Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - ALIMERA SCIENCES INC | pr.htm |
| 8-K - 8-K - ALIMERA SCIENCES INC | alim8k.htm |

Third Quarter 2017
Results Conference Call
Thursday, November 2, 2017
Exhibit 99.2

Safe Harbor Statement
This presentation contains “forward-looking statements,” within the meaning of the Private Securities Litigation Reform Act of 1995,
regarding, among other things, the opportunity for further growth in 2017 for ILUVIEN and the potential approval and commercialization of
ILUVIEN for the treatment of posterior uveitis in Europe. Such forward-looking statements are based on current expectations and involve
inherent risks and uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ
materially from those projected in its forward-looking statements. Words such as “anticipate,” “believe,” “estimate,” “expect,” “intend,”
“may,” “plan,” “contemplate,” “predict,” “project,” “target,” “likely,” “potential,” “continue,” “ongoing,” “will,” “would,” “should,” “could,”
or the negative of these terms and similar expressions are intended to identify forward-looking statements, although not all forward-looking
statements contain these identifying words. Such forward-looking statements are based on current expectations and involve inherent risks
and uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ materially from
those projected in its forward-looking statements. Meaningful factors which could cause actual results to differ include, but are not limited
to, continued market acceptance of ILUVIEN in the U.S. and Europe, including physicians' ability to obtain reimbursement, as well as other
factors discussed in the “Risk Factors” and “Management's Discussion and Analysis of Financial Condition and Results of Operations” sections
of Alimera's Annual Report on Form 10-K for the year ended December 31, 2016 and for the quarter ended June 30, 2017, which are on file
with the Securities and Exchange Commission (SEC) and available on the SEC's website at http://www.sec.gov. Additional factors may also be
set forth in those sections of Alimera’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2017, to be filed with the SEC in
the fourth quarter of 2017. In addition to the risks described above and in Alimera's Annual Report on Form 10-K, Quarterly Reports on Form
10-Q, Current Reports on Form 8-K and other filings with the SEC, other unknown or unpredictable factors also could affect Alimera's results.
There can be no assurance that the actual results or developments anticipated by Alimera will be realized or, even if substantially realized,
that they will have the expected consequences to, or effects on, Alimera. Therefore, no assurance can be given that the outcomes stated in
such forward-looking statements and estimates will be achieved. All forward-looking statements contained in this presentation are expressly
qualified by the cautionary statements contained or referred to herein. Alimera cautions investors not to rely too heavily on the forward-
looking statements Alimera makes or that are made on its behalf. These forward-looking statements speak only as of the date of this
presentation (unless another date is indicated). Alimera undertakes no obligation, and specifically declines any obligation, to publicly update
or revise any such forward-looking statements, whether as a result of new information, future events or otherwise.
2 © 2017 Alimera Sciences, Inc., All Rights Reserved

$8.3
M
$9.8
M
3Q16 3Q17
3
Third Quarter 2017 Highlights
Revenue Growth
• 18% revenue growth compared to the
third quarter of 2016.
• Partnership announced in France; first
units sold in Italy.
• Rights to posterior uveitis secured for
ILUVIEN in the EU, Middle East &
Africa.
18%
© 2017 Alimera Sciences, Inc., All Rights Reserved

USER Study Overview
4 © 2017 Alimera Sciences, Inc., All Rights Reserved
Center Patients Treated, n (%) Eyes Treated, n
All Centers 130 (100%) 160
Cincinnati Eye 37 (28.5%) 49
Georgia Retina 15 (11.5%) 19
Retina Health Institute 53 (40.8%) 59
Southern Eye 25 (19.2%) 33
Patients must have received ILUVIEN prior to January 1, 2016 for the treatment of DME in at least one eye.
Determine correlation in
ocular outcomes to DME
treatments prior and post
ILUVIEN administration
Compare number of DME
treatments before and
after ILUVIEN
administration
Compare IOP signals
associated with ILUVIEN to
IOP signals reported in
FAME.
Objectives

Change in Visual Acuity,
Entire Population
5 © 2017 Alimera Sciences, Inc., All Rights Reserved

