF OUR MATERIAL AGREEMENTS, WE MAY NOT BE ABLE TO
ENFORCE OUR RIGHTS WITHIN THE PRC OR ELSEWHERE, WHICH COULD RESULT IN A
SIGNIFICANT LOSS OF BUSINESS, BUSINESS OPPORTUNITIES OR CAPITAL.
Chinese
law governs many of our material agreements, some of which may be with Chinese
governmental agencies. We cannot assure you that we will be able to enforce any
of our material agreements or that remedies will be available outside of the
PRC. The system of laws and the enforcement of existing laws and
contracts in the PRC may not be as certain in implementation and interpretation
as in the United States. The Chinese judiciary is relatively inexperienced in
enforcing corporate and commercial law, leading to a higher than usual degree of
uncertainty as to the outcome of any litigation. The inability to enforce or
obtain a remedy under any of our future agreements could result in a significant
loss of business, business opportunities or capital.
BECAUSE
OUR FUNDS ARE HELD IN BANKS IN UNINSURED PRC BANK ACCOUNTS, THE FAILURE OF ANY
BANK IN WHICH WE DEPOSIT OUR FUNDS COULD AFFECT OUR ABILITY TO CONTINUE IN
BUSINESS.
Funds on
deposit at banks and other financial institutions in the PRC are often
uninsured. A significant portion of our assets are in the form of
cash deposited with banks in the PRC, and in the event of a bank failure, we may
not have access to our funds on deposit. Depending upon the amount of
money we maintain in a bank that fails, our inability to have access to our cash
could impair our operations, and, if we are not able to access funds to pay our
suppliers, employees and other creditors, we may be unable to continue in
business.
OUR
BUSINESS COULD BE SEVERELY HARMED IF THE CHINESE GOVERNMENT CHANGES ITS
POLICIES, LAWS, REGULATIONS, TAX STRUCTURE OR ITS CURRENT INTERPRETATIONS OF ITS
LAWS, RULES AND REGULATIONS RELATING TO OUR OPERATIONS IN CHINA.
Our
business is located in Guangxi province, China and virtually all of our assets
are located in China. We generate our sales revenue only from
customers located in China. Our results of operations, financial
state of affairs and future growth are, to a significant degree, subject to
China’s economic, political and legal development and related uncertainties. Our
operations and results could be materially affected by a number of factors,
including, but not limited to
|
·
|
Changes
in policies by the Chinese government resulting in changes in laws or
regulations or the interpretation of laws or
regulations,
|
|
·
|
changes
in taxation,
|
|
·
|
changes
in employment restrictions,
|
|
·
|
import
duties, and
|
|
·
|
currency
revaluation.
|
Over the
past several years, the Chinese government has pursued economic reform policies
including the encouragement of private economic activities and greater economic
decentralization. If the Chinese government does not continue to pursue its
present policies that encourage foreign investment and operations in China, or
if these policies are either not successful or are significantly altered, then
our business could be harmed. Following the Chinese government’s
policy of privatizing many state-owned enterprises, the Chinese government has
attempted to augment its revenues through increased tax
collection. It also exercises significant control over China’s
economic growth through the allocation of resources, controlling payment of
foreign currency-denominated obligations, setting monetary policy and providing
preferential treatment to particular industries or
companies. Continued efforts to increase tax revenues could result in
increased taxation expenses being incurred by us. Economic
development may be limited as well by the imposition of austerity measures
intended to reduce inflation, the inadequate development of infrastructure and
the potential unavailability of adequate power and water supplies,
transportation and communications. In addition, the Chinese
government continues to play a significant role in regulating industry by
imposing industrial policies.
THE
CHINESE LAWS AND REGULATIONS WHICH GOVERN OUR CURRENT BUSINESS OPERATIONS ARE
SOMETIMES VAGUE AND UNCERTAIN AND MAY BE CHANGED IN A WAY THAT HURTS OUR
BUSINESS.
China’s
legal system is a civil law system based on written statutes, in which system
decided legal cases have little value as precedents, unlike the common law
system prevalent in the United States. There are substantial uncertainties
regarding the interpretation and application of Chinese laws and regulations,
including but not limited to the laws and regulations governing our business, or
the enforcement and performance of our arrangements with customers in the event
of the imposition of statutory liens, death, bankruptcy and criminal
proceedings. The Chinese government has been developing a
comprehensive system of commercial laws, and considerable progress has been made
in introducing laws and regulations dealing with economic matters such as
foreign investment, corporate organization and governance, commerce, taxation
and trade. However, because these laws and regulations are relatively
new, and because of the limited volume of published cases and judicial
interpretation and their lack of force as precedents, interpretation and
enforcement of these laws and regulations involve significant uncertainties. New
laws and regulations that affect existing and proposed future businesses may
also be applied retroactively. We are considered an FIE under
Chinese laws, and as a result, we must comply with Chinese laws and
regulations. We cannot predict what effect the interpretation of
existing or new Chinese laws or regulations may have on our
business. If the relevant authorities find us to be in violation of
Chinese laws or regulations, they would have broad discretion in dealing with
such a violation, including, without limitation: levying fines; revoking our
business and other licenses; requiring that we restructure our ownership or
operations; and requiring that we discontinue any portion or all of our
business.
A
SLOWDOWN OR OTHER ADVERSE DEVELOPMENTS IN THE CHINESE ECONOMY MAY MATERIALLY AND
ADVERSELY AFFECT OUR CUSTOMERS’ DEMAND FOR OUR SERVICES AND OUR
BUSINESS.
All of
our operations are conducted in China and all of our revenues are generated from
sales to businesses operating in China. Although the Chinese economy
has grown significantly in recent years, such growth may not continue. We do not
know how sensitive we are to a slowdown in economic growth or other adverse
changes in Chinese economy which may affect demand for precision steel
products. A slowdown in overall economic growth, an economic downturn
or recession or other adverse economic developments in China may materially
reduce the demand for our services and in turn reduce our results of
operations.
FAILURE
TO COMPLY WITH THE U.S. FOREIGN CORRUPT PRACTICES ACT AND CHINESE
ANTI-CORRUPTION LAWS COULD SUBJECT US TO PENALTIES AND OTHER ADVERSE
CONSEQUENCES.
Our
executive officers, employees and other agents may violate applicable law in
connection with the marketing or sale of our products, including China’s
anti-corruption laws and the U.S. Foreign Corrupt Practices Act, or the FCPA,
which generally prohibits United States companies from engaging in bribery or
other prohibited payments to foreign officials for the purpose of obtaining or
retaining business. In addition, we are required to maintain records
that accurately and fairly represent our transactions and have an adequate
system of internal accounting controls. Foreign companies, including
some that may compete with us, are not subject to these prohibitions, and
therefore may have a competitive advantage over us. The PRC also strictly
prohibits bribery of government officials. However, corruption,
extortion, bribery, pay-offs, theft and other fraudulent practices occur from
time-to-time in the PRC.
While we
intend to implement measures to ensure compliance with the FCPA and Chinese
anti-corruption laws by all individuals involved with our company, our employees
or other agents may engage in such conduct for which we might be held
responsible. If our employees or other agents are found to have
engaged in such practices, we could suffer severe penalties and other
consequences that may have a material adverse effect on our business, financial
condition and results of operations. In addition, our brand and
reputation, our sales activities or the price of our ordinary shares could be
adversely affected if we become the target of any negative publicity as a result
of actions taken by our employees or other agents.
THE
IMPLEMENTATION OF THE NEW PRC EMPLOYMENT CONTRACT LAW AND INCREASES IN THE LABOR
COSTS IN CHINA MAY HURT OUR BUSINESS AND PROFITABILITY.
A new
employment contract law became effective on January 1, 2008 in China. It imposes
more stringent requirements on employers in relation to entry into fixed-term
employment contracts, recruitment of temporary employees and dismissal of
employees. In addition, under the newly promulgated Regulations on Paid Annual
Leave for Employees, which also became effective on January 1, 2008, employees
who have worked continuously for more than one year are entitled to paid
vacation ranging from 5 to 15 days, depending on the length of the employee’s
service. Employees who waive such vacation entitlements at the request of the
employer will be compensated for three times their normal daily salaries for
each vacation day so waived. As a result of the new law and regulations, our
labor costs may increase. There is no assurance that disputes, work stoppages or
strikes will not arise in the future. Increases in the labor costs or future
disputes with our employees could damage our business, financial condition or
operating results.
UNDER
THE NEW EIT LAW, CHINA BAICAOTANG AND LIUZHOU BCT MAY BE CLASSIFIED AS “RESIDENT
ENTERPRISES” OF CHINA, WHICH MAY SUBJECT CHINA BAICAOTANG AND LIYZHOU BAICAOTANG
TO PRC INCOME TAX ON THEIR TAXABLE GLOBAL INCOME.
China
passed a new Enterprise Income Tax Law, or the New EIT Law, and its
implementation regulations, both of which became effective on January 1,
2008. Under the New EIT Law, an enterprise established outside of
China with “de facto management bodies” within China is considered a “resident
enterprise,” meaning that it can be treated in a manner similar to a Chinese
domestic enterprise for enterprise income tax purposes. The implementing rules
of the New EIT Law
define de
facto management as “substantial and overall management and control over the
production and operations, personnel, accounting, and properties” of the
enterprise. On April 22, 2009, the State Administration of Taxation
issued the Notice Concerning Relevant Issues Regarding Cognizance of Chinese
Investment Controlled Enterprises Incorporated Offshore as Resident Enterprises
pursuant to Criteria of de facto Management Bodies, or the Notice, further
interpreting the application of the New EIT Law and its implementation with
respect to non-Chinese enterprises or group controlled offshore
entities. Pursuant to the Notice, an enterprise incorporated in an
offshore jurisdiction and controlled by a Chinese enterprise or group will be
classified as a “non-domestically incorporated resident enterprise” if (i) its
senior management in charge of daily operations reside or perform their duties
mainly in China; (ii) its financial or personnel decisions are made or approved
by bodies or persons in China; (iii) substantial assets and properties,
accounting books, corporate chops, board and shareholder minutes are kept in
China; and (iv) at least half of its directors with voting rights or senior
management often resident in China. A resident enterprise would be
generally subject to the uniform 25% enterprise income tax rate as to its
worldwide income. Although the Notice is directly applicable to
enterprises registered in an offshore jurisdiction and controlled by Chinese
domestic enterprises or groups, it is uncertain whether the PRC tax authorities
will make reference to the Notice when determining the resident status of other
offshore companies, such as Liuyzhou Baicaotang. Since substantially all of our
management is currently based in China, it is likely we may be treated as a
Chinese resident enterprise for enterprise income tax purposes. The
tax consequences of such treatment are currently unclear, as they will depend on
how local tax authorities apply or enforce the New EIT Law or the implementation
regulations.
In
addition, under the New EIT Law and implementation regulations, PRC income tax
at the rate of 10% is applicable to dividends payable to investors that are
“non-resident enterprises” (and that do not have an establishment or place of
business in the PRC, or that have such establishment or place of business but
the relevant income is not effectively connected with the establishment or place
of business) to the extent that such dividends have their source within the PRC
unless there is an applicable tax treaty between the PRC and the jurisdiction in
which an overseas holder resides which reduces or exempts the relevant
tax. Similarly, any gain realized on the transfer of shares by such
investors is also subject to the 10% PRC income tax if such gain is regarded as
income derived from sources within the PRC.
If we are
considered a PRC “resident enterprise”, it is unclear whether the dividends we
pay with respect to our shares, or the gain you may realize from the transfer of
our shares, would be treated as income derived from sources within the PRC and
be subject to PRC tax. If we are required under the New EIT Law to
withhold PRC income tax on our dividends payable to our foreign shareholders, or
if you are required to pay PRC income tax on the transfer of your shares, the
value of your investment in our shares may be materially and adversely
affected.
IT
MAY BE DIFFICULT TO AFFECT SERVICE OF PROCESS AND ENFORCEMENT OF LEGAL JUDGMENTS
UPON OUR COMPANY AND OUR OFFICERS AND DIRECTORS BECAUSE THEY RESIDE OUTSIDE THE
UNITED STATES.
As our
operations are presently based in PRC and a majority of our directors and all of
our officers reside in PRC, service of process on our company and such directors
and officers may be difficult to effect within the United States. Also, our main
assets are located in PRC and any judgment obtained in the United States against
us may not be enforceable outside the United States.
Risks
Associated with our Common Stock in General
OUR
SHARES OF COMMON STOCK HAVE LIMITED TRADING AND THERE CAN BE NO ASSURANCE THAT
THERE WILL BE AN ACTIVE MARKET FOR OUR SHARES OF COMMON STOCK EITHER NOW OR IN
THE FUTURE.
Our
shares of Common Stock began trading on the OTCBB on March 3, 2010 and there has
been limited trading and the price if traded may not reflect our value. There
can be no assurance that there will be an active market for our shares of Common
Stock either now or in the future. The market liquidity will be dependent on the
perception of our operating business and any steps that our management might
take to bring us to the awareness of investors. There can be no assurance given
that there will be any awareness generated. Consequently, investors may not be
able to liquidate their investment or liquidate it at a price that reflects the
value of the business. If a more active market should develop, the price may be
highly volatile. Because there may be a low price for our shares of Common
Stock, many brokerage firms may not be willing to effect transactions in the
securities. Even if an investor finds a broker willing to effect a transaction
in the shares of our Common Stock, the combination of brokerage commissions,
transfer fees, taxes, if any, and any other selling costs may exceed the selling
price. Further, many lending institutions will not permit the use of such shares
of Common Stock as collateral for any loans.
WE
MAY BE SUBJECT TO THE PENNY STOCK RULES WHICH WILL MAKE THE SHARES OF OUR COMMON
STOCK MORE DIFFICULT TO SELL.
We may be
subject now and in the future to the SEC’s “penny stock” rules if our shares of
Common Stock sell below $5.00 per share. Penny stocks generally are
equity securities with a price of less than $5.00. The penny stock rules require
broker-dealers to deliver a standardized risk disclosure document prepared by
the SEC which provides information about penny stocks and the nature and level
of risks in the penny stock market. The broker-dealer must also provide the
customer with current bid and offer quotations for the penny stock, the
compensation of the broker-dealer and its salesperson, and monthly account
statements showing the market value of each penny stock held in the customer’s
account. The bid and offer quotations, and the broker-dealer and salesperson
compensation information must be given to the customer orally or in writing
prior to completing the transaction and must be given to the customer in writing
before or with the customer’s confirmation.
In
addition, the penny stock rules require that prior to a transaction, the broker
dealer must make a special written determination that the penny stock is a
suitable investment for the purchaser and receive the purchaser’s written
agreement to the transaction. The penny stock rules are
burdensome and may reduce purchases of any offerings and reduce the trading
activity for shares of our Common Stock. As long as our shares of Common Stock
are subject to the penny stock rules, the holders of such shares of Common Stock
may find it more difficult to sell their securities.
OUR
SHAREHOLDERS WILL EXPERIENCE DILUTION AS A RESULT OF THE CONVERSION OF OUR CLASS
A WARRANTS OR ISSUANCE OF SECURITIES IN FUTURE FINANCINGS.
As of the
date hereof, we have Investor Warrants and Agent Warrants outstanding which are
exercisable for 2,111,235 shares of common stock. To the extent such warrants
are exercised and converted, there will be further dilution. In addition, in the
event that any future financing should be in the form of securities convertible
into, or exchangeable for, equity securities, investors may experience
additional dilution upon the conversion or exchange of such
securities.
There are
additional authorized but unissued shares of our Common Stock that may be later
issued by our management for any purpose without the consent or vote of the
stockholders. Our current shareholders may be further diluted in their
percentage ownership in the event additional shares are issued by us in the
future.
OUR
COMMON STOCK IS SUBJECT TO PRICE VOLATILITY UNRELATED TO OUR
OPERATIONS.
The
market price of our common stock could fluctuate substantially due to a variety
of factors, including market perception of our ability to achieve our planned
growth, quarterly operating results of other companies in the same industry,
trading volume in our common stock, changes in general conditions in the economy
and the financial markets or other developments affecting our competitors or us.
In addition, the stock market is subject to extreme price and volume
fluctuations. This volatility has had a significant effect on the market price
of securities issued by many companies for reasons unrelated to their operating
performance and could have the same effect on our common stock.
OUR
CHIEF FINANCIAL OFFICER OWNS A SUBSTANTIAL PORTION OF OUR OUTSTANDING COMMON
STOCK, WHICH WILL ENABLE HER TO INFLUENCE MANY SIGNIFICANT CORPORATE ACTIONS AND
IN CERTAIN CIRCUMSTANCES MAY PREVENT A CHANGE IN CONTROL THAT WOULD OTHERWISE BE
BENEFICIAL TO OUR SHAREHOLDERS.
As of the
date hereof, our Chief Financial Officer, Ms. Zhang, controls approximately
58.9%of our outstanding shares of common stock that are entitled to vote on all
corporate actions. These stockholders, acting together, could have a substantial
impact on matters requiring the vote of the shareholders, including the election
of our directors and most of our corporate actions. This control could delay,
defer or prevent others from initiating a potential merger, takeover or other
change in our control, even if these actions would benefit our shareholders and
us. This control could adversely affect the voting and other rights of our other
shareholders and could depress the market price of our common
stock.
IF
WE FAIL TO MAINTAIN AN EFFECTIVE SYSTEM OF INTERNAL CONTROLS, WE MAY NOT BE ABLE
TO ACCURATELY REPORT OUR FINANCIAL RESULTS OR PREVENT FRAUD.
Since we
operated as a private enterprise without public reporting obligations prior to
the Share Exchange, we have committed limited personnel and resources to the
development of the external reporting and compliance obligations that would be
required of a public company. If our financial reporting systems or procedures
fail, we may not be able to provide accurate financial statements on a timely
basis or comply with the Sarbanes-Oxley Act of 2002 as it applies to us. Any
failure of our ability to provide accurate financial statements could cause the
trading price of our common stock to decrease substantially.
|
Unresolved
Staff Comments
|
Not
Applicable.
All land
in the PRC is owned by the government and cannot be sold to any individual or
entity. Instead, the government grants or allocates landholders a “land use
right,” which we sometimes refer to informally as land ownership. There are four
ways of acquiring land use rights in the PRC:
|
·
|
Grant
of the right to use land;
|
|
·
|
Assignment
of the right to use land;
|
|
·
|
Lease
of the right to use land; and
|
|
·
|
Allocation
of the right to use land.
|
Granted
land use rights are provided by the government in exchange for a grant fee, and
carry the rights to pledge, mortgage, lease and transfer the land within the
term of the grant. Land is granted for a fixed term, generally 70 years for
residential use, 50 years for industrial use, and 40 years for commercial and
other use. The term is renewable in theory. Unlike in western nations, granted
land must be used for the specific purpose for which it was
granted.
