Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - Aquestive Therapeutics, Inc. | ex99_1.htm |
| 8-K - 8-K - Aquestive Therapeutics, Inc. | form8k.htm |
Exhibit 99.2

Corporate UpdateDecember 2018

2 Forward Looking Statements Certain statements in this presentation and associated oral statements
made by management may constitute "forward-looking statements.” Words such as “believes”, “expects”, “projects”, “future” and similar expressions often identify such forward-looking statements. These forward-looking statements are based on
our current expectations and beliefs and are subject to a number of risk factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Such risks and uncertainties
include, but are not limited to, the risk of the Company completing its development work, the risks of delays in FDA approval (or failure to approve) of our drug candidates as well as the risks inherent in commercializing a new product
(including technology risks, market risks, financial risks and implementation risks, and other risks and uncertainties affecting the Company including those described in our Form S-1 with the Securities and Exchange Commission (“SEC”). The
Company disclaims and is not under any obligation to revise any forward-looking statements, including, without limitation, financial estimates, whether as a result of new information, future events, or otherwise, except as required by
applicable law. This presentation also contains estimates and other statistical data made by independent parties relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations,
and you are cautioned not to give undue weight to such estimates. Neither we nor any other person makes any representation as to the accuracy or completeness of such data or undertakes any obligation to update such data after the date of this
presentation. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. By attending or
receiving this presentation you acknowledge that you will be solely responsible for your own assessment of the market and our market position and that you will conduct your own analysis and be solely responsible for forming your own view of
the potential future performance of our business.All third-party trademarks, including names, logos and brands, referenced by us in this presentation are property of their respective owners. All references to third-party trademarks are for
identification purposes only. Such use should not be construed as an endorsement of the products or services of us or this potential offering.* LibervantTM, the preliminary brand name for Diazepam Buccal Film, was conditionally accepted by
the U.S. Food and Drug Administration (FDA) and will be submitted for final FDA review and approval in the Diazepam Buccal Film New Drug Application (NDA).

