Attached files
| file | filename |
|---|---|
| EX-32 - EX-32 - NEUROCRINE BIOSCIENCES INC | d520757dex321.htm |
| EX-31.2 - EX-31.2 - NEUROCRINE BIOSCIENCES INC | d520757dex312.htm |
| EX-31.1 - EX-31.1 - NEUROCRINE BIOSCIENCES INC | d520757dex311.htm |
| EX-23.1 - EX-23.1 - NEUROCRINE BIOSCIENCES INC | d520757dex231.htm |
| EX-21.1 - EX-21.1 - NEUROCRINE BIOSCIENCES INC | d520757dex211.htm |
| EX-10.26 - EX-10.26 - NEUROCRINE BIOSCIENCES INC | d520757dex1026.htm |
| EX-10.17 - EX-10.17 - NEUROCRINE BIOSCIENCES INC | d520757dex1017.htm |
| EX-3.2 - EX-3.2 - NEUROCRINE BIOSCIENCES INC | d520757dex32.htm |
| EX-3.1 - EX-3.1 - NEUROCRINE BIOSCIENCES INC | d520757dex31.htm |
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
(Mark One)
| ☑ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2017
OR
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 0-22705
NEUROCRINE BIOSCIENCES, INC.
(Exact name of registrant as specified in its charter)
| Delaware | 33-0525145 | |
| (State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification Number) | |
| 12780 El Camino Real, San Diego, CA | 92130 | |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code:
(858) 617-7600
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
Name of Each Exchange on Which Registered | |
| Common Stock, $0.001 par value | The NASDAQ Stock Market |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☑ No ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☑
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☑ No ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ☑ No ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ☑
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer ☑ |
Accelerated filer ☐ | Non-accelerated filer ☐ (Do not check if a smaller reporting company) |
Smaller reporting company ☐ | |||
| Emerging growth company ☐ |
||||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☑
The aggregate market value of the common equity held by non-affiliates of the registrant as of June 30, 2017 totaled approximately $3,407,988,476 based on the closing price for the registrant’s Common Stock on that day as reported by the NASDAQ Stock Market. Such value excludes Common Stock held by executive officers, directors and 10% or greater stockholders as of June 30, 2017. The identification of 10% or greater stockholders as of June 30, 2017 is based on applicable Schedule 13G and amended Schedule 13G reports. This calculation does not reflect a determination that such parties are affiliates for any other purposes.
As of February 2, 2018, there were 89,273,537 shares of the registrant’s Common Stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
| Document Description |
10-K Part | |||
| Portions of the registrant’s notice of annual meeting of stockholders and proxy statement to be filed pursuant to Regulation 14A within 120 days after registrant’s fiscal year end of December 31, 2017 are incorporated by reference into Part III of this report |
III | |||
Table of Contents
INGREZZA® is a registered trademark of Neurocrine Biosciences, Inc. Any other brand names or trademarks appearing in this Annual Report that are not the property of Neurocrine Biosciences, Inc. are the property of their respective holders.
2
Table of Contents
FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K and the information incorporated herein by reference contain forward-looking statements that involve a number of risks and uncertainties. Although our forward-looking statements reflect the good faith judgment of our management, these statements can only be based on facts and factors currently known by us. Consequently, these forward-looking statements are inherently subject to risks and uncertainties, and actual results and outcomes may differ materially from results and outcomes discussed in the forward-looking statements.
Forward-looking statements can be identified by the use of forward-looking words such as “believes,” “expects,” “hopes,” “may,” “will,” “plan,” “intends,” “estimates,” “could,” “should,” “would,” “continue,” “seeks,” “pro forma,” or “anticipates,” or other similar words (including their use in the negative), or by discussions of future matters such as the development of new products, technology enhancements, possible changes in legislation and other statements that are not historical. These statements include but are not limited to statements under the captions “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business,” as well as other sections in this report. You should be aware that the occurrence of any of the events discussed under the heading “Item 1A. Risk Factors” and elsewhere in this report could substantially harm our business, results of operations and financial condition and that if any of these events occurs, the trading price of our common stock could decline and you could lose all or a part of the value of your shares of our common stock.
The cautionary statements made in this report are intended to be applicable to all related forward-looking statements wherever they may appear in this report. We urge you not to place undue reliance on these forward-looking statements, which speak only as of the date of this report. Except as required by law, we assume no obligation to update our forward-looking statements, even if new information becomes available in the future.
| ITEM 1. | BUSINESS |
Overview
We were originally incorporated in California in January 1992 and were reincorporated in Delaware in May 1996.
We are a company focused on discovering, developing, and commercializing innovative and life-changing pharmaceuticals, in diseases with high unmet medical needs, through our novel R&D platform, focused on neurological and endocrine based diseases and disorders. Our portfolio of products and product candidates is led by INGREZZA® (valbenazine), a vesicular monoamine transporter 2 (VMAT2) inhibitor for the treatment of movement disorders which was approved by the U.S. Food and Drug Administration (FDA) on April 11, 2017 for the treatment of tardive dyskinesia (TD). Our three lead late-stage clinical programs are INGREZZA (valbenazine) for Tourette syndrome, elagolix, a gonadotropin-releasing hormone (GnRH) antagonist for women’s health that is partnered with AbbVie Inc. (AbbVie), and opicapone, a highly-selective catechol-O-methyltransferase inhibitor (COMT inhibitor) that is an adjunct therapy to preparations of levodopa/DOPA decarboxylase inhibitors for adult patients with Parkinson’s disease and was in-licensed from BIAL – Portela & CA, S.A. (BIAL).
We believe that INGREZZA has the potential to address important unmet medical needs in neurological and psychiatric disorders beyond TD and we plan to continue to study the use of INGREZZA in other disease states. We are currently investigating the utilization of INGREZZA in Tourette syndrome. We have completed three clinical trials in Tourette’s patients and a Phase IIb study in children with Tourette syndrome is currently ongoing. We intend to utilize the results of these studies to discuss with the FDA a plan for New Drug Application (NDA) submission for INGREZZA in Tourette syndrome patients.
3
Table of Contents
Our partner AbbVie has successfully completed the placebo-controlled portion of two Phase III studies of elagolix in women with endometriosis. Based on the positive results of these studies, AbbVie submitted an NDA to the FDA for elagolix to treat women with endometriosis during the third quarter of 2017. The NDA was accepted for priority review by the FDA and given a Prescription Drug User Fee Act (PDUFA) date in the second quarter of 2018. In addition, AbbVie is also assessing elagolix in women with uterine fibroids. The Phase III program began in early 2016 with two replicate studies of women with heavy uterine bleeding associated with uterine fibroids. AbbVie expects initial top-line efficacy data from the Phase III uterine fibroids program to be available in the first quarter of 2018, and based on the results of the study anticipates a subsequent NDA submission in 2019.
On February 9, 2017, we entered into an exclusive licensing agreement with BIAL for the development and commercialization of opicapone in the United States and Canada. Opicapone is a once-daily, peripherally-acting, highly-selective COMT inhibitor that was approved in June 2016 by the European Medicines Agency (EMA) as an adjunct therapy to preparations of levodopa/DOPA decarboxylase inhibitors for adult patients with Parkinson’s disease and end-of-dose motor fluctuations who cannot be stabilized on those combinations. We held a meeting with the FDA in January 2018 to discuss a potential NDA submission for opicapone and we are currently awaiting the final summary meeting minutes which we expect to receive in February 2018. We intend to commercialize opicapone in the United States and Canada upon the receipt of applicable regulatory approvals.
Our Product Pipeline
The following table summarizes our approved products, our most advanced product candidates currently in clinical development and those currently in research and is followed by detailed descriptions of each program:
| Program |
Target Indication(s) |
Status |
Rights | |||
| Approved products: |
||||||
|
INGREZZA® (valbenazine) |
Tardive Dyskinesia | Marketed | Neurocrine/Mitsubishi Tanabe (Asia-Pacific) | |||
| Product candidates in clinical development: |
||||||
| elagolix |
Endometriosis | Phase III | AbbVie | |||
| elagolix |
Uterine Fibroids | Phase III | AbbVie | |||
| opicapone |
Parkinson’s Disease | Phase III | Neurocrine (U.S. and Canada)/BIAL | |||
|
INGREZZA® (valbenazine) |
Tourette Syndrome | Phase IIb | Neurocrine/Mitsubishi Tanabe (Asia-Pacific) | |||
| NBI-74788 |
Classic Congenital Adrenal Hyperplasia |
Phase II | Neurocrine | |||
| Research programs: |
||||||
| Neurological/Neuropsychiatric (e.g. VMAT2 Inhibitors) |
Movement Disorders, Bipolar Disorder and Schizophrenia |
Research | Neurocrine | |||
| CNS Disorders (Targeted by G Protein-Coupled Receptors and Ion Channels) |
Epilepsy, Essential Tremor, Dystonia, Other Indications | Research | Neurocrine | |||
“Marketed” indicates that we have received FDA regulatory approval of the product, for the specified target indication.
4
Table of Contents
“Phase III” indicates that we or our collaborators are conducting large-scale, multicenter comparative clinical trials on patients afflicted with a target disease in order to provide substantial evidence for efficacy and safety of the product candidate.
“Phase II” indicates that we or our collaborators are conducting clinical trials on groups of patients afflicted with a specific disease in order to determine preliminary efficacy, optimal dosages and expanded evidence of safety of the product candidate.
“Research” indicates identification and evaluation of compound(s) in laboratory and preclinical models.
INGREZZA® (valbenazine) – Vesicular Monoamine Transporter 2 Inhibitor (VMAT2)
VMAT2 is a protein concentrated in the human brain that is essential for the transmission of nerve impulses between neurons. VMAT2 is primarily responsible for packaging and transporting monoamines (dopamine, norepinephrine, serotonin, and histamine) in neurons. Specifically, dopamine enables neurotransmission among nerve cells that are involved in voluntary and involuntary motor control. Disease states such as TD, Tourette syndrome, Huntington’s chorea, schizophrenia, and tardive dystonia are characterized in part by a hyperdopaminergic state in the brain, and modulation of neuronal dopamine levels may provide symptomatic benefits for patients with these conditions, among others.
INGREZZA as a Treatment for Tardive Dyskinesia. TD is defined by hyperkinetic involuntary movements which arise after months or years of treatment with dopamine receptor blocking agents, e.g. antipsychotics used for treating schizophrenia, bipolar disorder, and depression, and Reglan® (metoclopramide) for nausea and vomiting and gastric emptying in patients with gastroparesis. Features of TD may include grimacing, tongue protrusion, lip smacking, puckering and pursing of the lips, and rapid eye blinking. Rapid movements of the extremities may also occur. The impact on daily function and the quality of life for individuals suffering from TD can be substantial. While the prevalence rates of TD can vary greatly in accordance with the population being studied, it is estimated that approximately 500,000 individuals are affected by TD in the United States alone (Kantar Health).
On April 11, 2017, INGREZZA became the first drug approved by the FDA for the treatment of TD. INGREZZA provides a once-daily dosing treatment option for TD causing reversible reduction of dopamine release at the nerve terminal by selectively inhibiting the pre-synaptic VMAT2. In vitro, INGREZZA is a highly selective inhibitor of human VMAT2 showing little or no affinity for VMAT1, other receptors, such as dopamine D2 receptors, other transporters or ion channels. INGREZZA for TD has two dosing options (40 mg and 80 mg) with 40 mg taken for the first seven days of treatment with an option of 40 mg or 80 mg thereafter depending on a patient’s dosing needs. INGREZZA was generally well tolerated during our clinical trials with no apparent drug-drug interactions with the most common emergent adverse event being mild and transient somnolence.
In connection with the FDA approval of INGREZZA for TD, we have committed to conduct certain post-marketing studies including Phase 1 (e.g., pharmacokinetics (PK) in volunteers with renal impairment) and Phase 4 (e.g., randomized placebo-controlled withdrawal in TD patients). We expect to conduct these studies over the next four years in accordance with FDA guidance.
Tourette syndrome. Tourette syndrome is a neurological disorder that consists of rapid, non-rhythmic stereotyped motor and vocal tics. Motor tics are typically characterized by facial grimacing, head jerks, extremity movements and other dystonic movements. Vocal tics typically include grunting, throat clearing, and repeating words and phrases. The average age of onset for Tourette syndrome is approximately six years, with symptoms reaching their peak severity at approximately age ten. Tourette syndrome is more commonly diagnosed in males than females and may also be associated with attention deficit hyperactivity disorder and obsessive compulsive disorder. We were granted FDA Orphan Drug Designation for valbenazine for the treatment of pediatric patients with Tourette s syndrome in 2017. Orphan drug designation is granted by the FDA to drugs that are intended to treat rare diseases or conditions affecting fewer than 200,000 people in the United States. The orphan drug
5
Table of Contents
designation allows the orphan drug indication for the drug to be eligible for a seven-year period of U.S. marketing exclusivity upon approval of the drug, as well as other development assistance and financial incentives.
We have completed juvenile rodent preclinical studies of INGREZZA and based on the results of these preclinical studies, we initiated the T-Force Study in children and adolescents with Tourette syndrome in early 2015. The T-Force Study was an open-label, multiple ascending dose, pharmacokinetic and pharmacodynamic study to evaluate the safety, tolerability and exposure-response of INGREZZA in children and adolescents with Tourette syndrome. A total of 28 patients were evaluated over 14 days of once daily dosing followed by 7 days off-drug at 10 study centers in the United States. The study was divided into two dosing groups consisting of children (ages 6-11) and adolescents (ages 12-18), and each age group was further divided into three dosing cohorts. Subsequent dose escalations for children and adolescents were based, in part, on the pharmacokinetic and safety data from the previous cohort in each age group. The Yale Global Tic Severity Scale was also assessed and after two weeks of treatment showed a mean reduction of 31% from baseline scores, with over half of the subjects considered clinical responders. Based on the results of the T-Force study, we initiated the Phase II program in Tourette syndrome.
The T-Forward study was a randomized, double-blind, placebo-controlled, multi-dose, parallel group, study that enrolled 124 adults with moderate to severe Tourette syndrome. Two once-daily fixed doses of INGREZZA were evaluated versus placebo in a 1:1:1 randomization. The three-arm study included eight weeks of dosing followed by two weeks off-drug at 32 study centers in the United States to assess the safety, tolerability and efficacy of INGREZZA in Tourette patients. The primary endpoint of this study was a change from baseline of placebo versus active scores utilizing the Yale Global Tic Severity Scale at the end of week 8. Tourette symptoms were also be evaluated via the Premonitory Urge for Tics Scale as well as Clinical Global Impression of Change scales, among others. While the T-Forward study showed a significant improvement in overall symptoms of Tourette syndrome as evidenced by the Clinical Global Impression of Change (p=0.015), the pre-specified primary endpoint, the change-from-baseline in the Yale Global Tic Severity Scale at week 8 was not met (p=0.18). Adverse events were dose dependent and consistent with earlier clinical studies.
The T-Force GREEN study was a multicenter, randomized, double-blind, placebo-controlled, multi-dose, parallel group, Phase II study to evaluate the safety, tolerability and efficacy of INGREZZA in up to 90 pediatric patients with moderate to severe Tourette syndrome. Once-daily fixed doses of INGREZZA were evaluated versus placebo in a 1:1:1 randomization. The three-arm study evaluated 98 children and adolescents over six weeks of dosing followed by two weeks off-drug at approximately 40 study centers in the United States. The primary endpoint of this study is the change from baseline of the Yale Global Tic Severity Scale between placebo and active treatment groups at the end of week six. Exposure-response analysis showed that selected doses for the T-Force GREEN study were below the therapeutic range for adequate tic reduction in the majority of subjects. For the subset of subjects with pharmaceutical exposure in the appropriate range, there was a substantial reduction in tics. For subjects with sub-therapeutic exposure, tic reduction was comparable to placebo. Adverse events were consistent with those observed in previous INGREZZA studies.
Based on the results of the T-Force GREEN study and earlier studies of INGREZZA in Tourette patients, we initiated the T-Force GOLD study in October 2017. This study is a multicenter, randomized, double-blind, placebo-controlled, parallel group, Phase IIb study to evaluate the safety, tolerability, efficacy and optimal dose of once-daily INGREZZA in up to 120 pediatric patients with moderate to severe Tourette syndrome. Patients will receive either once-daily dosing of INGREZZA or placebo using a 1:1 randomization over 12 weeks of dosing followed by two weeks off-drug. The primary endpoint of this study is the change from baseline of the Yale Global Tic Severity Scale between placebo and active treatment groups at the end of week 12. Tourette symptoms will also be evaluated via the Premonitory Urge for Tics Scale as well as Clinical Global Impression Scales. Top-line data from this study is expected in late 2018.
6
Table of Contents
elagolix – Gonadotropin-Releasing Hormone (GnRH) Antagonist
GnRH is a peptide that stimulates the secretion of the pituitary hormones that are responsible for sex steroid production and normal reproductive function. Researchers have found that chronic administration of GnRH agonists, after initial stimulation, reversibly shuts down this transmitter pathway and is clinically useful in treating hormone-dependent diseases such as endometriosis and uterine fibroids. Several companies have developed peptide GnRH agonists on this principle, such as Lupron® and Zoladex®. However, since these molecules are peptides, they must be injected via a depot formulation rather than the preferred oral route of administration. In addition, GnRH agonists can take up to several weeks to exert their desired effect once the initial stimulation has occurred, a factor not seen with the use of GnRH antagonists. Upon administration, GnRH agonists have shown a tendency to exacerbate the condition via a hormonal flare. More importantly the profound suppression effect observed with GnRH agonists is similar to that seen after menopause and can be associated with hot flushes and the loss of bone mineral density.
Orally active, nonpeptide GnRH antagonists potentially offer several advantages over injectable GnRH peptide drugs, including rapid onset of hormone suppression without hormonal flare. Also, injection site reactions commonly observed in peptide depots are avoided and dosing can be rapidly discontinued if necessary – a clinical management option not available with long-acting depot injections. Additionally, by using GnRH antagonists, it may be possible to alter the level of pituitary GnRH suppression thereby titrating circulating hormone levels.
In June 2010, we entered into an exclusive worldwide collaboration with AbbVie to develop and commercialize elagolix and all next-generation non-peptide GnRH antagonists (collectively, GnRH Compounds) for women’s and men’s health indications. Under the terms of the agreement, AbbVie is responsible for all development, marketing and commercialization costs and has primary responsibility for all regulatory interactions with the FDA related to elagolix and other GnRH Compounds covered by the collaboration. AbbVie is currently in Phase III evaluation of elagolix in two indications, endometriosis and uterine fibroids.
Endometriosis. Endometriosis is associated with a multitude of symptoms, some of the most common of which include pain related both to menstruation (dysmenorrhea) and sexual intercourse (dyspareunia) as well as chronic pelvic pain throughout the menstrual cycle, infertility, and menorrhagia, among many others. The wide range of symptoms associated with endometriosis serves to complicate and delay diagnosis due to the significant overlap of symptoms with the disease profiles of other conditions. The World Endometriosis Research Foundation estimates that there are over 170 million women worldwide who suffer from endometriosis, including approximately 7.5 million women in the United States alone. We believe that the availability of an oral treatment, lacking the side effect profile of the currently available peptide GnRH agonists, may be a desirable alternative to current pharmaceutical therapies and ultimately encourage a significantly higher treatment rate.
The endometriosis Phase III program evaluated two separate doses of elagolix (150mg once daily and 200mg twice daily) over a 24-week treatment period. The initial randomized, parallel, double-blind, placebo-controlled pivotal trial (Violet PETAL) enrolled 872 women in approximately 160 clinical sites throughout the United States, Canada and Puerto Rico. The co-primary endpoints were a comparison of the daily non-menstrual pelvic pain and daily dysmenorrhea scores during the third month of treatment to the respective daily baseline scores utilizing a responder analysis. Maintenance of response at month six was also assessed utilizing the same daily scales.
In January 2015, AbbVie announced the top-line results of the initial six months of placebo-controlled dosing of the Violet PETAL study. After six months of continuous treatment, both doses of elagolix (150mg once daily and 200mg twice daily) met the study’s co-primary endpoints (p<0.001) of reducing scores of non-menstrual pelvic pain and dysmenorrhea associated with endometriosis, at month three, as well as at month six.
The observed safety profile of elagolix in the Violet PETAL study was consistent with observations from earlier clinical studies. Among the most common adverse events were hot flush, headache, nausea and fatigue.
7
Table of Contents
While most adverse events were similar across treatment groups, some, such as hot flush and bone mineral density loss, were dose-dependent. Overall discontinuation rates were similar across treatment groups and discontinuations specifically due to adverse events were 5.9%, 6.4%, and 9.7% for placebo, 150 mg once daily and 200 mg twice daily, respectively.
Additional efficacy and safety endpoints for the patients enrolled in the Violet PETAL study were measured through one year of continuous dosing as well as for a period of time after the final dose. The one-year dosing portion of this study concluded in mid-2015. In July 2015, AbbVie announced that the efficacy and safety data at one year was consistent with the data witnessed at six months.
In February 2016, AbbVie announced the top-line results from the second of the two Phase III elagolix endometriosis clinical trials, the Solstice Study, a multinational study designed to evaluate the efficacy and safety of elagolix in 815 premenopausal women with endometriosis. The top-line results from this trial were consistent with those of the Violet PETAL Study; after six months of treatment, both doses of elagolix (150 mg once daily and 200 mg twice daily) met the Solstice Study’s co-primary endpoints of reducing scores of non-menstrual pelvic pain and menstrual pain (or dysmenorrhea) associated with endometriosis at month three, as well as month six. The observed safety profile of elagolix in the Solstice Study was consistent with observations from prior studies. Among the most common adverse events were hot flush, headache, and nausea. While most adverse events were similar across treatment groups some, such as hot flush and bone mineral density loss, were dose-dependent. Overall discontinuation rates were similar across treatment groups (25.3%, 21.2%, and 19.7% for placebo, 150 mg once daily and 200 mg twice daily, respectively); discontinuations specifically due to treatment emergent adverse events were 6.1%, 4.4%, and 10.0% for placebo, 150 mg once daily and 200 mg twice daily, respectively. Patients in the Solstice Study were eligible to continue on in either post-treatment follow-up or a blinded extension study for an additional six-month safety and efficacy evaluation of elagolix.
During the third quarter of 2017, AbbVie submitted an NDA for elagolix for the treatment of endometriosis to the FDA. The NDA was accepted for priority review by the FDA and given a PDUFA date in the second quarter of 2018.
Uterine Fibroids. Uterine fibroids are benign hormonally responsive tumors that form in the wall of the uterus. They are the most common solid tumor in women with a prevalence rate of at least 25% (American College of Obstetricians and Gynecologists). While many women do not have symptoms, depending on the size, location and number, uterine fibroids can cause symptoms such as: longer, more frequent, or heavy menstrual bleeding, menstrual pain, vaginal bleeding at time other than menstruation, pain in the abdomen or lower back, pain during sex, difficulty urinating, frequent urination, constipation or rectal pain. Due to the severity of symptoms, treatment sometimes requires surgery, including the removal of the uterus. In fact, uterine fibroids is a leading indication for hysterectomy in the United States, with approximately 250,000 hysterectomies performed each year related to uterine fibroids (Whiteman et al AJOG 2008, 198, e1). We believe that a safe and effective oral therapy would be a preferred treatment regimen rather than surgical intervention.
AbbVie conducted a Phase IIb clinical trial that enrolled approximately 570 women with heavy uterine bleeding due to uterine fibroids at approximately 100 sites in the United States, Canada, Puerto Rico, Chile and the United Kingdom. The trial was a 24-week, randomized, double-blind, multicenter, placebo-controlled, two cohort design study that evaluated the safety and efficacy of two different elagolix treatment regimens (300mg twice daily and 600mg once daily) alone and in combination with two different strengths of hormonal add-back therapy (estradiol/norethindrone acetate). The primary endpoint of the study was an assessment of uterine blood loss after six months of treatment. Secondary efficacy endpoints included change in uterine volume, fibroid volume, and menstrual patterns. Safety assessments of bone mineral density, comparing baseline to month six, were performed via DXA scan. Patients were also followed off drug for up to six months.
Results from this Phase IIb study show elagolix reduced heavy menstrual bleeding in all treatment arms. The study’s primary endpoint, a composite design where subjects had to achieve a menstrual blood loss (MBL)
8
Table of Contents
volume of less than 80 mL as well as a 50 percent or greater reduction in MBL volume from baseline at the final study month, was met for all dosing regimens (p<0.001) as assessed utilizing a quantitative measure of reduction in uterine blood flow, the alkaline hematin method.
Among the most common adverse events were hot flush, headache, nausea, and vomiting. Some adverse events such as hot flush were more frequent in the elagolix only treatment arms versus the placebo and elagolix with hormonal add back therapy treatment arms. Reduction in bone mineral density associated with elagolix alone was attenuated when elagolix was co-administered with hormonal add-back therapy.
AbbVie initiated Phase III studies of elagolix in patients with uterine fibroids in early 2016. The Phase III program includes two replicate, pivotal, six-month efficacy and safety studies followed by a six-month safety and efficacy extension study. AbbVie is evaluating 300mg of elagolix dosed twice daily both alone and in combination with hormonal add-back therapy (estradiol/norethindrone acetate). The primary endpoint in these Phase III studies will be the same as that employed in the Phase IIb study: percent of subjects with reduction in uterine blood flow as measured by the alkaline hematin method. AbbVie expects initial top-line data from this Phase III program in the first quarter of 2018, and based on the results of the study anticipates a subsequent NDA submission in 2019.
opicapone, Catechol-O-methyltransferase (COMT) Inhibitor
COMT inhibitors are utilized to prolong the duration of effect of levodopa which is utilized as a primary treatment option for Parkinson’s disease patients. Administration of levodopa often results in adequate control of Parkinson’s symptoms, also referred to as “on-time,” however, there are periods of the day where the effects of levodopa wear off and motor symptoms worsen, these are considered “off-time.” Opicapone is a novel, once-daily, peripherally-acting, highly-selective COMT inhibitor utilized as adjunct therapy to levodopa in Parkinson’s patients. Opicapone works through decreasing the conversion rate of levodopa into 3-O-methyldopa, thereby reducing the off-time period in patients with Parkinson’s and extending the on-time period.
In February 2017, we entered into an exclusive license agreement with BIAL for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. Under the terms of the agreement, we will be responsible for the development and commercialization of opicapone in the United States and Canada.
Parkinson’s Disease. Parkinson’s disease is a chronic and progressive movement disorder that affects approximately one million people in the United States. The disease is characterized by a loss of neurons in the substantia nigra, the area of the brain where dopamine is produced. Dopamine production and synthesis is necessary for coordination and movement. As Parkinson’s progresses, dopamine production steadily decreases resulting in tremor, slowed movement (bradykinesia), impaired posture and balance, and speech and writing problems. There is no present cure for Parkinson’s and management consists of controlling the motor symptoms primarily through administration of levodopa therapies. While this improves the control of Parkinson’s symptoms, as the disease progresses the beneficial effects of levodopa begin to wear off, symptoms worsen and patients experience end-of-dose motor fluctuations. These end of dose motor fluctuations are improved with the addition of a COMT inhibitor to levodopa.
In June 2016, the EMA authorized ONGENTYS® (opicapone) as an adjunct therapy to preparations of levodopa/DOPA decarboxylase inhibitors (DDCIs) in adult patients with Parkinson’s disease and end-of-dose motor fluctuations who cannot be stabilized on those combinations. This approval was based on data from a clinical development program that included 28 clinical studies of more than 900 patients treated with opicapone in 30 countries worldwide.
The two pivotal Phase III studies utilized for European approval, BIPARK-I and BIPARK-II, demonstrated that opicapone once-daily achieved a statistically significant decrease in off-time periods for Parkinson’s patients
9
Table of Contents
compared to placebo. The BIPARK-I study was a placebo-controlled study of approximately 600 patients that also included entacapone as an active comparator. The results of this study also showed that once-daily opicapone was non-inferior to entacapone which is dosed multiple times per day. The BIPARK-II study was a placebo-controlled study of approximately 400 patients that also showed a significant decrease in off-time periods for Parkinson’s patients. In both studies, opicapone was associated with significant improvements in both patient and clinician global assessments of change. The data from these two Phase III trials also demonstrated that opicapone improved motor fluctuations in levodopa-treated patients regardless of concomitant dopamine agonist or monoamine oxidase type B inhibitors used. Opicapone was generally well tolerated and was not associated with relevant electrocardiographic or hepatic adverse events.
Both of the BIPARK Phase III trials included a one-year open-label extension where opicapone sustained the decrease in off-time and increase in on-time periods that was demonstrated during the double-blind placebo-controlled portion of the studies.
We held a meeting with the FDA in January 2018 to discuss a potential NDA submission for opicapone and we are currently awaiting the final summary meeting minutes which we expect to receive in February 2018. We intend to commercialize opicapone in the United States and Canada, subject to regulatory approvals.
NBI-74788- Corticotropin-Releasing Factor (CRF) Receptor1 Antagonist
CRF is a hypothalamic hormone released directly into the hypophyseal portal vasculature which acts on specific CRF1 receptors on corticotropes in the anterior pituitary to stimulate the release of adrenocorticotropin hormone (ACTH). The primary role of ACTH is the stimulation of the synthesis and release of adrenal steroids including cortisol. Cortisol from the adrenals have a negative feedback role at the level of the hypothalamus that decreases CRF release as well as at the level of the pituitary to inhibit the release of ACTH. This tight control loop is known as the hypothalamic-pituitary-adrenal (HPA) axis. Blockade of CRF receptors at the pituitary has been shown to decrease the release of ACTH and subsequently attenuate the production and release of adrenal steroids.
Classic Congenital Adrenal Hyperplasia. Classic congenital adrenal hyperplasia (classic CAH) is a group of autosomal recessive genetic disorders that affects approximately 20,000-30,000 people in the United States and results in an enzyme deficiency altering the production of adrenal steroids. Because of this deficiency, the adrenal glands have little to no cortisol biosynthesis resulting in a potentially life-threatening condition. If left untreated, classic CAH can result in salt wasting, dehydration and eventually death. Even with cortisol replacement, persistent elevation of ACTH from the pituitary gland results in excessive androgen levels leading to virilization of females including precocious puberty, menstrual irregularity, short stature, hirsutism, acne and fertility problems.
Corticosteroids are the current standard of care for classic CAH and are used chronically to both correct the endogenous cortisol deficiency and to reduce the excessive ACTH levels and androgen excess. However, the dose and duration of steroid use required to suppress ACTH is well above the normal physiological level of cortisol; resulting in metabolic syndrome, bone loss, growth impairment, and Cushing’s syndrome as common and serious side effects.
NBI-74788 is a potent, selective, orally-active, CRF receptor antagonist as demonstrated in a range of in vitro and in vivo assays. Blockade of CRF receptors at the pituitary has been shown to decrease the release of ACTH, which in turn decreases the production of adrenal steroids including androgens, and potentially the symptoms associated with classic CAH. Lower ACTH levels would also reduce the amount of exogenous corticosteroid necessary for classic CAH patients to thrive avoiding the side-effects currently associated with excessive steroid therapy.
We conducted a Phase I single ascending dose study of NBI-74788 in healthy volunteers in 2017. Based on the positive results of this Phase I study we initiated a Phase II clinical trial of NBI-74788 in adult patients with
10
Table of Contents
refractory classic CAH. This pilot study is designed to be an open-label, pharmacokinetic/pharmacodynamic study assessing two single, ascending doses of NBI-74788 in up to twenty study participants. Key pharmacodynamic biomarker measurements include ACTH, 17-hydroxyprogesterone (17-OHP), androgen, and cortisol levels collected the morning following bedtime dosing.
We intend to apply for orphan drug designation for NBI-74788 in the treatment of congenital adrenal hyperplasia. Orphan drug designation is granted by the FDA to medicines intended for the treatment, diagnosis or prevention of rare diseases or disorders that affect fewer than 200,000 people in the United States and provides sponsors with development and commercial incentives for such designated compounds and medicines.
Research Programs
Our research and development focus is on addressing diseases and disorders of the central nervous and endocrine systems, which include therapeutic categories ranging from HPA disorders to stress-related disorders and neurological/neuropsychiatric diseases. Central nervous system and endocrinology drug therapies are among the largest therapeutic categories, accounting for over $150 billion in worldwide drug sales according to GlobalData (2014).
Neurological/Neuropsychiatric: VMAT2 Inhibitors
VMAT2 inhibition results in the modulation of dopamine pathways which may also be useful for patients suffering from schizophrenia. Approximately 2.2 million people in the Unites States suffer from schizophrenia at an estimated annual cost of $62 billion. Our discovery efforts around VMAT2 inhibitors also focus on developing novel therapies for schizophrenia sufferers.
Essential Tremor
Essential tremor is one of the most common neurological disorders in adults, impacting an estimated 10 million individuals in the United States (International Essential Tremor Foundation). The disorder is characterized by involuntary, rhythmic, oscillatory movements that most often affect the upper limbs. As the disease progresses, tremor severity often increases and spreads to other parts of the body. Essential tremor has a significant impact on the activities of daily living often resulting in functional disability as the disease progresses and is associated with a high comorbidity rate of social phobia, depression and anxiety. Current pharmacological therapies utilized in the treatment of essential tremor include propranolol and primidone. Deep brain stimulation, an invasive procedure involving the implantation of electrodes within certain areas of the brain, is sometimes utilized for severe essential tremor.
CNS Disorders (Targeted by G Protein-Coupled Receptors and Ion Channels)
G Protein-Coupled Receptors (GPCRs) are the largest known gene superfamily of the human genome. Greater than thirty percent of all marketed prescription drugs act on GPCRs; which makes this class of proteins historically the most successful therapeutic target family. However, only a small fraction of the GPCR gene superfamily has been exploited. Ion channels appear to be represented by approximately 400 genes in the human genome and are currently the targets for approximately seven percent of the current marketed drugs. Next-generation therapies derived from targeting GPCRs and ion channels will be discovered through the understanding of the complex relationships of drug/receptor interactions and their subsequent impact on efficacy, downstream signaling networks and regulation.
