Attached files
| file | filename |
|---|---|
| EX-23.1 - EXHIBIT 23.1 - Fibrocell Science, Inc. | fcsc-20151231ex231.htm |
| EX-21 - EXHIBIT 21 - Fibrocell Science, Inc. | fcsc-20151231ex21.htm |
| EX-32.1 - EXHIBIT 32.1 - Fibrocell Science, Inc. | fcsc-20151231ex321.htm |
| EX-23.2 - EXHIBIT 23.2 - Fibrocell Science, Inc. | fcsc-20151231ex232.htm |
| EX-32.2 - EXHIBIT 32.2 - Fibrocell Science, Inc. | fcsc-20151231ex322.htm |
| EX-31.2 - EXHIBIT 31.2 - Fibrocell Science, Inc. | fcsc-20151231ex312.htm |
| EX-31.1 - EXHIBIT 31.1 - Fibrocell Science, Inc. | fcsc-20151231ex311.htm |
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
________________________________________________________________
FORM 10-K
ý Annual Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
For the fiscal year ended December 31, 2015
OR
¨ Transition Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
________________________________________________________________

Fibrocell Science, Inc.
(Exact name of registrant as specified in its Charter.)
Delaware | 001-31564 | 87-0458888 |
(State or other jurisdiction of incorporation) | (Commission File Number) | (I.R.S. Employer Identification No.) |
405 Eagleview Boulevard
Exton, Pennsylvania 19341
(Address of principal executive offices, including zip code)
(484) 713-6000
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Title of Each Class | Name of each exchange on which registered | |
Common Stock, $.001 par value | The NASDAQ Stock Market LLC | |
Securities registered pursuant to Section 12(g) of the Act: None
____________________________________________________________________________________________________________
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes o No ý
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No ý
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ý No o
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ý No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ý
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ¨ | Accelerated filer x |
Non-accelerated filer ¨ (Do not check if a smaller reporting company) | Smaller reporting company ¨ |
Indicate by check mark whether the registrant is a shell company (as defined in the Exchange Act Rule 12b-2). Yes o No ý
The aggregate market value of the registrant's common stock held by non-affiliates was $134.9 million as of June 30, 2015 (the last business day of the registrant’s most recently completed second fiscal quarter), based on a total of 25,594,501 shares of common stock held by non-affiliates and on a closing price of $5.27 as reported on NASDAQ on June 30, 2015.
As of March 4, 2016, there were 43,898,785 shares of the registrant's common stock, par value $0.001 per share, outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive proxy statement for its 2016 annual meeting of stockholders are incorporated by reference into Part III of this Form 10-K where indicated. Such definitive proxy statement will be filed with the U.S. Securities and Exchange Commission within 120 days after the year ended December 31, 2015.
TABLE OF CONTENTS
Page | ||
_________________________
Unless the context otherwise indicates, references in this Form 10-K to “Fibrocell,” “the Company,” “we,” “us” and “our” refer to Fibrocell Science, Inc. and its subsidiaries.
Fibrocell, Fibrocell Science and LAVIV® are trademarks of Fibrocell. Other trademarks, trade names and service marks appearing in this Form 10-K are the property of their respective owners.
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Form 10-K contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. These statements include, among others, statements about:
• | the sufficiency of our cash and cash equivalents to fund our operations into the fourth quarter of 2016; |
• | our ability to obtain additional capital in sufficient amounts and on terms acceptable to us, and the consequences of failing to do so; |
• | future expenses and capital expenditures; |
• | our expectation to announce primary endpoint results of our Phase II clinical trial for azficel-T in the second quarter of 2016; |
• | our interpretation of the U.S. Food and Drug Administration's ("FDA") feedback relating to our Investigational New Drug ("IND") application for FCX-007 and our plans to address such feedback and submit an amended IND in the first quarter of 2016; |
• | the initiation, design and timing of our planned Phase I/II clinical trial for FCX-007; |
• | our expectation to submit an IND for FCX-013 to the FDA in 2017; |
• | the implication of results from pre-clinical and clinical trials and other research activities; |
• | our ability to obtain orphan drug designation for FCX-013; |
• | our ability to complete, or obtain modifications to, the postmarketing study required by the FDA for LAVIV; |
• | the potential advantages of our product candidates and technologies; |
• | the scope and duration of intellectual property protection; and |
• | the effect of legal and regulatory developments; |
as well as other statements relating to our future operations, financial performance or financial condition, prospects or other future events. Forward-looking statements appear primarily in the sections of this Form 10-K entitled “Item 1—Business,” “Item 1A—Risk Factors,” “Item 7—Management's Discussion and Analysis of Financial Condition and Results of Operations,” “Item 7A—Quantitative and Qualitative Disclosures About Market Risk,” and “Item 8—Financial Statements and Supplementary Data.” In some cases, you can identify forward-looking statements by words such as “may,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “ongoing,” “scheduled” and similar expressions, although not all forward-looking statements contain these identifying words.
Forward-looking statements are based upon current expectations and assumptions and are subject to a number of known and unknown risks, uncertainties and other factors that could cause actual results to differ materially and adversely from those expressed or implied by such statements. Factors that could cause or contribute to such differences include, but are not limited to, those discussed in this Form 10-K and in particular the risks and uncertainties discussed under "Item 1A—Risk Factors" of this Form 10-K. As a result, you should not place undue reliance on forward-looking statements.
Additionally, the forward-looking statements contained in this Form 10-K represent our views only as of the date of this Form 10-K (or any earlier date indicated in such statement). While we may update certain forward-looking statements from time to time, we specifically disclaim any obligation to do so, even if new information becomes available in the future.
The foregoing cautionary statements are intended to qualify all forward-looking statements wherever they may appear in this Form 10-K. For all forward-looking statements, we claim the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995.
This Form 10-K also contains estimates, projections and other information concerning our industry, our business, and the markets for certain diseases, including data regarding the estimated size of those markets, and the incidence and prevalence of certain medical conditions. Information that is based on estimates, forecasts, projections, market research or similar methodologies is inherently subject to uncertainties and actual events or circumstances may differ materially from events and circumstances reflected in this information. Unless otherwise expressly stated, we obtained this industry, business, market and other data from reports, research surveys, studies and similar data prepared by market research firms and other third parties, industry, medical and general publications, government data and similar sources.
1
Part I
Item 1. Business
Overview
We are an autologous cell and gene therapy company translating personalized biologics into medical breakthroughs. Our approach to personalized biologics is distinctive. We target the underlying cause of disease by using fibroblast cells from a patient's skin to create localized therapies—with or without genetic modification—that are compatible with the unique biology of the patient.
We are focused on discovering and developing localized therapies for diseases affecting the skin, connective tissue and joints to improve the lives of patients and their families. In that regard, we commit significant resources to our research and development programs. Currently, all of our research and development operations and focus are on gaining regulatory approvals to commercialize our product candidates in the United States, however, we may seek to expand into foreign markets in the future.
Our current pre-clinical and clinical development program pipeline consists of the following product candidates:
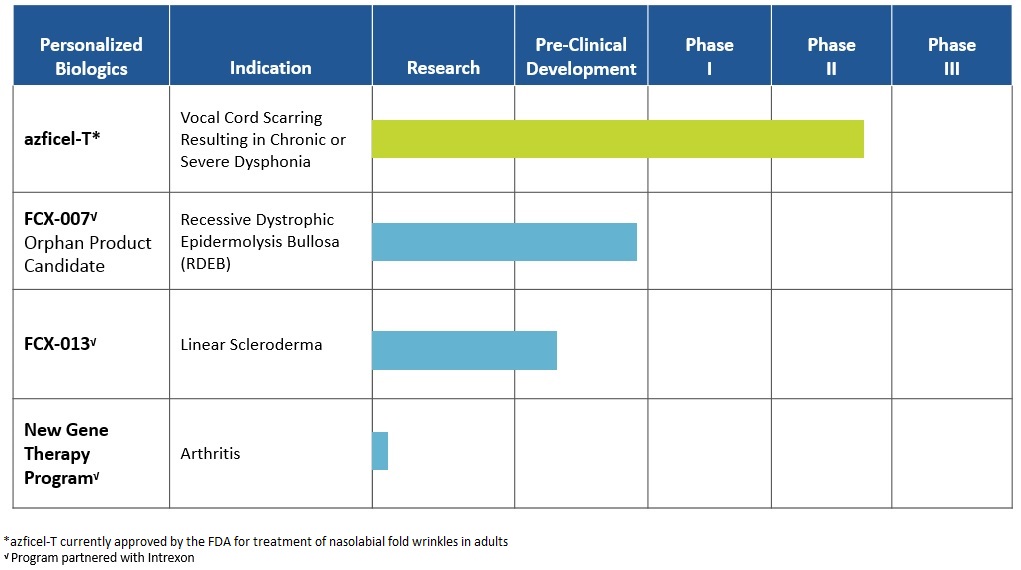
2
Our most advanced development program is azficel-T for the treatment of vocal cord scarring resulting in chronic or severe dysphonia. We are currently investigating this indication in a Phase II clinical trial. We have completed dosing in this trial and expect to announce primary endpoint results in the second quarter of 2016.
In collaboration with Intrexon Corporation ("Intrexon"), we have two gene-therapy product candidates in pre-clinical development and a third gene-therapy program in the research phase. Our lead gene-therapy product candidate, FCX-007, is in late-stage pre-clinical development for the treatment of recessive dystrophic epidermolysis bullosa (“RDEB”). RDEB is a devastating, rare, congenital, painful, progressive, blistering skin disease that often leads to premature death. FCX-007 has received orphan drug designation as well as rare pediatric disease designation from the FDA. Our second gene-therapy product candidate, FCX-013, is in pre-clinical development for the treatment of linear scleroderma. Linear scleroderma is a localized autoimmune skin disorder that manifests as excess production of extracellular matrix that causes tightening and hardening of the skin and is characterized by linear areas of fibrosis, or skin thickening. We plan to seek orphan drug designation for FCX-013. Our third gene-therapy program is focused on the treatment of arthritis and is in the research phase.
Our Strategy
Our strategy is to develop and commercialize transformational therapies for diseases affecting the skin, connective tissue and joints to improve the lives of patients and their families. Key elements of our strategy are:
• | Leveraging our FDA-approved BLA for azficel-T to investigate and seek approval for additional therapeutic indications, including but not limited to vocal cord scarring resulting in chronic or severe dysphonia; |
• | Advancing our gene-therapy product candidates, FCX-007 and FCX-013, into human clinical trials; |
• | Advancing our gene-therapy program focused on arthritis through research and into pre-clinical development; and |
• | Leveraging our FDA-inspected, cGMP manufacturing facility and our expertise in cell therapy manufacturing to advance the development of our autologous cell and gene therapy pipeline. |
Our Personalized Biologics Platform
The foundation of our personalized biologics platform is our proprietary autologous fibroblast technology. Fibroblasts are the most common cell in skin and connective tissue and are responsible for synthesizing extracellular matrix proteins, including collagen and other growth factors, that provide structure and support. Because fibroblasts naturally reside in the localized environment of the skin and connective tissue, they represent an ideal therapeutic agent for the treatment of diseases affecting the skin, connective tissue and joints. Utilizing our autologous fibroblast technology, we use a patient's fibroblast cells to create therapies—with or without genetic modification—that are compatible with the unique biology of the patient (i.e., autologous).

3
One Powerful Platform Drives Two Product Engines
Our personalized biologics platform is comprised of two separate product engines, each of which uses our proprietary autologous fibroblast technology.
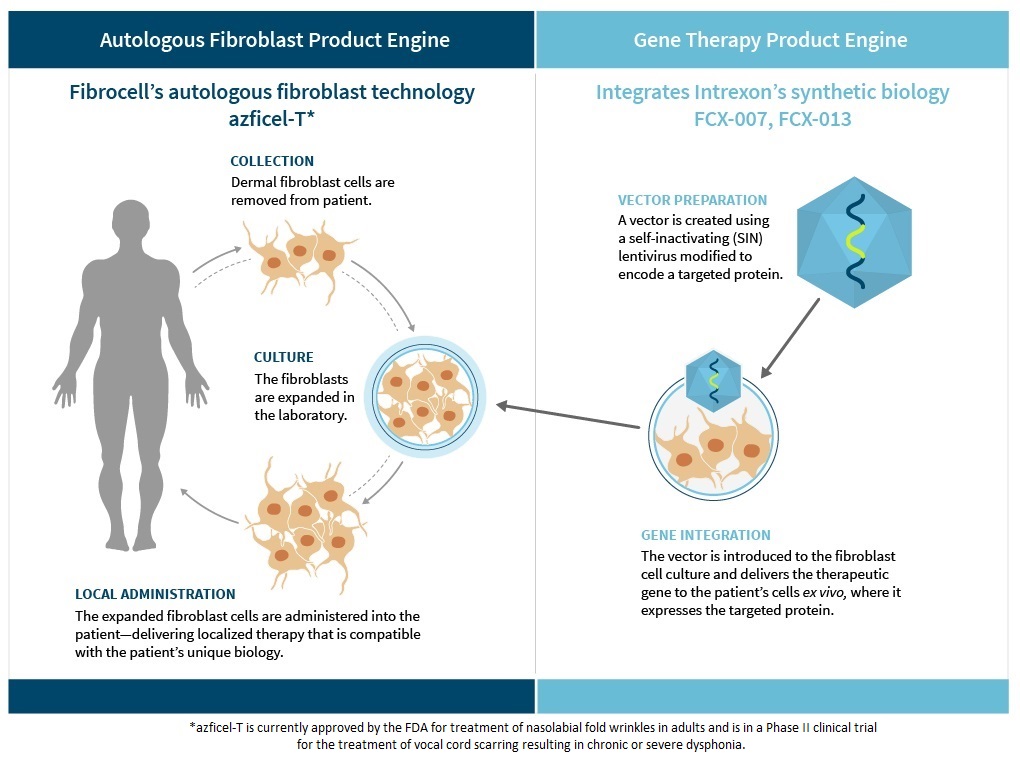
Autologous Fibroblast Product Engine
Our Autologous Fibroblast Product Engine utilizes fibroblast cells to uniquely target the localized environment of skin and connective tissue, which are generally difficult to treat with systemic drug therapies due to limited blood flow to these areas. In 2011, we obtained FDA-approval of LAVIV (azficel-T). LAVIV uses autologous fibroblast cells to improve the appearance of moderate to severe nasolabial fold wrinkles in adults. Although we shifted our strategic focus away from the aesthetics market in 2013, we are leveraging our FDA-approved biologics license application ("BLA") for LAVIV to investigate the use of azficel-T in other therapeutics areas. Currently, we are investigating azficel-T in a Phase II clinical trial for the treatment of vocal cord scarring resulting in chronic or severe dysphonia.
Gene Therapy Product Engine
Our Gene Therapy Product Engine integrates our autologous fibroblast technology with the synthetic biology technology of our collaborator, Intrexon. By combining these technologies, we are genetically modifying autologous fibroblast cells to express targeted proteins in order to address the underlying cause of debilitating diseases affecting the skin, connective tissue and joints. Through our collaboration with Intrexon, we have access to:
• | Intrexon’s proprietary vector technology, which is designed to facilitate the assembly and delivery of the necessary target gene constructs for delivery to autologous fibroblast cells. Access to this technology allows us to rapidly screen and construct genetic therapeutic solutions. |
4
• | Intrexon’s proprietary RheoSwitch Therapeutic System® (RTS®) technology. The RTS® biologic switch is activated by an orally-administered compound that provides the ability to control level and timing of protein expression in those diseases where such control is ideal. |
Currently, we have two gene-therapy product candidates in pre-clinical development, FCX-007 for the treatment of RDEB and FCX-013 for the treatment of linear scleroderma. A third gene-therapy program, focused on the treatment of arthritis, is in the research phase.
Advantages of Our Approach
We believe our personalized biologics approach provides the following distinct advantages for creating cell and gene therapies:
• | Localized administration—avoids side effects typically associated with systemic therapy |
• | Reduced rejection concerns—because autologous fibroblasts are compatible with the unique biology of each patient |
• | Fibroblast cells are genetically modified ex vivo—to enable testing for safety and confirmation of protein expression levels prior to administration to the patient |
• | Demonstrated expertise in manufacturing our fibroblast cell therapy |
Development Programs
Our development programs are focused on diseases affecting the skin, connective tissue and joints for which there are high unmet needs. Our programs consist of the following:
Program | Potential Indication | Status | ||
azficel-T | Vocal Cord Scarring resulting in Chronic or Severe Dysphonia | Phase II Clinical Trial | ||
FCX-007 | RDEB | Pre-clinical | ||
FCX-013 | Linear Scleroderma | Pre-clinical | ||
New Gene Therapy Program | Arthritis | Research | ||
azficel-T for Vocal Cord Scarring
Vocal cord scarring is caused by damage to the fibroblast layer of the vocal cords which reduces vocal cord elasticity and airflow, affecting voice tone and volume. This reduction in vocal capacity is referred to as dysphonia, severe cases of which can lead to a total loss of voice. Current treatments for vocal cord scarring, which include voice therapy and surgery through the use of injection (collagen, fat, calcium, hyaluronic acid) or implant (PTFE, silastic), only address the symptoms of vocal cord scarring and have inconsistent efficacy.
azficel-T is in development to treat patients suffering from vocal cord scarring that is either idiopathic or age-related, of which we estimate there to be approximately 64,000 in the U.S. We believe azficel-T restores the extracellular matrix to repair damage to the fibroblast layer of the vocal cords, thereby improving voice quality. This program is being conducted under an IND that cross-references our FDA-approved BLA for LAVIV, which allows us to leverage the safety, chemistry and manufacturing data contained in the BLA.
Phase I Trial
In our Phase I open label clinical trial of azficel-T for the treatment of vocal cord scarring resulting in chronic or severe dysphonia, we examined the safety and efficacy of azficel-T injections for patients who had failed to improve following currently available treatments. Five patients were treated in the clinical trial. For patients with only one impacted vocal cord, azficel-T was injected into the vocal cord over three separate injection sessions approximately 28 days apart. For patients with both vocal cords impacted, azficel-T was injected into the vocal cords over six separate injection sessions approximately 14 days apart, alternating vocal cords at each injection session.
The voice quality of each patient was evaluated using Mucosal Wave Grade assessment, Voice Handicap Index and patient-assessed voice quality prior to treatment and at 4 and 12 months following treatment. For the Mucosal Wave Grade assessment, each patient phonates a sustained vowel while the vocal cord vibration is recorded using a videostroboscope. The
5
recording is then assessed and scored on a 1 (no wave present) to 5 (normal wave) scale. The pliability of the vocal cords can be visualized as waves of mucosa on the vocal cord surface during vibration. The deficiency of the vocal cord tissue results in lack of pliability of the cord, resulting in a reduction of score. The Voice Handicap Index is a questionnaire to quantify the functional, physical and emotional impacts of a voice disorder on a patient's quality of life. The scale contains 30 items in three categories, scored 0 (never) to 4 (always) per item. An 18-point reduction in the overall Voice Handicap Index score is considered to be a significant change in voice quality.
The data from this trial showed a positive trend of sustained improvement in a majority of the patients in the Mucosal Wave Grade assessment and Voice Handicap Index. The mean results for all patients are set forth below:
◦ | Mucosal Wave Grade |
▪ | Mean baseline result = 1.3 |
▪ | Mean 4-month result = 3 (1.7 improvement over baseline) |
▪ | Mean 12-month result = 3.22 (1.92 improvement over baseline) |
◦ | Voice Handicap Index |
▪ | Mean baseline result = 83.8 |
▪ | Mean 4-month result = 55.8 (-28 improvement over baseline) |
▪ | Mean 12-month result = 52.6 (-31.2 improvement over baseline) |
No serious adverse events were reported in the study. Three of the five patients reported ear pain with a majority of the cases being mild or severe in severity. Voice alternation was noted in one subject with a duration of one day, and was categorized as possibly related to treatment. All other reported adverse events were considered to be unrelated to the study treatment and there were no laboratory abnormalities or other untoward events that were considered related to study treatment. Based on these results, azficel-T was well-tolerated in this patient population.
Phase II Trial
Our Phase II clinical trial that is currently in progress is a double-blind, randomized, placebo-controlled trial that is designed to test the safety and efficacy of azficel-T injections in patients with chronic dysphonia caused by vocal cord scarring or atrophy. In this clinical trial, patients were administered the same dose at the same time intervals as in the Phase I clinical trial. We have completed dosing for all 21 patients currently enrolled in the trial. Efficacy endpoints will be assessed four months after administration of final treatment on three different scales: Voice Handicap Index, Mucosal Wave Grade, and GRBAS (grade, roughness, breathiness, asthenia & strain). For GRBAS, a recording of the patient's voice is assessed for grade, roughness, breathiness, asthenia and strain, and scored on a four-point scale where, 0 = normal, 1 = mild, 2= moderate, and 3 = severe. We expect to report primary endpoint results in the second quarter of 2016. As of March 8, 2016, no treatment-related serious adverse events have been reported.
FCX-007 for RDEB
Recessive dystrophic epidermolysis bullosa is a congenital, progressive, devastatingly painful and debilitating genetic disorder that often leads to death, and is the most severe form of dystrophic epidermolysis bullosa. RDEB is caused by a mutation of the COL7A1 gene. The COL7A1 gene encodes for type VII collagen (“COL7”), a protein that forms anchoring fibrils. Anchoring fibrils hold together the layers of skin, and without them, skin layers separate causing severe blistering, open wounds and scarring in response to any kind of friction, including normal daily activities like rubbing or scratching. Children who inherit this condition are often called “butterfly children” because their skin can be as fragile as a butterfly’s wings. We estimate that there are approximately 1,100 - 2,500 people suffering from RDEB in the U.S. Current treatments for RDEB, which include daily bandaging, hydrogel dressings, antibiotics, feeding tubes, and surgery (hand and esophageal), only address the symptoms of the disorder.
6

Our lead gene-therapy product candidate, FCX-007, is in pre-clinical development for the treatment of RDEB. FCX-007 is an autologous fibroblast cell genetically modified to express COL7 for localized treatment of RDEB. FCX-007 is designed to be delivered to the dermal layer where the genetically-modified fibroblast cells will produce COL7 to promote the formation of anchoring fibrils to aid in wound closure and prevention of wound recurrence. We are developing FCX-007 in collaboration with Intrexon.
FCX-007 was evaluated for toxicology and biological proof-of-concept in RDEB and normal human skin xenografts implanted onto severe combined immunodeficiency (SCID) mice. Toxicology results for FCX-007 showed, at two- and six-weeks post-administration, that there were:
• | No treatment-related findings; |
• | No tumors in the skin grafts or other organs; |
• | No statistical changes in blood chemistry; and |
• | No apparent systemic distribution of the vector. |
In addition, data supporting the biological proof-of-concept indicated FCX-007 cells in a human skin xenograft SCID mice model expressed COL7 that localized to the basement membrane zone where anchoring fibrils are formed suggesting that the biological mechanism of action of FCX-007 was verified.
We submitted an IND for FCX-007 to the FDA in July 2015. In September 2015, we received feedback from the FDA on the IND which required us to delay the initiation of our proposed Phase I/II clinical trial. The FDA’s feedback related to the areas of chemistry, manufacturing and controls, toxicology and our proposed Phase I/II clinical trial protocol. Although the hybrid pharmacology/toxicology study performed based on the injection of FCX-007 into human skin that was xenografted onto SCID mice was included in the IND and showed no signs of toxicity, the FDA requested that we execute a toxicology-specific study in which FCX-007 is injected in non-grafted SCID mice. We have initiated this new toxicology study, and we expect to amend the IND in response to the FDA’s feedback and to include data from the new study in the first quarter of 2016. Subject to successful completion of the new toxicology study and satisfactorily addressing the FDA’s other feedback, we expect to initiate a Phase I/II clinical trial for FCX-007 in the second quarter of 2016.
FCX-013 for Linear Scleroderma
Linear scleroderma is a localized autoimmune skin disorder that manifests as excess production of extracellular matrix, specifically type I collagen and type III collagen, resulting in fibrosis and linear scars. The linear areas of skin thickening may extend to underlying tissue and muscle in children which may impair growth and development. Lesions appearing across joints can be painful, impair motion and may be permanent. Current treatments for linear scleroderma, which include systemic or topical corticosteroids, UVA light therapy and physical therapy, only address the symptoms of the disorder. We estimate the U.S. population of patients who have linear scleroderma over a major joint and exhibit severe joint pain to be approximately 40,000.
Our second gene-therapy product candidate, FCX-013, is in pre-clinical development for the treatment of linear scleroderma. FCX-013 is an autologous fibroblast cell genetically modified to express a protein to breakdown collagen accumulation at the site of the localized disease. FCX-013 incorporates Intrexon’s proprietary RTS® switch, a biologic switch activated by an orally administered compound to control protein expression once the initial fibrosis has been resolved.
7
FCX-013 is designed to be injected under the skin at the location of the fibrosis where the genetically-modified fibroblast cells will produce a protein to break down excess collagen accumulation. The patient takes an oral compound to facilitate protein expression. Once the fibrosis is resolved, the patient will stop taking the oral compound which will stop further production of the subject protein by FCX-013.
We have successfully completed a proof-of-concept study for FCX-013 in which the primary objective was to determine whether FCX-013 had the potential to reduce dermal thickness in fibrotic tissue. In this study, FCX-013 was evaluated in a bleomycin-induced scleroderma model utilizing SCID mice. Data from the study demonstrated that FCX-013 reduced dermal thickness of fibrotic tissue to levels similar to that of the non-treated control and further reduced the thickness of the sub-dermal muscle layer. FCX-013 will now be advanced into dose-ranging and toxicology/biodistribution studies for product optimization. We expect to submit an IND for FCX-013 to the FDA in 2017.
New Gene Therapy Program for Arthritis
Arthritis is a broad term that covers a group of more than 100 different types of diseases that affect the joints, as well as connective tissues and organs, including the skin. According to the Centers for Disease Control and Prevention, arthritis—characterized by joint inflammation, pain, and decreased range of motion—is the United States’ most common cause of disability affecting more than 52 million adults as well as 300,000 children at a cost exceeding $120 billion.
Our third gene-therapy program is focused on the treatment of arthritis. This program is in the research phase and is being undertaken in collaboration with Intrexon. Our goal is to deliver a protein therapy locally to the joint to provide sustained efficacy while avoiding key side effects typically associated with systemic therapy.
Commercial Program
LAVIV (azficel-T) for Nasolabial Fold Wrinkles
LAVIV (azficel-T) was approved by the FDA in June 2011 for the improvement of the appearance of moderate to severe nasolabial fold wrinkles in adults. LAVIV utilizes our proprietary autologous fibroblast technology. In 2013, we shifted our strategic focus away from the aesthetic market and towards developing treatments for diseases affecting the skin, connective tissue and joints. As a result, we no longer actively market or promote LAVIV but we continue to accept prescriptions, from which we expect a nominal amount of revenue during 2016.
A condition to the FDA's approval of LAVIV was that we conduct a 2,700 patient post-marketing study by the end of 2016 to assess the risk of skin cancer (such as basal cell cancer) in the area of LAVIV injections and the risk of immune-mediated hypersensitivity reactions (such as leukocytoclastic vasculitis). We have initiated enrollment in this study and have submitted the required biannual interim reports to the FDA, with the most recent being in January 2016. However, given the limited use of LAVIV, we have experienced difficulties in recruiting a sufficient number of patients for this study. We are actively engaged in discussions with the FDA about how to fulfill the study size requirement in light of the limited population of LAVIV users.
Intrexon Collaborations
2012 Exclusive Channel Collaboration Agreement ("2012 ECC")
In October 2012, we entered into an Exclusive Channel Collaboration Agreement, with Intrexon, which was amended in June 2013 and January 2014 (as amended, the "2012 ECC") pursuant to which we are Intrexon's exclusive channel collaborator in the research, development and commercialization of products in the following areas (the "2012 Fields"):
• | the enhanced production and purification of autologous fibroblasts (without genetic modification) for all aesthetic and therapeutic indications; |
• | the enhanced production and purification of autologous dermal cells (without genetic modification) for aesthetic and therapeutic treatment of dermal, vocal cord, and periodontal indications; |
• | the development of genetically modified autologous fibroblasts for all aesthetic and therapeutic indications where an autologous fibroblast itself is the principal effector of the product in contrast to the use of autologous fibroblasts as the source of expression of a systemically available therapeutic protein in which that protein (and not the fibroblast) is the principal therapeutic effector; |
8
• | the development of genetically modified autologous dermal cells for aesthetic and therapeutic treatment of dermal, vocal cord, and periodontal indications; |
• | autologous fibroblasts genetically modified to express a therapeutic protein and/or bioactive ribonucleic acid for the treatment of autoimmune and non-infectious inflammatory disorders that manifest in cutaneous tissues, fascia and/or muscle; and |
• | autologous human fibroblasts with gene therapy to express bioactive Tenascin-X locally to correct connective tissue disorders associated with Ehlers-Danlos Syndrome (hypermobility type). |
Pursuant to the terms of the 2012 ECC, Intrexon has granted us a license to use its proprietary technologies and other intellectual property to research, develop and commercialize products in the 2012 Fields within the United States. We are responsible for all costs incurred in connection with the research, development and commercialization of products under the 2012 ECC and own all clinical data, regulatory filings and regulatory approvals relating to such products. We engage Intrexon for support services for the research and development of products under the 2012 ECC, and reimburse Intrexon for its cost for time and materials for such services.
We are required to pay Intrexon quarterly cash royalties on all products developed under the 2012 ECC in an amount equal to 7% of aggregate annualized net sales up to $100 million, plus 14% on aggregate annualized net sales greater than $100 million. We are also required to pay Intrexon half of any sublicensing revenues we receive from third parties in consideration for sublicenses granted by us with respect to products developed under the 2012 ECC, but only to the extent such sublicensing revenues are not included in net sales subject to royalties. Sales from LAVIV (azficel-T), including new indications, or other products that we develop and commercialize outside of the 2012 ECC are not subject to royalty payments unless we are able to reduce the product's cost of goods sold through the 2012 ECC, in which case, we are required to pay quarterly cash royalties on such products equal to one-third of the cost of goods sold savings less any such savings developed by us outside of the 2012 ECC.
The 2012 ECC may be terminated by Intrexon if we fail to exercise diligent efforts in developing products through the collaboration or if we elect not to pursue the development of a therapy identified by Intrexon within the 2012 Field and that qualifies as a “Superior Therapy” as defined in the 2012 ECC. Upon such termination, the products covered by the 2012 ECC in active and ongoing Phase II clinical trials or later stage development shall be entitled to be continued by us with a continuation of the related milestone, royalty and other payment obligations for such products, and all rights to products covered by the 2012 ECC still in an earlier stage of development shall revert to Intrexon.
In September 2015, we and Intrexon entered into a letter of agreement pursuant to which we mutually agreed to terminate our collaboration with respect to the development of potential therapies to treat Ehlers-Danlos Syndrome (hypermobility type) due to technical hurdles. As a result, we no longer have any rights or obligations under the 2012 ECC with respect to the development of “autologous human fibroblasts genetically modified to express bioactive Tenascin-X locally to correct connective tissue disorders”.
Currently, we are in pre-clinical development of two gene-therapy product candidates, FCX-007 and FCX-013, under the 2012 ECC.
2015 Exclusive Channel Collaboration Agreement ("2015 ECC")
In December 2015, we entered into an additional Exclusive Channel Collaboration Agreement with Intrexon
(the "2015 ECC") pursuant to which we are Intrexon's exclusive channel collaborator in the research, development
and commercialization of products for the treatment of chronic inflammation and degenerative diseases of human joints through intra-articular or other local administration of genetically-modified fibroblasts (the "2015 Field"). The collaboration leverages our autologous fibroblast technology with Intrexon's synthetic biology technology to identify and develop cell-based therapeutics that will be genetically modified to express one or more proteins at sites of joint inflammation. We believe this treatment approach has the potential to overcome the limitations of existing therapies for chronic inflammation and degenerative diseases of the joint, including arthritis and related conditions.
Pursuant to the terms of the 2015 ECC, Intrexon has granted us a license to use its proprietary technologies and
other intellectual property to develop and commercialize products in the 2015 Field throughout the world. We are responsible for all costs incurred in connection with the research, development and commercialization of products under the 2015 ECC and own all clinical data, regulatory filings and regulatory approvals relating to such products. We engage Intrexon for support services in connection with the research and development of products under the 2015 ECC, and reimburse Intrexon for its cost for time and materials for such services.
9
For each product that we develop under the 2015 ECC, we are required to pay Intrexon development milestones of up to $30 million and commercialization milestones of up to $22.5 million, a low double-digit royalty on our net sales of such products and half of any sublicensing revenues we receive from third parties in consideration for sublicenses granted by us with respect to such products but only to the extent such sublicensing revenues are not included in net sales subject to royalties.
The 2015 ECC may be terminated by Intrexon if we fail to exercise diligent efforts in developing products through the collaboration or if we elect not to pursue the development of a therapy identified by Intrexon within the 2015 Field and that qualifies as a “Superior Therapy” as defined in the 2015 ECC. Upon such termination, the products covered by the 2015 ECC in active and ongoing Phase II clinical trials or later stage development shall be entitled to be continued by us with a continuation of the related milestone, royalty and other payment obligations for such products, and all rights to products covered by the 2015 ECC still in an earlier stage of development shall revert to Intrexon.
Currently, we are in the research phase for a gene-therapy product for arthritis under the 2015 ECC.
UCLA Collaboration
In May 2014, we entered into a research agreement with The Regents of the University of California ("UCLA") pursuant to which we and UCLA agreed to undertake collaborative research activities over a three to five year period to develop novel intellectual property and products relating to (a) somatic dermal derivatives and (b) human induced pluripotent stem cells. In connection with this collaboration, we and UCLA also entered into an exclusive license agreement in June 2014 pursuant to which UCLA granted us an exclusive, sublicensable right and license to use certain intellectual property developed under this program for all research, development and commercialization purposes. In consideration for the license, we are required to pay to UCLA a license issue fee, a license maintenance fee (waived if earned royalties are paid), certain one-time milestone payments, earned royalties on net sales of licensed products (including sales by affiliates) and a percentage of amounts received from sublicensing activities. We are also subject to minimum annual royalty payments to UCLA beginning after the first commercial sale of a licensed product.
We had previously been a party to additional collaborations with UCLA relating to isolating stem cell sub-populations in the skin; however, we terminated these programs and related agreements during 2015.
