Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - Fibrocell Science, Inc. | a031618finalfcscfy2017ea.htm |
| 8-K - 8-K - Fibrocell Science, Inc. | fcsc31918form8-k.htm |

Corporate Presentation
March 19, 2018

Forward-Looking Statements
This presentation and our accompanying remarks contain “forward-looking statements” within the meaning of the U.S.
Private Securities Litigation Reform Act of 1995. All statements that are not historical facts are hereby identified as forward-
looking statements for this purpose and include, among others, statements relating to: the potential advantages of our
product candidates; the initiation, design and timing of pre-clinical studies and clinical trials and activities and the reporting
of the results thereof; the timing of regulatory submissions and actions; expected milestones; and all other statements
relating to our future operations, future financial performance, future financial condition, prospects or other future events.
Forward-looking statements are based upon our current expectations and assumptions and are subject to a number of
known and unknown risks, uncertainties and other factors that could cause actual results to differ materially and adversely
from those expressed or implied by such statements. Factors that could cause or contribute to such differences include,
among others: that interim clinical trial results are not necessarily indicative of final clinical results and final clinical trial
results may not be positive with regard to safety or efficacy of FCX-007 or FCX-013; uncertainties and delays relating to the
initiation, enrollment and completion of pre-clinical studies and clinical trials, including with respect to FCX-007, FCX-013 and
our other product candidates; whether pre-clinical study and clinical trial results will validate and support the safety and
efficacy of our product candidates; the risk that results seen in pre-clinical studies may not be replicated in humans; varying
interpretation of pre-clinical and clinical data; unanticipated or excess costs relating to the development of our gene therapy
product candidates; our ability to obtain additional capital to continue to fund operations; our ability to maintain our
collaboration with Intrexon Corporation; and the other factors discussed under the caption “Item 1A. Risk Factors” in our
most recent Form 10-K and Form 10-Qs which are available through the “Investors—SEC Filings” page of our website at
www.fibrocell.com. As a result, you should not place undue reliance on forward-looking statements.
The forward-looking statements made in connection with this presentation represent our views only as of the date of this
presentation (or any earlier date indicated in such statement). While we may update certain forward-looking statements
from time to time, we specifically disclaim any obligation to do so, even if new information becomes available in the future.
2

Investment Highlights
3
•Medical breakthroughs for rare diseases of the skin and connective tissue
• Unmet needs with no approved therapies
• Significant mortality and morbidity impact on pediatric populations
Focus on Rare Skin
Diseases
• Autologous fibroblasts derived from skin are the vehicle to deliver target proteins
locally to the site of disease
• Extensive experience culturing dermal fibroblasts, including commercial scale
Proprietary Ex-Vivo Gene
Modified Fibroblast
Platform
• FCX-007 for the treatment of Recessive Dystrophic Epidermolysis Bullosa (RDEB)
Obtained FDA allowance to initiate enrollment of pediatric patients in Phase 2
Well tolerated safety and positive early trends noted in pharmacology and wound healing
• FCX-013 for the treatment of moderate to severe Localized Scleroderma
IND allowed by FDA, expect enrollment to begin 3Q2018
Two Clinical Trial Programs
• Regulatory advantages including more frequent communications with FDA, eligibility
for Accelerated Approval and Priority Review, and Rolling Review
• Two Rare Pediatric Disease Designations to potentially receive Priority Review Vouchers
(PRVs) upon market authorization
Multiple FDA Designations
• In-house 13,000 square foot cGMP cell therapy manufacturing facility supporting both
FCX-007 and FCX-013
• Existing capacity to serve the U.S. RDEB market
Internal Manufacturing
Infrastructure

Development Pipeline
4
Program Condition Target Research Pre-Clinical
Phase 1/2
Clinical Trials FDA Designations
FCX-007 Recessive Dystrophic
Epidermolysis Bullosa
(RDEB)
Type VII
Collagen
FCX-013 Moderate to Severe
Localized Scleroderma
MMP-1
Research Arthritis and Related
Conditions
TBD
Orphan Drug
Rare Pediatric Disease
Fast Track
Orphan Drug
Rare Pediatric Disease
Portfolio being developed in collaboration with