Our GPCR research platform has met this requirement by integrating drug discovery research efforts with a suite of assays and assay systems and automated analytical techniques. This process, now also applied to ion channels, provides an unbiased profile of pharmacological protein/ligand interactions coupled with in vivo efficacy using discrete animal models allowing for rapid discovery of initial leads and advancement into
11
Table of Contents
preclinical and clinical development. Importantly, this design cycle is not limited to GPCRs or ion channel targets, but can be utilized for other recently identified proteins that play a role in human disease where current treatments or therapies are either inadequate or nonexistent.
Our Business Strategy
Our goal is to become the leading biopharmaceutical company focused on neurological and endocrine-related diseases and disorders. The following are the key elements of our business strategy:
Continuing to Advance and Build Our Product Portfolio Focused on Neurological and Endocrine-Related Diseases and Disorders. We believe that by continuing to advance and extend our product pipeline, we can mitigate some of the clinical development risks associated with drug development. We currently have multiple programs in various stages of research and development. Our three lead late-stage clinical programs are elagolix, a GnRH antagonist in Phase III development for endometriosis and uterine fibroids that is partnered with AbbVie, INGREZZA (valbenazine), our VMAT2 inhibitor for the treatment of movement disorders which is in Phase IIb development for Tourette syndrome, and opicapone, a highly-selective COMT inhibitor that is an adjunct therapy to preparations of levodopa/DOPA decarboxylase inhibitors for adult patients with Parkinson’s disease and was in-licensed from BIAL. We take a portfolio approach to managing our pipeline that balances the size of the market opportunities with clear and defined clinical and regulatory paths to approval. By doing so, we focus our internal development resources on innovative therapies with improved probabilities of technical and commercial success.
Maintaining Certain Commercial Rights to Our Product Portfolio to Evolve into a Fully-Integrated Pharmaceutical Company. In April 2017, we received approval from the FDA for INGREZZA for the treatment of TD. We market INGREZZA for TD in the United States through our specialty sales force focused primarily on physicians who treat TD patients, including psychiatrists and neurologists. The commercial launch of INGREZZA, with our field-based sales team of approximately 160 personnel, occurred on May 1, 2017. This entire commercial team is comprised of experienced professionals in marketing, access and reimbursement, managed markets, market research, commercial operations, and sales force planning and management. In addition, our commercial infrastructure includes capabilities in manufacturing, medical affairs, quality control, and compliance. We intend to retain commercial rights to certain products, including INGREZZA, that we can effectively and efficiently develop, secure regulatory approval and commercialize, which includes products with a concentrated prescriber base and well-defined patient population that can be accessed with an efficient patient and prescriber outreach program.
Selectively Establishing Corporate Collaborations with Global Pharmaceutical Companies to Assist in the Development of Our Products and Mitigate Financial Risk while Retaining Significant Commercial Upside. We leverage the development, regulatory and commercialization expertise of our corporate collaborators to accelerate the development of certain of our product candidates, while typically retaining co-promotional rights, and at times commercial rights, in North America, as in the case of our collaboration with Mitsubishi Tanabe. We intend to further leverage our resources by selectively entering into additional strategic alliances to enhance our internal development and commercialization capabilities by licensing our technology.
Identifying Novel Drugs to Address Unmet Market Opportunities. We seek to identify and validate novel drugs on characterized targets for internal development or collaboration. For example, GnRH antagonists, compounds designed to reduce the secretions of sex steroids, may represent the first novel non-peptide, non-injectable means of treatment of endometriosis. We believe the creativity and productivity of our discovery research group will continue to be a critical component for our continued success. Research and development costs were $121.8 million, $94.3 million and $81.5 million for the years ended December 31, 2017, 2016 and 2015, respectively.
Acquiring Rights to Complementary Drug Candidates and Technologies. We plan to continue to selectively acquire rights to products in various stages of development to take advantage of our drug development
12
Table of Contents
capabilities. For example, in February 2017, we entered into an exclusive license agreement with BIAL for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. Under the terms of the agreement, we will be responsible for the development and commercialization of opicapone in the United States and Canada.
Our Corporate Collaborations and Strategic Alliances
One of our business strategies is to utilize strategic alliances to enhance our development and commercialization capabilities. The following is a summary of our significant collaborations/alliances:
AbbVie Inc. (AbbVie). In June 2010, we announced an exclusive worldwide collaboration with AbbVie to develop and commercialize elagolix and all next-generation GnRH antagonists (collectively, GnRH Compounds) for women’s and men’s health. AbbVie made an upfront payment of $75 million and has agreed to make additional development and regulatory event based payments of up to $480 million and up to an additional $50 million in commercial event based payments. Under the terms of the agreement, AbbVie is responsible for all development, marketing and commercialization costs. We received funding for certain internal collaboration expenses which included reimbursement from AbbVie for internal and external expenses related to the GnRH Compounds and personnel funding through the end of 2012. We will be entitled to a percentage of worldwide sales of GnRH Compounds for the longer of ten years or the life of the related patent rights. Under the terms of our agreement with AbbVie, the collaborative development effort between the parties to advance GnRH compounds towards commercialization was governed by a joint development committee with representatives from both us and AbbVie. The collaborative development portion of the agreement concluded, as scheduled, on December 31, 2012. AbbVie may terminate the collaboration at its discretion upon 180 days written notice to us. In such event, we would be entitled to specified payments for ongoing clinical development and related activities and all GnRH Compound product rights would revert to us. Since the inception of the agreement, we have recorded revenues of $75.0 million related to the amortization of up-front license fees, $75.0 million in milestone revenue, and $37.0 million of sponsored development revenue.
Mitsubishi Tanabe Pharma Corporation (Mitsubishi Tanabe). In March 2015, we entered into a collaboration and license agreement with Mitsubishi Tanabe for the development and commercialization of INGREZZA for movement disorders in Japan and other select Asian markets. Mitsubishi Tanabe made an up-front license fee payment of $30 million and has agreed to make additional development and commercialization event-based payments totaling up to $85 million, payments for the manufacture of pharmaceutical products, and royalties on product sales in select territories in Asia. Under the terms of the agreement, Mitsubishi Tanabe is responsible for all development, marketing and commercialization costs in Japan and other select Asian markets with the exception of a single Huntington’s chorea clinical trial to be performed by us, at an estimated cost of approximately $12 million, should Mitsubishi Tanabe request the clinical trial. We will be entitled to a percentage of sales of INGREZZA in Japan and other select Asian markets for the longer of ten years or the life of the related patent rights. Mitsubishi Tanabe may terminate the agreement at its discretion upon 180 days’ written notice to us. In such event, all INGREZZA product rights for Japan and other select Asian markets would revert to us. Since the inception of the agreement, we have recorded revenues of $19.8 million related to the up-front license fee, and $15.0 million in milestone revenue.
BIAL – Portela & Ca, S.A. (BIAL). In February 2017, we entered into an exclusive license agreement with BIAL for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. Under the terms of the agreement, we are responsible for the management and cost of all opicapone development and commercialization activities in the United States and Canada. Under the terms of the agreement, we paid BIAL an upfront license fee of $30 million, and we may also be required to pay up to an additional $115 million in milestone payments associated with the regulatory approval and net sales of products containing opicapone. In addition, we will pay BIAL a percentage of net sales (with a floor minimum) in exchange for the manufacture and supply of opicapone drug product.
13
Table of Contents
Upon commercialization, BIAL and Neurocrine will agree on annual sales forecasts. If we fail to meet the minimum sales requirements for a particular year, we will be required to pay BIAL an amount corresponding to the difference between the actual net sales and the minimum sales requirements for such year, and if we fail to meet the minimum sales requirements for any two consecutive years, BIAL may terminate the agreement. The agreement also contemplates that we will purchase, and BIAL will supply, all drug product and investigation medicinal product for our development and commercialization activities. BIAL has the right to co-promote opicapone within the United States and Canada during certain periods of time. If BIAL exercises its option to co-promote the licensed products, we will enter into a co-promotion agreement with BIAL at a future time.
The agreement, unless terminated earlier, will continue on a licensed product-by-licensed product and country-by-country basis until a generic product in respect of such licensed product under the agreement is sold in a country and sales of such generic product are greater than a specified percentage of total sales of such licensed product in such country. Either party may terminate the agreement earlier if the other party materially breaches the agreement and does not cure the breach within a specified notice period, or upon the other party’s insolvency. BIAL may terminate the agreement if we fail to use commercially reasonable efforts or fail to file an NDA for a licensed product by a specified date or under certain circumstances involving a change of control. In certain circumstances where BIAL elects to terminate the agreement in connection with our change of control, BIAL shall pay us a termination fee. We can terminate the agreement at any time for any reason upon six months written notice to BIAL if prior to the first NDA approval in the United States, and upon nine months written notice to BIAL if such notice is given after the first NDA approval in the United States. If our termination request occurs prior to the first NDA approval in the United States, we will have to pay BIAL a termination fee except under certain conditions specified in the agreement.
Intellectual Property
We seek to protect our lead compounds, compound libraries, expressed proteins, synthetic organic processes, formulations, assays, cloned targets, screening technology and other technologies by filing, or by causing to be filed on our behalf, patent applications in the United States and abroad. Additionally, we have licensed from institutions the rights to issued United States patents, pending United States patent applications, and issued and pending foreign filings. We face the risk that one or more of the above patent applications may be denied. We also face the risk that issued patents that we own or license may be challenged or circumvented or may otherwise not provide protection for any commercially viable products we develop.
The technologies we use in our research, as well as the drug targets we select, may infringe the patents or violate the proprietary rights of third parties. If this occurs, we may be required to obtain licenses to patents or proprietary rights of others in order to continue with the commercialization of our products.
In addition to the granted and potential patent protection, the United States, the European Union and Japan all provide data and marketing exclusivity for new medicinal compounds. If this protection is available, no competitor may use the original applicant’s data as the basis of a generic marketing application during the period of data and marketing exclusivity. This period of exclusivity is generally five years in the United States, six years in Japan and ten years in the European Union, measured from the date of FDA, or corresponding foreign regulatory authority, approval.
Elagolix, our small molecule GnRH antagonist currently in clinical trials for the treatment of endometriosis and uterine fibroids, is covered by six issued U.S. patents relating to composition of matter, pharmaceutical compositions, and methods of use. U.S. Patent Nos. 6,872,728, 7,179,815 and 7,462,625 are due to expire in 2021 (not including potential patent term extensions of up to five years) while U.S. Patent Nos. 7,056,927, 7,176,211 and 7,419,983 are due to expire in 2024 (not including potential patent term extensions of up to five years).
INGREZZA (valbenazine), our highly selective VMAT2 inhibitor, currently in clinical trials for the treatment Tourette syndrome, is covered by U.S. Patent No. 8,039,627, which expires in 2029 (not including a
14
Table of Contents
potential patent term extension of up to two years) and U.S. Patent No. 8,357,697, which expires in 2027. INGREZZA is also covered by European Patent No. 2,081,929, which expires in 2027.
Opicapone, a highly selective COMT inhibitor for Parkinson’s disease is covered by U.S. Patent No. 8,168,793, among others, which expires in 2029 (not including a potential patent term extension of up to five years).
Manufacturing and Distribution
We currently rely on, and expect to continue to rely on, contract manufacturers to produce INGREZZA, as well as for our existing and future product candidates. We believe this outsourcing manufacturing strategy will enable us to direct our financial resources to our commercialization efforts without devoting the resources and capital required to build manufacturing facilities.
We have established an internal pharmaceutical development group to develop manufacturing methods for our product candidates, to optimize manufacturing processes, and to select and transfer these manufacturing technologies to our suppliers. We have also established an internal commercial supply team to manage all aspects related to the INGREZZA commercial supply chain. We have entered into long-term contracts with multiple manufacturers to ensure adequate product supply and to mitigate risk.
There currently are a limited number of these manufacturers. Furthermore, some of the contract manufacturers that we have identified to date only have limited experience at manufacturing, formulating, analyzing and packaging our products and product candidates in quantities sufficient for conducting clinical trials or for commercialization. We attempt to acquire adequate inventory of materials and/or finished product to avoid significant supply disruption.
Additionally, we have retained third-party service providers to perform a variety of functions related to the distribution of INGREZZA, including shipping, warehousing, customer service, order-taking and processing, invoicing, collections, and other distribution-related activities.
We have entered into distribution agreements for INGREZZA with a limited number of select pharmacies (SP) and select distributors (SD), and all of our product sales are to these customers. SPs subsequently dispense INGREZZA to patients based on the fulfillment of a prescription and SDs sell INGREZZA primarily to government facilities and in-patient hospital pharmacies. Our agreements with SPs and SDs provide for transfer of title to the product at the time the product is delivered to the SP or SD. Our three largest customers represented 99% of our product revenue for the year ended December 31, 2017.
Master Manufacturing Services Agreement and Product Agreement
We entered into a Master Manufacturing Services Agreement with Patheon UK Limited (Patheon) in November 2016, and a related Amended and Restated Product Agreement in July 2017, for Patheon’s manufacture of commercial supplies of INGREZZA at Patheon’s manufacturing site. Under the terms of the agreements, we are responsible for supplying the active pharmaceutical ingredients for INGREZZA to Patheon. Patheon is responsible for manufacturing the INGREZZA capsules, conducting quality control, quality assurance, validation activities, stability testing, packaging and providing related services for the manufacture of the INGREZZA capsules.
Pursuant to the agreements, we have agreed to order from Patheon certain annual binding minimum amounts of INGREZZA capsules based on an agreed upon pricing schedule. The agreements have an initial term ending in December 2021, and will automatically renew after the initial term for successive terms of two years, unless either party gives notice of its intention to terminate the agreements within at least 18 months prior to the end of the then current term.
15
Table of Contents
Commercial Packaging Agreement
We entered into a Commercial Packaging Agreement with AndersonBrecon Inc., doing business as PCI of Illinois (PCI), in December 2016, for PCI’s commercial packaging services. Under the terms of the agreement, PCI will be responsible for, among other things, the packaging of certain of our products, tooling purchases and repair, analytical work, stability testing, auditing of suppliers and storage. We are responsible for supplying the product materials to PCI. Pursuant to the agreement, we have agreed to submit rolling forecasts, some of which will be binding on us. We will compensate PCI for services rendered, based on an agreed upon fee schedule and subject to certain price adjustments.
The agreement has an initial term ending in September 2019, unless earlier terminated in accordance with its terms. The agreement will automatically renew after the initial term for successive terms of two years, unless either party gives notice of its intention to terminate the agreement at least one year prior to the end of the then current term.
Marketing and Sales
During 2017, we established the balance of our commercial team to launch INGREZZA in TD. This commercial team is comprised of experienced professionals in marketing, access and reimbursement, managed markets, market research, commercial operations, and sales force planning and management.
We have built a specialty sales force in the United States of approximately 160 experienced sales professionals. This specialty sales force focuses on promotion to physicians who treat TD patients, primarily neurologists and psychiatrists.
In addition, our commercial infrastructure includes capabilities in manufacturing, medical affairs, quality control, and compliance.
Government Regulation
Our business activities, which include the manufacture and marketing of INGREZZA as well as our other potential products currently in research and development, are subject to extensive regulation by the United States and other countries. Regulation by government authorities in the United States and foreign countries is a significant factor in the development, manufacture, distribution, marketing and sale of our proposed products and in our ongoing research and product development activities. All of our products in development will require regulatory approval by government agencies prior to commercialization. In particular, human therapeutic products are subject to rigorous preclinical studies and clinical trials and other approval procedures of the FDA and similar regulatory authorities in foreign countries. The process of obtaining these approvals and the subsequent compliance with appropriate federal and state statutes and regulations require the expenditure of substantial time and financial resources. In the United States, various federal and state statutes and regulation also govern or influence testing, manufacturing, safety, labeling, storage, and record-keeping of human therapeutic products and their marketing. Recent federal legislation imposes additional obligations on pharmaceutical manufacturers regarding product tracking and tracing.
In addition, federal and state healthcare laws restrict business practices in the pharmaceutical industry. These laws include, without limitation, federal and state fraud and abuse laws, false claims laws, data privacy and security laws, as well as transparency laws regarding payments or other items of value provided to healthcare providers.
The federal Anti-Kickback Statute makes it illegal for any person or entity, including a prescription drug manufacturer (or a party acting on its behalf) to knowingly and willfully, directly or indirectly, solicit, receive, offer, or pay any remuneration that is intended to induce the referral of business, including the purchase, order,
16
Table of Contents
lease of any good, facility, item or service for which payment may be made under a federal healthcare program, such as Medicare or Medicaid. The term “remuneration” has been broadly interpreted to include anything of value.
Federal civil and criminal false claims laws, including the federal civil False Claims Act, and the federal civil monetary penalties law, which prohibit among other things, any person or entity from knowingly presenting, or causing to be presented, for payment to, or approval by, federal programs, including Medicare and Medicaid, claims for items or services, including drugs, that are false or fraudulent or not provided as claimed and knowingly making, or causing to be made, a false record or statement material to a false or fraudulent claim to avoid, decrease or conceal an obligation to pay money to the federal government.
The federal Health Insurance Portability and Accountability Act of 1996 (HIPAA) created additional federal criminal statutes that prohibit among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private third-party payors, knowingly and willfully stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense, and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services.
Also, many states have similar fraud and abuse statutes or regulations that may be broader in scope and may apply regardless of payor, in addition to items and services reimbursed under Medicaid and other state programs. Additionally, to the extent that our product is sold in a foreign country, we may be subject to similar foreign laws.
HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act and their implementing regulations, requires certain types of individuals and entities to abide by standards relating to the privacy and security of individually identifiable health information, including the adoption of administrative, physical and technical safeguards to protect such information. In addition, certain state laws govern the privacy and security of health information in certain circumstances, some of which are more stringent than HIPAA and many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
The federal Physician Payments Sunshine Act requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with specific exceptions, to report annually to the Centers for Medicare & Medicaid Services (CMS) information related to payments or other transfers of value made to physicians and teaching hospitals, and applicable manufacturers and applicable group purchasing organizations to report annually to CMS ownership and investment interests held by the physicians and their immediate family members.
Failure to comply with these laws, where applicable, can result in significant penalties, including the imposition of significant civil, criminal and administrative penalties, damages, monetary fines, disgorgement, individual imprisonment, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
17
Table of Contents
Preclinical studies generally are conducted in laboratory animals to evaluate the potential safety and efficacy of a product. Drug developers submit the results of preclinical studies to the FDA as a part of an IND application before clinical trials can begin in humans. Typically, clinical evaluation involves a time consuming and costly three-phase process.
| Phase I | Clinical trials are conducted with a small number of subjects to determine the early safety profile, maximum tolerated dose and pharmacological properties of the product in human volunteers. | |
| Phase II | Clinical trials are conducted with groups of patients afflicted with a specific disease in order to determine preliminary efficacy, optimal dosages and expanded evidence of safety. | |
| Phase III | Large-scale, multi-center, comparative clinical trials are conducted with patients afflicted with a specific disease in order to determine safety and efficacy as primary support for regulatory approval by the FDA to market a product candidate for a specific disease. | |
The FDA closely monitors the progress of each of the three phases of clinical trials that are conducted in the United States and may, at its discretion, re-evaluate, alter, suspend or terminate the testing based upon the data accumulated to that point and the FDA’s assessment of the risk/benefit ratio to the patient. To date, we have also conducted some of our clinical trials in Europe, Canada, Oceania and South Africa. Clinical trials conducted in foreign countries are also subject to oversight by regulatory authorities in those countries.
Once Phase III trials are completed, drug developers submit the results of preclinical studies and clinical trials to the FDA in the form of an NDA or a biologics license application for approval to commence commercial sales. In most cases, the submission of an NDA is subject to a substantial application user fee. Under the PDUFA guidelines that are currently in effect, the FDA has a goal of ten months from the date of filing of a standard NDA for a new molecular entity to review and act on the submission. This review typically takes twelve months from the date the NDA is submitted to FDA because the FDA has approximately two months to make a “filing” decision.
In addition, under the Pediatric Research Equity Act of 2003 (PREA) as amended and reauthorized, certain NDAs or supplements to an NDA must contain data that are adequate to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults or full or partial waivers from the pediatric data requirements.
The FDA also may require submission of a risk evaluation and mitigation strategy (REMS) plan to ensure that the benefits of the drug outweigh its risks. The REMS plan could include medication guides, physician communication plans, assessment plans, and/or elements to assure safe use, such as restricted distribution methods, patient registries, or other risk minimization tools.
The FDA conducts a preliminary review of all NDAs within the first 60 days after submission, before accepting them for filing, to determine whether they are sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an NDA for filing. In this event, the application must be resubmitted with the additional information. The resubmitted application is also subject to review before the FDA accepts it for filing. Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews an NDA to determine, among other things, whether the drug is safe and effective and whether the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product’s continued safety, quality and purity.
The FDA may refer an application for a novel drug to an advisory committee. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
18
Table of Contents
Before approving an NDA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with current Good Manufacturing Practice requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving an NDA, the FDA may inspect one or more clinical trial sites to assure compliance with Good Clinical Practice requirements.
After evaluating the NDA and all related information, including the advisory committee recommendation, if any, and inspection reports regarding the manufacturing facilities and clinical trial sites, the FDA may issue an approval letter, or, in some cases, a complete response letter. A complete response letter generally contains a statement of specific conditions that must be met in order to secure final approval of the NDA and may require additional clinical or preclinical testing in order for FDA to reconsider the application. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. If and when those conditions have been met to the FDA’s satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Even if the FDA approves a product, it may limit the approved indications for use of the product, require that contraindications, warnings or precautions be included in the product labeling, require that post-approval studies, including Phase IV clinical trials, be conducted to further assess a drug’s safety after approval, require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution and use restrictions or other risk management mechanisms under a REMS, which can materially affect the potential market and profitability of the product. The FDA may prevent or limit further marketing of a product based on the results of post-marketing studies or surveillance programs. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval.
We will also have to complete an approval process similar to that in the United States in virtually every foreign target market for our products in order to commercialize our product candidates in those countries. The approval procedure and the time required for approval vary from country to country and may involve additional testing. Foreign approvals may not be granted on a timely basis, or at all. In addition, regulatory approval of prices is required in most countries other than the United States. The resulting prices may not be sufficient to generate an acceptable return to us or our corporate collaborators.
Special FDA Expedited Review and Approval Programs
The FDA has various programs, including fast track designation, accelerated approval, priority review, and breakthrough therapy designation, which are intended to expedite or simplify the process for the development and FDA review of drugs that are intended for the treatment of serious or life threatening diseases or conditions and demonstrate the potential to address unmet medical needs. The purpose of these programs is to provide important new drugs to patients earlier than under standard FDA review procedures.
To be eligible for a fast track designation, the FDA must determine, based on the request of a sponsor, that a product is intended to treat a serious or life-threatening disease or condition and demonstrates the potential to address an unmet medical need. The FDA will determine that a product will fill an unmet medical need if it will provide a therapy where none exists or provide a therapy that may be potentially superior to existing therapy based on efficacy or safety factors. The FDA may review sections of the NDA for a fast track product on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the NDA, the FDA agrees to accept sections of the NDA and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the NDA.
The FDA may give a priority review designation to drugs that offer major advances in treatment, or provide a treatment where no adequate therapy exists. A priority review means that the goal for the FDA to review an
19
Table of Contents
application is six months, rather than the standard review of ten months under current PDUFA guidelines. These six and ten month review periods are measured from the filing date rather than the receipt date for NDAs for new molecular entities, which typically adds approximately two months to the timeline for review and decision from the date of submission. Most products that are eligible for fast track designation are also likely to be considered appropriate to receive a priority review.
In addition, products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may be eligible for accelerated approval and may be approved on the basis of adequate and well-controlled clinical trials establishing that the drug product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA may require a sponsor of a drug receiving accelerated approval to perform post-marketing studies to verify and describe the predicted effect on irreversible morbidity or mortality or other clinical endpoint, and the drug may be subject to accelerated withdrawal procedures.
A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. Drugs designated as breakthrough therapies are also eligible for accelerated approval. The FDA must take certain actions, such as holding timely meetings and providing advice, intended to expedite the development and review of an application for approval of a breakthrough therapy.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.
Post-Approval Requirements
Drugs manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior FDA review and approval. There also are continuing, annual user fee requirements for any marketed products and the establishments at which such products are manufactured, as well as new application fees for supplemental applications with clinical data.
The FDA may impose a number of post-approval requirements as a condition of approval of an NDA. For example, the FDA may require post-marketing testing, including Phase IV clinical trials, and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies, and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with current Good Manufacturing Practices (cGMP) requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP requirements and impose reporting and documentation requirements upon the sponsor and any third-party manufacturers that the sponsor may decide to use. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance.
20
Table of Contents
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, among other things:
| • | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| • | fines, warning letters or holds on post-approval clinical trials; |
| • | refusal of the FDA to approve pending NDAs or supplements to approved NDAs, or suspension or revocation of product approvals; |
| • | product seizure or detention, or refusal to permit the import or export of products; or |
| • | injunctions or the imposition of civil or criminal penalties. |
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability.
Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any product candidates for which we obtain regulatory approval. In the United States and markets in other countries, sales of any products for which we receive regulatory approval will depend, in part, on the extent to which third-party payors provide coverage and establish adequate reimbursement levels for such drug products.
In the United States, third-party payors include federal and state healthcare programs, government authorities, private managed care providers, private health insurers and other organizations. Third-party payors are increasingly challenging the price, examining the medical necessity and reviewing the cost-effectiveness of medical drug products and medical services, in addition to questioning their safety and efficacy. Such payors may limit coverage to specific drug products on an approved list, also known as a formulary, which might not include all of the FDA-approved drugs for a particular indication. We may need to conduct expensive pharmaco-economic studies in order to demonstrate the medical necessity and cost-effectiveness of our products, in addition to the costs required to obtain the FDA approvals. Nonetheless, our products or product candidates, including INGREZZA, may not be considered medically necessary or cost-effective.
Moreover, the process for determining whether a third-party payor will provide coverage for a drug product may be separate from the process for setting the price of a drug product or for establishing the reimbursement rate that such a payor will pay for the drug product. A payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Further, one payor’s determination to provide coverage for a drug product does not assure that other payors will also provide coverage for the drug product. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development.
The marketability of any product or product candidates for which we or our collaborators receive regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide adequate coverage and reimbursement. In addition, emphasis on managed care in the United States has increased and we
21
Table of Contents
expect will continue to increase the pressure on pharmaceutical pricing. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
Healthcare Reform
The United States and some foreign jurisdictions are considering or have enacted a number of legislative and regulatory proposals to change the healthcare system in ways that could affect our ability to sell our products profitably. Among policy makers and payors in the United States and elsewhere, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality or expanding access. In the United States, the pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives.
By way of example, in March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, collectively the ACA, was signed into law, which intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add transparency requirements for the healthcare and health insurance industries, impose taxes and fees on the health industry and impose additional health policy reforms. Among the provisions of the ACA of importance to our potential drug candidates are:
| • | an annual, nondeductible fee on any entity that manufactures or imports specified branded prescription drugs and biologic agents, apportioned among these entities according to their market share in certain government healthcare programs; |
| • | an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program to 23.1% and 13.0% of the average manufacturer price for branded and generic drugs, respectively; |
| • | a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; |
| • | extension of a manufacturer’s Medicaid rebate liability to covered drugs dispensed to individuals who are enrolled in Medicaid managed care organizations; |
| • | expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to certain individuals with income at or below 133% of the federal poverty level, thereby potentially increasing a manufacturer’s Medicaid rebate liability; |
| • | a new Medicare Part D coverage gap discount program, in which manufacturers must agree to offer 50% point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for a manufacturer’s outpatient drugs to be covered under Medicare Part D; |
| • | expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program; and |
| • | a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research. |
We expect that the ACA, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and lower reimbursement, and in additional downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare or other government-funded programs may result in a similar reduction in payments from private payor.
Some of the provisions of the ACA have yet to be fully implemented, and there have been legal and political challenges to certain aspects of the ACA. Since January 2017, President Trump has signed two executive orders
22
Table of Contents
and other directives designed to delay, circumvent, or loosen certain requirements mandated by the ACA. Concurrently, Congress has considered legislation that would repeal or repeal and replace all or part of the ACA. While Congress has not passed repeal legislation, the newly enacted federal income tax law includes a provision repealing, effective January 1, 2019, the tax-based shared responsibility payment imposed by the ACA on certain individuals who fail to maintain qualifying health coverage for all or part of a year that is commonly referred to as the “individual mandate”. Congress may consider other legislation to repeal or replace elements of the ACA.
Other legislative changes have been proposed and adopted since the ACA was enacted. These changes include aggregate reductions to Medicare payments to providers of up to 2% per fiscal year pursuant to the Budget Control Act of 2011, which began in 2013 and will remain in effect through 2025 unless additional Congressional action is taken. The American Taxpayer Relief Act of 2012, among other things, further reduced Medicare payments to several providers, including hospitals and cancer treatment centers, increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
Additional changes that may affect our business include the expansion of new programs such as Medicare payment for performance initiatives for physicians under the Medicare Access and CHIP Reauthorization Act of 2015(MACRA), which will be fully implemented in 2019. At this time it is unclear how the introduction of the Medicare quality payment program will impact overall physician reimbursement. Also, there has been heightened governmental scrutiny recently over pharmaceutical pricing practices in light of the rising cost of prescription drugs and biologics. Such scrutiny has resulted in several recent Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for products. At the federal level, Congress and the Trump administration have each indicated that it will continue to seek new legislative and/or administrative measures to control drug costs. At the state level, legislatures have become increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing.
Competition
The biotechnology and pharmaceutical industries are subject to rapid and intense technological change. We face, and will continue to face, competition in the development and marketing of our product candidates from biotechnology and pharmaceutical companies, research institutions, government agencies and academic institutions. Competition may also arise from, among other things:
| • | other drug development technologies; |
| • | methods of preventing or reducing the incidence of disease, including vaccines; and |
| • | new small molecule or other classes of therapeutic agents. |
Developments by others may render our products, product candidates or technologies obsolete or noncompetitive. We are performing research on or developing products for the treatment of several disorders including endometriosis, TD, uterine fibroids, Tourette syndrome, classic congenital adrenal hyperplasia, essential tremor, pain, and other neurological and endocrine-related diseases and disorders.
In April 2017, INGREZZA (valbenazine), was approved by the FDA for TD, and is also currently in Phase IIb development for Tourette syndrome. There are currently two FDA approved drug therapies for TD; INGREZZA® and AUSTEDO® (deutetrabenazine), a deuterium labeled VMAT2 inhibitor which was developed by Teva Pharmaceutical Industries Ltd. (Teva). In addition, off-label treatment regimens for TD consist of utilizing various atypical antipsychotic medications (e.g., clozapine), benzodiazepines (off-label) or botulinum toxin injections to treat the movements associated with TD.
23
Table of Contents
Generic neuroleptic medications (pimozide and haloperidol) as well as ABILIFY® (apriprizole) are approved by the FDA to control the tics associated with Tourette syndrome. Teva is also investigating AUSTEDO for utility in Tourette syndrome. Other potential indications for our VMAT2 inhibitor include the chorea associated with Huntington’s disease, schizophrenia and tardive dystonia. Currently, Austedo (deutetrabenazine) and XENAZINE® (tetrabenazine), marketed by Lundbeck, as well as its generic alternatives to Xenazine, are approved for the chorea associated with Huntington’s disease.
We, in conjunction with our partner AbbVie, are developing elagolix for the treatment of heavy menstrual bleeding associated with uterine fibroids. There are no current pharmaceutical therapies approved in the United States for the chronic treatment of uterine fibroids. During 2017, Allergan Pharmaceuticals, Inc. filed an NDA with the FDA for potential use of ESMYA® (ulipristal), a selective progesterone receptor modulator in the treatment of heavy menstrual bleeding associated with uterine fibroids. ObsEva SA has initiated a Phase IIb endometriosis study with its GnRH receptor antagonist OBE2109 and has initiated Phase III studies of uterine fibroids patients with the same molecule. Myovant Sciences, Inc. is investigating its GnRH receptor antagonist, relugolix, in Phase III trials of endometriosis, uterine fibroids and prostate cancer patients. LUPRON DEPOT®, marketed by AbbVie, is approved for short-term use to improve the outcome of uterine fibroid surgery. However, approximately 250,000 hysterectomies are performed annually in the United States as a direct result of uterine fibroids, as well as myomectomies (surgery) to remove the fibroids. Our oral small molecule pharmaceutical agent, elagolix, would compete directly with these current invasive standards of care.
LUPRON DEPOT and SYNAREL® and depo-subQ provera104®, marketed by Pfizer, are products that have been approved for the treatment of endometriosis, infertility, and central precocious puberty. These drugs, and any generic alternatives, may compete with any small molecule non-peptide GnRH antagonists we, in conjunction with our collaborative partner AbbVie, develop for these indications. Approximately 130,000 hysterectomies are performed annually in the United States as a direct result of endometriosis, as well as a significant number of laparoscopic procedures to ablate endometrial explants. Our oral small molecule pharmaceutical agent, elagolix, would also compete directly with these current invasive standards of care.
Opicapone is a COMT inhibitor to be utilized as an adjunct therapy in the treatment of Parkinson’s disease. COMT inhibitors prolong the duration of effect of levodopa which is the primary treatment option for Parkinson’s disease patients. There are currently two FDA approved COMT inhibitors, COMTAN® (entacapone) originally developed by Orion Pharma and TASMAR® (tolcapone) originally developed by Hoffman-LaRoche Inc. Opicapone would compete directly with these two drugs and their generic equivalents.
NBI-74788 is currently being investigated for the treatment of classic CAH, for which there are limited therapies. High doses of corticosteroids are the current standard of care to both correct the endogenous cortisol deficiency as well as reduce the excessive ACTH levels. However, the level of dose as well as the duration of steroid use required to suppress ACTH is well above the normal physiological level of cortisol; resulting in metabolic syndrome, bone loss, growth impairment, and Cushing’s syndrome as common and serious side effects. Both Millendo Therapeutics, with its ACAT1 inhibitor ATR-101, and Spruce Biosciences, with its CRF antagonist SPR001 are in clinical development for classic CAH.
