Attached files
| file | filename |
|---|---|
| EX-32.2 - TearLab Corp | ex32-2.htm |
| EX-32.1 - TearLab Corp | ex32-1.htm |
| EX-31.2 - TearLab Corp | ex31-2.htm |
| EX-31.1 - TearLab Corp | ex31-1.htm |
| EX-23.1 - TearLab Corp | ex23-1.htm |
| EX-10.29 - TearLab Corp | ex10-29.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
| [X] | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2016
Or
| [ ] | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from _________________ to _________________
Commission file number: 000-51030
TearLab Corp.
(Exact name of registrant as specified in its charter)
| Delaware | 59-3434771 | |
| (State
or other jurisdiction of incorporation or organization) |
(I.R.S.
Employer Identification Number) |
9980 Huennekens St., Suite 100 San Diego, California |
92121 | |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: (858) 455-6006
SECURITIES REGISTERED PURSUANT TO SECTION 12(b) OF THE ACT:
| Title of each class | Name of each exchange on which registered | |
COMMON
STOCK, $0.001 PAR VALUE |
1The Nasdaq Stock Market LLC (The Nasdaq Capital Market) |
SECURITIES REGISTERED PURSUANT TO SECTION 12(G) OF THE ACT: NONE
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes [ ] No [X]
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes [ ] No [X]
Indicate by check mark whether the Registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes [X] No [ ]
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes [X] No [ ]
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of the Registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. [X]
Indicate by check mark whether the Registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer [ ] | Accelerated filer [ ] | Non-accelerated filer [ ] | Smaller reporting company [X] |
| (Do not check if a smaller reporting company) | |||
Indicate by check mark if the Registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). [ ] No [X]
The aggregate market value of the voting common stock held by non-affiliates of the Registrant, based on the closing sale price of the Registrant’s common stock on June 30, 2016, the last business day of the Registrant’s most recently completed second fiscal quarter, as reported on the NASDAQ Capital Market on June 30, 2016 was approximately $32.7 million. The Registrant has no non-voting common stock. Shares of common stock held by each executive officer and director and by each person who owns 10% or more of the outstanding common stock, based on filings with the Securities and Exchange Commission, have been excluded from this computation since such persons may be deemed to be affiliates of the Registrant. The determination of affiliate status for this purpose is not necessarily a conclusive determination for other purposes
As of March 3, 2017, there were 5,367,156 shares of the Registrant’s common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the Registrant’s Proxy Statement for the 2017 Annual Meeting of Stockholders of the Registrant to be held on June 23, 2017 are incorporated by reference into Part III of this Form 10-K.
The Registrant makes available free of charge on or through its website (http://www.tearlab.com) its Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and any amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934. The material is made available through the Registrant’s website as soon as reasonably practicable after the material is electronically filed with or furnished to the U.S. Securities and Exchange Commission, or SEC. All of the Registrant’s filings may be read or copied at the SEC’s Public Reference Room at 100 F Street, N.E., Room 1580, Washington D.C. 20549. Information on the hours of operation of the SEC’s Public Reference Room can be obtained by calling the SEC at 1-800-SEC-0330. The SEC maintains a website (http://www.sec.gov) that contains reports and proxy and information statements of issuers that file electronically.
TEARLAB CORPORATION
Form 10-K – ANNUAL REPORT
For the Fiscal Year Ended December 31, 2016
Table of Contents
PART
I
SPECIAL NOTE REGARDING FORWARD LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements relating to future events and our future performance within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. In some cases, you can identify forward-looking statements by terms such as “may”, “will”, “should”, “could”, “would”, “hope”, “expects”, “plans”, “intends”, “anticipates”, “believes”, “estimates”, “projects”, “predicts”, “potential” and similar expressions intended to identify forward-looking statements. These forward-looking statements include, without limitation, statements relating to future events, future results, and future economic conditions in general and statements about:
| ● | The adequacy of our funding and our forecast of the period of time through which our financial resources will be adequate to support our operations | |
| ● | Our future strategy, structure, and business prospects; | |
| ● | Our ability to continue as a going concern; | |
| ● | Our ability to obtain additional financing for working capital on acceptable terms and in a timely manner; | |
| ● | The continued commercialization of our current product; | |
| ● | Our ability to expand into next generation products; | |
| ● | Our ability to meet the financial covenants under our credit facilities; | |
| ● | Use of cash, cash needs and ability to raise capital; | |
| ● | The size and growth of the potential markets for our product and technology; | |
| ● | The effect of our strategy to streamline our organization and lower our costs; | |
| ● | The adequacy of current, and the development of new distributor, reseller, and supplier relationships, and our efforts to expand relationships with distributors and resellers in additional countries; | |
| ● | Our anticipated expansion of United States and international sales and operations; | |
| ● | Our ability to obtain and protect our intellectual property and proprietary rights; | |
| ● | The results of our clinical trials; | |
| ● | Our ability to maintain reimbursement for our product and support our pricing strategies; | |
| ● | Our plan to continue to develop and execute our conference and podium strategy to ensure visibility and evidence-based positioning of the TearLab® Osmolarity System among eye care professionals; | |
| ● | Our ability to attract and retain a sufficient number of scientists, clinicians, sales personnel and other key personnel with extensive experience in medical technology, who are in short supply; | |
| ● | Our beliefs about our employee relations; | |
| ● | Our efforts to assist our customers in obtaining their CLIA waiver or providing them with support from certified professionals; and | |
| ● | Our ability to remain listed on the NASDAQ Capital Market. |
These statements involve known and unknown risks, uncertainties and other factors, including the risks described in Part I, Item 1A of this Annual Report on Form 10-K, which may cause our actual results, performance or achievements to be materially different from any future results, performances, time frames or achievements expressed or implied by the forward-looking statements. Given these risks, uncertainties and other factors, you should not place undue reliance on these forward-looking statements. Information regarding market and industry statistics contained in this Annual Report on Form 10-K is included based on information available to us that we believe is accurate. It is generally based on academic and other publications that are not produced for purposes of securities offerings or economic analysis.
| -2- |
Corporate Information
TearLab Corp. was incorporated as OccuLogix, Inc. in Delaware in 2002. Unless the context requires otherwise, in this report the terms “the Company,” “we,” “us” and “our” refer to TearLab Corp. and our subsidiaries. References to “$” or “dollars” shall mean U.S. dollars unless otherwise indicated. References to “C$” shall mean Canadian dollars
Overview
We are an in-vitro diagnostic company based in San Diego, California. We have commercialized a proprietary tear testing platform, the TearLab® Osmolarity System that enables eye care practitioners to test for highly sensitive and specific biomarkers using nanoliters of tear film at the point-of-care. Our first product measures tear film osmolarity for the diagnosis of Dry Eye Disease or DED. Our results are included in our financial statements, which are included under Item 8 to this Annual Report on Form 10-K.
TearLab Research, Inc.
TearLab Research, Inc. (“TearLab Research”), our wholly-owned subsidiary, develops technologies to enable eye care practitioners to test a wide range of biomarkers (chemistries, metabolites, genes and proteins) at the point-of-care. Commercializing that tear testing platform is now the focus of our business.
Our product, the TearLab® Osmolarity System, enables the rapid measurement of tear osmolarity in the doctor’s office. Osmolarity is a quantitative and highly specific biomarker that has been shown to assist in the diagnosis and management of DED. Market Scope estimates that there are 19 million people suffering from dry eye in the US and nearly 337 million worldwide. Postmenopausal women make up the largest portion of the dry eye population across all regions of the world (US, West Europe, Japan, China, India, Latin America, and Rest of World). The innovation of the TearLab® Osmolarity System is its ability to precisely and rapidly measure osmolarity in nanoliter volumes of tear samples, using a highly efficient and novel tear collection system at the point of care. Historically, eye care researchers have relied on expensive instruments to perform tear biomarker analysis. In addition to their cost, these conventional systems are slow, highly variable in their measurement readings, and not categorized as waived by the United States Food and Drug Administration (the “FDA”), under regulations promulgated under the Clinical Laboratory Improvement Amendments, (“CLIA”).
The TearLab® Osmolarity System consists of the following three components: (1) the TearLab disposable, which is a single-use microfluidic microchip; (2) the TearLab Pen, which is a hand-held device that interfaces with the TearLab disposable; and (3) the TearLab Reader, which is a small desktop unit that allows for the docking of the TearLab Pen and provides a quantitative reading for the operator.
In October 2008, the TearLab® Osmolarity System received CE mark approval, clearing the way for sales in the European Union and all countries recognizing the CE mark. On December 8, 2009, TearLab announced that Health Canada issued a Medical Device License for the TearLab® Osmolarity System.
| -3- |
On May 19, 2009, we announced that we received 510(k) clearance from the FDA. On January 23, 2012, we announced the FDA had granted the waiver categorization under CLIA for the TearLab® Osmolarity System. The CLIA waiver reduces the regulatory paperwork and related administrative time for customers.
Currently, the TearLab®Osmolarity System is commercialized in over 40 countries. In the United States, the TearLab Osmolarity System is sold direct. In markets outside of the US, the TearLab® Osmolarity System is sold through distributors.
OcuHub LLC
On March 14, 2014, TearLab purchased the assets of the OcuHub business unit from AOAExcel, Inc., the for-profit subsidiary of the American Optometric Association (“AOA”). As of the close of the transaction, the OcuHub purchased assets and business were operated out of OcuHub Holdings, Inc., a wholly-owned subsidiary of TearLab and renamed OcuHub LLC (“OcuHub”). The purchase price was $1.4 million in cash and the assumption of net liabilities of $163,000.
On April 8, 2016, OcuHub Holdings, Inc., completed the sale of 10,167.5 units of OcuHub to an OcuHub executive and an unrelated third party. OcuHub Holdings, Inc. currently owns approximately 10.5% of OcuHub on a fully diluted basis (including all outstanding options and profits interests).
Industry
Point-of-care Testing and DED
The market research firm, “Markets and Markets” reports the global market for point-of-care testing will reach $37.0 billion annually by 2021. Approximately 75% of all laboratory tests today are performed at centralized clinical laboratories. However, diagnostic testing is increasingly being performed at the point-of-care due to several factors, including a need for rapid testing in acute care situations, the benefits of patient monitoring and disease management, streamlining therapeutic decision making and the overall trend toward personalized medicine. We believe that advances in bio-detection technologies that can simplify and accelerate the rate of performing complex diagnostic tests at the point-of-care will drive utilization and overall point-of-care testing market growth.
Each time a person blinks, his or her eyes are resurfaced with a thin layer of a complex fluid known as the tear film. The tear film works to protect eyes from the outside world. Bacteria, viruses, sand, freezing winds and salt water (inclusive of most environmental factors) will not damage eyes when the tear film is intact. However, when compromised, a deficient tear film can be an exceedingly painful and disruptive condition. The tear film consists of three components: (i) an innermost glycocalyx (produced by the surface cells); (ii) the aqueous layer (the water in tears, produced by the lacrimal gland); and (iii) an oily lipid layer which limits evaporation of the tears (produced by the meibomian glands, located at the margins of the eyelids). The apparatus of the ocular surface forms an integrated unit. When working correctly, the tear film presents a smooth optical surface essential for clear vision and proper immunity. Androgen deficiency, contact lens wear and chronic inflammation of the lacrimal or meibomian gland may lead to the condition known as dry eye, which has been likened to arthritis of the eye, and results in a compromised, fragile tear film. In turn, the unstable tear film undermines vision, altering focus between every blink. An unstable tear film is the equivalent of a smudge atop the lens of a camera. It doesn’t matter how many megapixels your camera has, if the first lens is compromised, the image will be fuzzy.
| -4- |
DED is often a result of aging, diabetes, cancer therapy, HIV, autoimmune diseases such as Sjögren’s syndrome and rheumatoid arthritis, LASIK surgery, contact lens wear, menopause and as a side effect of hormone replacement therapy. Numerous commonly prescribed and over-the-counter medications also can cause, or contribute to, the manifestation of DED.
Discomfort and dryness are the most commonly reported symptoms of contact lens wear. These symptoms can lead to contact lens drop out if severe and/or persistent. In 2010, Contact Lens Spectrum reported that 16% of contact lens wearers permanently dropout of contact lens wear each year. In addition, there are approximately 600,000 LASIK procedures performed in the U.S each year with up to 60% reporting dry eye symptoms 1 month post-LASIK.
Diagnostic Alternatives for Dry Eye Disease
Existing diagnostic tools are highly subjective, do not correlate well with symptoms, are invasive for patients and may require up to an hour of operator time to perform. All of these factors have constrained the diagnosis and treatment of the DED patient population. As physicians have not had access to objective, quantitative diagnostic assays that correlate well with and the severity of DED disease, it has been difficult for them to objectively differentiate DED symptoms from other eye diseases that present with very similar symptoms, such as ocular allergies, conjunctivochalasis or infectious bacterial or viral diseases. To treat DED effectively and to mitigate the emotional and physical effects of this disease, it is important to equip physicians with objective, quantitative measurements of disease pathogenesis so they can determine more accurately the most efficacious treatments for their patients.
Osmolarity in DED presents itself as an increase in the salt concentration of the tear film. For over 50 years, studies have shown that tear film osmolarity is an ideal biomarker for diagnosing DED, providing an objective, quantitative measurement of disease pathogenesis. Measuring osmolarity also serves as an effective disease management tool by providing physicians with an ability to personalize therapeutic intervention and to track patient outcomes quantitatively. Osmolarity testing could also provide physicians with a tool to identify patients at risk for dropping out of contact lens wear early in disease progression, as well as an invaluable test to guide the type and duration of therapy prior to, and following refractive surgery.
The main challenge in measuring osmolarity at the point-of-care is the small volume of tear available for testing. Older laboratory osmometers require upwards of ten microliters of fluid to produce a single reading. In addition, these instruments are not particularly suitable for use in a physician’s office, since they require continual calibration, cleaning and maintenance. Existing osmometers currently are marketed primarily to reference and hospital laboratories for the measurement of osmolarity in blood, urine and other serum samples.
TearLab’s Product
Our TearLab® Osmolarity System is an integrated testing system comprised of: (1) the TearLab disposable, which is a single-use microfluidic microchip; (2) the TearLab Pen, which is a hand-held device that interfaces with the TearLab disposable; and (3) the TearLab Reader, which is a small desktop unit that allows for the docking of the TearLab Pen and provides a quantitative reading for the operator. The innovation of the TearLab® Osmolarity System is its ability to measure precisely, rapidly, and inexpensively biomarkers in nanoliter volumes of tear samples or approximately 1,000 times less volume than required for older laboratory devices.
The operator of the TearLab® Osmolarity System, most commonly a technician, collects the tear sample from the patient’s eye in the TearLab disposable, using the TearLab Pen. After the tear has been collected, the operator places the Pen into the Reader. The TearLab Reader then will display an osmolarity reading to the operator. Following the completion of the test, the TearLab disposable will be discarded and a new TearLab disposable will be readied for the next test. The entire process, from sample to answer, should require approximately two minutes or less to complete.
| -5- |
Competition
The medical device industry is highly competitive and we face potential competition from medical device companies worldwide. There are several laboratory technologies that claim to measure the osmolarity of nanoliter tear samples. The i-Pen manufactured by i-Med Pharma Inc. has approval from Health Canada and a CE mark. The LacriPen, developed by LacriSciences, LLC (Washington, DC, US), does not have a CE Mark, FDA 510(k) clearance or a CLIA waiver, but has stated to be in clinical trials. Another investigational device aimed at dry eye diagnosis, the TeaRx, manufactured by DiagnosTear Ltd., a division of BioLight Life Sciences Investments of Tel Aviv, Israel, announced positive correlations between TeaRx diagnostic parameters and benchmarks used to test for dry eye syndrome. Another non-osmolarity based in vitro diagnostic test for dry eye has been developed by Rapid Pathogen Screening, Inc. (RPS), of Sarasota Florida. RPS has commercialized a tear test for dry eye that measures MMP-9, an inflammatory biomarker. This test is FDA cleared and has obtained a CLIA waiver. Another company, ATD (Advanced Tear Diagnostics) has a CLIA classification of Moderately Complex in the United States, and markets products that measure lactoferrin and IgE in human tears for the diagnosis of aqueous deficient dry eye disease and ocular allergy, respectively.
Tear film break-up time, or TBUT, is a non-laboratory test performed to evaluate tear film stability during an examination of the ocular surface with a slit lamp by an eye care practitioner. However, it is subjective, requires a physician to instill a carefully controlled amount of fluorescein dye into the eye and requires a stopwatch to determine the endpoint. TBUT has been shown to be unreliable as a determinant of DED since shortened TBUT does not always correlate well with other signs or symptoms.
Other office-based tests performed during a standard eye care examination like impression cytology and corneal staining, although indicative of relatively late stage phenomena in DED, are subjective, qualitative and generally do not correlate to disease pathogenesis. We believe the Schirmer Test, to determine tear fluid volume, is an imprecise marker of tear function since its diagnostic results vary significantly.
Principal Suppliers
We rely on two suppliers based in the United States for the manufacture of the Readers and Pens which are key components of the TearLab® Osmolarity System. We also rely on a single supplier, MiniFAB (Aust) Pty Ltd. located in Australia, for the manufacture of the test cards which is also a key component of the TearLab® Osmolarity System.
Patents and Proprietary Rights
We own or have exclusive licenses to multiple patents and applications relating to the TearLab® Osmolarity System and related technology and processes:
| ● | eleven issued U.S. patents; relating to the TearLab® Osmolarity System and related technology and processes and have applied for a number of other patents in the United States and other jurisdictions; | |
| ● | twenty five issued patents in the rest of the world; and | |
| ● | eighteen applications pending. |
| -6- |
We intend to rely on know-how, continuing technological innovation and in-licensing opportunities to further develop our proprietary position. Our ability to obtain intellectual property protection for the TearLab® Osmolarity System and related technology and processes, and our ability to operate without infringing on the intellectual property rights of others and to prevent others from infringing on our intellectual property rights, will have a substantial impact on our ability to succeed in our business. Although we intend to seek to protect our proprietary position by, among other methods, continuing to file patent applications, the patent position of companies like TearLab is generally uncertain and involves complex legal and factual questions. Our ability to maintain and solidify a proprietary position for our technology will depend on our success in obtaining effective claims and enforcing those claims once granted. We do not know whether any part of our patent applications will result in the issuance of any patents. Our issued patents, those that may be issued in the future or those licensed to us, may be challenged, invalidated or circumvented, which could limit our ability to stop would-be competitors from marketing tests identical to the TearLab® Osmolarity System.
In addition to patent protection, we have registered the TearLab trademark in the United States, the European Union, Japan, Korea, Mexico, the Russian Federation, Australia, Canada, China and Turkey.
Government Regulation
Government authorities in the United States and other countries extensively regulate, among other things, the research, development, testing, manufacture, labeling, promotion, advertising, distribution and marketing of our product, which is a medical device. In the United States, the FDA regulates medical devices under the Federal Food, Drug, and Cosmetic Act and implementing regulations. Failure to comply with the applicable FDA requirements, both before and after approval, may subject us to administrative and judicial sanctions, such as a delay in approving or refusal by the FDA to approve pending applications, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, administrative fines or criminal prosecution.
Unless exempted by regulation, medical devices may not be commercially distributed in the United States until they have been cleared or approved by the FDA. Medical devices are classified into one of the three classes, Class I, II or III, on the basis of the controls necessary to reasonably assure their safety and effectiveness. Class I devices are subject to general controls, such as labeling, pre-market notification and adherence to good manufacturing practices. The TearLab® Osmolarity System is a Class I, non-exempt device and qualifies for the 510(k) procedure. Under the FDA’s Section 510(k) procedure, the manufacturer provides a pre-market notification that it intends to begin marketing the product, and shows that the product is substantially equivalent to another legally marketed product, that it has the same intended use and is as safe and effective as a legally marketed device and does not raise different questions of safety and effectiveness than does a legally marketed device. In some cases, the submission must include data from human clinical studies. Marketing may commence when the FDA issues a clearance letter finding substantial equivalence. On May 19, 2009, we announced that we received FDA 510(k) clearance of the TearLab® Osmolarity System.
After a device receives 510(k) clearance, any modification to the device that could significantly affect its safety or effectiveness, or that would constitute a major change in its intended use, would require a new 510(k) clearance or an approval of a Premarket Approval, or PMA. A PMA is the FDA process of scientific or regulatory review to evaluate the safety and effectiveness of Class III medical devices which are those devices which support or sustain human life, are of substantial importance in preventing impairment of human health, or which present a potential, unreasonable risk of illness or injury. Although the FDA requires the manufacturer to make the initial determination regarding the effect of a modification to the device that is subject to 510(k) clearance, the FDA can review the manufacturer’s determination at any time and require the manufacturer to seek another 510(k) clearance or an approval of a PMA.
| -7- |
CLIA is intended to ensure the quality and reliability of clinical laboratories in the United States by mandating specific standards in the areas of personnel qualifications, administration, and participation in proficiency testing, patient test management, quality control, quality assurance and inspections. The regulations promulgated under CLIA establish three levels of in vitro diagnostic tests: (1) waiver; (2) moderately complex; and (3) highly complex. The standards applicable to a clinical laboratory depend on the level of diagnostic tests it performs. A CLIA waiver is available to clinical laboratory test systems if they meet certain requirements established by the statute. Waived tests are simple laboratory examinations and procedures employing methodologies that are so simple and accurate as to render the likelihood of erroneous results negligible or to pose no reasonable risk of harm to patients if the examinations or procedures are performed incorrectly. These tests are waived from regulatory oversight of the user other than the requirement to follow the manufacturer’s labeling and directions for use.
On January 23, 2012, we announced that after reviewing and accepting labeling submitted to it by the Company, the FDA had granted the waiver categorization under CLIA for the TearLab® Osmolarity System.
Regardless of whether a medical device requires FDA clearance or approval, a number of other FDA requirements apply to the device, its manufacturer and those who distribute it. Device manufacturers must be registered and their products listed with the FDA, and certain adverse events and product malfunctions must be reported to the FDA. The FDA also regulates the product labeling, promotion and, in some cases, advertising of medical devices. In addition, manufacturers and their suppliers must comply with the FDA’s quality system regulation which establishes extensive requirements for quality and manufacturing procedures. Thus, suppliers, manufacturers and distributors must continue to spend time, money and effort to maintain compliance, and failure to comply can lead to enforcement action. The FDA periodically inspects facilities to ascertain compliance with these and other requirements.
Clinical, Regulatory, Research and Development Expenditure
Our clinical, regulatory, research and development expense was $5.2 million and $7.0 million for the years ended December 31, 2016 and 2015, respectively.
Employees
On December 31, 2016, we had 75 full-time employees. None of our employees are covered by a collective bargaining agreement.