5
Our Proprietary Fibroblast Platform
Vector
FCX-007 autologous dermal fibroblasts in culture
Lentiviral vector gene transfer
• 3rd generation and self-
inactivating
• Accommodates large gene
constructs
• Transduces both dividing and non-
dividing cells
• Target gene integration for long-
term expression of the protein
Autologous fibroblasts
• Readily sourced, as fibroblasts
are the most common cell type
in skin and connective tissue
• Reduced rejection and
immunogenicity concerns
• Established manufacturing
process and regulatory
pathway Ex-vivo Gene
Modified Cells
Delivery Vehicle Viral Vector
Ex-Vivo Gene Modified Cells
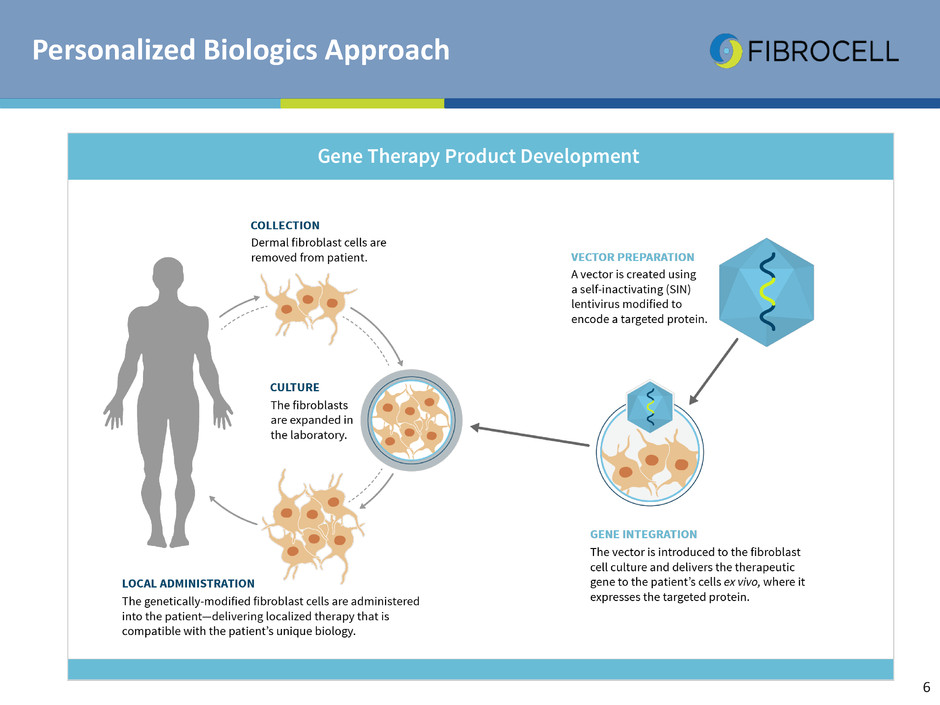
Personalized Biologics Approach
6

