Attached files
| file | filename |
|---|---|
| EX-32.1 - EXHIBIT 32.1 - BIOCRYST PHARMACEUTICALS INC | exh_321.htm |
| EX-31.1 - EXHIBIT 31.1 - BIOCRYST PHARMACEUTICALS INC | exh_311.htm |
| EX-23 - EXHIBIT 23 - BIOCRYST PHARMACEUTICALS INC | exh_23.htm |
| EX-21 - EXHIBIT 21 - BIOCRYST PHARMACEUTICALS INC | exh_21.htm |
| EX-10.83 - EXHIBIT 10.83 - BIOCRYST PHARMACEUTICALS INC | exh_1083.htm |
| EX-10.18 - EXHIBIT 10.18 - BIOCRYST PHARMACEUTICALS INC | exh_1018.htm |
| EX-4.1 - EXHIBIT 4.1 - BIOCRYST PHARMACEUTICALS INC | exh_41.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
________________________
Form 10-K
________________________
| ☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2019
OR
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934. |
For the transition period from to .
Commission File Number 000-23186
________________________
BIOCRYST PHARMACEUTICALS, INC.
(Exact name of registrant as specified in its charter)
________________________
| DELAWARE | 62-1413174 |
|
(State of other jurisdiction of incorporation or organization) |
(I.R.S. employer identification no.) |
4505 Emperor Blvd., Suite 200, Durham, North Carolina 27703
(Address of principal executive offices)
(919) 859-1302
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol(s) | Name of Each Exchange on Which Registered |
| Common Stock, $.01 Par Value | BCRX | The NASDAQ Global Select Market |
Securities registered pursuant to Section 12(g) of the Act:
Title of class
None
________________________
Indicate by a check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No ☐
Indicate by a check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by a check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by a check mark whether the registrant submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (Section 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☒ | |||
| Non-accelerated filer | ☐ | Smaller reporting company | ☐ | |||
| Emerging growth company | ☐ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by a check mark whether the registrant is a shell company (as defined in Exchange Act Rule 12b-2). Yes ☐ No ☒.
The Registrant estimates that the aggregate market value of the Common Stock on June 30, 2019 (based upon the closing price shown on the NASDAQ Global Select Market on June 30, 2019) held by non-affiliates was $413,929,804
The number of shares of Common Stock, par value $0.01, of the Registrant outstanding as of January 31, 2020 was 154,191,951 shares.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the Registrant’s definitive Proxy Statement to be filed in connection with the solicitation of proxies for its 2020 Annual Meeting of Stockholders are incorporated by reference into Items 10, 11, 12, 13 and 14 under Part III hereof.
TABLE OF CONTENTS
| 3 |
| ITEM 1. | BUSINESS |
Forward-Looking Statements
This report includes forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, which are subject to the “safe harbor” created in Section 21E. In particular, statements about our expectations, beliefs, plans, objectives or assumptions of future events or performance are contained or incorporated by reference in this report. All statements other than statements of historical facts contained herein are forward-looking statements. We have based these forward-looking statements on our current expectations about future events. While we believe these expectations are reasonable, forward-looking statements are inherently subject to risks and uncertainties, many of which are beyond our control. Our actual results may differ materially from those suggested by these forward-looking statements for various reasons, including those discussed in this report under the heading “Risk Factors.” Given these risks and uncertainties, you are cautioned not to place undue reliance on our forward-looking statements. The forward-looking statements included in this report are made only as of the date hereof. We do not undertake, and specifically decline, any obligation to update any of these statements or to publicly announce the results of any revisions to any forward-looking statements to reflect future events or developments. When used in the report, unless otherwise indicated, “we,” “our,” “us,” the “Company” and “BioCryst” refer to BioCryst Pharmaceuticals, Inc.
Our Business
We are a biotechnology company that discovers novel, oral, small-molecule medicines. We focus on oral treatments for rare diseases in which significant unmet medical needs exist and an enzyme plays the key role in the biological pathway of the disease. We integrate the disciplines of biology, crystallography, medicinal chemistry and computer modeling to discover and develop small molecule pharmaceuticals through the process known as structure-guided drug design. Structure-guided drug design is a drug discovery approach by which we design synthetic compounds from detailed structural knowledge of the active sites of enzyme targets associated with particular diseases. We use X-ray crystallography, computer modeling of molecular structures and advanced chemistry techniques to focus on the three-dimensional molecular structure and active site characteristics of the enzymes that control cellular biology. Enzymes are proteins that act as catalysts for many vital biological reactions. Our goal generally is to design a compound that will fit in the active site of an enzyme and thereby prevent its catalytic activity. Molecules from our discovery efforts which are commercially available or that are in active development are summarized in the table below:
| Drug/Drug Candidate | Drug Class |
Therapeutic Area(s) |
Phase | Rights | |||||
|
Berotralstat (BCX7353) |
Oral Serine Protease Inhibitor Targeting Kallikrein (once-daily treatment) | Hereditary Angioedema (“HAE”) |
NDA accepted for review
|
BioCryst (worldwide, except Japan)
| |||||
| Oral Serine Protease Inhibitor Targeting Kallikrein (once-daily treatment) | Hereditary Angioedema (“HAE”) | JNDA accepted for review |
Torii Pharmaceutical Co., Ltd. (Japan) | ||||||
| Distinct and different oral dose formulation for acute treatment | Hereditary Angioedema (“HAE”) | Phase 3 | BioCryst (worldwide) | ||||||
| BCX9930 | Oral Factor D Inhibitor | Complement-mediated diseases | Phase 1 | BioCryst (worldwide) | |||||
|
BCX9250
|
Oral Activin Receptor-Like Kinase-2 Inhibitors | Fibrodysplasia Ossificans Progressiva (“FOP”) | Phase 1 | BioCryst (worldwide) | |||||
|
RAPIVAB® (peramivir injection) |
Intravenous Neuraminidase Inhibitor | Acute uncomplicated Influenza |
Approved (U.S., Australia & Canada)
|
Seqirus UK Limited (worldwide, except Japan, Taiwan, Korea and Israel)* BioCryst retains full U.S. Government stockpiling rights | |||||
| 4 |
|
ALPIVABTM (peramivir injection) |
Intravenous Neuraminidase Inhibitor | Acute uncomplicated Influenza | Approved (European Union) | Seqirus UK Limited (worldwide, except Japan, Taiwan, Korea and Israel)* | |||||
|
RAPIACTA® (peramivir injection)
|
Intravenous Neuraminidase Inhibitor | Uncomplicated seasonal influenza |
Approved (Japan & Taiwan) |
Shionogi & Co., Ltd. (Japan & Taiwan)
| |||||
|
PERAMIFLU® (peramivir injection) |
Intravenous Neuraminidase Inhibitor | Uncomplicated seasonal influenza |
Approved (Korea) |
Green Cross Corporation (Korea) | |||||
| Galidesivir (BCX4430) | RNA dependent-RNA Polymerase Inhibitor | Broad spectrum antiviral for 20 RNA viruses, including Marburg, Yellow Fever, and Ebola | Phase 1 |
BioCryst (worldwide) | |||||
|
Mundesine® (forodesine) |
Oral Purine Nucleoside Phosphorylase Inhibitor | Oncology - PTCL | Approved (Japan) |
Mundipharma International Corporation Limited (worldwide) | |||||
* See “Business—Collaborations and In-License Relationships—Seqirus UK Limited”
Business Strategy
Our business strategy is to create shareholder value by focusing our discovery and development efforts on oral drugs for rare diseases for which a significant unmet medical need exists. Our strategy also includes efficiently commercializing these drugs in the United States and certain other regions upon regulatory approval. By focusing on rare disease markets, we believe that we will be able to more effectively control the costs of, and our strategic allocation of financial resources toward, post-approval commercialization.
We select disease targets and product candidates in which a small molecule would offer a significant benefit over existing products or would be the first to market. We strive to advance our product candidate portfolio from discovery to commercial markets efficiently by utilizing a small group of talented and highly-skilled employees working in conjunction with strategic outsource partners. BioCryst is unique in its approach to treat orphan diseases with orally-administered, small molecules, identified by utilizing crystallography and structure-guided drug design. The principal elements of our strategy are:
| • | Focusing on High Value-Added Structure-Guided Drug Design Technologies. We utilize structure-guided drug design in order to most efficiently develop new therapeutic candidates. Structure-guided drug design is a process by which we design a product candidate through detailed analysis of the enzyme target, which the product candidate must inhibit in order to stop the progression of the disease or disorder. We believe that structure-guided drug design is a powerful tool for the efficient development of small-molecule product candidates that have the potential to be safe and effective. Our structure-guided drug design technologies typically allow us to design and synthesize multiple product candidates that inhibit the same enzyme target, with the goal of establishing broad intellectual property protection and formulating compounds with competitive advantages. |
| • | Selecting Inhibitors that are Promising Product Candidates. We start by selecting disease targets with well-understood biology and characteristics that fit with our ability to utilize structure-guided drug design capabilities to build potent and specific enzyme inhibitors. Next, we narrow our selection of these product candidates based on product characteristics, such as initial indications of safety and biologic activity on the target. |
| 5 |
| • | Developing our Product Candidates Efficiently. An important element of our business strategy is to efficiently progress our product candidates through the development process. In order to accomplish this, we typically strive for disease targets with a defined clinical and regulatory pathway for approval. In addition, we control fixed costs and overhead by outsourcing with strategic partners and contractors or entering into license agreements with third parties, including the U.S. Government. By contracting with the U.S. Government and outsourcing certain aspects of our operations, we are able to control overhead costs and focus financial resources directly where they provide the most benefit and reduce our business risk. |
| • | Commercializing our Product Candidates Globally. A core part of our strategy is to commercialize our rare disease products globally. We are building the structure and expertise to commercialize our products in markets where we believe we can do this efficiently and effectively, such as the United States and Europe. We also will seek licensing or distribution partners in certain markets where we determine this to be the more effective approach. |
We are a Delaware corporation originally founded in 1986. Our corporate headquarters is located at 4505 Emperor Blvd., Suite 200, Durham, North Carolina 27703 and the corporate telephone number is (919) 859-1302. For more information about us, please visit our website at www.biocryst.com. The information on our website is not incorporated into this Form 10-K.
Hereditary Angioedema (“HAE”) Drug Candidate
HAE is a rare, severely debilitating and potentially fatal genetic condition with a prevalence of between 1 in 33,000 to 1 in 67,000 people. HAE symptoms include recurrent episodes of edema in various locations, including the hands, feet, face, genitalia and airway. Airway swelling is particularly dangerous and can lead to death by asphyxiation. In addition, patients often have bouts of severe abdominal pain, nausea and vomiting caused by swelling in the intestinal wall. By inhibiting plasma kallikrein, our HAE drug candidates suppress bradykinin production. Bradykinin is the mediator of acute swelling attacks in HAE patients.
Berotralstat (BCX7353): Berotralstat is a second generation HAE compound and our most advanced molecule that is being developed as a once-daily oral therapy for the prevention of HAE attacks. We successfully completed our pivotal Phase 3 clinical trial, APeX-2, and reported 48-week data from our ongoing long-term safety clinical trial, APeX-S, in 2019. Based on the data from our clinical program, including APeX-2 and APeX-S, we submitted a New Drug Application (“NDA”) to the Food and Drug Administration (“FDA”) in December 2019 for approval of oral, once daily berotralstat for the prevention of HAE attacks. In February 2020, the FDA notified us that they had accepted and filed our NDA for review and that our Prescription Drug User Fee Act (“PDUFA”) date for the NDA is December 3, 2020.
In addition, we have completed APeX-J, a clinical trial of berotralstat for the prevention of HAE attacks designed to support Japanese marketing authorization in conjunction with our other berotralstat clinical trials. On February 3, 2020, we announced we had submitted a new drug application (“JNDA”) to the Japanese Pharmaceuticals and Medical Devices Agency (“PDMA”) for approval of oral, once daily berotralstat for the prevention of HAE attacks. We expect approval of berotralstat in Japan in the second half of 2020.
The FDA has granted Fast Track Designation to berotralstat for the prevention of angioedema attacks in patients with HAE, and the United Kingdom’s Medicines and Healthcare products Regulatory Agency has granted a Promising Innovative Medicine designation to berotralstat. We have also received orphan drug status from the FDA, European Medicines Agency (“EMA”), and the PDMA for berotralstat. In addition, in 2015, berotralstat was designated under Japan’s Ministry of Health Labor & Welfare (“MHLW”) Sakigake fast track review system, which provides for additional interactions with the MHLW from early development through filing, prioritized development and review, and introduction of the product as soon as possible to address a serious unmet medical need.
APeX-2 Phase 3 Trial: APeX-2 is a Phase 3 double-blinded, placebo-controlled, three-arm clinical trial evaluating two dose levels of berotralstat administered orally once-daily as a preventive treatment to reduce the frequency of attacks in patients with HAE. APeX-2 tested once-daily berotralstat at 110 mg and 150 mg for prevention of angioedema attacks. The trial enrolled patients with Type I and II HAE in the United States, Canada and Europe. The primary efficacy endpoint of APeX-2 is the rate of angioedema attacks over 24 weeks of study drug administration. The trial enrolled and randomized 121 patients. The APeX-2 trial has been amended to extend the duration of dosing to monitor the long-term safety of the trial. Patients may continue in the trial on open-label berotralstat.
On May 21, 2019, we announced our Phase 3 APeX-2 trial of oral, once-daily berotralstat for the prevention of HAE attacks achieved its primary endpoint for both dose levels (110 mg and 150 mg), with the 150 mg dose reducing the attack rate in HAE patients by 44 percent (p<0.001) compared to placebo. Fifty percent of patients receiving 150 mg berotralstat in APeX-2 had a ≥ 70 percent reduction in their HAE attack rate compared to baseline, compared to 15 percent of placebo patients (p=0.002). Of 108 patients who completed 24 weeks of study drug treatment, 100 percent continued into the ongoing 48-week extension phase of the trial. Both the 110 mg and 150 mg dose levels of oral, once-daily berotralstat were generally safe and well-tolerated. No drug-related serious adverse events were reported.
APeX-S Long-term Safety Trial: APeX-S is an open label, long-term safety trial evaluating two dose levels (110 mg and 150 mg) of berotralstat administered orally once-daily as a preventive treatment in patients with Type I and II HAE. The APeX-S trial has been amended to extend the duration of dosing through 96 weeks to monitor the safety and effectiveness of long term treatment with berotralstat.
On November 6, 2019, we announced 48-week data from APeX-S showing that APeX-S patients taking 150 mg of berotralstat had similar attack control as those in APeX-2. Patients completing 48 weeks of treatment on 150 mg of berotralstat (n=73) had a median attack rate of zero attacks per month in six of the 12 months, including month 12 (week 48). An integrated 48-week analysis across both APeX-2 and APeX-S showed no new safety findings. Berotralstat was safe and generally well tolerated in a total of 342 patients with a total of 232 patient-years of daily oral dosing. The most common adverse event was the common cold, which occurred with similar frequency in berotralstat and placebo patients. Gastrointestinal events led to discontinuation of berotralstat in three percent of patients. Drug-related serious adverse events occurred in three of 342 subjects (0.9%) and resolved after stopping or interrupting BCX7353 dosing. In APeX-S, alanine aminotransferase levels >3xULN were seen in 14 of 49 patients who discontinued androgens within 28 days prior to study entry, compared to one of 104 patients who discontinued androgens more than 28 days prior to study entry and zero of 74 patients who had never used androgens.
| 6 |
APeX-J Trial: On January 12, 2020, we reported data from our APeX-J trial in Japan, designed to support potential Japanese approval of berotralstat for the prevention of HAE attacks. APeX-J met its primary endpoint (p=0.003) for prevention of HAE attacks, and berotralstat was safe and generally well-tolerated.
ZENITH-1 Phase 2 Trial: We have also been evaluating berotralstat, in a distinct oral formulation, for the treatment of acute HAE attacks. In 2019, we completed ZENITH-1, an adaptive dose-ranging proof-of-concept Phase 2 clinical trial evaluating efficacy, safety and tolerability for the oral treatment of acute HAE attacks. We expect the market for products for the treatment of acute HAE attacks to be less significant than the HAE prophylaxis market and have prioritized our development strategies accordingly.
ZENITH-1 is a clinical trial studying three dose levels of a liquid formulation of berotralstat given as a single oral dose for the acute treatment of angioedema attacks in patients with HAE. ZENITH-1 is a randomized, double-blind, placebo-controlled, adaptive dose-ranging trial of the efficacy, safety and tolerability of berotralstat for treatment of acute angioedema attacks, and enrolled subjects with Type I and II HAE. Blinded study drug was dosed as an oral liquid after onset of symptoms, for up to three attacks in each subject, with each subject receiving both berotralstat (for two attacks) and placebo (for one attack) in a randomized sequence. The trial was structured for three consecutive cohorts testing single doses of 750 mg, 500 mg and 250 mg.
On February 23, 2019, we reported topline data from the completed Phase 2 ZENITH-1 trial. Data from the complete trial confirmed previously-reported results showing a single dose of oral 750 mg berotralstat was well-tolerated and superior to placebo (p<0.05) against the majority of efficacy endpoints evaluated in HAE patients suffering an acute attack, and demonstrated a clear dose response across the three dose levels evaluated, 250 mg, 500 mg and 750 mg. With the 750 mg dose, compared to placebo, improvements in symptoms and Visual Analog Scale (“VAS”) scores were demonstrated as early as one hour after oral berotralstat dosing (the first timepoint evaluated), and were sustained through 24 hours. Through 24 hours, standard of care (“SOC”) medication use was reduced by 31.6% after berotralstat compared with placebo (p=0.0029), and no or mild symptoms were reported in 64.1% of attacks treated with berotralstat compared with 32.3% of attacks treated with placebo (p=0.0038).
Berotralstat was generally safe and well-tolerated with no notable differentiation from the adverse event profile of placebo. The most commonly reported adverse events were diarrhea, abdominal pain, nausea, headache and nasopharyngitis. There were three discontinuations in the trial: one following a berotralstat 750 mg dose due to a transient, localized rash; one following a berotralstat 500 mg dose due to grade 2 vomiting and nausea and one following a placebo dose due to abdominal pain. With the exception of an unrelated ankle fracture, there were no grade 3 or 4 adverse events, and no grade 3 or 4 laboratory abnormalities.
Complement-Mediated Diseases
The complement system is part of the body’s natural immune system and is responsible for helping the body eliminate microbes (including viral and bacterial infections) and damaged cells. It is comprised of proteins which are primarily produced in the liver and circulate in the blood. Once activated, the complement system stimulates inflammation, phagocytosis and cell lysis. Excessive or uncontrolled activation of the complement system can cause severe, and potentially fatal, immune and inflammatory disorders. The complement system comprises biological cascades of amplifying enzyme cleavages involving more than 30 proteins and protein fragments, and may be activated through three pathways: the classical pathway (initiated by antibody-antigen complexes), the lectin pathway (initiated by lectin binding) and the alternative pathway (initiated by microbial surfaces). The alternative pathway also provides a critical amplification loop for all three pathways, regardless of the initiating mechanism. Factor D is an essential enzyme in the alternative pathway, thus making Factor D an attractive target to address complement-mediated diseases. Several rare diseases are known to be mediated by dysregulation of the complement system.
On June 27, 2019, we announced that we began enrollment of a Phase 1 trial of BCX9930, an oral Factor D inhibitor discovered and developed by us, for the treatment of complement-mediated diseases. The objectives of the trial are to evaluate the safety and tolerability of single and multiple ascending doses of BCX9930 in healthy subjects and to characterize the pharmacokinetic (“PK”) and pharmacodynamic (“PD”) profiles of BCX9930 in single and multiple ascending doses of BCX9930 in healthy subjects (parts 1 and 2). In part three of the trial, there is an additional objective to demonstrate proof of concept in paroxysmal nocturnal hemoglobinuria (“PNH”) patients by evaluating key biomarkers of effectiveness in PNH patients taking BCX9930. On October 28, 2019, we announced results from the ongoing Phase 1 trial of BCX9930 in 72 healthy volunteers. BCX9930 was safe and generally well tolerated, and showed rapid, sustained and >95% suppression of the alternative pathway (“AP”) of the complement system at 100 mg every 12 hours, as measured by the AP Wieslab® assay. In part 1 of the trial, a single ascending dose (“SAD”) assessment, six cohorts of healthy volunteers received a single dose of 10 mg, 30 mg, 100 mg, 300 mg, 600 mg or 1200 mg of oral BCX9930 or placebo (each SAD cohort randomized 6:2). In part 2 of the trial, the multiple ascending dose (“MAD”) assessment, two cohorts of healthy volunteers received 50 mg or 100 mg of oral BCX9930 or placebo (each MAD cohort randomized 10:2) administered every 12 hours for seven days. Healthy volunteers in the MAD cohorts were prophylactically dosed with the broad-spectrum antibiotic, amoxicillin/clavulanate to lower the risk of meningococcal infection during suppression of the complement system. BCX9930 was safe and generally well tolerated at all doses studied. There were no serious adverse events. A clinically benign rash was observed in some healthy volunteers in the MAD assessment (two in the 50 mg cohort, seven in the 100 mg cohort), which was self-limited and resolved in 4-8 days after onset. There were no discontinuations from the trial.
| 7 |
We have completed an additional MAD cohort with 50 mg of oral BCX9930 or placebo administered every 12 hours for 14 days, with vaccination instead of an antibiotic. In the additional MAD cohort, a benign rash (similar to prior MAD cohorts) that was self-limited and resolved in 4 to 8 days post-onset was seen in seven healthy volunteers; the protocol allowed two of these healthy volunteers with more limited surface area affected by the rash to continue receiving BCX9930. Both of these healthy volunteers successfully dosed-through benign rash, with rash resolving on-drug, in both patients; biopsies of rashes from multiple subjects confirm the benign nature of the rash. The protocol for part three of the trial in PNH patients allows any patient who develops a clinically benign rash to continue dosing with BCX9930.
Based on the safety, tolerability, PK and PD dose-response results from parts 1 and 2 of the Phase 1 trial, we plan to complete additional MAD dosing cohorts and advance to part 3 of the trial, a proof of concept (“PoC”) study of BCX9930 in PNH patients who are poor responders to eculizumab or ravulizumab, and treatment-naïve patients. We have also successfully dosed MAD cohorts of 200 milligrams twice a day and 400 milligrams twice a day. On March 5, 2020 we announced that we had dosed the first PNH patients in part three of the trial. These patients were naïve to eculizumab and ravulizumab. We expect to report data from the PoC study in PNH patients in the second quarter of 2020.
Fibrodysplasia Ossificans Progressiva (“FOP”)
FOP is an ultra-rare disease that affects approximately 1 in 2 million people worldwide. FOP is a rare, severely disabling condition characterized by the irregular formation of bone outside the normal skeleton, also known as heterotopic ossification (“HO”). It occurs in approximately 1 in 2 million people worldwide. HO can occur in muscles, tendons and soft tissue. FOP patients progressively become bound by this irregular ossification, with restricted movement and fused joints, resulting in deformities and premature mortality. In patients with FOP, minor trauma can result in rapid development of painful inflammatory masses. These progress over several weeks resulting in the replacement of the affected soft tissue by permanent bone masses. There is no cure for this condition, and there are no approved treatments for FOP.
In 2018, we announced the advancement of a program exploring activin receptor-like kinase-2 (“ALK2”) inhibitors for treatment of FOP. ALK2 enzyme is a part of the normal signaling pathway for bone formation and responds to binding its specific ligands (bone morphogenic proteins, or BMPs), by stimulating normal bone growth and renewal in healthy children and adults. Specific activating mutations of the ALK2 gene are seen in all cases of FOP. An activating mutation in ALK2 is necessary for the disease to occur, making the ALK2 kinase an ideal drug target for treatment of FOP with an ALK2 kinase inhibitor
The goal of our ALK2 inhibitor program is to discover and develop orally administered kinase inhibitor drug candidates that are able to slow or prevent HO. Our lead compound, BCX9250, reduced HO in an experimental model of ALK2-driven HO in laboratory rats, with up to 89 percent reduction in volume of HO compared to controls.
On November 1, 2019, we announced that we had begun a Phase 1 clinical trial with oral BCX9250 for the treatment of FOP. The Phase 1 trial will evaluate single and multiple ascending doses of oral BCX9250 in healthy volunteers. We expect to report the results from the trial in the second half of 2020.
Peramivir injection (RAPIVAB®, ALPIVABTM, RAPIACTA®, PERAMIFLU®)
Peramivir was approved in Japan and Korea in 2010, the United States in 2014, in Taiwan in 2016, in Canada in 2017, and in the European Union (“EU”) and Australia in 2018. A Supplemental New Drug Application (“sNDA”) was approved in the United States in September 2017, extending its availability for the treatment of acute uncomplicated influenza to pediatric patients two years and older. In the United States, peramivir is indicated for the treatment of acute uncomplicated influenza in patients who have been symptomatic for no more than two days. In May 2018, peramivir, with the brand name of ALPIVAB, received approval from the European Medicines Agency (“EMA”), although ALPIVAB is not currently commercially available in the EU.
On June 17, 2015, we announced that we licensed RAPIVAB (peramivir injection) for the treatment of influenza to CSL Limited (“CSL”), a global biopharmaceutical company. Under this license agreement (the “SUL Agreement”), RAPIVAB and ALPIVAB were licensed to and expected to be commercialized by CSL's subsidiary, Seqirus UK Limited (“SUL”), which specializes in influenza prevention through the supply of seasonal and pandemic vaccine to global markets. SUL was to manufacture, commercialize and exercise decision-making authority with respect to the development and commercialization of RAPIVAB and ALPIVAB within the Territory (as defined in the SUL Agreement) and be responsible for all related costs, including sales and promotion.
| 8 |
Under the terms of the SUL Agreement, we were responsible for fulfilling all post-marketing approval commitments in connection with the FDA's approval of the new drug application (“NDA”), and upon fulfillment would transfer ownership of and financial responsibility for the NDA to SUL.
On March 4, 2020, the International Court of Arbitration of the International Chamber of Commerce delivered a Partial Arbitration Award in the arbitration matter between us and SUL with respect to the SUL Agreement. See “Business—Collaborations and In-License Relationships—Seqirus UK Limited.”
In September 2018, the U.S. Department of Health and Human Services awarded us a $34.7 million contract for the procurement of up to 50,000 doses of RAPIVAB over a five-year period to supply the Strategic National Stockpile for use in a public health emergency. We delivered two shipments under this contract in 2019 for a total price of approximately $13.9 million, and we expect to deliver at least one shipment within the award in 2020, totaling approximately $6.9 million.
RAPIVAB was developed under a $234.8 million contract from the Biomedical Advanced Research and Development Authority within the United States Department of Health and Human Services (“BARDA/HHS”). See “Collaborations and In-License Relationships” below for a further discussion of this development contract.
In January 2010, our partner Shionogi & Co., Ltd. (“Shionogi”) received the first approval for peramivir injection and launched it in Japan under the commercial name RAPIACTA. It is approved for the treatment of adults, children and infants with uncomplicated seasonal influenza and those patients at high-risk for complications associated with influenza. In August 2010, Green Cross Corporation (“Green Cross”) received marketing and manufacturing approval from the Korean Food & Drug Administration under the commercial name PERAMIFLU to treat patients with influenza A & B viruses, including pandemic H1N1 and avian influenza. See “Collaborations and In-License Relationships” below for a further discussion of these outlicense arrangements.
Galidesivir (BCX4430)
Galidesivir is a broad-spectrum antiviral (“BSAV”) that has been shown to be active against more than 20 RNA viruses in nine different families, including filoviruses, togaviruses, bunyaviruses, arenaviruses, paramyxoviruses, coronaviruses and flaviviruses. In animal studies, galidesivir has demonstrated survival benefits against a variety of serious pathogens, including Marburg, Yellow Fever, Ebola, and Zika viruses and from exposures to aerosolized Marburg virus, an experimental condition designed to mimic an exposure scenario that could result during a bioterrorist attack. Galidesivir research program and is currently being developed under contracts with the National Institute of Allergy and Infectious Diseases (“NIAID/HHS”) and the U.S. Department of Health and Human Services (“BARDA/HHS”).
The objective of our BSAV program is to develop galidesivir as a broad-spectrum therapeutic for viruses that pose a threat to national health and security. The primary focus of the program is treatment of hemorrhagic fever viruses. NIAID/HHS funding has supported galidesivir’s development as a treatment for Marburg virus, Yellow Fever and Ebola virus. Since September 2013, NIAID/HHS has supported the development of galidesivir as a therapeutic for Ebola and Marburg viruses. As of the date hereof, all options under this contract have been awarded and the total NIAID/HHS contract amount is $43.0 million. Since March 2015, BARDA/HHS has supported the development of galidesivir as a potential treatment for filoviruses. The total BARDA/HHS contract value to advance the program through toxicology studies and manufacturing work to support a new drug application is $39.1 million if all contract options are exercised. As of the date hereof, a total of $20.6 million has been awarded under exercised options within this contract.
On May 9, 2019, we announced the completion of a randomized, placebo-controlled Phase 1 clinical trial to evaluate intravenous (IV) galidesivir in healthy volunteers. In the trial, galidesivir was generally safe and well tolerated. This placebo-controlled trial evaluated the safety, tolerability and pharmacokinetics of escalating doses of galidesivir in four single-dose cohorts of 5mg/kg, 10 mg/kg, 15 mg/kg and 20 mg/kg, with a total of 24 volunteers receiving galidesivir by IV infusion. Drug exposures (Cmax and AUC) at the highest dose were 20,500 ng/mL and 44,600 hr.ng/mL, similar to or greater than drug exposures needed in nonclinical galidesivir treatment experiments in Marburg virus disease and Yellow Fever. The clinical pharmacokinetics of galidesivir IV were similar to that of galidesivir administered by intramuscular (IM) injection in a previous single ascending dose and multiple ascending dose phase 1 study in healthy subjects. In that trial, galidesivir was generally safe and well tolerated at doses up to 10 mg/kg/day for 7 days.
We are in the process of initiating an exploratory Phase 1b clinical trial evaluating galidesivir in Yellow Fever patients in Brazil.
We are in active dialogue with NIAID, relevant U.S. public health authorities, and clinical investigators as they assess potential approaches to evaluate investigational antiviral drugs for treatment of COVID-19, with the goal of determining if galidesivir is effective against this strain, assessing whether galidesivir should be tested in new or existing clinical trials in patients with COVID-19, and expanding the current supply of the drug.
Mundesine (forodesine)
Mundesine is a Purine Nucleoside Phosphorylase (“PNP”) inhibitor developed by Mundipharma as a treatment for cancer under a world-wide license agreement. PNP is a purine salvage pathway enzyme. High doses of PNP inhibitors could be useful in the treatment of hematological malignancies. Mundipharma has received orphan drug status for Mundesine, and, following its successful completion of a Phase 2 pivotal study in recurrent/refractory peripheral T-cell lymphoma (“PTCL”) patients in Japan, Mundesine was approved in April 2017 by the MHLW in Japan. We are currently entitled to receive royalties on Mundesine.
| 9 |
Collaborations and In-License Relationships
Torii Pharmaceutical Co., Ltd. (“Torii”)
On November 5, 2019, we announced we had entered into the Torii Agreement, granting Torii the exclusive right to commercialize berotralstat for the prevention of HAE attacks in Japan.
Under the Torii Agreement, we received an upfront, non-refundable payment of $22.0 million and may be eligible to receive an additional milestone payment of either $20.0 million if the PMDA grants regulatory approval on or before December 31, 2020, or $15.0 million if regulatory approval is granted on or before December 31, 2021. In either case, the regulatory milestone payment is contingent upon receipt of a reimbursement price approval from Japan’s National Health Insurance system in excess of the threshold specified in the Torii Agreement.
In addition, we will be entitled under the Torii Agreement to receive tiered royalty payments based on the amount of annual net sales of berotralstat in Japan during each calendar year. If berotralstat maintains its Sakigake designation during the PMDA review, the tiered royalty rate will range from 20% to 40% of net sales, otherwise, the tiered royalty rate will range from 15% to 35% of net sales. Torii’s royalty payment obligations are subject to customary reductions in certain circumstances, but may not be reduced by more than 50% of the amount that otherwise would have been payable to us in the applicable calendar quarter. Torii’s royalty payment obligations commence upon the first commercial sale of berotralstat in Japan and expire upon the later of (i) the tenth anniversary of the date of first commercial sale of berotralstat in Japan, (ii) the expiration of our patents covering berotralstat, and (iii) the expiration of regulatory exclusivity for berotralstat in Japan. We will be responsible for supplying Torii with its required amounts of berotralstat. The activities of the parties pursuant to the Torii Agreement will be overseen by a Joint Steering Committee, to be composed of an equal number of representatives from each party to coordinate the development and commercialization of berotralstat in Japan.
Under the Torii Agreement, we have granted Torii a right of first negotiation (“ROFN”) to commercialize berotralstat in Japan for the acute treatment of HAE attacks if we develop berotralstat for such indication and to commercialize any additional kallikrein inhibitor that we may develop in the future for use in HAE in Japan. Under both ROFNs, if the parties do not agree to terms with respect to a definitive amendment to the Torii Agreement or new agreement, as applicable, the terms of the amendment or agreement would be set by a third party arbitrator.
U.S. Department of Health and Human Services
On September 6, 2018, we announced that the U.S. Department of Health and Human Services (“HHS”) had awarded us a $34.7 million contract for the procurement of up to 50,000 doses of RAPIVAB® (peramivir injection) over a five-year period, including an initial base order of 10,000 doses. On September 26, 2019, we announced that HHS had exercised its option to purchase an additional 10,000 doses of RAPIVAB. We delivered a total of 20,000 doses of RAPIVAB and recorded approximately $13.9 million of product sales in the fourth quarter of 2019. The RAPIVAB purchase by the HHS will supply the Strategic National Stockpile, the nation’s largest supply of life-saving pharmaceuticals and medical supplies for use in a public health emergency.
On March 31, 2015, we announced that BARDA/HHS awarded us a contract for the continued development of galidesivir as a potential treatment for diseases caused by RNA pathogens, including filoviruses. This BARDA/HHS contract has a potential value of $39.1 million if all contract options are exercised. As of the date hereof, a total of $20.6 million has been awarded under exercised options within this contract. In September 2013, NIAID/HHS contracted with us for the development of galidesivir as a treatment for Marburg, and subsequently, Yellow Fever and Ebola virus disease. All options under this contract have been awarded and the total contract value is $43.0 million. The contracts with BARDA/HHS and NIAID/HHS are cost-plus-fixed-fee contracts. That is, we are entitled to receive reimbursement for all costs incurred in accordance with the contracts provisions that are related to the development of peramivir and galidesivir plus a fixed fee, or profit. BARDA/HHS and NIAID/HHS will make periodic assessments of progress and the continuation of the contract is based on our performance, the timeliness and quality of deliverables, and other factors. The government has rights under certain contract clauses to terminate these contracts. These contracts are also terminable by the government at any time for breach or without cause.
Seqirus UK Limited
On June 16, 2015, we and SUL, a limited company organized under the laws of the United Kingdom and a subsidiary of CSL, a company organized under the laws of Australia, entered into the SUL Agreement granting SUL and its affiliates worldwide rights to develop, manufacture and commercialize peramivir for the treatment of influenza except for the rights to conduct such activities in Israel, Japan, Korea and Taiwan (the permitted geographies together constituting the “Territory”). We retain all rights and associated economics to procure pandemic stockpiling orders for RAPIVAB from the U.S. Government, while SUL has the right to pursue government stockpiling outside the U.S. Pursuant to the SUL Agreement, peramivir is being commercialized by CSL’s subsidiary, SUL, which specializes in influenza prevention through the supply of seasonal and pandemic vaccine to global markets. SUL is responsible for the manufacture, commercialization and decision-making authority with respect to the development and commercialization of peramivir within the Territory and is responsible for all related costs, including sales and promotion. We exercise sole decision-making authority with regard to the development and commercialization of peramivir outside of the Territory and are responsible for all associated costs.
| 10 |
Under the terms of the SUL Agreement, we are responsible for fulfilling all post-marketing approval commitments in connection with the FDA’s approval of the NDA, and upon fulfillment will transfer ownership of and financial responsibility for the NDA to SUL. Pursuant to rights to sell ALPIVAB in the EU, we were responsible for regulatory filings and interactions with the European Medicines Agency. In accordance with the SUL Agreement, we and SUL formed a joint steering committee, composed of an equal number of representatives from each party, to oversee, review and coordinate the conduct and progress of the commercialization of peramivir in the Territory and any additional development.
Under the terms of the SUL Agreement, we received an upfront payment of $33.7 million, and have achieved all development milestones under the contract totaling $12.0 million. We are also entitled under the SUL Agreement to receive tiered royalties at a percentage rate beginning in the mid-teens contingent upon meeting minimum thresholds of net sales, as well as a low-thirties percentage of the gross profit from government stockpiling purchases made outside the U.S. Specifically, we receive tiered royalties at a percentage rate in the mid-teens to low-forties on net sales in the U.S. during a Contract Year (defined as July 1 - June 30) and tiered royalties at a percentage rate in the mid-teens to mid-twenties on net sales in the Territory, other than in the U.S., during a Calendar Year, each subject to certain downward adjustments for circumstance or events impacting the overall market opportunity. SUL’s royalty payment obligations commence on the date of the SUL Agreement and expire, on a country-by-country basis, upon the later of (i) the expiration of legal exclusivity in such country and (ii) ten years from the date of the SUL Agreement (the “Royalty Term”). We developed RAPIVAB under a license from UAB and will owe sublicense payments to them on any future milestone payments and/or royalties received by us from SUL.
The term of the SUL Agreement shall continue on a country-by-country basis until the expiration of the last-to-expire Royalty Term in any such country in the Territory. Either party may terminate the SUL Agreement in its entirety if the other party breaches a payment obligation, otherwise materially breaches the SUL Agreement, subject to applicable cure periods, or if the other party suffers an insolvency event. We may also terminate the SUL Agreement if SUL or any of its affiliates seek to challenge the validity of our patents. Termination does not affect a party’s rights which have accrued prior thereto, but there are no stated payments in connection with termination other than payments of obligations previously accrued. For all terminations exercised by us, the SUL Agreement provides for the termination of any sublicenses granted by SUL to third parties, and in the case of termination by us for cause, the ceasing of SUL’s activities with respect to RAPIVAB, the discontinued use of all of our intellectual property and the termination of licenses and rights previously granted to SUL. If requested by us, SUL shall also promptly sell to us all licensed product it then holds in stock, otherwise, SUL may continue to sell such licensed product for designated periods.
On March 4, 2020, the International Court of Arbitration of the International Chamber of Commerce (“ICC Tribunal”) delivered a Partial Arbitration Award (the “Partial Arbitration Award”) in the arbitration matter between us and SUL with respect to the SUL Agreement. In the Partial Arbitration Award, the ICC Tribunal found that, during the term, SUL materially breached and abandoned its core duties to us under the Diligent Efforts (as defined in the SUL Agreement) requirements of the SUL Agreement as applicable in the U.S. The ICC Tribunal granted a declaratory judgment in favor of us terminating the SUL Agreement and restoring all rights to peramivir to us as of March 17, 2020 (or such other date as the parties agree). The ICC Tribunal also awarded us attorneys’ fees and expenses incurred in securing the declaratory judgment as well as the costs incurred by us in the arbitration. Finally, the ICC Tribunal found that SUL breached the SUL Agreement by failing to pay the milestone payment due to us within 30 days of the approval of peramivir for adult use in the European Union and awarded us $5.0 million (plus interest) for this claim. The ICC Tribunal retained jurisdiction for further proceedings relating to the award of attorneys’ fees and for any dispute relating to the return to us of all rights to peramivir in the Territory.
Shionogi & Co., Ltd. (“Shionogi”). On February 28, 2007, we entered into a License, Development and Commercialization Agreement (as amended, supplemented or otherwise modified, the “Shionogi Agreement”), an exclusive license agreement with Shionogi to develop and commercialize peramivir in Japan for the treatment of seasonal and potentially life-threatening human influenza. In October 2008, we and Shionogi amended the Shionogi Agreement to expand the territory covered by the agreement to include Taiwan. Under the terms of the Shionogi Agreement, Shionogi obtained rights to injectable formulations of peramivir in Japan in exchange for a $14.0 million upfront payment. The license provided for development milestone payments (up to $21.0 million), which have all been paid, and for commercial milestone payments (up to $95.0 million) in addition to double-digit (between 10% and 20%) royalty payments on product sales of peramivir.