Manufacturing
We lease and operate our own manufacturing facility located in Exton, Pennsylvania. We use this facility to manufacture our non-genetically modified products. We outsource the manufacturing for our genetically-modified product candidates to a contract manufacturer with a facility located in Mountain View, California. We and our contract manufacturer are subject to routine inspections in accordance with the FDA's Current Good Manufacturing Practices (“cGMP”). We believe that we and our contract manufacturer have adequate manufacturing capacity to satisfy our clinical demands, as well as the limited commercial demand we expect during 2016.
The fibroblast cells that constitute our product candidates are grown by our proprietary manufacturing process which begins with the collection of skin biopsies from behind the ear on the patient’s skin. The biopsies are then sent to us for processing according to cGMP and, for our genetically-modified product candidates, cell engineering. The cells are then expanded using tissue culture techniques and cryo-preserved. When a treatment is requested, the cells are further processed and prepared for shipment.
All component parts, including raw materials and other supplies utilized in our manufacturing process are available from various third party suppliers and manufacturers in quantities adequate to meet our needs. We seek to ensure continuity of supply of such component parts, raw materials and supplies using a strategy of dual sourcing, where possible. Some of our raw materials are currently sourced from one vendor; however, alternate vendors are available should they be required, although we would need sufficient lead time to qualify those vendors.
We use certain hazardous chemicals and biological materials in our manufacturing process which are subject to a variety of federal, state and local laws and regulations governing, among other matters, the use, generation, manufacture, transportation, storage, handling, disposal of and human exposure to these materials, including regulation by governmental regulatory agencies, such as the Occupational Safety and Health Administration and the U.S. Environmental Protection Agency. We incur capital and operating expenditures and other costs in the ordinary course of our business in complying with these laws and regulations. We dispose of minimal hazardous biological waste as a result of our manufacturing process.
10
Intellectual Property
We believe that patents, trademarks, copyrights and other proprietary rights are important to our business. We also rely on trade secrets, know-how and continuing technological innovations to develop and maintain our competitive position. We seek to protect our intellectual property rights by a variety of means, including obtaining patents, maintaining trade secrets and proprietary know-how and technological innovation to operate, without infringing on the proprietary rights of others and to prevent others from infringing on our proprietary rights.
As of December 31, 2015, we own or license 11 issued U.S. patents, 9 pending U.S. patent applications, 10 granted foreign patents, 1 pending international patent and 14 pending foreign patent applications. Our issued patents and patent applications primarily cover the method of using autologous cell fibroblasts for the repair of skin and soft tissue defects and the use of autologous fibroblast cells for tissue regeneration. In particular, we own issued patents in the U.S. and in other countries that are directed to methods of long-term augmentation of subcutaneous or dermal tissue by injecting an effective amount of a suspension of autologous passaged dermal fibroblasts into subadjacent tissue, which covers the approved use of LAVIV, as well as azficel-T for the treatments of vocal cord scarring and restrictive burn scarring, and which are set to expire in the U.S. in July 2020. In addition, we own an issued U.S. patent and pending applications in Australia, Canada, China, Europe, India, Japan, South Korea, Hong Kong and the U.S. directed to dosage formulations for injection containing particular amounts of autologous human fibroblasts and uses thereof, which also covers LAVIV as well as azficel-T for the treatments of vocal cord and restrictive burn scarring, and which naturally expire in 2030 and 2031. We also own pending applications in the U.S. and several foreign countries related to topical formulations of autologous dermal fibroblasts and uses thereof, the earliest of which, if issued, would naturally expire in 2027.
Competition
There is significant competition in the biopharmaceutical industry which can be attributed to companies ranging from small specialized biotechnology firms to large well-established pharmaceutical companies. More specifically, there are many companies currently competing in drug development for new therapies for the treatment of diseases affecting the skin, connective tissue and joints, our focus area. Some of our competitors have substantially greater financial resources and larger research and development organizations. In addition, our experience in clinical trials, obtaining FDA and other regulatory approvals, manufacturing and commercialization of products may be more limited.
Product competition is based on a variety of factors, including but not limited to: product safety, efficacy, convenience of dosing, availability, price, as well as brand recognition. Our product candidates, if approved for commercial use, will contend with treatments offered by our competitors. Although we believe the autologous nature and localized treatment approach of our product candidates provide advantages over our competitors, existing and new treatments may also possess certain advantages. Additionally, the development of other drug technologies and methods of treating diseases are occurring at a rapid pace. These developments may render our products or technologies obsolete or noncompetitive. Currently, we believe the primary competitors for our product candidates are as follows:
azficel-T for Vocal Cord Scarring. Our product candidate azficel-T is being developed to treat vocal cord scarring resulting in chronic or severe dysphonia. Current treatments for vocal cord scarring, which include voice therapy and surgery through the use of injection (collagen, fat, calcium, hyaluronic acid) or implant (PTFE, silastic), only address the symptoms of this condition. PROLARYN™ (injectable implant) is currently marketed by Merz North America, Inc. as a treatment for dysphonia and vocal cord insufficiency.
FCX-007 for RDEB. Our product candidate FCX-007 is being developed for the treatment of RDEB. Current treatments for RDEB, which include bandaging, antibiotics, feeding tubes, and surgery (hand and esophageal), only address the symptoms of this disorder. There are currently no products approved by the FDA for the treatment of RDEB. We are aware of a potentially competing product, ZORBLISA™ (SD-101 Cream), which is being developed by Amicus Therapeutics and is in a Phase III trial for the treatment of epidermolysis bullosa.
FCX-013 for Linear Scleroderma. Our product candidate FCX-013 is being developed for the treatment of linear scleroderma. Current treatments for linear scleroderma, which include systemic or topical corticosteroids, UVA light therapy, and physical therapy, only address the symptoms of the disorder. There are currently no products approved by the FDA for the treatment of linear scleroderma. We are aware of a potentially competing product, ECCS-50 Cellular Therapy, which is being developed by Cytori Therapeutics and is in a Phase III clinical trial for the treatment of scleroderma that affects the hands. We are also aware that miRagen Therapeutics has a product candidate utilizing microRNA biology in a Phase I clinical trial for the treatment of systemic and localized scleroderma.
11
Research and Development
We expense research and development costs as they are incurred. For the years ended December 31, 2015, 2014 and 2013, we incurred total research and development expenses of $25.9 million, $17.7 million and $13.8 million, respectively. Additionally, for the year ended December 31, 2015, we incurred expenses of $0.3 million related to a research and development agreement that the Company has with a third party to investigate potential new non-pharmaceutical applications for the Company's conditioned fibroblast media technology. Expenses pertaining to this collaboration agreement are classified under the caption "Cost of collaboration revenue" in the Consolidated Statements of Operations. No such expenses were incurred during the years ended December 31, 2014 or 2013.
Government Regulation
We are subject to extensive government regulation, principally by the FDA and state and local authorities in the United States and by comparable agencies in foreign countries. Governmental authorities in the United States extensively regulate the pre-clinical and clinical testing, safety, efficacy, research, development, manufacturing, labeling, storage, record-keeping, advertising, promotion, import, export, marketing and distribution, among other things, of pharmaceutical and biologic products under various federal laws including the Federal Food, Drug and Cosmetic Act ("FFDCA"), the Public Health Service Act ("PHSA") and under comparable laws by the states and in most foreign countries.
Domestic Regulation
In the United States, the FDA, under the FFDCA, the PHSA, and other federal statutes and regulations, subjects pharmaceutical and biologic products to rigorous review. If we do not comply with applicable requirements, we may be fined, the government may refuse to approve our marketing applications or allow us to manufacture or market our products or product candidates, and we may be criminally prosecuted. The FDA also has the authority to discontinue or suspend manufacture or distribution, require a product withdrawal or recall or revoke previously granted marketing authorizations if we fail to comply with regulatory standards or if we encounter problems during commercial operations.
FDA Approval Process
To obtain approval of a new product from the FDA, we must, among other requirements, submit data demonstrating the product’s safety and efficacy as well as detailed information on the manufacture and composition of the product candidate. In most cases, this entails extensive laboratory tests and pre-clinical and clinical trials. This testing and the preparation of necessary applications and processing of those applications by the FDA are expensive and typically take many years to complete. The FDA may deny our applications or may not act quickly or favorably in reviewing these applications, and we may encounter significant difficulties or costs in our efforts to obtain FDA approvals that could delay or preclude us from marketing any products we may develop. The FDA also may require post-marketing testing and surveillance to monitor the effects of approved products or place conditions on any approvals that could restrict the commercial applications of these products. Regulatory authorities may withdraw product approvals if we fail to comply with regulatory standards or if we encounter problems following initial marketing. With respect to patented products or technologies, delays imposed by the governmental approval process may materially reduce the period during which we may have the exclusive right to exploit the products or technologies.
The FDA does not apply a single regulatory scheme to human tissues and the products derived from human tissue. On a product-by-product basis, the FDA may regulate products as drugs, biologics, or medical devices, in addition to regulating them as human cells, tissues, or cellular or tissue-based products (“HCT/P”), depending on whether or not the particular product triggers any of an enumerated list of regulatory factors. A fundamental difference in the treatment of products under these classifications is that the FDA generally permits HCT/Ps that do not trigger any of those regulatory factors to be commercially distributed without regulatory approval. In contrast, products that trigger those factors, such as if they are more than minimally manipulated when processed or manufactured, are regulated as drugs, biologics, or medical devices and require FDA approval. We have determined that LAVIV and our product candidates trigger regulatory factors that make them biologics, in addition to an HCT/P, and consequently, we must obtain approval from the FDA before marketing such products and must also satisfy all regulatory requirements for HCT/Ps.
The process required by the FDA before a new drug or biologic may be marketed in the United States generally involves the following:
• | completion of pre-clinical laboratory tests or studies and formulation studies; |
12
• | submission to the FDA of an IND application for a new drug or biologic, which must become effective before human clinical trials may begin; |
• | performance of adequate and well-controlled human clinical trials to establish the safety and efficacy of the proposed drug or biologic for its intended use; |
• | detailed information on product characterization and manufacturing process; and |
• | submission and approval of a New Drug Application (“NDA”) for a drug, or a BLA for a biologic. |
Pre-clinical tests include laboratory evaluation of product chemistry formulation and stability, as well as animal and other studies to evaluate toxicity. Under FDA regulations, the results of any pre-clinical testing, together with manufacturing information and analytical data, are submitted to the FDA as part of an IND application. The FDA requires a 30-day waiting period after the filing of each IND application before clinical trials may begin, in order to ensure that human research patients will not be exposed to unreasonable health risks. At any time during this 30-day period or at any time thereafter, the FDA may halt proposed or ongoing clinical trials, may authorize trials only on specified terms, or may require additional trials. The IND application process may become extremely costly and substantially delay development of our products. Moreover, positive results of pre-clinical tests will not necessarily indicate positive results in clinical trials.
The sponsor typically conducts human clinical trials in three sequential phases, which may overlap. These phases generally include the following:
• | Phase I: The product candidate is usually first introduced into healthy humans or, on occasion, into patients, and is tested for safety, dosage tolerance, absorption, distribution, excretion and metabolism; |
• | Phase II: The product candidate is introduced into a limited patient population to: |
• | assess its efficacy in specific, targeted indications; |
• | assess dosage tolerance and optimal dosage; and |
• | identify possible adverse effects and safety risks. |
• | Phase III: These are commonly referred to as pivotal studies. If a product candidate is found to have an acceptable safety profile and to be potentially effective in Phase II clinical trials, clinical trials in Phase III will be initiated to further demonstrate clinical efficacy, optimal dosage and safety within an expanded and diverse patient population at geographically dispersed clinical trial sites; and |
• | If the FDA does ultimately approve the product candidate, it may require post-marketing testing, including potentially expensive Phase IV studies, to confirm or further evaluate its safety and effectiveness. Continued ability to commercialize the product may be based on the successful completion of these additional studies. |
Before proceeding with a trial, the sponsor may seek a written agreement from the FDA regarding the design, size, and conduct of a clinical trial. This is known as a Special Protocol Assessment (“SPA”). Among other things, SPAs can cover clinical trials for pivotal studies whose data will form the primary basis to establish a product’s efficacy. SPAs thus help establish up-front agreement with the FDA about the adequacy of a clinical trial design to support a regulatory approval, but the agreement is not binding if new circumstances arise. Even if the FDA agrees to a SPA, the agreement may be changed by the sponsor or the FDA on written agreement by either parties, or if a senior FDA official determines that a substantial scientific issue essential to determining the safety or effectiveness of the product was identified after the testing began. There is no guarantee that a study will ultimately be adequate to support an approval, even if the study is subject to a SPA. The FDA retains significant latitude and discretion in interpreting the terms of the SPA and the data and results from any study that is the subject of the SPA.
Clinical trials must meet requirements for Institutional Review Board (“IRB”) oversight, patient informed consent and the FDA’s Good Clinical Practice (“GCP”). Prior to commencement of each clinical trial, the sponsor must submit to the FDA a clinical plan, or protocol, accompanied by the approval of the committee responsible for overseeing clinical trials at the clinical trial sites. The FDA or the IRB at each institution at which a clinical trial is being performed may order the temporary or permanent discontinuation of a clinical trial at any time if it believes that the clinical trial is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. Data safety monitoring committees, which monitor certain studies to protect the welfare of study patients, may also require that a clinical trial be discontinued or modified.
13
The sponsor must submit to the FDA the results of the pre-clinical and clinical trials, together with, among other things, detailed information on the manufacturing and composition of the product, and proposed labeling, in the form of an NDA, or, in the case of a biologic, a BLA. The applicant must also submit with the NDA or BLA a substantial user fee payment, unless a waiver or reduction applies. In some cases, a sponsor may be able to expand the indications in an approved NDA or BLA through a submission of a Prior Approval Supplement. Each NDA or BLA submitted for FDA approval is usually reviewed for administrative completeness and reviewability within 60 days following submission of the application. If deemed complete, the FDA will “file” the NDA or BLA, thereby triggering substantive review of the application. The FDA can refuse to file any NDA or BLA that it deems incomplete or not properly reviewable. Once the submission has been accepted for filing, the FDA will review the application and will usually respond to the applicant in accordance with performance goals the FDA has established for the review of NDAs and BLAs - six months from the receipt of the application for priority applications and ten to twelve months for regular applications. The review process is often significantly extended by FDA requests for additional information, pre-clinical studies or clinical trials, clarification, or a risk evaluation and mitigation strategy (“REMS”) or by changes to the application submitted by the applicant in the form of amendments. The FDA may refer applications for novel product candidates which present difficult questions of safety or efficacy to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation, and a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Before approving an NDA or BLA, the FDA will often inspect the facilities at which the product is manufactured and will not approve the product unless the manufacturing facilities are in compliance with cGMP requirements which govern the manufacture, holding and distribution of a product. Manufacturers of human cellular or tissue-based biologics also must comply with the FDA’s Good Tissue Practices, as applicable, and the general biological product standards.
It is possible that our product candidates will not successfully proceed through this approval process or that the FDA will not approve them in any specific period of time, or at all. The FDA may deny or delay approval of applications that do not meet applicable regulatory criteria, or if the FDA determines that the clinical data does not adequately establish the safety and efficacy of the product. Satisfaction of FDA pre-market approval requirements for a new biologic is a process that may take a number of years and the actual time required may vary substantially based upon the type, complexity and novelty of the product or disease. The FDA reviews these applications and, when and if it decides that adequate data is available to show that the product is both safe and effective and that other applicable requirements have been met, approves the drug or biologic for marketing. Government regulation may delay or prevent marketing of potential products for a considerable period of time and imposes costly procedures upon our activities. Success in early stage clinical trials does not assure success in later stage clinical trials. Data obtained from clinical activities is not always conclusive and may be susceptible to varying interpretations that could delay, limit or prevent regulatory approval. Upon approval, a product candidate may be marketed only for those indications approved in the NDA or BLA and will be subject to labeling and promotional requirements or limitations, including warnings, precautions, contraindications and use limitations, which could materially impact profitability. Once approved, the FDA may withdraw the product approval if compliance with pre- and post-market regulatory standards and requirements are not maintained or if safety, efficacy or other problems occur after the product reaches the marketplace.
The FDA may, during its review of an NDA or BLA, ask for additional study data. If the FDA does ultimately approve the product, approval may be subject to limitations based on the FDA's interpretation of the existing pre-clinical and clinical data and the FDA may require post-marketing testing, including potentially expensive Phase IV studies, to confirm or otherwise further evaluate the safety and effectiveness of the product. The FDA also may require, as a condition to approval or continued marketing of a drug, a REMS to ensure that the benefits of a drug or biologic product outweigh its risks. REMS can include additional educational materials for healthcare professionals and patients such as Medication Guides and Patient Package Inserts, a plan for communicating information to healthcare professionals, and restricted distribution of the product. In addition, the FDA may, in some circumstances, impose restrictions on the use of the product, which may be difficult and expensive to administer and may require prior approval of promotional materials. Following approval, the FDA may require labeling changes or impose new post-approval study, risk management, or distribution restriction requirements.
The FDA has developed four distinct approaches intended to make drugs that address unmet medical needs for serious or life threatening conditions available as rapidly as possible, especially when the drugs are the first available treatment or have advantages over existing treatments: accelerated approval, fast track, breakthrough therapy, and priority review.
• | Accelerated Approval. The FDA may grant “accelerated approval” status to drugs or biologics that treat serious or life-threatening illnesses and that provide meaningful therapeutic benefits to patients over existing treatments. Under this pathway, the FDA may approve a product based on surrogate endpoints, or clinical endpoints other than survival or irreversible morbidity. When approval is based on surrogate endpoints or clinical endpoints other than survival or morbidity, the sponsor will be required to conduct additional post-approval clinical trials to verify and describe clinical benefit. Under the agency's accelerated approval regulations, if the FDA concludes that a |
14
product that has been shown to be effective can be safely used only if distribution or use is restricted, it may require certain post-marketing restrictions as necessary to assure safe use. In addition, for products approved under accelerated approval, sponsors will be required to submit all copies of their promotional materials, including advertisements, to the FDA at least thirty days prior to initial dissemination unless otherwise informed by the FDA. After a hearing, the FDA may withdraw a previously granted accelerated approval if, for instance, post-marketing studies fail to verify any clinical benefit, it becomes clear that restrictions on the distribution of the product are inadequate to ensure its safe use, or if a sponsor fails to comply with the conditions of the accelerated approval.
• | Breakthrough Therapy. The FDA may grant “breakthrough therapy” status to drugs or biologics designed to treat, alone or in combination with another drug(s) or biologic(s), a serious or life-threatening disease or condition and for which preliminary evidence suggests a substantial improvement on clinically-meaningful endpoints over existing therapies. Such products need not address an unmet need, but are nevertheless eligible for expedited review if they offer the potential for an improvement over existing therapies. Breakthrough therapy status entitles the sponsor to earlier and more frequent meetings with the FDA regarding the development of nonclinical and clinical data and permits the FDA to offer product development or regulatory advice for the purpose of shortening the potential time to product approval. Breakthrough therapy status does not guarantee that a product will be developed or reviewed more quickly and does not ensure FDA approval. |
• | Fast Track. The FDA may grant “fast track” status to drugs or biologics that treat serious diseases or illness and fill an unmet medical need. Fast track is a process designed to expedite the review of such products by providing, among other things, more frequent meetings with the FDA to discuss the product's development plan, more frequent written correspondence from the FDA about trial design, eligibility for accelerated approval, and rolling review, which allows submission of individually completed sections of a NDA or BLA for the FDA's review before the entire filing is completed. Fast track status does not ensure that a product will be developed more quickly or receive FDA approval more quickly, if at all. |
• | Priority Review. The FDA may grant “priority review” status to products that, if approved, would be significant improvements in safety or effectiveness of the treatment, diagnosis or prevention of serious conditions. Priority review is intended to reduce the time it takes for the FDA to review a NDA or BLA. |
Additionally, there are various designations available to drugs and biologics which provide a sponsor with incentives to support approval of the product candidate, including, but is not limited to, orphan drug designation and rare pediatric disease designation.
Orphan Drug Designation
Under the U.S. Orphan Drug Act, the FDA may grant orphan drug designation to drugs or biologics intended to treat a "rare disease or condition," which is defined as having a prevalence of less than 200,000 individuals in the U.S. Orphan drug designation must be requested before submitting a NDA or BLA for the product. Orphan drug designation does not shorten the regulatory review and approval process, nor does it provide any advantage in the regulatory review and approval process. However, if an orphan drug later receives approval for the indication for which it has designation, the relevant regulatory authority may not approve any other applications to market the same drug for the same indication, except in very limited circumstances, for seven years in the U.S. Although obtaining approval to market a product with orphan drug exclusivity may be advantageous, we cannot be certain:
• | that we will be the first to obtain approval for any drug for which we obtain orphan drug designation; |
• | that orphan drug designation will result in any commercial advantage or reduce competition; or |
• | that the limited exceptions to this exclusivity will not be invoked by the relevant regulatory authority. |
Additionally, orphan drug exclusive marketing rights may be lost under certain conditions, such as if the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug.
FCX-007 has received orphan drug designation from the FDA and we plan to seek orphan drug designation for FCX-013.
15
Rare Pediatric Disease Designation
FCX-007 has received rare pediatric disease designation from the FDA for the treatment of RDEB. The FDA defines a "rare pediatric disease" as a disease that affects fewer than 200,000 individuals in the U.S. primarily under the age of 18 years old. Under the FDA's Rare Pediatric Disease Priority Review Voucher program, upon the approval of a NDA or BLA of a product for the treatment of a rare pediatric disease, the sponsor of such application is eligible for a Rare Pediatric Disease Priority Review Voucher. Currently, the Priority Review Voucher can be used to obtain priority review for any subsequent NDA or BLA and may be sold or transferred an unlimited number of times. Congress has extended the Priority Review Voucher Program until September 30, 2016. Because this program has been subject to criticism, including by the FDA, it is possible that even if we obtain approval for FCX-007 and qualify for a Priority Review Voucher, the program may no longer be in effect at the time of FCX-007's approval.
Ongoing FDA Requirements and Post-Marketing Obligations
The Food and Drug Administration Amendments Act of 2007 expanded FDA authority over drug products after approval. All approved drug products are subject to continuing regulation by the FDA, including record-keeping requirements, reporting of adverse experiences with the product, sampling and distribution requirements, notifying the FDA and gaining its approval of certain manufacturing or labeling changes, complying with certain electronic records and signature requirements, submitting periodic reports to the FDA, maintaining and providing updated safety and efficacy information to the FDA, and complying with FDA promotion and advertising requirements. Failure to comply with the statutory and regulatory requirements can subject a manufacturer to possible legal or regulatory action, such as warning letters, suspension of manufacturing, seizure of product, injunctive action, criminal prosecution, or civil penalties.
The FDA may require post-marketing studies or clinical trials to develop additional information regarding the safety of a product. These studies or trials may involve continued testing of a product and development of data, including clinical data, about the product’s effects in various populations and any side effects associated with long-term use. The FDA may require post-marketing studies or trials to investigate possible or known serious risks or signals of serious risks, or to identify unexpected serious risks, and may require periodic status reports if new safety information develops. Failure to conduct these studies in a timely manner may result in substantial civil fines, or withdrawal of product approval.
A condition to the FDA's approval of LAVIV was that we conduct a 2,700 patient post-marketing study by the end of 2016 to assess the risk of skin cancer (such as basal cell cancer) in the area of LAVIV injections and the risk of immune-mediated hypersensitivity reactions (such as leukocytoclastic vasculitis). We have initiated enrollment in this study and have submitted the required biannual interim reports to the FDA, with the most recent being in January 2016. However, given the limited use of LAVIV, we have experienced difficulties in recruiting a sufficient number of patients for this study. We are actively engaged in discussions with the FDA about how to fulfill the study size requirement in light of the limited population of LAVIV users.
Also, newly discovered or developed safety or efficacy data may require changes to a product’s approved labeling, including the addition of new warnings and contraindications, additional pre-clinical studies or clinical trials, or even in some instances, withdrawal of the approval. Violations of regulatory requirements at any stage, including after approval, may result in various adverse consequences, including the FDA’s withdrawal of an approved product from the market, other voluntary or FDA-initiated action that could delay or restrict further marketing, and the imposition of civil fines and criminal penalties against the manufacturer and NDA or BLA holder. In addition, later discovery of previously unknown problems may result in restrictions on the product, manufacturer or NDA or BLA holder, including withdrawal of the product from the market.
The labeling, advertising, promotion, marketing and distribution of a drug or biologic product also must be in compliance with FDA and Federal Trade Commission (“FTC”) requirements which include, among others, promotional activities, standards and regulations for direct-to-consumer advertising, promotional activities involving the internet, and industry sponsored scientific and educational activities. In general, all product promotion must be consistent with the labeling approved by the FDA for such product, contain a balanced presentation of information on the product’s uses, benefits, risks, and important safety information and limitations on use, and otherwise not be false or misleading. The FDA, as well as the FTC, have very broad enforcement authority, and failure to abide by these regulations can result in penalties, including the issuance of a warning letter directing a company to correct deviations from regulatory standards and enforcement actions that can include seizures, injunctions and criminal prosecution. Failure to comply with applicable FDA requirements and restrictions also may subject a company to adverse publicity and enforcement action by the FDA, the U.S. Department of Justice (“DOJ”) or the Office of the Inspector General of the U.S. Department of Health and Human Services (“HHS”) as well as state authorities. This could subject the company to a range of penalties that could have a significant commercial impact, including
16
civil and criminal fines and agreements that materially restrict the manner in which a company promotes or distributes its products.
Drug and biologic manufacturers and their subcontractors are required to register their establishments with the FDA and certain state agencies, and to list their products with the FDA. The FDA periodically inspects manufacturing facilities in the United States and abroad in order to assure compliance with the applicable cGMP regulations and other requirements. Facilities also are subject to inspections by other federal, foreign, state or local agencies. In complying with the cGMP regulations, manufacturers must continue to assure that the product meets applicable specifications, regulations and other post-marketing requirements. Failure to comply with these requirements subjects the manufacturer to possible legal or regulatory action, such as suspension of manufacturing or recall or seizure of product.
Sponsors and their third-party contractors are also subject to various laws and regulations governing laboratory practices, the experimental use of animals and the use and disposal of hazardous or potentially hazardous substances in connection with their research. In each of the above areas, the FDA has broad regulatory and enforcement powers, including the ability to levy fines and civil penalties, suspend or delay issuance of approvals, seize or recall products and deny or withdraw approvals.
Furthermore, new government requirements may be established that could delay or prevent regulatory approval of our products under development, or affect the conditions under which approved products are marketed.
HIPAA Requirements
Other federal legislation may affect our ability to obtain certain health information in conjunction with our research activities. We may be subject to data privacy and security regulation by both the federal government and the states in which we conduct our business. The Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”), and its implementing regulations, imposes requirements relating to the privacy, security and transmission of individually identifiable health information. Among other things, HITECH makes HIPAA’s privacy and security standards directly applicable to “business associates”— independent contractors or agents of covered entities that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions. In addition, state laws govern the privacy and security of health information in specified circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
Other U.S. Regulatory Requirements
In the United States, the research, manufacturing, distribution, sale, and promotion of drug and biologic products are potentially subject to regulation by various federal, state and local authorities in addition to the FDA, including the Centers for Medicare and Medicaid Services (formerly the Health Care Financing Administration), other divisions of the HHS (e.g., the Office of Inspector General), the DOJ and individual U.S. Attorney offices within the DOJ, and state and local governments. For example, sales, marketing and scientific/educational grant programs must comply with the anti-fraud and abuse provisions of the Social Security Act, the False Claims Act, and similar state laws, each as amended.
If a drug or biologic product is reimbursed by Medicare or Medicaid, pricing and rebate programs must comply with, as applicable, the Medicare Modernization Act as well as the Medicaid rebate requirements of the Omnibus Budget Reconciliation Act of 1990 (“OBRA”) and the Veterans Health Care Act of 1992 (“VHCA”), each as amended. Among other things, the OBRA requires pharmaceutical manufacturers to pay rebates on prescription products to state Medicaid programs and empowers states to negotiate rebates on pharmaceutical prices, which may result in prices for our future products that will likely be lower than the prices we might otherwise obtain. If products are made available to authorized users of the Federal Supply Schedule of the General Services Administration, additional laws and requirements apply. Under the VHCA, drug companies are required to offer some products at a reduced price to a number of federal agencies including the U.S. Department of Veterans Affairs and the U.S. Department of Defense, the Public Health Service and some private Public Health Service designated entities in order to participate in other federal funding programs including Medicaid. Participation under the VHCA requires submission of pricing data and calculation of discounts and rebates pursuant to complex statutory formulas, as well as the entry into government procurement contracts governed by the Federal Acquisition Regulation. All of these activities are also potentially subject to federal and state consumer protection, unfair competition, and other laws.
17
In March 2010, President Obama signed one of the most significant healthcare reform measures in decades. The Affordable Care Act, substantially changes the way healthcare will be financed by both governmental and private insurers, and significantly impacts the pharmaceutical industry. The Affordable Care Act was a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for health care and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. The Affordable Care Act has resulted in, and we expect it will continue to result in, downward pressure on coverage and the price of products covered by Medicare and other government programs. Any reduction in reimbursement from Medicare and other government programs may result in a similar reduction in payments and coverage from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize our products. In addition, it is possible that there will be further legislation or regulation that could harm our business, financial condition and results of operations.
The federal Anti-Kickback Statute prohibits, among other things, knowingly and willfully offering, paying, soliciting or receiving remuneration to induce or in return for purchasing, leasing, ordering or arranging for the purchase, lease or order of any healthcare item or service reimbursable under Medicare, Medicaid or other federally financed healthcare programs. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on one hand and prescribers, purchasers, and formulary managers on the other. Although there are a number of statutory exemptions and regulatory safe harbors protecting some business arrangements from prosecution, the exemptions and safe harbors are drawn narrowly and practices that involve remuneration intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not qualify for an exemption or safe harbor. Our practices may not in all cases meet all of the criteria for safe harbor protection from federal Anti-Kickback Statute liability. The reach of the Anti-Kickback Statute was broadened by the Affordable Care Act, which, among other things, amends the intent requirement of the federal Anti-Kickback Statute. Pursuant to the statutory amendment, a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation. In addition, the Affordable Care Act provides that the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act (discussed below) or the civil monetary penalties statute, which imposes penalties against any person who is determined to have presented or caused to be presented a claim to a federal health program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent.
The federal False Claims Act prohibits any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government or knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government. As a result of a modification made by the Fraud Enforcement and Recovery Act of 2009, a claim includes “any request or demand” for money or property presented to the U.S. government. Recently, several pharmaceutical and other healthcare companies have been prosecuted under these laws for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. Other companies have been prosecuted for causing false claims to be submitted because of the companies’ marketing of the product for unapproved, and thus non-reimbursable, uses. HIPAA created new federal criminal statutes that prohibit knowingly and willfully executing a scheme to defraud any healthcare benefit program, including private third-party payors and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. Also, many states have similar fraud and abuse statutes or regulations that apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
International Regulation
The regulation of our product candidates outside of the United States varies by country. Certain countries regulate human tissue products as a pharmaceutical product, which would require us to make extensive filings and obtain regulatory approvals before selling our product candidates. Certain other countries classify our product candidates as human tissue for transplantation but may restrict its import or sale. Other countries may have no application regulations regarding the import or sale of products similar to our product candidates, creating uncertainty as to what standards we may be required to meet.
Employees
As of December 31, 2015, we had 52 full-time employees, all located in the United States. Of these full-time employees, 39 are engaged in research, development and manufacturing (including facilities) functions and 13 are engaged in finance, legal, human resources, information technology, and other general administrative functions. None of our employees are covered by a collective bargaining agreement, and we consider our relations with our employees to be good.
18
Corporate Information
We were incorporated under the laws of the State of Delaware in September 1992. Our corporate office is located at 405 Eagleview Boulevard, Exton, Pennsylvania 19341. Our telephone number is (484) 713-6000. We maintain an Internet website at www.fibrocell.com. The information contained on our website is not incorporated by reference into this Form 10-K.
We file reports, proxy and information statements and other information with the SEC. We make available free of charge under the “Investors—SEC Filings” section of our website all of our filings with the SEC, including our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy statements and amendments to such documents, each of which is provided on our website as soon as reasonably practicable after we electronically file the information with the SEC.
The public may read and copy any materials we file with the SEC at the SEC's Public Reference Room at 100 F Street, NE, Washington, DC 20549. The public may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. Additionally, the SEC maintains a website (www.sec.gov) that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC, including us.
Item 1A. Risk Factors
Our business is subject to substantial risks and uncertainties. The occurrence of any of the following risks and uncertainties, either alone or taken together, could materially and adversely affect our business, financial condition, results of operations or prospects. In these circumstances, the market price of our common stock could decline and you may lose all or part of your investment. The risks and uncertainties described below are not the only ones we face. Risks and uncertainties of general applicability and additional risks and uncertainties not currently known to us or that we currently deem to be immaterial may also materially and adversely affect our business, financial condition, results of operations or prospects.
Risks Related to our Financial Position and Need for Additional Capital
We need to obtain additional capital to continue as a going concern. If we are unable to obtain sufficient capital, we will need to curtail and reduce our operations and costs, and modify our business strategy.
Our principal sources of liquidity are cash and cash equivalents of $29.3 million as of December 31, 2015. As of December 31, 2015, we had working capital of $15.6 million. We believe that our existing cash and cash equivalents will be sufficient to fund our operations into the fourth quarter of 2016. However, changing circumstances may cause us to consume capital faster than we currently anticipate, and we may need to spend more money than currently expected because of such circumstances.
To meet our capital needs, we are considering multiple alternatives, including but not limited to, equity financings, debt financings, corporate collaborations, partnerships and other strategic transactions and funding opportunities. However, there can be no assurance that we will be able to complete any such transaction on acceptable terms or otherwise. These factors raise substantial doubt about our ability to continue as a going concern. As a result, our independent registered public accounting firm has included an explanatory paragraph in its report on our financial statements for the year ended December 31, 2015 related to our ability to continue as a going concern.
If we raise additional funds by issuing equity securities, our stockholders will experience dilution. Debt financing, if available, will result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. Any debt financing or additional equity that we raise may contain terms, such as liquidation and other preferences, which are not favorable to us or our stockholders. If we raise additional funds through collaboration or partnership arrangements with third parties, it may be necessary to relinquish valuable rights to our technologies, future revenue streams or product candidates or to grant licenses on terms that may not be favorable to us.