6 © 2017 Alimera Sciences, Inc., All Rights Reserved
Minimal IOP Increase

7 © 2017 Alimera Sciences, Inc., All Rights Reserved
IOP Related Events - DME
(N eyes = 160)
IOP-Related Event
Before ILUVIEN
Administration
After ILUVIEN
Administration
P-Value
N % N %
IOP Elevation to over 21 mmHg 61 38.1% 49 30.6% 0.195
IOP Elevation to over 25 mmHg 24 15.0% 24 15.0% 1.000
IOP Elevation to over 30 mmHg 9 5.6% 8 5.0% 1.000
Trabeculoplasty 3 1.9% 2 1.3% 1.000
Trabeculectomy 0 0.0% 0 0.0% NA
Incisional IOP-Lowering Surgery 2 1.3% 2 1.3% 1.00
IOP elevation increase of 10 mmHg or
more after ILUVIEN
NA NA 29 18.1% 1.00

8 © 2017 Alimera Sciences, Inc., All Rights Reserved
Expect approval
during 2018 and
expect to begin
marketing ILUVIEN
for Posterior Uveitis
in 2019
Fluocinolone Acetonide for Non-Infectious Uveitis
Affecting The Posterior Segment (NIU-PS)
Primary End Point at 6
Months – Reduction in the
Recurrence of Uveitis
Sham Injection
(N = 94)
69 (73.4%)
FAc Insert
(N = 188)
50 (26.6%)
73.4
%
26.6
%
Sham Fluocinolone
Acetonide
Percentage of Patients with
Uveitis Recurrence
Pooled Data

IOP-Related Event FAc Insert (N=188) Sham Injection (N=94)
N % N %
IOP Elevation to over 25 mmHg 36 19.1% 7 (7.4%)
IOP Elevation to over 30 mmHg 22 11.7% 3 3.2%
IOP-lowering medication 42 22.3% 14 14.9%
IOP-lowering Surgery 3 1.6% 2 2.1%
Cataract Surgery (N/Phakic) 17/103 16.5% 4/56 7.1%
9
Expect approval
during 2018 and
expect to begin
marketing ILUVIEN
for Posterior Uveitis
in 2019
IOP Related Events – Posterior Uveitis
© 2017 Alimera Sciences, Inc., All Rights Reserved

10
Uveitis Indication for ILUVIEN
© 2017 Alimera Sciences, Inc., All Rights Reserved
• Announced acquisition of
rights for Posterior Uveitis for
ILUVIEN in Europe, the Middle
East and Africa
• Filing for label variation with
the MHRA in the 17 EU
countries where ILUVIEN is
approved in the first quarter of
2018
Expect approval
during 2018 and
expect to begin
marketing ILUVIEN
for Posterior Uveitis
in 2019
First European
Approval Expected
2018
Plan to Begin Marketing
Where Approved
2019

