Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - BIOGEN INC. | titration1.htm |
| 8-K - 8-K - BIOGEN INC. | december920168-k.htm |

Interim analysis presented at CTAD 2016
Aducanumab 24-Month Data From PRIME: A Randomized
Double-Blind, Placebo-Controlled Phase 1b Study in
Patients With Prodromal or Mild Alzheimer’s Disease
Vissia Viglietta,1 John O’Gorman,1 Leslie Williams,1 Tianle Chen,1
Ahmed Enayetallah,1 Ping Chiao,1 Christoph Hock,2 Roger M. Nitsch,2
Samantha Budd Haeberlein,1 Alfred Sandrock1
1Biogen, Cambridge, MA, USA; 2Neurimmune, Schlieren-Zurich, and University of Zurich, Switzerland

Interim analysis presented at CTAD 2016
Disclosures
This study was funded by Biogena
VV, JO, LW, TC, AE, PC, SBH, and AS are employees and
shareholders of Biogen
CH and RMN are employees and shareholders of Neurimmune
aMedical writing support for this presentation was provided by Erin Bekes, PhD, of Complete Medical Communications and funded by Biogen.

Interim analysis presented at CTAD 2016
Overview
Aducanumab is a human monoclonal antibody selective for aggregated
forms of Aβ, including soluble oligomers and insoluble fibrils
PRIME is an ongoing Phase 1b study assessing the safety, tolerability, PK
and PD of aducanumab in patients with prodromal or mild Alzheimer’s
disease
Here we present 24-month data from the 12-month placebo-controlled period
and the first 12 months of the LTE period of PRIME
- Data from the titration cohort are not reported because 24-month data are not yet
available for this cohort
Primary endpoint in the LTE was safety/tolerability
Exploratory endpoints included:
• Changes in amyloid PET
• Measures of clinical decline on the CDR–SB and MMSE
CDR-SB, Clinical Dementia Rating-Sum of Boxes; MMSE, Mini-Mental State Examination; PD, pharmacodynamics; PET, positron emission
tomography; PK, pharmacokinetics; LTE, long-term extension

Interim analysis presented at CTAD 2016
• Randomization: 3:1 active: placebo within cohorts, fixed-dose cohorts stratified by ApoE ε4 status
• Patients randomized to placebo or aducanumab 1 mg/kg in the placebo-controlled period were switched to aducanumab
3 mg/kg or titration in the LTE (“switchers”). Patients randomized to aducanumab 3, 6, or 10 mg/kg or titration in the
placebo-controlled period were assigned to continue in the same dose group in the LTE (“continuers”)
PRIME Study Design:
Placebo-Controlled and LTE Periods
aData from the titration cohort are not included in this analysis as 24-month data from this cohort are not yet available. bFor patients switched
from placebo to titration in the LTE, titration denotes 2 doses of 3 mg/kg followed by subsequent doses of 6 mg/kg.
1 mg/kg
3 mg/kg
Placebo
Titration (ApoE ε4 carriers; 1→10 mg/kg)a
Placebo (ApoE ε4 carriers)a
6 mg/kg
Placebo
Placebo-controlled period
10 mg/kg
Placebo
LTE period
All patients receive aducanumab
3 mg/kg
Aducanumab 3 mg/kg or titration (3→6 mg/kg)b
Aducanumab titration (ApoE ε4 carriers;
1→10 mg/kg)a
Aducanumab titration (3→6 mg/kg)b
3 mg/kg
Titration (ApoE ε4 carriers; 1→10 mg/kg)a
6 mg/kg
10 mg/kg
Aducanumab titration (3→6 mg/kg)b

Interim analysis presented at CTAD 2016
Timeline of Dose Administration and
Key Assessments in PRIME
aSchedule of brain MRIs for fixed-dose cohorts (Arms 1-7)
MRI, magnetic resonance imaging
IV infusions every 4 weeks over 52 weeks (14 total)
active or placebo
IV infusions every 4 weeks over a total of 3 years
All patients received active treatment
6 52-5418 30 42
LTE
Years 2-3Screening
62 70 76-78 90 102 108-110
Placebo controlled period
Week
MRIa
Amyloid
PET
Clinical
Tests
LTE (Year 1)
24-26