If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we will need to curtail and reduce our operations and costs, and modify our business strategy which may require us to, among other things:
• | significantly delay, scale back or discontinue the development or commercialization of one or more of our product candidates or one or more of our other research and development initiatives; |
• | seek collaborators for one or more of our current or future product candidates at an earlier stage than otherwise would be desirable or on terms that are less favorable than might otherwise be available; |
19
• | sell or license on unfavorable terms our rights to technologies or product candidates that we otherwise would seek to develop or commercialize ourselves; or |
• | seek bankruptcy protection which may result in the termination of agreements pursuant to which we license important intellectual property rights including our exclusive collaboration agreements with Intrexon. |
We have incurred significant losses since our inception and anticipate that we will continue to incur losses in the future.
We have incurred losses since our inception, have not generated significant revenue from commercial sales of our products, and have never been profitable. Since 2013, which is when we changed our business strategy to focus on therapeutic indications for azficel-T and on diseases affecting the skin and connective tissue in collaboration with our partner Intrexon, we have reduced sales and marketing efforts of LAVIV. We fulfilled a nominal amount of prescriptions for LAVIV aesthetic procedures in 2014 and 2015 and will continue to do so in 2016. Investment in drug development is highly speculative because it entails substantial upfront capital expenditures and significant risk that a product candidate will fail to gain regulatory approval or become commercially viable. We continue to incur significant research, development and other expenses related to our ongoing operations including development of our product candidates and operation of our manufacturing facility. As a result, we are not profitable and have incurred losses in each period since we emerged from bankruptcy in September 2009. For the year ended December 31, 2015, we reported a net loss of $34.5 million, and we had an accumulated deficit of $147.2 million at December 31, 2015.
We expect to continue to incur significant expenses and increasing operating losses for the foreseeable future. We anticipate that our expenses will continue to be significant if and as we:
• | continue our research and pre-clinical and clinical development of our product candidates; |
• | initiate additional pre-clinical, clinical or other studies or trials for our product candidates, including under our collaboration agreements with Intrexon; |
• | continue or expand our collaborations with Intrexon and our other collaborators; |
• | further develop the manufacturing process for our product candidates; |
• | continue to maintain a cGMP manufacturing facility; |
• | change or add additional manufacturers or suppliers; |
• | seek regulatory approvals for our product candidates that successfully complete clinical trials; |
• | establish a sales, marketing and distribution infrastructure to commercialize any products for which we may obtain regulatory approval; |
• | seek to identify and validate additional product candidates; |
• | acquire or in-license other product candidates and technologies; |
• | maintain, protect and expand our intellectual property portfolio; |
• | attract and retain skilled personnel; |
• | create additional infrastructure to support our product development and planned future commercialization efforts; and |
• | experience any delays or encounter issues with any of the above. |
The net losses we incur may fluctuate significantly from quarter to quarter and year to year, such that a period-to-period comparison of our results of operations may not be a good indication of our future performance. In any particular quarter or quarters, our operating results could be below the expectations of securities analysts or investors, which could cause our stock price to decline.
We do not generate significant revenues from product sales and may never be profitable.
Our ability to generate revenue and achieve profitability depends on our ability, alone or with strategic collaboration partners, to successfully complete the development of, and obtain the regulatory approvals necessary for, the manufacture and commercialization of our product candidates. Other than from the sale of LAVIV, we do not anticipate generating revenues from product sales for the foreseeable future, if ever. Our ability to generate future revenues from product sales depends heavily on our success in:
20
• | completing research and pre-clinical and clinical development of our product candidates; |
• | seeking and obtaining regulatory approvals for product candidates for which we complete clinical trials; |
• | developing a sustainable, scalable, reproducible, and transferable manufacturing process for our product candidates; |
• | establishing and maintaining supply and manufacturing relationships with third parties that can provide adequate (in amount and quality) products and services to support clinical development and the market demand for our product candidates, if approved; |
• | launching and commercializing product candidates for which we obtain regulatory approval, either by collaborating with a partner or, if launched independently, by establishing a sales force, marketing, sales operations and distribution infrastructure; |
• | obtaining market acceptance of our product candidates and cell therapy and gene therapy as viable treatment options; |
• | addressing any competing technological and market developments; |
• | implementing additional internal systems and infrastructure, as needed; |
• | identifying and validating new product candidates; |
• | negotiating favorable terms in any collaboration, licensing or other arrangements into which we may enter; |
• | maintaining, protecting and expanding our portfolio of intellectual property rights, including patents, trade secrets and know-how; and |
• | attracting, hiring and retaining qualified personnel. |
Even if one or more of the product candidates that we develop is approved for commercial sale, we anticipate incurring significant costs associated with commercializing any approved product candidate. Our expenses could increase beyond expectations if we are required by the FDA or other regulatory agencies, domestic or foreign, to perform clinical trials or other studies in addition to those that we currently anticipate. Even if we are able to generate revenues from the sale of any approved products, we may not become profitable and may need to obtain additional funding to continue operations.
We will seek to raise additional funds in the future, which may be dilutive to stockholders or impose operational restrictions.
We will need to raise additional capital in the future to help fund our clinical trials, our collaboration efforts with Intrexon and for the development and commercialization of our product candidates. If we raise additional capital through the issuance of equity securities, such as through our "at-the-market" equity program with Cantor Fitzgerald & Co., the percentage ownership of our current stockholders will be reduced. We may also issue equity as part of license issue fees to our licensors, compensate consultants or settle outstanding payables. Our stockholders may experience additional dilution in net book value per share and any additional equity securities may have rights, preferences and privileges senior to those of the holders of our common stock. Debt financing, if available, will result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. Any debt financing or additional equity that we raise may contain terms, such as liquidation and other preferences, which are not favorable to us or our stockholders. If we raise additional funds through corporate collaboration, partnership or other strategic transactions, it may be necessary to relinquish valuable rights to our product candidates, our technologies or future revenue streams or to grant licenses or sell assets on terms that may not be favorable to us. If we cannot raise additional funds, we will have to delay our development activities or cease operations.
We have a limited operating history, which may make it difficult for you to evaluate the success of our business to date and to assess our future viability.
We have a limited operating history and our primary business activities consist of pre-clinical development and conducting clinical trials, pursuing our collaborations with Intrexon and commercializing LAVIV. As such, our historical financial data is of limited value in estimating future operating expenses. Our budgeted expense levels are based in part on our expectations concerning the costs of our pre-clinical development, clinical trials and our collaborations with Intrexon, which depend on the success of such activities, and our ability to effectively and efficiently conduct such trials, pre-clinical development and our expectations related to our efforts to achieve FDA approval with respect to our product candidates. Our limited operating history and clinical trial experience make these costs difficult to forecast accurately. We may be unable to adjust our operations in a timely manner to compensate for any unexpected increase in costs. Further, our fixed manufacturing
21
costs and operating expenses may increase significantly as we expand our operations. Accordingly, a significant increase in costs could have an immediate and material adverse effect on our business, results of operations and financial condition.
We may acquire other assets or businesses, or form collaborations or make investments in other companies or technologies that could harm our operating results, dilute our stockholders’ ownership, incur debt or cause us to incur significant expense.
As part of our business strategy, we may pursue acquisitions of assets or businesses, or strategic alliances and collaborations, to expand our existing technologies and operations. We may not identify or complete these transactions in a timely manner, on a cost-effective basis, or at all, and we may not realize the anticipated benefits of any such transaction, any of which could have a detrimental effect on our financial condition, results of operations and cash flows. We may not be able to find suitable acquisition candidates, and if we make any acquisitions, we may not be able to integrate these acquisitions successfully into our existing business and we may incur debt or assume unknown or contingent liabilities in connection therewith. Integration of an acquired company or assets may also disrupt ongoing operations, require the hiring of additional personnel and the implementation of additional internal systems and infrastructure, especially the acquisition of commercial assets, and require management resources that would otherwise focus on developing our existing business. We may not be able to find suitable collaboration partners or identify other investment opportunities, and we may experience losses related to any such investments.
To finance any acquisitions or collaborations, we may choose to issue debt or shares of our common stock as consideration. Any such issuance of shares would dilute the ownership of our stockholders. If the price of our common stock is low or volatile, we may not be able to acquire other assets or companies or fund a transaction using our stock as consideration. Alternatively, it may be necessary for us to raise additional capital for acquisitions through public or private financings. Additional capital may not be available on terms that are favorable to us, or at all.
Risks Related to Clinical Development, Regulatory Approval and Commercialization of Our Product Candidates
Our gene-therapy product candidates are based on novel technology, which makes it difficult to predict the time and cost of product development and subsequently obtaining regulatory approval. At the moment, no gene therapy products have been approved in the United States and only one product has been approved in the European Union.
Our gene-therapy product candidates, including FCX-007 and FCX-013, are based on novel technology. Our future success depends on the successful development of this therapeutic approach. There can be no assurance that any development problems we experience in the future related to our gene-therapy product candidates will not cause significant delays or unanticipated costs, or that such development problems can be solved. We may also experience delays in developing a sustainable, reproducible and commercial-scale manufacturing process or transferring that process to commercial partners, which may prevent us from completing our clinical studies or commercializing our gene-therapy products on a timely or profitable basis, if at all.
In addition, the clinical trial requirements of the FDA, the European Medicines Agency, or EMA, and other regulatory agencies and the criteria these regulators use to determine the safety and efficacy of a product candidate vary substantially according to the type, complexity, novelty and intended use and market of the potential products. The regulatory approval process for gene-therapy product candidates such as ours can be more expensive and take longer than for other, better known or more extensively studied pharmaceutical or other product candidates. At the moment, only one gene-therapy product, UniQure’s Glybera, which received marketing authorization in the European Union, or EU, in 2012, has been approved in the Western world, which makes it difficult to determine how long it will take or how much it will cost to obtain regulatory approvals for our gene-therapy product candidates in the United States, the EU or other jurisdictions. Approvals by the EMA and the European Commission may not be indicative of what the FDA may require for approval.
Regulatory requirements governing gene and cell therapy products have evolved and may continue to change in the future. For example, the FDA has established the Office of Cellular, Tissue and Gene Therapies within its Center for Biologics Evaluation and Research, or CBER, to consolidate the review of gene therapy and related products, and the Cellular, Tissue and Gene Therapies Advisory Committee to advise CBER on its review. Gene therapy clinical trials conducted at institutions that receive funding for recombinant DNA research from the U.S. National Institutes of Health, or NIH, are also subject to review by the NIH Office of Biotechnology Activities’ Recombinant DNA Advisory Committee, or RAC. Although the FDA decides whether individual gene therapy protocols may proceed, the RAC review process can impede the initiation of a clinical study, even if the FDA has reviewed the study and approved its initiation. Clinical trial sites in the United States that receive NIH funding for research involving recombinant or synthetic nucleic acid molecules are required to follow RAC recommendations, or risk losing NIH funding for such research or needing NIH pre-approval before conducting such research. In addition, the
22
FDA can put an investigational new drug application, or IND, on clinical hold if the information in an IND is not sufficient to assess the risks in pediatric patients. Before a clinical study can begin at any institution, that institution’s institutional review board, or IRB, and its Institutional Biosafety Committee will have to review the proposed clinical study to assess the safety of the trial. Moreover, serious adverse events or developments in clinical trials of gene therapy product candidates conducted by others may cause the FDA or other regulatory bodies to initiate a clinical hold on our clinical trials or otherwise change the requirements for approval of any of our product candidates.
These regulatory review agencies, committees and advisory groups and the new requirements and guidelines they promulgate may lengthen the regulatory review process, require us to perform additional or larger studies, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of these treatment candidates or lead to significant post-approval studies, limitations or restrictions. As we advance our product candidates, we will be required to consult with these regulatory and advisory groups and comply with applicable requirements and guidelines. If we fail to do so, we may be required to delay or discontinue development of our product candidates. Delay or failure to obtain, or unexpected costs in obtaining, the regulatory approval necessary to bring a potential product to market could decrease our ability to generate sufficient product revenue to maintain our business.
The results seen in pre-clinical studies of our product candidates may not be replicated in humans.
Although we have seen positive results in pre-clinical studies of FCX-007 and FCX-013, we may not see positive
results when these and any other product candidates undergo future clinical trials in humans. Pre-clinical studies are not
designed to test the efficacy of a product candidate in humans, but rather to:
• | test short-term safety; |
• | establish biological plausibility; |
• | identify biologically active dose levels; |
• | establish feasibility and reasonable safety of the investigational product's proposed clinical route of administration; |
• | identify physiologic parameters that can guide clinical monitoring; and/or |
• | establish proof of concept, or the feasibility and rationale for use of an investigational product in the targeted patient population. |
Success in pre-clinical studies does not ensure that later studies or any clinical trials will be successful nor does it predict future results. The rate of failure in drug development is quite high, and many companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in pre-clinical studies and earlier clinical trials. Product candidates may fail to show desired safety and efficacy when used with human patients. Negative or inconclusive results from any of our ongoing pre-clinical studies could result in delays, modifications, or abandonment of clinical trials and the termination of our development of a product candidate.
In September 2015, we received feedback from the FDA relating to our IND for FCX-007. If we are unable to adequately address the FDA’s feedback, we will not be permitted to commence clinical trials of FCX-007, the development of FCX-007 will be further delayed and we will incur additional costs.
In September 2015, we received feedback from the FDA relating to our IND application for FCX-007, which required us to delay the initiation of our proposed Phase I/II clinical trial. The FDA’s feedback related to the areas of CMC, toxicology and our proposed Phase I/II clinical trial protocol. Although the prior hybrid pharmacology/toxicology study performed based on the injection of FCX-007 into human skin that was xenografted onto SCID (severe combined immunodeficiency) mice was included in the IND and showed no signs of toxicity, the FDA requested that we execute an additional toxicology-specific study in which FCX-007 will be injected in non-grafted SCID mice. We have initiated this new toxicology study, and we expect to amend the IND in response to the FDA’s feedback and to include data from the new study in the first quarter of 2016. If we are unable to successfully complete the additional toxicology study and adequately address the FDA’s other feedback, we will not be permitted to commence clinical trials, the development of FCX-007 will be further delayed and we will be required to incur additional costs in the development of FCX-007.
In previous clinical trials involving viral vectors for gene therapy, some patients experienced serious adverse events, including the development of leukemia due to vector-related insertional oncogenesis. If our vectors demonstrate a similar effect, we may be required to halt or delay further clinical development of our product candidates.
23
A significant risk in any gene therapy product based on viral vectors is that the vector will insert in or near cancer-causing oncogenes leading to uncontrolled clonal proliferation of mature cancer cells in the patient. For example, in 2003, 20 patients treated for X-linked severe combined immunodeficiency in two gene therapy trials using a murine, or mouse-derived, gamma-retroviral vector showed correction of the disease, but the trials were terminated after five patients developed leukemia (four of whom were subsequently cured). The cause of these adverse events was shown to be insertional oncogenesis, which is the process whereby the corrected gene inserts in or near a gene that is important in a critical cellular process like growth or division, and this insertion results in the development of a cancer (often leukemia). Using molecular diagnostic techniques, it was determined that clones from these patients showed retrovirus insertion in proximity to the promoter of the LMO2 proto-oncogene. Earlier generation retroviruses like the one used in these two trials have been shown to preferentially integrate in regulatory regions of genes that control cell growth.
These well-publicized adverse events led to the development of new viral vectors, such as lentiviral vectors like the ones we utilize for FCX-007 and FCX-013, with potentially improved safety profiles and also the requirement of enhanced safety monitoring in gene therapy clinical trials, including periodic analyses of the therapy’s genetic insertion sites. Notwithstanding the potential safety improvements of lentiviral vectors, the risk of insertional oncogenesis remains a significant concern for gene therapy and we cannot assure that it will not occur in any of our clinical trials. There is also the potential risk of delayed adverse events following exposure to gene therapy products due to persistent biological activity of the genetic material or other components of products used to carry the genetic material. The FDA has stated that lentiviral vectors possess characteristics that may pose risks of delayed adverse events. If any such adverse events occur, further advancement of our clinical trials could be halted or delayed, which would have a material adverse effect on our business and operations.
We may find it difficult to enroll patients in our clinical trials, which could delay or prevent clinical trials of our product candidates.
Identifying and qualifying patients to participate in clinical trials of our product candidates is critical to our success. The timing of our clinical trials depends on the speed at which we can recruit patients to participate in testing our product candidates. We have experienced delays in some of our clinical trials, and we may experience similar delays in the future. If patients are unwilling to participate in our clinical trials because of negative publicity from adverse events in cell and gene therapies or for other reasons, including competitive clinical trials for similar patient populations, the timeline for recruiting patients, conducting clinical trials and obtaining regulatory approval of potential products may be delayed. These delays could result in increased costs, delays in advancing our product candidates, delays in testing the effectiveness of our technology or termination of the clinical trials altogether.
We may not be able to identify, recruit and enroll a sufficient number of patients, or those with required or desired characteristics to achieve diversity in a clinical trial or complete our clinical trials in a timely manner. Patient enrollment is affected by a variety factors including, among others:
• | severity of the disease under investigation; |
• | design of the study protocol; |
• | prevalence of the disease/size of the patient population; |
• | eligibility criteria for the clinical trial in question; |
• | perceived risks and benefits of the product candidate under study; |
• | proximity and availability of clinical trial sites for prospective patients; |
• | availability of competing therapies and clinical trials; |
• | efforts to facilitate timely enrollment in clinical trials; |
• | patient referral practices of physicians; and |
• | ability to monitor patients adequately during and after treatment. |
Our current product candidates are being developed to treat rare diseases with limited patient pools from which to draw for clinical trials and the process of finding and diagnosing patients may prove costly. We have estimated that there are approximately 1,100 to 2,500 U.S. patients with RDEB and approximately 40,000 U.S. patients with linear scleroderma over a major joint who exhibit severe joint pain. We may not be able to initiate or continue clinical trials if we cannot enroll a sufficient number of eligible patients to participate in the clinical trials required by the FDA or other regulatory agencies. If we have difficulty enrolling a sufficient number of patients to conduct our clinical trials as planned, we may need to delay, limit or terminate ongoing or planned clinical trials, any of which would have an adverse effect on our business.
24
Clinical trials may fail to demonstrate the safety or efficacy of our product candidates, which could prevent or significantly delay regulatory approval of our product candidates and harm our business.
Prior to receiving approval to commercialize any of our product candidates, we must demonstrate with substantial evidence from well-controlled clinical trials, and to the satisfaction of the FDA and other regulatory authorities in the United States and abroad, that our product candidates are both safe and effective. We will need to demonstrate our product candidates’ efficacy and monitor their safety throughout the process. If our current or future clinical trials are unsuccessful, regulatory approval of our product candidates could be delayed or prevented and our business could be harmed.
All of our product candidates are subject to the risks of failure inherent in drug development. The results of early-stage clinical trials of our product candidates do not necessarily predict the results of later-stage clinical trials. Product candidates in later-stage clinical trials may fail to demonstrate desired safety and efficacy traits despite having successfully progressed through initial clinical testing. Even if we believe the data collected from clinical trials of our product candidates is promising, this data may not be sufficient to support approval by the FDA or any other U.S. or foreign regulatory approval. The FDA may also reject any of our completed clinical trials as inadequate to support approval if the study design does not include specific safety monitoring measures. Pre-clinical and clinical data can be interpreted in different ways. Accordingly, FDA officials could reach different conclusions in assessing such data than we do which could delay, limit or prevent regulatory approval. In addition, the FDA, other regulatory authorities, our IRB or we may suspend or terminate clinical trials at any time.
Obtaining FDA and other regulatory approvals is complex, time consuming and expensive, and the outcomes are uncertain.
The process of obtaining FDA and other regulatory approvals is time consuming, expensive and difficult. Clinical trials are required to establish the safety and efficacy of product candidates. Applications to market product candidates must be submitted to the FDA which must be reviewed for approval and approved by the FDA before product candidates may be marketed and clinical trials, manufacturing, and the marketing of products, if approved, are subject to strict regulatory compliance. The commencement and completion of clinical trials for any of our product candidates could be delayed or prevented by a variety of factors, including:
• | delays in obtaining regulatory approvals to commence a study or trial; |
• | delays in identifying and reaching agreement on acceptable terms with prospective clinical trial sites; |
• | delays or failures in obtaining approval of our clinical trial protocol from an IRB to conduct a clinical trial at a prospective study site; |
• | delays in the enrollment of patients; |
• | manufacturing difficulties; |
• | failure of our clinical trials and clinical investigators to be in compliance with the FDA’s GCP; |
• | failure of our third-party contract research organizations, clinical site organizations or other clinical trial managers, to satisfy their contractual duties, comply with regulations or meet expected deadlines; |
• | lack of efficacy during clinical trials; or |
• | unforeseen safety issues. |
We do not know whether our clinical trials will need to be restructured or will be completed on schedule, if at all, or whether they will provide data necessary to support regulatory approval. Significant delays in clinical trials will impede our ability to commercialize our product candidates and generate revenue, and could significantly increase our development costs.
In addition, we utilize bovine-sourced materials to manufacture our product candidates. It is possible that future FDA regulations may require us to change the source of the bovine-sourced materials we use in our products or to cease using bovine-sourced materials. If we are required to use alternative materials in our products, and in the event that such alternative materials are available to us, or if we choose to change the materials used in our products in the future, we would need to validate the new manufacturing process and run comparability trials with the reformulated product, which could delay our submission for regulatory approval of our product candidates and negatively impact the development and potential commercialization of our product candidates.
25
If we fail to obtain the necessary regulatory approvals, or if such approvals are limited, we will not be able to commercialize our product candidates, and we will not generate product revenues.
Even if we comply with all FDA pre-approval regulatory requirements, the FDA may determine that our product candidates are not safe or effective, and we may never obtain regulatory approval for such product candidates. If we fail to obtain regulatory approval for some or all of our product candidates, we will have fewer commercial products, if any, and correspondingly lower product revenues, if any. Even if our product candidates receive regulatory approval, such approval may involve limitations on the indications and conditions of use or marketing claims for our products. Further, later discovery of previously unknown problems or AEs could result in additional regulatory restrictions, including withdrawal of products and addition of warnings or other statements on the product label.
With respect to LAVIV, which was approved in June 2011, as part of approval the FDA required us, based on clinical trial data, to conduct a 2,700 patient post-marketing study evaluating the risk of skin cancer in the injection site. This study is required to be completed by the end of 2016. We have been engaged in discussions with the FDA on the design of the post-marketing study, as we believe the original study design, especially the sample size, is no longer consistent with our current emphasis on azficel-T for vocal cord scarring and our decreased involvement with cosmetic applications. The FDA acknowledged that commercial volume since the marketing of LAVIV is inadequate to meet the original enrollment rate proposed but has required submission of actual enrollment data before considering a revision. We have initiated enrollment in the post-marketing study and have submitted the required biannual interim reports to the FDA containing this enrollment data, with the most recent being in January 2016. We continue to be actively engaged in discussions with the FDA about how to fulfill the original post-marketing study sample size requirement in light of the limited population of LAVIV users. Although we believe we will be able to reach an agreement with the FDA on this post-marketing study, to the extent we are unable to complete an acceptable post-marketing study, the FDA may determine to take action against us, including the withdrawal of its approval of LAVIV.
In jurisdictions outside the United States, we must receive marketing authorizations from the appropriate regulatory authorities before commercializing our product candidates. Regulatory approval processes outside the United States generally include requirements and risks similar to, and in many cases in excess of, the risks associated with FDA approval.
Our failure to comply with extensive governmental regulation may significantly affect our operating results.
Even if we obtain regulatory approval for some or all of our product candidates, we will continue to be subject to extensive ongoing requirements by the FDA, as well as by a number of foreign, national, state and local agencies. These regulations will impact many aspects of our operations, including testing, research and development, manufacturing, safety, efficacy, labeling, storage, quality control, AE reporting, import and export, record keeping, approval, distribution, advertising and promotion of our future products. We must also submit new or supplemental applications and obtain FDA approval for certain changes to an approved product, product labeling or manufacturing process. Application holders must also submit advertising and other promotional material to the FDA and report on ongoing clinical trials. The FDA enforces post-marketing regulatory requirements, including the cGMP requirements, through periodic unannounced inspections. We do not know whether we will pass any future FDA inspections. Failure to pass an inspection could disrupt, delay or shut down our manufacturing operations. Failure to comply with applicable regulatory requirements could result in, among other things:
• | administrative or judicial enforcement actions; |
• | changes to advertising; |
• | failure to obtain regulatory approvals for our product candidates; |
• | revocation or suspension of regulatory approvals of products; |
• | product seizures or recalls; |
• | court-ordered injunctions; |
• | import detentions; |
• | delay, interruption or suspension of product manufacturing, distribution, marketing and sales; or |
• | civil or criminal sanctions. |
The discovery of previously unknown problems with our products may result in restrictions on the products, including withdrawal from the market. In addition, the FDA may revisit and change its prior determinations with regard to the safety or efficacy of our future products. If the FDA’s position changes, we may be required to change our labeling or cease to
26
manufacture and market our future products. Even prior to any formal regulatory action, we could voluntarily decide to cease the distribution and sale or recall any of our future products if concerns about their safety or efficacy develop.
In their regulation of advertising and other promotion, the FDA and the FTC may issue correspondence alleging that some advertising or promotional practices are false, misleading or deceptive. The FDA and FTC are authorized to impose a wide array of sanctions on companies for such advertising and promotion practices, which could result in any of the following:
• | incurring substantial expenses, including fines, penalties, legal fees and costs to comply with the FDA’s requirements; |
• | changes in the methods of marketing and selling products; |
• | taking FDA mandated corrective action, which may include placing advertisements or sending letters to physicians rescinding previous advertisements or promotions; or |
• | disruption in the distribution of products and loss of sales until compliance with the FDA’s position is obtained. |
Improper promotional activities may also lead to investigations by federal or state prosecutors, and result in criminal and civil penalties. If we become subject to any of the above requirements, it could be damaging to our reputation and restrict our ability to sell or market our future products, and our business condition could be adversely affected. We may also incur significant expenses in defending ourselves.
Physicians may prescribe pharmaceutical or biologic products for uses that are not described in a product’s labeling or differ from those tested by us and approved by the FDA. While such “off-label” uses are common and the FDA does not regulate physicians’ choice of treatments, the FDA does restrict a manufacturer’s communications on the subject of off-label use. Companies cannot promote FDA-approved pharmaceutical or biologic products for off-label uses, but under certain limited circumstances they may disseminate to practitioners’ articles published in peer-reviewed journals. To the extent allowed by the FDA, we may disseminate peer-reviewed articles on our future products, if approved, to practitioners. If, however, our activities fail to comply with the FDA’s regulations or guidelines, we may be subject to warnings from, or enforcement action by, the FDA or other regulatory or law enforcement authorities.
Our sales, marketing, and scientific/educational grant programs, if any in the future, must also comply with applicable requirements of the anti-fraud and abuse provisions of the Social Security Act, the False Claims Act, the federal anti-kickback law, and similar state laws, each as amended. Pricing and rebate programs must comply with the Medicaid rebate requirements of the Omnibus Budget Reconciliation Act of 1990 and the Veteran’s Health Care Act of 1992, each as amended. If products are made available to authorized users of the Federal Supply Schedule of the General Services Administration, additional laws and requirements apply. All of these activities are also potentially subject to federal and state consumer protection and unfair competition laws. The distribution of product samples to physicians must comply with the requirements of the Prescription Drug Marketing Act.
Depending on the circumstances, failure to meet post-approval requirements can result in criminal prosecution, fines or other penalties, injunctions, recall or seizure of products, total or partial suspension of production, denial or withdrawal of pre-marketing product approvals, or refusal to allow us to enter into supply contracts, including government contracts. Any government investigation of alleged violations of law could require us to expend significant time and resources in response, and could generate negative publicity.
We are subject to significant regulation with respect to the manufacturing of our products.
Components of a finished therapeutic product approved for commercial sale or used in late-stage clinical trials must be manufactured in accordance with current Good Manufacturing Practices. These regulations govern manufacturing processes and procedures and the implementation and operation of quality systems to control and assure the quality of investigational products and products approved for sale. Our facilities and quality systems and the facilities and quality systems of some or all of our third party contractors and suppliers must pass inspection for compliance with the applicable regulations as a condition of FDA approval of our products. In addition, the FDA may, at any time, audit or inspect a manufacturing facility, including our manufacturing facility, involved with the preparation of LAVIV, or our associated quality systems for compliance with the regulations applicable to the activities being conducted. The FDA also may, at any time following approval of a product for sale, audit our manufacturing facilities or those of our third party contractors. If any such inspection or audit identifies a failure to comply with applicable regulations or if a violation of our product specifications or applicable regulation occurs independent of such an inspection or audit, we or the FDA may require remedial measures that may be costly and/or time consuming for us or a third party to implement and that may include the temporary or permanent suspension of a clinical trial or commercial
27
sales, recalls, market withdrawals, seizures or the temporary or permanent closure of a facility. Any such remedial measures imposed upon us or third parties with whom we contract could materially harm our business.
If we fail to obtain or maintain orphan drug exclusivity for any of our product candidates, our competitors may sell products to treat the same conditions and our operations will be adversely impacted.
As part of our business strategy, we have obtained FDA orphan designation for FCX-007 and intend to seek orphan drug designation for FCX-013. Under the Orphan Drug Act, the FDA may designate a product as an orphan drug if it is intended to treat a rare disease or condition, defined as a patient population of fewer than 200,000 in the U.S. The first company to obtain FDA approval for a designated orphan drug for a given rare disease receives marketing exclusivity for use of that drug for the stated condition for a period of seven years. Orphan drug exclusive marketing rights may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug or access to the biologic.
Because the extent and scope of patent protection for some of our product candidates is limited, orphan drug designation is especially important for our products that are eligible for orphan drug designation. For eligible product candidates, we plan to rely on the exclusivity period under the Orphan Drug Act to maintain a competitive position. If we do not obtain orphan drug exclusivity for our product candidates that do not have broad patent protection, our competitors may then sell the same drug or biologic to treat the same condition which could adversely affect our operations.
Even though we have obtained orphan drug designation for FCX-007 and even if we obtain orphan drug designation for other product candidates, due to the uncertainties associated with developing biopharmaceutical products, we may not be the first to obtain regulatory approval for any particular orphan indication, which means that we may not obtain orphan drug exclusivity and could also potentially be blocked from approval until the first product’s orphan drug exclusivity period expires. Further, even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs can be approved for the same condition. Even after an orphan drug is approved and granted orphan drug exclusivity, the FDA can subsequently approve the same drug for the same condition if the FDA concludes that the later drug is safer, more effective or makes a major contribution to patient care. Orphan drug designation does not shorten the regulatory review and approval process, nor does it provide any additional opportunities for review and guidance from the FDA during the review and approval process.
Even if we were to obtain approval for FCX-007 with the rare pediatric disease designation, the Rare Pediatric Disease Priority Review Voucher Program may no longer be in effect at the time of such approval.
FCX-007 has received rare pediatric disease designation from the FDA for the treatment of RDEB. The FDA defines a "rare pediatric disease" as a disease that affects fewer than 200,000 individuals in the U.S. primarily under the age of 18 years old. Under the FDA's Rare Pediatric Disease Priority Review Voucher program, upon the approval of a NDA or BLA for the treatment of a rare pediatric disease, the sponsor of such application would be eligible for a Rare Pediatric Disease Priority Review Voucher that can be used to obtain priority review for a subsequent NDA or BLA. The Priority Review Voucher may be sold or transferred an unlimited number of times. Congress has extended the Priority Review Voucher Program until September 30, 2016. This program has been subject to criticism, including by the FDA, and it is possible that even if we obtain approval for FCX-007 and qualify for such a Priority Review Voucher, the program may no longer be in effect at the time of approval.
We are largely dependent on the future commercial success of our product candidates.
Our ability to generate revenues and become profitable will depend in large part on the future commercial success of our product candidates. If any product that we commercialize in the future does not gain an adequate level of acceptance among physicians, patients and third parties, we may not generate significant product revenues or become profitable. Market acceptance of our products, and any other products that we commercialize, by physicians, patients and third party payors will depend on a number of factors, some of which are beyond our control, including:
• | The efficacy, safety and other potential advantages in relation to alternative treatments; |
• | The relative convenience and ease of administration; |
• | The availability of adequate coverage or reimbursement by third parties, such as insurance companies and other healthcare payors, and by government healthcare programs, including Medicare and Medicaid; |
• | The prevalence and severity of adverse events; |
• | The cost of treatment in relation to alternative treatments, including generic products; |
28
• | The extent and strength of our third party manufacturer and supplier support; |
• | The extent and strength of marketing and distribution support; |
• | The limitations or warnings contained in a product’s FDA approved labeling; and |
• | Distribution and use restrictions imposed by the FDA or to which we agree as part of a mandatory risk evaluation and mitigation strategy or voluntary risk management plan |
For example, even if our products have been approved by the FDA, physicians and patients may not immediately be receptive to them and may be slow to adopt them. In addition, even though we believe our product candidates have significant advantages to other treatment options, because no head-to-head trials comparing our product candidates to competing products will have been conducted, the prescribing information approved by the FDA would not contain claims that our product is safer or more effective than competitive products. Accordingly, promotion of our products will not reflect any comparative advantages that may exist. If our products do not achieve an adequate level of acceptance among physicians, patients and third party payors, we may not generate meaningful revenues from and we may not become profitable.
In order to commercialize any future product candidates, we will need to increase our manufacturing capacity and improve our manufacturing capabilities, which will require significant expenditures and regulatory approval.
We currently have limited manufacturing capacity. In order to commercialize any future product candidates, we will need to increase our manufacturing capacity. We are developing enhancements and alternatives to our current manufacturing process. If we have difficulties in increasing our manufacturing capacity and improving our capabilities, we will be limited in our ability to manufacture and commercialize our product candidates, if they are approved for marketing; and we may not be able to decrease our manufacturing costs. These difficulties could adversely affect our financial performance and damage our reputation. Even if we are successful in developing such enhancements or finding alternatives to our current process, such manufacturing changes will require additional expenditures, for which we may be required to seek external financing. In addition, our ability to increase our manufacturing capacity or modify our manufacturing processes will be subject to additional FDA review and approval.
Negative public opinion and increased regulatory scrutiny of gene therapies may damage public perception of our product candidates or adversely affect our ability to conduct our business or obtain regulatory approvals for our gene therapy product candidates.
Public perception may be influenced by claims that gene therapies are unsafe, and gene therapies may not gain the acceptance of the public or the medical community. In particular, our success will depend upon physicians specializing in the treatment of those diseases that our product candidates target prescribing treatments that involve the use of our product candidates in lieu of, or in addition to, existing treatments they are already familiar with and for which greater clinical data may be available. More restrictive government regulations or negative public opinion would have a negative effect on our business or financial condition and may delay or impair the development and commercialization of our product candidates or demand for any products we may develop. Adverse events in our clinical trials, even if not ultimately attributable to our product candidates and the resulting publicity could result in increased governmental regulation, unfavorable public perception, potential regulatory delays in the testing or approval of our potential product candidates, stricter labeling requirements for those product candidates that are approved and a decrease in demand for any such product candidates.
