Attached files
| file | filename |
|---|---|
| EX-32.2 - EXHIBIT 32.2 - Edesa Biotech, Inc. | tv479298_ex32-2.htm |
| EX-32.1 - EXHIBIT 32.1 - Edesa Biotech, Inc. | tv479298_ex32-1.htm |
| EX-31.2 - EXHIBIT 31.2 - Edesa Biotech, Inc. | tv479298_ex31-2.htm |
| EX-31.1 - EXHIBIT 31.1 - Edesa Biotech, Inc. | tv479298_ex31-1.htm |
| EX-23.1 - EXHIBIT 23.1 - Edesa Biotech, Inc. | tv479298_ex23-1.htm |
| EX-21 - EXHIBIT 21 - Edesa Biotech, Inc. | tv479298_ex21.htm |
| EX-14.1 - EXHIBIT 14.1 - Edesa Biotech, Inc. | tv479298_ex14-1.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| þ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended September 30, 2017
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 001-37619
Stellar Biotechnologies, Inc.
(Exact name of registrant as specified in its charter)
| British Columbia, Canada | N/A |
|
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
|
332 E. Scott Street Port Hueneme, California |
93041 |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: (805) 488-2800
Securities registered pursuant to Section 12(b) of the Act:
| Name of each exchange on which | |
| Title of each class | registered |
| Common Shares, without par value | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ¨ | Accelerated filer ¨ |
| Non-accelerated filer x (Do not check if a smaller reporting company) | Smaller reporting company ¨ |
| Emerging growth company x |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. x
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ¨ No x
As of March 31, 2017, the last business day of the registrant’s most recently completed second fiscal quarter, the aggregate market value of the registrant’s outstanding common shares held by non-affiliates was approximately $16,119,024, which was calculated based on 10,136,258 common shares outstanding as of that date, of which 9,888,972 common shares were held by non-affiliates at the closing price of the registrant’s common shares on The Nasdaq Capital Market on such date.
As of November 30, 2017, the registrant had 10,520,096 common shares issued and outstanding.
Documents incorporated by reference: NONE
Stellar Biotechnologies, Inc.
Annual Report ON FORM 10-K
Fiscal Year Ended September 30, 2017
Table of Contents
| Page 2 |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the Securities Act) and Section 21E of the Securities Exchange Act of 1934, as amended (the Exchange Act) and, as such, may involve known and unknown risks, uncertainties and assumptions. Forward-looking statements are based upon our current expectations, speak only as of the date hereof, and are subject to change. Forward-looking statements are those that predict or describe future events or trends and that do not relate solely to historical matters. You can generally identify forward-looking statements as those statements containing the words “anticipate,” “believe,” “plan,” “estimate,” “expect,” “intend,” “may,” “will,” “would,” “could,” “should,” “might,” “potential,” “continue” or other similar expressions. You should not rely on our forward-looking statements as they are not a guarantee of future performance. There can be no assurance that forward-looking statements will prove to be accurate because the matters they describe are subject to assumptions, known and unknown risks, uncertainties and other unpredictable factors, many of which are beyond our control. Our actual results could differ materially and adversely from those expressed in any forward-looking statements as a result of various factors, some of which are listed under the “Risk Factors” section of this Annual Report. Risks and uncertainties include, among others, the availability of funds and resources to pursue our research and development projects, the successful and timely completion of preclinical or clinical studies by third parties in which our products are utilized, our ability to meet the goals of our joint ventures and strategic partnerships, the degree of market acceptance for our products or for other companies’ products in which our products are components, our ability to take advantage of business opportunities in the pharmaceutical industry, changes in our strategy or development plans, our ability to protect our intellectual property, uncertainties related to governmental regulations and regulatory processes, the volatility of our common share price, the effect of competition, the effect of technological changes, reliance on key personnel, and general changes in economic or business conditions. Except as required by law, we undertake no obligation to update forward-looking statements.
As used in this Annual Report on Form 10-K, “Stellar,” “the Company,” “we,” “us,” and “our” refer to Stellar Biotechnologies, Inc. and our consolidated subsidiaries, except where the context otherwise requires.
Our logo, Stellar KLH and other trademarks or service marks of Stellar Biotechnologies, Inc. appearing in this Annual Report on Form 10-K are the property of Stellar Biotechnologies, Inc. This Annual Report on Form 10-K contains additional trade names, trademarks and service marks of other companies. We do not intend our use or display of other companies’ trade names, trademarks or service marks to imply relationships with, or endorsement or sponsorship of us by, these other companies.
| Page 3 |
| Item 1. | BUSINESS. |
Overview
Stellar Biotechnologies, Inc. is a biotechnology company engaged in the aquaculture, research and development, manufacture and commercialization of Keyhole Limpet Hemocyanin (KLH). KLH is an immune-stimulating protein with an extensive history of safe and effective use in immunological applications.
Immunotherapies (also known as therapeutic vaccines) are an emerging class of treatments that involve using the body’s own immune system to target and treat disease. Today, multiple companies and institutions are developing drugs that combine disease-targeting agents with KLH. These disease-targeting agents do not evoke a robust immune response by themselves and thus require a carrier molecule like KLH.
The versatility of the KLH molecule and its use in multiple drug development pipelines provide numerous commercial opportunities for us. KLH is currently utilized in immunotherapies in clinical or pre-clinical development for Alzheimer’s disease, metastatic breast cancer, type 1 diabetes, dermatomyositis, systemic lupus erythematous, ovarian cancer and various other cancers and diseases. The successful commercialization of one or more of these drug development pipelines, especially in a major indication, could have a significant impact on the industry’s ability to produce sufficient quantities of KLH. The protein is derived only from the Giant Keyhole Limpet, a scarce ocean mollusk that is native to a limited stretch of Pacific Ocean coastline. Due in part to the inherent limitations of utilizing of wild sources of KLH, we believe that aquaculture production methods, like the methods we practice, will be required to provide scalable, fully traceable supplies of KLH.
Based upon our specialized knowledge of aquaculture science and KLH, we have built unique land-based aquaculture, laboratory and production facilities in Port Hueneme, California, and developed production and manufacturing processes to produce medical-grade KLH using Current Good Manufacturing Practices (GMP). Using our proprietary aquaculture technology, we can support the marine mollusk from embryo to protein-producing adult, and we now support multiple generations of limpets grown entirely within our land-based aquaculture facility. Other KLH suppliers do not have this capability and thus are reliant on scarce, wild populations of limpets.
We market and sell our KLH products under the brand Stellar KLH. Our customers and partners include multinational biotechnology and pharmaceutical companies, academic institutions, clinical research organizations and research centers. We have multiple agreements to license and supply Stellar KLH and other technology in exchange for fees, revenues or royalties. Our customers manage and fund all product development and regulatory submissions for their respective drug products that utilize our KLH protein.
Competitive Strengths
We believe that we possess a number of competitive strengths that position us to become the world leader in the sustainable manufacture of GMP grade KLH and KLH-conjugated vaccines, including:
| • | Fully permitted, land-based aquaculture facility produces a barrier to market entry. Our proprietary methods, infrastructure and aquaculture facility give us the capability to support the source animal in aquaculture. Due to the time needed to raise the source animal to maturity, and the time needed to build and validate facilities and manufacturing processes, including water discharge permits, we believe that we have a five to seven year lead over any new market entrants attempting to produce KLH in a similar manner. Due to its exceptional size and complexity, KLH has not been reproduced synthetically. |
| • | Fully traceable, GMP grade product offerings benefit commercialization programs. Using our proprietary production and manufacturing methods, we are able to produce a high quality, GMP grade KLH product that is fully traceable and controlled from native source to finished product, which we believe are important considerations for our pharmaceutical partners as they pursue later-stage trials and commercial introductions subject to more rigorous regulatory standards than early-stage research. Due to the known origin of material and continuity of data, we believe we are able to create a more consistent, high quality, immunogenic product than other KLH proteins in the market. In contrast, commercial supplies of KLH from other sources have historically differed widely in their source, traceability, purity, form and preparation, as well as their immunogenicity (their ability to stimulate an immune response). We believe that we are the only company that offers GMP grade KLH supported by fully traceable manufacturing methods. |
| • | Multiple supply and collaboration agreements reduce single-customer dependence. We believe that our supply and collaboration agreements with drug developers, which include binding orders, allow us to better manage our working capital as well as build long- term relationships. Our manufacturing and quality experts work closely with our collaboration partners and customers to deliver KLH products according to their specifications. We believe that our long-term relationships and collaborative approach have helped build customer trust and loyalty. |
| Page 4 |
| • | Business model leverages growth potential. We believe we have an attractive business model due to the unique nature of our product offerings, embedded growth opportunities within our existing customer base and operating leverage. As we increase production volumes and sales, we expect our operating expenses to decrease as a percentage of revenue, providing for greater operating leverage. In addition, we have established a model via our joint venture, Neostell, S.A.S., to participate in the manufacturing of KLH-conjugated vaccines, which provides additional revenue and growth opportunities. |
| • | Intellectual property portfolio includes protection for specialized systems and technologies. We have intellectual property related to KLH development and manufacturing and to the environmental protection of the Giant Keyhole Limpet, including patents, trade secrets and know-how related to specialized aquaculture systems and technologies; spawning, selection and maintenance of the species; non- lethal KLH protein extraction methods; and the processing, purification and production of KLH formulations. |
| • | Safety profile and extensive citations in scientific literature contribute to the appeal of KLH as a carrier platform for immunotherapies. KLH has been used for decades in immune system testing, it has an extensive safety record, and continues to be selected for new immunotherapies preparing to enter clinical testing. According to a search on PubMed, a service of the U.S. National Library of Medicine, there are more than 3,600 publications referencing Keyhole Limpet Hemocyanin in biomedical literature. |
| • | Sustainability practices protect marine source and promote scalability Our KLH protein is produced using environmentally sound, sustainable practices intended to protect and renew the live marine source. We believe this is a critical component of ensuring long-term, scalable supplies, since rapid growth in demand has had severe consequences to other related species. In California, for example, failure to manage wild populations of abalone resulted in dramatic declines and eventually led to closure of commercial abalone harvests. |
| • | Leadership team provides extensive aquaculture production and related industry expertise. Our leadership team includes industry experts who have extensive experience in the field of aquaculture and Giant Keyhole Limpet production, and possess a deep understanding of a variety of biotechnology businesses. Our President and CEO has more than 40 years of experience leading commercial aquaculture businesses and projects focused on mollusk domestication and production. |
Our Strategy
We intend to develop and expand the market for KLH and KLH-conjugated vaccines. Our near-term focus is to support the further development of third party drug candidates utilizing Stellar KLH and to expand our customer base. This strategy seeks to preserve the opportunity for Stellar to share in the successful development and commercialization of product candidates utilizing our licensed KLH products. In addition to fees, revenues or royalties we may receive, we believe that the successful development of third party drug candidates will further validate our technologies, increase awareness and promote broader adoption of our products by additional third parties. Key elements of our business strategy include:
| • | Expand infrastructure and capacity while prudently managing our working capital. We currently have multiple customers with KLH-based drug candidates in Phase 2 studies. While the outcome of these clinical studies cannot be predicted, we are preparing for the possible impact that favorable clinical results could have on the KLH market and the company’s supply capabilities. We plan to incrementally increase our infrastructure, manufacturing capabilities and KLH production capacity based on our customers’ forecasts and the anticipated future requirements of commercial-scale vaccine manufacturing, which we estimate could require multiple kilograms of GMP grade KLH per year. In order to produce such volumes and to provide our customers with greater certainty of future supply, we intend to have the capacity to support commercial drug launches in a variety of indications, with planned redundancy at multiple locations. We also plan to increase efficiency and throughput capacity by optimizing our manufacturing and purification processes. |
| • | Pursue additional supply and collaboration agreements. We plan to continue pursuing opportunities for commercial growth that build on our strengths and core competencies in KLH development and manufacturing, including additional supply and collaboration agreements. We regularly engage in discussions with various entities involved in immunotherapies, in connection with opportunities for licensing, supply and collaborative research. |
| • | Support continuing development of our Neostell Growth Initiative. In July 2016, we formed Neostell S.A.S., a joint venture with Neovacs S.A, to produce Neovacs’ Kinoid immunotherapy product candidates which utilize Stellar KLH as a carrier molecule. In addition to expanding our market opportunities related to manufacturing of Neovacs’ KLH-conjugated vaccines, this joint venture provides the opportunity to manufacture and sell KLH-based immunotherapies for third party customers. |
| Page 5 |
| • | Continue innovation and new product development. We plan to expand our KLH technology portfolio through ongoing research and development. Our research and development activities are focused primarily on the aquaculture of the Giant Keyhole Limpet; improvements in KLH protein characterization and manufacturing; the development of functional assays; and new uses for KLH in immunotherapy and immunodiagnostic applications. We believe that these activities provide long-term strategic, revenue and clinical opportunities by extending the commercial use of Stellar KLH and furthering our understanding of the KLH molecule. |
| • | Pursue additional markets for our technology and products. We intend to evaluate additional markets for our current products and technologies. Due to the immune-stimulating characteristics of KLH, we believe the protein could have broader applications in the medical field or other markets. |
Keyhole Limpet Hemocyanin (KLH)
KLH is a safe, potent, immune-stimulating protein. Specifically, it is a very large, high molecular weight, oxygen-carrying glycoprotein. In addition to the native molecule, KLH can be chemically dissociated into a subunit formulation commonly used in the production of immunotherapies. Both the native, high molecular weight molecule and subunit forms of KLH are excellent immune stimulants. The KLH molecular structure offers numerous sites for conjugation, and can generate multiple product configurations. Because of its large size, immune-stimulating properties, numerous sites for conjugation, and safety profile, KLH is used by researchers and product developers as a vaccine carrier protein. However, due to its exceptional size and complexity, KLH has not been reproduced synthetically.
KLH can be used as a carrier molecule, or it can be used as a finished, injectable product in the immunodiagnostic market.
As a carrier molecule, KLH is combined, or conjugated, to vaccine antigens that are used to promote the generation of antibody and cell-mediated immune responses against targeted diseases. By themselves, the small haptens (partial antigens) and vaccine antigens used to target these diseases are not usually immunogenic enough to awaken the immune system and therefore, require a carrier molecule or adjuvant, like KLH, in order to be effective. The combination of an antigen against specific pathogenic targets, such as tumors, and over-expressed proteins, conjugated to the immunogenic KLH molecule, is the basis for a promising new class of drugs in development known as active immunotherapies or therapeutic vaccines. Unlike preventative vaccines, active immunotherapies are designed to stimulate the body’s own immune system to generate an immune response to target and attack an existing disease or condition. We believe immunotherapies are, and will continue to be, one of the fastest-growing sectors of pharmaceutical research and development. KLH is an important component for drugs used in clinical development, including major indications such as Alzheimer’s disease, metastatic breast cancer, systemic lupus erythematous, dermatomyositis, ovarian cancer and various other cancers and diseases. New indications expected to enter clinical trials, such as type 1 diabetes, point to expanding clinical potential for KLH.
As a finished injectable product, KLH has been used extensively by pharmaceutical companies and researchers as a safe, immune-stimulating antigen in drug-screening, drug immunotoxicology, and assessment of immune status. KLH is a standard immunogen in T-Cell Dependent Antibody Response (TDAR), a functional assay which is widely recognized as a standard test for monitoring the effects of drugs on the immune system.
KLH protein is derived only from the hemolymph of the Giant Keyhole Limpet (Megathura crenulata), a mollusk native only to a limited stretch of the Pacific Ocean coastline along Southern California and Baja California, Mexico. Historically, suppliers other than us have obtained KLH protein directly from wild and sensitive populations of Giant Keyhole Limpet, or have utilized lethal production processes. Based on publicly available information and reports, commercial supplies of KLH differ widely in their source, traceability, purity, form, and preparation, as well as in immunogenicity (their ability to stimulate an immune response). We believe that highly-specialized aquaculture manufacturing methods, like the methods we practice, protect the KLH molecule’s source species and provide sustainable, scalable supplies of quality KLH protein. The concept of sustainability involves sound, responsible management of environmental resources and, especially where biological systems are concerned, includes protecting native species so that the species thrive and remain diverse and productive over time. Further, we believe that environmentally sound methods associated with professional and specialized aquaculture can minimize variability in KLH products and assure full traceability to their biological source.
Our Technology
We have spent more than 15 years developing and optimizing sustainable KLH production methods, specifically focused on protection of the Giant Keyhole Limpet and a patented, non-lethal method to extract KLH protein. We believe our proprietary methods will provide a scalable supply of GMP grade KLH and meet pharmaceutical industry standards for immune response, consistency, purity, and traceability while protecting the natural source species.
| Page 6 |
Our proprietary aquaculture technology involves methods we developed and optimized to control the reproduction and growth of the Giant Keyhole Limpet including, but not limited to, culture systems, nutritional requirements and the recirculation of seawater. We achieved a significant milestone in aquaculture science by developing the capability to sustain the complete life cycle of the Giant Keyhole Limpet. Using our proprietary methods, we can support the marine mollusk from embryo to protein-producing adult, and we now support multiple generations of limpets grown entirely within our land-based aquaculture facility. We believe that other KLH suppliers do not have this capability and thus are reliant on scarce, wild populations of limpets.
The aquaculture cycle to raise Giant Keyhole Limpets from fertilized eggs to maturity for KLH production is approximately five years, with multiple complex larval and juvenile stages. The hemolymph circulatory fluid, which contains KLH, is extracted in a non-lethal manner utilizing our patented methods. Once extracted, the hemolymph is processed and purified through our proprietary methods, which are protected as trade secrets. KLH can be extracted from mature limpets multiple times per year.
We currently maintain a production inventory of limpets sufficient for an annual capacity of up to 1,500 grams/year of KLH pharmaceutical intermediate, which can be further processed and purified to produce various final product grades and formulations. We believe we can continue to scale up capacity to meet anticipated customer demand in the near term. Given sufficient funding to continue scale- up, our projected production capacity is up to 20,000 grams (20 kg) of KLH pharmaceutical intermediate in five to seven years. We plan to incrementally increase hatchery production of limpets and expand aquaculture infrastructure, which will thereby increase our KLH production capacity, in order to meet the anticipated future multi-kilogram KLH requirements of immunotherapy commercialization.
In December 2016, we initiated plans to optimize our protein manufacturing processes at our primary facility in Port Hueneme, California, including the evaluation and use of new equipment. This initiative is intended to increase the scalability and throughput capacity of existing manufacturing systems, which were originally developed to provide clinical development stage quantities of our Stellar KLH products.
We rely on contract manufacturing organizations and contract testing organizations for certain steps of cGMP processing and quality control testing. The services performed by these contract vendors have included sterile fill/finish and product release testing.
As a result of these operational capabilities, we believe we will be able to supply GMP grade KLH in commercial quantities to meet the anticipated long-term demand within the pharmaceutical industry, while protecting the natural source species. We base these beliefs on our intellectual property, achievements in aquaculture science, KLH production capacity, KLH sustainable manufacturing know-how, and survey data used to estimate populations of Giant Keyhole Limpets in the wild.
Our Aquaculture and KLH Production Facilities
We maintain research and manufacturing facilities directly along the Pacific Ocean with dedicated, land-based aquaculture operations in Port Hueneme, California. We have approximately 37,000 square feet of leased aquaculture, manufacturing and laboratory space. We believe our waterfront location is a proprietary asset that allows our marine scientists to work in close proximity to naturally resident Giant Keyhole Limpet colonies, and to be at the forefront in developing protective measures and environmentally sound practices for KLH production. At this location, our seawater supply and discharge system is fully permitted, which we believe is a competitive strength due in part to the time required and uncertainties related to obtaining new water discharge permits in the State of California.
Our aquaculture operations include, among other specialized infrastructure, systems for spawning, larval development, and maturation of limpets, recirculating seawater supply systems and environmental controls. Our facility currently includes multiple production tanks and numerous individual limpet production modules in two independent aquaculture production systems. Each closed recirculating system is equipped with temperature controlled seawater distribution, filtration and treatment equipment. The facility also contains a fabrication shop for production of equipment and culture apparatus.
Additional Aquaculture and KLH Production Locations
In January 2017, we established a wholly owned Mexican subsidiary under the name BioEstelar, S.A. de C.V. to support our plan to establish additional aquaculture capabilities in Baja California, including the development of regional marine resources, aquaculture and raw material processing for Stellar’s KLH products. Since 2015, we have leased undeveloped land in Baja California as part of multi-year site suitability studies. We have a related, exclusive collaboration agreement with the lessor to collaborate on the design, expansion and development of marine aquaculture resources for hatchery and maturation of Giant Keyhole Limpets on the leased property. The collaboration agreement expires in June 2018, unless terminated earlier. We believe this expansion in Mexico will support our goal to meet the anticipated long-term industry demand for KLH protein.
| Page 7 |
Research and Development
Our research and development is focused primarily on the aquaculture of the Giant Keyhole Limpet; improvements in KLH protein characterization and manufacturing; the development of functional assays; and new uses for KLH in immunotherapy and immunodiagnostic applications. These activities involve both internal programs and external collaborations with other biopharmaceutical companies or research organizations.
Our internal research has included, among other activities, improvement of methods for the culture and growth of Giant Keyhole Limpet, developing proprietary formulated limpet diets, innovations in aquaculture systems and infrastructure, biophysical and biochemical characterization of the KLH molecule, analytical processes to enhance performance of our products, KLH manufacturing process improvements, and new KLH formulations and KLH-related technologies.
Our external collaborations have historically involved both development and evaluation projects, with multiple biopharmaceutical companies and research institutions, for the use of Stellar KLH in their programs. We believe that these collaborations provide for strategic, revenue and clinical opportunities for our future business by extending the commercial use of Stellar KLH and furthering our understanding of the KLH molecule.
For the years ended September 30, 2017, 2016 and 2015, our research and development expense were $1.97 million, $1.73 million and $1.03 million, respectively. These amounts related mainly to research and development in aquaculture, improvements in analytical, manufacturing, and purification processes, stability studies and formulation development.
Our Stellar KLH Products
We offer Stellar KLH protein in various grades, formulations, custom configurations and fill finishes for both drug development and research applications. Our portfolio includes GMP and research grade products intended for: conjugation as a carrier molecule in therapeutic vaccines; assessing immune function; and, in immunotoxicology studies, for monitoring the immunomodulatory effects of drug candidates. We also offer KLH-based in vitro diagnostic kits for research and preclinical use.
We currently have limited revenue from sales of our Stellar KLH products. The list price for bulk Stellar KLH protein ranges from approximately $15,000 to $50,000 per gram, depending on the purity, grade, preparation, packaging configuration and volume ordered. While our customer base has not changed significantly from year to year, product sales volumes have been highly dependent and subject to variability associated with the rate of development and progression of clinical studies of third-party immunotherapies and other technologies that utilize our products. The rate of progression towards later stage studies is expected to continue to affect the timing and volume of future product sales. The advancement and commercial success of third-party products utilizing Stellar KLH is dependent upon many factors, including available capital, trial recruitment and progress, and regulatory review.
Revenues from the sale of products and contract services revenues in fiscal years 2017, 2016 and 2015 are as follows:
| 2017 | 2016 | 2015 | ||||||||||
| Product sales | $ | 178,287 | $ | 1,239,689 | $ | 563,689 | ||||||
| Contract services revenue | 50,000 | 32,000 | 195,000 | |||||||||
The geographic breakdown of revenues in fiscal years 2017, 2016 and 2015 are as follows:
| 2017 | 2016 | 2015 | ||||||||||
| Europe | 64 | % | 43 | % | 53 | % | ||||||
| North America | 33 | % | 12 | % | 9 | % | ||||||
| Asia | 3 | % | 45 | % | 38 | % | ||||||
| Page 8 |
Drug Master Files for Stellar KLH
We have submitted Type II Master Files for our high molecular weight and subunit KLH product formulations to the FDA Center for Biologics Evaluation and Research (CBER) and Type II Drug Master Files to the FDA Center for Drug Evaluation and Research (CDER). A Master File is a confidential, detailed dossier kept on file at the FDA that contains the proprietary information on the manufacture and safety of a drug component. These files can be used to support the regulatory approval process for customers’ immunotherapy products that use our Stellar KLH, while allowing us to control access to our manufacturing data.
Customers
We primarily market and distribute our products directly to biotechnology and pharmaceutical companies, academic institutions, clinical research organizations and research centers. Products are shipped to our customers from our facilities in Port Hueneme, California using a common carrier chosen by the customer. The geographic markets of our customers are principally Europe, North America and Asia.
The customers that represent 10% or more of our total consolidated revenue in fiscal years 2017, 2016 and 2015 are as follows:
| Customer | Percentage | |||
| 2017 | ||||
| Araclon Biotech, SL | 57 | % | ||
| Matrivax R&D Corporation | 22 | % | ||
| 2016 | ||||
| OBI Pharma, Inc. | 41 | % | ||
| Eurogentec | 25 | % | ||
| Neovacs SA | 10 | % | ||
| 2015 | ||||
| Araclon Biotech, SL | 19 | % | ||
| Amaran Biotechnology, Inc. | 19 | % | ||
| OBI Pharma, Inc. | 17 | % | ||
| AXON Neuroscience SE | 16 | % | ||
| Neovacs SA | 15 | % | ||
Supply Agreements, Collaboration Agreements and Contracts
We have entered into, and intend to continue to enter into, agreements with third parties that will allow us to supply Stellar KLH in exchange for fees, revenues or royalties. Supply agreements generally involve a customer’s commitment to purchase our Stellar KLH for use as a carrier molecule in the customer’s own immunotherapy products or as a finished product in their development programs. In return, we license and provide exclusive or priority supply in a given field and territory, and provide technical and regulatory support. When applicable, we also agree to maintain a master file with the U.S. Food and Drug Administration (FDA) for the KLH product. Our current supply agreements are limited to clinical trials and typically have an initial multi-year term, which may be renewed by customers for additional one-year periods. Our supply agreements also typically provide us with first negotiation rights for the supply of KLH in connection with potential future commercialization of a customer’s products.
To date, our Stellar KLH protein has been used in research and development, preclinical and clinical phases of development but has not yet been used in any commercialized and marketed drug products. Quantities required for clinical trials depend on, among other variables, the nature of the trial, the clinical indication, the number of patients enrolled, dosing regimens and vaccine manufacturing processes.
| Page 9 |
We have supply agreements with Araclon Biotech SL, a privately-held biotechnology company headquartered in Spain and majority-owned by global healthcare company Grifols, who is developing beta amyloid-targeting active immunotherapies for neurodegenerative diseases with a primary focus on Alzheimer's disease; Amaran Biotechnology, Inc., a biopharmaceuticals manufacturer based in Taiwan that manufactures a KLH conjugate vaccine for OBI Pharma, Inc., a publicly-listed Taiwan biotech company; and French biotechnology company Neovacs S.A, for the use of Stellar KLH in the development and manufacture of Neovacs’ active immunotherapies. As previously disclosed, our agreement with Neovacs provides for Neovacs to purchase Stellar KLH for use in its proprietary KLH-based Kinoid immunotherapies in the European Union, Latin America, Asia, the U.S. and Canada. Our customers manage and fund all product development and regulatory submissions for their respective drug products that utilize Stellar KLH.
Neostell Joint Venture Agreement
In May 2016, we entered into a joint venture agreement with Neovacs S.A., a publicly-held biotechnology company in Paris, France for the formation of a joint venture company to manufacture and sell conjugated therapeutic vaccines. In July 2016, Neostell S.A.S., a French simplified stock corporation (Neostell), was formed to carry out the business of the joint venture. Neostell is expected to produce Neovacs’ Kinoid immunotherapy product candidates which utilize Stellar KLH as a carrier molecule. Neostell may also manufacture and sell other KLH-based immunotherapy products for third-party customers worldwide.
We hold a 30% equity interest in the joint venture in exchange for an initial capital contribution of €120,000. One-half of the initial contribution, approximately $67,000, was paid in June 2016 with the balance due upon the occurrence of certain defined future events. We will also provide additional financing to Neostell, as may be required, on a pro rata basis in line with our equity interest. If Neostell does not achieve certain milestones by December 31, 2017, it will be dissolved, unless the parties mutually agree to pursue the joint venture arrangement, or either party decides to purchase the equity interests of the other party. As of the date of this Annual Report, these milestones have not been achieved, and the parties have discussed their mutual desire to extend the deadline. Each of the parties is entitled, upon the occurrence of certain defined events, to acquire the interest of the other party. Except as otherwise described herein, the joint venture has an initial ten-year term, renewable for successive five-year terms. If either party provides notice at least six months prior to the expiration date of an applicable term that it does not wish to continue its participation in the joint venture, the other party will have a right to acquire all of such terminating party’s equity interests in Neostell.
In connection with the formation of Neostell and the execution of its strategy, the parties intend over time to enter into an exclusive supply agreement within a limited field of use for Stellar to supply KLH to Neostell, a supply agreement designating Neostell as the exclusive manufacturer and supplier of the Neovacs’ vaccines, and services agreements for the provision of various knowledge and expertise by each of the parties. Neovacs will also license certain of its intellectual property to Neostell.