Interim analysis presented at CTAD 2016
Dosed in placebo-controlled
period
Completed treatment in the
placebo-controlled period
166 randomized
(165 dosed)
1 mg/kg
31
Pooled
placebo
40
3 mg/kg
32
6 mg/kg
30
10 mg/kg
32
2430 26 25 20
Patient Disposition at 24 Months
Analysis of data from fixed-dose arms up to Month 24.
Discontinued treatment
in the first year of the LTE
AE
Other
Death
Disease progression
4
0
4
0
0
8
7
0
0
1
8
2
6
0
0
2
0
1
1
0
4
3
1
0
0
Dosed in the LTE 1929 26 24 19
Switchers Continuers
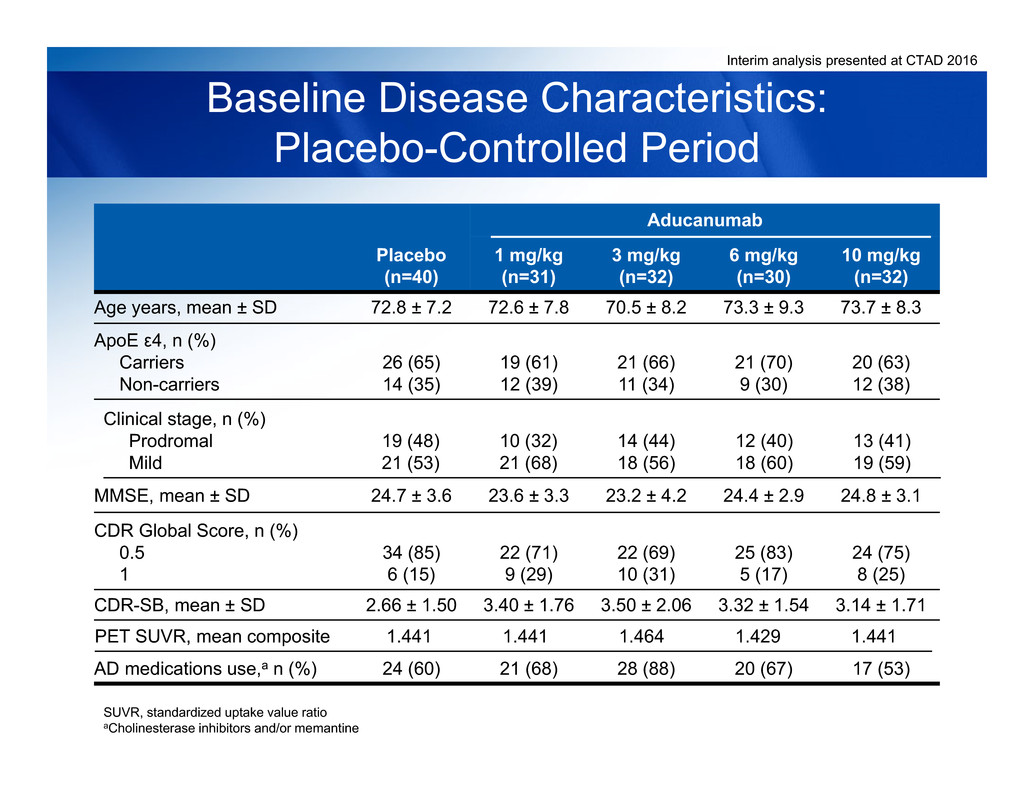
Interim analysis presented at CTAD 2016
Baseline Disease Characteristics:
Placebo-Controlled Period
SUVR, standardized uptake value ratio
aCholinesterase inhibitors and/or memantine
Placebo
(n=40)
Aducanumab
1 mg/kg
(n=31)
3 mg/kg
(n=32)
6 mg/kg
(n=30)
10 mg/kg
(n=32)
ApoE ε4, n (%)
Carriers
Non-carriers
26 (65)
14 (35)
19 (61)
12 (39)
21 (66)
11 (34)
21 (70)
9 (30)
20 (63)
12 (38)
Clinical stage, n (%)
Prodromal
Mild
19 (48)
21 (53)
10 (32)
21 (68)
14 (44)
18 (56)
12 (40)
18 (60)
13 (41)
19 (59)
MMSE, mean ± SD 24.7 ± 3.6 23.6 ± 3.3 23.2 ± 4.2 24.4 ± 2.9 24.8 ± 3.1
CDR Global Score, n (%)
0.5
1
34 (85)
6 (15)
22 (71)
9 (29)
22 (69)
10 (31)
25 (83)
5 (17)
24 (75)
8 (25)
Age years, mean ± SD 72.8 ± 7.2 72.6 ± 7.8 70.5 ± 8.2 73.3 ± 9.3 73.7 ± 8.3
CDR-SB, mean ± SD 2.66 ± 1.50 3.40 ± 1.76 3.50 ± 2.06 3.32 ± 1.54 3.14 ± 1.71
PET SUVR, mean composite 1.441 1.441 1.464 1.429 1.441
AD medications use,a n (%) 24 (60) 21 (68) 28 (88) 20 (67) 17 (53)