FCX-007 – Recessive Dystrophic Epidermolysis Bullosa
FCX-007 is a dermal fibroblast transduced with a lentiviral vector encoded with the gene for
type VII collagen
Orphan Drug Designation Rare Pediatric Disease Designation Fast Track Designation
7
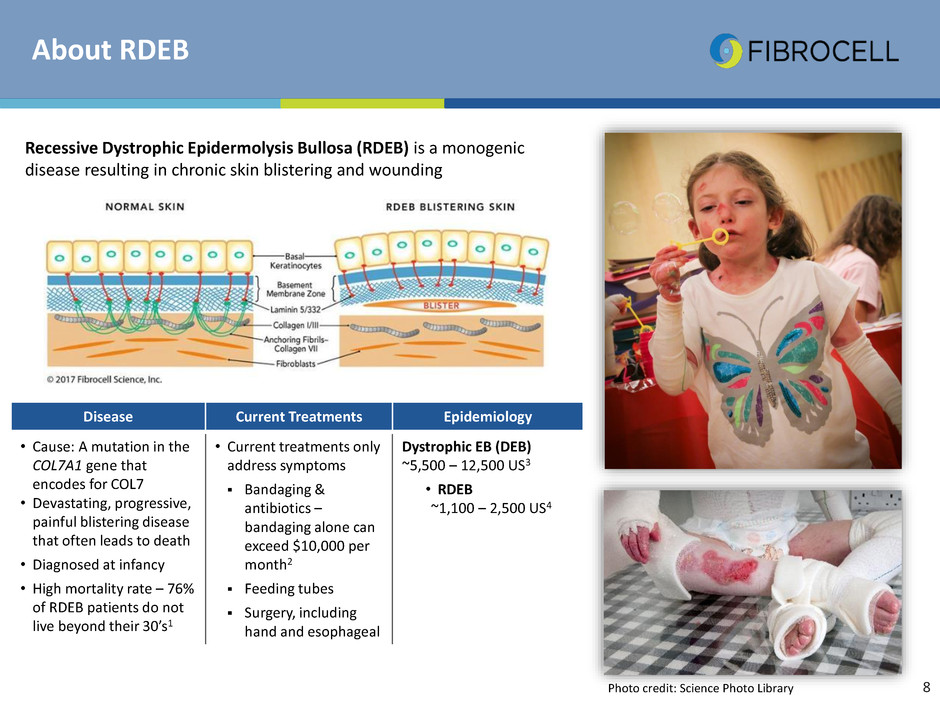
About RDEB
8
Recessive Dystrophic Epidermolysis Bullosa (RDEB) is a monogenic
disease resulting in chronic skin blistering and wounding
Photo credit: Science Photo Library
Disease Current Treatments Epidemiology
• Cause: A mutation in the
COL7A1 gene that
encodes for COL7
• Devastating, progressive,
painful blistering disease
that often leads to death
• Diagnosed at infancy
• High mortality rate – 76%
of RDEB patients do not
live beyond their 30’s1
• Current treatments only
address symptoms
Bandaging &
antibiotics –
bandaging alone can
exceed $10,000 per
month2
Feeding tubes
Surgery, including
hand and esophageal
Dystrophic EB (DEB)
~5,500 – 12,500 US3
• RDEB
~1,100 – 2,500 US4

FCX-007 Phase 1/2 Clinical Trial Design
9
A Phase 1/2 Trial of FCX-007 (Genetically-Modified Autologous Human Dermal
Fibroblasts) for Recessive Dystrophic Epidermolysis Bullosa (RDEB)
NCT02810951
Title
To evaluate the safety of a single intradermal injection session of FCX-007Primary Objective
To evaluate wound healing and pharmacology of FCX-007Secondary Objective
Stanford University School of Medicine and Children’s Hospital ColoradoInvestigator Sites
• Four patients enrolled and dosed, continuing follow-up visits through 25 weeks
• Next interim data readout expected in 2Q2018
Phase 1 Status
• Obtained FDA allowance to initiate enrollment of pediatric patients
• Six patients ages seven and older targeted for enrollment
• One adult patient enrolled; expect completion of enrollment 3Q2018
Phase 2 Status

FCX-007 Interim Data Readout
• Interim readout of data for three NC1+ adult Phase 1 patients, 12 weeks post-
administration
• Single injection session of FCX-007 to target wounds or preliminary safety assessment
• Five wounds treated over three patients ranging from 4.4cm2 to 13.1cm2 in size
• FCX-007 was well tolerated through 12 weeks post-administration
No serious adverse events or product related adverse events
No replication competent lentivirus (RCL) detected
Low to no autoantibody response detected
• Positive early trends noted in pharmacology signals and wound healing
• The Data Safety Monitoring Board (DSMB) concluded that safety and potential benefit
were established, and allowed continuation of enrollment and dosing
10

FCX-007 Interim Readout: Wound Healing
• Digital images captured and tracings performed
Skin tattoos and transparent overlays used as landmarks
• Wounds monitored in the study prior to dosing were open
at each monitoring visit up to 8 months
• A matched size untreated wound for each treated wound
on the same patient was also monitored and evaluated
• Compared to the baseline measurement collected at Day 0
pre-administration, wounds healing after a single injection
session of FCX-007 resulted as follows:
11
* Increase in size for one wound from the data set may have potentially
destabilized due to biopsy samples collected in the center of the wound bed
Day 0 Pre-Administration 4 Weeks 12 Weeks
75% healed 100% healed
Treated area: 10.3cm2
Right posterior leg
• White arrows denote tracking tattoo
• Dash lines represent target treatment area
• Day 0 pen markings denote injection locations
• 4 week solid lines denote remaining wounded areas
Post-
Baseline
Visit
Treated or
Untreated
Wounds
Percent Healing Number of
Wounds Meeting
Criteria
4 weeks Treated > 75% healed 100% (5/5)
Untreated < 25% healed 80% (4/5)
12 weeks Treated > 70% healed 80% (4/5)*
Untreated < 25% healed 80% (4/5)