Intellectual Property and License Agreements
We hold important proprietary intellectual property related to KLH development and manufacture and to the environmental protection of the Giant Keyhole Limpet including, but not limited to, patents and trade secrets related to specialized aquaculture systems and technologies; spawning, selection and maintenance of the Giant Keyhole Limpet; non-lethal KLH protein extraction methods; and the processing, purification and production of KLH formulations. Our proprietary methods also include methods for the control of larval development, metamorphosis and maturation of the Giant Keyhole Limpets, which we protect as trade secrets.
Our success depends in part on our ability to obtain and maintain proprietary protection for our product technology and know-how, to operate without infringing proprietary rights of others, and to prevent others from infringing our proprietary rights. We seek to protect our proprietary position by, among other methods, filing, when possible, U.S. and foreign patent applications relating to our technology, inventions and improvements that are important to our business. We also rely on trade secrets, know-how, continuing technological innovation, and in-licensing opportunities to develop and maintain our proprietary position. We require our employees, consultants and advisors to execute confidentiality agreements in connection with their employment, consulting or advisory relationship with us. We also require our employees, and to the extent practicable, our consultants and advisors with whom we expect to work on our products to agree to disclose and assign to us all inventions made in the course of our working relationship with them, while using our intellectual property or which relate to our business.
| Page 10 |
We hold patent protection for our non-lethal extraction methods of hemocyanin in the United States and other countries, including one issued patent in the United States, U.S. Patent No. 6,852,338, which currently expires in 2023, and covers a two-step method for obtaining hemolymph from a live gastropod mollusk. This U.S. patent was originally granted to our Chief Executive Officer, Frank Oakes, who assigned the patent to the Company in August 2002. Foreign patent counterparts were granted in Canada, France and Germany. In August 2011 we acquired an exclusive, worldwide sub-licensable and royalty-free license to the technology we developed under collaboration with Bayer Innovation GmbH (Bayer) for the improved production method and process yields for Stellar KLH. The license included a carve-out by Bayer to use the technology in certain non-Hodgkin Lymphoma active immunotherapies, but we may exclusively commercialize the technology in other fields.
The scope of any patent protection may not exclude competitors or provide competitive advantages to us, and any of our patents may not be held valid if subsequently challenged, and others may claim rights in or ownership of our patents and proprietary rights. Furthermore, others may develop products similar to our products and may duplicate any of our products or design around our patents.
Our trademarks include, but are not limited to, “Powering and Improving Immunotherapy™”, “Stellar KLH™” and “KLH Site™”. In addition to patents and trademarks, we rely on trade secrets and other intellectual property laws, nondisclosure agreements and other measures to protect our intellectual property rights.
Competition
The immunotherapy industry is rapidly evolving and new competitors with competing technologies and products are regularly entering clinical development and the market. We compete on the basis of: the advantages and disadvantages of Stellar KLH as compared to other KLH proteins manufactured by our competitors; our ability to educate the industry about the high quality, and sustainable and traceable features, of Stellar KLH; our ability to supply scalable quantities of GMP grade KLH; product efficacy; customer service; and the price and demonstrated cost-effectiveness of Stellar KLH as compared to our competitors. We believe that our products and services currently compete favorably with respect to such factors. However, we may not be able to maintain our competitive position against current and potential competitors. We compete directly with Biosyn Corporation, a pharmaceutical and biotechnology company which manufactures KLH starting material and offers clinical and research grade KLH products. We also compete directly with SAFC, a division of Sigma-Aldrich, which offers clinical and research grade KLH products. In addition to competition from current suppliers of KLH, we also face indirect competition from developers of other carrier proteins, adjuvants or therapeutic vaccine platforms. We are unable to predict what effect evolution of the KLH and immunotherapy industries and potential new entrants may have on price, selling strategies, intellectual property or our competitive position.
Government Regulation
Our operations, including our aquaculture and harvesting activities, as well as production operations, manufacturing site development, and drug research, development and sales, are subject to complex regulation at the local, state and federal levels in the United States by a number of regulatory agencies including, but not limited to, the U.S. Food and Drug Administration, the U.S. Environmental Protection Agency, the U.S. Fish and Wildlife Service, the U.S. Secretary of the Navy, the Regional Water Quality Control Board Los Angeles Region, the California Department of Fish and Wildlife, the California Coastal Commission, the California Air Pollution Control Board, the County of Ventura, and the City of Port Hueneme.
We are subject to laws and regulations covering clean water and waste discharge, and are required to hold licenses for the aquaculture production and wild harvesting of the Giant Keyhole Limpet. Our aquaculture facility is subject to regulation by the California Department of Fish and Wildlife and the Regional Water Quality Control Board, Los Angeles Region. These agencies impose regulations that restrict any activity that could pose a potential risk to the California marine environment including, but not limited to, seawater waste discharge limitations specified in our National Pollution Discharge Elimination Systems (NPDES) permit. We regularly monitor our KLH production and manufacturing processes for compliance with applicable regulations.
In addition to regulations in the United States, we may be subject to a variety of foreign regulations related to research, manufacturing, and the commercial sale and distribution of our products, to the extent we choose to manufacture, sell or distribute any products outside of the United States. The requirements governing our activities in jurisdictions outside the United States vary greatly from country to country.
In Mexico, our current research and development activities and collaborations, and potential future operations, are subject to regulation, permitting and oversight by the Secretariat of Agriculture, Livestock, Rural Development, Fisheries and Food (SAGARPA), including the National Service of Health, Food Safety and Quality (SENASICA), the National Commission of Fisheries and Aquaculture (CONAPESCA), and the National Institute of Fisheries and Aquaculture (INAPESCA), all of which are administrative bodies of SAGARPA. We are also subject to regulation, permitting and oversight by the Secretariat of the Environment and Natural Resources (SEMARNAT), the Secretariat of Health’s Federal Commission for the Protection Against Sanitary Risks (COFEPRIS), and by and other state and local agencies.
| Page 11 |
Good Manufacturing Practices
The FDA and other regulatory agencies regulate and inspect equipment, facilities and processes used in the manufacture of pharmaceutical and biologic products prior to approving a product. If, after receiving approval from regulatory agencies, a company makes a material change in manufacturing equipment, location or process, additional regulatory review and approval may be required. All facilities and manufacturing techniques used for the manufacture of our products must comply with applicable regulations governing the production of pharmaceutical products known as Current Good Manufacturing Practices. These requirements include, among other things, quality control, quality assurance and the maintenance of records and documentation. We are responsible for regularly assessing compliance with GMP requirements through record reviews and periodic audits and for ensuring that we take corrective action for any identified deficiencies.
The FDA and other regulatory agencies also conduct regular, periodic visits to re-inspect equipment, facilities and processes following initial approval of a product. If, as a result of these inspections, it is determined that our equipment, facilities or processes do not comply with applicable regulations and conditions of product approval, regulatory agencies may issue warning or similar letters or may seek civil, criminal, or administrative sanctions against us. To date, we have not been subject to inspection by the FDA or other drug regulatory agency because none of our customers or partners has filed an application in any country for marketing approval of a product encompassing our Stellar KLH protein.
New Drug Development
None of our KLH products have been subject to approval as a drug by any regulatory authority. However, a number of our customers and strategic partners are utilizing Stellar KLH in the development of pharmaceuticals and immunotherapies that are subject to the regulatory approval process in various jurisdictions. The regulatory approval process for new drugs under development by our customers is typically long and expensive. Clinical trials that they conduct may not be successful and such products may not receive regulatory approval. Delays by our customers in obtaining, or the inability to obtain, regulatory approvals for their products which use Stellar KLH will have a direct effect on the demand for our products.
Employees
As of November 30, 2017, we had 28 employees. We consider our employee relations to be good. None of our employees are represented by a labor union or collective bargaining agreement.
Corporate Information
We operate through our wholly-owned subsidiary, Stellar Biotechnologies, Inc., a California corporation which was organized September 9, 1999. We acquired the subsidiary on April 12, 2010 through a reverse merger and began trading on the TSX Venture Exchange (TSX-V) under the symbol “KLH” on April 19, 2010. We were originally incorporated in Canada on June 12, 2007 under the name China Growth Capital, Inc. and subsequently changed our name to CAG Capital, Inc. on April 15, 2008. We began trading on the TSX Venture Exchange as a Canadian capital pool company on August 29, 2008, and became a British Columbia corporation on November 25, 2009. Our reverse merger in April 2010 constituted our “qualifying transaction” under Canadian law, at which time we changed our name to Stellar Biotechnologies, Inc. In January 2013, we began trading on the U.S. OTCQB Marketplace Exchange under the symbol “SBOTF” and, on November 5, 2015, our common shares began trading on The Nasdaq Capital Market (Nasdaq) under the symbol “SBOT.” On March 29, 2016, at our request, our common shares were voluntarily delisted from the TSX-V in Canada and ceased trading on the TSX-V as of the close of business on April 8, 2016.
Our executive offices are located at 332 East Scott Street, Port Hueneme, California 93041. Our phone number is (805) 488-2800. Our website address is www.stellarbiotechnologies.com. The contents of our website are not part of this Annual Report on Form 10-K for any purpose or otherwise incorporated by reference. Our website address is included for information only.
Available Information
We file or furnish periodic reports, including our annual reports on Form 10-K, our quarterly reports on Form 10-Q and current reports on Form 8-K, proxy statements and other information with the SEC, and amendments to such reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange of 1934, as amended, as soon as reasonably practicable after we electronically file or furnish such materials to the SEC. Such reports and filings may be obtained by visiting the Public Reference Room of the SEC at 100 F Street, NE, Washington, D.C. 20549, by calling the SEC at (800) SEC-0330 or by sending an email to the SEC at publicinfo@sec.gov. In addition, the SEC maintains a website (www.sec.gov) that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC. Our reports, proxy statements and other information are also made available, free of charge, on our investor relations website at ir.stellarbiotechnologies.com as soon as reasonably practicable after we electronically file such information with the SEC. References to our corporate website address (www.stellarbiotechnologies.com) in this Annual Report on Form 10-K are intended to be inactive textual references only, and none of the information contained on our website is part of this report or incorporated in this report by reference.
| Page 12 |
| Item 1A. | RISK FACTORS. |
Certain factors may have a material adverse effect on our business, financial conditions and results of operations. You should carefully consider the risks and uncertainties described below together with all of the other information contained in this Annual Report on Form 10-K, including our financial statements and the related notes, before deciding to invest in our common shares. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties not presently known to us or that we currently believe to be immaterial may also adversely affect our business. If any of the following risks actually occurs, our business, financial condition, results of operations and future prospects could be materially and adversely affected.
Risks Related to Our Business
We have a history of net losses and limited cash flow to sustain our operations.
We currently have limited revenue from product sales of Stellar KLH, and anticipate our planned total operating expenses will be greater than our revenues for the foreseeable future. We incurred net losses of $5.03 million in fiscal 2017, $5.03 million in fiscal 2016, and $2.84 million in fiscal 2015. As of September 30, 2017, we have an accumulated deficit of $45.4 million since inception. To date, we have not paid dividends on our common shares and do not anticipate doing so in the foreseeable future. We have historically relied upon the sale of common shares to help fund our operations and meet our obligations. Any future additional equity financing would cause dilution to current shareholders. If we do not have sufficient capital for our operations, management would be forced to reduce or discontinue our activities, which would have a negative effect on our operations and financial condition.
We depend heavily on the success and market acceptance of Stellar KLH and we may never recoup our investment into its research and development.
We have invested a significant portion of our time and financial resources into the development of Stellar KLH. We anticipate that in the near term our ability to generate revenues will depend solely on the commercial success of Stellar KLH, which depends upon its market acceptance by purchasers in the pharmaceutical market and the future market demand and medical need for products and research utilizing KLH. The degree of market acceptance of Stellar KLH depends on a number of factors including: the advantages and disadvantages of Stellar KLH as compared to other KLH proteins; our ability to educate the industry about the high quality, sustainable and traceable qualities of Stellar KLH; product efficacy; customer service; and the price and demonstrated cost-effectiveness of Stellar KLH as compared to our competitors.
Our customers face uncertainties related to regulatory approval, which could reduce the market for our products.
A primary market for our Stellar KLH products is its use as a component of active immunotherapies under development. The pharmaceutical industry is subject to significant government regulation, which varies from country to country. Many of the products being developed by our customers that utilize our Stellar KLH are not yet approved for commercial sale. Before regulatory approvals for the commercial sale of any drug is granted, it must be demonstrated through preclinical research and clinical trials to be safe and effective for its intended use in humans. The process to determine safety and efficacy, including clinical trials, is expensive, prolonged and uncertain. The time necessary to complete these processes and clinical trials, and to submit applications for regulatory approvals, is difficult to predict and is subject to numerous factors outside of our control. Such clinical trials may not be successful. Larger or later stage clinical trials may not produce the same results as earlier trials. Successful results in clinical trials may not result in regulatory approval, due to certain factors including unacceptable side effects or safety issues. If regulatory approval is granted for any drug or product that utilizes Stellar KLH, it will be subject to ongoing regulatory requirements, which include registration, manufacturing, labeling, advertising and promotion, packaging, distribution, record keeping and reporting, and storage. Manufacturing facilities, both those operated by us and by our contractors, would be subject to continual review and inspection, and failure to meet these regulatory requirements can interrupt, delay, or shut down these facilities. Previously unknown problems may result in regulatory restrictions on such products, including withdrawal from the marketplace. Delays in obtaining regulatory approvals for products developed by our customers that use Stellar KLH, or failure to obtain or maintain regulatory approvals altogether, would have a negative effect on market demand for our Stellar KLH products, and have a negative effect on our operations and financial condition.
| Page 13 |
Our business is geographically concentrated and if a catastrophic event, such as a hurricane, an earthquake or coastal flooding, were to impact our facilities, our business may be disrupted which could result in serious harm to our business, results of operations and financial condition.
Our aquaculture operations, research and manufacturing facilities, laboratory space, and executive offices are all located in Port Hueneme, California, a coastal city located along the Pacific Ocean. To date, we have conducted all of our aquaculture operations, research and manufacturing at these facilities and we currently have no active backup facilities or second sites. In January 2017, we established a wholly owned Mexican subsidiary to support our plan to establish additional aquaculture capabilities in Baja California, including the development of regional marine resources, aquaculture and raw material processing for Stellar’s KLH products. However, we do not anticipate the site to be available for manufacture and production until 2019 at the earliest. There can be no assurance that these expansion plans will result in successful development of additional sites of research and manufacturing and KLH production outside of our Port Hueneme location. If a hurricane, an earthquake or other natural disaster, including coastal flooding, or a virus affecting our limpet colony, were to impact our facilities, we may be unable to manufacture our KLH products, which would have a serious disruptive impact on our business and a material adverse effect on our results of operations and financial condition. While we carry personal property insurance, such insurance may not be adequate to compensate us for losses from any damage or interruption of our business operations resulting from a hurricane, an earthquake, coastal flooding or other catastrophic event.
Government and geopolitical changes may impede the implementation of our strategy outside the United States.
Changes in geopolitical policies of the United States, such as changes in U.S. support for existing treaty and trade relationships with other countries, may adversely impact (i) the ability or willingness of non-U.S. companies to transact business in the United States, including with Stellar (ii) regulation and trade agreements affecting U.S. companies, (iii) global stock markets (including The Nasdaq Capital Market on which our common shares are traded), and (iv) general global economic conditions. These factors are outside of our control, but may nonetheless cause us to adjust our strategy in order to compete effectively in global markets.
Our joint venture with Neovacs involves numerous risks that could adversely impact our financial results.
In May 2016, we entered into a strategic relationship with Neovacs S.A. to manufacture and sell conjugated therapeutic vaccines through a newly-formed joint venture entity in France called Neostell S.A.S. This relationship is subject to various risks that could adversely affect the value of our investments and our results of operations. These risks include the following:
| • | our interests could diverge from those of Neovacs or we may not be able to agree on ongoing manufacturing and operational activities, or on the amount, timing, or nature of further investments in Neostell; |
| • | we may experience difficulties in transferring technology to Neostell; |
| • | we may experience difficulties and delays in manufacturing and production at Neostell; |
| • | as a minority partner, our control over the operations of Neostell is limited; |
| • | Neovacs may be unable to meet its commitments to us or to Neostell, which may pose credit risks for our transactions with them; |
| • | due to differing business models or long-term business goals, we and Neovacs may not participate to the same extent on funding capital investments in Neostell; |
| • | our working capital or cash flows may be inadequate to fund increased capital requirements in Neostell; |
| • | we may experience difficulties or delays in collecting amounts due to us from Neostell and/or Neovacs due to multinational financial regulations or geopolitical forces beyond our control; and |
| • | shifts in the geopolitical landscape may result in tax, legal, or regulatory changes in the United States, France and/or the European Union, thereby necessitating amendments to the agreements with Neovacs and/or the structure of the joint venture. |
If our joint venture with Neovacs is unsuccessful, our business, results of operations, or financial condition may be materially adversely affected.
| Page 14 |
Our expansion plans include the design and development of aquaculture infrastructure and KLH production in Mexico which presents substantial risks to our business and personnel. We may never recoup our investment into this location.
We plan to establish additional aquaculture capabilities in Baja California, including the development of regional marine resources, aquaculture and raw material processing for Stellar’s KLH products. There are certain administrative, legal, governmental and societal risks to operating in Mexico that could adversely impact our ability to expand our operations there. Any one or more of the risks that could adversely affect our ability to successfully implement our expansion and therefore ultimately have a material adverse effect on our business, financial condition and results of operations include, without limitation:
| • | geopolitical factors could adversely impact the ongoing relationship between the United States and Mexico and/or the continuity of the North American Free Trade Agreement, or NAFTA, in its present form; |
| • | regional political and economic instability; |
| • | ability to hire and maintain a significant work force; |
| • | burdensome and evolving government regulations; |
| • | cooperation of various departments of the Mexican government in issuing permits, and inspecting our operations on a timely basis; |
| • | providing adequate security for our employees; and |
| • | change in the value of the Mexican peso. |
In addition, our international operations are governed by the U.S. Foreign Corrupt Practices Act and similar anti-corruption laws outside the U.S. Global enforcement of anti-corruption laws has increased substantially in recent years, with more enforcement proceedings by U.S. and foreign governmental agencies and the imposition of significant fines and penalties. While we have implemented policies and procedures to enhance compliance with these laws, our international operations create the risk that there may be unauthorized payments or offers of payments made by employees, consultants, sales agents or distributors. Any alleged or actual violations of these laws may subject us to government investigations, significant criminal or civil sanctions and other liabilities, and negatively affect our reputation.
Our sales in international markets subject us to foreign currency exchange and other risks and costs, which could harm our business.
Substantial portions of our revenues are derived from outside the United States; primarily from Europe and Asia. We anticipate that revenues from international customers will continue to represent a substantial portion of our revenues for the foreseeable future. All our revenues are generated in U.S. dollars. However, if the effective price of our products were to increase as a result of fluctuations in foreign currency exchange rates, demand for our products could decline and adversely affect our results of operations and financial condition.
We compete with other companies in KLH production and manufacturing that may have greater resources than we do.
The immunotherapy industry is rapidly evolving and new competitors with competing technologies and products are regularly entering the market. Our Stellar KLH products are similar to KLH-based products produced by other companies. While we believe we are the only company that offers GMP grade KLH supported by fully traceable manufacturing methods, we may not be able to maintain our competitive position against current and potential competitors. We compete directly with Biosyn Corporation, a pharmaceutical and biotechnology company which manufactures KLH starting material and offers clinical and research grade KLH products. We also compete directly with SAFC, a division of Sigma-Aldrich, which offers clinical and research grade KLH products. Some of our competitors, both public and private, have greater financial and personnel resources than us, and have greater sales and marketing experience in the industry than us. If they are able to produce and sell comparable KLH products for less than us, it will have a negative effect on our operations and financial position. In addition to competition from current suppliers of KLH, we also face indirect competition from developers of other carrier proteins, adjuvants or therapeutic vaccine platforms. We are unable to predict what effect evolution of the KLH and immunotherapy industries and potential new entrants may have on price, selling strategies, intellectual property or our competitive position.
| Page 15 |
We may not be able to meet demand for KLH from either internally raised or ocean harvest sources.
We are dependent upon a supply of Giant Keyhole Limpets (Megathura crenulata) for KLH production. The range of the Giant Keyhole Limpet in the wild is limited, and due to the lack of a regulated harvest, the wild stocks of Giant Keyhole Limpets are believed to be declining. If the wild stocks are depleted, and our hatchery and aquaculture operations are unable to produce sufficient supplies of captive Giant Keyhole Limpets to meet demand, it would have a negative effect on our operations and financial condition.
We may not be able to manufacture our products in commercial quantities and currently depend on third parties for certain steps in our manufacturing operations, which could prevent us from marketing our products.
The manufacture of pharmaceutical starting materials like KLH requires significant expertise, including the development of advanced manufacturing techniques and process controls. We may encounter difficulties in production, particularly in scaling up production. These problems include difficulties with production costs and yields, quality control, including stability of the product and quality assurance testing, shortages of qualified personnel, as well as compliance with federal, state and foreign regulations.
In addition, we contract with third party vendors, including contract manufacturing organizations and contract testing organizations, for certain steps in the manufacture and testing of our products, and may be unable to establish and maintain relationships with qualified vendors in order to produce sufficient supplies of our finished products.
We are currently dependent upon a small number of contractors and locations for certain steps in our manufacturing operations, namely product release testing. We do not currently have backup manufacturing capacity for some of our key products. If we are unable to retain our current contractors, or are unable to obtain new contractors to provide manufacturing services in a timely manner and on similar terms, it will have a negative effect on our operations. Further, these contract manufacturers and testing organizations provide services to many biotechnology and research companies, and such third party contractors may not provide acceptable quality, quantity or costs required by us. In addition, they may not be able to provide the services required on a schedule acceptable to us. These issues may result in us being unable to manufacture our products in the required quantities or at an acceptable cost, which would have a negative effect on our operations and financial condition.
We have been, and expect to continue to be in the future, significantly dependent on collaboration and supply agreements for the development and sales of Stellar KLH.
In conducting our research and development and commercialization activities, we currently rely, and expect to continue to rely, on collaboration and supply agreements with third parties, such as contract research organizations, commercial partners, universities, governmental agencies and not-for-profit organizations, for strategic, technological, and financial resources. The inability to secure agreements on acceptable terms, the termination of these relationships, changes in our strategy or development plans or those of third parties, or failure to perform by us or third parties who are subject to regulatory, competitive and other risks, under their respective agreements or arrangements with us, would substantially disrupt or delay our research and development and commercialization activities, including anticipated commercial sales. Any such loss would likely increase our expenses and materially harm our business, financial condition and results of operation.
We have limited marketing, sales and distribution experience and capabilities. We will need to establish sales and marketing capabilities or enter into agreements with third parties to market and sell our products.
We currently have limited experience in the marketing, sales and distribution of KLH-based therapeutic or diagnostic products. Depending on market acceptance of our Stellar KLH products, we may need to expand our capabilities. We may not be able to establish such additional capabilities in-house, and then will need to enter into agreements with third parties to successfully perform these tasks. If we contract or make arrangements with third parties for the sales and marketing of our products, our revenues will be dependent on the efforts of these third parties, whose efforts may not be successful. If we market any of our products directly, we must either internally develop or acquire a marketing and sales force, which would require substantial resources and management attention.
| Page 16 |
We rely on the significant experience and specialized expertise of our Chief Executive Officer and other members of our senior management team, and we will need to hire and retain other highly skilled personnel to maintain and grow our business.
Our ability to be successful in the highly competitive biotechnology and pharmaceutical industries depends in large part upon our ability to attract and retain highly qualified managerial, scientific, medical, sales and other personnel. Our performance is substantially dependent on the research and development and business development expertise of Frank Oakes, our President and Chief Executive Officer, and other executive officers. We do not have employment agreements currently in effect with Mr. Oakes and other executive officers, and they are free to leave their employment with us at any time.
There is little possibility that this dependence will decrease in the near term. The loss of the services of Mr. Oakes, or the increased demands placed on our key executives and personnel by our continued growth, could adversely affect our financial performance and our ability to execute our strategies. Our continued success also depends on our ability to attract and retain qualified team members to meet our future growth needs. We may not be able to attract and retain necessary team members to operate our business.
In addition, our future success depends on our ability to identify, attract, hire, train, retain and motivate highly skilled technical, managerial and research personnel in all areas within our organization. We plan to continue to grow our business and will need to hire additional personnel to support this growth. We believe that there are only a limited number of individuals with the requisite skills to serve in many of our key positions, and we compete for key personnel with other biotechnology companies, as well as universities and research institutions. It is often difficult to hire and retain these persons, and we may be unable to timely replace key persons if they leave or be unable to fill new positions, as they become available, requiring key persons with appropriate experience. If we fail to attract, integrate and retain the necessary personnel, our ability to maintain and grow our business could suffer significantly.
We are subject to the risk of product liability claims, for which we may not have, or be able to obtain, adequate insurance coverage.
The pharmaceutical industry is subject to product liability claims in the event of adverse effects, even in respect to products that have received regulatory approval for commercial sale. Such claims might be made directly by consumers, healthcare providers or by pharmaceutical companies, or others selling or utilizing our Stellar KLH products. Although we currently maintain liability insurance for our products, we may not be able to obtain or maintain sufficient and affordable insurance coverage for all claims that may occur. The cost of any product liability litigation or other proceeding, even if resolved in our favor, could be substantial. In addition, our inability to obtain or maintain sufficient insurance coverage at an acceptable cost or to otherwise protect against potential product liability claims could prevent or inhibit the development and commercial production and sale of our products, which could adversely affect our business, financial condition and results of operations.
Our activities are subject to regulation in the United States and in the foreign jurisdictions in which we operate. Failure to comply with applicable laws and regulations could adversely impact our operations.
Our operations, including our aquaculture and harvesting activities, and our production activities, are subject to regulation at the local, state and federal levels in the United States by a number of regulatory agencies including, but not limited to, the U.S. Food and Drug Administration, the U.S. Environmental Protection Agency, the U.S. Fish and Wildlife Service, the U.S. Secretary of the Navy, The Regional Water Quality Control Board, the California Department of Fish and Wildlife, and similar foreign agencies. In addition to regulations in the United States, we may be subject to a variety of foreign regulations related to research, manufacturing, and the commercial sales and distribution of our products, to the extent we choose to manufacture, sell or distribute any products outside of the United States. If we are unable to comply with laws and regulations in the United States and elsewhere, our operations could be restricted, or sanctions could be imposed on us, if we are found to not be in compliance with any such regulation.
We may face environmental risks related to handling regulated substances and hazardous materials.
Our research and clinical development activities, as well as the manufacture of materials and products, are subject to federal, state and local laws, rules, regulations and policies governing the use, generation, manufacture, storage, air emission, effluent discharge, handling and disposal of certain materials, biological specimens and wastes. We may be required to incur significant costs to comply with environmental and health and safety regulations in the future. Our research and clinical development, both now and in the future, may involve the controlled use of hazardous materials, including but not limited to certain hazardous chemicals. We cannot completely eliminate the risk of accidental contamination or injury from these materials. In the event of such an occurrence, we could be held liable for any damages that result and any such liability could exceed our resources.
| Page 17 |
We deal with hazardous materials and must comply with environmental, health and safety laws and regulations, which can be expensive and restrict how we do business and/or give rise to significant liabilities.
As we operate a manufacturing facility, we are subject to various environmental, health and safety laws and regulations, including those governing air emissions, water and wastewater discharges, noise emissions, the use, management and disposal of hazardous materials and wastes, and the cleanup of contaminated sites. The cost of compliance with these laws and regulations could be significant. In the event of a violation of these requirements, including from accidental contamination or injury, we could be held liable for damages exceeding our available financial resources. We could be subject to monetary fines, penalties or third party damage claims as a result of violations of such laws and regulations or noncompliance with environmental permits required at our facility. As an operator of real property and a generator of hazardous materials and wastes, we also could be subject to environmental cleanup liability, in some cases without regard to fault or whether we were aware of the conditions giving rise to such liability. In addition, we may be subject to liability and may be required to comply with new or existing environmental laws regulating pharmaceuticals in the environment. Environmental laws or regulations (or their interpretation) may become more stringent in the future. If any such future revisions require significant changes in our operations, or if we engage in the development and manufacturing of new products or otherwise expand our operations requiring new or different environmental controls, we will have to dedicate additional management resources and incur additional expenses to comply with such laws and regulations.
In the event of an accident, applicable authorities may curtail our use of hazardous materials and interrupt our business operations. In addition, with respect to our manufacturing facility, we may incur substantial costs to comply with environmental regulations and may become subject to the risk of accidental contamination or injury from the use of hazardous materials in our manufacturing process.
Risks Related to Intellectual Property
The inability to protect our intellectual property rights could result in competitive harm to our Company.