Corporate Highlights 3 Broad, Late-Stage Product Pipeline Two late-stage CNS product candidates
aimed at improving treatment paradigmsComplex molecule product candidates in immunology and endocrinology targeting large market opportunities Commercial-Stage, Innovative Technology & Global IP Received FDA approval and launched
SYMAPAZAN™, adjunctive treatment of seizures associated with Lennox-Gastaut syndromeCommercial team with numerous product launches, including Diastat, Onfi and others in CNS spaceWorld-class manufacturing capabilitiesPharmFilm technology
allows unmet patient needs to be addressed in novel ways Extensive patents or patent applications provide protection to 2037 Revenue and Cash Flow from Successful Partnerships Multiple collaborations covering products from early-stage to
commercial~$50.6 million in revenue generated from licensed products and collaborations for first nine months 2018Cash flows re-invested in development of proprietary pipelineExperienced management team and board with track record of
developing and commercializing pharmaceutical products
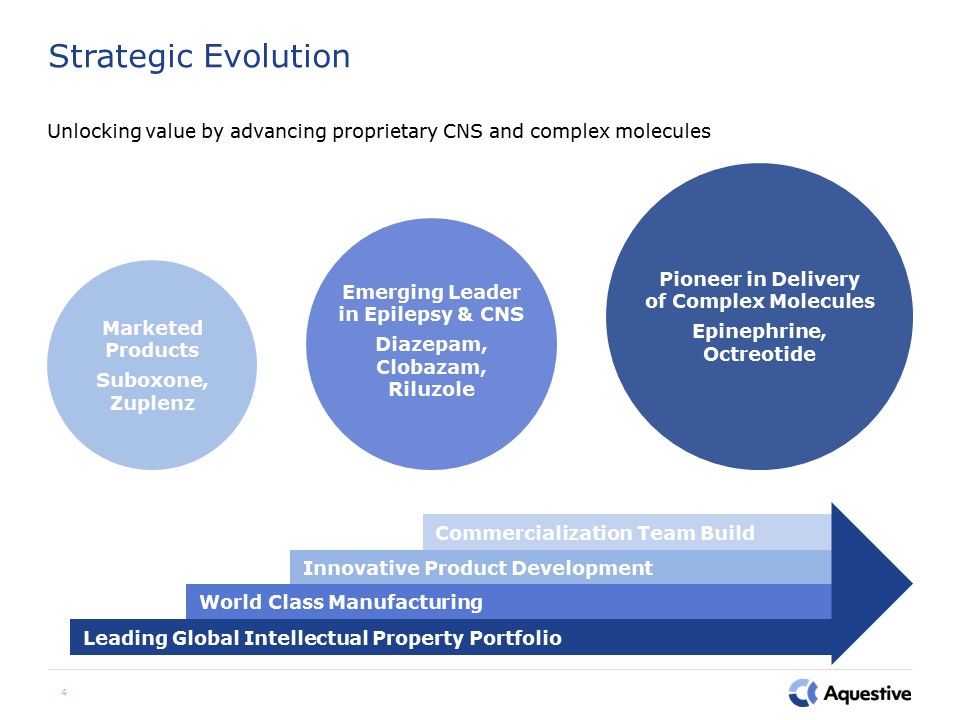
4 Strategic Evolution Unlocking value by advancing proprietary CNS and complex molecules Emerging
Leader in Epilepsy & CNSDiazepam,Clobazam, Riluzole Pioneer in Deliveryof Complex MoleculesEpinephrine, Octreotide Marketed ProductsSuboxone, Zuplenz Commercialization Team Build Innovative Product Development World Class
Manufacturing Leading Global Intellectual Property Portfolio
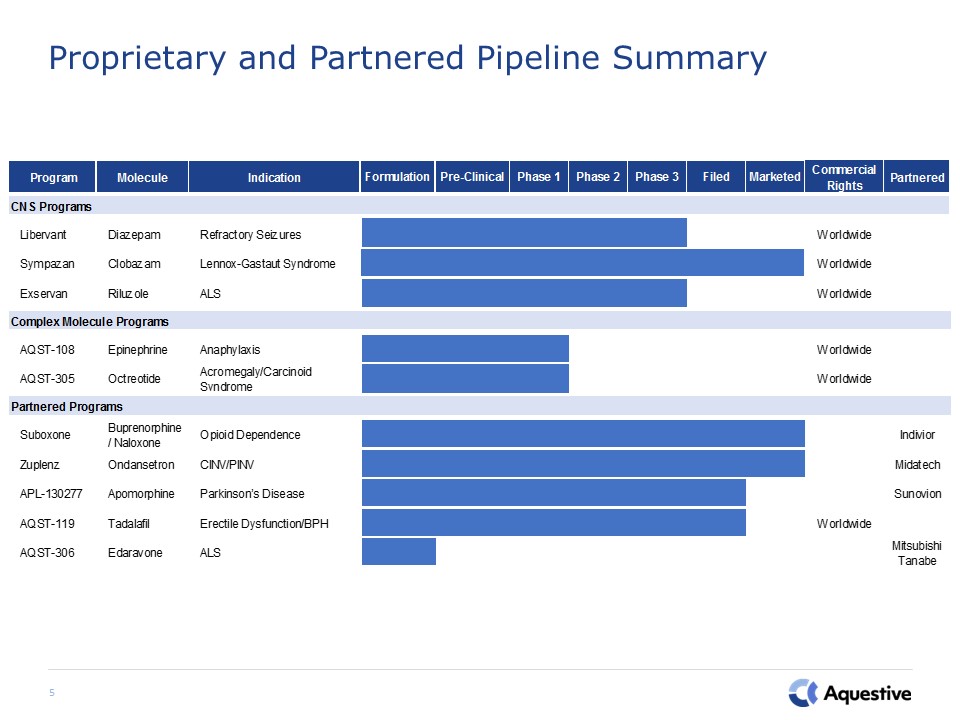
Proprietary and Partnered Pipeline Summary 5
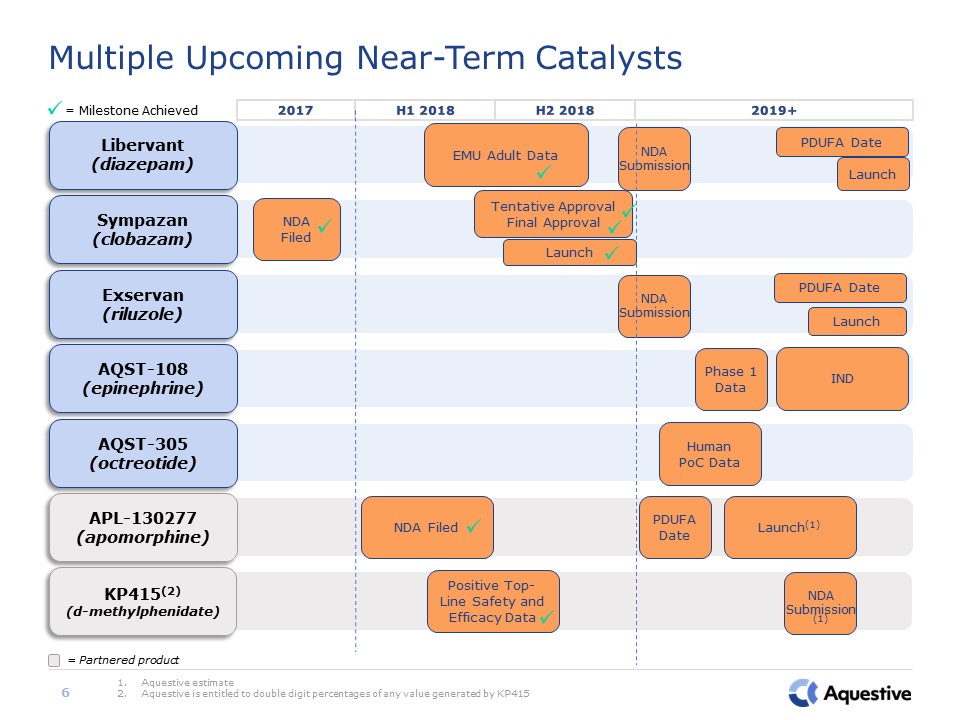
AQST-305(octreotide) Human PoC Data APL-130277(apomorphine) NDA Filed PDUFA
Date Launch(1) AQST-108(epinephrine) IND Phase 1 Data Exservan(riluzole) PDUFA Date Launch KP415(2)(d-methylphenidate) Positive Top-Line Safety and Efficacy Data Multiple Upcoming Near-Term Catalysts Aquestive
estimateAquestive is entitled to double digit percentages of any value generated by KP415 = Milestone Achieved 6 = Partnered product 2017 H1 2018 2019+ H2 2018 Sympazan(clobazam) NDA Filed Libervant(diazepam) EMU
Adult Data PDUFA Date Launch NDA Submission NDA Submission NDA Submission (1) Tentative ApprovalFinal Approval Launch