Available Information
Our corporate Internet address is www.tearlab.com. At the Investor Relations section of this website, we make available free of charge our Annual Report on Form 10-K, our Annual Proxy statement, our quarterly reports on Form 10-Q, any Current Reports on Form 8-K, and any amendments to these reports, as soon as reasonably practicable after we electronically file them with, or furnish them to, the U.S. Securities and Exchange Commission, or the SEC. The information found on our website is not part of this Annual Report on Form 10-K. In addition to our website, the SEC maintains an Internet site at www.sec.gov that contains reports, proxy and information statements, and other information regarding us and other issuers that file electronically with the SEC.
| -8- |
Risks Relating to Our Financial Condition
We have limited working capital and a history of losses that raise substantial doubts as to whether we will be able to continue as a going concern.
We have prepared our consolidated financial statements on the basis that we would continue as a going concern. However, we have incurred losses in each year since our inception. We do not currently have any available borrowing under our term loan or credit facility.
Our consolidated financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary if we were not able to continue as a going concern. If we are unable to generate positive cash flows from operations, we would need to undertake a review of potential business alternatives, which may include, but are not limited to, a merger or sale of the company or ceasing operations and winding down the business.
We have incurred losses since inception and anticipate that we will incur continued losses for the foreseeable future.
We have incurred losses in each year since our inception. As of December 31, 2016, we had an accumulated deficit of $512.7 million. Our losses have resulted primarily from expenses incurred in research and development of our product candidates from the former retina and glaucoma business divisions and to the development and commercialization of our tear testing platform. We do not know when or if we will successfully commercialize the TearLab® Osmolarity System in the United States or in international markets on a scale that will allow us to achieve and sustain profitability. As a result, and because of the numerous risks and uncertainties facing us, it is difficult to provide the extent of any future losses or the time required to achieve profitability, if at all. Any failure to become and remain profitable would require us to undertake a review of the potential business alternatives discussed above.
We may need to raise additional capital at some point in the future if we do not achieve our business objectives. Such capital, if needed, may not be available to us on reasonable terms, if at all, when or as we require additional funding. If we issue additional shares of our common stock or other securities that may be convertible into, or exercisable or exchangeable for, our common stock, our existing stockholders would experience further dilution.
We may be required to raise additional capital from time to time in the future if we do not execute on our current business objectives and continue to grow our revenue at a rate sufficient to fund our operations with our current cash on hand. Such financings may involve the issuance of debt, equity and/or securities convertible into or exercisable or exchangeable for our equity securities. These financings may not be available to us on reasonable terms or at all when and as we require funding. Any failure to obtain additional working capital when required would have a material adverse effect on our business and financial condition and our ability to continue as a going concern and would be expected to result in a decline in our stock price. If we consummate such financings, the terms of such financings may adversely affect the interests of our existing stockholders. Any issuances of our common stock, preferred stock, or securities such as warrants or notes that are convertible into, exercisable or exchangeable for, our capital stock, would have a dilutive effect on the voting and economic interest of our existing stockholders.
| -9- |
We may not be able to generate sufficient cash to service our indebtedness, which currently consists of our credit facility with CRG. We may not be able to satisfy our minimum revenue and cash covenants, as required by the CRG term loan. If our annual sales revenue levels do not meet or exceed the levels required by the CRG covenants, we will be required to raise additional equity or subordinated debt, with the proceeds paid to reduce the outstanding principal of the CRG term loan. This financing could dilute existing shareholders and impact the value of their investment.
On March 4, 2015, we executed a term loan agreement with CRG as lenders which we refer to as the Term Loan Agreement, providing us with access of up to $35.0 million under the Term Loan Agreement. We entered into an amendment of the Term Loan Agreement with CRG on August 6, 2015. We received $25.0 million in gross proceeds during 2015. We were unable to access a third tranche of $10.0 million because we did not achieve at least $38.0 million in twelve-month sales revenue prior to June 30, 2016, as required to access the third tranche.
Our ability to make scheduled payments or to refinance our debt obligations depends on numerous factors, including the amount of our cash reserves and our actual and projected financial and operating performance. These amounts and our performance are subject to certain financial and business factors, as well as prevailing economic and competitive conditions, some of which may be beyond our control. We cannot assure you that we will maintain a level of cash reserves or cash flows from operating activities sufficient to permit us to pay the principal, premium, if any, and interest on our existing or future indebtedness. If our cash flows and capital resources are insufficient to fund our debt service obligations, we may be forced to reduce or delay capital expenditures, sell assets or operations, seek additional capital or restructure or refinance our indebtedness. We cannot assure you that we would be able to take any of these actions, or that these actions would permit us to meet our scheduled debt service obligations. In addition, in the event of our breach of the Term Loan Agreement, we may be required to repay any outstanding amounts earlier than anticipated, and the lenders may foreclose on their security interest in our assets.
The CRG loan is collateralized by all our assets. Additionally, the terms of the Term Loan Agreement contain various affirmative and negative covenants that we agreed to. Among them are requirements that we must attain minimum annual revenue and minimum cash threshold levels. The minimum annual revenue threshold levels required by the Term Loan are $31.0 million, $36.0 million, $45.0 million, and $55.0 million for calendar years 2017, 2018, 2019 and 2020, respectively. The minimum cash balance required is $5.0 million, subject to certain conditions.
If we do not have annual revenue greater or equal to the annual revenue covenant in a calendar year, we will have to raise subordinated debt or equity, which we refer to as the CRG Equity Cure, equal to twice the difference between the annual revenue and the revenue covenant, with the total proceeds from this financing to be used to reduce the principal of the Term Loan Agreement. We cannot assure you that we will be able to achieve the annual revenue thresholds and the minimum cash threshold. We cannot assure you that we would be able to raise the financing for the CRG Equity Cure, if required. In addition, in the event of our breach of the Term Loan Agreement, we may be required to repay any outstanding amounts earlier than anticipated, and the lenders may foreclose on their security interest in our assets.
Borrowings under the Term Loan Agreement are subject to certain conditions, including the non-occurrence of a material adverse change in our business or operations (financial or otherwise), or a material impairment of the prospect of repayment of obligations.
| -10- |
Our existing Term Loan Agreement contains restrictive and financial covenants that may limit our operating flexibility.
The Term Loan Agreement contains certain restrictive covenants that limit our ability to incur additional indebtedness and liens, merge with other companies or consummate certain changes of control, acquire other companies, engage in new lines of business, make certain investments, pay dividends, transfer or dispose of assets, amend certain material agreements or enter into various specified transactions. We therefore may not be able to engage in any of the foregoing transactions unless we obtain the consent of CRG or terminate the Term Loan Agreement. There is no guarantee that we will be able to generate sufficient cash flow or sales to meet the financial covenants or pay the principal and interest under the agreement. Furthermore, there is no guarantee that future working capital, borrowings or equity financing will be available to repay or refinance the amounts outstanding under the Term Loan Agreement.
Our financial results may vary significantly from year-to-year due to a number of factors, which may lead to volatility in the trading price of our common stock.
Our annual and quarterly revenue and results of operations have varied in the past and may continue to vary significantly from year-to-year and quarter-to-quarter. The variability in our annual and quarterly results of operations may lead to volatility in our stock price as research analysts and investors respond to these annual fluctuations. These fluctuations are due to numerous factors that are difficult to forecast, including:
| ● | fluctuations in demand for our products; | |
| ● | changes in customer budget cycles and capital spending; | |
| ● | seasonal variations in customer operations that could occur during holiday or summer vacation periods; | |
| ● | tendencies among some customers to defer purchase decisions to the end of the quarter; | |
| ● | the unit value of our systems; | |
| ● | changes in our pricing and sales policies or the pricing and sales policies of our competitors; | |
| ● | changes in reimbursement levels that might negatively impact our pricing policies; | |
| ● | our ability to design, manufacture and deliver products to our customers in a timely and cost effective manner; | |
| ● | quality control or yield problems in our manufacturing suppliers; | |
| ● | our ability to timely obtain adequate quantities of the components used in our products; | |
| ● | new product introductions and enhancements by us and our competitors; | |
| ● | unanticipated increases in costs or expenses; | |
| ● | our complex, variable and, at times, lengthy sales cycle; | |
| ● | global economic conditions; and | |
| ● | fluctuations in foreign currency exchange rates. |
In addition, we may experience seasonal variations in our customer operations such as could occur during holiday vacation periods. For example, one of our principal target markets consists of private ophthalmic and optometric practices, and our quarterly operating results could be adversely affected by summer or holiday vacation periods. The foregoing factors, as well as other factors, could materially and adversely affect our quarterly and annual results of operations. In addition, a significant amount of our operating expenses are relatively fixed due to our manufacturing, research and development, and sales and general administrative efforts. Any failure to adjust spending quickly enough to compensate for a revenue shortfall could magnify the adverse impact of such revenue shortfall on our results of operations. We expect that our sales will continue to fluctuate on a quarterly basis and our financial results for some periods may differ from those projected by securities analysts, which could significantly decrease the price of our common stock.
| -11- |
Risks Related to our Business
Our near-term success is highly dependent on the success of the TearLab® Osmolarity System, and we cannot be certain that we will successfully increase such sales.
The TearLab® Osmolarity System is currently our only product. Our product is currently sold outside of the United States pursuant to CE mark approval; in Canada pursuant to a Health Canada Medical Device License; and in the United States as a result of having received 510(k) approval from the FDA to market the TearLab® Osmolarity System to those reference and physician operated laboratories with CLIA waiver certifications. Even though the TearLab® Osmolarity System has received all regulatory approvals in the United States, it may never generate sufficient sales to achieve profitability. If the TearLab® Osmolarity System is not as successfully commercialized as expected, we may not be able to generate sufficient revenue to become profitable or continue our operations. Any failure of the TearLab® Osmolarity System to be successfully commercialized in the United States would have a material adverse effect on our business, operating results, financial condition and cash flows and could result in a substantial decline in the price of our common stock.
Our near-term success is highly dependent on increasing sales of the TearLab® Osmolarity System outside the United States, and we cannot be certain that we will successfully increase such sales.
Our product is currently sold outside of the United States pursuant to CE mark approval and Health Canada Approval in Canada. Our near-term success is highly dependent on increasing our international sales. We may also be required to register our product with health departments in our foreign market countries. A failure to successfully register in such markets would negatively affect our sales in any such markets. In addition, import taxes are levied on our product in certain foreign markets. Other countries may adopt taxation codes on imported products. Increases in such taxes or other restrictions on our product could negatively affect our ability to import, distribute and price our product.
We have outstanding liabilities, which could adversely affect our ability to adjust our business to respond to competitive pressures and to obtain sufficient funds to satisfy our future research and development needs, and to defend our intellectual property.
As of December 31, 2016, our total liabilities were $32.3 million including $26.4 million of long-term obligations under our Term Loan Agreement. Our significant liability service requirements could adversely affect our ability to operate our business and may limit our ability to take advantage of potential business opportunities. For example:
| ● | our liabilities increase our vulnerability to economic downturns and adverse competitive and industry conditions and could place us at a competitive disadvantage compared to those of our competitors that are less leveraged; | |
| ● | our liabilities could limit our flexibility in planning for, or reacting to, changes in our business and our industry and could limit our ability to pursue other business opportunities, borrow money for operations or capital in the future and implement our business strategies; and | |
| ● | our liabilities may restrict us from raising additional funds on satisfactory terms to fund working capital, capital expenditures, product development efforts, strategic acquisitions, investments and alliances, and other general corporate requirements. |
If we are at any time unable to generate sufficient cash flow to service our liabilities when payment is due, we may be required to attempt to renegotiate the terms of the instruments relating to the liabilities, seek to refinance all or a portion of the liabilities or obtain additional financing. There can be no assurance that we will be able to successfully renegotiate such terms, that any such refinancing would be possible or that any additional financing could be obtained on terms that are favorable or acceptable to us.
| -12- |
We will face challenges in bringing the TearLab® Osmolarity System to market in the United States and may not succeed in executing our business plan.
There are numerous risks and uncertainties inherent in the development of new medical technologies. In addition to our requirement for additional capital, our ability to bring the TearLab® Osmolarity System to market in the United States and to execute our business plan successfully is subject to the following risks, among others:
| ● | Our clinical trials may not succeed. Clinical testing is expensive and can take longer than originally anticipated. The outcomes of clinical trials are uncertain, and failure can occur at any stage of the testing. We could encounter unexpected problems, which could result in a delay in efforts to complete clinical trials supporting our commercialization efforts. | |
| ● | The TearLab® Osmolarity System is rated under a CLIA waiver certification which requires our customers to be certified under the CLIA waiver requirements to be reimbursed under Medicare, including certain parallel state requirements. If our customers are unwilling or unable to comply with such requirements, it could have an adverse effect on their acceptance of and on our ability to market the TearLab® Osmolarity System in the United States. | |
| ● | Our suppliers and we will be subject to numerous FDA requirements covering the design, testing, manufacturing, quality control, labeling, advertising, promotion and export of the TearLab® Osmolarity System and other matters. If our suppliers or we fail to comply with these regulatory requirements, the TearLab® Osmolarity System could be subject to restrictions or withdrawals from the market and we could become subject to penalties. | |
| ● | Even though we successfully obtained the sought-after FDA approvals, we may be unable to commercialize the TearLab® Osmolarity System successfully in the United States. Successful commercialization will depend on a number of factors, including, among other things, achieving widespread acceptance of the TearLab® Osmolarity System among physicians, establishing adequate sales and marketing capabilities, addressing competition effectively, obtaining and enforcing patents to protect proprietary rights from use by would-be competitors, key personnel retention and ensuring sufficient manufacturing capacity and inventory to support commercialization plans. |
Our business is subject to health care industry and government cost-containment measures that could result in reduced sales of our TearLab® Osmolarity System.
Most of our customers rely on third-party payers, including government programs and private health insurance plans, to reimburse some or all of the cost of the procedures which use our TearLab® Osmolarity System. The continuing efforts of governmental authorities, insurance companies, and other health care payers to contain or reduce these costs could lead to patients being unable to obtain approval for payment from these third-party payers. If patients cannot obtain third-party payer payment approval, the use of our TearLab® Osmolarity System may decline significantly and our customers may reduce or eliminate the use of our system. The cost-containment measures that health care providers are instituting, both in the U.S. and internationally, could harm our ability to operate profitably. For example, managed care organizations have successfully negotiated volume discounts for pharmaceuticals. While this type of discount pricing does not currently exist for the medical systems we supply, if managed care or other organizations were able to affect discount pricing for such systems, it could result in lower prices to our customers from their customers and, in turn, reduce the amounts we can charge our customers for our products.
| -13- |
In addition to general health care industry cost-containment, the Centers for Medicare and Medicaid Services (CMS) released its final rule implementing section 216(a) of the Protecting Access to Medicare Act of 2014 (PAMA) that will require reporting entities to report private payer rates paid to laboratories for lab tests, which will be used to calculate Medicare payment rates. This final rule also announces CMS’ decision to move the implementation date for the private payer rate-based fee schedule to January 1, 2018. Reporting entities, which would primarily be certain qualifying customers in the U.S. that derive a certain percentage and volume of their revenue from laboratory tests from Medicare, will report private payer rates for our laboratory tests which will serve under the act as a baseline for future reimbursement. Although it is very early to understand if reimbursement for our product or future products will be impacted, the act does provide a maximum reimbursement reduction of 10% per year for the years 2018 through 2020. For years 2021 through 2023, the maximum reduction on the reimbursement rate is 15%. Should reimbursement for our products be reduced as a result of PAMA, this could negatively impact our pricing and commercialization of our products in the U.S.
If we are subject to regulatory enforcement action as a result of our failure to comply with regulatory requirements, our commercial operations would be harmed.
While we received the 510(k) clearance and CLIA waiver that we were seeking, we are subject to significant ongoing regulatory requirements, and if we fail to comply with these requirements, we could be subject to enforcement action by the FDA or state agencies, including:
| ● | adverse publicity, warning letters, fines, injunctions, consent decrees and civil penalties; | |
| ● | repair, replacement, refunds, recall or seizure of our product; | |
| ● | operating restrictions or partial suspension or total shutdown of production; | |
| ● | delay or refusal of our requests for 510(k) clearance or premarket approval of new products or of new intended uses or modifications to our existing product; | |
| ● | refusal to grant export approval for our products; | |
| ● | withdrawing 510(k) clearances or premarket approvals that have already been granted; and | |
| ● | criminal prosecution. |
If the government initiated any of these enforcement actions, our business could be harmed.
We are required to demonstrate and maintain compliance with the FDA’s Quality System Regulation, or the QSR. The QSR is a complex regulatory scheme that covers the methods and documentation of the design, testing, control, manufacturing, labeling, quality assurance, packaging, storage and shipping of our products. The FDA must determine that the facilities which manufacture and assemble our products that are intended for sale in the United States, as well as the manufacturing controls and specifications for these products, are compliant with applicable regulatory requirements, including the QSR. The FDA enforces the QSR through periodic unannounced inspections. The FDA has not yet inspected our facilities, and we cannot assure you that we will pass any future FDA inspection. Our failure, or the failure of our suppliers, to take satisfactory corrective action in response to an adverse QSR inspection could result in enforcement actions, including a public warning letter, a shutdown of our manufacturing operations, a recall of our product, civil or criminal penalties or other sanctions, which would significantly harm our available inventory and sales and cause our business to suffer.
| -14- |
If we are unable to fully comply with federal and state “fraud and abuse laws,” we could face substantial penalties, which may adversely affect our business, financial condition and results of operations.
We are subject to various laws pertaining to health care fraud and abuse, including the U.S. Anti- Kickback Statute, physician self-referral laws (the “Stark Law”), the U.S. False Claims Act, the U.S. False Statements Statute, the Physician Payment Sunshine Act, and state law equivalents to these U.S. federal laws, which may not be limited to government-reimbursed items and may not contain identical exceptions. Violations of these laws are punishable by criminal and civil sanctions, including, in some instances, civil and criminal penalties, damages, fines, exclusion from participation in U.S. federal and state health care programs, including Medicare and Medicaid, and the curtailment or restructuring of operations. Any action against us for violation of these laws could have a significant impact on our business. In addition, we are subject to the U.S. Foreign Corrupt Practices Act. Any action against us for violation by us or our agents or distributors of this act could have a significant impact on our business.
If we fail to comply with contractual obligations and applicable laws and regulations governing the handling of patient identifiable medical information, we could suffer material losses or be adversely affected by exposure to material penalties and liabilities.
Many, if not all of our customers, are covered entities under the Health Insurance Portability and Accountability Act of August 1996 or HIPAA. As part of the operation of our business, we provide reimbursement assistance to certain of our customers and as a result we act in the capacity of a business associate with respect to any patient-identifiable medical information, or PHI, we receive in connection with these services. We and our customers must comply with a variety of requirements related to the handling of patient information, including laws and regulations protecting the privacy, confidentiality and security of PHI. The provisions of HIPAA require our customers to have business associate agreements with us under which we are required to appropriately safeguard the PHI we create or receive on their behalf. Further, we and our customers are required to comply with HIPAA security regulations that require us and them to implement certain administrative, physical and technical safeguards to ensure the confidentiality, integrity and availability of electronic PHI, or EPHI. We are required by regulation and contract to protect the security of EPHI that we create, receive, maintain or transmit for our customers consistent with these regulations. To comply with our regulatory and contractual obligations, we may have to reorganize processes and invest in new technologies. We also are required to train personnel regarding HIPAA requirements. If we, or any of our employees or consultants, are unable to maintain the privacy, confidentiality and security of the PHI that is entrusted to us, we and/or our customers could be subject to civil and criminal fines and sanctions and we could be found to have breached our contracts with our customers. Under the Health Information Technology for Economic and Clinical Health Act, or HITECH Act, and recent omnibus revisions to the HIPAA regulations, we are directly subject to HIPAA’s criminal and civil penalties for breaches of our privacy and security obligations and are required to comply with security breach notification requirements. The direct applicability of the HIPAA privacy and security provisions and compliance with the notification requirements requires us to incur additional costs and may restrict our business operations.
Our patents may not be valid, and we may not obtain and enforce patents to protect our proprietary rights from use by would-be competitors. Companies with other patents could require us to stop using or pay to use required technology.
Our owned and licensed patents may not be valid, and we may not obtain and enforce patents to maintain trade secret protection for our technology. The extent to which we are unable to do so could materially harm our business.
| -15- |
We have applied for, and intend to continue to apply for, patents relating to the TearLab® Osmolarity System and related technology and processes. Such applications may not result in the issuance of any patents, and any patents now held or that may be issued may not provide adequate protection from competition. Furthermore, it is possible that patents issued or licensed to us may be challenged successfully. In that event, if we have a preferred competitive position because of any such patents, any preferred position would be lost. If we are unable to secure or to continue to maintain a preferred position, the TearLab® Osmolarity System could become subject to competition from the sale of similar competing products.
Patents issued or licensed to us may be infringed by the products or processes of others. The cost of enforcing patent rights against infringers, if such enforcement is required, could be significant and the time demands could interfere with our normal operations. There has been substantial litigation and other proceedings regarding patent and other intellectual property rights in the pharmaceutical, biotechnology and medical technology industries. We are currently involved in litigation defending our patent rights in Canada. Efforts to defend our rights could incur significant costs and may or may not be resolved in our favor. We could become a party to additional patent litigation and other proceedings. The cost to us of any patent litigation, even if resolved in our favor, could be substantial. Some of our would-be competitors may sustain the costs of such litigation more effectively than we can because of their greater financial resources. Litigation also may absorb significant management time.
Unpatented trade secrets, improvements, confidential know-how and continuing technological innovation are important to our future scientific and commercial success. Although we attempt, and will continue to attempt, to protect our proprietary information through reliance on trade secret laws and the use of confidentiality agreements with corporate partners, collaborators, employees and consultants and other appropriate means, these measures may not effectively prevent disclosure of our proprietary information, and, in any event, others may develop independently, or obtain access to, the same or similar information.
Certain of our patent rights are licensed to us by third parties. If we fail to comply with the terms of these license agreements, our rights to those patents may be terminated, and we will be unable to conduct our business.
It is possible that a court may find us to be infringing upon validly issued patents of third parties. In that event, in addition to the cost of defending the underlying suit for infringement, we may have to pay license fees and/or damages and may be enjoined from conducting certain activities. Obtaining licenses under third-party patents can be costly, and such licenses may not be available at all.
We may face future product liability claims.