Allocated
land use rights are generally provided by the government for an indefinite
period (usually to state-owned entities) and cannot be pledged, mortgaged,
leased, or transferred by the user unless otherwise approved by the competent
government authorities. Allocated land can be reclaimed by the government at any
time. Allocated land use rights may be converted into granted land use rights
upon the payment of a grant fee to the government.
Our land
use rights are set forth below:
Liuzhou
BCT
Liuzhou Baicaotang owns properties listed below:
|
1
|
2,753.5
square meter land use rights, the term of which is to be expired on
November 14, 2053, located at No. 102, Chengzhan Road, Liuzhou City,
Guangxi Province, PRC, as its corporate headquarter.
|
|
2
|
Approximately
321.7 square meter land use rights, the term of which expires on March 8,
2047, located at Building 2, 197 No. 3 Middle Road, Liuzhou City, Guangxi
province;
|
|
3
|
Approximately
10.7 square meter land use rights, the term of which is to be expired on
December 28, 2044, located at No. 1-3 XingLong Building, Zhongshan Middle
Rd., Liuzhou City, Guangxi
province;
|
|
4
|
Approximately
75.2 square meter land use rights, the term of which is to be expired on
August 4, 2043, located at Floor 1, 197 No. 3 Middle Road, Liuzhou City,
Guangxi province;
|
|
5
|
Approximately
346.85 square meter land use rights, located at No. 10 Shizi Rd., Luzhai
Town, Luzhai County, Guangxi
province;
|
|
6
|
Approximately
5,655.6 square meter land use rights, the term of which is to be expired
on November 14, 2053, located at No. 6 Changfeng Rd. Liuzhou City, Guangxi
province;
|
|
7
|
Approximately
886.7 square meter land use rights, located at No. 4 Changfeng Rd.,
Liuzhou City, Guangxi province;
|
|
8
|
Approximately
1,308.5 square meter land use rights, the term of which is to be expired
on August 4, 2053, located at No. 4 Changfeng Rd., Liuzhou City, Guangxi
province;
|
|
9
|
Approximately
1,084.5 square meter land use rights, the term of which is to be expired
on November 14, 2053, located at No. 4 Changfeng Rd., Liuzhou City,
Guangxi province;
|
|
10
|
Approximately
1,558.05 square meter land use rights, the term of which is to be expired
on May 28, 2057, located at Desheng Village, Litang Town Bingyang County,
Guangxi province;
|
|
11
|
Approximately
380.6 square meter land use rights, the term of which is to be expired on
August 4, 2043, located at No. 15 May First Rd,, Liuzhou City, Guangxi
province; and
|
|
12
|
Approximately
2,048 square meter land use rights, the term of which is to be expired on
January 16, 2074, located at 9 District, Liupai Zhen, Tian’e County,
Guangxi province.
|
Hefeng
Pharmaceutical
Hefeng
Pharmaceutical owns a 44,982.18 square meter business facility located at 3
Development District, Donglan County, which is used as its principal executive
offices and plant.
BCT
Retail
The
principal executive offices of BCT Retail are located at No. 102, Chengzhan
Road, Liuzhou City, Guangxi Province, PRC. Such office space that Baicaotang
Retail is using is owned by Liuzhou BCT. Baicaotang Retail has used the
office space free of charge and without any lease agreement. Baicaotang Retail
currently operates 66 retail chain stores. Most of the chain stores managed by
Baicaotang Retail are located in the following towns, counties and
municipal cities: Liuzhou City, Ronghui County, Sangjiang County, Liujiang
County, Nandan County, Yongfu County, Bama County, Binyang County, Yongan
County, Laibin City, and Rongan County.
The space
of the retail chain stores ranges in size from 85 square meters to 168 square
meters. Except for two retail stores which are owned by Liuzhou BCT
and used by Baicaotang Retail free of charge and without any lease agreement,
all other chain stores are leased. The monthly rents
of the chain stores range from approximately $140.00 to $2,500.00, and most of
the leases have a three year term.
All the
lease agreements have similar terms and provisions.
Neither
we nor any of our subsidiaries is a party to any pending legal
proceedings.
None.
PART
II
|
Market
for Common Equity, Related Stockholder Matters and Issuer
Purchases of Equity Securities
|
Market
Information
Our
common stock is listed on the OCTBB under the symbol “CNBI”. Prior to
March 3, 2010, our common stock was not traded on the OCTBB. Since
March 3, 2010, the trading in our stock has been very limited. Consequently, our
historical prices may not be an accurate indication of the future prices of our
common stock.
The
following table sets forth the high and low bid prices, on the OTCBB, as
reported and summarized by the OTCBB from March 3 to March 29,
2010.
|
High
|
Low
|
|||||||
|
2010:
|
||||||||
|
March
3 through March 29, 2010
|
$ | 2.00 | $ | 2.00 | ||||
As of
March 29, 2010, the last reported sale price of our common stock was $2.75 per
share.
As of
March 25, 2010, there were 38,154,340 shares of our common stock outstanding and
we had approximately 182 shareholders of record of our common stock. This does
not reflect the number of persons or entities who hold stock in nominee or
“street” name through various brokerage firms.
Dividends
During
the fiscal years ended December 31, 2008, Liuzhou BCT declared and paid to its
original shareholders cash dividends in the aggregate amount of
$6,940,000.
The
declaration or payment of any future cash dividend will be at the discretion of
the Board and will depend upon the earnings (if any), capital requirements and
financial position of the company, general economic conditions, and other
pertinent factors. It is our present intention not to declare or pay any cash
dividends in the foreseeable future, but rather to reinvest earnings (if any),
in our business operations.]
Securities
authorized for issuance under equity compensation plans
We do not
have any equity compensation plans in effect.
Issuer
Purchases of Equity Securities
None.
|
Selected
Consolidated Financial Data
|
The
following selected consolidated statement of income and comprehensive income
data for the fiscal years ended December 31, 2009 and 200 and the
consolidated balance sheet data as of December 31, 2009 and 2008 and are derived
from the audited consolidated financial statements of China Baicaotang Medicine
Limited included elsewhere in this prospectus. The audited
consolidated financial statements have been prepared in accordance with U.S.
GAAP and have been audited by PKF, Certified Public Accountants, Hong Kong,
China, a member firm of PKF International Limited network of legally
independent firms. The results of operations for past
accounting periods are not necessarily indicative of the results to be expected
for any future periods.
|
Statements
of Income and Comprehensive Income
|
Year
ended December 31,
|
||||||
|
2009
|
2008
|
||||||
|
Revenue
|
|||||||
|
Sales
|
$
|
136,086,708
|
$
|
108,991,329
|
|||
|
Cost of sales
|
100,578,769
|
79,361,987
|
|||||
|
Gross profit
|
35,507,939
|
29,629,342
|
|||||
|
Operating
expenses
|
|||||||
|
Administrative
expenses
|
4,598,800
|
3,341,605
|
|||||
|
Research and development
expenses
|
99,688
|
763,995
|
|||||
|
Selling
expenses
|
3,865,980
|
2,122,153
|
|||||
|
8,564,468
|
6,227,753
|
||||||
|
Income
from operations
|
26,943,471
|
23,401,589
|
|||||
|
Interest income
|
16,100
|
29,315
|
|||||
|
Government
grants
|
29,320
|
-
|
|||||
|
Other income
|
95,345
|
143,426
|
|||||
|
Finance costs
|
(1,413,873
|
)
|
(1,260,290
|
)
|
|||
|
Income
before income taxes
|
25,670,363
|
22,314,040
|
|||||
|
Income
taxes
|
(6,261,798
|
)
|
(5,656,878
|
)
|
|||
|
Net
income attributable to China Baicaotang Medicine Limited
|
|||||||
|
common
stockholders
|
$19,408,565
|
$
|
16,657,162
|
||||
|
Other
comprehensive income
|
|||||||
|
Foreign currency translation
adjustments
|
57,322
|
1,142,614
|
|||||
|
Total
comprehensive income
|
$
|
19,465,887
|
$
|
17,799,776
|
|||
|
Earnings
per share: basic and diluted
|
$
|
0.61
|
$
|
0.52
|
|||
|
Weighted
average number of shares
|
|||||||
|
outstanding:
basic and diluted
|
32,013,943
|
31,963,669
|
|||||
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Balance
Sheet Data:
|
||||||||
|
Cash
and cash equivalents
|
$ | 13,304,158 | $ | 1,265,184 | ||||
|
Total
assets
|
$ | 93,060,792 | $ | 65,814,348 | ||||
|
Total
liabilities
|
$ | 39,682,483 | $ | 37,231,282 | ||||
|
Stockholders’
equity
|
$ | 53,378,309 | $ | 28,583,066 | ||||
|
|
||||||||
|
Total
liabilities and stockholders’ equity
|
$ | 93,060,792 | $ | 65,814,348 | ||||
|
Management’s
Discussion and Analysis of Financial Condition and Results of
Operations
|
The
following discussion should be read along with our financial statements and
notes thereto. This section includes a number of forward-looking statements that
reflect our current views with respect to future events and financial
performance. Forward-looking statements are often identified by words like
believe, expect, estimate, anticipate, intend, project and similar expressions,
or words which, by their nature, refer to future events. You should not place
undue certainty on these forward-looking statements. These forward-looking
statements are subject to certain risks and uncertainties that could cause
actual results to differ materially from our predictions.
Business
Review
We are
engaged in pharmaceutical distribution, retail pharmacy and manufacture of
pharmaceuticals and medical-related products through our three subsidiaries
Liuzhou BCT, Hefeng Pharmaceutical, and BCT Retail, each of which is located in
Guangxi Province, China.
We have
integrated operations in the following three business segments.
Pharmaceutical
distribution segment:
We
provide a comprehensive offering of pharmaceutical and healthcare product,
included branded and generic prescription medicines, over-the counter medicines,
Western and Chinese medicines, as well as personal care products and medical
supplies, Chinese herbs, and medical instrument from manufacturers
and suppliers through distribution to its customers, including hospitals, retail
drug stores, other pharmaceutical wholesalers, clinics, medical centers, and
individuals located mainly in Guangxi Province except for the other
pharmaceutical wholesalers. Over 8,000 products are distributed in
compliance with China’s regulations over the pharmaceutical industry. For
the year ended December 31, 2009, our pharmaceutical distribution segment
accounted for approximately 71.4% of our total revenue after elimination of
inter-segment sales.
Revenue
derived from Chinese herbal medicine, family planning products, medical
instruments, injection drugs and other packaged medicine
drugs constituted 0.96%, 0.1%, 0.49%, 19.06% and 79.38% of our
pharmaceutical distribution segment’s total revenue, respectively.
Retail
pharmacy segment:
Baicaotang
Retail operates the largest regional wholesale network in Guangxi province,
consisting of 66 directly owned retail stores in Guangxi province under the
registered name “Baicaotang 百草堂.” Our retail
stores provide high-quality convenient and professional pharmaceutical services,
and supply a wide variety of medicines for selling prescription medicines,
over-the-counter medicines, Chinese herbal medicine, roughly processed Chinese
herbal medicine, family planning products, and other pharmaceutical products and
healthcare products. For the year ended
December 31, 2009, our retail pharmacy segment accounted for approximately 22.9%
of our total revenue after elimination of the inter-segment
sales.
Manufacturing
pharmacy segment:
Located
in Donglan District, Guangxi province and built on approximately 40,000 square
meters of land which we own, Hefeng Pharmaceutical has four product processing
units: (1) Chinese herbal medicine abstraction unit for raw material and
medicine paste with 100 tons of annual abstraction capacity; (2) granular
formulation unit with an annual production capacity of 2 billion packages; (3)
pill formulation unit with annual production capacity of 3 billion pills, and
(4) liquid formulation unit with an annual production capacity of 0.1 billion
injections. We manufacture and sell both the generic and clinic drugs all over
the China. For the year ended December 31, 2009, our manufacturing
pharmacy segment accounted for approximately 5.7% of our total revenue after
elimination of the inter-segment sales.
RESULTS
OF OPERATIONS
The
following table sets for the key components of our results of operations for the
periods indicated .
|
Year
ended December 31
|
|||||||||||
|
2009
|
2008
|
||||||||||
|
’000
|
%
of total sales
|
’000
|
%
of total sales
|
||||||||
|
Sales
revenue
|
$
|
136,087
|
$
|
108,991
|
|||||||
|
Cost
of sales
|
100,579
|
73.9
|
79,362
|
72.8
|
|||||||
|
Gross
profit
|
35,508
|
26.1
|
29,629
|
27.2
|
|||||||
|
Operating
expenses
|
|||||||||||
|
Administrative
expenses
|
4,599
|
3.4
|
3,341
|
3.1
|
|||||||
|
Research
and development expenses
|
100
|
-
|
764
|
0.7
|
|||||||
|
Selling
expenses
|
3,866
|
2.8
|
2,122
|
1.9
|
|||||||
|
8,565
|
6.3
|
6,227
|
5.7
|
||||||||
|
Income
from operations
|
26,943
|
19.8
|
23,402
|
21.5
|
|||||||
|
Interest
income
|
16
|
-
|
29
|
- | |||||||
|
Other
income
|
125
|
-
|
143
|
- | |||||||
|
Finance
costs
|
(1,414
|
)
|
1.0
|
(1,260
|
)
|
1.2
|
|||||
|
Income
before income taxes
|
25,670
|
18.9
|
22,314
|
20.5
|
|||||||
|
Income
taxes
|
(6,262
|
)
|
4.6
|
(5,657
|
)
|
5.2
|
|||||
|
Net
income
|
$
|
19,408
|
14.3
|
$
|
16,557
|
15.3
|
|||||
|
Other
comprehensive income
|
|||||||||||
|
Foreign
currency translation adjustments
|
57
|
-
|
1,142
|
1.0
|
|||||||
|
Total
comprehensive income
|
$
|
19,465
|
14.3
|
$
|
17,799
|
16.3
|
|||||
The table
below sets forth a breakdown of our external segment revenue after elimination
of inter-segment sales, and each segment revenue item as a percentage of our
total revenue, as well as our inter-segment sales for the year ended December
31, 2009 and December 31, 2008. For the year ended December 31, 2009,
we had approximately $22.5 million of inter-segment revenue, which includes
approximately $21.7 million in sales from our pharmaceutical distribution
segment to our retail pharmacy segment, and approximately $762,000 in sales from
our manufacturing pharmacy segment to our distribution pharmacy
segment External segment revenue refers to segment revenue
after inter-segment elimination.
|
December
31
|
||||||||||||||||
|
2009
|
2008
|
|||||||||||||||
|
External
Segment revenue
|
’000 |
%
|
’000 |
%
|
||||||||||||
|
Pharmaceutical
distribution
|
$ | 97,137 | 71.4 | $ | 72,807 | 66.8 | ||||||||||
|
Retail
pharmacy
|
31,223 | 22.9 | 28,593 | 26.2 | ||||||||||||
|
Manufacturing
pharmacy
|
7,727 | 5.7 | 7,591 | 7 | ||||||||||||
| 136,087 | 100.0 | 108,991 | 100.0 | |||||||||||||
|
Inter-segment
revenue
|
22,491 | N/A | 21,656 | N/A | ||||||||||||
Sales
Revenue.
During
the year ended December 31, 2009, we had sales revenue of $136.1 million, as
compared to sales revenue of $109 million during the year ended December 31,
2008, an increase of $27.1 million or approximately 24.9%. This increase
was mainly attributable to an increase in sales revenue of $24.3 million from
our pharmaceutical distribution operations. Sales revenue derived from our
pharmaceutical distribution segment amounted to $97.1million, which accounted
for 71.4% of our total sales revenue. In addition, the respective increase of
$2.6 million and $0.1 million of our retail and manufacturing segments also
contributed to the increase in the sales revenue during the period
Pharmaceutical
distribution segment
Revenue
from our pharmaceutical distribution segment increased by 33.4% from $72.8
million for the year ended December 31, 2008 to $97.1 million for the year ended
December 31, 2009. The increase in sales revenue from our pharmaceutical
distribution segment was the result of an increase in the quantity and range of
products sold to our existing hospital clients, which was attributable
to:
|
·
|
China’s
expansion of the healthcare security scheme coverage to city residents,
including unemployed population, student and self-employed
person,
|
|
·
|
the
establishment of basic drugs catalogue by hospitals for full reimbursement
basis upon medical social insurance
scheme,
|
|
·
|
the
increase of the reimbursement ratio covered by medical social insurance
scheme, and
|
|
·
|
the
implementation of New Rural Corporative-Medicare scheme for rural
population as well.
|
An
additional factor contributing to the increase in our sales revenue from our
pharmaceutical distribution segment was additional sales resulting from a six
city and townships’ distribution business bid we won by middle of
2009. Further, the penetration of the group’s distribution network
into community clinics and health care institutions also contributed to the
increase in sales revenue.
Retail
pharmacy segment.
Revenue
from our retail pharmacy segment increased by 9.2% from $28.6 million
for the year ended December 31, 2008 to $31.2 million for the year ended
December 31, 2009. The increase was partly contributed by sales
derived through medi-care insurance cards as a result
of an increase in the portion of pharmacy products entitled to be
reimbursed by the PRC government, as well as an increase in the number of people
covered by insurance. Further, the prevalence of and concerns related to swine
flu in 2009 also boosted the demand for pharmacy products at our drug
stores.
Manufacturing
pharmacy segment
Revenue
from our manufacturing pharmacy segment increased by 2% from $7.6 million for
the year ended December 31, 2008 to $7.7 million for the year ended December 31,
2009.
Cost of Sales. Cost of Sales
was $100.6 million for the year ended December 31, 2009 as compared to $79.4
million for the year ended December 31, 2009. Our cost of sales
consist the cost of merchandise and raw materials and other costs. Other costs
include direct labor, depreciation and other costs. The increase was primarily
due to an increase in the costs of purchasing merchandise following the increase
in the Group’s revenue from sales.
Gross Profit. Gross profit
was $35.5 million for the year ended December 31, 2009 as compared to $29.6
million for the year ended December 31, 2008, representing an increase of $5.9
million or approximately 19.9%. Our gross profit margin was 26.1% and
27.2% for the year ended December 31, 2009 and December 31, 2008 respectively.
The gross profit margin was relatively stable in which we maintain the margin
between our cost of purchasing pharmaceutical products from our suppliers and
our prices of pharmaceutical products sold to our hospital, pharmaceutical
distributor and other customers. For hospital customers, we establish a pricing
range aligned with our suppliers through the PRC Government-mandated collective
tender process. For other pharmaceutical product distributors, we arrange three
party negotiations with distributors and suppliers. The slightly decrease in
profit margin was mainly attributed to a larger percentage of wholesale accounts
that we sold products to compared to 2008.
Pharmaceutical
distribution segment
The
respective gross profit margin for our pharmaceutical distribution segment was
approximately 20.8% and 23% for the year ended December 31, 2009 and 2008,
respectively. In order to penetrate and capture the growth of the market upon
medical insurance scheme, we attempted to satisfy the demand of hospital
customers by procuring from other eligible supplier within the medicine
catalogue but at lower margins because of pre-determined/set pricing under the
medicine catalogue. This results in slight decrease in the gross profit margin
between year 2009 and year 2008. .