Future sales of our products are subject to adequate coverage, pricing and reimbursement from third-party payors, which are subject to increasing and intense pressure from political, social, competitive and other sources. Our inability to obtain and maintain adequate coverage, pricing or reimbursement, could have an adverse effect on our business.
Future sales of our product candidates, should they receive regulatory approval and be commercialized, are dependent, in large part, on the availability and extent of coverage, pricing and reimbursement from government health administration authorities, private health insurers and other organizations. When a new pharmaceutical product is approved, the availability of government and private reimbursement for that product may be uncertain, as is the pricing and amount for which that product will be reimbursed. The manner and level at which reimbursement is provided for services related to our product candidates (e.g., for administration of our products to patients) is also important. Inadequate reimbursement for such services may lead to physician resistance and adversely affect our ability to market or sell our products.
Pricing and reimbursement for our products and services related to our products may be adversely affected by a number of factors, including:
29
• | changes in federal, state or foreign government regulations or private third-party payors' reimbursement policies; |
• | pressure by employers on private health insurance plans to reduce costs; and |
• | consolidation and increasing assertiveness of payors, including managed care organizations, health insurers, pharmacy benefit managers, government health administration authorities, private health insurers and other organizations, seeking price discounts or rebates in connection with the placement of our products on their formularies and, in some cases, the imposition of restrictions on access or coverage of particular drugs or pricing determined based on perceived value. |
Our failure to maintain adequate coverage, pricing, or reimbursement for our products and services related to our products would have an adverse effect on our business, revenues and results of operation, could curtail or eliminate our ability to adequately fund research and development programs for the discovery and commercialization of new products, and could cause a decline in our stock price.
Drug pricing and other health care costs are under significant scrutiny in the U.S are subject to intense political and societal pressures which we anticipate will continue and escalate on a global basis. As a result, our business and reputation may be harmed, our stock price may be adversely impacted and experience periods of volatility, and our results of operations may be adversely impacted.
If the market opportunities for our product candidates are smaller than we believe they are, our results of operations may be adversely affected and our business may suffer.
We focus our research and product development on treatments of diseases affecting the skin, connective tissue and joints. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who have the potential to benefit from treatment with our product candidates, are based on estimates. These estimates may prove to be incorrect and new studies or clinical trials may change the estimated incidence or prevalence of these diseases. The number of patients in the U.S. and elsewhere may turn out to be lower than expected, may not be otherwise amenable to treatment with our products, or new patients may become increasingly difficult to identify or gain access to, all of which would adversely affect our results of operations and our business.
If any of our approved products were to become the subject of problems related to their efficacy, safety, or otherwise, our business would be seriously harmed.
LAVIV, in addition to any other of our product candidates that may be approved by the FDA, will be subject to continual review by the FDA, and we cannot assure you that newly discovered or developed safety issues will not arise. For all of our product candidates, the FDA has required us to pay special attention to potential skin cancer and hypersensitivity reactions at the site of injection and, while we have seen no issues to date, we cannot rule out that issues may arise in the future. With the use of any newly marketed drug by a wider patient population, serious adverse events ("AE's") may occur from time to time that initially do not appear to relate to the drug itself. Any safety issues could cause us to suspend or cease marketing of our approved products, cause us to modify how we market our approved products, subject us to substantial liabilities, and adversely affect our financial condition and business.
If physicians do not follow our established protocols, the efficacy and safety of our product candidates may be adversely affected.
We are dependent on physicians and other healthcare professionals to follow our established protocols both as to the administration and the handling of our product candidates in connection with our clinical trials, and we continue to be dependent on physicians and other healthcare professionals to follow such protocols after our product candidates are commercialized. The treatment protocol requires each physician to verify the patient’s name and date of birth with the patient and the patient records immediately prior to injection. In the event more than one patient’s cells are delivered to a physician or we deliver the wrong patient’s cells to the physician, which has occurred in the past, it is the physician’s obligation to follow the treatment protocol and assure that the patient is treated with the correct cells. If the physicians and other healthcare professionals do not follow our protocol, the efficacy and safety of our product candidates may be adversely affected.
30
Our competitors in the pharmaceutical, medical device and biotechnology industries may have superior products, manufacturing capabilities, financial resources or marketing position.
The human healthcare products and services industry is extremely competitive. Our competitors include major pharmaceutical, medical device and biotechnology companies. Most of these competitors have more extensive research and development and marketing and manufacturing capabilities than we do, as well as greater financial resources. Our future success will depend on our ability to develop and market effectively our products against those of our competitors. If our products cannot compete effectively in the marketplace, our results of operations and financial position will suffer.
Our cell and gene therapy product candidates may face competition in the future from biosimilars.
With the enactment of the Biologics Price Competition and Innovation Act of 2009 ("BPCIA"), as part of the Patient Protection and Affordable Care Act, an abbreviated pathway for the approval of follow-on biological products was created. The new abbreviated regulatory pathway establishes legal authority for the FDA to review and approve biosimilar biologics, including the possible designation of a biosimilar as “interchangeable” with an existing brand product. Under the BPCIA, an application for a biosimilar product cannot be approved by the FDA until 12 years after the original branded product was approved under a BLA. This data exclusivity does not prevent another company from developing a product that is highly similar to the original branded product, generating its own data and seeking approval. Data exclusivity only assures that another company cannot rely upon the data within the innovator’s application to support the biosimilar product’s approval.
In his proposed budget for fiscal year 2014, President Obama proposed to cut this 12-year period of exclusivity down to seven years. He also proposed to prohibit additional periods of exclusivity due to minor changes in product formulations, a practice often referred to as “evergreening.” It is possible that Congress may take these or other measures to reduce or eliminate periods of exclusivity.
Although final implementation of the BPCIA is not yet complete, such FDA implementation could have a material adverse effect on the future commercial prospects for our product candidates.
We may be liable for product liability claims not covered by insurance.
Physicians, patients and clinical trial participants who have used our products in the past or who use them in the future may bring product liability claims against us. While we have taken, and continue to take, what we believe are appropriate precautions, we may be unable to avoid significant liability exposure. We currently keep in force product liability insurance, although such insurance may not be adequate to fully cover any potential claims or may lapse in accordance with its terms prior to the assertion of claims. We may be unable to obtain product liability insurance in the future, or we may be unable to do so on acceptable terms. Any insurance we obtain or have obtained in the past may not provide adequate coverage against any asserted claims. In addition, regardless of merit or eventual outcome, product liability claims may result in:
•diversion of management’s time and attention;
•expenditure of large amounts of cash on legal fees, expenses and payment of damages;
•decreased demand for our products or any of our future products and services; or
•injury to our reputation.
If we are the subject of any future product liability claims, our business could be adversely affected, and if these claims are in excess of insurance coverage, if any, that we may possess, our financial position will suffer.
Risks Related to Our Dependence on Third Parties
We will incur additional expenses in connection with our exclusive channel collaboration agreements with Intrexon.
Pursuant to our exclusive channel collaboration agreements with Intrexon, we are responsible for future research, development and commercialization expenses of product candidates developed under such collaborations, including FCX-007, FCX-013 and our gene therapy program for arthritis, the effect of which we expect will increase the level of our overall research and development expenses going forward. Although all manufacturing, pre-clinical studies and human clinical trials are expensive and difficult to design and implement, costs associated with the manufacturing, research and development of gene therapy product candidates are generally greater in comparison to small molecule product candidates. We have added personnel and expect to add additional personnel, either directly or through consulting arrangements, to support our exclusive channel collaborations with Intrexon.
31
Because development activities are determined pursuant to a joint steering committee comprised of Intrexon and ourselves and we have limited experience, future development costs associated with this program may be difficult to anticipate and exceed our expectations. Our actual cash requirements may vary materially from our current expectations for a number of other factors that may include, but are not limited to, unanticipated technical challenges, changes in the focus and direction of our development activities or adjustments necessitated by changes in the competitive landscape in which we operate. If we are unable to continue to financially support such collaboration due to our own working capital constraints, we may be forced to discontinue the collaboration or delay our activities.
We may not be able to retain the exclusive rights licensed to us by Intrexon to develop and commercialize our gene therapy product candidates.
Pursuant to our exclusive channel collaboration agreements, we are using Intrexon’s technology in connection with various product candidates. The collaboration agreements grant us a license to use patents and other intellectual property of Intrexon in connection with the research, development, and commercialization of collaboration products within “Fields” that we set forth above in the “Item 1. Business - Intrexon Collaboration”.
The exclusive channel collaboration agreements may be terminated by Intrexon if we fail to exercise diligent efforts in developing products through the collaboration or if we elect not to pursue the development of a therapy in a “Field” identified by Intrexon that is a “Superior Therapy” as defined in the agreement. Upon such termination, the products covered by the applicable exclusive channel collaboration agreement in active and ongoing Phase II or III clinical trials or later stage development through the exclusive channel collaboration agreement shall be entitled to be continued by us with a continuation of the related royalties for such products, and all rights to products covered by the exclusive channel collaboration agreement still in an earlier stage of development shall revert to Intrexon.
There can be no assurance that we will be able to successfully perform under the exclusive channel collaboration agreements and if either agreement is terminated it may prevent us from achieving our business objectives and our business may be harmed.
We depend on third parties to conduct our pre-clinical studies and clinical trials, which may result in costs and delays that prevent us from obtaining regulatory approval or successfully commercializing our product candidates.
We engage third parties to perform various aspects of our pre-clinical studies and clinical trials. For instance, we obtain genetically-modified material from a sole source supplier in connection with the pre-clinical development of RDEB. We depend on these third parties to perform these activities on a timely basis in accordance with the protocol, good laboratory practices, good clinical practices, and other regulatory requirements. Our reliance on these third parties for pre-clinical and clinical development activities reduces our control over these activities. Accordingly, if these parties do not successfully carry out their contractual duties or obligations or meet expected deadlines, our pre-clinical studies and clinical trials may be extended, delayed, terminated or our data may be rejected by the FDA. For example, our sole source supplier of genetically-modified material in connection with the pre-clinical development of RDEB were to cease to be able to supply genetically-modified material to us, or decline to supply genetically-modified material to us, our RDEB program would be delayed until we obtained an alternative source, which could take a considerable length of time. If it became necessary to replace a third party that was assisting with one of our pre-clinical studies or clinical trials, we believe that there are a number of other third-party contractors that could be engaged to continue these activities, although it may result in a delay of the applicable pre-clinical study or clinical trial. If there are delays in testing or obtaining regulatory approvals as a result of a third party’s failure to perform, our drug discovery and development costs will likely increase, and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates.
We have limited manufacturing capacity and any manufacturing difficulties, disruptions or delays could limit supply of our products and or adversely affect our ability to conduct our clinical trials.
Manufacturing biologic products is difficult, complex and highly regulated. We currently manufacture LAVIV and our non-genetically modified cell therapy product candidates at our facility in Exton, PA. We outsource the manufacturing of our genetically modified product candidates to a contract manufacturer in Mountain View, CA. Our ability to adequately and timely manufacture and supply our products is dependent on the operation of our sole facility and those of our contract manufacturer, which may be impacted by, among other things:
• | availability, performance, or contamination of raw materials and components used in the manufacturing process, particularly those for which we have no other source or supplier; |
• | quality of incoming biopsy samples; |
32
• | capacity of our facility and those of contract manufacturer; |
• | the performance of information technology systems; |
• | compliance with regulatory requirements; |
• | inclement weather and natural disasters; |
• | changes in forecasts of future demand for product components; |
• | timing and actual number of production runs for product components; |
• | potential facility contamination by microorganisms or viruses; |
• | updating of manufacturing specifications; and |
• | product quality success rates and yields. |
If the efficient manufacture and supply of our products is interrupted, we may experience delayed shipments or supply constraints. If we are at any time unable to provide an uninterrupted supply of our products to patients, we may lose patients and physicians may elect to prescribe competing therapeutics instead of our products, which could materially and adversely affect our product sales and results of operations. In addition, if we are unable to supply our clinical trials due to manufacturing limitations, our trials may be delayed or compromised.
Our manufacturing processes and those of our contract manufacturer must undergo a potentially lengthy FDA approval process, as well as other regulatory approval processes, and are subject to continued review by the FDA and other regulatory authorities. It is a multi-year process to build and license a new manufacturing facility and it can take significant time to qualify and license a contract manufacturer.
If regulatory authorities determine that we or our contract manufacturer or certain of our third-party service providers have violated regulations or if they restrict, suspend or revoke our prior approvals, they could prohibit us from manufacturing our products or conducting clinical trials or selling our marketed products until we or the affected third-party service providers comply, or indefinitely. Because our third-party service providers are subject to FDA and, potentially, in the future, foreign regulatory authorities, alternative qualified third-party service providers may not be available on a timely basis or at all. If we or our third-party service providers cease or interrupt production or if our third-party service providers fail to supply materials, products or services to us, we may experience delayed shipments, and supply constraints for our products.
Our research, development and manufacturing operations depend on one facility each for both our cell therapy and gene therapy product candidates. If such facility is destroyed or is out of operation for a substantial period of time, our business will be adversely impacted.
We currently conduct our research, development and manufacturing operations related to LAVIV and our non-genetically modified product candidates in one facility located in Exton, Pennsylvania. We currently outsource our research, development and manufacturing operations related to our gene therapy product candidates to a contract manufacturer that uses one facility located in Mountain View, California.
If regulatory, manufacturing or other problems require us to discontinue production at either facility, we will not be able to supply our product to our customers or have supplies for our clinical trials, which would adversely impact our business. If either facility or the equipment in it is significantly damaged or destroyed by fire, flood, power loss or similar events, we may not be able to quickly or inexpensively replace our manufacturing capacity or replace our facility at all. In the event of a temporary or protracted loss of either facility or equipment, we might not be able to transfer manufacturing to a third party. Even if we could transfer manufacturing to a third party, the shift would likely be expensive and time-consuming, particularly since the new facility would need to comply with the necessary regulatory requirements and we would need FDA approval before any products manufactured at that facility could be sold or used.
Risks Related to Our Intellectual Property
If we or our licensors are unable to protect our intellectual property rights or if our intellectual property rights are inadequate for our technologies, products and product candidates, our competitive position could be harmed.
Our commercial success will depend in large part on our, and our licensors, ability to obtain and maintain patent and other intellectual property protection in the U.S. and other countries with respect to our proprietary technology and products. We rely on trade secret, patent, copyright and trademark laws, and confidentiality, licensing and other agreements with
33
employees and third parties, all of which offer only limited protection. We seek to protect our proprietary position by filing and prosecuting patent applications in the U.S. and abroad related to our novel technologies and products that are important to our business.
The patent positions of biopharmaceutical companies generally are highly uncertain, involve complex legal and factual questions and have in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial value of our patents, including those patent rights licensed to us by third parties, are highly uncertain. The steps we or our licensors have taken to protect our proprietary rights may not be adequate to preclude misappropriation of our proprietary information or infringement of our intellectual property rights, both inside and outside the U.S. Further, the examination process may require us or our licensors to narrow the claims for our pending patent applications, which may limit the scope of patent protection that may be obtained if these applications issue. The rights already granted under any of our currently issued patents or those licensed to us and those that may be granted under future issued patents may not provide us with the proprietary protection or competitive advantages we are seeking. If we or our licensors are unable to obtain and maintain patent protection for our technology and products, or if the scope of the patent protection obtained is not sufficient, our competitors could develop and commercialize technology and products similar or superior to ours, and our ability to successfully commercialize our technology and products may be adversely affected. It is also possible that we or our licensors will fail to identify patentable aspects of inventions made in the course of our development and commercialization activities before it is too late to obtain patent protection on them.
With respect to patent rights, we do not know whether any of the pending patent applications for any of our therapies will result in the issuance of patents that protect our technology or products, or if any of our or our licensors’ issued patents will effectively prevent others from commercializing competitive technologies and products. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the U.S. and other jurisdictions are typically not published until 18 months after filing or in some cases not at all, until they are issued as a patent. Therefore, we cannot be certain that we or our licensors were the first to make the inventions claimed in our owned or licensed patents or pending patent applications, or that we or our licensors were the first to file for patent protection of such inventions.
Our issued patents, those that may be issued in the future or those licensed or acquired by us, may be challenged, invalidated or circumvented, and the rights granted under any issued patent may not provide us with proprietary protection or competitive advantages against competitors with similar technology. In particular, we do not know if competitors will be able to design variations on our treatment methods to circumvent our current and anticipated patent claims. Furthermore, competitors may independently develop similar technologies or duplicate any technology developed by us.
Our pending applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issues from such applications. Because the issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, issued patents that we own or have licensed from third parties may be challenged in the courts or patent offices in the U.S. and abroad. Such challenges may result in the loss of patent protection, the narrowing of claims in such patents or the invalidity or unenforceability of such patents, which could limit our ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection for our technology and products. Protecting against the unauthorized use of our or our licensor’s patented technology, trademarks and other intellectual property rights is expensive, difficult and may in some cases not be possible. In some cases, it may be difficult or impossible to detect third-party infringement or misappropriation of our intellectual property rights, even in relation to issued patent claims, and proving any such infringement may be even more difficult.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain and could harm our business.
Our commercial success depends upon our ability to develop, manufacture, and if approved, market and sell our product candidates and to use our related proprietary technologies. We may become party to, or threatened with, future adversarial proceedings or litigation regarding intellectual property rights with respect to our products or product candidates, including interference, post grant review, inter partes review or derivation proceedings before the U.S. Patent and Trademark Office, or USPTO. Third parties may assert infringement claims against us based on existing patents or patents that may be granted in the future. If we are found to infringe a third party’s intellectual property rights, we could be required to obtain a license from such third party to continue commercializing our products. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Under certain circumstances, we could be forced, including by court order, to cease commercializing our products and then expend time and funding to redesign our product and/or product candidates so that it does not infringe others’ patents while still allowing us to compete in the market with a substantially similar product. In addition, in any such proceeding or litigation, we could be found liable for monetary damages. A finding of infringement could prevent us from commercializing our products or force us to cease some of our business operations, which could materially
34
harm our business. Any claims by third parties that we have misappropriated their confidential information or trade secrets could have a similar negative impact on our business. In addition, our involvement in any of these proceedings may cause us to incur substantial costs and result in diversion of management and technical personnel. Furthermore, parties making claims against us may be able to obtain injunctive or other equitable relief that could effectively block our ability to develop, commercialize and sell products, and could result in the award of substantial damages against us.
We believe that use of our product candidates in clinical trials falls within the scope of the exemptions provided by 35 U.S.C. Section 271(e) in the U.S., which exempts from patent infringement liability activities reasonably related to the development and submission of information to the FDA. As our product candidates progresses toward regulatory approval and commercialization, the possibility of a patent infringement claim against us increases. We attempt to ensure that our product candidates and the methods we employ to manufacture them, as well as the methods for their use we intend to promote, do not infringe other parties’ patents and other proprietary rights. There can be no assurance they do not, however, and competitors or other parties may assert that we infringe their proprietary rights in any event.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on all of our product candidates throughout the world would be prohibitively expensive, and our or our licensors’ intellectual property rights in some countries outside the U.S. can be less extensive than those in the U.S. In addition, the laws and practices of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the U.S. Consequently, we and our licensors may not be able to prevent third parties from practicing our and our licensors’ inventions in all countries outside the U.S., or from selling or importing products made using our and our licensors’ inventions in and into the U.S. or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products, and may export otherwise infringing products to territories where we or our licensors have patent protection, but where enforcement is not as strong as that in the U.S. These products may compete with our products in jurisdictions where we do not have any issued patents and our patent claims or other intellectual property rights may not be effective or sufficient to prevent them from so competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biopharmaceuticals, which could make it difficult for us to stop the infringement of our or our licensor’s patents or marketing of competing products in violation of our proprietary rights generally in those countries. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business, could put our and our licensors’ patents at risk of being invalidated or interpreted narrowly and our and our licensors’ patent applications at risk of not issuing and could provoke third parties to assert claims against us or our licensors. We or our licensors may not prevail in any lawsuits that we or our licensors initiate and the damages or other remedies awarded, if any, may not be commercially meaningful.
The laws of certain foreign countries may not protect our rights to the same extent as the laws of the U.S., and these foreign laws may also be subject to change. For example, methods of treatment and manufacturing processes may not be patentable in certain jurisdictions, and the requirements for patentability may differ in certain countries, particularly developing countries. Furthermore, generic drug manufacturers or other competitors may challenge the scope, validity or enforceability of our or our licensors’ patents, requiring us or our licensors to engage in complex, lengthy and costly litigation or other proceedings. Generic drug manufacturers may develop, seek approval for, and launch generic versions of our products. Many countries, including European Union countries, India, Japan and China, have compulsory licensing laws under which a patent owner may be compelled under certain circumstances to grant licenses to third parties. In those countries, we and our licensors may have limited remedies if patents are infringed or if we or our licensors are compelled to grant a license to a third party, which could materially diminish the value of those patents. This could limit our potential revenue opportunities. Accordingly, our and our licensors’ efforts to enforce intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we own or license.
Patent terms may be inadequate to protect our competitive position on our products for an adequate amount of time.
Given the amount of time required for the development, testing and regulatory review of products, such as LAVIV, and our other products candidates patents protecting such candidates might expire before or shortly after such candidates are commercialized. We expect to seek extensions of patent terms in the U.S. and, if available, in other countries where we are prosecuting patents. In the U.S., the Drug Price Competition and Patent Term Restoration Act of 1984 permits a patent term extension of up to five years beyond the normal expiration of the patent, which is limited to the approved indication (or any additional indications approved during the period of extension). However, the applicable authorities, including the FDA and the USPTO in the U.S., and any equivalent regulatory authority in other countries, may not agree with our assessment of whether
35
such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request. If this occurs, our competitors may be able to take advantage of our investment in development and clinical trials by referencing our clinical and pre-clinical data and launch their product earlier than might otherwise be the case.
Changes in patent law, including recent patent reform legislation, could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
As is the case with other biopharmaceutical companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biopharmaceutical industry involve technological and legal complexity, and obtaining and enforcing pharmaceutical patents is costly, time-consuming, and inherently uncertain. Changes in either the patent laws or interpretation of the patent laws in the U.S. and other countries may diminish the value of our patents or narrow the scope of our patent protection. For example, the U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to our and our licensors’ ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our and our licensors’ ability to obtain new patents or to enforce existing patents and patents we and our licensors may obtain in the future. Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our and our licensors’ patent applications and the enforcement or defense of our or our licensors’ issued patents.
In September 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications will be prosecuted and may also affect patent litigation. In particular, under the Leahy-Smith Act, the U.S. transitioned in March 2013 to a “first to file” system in which the first inventor to file a patent application will be entitled to the patent. Third parties are allowed to submit prior art before the issuance of a patent by the USPTO and may become involved in opposition, derivation, reexamination, post grant review, inter-partes review or interference proceedings challenging our patent rights or the patent rights of our licensors. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our or our licensors’ patent rights, which could adversely affect our competitive position. The Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents and those licensed to us.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submissions, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we or our licensors fail to maintain the patents and patent applications covering our product candidates, our competitive position would be adversely affected.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
To protect our proprietary know-how and our inventions for which patents may be unobtainable or difficult to obtain, and to maintain our competitive position, we rely on trade secret protection and confidentiality agreements. To this end, it is our general policy to require our employees, consultants, advisors, and contractors to enter into agreements that prohibit the disclosure of confidential information and, where applicable, require disclosure and assignment to us of the ideas, developments, discoveries, and inventions important to our business. These agreements may not provide adequate protection for our trade secrets, know-how or other proprietary information in the event of any unauthorized use or disclosure or the lawful development by others of such information. Moreover, we may not be able to obtain adequate remedies for any breaches of these agreements. Our trade secrets may also be obtained by third parties by other means, such as breaches of our physical or computer security systems. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States
36
are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they communicate it, from using that technology or information to compete with us. If any of our trade secrets, know-how or other proprietary information is disclosed, the value of our trade secrets, know-how and other proprietary rights would be significantly impaired and our business and competitive position would suffer.
We may become involved in lawsuits to protect or enforce our intellectual property, which could be expensive, time consuming and unsuccessful and have a material adverse effect on the success of our business.
Competitors may infringe our patents or misappropriate or otherwise violate our intellectual property rights. To counter infringement or unauthorized use, litigation may be necessary in the future to enforce or defend our intellectual property rights, to protect our trade secrets or to determine the validity and scope of our own intellectual property rights or the proprietary rights of others. Also, third parties may initiate legal proceedings against us or our licensors to challenge the validity or scope of intellectual property rights we own or control. These proceedings can be expensive and time consuming. Many of our current and potential competitors have the ability to dedicate substantially greater resources to defend their intellectual property rights than we can. Accordingly, despite our efforts, we may not be able to prevent third parties from infringing upon or misappropriating our intellectual property. Litigation could result in substantial costs and diversion of management resources, which could harm our business and financial results. In addition, in an infringement proceeding, a court may decide that a patent owned by or licensed to us is invalid or unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated, held unenforceable or interpreted narrowly. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of shares of our common stock.
We may be subject to claims by third parties asserting that our licensors, employees or we have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property.
Many of our employees and our licensors’ employees, including our senior management, were previously employed at universities or at other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Some of these employees, including each member of our senior management, executed proprietary rights, non-disclosure and non-competition agreements, or similar agreements, in connection with such previous employment. Although we try to ensure that our employees do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or these employees have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such third party. Litigation may be necessary to defend against such claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel or sustain damages. Such intellectual property rights could be awarded to a third party, and we could be required to obtain a license from such third party to commercialize our technology or products. Such a license may not be available on commercially reasonable terms or at all. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management.
Intellectual property rights do not necessarily address all potential threats to our competitive advantage.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations, and may not adequately protect our business, or permit us to maintain our competitive advantage. The following examples are illustrative:
• | others may be able to make compounds that are the same as or similar to our product candidates, but that are not covered by the claims of the patents that we own or have exclusively licensed; |
• | we or our licensors or any strategic partners might not have been the first to make the inventions covered by the issued patents or pending patent applications that we own or have exclusively licensed; |
• | we or our licensors might not have been the first to file patent applications covering certain of our inventions; |
• | others may independently develop similar or alternative technologies or duplicate any of our technologies without infringing our intellectual property rights; |
• | it is possible that our pending patent applications will not lead to issued patents; |
37
• | issued patents that we own or have exclusively licensed may not provide us with any competitive advantages, or may be held invalid or unenforceable as a result of legal challenges; |
• | our competitors might conduct research and development activities in the U.S. and other countries that provide a safe harbor from patent infringement claims for certain research and development activities, as well as in countries where we do not have patent rights and then use the information learned from such activities to develop competitive products for sale in our major commercial markets; |
• | we may not develop additional proprietary technologies that are patentable; and |
• | the patents of others may have an adverse effect on our business. |
Risks Related to Business Operations
We are dependent on our executives and other key professionals and the loss of any of these individuals could harm our business.
We are dependent on the efforts of our executives and other key scientific, manufacturing and quality personnel. The loss of any of these individuals, or our inability to recruit and train additional key personnel in a timely manner, could materially and adversely affect our business and our future prospects. A loss of one or more of our current executives or other key professionals could severely and negatively impact our operations. All of our employees, including our chief executive officer, are employed “at-will,” and any of them may elect to pursue other opportunities at any time. We have no present intention of obtaining key man life insurance on any of our executive officers or key professionals.
We may need to attract, train and retain additional experienced executives and other key professionals in the future.
In the future, we may need to seek additional executives and other key professionals. There is a high demand for experienced executive, scientific, manufacturing and quality personnel in our industry. We do not know whether we will be able to attract, train and retain such experienced personnel in the future, which could have a material adverse effect on our business, financial condition and results of operations.
Our results of operations may be adversely affected by current and potential future healthcare reforms.
In the U.S., federal and state legislatures, health agencies and third-party payors continue to focus on containing the cost of health care. Legislative and regulatory proposals and enactments to reform health care insurance programs could significantly influence the manner in which our products are prescribed and purchased. For example, provisions of the Patient Protection and Affordable Care Act ("PPACA") have resulted in changes in the way health care is paid for by both governmental and private insurers, including increased rebates owed by manufacturers under the Medicaid Drug Rebate Program, annual fees and taxes on manufacturers of certain branded prescription drugs, the requirement that manufacturers participate in a discount program for certain outpatient drugs under Medicare Part D and the expansion of the number of hospitals eligible for discounts under Section 340B of the Public Health Service Act. These changes have had and are expected to continue to have a significant impact on our business.
There is also significant economic pressure on state budgets that may result in states increasingly seeking to achieve budget savings through mechanisms that limit coverage or payment for our drugs. In recent years, some states have considered legislation and ballot initiatives that would control the prices of drugs, including laws to allow importation of pharmaceutical products from lower cost jurisdictions outside the U.S. and laws intended to impose price controls on state drug purchases. State Medicaid programs are increasingly requesting manufacturers to pay supplemental rebates and requiring prior authorization by the state program for use of any drug for which supplemental rebates are not being paid. Government efforts to reduce Medicaid expenses may lead to increased use of managed care organizations by Medicaid programs. This may result in managed care organizations influencing prescription decisions for a larger segment of the population and a corresponding constraint on prices and reimbursement for our products. In addition, under the PPACA, as states implement their health care marketplaces or operate under the federal exchange, the impact on drug manufacturers will depend in part on the formulary and benefit design decisions made by insurance sponsors or plans participating in these programs. It is possible that we may need to provide discounts or rebates to such plans in order to maintain favorable formulary access for our future products, which could have an adverse impact on our sales and results of operations.
If we fail to comply with federal and state healthcare laws, including fraud and abuse and health information privacy and security laws, we could face substantial penalties and our business, results of operations, financial condition and prospects could be adversely affected.
38
As a biopharmaceutical company, even though we do not and will not control referrals of healthcare services or bill directly to Medicare, Medicaid or other third-party payors, certain federal and state healthcare laws and regulations pertaining to fraud and abuse and patients’ rights are and will be applicable to our business. For example, we could be subject to healthcare fraud and abuse and patient privacy regulation by both the federal government and the states in which we conduct our business. The laws that may affect our ability to operate include, among others:
• | The federal Anti-Kickback Statute, which constrains our business activities, including our marketing practices, educational programs, pricing policies, and relationships with healthcare providers or other entities, by prohibiting, among other things, soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce, or in return for, either the referral of an individual or the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
• | Federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payors that are false or fraudulent; |
• | The federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created new federal criminal statutes that prohibit, among other things, executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters; |
• | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, and its implementing regulations, which imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information; |
• | Requirements to report annually to CMS certain financial arrangements with physicians and teaching hospitals, as defined in the ACA and its implementing regulations, including reporting any “transfer of value” made or distributed to teaching hospitals, prescribers, and other healthcare providers and reporting any ownership and investment interests held by physicians and their immediate family members and applicable group purchasing organizations during the preceding calendar year; and |
• | State and foreign law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payor, including commercial insurers; state laws that require pharmaceutical companies to comply with the industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government that otherwise restricts certain payments that may be made to healthcare providers and entities; state laws that require drug manufacturers to report information related to payments and other transfer of value to physicians and other healthcare providers and entities; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. In addition, recent health care reform legislation has further strengthened these laws. For example, the ACA, among other things, amends the intent requirement of the federal anti-kickback and certain criminal healthcare fraud statutes. A person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it. Moreover, the ACA provides that the government may assert that a claim including items or services resulting from a violation of the federal anti-kickback statute constitutes a false or fraudulent claim for purposes of the False Claims Act.
To the extent that any of our product candidates is ultimately sold in a foreign country, we may be subject to similar foreign laws and regulations.
If we or our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including administrative, civil and criminal penalties, damages, fines, exclusion from participation in United States federal or state health care programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations any of which could materially adversely affect our ability to operate our business and our financial results. Although compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, the risks cannot be entirely eliminated. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. Moreover, achieving and sustaining compliance with applicable federal and state privacy, security and fraud laws may prove costly.
39
If we fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of our business.
We are subject to numerous environmental, health and safety laws and regulations, including those governing manufacturing and laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Our operations involve the use of hazardous and flammable materials, including chemicals and biological materials. Our operations also produce hazardous waste products. We generally contract with third parties for the disposal of these materials and wastes. We cannot eliminate the risk of contamination or injury from these materials. In the event of contamination or injury resulting from our use of hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials or other work-related injuries, this insurance may not provide adequate coverage against potential liabilities. In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts. Failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions.
Our business and operations would suffer in the event of computer system failures.
Despite the implementation of security measures, our internal computer systems, and those of our contract research organizations, contract manufacturing organization, and other third parties on which we rely, are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. System failures, accidents or security breaches could cause interruptions in our operations or the unauthorized transfer of our proprietary information, and could result in a material disruption of our clinical activities and business operations, in addition to possibly requiring substantial expenditures of resources to remedy. If such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs. For example, the loss of clinical trial data from completed or ongoing clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach was to result in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development of our product candidates could be delayed.
Our ability to use net operating loss carryforwards to reduce future tax payments may be limited or restricted.
We have generated significant net operating loss carryforwards (“NOL's”) as a result of our incurrence of losses and our conduct of research activities since inception. We generally are able to carry NOL's forward to reduce our tax liability in future years. However, our ability to utilize the NOL's is subject to the rules of Sections 382 and 383 of the Internal Revenue Code of 1986, as amended (the "Code"). Those sections generally restrict the use of NOL's after an “ownership change.” An ownership change occurs if, among other things, the stockholders (or specified groups of stockholders) who own or have owned, directly or indirectly, 5% or more of a corporation’s common stock or are otherwise treated as 5% stockholders under Section 382 of the Code and the United States Treasury Department regulations promulgated thereunder increase their aggregate percentage ownership of that corporation’s stock by more than 50 percentage points over the lowest percentage of the stock owned by these stockholders over the applicable testing period. In the event of an ownership change, Section 382 imposes an annual limitation on the amount of taxable income a corporation may offset with NOL carry forwards and Section 383 imposes an annual limitation on the amount of tax a corporation may offset with carry forwards. Any unused annual limitation may be carried over to later years until the applicable expiration date for the respective NOL carry forwards. We have completed several financings since our inception which we believe have resulted in “ownership changes” within the meaning of Section 382. We may also experience ownership changes in the future as a result of additional financings and subsequent shifts in our stock ownership. As a result, our NOL's may be subject to limitations and we may be required to pay taxes earlier and in larger amounts than would be the case if our NOL's were freely usable.
40
Risks Related to Ownership of our Common Stock
The trading price of the shares of our common stock has been highly volatile, and purchasers of our common stock could incur substantial losses.