FCX-007 Interim Readout: Pharmacology
12Texas Red – C7/fNC1 + DAPI + FITC – laminin 332Texas Red – C7/fNC1
• Variable pharmacology signals detected throughout the data set in each patient for one or more assays
through 12 weeks post-administration (qPCR, electron microscopy, immunofluorescence)
Anchoring fibrils have not been detected to date, whereas COL7 mRNA and COL7 protein have been confirmed in multiple
patient samples, including one that detected linear expression of COL7
• Linear expression of COL7 in the basement membrane zone (BMZ) and expression in the dermis detected
through 12 weeks post-administration

FCX-013 – Moderate to Severe Localized Scleroderma
FCX-007 is a dermal fibroblast transduced with a lentiviral vector encoded with the gene for
MMP-1, under control of the RheoSwitch Therapeutic System® (RTS®)
Orphan Drug Designation Rare Pediatric Disease Designation
13

About Localized Scleroderma
14
Disease Epidemiology
• Excess production of collagen
characterized by skin fibrosis
• Focus on moderate to severe
subtypes
• Thickening may extend to
underlying tissue and muscle
in children which may impair
growth in affected limbs or
forehead
• Lesions appearing across
joints impair motion and may
be permanent
• Localized Scleroderma
~160,000 sufferers US5
comprised of different sub-
types
~90,000 patients are
considered moderate to
severe6
Current Treatments
Current treatments:
• Systemic or topical
corticosteroids target
inflammation
• UVA light therapy
• Physical therapy
Photo: Reprinted from the Journal of the American Academy of Dermatology, Volume 59, Issue 3, Stéphanie Christen-Zaech, Miriam D. Hakim, F. Sule Afsar, Amy S. Paller.
Pediatric morphea (localized scleroderma): Review of 136 patients, Figure 1, pp. 385-396. Copyright Sept 2008. Used with permission from Elsevier Ltd.

FCX-013 Moving into Clinical Trials
15
• Product profile
Dermal fibroblasts genetically modified using lentivirus and encoded for matrix
metalloproteinase 1 (MMP-1), a protein responsible for breaking down collagen
Incorporates Intrexon’s RheoSwitch Therapeutic System® (RTS®) to control protein expression
• Activated by an oral ligand (veledimex)
• FDA allowed IND
• Completed GLP toxicology/biodistribution study
Bleomycin fibrosis model using immunocompromised mice (NOD/SCID)
No test article-related clinical observations, body weight changes, changes in clinical pathology
parameters, gross observations or organ weight change
No significant vector biodistribution to target organs
• Expect to initiate enrollment for a Phase 1/2 clinical study in 3Q2018

FCX-013 Phase 1/2 Clinical Trial Design
16
A Phase 1/2 Study of a Combination of FCX-013 (Genetically-Modified Human
Dermal Fibroblasts) plus Veledimex for the Treatment of Moderate to Severe
Scleroderma (Morphea)
Title
To evaluate the safety of a single intradermal injection session of FCX-013Primary Objective
To evaluate fibrosis resolution through the following measurements:
• Histology
• MRI, ultrasound and durometry
• Various scale assessments
Secondary Objectives
• 10 (approximately 5 per Phase)
• Staggered enrollment for first 3 adults
• Starting with adult patients; addition of pediatric patients after submission and
approval of safety and activity data from adult patients to FDA and the DSMB
Number of Patients
Initiate enrollment of Phase 1 adult patients in 3Q2018Status

FCX-013 Proof-of-Concept Study
17
• Study Design
Bleomycin treated SCID mouse model
N=30 mice over test and control groups
Assessed histologically for reduction of dermal
thickness and sub-dermal muscle in the presence
of FCX-013 and oral ligand
• Result
Bleomycin treatment resulted in skin fibrosis, measured by a significant increase in dermal thickness
Demonstrated that FCX-013 with ligand reduced the dermal thickness of fibrotic tissue to levels
similar to non-bleomycin (saline) with ligand treated skin
Further reduced the thickness of the sub-dermal muscle layer
Blecomycin treatments Ligand Treatment
D0 D28 D29 D39
Cell
injection
Harvest skin
samples
CONTROL:
Saline (no Bleo)
No Cells
TEST:
Bleomycin
FCX-013
CONTROL:
Bleomycin
Non-Modified Cells