Retail
pharmacy segment
The
respective gross margin for our retail pharmacy segment was approximately 32.7%
and 28.4% for the year ended December 31, 2009 and 2008, respectively. The
increase in gross profit margin was primarily attributed to our strategy of
promoting higher profit margin goods and selling more private label products
which have higher margins.
Manufacturing
pharmacy segment
The
respective gross profit margin for our manufacturing segment was approximately
65.9% and 63.1% approximately for the year ended December 31, 2009 and 2008,
respectively. We believe this increase was do to management focus on
optimizing our product portfolio to higher margin products
Selling, General and Administrative
Expenses. Selling, general and administrative expenses totaled $8.6
million for the year ended December 31, 2009, as compared to $6.2 million for
the year ended December 31, 2008, representing an increase of $2.4 million or
approximately 38.7%. The increase was partly attributable to the $1.8 million
increase in selling expenses for the year ended December 31, 2009, which was
primarily due to the increase in our marketing staff’s wages and salaries,
payment for staff welfare, commission and transportation costs in connection
with our increased sales and marketing activities. The percentage of our
distribution and selling expenses to our total revenue gradually increased from
1.9% to 2.8% and was primarily due to the increase of our efforts to penetrate
and capture market share. In addition, the increase of $1.3 million in
administrative expenses also contributed to the increase in our operating
expenses. The increase in administrative expenses for the year ended December
31, 2009 was primarily due to an increase in wages and salaries, staff benefits,
post-employment benefits, and the rental expenditures resulted from the renewal
of lease and the opening of new stores. The percentage of our administrative
expenses to our total revenue increased slightly from 3.1% in 2008 to 3.4% in
2009. The increase was in total operating expenses was partially offset by the
decrease of $0.7 million in research and development charges.
Net Income. As a result of
the above factors, we had net income of $19.4 million for the year ended
December 31, 2009 as compared to $16.7 million for the year ended December 31,
2008, representing an increase of $2.7 million or approximately
16.2%.
Earning per
share For the fiscal year ended December 31, 2009, our
earnings per share was $0.61, representing an increase of 17.3%, compared the
same period in 2008.
Liquidity
and Capital Resources
We
believe that our existing sources of liquidity, along with cash expected to be
generated from services will be sufficient to fund our operations, anticipated
capital expenditures, working capital and other financing requirements for at
least the next twelve months. We will continue to monitor our expenditures and
cash flow position and we do not believe that we shall be forced to enter into
any long or short term debt arrangements.
|
For
the Year
|
For
the Year
|
|||||||
|
Ended
|
Ended
|
|||||||
|
December
31,
|
December
31,
|
|||||||
|
2009
|
2008
|
|||||||
|
’000
|
‘000
|
|||||||
|
Net
cash provided by operating activities
|
$ |
5,900
|
$ |
11,734
|
||||
|
Net
cash used in investing activities
|
(346
|
)
|
(831
|
)
|
||||
|
Net
cash provided by/(used in) financing activities
|
6,417
|
(9,982
|
)
|
|||||
|
Foreign
currency translation
|
68
|
(176
|
)
|
|||||
|
Net
increase in cash and equivalents
|
12,039
|
745
|
||||||
|
Cash
and cash equivalents, beginning of year
|
1,265
|
520
|
||||||
|
Cash
and cash equivalents, end of year
|
13,304
|
1,265
|
||||||
Operating
Activities
Cash
provided by operating activities was $5.9 million for the year ended 2009
compared to $11.7 million for fiscal 2008, representing a decrease of $5.8
million or approximately 49.6%. Operating cash flows for 2009 reflects primarily
net cash receipts derived from business operations. Despite the increase in net
income in 2009 as compared to the same period of 2008, the reduction in net cash
from operating activities was primarily attributable to the slowdown of account
receivable repayment, which increased to 95 days in 2009 as compared to 70 days
in 2008. The increase in turnover days was attributable to an increase in the
portion of our sales to hospital under our pharmaceutical distribution
operations segment to 74% in 2009 from 52% in 2008. Hospitals, which
are owned by the PRC government, have comparatively longer payment cycles,
especially at the period of time when more investment was spent by hospital to
meet the medical reform requirement promulgated by the PRC government. Such
decrease was, to certain degree, offset by our delay in cash payments to vendors
and the increase in our use of bills and restricted cash for creditor
settlement.
Investing
Activities
Cash used
in investing activities was $0.3 million for the twelve months of 2009, compared
to $0.8 million for fiscal year 2008. The decrease in cash used was primarily
due to the proceeds obtained from the disposal of the land in fiscal year 2009
and the reduction in the expenditures for the acquisition of property, plant and
equipment.
Financing
Activities
Cash
provided by financing activities was $6.4 million for the twelve months of
fiscal year 2009, compared to $9.9 million used in financing activities for
fiscal year 2008. The substantial increase was primarily due to the proceeds of
the private placement of shares amounting to $5.3 million in December 2009. In
addition, we did not declare nor pay any dividends in 2009. In 2008,
a dividend of $6,940,000 was paid to former shareholders of Liuzhou BCT.
Further, the increase was also attributable to the reduction in loan repayments
to directors and other parties.
Off
Balance Sheet Arrangements
We have
no off balance sheet arrangements.
|
Quantitative
and Qualitative Disclosures about Market
Risk
|
Not
applicable.
|
Financial
Statements and Supplementary Data
|
The
financial statements and supplementary data required by this item are included
in Part IV, Item 15 of this Report.
|
Changes
in and Disagreements with Accountants on Accounting and Financial
Disclosure
|
On
December 30, 2009, the board of directors (the “Board of Directors”) of China
Baicaotang Medicine Limited (f/k/a Purden Lake Resource Corp., hereinafter
referred to as the “Company”, “we”, “us” or “our”) terminated Bernstein &
Pinchuk LLP, an Independent Member of BDO SEIDMAN Alliance ( “Bernstein”) as the
independent registered public accounting firm of the Company, and engaged a new
independent registered public accounting firm, PKF, Certified Public
Accountants, Hong Kong, China, a member firm of PKF International Limited
network of legally independent firms (“PKF Hong Kong”) , to serve as the
Company’s independent directors. Pursuant to Item 304(a) of Regulation S-K under
the Securities Act of 1933, as amended, and under the Securities Exchange Act of
1934, as amended, the Company reports as follows:
|
(a)
|
(i)
|
Bernstein
was terminated as our independent registered public accounting firm
effective on December 30, 2009.
|
|
(ii)
|
For
the two most recent fiscal years ended March 31, 2009 and 2008,
Bernstein’s report on the financial statements did not contain any adverse
opinions or disclaimers of opinion, and were not qualified or modified as
to uncertainty, audit scope, or accounting principles, other than for a
going concern.
|
|
|
|
(iii)
|
The
termination of Bernstein and engagement of PKF Hong Kong were approved by
the Company’s Board of Directors.
|
|
(iv)
|
The
Company and Bernstein did not have any disagreements with regard to any
matter of accounting principles or practices, financial statement
disclosure, or auditing scope or procedure for the audited financials for
the fiscal years ended March 31, 2009 and 2008, and subsequent interim
periods ended June 30 and September 30, 2009 and through the date of
dismissal, which disagreements, if not resolved to the satisfaction of
Bernstein, would have caused it to make reference to the subject matter of
the disagreements in connection with its reports.
|
|
|
(v)
|
During
our fiscal years ended March 31, 2009 and 2008, and subsequent interim
periods ended June 30 and September 30, 2009 and through the date of
dismissal, the Company did not experience any reportable
events.
|
|
(b)
|
(i)
|
On
December 30, 2009, the Company engaged PKF Hong Kong to serve as its
independent registered public accounting firm.
|
|
(ii)
|
Prior
to engaging PKF Hong Kong, the Company had not consulted PKF
Hong Kong regarding the application of accounting principles to a
specified transaction, completed or proposed, the type of audit opinion
that might be rendered on its financial statements or a reportable event,
nor did the Company consult with PKF Hong Kong regarding any disagreements
with its prior auditor on any matter of accounting principles or
practices, financial statement disclosure, or auditing scope or procedure,
which disagreements, if not resolved to the satisfaction of the prior
auditor, would have caused it to make a reference to the subject matter of
the disagreements in connection with its reports.
|
|
|
(iii)
|
The
Company did not have any disagreements with PKF Hong Kong, and
therefore did not discuss any past disagreements with PKF Hong
Kong.
|
|
|
(c)
|
The
Company requested Bernstein to furnish with a letter addressed to the SEC
stating whether it agrees with the statements made by us regarding
Bernstein.
|
|
Controls
and Procedures
|
Disclosure
Controls and Procedures.
As
required by Rule 13a-15 under the Exchange Act, in connection with filing of
this Annual Report on Form 10-K, our management, including our chief executive
officer and chief financial officer, evaluated the effectiveness of the design
and operation of our disclosure controls and procedures (as defined in Rule
13a-15(e) and 15d-15(e) under the Exchange Act) as of December 31, 2009 and has
determined that our disclosure controls and procedures were not
effective. In additions to the reasons set forth below, there is also
a strong reliance on the external consultants, including our attorneys, to
review and edit the annual and quarterly filings and to ensure compliance with
SEC disclosure requirements.
Internal
Controls Over Financial Reporting
In
connection with the filing and preparation of the 2008 10-K management assessed
the effectiveness of our internal control over financial reporting as of
December 31, 2009. In making this assessment, management used the
criteria set forth by the Committee of Sponsoring Organizations of the Treadway
Commission in Internal Control
— Integrated Framework. Based on management’s assessment using
those criteria, management concluded that our internal control over financial
reporting was not effective as of December 31, 2009.
We
identified the following material weaknesses in our internal control over the
financial reporting year 2009.
On
December 30, 2009, as a result of the completion of a share exchange
agreement between the Company and the shareholders of Ingenious, we acquired
operating subsidiaries in the PRC. Because these operating
subsidiaries did not have financial controls and procedures appropriate for
subsidiaries of a public company, the Company has began the process of
incorporating the operations acquired into its financial systems and upgrading
its financial controls and procedures As of December 31, 2009, the Company had
not established adequate financial reporting monitoring activities to mitigate
the risk of management override., specifically because we did not have an audit
committee and internal audit department to carry out the formal corporate
governance function and left the function to certain assigned person with no
segregation of duties. However, although our controls were not effective, these
significant weaknesses did not result in any material misstatements in our
financial statements
REMEDIATION
OF MATERIAL WEAKNESSES IN INTERNAL CONTROL
The
Company will continue to work with its external auditors and attorneys as it
relates to any changes to SEC
disclosure
requirements. The Company has found that this approach worked well in the past
and believes it to be the effective solution available for the foreseeable
future.
The
company has recently engaged Ernst & Young (China) Advisory Limited to work
with us to identify the key internal control problems and provide
recommendations over the control procedures. We will assign staff with complete
segregation of responsibilities to carry out the internal control
function in the future to minimize any risk of inadequate controls.
This
Annual Report does not include an attestation report of the Company's registered
public accounting firm regarding internal control over financial reporting.
Management's report was not subject to attestation by the Company's registered
public accounting firm pursuant to temporary rules of the SEC that permit the
Company to provide only management's report in this annual report.
Changes in Internal Control over
Financial Reporting. There were no changes in our internal
control over financial reporting that occurred during the fourth quarter of
fiscal year 2009 that has materially affected, or is reasonably likely to
materially affect, our internal control over financial reporting.
|
OTHER
INFORMATION
|
None.
PART
III
|
DIRECTORS
AND EXECUTIVE OFFICERS AND CORPORATE
GOVERNANCE
|
The
following table sets forth the name and age of officers and director as of March
30 Our executive officers are elected annually by our board of directors. Our
executive officers hold their offices until they resign, are removed by the
Board, or his successor is elected and qualified.
Directors
and Executive Officers
|
Name
|
Age
|
Position
|
|
Hui
Tian Tang
|
49
|
Chairman
& Chief Executive Officer
|
|
Xiao
Yan Zhang
|
37
|
Director
& Chief Financial Officer
|
Mr.
Hui Tian Tang
Mr. Tang
was appointed as our CEO on December 30, 2009 and as our Chairman on January 14,
2010. Mr. Tang is a registered pharmacist and has been
the president of Liuzhou BCT since 2001. In 1993, he was hired as the
general manager by Liuzhou BCT (f/k/a Guangxi Liuzhou Wholesaler) and was
promoted to president of the company in 2001. Mr. Tang has over 25 years
experience in the traditional Chinese medicine industry. As the president of
Liuzhou BCT, Mr. Tang has been responsible for the formulation of strategies,
decision-making on investment projects and development directions on the
operations and overall business management, and led us successfully through the
privatization process in 2001. Prior to his employment with Liuzhou BCT in 1993,
Mr. Tang was employed by Guangxi Jinchengjiang Medicine Wholesaler Group from
1983 to 1993 where he was deputy general manager. In July 1983 Mr.
Tang received a Bachelor of Chinese Pharmacy from Guangxi Chinese Medicine
University.
Ms.
Xiao Yan Zhang
Ms. Zhang
joined Liuzhou BCT in May 2008 as our Corporate Strategy VP and was appointed as
our CFO on December 30, 2009 and as a director on January 14,
2010. Prior to joining Liuzhou BCT, from 2006 to 2008 she was a
corporate finance advisor to First Asia Finance Group, in Hong
Kong. Ms. Zhang is an Associate Member of CPA
Australia. She received a Masters degree in accounting from Curtin
University of Technology, Australia in 2007, an MBA in International Business
from CMSD Switzerland in 2001 and a BA (Honors) in Marketing from Portsmouth
University, UK in 2004.
In
general, we seek directors:
|
·
|
with
established strong professional reputations and experience in areas
relevant to the strategy and operations of our
businesses;
|
|
·
|
who
possess the qualities of integrity and candor, who have strong analytical
skills and who are willing to engage management and each other in a
constructive and collaborative
fashion;
|
|
·
|
who
have an understanding of business practices in China, and the ability to
communicate in Mandarin Chinese;
and
|
|
·
|
who
have the ability and commitment to devote significant time and energy to
service on the Board and its
committees.
|
When
considering whether directors and nominees have the experience, qualifications,
attributes or skills, taken as a whole, to enable our Board of Directors to
satisfy its oversight responsibilities effectively in light of our business and
structure, the Board of Directors focused primarily on each person’s background
and experience as reflected in the information discussed in each of the
directors’ individual biographies set forth above. We believe that our
directors provide an
appropriate
mix of experience and skills relevant to the size and nature of our business. In
particular, the members of our Board of Directors considered the following
important characteristics: (i) Mr. Tang has worked for Liuzhou BCT since
1993 and has significant expertise in the business operations of the Company and
the pharmaceutical business, and (ii) Ms. Zhang worked for Liuzhou BCT for
more than 18 months prior to joining the Board and has significant financial
expertise.
Family
Relationship
There are
no family relationships between any of our directors or executive officers and
any other directors or executive officers.
Corporate
Governance
Director
Independence
We do not
currently have any independent directors serving on our board of
directors.
Board
Committees
We do no
have currently have any Board committees. Our board of directors currently
performs the functions that would be delegated to the audit
committee.
Code
of Ethics
We have
adopted a code of ethics that applies to our officers, employees and directors,
including our Chief Executive Officer and senior executives. A copy
of the Code of Ethics is included as Exhibit 14.1 to our Current Report on Form
8-K, filed with the SEC on December 31, 2009. A printed copy of the Code of
Ethics may also be obtained free of charge by writing to us at our headquarters
located at No. 102, Chengzhan Road, Liuzhou City, Guangxi Province, PRC 545007,
Attention: Company Secretary.
Involvement
in Certain Legal Proceedings
There
have been no events under any bankruptcy act, no criminal proceedings and no
judgments, injunctions, orders or decrees material to the evaluation of the
ability and integrity of any director, executive officer, promoter or control
person of our Company during the past ten years.
Director
Compensation
None of
the directors have received compensation for their respective services rendered
to the Company for the year ended December 31, 2009
Conflicts
of Interest
Certain
potential conflicts of interest are inherent in the relationships between our
officers and directors, and us.
From time
to time, one or more of our affiliates may form or hold an ownership interest in
and/or manage other businesses both related and unrelated to the type of
business that we own and operate. These persons expect to continue to form, hold
an ownership interest in and/or manage additional other businesses which may
compete with ours with respect to operations, including financing and marketing,
management time and services and potential customers. These activities may give
rise to conflicts between or among the interests of us and other businesses with
which our affiliates are associated. Our affiliates are in no way prohibited
from undertaking such activities, and neither we nor our shareholders will have
any right to require participation in such other activities.
Further,
because we intend to transact business with some of our officers, directors and
affiliates, as well as with firms in which some of our officers, directors or
affiliates have a material interest, potential conflicts may arise between the
respective interests of us and these related persons or entities. We believe
that such transactions will be effected on terms at least as favorable to us as
those available from unrelated third parties.
With
respect to transactions involving real or apparent conflicts of interest, we
have adopted policies and procedures which require that: (i) the fact of the
relationship or interest giving rise to the potential conflict be disclosed or
known to the directors who authorize or approve the transaction prior to such
authorization or approval, (ii) the transaction be approved by a majority of our
disinterested outside directors, and (iii) the transaction be fair and
reasonable to us at the time it is authorized or approved by our
directors.
|
EXECUTIVE
COMPENSATION
|
Compensation
Discussion and Analysis
We strive
to provide our named executive officers with a competitive base salary that is
in line with their roles and responsibilities when compared to peer companies of
comparable size in the same or similar locality.
It is not
uncommon for companies with operations primarily in China operations to have
base salaries and bonuses as the sole and only form of compensation. The base
salary level is established and reviewed based on the level of responsibilities,
the experience and tenure of the individual and the current and potential
contributions of the individual. The base salary is compared to similar
positions within comparable peer companies and with consideration of the
executive’s relative experience in his or her position. Based on an evaluation
of available information with respect to the base salaries of executives of our
competitors, the base salary and bonus paid to our named executive officers is
in line with our competitors. Base salaries are reviewed periodically and at the
time of promotion or other changes in responsibilities.
We plan
to implement a more comprehensive compensation program appropriate for
executives of a public company, which takes into account other elements of
compensation, including without limitation, short and long term compensation,
cash and non-cash, and other equity-based compensation such as stock options. We
expect that such compensation programs shall be comparative to our peers in the
industry and aimed to retain and attract talented individuals.