Our stock began trading on NYSE MKT on May 17, 2013 and then on the NASDAQ Stock Market LLC ("NASDAQ") on August 29, 2014. Between May 17, 2013 and December 31, 2015, our common stock has traded between $2.28 and $7.60. Our stock price could be subject to wide fluctuations in response to a variety of factors, which include:
• | whether our clinical trials can be conducted within the timeframe that we expect and whether such trials will yield positive results; |
• | whether our collaborations with Intrexon can be advanced with positive results within the timeframe and budget that we expect; |
• | changes in laws or regulations applicable to our products or product candidates, including but not limited to clinical trial requirements for approvals; |
• | unanticipated serious safety concerns related to the use of our products or product candidates; |
• | a decision to initiate a clinical trial, not to initiate a clinical trial or to terminate an existing clinical trial; |
• | our ability to increase our manufacturing capacity and reduce our manufacturing costs through the improvement of our manufacturing process, our ability to validate any such improvements with the relevant regulatory agencies and our ability to accomplish the foregoing on a timely basis; |
• | adverse regulatory decisions; |
• | the introduction of new products or technologies offered by us or our competitors; |
• | negative public opinion or perception of cell and gene therapies; |
• | the inability to effectively manage our growth; |
• | actual or anticipated variations in quarterly operating results; |
• | the failure to meet or exceed the estimates and projections of the investment community; |
• | the perception of the biopharmaceutical industry by the public, legislatures, regulators and the investment community; |
• | the overall performance of the U.S. equity capital markets and general political and economic conditions; |
• | announcements of significant acquisitions, strategic partnerships, joint ventures, collaborations or capital commitments by us or our competitors; |
• | disputes or other developments relating to proprietary rights, including patents, litigation matters and our ability to obtain patent protection for our technologies; |
• | additions or departures of key personnel; |
• | the trading volume of our common stock; and |
• | other events or factors, many of which are beyond our control. |
In addition, the stock market in general, and the stock of biotechnology companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our actual operating performance.
Randal J. Kirk and certain of his affiliates (including Intrexon) own a substantial percentage of our common stock and will be able to exert significant influence over matters subject to stockholder approval.
As of March 4, 2016, Randal J. Kirk and certain of his affiliates (including Intrexon, our collaboration partner on our gene therapy programs) beneficially owned approximately 16.6 million shares, or approximately 38%, of our common stock. Mr. Kirk and his affiliates may have interests that conflict with our other stockholders and, if acting together, have the ability to significantly influence the outcome of matters submitted to our stockholders for approval, including the election and removal of directors and any merger, consolidation or sale of all or substantially all of our assets. This concentration of ownership could delay or prevent any acquisition of our company on terms that other stockholders may desire.
41
Additionally, two of our directors, Julian Kirk (who is the son of Randal J. Kirk) and Marcus Smith, are employees of Third Security, LLC, which is an affiliate of Randal J. Kirk.
Our operating results may fluctuate significantly in the future, which may cause our results to fall below the expectations of securities analysts, stockholders and investors.
Our operating results may fluctuate significantly in the future as a result of a variety of factors, many of which are outside of our control. These factors include, but are not limited to:
• | the timing, implementation and cost of our pre-clinical studies and clinical trials; |
• | expenses in connection with our exclusive channel collaboration agreements with Intrexon; |
• | the timely and successful implementation of improved manufacturing processes; |
• | our ability to attract and retain personnel with the necessary strategic, technical and creative skills required for effective operations; |
• | the amount and timing of expenditures by practitioners and their patients; |
• | introduction of new technologies; |
• | product liability litigation, class action and derivative action litigation, or other litigation; |
• | the amount and timing of capital expenditures and other costs relating to the expansion of our operations; |
• | the state of the debt and/or equity capital markets at the time of any proposed offering we choose to initiate; |
• | our ability to successfully integrate new acquisitions into our operations; |
• | government regulation and legal developments regarding our product candidates in the United States and in the foreign countries in which we may operate in the future; and |
• | general economic conditions. |
As a strategic response to changes in the competitive environment, we may from time to time make pricing, service, technology or marketing decisions or business or technology acquisitions that could have a material adverse effect on our operating results. Due to any of these factors, our operating results may fall below the expectations of securities analysts, stockholders and investors in any future period, which may cause our stock price to decline.
Future sales of our common stock may depress our stock price.
The market price of our common stock could decline as a result of sales of substantial amounts of our common stock in the public market, or as a result of the perception that these sales could occur, which could occur if we issue a large number of shares of common stock (or securities convertible into our common stock) in connection with a future financing, as our common stock is trading at low levels. These factors could make it more difficult for us to raise funds through future offerings of common stock or other equity securities. As of the date of this report, all of our outstanding shares held by non-affiliates are freely transferable without restriction or further registration under the Securities Act. In addition to our common stock outstanding, as of December 31, 2015, we had warrants and options outstanding that were exercisable for a total of 7,146,718 shares of common stock.
We have not declared any dividends on our common stock to date, and we have no intention of declaring dividends in the foreseeable future.
The decision to pay cash dividends on our common stock rests with our Board of Directors and will depend on our earnings, unencumbered cash, capital requirements and financial condition. We do not anticipate declaring any dividends in the foreseeable future, as we intend to use any excess cash to fund our operations. Investors in our common stock should not expect to receive dividend income on their investment, and investors will be dependent on the appreciation of our common stock to earn a return on their investment.
42
Provisions in our charter documents could prevent or delay stockholders’ attempts to replace or remove current members of our Board of Directors.
Our charter documents provide for staggered terms for the members of our Board of Directors. Our Board of Directors is divided into three staggered classes, and each director serves a term of three years. At stockholders’ meetings, only those directors comprising one of the three classes will have completed their term and be subject to re-election or replacement.
In addition, our Board of Directors is authorized to issue “blank check” preferred stock, with designations, rights and preferences as they may determine. Accordingly, our Board of Directors has in the past and may in the future, without stockholder approval, issue shares of preferred stock with dividend, liquidation, conversion, voting or other rights that could adversely affect the voting power or other rights of the holders of our common stock. This type of preferred stock could also be issued to discourage, delay or prevent a change in our control.
The use of a staggered Board of Directors and the ability to issue “blank check” preferred stock are traditional anti-takeover measures. These provisions in our charter documents make it difficult for a majority stockholder to gain control of the Board of Directors and of our company. These provisions may be beneficial to our management and our Board of Directors in a hostile tender offer and may have an adverse impact on stockholders who may want to participate in such a tender offer, or who may want to replace some or all of the members of our Board of Directors.
Provisions in our bylaws provide for indemnification of officers and directors, which could require us to direct funds away from our business and future products.
Our bylaws provide for the indemnification of our officers and directors. We have in the past and may in the future be required to advance costs incurred by an officer or director and to pay judgments, fines and expenses incurred by an officer or director, including reasonable attorneys’ fees, as a result of actions or proceedings in which our officers and directors are involved by reason of being or having been an officer or director of our company. Funds paid in satisfaction of judgments, fines and expenses may be funds we need for the operation of our business and the development of our product candidates, thereby affecting our ability to attain profitability.
Provisions of the warrants issued in connection with certain of our prior financings provide for preferential treatment to the holders of the warrants and could impede a sale of the Company.
The warrants we issued in connection with certain of our prior financings gives each holder the option to receive a cash payment based on a Black-Scholes valuation upon our change of control or upon our failure to be listed on any trading market. We are required, at the warrant holder’s option, exercisable at any time concurrently with, or within 30 days after, the announcement of a fundamental transaction, to redeem all or any portion of these warrants from the warrant holder by paying to the holder an amount of cash equal to the Black-Scholes value of the remaining unexercised portion of the warrant on or prior to the date of the consummation of such fundamental transaction.
An active market for our common stock may not be sustained.
In the past, we have had a limited, volatile and sporadic public trading market for our common stock. Although our common stock is listed on NASDAQ, an active trading market for our common stock may not be sustained, especially given the large percentage of our common stock held by our affiliates. If an active market for our common stock is not sustained, it may be difficult for our stockholders to sell shares without depressing the market price for our common stock.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our common stock depends in part on the research and reports that securities or industry analysts publish about us or our business. We do not have any control over these analysts. If one or more of the analysts who cover us downgrade our stock or publish inaccurate or unfavorable research about our business, our stock price would likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our stock could decrease, which might cause our stock price and trading volume to decline.
Item 1B. Unresolved Staff Comments
None.
43
Item 2. Properties
Our corporate office and manufacturing facility are located at 405 Eagleview Boulevard, Exton, Pennsylvania. This location consists of approximately 17,500 square feet of manufacturing and laboratory space and 69,000 square feet of office space, which we lease pursuant to a lease agreement that expires on March 31, 2023. We believe this facility is suitable for our current needs.
Item 3. Legal Proceedings
We are not a party to any pending legal proceedings.
Item 4. Mine Safety Disclosure
Not applicable.
Part II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
Our common stock trades under the symbol “FCSC.” Our stock traded on the Over the Counter Bulletin Board (“OTCBB”) from October 21, 2009 until May 16, 2013. On May 17, 2013 our stock began trading on the NYSE MKT. On August 28, 2014, the Company delisted its common stock from the NYSE MKT and began trading on NASDAQ.
The following table sets forth, for the indicated periods in which we traded on the NYSE MKT and NASDAQ, the high and low intra-day sales prices per share for our common stock.
High | Low | ||||||
Year Ended December 31, 2015 | |||||||
First Quarter | $ | 5.99 | $ | 2.38 | |||
Second Quarter | $ | 6.40 | $ | 3.25 | |||
Third Quarter | $ | 7.60 | $ | 3.68 | |||
Fourth Quarter | $ | 6.18 | $ | 3.50 | |||
Year Ended December 31, 2014 | |||||||
First Quarter | $ | 6.30 | $ | 4.00 | |||
Second Quarter | $ | 5.23 | $ | 2.70 | |||
Third Quarter prior to August 29, 2014 (NYSE MKT until the date we began trading on the NASDAQ) | $ | 4.31 | $ | 2.88 | |||
Third Quarter (from August 29, 2014 to September 30, 2014) | $ | 3.40 | $ | 2.60 | |||
Fourth Quarter | $ | 3.25 | $ | 2.28 | |||
The closing price of our common stock on March 4, 2016 was $2.70 as reported on NASDAQ.
Holders of Record
As of March 4, 2016, there were 43,898,785 shares of our common stock outstanding. There were approximately 145 stockholders of record at March 4, 2016. Because many of our shares are held by brokers and other institutions on behalf of stockholders, we are unable to estimate the total number of stockholders represented by these record holders.
Dividends
We have never declared or paid any cash dividend on our common stock and our Board of Directors does not intend to do so in the foreseeable future. The declaration and payment of dividends in the future, of which there can be no assurance, will be determined by the Board of Directors in light of conditions then existing, including earnings, financial condition, capital requirements and other factors.
44
Securities Authorized for Issuance under Equity Compensation Plans
Information regarding securities authorized for issuance under equity compensation plans is incorporated by reference into the information in Part III, Item 12 of this Annual Report on Form 10-K.
Recent Sales of Unregistered Securities
All information regarding the issuance of our securities during the quarter ended December 31, 2015 have been previously disclosed in current reports we have filed on Form 8-K or in quarterly reports we have filed on Form 10-Q. We did not issue any unregistered equity securities during the quarter ended December 31, 2015.
Stock Performance Graph
The following graph compares the cumulative total return on our common stock relative to the cumulative total returns of the NASDAQ composite index and the NASDAQ Biotechnology index for the period from December 31, 2010 through December 31, 2015. An investment of $100 (with reinvestment of all dividends) is assumed to have been made in our common stock and in each of the indexes on December 31, 2010 and its relative performance is tracked through December 31, 2015.
As of | |||||||||||||||||||||||
12/31/2010 | 12/31/2011 | 12/31/2012 | 12/31/2013 | 12/31/2014 | 12/31/2015 | ||||||||||||||||||
Fibrocell Science, Inc. | $ | 100.00 | $ | 78.43 | $ | 29.41 | $ | 31.84 | $ | 20.31 | $ | 35.69 | |||||||||||
NASDAQ Composite Index | $ | 100.00 | $ | 98.20 | $ | 113.82 | $ | 157.44 | $ | 178.53 | $ | 188.75 | |||||||||||
NASDAQ Biotechnology Index | $ | 100.00 | $ | 111.81 | $ | 147.48 | $ | 244.24 | $ | 327.52 | $ | 364.93 | |||||||||||
The stock price performance included in this graph is not necessarily indicative of future stock price performance.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
We did not repurchase any of our equity securities during the year ended December 31, 2015.
Item 6. Selected Financial Data
The following selected consolidated financial data should be read in conjunction with Part II, Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations," and our Consolidated Financial Statements and the accompanying notes included in Part II, Item 8, "Financial Statements and Supplementary Data" of this Annual Report on Form 10-K.
45
The Consolidated Statements of Operations data for each of the years ended December 31, 2015, 2014, and 2013 and the Consolidated Balance Sheets data as of December 31, 2015 and 2014 are derived from our audited Consolidated Financial Statements included in Part II, Item 8, "Financial Statements and Supplementary Data" of this Annual Report on Form 10-K. The Consolidated Statements of Operations data for the years ended December 31, 2012 and 2011 and the Consolidated Balance Sheets data as of December 31, 2013, 2012, and 2011 are derived from our audited Consolidated Financial Statements that are not included in this Annual Report on Form 10-K. Our historical results are not necessarily indicative of our results in any future period.
Year Ended December 31, | ||||||||||||||||||||
2015 | 2014 | 2013 | 2012 | 2011 | ||||||||||||||||
($ in thousands, except per share data) | ||||||||||||||||||||
Consolidated Statements of Operations data: | ||||||||||||||||||||
Total revenue | $ | 492 | $ | 180 | $ | 200 | $ | 153 | $ | — | ||||||||||
Total cost of revenue | 722 | 2,312 | 7,501 | 7,804 | 13 | |||||||||||||||
Research and development expenses (1) | 25,892 | 17,735 | 13,762 | 10,193 | 7,171 | |||||||||||||||
Selling, general and administrative expenses (1) | 11,285 | 10,087 | 9,440 | 11,546 | 12,795 | |||||||||||||||
Warrant revaluation and other finance income (expense) | 2,929 | 3,930 | (1,053 | ) | 20,404 | 2,562 | ||||||||||||||
Loss from continuing operations | (34,453 | ) | (25,650 | ) | (31,554 | ) | (13,143 | ) | (23,931 | ) | ||||||||||
Net loss | (34,453 | ) | (25,650 | ) | (31,554 | ) | (12,687 | ) | (24,025 | ) | ||||||||||
Per Share Information (2): | ||||||||||||||||||||
Loss from continuing operations per common share - basic | $ | (0.82 | ) | $ | (0.63 | ) | $ | (1.06 | ) | $ | (1.47 | ) | $ | (10.91 | ) | |||||
Loss from continuing operations per common share - diluted | $ | (0.85 | ) | $ | (0.70 | ) | $ | (1.12 | ) | $ | (2.65 | ) | $ | (10.99 | ) | |||||
Net loss per common share - basic | $ | (0.82 | ) | $ | (0.63 | ) | $ | (1.06 | ) | $ | (1.42 | ) | $ | (10.96 | ) | |||||
Net loss per common share - diluted | $ | (0.85 | ) | $ | (0.70 | ) | $ | (1.12 | ) | $ | (2.60 | ) | $ | (11.04 | ) | |||||
(1) Included in both selling, general and administrative expenses and research and development expenses, combined, is $2.0 million, $1.2 million, $1.2 million, $1.2 million, and $2.9 million of stock-based compensation for the years ended December 31, 2015, 2014, 2013, 2012, and 2011, respectively.
(2) | The per share data has been retroactively adjusted to reflect the April 30, 2013 reverse stock split of 25 to 1. See Note 13 of the Consolidated Financial Statements for a description of our computation of basic and diluted loss per share attributable to common stockholders. |
As of December 31, | ||||||||||||||||||||
2015 | 2014 | 2013 | 2012 | 2011 | ||||||||||||||||
($ in thousands) | ||||||||||||||||||||
Consolidated Balance Sheets Data: | ||||||||||||||||||||
Cash and cash equivalents | $ | 29,268 | $ | 37,495 | $ | 60,033 | $ | 31,346 | $ | 10,799 | ||||||||||
Total current assets | 30,994 | 39,349 | 61,860 | 33,156 | 12,499 | |||||||||||||||
Total assets | 36,712 | 45,634 | 69,014 | 40,603 | 20,274 | |||||||||||||||
Total current liabilities | 15,365 | 3,493 | 3,593 | 1,554 | 9,611 | |||||||||||||||
Warrant liability, current and long term | 8,275 | 11,286 | 15,216 | 14,515 | 23,754 | |||||||||||||||
Total liabilities | 22,509 | 15,225 | 19,348 | 16,413 | 36,541 | |||||||||||||||
Total equity (deficit) and noncontrolling interest | 14,203 | 30,409 | 49,666 | 24,190 | (16,268 | ) | ||||||||||||||
We have never paid dividends on our common stock.
46
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
This Management’s Discussion and Analysis of Financial Conditions and Results of Operations should be read in conjunction with our financial statements and the related notes included elsewhere in this Form 10-K.
Overview
We are an autologous cell and gene therapy company translating personalized biologics into medical breakthroughs. We are focused on discovering and developing new therapies for the localized treatment of diseases affecting the skin, connective tissue and joints. Our approach to personalized biologics is distinctive based on our proprietary autologous fibroblast technology: targeting the underlying cause of disease by using cells from the skin to create localized therapies—with or without genetic modification—that are compatible with the unique biology of each patient.
Development Programs
Our most advanced development program is azficel-T for vocal cord scarring resulting in chronic or severe dysphonia. We are currently in a Phase II clinical trial for this indication. We have completed dosing in this trial and expect to announce primary endpoint results in the second quarter of 2016.
In collaboration with Intrexon, we are in pre-clinical development with two gene-therapy product candidates.
Our lead gene-therapy product candidate, FCX-007, has received orphan drug designation as well as rare pediatric disease
designation from the FDA and is in late-stage pre-clinical development for the treatment of RDEB, a devastating, rare, congenital, painful, progressive. blistering skin disease that typically leads to premature death. We expect to file an amended IND with the FDA and, subject to FDA approval, initiate our phase I/II clinical trial in the second quarter of 2016. We are also in pre-clinical development of our second gene-therapy product candidate, FCX-013, for the treatment of linear scleroderma, an excess production of extracellular matrix characterized by skin fibrosis and linear scars. We have completed our proof-of-concept study for FCX-013 and expect to file an IND in 2017. We plan to seek orphan drug designation for FCX-013.
In September 2015, we and Intrexon entered into a letter of agreement pursuant to which we mutually agreed to terminate our collaboration with respect to the development of potential therapies to treat Ehlers-Danlos Syndrome (hypermobility type) due to technical hurdles. As a result, we no longer have any rights or obligations under the 2012 ECC with respect to the development of “autologous human fibroblasts genetically modified to express bioactive Tenascin-X locally to correct connective tissue disorders”.
In December 2015, we expanded our collaboration with Intrexon to pursue the research, development and
commercialization of products for the treatment of chronic inflammation and degenerative diseases of human joints through intra-articular or other local administration of genetically modified fibroblasts. We are currently in the research phase for a gene therapy to treat arthritis under this collaboration. Our goal is to deliver a protein therapy locally to the joint to provide sustained efficacy while avoiding key side effects typically associated with systemic therapy.
We also have a scientific research collaboration with UCLA focused on media that promotes genomic stability in induced pluripotent stem cell cultures. We had previously been a party to additional collaborations with UCLA and the Massachusetts Institute of Technology relating to isolating stem cell sub-populations in the skin; however, we terminated these other programs and related agreements in 2015.
Commercial Programs
LAVIV (azficel-T) is an FDA-approved biological product that uses our proprietary autologous fibroblast technology for the improvement of the appearance of moderate to severe nasolabial fold wrinkles in adults. In 2013, we shifted our strategic focus to diseases affecting the skin, connective tissue and joints, resulting in the pre-clinical and clinical product candidates mentioned above. As a result, we no longer actively market or promote LAVIV to physicians but will continue to accept prescriptions, for which we expect a nominal amount in 2016.
See Part I, Item 1, "Business" of this Annual Report on Form 10-K for additional details regarding our development programs, commercial programs, and collaboration agreements.
Critical Accounting Policies
The following discussion and analysis of financial condition and results of operations are based upon our Consolidated Financial Statements, which have been prepared in conformity with U.S. generally accepted accounting principles (“GAAP”).
47
Preparing financial statements requires us to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses. Estimates are based on our historical operations, our future business plans and projected financial results, the terms of existing contracts, our observance of trends in the industry, information provided by our customers and information available from other outside sources, as appropriate. These estimates and assumptions are affected by the application of our accounting policies. Critical accounting policies and practices are both important to the portrayal of a company’s financial condition and results of operations, and require management’s most difficult, subjective or complex judgments, often as a result of the need to make estimates about the effects of matters that are inherently uncertain. Actual results could differ from such estimates due to changes in economic factors or other conditions that are outside the control of management. A summary of our significant accounting policies is described in Note 3 of the Consolidated Financial Statements contained in this Form 10-K.
Basis of Presentation
The following discussion should be read in conjunction with the Consolidated Financial Statements and the accompanying Notes of the Consolidated Financial Statements included in this Form 10-K.
The prior year financial data included in Part II, Item 7, "Management's Discussion and Analysis of Financial Condition and Results of Operations," contains certain reclassifications to the results of operations for the years ended December 31, 2014 and 2013 to conform to the presentation for the year ended December 31, 2015 in this Form 10-K. These reclassifications were made in conjunction with the Company’s strategic shift away from the aesthetics market (LAVIV) and towards diseases affecting the skin, connective tissue and joints. See Note 2 of the Consolidated Financial Statements included in this Form 10-K for additional details.
Results of Operations
Comparison of Years Ending December 31, 2015, 2014 and 2013
Revenue and Cost of Revenue
Revenue and cost of revenue were comprised of the following:
Year Ended December 31, | 2015 vs 2014 % Change | 2014 vs 2013 % Change | ||||||||||||||||
($ in thousands) | 2015 | 2014 | 2013 | |||||||||||||||
Revenue from product sales | $ | 270 | $ | 170 | $ | 200 | 58.8 | % | (15.0 | )% | (1) | |||||||
Collaboration revenue | 222 | 10 | — | 2,120.0 | % | 100.0 | % | (2) | ||||||||||
Total revenue | 492 | 180 | 200 | 173.3 | % | (10.0 | )% | |||||||||||
Cost of product sales | 426 | 2,312 | 7,501 | (81.6 | )% | (69.2 | )% | (3) | ||||||||||
Cost of collaboration revenue | 296 | — | — | 100.0 | % | — | % | (4) | ||||||||||
Total cost of revenue | 722 | 2,312 | 7,501 | (68.8 | )% | (69.2 | )% | |||||||||||
Gross loss | $ | (230 | ) | $ | (2,132 | ) | $ | (7,301 | ) | (89.2 | )% | (70.8 | )% | |||||
(1) | Revenue from product sales has remained relatively consistent, ranging from approximately $0.2 million to $0.3 million. Revenue from product sales solely relates to, and is recognized based on, the shipment of LAVIV injections to patients. Although the number of injections can fluctuate from period to period, product revenues continue to be, and are expected to remain, insignificant to our operations. |
(2) | Collaboration revenue is related to a research and development agreement that we have with a third party to investigate potential new non-pharmaceutical applications for our conditioned fibroblast media technology. We entered into to this agreement during the fourth quarter of 2014. Revenue recognized to date relates to an upfront license fee of approximately $0.1 million that is being amortized over the estimated total contract period and $0.2 million for a proof-of-concept study that was completed during the fourth quarter of 2015. |
(3) | Cost of product sales includes the costs related to the processing of cells for LAVIV, including direct and indirect costs. Beginning in 2014, cost of product sales was comprised of allocated costs associated with our manufacturing, facility, quality control and quality assurance operations as well as overhead costs. LAVIV has historically generated gross losses, primarily due to low volumes which result in a higher allocation of fixed overhead costs to commercial sales. This trend is expected to continue for 2016. |
48
Costs of product sales experienced a decrease in both 2015 ($1.9 million, or 81.6%) and 2014 ($5.2 million, or 69.2%), compared to their respective immediately preceding fiscal years. The decrease for both comparable periods is primarily due to our strategic shift away from the aesthetic market (LAVIV) and towards diseases affecting the skin, connection tissue and joints, as well as to an increase in clinical manufacturing, which reduces the allocation of manufacturing and fixed overhead costs to commercial sales and increases the allocation to research and development expenses.
(4) | Cost of collaboration revenue was approximately $0.3 million in 2015, and consists primarily of manufacturing costs related to a proof-of-concept study which begun and was completed in 2015. There were no such costs in 2014. |
Research and Development Expenses
For each of our research and development programs, we incur both direct and indirect expenses. We track direct research and development expenses by program, which include third party costs such as contract research, consulting and pre-clinical and clinical development costs. We do not allocate indirect research and development expenses, which may include regulatory, laboratory (equipment and supplies), personnel, facility, process development and other overhead costs (including depreciation and amortization), to specific programs, as these expenses are to be deployed across all of our product candidates. We expect research and development costs to continue to be significant for the foreseeable future as a result of our pre-clinical studies and clinical trials, as well as our ongoing collaborations with Intrexon.
As depicted below, we devoted significant resources towards research and development activities directly related to our pre-clinical and clinical development programs. For the years ended December 31, 2015, 2014 and 2013, direct research and development expenses represented approximately 68%, 62% and 77%, respectively, of total research and development expenses.
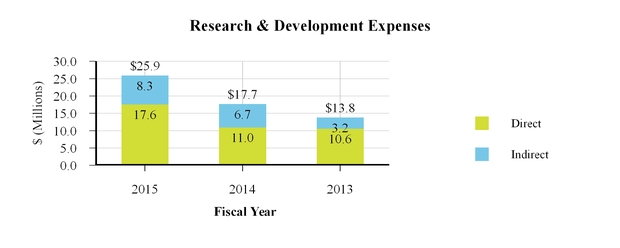
Total research and development expenses increased approximately $8.2 million, or 46%, to $25.9 million for the year ended December 31, 2015 as compared to $17.7 million for the year ended December 31, 2014. In 2015, we incurred $10.0 million for an up-front license fee for our latest exclusive channel collaboration with Intrexon. Additionally, we had increased spending of approximately $1.0 million related to the completed proof-of-concept study for FCX-013 and $0.8 million related to the IND filing for FCX-007, offset by a decrease in spending of $5.2 million in connection with supplemental stock issuance costs incurred in 2014 for the second amendment to our original channel collaboration with Intrexon, resulting in a $6.6 million increase in the direct component of total research and development expenses. We also had increased process development expenses of $1.7 million in 2015, which was the primary contributor to the $1.6 million increase in the indirect component of total research and development expenses.
Total research and development expenses increased approximately $4.0 million, or 29%, to $17.7 million for the year ended December 31, 2014 as compared to $13.8 million for the year ended December 31, 2013. The increase is primarily due to an increase in total indirect research and development costs of approximately $3.5 million, including $3.0 million of compensation and related expenses, as a result of the greater allocation of operational and overhead costs to research and development expenses associated with our strategic shift away from the aesthetic market (LAVIV) and an increase in clinical manufacturing. Similar costs would have been included in cost of product sales for the year ended December 31, 2013. There was also a minimal increase in total direct research and development costs of approximately $0.4 million.
49
Direct research and development costs, by major program, and indirect research and development costs, by major component, were as follows:
Year Ended December 31, | 2015 vs 2014 % Change | 2014 vs 2013 % Change | ||||||||||||||||
($ in thousands) | 2015 | 2014 | 2013 | |||||||||||||||
Direct costs: | ||||||||||||||||||
azficel-T for vocal cord scarring | $ | 1,149 | $ | 778 | $ | 322 | 47.7 | % | 141.6 | % | (1) | |||||||
FCX-007 | 4,572 | 3,800 | 1,773 | 20.3 | % | 114.3 | % | (2) | ||||||||||
FCX-013 | 1,541 | 558 | 7,140 | 176.2 | % | (92.2 | )% | (3) | ||||||||||
Arthritis | 10,187 | — | — | 100.0 | % | — | % | (4) | ||||||||||
Ehlers-Danlos Syndrome (hypermobility type) | — | 5,171 | — | (100.0 | )% | 100.0 | % | (5) | ||||||||||
Other | 139 | 679 | 1,315 | (79.5 | )% | (48.4 | )% | (6) | ||||||||||
Total direct costs | 17,588 | 10,986 | 10,550 | 60.1 | % | 4.1 | % | |||||||||||
Indirect costs: | ||||||||||||||||||
Regulatory costs | 957 | 972 | 923 | (1.5 | )% | 5.3 | % | |||||||||||
Intangible amortization | 551 | 551 | 551 | — | % | — | % | |||||||||||
Compensation and related expenses | 3,923 | 3,257 | 270 | 20.4 | % | 1,106.3 | % | (7) | ||||||||||
Process development | 1,847 | 101 | 1,223 | 1,728.7 | % | (91.7 | )% | (8) | ||||||||||
Other indirect R&D costs | 1,026 | 1,868 | 245 | (45.1 | )% | 662.4 | % | (9) | ||||||||||
Total indirect costs | 8,304 | 6,749 | 3,212 | 23.0 | % | 110.1 | % | |||||||||||
Total research and development expenses | $ | 25,892 | $ | 17,735 | $ | 13,762 | 46.0 | % | 28.9 | % | ||||||||
(1) | Costs to date on our azficel-T program for the treatment of vocal cord scarring are approximately $2.4 million as of December 31, 2015. These funds have been utilized to author and review clinical trial protocols, recruit investigator sites, recruit patients and execute our Phase I and II clinical trials. |
Cost increased approximately $0.4 million, or 47.7%, for the year ended December 31, 2015 as compared to 2014 and increased approximately $0.5 million, or 141.6%, for the year ended December 31, 2014 as compared to 2013 due to additional costs for patient enrollment, clinical site fees and clinical manufacturing costs related to our Phase II clinical trial which began enrollment in the second quarter of 2014.
Going forward, development investments for this program will support the completion of our Phase II clinical trial, for which we expect to report efficacy results in the second quarter of 2016, as well as clinical product manufacturing, statistical analyses, report generation, future clinical development and Phase III clinical trial costs.
(2) | Costs to date on our FCX-007 program are approximately $17.2 million as of December 31, 2015, and include $6.9 million of stock issuance costs incurred in connection with the 2012 ECC. Other costs to date for this program include costs for product and assay development, key opinion leader development, pre-clinical studies and manufacturing, design of the Phase I/II clinical trial protocol and IND enabling development work. |
Costs for our FCX-007 program increased approximately $0.8 million, or 20.3%, for the year ended December 31, 2015 compared to 2014 due to progression of FCX-007 pre-clinical development, specifically our animal studies and pre-clinical product manufacturing costs, as well as additional IND enabling development work performed related to the IND filed with the FDA in July 2015.
Costs for our FCX-007 program increased approximately $2.0 million, or 114.3%, for the year ended December 31, 2014 compared to 2013 due to the expansion of our pre-clinical development program and pre-clinical manufacturing costs.
Going forward, research and development investments for this program will support additional product and assay development, the additional toxicology-specific study and amended IND in response to the FDA's feedback from September 2015 and the execution of clinical trials, which we expect to initiate in the second quarter of 2016.
(3) | Costs to date on our FCX-013 program are approximately $9.2 million as of December 31, 2015, and include $6.4 million of stock issuance costs in connection with the 2013 amendment to the 2012 ECC. Other costs to date for this program include costs for product and assay development and pre-clinical work, including execution of our proof-of-concept study. |
50
Costs for our FCX-013 program increased approximately $1.0 million, or 176.2%, for the year ended December 31, 2015 compared to 2014 due to the advancement of our pre-clinical work, specifically for gene screening and selection, vector optimization as well as initiation of our proof-of-concept study.
Costs for our FCX-013 program decreased approximately $6.6 million, or 92.2%, for the year ended December 31, 2014 compared to 2013 primarily due to the $6.4 million nonrecurring up-front stock issuance costs incurred in 2013 as mentioned above.
Going forward, research and development investments for this program will support ongoing product and assay development, pre-clinical study execution, key opinion leader development, NIH RAC meeting preparation and design and execution of clinical trials. We achieved proof-of-concept data in the first quarter of 2016 and plan to submit an IND for FCX-013 to the FDA in 2017.
(4) | Costs to date on our arthritis program are approximately $10.2 million as of December 31, 2015, which includes a $10.0 million up-front technology access fee payable in connection with the 2015 ECC. See Part I, Item 1, "Business —Intrexon Collaborations" of this Form 10-K for additional details regarding our collaborations with Intrexon. |
Going forward, research and development investments for this program will support the establishment of program plans, gene candidate screening, product and assay development, proof-of-concept studies, pre-clinical studies and clinical trial execution.
(5) | Costs to date on our Ehlers-Danlos Syndrome (hypermobility type) program were approximately $5.2 million as of December 31, 2015, which represents the stock issuance costs in connection with the 2014 amendment to the 2012 ECC. This program was terminated in September 2015 due to technical hurdles. |
(6) | Costs for other direct programs are primarily related to validation and post-marketing monitoring studies for azficel-T for aesthetic indications, as well as costs incurred related to our azficel-T program for restrictive burn scarring ("RBS") program. Currently, we do not intend to advance this program. |
Total other direct costs decreased approximately $0.5 million, or 79.5%, for the year ended December 31, 2015 compared to 2014, primarily due to the reduction of efforts on our RBS program.
Total other direct costs decreased approximately $0.6 million, or 48.4%, for the year ended December 31, 2014 compared to 2013. The net decrease was primarily due to a decrease in costs for azficel-T of approximately $1.0 million. Comparable costs incurred for the year ended December 31, 2014 were internal costs, classified as process development in the table above, required to validate studies for azficel-T that had been performed by outside vendors during the year ended December 31, 2013. This decrease was offset by an increase in costs for our RBS program of approximately $0.3 million in the 2014 period due to additional costs for enrollment, clinical site fees and manufacturing costs related to our Phase II clinical trial.
(7) | Compensation and related expenses increased approximately $0.7 million, or 20.4%, for the year ended December 31, 2015 compared to 2014, primarily due to additional employees and increases in salaries, bonus and stock-based compensation. |
Compensation and related expenses increased approximately $3.0 million, or 1,106.3%, for the year ended December 31, 2014 compared to 2013, primarily due to a greater allocation of personnel costs to research and development previously captured in cost of product sales. The shift in allocation was a result of our strategic shift away from the aesthetic market (LAVIV) and an increase in clinical manufacturing.