If one or more of these competitive products or programs are successful, it may reduce or eliminate the market for our products.
Compared to us, many of our competitors and potential competitors have substantially greater:
| • | capital resources; |
| • | commercial experience; |
| • | research and development resources, including personnel and technology; |
| • | regulatory experience; |
24
Table of Contents
| • | preclinical study and clinical testing experience; |
| • | manufacturing and marketing experience; and |
| • | production facilities. |
Any of these competitive factors could harm our business, prospects, financial condition and results of operations, which could negatively affect our stock price.
Employees
As of December 31, 2017, we had approximately 400 full-time employees. None of our employees are represented by a collective bargaining arrangement, and we believe our relationship with our employees is good. In addition, we rely on a number of consultants to assist us.
Insurance
We maintain limited product liability insurance coverage for our clinical trials in the amount of $10 million per occurrence and $10 million in the aggregate. Upon FDA approval of INGREZZA we expanded our insurance coverage to include product liability insurance related to the sale of INGREZZA in the amount of $20 million per occurrence and $20 million in the aggregate. However, insurance coverage is becoming increasingly expensive, and we may not be able to obtain or maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability.
Available Information
Our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to reports filed pursuant to Sections 13(a) and 15(d) of the Securities Exchange Act of 1934, as amended, are available free of charge on our website at www.neurocrine.com, as soon as reasonably practicable after such reports are available on the Securities and Exchange Commission website at www.sec.gov.
Additionally, copies of our Annual Report will be made available, free of charge, upon written request.
25
Table of Contents
| ITEM 1A. | RISK FACTORS |
The following information sets forth risk factors that could cause our actual results to differ materially from those contained in forward-looking statements we have made in this Annual Report on Form 10-K and those we may make from time to time. If any of the following risks actually occur, our business, operating results, prospects or financial condition could be harmed. Additional risks not presently known to us, or that we currently deem immaterial, may also affect our business operations.
Risks Related to Our Company
We have limited marketing experience, and have only recently begun establishing our sales force, distribution and reimbursement capabilities, and we may not be able to successfully commercialize INGREZZA, or any of our product candidates if they are approved in the future.
Our ability to produce revenues ultimately depends on our ability to sell our products and secure adequate third-party reimbursement if and when they are approved by the FDA. We currently have limited experience in marketing and selling pharmaceutical products. With respect to INGREZZA in particular, we have only recently hired our sales force to sell INGREZZA, and have only recently begun establishing our distribution and reimbursement capabilities, all of which will be necessary to successfully commercialize INGREZZA. While we have recently hired personnel, and engaged consultants with experience marketing and selling pharmaceutical products, there can be no guarantee that we will be able to establish or maintain the personnel, systems, arrangements and capabilities necessary to successfully commercialize INGREZZA or any product candidate approved by the FDA in the future. If we fail to establish or maintain successful marketing, sales and reimbursement capabilities or fail to enter into successful marketing arrangements with third parties, our product revenues may suffer.
We currently depend on a single source supplier for each of the production of INGREZZA and its active pharmaceutical ingredients. The loss of either of these suppliers, or delays or problems in the supply of INGREZZA, could materially and adversely affect our ability to successfully commercialize INGREZZA.
The manufacture of pharmaceutical products requires significant expertise and capital investment, including the development of process controls required to consistently produce the active pharmaceutical ingredients and the finished product in sufficient quantities while meeting detailed product specifications on a repeated basis. Manufacturers of pharmaceutical products may encounter difficulties in production, including difficulties with production costs and yields, process controls, quality control and quality assurance, including testing of stability, impurities and impurity levels and other product specifications by validated test methods, and compliance with strictly enforced United States, state and non-United States regulations. If our third-party suppliers for INGREZZA encounter these or any other manufacturing, quality or compliance difficulties, we may be unable to meet commercial demand for INGREZZA, which could materially and adversely affect our ability to successfully commercialize INGREZZA.
In addition, if our suppliers fail or refuse to supply us with INGREZZA or its active pharmaceutical ingredient for any reason, it would take a significant amount of time and expense to qualify a new supplier. The FDA and similar international regulatory bodies must approve manufacturers of the active and inactive pharmaceutical ingredients and certain packaging materials used in pharmaceutical products. The loss of a supplier could require us to obtain regulatory clearance and to incur validation and other costs associated with the transfer of the active pharmaceutical ingredients or product manufacturing processes. If there are delays in qualifying new suppliers or facilities or a new supplier is unable to meet FDA or a similar international regulatory body’s requirements for approval, there could be a shortage of INGREZZA, which could materially and adversely affect our ability to successfully commercialize INGREZZA.
26
Table of Contents
We have no manufacturing capabilities. If third-party manufacturers of INGREZZA or any of our product candidates fail to devote sufficient time and resources to our concerns, or if their performance is substandard, our clinical trials and product introductions may be delayed and our costs may rise.
We have in the past utilized, and intend to continue to utilize, third-party manufacturers to produce the drug compounds we use in our clinical trials and for the commercialization of our products. We have limited experience in manufacturing products for commercial purposes and do not currently have any manufacturing facilities. Consequently, we depend on, and will continue to depend on, several contract manufacturers for all production of products for development and commercial purposes, including INGREZZA. If we are unable to obtain or retain third-party manufacturers, we will not be able to develop or commercialize our products, including INGREZZA. The manufacture of our products for clinical trials and commercial purposes is subject to specific FDA regulations, including current Good Manufacturing Practice regulations. Our third-party manufacturers might not comply with FDA regulations relating to manufacturing our products for clinical trials and commercial purposes or other regulatory requirements now or in the future. Our reliance on contract manufacturers also exposes us to the following risks:
| • | contract manufacturers may encounter difficulties in achieving volume production, quality control and quality assurance, and also may experience shortages in qualified personnel. As a result, our contract manufacturers might not be able to meet our clinical schedules or adequately manufacture our products in commercial quantities when required; |
| • | switching manufacturers may be difficult because the number of potential manufacturers is limited. It may be difficult or impossible for us to find a replacement manufacturer quickly on acceptable terms, or at all; |
| • | our contract manufacturers may not perform as agreed or may not remain in the contract manufacturing business for the time required to successfully produce, store or distribute our products; and |
| • | drug manufacturers are subject to ongoing periodic unannounced inspection by the FDA, the United States Drug Enforcement Administration, and other agencies to ensure strict compliance with current Good Manufacturing Practices and other government regulations and corresponding foreign standards. We do not have control over third-party manufacturers’ compliance with these regulations and standards. |
Our current dependence upon third parties for the manufacture of our products may reduce our profit margin, if any, on the sale of INGREZZA or our future products and our ability to develop and deliver products on a timely and competitive basis.
Our clinical trials may fail to demonstrate the safety and efficacy of our product candidates, which could prevent or significantly delay their regulatory approval.
Before obtaining regulatory approval for the sale of any of our potential products, we must subject these product candidates to extensive preclinical and clinical testing to demonstrate their safety and efficacy for humans. Clinical trials are expensive, time-consuming and may take years to complete.
In connection with the clinical trials of our product candidates, we face the risks that:
| • | the FDA or similar foreign regulatory authority may not allow an Investigational New Drug (IND) application or foreign equivalent filings required to initiate human clinical studies for our drug candidates or the FDA may require additional preclinical studies as a condition of the initiation of Phase I clinical studies, or additional clinical studies for progression from Phase I to Phase II, or Phase II to Phase III, or for NDA approval; |
| • | the product candidate may not prove to be effective or as effective as other competing product candidates; |
27
Table of Contents
| • | we may discover that a product candidate may cause harmful side effects or results of required toxicology studies may not be acceptable to the FDA; |
| • | the results may not replicate the results of earlier, smaller trials; |
| • | the FDA or similar foreign regulatory authorities may require use of new or experimental endpoints that may prove insensitive to treatment effects; |
| • | we or the FDA or similar foreign regulatory authorities may suspend the trials; |
| • | the results may not be statistically significant; |
| • | patient recruitment may be slower than expected; |
| • | patients may drop out of the trials; and |
| • | regulatory requirements may change. |
These risks and uncertainties impact all of our clinical programs. Specifically, our VMAT2 inhibitor program will be impacted if any of the events above lead to delayed timelines for the enrollment in, or completion of, clinical trials of INGREZZA for Tourette syndrome. With respect to our gonadotropin-releasing hormone (GnRH) program with AbbVie, any of the clinical, regulatory or operational events described above could delay timelines for the elagolix endometriosis program, for which an NDA has been submitted to the FDA; or the completion of the Phase III uterine fibroids program. Likewise, any of these events could change our planned clinical and regulatory activities for the opicapone program in Parkinson’s disease. Additionally, any of these events described above could result in suspension of a program and/or obviate any filings for necessary regulatory approvals.
In addition, late-stage clinical trials are often conducted with patients having the most advanced stages of disease. During the course of treatment, these patients can die or suffer other adverse medical effects for reasons that may not be related to the pharmaceutical agent being tested but which can nevertheless adversely affect clinical trial results. Any failure or substantial delay in completing clinical trials for our product candidates may severely harm our business.
Even if the clinical trials are successfully completed, we cannot guarantee that the FDA or foreign regulatory authorities will interpret the results as we do, and more trials could be required before we submit our product candidates for approval. To the extent that the results of the trials are not satisfactory to the FDA or foreign regulatory authorities for support of a marketing application, approval of our product candidates may be significantly delayed, or we may be required to expend significant additional resources, which may not be available to us, to conduct additional trials in support of potential approval of our product candidates.
We depend on our current collaborators for the development and commercialization of our product candidates that we out-license and in-license, and may need to enter into future collaborations to develop and commercialize certain of our product candidates.
Our strategy for fully developing and commercializing elagolix is dependent upon maintaining our current collaboration agreement with AbbVie. This collaboration agreement provides for significant future payments should certain development, regulatory and commercial milestones be achieved, and royalties on future sales of elagolix. Under this agreement, AbbVie is responsible for, among other things, conducting clinical trials and obtaining required regulatory approvals for elagolix; as well as manufacturing and commercialization of elagolix in the event it receives regulatory approval.
Because of our reliance on AbbVie, the development and commercialization of elagolix could be substantially delayed, and our ability to receive future funding could be substantially impaired, if AbbVie:
| • | failed to gain the requisite regulatory approval of elagolix; |
28
Table of Contents
| • | did not successfully launch and commercialize elagolix; |
| • | did not conduct its collaborative activities in a timely manner; |
| • | did not devote sufficient time and resources to our partnered program; |
| • | terminated its agreement with us; |
| • | developed, either alone or with others, products that may compete with elagolix; |
| • | disputed our respective allocations of rights to any products or technology developed during our collaboration; or |
| • | merged with a third party that wants to terminate our agreement. |
In March 2015, we entered into a collaboration and license agreement with Mitsubishi Tanabe to develop and commercialize INGREZZA in Japan and other select Asian markets. We will rely on Mitsubishi Tanabe to achieve certain development, regulatory and commercial milestones which, if achieved, could generate significant future revenue for us. Our collaboration with Mitsubishi Tanabe is subject to risks and uncertainties similar to those described above. In addition, we may need to enter into other out-licensing collaborations to assist in the development and commercialization of other product candidates we are developing now or may develop in the future, and any such future collaborations would be subject to similar risks and uncertainties.
In February 2017, we entered into a license agreement with BIAL for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. Under the terms of the agreement, we are responsible for the management of all opicapone development and commercialization activities; however, we will depend on BIAL to supply all drug product and investigation medicinal product for our development and commercialization activities. In addition, pursuant to the license agreement, the parties have established a joint steering committee with overall coordination and strategic oversight over activities under the agreement and to provide a forum for regular exchange of information, and BIAL has the right to co-promote licensed products during certain periods of time and to engage in certain marketing-related activities in cooperation with us. Accordingly, our strategy for developing and commercializing opicapone is dependent upon maintaining our current collaboration with BIAL. Because of our reliance on BIAL for certain aspects related to the development and commercialization of opicapone, any disagreement with BIAL, or BIAL’s decision to not devote sufficient time and resources to our collaboration or to not conduct activities in a timely manner, could substantially delay and/or prohibit our ability to develop and commercialize opicapone.
These issues and possible disagreements with AbbVie, Mitsubishi Tanabe, BIAL or any future corporate collaborators could lead to delays in the collaborative research, development or commercialization of our product candidates. Furthermore, disagreements with these parties could require or result in litigation or arbitration, which would be time-consuming and expensive. If any of these issues arise, it may delay the development and commercialization of drug candidates and, ultimately, our generation of product revenues.
We do not and will not have access to all information regarding the product candidates we licensed to AbbVie.
We do not and will not have access to all information regarding the products being developed and potentially commercialized by AbbVie, including potentially material information about clinical trial design and execution, safety reports from clinical trials, spontaneous safety reports if a product candidate is later approved and marketed, regulatory affairs, process development, manufacturing, marketing and other areas known by AbbVie. In addition, we have confidentiality obligations under our agreement with AbbVie. Thus, our ability to keep our shareholders informed about the status of product candidates under our collaboration with AbbVie will be limited by the degree to which AbbVie keeps us informed and allows us to disclose such information to the public. If AbbVie fails to keep us informed about the clinical development and regulatory approval of our collaboration and product candidates licensed to it, we may make operational and investment decisions that we
29
Table of Contents
would not have made had we been fully informed, which may materially and adversely affect our business and operations.
In April 2017, we received FDA approval for INGREZZA for TD and that approval subjects us to ongoing obligations and continued regulatory review, which may result in significant additional expense and market withdrawal. Additionally, our other product candidates, if approved, could be subject to labeling and other restrictions and market withdrawal and we may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our products.
We received FDA regulatory approval for INGREZZA in April 2017. This approval and other regulatory approvals for any of our product candidates may also be subject to limitations on the approved indicated uses for which the product may be marketed or to the conditions of approval, or contain requirements for potentially costly post-marketing testing, including Phase IV clinical trials, and surveillance to monitor the safety and efficacy of the product candidate. With respect to the FDA’s approval of INGREZZA for TD, we are subject to certain post-marketing requirements and commitments. Failure to comply with these post-marketing requirements and commitments could result in withdrawal of our marketing approval for INGREZZA. In addition, with respect to INGREZZA, and any product candidate that the FDA or a comparable foreign regulatory authority approves, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and recordkeeping for the product will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with current Good Manufacturing Practices for any clinical trials that we conduct post-approval. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with our third-party manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
| • | restrictions on the marketing or manufacturing of the product, withdrawal of the product from the market, or voluntary or mandatory product recalls; |
| • | fines, warning letters or holds on clinical trials; |
| • | refusal by the FDA to approve pending applications or supplements to approved applications filed by us, or suspension or revocation of product license approvals; |
| • | product seizure or detention, or refusal to permit the import or export of products; and |
| • | product injunctions or the imposition of civil or criminal penalties. |
The FDA’s policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of any of our product candidates or future indications for currently approved products. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained, which would adversely affect our business, prospects and ability to achieve or sustain profitability.
Governmental and third-party payors may impose sales and pharmaceutical pricing controls on our products or limit coverage and/or reimbursement for our products that could limit our product revenues and delay sustained profitability.
Our ability to commercialize any products successfully, including INGREZZA, will depend in part on the extent to which coverage and adequate reimbursement for these products and related treatments will be available. The continuing efforts of government and third-party payors to contain or reduce the costs of health care through various means may reduce our potential revenues. These payors’ efforts could decrease the price that we receive for any products we may develop and sell in the future.
30
Table of Contents
Assuming we obtain coverage for a given product by a third-party payor, the resulting reimbursement payment rates may not be adequate or may require co-payments that patients find unacceptably high. Patients who are prescribed medications for the treatment of their conditions, and their prescribing physicians, generally rely on third-party payors to reimburse all or part of the costs associated with their prescription drugs. Patients are unlikely to use our products unless coverage is provided and reimbursement is adequate to cover all or a significant portion of the cost of our products. Coverage decisions may depend upon clinical and economic standards that disfavor new drug products when more established or lower cost therapeutic alternatives are already available or subsequently become available regardless of whether they are approved by the FDA for that particular use.
Government authorities and other third-party payors are developing increasingly sophisticated methods of controlling healthcare costs, such as by limiting coverage and the amount of reimbursement for particular medications. Further, no uniform policy requirement for coverage and reimbursement for drug products exists among third-party payors in the United States. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our products to each payor separately, with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance. In addition, communications from government officials regarding health care costs and pharmaceutical pricing could have a negative impact on our stock price, even if such communications do not ultimately impact coverage or reimbursement decisions for our products.
There may also be significant delays in obtaining coverage and reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which the drug is approved by the FDA or comparable foreign regulatory authorities. Moreover, eligibility for coverage and reimbursement does not imply that a drug will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. If coverage and reimbursement are not available or reimbursement is available only to limited levels, we may not successfully commercialize INGREZZA or any other product candidate for which we obtain marketing approval. Our inability to promptly obtain coverage and profitable reimbursement rates from both government-funded and private payors for any approved products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
If physicians and patients do not accept INGREZZA or any of our other products, we may not generate sufficient revenue.
The commercial success of INGREZZA or any of our other products, if approved for marketing, will depend upon the acceptance of those products as safe and effective by the medical community and patients.
The market acceptance of INGREZZA or any of our other products could be affected by a number of factors, including:
| • | the timing of receipt of marketing approvals for indications; |
| • | the safety and efficacy of the products; |
| • | the pricing of our products; |
| • | the availability of coverage and adequate reimbursement for the products; |
| • | the success of existing products addressing our target markets or the emergence of equivalent or superior products; and |
| • | the cost-effectiveness of the products. |
In addition, market acceptance depends on the effectiveness of our marketing strategy and distribution support, and, to date, although we have hired experienced sales and marketing professionals, we have very
31
Table of Contents
limited sales and marketing experience. If the medical community and patients do not ultimately accept our products as being safe, effective, superior and/or cost-effective, we may not generate sufficient revenue.
Because the development of our product candidates is subject to a substantial degree of technological uncertainty, we may not succeed in developing any of our product candidates.
All of our product candidates are currently in research or clinical development with the exception of INGREZZA, which has been approved by the FDA for TD. Only a small number of research and development programs ultimately result in commercially successful drugs. Potential products that appear to be promising at early stages of development may not reach the market for a number of reasons. These reasons include the possibilities that the potential products may:
| • | be found ineffective or cause harmful side effects during preclinical studies or clinical trials; |
| • | fail to receive necessary regulatory approvals on a timely basis or at all; |
| • | be precluded from commercialization by proprietary rights of third parties; |
| • | be difficult to manufacture on a large scale; or |
| • | be uneconomical to commercialize or fail to achieve market acceptance. |
If any of our product candidates encounters any of these potential problems, we may never successfully market that product candidate.
Our indebtedness and liabilities could limit the cash flow available for our operations, expose us to risks that could adversely affect our business, financial condition and results of operations.
To date, we have sold $517.5 million aggregate principal amount of 2.25% convertible senior notes due 2024 (2024 Notes). We may also incur additional indebtedness to meet future financing needs. Our indebtedness could have significant negative consequences for our security holders and our business, results of operations and financial condition by, among other things:
| • | increasing our vulnerability to adverse economic and industry conditions; |
| • | limiting our ability to obtain additional financing; |
| • | requiring the dedication of a substantial portion of our cash flow from operations to service our indebtedness, which will reduce the amount of cash available for other purposes; |
| • | limiting our flexibility to plan for, or react to, changes in our business; |
| • | diluting the interests of our existing stockholders as a result of issuing shares of our common stock upon conversion of the 2024 Notes; and |
| • | placing us at a possible competitive disadvantage with competitors that are less leveraged than us or have better access to capital. |
Our business may not generate sufficient funds, and we may otherwise be unable to maintain sufficient cash reserves, to pay amounts due under the 2024 Notes and any additional indebtedness that we may incur. In addition, our cash needs may increase in the future. In addition, any future indebtedness that we may incur may contain financial and other restrictive covenants that limit our ability to operate our business, raise capital or make payments under our other indebtedness. If we fail to comply with these covenants or to make payments under our indebtedness when due, then we would be in default under that indebtedness, which could, in turn, result in that and our other indebtedness becoming immediately payable in full.
32
Table of Contents
The conditional conversion feature of the 2024 Notes, if triggered, may adversely affect our financial condition and operating results.
In the event the conditional conversion feature of the 2024 Notes is triggered, holders of 2024 Notes will be entitled to convert their 2024 Notes at any time during specified periods at their option. If one or more of the holders of the 2024 Notes elects to convert their notes, unless we satisfy our conversion obligation by delivering only shares of our common stock, we would be required to settle all or a portion of our conversion obligation through the payment of cash, which could adversely affect our liquidity. Furthermore, even if holders of the 2024 Notes did not elect to convert their notes, we could be required under applicable accounting rules to reclassify all or a portion of the outstanding principal of the 2024 Notes as a current rather than long-term liability, which would result in a material reduction of our net working capital.
We have a history of losses and expect to increase our expenses for the foreseeable future, and we may never achieve sustained profitability.
Since our inception, we have incurred significant net losses and negative cash flow from operations. As a result of historical operating losses, we had an accumulated deficit of approximately $1.2 billion as of December 31, 2017. We do not expect to be profitable for the year ending December 31, 2018.
In April 2017, we received FDA approval of INGREZZA for TD, however we have not yet obtained regulatory approvals for any other product candidates. Even if we succeed in commercializing INGREZZA or developing and commercializing any of our other product candidates, we may not be profitable. We also expect to continue to incur significant operating and capital expenditures as we:
| • | commercialize INGREZZA for TD; |
| • | seek regulatory approvals for our product candidates; |
| • | develop, formulate, manufacture and commercialize our product candidates; |
| • | in-license or acquire new product development opportunities; |
| • | implement additional internal systems and infrastructure; and |
| • | hire additional clinical, scientific, sales and marketing personnel. |
We expect to increase our expenses and other investments in the coming years as we fund our operations, in-licensing or acquisition opportunities, and capital expenditures. We will need to generate significant revenues to achieve and maintain profitability and positive cash flow on an annual basis. We may not be able to generate these revenues, and we may never achieve profitability on an annual basis in the future. Our failure to achieve or maintain profitability on an annual basis could negatively impact the market price of our common stock. Even if we become profitable on an annual basis, we cannot assure you that we would be able to sustain or increase profitability on an annual basis.
We have recently increased the size of our organization, and will need to continue to increase the size of our organization. We may encounter difficulties with managing our growth, which could adversely affect our results of operations.
As of December 31, 2017, we had approximately 400 employees. Although we substantially increased the size of our organization during 2017, we may need to add additional qualified personnel and resources, especially now that we have a commercial sales force. Our current infrastructure may be inadequate to support our development and commercialization efforts and expected growth. Future growth will impose significant added responsibilities on members of management, including the need to identify, recruit, maintain and integrate additional employees, and may take time away from running other aspects of our business, including development and commercialization of our product candidates.
33
Table of Contents
Our future financial performance and our ability to commercialize INGREZZA and any other product candidates that receive regulatory approval will depend, in part, on our ability to manage any future growth effectively. In particular, as we commercialize INGREZZA, we will need to support the training and ongoing activities of our sales force and will likely need to continue to expand the size of our employee base for managerial, operational, financial and other resources. To that end, we must be able to:
| • | manage our development efforts effectively; |
| • | integrate additional management, administrative and manufacturing personnel; |
| • | further develop our marketing and sales organization; and |
| • | maintain sufficient administrative, accounting and management information systems and controls. |
We may not be able to accomplish these tasks or successfully manage our operations and, accordingly, may not achieve our research, development, and commercialization goals. Our failure to accomplish any of these goals could harm our financial results and prospects.
We license some of our core technologies and drug candidates from third parties. If we default on any of our obligations under those licenses, or violate the terms of these licenses, we could lose our rights to those technologies and drug candidates or be forced to pay damages.
We are dependent on licenses from third parties for some of our key technologies. These licenses typically subject us to various commercialization, reporting and other obligations. If we fail to comply with these obligations, we could lose important rights. If we were to default on our obligations under any of our licenses, we could lose some or all of our rights to develop, market and sell products covered by these licenses. For example, BIAL may terminate our license agreement, pursuant to which we have rights to develop and commercialize opicapone, if we fail to use commercially reasonable efforts, fail to file an NDA for a licensed product by a specified date, or otherwise breach the license agreement. In addition, if we were to violate any of the terms of our licenses, we could become subject to damages. For example, on December 1, 2015, The Mount Sinai School of Medicine of the City University of New York (Mount Sinai) filed a complaint against us, seeking unspecified monetary damages, future sublicensing fees and attorney’s fees, alleging that we violated the terms of our license with Mount Sinai by inappropriately sublicensing Mount Sinai technology to AbbVie. While we believe that we have meritorious defenses to the claims made in the complaint and intend to vigorously defend ourselves against such claims, we are not able to predict the ultimate outcome of this action. Likewise, if we were to lose our rights under a license to use proprietary research tools, it could adversely affect our existing collaborations or adversely affect our ability to form new collaborations. We also face the risk that our licensors could, for a number of reasons, lose patent protection or lose their rights to the technologies we have licensed, thereby impairing or extinguishing our rights under our licenses with them.
The independent clinical investigators and contract research organizations that we rely upon to conduct our clinical trials may not be diligent, careful or timely, and may make mistakes, in the conduct of our trials.
We depend on independent clinical investigators and contract research organizations (CROs) to conduct our clinical trials under their agreements with us. The investigators are not our employees, and we cannot control the amount or timing of resources that they devote to our programs. If our independent investigators fail to devote sufficient time and resources to our drug development programs, or if their performance is substandard, or not in compliance with Good Clinical Practices, it may delay or prevent the approval of our FDA applications and our introduction of new drugs. The CROs we contract with for execution of our clinical trials play a significant role in the conduct of the trials and the subsequent collection and analysis of data. Failure of the CROs to meet their obligations could adversely affect clinical development of our products. Moreover, these independent investigators and CROs may also have relationships with other commercial entities, some of which may compete with us. If independent investigators and CROs assist our competitors at our expense, it could harm our competitive position.
34
Table of Contents
We may be subject to claims that we or our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
As is commonplace in the biotechnology industry, we employ individuals who were previously employed at other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although no claims against us are currently pending, we may be subject to claims that these employees or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
If we are unable to retain and recruit qualified scientists or if any of our key senior executives discontinues his or her employment with us, it may delay our development efforts or impact our commercialization of INGREZZA or any product candidate approved by the FDA.
We are highly dependent on the principal members of our management and scientific staff. The loss of any of these people could impede the achievement of our objectives, including the successful commercialization of INGREZZA or any product candidate approved by the FDA. Furthermore, recruiting and retaining qualified scientific personnel to perform research and development work in the future, along with personnel with experience marketing and selling pharmaceutical products, is critical to our success. We may be unable to attract and retain personnel on acceptable terms given the competition among biotechnology, pharmaceutical and health care companies, universities and non-profit research institutions for experienced scientists and individuals with experience marketing and selling pharmaceutical products. In addition, we rely on a significant number of consultants to assist us in formulating our research and development strategy and our commercialization strategy. Our consultants may have commitments to, or advisory or consulting agreements with, other entities that may limit their availability to us.
If the market opportunities for our products and product candidates are smaller than we believe they are, our revenues may be adversely affected and our business may suffer.
Certain of the diseases that INGREZZA and our product candidates are being developed to address are in underserved and underdiagnosed populations. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who will seek treatment utilizing our products or product candidates, may not be accurate. If our estimates of the prevalence or number of patients potentially on therapy prove to be inaccurate, the market opportunities for INGREZZA and our product candidates may be smaller than we believe they are, our prospects for generating expected revenue may be adversely affected and our business may suffer.
We could face liability if a regulatory authority determines that we are promoting INGREZZA, or any of our product candidates that receives regulatory approval, for “off-label” uses.
A company may not promote “off-label” uses for its drug products. An off-label use is the use of a product for an indication that is not described in the product’s FDA-approved label in the United States or for uses in other jurisdictions that differ from those approved by the applicable regulatory agencies. Physicians, on the other hand, may prescribe products for off-label uses. Although the FDA and other regulatory agencies do not regulate a physician’s choice of drug treatment made in the physician’s independent medical judgment, they do restrict promotional communications from companies or their sales force with respect to off-label uses of products for which marketing clearance has not been issued. A company that is found to have promoted off-label use of its product may be subject to significant liability, including civil and criminal sanctions. We intend to comply with the requirements and restrictions of the FDA and other regulatory agencies with respect to our promotion of our products, including INGREZZA, but we cannot be sure that the FDA or other regulatory agencies will agree that we have not violated their restrictions. As a result, we may be subject to criminal and civil liability. In addition, our management’s attention could be diverted to handle any such alleged violations. A significant number of
35
Table of Contents
companies have been the target of inquiries and investigations by various United States federal and state regulatory, investigative, prosecutorial and administrative entities in connection with the promotion of products for unapproved uses and other sales practices, including the Department of Justice and various United States Attorneys’ Offices, the Office of Inspector General of the Department of Health and Human Services, the FDA, the Federal Trade Commission and various state Attorneys General offices. These investigations have alleged violations of various United States federal and state laws and regulations, including claims asserting antitrust violations, violations of the federal False Claims Act, the Prescription Drug Marketing Act, anti-kickback laws, and other alleged violations in connection with the promotion of products for unapproved uses, pricing and Medicare and/or Medicaid reimbursement. If the FDA or any other governmental agency initiates an enforcement action against us or if we are the subject of a qui tam suit and it is determined that we violated prohibitions relating to the promotion of products for unapproved uses, we could be subject to substantial civil or criminal fines or damage awards and other sanctions such as consent decrees and corporate integrity agreements pursuant to which our activities would be subject to ongoing scrutiny and monitoring to ensure compliance with applicable laws and regulations. Any such fines, awards or other sanctions would have an adverse effect on our revenue, business, financial prospects, and reputation.
Because our operating results may vary significantly in future periods, our stock price may decline.
Our quarterly revenues, expenses and operating results have fluctuated in the past and are likely to fluctuate significantly in the future. Our financial results are unpredictable and may fluctuate, for among other reasons, due to commercial sales of INGREZZA, the impact of Medicare Part D coverage; our achievement of product development objectives and milestones, clinical trial enrollment and expenses, research and development expenses and the timing and nature of contract manufacturing and contract research payments. In addition, we recently received regulatory approval from the FDA for INGREZZA in TD and our revenues will be dependent on our ability to sell INGREZZA and to secure adequate third-party reimbursement. A high portion of our costs are predetermined on an annual basis, due in part to our significant research and development costs. Thus, small declines in revenue could disproportionately affect financial results in a quarter. Because of these factors, our financial results in one or more future quarters may fail to meet the expectations of securities analysts or investors, which could cause our stock price to decline.
The recently passed comprehensive tax reform bill could adversely affect our business and financial condition.
On December 22, 2017, President Trump signed into law new legislation that significantly revises the Internal Revenue Code of 1986, as amended. The newly enacted federal income tax law, among other things, contains significant changes to corporate taxation, including reduction of the corporate tax rate from a top marginal rate of 35% to a flat rate of 21%, limitation of the tax deduction for interest expense to 30% of adjusted earnings (except for certain small businesses), limitation of the deduction for net operating losses to 80% of current year taxable income and elimination of net operating loss carrybacks, one time taxation of offshore earnings at reduced rates regardless of whether they are repatriated, elimination of U.S. tax on foreign earnings (subject to certain important exceptions), immediate deductions for certain new investments instead of deductions for depreciation expense over time, and modifying or repealing many business deductions and credits. Notwithstanding the reduction in the corporate income tax rate, the overall impact of the new federal tax law is uncertain and our business and financial condition could be adversely affected. In addition, it is uncertain if and to what extent various states will conform to the newly enacted federal tax law. The impact of this tax reform on holders of our common stock is also uncertain and could be adverse. We urge our stockholders to consult with their legal and tax advisors with respect to this legislation and the potential tax consequences of investing in or holding our common stock.
Our effective tax rate may fluctuate, and we may incur obligations in tax jurisdictions in excess of accrued amounts.
Our effective tax rate is derived from a combination of applicable tax rates in the various places that we operate. In preparing our financial statements, we estimate the amount of tax that will become payable in each of
36
Table of Contents
such places. Nevertheless, our effective tax rate may be different than experienced in the past due to numerous factors, including passage of the newly enacted federal income tax law, changes in the mix of our profitability from state to state, the results of examinations and audits of our tax filings, our inability to secure or sustain acceptable agreements with tax authorities, changes in accounting for income taxes and changes in tax laws. Any of these factors could cause us to experience an effective tax rate significantly different from previous periods or our current expectations and may result in tax obligations in excess of amounts accrued in our financial statements.
Our ability to use net operating losses to offset future taxable income may be subject to limitations.
As of December 31, 2017, we had federal and state income tax net operating loss carry forwards of approximately $978.7 million and $535.3 million, respectively. These net operating loss carry forwards could expire unused and be unavailable to offset future income tax liabilities. Under the newly enacted federal income tax law, federal net operating losses incurred in 2018 and in future years may be carried forward indefinitely, but the deductibility of such federal net operating losses is limited. It is uncertain if and to what extent various states will conform to the newly enacted federal tax law. In addition, under Section 382 of the Internal Revenue Code of 1986, as amended, and corresponding provisions of state law, if a corporation undergoes an “ownership change,” which is generally defined as a greater than 50% change, by value, in its equity ownership over a three-year period, the corporation’s ability to use its pre-change net operating loss carry forwards and other pre-change tax attributes to offset its post-change income or taxes may be limited. We have determined that no ownership changes have occurred through December 31, 2016 and we are currently evaluating whether any ownership changes occurred through December 31, 2017. We may experience ownership changes in the future as a result of subsequent shifts in our stock ownership, some of which may be outside of our control. If an ownership change occurs and our ability to use our net operating loss carry forwards is materially limited, it would harm our future operating results by effectively increasing our future tax obligations.
The price of our common stock is volatile.