The testing, manufacturing, marketing and sale of therapeutic and diagnostic products entail significant inherent risks of allegations of product liability. Our past use of the RHEO™ System and the components of the SOLX Glaucoma System in clinical trials and the commercial sale of those products may have exposed us to potential liability claims. Our use of the TearLab® Osmolarity System and its commercial sale could also expose us to liability claims. All of such claims might be made directly by patients, health care providers or others selling the products. We carry clinical trials and product liability insurance to cover certain claims that could arise, or that could have arisen, during our clinical trials or during the commercial use of our products. We currently maintain clinical trials and product liability insurance with aggregate annual coverage limits of $2.0 million. Such coverage, and any coverage obtained in the future, may be inadequate to protect us in the event of successful product liability claims, and we may not increase the amount of such insurance coverage or even renew it. A successful product liability claim could materially harm our business. In addition, substantial, complex or extended litigation could result in the incurrence of large expenditures and the diversion of significant resources.
| -16- |
If we do not introduce new commercially successful products in a timely manner, our products may become obsolete over time, customers may not buy our products and our revenue and profitability may decline.
Demand for our products may change in ways we may not anticipate because of:
| ● | evolving customer needs; | |
| ● | the introduction of new products and technologies; and | |
| ● | evolving industry standards. |
Without the timely introduction of new commercially successful products and enhancements, our products may become obsolete over time, in which case our sales and operating results would suffer. The success of our new product offerings will depend on several factors, including our ability to:
| ● | properly identify and anticipate customer needs; | |
| ● | commercialize new products in a cost-effective and timely manner; | |
| ● | manufacture and deliver products in sufficient volumes on time; | |
| ● | obtain and maintain regulatory approval for such new products; | |
| ● | differentiate our offerings from competitors’ offerings; | |
| ● | achieve positive clinical outcomes; and | |
| ● | provide adequate medical and/or consumer education relating to new products. |
Moreover, innovations generally will require a substantial investment in research and development before we can determine the commercial viability of these innovations and we may not have the financial resources necessary to fund these innovations. In addition, even if we successfully develop enhancements or new generations of our products, these enhancements or new generations of products may not produce revenue in excess of the costs of development and they may be quickly rendered obsolete by changing customer preferences or the introduction by our competitors of products embodying new technologies or features.
We rely on a limited number of suppliers of each of the key components of the TearLab® Osmolarity System and are vulnerable to fluctuations in the availability and price of our suppliers’ products and services.
We purchase each of the key components of the TearLab® Osmolarity System from a limited number of third-party suppliers. Our suppliers may not provide the components or other products needed by us in the quantities requested, in a timely manner or at a price we are willing to pay. In the event we are unable to renew our agreements with our suppliers or they become unable or unwilling to continue to provide important components in the required volumes and quality levels or in a timely manner, or if regulations affecting the components change, we may be required to identify and obtain acceptable replacement supply sources. We may not be able to obtain alternative suppliers or vendors on a timely basis, or at all, which could disrupt or delay, or halt altogether, our ability to manufacture or deliver the TearLab® Osmolarity System. If any of these events should occur, our business, financial condition, cash flows and results of operations could be materially adversely affected.
| -17- |
We face intense competition, and our failure to compete effectively could have a material adverse effect on our results of operations.
We face intense competition in the markets for ophthalmic products and these markets are subject to rapid and significant technological change. We have numerous potential competitors in the United States and abroad, including one direct competitor recently launched in Canada. We face potential competition from industry participants marketing conventional technologies for the measurement of osmolarity and other in-lab testing technologies, and commercially available methods, such as the Schirmer Test and ocular surface staining. Many of our potential competitors have substantially more resources and a greater marketing scale than we do. If we are unable to develop and produce or market our products to effectively compete against our competitors, our operating results will materially suffer.
If we lose key personnel, or we do not attract and retain highly qualified personnel on a cost-effective basis, it would be more difficult for us to manage our existing business operations and to identify and pursue new growth opportunities.
Our success depends, in large part, upon our ability to attract and retain highly qualified scientific, clinical, manufacturing and management personnel. In addition, any difficulties in retaining key personnel or managing this growth could disrupt our operations. Future growth will require us to continue to implement and improve our managerial, operational and financial systems, and to continue to recruit, train and retain, additional qualified personnel, which may impose a strain on our administrative and operational infrastructure. The competition for qualified personnel in the medical technology field is intense. We are highly dependent on our continued ability to attract, motivate and retain highly qualified management, clinical and scientific personnel.
Due to our limited resources, we may not effectively recruit, train and retain additional qualified personnel. If we do not retain key personnel or manage our growth effectively, we may not implement our business plan effectively.
Furthermore, we have not entered into non-competition agreements with our key employees. In addition, we do not maintain “key person” life insurance on any of our officers, employees or consultants. The loss of the services of existing personnel, the failure to recruit additional key scientific, technical and managerial personnel in a timely manner, and the loss of our employees to our competitors would harm our research and development programs and our business.
If we fail to establish and maintain proper and effective internal controls, our ability to produce accurate financial statements on a timely basis could be impaired, which would adversely affect our consolidated operating results, our ability to operate our business and our stock price.
Ensuring that we have adequate internal financial and accounting controls and procedures in place to produce accurate financial statements on a timely basis is a costly and time-consuming effort that needs to be re-evaluated frequently. Failure on our part to maintain effective internal financial and accounting controls would cause our financial reporting to be unreliable, could have a material adverse effect on our business, operating results, financial condition and cash flows, and could cause the trading price of our common stock to fall dramatically.
Maintaining proper and effective internal controls will require substantial management time and attention and may result in our incurring substantial incremental expenses, including with respect to increasing the breadth and depth of our finance organization to ensure that we have personnel with the appropriate qualifications and training in certain key accounting roles and adherence to certain control disciplines within the accounting and reporting function. Any failure in internal controls or any errors or delays in our financial reporting would have a material adverse effect on our business and results of operations and could have a substantial adverse impact on the trading price of our common stock.
| -18- |
Our management is responsible for establishing and maintaining adequate internal control over financial reporting to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of financial statements for external purposes in accordance with U.S. GAAP. Our management does not expect that our internal control over financial reporting will prevent or detect all errors and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Our management has identified control deficiencies in the past and may identify additional deficiencies in the future.
We cannot be certain that the actions we are taking to improve our internal controls over financial reporting will be sufficient or that any changes in processes and procedures can be completed in a timely manner. In future periods, if the process required by Section 404 of the Sarbanes-Oxley Act of 2002 reveals material weaknesses or significant deficiencies, the correction of any such material weaknesses or significant deficiencies could require additional remedial measures which could be costly and time-consuming. In addition, we may be unable to produce accurate financial statements on a timely basis. Any of the foregoing could cause investors to lose confidence in the reliability of our consolidated financial statements, which could cause the market price of our common stock to decline and make it more difficult for us to finance our operations and growth.
Risks Related to Our Common Stock
Our common stock may be delisted from The NASDAQ Capital Market if we cannot maintain compliance with NASDAQ’s continued listing requirements.
In order to maintain our listing on NASDAQ, we are required to comply with NASDAQ requirements, which include, but are not limited to, maintaining a minimum stockholders’ equity or market capitalization and minimum bid price requirement. In particular, we are required to (i) maintain a minimum bid price of $1.00, and we have traded below that threshold since February 2, 2016, and (ii) maintain a minimum market capitalization of $35 million. On March 16, 2016, we received a notice from NASDAQ stating that we were not in compliance with Nasdaq Listing Rule 5550(a)(2) (the “Minimum Bid Price Rule”) because our common stock failed to maintain a minimum closing bid price of $1.00 for 30 consecutive business days. This notice had no immediate effect on the NASDAQ listing or trading of the Company’s common stock.
On September 16, 2016, we received a letter from the NASDAQ Listing Qualifications Staff (the “Staff”). The Staff had determined to delist our securities from the NASDAQ Capital Market due to our continued non-compliance with the Minimum Bid Price Rule within 180 calendar days. To regain compliance with the Minimum Bid Price Rule, we filed a certificate of amendment to our amended and restated certificate of incorporation to effect (i) a reverse stock split of all of the outstanding shares of our common stock and those shares held by us in treasury stock, if any, in a ratio of one-for ten, and (ii) a reduction in the total number of authorized shares of common stock from 95,000,000 to 9,500,000. As of March 3, 2017, the last reported sale price of our common stock was $4.20.
Additionally, on November 8, 2016, we received notice from the Staff indicating that the Company did not satisfy Nasdaq Listing Rule 5550(b)(2), insofar as the Company’s market value of listed securities (“MVLS”) had closed below $35 million for the previous 30 consecutive business days. This notice has no immediate effect on the NASDAQ listing or trading of the Company’s common stock.
In accordance with Nasdaq Listing Rule 5810(c)(3)(C), the Company was granted a 180-calendar day grace period within which to regain compliance with the MVLS requirement, through May 8, 2017. To evidence compliance with the MVLS requirement, the Company’s MVLS must close at $35 million or more for a minimum of 10 consecutive business days during the 180-calendar day compliance period.
| -19- |
If we fail to regain compliance with the applicable requirements, our stock may be delisted. Delisting from the NASDAQ Capital Market could make trading our common stock more difficult for investors, potentially leading to declines in our share price and liquidity. Without a NASDAQ Capital Market listing, stockholders may have a difficult time getting a quote for the sale or purchase of our stock, the sale or purchase of our stock would likely be made more difficult and the trading volume and liquidity of our stock could decline. Delisting from the NASDAQ Capital Market could also result in negative publicity and could also make it more difficult for us to raise additional capital. The absence of such a listing may adversely affect the acceptance of our common stock as currency or the value accorded by other parties. Further, if we are delisted, we would also incur additional costs under state blue sky laws in connection with any sales of our securities. These requirements could severely limit the market liquidity of our common stock and the ability of our stockholders to sell our common stock in the secondary market. If our common stock is delisted by NASDAQ, our common stock may be eligible to trade on an over-the-counter quotation system, such as the OTCQB market, where an investor may find it more difficult to sell our stock or obtain accurate quotations as to the market value of our common stock. We cannot assure you that our common stock, if delisted from the NASDAQ Capital Market, will be listed on another national securities exchange or quoted on an over-the counter quotation system.
If our common stock is delisted, it would come within the definition of “penny stock” as defined in the Securities Exchange Act of 1934, or the Exchange Act, and would be covered by Rule 15g-9 of the Exchange Act. That Rule imposes additional sales practice requirements on broker-dealers who sell securities to persons other than established customers and accredited investors. For transactions covered by Rule 15g-9, the broker-dealer must make a special suitability determination for the purchaser and receive the purchaser’s written agreement to the transaction prior to the sale. Consequently, Rule 15g-9, if it were to become applicable, would affect the ability or willingness of broker-dealers to sell our securities, and accordingly would affect the ability of stockholders to sell their securities in the public market. These additional procedures could also limit our ability to raise additional capital in the future.
The trading price of our common stock may be volatile.
The market prices for, and the trading volumes of, securities of medical device companies, such as ours, have been historically volatile. The market has experienced, from time to time, significant price and volume fluctuations unrelated to the operating performance of particular companies. The market price of our common shares may fluctuate significantly due to a variety of factors, including:
| ● | the results of pre-clinical testing and clinical trials by us, our collaborators and/or our competitors; | |
| ● | technological innovations or new diagnostic products; | |
| ● | governmental regulations and reimbursement levels; | |
| ● | developments in patent or other proprietary rights; | |
| ● | litigation; | |
| ● | public concern regarding the safety of products developed by us or others; | |
| ● | comments by securities analysts; | |
| ● | the issuance of additional shares to obtain financing or for acquisitions; | |
| ● | general market conditions in our industry or in the economy as a whole; | |
| ● | political instability, natural disasters, war and/or events of terrorism; and | |
| ● | failure to maintain our NASDAQ listing. |
In addition, the stock market has experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of individual companies. Broad market and industry factors may seriously affect the market price of our stock, regardless of actual operating performance. In the past, securities class action litigation often follows periods of volatility in the overall market and market price of a particular company’s securities. This litigation, if instituted against us, could result in substantial costs and a diversion of our management’s attention and resources.
| -20- |
Because we do not expect to pay dividends on our common stock, stockholders will benefit from an investment in our common stock only if it appreciates in value.
We have never paid cash dividends on our common stock and have no present intention to pay any dividends in the future. We are not profitable and may not earn sufficient revenue to meet all operating cash needs. As a result, we intend to use all available cash and liquid assets in the development of our business. Any future determination about the payment of dividends will be made at the discretion of our board of directors and will depend upon our earnings, if any, our capital requirements, our operating and financial conditions and on such other factors as our board of directors may deem relevant. As a result, the success of an investment in our common stock will depend upon any future appreciation in its value. There is no guarantee that our common stock will appreciate in value or even maintain the price at which stockholders have purchased their shares.
Warrant holders will not be entitled to any of the rights of common stockholders, but will be subject to all changes made with respect thereto.
If you hold warrants, you will not be entitled to any rights with respect to our common stock (including, without limitation, voting rights and rights to receive any dividends or other distributions on our common stock), but you will be subject to all changes affecting our common stock. You will have rights with respect to our common stock only if you receive our common stock upon exercise of the warrants and only as of the date when you become a record owner of the shares of our common stock upon such exercise. For example, if a proposed amendment to our charter or bylaws requires stockholder approval and the record date for determining the stockholders of record entitled to vote on the amendment occurs prior to the date that you are deemed to be the owner of the shares of our common stock due upon exercise of your warrants, you will not be entitled to vote on the amendment; although, you will nevertheless be subject to any changes in the powers, preferences or special rights of our common stock.
We can issue shares of preferred stock that may adversely affect the rights of holders of our common stock.
Our certificate of incorporation authorizes us to issue up to 10.0 million shares of preferred stock with designations, rights, and preferences determined from time to time by our board of directors. Accordingly, our board of directors is empowered, without stockholder approval, to issue preferred stock with dividend, liquidation, conversion, voting or other rights superior to those of holders of our common stock. For example, an issuance of shares of preferred stock could:
| ● | adversely affect the voting power of the holders of our common stock; | |
| ● | make it more difficult for a third party to gain control of us; | |
| ● | discourage bids for our common stock at a premium; | |
| ● | limit or eliminate any payments that the holders of our common stock could expect to receive upon our liquidation; or | |
| ● | otherwise adversely affect the market price or our common stock. |
| -21- |
Our world-wide headquarters, located in San Diego, California, occupies 14,700 square feet of commercial and industrial space. We use this space for administrative, quality assurance, manufacturing, and research and development activities. The current arrangement extends to June 2018. The total future minimum obligation under this lease is $481,000 for the remaining term of the arrangement with the 2017 obligation being $319,000.
We also lease 7,700 square feet of commercial space in Southlake, Texas for our customer care, marketing, finance, and administrative functions. The lease has a term through February 2018, but we have the option to terminate at any time.
We believe that the California and Texas facilities are suitable and adequate to support our current operations. We believe that if our existing facilities are not adequate to meet our business requirements long-term, additional space will be available on commercially reasonable terms.
We are not aware of any material litigation involving us that is outstanding, threatened or pending.
ITEM 4. Mine Safety Disclosures.
Not applicable.
| -22- |
PART II
ITEM 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Market for Common Equity
Our common stock trades on the NASDAQ Capital Market (“NASDAQ”) under the symbol “TEAR” and the Toronto Stock Exchange (“TSX”) under the symbol “TLB”.
The following table sets forth the range of high and low sales prices per share of our common stock on both the NASDAQ and the TSX for the fiscal periods indicated. On February 23, 2017 we effected a 1-for-10 reverse stock split of our common stock. All prices in the table below have been adjusted to reflect the effect of the reverse stock split.
| Common Stock Prices | ||||||||||||||||
| Fiscal 2016 | Fiscal 2015 | |||||||||||||||
| High | Low | High | Low | |||||||||||||
| NASDAQ Capital Market | ||||||||||||||||
| First Quarter | $ | 16.50 | $ | 6.00 | $ | 31.50 | $ | 15.00 | ||||||||
| Second Quarter | 9.10 | 6.00 | 26.00 | 19.60 | ||||||||||||
| Third Quarter | 9.00 | 5.80 | 30.30 | 18.00 | ||||||||||||
| Fourth Quarter | 6.94 | 3.90 | 22.50 | 11.50 | ||||||||||||
| TSX | ||||||||||||||||
| First Quarter | C$ | 22.60 | C$ | 7.90 | C$ | 40.00 | C$ | 19.20 | ||||||||
| Second Quarter | 12.40 | 7.80 | 32.30 | 25.00 | ||||||||||||
| Third Quarter | 11.60 | 8.00 | 37.90 | 24.30 | ||||||||||||
| Fourth Quarter | 9.10 | 5.10 | 29.20 | 16.10 | ||||||||||||
The closing share price for our common stock on March 3, 2017 as reported by NASDAQ, was $4.20. The closing share price for our common stock on March 3, 2017, as reported by TSX, was C$5.85.
As of March 3, 2017, there were approximately 80 active stockholders of record of our common stock.
Dividend Policy
We have never declared or paid any cash dividends on shares of our capital stock. We currently intend to retain all available funds to support operations and to finance the growth and development of our business. Any determination related to payments of future dividends will be at the discretion of our board of directors after taking into account various factors that our board of directors deems relevant, including our financial condition, operating results, current and anticipated cash needs, plans for expansion and debt restrictions, if any.
Sales of Unregistered Securities
On March 31, 2016, we issued an aggregate of 38,580 shares of common stock of the Company (adjusted for a 1-for-10 reverse stock split) to certain former investors (collectively, “OcuHub Investors”) in OcuHub, in accordance with the OcuHub Investors’ exercise of their right to exchange their equity in OcuHub for shares of common stock of the Company pursuant to the terms of OcuHub’s limited liability company agreement dated as of July 31, 2014. We relied on the exemption from registration contained in Section 4(2) of the Securities Act of 1933, as amended, and Regulation D, Rule 506 thereunder, for such issuance of shares of common stock of the Company.
| -23- |
Stock Performance Graph
The following graph compares the cumulative total stockholder return data for our common stock to the cumulative return of (i) the NASDAQ Composite Index and (ii) the NASDAQ Medical Equipment Index for the period beginning December 31, 2011, and ending on December 31, 2016. The graph assumes that $100 was invested on December 31, 2011, and assumes reinvestment of dividends. The stock price performance on the following graph is not necessarily indicative of future stock price performance.
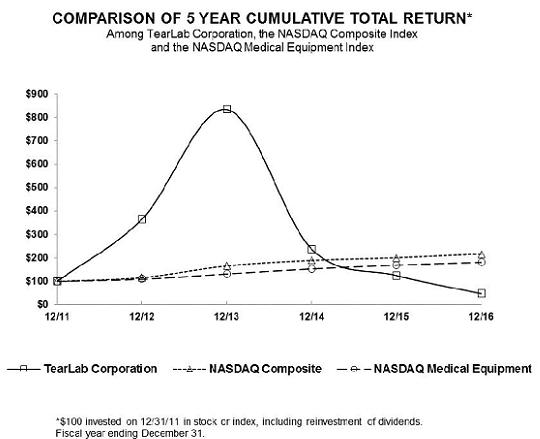
This performance graph shall not be deemed ‘‘filed’’ for purposes of Section 18 of the Exchange Act, or incorporated by reference into any filing of TearLab Corp. under the Securities Act, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| -24- |
ITEM 6. Selected Financial Data.
The following selected financial data should be read in conjunction with our consolidated financial statements, the related notes thereto and the information contained in “Item 7 – Management’s Discussion and Analysis of Financial Condition and Results of Operations.” The consolidated statement of loss data, consolidated balance sheet data, and consolidated other data set forth below as of and for the years ended December 31, 2016, 2015, 2014, 2013, and 2012 have been derived from our audited consolidated financial statements. Historical results are not necessarily indicative of the results to be expected for future periods; the risk factors set forth in Part I, Item 1A, “Risk Factors,” of this report also discuss material uncertainties that could cause the data reflected below not to be indicative of our future financial condition or results of operations. We declared no cash dividends during the periods presented. On February 27, 2017 we effected a 1-for-10 reverse stock split of our common stock. All share amounts and per share prices in the table below have been adjusted to reflect the effect of the reverse stock split.
Table is in thousands except per share data.
| Year Ended December 31, | ||||||||||||||||||||
| 2012 | 2013 | 2014 | 2015 | 2016 | ||||||||||||||||
| Consolidated Statements of Comprehensive Loss Data: | ||||||||||||||||||||
| Revenue | $ | 3,960 | $ | 14,645 | $ | 19,718 | $ | 25,156 | $ | 28,014 | ||||||||||
| Costs and operating expenses | ||||||||||||||||||||
| Cost of goods sold | 2,295 | 8,146 | 10,096 | 12,557 | 12,318 | |||||||||||||||
| Amortization of intangible assets | 1,215 | 1,215 | 1,462 | 1,501 | 1,066 | |||||||||||||||
| General and administrative | 4,770 | 8,942 | 13,555 | 14,935 | 11,057 | |||||||||||||||
| Clinical, regulatory and research and development | 2,241 | 1,095 | 2,792 | 6,951 | 5,152 | |||||||||||||||
| Sales and marketing | 5,471 | 13,021 | 16,817 | 19,349 | 14,397 | |||||||||||||||
| Impairment of long-lived assets | - | - | - | 1,372 | - | |||||||||||||||
| Total costs and operating expenses | 15,992 | 32,419 | 44,722 | 56,665 | 43,990 | |||||||||||||||
| Other income (expense), net | (7,280 | ) | (11,216 | ) | 1,282 | (1,720 | ) | (3,944 | ) | |||||||||||
| Loss from operations before income taxes | (19,312 | ) | (28,990 | ) | (23,722 | ) | (33,229 | ) | (19,920 | ) | ||||||||||
| Recovery of income taxes | - | - | - | - | - | |||||||||||||||
| Net loss | $ | (19,312 | ) | (28,990 | ) | (23,722 | ) | (33,229 | ) | (19,920 | ) | |||||||||
| Per Share Data: | ||||||||||||||||||||
| Net loss per share — basic | $ | (7.58 | ) | $ | (9.42 | ) | $ | (7.06 | ) | $ | (9.86 | ) | $ | (4.29 | ) | |||||
| Net loss per share — diluted | $ | (7.58 | ) | $ | (9.42 | ) | $ | (7.38 | ) | $ | (9.92 | ) | $ | (4.29 | ) | |||||
| Weighted average number of shares used in per share calculations — basic | 2,549 | 3,076 | 3,358 | 3,370 | 4,648 | |||||||||||||||
| Weighted average number of shares used in per share calculations —diluted | 2,549 | 3,076 | 3,372 | 3,373 | 4,648 | |||||||||||||||
| -25- |
| As of December 31, | ||||||||||||||||||||
| 2012 | 2013 | 2014 | 2015 | 2016 | ||||||||||||||||
| Consolidated Balance Sheet Data: | ||||||||||||||||||||
| Cash and cash equivalents | $ | 15,437 | $ | 37,778 | $ | 16,338 | $ | 13,838 | $ | 15,471 | ||||||||||
| Working capital | 9,341 | 34,297 | 16,297 | 14,139 | 16,270 | |||||||||||||||
| Total assets | 24,139 | 49,957 | 31,031 | 28,351 | 26,627 | |||||||||||||||
| Current liabilities | 9,295 | 8,589 | 6,397 | 7,482 | 5,899 | |||||||||||||||
| Long-term debt, net of unamortized discount | - | - | - | 24,859 | 26,449 | |||||||||||||||
| Total liabilities | 9,295 | 8,589 | 6,397 | 32,341 | 32,348 | |||||||||||||||
| Exchange right | - | - | 250 | 250 | - | |||||||||||||||
| Common stock | 29 | 33 | 34 | 34 | 54 | |||||||||||||||
| Additional paid-in capital | 421,662 | 477,172 | 483,909 | 488,514 | 506,933 | |||||||||||||||
| Accumulated deficit | (406,847 | ) | (435,837 | ) | (459,559 | ) | (492,788 | ) | (512,708 | ) | ||||||||||
| Total stockholders’ equity (deficit) | 14,844 | 41,368 | 24,384 | (4,240 | ) | (5,721 | ) | |||||||||||||
| -26- |
ITEM 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and related notes, included in Item 8 of this Report. Unless otherwise specified, all dollar amounts are in U.S. dollars.