Summary
Compensation Table— Fiscal Years Ended December 31, 2009 and 2008
The
following table sets forth information concerning all cash and non-cash
compensation awarded to, earned by or paid to the named person for services
rendered in all capacities during the noted periods.
|
Name
and Principal Position
(1)
|
Year
Ended December 31
|
Salary
($)
|
Bonus
($)
|
Stock
Awards
($)
|
Option
Awards
($)
|
Non-Equity
Incentive Plan Compensation Earnings
($)
|
Non-Qualified
Deferred Compensation Earnings
($)
|
All
Other Compensation
($)
|
Total
($)
|
|
Lisa
Lopomo
|
2009
|
0
|
0
|
0
|
0
|
0
|
0
|
0
|
0
|
|
former
CEO and
|
2008
|
0
|
0
|
0
|
0
|
0
|
0
|
0
|
0
|
|
Director
(1)
|
|||||||||
|
Hui
Tian Tang
|
2009
|
27,027
|
0
|
0
|
0
|
0
|
0
|
0
|
27,027
|
|
Chairman
& CEO(2)
|
2008
|
27,027
|
0
|
0
|
0
|
0
|
0
|
0
|
27,027
|
|
Xiaoyan
Zhang
|
2009
|
0
|
0
|
0
|
0
|
0
|
0
|
0
|
0 |
|
CFO
and Director(2)
|
|
(1)
|
On
December 30, 2009, Ms. Lisa Lopomo tendered her letter of resignation to
resign as CEO, effective December 30, 2009 and to resign from
our board of directors y, effective on January 14,
2010.
|
|
(2)
|
On
December 30, 2009, Hui Tian Tang and Xiaoyan Zhang were elected as to the
board of directors of the Company, effective January 14, 2010 which is 10
days following the filing of an information statement required by Rule
14f-1 promulgated under the Exchange Act. In addition, effective on
December 30, 2009, Hui Tian Tang and Xiaoyan Zhang were also elected to
serve as the CEO and CFO of the Company
respectively.
|
|
(3)
|
Represents
amounts paid to Mr. Tang by Liuzhou
BCT.
|
Employment
Agreements
Liuzhou
BCT entered into an employment agreement with Mr. Hui Tian Tang pursuant to
which Mr. Tang was hired as the President of Liuzhou BCT and received a salary
of $27,027 per year in 208 and 2009 We currently have an oral
employment agreement beginning on January 1, 2010 with Mr. Tang to employ him
has our CEO, pursuant to which we have agreed to pay him 90,000 Hong Kong
Dollars (HK$) ($11,688) per month and a discretionary bonus based upon our 2010
financial performance We currently have an oral employment agreement
beginning on January 1, 2010 with Xiaoyan Zhang to employ her as our CFO,
pursuant to which we have agreed to pay her HKD70,000 ($9,091) per month and a
discretionary bonus based upon our 2010 financial performance. We are
currently in the process of formalizing each of these agreements.
Outstanding
Equity Awards at Fiscal Year End
None of
our executive officers received any equity awards, including, options,
restricted stock or other equity incentives during the fiscal year ended
December 31, 2009.
Compensation
of Directors
During
the 2008 and 209 fiscal years, no member of our board of directors received any
compensation solely for service as a director. Our directors will not receive a
fee for attending each board of directors meeting or meeting of a committee of
the board of directors. All directors will be reimbursed for their reasonable
out-of-pocket expenses incurred in connection with attending board of director
and committee meetings.
Compensation
Committee Interlocks and Insider Participation
During
the last fiscal year we did not have a standing Compensation Committee. Our
board of directors was responsible for the functions that would otherwise be
handled by the compensation committee.
Indemnification
of Directors and Executive Officers and Limitation of Liability
Delaware
General Corporation Law does not limit the extent to which a company’s articles
of association may provide for indemnification of officers and directors, except
to the extent any such provision may beheld by the courts of the State of
Delaware to be contrary to public policy, such as to provide indemnification
against civil fraud or the consequences of committing a crime. Our
articles of association provide for indemnification of our officers and
directors for any liability incurred in their capacities as such, except through
their own willful negligence or default.
Insofar
as indemnification for liabilities arising under the Securities Act may be
permitted to directors, officers or persons controlling us pursuant to the
foregoing provisions, we have been informed that in the opinion of the
Securities and Exchange Commission such indemnification is against public policy
as expressed in the Securities Act and is theretofore
unenforceable.
|
Security
Ownership of Certain Beneficial Owners and Management and Related
Stockholder Matters
|
The
following table sets forth certain information regarding beneficial ownership of
our common stock effective March 29, 2010 by (i) each person (or group of
affiliated persons) who is known by us to own more than five percent of the
outstanding shares of our common stock, (ii) each director, and named executive
officer, and (iii) all of our directors and executive officers as a group. As of
March 1, 2010, we had 38,154,340 shares of common stock issued and
outstanding.
Beneficial
ownership is determined in accordance with SEC rules and generally includes
voting or investment power with respect to securities. Unless otherwise noted,
the principal address of each of the stockholders, directors and officers listed
below is c/o Guangxi Liuzhou Baicaotang Medicine Limited, No. 102 Chengzhan
Road, Liuzhou City, Guangxi Province, PRC.
All share
ownership figures include shares of our Common Stock and securities convertible
or exchangeable into shares of our Common Stock within sixty (60) days of March
29, 2010, which are deemed outstanding and beneficially owned by such person for
purposes of computing his or her percentage ownership, but not for purposes of
computing the percentage ownership of any other person.
|
Names
and Addresses of Beneficial Owners
|
Amount
and Nature of Beneficial Ownership (1)
|
Percent
of Class (2)
|
|
Xiao
Yan Zhang (3)
|
22,480,000
|
58.9%
|
|
All
directors and executive officers as a group (2 persons)
|
22,480,000
|
58.9%
|
|
(1)
|
Beneficial
ownership is determined in accordance with the rules of the SEC and
generally includes voting or investment power with respect to securities.
Shares of common stock subject to securities anticipated to be exercisable
or convertible at or within 60 days of the date hereof, are deemed
outstanding for computing the percentage of the person holding such option
or warrant but are not deemed outstanding for computing the percentage of
any other person. The indication herein that shares are anticipated to be
beneficially owned is not an admission on the part of the listed
stockholder that he, she or it is or will be a direct or indirect
beneficial owner of those shares.
|
|
(2)
|
Based
upon 38,154,340 shares of common stock issued and
outstanding.
|
|
(3)
|
Xiao
Yan Zhang is a citizen of Hong Kong and our Chief Financial Officer,
Secretary and director. Pursuant to an Earn-in Agreement dated December
30, 2009 by and among Ms. Zhang and certain former shareholders of Liuzhou
BCT (the “Former Shareholders”), such Former Shareholders have a call
right to purchase up to 22,480,000 shares of our common
stock from Ms. Zhang for a nominal amount per share provided
that the Company meets certain performance targets for fiscal 2010 and
2011. For the 2010 and 2011 fiscal years the performance
targets for the Company are $26 million and $28 million after tax audited
net income, respectively. If the 2010 performance target is
met, the Former Shareholders have the right to acquire 50% of shares held
by Ms. Zhang over which they have a call right. If the 2011
performance target is met the Former Shareholders have the right to
acquire the other 50% of the shares held by Ms. Zhang over which they have
a call right. The number of shares which can be acquired by the
Former Shareholders under the Earn-In Agreement is in proportion to their
former relative ownership interest in Liuzhou BCT. Our Chairman
and CEO, Hui Tian Tang, is one of the Former Shareholders and has the
right to acquire up to 2,241,193 shares of our common stock under the
Earn-In Agreement if both performance targets are
met.
|
|
Certain
Relationships and Related Transactions, and Director
Independence
|
China
Baicaotang Medicine Limited
Effective
December 23, 2009, the Company entered into lock-up agreements with the original
shareholders of Ingenious, consisting of Xiao Yan Zhang, Lei Ying, Chunqi
Cao, Bixun Su, Hui Tian Tang, Yik Kwok Wah, and Yik Li Yee, pursuant to which
these shareholders will be refrained from selling any of our securities from the
date of the Subscription Agreement for twelve (12) months after the earlier of:
(i) the effective date of the Registration Statement of which this prospectus is
a part; or (ii) the date that the shares of common stock may be sold under Rule
144, without limitation.
Liuzhou
BCT
On March
31, 2009, Liuzhou BCT entered into a Shares Pledge Agreement with Property
Management, pursuant to which Property Management pledged 100% of its equity
interest in BCT Retail to Liuzhou BCT to guarantee a loan of approximately
$196,618 (RMB 1,377,000) from Liuzhou BCT.
On
January 15, 2009, Liuzhou BCT entered into a loan agreement with Agricultural
Bank of China Liuzhou Branch in the amount of approximately $660,150 (RMB
4,500,000), pursuant to which both the Baicaotang Property Development Limited
and Wuxuan Baicaotang Medicine Limited pledge part of their land and property to
the bank as security interest for the loan.
On
February 8, 2007, Mr. Hui Tian Tang entered into a loan agreement with
Industrial and Commercial Bank of China Guangxi Branch in the amount of
approximately $234,720 (RMB1,600,000), pursuant to which Guangxi Liuzhou BCT
Medicine Limited, Guigang Branch pledge part of its assets to the bank as
security interest for the loan. Mr. Hui Tang then lent the full amount of the
above loan to the Liuzhou BCT for working capital. On December 31, 2008, a
mutual agreement was signed between Mr. Tang and Liuzhou BCT, pursuant to which
Liuzhou BCT assume the obligation to repay the principal amount and accrued
interest from January 1, 2009 onwards.
On
February 12, 2007, Jiang You Ru, the director of Liuzhou BCT entered into a loan
agreement with Industrial and Commercial Bank of China Guangxi Branch in the
amount of approximately $264,060 (RMB1,800,000), pursuant to which Guangxi
Liuzhou BCT Medicine Limited, Guigang Branch pledge part of its assets to the
bank as security interest for the loan. Mr. Jiang then lent the full amount of
the above loan to the Liuzhou BCT for working capital. On December 31, 2008, a
mutual agreement was signed between Mr. Tang and Liuzhou BCT, pursuant to which
Liuzhou BCT assume the obligation to repay the principal amount and accrued
interest from January 1, 2009 onwards.
In
addition, we also entered into the following transactions with related parties
as follows:
|
Year
ended December 31,
|
||||
|
2009
|
||||
|
Sales
of goods
|
||||
|
Liucheng
Medicine Limited
|
$ | 311,759 | ||
|
Guangxi
Tianhu Medicine Limited
|
$ | 271,890 | ||
|
Guangxi
Liuzhou Baicaotang Medicine Limited, Guigang Branch
|
$ | 1,273,381 | ||
|
Wuxuan
Baicaotang Medicine Limited
|
$ | 208,475 | ||
Hefeng Pharmaceutical
From 2006
to 2009, Mr. Jing Hua Li, the General Manager of Liuzhou Baicaotang, entered
into a series of loan agreements with Hefeng Pharmaceutical, pursuant to which
Hefeng Pharmaceutical borrowed an aggregate of approximately$586,563 at monthly
interests rates ranging from 5.8% to 6.8%. All of the loan agreements have
similar terms and provisions.
Liuzhou
Retail
On June
18, 2009, Liuzhou Retail entered into a loan agreement with Rurol Credence
Cooperation of Guangxi in the amount of approximately $1,613,700 (RMB
11,000,000), pursuant to which Baicaotang Property Development Limited pledge
part of its premise to the bank as security interest for the loan
|
Principal
Accounting Fees and Services
|
Our
independent accountants for the audit of our annual financial statements for our
fiscal years ended December 31, 2009 and 2008, was PKF, Certified Public
Accountants, Hong Kong, China, a member firm of PKF International Limited
network of legally independent firms (“PKF Hong Kong”) . The
following table shows the fees paid or accrued by us to PKF Hong
Kong.
|
2009
|
2008
|
|||||||
|
Audit
Fees
|
$
|
137,000
|
$
|
70,000
|
||||
|
Audit-Related
Fees
|
$
|
0
|
$
|
0
|
||||
|
Tax
Fees
|
$
|
0
|
$
|
0
|
||||
|
All
other fees
|
$
|
0
|
$
|
0
|
|
|||
Audit
Fees were for professional services rendered for the audit of our company’s
annual financial statements, the review of quarterly financial statements and
the preparation of statutory and regulatory
filings.
Pre-Approval
of Services.
We do not have an audit committee and as a
result, our entire board of directors performed the duties of an audit
committee. Our board of directors evaluated and approved in advance the scope
and cost of the engagement of an auditor before the auditor rendered audit
services.
PART IV
|
Exhibits,
Financial Statement Schedules
|
(a) The
following documents are filed as part of this Report:
1. Financial
Statements.
Incorporated
by reference from the financial statements and notes thereto that are set forth
in Item 8 of this Annual Report on Form 10-K.
2. Financial
Statement Schedules.
No
schedules have been filed because they are not applicable or not required, or
the information is included in the Consolidated Financial Statements or Notes
thereto.
(b) Exhibits.
The
exhibits listed on the accompanying index to exhibits immediately following the
financial statements are filed as part of, or hereby incorporated by reference
into, this Report.
China
Baicaotang Medicine Limited
Consolidated
Financial Statements
Index to
Consolidated Financial Statements
|
Pages
|
||
|
Report
of Independent Registered Public Accounting Firm
|
F-1
|
|
|
Consolidated
Statements of Income and Comprehensive Income
|
F-2
|
|
|
Consolidated
Balance Sheets
|
F3
- F4
|
|
|
Consolidated
Statements of Cash Flows
|
F5
- F6
|
|
|
Consolidated
Statements of Stockholders’ Equity
|
F-7
|
|
|
Notes
to Consolidated Financial Statements
|
F8
- F36
|
Report
of Independent Registered Public Accounting Firm
To the
Directors and Stockholders of
China
Baicaotang Medicine Limited
We have
audited the accompanying consolidated balance sheets of China Baicaotang
Medicine Limited (the “Company”) and its subsidiaries as of December 31, 2009
and 2008, and the related consolidated statements of Income and comprehensive
Income, stockholders’ equity and cash flows for each of the two years in the
period ended December 31, 2009. These financial statements are the
responsibility of the Company’s management. Our responsibility is to
express an opinion on these financial statements based on our
audits.
We
conducted our audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require
that we plan and perform the audit to obtain reasonable assurance about whether
the financial statements are free of material misstatement. An audit
includes examining, on a test basis, evidence supporting the amounts and
disclosures in the financial statements. An audit also includes
assessing the accounting principles used and significant estimates made by
management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis
for our opinion.
In our
opinion, the financial statements referred to above present fairly, in all
material respects, the consolidated financial position of the Company and its
subsidiaries as of December 31, 2009 and 2008, and the consolidated results
of their operations and their cash flows for each of the two years in the period
ended December 31, 2009 in conformity with accounting principles generally
accepted in the United States of America.