In December 2017, we, on behalf of Royalty Sub (defined below), instituted arbitration proceedings against Shionogi in order to resolve a dispute with Shionogi under the Shionogi Agreement regarding the achievement of sales milestones and escalating royalties. The arbitration proceedings have concluded, with the decision that no sale milestones have been achieved and that the royalties will remain the same. The costs associated with the arbitration proceedings are recoverable from the assets of Royalty Sub in accordance with the terms of the indenture and servicing agreement relating to the PhaRMA Notes.
Generally, all payments under the Shionogi Agreement are non-refundable and non-creditable, but they are subject to audit. Shionogi is responsible for all development, regulatory, and marketing costs in Japan. The term of the Shionogi Agreement is from February 28, 2007 until terminated. Either party may terminate in the event of an uncured breach. Shionogi has the right of termination without cause. In the event of termination, all license and rights granted to Shionogi shall terminate and shall revert back to us. We developed peramivir under a license from the University of Alabama Birmingham (“UAB”) and have paid sublicense payments to UAB on the upfront payments and will owe sublicense payments on any future event payments and/or royalties received by us from Shionogi.
| 11 |
Shionogi Royalty Monetization and Non-Recourse Notes Payable. On March 9, 2011, we completed a $30.0 million financing transaction to monetize certain future royalty and milestone payments under the Shionogi Agreement, pursuant to which JPR Royalty Sub LLC (“Royalty Sub”) a wholly-owned subsidiary of BioCryst, issued the PhaRMA Notes discussed below. We received net proceeds of $22.7 million from this transaction.
As part of the transaction, we entered into a purchase and sale agreement dated as of March 9, 2011 with Royalty Sub, whereby we transferred to Royalty Sub, among other things, (i) our rights to receive commercial royalty and milestone payments from Shionogi arising under the Shionogi Agreement, and (ii) the right to receive payments under a Japanese yen/US dollar foreign currency hedge arrangement (as further described below, the “Currency Hedge Agreement”) put into place by us in connection with the transaction. Royalty payments are paid by Shionogi in Japanese yen and milestone payments are paid in U.S. dollars. Our collaboration with Shionogi was not impacted by this transaction.
On March 9, 2011, Royalty Sub completed a private placement to institutional investors of $30.0 million in aggregate principal amount of its PhaRMA Senior Secured 14.0% Notes due 2020 (the “PhaRMA Notes”). The PhaRMA Notes were issued by Royalty Sub under an Indenture, dated as of March 9, 2011 (the “Indenture”), by and between Royalty Sub and U.S. Bank National Association, as Trustee. Principal and interest on the PhaRMA Notes issued are payable from, and are secured by, the rights to royalty and milestone payments under the Shionogi Agreement transferred by us to Royalty Sub and payments, if any, made to Royalty Sub under the Currency Hedge Agreement. The PhaRMA Notes bear interest at 14.0% per annum, payable annually in arrears on September 1st of each year (the “Payment Date”). We remain entitled to receive any royalties and milestone payments related to sales of peramivir by Shionogi following repayment by Royalty Sub of the PhaRMA Notes.
Royalty Sub’s obligations to pay principal and interest on the PhaRMA Notes are obligations solely of Royalty Sub and are without recourse to any other person, including the Company, except to the extent of our pledge of our equity interests in Royalty Sub in support of the PhaRMA Notes. We may, but are not obligated to, make capital contributions to a capital account that may be used to redeem, or on up to one occasion pay any interest shortfall on, the PhaRMA Notes.
In September 2014, Royalty Sub was unable to pay the full amount of interest payable in September 2013 by the next succeeding Payment Date for the PhaRMA Notes, which was September 1, 2014. This inability constituted an event of default under the terms of the Indenture. Accordingly, we have classified the PhaRMA Notes and related accrued interest as current liabilities on our balance sheet since that time. As of December 31, 2019, the PhaRMA Notes remain in default.
As a result of the continuing event of default under the PhaRMA Notes, the holders of the PhaRMA Notes may pursue acceleration of the PhaRMA Notes, may foreclose on the collateral securing the PhaRMA Notes and the equity interest in Royalty Sub and exercise other remedies available to them under the Indenture in respect of the PhaRMA Notes. In such event, we may not realize the benefit of future royalty payments that might otherwise accrue to us following repayment of the PhaRMA Notes and we might otherwise be adversely affected. Due to the non-recourse nature of the PhaRMA Notes, in the event of any potential acceleration or foreclosure, we believe the primary impact to us would be the loss of future royalty payments from Shionogi and legal costs associated with retiring the PhaRMA Notes. In addition, we may incur costs associated with liquidating the related Currency Hedge Agreement, which would no longer be required in the event of foreclosure or if the PhaRMA Notes cease to be outstanding.
The PhaRMA Notes have a final legal maturity date of December 1, 2020, at which time the outstanding principal amount of the PhaRMA Notes, together with all accrued and unpaid interest, will be due in full. The failure by Royalty Sub to repay in full the outstanding principal amount of the PhaRMA Notes, together with any accrued and unpaid interest, at the December 1, 2020 final maturity date would constitute an additional event of default under the PhaRMA Notes. We do not currently expect that Royalty Sub will be able to repay the PhaRMA Notes at final maturity. We cannot predict whether holders of PhaRMA Notes will seek to pursue any remedies as a result of the continuing event of default with respect to the PhaRMA Notes or if Royalty Sub fails to pay the PhaRMA Notes in full at final maturity. The PhaRMA Notes are the obligation of Royalty Sub. As a result, we do not currently expect the continuing event of default on the PhaRMA Notes, or a failure by Royalty Sub to repay the PhaRMA Notes at final maturity, to have a significant impact on our future results of operations or cash flows. However, there can be no assurance that this will be the case or that we will not otherwise be adversely affected as a result the continuing event of default under the PhaRMA Notes or a failure by Royalty Sub to repay the PhaRMA Notes at maturity.
The Indenture does not contain any financial covenants. The Indenture includes customary representations and warranties of Royalty Sub, affirmative and negative covenants of Royalty Sub, Events of Default and related remedies, and provisions regarding the duties of the Trustee, indemnification of the Trustee, and other matters typical for indentures used in structured financings of this type. The PhaRMA Notes are redeemable at the option of Royalty Sub at any time at a redemption price equal to 100% of the outstanding principal balance of the PhaRMA Notes being redeemed, plus accrued and unpaid interest through the redemption date on the PhaRMA Notes being redeemed.
Foreign Currency Hedge. In connection with the issuance by Royalty Sub of the PhaRMA Notes, we entered into a Currency Hedge Agreement to hedge certain risks associated with changes in the value of the Japanese yen relative to the U.S. dollar. Under the Currency Hedge Agreement, we have the right to purchase dollars and sell yen at a rate of 100 yen per dollar for which we may be required to pay a premium in 2020. A payment of $2.0 million will be required if, on May 18, 2020, the U.S. dollar is worth 100 yen or less, as determined in accordance with the Currency Hedge Agreement. The final tranche of the options under the Currency Hedge Agreement will expire in November of 2020.
| 12 |
The Currency Hedge Agreement does not qualify for hedge accounting treatment; therefore, mark-to-market adjustments are recognized in our Consolidated Statements of Comprehensive Loss. Cumulative mark-to-market adjustments resulted in losses of $0.4 million, $1.0 million and $1.8 million for the twelve months ended December 30, 2019, 2018, and 2017, respectively. In addition, realized currency exchange gains of $0.9 million, $1.0 million and $1.0 million were recognized in 2019, 2018 and 2017, respectively, related to the exercise of the U.S. dollar/Japanese yen currency option under the Currency Hedge Agreement. We are also required to post collateral in connection with the mark-to-market adjustments based on defined thresholds. As of December 31, 2019, no collateral was posted under the Currency Hedge Agreement. We will not be required at any time to post collateral exceeding the maximum premium payments remaining payable under the Currency Hedge Agreement. The maximum amount of hedge collateral we would be required to post is $2.0 million. We are required to maintain a foreign currency hedge at 100 yen per dollar under the agreements governing the PhaRMA Notes.
Green Cross Corporation (“Green Cross”). In June 2006, we entered into an agreement with Green Cross to develop and commercialize peramivir in Korea. Under the terms of the agreement, Green Cross is responsible for all development, regulatory, and commercialization costs in Korea. We received a one-time license fee of $250,000. The license provides that we will share in profits resulting from the sale of peramivir in Korea, including the sale of peramivir to the Korean government for stockpiling purposes. Furthermore, Green Cross will pay us a premium over its cost to supply peramivir for development and any future marketing of peramivir products in Korea. Both parties have the right to terminate in the event of an uncured material breach. In the event of termination, all rights, data, materials, products and other information would be transferred to us.
In August 2010, we announced that Green Cross had received marketing and manufacturing approval from the Korean Food & Drug Administration for i.v. peramivir, under the commercial name PERAMIFLU ®. PERAMIFLU is intended to treat patients with influenza A & B viruses, including pandemic H1N1 and avian influenza. Green Cross received the indication of single dose administration of 300 mg i.v. peramivir.
Mundipharma. We are party to an exclusive, royalty bearing right and license agreement with Mundipharma for the development and commercialization of Mundesine for use in oncology. The agreement, as amended and restated, provides for the possibility of future event payments totaling $15.0 million for achieving specified regulatory events for certain indications and provides that we will receive tiered royalties ranging from mid- to high-single digit percentages of net product sales in each country where Mundesine is sold by Mundipharma. We licensed forodesine and other PNP inhibitors from AECOM/IRL and will owe sublicense payments to AECOM/IRL on all milestone payments and royalties received by us from Mundipharma.
Albert Einstein College of Medicine of Yeshiva University and Industrial Research, Ltd. (“AECOM” and “IRL” respectively). In June 2000, we licensed a series of potent inhibitors of PNP from AECOM and IRL (collectively, the “Licensors”). The lead product candidate from this collaboration is forodesine. We have obtained worldwide exclusive rights to develop and ultimately distribute it, or any other, product candidates that might arise from research on these inhibitors. We have the option to expand our license agreement with the Licensors to include other inventions in the field made by the investigators or employees of the Licensors. Under this agreement, as amended and restated, we have agreed to use commercially reasonable efforts to develop these drugs and to pay certain milestone payments for each licensed product (which range in the aggregate from $1.4 million to almost $4.0 million per indication) for future development, single digit royalties on net sales of any resulting product made by us, and to share a portion of future payments received from other third-party partners, if any. In addition, we have agreed to pay annual license fees, which can range from $150,000 to $500,000, that are creditable against actual royalties and other payments due to the Licensors. The Licensors have also granted us an exclusive worldwide license of galidesivir for any antiviral use.
The University of Alabama at Birmingham (“UAB”). We currently have agreements with UAB for influenza neuraminidase and complement inhibitors. Under the terms of these agreements, UAB performed specific research for us in return for research payments and license fees. UAB has granted us certain rights to any discoveries in these areas resulting from research developed by UAB or jointly developed with us. We have agreed to pay single digit royalties on sales of any resulting product and to share in future payments received from other third-party partners. We have completed the research under the UAB agreements. These two agreements have initial 25-year terms, are automatically renewable for five-year terms throughout the life of the last patent and are terminable by us upon three months’ notice and by UAB under certain circumstances. Upon termination both parties shall cease using the other parties’ proprietary and confidential information and materials, the parties shall jointly own joint inventions and UAB shall resume full ownership of all UAB licensed products. There is currently no activity between us and UAB on these agreements, but when we license this technology, such as in the case of the Shionogi and Green Cross agreements, or commercialize products related to these programs, we will owe sublicense fees or royalties on amounts it receives.
| 13 |
Government Contracts
NIAID/HHS. In September 2013, NIAID/HHS contracted with us for the development of galidesivir as a treatment for Marburg, and subsequently, Yellow Fever and Ebola virus. All options under this contract have been awarded and the total contract value is $43.0 million. The goals of this contract, including amendments, are to file IND applications for i.v. and i.m. galidesivir for the treatment of Marburg virus disease and other hemorrhagic fever viruses, to study galidesivir as a treatment for Yellow Fever and Ebola virus disease and to conduct a Phase 1 human clinical trial.
BARDA/HHS. In March 2015, BARDA/HHS awarded us a contract for the continued development of galidesivir as a potential treatment for diseases caused by RNA pathogens, including filoviruses. This BARDA/HHS contract has a potential value of $39.1 million if all contract options are exercised. As of the date hereof, a total of $20.6 million has been awarded under exercised options within this contract.
The contracts with NIAID/HHS and BARDA/HHS are cost-plus-fixed-fee contracts. That is, we are entitled to receive reimbursement for all costs incurred in accordance with the contracts provisions that are related to the development of peramivir and galidesivir plus a fixed fee, or profit. NIAID/HHS and BARDA/HHS will make periodic assessments of progress and the continuation of the contract is based on our performance, the timeliness and quality of deliverables, and other factors. The government has rights under certain contract clauses to terminate these contracts. These contracts are also terminable by the government at any time for breach or without cause.
HHS Procurement Contract. In September 2018, the U.S. Department of Health and Human Services awarded us a $34.7 million contract for the procurement of up to 50,000 doses of RAPIVAB over a five-year period to supply the Strategic National Stockpile for use in a public health emergency. We delivered two shipments under this contract in 2019 for a total price of approximately $13.9 million. This contract contains a number of terms and conditions that are customary for government contracts of this nature, including provisions giving the government the right to terminate the contract at the government’s discretion.
Patents and Proprietary Information
Our success will depend in part on our ability to obtain and enforce patent protection for our products, methods, processes and other proprietary technologies, preserve our trade secrets, and operate without infringing on the proprietary rights of other parties, both in the United States and in other countries. We own or have rights to certain proprietary information, proprietary technology, issued and allowed patents and patent applications which relate to compounds we are developing. We actively seek, when appropriate, protection for our products, proprietary technology and proprietary information by means of U.S. and foreign patents, trademarks and contractual arrangements. In addition, we rely upon trade secrets and contractual arrangements to protect certain of our proprietary information, proprietary technology and products.
The patent positions of companies like ours are generally uncertain and involve complex legal and factual questions. Our ability to maintain and solidify our proprietary position for our technology will depend on our success in obtaining effective patent claims and enforcing those claims once granted. We do not know whether any of our patent applications or those patent applications that we license will result in the issuance of any patents. Our issued patents and those that may issue in the future, or those licensed to us, may be challenged, invalidated, rendered unenforceable or circumvented, which could limit our ability to stop competitors from marketing related products or the length of term of patent protection that we may have for our products. In addition, the rights granted under any issued patents may not provide us with competitive advantages against competitors with similar compounds or technology. Furthermore, our competitors may independently develop similar technologies or duplicate any technology developed by us in a manner that does not infringe our patents or other intellectual property. Because of the extensive time required for development, testing and regulatory review of a potential product, it is possible that, before any of our product candidates or those developed by our partners can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thereby reducing any advantage of the patent.
As of December 31, 2019, we have been issued approximately 20 U.S. patents that expire between 2020 and 2037 and that relate to our kallikrein inhibitor compounds, neuraminidase inhibitor compounds, BSAV compounds and PNP compounds. We have licensed a number of compounds protected by certain composition of matter patents from AECOM and IRL, plus additional manufacturing patents, totaling seven additional U.S. patents that expire between 2020 and 2029. Additionally, we have approximately 21 Patent Cooperation Treaty or U.S. patent applications pending related to kallikrein inhibitor compounds, neuraminidase inhibitor compounds, BSAV compounds, PNP compounds, FOP program compounds, and other complement-mediated disease program compounds. Our pending applications may not result in issued patents, our patents may not cover the products of interest or may not be enforceable in all, or any jurisdictions and our patents may not provide us with sufficient protection against competitive products or otherwise be commercially viable. After expiration of composition of matter patents for our products and product candidates, we may rely on data exclusivity, or in some cases, method of use patents. The enforceability of these patents varies from jurisdiction to jurisdiction and may not be allowed or enforceable in some territories where we may seek approval. We may not have the funds to continue patent prosecution or to defend all of our existing patents in our current patent estate and may selectively abandon patents or patent families worldwide or in certain territories.
| 14 |
Our success is also dependent upon the skills, knowledge and experience of our scientific and technical personnel, none of which is patentable. To help protect our rights, we require all employees, consultants, advisors and partners to enter into confidentiality agreements, which prohibit the disclosure of confidential information to anyone outside of BioCryst and, where possible, require disclosure and assignment to us of their ideas, developments, discoveries and inventions. These agreements may not provide adequate protection for our trade secrets, know-how or other proprietary information in the event of any unauthorized use or disclosure or the lawful development by others of such information.
Competition
The pharmaceutical and biotechnology industries are intensely competitive. Many companies, including biotechnology, chemical and pharmaceutical companies, are actively engaged in activities similar to ours, including research, development, and commercialization of drugs for the treatment of rare medical conditions. Many of these companies have substantially greater financial and other resources, larger research and development staffs, and more extensive commercial and manufacturing organizations than we do. In addition, many have considerable experience in preclinical testing, clinical trials and other regulatory approval procedures. In addition, there are also academic institutions, governmental agencies and other research organizations who conduct research in areas in which we are working. We expect to encounter significant competition for any of the pharmaceutical products we plan to develop. Companies that successfully complete clinical trials, obtain required regulatory approvals and commence commercial marketing and sales of their products may achieve a significant competitive advantage.
HAE: HAE is an autosomal dominant disease characterized by painful, unpredictable, recurrent attacks of inflammation affecting the hands, feet, face, abdomen, urogenital tract, and the larynx. The inflammation can be disfiguring, debilitating, or in the case of laryngeal attacks, life-threatening. Prevalence for HAE is uncertain but is estimated to be approximately 1 case per 33,000 to 67,000 persons without known differences among ethnic groups and is caused by deficient (Type I) or dysfunctional (Type II) levels of C1-Inhibitor (“C1-INH”), a naturally occurring molecule that is known to inhibit kallikrein, bradykinin, and other serine proteases in the blood. If left untreated, HAE can result in a mortality rate as high as 40% primarily due to upper airway obstruction. There are several licensed therapies for HAE, including the following:
| • | C1-INH replacement therapy is available as an acute therapy (Berinert®) and as a prophylactic therapy (Haegarda® and Cinryze®). These therapies are dosed subcutaneously and intravenously. Recombinant C1-INH (Ruconest ®) is also available as an acute therapy. | |
| • | Kallikrein Inhibitors - Kalbitor® (ecallantide) is a specific recombinant plasma kallikrein inhibitor that is dosed subcutaneously by healthcare providers to treat acute HAE attacks. Takhzyro™ (lanadelumab-flyo) is a monoclonal antibody approved for prophylaxis of HAE attacks and can be self-administered as a subcutaneous injection. | |
| • | Bradykinin receptor antagonist - Firazyr® (icatibant) is the treatment of acute attacks and is administered by subcutaneous administration. Two generic forms of icatibant were approved in July 2019 and more may be approved in the future. | |
| • | Other medications - Prophylactic administration of synthetic attenuated androgens (generically available as danazol or stanozolol) has been utilized to reduce the frequency or severity of attacks. However, long-term use of danazol or stanozolol may result in liver damage, virilization and arterial hypertension. Six-month liver function tests, annual lipid profiles, and biennial hepatic ultrasound are recommended for patients on chronic androgen therapy. |
In addition to berotralstat, we are aware of a number of HAE therapies in clinical development, which include:
| Company | Asset | Mechanism of Action | Route of Administration | Trial Phase | Role in Therapy |
| KalVista | KVD-900 | Kallikrein inhibitor | Oral | II | Acute treatment |
| KVD-824 | Kallikrein inhibitor | Oral | I | Prophylaxis | |
| Pharvaris | PHA121 | B2 bradykinin antagonist | Oral | I | Prophylaxis |
| Attune | ATN-249 | Kallikrein inhibitor | Oral | I | Prophylaxis |
| CSL | CSL312 | Anti-factor XII mAb | IV/Subcutaneous | II | Prophylaxis |
| Ionis | IONIS-PKK-LRx | Antisense inhibitor of prekallikrein | Subcutaneous | II | Prophylaxis |
Complement-mediated diseases: Several rare diseases are known to be mediated by defects of the complement system, including paroxysmal nocturnal hemoglobinuria (“PNH”), atypical hemolytic uremic syndrome (“aHUS”), complement 3 glomerulopathy (“C3G”), and myasthenia gravis. Alexion Pharmaceuticals, Inc.’s Soliris® (eculizumab) is a C5 inhibitor approved for PNH, aHUS, myasthenia gravis, and neuromyelitis optica spectrum disorder. Soliris had global sales of over $3.9 billion in 2019. Alexion also recently received FDA approval for Ultomiris™ (ravulizumab), a longer-acting C5 inhibitor, as a treatment for PNH in late 2018 and aHUS in late 2019. Global sales for Ultomiris were $339 million in 2019. In addition, Alexion acquired Achillion, a developer of oral Factor D inhibitors, in early 2020.
| 15 |
In addition to BCX9930, we are aware of a number of complement pathway-based products in development, which include:
| Company | Asset | Mechanism of Action | Route of Administration | Trial Phase |
| Apellis | Pegcetacoplan (APL-2) | C3 Inhibitor | Subcutaneous | III |
| Akari | Nomacopan | C5 Inhibitor | Subcutaneous | III |
| Regeneron | Pozelimab | C5 Inhibitor | IV / Subcutaneous | III |
| Omeros | Narsoplimab | MASP-2 | IV / Subcutaneous | III |
| Alexion | Danicopan (ACH-4471) | Factor D Inhibitor | Oral | II |
| ACH-5228 | Factor D Inhibitor | Oral | I | |
| Novartis | LNP023 | Factor B Inhibitor | Oral | II |
| Tesidolumab | C5 Inhibitor | IV | II | |
| ChemoCentryx | Avacopan | C5aR Inhibitor | Oral | II |
| Ra / UCB | Zilucoplan | C5 Inhibitor | Subcutaneous | II |
| Alnylam | Cemdisiran | C5 Inhibitor | Subcutaneous | II |
| Chugai | SKY59/RG6107 | C5 Inhibitor | IV | I |
Amgen (Phase 3), Samsung, and Isuabxis are also in clinical trials developing biosimilars of eculizumab.
FOP: FOP is a rare, severely disabling condition characterized by the irregular formation of bone outside the normal skeleton, also known as heterotopic ossification (“HO”). HO can occur in muscles, tendons and soft tissue. FOP patients progressively become bound by this irregular ossification, with restricted movement and fused joints, resulting in deformities and premature mortality. There are currently no approved treatments for FOP.
In addition to BCX9250, we are aware of a number of FOP therapies in clinical development, which include:
| Company | Asset | Mechanism of Action | Route of Administration | Trial Phase |
| Ipsen | Palovarotene | Retinoic Acid Receptor (RAR) Gamma Agonist | Oral | III |
| BLU-782 | ALK-2 inhibitor | Oral | I | |
| Regeneron | Garetosmab | Anti-activin A | Intravenous | II |
| Incyte | INCB00928 | ALK-2 inhibitor | - | I |
Antivirals: The pharmaceutical market for products that prevent or treat influenza is very competitive. Key competitive factors for RAPIVAB (peramivir injection) include, among others, efficacy, ease of use, safety, price and cost-effectiveness, storage and handling requirements and reimbursement. A number of products are currently available in the U.S. and/or other counties, including Japan, for the prevention or treatment of influenza, including seasonal flu vaccines, F. Hoffmann-La Roche Ltd.’s (“Roche”) TAMIFLU® (oseltamivir), generic oseltamivir, GlaxoSmithKline plc’s (“GSK”) RELENZA®, Genentech and Shiongi’s XOFLUZA™ and Daiichi Sankyo Co., Ltd.’s INAVIR®. In addition, FUJIFILM Corporation’s favipiravir, a polymerase inhibitor, is approved in Japan.
Various government entities throughout the world are offering incentives, grants and contracts to encourage additional investment into preventative and therapeutic agents against influenza, which may have the effect of further increasing the number of our competitors and/or providing advantages to certain competitors.
Galidesivir is a product candidate in our BSAV research program and is currently being developed under contracts with NIAID/HHS and BARDA/HHS. The objective of our BSAV program is to develop galidesivir as a broad-spectrum therapeutic for viruses that pose a threat to national health and security. The U.S. Government is investing in a number of programs intended to address gaps in its medical countermeasure plan. Currently, there are five investigational therapeutics under a compassionate use/expanded access framework that can be available in an outbreak setting to treat Ebola virus disease.
We are in active dialogue with NIAID, relevant U.S. public health authorities, and clinical investigators as they assess potential approaches to evaluate investigational antiviral drugs for treatment of COVID-19, with the goal of determining if galidesivir is effective against this strain, assessing whether galidesivir should be tested in new or existing clinical trials in patients with COVID-19, and expanding the current supply of the drug.
In order to compete successfully in these and other therapeutic areas, we must develop proprietary positions in patented drugs for therapeutic markets that have not been satisfactorily addressed by conventional research strategies and, in the process, expand our expertise in structure-based drug design. Our product candidates, even if successfully tested and developed, may not be adopted by physicians over other products and may not offer economically feasible alternatives to other therapies.
| 16 |
Research and Development
We initiated our research and development activities in 1986. We have assembled a scientific research staff with expertise in a broad base of advanced research technologies including protein biochemistry, X-ray crystallography, chemistry and pharmacology. Our research facilities, located in Birmingham, Alabama, include protein biochemistry and organic synthesis laboratories, testing facilities, X-ray crystallography, computer and graphics equipment and facilities to make product candidates on a small scale for early stage clinical trials.
Compliance
We conduct our business in an ethical, fair, honest and lawful manner. We act responsibly, respectfully and with integrity in our relationships with patients, health care professionals, collaborators, governments, regulatory entities, stockholders, suppliers and vendors.
In order to ensure compliance with applicable laws and regulations, our Chief Financial Officer (or in the absence of the Chief Financial Officer, the Principal Accounting Officer), Chief Legal Officer and Vice President of Human Resources oversee compliance training, education, auditing and monitoring; enforce disciplinary guidelines for any infractions of our corporate polices; implement new policies and procedures; respond to any detected issues; and undertake corrective action procedures. Our controls address compliance with laws and regulations that govern public pharmaceutical companies including, but not limited to, the following: federal and state law, such as the Sarbanes-Oxley Act of 2002 and the U.S. Foreign Corrupt Practices Act of 1977; NASDAQ listing requirements; the regulations of the Financial Industry Regulatory Authority, the Securities and Exchange Commission (“SEC”), the FDA, and the United States Department of Health and Human Services; and applicable laws and regulations administered by foreign regulatory authorities, including those of the European Union, the U.K., and Japan. Our standard operating procedures are designed to provide a framework for corporate governance in accordance with ethical standards and best legal practices.
Government Regulation
The FDA regulates the pharmaceutical and biotechnology industries in the U.S., and our product candidates are subject to extensive and rigorous domestic government regulations prior to commercialization. The FDA regulates, among other things, the development, testing, manufacture, safety, efficacy, record-keeping, labeling, storage, approval, advertising, promotion, sale and distribution of pharmaceutical products. In foreign countries, our products are also subject to extensive regulation by foreign governments. These government regulations will be a significant factor in the production and marketing of any pharmaceutical products that we develop. Failure to comply with applicable FDA and other regulatory requirements at any stage during the regulatory process may subject us to sanctions, including:
| • | delays; | ||
| • | warning letters; | ||
| • | fines; | ||
| • | product recalls or seizures; | ||
| • | injunctions; | ||
| • | penalties; | ||
| • | refusal of the FDA to review pending market approval applications or supplements to approval applications; | ||
| • | total or partial suspension of production; | ||
| • | civil penalties; | ||
| • | withdrawals of previously approved marketing applications; and | ||
| • | criminal prosecutions. |
The regulatory review and approval process is lengthy, expensive and uncertain. Before obtaining regulatory approvals for the commercial sale of any products, we or our partners must demonstrate that our product candidates are safe and effective for use in humans. The approval process takes many years, substantial expenses may be incurred and significant time may be devoted to clinical development.
| 17 |
The policies of the FDA and foreign regulatory authorities may change and additional government regulations may be enacted which could prevent or delay regulatory approval of our product candidates or approval of new indications for our existing products. We cannot predict the likelihood, nature or extent of adverse governmental regulation that might arise from future legislative or administrative action, either in the United States or abroad.
FDA Regulation
Before testing potential product candidates in humans, we carry out laboratory and animal studies to determine safety and biological activity. After completing preclinical trials, we must file an IND, including a proposal to begin clinical trials, with the FDA. Thirty days after filing an IND, a Phase 1 human clinical trial can start, unless the FDA places a hold on the trial.
Clinical trials to support a NDA are typically conducted in three sequential phases, but the phases may overlap.
Phase 1—During Phase 1, the initial introduction of the drug into healthy volunteers, the drug is tested to assess metabolism, pharmacokinetics and pharmacological actions and safety, including side effects associated with increasing doses.
Phase 2—Phase 2 usually involves trials in a limited patient population to: (1) assess the efficacy of the drug in specific, targeted indications; (2) assess dosage tolerance and optimal dosage; and (3) identify possible adverse effects and safety risks.
Phase 3 (pivotal)—If a compound is found to be potentially effective and to have an acceptable safety profile in Phase 2 evaluations, Phase 3 clinical trials, also called pivotal studies, major studies or advanced clinical trials, are undertaken to further demonstrate clinical efficacy and to further test for safety within an expanded patient population at geographically dispersed clinical trial sites. In general, the FDA requires that at least two adequate and well-controlled Phase 3 clinical trials be conducted.
Initiation and completion of the clinical trial phases are dependent on several factors including things that are beyond our control. For example, the clinical trials cannot begin at a particular site until that site receives approval from its Institutional Review Board (“IRB”), which reviews the protocol and related documents. This process can take from several weeks to several months. In addition, clinical trials are dependent on patient enrollment, but the rate at which patients enroll in the study depends on:
| • | willingness of investigators to participate in a study; | ||
| • | ability of clinical sites to obtain approval from their IRB; | ||
| • | the availability of the required number of eligible subjects to be enrolled in a given trial; | ||
| • | the availability of existing or other experimental drugs for the disease we intend to treat; | ||
| • | the willingness of patients to participate; and | ||
| • | the patients meeting the eligibility criteria. |
Delays in planned patient enrollment may result in increased expense and longer development timelines.
After successful completion of the required clinical testing, generally an NDA is submitted. Upon receipt of the NDA, the FDA will review the application for completeness. Within 60 days, the FDA will determine if the application is sufficiently complete to warrant full review and will consider the application “filed” at that time. Also upon receipt of the application, the FDA will assign a review priority to the application. Priority review applications are usually reviewed within 6 months; standard review applications are usually reviewed within 10 months. The FDA may refer NDAs for new molecular entities to an appropriate advisory committee for review and evaluation in regards to providing a recommendation as to whether the application should be approved. The FDA is not bound to follow the recommendation of an advisory committee.
Following the review of the application, which may include requests for additional information from the sponsor and results from inspections of manufacturing and clinical sites, the FDA will issue an “action letter” on the application. The action letter will either be an “approval letter,” in which case the product may be lawfully marketed in the United States, or a “complete response letter.” A complete response letter will state that the FDA cannot approve the NDA in its present form and, usually, will describe all of the specific deficiencies that the FDA has identified in the application. The complete response letter, when possible, will include the FDA’s recommended actions to place the application in a condition for approval. Deficiencies can be minor (e.g., labeling changes) or major (e.g., requiring additional clinical trials). A complete response letter may also be issued before the FDA conducts the required facility inspection and/or reviews labeling, leaving the possibility that additional deficiencies in the original NDA could be subsequently cited. An applicant receiving a complete response letter is permitted to resubmit the NDA addressing the identified deficiencies (in which case a new two or six-month review cycle will begin), or withdraw the NDA. The FDA may consider a failure to take action within one year of a complete response letter to be a request to withdraw, unless the applicant has requested an extension of time in which to resubmit. If the FDA approves an NDA, the marketing of the product will be limited to the particular disease states and conditions of use that are described in the product label.
| 18 |
We and all of our contract manufacturers are also required to comply with the applicable FDA current Good Manufacturing Practice, or cGMP, regulations during clinical development and to ensure that the product can be consistently manufactured to meet the specifications submitted in an NDA. The cGMP regulations include requirements relating to product quality as well as the corresponding maintenance of records and documentation. Manufacturing facilities must be approved by the FDA before they can be used to manufacture our products. Based on an inspection, the FDA determines whether manufacturing facilities are in compliance with applicable regulations. Manufacturing facilities in non-United States countries that are utilized to manufacture drugs for distribution into the United States are also subject to inspection by the FDA. Additionally, failure to comply with local regulatory requirements could affect production and availability of product in relevant markets.
Foreign Regulation
In addition to regulations in the United States, we are subject to a variety of foreign regulatory requirements governing human clinical trials and marketing approval for drugs. The foreign regulatory approval process includes all of the risks associated with FDA approval set forth above, as well as additional country-specific regulations. Whether or not we obtain FDA approval for a product, we must obtain approval of a product by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval. The requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from country to country.
For example, under European Union regulatory systems, we may submit marketing authorizations either under a centralized or decentralized procedure. The centralized procedure provides for the grant of a single marketing authorization that is valid for all European Union member states. The decentralized procedure provides for mutual recognition of national approval decisions, and the holder of a national marketing authorization may submit an application to the remaining member states. The United Kingdom’s exit from the EU, or Brexit, has caused political and economic uncertainty, including in the regulatory framework applicable to our operations and product candidates, and this uncertainty may persist for years.
Under the Japanese regulatory system administered by the Pharmaceuticals and Medical Devices Agency (“PMDA”), pre-marketing approval and clinical studies are required for all pharmaceutical products. To obtain manufacturing/ marketing approval, we must submit an application for approval to the MHLW with results of nonclinical and clinical studies to show the quality, efficacy and safety of a new drug. A data compliance review, good Clinical Practices, or GCP, on-site inspection, cGMP audit and detailed data review are undertaken by the PMDA. The application is then discussed by the committees of the Pharmaceutical Affairs and Food Sanitation Council (“PAFSC”). Based on the results of these reviews, the final decision on approval is made by Ministry of Health, Labour and Welfare (“MHLW”). In Japan, the National Health Insurance system maintains a Drug Price List specifying which pharmaceutical products are eligible for reimbursement, and the MHLW sets the prices of the products on this list. After the approval, negotiations regarding the reimbursement price with MHLW will begin. The price will be determined within 60 to 90 days unless the applicant disagrees, which may result in extended pricing negotiations. The government generally introduces price cut rounds every other year and also mandates price decreases for specific products. New products judged innovative or useful, that are indicated for pediatric use, or that target orphan or small population diseases, however, may be eligible for a pricing premium. The Japanese government has also promoted the use of generics, where available.
Human Resources
As of January 31, 2020, we had approximately 140 employees, of whom approximately 80 were engaged in the research and development function of our operations. Our research and development staff, approximately 35 of whom hold Ph.D. or M.D. degrees, have diversified experience in biochemistry, pharmacology, X-ray crystallography, synthetic organic chemistry, computational chemistry, medicinal chemistry, clinical development and regulatory affairs.
Our employees are not represented by any collective bargaining agreements, and we have never experienced a work stoppage. Employees are required to execute confidentiality and assignment of intellectual property agreements. We consider our relations with our employees to be satisfactory.
Available Information
Our website address is www.biocryst.com. We make available, free of charge, at our website our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. We also make available at our website copies of our audit committee charter, compensation committee charter, corporate governance and nominating committee charter and our code of business conduct, which applies to all our employees as well as the members of our Board of Directors. Any amendment to, or waiver from, our code of business conduct will be posted on our website.
| 19 |
Financial Information
For information related to our revenues, profits, net loss and total assets, in addition to other financial information, please refer to the Financial Statements and Notes to Financial Statements contained in this Annual Report. Financial information about revenues derived from foreign countries is included in Note 1 to the Financial Statements contained in this Annual Report.
| ITEM 1A. | RISK FACTORS |
An investment in our stock involves risks. You should carefully read this entire report and consider the following uncertainties and risks, which may adversely affect our business, financial condition or results of operations, along with all of the other information included in our other filings with the Securities and Exchange Commission, before deciding to buy our common stock.
Risks Relating to Our Business
We have incurred losses since our inception, expect to continue to incur such losses, and may never be profitable.
Since our inception, we have not achieved sustained profitability. We expect to incur additional losses for the foreseeable future, and our losses could increase as our research and development efforts and commercial activities progress. We expect that such losses will fluctuate from quarter to quarter and losses and fluctuations may be substantial. To become profitable, we, or our collaborative partners, must successfully manufacture and develop product candidates, receive regulatory approval, and successfully commercialize and/or enter into profitable commercialization arrangements with other parties. It could be several years, if ever, before we receive significant revenue from any current or future license agreements or revenues directly from product sales.
Because of the numerous risks and uncertainties associated with developing our product candidates and their potential for commercialization, we are unable to predict the extent of any future losses. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. If we are unable to achieve and sustain profitability, the market value of our common stock will likely decline.
Our success depends upon our ability to advance our products through the various stages of development, especially through the clinical trial process, and to receive regulatory approval for the commercial sale of our products.
To receive the regulatory approvals necessary for the commercial sale of our product candidates, we or our partners must demonstrate through preclinical studies and clinical trials that each product candidate is safe and effective. The development process and related regulatory process are complex and uncertain. The preclinical and clinical development of our product candidates is susceptible to the risk of failure inherent at any stage of drug development, including failure to demonstrate efficacy and safety, the occurrence of adverse events that are severe or medically or commercially unacceptable, our or our partners’ failure to comply with trial protocols, applicable regulatory requirements, and industry standards, or a determination by the FDA or any comparable foreign regulatory authority that a product candidate may not continue development or be approved in accordance with our development plans or at all. We cannot guarantee that any preclinical studies and clinical trials will be conducted as planned or completed on schedule, if at all, or that the results of such trials will be sufficient to support regulatory approval for our product candidates.
20
Progression of our product candidates through the clinical development process is dependent upon our trials indicating that our product candidates have adequate safety and efficacy in the patients being treated by achieving pre-determined safety and efficacy endpoints according to the clinical trial protocols. Failure to achieve any of these endpoints in any of our programs, including berotralstat, BCX9930, BCX9250, galidesivir, and our other rare disease product candidates, could result in delays in or modifications to our trials or require the performance of additional unplanned trials. If any of our product candidates is associated with adverse events or undesirable side effects or has properties that are unexpected, we may need to abandon development or limit development of that product candidate to certain uses or subpopulations in which the undesirable side effects or other characteristics are less prevalent, less severe or more acceptable from a risk-benefit perspective. Product candidates that initially show promise in clinical or preclinical testing could later be found to cause undesirable or unexpected side effects that could result in delays in the development of our product candidates, significant unexpected costs, or the termination of programs. The development plans for our product candidates, including our clinical trials, may not be adequately designed or executed, which could negatively affect the outcome and analysis of study results. Because of the cost and duration of clinical trials, we may decide to discontinue development of product candidates that are unlikely to show favorable results in clinical trials, unlikely to help advance a product to the point of a meaningful collaboration, or unlikely to have reasonable commercial potential.
Undesirable or inconclusive data in our pre-clinical studies and clinical trials or side effects in humans could result in the U.S. Food and Drug Administration (the “FDA”) or foreign regulatory authorities (including, e.g., the European Medicines Agency (“EMA”), the Japanese Ministry of Health, Labor & Welfare (“MHLW”) or the U.K. Medicines and Healthcare products Regulatory Agency (“MHRA”) refusing to approve a product candidate for any targeted indications or imposing restrictions or warnings that could impact development or the ultimate commercial viability of a product candidate. In addition, the FDA or foreign regulatory authorities may determine that study data from our product candidates necessitates additional studies or study designs which differ from our planned development strategy, and such regulatory authorities may also require patient monitoring and testing or may implement restrictions or other conditions on our development activities, any of which could materially impact the cost and timing of our planned development strategy. We, our partners, the FDA, or foreign regulatory authorities may suspend or terminate clinical trials at any time if we or they believe the trial participants face unacceptable health risks.