Overview
We are an in vitro diagnostic company that has developed a proprietary tear testing platform, the TearLab® Osmolarity System. The TearLab test measures tear film osmolarity for diagnosis of DED. Tear osmolarity is a quantitative and highly specific biomarker that has been shown to correlate with DED. The TearLab test enables the rapid measurement of tear osmolarity in a doctor’s office. Commercializing our Point-of-Care tear testing platform is the focus of our business.
In October 2008, the TearLab® Osmolarity System received CE mark approval, clearing the way for sales in the European Union and all countries recognizing the CE mark. In connection with the CE mark clearance, we have entered into multi-year agreements with numerous distributors for distribution of the TearLab® Osmolarity System. Currently, we have signed distribution agreements in South America, Europe, Asia, Canada, and Australia. We sell directly to the customer in the United States.
On May 19, 2009, we announced that we received 510(k) clearance from the FDA. On January 23, 2012, we announced the FDA had granted the waiver categorization under CLIA for the TearLab® Osmolarity System. The CLIA waiver reduces the regulatory paperwork and related administrative time for customers.
On October 19, 2010 we announced that a unique, new Current Procedural Terminology, or CPT, code that will apply to the TearLab Osmolarity test had been published by the American Medical Association, or AMA. The code became effective January 1, 2011. The CPT code for the TearLab Osmolarity test is: 83861; Microfluidic analysis utilizing an integrated collection and analysis device, tear osmolarity (For microfluidic tear osmolarity of both eyes, report 83861 twice). This code falls under the Chemistry sub-section of the Pathology and Laboratory section of the CPT Codebook and was listed under the 2010 Clinical Laboratory Fee Schedule by the Centers for Medicare and Medicaid Services, or CMS. At 2016 reimbursement rates, payment code 83861 would be reimbursed in every state by CMS at $22.50 per eye. This decision by CMS provides level reimbursement for and equal access to the TearLab Osmolarity Test across all of the United States.
RESULTS OF OPERATIONS
Revenue, Cost of Goods Sold and Gross Profit (in thousands)
| For the years ended December 31, | ||||||||||||||
| % of | % of | |||||||||||||
| 2016 | Revenue | 2015 | Revenue | |||||||||||
| Revenue | $ | 28,014 | 100.0 | % | $ | 25,156 | 100.0 | % | ||||||
| Cost of goods sold | 12,318 | 44.0 | % | 12,557 | 49.9 | % | ||||||||
| Gross profit | $ | 15,696 | 56.0 | % | $ | 12,599 | 50.1 | % | ||||||
| -27- |
Revenue
TearLab revenue consists primarily of sales of the TearLab® Osmolarity System, which is a hand-held tear film test for the measurement of tear osmolarity, a quantitative and highly specific biomarker that has shown to correlate with DED.
The TearLab® Osmolarity System consists of the following three components: (1) the TearLab disposable, which is a single-use microfluidic lab test card; (2) the TearLab pen, which is a hand-held device that interfaces with the TearLab disposable; and (3) the TearLab reader, which is a small desktop unit that allows for the docking of the TearLab disposable and the TearLab pen and provides a quantitative reading for the operator.
Having received 510(k) approval from the FDA in the United States, we sell to customers in the United States who hold CLIA moderate and high complexity certificates and actively support and assist our customers to obtain their moderate complexity CLIA certificates or provide them with support from certified professionals. Having obtained a CLIA waiver certificate in early 2012, we continue to actively support our customers in obtaining their CLIA waiver documentation which will allow us to sell our product to the approximately 50,000 eye care practitioners in the United States that are candidates to operate under a CLIA waiver certification.
We are working with our established distributors in Canada, Europe, South America and Asia to increase sales. The ability for re-imbursement to be obtained in many of those countries where we have distributors will facilitate our ability to increase sales and stimulate the commercialization process. In countries where we have distributors, we are supporting physicians in local clinical trials and providing them with the required guidance to understand the relationship between DED and osmolarity and how to manage their patients with objective diagnostic data.
Revenue increased $2.9 million or 11% for the year ended December 31, 2016 compared to the prior year. The increase in 2016 was due to a $1.7 million increase in test card sales volume and a $1.1 million increase in the sale of TearLab® Osmolarity System readers.
Cost of Goods Sold
TearLab cost of sales includes costs of goods sold, depreciation of reader systems, warranty, and royalty costs. Our cost of goods sold consists primarily of costs for the manufacture of the TearLab® Osmolarity System, including the costs we incur for the purchase of component parts from our suppliers, applicable freight and shipping costs, fees related to merchant services, warehousing and logistics inventory management.
Our cost of sales for the year ended December 31, 2016 decreased $0.2 million or 2% compared to the prior year. The decrease in cost of sales was in spite of higher revenue and test card sales volume, as our per unit cost savings were almost entirely offset by the higher volume of test card sales.
Gross Profit
Gross profit for the year ended December 31, 2016 increased by $3.1 million or 25% compared to the prior year on higher revenue. Gross margin improved to 56%, compared to 50% in the prior year. The improvement in gross margin is primarily attributable to an increase in the make-up of accounts under the FLEX program and the moratorium in the United States on the medical device excise tax.
| -28- |
Operating Expenses (in thousands)
| For the years ended December 31, | ||||||||||||||
| % of | % of | |||||||||||||
| 2016 | Revenue | 2015 | Revenue | |||||||||||
| Sales and marketing | 14,397 | 51.4 | % | 19,349 | 76.9 | % | ||||||||
| Clinical, regulatory and research and development | 5,152 | 18.4 | % | 6,951 | 27.6 | % | ||||||||
| General and administrative | 11,057 | 39.5 | % | 14,935 | 59.4 | % | ||||||||
| Amortization of intangible assets | $ | 1,066 | 3.8 | % | $ | 1,501 | 6.0 | % | ||||||
| Impairment of long-lived assets | - | 0.0 | % | 1,372 | 5.5 | % | ||||||||
| Total operating expenses | $ | 31,672 | 113.1 | % | $ | 44,108 | 175.3 | % | ||||||
Sales and Marketing Expenses
For the year ended December 31, 2016, sales and marketing expenses decreased by $5.0 million or 26% as compared with the prior year. The decrease is due primarily to cost savings from our February 2016 organizational restructuring and approximately $0.6 million from the divestiture of our former subsidiary, OcuHub LLC.
Clinical, Regulatory and Research and Development
For the year ended December 31, 2016, clinical, regulatory and research and development expenses decreased by $1.8 million or 26%, as compared with the prior year. The decrease was primarily due to a decrease in external product development costs related to expenses for our next generation diagnostic platform.
General and Administrative Expenses
General and administrative expenses decreased $3.9 million or 26% for the year ended December 31, 2016 compared to 2015. The decrease in general and administrative expenses was due to $1.9 million of cost savings from our divestiture of OcuHub, $1.1 million higher professional services costs in 2015 from transitioning our office support team from California to Texas, and $0.7 million reduced stock compensation in 2016 compared to 2015.
Amortization of Intangible Assets
Amortization expense of intangible assets for the year ended December 31, 2016 decreased $0.4 million or 29% compared to 2015. The decrease was due in part to $313,000 of amortization of OcuHub related intangibles in 2015, which did not recur in 2016, and TearLab® technology acquired with the acquisition of TearLab Research, Inc. in 2006 fully amortizing in November 2016.
Impairment of Long-lived Assets
As more fully described in Note 4 to the Consolidated Financial Statements, the Company performs an evaluation of long-lived assets and intangible assets for impairment when certain indicators of impairment are present. During the year ended December 31, 2015, the performance of the OcuHub subsidiary indicated potential impairment. The evaluation resulted in an impairment of $1.4 million to the intangible and tangible long-lived assets of OcuHub, recorded to impairment of long-lived assets on the Consolidated Statements of Operations. The assessment for the year ended December 31, 2016 did not result in a recorded impairment.
| -29- |
Other Income (Expense), Net (in thousands)
| For the years ended December 31, | ||||||||||||||
| % of | % of | |||||||||||||
| 2016 | Revenue | 2015 | Revenue | |||||||||||
| Interest expense | $ | (4,003 | ) | -14.3 | % | $ | (2,023 | ) | -8.0 | % | ||||
| Changes in fair value of warrant obligations | 28 | 0.1 | % | 227 | 0.9 | % | ||||||||
| Other, net | 31 | 0.1 | % | 76 | 0.3 | % | ||||||||
| Total other income (expense) | $ | (3,944 | ) | -14.1 | % | $ | (1,720 | ) | -6.8 | % | ||||
Interest Expense
Interest expense is generated from our long-term debt under the Term Loan Agreement, which the Company entered into in March 2015. Interest expense increased in 2016 on larger average balances of long-term debt outstanding.
Changes in Fair Value of Warrant Obligation
During 2015 and thru the second quarter of 2016, the Company had outstanding warrants that were presented as liabilities, recorded at their fair value as of the end of each reporting period. Adjustments to the warrant obligations were recorded as a gain or loss in the applicable reporting period to other income (expense) in the Consolidated Statements of Operations and Comprehensive Loss. On June 30, 2016, all outstanding warrants presented as a liability on the Consolidated Balance Sheets expired.
Other Income (Expense)
Other expense for the years ended December 31, 2016 and 2015 consists primarily of foreign exchange gains and losses, based on fluctuations of the Company’s foreign denominated currencies.
Liquidity and Capital Resources (in thousands)
| As of December 31, | ||||||||
| 2015 | 2015 | |||||||
| Cash and cash equivalents | $ | 15,471 | $ | 13,838 | ||||
| Percentage of total assets | 58.1 | % | 48.8 | % | ||||
| Working capital | $ | 16,270 | $ | 14,139 | ||||
Financial Condition
Based on our current rate of cash consumption, we estimate we will need additional capital during the first quarter of 2018 and our prospects for obtaining that capital are uncertain. The Company may be able to raise either additional debt financing or additional equity financing. However, the Company can make no assurances that it will be able to raise the required additional capital, either through debt or equity financing, on acceptable terms or at all. Unless we succeed in raising additional capital or we significantly reduce the cash consumed in the operations of the Company, we anticipate that we will be unable to continue our operations through the end of the first quarter of 2018, without violating an existing covenant on the Term Loan Agreement. As a result of the Company’s historical losses and financial condition, there is substantial doubt about the Company’s ability to continue as a going concern.
| -30- |
Our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement and involves risks and uncertainties. Actual results could vary as a result of a number of factors. We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect. Our future funding requirements will depend on many factors, including but not limited to:
| ● | Our ability to execute our commercial strategy with our current resources | |
| ● | whether government and third-party payers agree to reimburse the TearLab® Osmolarity System; | |
| ● | whether eye care professionals engage in the process of obtaining their CLIA waiver certification; | |
| ● | the costs and timing of building the infrastructure to market and sell the TearLab Osmolarity System;•ἛἛ | |
| ● | the cost and results of continuing development of the TearLab® Osmolarity System; | |
| ● | the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights and | |
| ● | the effect of competing technological and market developments. |
At the present time, our only product is the TearLab® Osmolarity System, and although we have received 510(k) approval from the FDA and a CLIA waiver approval from the FDA, at this time, we do not know when we can expect to begin to generate sufficient revenue from the TearLab® Osmolarity System in the United States to fully fund our operations. If events or circumstances occur such that we do not meet our plans to fund the business, we may be required to reduce operating expenses and reduce the planned levels of inventory and fixed assets which could have an adverse impact on our ability to achieve our intended business objectives.
Indebtedness
On March 4, 2015, the Company executed the Term Loan Agreement with CRG as lenders providing the Company with access of up to $35.0 million under the Term Loan Agreement. The Company entered into a second amendment to the Term Loan Agreement with CRG on August 6, 2015. The Company received $15.0 million in gross proceeds under the Term Loan Agreement on March 4, 2015, and an additional $10.0 million on October 6, 2015. We were unable to access a third tranche of $10.0 million because we did not attain at least $38.0 million in trailing twelve-month revenue prior to June 30, 2016, as required to access the third tranche. As part of the second amendment to the Term Loan Agreement, CRG received warrants to purchase 35,000 common shares in the Company at a price of $50.00 per share (affected for a 1-for-10 reverse stock split). The warrants have a life of five years. On April 7, 2016, the Term Loan Agreement was further amended, under the fourth amendment, to change the required minimum revenue levels. In addition, the fourth amendment reduced the exercise price of the warrants CRG received under the second amendment to $15.00 per share and granted CRG additional warrants to purchase an additional 35,000 common shares in the Company at a price of $15.00 per share. The Term Loan Agreement has a term of six years and bears interest at 13% per annum, with quarterly payments of interest only for the first four years. At the Company’s option, during the first four years a portion of the interest payments amounting to a 4.5% per annum rate may be deferred and paid together with the principal in the fifth and sixth years.
The Term Loan Agreement is collateralized by all assets of the Company. Additionally, the terms of the Term Loan Agreement contain various affirmative and negative covenants agreed to by the Company. Among them, the Company must attain minimum certain annual revenue and minimum cash threshold levels. The minimum annual revenue threshold level required by the Term Loan Agreement for calendar year 2016 is $27.0 million. The minimum cash balance required is $5.0 million, subject to certain conditions.
| -31- |
If the Company does not have annual revenue greater or equal to the annual revenue covenant in a calendar year, the Company will have to raise subordinated debt or equity (the “CRG Equity Cure”) equal to twice the difference between the annual revenue and the revenue covenant, with the total proceeds from this financing to be used to reduce the principal of the Term Loan Agreement. In the event the Company does not achieve the minimum revenue threshold and it cannot complete the CRG Equity Cure, it may be in default of the Term Loan Agreement. In the event of a default, we may be required to repay any outstanding amounts earlier than anticipated, and the lenders may foreclose on their security interest in our assets.
Ongoing Sources and Uses of Cash
We anticipate that our cash and cash equivalents and cash generated from business operations will be sufficient to sustain our operations until the first quarter of 2018. We continually evaluate various financing possibilities but we typically expect our primary source of cash will be related to the collection of accounts receivable. Our accounts receivable collections will be impacted by our ability to grow our point-of-care revenue.
We expect to use our cash primarily to fund our operating expenses and pursue and maintain our patents and trademarks. In addition, dependent on available funds, we expect to expend cash to improve production capability of the TearLab test, to further improve the performance of the TearLab test, and to pursue next generation products using our lab-on-a-chip technology.
Changes in Cash Flows (in thousands)
| Years ended December 31, | ||||||||
| 2015 | 2014 | |||||||
| Cash used in operating activities | $ | (12,516 | ) | $ | (23,703 | ) | ||
| Cash used in investing activities | (1,434 | ) | (3,388 | ) | ||||
| Cash provided by financing activities | 15,583 | 24,591 | ||||||
| Net decrease in cash and cash equivalents during the period | $ | 1,633 | $ | (2,500 | ) | |||
Cash Used in Operating Activities
Net cash used in our operating activities for 2016 was $12.5 million. Net cash used in operating activities was less than our net loss for the year of $19.9 million primarily due to stock compensation expense, deferred interest on our long-term debt, depreciation and amortization. In aggregate, these non-cash expenses totaled $7.7 million during the year.
Net cash used to fund our operating activities during the year ended December 31, 2015 was $23.7 million. Net cash used in operating activities was less than our net loss for the year of $33.2 million primarily due to stock compensation expense, depreciation and amortization, and an impairment to long-lived assets during the year. In aggregate, these non-cash expenses totaled $9.1 million during the year.
| -32- |
The net change in non-cash working capital and non-current asset balances related to operations for the years ended December 31, 2016 and 2015 consists of the following (in thousands):
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Accounts receivable, net | $ | 742 | $ | (540 | ) | |||
| Inventory | 780 | (987 | ) | |||||
| Prepaid expenses and other assets | (458 | ) | 100 | |||||
| Other non-current assets | (44 | ) | (24 | ) | ||||
| Accounts payable | (335 | ) | 90 | |||||
| Accrued liabilities | (1,093 | ) | 1,318 | |||||
| Deferred rent/revenue | (24 | ) | (60 | ) | ||||
| $ | (432 | ) | $ | (103 | ) | |||
Explanations of the more significant net changes in working capital and non-current asset balances are as follows:
| ● | Accounts receivable decreased in 2016 due to improved collection efforts. Accounts receivable increased in 2015 on increased year-over-year revenue; | |
| ● | Inventory decreased in 2016 due to a focused effort to reduce the number of days sales on hand and cost savings under a new supply agreement with our supplier of test cards. Inventory increased in 2015 as we built our inventory to a level to satisfy anticipated near-term demand; | |
| ● | Prepaid expenses and other assets increased in 2016 due primarily to timing of payments on annual insurance policies and certain manufacturing deposits; | |
| ● | Accounts payable and accrued liabilities decreased in 2016 due to lower unbilled progress on product development, accrued fees under our former supply agreement for test cards that did not occur in 2016, and lower accrued professional service fees. Accrued liabilities increased in 2015, primarily due to unbilled progress on the development associated with the next generation diagnostic products. |
Cash used in Investing Activities
Net cash used in investing activities for the years ended December 31, 2016 and 2015 was $1.4 million and $3.4 million, respectively, which we used to acquire fixed assets, primarily the TearLab® Osmolarity System readers.
Cash Provided by Financing Activities
Net cash provided by financing activities for the year ended December 31, 2016 was $15.6 million, which was predominantly the proceeds from the issuance of common and preferred stock during the year. Net cash provided by financing activities for the year ended December 31, 2015 was $24.6 million and relates to proceeds received during the year under our Term Loan Agreement.
| -33- |
The following table summarizes our contractual commitments as of December 31, 2016 and the effect those commitments are expected to have on liquidity and cash flow in future periods (in thousands):
| Less Than | 1 To 3 | More Than | ||||||||||||||
| Contractual obligation | Total | 1 Year | Years | 3 Years | ||||||||||||
| Operating leases | $ | 536 | $ | 354 | $ | 182 | $ | - | ||||||||
| Royalty payments | 770 | 70 | 140 | 560 | ||||||||||||
| Marketing service agreements | 225 | 100 | 125 | - | ||||||||||||
| Purchase obligations | 1,143 | 1,143 | - | - | ||||||||||||
| Long-term debt | 25,000 | - | 9,375 | 15,625 | ||||||||||||
| Interest payments | 15,294 | 3,494 | 7,364 | 4,436 | ||||||||||||
| Total contractual obligations | $ | 42,968 | $ | 5,161 | $ | 17,168 | $ | 20,621 | ||||||||
Off-Balance-Sheet Arrangements
As of December 31, 2016, we did not have any material off-balance-sheet arrangements.
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations is based upon our audited consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States (“US GAAP”). The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, sales and expenses, and related disclosure of contingent assets and liabilities. On an ongoing basis, we evaluate our estimates, including those related to our intangible assets, uncollectible receivables, inventories, debt and equity instruments and stock-based compensation. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Because this can vary in each situation, actual results may differ from these estimates under different assumptions or conditions.
We believe the following critical accounting policies affect our more significant judgments and estimates used in the preparation of our audited consolidated financial statements.
Revenue recognition
Revenue is recognized when all four of the following criteria are met: (i) persuasive evidence that an arrangement exists; (ii) delivery of the products has occurred; (iii) the selling price is fixed or determinable; and (iv) collectability is reasonably assured. The Company records revenue when all of its obligations are completed, which is generally upon shipment of the Company’s products. Amounts received in excess of revenue recognizable are deferred.
Our revenue is primarily derived from the sale of disposable test cards. We sell our proprietary TearLab® Osmolarity System and related test cards to our customers, who are primarily eye care professionals, for use in osmolarity testing procedures. Our products are generally shipped from our primary distribution and warehousing operations facility located in San Diego, California. Sales are direct to customers in the United States and to distributors in the rest of the world.
| -34- |
The Company enters into contracts where revenue is derived either from agreements whereby the customer is provided the right to use the TearLab® Osmolarity System (reader equipment) at no separate cost to the customer in consideration for a minimum or implied purchase commitment of disposables over the related contract term (referred to as either “Use Agreements”, “Masters Agreements” or “Flex Agreements”), or from agreements with sales of multiple deliverables, such as the reader equipment and disposable test cards (referred to as “Purchase Agreements”).
Purchase commitments for Use Agreements and Flex Agreements are expressed in the agreement for a specified period of time (generally one to three years), and the purchase commitment for Masters Agreements is implied for large physician practices with an expectation of purchasing certain levels of test cards. The Company recovers the cost of providing the reader equipment in the amount charged for disposables. These agreements are treated as operating leases as collectability of the minimum lease payments is not reasonably predictable at the outset of the arrangement. Accordingly, revenue is recognized over the defined contract term as disposable test cards are shipped. Revenue under such agreements is allocated between the lease of the reader equipment and the sale of the disposables based upon each component’s relative fair value. When reader equipment is placed with a customer at no separate cost, the Company retains title to the equipment and it remains capitalized on the Company’s consolidated balance sheet as equipment classified within fixed assets, net. After it has been shipped to a customer location, equipment is depreciated on a straight-line basis over its estimated useful life and depreciation expense is included in cost of goods sold within the Consolidated Statement of Operations and Comprehensive Loss.