PKF
Certified
Public Accountants
Hong
Kong, China
March 31,
2010
China
Baicaotang Medicine Limited
Consolidated
Statements of Income and Comprehensive Income
(Stated
in US Dollars)
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Revenue
|
||||||||
|
Sales
|
$ | 136,086,708 | $ | 108,991,329 | ||||
|
Cost of sales
|
100,578,769 | 79,361,987 | ||||||
|
Gross profit
|
35,507,939 | 29,629,342 | ||||||
|
Operating
expenses
|
||||||||
|
Administrative
expenses
|
4,598,800 | 3,341,605 | ||||||
|
Research and development
expenses
|
99,688 | 763,995 | ||||||
|
Selling
expenses
|
3,865,980 | 2,122,153 | ||||||
| 8,564,468 | 6,227,753 | |||||||
|
Income
from operations
|
26,943,471 | 23,401,589 | ||||||
|
Interest income
|
16,100 | 29,315 | ||||||
|
Government grants - Note
5
|
29,320 | - | ||||||
|
Other income - Note
6
|
95,345 | 143,426 | ||||||
|
Finance costs - Note
7
|
(1,413,873 | ) | (1,260,290 | ) | ||||
|
Income
before income taxes
|
25,670,363 | 22,314,040 | ||||||
|
Income
taxes - Note 8
|
(6,261,798 | ) | (5,656,878 | ) | ||||
|
Net
income attributable to China Baicaotang Medicine Limited
|
||||||||
|
common
stockholders
|
$ | 19,408,565 | $ | 16,657,162 | ||||
|
Other
comprehensive income
|
||||||||
|
Foreign currency translation
adjustments
|
57,322 | 1,142,614 | ||||||
|
Total
comprehensive income
|
$ | 19,465,887 | $ | 17,799,776 | ||||
|
Earnings
per share: basic and diluted - Note 9
|
$ | 0.61 | $ | 0.52 | ||||
|
Weighted
average number of shares
|
||||||||
|
outstanding:
basic and diluted
|
32,013,943 | 31,963,669 | ||||||
See the
accompanying notes to consolidated financial statements
China
Baicaotang Medicine Limited
Consolidated
Balance Sheets
(Stated
in US Dollars)
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
ASSETS
|
||||||||
|
Current
assets
|
||||||||
|
Cash and cash
equivalents
|
$ | 13,304,158 | $ | 1,265,184 | ||||
|
Restricted cash - Note
10
|
1,155,779 | 1,228,011 | ||||||
|
Trade receivables, net - Note
11
|
35,410,039 | 20,976,203 | ||||||
|
Amounts due from related
companies - Note 20
|
4,275,586 | 4,658,061 | ||||||
|
Other receivables, prepayments
and deposits - Note 12
|
2,526,398 | 1,252,527 | ||||||
|
Inventories - Note
13
|
8,745,525 | 6,425,765 | ||||||
|
Deferred taxes - Note
8
|
60,164 | 60,164 | ||||||
|
Total current
assets
|
65,477,649 | 35,865,915 | ||||||
|
Goodwill - Note
14
|
107,968 | 107,968 | ||||||
|
Other intangible assets - Note
14
|
660,034 | 786,049 | ||||||
|
Property, plant and equipment,
net - Note 15
|
12,171,689 | 12,413,274 | ||||||
|
Land use rights - Note
16
|
13,979,753 | 15,667,168 | ||||||
|
Deposits for acquisition of
property, plant and equipment
|
- | 299,325 | ||||||
|
Deferred taxes - Note
8
|
663,699 | 674,649 | ||||||
|
TOTAL
ASSETS
|
$ | 93,060,792 | $ | 65,814,348 | ||||
See the accompanying notes to consolidated financial statements
China
Baicaotang Medicine Limited
Consolidated
Balance Sheets (Cont’d)
(Stated
in US Dollars)
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
LIABILITIES
AND STOCKHOLDERS’ EQUITY
|
||||||||
|
LIABILITIES
|
||||||||
|
Current
liabilities
|
||||||||
|
Trade payables
|
$ | 19,159,212 | $ | 15,007,291 | ||||
|
Bills payable - Note
10
|
2,239,604 | 1,448,293 | ||||||
|
Other payables and accrued
expenses - Note 17
|
3,194,612 | 3,230,577 | ||||||
|
Amounts due to directors - Note
19
|
1,008,111 | 1,270,929 | ||||||
|
Amounts due to related
companies - Note 20
|
128,579 | 126,440 | ||||||
|
Income tax
payable
|
562,603 | 954,666 | ||||||
|
Secured bank loans - Note
21
|
7,136,069 | 10,291,005 | ||||||
|
Other loans - Note
22
|
2,361,258 | 2,432,139 | ||||||
|
Retirement benefit costs - Note
5
|
59,158 | 89,016 | ||||||
|
Total current
liabilities
|
35,849,206 | 34,850,356 | ||||||
|
Secured long-term bank loans -
Note 21
|
3,631,957 | 2,200,500 | ||||||
|
Retirement benefit costs - Note
5
|
201,320 | 180,426 | ||||||
|
TOTAL
LIABILITIES
|
39,682,483 | 37,231,282 | ||||||
|
COMMITMENTS AND CONTINGENCIES
- Note 23
|
||||||||
|
STOCKHOLDERS’
EQUITY
|
||||||||
|
Common stock: par value $0.001
per share
|
||||||||
|
Authorized
100,000,000 shares in 2009 and
2008;
37,089,370 shares issued and
|
||||||||
|
outstanding
as of December 31, 2009 and 32,000,000 shares issued and outstanding as of
December 31, 2008 - Note 24
|
37,089 | 32,000 | ||||||
|
Additional paid-in
capital - Note 24
|
14,920,899 | 9,596,632 | ||||||
|
Statutory and
surplus reserves - Note 25
|
2,605,901 | 1,431,174 | ||||||
|
Accumulated other
comprehensive income
|
2,110,270 | 2,052,948 | ||||||
|
Retained
earnings
|
33,704,150 | 15,470,312 | ||||||
|
TOTAL
STOCKHOLDERS’ EQUITY
|
53,378,309 | 28,583,066 | ||||||
|
|
||||||||
|
TOTAL
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
$ | 93,060,792 | $ | 65,814,348 | ||||
See the
accompanying notes to consolidated financial statements
China
Baicaotang Medicine Limited
Consolidated
Statements of Cash Flows
(Stated
in US Dollars)
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Cash
flows from operating activities
|
||||||||
|
Net income attributable to
China Baicaotang Medicine Limited
|
||||||||
|
common
stockholders
|
$ | 19,408,565 | $ | 16,657,162 | ||||
|
Adjustments to reconcile net
income to net
|
||||||||
|
cash provided by operating
activities :-
|
||||||||
|
Depreciation
|
598,276 | 632,900 | ||||||
|
Amortization of other intangible
assets
|
118,565 | 200,227 | ||||||
|
Amortization of land use
rights
|
313,533 | 334,206 | ||||||
|
Deferred taxes
|
10,943 | (52,835 | ) | |||||
|
Gain on disposal of land use
rights
|
(17,814 | ) | - | |||||
|
Recovery of doubtful
debts
|
- | (84,091 | ) | |||||
|
Written off of other
receivables
|
174,524 | - | ||||||
|
Changes in operating assets and
liabilities :-
|
||||||||
|
Trade receivables
|
(14,423,654 | ) | (9,394,900 | ) | ||||
|
Other receivables, prepayments
and deposits
|
(1,447,184 | ) | 199,870 | |||||
|
Amounts due to (from) related
companies
|
(1,020,253 | ) | 324,261 | |||||
|
Inventories
|
(2,318,179 | ) | 3,003,812 | |||||
|
Trade payables
|
4,149,091 | 192,095 | ||||||
|
Bills payable
|
790,772 | (180,766 | ) | |||||
|
Other payables and accrued
expenses
|
(35,940 | ) | (442,348 | ) | ||||
|
Retirement benefit
costs
|
(8,959 | ) | 680 | |||||
|
Income tax
payable
|
(391,796 | ) | 344,490 | |||||
|
Net
cash flows provided by operating activities
|
5,900,490 | 11,734,763 | ||||||
|
Cash
flows from investing activities
|
||||||||
|
Payments to acquire and for
deposits for
|
||||||||
|
acquisition of
property, plant and equipment
|
||||||||
|
and land use
rights
|
(71,328 | ) | (721,910 | ) | ||||
|
Net cash received from the
RTO
|
22,694 | - | ||||||
|
Cash received from disposal of
land use rights
|
1,391,588 | - | ||||||
|
Cash received from acquisition
of Hefeng - Note 4
|
- | 631,818 | ||||||
|
Advanced to related
companies
|
(1,689,557 | ) | (741,043 | ) | ||||
|
Net
cash flows used in investing activities
|
$ | (346,603 | ) | $ | (831,135 | ) | ||
See the
accompanying notes to consolidated financial statements
China Baicaotang Medicine Limited
Consolidated
Statements of Cash Flows (Cont’d)
(Stated
in US Dollars)
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Cash
flows from financing activities
|
||||||||
|
Advance from related
companies
|
$ | 3,094,189 | $ | 72,368 | ||||
|
Restricted cash
|
72,183 | (250,089 | ) | |||||
|
Repayments to
directors
|
(262,639 | ) | (1,016,329 | ) | ||||
|
Dividend paid to former
stockholders of Baicaotang Medicine
|
- | (6,940,000 | ) | |||||
|
Proceeds from bank
loans
|
9,587,885 | 10,688,723 | ||||||
|
Repayment of bank
loans
|
(11,310,190 | ) | (11,229,285 | ) | ||||
|
Cash received from private
placement
|
5,306,662 | - | ||||||
|
Proceeds from other
loans
|
590,945 | 1,317,891 | ||||||
|
Repayment of other
loans
|
(661,777 | ) | (2,642,631 | ) | ||||
|
Proceeds from issue of common
stock by Baicaotang Medicine
|
- | 17,569 | ||||||
|
Net
cash flows provided by (used in) financing activities
|
6,417,258 | (9,981,783 | ) | |||||
|
Effect
of foreign currency translation on cash
|
||||||||
|
and
cash equivalents
|
67,829 | (176,397 | ) | |||||
|
Net
increase in cash and cash equivalents
|
12,038,974 | 745,448 | ||||||
|
Cash
and cash equivalents - beginning of year
|
1,265,184 | 519,736 | ||||||
|
Cash
and cash equivalents - end of year
|
$ | 13,304,158 | $ | 1,265,184 | ||||
|
Supplemental
disclosures for cash flow information :-
|
||||||||
|
Cash paid for
|
||||||||
|
-
Interest
|
$ | 1,291,082 | $ | 1,227,364 | ||||
|
- Income
taxes
|
$ | 6,642,573 | $ | 5,365,222 | ||||
|
Non-cash investing
activity
|
||||||||
|
Acquisition
of Hefeng in form of non-cash
|
||||||||
|
contribution
- Note 4
|
$ | - | $ | 4,982,223 | ||||
See the
accompanying notes to consolidated financial statements
China Baicaotang Medicine Limited
Consolidated
Statements of Stockholders’ Equity
(Stated
in US Dollars)
|
Statutory
|
Accumulated
|
|||||||||||||||||||||||||||
|
Additional
|
and
surplus
|
other
|
||||||||||||||||||||||||||
|
Common
stock
|
paid-in
|
reserves
|
comprehensive
|
Retained
|
||||||||||||||||||||||||
|
No.
of shares
|
Amount
|
capital
|
(Note
25)
|
income
|
earnings
|
Total
|
||||||||||||||||||||||
|
Balance,
January 1, 2008
|
26,419,200 | $ | 26,419 | $ | 4,602,421 | $ | 329,562 | $ | 910,334 | $ | 6,854,762 | $ | 12,723,498 | |||||||||||||||
|
Net
income
|
- | - | - | - | - | 16,657,162 | 16,657,162 | |||||||||||||||||||||
|
Foreign
currency translation adjustments
|
- | - | - | - | 1,142,614 | - | 1,142,614 | |||||||||||||||||||||
|
Acquisition
of Hefeng – Note 3 and 4
|
4,300,800 | 4,301 | 4,977,922 | - | - | - | 4,982,223 | |||||||||||||||||||||
|
Increase
in paid up capital of Baicaotang
|
||||||||||||||||||||||||||||
|
medicine
in form of cash consideration
|
||||||||||||||||||||||||||||
|
before
the Reorganization as stated in
|
||||||||||||||||||||||||||||
|
Note
2
|
1,280,000 | 1,280 | 16,289 | - | - | - | 17,569 | |||||||||||||||||||||
|
Appropriation
to reserves
|
- | - | - | 1,101,612 | - | (1,101,612 | ) | - | ||||||||||||||||||||
|
Dividend
– Note 18
|
- | - | - | - | - | (6,940,000 | ) | (6,940,000 | ) | |||||||||||||||||||
|
Balance,
December 31, 2008
|
32,000,000 | 32,000 | 9,596,632 | 1,431,174 | 2,052,948 | 15,470,312 | 28,583,066 | |||||||||||||||||||||
|
Net
income
|
- | - | - | - | - | 19,408,565 | 19,408,565 | |||||||||||||||||||||
|
Foreign
currency translation adjustments
|
- | - | - | - | 57,322 | - | 57,322 | |||||||||||||||||||||
|
Appropriation
to reserves
|
- | - | - | 1,174,727 | - | (1,174,727 | ) | - | ||||||||||||||||||||
|
Recapitalization
|
2,600,000 | 2,600 | 20,094 | - | - | - | 22,694 | |||||||||||||||||||||
|
Private
placement – Note 24(b)
|
2,489,370 | 2,489 | 5,304,173 | - | - | - | 5,306,662 | |||||||||||||||||||||
|
Balance,
December 31, 2009
|
37,089,370 | $ | 37,089 | $ | 14,920,899 | $ | 2,605,901 | $ | 2,110,270 | $ | 33,704,150 | $ | 53,378,309 | |||||||||||||||
See the
accompanying notes to consolidated financial statements
|
1.
|
Corporate
information
|
|
(i)
|
China
Baicaotang Medicine Limited (the “Company”), formerly known as Purden Lake
Resource Corp. which changed its name on December 24, 2009, was
incorporated in the State of Delaware on November 30, 2006 as a limited
liability company with authorized capital stock consists of 100,000,000
shares of Common Stock, par value $0.001. Prior to the completion of
reverse takeover transaction (“RTO”) on December 30, 2009 as mentioned in
Note 2 (iv), the Company was a development stage company for acquisition,
exploration and development of natural resource properties. Following the
completion of RTO on December 30, 2009, the Company commenced to be
engaged in distribution, retail and production of drugs in the People’s
Republic of China (the “PRC”).
|
|
(ii)
|
Ingenious
Paragon Global Limited (“Ingenious”) was incorporated in the British
Virgin Islands (the “BVI”) on May 29, 2008 as a limited liability company
with authorized, issued and paid up capital of $50,000, divided into
50,000 common shares of $1 par value each. Prior to the
completion of RTO on December 30, 2009, the 50,000 common shares were held
by Xiao Yan Zhang, the Chief Financial Officer of the Company and its
directors were Hui Tian Tang, Jing Hua Li, JYou Ru Jiang, Chun Lin Liu,
Wen De Wei and Bang Fu wang. The principal activity of
Ingenious is investment holding.
|
|
(iii)
|
Forever
Well Asia Pacific Ltd. (“Forever Well”) was incorporated in Hong Kong on
January 10, 2008 as a limited liability company with authorized, issued
and paid up capital of HK$10,000, divided into 10,000 common shares of
HK$1 par value each. The principal activity of Forever Well is
investment holding. Before the acquisition by Ingenious as
stated in Note 2(ii), the 10,000 common shares were held by Ping Ki Yueand
its directors were Zhi Wen Zheng and Han Xin
Hong.
|
|
(iv)
|
Baicaotang
Medicine Ltd. (“Baicaotang Medicine”) was established on April 3, 1986 in
the PRC as a State-Owned Enterprise. On June 20, 2001,
Baicaotang Medicine was transformed into a joint stock enterprise through
management buyout by certain of its management and employees. On December
29, 2007, Baicaotang Medicine was transformed into a limited
company. Baicaotang Medicine is engaged in the distribution of
drugs in the PRC. Before the acquisition by Forever Well as
stated in Note 2(iii), the registered and paid up capital was
RMB10,000,000 which were held as to 23.83% by Hui Tian Tang, 13.44% by
Jing Hua Li, 10.81% by Wen De Wei, 6.81% by You Ru Jiang, 6.81% by Chun
Lin Liu and 6.81% by Bang Fu Wang, who are also the directors
of Ingenious and Baicaotang Medicine. The remaining 31.49% were
held by nine stockholders, who are the employees of Baicaotang
Medicine.
|
|
(v)
|
Guangxi
Lizhou Baicaotang Medicine (Retail Chain) Ltd. (“Baicaotang Retail”) is a
wholly owned subsidiary of Baicaotang Medicine and was established on
October 30, 2001 with registered and paid up capital of
RMB300,000. Baicaotang Retail is engaged in the retail of drugs
in the PRC.
|
|
1.
|
Corporate
information (Cont’d)
|
|
(vi)
|
Guangxi
Hefeng Pharmaceutical Co Ltd. (“Hefeng”) was established on September 18,
2000 with registered and paid up capital of
RMB5,000,000. Hefeng is engaged in the production and sale of
drugs for healing of hepatitis, cough, parkinson’s disease in the
PRC. On December 31, 2007, Baicaotang Medicine entered into an
agreement with Jing Hua Li to acquire his entire interest in
Hefeng at a consideration of RMB36,340,064 (equivalent to $4,982,223)
which was satisfied by issuance of 13.44% shares of Baicaotang
Medicine. The acquisition was completed on January 2, 2008,
which is the date Baicaotang Medicine obtained the control over Hefeng by
appointing directors into the board of directors of
Hefeng.
|
|
2.
|
Reorganization
|
| To rationalize the group structure, the Company, Ingenious, Forever Well, Baicaotang Medicine and Baicaotang Retail reorganized their group structure (the “Reorganization”) as follows :- |
|
(i)
|
Due
to certain regulatory restrictions on a wholly owned foreign enterprise or
its wholly owned subsidiary to hold over 50% equity interest in any PRC
company which operates more than 30 drug stores in the PRC (the “Drug
Stores Restrictions”), Baicaotang Medicine sold its 51% equity interest in
Baicaotang Retail to Liuzhou Baicaotang Property Management Company Ltd.
(“Baicaotang Property”), of which the directors of Ingenious are the
controlling stockholders of Baicaotang Property, at a consideration of
RMB153,000 on April 1, 2008. Afterwards, Baicaotang Property
pledged its 51% equity interest in Baicaotang Retail to Baicaotang
Medicine to secure a loan, amounted to RMB153,000, granted by Baicaotang
Medicine to Baicaotang Property up to December 31, 2015 for the
acquisition of 51% equity interest in Baicaotang
Retail. According to Repurchase Agreement dated July 31, 2008,
Baicaotang Medicine was entitled a preemptive right to repurchase the 51%
equity interest in Baicaotang Retail from Baicaotang Property up to the
earlier of December 31, 2017 or the removal of the Drug Stores
Restrictions. Before the execution of the preemptive right by
Baicaotang Medicine, the rights and obligations as a stockholder of the
51% equity interest in Baicaotang Retail are still vested in Baicaotang
Medicine and the appointment of the board of directors and management is
controlled by Baicaotang Medicine.
|
|
(ii)
|
On
June 30, 2008, the Ingenious acquired entire equity interest in Forever
Well at a cash consideration of HK$10,000, which is the issued and fully
paid up capital of Forever Well.
|
|
(iii)
|
On
March 28, 2008, Forever Well entered into an agreement with the
stockholders of Baicaotang Medicine to acquire their entire equity
interest in Baicaotang Medicine at a cash consideration of RMB10,000,000,
which is the registered and fully paid up capital of Baicaotang
Medicine. The acquisition was completed on August 4,
2008.
|
|
2.
|
Reorganization
(Cont’d)
|
|
(iv)
|
On
December 23, 2009, the Company entered into a Share Exchange Agreement
with the shareholders of Ingenious to acquire their 100% of the issued and
outstanding common shares in Ingenious by issuance of 32,000,000 shares of
the Company’s common stock, par value $0.001 per share (the “Exchange
Transaction”).
|
Following
the completion of the Exchange Transaction on December 30, 2009, 2,900,000
shares of the Company’s common stock of $0.001 each, which are held by a
Company’s shareholder, Lisa Lopomo, were cancelled on December 30, 2009.
The
Exchange Transaction, which was completed on December 30, 2009, constituted a
RTO and thereafter Ingenious became a wholly owned subsidiary of the
Company.
|
(v)
|
On
December 23, 2009, Zhang Xiao Yan entered into an Earn-in Agreement with
the former stockholders of Baicaotang Medicine of which the former
stockholders of Baicaotang Medicine are given the rights to acquire
22,480,000 common shares (Earn-in Shares) of the Company at $300,000 based
on their former respective equity interest in Baicaotang immediate before
the acquisition by Forever Well as stated in Note 2 (iii) provided that
the Company meets the profit targets, representing audited net income
after tax of $26 million for the year of 2010 and $28 million for the year
of 2011. The former stockholders are allowed to acquire 50% and 50% of
Earn-in Shares upon the profit targets have been met for the years of 2010
and 2011 respectively.
|
|
3.
|
Basis
of presentation
|
Except
for Hefeng, accounting for recapitalization is adopted for the preparation of
consolidated financial statements to present the combined results of operations
and financial position of the Company, Ingenious, Forever Well, Baicaotang
Medicine and Baicaotang Retail as if the current group structure, which means
that Ingenious, Forever Well, Baicaotang Medicine and Baicaotang Retail are
wholly owned subsidiaries of the Company, had been in existence at the beginning
of the reporting period. The 13.44% equity interest of Baicaotang
Medicine transferred for the acquisition of Hefeng as stated in Note 1 (vi) was
accounted for as 13.44% of issued 32,000,000 common stock of the Company,
amounted to $4,301 with $4,977,922 recorded in additional paid in capital during
the year ended December 31, 2008. Purchase accounting was adopted to
reflect the post-acquisition results of Hefeng, which was acquired by Baicaotang
Medicine on January 2, 2008, in these consolidated financial statements of the
Company.
|
4.
|
Acquisition
|
On
December 31, 2007, in order to diversify its business activities, Baicaotang
Medicine entered into an agreement with Li Jing Hua to acquire his entire
interest in Hefeng at a consideration of RMB36,340,064 (equivalent to
$4,982,223) which was satisfied by issuance of 13.44% shares of Baicaotang
Medicine. The acquisition was completed on January 2,
2008.
The
following table summarizes the allocation of the purchase price reflecting the
amounts assigned to Hefeng’s each major class of assets acquired and liabilities
assumed at the date of acquisition :-
|
January
2,
|
||||
|
2008
|
||||
|
Current
assets
|
$ | 3,601,575 | ||
|
Property,
plant and equipment, net
|
4,696,303 | |||
|
Land
use right
|
1,695,927 | |||
|
Goodwill
|
107,968 | |||
|
Intangible
assets
|
925,044 | |||
|
Deferred
tax assets
|
136,735 | |||
|
Current
liabilities
|
(3,439,329 | ) | ||
|
Long-term
bank loan
|
(2,742,000 | ) | ||
|
Net
assets acquired
|
$ | 4,982,223 | ||
Satisfied
by :-
|
The
fair value of 13.44% equity interest in Baicaotang
Medicine
|
$ | 4,982,223 | ||
|
Net
cash received from the acquisition of Hefeng
|
$ | 631,818 |
As of
December 31, 2008, the consolidated balance sheet includes a goodwill identified
upon the acquisition of 100% equity interest in Hefeng amounting to $107,968
which represents the excess of the purchase price representing the fair value of
13.44% equity of the Baicaotang Medicine over the attributable share of fair
value of acquired identifiable net assets of Hefeng of $4.87 million at the time
of acquisition on January 2, 2008.