Our ability to successfully complete the clinical development process is dependent upon many factors, including but not limited to:
| • | our or our partners’ ability to secure suitable clinical sites and investigators and to enroll and maintain an adequate number of patients on a timely basis or at all; | |
| • | patients that enroll in a clinical trial may not comply with the clinical trial protocol or maintain contact with investigators to provide complete data during and after treatment; | |
| • | our product candidates may not prove to be either safe or effective or may produce unfavorable or inconclusive results; | |
| • | we or our partners may decide, or be required by regulatory authorities, to suspend or terminate clinical research for various reasons, including a finding that the participants are being exposed to unacceptable health risks, undesirable side effects or other unexpected characteristics of the product candidate, noncompliance with regulatory requirements or their standards of conduct, or findings of undesirable effects caused by a chemically or mechanistically similar product or product candidate; | |
| • | regulatory authorities may disagree with our or our partners’ clinical trial protocols or our or their interpretation of data from preclinical studies and clinical trials; | |
| • | clinical protocols or study procedures may not be adequately designed or followed by the investigators; | |
| • | formulation improvements may not work as expected, which could negatively impact commercial demand for our product candidates; | |
| • | regulatory authorities may fail to approve or subsequently find fault with the manufacturing processes or facilities of third party manufactures with which we or our partners enter into agreements for clinical and commercial supplies; | |
| • | the supply or quantity of raw materials or manufactured product candidates or other materials necessary to conduct development activities may be insufficient, inadequate, or unavailable at an acceptable cost, and we or our partners may experience interruptions in supply; | |
| • | our or our partners’ development plans may be delayed or changed as a result of changes in development strategy, the impact of new or different regulations, requirements, and guidelines, or other unexpected events or conditions; | |
| • | the cost of pre-clinical studies and clinical trials may be greater than we anticipate; and | |
| • | third-party contractors, including those manufacturing our product candidates or components or ingredients thereof, or conducting clinical trials or laboratory testing on our or our partners’ behalf, may fail to comply with regulatory requirements and industry standards or meet their contractual obligations in a timely manner or at all. |
21
Clinical trials are lengthy and expensive. Many of the factors listed above could result in increased clinical development costs or longer clinical development times for any of our programs. We or our partners incur substantial expense for, and devote significant time to, preclinical testing and clinical trials, yet we cannot be certain that the tests and trials will ever result in the commercial sale of a product. Even if we or our partners successfully complete clinical trials for our product candidates, we or our partners might not file the required regulatory submissions in a timely manner and may not receive regulatory approval for the product candidates, in which case would be unable to generate any revenues from product sales or licensing arrangements.
If our development collaborations with third parties, such as our development partners, contractors and contract research organizations, fail, the development of our product candidates will be delayed or stopped.
We rely heavily upon third parties for many important stages of our product candidate development, including but not limited to:
| • | discovery of natural proteins that cause or enable biological reactions necessary for the progression of the disease or disorder, called enzyme targets; | |
| • | execution of certain pharmacology preclinical studies and late-stage development for our compounds and product candidates; | |
| • | management of our Phase 1, 2 and 3 clinical trials, including medical monitoring, laboratory testing, and data management; | |
| • | execution of toxicology studies that may be required to obtain approval for our product candidates; | |
| • | formulation improvement strategies and methods; | |
| • | manufacturing the starting materials and drug substance required to formulate our products and the product candidates to be used in our clinical trials, toxicology studies and any potential commercial product; and | |
| • | management of certain regulatory interactions outside of the United States. | |
Our failure to engage in successful collaborations at any one of these stages would greatly impact our business. If we do not license enzyme targets or inhibitors from academic institutions or from other biotechnology companies on acceptable terms, our drug development efforts would suffer. Similarly, if the contract research organizations or third-party contractors that conduct our initial or late-stage clinical trials, conduct our toxicology or other studies, manufacture our starting materials, drug substance and product candidates, provide laboratory testing or other services in connection with our clinical trials, or assist with our regulatory function breach their obligations to us, perform their services inconsistent with industry standards, or fail to comply with regulatory requirements, this would delay or prevent both the development of our product candidates and the availability of any potential commercial product.
If we lose our relationship with any one or more of these parties, we could experience a significant delay in both identifying another comparable provider and then contracting for its services. We may be unable to retain an alternative provider on reasonable terms, if at all. Even if we locate an alternative provider, it is likely that this provider may need additional time to respond to our needs and may not provide the same type or level of service as the original provider. In addition, any provider that we retain will be subject to applicable FDA current Good Laboratory Practices, current Good Manufacturing Practices (“cGMP”) and current Good Clinical Practices, and comparable foreign standards. We do not have control over compliance with these regulations by these providers. Consequently, if these practices and standards are not adhered to by these providers, the development and commercialization of our product candidates could be delayed. If any of the foregoing risks are realized, our business, financial condition and results of operations could be materially adversely affected.
If we fail to obtain additional financing or acceptable partnership arrangements, we may be unable to complete the development and commercialization of our product candidates or continue operations.
As our programs advance, our costs are likely to increase. Our current and planned discovery, development, approval, and commercialization efforts will require significant capital. Our expenses, revenues and cash utilization rate could vary significantly depending on many factors, including: our ability to ability to obtain regulatory approval for and successfully commercialize our product candidates, including berotralstat, BCX9930, BCX9250, and galidesivir; our ability to raise additional capital; the amount of funding we receive from partnerships with third parties for the development and commercialization of our product candidates (including, our collaborations with Torii Pharmaceutical, Ltd. (“Torii”) and the U.S. Department of Health and Human Services (“BARDA/HHS and NIAID/HHS”); the commercial success of peramivir achieved by our partners; the amount or profitability of any orders for peramivir or galidesivir by any government agency or other party; the progress and results of our current and proposed clinical trials for our product candidates; and the progress made in the manufacture of our lead products and the progression of our other programs.
22
In order to continue future operations, progress our drug development programs, and commercialize our current product candidates, we will be required to raise additional capital. In addition to seeking strategic partnerships, transactions and government funding, we may decide to access the equity or debt markets, incur additional borrowings, or seek other sources to meet liquidity needs at any time. Additional funding, whether through additional sales of securities, additional borrowings, royalty or other monetization transactions, collaborative arrangements with partners, including corporate partners such as Torii and governmental agencies such as BARDA/HHS or NIAID/HHS, or from other sources, may not be available when needed or on terms acceptable to us. The issuance of preferred or common stock or convertible securities, with terms and prices significantly more favorable than those of our currently outstanding common stock, could have the effect of diluting or adversely affecting the holdings or rights of our existing stockholders. Additional borrowings may subject us to more restrictive covenants than are currently applicable to us under our secured credit facility with MidCap Financial, a Delaware statutory trust (“MidCap”), pursuant to the terms and conditions of our Second Amended and Restated Credit and Security Agreement, dated as of February 5, 2019, with MDCP, LLC, MidCap, and the lenders thereto (the “Second Amended and Restated Senior Credit Facility”). In addition, collaborative arrangements may require us to transfer certain material rights to such corporate partners. Insufficient funds or lack of an acceptable partnership may require us to delay, scale-back or eliminate certain of our research and development programs.
Our ability to raise additional capital when needed or at all may be limited and may greatly depend upon the success of our current drug development programs, including the progress, timeline and ultimate outcome of the development programs for our kallikrein inhibitors such as berotralstat (including but not limited to formulation progress, long-term human safety studies, and carcinogenicity, drug-drug interaction, toxicity, or other required studies), BCX9250 for the treatment of FOP, BCX9930 for diseases of the complement system, our broad-spectrum antiviral program, and other rare disease product candidates, as well as any post-approval studies for RAPIVAB. In addition, constriction and volatility in the equity and debt markets may restrict our future flexibility to raise capital when such needs arise. Furthermore, we have exposure to many different industries, financing partners and counterparties, including commercial banks, investment banks and partners (which include investors, licensing partners, and the U.S. Government) which may be unstable or may become unstable in the current economic and political environment. Any such instability may impact these parties’ ability to fulfill contractual obligations to us or they might limit or place burdensome conditions upon future transactions with us. Also, it is possible that suppliers may be negatively impacted. Any such unfavorable outcomes in our current programs or unfavorable economic conditions could place severe downward pressure on the price of our common stock and may decrease opportunities to raise capital in the capital or credit markets, and further could reduce the return available on invested corporate cash, which, if severe and sustained, could have a material and adverse impact on our results of operations and cash flows and limit our ability to continue development of our product candidates.
We may not be able to continue as a going concern if we do not obtain additional capital.
We have sustained operating losses for the majority of our corporate history and expect that our 2020 expenses will exceed our 2020 revenues. We expect to continue to incur operating losses and negative cash flows until revenues reach a level sufficient to support ongoing operations.
Our liquidity needs will be largely determined by the success of operations in regards to the progression of our product candidates in the future. Our plans to alleviate the doubt regarding our ability to continue as a going concern primarily include our ability to control the timing and spending on our research and development programs and raising additional funds through equity financings. We also may consider other plans to fund operations including: (1) securing or increasing U.S. Government funding of our programs, including obtaining additional and delivering on procurement contracts; (2) out-licensing rights to certain of our products or product candidates, pursuant to which the we would receive cash milestones and/or royalties; (3) raising additional capital through equity or debt financings or from other sources, including royalty or other monetization transactions; (4) obtaining additional product candidate regulatory approvals, which would generate revenue, milestones and cash flow; (5) reducing spending on research and development programs, including by discontinuing and suspending development; and/or (6) restructuring operations to change our overhead structure.
There can be no assurance that any of our plans will be successful or that additional capital will be available to us on reasonable terms, or at all, when needed. If we are unable to obtain sufficient additional capital, we may be forced to curtail operations, delay or stop ongoing clinical trials, cease operations altogether or file for bankruptcy.
If we or our partners do not obtain and maintain governmental approval for our product candidates, we or our partners will not be able to commercialize and sell these potential products, which would significantly harm our business because we will receive no revenue.
We or our partners must obtain regulatory approval before marketing or selling our products. If the FDA or a comparable foreign regulatory authority delays or denies regulatory approval of one of our product candidates, or revokes approval of a previously approved product, we would be unable to market or sell the product in the applicable jurisdiction and would not receive revenue from sales or licensing arrangements related thereto, which could have a material and adverse impact on our business.
23
The process of preparing for and obtaining regulatory approval in any jurisdiction may be lengthy and expensive, and approval is never certain. Because of the risks and uncertainties inherent to the development process, our product candidates could take a significantly longer time to gain regulatory approval than we expect or may never gain approval. As discussed under “Risk Factors—Risks Relating to Our Business—Our success depends upon our ability to advance our products through the various stages of development, especially through the clinical trial process, and to receive regulatory approval for the commercial sale of our products,” we or our partners may experience any number of unfavorable outcomes during or as a result of pre-clinical studies and clinical trials that could delay or prevent regulatory approval of our product candidates, or negatively impact our management’s credibility, our value and our operating results.
Even if the FDA or foreign regulatory authorities approve a product candidate, the approval may limit the indicated uses for a product candidate and/or may require post-approval studies that could impair the commercial viability of a product candidate. If we receive approval to market our potential products, whether in the United States or internationally, we will continue to be subject to extensive regulatory requirements. These requirements are wide ranging and govern, among other things:
| • | adverse drug experience reporting regulations; | |
| • | product promotion; | |
| • | product manufacturing, including good manufacturing practice requirements; and | |
| • | product changes or modifications. |
Our failure to comply with existing or future regulatory requirements for regulatory approval, or our loss of, or changes to, previously obtained approvals, could impair our ability to generate any revenues from product sales or licensing arrangements, which could have a material adverse effect on our business, financial condition, and results of operations.
We focus on rare diseases, which may create additional risks and challenges.
Because we focus on developing drugs as treatments for rare diseases, we may seek orphan drug, breakthrough therapy or fast track designations for our product candidates in the United States or the equivalent designations elsewhere in the world. Often, regulatory authorities have broad discretion in determining whether or not to grant such designations. We cannot guarantee that our product candidates will receive orphan drug status from the FDA or equivalent designations from other regulatory authorities. We also cannot guarantee that we will receive breakthrough therapy, fast track, or equivalent designations, which provide certain potential benefits such as more frequent meetings with the applicable regulatory authorities to discuss development plans, intensive guidance on efficient drug development programs, and potential eligibility for rolling review or priority review. Even if we are successful in obtaining any such designations for our product candidates, such designations may not lead to faster development or regulatory review or approval and do not increase the likelihood that our product candidates will receive marketing approval. For instance, although berotralstat for HAE prophylaxis has received Fast Track designation from the FDA, Sakigake designation from the Japanese Pharmaceuticals and Medical Devices Agency (“PDMA”), and Promising Innovative Medicine designation from the MHRA, as well as orphan drug status from the FDA, EMA, and the MHLW, we may not experience a faster development, review or approval process compared to the conventional process in the relevant jurisdictions. We may not be able to obtain or maintain these designations for berotralstat or other product candidates that receive them, and our competitors may obtain these designations for their product candidates, which could impact our ability to develop and commercialize our product candidates or compete with such competitors, which may adversely impact our business, financial condition or results of operations.
The commercial viability of any approved product could be compromised if the product is less effective than expected, causes undesirable side effects that were not previously identified, or fails to achieve market acceptance within the medical community.
If after obtaining regulatory approval of a product we or others discover that it is less effective than previously believed or causes undesirable side effects that were not previously identified, any of the following adverse events could occur:
| • | regulatory authorities may withdraw their approval of, or impose marketing or manufacturing restrictions on, the product, or require us or our partners to create a medication guide outlining the risks of unidentified side effects for distribution to patients; |
| • | we or our partners may be required to recall the product, change the way the product is administered, conduct additional clinical trials, or be subject to civil or criminal penalties; |
| • | the product may become less competitive and our reputation may suffer. |
24
Even after receiving regulatory approval, any product could fail to gain sufficient, or even any, market acceptance by physicians, patients, third party payors, health authorities and others in the medical community. For example, physicians are often reluctant to switch their patients from existing therapies even when new and potentially more effective or convenient treatments enter the market. Further, patients often acclimate to the therapy that they are currently taking and do not want to switch unless their physicians recommend switching products or they are required to switch therapies due to lack of reimbursement for existing therapies. If an approved product does not achieve an adequate level of market acceptance, it may not generate significant revenues. The occurrence of any of the foregoing could have a material and adverse impact on our business.
If we fail to successfully commercialize or establish collaborative relationships to commercialize certain of our product candidates, or if any partner terminates or fails to perform its obligations under agreements with us, potential revenues from commercialization of our product candidates could be reduced, delayed or eliminated.
Our business strategy is to increase the asset value of our product candidate portfolio. We believe this is best achieved by retaining full product rights or through collaborative arrangements with third parties as appropriate. As needed, potential third-party relationships could include preclinical development, clinical development, regulatory approval, marketing, sales and distribution of our product candidates.
Currently, we have established collaborative relationships with Torii for the commercialization of berotralstat in Japan, with each of SUL, Shionogi and Green Cross for the development and commercialization of peramivir, and with Mundipharma for the development and commercialization of Mundesine (forodesine). The process of establishing and implementing collaborative relationships is difficult, time-consuming and involves significant uncertainty, including:
| • | our partners may seek to renegotiate or terminate their relationships with us due to unsatisfactory commercial, regulatory or clinical results, including post approval clinical commitments, a change in business strategy, a change of control or other reasons; | ||
| • | our contracts for collaborative arrangements may expire; | ||
| • | our partners may choose to pursue alternative technologies, including those of our competitors; | ||
| • | we may have disputes with a partner that could lead to litigation or arbitration, such as the recent arbitration proceeding between us and SUL, which could result in substantial costs and divert the attention of our management; | ||
| • | we do not have day to day control over the activities of our partners and have limited control over their decisions; | ||
| • | our ability to generate future event payments and royalties from our partners depends upon their abilities to establish the safety and efficacy of our product candidates, obtain regulatory approvals and achieve market acceptance of products developed from our product candidates; | ||
| • | we or our partners may fail to properly initiate, maintain or defend our intellectual property rights, where applicable, or a party may utilize our proprietary information in such a way as to invite litigation that could jeopardize or potentially invalidate our proprietary information or expose us to potential liability; | ||
| • | we or our partners may not devote sufficient capital or resources towards our product candidates; and | ||
| • | we or our partners may not comply with applicable government regulatory requirements. |
If we or our partners fail to fulfill our responsibilities in a timely manner, or at all, our commercialization efforts related to that collaboration could be reduced, delayed or terminated, or it may be necessary for us to assume responsibility for activities that would otherwise have been the responsibility of our partner. If we are unable to establish and maintain collaborative relationships on acceptable terms, we may have to delay or discontinue further development of one or more of our product candidates, undertake commercialization activities at our own expense or find alternative sources of funding. Any delay in the development or commercialization of our product candidates would severely affect our business, because if our product candidates do not progress through the development process or reach the market in a timely manner, or at all, we may not receive any revenues from product sales or licensing arrangements.
The results of our partnership with Torii may not meet our current expectations.
We have an agreement with Torii for the development and commercialization of berotralstat in Japan (the “Torii Agreement”). We do not have a history of working with Torii and cannot predict the success of this collaboration. Our ability to realize the expected benefits of this collaboration, including with respect to the receipt or amounts of potential milestone or royalty payments, is subject to a number of risks, including that applicable regulatory agencies may not provide adequate regulatory clearances or reimbursement approvals on a timely basis or at all, the commercial potential of berotralstat may not meet our current expectations, we or Torii may fail to comply with our respective obligations under the Torii Agreement, and third parties may fail to perform their obligations to us on a timely basis or at all.
25
The Torii Agreement provides for a potential milestone payment depending on the receipt and timing of regulatory approval and contingent upon receipt of a reimbursement price approval from Japan’s National Health Insurance system in excess of the threshold specified in the Torii Agreement, either of which we may not receive on a timely basis or at all. The Torii Agreement also provides that we will be entitled to receive tiered royalty payments, the amounts of which will depend upon the amount of annual net sales of berotralstat in Japan during each calendar year, whether berotralstat maintains its Sakigake designation, and other factors. We remain responsible for regulatory activities with respect to berotralstat in Japan for one year after the first commercial sale. We expect to use third parties to satisfy many of our obligations under the Torii Agreement, including but not limited to our regulatory and other responsibilities in Japan. If our interactions, or those of our third party agents, are unsuccessful, we could fail to meet our obligations under the Torii Agreement, fail to receive regulatory approval of berotralstat on a timely basis or at all, receive approval of berotralstat on a narrower scope than currently anticipated, or fail to receive reimbursement authorization in excess of the specified threshold, which could negatively impact the commercial success and the partnership, impact the economic benefit expected or require additional development of berotralstat.
Torii may terminate the Torii Agreement under certain limited circumstances, including the receipt of notice that certain additional development activities are required for regulatory approval of berotralstat, if regulatory approval of berotralstat is not received prior to December 31, 2022, or upon one year’s written notice after the sixth anniversary of the first commercial sale of berotralstat in Japan. If the Torii Agreement is terminated in connection with these provisions, we will no longer be entitled to receive any milestone or royalty payments thereunder, which could have a material adverse impact on our business and results of operations.
Torii will have sole control over and decision-making authority with respect to commercialization activities for berotralstat for the prevention of HAE attacks in Japan, subject to oversight from a joint steering committee. Therefore, our receipt of, and the amounts of, any royalty payments under the Torii Agreement are dependent upon Torii’s successful performance of such commercialization activities. In addition, competitive products and variations in patient demand, prescription levels, reimbursement determinations or other factors may limit the commercial potential of berotralstat in Japan, which could materially reduce the amount of any royalties we would be entitled to receive under the Torii Agreement.
Under the Torii Agreement, we will be responsible for supplying Torii with its required amounts of berotralstat for commercial sale. If due to the failure of our third-party contract manufacturers to produce sufficient drug product we fail to supply to Torii the required amounts of berotralstat, then Torii’s ability to successfully commercialize berotralstat in Japan could be materially impaired, and we may receive less royalty income under the Torii Agreement, or none at all.
Any of the foregoing risks could materially adversely impact our ability to obtain regulatory approval of berotralstat in Japan, the price of berotralstat in Japan, and to perform our obligations under the Torii Agreement, which could materially reduce the economic benefits of the Torii Agreement to us and impair or result in the termination of our collaboration with Torii.
We do not have a great deal of experience in commercializing our products or technologies, and our future revenue generation is uncertain.
We do not have a great deal of experience in commercializing our product candidates or technologies. We currently have limited sales, marketing and distribution capabilities, and we may be unable to establish or sufficiently increase these capabilities for products we currently, or plan to, commercialize. Our ability to receive revenue from products we or our partners commercialize is subject to several risks, including:
| • | we or our partners may fail to successfully complete clinical trials, or satisfy post-marketing commitments, sufficient to obtain and keep regulatory agency marketing approval; | |
| • | many competitors are more experienced and have significantly more resources, and their products could reach the market faster, be more cost effective or have a better efficacy or tolerability profile than our product candidates; | |
| • | we may fail to employ a comprehensive and effective intellectual property strategy, which could result in decreased commercial value of our Company and our products; | |
| • | we may fail to employ a comprehensive and effective regulatory strategy, which could result in a delay or failure in commercialization of our products; | |
| • | our ability to successfully commercialize our products is affected by the competitive landscape, which cannot be fully known at this time; | |
| • | reimbursement is constantly changing, which could greatly affect usage of our products; and | |
| • | future revenue from product sales would depend on our ability to successfully complete clinical studies, obtain regulatory approvals, and manufacture, market and commercialize our approved drugs. |
26
We expect to expand our development and regulatory capabilities and implement sales, marketing and distribution capabilities, and as a result, we may encounter difficulties in managing our growth, which could disrupt our operations.
We expect to experience significant growth in the number of our employees and the scope of our operations, particularly in the areas of drug development, regulatory affairs and, if any of our product candidates currently in development receive marketing approval, sales, marketing and distribution. To manage our anticipated future growth, we must continue to implement and improve our managerial, operational and financial systems, expand our facilities and continue to recruit and train additional qualified personnel. Due to our limited financial resources and the limited experience of our management team in managing a company with such anticipated growth, we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel. The expansion of our operations may lead to significant costs and may divert our management and business development resources. Any inability to manage growth could delay the execution of our business plans or disrupt our operations.
Because we have limited manufacturing experience, we depend on third-party manufacturers to manufacture our products, product candidates and the materials for our product candidates. Often, especially early in the development and commercialization process, we have only one source for manufacturing. If we cannot rely on existing third-party manufacturers, we will be required to incur significant costs and potential delays in finding new third-party manufacturers.
We have limited manufacturing experience and only a small scale manufacturing facility. We currently rely upon a very limited number of third-party manufacturers to manufacture the materials required for our products, product candidates and most of the preclinical and clinical quantities of our product candidates. We depend on these third-party manufacturers to perform their obligations in a timely manner and in accordance with applicable governmental regulations. Our third-party manufacturers, which may be the only manufacturer we have engaged for a particular product, may encounter difficulties with meeting our requirements, including but not limited to problems involving:
| • | inconsistent production yields; | |
| • | product liability claims or recalls of commercial product; | |
| • | difficulties in scaling production to commercial and validation sizes; | |
| • | interruption of the delivery of materials required for the manufacturing process; | |
| • | scheduling of plant time with other vendors or unexpected equipment failure; | |
| • | potential catastrophes that could strike their facilities or have an effect on infrastructure; | |
| • | potential impurities in our drug substance or products that could affect availability of product for our clinical trials or future commercialization; | |
| • | poor quality control and assurance or inadequate process controls; and | |
| • | lack of compliance or cooperation with regulations and specifications or requests set forth by the FDA or other foreign regulatory agencies or local customs, particularly associated with berotralstat, BCX9930, BCX9250, galidesivir, peramivir and our early stage compounds. |
These contract manufacturers may not be able to manufacture the materials required for our product candidates at a cost or in quantities necessary to make them commercially viable. Our raw materials, drug substances, and product candidates are manufactured by a limited group of suppliers, including some at a single facility. If any of these suppliers were unable to produce these items, this could significantly impact our supply of product candidate material for further preclinical testing and clinical trials. We also have no control over whether third-party manufacturers breach their agreements with us or whether they may terminate or decline to renew agreements with us. To date, our third-party manufacturers have met our manufacturing requirements, but they may not continue to do so. Furthermore, changes in the manufacturing process or procedure, including a change in the location where the drug is manufactured or a change of a third-party manufacturer, may require prior review and approval in accordance with the FDA’s cGMP and comparable foreign requirements. This review may be costly and time-consuming and could delay or prevent the launch of a product. The FDA or foreign regulatory authorities may at any time implement new standards, or change their interpretation and enforcement of existing standards for manufacture, packaging or testing of products. If we or our contract manufacturers are unable to comply, we or they may be subject to regulatory action, civil actions or penalties any of which could be costly to us and could result in a delay or shortage of product.
27
If we are unable to maintain current manufacturing or other contract relationships, or enter into new agreements with additional manufacturers on commercially reasonable terms, or if there is poor manufacturing performance or failure to comply with any regulatory agency on the part of any of our third-party manufacturers, we may not be able to complete development of, obtain timely approval of, or commercialize, our product candidates.
Commercialization of peramivir by our partners is subject to the potential commercialization risks described herein and numerous additional risks. Any potential revenue benefits to us in the form of milestone payments, royalties or other consideration are highly speculative.
Commercialization success of peramivir is uncertain and is subject to all the risks and uncertainties disclosed in our other risk factors relating to drug development and commercialization. In addition, commercialization of peramivir products is subject to further risks and may be negatively impacted by a number of factors, including, but not limited to, the following:
| • | peramivir may not prove to be adequately safe and effective for market approval in markets other than the United States, Canada, Japan, Korea, Taiwan, Australia and the European Union (“EU”); | ||
| • | necessary funding for post-marketing commitments and further development of peramivir may not be available timely, at all, or in sufficient amounts; | ||
| • | flu prevention or pandemic treatment concerns may not materialize at all, or in the near future; | ||
| • | advances in flu vaccines or other antivirals, including competitive i.v. antivirals, could substantially replace potential demand for peramivir; | ||
| • | a limited number of governmental entities are expected to be the primary potential stockpiling customers for peramivir and if we are not successful at marketing peramivir to these entities for any reason, we will not receive substantial revenues from stockpiling orders; | ||
| • | government and third party payors may not provide sufficient coverage or reimbursement which would negatively impact the demand for peramivir; | ||
| • | we may not be able to supply commercial material to our partners and our partners may not be able to maintain or establish sufficient and acceptable commercial manufacturing, either directly or through third-party manufacturers; | ||
| • | the commercial demand and acceptance for peramivir by healthcare providers and by patients may not be sufficient to result in substantial revenues of peramivir to our partners and may result in little to no milestones or royalties to us; | ||
| • | effectiveness of marketing and commercialization efforts for peramivir by our partners; | ||
| • | market satisfaction with existing alternative therapies; | ||
| • | perceived efficacy relative to other available therapies; | ||
| • | disease prevalence; | ||
| • | cost of treatment; | ||
| • | pricing and availability of alternative products; | ||
| • | marketing and sales activities of competitors; | ||
| • | shifts in the medical community to new treatment paradigms or standards of care; and | ||
| • | relative convenience and ease of administration. | ||
28
We face intense competition, and if we are unable to compete effectively, the demand for our products, if any, may be reduced.
The biotechnology and pharmaceutical industries are highly competitive and subject to rapid and substantial technological change. There are many companies seeking to develop products for the same indications that we currently target. Our competitors in the United States and elsewhere are numerous and include, among others, major multinational pharmaceutical and chemical companies and specialized biotechnology firms. Most of these competitors have greater resources than we do, including greater financial resources, larger research and development staffs and more experienced marketing and manufacturing organizations. In addition, most of our competitors have greater experience than we do in conducting clinical trials and obtaining FDA and other regulatory approvals. Accordingly, our competitors may succeed in obtaining FDA or other regulatory approvals of product candidates more rapidly than we do. Companies that complete clinical trials, obtain required regulatory approvals, and commence commercial sale of their drugs before we do may achieve a significant competitive advantage, including patent and FDA exclusivity rights that would delay our ability to market products. We face, and will continue to face, competition in the licensing of potential product candidates for desirable disease targets, licensing of desirable product candidates, and development and marketing of our product candidates from academic institutions, government agencies, research institutions and biotechnology and pharmaceutical companies. Competition may also arise from, among other things:
| • | other drug development technologies; | |
| • | methods of preventing or reducing the incidence of disease, including vaccines; and | |
| • | new small molecule or other classes of therapeutic agents. |
Developments by others may render our product candidates or technologies obsolete or noncompetitive.
We are performing research on or developing products for the treatment of several rare diseases, including HAE, diseases of the complement system, and FOP, as well as developing broad spectrum antivirals for use as medical countermeasures. We expect to encounter significant competition for any of the pharmaceutical products we are developing and plan to develop. Companies that complete clinical trials, obtain required funding or government support, obtain required regulatory approvals and commence commercial sales or stockpiling orders of their products before their competitors may achieve a significant competitive advantage. There are licensed therapies for HAE (including Barinert®, Haegarda®, Cinryze®, Kalbitor®, Takzyro®, Finrazyr® (icatibant) and generic icatibant), therapies for certain complement-mediated diseases such as PNH, aHUS, myasthenia gravis, and neuroyelitis optica specrtum disorder (Soliris® and UltomirisTM), products for the prevention or treatment of influenza (seasonal flu vaccines, Tamiflu® (oseltamivir), generic oseltamivir, Relenza®, and Inavir®, favipiravir, and Xofluza™), and a number additional of products in clinical development in these therapeutic areas and for the treatment of FOP. In addition, various government entities throughout the world may offering incentives, grants and contracts to encourage additional investment into preventative and therapeutic agents against viruses such as influenza, coronavirus, Ebola, and others, which may have the effect of further increasing the number of our competitors and/or providing advantages to certain competitors. See “Business—Competition” for further discussion of our competitors, competitive products or programs, and the competitive conditions in these and other therapeutic areas.
If one or more of our competitors’ products or programs, including potential competitors not currently identified, are successful, the market for our products may be reduced or eliminated.
Compared to us, many of our competitors and potential competitors have substantially greater:
| • | capital resources; | |
| • | research and development resources, including personnel and technology; | |
| • | regulatory experience; | |
| • | preclinical study and clinical testing experience; | |
| • | manufacturing and marketing experience; and | |
| • | production facilities. |
Any of these competitive factors could impede our funding efforts, render technology and product candidates noncompetitive or eliminate or reduce demand for our product candidates.
29
We are subject to various laws and regulations related to our products and product candidates and, if we or our partners do not comply with these laws and regulations, we could face substantial penalties.
Our or our partners’ activities related to approved products, such as RAPIVAB/ALPIVAB (peramivir), or, following their regulatory approval, any of our product candidates under development, such as berotralstat, BCX9930, BCX925, and galidesivir, are subject to regulatory and law enforcement authorities in the United States (including the FDA, the Federal Trade Commission, the Department of Justice, and state and local governments) and their foreign equivalents (including the EMA, MHLW, MHRA, and others).
We are responsible for reporting adverse drug experiences, have responsibility for certain post-approval studies, and may have responsibilities and costs related to a recall or withdrawal of RAPIVAB/ALPIVAB from sale in the jurisdictions in which it is approved. We may also incur liability associated with RAPIVAB/ALPIVAB manufacturing contracted by us or in support of any of our partners. We are required to maintain records and provide data and reports to regulatory agencies related to RAPIVAB/ALPIVAB (e.g. risk evaluation and mitigation strategies, track and trace requirements, adverse events), and we may incur certain promotional regulatory and government pricing risks, all of which could have a material adverse impact on our operations and financial condition. Similar responsibilities would apply upon regulatory approval of any of our other product candidates currently under development.
In addition, we are subject to the federal physician sunshine act and certain similar physician payment and drug pricing transparency legislation in various states. We are also subject to various federal and state laws pertaining to health care “fraud and abuse,” including both federal and state anti-kickback and false claims laws. Outside of the United States, we may be subject to analogous foreign laws and regulations in the various jurisdictions in which we operate. These laws and regulations apply to our or our partners’ operations, sales and marketing practices, price reporting, and relationships with physicians and other customers and third-party payors. Anti-kickback laws generally prohibit a manufacturer from soliciting, offering, receiving, or paying any remuneration to generate business, including the purchase or prescription of a particular drug. Although the specific provisions of these laws vary, their scope is generally broad and there may be no regulations, guidance or court decisions that clarify how the laws apply to particular industry practices. False claims laws prohibit anyone from knowingly and willingly presenting, or causing to be presented for payment to third party payors (including Medicare and Medicaid) claims for reimbursement or services that are false or fraudulent, claims for items or services not provided as claimed, or claims for medically unnecessary items or services. The sunshine provisions apply to manufacturers with products reimbursed under certain government programs and require those manufacturers to disclose annually to the federal government certain payments made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors) and teaching hospitals, as well as, ownership and investment interests held by physicians (as defined above) and their immediate family members. State laws may also require disclosure of pharmaceutical pricing information and marketing expenditures. Although we seek to comply with these statutes, it is possible that our practices, or those of our partners, might be challenged under health care fraud and abuse, anti-kickback, false claims or similar laws. Violations of the physician sunshine act and similar legislation or the fraud and abuse laws may be punishable by civil or criminal sanctions, including fines and civil monetary penalties, and future exclusion from participation in government healthcare programs.
The principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time and receive compensation in connection with such services. Under certain circumstances, we may be required to report some of these relationships to certain regulatory authorities, including the FDA and comparable foreign regulatory authorities. Consequently, the FDA or other regulatory authority may conclude that a financial relationship between us and a principal investigator creates a conflict of interest or otherwise affects interpretation of the study. In the event of a conflict of interest with respect to a study, the integrity of the data generated at the applicable clinical trial site may be questioned or the utility of the clinical trial itself may be jeopardized. This could result in a delay in approval, or rejection, of our marketing applications by the FDA or other regulatory authority, as the case may be, and may ultimately lead to the denial of marketing approval of one or more of our product candidates.
We have a number of outstanding post-approval commitments to the FDA and EMA that we retain, which we may not complete successfully or on time for any number of reasons, including but not limited to lack of funds to complete the studies and insufficient interest by appropriate sites, investigators or study subjects. For example, as a condition of the approval of RAPIVAB/ALPIVAB, we were required to complete pediatric patient trials and to submit the final results of these clinical trials to the FDA and EMA. We may be subject to penalties if we fail to comply with post-approval legal and regulatory requirements and our products could be subject to continual recordkeeping and reporting requirements, review and periodic inspections by the FDA and other regulatory bodies. Regulatory approval of a product may be subject to limitations on the indicated uses for which the product may be marketed or to the other restrictive conditions of approval that limit our ability to promote, sell or distribute a product. Furthermore, the approval of RAPIVAB/ALPIVAB and any other future product candidates may be subject to requirements for costly post-approval testing and surveillance to monitor its safety or efficacy.
Advertising and promotion are subject to stringent FDA rules and oversight and as an NDA-holder we may be held responsible for any advertising and promotion that is not in compliance with the rules and regulations. In particular, the claims in all promotional materials and activities must be consistent with the FDA approvals for approved products, and must be appropriately substantiated and fairly balanced with information on the safety risks and limitations of the products. Adverse event information concerning approved products must be reviewed and as an NDA-holder we are required to make expedited and periodic adverse event reports to the FDA and other regulatory authorities. In addition, the research, manufacturing, distribution, sale and promotion of products are potentially subject to regulation by various federal, state and local authorities in addition to the FDA, including the Centers for Medicare and Medicaid Services, other divisions of the U.S. Department of Health and Human Services, the U.S. Department of Justice and individual U.S. Attorney offices within the Department of Justice, state and local governments, and foreign equivalents of the foregoing. All of these activities are also potentially subject to healthcare false claims and fraud and abuse laws, as well as consumer protection and unfair competition laws.
30
If our operations with respect to RAPIVAB/ALPIVAB or our other products that are subject to healthcare laws and regulations are found to be in violation of any of the healthcare fraud and abuse laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including civil and criminal penalties, damages, fines and the curtailment or restructuring of our operations. Any penalties, damages, fines, curtailment or restructuring of our operations could adversely affect our ability to operate our business and our financial results. Although compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, the risks cannot be entirely eliminated. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management's attention from the operation of our business. Moreover, achieving and sustaining compliance with all applicable fraud and abuse laws may be costly.
International expansion of our business exposes us to business, regulatory, political, operational, financial, and economic risks.
Our business strategy includes international expansion, including the commercialization of products outside of the United States. We currently conduct clinical studies and regulatory activities and have hired, and expect to continue hiring, employees outside of the United States. Doing business internationally involves a number of risks, including but not limited to:
| • | multiple, conflicting, and changing laws and regulations such as privacy and data regulations, transparency regulations, tax laws, export and import restrictions, employment laws, regulatory requirements, and other governmental approvals, permits, and licenses; |
| • | introduction of new health authority requirements and/or changes in health authority expectations; |
| • | failure by us or our partners to obtain and maintain regulatory approvals for the use of our products in various countries; |
| • | complexities and difficulties in obtaining protection for, and enforcing, our intellectual property; |
| • | difficulties in staffing and managing foreign operations; |
| • | complexities associated with managing multiple payor reimbursement regimes, government payors, or patient self-pay systems; |
| • | limits on our ability to penetrate international markets; |
| • | financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for our products, and exposure to foreign currency exchange rate fluctuations; |
| • | natural disasters and political and economic instability, including wars, terrorism, political unrest, results of certain elections and votes, actual or threatened public health emergencies and outbreak of disease (including for example, the recent coronavirus outbreak), boycotts, adoption or expansion of government trade restrictions, and other business restrictions; |
| • | certain expenses including, among others, expenses for travel, translation, and insurance; |
| • | regulatory and compliance risks that relate to maintaining accurate information and control over commercial operations and activities that may fall within the purview of the U.S. Foreign Corrupt Practices Act, or FCPA, including its books and records provisions or anti-bribery provisions, or the U.K. Bribery Act and similar foreign laws and regulations; and |
| • | regulatory and compliance risks relating to doing business with any entity that is subject to sanctions administered by the Office of Foreign Assets Control of the U.S. Department of the Treasury. |
Any of these factors could significantly harm our future international expansion of operations and, consequently, our business and results of operations.
Additionally, in some countries, such as Japan and the countries of the EU, the pricing of prescription pharmaceuticals is subject to governmental control and access. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a product. To obtain reimbursement or pricing approval in some countries, we or our partners may be required to conduct a clinical trial that compares the cost-effectiveness of our product to other available therapies. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business could be materially harmed.
31
Our employees and consultants may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements, which could cause significant liability for us and harm our reputation.
We are subject to the risk of fraud or other misconduct by our employees and consultants, including intentional failures to comply with FDA regulations or similar regulations of comparable foreign regulatory authorities, provide accurate information to the FDA or comparable foreign regulatory authorities, comply with manufacturing standards we have established, comply with federal and state healthcare fraud and abuse laws and regulations and similar laws and regulations established and enforced by comparable foreign regulatory authorities, report financial information or data accurately or disclose unauthorized activities to us. Employee and consultant misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter employee and consultant misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws, standards or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of significant fines or other sanctions.
We and our partners may be subject to new legislation, regulatory proposals and healthcare payor initiatives that may increase our costs of compliance and adversely affect our or our partners’ ability to market our products, obtain collaborators and raise capital.
The Patient Protection and Affordable Care Act, or PPACA, made extensive changes to the delivery of health care in the U.S. The PPACA included numerous provisions that affect pharmaceutical companies, some of which became effective immediately and others of which have taken effect over the past several years. For example, the PPACA expanded health care coverage to the uninsured through private health insurance reforms and an expansion of Medicaid. The PPACA also imposed substantial costs on pharmaceutical manufacturers, such as an increase in liability for rebates paid to Medicaid, new drug discounts that must be offered to certain enrollees in the Medicare prescription drug benefit, an annual fee imposed on all manufacturers of brand prescription drugs in the U.S., and an expansion of an existing program requiring pharmaceutical discounts to certain types of hospitals and federally subsidized clinics. The PPACA also contains cost containment measures that could reduce reimbursement levels for health care items and services generally, including pharmaceuticals. It also required reporting and public disclosure of payments and other transfers of value provided by pharmaceutical companies to physicians and teaching hospitals.
We expect that the current presidential administration and U.S. Congress may continue to seek to modify, repeal, or otherwise invalidate all, or certain provisions of, the PPACA or undertake other reforms that impact the pharmaceutical industry. For instance, the Trump administration has taken several executive actions, including the issuance of a number of executive orders, that could impose significant burdens on, or otherwise materially delay, the FDA’s ability to engage in routine regulatory and oversight activities such as implementing statutes through rulemaking, issuance of guidance, and review and approval of marketing applications. An under-staffed FDA could result in delays in the FDA’s responsiveness or in its ability to review submissions or applications within the established Prescription Drug User Fee Act time frames, issue regulations or guidance, or implement or enforce regulatory requirements in a timely fashion or at all. There is still significant uncertainty with respect to the impact that the current presidential administration and the U.S. Congress may have on the PPACA specifically and the healthcare industry generally, and any changes will likely take time to unfold. As such, we cannot predict what effect the PPACA or other healthcare reform initiatives that may be adopted in the future will have on our business.