Our success and ability to maintain our competitive position depends on our ability to protect our intellectual property, including by obtaining patent protection in the United States and other countries, or through protection of our trade secrets, including unpatented know-how, technology and other proprietary information. When appropriate, we seek to protect our proprietary position by filing patent applications in the United States and other countries. If we are unable to protect our intellectual property, whether by obtaining patents or through trade secret protection, our competitors could develop and commercialize products similar or identical to ours.
We may not have adequate remedies for any infringement or funds to take action against those infringing any of our intellectual property rights, or if our trade secrets otherwise become known or independently developed by competitors. There can be no assurance that any current or future patents held, licensed by or applied for by us will be upheld, if challenged, or that the protections afforded will not be circumvented by others. The patent positions of biotechnology and pharmaceutical companies, which often involve licensing agreements, are frequently uncertain and involve complex legal and factual questions. In addition, the coverage claimed in a patent application can be significantly reduced before the patent is issued. Consequently, our patents, patent applications and licensed rights may not provide protection against competitive technologies or may be held invalid if challenged or could be circumvented. If we enter litigation in regards to our business or to protect or enforce our patents, it may involve substantial expenditures and require significant management attention, even if we ultimately prevail.
The patent position of biotechnology companies is generally highly uncertain. The degree of patent protection we require may be unavailable or severely limited in some cases and may not adequately protect our rights, provide sufficient exclusivity, or preserve our competitive advantage. For example:
| • | we might not have been the first to invent or the first to file patent applications on the inventions covered by each of our pending patent applications; |
| • | others may independently develop similar or alternative technologies or duplicate any of our technologies; |
| • | the patents of others may have an adverse effect on our business; |
| • | any patents we obtain or license from others in the future may not encompass commercially viable products, may not provide us with any competitive advantages or may be challenged by third parties; |
| • | any patents we have obtained, will obtain or license from others in the future may not be valid or enforceable; and |
| • | we may not develop additional proprietary technologies that are patentable. |
Patents have a limited lifespan. In the United States, the natural expiration of a utility patent typically is generally 20 years after it is filed. Various extensions may be available; however, the life of a patent, and the protection it affords, is limited.
| Page 18 |
In addition, some of our technologies are not covered by any patent application and we rely instead on confidentiality agreements and trade secret law to protect such intellectual property rights. We require all of our employees and consultants to sign confidentiality agreements. The agreements also oblige our employees, and to the extent practicable, our consultants, and advisors, to assign to us ideas, developments, discoveries and inventions made by such persons in connection with their work with us. We cannot be sure that these agreements will maintain confidentiality, will prevent disclosure, or will protect our proprietary information or intellectual property, or that others will not independently develop substantially equivalent proprietary information or intellectual property.
The failure of our patents, patent applications, applicable intellectual property law or our confidentiality agreements to protect our intellectual property and other proprietary information, including our trade secrets, could have a material adverse effect on our competitive advantages and on our operations and financial position.
Changes in patent law could diminish the value of patents in general, thereby impairing our ability to protect our products and our technologies. There are numerous recent changes to the U.S. patent laws and proposed changes to the rules of the United States Patent and Trademark Office (USPTO) that may have a significant impact on our ability to obtain and enforce intellectual property rights. In particular, the Leahy-Smith America Invents Act (Leahy-Smith Act) was adopted in September 2011. The Leahy-Smith Act includes a number of significant changes to U.S. patent law, including provisions that affect the way patent applications will be prosecuted and may also affect patent litigation. Under the Leahy-Smith Act, the United States transitioned from a “first-to-invent” system to a “first-inventor-to-file” system for patent applications filed on or after March 16, 2013. With respect to patent applications filed on or after March 16, 2013, if we are the first to invent but not the first to file a patent application, we may not be able to fully protect our intellectual property rights and may be found to have violated the intellectual property rights of others if we continue to operate in the absence of a patent issued to us. Many of the substantive changes to patent law associated with the Leahy-Smith Act have recently become effective. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of any patents that issue, all of which could have a material adverse effect on our business and financial condition.
In addition, patent reform legislation may pass in the future that could lead to additional uncertainties and increased costs surrounding the prosecution, enforcement, and defense of patent applications and any patents we may obtain. Furthermore, the U.S. Supreme Court and the U.S. Court of Appeals for the Federal Circuit have made, and will likely continue to make, changes in how the patent laws of the United States are interpreted. Similarly, foreign courts have made, and will likely continue to make, changes in how the patent laws in their respective jurisdictions are interpreted. We cannot predict future changes in the interpretation of patent laws or changes to patent laws that might be enacted into law by United States and foreign legislative bodies. Those changes may materially affect our patents and patent applications or any patents we may obtain and our ability to obtain and enforce or defend additional patent protection in the future.
We may not be able to adequately protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on our products and technologies in all countries throughout the world would be prohibitively expensive. The requirements for patentability may differ in certain countries, particularly developing countries, and the breadth of patent claims allowed can be inconsistent. In addition, the laws of some foreign countries may not protect our intellectual property rights to the same extent as laws in the United States. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories where we have patent protection, but enforcement on infringing activities is inadequate.
We seek to protect our proprietary position by, among other methods, filing, when possible, U.S. and foreign patent applications relating to our technology, inventions and improvements that are important to our business. We have obtained patent protection for our non-lethal extraction methods of hemocyanin in the United States and other countries. We also rely on trade secrets, know-how, continuing technological innovation, and in-licensing opportunities to develop and maintain our proprietary position.
We plan to file other international patent applications directed to patentable features of our products and technologies from time to time. If patent rights are obtained in foreign jurisdictions, proceedings to enforce such rights could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly, could put our pending patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Thus, we may not be able to stop a competitor from marketing and selling in foreign countries products that are the same as or similar to our product.
| Page 19 |
We may become involved in lawsuits to protect or enforce our patents and patent applications, any patents that may be issued to us or other intellectual property, which could be expensive, time consuming and unsuccessful.
Competitors may infringe our patents or patent applications, or other of our intellectual property. To counter infringement or unauthorized use, we may be required to file infringement or misappropriation claims, which can be expensive and time consuming. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us alleging that we infringe their patents or claiming that our patents are invalid or unenforceable. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, non-enablement or lack of statutory subject matter. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant material information from the USPTO, or made a materially misleading statement, during prosecution. Third parties may also raise similar validity claims before the USPTO in post-grant proceedings such as ex parte reexaminations, inter partes review, or post-grant review, or oppositions or similar proceedings outside the United States, in parallel with litigation or even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable. We cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution. For any patents and patent applications we may license, we may have limited or no right to participate in the defense of any such patents against challenge by a third party. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of any future patent protection on our products. Such a loss of patent protection could harm our business. In addition, in a patent infringement proceeding, a court may decide that our patent applications or patents, if issued, are invalid or unenforceable, in whole or in part, construe the patent’s claims narrowly, or refuse to stop the other party from using the technology at issue on the grounds that our patent applications do not cover the technology. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated or interpreted narrowly.
Our trade secrets are difficult to protect and misappropriation could reduce the market for our products.
We may not be able to obtain adequate remedies for the unauthorized use or disclosure of our proprietary information, including our trade secrets. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time consuming, and the outcome is unpredictable. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they communicate it, from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor, our competitive position could be harmed.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain and could have a material adverse effect on the success of our business.
Our success depends, in part, on our ability to operate without infringing the patents and other proprietary intellectual property rights of third parties. This is generally referred to as having the “freedom to operate.” The biotechnology and pharmaceutical industries are characterized by extensive litigation regarding patents and other intellectual property rights. The defense and prosecution of intellectual property claims, interference proceedings and related legal and administrative proceedings, both in the United States and internationally, involve complex legal and factual questions. As a result, such proceedings are lengthy, costly and time-consuming, and their outcome is highly uncertain. We may become involved in protracted and expensive litigation in order to determine the enforceability, scope and validity of the proprietary rights of others, or to determine whether we have the freedom to operate with respect to the intellectual property rights of others.
Patent applications in the United States are, in most cases, maintained in secrecy until approximately 18 months after the patent application is filed. The publication of discoveries in the scientific or patent literature frequently occurs substantially later than the date on which the underlying discoveries were made. Therefore, patent applications relating to a product or method similar to ours may have already been filed by others without our knowledge. In the event that a third party has also filed a patent application covering our products, methods or other claims, we may have to participate in an adversarial proceeding, such as an interference or derivation proceeding in the USPTO or similar proceedings in other countries, to determine the priority of invention. In the event an infringement claim is brought against us, we may be required to pay substantial legal fees and other expenses to defend such a claim and, if we are unsuccessful in defending the claim, we may be subject to injunctions or damage awards.
In the future, the USPTO or a foreign patent office may grant patent rights to our claims to third parties. Subject to the issuance of these future patents, the claims of which will be unknown until issued, we may need to obtain a license or sublicense to these rights in order to have the appropriate freedom to further use, develop or commercialize such products or methods. Any required licenses may not be available to us on acceptable terms, if at all. If it is determined that we have infringed an issued patent and do not have the freedom to operate, we could be subject to injunctions, and compelled to pay significant damages, including punitive damages, which could harm our business.
| Page 20 |
We may become involved in lawsuits to protect or enforce our patents and patent applications, any patents that may be issued to us or other intellectual property, which could be expensive, time consuming and unsuccessful.
If we become involved in any patent litigation or other legal proceedings, we could incur substantial expense, and the efforts of our technical and management personnel could be significantly diverted. A negative outcome of such litigation or proceedings may expose us to the loss of our proprietary position or to significant liabilities, or require us to seek licenses that may not be available from third parties on commercially acceptable terms, if at all. We may be restricted or prevented from using or developing methods, or manufacturing and selling our products in the event of an adverse determination in a judicial or an administrative proceeding, or if we fail to obtain necessary licenses. Further, even if we are successful in defending against claims of infringement, such litigation could be burdensome and costly, and divert management’s attention away from executing our business plan.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties.
Certain of our employees were previously employed at other biotechnology or pharmaceutical companies. We may be subject to claims that we or our employees, consultants or independent contractors have inadvertently or otherwise improperly used or disclosed confidential information of these third parties or our employees’ former employers. We may also be subject to claims that former employees, consultants, independent contractors or other third parties have an ownership interest in our patents or other intellectual property. Litigation may be necessary to defend against these and other claims challenging our right to and use of confidential and proprietary information. If we fail in defending any such claims, we may lose our rights to such information, in addition to paying monetary damages. Such an outcome could have a material adverse effect on our business. Even if we are successful in defending against these claims, litigation could result in substantial cost and be a distraction to our management and employees.
Risks Related to Ownership of Our Securities
The price of our common shares may be subject to substantial volatility.
Although our common shares are listed on The Nasdaq Capital Market in the United States, there can be no assurance that an active public market will be sustained for our common shares. If there is a thin trading market or “float” for our common shares, the market price for our common shares may fluctuate significantly more than the stock market as a whole. Without a large float, our common shares would be less liquid than the stock of companies with broader public ownership and, as a result, the trading price of our common shares may be more volatile.
Furthermore, the stock market is subject to significant price and volume fluctuations, and the price of our common shares has been in the past, and may continue in the future to be subject to wide fluctuations in response to several factors, including:
| • | our quarterly or annual operating results; |
| • | our cash and cash equivalents position; |
| • | changes in our earnings estimates; |
| • | investment recommendations by securities analysts following our business or our industry; |
| • | additions or departures of key personnel; |
| • | changes in the business, earnings estimates or market perceptions of our competitors; |
| • | our failure to achieve operating results consistent with securities analysts’ projections; |
| • | announcements or the expectation of raising additional financing; |
| • | sales of our common shares by us, our insiders or other shareholders; |
| • | the status of our listing on the Nasdaq; |
| • | changes in industry, general market or economic conditions; and |
| • | announcements of legislative or regulatory changes in the United States and in other countries where we transact business. |
| Page 21 |
The stock markets in general, and the small-cap biotech market, in particular, have experienced extreme price and volume fluctuations in recent years that have significantly affected the quoted prices of the securities of many companies, including companies in our industry. The changes often appear to occur without regard to specific operating performance. The price of our common shares could fluctuate based upon factors that have little or nothing to do with our company and these fluctuations could materially reduce our share price.
Our common shares are thinly traded and there may not be an active, liquid trading market for our common shares.
There is no guarantee that an active trading market for our common shares will be maintained on Nasdaq, or that the volume of trading will be sufficient to allow for timely trades. Investors may not be able to sell our common shares quickly or at the latest market price if trading in our shares is not active or if trading volume is limited. In addition, if trading volume in our common shares is limited, trades of relatively small numbers of shares may have a disproportionate effect on the market price of our common shares.
If we cannot meet Nasdaq’s continuing listing requirements and Nasdaq rules, Nasdaq may delist our securities, which could negatively affect our company and the price of our securities.
Although our shares are currently listed on Nasdaq, in the future, we may not be able to meet the continued listing requirements of Nasdaq, which require, among other things, a minimum bid price of $1.00 per share for common shares listed on the exchange. If we are unable to satisfy the Nasdaq criteria for maintaining our listing, our securities could be subject to delisting. Trading of our securities may still be eligible for an over-the-counter market or electronic bulletin board. As a consequence of any such delisting, our shareholders would likely find it more difficult to dispose of, or to obtain accurate quotations as to the prices of our securities.
We may require additional financing or financings, which would result in substantial dilution to existing shareholders.
While the Company plans to finance company operations for the next twelve months with cash on hand and product sales, management expects to continue incurring losses for the foreseeable future and will need to raise additional capital to pursue our business plan beyond December 2018. In addition, we may decide to expand operations, undertake strategic acquisitions or determine some other business need. Financing could include debt and/or equity financings, including transactions with strategic customers and partners that may include debt and/or equity arrangements. Such sources of financing may not be available on acceptable terms, if at all. Failure to obtain such financing may cause us to curtail operations and/or result in delay or indefinite postponement of research and development of our Stellar KLH, expansion initiatives, capital expenditures and other operational priorities. Any transaction involving the issuance of previously authorized but unissued common shares, or securities convertible into common shares, could result in dilution, possibly substantial, to present and prospective holders of common shares and may be on terms less favorable to us.
We could be deemed a “passive foreign investment company” in the future, which could have negative consequences for U.S. investors.
We would be designated as a “passive foreign investment company”, or a PFIC, under the meaning of Section 1297 of the United States Internal Revenue Code of 1986, as amended, or the Code, if (a) 75% or more of our gross income is “passive income” (generally, dividends, interest, rents, royalties and gains from the disposition of assets producing passive income) in any taxable year, or (b) at least 50% of the average value of our assets produce, or are held for the production of, passive income. If we are designated a PFIC for any taxable year during which a U.S. shareholder holds our common shares, it would likely result in materially adverse U.S. federal income tax consequences for such U.S. shareholder, including, but not limited to, any gain from the sale of our common shares would be taxed as ordinary income, as opposed to capital gain, and such gain and certain distributions on our common shares would be subject to an interest charge, except in certain circumstances. In addition, U.S. shareholders should be aware that there can be no assurances that we would be able to satisfy the record keeping requirements that apply to a PFIC, or that we would supply U.S. shareholders with the information that such U.S. shareholders require to make certain elections available under the Code that are intended to mitigate the adverse tax consequences of the PFIC rules. The PFIC rules are extremely complex. A U.S. shareholder of our common shares is encouraged to consult a tax advisor regarding the PFIC rules and the U.S. federal income tax consequences of the acquisition, ownership and disposition of our common shares.
| Page 22 |
We are governed by the corporate laws in British Columbia, Canada which in some cases have a different effect on shareholders than the corporate laws in Delaware.
The material differences between the British Columbia Business Corporations Act (BCBCA) as compared to the Delaware General Corporation Law (DGCL) which may be of most interest to shareholders include the following: (i) for material corporate transactions (such as amalgamations, other extraordinary corporate transactions, amendments to the notice of articles and amendments to the Articles), the BCBCA generally requires a two-thirds majority vote by shareholders (and, in addition, especially where the holders of a class of shares are being affected differently from others, approval will be required by holders of two-thirds of the shares of such class voting in a meeting called for that purpose), whereas the DGCL generally only requires a majority vote of shareholders for similar material corporate transactions; (ii) quorum for shareholders meetings is not prescribed under the BCBCA and is 33-1/3% under our Articles (to assure compliance with Nasdaq corporate governance requirements); whereas, under the DGCL, quorum requires the holders of a majority of the shares entitled to vote to be present; and (iii) our Articles require a two-thirds majority vote of shareholders to pass a resolution for one or more directors to be removed, whereas the DGCL requires only the affirmative vote of a majority of the shareholders. Accordingly, certain provisions of our corporate governance under the laws of British Columbia may be disadvantageous to our shareholders.
Risks Related to an Emerging Growth Company
We are an “emerging growth company” under the JOBS Act of 2012 and we cannot be certain if the reduced disclosure requirements applicable to emerging growth companies will make our common shares less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012 (JOBS Act), and as a result, we may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging growth companies.” We may take advantage of these reporting exemptions until we are no longer an “emerging growth company.” We will remain an “emerging growth company” until the earlier of: (i) the last day of the fiscal year (a) following the fifth anniversary of the date of our first sale of common equity securities pursuant to an effective registration statement under the Securities Act of 1933, (b) in which we have more than $1.07 billion in annual revenues ($1.0 billion threshold adjusted for inflation effective April 2017), or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common shares that is held by non-affiliates exceeded $700 million as of the prior March 31st and (ii) the date on which we have issued more than $1.0 billion in non-convertible debt securities during the prior three-year period. While we became a reporting company following the effectiveness of our Form 20-F, filed with the Securities and Exchange Commission on February 3, 2012, our first sale of common equity securities pursuant to an effective registration statement under the Securities Act of 1933 was July 6, 2016. We may choose to take advantage of some but not all of these reduced reporting burdens.
For so long as we remain an emerging growth company, we will not be required to:
| • | have an auditor report on our internal control over financial reporting pursuant to Section 404(b) of Sarbanes-Oxley; |
| • | comply with any requirement that may be adopted by the Public Company Accounting Oversight Board (PCAOB), regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements (auditor discussion and analysis); |
| • | submit certain executive compensation matters to shareholders advisory votes pursuant to the “say on frequency” and “say on pay” provisions (requiring a non-binding shareholder vote to approve compensation of certain executive officers) and the “say on golden parachute” provisions (requiring a non-binding shareholder vote to approve golden parachute arrangements for certain executive officers in connection with mergers and certain other business combinations) of the Dodd-Frank Wall Street Reform and Consumer Protection Act of 2010; and |
| • | include detailed compensation discussion and analysis in our filings under the Exchange Act, and, instead, may provide a reduced level of disclosure concerning executive compensation. |
In addition, Section 107 of the JOBS Act also provides that an “emerging growth company” can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected not to take advantage of the extended transition period for complying with new or revised accounting standards.
If we take advantage of any of these reduced reporting burdens in future filings, the information that we provide our security holders may be different than information such security holders might receive from other public companies in which they hold equity interests. We cannot predict if investors will find our common shares less attractive because we may rely on these exemptions. If some investors find our common shares less attractive as a result, there may be a less active trading market for our common shares and our share price may be more volatile.
| Page 23 |
| Item 1B. | UNRESOLVED STAFF COMMENTS. |
None.
| Item 2. | PROPERTIES. |
We currently lease 4,300 square feet of executive office and laboratory space in Port Hueneme, California under a lease which was renewed in July 2016 for a two-year term, with options to renew for three successive two-year terms.
Our aquaculture and KLH manufacturing operations are located on approximately 37,000 square feet of oceanfront land in the Port Hueneme Aquaculture Business Park. Our facilities here include specialized aquaculture infrastructure, seawater supply and discharge systems, laboratories, manufacturing and administrative offices. We have two sublease agreements which expire in September and October 2020, respectively, with options to extend the leases for two additional five-year terms.
We also currently lease undeveloped land in Baja California, Mexico under a lease agreement which we entered into in June 2015, with a three-year term, which lease agreement is terminable at will at any time with 30 days prior notice by either party. We are utilizing the undeveloped land to conduct suitability studies for the potential development of an additional aquaculture locale and future expansion of production. We also have a short-term lease for office space in a business center located in Ensenada, Baja California. This office serves as the administrative headquarters of our BioEstelar subsidiary.
| Item 3. | LEGAL PROCEEDINGS. |
From time to time, we may be involved in legal proceedings, claims and litigation arising in the ordinary course of business, including contract disputes, employment matters and intellectual property disputes. We are not currently a party to any material legal proceedings or claims outside the ordinary course of business. Regardless of outcome, litigation can have an adverse impact on us because of defense and settlement costs, diversion of management resources and other factors.
| Item 4. | MINE SAFETY DISCLOSURES. |
Not applicable.
| Page 24 |
| Item 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES. |
Market Information
Our common shares trade on The Nasdaq Capital Market in the United States under the symbol “SBOT” since November 5, 2015.
From January 15, 2013 through November 4, 2015, our common shares were traded in the United States on the U.S. OTCQB Marketplace Exchange under the symbol “SBOTF.” From April 19, 2010 to April 8, 2016 our common shares traded on the TSX Venture Exchange in Canada under the symbol “KLH.”
The table below lists the high and low closing prices for our common shares for each fiscal quarter during 2017 and 2016 as reported by Nasdaq, Inc. or OTC Markets Group, Inc., as applicable. The OTC quotations reflect inter-dealer prices, without retail mark-up, mark-down or commission, and may not necessarily represent actual transactions.
Common Shares Trading Activity
Nasdaq Capital Market and OTCQB Marketplace
| US Dollars | ||||||||
| Period | High | Low | ||||||
| Fiscal Year 2017 | ||||||||
| Fourth Quarter Ended 9/30/17 | $ | 1.44 | $ | 1.11 | ||||
| Third Quarter Ended 6/30/17 | $ | 1.64 | $ | 1.12 | ||||
| Second Quarter Ended 3/31/17 | $ | 2.16 | $ | 1.53 | ||||
| First Quarter Ended 12/31/16 | $ | 2.50 | $ | 1.87 | ||||
| Fiscal Year 2016 | ||||||||
| Fourth Quarter Ended 9/30/16 | $ | 3.82 | $ | 2.13 | ||||
| Third Quarter Ended 6/30/16 | $ | 4.70 | $ | 2.44 | ||||
| Second Quarter Ended 3/31/16 | $ | 6.85 | $ | 4.81 | ||||
| First Quarter Ended 12/31/15 (after 11/4/15) | $ | 9.41 | $ | 6.49 | ||||
| First Quarter Ended 12/31/15 (through 11/4/15) * | $ | 8.56 | $ | 6.75 | ||||
* OTC quotations | ||||||||
Holders
As of November 30, 2017, we had 10,520,096 common shares outstanding, with 19 shareholders of record. The number of record shareholders was determined from the records of our stock transfer agent and does not reflect persons or entities that hold their shares in nominee or “street” name through various brokerage firms.
Securities Authorized for Issuance Under Equity Compensation Plans
See Item 12 “Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters” of this report.
Dividends
We have not declared any dividends on our common shares since our incorporation and do not anticipate that we will do so in the foreseeable future. Our present policy is to retain future earnings, if any, for use in our operations and the expansion of our business.
| Page 25 |
Performance Graph
The following performance graph and related information shall not be deemed to be “soliciting material” or to be “filed” with the SEC, nor shall such information be incorporated by reference into any future filing under the Securities Act, except to the extent that we specifically incorporate it by reference into such filing.
The graph set forth below compares the cumulative total return of our common shares to the Nasdaq Composite Index and the Nasdaq Biotechnology Index based on the period from August 31, 2012 through the Company’s fiscal year end on September 30, 2017. The graph assumes $100 was invested on August 31, 2012 in our common shares and in each of the comparative indices and assumes reinvestment of dividends, if any.
The comparisons shown in the graph below are based on historical data. We caution that the stock price performance showing in the graph below is not necessarily indicative of, nor is it intended to forecast, the potential future performance of our common shares. Information used in the graph was obtained from S&P Capital IQ, a source believed to be reliable, but we are not responsible for any errors or omissions in such information. Please also note that, due to the fact that the graph begins in August and includes a transition period resulting from a change in fiscal year-end, the horizontal segments of the graph do not represent equal time intervals.
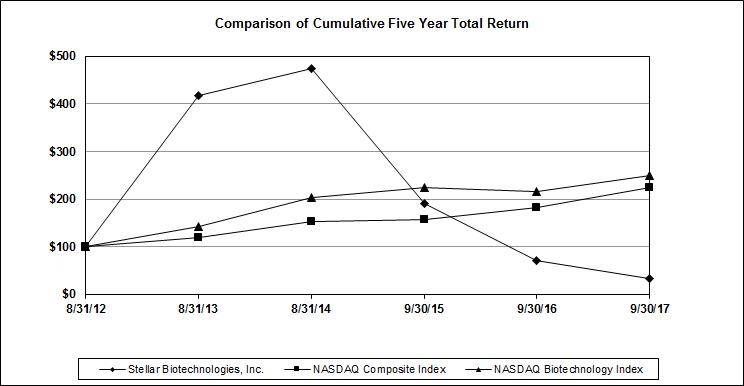
Prepared by S&P Global Market Intelligence
| Base | Years Ending | |||||||||||||||||||||||
| Period | ||||||||||||||||||||||||
| Company / Index | 8/31/12 | 8/31/13 | 8/31/14 | 9/30/15 | 9/30/16 | 9/30/17 | ||||||||||||||||||
| Stellar Biotechnologies, Inc. | $ | 100 | $ | 417.91 | $ | 474.63 | $ | 191.04 | $ | 71.64 | $ | 33.87 | ||||||||||||
| NASDAQ Composite Index | $ | 100 | $ | 118.75 | $ | 153.37 | $ | 156.60 | $ | 182.32 | $ | 225.50 | ||||||||||||
| NASDAQ Biotechnology Index | $ | 100 | $ | 142.71 | $ | 204.08 | $ | 224.20 | $ | 215.04 | $ | 249.29 | ||||||||||||
| Page 26 |
Recent Sales of Unregistered Securities; Use of Proceeds from Registered Securities
None.
Purchase of Equity Securities by the Issuer and Affiliated Purchasers
None.
| Item 6. | SELECTED FINANCIAL DATA. |
Our selected financial data in the table below is derived from our audited financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States (US GAAP). Our auditors for the fiscal years ended September 30, 2017, 2016 and 2015 and August 31, 2014, Moss Adams LLP, conducted the audits in accordance with United States generally accepted auditing standards, and the standards of the Public Company Accounting Oversight Board. Our auditors for the fiscal year ended August 31, 2013, D&H Group LLP, conducted the audit in accordance with Canadian generally accepted auditing standards, and the standards of the Public Company Accounting Oversight Board. You should read these selected financial data together with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in Item 7 and our audited financial statements and notes thereto that are included in this Annual Report on Form 10-K.
Selected Financial Data
Expressed in U.S. dollars
| Year Ended September 30, 2017 | Year Ended September 30, 2016 | Year Ended September 30, 2015 | Year Ended August 31, 2014 | Year Ended August 31, 2013 | ||||||||||||||||
| Revenues | $ | 228,287 | $ | 1,271,689 | $ | 758,689 | $ | 372,132 | $ | 545,469 | ||||||||||
| Net loss | (5,030,648 | ) | (5,026,080 | ) | (2,843,029 | ) | (8,439,523 | ) | (14,495,779 | ) | ||||||||||
| Net loss per share | (0.49 | ) | (0.57 | ) | (0.36 | ) | (1.11 | ) | (2.81 | ) | ||||||||||
| Total assets | 7,720,005 | 12,937,804 | 10,385,927 | 14,473,962 | 8,513,358 | |||||||||||||||
| Long-term obligations | - | - | - | 5,352,663 | 6,835,199 | |||||||||||||||
Supplementary Financial Information
Selected Quarterly Financial Data
U.S. dollars are shown in thousands, except per share data
| For the Year Ended September 30, | For the Year Ended September 30, | |||||||||||||||||||||||||||||||
| 2017 | 2016 | |||||||||||||||||||||||||||||||
| Q4 | Q3 | Q2 | Q1 | Q4 | Q3 | Q2 | Q1 | |||||||||||||||||||||||||
| Revenues | $ | 3 | $ | 21 | $ | 63 | $ | 141 | $ | 277 | $ | 181 | $ | 326 | $ | 488 | ||||||||||||||||
| Net loss for period | (1,221 | ) | (1,220 | ) | (1,104 | ) | (1,486 | ) | (1,349 | ) | (1,194 | ) | (861 | ) | (1,622 | ) | ||||||||||||||||
| Net loss per share - basic and diluted | (0.11 | ) | (0.12 | ) | (0.11 | ) | (0.15 | ) | (0.14 | ) | (0.14 | ) | (0.10 | ) | (0.19 | ) | ||||||||||||||||
| Page 27 |
Fluctuations in net income (loss) between quarters can be mainly attributed to fluctuations in revenue and changes in noncash gains and losses shown in thousands as follows:
| For the Year Ended September 30, | For the Year Ended September 30, | |||||||||||||||||||||||||||||||
| 2017 | 2016 | |||||||||||||||||||||||||||||||
| Q4 | Q3 | Q2 | Q1 | Q4 | Q3 | Q2 | Q1 | |||||||||||||||||||||||||
| Loss in fair value of warrant liability | $ | - | $ | - | $ | - | $ | - | $ | - | $ | - | $ | - | $ | (212 | ) | |||||||||||||||
| Foreign exchange gain (loss) | 140 | 64 | 35 | (77 | ) | (33 | ) | (8 | ) | 227 | (109 | ) | ||||||||||||||||||||
|
|
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS. |
This discussion contains forward-looking statements that involve risks and uncertainties that could cause actual results or events to differ materially from those expressed or implied by such forward-looking statements as a result of many important factors, including those set forth in Part I of this Annual Report on Form 10-K under the caption “Risk Factors.” Please see “Special Note Regarding Forward-Looking Statements” in Part I above. We do not undertake any obligation to update forward-looking statements to reflect events or circumstances occurring after the date of this Annual Report.
