Attached files
| file | filename |
|---|---|
| EX-99.2 - EXHIBIT 99.2 - Aquestive Therapeutics, Inc. | ex99_2.htm |
| 8-K - 8-K - Aquestive Therapeutics, Inc. | form8k.htm |
Exhibit 99.1

Business UpdateDecember 2018

2 Forward Looking Statements Certain statements in this presentation and associated oral statements
made by management may constitute "forward-looking statements.” Words such as “believes”, “expects”, “projects”, “future” and similar expressions often identify such forward-looking statements. These forward-looking statements are based on our
current expectations and beliefs and are subject to a number of risk factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Such risks and uncertainties include,
but are not limited to, the risk of the Company completing its development work, the risks of delays in FDA approval (or failure to approve) of our drug candidates as well as the risks inherent in commercializing a new product (including
technology risks, market risks, financial risks and implementation risks, and other risks and uncertainties affecting the Company including those described in our Form S-1 with the Securities and Exchange Commission (“SEC”). The Company
disclaims and is not under any obligation to revise any forward-looking statements, including, without limitation, financial estimates, whether as a result of new information, future events, or otherwise, except as required by applicable law.
This presentation also contains estimates and other statistical data made by independent parties relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are
cautioned not to give undue weight to such estimates. Neither we nor any other person makes any representation as to the accuracy or completeness of such data or undertakes any obligation to update such data after the date of this presentation.
In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. By attending or receiving this
presentation you acknowledge that you will be solely responsible for your own assessment of the market and our market position and that you will conduct your own analysis and be solely responsible for forming your own view of the potential
future performance of our business.All third-party trademarks, including names, logos and brands, referenced by us in this presentation are property of their respective owners. All references to third-party trademarks are for identification
purposes only. Such use should not be construed as an endorsement of the products or services of us or this potential offering.

3 Business Update – December 2018 SympazanTM (clobazam) Oral Film launched in November; marketing and
sales efforts initiated nationwideLibervantTM (diazepam) Buccal Film* progressing toward NDA submission, pending outcome of pre-NDA meetingAquestive’s PharmFilm patent portfolio not impacted by lifting of DRL Preliminary Injunction; Suboxone®
Sublingual Film (CIII)** and Authorized Generic will continue to be a meaningful part of partnered revenue baseStrong cash position from IPO to support commercialization and development plans with several non-dilutive capital options available,
if needed *LibervantTM, the preliminary brand name for Diazepam Buccal Film, was conditionally accepted by the U.S. Food and Drug Administration (FDA) and will be submitted for final FDA review and approval in the Diazepam Buccal Film New
Drug Application (NDA)** SUBOXONE® and SUBUTEX® are registered trademarks of Indivior UK Limited.

4 SympazanTM (clobazam) Launch Received FDA approval as expected on November 1st for treatment of
seizures associated with Lennox-Gastaut syndrome (LGS) in patients 2 years of age or olderPharmacokinetic (PK) data presented at American Epilepsy Society 2018 Annual Meeting and published in Epilepsia1Commercial organization with 50+
experienced professionals, in markets nationwide actively meeting with prescribers and payers – first scripts written Engagements will help to educate key customers on value of PharmFilm technology, in advance of a potential 2019 Libervant
launch90% overlap in high volume prescriber base Key corporate milestone in Aquestive’s transformation from a CDMO to a specialty pharmaceutical company 1 “Pharmacokinetics of clobazam oral soluble film,”
https://onlinelibrary.wiley.com/doi/abs/10.1111/epi.14581

5 LibervantTM (diazepam) Buccal Film Completed six (6) studies to date, including pivotal Adult EMU
study, in line with FDA interactions to dateData presented at American Epilepsy Study (AES) meeting Long-term safety study ongoing and progressing wellContinue to enroll patients in Pediatric EMU and safety studiesUpcoming pre-NDA meeting with
FDA will inform submission timing and strategy; Seeking to gain the FDA’s alignment on following criteria:Appropriate characterization of pharmacokinetic data in healthy volunteers and patientsProposal for appropriate dosing regimen based on
studies and population PK modelProposal for providing safety data updates Progressing toward NDA submission, pending outcome of pre-NDA meeting in mid-December