The continuing efforts of the government, insurance companies, managed care organizations and other payors of health care services to contain or reduce costs of health care could result in decreased net revenues from our pharmaceutical products and decrease potential returns from our development efforts. In addition, pharmaceutical and device manufacturers are also required to report and disclose certain payments and transfers of value to, and investment interests held by, physicians and their immediate family members during the preceding calendar year. Failure to submit required information may result in civil monetary penalties for payments, transfers of value or ownership or investment interests not reported in an annual submission. Compliance with the PPACA and state laws with similar provisions is difficult and time consuming, and companies that do not comply with these state laws face civil penalties. Because of the breadth of these laws and the narrowness of the safe harbors, it is possible that some of our business activities could be subject to challenge under one or more of such laws. Such a challenge could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
In addition, there have been a number of other legislative and regulatory proposals aimed at changing the pharmaceutical industry. For example, legislation has been enacted in certain states and at a federal level that requires development of an electronic pedigree to track and trace each prescription drug at the saleable unit level through the distribution system. Compliance with these electronic pedigree requirements may increase our operational expenses and impose significant administrative burdens. In addition, our compliance may be deemed insufficient and we could face a material adverse effect on our business, financial condition, results of operations and growth prospects. As a result of these and other new proposals, we may determine to change our current manner of operation, provide additional benefits or change our contract arrangements, any of which could have a material adverse effect on our business, financial condition and results of operations.
32
Adequate coverage and reimbursement in the U.S. and other markets is critical to the commercial success of RAPIVAB or any other product that we might bring to market. Recently in the U.S. there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several Congressional inquiries and proposed bills designed to, among other things, reform government program reimbursement methodologies. Individual states in the United States have been increasingly active in passing legislation and implementing regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. Regional health care authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other health care programs. Third-party payors are increasingly challenging the prices charged for medical products and services and, in some cases, imposing restrictions on the coverage of particular drugs. Many third-party payors negotiate the price of medical services and products and develop formularies which establish pricing and reimbursement levels. Exclusion of a product from a formulary can lead to its sharply reduced usage in the third-party payor’s patient population. The process for obtaining coverage can be lengthy and costly, and we expect that it could take several months before a particular payor initially reviews our product and makes a decision with respect to coverage. For example, third-party payors may require cost-benefit analysis data from us in order to demonstrate the cost-effectiveness of RAPIVAB or any other product we might bring to market. For any individual third-party payor, we may not be able to provide data sufficient to gain reimbursement on a similar or preferred basis to competitive products, or at all which may have a material adverse effect on our business, financial condition and results of operations.
If we fail to adequately protect or enforce our intellectual property rights or secure rights to patents of others, the value of those rights would diminish.
Our success will depend in part on our ability and the abilities of our partners to obtain, protect and enforce viable intellectual property rights including but not limited to trade name, trademark and patent protection for our Company and its products, methods, processes and other technologies we may license or develop, to preserve our trade secrets, and to operate without infringing the proprietary rights of third parties both domestically and abroad. The patent position of biotechnology and pharmaceutical companies is generally highly uncertain, involves complex legal and factual questions and has recently been the subject of much litigation. Neither the United States Patent and Trademark Office (“USPTO”), the Patent Cooperation Treaty offices, nor the courts of the United States and other jurisdictions have consistent policies nor predictable rulings regarding the breadth of claims allowed or the degree of protection afforded under many biotechnology and pharmaceutical patents. Further, we may not have worldwide patent protection for all of our product candidates and our intellectual property rights may not be legally protected or enforceable in all countries throughout the world. In some jurisdictions, some of our product candidates in certain programs, including our HAE program, may have short or no composition of matter patent life and we may therefore rely on orphan drug exclusivity or data exclusivity. There can be no assurance that we will obtain orphan drug exclusivity or data exclusivity in every jurisdiction. Further, in some jurisdictions, we may rely on formulation patents or method of use patents. Both the ability to achieve issuance and the enforcement of formulation and method of use patents can be highly uncertain and can vary from jurisdiction to jurisdiction, and such patents may therefore not adequately prevent competitors and potential infringers in some jurisdictions. The validity, scope, enforceability and commercial value of the rights protected by such patents, therefore, is highly uncertain.
We also rely on trade secrets to protect technology in cases when we believe patent protection is not appropriate or obtainable. However, trade secrets are difficult to protect. If we cannot maintain the confidentiality of our technology and other confidential information in connection with our collaborators and advisors, our ability to receive patent protection or protect our proprietary information may be imperiled.
We may be involved in legal proceedings to protect or enforce our patents, the patents of our partners or our other intellectual property rights, which could be expensive, time consuming and unsuccessful.
Competitors may infringe or otherwise violate our patents, the patents of our licensors or our other intellectual property rights. To counter infringement or unauthorized use, we may be required to file legal claims, which can be expensive and time-consuming and unsuccessful. An adverse result in any legal proceeding could put one or more of our patents at risk. Our success depends in part on avoiding the infringement of other parties’ patents and other intellectual property rights as well as avoiding the breach of any licenses relating to our technologies and products. In the United States, patent applications filed in recent years are confidential for 18 months, while older applications are not published until the patent issues. As a result, avoiding patent infringement may be difficult and we may inadvertently infringe third-party patents or proprietary rights. These third parties could bring claims against us, our partners or our licensors that even if resolved in our favor, could cause us to incur substantial expenses and, if resolved against us, could additionally cause us to pay substantial damages. Further, if a patent infringement suit were brought against us, our partners or our licensors, we or they could be forced to stop or delay research, development, manufacturing or sales of any infringing product in the country or countries covered by the patent we infringe, unless we can obtain a license from the patent holder. Such a license may not be available on acceptable terms, or at all, particularly if the third party is developing or marketing a product competitive with the infringing product. Even if we, our partners or our licensors were able to obtain a license, the rights may be nonexclusive, which would give our competitors access to the same intellectual property.
33
If we or our partners are unable or fail to adequately initiate, protect, defend or enforce our intellectual property rights in any area of commercial interest or in any part of the world where we wish to seek regulatory approval for our products, methods, processes and other technologies, the value of the product candidates to produce revenue would diminish. Additionally, if our products, methods, processes, and other technologies or our commercial use of such products, processes, and other technologies, including but not limited to any trade name, trademark or commercial strategy infringe the proprietary rights of other parties, we could incur substantial costs. The USPTO and the patent offices of other jurisdictions have issued to us a number of patents for our various inventions and we have in-licensed several patents from various institutions. We have filed additional patent applications and provisional patent applications with the USPTO. We have filed a number of corresponding foreign patent applications and intend to file additional foreign and U.S. patent applications, as appropriate. We have also filed certain trademark and trade name applications worldwide. We cannot assure you as to:
| • | the degree and range of protection any patents will afford against competitors with similar products; | |
| • | if and when patents will issue; | |
| • | if patents do issue we cannot be sure that we will be able to adequately defend such patents and whether or not we will be able to adequately enforce such patents; or | |
| • | whether or not others will obtain patents claiming aspects similar to those covered by our patent applications. |
If the USPTO or other foreign patent office upholds patents issued to others or if the USPTO grants patent applications filed by others, we may have to:
| • | obtain licenses or redesign our products or processes to avoid infringement; | |
| • | stop using the subject matter claimed in those patents; or | |
| • | pay damages. |
We may initiate, or others may bring against us, litigation or administrative proceedings related to intellectual property rights, including proceedings before the USPTO or other foreign patent office. Any judgment adverse to us in any litigation or other proceeding arising in connection with a patent or patent application could materially and adversely affect our business, financial condition and results of operations. In addition, the costs of any such proceeding may be substantial whether or not we are successful.
Our success is also dependent upon the skills, knowledge and experience, none of which is patentable, of our scientific and technical personnel. To help protect our rights, we require all employees, consultants, advisors and partners to enter into confidentiality agreements that prohibit the disclosure of confidential information to anyone outside of our company and require disclosure and assignment to us of their ideas, developments, discoveries and inventions. These agreements may not provide adequate protection for our trade secrets, know-how or other proprietary information in the event of any unauthorized use or disclosure or the lawful development by others of such information, and if any of our proprietary information is disclosed, our business will suffer because our revenues depend upon our ability to license or commercialize our product candidates and any such events would significantly impair the value of such product candidates.
We face risks related to our government-funded programs; if BARDA/HHS or NIAID/HHS were to eliminate, reduce or delay funding from our contracts, this would have a significant negative impact on the programs associated with such funding and could have a significant negative impact on our revenues and cash flows.
We have completed work under a contract with BARDA/HHS for the development of RAPIVAB and have entered into contracts with BARDA/HHS and NIAID/HHS for the development of galidesivir as a treatment for diseases caused by RNA pathogens, including Marburg virus disease, Yellow Fever and Ebola virus disease. In contracting with these government agencies, we are subject to various U.S. Government contract requirements, including general clauses for a cost-reimbursement research and development contract, which may limit our reimbursement or, if we are found to be in violation, could result in contract termination. If the U.S. Government terminates any of its contracts with us for its convenience, or if we default by failing to perform in accordance with the contract schedule and terms, significant negative impact on our cash flows and operations could result.
34
U.S. Government contracts typically contain a number of extraordinary provisions that would not typically be found in commercial contracts and which may create a disadvantage and additional risks to us as compared to competitors that do not rely on U.S. Government contracts. These risks include the ability of the U.S. Government to unilaterally:
| • | terminate or reduce the scope of our contract with or without cause; | |
| • | interpret relevant regulations (federal acquisition regulation clauses); | |
| • | require performance under circumstances which may not be favorable to us; | |
| • | require an in process review where the U.S. Government will review the project and its options under the contract; | |
| • | control the timing and amount of funding, which impacts the development progress of our programs; and | |
| • | audit and object to our contract-related costs and fees, including allocated indirect costs. |
The U.S. Government may terminate its contracts with us either for its convenience or if we default by failing to perform in accordance with the contract schedule and terms. Termination for convenience provisions generally enable us to recover only our costs incurred or committed, and settlement expenses and profit on the work completed prior to termination. Termination does not permit these recoveries under default provisions. In the event of termination or upon expiration of a contract, the U.S. Government may dispute wind-down and termination costs and may question prior expenses under the contract and deny payment of those expenses. Should we choose to challenge the U.S. Government for denying certain payments under a contract, such a challenge could subject us to substantial additional expenses which we may or may not recover. Further, if the U.S. Government terminates its contracts with us for its convenience, or if we default by failing to perform in accordance with the contract schedule and terms, significant negative impact on our cash flows and operations could result.
As a U.S. Government contractor, we are required to comply with applicable laws, regulations and standards relating to our accounting practices and are subject to periodic audits and reviews. As part of any such audit or review, the U.S. Government may review the adequacy of, and our compliance with, our internal control systems and policies, including those relating to our purchasing, property, estimating, compensation and management information systems. Audits under the active BARDA/HHS and NIAID/HHS galidesivir contracts may occur at the election of the U.S. Government and have been concluded through fiscal 2015; all subsequent fiscal years are still open and auditable. Based on the results of its audits, the U.S. Government may adjust our contract-related costs and fees, including allocated indirect costs. This adjustment could impact the amount of revenues reported on a historic basis and could impact our cash flows under the contracts prospectively. In addition, in the event BARDA/HHS or NIAID/HHS determines that certain costs and fees were unallowable or determines that the allocated indirect cost rate was higher than the actual indirect cost rate, BARDA/HHS or NIAID/HHS would be entitled to recoup any overpayment from us as a result. In addition, if an audit or review uncovers any improper or illegal activity, we may be subject to civil and criminal penalties and administrative sanctions, including termination of our contracts, forfeiture of profits, suspension of payments, fines and suspension or prohibition from doing business with the U.S. Government. We could also suffer serious harm to our reputation if allegations of impropriety were made against us. In addition, under U.S. Government purchasing regulations, some of our costs may not be reimbursable or allowed under our contracts. Further, as a U.S. Government contractor, we are subject to an increased risk of investigations, criminal prosecution, civil fraud, whistleblower lawsuits and other legal actions and liabilities as compared to private sector commercial companies.
We face an inherent risk of liability in the event that the use or misuse of our products results in personal injury or death and our product liability insurance coverage may be insufficient.
If the use or misuse of peramivir, forodesine or any other regulatory body-approved products we or a partner may sell in the future harms people, we may be subject to costly and damaging product liability claims brought against us by consumers, healthcare providers, pharmaceutical companies, third-party payors or others. The use of our product candidates in clinical trials, including post marketing clinical studies, could also expose us to product liability claims. We cannot predict all of the possible harms or side effects that may result from the use of our products or the testing of product candidates and, therefore, the amount of insurance coverage we currently have may not be adequate to cover all liabilities or defense costs we might incur. A product liability claim or series of claims brought against us could give rise to a substantial liability that could exceed our resources. Even if claims are not successful, the costs of defending such claims and potential adverse publicity could be harmful to our business.
35
We face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials and will face even greater risks upon any commercialization by us of our product candidates. We have product liability insurance covering our clinical trials. Clinical trial and product liability insurance is becoming increasingly expensive. As a result, we may be unable to obtain sufficient insurance or increase our existing coverage at a reasonable cost to protect us against losses that could have a material adverse effect on our business. An individual may bring a product liability claim against us if one of our products or product candidates causes, or is claimed to have caused, an injury or is found to be unsuitable for consumer use. Any product liability claim brought against us, with or without merit, could result in:
| • | liabilities that substantially exceed our product liability insurance, which we would then be required to pay from other sources, if available; |
| • | an increase of our product liability insurance rates or the inability to maintain insurance coverage in the future on acceptable terms, or at all; | ||
| • | withdrawal of clinical trial volunteers or patients; | ||
| • | damage to our reputation and the reputation of our products, resulting in lower sales; | ||
| • | regulatory investigations that could require costly recalls or product modifications; | ||
| • | litigation costs; and | ||
| • | the diversion of management’s attention from managing our business. | ||
There are risks related to the potential government use or sale of our antivirals.
Government use or sale, in emergency situations or otherwise, of our antivirals—including peramivir for the treatment of influenza or galidesivir as a potential treatment for the COVID-19 novel coronavirus—may result in risks to us or our collaborative partners. There can be no assurance that government use of our antivirals (whether as indicated or outside of their current indications) will prove to be generally safe, well-tolerated and effective. Any government sale or use (on an emergency basis or otherwise) of our antivirals in any country may create liabilities for us or our partners.
We have entered into a contract with the CDC for the procurement of up to 50,000 doses of RAPIVAB (peramivir injection) over a five-year period. In addition, we are in active dialogue with NIAID, relevant U.S. public health authorities, and clinical investigators as they assess potential approaches to evaluate investigational antiviral drugs for treatment of COVID-19, with the goal of determining if galidesivir is effective against this strain, assessing whether galidesivir should be tested in new or existing clinical trials in patients with COVID-19, and expanding the current supply of the drug. There can be no assurance that we or our manufacturers will be able to fully meet the demand for such antivirals with respect to these or future arrangements. Further, we may not receive a favorable purchase price for future orders of our antivirals by governmental entities. Our competitors may develop products that could compete with or replace any antivirals selected for government sale or use. We may face competition in markets where we have no existing intellectual property protection or are unable to successfully enforce our intellectual property rights.
There can be no assurance that the non-U.S. partnerships that we have entered into for peramivir will result in any order for peramivir in those countries or that peramivir will be approved for any use or will achieve market approval in additional countries. There can be no assurance that galidesivir will be approved for use in any countries. In the event that any emergency use or market approval is granted in any country, there can be no assurance that any government order or commercialization of the applicable product or product candidate in such countries will be substantial or will be profitable to us.
There can be no assurance that the non-U.S. partnerships that we have entered into for peramivir will result in any order for peramivir in those countries or that peramivir will be approved for any use or will achieve market approval in additional countries. There can be no assurance that galidesivir will be approved for use in any countries. In the event that any emergency use or market approval is granted in any country, there can be no assurance that any government order or commercialization of the applicable product or product candidate in such countries will be substantial or will be profitable to us.
If we fail to reach milestones or to make annual minimum payments or otherwise breach our obligations under our license agreements, our licensors may terminate our agreements with them and seek additional remedies.
If we are unable or fail to meet payment obligations, performance milestones relating to the timing of regulatory filings, product supply obligations, post approval commitments, or development and commercial diligence obligations; are unable or fail to make milestone payments or material data use payments in accordance with applicable provisions; or fail to pay the minimum annual payments under any of our in-licenses relating to our products or product candidates, our licensors may terminate the applicable license or seek other available remedies. As a result, our development of the respective product candidate or commercialization of the product would cease.
Royalties and milestone payments from Shionogi under our license agreement with Shionogi (the "Shionogi Agreement") are required to be used by Royalty Sub to service its obligations under its PhaRMA Notes, and generally will not be available to us for other purposes unless and until Royalty Sub has repaid in full its obligations under the PhaRMA Notes.
In March 2011, our wholly-owned subsidiary Royalty Sub issued $30.0 million in aggregate principal amount of PhaRMA Notes. The PhaRMA Notes are secured principally by (i) certain royalty and milestone payments under the Shionogi Agreement, pursuant to which Shionogi licensed from us the rights to market peramivir in Japan and Taiwan, (ii) rights to certain payments under a Japanese yen/U.S. dollar Currency Hedge Agreement put into place by us in connection with the issuance of the PhaRMA Notes and (iii) the pledge by us of our equity interest in Royalty Sub. Payments from Shionogi to us on non-governmental sales under the Shionogi Agreement will generally not be available to us for other purposes unless and until Royalty Sub has repaid in full its obligations under the PhaRMA Notes. Accordingly, these funds have been and will continue to be required to be dedicated to Royalty Sub’s debt service and not available to us for product development or other purposes. Since September 1, 2014, payments from Shionogi have been insufficient for Royalty Sub to service its obligations under the PhaRMA Notes, resulting in a continuing event of default with respect to the PhaRMA Notes since that time. As a result of the continuing event of default, the holders of the PhaRMA Notes may be able to pursue acceleration of the PhaRMA Notes and foreclose on the collateral securing the PhaRMA Notes and our equity interest in Royalty Sub and may exercise other remedies available to them under the indenture or other documents related to the PhaRMA Notes. In such event, we may not realize the benefit of future royalty payments that might otherwise accrue to us following repayment of the PhaRMA Notes, we may incur legal costs and we might otherwise be adversely affected.
36
The PhaRMA Notes have a final legal maturity date of December 1, 2020, at which time the outstanding principal amount of the PhaRMA Notes, together with accrued and unpaid interest, will be due in full. The failure by Royalty Sub to repay in full the outstanding principal amount of the PhaRMA Notes, together with any accrued and unpaid interest, at the December 1, 2020 final maturity date would constitute an additional event of default under the PhaRMA Notes. We do not currently expect that Royalty Sub will be able to repay the PhaRMA Notes at final maturity. We cannot predict whether holders of PhaRMA Notes will seek to pursue any remedies as a result of the continuing event of default with respect to the PhaRMA Notes or at final maturity if Royalty Sub fails to pay the PhaRMA Notes in full at final maturity. The PhaRMA Notes are the obligation of Royalty Sub. As a result, we do not currently expect the continuing event of default on the PhaRMA Notes, or a failure by Royalty Sub to repay the PhaRMA Notes at final maturity, to have a significant impact on our future results of operations or cash flows. However, we cannot assure you that this will be the case or that we will not otherwise be adversely affected as a result the continuing event of default under the PhaRMA Notes or a failure by Royalty Sub to repay the PhaRMA Notes at maturity.
Because a continuing event of default exists under the PhaRMA Notes, the holders of the PhaRMA Notes may be able to pursue acceleration of the PhaRMA Notes and foreclose on the collateral securing the PhaRMA Notes and our equity interest in Royalty Sub. In addition, we do not currently expect that Royalty Sub will be able to repay the PhaRMA Notes at final maturity on December 1, 2020. As a result, we may not realize the benefit of future royalty payments that might otherwise accrue to us following repayment of the PhaRMA Notes and we could otherwise be adversely affected.
As Royalty Sub has been unable to service its obligations under the PhaRMA Notes and a continuing event of default exists under the PhaRMA Notes, the holders of the PhaRMA Notes may be able to pursue acceleration of the PhaRMA Notes and foreclose on the collateral securing the PhaRMA Notes and our equity interest in Royalty Sub and may exercise other remedies available to them under the indenture or other documents related to the PhaRMA Notes. In such event, we may not realize the benefit of future royalty payments that might otherwise accrue to us following repayment of the PhaRMA Notes, we may incur legal costs and we might otherwise be adversely affected. In addition, the PhaRMA Notes have a final legal maturity date of December 1, 2020, at which time the outstanding principal amount of the PhaRMA Notes, together with accrued and unpaid interest, will be due in full. The failure by Royalty Sub to repay in full the outstanding principal amount of the PhaRMA Notes, together with any accrued and unpaid interest, at the December 1, 2020 final maturity date would constitute an additional event of default under the PhaRMA Notes. We do not currently expect that Royalty Sub will be able to repay the PhaRMA Notes at final maturity. We cannot predict whether holders of PhaRMA Notes will seek to pursue any remedies as a result of the continuing event of default with respect to the PhaRMA Notes or at final maturity if Royalty Sub fails to pay the PhaRMA Notes in full at final maturity. The PhaRMA Notes are the obligation of Royalty Sub. As a result, we do not currently expect the continuing event of default on the PhaRMA Notes, or a failure by Royalty Sub to repay the PhaRMA Notes at final maturity, to have a significant impact on our future results of operations or cash flows. However, we cannot assure you that this will be the case or that we will not otherwise be adversely affected as a result the continuing event of default under the PhaRMA Notes or a failure by Royalty Sub to repay the PhaRMA Notes at maturity.
We may be required to pay premiums under the Currency Hedge Agreement entered into by us in connection with the issuance of the PhaRMA Notes. In addition, because our potential obligations under the foreign currency hedge are marked to market, we may experience additional quarterly volatility in our operating results and cash flows attributable to the Currency Hedge Agreement.
In connection with the issuance by Royalty Sub of the PhaRMA Notes, we entered into a Currency Hedge Agreement to hedge certain risks associated with changes in the value of the Japanese yen relative to the U.S. dollar. Under the foreign currency hedge agreement, we may be required to pay an annual premium in the amount of $2.0 million in May 2020. Such payment will be required if, on May 18, 2020, the spot rate of exchange for Japanese yen-U.S. dollars (determined in accordance with the Currency Hedge Agreement) is such that the U.S. dollar is worth 100 yen or less. We are required to mark to market our potential obligations under the currency hedge and post cash collateral, which may cause us to experience additional quarterly volatility in our operating results and cash flows as a result. Additionally, we may be required to pay significant premiums or a termination fee under the foreign currency hedge agreement entered into by us in connection with the issuance of the PhaRMA Notes. We are required to maintain a foreign currency hedge at 100 yen per dollar under the agreements governing the PhaRMA Notes.
Our Second Amended and Restated Senior Credit Facility contains restrictions that limit our flexibility in operating our business. We may be required to make a prepayment or repay the outstanding indebtedness earlier than we expect if a prepayment event or an event of default occurs, including a material adverse change with respect to us, which could have a material adverse effect on our business.
37
The Second Amended and Restated Senior Credit Facility contains various covenants that limit our ability to engage in specified types of transactions. These covenants limit our ability to, among other things:
| • | convey, sell, lease, license, transfer or otherwise dispose of certain parts of our business or property; | |
| • | change the nature of our business; |
| • | liquidate or dissolve; | |
| • | enter into certain change in control or acquisition transactions; | |
| • | incur or assume certain debt, including accessing additional tranches of debt under the Senior Credit Facility; | |
| • | grant certain types of liens on our assets; | |
| • | modify, liquidate or transfer assets in certain collateral accounts; | |
| • | pay dividends or make certain distributions to our stockholders; | |
| • | make certain investments; | |
| • | enter into material transactions with affiliates; and | |
| • | modify existing debt or collaboration arrangements. |
The restrictive covenants contained in the Second Amended and Restated Senior Credit Facility could cause us to be unable to pursue business opportunities that we or our stockholders may consider beneficial without the lender’s permission or without repaying all Second Amended and Restated Senior Credit Facility obligations.
A breach of any of these covenants could result in an event of default under the Second Amended and Restated Senior Credit Facility. An event of default will also occur if, among other things, a material adverse change in our business, operations or condition occurs, which could potentially include negative results in clinical trials, or a material impairment of the prospect of our repayment of any portion of the amounts we owe under the Second Amended and Restated Senior Credit Facility occurs. In the case of a continuing event of default under the agreement, the lender could elect to declare all amounts outstanding to be immediately due and payable, proceed against the collateral in which we granted to the lender a security interest under the Second Amended and Restated Senior Credit Facility, or otherwise exercise the rights of a secured creditor. Amounts outstanding under the Second Amended and Restated Senior Credit Facility are secured by substantially all of our assets and those of our subsidiaries, excluding certain specified assets but including proceeds from those assets.
Our actual or perceived failure to comply with European governmental regulations and other legal obligations related to privacy, data protection and information security could harm our business.
EU member states, Switzerland and other countries have adopted data protection laws and regulations, which impose significant compliance obligations. For example, the General Data Protection Regulation (“GDPR”) imposes strict requirements on controllers and processors of personal data, including special protections for “special category data,” which includes health, biometric and genetic information of data subjects located in the EU. Further, GDPR provides a broad right for EU member states to create supplemental national laws, for example relating to the processing of health, genetic and biometric data, which could further limit our ability to use and share such data or could cause our costs to increase and harm our business and financial condition. GDPR grants individuals the opportunity to object to the processing of their personal information, allows them to request deletion of personal information in certain circumstances, and provides the individual with an express right to seek legal remedies in the event the individual believes his or her rights have been violated. Further, the GDPR imposes strict rules on the transfer of personal data out of the EU to the United States or other regions that have not been deemed to offer “adequate” privacy protections.
Failure to comply with the requirements of the GDPR and the related national data protection laws of the EU member states, which may deviate slightly from the GDPR, may result in significant fines of up to 4% of global revenues, or €20.0 million, whichever is greater, and in addition to such fines, our failure to comply with the requirements of GDPR may subject us to litigation and/or adverse publicity, which could have material adverse effect on our reputation and business. As a result of the implementation of the GDPR, we are required to put in place additional mechanisms to ensure compliance with the new data protection rules. For example, the GDPR requires us to make more detailed disclosures to data subjects, requires disclosure of the legal basis on which we can process personal data, makes it harder for us to obtain valid consent for processing, will require the appointment of a data protection officer where sensitive personal data (i.e., health data) is processed on a large scale, introduces mandatory data breach notification throughout the EU, imposes additional obligations on us when we are contracting with service providers and requires us to adopt appropriate privacy governance including policies, procedures, training and data audit.
38
We are subject to the supervision of local data protection authorities in those jurisdictions where we undertake clinical trials. We depend on a number of third parties in relation to the provision of our services, a number of which process personal data of EU individuals on our behalf. With each such provider we are required to enter into contractual arrangements under which they are contractually obligated to only process personal data according to our instructions, and conduct diligence to ensure that they have sufficient technical and organizational security measures in place.
We are also subject to evolving European privacy laws on electronic marketing and cookies. The EU is in the process of replacing the e-Privacy Directive (2002/58/EC) with a new set of rules taking the form of a regulation that will be directly implemented in the laws of each European member state. While this e-Privacy Regulation was originally intended to be adopted on May 25, 2018, it is still going through the European legislative process and the timing of its adoption remains unclear.
The United Kingdom’s decision to withdraw from the EU could result in increased regulatory and legal complexity, which may make it more difficult for us to do business in Europe and impose additional challenges in securing regulatory approval of our product candidates in Europe.
The United Kingdom’s exit from the EU, or Brexit, has caused political and economic uncertainty, including in the regulatory framework applicable to our operations and product candidates, and this uncertainty may persist for years. Brexit could, among other outcomes, disrupt the free movement of goods, services and people between the United Kingdom and the EU, and result in increased legal and regulatory complexities, as well as potential higher costs of conducting business in Europe. For instance, preparations for Brexit have resulted in the decision to move the EMA from the United Kingdom to the Netherlands. This transition may cause disruption or delays in granting clinical trial authorization or opinions for marketing authorization, disruption of importation and export of active substance and other components of new drug formulations, and disruption of the supply chain for clinical trial product and final authorized formulations.
The cumulative effects of the disruption to the regulatory framework may add considerably to the development lead time to marketing authorization and commercialization of products in the EU and/or the United Kingdom. It is possible that there will be increased regulatory complexities, which can disrupt the timing of our clinical trials and regulatory approvals. In addition, changes in, and legal uncertainty with regard to, national and international laws and regulations may present difficulties for our clinical and regulatory strategy. Any delay in obtaining, or an inability to obtain, any marketing approvals, as a result of Brexit or otherwise, would prevent us from commercializing our product candidates in the United Kingdom and/or the EU and restrict our ability to generate revenues and achieve and sustain profitability.
In addition, as a result of Brexit, other European countries may seek to conduct referenda with respect to their continuing membership with the EU. Given these possibilities and others we may not anticipate, as well as the absence of comparable precedent, it is unclear what financial, regulatory and legal implications the withdrawal of the United Kingdom from the EU would have and how such withdrawal would affect us, and the full extent to which our business could be adversely affected.
Natural disasters, epidemic or pandemic disease outbreaks, trade wars, political unrest or other events could disrupt our business or operations or those of our development partners, manufacturers, regulators or other third parties with whom we conduct business now or in the future.
A wide variety of events beyond our control, including natural disasters, epidemic or pandemic disease outbreaks (such as the recent novel coronavirus outbreak), trade wars, political unrest or other events could disrupt our business or operations or those of our development partners (such as Torii), manufacturers, regulatory authorities, or other third parties with whom we conduct business. These events may cause businesses and government agencies to be shut down, supply chains to be interrupted, slowed, or rendered inoperable, and individuals to become ill, quarantined, or otherwise unable to work and/or travel due to health reasons or governmental restrictions. If our operations or those of third parties with whom we have business are impaired or curtailed as a result of these events, the development and commercialization of our products and product candidates could be impaired or halted, which could have a material adverse impact on our business. For example, the recent novel coronavirus outbreak has resulted in widespread illness, quarantine, and interruption of business in parts of Asia. If these interruptions impair our or Torii’s ability to perform under the Torii Agreement, or our or our partners’ regulatory interactions in Japan, including with respect to the pending Japanese NDA with respect to berotralstat for the treatment of HAE, then the timing and success of our development and commercialization of berotralstat in Japan could be severely impacted.
We are subject to legal proceedings, which could result in losses or unexpected expenditure of time and resources.
From time to time, we may be involved in disputes, called upon to initiate legal proceedings or to defend ourselves in such legal proceedings relating to our business. Due to the inherent uncertainties in legal proceedings, we cannot accurately predict the ultimate outcome of any such proceedings. An unfavorable outcome in any such proceedings could have an adverse impact on our business, financial condition and results of operations. If our stock price is volatile, we may become involved in securities class action lawsuits in the future. Any current or future dispute resolution or legal proceeding, regardless of the merits of any such proceeding, could result in substantial costs and a diversion of management’s attention and resources that are needed to successfully run our business.
39
Insurance coverage is increasingly more costly and difficult to obtain or maintain.
While we currently have insurance for our business, property, directors and officers, and our products, insurance is increasingly more costly and narrower in scope, and we may be required to assume more risk in the future. If we are subject to claims or suffer a loss or damage in excess of our insurance coverage, we will be required to bear any loss in excess of our insurance limits. If we are subject to claims or suffer a loss or damage that is outside of our insurance coverage, we may incur significant uninsured costs associated with loss or damage that could have an adverse effect on our operations and financial position. Furthermore, any claims made on our insurance policies may impact our ability to obtain or maintain insurance coverage at reasonable costs or at all.
If our facility incurs damage or power is lost for a significant length of time, our business will suffer.
We store clinical and stability samples at our facility that could be damaged if our facility incurs physical damage or in the event of an extended power failure. We have backup power systems in addition to backup generators to maintain power to all critical functions, but any loss of these samples could result in significant delays in our drug development process.
In addition, we store most of our preclinical and clinical data at our facilities. Duplicate copies of most critical data are secured off-site. Any significant degradation or failure of our computer systems could cause us to inaccurately calculate or lose our data. Loss of data could result in significant delays in our drug development process and any system failure could harm our business and operations.
A significant disruption in our information technology systems or a cyber-security breach could adversely affect our business.
We are increasingly dependent on information technology systems to operate our business. In addition, the FDA and comparable foreign regulatory authorities regulate, among other things, the record keeping and storage of data pertaining to potential pharmaceutical products. We currently store most of our preclinical research data, our clinical data and our manufacturing data at our facility. While we do store duplicate copies of most of our clinical data offsite and a significant portion of our data is included in regular backups of our systems, we could lose important data if our facility incurs damage, or if our vendor data systems fail, suffer damage or are destroyed.
Like other companies in our industry, our networks and infrastructure may be vulnerable to cyber-attacks or intrusions, including by computer hackers, foreign governments, foreign companies or competitors, or may be breached by employee error, malfeasance or other disruption. A breakdown, invasion, corruption, destruction or interruption of critical information technology systems could negatively impact operations. If our systems are damaged, fail to function properly or otherwise become unavailable, we may incur substantial costs to repair or replace them, and we may experience loss of critical data and interruptions or delays in our ability to perform critical functions, which could adversely affect our business, financial condition or results of operations. Any compromise of our data security could also result in a violation of applicable privacy and other laws, significant legal and financial exposure, damage to our reputation, loss or misuse of the information and a loss of confidence in our data security measures, which could harm our business. There can be no assurance that our efforts to protect our data and information technology systems will prevent breakdowns or breaches in our systems, or those of third parties with which we do business, and any such events could adversely affect our business.
If we fail to retain our existing key personnel or fail to attract and retain additional key personnel, the development of our product candidates and commercialization of our products and the related expansion of our business will be delayed or stopped.
We are highly dependent upon our senior management and scientific team, the unexpected loss of whose services might impede the achievement of our development and commercial objectives. Competition for key personnel with the experience that we require is intense and is expected to continue to increase. Our inability to attract and retain the required number of skilled and experienced management, commercial, operational and scientific personnel will harm our business because we rely upon these personnel for many critical functions of our business.
If because of our use of hazardous materials, we violate any environmental controls or regulations that apply to such materials, we may incur substantial costs and expenses in our remediation efforts.
Our research and development involves the controlled use of hazardous materials, chemicals and various radioactive compounds. We are subject to federal, state and local laws and regulations governing the use, storage, handling and disposal of these materials and some waste products. Accidental contamination or injury from these materials could occur. In the event of an accident, we could be liable for any damages that result and any liabilities could exceed our resources. Compliance with environmental laws and regulations or a violation of such environmental laws and regulations could require us to incur substantial unexpected costs, which would materially and adversely affect our results of operations.
40
Risks relating to investing in our common stock
Our existing principal stockholders hold a substantial amount of our common stock and may be able to influence significant corporate decisions, which may conflict with the interest of other stockholders.
Several of our stockholders own greater than 5% of our outstanding common stock. Our top ten stockholders own more than 50% of BioCryst and can individually, and as a group, influence our operations based upon their concentrated ownership. These stockholders, if they act together, may be able to influence the outcome of matters requiring approval of the stockholders, including the election of our directors and other corporate actions.
Our stock price has been, and is likely to continue to be, highly volatile, which could cause the value of an investment in our common stock to decline significantly.
The market prices for securities of biotechnology companies in general have been highly volatile and may continue to be highly volatile in the future. Moreover, our stock price has fluctuated frequently, and these fluctuations are often not related to our financial results. For the twelve months ended December 31, 2019, the 52-week range of the market price of our stock was from $1.38 to $9.95 per share. The following factors, in addition to other risk factors described in this section, may have a significant impact on the market price of our common stock:
| • | announcements of technological innovations or new products by us or our competitors; | |
| • | developments or disputes concerning patents or proprietary rights; | |
| • | additional dilution through sales of our common stock or other derivative securities; |
| • | status of new or existing licensing or collaborative agreements and government contracts; | |
| • | announcements relating to the status of our programs; | |
| • | developments and announcements regarding new and virulent strains of influenza; | |
| • | we or our partners achieving or failing to achieve development milestones; | |
| • | publicity regarding actual or potential medical results relating to products under development by us or our competitors; | |
| • | publicity regarding certain public health concerns for which we are or may be developing treatments; | |
| • | regulatory developments in both the United States and foreign countries; | |
| • | public concern as to the safety of pharmaceutical products; | |
| • | actual or anticipated fluctuations in our operating results; | |
| • | changes in financial estimates or recommendations by securities analysts; | |
| • | changes in the structure of healthcare payment systems, including developments in price control legislation; | |
| • | announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures or capital commitments; | |
| • | additions or departures of key personnel or members of our board of directors; | |
| • | purchases or sales of substantial amounts of our stock by existing stockholders, including officers or directors; | |
| • | economic and other external factors or other disasters or crises; and | |
| • | period-to-period fluctuations in our financial results. | |
41
Future sales and issuances of securities may dilute the ownership interests of our current stockholders and cause our stock price to decline.
Future sales of our common stock by current stockholders into the public market could cause the market price of our stock to fall. As of January 31, 2020, there were 154,191,951 shares of our common stock outstanding. We may from time to time issue securities in relation to a license arrangement, collaboration, merger or acquisition. We may also sell, for our own account, shares of common stock or other equity securities, from time to time at prices and on terms to be determined at the time of sale.
As of January 31, 2020, there were 21,044,021 stock options outstanding and 974,781 shares available for issuance under our Amended and Restated Stock Incentive Plan, 1,469,224 stock options outstanding and 30,776 shares available for issuance under our Inducement Equity Incentive Plan and 9,484 shares available for issuance under our Employee Stock Purchase Plan. In addition, we could also make equity grants outside of our Stock Incentive Plan or Inducement Equity Incentive Plan. The shares underlying existing stock options, restricted stock units and possible future stock options, stock appreciation rights and stock awards have been registered pursuant to registration statements on Form S-8.
If some or all of such shares are sold or otherwise issued into the public market over a short period of time, our current stockholders’ ownership interests may be diluted and the value of all publicly traded shares is likely to decline, as the market may not be able to absorb those shares at then-current market prices. Additionally, such sales and issuances may make it more difficult for us to sell equity securities or equity-related securities in the future at a time and price that our management deems acceptable, or at all.
In March 2017, we entered into a Registration Rights Agreement with entities affiliated with Baker Bros. Advisors LP (the “Baker Entities”) to provide that, if requested, we will register the shares of our common stock beneficially owned by the Baker Entities for resale under the Securities Act. Our registration obligations pursuant to the Registration Rights Agreement cover all shares then held or thereafter acquired by the Baker Entities, for up to ten years, and include our obligation to facilitate certain underwritten public offerings of our common stock by the Baker Entities in the future. On May 10, 2017, we filed a registration statement on Form S-3 with respect to 11,710,951 shares of common stock held by the Baker Entities. Subsequently, on November 21, 2019, certain of the Baker Entities acquired pre-funded warrants to purchase 11,764,706 shares of our common stock at a price of $1.69 per warrant. Each warrant has an exercise price of $0.01 per share. If the Baker Entities, by exercising their underwriting rights or otherwise, sell a large number of our shares, or the market perceives that the Baker Entities intend to sell a large number of our shares, this could adversely affect the market price of our common stock.
We have anti-takeover provisions in our corporate charter documents that may result in outcomes with which you do not agree.
Our board of directors has the authority to issue up to 4,800,000 shares of undesignated preferred stock and to determine the rights, preferences, privileges and restrictions of those shares without further vote or action by our stockholders. The rights of the holders of any preferred stock that may be issued in the future may adversely affect the rights of the holders of common stock. The issuance of preferred stock could make it more difficult for third parties to acquire a majority of our outstanding voting stock.
In addition, our certificate of incorporation provides for staggered terms for the members of the board of directors and supermajority approval of the removal of any member of the board of directors and prevents our stockholders from acting by written consent. Our certificate also requires supermajority approval of any amendment of these provisions. These provisions and other provisions of our by-laws and of Delaware law applicable to us could delay or make more difficult a merger, tender offer or proxy contest involving us.
We have never paid dividends on our common stock and do not anticipate doing so in the foreseeable future.
We have never paid cash dividends on our stock. We currently intend to retain all future earnings, if any, for use in the operation of our business. Accordingly, we do not anticipate paying cash dividends on our common stock in the foreseeable future.