Operating and Financial Review and Prospects
Overview
Our financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (GAAP) and include the accounts of the Company and our wholly-owned subsidiaries, Stellar Biotechnologies, Inc. and BioEstelar S.A. de C.V.
In the past, operations of the Company have primarily been funded by the issuance of common shares, exercise of warrants, grant revenues, contract services revenue and product sales. In July 2016, the Company closed a $6.75 million registered direct offering. Management believes the Company’s working capital is sufficient to support the Company’s operations for the next 12 months. Management also seeks to expand the customer base for existing marketed products, and is currently evaluating opportunities to secure additional financing through debt and/or equity financings, including transactions with strategic customers and partners that may include debt and/or equity arrangements.
Results of Operations
Fiscal Year Ended September 30, 2017
Our total revenues decreased by $1.04 million to $.23 million for fiscal 2017 compared to $1.27 million for fiscal 2016 primarily due to a decrease in product sales. While our customer base has not changed significantly, product sales volumes are subject to variability associated with the rate of development and progression of clinical studies of third-party products that utilize Stellar KLH. For fiscal 2017, product sales consisted of KLH for clinical and pre-clinical studies and immune system assays. For fiscal 2016, product sales primarily consisted of higher volume orders for later stage clinical studies. The rate of progression toward later stage studies is expected to continue to affect the timing and volume of future product sales
Our total expenses decreased by $.73 million to $5.45 million for fiscal 2017 compared to $6.18 million for fiscal 2016:
| · | Our costs of sales and contract services decreased by $.57 million to $.25 million for fiscal 2017 compared to $.82 million for fiscal 2016 primarily due to decreased product sales. | |
| · | Our research and development expenses increased by $.24 million to $1.97 million for fiscal 2017 compared to $1.73 million for fiscal 2016. The increase was primarily due to research and development activities intended to increase the scalability and throughput capacity of existing manufacturing systems, including additional research and development in aquaculture, both in the U.S. and for our aquaculture feasibility assessment in Baja California, Mexico; improvements in analytical, manufacturing, and purification processes; stability studies; and formulation development. | |
| · | Our general and administrative expenses decreased by $.38 million to $2.94 million for fiscal 2017 compared to $3.32 million for fiscal 2016 primarily due to management’s actions to reduce corporate expenses, including travel and professional fees, as well as lower legal fees and public company expenses. |
| Page 28 |
Our other income (loss) increased by $.31 million to an overall gain of $.19 million for fiscal 2017 compared to an overall loss of $.11 million for fiscal 2016. The increase was primarily due to a noncash change in fair value of warrant liability related to warrants with Canadian dollar exercise prices. All such warrants were exercised or expired by December 2015 and, consequently, there was no warrant liability and no gain/loss in fair value of warrant liability for fiscal 2017 compared to a loss of $.21 million for fiscal 2016. Foreign exchange gain (loss) was a gain of $.16 million for the fiscal 2017 compared to a gain of $.08 million for fiscal 2016 due to fluctuations in exchange rates and decreased amounts held in Canadian cash and cash equivalents.
Our net loss for fiscal 2017 was $5.03 million, or $0.49 per basic share, compared to a net loss of $5.03 million, or $0.57 per basic share, for fiscal 2016.
Fiscal Year Ended September 30, 2016
Our total revenues increased by $.51 million to $1.27 million for fiscal 2016 compared to $.76 million for fiscal 2015. Product sales increased by $.68 million to $1.24 million for fiscal 2016 compared to $.56 million for fiscal 2015 primarily due to an increase in the number of customers and greater product sales volume, including sales under supply agreements and custom manufactured products. Contract services revenue decreased by $.17 million to $.03 million for fiscal 2016 compared to $.20 million for fiscal 2015 as a result of the successful conclusion of a collaboration agreement in December 2015.
Our total expenses increased by $1.08 million to $6.18 million for fiscal 2016 compared to $5.10 million for fiscal 2015.
Our costs of sales and contract services increased by $.24 million to $.82 million for fiscal 2016 compared to $.58 million for fiscal 2015, due to increased product sales.
Our research and development expenses increased by $.70 million to $1.73 million fiscal 2016 compared to $1.03 million for fiscal 2015. The increase was a result of additional research and development in aquaculture, both in the U.S. and for our aquaculture feasibility assessment in Baja California, Mexico; improvements in analytical, manufacturing, and purification processes; stability studies; and formulation development.
Our general and administrative expenses increased by $.09 million to $3.32 million for fiscal 2016 compared to $3.23 million for fiscal 2015. The increase resulted from increased corporate expenses, including our Nasdaq listing fees; compensation increases; and expanded business development and investor relations activities; offset by decreases in legal fees due to the Form S-3 shelf registration statement and our transition to reporting as a U.S. domestic issuer during fiscal 2015.
Other income decreased by $1.64 million to an overall loss of $.11 million for fiscal 2016 compared to an overall gain of $1.53 million for fiscal 2015 primarily due to a noncash change in fair value of warrant liability, which fluctuated to a loss of $.21 million for fiscal 2016 compared to a gain of $2.13 million in fiscal 2015. All warrants with Canadian dollar exercise prices were exercised or expired by December 2015 and, consequently, there was no warrant liability and no gain/loss in fair value of warrant liability after that time. These fair value gains and losses occur in inverse relation to changes in our share price that affect the Black Scholes valuation model. The loss in fiscal 2016 is a result of the increase in our share price from September 30, 2015 to the exercise dates of the warrants compared to the gain in fiscal 2015 as a reflection of both the decrease in our share price from September 30, 2014 to the exercise dates of warrants during the year and the decrease in our share price from $11.90 at September 30, 2014 to $6.40 for warrants outstanding at September 30, 2015. Our foreign exchange gain in fiscal 2016 was $.08 million compared to a foreign exchange loss of $.65 million in fiscal 2015. The change over the prior year was due to improved exchange rates for our Canadian cash and cash equivalents.
Our net loss for fiscal 2016 was $5.03 million, or $0.57 per basic share, compared to a net loss of $2.84 million, or $0.36 per basic share, for fiscal 2015. The increase in net loss of approximately $2.19 million for fiscal 2016 was primarily due to significant fluctuations in non-cash gain/loss in fair value of warrant liability and non-cash foreign exchange gain/loss, as well as increased research and development expenses, which were offset by increased product sales.
Capital Expenditures
Our capital expenditures, which primarily consist of scientific, manufacturing, and aquaculture equipment, and facility leasehold improvements for the previous three fiscal years are as follows:
| 2017 | $ | 302,733 | ||
| 2016 | 402,271 | |||
| 2015 | 274,589 |
Capital expenditures include $145,318 of construction in progress, primarily for aquaculture site improvements and installation of lab equipment.
| Page 29 |
Liquidity and Capital Resources
Company operations have historically been funded by the issuance of common shares, exercise of warrants, grant revenues, contract services revenue and product sales. For the fiscal years 2017, 2016, and 2015, the Company reported net losses of approximately $5.0 million, $5.0 million, and $2.8 million, respectively. As of September 30, 2017, the Company had an accumulated deficit of approximately $45.4 million and working capital of approximately $6.4 million. While the Company plans to finance company operations for the next twelve months with cash on hand and product sales, management expects to continue incurring losses for the foreseeable future and will need to raise additional capital to pursue our business plan beyond December 2018. Management is taking action to ensure the Company will continue as a going concern for at least one year beyond the date of the issuance of the Company’s financial statements. First, management has flexibility to adjust planned expenditures based on a number of factors including the size and timing of capital expenditures, staffing levels, inventory levels, and the status of customer clinical trials. Management also seeks to expand the customer base for existing marketed products, and is currently evaluating opportunities to secure additional financing through debt and/or equity financings, including transactions with strategic customers and partners that may include debt and/or equity arrangements. We have not secured any commitment for new financing at this time, nor can we provide any assurance that new financing will be available on commercially acceptable terms, if needed.
In July 2016, we completed a registered direct offering of an aggregate of 1,687,500 of our common shares, and a concurrent private placement of warrants to purchase up to an aggregate of 1,265,626 common shares with an exercise price of $4.50 per share, resulting in net proceeds of approximately $6 million.
We have filed with the Securities and Exchange Commission, and the Securities and Exchange Commission declared effective, a universal shelf registration statement of up to $100 million worth of registered equity securities, of which we utilized approximately $6.75 million in our July 2016 offering. Under this effective registration statement, we may issue registered securities, from time to time, in one or more separate offerings or other transactions with the size, price and terms to be determined at the time of issuance. Pursuant to General Instruction I.B.6 of Form S-3, in no event will we sell securities in a public primary offering with a value of more than one-third of the aggregate market value of our common shares held by non-affiliates in any twelve-month period, so long as the aggregate market value of our common shares held by non-affiliates remains below $75 million. Registered securities issued using our existing shelf may be used to raise additional capital to fund our working capital, R&D and other corporate needs.
Research and Development
Our core business is developing and commercializing Keyhole Limpet Hemocyanin for use in immunotherapy and immunodiagnostic applications. Our internal research has included, among other activities, continual improvement of methods for the culture and growth of Giant Keyhole Limpet, innovations in aquaculture systems and infrastructure, biophysical and biochemical characterization of the KLH molecule, analytical processes to enhance performance of our products, KLH manufacturing process improvements, new KLH formulations, and early development of potential new KLH-based immunotherapies.
Research and development costs, including materials, KLH designated for internal research use only and salaries of employees directly involved in research and development efforts, are expensed as incurred.
The following table includes our research and development costs for each of the most recent three fiscal years:
| 2017 | $ | 1,973,400 | ||
| 2016 | 1,729,445 | |||
| 2015 | 1,029,489 |
Off Balance Sheet Arrangements
We do not have any off balance sheet arrangements that have or are reasonably likely to have a current or future material effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures, or capital resources.
Disclosure of Contractual Obligations
We currently lease 4,300 square feet of executive office and laboratory space in Port Hueneme, California under a lease which was renewed in July 2016 for a two-year term, with options to renew for three successive two-year terms.
| Page 30 |
Our aquaculture and KLH manufacturing operations are located on approximately 37,000 square feet of oceanfront land in the Port Hueneme Aquaculture Business Park. Our facilities here include specialized aquaculture infrastructure, seawater supply and discharge systems, laboratories, manufacturing and administrative offices. We have two sublease agreements which expire in September and October 2020, respectively, with options to extend the leases for two additional five-year terms.
We also currently lease undeveloped land in Baja California, Mexico under a lease agreement which we entered into in June 2015, with a three-year term, which lease agreement is terminable at will at any time with 30 days prior notice by either party. We are utilizing the undeveloped land to conduct suitability studies for the potential development of an additional aquaculture locale and future expansion of production. We also have a short-term lease for office space in a business center located in Ensenada, Baja California. This office serves as the administrative headquarters of our BioEstelar subsidiary.
We have purchase commitments for contract research organizations, consultants and construction contractors.
The approximate amounts of our contractual obligations are as follows:
Contractual Obligations
As of September 30, 2017
| Less than 1 | 1-3 | 3-5 | More than | |||||||||||||||||
| Total | year | years | years | 5 years | ||||||||||||||||
| Operating lease obligations | $ | 378,000 | $ | 160,000 | $ | 212,000 | $ | 6,000 | $ | - | ||||||||||
| Purchase obligations | 252,000 | 186,900 | 65,100 | - | - | |||||||||||||||
| Total | $ | 630,000 | $ | 346,900 | $ | 277,100 | $ | 6,000 | $ | - | ||||||||||
Significant Accounting Policies and Estimates
Our consolidated financial statements, which are indexed under Item 15 of this Annual Report on Form 10-K, have been prepared in accordance with accounting principles generally accepted in the United States, which require that the management make certain assumptions and estimates and, in connection therewith, adopt certain accounting policies. Our significant accounting policies are set forth in Note 3 in the Notes to Consolidated Financial Statements. Of those policies, we believe that the policies discussed below may involve a higher degree of judgment or may otherwise be more relevant to our financial condition and results of operations.
Investments
Investments at September 30, 2017 and 2016 consisted of U.S. Treasury bills with original maturities between 13 and 52 weeks. They are classified as held-to-maturity and are reported at amortized cost, which approximates fair value. We regularly review these investments to determine whether any decline in fair value below the amortized cost basis has occurred that is other than temporary. If a decline in fair value has occurred that is determined to be other than temporary, the cost basis of the investment is written down to fair value.
Inventory
We record inventory at the lower of cost or market, with market not in excess of net realizable value. Raw materials are measured using FIFO (first-in first-out) cost. Work in process and finished goods are measured using average cost. Raw materials include inventory of manufacturing supplies. Work in process includes manufacturing supplies, direct and indirect labor, contracted manufacturing and testing, and allocated manufacturing overhead for inventory in process at the end of the year. Finished goods include products that are complete and available for sale. The Company recorded work in process and finished goods inventory only for those products with recent sales levels to evaluate net realizable value.
| Page 31 |
Warrant Liability
Our equity offerings in prior years included the issuance of warrants with exercise prices denominated in Canadian dollars. As a result of having exercise prices denominated in a currency other than our functional currency, our warrants with Canadian dollar exercise prices met the definition of derivatives and were therefore classified as derivative liabilities measured at fair value with noncash adjustments to fair value recognized through the consolidated statements of operations. Upon exercise of these warrants, the fair value of warrants included in derivative liabilities was reclassified to common shares. If these warrants expired, the related decrease in warrant liability was recognized as gain in fair value of warrant liability. There was no cash flow impact as a result of this accounting treatment. The fair value of the warrants was determined using the Black-Scholes option valuation model at the end of each reporting period.
All warrants with exercise prices denominated in Canadian dollars were exercised or expired by December 2015. Therefore, there is no outstanding warrant liability at September 30, 2017.
Revenue Recognition
Product Sales
The Company recognizes product sales when KLH product is shipped (for which the risk is typically transferred upon delivery to the shipping carrier) and there is persuasive evidence of an arrangement, the fee is fixed or determinable, and collectability is reasonably assured. The Company documents arrangements with customers with purchase orders and sales agreements.
Product sales include sales made under supply agreements with customers for a fixed price per gram of KLH products based on quantities ordered. Supply agreements are typically on a non-exclusive basis except within that customer’s field of use.
Contract services revenue
The Company recognizes contract services revenue when contract services have been performed and reasonable assurance exists regarding measurement and collectability. An appropriate amount will be recognized as revenue in the period that the Company is assured of fulfilling the contract requirements. Amounts received in advance of performance of contract services are recorded as deferred revenue.
Contract services include services performed under collaboration agreements and technology transfer and purchase agreement.
Share-Based Compensation
We grant options to buy common shares of the Company to our directors, officers, employees and consultants, and grant other equity-based instruments to non-employees.
The fair value of share-based compensation is measured on the date of grant, using the Black-Scholes option valuation model and is recognized over the vesting period net of estimated forfeitures for employees or the service period for non-employees. The Black-Scholes option valuation model requires the input of subjective assumptions, including price volatility of the underlying shares, risk-free interest rate, dividend yield, and expected life of the option.
Foreign Exchange
Items included in the financial statements of our subsidiaries are measured using the currency of the primary economic environment in which the entity operates (the functional currency). Our functional currency and the functional currency of our subsidiaries is the U.S. dollar.
Transactions in currencies other than the U.S. dollar are recorded at exchange rates prevailing on the dates of the transactions.
Recent Accounting Pronouncements
Recent accounting pronouncements are contained in Note 3 to the financial statements, which are indexed under Item 15 of this this Annual Report on Form 10-K.
| Page 32 |
CERTAIN INCOME TAX CONSIDERATIONS
United States Federal Income Taxation
As used below, a “U.S. holder” is a beneficial owner of a common share that is, for U.S. federal income tax purposes, (i) a citizen or resident alien individual of the United States, (ii) a corporation (or an entity treated as a corporation) created or organized under the law of the United States, any State thereof or the District of Columbia, (iii) an estate the income of which is subject to U.S. federal income tax without regard to its source or (iv) a trust if (1) a court within the United States is able to exercise primary supervision over the administration of the trust, and one or more United States persons have the authority to control all substantial decisions of the trust, or (2) the trust has a valid election in effect under applicable U.S. Treasury Regulations to be treated as a United States person. For purposes of this discussion, a “non-U.S. holder” is a beneficial owner of a common share that is (i) a nonresident alien individual, (ii) a corporation (or an entity treated as a corporation) created or organized in or under the law of a country other than the United States or a political subdivision thereof or (iii) an estate or trust that is not a U.S. Holder. If a partnership (including for this purpose any entity treated as a partnership for U.S. federal tax purposes) is a beneficial owner of a common share, the U.S. federal tax treatment of a partner in the partnership generally will depend on the status of the partner and the activities of the partnership. A holder of a common share that is a partnership and partners in that partnership should consult their own tax advisers regarding the U.S. federal income tax consequences of holding and disposing of common shares. We have not sought a ruling from the Internal Revenue Service (IRS) or an opinion of counsel as to any U.S. federal income tax consequence described herein. The IRS may disagree with the description herein, and its determination may be upheld by a court. This discussion does not address U.S. federal tax laws other than those pertaining to U.S. federal income taxation (such as estate or gift tax laws), nor does it address any aspects of U.S. state or local or non-U.S. taxation.
GIVEN THE COMPLEXITY OF THE TAX LAWS AND BECAUSE THE TAX CONSEQUENCES TO ANY PARTICULAR SHAREHOLDER MAY BE AFFECTED BY MATTERS NOT DISCUSSED HEREIN, SHAREHOLDERS ARE URGED TO CONSULT THEIR OWN TAX ADVISORS WITH RESPECT TO THE SPECIFIC TAX CONSEQUENCES OF THE ACQUISITION, OWNERSHIP AND DISPOSITION OF COMMON SHARES, INCLUDING THE APPLICABILITY AND EFFECT OF STATE, LOCAL AND NON-U.S. TAX LAWS, AS WELL AS U.S. FEDERAL TAX LAWS.
Taxation of Dividends
U.S. Holders. In general, subject to the passive foreign investment company rules discussed below, a distribution on a common share will constitute a dividend for U.S. federal income tax purposes to the extent that it is made from a corporation’s current or accumulated earnings and profits as determined under U.S. federal income tax principles. If a distribution exceeds the current and accumulated earnings and profits of the distributing corporation, it will generally be treated as a non-taxable reduction of basis to the extent of the U.S. holder’s tax basis in the common share on which it is paid, and to the extent it exceeds that basis it will be treated as capital gain. The Company has not and does not plan to maintain calculations of earnings and profits under U.S. federal income tax principles. Accordingly, it is unlikely that U.S. holders will be able to establish that a distribution by the Company is in excess of its current and accumulated earnings and profits (as computed under U.S. federal income tax principles). Therefore, a U.S. holder should expect that a distribution by the Company will generally be taxable in its entirety as a dividend to U.S. holders for U.S. federal income tax purposes even though the distribution may be treated in whole or in part as a non-taxable distribution for Canadian tax purposes.
The gross amount of any dividend on a common share (which will include the amount of any Canadian taxes withheld with respect to such dividend) generally will be subject to U.S. federal income tax as foreign source dividend income, and will not be eligible for the corporate dividends received deduction. The amount of a dividend paid in Canadian dollars will be its value in U.S. dollars based on the prevailing spot market exchange rate in effect on the day the U.S. holder receives the dividend. A U.S. holder will have a tax basis in any distributed Canadian dollars equal to their U.S. dollar value on the date of receipt, and any gain or loss realized on a subsequent conversion or other disposition of such Canadian dollars generally will be treated as U.S. source ordinary income or loss. If dividends paid in Canadian dollars are converted into U.S. dollars on the date they are received by a U.S. holder, the U.S. holder generally should not be required to recognize foreign currency gain or loss in respect of the dividend income.
| Page 33 |
Subject to certain exceptions for short-term and hedged positions, as well as the passive foreign investment rules, a dividend that a non-corporate holder receives on a common share will generally be subject to a maximum federal income tax rate of 20% if the dividend is a “qualified dividend.” A dividend on a common share will be a qualified dividend if (i) either (a) the common shares are readily tradable on an established market in the United States or (b) we are eligible for the benefits of a comprehensive income tax treaty with the United States that the Secretary of the Treasury determines is satisfactory for purposes of these rules and that includes an exchange of information program, and (ii) we were not, in the year prior to the year the dividend was paid, and are not, in the year the dividend is paid, a passive foreign investment company (PFIC). The common shares are listed on The Nasdaq Capital Market which should be treated as an established securities market in the United States. In any event, the U.S. Canada Income Convention (the Treaty) satisfies the requirements of clause (i)(b), the Company is incorporated in and tax resident of Canada and should be entitled to the benefits of the Treaty. Based on our audited financial statements, income tax returns and relevant market and shareholder data, we believe that we likely will not be classified as a PFIC in the September 30, 2017 taxable year. There can be no assurance, however, that the Company will not be considered to be a PFIC for any particular year in the future because PFIC status is factual in nature, depends upon factors not wholly within the Company’s control, generally cannot be determined until the close of the taxable year in question, and is determined annually. Accordingly, no assurance can be made that a dividend paid, if any, would be a “qualified dividend.” In addition, as described in the section below entitled “Passive Foreign Investment Company Rules,” if we were a PFIC in a year while a U.S. holder held a common share, and if the U.S. holder has not made a qualified electing fund election effective for the first year the U.S. holder held the common share, the common share remains an interest in a PFIC for all future years or until such an election is made. The IRS takes the position that such rule will apply for purposes of determining whether a common share is an interest in a PFIC in the year a dividend is paid or in the prior year, even if we do not satisfy the tests to be a PFIC in either of those years. Even if dividends on the common shares would otherwise be eligible for qualified dividend treatment, in order to qualify for the reduced qualified dividend tax rates, a non-corporate holder must hold the common share on which a dividend is paid for more than 60 days during the 120-day period beginning 60 days before the ex-dividend date, disregarding for this purpose any period during which the non-corporate holder has an option to sell, is under a contractual obligation to sell or has made (and not closed) a short sale of substantially identical stock or securities, is the grantor of an option to buy substantially identical stock or securities or, pursuant to U.S. Treasury regulations, has diminished such holder’s risk of loss by holding one or more other positions with respect to substantially similar or related property. In addition, to qualify for the reduced qualified dividend tax rates, the non-corporate holder must not be obligated to make related payments with respect to positions in substantially similar or related property. Payments in lieu of dividends from short sales or other similar transactions will not qualify for the reduced qualified dividend tax rates.
A non-corporate holder that receives an extraordinary dividend (generally, any dividend that is in excess of 10% of the holder's adjusted basis in the common share on which the dividend is paid) that is eligible for the reduced qualified dividend rates must treat any loss on the sale of the common share as a long-term capital loss to the extent of the dividend. For purposes of determining the amount of a non-corporate holder’s deductible investment interest expense, a dividend is treated as investment income only if the non-corporate holder elects to treat the dividend as not eligible for the reduced qualified dividend tax rates. Special limitations on foreign tax credits with respect to dividends subject to the reduced qualified dividend tax rates apply to reflect the reduced rates of tax.
The U.S. Treasury has announced its intention to promulgate rules pursuant to which non-corporate holders of stock of non-U.S. corporations, and intermediaries through which the stock is held, will be permitted to rely on certifications from issuers to establish that dividends are treated as qualified dividends. Because those procedures have not yet been issued, it is not clear whether we will be able to comply with them.
Non-corporate holders of common shares are urged to consult their own tax advisers regarding the availability of the reduced qualified dividend tax rates with respect to dividends, if any, received on the common shares in the light of their own particular circumstances.
Any Canadian withholding tax imposed on dividends received with respect to the common shares will be treated as a foreign income tax eligible for credit against a U.S. holder’s U.S. federal income tax liability, subject to generally applicable limitations under U.S. federal income tax law. For purposes of computing those limitations separately under current law for specific categories of income, a dividend generally will constitute foreign source “passive category income” or, in the case of certain holders, “general category income.” A U.S. holder will be denied a foreign tax credit with respect to Canadian income tax withheld from dividends received with respect to the common shares to the extent the U.S. holder has not held the common shares for at least 16 days of the 30-day period beginning on the date which is 15 days before the ex-dividend date or to the extent the U.S. holder is under an obligation to make related payments with respect to substantially similar or related property. Any days during which a U.S. holder has substantially diminished its risk of loss on the common shares are not counted toward meeting the 16-day holding period required by the statute. The rules relating to the determination of the foreign tax credit are complex, and U.S. holders are urged to consult with their own tax advisers to determine whether and to what extent they will be entitled to foreign tax credits as well as with respect to the determination of the foreign tax credit limitation. Alternatively, any Canadian withholding tax may be taken as a deduction against taxable income, provided the U.S. holder takes a deduction and not a credit for all foreign income taxes paid or accrued in the same taxable year. In general, special rules will apply to the calculation of foreign tax credits in respect of dividend income that is subject to preferential rates of U.S. federal income tax.
| Page 34 |
Non-U.S. Holders. A dividend paid to a non-U.S. holder of a common share will generally not be subject to U.S. federal income tax unless the dividend is effectively connected with the conduct of trade or business by the non-U.S. holder within the United States (and is attributable to a permanent establishment or fixed base the non-U.S. holder maintains in the United States if an applicable income tax treaty so requires as a condition for the non-U.S. holder to be subject to U.S. taxation on a net income basis on income from the common share). A non-U.S. holder generally will be subject to tax on an effectively connected dividend in the same manner as a U.S. holder. A corporate non-U.S. holder under certain circumstances may also be subject to an additional “branch profits tax,” the rate of which may be reduced pursuant to an applicable income tax treaty.
Taxation of Capital Gains
U.S. Holders. Subject to the passive foreign investment company rules discussed below, on a sale or other taxable disposition of a common share, a U.S. holder will recognize capital gain or loss in an amount equal to the difference between the U.S. holder’s adjusted basis in the common share and the amount realized on the sale or other disposition, each determined in U.S. dollars. Such capital gain or loss will be long-term capital gain or loss if at the time of the sale or other taxable disposition the common share has been held for more than one year. In general, any adjusted net capital gain of an individual is subject to a maximum federal income tax rate of 20%. Capital gains recognized by corporate U.S. holders generally are subject to U.S. federal income tax at the same rate as ordinary income. The deductibility of capital losses is subject to limitations.
Any gain a U.S. holder recognizes generally will be U.S. source income for U.S. foreign tax credit purposes, and, subject to certain exceptions, any loss will generally be a U.S. source loss. If a Canadian tax is paid on a sale or other disposition of a common share, the amount realized will include the gross amount of the proceeds of that sale or disposition before deduction of the Canadian tax. The generally applicable limitations under U.S. federal income tax law on crediting foreign income taxes may preclude a U.S. holder from obtaining a foreign tax credit for any Canadian tax paid on a sale or other disposition of a common share. The rules relating to the determination of the foreign tax credit are complex, and U.S. holders are urged to consult with their own tax advisers regarding the application of such rules. Alternatively, any Canadian tax paid on the sale or other disposition of a common share may be taken as a deduction against taxable income, provided the U.S. holder takes a deduction and not a credit for all foreign income taxes paid or accrued in the same taxable year.
Non-U.S. Holders. A non-U.S. holder will not be subject to U.S. federal income tax on gain recognized on a sale or other disposition of a common share unless (i) the gain is effectively connected with the conduct of trade or business by the non-U.S. holder within the United States (and is attributable to a permanent establishment or fixed base the non-U.S. holder maintains in the United States if an applicable income tax treaty so requires as a condition for the non-U.S. holder to be subject to U.S. taxation on a net income basis on income from the common share), or (ii) in the case of a non-U.S. holder who is an individual, the holder is present in the United States for 183 or more days in the taxable year of the sale or other disposition and certain other conditions apply. Any effectively connected gain of a corporate non-U.S. holder may also be subject under certain circumstances to an additional “branch profits tax,” the rate of which may be reduced pursuant to an applicable income tax treaty.