(8) | Process development costs increased approximately $1.7 million, or 1,728.7%, for the year ended December 31, 2015 compared to 2014, primarily due to an increase in internal process development work as additional resources were directed towards optimizing our current manufacturing processes, benefiting several of our ongoing development programs. |
Process development costs decreased approximately $1.1 million, or 91.7%, for the year ended December 31, 2014 compared to 2013, primarily due to lower costs incurred with Intrexon related to process improvements for azficel-T. In both 2013 and 2014, there was minimal internal process development work performed.
(9) | Other indirect R&D costs decreased approximately $0.8 million, or 45.1%, for the year ended December 31, 2015 compared to 2014, primarily due to an increased focus on internal process development, resulting in a higher allocation of fixed overhead costs to process development manufacturing instead of other indirect R&D costs. |
51
Other indirect R&D costs increased approximately $1.6 million, or 662.4%, for the year ended December 31, 2014 compared to 2013, primarily due to the greater allocation of unabsorbed fixed overhead costs for our manufacturing facility to other indirect R&D costs as a result of our strategic shift away from the aesthetic market (LAVIV) and an increase in clinical manufacturing.
Selling, General and Administrative Expenses
Selling, general and administrative expenses were comprised of the following:
Year Ended December 31, | 2015 vs 2014 % Change | 2014 vs 2013 % Change | ||||||||||||||||
($ in thousands) | 2015 | 2014 | 2013 | |||||||||||||||
Compensation and related expenses | $ | 4,844 | $ | 3,976 | $ | 3,841 | 21.8 | % | 3.5 | % | (1) | |||||||
Professional fees | 3,569 | 3,073 | 1,956 | 16.1 | % | 57.1 | % | (2) | ||||||||||
Facilities and related expenses and other | 2,872 | 3,038 | 3,643 | (5.5 | )% | (16.6 | )% | (3) | ||||||||||
Total selling, general and administrative expenses | $ | 11,285 | $ | 10,087 | $ | 9,440 | 11.9 | % | 6.9 | % | ||||||||
(1) | Compensation and related expenses increased approximately $0.9 million, or 21.8%, for the year ended December 31, 2015 compared to 2014. The increase is primarily due to increases of approximately $0.3 million in salaries and wages (including benefits), $0.7 million in stock-based compensation expense and $0.1 million in bonuses, offset by decreases in severance costs of approximately $0.2 million. |
Compensation and related expenses increased approximately $0.1 million, or 3.5%, for the year ended December 31, 2014 compared to 2013. The increase is primarily due to increases of approximately $0.2 million in employee bonuses, $0.1 million in severance costs and $0.1 million in stock-based compensation expense, offset by a decrease of $0.3 million in salaries and wages (including benefits).
(2) | Professional fees increased approximately $0.5 million, or 16.1%, for the year ended December 31, 2015 compared to 2014. The increase is primarily due to an increase in legal fees of approximately $0.9 million related to litigation and contract negotiation costs. Increased legal fees were offset by a decrease in accounting and audit related fees of approximately $0.2 million in relation to the 2014 warrant restatement project and a decrease of $0.2 million in investor relations services. |
Professional fees increased approximately $1.1 million, or 57.1%, for the year ended December 31, 2014 compared to 2013. The increase is primarily due to an increase of approximately $0.7 million in accounting and audit related fees for our 2014 warrant restatement project, an increase in legal fees of approximately $0.4 million due to negotiations with respect to our corporate contracts and the warrant restatement project and an increase of $0.3 million in temporary staffing and consulting costs. These increases were offset by a decrease in marketing expense of approximately $0.3 million due to our strategic shift away from the aesthetic market (LAVIV).
(3) | Facilities and related expenses and other decreased approximately $0.2 million, or 5.5%, for the year ended December 31, 2015 compared to 2014. The decrease is primarily due to a $0.1 million decrease in state franchise taxes and a $0.2 million decrease in office and other general expenses, offset by a $0.1 million increase in rent and utilities expenses. |
Facilities and related expenses and other decreased approximately $0.6 million, or 16.6%, for the year ended December 31, 2014 compared to 2013. The decrease is primarily due to a $0.1 million decrease in corporate travel expenses, a $0.1 million decrease in depreciation, and $0.4 million decrease in office and other general expenses.
Warrant revaluation and other finance income (expense)
During the years ended December 31, 2015, 2014 and 2013, we recorded non-cash income of approximately $2.9 million, non-cash income of approximately $3.9 million and non-cash expense of approximately $1.1 million for warrant revaluation and other finance charges in our Consolidated Statements of Operations, respectively. Due to the nature and inputs of the model used to assess the fair value of our outstanding warrants, it is not abnormal to experience significant fluctuations from year to year. These fluctuations are due to a variety of factors including changes in our stock price and those of our peers, changes in the remaining contractual life of the warrants, and changes in management's estimated probability of certain events occurring that would impact the warrants.
52
Other Income
During the year ended December 31, 2014, we recorded approximately $0.4 million of other income, primarily in a settlement agreement with one of our suppliers. There was no such income during the years ended December 31, 2015 or 2013.
Net Loss
Net loss increased approximately $8.8 million to $34.5 million for the year ended December 31, 2015, as compared to $25.7 million for the year ended December 31, 2014. The increase is primarily due to an overall net increase in operating expenses of approximately $7.8 million, as more fully described at the component level above, and decreases in warrant revaluation income and other income of approximately $1.0 million and $0.4 million, respectively. This was offset by an increase in total revenues of approximately $0.3 million.
Net loss decreased approximately $5.9 million to $25.7 million for the year ended December 31, 2014, as compared to $31.6 million for the year ended December 31, 2013. The decrease is primarily due to the increase in warrant revaluation income of approximately $5.0 million, as more fully described above, an overall decrease in cost of product sales and operating expenses of approximately $0.6 million and an increase in other income of approximately $0.4 million.
Financial Condition, Liquidity and Capital Resources
Financial Condition
We have experienced losses since our inception. As of December 31, 2015, we had an accumulated deficit of $147.2 million. The process of developing and commercializing our product candidates requires significant research and development efforts and clinical trial work, as well as significant manufacturing and process development. These activities, together with our selling, general and administrative expenses, are expected to continue to result in significant operating losses for the foreseeable future.
Our financial condition is summarized as follows:
As of December 31, | Change | ||||||||||||
($ in thousands) | 2015 | 2014 | $ | % | |||||||||
Financial assets: | |||||||||||||
Cash and cash equivalents | $ | 29,268 | $ | 37,495 | $ | (8,227 | ) | (21.9 | )% | ||||
Working capital: | |||||||||||||
Total current assets | 30,994 | 39,349 | (8,355 | ) | (21.2 | )% | |||||||
Total current liabilities | (15,365 | ) | (3,493 | ) | (11,872 | ) | 339.9 | % | |||||
Net working capital | $ | 15,629 | $ | 35,856 | $ | (20,227 | ) | (56.4 | )% | ||||
Liquidity and Capital Resources
Our principal sources of liquidity are cash and cash equivalents of $29.3 million as of December 31, 2015. As of December 31, 2015, we had net working capital of $15.6 million. Net working capital decreased approximately $20.2 million, or 56.4%, from December 31, 2014 to December 31, 2015. This decrease is the result of a net consumption of cash described below and an increase in accounts payable for the $10 million up-front technology access fee associated with the 2015 ECC we entered into with Intrexon. We believe that our existing cash and cash equivalents will be sufficient to fund our operations into the fourth quarter of 2016, however, changing circumstances may cause us to consume capital faster than we currently anticipate, and we may need to spend more money than currently expected because of such circumstances. We will require additional capital to fund operations beyond that point. To meet our capital needs, we are considering multiple alternatives, including but not limited to, equity financings, debt financings, corporate collaborations, partnerships and other strategic transactions and funding opportunities. However, there can be no assurance that we will be able to complete any such transaction on acceptable terms or otherwise. These factors raise substantial doubt about our ability to continue as a going concern. As a result, our independent registered public accounting firm has included an explanatory paragraph in its audit report on our Consolidated Financial Statements for the year ended December 31, 2015 related to our ability to continue as a going concern.
53
Also, see Risks Related to Our Financial Position and Need for Additional Capital included within Part I, Item 1A, "Risk Factors" of this Form 10-K.
Future Funding Requirements
We will require additional capital in 2016 and prior to our business achieving significant net cash from operations. Our future capital requirements may be substantial, and will depend on many factors, including, but not limited to:
• | The cost of activities and outcomes related to our amended IND filing for FCX-007 which is expected in the second quarter of 2016 and pre-clinical activities and outcomes related to FCX-013, for which we expect to file an IND with the FDA in 2017; |
• | The timing and cost of our ongoing clinical program for azficel-T for the treatment of vocal cord scarring resulting in chronic or severe dysphonia; |
• | The cost of additional clinical trials in order to obtain regulatory approvals for our product candidates; |
• | The cost of regulatory submissions, as well as the preparation, initiation and execution of clinical trials in potential new clinical indications; |
• | The cost of maintaining an adequate and compliant manufacturing facility; and |
• | The cost of filing, surveillance around, prosecuting, defending and enforcing patent claims. |
If we raise additional funds by issuing equity securities, our stockholders will experience dilution. Debt financing, if available, will result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. Any debt financing or additional equity that we raise may contain terms, such as liquidation and other preferences, which are not favorable to us or our stockholders. If we raise additional funds through collaboration or partnership arrangements with third parties, it may be necessary to relinquish valuable rights to our technologies, future revenue streams or product candidates or to grant licenses on terms that may not be favorable to us.
If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we will need to curtail and reduce our operations and costs and modify our business strategy which may require us to, among other things:
• | significantly delay, scale back or discontinue the development or commercialization of one or more of our product candidates or one or more of our other research and development initiatives; |
• | seek collaborators for one or more of our current or future product candidates at an earlier stage than otherwise would be desirable or on terms that are less favorable than might otherwise be available; |
• | sell or license on unfavorable terms our rights to technologies or product candidates that we otherwise would seek to develop or commercialize ourselves; or |
• | seek bankruptcy protection which may result in the termination of agreements pursuant to which we license certain intellectual property rights including our exclusive collaboration agreements with Intrexon. |
Research and Development Activities
We plan to conduct further research and development in the next 12 months to support our ongoing development programs. We plan to conduct research and development activities with our own laboratory staff and also by engaging contract research organizations, clinical research organizations and contract manufacturing organizations. We also plan to incur costs for clinical and medical advisors and consulting and collaboration fees. We further anticipate that our research and development costs will increase during the next 12 months due to our arthritis program.
Selling, General and Administrative Activities
Selling, general and administrative costs in the next 12 months will consist primarily of legal, accounting, investor relations and other professional fees, board fees and expenses, insurance, rent and facility support expenses. We anticipate that selling, general and administrative expenses, primarily facilities and administrative expenses, will continue to increase to support our anticipated growth.
54
Capital Expenditures
In the next 12 months, we expect to increase our capital expenditures for laboratory and computer software as we maintain and improve upon our manufacturing facility.
Cash Flows
The following table summarizes our cash flow activity:
Year Ended December 31, | 2015 vs 2014 % Change | 2014 vs 2013 % Change | |||||||||||||||
($ in thousands) | 2015 | 2014 | 2013 | ||||||||||||||
Net cash flows provided by (used in): | |||||||||||||||||
Operating activities | $ | (24,106 | ) | $ | (22,296 | ) | $ | (20,075 | ) | 8.1 | % | 11.1 | % | ||||
Investing activities | $ | (245 | ) | $ | (242 | ) | $ | (360 | ) | 1.2 | % | (32.8 | )% | ||||
Financing activities | $ | 16,124 | $ | — | $ | 49,122 | 100.0 | % | (100.0 | )% | |||||||
Operating Activities. Cash used in operating activities during the year ended December 31, 2015 was $24.1 million, an increase of $1.8 million over the year ended December 31, 2014. Cash used in operating activities during the year ended December 31, 2014 was $22.3 million, an increase of $2.2 million over the year ended December 31, 2013. The increases in both 2015 and 2014 compared to the immediately preceding year were primarily due to increased spending with our partner Intrexon for the development of our product candidates FCX-007 and FCX-013.
Investing Activities. Cash used in investing activities, which was approximately $0.2 million for both the years ended December 31, 2015 and 2014 and $0.3 million for the year ended December 31, 2013, related primarily to the purchases of property and equipment.
Financing Activities. During the year ended December 31, 2015, we received net proceeds of approximately $15.9 million from the issuance of common stock related to our July 2015 financing. There were no financing activities for the year ended December 31, 2014. During the year ended December 31, 2013, we received net proceeds of $47.1 million from the issuance of common stock related to our October 2013 financing, and $2.0 million from a common stock subscription receivable.
Off-Balance Sheet Transactions
We do not engage in material off-balance sheet transactions.
Contractual Obligations
The following table summarizes our minimum contractual obligations as of December 31, 2015:
Payments due by period | |||||||||||||||||||||||||||
($ in thousands) | Total | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 and thereafter | ||||||||||||||||||||
License fee obligations (1) | $ | 48 | $ | 8 | $ | 8 | $ | 8 | $ | 8 | $ | 8 | $ | 8 | |||||||||||||
Research obligations (2) | 202 | 202 | — | — | — | — | — | ||||||||||||||||||||
Operating lease obligations (3) | 9,958 | 1,254 | 1,254 | 1,254 | 1,416 | 1,471 | 3,309 | ||||||||||||||||||||
Collaboration with related party (4) | 10,000 | 10,000 | — | — | — | — | — | ||||||||||||||||||||
Total | $ | 20,208 | $ | 11,464 | $ | 1,262 | $ | 1,262 | $ | 1,424 | $ | 1,479 | $ | 3,317 | |||||||||||||
(1) | Obligation for license agreement with UCLA. The amounts in the table assume the foregoing agreement is continued through its respective term. The agreement may be terminated at the option of either party. In such event, our obligation would be limited to costs through the date of such termination. |
(2) | Research obligations under the research agreement with UCLA stated are based upon budgeted project costs agreed to during the term of the agreement. |
(3) | Operating lease obligations are stated based on the lease agreement, as amended, for the office, warehouse and laboratory facilities in Exton, Pennsylvania, executed in February 2012. |
(4) | Up-front technology access fee pursuant to 2015 ECC with Intrexon. Paid in January 2016. |
55
Collaborations with Related Party
We are party to two separate exclusive channel collaboration agreements with Intrexon, a related party, pursuant to which we became Intrexon's exclusive channel collaborator in the research, development and commercialization of certain products as defined in the respective agreements. In connection with these exclusive channel collaboration agreements, the Company engages Intrexon for support services for the research and development of product candidates covered under the respective agreements and reimburses Intrexon for its cost for time and materials for such services.
For the years ended December 31, 2015, 2014 and 2013, the Company incurred expenses of $15.9 million, $9.4 million and $10.1 million, respectively, for payments to Intrexon. Of the costs incurred during the the years ended December 31, 2014 and 2013, $5.2 million and $6.4 million, respectively, related to non-cash supplemental stock issuance costs. As of December 31, 2015 and 2014, the Company had outstanding payables with Intrexon of $10.7 million and $1.0 million, respectively.
For additional details, see Part I, Item 1, "Business - Intrexon Collaborations," and Note 12, Related Party Transactions to the Consolidated Financial Statements, included as part of this Form 10-K.
Recently Issued Accounting Pronouncements
See Note 3 of the Consolidated Financial Statements for discussion on recently issued accounting pronouncements.
Item 7A. Quantitative and Qualitative Disclosure about Market Risk
We are exposed to market risk, primarily related to changes in interest rates impacting our cash and cash equivalents. Although multiple foreign subsidiaries are included in our Consolidated Financial Statements, operations at these entities are not material and there are currently no significant financial assets or liabilities denominated in a foreign currency in which exchange rate fluctuations would result in a material adverse impact to our results of operations, financial position, or cash flows.
Interest Rate Risk
The primary objective of our investment activities is to preserve our capital until it is required to fund operations. As
of December 31, 2015, our exposure to interest rate risk is confined to cash and cash equivalents of approximately $29.3 million, which include investments in U.S. money market funds and instruments having maturities of three months or less. None of these investments are entered into for trading purposes. Our cash flow and earnings are subject to immaterial fluctuations due to changes in interest rates in our investment portfolio, and we do not believe that a 10% change in average interest rates would have a significant impact on our interest income.
Item 8. Financial Statements and Supplementary Data
The information required by Item 8 including the financial statements and notes thereto, and report of the independent registered public accounting firm thereon, are included in this Form 10-K as set forth in the “Index to Consolidated Financial Statements” on page F 1.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
Not applicable.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Our management, including our principal executive officer and principal financial officer, has evaluated the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act), as of the end of the period covered by this Form 10-K. Based upon that evaluation, our Chief Executive Officer and Chief Financial Officer, concluded that, as of December 31, 2015, our disclosure controls and procedures were effective to provide reasonable assurance that (a) the information required to be disclosed by us in the reports that we file or submit under the Securities Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms, and (b) such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure.
56
In designing and evaluating our disclosure controls and procedures, our management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and our management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Management’s Report on Internal Control over Financial Reporting
Management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting, based on the framework in the Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission ("COSO 2013").
Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles in the United States of America. Our internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of our assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Based on our evaluation under the framework in COSO 2013, our management concluded that our internal control over financial reporting was effective as of December 31, 2015.
The effectiveness of our internal control over financial reporting as of December 31, 2015 has been audited by PricewaterhouseCoopers LLP, an independent registered public accounting firm, as stated in their report, which is included in this Annual Report on Form 10-K in the accompanying Consolidated Financial Statements.
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting during the quarter ended December 31, 2015 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
None.
Part III
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this item is incorporated by reference to our Proxy Statement for the 2016 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days of the fiscal year ended December 31, 2015.
Our Board of Directors has adopted a written Code of Conduct and Business Ethics applicable to all officers, directors and employees, which is available on our website (www.fibrocell.com) under "Corporate Governance" within the “Investors” section. We intend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding amendment to, or waiver from, a provision of this Code and by posting such information on the website address and location specified above.
57
Item 11. Executive Compensation
The information required by this item is incorporated by reference to our Proxy Statement for the 2016 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days of the fiscal year ended December 31, 2015.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this item is incorporated by reference to our Proxy Statement for the 2016 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days of the fiscal year ended December 31, 2015.
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this item is incorporated by reference to our Proxy Statement for the 2016 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days of the fiscal year ended December 31, 2015.
Item 14. Principal Accountant Fees and Services
The information required by this item is incorporated by reference to our Proxy Statement for the 2016 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days of the fiscal year ended December 31, 2015.
Part IV
Item 15. Exhibits and Financial Statement Schedule
(a) (1) Consolidated Financial Statements.
The Consolidated Financial Statements are filed as part of this report. See the index to the Consolidated Financial Statements on page F-1.
(2) Consolidated Financial Statement Schedule.
Schedules are omitted because they are not applicable, or are not required, or because the information is included in the Consolidated Financial Statements and notes thereto.
(3) The exhibits listed under Item 15(b), which are incorporated herein by reference, are filed or furnished as part of this report or are incorporated into this report by reference.
(b) Exhibits.
See the Exhibit Index immediately following the signature page of this Form 10-K.
58
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
FIBROCELL SCIENCE, INC. | ||
By: | /s/ David Pernock | |
David Pernock | ||
Chief Executive Officer | ||
Date: March 10, 2016 | ||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the date indicated.
Signature | Title | Date | ||
/s/ David Pernock | Chief Executive Officer and Chairman of the Board of Directors | March 10, 2016 | ||
David Pernock | (Principal Executive Officer) | |||
/s/ Keith A. Goldan | SVP and Chief Financial Officer | March 10, 2016 | ||
Keith A. Goldan | (Principal Financial and Accounting Officer) | |||
/s/ Kelvin Moore | Director | March 10, 2016 | ||
Kelvin Moore | ||||
/s/ Marc Mazur | Director | March 10, 2016 | ||
Marc Mazur | ||||
/s/ Julian Kirk | Director | March 10, 2016 | ||
Julian Kirk | ||||
/s/ Marcus Smith | Director | March 10, 2016 | ||
Marcus Smith | ||||
/s/ Christine St.Clare | Director | March 10, 2016 | ||
Christine St.Clare | ||||
/s/ Douglas J. Swirsky | Director | March 10, 2016 | ||
Douglas J. Swirsky | ||||
59
EXHIBIT INDEX
EXHIBIT NO. | IDENTIFICATION OF EXHIBIT | |
2.1 | Debtors’ First Amended Joint Plan of Reorganization dated July 30, 2009 and Disclosure Statement (incorporated by reference to as Exhibit 10.2 to the Company’s Form 10-Q for quarter ended June 30, 2009, filed on August 12, 2009 and as Exhibit 99.1 to our Form 8-K filed September 2, 2009) | |
3.1 | Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to our Form 8-K filed December 13, 2012) | |
3.2 | Certificate of Amendment of the Restated Certificate of Incorporation filed April 26, 2013 (incorporated by reference to Exhibit 3.1 of the Form 8-K filed on April 29, 2013) | |
3.3 | Certificate of Amendment to the Company’s Restated Certificate of Incorporation, as amended, filed July 19, 2013 (incorporated by reference to Exhibit 3.1 of the Form 8-K filed on July 22, 2013) | |
3.4 | Fourth Amended and Restated Bylaws (incorporated by reference to Exhibit 3.1 to our Form 10-Q filed on May 8, 2015) | |
3.5 | Amendment to Fourth Amended and Restated Bylaws (incorporated by reference to Exhibit 3.2 to our Form 10-Q filed on May 8, 2015) | |
4.1 | Form of Common Stock Certificate (incorporated by reference to Exhibit 4.1 to our Form 10-Q filed November 23, 2009) | |
4.2 | Form of Class A/B Common Stock Purchase Warrant issued in October 2009 offering (incorporated by reference to Exhibit 4.1 to our Form 8-K filed October 14, 2009) | |
4.3 | Form of Placement Agent Warrant issued in November 2009 offering (incorporated by reference to Exhibit 4.2 to our Form 10-Q filed November 23, 2009) | |
4.4 | Form of Common Stock Purchase Warrant issued in March 2010 offering (incorporated by reference to Exhibit 4.1 to our Form 8-K filed March 3, 2010) | |
4.5 | Form of Common Stock Purchase Warrant issued in July 2010 Series B Preferred Stock offering (incorporated by reference to Exhibit 4.1 to our Form 8-K filed July 20, 2010) | |
4.6 | Form of Placement Agent Warrant issued in July 2010 Series B Preferred Stock offering (incorporated by reference to Exhibit 4.2 to our Form 8-K filed July 20, 2010) | |
4.7 | Form of Common Stock Purchase Warrant used for Series B Preferred Stock offering (incorporated by reference to Exhibit 4.1 of the Form 8-K filed October 22, 2010). | |
4.8 | Form of Common Stock Purchase Warrant used for the Series D Preferred Stock offering (incorporated by reference to Exhibit 4.1 of the Form 8-K filed February 15, 2011). | |
4.9 | Form of Common Stock Purchase Warrant issued in August 2011 offering (incorporated by reference to Exhibit 4.1 to our Form 8-K filed August 4, 2011) | |
4.10 | Form of Amended and Restated Common Stock Purchase Warrant issued to our prior 12.5% Note holders (incorporated by reference to Exhibit 10.5 of the Form 8-K filed October 9, 2012). | |
10.1 | Lease Agreement between Isolagen, Inc. and The Hankin Group dated April 7, 2005 (incorporated by reference to Exhibit 10.1 to our Form 8-K filed on April 12, 2005) | |
10.2 | Amendment to Lease Agreement between Fibrocell Science, Inc. and The Hankin Group dated February 17, 2012 (incorporated by reference to Exhibit 10.17 of our Annual Report on Form 10-K for the fiscal year ended December 31, 2011) | |
10.3 | Securities Purchase Agreement dated October 5, 2012 (incorporated by reference to Exhibit 10.1 to our Form 8-K filed October 9, 2012) | |
10.4 | Registration Rights Agreement dated October 5, 2012 (incorporated by reference to Exhibit 10.2 to our Form 8-K filed October 5, 2012) | |
10.5 | Stock Issuance Agreement dated October 5, 2012 between the Company and Intrexon Corporation (incorporated by reference to Exhibit 10.3 to our Form 8-K filed October 5, 2012) | |
10.6 | Amendment and Conversion Agreement dated October 5, 2012 between the Company and the Holders of the Company’s Notes (incorporated by reference to Exhibit 10.4 to our Form 8-K filed October 5, 2012) | |
10.7 | Exclusive Channel Collaboration Agreement between Intrexon Corporation and Fibrocell Science, Inc. (incorporated by reference to Exhibit 10.21 of the Form 10-K filed on April 1, 2013) | |
10.8 | First Amendment to Exclusive Channel Collaboration Agreement between the Company and Intrexon Corporation dated June 28, 2013 (incorporated by reference to Exhibit 10.1 of the Form 8-K filed on July 1, 2013) | |
10.9 | Supplemental Stock Issuance Agreement between the Company and Intrexon Corporation dated June 28, 2013 (incorporated by reference to Exhibit 10.2 of the Form 8-K filed on July 1, 2013) | |
10.10 | Second Amendment to Exclusive Channel Collaboration Agreement between the Company and Intrexon Corporation dated January 10, 2014 (incorporated by reference to Exhibit 10.1 of the Form 8-K filed on January 13, 2014) | |
60
10.11 | Supplemental Stock Issuance Agreement between the Company and Intrexon Corporation dated January 10, 2014 (incorporated by reference to Exhibit 10.2 of the Form 8-K filed on January 13, 2014) | |
10.12 | Letter Agreement to Exclusive Channel Collaboration Agreement, as amended, between Fibrocell Science, Inc. and Intrexon Corporation, dated September 29, 2015 (incorporated by reference to Exhibit 10.2 to our Form 10-Q filed on November 5, 2015) | |
10.13 | U | Exclusive Channel Collaboration Agreement, dated December 31, 2015, between Fibrocell Science, Inc. and Intrexon Corporation (incorporated by reference to Exhibit 99.1 to our Form 8-K filed on January 4, 2016) |
10.14 | The Regents of the University of California Research Agreement (incorporated by reference to Exhibit 10.2 of the Form 10-Q filed on November 14, 2013) | |
10.15 | U | Exclusive License Agreement, dated June 1, 2014, by and between The Regents of the University of California and the Company (incorporated by reference to Exhibit 10.2 to our Quarterly Report on Form 10-Q/A filed on October 7, 2014) |
**10.16 | Fibrocell Science, Inc. 2009 Equity Incentive Plan, as amended (incorporated by reference to Exhibit 10.1 to our Current Report on Form 8-K filed on June 20, 2014) | |
10.17 | Form of Nonqualified Stock Option Agreement for Employee Grants under Fibrocell Science, Inc. 2009 Equity Incentive Plan (incorporated by reference to Exhibit 10.1 to our Form 10-Q filed on May 8, 2015 | |
10.18 | Form of Nonqualified Stock Option Agreement for Director Grants under Fibrocell Science, Inc. 2009 Equity Incentive Plan (incorporated by reference to Exhibit 10.2 to our Form 10-Q filed on May 8, 2015 | |
10.19 | Form of Incentive Stock Option Agreement for Employee Grants under Fibrocell Science, Inc. 2009 Equity Incentive Plan (incorporated by reference to Exhibit 10.3 to our Form 10-Q filed on May 8, 2015 | |
**10.20 | Amendment to Stock Option Agreement by and between the Company and David Pernock dated March 11, 2015 (incorporated by reference to Exhibit 10.22 to our Annual Report on Form 10-K filed on March 13, 2015) | |
**10.21 | Employment Agreement between the Company and David Pernock dated November 15, 2013 (incorporated by reference to Exhibit 10.1 to our Form 8-K filed November 18, 2013) | |
**10.22 | Employment Agreement between the Company and Keith A. Goldan dated March 18, 2015 (incorporated by reference to Exhibit 10.1 of the Form 8-K filed on March 18, 2015) | |
**10.23 | Employment Agreement between the Company and Michael F. Marino dated June 1, 2015 (incorporated by reference to Exhibit 10.1 of the Form 10-Q filed on August 7, 2015) | |
**10.24 | Employment Agreement between the Company and John Maslowski dated September 14, 2015 (incorporated by reference to Exhibit 10.1 of the Form 8-K filed on September 16, 2015) | |
*21 | List of Subsidiaries | |
*23.1 | Consent of PricewaterhouseCoopers LLP | |
*23.2 | Consent of BDO USA, LLP | |
*31.1 | Certification of Principal Executive Officer pursuant to Rule 13a-14(a) and 15d-14(a), required under Section 302 of the Sarbanes-Oxley Act of 2002 | |
*31.2 | Certification of Principal Financial Officer pursuant to Rule 13a-14(a) and 15d-14(a), required under Section 302 of the Sarbanes-Oxley Act of 2002 | |
*32.1 | Certification of Principal Executive Officer pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
*32.2 | Certification of Principal Financial Officer pursuant to 18 U.S.C. Section 1350 as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
101.INS | XBRL Instance Document | |
101.SCH | XBRL Taxonomy Extension Schema Document | |
101.CAL | XBRL Taxonomy Extension Calculation Linkbase Document | |
101.LAB | XBRL Taxonomy Extension Label Linkbase Document | |
101.PRE | XBRL Taxonomy Extension Presentation Linkbase Document | |
101.DEF | XBRL Taxonomy Extension Definition Linkbase Document | |
* | Filed herewith. |
** | Indicates management contract or compensatory plan or arrangement. |
U | Confidential treatment has been granted as to certain portions of this exhibit pursuant to Rule 406 of the Securities Act of 1933, as amended, or Rule 24b-2 of the Securities Exchange Act of 1934, as amended. |
61
Fibrocell Science, Inc.
Index to Consolidated Financial Statements
PAGE | |
F 1
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of
Fibrocell Science, Inc.
In our opinion, the accompanying consolidated balance sheet as of December 31, 2015 and the related consolidated statements of operations, stockholders’ equity, and cash flows for the year then ended present fairly, in all material respects, the financial position of Fibrocell Science, Inc. and its subsidiaries at December 31, 2015, and the results of their operations and their cash flows for the year then ended in conformity with accounting principles generally accepted in the United States of America. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2015, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The Company's management is responsible for these financial statements, for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in "Management’s Report on Internal Control over Financial Reporting” appearing under Item 9A. Our responsibility is to express opinions on these financial statements and on the Company's internal control over financial reporting based on our integrated audit. We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement and whether effective internal control over financial reporting was maintained in all material respects. Our audit of the financial statements included examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audit also included performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinions.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has suffered recurring losses from operations and has an accumulated deficit that raises substantial doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ PricewaterhouseCoopers LLP
Philadelphia, Pennsylvania
March 10, 2016
F 2
Report of Independent Registered Public Accounting Firm
Board of Directors and Stockholders
Fibrocell Science, Inc.
Exton, Pennsylvania
We have audited the accompanying consolidated balance sheet of Fibrocell Science, Inc. as of December 31, 2014 and the related consolidated statements of operations, stockholders’ equity, and cash flows for each of the two years in the period ended December 31, 2014. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Fibrocell Science, Inc. at December 31, 2014, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2014, in conformity with accounting principles generally accepted in the United States of America.
/s/ BDO USA, LLP
Philadelphia, Pennsylvania
March 13, 2015
F 3
Fibrocell Science, Inc.
Consolidated Balance Sheets
($ in thousands, except share data)
As of December 31, | |||||||
2015 | 2014 | ||||||
Assets | |||||||
Current assets: | |||||||
Cash and cash equivalents | $ | 29,268 | $ | 37,495 | |||
Accounts receivable, net of allowance for doubtful accounts of $12 and $17, respectively | — | 4 | |||||
Inventory | 482 | 571 | |||||
Prepaid expenses and other current assets | 1,244 | 1,279 | |||||
Total current assets | 30,994 | 39,349 | |||||
Property and equipment, net | 1,582 | 1,598 | |||||
Intangible assets, net of accumulated amortization of $2,204 and $1,653, respectively | 4,136 | 4,687 | |||||
Total assets | $ | 36,712 | $ | 45,634 | |||
Liabilities and Stockholders’ Equity | |||||||
Current liabilities: | |||||||
Accounts payable | $ | 499 | $ | 602 | |||
Related party payable | 10,720 | 1,001 | |||||
Accrued expenses | 1,779 | 1,196 | |||||
Deferred revenue | 457 | 416 | |||||
Warrant liability, current | 1,910 | 278 | |||||
Total current liabilities | 15,365 | 3,493 | |||||
Warrant liability, long term | 6,365 | 11,008 | |||||
Deferred rent | 779 | 724 | |||||
Total liabilities | 22,509 | 15,225 | |||||
Commitments and contingencies (Note 14) | |||||||
Stockholders’ equity: | |||||||
Preferred stock, $0.001 par value; 5,000,000 shares authorized; no shares outstanding | — | — | |||||
Common stock, $0.001 par value; 100,000,000 shares authorized; 43,898,785 and 40,856,815 shares issued and outstanding, respectively | 44 | 41 | |||||
Additional paid-in capital | 161,330 | 143,086 | |||||
Accumulated deficit | (147,171 | ) | (112,718 | ) | |||
Total stockholders’ equity | 14,203 | 30,409 | |||||
Total liabilities and stockholders’ equity | $ | 36,712 | $ | 45,634 | |||
The accompanying notes are an integral part of these consolidated financial statements.
F 4
Fibrocell Science, Inc.
Consolidated Statements of Operations
($ in thousands, except share and per share data)
Year Ended December 31, | |||||||||||
2015 | 2014 | 2013 | |||||||||
Revenue from product sales | $ | 270 | $ | 170 | $ | 200 | |||||
Collaboration revenue | 222 | 10 | — | ||||||||
Total revenue | 492 | 180 | 200 | ||||||||
Cost of product sales | 426 | 2,312 | 7,501 | ||||||||
Cost of collaboration revenue | 296 | — | — | ||||||||
Total cost of revenue | 722 | 2,312 | 7,501 | ||||||||
Gross loss | (230 | ) | (2,132 | ) | (7,301 | ) | |||||
Research and development expenses | 9,968 | 8,342 | 3,638 | ||||||||
Research and development expenses - related party | 15,924 | 9,393 | 10,124 | ||||||||
Selling, general and administrative expenses | 11,285 | 10,087 | 9,440 | ||||||||
Operating loss | (37,407 | ) | (29,954 | ) | (30,503 | ) | |||||
Other income (expense): | |||||||||||
Warrant revaluation and other finance income (expense) | 2,929 | 3,930 | (1,053 | ) | |||||||
Other income | 17 | 368 | — | ||||||||
Interest income | 8 | 6 | 2 | ||||||||
Loss before income taxes | (34,453 | ) | (25,650 | ) | (31,554 | ) | |||||
Income tax benefit | — | — | — | ||||||||
Net loss | $ | (34,453 | ) | $ | (25,650 | ) | $ | (31,554 | ) | ||
Per Share Information: | |||||||||||
Net loss | |||||||||||
— Basic | $ | (0.82 | ) | $ | (0.63 | ) | $ | (1.06 | ) | ||
— Diluted | $ | (0.85 | ) | $ | (0.70 | ) | $ | (1.12 | ) | ||
Weighted average number of common shares outstanding | |||||||||||
— Basic | 42,178,397 | 40,789,445 | 29,830,207 | ||||||||
— Diluted | 42,351,346 | 40,969,399 | 30,196,616 | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F 5
Fibrocell Science, Inc.