Experienced Management Team & Board of Directors 7 Keith Kendall President, Chief Executive
Officer & Director Mark SchobelChief Innovation & Technology Officer John MaxwellSVP, Chief Financial Officer Theresa WoodSVP, Human Resources and Organizational Development Peter BoydSVP, Operations and Value Delivery Dan
BarberSVP, Chief Strategy and Development Officer Ken MarshallCommercial Leader Doug BrattonFounding Partner and Chief Investment Officer, Crestline Investors / Ed Bass Group Greg Brown, M.D.Founding Managing Director, Healthcare Royalty
Partners Sandy CostaFormer President & COO, Quintiles James S. ScibettaCEO of Maverick Therapeutics, former President of Pacira Pharmaceuticals Board of Directors Leadership Team Nancy LurkerCEO of EyePoint Pharmaceuticals (NASDAQ:
EYPT), former CEO of PDI Corporation John CochranPartner and COO, Crestline Investors / Ed Bass Group Lori BraenderSVP, General Counsel

8 PharmFilm Technology First FDA approved oral films for lingual, sublingual and combination drug
deliveryMultiple advantages versus IV, tablet and liquid formulationsWorld leader in supplying oral films for prescription pharmaceutical products FDA- and DEA-inspected facilities with capacity sufficient for commercial quantities of
products and product candidates currently in developmentRobust intellectual property estate with at least 200 approved patents and more than 75 pending patents

CNS Product Portfolio & Strategy

Sympazan(clobazam oral film)Launch underway

11 Sympazan: Positioning in LGS Treatment Paradigm Patients with Epilepsy 3.4M Patients with
Refractory Seizures 1.2M LGS Patients (55K) Atypical LGS Crushed Tablet or Solution Administered via G-tube Solutions Tablets Crushed Tablet, Mixed with Food or Dissolved Ability to Swallow Compliance Low
Moderate High High Low Lennox-Gastaut Syndrome Market Lennox-Gastaut syndrome (LGS) is a rare, intractable form of epilepsy affecting ~55K patients in the US Medication administration perceived as significant unmet
needApproximately 30-40% of LGS patients experience dysphagia Caregivers struggle through decades of difficult treatment regimens, dosing alternatives to address this daily challengeONFIApproximately 500K prescriptions annually Available only
in tablet and suspension formulations LGS patients often cannot, or refuse to, swallowLeads to caregivers crushing tablets or using liquid formulationsCan result in patient receiving sub-optimal dose

Commercialization Strategy Commercial leadership with multiple product starts, including launches of
Diastat and ONFI30 person sales team, led by three seasoned Regional Directors, designed to support high prescribing HCPs in epilepsy space Completed home study and live training at National Sales Meeting alongside 30 cliniciansDeployed to
introduce company, educated value of PharmFilm, schedule appointments and peer programsEstimated >90% prescribing physician overlap between Libervant and Sympazan to enable dedicated sales team to cover most of the marketPlan to expand
ultimate sales team size of ~50 with 5 first line managers ahead of Libervant launchBuilt distribution network to ensure supply in pharmacyUS-focused commercialization strategy with option to partner ex-US 12

Libervant(diazepam buccal film)
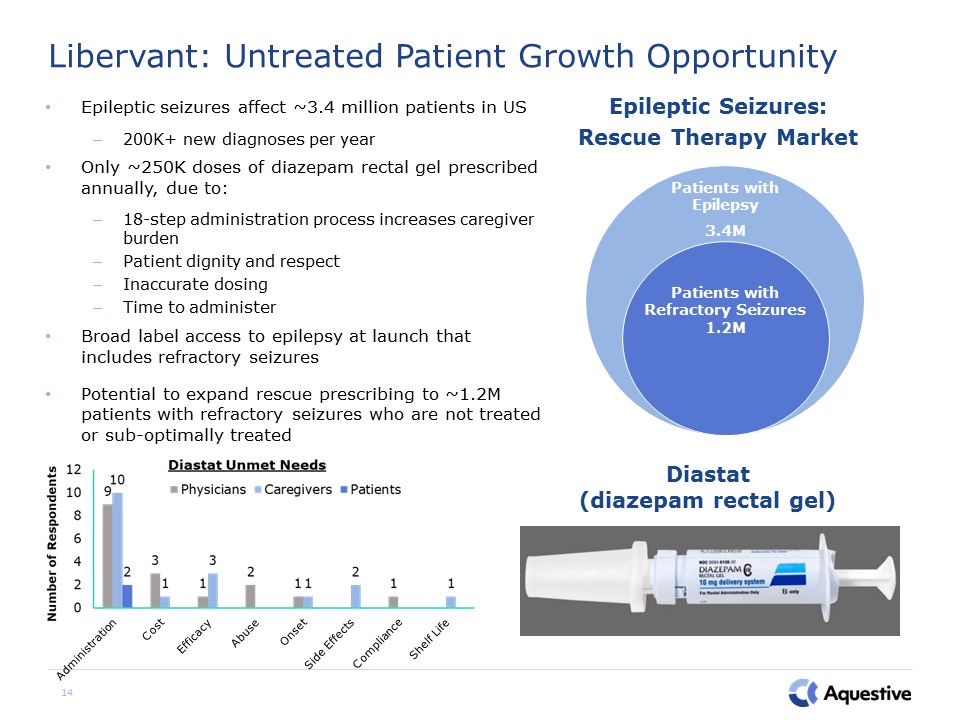
14 Libervant: Untreated Patient Growth Opportunity Epileptic seizures affect ~3.4 million patients
in US200K+ new diagnoses per yearOnly ~250K doses of diazepam rectal gel prescribed annually, due to:18-step administration process increases caregiver burdenPatient dignity and respectInaccurate dosingTime to administerBroad label access to
epilepsy at launch that includes refractory seizures Potential to expand rescue prescribing to ~1.2M patients with refractory seizures who are not treated or sub-optimally treated Patients with Epilepsy 3.4M Patients with Refractory
Seizures 1.2M Diastat (diazepam rectal gel) Epileptic Seizures: Rescue Therapy Market Administration Cost Efficacy Abuse Onset Side Effects Shelf Life Compliance