Passive Foreign Investment Company Rules
A special set of U.S. federal income tax rules applies to a foreign corporation that is a PFIC for U.S. federal income tax purposes. As noted above, based on our audited financial statements, income tax returns, and relevant market and shareholder data, we believe that we likely will not be classified as a PFIC in the September 30, 2017 taxable year. There can be no assurance, however, that the Company will not be considered to be a PFIC for any particular year in the future because PFIC status is factual in nature, depends upon factors not wholly within the Company’s control, generally cannot be determined until the close of the taxable year in question, and is determined annually.
In general, a non-US corporation is a PFIC if in any taxable year either (i) at least 75% of its gross income is “passive income” or (ii) at least 50% of the quarterly average value of its assets is attributable to assets that produce or are held to produce “passive income.” In applying these tests, the Company generally is treated as holding its proportionate share of the assets and receiving its proportionate share of the income of any other corporation in which the Company owns at least 25% by value of the shares. Passive income for this purpose generally includes dividends, interest, royalties, rent and capital gains, but generally does not include certain royalties derived in an active business. Passive assets are those assets that are held for production of passive income or do not produce income at all. Thus cash will be a passive asset. Interest, including interest on working capital, is treated as passive income for purposes of the income test. Without taking into account the value of its goodwill, more than 50% of the Company’s assets by value would be passive so that the Company would be a PFIC under the asset test. Based upon its current operations, its goodwill (the value of which should be based upon the Company’s market capitalization) will likely be attributable to its activities that will generate active income and to such extent, should be treated as an active asset. The determination of whether a foreign corporation is a PFIC is a factual determination made annually and is therefore subject to change. Subject to exceptions pursuant to certain elections that generally require the payment of tax, once stock in a foreign corporation is stock in a PFIC in the hands of a particular shareholder that is a United States person, it remains stock in a PFIC in the hands of that shareholder.
| Page 35 |
If we are treated as a PFIC, contrary to the tax consequences described in “Taxation of Dividends” and “Taxation of Capital Gains” above, a U.S. holder that does not make an election described in the succeeding two paragraphs would be subject to special rules with respect to (i) any gain realized on a sale or other disposition of a common share (for purposes of these rules, a disposition of a common share includes many transactions on which gain or loss is not realized under general U.S. federal income tax rules) and (ii) any “excess distribution” by the Company to the U.S. holder (generally, any distribution during a taxable year in which distributions to the U.S. holder on the common share exceed 125% of the average annual taxable distributions (whether actual or constructive and whether or not out of earnings and profits) the U.S. holder received on the common share during the preceding three taxable years or, if shorter, the U.S. holder’s holding period for the common share). Under those rules, (i) the gain or excess distribution would be allocated ratably over the U.S. holder’s holding period for the common share, (ii) the amount allocated to the taxable year in which the gain or excess distribution is realized would be taxable as ordinary income in its entirety and not as capital gain, would be ineligible for the reduced qualified dividend rates, and could not be offset by any deductions or losses, and (iii) the amount allocated to each prior year, with certain exceptions, would be subject to tax at the highest tax rate in effect for that year, and the interest charge generally applicable to underpayments of tax would be imposed in respect of the tax attributable to each of those years.
The special PFIC rules described above will not apply to a U.S. holder if the U.S. holder makes a timely election, which remains in effect, to treat the Company as a “qualified electing fund” (QEF) in the first taxable year in which the U.S. holder owns a common share and the Company is a PFIC and if the Company complies with certain requirements. Instead, a shareholder of a QEF generally is currently taxable on a pro rata share of the Company’s ordinary earnings and net capital gain as ordinary income and long-term capital gain, respectively. Neither that ordinary income nor any actual dividend from the Company would qualify for the 20% maximum federal income tax rate on dividends described above if the Company is a PFIC in the taxable year the ordinary income is realized or the dividend is paid or in the preceding taxable year. A QEF election cannot be made unless the Company provides U.S. Holders the information and computations needed to report income and gains pursuant to a QEF election. The Company expects that will not provide this information. It is, therefore, likely that U.S. holders would not be able to make a QEF election in any year we are a PFIC.
In lieu of a QEF election, a U.S. holder of stock in a PFIC that is considered marketable stock could elect to mark the stock to market annually, recognizing as ordinary income or loss each year an amount equal to the difference as of the close of the taxable year between the fair market value of the stock and the U.S. holder’s adjusted basis in the stock. Losses would be allowed only to the extent of net mark-to-market gain previously included in income by the U.S. holder under the election for prior taxable years. A U.S. holder’s adjusted basis in the common shares will be adjusted to reflect the amounts included or deducted with respect to the mark-to-market election. If the mark-to-market election were made, the rules set forth in the second preceding paragraph would not apply for periods covered by the election. A mark-to-market election will not apply during any later taxable year in which the Company does not satisfy the tests to be a PFIC. In general, the common shares will be marketable stock if the common shares are traded, other than in de minimis quantities, on at least 15 days during each calendar quarter on a national securities exchange that is registered with the SEC or on a designated national market system or on any exchange or market that the Treasury Department determines to have rules sufficient to ensure that the market price accurately represents the fair market value of the stock. Under current law, the mark-to-market election may be available to U.S. holders of common shares because the common shares are listed on The Nasdaq Capital Market and the TSX Venture Exchange (at least one of which should constitute a qualified exchange for this purpose), although there can be no assurance that the common shares will be “regularly traded” for purposes of the mark-to-market election.
If we are treated as a PFIC, each U.S. holder generally will be required to file a separate annual information return with the United States Internal Revenue Service (IRS) with respect to the Company (and any lower-tier PFICs). A failure to file this return will suspend the statute of limitations with respect to any tax return, event, or period to which such report relates (potentially including with respect to items that do not relate to a U.S. holder’s investment in the common shares). Given the complexities of the PFIC rules and their potentially adverse tax consequences, U.S. holders of common shares are urged to consult their tax advisers about the PFIC rules.
Medicare Surtax on Net Investment Income
Non-corporate U.S. Holders whose income exceeds certain thresholds generally will be subject to 3.8% surtax on their “net investment income” (which generally includes, among other things, dividends on, and capital gain from the sale or other taxable disposition of, the common shares). Absent an election to the contrary, if a QEF election is available and made, QEF inclusions will not be included in net investment income at the time a U.S. Holder includes such amounts in income, but rather will be included at the time distributions are received or gains are recognized. Non-corporate U.S. Holders should consult their own tax advisors regarding the possible effect of such tax on their ownership and disposition of the common shares, in particular the applicability of this surtax with respect to a non-corporate U.S. Holder that makes a QEF or mark-to-market election in respect of their common shares.
| Page 36 |
Information Reporting and Backup Withholding
Dividends paid on, and proceeds from the sale or other disposition of, a common share to a U.S. holder generally may be subject to information reporting requirements and may be subject to backup withholding unless the U.S. holder provides an accurate taxpayer identification number or otherwise establishes an exemption. The amount of any backup withholding collected from a payment to a U.S. holder will be allowed as a credit against the U.S. holder’s U.S. federal income tax liability and may entitle the U.S. holder to a refund, provided certain required information is furnished to the Internal Revenue Service. A non-U.S. holder generally will be exempt from these information reporting requirements and backup withholding tax but may be required to comply with certain certification and identification procedures in order to establish its eligibility for exemption.
Under U.S. federal income tax law and U.S. Treasury Regulations, certain categories of U.S. holders must file information returns with respect to their investment in, or involvement in, a foreign corporation. U.S. holders are urged to consult with their own tax advisors concerning such reporting requirements.
Reporting Obligations of Individual Owners of Foreign Financial Assets
Section 6038D of the Code generally requires U.S. individuals (and possibly certain entities that have U.S. individual owners) to file IRS Form 8938 if they hold certain “specified foreign financial assets,” the aggregate value of which exceeds $50,000 on the last day of the year or $75,000 at any time during the year. The definition of specified foreign financial assets includes not only financial accounts maintained in foreign financial institutions, but also, unless held in accounts maintained by a financial institution, any stock or security issued by a non-US. person, any financial instrument or contract held for investment that has an issuer or counterparty other than a U.S. person and any interest in a foreign entity. Persons who are required to report foreign financial assets and fail to do so may be subject to substantial penalties.
Canadian Federal Income Tax Consequences
The following summary of the material Canadian federal income tax consequences is stated in general terms and is not intended to be legal or tax advice to any particular shareholder. Each shareholder or prospective shareholder is urged to consult his or her own tax advisor regarding the tax consequences of his or her purchase, ownership and disposition of common shares. The tax consequences to any particular holder of common shares will vary according to the status of that holder as an individual, trust, corporation or member of a partnership, the jurisdiction in which that holder is subject to taxation, the place where that holder is resident and, generally, according to that holder’s particular circumstances.
This summary is applicable only to holders who are resident in the United States for income tax purposes, have never been resident in Canada for income tax purposes, deal at arm’s length with the Company, hold their common shares as capital property and who will not use or hold the common shares in carrying on business in Canada. Special rules, which are not discussed in this summary, may apply to a United States holder that is an issuer that carries on business in Canada and elsewhere.
This summary is based upon the provisions of the Income Tax Act (Canada) and the regulations thereunder (collectively, the Tax Act or ITA) and the Canada-United States Tax Convention (the Tax Convention) at the date of this Annual Report and the current administrative practices of the Canada Revenue Agency. This summary does not take into account provincial income tax consequences. The comments in this summary that are based on the Tax Convention are applicable to U.S. holders only if they qualify for benefits under the Tax Convention. Management urges each holder to consult his own tax advisor with respect to the income tax consequences applicable to him in his own particular circumstances.
Non-Resident Holders
The summary below is restricted to the case of a holder (a Holder) of one or more common shares who for the purposes of the Tax Act is a non-resident of Canada, holds his common shares as capital property and deals at arm’s length with the Company.
Dividends
A Holder will be subject to Canadian withholding tax (Part XIII Tax) equal to 25%, or such lower rates as may be available under an applicable tax treaty, of the gross amount of any dividend paid or deemed to be paid on his common shares. The Company will be required to withhold the applicable amount of Part XIII Tax from each dividend so paid and remit the withheld amount directly to the Receiver General for Canada for the account of the Holder.
| Page 37 |
Disposition of Common Shares
A Holder who disposes of common shares, including by deemed disposition on death, will not be subject to Canadian tax on any capital gain thereby realized unless the common share constituted “taxable Canadian property” as defined by the Tax Act. Generally, a common share of a public corporation will not constitute taxable Canadian property of a Holder unless he held the common share as capital property used by him carrying on a business in Canada, or he, persons with whom he did not deal at arm’s length or partnerships in which he or persons with whom he did not deal at arm’s length held an interest, alone or together held or held options to acquire, at any time within the 60 months preceding the disposition, 25% or more of the issued shares of any class of the capital shares of the Company and at any time during the 60 months preceding the disposition more than 50% of the value of the common shares is derived from, or from an interest in, Canadian real estate, including Canadian resource or timber resource properties.
Holders Resident in the United States
A Holder who is a resident of the United States and realizes a capital gain on disposition of common shares that was taxable Canadian property will, if qualified for benefits under the Tax Convention, generally be exempt from Canadian tax thereon unless (a) more than 50% of the value of the common shares is derived from, or from an interest in, Canadian real estate, including Canadian natural resource properties, (b) the common shares formed part of the business property of a permanent establishment that the Holder has or had in Canada within the 12 months preceding disposition, or (c) the Holder (i) was a resident of Canada at any time within the ten years immediately preceding the disposition, and for a total of 120 months during any period of 20 consecutive years, preceding the disposition, (ii) owned the common shares when he ceased to be resident in Canada, and (iii) the common shares were not subject to a deemed disposition on the Holder’s departure from Canada.
Inclusion in Taxable Income
A Holder who is subject to Canadian tax in respect of a capital gain realized on disposition of common shares must include one half of the capital gain (“taxable capital gain”) in computing his taxable income earned in Canada. The Holder may, subject to certain limitations, deduct one half of any capital loss (“allowable capital loss”) arising on disposition of taxable Canadian property from taxable capital gains realized in the year of disposition in respect to taxable Canadian property and, to the extent not so deductible, from such taxable capital gains of any of the three preceding years or any subsequent year.
Subject to certain exceptions, a non-resident person who disposes of taxable Canadian property must notify the Canada Revenue Agency either before or after the disposition (within ten days of the disposition).
| Item 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK. |
We are exposed to financial market risks associated with foreign exchange rates, concentration of credit, and liquidity. In accordance with our policies, we manage our exposure to various market-based risks and where material, these risks are reviewed and monitored by our Board of Directors.
Foreign Exchange Risk
Our exposure to foreign exchange risk is primarily related to fluctuations between the Canadian dollar and the U.S. dollar. Funds held in Mexican pesos are nominal. We incur operating expenses and capital expenditures mostly in U.S. dollars, with some operating expenses incurred in Canadian dollars which are subject to foreign currency fluctuations. The fluctuation of the U.S. dollar in relation to the Canadian dollar will have an impact upon our profitability and may also affect the value of our assets and the amount of shareholders’ equity. We have not entered into any agreements or purchased any instruments to hedge possible currency risks. At September 30, 2017, we held approximately CDN$1.5 million in cash and cash equivalents in Canadian dollars and the U.S. dollar was equal to 1.2458 Canadian dollars. Based on the exposure at September 30, 2017, a 10% annual change in the Canadian/U.S. exchange rate over the prior year would impact our net loss by approximately $122,000.
Concentration of Credit Risk
We are potentially subject to financial instrument concentration of credit risk through our cash equivalents, US Treasury bills and accounts receivables. We place our cash and cash equivalents in 4 week US Treasury bills or financial institutions believed to be credit worthy and perform periodic evaluations of their relative credit standing. We place short-term investments in 13 to 52 week US Treasury bills. Accounts receivables can be potentially exposed to a concentration of credit risk with our major customers.
| Page 38 |
The Company had concentrations of revenues in fiscal 2017, 2016 and 2015 as follows:
| 2017 | 2016 | 2015 | ||||||||||
| Product sales and contract services revenue | 79% from 2 customers | 76% from 3 customers | 82% from 3 customers | |||||||||
The Company had concentrations of accounts receivable at fiscal year-end 2016 as follows:
| 2016 | ||||
| Accounts receivable | 100 % from 1 customer | |||
There were no customer accounts receivable at September 30, 2017.
We assess the collectability of our accounts receivable through a review of our current aging, as well as an analysis of our historical collection rate, general economic conditions and credit status of our customers. As of September 30, 2017 and 2016, all outstanding accounts receivable were deemed to be fully collectible, and therefore, no allowance for doubtful accounts was recorded. We determine terms and conditions for our customers primarily based on the volume purchased by the customer, customer creditworthiness and past transaction history.
Management works to mitigate our concentration of credit risk with respect to accounts receivable through our credit evaluation policies, reasonably short payment terms and geographical dispersion of sales. Revenue includes export sales to foreign companies located principally in Europe and Asia.
Liquidity Risk
Liquidity risk is the risk that we will not be able to meet our financial obligations as they fall due. We attempt to manage liquidity risk by maintaining sufficient cash and cash equivalent and short-term investment balances. Liquidity requirements are managed based on expected cash flows to ensure that there is sufficient capital in order to meet our short-term obligations. At September 30, 2017 and 2016, we had cash and cash equivalents and short-term investment balances totaling $6.6 million and $11.4 million, respectively, to settle current liabilities of $.32 million and $.62 million, respectively.
| Item 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA. |
The financial statements and related financial information required to be filed hereunder are indexed under Item 15 of this Annual Report on Form 10-K and are incorporated herein by reference.
| Item 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE. |
There were no reportable events under this item during the past two fiscal years.
| Item 9A. | CONTROLS AND PROCEDURES. |
Disclosure Controls and Procedures
Our management is responsible for establishing and maintaining disclosure controls and procedures to provide reasonable assurance that material information related to our Company, including our consolidated subsidiaries, is made known to senior management, including our Chief Executive Officer and the Chief Financial Officer, by others within those entities on a timely basis so that appropriate decisions can be made regarding public disclosure.
We carried out an evaluation, under the supervision and with the participation of our management, including our Principal Executive Officer and our Principal Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e)) under the Securities and Exchange Act of 1934, as amended) as of September 30, 2017. Our Chief Executive Officer and Chief Financial Officer concluded that the disclosure controls and procedures as of September 30, 2017, were effective.
| Page 39 |
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for designing, establishing and maintaining a system of internal controls over financial reporting (as defined in Exchange Act Rule 13a-15(f)) to provide reasonable assurance that the financial information prepared by us for external purposes is reliable and has been recorded, processed and reported in an accurate and timely manner in accordance with accounting principles generally accepted in the United States. The Board of Directors is responsible for ensuring that management fulfills its responsibilities. The Audit Committee fulfills its role of ensuring the integrity of the reported information through its review of the interim and annual financial statements. Management reviewed the results of their assessment with our Audit Committee.
Because of its inherent limitations, our internal control over financial reporting may not prevent or detect all possible misstatements or frauds. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with policies or procedures may deteriorate.
Management has used the criteria issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in “Internal Control — Integrated Framework (2013)” to evaluate the effectiveness of our internal control over financial reporting. Management has assessed the effectiveness of our internal control over financial reporting and concluded that such internal control over financial reporting was effective as of September 30, 2017.
Attestation Report of Our Registered Public Accounting Firm
This Annual Report does not include an attestation report from our independent registered public accounting firm. We are an “emerging growth company,” as defined under the JOBS Act, and are subject to reduced public company reporting requirements. The JOBS Act provides that an emerging growth company is not required to have the effectiveness of such company’s internal control over financial reporting audited by its external auditors for as long as such company is deemed to be an emerging growth company.
Limitations on the Effectiveness of Controls
Our management, including the Chief Executive Officer and Chief Financial Officer, does not expect that our disclosure controls or our internal controls will prevent all error and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, within our Company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the control. The design of any system of controls is also based, in part, upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed achieving its stated goals under all potential future conditions; over time, control may become inadequate because of changes in conditions, or the degree of compliance with the policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting that occurred during the fourth quarter ended September 30, 2017 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
| Item 9B. | OTHER INFORMATION. |
None.
| Page 40 |
| Item 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE. |
Directors
Our directors and their ages as of November 30, 2017 are set forth below.
| Name | Age | Position(s) Held | Director Since | |||
| Tessie M. Che, Ph.D. | 67 | Director | September 25, 2013 | |||
| Paul Chun (1)(2)(3) | 36 | Director | December 8, 2016 | |||
| David L. Hill, Ph.D. (1)(2)(3) | 66 | Director | May 17, 2011 | |||
| Daniel E. Morse, Ph.D. | 76 | Director | April 9, 2010 | |||
| Frank R. Oakes | 67 |
President, Chief Executive Officer and Chairman of our Board of Directors |
April 9, 2010 | |||
| Charles V. Olson, D.Sc. (2) | 60 | Director | December 8, 2016 | |||
| Mayank D. Sampat (1)(2)(3) | 62 | Director | August 15, 2012 |
(1) Member of Audit Committee.
(2) Member of Compensation Committee.
(3) Member of the Nominating and Corporate Governance Committee.
There are no family relationships between any of our directors or executive officers.
Biographies and Qualifications. The biographies of our directors and certain information regarding each director’s experience, attributes, skills and/or qualifications that led to the conclusion that the director should be serving as a director of our Company are as follows:
Tessie M. Che, Ph.D. has been a director of Stellar since September 2013. Dr. Che is currently General Manager and Chair of the Board of Directors of Amaran Biotechnology Inc., a privately-held biopharmaceuticals manufacturer based in Taiwan, a position she has held since 2012. She is also a director of OBI Pharma USA, a wholly-owned subsidiary of OBI Pharma, Inc., a publicly traded biotechnology corporation in Taiwan. From 1998 to 2011 she served as COO and Sr. V.P., Corporate Affairs of Optimer Pharmaceuticals Inc., a company she co-founded. At Optimer, Dr. Che guided the company’s CMC team to the successful registration and commercialization of DificidTM in the U.S., Canada and Europe. Prior to Optimer, Dr. Che’s experience includes 20 years in research, operations and management at global companies, including Exxon Mobil Corp., Aventis Pharmaceuticals Inc., and EniChem SpA. Dr. Che holds bachelor degrees in chemistry from Illinois State University and Fu-Jen Catholic University (Taiwan) and a PhD in physical-inorganic chemistry from Brandeis University. She has authored numerous scientific publications and holds over 20 U.S. patents. Dr. Che has extensive scientific, operational, manufacturing, quality assurance, product development and senior management experience in the pharmaceutical and biotechnology industries, as well as experience serving on a board of directors within our industry.
Paul Chun has been a director of Stellar since December 2016 and serves as the chair of the Nominating and Governance Committee. He is a Managing Partner of Eldred Advisors LLC, a life sciences advisory firm he founded in May 2016. From November 2015 to April 2016, he served as Director of Strategy and Corporate Development at Kiromic, LLC. From May 2011 to October 2015, Mr. Chun served as a life sciences principal with Westwicke Partners, LLC, a capital markets advisory firm. During his tenure at Westwicke, he supported the capital markets and investor engagement objectives of private and public biopharma companies, including the support of multiple initial public offerings and other strategic transactions. Prior to Westwicke, he held various roles in investment research and corporate finance, including at Amgen, Inc., Tavistock Life Sciences and Goldman, Sachs & Co. He received his bachelors in biological sciences from Columbia University. Mr. Chun has broad experience in therapeutics development and commercialization, valuation, corporate development and finance.
David L. Hill, Ph.D. has been a director of Stellar since May 2011, and serves as the chair of the Compensation Committee. He served as Scientific Director for the ART Reproductive Center, Beverly Hills, California, from December 1999 until his retirement in December 2016. He is also an Assistant Clinical Professor in the Dept. of Obstetrics and Gynecology at the David Geffen School of Medicine, University of California, Los Angeles, and a Research Assistant IV at Cedars-Sinai Medical Center, Los Angeles, California. Dr. Hill received his Ph.D. in Biological Sciences from the Department of Pathology, School of Life Sciences, University of Connecticut and completed a Postdoctoral Fellowship at the Dana Farber Cancer Institute through an appointment by the Department of Physiology and Biophysics, Harvard Medical School, Boston, Massachusetts. Dr. Hill has extensive scientific and clinical research experience in our industry.
| Page 41 |
Daniel E. Morse, Ph.D. has been a director of Stellar since April 2010. Dr. Morse is the Wilcox Professor Emeritus of Molecular Genetics and Biochemistry Biotechnology, Biomolecular Science and Engineering, a position he has held since 2008, and Director of the Marine Biotechnology Center, at the University of California, Santa Barbara, a position he has held since 1986. Previously, he served as Director of the UCSB-MIT-Caltech Institute of Collaborative Biotechnologies from 2003 to 2010, and also served as Stellar’s Executive Vice-President, Science & Technology from 2010 until December 2011. Dr. Morse is an expert in the structure and function of the KLH molecule and internationally recognized expert in protein chemistry, molecular biology, molluscan reproductive biology, and aquaculture, and has an intimate understanding of our technology.
Frank R. Oakes was appointed our President and Chief Executive Officer and Chairman of our Board of Directors in April 2010. Prior to that time, he served as founder and Chief Executive Officer of Stellar’s California subsidiary since 1999. He has more than 40 years of management experience in aquaculture including a decade as Chief Executive Officer of The Abalone Farm, Inc., during which he led the company through the R&D, capitalization, and commercialization phases of development to become the largest abalone producer in the United States. Mr. Oakes is the inventor of our patented method for non-lethal extraction of hemolymph from a live gastropod mollusk. He was the principal investigator on our Small Business Innovation Research (SBIR) grant from the National Science Foundation and was principal investigator on our Phase I and II SBIR grants from the NIH’s Center for Research Resources, and a California Technology Investment Partnership (CalTIP) grant from the Department of Commerce. Mr. Oakes has consulted and lectured for the aquaculture industry around the world. He received his Bachelor of Science degree from California State Polytechnic University, San Luis Obispo and is a graduate of the Los Angeles Regional Technology Alliance University’s management-training program. Mr. Oakes is a valuable member of our Board due to his depth of operating, strategic, and senior management experience in our industry, specifically as related to aquaculture. Additionally, Mr. Oakes holds an intimate knowledge of Stellar due to his longevity in the industry and with us.
Charles V. Olson, D.Sc. has been a director of Stellar since December 2016 and a member of our scientific advisory board since June 2014. Since September 2017, he has served at Applied Molecular Transport Inc., as the Vice President of Biologics. He has also been a Principal Biotechnology Consultant for Compass Biotechnology LLC since 2006. Dr. Olson previously held senior and executive management positions at Anthera Pharmaceuticals Inc. from April 2010 to August 2017, NGM Bioharmaceuticals Inc, Coherus BioSciences Inc, Nexbio Inc., Cell Genesys, Inc., Biomarin Pharmaceuticals, Inc, and Onyx Pharmaceuticals, Inc. After graduate school, Dr. Olson was a Research Scientist at Kaiser Hospitals, followed by Scientist and Senior Scientist positions at Genentech and Bayer, respectively. He holds a B.A. in biology and chemistry from Westmont College, an M.A. in chemistry from the University of California at Santa Barbara and a D.Sc. in biochemistry. Dr. Olson has extensive scientific, manufacturing operations, process development, and senior management experience in the biopharmaceutical industry.
Mayank (Mike) D. Sampat has been a director of Stellar since August 2012, and serves as the chair of the Audit Committee. Mr. Sampat is an independent consultant providing business services to companies seeking expertise in financial planning and analysis, accounting and financial reporting, M&A transactions support and financial system implementation. He previously held the positions of controller at Precision Toxicology, LLC, a healthcare focused clinical laboratory specializing in providing quantitative drug testing, from February 2015 to May 2016, Zpower, LLC, an emerging manufacturer in the microbattery industry, from June 2012 to September 2014, and Imaging Advantage LLC from September 2010 to June 2012, and the position of Chief Financial Officer for Gamma Medica-Ideas, a supplier of imaging equipment to the medical industry, from September 2007 to June 2010. Mr. Sampat received a BBA in accounting from Bombay University and his MBA in Finance at Mercer University. Mr. Sampat is a seasoned finance and accounting executive, having worked with multiple companies ranging from startups to large Fortune 100 companies.
Executive Officers
Set forth below is certain information with respect to the names, ages, and positions of our executive officers as of November 30, 2017. Biographical information pertaining to Mr. Oakes, who is a director and an executive officer, may be found in the above section entitled “Directors.” The executive officers serve at the pleasure of our Board of Directors.
| Name | Age | Position(s) Held | Date of Appointment | |||
| Frank R. Oakes | 67 | President, Chief Executive Officer and Chairman of our Board of Directors | April 9, 2010 | |||
| Kathi Niffenegger, CPA | 60 | Chief Financial Officer and Corporate Secretary | November 1, 2013 | |||
| Gregory T. Baxter, Ph.D. | 58 | Executive Vice President of Corporate Development | December 1, 2016 |
Kathi Niffenegger, CPA was appointed Chief Financial Officer in November 2013 and Corporate Secretary in June 2013. She initially joined Stellar in May 2012 as Controller, after previously serving as the company’s outside Certified Public Accountant for more than 12 years. Ms. Niffenegger has more than 30 years of experience in accounting and finance in a range of industries. She held positions of increasing responsibility in the audit division of Glenn Burdette CPAs from 1988 to 2012 and served most recently as technical partner. She obtained CFO experience at Martin Aviation, and began her career at Peat, Marwick, Mitchell & Co. (now KPMG LLP). Ms. Niffenegger has held leadership roles for audits of manufacturing, aquaculture, pharmaceutical and governmental grant clients, and developed specific expertise in cost accounting systems and internal controls. Ms. Niffenegger holds a B.S. degree in Business Administration, Accounting from California State University, Long Beach and is a member of the American Institute of Certified Public Accountants (AICPA).
| Page 42 |
Gregory T. Baxter, Ph.D. joined Stellar’s executive management team in December 2016 following his service on the company’s Board of Directors, which he joined in August 2012. Dr. Baxter has served as an executive and scientist for several biotechnology corporations and foundations. Since 2001, Dr. Baxter has been a Senior Scientist in the Department of Clinical Drug Development for CCS Associates Inc., a scientific research consulting firm specializing in technical and support services for clinical research, design strategies for preclinical studies, chemical information sciences and research and development support for translational science. His prior experience includes serving as Program Director for the National Science Foundation (NSF) Division of Industrial Innovation and Partnerships, Founder and CSO of Hurel Corporation, Founder and CEO of Aegen Biosciences and Research Scientist for Molecular Device Corporation. He also serves as Adjunct Associate Professor at Cornell University in the College of Chemical Engineering and on the Founders Board of Stanford University's StartX Med Program. Dr. Baxter received his B.A. and Ph.D. in Biochemistry/Molecular Biology from University of California, Santa Barbara.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Exchange Act, requires that our directors, executive officers, and greater-than-10% shareholders file reports with the SEC on their initial beneficial ownership of our common shares and any subsequent changes. To our knowledge, based solely on a review of copies of such reports furnished to us by our officers and directors, we believe that, during the fiscal year ended September 30, 2017, no person required to file reports under Section 16(a) of the Exchange Act failed to file such reports on a timely basis during such fiscal year.