The market prices for securities of biotechnology and pharmaceutical companies historically have been highly volatile, and the market for these securities has from time to time experienced significant price and volume fluctuations that are unrelated to the operating performance of particular companies. Furthermore, the price of our common stock may be dependent upon the valuations and recommendations of the analysts who cover our business. If our results do not meet these analysts’ forecasts, the expectations of our investors or the financial guidance we provide to investors in any period, which is based on assumptions that may be incorrect or that may change from quarter to quarter, the market price of our common stock could decline. During 2017, the price of our common stock has ranged from approximately $38.00 per share to approximately $78.00 per share. The market price of our common stock may fluctuate in response to many factors, including:
| • | sales of INGREZZA; |
| • | the status and cost of our post-marketing commitments for INGREZZA; |
| • | the results of our clinical trials; |
| • | developments concerning new and existing collaboration agreements; |
| • | announcements of technological innovations or new therapeutic products by us or others; |
| • | general economic and market conditions, including economic and market conditions affecting the biotechnology industry; |
| • | developments in patent or other proprietary rights; |
| • | developments related to the FDA; |
| • | future sales of our common stock by us or our stockholders; |
| • | comments by securities analysts; |
37
Table of Contents
| • | additions or departures of key personnel; |
| • | fluctuations in our operating results; |
| • | developments related to on-going litigation; |
| • | government regulation; |
| • | health care reimbursement; |
| • | failure of any of our product candidates, if approved, to achieve commercial success; and |
| • | public concern as to the safety of our drugs. |
If we cannot raise additional funding, we may be unable to complete development of our product candidates or establish commercial and manufacturing capabilities in the future.
We may require additional funding to commercialize INGREZZA for TD, to continue our research and product development programs, to conduct preclinical studies and clinical trials, for operating expenses and to pursue regulatory approvals for additional product candidates and indications, for the costs involved in filing and prosecuting patent applications and enforcing or defending patent claims, if any, product in-licensing and any possible acquisitions, establish manufacturing capabilities in the future and we may require additional funding to expand commercial and marketing efforts for other product candidates or indications. We believe that our existing capital resources, together with investment income, and future payments due under our strategic alliances, will be sufficient to satisfy our current and projected funding requirements for at least the next 12 months. However, these resources might be insufficient to conduct research and development programs, fully commercialize products and operate the company to the full extent currently planned. If we cannot obtain adequate funds, we may be required to curtail significantly our commercial plans or one or more of our research and development programs or obtain funds through additional arrangements with corporate collaborators or others that may require us to relinquish rights to some of our technologies or product candidates.
Our future capital requirements will depend on many factors, including:
| • | the commercial success of INGREZZA; |
| • | debt service obligations on the 2024 Notes; |
| • | continued scientific progress in our research and development programs; |
| • | the magnitude and complexity of our research and development programs; |
| • | progress with preclinical testing and clinical trials; |
| • | the time and costs involved in obtaining regulatory approvals; |
| • | the costs involved in filing and pursuing patent applications, enforcing patent claims, or engaging in interference proceedings or other patent litigation; |
| • | competing technological and market developments; |
| • | the establishment of additional strategic alliances; |
| • | developments related to on-going litigation; |
| • | the cost of commercialization activities and arrangements, including manufacturing of our product candidates; and |
| • | the cost of product in-licensing and any possible acquisitions. |
We intend to seek additional funding through strategic alliances, and may seek additional funding through public or private sales of our securities, including equity securities. For example, for so long as we continue to satisfy the requirements to be deemed a well-known seasoned issuer, we can utilize a shelf registration statement
38
Table of Contents
currently on file with the Securities and Exchange Commission (SEC), to allow us to issue an unlimited number of securities from time to time. In addition, during the second quarter of 2017, we issued the 2024 Notes and we have previously financed capital purchases and may continue to pursue opportunities to obtain additional debt financing in the future. Additional equity or debt financing might not be available on reasonable terms, if at all. Any additional equity financings will be dilutive to our stockholders and any additional debt financings may involve operating covenants that restrict our business.
Compliance with changing regulation of corporate governance and public disclosure may result in additional expenses.
Changing laws, regulations and standards relating to corporate governance and public disclosure, including the Sarbanes-Oxley Act of 2002, the Dodd-Frank Wall Street Reform and Consumer Protection Act, new SEC regulations and NASDAQ rules, are creating uncertainty for companies such as ours. These laws, regulations and standards are subject to varying interpretations in some cases due to their lack of specificity, and as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies, which could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. We are committed to maintaining high standards of corporate governance and public disclosure. As a result, our efforts to comply with evolving laws, regulations and standards have resulted in, and are likely to continue to result in, increased sales, general and administrative expenses and management time related to compliance activities. If we fail to comply with these laws, regulations and standards, our reputation may be harmed and we might be subject to sanctions or investigation by regulatory authorities, such as the SEC. Any such action could adversely affect our financial results and the market price of our common stock.
Risks Related to Our Industry
Health care reform measures and other recent legislative initiatives could adversely affect our business.
The business and financial condition of pharmaceutical and biotechnology companies are affected by the efforts of governmental and third-party payors to contain or reduce the costs of health care. In the United States, comprehensive health care reform legislation was enacted by the Federal government and we expect that there will continue to be a number of federal and state proposals to implement government control over the pricing of prescription pharmaceuticals. In addition, increasing emphasis on reducing the cost of health care in the United States will continue to put pressure on the rate of adoption and pricing of prescription pharmaceuticals. Moreover, in some foreign jurisdictions, pricing of prescription pharmaceuticals is already subject to government control. Additionally, other recent federal and state legislation imposes new obligations on manufacturers of pharmaceutical products, among others, related to product tracking and tracing. Among the requirements of this new legislation, manufacturers are required to provide certain information regarding the drug product to individuals and entities to which product ownership is transferred, label drug product with a product identifier, and keep certain records regarding distribution of the drug product. Further, under this new legislation, manufacturers will have drug product investigation, quarantine, disposition, notification and purchaser license verification responsibilities related to counterfeit, diverted, stolen, and intentionally adulterated products, as well as products that are the subject of fraudulent transactions or which are otherwise unfit for distribution such that they would be reasonably likely to result in serious health consequences or death.
Additionally, in March 2010, the ACA was signed into law, which was intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add transparency requirements for the healthcare and health insurance industries, impose taxes and fees on the health industry and impose additional health policy reforms. Among the provisions of the ACA of importance to our potential drug candidates are:
| • | an annual, nondeductible fee on any entity that manufactures or imports specified branded prescription drugs and biologic agents, apportioned among these entities according to their market share in certain government healthcare programs; |
39
Table of Contents
| • | an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program to 23.1% and 13.0% of the average manufacturer price for branded and generic drugs, respectively; |
| • | a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected; |
| • | extension of a manufacturer’s Medicaid rebate liability to covered drugs dispensed to individuals who are enrolled in Medicaid managed care organizations; |
| • | expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to certain individuals with income at or below 133% of the federal poverty level, thereby potentially increasing a manufacturer’s Medicaid rebate liability; |
| • | a new Medicare Part D coverage gap discount program, in which manufacturers must agree to offer 50% point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for a manufacturer’s outpatient drugs to be covered under Medicare Part D; |
| • | expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program; and |
| • | a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research. |
Some of the provisions of the ACA have yet to be fully implemented, and there have been legal and political challenges to certain aspects of the ACA. Since January 2017, President Trump has signed two executive orders and other directives designed to delay, circumvent, or loosen certain requirements mandated by the ACA. Concurrently, Congress has considered legislation that would repeal or repeal and replace all or part of the ACA. While Congress has not passed repeal legislation, the newly enacted federal income tax law includes a provision repealing, effective January 1, 2019, the tax-based shared responsibility payment imposed by the ACA on certain individuals who fail to maintain qualifying health coverage for all or part of a year that is commonly referred to as the “individual mandate”. Congress may consider other legislation to repeal or replace elements of the ACA.
Other legislative changes have been proposed and adopted since the ACA was enacted. These changes include aggregate reductions to Medicare payments to providers of up to 2% per fiscal year pursuant to the Budget Control Act of 2011, which began in 2013 and will remain in effect through 2025 unless additional Congressional action is taken. The American Taxpayer Relief Act of 2012, among other things, further reduced Medicare payments to several providers, including hospitals and cancer treatment centers, increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
Additional changes that may affect our business include the expansion of new programs such as Medicare payment for performance initiatives for physicians under MACRA, which will be fully implemented in 2019. At this time it is unclear how the introduction of the Medicare quality payment program will impact overall physician reimbursement. Also, there has been heightened governmental scrutiny recently over pharmaceutical pricing practices in light of the rising cost of prescription drugs and biologics. Such scrutiny has resulted in several recent Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for products. At the federal level, Congress and the Trump administration have each indicated that it will continue to seek new legislative and/or administrative measures to control drug costs. At the state level, legislatures have become increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing.
40
Table of Contents
We expect that the ACA, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and lower reimbursement, and in additional downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare or other government-funded programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our drugs.
We are currently unable to predict what additional legislation or regulation, if any, relating to the health care industry may be enacted in the future or what effect recently enacted federal legislation or any such additional legislation or regulation would have on our business. The pendency or approval of such proposals or reforms could result in a decrease in our stock price or limit our ability to raise capital or to enter into collaboration agreements for the further development and commercialization of our programs and products.
We face intense competition, and if we are unable to compete effectively, the demand for our products may be reduced.
The biotechnology and pharmaceutical industries are subject to rapid and intense technological change. We face, and will continue to face, competition in the development and marketing of our products and product candidates from academic institutions, government agencies, research institutions and biotechnology and pharmaceutical companies.
Competition may also arise from, among other things:
| • | other drug development technologies; |
| • | methods of preventing or reducing the incidence of disease, including vaccines; and |
| • | new small molecule or other classes of therapeutic agents. |
Developments by others may render our product candidates or technologies obsolete or noncompetitive.
We are commercializing and performing research on or developing products for the treatment of several disorders including endometriosis, TD, uterine fibroids, Tourette syndrome, essential tremor, classic congenital adrenal hyperplasia, pain, and other neurological and endocrine-related diseases and disorders, and there are a number of competitors to our products and product candidates. If one or more of our competitors’ products or programs are successful, the market for our products may be reduced or eliminated. For example, in August 2017, Teva received approval for AUSTEDO® to treat TD.
Compared to us, many of our competitors and potential competitors have substantially greater:
| • | capital resources; |
| • | research and development resources, including personnel and technology; |
| • | regulatory experience; |
| • | preclinical study and clinical testing experience; |
| • | manufacturing, marketing and distribution experience; and |
| • | production facilities. |
If we are unable to protect our intellectual property, our competitors could develop and market products based on our discoveries, which may reduce demand for our products.
Our success will depend on our ability to, among other things:
| • | obtain patent protection for our products; |
41
Table of Contents
| • | preserve our trade secrets; |
| • | prevent third parties from infringing upon our proprietary rights; and |
| • | operate without infringing upon the proprietary rights of others, both in the United States and internationally. |
Because of the substantial length of time and expense associated with bringing new products through the development and regulatory approval processes in order to reach the marketplace, the pharmaceutical industry places considerable importance on obtaining patent and trade secret protection for new technologies, products and processes. Accordingly, we intend to seek patent protection for our proprietary technology and compounds. However, we face the risk that we may not obtain any of these patents and that the breadth of claims we obtain, if any, may not provide adequate protection of our proprietary technology or compounds.
We also rely upon unpatented trade secrets and improvements, unpatented know-how and continuing technological innovation to develop and maintain our competitive position, which we seek to protect, in part, through confidentiality agreements with our commercial collaborators, employees and consultants. We also have invention or patent assignment agreements with our employees and some, but not all, of our commercial collaborators and consultants. However, if our employees, commercial collaborators or consultants breach these agreements, we may not have adequate remedies for any such breach, and our trade secrets may otherwise become known or independently discovered by our competitors.
In addition, although we own a number of patents, the issuance of a patent is not conclusive as to its validity or enforceability, and third parties may challenge the validity or enforceability of our patents. We cannot assure you how much protection, if any, will be given to our patents if we attempt to enforce them and they are challenged in court or in other proceedings. It is possible that a competitor may successfully challenge our patents or that challenges will result in limitations of their coverage. Moreover, competitors may infringe our patents or successfully avoid them through design innovation. To prevent infringement or unauthorized use, we may need to file infringement claims, which are expensive and time-consuming. In addition, in an infringement proceeding a court may decide that a patent of ours is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover its technology. Interference proceedings declared by the United States Patent and Trademark Office may be necessary to determine the priority of inventions with respect to our patent applications or those of our licensors. Litigation or interference proceedings may fail and, even if successful, may result in substantial costs and be a distraction to management. We cannot assure you that we will be able to prevent misappropriation of our proprietary rights, particularly in countries where the laws may not protect such rights as fully as in the United States.
If we fail to obtain or maintain orphan drug designation or other regulatory exclusivity for some of our product candidates, our competitive position would be harmed.
A product candidate that receives orphan drug designation can benefit from a streamlined regulatory process as well as potential commercial benefits following approval. Currently, this designation provides market exclusivity in the United States and the EU for seven years and ten years, respectively, if a product is the first such product approved for such orphan indication. This market exclusivity does not, however, pertain to indications other than those for which the drug was specifically designated in the approval, nor does it prevent other types of drugs from receiving orphan designations or approvals in these same indications. Further, even after an orphan drug is approved, the FDA can subsequently approve the same drug for the same condition if the FDA concludes that the new drug is clinically superior to the orphan product or a market shortage occurs.
In the EU, orphan exclusivity may be reduced to six years if the drug no longer satisfies the original designation criteria or can be lost altogether if the marketing authorization holder consents to a second orphan drug application or cannot supply enough drug, or when a second applicant demonstrates its drug is “clinically superior” to the original orphan drug. Valbenazine has received an orphan drug designation for the treatment of
42
Table of Contents
pediatric patients with Tourette syndrome from the FDA. If we seek orphan drug designations for other indications or in other jurisdictions, we may fail to receive such orphan drug designations and, even if we succeed, such orphan drug designations may fail to result in or maintain orphan drug exclusivity upon approval, which would harm our competitive position.
The technologies we use in our research as well as the drug targets we select may infringe the patents or violate the proprietary rights of third parties.
We cannot assure you that third parties will not assert patent or other intellectual property infringement claims against us or our collaborators with respect to technologies used in potential products. If a patent infringement suit were brought against us or our collaborators, we or our collaborators could be forced to stop or delay developing, manufacturing or selling potential products that are claimed to infringe a third party’s intellectual property unless that party grants us or our collaborators rights to use its intellectual property. In such cases, we could be required to obtain licenses to patents or proprietary rights of others in order to continue to commercialize our products. However, we may not be able to obtain any licenses required under any patents or proprietary rights of third parties on acceptable terms, or at all. Even if our collaborators or we were able to obtain rights to the third party’s intellectual property, these rights may be non-exclusive, thereby giving our competitors access to the same intellectual property. Ultimately, we may be unable to commercialize some of our potential products or may have to cease some of our business operations as a result of patent infringement claims, which could severely harm our business.
Our employees, independent contractors, principal investigators, consultants, commercial partners and vendors may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees and independent contractors, such as principal investigators, consultants, commercial partners and vendors, or by employees of our commercial partners could include failures to comply with FDA regulations, to provide accurate information to the FDA, to comply with manufacturing standards we have established, to comply with federal and state healthcare fraud and abuse laws, to report financial information or data accurately, to maintain the confidentiality of our trade secrets or the trade secrets of our commercial partners, or to disclose unauthorized activities to us. In particular, sales, marketing and other business arrangements in the healthcare industry are subject to extensive laws intended to prevent fraud, kickbacks, self-dealing and other abusive practices. Employee and independent contractor misconduct could also involve the improper use of individually identifiable information, including, without limitation, information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation.
Any relationships with healthcare professionals, principal investigators, consultants, customers (actual and potential) and third-party payors in connection with our current and future business activities are and will continue to be subject, directly or indirectly, to federal and state healthcare laws. If we are unable to comply, or have not fully complied, with such laws, we could face penalties, contractual damages, reputational harm, diminished profits and future earnings and curtailment or restructuring of our operations.
Our business operations and activities may be directly, or indirectly, subject to various federal and state healthcare laws, including without limitation, fraud and abuse laws, false claims laws, data privacy and security laws, as well as transparency laws regarding payments or other items of value provided to healthcare providers. These laws may restrict or prohibit a wide range of business activities, including, but not limited to, research, manufacturing, distribution, pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. These laws may impact, among other things, our current activities with principal investigators and research subjects, as well as current and future sales, marketing, patient co-payment assistance and education programs.
43
Table of Contents
Such laws include:
| • | the federal Anti-Kickback Statute which prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made under a federal healthcare program such as Medicare and Medicaid; |
| • | the federal civil and criminal false claims, including the civil False Claims Act, and civil monetary penalties laws, which impose criminal and civil penalties against individuals or entities for, among other things, knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government; |
| • | the federal Health Insurance Portability and Accountability Act of 1996 (HIPAA) which imposes criminal and civil liability for, among other things, executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters; |
| • | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act and its implementing regulations, which also imposes obligations, including mandatory contractual terms, on certain types of individuals and entities, with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| • | the federal Physician Payments Sunshine Act, which requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with specific exceptions, to report annually to CMS information related to payments or other transfers of value made to physicians and teaching hospitals, and applicable manufacturers and applicable group purchasing organizations to report annually to CMS ownership and investment interests held by the physicians and their immediate family members; and |
| • | analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, which may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third party payors, including private insurers; state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; and state and foreign laws governing the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
Efforts to ensure that our business arrangements will comply with applicable healthcare laws may involve substantial costs. It is possible that governmental and enforcement authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other healthcare laws. If our operations or activities are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to, without limitation, civil, criminal and administrative penalties, damages, monetary fines, disgorgement, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, contractual damages, reputational harm, diminished profits and future earnings and curtailment or restructuring of our operations, any of which could adversely affect our ability to operate.
In addition, any sales of our product once commercialized outside the United States will also likely subject us to foreign equivalents of the healthcare laws mentioned above, among other foreign laws.
44
Table of Contents
We face potential product liability exposure far in excess of our limited insurance coverage.
The use of any of our potential products in clinical trials, and the sale of any approved products, including INGREZZA, may expose us to liability claims. These claims might be made directly by consumers, health care providers, pharmaceutical companies or others selling our products. We have obtained limited product liability insurance coverage for our clinical trials in the amount of $10 million per occurrence and $10 million in the aggregate. However, our insurance may not reimburse us or may not be sufficient to reimburse us for any expenses or losses we may suffer. Moreover, insurance coverage is becoming increasingly expensive, and we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability. Upon FDA approval of INGREZZA we expanded our insurance coverage to include product liability insurance related to the sale of INGREZZA in the amount of $20 million per occurrence and $20 million in the aggregate. However, we may be unable to obtain commercially reasonable product liability insurance for any products approved in the future for marketing. On occasion, juries have awarded large judgments in class action lawsuits based on drugs that had unanticipated side effects. A successful product liability claim or series of claims brought against us would decrease our cash reserves and could cause our stock price to fall.
Our activities involve hazardous materials, and we may be liable for any resulting contamination or injuries.
Our research activities involve the controlled use of hazardous materials. We cannot eliminate the risk of accidental contamination or injury from these materials. If an accident occurs, a court may hold us liable for any resulting damages, which may harm our results of operations and cause us to use a substantial portion of our cash reserves, which would force us to seek additional financing.
Cyber security breaches and other disruptions could compromise our information and expose us to liability, which would cause our business and reputation to suffer.
In the ordinary course of our business, we collect and store confidential and sensitive electronic information on our networks and in our data centers. This information includes, among other things, our intellectual property and proprietary information, the confidential information of our collaborators and licensees, and the personally identifiable information of our employees. It is important to our operations and business strategy that this electronic information remains secure and is perceived to be secure. Despite security measures, however, our information technology and network infrastructure may be vulnerable to attacks by hackers or breached due to employee error, malfeasance, or other disruptions. Any such cyber-attack or breach could compromise our networks and data centers and the information stored there could be accessed, publicly disclosed, lost, or stolen. Any such access, disclosure, or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, delays and impediments to our discovery, development, and commercialization efforts, and damage to our reputation.
45
Table of Contents
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
We lease our corporate headquarters which consists of approximately 140,000 square feet of laboratory and office space located at 12780 El Camino Real in San Diego, California. The lease expires in December 2029 with options to extend the term of the lease for up to two consecutive ten year periods.
We believe that our property and equipment are generally well maintained and in good operating condition.
| ITEM 3. | LEGAL PROCEEDINGS |
The information set forth under Note 7 “Commitments and Contingencies” to our consolidated financial statements included in Part II, Item 8 of this Annual Report on From 10-K is incorporated herein by reference.
| ITEM 4. | MINE SAFETY DISCLOSURES |
None.
46
Table of Contents
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Our common stock is traded on the NASDAQ Global Select Market under the symbol “NBIX.” The following table sets forth for the periods indicated the high and low sale price for our common stock. These prices do not include retail markups, markdowns or commissions.
| High | Low | |||||||
| Year Ended December 31, 2017 |
||||||||
| 1st Quarter |
$ | 47.43 | $ | 38.38 | ||||
| 2nd Quarter |
55.38 | 39.21 | ||||||
| 3rd Quarter |
61.51 | 44.75 | ||||||
| 4th Quarter |
78.05 | 57.71 | ||||||
| Year Ended December 31, 2016 |
||||||||
| 1st Quarter |
$ | 55.94 | $ | 31.25 | ||||
| 2nd Quarter |
53.00 | 39.01 | ||||||
| 3rd Quarter |
55.15 | 44.69 | ||||||
| 4th Quarter |
54.91 | 37.35 | ||||||
As of February 6, 2018, there were approximately 51 stockholders of record of our common stock. We have not paid any cash dividends on our common stock since inception and do not anticipate paying cash dividends in the foreseeable future.
Information about our equity compensation plans is incorporated herein by reference to Item 12 of Part III of this Annual Report on Form 10-K.
Recent Sales of Unregistered Securities
There were no unregistered sales of equity securities during fiscal 2017.
47
Table of Contents
Stock Performance Graph and Cumulative Total Return*
The graph below shows the cumulative total stockholder return assuming the investment of $100 on December 31, 2012 (and the reinvestment of dividends thereafter) in each of (i) Neurocrine Biosciences, Inc.’s common stock, (ii) the NASDAQ Composite Index and (iii) the NASDAQ Biotechnology Index. The comparisons in the graph below are based upon historical data and are not indicative of, or intended to forecast, future performance of our common stock or Indexes.
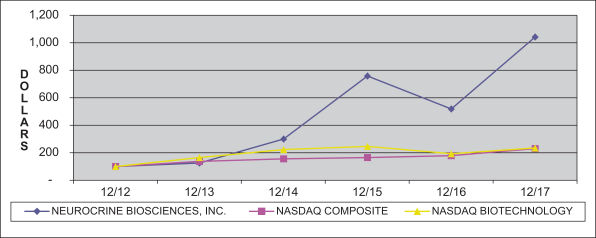
| * | The material in this section is not “soliciting material”, is not deemed “filed” with the SEC and is not to be incorporated by reference into any of our SEC filings whether made before or after the date hereof and irrespective of any general incorporation language in any such SEC filing except to the extent we specifically incorporate this section by reference. |
48
Table of Contents
| ITEM 6. | SELECTED FINANCIAL DATA |
The following selected financial data have been derived from our audited financial statements. The information set forth below is not necessarily indicative of our results of future operations and should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the financial statements and notes thereto appearing elsewhere in this Annual Report on Form 10-K.
| 2017 | 2016 | 2015 | 2014 | 2013 | ||||||||||||||||
| (In thousands, except for net (loss) income per share data) | ||||||||||||||||||||
| STATEMENT OF COMPREHENSIVE LOSS DATA |
||||||||||||||||||||
| Revenues: |
||||||||||||||||||||
| Product sales, net |
$ | 116,626 | $ | — | $ | — | $ | — | $ | — | ||||||||||
| Milestones and license fees |
45,000 | 15,000 | 19,769 | — | 2,919 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total revenues |
161,626 | 15,000 | 19,769 | — | 2,919 | |||||||||||||||
| Operating expenses: |
||||||||||||||||||||
| Cost of product sales |
1,254 | — | — | — | — | |||||||||||||||
| Research and development |
121,827 | 94,291 | 81,491 | 46,425 | 39,248 | |||||||||||||||
| Sales, general and administrative |
169,906 | 68,081 | 32,480 | 17,986 | 13,349 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total operating expenses |
292,987 | 162,372 | 113,971 | 64,411 | 52,597 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Loss from operations |
(131,361 | ) | (147,372 | ) | (94,202 | ) | (64,411 | ) | (49,678 | ) | ||||||||||
| Other income (expense): |
||||||||||||||||||||
| Deferred gain on real estate |
2,124 | 3,423 | 3,325 | 3,226 | 3,133 | |||||||||||||||
| Interest expense |
(19,523 | ) | — | — | — | — | ||||||||||||||
| Other income, net |
6,218 | 2,859 | 1,948 | 643 | 455 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total other income (expense), net |
(11,181 | ) | 6,282 | 5,273 | 3,869 | 3,588 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net loss |
$ | (142,542 | ) | $ | (141,090 | ) | $ | (88,929 | ) | $ | (60,542 | ) | $ | (46,090 | ) | |||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Net loss per common share: |
||||||||||||||||||||
| Basic |
$ | (1.62 | ) | $ | (1.63 | ) | $ | (1.05 | ) | $ | (0.81 | ) | $ | (0.69 | ) | |||||
| Diluted |
$ | (1.62 | ) | $ | (1.63 | ) | $ | (1.05 | ) | $ | (0.81 | ) | $ | (0.69 | ) | |||||
| Shares used in calculation of net loss per common share: |
||||||||||||||||||||
| Basic |
88,089 | 86,713 | 84,496 | 74,577 | 66,989 | |||||||||||||||
| Diluted |
88,089 | 86,713 | 84,496 | 74,577 | 66,989 | |||||||||||||||
| BALANCE SHEET DATA |
||||||||||||||||||||
| Cash, cash equivalents and investments |
$ | 763,290 | $ | 350,840 | $ | 461,679 | $ | 231,301 | $ | 145,739 | ||||||||||
| Working capital |
500,493 | 280,028 | 358,359 | 182,539 | 136,763 | |||||||||||||||
| Total assets |
817,591 | 365,086 | 474,785 | 243,033 | 154,676 | |||||||||||||||
| Long-term debt |
369,618 | — | — | — | — | |||||||||||||||
| Accumulated deficit |
(1,198,866 | ) | (1,056,324 | ) | (915,234 | ) | (826,305 | ) | (765,763 | ) | ||||||||||
| Total stockholders’ equity |
372,138 | 314,877 | 424,454 | 208,699 | 120,410 | |||||||||||||||
49
Table of Contents
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
The following Management’s Discussion and Analysis of Financial Condition and Results of Operations section contains forward-looking statements pertaining to, among other things, the commercialization of our product and product candidates, the expected continuation of our collaborative agreements, the receipt of research and development payments thereunder, the future achievement of various milestones in product development and the receipt of payments related thereto, the potential receipt of royalty payments, preclinical testing and clinical trials of potential products, the period of time that our existing capital resources will meet our funding requirements, and our financial results of operations. Our actual results could differ materially from those anticipated in these forward-looking statements as a result of various risks and uncertainties, including those set forth in this Annual Report on Form 10-K under the heading “Item 1A. Risk Factors.” See “Forward-Looking Statements” in Part I of this Annual Report on Form 10-K.
Overview
We discover and develop innovative and life-changing pharmaceuticals, in diseases with high unmet medical needs, through our novel research and development (R&D) platform, focused on neurological, psychological and endocrine based diseases and disorders. Utilizing a portfolio approach to drug discovery, we have multiple small molecule drug candidates at various stages of pharmaceutical development. We develop proprietary pharmaceuticals for our pipeline, as well as collaborate with other pharmaceutical companies on our discoveries.
On April 11, 2017, the U.S. Food and Drug Administration (FDA) approved INGREZZA® (valbenazine) capsules for the treatment of adults with tardive dyskinesia (TD). We market INGREZZA for TD in the United States through our specialty sales force focused primarily on physicians who treat TD patients, including psychiatrists and neurologists. The commercial launch of INGREZZA, with our field-based sales team of approximately 160 personnel, occurred on May 1, 2017.
Our three lead late-stage clinical programs are INGREZZATM (valbenazine) for Tourette syndrome, elagolix, a gonadotropin-releasing hormone (GnRH) antagonist for women’s health that is partnered with AbbVie Inc. (AbbVie), and opicapone, a highly-selective catechol-O-methyltransferase inhibitor (COMT inhibitor) that is an adjunct therapy to preparations of levodopa/DOPA decarboxylase inhibitors for adult patients with Parkinson’s disease and was in-licensed from BIAL – Portela & CA, S.A. (BIAL).
We have funded our operations primarily through private and public offerings of our common stock, debt securities and payments received under collaboration agreements. While we independently develop many of our product candidates, we have entered into collaborations for several of our programs, and intend to rely on our product revenues and existing and future collaborators to meet funding requirements. We expect to generate future operating losses as product candidates are advanced through the various stages of clinical development and as we proceed with the commercial launch of INGREZZA. As of December 31, 2017, we had an accumulated deficit of approximately $1.2 billion and expect to incur an operating loss in 2018.
We currently have three major collaborations. Two of these collaborations involve out-licensing of our proprietary technology to pharmaceutical partners. In June 2010, we announced an exclusive worldwide collaboration with AbbVie to develop and commercialize elagolix and all next-generation GnRH antagonists (collectively, GnRH Compounds) and in March 2015, we entered into a collaboration and license agreement with Mitsubishi Tanabe Pharma Corporation (Mitsubishi Tanabe) for the development and commercialization of INGREZZA for movement disorders in Japan and other select Asian markets. The third collaboration agreement is one in which we in-licensed technology from BIAL for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. These three agreements are discussed below under the heading “Critical Accounting Policies.”
50
Table of Contents
Critical Accounting Policies
Our discussion and analysis of our financial condition and results of operations is based upon financial statements that we have prepared in accordance with accounting principles generally accepted in the United States. The preparation of these financial statements requires management to make estimates and judgments that affect the reported amounts of assets, liabilities and expenses, and related disclosures. On an on-going basis, we evaluate these estimates, including those related to revenue recognition, clinical trial accruals (research and development expense), convertible debt, accounts receivable, inventory, cost of product sales and share-based compensation. Estimates are based on historical experience, information received from third parties and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions. Historically, revisions to our estimates have not resulted in a material change to the financial statements. The items in our financial statements requiring significant estimates and judgments are as follows:
Product Revenue Recognition
Our net product sales consist of U.S. sales of INGREZZA and are recognized when (i) persuasive evidence of an arrangement exists, (ii) delivery has occurred and title to the product and associated risk of loss has passed to the customer, (iii) the price is fixed or determinable, and (iv) collectability is reasonably assured. INGREZZA was approved by the FDA on April 11, 2017 and we commenced shipments of INGREZZA to select pharmacies (SPs) and a select distributor (SD) in late April 2017. SPs dispense product to a patient based on the fulfillment of a prescription and SDs sell product to government facilities, long-term care pharmacies or in-patient hospital pharmacies. Our agreements with SPs and the SD provide for transfer of title to the product at the time the product is delivered to the SP or SD. In addition, except for limited circumstances, the SPs and SD have no right of product return to us. This limited right of return results in the SP or SD using a just-in-time inventory model and which results in frequent product deliveries from our central distribution center. Our agreements with our SPs and SD provide data related to prescription fulfillment including the insurance coverage, copay amounts, deductibles and other data necessary to determine net product sales.
We record revenue when the product is delivered to our SPs or SD, which is an approach frequently referred to as the “sell-in” revenue recognition model. We recognize revenue from product sales net of allowances for distribution fees, rebates, chargebacks, and co-payment assistance. Reserves are established for these allowances upon receipt of INGREZZA by the SPs or SD and are classified as: (i) an allowance against accounts receivable if the amount is payable to the SP or SD, or (ii) an accrued liability if the amount is payable to a party other than the SP or SD. Allowances against accounts receivable relate to chargebacks and distributor fees and accruals relate primarily to government rebates.
Distribution Fees: Distribution fees include fees paid to the SPs and SD for data, prompt payment discounts and other off-invoice discounts. These fees are recorded based on contractual terms.
Government Rebates: Rebates include mandated discounts under the Medicaid Drug Rebate Program and the Medicare Part D prescription drug benefit. Rebates are amounts owed after the final dispensing of the product to a benefit plan participant and result from contractual agreements with, or statutory requirements pertaining to, Medicaid and Medicare benefit providers. The allowance for rebates is based on statutory discount rates, expected utilization and expected trends in net reimbursement. Our expected utilization of rebates is based on prescription level detail provided by our SPs that includes insurer amounts for each prescription filled during the period as well as the limited amount of product that is in the SPs warehouse at period end.
Chargebacks: Chargebacks are discounts that relate to contracts with government and other entities purchasing from the SD at a discounted price. The SD charges back to us the difference between the price initially paid by the SD and the discounted price paid to the SD by these entities. Chargebacks are recorded based on known and expected SD sales to these entities.
51
Table of Contents
Co-Payment Assistance: We offer co-payment assistance to commercially insured patients meeting certain eligibility requirements. Co-payment assistance is recorded based on actual and expected program participation. We base our estimate of expected program participation on prescription level detail provided by our SPs that includes copay amounts for each prescription filled during the period as well as the limited amount of product that is in the SPs warehouse at period end. While we obtain this detailed information, we must make estimates of the actual level of rebates, which we base on the detailed information as well as our experience.
Due to estimates and assumptions inherent in determining the government rebate and copay assistance amounts, the actual rebates and copay assistance paid may be different than our estimates.
Collaboration Agreement Revenue Recognition
Since 2011, we have followed the Accounting Standards Codification (ASC) for Revenue Recognition – Multiple-Element Arrangements, if applicable, to determine the recognition of revenue under license and collaboration agreements. The ASC provides guidance relating to the separation of deliverables included in an arrangement into different units of accounting and the allocation of consideration to the units of accounting. The evaluation of multiple-element arrangements requires management to make judgments about (i) the identification of deliverables, (ii) whether such deliverables are separable from the other aspects of the contractual relationship, (iii) the estimated selling price of each deliverable, and (iv) the expected period of performance for each deliverable.