Financial Overview
Rick Eiswirth, President & CFO

$8.3M
$2.1M
$6.2M
$9.8M
$2.6M
$7.1M
Global Revenue International Revenue U.S. Revenue
12
3Q17 Financial Overview
+18%
+24%
+15%
© 2017 Alimera Sciences, Inc., All Rights Reserved
3Q17 3Q16
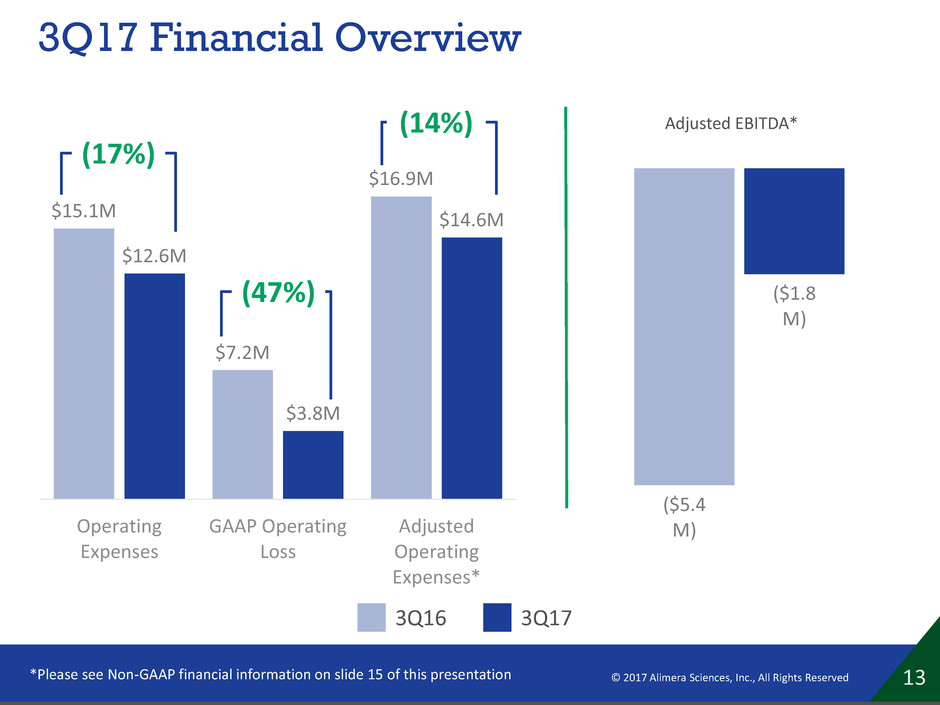
$15.1M
$7.2M
$16.9M
$12.6M
$3.8M
$14.6M
Operating
Expenses
GAAP Operating
Loss
Adjusted
Operating
Expenses*
13 © 2017 Alimera Sciences, Inc., All Rights Reserved *Please see Non-GAAP financial information on slide 15 of this presentation
($5.4
M)
($1.8
M)
Adjusted EBITDA*
(17%)
(47%)
(14%)
3Q17 Financial Overview
3Q17 3Q16

14
Closing Remarks
© 2017 Alimera Sciences, Inc., All Rights Reserved
• Continue to focus on profitable revenue growth.
• USER Study data expected to drive further acceptance of
ILUVIEN in the U.S.
• Continue to build product pipeline for ILUVIEN with first
approvals of posterior uveitis expected during 2018 in Europe.

15
Non-GAAP Financial Information
© 2017 Alimera Sciences, Inc., All Rights Reserved
Alimera believes that the non-GAAP financial information provided in this presentation can assist investors in the overall understanding of its
financial performance when considered together with GAAP figures.
This presentation contains a discussion of certain non-GAAP financial measures, as defined in Regulation G of the Securities Exchange Act of
1934, as amended. Alimera reports its financial results in compliance with GAAP, but believes that the non-GAAP measures of Adjusted
EBITDA and Adjusted Operating Expenses will be a more relevant measure of Alimera's operating performance. For the purpose of this
presentation, “Adjusted EBITDA” is adjusted earnings before interest, taxes, depreciation, amortization, non-cash stock-based compensation
expense, and to the extent they are included in the calculation of earnings, net unrealized gain (loss) from foreign currency exchange
transactions and gains (losses) from the change in the fair value of derivative warrant liability. “Adjusted Operating Expenses” is operating
expenses excluding depreciation, amortization and non-cash stock-based compensation expense. Alimera uses Adjusted EBITDA and Adjusted
Operating Expenses in the management of its business and Alimera's lender uses Adjusted EBITDA as a financial covenant measurement.
Accordingly, Adjusted EBITDA and Adjusted Operating Expenses for the second quarter of 2017 have been presented in certain instances
excluding items identified in the reconciliations provided.
These non-GAAP financial measures, as presented, may not be comparable to similarly titled measures reported by other companies since not
all companies may calculate these measures in an identical manner and, therefore, they are not necessarily an accurate measure of
comparison between companies.
The presentation of these non-GAAP financial measures is not intended to be considered in isolation or as a substitute for guidance prepared
in accordance with GAAP. The principal limitation of these non-GAAP financial measures is that they exclude significant elements that are
required by GAAP to be recorded in Alimera's financial statements. In addition, they are subject to inherent limitations as they reflect the
exercise of judgments by management in determining these non-GAAP financial measures. In order to compensate for these limitations,
Alimera presents its non-GAAP financial results in connection with its GAAP results. Investors are encouraged to review the reconciliation of
our non-GAAP financial measures to their most directly comparable GAAP financial measure.

Third Quarter 2017
Results Conference Call
Thursday, November 2, 2017