Information Regarding Forward-Looking Statements
This filing contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, which are subject to the “safe harbor” created in Section 21E. All statements other than statements of historical facts contained in this filing are forward-looking statements. These forward-looking statements can generally be identified by the use of words such as “may,” “will,” “intends,” “plans,” “believes,” “anticipates,” “expects,” “estimates,” “predicts,” “potential,” the negative of these words or similar expressions. Statements that describe our future plans, strategies, intentions, expectations, objectives, goals or prospects are also forward-looking statements. Discussions containing these forward-looking statements are principally contained in “Business,” “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” as well as any amendments we make to those sections in filings with the SEC. These forward-looking statements include, but are not limited to, statements about:
| • | the preclinical development, clinical development, commercialization, or post-marketing studies of our product candidates and products, including our acute and prophylactic HAE programs, BCX9930, BCX9250, peramivir, galidesivir, and early stage discovery programs; |
42
| • | the potential funding from our contracts with NIAID/HHS and BARDA/HHS for the development of galidesivir; |
| • | the potential for government stockpiling orders of peramivir and galidesivir, additional regulatory approvals of peramivir, or milestones, royalties or profit from sales of peramivir by us or our partners; |
| • | the potential use of peramivir as a treatment for H1N1, H5N1, and H7N9 or other strains of influenza; |
| • | the implementation of our business model, strategic plans for our business, products, product candidates and technology; | |
| • | our ability to establish and maintain collaborations or out-license rights to our product candidates; |
| • | the outcome, cost and timing of any resolution of disputes and legal proceedings, including but not limited to the dispute with our partner SUL; |
| • | plans, programs, progress and potential success of our collaborations, including SUL for peramivir, Mundipharma for mundesine, Torii for BCX7353 in Japan and Shionogi and Green Cross for peramivir in their territories; |
| • | our and MDCP’s ability to satisfy obligations under our Second Amended and Restated Senior Credit Facility; |
| • | Royalty Sub’s ability to service its payment obligations in respect of the PhaRMA Notes; |
| • | the Currency Hedge Agreement entered into by us in connection with the issuance by Royalty Sub of the PhaRMA Notes; | |
| • | the scope of protection we are able to establish and maintain for intellectual property rights covering our product candidates and technology; |
| • | our ability to operate our business without infringing the intellectual property rights of others; |
| • | estimates of our expenses, revenues, capital requirements, annual cash utilization, and our needs for additional financing; | |
| • | our ability to continue as a going concern; |
| • | the timing or likelihood of regulatory filings or regulatory agreements, deferrals, and approvals; |
| • | the timing or likelihood of entering into a U.S. government stockpile order and our ability to execute any such order; |
| • | our ability to raise additional capital to fund our operations or repay our recourse debt obligations; |
| • | our ability to comply with the covenants as set forth in the agreements governing our debt obligations; |
| • | our financial performance; |
| • | the timing and success of our anticipated commercialization of BCX7353 in the U.S. and elsewhere; and |
| • | competitive companies, technologies and our industry. |
These statements relate to future events or to our future financial performance and involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by these forward-looking statements. Factors that may cause actual results to differ materially from current expectations include, among other things, those listed under “Risk Factors.” Any forward-looking statement reflects our current views with respect to future events and is subject to these and other risks, uncertainties and assumptions relating to our operations, results of operations, industry and future growth. Except as required by law, we assume no obligation to update or revise these forward-looking statements for any reason, even if new information becomes available in the future.
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
We lease property in both Durham, North Carolina and Birmingham, Alabama. Our headquarters, including our clinical and regulatory operations, are based in Durham, while our principal research facility is located in Birmingham. We currently lease approximately 33,000 square feet in Durham through December 31, 2020, sublease approximately 7,000 square feet in Durham through January 26, 2021 and lease approximately 34,000 square feet in Birmingham through October 31, 2026. We believe that our facilities are adequate for our current and planned future operations.
| ITEM 3. | LEGAL PROCEEDINGS |
None.
| ITEM 4. | MINE SAFETY DISCLOSURES |
Not applicable.
43
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock trades on the NASDAQ Global Select Market under the symbol BCRX.
Holders
As of January 31, 2020, there were approximately 145 holders of record of our common stock.
Dividends
We have never paid cash dividends and do not anticipate paying cash dividends in the foreseeable future.
Stock Performance Graph
This performance graph is not “soliciting material,” is not deemed filed with the SEC and is not to be incorporated by reference in any filing by us under the Securities Act or the Exchange Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing. The stock price performance shown on the graph is not necessarily indicative of future price performance.
PERFORMANCE GRAPH FOR BIOCRYST
Indexed Comparison Since 2014
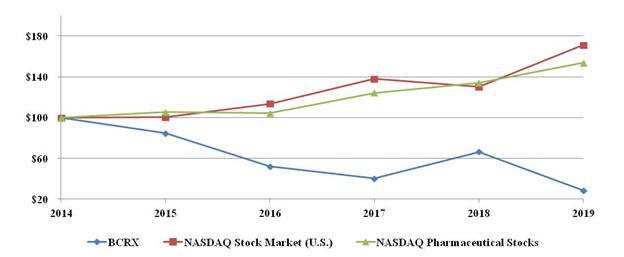
| Beginning Investment 12/31/14 | Investment at 12/31/15 | Investment at 12/31/16 | Investment at 12/31/17 | Investment at 12/31/18 | Investment at 12/31/19 | |||||||||||||||||||
| BioCryst Pharmaceuticals, Inc. | $ | 100.00 | $ | 84.87 | $ | 52.06 | $ | 40.38 | $ | 66.37 | $ | 28.37 | ||||||||||||
| NASDAQ Stock Market (U.S.) | 100.00 | 100.48 | 113.55 | 137.83 | 130.33 | 170.96 | ||||||||||||||||||
| NASDAQ Pharmaceutical Stocks | 100.00 | 105.43 | 104.29 | 124.23 | 134.11 | 153.57 | ||||||||||||||||||
The above graph measures the change in a $100 investment in our common stock based on its closing price of $12.16 on December 31, 2014 and its year-end closing price thereafter. Our relative performance is then compared with the CRSP Total Return Indexes for the NASDAQ Stock Market (U.S.) and NASDAQ Pharmaceutical Stocks.
Recent Sales of Unregistered Securities: None.
Issuer Purchases of Equity Securities
There were no repurchases of our common stock or shares surrendered to satisfy tax obligations during the fourth quarter of 2019.
44
| ITEM 6. | SELECTED FINANCIAL DATA |
The selected Statement of Operations Data and Balance Sheet data with respect to the years ended December 31, 2019, 2018, 2017, 2016 and 2015 set forth below are derived from our consolidated financial statements. The selected financial data set forth below should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” contained in Item 7 below and our consolidated financial statements and the notes thereto appended to this annual report.
| Years Ended December 31, | ||||||||||||||||||||
| 2019 | 2018 | 2017 | 2016 | 2015 | ||||||||||||||||
| (In thousands, except per share amounts) | ||||||||||||||||||||
| Statement of Operations Data: | ||||||||||||||||||||
| Total revenues | $ | 48,835 | $ | 20,653 | $ | 25,186 | $ | 26,353 | $ | 48,257 | ||||||||||
| Cost of product sold | 3,726 | - | 1,142 | 2,297 | 1,368 | |||||||||||||||
| Research and development expenses | 107,068 | 84,888 | 66,962 | 61,008 | 72,758 | |||||||||||||||
| Selling, general and administrative expenses | 37,121 | 29,514 | 13,933 | 11,253 | 13,047 | |||||||||||||||
| Royalty expense | 375 | 471 | 560 | 402 | 528 | |||||||||||||||
| Loss from operations | (99,455 | ) | (94,220 | ) | (57,411 | ) | (48,607 | ) | (39,444 | ) | ||||||||||
| Net loss | (108,897 | ) | (101,253 | ) | (65,782 | ) | (55,144 | ) | (43,019 | ) | ||||||||||
| Basic and diluted net loss per share | $ | (0.94 | ) | $ | (0.98 | ) | $ | (0.78 | ) | $ | (0.75 | ) | $ | (0.59 | ) | |||||
| Weighted average shares outstanding | 115,600 | 103,185 | 84,451 | 73,699 | 72,901 | |||||||||||||||
| As of December 31, | ||||||||||||||||||||
| 2019 | 2018 | 2017 | 2016 | 2015 | ||||||||||||||||
| (In thousands) | ||||||||||||||||||||
| Balance Sheet Data: | ||||||||||||||||||||
| Cash, cash equivalents and investments | $ | 137,777 | $ | 128,387 | $ | 158,978 | $ | 65,122 | $ | 100,858 | ||||||||||
| Receivables | 22,146 | 4,293 | 6,117 | 8,768 | 6,243 | |||||||||||||||
| Inventory | - | 1,649 | - | 500 | 1,612 | |||||||||||||||
| Total assets | 175,282 | 146,841 | 178,259 | 89,847 | 122,359 | |||||||||||||||
| Long-term deferred revenue | - | - | - | 8,184 | 9,674 | |||||||||||||||
| Non-recourse notes payable | 29,561 | 29,121 | 28,682 | 28,243 | 27,804 | |||||||||||||||
| Senior credit facility | 50,309 | 29,952 | 23,214 | 22,777 | - | |||||||||||||||
| Accumulated deficit | (840,628 | ) | (731,969 | ) | (631,843 | ) | (566,061 | ) | (510,917 | ) | ||||||||||
| Total stockholders’ equity | 38,252 | 49,235 | 83,767 | 1,578 | 47,724 | |||||||||||||||
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
This Annual Report on Form 10-K contains certain statements of a forward-looking nature relating to future events or the future financial performance of BioCryst. Such statements are only predictions and the actual events or results may differ materially from the results discussed in the forward-looking statements. Factors that could cause or contribute to such differences include those discussed below and elsewhere in this report, as well as those discussed in other filings made by BioCryst with the Securities and Exchange Commission.
The following Management’s Discussion and Analysis (“MD&A”) is intended to help the reader understand our results of operations and financial condition. MD&A is provided as a supplement to, and should be read in conjunction with, our audited financial statements and the accompanying notes to the financial statements and other disclosures included in this Annual Report on Form 10-K (including the disclosures under “Item 1A. Risk Factors”).
Cautionary Statement
The discussion herein contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), which are subject to the “safe harbor” created in Section 21E. Forward looking statements regarding our financial condition and our results of operations that are based upon our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted within the United States (“U.S. GAAP”), as well as projections for the future. The preparation of these financial statements requires our management to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent assets and liabilities. We evaluate our estimates on an ongoing basis. Our estimates are based on historical experience and on various other assumptions that are believed to be reasonable under the circumstances. The results of our estimates form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources.
45
We operate in a highly competitive environment that involves a number of risks, some of which are beyond our control. We are subject to risks common to biotechnology and biopharmaceutical companies, including risks inherent in our drug discovery, drug development and commercialization efforts, clinical trials, uncertainty of regulatory actions and marketing approvals, reliance on collaborative partners, enforcement of patent and proprietary rights, the need for future capital, competition associated with products, potential competition associated with our product candidates and retention of key employees. In order for any of our product candidates to be commercialized, it will be necessary for us, or our collaborative partners, to conduct clinical trials, demonstrate efficacy and safety of the product candidate to the satisfaction of regulatory authorities, obtain marketing approval, enter into manufacturing, distribution and marketing arrangements, and obtain market acceptance and adequate reimbursement from government and private insurers. We cannot provide assurance that we will generate significant revenues or achieve and sustain profitability in the future. In addition, we can provide no assurance that we will have sufficient funding to meet our future capital requirements. Statements contained in Management’s Discussion and Analysis of Financial Condition and Results of Operations and elsewhere in this report which are not historical facts are, or may constitute, forward-looking statements. Forward-looking statements involve known and unknown risks that could cause our actual results to differ materially from expected results. The most significant known risks are discussed in the section entitled “Risk Factors.” Although we believe the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. We caution you not to place undue reliance on any forward-looking statements.
Our revenues are difficult to predict and depend on numerous factors, including the prevalence and severity of influenza in regions for which peramivir has received regulatory approval, seasonality of influenza, commercialization efforts and resources dedicated to our products by our collaborative partners, ongoing discussions with government agencies regarding future peramivir and/or galidesivir development and stockpiling procurement, as well as entering into, or modifying, licensing agreements for our product candidates. Furthermore, revenues related to our collaborative development activities are dependent upon the progress toward and the achievement of developmental milestones by us or our collaborative partners.
Our operating expenses are also difficult to predict and depend on several factors, including research and development expenses (and whether these expenses are reimbursable under government contracts), drug manufacturing, and clinical research activities, the ongoing requirements of our development programs, and the availability of capital and direction from regulatory agencies, which are difficult to predict. Management may be able to control the timing and level of research and development and general and administrative expenses, but many of these expenditures will occur irrespective of our actions due to contractually committed activities and/or payments.
As a result of these factors, we believe that period to period comparisons are not necessarily meaningful and you should not rely on them as an indication of future performance. Due to all of the foregoing factors, it is possible that our operating results will be below the expectations of market analysts and investors. In such event, the prevailing market price of our common stock could be materially adversely affected.
Overview
We are a biotechnology company that discovers novel, oral, small-molecule medicines. We focus on oral treatments for rare diseases in which significant unmet medical needs exist and an enzyme plays the key role in the biological pathway of the disease. We integrate the disciplines of biology, crystallography, medicinal chemistry and computer modeling to discover and develop small molecule pharmaceuticals through the process known as structure-guided drug design.
Critical Accounting Policies and Estimates
The accompanying discussion and analysis of our financial condition and results of operations are based upon our consolidated financial statements and the related disclosures, which have been prepared in accordance with U.S. GAAP. The preparation of these consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenues, expenses and related disclosure of contingent assets and liabilities. We evaluate our estimates, judgments and the policies underlying these estimates on a periodic basis, as situations change, and regularly discuss financial events, policies, and issues with members of our audit committee and our independent registered public accounting firm. We routinely evaluate our estimates and policies regarding revenue recognition, administration, inventory and manufacturing, taxes, stock-based compensation, research and development, consulting and other expenses and any associated liabilities.
46
Recent Corporate Highlights
Berotralstat (BCX7353)
Berotralstat is a second generation HAE compound and our lead molecule that is being developed as a once-daily oral therapy for the prevention of HAE attacks. We successfully completed our pivotal Phase 3 clinical trial, APeX-2, and reported 48-week data from our ongoing long-term safety clinical trial, APeX-S, in 2019. Based on the data from our clinical program, including APeX-2 and APeX-S, we submitted a new drug application to the FDA in December 2019 for approval of oral, once daily berotralstat for the prevention of HAE attacks. In February 2020, the FDA notified us that they had accepted and filed our NDA for review and that our Prescription Drug User Fee Act (“PDUFA”) date for the NDA is December 3, 2020. In the NDA filing acceptance letter, the FDA stated that they are not currently planning to hold an advisory committee meeting to discuss the NDA.
In addition, we have completed APeX-J, a clinical trial of berotralstat for the prevention of HAE attacks designed to support Japanese marketing authorization in conjunction with our other berotralstat clinical trials. On February 3, 2020, we announced we had submitted a new drug application (“JNDA”) to the Japanese Pharmaceuticals and Medical Devices Agency (“PDMA”) for approval of oral, once daily berotralstat for the prevention of HAE attacks. We expect approval of berotralstat in Japan in the second half of 2020.
In anticipation of a commercial launch of berotralstat, we are in the process of developing our business infrastructure, personnel, partnerships, and marketing strategies to position berotralstat for success in the commercial market, which we anticipate—based on proprietary market research, including analyses of HAE prevalence in the U.S. and market research studies with HAE patients, physicians, and payors in the U.S.—has the potential to reach a global peak of over $500 million in annual sales. These expectations are subject to numerous risks and uncertainties that may cause our actual results, performance or achievements to be materially different. There can be no assurance that regulatory approval of berotralstat will be granted in a timely fashion or at all, that our commercialization methods and strategies will succeed, or that the market for berotralstat will develop in line with our current expectations. See the “Risk Factors” section of this Annual Report on Form 10-K, including the information under “Risk Factors—We do not have a great deal of experience in commercializing our products or technologies, and our future revenue generation is uncertain” for further discussion of these risks.
Torii Collaboration: On November 5, 2019, we announced we had entered into a Commercialization and License Agreement (the “Torii Agreement”) with Torii Pharmaceutical Co., Ltd., a corporation organized under the laws of Japan (“Torii”), granting Torii the exclusive right to commercialize berotralstat for the prevention of HAE attacks, in Japan.
Under the Torii Agreement, we received an upfront, non-refundable payment of $22.0 million. We realized $20.1 million of revenue in the fourth quarter of 2019 associated with the upfront payment and expect to recognize the remaining $1.9 million in fiscal 2020 once we complete performance obligations under the agreement. We may also be eligible to receive an additional milestone payment of either $20.0 million if the PMDA grants regulatory approval on or before December 31, 2020, or $15.0 million if regulatory approval is granted on or before December 31, 2021. In either case, the regulatory milestone payment is contingent upon receipt of a reimbursement price approval from Japan’s National Health Insurance system in excess of the threshold specified in the Torii Agreement.
In addition, we will be entitled under the Agreement to receive tiered royalty payments based on the amount of annual net sales of berotralstat in Japan during each calendar year. If berotralstat maintains its Sakigake designation during the PMDA review, the tiered royalty rate will range from 20% to 40% of net sales, otherwise, the tiered royalty rate will range from 15% to 35% of net sales. Torii’s royalty payment obligations are subject to customary reductions in certain circumstances, but may not be reduced by more than 50% of the amount that otherwise would have been payable to us in the applicable calendar quarter. Torii’s royalty payment obligations commence upon the first commercial sale of berotralstat in Japan and expire upon the later of (i) the tenth anniversary of the date of first commercial sale of berotralstat in Japan, (ii) the expiration of our patents covering berotralstat, and (iii) the expiration of regulatory exclusivity for berotralstat in Japan. We will be responsible for supplying Torii with its required amounts of berotralstat. The activities of the parties pursuant to the Torii Agreement will be overseen by a Joint Steering Committee, to be composed of an equal number of representatives from each party to coordinate the development and commercialization of berotralstat in Japan.
APeX-2 Phase 3 Trial: APeX-2 is a Phase 3 double-blinded, placebo-controlled, three-arm clinical trial evaluating two dose levels of berotralstat administered orally once-daily as a preventive treatment to reduce the frequency of attacks in patients with HAE. APeX-2 tested once-daily berotralstat at 110 mg and 150 mg for prevention of angioedema attacks. The trial enrolled patients with Type I and II HAE in the United States, Canada and Europe. The primary efficacy endpoint of APeX-2 is the rate of angioedema attacks over 24 weeks of study drug administration. The trial enrolled and randomized 121 patients. The APeX-2 trial has been amended to extend the duration of dosing to monitor the long-term safety of the trial. Patients may continue in the trial on open-label berotralstat.
47
On May 21, 2019, we announced our Phase 3 APeX-2 trial of oral, once-daily berotralstat for the prevention of HAE attacks achieved its primary endpoint for both dose levels (110 mg and 150 mg), with the 150 mg dose reducing the attack rate in HAE patients by 44 percent (p<0.001) compared to placebo. Fifty percent of patients receiving 150 mg berotralstat in APeX-2 had a ≥ 70 percent reduction in their HAE attack rate compared to baseline, compared to 15 percent of placebo patients (p=0.002). Of 108 patients who completed 24 weeks of study drug treatment, 100 percent continued into the ongoing 48-week extension phase of the trial. Both the 110 mg and 150 mg dose levels of oral, once-daily berotralstat were generally safe and well-tolerated. No drug-related serious adverse events were reported.
APeX-S Long-term Safety Trial: APeX-S is an open label, long-term safety trial evaluating two dose levels (110 mg and 150 mg) of berotralstat administered orally once-daily as a preventive treatment in patients with Type I and II HAE. The APeX-S trial has been amended to extend the duration of dosing through 240 weeks to monitor the safety and effectiveness of long term treatment with berotralstat.
On November 6, 2019, we announced 48-week data from APeX-S showing that APeX-S patients taking 150 mg of berotralstat had similar attack control as those in APeX-2. Patients completing 48 weeks of treatment on 150 mg of berotralstat (n=73) had a median attack rate of zero attacks per month in six of the 12 months, including month 12 (week 48). The integrated 48-week analysis across both APeX-2 and APeX-S showed no new safety findings. Berotralstat was safe and generally well tolerated in a total of 342 patients with a total of 232 patient-years of daily oral dosing. The most common adverse event was the common cold, which occurred with similar frequency in berotralstat and placebo patients. Gastrointestinal events led to discontinuation of berotralstat in three percent of patients. Drug-related serious adverse events occurred in three of 342 subjects (0.9%) and resolved after stopping or interrupting BCX7353 dosing. In APeX-S, alanine aminotransferase levels >3xULN were seen in 14 of 49 patients who discontinued androgens within 28 days prior to study entry, compared to one of 104 patients who discontinued androgens more than 28 days prior to study entry and zero of 74 patients who had never used androgens. These observations support a proposed four-week washout period for current androgen patients before beginning therapy with BCX7353.
APeX-J Trial: On January 12, 2020, we reported data from our APeX-J trial in Japan, designed to support potential Japanese approval of berotralstat for the prevention of HAE attacks. APeX-J met its primary endpoint (p=0.003) for prevention of HAE attacks, and berotralstat was safe and generally well-tolerated.
We have also been developing berotralstat treatment of acute HAE attacks. In 2019, we completed ZENITH-1, an adaptive dose-ranging proof-of-concept Phase 2 clinical trial evaluating efficacy, safety and tolerability for the oral treatment of acute HAE attacks.
ZENITH-1 Trial: ZENITH-1 is a clinical trial studying three dose levels of a liquid formulation of berotralstat given as a single oral dose for the acute treatment of angioedema attacks in patients with HAE. ZENITH-1 is a randomized, double-blind, placebo-controlled, adaptive dose-ranging trial of the efficacy, safety and tolerability of berotralstat for treatment of acute angioedema attacks, and enrolled subjects with Type I and II HAE. Blinded study drug was dosed as an oral liquid after onset of symptoms, for up to three attacks in each subject, with each subject receiving both berotralstat (for two attacks) and placebo (for one attack) in a randomized sequence. The trial was structured for three consecutive cohorts testing single doses of 750 mg, 500 mg and 250 mg.
On February 23, 2019, we reported topline data from the completed Phase 2 ZENITH-1 trial. Data from the complete trial confirmed previously-reported results showing a single dose of oral 750 mg berotralstat was well-tolerated and superior to placebo (p<0.05) against the majority of efficacy endpoints evaluated in HAE patients suffering an acute attack, and demonstrated a clear dose response across the three dose levels evaluated, 250 mg, 500 mg and 750 mg. With the 750 mg dose, compared to placebo, improvements in symptoms and Visual Analog Scale (“VAS”) scores were demonstrated as early as one hour after oral berotralstat dosing (the first timepoint evaluated), and were sustained through 24 hours. Through 24 hours, standard of care (“SOC”) medication use was reduced by 31.6% after berotralstat compared with placebo (p=0.0029), and no or mild symptoms were reported in 64.1% of attacks treated with berotralstat compared with 32.3% of attacks treated with placebo (p=0.0038).
Berotralstat was generally safe and well-tolerated with no notable differentiation from the adverse event profile of placebo. The most commonly reported adverse events were diarrhea, abdominal pain, nausea, headache and nasopharyngitis. There were three discontinuations in the trial: one following a berotralstat 750 mg dose due to a transient, localized rash; one following a berotralstat 500 mg dose due to grade 2 vomiting and nausea and one following a placebo dose due to abdominal pain. With the exception of an unrelated ankle fracture, there were no grade 3 or 4 adverse events, and no grade 3 or 4 laboratory abnormalities.
ZENITH-2, a Phase 3 clinical trial of oral berotralstat (750 mg) for the acute treatment of HAE could begin in 2020, upon reaching agreement with regulatory agencies on trial protocol and additional formulation work on the acute oral formulation.
Complement-Mediated Diseases
On June 27, 2019, we announced that we began enrollment of a Phase 1 trial of BCX9930, an oral Factor D inhibitor discovered and developed by us, for the treatment of complement-mediated diseases. The objectives of the trial are to evaluate the safety and tolerability of single and multiple ascending doses of BCX9930 in healthy subjects and to characterize the pharmacokinetic (“PK”) and pharmacodynamic (“PD”) profiles of BCX9930 in single and multiple ascending doses of BCX9930 in healthy subjects (parts 1 & 2). In part three of the trial, there is an additional objective to demonstrate proof of concept in paroxysmal nocturnal hemoglobinuria (“PNH”) patients by evaluate key biomarkers of effectiveness in PNH patients taking BCX9930.
48
On October 28, 2019, we announced results from the ongoing Phase 1 trial of BCX9930 in 72 healthy volunteers. BCX9930 was safe and generally well tolerated, and showed rapid, sustained and >95% suppression of the alternative pathway (“AP”) of the complement system at 100 mg every 12 hours, as measured by the AP Wieslab® assay. In part 1 of the trial, a single ascending dose (“SAD”) assessment, six cohorts of healthy volunteers received a single dose of 10 mg, 30 mg, 100 mg, 300 mg, 600 mg or 1200 mg of oral BCX9930 or placebo (each SAD cohort randomized 6:2). In part 2 of the trial, the multiple ascending dose (“MAD”) assessment, two cohorts of healthy volunteers received 50 mg or 100 mg of oral BCX9930 or placebo (each MAD cohort randomized 10:2) administered every 12 hours for seven days. Healthy volunteers in the MAD cohorts were prophylactically dosed with the broad-spectrum antibiotic, amoxicillin/clavulanate. BCX9930 was safe and generally well tolerated at all doses studied. There were no serious adverse events. A clinically benign rash was observed in some healthy volunteers in the MAD assessment (two in the 50 mg cohort, seven in the 100 mg cohort), which was self-limited and resolved in 4-8 days after onset. There were no discontinuations from the trial.
On January 12, 2020, we announced that we have completed an additional MAD cohort with 50 mg of oral BCX9930 or placebo administered every 12 hours for 14 days, with vaccination instead of an antibiotic. In the additional MAD cohort, a benign rash (similar to prior MAD cohorts) that was self-limited and resolved in 4 to 8 days post-onset was seen in seven healthy volunteers; the protocol allowed two of these healthy volunteers with more limited surface area affected by the rash to continue receiving BCX9930. Both of these healthy volunteers successfully dosed-through benign rash, with rash resolving on-drug, in both patients; biopsies of rashes from multiple subjects confirm the benign nature of the rash. The protocol for part three of the trial in PNH patients allows any patient who develops a clinically benign rash to continue dosing with BCX9930.
Based on the safety, tolerability, PK and PD dose-response results from parts 1 and 2 of the Phase 1 trial, we plan to complete additional MAD dosing cohorts and advance to part 3 of the trial, a proof of concept (“PoC”) study of BCX9930 in PNH patients who are poor responders to eculizumab or ravulizumab, and treatment-naïve patients. We have also successfully dosed MAD cohorts of 200 milligrams twice a day and 400 milligrams twice a day. On March 5, 2020 we announced that we had dosed the first PNH patients in part three of the trial. These patients were naïve to eculizumab and ravulizumab. We expect to report data from the PoC study in PNH patients in the second quarter of 2020.
Fibrodysplasia Ossificans Progressiva (“FOP”)
The goal of the ALK2 inhibitor project program at BioCryst is to discover and develop orally administered kinase inhibitor drug candidates that are able to slow or prevent the progressive formation of bone in soft tissues, also known as heterotopic ossification (“HO”). Our lead compound, BCX9250, reduced HO in an experimental model of ALK2-driven HO in laboratory rats, with up to 89 percent reduction in volume of HO compared to controls. On November 1, 2019, we announced that we had begun a Phase 1 clinical trial with oral BCX9250 for the treatment of FOP. The Phase 1 trial will evaluate single and multiple ascending doses of oral BCX9250 in healthy volunteers. We expect to report the results from the trial in the second half of 2020.
RAPIVAB/ALPIVAB/RAPIACTA/PERAMIFLU (peramivir injection)
In September 2018, the Centers for Disease Control and Prevention awarded us a $34.7 million contract for the procurement of up to 50,000 doses of RAPIVAB over a five-year period to supply the Strategic National Stockpile for use in a public health emergency. We delivered two shipments within the award in 2019, totaling approximately $13.9 million, and we expect to deliver at least one shipment within the award in 2020, totaling approximately $6.9 million.
Galidesivir (formerly BCX4430)
On May 9, 2019, we announced the completion of a randomized, placebo-controlled Phase 1 clinical trial to evaluate intravenous (IV) galidesivir in healthy volunteers. In the trial, galidesivir was generally safe and well tolerated. This placebo-controlled trial evaluated the safety, tolerability and pharmacokinetics of escalating doses of galidesivir in four single-dose cohorts of 5mg/kg, 10 mg/kg, 15 mg/kg and 20 mg/kg, with a total of 24 volunteers receiving galidesivir by IV infusion. Drug exposures (Cmax and AUC) at the highest dose were 20,500 ng/mL and 44,600 hr.ng/mL, similar to or greater than drug exposures needed in nonclinical galidesivir treatment experiments in Marburg virus disease and Yellow Fever.
We are in the process of initiating an exploratory Phase 1b clinical trial evaluating galidesivir in Yellow Fever patients in Brazil.
We are in active dialogue with NIAID, relevant U.S. public health authorities, and clinical investigators as they assess potential approaches to evaluate investigational antiviral drugs for treatment of COVID-19, with the goal of determining if galidesivir is effective against this strain, assessing whether galidesivir should be tested in new or existing clinical trials in patients with COVID-19, and expanding the current supply of the drug.
Modified Secured Credit Facility
On February 6, 2019, we entered into a $100.0 million secured credit facility (the “Second Amended and Restated Senior Credit Facility”) with MidCap Financial as administrative agent and lender (“MidCap”), pursuant to the terms and conditions of that certain Second Amended and Restated Credit and Security Agreement, dated as of February 6, 2019 (the “Credit Agreement”). The Second Amended and Restated Senior Credit Facility will be available in three tranches, with (i) the first tranche comprised of $50.0 million funded at closing of the Credit Agreement, which included $30.0 million of proceeds that were deemed rolled over from the outstanding principal amount under the prior Amended and Restated Senior Credit Facility dated as of July 20, 2018 (the “Amended and Restated Senior Credit Facility”), (ii) the second tranche to be comprised of $30.0 million, and (iii) the third tranche to be comprised of $20.0 million, with the second and third tranches to be funded upon the completion of certain contingencies related to our development activities and the establishment of certain financial covenants. On September 10, 2019 the Company executed the first amendment to the Second Amended and Restated Credit Facility which extended the commitment termination date for the second tranche to November 30, 2019. On November 30, 2019, the Company’s access to the second tranche expired.
49
The Second Amended and Restated Senior Credit Facility refinanced and replaced our prior Amended and Restated Senior Credit Facility dated as of July 20, 2018. The Second Amended and Restated Senior Credit Facility bears a variable interest rate of LIBOR (which shall not be less than 0.5%) plus 8%. The Second Amended and Restated Senior Credit Facility includes an interest-only payment period through June 2020 and scheduled monthly principal and interest payments for the subsequent 30 months. We used a portion of the proceeds of the Second Amended and Restated Senior Credit Facility to pay off outstanding amounts under our prior Amended and Restated Senior Credit Facility and the remainder will be used for general corporate purposes.
Results of Operations
Year Ended December 31, 2019 Compared to 2018
Total 2019 revenues increased to $48.8 million as compared to 2018 revenues of $20.7 million. The increase in 2019 revenue was primarily due to recognition of $20.1 million of a $22.0 million upfront milestone payment from Torii, $13.9 million of RAPIVAB product sales under our U.S. Department of Health and Human Services procurement contract and $3.7 million of peramivir product sales to our licensing partners. The increase in revenues was partially offset by the recognition of peramivir milestones recognized in 2018 that did not recur in 2019. Revenues in 2019 included $6.3 million of royalty revenue from SUL, Shionogi and Green Cross associated with sales of peramivir in the United States, Japan and Korea, $4.9 million of reimbursement of collaborative expenses from NIAID/HHS and BARDA/HHS under U.S. Government development contracts, $3.7 million of peramivir product revenue from inventory sales to our commercial partners, $13.9 million of RAPIVAB product revenue from inventory sales to BARDA/HHS under our Government procurement contract and $20.1 million associated with milestone revenue and collaborative revenue from corporate partnerships. Revenues in 2018 included $5.9 million of royalty revenue from SUL, Shionogi and Green Cross associated with sales of peramivir in the United States, Japan and Korea, $2.6 million of reimbursement of collaborative expenses from NIAID/HHS and BARDA/HHS under U.S. Government development contracts and $12.0 million associated with milestone revenue and collaborative revenue from corporate partnerships. Our future RAPIVAB revenue will be difficult to predict because of volatility in prevalence, timing and severity of influenza season to season as well as variable commercialization efforts and resources dedicated to our products by us and our collaborative partners.
Research and Development (“R&D”) expenses increased to $107.1 million in 2019 from $84.9 million in 2018. The increase in 2019 R&D expenses, as compared to 2018, is primarily due to increased spending on our complement-mediated diseases program and other preclinical development initiatives.
The following table summarizes our R&D expenses for the periods indicated (amounts are in thousands).
| 2019 | 2018 | 2017 | ||||||||||
| R&D expenses by program: | ||||||||||||
| Berotralstat (BCX7353) | $ | 57,059 | $ | 53,993 | $ | 40,974 | ||||||
| BCX9930 | 26,640 | 10,189 | 6,609 | |||||||||
| FOP | 6,167 | 8,871 | 3,494 | |||||||||
| Galidesivir | 4,680 | 2,428 | 3,757 | |||||||||
| Peramivir | 2,143 | 1,936 | 4,872 | |||||||||
| Other 2nd generation HAE compounds | 6 | 357 | 1,111 | |||||||||
| Other research, preclinical and development costs | 10,373 | 7,114 | 6,145 | |||||||||
| Total R&D expenses | $ | 107,068 | $ | 84,888 | $ | 66,962 | ||||||
R&D expenses include all direct and indirect expenses and are allocated to specific programs at the point of development of a lead product candidate. Direct expenses are charged directly to the program to which they relate and indirect expenses are allocated based upon internal direct labor hours dedicated to each respective program. Direct expenses consist of compensation for R&D personnel and costs of outside parties to conduct laboratory studies, develop manufacturing processes, manufacture the product candidates, conduct and manage clinical trials, as well as other costs related to our clinical and preclinical studies. Indirect R&D expenses consist of lab supplies and services, facility expenses, depreciation of development equipment and other overhead of our research and development efforts. R&D expenses vary according to the number of programs in clinical development and the stage of development of our clinical programs. Later stage clinical programs tend to cost more than earlier stage programs due to the longer length of time of the clinical trials and the higher number of patients enrolled in these clinical trials.
50
Selling, general and administrative (“SG&A”) expenses increased to $37.1 million in 2019 compared to $29.5 million in 2018. The increase of $7.6 million was primarily due to increased spending on commercial activities and medical affairs to support the anticipated U.S. commercial launch of berotralstat in 2020. The increased commercial and medical affairs costs were partially offset by the non-recurring merger-related costs associated with our terminated merger with Idera and a $4.9 million reserve for collectability of the EMA approval milestone of peramivir. We expect our SG&A expenses to continue to increase as we near the expected commercial launch of berotralstat.
Interest expense, which is related to the non-recourse notes issued in conjunction with the non-dilutive RAPIACTA royalty monetization transaction in March 2011 and borrowings under our secured credit facility with MidCap Financial (“MidCap”), pursuant to the terms and conditions of that certain Second Amended and Restated Credit and Security Agreement, dated as of February 6, 2018 (the “Second Amended and Restated Senior Credit Facility”), increased to $11.9 million in 2019 as compared to $9.2 million in 2018. The increase in interest expense is primarily related to an increase in the outstanding balance of the the Second Amended and Restated Senior Credit Facility in February 2019 and increased interest expense associated with our non-recourse notes payable. In addition, a mark to market loss of $0.4 million was recognized in 2019 related to the foreign currency hedge entered into in conjunction with the royalty monetization transaction, compared to a mark to market loss of $1.0 million in 2018, both resulting from changes in the U.S. dollar/Japanese yen exchange rate during the respective years. In addition, realized currency exchange gains of $0.9 million were recognized in each of 2019 and 2018, respectively, related to the exercise of a U.S. dollar/Japanese yen currency option under our foreign currency hedge. We entered into a foreign currency hedge agreement to hedge changes in the value of the Japanese yen relative to the U.S. dollar associated with the RAPIACTA royalty monetization. The currency hedge does not qualify for hedge accounting treatment and therefore mark to market adjustments are recognized in our Consolidated Statements of Comprehensive Loss. Although we cannot predict the future yen/dollar exchange rate, the applicable foreign currency rates moved such that we currently have no collateral posted; however, it is possible that collateral will be required to be posted in the future. We are unable to predict future changes in the yen/dollar exchange rate or increases/decreases in our hedge gains/losses associated with the currency hedge agreement.
Liquidity and Capital Resources
Cash expenditures have exceeded revenues since our inception and we expect our 2019 operating expenses to exceed our 2019 revenues. Our operations have principally been funded through public offerings and private placements of equity securities; cash from collaborative and other research and development agreements, including U.S. Government contracts for RAPIVAB and galidesivir; and to a lesser extent, the PhaRMA Notes financing and the Senior Credit Facility, the Amended and Restated Credit Facility, and the Second Amended and Restated Credit Facility (defined below). To date, we have been awarded a BARDA/HHS RAPIVAB development contract totaling $234.8 million, which expired on June 30, 2014, a NIAID/HHS galidesivir development contract totaling $43.0 million, which is ongoing, and a BARDA/HHS galidesivir development contract totaling $39.1 million, which is also ongoing. The total amount of NIAID/HHS and BARDA/HHS galidesivir funding obligated under awarded options is $43.0 million and $20.6 million, respectively. We may issue securities through private placement transactions or registered public offerings pursuant to a registration statement filed with the SEC. In addition to the above, we have received funding from other sources, including other collaborative and other research and development agreements; government grants; equipment lease financing; facility leases; research grants; and interest income on our investments.
As of December 31, 2019, we had net working capital of $72.0 million, an increase of approximately $27.1 million from $44.9 million at December 31, 2018. The increase in working capital was principally due to proceeds from the Second Amended and Restated Senior Credit Facility, the November 2019 public offering of our common stock, the November 2019 offering of pre-funded warrants to purchase our common stock and the upfront milestone associated with the Torii license agreement partially offset by our normal operating expenses associated with the development of our product candidates. Our principal sources of liquidity at December 31, 2019 were approximately $114.1 million in cash and cash equivalents and approximately $22.1 million in investments considered available-for-sale. On February 6, 2019, we replaced our prior $30.0 million Amended and Restated Senior Credit Facility with our Second Amended and Restated Senior Credit Facility. We anticipate our cash and investments will fund our operations into 2021.
We intend to contain costs and cash flow requirements by closely managing our third party costs and headcount, leasing scientific equipment and facilities, contracting with other parties to conduct certain research and development projects and using consultants. We expect to incur additional expenses, potentially resulting in significant losses, as we continue to pursue our research and development activities and begin to build a commercial infrastructure. We may incur additional expenses related to the filing, prosecution, maintenance, defense and enforcement of patent and other intellectual property claims and additional regulatory costs as our clinical programs advance through later stages of development. The objective of our investment policy is to ensure the safety and preservation of invested funds, as well as maintaining liquidity sufficient to meet cash flow requirements. We place our excess cash with high credit quality financial institutions, commercial companies, and government agencies in order to limit the amount of our credit exposure. We have not realized any significant losses on our investments.
51
We plan to finance our needs principally from the following:
| • | lease, royalty or loan financing and future public or private equity financing; |
| • | our existing capital resources and interest earned on that capital; |
| • | payments under existing and executing new contracts with the U.S. Government; and |
| • | payments under current or future collaborative and licensing agreements with corporate partners. |
As our programs continue to advance, our costs will increase. Our current and planned clinical trials, plus the related development, manufacturing, regulatory approval process requirements and additional personnel resources and testing required for the continuing development of our product candidates will consume significant capital resources and will increase our expenses. Our expenses, revenues and cash utilization rate could vary significantly depending on many factors, including our ability to raise additional capital, the development progress of our collaborative agreements for our product candidates, the amount and timing of funding we receive from existing U.S. Government contracts for galidesivir, the amount of funding or assistance, if any, we receive from new U.S. Government contracts or other new partnerships with third parties for the development and or commercialization of our product candidates, the progress and results of our current and proposed clinical trials for our most advanced product candidates, the progress made in the manufacturing of our lead product candidates, commercializing our products, and the overall progression of our other programs.