Code of Ethics
We have adopted a Code of Ethics and Business Conduct that applies to all of our directors, officers, and employees. A copy of our Code of Ethics and Business Conduct is available on the Investor Relations section of our website at http://ir.stellarbiotechnologies.com. We intend to satisfy the SEC’s disclosure requirements regarding amendments to, or waivers of, our Code of Ethics and Business Conduct by posting such information on our website. Copies of our Code of Ethics and Business Conduct may be obtained, free of charge, by writing to our Corporate Secretary, Stellar Biotechnologies, Inc., 332 East Scott Street, Port Hueneme, California 93041.
Nominations for Board of Directors
The Board of Directors has approved an advance notice policy, which was subsequently approved by our shareholders, that requires advance notice be given to us in certain circumstances where nominations of persons for election to the Board are made by our shareholders.
In the case of an annual meeting of shareholders, notice to the Company must be made not less than 30 days nor more than 65 days prior to the date of the annual meeting. However, in the event that the annual meeting is to be held on a date that is less than 40 days after the date on which the first public announcement of the date of the annual meeting was made, notice may be made not later than the close of business on the tenth (10th) day following such public announcement.
In the case of a special meeting of shareholders (which is not also an annual meeting), notice to the Company must be made not later than the close of business on the fifteenth (15th) day following the day on which the first public announcement of the date of the special meeting was made.
Information about our Board Committees
Our Board of Directors has appointed an Audit Committee, a Compensation Committee, and a Nominating and Corporate Governance Committee. The Board of Directors has determined that each director who serves on these committees is “independent,” as that term is defined by the listing rules of Nasdaq and rules of the Securities and Exchange Commission. The Board of Directors has adopted written charters for its Audit Committee, Compensation Committee, and Nominating and Corporate Governance Committee. Copies of these charters are available on our website at http://ir.stellarbiotechnologies.com. In addition, our board of directors appointed a temporary Strategic Investments Committee to approve actions related to potential strategic investments and a Pricing Committee to approve actions related to our capital raising transactions. There was no requirement for directors who served on these committees to be “independent”.
| Page 43 |
Audit Committee
Our Audit Committee is composed of Paul Chun, David Hill, and Mayank Sampat (chair). The purpose of the Audit Committee is to oversee our accounting and financial reporting processes and the audits of our financial statements. In that regard, the Audit Committee assists the Board in monitoring: (a) the integrity of our financial statements; (b) our independent auditor’s qualifications, independence, and performance; (c) the performance of our system of internal controls, financial reporting, and disclosure controls; and (d) our compliance with legal and regulatory requirements. To fulfill this obligation and perform its duties, the Audit Committee maintains effective working relationships with the Board, management, our internal auditor, and our independent auditor.
Mayank Sampat is the Chair of our Audit Committee and has extensive financial experience. He received an MBA in Finance from Mercer University, and has served in several financial positions with other companies, including several years as Chief Financial Officer for a medical equipment manufacturer. Mr. Sampat is considered to be “independent” as defined pursuant to the listing rules of the Nasdaq. The Board has determined that Mr. Sampat is an “audit committee financial expert” as defined in Item 407(d)(5)(ii) of Regulation S-K.
Compensation Committee
Our Compensation Committee is composed of Paul Chun, David Hill (chair), Charles Olson and Mayank Sampat. The purpose of the Compensation Committee is to oversee the Board’s responsibilities relating to compensation, including (i) the approval of compensation for our Company’s Chief Executive Officer and (ii) the review of compensation for our other executive officers. It has overall responsibility for approving and evaluating all of our compensation plans, policies and programs as such plans, policies and programs affect executive officers.
Nominating and Corporate Governance Committee
Our Nominating and Corporate Governance Committee is composed of Paul Chun (chair), David Hill and Mayank Sampat. The purpose of the Nominating and Corporate Governance Committee is to identify individuals qualified to become Board members; recommend to the Board individuals to serve as directors; advise the Board with respect to Board composition, procedures and committees; develop, recommend to the Board and annually review a set of corporate governance principles applicable to the Company; and oversee any related matters required by the federal securities laws.
| Item 11. | EXECUTIVE COMPENSATION. |
Executive Compensation
Our named executive officers for 2017 were Frank R. Oakes, Chief Executive Officer, President and Executive Chairman of our Board of Directors; Kathi Niffenegger, CPA, Chief Financial Officer and Corporate Secretary; and Gregory T. Baxter, Ph.D., our Executive Vice President of Corporate Development.
Summary Compensation Table
The following table sets forth information regarding the compensation awarded to, earned by or paid to the named executive officers.
| Name and Principal Position | Fiscal Year | Salary ($) | Bonus ($) | Stock Awards ($) (1) | Option Awards ($) (2) |
All Other Compensation ($) |
Total ($) | |||||||||||||||||||||||
| Frank R. Oakes | 2017 | $ | 257,100 | $ | 25,000 | $ | 296,969 | (3) | $ | - | $ | 23,669 | (4) | 602,738 | ||||||||||||||||
| President, Chief Executive | 2016 | 250,100 | 120,000 | - | - | 59,737 | 429,837 | |||||||||||||||||||||||
| Officer and Chairman of our | ||||||||||||||||||||||||||||||
| Board of Directors | ||||||||||||||||||||||||||||||
| Kathi Niffenegger, CPA | 2017 | 202,560 | 20,000 | - | 19,744 | 18,526 | (5) | 260,830 | ||||||||||||||||||||||
| Chief Financial Officer and | 2016 | 196,560 | 47,250 | - | 61,148 | 19,004 | 323,962 | |||||||||||||||||||||||
| Corporate Secretary | ||||||||||||||||||||||||||||||
| Gregory T. Baxter, Ph.D. | 2017 | 157,372 | 500 | - | 15,605 | 18,406 | (7) | 191,883 | ||||||||||||||||||||||
| Executive Vice President of Corporate Development (6) |
2016 | - | - | - | - | 11,800 | 11,800 | |||||||||||||||||||||||
| Page 44 |
| (1) | The amounts shown in this column represent the aggregate grant date fair value of the share awards based on the closing price on Nasdaq, not the actual amounts paid to or realized by the named executive officer during the covered fiscal year. It differs from the amounts recorded in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification 718, which were based on the share value at the inception of the performance share plan in April 2010 expensed over the estimated vesting period ended August 31, 2012. The vesting requirements of these awards are set forth in Note 8 to our audited consolidated financial statements for the year ended September 30, 2017 included in this Annual Report. |
| (2) | The amounts shown in this column represent the aggregate grant date fair value of the share option awards computed in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification 718, not the actual amounts paid to or realized by the named executive officers during the covered fiscal year. The assumptions used in determining grant date fair value of these awards are set forth in Note 8 to our audited consolidated financial statements for the year ended September 30, 2017 included in this Annual Report. |
| (3) | 235,690 shares were issued under our Performance Share Plan. |
| (4) | Represents (i) $15,719 in health insurance and (ii) $7,950 in 401(k) Company contributions. |
| (5) | Represents (i) $11,984 in health insurance and (ii) $6,542 in 401(k) Company contributions. |
| (6) | Dr. Baxter’s employment with the Company began December 1, 2016. Dr. Baxter was a director of the Company from August 15, 2012 until December 1, 2016. |
| (7) | Represents (i) $8,656 in health insurance, (ii) $1,050 in director fees and (iii) $8,700 in consultant fees prior to becoming an employee. |
Employment Agreements
We do not have employment agreements currently in effect with any of our named executive officers. Like our other employees, our executives are eligible for annual salary increases and discretionary equity grants.
Performance Share Plan
Under the merger agreement between our Company and our California subsidiary, we allotted 1,000,000 common shares (the Performance Shares) under a performance share plan (the Plan). The performance shares were reserved for issuance to certain officers, directors and employees of the Company upon achievement of three milestones related to completion of method development for commercial-scale manufacture of KLH, compilation and regulatory submittal of all required chemistry, manufacturing and control data and completion of preclinical toxicity and immunogenicity testing of products. Share-based compensation was recorded over the estimated vesting period ending in August 2012.
As each milestone was met as determined by our Board of Directors, one third of the Performance Shares were available to be released to the Plan participants. The three milestones were met on or before August 2012, and all Performance Shares had been issued to non-director employees by August 2012. In December 2013, the Board issued 151,515 Performance Shares to a former director of the Company. The Board issued the remaining Performance Shares in June 2017, of which (a) 13,468 Performance Shares were issued to a former director of the Company, (b) 235,690 Performance Shares were issued to Mr. Oakes, the Company’s current President, CEO and Chairman and an eligible participant in the Plan, and (c) 134,680 Performance Shares were issued to Dr. Morse, a current director of the Company and an eligible participant in the Plan. Since all Performance Shares under the Plan have been issued, the Plan was terminated.
| Page 45 |
Outstanding Equity Awards at 2017 Fiscal Year-End
The following table summarizes the equity awards made to our named executive officers that were outstanding at September 30, 2017.
| Option Awards | ||||||||||||||||
| Name | Award grant date | Number of securities underlying unexercised options (#) exercisable |
Number of securities underlying unexercised options (#) unexercisable (1) |
Option exercise prices ($) |
Option expiration date | |||||||||||
| Frank R. Oakes | 8/8/11 | 42,560 | - | $ | CDN6.50 | 8/8/18 | ||||||||||
| 4/13/12 | 37,560 | - | CDN4.20 | 4/13/19 | ||||||||||||
| Kathi Niffenegger, CPA | 6/18/12 | 9,000 | - | CDN2.90 | 6/18/19 | |||||||||||
| 12/19/12 | 5,000 | - | CDN2.50 | 12/19/19 | ||||||||||||
| 5/14/13 | 9,000 | - | CDN5.80 | 5/14/20 | ||||||||||||
| 11/1/13 | 10,000 | - | US18.30 | 11/1/20 | ||||||||||||
| 11/12/14 | 9,000 | - | CDN15.20 | 11/12/21 | ||||||||||||
| 12/22/15 | 10,000 | - | US7.24 | 12/22/22 | ||||||||||||
| 12/20/16 | 3,333 | 6,667 | US2.03 | 12/20/23 | ||||||||||||
| Gregory T. Baxter, Ph.D. | 8/16/12 | 7,000 | - | CDN3.70 | 8/16/19 | |||||||||||
| 11/12/14 | 1,250 | - | CDN15.20 | 11/12/21 | ||||||||||||
| 3/28/17 | - | 10,000 | US1.60 | 3/28/24 | ||||||||||||
| (1) | Our options vesting policy is described in the Outstanding Equity Awards Narrative Disclosure section. |
Outstanding Equity Awards Narrative Disclosure
Incentive Compensation Plan
We adopted an Incentive Compensation Plan in 2017 (the 2017 Plan) administered by the Board of Directors, which amended and restated the 2013 fixed share option plan (the 2013 Plan). Options, restricted shares and restricted share units are eligible for grant under the 2017 Plan. The number of shares available for issuance under the 2017 Plan is 1,597,000, including shares available for the exercise of outstanding options under the 2013 Plan. The purpose of the 2017 Plan is to advance the interests of the Company by encouraging equity participation through the acquisition of common shares of the Company. Our Board is responsible for the general administration of the 2017 Plan and the proper execution of its provisions, its interpretation and the determination of all questions arising thereunder. Specifically, the Board has the power to, among other things:
| · | allot common shares for issuance in connection with the exercise of options; |
| · | grant options, restricted shares or restricted share units; |
| · | amend, suspend, terminate or discontinue the plan; and |
| · | delegate all or a portion of its administrative powers as it may determine to one or more committees. |
Options, restricted shares or restricted share units may be awarded to our directors, officers, employees and consultants.
Options to purchase 410,970 common shares at prices ranging from CDN$2.50 to CDN$18.70 and $1.60 to $18.40 are outstanding at September 30, 2017. No restricted shares or restricted share units have been granted as of September 30, 2017.
Options granted during fiscal 2017 to employees and consultants under the 2017 Plan totaled 71,600 options to purchase common shares, at exercise prices ranging from $1.60 to $2.03.
| Page 46 |
Options Vesting Policy
Options granted for past service are subject to the following vesting schedule: (a) one-third of the option shall vest on the date of grant; (b) one-third of the option shall vest 12 months from the date of grant; and (c) the remaining one-third of the option shall vest 18 months from the date of grant. Options granted for future service are subject to the following vesting schedule: (x) one-third shall vest at 12 months from the date of grant, (y) one-third shall vest at 24 months from the date of grant and (z) one-third shall vest at 36 months from the date of grant.
Grants of Plan-Based Awards
The following table provides information regarding grants of plan-based awards to our named executive officers during fiscal year 2017.
| Name | Grant Date | All Other Stock Awards: Number of Shares of Stock or Units (#) | All Other Option Awards: Number of Securities Underlying Options (#) | Exercise or Base Price of Option Awards ($/Sh) | Grant Date Fair Value of Stock and Option Awards ($)(1) | ||||||||||||||
| Frank R. Oakes | 6/26/17 | 235,690 | - | $ | - | $ | 296,969 | (2) | |||||||||||
| Kathi Niffenegger, CPA | 12/20/16 | - | 10,000 | 2.03 | 19,744 | (3) | |||||||||||||
| Gregory T. Baxter, Ph.D. | 3/28/17 | - | 10,000 | 1.60 | 15,605 | (4) | |||||||||||||
| (1) | The amounts shown in this column for share awards represent the aggregate grant date fair value of the share awards based on the closing price on Nasdaq, not the actual amounts paid to or realized by the named executive officer during the covered fiscal year. It differs from the amounts recorded in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification 718, which were based on the share value at the inception of the performance share plan in April 2010 expensed over the estimated vesting period ended August 31, 2012. The vesting requirements of these awards are set forth in Note 8 to our audited consolidated financial statements for the year ended September 30, 2017 included in this Annual Report.
The amounts shown in this column for share option awards represent the aggregate grant date fair value of the share option awards computed in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification 718, not the actual amounts paid to or realized by the named executive officers during the fiscal year. The assumptions used in determining grant date fair value of these awards are set forth in Note 8 to our audited consolidated financial statements for the year ended September 30, 2017 included in this Annual Report. |
| (2) | The shares were issued under our Performance Share Plan and are fully vested. |
| (3) | The option awards were issued under our 2017 Plan for past service, and vest in thirds annually beginning December 2016. |
| (4) | The option awards were issued under our 2017 Plan for future service, and vest in thirds annually beginning March 2018. |
Retirement Benefits
We have established a 401(k) plan to provide retirement benefits to eligible executive officers and employees. Employees may enter the plan after they have been employed by us for at least three consecutive months. Under the plan, we contribute a flat non-elective contribution of 3% of eligible compensation for each plan participant at the end of the calendar year. Any Company contributions we made to the plan for our named executive officers are reflected in the “All Other Compensation” column of the Summary Compensation Table above.
Other than the funds contributed under our 401(k) plan, no other funds were set aside or accrued by us during fiscal 2017 to provide pension, retirement or similar benefits for our named executive officers.
| Page 47 |
Director Compensation
The following table sets forth information regarding the compensation of our non-employee directors for the fiscal year ended September 30, 2017.
| Name | Fees Earned or Paid in Cash ($) |
Stock Awards ($) (1) |
Option |
All Other Compensation ($) |
Total ($) | ||||||||||||||||||
| Tessie M. Che, Ph.D. | $ | 1,000 | $ | - | $ | 8,996 | (4) | $ | - | $ | 9,996 | ||||||||||||
| Paul Chun | 8,850 | - | 8,996 | (5) | - | 17,846 | |||||||||||||||||
| David L. Hill, Ph.D. | 10,250 | - | 8,996 | (4) | - | 19,246 | |||||||||||||||||
| Daniel E. Morse, Ph.D. | 5,700 | 169,697 | (3) | 8,996 | (4) | 900 | (6) | 185,293 | |||||||||||||||
| Charles V. Olson, D.Sc. | 4,700 | - | 8,996 | (5) | 5,775 | (7) | 19,471 | ||||||||||||||||
| Mayank D. Sampat | 10,250 | - | 8,996 | (4) | - | 19,246 | |||||||||||||||||
| (1) | The amounts shown in this column represent the aggregate grant date fair value of the share awards based on the closing price on Nasdaq, not the actual amounts paid to or realized by the named executive officer during the covered fiscal year. It differs from the amounts recorded in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification 718, which were based on the share value at the inception of the performance share plan in April 2010 expensed over the estimated vesting period ended August 31, 2012. The vesting requirements of these awards are set forth in Note 8 to our audited consolidated financial statements for the year ended September 30, 2017 included in this Annual Report. |
| (2) | The amounts shown in this column represent the aggregate grant date fair value of the share option awards computed in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification 718, not the actual amounts paid to or realized by the directors during the fiscal year. The assumptions used in determining grant date fair value of these awards are set forth in Note 8 to our audited consolidated financial statements for the year ended September 30, 2017 included in this Annual Report. |
| (3) | 134,680 shares were issued under our Performance Share Plan and are fully vested. |
| (4) | The option awards were issued under our 2017 Incentive Compensation Plan for past service, with 2,500 options vesting in thirds beginning December 2016 and 2,500 options vesting in thirds beginning March 2017. |
| (5) | The option awards were issued under our 2017 Incentive Compensation Plan, with 2,500 options for future service vesting in thirds beginning December 2017 and 2,500 options for past service vesting in thirds beginning March 2017. |
| (6) | Represents amount for service as member of our Scientific Advisory Board. |
| (7) | Represents (i) $5,425 in consultant fees and (ii) $350 for service as member of our Scientific Advisory Board. |
Outstanding Equity Awards at 2017 Fiscal Year-End
The following table summarizes the equity awards made to our directors that were outstanding at September 30, 2017.
| Name | Outstanding Options (#) | |||
| Tessie M. Che, Ph.D. | 12,000 | |||
| Paul Chun | 5,000 | |||
| David L. Hill, Ph.D. | 15,000 | |||
| Daniel E. Morse, Ph.D. | 31,600 | |||
| Charles V. Olson, D.Sc. | 6,250 | |||
| Mayank D. Sampat | 12,000 | |||
| Page 48 |
Narrative to Director Compensation Table
Non-Employee Director Compensation Policy
Pursuant to our non-employee director compensation policy, non-employee directors receive $1,000 for each Board meeting attended in person and $350 for each Board meeting attended by telephone. Members of Board committees also receive $350 for each committee meeting attended. Non-executive directors may also receive share option awards at the discretion of the Board of Directors.
Non-Employee Directors on our Scientific Advisory Board
Dr. Morse and Dr. Olson are members of our Scientific Advisory Board. As compensation for their services, the members of our Scientific Advisory Board receive certain advisory fees and expense reimbursements. Amounts for their services as members of our Scientific Advisory Board are reflected in the Director Compensation table above.
Compensation Committee Interlocks and Insider Participation
The members of our Compensation Committee during the fiscal year ended September 30, 2017 were Paul Chun, David Hill (chairman), Charles Olson and Mayank Sampat.
None of the individuals who served as a member of the Compensation Committee during fiscal 2017 was at any time during fiscal 2017 an officer or employee of our Company.
| Item 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS. |
Equity Compensation Plan Information
The following table provides certain information as of September 30, 2017 about our common shares that may be issued under our equity compensation plans, which consists of our 2017 Incentive Compensation Plan:
| Plan category | Number of securities to be issued upon exercise of outstanding options, warrants and rights | Weighted-average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) | |||||||||
| (a) | (b) | (c) | ||||||||||
| Equity compensation plans approved by security holders | 410,970 | $ | 5.74 | 1,186,030 | ||||||||
| Equity compensation plans not approved by security holders | N/A | N/A | N/A | |||||||||
| Total | 410,970 | $ | 5.74 | 1,186,030 | ||||||||
Security Ownership of Certain Beneficial Owners and Management
The following tables sets forth certain information as of November 30, 2017, with respect to the beneficial ownership of our common shares by: (1) all of our directors; (2) our named executive officers listed in the Summary Compensation Table; (3) all of directors and executive officers as a group; and (4) each person known by us to beneficially own more than 5% of our outstanding common shares.
We have determined beneficial ownership in accordance with the rules of the SEC. Except as indicated by the footnotes below, we believe, based on the information furnished to us, that the persons and entities named in the table below have sole voting and investment power with respect to all common shares that they beneficially own, subject to applicable community property laws.
Common shares subject to options or warrants currently exercisable or exercisable within 60 days of November 30, 2017 are deemed outstanding for computing the share ownership and percentage of the person holding such options and warrants, but are not deemed outstanding for computing the percentage of any other person. The percentage ownership of our common shares of each person or entity named in the following table is based on 10,520,096 common shares outstanding as of November 30, 2017.
| Page 49 |
Directors and Officers
| Name and Address of Beneficial Owner (1) | Amount and Nature of Beneficial Ownership | Percent of Shares Beneficially Owned | ||||||
| Frank R. Oakes | 464,587 | (2) | 4.4 | % | ||||
| Kathi Niffenegger, CPA | 58,667 | (3) | * | |||||
| Gregory T. Baxter, Ph.D. | 8,250 | (4) | * | |||||
| Tessie M. Che, Ph.D. | 9,500 | (5) | * | |||||
| Paul Chun | 1,666 | (6) | * | |||||
| David L. Hill, Ph.D. | 14,500 | (7) | * | |||||
| Daniel E. Morse, Ph.D. | 125,609 | (8) | 1.2 | % | ||||
| Charles V. Olson, D.Sc. | 2,916 | (9) | * | |||||
| Mayank D. Sampat | 9,500 | (10) | * | |||||
| All directors and executive officers as a group (9 persons) | 695,195 | (11) | 6.5 | % | ||||
* Percentage of shares beneficially owned does not exceed one percent.
| (1) | Unless otherwise indicated, the address of each beneficial owner is c/o Stellar Biotechnologies, Inc., 332 E. Scott Street, Port Hueneme, California 93041. |
| (2) | This amount includes (i) 80,120 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017; and excludes (ii) 20,867 common shares and 6,200 common shares issuable upon the exercise of outstanding options currently exercisable or exercisable within 60 days of November 30, 2017 which are held by Mr. Oakes’ spouse who has sole voting and dispositive power over the securities, and as to which Mr. Oakes disclaims beneficial ownership. Mr. Oakes does not have the power to vote or dispose of, or to direct the voting or disposition of, the shares held by his spouse, or with respect to any shares acquired under her outstanding options. |
| (3) | Represents 58,667 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (4) | Represents 8,250 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (5) | Represents 9,500 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (6) | Represents 1,666 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (7) | This amount includes 12,500 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (8) | This amount includes 29,100 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (9) | Represents 2,916 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (10) | Represents 9,500 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
| (11) | This amount includes 212,219 shares issuable upon the exercise of options currently exercisable or exercisable within 60 days of November 30, 2017. |
Shareholders Known by Us to Own 5% or More of Our Common Shares
| Name and Address of Beneficial Owner | Amount
and | Percent of Shares Beneficially Owned | ||||||
| Ernesto Echavarria Blvd. Anaya 1225 Culiacan Sinaloa, Mexico 80040 | 1,411,310 | 13.4 | % | |||||
| Page 50 |
| Item 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE. |
Related Party Transactions
Patent Royalty Agreement
On August 14, 2002, through our California subsidiary, we entered into an agreement with Frank Oakes, our Chief Executive Officer, where he would receive royalty payments in exchange for the assignment of his rights to U.S. Patent No. 6,852,338 to us. The royalty is 5% of gross receipts from products using this invention in excess of $500,000 annually. Our current operations utilize this invention. Patent royalties of $35,516 were paid to Mr. Oakes for the year ended September 30, 2016. No royalties were paid for the year ended September 30, 2017.
Collaboration Agreement
In December 2013, we entered into a collaboration agreement (the Amaran Agreement) with Amaran Biotechnology, Inc. to develop and evaluate methods for Amaran’s potential manufacture of the OBI-822 (Adagloxad Simolenin) active immunotherapy using our GMP grade Stellar KLH. The Amaran Agreement expired by its terms on December 7, 2015.
Revenues received from Amaran under the Amaran Agreement totaled $32,000 during the fiscal year ended September 30, 2016. The terms of the collaboration with Amaran also provided for negotiation of a commercial supply agreement for Stellar KLH in the future, which was executed in February 2017.
Tessie Che, a member of our Board of Directors, currently serves as general manager and chair of the board of directors of Amaran.
Policies and Procedures for Review of Related Party Transactions
The Audit Committee reviews, approves and oversees any transaction between us and any “related person” (as defined in Item 404 of Regulation S-K) and any other potential conflict of interest situations, on an ongoing basis. Under these policies and procedures, the Audit Committee is to be informed of transactions subject to review before their implementation. The procedures establish our practices for obtaining and reporting information to the Audit Committee regarding such transactions on a periodic and an as-needed basis. The policy provides that such transactions are to be submitted for approval before they are initiated but also provides for ratification of such transactions. No director who is interested in a transaction may participate in the Audit Committee’s determinations as to the appropriateness of such transaction.
Director Independence
In evaluating the independence of our Board members and the composition of the committees of our Board of Directors, the Board of Directors utilizes the definition of “independence” as that term is defined by the Securities Exchange Act of 1934, and the Nasdaq Listing Rules. Using this standard, the Board of Directors has determined that Paul Chun, David Hill, Daniel Morse, Charles Olson and Mayank Sampat are “independent directors.” This means that our Board of Directors is composed of a majority of independent directors as required by the rules of Nasdaq.
| Item 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES. |
The following table shows the aggregate fees paid or accrued for audit and other services provided for the years ended September 30, 2017 and 2016 rendered by Moss Adams LLP.
Principal Accountant Fees and Services
| Type of Service | Fiscal Year 2017 | Fiscal Year 2016 | ||||||
| Audit Fees | $ | 190,000 | $ | 238,000 | ||||
| Audit-Related Fees | - | - | ||||||
| Tax Fees | - | - | ||||||
| All Other Fees | - | - | ||||||
| Total | $ | 190,000 | $ | 238,000 | ||||
Audit Fees consisted of fees incurred for professional services rendered for audits of the years ended September 30, 2017 and 2016 and include procedures related to registrations and offerings.
| Page 51 |
Pre-Approval Policies and Procedures
The Audit Committee is directly responsible for the appointment, compensation and oversight of our auditors. It has established procedures for the receipt, retention, and treatment of complaints received by us regarding accounting, internal accounting controls, or auditing matters, and the confidential, anonymous submission by our employees of concerns regarding questionable accounting or auditing matters. The Audit Committee also has the authority and the funding to engage independent counsel and other outside advisors.
The Audit Committee pre-approves all audit and permissible non-audit services provided by the independent registered public accounting firm. These services may include audit services, audit-related services, tax services and other services. Pre-approval is generally provided for up to one year, and any pre-approval is detailed as to the particular service or category of services and is generally subject to an amount or range of estimated fees. All proposed engagements of the auditor for audit and permitted non-audit services are submitted to the Audit Committee for approval prior to the beginning of any such services. Our auditors are required to periodically report to the Audit Committee regarding the extent of services provided by the independent registered public accounting firm in accordance with the pre-approval, and the fees for the services performed to date. The Audit Committee may also pre-approve particular services on a case-by-case basis. The Audit Committee pre-approved 100% of the audit services performed by our independent registered public accounting firm for the fiscal year ended September 30, 2017.
| Page 52 |
| Item 15. | EXHIBITS, FINANCIAL STATEMENT SCHEDULES. |
| (a) | The following documents are filed as a part of this Annual Report: |
(1) Financial Statements
The list of consolidated financial statements and notes required by this Item 15 (a) (1) is set forth in the “Index to Financial Statements” on page F-1 of this Annual Report.