Interim analysis presented at CTAD 2016
PET AMYLOID IMAGING
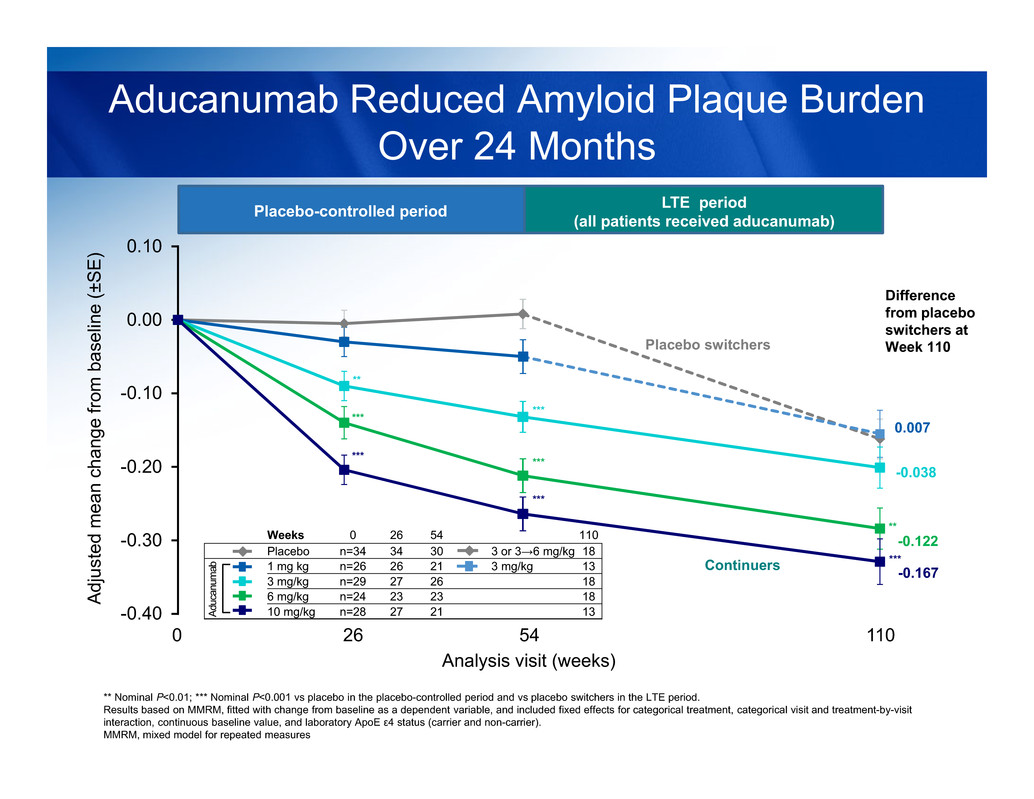
-0.40
-0.30
-0.20
-0.10
0.00
0.10
Aducanumab Reduced Amyloid Plaque Burden
Over 24 Months
** Nominal P<0.01; *** Nominal P<0.001 vs placebo in the placebo-controlled period and vs placebo switchers in the LTE period.
Results based on MMRM, fitted with change from baseline as a dependent variable, and included fixed effects for categorical treatment, categorical visit and treatment-by-visit
interaction, continuous baseline value, and laboratory ApoE ε4 status (carrier and non-carrier).
MMRM, mixed model for repeated measures
A
d
j
u
s
t
e
d
m
e
a
n
c
h
a
n
g
e
f
r
o
m
b
a
s
e
l
i
n
e
(
±
S
E
)
Placebo-controlled period LTE period
(all patients received aducanumab)
Weeks 0 26 54 110
Placebo n=34 34 30 3 or 3→6 mg/kg 18
1 mg kg n=26 26 21 3 mg/kg 13
3 mg/kg n=29 27 26 18
6 mg/kg n=24 23 23 18
10 mg/kg n=28 27 21 13
A
d
u
c
a
n
u
m
a
b
Analysis visit (weeks)
110
Difference
from placebo
switchers at
Week 110
0.007
54260
***
**
***
******
***
**
***
-0.038
-0.167
-0.122
Placebo switchers
Continuers