6 Suboxone® Sublingual Film Appeals court lifted Preliminary Injunction, allowing DRL to enter market
“at risk”; IP litigation continuesNo impact to Aquestive’s patent portfolio, no validity or infringement decision renderedProprietary products have additional unique patents expected to extend protection into the late 2030sRevenues from
Suboxone are tied to manufacturing volume (not price), and derived from several current and potential sources where Aquestive is the sole and exclusive provider:US Branded orders from IndiviorAuthorized Generic orders from SandozAdditional
non-U.S. volume, which is growing as a percent of business Suboxone will remain a significant revenue contributor 4+ years

7 Aquestive Capital Resources Proceeds from July IPO positions company to commercially launch its
proprietary CNS productsImpact of Suboxone is manageable as variable cost structure will be adjusted to minimize any impact of lost revenuePotential monetizable royalty streams represent non-dilutive capital optionsAPL-130277 (Sunovion) - early
2019 PDUFAKP-415, -484, -879 (Kempharm) Solid cash and balance sheet available to fund current and future programsCompany strategy and plans were designed purposefully to shift from Suboxone/Indivior revenue base to proprietary
products Multi-dimensional business with the resources to fund growth prospects behind CNS franchise, early pipeline and diverse partner portfolio
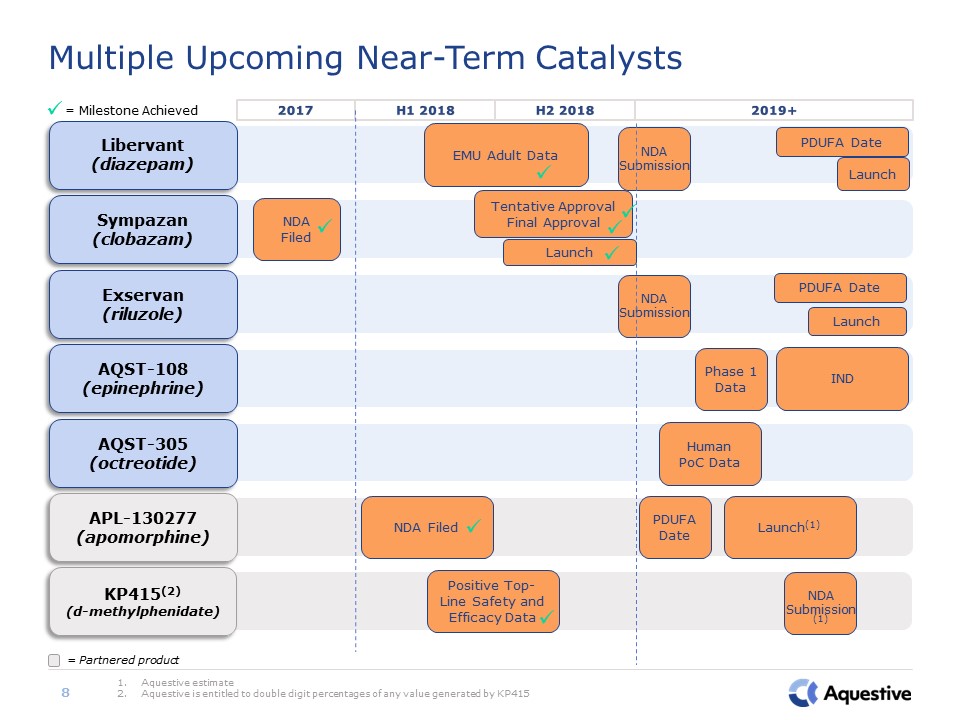
AQST-305(octreotide) Human PoC Data APL-130277(apomorphine) NDA Filed PDUFA
Date Launch(1) AQST-108(epinephrine) IND Phase 1 Data Exservan(riluzole) PDUFA Date Launch KP415(2)(d-methylphenidate) Positive Top-Line Safety and Efficacy Data Multiple Upcoming Near-Term Catalysts Aquestive estimateAquestive
is entitled to double digit percentages of any value generated by KP415 = Milestone Achieved 8 = Partnered product 2017 H1 2018 2019+ H2 2018 Sympazan(clobazam) NDA Filed Libervant(diazepam) EMU Adult Data PDUFA
Date Launch NDA Submission NDA Submission NDA Submission (1) Tentative ApprovalFinal Approval Launch
Learn more about Aquestive Therapeutics, Inc. 9 December 12, 201810:40 am2018 Prescriptions for
Success Healthcare ConferenceNew York, New York Online at:investors.aquestive.com

Thank you