The
Company with advice from an independent appraiser, has identified all assets
acquired (including intangible assets which meets either the separability
criterion or the contractual-legal criterion in accordance with ASC 805 as of
the date of acquisition with a conclusion that certain significant identifiable
intangible assets inclusive of trademark, product licences and customers
contracts was identified and recognized.
|
4.
|
Acquisition
|
The
following unaudited pro forma financial information presents the combined
results of operations of the Company with the operations of Hefeng as if the
acquisition had occurred as of the beginning of fiscal year 2007 :-
|
(Pro
Forma)
|
||||||||
|
Year
ended December 31,
|
||||||||
|
2008
|
2007
|
|||||||
|
Revenue
|
$ | 108,991,329 | $ | 70,522,398 | ||||
|
Net
income
|
$ | 16,657,162 | $ | 8,275,088 | ||||
|
Earnings
per share : basic and diluted
|
$ | 0.52 | $ | 0.27 | ||||
This
unaudited pro forma financial information is presented for informational
purposes only. The unaudited pro forma financial information may not
necessarily reflect the future results of operations or the results of
operations had the Company owned and operated this business as of the beginning
of the period presented.
|
5.
|
Summary
of significant accounting policies
|
Basis of
consolidation
The
consolidated financial statements include the accounts of the Company,
Ingenious, Forever Well, Baicaotang Medicine, Baicaotang Retail and
Hefeng. All significant inter-company accounts and transactions have
been eliminated in consolidation.
Use of
estimates
In
preparing financial statements in conformity with accounting principles
generally accepted in the United States of America, management makes estimates
and assumptions that affect the reported amounts of assets and liabilities and
disclosures of contingent assets and liabilities at the dates of the financial
statements, as well as the reported amounts of revenues and expenses during the
reporting periods. These accounts and estimates include, but are not
limited to, the valuation of accounts receivable, inventories and the estimation
on useful lives of property, plant and equipment and intangible
assets. Actual results could differ from those
estimates.
Concentrations of credit
risk
Financial
instruments that potentially subject the Company to significant concentrations
of credit risk consist principally of cash and cash equivalents, restricted
cash, trade receivables and amounts due from related companies. As of
December 31, 2009 and 2008, substantially all of the Company’s cash and cash
equivalents and restricted cash were held by major financial institutions
located in the PRC, which management believes are of high credit
quality. With respect to trade receivables, the Company extends
credit based on an evaluation of the customer’s financial
condition. The Company generally does not require collateral for
trade receivables and maintains an allowance for doubtful accounts of trade
receivables.
During
the years ended December 31, 2009 and 2008, no customers were identified with
who accounted for 10% or more of the Company’s consolidated sales and no
customers asset for or constituted 10% or more of the Company’s trade
receivables.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Cash and cash
equivalents
Cash and
cash equivalents include all cash, deposits in banks and other highly liquid
investments with initial maturities of three months or less. As of
December 31, 2009, the cash and cash equivalents were mainly denominated in
Renminbi (“RMB”) and United States Dollars were placed with banks in the PRC and
Hong Kong. For those denominated in RMB, they are not freely convertible into
foreign currencies and the remittance of these funds out of the PRC is subject
to exchange control restrictions imposed by the PRC government. The
remaining insignificant balance of cash and cash equivalents were denominated in
HK dollars.
Restricted
Cash
Deposits
in banks pledged as securities for bills payable (Note 10) that are restricted
in use are classified as restricted cash under current assets.
Allowance for doubtful
accounts
The
Company establishes an allowance for doubtful accounts based on management’s
assessment of the collectibility of trade receivables. A considerable
amount of judgment is required in assessing the amount of the allowance, the Company
considers the historical level of credit losses and applies percentages to aged
receivable categories. The Company makes judgments about the creditworthiness of
each customer based on ongoing credit evaluations, and monitors current economic
trends that might impact the level of credit losses in the future. If
the financial condition of the customers were to deteriorate, resulting in their
inability to make payments, a larger allowance may be required.
Based on
the above assessment, during the reporting years, the management establishes the
general provisioning policy to make allowance equivalent to 40% of gross amount
of trade receivables due between six months and one year and 100% of gross
amount of accounts receivable due over 1 year. Additional specific
provision is made against trade receivables whenever they are considered to be
doubtful.
Bad debts
are written off when identified. The Company extends unsecured credit
to customers ranging from three to six months in the normal course of business.
The Company does not accrue interest on trade accounts receivable.
Historically,
losses from uncollectible accounts have not significantly deviated from the
general allowance estimated by the management and no significant additional bad
debts have been written off directly to the profit and loss. This
general provisioning policy has not changed in the past since establishment and
the management considers that the aforementioned general provisioning policy is
adequate and not too excessive and does not expect to change this established
policy in the near future.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Inventories
Inventories
are stated at the lower of cost or market value. Cost is determined
on weighted average basis and includes all expenditures incurred in bringing the
goods to the point of sale and putting them in a saleable
condition. In assessing the ultimate realization of inventories, the
management makes judgments as to future demand requirements compared to current
or committed inventory levels. The Company’s reserve requirements
generally increase with its projected demand requirements; decrease due to
market conditions, product life cycle changes. The Company estimates the demand
requirements based on market conditions, forecasts prepared by its customers,
sales contracts and orders in hand. Inventory quantities and expiry
dates are regularly reviewed and provisions for excess or obsolete inventory are
recorded primarily based on the expiry dates and the Company’s forecast of
future demand and market conditions.
No
provision for excess or obsolete inventory was made for the years ended December
31, 2009 and 2008.
Property, plant and
equipment
Property,
plant and equipment are stated at cost less accumulated
depreciation. Cost represents the purchase price of the asset and
other costs incurred to bring the asset into its existing use.
Depreciation
is provided on straight-line basis over their estimated useful
lives. The principal depreciation rates are as follows
:-
|
Annual
rate
|
Residual
value
|
|||||||
|
Buildings
|
2.54% - 9.84 | % |
Nil
- 2%
|
|||||
|
Plant
and machinery
|
7% - 18.4 | % |
Nil
- 10%
|
|||||
|
Motor
vehicles
|
6% -18.4 | % | 10 | % | ||||
|
Furniture,
fixtures and equipment
|
6% -18.4 | % | 10 | % | ||||
Construction
in progress mainly represents expenditures in respect of the construction of a
new production line and improving the manufacturing process. All
direct costs relating to the new production line and improving the manufacturing
process are capitalized as construction in progress. No depreciation is provided
in respect of construction in progress.
Maintenance
or repairs are charged to expense as incurred. Upon sale or
disposition, the applicable amounts of asset cost and accumulated depreciation
are removed from the accounts and the net amount less proceeds from disposal is
charged or credited to income.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Goodwill and intangible
assets
The
Company applies the provisions of ASC 805, “Goodwill and Intangible Assets”
(“ASC 805”). Under ASC 805, goodwill and intangible assets with indefinite
useful lives are not amortized, but instead are tested for impairment at least
annually.
Goodwill
with infinite useful life are stated at cost less accumulated
impairment. The Company completed the annual impairment tests as of
December 31, 2009.
Pharmaceutical
licences, customer contracts, trademarks, know-how and patents are stated at
cost less accumulated amortization. Amortization is provided on a straight-line
over their useful lives as follows :-
| Pharmaceutical licences | 10 years | ||
| Customer contracts, trademarks, know-how and patents | 1-3 years |
Land use
right
Land use
rights are stated at cost less accumulated amortization. Amortization
is provided using the straight-line method over the terms of the lease of 40 to
70 years obtained from the relevant PRC land authority.
Impairment of long-lived
assets
Long-lived
assets are tested for impairment in accordance with ASC 360-10-45 “Impairment or
Disposal of Long-Lived Assets” (previously SFAS No. 144). The Company
periodically evaluates potential impairment whenever events or changes in
circumstances indicate that the carrying amount of the assets may not be
recoverable. The Company recognizes impairment of long-lived assets
in the event that the net book values of such assets exceed the future
undiscounted cashflows attributable to such assets. During the
reporting periods, the Company has not identified any indicators that would
require testing for impairment.
Government
grants
Receipts
of government grants to encourage research and development activities, which are
non-refundable, are credited to deferred income upon
receipt. Government grants are used for purchases of property, plant
and equipment, to subsidize the research and development expenses incurred, for
compensation expenses already incurred or for good performance of the
Company.
Grants
applicable to purchase of property, plant and equipment are amortized over the
life of the depreciable assets. For research and development
expenses, the Company matches and offsets the government grants with the
expenses of the research and development activities as specified in the grant
approval document in the corresponding period when such expenses are
incurred. Government grants received as compensation for expenses
already incurred in the prior period or for good performance of the Company are
recognized as income in the period they become recognizable.
During
the year ended December 31, 2009, the Company received government grants of
$29,320 for good performance. The Company recognized this amount as income for
the year. No government grant was received and recognized as income
for the year ended December 31, 2008.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Revenue
recognition
Revenue
from sales of the Company’s products is recognized when the significant risks
and rewards of ownership have been transferred to the buyer at the time when the
products are delivered to its customers, the sales price is fixed or
determinable and collection is reasonably assured.
Advertising, research and
development expenses
Advertising
and research and development expenses are charged to expense as
incurred.
Advertising
expenses amounting to $123,195 and $158,997 for the year ended December 31, 2009
and 2008 are included in selling expenses.
Research
and development expenses consist primarily of remuneration for research and
development staff and material costs for research and development.
Research
and development expenses amounting to $99,688 and $763,995 for the year ended
December 31, 2009 and 2008 are included in operating expenses.
Retirement benefit
costs
Baicaotang
Medicine adopted a retirement scheme to provide for eligible staff employed
prior to April 23, 2002. The eligible staff are entitled to receive
certain amounts based on their years of service in Baicaotang Medicine up to
April 23, 2002, upon their termination of employment relationship with the
Company or retirement. The obligation of retirement benefit costs is
recorded at the present value of the cost expected to settle the obligation and
is recognised when the retirement scheme has been approved. The staff
employed after April 23, 2002 is not entitled to this retirement
scheme.
As of
December 31, 2009, the discount rate of 7.14%, which represented Baicaotang ’s
weighted average borrowing rate of Renminbi loans in the PRC, was adopted to
calculate the present value of retirement benefits cost.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Income
taxes
The
Company uses the asset and liability method of accounting for income taxes
pursuant to ASC 740 "Income Taxes” (previously SFAS No. 109). Under
the asset and liability method of ASC 740, deferred tax assets and liabilities
are recognized for the future tax consequences attributable to temporary
differences between the financial statements carrying amounts of existing assets
and liabilities and loss carryforwards and their respective tax
bases. Deferred tax assets and liabilities are measured using enacted
tax rates expected to apply to taxable income in the years in which those
temporary differences are expected to be recovered or settled.
Dividends
Dividends
are recorded in Company’s financial statements in the period in which they are
declared.
Off-balance sheet
arrangements
The
Company does not have any off-balance sheet arrangements.
Comprehensive
income
The
Company has adopted ASC 220, “Comprehensive Income”, which establishes standards
for reporting and display of comprehensive income (loss), its components and
accumulated balances. Components of comprehensive income (loss) include net
income (loss) and foreign currency translation adjustments.
Foreign currency
translation
The
functional currency of the Company is RMB and RMB is not freely convertible into
foreign currencies. The Company maintains its financial statements in the
functional currency. Monetary assets and liabilities denominated in
currencies other than the functional currency are translated into the functional
currency at rates of exchange prevailing at the balance sheet
date. Transactions denominated in currencies other than the
functional currency are translated into the functional currency at the exchanges
rates prevailing at the dates of the transaction. Exchange gains or
losses arising from foreign currency transactions are included in the
determination of net income for the respective periods.
For
financial reporting purposes, the financial statements of the Company which are
prepared using the functional currency have been translated into United States
dollars. Assets and liabilities are translated at the exchange rates
at the balance sheet dates and revenue and expenses are translated at the
average exchange rates and stockholders’ equity is translated at historical
exchange rates. Any translation adjustments resulting are not
included in determining net income but are included in foreign exchange
adjustment to other comprehensive income, a component of stockholders’
equity. The exchange rates in effect at December 31, 2009, 2008 and
2007 were RMB1 for $0.1467, $0.1467 and $0.1371 respectively. There
is no significant fluctuation in exchange rate for the conversion of RMB to US
dollars after the balance sheet date.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Basic and diluted earnings
per share
The
Company reports basic earnings per share in accordance with ASC 260, “Earnings
Per Share” (previously SFAS No. 128). Basic earnings (loss) per share
is computed using the weighted average number of shares outstanding during the
periods presented. The weighted average number of shares of the
Company represents the common stock outstanding during the reporting
periods. Diluted earnings per share is computed using the weighted
average number of common shares outstanding during the periods plus the effect
of dilutive securities outstanding during the periods. At December
31, 2009, the Company had outstanding warrants exercisable into shares of common
stock.
Fair value of financial
instruments
The
Company adopted ASC 820 (previously Statement of Financial Accounting Standards
("SFAS") No. 157) on January 1, 2008. The adoption of ASC 820 did not
materially impact the Company's financial position, results of operations or
cash flows.
ASC 820
requires the disclosure of the estimated fair value of financial instruments
including those financial instruments for which fair value option was not
elected. The carrying amounts of the financial assets and liabilities
approximate to their fair values due to short maturities or the applicable
interest rates approximate the current market rates.
Recently issued accounting
pronouncements
FASB
Accounting Standards Codification (Accounting Standards Update “ASU” 2009-1). In
June 2009, the Financial Accounting Standard Board (“FASB”) approved its
Accounting Standards Codification (“Codification”) as the single source of
authoritative United States accounting and reporting standards applicable for
all non-governmental entities, with the exception of the SEC and its staff. The
Codification is effective for interim or annual financial periods ending after
September 15, 2009 and impacts our financial statements as all future references
to authoritative accounting literature will be referenced in accordance with the
Codification. There have been no changes to the content of our financial
statements or disclosures as a result of implementing the
Codification.
As a
result of our implementation of the Codification during the quarter ended
September 30, 2009, previous references to new accounting standards and
literature are no longer applicable. In the current financial statements, we
will provide reference to both new and old guidance to assist in understanding
the impacts of recently adopted accounting literature, particularly for guidance
adopted since the beginning of the current fiscal year but prior to the
Codification.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Recently issued accounting
pronouncements (cont’d)
Noncontrolling Interests (Included
in amended Topic ASC 810 “Consolidation”, previously SFAS No. 160
“Noncontrolling Interests in Consolidated Financial Statements”, an amendment of
ARB No. 51). The amended topic establishes accounting and reporting
standards for the noncontrolling interest in a subsidiary and for the
deconsolidation of a subsidiary. It clarifies that a noncontrolling interest in
a subsidiary is an ownership interest in the consolidated entity that should be
reported as equity in the consolidated financial statements. The
adoption of this amended topic has no material impact on the Company’s financial
statements.
Business Combinations (Included in
amended Topic ASC 805 “Business Combinations”, previously SFAS No. 141(R)).
This ASC guidance addresses the accounting and disclosure for
identifiable assets acquired, liabilities assumed, and noncontrolling interests
in a business combination. The adoption of this amended topic has no material
impact on the Company’s financial statements.
Intangibles-Goodwill and Other
(Included in amended Topic ASC 350”, previously FASB staff position (“FSP”) FAS
142-3, Determination of the Useful Life of Intangible Assets). The
amended topic amends the factors an entity should consider in developing renewal
or extension assumptions used in determining the useful life of recognized
intangible assets under FASB Statement No. 142, “Goodwill and Other
Intangible Assets”. This new guidance applies prospectively to intangible assets
that are acquired individually or with a group of other assets in business
combinations and asset acquisitions. The amended topic is effective for
financial statements issued for fiscal years and interim periods beginning after
December 15, 2008. Early adoption is prohibited. The adoption of this
amended topic has no material effect on the Company's financial
statements.
Business Combinations (Included in
amended Topic ASC 805, previously FSP No. 141R-1 “Accounting for Assets Acquired
and Liabilities Assumed in a Business Combination That Arise from
Contingencies”). Amended topic ASC 805 amends the requirements for the
provisions in FASB Statement 141R for the initial recognition and measurement,
subsequent measurement and accounting, and disclosures for assets and
liabilities arising from contingencies in business combinations. The amended
topic eliminates the distinction between contractual and non-contractual
contingencies, including the initial recognition and measurement criteria and
instead carries forward most of the provisions for acquired contingencies. The
amended topic is effective for contingent assets and contingent liabilities
acquired in evaluating the impact. The adoption of this amended topic has no
material impact on the Company’s financial statements.
Fair Value Measurements and
Disclosures (Included in amended Topic ASC 820, previously FSP No. 157-4,
“Determining Whether a Market is Not Active and a Transaction Is Not
Distressed”.) The amended topic clarifies when markets are illiquid or
that market pricing may not actually reflect the “real” value of an asset. If a
market is determined to be inactive and market price is reflective of a
distressed price then an alternative method of pricing can be used, such as a
present value technique to estimate fair value. The amended topic identifies
factors to be considered when determining whether or not a market is inactive.
The amended topic would be effective for interim and annual periods ending after
June 15, 2009, with early adoption permitted for periods ending after March 15,
2009 and shall be applied prospectively. The adoption of this amended topic has
no material effect on the Company's financial statements.
|
5.
|
Summary
of significant accounting policies
(cont’d)
|
Recently issued accounting
pronouncements (cont’d)
Investments - Debt and Equity
Securities - Overall - Transition and Open Effective Date Information (Included
in amended Topic ASC 320, previously FASB Staff Position No. 115-2 and Statement
of Financial Accounting Standards No. 124-2, “Recognition and Presentation of
Other-Than-Temporary Impairments”). The amended topic amends the
other-than-temporary impairment guidance in U.S. GAAP for debt securities
through increased consistency in the timing of impairment recognition and
enhanced disclosures related to the credit and noncredit components of impaired
debt securities that are not expected to be sold. In addition, increased
disclosures are required for both debt and equity securities regarding expected
cash flows, credit losses, and securities with unrealized losses. The adoption
of this amended topic has no material impact on the Company’s financial
statements.