15 Libervant: Proprietary Formulation with Potential to Expand Patient Usage Buccal film
administration of diazepamDissolves quickly and demonstrates rapid achievement of therapeutic blood levelsIn development as rescue therapy for breakthrough epileptic seizuresAlternative to Diastat® (diazepam rectal gel)Adult EMU study topline
data to be presented as two late-breaking poster presentations at AES, December 2018Pre-NDA meeting scheduled for mid December 2018Prepared to commence rolling submission before year end assume positive meeting outcomeGranted orphan drug
designation and fast track designation
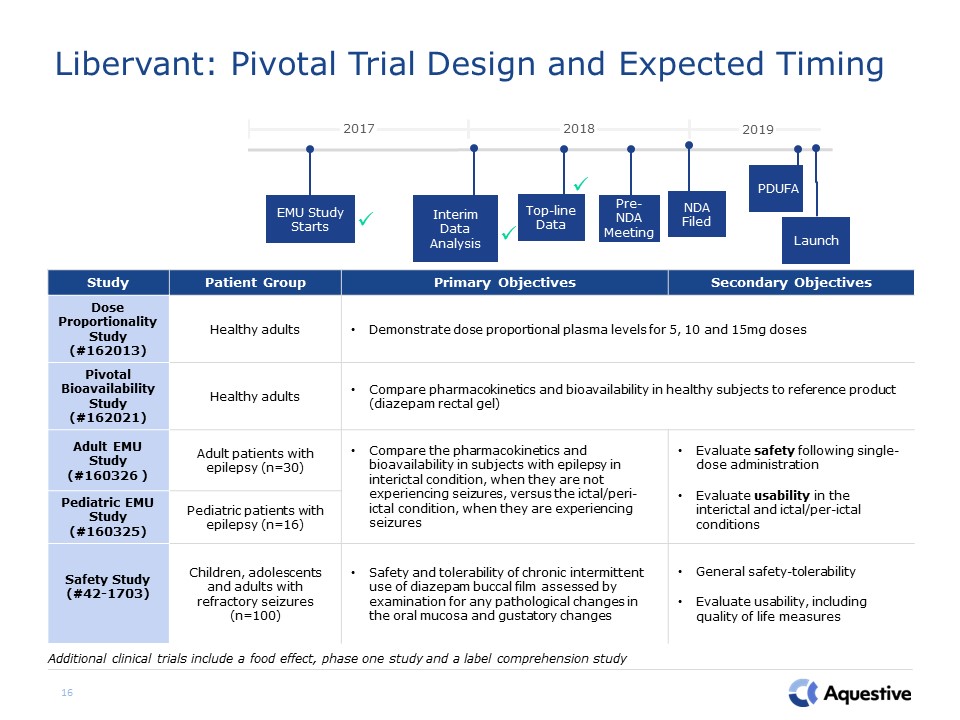
Libervant: Pivotal Trial Design and Expected Timing 16 Additional clinical trials include a food
effect, phase one study and a label comprehension study Study Patient Group Primary Objectives Secondary Objectives Dose Proportionality Study (#162013) Healthy adults Demonstrate dose proportional plasma levels for 5, 10 and 15mg
doses Pivotal Bioavailability Study (#162021) Healthy adults Compare pharmacokinetics and bioavailability in healthy subjects to reference product (diazepam rectal gel) Adult EMU Study(#160326 ) Adult patients with epilepsy
(n=30) Compare the pharmacokinetics and bioavailability in subjects with epilepsy in interictal condition, when they are not experiencing seizures, versus the ictal/peri-ictal condition, when they are experiencing seizures Evaluate safety
following single-dose administration Evaluate usability in the interictal and ictal/per-ictal conditions Pediatric EMU Study (#160325) Pediatric patients with epilepsy (n=16) Safety Study (#42-1703) Children, adolescents and adults
with refractory seizures (n=100) Safety and tolerability of chronic intermittent use of diazepam buccal film assessed by examination for any pathological changes in the oral mucosa and gustatory changes General safety-tolerabilityEvaluate
usability, including quality of life measures Launch EMU Study Starts 2018 2019 2017 PDUFA Pre-NDA Meeting Top-line Data Interim Data Analysis NDA Filed
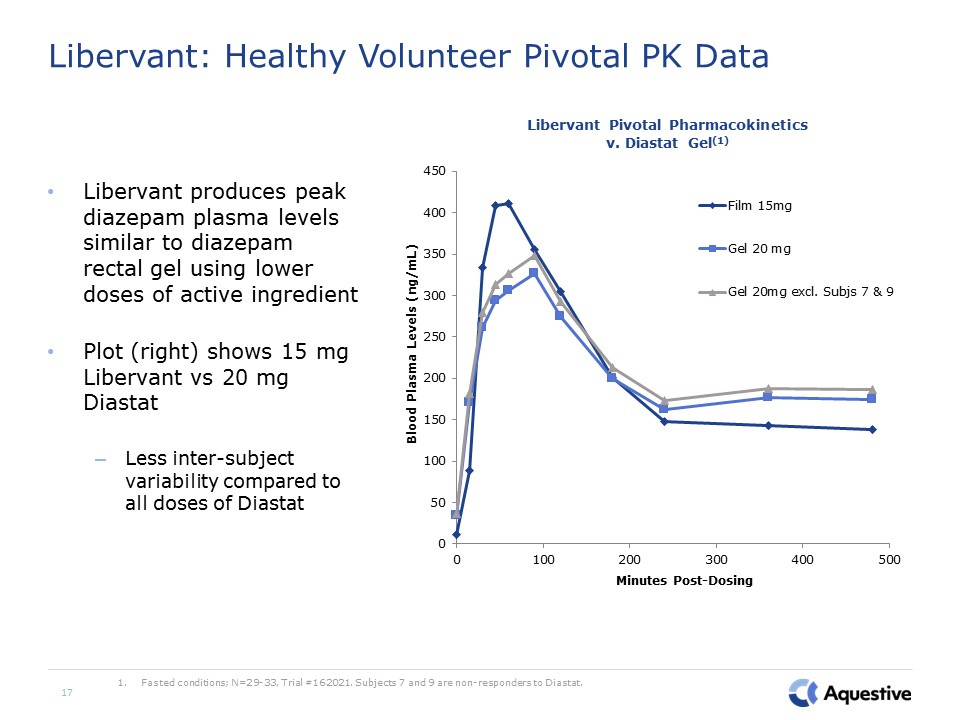
17 Libervant: Healthy Volunteer Pivotal PK Data Libervant produces peak diazepam plasma levels
similar to diazepam rectal gel using lower doses of active ingredientPlot (right) shows 15 mg Libervant vs 20 mg DiastatLess inter-subject variability compared to all doses of Diastat Fasted conditions; N=29-33, Trial #162021. Subjects 7 and
9 are non-responders to Diastat.