To determine the units of accounting under a multiple-element arrangement, we evaluate certain separation criteria, including whether the deliverables have stand-alone value, based on the relevant facts and circumstances for each arrangement. The selling prices of deliverables under an arrangement may be derived using vendor specific objective evidence (VSOE), third-party evidence, or a best estimate of selling price (BESP), if VSOE or third-party evidence is not available. For most pharmaceutical licensing and collaboration agreements, BESP is utilized. Establishing BESP involves our judgment and considers multiple factors, including market conditions and company-specific factors, including those factors contemplated in negotiating the agreements, as well as internally developed models that include assumptions related to market opportunity, discounted cash flows, estimated development costs, probability of success and the time needed to commercialize a product candidate pursuant to the agreement. In validating the BESP, we consider whether changes in key assumptions used to determine the BESP will have a significant effect on the allocation of the arrangement consideration between the multiple deliverables. The allocated consideration for each unit of accounting is recognized over the related obligation period in accordance with the applicable revenue recognition criteria.
If there are deliverables in an arrangement that are not separable from other aspects of the contractual relationship, they are treated as a combined unit of accounting, with the allocated revenue for the combined unit recognized in a manner consistent with the revenue recognition applicable to the final deliverable in the combined unit. Payments received prior to satisfying the relevant revenue recognition criteria are recorded as deferred revenue in the accompanying balance sheets and recognized as revenue when the related revenue recognition criteria are met.
We typically receive up-front payments when licensing our intellectual property, which often occurs in conjunction with an R&D agreement. We recognize revenue attributed to the license upon delivery, provided that the license has stand-alone value.
Milestones are recognized when earned, as evidenced by written acknowledgment from the collaborator or other persuasive evidence that the milestone has been achieved, provided that the milestone event is substantive. A milestone event is generally considered to be substantive if its achievability was not reasonably assured at the inception of the agreement. We assess whether a milestone is substantive at the inception of each agreement.
52
Table of Contents
Cost of Product Sales
Cost of product sales primarily consists of third-party manufacturing costs, storage, freight and other costs associated with sales of INGREZZA. Cost of product sales may also include period costs related to certain inventory manufacturing services, inventory adjustment charges, and manufacturing variances.
Accounts Receivable
Accounts receivable are recorded net of customer allowances for prompt payment discounts, chargebacks, and any allowance for doubtful accounts. We estimate the allowance for doubtful accounts based on existing contractual payment terms, actual payment patterns of its customers and individual customer circumstances. To date, we have determined that an allowance for doubtful accounts is not required.
Inventory
Inventory is stated at the lower of cost or estimated net realizable value. We currently use actual costing methodologies to determine the cost basis for our inventories. Inventory is valued on a first-in, first-out basis and consists primarily of third-party manufacturing costs. We capitalize inventory costs associated with our products upon regulatory approval when, based on our judgment, future commercialization is considered probable and the future economic benefit is expected to be realized; otherwise, such costs are expensed. Prior to FDA approval of INGREZZA, all costs related to its manufacturing were charged to research and development expense in the period incurred. At December 31, 2017, our physical inventory consisted primarily of active pharmaceutical product that had been produced prior to FDA approval of INGREZZA with no cost basis as the cost associated with producing this material was expensed rather than capitalized in accordance with accounting guidance. Additionally, bulk drug production, finished bottling and other activities that occurred post FDA approval are included in inventory at December 31, 2017. We provide reserves for potential excess, dated or obsolete inventory based on an analysis of forecasted demand compared to quantities on hand and any firm purchase orders, as well as product shelf life. To date, we have determined that such reserves are not required.
Research and Development Expense
R&D expense consists primarily of salaries, payroll taxes, employee benefits, and share-based compensation charges, for those individuals involved in ongoing research and development efforts; as well as scientific contractor fees, preclinical and clinical trial costs, research and development facilities costs, laboratory supply costs, and depreciation of scientific equipment. All such costs are charged to R&D expense as incurred. These expenses result from our independent R&D efforts as well as efforts associated with collaborations and in-licensing arrangements. In addition, we fund R&D and clinical trials at other companies and research institutions under agreements, which are generally cancelable. We review and accrue clinical trials expense based on work performed, which relies on estimates of total costs incurred based on patient enrollment, completion of studies and other events. We follow this method since reasonably dependable estimates of the costs applicable to various stages of a research agreement or clinical trial can be made. Accrued clinical costs are subject to revisions as trials progress. Revisions are charged to expense in the period in which the facts that give rise to the revision become known. Historically, revisions have not resulted in material changes to R&D expense; however a modification in the protocol of a clinical trial or cancellation of a trial could result in a charge to our results of operations.
Share-Based Compensation
We grant stock options to purchase our common stock to our employees and directors under our 2011 Equity Incentive Plan (the 2011 Plan) and grant stock options to certain employees pursuant to Employment Commencement Nonstatutory Stock Option Agreements (inducement grants). We also grant certain employees stock bonuses and restricted stock units (RSUs) under the 2011 Plan, and grant certain employees stock options
53
Table of Contents
and RSUs under the Neurocrine Biosciences, Inc. Inducement Plan (Inducement Plan). Additionally, we have outstanding options that were granted under previous equity plans from which we no longer make grants. Share-based compensation expense related to these equity instruments for the years ended December 31, 2017, 2016 and 2015 was $42.5 million, $28.5 million and $28.4 million, respectively.
Stock option awards and RSUs generally vest over a three to four year period and expense is ratably recognized over those same time periods. For RSUs with performance-based vesting requirements (PRSUs), no expense is recorded until the performance condition is probable of being achieved; upon which expense is then recognized ratably over the expected performance period. During 2017, 2016 and 2015, we recognized approximately $0.4 million, $1.8 million and $8.8 million, respectively, in expense related to certain PRSUs as it became probable that pre-defined performance conditions would be met primarily due to the Phase III results of the Kinect 3 clinical study which were unblinded during the third quarter of 2015.
For purposes of calculating share-based compensation, we estimate the fair value of share-based compensation awards using a Black-Scholes option-pricing model. The determination of the fair value of share-based compensation awards utilizing the Black-Scholes model is affected by our stock price and a number of assumptions, including but not limited to expected stock price volatility over the term of the awards and the expected term of stock options. Our stock options have characteristics significantly different from those of traded options, and changes in the assumptions can materially affect the fair value estimates. For example, an increase in the underlying stock price results in a significant increase in the Black-Scholes option-pricing.
If factors change and we employ different assumptions, share-based compensation expense may differ significantly from what we have recorded in the past. If there is a difference between the assumptions used in determining share-based compensation expense and the actual factors which become known over time, we may change the input factors used in determining share-based compensation expense for future grants. These changes, if any, may materially impact our results of operations in the period such changes are made. For actual forfeitures, we recognize the adjustment to compensation expense in the period the forfeitures occur.
Convertible Debt
We account for convertible debt instruments that may be settled in cash upon conversion by separating the liability and equity components of the instruments in a manner that reflects our nonconvertible debt borrowing rate. In May 2017, we issued $517.5 million aggregate principal amount of 2.25% Convertible Senior Notes due 2024 (the 2024 Notes). We determined the carrying amount of the liability component of the 2024 Notes by using assumptions that market participants would use in pricing a debt instrument, including market interest rates, credit standing, yield curves and volatilities. Determining the fair value of the debt component requires the use of accounting estimates and assumptions. These estimates and assumptions are judgmental in nature and could have a significant impact on the determination of the debt component, and the associated non-cash interest expense.
Debt acquisition costs related to the 2024 Notes were $14.7 million. In addition, we allocated $149.2 million to the equity component of the convertible debt instrument. We are amortizing the debt acquisition costs and the equity component over the life of the 2024 Notes as additional non-cash interest expense utilizing the effective interest method.
Collaboration Agreements
Mitsubishi Tanabe Pharma Corporation (Mitsubishi Tanabe). On March 31, 2015, we entered into a collaboration and license agreement with Mitsubishi Tanabe for the development and commercialization of INGREZZA (valbenazine) for movement disorders in Japan and other select Asian markets. Payments to us under this agreement include an up-front license fee of $30 million, up to $85 million in development and commercialization event-based payments, payments for the manufacture of pharmaceutical products, and royalties on product sales in select territories in Asia. Under the terms of the agreement, Mitsubishi Tanabe is
54
Table of Contents
responsible for all third-party development, marketing and commercialization costs in Japan and other select Asian markets with the exception of a single Huntington’s chorea clinical trial to be performed by us, at an estimated cost of approximately $12 million, should Mitsubishi Tanabe request the clinical trial. We will be entitled to a percentage of sales of INGREZZA in Japan and other select Asian markets for the longer of ten years or the life of the related patent rights.
Under our agreement with Mitsubishi Tanabe, the collaboration effort between the parties to advance INGREZZA towards commercialization in Japan and other select Asian markets is governed by a joint steering committee and joint development committee with representatives from both us and Mitsubishi Tanabe. There are no performance, cancellation, termination or refund provisions in the agreement that would have a material financial consequence to us. We do not directly control when event-based payments will be achieved or when royalty payments will begin. Mitsubishi Tanabe may terminate the agreement at its discretion upon 180 days’ written notice to us. In such event, all INGREZZA product rights for Japan and other select Asian markets would revert to us.
We have identified the following deliverables associated with the Mitsubishi Tanabe agreement: INGREZZA technology license and existing know-how, development activities to be performed as part of the collaboration, and the manufacture of pharmaceutical products. The respective standalone value from each of these deliverables has been determined by applying the BESP method and the revenue was allocated based on the relative selling price method with revenue recognition timing to be determined either by delivery or the provision of services.
As discussed above, the BESP method required the use of significant estimates. We used an income approach to estimate the selling price for the technology license and an expense approach for estimating development activities and the manufacture of pharmaceutical products. The development activities and the manufacture of pharmaceutical products are expected to be delivered throughout the duration of the agreement. The technology license and existing know-how was delivered on the effective date of the agreement.
We also evaluated the event-based payments under the Milestone Method and concluded only one immaterial event-based payment represents a substantive milestone. Event-based payments will be recognized when earned.
We are eligible to receive tiered royalty payments based on product sales in Japan and other select Asian markets. Royalties will be recognized as earned in accordance with the terms of the agreement, when product sales are reported by Mitsubishi Tanabe, the amount can be reasonably estimated, and collectability is reasonably assured.
For the year ended December 31, 2017, we recognized $15 million in development event-based payments resulting from Mitsubishi Tanabe’s initiation of Phase II/III development of INGREZZA in TD in Asia. No revenue was recognized under the Mitsubishi Tanabe agreement for the year ended December 31, 2016. For the year ended December 31, 2015, we recognized revenue under this agreement of $19.8 million associated with the delivery of a technology license and existing know-how. In accordance with our continuing performance obligations, $10.2 million of the $30 million up-front payment is being deferred and recognized in future periods. Under the terms of the agreement, there is no general obligation to return the up-front payment for any non-contingent deliverable.
AbbVie Inc. (AbbVie). In June 2010, we announced an exclusive worldwide collaboration with AbbVie to develop and commercialize elagolix and all next-generation GnRH antagonists (collectively, GnRH Compounds) for women’s and men’s health. AbbVie made an upfront payment of $75 million and agreed to make additional development and regulatory event-based payments of up to $480 million and up to an additional $50 million in commercial event-based payments. We assessed event-based payments under the revised authoritative guidance for research and development milestones and determined that event-based payments prior to commencement of a
55
Table of Contents
Phase III clinical study, as defined in the agreement, meet the definition of a milestone in accordance with authoritative guidance as (i) they are events that can only be achieved in part on our past performance, (ii) there is substantive uncertainty at the date the arrangement was entered into that the event will be achieved and (iii) they result in additional payments being due to us. Development and regulatory event-based payments subsequent to the commencement of a Phase III clinical study, however, currently do not meet these criteria as their achievement is based on the performance of AbbVie. As of December 31, 2017, $455 million remains outstanding in future event-based payments under the agreement. However, none of the remaining event-based payments meet the definition of a milestone in accordance with authoritative accounting guidance and will be recognized when earned as we have no ongoing obligations.
Under the terms of the agreement, AbbVie is responsible for all third-party development, marketing and commercialization costs. We will be entitled to a percentage of worldwide sales of GnRH Compounds for the longer of ten years or the life of the related patent rights. AbbVie may terminate the collaboration at its discretion upon 180 days’ written notice to us. In such event all GnRH Compound product rights would revert to us.
During 2017, event-based revenue of $30.0 million was recognized based on AbbVie’s New Drug Application (NDA) submission for elagolix in endometriosis being accepted as filed by the FDA. During 2016, event-based revenue of $15.0 million was recognized related to AbbVie’s initiation of Phase III development of elagolix in uterine fibroids. No revenue was recognized during 2015 under this collaboration agreement.
BIAL – Portela & CA, S.A. (BIAL). In February 2017, we entered into an exclusive license agreement with BIAL for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. Under the terms of the agreement, we are responsible for the management and cost of all opicapone development and commercialization activities in the United States and Canada.
Under the terms of the agreement, we paid BIAL an upfront license fee of $30 million, which was expensed in the first quarter of 2017 as in process research and development. We may also be required to pay up to an additional $115 million in milestone payments associated with the regulatory approval and net sales of products containing opicapone. Prior to FDA approval of opicapone, we may be required to pay up to $20 million in milestones based on certain regulatory and clinical results and FDA acceptance of our NDA filing for opicapone. Upon commercialization of opicapone, we have agreed to determine certain annual sales forecasts. In the event that we fail to meet the minimum sales requirements for a particular year, we will be required to pay BIAL an amount corresponding to the difference between the actual net sales and the minimum sales requirements for such year, and if we fail to meet the minimum sales requirements for any two years, BIAL may terminate the agreement.
The agreement, unless terminated earlier, will continue on a licensed product-by-licensed product and country-by-country basis until a generic product in respect of such licensed product under the agreement is sold in a country and sales of such generic product are greater than a specified percentage of total sales of such licensed product in such country. Upon our written request prior to the estimated expiration of the term in respect of a licensed product, the parties shall negotiate a good faith continuation of BIAL’s supply of such licensed product after the term. After the term, and if BIAL is not supplying a certain licensed product, we shall pay BIAL a trademark royalty based on the net sales of such licensed product. Either party may terminate the agreement earlier if the other party materially breaches the agreement and does not cure the breach within a specified notice period, or upon the other party’s insolvency. BIAL may terminate the agreement if we fail to use commercially reasonable efforts or fail to file an NDA for a licensed product by a specified date or under certain circumstances involving our change of control. In certain circumstances where BIAL elects to terminate the agreement in connection with our change of control, BIAL shall pay us a termination fee. We may terminate the agreement at any time for any reason upon six months written notice to BIAL if prior to the first NDA approval in the United States, and upon nine months written notice to BIAL if such notice is given after the first NDA approval in the United States. If our termination request occurs prior to the first NDA approval in the United States, we will have to pay BIAL a termination fee except under certain conditions specified in the agreement.
56
Table of Contents
Results of Operations for Years Ended December 31, 2017, 2016 and 2015
Revenue
Product Sales, net
In April 2017, the FDA approved INGREZZA for the treatment of TD. INGREZZA became available for prescription in late April 2017. Net product sales were $116.6 million for the year ended December 31, 2017 with no similar net product sales during 2016 or 2015.
Milestones and License Fees
The following table summarizes our collaboration revenue during the periods presented:
| Year
Ended December 31, |
||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| (In millions) | ||||||||||||
| Revenues under collaboration agreements: |
||||||||||||
| Mitsubishi Tanabe Pharma, Inc. |
$ | 15.0 | $ | — | $ | 19.8 | ||||||
| AbbVie, Inc. |
30.0 | 15.0 | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Total revenues |
$ | 45.0 | $ | 15.0 | $ | 19.8 | ||||||
|
|
|
|
|
|
|
|||||||
During 2017, we recognized $15.0 million in development event-based payments resulting from Mitsubishi Tanabe’s initiation of Phase II/III development of INGREZZA in TD in Asia. During October 2017, AbbVie’s NDA submission for elagolix in endometriosis was accepted as filed by the FDA. This NDA filing acceptance generated a $30.0 million milestone event payment to us.
During 2016, we recognized $15.0 million in event-based revenue under our collaboration agreement with AbbVie as a result of AbbVie initiating Phase III clinical studies of elagolix in patients with uterine fibroids.
During 2015, we entered into our collaboration and license agreement with Mitsubishi Tanabe for the development and commercialization of our VMAT2 inhibitor INGREZZA for movement disorders in Japan and other select Asian markets. Payments from Mitsubishi Tanabe under this agreement included an up-front license fee of $30 million. During 2015, we recorded revenues of $19.8 million related to the up-front license fee.
Operating Expenses
Cost of Product Sales
Cost of product sales was $1.3 million for the year ended December 31, 2017. Product sold during the year ended December 31, 2017 included drug product that was previously charged to research and development expense prior to FDA approval of INGREZZA for TD. This minimal cost drug product had a positive impact on our cost of product sales and related product gross margins for the year ended December 31, 2017. We will continue to have a lower cost of product sales that excludes the cost of the API that was produced prior to FDA approval until we manufacture API and sell INGREZZA that includes this newly manufactured API. We expect that this will be the case for the near-term and as a result, our cost of product sales in the near-term will be less than we anticipate it will be in future periods. No similar cost of product sales was recognized during 2016 or 2015.
Research and Development
Our R&D expenditures include costs related to preclinical and clinical trials, scientific personnel, equipment, consultants, sponsored research, share-based compensation and allocated facility costs. We do not
57
Table of Contents
track fully burdened R&D costs separately for each of our drug candidates. We review our R&D expenses by focusing on four categories: external development, personnel, facility and depreciation, and other. External development expenses consist of costs associated with our external preclinical and clinical trials, including pharmaceutical development and manufacturing. Personnel expenses include salaries and wages, share-based compensation, payroll taxes and benefits for those individuals involved in ongoing research and development efforts. Other R&D expenses mainly represent lab supply expenses, scientific consulting expenses and other expenses.
The following table presents our total R&D expenses by category during the periods presented:
| Years Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| (In millions) | ||||||||||||
| External development expense: |
||||||||||||
| VMAT2 |
$ | 20.9 | $ | 32.4 | $ | 29.3 | ||||||
| CRF |
3.9 | 2.5 | 3.3 | |||||||||
| Other |
3.4 | 1.0 | 1.2 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total external development expense |
28.2 | 35.9 | 33.8 | |||||||||
| R&D personnel expense |
42.2 | 34.1 | 32.8 | |||||||||
| R&D facility and depreciation expense |
5.8 | 6.3 | 6.0 | |||||||||
| Other R&D expense |
45.6 | 18.0 | 8.9 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total research and development expense |
$ | 121.8 | $ | 94.3 | $ | 81.5 | ||||||
|
|
|
|
|
|
|
|||||||
R&D expense increased by $27.5 million, from $94.3 million in 2016 to $121.8 million in 2017. This increase in R&D expense was primarily driven by the $30 million paid to BIAL to in-license opicapone. Approximately $8.1 million of the increase in R&D expense was due to higher R&D personnel related expense, offset by a decrease in VMAT2 spending due to the wind down of the Phase III clinical program and NDA preparation activities related to INGREZZA for TD during 2016.
R&D expense increased from $81.5 million in 2015 to $94.3 million in 2016. This increase was primarily due to a $9.1 million increase in other R&D expenses related to efforts around our NDA filing of INGREZZA for TD, including $2.4 million for the related FDA filing fee and an increase in scientific consulting expense of approximately $6.6 million. Additionally, external development expenses related to our INGREZZA Phase III clinical program in TD and Phase II program in Tourette syndrome increased by $3.1 million from 2015 to 2016.
We expect ongoing research and development expenses in 2018 to be higher than 2017 levels due to increased headcount in research and development, increased clinical trial activity, and post-marketing studies of INGREZZA for TD.
Sales, General and Administrative
Sales, general and administrative expense increased to $169.9 million for 2017 compared with $68.1 million during the same period in 2016. The $101.8 million increase in sales, general and administrative expense is primarily due to the launch of our commercial efforts for INGREZZA in April 2017 and is primarily due to higher personnel related costs (increased by $56.7 million), with share-based compensation costs accounting for $8.2 million of this increase in personnel costs. Additionally, external costs related to market research, patient support, commercial launch activities and other professional services were $36.6 million higher for 2017 when compared to 2016.
Sales, general and administrative expenses were $68.1 million in 2016 compared to $32.5 million in 2015. The $35.6 million increase in general and administrative expense from 2015 to 2016 was primarily due to higher
58
Table of Contents
personnel related costs associated with an increase in headcount to support our commercial launch preparations for INGREZZA in TD (increased by $13.5 million). Additionally, external costs related to market research, commercial launch preparation and other professional services were $20.2 million higher for 2016 when compared to 2015.
We expect our general and administrative expenses in 2018 to increase significantly from 2017 expense levels due to having a full year of commercialization activities related to INGREZZA for TD.
Interest Expense
As discussed above, during May 2017, we issued $517.5 million of convertible debt which resulted in $19.5 million in interest expense during 2017. $11.8 million of which relates to amortization of debt discount and debt acquisition costs. The remaining $7.7 million relates to the stated interest of which $6.2 million was paid in cash during the year. There was no related interest expense in 2016 or 2015.
We expect our 2018 interest expense to increase significantly from 2017 expense levels due to having a full year of convertible debt outstanding.
Net Loss
Our net loss for 2017 was $142.5 million, or $1.62 net loss per common share, our net loss for 2016 was $141.1 million, or $1.63 net loss per common share, and our net loss for 2015 was $88.9 million, or $1.05 net loss per common share.
Our net loss was fairly consistent from 2016 to 2017. Operating expenses increased by $130.6 million from 2016 to 2017 primarily due to the in-licensing of opicapone and the costs associated with the commercial launch of INGREZZA for TD. This increase in operating expenses was offset by a $146.6 million increase in revenue from 2016 to 2017, primarily driven by sales of INGREZZA.
The increase in our net loss from 2015 to 2016 was primarily a result of higher overall expenses associated with INGREZZA clinical activities and commercial launch preparations. In addition, revenue for 2016 decreased by $4.8 million due to the $19.8 million in revenue recognized from the up-front license fee from Mitsubishi Tanabe in 2015 being greater than the $15.0 million milestone payment received from AbbVie during 2016.
For 2018, although we anticipate significantly higher sales, we expect to have a net loss due to higher operating expenses to execute our clinical programs and and higher general and administrative expenses as we continue to execute on our commercialization plan for INGREZZA in TD. Ongoing operational R&D expenses (costs excluding the in-licensing fee of $30 million for opicapone) for 2018 are expected to be higher than 2017 levels due to increased headcount in research and development and increased clinical trial activity. Revenue is expected to increase in 2018 due to an anticipated event-based payment from AbbVie under our collaboration agreement, as well as anticipated increased product sales of INGREZZA in TD.
Liquidity and Capital Resources
At December 31, 2017, our cash, cash equivalents, and investments totaled $763.3 million compared with $350.8 million at December 31, 2016.
Net cash used in operating activities during 2017 was $94.3 million compared to $106.2 million in 2016. The net loss from 2017 increased by $1.4 million over 2016 levels but included an increase in non-cash share based compensation expense of $14.1 million and the non-cash impact of the amortization of debt discount associated with the Senior Convertible Notes during 2017 of approximately $10.9 million.
59
Table of Contents
Net cash used in operating activities during 2016 was $106.2 million compared to $38.0 million in 2015. The $68.2 million change in cash flows from operating activities is primarily due to an increase in net loss of approximately $52.2 million. In addition, during 2015, we received $30.0 million from Mitsubishi Tanabe as an upfront licensing payment; of which approximately $10.2 million was accounted for as deferred revenue.
Net cash used in investing activities was $250.9 million in 2017 and $195.8 million in 2015 as compared to $112.9 million of net cash provided by investing activities in 2016. The fluctuation in net cash used in investing activities resulted primarily from the timing differences in investment purchases, sales and maturities, and the fluctuation of our portfolio mix between cash equivalents and short-term investment holdings. The average term to maturity in our investment portfolio is less than one year.
Net cash provided by financing activities during 2017 was $516.6 million compared to $2.4 million and $277.0 million in 2016 and 2015, respectively. The increase in cash provided by financing activities from 2016 to 2017 was primarily due to net proceeds of approximately $502.8 million from our offering of senior convertible debt in May 2017. Cash provided by financing activities during 2015 included approximately $270.7 million from our public offering of common stock in February 2015. During 2017, 2016 and 2015 stock option exercises yielded approximately $13.9 million, $2.4 million and $6.3 million, respectively, in cash proceeds.
Equity Financing. In February 2015, we completed a public offering of common stock in which we sold 8.0 million shares of our common stock at an offering price of $36.00 per share. The shares were sold pursuant to a shelf registration statement with the Securities and Exchange Commission (SEC). The net proceeds generated from this transaction, after underwriting discounts and commissions and offering costs, were approximately $270.7 million.
Shelf Registration Statement. In February 2017, we filed an automatic shelf registration statement which immediately became effective by rule of the SEC. For so long as we continue to satisfy the requirements to be deemed a well-known seasoned issuer, this shelf registration statement allows us to issue an unlimited number of securities from time to time. As of December 31, 2017, we had not sold any securities under this shelf registration statement.
Convertible Debt. In May 2017, we issued $517.5 million aggregate principal amount of 2.25% Convertible Senior Notes due May 2024 (2024 Notes). Debt issuance costs of approximately $14.7 million were primarily comprised of initial purchasers’ discounts and commissions, legal, accounting, and other professional fees, a portion of which was capitalized and are recorded as a reduction to long-term debt and is being amortized as interest expense using the effective interest method over the seven-year term of the 2024 Notes. The remaining debt issuance costs were allocated as a component of equity in additional paid-in capital.
The 2024 Notes will be convertible into cash, shares of common stock, or a combination of cash and shares of common stock based on an initial conversion rate, subject to adjustment, of 13.1711 shares per $1,000 principal amount of the 2024 Notes (which represents an initial conversion price of approximately $75.92 per share). The 2024 Notes will mature on May 15, 2024, unless earlier converted, redeemed or repurchased. Prior to the close of business on the business day immediately preceding January 15, 2024, the 2024 Notes will be convertible at the option of the holders only upon satisfaction of certain conditions. Thereafter, the 2024 Notes will be convertible at the option of the holders at any time up until the close of business on the scheduled trading day immediately preceding May 15, 2024. We may not redeem the 2024 Notes prior to May 15, 2021. On or after this date, at our election, we may redeem all, or any portion, of the 2024 Notes for cash if the last reported sales price of our common stock, for at least 20 trading days (whether or not consecutive) during the period of 30 consecutive trading days ending on the last trading day immediately preceding the date which we send the related redemption notice, is more than 130% of the conversion price of the 2024 Notes in effect on each applicable trading day.
60
Table of Contents
Factors That May Affect Future Financial Condition and Liquidity
We anticipate increases in expenditures as we execute on our commercialization plan for INGREZZA and continue our R&D activities. Because of our limited financial resources, our strategies to develop some of our programs include collaborative agreements with major pharmaceutical companies and sales of our securities in both public and private offerings. Our collaborative agreements typically include a partial recovery of our research costs through license fees, contract research funding and milestone revenues. Our collaborators are also financially and managerially responsible for clinical development and commercialization. In these cases, the estimated completion date would largely be under the control of the collaborator. We cannot forecast, with any degree of certainty, which other proprietary products or indications, if any, will be subject to future collaborative arrangements, in whole or in part, and how such arrangements would affect our capital requirements.
Our in-license, research and clinical development agreements are generally cancelable with written notice within 180 days or less. In addition to the minimum annual payments due under certain in-license and research agreements, including a $30 million upfront license fee paid to BIAL in February 2017, we may be required to pay up to approximately $132 million in milestone payments, plus sales royalties, in the event that all scientific research, development and commercialization milestones under these agreements are achieved.
From time to time, we may be subject to legal proceedings and claims in the ordinary course of business. On December 1, 2015, Icahn School of Medicine at Mount Sinai (Mount Sinai) filed a complaint against us in the United States District Court for the Southern District of New York: Icahn School of Medicine at Mount Sinai v. Neurocrine Biosciences, Inc., Case No. 1:15-cv-09414. In the complaint, Mount Sinai alleges that we, by entering into an exclusive worldwide collaboration with AbbVie to develop and commercialize next-generation GnRH antagonists, breached our license agreement with Mount Sinai dated August 27, 1999 (Mount Sinai License). Mount Sinai is seeking unspecified monetary damages, future sublicensing fees and attorney’s fees. In January 2016, we filed a motion to dismiss this complaint in its entirety. In June 2016, the Court denied the motion in part and granted the motion in part, ruling that while Mount Sinai could continue its lawsuit against us, there was no requirement for us to obtain Mount Sinai’s consent prior to licensing the next-generation GnRH antagonists to AbbVie. In July 2016, we filed our answer denying Mount Sinai’s allegations, and filed counterclaims against Mount Sinai alleging patent misuse, non-infringement of Mount Sinai’s patents, and that Mount Sinai’s patents that are subject to the Mount Sinai License are invalid. Mount Sinai has filed a motion to dismiss our counterclaims and affirmative defenses. We believe that we have meritorious defenses to the claims made in the complaint and intend to vigorously defend ourselves against such claims, but we are not able to predict the ultimate outcome of this action, or estimate any potential loss.
We lease our office and research laboratories under an operating lease with an initial term that expires at the end of 2029.
As of December 31, 2017, the total estimated future annual minimum lease payments under our non-cancelable operating lease obligations are as follows (in thousands):
| Payment Amount |
||||
| Year ending: |
||||
| 2018 |
$ | 7,368 | ||
| 2019 |
7,589 | |||
| 2020 |
7,844 | |||
| 2021 |
8,079 | |||
| 2022 and thereafter |
74,000 | |||
|
|
|
|||
| Total future minimum lease payments |
$ | 104,880 | ||
|
|
|
|||
As discussed above, the 2024 Notes (face value of $517.5 million) will mature on May 15, 2024, unless earlier converted, redeemed or repurchased. We may not redeem the 2024 Notes prior to May 15, 2021. On or
61
Table of Contents
after this date, at our election, we may redeem all, or any portion, of the 2024 Notes under certain circumstances. The 2024 Notes do not contain any financial or operating covenants or any restrictions on the payment of dividends, the issuance of other indebtedness or the issuance or repurchase of securities by us. There are customary events of default with respect to the 2024 Notes, including that upon certain events of default, 100% of the principal and accrued and unpaid interest on the 2024 Notes will automatically become due and payable.
The funding necessary to execute our business strategies is subject to numerous uncertainties, which may adversely affect our liquidity and capital resources. Marketing of approved pharmaceuticals and completion of clinical trials may take several years or more, but the length of time generally varies substantially according to the type, complexity, novelty and intended use of a product candidate. It is also important to note that if a clinical candidate is identified, the further development of that candidate can be halted or abandoned at any time due to a number of factors. These factors include, but are not limited to, funding constraints, safety or a change in market demand.
The nature and efforts required to develop our product candidates into commercially viable products include research to identify a clinical candidate, preclinical development, clinical testing, FDA approval and commercialization. For each drug candidate that successfully completes all stages of R&D, and is commercialized, total R&D spending in the pharmaceutical industry may exceed $2 billion. Additionally, the stages of research and development can take in excess of ten years to complete for each drug candidate.
We test our potential product candidates in numerous preclinical studies to identify disease indications for which our product candidates may show efficacy. We may conduct multiple clinical trials to cover a variety of indications for each product candidate. As we obtain results from trials, we may elect to discontinue clinical trials for certain product candidates or for certain indications in order to focus our resources on more promising product candidates or indications. The duration and the cost of clinical trials may vary significantly over the life of a project as a result of differences arising during the clinical trial protocol, including, among others, the following:
| • | we or the FDA or similar foreign regulatory authorities may suspend the trials; |
| • | we may discover that a product candidate may cause harmful side effects; |
| • | patient recruitment may be slower than expected; and |
| • | patients may drop out of the trials. |
For each of our programs, we periodically assess the scientific progress and merits of the programs to determine if continued R&D is economically viable. Certain of our programs have been terminated due to the lack of scientific progress and lack of prospects for ultimate commercialization. Because of the uncertainties associated with R&D of these programs, we may not be successful in achieving commercialization. As such, the ultimate timeline and costs to commercialize a product cannot be accurately estimated.
Other than INGREZZA, which has been approved by the FDA for the treatment of TD, our product candidates have not yet achieved FDA regulatory approval, which is required before we can market them as therapeutic products in the United States. In order to proceed to subsequent clinical trial stages and to ultimately achieve regulatory approval, the FDA must conclude that our clinical data establish safety and efficacy. We must satisfy the requirements of similar regulatory authorities in foreign countries in order to market products in those countries. The results from preclinical testing and early clinical trials may not be predictive of results in later clinical trials. It is possible for a candidate to show promising results in clinical trials, but subsequently fail to establish sufficient safety and efficacy data necessary to obtain regulatory approvals.
As a result of the uncertainties discussed above, among others, the duration and completion costs of our R&D projects, clinical trials, and post-marketing studies are difficult to estimate and are subject to considerable
62
Table of Contents
variation. Our inability to complete our R&D projects in a timely manner or our failure to enter into collaborative agreements, when appropriate, could significantly increase our capital requirements and could adversely impact our liquidity. These uncertainties could force us to seek additional, external sources of financing from time to time in order to continue with our business strategy. Our inability to raise additional capital, or to do so on terms reasonably acceptable to us, would jeopardize the future success of our business.
We currently have limited experience in marketing and selling pharmaceutical products. If we fail to maintain successful marketing, sales, and reimbursement capabilities, or fail to enter into successful arrangements with third parties, our product revenues may suffer. We also may be required to make further substantial expenditures if unforeseen difficulties arise in other areas of our business. In particular, our future capital requirements will depend on many factors, including:
| • | continued scientific progress in our R&D programs; |
| • | the magnitude of our R&D programs; |
| • | progress with preclinical testing and clinical trials; |
| • | the time and costs involved in obtaining regulatory approvals; |
| • | the costs involved in filing and pursuing patent applications and enforcing patent claims; |
| • | competing technological and market developments; |
| • | the establishment of additional collaborations and strategic alliances; |
| • | developments related to on-going litigation; |
| • | the cost of manufacturing facilities and of commercialization activities and arrangements; and |
| • | the cost of product in-licensing and any possible acquisitions. |
We believe that our existing capital resources, together with investment income and future payments due under our strategic alliances, will be sufficient to satisfy our current and projected funding requirements for at least the next 12 months. However, we cannot guarantee that our existing capital resources and anticipated revenues will be sufficient to conduct and complete all of our research and development programs or commercialization activities as planned.