Consolidated Statements of Stockholders’ Equity
($ in thousands, except share data)
Common Stock | Subscription receivable | Additional paid-in capital | Accumulated deficit | Total Equity | ||||||||||||||||||
Shares | Amount | |||||||||||||||||||||
Balance, December 31, 2012 | 26,229,909 | $ | 26 | $ | (2,004 | ) | $ | 81,682 | $ | (55,514 | ) | $ | 24,190 | |||||||||
Proceeds from equity financing, net | 12,311,698 | 12 | — | 47,106 | — | 47,118 | ||||||||||||||||
Subscription received | — | — | 2,004 | — | — | 2,004 | ||||||||||||||||
Issuance of common stock - related party | 1,243,781 | 2 | — | 6,404 | — | 6,406 | ||||||||||||||||
Stock-based compensation expense | — | — | — | 1,150 | — | 1,150 | ||||||||||||||||
Exercise of warrants | 46,837 | — | — | 352 | — | 352 | ||||||||||||||||
Net loss | — | — | — | — | (31,554 | ) | (31,554 | ) | ||||||||||||||
Balance, December 31, 2013 | 39,832,225 | $ | 40 | $ | — | $ | 136,694 | $ | (87,068 | ) | $ | 49,666 | ||||||||||
Issuance of common stock - related party | 1,024,590 | 1 | — | 5,153 | — | 5,154 | ||||||||||||||||
Stock-based compensation expense | — | — | — | 1,239 | — | 1,239 | ||||||||||||||||
Net loss | — | — | — | — | (25,650 | ) | (25,650 | ) | ||||||||||||||
Balance, December 31, 2014 | 40,856,815 | $ | 41 | $ | — | $ | 143,086 | $ | (112,718 | ) | $ | 30,409 | ||||||||||
Proceeds from equity financing, net | 2,974,136 | 3 | — | 15,869 | — | 15,872 | ||||||||||||||||
Stock-based compensation expense | — | — | — | 2,038 | — | 2,038 | ||||||||||||||||
Exercise of stock options | 56,250 | — | — | 255 | — | 255 | ||||||||||||||||
Exercise of warrants | 11,584 | — | — | 82 | — | 82 | ||||||||||||||||
Net loss | — | — | — | — | (34,453 | ) | (34,453 | ) | ||||||||||||||
Balance, December 31, 2015 | 43,898,785 | $ | 44 | $ | — | $ | 161,330 | $ | (147,171 | ) | $ | 14,203 | ||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F 6
Fibrocell Science, Inc.
Consolidated Statements of Cash Flows
($ in thousands)
Year Ended December 31, 2015 | Year Ended December 31, 2014 | Year Ended December 31, 2013 | |||||||||
Cash flows from operating activities: | |||||||||||
Net loss | $ | (34,453 | ) | $ | (25,650 | ) | $ | (31,554 | ) | ||
Adjustments to reconcile net loss to net cash used in operating activities: | |||||||||||
Stock issued to related party for exclusive channel collaboration agreement | — | 5,154 | 6,406 | ||||||||
Stock-based compensation expense | 2,038 | 1,239 | 1,150 | ||||||||
Warrant revaluation and other finance (income) expense | (2,929 | ) | (3,930 | ) | 1,053 | ||||||
Deferred tax benefit | — | — | — | ||||||||
Loss on disposal of property and equipment | 56 | 13 | 5 | ||||||||
Depreciation and amortization | 767 | 883 | 863 | ||||||||
(Recovery of) provision for doubtful accounts | (5 | ) | 12 | (20 | ) | ||||||
Change in operating assets and liabilities: | |||||||||||
Accounts receivable | 9 | 12 | 54 | ||||||||
Inventory | 89 | 26 | (120 | ) | |||||||
Prepaid expenses and other current assets | 35 | (76 | ) | 69 | |||||||
Other assets | — | 214 | (215 | ) | |||||||
Accounts payable | (114 | ) | (1,065 | ) | 807 | ||||||
Related party payable | 9,719 | (290 | ) | 1,230 | |||||||
Accrued expenses and deferred rent | 641 | 894 | 188 | ||||||||
Deferred revenue | 41 | 268 | 9 | ||||||||
Net cash used in operating activities | (24,106 | ) | (22,296 | ) | (20,075 | ) | |||||
Cash flows from investing activities: | |||||||||||
Purchase of property and equipment | (271 | ) | (242 | ) | (360 | ) | |||||
Proceeds from the sale of property and equipment | 26 | — | — | ||||||||
Net cash used in investing activities | (245 | ) | (242 | ) | (360 | ) | |||||
Cash flows from financing activities: | |||||||||||
Proceeds from equity financing, net | 15,872 | — | 47,118 | ||||||||
Proceeds from the exercise of stock options | 255 | — | — | ||||||||
Subscription received | — | — | 2,004 | ||||||||
Principle payments on capital lease obligations | (3 | ) | — | — | |||||||
Net cash provided by financing activities | 16,124 | — | 49,122 | ||||||||
Net (decrease) increase in cash and cash equivalents | (8,227 | ) | (22,538 | ) | 28,687 | ||||||
Cash and cash equivalents, beginning of period | 37,495 | 60,033 | 31,346 | ||||||||
Cash and cash equivalents, end of period | $ | 29,268 | $ | 37,495 | $ | 60,033 | |||||
Supplemental disclosures of cash flow information: | |||||||||||
Non-cash investing and financing activities: | |||||||||||
Property and equipment in accounts payable | $ | 11 | $ | — | $ | — | |||||
Reduction of warrant liability upon cashless exercise of warrants | $ | 82 | $ | — | $ | 352 | |||||
The accompanying notes are an integral part of these consolidated financial statements.
F 7
Note 1. Business and Organization
Organization
Fibrocell Science, Inc. (as used herein, “we,” “us,” “our,” “Fibrocell” or the “Company”) is the parent company of Fibrocell Technologies, Inc. (“Fibrocell Tech”) and Fibrocell Science Hong Kong Limited (“Fibrocell Hong Kong”), a company organized under the laws of Hong Kong. Fibrocell Tech is the parent company of Isolagen International, S.A., a company organized under the laws of Switzerland (“Isolagen Switzerland”). In 2015, the Company's board of directors approved the formal dissolution of Isolagen Australia Pty Limited, a company organized under the laws of Australia (“Isolagen Australia”), and Isolagen Europe Limited, a company organized under the laws of the United Kingdom (“Isolagen Europe”), both of which were wholly owned subsidiaries of Fibrocell Tech. As these entities had previously ceased to operate, there was no impact to the Consolidated Financial Statements. The Company’s remaining international activities are currently immaterial.
Business Overview
The Company is an autologous cell and gene therapy company translating personalized biologics into medical breakthroughs. The Company is focused on discovering and developing therapies for the localized treatment of diseases affecting the skin, connective tissue and joints. All of the Company's product candidates incorporate its proprietary autologous fibroblast technology. Currently, all of the Company's research and development efforts focus on gaining regulatory approvals and commercialization of its product candidates in the United States.
Liquidity and Financial Condition
The Company expects to continue to incur losses and will require additional capital to advance its product candidates through development to commercialization. As of December 31, 2015, the Company had cash and cash equivalents of approximately $29.3 million and working capital of approximately $15.6 million. The Company believes that its cash and cash equivalents at December 31, 2015 will be sufficient to fund operations into the fourth quarter of 2016. The Company will require additional capital to fund operations beyond that point. To meet its capital needs, the Company intends to raise additional capital through debt or equity financings, collaborations, partnerships or other strategic transactions. However, there can be no assurance that the Company will be able to complete any such transaction on acceptable terms or otherwise. The failure of the Company to obtain sufficient funds on acceptable terms when needed could have a material adverse effect on the Company’s business, results of operations and financial condition. These conditions raise substantial doubt about its ability to continue as a going concern. Consequently, the audit report prepared by the Company’s independent registered public accounting firm relating to its Consolidated Financial Statements for the year ended December 31, 2015 includes a going concern explanatory paragraph.
Note 2. Basis of Presentation
General
The accompanying Consolidated Financial Statements have been prepared in accordance with U.S. generally accepted accounting principles (“GAAP”) and include the accounts of Fibrocell and its wholly owned subsidiaries. The accompanying Consolidated Financial Statements should be read in conjunction with the Notes to the Consolidated Financial Statements ("Notes"). All intercompany accounts and transactions have been eliminated in consolidation. The Company's foreign operations are immaterial and it has no unrealized gains or losses from the sale of investments. As a result, it does not have any items that would be classified as other comprehensive income in such a statement.
Reclassifications
The prior year financial statements contain certain reclassifications to the results of operations for the year ended December 31, 2014 to conform to the presentation for the year ended December 31, 2015 in this Form 10-K. Compensation and related expenses for manufacturing and facilities personnel of approximately $1.5 million were reclassified from selling, general and administrative expenses to research and development expenses for the year ended December 31, 2014. This reclassification was made in conjunction with the Company’s strategic shift away from the aesthetic market (LAVIV) and towards diseases affecting the skin, connective tissue and joints.
F 8
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 2. Basis of Presentation (continued)
On April 30, 2013, the Company completed a 25-for-1 reverse stock split. All common share and per share data included in these Consolidated Financial Statements reflect this reverse stock split.
Note 3. Summary of Significant Accounting Policies
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates, judgments, and assumptions that affect the reported amounts of assets, liabilities, equity, revenues and expenses, and related disclosure of contingencies in the accompanying Consolidated Financial Statements and Notes. In addition, management’s assessment of the Company’s ability to continue as a going concern involves the estimation of the amount and timing of future cash inflows and outflows. On an ongoing basis, the Company evaluates its estimates, judgments and methodologies. The Company bases its estimates on historical experience and on various other assumptions that are believed to be reasonable. Actual results may differ materially from those estimates.
Segment Information
The Company has determined that it operates in only one segment, as it only reports operational results on an aggregate basis to its chief operating decision maker. Additionally, all of the Company's revenues are derived from within, research development activities occur in, and assets are located in, the United States.
Cash and Cash Equivalents
The Company considers highly liquid investments with an original maturity of three months or less when purchased to be cash equivalents.
Concentration of Credit Risk
As of December 31, 2015, the Company maintains its operating cash with one major U.S. domestic bank and the remainder of its cash and cash equivalents as a money market fund with one major global bank. Federal insurance coverage on operating cash amounted to $250,000 per depositor at each financial institution, and the Company’s non-interest bearing cash balances may exceed federally insured limits. The terms of these deposits are on demand to minimize risk. The Company has not incurred losses related to these deposits.
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable are recorded at the invoiced amount, net of related cash discounts, and do not bear interest. The Company does not have any off-balance sheet exposure related to its customers. The Company maintains an allowance for doubtful accounts related to its accounts receivable that have been deemed to have a high risk of collectability. Management reviews its accounts receivable on a monthly basis to determine if any receivables may be uncollectible. Management analyzes historical collection trends and changes in its customer payment patterns, customer concentration and creditworthiness when evaluating the adequacy of its allowance for doubtful accounts. In its overall allowance for doubtful accounts, the Company includes any receivable balances that are determined to be uncollectible. Based on the information available, management believes the allowance for doubtful accounts is adequate; however, actual write-offs might exceed the recorded allowance.
The following table summarizes the changes in the allowance for doubtful accounts for the indicated periods:
Balance at beginning of year | Additions / deductions charged to earnings | Uncollectible receivables written off, net of recoveries | Balance at end of year | |||||||||||
December 31, 2015 | $ | 17 | (5 | ) | — | $ | 12 | |||||||
December 31, 2014 | $ | 5 | 14 | (2 | ) | $ | 17 | |||||||
December 31, 2013 | $ | 25 | (20 | ) | — | $ | 5 | |||||||
F 9
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 3. Summary of Significant Accounting Policies (continued)
Inventory
Inventories are determined at the lower of cost or market value, with cost determined under specific identification and on the first-in-first-out method. Inventories consist of raw materials and work-in-process. On a periodic basis, we analyze our inventory levels and reserve for inventory that is expected to expire prior to being sold, inventory that has a cost basis in excess of its expected net realizable value, inventory in excess of expected sales requirements, or inventory that fails to meet commercial sale specifications. Expired inventory is disposed of and the related costs are written off to the reserve for inventory obsolescence or charged against cost of product sales. Any remaining deferred revenue associated with such inventory is recognized at that time as the Company has fulfilled its obligations to the customer. The Company has determined that no inventory reserve was necessary at December 31, 2015 or December 31, 2014. See Note 4 for additional details.
Property and Equipment
Property and equipment is carried at acquisition cost less accumulated depreciation. Depreciation is computed on a straight-line basis over the estimated useful life of the asset. The cost of repairs and maintenance is charged to expense as incurred. In the first quarter of 2014, the Company adjusted its useful lives to more accurately reflect the expected consumption of the economic benefit of these assets as noted in the following table:
Property and equipment category | Useful life | |
Laboratory equipment | 6 years | |
Computer equipment and software | 3 years | |
Furniture and fixtures | 10 years | |
Leasehold improvements | Lesser of remaining lease term or life of asset | |
In accordance with Financial Accounting Standards Board (“FASB”) Accounting Standard Codification (“ASC”) ASC Topic 250, Accounting Changes and Error Corrections, the Company accounted for this change in useful lives as a change in estimate, with prospective application only. The impact of this change in estimate on depreciation expense was immaterial to the results in the Consolidated Statements of Operations.
Intangible Assets
Intangible assets are research and development assets related to the Company’s primary study on azficel-T that were capitalized on the balance sheet upon emergence from bankruptcy. The portion of the reorganization value which was attributed to identifiable intangible assets was $6.3 million. Azficel-T has three current or target indications: the Company's FDA-approved product LAVIV and a clinical development program for azficel-T for the treatment of vocal cord scarring resulting in chronic or severe dysphonia. Effective January 1, 2012, the Company launched LAVIV and as a result, the research and development intangible assets related to the Company’s primary study were considered to be finite-lived intangible assets and began amortizing over 12 years, the estimated useful life of the assets which is analogous with the exclusivity period granted to the Company under the BLA. For each of the years ended December 31, 2015, 2014 and 2013, amortization expense was approximately $0.6 million. The Company expects to amortize approximately $0.6 million for each of the next five years.
Finite-lived intangible assets are recorded at cost, net of accumulated amortization and, if applicable, impairment charges. Amortization of finite-lived intangible assets is provided over their estimated useful lives on a straight-line basis. The Company reviews the estimated remaining useful life of its intangible assets on an annual basis with any changes, if applicable, accounted for prospectively. In accordance with ASC 360-10-35, Impairment or Disposal of Long-Lived Assets, the Company reviews its finite-lived intangible assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. There was no impairment expense recognized for the years ended December 31, 2015, 2014 or 2013. Clinical trials and the development of biopharmaceutical products is a lengthy and complex process with significant uncertainty and risk. If development programs for azficel-T are not successful and the Company does not obtain regulatory approval, or there is a lack of commercial viability of the product(s), the Company's intangible assets may become impaired.
F 10
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 3. Summary of Significant Accounting Policies (continued)
Warrant Liability
The Company accounts for stock warrants as either equity instruments or derivative liabilities depending on the specific terms of the warrant agreement. Stock warrants are accounted for as a derivative in accordance with ASC 815, Derivatives and Hedging (“ASC 815”) if the stock warrants contain “down-round protection” or other terms that could potentially require “net cash settlement” and therefore, do not meet the scope exception for treatment as a derivative. Since “down-round protection” is not an input into the calculation of the fair value of the warrants, the warrants cannot be considered indexed to the Company’s own stock which is a requirement for the scope exception as outlined under ASC 815. Warrant instruments that could potentially require “net cash settlement” in the absence of express language precluding such settlement and those which include “down-round provisions” are initially classified as derivative liabilities at their estimated fair values, regardless of the likelihood that such instruments will ever be settled in cash. The Company will continue to classify the fair value of the warrants that contain “down-round protection” and “net cash settlement” as a liability until the warrants are exercised, expire or are amended in a way that would no longer require these warrants to be classified as a liability. For additional discussion on warrants, see Note 7.
Revenue Recognition
Revenue from Product Sales. In June 2011, the FDA approved the Company's BLA for LAVIV for the improvement of the appearance of moderate to severe nasolabial fold wrinkles in adults. The Company recognizes revenue from product sales in accordance with ASC 605, Revenue Recognition (“ASC 605”). In general, ASC 605 requires that four basic criteria must be met before revenue can be recognized: (1) persuasive evidence of an arrangement exists, (2) delivery has occurred or services rendered, (3) the fee is fixed and determinable and (4) collectability is reasonably assured.
In general, prepayments are received at three different stages of the treatment: (1) the biopsy stage, (2) the cell harvest stage, and (3) the injection stage. As one full course of LAVIV therapy includes three series of injections, prepayments are deferred and revenue is recognized on a prorata basis as each of the three series of injections is shipped to the physician. Currently, the Company no longer actively markets or promotes LAVIV to physicians or patients; however, prescriptions are still being accepted and filled.
Collaboration Revenue. The Company follows ASC 605-25, Revenue Recognition – Multiple-Element Arrangements ("ASC 605-25") and ASC 808, Collaborative Arrangements, if applicable, to determine the recognition of revenue under its collaborative research, development and commercialization agreements. The terms of these agreements generally contain multiple elements, or deliverables, which may include (i) grants of licenses, or options to obtain licenses, to our intellectual property, (ii) research and development services, (iii) clinical and commercial manufacturing, and/or (iv) participation on joint research and/or joint development committees. The payments the Company may receive under these arrangements typically include one or more of the following: non-refundable, up-front license fees; funding of research and/or development efforts including proof-of-concept studies and product development; amounts due upon the achievement of specified objectives or milestones such as obtaining patents, trademarks and certain regulatory approvals, and achievement of commercialization of products; and/or royalties on future product sales.
Each of the required deliverables under such an arrangement are evaluated, in accordance with ASC 605-25, to determine whether it qualifies as a separate unit of accounting based on whether the deliverable has “stand-alone value” to the customer. The arrangement’s consideration that is fixed or determinable is then allocated to each separate unit of accounting based on the relative selling price of each deliverable. In general, the consideration allocated to each unit of accounting is recognized as the related goods or services are delivered, limited to the consideration that is not contingent upon future deliverables. Collaboration revenue is recognized on a gross basis, in accordance with the criteria set forth in ASC 605-45, Revenue Recognition: Principal Agent Considerations.
Collaboration revenue for the years ended December 31, 2015 and 2014 is related to a research and development agreement that the Company has with a third party to investigate potential new non-pharmaceutical applications for the Company's conditioned fibroblast media technology. Revenue recognized to date from this collaboration relates to an upfront license fee that is being amortized over the estimated contract period and a proof-of-concept study which was completed as of December 31, 2015. Deferred revenue related to this collaboration as of December 31, 2015 includes the unamortized balance of the upfront license fee. There was no such collaboration revenue for the year ended December 31, 2013.
F 11
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 3. Summary of Significant Accounting Policies (continued)
The Company will recognize future milestone payments when earned provided that (1) the milestone event is substantive in that it can only be achieved based in whole or in part on either the Company’s performance or on the occurrence of a specific outcome resulting from the Company’s performance and its achievability was not reasonably assured at the inception of the agreement; (2) the Company does not have ongoing performance obligations related to the achievement of the milestone; and (3) it would result in the receipt of additional payments. A milestone payment is considered substantive if all of the following conditions are met: (a) the milestone payment is non-refundable; (b) achievement of the milestone was not reasonably assured at the inception of the arrangement; (c) substantive effort is involved to achieve the milestone; and (d) the amount of the milestone payment appears reasonable in relation to the effort expended, the other milestones in the arrangement and the related risk associated with the achievement of the milestone.
Cost of Revenue
Cost of revenue includes expenses related to revenue from product sales and collaboration revenue.
Cost of Product Sales. Costs include the expense to manufacture LAVIV, including direct and indirect costs. Beginning in 2014, cost of product sales is comprised of allocated costs associated with the Company’s manufacturing, facility, quality control, and quality assurance operations as well as overhead costs. The principal reason for the relatively small level of revenue as compared to the cost of product sales is that in late 2013 the Company shifted its strategic focus away from the aesthetic market (LAVIV) and towards developing treatments for diseases affecting the skin, connective tissue and joints. Costs incurred for shipping and handling during the biopsy stage (to/from physicians) are included in cost of product sales. Costs related to shipping and handling of injections (to physicians) are included in selling, general and administrative expenses.
Cost of Collaboration Revenue. Costs directly related to deliverables in a revenue-generating collaboration are
charged to cost of collaboration revenue as incurred.
Research and Development Expenses
Research and development costs are expensed as incurred and include employee salaries and benefits, costs incurred with third party contractors to perform research, conduct clinical trials, develop and manufacture drug materials and delivery devices, and a portion of facilities costs. Research and development expenses also include costs to manufacture product for clinical trial use and to develop manufacturing, cell collection and logistical process improvements.
Clinical trial costs are a significant component of research and development expenses, often with third party service providers. Invoicing from third party contractors for services performed can lag several months. The Company accrues the costs of services rendered in connection with third party contractor activities based on its estimate of management fees, site management and monitoring costs and data management costs incurred in a given period.
Stock-Based Compensation
The Company follows ASC 718, Compensation – Stock Compensation, or ASC 505-50, Equity – Equity Based Payments to Non-Employees, where applicable. The Company accounts for stock-based awards to employees using the fair value based method to determine compensation for all arrangements where shares of stock or equity instruments are issued for compensation. In addition, the Company accounts for stock-based compensation to non-employees in accordance with the accounting guidance for equity instruments that are issued to other than employees. The Company uses a Black-Scholes option-pricing model to determine the fair value of each option grant as of the date of grant for expense incurred. The Black-Scholes model requires inputs for risk-free interest rate, dividend yield, expected stock price volatility and expected life of the options. The value of the award that is ultimately expected to vest is recognized as expense on a straight-line basis over the requisite service period. See Note 10 for additional details.
Income Taxes
An asset and liability approach is used for financial accounting and reporting for income taxes. Deferred income taxes arise from temporary differences between income tax and financial reporting and principally relate to recognition of revenue and expenses in different periods for financial and tax accounting purposes and are measured using currently enacted tax rates and laws. In addition, a deferred tax asset can be generated by a net operating loss carryover. If it is more likely than not that some portion or all of a deferred tax asset will not be realized, a valuation allowance is recognized.
F 12
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 3. Summary of Significant Accounting Policies (continued)
In the event the Company is charged interest or penalties related to income tax matters, the Company would record such interest as interest expense and would record such penalties as other expense in the Consolidated Statements of Operations. No such charges have been incurred by the Company. For each of the years ended December 31, 2015, 2014 and 2013, the Company had no uncertain tax positions. See Note 11 for additional details.
Loss Per Share Data
Basic loss per share is computed by dividing net loss for the period by the weighted average number of shares of common stock outstanding during that period. The diluted loss per share calculation gives effect to dilutive stock options, warrants and other potentially dilutive common stock equivalents outstanding during the period. Diluted loss per share is based on the treasury stock method and includes the effect from the potential issuance of common stock, such as shares issuable pursuant to the exercise of stock options and warrants, assuming the exercise of all "in-the-money" stock options and warrants based on the average market price during the period. Common stock equivalents have been excluded where their inclusion would be anti-dilutive. See Note 13 for additional details.
Fair Value of Financial Instruments
The carrying values of certain of the Company’s financial instruments, including cash and cash equivalents and accounts payable, approximate fair value due to their short maturities. Warrant liability is also recorded at fair value. The fair values of the Company’s long term obligations are based on assumptions concerning the amount and timing of estimated future cash flows and assume discount rates reflecting varying degrees of risk. The carrying values of the Company’s long term obligations approximate their fair values. See Note 9 for additional details.
Recently Issued Accounting Pronouncements
In April 2015, the FASB issued ASU 2015-05, Customer's Accounting for Fees Paid in a Cloud Computing Arrangement ("ASU 2015-05"), which provides additional guidance to customers about whether a cloud computing arrangement includes a software license. Under ASU 2015-05, if a software cloud computing arrangement contains a software license, customers should account for the license element of the arrangement in a manner consistent with the acquisition of other software licenses. If the arrangement does not contain a software license, customers should account for the arrangement as a service contract. ASU 2015-05 also removes the requirement to analogize to ASC 840-10, Leases, to determine the asset acquired in a software licensing arrangement. An entity can elect to adopt the amendments either prospectively to all arrangements entered into or materially modified after the effective date or retrospectively. The guidance is effective for public companies with annual reporting periods beginning after December 15, 2015, including interim periods within that reporting period. Earlier application is permitted. The Company does not believe the impact of adoption of ASU 2015-05 to be material to its Consolidated Financial Statements.
In July 2015, the FASB issued ASU 2015-11, Inventory (Topic 330): Simplifying the Measurement of Inventory, which requires an entity to measure in scope inventory at the lower of cost and net realizable value rather than at the lower of cost or market, where market could be replacement cost, net realizable value, or net realizable value less an approximately normal profit margin. The amendments do not apply to inventory that is measured using last-in, first-out (LIFO) or the retail inventory method. The amendments apply to all other inventory, which includes inventory that is measured using first-in, first-out (FIFO) or average cost. The guidance is effective for public companies with annual reporting periods beginning after December 15, 2016, including interim periods within that reporting period, and should be applied prospectively. Earlier application is permitted. The Company does not believe the impact of adoption of ASU 2015-11 to be material to its Consolidated Financial Statements.
In August 2015, the FASB issued ASU 2015-14, Revenue from Contracts with Customers (Topic 606): Deferral of the Effective Date, which defers by one year the effective date of ASU 2014-09, Revenue from Contracts with Customers. The guidance is effective for public companies with annual reporting periods beginning after December 15, 2017, including interim periods within that reporting period. Earlier application is permitted. The Company is currently evaluating the effects of the adoption of ASU 2014-09 deferred by this update on its Consolidated Financial Statements but does not believe the impact to be material.
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which is intended to increase transparency and comparability among organizations by recognizing lease assets and lease liabilities on the balance sheet (including by lessees
F 13
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 3. Summary of Significant Accounting Policies (continued)
for those leases classified as operating leases under previous GAAP) and disclosing key information about leasing arrangements. The guidance is effective for public companies with annual reporting periods beginning after December 15, 2018, including interim periods within that reporting period. Earlier application is permitted. The Company believes the proposed ASU would increase its total assets and total liabilities on its Consolidated Balance Sheet as a result of capitalizing the current operating lease, as amended, for the Exton, PA facility. The Company does not anticipate any other impacts to its Consolidated Financial Statements.
Management does not believe that any other recently issued, but not yet effective, accounting pronouncements, if adopted, are applicable to the Company or would have a material impact on its Consolidated Financial Statements.
Recently Adopted Accounting Pronouncements
In November 2015, the FASB issued ASU 2015-17, Income Taxes (Topic 740): Balance Sheet Classification of Deferred Taxes ("ASU 2015-17"), which simplifies the reporting requirements of deferred taxes by requiring all organizations to classify all deferred tax assets and liabilities, along with any related valuation allowance, as noncurrent. The guidance is effective for public companies with annual reporting periods beginning after December 15, 2016, including interim periods within that reporting period. Early application is permitted. The Company has elected to early adopt ASU 2015-17 as of December 31, 2015, with retrospective application. The adoption of this ASU had no impact on the Company's Consolidated Financial Statements in the current year or previously reported periods because of the Company's full valuation allowance.
Note 4. Inventory
Inventories consist of raw materials and work-in-process intended for use in the manufacture of LAVIV. Raw materials may be used for clinical trials and are charged to research and development expense when consumed.
Inventories consisted of the following as of:
($ in thousands) | December 31, 2015 | December 31, 2014 | |||||
Raw materials (LAVIV and product candidates) | $ | 338 | $ | 357 | |||
Work-in-process (LAVIV) | 144 | 214 | |||||
Total inventory | $ | 482 | $ | 571 | |||
The Company determined that no reserve for expiration was needed at December 31, 2015 or December 31, 2014. No inventory expiration/obsolescence expense was recognized during the years ended December 31, 2015, 2014 or 2013.
Note 5. Property and Equipment
Property and equipment consisted of the following as of:
($ in thousands) | December 31, 2015 | December 31, 2014 | |||||
Laboratory equipment | $ | 1,416 | $ | 1,279 | |||
Computer equipment and software | 296 | 206 | |||||
Furniture and fixtures | 53 | 49 | |||||
Leasehold improvements | 903 | 772 | |||||
Construction-in-process | 156 | 343 | |||||
Total property and equipment, gross | 2,824 | 2,649 | |||||
Less: Accumulated depreciation | (1,242 | ) | (1,051 | ) | |||
Total property and equipment, net | $ | 1,582 | $ | 1,598 | |||
Depreciation expense was approximately $0.2 million, $0.3 million and $0.3 million for the years ended December 31, 2015, 2014 and 2013, respectively.
F 14
Note 6. Accrued Expenses
Accrued expenses consisted of the following as of:
($ in thousands) | December 31, 2015 | December 31, 2014 | |||||
Accrued professional fees | $ | 824 | $ | 402 | |||
Accrued compensation | 755 | 540 | |||||
Accrued other | 200 | 254 | |||||
Total accrued expenses | $ | 1,779 | $ | 1,196 | |||
Note 7. Warrants
The Company accounts for stock warrants as either equity instruments or derivative liabilities depending on the specific terms of the warrant agreement. See Note 3 for further details on accounting policies related to the Company's stock warrants. As of December 31, 2015 and 2014, all of the Company's outstanding stock warrants were classified as derivative liabilities.
Liability-classified Warrants
The following table summarizes outstanding liability-classified warrants to purchase common stock as of:
Number of Warrants | |||||||||||
December 31, 2015 | December 31, 2014 | Exercise Price | Expiration Dates | ||||||||
Issued in Series A, B and D Preferred Stock offerings (1) | 1,970,594 | 2,247,118 | $ | 6.25 | Oct 2015 - Dec 2016 | ||||||
Issued in March 2010 financing | 319,789 | 393,416 | $ | 6.25 | Mar 2016 | ||||||
Issued in June 2011 financing | 6,113 | 6,113 | $ | 22.50 | Jun 2016 | ||||||
Issued in August 2011 financing | 565,759 | 565,759 | $ | 18.75 | Aug 2016 | ||||||
Issued to placement agents in August 2011 financing | 50,123 | 50,123 | $ | 13.635 | Aug 2016 | ||||||
Issued in Series B, D and E Preferred Stock offerings (2) | 60,000 | 76,120 | $ | 2.50 | Nov 2015 - Dec 2017 | ||||||
Issued with Convertible Notes | 1,125,578 | 1,125,578 | $ | 2.50 | Jun 2018 | ||||||
Issued in Series E Preferred Stock offering | 1,568,823 | 1,568,823 | $ | 7.50 | Dec 2018 | ||||||
Total | 5,666,779 | 6,033,050 | |||||||||
(1) | As of December 31, 2015, all Series A warrants had expired. |
(2) | As of December 31, 2015, the Series B and D warrants in this group had either been exercised or expired. |
The table below is a summary of the Company's warrant activity for the year ended December 31, 2015.
Number of warrants | Weighted average exercise price | |||||
Outstanding at December 31, 2014 | 6,033,050 | $ | 7.08 | |||
Granted | — | — | ||||
Exercised (1) | (90,947 | ) | 6.20 | |||
Expired | (275,324 | ) | 6.05 | |||
Outstanding at December 31, 2015 | 5,666,779 | $ | 7.14 | |||
(1) | All warrants were exercised on a cashless basis resulting in the issuance of 11,584 shares of the Company's common stock. |
F 15
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 7. Warrants (continued)
The foregoing warrants are recorded as liabilities at their estimated fair value at the date of issuance, with subsequent changes in estimated fair value recorded in "Warrant revaluation and other finance income (expense)" in the Company’s Consolidated Statement of Operations in each subsequent period. The change in estimated fair value of the Company's warrant liability for the years ended December 31, 2015, 2014 and 2013 resulted in non-cash income of $2.9 million, non-cash income of $3.9 million, and non-cash expense of $1.1 million, respectively. The Company utilizes the Monte Carlo simulation valuation method to value its liability-classified warrants.
The estimated fair value of these warrants is determined using Level 3 inputs. Inherent in the Monte Carlo valuation model are assumptions related to expected stock-price volatility, expected life, risk-free interest rate and dividend yield. The Company estimates the volatility of its common stock based on the median of peer group historical volatility for a period that matches the weighted average of the expected remaining life of the warrants. The risk-free interest rate is based on the U.S. Treasury zero-coupon yield curve on the grant date for a maturity similar to the weighted average of the expected remaining life of the warrants. The expected life of the warrants is assumed to be equivalent to their remaining contractual term. The dividend rate is based on the historical rate, which the Company anticipates to remain at zero.
The following table summarizes the calculated aggregate fair values, along with the assumptions utilized in each calculation:
($ in thousands, except per share data) | December 31, 2015 | December 31, 2014 | December 31, 2013 | ||||||||
Calculated aggregate value | $ | 8,275 | $ | 11,286 | $ | 15,216 | |||||
Weighted average exercise price per share of warrant | $ | 7.14 | $ | 7.08 | $ | 7.08 | |||||
Closing price per share of common stock | $ | 4.55 | $ | 2.59 | $ | 4.06 | |||||
Volatility | 85.2 | % | 67.6 | % | 70.0 | % | |||||
Weighted average remaining expected life | 1 year, 8 months | 2 years, 7 months | 3 years, 6 months | ||||||||
Risk-free interest rate | 0.98 | % | 0.86 | % | 1.20 | % | |||||
Dividend yield | — | % | — | % | — | % | |||||
Note 8. Equity
Common Stock - Subscription Receivable
In July of 2013, the Company collected a subscription receivable in the amount of approximately $2.0 million, related to its October 2012 private placement offering.