Fibrocell’s existing cGMP cell therapy manufacturing facility in
Exton, PA has been designated as the production site for FCX-007
and FCX-013
• ~13,000 square foot facility includes:
Cleanroom for cell culture manufacturing
On-site quality control testing laboratories
Cryogenic storage
Warehouse and distribution areas
Centrally monitored
• Previous experience with commercial cell therapy manufacturing
• Site for remaining clinical and future commercial manufacture of
FCX-007 and FCX-013
• Existing capacity to serve the U.S. RDEB market commercially
• In-house manufacturing expertise for fibroblast-based autologous
cell and gene therapy products
• Multiple site inspections by FDA
Manufacturing Experience and Solutions
18

2Q2018
Interim Phase 1 data
readout and trial
update
Dose first Phase 2
patient
FCX-007 Anticipated Milestones 2018-2019
January 2018
FDA allowance of
pediatric patient
enrollment in Phase 2
March 2018
FDA allowance of IND
3Q2018
Complete enrollment
of Phase 2 patients
3Q2018
Initiate enrollment
of Phase 1 patients
1Q2019
Interim data readout
and trial update
FCX-007
FCX-013
Category Amount
Cash at December 31, 2017 $17.4M
Share Count (Outstanding/Fully Diluted) 28.3M/63.6M as of February 28, 2018
Cash Runway Cash into 1Q2019
19

Investment Highlights
20
•Medical breakthroughs for rare diseases of the skin and connective tissue
• Unmet needs with no approved therapies
• Significant mortality and morbidity impact on pediatric populations
Focus on Rare Skin
Diseases
• Autologous fibroblasts derived from skin are the vehicle to deliver target proteins
locally to the site of disease
• Extensive experience culturing dermal fibroblasts, including commercial scale
Proprietary Ex-Vivo Gene
Modified Fibroblast
Platform
• FCX-007 for the treatment of Recessive Dystrophic Epidermolysis Bullosa (RDEB)
Obtained FDA allowance to initiate enrollment of pediatric patients in Phase 2
Well tolerated safety and positive early trends noted in pharmacology and wound healing
• FCX-013 for the treatment of moderate to severe Localized Scleroderma
IND allowed by FDA, expect enrollment to begin 3Q2018
Two Clinical Trial Programs
• Regulatory advantages including more frequent communications with FDA, eligibility
for Accelerated Approval and Priority Review, and Rolling Review
• Two Rare Pediatric Disease Designations to potentially receive Priority Review Vouchers
(PRVs) upon market authorization
Multiple FDA Designations
• In-house 13,000 square foot cGMP cell therapy manufacturing facility supporting both
FCX-007 and FCX-013
• Existing capacity to serve the U.S. RDEB market
Internal Manufacturing
Infrastructure

References
1 Fine, J. et. al. (ed.). Epidermolysis Bullosa: Clinical, Epidemiologic, and Laboratory Advances and the Findings of the
National Epidermolysis Bullosa Registry. The John Hopkins University Press, Baltimore, MD, 1999.
2 The Dystrophic Epidermolysis Research Association of America (DebRA). EB brochure, page 6:
http://www.debra.org/DebraBrochure; accessed 11/10/17.
3 DEBRA International. What is EB Infographic: http://www.debra-international.org/what-is-eb.html; accessed
11/10/17.
4 Murauer, E, Koller, U, Pellegrini, G, De Luca, M, Bauer, J. Advances in Gene/Cell Therapy in Epidermolysis Bullosa. The
Keio Journal of Medicine. 2015; 64.
5 Peterson LS et al. The epidemiology of morphea (localized scleroderma) in Olmsted County 1960-1993. J Rheumatol.
1997; 24:73-80.
6Leitenberger, et. al. Distinct autoimmune syndromes in morphea: a case study of 245 adult and pediatric cases. Arch
Dermatol. 2009 May; 145(5):545-550.
21