(2) Financial Statement Schedules
All schedules have been omitted because the required information is included in the financial statements or notes thereto.
| (b) | Exhibits |
The exhibits listed on the Exhibit Index below are filed as part of this Annual Report.
| Page 53 |
EXHIBIT INDEX
| Exhibit No. | Description |
| 4.1 | Form of Warrant dated June 30, 2016 (included as Exhibit 4.1 to the Company’s Current Report on Form 8-K filed on June 30, 2016, and incorporated herein by reference). |
| 10.8 @ | Fixed Share Option Plan dated December 18, 2013 (included as Exhibit 10.11 to the Company’s Annual Report on Form 10-K filed on November 14, 2014, and incorporated herein by reference). |
| 10.9 @ | 2017 Incentive Compensation Plan (included as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed on March 29, 2017, and incorporated herein by reference). |
| Page 54 |
| 10.10 @ | Performance Share Plan dated April 9, 2010 (included as Exhibit 10(d) to the Company’s Registration Statement on Form 20-F filed on February 3, 2012, and incorporated herein by reference). |
| 10.11 | Advance Notice Policy, adopted October 31, 2013 (included as Exhibit 10.14 to the Company’s Annual Report on Form 10-K filed on November 14, 2014, and incorporated herein by reference). |
| 10.20 | Form of Securities Purchase Agreement, dated June 30, 2016 (included as Exhibit 10.1 to the Company’s Current Report on Form 8-K filed on June 30, 2016, and incorporated herein by reference). |
| 14.1 | Code of Ethics and Business Conduct (filed herewith). |
| 21 | Subsidiaries of Stellar Biotechnologies, Inc. (filed herewith). |
| 23.1 | Consent of Moss Adams LLP (filed herewith). |
| 32.1 ^ | Certification of the Chief Executive Officer pursuant to 18 U.S.C. 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (filed herewith). |
| 32.2 ^ | Certification of the Chief Financial Officer pursuant to 18 U.S.C. 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (filed herewith). |
| Page 55 |
| 101.INS | XBRL Instance Document |
| 101.SCH | XBRL Taxonomy Extension Schema Document |
| 101.CAL | XBRL Taxonomy Calculation Linkbase Document |
| 101.DEF | XBRL Taxonomy Extension Definition Linkbase Document |
| 101.LAB | XBRL Taxonomy Label Linkbase Document |
| 101.PRE | XBRL Taxonomy Presentation Linkbase Document |
| @ | Management contract or compensatory plan or arrangement. |
| # | Confidential treatment has been granted for certain portions of this exhibit. Original copies have been filed separately with the Securities and Exchange Commission pursuant to Rule 24B-2 of the Securities Exchange Act of 1934, as amended. |
| ^ | A signed original of this written statement required by Section 906 has been provided and will be retained by the Company and furnished to the Securities and Exchange Commission or its staff upon request. |
| Page 56 |
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| Date: December 1, 2017 | STELLAR BIOTECHNOLOGIES, INC. |
| /s/ Frank R. Oakes | |
| Frank R. Oakes | |
| President and Chief Executive Officer | |
| (Principal Executive Officer) |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signature | Title | Date | ||
| /s/ Frank R. Oakes | President, Chief Executive Officer, and | December 1, 2017 | ||
| Frank R. Oakes | Chairman of the Board of Directors (Principal Executive Officer) | |||
| /s/ Kathi Niffenegger | Chief Financial Officer | December 1, 2017 | ||
| Kathi Niffenegger | (Principal Financial and Accounting Officer) | |||
| /s/ Tessie M. Che | Director | December 1, 2017 | ||
| Tessie M. Che | ||||
| /s/ Paul Chun | Director | December 1, 2017 | ||
| Paul Chun | ||||
| /s/ David L. Hill | Director | December 1, 2017 | ||
| David L. Hill | ||||
| /s/ Daniel E. Morse | Director | December 1, 2017 | ||
| Daniel E. Morse | ||||
| /s/ Charles V. Olson | Director | December 1, 2017 | ||
| Charles V. Olson | ||||
| /s/ Mayank D. Sampat | Director | December 1, 2017 | ||
| Mayank D. Sampat |
| Page 57 |
STELLAR BIOTECHNOLOGIES, INC.
INDEX TO FINANCIAL STATEMENTS
| F-1 |
Consolidated Financial Statements
For the Years Ended September 30, 2017, 2016 and 2015
| F-2 |
Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders
Stellar Biotechnologies, Inc.
We have audited the accompanying consolidated balance sheets of Stellar Biotechnologies, Inc. (the Company) as of September 30, 2017 and 2016, and the related consolidated statements of operations, changes in equity and cash flows for the fiscal years ended September 30, 2017, 2016 and 2015. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the consolidated financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall consolidated financial statement presentation. We believe that our audits provide reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the consolidated financial position of Stellar Biotechnologies, Inc. as of September 30, 2017 and 2016, and the consolidated results of its operations and its cash flows for fiscal years ended September 30, 2017, 2016 and 2015, in conformity with accounting principles generally accepted in the United States of America.
| /s/ Moss Adams LLP | |
| Los Angeles, California | |
| December 1, 2017 |
| F-3 |
| Stellar Biotechnologies, Inc. |
| Consolidated Balance Sheets |
| September 30, | September 30, | |||||||
| 2017 | 2016 | |||||||
| Assets: | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 4,570,951 | $ | 7,416,904 | ||||
| Accounts receivable | 1,287 | 85,813 | ||||||
| Short-term investments | 1,994,401 | 3,988,794 | ||||||
| Inventory | 68,114 | 249,430 | ||||||
| Prepaid expenses | 123,694 | 358,714 | ||||||
| Total current assets | 6,758,447 | 12,099,655 | ||||||
| Noncurrent assets: | ||||||||
| Equity investment in joint venture | 66,695 | 66,695 | ||||||
| Property, plant and equipment, net | 879,523 | 756,114 | ||||||
| Deposits | 15,340 | 15,340 | ||||||
| Total noncurrent assets | 961,558 | 838,149 | ||||||
| Total Assets | $ | 7,720,005 | $ | 12,937,804 | ||||
| Liabilities and Shareholders' Equity: | ||||||||
| Current liabilities: | ||||||||
| Accounts payable and accrued liabilities | $ | 320,947 | $ | 623,644 | ||||
| Total Current Liabilities | 320,947 | 623,644 | ||||||
| Commitments (Note 7) | ||||||||
| Shareholders' equity: | ||||||||
| Common shares, unlimited common shares authorized, no par value, 10,520,096 and 10,136,258 issued and outstanding at September 30, 2017 and 2016 | 48,351,701 | 47,280,792 | ||||||
| Accumulated share-based compensation | 4,439,400 | 5,394,763 | ||||||
| Accumulated deficit | (45,392,043 | ) | (40,361,395 | ) | ||||
| Total Shareholders' Equity | 7,399,058 | 12,314,160 | ||||||
| Total Liabilities and Shareholders' Equity | $ | 7,720,005 | $ | 12,937,804 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-4 |
| Stellar Biotechnologies, Inc. |
| Consolidated Statements of Operations |
| Years Ended | ||||||||||||
| September 30, | September 30, | September 30, | ||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Revenues: | ||||||||||||
| Product sales | $ | 178,287 | $ | 1,239,689 | $ | 563,689 | ||||||
| Contract services revenue | 50,000 | 32,000 | 195,000 | |||||||||
| 228,287 | 1,271,689 | 758,689 | ||||||||||
| Expenses: | ||||||||||||
| Costs of sales and contract services | 250,042 | 818,566 | 580,824 | |||||||||
| Costs of aquaculture | 284,411 | 309,262 | 259,423 | |||||||||
| Research and development | 1,973,400 | 1,729,445 | 1,029,489 | |||||||||
| General and administrative | 2,944,980 | 3,322,772 | 3,227,545 | |||||||||
| 5,452,833 | 6,180,045 | 5,097,281 | ||||||||||
| Loss from Operations | (5,224,546 | ) | (4,908,356 | ) | (4,338,592 | ) | ||||||
| Other Income (Loss) | ||||||||||||
| Foreign exchange gain (loss) | 162,028 | 76,800 | (653,333 | ) | ||||||||
| Gain (loss) in fair value of warrant liability | - | (211,956 | ) | 2,131,062 | ||||||||
| Investment income | 32,670 | 24,632 | 54,634 | |||||||||
| 194,698 | (110,524 | ) | 1,532,363 | |||||||||
| Loss Before Income Tax | (5,029,848 | ) | (5,018,880 | ) | (2,806,229 | ) | ||||||
| Income tax expense | 800 | 7,200 | 36,800 | |||||||||
| Net Loss | $ | (5,030,648 | ) | $ | (5,026,080 | ) | $ | (2,843,029 | ) | |||
| Loss per common share: | ||||||||||||
| Basic and diluted | $ | (0.49 | ) | $ | (0.57 | ) | $ | (0.36 | ) | |||
| Weighted average number of common shares outstanding: | ||||||||||||
| Basic and diluted | 10,237,213 | 8,826,312 | 7,956,962 | |||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-5 |
| Stellar Biotechnologies, Inc. |
| Consolidated Statements of Cash Flows |
| Years Ended | ||||||||||||
| September 30, | September 30, | September 30, | ||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Cash Flows Used In Operating Activities: | ||||||||||||
| Net loss | $ | (5,030,648 | ) | $ | (5,026,080 | ) | $ | (2,843,029 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 179,322 | 149,565 | 159,521 | |||||||||
| Share-based compensation | 115,546 | 259,379 | 267,222 | |||||||||
| Foreign exchange (gain) loss | (162,028 | ) | (76,800 | ) | 653,333 | |||||||
| (Gain) loss in fair value of warrant liability | - | 211,956 | (2,131,062 | ) | ||||||||
| Changes in working capital items: | ||||||||||||
| Accounts receivable | 84,573 | 71,827 | (113,917 | ) | ||||||||
| Inventory | 181,316 | 307,850 | (522,389 | ) | ||||||||
| Prepaid expenses | 235,001 | (197,150 | ) | (45,758 | ) | |||||||
| Deposits | - | 560 | - | |||||||||
| Accounts payable and accrued liabilities | (302,731 | ) | (33,403 | ) | 77,018 | |||||||
| Deferred revenue | (173,333 | ) | 86,666 | |||||||||
| Net cash used in operating activities | (4,699,649 | ) | (4,505,629 | ) | (4,412,395 | ) | ||||||
| Cash Flows From Investing Activities: | ||||||||||||
| Acquisition of property, plant and equipment | (302,733 | ) | (402,271 | ) | (274,589 | ) | ||||||
| Purchase of short-term investments | (5,005,607 | ) | (11,995,450 | ) | (13,677 | ) | ||||||
| Proceeds on sales and maturities of short-term investments | 7,000,000 | 13,021,827 | 410,736 | |||||||||
| Contribution to joint venture | - | (66,695 | ) | - | ||||||||
| Net cash provided by investing activities | 1,691,660 | 557,411 | 122,470 | |||||||||
| Cash Flows From Financing Activities: | ||||||||||||
| Proceeds from issuance of common shares, net | - | 6,277,500 | - | |||||||||
| Payments for share issuance costs | - | (332,764 | ) | - | ||||||||
| Proceeds from exercise of warrants and options | - | 1,368,260 | 106,777 | |||||||||
| Net cash provided by financing activities | - | 7,312,996 | 106,777 | |||||||||
| Effect of exchange rate changes on cash and cash equivalents | 162,036 | 96,623 | (629,808 | ) | ||||||||
| Net change in cash and cash equivalents | (2,845,953 | ) | 3,461,401 | (4,812,956 | ) | |||||||
| Cash and cash equivalents - beginning of year | 7,416,904 | 3,955,503 | 8,768,459 | |||||||||
| Cash and cash equivalents - end of year | $ | 4,570,951 | $ | 7,416,904 | $ | 3,955,503 | ||||||
| Cash (demand deposits) | $ | 3,847,655 | $ | 972,412 | $ | 3,955,503 | ||||||
| Cash equivalents | 723,296 | 6,444,492 | - | |||||||||
| Cash and cash equivalents | $ | 4,570,951 | $ | 7,416,904 | $ | 3,955,503 | ||||||
| Supplemental cash flow information: | ||||||||||||
| Cash paid during the year for taxes | $ | 800 | $ | 7,200 | $ | 36,800 | ||||||
| Supplemental disclosure of non-cash transactions: | ||||||||||||
| Share issuance costs withheld from escrow proceeds | $ | - | $ | 472,500 | $ | - | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-6 |
| Stellar Biotechnologies, Inc. |
| Consolidated Statements of Changes in Equity |
| Accumulated | Total | |||||||||||||||||||
| Common | Share-Based | Accumulated | Shareholders' | |||||||||||||||||
| Shares | Shares | Compensation | Deficit | Equity | ||||||||||||||||
| Balance - September 30, 2014 | 7,941,985 | $ | 37,883,877 | $ | 5,073,144 | $ | (32,492,286 | ) | $ | 10,464,735 | ||||||||||
| Proceeds from exercise of warrants | 4,020 | 12,609 | - | - | 12,609 | |||||||||||||||
| Transfer to common shares on exercise of warrants | - | 10,000 | (426 | ) | - | 9,574 | ||||||||||||||
| Proceeds from exercise of options | 38,753 | 94,168 | - | - | 94,168 | |||||||||||||||
| Transfer to common shares on exercise of options | - | 113,561 | (113,561 | ) | - | - | ||||||||||||||
| Share-based compensation | - | - | 267,222 | - | 267,222 | |||||||||||||||
| Net loss | - | - | - | (2,843,029 | ) | (2,843,029 | ) | |||||||||||||
| Balance - September 30, 2015 | 7,984,758 | $ | 38,114,215 | $ | 5,226,379 | $ | (35,335,315 | ) | $ | 8,005,279 | ||||||||||
| Issuance of common shares | 1,687,500 | 6,750,000 | - | - | 6,750,000 | |||||||||||||||
| Share issuance costs | - | (805,264 | ) | - | - | (805,264 | ) | |||||||||||||
| Proceeds from exercise of warrants | 464,000 | 1,368,260 | - | - | 1,368,260 | |||||||||||||||
| Transfer to common shares on exercise of warrants | - | 1,853,581 | (90,995 | ) | - | 1,762,586 | ||||||||||||||
| Share-based compensation | - | - | 259,379 | - | 259,379 | |||||||||||||||
| Net loss | - | - | - | (5,026,080 | ) | (5,026,080 | ) | |||||||||||||
| Balance - September 30, 2016 | 10,136,258 | $ | 47,280,792 | $ | 5,394,763 | $ | (40,361,395 | ) | $ | 12,314,160 | ||||||||||
| Issuance of performance shares | 383,838 | 1,070,909 | (1,070,909 | ) | - | - | ||||||||||||||
| Share-based compensation | - | - | 115,546 | - | 115,546 | |||||||||||||||
| Net loss | - | - | - | (5,030,648 | ) | (5,030,648 | ) | |||||||||||||
| Balance - September 30, 2017 | 10,520,096 | $ | 48,351,701 | $ | 4,439,400 | $ | (45,392,043 | ) | $ | 7,399,058 | ||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-7 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
| 1. | Nature of Operations |
Stellar Biotechnologies, Inc. (the Company) is organized under the laws of British Columbia, Canada. The Company’s business is the aquaculture, research and development, manufacture and commercialization of Keyhole Limpet Hemocyanin (KLH). The Company markets and distributes its KLH products to biotechnology and pharmaceutical companies, academic institutions, and clinical research organizations primarily in Europe, North American and Asia. The Company’s common shares have been listed for trading on The Nasdaq Capital Market in the United States under the symbol SBOT since November 5, 2015. From January 15, 2013 through November 4, 2015, the Company’s common shares were quoted in the United States on the U.S. OTCQB Marketplace Exchange under the symbol SBOTF. From April 19, 2010 to April 8, 2016 the Company’s common shares were listed in Canada on the TSX Venture Exchange as a Tier 2 issue under the trading symbol KLH.
In April 2010, the Company changed its name from CAG Capital, Inc. to Stellar Biotechnologies, Inc. and completed a reverse merger transaction with Stellar Biotechnologies, Inc., a California corporation, which was founded in September 1999, and remains the Company’s wholly-owned subsidiary and principal operating entity. In January 2017, the California subsidiary and the Company established a wholly-owned Mexican subsidiary under the name BioEstelar, S.A. de C.V. in Ensenada, Baja California to perform aquaculture research and development activities in Mexico. The Company’s executive offices are located at 332 E. Scott Street, Port Hueneme, California, 93041, USA, and its registered and records office is Royal Centre, 1055 West Georgia Street, Suite 1500, Vancouver, BC, V6E 4N7, Canada.
Functional Currency
The consolidated financial statements of the Company are presented in U.S. dollars, which is the Company’s functional currency, unless otherwise stated.
Management Plans
Company operations have historically been funded by the issuance of common shares, exercise of warrants, grant revenues, contract services revenue and product sales. For the fiscal years 2017, 2016, and 2015, the Company reported net losses of approximately $5.0 million, $5.0 million, and $2.8 million, respectively. As of September 30, 2017, the Company had an accumulated deficit of approximately $45.4 million and working capital of approximately $6.4 million. While the Company plans to finance company operations for the next twelve months with cash on hand and product sales, management expects to continue incurring losses for the foreseeable future and will need to raise additional capital to pursue our business plan beyond December 2018. Management is taking action to ensure the Company will continue as a going concern for at least one year beyond the date of the issuance of the Company’s financial statements. First, management has flexibility to adjust planned expenditures based on a number of factors including the size and timing of capital expenditures, staffing levels, inventory levels, and the status of customer clinical trials. Management also seeks to expand the customer base for existing marketed products, and is currently evaluating opportunities to secure additional financing through debt and/or equity financings, including transactions with strategic customers and partners that may include debt and/or equity arrangements.
| 2. | Basis of Presentation |
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States (U.S. GAAP) and include the accounts of the Company, its wholly-owned subsidiaries, Stellar Biotechnologies, Inc., a California corporation in the U.S. and BioEstelar, S.A. de C.V. a Baja California corporation in Mexico. All significant intercompany balances and transactions have been eliminated in consolidation.
| F-8 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
| 3. | Significant Accounting Policies |
| a) | Use of Estimates |
The preparation of financial statements in conformity with U.S. GAAP requires management to make certain estimates, judgments and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the dates of the consolidated financial statements and the reported amounts of revenues and expenses during the reported periods. These estimates include warrant liabilities, share-based compensation, intangible assets, valuation of accounts receivable, valuation of inventory, and income taxes. Actual outcomes could differ from these estimates. These consolidated financial statements include estimates, which by their nature are uncertain. The impacts of such estimates are pervasive throughout the consolidated financial statements, and may require accounting adjustments based on future periods if the revision affects both current and future periods. These estimates are based on historical experience, current and future economic conditions and other factors, including expectations of future events that are believed to be reasonable under the circumstances.
| b) | Cash and Cash Equivalents |
Cash and cash equivalents consist of demand deposits with financial institutions and highly liquid investments which are readily convertible into cash with maturities of three months or less when purchased.
| c) | Investments |
Investments at September 30, 2017 and 2016 consisted of U.S. Treasury bills with original maturities between 13 and 52 weeks. They are classified as held-to-maturity and are reported at amortized cost, which approximates fair value. The Company regularly reviews these investments to determine whether any decline in fair value below the amortized cost basis has occurred that is other than temporary. If a decline in fair value has occurred that is determined to be other than temporary, the cost basis of the investment is written down to fair value.
| d) | Allowance for Doubtful Accounts Receivable |
The Company assesses the collectability of its accounts receivable through a review of its current aging, as well as an analysis of its historical collection rate, general economic conditions and credit status of its customers. As of September 30, 2017 and 2016, all outstanding accounts receivable were deemed to be fully collectible, and therefore, no allowance for doubtful accounts was recorded.
| e) | Inventory |
The Company records inventory at the lower of cost or market, with market not in excess of net realizable value. Raw materials are measured using FIFO (first-in first-out) cost. Work in process and finished goods are measured using average cost.
| f) | Property, Plant and Equipment |
Property, plant and equipment are recorded at cost less accumulated depreciation and accumulated impairment losses, if any. Depreciation is recorded on the straight-line method over useful lives ranging from 1.5 to 15 years. Leasehold improvements are depreciated over the shorter of the useful life of the improvement or remaining term of lease. Maintenance and repairs are charged to operations as incurred.
| g) | Impairment of Long-Lived Assets |
If indicators of impairment exist, the Company assesses the recoverability of the affected long-lived assets by determining whether the carrying value of such assets can be recovered through undiscounted future operating cash flows. If impairment is indicated, the amount of such impairment is measured by comparing the carrying value of the asset to the fair value of the asset and the Company records the impairment as a reduction in the carrying value of the related asset and a charge to operating results. Estimating the undiscounted future cash flows associated with long-lived assets requires judgment, and assumptions could differ materially from actual results.
| h) | Fair Value of Financial Instruments |
The Company uses the fair value measurement framework for valuing financial assets and liabilities measured on a recurring basis in situations where other accounting pronouncements either permit or require fair value measurements. See Note 10 for fair value measurements.
| F-9 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
| i) | Revenue Recognition |
Product Sales
The Company recognizes product sales when KLH product is shipped (for which the risk is typically transferred upon delivery to the shipping carrier) and there is persuasive evidence of an arrangement, the fee is fixed or determinable, and collectability is reasonably assured. The Company documents arrangements with customers with purchase orders and sales agreements.
Product sales include sales made under supply agreements with customers for a fixed price per gram of KLH products based on quantities ordered. Supply agreements are typically on a non-exclusive basis except within that customer’s field of use.
Contract services revenue
The Company recognizes contract services revenue when contract services have been performed and reasonable assurance exists regarding measurement and collectability. An appropriate amount will be recognized as revenue in the period that the Company is assured of fulfilling the contract requirements. Amounts received in advance of performance of contract services are recorded as deferred revenue.
Contract services include services performed under collaboration agreements and technology transfer and purchase agreement.
| j) | Research and Development |
Research and development expenses principally consist of personnel costs related to the Company’s research and development staff as well as depreciation of research and development assets. Research and development expenses also include costs incurred for laboratory supplies, KLH designated for internal research use only, reimbursable costs associated with collaborative agreements, third-party contract payments, consultants, facility and related overhead costs. Research and development costs are expensed as incurred.
| k) | Share-Based Compensation |
The Company grants options to buy common shares of the Company to its directors, officers, employees and consultants, and grants other equity-based instruments to non-employees.
The fair value of share-based compensation is measured on the date of grant, using the Black-Scholes option valuation model and is recognized over the vesting period net of estimated forfeitures for employees or the service period for non-employees. The Black-Scholes option valuation model requires the input of subjective assumptions, including price volatility of the underlying stock, risk-free interest rate, dividend yield, and expected life of the option.
| l) | Foreign Exchange |
Items included in the financial statements of the Company’s subsidiaries are measured using the currency of the primary economic environment in which the entity operates (the functional currency). The functional currency of the parent and its subsidiaries is the U.S. dollar.
Transactions in currencies other than the U.S. dollar are recorded at exchange rates prevailing on the dates of the transactions.
| m) | Income Taxes |
Income tax expense comprises current and deferred tax. Income tax is recognized in income or loss except to the extent that it relates to items recognized directly in equity. Current tax expense is the expected tax payable on taxable income for the year, using tax rates enacted or substantively enacted at year-end, adjusted for amendments to tax payable with regards to previous years.
Deferred tax is recorded using the liability method, providing for temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for taxation purposes. Temporary differences are not provided for relating to goodwill not deductible for tax purposes. The amount of deferred tax provided is based on the expected manner of realization or settlement of the carrying amount of assets and liabilities, using tax rates enacted or substantively enacted at the balance sheet date.
| F-10 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
A deferred tax asset is recognized only to the extent that it is more likely than not that future taxable profits will be available against which the asset can be utilized. To the extent that the Company does not consider it more likely than not that a deferred tax asset will be recovered, it provides a valuation allowance against that excess.
The Company periodically evaluates its tax positions to determine whether it is more likely than not that a tax position will be sustained upon examination by the appropriate taxing authorities. The Company has not incurred any interest or penalties as of September 30, 2017 with respect to uncertain income tax matters. The Company does not expect that there will be unrecognized tax benefits of a significant nature that will increase or decrease within 12 months of the reporting date.
The Company files income tax returns in the U.S. federal and state jurisdictions and in Canada. Mexico tax returns are on a calendar year basis. Management believes that there are no material uncertain tax positions that would impact the accompanying consolidated financial statements. The Company's policy is to recognize interest and penalties related to unrecognized tax benefits in income tax expense. The Company may be subject to examination by the Internal Revenue Service for tax years 2013 through 2016 and by the Canada Revenue Agency for tax years 2013 through 2017. The Company may also be subject to examination on certain state, local and other foreign jurisdictions for the tax years 2012 through 2017.
| n) | Earnings (Loss) Per Share |
Basic earnings (loss) per share is calculated by dividing income available to common shareholders by the weighted average number of common shares outstanding during the period.
The computation of diluted earnings (loss) per share assumes the conversion, exercise or contingent issuance of securities only when such conversion, exercise or issuance would have a dilutive effect on earnings (loss) per share. The dilutive effect of convertible securities is reflected in diluted earnings per share by application of the “if converted” method. The dilutive effect of outstanding options and warrants and their equivalents is reflected in diluted earnings per share by application of the treasury stock method. Conversion of outstanding warrants, broker units and options would have an antidilutive effect on loss per share for the years ended September 30, 2017, 2016 and 2015 and are therefore excluded from the computation of diluted loss per share.
| o) | Segments |
The Company operates in one reportable segment and, accordingly, no segment disclosures have been presented. All equipment, leasehold improvements and other fixed assets owned by the Company are physically located within the United States (except for insignificant leasehold improvements under evaluation in Baja California, Mexico), and all supply, collaboration and licensing agreements are denominated in U.S. dollars.
| p) | Recent Accounting Pronouncements |
In May 2014, the Financial Accounting Standards Board (FASB) issued guidance codified in Accounting Standards Codification (ASC) 606 Revenue Recognition – Revenue from Contracts with Customers which amends the guidance in ASC 605, Revenue Recognition and adds a new Subtopic to the Codification, ASC 340-40, Other Assets and Deferred Costs: Contracts with Customers. The standard’s core principle is that a company will recognize revenue when it transfers promised goods or services to customers in an amount that reflects the consideration to which the company expects to be entitled in exchange for those goods or services. To achieve that core principle, an entity should apply the following steps: Step 1: Identify the contract(s) with a customer; Step 2: Identify the performance obligations in the contract; Step 3: Determine the transaction price; Step 4: Allocate the transaction price to the performance obligations in the contract; and Step 5: Recognize revenue when (or as) the entity satisfies a performance obligation. ASC 606 permits two methods of adoption: retrospectively to each prior reporting period presented (full retrospective method), or retrospectively with the cumulative effect of initially applying the guidance recognized at the date of initial application (the modified retrospective method). In August 2015, the FASB issued an accounting update to defer the effective date by one year for public entities such that it is now effective for public entities for annual reporting periods beginning after December 15, 2017, including interim periods within those years, with early application permitted by one year. Subsequently, the FASB issued supplemental adoption guidance and clarification to ASC 606 related to principal vs. agent considerations, identifying performance obligations and licensing, technical corrections and improvements, which must be adopted at the same time as ASC 606. These standards are effective for the Company during the fiscal year ending September 30, 2019. Management is in the process of assessing the impact this guidance will have on the Company’s consolidated financial statements. We anticipate adoption of ASC 606 using the modified retrospective method with a cumulative catch-up adjustment to the opening balance sheet of retained earnings at the effective date, during the first quarter of fiscal 2019. The Company will continue to review separate performance obligations, potential disclosures, and the method of adoption in order to complete the evaluation of the impact on the consolidated financial statements.
| F-11 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
In July 2015, FASB issued ASU 2015-11, Inventory (Topic 330): Simplifying the Measurement of Inventory. ASU 2015-11 indicates that an entity should measure inventory at the lower of cost and net realizable value. Net realizable value is the estimated selling prices in the ordinary course of business, less reasonably predictable costs of completion, disposal, and transportation. The ASU does not apply to inventory measured using LIFO or the retail inventory method. It does apply to all other inventory, including inventory measured using FIFO or average cost. The guidance in ASU 2015-11 is effective for public entities for annual reporting periods beginning after December 15, 2016, including interim periods within those years. The provisions should be applied prospectively with early application permitted as of the beginning of an interim or annual reporting period. These standards are effective for the Company during the fiscal year ending September 30, 2018. Management believes ASU 2015-11 will not have a significant impact on the Company’s consolidated financial statements.
In January 2016, the FASB issued ASU 2016-01, Financial Instruments-Overall (Subtopic 825-10): Recognition and Measurement of Financial Assets and Financial Liabilities, which primarily affects the accounting for equity investments, financial liabilities under the fair value option, and the presentation and disclosure requirements for financial instruments. In addition, ASU 2016-01 clarified guidance related to the valuation allowance assessment when recognizing deferred tax assets resulting from unrealized losses on available-for-sale debt securities. The guidance is effective for public entities for fiscal years beginning after December 15, 2017, including interim periods within those years, with early adoption permitted. These standards are effective for the Company during the fiscal year ending September 30, 2019. Management is in the process of assessing the impact of ASU 2016-01 on the Company’s consolidated financial statements.
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which establishes a new lease accounting model for lessees. The updated guidance requires an entity to recognize assets and liabilities on the balance sheet arising from a lease for both financing and operating leases, along with additional qualitative and quantitative disclosures. The amended guidance is effective for public entities for fiscal years beginning after December 15, 2018, including interim periods within those years, with early adoption permitted. These standards are effective for the Company during the fiscal year ending September 30, 2020. Management is in the process of assessing the impact of ASU 2016-02 on the Company’s consolidated financial statements. We anticipate adoption of ASU 2016-02, will result in lease liabilities and right-of-use assets on the Company’s consolidated financial statements for several long-term operating leases.
In March 2016, the FASB issued ASU 2016-09, Compensation - Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting, which is part of the FASB’s Simplification Initiative. The updated guidance simplifies the accounting for share-based payment transactions, including the income tax consequences, classification of awards as either equity or liabilities, and classification on the statement of cash flows. The amended guidance is effective for public entities for fiscal years beginning after December 15, 2016, including interim periods within those years, with early adoption permitted. These standards are effective for the Company during the fiscal year ending September 30, 2018. Management believes ASU 2016-09 will not have a significant impact on the Company’s consolidated financial statements.