Interim analysis presented at CTAD 2016
CLINICAL ENDPOINTS
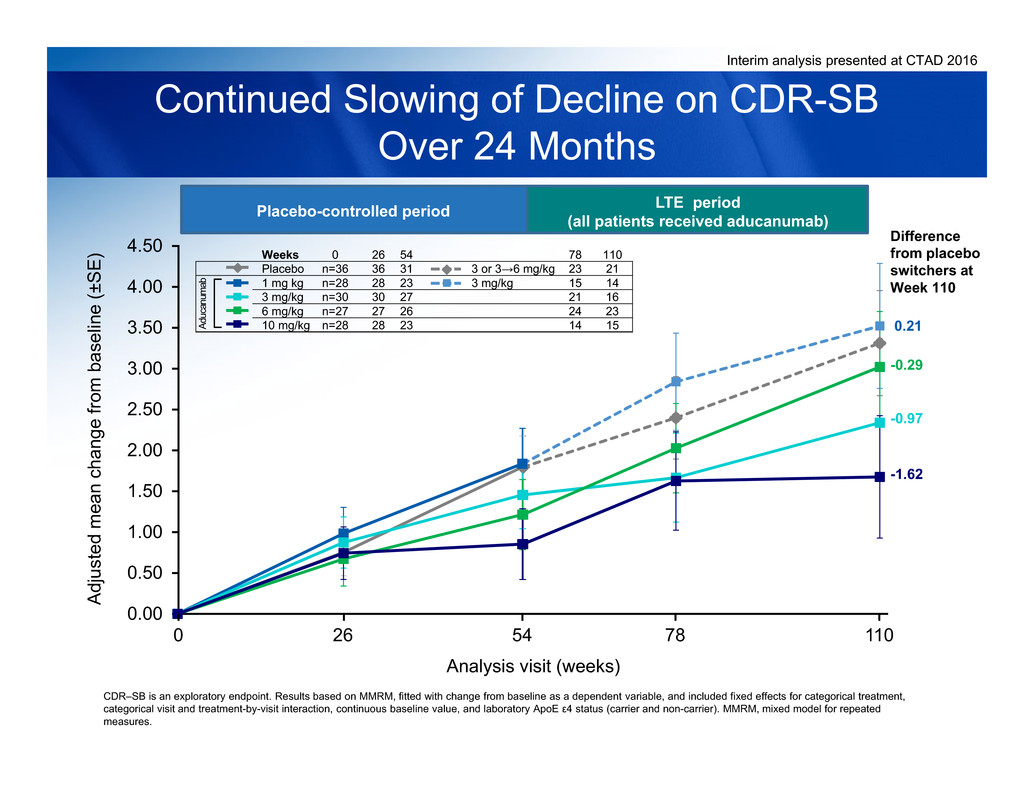
Interim analysis presented at CTAD 2016
0.00
0.50
1.00
1.50
2.00
2.50
3.00
3.50
4.00
4.50
Continued Slowing of Decline on CDR-SB
Over 24 Months
CDR‒SB is an exploratory endpoint. Results based on MMRM, fitted with change from baseline as a dependent variable, and included fixed effects for categorical treatment,
categorical visit and treatment-by-visit interaction, continuous baseline value, and laboratory ApoE ε4 status (carrier and non-carrier). MMRM, mixed model for repeated
measures.
Placebo-controlled period LTE period
(all patients received aducanumab)
A
d
j
u
s
t
e
d
m
e
a
n
c
h
a
n
g
e
f
r
o
m
b
a
s
e
l
i
n
e
(
±
S
E
)
Analysis visit (weeks)
Weeks 0 26 54 78 110
Placebo n=36 36 31 3 or 3→6 mg/kg 23 21
1 mg kg n=28 28 23 3 mg/kg 15 14
3 mg/kg n=30 30 27 21 16
6 mg/kg n=27 27 26 24 23
10 mg/kg n=28 28 23 14 15A d
u
c
a
n
u
m
a
b
Difference
from placebo
switchers at
Week 110
0.21
-0.97
-1.62
-0.29
11054260 78