Revenue recognition for Purchase Agreements with multiple deliverables is based on the individual units of accounting determined to exist in the contract, with the test cards and the TearLab reader considered the individual units. Test cards are essential to the operation of a TearLab reader, there is no alternative vendor for the test cards and no indication that a secondary market for the TearLab readers is established. Therefore, the deliverables under the contracts entered into during 2016 and 2015 do not meet criteria for separation under the multiple-element arrangements guidance. Consideration is allocated at the inception of the contract to all deliverables based on their relative selling price, as determined by our selling price of similar individual items on a stand-alone basis. The Company recognizes revenue for each of the elements only when it determines that all applicable recognition criteria have been met.
Although the Company typically has a no return policy for its products, we have established a return reserve for product sales that contain an implicit right of return. Reserves for estimated returns or refunds are netted against accounts receivable and reduce revenue at the time of shipment based on historical experience.
Valuation of intangible and other long-lived assets
We periodically assess the carrying value of intangible and other long-lived assets, which requires us to make assumptions and judgments regarding the future cash flows of these assets. The assets are considered to be impaired if we determine that the carrying value may not be recoverable based upon our assessment of the following events or changes in circumstances:
| ● | the asset’s ability to continue to generate income from operations and positive cash flow in future periods; | |
| ● | loss of legal ownership or title to the asset; | |
| ● | significant changes in our strategic business objectives and utilization of the asset(s); and | |
| ● | the impact of significant negative industry or economic trends. |
| -35- |
If the assets are considered to be impaired, the impairment is the amount by which the carrying value of the assets exceeds the fair value of the assets. Fair value is determined by a combination of available third party sources and discounted cash flows. In addition, we base the useful lives and related amortization or depreciation expense on our estimate of the period that the assets will generate revenue or otherwise be used by us. We also periodically review the lives assigned to our intangible assets to ensure that our initial estimates do not exceed any revised estimated periods from which we expect to realize cash flows from the technologies. If a change were to occur in any of the above-mentioned factors or estimates, the likelihood of a material change in our reported results would increase.
As of December 31, 2016, the net book value of fixed assets equaled $4.2 million.
Warrant liabilities
The Company issued several rounds of warrants related to various debt and equity transactions that occurred in 2011. The Company accounts for its warrants issued in accordance with US GAAP accounting guidance applicable to derivative instruments, which requires every derivative instrument within its scope to be recorded on the balance sheet as either an asset or liability measured at its fair value, with changes in fair value recognized in earnings. Based on this guidance, the Company determined that these warrants did not meet the criteria for classification as equity. Accordingly, the Company classified the warrants as current liabilities. The warrants were subject to remeasurement at each balance sheet date, with any change in fair value recognized as a component of other income (expense), net in the statements of operations. We estimated the fair value of these warrants at the respective balance sheet dates using the Black-Scholes Merton option pricing model based on the estimated market value of the underlying common stock at the valuation measurement date, the remaining contractual term of the warrant, risk-free interest rates and expected dividends on and expected volatility of the price of the underlying common stock. Option pricing models employ subjective factors to estimate warrant liability; and, therefore, the assumptions used in the Black-Scholes Merton option pricing model are judgmental. During 2016, all remaining warrants from the 2011 issuances expired.
The Company issued warrants in 2015 and 2016 associated with our Term Loan Agreement and our 2016 issuance of common and preferred stock. These warrants did meet the criteria for classification as equity. We estimated the fair value of these warrants at the date of issuance using the Black-Scholes Merton option pricing model as described above. The fair value of the warrants issued in 2015 and 2016 were included as a component of equity on the Consolidated Balance Sheets and are not adjusted for fair value at the balance sheet date.
For information on the recent accounting pronouncements impacting our business, see Note 2 of the Notes to Consolidated Financial Statements included in Item 8.
Allowance for doubtful accounts
We maintain an allowance for doubtful accounts to reflect estimated losses resulting from our customers’ inability to make required payments. On an on-going basis, we evaluate the collectability of accounts receivable based on a combination of factors, including historical collection trends, current economic factors, and the assessment of collectability of specific accounts.
| -36- |
ITEM 7A. Quantitative and Qualitative Disclosures about Market Risk.
Currency Fluctuations and Exchange Risk
Our sales are denominated primarily in U.S. dollars. Most of our expenses are denominated in U.S. dollars, however, some of our research and development expenses are in Australian dollars and a minor portion of our other expenses are in Canadian dollars, Australian dollars, euro and pounds sterling. We cannot predict any future trends in the exchange rate of the Canadian dollar, Australian dollar, euro or pound sterling against the U.S. dollar. Any strengthening of the Canadian dollar, Australian dollar, euro or pound sterling in relation to the U.S. dollar would increase the U.S. dollar cost of our operations, and affect our U.S. dollar measured results of operations. We maintain bank accounts in Canadian dollars to meet short term operating requirements. We do not engage in any hedging or other transactions intended to manage these risks. In the future, we may undertake hedging or other similar transactions or invest in market risk sensitive instruments if we determine that is advisable to offset these risks.
Interest Rate Risk
Our long-term debt carries a fixed rate of 13% interest. A decrease in market interest rates would increase the fair value of our long-term debt.
| -37- |
ITEM 8. Financial Statements and Supplementary Data
Consolidated Financial Statements
| -38- |
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders
TearLab Corporation
We have audited the accompanying consolidated balance sheets of TearLab Corporation (the “Company”) as of December 31, 2016 and 2015, and the related consolidated statements of operations and comprehensive loss, stockholders’ equity (deficit), and cash flows for each of the two years in the period ended December 31, 2016, and the related notes to the consolidated financial statements. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of the Company as of December 31, 2016 and 2015, and the consolidated results of their operations and their cash flows for each of the two years in the period ended December 31, 2016, in conformity with accounting principles generally accepted in the United States of America.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has incurred recurring operating losses and is dependent on additional financing to fund operations. These conditions raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters are described in Note 1 to the consolidated financial statements. The consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from the outcome of this uncertainty.
| /s/ Mayer Hoffman McCann P.C. | |
| San Diego, California | |
| March 10, 2017 |
| -39- |
TearLab Corporation
(in thousands, except share and per share data)
| December 31, 2016 | December 31, 2015 | |||||||
| ASSETS | ||||||||
| Current assets | ||||||||
| Cash | $ | 15,471 | $ | 13,838 | ||||
| Accounts receivable, net | 2,279 | 3,021 | ||||||
| Inventory | 3,193 | 3,972 | ||||||
| Prepaid expenses and other current assets | 1,226 | 790 | ||||||
| Total current assets | 22,169 | 21,621 | ||||||
| Fixed assets, net | 4,178 | 5,352 | ||||||
| Patents and trademarks, net | 26 | 52 | ||||||
| Intangible assets, net | 34 | 1,145 | ||||||
| Other non-current assets | 220 | 181 | ||||||
| Total assets | $ | 26,627 | $ | 28,351 | ||||
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 1,858 | $ | 2,292 | ||||
| Accrued liabilities | 3,958 | 5,047 | ||||||
| Deferred Rent | 83 | 114 | ||||||
| Obligations under warrants | - | 29 | ||||||
| Total current liabilities | 5,899 | 7,482 | ||||||
| Long-term debt, net | 26,449 | 24,859 | ||||||
| Total liabilities | 32,348 | 32,341 | ||||||
| Exchange right | - | 250 | ||||||
| Commitments and contingencies (Note 10) | ||||||||
| Stockholders’ deficit | ||||||||
| Capital stock | ||||||||
| Preferred Stock, $0.001 par value, 10,000,000 authorized, 2,764 issued and outstanding at December 31, 2016 and none issued and outstanding at December 31, 2015 | - | - | ||||||
| Common stock, $0.001 par value, 9,500,000* and 6,500,000* authorized, 5,360,198* and 3,376,090* issued and outstanding at December 31, 2016 and December 31, 2015, respectively | 54 | 34 | ||||||
| Additional paid-in capital | 506,933 | 488,514 | ||||||
| Accumulated deficit | (512,708 | ) | (492,788 | ) | ||||
| Total stockholders’ deficit | (5,721 | ) | (4,240 | ) | ||||
| Total liabilities and stockholders’ deficit | $ | 26,627 | $ | 28,351 | ||||
* Restated for a 1-for-10 reverse stock split effected February 27, 2017.
See accompanying notes to consolidated financial statements.
| -40- |
TearLab Corporation
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(in thousands, except per share data)
| Years Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Revenue | ||||||||
| Product sales | $ | 23,809 | $ | 20,325 | ||||
| Reader equipment rentals | 4,205 | 4,831 | ||||||
| Total revenue | 28,014 | 25,156 | ||||||
| Cost of goods sold | ||||||||
| Cost of goods sold (excluding amortization of intangible assets) | 10,188 | 10,825 | ||||||
| Cost of goods sold - reader equipment depreciation | 2,130 | 1,732 | ||||||
| Gross profit | 15,696 | 12,599 | ||||||
| Operating expenses | ||||||||
| Sales and marketing | 14,397 | 19,349 | ||||||
| Clinical, regulatory and research & development | 5,152 | 6,951 | ||||||
| General and administrative | 11,057 | 14,935 | ||||||
| Amortization of intangible assets | 1,066 | 1,501 | ||||||
| Impairment of long-lived assets | - | 1,372 | ||||||
| Total operating expenses | 31,672 | 44,108 | ||||||
| Loss from operations | (15,976 | ) | (31,509 | ) | ||||
| Other income (expense) | ||||||||
| Interest expense | (4,003 | ) | (2,023 | ) | ||||
| Changes in fair value of warrant obligations | 28 | 227 | ||||||
| Other, net | 31 | 76 | ||||||
| Total other income (expense) | (3,944 | ) | (1,720 | ) | ||||
| Net loss and comprehensive loss | $ | (19,920 | ) | $ | (33,229 | ) | ||
| Weighted average shares outstanding - basic * | 4,647,983 | 3,369,808 | ||||||
| Net loss per share – basic * | $ | (4.29 | ) | $ | (9.86 | ) | ||
| Weighted average shares outstanding - diluted * | 4,647,983 | 3,373,180 | ||||||
| Net loss per share – diluted * | $ | (4.29 | ) | $ | (9.92 | ) | ||
* Restated for a 1-for-10 reverse stock split effected February 27, 2017.
See accompanying notes to consolidated financial statements.
| -41- |
TearLab Corporation
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY (DEFICIT)
(in thousands, except share data)
| Series
A Convertible | Additional | Stockholders’ | ||||||||||||||||||||||||||
| Common stock | Preferred stock | paid-in | Accumulated | Equity | ||||||||||||||||||||||||
| Shares* | Amount | Shares | Amount | Capital | Deficit | (deficit) | ||||||||||||||||||||||
| Balance, December 31, 2014 | 3,364,130 | $ | 34 | - | $ | - | $ | 483,909 | $ | (459,559 | ) | $ | 24,384 | |||||||||||||||
| Stock-based compensation | - | - | - | - | 4,117 | - | 4,117 | |||||||||||||||||||||
| Common Stock Warrants issued | - | - | - | - | 290 | - | 290 | |||||||||||||||||||||
| Options exercised | 6,539 | - | - | - | 99 | - | 99 | |||||||||||||||||||||
| Issuance of employee purchase plan shares | 5,421 | - | - | - | 99 | - | 99 | |||||||||||||||||||||
| Net loss and comprehensive loss | - | - | - | - | - | (33,229 | ) | (33,229 | ) | |||||||||||||||||||
| Balance, December 31, 2015 | 3,376,090 | 34 | - | - | 488,514 | (492,788 | ) | (4,240 | ) | |||||||||||||||||||
| Common stock issued | 1,861,090 | 19 | - | - | 9,613 | - | 9,632 | |||||||||||||||||||||
| Series A Convertible Preferred stock issued | - | - | 3,292 | - | 2,275 | - | 2,275 | |||||||||||||||||||||
| Series A Convertible Preferred conversion to common | 70,333 | 1 | (528 | ) | - | (1 | ) | - | - | |||||||||||||||||||
| Exchanged shares | 38,580 | - | - | - | 250 | - | 250 | |||||||||||||||||||||
| Stock-based compensation | - | - | - | - | 2,447 | - | 2,447 | |||||||||||||||||||||
| Common Stock Warrants issued | - | - | - | - | 3,709 | - | 3,709 | |||||||||||||||||||||
| Issuance of employee purchase plan shares | 14,105 | - | - | - | 126 | - | 126 | |||||||||||||||||||||
| Net loss and comprehensive loss | - | - | - | - | - | (19,920 | ) | (19,920 | ) | |||||||||||||||||||
| Balance, December 31, 2016 | 5,360,198 | $ | 54 | 2,764 | $ | - | $ | 506,933 | $ | (512,708 | ) | $ | (5,721 | ) | ||||||||||||||
* Restated for a 1-for-10 reverse stock split effected February 27, 2017.
See accompanying notes to consolidated financial statements.
| -42- |
TearLab Corporation
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| OPERATING ACTIVITIES | ||||||||
| Net loss for the period | $ | (19,920 | ) | $ | (33,229 | ) | ||
| Adjustments to reconcile net loss to cash used in operating activities: | ||||||||
| Stock-based compensation | 2,447 | 4,117 | ||||||
| Depreciation of fixed assets | 2,364 | 2,075 | ||||||
| Amortization of patents and trademarks | 28 | 28 | ||||||
| Amortization of intangible assets | 1,111 | 1,512 | ||||||
| Impairment of long-lived assets | - | 1,372 | ||||||
| Changes in fair value of warrant obligations | (29 | ) | (227 | ) | ||||
| (Gain) loss on disposal of fixed assets | 165 | (3 | ) | |||||
| Deferred interest on long-term debt | 1,517 | 681 | ||||||
| Amortization of deferred financing charges | 233 | 74 | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable, net | 742 | (540 | ) | |||||
| Inventory | 780 | (987 | ) | |||||
| Prepaid expenses and other assets | (458 | ) | 100 | |||||
| Other non-current assets | (44 | ) | (24 | ) | ||||
| Accounts payable | (335 | ) | 90 | |||||
| Accrued liabilities | (1,093 | ) | 1,318 | |||||
| Deferred rent/revenue | (24 | ) | (60 | ) | ||||
| Cash used in operating activities | (12,516 | ) | (23,703 | ) | ||||
| INVESTING ACTIVITIES | ||||||||
| Additions to fixed assets, net of proceeds | (1,434 | ) | (3,388 | ) | ||||
| Cash used in investing activities | (1,434 | ) | (3,388 | ) | ||||
| FINANCING ACTIVITIES | ||||||||
| Proceeds from the issuance of shares and warrants | 15,457 | - | ||||||
| Proceeds from the issuance of term loan | - | 24,393 | ||||||
| Proceeds from the issuance of employee stock purchase plan shares | 126 | 99 | ||||||
| Proceeds from the exercise of stock options | - | 99 | ||||||
| Cash provided by financing activities | 15,583 | 24,591 | ||||||
| Net Increase (decrease) in cash and cash equivalents during the year | 1,633 | (2,500 | ) | |||||
| Cash and cash equivalents, beginning of year | 13,838 | 16,338 | ||||||
| Cash and cash equivalents, end of year | $ | 15,471 | $ | 13,838 | ||||
| Supplemental Cash flow information | ||||||||
| Cash paid for interest | $ | 2,258 | $ | 1,192 | ||||
| Supplemental disclosure of noncash investing and financing activities | ||||||||
| Issuance of Warrants | $ | 3,709 | $ | 290 | ||||
See accompanying notes to consolidated financial statements.
| -43- |
TearLab Corporation
Notes to Consolidated Financial Statements
(expressed in thousands of U.S. dollars except per share amounts and as otherwise noted)
1. Basis of Presentation
Nature of Operations
TearLab Corporation (“TearLab” or the “Company”), a Delaware corporation, is an ophthalmic device company that is commercializing a proprietary in vitro diagnostic tear testing platform, the TearLab® Osmolority System to test for dry eye disease, or DED, which enables eye care practitioners to test for highly sensitive and specific biomarkers using nanoliters of tear film at the point-of-care and operates in one business segment.
The accompanying consolidated financial statements include the accounts of the Company and all of its wholly owned subsidiaries, TearLab Research, Inc. (“TearLab Research”), Occulogix Canada Corporation, and OcuHub Holdings, Inc. (formerly, Occulogix LLC). On April 8, 2016 OcuHub Holdings, Inc., a wholly-owned subsidiary of the Company, completed the sale of 10,167.5 units of OcuHub LLC, (“OcuHub”), reducing OcuHub Holdings, Inc.’s ownership in OcuHub to 10.5% on a fully-diluted basis. Prior to the sale, the accounts of OcuHub were included in the consolidated financial statements. In the twelve months ended December 31, 2016, the Company recorded a gain on the sale of OcuHub of $75, included in Other income (expense) on the Consolidated Statements of Operations and Comprehensive Loss. Intercompany accounts and transactions have been eliminated on consolidation.
On February 23, 2017, the Company’s stockholders authorized the board of directors to implement a reverse stock split, along with a corresponding reduction in the number of shares authorized. On February 27, 2017, the Company effected a 1-for-10 reverse stock split of its common stock. All common stock share amounts and prices per share of common stock in these Consolidated Financial Statements have been retroactively adjusted to reflect the reverse stock split.
Liquidity and Going Concern
The accompanying consolidated financial statements have been prepared on the going concern basis, which assumes that the Company will continue to operate as a going concern and which contemplates the realization of assets and the satisfaction of liabilities and commitments in the normal course of business. The Company has sustained substantial losses of $19,920 and $33,229 for the years ended December 31, 2016 and 2015, respectively. Based on the Company’s current rate of cash consumption, the Company estimates it will need additional capital in the first quarter of 2018 and its prospects for obtaining that capital are uncertain. The Company may be able to raise either additional debt financing or additional equity financing. However, the Company can make no assurances that it will be able to raise the required additional capital, either through debt or equity financing, on acceptable terms or at all. Unless the Company succeeds in raising additional capital or significantly reduces the cash consumed in the operations of the Company, the Company anticipates that it will be unable to continue operations through the end of the first quarter of 2018 without violating an existing covenant on the Term Loan Agreement (defined below). As a result of the Company’s historical losses and financial condition, there is substantial doubt about the Company’s ability to continue as a going concern.
| -44- |
2. SIGNIFICANT ACCOUNTING POLICIES
Use of estimates
The preparation of financial statements in conformity with U.S. Generally Accepted Accounting Principles (‘‘GAAP’’) requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenue and expenses during the reporting periods. Actual results could differ from those estimates. The principal areas of judgment relate to revenue and inventory reserves, allowance for doubtful accounts, impairment of long-lived and intangible assets, valuation allowance on deferred tax assets and the fair value of stock options and warrants.
Concentrations and risk
Credit risk
Financial instruments, which potentially subject the Company to concentrations of credit risk, consist primarily of cash and accounts receivable. The Company limits its exposure to credit loss by placing its cash with high credit quality financial institutions.
During 2016 and 2015, the Company derived its revenue from the sale of the TearLab® Osmolarity product. There were no customers representing revenue in excess of 10% in the years ended December 31, 2016 or 2015.
Currently, there are two suppliers for the reader and pen components of the TearLab® Osmolarity System and one supplier for the test cards. The Company expects to maintain the relationships with these suppliers.
Fair value of financial instruments
The Company’s financial instruments consist principally of cash and cash equivalents, accounts receivable, accounts payable, accrued expenses and term debt. The carrying amounts of financial instruments such as cash equivalents, accounts receivable, accounts payable and accrued expenses approximate the related fair values due to the short-term maturities of these instruments. The term debt is presented net of any unamortized premiums or discounts, which approximates fair value.
Cash and cash equivalents
The Company considers all highly liquid investments that are readily convertible into cash and have an original maturity of three months or less at the time of purchase to be cash equivalents.
Accounts receivable and allowance for doubtful accounts
The Company’s accounts receivable consist primarily of trade receivables from customers and are generally unsecured and due within 30 days. The carrying value of accounts receivable approximates their fair value due to their short term nature. The Company evaluates the collectability of its accounts receivable based on a combination of factors and calculates an allowance for doubtful accounts based on the estimated proportion of aged receivables deemed uncollectable. Expected credit losses related to trade receivables are recorded as an allowance for doubtful accounts. The allowance for doubtful accounts is charged to sales and marketing expense and accounts receivable are written off as uncollectible and deducted from the allowance after appropriate collection efforts have been exhausted. The activities in the allowance for doubtful accounts are as follows:
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Balance at the beginning of the year | $ | 589 | $ | 424 | ||||
| Charges to bad debt expense | 217 | 271 | ||||||
| Write-off and recoveries | (157 | ) | (106 | ) | ||||
| Balance at the end of the year | $ | 649 | $ | 589 | ||||
| -45- |
Inventory
Inventory is recorded at the lower of cost (based on first in, first out basis) or market and consists of purchased finished goods. Inventory is periodically reviewed for evidence of slow-moving or obsolete items, and the estimated reserve is based on management’s reviews of inventories on hand, compared to estimated future usage and sales, reviewing product shelf-life, and assumptions about the likelihood of obsolescence. Once written down, the adjustments are considered permanent and are not reversed until the related inventory is sold.
Fixed assets
Property and equipment are carried at cost less accumulated depreciation and amortization. Depreciation is computed using the straight-line method over the estimated useful lives of the assets, ranging from three to seven years. Maintenance and repairs are expensed as incurred. The Company reviews property and equipment for impairment whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. An impairment loss would be recognized when estimated future undiscounted cash flows relating to the asset are less than its carrying amount. An impairment loss is measured as the amount by which the carrying amount of an asset exceeds its fair value.
Impairment of long-lived assets
The Company periodically assesses the carrying value of intangible and other long-lived assets, and whenever events or changes in circumstances indicate that the carrying amount of an asset might not be recoverable. The assets are considered to be impaired if the Company determines that the carrying value may not be recoverable based upon its assessment, which includes consideration of the following events or changes in circumstances:
| ● | the asset’s ability to continue to generate income from operations and positive cash flow in future periods; | |
| ● | loss of legal ownership or title to the asset; | |
| ● | significant changes in the Company’s strategic business objectives and utilization of the asset(s); and | |
| ● | the impact of significant negative industry or economic trends. |
If the assets are considered to be impaired, the impairment recognized is the amount by which the carrying value of the assets exceeds the fair value of the assets. Fair value is determined by the application of discounted cash flow models to project cash flows from the asset. In addition, the Company bases the useful lives and related amortization or depreciation expense on an estimate of the period that the assets will generate revenue or otherwise be used. The Company also periodically reviews the lives assigned to long-lived assets to ensure that the initial estimates do not exceed any revised estimated periods from which the Company expects to realize cash flows from its assets.
Patents and trademarks
Patents and trademarks are recorded at historical cost and are amortized using the straight-line method over their estimated useful lives, not to exceed 15 years.
| -46- |
Intangible Assets
Intangible assets are recorded at historical cost and are amortized using the straight-line method over their estimated useful life.