With the funds available at December 31, 2019, we believe our financial resources will be sufficient to fund our operations into 2021. We have sustained operating losses for the majority of our corporate history and expect that our 2020 expenses will exceed our 2020 revenues. We expect to continue to incur operating losses and negative cash flows until revenues reach a level sufficient to support ongoing operations. Accordingly, our planned operations raise doubt about our ability to continue as a going concern throughout 2021. Our liquidity needs will be largely determined by the success of operations in regards to the progression of our product candidates in the future. We also may consider other plans to fund operations through 2021 including: (1) securing or increasing U.S. Government funding of our programs, including obtaining procurement contracts; (2) out-licensing rights to certain of our products or product candidates, pursuant to which the we would receive cash milestones; (3) raising additional capital through equity or debt financings or from other sources; (4) obtaining additional product candidate regulatory approvals, which would generate revenue, milestones and cash flow; (5) reducing spending on one or more research and development programs, including by discontinuing development; and/or (6) restructuring operations to change our overhead structure. We may issue securities, including common stock, preferred stock, depositary shares, stock purchase contracts, warrants and units, through private placement transactions or registered public offerings. Our future liquidity needs, and ability to address those needs, will largely be determined by the success of our product candidates, timing, scope and magnitude of its commercial expenses and key development and regulatory events and our decisions in the future.
Our long-term capital requirements and the adequacy of our available funds will depend upon many factors, including:
| • | our ability to perform under our government contracts and receive reimbursement, and receive stockpiling procurement contracts; | ||
| • | the magnitude of work under our government contracts; | ||
| • | the progress and magnitude of our research, drug discovery and development programs; | ||
| • | changes in existing collaborative relationships or government contracts; | ||
| • | our ability to establish additional collaborative relationships with academic institutions, biotechnology or pharmaceutical companies and governmental agencies or other third parties; | ||
| • | the extent to which our partners, including governmental agencies, will share in the costs associated with the development of our programs or run the development programs themselves; | ||
| • | our ability to negotiate favorable development and marketing strategic alliances for certain product candidates or a decision to build or expand internal development and commercial capabilities; | ||
| • | successful commercialization of marketed products by either us or a partner; |
| • | the scope and results of preclinical studies and clinical trials to identify and develop product candidates; | ||
| • | our ability to engage sites and enroll subjects in our clinical trials; | ||
| • | the scope of manufacturing of our product candidates to support our preclinical research and clinical trials; |
52
| • | increases in personnel and related costs to support the development and commercialization of our product candidates; | ||
| • | the scope of manufacturing of our drug substance and product candidates required for future NDA filings; | ||
| • | competitive and technological advances; | ||
| • | the time and costs involved in obtaining regulatory approvals; | ||
| • | post-approval commitments for RAPIVAB and other products that receive regulatory approval; and | ||
| • | the costs involved in all aspects of intellectual property strategy and protection including the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims. |
We expect that we will be required to raise additional capital to complete the development and commercialization of our current product candidates and we may seek to raise capital in the future. Additional funding, whether through additional sales of equity or debt securities, collaborative or other arrangements with corporate partners or from other sources, including governmental agencies in general and existing government contracts specifically, may not be available when needed or on terms acceptable to us. The issuance of preferred or common stock or convertible securities, with terms and prices significantly more favorable than those of the currently outstanding common stock, could have the effect of diluting or adversely affecting the holdings or rights of our existing stockholders. In addition, collaborative arrangements may require us to transfer certain material rights to such corporate partners. Insufficient funds may require us to delay, scale back or eliminate certain of our research and development programs. Our future working capital requirements, including the need for additional working capital, will be largely determined by the advancement of our portfolio of product candidates as well as rate of reimbursement by U.S. Government agencies of our galidesivir expenses and any future decisions regarding the future of the RAPIVAB and galidesivir programs, including those relating to stockpiling procurement. More specifically, our working capital requirements will be dependent on the number, magnitude, scope and timing of our development programs; regulatory approval of our product candidates; obtaining funding from collaborative partners; the cost, timing and outcome of regulatory reviews, regulatory investigations, and changes in regulatory requirements; the costs of obtaining patent protection for our product candidates; the timing and terms of business development activities; the rate of technological advances relevant to our operations; the efficiency of manufacturing processes developed on our behalf by third parties; the timing, scope and magnitude of commercial spending, and the level of required administrative support for our daily operations.
The restrictive covenants contained in the Second Amended and Restated Senior Credit Facility could cause us to be unable to pursue business opportunities that we or our stockholders may consider beneficial without the lender’s permission or without repaying all Second Amended and Restated Senior Credit Facility obligations. These covenants limit our ability to, among other things, convey, sell, lease, license, transfer or otherwise dispose of certain parts of our business or property; change the nature of our business; liquidate or dissolve; enter into certain change in control or acquisition transactions; incur or assume certain debt; grant certain types of liens on our assets; modify, liquidate or transfer assets in certain collateral accounts; pay dividends or make certain distributions to our stockholders; make certain investments; enter into material transactions with affiliates; and modify existing debt or collaboration arrangements. A breach of any of these covenants could result in an event of default under the Second Amended and Restated Senior Credit Facility.
Financial Outlook for 2020
Based upon our development and commercial plans, expected operations and our awarded government contracts, we expect 2020 operating cash usage to be in the range of $125 to $150 million, and expect our total 2020 operating expenses to be in the range of $135 to $160 million. Our operating expense range excludes equity-based compensation expense due to the difficulty in accurately projecting this expense as it is significantly impacted by the volatility and price of our stock, as well as vesting of our outstanding performance-based stock options. Our operating cash forecast excludes any impact of our royalty monetization, hedge collateral posted or returned, and any other non-routine cash outflows or inflows. Our ability to remain within our operating expense and operating cash target ranges is subject to multiple factors, including unanticipated or additional general development and administrative costs and other factors described under the Risk Factors located elsewhere in this report.
Off-Balance Sheet Arrangements
As of December 31, 2019, we are not involved in any unconsolidated entities or off–balance sheet arrangements.
53
Contractual Obligations
In the table below, we set forth our enforceable and legally binding obligations and future commitments and obligations related to all contracts that we are likely to continue regardless of the fact that they are cancelable as of December 31, 2019. Some of the amounts we include in this table are based on management’s estimates and assumptions about these obligations, including their duration, the possibility of renewal, anticipated actions by third parties, and other factors. Because these estimates and assumptions are necessarily subjective, the obligations we will actually pay in future periods may vary from those reflected in the table.
| Payments Due by Period (In thousands) | ||||||||||||||||||||
| Contractual Obligations | Total | Less Than 1 Year | 1-3 Years | 3-5 Years | More Than 5 Years | |||||||||||||||
| Operating lease obligations | $ | 10,873 | $ | 1,465 | $ | 1,126 | $ | 1,033 | $ | 7,249 | ||||||||||
| Purchase obligations(1) | 59,782 | 59,782 | - | - | - | |||||||||||||||
| Contingent license obligations | 1,200 | 150 | 300 | 300 | 450 | |||||||||||||||
| Non-recourse notes payable(2) | 48,335 | 48,335 | - | - | - | |||||||||||||||
| Senior credit facility | 61,966 | 15,106 | 46,860 | - | - | |||||||||||||||
| Total | $ | 182,156 | $ | 124,838 | $ | 48,286 | $ | 1,333 | $ | 7,699 | ||||||||||
| (1) | Purchase obligations include commitments related to clinical development, manufacturing and research operations and other purchase commitments. |
| (2) | Assumes the PhaRMA Notes will be repaid at maturity and the related interest costs will accrue and be paid annually through maturity. This assumption is based on the unpredictable nature of the royalty payments from Shionogi, which are designated for both principal and interest payments on the PhaRMA Notes. |
Under the Currency Hedge Agreement, we have the right to purchase dollars and sell yen at a rate of 100 yen per dollar for which we may be required to pay a premium in 2020. A payment of $2.0 million will be required if, on May 18, the dollar is worth 100 yen or less. As of December 31, 2019, we have no hedge collateral posted against the Currency Hedge Agreement. Because the posting of additional collateral and payment of annual premiums is contingent on the value of the yen relative to the dollar and other factors, such payments have been excluded from the foregoing table.
In addition to the above, we have committed to make potential future “sublicense” payments to third-parties as part of in-licensing and development programs. Payments under these agreements generally become due and payable only upon achievement of certain developmental, regulatory and/or commercial milestones. Because the achievement of these milestones is neither probable nor reasonably estimable, such contingencies have not been recorded on our balance sheet.
Critical Accounting Policies
We have established various accounting policies that govern the application of U.S. GAAP, which were utilized in the preparation of our consolidated financial statements. Certain accounting policies involve significant judgments and assumptions by management that have a material impact on the carrying value of certain assets and liabilities. Management considers such accounting policies to be critical accounting policies. The judgments and assumptions used by management are based on historical experience and other factors, which are believed to be reasonable under the circumstances. Because of the nature of the judgments and assumptions made by management, actual results could differ from these judgments and estimates, which could have a material impact on the carrying values of assets and liabilities and the results of operations.
While our significant accounting policies are more fully described in Note 1 to our consolidated financial statements included in this Annual Report on Form 10-K for the year ended December 31, 2019, we believe that the following accounting policies are the most critical to aid you in fully understanding and evaluating our reported financial results and affect the more significant judgments and estimates that we use in the preparation of our financial statements.
Inventory
Our inventories consist of peramivir finished goods and work in process, which are valued at the lower of cost or net realizable value using the first-in, first-out (i.e., FIFO) method. Cost includes materials, labor, overhead, shipping and handling costs. Our inventories are subject to expiration dating. We regularly evaluate the carrying value of our inventories and provide valuation reserves for any estimated obsolete, short-dated or unmarketable inventories. In addition, we may experience spoilage of our raw materials and supplies. Our determination that a valuation reserve might be required, in addition to the quantification of such reserve, requires us to utilize significant judgment. In connection with the FDA approval of RAPIVAB and other regulatory approvals, we began capitalizing costs associated with the production of peramivir inventories.
54
Accrued Expenses
We enter into contractual agreements with third-party vendors who provide research and development, manufacturing, and other services in the ordinary course of business. Some of these contracts are subject to milestone-based invoicing and services are completed over an extended period of time. We record liabilities under these contractual commitments when an obligation has been incurred. This accrual process involves reviewing open contracts and purchase orders, communicating with our applicable personnel to identify services that have been performed and estimating the level of service performed and the associated cost when we have not yet been invoiced or otherwise notified of actual cost. The majority of our service providers invoice us monthly in arrears for services performed. We make estimates of our accrued expenses as of each balance sheet date based on the facts and circumstances known to us. We periodically confirm the accuracy of our estimates with the service providers and make adjustments if necessary. Examples of estimated accrued expenses include:
| • |
fees paid to Clinical Research Organizations (“CROs”) in connection with preclinical and toxicology studies and clinical trials; |
||
| • |
fees paid to investigative sites in connection with clinical trials; |
||
| • |
fees paid to contract manufacturers in connection with the production of our raw materials, drug substance and product candidates; and |
||
| • | professional fees. |
We base our expenses related to clinical trials on our estimates of the services received and efforts expended pursuant to contracts with multiple research institutions and CROs that conduct and manage clinical trials on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. Payments under some of these contracts depend on factors such as the successful enrollment of patients and the completion of clinical trial milestones. In accruing service fees, we estimate the time period over which services will be performed and the level of effort expended in each period. If the actual timing of the performance of services or the level of effort varies from our estimate, we will adjust the accrual accordingly. If we do not identify costs that we have begun to incur or if we underestimate or overestimate the level of these costs, our actual expenses could differ from our estimates.
Revenue Recognition
We adopted the provisions of ASC 606 as of January 1, 2018 using the modified retrospective method as applied to contracts that were not completed as of that date. As a result, financial information for reporting periods beginning after January 1, 2018 are presented under ASC 606, while comparative financial information has not been adjusted and continues to be reported in accordance with our historical accounting policy for revenue recognition prior to the adoption of ASC 606.
Collaborative and Other Research and Development Arrangements and Royalties
We recognize revenue when we satisfy a performance obligation by transferring promised goods or services to a customer. Revenue is measured at the transaction price that is based on the amount of consideration that we expect to receive in exchange for transferring the promised goods or services to the customer. The transaction price includes estimates of variable consideration to the extent it is probable that a significant reversal of revenue recognized will not occur.
We have collaboration and license agreements with a number of third parties as well as research and development agreements with certain government entities. Our primary sources of revenue are license, service, royalty and product sale revenues from these collaborative and other research and development arrangements.
Revenue from license fees, royalty payments, milestone payments, and research and development fees are recognized as revenue when the earnings process is complete and we have no further continuing performance obligations or we have completed the performance obligations under the terms of the agreement.
Arrangements that involve the delivery of more than one performance obligation are initially evaluated as to whether the intellectual property licenses granted by us represent distinct performance obligations. If they are determined to be distinct, the value of the intellectual property licenses would be recognized up-front while the research and development service fees would be recognized as the performance obligations are satisfied. Variable consideration is assessed at each reporting period as to whether it is not subject to significant future reversal and, therefore, should be included in the transaction price at the inception of the contract. If a contract includes a fixed or minimum amount of research and development support, this also would be included in the transaction price. Changes to collaborations, such as the extensions of the research term or increasing the number of targets or technology covered under an existing agreement, are assessed for whether they represent a modification or should be accounted for as a new contract. For contracts with multiple performance obligations, revenue is allocated to each performance obligation based on its relative standalone selling price. Standalone selling prices are based on observable prices at which we separately sell the products or services. If a standalone selling price is not directly observable, then we estimate the standalone selling price considering market conditions and entity-specific factors. Analyzing the arrangement to identify performance obligations requires the use of judgment, and each may be an obligation to deliver services, a right or license to use an asset, or another performance obligation.
55
Milestone payments are recognized as licensing revenue upon the achievement of specified milestones if (i) the milestone is substantive in nature and the achievement of the milestone was not probable at the inception of the agreement; and (ii) we have a right to payment. Any milestone payments received prior to satisfying these revenue recognition criteria are recorded as deferred revenue.
Reimbursements received for direct out-of-pocket expenses related to research and development costs are recorded as revenue in the Consolidated Statements of Comprehensive Loss rather than as a reduction in expenses. Under our contracts with the Biomedical Advanced Research and Development Authority within the United States Department of Health and Human Services (”BARDA/HHS”) and the National Institute of Allergy and Infectious Diseases (“NIAID/HHS”), revenue is recognized as reimbursable direct and indirect costs are incurred.
Under certain of our license agreements, we receive royalty payments based upon our licensees’ net sales of covered products. Royalties are recognized at the later of when (i) the subsequent sale or usage occurs, or (ii) the performance obligation to which some or all of the sales-based or usage-based royalty has been satisfied.
Product Sales
Our principal sources of product sales are sales of peramivir to our licensing partners and sales of RAPIVAB to the U.S. Department of Health and Human Services under our procurement contract. We recognize revenue for sales when the customer obtains control of the product, which generally occurs upon delivery.
Contract Balances
The timing of revenue recognition, billings and cash collections results in billed accounts receivable, unbilled receivables (contract assets) and deferred revenue and billings in excess of revenue recognized (contract liabilities) on the Consolidated Balance Sheets.
Contract assets - Our long-term contracts are billed as work progresses in accordance with the contract terms and conditions, either at periodic intervals or upon achievement of certain milestones. Often this results in billing occurring subsequent to revenue recognition, resulting in contract assets. Contract assets are generally classified as current assets in the Consolidated Balance Sheets.
Contract liabilities - We often receive cash payments from customers in advance of our performance, resulting in contract liabilities. These contract liabilities are classified as either current or long-term in the Consolidated Balance Sheets based on the timing of when we expect to recognize the revenue.
Contract Costs
We may incur direct and indirect costs associated with obtaining a contract. Incremental contract costs that we expect to recover are capitalized and amortized over the expected term of the contract. Non-incremental contract costs and costs that we expect to recover are expensed as incurred.
Research and Development Expenses
Our research and development costs are charged to expense when incurred. Advance payments for goods or services that will be used or rendered for future research and development activities are deferred and capitalized. Such amounts are recognized as expense when the related goods are delivered or the related services are performed. Research and development expenses include, among other items, personnel costs, including salaries and benefits, manufacturing costs, clinical, regulatory, and toxicology services performed by CROs, materials and supplies, and overhead allocations consisting of various administrative and facilities related costs. Most of our manufacturing and clinical and preclinical studies are performed by third-party CROs. Costs for studies performed by CROs are accrued by us over the service periods specified in the contracts and estimates are adjusted, if required, based upon our on-going review of the level of services actually performed.
56
Additionally, we have license agreements with third parties, such as the Albert Einstein College of Medicine of Yeshiva University, Industrial Research, Ltd. and the University of Alabama at Birmingham (“UAB”), which require fees related to sublicense agreements or maintenance fees. We expense sublicense payments as incurred unless they are related to revenues that have been deferred, in which case the expenses are deferred and recognized over the related revenue recognition period. We expense maintenance payments as incurred.
Deferred collaboration expenses represent sub-license payments paid to our academic partners upon receipt of consideration from various commercial partners, and other consideration to our academic partners for modification to existing license agreements. These deferred expenses would not have been incurred without receipt of such payments or modifications from our commercial partners and are being expensed in proportion to the related revenue being recognized. We believe that this accounting treatment appropriately matches expenses with the associated revenue.
We group our R&D expenses into two major categories: direct external expenses and indirect expenses. Direct expenses consist of compensation for R&D personnel and costs of outside parties to conduct laboratory studies, develop manufacturing processes and manufacture the product candidate, conduct and manage clinical trials, as well as other costs related to our clinical and preclinical studies. These costs are accumulated and tracked by active program. Indirect expenses consist of lab supplies and services, facility expenses, depreciation of development equipment and other overhead of our research and development efforts. These costs apply to work on non-active product candidates and our discovery research efforts.
Stock-Based Compensation
All share-based payments, including grants of stock option awards and restricted stock unit awards, are recognized in our Consolidated Statements of Comprehensive Loss based on their fair values. Stock-based compensation cost is estimated at the grant date based on the fair value of the award and is recognized as expense on a straight-line basis over the requisite service period of the award. Determining the appropriate fair value model and the related assumptions for the model requires judgment, including estimating the life of an award, the stock price volatility, and the expected term. We utilize the Black-Scholes option-pricing model to value our awards and recognize compensation expense on a straight-line basis over the vesting periods. The estimation of share-based payment awards that will ultimately vest requires judgment, and to the extent actual results or updated estimates differ from our current estimates, such amounts will be recorded as a cumulative adjustment in the period estimates are revised. In addition, we have outstanding performance-based stock options for which no compensation expense is recognized until “performance” has occurred. Significant management judgment is also required in determining estimates of future stock price volatility and forfeitures to be used in the valuation of the options. Actual results, and future changes in estimates, may differ substantially from our current estimates.
Foreign Currency Hedge
In connection with our issuance of the PhaRMA Notes, we entered into a foreign Currency Hedge Agreement to hedge certain risks associated with changes in the value of the Japanese yen relative to the U.S. dollar. Under the Currency Hedge Agreement, we have the right to purchase dollars and sell yen at a rate of 100 yen per dollar for which we may be required to pay a premium in 2020, provided the Currency Hedge Agreement remains in effect. A payment of $2.0 million will be required if, on May 18, the U.S. dollar is worth 100 yen or less as determined in accordance with the Currency Hedge Agreement. In conjunction with establishing the Currency Hedge Agreement, we will be required to post collateral to the counterparty, which may cause us to experience additional quarterly volatility in our financial results. We will not be required at any time to post collateral exceeding the maximum premium payments remaining payable under the Currency Hedge Agreement. As of December 31, 2019, the maximum amount of hedge collateral we may be required to post is $2.0 million.
The Currency Hedge Agreement does not qualify for hedge accounting treatment and therefore mark to market adjustments will be recognized in our Consolidated Statements of Comprehensive Loss. Mark to market adjustments are determined by quoted prices in markets that are not actively traded and for which significant inputs are observable directly or indirectly, representing the Level 2 in the fair value hierarchy as defined by generally accepted accounting principles (“U.S. GAAP”). We are also required to post collateral in connection with the mark to market adjustments based on defined thresholds. As of December 31, 2019, no collateral was posted under the agreement.
Tax
We account for uncertain tax positions in accordance with U.S. GAAP. Significant management judgment is required in determining our provision for income taxes, deferred tax assets and liabilities and any valuation allowance recorded against net deferred tax assets. We have recorded a valuation allowance against all potential tax assets, due to uncertainties in our ability to utilize deferred tax assets, primarily consisting of certain net operating losses carried forward, before they expire. The valuation allowance is based on estimates of taxable income in each of the jurisdictions in which we operate and the period over which our deferred tax assets will be recoverable.
57
Impact of Inflation
We do not believe that our operating results have been materially impacted by inflation during the past three years. However, we cannot be assured that our operating results will not be adversely affected by inflation in the future. We will continually seek to mitigate the adverse effects of inflation on the services that we use through improved operating efficiencies and cost containment initiatives.
Recent Accounting Pronouncements
Note 12 to the Consolidated Financial Statements included in Item 8 of this Annual Report on Form 10-K discusses accounting pronouncements recently issued or proposed but not yet required to be adopted.
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK. |
Interest Rate Risk
We are subject to interest rate risk on our investment portfolio and borrowings under our fixed-interest rate PhaRMA Notes and our variable-interest rate Second Amended and Restated Senior Credit Facility. The interest rate applicable to our borrowings under the PhaRMA Notes is fixed at 14.0% and the Second Amended and Restated Senior Credit Facility bears a floating interest rate based on LIBOR. Increases in interest rates could therefore increase the associated interest payments that we are required to make on the Senior Credit Facility. As of December 31, 2019, our Second Amended and Restated Senior Credit Facility had an interest rate of 9.7%.
We invest in marketable securities in accordance with our investment policy. The primary objectives of our investment policy are to preserve capital, maintain proper liquidity to meet operating needs and maximize yields. Our investment policy specifies credit quality standards for our investments and limits the amount of credit exposure to any single issue, issuer or type of investment. We place our excess cash with high credit quality financial institutions, commercial companies, and government agencies in order to limit the amount of credit exposure. Some of the securities we invest in may have market risk. This means that a change in prevailing interest rates may cause the principal amount of the investment to fluctuate.
Our investment exposure to market risk for changes in interest rates relates to the increase or decrease in the amount of interest income we can earn on our portfolio, changes in the market value due to changes in interest rates and other market factors as well as the increase or decrease in any realized gains and losses. Our investment portfolio includes only marketable securities and instruments with active secondary or resale markets to help ensure portfolio liquidity. A hypothetical 100 basis point increase or decrease in interest rates along the entire interest rate yield curve would not significantly affect the fair value of our interest sensitive financial instruments, including our borrowings, but may affect our future earnings and cash flows. We generally have the ability to hold our fixed-income investments to maturity and therefore do not expect that our operating results, financial position or cash flows will be materially impacted due to a sudden change in interest rates. However, our future investment income may fall short of expectations due to changes in interest rates, or we may suffer losses in principal if forced to sell securities which have declined in market value due to changes in interest rates or other factors, such as changes in credit risk related to the securities’ issuers. To minimize this risk, we schedule our investments to have maturities that coincide with our expected cash flow needs, thus avoiding the need to redeem an investment prior to its maturity date. Accordingly, we do not believe that we have material exposure to interest rate risk arising from our investments. Generally, our investments are not collateralized. We have not realized any significant losses from our investments.
We do not use interest rate derivative instruments to manage exposure to interest rate changes. We ensure the safety and preservation of invested principal funds by limiting default risk, market risk and reinvestment risk. We reduce default risk by investing in investment grade securities.
Foreign Currency Risk
The majority of our transactions and the greatest magnitude of these transactions occur in U.S. dollars and we do not have significant operating subsidiaries or significant investments in foreign countries as of December 31, 2019. Therefore, we are not subject to significant foreign currency exchange risk in our normal operations.
In connection with the issuance by Royalty Sub of the PhaRMA Notes, we entered into a Currency Hedge Agreement to hedge certain risks associated with changes in the value of the Japanese yen relative to the U.S. dollar. Under the Currency Hedge Agreement, we are required to post collateral based on our potential obligations under the Currency Hedge Agreement as determined by periodic mark to market adjustments. Provided the Currency Hedge Agreement remains in effect, we may be required to pay an annual premium in the amount of $2.0 million in May 2020. Such payment will be required if, on May 18, the spot rate of exchange for Japanese yen-U.S. dollars (determined in accordance with the Currency Hedge Agreement) is such that the U.S. dollar is worth 100 yen or less. As of December 31, 2019, the maximum amount of hedge collateral we may be required to post is $2.0 million.
58
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
BIOCRYST PHARMACEUTICALS, INC.
(In thousands, except per share amounts)
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| ASSETS | ||||||||
| Cash and cash equivalents | $ | 114,172 | $ | 26,731 | ||||
| Restricted cash | 1,551 | 1,544 | ||||||
| Investments | 22,054 | 77,736 | ||||||
| Receivables from collaborations | 22,146 | 4,293 | ||||||
| Inventory | - | 1,649 | ||||||
| Prepaid expenses and other current assets | 4,422 | 2,399 | ||||||
| Total current assets | 164,345 | 114,352 | ||||||
| Investments | - | 22,376 | ||||||
| Property and equipment, net | 7,347 | 9,135 | ||||||
| Other assets | 3,590 | 978 | ||||||
| Total assets | $ | 175,282 | $ | 146,841 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Accounts payable | $ | 13,988 | $ | 7,769 | ||||
| Accrued expenses | 21,365 | 15,891 | ||||||
| Interest payable | 14,904 | 11,848 | ||||||
| Deferred collaboration revenue | 2,120 | 221 | ||||||
| Lease financing obligation | 1,377 | 47 | ||||||
| Senior credit facility | 9,020 | 4,580 | ||||||
| Non-recourse notes payable | 29,561 | 29,121 | ||||||
| Total current liabilities | 92,335 | 69,477 | ||||||
| Deferred rent | - | 54 | ||||||
| Lease financing obligation | 3,406 | 2,703 | ||||||
| Senior credit facility | 41,289 | 25,372 | ||||||
| Stockholders’ equity: | ||||||||
| Preferred stock, $0.001 par value; shares authorized — 5,000; no shares outstanding | - | - | ||||||
| Common stock, $0.01 par value; shares authorized — 200,000; shares issued and outstanding — 154,082 at December 31, 2019 and 110,063 at December 31, 2018 | 1,541 | 1,101 | ||||||
| Additional paid-in capital | 877,300 | 780,400 | ||||||
| Accumulated other comprehensive income (loss) | 39 | (297 | ) | |||||
| Accumulated deficit | (840,628 | ) | (731,969 | ) | ||||
| Total stockholders’ equity | 38,252 | 49,235 | ||||||
| Total liabilities and stockholders’ equity | $ | 175,282 | $ | 146,841 | ||||
See accompanying notes to consolidated financial statements.
59
BIOCRYST PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(In thousands, except per share amounts)
| Year Ended December 31, | ||||||||||||
| 2019 | 2018 | 2017 | ||||||||||
| Revenues | ||||||||||||
| Product sales, net | $ | 17,533 | $ | - | $ | 1,501 | ||||||
| Royalty revenue | 6,303 | 6,101 | 10,543 | |||||||||
| Collaborative and other research and development | 24,999 | 14,552 | 13,142 | |||||||||
| Total revenues | 48,835 | 20,653 | 25,186 | |||||||||
| Expenses | ||||||||||||
| Cost of products sold | 3,726 | - | 1,142 | |||||||||
| Research and development | 107,068 | 84,888 | 66,962 | |||||||||
| Selling, general and administrative | 37,121 | 29,514 | 13,933 | |||||||||
| Royalty | 375 | 471 | 560 | |||||||||
| Total operating expenses | 148,290 | 114,873 | 82,597 | |||||||||
| Loss from operations | (99,455 | ) | (94,220 | ) | (57,411 | ) | ||||||
| Interest and other income | 1,933 | 2,252 | 1,015 | |||||||||
| Interest expense | (11,892 | ) | (9,176 | ) | (8,565 | ) | ||||||
| Gain (loss) on foreign currency derivative | 517 | (108 | ) | (821 | ) | |||||||
| Net loss | $ | (108,897 | ) | $ | (101,252 | ) | $ | (65,782 | ) | |||
| Unrealized gain (loss) on available for sale investments | $ | 336 | $ | (54 | ) | $ | (231 | ) | ||||
| Net comprehensive loss | $ | (108,561 | ) | $ | (101,306 | ) | $ | (66,013 | ) | |||
| Basic and diluted net loss per common share | $ | (0.94 | ) | $ | (0.98 | ) | $ | (0.78 | ) | |||
| Weighted average shares outstanding | 115,600 | 103,185 | 84,451 | |||||||||
See accompanying notes to consolidated financial statements.
60
BIOCRYST PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands, except per share amounts)
| Year Ended December 31, | ||||||||||||
| 2019 | 2018 | 2017 | ||||||||||
| Operating activities: | ||||||||||||
| Net loss | $ | (108,897 | ) | $ | (101,252 | ) | $ | (65,782 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 724 | 770 | 704 | |||||||||
| Loss (gain) on disposal of property and equipment | - | 7 | (12 | ) | ||||||||
| Stock-based compensation expense | 17,719 | 9,396 | 12,621 | |||||||||
| Amortization of debt issuance costs | 1,278 | 885 | 876 | |||||||||
| Amortization of premium/discount on investments | 117 | 110 | 157 | |||||||||
| Change in fair value of foreign currency derivative | 347 | 1,049 | 966 | |||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Receivables | (17,853 | ) | 1,824 | 2,651 | ||||||||
| Inventory | 1,649 | (1,649 | ) | 500 | ||||||||
| Prepaid expenses and other assets | (1,364 | ) | (866 | ) | 951 | |||||||
| Accounts payable and accrued expenses | 11,741 | 4,487 | 3,842 | |||||||||
| Interest payable | 3,056 | (247 | ) | 3,105 | ||||||||
| Deferred revenue | 1,899 | (7,079 | ) | (1,722 | ) | |||||||
| Net cash used in operating activities: | (89,584 | ) | (92,565 | ) | (41,143 | ) | ||||||
| Investing activities: | ||||||||||||
| Acquisition of property and equipment | (343 | ) | (366 | ) | (328 | ) | ||||||
| Proceeds from sale of property and equipment | - | - | 12 | |||||||||
| Purchases of investments | (3,018 | ) | (62,614 | ) | (107,787 | ) | ||||||
| Sales and maturities of investments | 81,295 | 67,748 | 43,461 | |||||||||
| Net cash provided by (used in) investing activities: | 77,934 | 4,768 | (64,642 | ) | ||||||||
| Financing activities: | ||||||||||||
| Sale of common stock, net | 58,500 | 53,400 | 134,000 | |||||||||
| Sale of pre-funded warrants | 19,882 | - | - | |||||||||
| Net proceeds from common stock issued under stock-based compensation plans | 1,239 | 2,852 | 1,581 | |||||||||
| Proceeds from senior credit facility | 19,477 | 10,353 | - | |||||||||
| Payment of senior credit facility | - | (4,025 | ) | - | ||||||||
| (Decrease) increase in lease financing obligation | - | (76 | ) | 122 | ||||||||
| Net cash provided by financing activities: | 99,098 | 62,504 | 135,703 | |||||||||
| Increase (decrease) in cash, cash equivalents and restricted cash | 87,448 | (25,293 | ) | 29,918 | ||||||||
| Cash, cash equivalents and restricted cash at beginning of year | 28,275 | 53,568 | 23,650 | |||||||||
| Cash, cash equivalents and restricted cash at end of year | $ | 115,723 | $ | 28,275 | $ | 53,568 | ||||||
See accompanying notes to consolidated financial statements.
61
BIOCRYST PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(In thousands, except per share amounts)
| Common Stock | Additional Paid-In Capital | Accumulated Other Comprehensive (Loss) Income | Accumulated Deficit | Total Stockholders’ Equity | ||||||||||||||||
| Balance at December 31, 2016 | $ | 738 | $ | 566,913 | $ | (12 | ) | $ | (566,061 | ) | $ | 1,578 | ||||||||
| Net loss | - | - | - | (65,782 | ) | (65,782 | ) | |||||||||||||
| Other comprehensive (loss) | - | - | (231 | ) | - | (231 | ) | |||||||||||||
| Exercise of stock options, 609 shares, net | 6 | 1,230 | - | - | 1,236 | |||||||||||||||
| Employee stock purchase plan sales, 95 shares, net | 1 | 344 | - | - | 345 | |||||||||||||||
| Issuance of common stock, 23,925 shares, net | 239 | 133,761 | - | - | 134,000 | |||||||||||||||
| Stock-based compensation expense | - | 12,621 | - | - | 12,621 | |||||||||||||||
| Balance at December 31, 2017 | 984 | 714,869 | (243 | ) | (631,843 | ) | 83,767 | |||||||||||||
| Impact to retained earnings from adoption of ASC 606 | - | - | - | 1,126 | 1,126 | |||||||||||||||
| Net loss | - | - | - | (101,252 | ) | (101,252 | ) | |||||||||||||
| Other comprehensive (loss) | - | - | (54 | ) | - | (54 | ) | |||||||||||||
| Exercise of stock options, 1,106 shares, net | 11 | 2,490 | - | - | 2,501 | |||||||||||||||
| Employee stock purchase plan sales, 92 shares, net | 1 | 350 | - | - | 351 | |||||||||||||||
| Issuance of common stock, 10,455 shares, net | 105 | 53,295 | - | - | 53,400 | |||||||||||||||
| Stock-based compensation expense | - | 9,396 | - | - | 9,396 | |||||||||||||||
| Balance at December 31, 2018 | 1,101 | 780,400 | (297 | ) | (731,969 | ) | 49,235 | |||||||||||||
| Impact to retained earnings from adoption of ASC 842 | - | - | - | 238 | 238 | |||||||||||||||
| Net loss | - | - | - | (108,897 | ) | (108,897 | ) | |||||||||||||
| Other comprehensive income | - | - | 336 | - | 336 | |||||||||||||||
| Exercise of stock options, 283 shares, net | 3 | 832 | - | - | 835 | |||||||||||||||
| Employee stock purchase plan sales, 115 shares, net | 1 | 403 | - | - | 404 | |||||||||||||||
| Issuance of common stock, 43,621 shares, net | 436 | 58,064 | - | - | 58,500 | |||||||||||||||
| Issuance of pre-funded warrants, 11,765 warrants | - | 19,882 | 19,882 | |||||||||||||||||
| Stock-based compensation expense | - | 17,719 | - | - | 17,719 | |||||||||||||||
| Balance at December 31, 2019 | $ | 1,541 | $ | 877,300 | $ | 39 | $ | (840,628 | ) | $ | 38,252 | |||||||||
See accompanying notes to consolidated financial statements.
62
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Note 1 — Significant Accounting Policies and Concentrations of Risk
The Company
BioCryst Pharmaceuticals, Inc. (the “Company”) is a biotechnology company that discovers novel, oral, small-molecule medicines. The Company focuses on the treatment of rare diseases in which significant unmet medical needs exist and an enzyme plays the key role in the biological pathway of the disease. The Company was incorporated in Delaware in 1986 and its headquarters is located in Durham, North Carolina. The Company integrates the disciplines of biology, crystallography, medicinal chemistry and computer modeling to discover and develop small molecule pharmaceuticals through the process known as structure-guided drug design. BioCryst has incurred losses and negative cash flows from operations since inception.
With the funds available at December 31, 2019, the Company believes these resources will be sufficient to fund its operations into 2021. The Company has sustained operating losses for the majority of its corporate history and expects that its 2020 expenses will exceed its 2020 revenues. The Company expects to continue to incur operating losses and negative cash flows until revenues reach a level sufficient to support ongoing operations. Accordingly, its planned operations raise doubt about its ability to continue as a going concern through 2021. The Company’s liquidity needs will be largely determined by the success of operations in regards to the progression of its product candidates in the future. The Company also may consider other plans to fund operations through 2021 including: (1) securing or increasing U.S. Government funding of its programs, including obtaining procurement contracts; (2) out-licensing rights to certain of its products or product candidates, pursuant to which the Company would receive cash milestones; (3) raising additional capital through equity or debt financings or from other sources; (4) obtaining additional product candidate regulatory approvals, which would generate revenue, milestones and cash flow; (5) reducing spending on one or more research and development programs, including by discontinuing development; and/or (6) restructuring operations to change its overhead structure. The Company may issue securities, including common stock, preferred stock, depositary shares, stock purchase contracts, warrants and units, through private placement transactions or registered public offerings in the future. The Company’s future liquidity needs, and ability to address those needs, will largely be determined by the success of its product candidates, timing, scope and magnitude of its commercial expenses and key development and regulatory events and its decisions in the future.
Basis of Presentation
The consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries, JPR Royalty Sub LLC (“Royalty Sub”) and MDCP, LLC (“MDCP”). Both subsidiaries were formed to facilitate financing transactions for the Company. Royalty Sub was formed in connection with a $30,000 financing transaction the Company completed on March 9, 2011. See Note 3, Royalty Monetization, for a further description of this transaction. MDCP was formed in connection with a $23,000 senior credit facility that the Company closed on September 23, 2016 and subsequently amended and restated on each of July 20, 2018 and February 6, 2019. See Note 4 for a further description of these transactions. All intercompany transactions and balances have been eliminated.
The Company’s consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States (“U.S. GAAP”). Such consolidated financial statements reflect all adjustments that are, in management’s opinion, necessary to present fairly, in all material respects, the Company’s consolidated financial position, results of operations, and cash flows. There were no adjustments other than normal recurring adjustments.
Cash and Cash Equivalents
The Company generally considers cash equivalents to be all cash held in commercial checking accounts, certificates of deposit, money market accounts or investments in debt instruments with maturities of three months or less at the time of purchase. The carrying value of cash and cash equivalents approximates fair value due to the short-term nature of these items.
Restricted Cash
Restricted cash as of December 31, 2019 and 2018 reflects $134 and 131, respectively, in royalty revenue paid by Shionogi & Co., Ltd. (“Shionogi”) designated for interest on the PhaRMA Notes (defined in Note 3) and $1,417 and 1,413, respectively, the Company is required to maintain as collateral for a letter of credit associated with the lease execution and build-out of its new Birmingham research facilities.
Investments
The Company invests in high credit quality investments in accordance with its investment policy, which is designed to minimize the possibility of loss. The objective of the Company’s investment policy is to ensure the safety and preservation of invested funds, as well as maintaining liquidity sufficient to meet cash flow requirements. The Company places its excess cash with high credit quality financial institutions, commercial companies, and government agencies in order to limit the amount of its credit exposure. In accordance with its policy, the Company is able to invest in marketable debt securities that may consist of U.S. Government and government agency securities, money market and mutual fund investments, municipal and corporate notes and bonds, commercial paper and asset or mortgage-backed securities, among others. The Company’s investment policy requires it to purchase high-quality marketable securities with a maximum individual maturity of three years and requires an average portfolio maturity of no more than 18 months. Some of the securities the Company invests in may have market risk. This means that a change in prevailing interest rates may cause the principal amount of the investment to fluctuate. To minimize this risk, the Company schedules its investments with maturities that coincide with expected cash flow needs, thus avoiding the need to redeem an investment prior to its maturity date. Accordingly, the Company does not believe it has a material exposure to interest rate risk arising from its investments. Generally, the Company’s investments are not collateralized. The Company has not realized any significant losses from its investments.
63
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
The Company classifies all of its investments as available-for-sale. Unrealized gains and losses on investments are recognized in comprehensive loss, unless an unrealized loss is considered to be other than temporary, in which case the unrealized loss is charged to operations. The Company periodically reviews its investments for other than temporary declines in fair value below cost basis and whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The Company believes the individual unrealized losses represent temporary declines primarily resulting from interest rate changes. Realized gains and losses are reflected in interest and other income in the Consolidated Statements of Comprehensive Loss and are determined using the specific identification method with transactions recorded on a settlement date basis. Investments with original maturities at date of purchase beyond three months and which mature at or less than 12 months from the balance sheet date are classified as current. Investments with a maturity beyond 12 months from the balance sheet date are classified as long-term. At December 31, 2019, the Company believes that the cost of its investments is recoverable in all material respects.