18 Libervant: No ‘Non-Responders’ Current standard of care (Diastat rectal gel):Exhibits a population
subset that does not obtain expected plasma concentrations of diazepamLibervant: Consistent plasma concentrations Even Diastat ‘non-responders’ received acceptable plasma concentrations with Libervant Performed under fasted conditions; Trial
#162021
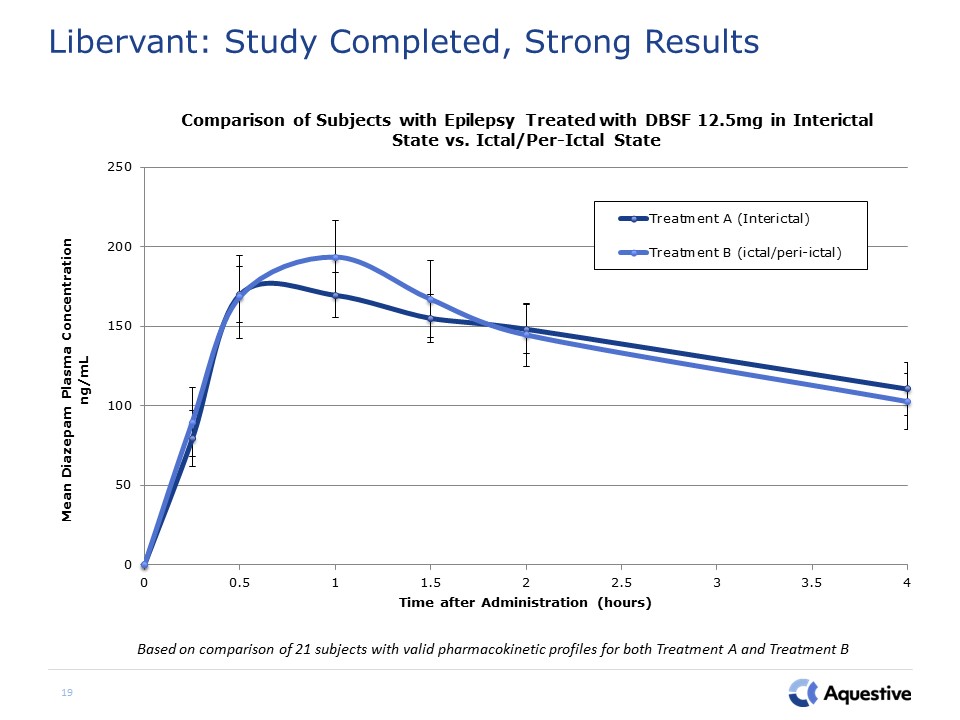
Libervant: Study Completed, Strong Results 19 Based on comparison of 21 subjects with valid
pharmacokinetic profiles for both Treatment A and Treatment B