Product Warranties
The Company generally provides a one year warranty on its TearLab® Osmolarity System and related disposables. The Company accrues the estimated cost of this warranty at the time revenue is recognized and charges warranty expense to cost of goods sold. Warranty reserves are established based on historical experience with failure rates and the number of systems covered by warranty. Warranty reserves are depleted as systems and disposables are replaced. The Company reviews warranty reserves quarterly and, if necessary, make adjustments. The activities in the warranty reserve are as follows:
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Balance at the beginning of the year | $ | 94 | $ | 121 | ||||
| Charges to cost of goods sold | 141 | 73 | ||||||
| Costs applied to liability | (111 | ) | (100 | ) | ||||
| Balance at the end of the year | $ | 124 | $ | 94 | ||||
Income Taxes
A deferred tax asset or liability is determined based on the difference between the financial statement and tax basis of assets and liabilities as measured by the enacted tax rates which will be in effect when these differences reverse. The Company provides a valuation allowance against net deferred tax assets unless, based upon the available evidence, it is more likely than not that the deferred tax assets will be realized.
Revenue recognition
Revenue is recognized when all four of the following criteria are met: (i) persuasive evidence that an arrangement exists; (ii) delivery of the products has occurred; (iii) the selling price is fixed or determinable; and (iv) collectability is reasonably assured. The Company records revenue when all of its obligations are completed, which is generally upon shipment of the Company’s products. Amounts received in excess of revenue recognizable are deferred.
The Company sells its proprietary TearLab® Osmolarity System and related test cards to external customers, who are primarily eye care professionals, for use in osmolarity testing procedures. Revenue is primarily derived from the sale of disposable test cards. Products are generally shipped from a distribution and warehousing facility located in San Diego, California. The Company’s sales are currently direct to customers in the United States and distributors in the rest of the world.
The Company enters into contracts where revenue is derived either from agreements whereby the customer is provided the right to use the TearLab® Osmolarity System (reader equipment) at no separate cost to the customer in consideration for a minimum or implied purchase commitment of disposable test cards over the related contract term (referred to as either “Use Agreements”, “Masters Agreements” or “Flex Agreements”), or from agreements with sales of multiple deliverables, such as the reader equipment and disposable test cards (referred to as “Purchase Agreements”).
| -47- |
Purchase commitments for Use Agreements and Flex Agreements are expressed in the agreement for a specified period of time (generally one to three years). The purchase commitment for Masters Agreements is implied for large physician practices with an expectation of purchasing certain levels of test cards. The Company recovers the cost of providing the reader equipment in the amount charged for disposable test cards. These agreements are treated as operating leases as collectability of the minimum lease payments is not reasonably predictable at the outset of the arrangement. Accordingly, revenue is recognized over the defined contract term as disposable test cards are shipped. Revenue under such agreements is allocated between the lease of the reader equipment and the sale of the disposables based upon each component’s relative fair value. When reader equipment is placed with a customer at no separate cost, the Company retains title to the equipment and it remains capitalized on the Company’s Consolidated Balance Sheet as equipment classified within fixed assets, net. The equipment is depreciated on a straight-line basis once shipped to a customer location over its estimated useful life and depreciation expense is included in cost of goods sold within the Consolidated Statements of Operations and Comprehensive Loss.
Revenue recognition for Purchase Agreements with multiple deliverables is based on the individual units of accounting determined to exist in the contract. A delivered item is considered a separate unit of accounting when the delivered item has value to the customer on a stand-alone basis. Items are considered to have stand-alone value when they are sold separately by any vendor or when the customer could resell the item on a stand-alone basis. Considering that test cards are essential to the operation of a TearLab reader, there is no alternative vendor for the test cards and no indication that a secondary market for the TearLab readers is established, the deliverables under the contracts do not meet criteria for separation under the multiple-element arrangements guidance. Consideration is allocated at the inception of the contract to all deliverables based on their relative selling price as determined by the selling price of similar individual items on a stand-alone basis. The Company recognizes revenue for each of the elements only when it determines that all applicable recognition criteria have been met.
Amounts billed to customers for shipping and handling of a sales transaction are included as revenue. For the years ended December 31, 2016 and 2015, the Company recognized revenue from shipping and handling of $170 and $194, respectively.
Although the Company has a no return policy for its products, the Company has established a return reserve for product sales that contain an implicit right of return. The Company reserves for estimated returns or refunds by reducing revenue at the time of shipment based on historical experience. The reserve of $13 and $67 as of December 31, 2016 and 2015, respectively, has been recorded as a reduction of revenue and is included in accounts receivable. The activities in the return reserve are as follows:
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Balance at the beginning of the year | $ | 67 | $ | 77 | ||||
| Provision | 71 | 265 | ||||||
| Write-off and recoveries | (125 | ) | (275 | ) | ||||
| Balance at the end of the year | $ | 13 | $ | 67 | ||||
Cost of goods sold
Cost of goods sold includes the costs the Company incurs for the purchase of the TearLab® Systems sold and related freight and shipping costs, fees related to merchant services, warehousing and logistics inventory management associated with conducting business and depreciation of reader equipment. The Company recorded $1,422 and $1,489 in shipping and handling fees for the years ended December 31, 2016 and 2015, respectively.
Clinical, regulatory and research & development costs
Clinical and regulatory costs attributable to the performance of contract services are recognized as an expense as the services are performed. Non-refundable, up-front fees paid in connection with these contracted services are deferred and recognized as an expense over the estimated term of the related contract.
| -48- |
Stock-based compensation
The Company accounts for stock-based compensation expense for its directors and employees in accordance with US GAAP guidance related to stock-based compensation. Under this guidance, stock-based compensation cost is estimated at the grant date based on the fair value of the award and is recognized as an expense ratably over the requisite service period of the award. The Company uses the Black-Scholes Merton option pricing model for determining the fair value for all its awards and will recognize compensation cost on a straight-line basis over the awards’ vesting periods.
Advertising Costs
Advertising costs are expensed as incurred. Total advertising costs for each of the years ended December 31, 2016 and 2015 were $378 and $442, respectively.
Warrant liabilities
The Company issued several rounds of warrants related to various debt and equity transactions which occurred in 2011. The Company accounts for its warrants issued in accordance with the US GAAP accounting guidance under Accounting Standards Codification (ASC) 815 applicable to derivative instruments, which requires every derivative instrument within its scope to be recorded on the balance sheet as either an asset or liability measured at its fair value, with changes in fair value recognized in earnings. Based on this guidance, the Company determined that the Company’s warrants do not meet the criteria for classification as equity. Accordingly, the Company classified the warrants as current liabilities. The warrants are subject to remeasurement at each balance sheet date with any change in fair value recognized as a component of other income (expense), in the statements of operations and comprehensive loss. Warrants are also remeasured at fair value immediately prior to being exercised, and the resulting fair value is reclassified into additional paid-in capital, net of any applicable exercise proceeds. The Company estimated the fair value of these warrants at the respective balance sheet dates using the Black-Scholes Merton option pricing model based on the estimated market value of the underlying common stock at the valuation measurement date, the remaining contractual term of the warrant, risk-free interest rates and expected dividends on and expected volatility of the price of the underlying common stock. There is subjectivity involved when using option pricing models to estimate the warrant liability and the assumptions used in the Black-Scholes Merton option pricing model are judgmental. The warrants classified as liabilities were either exercised or expired and no liability remains at December 31, 2016.
Foreign currency transactions
The Company’s functional and reporting currency is the U.S. dollar. The assets and liabilities of the Company’s Canadian operations are maintained in U.S. dollars. Monetary assets and liabilities denominated in foreign currencies are translated into U.S. dollars at exchange rates in effect at the consolidated balance sheet dates, and non-monetary assets and liabilities are translated at exchange rates in effect on the date of the transaction. Revenue and expenses are translated into U.S. dollars at average exchange rates prevailing during the year. Resulting exchange gains of $54 and $76 are included in other income (expense) for the years ended December 31, 2016 and 2015, respectively.
| -49- |
Geographic information
The following table provides geographic information related to the Company’s revenue based on the geographic location to which it delivers the product:
| For the year ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| United States | $ | 26,391 | $ | 23,597 | ||||
| Rest of the world | 1,623 | 1,559 | ||||||
| Total | $ | 28,014 | $ | 25,156 | ||||
Comprehensive Income (Loss)
Comprehensive income (loss) is defined as the change in equity during a period from transactions and other events and circumstances from non-owner sources. Comprehensive income (loss) generally includes unrealized gains or losses on the Company’s marketable securities and foreign currency translation adjustments. In all the periods presented, the Company’s comprehensive loss equaled the net loss for the period.
Recent accounting pronouncements
In August 2016, the Financial Accountings Standards Board (the “FASB”) issued Accounting Standards Update No. 2016-15, Statement of Cash Flows (“ASU 2016-35”), to reduce diversity in practice of how certain transactions are classified in the statement of cash flows. ASU 2016-15 is effective for fiscal and interim periods beginning after December 15, 2017. The Company is currently evaluating the impact the adoption of the new standard will have on its financial statements.
In March 2016, the FASB issued Accounting Standards Update No. 2016-09, Improvement to Employee Share-Based Payment Accounting (“ASU 2016-09”), which involves several aspects of accounting for share-based payment transactions, including income tax consequences, classification of awards as liabilities or equity, and classifications on the statement of cash flows. ASU 2016-09 is effective for fiscal and interim periods beginning after December 15, 2016. The Company does not expect the guidance to have a material impact on the Company.
In February 2016, the FASB issued Accounting Standards Update No. 2016-02, Leases (“ASU 2016-02”). ASU 2016-02 establishes a right-of-use model that requires a lessee to record an asset and liability on the balance sheet for all leases with terms longer than twelve months. ASU 2016-02 is effective for fiscal years and interim periods beginning after December 15, 2018. A modified retrospective transition approach is required for lessees for capital and operating leases existing at, or entered into after, the beginning of the earliest comparative period presented in the financial statements. The Company is currently evaluating the impact of the new standard on its financial statements.
In May 2014, the FASB issued Accounting Standards Update No. 2014-09, Revenue from Contracts with Customers (“ASU 2014-09”), which requires an entity to recognize the amount of revenue to which it expects to be entitled for the transfer of promised goods or services to customers. Early application is not permitted. On April 1, 2015, the FASB voted to propose a deferral of the effective date of the standard by one year which would result in the new standard being effective for the Company at the beginning of its first quarter of fiscal year 2018. During 2017, the Company will assess the impact of the new standard, including possible transition alternatives, on the Company’s financial statements.
| -50- |
3. BALANCE SHEET DETAILS
Accounts receivable
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Trade receivables | $ | 2,928 | $ | 3,610 | ||||
| Allowance for doubtful accounts | (649 | ) | (589 | ) | ||||
| $ | 2,279 | $ | 3,021 | |||||
Inventory
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Finished goods | $ | 3,210 | $ | 4,002 | ||||
| Inventory reserves | (17 | ) | (30 | ) | ||||
| $ | 3,193 | $ | 3,972 | |||||
The Company evaluates inventory for estimated excess quantities and obsolescence, based on expected future sales levels and projections of future demand and expiration dates of inventory, with the excess inventory provided for. In addition, the Company assesses the impact of changing technology and market conditions.
Prepaid expenses and other current assets
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Prepaid trade shows | $ | 217 | $ | 246 | ||||
| Prepaid insurance | 326 | 87 | ||||||
| Manufacturing deposits | 282 | 154 | ||||||
| Subscriptions | 297 | 128 | ||||||
| Other fees and services | 66 | 146 | ||||||
| Other current assets | 38 | 29 | ||||||
| $ | 1,226 | $ | 790 | |||||
Fixed assets, net
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Capitalized TearLab equipment | $ | 9,095 | 8,349 | |||||
| Leasehold improvements | 60 | 61 | ||||||
| Computer equipment and software | 932 | 1,023 | ||||||
| Furniture and office equipment | 271 | 278 | ||||||
| Medical equipment | 500 | 431 | ||||||
| $ | 10,858 | $ | 10,142 | |||||
| Less accumulated depreciation | (6,680 | ) | (4,790 | ) | ||||
| $ | 4,178 | $ | 5,352 | |||||
| -51- |
Depreciation expense was $2,364 and $2,075 for the years ended December 31, 2016 and 2015, respectively. During the year ended December 31, 2015, the Company determined the assets of OcuHub were impaired and recorded a charge of $343 to fixed assets, which is included with the impairment of the OcuHub intangible assets and presented as an impairment of long-lived assets on the Consolidated Statements of Operations and Comprehensive Loss.
Accrued liabilities
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Due to professionals | $ | 81 | $ | 256 | ||||
| Due to employees and directors | 2,200 | 2,130 | ||||||
| Sales and use tax liabilities | 247 | 231 | ||||||
| Royalty liability | 854 | 753 | ||||||
| Warranty | 124 | 94 | ||||||
| Other | 452 | 1,583 | ||||||
| $ | 3,958 | $ | 5,047 | |||||
4. INTANGIBLE ASSETS
The Company’s intangible assets consist of the value of TearLab® Technology acquired in the acquisition of TearLab Research, Inc., a wholly-owned subsidiary of the Company, the value of the OcuHub platform technology acquired in the acquisition of the OcuHub business unit in March 2014 and a prescriber list. The TearLab Technology, which consists of a disposable lab card and card reader, supported by an array of patents and patent applications that are either held or in-licensed by the Company, was fully amortized in November 2016. Amortization expense was $1,139 and $1,540 for the years ended December 31, 2016 and 2015, respectively.
During the year ended December 31, 2015 the Company determined the OcuHub platform technology was no longer expected to generate any future cash flows for the Company. Accordingly the Company recorded an impairment to the OcuHub platform technology of $1,029 in 2015. The impairment is included with the impairment of the OcuHub fixed assets and presented in the Consolidated Statements of Operations and Comprehensive Loss as an impairment of long-lived assets.
Intangible assets subject to amortization consist of the following:
| Remaining | Gross | Net Book | ||||||||||||||
| Useful Life | Value at | Accumulated | Value at | |||||||||||||
| (Years) | December 31, 2016 | Amortization | December 31, 2016 | |||||||||||||
| TearLab® technology | 0 | $ | 12,172 | $ | (12,172 | ) | $ | - | ||||||||
| Patents and trademarks | 2 | 271 | (245 | ) | 26 | |||||||||||
| Prescriber list | 1 | 90 | (56 | ) | 34 | |||||||||||
| Total | $ | 12,533 | $ | (12,473 | ) | $ | 60 | |||||||||
| -52- |
| Remaining | Gross | Net Book | ||||||||||||||
| Useful Life | Value at | Accumulated | Value at | |||||||||||||
| (Years) | December 31, 2015 | Amortization | December 31, 2015 | |||||||||||||
| TearLab® technology | 1 | $ | 12,172 | $ | (11,106 | ) | $ | 1,066 | ||||||||
| Patents and trademarks | 3 | 268 | (216 | ) | 52 | |||||||||||
| Prescriber list | 2 | 90 | (11 | ) | 79 | |||||||||||
| Total | $ | 12,530 | $ | (11,333 | ) | $ | 1,197 | |||||||||
Estimated future amortization expense related to intangible assets with finite lives at December 31, 2016 is as follows:
| Amortization | ||||
| of intangible | ||||
| assets | ||||
| 2017 | $ | 58 | ||
| 2018 | 2 | |||
| $ | 60 | |||
5. TERM LOAN
On March 4, 2015, the Company executed a term loan agreement (the “Term Loan Agreement”) with CRG LP and certain of its affiliate funds (“CRG”) as lenders providing the Company with access of up to $35,000 under the arrangement. The Company received $15,000 in gross proceeds under the arrangement on March 4, 2015, and an additional $10,000 on October 6, 2015. The Term Loan Agreement matures on March 31, 2021 and bears interest at 13% per annum, with quarterly payments of interest only for the first four years. While interest on the loan is accrued at 13% per annum, the Company may elect to make interest-only payments at 8.5% per annum. The unpaid interest of 4.5% is added to the principal of the loan and is subject to additional accrued interest (“PIK interest”). The accrued interest can be deferred and paid together with the principal in the fifth and sixth years.
As part of the amended Term Loan Agreement, and funding of the second tranche, CRG received 35,000 warrants dated as of October 6, 2015 to purchase common shares of the Company at a price of $50.00 per share (the “CRG Warrants”). The CRG Warrants have a five-year life. The CRG Warrants are classified as equity on the Consolidated Balance Sheets as of December 31, 2016 and 2015. The CRG Warrants were valued at their issuance date using the Black-Scholes Merton model. The related reduction of the long-term debt will be amortized over the life of the debt.
The loan is collateralized by all assets of the Company. Additionally, the terms of the Term Loan Agreement contain various affirmative and negative covenants agreed to by the Company. Among them, the Company must attain minimum certain annual revenue and minimum cash threshold levels. On April 7, 2016, the Company amended the Term Loan Agreement to change the required minimum revenue levels. The amended minimum revenue is $27,000 for 2016, $31,000 for 2017, $36,000 for 2018, $45,000 for 2019 and $55,000 for 2020. The minimum cash balance required is $5,000 subject to certain conditions. The amendment also reduced the exercise price of the CRG Warrants from $50.00 per share to $15.00 per share and the Company issued CRG additional warrants to purchase 35,000 common shares of the Company’s stock at $15.00 per share, which expire 5 years after issuance.
| -53- |
If the Company does not have annual revenue greater or equal to the annual revenue covenant in a calendar year, the Company will have the right within 90 days of the end of the respective calendar year to raise subordinated debt or equity (the “CRG Equity Cure”) equal to twice the difference between the annual revenue and the revenue covenant, with the total proceeds from this financing to be used to reduce the principal of the Term Loan Agreement. In the event of a default, the Company may be required to repay any outstanding amounts earlier than anticipated, and CRG may foreclose on their security interest in the Company’s assets.
At December 31, 2016, the Company was in compliance with all of the covenants.
As part of the amended Term Loan Agreement, on the earlier of the maturity date or the date the term loan is paid off in full, the Company will pay a facility fee equal to 6.5% of the aggregate principal amount of the loan, excluding accrued PIK interest (the “Facility Fee”). The Facility Fee is being accrued to interest expense using the effective interest method.
The Company incurred financing and legal fees associated with the debt of $606, which were recorded as a direct discount to the debt and are being amortized using the effective interest method. The Company presents the debt issuance costs related to the recognized debt liability on the Consolidated Balance Sheet as a reduction of the liability.
The Term Loan Agreement provided for prepayment fees of 5% of the outstanding balance of the loan if the loan was repaid prior to March 31, 2016. The prepayment fee is reduced 1% per year for each subsequent year until maturity.
The following is a summary of the Term Loan Agreement as of December 31, 2016 and related maturities of outstanding principle:
| Principle balance outstanding | $ | 25,000 | ||
| PIK interest | 1,877 | |||
| Facility fee | 321 | |||
| less discount on term loan: | ||||
| deferred financing fees, net | (432 | ) | ||
| fair value of detachable warrants, net | (317 | ) | ||
| Total term loan | $ | 26,449 |
Principal due for each of the next 5 years and in the aggregate thereafter:
| 2017 | - | |||
| 2018 | - | |||
| 2019 | 10,079 | |||
| 2020 | 13,439 | |||
| 2021 | 3,680 | |||
| Total principal, PIK interest and facility fee due | 27,198 | |||
| Less: discount on term loan | (749 | ) | ||
| Total term loan | $ | 26,449 |
6. STOCKHOLDERS’ EQUITY
(a) Authorized share capital
On February 23, 2017, the Company’s stockholders authorized the board of directors to implement a reverse stock split, along with a corresponding reduction in the number of shares authorized. On February 27, 2017, the Company effected a 1-for-10 reverse stock split of its common stock. All common stock share amounts and prices per share of common stock have been retroactively adjusted to reflect the reverse stock split.
| -54- |
On June 24, 2016, the Company’s stockholders approved an amendment to the Company’s certificate of incorporation to increase the total number of authorized shares of common stock of the Company to 9,500,000 from 6,500,000. Each share of common stock has a par value of $0.001 per share. The total number of authorized shares of preferred stock of the Company is 10,000,000. Each share of preferred stock has a par value of $0.001 per share.
(b) Common and preferred shares
On May 9, 2016 the Company issued 1,861,090 shares of common stock, 3,291.8 shares of Series A Convertible Preferred Stock (“Preferred Stock”) and Series A warrants to purchase 1,150,000 shares of common stock (“Series A Warrants”) for gross proceeds of $17,250, less issuance costs of $1,793. Additionally, the Company granted the placement agent warrants to purchase 103,500 shares of common stock with an exercise price of $11.25 per share. The Preferred Stock is convertible, subject to certain limitations, into an aggregate of 438,910 shares of common stock, contains no voting rights, participates in any common stock dividends, and is treated as if converted upon any ordinary liquidation event. The common stock, the Series A Convertible Preferred Stock, and the Series A Warrants are all included in equity in the Company’s Consolidated Balance Sheets as of December 31, 2016. The net proceeds were allocated to common stock, Preferred Stock, and Series A Warrants based on their relative fair values, as follows:
| Common stock | $ | 9,632 | ||
| Preferred stock | 2,275 | |||
| Series A warrants | 3,550 | |||
| Net proceeds | $ | 15,457 |
On August 2, 2016, 527.5 shares of Series A Convertible Preferred stock were converted into 70,333 shares of common stock.
(c) Stock incentive plan
The Company has a stock incentive plan, the 2002 Stock Incentive Plan (the “Stock Incentive Plan”), under which up to 720,000 options are available for grant to employees, directors and consultants. Options granted under the Stock Incentive Plan may be either incentive stock options or non-statutory stock options. Under the terms of the Stock Incentive Plan, the exercise price per share for an incentive stock option shall not be less than the fair market value of a share of stock on the effective date of grant and the exercise price per share for non-statutory stock options shall not be less than 85% of the fair market value of a share of stock on the date of grant. No option granted to a holder of more than 10% of the Company’s common stock shall have an exercise price per share less than 110% of the fair market value of a share of stock on the effective date of grant.
Options granted are typically service-based options. Generally, options expire 10 years after the date of grant. No incentive stock options granted to a 10% owner optionee shall be exercisable after the expiration of five years after the effective date of grant of such option, no option has been granted to a prospective employee, prospective consultant or prospective director prior to the date on which such person commences service, and with the exception of an option granted to an officer, director or consultant, no incentive option shall become exercisable at a rate less than 20% per annum over a period of five years from the effective date of grant of such option unless otherwise approved by the Board.