We may require additional funding to effectively commercialize INGREZZA, to continue our research and product development programs, to conduct preclinical studies and clinical trials, for operating expenses, to pursue regulatory approvals for our product candidates, for the costs involved in filing and prosecuting patent applications and enforcing or defending patent claims, if any, the cost of product in-licensing and any possible acquisitions, and we may require additional funding to establish manufacturing and marketing capabilities in the future. We may seek to access the public or private equity markets whenever conditions are favorable. For example, we have an effective shelf registration statement on file with the SEC which allows us to issue an unlimited number of shares of our securities from time to time. In addition, during the second quarter of 2017, we issued $517.5 million of convertible debt and we have previously financed capital purchases and may continue to pursue opportunities to obtain additional debt financing in the future. We may also seek additional funding through strategic alliances or other financing mechanisms. We cannot assure you that adequate funding will be available on terms acceptable to us, if at all. Any additional equity financings will be dilutive to our stockholders and any additional debt may involve operating covenants that may restrict our business. If adequate funds are not available through these means, we may be required to curtail significantly one or more of our research or development programs or obtain funds through arrangements with collaborators or others. This may require us to relinquish rights to certain of our technologies, products or product candidates. To the extent that we are unable to obtain third-party funding for such expenses, we expect that increased expenses will result in increased cash flow losses from operations. We cannot assure you that we will successfully develop our products under development or that our approved products will generate revenues sufficient to enable us to earn a profit.
63
Table of Contents
Interest Rate Risk
We are exposed to interest rate risk on our short-term investments. The primary objective of our investment activities is to preserve principal while at the same time maximizing yields without significantly increasing risk. To achieve this objective, we invest in highly liquid and high quality government and other debt securities. To minimize our exposure due to adverse shifts in interest rates, we invest in short-term securities and ensure that the maximum average maturity of our investments does not exceed 12 months. If a 10% change in interest rates were to have occurred on December 31, 2017, this change would not have had a material effect on the fair value of our investment portfolio as of that date. Due to the short holding period of our investments, we have concluded that we do not have a material financial market risk exposure.
New Accounting Pronouncements
In May 2014 the FASB issued Accounting Standards Update (ASU) No. 2014-09, Revenue from Contracts with Customers (Topic 606), which supersedes all existing revenue recognition requirements, including most industry-specific guidance. This new standard requires a company to recognize revenue when it transfers goods or services to customers in an amount that reflects the consideration that the company expects to receive for those goods or services. The FASB subsequently issued amendments to ASU No. 2014-09 that have the same effective date and transition date. These new standards became effective for us on January 1, 2018, and will be adopted using the modified retrospective method through a cumulative-effect adjustment, if any, directly to retained earnings as of that date. We have performed a review of these new standards as compared to our current accounting policies for customer contracts and collaborative relationships. As of December 31, 2017, we have not identified any accounting changes that would materially impact the amount of reported revenues with respect to our product revenues. We are currently evaluating the impact of the new standard on historical revenue recorded for our two collaboration agreements. This ongoing evaluation is dependent upon the resolution of certain questions relating to the application of the new revenue recognition guidance for collaboration agreements which will ultimately determine the impact, if any, the adoption of this standard may have on the consolidated financial statements.
We expect the accounting for contingent milestone payments to be the most significant change in accounting for our license and collaboration agreements. Topic 605 provides guidance specific to the accounting for milestone payments, including the ability to defer the recognition of any milestones until received and, if certain criteria are met, the ability to recognize milestone payments as revenue when received. However, Topic 606 does not contain guidance specific to milestone payments, thereby requiring potential milestone payments to be considered in accordance with the overall model of Topic 606. As a result, revenues from contingent milestone payments may be recognized earlier under Topic 606 than under Topic 605, based on an assessment of the probability of achievement of the milestone and the likelihood of a significant reversal of such milestone revenue at each reporting date. This assessment may result in recognizing milestone revenue before the milestone event has been achieved. In addition, Topic 606 changes guidance regarding the accounting for variable consideration received from licensees, which may impact the estimation of, and determination of the timing of, the related revenue recognition.
In February 2016, the FASB issued Accounting Standards Update 2016-02 Leases. This update amends the current accounting guidance for lease transactions. Under the new guidance, a lessee will be required to recognize both assets and liabilities for any leases in excess of twelve months. Additionally, certain qualitative and quantitative disclosures will also be required in the financial statements. We are required to adopt this new guidance beginning in 2019 and early adoption is permitted. We are in the process of determining the effects the adoption of this update will have on our consolidated financial statements.
In November 2016, the FASB issued ASU 2016-18, “Statement of Cash Flows (Topic 230): Restricted Cash”, which clarifies the presentation of restricted cash and restricted cash equivalents in the statements of cash flows. Under the ASU, restricted cash and restricted cash equivalents are included with cash and cash equivalents
64
Table of Contents
when reconciling the beginning-of-period and end-of-period total amounts presented on the statements of cash flows. The ASU is intended to reduce diversity in practice in the classification and presentation of changes in restricted cash on the statement of cash flows. The ASU requires that the statement of cash flows explain the change in total cash and equivalents and amounts generally described as restricted cash or restricted cash equivalents when reconciling the beginning-of-period and end-of-period total amounts. The ASU also requires a reconciliation between the total of cash and equivalents and restricted cash presented on the statement of cash flows and the cash and equivalents balance presented on the balance sheet. The ASU was effective for us on January 1, 2018. We do not expect the adoption of the ASU will have a material effect on our results of operations, financial condition or cash flows.
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Information required by this item is contained in “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations – Interest Rate Risk.” Such information is incorporated herein by reference.
65
Table of Contents
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
NEUROCRINE BIOSCIENCES, INC.
INDEX TO THE CONSOLIDATED FINANCIAL STATEMENTS
66
Table of Contents
Report of Independent Registered Public Accounting Firm
To the shareholders and the board of directors of
Neurocrine Biosciences, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Neurocrine Biosciences, Inc. (the “Company”) as of December 31, 2017 and 2016, the related consolidated statements of operations and comprehensive loss, stockholders’ equity, and cash flows, for each of the three years in the period ended December 31, 2017 and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2017 and 2016, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2017, in conformity with US generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company’s internal control over financial reporting as of December 31, 2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated February 13, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the US federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures include examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 1992.
San Diego, California
February 13, 2018
67
Table of Contents
NEUROCRINE BIOSCIENCES, INC.
(In thousands, except for par value and share totals)
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| ASSETS | ||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 254,712 | $ | 83,267 | ||||
| Short-term investments, available-for-sale |
261,217 | 224,083 | ||||||
| Accounts receivable |
31,127 | — | ||||||
| Other current assets |
7,863 | 3,092 | ||||||
|
|
|
|
|
|||||
| Total current assets |
554,919 | 310,442 | ||||||
| Property and equipment, net |
10,811 | 6,271 | ||||||
| Long-term investments, available-for-sale |
247,361 | 43,490 | ||||||
| Restricted cash |
4,500 | 4,883 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 817,591 | $ | 365,086 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities: |
||||||||
| Accounts payable and accrued liabilities |
$ | 53,520 | $ | 26,182 | ||||
| Current portion of deferred rent |
— | 470 | ||||||
| Current portion of cease-use liability |
62 | 236 | ||||||
| Current portion of deferred gain on sale of real estate |
731 | 3,526 | ||||||
| Other current liabilities |
113 | — | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
54,426 | 30,414 | ||||||
| Deferred gain on sale of real estate |
8,043 | 7,372 | ||||||
| Deferred revenue |
10,231 | 10,231 | ||||||
| Deferred rent |
3,135 | 1,462 | ||||||
| Convertible senior notes |
369,618 | — | ||||||
| Cease-use liability |
— | 617 | ||||||
| Other liabilities |
— | 113 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
445,453 | 50,209 | ||||||
| Commitments and contingencies |
||||||||
| Stockholders’ equity: |
||||||||
| Preferred stock, $0.001 par value; 5,000,000 shares authorized; no shares issued and outstanding |
— | — | ||||||
| Common stock, $0.001 par value; 220,000,000 shares authorized; issued and outstanding shares were 88,793,903 and 86,883,300 at December 31, 2017 and 2016, respectively |
89 | 87 | ||||||
| Additional paid-in capital |
1,572,765 | 1,371,432 | ||||||
| Accumulated other comprehensive loss |
(1,850 | ) | (318 | ) | ||||
| Accumulated deficit |
(1,198,866 | ) | (1,056,324 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders’ equity |
372,138 | 314,877 | ||||||
|
|
|
|
|
|||||
| Total liabilities and stockholders’ equity |
$ | 817,591 | $ | 365,086 | ||||
|
|
|
|
|
|||||
See accompanying notes.
68
Table of Contents
NEUROCRINE BIOSCIENCES, INC.
Consolidated Statements of Operations and Comprehensive Loss
(In thousands, except net loss per share data)
| Year Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Revenues: |
||||||||||||
| Product sales, net |
$ | 116,626 | $ | — | $ | — | ||||||
| Milestones and license fees |
45,000 | $ | 15,000 | $ | 19,769 | |||||||
|
|
|
|
|
|
|
|||||||
| Total revenues |
161,626 | 15,000 | 19,769 | |||||||||
| Operating expenses: |
||||||||||||
| Cost of product sales |
1,254 | — | — | |||||||||
| Research and development |
121,827 | 94,291 | 81,491 | |||||||||
| Sales, general and administrative |
169,906 | 68,081 | 32,480 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
292,987 | 162,372 | 113,971 | |||||||||
|
|
|
|
|
|
|
|||||||
| Loss from operations |
(131,361 | ) | (147,372 | ) | (94,202 | ) | ||||||
| Other income: |
||||||||||||
| Deferred gain on real estate |
2,124 | 3,423 | 3,325 | |||||||||
| Interest expense |
(19,523 | ) | — | — | ||||||||
| Investment income, net |
6,238 | 2,838 | 1,928 | |||||||||
| Other (expense) income, net |
(20 | ) | 21 | 20 | ||||||||
|
|
|
|
|
|
|
|||||||
| Total other income |
(11,181 | ) | 6,282 | 5,273 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net loss |
$ | (142,542 | ) | $ | (141,090 | ) | $ | (88,929 | ) | |||
|
|
|
|
|
|
|
|||||||
| Net loss per common share: |
||||||||||||
| Basic |
$ | (1.62 | ) | $ | (1.63 | ) | $ | (1.05 | ) | |||
|
|
|
|
|
|
|
|||||||
| Diluted |
$ | (1.62 | ) | $ | (1.63 | ) | $ | (1.05 | ) | |||
|
|
|
|
|
|
|
|||||||
| Shares used in the calculation of net loss per common share: |
||||||||||||
| Basic |
88,089 | 86,713 | 84,496 | |||||||||
|
|
|
|
|
|
|
|||||||
| Diluted |
88,089 | 86,713 | 84,496 | |||||||||
|
|
|
|
|
|
|
|||||||
| Other comprehensive loss: |
||||||||||||
| Net loss |
$ | (142,542 | ) | $ | (141,090 | ) | $ | (88,929 | ) | |||
| Net unrealized (losses) gains on available-for-sale securities |
(1,532 | ) | 659 | (700 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Comprehensive loss |
$ | (144,074 | ) | $ | (140,431 | ) | $ | (89,629 | ) | |||
|
|
|
|
|
|
|
|||||||
See accompanying notes.
69
Table of Contents
NEUROCRINE BIOSCIENCES, INC.
Consolidated Statements of Stockholders’ Equity
(In thousands)
| Common Stock | Accumulated Other Comprehensive (Loss) Gain |
Accumulated Deficit |
Total Stockholders’ Equity |
|||||||||||||||||||||
| Shares | Amount | Additional Paid in Capital |
||||||||||||||||||||||
| BALANCE AT DECEMBER 31, 2014 |
76,466 | $ | 76 | $ | 1,035,205 | $ | (277 | ) | $ | (826,305 | ) | $ | 208,699 | |||||||||||
| Net loss |
— | — | — | — | (88,929 | ) | (88,929 | ) | ||||||||||||||||
| Unrealized losses on investments |
— | — | — | (700 | ) | — | (700 | ) | ||||||||||||||||
| Share-based compensation |
— | — | 28,392 | — | — | 28,392 | ||||||||||||||||||
| Issuance of common stock for restricted share units vested |
503 | 1 | — | — | — | 1 | ||||||||||||||||||
| Issuance of common stock for option exercises |
1,308 | 1 | 6,303 | — | — | 6,304 | ||||||||||||||||||
| Issuance of common stock, net of offering costs |
7,986 | 8 | 270,679 | — | — | 270,687 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| BALANCE AT DECEMBER 31, 2015 |
86,263 | $ | 86 | $ | 1,340,579 | $ | (977 | ) | $ | (915,234 | ) | $ | 424,454 | |||||||||||
| Net loss |
— | — | — | — | (141,090 | ) | (141,090 | ) | ||||||||||||||||
| Unrealized losses on investments |
— | — | — | 659 | — | 659 | ||||||||||||||||||
| Share-based compensation |
— | — | 28,464 | — | — | 28,464 | ||||||||||||||||||
| Issuance of common stock for restricted share units vested |
284 | — | — | — | — | — | ||||||||||||||||||
| Issuance of common stock for option exercises |
336 | 1 | 2,389 | — | — | 2,390 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| BALANCE AT DECEMBER 31, 2016 |
86,883 | $ | 87 | $ | 1,371,432 | $ | (318 | ) | $ | (1,056,324 | ) | $ | 314,877 | |||||||||||
| Net loss |
— | — | — | — | (142,542 | ) | (142,542 | ) | ||||||||||||||||
| Unrealized gains on investments |
— | — | — | (1,532 | ) | — | (1,532 | ) | ||||||||||||||||
| Share-based compensation |
— | — | 42,522 | — | 42,522 | |||||||||||||||||||
| Issuance of common stock for restricted share units vested |
562 | 1 | — | — | — | 1 | ||||||||||||||||||
| Issuance of common stock for option exercises |
1,349 | 1 | 13,863 | — | — | 13,864 | ||||||||||||||||||
| Equity component of convertible debt, net of issuance costs |
— | — | 144,948 | — | — | 144,948 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| BALANCE AT DECEMBER 31, 2017 |
88,794 | $ | 89 | $ | 1,572,765 | $ | (1,850 | ) | $ | (1,198,866 | ) | $ | 372,138 | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
See accompanying notes.
70
Table of Contents
NEUROCRINE BIOSCIENCES, INC.
Consolidated Statements of Cash Flows
(In thousands)
| Years Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| CASH FLOW FROM OPERATING ACTIVITIES |
||||||||||||
| Net loss |
$ | (142,542 | ) | $ | (141,090 | ) | $ | (88,929 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||||||
| Depreciation and amortization |
2,400 | 1,453 | 1,009 | |||||||||
| Gain on sale of assets, net |
(2,104 | ) | (3,431 | ) | (3,334 | ) | ||||||
| Cease-use expense |
(544 | ) | (584 | ) | (85 | ) | ||||||
| Amortization of debt issuance costs |
848 | — | — | |||||||||
| Amortization of debt discount |
10,937 | — | — | |||||||||
| Deferred revenues |
— | — | 10,231 | |||||||||
| Deferred rent |
1,203 | (294 | ) | (16 | ) | |||||||
| Amortization of premiums on investments |
1,756 | 3,520 | 6,032 | |||||||||
| Non-cash share-based compensation expense |
42,522 | 28,464 | 28,392 | |||||||||
| Change in operating assets and liabilities: |
||||||||||||
| Accounts receivable |
(31,127 | ) | — | — | ||||||||
| Inventory |
(1,024 | ) | — | — | ||||||||
| Other current assets |
(3,747 | ) | 1,791 | (489 | ) | |||||||
| Accounts payable and accrued liabilities |
27,338 | 4,398 | 9,841 | |||||||||
| Cease-use liability |
(247 | ) | (300 | ) | (610 | ) | ||||||
| Other liabilities |
— | (108 | ) | (39 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Net cash used in operating activities |
(94,331 | ) | (106,181 | ) | (37,997 | ) | ||||||
| CASH FLOW FROM INVESTING ACTIVITIES |
||||||||||||
| Purchases of investments |
(583,408 | ) | (298,776 | ) | (449,052 | ) | ||||||
| Sales/maturities of investments |
339,088 | 415,826 | 255,123 | |||||||||
| Deposits and restricted cash |
383 | (92 | ) | 40 | ||||||||
| Proceeds from sales of property and equipment |
7 | 13 | 9 | |||||||||
| Purchases of property and equipment |
(6,940 | ) | (4,108 | ) | (1,934 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net cash (used in) provided by investing activities |
(250,870 | ) | 112,863 | (195,814 | ) | |||||||
| CASH FLOW FROM FINANCING ACTIVITIES |
||||||||||||
| Issuance of common stock |
13,865 | 2,390 | 276,992 | |||||||||
| Proceeds from issuance of senior convertible notes, net |
502,781 | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by financing activities |
516,646 | 2,390 | 276,992 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net change in cash and cash equivalents |
171,445 | 9,072 | 43,181 | |||||||||
| Cash and cash equivalents at beginning of the year |
83,267 | 74,195 | 31,014 | |||||||||
|
|
|
|
|
|
|
|||||||
| Cash and cash equivalents at end of the year |
$ | 254,712 | $ | 83,267 | $ | 74,195 | ||||||
|
|
|
|
|
|
|
|||||||
| SUPPLEMENTAL DISCLOSURES |
||||||||||||
| Interest paid |
$ | 6,242 | $ | — | $ | — | ||||||
| Taxes paid |
$ | — | $ | — | $ | — | ||||||
See accompanying notes.
71
Table of Contents
NEUROCRINE BIOSCIENCES, INC.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
December 31, 2017
NOTE 1. ORGANIZATION AND SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Business Activities. Neurocrine Biosciences, Inc. (the Company or Neurocrine) was incorporated in California in 1992 and reincorporated in Delaware in 1996. The Company discovers and develops innovative and life-changing pharmaceuticals, in diseases with high unmet medical needs through its novel research and development (R&D) platform focused on neurological, psychological, and endocrine based diseases and disorders. In April 2017, the Company received approval from the United States Food and Drug Administration (FDA) for INGREZZA® (valbenazine), a vesicular monoamine transporter 2 (VMAT2) inhibitor for the treatment of TD. The Company’s three lead late-stage clinical programs are elagolix, a gonadotropin-releasing hormone (GnRH) antagonist for women’s health that is partnered with AbbVie Inc. (AbbVie), INGREZZA for Tourette syndrome and opicapone, a highly-selective catechol-O-methyltransferase inhibitor for the treatment of Parkinson’s disease.
Neurocrine Continental, Inc., is a Delaware corporation and a wholly owned subsidiary of the Company. The Company also has two wholly-owned Irish subsidiaries, Neurocrine Therapeutics, Ltd. and Neurocrine Europe, Ltd. which were formed in December 2014, both of which are inactive.
Principles of Consolidation. The consolidated financial statements include the accounts of Neurocrine as well as its wholly owned subsidiaries. The Company does not have any significant interests in any variable interest entities. All intercompany transactions and balances have been eliminated in consolidation.
Use of Estimates. The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America (GAAP) requires management to make estimates and assumptions that affect the amounts reported in the financial statements and the accompanying notes. Actual results could differ from those estimates.
Cash Equivalents. The Company considers all highly liquid investments that are readily convertible into cash and have an original maturity of three months or less at the time of purchase to be cash equivalents.
Short-Term and Long-Term Investments Available-for-Sale. Certain investments are classified as available-for-sale and, in accordance with authoritative guidance, are carried at fair value, with the unrealized gains and losses reported in other comprehensive loss. The amortized cost of debt securities in this category is adjusted for amortization of premiums and accretion of discounts to maturity. Such amortization and accretion is included in investment income. Realized gains and losses and declines in value judged to be other-than-temporary, if any, on available-for-sale securities are included in other income or expense. The cost of securities sold is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in investment income.
Concentration of Credit Risk. Financial instruments that potentially subject the Company to concentrations of credit risk consist primarily of cash, cash equivalents, investments and accounts receivables. The Company has established guidelines to limit its exposure to credit risk by placing investments with high credit quality financial institutions, diversifying its investment portfolio and placing investments with maturities that maintain safety and liquidity.
Inventory. Inventory is stated at the lower of cost or estimated net realizable value. The Company currently uses actual costing to determine the cost basis for its inventory. Inventory is valued on a first-in, first-out basis and consists primarily of third-party manufacturing costs. The Company capitalizes inventory costs associated with its products upon regulatory approval when, based on management’s judgment, future commercialization is considered probable and the future economic benefit is expected to be realized; otherwise, such costs are expensed.
72
Table of Contents
Prior to FDA approval of INGREZZA, all costs related to its manufacturing were charged to research and development expense in the period incurred. At December 31, 2017, the Company’s physical inventory included active pharmaceutical product that had been produced prior to FDA approval of INGREZZA and accordingly has no cost basis as the cost associated with producing this material was expensed rather than capitalized in accordance with authoritative guidance. Additionally, manufacturing of bulk drug product, finished bottling and other labeling activities that occurred post FDA approval are included in the inventory value at December 31, 2017.
The Company provides reserves for potential excess, dated or obsolete inventory based on an analysis of forecasted demand compared to quantities on hand and any firm purchase orders, as well as product shelf life. To date, the Company has determined that such reserves are not required.
Cost of Product Sales. Cost of product sales consists of third-party manufacturing costs, transportation and freight, and indirect overhead costs associated with the manufacture and distribution of INGREZZA. Cost of product sales may also include period costs related to certain inventory manufacturing services, inventory adjustment charges as well as manufacturing variances. A significant portion of the cost of producing the product sold during the year ended December 31, 2017 was expensed as R&D prior to the Company’s New Drug Application (NDA) approval for INGREZZA and therefore is not included in the cost of product sales during this period.
Accounts Receivable. Accounts receivable are recorded net of customer allowances for prompt payment discounts, chargebacks, and any allowance for doubtful accounts. The Company estimates the allowance for doubtful accounts based on existing contractual payment terms, actual payment patterns of its customers and individual customer circumstances. To date, the Company has determined that an allowance for doubtful accounts is not required.
Advertising Expense. In connection with the FDA approval and commercial launch of INGREZZA in 2017, the Company began to incur advertising costs. Advertising costs are expensed when services are performed or goods are delivered. The Company incurred $10.1 million in advertising costs in 2017 related to its marketed product, INGREZZA. No advertising costs were capitalized as prepaid expenses at December 31, 2017.
Property and Equipment. Property and equipment are stated at cost and depreciated over the estimated useful lives of the assets using the straight-line method. Equipment is depreciated over an average estimated useful life of three to seven years. Leasehold improvements are depreciated over the shorter of their estimated useful lives or the remaining lease term.
Industry Segment and Geographic Information. The Company operates in a single industry segment – the discovery, development and marketing of therapeutics for the treatment of neurological and endocrine based diseases and disorders. The Company had no foreign based operations during any of the years presented. All revenue is generated in the United States.
Impairment of Long-Lived Assets. The Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. If indicators of impairment exist, the Company assesses the recoverability of the affected long-lived assets by determining whether the carrying value of such assets can be recovered through undiscounted future operating cash flows. If the carrying amount is not recoverable, the Company measures the amount of any impairment by comparing the carrying value of the asset to the present value of the expected future cash flows associated with the use of the asset.
Fair Value of Financial Instruments. Certain financial instruments, including cash and cash equivalents, accounts receivable, accounts payable, and accrued liabilities, are carried at cost, which management believes approximates fair value because of the short-term maturity of these instruments. Convertible senior notes were recorded at the estimated value of a similar non-convertible instrument on the date of issuance and accretes to
73
Table of Contents
face value of the notes over the seven year term. The fair value of the 2024 Notes is estimated utilizing market quotations from an over-the-counter trading market. As of December 31, 2017, the fair value approximated 128 percent of the face principal amount of the convertible senior notes.
Research and Development Expenses. R&D expenses consist primarily of salaries, payroll taxes, employee benefits, and share-based compensation charges, for those individuals involved in ongoing R&D efforts; as well as scientific contractor fees, preclinical and clinical trial costs, R&D facilities costs, laboratory supply costs, and depreciation of scientific equipment. All such costs are charged to R&D expense as incurred. These expenses result from the Company’s independent R&D efforts as well as efforts associated with collaborations and in-licensing arrangements. In addition, the Company funds R&D at other companies and research institutions under agreements, which are generally cancelable. The Company reviews and accrues clinical trial expenses based on work performed, which relies on estimates of total costs incurred based on patient enrollment, completion of patient studies and other events. The Company follows this method since reasonably dependable estimates of the costs applicable to various stages of a research agreement or clinical trial can be made. Accrued clinical costs are subject to revisions as trials progress. Revisions are charged to expense in the period in which the facts that give rise to the revision become known.
Revenue Recognition.
Product Sales, Net. The Company’s net product sales consist of U.S. sales of INGREZZA and are recognized when (i) persuasive evidence of an arrangement exists, (ii) delivery has occurred and title to the product and associated risk of loss has passed to the customer, (iii) the price is fixed or determinable, and (iv) collectability is reasonably assured.
INGREZZA was approved by the FDA on April 11, 2017 and the Company commenced shipments of INGREZZA to select pharmacies (SPs) and a select distributor (SD) in late April 2017. The SPs dispense product to a patient based on the fulfillment of a prescription and the SD sells product to government facilities, long-term care pharmacies or in-patient hospital pharmacies. The Company’s agreements with SPs and SDs provide for transfer of title to the product at the time the product is delivered to the SP or SD. In addition, except for limited circumstances, the SPs and SDs have no right of product return to the Company.
The Company has determined it can reasonably estimate its sales allowances at the time title and risk of loss transfers to the SP or SD. Therefore, the Company records revenue when the product is delivered to the SPs or SD, which is an approach frequently referred to as the “sell-in” revenue recognition model.
The Company recognizes revenue from product sales net of the following allowances:
Distribution Fees: Distribution fees include fees paid to the SPs and SD for data, prompt payment discounts and other off-invoice discounts. Distribution fees are recorded as an offset to gross revenue based on contractual terms at the time revenue from the sale is recognized.
Government Rebates: Government rebates include mandated discounts under the Medicaid Drug Rebate Program and the Medicare Part D prescription drug benefit. Rebates are amounts owed after the final dispensing of the product to a benefit plan participant and result from contractual agreements with, or statutory requirements pertaining to, Medicaid and Medicare benefit providers. The allowance for rebates is based on statutory discount rates, expected utilization and expected trends in net reimbursement. Expected utilization of rebates is based on prescription level detail provided by the SPs that includes insurer amounts for each prescription filled during the period as well as the limited amount of product that is in the SPs warehouse at period end.
Chargebacks: Chargebacks are discounts that relate to contracts with government and other entities purchasing from the SD at a discounted price. The SD charges back to the Company the difference between the price initially paid by the SD and the discounted price paid to the SD by these entities. Chargebacks are recorded as an offset to gross revenue at the time revenue from the product sale is recognized based on known and expected SD sales to these entities.
74
Table of Contents
Co-Payment Assistance: The Company offers co-payment assistance to commercially insured patients meeting certain eligibility requirements. Co-payment assistance is recorded as an offset to gross revenue at the time revenue from the product sale is recognized based on expected and actual program participation. Expected program participation is based on prescription level detail provided by the SPs that includes copay amounts for each prescription filled during the period as well as the limited amount of product that is in the SPs warehouse at period end.
Product Returns: The Company offers the SPs and SD limited product return rights for damages and shipment errors provided it is within a very limited period after the original shipping date as set forth in the applicable individual distribution agreement. The Company does not allow product returns for product that has been dispensed to a patient or for drug expiration. The Company receives real-time shipping reports and inventory reports from the SPs and SD and has the ability to control the amount of product that is sold to the SPs and SD. It is also able to make a reasonable estimate of potential product returns due to the limited time-frame allowed for the SPs and SD to process returns due to shipment error or damaged product.
Collaborative Research and Development Agreements.
Since 2011, the Company has followed the Accounting Standards Codification (ASC) for Revenue Recognition – Multiple-Element Arrangements, if applicable, to determine the recognition of revenue under license and collaboration agreements. The ASC provides guidance relating to the separation of deliverables included in an arrangement into different units of accounting and the allocation of consideration to the units of accounting. The evaluation of multiple-element arrangements requires management to make judgments about (i) the identification of deliverables, (ii) whether such deliverables are separable from the other aspects of the contractual relationship, (iii) the estimated selling price of each deliverable, and (iv) the expected period of performance for each deliverable.
To determine the units of accounting under a multiple-element arrangement, the Company evaluates certain separation criteria, including whether the deliverables have stand-alone value, based on the relevant facts and circumstances for each arrangement. The selling prices of deliverables under an arrangement may be derived using vendor specific objective evidence (VSOE), third-party evidence, or a best estimate of selling price (BESP), if VSOE or third-party evidence is not available. For most pharmaceutical licensing and collaboration agreements, BESP is utilized. Establishing BESP involves judgment and considers multiple factors, including market conditions and company-specific factors, including those factors contemplated in negotiating the agreements, as well as internally developed models that include assumptions related to market opportunity, discounted cash flows, estimated development costs, probability of success and the time needed to commercialize a product candidate pursuant to the agreement. In validating the BESP, the Company considers whether changes in key assumptions used to determine the BESP will have a significant effect on the allocation of the arrangement consideration between the multiple deliverables. The allocated consideration for each unit of accounting is recognized over the related obligation period in accordance with the applicable revenue recognition criteria.
If there are deliverables in an arrangement that are not separable from other aspects of the contractual relationship, they are treated as a combined unit of accounting, with the allocated revenue for the combined unit recognized in a manner consistent with the revenue recognition applicable to the final deliverable in the combined unit. Payments received prior to satisfying the relevant revenue recognition criteria are recorded as deferred revenue in the accompanying balance sheets and recognized as revenue when the related revenue recognition criteria are met.
The Company typically receives up-front payments when licensing its intellectual property, which often occurs in conjunction with an R&D agreement. The Company will recognize revenue attributed to the license upon delivery, provided that the license has stand-alone value.
Milestones are recognized when earned, as evidenced by written acknowledgment from the collaborator or other persuasive evidence that the milestone has been achieved, provided that the milestone event is substantive.
75
Table of Contents
A milestone event is generally considered to be substantive if its achievability was not reasonably assured at the inception of the agreement. The Company assesses whether a milestone is substantive at the inception of each agreement.
Share-Based Compensation. The Company estimates the fair value of stock options using the Black-Scholes option pricing model on the date of grant. Restricted stock units are valued based on the closing price of the Company’s common stock on the date of grant. The fair value of equity instruments expected to vest are recognized and amortized on a straight-line basis over the requisite service period of the award, which is generally three to four years; however, certain provisions in the Company’s equity compensation plans provide for shorter vesting periods under certain circumstances. Additionally, the Company has granted certain performance-based equity awards that vest upon the achievement of certain pre-defined Company-specific performance criteria. Expense related to these performance-based equity awards is generally recognized ratably over the performance period once the pre-defined performance based criteria for vesting becomes probable.
Investment Income, Net. Investment income, net is comprised of interest and dividends earned on cash, cash equivalents and investments as well as gains and losses realized from activity in the Company’s investment portfolio. For the years ended December 31, 2017, 2016 and 2015, the investment income was $6.2 million, $2.8 million and $1.9 million, respectively and primarily consisted of interest income.
Net Loss Per Share. The Company computes basic net loss per share using the weighted average number of common shares outstanding during the period. Diluted net income per share is based upon the weighted average number of common shares and potentially dilutive securities (common share equivalents) outstanding during the period. Common share equivalents outstanding, determined using the treasury stock method, are comprised of shares that may be issued under the Company’s stock option agreements or convertible debt. Common share equivalents are excluded from the diluted net loss per share calculation because of their anti-dilutive effect.
Due to the Company’s net loss position in 2017, 2016 and 2015, approximately 3.4 million, 3.8 million and 4.1 million, respectively, of common share equivalents related to outstanding restricted stock units and employee options were excluded from the diluted common shares outstanding. For the years ended December 31, 2017, 2016 and 2015, there were employee stock options, calculated on a weighted average basis, to purchase 0.4 million, 0.4 million, and 0.1 million shares of our common stock with an exercise price greater than the average market price of the underlying common shares.
Additionally, during May 2017 the Company issued $517.5 million of convertible debt which under certain circumstances may convert to common shares outstanding. If converted, this $517.5 million of convertible debt would represent approximately 6.8 million common shares.
Impact of Recently Issued Accounting Standards.
In May 2014 the FASB issued Accounting Standards Update (ASU) No. 2014-09, Revenue from Contracts with Customers (Topic 606), which supersedes all existing revenue recognition requirements, including most industry-specific guidance. This new standard requires a company to recognize revenue when it transfers goods or services to customers in an amount that reflects the consideration that the company expects to receive for those goods or services. The FASB subsequently issued amendments to ASU No. 2014-09 that have the same effective date and transition date. These new standards became effective for us on January 1, 2018, and will be adopted using the modified retrospective method through a cumulative-effect adjustment, if any, directly to retained earnings as of that date. The Company has performed a review of these new standards as compared to our current accounting policies for customer contracts and collaborative relationships. As of December 31, 2017, the Company has not identified any accounting changes that would materially impact the amount of reported revenues with respect to our product revenues. The Company is currently evaluating the impact of the new standard on historical revenue recorded for our two collaboration agreements. This ongoing evaluation is
76
Table of Contents
dependent upon the resolution of certain questions relating to the application of the new revenue recognition guidance for collaboration agreements which will ultimately determine the impact, if any, the adoption of this standard may have on the consolidated financial statements.