Interim analysis presented at CTAD 2016
-7
-6
-5
-4
-3
-2
-1
0
1
Continued Slowing of Decline on MMSE
Over 24 Months
*Nominal P<0.05 (vs placebo [Week 52] or placebo switchers [Weeks 76 and 108])
MMSE is an exploratory endpoint. Results based on MMRM, fitted with change from baseline as a dependent variable, and included fixed effects for categorical treatment,
categorical visit and treatment-by-visit interaction, continuous baseline value, and laboratory ApoE ε4 status (carrier and non-carrier). MMRM, mixed model for repeated
measures.
Analysis visit (weeks)
A
d
j
u
s
t
e
d
m
e
a
n
c
h
a
n
g
e
f
r
o
m
b
a
s
e
l
i
n
e
(
±
S
E
)
Placebo-controlled period LTE period
(all patients received aducanumab)
Weeks 0 24 52 76 108
Placebo n=37 36 32 3 or 3→6 mg/kg 24 21
1 mg kg n=26 26 25 3 mg/kg 15 15
3 mg/kg n=29 29 26 21 17
6 mg/kg n=28 28 26 24 23
10 mg/kg n=30 29 25 14 15A d
u
c
a
n
u
m
a
b
10854260 78
*
Difference
from placebo
switchers at
Week 108
1.65
2.47
3.27
0.06
*
*

Interim analysis presented at CTAD 2016
SAFETY AND
TOLERABILITY
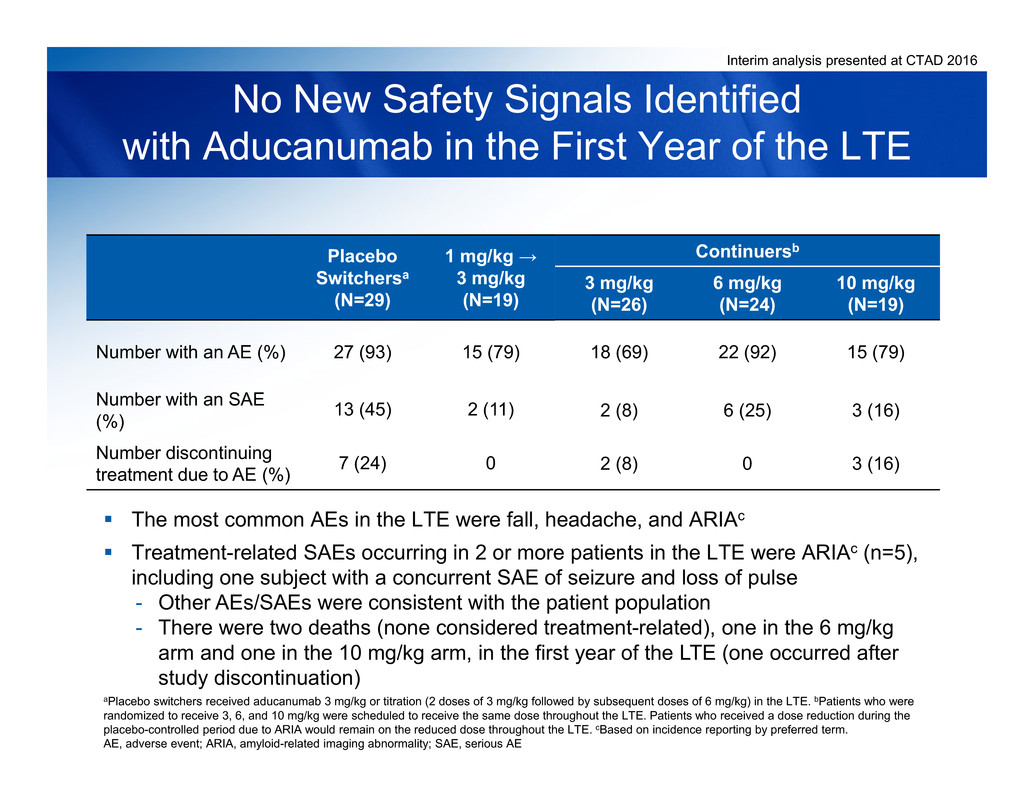
Interim analysis presented at CTAD 2016
Continuersb
3 mg/kg
(N=26)
6 mg/kg
(N=24)
10 mg/kg
(N=19)
Number with an AE (%) 18 (69) 22 (92) 15 (79)
Number with an SAE
(%) 2 (8) 6 (25) 3 (16)
Number discontinuing
treatment due to AE (%) 2 (8) 0 3 (16)
No New Safety Signals Identified
with Aducanumab in the First Year of the LTE
aPlacebo switchers received aducanumab 3 mg/kg or titration (2 doses of 3 mg/kg followed by subsequent doses of 6 mg/kg) in the LTE. bPatients who were
randomized to receive 3, 6, and 10 mg/kg were scheduled to receive the same dose throughout the LTE. Patients who received a dose reduction during the
placebo-controlled period due to ARIA would remain on the reduced dose throughout the LTE. cBased on incidence reporting by preferred term.
AE, adverse event; ARIA, amyloid-related imaging abnormality; SAE, serious AE
The most common AEs in the LTE were fall, headache, and ARIAc
Treatment-related SAEs occurring in 2 or more patients in the LTE were ARIAc (n=5),
including one subject with a concurrent SAE of seizure and loss of pulse
- Other AEs/SAEs were consistent with the patient population
- There were two deaths (none considered treatment-related), one in the 6 mg/kg
arm and one in the 10 mg/kg arm, in the first year of the LTE (one occurred after
study discontinuation)
Placebo
Switchersa
(N=29)
1 mg/kg →
3 mg/kg
(N=19)
27 (93) 15 (79)
13 (45) 2 (11)
7 (24) 0