The Company accounts for stock-based compensation under the authoritative guidance which requires that share-based payment transactions with employees be recognized in the financial statements based on their fair value and recognized as compensation expense over the vesting period. The amount of expense recognized during the period is affected by subjective assumptions, including: estimates of the Company’s future volatility, the expected term for its stock options, option exercise behavior, the number of options expected to ultimately vest, and the timing of vesting for the Company’s share-based awards. The weighted-average fair value of stock options granted during the years ended December 31, 2016 and 2015 was $4.30 and $13.39, respectively.
| -55- |
The following table sets forth the total stock-based compensation expense resulting from stock options and the employee stock purchase plan included in the Company’s Consolidated Statements of Operations and Comprehensive Loss (in thousands):
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| General and administrative | $ | 1,359 | $ | 2,013 | ||||
| Clinical, regulatory and research and development | 327 | 471 | ||||||
| Sales and marketing | 761 | 1,633 | ||||||
| Stock-based compensation expense before income taxes | $ | 2,447 | $ | 4,117 | ||||
The estimated fair value of stock options for the periods presented was determined using the Black-Scholes Merton option pricing model with the following weighted-average assumptions:
| Years ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Volatility | 76 | % | 89 | % | ||||
| Weighted average expected life of the options | 6 | 5.39 | ||||||
| Risk-free interest rate | 1.23 | % | 1.60 | % | ||||
| Dividend yield | 0.00 | % | 0.00 | % | ||||
The Company’s computation of expected volatility is based on the historical volatility of the Company’s common stock over a period of time equal to the expected term of the stock options. Due to the lack of sufficient historical data, the Company’s computation of weighted average expected life was estimated as the mid-point between the vesting date and the end of the contractual period. The risk-free interest rate for an award is based on the U.S. Treasury yield curve with a term equal to the expected life of the award on the date of grant.
A summary of the options issued during the year ended December 31, 2016 and the total number of options outstanding as of that date are set forth below:
| Weighted | ||||||||||||||||
| Average | ||||||||||||||||
| Number of | Weighted | Remaining | Aggregate | |||||||||||||
| Options | Average | Contractual | Intrinsic Value | |||||||||||||
| Outstanding | Exercise Price | Life (years) | (in thousands) | |||||||||||||
| Outstanding, December 31, 2014 | 636,313 | 47.46 | 6.57 | 2,155 | ||||||||||||
| Granted | 128,546 | 20.91 | ||||||||||||||
| Exercised | (6,539 | ) | 15.19 | |||||||||||||
| Forfeited/cancelled/expired | (66,409 | ) | 59.80 | |||||||||||||
| Outstanding, December 31, 2015 | 691,911 | $ | 41.66 | 6.08 | $ | 184 | ||||||||||
| Granted | 156,637 | 6.90 | ||||||||||||||
| Exercised | - | - | ||||||||||||||
| Forfeited/cancelled/expired | (134,932 | ) | 45.95 | |||||||||||||
| Outstanding, December 31, 2016 | 713,616 | $ | 33.19 | 5.92 | $ | - | ||||||||||
| Vested or expected to vest, December 31, 2016 | 705,094 | $ | 33.28 | 5.90 | $ | 0 | ||||||||||
| Exercisable, December 31, 2016 | 495,321 | $ | 41.51 | 4.61 | $ | 0 | ||||||||||
| -56- |
The aggregate intrinsic value at December 31, 2016 represents the total pre-tax intrinsic value, calculated as the difference between the Company’s closing stock price on the last trading day of the respective fiscal year and the exercise price, multiplied by the number of shares that would have been received by the option holders if the options that could be exercised had been exercised on such date.
Net cash proceeds from the exercise of common stock options were $0 and $99 for the years ended December 31, 2016 and 2015, respectively. No income tax benefit was realized from stock option exercises during the years ended December 31, 2016 and 2015. The Company presents excess tax benefits from the exercise of stock options, if any, as financing cash flows rather than operating cash flows.
The total intrinsic value of options exercised was $0 and $76 for the years ended December 31, 2016 and 2015, respectively. The total fair value of stock options vested during the years ended December 31, 2016 and 2015 was $2,619 and $3,981, respectively.
As of December 31, 2016, total unrecognized compensation cost related to stock options of $1,321 is expected to be recognized over a weighted-average period of 1.23 years.
As of December 31, 2016, the Company had 17,157 options remaining in the Stock Option Plan available for grant.
(d) Employee Stock Purchase Plan
In July 2014, the Company’s Board of Directors adopted the 2014 Employee Stock Purchase Plan (the “ESPP”) which was approved by the Company’s stockholders in June 2014 at the Company’s Annual Meeting of Stockholders. A total of 67,150 shares of the Company’s common stock are reserved for issuance under the plan, which permits eligible employees to purchase common stock at a discount through payroll deductions.
The price at which stock is purchased under the ESPP is equal to 90% of the fair market value of the common stock on the first or the last day of the offering period, whichever is lower. Generally, each offering under the ESPP will be for a period of six months as determined by the Company’s Board of Directors. Employees may invest up to 20% of their gross compensation through payroll deductions. In no event may an employee purchase more than $25 worth of stock in the plan during each calendar year or purchase more than 500 shares per offering period. During the year ended December 31, 2016 and 2015, the Company recorded $11 and $20 of expense, respectively, under the ESPP. During the year ended December 31, 2016 and 2015 the Company issued 14,105 and 5,421 shares of common stock, respectively, under the ESPP. In January 2017 the Company issued an additional 7,512 shares of Common stock under the ESPP.
(e) Warrants
On June 30, 2011, the Company closed a private placement financing in which 384,615 shares of common stock and warrants (‘‘2011 Warrants’’) to purchase 384,615 shares of common stock for gross proceeds of approximately $7,000. The exercise price of the warrants was $18.60 per share. The 2011 Warrants expired on June 30, 2016. There were 21,960 of the 2011 Warrants that expired unexercised. Prior to their expiration, the 2011 Warrants were recorded as a liability on the Company’s Consolidated Balance Sheets and remeasured each period using the Black-Scholes Merton option-pricing model. There were no exercises of 2011 Warrants during the twelve months ended December 31, 2016 or 2015. The liability for the 2011 Warrants outstanding as of December 31, 2015 had a value of $29. The value of the 2011 Warrants outstanding as of December 31, 2015 was recorded as a Change in fair value of warrant obligations in the Consolidated Statements of Operations and Comprehensive Loss during the twelve months ended December 31, 2016, reflecting the expiration of the instruments and the associated liability.
| -57- |
On October 8, 2015, as part of the second amendment to the Term Loan Agreement and funding of the $10,000 tranche, CRG received warrants to purchase 35,000 common shares in the Company at a price of $50.00 per share (the “CRG Warrants”). The CRG Warrants are exercisable any time prior to October 8, 2020. The CRG Warrants are classified as equity on the Consolidated Balance Sheets as of December 31, 2016 and 2015, respectively. The CRG Warrants were valued at $290 upon issuance using the Black-Scholes Merton model assuming volatility of 73%, an expected life of 5.0 years, a risk-free interest rate of 1.71%, and 0% dividend yield. No CRG Warrants were exercised during the twelve months ended December 31, 2016 or 2015.
On April 8, 2016, the Company further amended its Term Loan Agreement. As part of the amendment, the exercise price of the CRG Warrants was changed to allow the holder to purchase 35,000 common shares in the Company at a price of $15.00 per share and CRG was issued an additional 35,000 warrants to purchase common shares at an exercise price of $15.00 (the “2016 CRG Warrants”). The modification to the terms of the CRG Warrants resulted in a change in fair value of $54 which was included as interest expense for the twelve months ended December 31, 2016. The change in fair value was calculated using the Black-Scholes Merton model with both exercise prices, assuming volatility of 76%, an expected life of 4.5 years, a risk-free interest rate of 1.06%, and 0% dividend yield. The 2016 CRG Warrants were valued at $106 upon issuance using the Black-Scholes Merton model assuming volatility of 76%, an expected life of 5.0 years, a risk-free interest rate of 1.30% and 0% dividend yield.
On May 9, 2016, the Company issued Series A Warrants to purchase 1,253,500 shares of common stock for $11.25 per common share attached to shares of common and Series A Convertible Preferred Stock issued on the same date. The Series A Warrants can be exercised after May 9, 2017 (the “Initial Exercise Date”) and expire 5 years after the Initial Exercise Date. Fair value of the Series A Warrants, for purposes of allocating the net proceeds of the equity offering, was determined using the Black-Scholes Merton model assuming volatility of 76%, an expected life of 6.0 years, a risk-free interest rate of 1.30%, and 0% dividend yield.
The following table provides activity for warrants issued and outstanding during the two years ended December 31, 2016:
| Number of | Weighted average | |||||||
| warrants | exercise | |||||||
| outstanding | price | |||||||
| Outstanding, December 31, 2014 | 29,367 | $ | 17.90 | |||||
| Issued | 35,000 | 50.00 | ||||||
| Exercised | - | - | ||||||
| Expired | - | - | ||||||
| Outstanding, December 31, 2015 | 64,367 | $ | 35.35 | |||||
| Issued | 1,288,500 | 11.35 | ||||||
| Exercised | - | - | ||||||
| Expired | (29,367 | ) | 17.90 | |||||
| Outstanding, December 31, 2016 | 1,323,500 | $ | 10.65 | |||||
(f) Exchange Right
In August 2014, the Company sold membership units in OcuHub LLC, a Delaware limited liability company, which was a wholly owned subsidiary of TearLab Corporation at the time, in exchange for 2% ownership of OcuHub LLC. In connection with the sale of the membership units, the new members received an exchange right allowing the units to be exchanged upon written notice and during a specified exchange window for shares in the Company’s common stock. On March 31, 2016, the members exchanged the ownership interest in OcuHub LLC for 38,580 shares of the Company’s common stock.
| -58- |
7. INCOME TAXES
Geographic sources of loss from continuing operations before income tax are as follows:
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Domestic | $ | 19,242 | $ | 32,099 | ||||
| Foreign | 678 | 1,130 | ||||||
| Loss before income taxes | $ | 19,920 | $ | 33,229 | ||||
Significant components of the Company’s deferred tax assets and liabilities are as follows:
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Deferred tax assets | ||||||||
| Intangible assets | $ | 272 | $ | 1,307 | ||||
| Stock options | 4,135 | 3,615 | ||||||
| Accruals and others | 1,359 | 1,060 | ||||||
| Net operating loss carryforwards | 37,215 | 30,200 | ||||||
| 42,981 | 36,182 | |||||||
| Valuation allowance | (43,138 | ) | (36,182 | ) | ||||
| Deferred tax asset | $ | (157 | ) | $ | - | |||
| Deferred tax liability | ||||||||
| Fixed Assets | $ | 157 | $ | - | ||||
| Deferred tax liability | $ | 157 | $ | - | ||||
| Deferred taxes, net | $ | - | $ | - | ||||
The following is a reconciliation of the recovery of income taxes between those that are expected, based on enacted tax rates and laws, to those currently reported:
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Loss for the year before income taxes | $ | (19,920 | ) | $ | (33,229 | ) | ||
| Expected recovery of income taxes | $ | (6,773 | ) | $ | (11,298 | ) | ||
| State income tax, net of federal benefit | (416 | ) | (518 | ) | ||||
| Stock based compensation | 99 | 239 | ||||||
| Warrants | (10 | ) | (75 | ) | ||||
| Deferred state tax rate adjustment | - | (227 | ) | |||||
| Adjustments to deferred tax assets | 91 | 552 | ||||||
| Non-deductible expense and other | 53 | 118 | ||||||
| Change in valuation allowance | 6,956 | 11,209 | ||||||
| Total recovery of income taxes | $ | - | $ | - | ||||
| -59- |
Income taxes are recorded in accordance with authoritative guidance for accounting for income taxes, which requires the recognition of deferred tax assets and liabilities to reflect the future tax consequences of the events that have been recognized in the Company’s consolidated financial statements or tax returns. Measurement of the deferred items is based on enacted tax laws. In the event of differences between the financial reporting bases and tax bases of the Company’s assets, an assessment regarding the Company’s ability to realize the future benefits of the deferred tax assets is required. The Company records a valuation allowance to reduce the deferred tax assets to the amount that is more likely than not to be realized. Management has considered future taxable income and ongoing tax planning strategies in assessing the need for the valuation allowance. In the event the Company were to determine that it would be able to realize the deferred tax assets in the future in excess of their net recorded amounts, an adjustment to the deferred tax assets would increase the income in the period such determination was made. Likewise, should the Company determine that it would not be able to realize all or part of the net deferred tax assets in the future, an adjustment to the deferred tax assets would be charged to income in the period such determination was made.
In July 2006, the FASB issued additional guidance which requires the impact of an uncertain income tax position on the income tax return must be recognized at the largest amount that is more-likely-than-not to be sustained upon audit by the relevant taxing authority. An uncertain income tax position will not be recognized if it has less than a 50% likelihood of being sustained. Additionally, the provisions under this guidance also provides guidance on derecognition, measurement, classification, interest and penalties, accounting in interim periods, disclosure and transition. The Company adopted the provisions under this guidance on January 1, 2007. The adoption of these provisions had no impact on the Company’s consolidated financial position or results of operations. At December 31, 2016 and 2015, the Company has no unrecognized income tax benefits and no material uncertain tax positions.
During the year ended December 31, 2012, a change of ownership for tax purposes causing Section 382 restrictions on the utilization of net operating losses occurred. No additional ownership changes are expected to arise through 2016. The ownership change in 2012, while limiting the annual utilization of net operating losses, will not cause the carryforwards generated subsequent to the Company’s last ownership change in October 2008 to expire unused. In general, an ownership change, as defined by Section 382, results from transactions increasing ownership of certain stockholders or public groups in the stock of the corporation by more than 50 percentage points over a three-year period. Utilization of net operating loss carryforwards are subject to annual limitations under Section 382 of the Internal Revenue Code of 1986, and similar state provisions whenever an ownership change has occurred. The ownership changes described above will limit the annual amount of net operating loss carryforwards that can be utilized to offset future taxable income.
At December 31, 2016, the Company had federal net operating loss carryforwards of approximately $100,280, state net operating loss carryforwards of approximately $64,473 and Canadian net operating loss carryforwards of approximately $8,683. The federal net operating loss carryforwards and the state net operating loss carryforwards begin to expire in 2028, and the Canadian net operating loss carryforwards begin to expire in 2026. The federal and state net operating loss carryovers reflected above do not include any net operating loss carryover which would expire unutilized solely as a result of Section 382 limitations arising in connection with the 2008 ownership change. As of December 31, 2016, the Company has approximately $1,515 of net operating loss carryforwards related to stock option exercises which will result in an increase to additional paid-in capital and a decrease in income taxes payable at the time when the tax loss carryforwards are utilized.
The Company’s policy is to recognize interest and penalties related to income tax matters in other expense. Because the Company has generated net operating losses since inception for both state and federal purposes, no additional tax liability, penalties or interest have been recognized for balance sheet or income statement purposes as of and for the two years ended December 31, 2016.
The Company does not expect a significant change in the amount of its unrecognized tax benefits within the next 12 months. Therefore, it is not expected that the change in the Company’s unrecognized tax benefits will have a significant impact on the Company’s results of operations or financial position.
All of the federal income tax returns for the Company and its subsidiaries remain open since their respective dates of incorporation due to the existence of net operating losses. The Company and its subsidiaries have not been, nor are they currently, under examination by the Internal Revenue Service or the Canada Revenue Agency.
| -60- |
State and provincial income tax returns are generally subject to examination for a period of between three and five years after their filing. However, due to the existence of net operating losses, all state income tax returns of the Company and its subsidiaries since their respective dates of incorporation are subject to re-assessment. The state impact of any federal changes remains subject to examination by various states for a period of up to one year after formal notification to the states. The Company and its subsidiaries have not been, nor are they currently, under examination by any state tax authority.
8. NET INCOME (LOSS) PER SHARE
Basic earnings per share (’‘EPS”) excludes dilutive securities and is computed by dividing net loss available to common stockholders by the weighted average number of shares of common stock outstanding for the year. Diluted EPS reflects the potential dilution that could occur if securities or other contracts to issue common stock were exercised or converted and the resulting additional shares are dilutive because their inclusion decreases the amount of EPS.
The following are potentially dilutive securities which have not been used in the calculation of diluted loss per share as they are anti-dilutive:
| Year Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| (in thousands of shares) | ||||||||
| Stock options | 714 | 692 | ||||||
| Warrants | 1,324 | 42 | ||||||
| ESPP shares | 8 | 7 | ||||||
| Convertible preferred shares | 369 | - | ||||||
| Exchange rights | - | 12 | ||||||
| Total | 2,415 | 753 | ||||||
The following table is a reconciliation of the weighted average shares outstanding used for basic and diluted loss per share:
| Year Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| (in thousands of shares) | ||||||||
| Weighted average shares outstanding - basic | 4,648 | 3,370 | ||||||
| Dilutive potential common shares | - | 3 | ||||||
| Weighted average shares outstanding - fully diluted | 4,648 | 3,373 | ||||||
9. EMPLOYEE RETIREMENT PLAN
The Company has a defined contribution, 401(k) retirement plan under which all full-time employees may contribute up to 90% of their annual salary, within IRS limits. The Company has not made any contributions to the retirement plan in the years ended December 31, 2016 and 2015.
10. COMMITMENTS AND CONTINGENCIES
Commitments
The Company has commitments relating to operating leases recognized on a straight line basis over the term of the lease for rental of office space and equipment from unrelated parties, expiring at various times through June 30, 2018. The total future minimum obligation under these various leases for 2017 and 2018 are $354, and $182, respectively. Rent expensed under these leases was $494 and $519 for the years ended December 31, 2016 and 2015 respectively. The Company leases office facilities under an operating lease agreement. The initial term of the lease is five years and includes periodic rent increases and a renewal option.
| -61- |
Effective October 1, 2006, the Company entered into a patent license and royalty agreement with the University of California San Diego to obtain a second exclusive license to make, use, sell, offer for sale, and import existing TearLab technology. The Company is required to make royalty payments of $35 or 5.5% of gross sales per year, whichever is higher. Additionally, the Company is required to pay a royalty of 30% of any sublicense fees it receives prior to receiving FDA approval and 25% of any sub-license fees it receives after FDA approval. The Company incurred fees of $1,398 and $1,370 under this agreement during the years ended December 31, 2016 and 2015, respectively. The Company had $790 and $734 in accrued royalties at December 31, 2016 and 2015, respectively. Future minimum royalty payments under this agreement as of December 31, 2016 are as follows:
| 2017 | $ | 35 | ||
| 2018 | 35 | |||
| 2019 | 35 | |||
| 2020 | 35 | |||
| 2021 | 35 | |||
| Thereafter | 210 | |||
| Total | $ | 385 |
On March 12, 2003, the Company entered into a patent license and royalty agreement with the University of California San Diego to obtain an exclusive license to make, use, sell, offer for sale, and import TearLab technology in development. Starting in 2009, the Company was required to make minimum royalty payments of $35 or 5.5% of gross sales per year, whichever is higher. However, if this new technology is combined with existing technology, the maximum royalty payable on the sale of the combined products would be 5.5% of gross sales per year. As the new technology is currently in development, there is no revenue and the minimum royalty payment of $35 is applicable. Future minimum royalty payments under this agreement as of December 31, 2016 are as follows:
| 2017 | $ | 35 | ||
| 2018 | 35 | |||
| 2019 | 35 | |||
| 2020 | 35 | |||
| 2021 | 35 | |||
| Thereafter | 210 | |||
| Total | $ | 385 |
On March 7, 2016, the Company, through its subsidiary, TearLab Research, Inc., entered into a supply and development agreement (“Supply Agreement”) with MiniFAB (Aust) Pty Ltd (“MiniFAB”). The agreement is an exclusive supply agreement through June 2021, which will provide 16% savings on the purchase and delivery of individual osmolarity test cards following a transition period to account for ending inventory at December 31, 2015 and other elements of the prior agreement. The savings consist of lower prices for the purchase of the test cards and for freight costs to ship the cards to the Company’s distribution facility. The lower purchase price will remain in place until the earlier of the date that the Company reaches an annual volume of 4.5 million test cards and March 31, 2018. The savings from freight costs will remain in place throughout the agreement. The Supply Agreement requires that, in any given 6 calendar months, the Company must place aggregate purchase orders equal to at least 50% of the orders forecasted for that 6 month period at its onset. The Supply Agreement can be extended by either party for a term of five years with the option for the Company to buyout the exclusive supply provision during any extended term. This Supply Agreement replaces the Manufacturing Agreement between MiniFAB and the Company from August 2011.
| -62- |
In April 2014, the Company guaranteed a marketing agreement entered into by OcuHub LLC, which was a wholly-owned subsidiary of the Company at the time. The underlying marketing agreement calls for an annual marketing fee of $100, with payments due quarterly through March 31, 2019. Should OcuHub LLC fail to perform its obligations under the terms of the marketing agreement, the Company could be required to make the $25 quarterly payments, through the March 31, 2019 initial term of the agreement. The Company has not accrued for any liability under the guarantee.
In the normal course of business, the Company enters into purchase obligations for future goods and services needed for the operations of the business. Such commitments are not in excess of expected requirements and are not reasonably likely to result in performance penalties or payments that would have a material adverse effect on the Company’s liquidity.
Contingencies
During the ordinary course of business activities, the Company may be contingently liable for litigation and a party to claims. Currently the Company is not party to any litigation.
11. QUARTERLY FINANCIAL DATA (UNAUDITED)
The following tables in the opinion of management, reflect all adjustments, consisting of normal recurring adjustments necessary for the fair presentation of results for the periods presented (in thousands, except per share data):
| Fiscal 2016 Quarter Ended | ||||||||||||||||
| March 31 | June 30 | September 30 | December 31 | |||||||||||||
| Revenue | $ | 6,767 | $ | 6,903 | $ | 7,223 | $ | 7,121 | ||||||||
| Loss from operations | (6,336 | ) | (3,406 | ) | (2,941 | ) | (3,293 | ) | ||||||||
| Net loss | $ | (7,254 | ) | $ | (4,344 | ) | $ | (3,982 | ) | $ | (4,340 | ) | ||||
| Weighted average number of shares outstanding, basic | 3,383 | 4,485 | 5,335 | 5,360 | ||||||||||||
| Net loss per common share basic | $ | (2.14 | ) | $ | (0.97 | ) | $ | (0.75 | ) | $ | (0.81 | ) | ||||
| Weighted average number of shares outstanding diluted | 3,383 | 4,485 | 5,335 | 5,360 | ||||||||||||
| Net loss per common share diluted | $ | (2.14 | ) | $ | (0.97 | ) | $ | (0.75 | ) | $ | (0.81 | ) | ||||
| Fiscal 2015 Quarter Ended | ||||||||||||||||
| March 31 | June 30 | September 30 | December 31 | |||||||||||||
| Revenue | $ | 5,408 | $ | 6,345 | $ | 6,612 | $ | 6,791 | ||||||||
| Loss from operations | (8,071 | ) | (7,702 | ) | (7,543 | ) | (8,193 | ) | ||||||||
| Net loss | $ | (8,168 | ) | $ | (8,072 | ) | $ | (8,064 | ) | $ | (8,925 | ) | ||||
| Weighted average number of shares outstanding, basic | 3,364 | 3,366 | 3,373 | 3,376 | ||||||||||||
| Net loss per common share basic | $ | (2.43 | ) | $ | (2.40 | ) | $ | (2.39 | ) | $ | (2.64 | ) | ||||
| Weighted average number of shares outstanding diluted | 3,369 | 3,370 | 3,377 | 3,376 | ||||||||||||
| Net loss per common share diluted | $ | (2.42 | ) | $ | (2.39 | ) | $ | (2.39 | ) | $ | (2.64 | ) | ||||
| (i) | Net loss per share basic and diluted are computed independently for the quarters presented. Therefore, the sum of the quarterly per share information may not be equal to the annual per share information. Also totals may not add to the financial statements due to rounding. |
| -63- |
12. SUBSEQUENT EVENTS
On February 24, 2017, the Company’s stockholders authorized the board of directors to implement a reverse stock split, along with a corresponding reduction in the number of shares authorized. On February 27, 2017, the Company effected a 1-for-10 reverse stock split of its common stock. All common stock share amounts and prices per share of common stock in these Consolidated Financial Statements have been retroactively adjusted to reflect the reverse stock split.