The following tables summarize the fair value of the Company’s investments by type. The estimated fair values of the Company’s fixed income investments are classified as Level 2 in the fair value hierarchy as defined in U.S. GAAP. These valuations are based on observable direct and indirect inputs, primarily quoted prices of similar, but not identical, instruments in active markets or quoted prices for identical or similar instruments in markets that are not active. These fair values are obtained from independent pricing services which utilize Level 2 inputs.
| December 31, 2019 | ||||||||||||||||||||
| Amortized Cost | Accrued Interest | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | ||||||||||||||||
| Obligations of U.S. Government and its agencies | $ | 10,488 | $ | 50 | $ | 23 | $ | - | $ | 10,561 | ||||||||||
| Corporate debt securities | 9,742 | 59 | 10 | (1 | ) | 9,810 | ||||||||||||||
| Certificates of deposit | 1,669 | 7 | 7 | - | 1,683 | |||||||||||||||
| Total investments | $ | 21,899 | $ | 116 | $ | 40 | $ | (1 | ) | $ | 22,054 | |||||||||
| December 31, 2018 | ||||||||||||||||||||
| Amortized Cost | Accrued Interest | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | ||||||||||||||||
| Obligations of U.S. Government and its agencies | $ | 50,613 | $ | 176 | $ | 15 | $ | (131 | ) | $ | 50,673 | |||||||||
| Corporate debt securities | 45,793 | 254 | 4 | (171 | ) | 45,880 | ||||||||||||||
| Certificates of deposit | 3,559 | 14 | - | (14 | ) | 3,559 | ||||||||||||||
| Total investments | $ | 99,965 | $ | 444 | $ | 19 | $ | (316 | ) | $ | 100,112 | |||||||||
64
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
The following table summarizes the scheduled maturity for the Company’s investments at December 31, 2019 and 2018.
| 2019 | 2018 | |||||||
| Maturing in one year or less | $ | 22,054 | $ | 77,736 | ||||
| Maturing after one year through two years | - | 22,376 | ||||||
| Total investments | $ | 22,054 | $ | 100,112 | ||||
Receivables from Collaborations
Receivables from collaborations are recorded for amounts due to the Company related to reimbursable research and development costs from the U.S. Department of Health and Human Services, royalty receivables from Shionogi, Green Cross Corporation (“Green Cross”), Mundipharma International Holdings Limited (“Mundipharma”) and Seqirus UK Limited (“SUL”), and product sales to SUL. These receivables are evaluated to determine if any reserve or allowance should be established at each reporting date.
At December 31, 2019 and December 31, 2018, the Company had the following receivables:
| December 31, 2019 | ||||||||||||
| Billed | Unbilled | Total | ||||||||||
| U.S. Department of Health and Human Services | $ | 1,353 | $ | 15,023 | $ | 16,376 | ||||||
| Shionogi & Co. Ltd. | 1,336 | 4 | 1,340 | |||||||||
| Green Cross Corporation | 2,924 | 8 | 2,932 | |||||||||
| Mundipharma International Holdings Limited | 56 | - | 56 | |||||||||
| Seqirus UK Limited | 1,091 | 351 | 1,442 | |||||||||
| Total receivables | $ | 6,760 | $ | 15,386 | $ | 22,146 | ||||||
| December 31, 2018 | ||||||||||||
| Billed | Unbilled | Total | ||||||||||
| U.S. Department of Health and Human Services | $ | - | $ | 1,525 | $ | 1,525 | ||||||
| Shionogi & Co. Ltd. | 854 | - | 854 | |||||||||
| Green Cross Corporation | 876 | 28 | 904 | |||||||||
| Mundipharma International Holdings Limited | 44 | - | 44 | |||||||||
| Seqirus UK Limited | 940 | 26 | 966 | |||||||||
| Total receivables | $ | 2,714 | $ | 1,579 | $ | 4,293 | ||||||
Monthly invoices are submitted to the U.S. Department of Health and Human Services related to reimbursable research and development costs. The Company is also entitled to monthly reimbursement of indirect costs based on rates stipulated in the underlying contract. The Company’s calculations of its indirect cost rates are subject to audit by the U.S. Government.
Receivables from Product Sales
Receivables from product sales are recorded for amounts due to the Company related to sales of RAPIVAB. These receivables are evaluated to determine if any reserve or allowance should be established at each reporting date.
Inventory
At December 31, 2019 and December 31, 2018, the Company’s inventory consisted primarily of peramivir work in process and is being manufactured for the Company’s partners and the U.S. Government. Inventory is stated at the lower of cost and net realizable value, determined under the first-in, first-out (“FIFO”) method, or market. The Company expenses costs related to the production of inventories as research and development expenses in the period incurred until such time it is believed that future economic benefit is expected to be recognized, which generally is reliant upon receipt of regulatory approval. Upon regulatory approval, the Company will capitalize subsequent costs related to the production of inventories.
65
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Property and Equipment
Property and equipment are recorded at cost. Depreciation is computed using the straight-line method over the estimated useful lives of the assets. Computer equipment is depreciated over a life of three years. Laboratory equipment, office equipment, and software are depreciated over a life of five years. Furniture and fixtures are depreciated over a life of seven years. Leasehold improvements are amortized over their estimated useful lives or the expected lease term, whichever is less. Property consists of a leased building which did not meet the sale-leaseback criteria and is recorded at its fair value, less depreciation. The building is being depreciated over a period equal to the expected term of the related lease.
In accordance with U.S. GAAP, the Company periodically reviews its property and equipment for impairment when events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable. Determination of recoverability is based on an estimate of undiscounted future cash flows resulting from the use of the asset and its eventual disposition. In the event that such cash flows are not expected to be sufficient to recover the carrying amount of the assets, the assets are written down to their estimated fair values. Property and equipment to be disposed of are reported at the lower of carrying amount or fair value less cost to sell.
Patents and Licenses
The Company seeks patent protection on all internally developed processes and products. All patent related costs are expensed to selling, general and administrative expenses when incurred as recoverability of such expenditures is uncertain.
Accrued Expenses
The Company generally enters into contractual agreements with third-party vendors who provide research and development, manufacturing, and other services in the ordinary course of business. Some of these contracts are subject to milestone-based invoicing and services are completed over an extended period of time. The Company records liabilities under these contractual commitments when it determines an obligation has been incurred, regardless of the timing of the invoice. This process involves reviewing open contracts and purchase orders, communicating with applicable Company personnel to identify services that have been performed on its behalf and estimating the level of service performed and the associated cost incurred for the service when the Company has not yet been invoiced or otherwise notified of actual cost. The majority of service providers invoice the Company monthly in arrears for services performed. The Company makes estimates of accrued expenses as of each balance sheet date in its financial statements based on the facts and circumstances. The Company periodically confirms the accuracy of its estimates with the service providers and makes adjustments if necessary. Examples of estimated accrued expenses include:
| • | fees paid to Clinical Research Organizations (“CROs”) in connection with preclinical and toxicology studies and clinical trials; | |||
| • | fees paid to investigative sites in connection with clinical trials; | |||
| • | fees paid to contract manufacturers in connection with the production of the Company’s raw materials, drug substance and drug products; and | |||
| • | professional fees. | |||
The Company bases its expenses related to clinical trials on its estimates of the services received and efforts expended pursuant to contracts with multiple research institutions and CROs that conduct and manage clinical trials on the Company’s behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. Payments under some of these contracts depend on factors such as the successful enrollment of patients and the completion of clinical trial milestones. In accruing service fees, the Company estimates the time period over which services will be performed and the level of effort expended in each period. If the actual timing of the performance of services or the level of effort varies from the estimate, the Company will adjust the accrual accordingly. As of December 31, 2019 and December 31, 2018, the carrying value of accrued expenses approximates their fair value due to their short-term settlement.
66
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Accrued expenses were comprised of the following:
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| Compensation and benefits | $ | 6,190 | $ | 4,659 | ||||
| Development costs | 11,302 | 7,564 | ||||||
| Inventory | 29 | 1,649 | ||||||
| Professional fees | 326 | 118 | ||||||
| Duties and taxes | 67 | 51 | ||||||
| Other | 3,451 | 1,850 | ||||||
| Total accrued expenses | $ | 21,365 | $ | 15,891 | ||||
Income Taxes
The liability method is used in the Company’s accounting for income taxes. Under this method, deferred tax assets and liabilities are determined based on differences between financial reporting and tax bases of assets and liabilities and are measured using the enacted tax rates and laws that are expected to be in effect when the differences are expected to reverse.
Accumulated Other Comprehensive Loss
Accumulated other comprehensive loss is comprised of unrealized gains and losses on available-for-sale investments and is disclosed as a separate component of stockholders’ equity. Amounts reclassified from accumulated other comprehensive loss are recorded as interest and other income on the Consolidated Statements of Comprehensive Loss. No reclassifications out of accumulated other comprehensive loss were recorded during 2019. During 2018, realized losses of $2 were reclassified out of accumulated other comprehensive loss.
Revenue Recognition
Collaborative and Other Research and Development Arrangements and Royalties
The Company recognizes revenue when it satisfies a performance obligation by transferring promised goods or services to a customer. Revenue is measured at the transaction price that is based on the amount of consideration that the Company expects to receive in exchange for transferring the promised goods or services to the customer. The transaction price includes estimates of variable consideration to the extent it is probable that a significant reversal of revenue recognized will not occur.
The Company has collaboration and license agreements with a number of third parties as well as research and development agreements with certain government entities. The Company’s primary sources of revenue are license, service, royalty and product sale revenues from these collaborative and other research and development arrangements.
Revenue from license fees, royalty payments, milestone payments, and research and development fees are recognized as revenue when the earnings process is complete and the Company has no further continuing performance obligations or the Company has completed the performance obligations under the terms of the agreement.
Arrangements that involve the delivery of more than one performance obligation are initially evaluated as to whether the intellectual property licenses granted by the Company represent distinct performance obligations. If they are determined to be distinct, the value of the intellectual property licenses would be recognized up-front while the research and development service fees would be recognized as the performance obligations are satisfied. For performance obligations based on services performed, the Company measures progress using an input method based on the effort we expend or costs we incur toward the satisfaction of performance obligation in relation to the total estimated effort of costs. Variable consideration is assessed at each reporting period as to whether it is not subject to significant future reversal and, therefore, should be included in the transaction price at the inception of the contract. If a contract includes a fixed or minimum amount of research and development support, this also would be included in the transaction price. Changes to collaborations, such as the extensions of the research term or increasing the number of targets or technology covered under an existing agreement, are assessed for whether they represent a modification or should be accounted for as a new contract. For contracts with multiple performance obligations, revenue is allocated to each performance obligation based on its relative standalone selling price. Standalone selling prices are based on observable prices at which the Company separately sells the products or services. If a standalone selling price is not directly observable, then the Company estimates the standalone selling price using either an adjusted market assessment approach or an expected cost plus margin approach, representing the amount that the Company believes the market is willing to pay for the product or service. Analyzing the arrangement to identify performance obligations requires the use of judgment, and each may be an obligation to deliver services, a right or license to use an asset, or another performance obligation.
67
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Milestone payments are recognized as licensing revenue upon the achievement of specified milestones if (i) the milestone is substantive in nature and the achievement of the milestone was not probable at the inception of the agreement; and (ii) the Company has a right to payment. Any milestone payments received prior to satisfying these revenue recognition criteria are recorded as deferred revenue.
Reimbursements received for direct out-of-pocket expenses related to research and development costs are recorded as revenue in the Consolidated Statements of Comprehensive Loss rather than as a reduction in expenses. Under the Company’s contracts with the Biomedical Advanced Research and Development Authority within the United States Department of Health and Human Services (”BARDA/HHS”) and the National Institute of Allergy and Infectious Diseases (“NIAID/HHS”), revenue is recognized as reimbursable direct and indirect costs are incurred.
Under certain of the Company’s license agreements, the Company receives royalty payments based upon its licensees’ net sales of covered products. Royalties are recognized at the later of when (i) the subsequent sale or usage occurs, or (ii) the performance obligation to which some or all of the sales-based or usage-based royalty has been satisfied.
Product Sales
The Company’s principal sources of product sales are sales of peramivir to our licensing partners and sales of RAPIVAB to the U.S. Department of Health and Human Services under the Company’s procurement contract. The Company recognizes revenue for sales when the customer obtains control of the product, which generally occurs upon delivery.
The Company recorded the following revenues for the years ended December 31:
| 2019 | 2018 | 2017 | ||||||||||
| Product sales, net | $ | 17,533 | $ | - | $ | 1,501 | ||||||
| Royalty revenue | 6,303 | 6,101 | 10,543 | |||||||||
| Collaborative and other research and development revenues: | ||||||||||||
| U.S. Department of Health and Human Services | 4,898 | 2,552 | 4,608 | |||||||||
| Torii Pharmaceutical Co., Ltd. | 20,101 | - | - | |||||||||
| Shionogi & Co. Ltd. | - | - | 1,184 | |||||||||
| Seqirus UK Limited | - | 12,000 | 7,350 | |||||||||
| Total collaborative and other research and development revenues | 24,999 | 14,552 | 13,142 | |||||||||
| Total revenues | $ | 48,835 | $ | 20,653 | $ | 25,186 | ||||||
Advertising
The Company engages in very limited distribution and direct-response advertising when promoting RAPIVAB. Advertising and promotional costs are expensed as the costs are incurred. The Company did not incur advertising and product promotion expenses in 2019, 2018 or 2017.
Research and Development Expenses
The Company’s research and development costs are charged to expense when incurred. Research and development expenses include all direct and indirect development costs related to the development of the Company’s portfolio of product candidates. Advance payments for goods or services that will be used or rendered for future research and development activities are deferred and capitalized. Such amounts are recognized as expense when the related goods are delivered or the related services are performed. Research and development expenses include, among other items, personnel costs, including salaries and benefits, manufacturing costs, clinical, regulatory, and toxicology services performed by CROs, materials and supplies, and overhead allocations consisting of various administrative and facilities related costs. Most of the Company’s manufacturing and clinical and preclinical studies are performed by third-party CROs. Costs for studies performed by CROs are accrued by the Company over the service periods specified in the contracts and estimates are adjusted, if required, based upon the Company’s on-going review of the level of services actually performed.
Additionally, the Company has license agreements with third parties, such as Albert Einstein College of Medicine of Yeshiva University (“AECOM”), Industrial Research, Ltd. (“IRL”), and the University of Alabama at Birmingham (“UAB”), which require fees related to sublicense agreements or maintenance fees. The Company expenses sublicense payments as incurred unless they are related to revenues that have been deferred, in which case the expenses are deferred and recognized over the related revenue recognition period. The Company expenses maintenance payments as incurred.
68
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Deferred collaboration expenses represent sub-license payments, paid to the Company’s academic partners upon receipt of consideration from various commercial partners, and other consideration paid to the Company’s academic partners for modification to existing license agreements. These deferred expenses would not have been incurred without receipt of such payments or modifications from the Company’s commercial partners and are being expensed in proportion to the related revenue being recognized. The Company believes that this accounting treatment appropriately matches expenses with the associated revenue.
Stock-Based Compensation
All share-based payments, including grants of stock option awards and restricted stock unit awards, are recognized in the Company’s Consolidated Statements of Comprehensive Loss based on their fair values. The fair value of stock option awards is estimated using the Black-Scholes option pricing model. The fair value of restricted stock unit awards is based on the grant date closing price of the common stock. Stock-based compensation cost is recognized as expense on a straight-line basis over the requisite service period of the award. In addition, we have outstanding performance-based stock options for which no compensation expense is recognized until “performance” is deemed to have occurred.
Interest Expense and Deferred Financing Costs
Interest expense for the years ended December 31, 2019, 2018 and 2017 was $11,892, $9,176 and $8,565, respectively, and primarily relates to the issuance of the PhaRMA Notes (defined in Note 3) and the Prior Credit Facility and Amended and Restated Senior Credit Facility (each defined in Note 4). Costs directly associated with the issuance of the PhaRMA Notes, the Prior Credit Facility and the Amended and Restated Senior Credit Facility have been capitalized and are netted against the non-recourse notes payable and senior credit facility on the Consolidated Balance Sheets. These costs are being amortized to interest expense over the terms of the PhaRMA Notes and the Amended and Restated Senior Credit Facility (as subsequently amended and restated) using the effective interest rate method. Amortization of deferred financing costs and original issue discount included in interest expense was $1,278, $885 and $876 for each of the years ended December 31, 2019, 2018 and 2017, respectively.
Currency Hedge Agreement
In connection with the issuance by Royalty Sub of the PhaRMA Notes, the Company entered into a Currency Hedge Agreement to hedge certain risks associated with changes in the value of the Japanese yen relative to the U.S. dollar. The Currency Hedge Agreement does not qualify for hedge accounting treatment; therefore mark to market adjustments are recognized in the Company’s Consolidated Statements of Comprehensive Loss. Cumulative mark to market adjustments for the years ended December 31, 2019, 2018 and 2017 resulted in losses of $347, $1,049 and $1,787, respectively. Mark to market adjustments are determined by a third-party pricing model which uses quoted prices in markets that are not actively traded and for which significant inputs are observable directly or indirectly, representing Level 2 in the fair value hierarchy as defined by U.S. GAAP. In addition, realized currency exchange gains of $863, $941 and $966 were recognized in 2019, 2018 and 2017, respectively, related to the exercise of a U.S. dollar/Japanese yen currency option under the Company’s foreign currency hedge. The Company is also required to post collateral in connection with the mark to market adjustments based on defined thresholds. As of December 31, 2019 and December 31, 2018, no hedge collateral was posted under the Currency Hedge Agreement.
Net Loss Per Share
Net loss per share is based upon the weighted average number of common shares outstanding during the period. Diluted loss per share is equivalent to basic net loss per share for all periods presented herein because common equivalent shares from unexercised stock options, outstanding warrants, and common shares expected to be issued under the Company’s employee stock purchase plan were anti-dilutive. The calculation of diluted earnings per share for the years ended December 31, 2019, 2018 and 2017 does not include 2,805, 2,274 and 2,067 respectively, of potential common shares as their impact would be anti-dilutive.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses, and related disclosure of contingent assets and liabilities. The most significant estimates in the Company’s consolidated financial statements relate to the valuation of stock options, and the valuation allowance for deferred tax assets resulting from net operating losses. These estimates are based on historical experience and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results could differ from those estimates.
69
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Significant Customers and Other Risks
Significant Customers
Other than royalty revenues, the Company’s primary sources of revenue that have an underlying cash flow stream are the reimbursement of galidesivir (formerly BCX4430) development expenses earned under cost-plus-fixed-fee contracts with BARDA/HHS and NIAID/HHS and sales of RAPIVAB (peramivir injection) under our procurement contract with the Centers for Disease Control and Prevention. The Company relies on BARDA/HHS and NIAID/HHS to reimburse predominantly all of the development costs for its galidesivir program. Accordingly, reimbursement of these expenses represents a significant portion of the Company’s collaborative and other research and development revenues. The completion or termination of the NIAID/HHS and BARDA/HHS galidesivir contracts could negatively impact the Company’s future Consolidated Statements of Comprehensive Loss and Cash Flows. The Company recognizes royalty revenue from the net sales of RAPIACTA by Shionogi; however, the underlying cash flow from these royalty payments, except for Japanese government stockpiling sales, goes directly to pay the interest, and then the principal, on the Company’s non-recourse notes payable. Payment of the interest and the ultimate repayment of principal of these notes will be entirely funded by future royalty payments derived from net sales of RAPIACTA. Further, the Company’s drug development activities are performed by a limited group of third party vendors. If any of these vendors were unable to perform their services, this could significantly impact the Company’s ability to complete its drug development activities.
Risks from Third Party Manufacturing and Distribution Concentration
The Company relies on single source manufacturers for active pharmaceutical ingredient and finished drug product manufacturing of product candidates in development. Delays in the manufacture or distribution of any product could adversely impact the commercial revenue and future procurement stockpiling of the Company’s product candidates in development.
Credit Risk
Cash equivalents and investments are financial instruments which potentially subject the Company to concentration of risk to the extent recorded on the Consolidated Balance Sheets. The Company deposits excess cash with major financial institutions in the United States. Balances may exceed the amount of insurance provided on such deposits. The Company believes it has established guidelines for investment of its excess cash relative to diversification and maturities that maintain safety and liquidity. To minimize the exposure due to adverse shifts in interest rates, the Company maintains a portfolio of investments with an average maturity of approximately 18 months or less. Other than product sale and collaborative partner receivables discussed above, the majority of the Company’s receivables from collaborations are due from the U.S. Government, for which there is no assumed credit risk.
Recent Accounting Pronouncements
In February 2016, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update No. 2016-02: Leases (Topic 842) (“ASU 2016-02”). The amendments in this update require lessees, among other things, to recognize right-of-use assets and lease liabilities on the balance sheet for all leases with terms greater than 12 months. This update also introduces new disclosure requirements for leasing arrangements. In July 2018, the FASB issued Accounting Standards Update No. 2018-11: Targeted Improvements to Leases (“ASU 2018-11”), which provides companies with an additional transition method that allows the effects of the adoption of the new standard to be recognized as a cumulative-effect adjustment to the opening balance of retained earnings in the period of adoption. The Company adopted ASU 2016-02 as of January 1, 2019 using the optional transition method set forth in ASU 2018-11.
ASU 2016-02 provides a number of optional practical expedients in transition. The Company elected the ‘package of practical expedients’, which permits it to not reassess under the new standard its prior conclusions about lease identification, lease classification and initial direct costs. The Company also elected the short-term lease recognition exemption for all leases that qualify. For those leases that qualify, the Company will not recognize right of use assets or lease liabilities, and this includes not recognizing right of use assets or lease liabilities for existing short-term leases of those assets in transition.
The most significant effects of adoption relate to (1) the recognition of new right of use assets and lease liabilities on its balance sheet for real estate operating leases; and (2) providing significant new disclosures about its leasing activities. For additional detail, see Note 5, Lease Obligations and Other Contingencies.
70
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
In June 2016, the FASB issued Accounting Standards Update No. 2016-13, Financial Instruments - Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments (“ASU 2016-13”) .ASU 2016-13 requires a financial asset (or a group of financial assets) measured at amortized cost basis to be presented at the net amount expected to be collected. In addition, ASU 2016-13 requires credit losses relating to available-for-sale debt securities to be recorded through an allowance for credit losses. ASU 2016-13 requires consideration of a broader range of reasonable and supportable information to develop credit loss estimates. ASU 2016-13 is effective for public companies for interim and annual periods beginning after December 15, 2019. The adoption of ASU 2016-13 is not expected to have a material effect on the Company's financial position, results of operations or cash flows.
In August 2018, the FASB issued Accounting Standards Update No. 2018-15, Intangibles - Goodwill and Other - Internal-Use Software (Subtopic 350-40) (“ASU 2018-15”). ASU 2018-15 aligns the requirements for capitalizing implementation costs incurred in a hosting arrangement that is a service contract with the requirements for capitalizing implementation costs incurred to develop or obtain internal-use software. The guidance requires entities to capitalize costs for certain implementation activities in the application development stage and expense the capitalized implementation costs over the expected term of the hosting arrangement. ASU 2018-15 is effective for fiscal years beginning after December 15, 2019, with early adoption permitted. The Company elected to adopt this standard early, beginning October 1, 2019 on a prospective basis. Adoption did not have a material effect on the Company's financial position, results of operations or cash flows.
Note 2 — Property and Equipment
Property and equipment consisted of the following at December 31:
| 2019 | 2018 | |||||||
| Furniture and fixtures | $ | 602 | $ | 573 | ||||
| Office equipment | 184 | 152 | ||||||
| Software | 1,159 | 1,125 | ||||||
| Laboratory equipment | 3,462 | 3,329 | ||||||
| Leased equipment | - | 143 | ||||||
| Leasehold improvements | 8,528 | 8,413 | ||||||
| Building | - | 1,495 | ||||||
| 13,935 | 15,230 | |||||||
| Less accumulated depreciation and amortization | (6,588 | ) | (6,095 | ) | ||||
| Property and equipment, net | $ | 7,347 | $ | 9,135 | ||||
Depreciation and amortization expense for the years ended December 31, 2019, 2018 and 2017 was $724, $770 and $704, respectively.
Note 3—Royalty Monetization
Overview
On March 9, 2011, the Company completed a $30,000 financing transaction to monetize certain future royalty and milestone payments under the Shionogi Agreement, pursuant to which Shionogi licensed from the Company the rights to market RAPIACTA in Japan and Taiwan. The Company received net proceeds of $22,691 from the transaction after transaction costs of $4,309 and the establishment of a $3,000 interest reserve account by Royalty Sub, available to help cover interest shortfalls in the future. All of the interest reserve account has been fully utilized with the September 2012 interest payment.
As part of the transaction, the Company entered into a purchase and sale agreement dated as of March 9, 2011 with Royalty Sub, whereby the Company transferred to Royalty Sub, among other things, (i) its rights to receive certain royalty and milestone payments from Shionogi arising under the Shionogi Agreement, and (ii) the right to receive payments under a Japanese yen/US dollar foreign currency hedge arrangement (as further described below, the “Currency Hedge Agreement”) put into place by the Company in connection with the transaction. Royalty payments will be paid by Shionogi in Japanese yen and milestone payments will paid in U.S. dollars. The Company’s collaboration with Shionogi was not impacted as a result of this transaction.
71
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Non-Recourse Notes Payable
On March 9, 2011, Royalty Sub completed a private placement to institutional investors of $30,000 in aggregate principal amount of its PhaRMA Senior Secured 14.0% Notes due 2020 (the “PhaRMA Notes”). The PhaRMA Notes were issued by Royalty Sub under an Indenture, dated as of March 9, 2011 (the “Indenture”), by and between Royalty Sub and U.S. Bank National Association, as Trustee. Principal and interest on the PhaRMA Notes issued are payable from, and are secured by, the rights to royalty and milestone payments under the Shionogi Agreement transferred by the Company to Royalty Sub and payments, if any, made to Royalty Sub under the Currency Hedge Agreement. The PhaRMA Notes bear interest at 14% per annum, payable annually in arrears on September 1st of each year. The Company remains entitled to receive any royalties and milestone payments related to sales of peramivir by Shionogi following repayment of the PhaRMA Notes.
Royalty Sub’s obligations to pay principal and interest on the PhaRMA Notes are obligations solely of Royalty Sub and are without recourse to any other person, including the Company, except to the extent of the Company’s pledge of its equity interests in Royalty Sub in support of the PhaRMA Notes. The Company may, but is not obligated to, make capital contributions to a capital account that may be used to redeem, or on up to one occasion pay any interest shortfall on, the PhaRMA Notes.
In September 2014, Royalty Sub was unable to pay the accrued interest obligation due September 3, 2013. Under the terms of the Indenture, Royalty Sub’s inability to pay the full amount of interest payable in September 2013 by the next succeeding Payment Date for the PhaRMA Notes, which was September 1, 2014, constituted an event of default. Accordingly, the PhaRMA Notes and related accrued interest have been classified as current liabilities on the December 31, 2014 balance sheet, and thereafter. As a result of the event of default under the PhaRMA Notes, the holders of the PhaRMA Notes may pursue acceleration of the PhaRMA Notes, may foreclose on the collateral securing the PhaRMA Notes and the equity interest in Royalty Sub and exercise other remedies available to them under the Indenture in respect of the PhaRMA Notes. In such event, the Company may not realize the benefit of future royalty payments that might otherwise accrue to it following repayment of the PhaRMA Notes and it might otherwise be adversely affected. Due to the non-recourse nature of the PhaRMA Notes, in the event of any potential acceleration or foreclosure, the primary impact to the Company would be the loss of future royalty payments from Shionogi and legal costs associated with retiring the PhaRMA Notes. In addition, the Company may incur costs associated with liquidating the related Currency Hedge Agreement, which would no longer be required in the event of foreclosure, or if the PhaRMA Notes cease to be outstanding. As the PhaRMA Notes are the obligation of Royalty Sub and non-recourse to the Company, the event of default of the PhaRMA Notes is not expected to have a significant impact on the Company’s future results of operations or cash flows. As of December 31, 2019, the PhaRMA Notes remain in default.
The Indenture does not contain any financial covenants. The Indenture includes customary representations and warranties of Royalty Sub, affirmative and negative covenants of Royalty Sub, Events of Default and related remedies, and provisions regarding the duties of the Trustee, indemnification of the Trustee, and other matters typical for indentures used in structured financings of this type.
As of December 31, 2019, the aggregate fair value of the PhaRMA Notes was estimated to be approximately 7% of its carrying value of $30,000. The estimated fair value of the PhaRMA Notes is classified as Level 3 in the fair value hierarchy as defined in U.S. GAAP.
The PhaRMA Notes are redeemable at the option of Royalty Sub at any time at a redemption price equal to the outstanding principal balance of the PhaRMA Notes being redeemed plus accrued and unpaid interest through the redemption date on the PhaRMA Notes being redeemed.
Foreign Currency Hedge
In connection with the issuance by Royalty Sub of the PhaRMA Notes, the Company entered into a Currency Hedge Agreement to hedge certain risks associated with changes in the value of the Japanese yen relative to the U.S. dollar. Under the Currency Hedge Agreement, the Company has the right to purchase dollars and sell yen at a rate of 100 yen per dollar for which the Company may be required to pay a premium in 2020, provided the Currency Hedge Agreement remains in effect. A payment of $1,950 will be required if, on May 18, the U.S. dollar is worth 100 yen or less as determined in accordance with the Currency Hedge Agreement.
The Currency Hedge Agreement does not qualify for hedge accounting treatment; therefore mark to market adjustments are recognized in the Company’s Consolidated Statements of Comprehensive Loss. Cumulative mark to market adjustments in 2019, 2018 and 2017 resulted in losses of $347, $1,049 and $1,787, respectively. In addition, realized currency exchange gains of $863, $941 and $966 were recognized in 2019, 2018 and 2017, respectively, related to the exercise of a U.S. dollar/Japanese yen currency option under the Company’s foreign currency hedge. The Company is also required to post collateral in connection with the mark to market adjustments based on defined thresholds. As of December 31, 2019 and 2018, no collateral was posted under the Currency Hedge Agreement. The Company will not be required at any time to post collateral exceeding the maximum premium payments remaining payable under the Currency Hedge Agreement. As of December 31, 2019, the maximum amount of hedge collateral the Company may be required to post is $1,950.
72
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Note 4 — Senior Credit Facility
On February 5, 2019, the Company entered into a $100,000 Senior Credit Facility with an affiliate of MidCap Financial Services, LLC, as administrative agent (the “Second Amended and Restated Senior Credit Facility”). Borrowings under the Second Amended and Restated Senior Credit Facility will be available in three tranches, with (i) the first tranche comprised of $50,000 funded at closing, which includes $30,000 of proceeds that were deemed rolled over from the outstanding principal amount under the Company’s prior credit agreement, (ii) the second tranche to be comprised of $30,000, and (iii) the third tranche to be comprised of $20,000, with the second and third tranches to be funded upon the completion of certain contingencies related to the Company’s development activities of its product candidates and the establishment of certain financial covenants. On September 10, 2019 the Company executed the first amendment to the Second Amended and Restated Credit Facility which extended the commitment termination date for the second tranche to November 30, 2019. On November 30, 2019, the Company’s access to the second tranche expired.
The Second Amended and Restated Senior Credit Facility refinanced and replaced the Amended and Restated Senior Credit Facility dated as of July 20, 2018 (the “Amended and Restated Senior Credit Facility”). The Second Amended and Restated Senior Credit Facility bears a variable interest rate of LIBOR (which shall not be less than 0.5%) plus 8%. The Second Amended and Restated Senior Credit Facility includes an interest-only payment period through June 2020 and scheduled monthly principal and interest payments for the subsequent 30 months. The Company used a portion of the proceeds of the Second Amended and Restated Senior Credit Facility to pay off outstanding amounts under the Amended and Restated Senior Credit Facility and the remainder will be used for general corporate purposes. Under the Second Amended and Restated Senior Credit Facility, the Company must maintain a minimum cash balance of $25,000 of unrestricted cash at all times.
As of December 31, 2019, the Company had borrowings of $50,000 under the Second Amended and Restated Senior Credit Facility bearing an interest rate of 9.7%. The carrying amount of the debt approximates its fair value based on prevailing interest rates as of the balance sheet date. The remaining scheduled principal repayments of the Amended and Restated Senior Credit Facility are as follows:
| Principal Payments | ||||
| 2020 | $ | 10,000 | ||
| 2021 | 20,000 | |||
| 2022 | 20,000 | |||
| Total | $ | 50,000 | ||
The debt agreement contains two provisions that if deemed probable would create the recognition of an embedded feature; however, we do not believe either provision is probable.
Note 5 — Lease Obligations and Other Contingencies
In February 2016, the FASB issued ASU 2016-02: Leases (Topic 842). This ASU requires a lessee to recognize a right-of-use asset and a lease liability on its balance sheet for most operating leases. ASU 2016-02 is effective for annual and interim periods beginning after December 15, 2018, including interim periods within those fiscal years. In July 2018, the FASB issued ASU 2018-11, Leases (Topic 842): Targeted Improvements , which provides companies with an additional optional transition method to apply the new standard to leases in effect at the adoption date through a cumulative effect adjustment. The Company adopted the new lease standard as of January 1, 2019 using this optional transition method.
The Company elected the package of practical expedients referenced in ASU 2016-02, which permits companies to retain original lease identification and classification without reassessing initial direct costs for existing leases. The Company also elected the practical expedient that exempts leases with an initial lease term of twelve months or less, as well as the practical expedient that allows companies to select, by class of underlying asset, not to separate lease and non-lease components. Adoption of this standard resulted in the recognition of a right-of-use asset and a lease liability on the Company’s January 1, 2019 Consolidated Balance Sheet of $3,621 and $4,822, respectively. There was no material impact on the Company’s Consolidated Statement of Comprehensive Loss, and the cumulative transition adjustment recorded to retained earnings upon adoption was $238.
The Company leases certain assets under operating leases, which primarily consisted of real estate leases, laboratory equipment leases and office equipment leases at December 31, 2018. Certain operating leases provide for renewal options, which can vary by lease. The right-of-use asset and lease liabilities on the Company’s Consolidated Balance Sheet represent payments over the lease term, which includes renewal options for certain real estate leases that we are likely to exercise. As part of the Company’s assessment of the lease term, the Company elected the hindsight practical expedient, which allows companies to use current knowledge and expectations when determining the likelihood to extend lease options. Renewal options for our leases range from 1 to 5 years in length and begin from 2023 through 2026. The weighted average lease term for the Company’s operating leases was 13.3 years. The discount rate used in the calculation of the Company’s right-of-use asset and lease liability was determined based on the stated rate within each contract when available, or the Company’s collateralized borrowing rate from lending institutions. The weighted average discount rate for the Company’s operating leases was 12.7%.
73
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
The Company has not made any residual value guarantees related to its operating leases; therefore, the Company has no corresponding liability recorded on its Consolidated Balance Sheets.
Aggregate lease expense under operating leases was $1,464 for the twelve month period ended December 31, 2019. Certain operating leases include rent escalation provisions, which the Company recognizes as expense on a straight-line basis. Lease expense for leases with an initial term of twelve months or less was not material.
Future lease payments for assets under operating leases as of December 31, 2019, are as follows:
| Remaining Maturities of Lease Liabilities Year Ending December 31, | Operating Leases | |||
| 2020 | $ | 1,465 | ||
| 2021 | 595 | |||
| 2022 | 531 | |||
| 2023 | 515 | |||
| 2024 | 518 | |||
| Thereafter | 7,249 | |||
| Total lease payments | 10,873 | |||
| Less imputed interest | 6,090 | |||
| Total | $ | 4,783 | ||
Of the Company’s total lease liability, $1,377 is a current liability and $3,406 is a long-term liability at December 31, 2019. The current and long-term portions of the Company’s lease liability are presented within “Accrued expenses” and “Other non-current liabilities” on the Consolidated Balance Sheets. Cash paid for amounts included in the measurement of lease liabilities was $1,457 for the year-ended December 31, 2019. The Company’s right-of use asset balance associated with operating leases totaled $3,590 at December 31, 2019. This amount is presented within “Other long-term assets” on the Consolidated Balance Sheets. Operating right-of-use assets are recorded net of accumulated amortization of $1,386 as of December 31, 2019, which was presented within Depreciation and amortization on the Consolidated Statements of Cash Flows.
Note 6 — Stockholders’ Equity
Sales of Common Stock
On November 8, 2017, the Company filed a $200,000 shelf registration statement on Form S-3 with the SEC. This shelf registration statement became effective on December 12, 2017 and allows the Company to sell securities, including common stock, preferred stock, depository shares, stock purchase contracts, warrants and units, from time to time at prices and on terms to be determined at the time of sale.
On August 6, 2018, the Company completed an underwritten public offering of 10,455 shares of its common stock, offered at a price to the public of $5.50 per share, including shares issued pursuant to the underwriters’ 30-day option to purchase additional shares, which was exercised in full. The net proceeds from this offering were approximately $53,400 after deducting underwriting discounts and commissions and estimated offering expenses.
On November 18, 2019, the Company completed an underwritten public offering of 43,621 shares of its common stock, offered at a price to the public of $1.45 per share, including shares issued pursuant to the underwriters’ 30-day option to purchase additional shares, which was exercised in full. The net proceeds from this offering were approximately $53,500 after deducting underwriting discounts and commissions and estimated offering expenses.
74
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
On November 21, 2019, the Company completed an offering of pre-funded warrants to purchase up to 11,765 shares of its common stock at a price of $1.69 per warrant. Each pre-funded warrant is exercisable at the holder’s option into 1 share of common stock at an exercise price of $0.01 per share. The net proceeds from this offering were $19,882, excluding any proceeds the Company may receive upon the subsequent exercise of the pre-funded warrants. All warrants issued in this offering remain outstanding at December 31, 2019.
Note 7 — Stock-Based Compensation
As of December 31, 2019, the Company had three stock-based employee compensation plans, the Amended and Restated Stock Incentive Plan (“Incentive Plan”), the Inducement Equity Incentive Plan (“Inducement Plan”) and the Employee Stock Purchase Plan (“ESPP”). The Incentive Plan was amended and restated on April 19, 2019 and approved by the Company’s stockholders on May 29, 2019. The Inducement Plan was adopted by the Board of Directors on April 24, 2019. The ESPP was amended and restated in March 2014 and approved by the Company’s stockholders in May 2014. Stock-based compensation expense of $17,719 ($17,164 of expense related to the Incentive Plan, $323 of expense related to the Inducement Plan, $232 of expense related to the ESPP) was recognized during 2019, while $9,396 ($9,223 of expense related to the Incentive Plan, $173 of expense related to the ESPP) was recognized during 2018, and $12,621 ($12,421 of expense related to the Incentive Plan, $200 of expense related to the ESPP) was recognized during 2017.
The Company accounts for stock-based compensation in accordance with FASB authoritative guidance regarding share-based payments. Total stock-based compensation was allocated as follows:
| Year Ended December 31, | ||||||||||||
| 2019 | 2018 | 2017 | ||||||||||
| Research and development | $ | 13,977 | $ | 6,867 | $ | 9,602 | ||||||
| General and administrative | 3,742 | 2,529 | 3,019 | |||||||||
| Total stock-based compensation expense | $ | 17,719 | $ | 9,396 | $ | 12,621 | ||||||
Stock Incentive Plan
The Company grants stock option awards and restricted stock unit awards to its employees, directors, and consultants under the Incentive Plan. Under the Incentive Plan, stock option awards are granted with an exercise price equal to the market price of the Company’s stock at the date of grant. Commencing March 1, 2011, stock option awards and restricted stock units granted to employees generally vest 25% each year until fully vested after four years. In August 2013, December 2014 and December 2019, the Company issued 1,032, 1,250 and 315 performance-based stock options, respectively. These awards vest upon successful completion of specific development milestones. As of December 31, 2019, 75% of the August 2013 grants have vested based upon achievement of three milestones. As of December 31, 2019, 30% of the December 2014 grants have vested. As of December 31, 2019, none of the December 2019 grants have vested and no compensation expense has been recognized. During 2019, the Company recognized $4,998 of stock compensation expense related to two milestones within the December 2014 grants for which achievement became probable. Stock option awards granted to non-employee directors of the Company generally vest over one year. All stock option awards have contractual terms of 10 years. The vesting exercise provisions of all awards granted under the Incentive Plan are subject to acceleration in the event of certain stockholder-approved transactions, or upon the occurrence of a change in control as defined in the Incentive Plan.