Common Stock - Stock Issuance Agreements with Intrexon
In connection with the execution of the first amendment to the 2012 exclusive channel collaboration agreement (the “Amendment”) on June 28, 2013 between the Company and Intrexon Corporation ("Intrexon"), the Company entered into a supplemental stock issuance agreement with Intrexon. The Company agreed to issue to Intrexon a number of shares of the Company's common stock based on a per share value of the closing price of the Company’s common stock on the NYSE MKT on the day prior to execution of the supplemental stock issuance agreement (the “Supplemental Access Fee Shares”). The Supplemental Access Fee Shares were issued upon the satisfaction of customary closing conditions, including the approval for the listing of the Supplemental Access Fee Shares on the NYSE MKT. The closing took place on July 26, 2013. The Company recorded a fair value of $6.4 million for 1,243,781 shares, on a per share value of $5.15 based on the closing price of the Company’s common stock on the closing date, issued to Intrexon and recorded as a research and development expense in the third quarter of 2013. See Note 12 for further discussion on the collaboration with Intrexon.
In connection with the execution of the second amendment to the 2012 exclusive channel collaboration agreement (the “Second Amendment”) on January 10, 2014 between the Company and Intrexon, the Company entered into a supplemental stock issuance agreement with Intrexon. The Company agreed to issue to Intrexon a number of shares of the Company's common stock based on a per share value of the closing price of the Company’s common stock on the NYSE MKT on the day prior to execution of the supplemental stock issuance agreement (the “Second Supplemental Access Fee Shares”). The Second Supplemental Access Fee Shares were issued upon the satisfaction of customary closing conditions, including the approval for the listing of the Second Supplemental Access Fee Shares on the NYSE MKT. The closing took place on January 24, 2014. The Company recorded a fair value of $5.2 million for 1,024,590 shares, on a per share value of $5.03 based on the closing
F 16
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 8. Equity (continued)
price of the Company’s common stock on the closing date, issued to Intrexon and recorded as a research and development expense in the first quarter of 2014. See Note 12 for further discussion on the collaboration with Intrexon.
Common Stock - 2013 Follow-on Public Offering
On October 1, 2013, the Company completed an underwritten public offering of 11,000,000 shares of common stock at a public offering price of $4.10 per share. The net proceeds to the Company, after underwriting discounts and commissions and estimated offering expenses, were approximately $42.1 million. The underwriters for the public offering of common stock partially exercised their over-allotment option to purchase an additional 1,311,698 shares of common stock at a public offering price of $4.10 per share. The partial exercise of the over-allotment option increased the aggregate net proceeds to the Company, after underwriting discounts and commissions and estimated offering expenses, from approximately $42.1 million to approximately $47.1 million.
Common Stock - 2015 Follow-on Public Offering
On July 27, 2015, the Company completed an underwritten public offering of shares of the Company's common stock at a price per share of $5.80 per share (the “2015 Offering”). The shares sold in the 2015 Offering included 2,586,206 shares of common stock plus an additional 387,930 shares of common stock pursuant to the exercise by the underwriters of the over-allotment option the Company granted to them. Total gross proceeds to the Company in the 2015 Offering (including the sale of shares of common stock pursuant to the exercise of the over-allotment option) totaled $17.3 million, and resulted in net proceeds of approximately $15.9 million after the deduction of underwriting discounts and other offering expenses.
Preferred Stock
The Company is authorized to issue 5,000,000 shares of preferred stock in one or more series and to fix the rights, preferences, privileges, and restrictions thereof. These rights, preferences and privileges could include dividend rights, conversion rights, voting rights, terms of redemption, liquidation preferences, sinking fund terms and the number of shares constituting any series or the designation of such series, any or all of which may be greater than the rights of common stock. The issuance of the Company’s preferred stock could adversely affect the voting power of holders of common stock and the likelihood that such holders will receive dividend payments and payments upon liquidation. In addition, the issuance of preferred stock could have the effect of delaying, deferring or preventing a change of control of the Company or other corporate action. There were no preferred shares issued or outstanding as of December 31, 2015 or December 31, 2014.
Note 9. Fair Value Measurements
Assets and Liabilities Measured at Fair Value on a Recurring Basis
The Company uses the accounting guidance included in ASC 820, Fair Value Measurement, on fair value measurements for financial assets and liabilities measured on a recurring basis. Fair value is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. The Company uses a fair value hierarchy, which distinguishes between assumptions based on market data (observable inputs) and an entity's own assumptions (unobservable inputs). The guidance requires fair value measurements be classified and disclosed in one of the following three categories:
• | Level 1: Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities; |
• | Level 2: Quoted prices in markets that are not active or inputs which are observable, either directly or indirectly, for substantially the full term of the asset or liability; |
• | Level 3: Prices or valuation techniques that require inputs that are both significant to the fair value measurement and unobservable (i.e., supported by little or no market activity). |
Determining which category an asset or liability falls within the hierarchy requires significant judgment. The Company evaluates its hierarchy disclosures each reporting period.
F 17
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 9. Fair Value Measurements (continued)
The following fair value hierarchy table presents information about each major category of the Company’s financial assets and liabilities measured at fair value on a recurring basis as of December 31, 2015 and 2014:
December 31, 2015 | |||||||||||||||
($ in thousands) | Level 1 | Level 2 | Level 3 | Total | |||||||||||
Assets: | |||||||||||||||
Cash and cash equivalents | $ | 29,268 | $ | — | $ | — | $ | 29,268 | |||||||
Liabilities: | |||||||||||||||
Warrant liability, current and long term | $ | — | $ | — | $ | 8,275 | $ | 8,275 | |||||||
December 31, 2014 | |||||||||||||||
($ in thousands) | Level 1 | Level 2 | Level 3 | Total | |||||||||||
Assets: | |||||||||||||||
Cash and cash equivalents | $ | 37,495 | $ | — | $ | — | $ | 37,495 | |||||||
Liabilities: | |||||||||||||||
Warrant liability, current and long term | $ | — | $ | — | $ | 11,286 | $ | 11,286 | |||||||
Changes in Level 3 Liabilities Measured at Fair Value on a Recurring Basis - Common Stock Warrants
The reconciliation of the Company's warrant liability measured at fair value on a recurring basis using unobservable inputs (Level 3) is as follows:
($ in thousands) | Warrant Liability | ||
Balance at December 31, 2012 | $ | 14,515 | |
Exercise of warrants | (352 | ) | |
Cancellation of warrants | (41 | ) | |
Change in fair value of warrant liability | 1,094 | ||
Balance at December 31, 2013 | $ | 15,216 | |
Change in fair value of warrant liability | (3,930 | ) | |
Balance at December 31, 2014 | $ | 11,286 | |
Exercise of warrants (1) | (82 | ) | |
Expiration of warrants (2) | (276 | ) | |
Change in fair value of warrant liability | (2,653 | ) | |
Balance at December 31, 2015 | $ | 8,275 | |
(1) | Warrants were exercised under the cashless exercise method pursuant to the corresponding warrant agreements. As a result of such exercises, the Company issued 11,584 shares of common stock. Consequently, these instruments were no longer classified as liabilities. These common stock warrants were remeasured to their fair value as of the exercise date with the change in fair value recorded to the Consolidated Statement of Operations. The fair value related to the shares issued in connection with the exercised warrants was reclassified from a liability to additional paid-in capital in the Consolidated Balance Sheets. |
(2) | Represents the fair value as of the beginning of the year for warrants expiring during the year and has been recorded to the Consolidated Statement of Operations for the year ended December 31, 2015. |
The fair value of the warrant liability is based on Level 3 inputs. For this liability, the Company developed its own assumptions that do not have observable inputs or available market data to support the fair value. See Note 7 for further discussion of the warrant liability.
Effect of Fibrocell's Stock Price and Volatility Assumptions on the Calculation of Fair Value of Warrant Liabilities.
The fair value of the Company's warrant liability is based on Level 3 inputs. As discussed in Note 7, the Company uses a Monte Carlo simulation valuation method to value its liability-classified warrants. The determination of fair value as of the
F 18
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 9. Fair Value Measurements (continued)
reporting date is affected by Fibrocell's stock price as well as assumptions regarding a number of subjective variables that do not have observable inputs or available market data to support the fair value. These variables include, but are not limited to, expected stock price volatility over the term of the warrants and the risk-free interest rate. The primary factors affecting the fair value of the warrant liability are the Company's stock price and volatility as well as certain assumptions by the Company as to the likelihood of provisions to the underlying warrant agreements being triggered. The methods described above and in Notes 3 and 7 may produce a fair value calculation that may not be indicative of net realizable value or reflective of future fair values. Furthermore, while the Company believes its valuation method is appropriate and consistent with other market participants, the use of different methodologies or assumptions to determine the fair value could result in a different fair value measurement at the reporting date.
Fair Value of Certain Financial Assets and Liabilities.
The Company believes that the fair values of its current assets and liabilities approximate their reported carrying amounts. There were no transfers between Level 1, 2 and 3 during the periods presented.
Note 10. Stock-Based Compensation
The Company's board of directors (the “Board”) adopted the 2009 Equity Incentive Plan (as amended to date, the “Plan”) effective September 3, 2009. The Plan is intended to further align the interests of the Company and its stockholders with its employees, including its officers, non-employee directors, consultants and advisers by providing equity-based incentives. The Plan allows for the issuance of up to 5,600,000 shares of the Company’s common stock. In addition, there were 206,000 options issued outside of the Plan to consultants in prior years.
The types of awards that may be granted under the Plan include options (both non-qualified stock options and incentive stock options), stock appreciation rights, stock awards, stock units, and other stock-based awards. The term of each award is determined by the Compensation Committee of the Board at the time each award is granted, provided that the terms of options do not exceed ten years. Vesting schedules for stock options granted to employees vary, but generally vest 25% per year, over four years. The Plan had 2,426,659 options available for grant as of December 31, 2015.
The Company recognizes compensation cost for stock-based awards based on their grant date fair value. During the years ended December 31, 2015, 2014 and 2013, the weighted average fair market value using the Black-Scholes option-pricing model for the options granted was $3.59, $2.64 and $2.79, respectively. The fair market value of the stock options at the date of grant was estimated using the Black-Scholes option-pricing model with the following weighted average assumptions for the years ended December 31:
2015 | 2014 | 2013 | ||||||
Expected life (1) | 6 years, 1 month | 5 years, 11 months | 5 years, 7 months | |||||
Interest rate | 1.6 | % | 1.9 | % | 1.6 | % | ||
Dividend yield | — | — | — | |||||
Volatility (2) | 103.2 | % | 70.0 | % | 71.0 | % | ||
(1) | The Company uses the simplified method for estimating the stock option term. |
(2) | The Company uses historical volatility of a peer group for estimating expected volatility of the Company's shares. |
Expected stock price volatility is based on historical volatility of the Company’s stock and the stock of the Company’s peer companies. The risk-free interest rate for periods within the contractual life of the option is based on the U.S. Treasury yield curve in effect at the time of the grant. The expected life for options granted represents the period of time that options granted are expected to be outstanding and is derived from the contractual terms of the options granted. The Company estimates future forfeitures of options based upon historical forfeiture rates.
Total stock-based compensation expense, net of estimated forfeitures, recognized using the straight-line attribution method in the Consolidated Statements of Operations for the years ended December 31, 2015, 2014 and 2013 was as follows:
F 19
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 10. Stock-Based Compensation (continued)
($ in thousands) | 2015 | 2014 | 2013 | ||||||||
Research and development: | |||||||||||
Stock-based compensation expense for employees | $ | 158 | $ | 38 | $ | 32 | |||||
Equity awards for non-employees issued for services | — | 3 | 4 | ||||||||
Selling, general and administrative: | |||||||||||
Stock-based compensation expense for employees and directors | 1,880 | 1,198 | 1,013 | ||||||||
Equity awards for non-employees issued for services | — | — | 101 | ||||||||
Total stock-based compensation expense | $ | 2,038 | $ | 1,239 | $ | 1,150 | |||||
($ in thousands, except share and per share data) | Number of shares | Weighted- average exercise price | Weighted- average remaining contractual term (in years) | Aggregate intrinsic value | ||||||||
Outstanding at December 31, 2012 | 562,025 | $ | 18.56 | 7 years | $ | — | ||||||
Granted | 1,532,000 | 4.15 | ||||||||||
Forfeited | (25,305 | ) | 14.71 | |||||||||
Outstanding at December 31, 2013 | 2,068,720 | $ | 7.93 | 8 years, 5 months | $ | 544 | ||||||
Granted | 348,000 | 4.19 | ||||||||||
Expired | (51,637 | ) | 21.61 | |||||||||
Forfeited | (278,633 | ) | 4.45 | |||||||||
Outstanding at December 31, 2014 | 2,086,450 | $ | 7.43 | 7 years, 2 months | $ | — | ||||||
Granted | 1,352,114 | 4.48 | ||||||||||
Exercised | (56,250 | ) | 4.53 | |||||||||
Expired | (65,250 | ) | 10.54 | |||||||||
Forfeited | (182,970 | ) | 5.91 | |||||||||
Outstanding at December 31, 2015 | 3,134,094 | $ | 6.23 | 8 years | $ | 1,630 | ||||||
Vested and expected to vest at December 31, 2015 | 3,015,584 | $ | 6.32 | 7 years, 11 months | $ | 1,556 | ||||||
Exercisable at December 31, 2015 | 1,479,939 | $ | 8.47 | 7 years, 1 month | $ | 624 | ||||||
The total fair value of shares vested during the years ended December 31, 2015, 2014 and 2013 was $1.5 million, $1.2 million, and $1.2 million, respectively.
During the year ended December 31, 2015, a total of 56,250 stock options with a total intrinsic value of approximately $0.04 million were exercised resulting in proceeds of approximately $0.3 million. There were no exercises of vested stock options during the years ended 2014 or 2013.
As of December 31, 2015, there was $4.1 million of total unrecognized compensation cost related to nonvested stock options which vest over time. That cost is expected to be recognized over a weighted average period of 2.7 years. As of December 31, 2015, there were 1,654,155 nonvested stock options with a weighted average exercise price of $4.23 and an average intrinsic value of $1.0 million.
Note 11. Income Taxes
Fibrocell Science, Inc. and Fibrocell Technologies, Inc. file a consolidated U.S. federal income tax return, and file U.S. state income tax returns in several jurisdictions as well. In general, the U.S. federal and state income tax returns remain open to examination by taxing authorities for tax years beginning in 2012 to present. However, if and when the Company claims net operating loss (“NOL”) carryforwards from years prior to 2012 against future taxable income, those losses may be examined by the taxing authorities. The Company's foreign subsidiaries file income tax returns in their respective jurisdictions.
F 20
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 11. Income Taxes (continued)
The components of the income tax expense (benefit) related to operations, were as follows:
Year ended December 31, | Year ended December 31, | Year ended December 31, | |||||||||
($ in thousands) | 2015 | 2014 | 2013 | ||||||||
U.S. Federal: | |||||||||||
Current | $ | — | $ | — | $ | — | |||||
Deferred | — | — | — | ||||||||
U.S. State: | |||||||||||
Current | — | — | — | ||||||||
Deferred | — | — | — | ||||||||
Income tax expense (benefit) | $ | — | $ | — | $ | — | |||||
The reconciliation between income tax expense (benefit) at the U.S. federal statutory rate and the amount recorded in the accompanying Consolidated Financial Statements were as follows:
Year ended December 31, | Year ended December 31, | Year ended December 31, | |||||||||
($ in thousands) | 2015 | 2014 | 2013 | ||||||||
Tax benefit at U.S. federal statutory rate | $ | (12,183 | ) | $ | (8,977 | ) | $ | (11,044 | ) | ||
Increase in domestic valuation allowance | 14,236 | 11,109 | 11,626 | ||||||||
State income taxes/(benefit) before valuation allowance, net of federal benefit | (1,122 | ) | (846 | ) | (1,026 | ) | |||||
Warrant revaluation and other finance (income)/expense | (1,026 | ) | (1,375 | ) | 369 | ||||||
Other | 95 | 89 | 75 | ||||||||
Income tax expense (benefit) | $ | — | $ | — | $ | — | |||||
The components of the Company’s net deferred tax assets and liabilities at December 31, 2015, 2014 and 2013 were as follows:
($ in thousands) | December 31, 2015 | December 31, 2014 | December 31, 2013 | ||||||||
Deferred tax liabilities: | |||||||||||
Intangible assets | $ | 1,764 | $ | 2,001 | $ | 2,247 | |||||
Total deferred tax liabilities | $ | 1,764 | $ | 2,001 | $ | 2,247 | |||||
Deferred tax assets: | |||||||||||
Loss carryforwards | $ | 77,194 | $ | 63,560 | $ | 54,253 | |||||
Capital loss carryforward | 840 | 841 | 844 | ||||||||
Property and equipment | 1,096 | 1,135 | 1,149 | ||||||||
License fees | 8,351 | 7,055 | 5,393 | ||||||||
Accrued expenses and other | 886 | 602 | 412 | ||||||||
Stock compensation | 3,445 | 2,953 | 2,698 | ||||||||
Total deferred tax assets before valuation allowance | 91,812 | 76,146 | 64,749 | ||||||||
Less: valuation allowance | (90,048 | ) | (74,145 | ) | (62,502 | ) | |||||
Total deferred tax assets | $ | 1,764 | $ | 2,001 | $ | 2,247 | |||||
Net deferred tax assets | $ | — | $ | — | $ | — | |||||
As of December 31, 2015, the Company had generated U.S. net operating loss carryforwards of approximately $197.8 million which expire from 2018 to 2035. The NOL carryforwards are available to reduce future taxable income. However, the NOL carryforwards may be, or become subject to, an annual limitation in the event of certain cumulative changes in the ownership interest of significant stockholders over a three year period in excess of 50%, as defined under Sections 382 and 383 of the Internal Revenue Code of 1986, as amended, as well as similar state tax provisions. This could limit the amount of NOL's that the Company can utilize annually to offset future taxable income or tax liabilities. The amount of the annual
F 21
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 11. Income Taxes (continued)
limitation, if any, will be determined based on the value of the Company immediately prior to an ownership change. Subsequent ownership changes may further affect the limitation in future years. If and when the Company utilizes the NOL carryforwards in a future period, it will perform an analysis to determine the effect, if any, of these loss limitation rules on the NOL carryforward balances. Additionally, U.S. tax laws limit the time during which these carryforwards may be applied against future taxes, therefore, the Company may not be able to take full advantage of these carryforwards for federal income tax purposes. In addition, the Company has NOL carryforwards in certain non-U.S. jurisdictions of approximately $0.2 million. However, it is not expected that these non-U.S. loss carryforwards will ever be utilized, so they are not included in the components of deferred taxes listed above. Finally, there are no unremitted earnings in foreign jurisdictions, so no provision for taxes thereupon is required.
As the Company has had cumulative losses and there is no assurance of future taxable income, valuation allowances have been recorded to fully offset deferred tax assets at December 31, 2015, 2014, and 2013. The valuation allowance increased by $15.9 million, $11.6 million, and $10.2 million during 2015, 2014, and 2013, respectively, primarily due to the impact from the current year net losses incurred.
Note 12. Related Party Transactions
Overview of Related Parties
The Company and Intrexon are parties to two distinct exclusive channel collaboration agreements, as more fully described below. Pursuant to these agreements, the Company engages Intrexon for support services for the research and development of product candidates covered under the respective agreements and reimburses Intrexon for its cost for time and materials for such work. Additionally, the Company's future commitments pursuant to the agreements include cash royalties and various developmental and commercial milestone payments as more fully described in Note 14.
Randal J. Kirk is the chairman of the board and chief executive officer of Intrexon and, together with his affiliates, owns more than 50% of Intrexon's common stock. Randal J. Kirk, together with his affiliates (including Intrexon) own approximately 38% of our common stock. Additionally, two of our directors, Julian Kirk (who is the son of Randal J. Kirk) and Marcus Smith, are employees of Third Security, LLC, which is an affiliate of Randal J. Kirk.
For the years ended December 31, 2015, 2014 and 2013, the Company incurred expenses of $15.9 million, $9.4 million and $10.1 million, respectively, for payments to Intrexon. Of the expenses incurred during the 2015 period, $10.0 million related to an up-front technology access fee pursuant to the 2015 ECC, $3.1 million related to direct expenses for work performed by Intrexon and $2.8 million related to pass-through costs. Of the costs incurred during the 2014 period, $5.2 million related to non-cash supplemental stock issuance costs pursuant to the second amendment to the 2012 ECC, $2.3 million related to direct expenses for work performed by Intrexon and $1.9 million related to pass-through costs. Of the costs incurred during the 2013 period, $6.4 million related to non-cash supplemental stock issuance costs pursuant to the first amendment to the 2012 ECC, $2.9 million related to direct expenses for work performed by Intrexon and $0.8 million related to pass-through costs. As of December 31, 2015 and 2014, the Company had outstanding payables with Intrexon of $10.7 million and $1.0 million, respectively.
Intrexon Collaboration - 2012 ECC
In October 2012, the Company entered into an Exclusive Channel Collaboration Agreement with Intrexon which was amended in June 2013 and January 2014 (as amended, the “2012 ECC”) pursuant to which the Company is Intrexon's exclusive channel collaborator in the research, development and commercialization of products in the following fields (the "2012 Fields"):
• | the enhanced production and purification of autologous fibroblasts, without gene therapy, for all aesthetic and therapeutic indications; |
• | the enhanced production and purification of autologous dermal cells, without gene therapy, for aesthetic and therapeutic treatment of dermal, vocal cord, and periodontal indications; |
• | the development of our gene therapies applied to autologous fibroblasts for all aesthetic and therapeutic indications; |
F 22
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 12. Related Party Transactions (continued)
• | the development of our gene therapies applied to autologous dermal cells for aesthetic and therapeutic treatment of dermal, vocal cord, and periodontal indications; |
• | autologous human fibroblasts with gene therapy to express a therapeutic protein and/or bioactive ribonucleic acid for the treatment of autoimmune and non-infectious inflammatory disorders that manifest in cutaneous tissues, fascia and/or muscle; and |
• | autologous human fibroblasts with gene therapy to express bioactive Tenascin-X locally to correct connective tissue disorders associated with Ehlers-Danlos Syndrome (hypermobility type). |
Pursuant to the terms of the 2012 ECC, Intrexon has granted the Company a license to use its proprietary technologies and other intellectual property to research, develop and commercialize products in the 2012 Fields within the United States. The Company is responsible for all costs incurred in connection with the research, development and commercialization of products under the 2012 ECC and will own all clinical data, regulatory filings and regulatory approvals relating to such products. The Company engages Intrexon for support services for the research and development of products under the 2012 ECC and reimburses Intrexon for its cost for time and materials for such services.
In September 2015, the Company and Intrexon entered into a letter of agreement pursuant to which the parties mutually agreed to terminate their collaboration with respect to the development of potential therapies to treat Ehlers-Danlos
Syndrome (hypermobility type) due to technical hurdles. As a result, the Company no longer has any rights or obligations under the 2012 ECC with respect to the development of “autologous human fibroblasts genetically modified to express bioactive Tenascin-X locally to correct connective tissue disorders”.
In connection with the execution of the 2012 ECC, including certain amendments, the Company entered into stock issuance agreements with Intrexon. See Note 8 for details.
The Company is required to pay Intrexon quarterly cash royalties on all products developed under the 2012 ECC in an amount equal to 7% on aggregate annualized net sales up to $100 million, plus 14% on aggregate annualized net sales greater than $100 million. The Company is also required to pay Intrexon half of any sublicensing revenues that it receives from third parties in consideration for sublicenses granted by the Company with respect to products developed under the 2012 ECC, but only to the extent such sublicensing revenues are not included in net sales subject to royalties. Sales from LAVIV (azficel-T), including new indications, or other products that the Company develops and commercializes outside of the 2012 ECC are not subject to royalty payments unless the Company is able to reduce the product's cost of goods sold through the 2012 ECC, in which case, the Company is required to pay quarterly cash royalties on such products equal to one third of such cost of goods sold savings.
Intrexon Collaboration - 2015 ECC
In December 2015, the Company entered into an additional Exclusive Channel Collaboration Agreement with Intrexon (the “2015 ECC”) pursuant to which the Company is Intrexon’s exclusive channel collaborator in the research, development and commercialization of products for the treatment of chronic inflammatory and degenerative diseases of human joints through intra-articular or other local administration of genetically modified fibroblasts (the “2015 Field”).
Pursuant to the terms of the 2015 ECC, Intrexon has granted the Company a license to use its proprietary technologies and other intellectual property to develop and commercialize collaboration products in the 2015 Field throughout the world. The Company is responsible for all costs incurred in connection with the development and commercialization of collaboration products and will own all clinical data, regulatory filings and regulatory approvals relating to such products. The Company engages Intrexon for support services in connection with the research and development of products under the 2015 ECC and reimburses Intrexon for its cost for time and materials for such services.
In consideration for the license and the other rights that the Company receives under the 2015 ECC, the Company paid Intrexon an up-front technology access fee of $10 million in cash in January 2016. For each collaboration product the Company develops under the 2015 ECC, the Company is required to pay Intrexon development milestones of up to $30 million and commercialization milestones of up to $22.5 million, a low double-digit royalty on its net sales of such products and half of any sublicensing revenues received from third parties for such products.
F 23
Note 13. Loss Per Share
Details in the computation of basic and diluted loss per share were as follows:
For the Twelve Months Ended December 31, | |||||||||||
($ in thousands except share and per share data) | 2015 | 2014 | 2013 | ||||||||
Loss per share — Basic: | |||||||||||
Numerator for basic loss per share | $ | (34,453 | ) | $ | (25,650 | ) | $ | (31,554 | ) | ||
Denominator for basic loss per share | 42,178,397 | 40,789,445 | 29,830,207 | ||||||||
Basic loss per common share | $ | (0.82 | ) | $ | (0.63 | ) | $ | (1.06 | ) | ||
Loss per share — Diluted: | |||||||||||
Numerator for basic loss per share | $ | (34,453 | ) | $ | (25,650 | ) | $ | (31,554 | ) | ||
Adjust: Warrant revaluation income (expense) for dilutive warrants | 1,529 | 2,840 | 2,270 | ||||||||
Numerator for diluted loss per share | $ | (35,982 | ) | $ | (28,490 | ) | $ | (33,824 | ) | ||
Denominator for basic loss per share | 42,178,397 | 40,789,445 | 29,830,207 | ||||||||
Plus: Incremental shares underlying “in the money” warrants outstanding | 172,949 | 179,954 | 366,409 | ||||||||
Denominator for diluted loss per share | 42,351,346 | 40,969,399 | 30,196,616 | ||||||||
Diluted loss per common share | $ | (0.85 | ) | $ | (0.70 | ) | $ | (1.12 | ) | ||
The following potentially dilutive securities have been excluded from the computations of diluted weighted average shares outstanding, as their effect would be anti-dilutive:
For the Twelve Months Ended December 31, | ||||||||
2015 | 2014 | 2013 | ||||||
Shares underlying “in the money” options outstanding | 1,888,348 | 555,750 | 348,250 | |||||
Shares underlying “out of the money” options outstanding | 1,173,590 | 1,700,272 | 820,047 | |||||
Shares underlying “in the money” warrants outstanding | 897,244 | 300,425 | 334,175 | |||||
Shares underlying “out of the money” warrants outstanding | 4,721,408 | 4,831,352 | 4,834,852 | |||||
Note 14. Commitments and Contingencies
Leases
On April 6, 2005, the Company entered into a non-cancellable operating lease (the “Lease”) for its office, warehouse and laboratory facilities in Exton, Pennsylvania. The lease agreement had an original term of 8 years. On February 17, 2012, the Company entered into an amended and restated lease (the “Amended Lease”) for an additional term of 10 years through the year 2023. The Lease and the Amended Lease provide for rent payments escalating on a periodic basis. In accordance with ASC 840-20, Operating Leases, the Company calculated the total minimum payments under the lease and divided them equally over the life of the lease to account for the lease on a straight-line basis. The difference between actual rent payments and payments accounted for using the straight-line basis are reflected as deferred rent on the Company's Consolidated Balance Sheets. The Company has the option to renew the lease for an additional 5 years at fair market value. Rental expense totaled approximately $1.6 million, $1.6 million and $1.5 million for the years ended December 31, 2015, 2014 and 2013, respectively.
UCLA Collaboration
On May 21, 2014, the Company entered into a research agreement with The Regents of the University of California ("UCLA"), under which the Company and UCLA agreed to undertake collaborative research activities over a three to five year period to develop novel intellectual property and products relating to (a) somatic dermal derivatives and (b) human induced pluripotent stem cells (the "UCLA Research Agreement"). Under the terms of this agreement, the Company agreed to pay UCLA for performance under each portion of the research projects based on budgeted amounts explicitly stated in the agreement, in the aggregate amount of approximately $0.5 million for years one through three. Payments for optional years
F 24
Fibrocell Science, Inc.
Notes to Consolidated Financial Statements
Note 14. Commitments and Contingencies (continued)
four and five in the aggregate amount of approximately $0.5 million are due upon election of each option set forth in the agreement. As of December 31, 2015, the Company has not elected to proceed with the option periods.
In connection with this collaboration, the Company and UCLA also entered into an exclusive license agreement on June 13, 2014, pursuant to which UCLA granted the Company an exclusive, sublicensable right and license to use certain intellectual property developed in collaboration with UCLA relating to media that promotes genomic stability in induced pluripotent stem cell cultures for all research, development and commercialization purposes (the "UCLA License Agreement"). In consideration of the license granted under this agreement, the Company agreed to pay to UCLA a license issue fee, a license maintenance fee (waived if earned royalties are paid), certain one-time milestone payments, earned royalties on net sales of all licensed products (including sales by affiliates) and a percentage of amounts received from sublicensing activities. The Company is subject to minimum annual royalty payments to UCLA beginning after the first commercial sale of a licensed product.
Collaboration with Related Party (Intrexon)
The Company is a party to two separate exclusive channel collaboration agreements with Intrexon, a related party. Pursuant to the agreements, the Company is Intrexon's exclusive channel collaborator in the research, development and commercialization of products in certain defined fields. The Company is required to pay future royalties, as well as development and commercialization milestones, under these agreements. See Note 12 for additional details.
Contractual Obligations
The following table summarizes the Company’s minimum contractual obligations as of December 31, 2015:
Payments due by period | |||||||||||||||||||||||||||
($ in thousands) | Total | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 and thereafter | ||||||||||||||||||||
License fee obligations (1) | $ | 48 | $ | 8 | $ | 8 | $ | 8 | $ | 8 | $ | 8 | $ | 8 | |||||||||||||
Research obligations (2) | 202 | 202 | — | — | — | — | — | ||||||||||||||||||||
Operating lease obligations (3) | 9,958 | 1,254 | 1,254 | 1,254 | 1,416 | 1,471 | 3,309 | ||||||||||||||||||||
Collaboration with related party (4) | 10,000 | 10,000 | — | — | — | — | — | ||||||||||||||||||||
Total | $ | 20,208 | $ | 11,464 | $ | 1,262 | $ | 1,262 | $ | 1,424 | $ | 1,479 | $ | 3,317 | |||||||||||||
(1) | Obligations under the UCLA License Agreement. The amounts in the table assume the foregoing agreement is continued through its respective term. The agreement may be terminated at the option of either party. In such event, the Company’s obligation would be limited to costs through the date of such termination. |
(2) | Obligations under the UCLA Research Agreement. The amounts stated are based upon budgeted project costs agreed to for the term of the agreement. |
(3) | Operating lease obligations are stated based on the Amended Lease agreement for the office, warehouse and laboratory facilities executed in February 2012. |
(4) | Up-front technology access fee pursuant to 2015 ECC with Intrexon. Paid in January 2016. |
Note 15. Subsequent Events
Common Stock - "At-The-Market" Equity Program
On January 21, 2016, the Company entered into a Controlled Equity Offering™ Sales Agreement (the “ATM Agreement”) with Cantor Fitzgerald & Co. (“Cantor Fitzgerald”) to implement an "At-The-Market" (“ATM”) equity program under which the Company, from time to time, may offer and sell shares of its common stock having an aggregate offering price of up to $50.0 million (the “Shares”) through Cantor Fitzgerald.
Subject to the terms and conditions of the Agreement, Cantor Fitzgerald will use its commercially reasonable efforts to sell the Shares from time to time, based upon the Company’s instructions. The Company has no obligation to sell any of the Shares, and may at any time suspend sales under the ATM Agreement or terminate the ATM Agreement. Cantor Fitzgerald will be entitled to a fixed commission of up to 3.0% of the gross proceeds from Shares sold.
F 25
Note 16. Quarterly Financial Information (unaudited)
This table summarizes the unaudited consolidated financial results of operations for the quarters ended:
($ in thousands, except per share data) | March 31, | June 30, | September 30, | December 31, | |||||||||||
2015 Quarter Ended | |||||||||||||||
Total revenue | $ | 194 | $ | 137 | $ | 79 | $ | 82 | |||||||
Total cost of revenue | 147 | 162 | 204 | 209 | |||||||||||
Gross loss | 47 | (25 | ) | (125 | ) | (127 | ) | ||||||||
Operating expenses | 6,911 | 7,334 | 6,698 | 16,234 | |||||||||||
Other income (expense) | (1,661 | ) | 603 | 5,302 | (1,290 | ) | |||||||||
Net loss | $ | (8,525 | ) | $ | (6,756 | ) | $ | (1,521 | ) | $ | (17,651 | ) | |||
Basic net loss per share | $ | (0.21 | ) | $ | (0.17 | ) | $ | (0.04 | ) | $ | (0.40 | ) | |||
Diluted net loss per share | $ | (0.21 | ) | $ | (0.17 | ) | $ | (0.07 | ) | $ | (0.40 | ) | |||
2014 Quarter Ended | |||||||||||||||
Total revenue | $ | 46 | $ | 58 | $ | 20 | $ | 56 | |||||||
Total cost of revenue | 793 | 547 | 512 | 460 | |||||||||||
Gross loss | (747 | ) | (489 | ) | (492 | ) | (404 | ) | |||||||
Operating expenses | 10,253 | 6,107 | 5,699 | 5,763 | |||||||||||
Other income (expense) | (3,009 | ) | 4,339 | 179 | 2,795 | ||||||||||
Net loss | $ | (14,009 | ) | $ | (2,257 | ) | $ | (6,012 | ) | $ | (3,372 | ) | |||
Basic net loss per share | $ | (0.35 | ) | $ | (0.06 | ) | $ | (0.15 | ) | $ | (0.08 | ) | |||
Diluted net loss per share | $ | (0.35 | ) | $ | (0.09 | ) | $ | (0.17 | ) | $ | (0.09 | ) | |||
F 26