On October 13, 2016, TearLab Corporation (the “Company”) and Physician Recommended Nutriceuticals (“PRN”) entered into a Cooperative Marketing Agreement (as amended, the “Marketing Agreement”) pursuant to which the Company and PRN will jointly promote PRN’s proprietary omega-3 formulations, including Dry Eye Omega Benefits®. The Marketing Agreement was amended and restated on March 3, 2017.
The Marketing Agreement has a 30 month term with an option for mutual renewal by both parties. In addition, the agreement has a demonstration period through December 31, 2017 whereby the agreement can be terminated by PRN if certain milestones are not achieved, or by the Company upon forfeiture of a portion of the Company’s marketing fee for the demonstration period. After the demonstration period is completed, the Company will earn a marketing fee based on a percentage of revenue of the promoted products that varies over the life of the agreement and can vary by different customer channels. The Marketing Agreement contains provisions for termination upon change of control as well as non-compete provisions.
| -64- |
ITEM 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
Not applicable.
ITEM 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our reports under the Securities Exchange Act of 1934, as amended, is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms, and that such information is accumulated and communicated to our management, including our chief executive officer and chief financial officer, as appropriate, to allow timely decisions regarding required disclosure. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefit of controls must be considered relative to their costs. In designing and evaluating the disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and management necessarily is required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
As of the end of the period covered by the report, we carried out an evaluation, under the supervision and with the participation of our management, including our chief executive officer and chief financial officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)). Based on that evaluation, our chief executive officer and chief financial officer concluded that, as of December 31, 2016 our disclosure controls and procedures were effective at the reasonable assurance level.
MANAGEMENT’S REPORT ON INTERNAL CONTROL OVER FINANCIAL REPORTING
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Because of its inherent limitations, internal control over financial reporting may not prevent or detect all misstatements. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation.
We conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework in Internal Control—Integrated Framework published by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework). Based on our evaluation under the framework, our management has concluded that the Company’s internal control over financial reporting was effective, at the reasonable assurance level, as of December 31, 2016.
Changes in Internal Control over Financial Reporting
There has been no change in our internal control over financial reporting that occurred during the most recent fiscal quarter that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
| -65- |
On October 13, 2016, TearLab Corporation (the “Company”) and Physician Recommended Nutriceuticals (“PRN”) entered into a Cooperative Marketing Agreement ( as amended, the “Marketing Agreement”) pursuant to which the Company and PRN will jointly promote PRN’s proprietary omega-3 formulations, including Dry Eye Omega Benefits®. The Marketing Agreement was amended and restated on March 3, 2017.
The Marketing Agreement has a 30 month term with an option for mutual renewal by both parties. In addition, the agreement has a demonstration period through December 31, 2017 whereby the agreement can be terminated by PRN if certain milestones are not achieved, or by the Company upon forfeiture of a portion of the Company’s marketing fee for the demonstration period. After the demonstration period is completed, the Company will earn a marketing fee based on a percentage of revenue of the promoted products that varies over the life of the agreement and can vary by different customer channels. The Marketing Agreement contains provisions for termination upon change of control as well as non-compete provisions.
The foregoing summary of the Marketing Agreement does not purport to be complete and is qualified in its entirety by reference to the Marketing Agreement, which is filed as an exhibit to this Annual Report on Form 10-K.
| -66- |
PART III
ITEM 10. Directors, Executive Officers and Corporate Governance
The information required by this item will be set forth in our Proxy Statement related to the 2017 Annual Meeting of Stockholders which we intend to file with the Securities and Exchange Commission within 120 days of the end of our fiscal year pursuant to General Instructions G(3) of form 10-K and is incorporated in this report by reference.
ITEM 11. Executive Compensation
The information required by this item will be set forth in our Proxy Statement related to the 2017 Annual Meeting of Stockholders which we intend to file with the Securities and Exchange Commission within 120 days of the end of our fiscal year pursuant to General Instructions G(3) of form 10-K and is incorporated in this report by reference.
ITEM 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this item will be set forth in our Proxy Statement related to the 2017 Annual Meeting of Stockholders which we intend to file with the Securities and Exchange Commission within 120 days of the end of our fiscal year pursuant to General Instructions G(3) of form 10-K and is incorporated in this report by reference.
ITEM 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this item will be set forth in our Proxy Statement related to the 2017 Annual Meeting of Stockholders which we intend to file with the Securities and Exchange Commission within 120 days of the end of our fiscal year pursuant to General Instructions G(3) of form 10-K and is incorporated in this report by reference.
ITEM 14. Principal Accounting Fees and Services
The information required by this item will be set forth in our Proxy Statement related to the 2017 Annual Meeting of Stockholders which we intend to file with the Securities and Exchange Commission within 120 days of the end of our fiscal year pursuant to General Instructions G(3) of form 10-K and is incorporated in this report by reference.
| -67- |
PART IV
ITEM 15. Exhibits, Financial Statement Schedules
(a) The following documents are filed as part of the report:
| (1) | Financial Statements included in PART II of this report: |
| (2) | List of exhibits required by Item 601 of Regulation S-K. See part (b) below. | |
| (b) | Exhibits: The following exhibits are filed as a part of this report: |
All other financial statement schedules have been omitted because they are not applicable, not required or the information required is shown in the financial statements or the notes thereto.
| Exhibit | ||||
| Number | Exhibit Description | Incorporated by Reference | ||
| 3.1 | Amended and Restated Certificate of Incorporation of the Registrant currently in effect | Exhibit 3.3 to the Registrant’s Current Report on Form 8-K filed with the Commission on October 9, 2008 (file no. 000-51030) | ||
| 3.2 | Amended and Restated By-Laws of the Registrant currently in effect | Exhibit 3.4 to the Registrant’s Registration Statement on Form S-1/A No. 3, filed with the Commission on November 16, 2004 (file no. 333-118024) | ||
| 3.3 | Certificate of Amendment to Amended and Restated Certificate of Incorporation | Exhibit 3.2 to the Registrant’s Current Report on Form 8-K filed with the Commission on October 9, 2008 (file no. 000-51030) | ||
| 3.4 | Certificate of Amendment to Amended and Restated Certificate of Incorporation | Exhibit 3.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on May 19, 2010 (file no. 000-51030) | ||
| 3.5 | Certificate of Amendment to Amended and Restated Certificate of Incorporation | Exhibit 3.4 to the Registrant’s Post Effective Amendment No. 1 to Form S-3 filed with the Commission on July 15, 2013 (file no. 333-189372) | ||
| 3.6 | Form of Certificate of Designation of Preferences, Rights and Limitations of Series A Convertible Preferred Stock | Exhibit 3.6 to the Registrant’s Registration Statement on Form S-1/A No. 2 filed with the Commission on April 29, 2016 (file no. 333-210326) |
| -68- |
| Exhibit | ||||
| Number | Exhibit Description | Incorporated by Reference | ||
| 3.7 | Certificate of Amendment to Amended and Restated Certificate of Incorporation | Exhibit 3.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on June 30, 2016 (file no. 000-51030) | ||
| 3.8 | Certificate of Amendment to Amended and Restated Certificate of Incorporation | Exhibit 3.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on February 27, 2017 (file no. 000-51030) | ||
| 4.1 | Form of Common Stock Purchase Warrant Agreement | Exhibit A to the Registrant’s free writing prospectus filed with the Commission on March 15, 2010 (file no. 333-157269) | ||
| 4.2 | Form of Common Stock Purchase Warrant Agreement | Exhibit 10.3 to the Registrant’s Current Report on Form 8-K filed with the Commission on July 16, 2009 (file no. 000-51030) | ||
| 4.3 | Form of Senior Indenture | Exhibit 4.1 to the Registrant’s Registration Statement on Form S-3 filed with the Commission on January 2, 2015 (file no. 333-201355) | ||
| 4.4 | Form of Subordinated Indenture | Exhibit 4.2 to the Registrant’s Registration Statement on Form S-3 filed with the Commission on January 2, 2015 (file no. 333-201355) | ||
| 4.5 | Form of warrant issued to certain affiliated funds of CRG LP (formerly known as Capital Royalty) pursuant to the terms of the Term Loan Agreement, dated as of March 4, 2015, as amended by the Omnibus Amendment Agreement, dated as of April 2, 2015, Amendment 2, dated August 6, 2015, Amendment 3, dated December 31, 2015, and Amendment 4, dated April 7, 2016, by and among TearLab Corporation, certain of its subsidiaries from time to time party thereto as guarantors and CRG LP (formerly known as Capital Royalty) and certain of its affiliate funds, as lenders, dated as of April 7, 2016 | Exhibit 4.1 to the Registrant’s Quarterly Report on Form 10-Q filed with the Commission on May 9, 2016 (file no. 000-51030) | ||
| 4.6 | Form of Series A Warrant | Exhibit 4.6 to the Registrant’s Registration Statement on Form S-1/A No. 2 filed with the Commission on April 29, 2016 (file no. 333-210326) | ||
| 10.1 | License Agreement between TearLab, Inc. and The Regents of the University of California dated March 12, 2003. | Exhibit 10.48 to the Registrant’s Annual Report on Form 10-K/A, filed with the Commission on March 29, 2007 (file no. 000-51030) (Portions of this exhibit have been omitted pursuant to a request for confidential treatment.) |
| -69- |
| Exhibit | ||||
| Number | Exhibit Description | Incorporated by Reference | ||
| 10.2 | Amendment No. 1, dated June 9, 2003, to the License Agreement between TearLab, Inc. and The Regents of the University of California dated March 12, 2003. | Exhibit 10.49 to the Registrant’s Annual Report on Form 10-K/A, filed with the Commission on March 29, 2007 (file no. 000-51030) | ||
| 10.3 | Amendment No. 2, dated September 5, 2005, to the License Agreement between TearLab, Inc. and The Regents of the University of California dated March 12, 2003. | Exhibit 10.50 to the Registrant’s Annual Report on Form 10-K/A, filed with the Commission on March 29, 2007 (file no. 000-51030) (Portions of this exhibit have been omitted pursuant to a request for confidential treatment.) | ||
| 10.4 | Amendment No. 3, dated July 7, 2006, to the License Agreement between TearLab, Inc. and The Regents of the University of California dated March 12, 2003. | Exhibit 10.51 to the Registrant’s Annual Report on Form 10-K/A, filed with the Commission on March 29, 2007 (file no. 000-51030) | ||
| 10.5 | Amendment No. 4, dated October 9, 2006, to the License Agreement between TearLab, Inc. and The Regents of the University of California dated March 12, 2003. | Exhibit 10.52 to the Registrant’s Annual Report on Form 10-K/A, filed with the Commission on March 29, 2007 (file no. 000-51030) | ||
| 10.6 | Terms of Business, dated February 5, 2007, between Invetech Pty Ltd. and TearLab, Inc. | Exhibit 10.30 to the Registrant’s Annual Report on Form 10-K, filed with the Commission on March 17, 2008 (file no. 000-51030) | ||
| 10.7 | Amendment No. 5, dated June 29, 2007, to the License Agreement between TearLab, Inc. and The Regents of the University of California dated March 12, 2003. (Portions of this exhibit have been omitted pursuant to a request for confidential treatment). | Exhibit 10.31 to the Registrant’s Annual Report on Form 10-K, filed with the Commission on March 17, 2008 (file no. 000-51030) | ||
| 10.8 | # | Securities Purchase Agreement, dated as of March 14, 2010, by and between the Registrant and certain investors. | Registrant’s free writing prospectus filed with the Commission on March 15, 2010 (file no. 333-157269) | |
| 10.9 | Form of Director and Affiliate Letter Agreement | Exhibit 10.5 to the Registrant’s Current Report on Form 8-K filed with the Commission on July 16, 2009 (file no. 000-51030) | ||
| 10.10 | Deed and Amendment, dated December 22, 2011, to Manufacturing and Development Agreement by and between TearLab Research, Inc. and MiniFAB AB (Aust) Pty Ltd. Dated August 1, 2011 (Portions of this exhibit have been omitted pursuant to a request for confidential treatment). | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on December 29, 2011 (file no. 000-51030) | ||
| 10.11 | Manufacturing and Development Agreement by and between TearLab Research, Inc. and MiniFAB (Aust) Pty Ltd, dated August 1, 2011. (Portions of this exhibit have been omitted pursuant to a request for confidential treatment). | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on August 25, 2011 (file no. 000-51030) |
| -70- |
| Exhibit | ||||
| Number | Exhibit Description | Incorporated by Reference | ||
| 10.12 | Purchase Agreement, dated as of April 11, 2012, by and between the Registrant and Craig-Hallum Capital Group LLC. | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on April 11, 2012 (file no. 000-51030) | ||
| 10.13 | # | Form Change of Control Severance Agreement (for US executives). | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on June 21, 2013 (file no. 000-51030) | |
| 10.14 | # | Form Change of Control Severance Agreement (for Canadian executives). | Exhibit 10.2 to the Registrant’s Current Report on Form 8-K filed with the Commission on June 21, 2013 (file no. 000-51030) | |
| 10.15 | # | Offer Letter, dated September 24, 2013, by and between the Registrant and Joseph Jensen. | Exhibit 10.1# to the Registrant’s Current Report on Form 8-K filed with the Commission on October 1, 2013 (file no. 000-50789) | |
| 10.16 | # | Nonstatutory Stock Option Agreement, dated October 21, 2013, by and between the Company and Joseph Jensen. | Exhibit 10.1# to the Registrant’s Current Report on Form 8-K filed with the Commission on October 21, 2013 (file no. 000-50789) | |
| 10.17 | Asset Purchase Agreement, dated March 14, 2014 by and among AOA Excel, Inc., Occulogix LLC and TearLab Corporation. | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on March 17, 2014 (file no. 000-51030) | ||
| 10.18 | Term Loan Agreement, dated as of March 4, 2015, by and among TearLab Corporation, certain of its subsidiaries from time to time party thereto as guarantors and CRG LP (formerly known as Capital Royalty) and certain of its affiliate funds, as lenders. | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on March 10, 2015 (file no. 000-51030) | ||
| 10.19 | # | Nonstatutory Stock Option Agreement, dated April 21, 2014 by and between the Company and Paul Smith | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on April 21, 2014 (file no. 000-51030) | |
| 10.20 | # | Offer Letter, dated May 15, 2015, by and between the Registrant and Wes Brazell | Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed with the Commission on July 6, 2015 (file no. 000-51030) | |
| 10.21 | # | 2002 Stock Option Plan, as amended effective as of February 5, 2015. | Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q filed with the Commission on August 7, 2015 (file no. 000-51030) | |
| 10.22 | # | OcuHub LLC 2015 Equity Incentive Plan | Exhibit 10.22 to Amendment No. 1 to the Registrant’s Annual Report on Form 10-K filed with the Commission on March 22, 2016 (file no. 000-51030) | |
| 10.23 | # | Option Agreement dated as of October 1, 2015 by and between OcuHub LLC and Elias Vamvakas | Exhibit 10.23 to Amendment No. 1 to the Registrant’s Annual Report on Form 10-K filed with the Commission on March 22, 2016 (file no. 000-51030) |
| -71- |
| Exhibit | ||||
| Number | Exhibit Description | Incorporated by Reference | ||
| 10.24 | # | Profits Interest Award Agreement dated as of October 1, 2015 by and between OcuHub LLC and Elias Vamvakas | Exhibit 10.24 to Amendment No. 1 to the Registrant’s Annual Report on Form 10-K filed with the Commission on March 22, 2016 (file no. 000-51030) | |
| 10.25 | Amendment to Term Loan Agreement, dated as of March 4, 2015, as amended by the Omnibus Amendment Agreement, dated as of April 2, 2015, and Amendment 2, dated August 6, 2015, by and among the Registrant, certain of its subsidiaries from time to time party thereto as guarantors and CRG LP (formerly known as Capital Royalty) and certain of its affiliate funds, as lenders, dated as of December 31, 2015 | Exhibit 10.25 to Amendment No. 1 to the Registrant’s Annual Report on Form 10-K filed with the Commission on March 22, 2016 (file no. 000-51030) | ||
| 10.26 | # | Employment Agreement, dated as of December 31, 2015, by and between the Registrant and Elias Vamvakas | Exhibit 10.26 to Amendment No. 1 to the Registrant’s Annual Report on Form 10-K filed with the Commission on March 22, 2016 (file no. 000-51030) | |
| 10.27 | * | Manufacturing, Supply and Development Agreement between MiniFAB (Aust) Pty Ltd and TearLab Research, Inc., dated March 7, 2016 | Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q filed with the Commission on May 9, 2016 (file no. 000-51030) | |
| 10.28 | Amendment to Term Loan Agreement, dated as of March 4, 2015, as amended by the Omnibus Amendment Agreement, dated as of April 2, 2015, Amendment 2, dated August 6, 2015, and Amendment 3, dated December 31, 2015, by and among the Registrant, certain of its subsidiaries from time to time party thereto as guarantors and CRG LP (formerly known as Capital Royalty) and certain of its affiliate funds, as lenders, dated as of April 7, 2016 | Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q filed with the Commission on May 9, 2016 (file no. 000-51030) | ||
| 10.29 | † | Amended and Restated Cooperative Marketing Agreement between PRN Physician Recommended Nutriceuticals, LLC and TearLab Research, Inc. | Filed herewith | |
| 21.1 | Subsidiaries of Registrant. | Exhibit 21.1 to the Registrant’s Registration Statement on Form S-1, filed with the Commission on July 28, 2011 (file no. 333-175861) | ||
| 23.1 | Consent of Independent Registered Public Accounting Firm | |||
| 24.1 | Power of Attorney (included on signature page). | |||
| 31.1 | CEO’s Certification required by Rule 13A-14(a) of the Securities Exchange Act of 1934. | |||
| 31.2 | CFO’s Certification required by Rule 13A-14(a) of the Securities Exchange Act of 1934. | |||
| 32.1 | CEO’s Certification of periodic financial reports pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, U.S.C. Section 1350. |
| -72- |
| Exhibit | ||||
| Number | Exhibit Description | Incorporated by Reference | ||
| 32.2 | CFO’s Certification of periodic financial reports pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, U.S.C. Section 1350. | |||
| 101.INS* | XBRL Instance. | |||
| 101.SCH* | XBRL Taxonomy Schema. | |||
| 101.CAL* | XBRL Taxonomy Extension Calculation. | |||
| 101.DEF* | XBRL Taxonomy Extension Definition. | |||
| 101.LAB* | XBRL Taxonomy Extension Labels. | |||
| 101.PRE* | XBRL Taxonomy Extension Presentation |
* * *
#Management compensatory plan, contract or arrangement
| * | Portions of the exhibit have been omitted pursuant to an order granted by the Securities and Exchange Commission for confidential treatment. |
| † | Portions of the exhibit have been omitted pursuant to a request for confidential treatment and the non-public information has been filed separately with the Securities and Exchange Commission. |
| ● | XBRL information is furnished and not filed or a part of a registration statement or prospectus for purposes of sections 11 or 12 of the Securities Act of 1933, as amended, is deemed not filed for purposes of section 18 of the Securities Exchange Act of 1924, as amended, and otherwise is not subject to liability under these sections. |
Copies of the exhibits filed with this Annual Report on Form 10-K or incorporated by reference herein do not accompany copies hereof for distribution to stockholders of the Registrant. The Registrant will furnish a copy of any of such exhibits to any stockholder requesting the same for a nominal charge to cover duplicating costs.
POWER OF ATTORNEY
The Registrant and each person whose signature appears below hereby appoint Elias Vamvakas, Joseph Jensen and Wes Brazell as attorney-in-fact with full power of substitution, severally, to execute in the name and on behalf of the Registrant and each such person, individually and in each capacity stated below, one or more amendments to this Annual Report on Form 10-K, which amendments may make such changes in this Annual Report as the attorney-in-fact acting in the premises deems appropriate and to file any such amendments to this Annual Report on Form 10-K with the Securities and Exchange Commission.
| -73- |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Annual Report on Form 10-K to be signed on its behalf by the undersigned thereunto duly authorized.
| Dated: March 10, 2017 | TearLab Corp. | |
| By: | /s/ Joseph Jensen | |
| Joseph Jensen | ||
| Chief Executive Officer | ||
Pursuant to the requirements of the Securities Exchange Act of 1934, this Annual Report on Form 10-K has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
| Dated: March 10, 2017 | By: | /s/ Joseph Jensen |
| Joseph Jensen | ||
| Chief Executive Officer and Director (principal executive officer) |
| Dated: March 10, 2017 | By: | /s/ Wes Brazell |
| Wes Brazell | ||
| Chief Financial Officer (principal financial and accounting officer) |
| Dated: March 10, 2017 | By: | /s/ Elias Vamvakas |
| Elias Vamvakas | ||
| Chairman of Board of Directors |
| Dated: March 10, 2017 | By: | /s/ Anthony Altig |
| Anthony Altig | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Thomas N. Davidson, Jr. |
| Thomas N. Davidson, Jr. | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Adrienne L. Graves |
| Adrienne L. Graves | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Joseph S. Jensen |
| Joseph S. Jensen | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Richard L. Lindstrom, M.D. |
| Richard L. Lindstrom, M.D. | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Donald Rindell |
| Donald Rindell | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Paul Karpecki |
| Paul Karpecki | ||
| Director |
| Dated: March 10, 2017 | By: | /s/ Brock Wright |
| Brock Wright | ||
| Director |
| -74- |