In June 2016, the FASB issued ASU 2016-13, Financial Instruments-Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, which includes provisions that require financial assets measured at amortized cost basis to be presented at the net amount expected to be collected and credit losses relating to available-for-sale debt securities to be recorded through an allowance for credit losses, which requires recognition of an estimate of all current expected credit losses. The guidance is effective for public entities for fiscal years beginning after December 15, 2019, including interim periods within those years, with early adoption permitted for fiscal years beginning after December 15, 2018. These standards are effective for the Company during the fiscal year ending September 30, 2021. Management is in the process of assessing the impact of ASU 2016-13 on the Company’s consolidated financial statements.
In May 2017, the FASB issued ASU 2017-09, Scope of Modification Accounting, which provides new guidance on changes to the terms or conditions of share-based payment awards that would be required to apply modification accounting under ASC 718, Compensation-Stock Compensation. The amendments are effective for annual reporting periods beginning after December 15, 2017 with early adoption permitted. These standards are effective for the Company during the fiscal year ending September 30, 2019. Management is in the process of assessing the impact of ASU 2017-09 on the Company's consolidated financial statements.
| F-12 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
| 4. | Investments |
Short-term investments consisted of the following:
| September 30, | September 30, | |||||||
| 2017 | 2016 | |||||||
| U.S. Treasury Bills | $ | 1,994,401 | $ | 3,988,794 | ||||
U.S. Treasury Bills are carried at amortized cost which approximates fair value and classified as held-to-maturity investments.
| 5. | Inventory |
Raw materials include inventory of manufacturing supplies. Work in process includes manufacturing supplies, direct and indirect labor, contracted manufacturing and testing, and allocated manufacturing overhead for inventory in process at the end of the year. Finished goods include products that are complete and available for sale. At September 30, 2017 and 2016, the Company recorded work in process and finished goods inventory only for those products with recent sales levels to evaluate net realizable value.
Inventory consisted of the following:
| September 30, | September 30, | |||||||
| 2017 | 2016 | |||||||
| Raw materials | $ | 21,761 | $ | 38,764 | ||||
| Work in process | - | 43,498 | ||||||
| Finished goods | 46,353 | 167,168 | ||||||
| $ | 68,114 | $ | 249,430 | |||||
| 6. | Property, Plant and Equipment, net |
Property, plant and equipment, net consisted of the following:
| September 30, | September 30, | |||||||
| 2017 | 2016 | |||||||
| Aquaculture system | $ | 126,257 | $ | 126,257 | ||||
| Laboratory facilities | 62,033 | 62,033 | ||||||
| Computer and office equipment | 117,840 | 102,030 | ||||||
| Tools and equipment | 982,439 | 894,319 | ||||||
| Vehicles | 77,994 | 49,347 | ||||||
| Leasehold improvements | 337,060 | 282,305 | ||||||
| 1,703,623 | 1,516,291 | |||||||
| Less: accumulated depreciation | (969,418 | ) | (793,057 | ) | ||||
| Depreciable assets, net | 734,205 | 723,234 | ||||||
| Construction in progress | 145,318 | 32,880 | ||||||
| $ | 879,523 | $ | 756,114 | |||||
Depreciation expense amounted to $179,322, $149,565 and $159,521 for the years ended September 30, 2017, 2016 and 2015, respectively.
| F-13 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
| 7. | Commitments |
Operating leases
The Company leases buildings and facilities used in its operations under two sublease agreements. In June 2015, the Company exercised its option to extend these sublease agreements for an additional five-year term beginning in October and November 2015. The Company negotiated an option to extend the leases for two additional five-year terms.
The Company leases facilities used for executive offices and laboratories. The Company must pay a portion of the common area maintenance. In July 2016, the Company extended this lease for a two-year term, with options to renew for three successive two-year terms.
The Company leases undeveloped land in Baja California, Mexico to assess the potential development of an additional aquaculture locale and expansion of production. The lease term is three years from June 2015 with options to extend the lease for 30 years. The Company may terminate early with 30 days’ notice. The rent has been prepaid, and is not included in the future minimum lease payments below. The Company has a related agreement with the lessor to collaborate on the design, expansion and development of marine aquaculture resources and KLH production facilities on the leased property. Under that agreement, the Company is responsible for certain leasehold improvements including construction of structures and a power-generating facility, which will be owned by the Company. The Company will reimburse the lessor for local operational support. The collaboration agreement expires in June 2018, unless terminated earlier.
Aggregate future minimum lease payments at September 30, 2017 are as follows:
| For The Year Ending September 30, | ||||
| 2018 | 160,000 | |||
| 2019 | 106,000 | |||
| 2020 | 106,000 | |||
| 2021 | 6,000 | |||
| $ | 378,000 | |||
Rent expense on these lease agreements amounted to approximately $238,000, $235,000 and $192,000 for the years ended September 30, 2017, 2016 and 2015, respectively.
Purchase obligations
The Company has commitments totaling approximately $252,000 at September 30, 2017, for signed agreements with contract research organizations, consultants and construction contractors. All purchase obligations are expected to be fulfilled within the next 12 months, except for approximately $65,100, which is expected to be fulfilled in the following fiscal year.
Supply agreements
The Company has commitments under supply agreements with customers for fixed prices per gram of KLH in connection with clinical trials on a non-exclusive basis except within that customer’s field of use. The expiration dates of these supply agreements range from October 2019 to February 2022, and are generally renewable upon written request of the customer.
Joint venture agreement
In May 2016, the Company entered into a joint venture agreement with another party for the formation of a joint venture company to manufacture and sell conjugated therapeutic vaccines. The joint venture is organized as a French simplified corporation.
| F-14 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
The Company holds a 30% equity interest in the joint venture in exchange for an initial capital contribution of €120,000. One-half of the initial contribution, approximately $67,000, was paid during the year ended September 30, 2016 with the balance due upon the occurrence of certain defined future events. The Company will also provide the joint venture additional financing as may be required, on a pro rata basis in line with our equity interest. If the joint venture does not achieve certain milestones by December 31, 2017, the joint venture will be dissolved, unless (i) the parties mutually agree to pursue the joint venture arrangement, or (ii) either party decides to purchase the equity interests of the other party. These milestones have not been achieved, and the parties have discussed their mutual desire to extend the deadline. Each of the parties is entitled, upon the occurrence of certain defined events, to acquire the interest of the other party. Except as described herein, the joint venture has an initial ten-year term, renewable for successive five-year terms. If either party provides notice at least six months prior to the expiration date of an applicable term that it does not wish to continue its participation in the joint venture, the other party will have a right to acquire all of such terminating party’s equity interests in the joint venture.
In connection with the formation of the joint venture and the execution of its strategy, the parties intend over time to enter into an exclusive supply agreement within a limited field of use for Stellar to supply KLH to the joint venture, a supply agreement designating the joint venture as the exclusive manufacturer and supplier of the other party’s vaccines, and services agreements for the provision of various knowledge and expertise by each of the parties.
Licensing agreement and technology transfer agreement
In July 2013, the Company acquired the exclusive, worldwide license to certain patented technology for the development of human immunotherapies against Clostridium difficile infection (C. diff) under a written agreement (the License Agreement) with a University (the Licensor). Annual license fees of $20,000 each were paid for the years ended September 30, 2016 and 2015. The Company also reimbursed patent filing, prosecution, and maintenance costs of approximately $12,000, $11,000 and $52,000 for the years ended September 30, 2017, 2016 and 2015, respectively. License fees and patent cost reimbursements have been accounted for as research and development expense in the accompanying consolidated statements of operations.
In March 2017, (i) the Company entered into an agreement to terminate the License Agreement, (ii) the Company concurrently entered into a technology transfer and purchase agreement (the Transfer Agreement) with a vaccine biotechnology company (the Transferee), and (iii) the Licensor and Transferee entered into a direct licensing arrangement relating to the patented C. diff technology. Under the Transfer Agreement, the Company transferred to the Transferee its proprietary rights and know-how of immunogens and vaccine technology for C. diff, in exchange for an upfront payment and a percentage of future fees, milestone payments, sublicensing income and royalties, if any, paid by the Transferee or its assigns to the Licensor.
As a result of the termination of the License Agreement, there are no early termination penalties and no further annual licensing fees, contingent milestone payments, royalties, sub-licensing fees or other financial obligations payable by the Company to the Licensor.
Retirement savings plan 401(k) contributions
The Company sponsors a 401(k) retirement savings plan that requires an annual non-elective safe harbor employer contribution of 3% of eligible employee wages. All employees over 21 years of age are eligible beginning the first payroll after 3 consecutive months of employment. Employees are 100% vested in employer contributions and in any voluntary employee contributions. Contributions to the 401(k) plan were approximately $62,000, $64,000 and $58,000 for the years ended September 30, 2017, 2016 and 2015, respectively.
Related party commitments:
Patent royalty agreement
On August 14, 2002, through its California subsidiary, the Company entered into an agreement with a director and officer of the Company, where he would receive royalty payments in exchange for assignment of his patent rights to the Company. The royalty is 5% of gross receipts from products using this invention in excess of $500,000 annually. The Company’s current operations utilize this invention. Royalty expense incurred during the years ended September 30, 2016 and 2015 was approximately $35,500 and $1,500. There was no royalty expense incurred during the year ended September 30, 2017.
| F-15 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
Collaboration agreement
In December 2013, the Company entered into a collaboration agreement with a privately-held Taiwanese biopharmaceuticals manufacturer which expired in accordance with its terms in December 2015. Under the terms of the agreement, the Company was responsible for the production and delivery of GMP grade KLH for evaluation as a carrier molecule in the collaboration partner’s potential manufacture of OBI-822 (Adagloxad Simolenin) active immunotherapy. The Company was also responsible for method development, product formulation, and process qualification for certain KLH reference standards. The collaboration partner was responsible for development objectives and product specifications. The agreement provided for the collaboration partner to pay fees for certain expenses and costs associated with the collaboration. Subject to certain conditions and timing, the collaboration also provided for the parties to negotiate a commercial supply agreement for Stellar KLH which was executed in February 2017.
A member of the Company’s Board of Directors currently serves as the manufacturer’s general manager and chair of its board of directors.
| 8. | Share Capital |
The Company had the following transactions in share capital:
| Years Ended | ||||||||||||
| September 30, | September 30, | September 30, | ||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Number of common shares issued | 383,838 | 2,151,500 | 42,773 | |||||||||
| Issuance of common shares | $ | - | $ | 6,750,000 | $ | - | ||||||
| Share issuance costs | - | (805,264 | ) | - | ||||||||
| Proceeds from exercise of warrants | - | 1,368,260 | 12,609 | |||||||||
| Transfer to common shares on issuance of performance shares | 1,070,909 | - | - | |||||||||
| Transfer to common shares on exercise of warrants | - | 1,853,581 | 10,000 | |||||||||
| Proceeds from exercise of options | - | - | 94,168 | |||||||||
| Transfer to common shares on exercise of options | - | - | 113,561 | |||||||||
| Share-based compensation | 115,546 | 259,379 | 267,222 | |||||||||
Reverse Share Split
On September 2, 2015, the Company effected a share consolidation (reverse split) of the Company's common shares at a ratio of 1-for-10. As a result of the reverse split, every ten shares of the issued and outstanding common shares, without par value, consolidated into one newly-issued outstanding common share, without par value. Each fractional share remaining after the reverse split that was less than one-half of a share was cancelled and each fractional share that was at least one-half of a share was changed to one whole share. The number of warrants, broker units, and options were proportionately adjusted by the split ratio and the exercise prices correspondingly increased by the same split ratio. All historical shares and exercise prices are presented on a post-split basis in these consolidated financial statements.
Performance Shares
Pursuant to a performance share plan approved by shareholders in 2010, 1,000,000 common shares were reserved for issuance to certain officers, directors and employees of the Company upon achievement of certain milestones related to completion of method development for commercial-scale manufacture of KLH, compilation and regulatory submittal of all required chemistry, manufacturing and control data and completion of preclinical toxicity and immunogenicity testing of products. Share-based compensation was recorded over the estimated vesting period ending in August 2012.
At September 30, 2017, all vested performance shares under the plan have been issued, and the performance share plan was terminated.
Black-Scholes option valuation model
The Company uses the Black-Scholes option valuation model to determine the fair value of warrants, broker units and share options. Option valuation models require the input of highly subjective assumptions including the expected price volatility. The Company has used historical volatility to estimate the volatility of the share price. Changes in the subjective input assumptions can materially affect the fair value estimates, and therefore the existing models do not necessarily provide a reliable single measure of the fair value of the Company’s warrants, broker units and share options.
| F-16 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
Warrants
A summary of the Company’s warrants activity is as follows:
| Number of Warrants | Weighted Average Exercise Price | ||||||||
| Balance - September 30, 2015 | 1,022,761 | $ | 9.04 | ||||||
| Granted | 1,265,626 | 4.50 | |||||||
| Granted | 40,000 | 4.00 | CDN $ | ||||||
| Exercised | (424,000 | ) | 4.00 | CDN $ | |||||
| Expired | (598,761 | ) | 13.33 | ||||||
| Expired | (40,000 | ) | 4.00 | CDN $ | |||||
| Balance - September 30, 2016 and 2017 | 1,265,626 | $ | 4.50 | ||||||
There are no outstanding warrants with exercise prices denominated in Canadian dollar at September 30, 2017.
The weighted average contractual life remaining on the outstanding warrants at September 30, 2017 is 51 months.
The following table summarizes information about the warrants outstanding at September 30, 2017:
| Exercise Price | Number of Warrants | Expiry Date | ||||||
| $ | 4.50 | 1,265,626 | January 6, 2022 | |||||
| 1,265,626 | ||||||||
Warrant Liability
All warrants with exercise prices denominated in Canadian dollars were exercised or expired. Therefore, there is no outstanding warrant liability at September 30, 2017.
Equity offerings conducted by the Company in prior years included the issuance of warrants with exercise prices denominated in Canadian dollars. The Company’s functional currency is the U.S. dollar. As a result of having exercise prices denominated in other than the Company’s functional currency, those warrants met the definition of derivatives and were therefore classified as derivative liabilities measured at fair value with adjustments to fair value recognized through the consolidated statements of operations. The fair value of those warrants was determined using the Black-Scholes option valuation model at the end of each reporting period. On the date those warrants were exercised, the fair value of warrant liability was reclassified to common shares along with the proceeds from the exercise. If those warrants expired, the related decrease in warrant liability was recognized in profit or loss, as part of the change in fair value of warrant liability. There was no cash flow impact as a result of this accounting treatment.
The fair value of warrants exercised was determined using the Black-Scholes option valuation model, using the following weighted average assumptions:
| Years Ended | ||||||||
| September 30, | September 30, | |||||||
| 2016 | 2015 | |||||||
| Risk free interest rate | 0.48 | % | 0.44 | % | ||||
| Expected life (years) | 0.04 | 0.40 | ||||||
| Expected share price volatility | 92 | % | 92 | % | ||||
There were no warrants exercised during the year ended September 30, 2017.
| F-17 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
The fair value of warrants granted was determined using the Black-Scholes option valuation model, using the following weighted average assumptions at the date of the grant:
| Year Ended | ||||
| September 30, | ||||
| 2016 | ||||
| Risk free interest rate | 0.52 | % | ||
| Expected life (years) | 0.01 | |||
| Expected share price volatility | 91 | % | ||
| Expected dividend yield | 0 | % | ||
There were no warrants granted during the years ended September 30, 2017 or 2015.
Broker units
The Company granted broker units as finders’ fees in conjunction with equity offerings in prior years. Broker units were fully vested when granted and allowed the holders to purchase equity units. A unit consisted of one common share and either one whole warrant or one half warrant.
A summary of broker units activity is as follows:
| Number of Units | Weighted Average Exercise Price | ||||||||
| Balance - September 30, 2015 | 46,600 | $ | 1.87 | ||||||
| Exercised | (40,000 | ) | 2.50 | CDN $ | |||||
| Expired | (6,600 | ) | 2.50 | CDN $ | |||||
| Balance - September 30, 2016 and 2017 | - | $ | - | ||||||
There were no broker units granted during the years ended September 30, 2017, 2016 and 2015.
Options
The Company has an incentive compensation plan adopted in 2017 (the Plan) administered by the Board of Directors, which amended and restated the 2013 fixed share option plan (the 2013 Plan). Options, restricted shares and restricted share units are eligible for grants under the Plan. The number of shares available for issuance under the Plan is 1,597,000, including shares available for the exercise of outstanding options under the 2013 Plan. No restricted shares or restricted share units have been granted as of September 30, 2017.
The exercise price of an option is set at the closing price of the Company’s common shares on the date of grant. Share options granted to directors, officers, employees and certain individual consultants for past service are subject to the following vesting schedule: (a) one-third shall vest immediately, (b) one-third shall vest at 12 months from the date of grant and (c) one-third shall vest at 18 months from the date of grant.
Share options granted to directors, officers, employees and certain individual consultants for future service are subject to the following vesting schedule: (x) one-third shall vest at 12 months from the date of grant, (y) one-third shall vest at 24 months from the date of grant and (z) one-third shall vest at 36 months from the date of grant.
| F-18 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
Share options granted to certain individual investor relations consultants are subject to the following vesting schedule: (aa) 25% shall vest at 3 months from the date of grant, (bb) 25% shall vest at 6 months from the date of grant, (cc) 25% shall vest at 12 months from the date of grant and (dd) 25% shall vest at 15 months from the date of grant.
Options have been granted under the Plan allowing the holders to purchase common shares of the Company as follows:
| Number of Options | Weighted Average Exercise Price | ||||||||
| Balance - September 30, 2015 | 557,638 | $ | 5.17 | ||||||
| Granted | 56,300 | 6.47 | |||||||
| Expired | (21,334 | ) | 10.70 | ||||||
| Expired | (53,501 | ) | 5.22 | CDN $ | |||||
| Balance - September 30, 2016 | 539,103 | $ | 5.29 | ||||||
| Granted | 71,600 | 1.89 | |||||||
| Expired | (28,233 | ) | 11.14 | ||||||
| Expired | (171,500 | ) | 2.90 | CDN $ | |||||
| Balance - September 30, 2017 | 410,970 | $ | 5.74 | ||||||
The weighted average contractual life remaining on the outstanding options is 35 months.
The following table summarizes information about the options under the Plan outstanding and exercisable at September 30, 2017:
| Number of Options | Exercisable at September 30, 2017 | Range of exercise prices | Expiry Dates | |||||||
| 115,110 | 115,110 | CDN$0.01 - 5.00 | Apr 2017-Dec 2019 | |||||||
| 79,900 | 17,733 | $0.01 - 5.00 | Sep 2023-Mar 2024 | |||||||
| 139,860 | 139,860 | CDN$5.01 - 10.00 | Oct 2017-Jun 2022 | |||||||
| 15,100 | 15,100 | $5.01 - 10.00 | Dec 2022 | |||||||
| 21,500 | 21,500 | CDN$15.01 - 20.00 | Nov 2018-Nov 2021 | |||||||
| 39,500 | 39,500 | $15.01 - 20.00 | Nov 2020 | |||||||
| 410,970 | 348,803 | |||||||||
The estimated fair value of the share options granted was determined using a Black-Scholes option valuation model with the following weighted average assumptions:
| Years Ended | ||||||||||||
| September 30, | September 30, | September 30, | ||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| Risk free interest rate | 1.44 | % | 1.01 | % | 1.65 | % | ||||||
| Expected life (years) | 7.00 | 7.00 | 7.00 | |||||||||
| Expected share price volatility | 166 | % | 117 | % | 115 | % | ||||||
| Expected dividend yield | 0 | % | 0 | % | 0 | % | ||||||
The weighted average fair value of share options granted during the years ended September 30, 2017, 2016 and 2015 was $1.84, $5.56, and $9.48, respectively.
As of September 30, 2017, the Company had approximately $62,000 of unrecognized share-based compensation expense, which is expected to be recognized over a period of 30 months.
| F-19 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
The intrinsic value of the options exercised during the year ended September 30, 2015 was $8.28. There were no options exercised during the years ended September 30, 2017 and 2016. There was no intrinsic value of the vested options at September 30, 2017.
| 9. | Income Taxes |
The breakdown of loss before income tax by jurisdiction is as follows:
| Years Ended | ||||||||||||
| September 30, | September 30, | September 30, | ||||||||||
| 2017 | 2016 | 2015 | ||||||||||
| U.S. | $ | (4,540,094 | ) | $ | (4,001,206 | ) | $ | (3,258,355 | ) | |||
| Canadian | (464,990 | ) | (1,026,520 | ) | 405,203 | |||||||
| Other foreign | (24,764 | ) | 8,846 | 46,923 | ||||||||
| Total Loss Before Income Tax | $ | (5,029,848 | ) | $ | (5,018,880 | ) | $ | (2,806,229 | ) | |||
Deferred income tax assets and liabilities of the Company are as follows:
| September 30, 2017 | September 30, 2016 | September 30, 2015 | ||||||||||
| Deferred income tax assets: | ||||||||||||
| Non-capital loss carry-forwards | $ | 12,164,100 | $ | 10,000,000 | $ | 8,028,900 | ||||||
| Research and development tax credits | 947,300 | 808,000 | 716,400 | |||||||||
| Deferred expenses | 34,300 | 70,000 | 82,900 | |||||||||
| Property, plant and equipment | 2,200 | 400 | 1,700 | |||||||||
| Share issuance costs | 142,600 | 207,200 | 67,800 | |||||||||
| Deferred income tax liabilities: | ||||||||||||
| U.S. federal benefit net of state taxes | (923,700 | ) | (764,500 | ) | (628,800 | ) | ||||||
| Valuation allowance | (12,366,800 | ) | (10,321,100 | ) | (8,268,900 | ) | ||||||
| Net deferred income tax asset (liability) | $ | - | $ | - | $ | - | ||||||
Realization of the deferred tax assets is dependent upon the generation of future taxable income, the amount and timing of which are uncertain. Accordingly, the net deferred tax assets have been fully offset by a valuation allowance.
As of September 30, 2017, the Company had federal net operating loss (NOL) carryforwards of approximately $25.3 million expiring 2030 through 2037, California NOL carryforwards of approximately $25.0 million expiring 2018 through 2037, and Canadian federal and provincial NOL carryforwards of approximately CDN$6.8 million expiring 2028 through 2037. Portions of these NOL carryforwards may be used to offset future taxable income, if any.
As of September 30, 2017, the Company also has federal and California research and development tax credit carryforwards of approximately $.45 million and $.50 million, respectively, available to offset future taxes. The federal credits begin expiring in 2030 and continue expiring through 2037. The state tax credits do not expire.
Under the provisions of Section 382 of the Internal Revenue Code, substantial changes in the Company's ownership limit the amount of net operating loss carryforwards and tax credit carryforwards that can be utilized annually in the future to offset taxable income. A valuation allowance has been established to reserve the potential benefits of these carryforwards in the Company's consolidated financial statements to reflect the uncertainty of future taxable income required to utilize available tax loss carryforwards and other deferred tax assets.
| F-20 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
The recovery of income taxes shown in the consolidated statements of operations differs from the amounts obtained by applying statutory rates to the loss before provision for income taxes due to the following:
| September 30, 2017 | September 30, 2016 | September 30, 2015 | ||||||||||
| Combined Canadian federal and provincial tax rates | 26.0 | % | 26.0 | % | 26.0 | % | ||||||
| Expected income tax (recovery)/expense | $ | (1,307,800 | ) | $ | (1,304,900 | ) | $ | (729,600 | ) | |||
| Nondeductible share-based payments | 30,000 | (67,400 | ) | 69,500 | ||||||||
| Nondeductible change in fair value of warrant liability | - | (55,100 | ) | (554,100 | ) | |||||||
| Effect of higher income tax rate in U.S. | (624,400 | ) | (550,600 | ) | (445,800 | ) | ||||||
| Foreign currency differences | (42,200 | ) | 20,000 | 169,900 | ||||||||
| Other | (174,500 | ) | (2,800 | ) | (43,300 | ) | ||||||
| Change in valuation allowance on deferred tax assets | 2,119,700 | 1,968,000 | 1,570,200 | |||||||||
| Income tax expense | $ | 800 | $ | 7,200 | $ | 36,800 | ||||||
The components of income tax provision (benefits) are as follows:
| September 30, 2017 | September 30, 2016 | September 30, 2015 | ||||||||||
| Current tax provision | ||||||||||||
| U.S. federal | $ | - | $ | - | $ | - | ||||||
| Canadian | - | - | - | |||||||||
| Other foreign | - | 6,400 | 36,000 | |||||||||
| State | 800 | 800 | 800 | |||||||||
| Deferred tax provision | ||||||||||||
| U.S. federal | (1,447,100 | ) | (1,265,700 | ) | (1,032,200 | ) | ||||||
| Canadian | (199,100 | ) | (303,300 | ) | (209,300 | ) | ||||||
| Other foreign | (5,200 | ) | - | - | ||||||||
| State | (468,300 | ) | (399,000 | ) | (328,700 | ) | ||||||
| Change in valuation allowance on deferred tax assets | 2,119,700 | 1,968,000 | 1,570,200 | |||||||||
| Total | $ | 800 | $ | 7,200 | $ | 36,800 | ||||||
| 10. | Fair Value of Financial Instruments |
The Company uses the fair value measurement framework for valuing financial assets and liabilities measured on a recurring basis in situations where other accounting pronouncements either permit or require fair value measurements.
Fair value of a financial instrument is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. The carrying value of certain financial instruments such as accounts receivable, accounts payable, accrued liabilities, and deferred revenue approximates fair value due to the short-term nature of such instruments. Short-term investments in U.S. Treasury Bills are recorded at amortized cost, which approximates fair value.
| F-21 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
The Company follows the fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. There are three levels of inputs that may be used to measure fair value:
| Level 1: | Quoted prices in active markets for identical or similar assets and liabilities. | |
| Level 2: | Quoted prices for identical or similar assets and liabilities in markets that are not active or observable inputs other than quoted prices in active markets for identical or similar assets and liabilities. | |
| Level 3: | Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. |
The Company reports its short-term investments in U.S. Treasury Bills at fair value using Level 1 inputs in the fair value hierarchy.
The following table summarizes fair values for those assets and liabilities with fair value measured on a recurring basis.
| Fair Value Measurements Using | ||||||||||||||||
| Quoted Prices in Active Markets for Identical Instruments (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Total Fair Value | |||||||||||||
| September 30, 2017 | ||||||||||||||||
| Assets | ||||||||||||||||
| Short-term investments in U.S. Treasury Bills | $ | 1,994,401 | $ | - | $ | - | $ | 1,994,401 | ||||||||
| September 30, 2016 | ||||||||||||||||
| Assets | ||||||||||||||||
| Short-term investments in U.S. Treasury Bills | $ | 3,988,794 | $ | - | $ | - | $ | 3,988,794 | ||||||||
| 11. | Concentrations of Credit Risk |
Credit risk is the risk of an unexpected loss if a customer or third party to a financial instrument fails to meet its contractual obligations. Financial instruments that potentially subject the Company to a concentration of credit risk consist primarily of cash and cash equivalents, U.S Treasury Bills, and accounts receivable. The Company estimates its maximum credit risk at the amount recorded on the balance sheet.
Management’s assessment of the Company’s credit risk for cash and cash equivalents is low as they are held in major financial institutions believed to be credit worthy or U.S. Treasury Bills with maturities of 90 days or less. The Company limits its exposure to credit loss for short-term investments by holding U.S. Treasury Bills with maturities of 1 year or less. Based on credit monitoring and history, the Company considers the risk of credit losses due to customer non-performance on accounts receivable to be low.
| F-22 |
| Stellar Biotechnologies, Inc. |
| Notes to Consolidated Financial Statements |
| For the Years Ended September 30, 2017, 2016 and 2015 |
The Company had the following concentrations of revenues by customers, each of which accounted for more than 10% of revenues in the applicable period:
| Years Ended | ||||||
| September 30, | September 30, | September 30, | ||||
| 2017 | 2016 | 2015 | ||||
| Product sales and contract services revenue | 79% from 2 customers |
76% from 3 customers |
86% from 5 customers | |||
The Company had the following concentrations of revenues by geographic areas:
| Years Ended | ||||||||||
| September 30, | September 30, | September 30, | ||||||||
| 2017 | 2016 | 2015 | ||||||||
| Europe | 64 | % | 43 | % | 53 | % | ||||
| North America | 33 | % | 12 | % | 9 | % | ||||
| Asia | 3 | % | 45 | % | 38 | % | ||||
The Company had the following concentrations of accounts receivable from its customers, each of which accounted for more than 10% in the applicable period:
| September 30, | ||
| 2016 | ||
| Accounts receivable | 100% from 1 customer |
There were no customer accounts receivable at September 30, 2017.
| F-23 |