75
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Related activity under the Incentive Plan is as follows:
| Awards Available | Options Outstanding | Weighted Average Exercise Price | ||||||||||
| Balance at December 31, 2016 | 2,273 | 12,095 | $ | 6.55 | ||||||||
| Plan amendment | 1,000 | - | - | |||||||||
| Restricted stock awards granted | (22 | ) | - | - | ||||||||
| Restricted stock awards cancelled | 12 | - | - | |||||||||
| Stock option awards granted | (3,915 | ) | 3,915 | 5.33 | ||||||||
| Stock option awards exercised | - | (438 | ) | 3.50 | ||||||||
| Stock option awards cancelled | 1,120 | (1,120 | ) | 9.72 | ||||||||
| Balance at December 31, 2017 | 468 | 14,452 | 6.06 | |||||||||
| Plan amendment | 4,400 | - | - | |||||||||
| Restricted stock awards granted | (13 | ) | - | - | ||||||||
| Stock option awards granted | (4,272 | ) | 4,272 | 7.15 | ||||||||
| Stock option awards exercised | - | (1,011 | ) | 2.92 | ||||||||
| Stock option awards cancelled | 222 | (222 | ) | 7.44 | ||||||||
| Balance at December 31, 2018 | 805 | 17,491 | 6.49 | |||||||||
| Plan amendment | 4,000 | - | - | |||||||||
| Restricted stock awards granted | (27 | ) | - | - | ||||||||
| Stock option awards granted | (4,511 | ) | 4,511 | 3.91 | ||||||||
| Stock option awards exercised | - | (251 | ) | 3.75 | ||||||||
| Stock option awards cancelled | 701 | (701 | ) | 6.82 | ||||||||
| Balance at December 31, 2019 | 968 | 21,050 | $ | 5.96 | ||||||||
For stock option awards granted under the Incentive Plan during 2019, 2018, and 2017, the fair value was estimated on the date of grant using a Black-Scholes option pricing model and the assumptions noted in the table below. The weighted average grant date fair value of these awards granted during 2019, 2018, and 2017 was $2.63, $4.92 and $3.63, respectively. The fair value of the stock option awards is amortized to expense over the vesting periods using a straight-line expense attribution method. The following explanations describe the assumptions used by the Company to value the stock option awards granted during 2019, 2018, and 2017. The expected life is based on the average of the assumption that all outstanding stock option awards will be exercised at full vesting and the assumption that all outstanding stock option awards will be exercised at the midpoint of the current date (if already vested) or at full vesting (if not yet vested) and the full contractual term. The expected volatility represents the volatility over the most recent period corresponding with the expected life. The Company has assumed no expected dividend yield, as dividends have never been paid to stockholders and will not be for the foreseeable future. The weighted average risk-free interest rate is the implied yield currently available on zero-coupon government issues with a remaining term equal to the expected term.
Inducement Equity Incentive Plan
The Company has the ability to grant stock option awards to newly-hired employees as inducements material to each employee entering employment with the Company. Stock option awards granted to newly hired employees generally vest 25% each year until fully vested after four years. Each stock option has a term of 10 years and is subject to the terms and conditions of the Inducement Equity Incentive Plan. The vesting and exercise provisions of all awards granted under the Inducement Equity Incentive Plan are subject to acceleration in the event of certain stockholder-approved transactions, or upon the occurrence of a change in control as defined in the Inducement Equity Incentive Plan.
During 2019, the Company’s Board of Directors approved the issuance of 1,379 options to newly hired employees. The options were granted with an exercise price equal to the Company’s market price on the respective dates of grant. The fair value was estimated on the date of grant using a Black-Scholes option pricing model and the assumptions noted in the table below. The weighted average grant date fair value of these stock option awards was $2.41. The weighted-average exercise price of the inducement grants issued during 2019 was $3.58. As of December 31, 2019, 1,329 of those options are outstanding.
76
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
The following table summarizes the key assumptions used by the Company to value the stock option awards granted during 2019, 2018 and 2017, respectively. The expected life is based on the average of the assumption that all outstanding stock option awards will be exercised at full vesting and the assumption that all outstanding stock option awards will be exercised at the midpoint of the current date (if already vested) or at full vesting (if not yet vested) and the full contractual term. The expected volatility represents the historical volatility on the Company’s publicly traded common stock. The Company has assumed no expected dividend yield, as dividends have never been paid to stock or option holders and will not be paid for the foreseeable future. The weighted average risk-free interest rate is the implied yield currently available on zero-coupon government issues with a remaining term equal to the expected term.
Weighted Average Assumptions for Stock Option Awards Granted to Employees and Directors under the Plans
| 2019 | 2018 | 2017 | ||||||||||
| Expected Life | 5.5 | 5.5 | 5.5 | |||||||||
| Expected Volatility | 81 | % | 82 | % | 82 | % | ||||||
| Expected Dividend Yield | 0.0 | % | 0.0 | % | 0.0 | % | ||||||
| Risk-Free Interest Rate | 1.8 | % | 2.7 | % | 2.0 | % | ||||||
The total intrinsic value of stock option awards exercised under the Incentive Plan was $1,127 during 2019, $4,504 during 2018 and $1,964 during 2017. The intrinsic value represents the total proceeds (fair market value at the date of exercise, less the exercise price, times the number of stock option awards exercised) received by all individuals who exercised stock option awards during the period. No stock option awards were exercised under the Inducement Plan in 2019.
The following table summarizes, at December 31, 2019, by price range: (1) for stock option awards outstanding under the Incentive Plan, the number of stock option awards outstanding, their weighted average remaining life and their weighted average exercise price; and (2) for stock option awards exercisable under the Plan, the number of stock option awards exercisable and their weighted average exercise price:
| Outstanding | Exercisable | ||||||||||||||||||||
| Range | Number |
Weighted Average Remaining Life |
Weighted Average Exercise Price |
Number |
Weighted Average Exercise Price | ||||||||||||||||
| $ | 0 | to | 3 | 1,929 | 5.5 | $ | 2.02 | 1,159 | $ | 1.64 | |||||||||||
| 3 | to | 6 | 11,724 | 7.2 | 4.17 | 5,085 | 4.55 | ||||||||||||||
| 6 | to | 9 | 4,941 | 7.7 | 7.14 | 1,830 | 6.98 | ||||||||||||||
| 9 | to | 12 | 3,086 | 5.3 | 10.78 | 2,013 | 10.91 | ||||||||||||||
| 12 | to | 15 | 604 | 5.1 | 12.29 | 509 | 12.31 | ||||||||||||||
| 15 | to | 18 | 95 | 5.5 | 15.39 | 95 | 15.39 | ||||||||||||||
| $ | 0 | to | 18 | 22,379 | 6.9 | $ | 5.82 | 10,691 | $ | 6.31 | |||||||||||
The weighted average remaining contractual life of stock option awards exercisable under the plans at December 31, 2019 was 4.9 years.
The aggregate intrinsic value of stock option awards outstanding and exercisable under the plans at December 31, 2019 was $3,926. The aggregate intrinsic value represents the value (the period’s closing market price, less the exercise price, times the number of in-the-money stock option awards) that would have been received by all stock option award holders under the plans had they exercised their stock option awards at the end of the year.
The total fair value of the stock option awards vested under the plans was $12,499 during 2019, $8,952 during 2018 and $9,310 during 2017.
As of December 31, 2019, the number of stock option awards vested and expected to vest under the plans is 20,324. The weighted average exercise price of these stock option awards is $5.83 and their weighted average remaining contractual life is 6.9 years.
77
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
The following table summarizes the changes in the number and weighted-average grant-date fair value of non-vested stock option awards during 2019:
| Non-Vested Stock Option Awards | Weighted Average Grant-Date Fair Value | |||||||
| Balance December 31, 2018 | 9,295 | $ | 4.78 | |||||
| Stock option awards granted | 5,891 | 2.58 | ||||||
| Stock option awards vested | (2,794 | ) | 4.48 | |||||
| Stock option awards forfeited | (704 | ) | 4.38 | |||||
| Balance December 31, 2019 | 11,688 | $ | 3.77 | |||||
As of December 31, 2019, there was approximately $26,552 of total unrecognized compensation cost related to non-vested employee stock option awards granted by the Company. That cost is expected to be recognized as follows: $9,746 in 2020, $7,950 in 2021, $6,743 in 2022 and $2,113 in 2023.
Employee Stock Purchase Plan
The Company has reserved a total of 1,475 shares of common stock to be purchased under the ESPP, of which 119 shares remain available for purchase at December 31, 2019. Eligible employees may authorize up to 15% of their salary to purchase common stock at the lower of 85% of the beginning or 85% of the ending price during six-month purchase intervals. No more than 3 shares may be purchased by any one employee at the six-month purchase dates and no employee may purchase stock having a fair market value at the commencement date of $25 or more in any one calendar year.
There were 115, 92 and 95 shares of common stock purchased under the ESPP in 2019, 2018, and 2017, respectively, at a weighted average price per share of $3.51, $3.83 and $3.61, respectively. Expense of $232, $173 and $200 related to the ESPP was recognized during 2019, 2018, and 2017, respectively. Compensation expense for shares purchased under the ESPP related to the purchase discount and the “look-back” option were determined using a Black-Scholes option pricing model. The weighted average grant date fair values of shares granted under the ESPP during 2019, 2018, and 2017, were $2.01, $1.89 and $2.18, respectively.
Note 8 — Income Taxes
The Company has incurred net losses since inception and, consequently, has not recorded any U.S. Federal and state income tax expense or benefit. The differences between the Company’s effective tax rate and the statutory tax rate in 2019, 2018, and 2017 are as follows:
| 2019 | 2018 | 2017 | ||||||||||
| Income tax benefit at federal statutory rate (21% for 2019, 2018 and 2017) | $ | (22,868 | ) | $ | (21,263 | ) | $ | (23,024 | ) | |||
| State and local income taxes net of federal tax benefit | (1,591 | ) | (2,547 | ) | (1,611 | ) | ||||||
| Permanent items | 691 | 503 | 910 | |||||||||
| Rate change | 625 | (29 | ) | 71,155 | ||||||||
| Expiration of attribute carryforwards | 3,976 | 2,183 | 918 | |||||||||
| Effect of ASU 2016-09 | - | - | (5,949 | ) | ||||||||
| Research and development tax credits | (4,938 | ) | (4,905 | ) | (1,977 | ) | ||||||
| Orphan drug credit | - | - | 564 | |||||||||
| Other | 281 | 18 | 1,639 | |||||||||
| Change in valuation allowance | 23,824 | 26,040 | (42,625 | ) | ||||||||
| Income tax expense | $ | - | $ | - | $ | - | ||||||
In December 2017, the Tax Cuts and Jobs Act (“TCJA”), was signed into law. Among other things, the TCJA permanently lowers the corporate federal income tax rate to 21% from the existing maximum rate of 35%, effective for tax years including or commencing January 1, 2018. As a result of the reduction of the corporate federal income tax rate to 21%, U.S. GAAP requires companies to revalue their deferred tax assets and deferred tax liabilities as of the date of enactment, with the resulting tax effects accounted for in the reporting period of enactment. This revaluation resulted in a provision of $73,474 to income tax expense in continuing operations and a corresponding reduction in the valuation allowance. As a result, there was no impact on the Company’s Consolidated Statements of Operations from the reduction in tax rate.
78
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
The Company adopted ASU 2016-09 during the quarter ended March 31, 2017. As a result of the adoption, the net federal and state operating losses deferred tax assets increased by $5,949 and were offset by a corresponding increase in the valuation allowance. The adoption of ASU 2016-09 had no impact on the Company’s Consolidated Balance Sheets or Consolidated Statements of Operations.
The Company recognizes the impact of a tax position in its financial statements if it is more likely than not that the position will be sustained on audit based on the technical merits of the position. The Company has concluded that it has an uncertain tax position pertaining to its research and development and orphan drug credit carryforwards. The Company has established these credits based on information and calculations it believes are appropriate and the best estimate of the underlying credit. Any changes to the Company’s unrecognized tax benefits are offset by an adjustment to the valuation allowance and there would be no impact on the Company’s financial statements. The Company does not expect its unrecognized tax benefits to change significantly over the next 12 months.
A reconciliation of the beginning and ending amount of unrecognized tax benefits is as follows:
| 2019 | 2018 | |||||||
| Balance at January 1, | $ | 5,976 | $ | 4,750 | ||||
| Additions to current period tax positions | 1,234 | 1,226 | ||||||
| Additions to prior period tax positions | - | - | ||||||
| Reductions to prior period tax provisions | - | - | ||||||
| Balance at December 31, | $ | 7,210 | $ | 5,976 | ||||
The Company’s ability to utilize the net operating loss and tax credit carryforwards in the future may be subject to substantial restrictions in the event of past or future ownership changes as defined in Section 382 of the Internal Revenue Code of 1986, as amended and similar state tax law.
Significant components of the Company’s deferred tax assets and liabilities are as follows:
| 2019 | 2018 | |||||||
| Deferred tax assets: | ||||||||
| Net federal and state operating losses | $ | 155,190 | $ | 137,234 | ||||
| Research and development credits | 63,275 | 59,509 | ||||||
| Stock-based compensation | 9,786 | 7,108 | ||||||
| Leasing obligations | 1,070 | - | ||||||
| Other | 3,801 | 5,258 | ||||||
| Total deferred tax assets | 233,122 | 209,109 | ||||||
| Deferred tax liabilities: | ||||||||
| Fixed assets | (114 | ) | (418 | ) | ||||
| Right of use asset | (803 | ) | - | |||||
| Foreign currency derivative | - | (231 | ) | |||||
| Total deferred tax liabilities | (917 | ) | (649 | ) | ||||
| Valuation allowance | (232,205 | ) | (208,460 | ) | ||||
| Net deferred tax assets | $ | - | $ | - | ||||
The majority of the Company’s deferred tax assets relate to net operating loss and research and development carryforwards that can only be realized if the Company is profitable in future periods. It is uncertain whether the Company will realize any tax benefit related to these carryforwards. Accordingly, the Company has provided a full valuation allowance against the net deferred tax assets due to uncertainties as to their ultimate realization. The valuation allowance will remain at the full amount of the deferred tax assets until it is more likely than not that the related tax benefits will be realized. The Company’s valuation allowance increased by $23,824 in 2019, increased by $26,040 in 2018, and decreased by $42,625 in 2017 primarily because of the remeasurement required by TCJA.
As of December 31, 2019, the Company had federal operating loss carryforwards of $653,524, state operating loss carryforwards of $509,639, and research and development and orphan drug credit carryforwards of $70,485, which will expire at various dates from 2020 through 2037. Federal losses, state losses, research and development credit carryforwards begin to expire in 2020.
79
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Tax years 2016-2019 remain open to examination by the major taxing jurisdictions to which the Company is subject. Additionally, years prior to 2016 are also open to examination to the extent of loss and credit carryforwards from those years. The Company recognizes interest and penalties accrued related to unrecognized tax benefits as components of its income tax provision. However, there were no provisions or accruals for interest and penalties in 2019, 2018 and 2017.
Note 9 — Employee 401(k) Plan
In January 1991, the Company adopted an employee retirement plan (“401(k) Plan”) under Section 401(k) of the Internal Revenue Code covering all employees. Employee contributions may be made to the 401(k) Plan up to limits established by the Internal Revenue Service. Company matching contributions may be made at the discretion of the Board of Directors. The Company made matching contributions of $926, $724 and $664, in 2019, 2018 and 2017, respectively.
Note 10 — Collaborative and Other Research and Development Contracts
National Institute of Allergy and Infectious Diseases (“NIAID/HHS”). In September 2013, NIAID/HHS contracted with the Company for the development of galidesivir as a treatment for Marburg virus disease. NIAID/HHS, part of the National Institutes of Health, made an initial award of $5,000 to the Company. The goals of this contract, including amendments, are to file IND applications for intravenous (“i.v.”) and intramuscular (“i.m.”) galidesivir for the treatment of Marburg virus disease and other hemorrhagic fever virus diseases, including Yellow Fever and Ebola virus disease, and to conduct an initial Phase 1 human clinical trial. As of December 31, 2019, the total NIAID/HHS contract amount to advance the program through the completion of the Phase I clinical program is $43,035. As of December 31, 2019, all options have been exercised under this contract.
U.S. Department of Health and Human Services (“BARDA/HHS”). On March 31, 2015, the Company announced that BARDA/HHS had awarded the Company a contract for the continued development of galidesivir as a potential treatment for diseases caused by RNA pathogens, including filoviruses. This BARDA/HHS contract includes a base contract of $16,265 to support galidesivir drug manufacturing, as well as $22,855 in additional development options that can be exercised by the government, bringing the potential value of the contract to $39,120. As of December 31, 2019, a total of $20,574 has been awarded under exercised options within this contract.
The contracts with NIAID/HHS and BARDA/HHS are cost-plus-fixed-fee contracts. That is, the Company is entitled to receive reimbursement for all costs incurred in accordance with the contract provisions that are related to the development of galidesivir plus a fixed fee, or profit. BARDA/HHS and NIAID/HHS will make periodic assessments of progress and the continuation of the contract is based on the Company’s performance, the timeliness and quality of deliverables, and other factors. The government has rights under certain contract clauses to terminate these contracts. These contracts are terminable by the government at any time for breach or without cause.
U.S. Department of Health and Human Services (“HHS”). On September 6, 2018, the Company announced that HHS had awarded the Company a $34,660 contract for the procurement of up to 50,000 doses of RAPIVAB (peramivir injection) over a five-year period. HHS’s purchase of RAPIVAB will supply the Strategic National Stockpile, the nation’s largest supply of potentially life-saving pharmaceuticals and medical supplies for use in a public health emergency. The Company delivered two shipments under this contract in 2019 for a total price of approximately $13,864, and we expect to deliver at least one shipment within the award in 2020, totaling approximately $6,932.
Torii Pharmaceutical Co., Ltd. (“Torii”). On November 5, 2019, the Company announced that it had entered into the Torii Agreement, granting Torii the exclusive right to commercialize berotralstat for the prevention of HAE attacks in Japan.
Under the Torii Agreement, the Company received an upfront, non-refundable payment of $22,000 and may be eligible to receive an additional milestone payment of either $20,000 if the PMDA grants regulatory approval on or before December 31, 2020, or $15,000 if regulatory approval is granted on or before December 31, 2021. In either case, the regulatory milestone payment is contingent upon receipt of a reimbursement price approval from Japan’s National Health Insurance system in excess of the threshold specified in the Torii Agreement.
In addition, the Company will be entitled under the Torii Agreement to receive tiered royalty payments based on the amount of annual net sales of berotralstat in Japan during each calendar year. If berotralstat maintains its Sakigake designation during the PMDA review, the tiered royalty rate will range from 20% to 40% of net sales, otherwise, the tiered royalty rate will range from 15% to 35% of net sales. Torii’s royalty payment obligations are subject to customary reductions in certain circumstances, but may not be reduced by more than 50% of the amount that otherwise would have been payable to the Company in the applicable calendar quarter. Torii’s royalty payment obligations commence upon the first commercial sale of berotralstat in Japan and expire upon the later of (i) the tenth anniversary of the date of first commercial sale of berotralstat in Japan, (ii) the expiration of our patents covering berotralstat, and (iii) the expiration of regulatory exclusivity for berotralstat in Japan. The Company will be responsible for supplying Torii with its required amounts of berotralstat. The activities of the parties pursuant to the Torii Agreement will be overseen by a Joint Steering Committee, to be composed of an equal number of representatives from each party to coordinate the development and commercialization of berotralstat in Japan.
80
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
Under the Torii Agreement, the Company has granted Torii a right of first negotiation (“ROFN”) to commercialize berotralstat in Japan for the acute treatment of HAE attacks if the Company develops berotralstat for such indication and to commercialize any additional kallikrein inhibitor that the Company may develop in the future for use in HAE in Japan. Under both ROFNs, if the parties do not agree to terms with respect to a definitive amendment to the Torii Agreement or new agreement, as applicable, the terms of the amendment or agreement would be set by a third party arbitrator.
The Company identified performance obligations related to (i) the license to develop and commercialize berotralstat, (ii) regulatory approval support and (iii) reimbursement pricing approval support. These were each determined to be distinct from the other performance obligations. The Company allocated the $22,000 upfront consideration to the identified performance obligations using estimation approaches to determine the standalone selling prices under ASC 606. Specifically, in determining the value related to the license, a valuation approach utilizing risk adjusted discounted cash flow projections was used and an expected cost plus margin approach was utilized for the other performance obligations. The Company recognized $20,101 in revenue for the twelve months ended December 31, 2019 including $19,344 associated with the license which was transferred to Torii at the execution of the Agreement and $757 related to the year to date services provided in the performance of the two approvals. As of December 31, 2019, $1,899 of the $22,000 upfront payment is expected to be recognized as revenue in 2020 as the services are delivered.
Seqirus UK Limited (“SUL”). On June 16, 2015, the Company and SUL, a limited company organized under the laws of the United Kingdom and a subsidiary of CSL Limited, a company organized under the laws of Australia, entered into a License Agreement (the “SUL Agreement”) granting SUL and its affiliates worldwide rights to develop, manufacture and commercialize RAPIVAB (peramivir injection) for the treatment of influenza except for the rights to conduct such activities in Israel, Japan, Korea and Taiwan (the permitted geographies together constituting the “Territory”). The Company retains all rights and associated economics to procure pandemic stockpiling orders for RAPIVAB from the U.S. Government, while SUL has the right to pursue government stockpiling outside the U.S.
Under the terms of the SUL Agreement, the Company is responsible for fulfilling all post-marketing approval commitments in connection with the FDA's approval of the NDA, and upon fulfillment will transfer ownership of and financial responsibility for the NDA to SUL. Pursuant to rights to sell ALPIVAB in the EU, the Company was also responsible for regulatory filings and interactions with the European Medicines Agency (“EMA”). In accordance with the SUL Agreement, the Company and SUL formed a joint steering committee, composed of an equal number of representatives from each party, to oversee, review and coordinate the conduct and progress of the commercialization of RAPIVAB in the Territory and any additional development. In October 2017, SUL transferred Canadian registration rights for RAPIVAB to the Company.
Under the terms of the SUL Agreement, the Company has received an upfront payment of $33,740 and has achieved all development milestones under the contract totaling $12,000. The Company is entitled under the SUL Agreement to receive tiered royalties at a percentage rate beginning in the mid-teens contingent upon meeting minimum thresholds of net sales, as well as a low-thirties percentage of the gross profit from government stockpiling purchases made outside the U.S. Specifically, the Company receives tiered royalties at a percentage rate in the mid-teens to low-forties on net sales in the U.S. during a Contract Year (defined as July 1 - June 30) and tiered royalties at a percentage rate in the mid-teens to mid-twenties on net sales in the Territory, other than in the U.S., during a Calendar Year, each subject to certain downward adjustments for circumstance or events impacting the overall market opportunity. SUL's royalty payment obligations commence on the date of the SUL Agreement and expire, on a country-by-country basis, upon the later of (i) the expiration of legal exclusivity in such country and (ii) ten years from the date of the SUL Agreement (the "Royalty Term"). The Company developed peramivir under a license from UAB and will owe sublicense payments to them on any future milestone payments and/or royalties received by the Company from SUL.
The Company and SUL entered arbitration proceedings that involved many items under the SUL Agreement including, but not limited to, the EMA approval milestone, which BioCryst maintains is due under the contract as well as appropriately commercializing peramivir in the Territory. On March 4, 2020, the International Court of Arbitration of the International Chamber of Commerce (“ICC Tribunal”) delivered a Partial Arbitration Award (the “Partial Arbitration Award”) in the arbitration matter between the Company and SUL with respect to the License Agreement dated June 16, 2015 between the Company and SUL (the “SUL Agreement”) relating to the commercialization of peramivir (RAPIVAB/ALPIVAB) worldwide (excluding Japan, Taiwan, Korea, and Israel, the “Territory”).
In the Partial Arbitration Award, the ICC Tribunal found that, during the term, SUL materially breached and abandoned its core duties to the Company under the Diligent Efforts (as defined in the SUL Agreement) requirements of the SUL Agreement as applicable in the U.S. The ICC Tribunal granted a declaratory judgment in favor of the Company terminating the SUL Agreement and restoring all rights to peramivir to the Company as of March 17, 2020 (or such other date as the parties agree). The ICC Tribunal also awarded the Company its attorneys’ fees and expenses incurred in securing the declaratory judgment as well as the costs incurred by the Company in the arbitration. Finally, the ICC Tribunal found that SUL breached the SUL Agreement by failing to pay the milestone payment due to the Company within 30 days of the approval of peramivir for adult use in the European Union and awarded the Company $5.0 million (plus interest) for this claim. The ICC Tribunal retained jurisdiction for further proceedings relating to the award of attorneys’ fees and for any dispute relating to the return to the Company of all rights to peramivir in the Territory.
Shionogi & Co., Ltd. (“Shionogi”). In February 2007, the Company entered into an exclusive license agreement with Shionogi to develop and commercialize peramivir in Japan for the treatment of seasonal and potentially life-threatening human influenza. Under the terms of the agreement, Shionogi obtained rights to injectable formulations of peramivir in Japan. The Company developed peramivir under a license from UAB and will owe sublicense payments to them on any future milestone payments and/or royalties received by the Company from Shionogi. In October 2008, the Company and Shionogi amended the license agreement to expand the territory covered by the agreement to include Taiwan. Shionogi has commercially launched peramivir under the commercial name RAPIACTA in Japan and Taiwan.
81
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
In December 2017, the Company, on behalf of Royalty Sub, instituted arbitration proceedings against Shionogi in order to resolve a dispute with Shionogi under the Shionogi Agreement regarding the achievement of sales milestones and escalating royalties. In the event that the Company prevails in the arbitration, any amounts realized in the arbitration or in respect of the milestone payments and escalating royalties that are the subject of the arbitration would be for the benefit of Royalty Sub and be used by Royalty Sub to service its obligations under the non-recourse PhaRMA Notes (except for any amounts realized by the Company in respect of royalties relating to sales to Japanese governmental entities, which amounts would be retained by the Company). The costs associated with the arbitration proceedings are expected to be paid out of the assets of Royalty Sub in accordance with the terms of the indenture and servicing agreement relating to the PhaRMA Notes, except to the extent such costs are recovered in connection with any arbitration award in favor of the Company and Royalty Sub if they prevail in the arbitration proceedings. Arbitration proceedings, like other legal proceedings, are inherently uncertain. The arbitration proceedings have concluded, with the decision that no sale milestones have been achieved and that the royalties will remain the same. The costs associated with the arbitration proceedings are recoverable from the assets of Royalty Sub in accordance with the terms of the indenture and servicing agreement relating to the PhaRMA.
Green Cross Corporation (“Green Cross”). In June 2006, the Company entered into an agreement with Green Cross to develop and commercialize peramivir in Korea. Under the terms of the agreement, Green Cross will be responsible for all development, regulatory, and commercialization costs in Korea. The Company received a one-time license fee of $250. The license also provides that the Company will share in profits resulting from the sale of peramivir in Korea, including the sale of peramivir to the Korean government for stockpiling purposes. Furthermore, Green Cross will pay the Company a premium over its cost to supply peramivir for development and any future marketing of peramivir products in Korea.
Mundipharma International Holdings Limited (“Mundipharma”). In February 2006, the Company entered into an exclusive, royalty bearing right and license agreement with Mundipharma for the development and commercialization of Mundesine, a Purine Nucleoside Phosphorylase (“PNP”) inhibitor, for use in oncology (the “Original Agreement”). Under the terms of the Original Agreement, Mundipharma obtained rights to Mundesine in markets across Europe, Asia, and Australasia in exchange for a $10,000 up-front payment.
On November 11, 2011, the Company entered into the Amended and Restated License and Development Agreement (the “Amended and Restated Agreement”) with Mundipharma, amending and restating the Original Agreement. Under the terms of the Amended and Restated Agreement, Mundipharma obtained worldwide rights to Mundesine. Commencing on November 11, 2011, Mundipharma controls the development and commercialization of Mundesine and assumes all future development and commercialization costs. The Amended and Restated Agreement provides for the possibility of future event payments totaling $15,000 for achieving specified regulatory events for certain indications and tiered royalties ranging from mid to high single-digit percentages of net product sales in each country where Mundesine is sold by Mundipharma. These royalties are subject to downward adjustments based on the then-existing patent coverage and/or the availability of generic compounds in each country.
Albert Einstein College of Medicine of Yeshiva University and Industrial Research, Ltd. (“AECOM” and “IRL” respectively). In June 2000, the Company licensed a series of potent inhibitors of PNP from AECOM and IRL, (collectively, the “Licensors”). The lead product candidates from this collaboration are forodesine and ulodesine. The Company has obtained worldwide exclusive rights to develop and ultimately distribute these, or any other, product candidates that might arise from research on these inhibitors. The Company has the option to expand the Agreement to include other inventions in the field made by the investigators or employees of the Licensors. The Company agreed to use commercially reasonable efforts to develop these drugs. In addition, the Company has agreed to pay certain milestone payments for each licensed product (which range in the aggregate from $1,400 to almost $4,000 per indication) for future development of these inhibitors, single digit royalties on net sales of any resulting product made by the Company, and to share approximately one quarter of future payments received from other third-party partners, if any. In addition, the Company has agreed to pay annual license fees, which can range from $150 to $500, that are creditable against actual royalties and other payments due to the Licensors. This agreement may be terminated by the Company at any time by giving 60 days advance notice or in the event of material uncured breach by the Licensors.
In May 2010, the Company amended the licensee agreement through which the Company obtained worldwide exclusive rights to develop and ultimately distribute any product candidates that might arise from research on a series of PNP inhibitors, including forodesine and ulodesine. Under the terms of the amendment, the Licensors agreed to accept a reduction of one-half in the percentage of future payments received from third-party sub licensees of the licensed PNP inhibitors that must be paid to the Licensors. This reduction does not apply to (i) any milestone payments the Company may receive in the future under its license agreement dated February 1, 2006 with Mundipharma and (ii) royalties received from its sub licensees in connection with the sale of licensed products, for which the original payment rate will remain in effect. The rate of royalty payments to the Licensors based on net sales of any resulting product made by the Company remains unchanged.
82
BIOCRYST PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(In thousands, except per share amounts)
On November 17, 2011, the Company further amended its agreements with the Licensors whereby the Licensors agreed to accept a reduction of one-half in the percentage of Net Proceeds (as defined) received by the Company under its Amended and Restated Agreement with Mundipharma that will be paid to AECOM/IRL.
On June 19, 2012, the Company further amended its agreements with AECOM/IRL whereby the parties clarified the definition of the field with respect to PNP inhibition and AECOM/IRL agreed to exclusive worldwide license of galidesivir to BioCryst for any antiviral use.
At its sole option and subject to certain agreed upon conditions, any future non-royalty payments due to be paid by the Company to AECOM/IRL under the license agreement may be made either in cash, in shares of the Company’s common stock, or in a combination of cash and shares.
On January 6, 2014, the Carbohydrate Chemistry Research Team from Callaghan Innovation Research Limited, formerly Industrial Research Limited, transferred to Victoria University of Wellington (“VUW”) to establish the Ferrier Research Institute. The intellectual property rights relating to this research team, and the contracts relating to that intellectual property were transferred to a wholly owned subsidiary of VUW, including the contracts to which BioCryst is a party. The parties executed novation agreements in order to effectuate the transfer. Except for a substitution of parties, the terms and conditions of the contracts are substantially the same
The University of Alabama at Birmingham (“UAB”). The Company currently has agreements with UAB for influenza neuraminidase and complement inhibitors. Under the terms of these agreements, UAB performed specific research for the Company in return for research payments and license fees. UAB has granted the Company certain rights to any discoveries in these areas resulting from research developed by UAB or jointly developed with the Company. The Company has agreed to pay single digit royalties on sales of any resulting product and to share in future payments received from other third-party partners. The Company has completed the research under the UAB agreements. These two agreements have initial 25-year terms, are automatically renewable for five-year terms throughout the life of the last patent and are terminable by the Company upon three months’ notice and by UAB under certain circumstances. Upon termination both parties shall cease using the other parties’ proprietary and confidential information and materials, the parties shall jointly own joint inventions and UAB shall resume full ownership of all UAB licensed products. There is currently no activity between the Company and UAB on these agreements, but when the Company licenses this technology, such as in the case of the Shionogi, Green Cross and SUL agreements, or commercializes products related to these programs, the Company will owe sublicense fees or royalties on amounts it receives.
Note 11 — Quarterly Financial Information (Unaudited)
| First | Second | Third | Fourth | |||||||||||||
| 2019 Quarters | ||||||||||||||||
| Revenues | $ | 5,887 | $ | 1,448 | $ | 1,775 | $ | 39,725 | ||||||||
| Net Loss | (31,054 | ) | (37,629 | ) | (37,592 | ) | (2,622 | ) | ||||||||
| Basic and diluted net loss per share | (0.28 | ) | (0.34 | ) | (0.34 | ) | (0.02 | ) | ||||||||
| 2018 Quarters | ||||||||||||||||
| Revenues | $ | 3,976 | $ | 12,494 | $ | 1,454 | $ | 2,729 | ||||||||
| Net Loss | (25,777 | ) | (18,446 | ) | (29,597 | ) | (27,432 | ) | ||||||||
| Basic and diluted net loss per share | (0.26 | ) | (0.19 | ) | (0.28 | ) | (0.25 | ) | ||||||||
83
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors of BioCryst Pharmaceuticals, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of BioCryst Pharmaceuticals, Inc. (the Company) as of December 31, 2019 and 2018, the related consolidated statements of comprehensive loss, stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2019, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2019 and 2018, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2019, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company’s internal control over financial reporting as of December 31, 2019, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated March 13, 2020 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 1993.
Raleigh, North Carolina
March 13, 2020
84
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors of BioCryst Pharmaceuticals, Inc.
Opinion on Internal Control Over Financial Reporting
We have audited BioCryst Pharmaceuticals Inc.’s internal control over financial reporting as of December 31, 2019, based on criteria established in Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, BioCryst Pharmaceuticals Inc. (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2019, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of BioCryst Pharmaceuticals, Inc. as of December 31, 2019 and 2018, the related consolidated statements of comprehensive loss, stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2019, and the related notes and our report dated March 13, 2020 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
Raleigh, North Carolina
March 13, 2020
85
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
| ITEM 9A. | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
We maintain a set of disclosure controls and procedures that are designed to ensure that information relating to BioCryst Pharmaceuticals, Inc. required to be disclosed in our periodic filings under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), is recorded, processed, summarized and reported in a timely manner under the Exchange Act of 1934. We carried out an evaluation as required by paragraph (b) of Rule 13a-15 or Rule 15d-15 under the Exchange Act, under the supervision and with the participation of management, including our Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rule 13a-15(e) or Rule 15d-15 under the Exchange Act). Based upon that evaluation, the Chief Executive Officer and Chief Financial Officer concluded that, as of December 31, 2019, our disclosure controls and procedures are effective. We believe that our disclosure controls and procedures will ensure that information required to be disclosed in the reports filed or submitted by us under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the Securities and Exchange Commission, and include controls and procedures designed to ensure that information required to be disclosed by us in such reports is accumulated and communicated to our management, including our Chairman and Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure.
Management’s Report on Internal Control Over Financial Reporting
Management of BioCryst Pharmaceuticals, Inc. is responsible for establishing and maintaining adequate internal control over financial reporting and for the assessment of the effectiveness of internal control over financial reporting. As defined by the Securities and Exchange Commission, internal control over financial reporting is a process designed by, or under the supervision of our principal executive and principal financial officers and effected by our Board of Directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of the financial statements in accordance with U.S. GAAP.
Our internal control over financial reporting is supported by written policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect our transactions and dispositions of our assets; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of the financial statements in accordance with U.S. GAAP, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In connection with the preparation of our annual financial statements, management has undertaken an assessment of the effectiveness of our internal control over financial reporting as of December 31, 2019, based on criteria established in Internal Control — Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 Framework) (the COSO Framework). Management’s assessment included an evaluation of the design of our internal control over financial reporting and testing of the operational effectiveness of those controls.
Based on this assessment, management has concluded that as of December 31, 2019, our internal control over financial reporting was effective. Management believes our internal control over financial reporting will provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with U.S. GAAP.
Ernst & Young LLP, the independent registered public accounting firm that audited our financial statements included in this report, has issued an attestation report on the Company’s internal control over financial reporting, a copy of which appears on page 82 of this annual report.
86
Changes in Internal Control over Financial Reporting
There have been no changes in our internal control over financial reporting that occurred during the quarter ended December 31, 2019 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
| ITEM 9B. | OTHER INFORMATION |
None.
87
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
The information required by this item is set forth under the captions “Items to be Voted upon — 1. Election of Directors,” “Executive Officers,” and “Corporate Governance” in our definitive Proxy Statement for the 2020 Annual Meeting of Stockholders and incorporated herein by reference.
| ITEM 11. | EXECUTIVE COMPENSATION |
The information required by this item is set forth under the captions “Compensation Discussion and Analysis,” “Summary Compensation Table,” “Grants of Plan-Based Awards in 2019,” “Outstanding Equity Awards at December 31, 2019,” “2019 Option Exercises and Stock Vested,” “Potential Payments Upon Termination or Change in Control,” “2019 Director Compensation,” “Compensation Committee Interlocks and Insider Participation” and “Compensation Committee Report” in our definitive Proxy Statement for the 2020 Annual Meeting of Stockholders and incorporated herein by reference.
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The information required by this item is set forth under the captions “Equity Compensation Plan Information” and “Security Ownership of Certain Beneficial Owners and Management” in our definitive Proxy Statement for the 2020 Annual Meeting of Stockholders and incorporated herein by reference.
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
The information required by this item is set forth under the captions “Certain Relationships and Related Transactions” and “Corporate Governance” in our definitive Proxy Statement for the 2020 Annual Meeting of Stockholders and incorporated herein by reference.
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information required by this item is set forth under the caption “Items to be Voted upon — 2. Ratification of Appointment of Independent Registered Public Accountants” in our definitive Proxy Statement for the 2020 Annual Meeting of Stockholders and incorporated herein by reference.
88
| ITEM 15. | EXHIBITS, FINANCIAL STATEMENT SCHEDULES |
(a) Financial Statements
The following financial statements appear in Item 8 of this Form 10-K:
No financial statement schedules are included because the information is either provided in the consolidated financial statements or is not required under the related instructions or is inapplicable and such schedules therefore have been omitted.
(b) Exhibits
89
90
91
92
93
94
95
| (31.1) | Certification of the Chief Executive Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| (32.1) | Certification pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | |
| (101) | Financial statements from the Annual Report on Form 10-K of BioCryst Pharmaceuticals, Inc. for the year ended December 31, 2019, formatted in XBRL: (i) Consolidated Balance Sheets, (ii) Consolidated Statements of Comprehensive Loss, (iii) Consolidated Statements of Cash Flows, (iv) Consolidated Statements of Stockholders’ Equity and (v) Notes to Consolidated Financial Statements. | |
| # | Confidential treatment granted. | |
| † | Portions of this exhibit have been omitted pursuant to Item 601(b)(10) of Regulation S-K. The Company agrees to furnish to the Securities and Exchange Commission a copy of any omitted portions of the exhibit upon request. | |
| * | Certain identified information has been excluded from this exhibit because it is both not material and would likely cause competitive harm to the Company if publicly disclosed. | |
| & | Management contracts. | |
| ( ) | Filed herewith. |
| ITEM 16. | FORM 10-K SUMMARY. |
None.
96
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized on March 13, 2020.
| BIOCRYST PHARMACEUTICALS, INC. | |||
| By: | /s/ Jon P. Stonehouse | ||
| Jon P. Stonehouse | |||
| Chief Executive Officer | |||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities indicated on March 13, 2020:
| Signature | Title(s) | |
|
/s/ Jon P. Stonehouse |
President, Chief Executive Officer and Director | |
| (Jon P. Stonehouse) | (Principal Executive Officer and Principal Financial Officer) | |
|
/s/ Michael L. Jones |
Executive Director, Finance & Principal Accounting Officer | |
| (Michael L. Jones) | (Principal Accounting Officer) | |
| /s/ George B. Abercrombie | Director | |
| (George B. Abercrombie) | ||
| /s/ Stephen Aselage | Director | |
| (Stephen Aselage) | ||
| /s/ Theresa Heggie | Director | |
| (Theresa Heggie) | ||
| /s/ Nancy Hutson | Director | |
| (Nancy Hutson, Ph.D.) | ||
| /s/ Robert A. Ingram | Director | |
| (Robert A. Ingram) | ||
| /s/ Kenneth B. Lee, Jr. | Director | |
| (Kenneth B. Lee, Jr.) | ||
| /s/ Alan G. Levin | Director | |
| (Alan G. Levin) | ||
| /s/ Helen Thackray, M.D. | Director | |
| (Helen Thackray, M.D.) | ||
97