The company expects the accounting for contingent milestone payments to be the most significant change in accounting for its license and collaboration agreements. Topic 605 provides guidance specific to the accounting for milestone payments, including the ability to defer the recognition of any milestones until received and, if certain criteria are met, the ability to recognize milestone payments as revenue when received. However, Topic 606 does not contain guidance specific to milestone payments, thereby requiring potential milestone payments to be considered in accordance with the overall model of Topic 606. As a result, revenues from contingent milestone payments may be recognized earlier under Topic 606 than under Topic 605, based on an assessment of the probability of achievement of the milestone and the likelihood of a significant reversal of such milestone revenue at each reporting date. This assessment may result in recognizing milestone revenue before the milestone event has been achieved. In addition, Topic 606 changes guidance regarding the accounting for variable consideration received from licensees, which may impact the estimation of, and determination of the timing of, the related revenue recognition.
In February 2016, the FASB issued Accounting Standards Update (ASU) 2016-02 Leases. This update amends the current accounting guidance for lease transactions. Under the new guidance, a lessee will be required to recognize both assets and liabilities for any leases in excess of twelve months. Additionally, certain qualitative and quantitative disclosures will also be required in the financial statements. The Company is required to adopt this new guidance beginning in 2019 and early adoption is permitted. The Company’s lease for its corporate headquarters is subject to this new guidance and the process of determining the impact of the adoption of this update will have on its consolidated financial statements is ongoing.
In November 2016, the FASB issued ASU 2016-18, “Statement of Cash Flows (Topic 230): Restricted Cash”, which clarifies the presentation of restricted cash and restricted cash equivalents in the statements of cash flows. Under this ASU, restricted cash and restricted cash equivalents are included with cash and cash equivalents when reconciling the beginning-of-period and end-of-period total amounts presented on the statements of cash flows. This ASU is intended to reduce diversity in practice in the classification and presentation of changes in restricted cash on the statement of cash flows. This ASU requires that the statement of cash flows explain the change in total cash and equivalents and amounts generally described as restricted cash or restricted cash equivalents when reconciling the beginning-of-period and end-of-period total amounts. This ASU also requires a reconciliation between the total of cash and equivalents and restricted cash presented on the statement of cash flows and the cash and equivalents balance presented on the balance sheet. This ASU was effective for the Company on January 1, 2018. The Company does not expect the adoption of this ASU to have a material effect on its results of operations, financial condition or cash flows.
NOTE 2. SIGNIFICANT COLLABORATIVE RESEARCH AND DEVELOPMENT AGREEMENTS
Mitsubishi Tanabe Pharma Corporation (Mitsubishi Tanabe). During 2015, the Company entered into a collaboration and license agreement with Mitsubishi Tanabe for the development and commercialization of INGREZZA for movement disorders in Japan and other select Asian markets. Payments to the Company under this agreement include an up-front license fee of $30 million, up to $85 million in development and commercialization event-based payments, payments for the manufacture of pharmaceutical products, and royalties on product sales in select territories in Asia. Under the terms of the agreement, Mitsubishi Tanabe is responsible for all third-party development, marketing and commercialization costs in Japan and other select Asian markets with the exception of a single Huntington’s chorea clinical trial to be performed by the Company, at an estimated cost of approximately $12 million, should Mitsubishi Tanabe request the clinical trial. The Company will be entitled to a percentage of sales of INGREZZA in Japan and other select Asian markets for the longer of ten years or the life of the related patent rights.
77
Table of Contents
Under the terms of the Company’s agreement with Mitsubishi Tanabe, the collaboration effort between the parties to advance INGREZZA towards commercialization in Japan and other select Asian markets is governed by a joint steering committee and joint development committee with representatives from both the Company and Mitsubishi Tanabe. There are no performance, cancellation, termination or refund provisions in the agreement that would have a material financial consequence to the Company. The Company does not directly control when event-based payments will be achieved or when royalty payments will begin. Mitsubishi Tanabe may terminate the agreement at its discretion upon 180 days’ written notice to the Company. In such event, all INGREZZA product rights for Japan and other select Asian markets would revert to the Company.
The Company has identified the following deliverables associated with the Mitsubishi Tanabe agreement: INGREZZA technology license and existing know-how, development activities to be performed as part of the collaboration, and the manufacture of pharmaceutical products. The respective standalone value from each of these deliverables has been determined by applying the BESP method and the revenue was allocated based on the relative selling price method with revenue recognition timing to be determined either by delivery or the provision of services.
As discussed above, the BESP method required the use of significant estimates. The Company used an income approach to estimate the selling price for the technology license and an expense approach for estimating development activities and the manufacture of pharmaceutical products. The development activities and the manufacture of pharmaceutical products are expected to be delivered throughout the duration of the agreement. The technology license and existing know-how was delivered on the effective date of the agreement.
For the year ended December 31, 2017, the Company recognized $15 million in development event-based payments resulting from Mitsubishi Tanabe’s initiation of Phase II/III development of INGREZZA in TD in Asia. No revenue was recognized under the Mitsubishi Tanabe agreement for the year ended December 31, 2016. For the year ended December 31, 2015, the Company recognized revenue under this agreement of $19.8 million associated with the delivery of a technology license and existing know-how. In accordance with our continuing performance obligations, $10.2 million of the $30 million up-front payment is being deferred and recognized in future periods. Under the terms of the agreement, there is no general obligation to return the up-front payment for any non-contingent deliverable.
AbbVie Inc. (AbbVie). In June 2010, the Company announced an exclusive worldwide collaboration with AbbVie, to develop and commercialize elagolix and all next-generation GnRH antagonists (collectively, GnRH Compounds) for women’s and men’s health. AbbVie made an upfront payment of $75 million and has agreed to make additional development and regulatory event-based payments of up to $480 million, of which $75 million has been earned to date, and up to an additional $50 million in commercial event-based payments. The Company has assessed event-based payments under the revised authoritative guidance for research and development milestones and determined that event-based payments prior to commencement of a Phase III clinical study, as defined in the agreement, meet the definition of a milestone in accordance with authoritative guidance as (1) they are events that can only be achieved in part on the Company’s past performance, (2) there is substantive uncertainty at the date the arrangement was entered into that the event will be achieved and (3) they result in additional payments being due to the Company. Development and regulatory event-based payments subsequent to the commencement of a Phase III clinical study, however, currently do not meet these criteria as their achievement is based on the performance of AbbVie. As of December 31, 2017, approximately $455 million remains outstanding in future event-based payments under the agreement as the performance is based solely on AbbVie. However, none of the remaining event-based payments meet the definition of a milestone in accordance with authoritative accounting guidance.
Under the terms of the agreement, AbbVie is responsible for all third-party development, marketing and commercialization costs. The Company will be entitled to a percentage of worldwide sales of GnRH Compounds for the longer of ten years or the life of the related patent rights. AbbVie may terminate the collaboration at its discretion upon 180 days’ written notice to the Company. In such event, the Company would be entitled to
78
Table of Contents
specified payments for ongoing clinical development and related activities and all GnRH Compound product rights would revert to the Company. During 2017, event-based revenue of $30.0 million was recognized based on AbbVie’s NDA submission for elagolix in endometriosis being accepted as filed by the FDA. During 2016, event-based revenue of $15.0 million was recognized related to AbbVie’s initiation of Phase III development of elagolix in uterine fibroids. No revenue was recognized during 2015 under this collaboration agreement.
NOTE 3. INVESTMENTS
Available-for-sale securities are carried at fair value, with the unrealized gains and losses reported in comprehensive loss. The amortized cost of debt securities in this category is adjusted for amortization of premiums and accretion of discounts to maturity. Such amortization and accretion is included in investment income. Realized gains and losses and declines in value judged to be other-than-temporary, if any, on available-for-sale securities are included in other income or expense. The cost of securities sold is based on the specific identification method. Interest and dividends on securities classified as available-for-sale are included in investment income.
Investments at December 31, 2017 and 2016 consisted of the following (in thousands):
| Years
Ended December 31, |
||||||||
| 2017 | 2016 | |||||||
| Certificates of deposit |
$ | — | $ | 960 | ||||
| Commercial paper |
75,362 | 49,245 | ||||||
| Corporate debt securities |
414,815 | 204,436 | ||||||
| Securities of government-sponsored entities |
18,401 | 12,932 | ||||||
|
|
|
|
|
|||||
| Total investments |
$ | 508,578 | $ | 267,573 | ||||
|
|
|
|
|
|||||
The following is a summary of investments classified as available-for-sale securities (in thousands):
| Contractual Maturity (in years) |
Amortized Cost |
Gross Unrealized Gains(1) |
Gross Unrealized Losses(1) |
Aggregate Estimated Fair Value |
||||||||||||||||
| December 31, 2016: |
||||||||||||||||||||
| Classified as current assets: |
||||||||||||||||||||
| Certificates of deposit |
Less than 1 | $ | 960 | $ | — | $ | — | $ | 960 | |||||||||||
| Commercial paper |
Less than 1 | 49,280 | 3 | (38 | ) | 49,245 | ||||||||||||||
| Corporate debt securities |
Less than 1 | 168,548 | 3 | (117 | ) | 168,434 | ||||||||||||||
| Securities of government-sponsored entities |
Less than 1 | 5,448 | — | (4 | ) | 5,444 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total short-term available-for-sale securities |
$ | 224,236 | $ | 6 | $ | (159 | ) | $ | 224,083 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Classified as non-current assets: |
||||||||||||||||||||
| Corporate debt securities |
1 to 2 | $ | 36,149 | $ | — | $ | (147 | ) | $ | 36,002 | ||||||||||
| Securities of government-sponsored entities |
1 to 2 | 7,506 | — | (18 | ) | 7,488 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total long-term available-for-sale securities |
$ | 43,655 | $ | — | $ | (165 | ) | $ | 43,490 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
79
Table of Contents
| Contractual Maturity (in years) |
Amortized Cost |
Gross Unrealized Gains(1) |
Gross Unrealized Losses(1) |
Aggregate Estimated Fair Value |
||||||||||||||||
| December 31, 2017: |
||||||||||||||||||||
| Classified as current assets: |
||||||||||||||||||||
| Commercial paper |
Less than 1 | $ | 75,396 | $ | 1 | $ | (35 | ) | $ | 75,362 | ||||||||||
| Corporate debt securities |
Less than 1 | 178,776 | — | (400 | ) | 178,376 | ||||||||||||||
| Securities of government-sponsored entities |
Less than 1 | 7,503 | — | (24 | ) | 7,479 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total short-term available-for-sale securities |
$ | 261,675 | $ | 1 | $ | (459 | ) | $ | 261,217 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Classified as non-current assets: |
||||||||||||||||||||
| Corporate debt securities |
1 to 2 | $ | 237,749 | $ | — | $ | (1,310 | ) | $ | 236,439 | ||||||||||
| Securities of government-sponsored entities |
1 to 2 | 11,004 | — | (82 | ) | 10,922 | ||||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total long-term available-for-sale securities |
$ | 248,753 | $ | — | $ | (1,392 | ) | $ | 247,361 | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||||||
| (1) | Unrealized gains and losses are included in other comprehensive loss. |
The following table presents gross unrealized losses and fair value for those available-for-sale investments that were in an unrealized loss position as of December 31, 2017 and 2016, aggregated by investment category and length of time that individual securities have been in a continuous loss position (in thousands):
| Less Than 12 Months | 12 Months or Greater | Total | ||||||||||||||||||||||
| Estimated Fair Value |
Unrealized Losses |
Estimated Fair Value |
Unrealized Losses |
Estimated Fair Value |
Unrealized Losses |
|||||||||||||||||||
| December 31, 2016: |
||||||||||||||||||||||||
| Commercial paper |
$ | 43,781 | $ | (38 | ) | $ | — | $ | — | $ | 43,781 | $ | (38 | ) | ||||||||||
| Corporate debt securities |
185,243 | (261 | ) | 9,144 | (3 | ) | 194,387 | (264 | ) | |||||||||||||||
| Securities of government-sponsored entities |
12,932 | (22 | ) | — | — | 12,932 | (22 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total |
$ | 241,956 | $ | (321 | ) | $ | 9,144 | $ | (3 | ) | $ | 251,100 | $ | (324 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| December 31, 2017: |
||||||||||||||||||||||||
| Commercial paper |
$ | 62,602 | $ | (35 | ) | $ | — | $ | — | $ | 62,602 | $ | (35 | ) | ||||||||||
| Corporate debt securities |
386,728 | (1,660 | ) | 28,087 | (50 | ) | 414,815 | (1,710 | ) | |||||||||||||||
| Securities of government-sponsored entities |
10,922 | (82 | ) | 7,479 | (24 | ) | 18,401 | (106 | ) | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Total |
$ | 460,252 | $ | (1,777 | ) | $ | 35,566 | $ | (74 | ) | $ | 495,818 | $ | (1,851 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
NOTE 4. FAIR VALUE MEASUREMENTS
Fair value is an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. As a basis for considering such assumptions, a three-tier fair value hierarchy has been established, which prioritizes the inputs used in measuring fair value as follows:
| Level 1: |
Observable inputs such as quoted prices in active markets; | |
| Level 2: |
Inputs include quoted prices for similar instruments in active markets and/or quoted prices for identical or similar instruments in markets that are not active near the measurement date; and | |
| Level 3: |
Unobservable inputs in which there is little or no market data, which require the reporting entity to develop its own assumptions. | |
80
Table of Contents
The Company classifies its cash equivalents and available for sale investments within Level 1 or Level 2. The fair value of the Company’s high quality investment grade corporate debt securities is determined using proprietary valuation models and analytical tools. These valuation models and analytical tools use market pricing or prices for similar instruments that are both objective and publicly available, including matrix pricing or reported trades, benchmark yields, broker/dealer quotes, issuer spreads, two-sided markets, benchmark securities, bids and/or offers. The Company did not reclassify any investments between levels in the fair value hierarchy during the years ended December 31, 2017 and 2016.
The Company’s assets which are measured at fair value on a recurring basis as of December 31, 2017 and 2016 were determined using the inputs described above (in millions):
| Fair Value Measurements Using | ||||||||||||||||
| Carrying Value |
Quoted Prices in Active Markets for Identical Assets (Level 1) |
Significant
Other Observable Inputs (Level 2) |
Significant Unobservable Inputs (Level 3) |
|||||||||||||
| December 31, 2016: |
||||||||||||||||
| Classified as current assets: |
||||||||||||||||
| Cash and money market funds |
$ | 73.6 | $ | 73.6 | $ | — | $ | — | ||||||||
| Certificates of deposit |
1.0 | 1.0 | — | — | ||||||||||||
| Commercial paper |
49.2 | — | 49.2 | — | ||||||||||||
| Securities of government-sponsored entities |
5.4 | — | 5.4 | — | ||||||||||||
| Corporate debt securities |
178.1 | — | 178.1 | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Subtotal |
307.3 | 74.6 | 232.7 | — | ||||||||||||
| Classified as long-term assets: |
||||||||||||||||
| Certificates of deposit |
4.9 | 4.9 | — | — | ||||||||||||
| Securities of government-sponsored entities |
7.5 | — | 7.5 | — | ||||||||||||
| Corporate debt securities |
36.0 | — | 36.0 | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
355.7 | 79.5 | 276.2 | — | ||||||||||||
| Less cash, cash equivalents and restricted cash |
(88.1 | ) | (78.5 | ) | (9.6 | ) | — | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total investments |
$ | 267.6 | $ | 1.0 | $ | 266.6 | $ | — | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| December 31, 2017: |
||||||||||||||||
| Classified as current assets: |
||||||||||||||||
| Cash and money market funds |
$ | 170.2 | $ | 170.2 | $ | — | $ | — | ||||||||
| Commercial paper |
159.9 | — | 159.9 | — | ||||||||||||
| Securities of government-sponsored entities |
7.5 | — | 7.5 | — | ||||||||||||
| Corporate debt securities |
178.4 | — | 178.4 | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Subtotal |
516.0 | 170.2 | 345.8 | — | ||||||||||||
| Classified as long-term assets: |
||||||||||||||||
| Cash and money market funds |
1.5 | 1.5 | ||||||||||||||
| Certificates of deposit |
3.0 | 3.0 | — | — | ||||||||||||
| Securities of government-sponsored entities |
10.9 | — | 10.9 | — | ||||||||||||
| Corporate debt securities |
236.4 | — | 236.4 | — | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
767.8 | 174.7 | 593.1 | — | ||||||||||||
| Less cash, cash equivalents and restricted cash |
(259.2 | ) | (174.6 | ) | (84.6 | ) | — | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total investments |
$ | 508.6 | $ | 0.1 | $ | 508.5 | $ | — | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
81
Table of Contents
The fair value of the Company’s outstanding 2.25% convertible senior notes due 2024 (the 2024 Notes) at December 31, 2017, calculated utilizing market quotations from an over-the-counter trading market for these notes (Level 2), was approximately $662.1 million. The carrying value of the 2024 Notes is discussed in Note 8 to the Consolidated Financial Statements included in this Annual Report .
NOTE 5. PROPERTY AND EQUIPMENT
Property and equipment, net, at December 31, 2017 and 2016 consisted of the following (in thousands):
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Tenant improvements |
2,019 | 1,530 | ||||||
| Furniture and fixtures |
1,303 | 942 | ||||||
| Equipment |
35,069 | 29,749 | ||||||
|
|
|
|
|
|||||
| 38,391 | 32,221 | |||||||
| Less accumulated depreciation |
(27,580 | ) | (25,950 | ) | ||||
|
|
|
|
|
|||||
| Property and equipment, net |
$ | 10,811 | $ | 6,271 | ||||
|
|
|
|
|
|||||
For each of the years ended December 31, 2017, 2016 and 2015, depreciation expense was $2.4 million, $1.5 million and $1.0 million, respectively. During 2017, 2016 and 2015, the Company recognized a gain of approximately $7,000, $8,000 and $9,000, respectively, related to disposal of capital equipment.
NOTE 6. ACCOUNTS PAYABLE AND ACCRUED LIABILITIES
Accounts payable and accrued liabilities at December 31, 2017 and 2016 consisted of the following (in thousands):
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Accrued employee related costs |
$ | 24,901 | $ | 9,559 | ||||
| Accounts payable |
5,648 | 5,085 | ||||||
| Accrued development costs |
4,799 | 5,543 | ||||||
| Other accrued liabilities |
18,172 | 5,995 | ||||||
|
|
|
|
|
|||||
| $ | 53,520 | $ | 26,182 | |||||
|
|
|
|
|
|||||
NOTE 7. COMMITMENTS AND CONTINGENCIES
In December 2007, the Company closed the sale of its facility and associated real property for a purchase price of $109 million. Concurrent with the sale, the Company retired the entire $47.7 million in mortgage debt previously outstanding with respect to the facility and associated real property, and received cash of $61.0 million net of transaction costs and debt retirement.
Upon the closing of the sale of the facility and associated real property, the Company entered into a lease agreement (Lease) whereby it leased back for an initial term of 12 years its corporate headquarters comprised of two buildings located at 12790 El Camino Real (Front Building) and 12780 El Camino Real (Rear Building) in San Diego, California. The Company also entered into a series of lease amendments (Amendments), beginning in late 2008 through 2011, through which it was able to vacate the Front Building, but continue to occupy the Rear Building. The ultimate result of this real estate sale was a net gain of $39.1 million which was deferred in accordance with authoritative guidance. The Company recognized $2.1 million, $3.4 million and $3.3 million of the deferred gain during the years ended December 31, 2017, 2016 and 2015, respectively. The Company will recognize the remaining $8.8 million of the deferred gain on a straight-line basis over the amended Lease term which will expire at the end of 2029.
82
Table of Contents
During 2017, the Company entered into an amendment to extend the current term of the Lease through December 31, 2029 (Term Amendment). Under the Term Amendment, the Company reduced its base rental rate by approximately 8% and will continue to pay base annual rent (subject to an annual fixed percentage increase), plus a 3.5% annual management fee, property taxes and other normal and necessary expenses associated with the Lease such as utilities, repairs and maintenance. Certain incentives were included in the Term Amendment, including approximately $13.1 million in various tenant improvement allowances, three months of rent abatement, and a reduction in the required security deposit amount from $4.7 million to $3.0 million. In lieu of a cash security deposit, Wells Fargo Bank, N.A. issued on the Company’s behalf a letter of credit in the amount of $3.0 million, which is secured by a deposit of equal amount with the same bank. The Company also has the right to extend the Lease for two consecutive ten-year terms as well as a right of first offer for future rental of adjacent office space owned by the landlord.
As of December 31, 2017, the Company had one sublease agreement for approximately 16,000 square feet of the Rear Building. This sublease is expected to result in approximately $0.1 million of rental income in 2018 with this sublease rental income being recorded as an offset to rent expense. The income generated under this sublease is lower than the Company’s financial obligation under the Lease for the Rear Building, as determined on a per square foot basis. Consequently, at the inception of a sublease, or in association with an amendment to a sublease, the Company is required to record a cease-use liability for the net present value of the estimated difference between the expected income to be generated under the sublease and future subleases and the Lease obligation over the remaining term of the Lease for the space that is occupied by the subtenant. As a result of the recent FDA approval for INGREZZA and the Term Amendment, the Company has determined that it will reoccupy the space at the conclusion of the sublease in March 2018. Additionally, during the first quarter of 2016, the Company terminated another previously existing sublease and began to reoccupy the related space to allow for expansion. These two sublease terminations resulted in reversal of cease use expense of approximately $0.5 million and $0.8 million during 2017 and 2016, respectively.
The following table sets forth changes to the accrued cease-use liability during 2017 and 2016 (in thousands):
| Years
Ended December 31, |
||||||||
| 2017 | 2016 | |||||||
| Beginning balance |
$ | 853 | $ | 1,983 | ||||
| Change in estimate |
(544 | ) | (830 | ) | ||||
| Payments |
(247 | ) | (300 | ) | ||||
|
|
|
|
|
|||||
| Ending balance |
$ | 62 | $ | 853 | ||||
|
|
|
|
|
|||||
Rent Expense. Gross rent expense was approximately $5.9 million for the year ended December 31, 2017, and $6.0 million for each of the years ended December 31, 2016 and 2015, respectively. For financial reporting purposes, the Company recognizes rent expense on a straight-line basis over the term of the lease. Accordingly, rent expense recognized in excess of rent paid is reflected as a liability in the accompanying consolidated balance sheets.
Lease Commitments. The Company leases its office and research laboratories under an operating lease that expires at the end of 2029.
83
Table of Contents
As of December 31, 2017, the total estimated future annual minimum lease payments under the Company’s non-cancelable building lease for the years ending after December 31, 2017 were as follows (in thousands):
| Payment Amount | ||||
| 2018 |
$ | 7,368 | ||
| 2019 |
7,589 | |||
| 2020 |
7,844 | |||
| 2021 |
8,079 | |||
| 2022 and thereafter |
74,000 | |||
|
|
|
|||
| Total future minimum lease payments |
$ | 104,880 | ||
|
|
|
|||
Product Liability. The Company’s business exposes it to liability risks from its potential drug products. A successful product liability claim or series of claims brought against the Company could result in payment of significant amounts of money and divert management’s attention from running the business. The Company may not be able to maintain insurance on acceptable terms, or the insurance may not provide adequate protection in the case of a product liability claim. To the extent that product liability insurance, if available, does not cover potential claims, the Company would be required to self-insure the risks associated with such claims. The Company believes that it carries reasonably adequate insurance for product liability claims.
Licensing and Research Agreements. The Company has entered into in-licensing agreements with various universities and research organizations, which are generally cancelable at the option of the Company with terms ranging from 0-180 days written notice. Under the terms of these agreements, the Company has received licenses to research tools, know-how and technology claimed in certain patents or patent applications. The Company is required to pay fees, milestones and/or royalties on future sales of products employing the technology or falling under claims of a patent, and some of the agreements require minimum royalty payments. Some of the agreements also require the Company to pay expenses arising from the prosecution and maintenance of the patents covering the in-licensed technology. The Company continually reassesses the value of the license agreements and cancels them when research efforts are discontinued on these programs. As of December 31, 2017, all in-licensed and research candidates are successfully developed, the Company may be required to pay milestone payments of approximately $132 million over the lives of these agreements, in addition to royalties on sales of the affected products at rates ranging up to 5%. Due to the uncertainties of the development process, the timing and probability of the milestone and royalty payments cannot be accurately estimated.
Litigation. From time to time, the Company may be subject to legal proceedings and claims in the ordinary course of business. On December 1, 2015, Icahn School of Medicine at Mount Sinai (Mount Sinai) filed a complaint against the Company in the United States District Court for the Southern District of New York: Icahn School of Medicine at Mount Sinai v. Neurocrine Biosciences, Inc., Case No. 1:15-cv-09414. In the complaint, Mount Sinai alleges that the Company, by entering into an exclusive worldwide collaboration with AbbVie to develop and commercialize next-generation GnRH antagonists, breached a license agreement with Mount Sinai dated August 27, 1999 (Mount Sinai License). Mount Sinai is seeking unspecified monetary damages, future sublicensing fees and attorney’s fees. In January 2016, the Company filed a motion to dismiss this complaint in its entirety. In June 2016, the Court denied the motion in part and granted the motion in part, ruling that while Mount Sinai could continue its lawsuit against the Company, there was no requirement by the Company to obtain Mount Sinai’s consent prior to licensing the next-generation GnRH antagonists to AbbVie. In July 2016, the Company filed its answer denying Mount Sinai’s allegations, and filed counterclaims against Mount Sinai. Mount Sinai filed a motion to dismiss the Company’s counterclaims and affirmative defenses, which the Court granted. A trial is anticipated to occur in the first half of 2018.The Company believes that it has meritorious defenses to the claims made in the complaint and intends to vigorously defend against such claims, but is not able to predict the ultimate outcome of this action, or estimate any potential loss.
The Company is not aware of any other proceedings or claims that it believes will have, individually or in the aggregate, a material adverse effect on its business, financial condition or results of operations.
84
Table of Contents
NOTE 8. CONVERTIBLE SENIOR NOTES
On May 2, 2017, the Company completed a private placement of $517.5 million in aggregate principal amount of 2.25% convertible senior notes due 2024 (2024 Notes) and entered into an indenture agreement (2024 Indenture) with respect to the 2024 Notes. The 2024 Notes accrue interest at a fixed rate of 2.25% per year, payable semiannually in arrears on May 15 and November 15 of each year, beginning on November 15, 2017. The 2024 Notes mature on May 15, 2024. The net proceeds from the issuance of the 2024 Notes were approximately $502.8 million, after deducting commissions and the offering expenses payable by the Company.
Holders of the 2024 Notes may convert the 2024 Notes at any time prior to the close of business on the business day immediately preceding May 15, 2024, only under the following circumstances:
(i) during any calendar quarter commencing after the calendar quarter ending on September 30, 2017 (and only during such calendar quarter), if the last reported sale price of the Company’s common stock for at least 20 trading days (whether or not consecutive) during a period of 30 consecutive trading days ending on the last trading day of the immediately preceding calendar quarter is greater than 130% of the conversion price on each applicable trading day;
(ii) during the five business-day period immediately after any five consecutive trading-day period (the ‘‘measurement period’’) in which the trading price (as defined in the 2024 Indenture) per $1,000 principal amount of the 2024 Notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price of the Company’s common stock and the conversion rate on each such trading day;
(iii) upon the occurrence of specified corporate events, including a merger or a sale of all or substantially all of the Company’s assets; or
(iv) if the Company calls the 2024 Notes for redemption, until the close of business on the business day immediately preceding the redemption date.
On or after January 15, 2024, until the close of business on the scheduled trading day immediately preceding May 15, 2024, holders may convert their 2024 Notes at any time.
Upon conversion, holders will receive the principal amount of their 2024 Notes and any excess conversion value, calculated based on the per share volume-weighted average price for each of the 30 consecutive trading days during the observation period (as more fully described in the 2024 Indenture). For both the principal and excess conversion value, holders may receive cash, shares of the Company’s common stock or a combination of cash and shares of the Company’s common stock, at the Company’s option.
It is the Company’s intent and policy to settle conversions through combination settlement, which essentially involves repayment of an amount of cash equal to the “principal portion” and delivery of the “share amount” in excess of the principal portion in shares of common stock or cash. In general, for each $1,000 in principal, the “principal portion” of cash upon settlement is defined as the lesser of $1,000, and the conversion value during the 25-day observation period as described in the indenture for the notes. The conversion value is the sum of the daily conversion value which is the product of the effective conversion rate divided by 25 days and the daily volume weighted average price (VWAP) of the Company’s common stock. The “share amount” is the cumulative “daily share amount” during the observation period, which is calculated by dividing the daily VWAP into the difference between the daily conversion value (i.e., conversion rate x daily VWAP) and $1,000.
The initial conversion rate for the 2024 Notes is 13.1711 shares of common stock per $1,000 principal amount, which is equivalent to an initial conversion price of approximately $75.92 per share of the Company’s common stock. The conversion rate will be subject to adjustment in some events but will not be adjusted for any accrued and unpaid interest. The initial conversion price of the 2024 Notes represents a premium of approximately 42.5% to the closing sale price of $53.28 per share of the Company’s common stock on the NASDAQ Global Select Market on April 26, 2017, the date that the Company priced the private offering of the 2024 Notes.
85
Table of Contents
In the event of conversion, holders would forgo all future interest payments, any unpaid accrued interest and the possibility of further stock price appreciation. Upon the receipt of conversion requests, the settlement of the 2024 Notes will be paid pursuant to the terms of the 2024 Indenture. In the event that all of the 2024 Notes are converted, the Company would be required to repay the $517.5 million in principal value and any conversion premium in any combination of cash and shares of its common stock (at the Company’s option).
Prior to May 15, 2021, the Company may not redeem the 2024 Notes. On or after May 15, 2021, the Company may redeem for cash all or part of the 2024 Notes if the last reported sale price (as defined in the 2024 Indenture) of the Company’s common stock has been at least 130% of the conversion price then in effect for at least 20 trading days (whether or not consecutive) during any 30 consecutive trading-day period ending on, and including, the trading day immediately before the date which the Company provides notice of redemption. The redemption price will equal the sum of (i) 100% of the principal amount of the 2024 Notes being redeemed, plus (ii) accrued and unpaid interest, including additional interest, if any, to, but excluding, the redemption date. No sinking fund is provided for the 2024 Notes.
If the Company undergoes a fundamental change, as defined in the 2024 Indenture, subject to certain conditions, holders of the 2024 Notes may require the Company to repurchase for cash all or part of their 2024 Notes at a repurchase price equal to 100% of the principal amount of the 2024 Notes to be repurchased, plus accrued and unpaid interest to, but excluding, the fundamental change repurchase date. In addition, if a ‘‘make-whole fundamental change’’ (as defined in the 2024 Indenture) occurs prior to January 15, 2024, the Company will, in certain circumstances, increase the conversion rate for a holder who elects to convert its notes in connection with the make-whole fundamental change.
The 2024 Notes are the Company’s general unsecured obligations that rank senior in right of payment to all of its indebtedness that is expressly subordinated in right of payment to the 2024 Notes, and equal in right of payment to the Company’s unsecured indebtedness.
While the 2024 Notes are currently classified on the Company’s consolidated balance sheet at December 31, 2017 as long-term, the future convertibility and resulting balance sheet classification of this liability will be monitored at each quarterly reporting date and will be analyzed dependent upon market prices of the Company’s common stock during the prescribed measurement periods. In the event that the holders of the 2024 Notes have the election to convert the 2024 Notes at any time during the prescribed measurement period, the 2024 Notes would then be considered a current obligation and classified as such.
Under current accounting guidance, an entity must separately account for the liability and equity components of convertible debt instruments (such as the 2024 Notes) that may be settled entirely or partially in cash upon conversion in a manner that reflects the issuer’s economic interest cost. The liability component of the instrument was valued in a manner that reflects the market interest rate for a similar nonconvertible instrument at the date of issuance. The initial carrying value of the liability component of $368.3 million was calculated using a 7.5% assumed borrowing rate. The equity component of $149.2 million, representing the conversion option, was determined by deducting the fair value of the liability component from the par value of the 2024 Notes and is recorded in additional paid-in capital on the consolidated balance sheet at the issuance date. That equity component is treated as a discount on the liability component of the 2024 Notes, which is amortized over the seven year term of the 2024 Notes using the effective interest rate method. The equity component is not re-measured as long as it continues to meet the conditions for equity classification.
The Company allocated the total transaction costs of approximately $14.7 million related to the issuance of the 2024 Notes to the liability and equity components of the 2024 Notes based on their relative values. Transaction costs attributable to the liability component are amortized to interest expense over the seven-year term of the 2024 Notes, and transaction costs attributable to the equity component are netted with the equity component in stockholders’ equity.
86
Table of Contents
The 2024 Notes do not contain any financial or operating covenants or any restrictions on the payment of dividends, the issuance of other indebtedness or the issuance or repurchase of securities by the Company. The 2024 Indenture contains customary events of default with respect to the 2024 Notes, including that upon certain events of default, 100% of the principal and accrued and unpaid interest on the 2024 Notes will automatically become due and payable.
Debt, net of discounts and deferred financing costs at December 31, 2017, consisted of the following:
| Principal |
$ | 517,500 | ||
| Deferred financing costs |
(9,652 | ) | ||
| Debt discount, net |
(138,230 | ) | ||
|
|
|
|||
| Net carrying amount |
$ | 369,618 | ||
|
|
|
NOTE 9. SHARE-BASED COMPENSATION
Share-Based Compensation Plans. In May 2011, the Company adopted the Neurocrine Biosciences, Inc. 2011 Equity Incentive Plan (the 2011 Plan) pursuant to which 17.0 million shares of Company common stock are authorized for issuance. The 2011 Plan provides for the grant of stock options that qualify as incentive stock options under Section 422 of the Internal Revenue Code of 1986, as amended (the Code), nonstatutory stock options, restricted stock awards, restricted stock unit awards (RSUs), stock appreciation rights, performance stock awards, performance-based restricted stock units (PRSUs) and other forms of equity compensation.