Libervant: Adult EMU Safety & Tolerability 20 Majority of the treatment-emergent adverse events
(TEAE) in this study were classified as unrelated to study drugThe most common adverse events that were classified as probably related to study drug were somnolence and dizziness and were relatively infrequent (overall frequency of 5.7% of
subjects and 2.9% of subjects, respectively) FREQUENCY OF PATIENTS EXPERIENCING TREATMENT-EMERGENT ADVERSE EVENTS CONSIDERED POSSIBLY OR PROBABLY RELATED TO STUDY DRUG AND NUMBER OF EVENTS - SAFETY POPULATION Adverse Events n
(%) Period A (N=33) Period B (N=33) Overall (N=35) Somnolence 2 (6.1%) 0 2 (5.7%) Dizziness 1 (3.0%) 0 1 (2.9%) Hypoaesthesia 1 (3.0%) 0 1 (2.9%) Nausea 0 1 (3.0%) 1 (2.9%) Paraethesia oral 1 (3.0%) 1 (3.0%) 1 (2.9%)

Exservan(riluzole oral film)

22 Exservan (Riluzole) Oral soluble film formulation of riluzole for the treatment of Amyotrophic
Lateral Sclerosis (ALS)Potential to be the only formulation of riluzole that doesn’t require water or liquid for swallowingCompleted a pilot PK and pivotal PK study, as well as a food effect studyAll studies successfully shown bioequivalence
to the reference listed drug, RilutekRecently completed Swallowing Safety Study primary objective of the study was to evaluate the effect, if any, on swallowing safety in subjects with ALSstudy successfully met its primary objective and shows
that AQST-117 has no adverse effect on swallowing safety in subjects with ALS.Expect to submit an NDA by 1Q19