Interim Disclosures about Fair Value
of Financial Instruments (Included in amended Topic ASC 825 “Financial
Instruments”, previously FSP SFAS No. 107-1). This guidance requires that
the fair value disclosures required for all financial instruments be included in
interim financial statements. This guidance also requires entities to disclose
the method and significant assumptions used to estimate the fair value of
financial instruments on an interim and annual basis and to highlight any
changes from prior periods. The amended topic was effective for interim periods
ending after September 15, 2009. The adoption of this amended topic has no
material impact on the Company’s financial statements.
Subsequent Events (Included in
amended Topic ASC 855 “Subsequent Events”, previously SFAS No. 165). The
amended topic establishes accounting and disclosure requirements for subsequent
events. The amended topic details the period after the balance sheet date during
which the Company should evaluate events or transactions that occur for
potential recognition or disclosure in the financial statements, the
circumstances under which the Company should recognize events or transactions
occurring after the balance sheet date in its financial statements and the
required disclosures for such events. The Company adopted this amended topic
effective June 1, 2009.
Accounting for Transfers of
Financial Assets (Included in amended Topic ASC 860 “Transfers and Servicing”,
previously SFAS No. 166, “Accounting for Transfers of Financial Assets - an
Amendment of FASB Statement No. 140.”). The amended topic addresses
information a reporting entity provides in its financial statements about the
transfer of financial assets; the effects of a transfer on its financial
position, financial performance, and cash flows; and a transferor’s continuing
involvement in transferred financial assets. Also, the amended topic removes the
concept of a qualifying special purpose entity, limits the circumstances in
which a transferor derecognizes a portion or component of a financial asset,
defines participating interest and enhances the information provided to
financial statement users to provide greater transparency. The amended topic is
effective for the first annual reporting period beginning after November 15,
2009 and will be effective for us as of January 1, 2010. The management is in
the process of evaluating the impact of adopting this amended topic on the
Company’s financial statements.
5. Summary
of significant accounting policies (cont’d)
Recently issued accounting
pronouncements (cont’d)
Consolidation of Variable Interest
Entities – Amended (Included in amended Topic ASC 810 “Consolidation”,
previously SFAS 167 “Amendments to FASB Interpretation No. 46(R)”). The
amended topic requires an enterprise to perform an analysis to determine the
primary beneficiary of a variable interest entity; to require ongoing
reassessments of whether an enterprise is the primary beneficiary of a variable
interest entity and to eliminate the quantitative approach previously required
for determining the primary beneficiary of a variable interest entity. The
amended topic also requires enhanced disclosures that will provide users of
financial statements with more transparent information about an enterprise’s
involvement in a variable interest entity. The amended topic is effective for
the first annual reporting period beginning after November 15, 2009 and will be
effective for us as of January 1, 2010. The management is in the process of
evaluating the impact of adopting this amended topic on the Company’s financial
statements.
In August
2009, the FASB issued Accounting Standards Update (“ASU”) No. 2009-05 (“ASU
2009-05”), an update to ASC 820, Fair Value Measurements and Disclosures. This
update provides amendments to reduce potential ambiguity in financial reporting
when measuring the fair value of liabilities. Among other provisions, this
update provides clarification that in circumstances in which a quoted price in
an active market for the identical liability is not available, a reporting
entity is required to measure fair value using one or more of the valuation
techniques described in ASU 2009-05. ASU 2009-05 became effective for the
Company’s annual financial statements for the year ended December 31, 2009. The
adoption of this ASU has no material impact on the Company’s financial
statements.
The FASB
issued Accounting Standards Update (ASU) No. 2009-13, Revenue Recognition (Topic
605): Multiple Deliverable Revenue Arrangements - A Consensus of the FASB
Emerging Issues Task Force.” This update provides application guidance on
whether multiple deliverables exist, how the deliverables should be separated
and how the consideration should be allocated to one or more units of
accounting. This update establishes a selling price hierarchy for determining
the selling price of a deliverable. The selling price used for each deliverable
will be based on vendor-specific objective evidence, if available, third-party
evidence if vendor-specific objective evidence is not available, or estimated
selling price if neither vendor-specific or third-party evidence is available.
The Company will be required to apply this guidance prospectively for revenue
arrangements entered into or materially modified after January 1, 2011; however,
earlier application is permitted. The management is in the process of evaluating
the impact of adopting this ASU on the Company’s financial
statements.
The FASB
issued ASU-2010-09 (Topic 855) to amend guidance on subsequent events to remove
the requirement for SEC filers (as defined in ASU 2010-09) to disclose the date
through which an entity has evaluated subsequent events. This change alleviates
potential conflicts with current SEC guidance. An SEC filer is still required to
evaluate subsequent events through the date financial statements are issued, but
disclosure of that date is no longer required. The amendments in ASU 2010-09
became effective upon issuance of the guidance.
|
6.
|
Other
income
|
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Rental
income
|
$ | 69,086 | $ | 86,008 | ||||
|
Sales
of raw material
|
209 | 36,270 | ||||||
|
Other
operating income
|
26,050 | 21,148 | ||||||
| $ | 95,345 | $ | 143,426 | |||||
|
7.
|
Finance
costs
|
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Bank
loans interest
|
$ | 1,007,423 | $ | 1,171,373 | ||||
|
Other
loans interest
|
257,111 | 58,296 | ||||||
|
Imputed
interest on retirement benefit costs
|
20,349 | 22,594 | ||||||
|
Bank
charges
|
92,969 | 4,482 | ||||||
|
Discounting
charges
|
35,994 | - | ||||||
|
Others
|
27 | 3,545 | ||||||
| $ | 1,413,873 | $ | 1,260,290 | |||||
|
8.
|
Income
taxes
|
United
States
China
Baicaotang Medicine Limited is subject to the United States of America Tax law
at tax rate of 34%. No provision for the US federal income taxes has
been made as the Company had no taxable income in this jurisdiction for the
reporting period.
BVI
Ingenious
was incorporated in the BVI and, under the current laws of the BVI, is not
subject to income taxes.
Hong
Kong
Forever
Well is subject to the Hong Kong Profits tax at tax rate of 16.5% (2008:
16.5%). No provision for the Hong Kong Profits tax has been made as
the Company had no taxable income in this jurisdiction since its
incorporation.
|
8.
|
Income
taxes (cont’d)
|
PRC
Corporate
income tax (“CIT”) to Baicaotang Medicine and Baicaotang Retail, was charged at
33%, of which 30% is for national tax and 3% is for local tax, of the assessable
profits before 2008. The PRC’s legislative body, the National
People’s Congress, adopted the unified CIT Law on March 16,
2007. This new tax law replaces the existing separate income tax laws
for domestic enterprises and foreign-invested enterprises and became effective
on January 1, 2008. Under the new tax law, a unified income tax
rates is set at 25% for both domestic enterprises and foreign-invested
enterprises. However, there is a transition period for enterprises,
whether foreign-invested or domestic, that are currently receiving preferential
tax treatments granted by relevant tax authorities. Enterprises that
are subject to an enterprise income tax rate lower than 25% may continue to
enjoy the lower rate and will transit into the new tax rate over a five-year
period beginning on the effective date of the CIT Law. Enterprises
that are currently entitled to exemptions for a fixed term will continue to
enjoy such treatment until the exemption term expires. Preferential
tax treatment will continue to be granted to industries and projects that
qualify for such preferential treatments under the new tax law. Accordingly,
Baicaotang Medicine and Baicaotang Retail was subject to tax rate of 25%
starting from fiscal year 2008.
In
accordance with the Circular of the State Council on Policies and Measures
Pertaining to the Development of the Western Region (“DOWR”), the companies are
entitled to preferential rate of 15% if it is engaged in the projects listed in
Guiding Catalogue and the revenue derived from it account for the amount over
70% of total revenue. As Hefeng met this DOWR requirement, it was
approved by the tax authority and was granted a preferential tax rate of 15% for
fiscal years 2003 to 2010. From fiscal year 2011, Hefeng will be subject to CIT
at rate of 25% under the new tax law.
The
components of the provision (benefit) for income taxes are :-
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Current
taxes - PRC
|
$ | 6,250,855 | $ | 5,709,713 | ||||
|
Deferred
taxes - PRC
|
10,943 | (52,835 | ) | |||||
| $ | 6,261,798 | $ | 5,656,878 | |||||
|
8.
|
Income
taxes (cont’d)
|
The
effective income tax expenses differs from the PRC statutory income tax rate of
25% for the year ended December 31, 2009 and 2008 in the PRC as follows
:-
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Provision
for income taxes at PRC
|
||||||||
|
statutory
income tax rate
|
$ | 6,417,591 | $ | 5,578,510 | ||||
|
Non-deductible
items for tax
|
229,848 | 216,461 | ||||||
|
Decrease
in deferred tax assets resulting
|
||||||||
|
from a reduction of PRC
statutory
|
||||||||
|
income tax rate from 33% to
25%
|
- | 127,379 | ||||||
|
Under
provision in prior year
|
122 | - | ||||||
|
Tax
holiday
|
(338,135 | ) | (260,751 | ) | ||||
|
Others
|
(47,628 | ) | (4,721 | ) | ||||
| $ | 6,261,798 | $ | 5,656,878 | |||||
During
the year ended December 31, 2009, the amount of benefit from tax holiday was
$338,135 (2008: $260,751) and the effect on earnings per share was $0.01
(2008: $0.01).
In July
2006, the FASB issued ASC 740-10-25 (previously Interpretation No. 48,
“Accounting for Uncertainty in Income Taxes”). This interpretation
requires recognition and measurement of uncertain income tax positions using a
“more-likely-than-not” approach. The Company adopted this ASC
740-10-25 on January 1, 2007. Under the new CIT Law effective on
January 1, 2008, the Company may be deemed to be a resident enterprise by the
PRC tax authorities. If the Company was deemed to be resident
enterprise, the Company may be subject to the CIT at 25% on the worldwide
taxable income and dividends paid from PRC subsidiaries to their overseas
holding companies may be exempted from 10% PRC withholding tax. Except for
certain immaterial interest income from bank deposits placed with financial
institutions outside the PRC, all of the Company’s income is generated from the
PRC operation. Given the immaterial amount of income generated from
outside the PRC and the PRC subsidiaries do not intend to pay dividends in the
foreseeable future, the management considers that the impact arising from
resident enterprise on the Company’s financial position is not
significant. The management evaluated the Company’s overall tax
positions and considered that no additional provision for uncertainty in income
taxes is necessary as of December 31, 2009.
Deferred
tax assets/(liabilities) as of December 31, 2009 and 2008 are composed of the
followings :-
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
The PRC
|
||||||||
|
Current deferred tax assets
:-
|
||||||||
|
Allowance for doubtful
debts
|
$ | 60,164 | $ | 60,164 | ||||
|
Non current deferred tax
assets
|
||||||||
|
(liabilities)
:-
|
||||||||
|
Depreciation of
property, plant
|
||||||||
|
and
equipment
|
$ | 1,860,796 | $ | 1,952,809 | ||||
|
Amortization of
land use right
|
(1,380,844 | ) | (1,446,503 | ) | ||||
|
Amortization of
intangible assets
|
183,747 | 168,343 | ||||||
| $ | 663,699 | $ | 674,649 | |||||
|
9.
|
Earnings
per share
|
During
the reporting periods, potential dilutive shares are excluded from the
computation of earnings per share if their effect is anti-dilutive. 1,493,305
anti-dilutive warrants were excluded from the calculation of earnings per share
for year of 2009. Accordingly, the basic and diluted earnings per share are the
same.
The per
share data reflects the reorganization of stockholders’ equity as if the
Reorganization occurred as of the beginning of the first period
presented.
|
10.
|
Restricted
cash and bills payable
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Bank
deposits held as collateral for bills
|
||||||||
|
payable
|
$ | 1,155,779 | $ | 1,228,011 | ||||
The
Company is requested by certain of its suppliers to settle by issuance of bills
for which the banks add their undertakings to guarantee their settlement at
maturity. These bills are interest free with maturity of three to six months
from date of issuance. As security for the banks undertakings, the Company is
required to deposit with such banks equal to 50% to 100% of the bills amount at
the time of issuance and pay bank charges. These deposits will be
used to settle the bills at maturity.
|
11.
|
Trade
receivables, net
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Trade
receivables
|
$ | 35,554,436 | $ | 21,120,600 | ||||
|
Less
: allowance for doubtful accounts
|
(144,397 | ) | (144,397 | ) | ||||
|
Net
|
$ | 35,410,039 | $ | 20,976,203 | ||||
An
analysis of the allowance for doubtful accounts for the years ended December 31,
2009 and 2008 is as follows :-
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Balance
at beginning of year
|
$ | 144,397 | $ | 215,225 | ||||
|
Recovery
of doubtful debts
|
- | (84,091 | ) | |||||
|
Translation
adjustments
|
- | 13,263 | ||||||
|
Balance
at end of year
|
$ | 144,397 | $ | 144,397 | ||||
No trade
receivables (2008: US$1,606,516) were pledged as collateral under certain loan
agreements as of December 31, 2009.
|
12.
|
Other
receivables, prepayments and
deposits
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Advances
to staff
|
$ | 432,700 | $ | 210,322 | ||||
|
Prepayments
|
1,365,600 | 959,289 | ||||||
|
Other
receivables
|
728,098 | 82,916 | ||||||
| $ | 2,526,398 | $ | 1,252,527 | |||||
|
13.
|
Inventories
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Raw
materials
|
$ | 722,109 | $ | 509,394 | ||||
|
Work-in-progress
|
148,954 | 119,680 | ||||||
|
Finished
goods
|
7,874,462 | 5,796,691 | ||||||
| $ | 8,745,525 | $ | 6,425,765 | |||||
|
14.
|
Goodwill
and other Intangible assets
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Goodwill
|
||||||||
|
Acquisition
of Hefeng - Note 14(a)
|
$ | 107,968 | $ | 107,968 | ||||
|
Other
intangible assets
|
||||||||
|
Pharmaceutical
licences
|
$ | 799,197 | $ | 799,197 | ||||
|
Customer
contracts
|
90,729 | 90,729 | ||||||
|
Trademarks,
know-how and patents
|
99,892 | 99,892 | ||||||
| 989,818 | 989,818 | |||||||
|
Accumulated
amortization
|
(329,784 | ) | (203,769 | ) | ||||
|
Net
|
$ | 660,034 | $ | 786,049 | ||||
|
14.
|
Goodwill
and other Intangible assets
(cont’d)
|
Notes
:-
|
|
(a)
|
The
amount represents goodwill identified upon acquisition of Hefeng as stated
in Note 4.
|
During
the years ended December 31, 2009 and 2008 amortization charge was $118,565 and
$200,227 respectively.
The
estimated aggregate amortization expenses for other intangible assets for the
five succeeding years is as follows :-
|
Year
|
||||
|
2010
|
$ | 113,217 | ||
|
2011
|
79,920 | |||
|
2012
|
79,920 | |||
|
2013
|
79,920 | |||
|
2014
|
79,920 | |||
| $ | 432,897 | |||
|
15.
|
Property,
plant and equipment, net
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Costs
:-
|
||||||||
|
Buildings
|
$ | 12,331,575 | $ | 12,091,574 | ||||
|
Plant
and machinery
|
1,291,734 | 1,278,098 | ||||||
|
Furniture,
fixtures and equipment
|
374,615 | 348,437 | ||||||
|
Motor
vehicles
|
367,132 | 334,466 | ||||||
| 14,365,056 | 14,052,575 | |||||||
|
Accumulated
depreciation
|
(2,544,420 | ) | (1,932,154 | ) | ||||
| 11,820,636 | 12,120,421 | |||||||
|
Construction
in progress
|
351,053 | 292,853 | ||||||
|
Net
|
$ | 12,171,689 | $ | 12,413,274 | ||||
|
|
(a)
|
An
analysis of buildings, plant and machinery pledged to banks for banking
loans (Note 21(d)(i)) is as follows
:-
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Costs
:-
|
||||||||
|
Buildings
|
$ | 7,409,774 | $ | 10,782,432 | ||||
|
Furniture,
fixtures and equipment
|
- | 929,092 | ||||||
| 7,409,774 | 11,711,524 | |||||||
|
Accumulated
depreciation
|
(1,290,533 | ) | (1,289,333 | ) | ||||
|
Net
|
$ | 6,119,241 | $ | 10,422,191 | ||||
|
15.
|
Property,
plant and equipment, net (cont’d)
|
|
|
(b)
|
During
the reporting periods, depreciation is included in
:-
|
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Cost
of sales and overheads of
|
||||||||
|
inventories
|
$ | 157,256 | $ | 152,816 | ||||
|
Selling
expenses
|
1,198 | 4,701 | ||||||
|
Administrative
expenses
|
439,822 | 475,383 | ||||||
| $ | 598,276 | $ | 632,900 | |||||
|
|
(c)
|
Construction
in progress
|
Construction
in progress mainly represents expenditures in respect of the construction of a
new production line and improving the manufacturing process.
|
16.
|
Land
use rights
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Land
use rights
|
$ | 16,039,294 | $ | 17,445,819 | ||||
|
Accumulated
amortization
|
(2,059,541 | ) | (1,778,651 | ) | ||||
|
Net
|
$ | 13,979,753 | $ | 15,667,168 | ||||
The
Company has obtained land use rights from the relevant PRC land authority for a
period of 40 to 70 years to use the land on which the office premises,
production facilities and warehouse of the Company are situated. As of December
31, 2009, and 2008, land use rights with carrying amount of $6,205,140 and
$7,788,781 respectively were pledged to a bank for the bank loans granted to the
Company (Note 21(d)(ii)).
During
the years ended December 31, 2009 and 2008, amortization amounted to $313,533
and $334,206 respectively.
During
the year ended December 31, 2009, land use right with carrying amounts of
$1,373,773 were disposed of at considerations, net direct costs, of $1,391,587
resulting in gain of $17,814.