The Company also issues stock options under the Neurocrine Biosciences, Inc. Inducement Plan (Inducement Plan) to certain employees. During 2017 and 2015, 410,000 and 120,000 stock options, respectively, and during 2017 and 2015, 12,500 and 50,000 RSUs, respectively, were granted pursuant to the Inducement Plan. The Company did not grant any options or RSUs pursuant to the Inducement Plan during 2016. These stock option grants have a four year vesting period and the RSUs have a three year cliff vesting period. The Company currently has approximately 0.4 million in stock options and RSUs outstanding under this Inducement Plan.
As of December 31, 2017, approximately 6.3 million shares of common stock remained available for the future grant of awards under the 2011 Plan. Only share awards made under the 2011 Plan that are subsequently cancelled due to forfeiture or expiration are returned to the share pool available for future grants.
The Company issues new shares upon the exercise of stock options, the issuance of stock bonus awards and vesting of RSUs and PRSUs, and has 14.1 million shares of common stock reserved for such issuance as of December 31, 2017.
Vesting Provisions of Share-Based Compensation. Stock options generally have terms from seven to ten years from the date of grant, and generally vest over a three to four-year period. The maximum contractual term for all options granted from the 2011 Plan is ten years. RSUs granted under the 2011 Plan generally have vesting periods of four years. PRSUs granted under the 2011 Plan vest based on the achievement of certain pre-defined Company-specific performance criteria and expire four to five years from the grant date.
Share-Based Compensation. The compensation cost that has been included in the statement of comprehensive loss for all share-based compensation arrangements is as follows (in thousands):
| Years Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Sales, general and administrative expense |
$ | 27,951 | $ | 16,770 | $ | 15,281 | ||||||
| Research and development expense |
14,571 | 11,694 | 13,111 | |||||||||
|
|
|
|
|
|
|
|||||||
| Share-based compensation expense |
$ | 42,522 | $ | 28,464 | $ | 28,392 | ||||||
|
|
|
|
|
|
|
|||||||
87
Table of Contents
Authoritative guidance requires that cash flows resulting from tax deductions in excess of the cumulative compensation cost recognized for options exercised be classified as cash inflows provided by financing activities and cash outflows used in operating activities. Due to the Company’s net tax loss position, no tax benefits have been recognized in the consolidated statements of cash flows.
Stock Options. The exercise price of all options granted during the years ended December 31, 2017, 2016 and 2015 was equal to the closing price of the Company’s common stock on the date of grant. The estimated fair value of each option award granted was determined on the date of grant using the Black-Scholes option-pricing valuation model with the following weighted-average assumptions for option grants during the three years ended December 31, 2017:
| Years Ended December 31, | ||||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Risk-free interest rate |
2.0 | % | 1.4 | % | 1.7 | % | ||||||
| Expected volatility of common stock |
58 | % | 60 | % | 66 | % | ||||||
| Dividend yield |
0.0 | % | 0.0 | % | 0.0 | % | ||||||
| Expected option term |
5.7 years | 5.6 years | 6.6 years | |||||||||
The Company estimates the fair value of stock options using a Black-Scholes option-pricing model on the date of grant. The fair value of equity instruments are recognized and amortized on a straight-line basis over the requisite service period. The Black-Scholes option-pricing model incorporates various and highly sensitive assumptions including expected volatility, expected term and interest rates. The expected volatility is based on the historical volatility of the Company’s common stock over the most recent period commensurate with the estimated expected term of the Company’s stock options. The expected option term is estimated based on historical experience as well as the status of the employee. For example, directors and officers have a longer expected option term than all other employees. The risk-free rate for periods within the contractual life of the option is based upon observed interest rates appropriate for the expected term of the Company’s employee stock options. The Company has never declared or paid dividends and has no plans to do so in the foreseeable future.
The Company’s determination of fair value is affected by the Company’s stock price as well as a number of assumptions that require judgment. The weighted-average fair values of options granted during the years ended December 31, 2017, 2016 and 2015, estimated as of the grant date using the Black-Scholes option valuation model, were $25.11, $21.49 and $23.24, respectively.
A summary of the status of the Company’s stock options as of December 31, 2017, 2016 and 2015 and of changes in options outstanding under the plans during the three years ended December 31, 2017 is as follows (in thousands, except for weighted average exercise price data):
| 2017 | 2016 | 2015 | ||||||||||||||||||||||
| Options | Weighted Average Exercise Price |
Options | Weighted Average Exercise Price |
Options | Weighted Average Exercise Price |
|||||||||||||||||||
| Outstanding at January 1 |
6,112 | $ | 20.01 | 5,507 | $ | 15.63 | 5,750 | $ | 9.31 | |||||||||||||||
| Granted |
1,807 | 46.55 | 1,077 | 40.19 | 1,159 | 37.21 | ||||||||||||||||||
| Exercised |
(1,353 | ) | 10.41 | (341 | ) | 7.60 | (1,315 | ) | 5.01 | |||||||||||||||
| Canceled |
(210 | ) | 43.05 | (131 | ) | 34.35 | (87 | ) | 46.08 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Outstanding at December 31 |
6,356 | $ | 28.83 | 6,112 | $ | 20.01 | 5,507 | $ | 15.63 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
Options outstanding at December 31, 2017 have a weighted average remaining contractual term of 6.7 years.
For the year ended December 31, 2017, 2016 and 2015 share-based compensation expense related to stock options was $28.2 million, $18.4 million and $13.6 million, respectively. As of December 31, 2017, there was
88
Table of Contents
approximately $44.0 million of unamortized compensation cost related to stock options, which is expected to be recognized over a weighted average remaining vesting period of approximately 2.7 years. As of December 31, 2017, there were approximately 4.2 million options exercisable with a weighted average exercise price of $21.22 and a weighted-average remaining contractual term of 5.7 years. The total intrinsic value, which is the amount by which the exercise price was exceeded by the sale price of the Company’s common stock on the date of sale, of stock option exercises during the years ended December 31, 2017, 2016, and 2015 was $61.4 million, $13.2 million and $43.6 million, respectively. As of December 31, 2017, the total intrinsic value of options outstanding and exercisable was $309.9 million and $234.9 million, respectively. Cash received from stock option exercises for the years ended December 31, 2017, 2016 and 2015 was $13.9 million, $2.4 million and $6.3 million, respectively.
Restricted Stock Units. The fair value of RSUs is based on the closing sale price of the Company’s common stock on the date of issuance. For the year ended December 31, 2017, 2016 and 2015, share-based compensation expense related to RSUs was $13.9 million, $8.3 million, and $6.0 million, respectively. As of December 31, 2017, there was approximately $30.0 million of unamortized compensation cost related to RSUs, which is expected to be recognized over a weighted average remaining vesting period of approximately 2.7 years.
The total intrinsic value of RSUs converted into common shares during the years ended December 31, 2017, 2016 and 2015 was $14.9 million, $12.2 million, and $5.7 million, respectively. The RSUs, at the election of eligible employees, may be subject to deferred delivery arrangement. The total intrinsic value of RSUs outstanding at December 31, 2017 was $83.8 million based on the Company’s closing stock price on that date.
A summary of the status of the Company’s RSUs as of December 31, 2017, 2016 and 2015 and of changes in RSUs outstanding under the plans for the three years ended December 31, 2017 is as follows (in thousands, except for weighted average grant date fair value per unit):
| 2017 | 2016 | 2015 | ||||||||||||||||||||||
| Number of Units |
Weighted Average Grant Date Fair Value per Unit |
Number of Units |
Weighted Average Grant Date Fair Value per Unit |
Number of Units |
Weighted Average Grant Date Fair Value per Unit |
|||||||||||||||||||
| Outstanding at January 1 |
883 | $ | 29.33 | 910 | $ | 24.23 | 669 | $ | 15.01 | |||||||||||||||
| Granted |
588 | 47.21 | 326 | 36.73 | 448 | 33.62 | ||||||||||||||||||
| Cancelled |
(41 | ) | 40.62 | (69 | ) | 32.50 | (16 | ) | 20.83 | |||||||||||||||
| Converted into common shares |
(350 | ) | 24.19 | (284 | ) | 20.71 | (191 | ) | 14.24 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
| Outstanding at December 31 |
1,080 | $ | 40.30 | 883 | $ | 29.33 | 910 | $ | 24.23 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||
Performance-Based Restricted Stock Units. During the years ended December 31, 2016 and 2015, the Company granted approximately 230,000 and 50,000 PRSUs, respectively, that vest based on the achievement of certain pre-defined Company-specific performance criteria and expire approximately four to five years from the grant date. No PRSUs were granted during the year ended December 31, 2017. The fair value of PRSUs is estimated based on the closing sale price of the Company’s common stock on the date of grant. Expense recognition for PRSUs commences when attainment of the performance based criteria is determined to be probable. During 2017, 2016 and 2015, the Company recognized approximately $0.4 million, $1.8 million and $8.8 million in expense related to PRSUs. At December 31, 2017, the total unrecognized estimated compensation expense related to these PRSUs was $7.5 million. The total intrinsic value of PRSUs converted into common shares during the year ended December 31, 2017 and 2015 was $8.8 million and $14.9 million, respectively. No PRSUs were converted into common shares during 2016. The total intrinsic value of PRSUs outstanding at December 31, 2017 was $16.3 million based on the Company’s closing stock price on that date.
89
Table of Contents
NOTE 10. STOCKHOLDERS’ EQUITY
Equity Financing
In February 2015, the Company completed a public offering of common stock in which the Company sold approximately 8.0 million shares of its common stock at an offering price of $36.00 per share. The net proceeds generated from this transaction, after underwriting discounts and commissions and offering costs, were approximately $270.7 million.
NOTE 11. BIAL AGREEMENT
In February 2017, the Company entered into an exclusive license agreement with BIAL – Portela & CA, S.A. (BIAL) for the development and commercialization of opicapone for the treatment of human diseases and conditions, including Parkinson’s disease, in the United States and Canada. Under the terms of the agreement, the Company is responsible for the management and cost of all opicapone development and commercialization activities in the United States and Canada.
Under the terms of the agreement, the Company paid BIAL an upfront license fee of $30 million, which was expensed in the first quarter of 2017 as in process research and development. The Company may also be required to pay up to an additional $115 million in milestone payments associated with the regulatory approval and net sales of products containing opicapone. Prior to FDA approval of opicapone, the Company may be required to pay up to $20 million in milestones based on certain regulatory and clinical results and FDA acceptance of the Company’s NDA filing for opicapone. Upon commercialization of opicapone, the Company has agreed to determine certain annual sales forecasts. In the event that the Company fails to meet the minimum sales requirements for a particular year, the Company will be required to pay BIAL an amount corresponding to the difference between the actual net sales and the minimum sales requirements for such year, and if the Company fails to meet the minimum sales requirements for any two years, BIAL may terminate the agreement.
The agreement, unless terminated earlier, will continue on a licensed product-by-licensed product and country-by-country basis until a generic product in respect of such licensed product under the agreement is sold in a country and sales of such generic product are greater than a specified percentage of total sales of such licensed product in such country. Upon the Company’s written request prior to the estimated expiration of the term in respect of a licensed product, the parties shall negotiate a good faith continuation of BIAL’s supply of such licensed product after the term. After the term, and if BIAL is not supplying a certain licensed product, the Company shall pay BIAL a trademark royalty based on the net sales of such licensed product. Either party may terminate the agreement earlier if the other party materially breaches the agreement and does not cure the breach within a specified notice period, or upon the other party’s insolvency. BIAL may terminate the agreement if the Company fails to use commercially reasonable efforts or fails to file an NDA for a licensed product by a specified date or under certain circumstances involving a change of control of the Company. In certain circumstances where BIAL elects to terminate the agreement in connection with the Company’s change of control, BIAL shall pay the Company a termination fee. The Company may terminate the agreement at any time for any reason upon six months written notice to BIAL if prior to the first NDA approval in the United States, and upon nine months written notice to BIAL if such notice is given after the first NDA approval in the United States. If the Company’s termination request occurs prior to the first NDA approval in the United States, the Company will have to pay BIAL a termination fee except under certain conditions specified in the agreement.
NOTE 12. CONCENTRATION RISK
The Company does not currently have any of its own manufacturing facilities, and therefore it depends on an outsourced manufacturing strategy for the production of INGREZZA for commercial use and for the production of its product candidates for clinical trials. The Company has contracts in place with one third-party manufacturer that is approved for the commercial production of INGREZZA’s capsules and one third-party manufacturer that is approved for the production of INGREZZA’s active pharmaceutical ingredient. Although
90
Table of Contents
there are potential sources of supply other than the Company’s existing suppliers, any new supplier would be required to qualify under applicable regulatory requirements.
The Company has entered into distribution agreements with a limited number of SPs and SDs, and all of the Company’s product sales are to these customers. The Company’s three largest customers represented 99% of the Company’s product revenue for the year ended December 31, 2017 and substantially all of the Company’s accounts receivable balance at December 31, 2017.
NOTE 13. INCOME TAXES
At December 31, 2017, the Company had deferred tax assets of $362.7 million and deferred tax liabilities of $31.8 million. Due to uncertainties surrounding the Company’s ability to generate future taxable income, a full valuation has been established to offset the net deferred tax assets and liabilities. Additionally, the future utilization of the Company’s net operating loss and R&D credit carry forwards to offset future taxable income may be subject to an annual limitation, pursuant to Internal Revenue Code Sections 382 and 383, as a result of ownership changes that could occur in the future. The Company has determined that no ownership changes have occurred through December 31, 2016 and is currently evaluating whether any ownership changes occurred through December 31, 2017.
At December 31, 2017, the Company had Federal and state income tax net operating loss carry forwards of approximately $978.7 million and $535.3 million, respectively. The Federal tax loss carry forwards will begin to expire in 2021, unless previously utilized.
The California net operating loss carry forwards will begin to expire in 2018 and net operating loss carry forwards related to other states will begin to expire in 2027.
In addition, the Company has Federal and California R&D tax credit carry forwards of $48.2 million and $34.6 million, respectively. The Federal R&D tax credit carry forwards begin expiring in 2018 and will continue to expire unless utilized. The California R&D tax credit carryforwards carry forward indefinitely. The Company also has Federal Alternative Minimum Tax credit carryforwards of approximately $100,000.
On December 22, 2017, the Tax Cuts and Jobs Act of 2017 (the 2017 Tax Act) was enacted reducing the corporate tax rate from 35% to 21% which is effective on January 1, 2018. The carrying value of the Company’s deferred tax assets is also determined by the enacted U.S. corporate income tax rate. Consequently, any changes in the U.S. corporate income tax rate have impacted the carrying value of the Company’s deferred tax assets. Under the new corporate income tax rate of 21%, deferred income taxes decreased but there is a corresponding decrease to the valuation allowance. Therefore, the 2017 Tax Act is expected to have no impact on the Company’s 2017 earnings. In accordance with Staff Accounting Bulletin 118, as of December 31, 2017, the Company has not completed its accounting for the tax effects of the enactment of the 2017 Tax Act; however, the Company has made a reasonable estimate of the effects on its existing deferred tax balances.
91
Table of Contents
Significant components of the Company’s deferred tax assets and liabilities as of December 31, 2017 and 2016 are listed below. A valuation allowance of $330.9 million and $463.5 million at December 31, 2017 and 2016, respectively, has been recognized to offset net deferred tax assets as realization of such assets is uncertain. Amounts are shown as of December 31 as of each respective year (in thousands):
| 2017 | 2016 | |||||||
| Deferred tax assets: |
||||||||
| Net operating losses |
$ | 238,500 | $ | 309,100 | ||||
| Research and development credits |
47,500 | 38,800 | ||||||
| Capitalized research and development |
47,500 | 80,200 | ||||||
| Share-based compensation expense |
14,600 | 17,400 | ||||||
| Deferred revenue |
2,800 | 4,300 | ||||||
| Deferred gain on sales leaseback |
2,100 | 3,800 | ||||||
| Intangibles |
1,700 | 4,800 | ||||||
| Cease-use expense |
— | 300 | ||||||
| Fixed assets |
— | 400 | ||||||
| Other |
8,000 | 4,400 | ||||||
|
|
|
|
|
|||||
| Total deferred tax assets |
362,700 | 463,500 | ||||||
| Deferred tax liabilities: |
||||||||
| Convertible debt |
(31,300 | ) | — | |||||
| Fixed assets |
(500 | ) | — | |||||
|
|
|
|
|
|||||
| Total deferred tax liabilities |
(31,800 | ) | — | |||||
|
|
|
|
|
|||||
| Net of deferred tax assets and liabilities |
330,900 | 463,500 | ||||||
| Valuation allowance |
(330,900 | ) | (463,500 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax assets |
$ | — | $ | — | ||||
|
|
|
|
|
|||||
The provision for income taxes on earnings subject to income taxes differs from the statutory Federal rate at December 31, 2017, 2016 and 2015, due to the following (in thousands):
| 2017 | 2016 | 2015 | ||||||||||
| Federal income taxes at 35% |
$ | (49,889 | ) | $ | (49,383 | ) | $ | (31,126 | ) | |||
| State income tax, net of Federal benefit |
(4,013 | ) | 2 | 2 | ||||||||
| Tax effect on non-deductible expenses |
433 | (321 | ) | 172 | ||||||||
| Share-based compensation expense |
(19,589 | ) | (5,077 | ) | 201 | |||||||
| Officer compensation |
2,163 | — | — | |||||||||
| Change in tax rate |
154,415 | — | 10,773 | |||||||||
| Expired tax attributes |
2,998 | 6,708 | 5,594 | |||||||||
| Research credits |
(8,352 | ) | (6,511 | ) | (6,638 | ) | ||||||
| Change in valuation allowance |
(79,966 | ) | 53,414 | 15,029 | ||||||||
| Uncertain tax positions |
2,756 | 957 | 5,940 | |||||||||
| Other |
(956 | ) | 211 | 53 | ||||||||
|
|
|
|
|
|
|
|||||||
| $ | — | $ | — | $ | — | |||||||
|
|
|
|
|
|
|
|||||||
Under the FASB’s accounting guidance related to uncertain tax positions, among other things, the impact of an uncertain income tax position on the income tax return must be recognized at the largest amount that is more-likely-than-not to be sustained upon audit by the relevant taxing authority. An uncertain income tax position will not be recognized if it has less than a 50% likelihood of being sustained. Additionally, the guidance provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosure and transition.
92
Table of Contents
The Company’s policy is to recognize interest and/or penalties related to income tax matters in income tax expense. The Company had no accrual for interest or penalties on the Company’s consolidated balance sheets at December 31, 2017 or December 31, 2016, and has not recognized interest and/or penalties in the statement of comprehensive loss for the years ended December 31, 2017 and December 31, 2016.
The Company is subject to taxation in the United States and various state jurisdictions. The Company’s tax years for 1998 (federal) and 2002 (California) and forward are subject to examination by the United States and California tax authorities due to the carry forward of unutilized net operating losses and R&D credits.
The following table summarizes the activity related to our unrecognized tax benefits (in thousands):
| 2017 | 2016 | 2015 | ||||||||||
| Balance as of the beginning of the year |
$ | 34,112 | $ | 33,074 | $ | 23,854 | ||||||
| Increases related to prior year tax positions |
— | 260 | 6,636 | |||||||||
| Increases related to current year tax positions |
3,291 | 2,211 | 2,584 | |||||||||
| Expiration of the statute of limitations for the assessment of taxes |
— | (1,433 | ) | — | ||||||||
|
|
|
|
|
|
|
|||||||
| Balance as of the end of the year |
$ | 37,403 | $ | 34,112 | $ | 33,074 | ||||||
|
|
|
|
|
|
|
|||||||
The Company, under authoritative guidance, excluded those deferred tax assets that are not more likely than not to be sustained under the technical merits of the tax position. These unrecognized tax benefits totaled $3.3 million for current year tax positions, as reflected in the tabular rollforward above.
As of December 31, 2017, the Company had $30.7 million of unrecognized tax benefits that, if recognized and realized, would affect the effective tax rate.
In the next twelve months, the Company does not expect a significant change in their unrecognized tax benefits.
NOTE 14. RETIREMENT PLAN
The Company has a 401(k) defined contribution savings plan (401(k) Plan). The 401(k) Plan is for the benefit of all qualifying employees and permits voluntary contributions by employees up to 60% of base salary limited by the IRS-imposed maximum. Employer contributions were $1.1 million, $0.6 million and $0.4 million for the years ended December 31, 2017, 2016 and 2015, respectively.
93
Table of Contents
NOTE 15. SELECTED QUARTERLY FINANCIAL DATA (UNAUDITED)
The following is a summary of the quarterly results of the Company for the years ended December 31, 2017 and 2016 (unaudited, in thousands, except for per share data):
| Year Ended December 31, | Year
Ended December 31 |
|||||||||||||||||||
| First Quarter |
Second Quarter |
Third Quarter |
Fourth Quarter |
|||||||||||||||||
| 2016: |
||||||||||||||||||||
| Revenues |
$ | 15,000 | $ | — | $ | — | $ | — | $ | 15,000 | ||||||||||
| Operating expenses |
35,857 | 41,828 | 38,436 | 46,251 | 162,372 | |||||||||||||||
| Net loss |
(19,264 | ) | (40,280 | ) | (36,887 | ) | (44,659 | ) | (141,090 | ) | ||||||||||
| Net loss per share: |
||||||||||||||||||||
| Basic and Diluted |
$ | (0.22 | ) | $ | (0.46 | ) | $ | (0.43 | ) | $ | (0.51 | ) | $ | (1.63 | ) | |||||
| Shares used in the calculation of net loss per share: |
||||||||||||||||||||
| Basic and Diluted |
86,497 | 86,694 | 86,784 | 86,874 | 86,713 | |||||||||||||||
| 2017: |
||||||||||||||||||||
| Revenues |
$ | — | $ | 6,335 | $ | 60,774 | $ | 94,517 | $ | 161,626 | ||||||||||
| Operating expenses |
79,932 | 63,603 | 66,769 | 82,683 | 292,987 | |||||||||||||||
| Net (loss) income |
(78,326 | ) | (59,985 | ) | (11,125 | ) | 6,894 | (142,542 | ) | |||||||||||
| Net (loss) income per share: |
||||||||||||||||||||
| Basic |
$ | (0.90 | ) | $ | (0.68 | ) | $ | (0.13 | ) | $ | 0.08 | $ | (1.62 | ) | ||||||
| Diluted |
$ | (0.90 | ) | $ | (0.68 | ) | $ | (0.13 | ) | $ | 0.07 | $ | (1.62 | ) | ||||||
| Shares used in the calculation of net (loss) income per share: |
||||||||||||||||||||
| Basic |
87,283 | 88,063 | 88,325 | 88,665 | 88,089 | |||||||||||||||
| Diluted |
87,283 | 88,063 | 88,325 | 92,659 | 88,089 | |||||||||||||||
94
Table of Contents
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
Not applicable.
| ITEM 9A. | CONTROLS AND PROCEDURES |
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our Exchange Act reports is recorded, processed, summarized and reported within the timelines specified in the SEC’s rules and forms, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can only provide reasonable assurance of achieving the desired control objectives, and in reaching a reasonable level of assurance, management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
As required by SEC Rule 13a-15(b), we carried out an evaluation, under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures as of the end of the year covered by this report. Based on the foregoing, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level.
95
Table of Contents
Management’s Report on Internal Control Over Financial Reporting
Internal control over financial reporting refers to the process designed by, or under the supervision of, our Chief Executive Officer and Chief Financial Officer, and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles, and includes those policies and procedures that:
(1) Pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets;
(2) Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorization of our management and directors; and
(3) Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
Internal control over financial reporting cannot provide absolute assurance of achieving financial reporting objectives because of its inherent limitations. Internal control over financial reporting is a process that involves human diligence and compliance and is subject to lapses in judgment and breakdowns resulting from human failures. Internal control over financial reporting also can be circumvented by collusion or improper management override. Because of such limitations, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk. Management is responsible for establishing and maintaining adequate internal control over financial reporting for the company.
Management has used the framework set forth in the report entitled Internal Control-Integrated Framework (2013 framework) published by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), known as COSO, to evaluate the effectiveness of our internal control over financial reporting. Based on this assessment, management has concluded that our internal control over financial reporting was effective as of December 31, 2017. Ernst & Young, LLP, our independent registered public accounting firm, has issued an attestation report on our internal control over financial reporting as of December 31, 2017, which is included herein.
There has been no change in our internal control over financial reporting during our most recent fiscal quarter that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
96
Table of Contents
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors of
Neurocrine Biosciences, Inc.
Opinion on Internal Control over Financial Reporting
We have audited Neurocrine Biosciences, Inc.’s internal control over financial reporting as of December 31, 2017, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), (the COSO criteria). In our opinion, Neurocrine Biosciences, Inc. (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2017, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2017 and 2016, the related consolidated statements of operations and comprehensive loss, stockholders’ equity, and cash flows, for each of the three years in the period ended December 31, 2017, and the related notes and our report dated February 13, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
San Diego, California
February 13, 2018
97
Table of Contents
Table of Contents
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
Information required by this item will be contained in our Definitive Proxy Statement for our 2018 Annual Meeting of Stockholders, to be filed pursuant to Regulation 14A with the Securities and Exchange Commission within 120 days of December 31, 2017. Such information is incorporated herein by reference.
We have adopted a code of ethics that applies to our Chief Executive Officer, Chief Financial Officer, and to all of our other officers, directors, employees and agents. The code of ethics is available at the Corporate Governance section of the Investors page on our website at www.neurocrine.com. We intend to disclose future amendments to, or waivers from, certain provisions of our code of ethics on the above website within four business days following the date of such amendment or waiver.
| ITEM 11. | EXECUTIVE COMPENSATION |
Information required by this item will be contained in our Definitive Proxy Statement for our 2018 Annual Meeting of Stockholders, to be filed pursuant to Regulation 14A with the Securities and Exchange Commission within 120 days of December 31, 2017. Such information is incorporated herein by reference.
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
Information required by this item will be contained in our Definitive Proxy Statement for our 2018 Annual Meeting of Stockholders, to be filed pursuant to Regulation 14A with the Securities and Exchange Commission within 120 days of December 31, 2017. Such information is incorporated herein by reference.
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
Information required by this item will be contained in our Definitive Proxy Statement for our 2018 Annual Meeting of Stockholders, to be filed pursuant to Regulation 14A with the Securities and Exchange Commission within 120 days of December 31, 2017. Such information is incorporated herein by reference.
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
Information required by this item will be contained in our Definitive Proxy Statement for our 2018 Annual Meeting of Stockholders, to be filed pursuant to Regulation 14A with the Securities and Exchange Commission within 120 days of December 31, 2017. Such information is incorporated herein by reference.
99
Table of Contents
| ITEM 15. | EXHIBITS, FINANCIAL STATEMENT SCHEDULES |
(a) Documents filed as part of this report.
1. List of Financial Statements. The following are included in Item 8 of this report:
Report of Independent Registered Public Accounting Firm
Consolidated Balance Sheets as of December 31, 2017 and 2016
Consolidated Statements of Comprehensive Loss for the years ended December 31, 2017, 2016 and 2015
Consolidated Statements of Stockholders’ Equity for the years ended December 31, 2017, 2016 and 2015
Consolidated Statements of Cash Flows for the years ended December 31, 2017, 2016 and 2015
Notes to the Consolidated Financial Statements (includes unaudited Selected Quarterly Financial Data)
2. List of all Financial Statement schedules. All schedules are omitted because they are not applicable or the required information is shown in the Financial Statements or notes thereto.
3. List of Exhibits required by Item 601 of Regulation S-K. See part (b) below.
(b) Exhibits. The following exhibits are filed as part of, or incorporated by reference into, this report:
100
Table of Contents
101
Table of Contents
| Exhibit Number |
Description | |
| 31.2 | Certification of Chief Financial Officer pursuant to Rules 13a-14 and 15d-14 promulgated under the Securities Exchange Act of 1934 | |
| 32*** | Certifications of Chief Executive Officer and Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
| 101.INS | XBRL Instance Document. | |
| 101.SCH | XBRL Taxonomy Extension Schema Document. | |
| 101.CAL | XBRL Taxonomy Extension Calculation Linkbase Document. | |
| 101.DEF | XBRL Taxonomy Extension Definition Linkbase Document. | |
| 101.LAB | XBRL Taxonomy Extension Label Linkbase Document. | |
| 101.PRE | XBRL Taxonomy Extension Presentation Linkbase Document. | |
| (1) | Incorporated by reference to the Company’s Registration Statement on Form S-1 (Registration No. 333-03172) |
| (2) | Incorporated by reference to Exhibit 4.1 of the Company’s Current Report on Form 8-K filed on May 2, 2017 |
| (3) | Incorporated by reference to Exhibit 99.1 of the Company’s Current Report on Form 8-K filed on May 2, 2017 |
| (4) | Incorporated by reference to Exhibit 10.1 of the Company’s Quarterly Report on Form 10-Q filed on July 30, 2009 |
| (5) | Incorporated by reference to Exhibit 10.1 of the Company’s Quarterly Report on Form 10-Q filed on November 1, 2017 |
| (6) | Incorporated by reference to Exhibit 10.3 of the Company’s Annual Report on Form 10-K filed on February 14, 2017 |
| (7) | Incorporated by reference to Exhibit 99.2 of the Company’s Current Report on Form 8-K filed on January 18, 2012 |
| (8) | Incorporated by reference to Exhibit 10.1 of the Company’s Quarterly Report on Form 10-Q filed on August 3, 2017 |
| (9) | Incorporated by reference to Exhibit 10.3 of the Company’s Quarterly Report on Form 10-Q filed on November 1, 2017 |
| (10) | Incorporated by reference to Exhibit 10.3 of the Company’s Current Report on Form 8-K filed on December 10, 2007; Exhibit 10.5 of the Company’s Annual Report on Form 10-K filed on February 9, 2015; and Exhibit 10.2 of the Company’s Quarterly Report on Form 10-Q filed on August 3, 2017 |
| (11) | Incorporated by reference to Exhibit 10.3 of the Company’s Quarterly Report on Form 10-Q filed on August 3, 2007 |
| (12) | Incorporated by reference to Exhibit 10.1 of the Company’s Quarterly Report on Form 10-Q filed on July 26, 2013 |
| (13) | Incorporated by reference to Exhibit 10.35 of the Company’s Annual Report on Form 10-K filed on February 11, 2008 |
| (14) | Incorporated by reference to Exhibit 10.36 of the Company’s Annual Report on Form 10-K filed on February 11, 2008 |
| (15) | Incorporated by reference to Exhibit 10.37 of the Company’s Annual Report on Form 10-K filed on February 11, 2008 |
| (16) | Incorporated by reference to Exhibit 99.1 of the Company’s Current Report on Form 8-K filed on May 24, 2017 |
| (17) | Incorporated by reference to Exhibit 99.1 of the Company’s Current Report on Form 8-K filed on June 1, 2015 |
102
Table of Contents
| (18) | Incorporated by reference to Exhibit 10.2 of the Company’s Quarterly Report on Form 10-Q filed on July 29, 2010 |
| (19) | Incorporated by reference to Exhibit 10.32 of the Company’s Annual Report on Form 10-K filed on February 10, 2011 |
| (20) | Incorporated by reference to Exhibit 10.1 of the Company’s Quarterly Report on Form 10-Q filed on July 29, 2015 |
| (21) | Incorporated by reference to Exhibit 10.1 of the Company’s Quarterly Report on Form 10-Q filed on April 30, 2015 |
| (22) | Incorporated by reference to Exhibit 10.2 of the Company’s Quarterly Report on Form 10-Q filed on October 31, 2011 |
| (23) | Incorporated by reference to Exhibit 10.2 of the Company’s Quarterly Report on Form 10-Q filed on November 1, 2017 |
| (24) | Incorporated by reference to Exhibit 99.3 of the Company’s Current Report on Form 8-K filed on April 25, 2017 |
| (25) | Incorporated by reference to Exhibit 99.1 of the Company’s Current Report on Form 8-K filed on April 25, 2017 |
| (26) | Incorporated by reference to Exhibit 99.2 of the Company’s Current Report on Form 8-K filed on April 25, 2017 |
| (27) | Incorporated by reference to Exhibit 99.4 of the Company’s Current Report on Form 8-K filed on April 25, 2017 |
| * | Confidential treatment has been granted with respect to certain portions of the exhibit. |
| ** | Management contract or compensatory plan or arrangement. |
| *** | These certifications are being furnished solely to accompany this annual report pursuant to 18 U.S.C. Section 1350, and are not being filed for purposes of Section 18 of the Securities Exchange Act of 1934 and are not to be incorporated by reference into any filing of Neurocrine Biosciences, Inc., whether made before or after the date hereof, regardless of any general incorporation language in such filing. |
Except as specifically noted above, the Company’s Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K have a Commission File Number of 000-22705.
(c) Financial Statement Schedules. See Item 15(a)(2) above.
103
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| NEUROCRINE BIOSCIENCES, INC. | ||
| A Delaware Corporation | ||
| By: | /s/ Kevin C. Gorman | |
| Kevin C. Gorman | ||
| Chief Executive Officer | ||
Date: February 13, 2018
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed by the following persons on behalf of the Registrant and in the capacities and on the dates indicated:
| Signature |
Title |
Date | ||
| /s/ Kevin C. Gorman Kevin C. Gorman |
Chief Executive Officer and Director (Principal Executive Officer) |
February 13, 2018 | ||
| /s/ Matthew C. Abernethy Matthew C. Abernethy |
Chief Financial Officer (Principal Financial and Accounting Officer) |
February 13, 2018 | ||
| /s/ William H. Rastetter William H. Rastetter |
Chairman of the Board of Directors |
February 13, 2018 | ||
| /s/ Gary A. Lyons Gary A. Lyons |
Director |
February 13, 2018 | ||
| /s/ George J. Morrow George J. Morrow |
Director |
February 13, 2018 | ||
| /s/ Corinne H. Nevinny Corinne H. Nevinny |
Director |
February 13, 2018 | ||
| /s/ Richard F. Pops Richard F. Pops |
Director |
February 13, 2018 | ||
| /s/ Alfred W. Sandrock, Jr. Alfred W. Sandrock, Jr. |
Director |
February 13, 2018 | ||
| /s/ Stephen A. Sherwin Stephen A. Sherwin |
Director |
February 13, 2018 | ||
104