Complex Molecule Candidates & Partnered Products

24 AQST-108 (epinephrine): Injection to Oral 2017 injectable epinephrine market estimated to be
approximately $1.7B and over 3.5M total prescriptionsPotential alternative to EpiPen® intramuscular injection, the market leader in anaphylaxis treatmentAQST-108 has the potential to:Reduce treatment burden Lower costs to healthcare
systemAllow for more accurate dosing and patient compliance during times of life threatening anaphylaxis after exposure to allergensExpect to initiate next clinical trial in 1H 2019 Source: IQVIA, IQVIA Combined Audit, 03/2018 Auto-injector
Delivery Sublingual Film
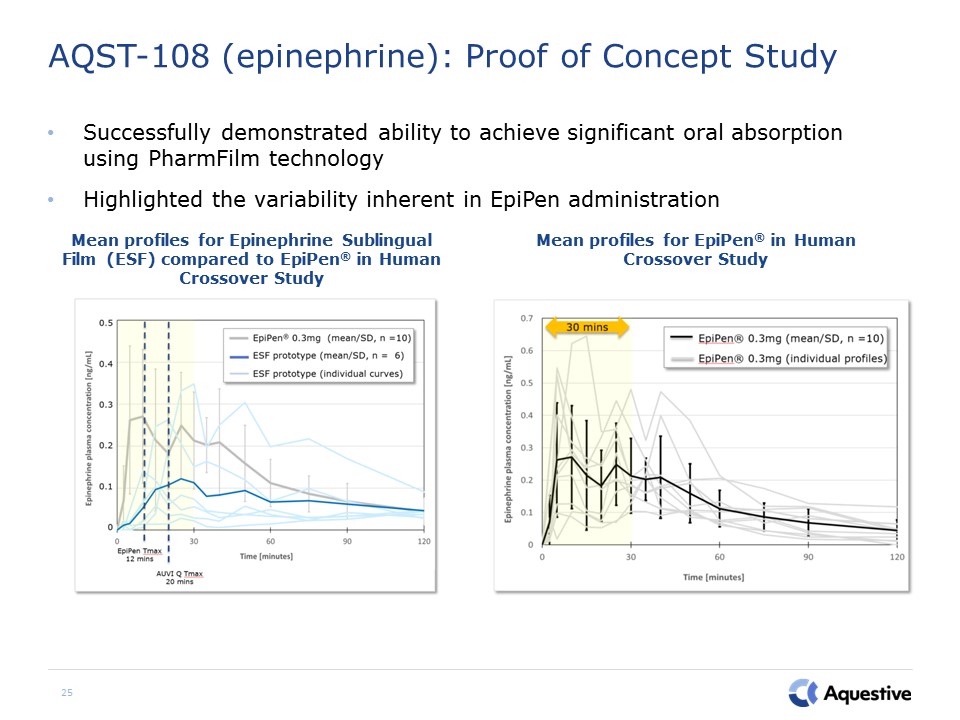
25 AQST-108 (epinephrine): Proof of Concept Study Successfully demonstrated ability to achieve
significant oral absorption using PharmFilm technologyHighlighted the variability inherent in EpiPen administration Mean profiles for Epinephrine Sublingual Film (ESF) compared to EpiPen® in Human Crossover Study Mean profiles for EpiPen®
in Human Crossover Study
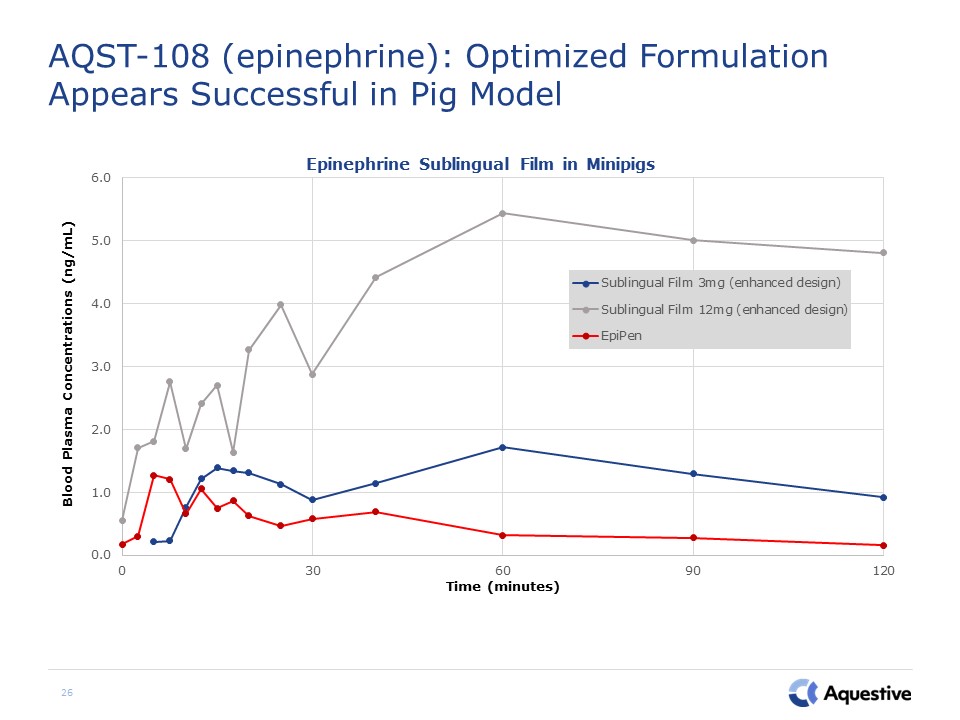
26 AQST-108 (epinephrine): Optimized Formulation Appears Successful in Pig Model