The
estimated aggregate amortization expenses for land use right for the five
succeeding years is as follows :-
|
Year
|
||||
|
2010
|
$ | 312,705 | ||
|
2011
|
312,705 | |||
|
2012
|
312,705 | |||
|
2013
|
312,705 | |||
|
2014
|
312,705 | |||
| $ | 1,563,525 | |||
|
17.
|
Other
payables and accrued expenses
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Accrued
audit fee
|
$ | 102,000 | $ | 160,000 | ||||
|
Other
accrued expenses
|
217,256 | 572,684 | ||||||
|
Deposits
received
|
995,387 | 819,908 | ||||||
|
Accrued
interest expenses
|
16,339 | 36,264 | ||||||
|
Accrued
staff costs - Note 17(a)
|
1,046,820 | 772,067 | ||||||
|
VAT
and other tax payables
|
471,690 | 407,034 | ||||||
|
Temporary
receipt
|
- | 146,700 | ||||||
|
Other
payables
|
345,120 | 315,920 | ||||||
| $ | 3,194,612 | $ | 3,230,577 | |||||
Notes
:-
|
|
a)
|
The
amount included accrued salaries and wages, staff welfare and accrued
social insurance to the PRC municipal and provincial governments which
cover pensions, unemployment and medical insurances and staff housing
fund.
|
|
18.
|
Dividends
|
Dividends
of $Nil and $6,940,000 declared during the years ended December 31, 2009 and
2008 respectively were made by Baicaotang Medicine to its former stockholders in
proportion to the percentage of their holdings of paid up capital.
|
19.
|
Amounts
due to directors
|
The
amounts are unsecured and repayable on demand. Except for the amounts of
$836,084 (2008:$744,817) as of December 31, 2009 which were interest bearing at
fixed rates ranging from 3.2% to 8.16% (2008:3.2%) per annum, the remaining
balances are interest-free.
|
20.
|
Amounts
due from/to related companies
|
The
related companies are controlled by certain Company’s directors collectively
inclusive of Hui Tian Tang. These amounts are interest-free, unsecured and
repayable on demand.
|
21.
|
Secured
bank loans
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Short-term
loans - Note 21(a)
|
$ | 7,070,940 | $ | 8,935,497 | ||||
|
Current
maturities of long-term bank
loan
|
65,129 | 1,355,508 | ||||||
| $ | 7,136,069 | $ | 10,291,005 | |||||
|
Long-term
bank loans - Note
21(b)
|
$ | 3,697,086 | $ | 3,556,008 | ||||
|
Less:
current maturities
|
(65,129 | ) | (1,355,508 | ) | ||||
| $ | 3,631,957 | $ | 2,200,500 | |||||
|
|
(a)
|
The
weighted average interest rates for short-term loans as of December 31,
2009 and 2008 were 7.14% and 10.96% per annum
respectively.
|
|
|
(b)
|
Except
for loans of $3,726,180 as of December 31, 2009 which were interest
bearing at fixed rates ranging from 6.138% to 9.855% per annum, the
remaining balances as of December 31, 2009 were interest bearing at
variable rates ranging from HIBOR plus 5.841% to HIBOR plus 7.02% per
annum respectively.
|
|
|
(c)
|
As
of December 31, 2009, the Company’s banking facilities were composed of
the following :-
|
|
Amount
|
||||||||||||
|
Facilities
granted
|
Granted
|
Utilized
|
Unused
|
|||||||||
|
Secured
bank loans
|
$ | 10,768,026 | $ | 10,768,026 | $ | - | ||||||
|
|
(d)
|
As
of December 31,2009, The above bank loans were secured by the following
:-
|
|
|
(i)
|
Property,
plant and equipment with carrying value of $6,119,241 (Note
15);
|
|
|
(ii)
|
Land
use rights with carrying value of $6,205,140 (Note
16);
|
|
|
(iii)
|
Buildings
and land use right owned by a related company which is controlled by
certain of the Company's
directors.
|
| 21. | Secured bank loans (cont’d) |
During
the reporting periods, there was no covenant requirement under the banking
facilities granted to the Company.
The
aggregate maturities of long-term bank loans as of December 31, 2009 are as
follows:
Fiscal
years ending on December 31,
|
Year
|
||||
|
2010
|
$ | 65,129 | ||
|
2011
|
1,757,354 | |||
|
2012
|
1,689,590 | |||
|
2013
and afterward
|
185,013 | |||
| $ | 3,697,086 | |||
|
22.
|
Other
loans
|
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Interest
bearing
|
||||||||
|
-
staff - Note 22(a)
|
$ | 377,067 | $ | 638,145 | ||||
|
-
third parties - Note 22(a)
|
1,544,091 | 1,793,994 | ||||||
| 1,921,158 | 2,432,139 | |||||||
|
Non
interest bearing
|
||||||||
|
-
third parties
|
440,100 | - | ||||||
| $ | 2,361,258 | $ | 2,432,139 | |||||
|
|
(a)
|
Interest
bearing at a fixed rate of 0.2667% to 0.6% per
month.
|
|
|
(b)
|
All
the other loans are unsecured and repayable on
demand.
|
|
23.
|
Commitments
and contingencies
|
|
|
a.
|
Capital
commitment
|
As of
December 31, 2009, the Company had no capital commitments in respect of the
acquisition of property, plant and equipment which were contracted for but not
provided in the consolidated financial statements.
|
|
b.
|
Operating lease
commitments
|
As of
December 31, 2009, the Company had non-cancelable operating leases for its
retail shops and future minimum lease payment to be paid are as
follows:-
|
Year
|
||||
|
2010
|
$ | 253,495 | ||
|
2011
|
223,273 | |||
|
2012
|
163,491 | |||
|
2013
and afterward
|
66,644 | |||
| $ | 706,903 | |||
The
rental expense relating to the operating leases was $309,833 and $229,013 for
the years ended December 31, 2009 and 2008 respectively.
|
|
c.
|
Operating lease
arrangement
|
As of
December 31, 2009, the Company leases its retail shops in PRC under an operating
lease arrangement until 2010. Future minimum lease payments to be received under
non-cancelable operating lease are as follows:-
|
As
of December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Within
one year
|
$ | 11,903 | $ | 8,950 | ||||
|
24.
|
Common
stock and additional paid-in
capital
|
|
Additional
|
||||||||||||
|
Number
of
|
paid-in
|
|||||||||||
|
shares
|
Amount
|
capital
|
||||||||||
|
Balance,
December 31, 2008
|
32,000,000 | $ | 32,000 | $ | 9,596,632 | |||||||
|
Recapitalization
- Note 24(d)
|
2,600,000 | 2,600 | 20,094 | |||||||||
|
Private
placement - Note 24(b)
|
2,489,370 | 2,489 | 5,304,173 | |||||||||
|
Balance,
December 31, 2009
|
37,089,370 | $ | 37,089 | $ | 14,920,899 | |||||||
|
(a)
|
On
December 30, 2009, the Company issued 32,000,000 shares of common stock at
par value $0.001 each to the shareholders of Ingenious in exchange for
their 100% issued and outstanding common stock in
Ingenious.
|
|
(b)
|
On
December 30, 2009, the Company completed a private placement for of
2,489,370 shares of common stock and warrants to purchase up to 1,244,368
shares of common stock at an exercise price per share of $3.81 per share
for gross proceed of $6,322,952 with related issuance expenses of
$1,016,290. In connection with the private placement, warrants to purchase
up to 248,937 shares of common stock at an exercise price of $3.05 per
shares were issued to Co-Placement
Agent.
|
|
(c)
|
On
December 30, 2009, 2,900,000 shares of the Company’s common stock, which
were held by a Company’s shareholder, Lisa Lopomo, were
cancelled.
|
|
(d)
|
The
Company’s issued and outstanding 2,600,000 shares of common stock prior to
the RTO were accounted for as $22,694, the net book value at the time of
RTO.
|
|
25.
|
Statutory
surplus reserves
|
Under PRC
regulations, Baicaotang Medicine, Baicaotang Retail and Hefeng may pay dividends
only out of their accumulated profits, if any, determined in accordance with PRC
GAAP. In addition, these companies are required to set aside at least
10% of their after-tax net profits each year, if any, to fund the statutory
reserves until the balance of the reserves reaches 50% of their registered
capital. The statutory reserves are not distributable in the form of
cash dividends to the Company and can be used to make up cumulative prior year
losses.
|
26.
|
Defined
contribution plan
|
Pursuant
to the relevant PRC regulations, the Company is required to make contributions
at a rate of 30.5% to employees’ salaries and wages to a defined contribution
retirement scheme organized by a state-sponsored social insurance plan in
respect of the retirement benefits for the Company’s employees in the
PRC. The only obligation of the Company with respect to retirement
scheme is to make the required contributions under the plan. No
forfeited contribution is available to reduce the contribution payable in the
future years. The defined contribution plan contributions were
charged to the consolidated statements of income. The Company contributed
$734,379 and $498,598 for the year ended December 31, 2009 and 2008,
respectively.
|
27.
|
Segment
information
|
The
Company uses the “management approach” in determining reportable operating
segments. The management approach considers the internal organization
and reporting used by the Company’s chief operating decision maker for making
operating decisions and assessing performance as the source for determining the
Company’s reportable segments. Management, including the chief
operating decision maker, reviews operating results solely by monthly revenue of
wholesales, retail and manufacturing sectors and operating results of the
Company and, as such, the Company has determined that the Company has three
operating segments as defined by ASC 280, “Segments Reporting” (previously SFAS
131)”: wholesales, retail and manufacturing.
|
Wholesale
|
Retail
|
Manufacturing
|
Eliminations
|
Total
|
||||||||||||||||||||||||||||||||||||
|
Year
ended
December
31,
|
Year
ended
December
31,
|
Year
ended
December
31,
|
Year
ended
December
31,
|
Year
ended
December
31,
|
||||||||||||||||||||||||||||||||||||
|
2009
|
2008
|
2009
|
2008
|
2009
|
2008
|
2009
|
2008
|
2009
|
2008
|
|||||||||||||||||||||||||||||||
|
Revenue
from external customers
|
$ | 97,137,585 | $ | 72,806,899 | $ | 31,222,577 | $ | 28,592,852 | $ | 7,726,546 | $ | 7,591,578 | $ | - | $ | - | $ | 136,086,708 | $ | 108,991,329 | ||||||||||||||||||||
|
Revenue
from intersegment
|
21,729,943 | 21,470,183 | - | - | 762,280 | 185,833 | (22,492,223 | ) | (21,656,016 | ) | - | - | ||||||||||||||||||||||||||||
|
Interest
income
|
15,116 | 25,386 | 984 | 3,332 | - | 597 | - | - | 16,100 | 29,315 | ||||||||||||||||||||||||||||||
|
Interest
expenses
|
902,642 | 945,838 | 37,826 | 14,767 | 344,415 | 291,658 | - | - | 1,284,883 | 1,252,263 | ||||||||||||||||||||||||||||||
|
Amortization
|
274,765 | 298,131 | - | - | 157,333 | 236,302 | - | - | 432,098 | 534,433 | ||||||||||||||||||||||||||||||
|
Depreciation
|
180,492 | 225,139 | 3,498 | 3,394 | 414,286 | 404,367 | - | - | 598,276 | 632,900 | ||||||||||||||||||||||||||||||
|
Segment
profit
|
15,187,570 | 13,992,806 | 7,198,703 | 5,938,155 | 3,472,102 | 2,426,031 | - | - | 25,858,375 | 22,356,992 | ||||||||||||||||||||||||||||||
|
Segment
assets
|
68,252,951 | 51,226,243 | 9,184,032 | 1,984,571 | 10,319,034 | 12,586,520 | - | - | 87,756,017 | 65,797,334 | ||||||||||||||||||||||||||||||
|
Expenditure
for segment assets
|
$ | 50,539 | $ | 149,081 | $ | 7,502 | $ | 5,674 | $ | 275,623 | $ | 567,155 | $ | - | $ | - | $ | 333,664 | $ | 721,910 | ||||||||||||||||||||
|
27.
|
Segment
information (cont’d)
|
A
reconciliation is provided for unallocated amounts relating to corporate
operations which is not included in the segment information.
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Total
consolidated revenue
|
$ | 136,086,708 | $ | 108,991,329 | ||||
|
Total
profit for reportable segments
|
$ | 25,858,375 | $ | 22,356,992 | ||||
|
Unallocated
amounts relating to
|
||||||||
|
relating
to operations:
|
||||||||
|
Finance costs
|
(2,791 | ) | (2,952 | ) | ||||
|
Other general
expenses
|
(185,221 | ) | (40,000 | ) | ||||
|
Income
before income taxes
|
$ | 25,670,363 | $ | 22,314,040 | ||||
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Assets
|
||||||||
|
Total
assets for reportable segments
|
$ | 87,756,017 | $ | 65,797,334 | ||||
|
Cash
and cash equivalents
|
5,078,973 | 16,671 | ||||||
|
Other
receivables
|
225,802 | 343 | ||||||
| $ | 93,060,792 | $ | 65,814,348 | |||||
All of
the Company’s long-lived assets and revenues classified based on the customers
are located in the PRC.
|
28.
|
Related
party transactions
|
Apart
from the transactions as disclosed elsewhere in these consolidated financial
statements, the Company has entered into following transactions with its related
parties :-
|
(i)
|
Sales
to related companies of which certain Company's directors have controlling
interests.
|
||||||||
|
Name
of related companies
|
Year
ended December 31,
|
||||||||
|
2009
|
2008
|
||||||||
|
Liucheng
Medicine Company
|
$ | 311,759 | $ | 438,371 | |||||
|
Guangxi
Tianhu Medicine Limited
|
$ | 271,890 | $ | 821,449 | |||||
|
Wuxuan
Baicaotang Medicine Limited
|
$ | 208,475 | $ | 487,781 | |||||
|
Guangxi
Liuzhou Baicaotang Medicine Limited, Guigang Branch
|
$ | 1,273,381 | $ | 1,262,429 | |||||
|
(ii)
|
During
the year of 2008, Baicaotang Medicine declared dividends of $6,940,000 to
the former stockholders of Baicaotang
Medicine.
|
|
29.
|
Subsequent
event
|
The
Company has evaluated its activities subsequent to December 31, 2009 and has
concluded that, except for the transaction described below, no subsequent events
have occurred that would require recognition or disclosure in the consolidated
financial statements.
On
February 1, 2010, the Company completed a private placement of 1,029,970 shares
of common stock and warrants to purchase up to 514,933 shares of common stock at
an exercise price of $3.81 per share for gross proceeds of $2,616,108. In
connection with the private placement, warrants to purchase up to 102,997 shares
of common stock at an exercise price of $3.05 were issued to Co-Placement Agent
on a cash or cashless basis.
In
accordance with the requirements of the Securities Exchange Act of 1934, the
registrant has duly caused this amended report to be signed on its behalf by the
undersigned thereunto duly authorized on March 31, 2010.
|
CHINA
BAICAOTANG MEDICINE LIMITED
|
|
|
/s/ Hui Tian Tang
|
|
|
By:
Hui Tian Tang
|
|
|
Chief
Executive Officer and Director
(principal
executive officer)
|
In
accordance with the requirements of the Securities Exchange Act of 1934, this
Amended Report was signed by the following persons in the capacities and on the
dates indicated.
|
Name
and Title
|
Date
|
|
|
/
s/ Hui Tian Tang
|
March
31, 2010
|
|
|
Hui
Tian Tang
Chief
Executive Officer and Director
(principal
executive officer)
|
|
/s/
Xia Yang Zhang
|
March
31, 2010
|
|
|
Xiao
Yang Zhang
Chief
Financial Officer and Director
(principal
financial officer and accounting officer)
|
EXHIBIT
INDEX
|
2.1
|
Share
Exchange Agreement Dated December 23, 2009 (1)
|
|
3.1
|
Certificate
of Incorporation. (2)
|
|
3.1
|
Certificate
of Amendment of Certificate of Incorporation. (1)
|
|
3.2
|
By-laws.
(2)
|
|
4.1
|
Form
of Warrant. (1)
|
|
4.2
|
Form
of Co-placement Agent Warrant.(3)
|
|
10.1
|
Subscription
Agreement Dated October 23, 2009 (1)
|
|
10.2
|
Co-Placement
Agent Agreement Dated October 21, 2009 (1)
|
|
10.3
|
Escrow
Deposit Agreement Dated October 21, 2009 (1)
|
|
10.4
|
Earn-In
Agreement dated October 22, 2009 (1)
|
|
10.5
|
Share
Transfer Agreement dated as of April 1, 2008 (1)
|
|
10.6
|
Shares
Pledge Agreement dated May 3, 2008 (1)
|
|
10.7
|
Shares
Pledge Agreement dated March 31, 2009 (1)
|
|
10.8
|
Shares
Repurchase Agreement dated July 31, 2008 (1)
|
|
10.9
|
Loan
Agreement and Pledge Agreement dated December 19, 2008 by and among
Liuzhou Baicaotang, Baicaotang Property Development Limited and Liuzhou
City Commercial Bank (1)
|
|
10.10
|
Loan
Agreement and Pledge Agreement dated December 29, 2008 by and among
Liuzhou Bcaicaotang Baicaotang Property Development Limited and Rurol
Credence Cooperation of Guangxi. (1)
|
|
10.11
|
Loan
Agreement and Pledge Agreement dated January 8, 2009 by and among Liuzhou
Bcaicaotang Baicaotang Property Development Limited and Agricultural Bank
of China. (1)
|
|
10.12
|
Form
of Loan Agreement by and between Li Jin Hua and Guangxi Hefeng
Pharmaceutical Co. Limited. (1)
|
|
10.13
|
Form
of Retail Chain Leasing Contract. (1)
|
|
10.14
|
Loan
Agreement dated February 8, 2007 by and between Hui Tian Tang and
Industrial and Commercial Bank of China Guangxi Branch
(3)
|
|
10.15
|
Confirmation
Agreement dated December 31, 2008 by and among Hui Tian Tang, Liuzhou
Baicaotang and Liuzhou Baicaotang Guigang Branch (3)
|
|
10.16
|
Loan
Agreement dated February 12, 2007 by and between Jiang You Ru and
Industrial and Commercial Bank of China Guangxi Branch
(3)
|
|
10.17
|
Confirmation
Agreement dated December 31, 2008 by and among Jiang You Ru, Liuzhou
Baicaotang and Liuzhou Baicaotang Guigang Branch (3)
|
|
10.18
|
Form
of Lock-Up Agreement. (3)
|
|
10.19
|
Employment
Agreement dated January 3rd, 2009 by and between Guangxi Liuzhou
Baicaotang Pharmaceutical Co., Ltd. and Hui Tian Tang.
(1)
|
|
10.20
|
Earn-in
Agreement dated December 30, 2009.(3)
|
|
10.21
|
Shares
Pledge Agreement dated May 3, 2008. (3)
|
|
10.22
|
Shares
Pledge Agreement dated March 31, 2009. (3)
|
|
10.23
|
Termination
Agreement dated March 2, 2010. (3)
|
|
14
|
Code
of Ethics and Business Conduct. (1)
|
|
21
|
List
of Subsidiaries. (3)
|
|
31.1
|
Certification
pursuant to Rule 13a-14(a) and 15d-14(a), as adopted pursuant to Section
302 of the Sarbanes-Oxley Act of 2002. *
|
|
31.2
|
Certification
pursuant to Rule 13a-14(a) and 15d-14(a), as adopted pursuant to Section
302 of the Sarbanes-Oxley Act of 2002. *
|
|
32.1
|
Certification
pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of
the Sarbanes-Oxley Act of 2002. *
|
|
(1)
|
Incorporated
by reference to the Company’s Form 8-K filed on December 31,
2009.
|
|
(2)
|
Incorporated
by reference to the Company’s Form SB-2 filed on August 22,
2007.
|
|
(3)
|
Incorporated
by reference to the Company’s Registration Statement on form S-1 filed on
March 3, 2010.
|
* Filed
herewith.
(1) Incorporated by
reference to the Company's Form 8-K filed on December 31, 2009
(2) Incorporated by
reference to the Company's Form SB-2 filed on August 22, 2007
(3) Incorporated by
reference to the Company's Registration Statement on Form S-1 filed on
March 3, 2010
I-3