Interim analysis presented at CTAD 2016
Patients with ≥1 post-
baseline MRI
ARIA-E,a n (%)
ApoE ε4 carrier
ApoE ε4 non-carrier
Isolated ARIA-H, n (%)
No Continuers Developed ARIA-E
During the First Year of the LTE
bPatients who were randomized to receive 3, 6, and 10 mg/kg were scheduled to receive the same dose throughout the LTE. Patients who received a dose
reduction during the placebo-controlled period due to ARIA would remain on the reduced dose throughout the LTE. cPlacebo switchers received aducanumab 3
mg/kg or titration (2 doses of 3 mg/kg followed by subsequent doses of 6 mg/kg) in the LTE.
ARIA-E, ARIA‒vasogenic edema; ARIA-H, ARIA‒microhemorrhages, macrohemorrhages, or superficial siderosis
No new ARIA-E cases or recurrence were observed among aducanumab
continuers
Continuersb
3 mg/kg 6 mg/kg 10 mg/kg
23 24 19
0/23 0/24 0/19
0/16 0/17 0/12
0/7 0/7 0/7
3/23 (13) 2/24 (8) 1/19 (5)
aARIA-E with or without ARIA-H
Placebo
Switchersc
1 mg/kg →
3 mg/kg
29 17
5/29 (17) 3/17 (18)
4/17 (24) 3/11 (27)
1/12 (8) 0/6
2/29 (7) 0/17

Interim analysis presented at CTAD 2016
Patients with ≥1 post-
baseline MRI
ARIA-Ea, n (%)
Continued treatment, n
(%)
Same dose
Reduced dose
Discontinued treatment, n
(%) 0 0 0
ApoE ε4 carriers 3 0 0 0 0
ApoE ε4 non-carriers 1 0 0 0 0
Discontinuations due to ARIA-E
During the First Year of the LTE
bPatients who were randomized to receive 3, 6, and 10 mg/kg were scheduled to receive the same dose throughout the LTE. Patients who received a dose
reduction during the placebo-controlled period due to ARIA would remain on the reduced dose throughout the LTE. cPlacebo switchers received aducanumab 3
mg/kg or titration (2 doses of 3 mg/kg followed by subsequent doses of 6 mg/kg) in the LTE.
Continuersb
3 mg/kg 6 mg/kg 10 mg/kg
23 24 19
0 0 0
0 0 0
0 0 0
0 0 0
aARIA-E with or without ARIA-H
Placebo
Switchersc
1 mg/kg →
3 mg/kg
29 17
5 (17) 3 (18)
1 ( 3) 3 (18)
0 2
1 1
4 (14) 0

Interim analysis presented at CTAD 2016
Summary
At 24 months, brain amyloid plaque burden continued to decrease in
aducanumab continuers
- This decrease was dose- and time-dependent
CDR‒SB and MMSE data suggest a clinical benefit in patients continuing
aducanumab over 24 months
No new ARIA-E cases or recurrence among aducanumab continuers
- ARIA-E incidence in aducanumab switchers was consistent with that
observed in the placebo-controlled portion of the study
These data continue to support further investigation of the clinical efficacy
and safety of aducanumab in patients with early AD in the ENGAGE and
EMERGE Phase 3 trials

Interim analysis presented at CTAD 2016
Acknowledgements
We thank all the patients and their family
members participating in the aducanumab studies
and the investigators and their staff conducting
these studies.