27 AQST-305 (octreotide): Gateway to Peptides and Biologics via Sublingual Film Alternative to
Sandostatin LAR®, a monthly depot injectionMarket leader in treatment of acromegaly and neuroendocrine tumors$843 million in sales in 2017(1)Twice daily administration of sublingual film eliminates need for monthly depot intramuscular
injectionSignificant patient burden associated with SandostatinPreclinical results demonstrated oral bioavailability levels of up to 10% vs. alternative technologies of <1%Dosed first patients in Q3 2018, with data by year-end 2018 IQVIA,
IQVIA Combined Audit, 03/2018Strasburgeret al, Patient reported outcomes of parenteral somatostatin analogue injections in 195 patients with acromegaly,Eur J Endocrinol, 12/18/15 EJE-15-1042 Pain Injection Site Reactions Sub-optimal
Symptom Control Emotional Impact Missed Work Days Issues Associated with Sandostatin(2):
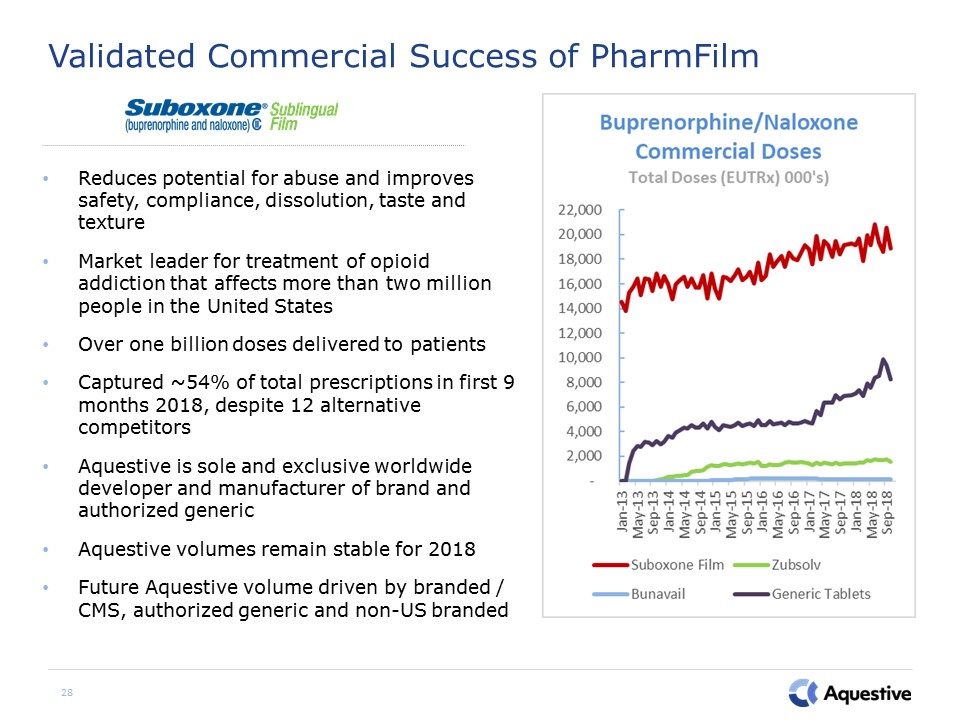
28 Validated Commercial Success of PharmFilm Reduces potential for abuse and improves safety,
compliance, dissolution, taste and textureMarket leader for treatment of opioid addiction that affects more than two million people in the United StatesOver one billion doses delivered to patientsCaptured ~54% of total prescriptions in first
9 months 2018, despite 12 alternative competitorsAquestive is sole and exclusive worldwide developer and manufacturer of brand and authorized genericAquestive volumes remain stable for 2018Future Aquestive volume driven by branded / CMS,
authorized generic and non-US branded
29 Apomorphine (APL-130277): Late-Stage Partnership with Sunovion Sunovion developed a sublingual
film formulation of apomorphine utilizing Aquestive’s PharmFilm technology Currently delivered via injection (Apokyn Pen)Aims to fulfill unmet need in patients with dysphagiaNDA under review for the treatment of patients with Parkinson’s
disease (PD) who experience motor fluctuations (OFF episodes) (affects ~500k U.S. patients)Reported positive top-line results from Phase 3 pivotal trial in January 2018PDUFA date in January 2019 Note: August 2016, Sunovion acquired Cynapsus,
whose single asset is based on Aquestive technology, for $624M.Source: US WorldMeds, LLC; 2014. Apokyn Pen: 7-Step Process(1)
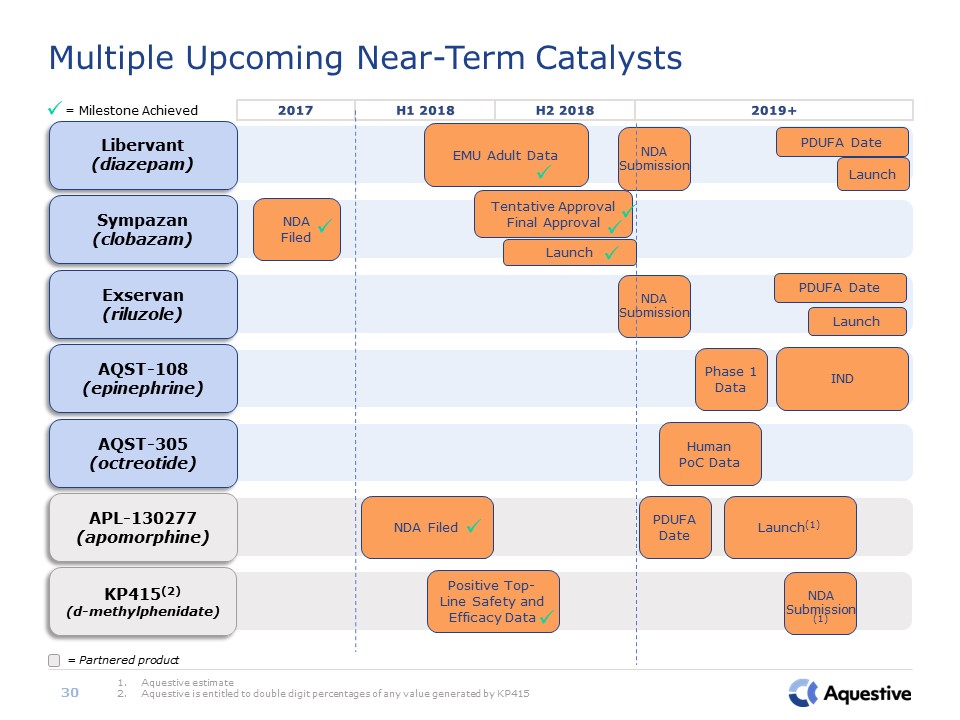
AQST-305(octreotide) Human PoC Data APL-130277(apomorphine) NDA Filed PDUFA
Date Launch(1) AQST-108(epinephrine) IND Phase 1 Data Exservan(riluzole) PDUFA Date Launch KP415(2)(d-methylphenidate) Positive Top-Line Safety and Efficacy Data Multiple Upcoming Near-Term Catalysts Aquestive
estimateAquestive is entitled to double digit percentages of any value generated by KP415 = Milestone Achieved 30 = Partnered product 2017 H1 2018 2019+ H2 2018 Sympazan(clobazam) NDA Filed Libervant(diazepam) EMU
Adult Data PDUFA Date Launch NDA Submission NDA Submission NDA Submission (1) Tentative ApprovalFinal Approval Launch

Thank you
