Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - ALIMERA SCIENCES INC | pressrelease.htm |
| 8-K - 8-K - ALIMERA SCIENCES INC | alim8k.htm |

Third Quarter 2016 Results
Conference Call
Thursday, November 3, 2016
Exhibit 99.2

Safe Harbor Statement
This presentation contains "forward-looking statements," within the meaning of the Private Securities Litigation Reform Act of 1995,
regarding, among other things, the opportunity growth potential of ILUVEN, the adoption of reimbursement coding in the Middle East, the
market size in the Middle East, Alimera’s need for additional financing, and the potential for Alimera to expand geographically, extend the
ILUVIEN label and become cash flow positive. Such forward-looking statements are based on current expectations and involve inherent risks
and uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ materially from
those projected in its forward-looking statements. Words such as “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,”
“contemplate,” “predict,” “project,” “target,” “likely,” “potential,” “continue,” “ongoing,” “will,” “would,” “should,” “could,” or the negative
of these terms and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements
contain these identifying words. Such forward-looking statements are based on current expectations and involve inherent risks and
uncertainties, including factors that could delay, divert or change any of them, and could cause actual results to differ materially from those
projected in its forward-looking statements. Meaningful factors which could cause actual results to differ include, but are not limited to
market acceptance and physician awareness of ILUVIEN in the U.S. and Europe, the regulatory environment in the Middle East and
elsewhere, the outcome of future clinical trials, Alimera’s ability to contain its operating expenses, as well as other factors discussed in the
"Risk Factors" and "Management's Discussion and Analysis of Financial Condition and Results of Operations" sections of Alimera's Annual
Report on Form 10-K for the year ended December 31, 2015 and Quarterly Reports on Form 10-Q for the quarters ended March 31, 2016
and June 30, 2016, which are on file with the Securities and Exchange Commission (SEC) and available on the SEC's website at
http://www.sec.gov. Additional factors may also be set forth in those sections of Alimera’s Quarterly Report on Form 10-Q for the nine
months ended September 30, 2016, to be filed with the SEC in the fourth quarter of 2016. In addition to the risks described above and in
Alimera's Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and other filings with the SEC, other
unknown or unpredictable factors also could affect Alimera's results. There can be no assurance that the actual results or developments
anticipated by Alimera will be realized or, even if substantially realized, that they will have the expected consequences to, or effects on,
Alimera. Therefore, no assurance can be given that the outcomes stated in such forward-looking statements and estimates will be achieved.
All forward-looking statements contained in this presentation are expressly qualified by the cautionary statements contained or referred to
herein. Alimera cautions investors not to rely too heavily on the forward-looking statements Alimera makes or that are made on its behalf.
These forward-looking statements speak only as of the date of this presentation (unless another date is indicated). Alimera undertakes no
obligation, and specifically declines any obligation, to publicly update or revise any such forward-looking statements, whether as a result of
new information, future events or otherwise.
© 2016 Alimera Sciences, Inc., All Rights Reserved 2

© 2016 Alimera Sciences, Inc., All Rights Reserved 3
Highlights Overall Revenue Growth
3Q15 3Q16
$8.3M
$6.9M
+20% • Grew revenues year over year,
both in the US and international
• Significant progress in obtaining
reimbursement in new markets
• Strengthened balanced sheet
• Amended term loan with
Hercules

© 2016 Alimera Sciences, Inc., All Rights Reserved 4
Real World Evidence - Europe
Location N° of eyes (pts) Follow-up
Medisoft 2nd data extract results (13 sites
in the UK)1
290
Eyes (258 pts)
Up to
722
days
IRISS 2nd data extract
(37 sites in UK, DE, PT)2
328
Eyes (292 pts)
Up to
763
days
1. Bailey C. Alimera Sciences symposium at EURETINA 2015.
2. Chakravarthy U. Alimera Sciences Advisory Board held on March 12 2016 in Paris, France.
© 2016 Alimera Sciences, Inc., All Rights Reserved 5
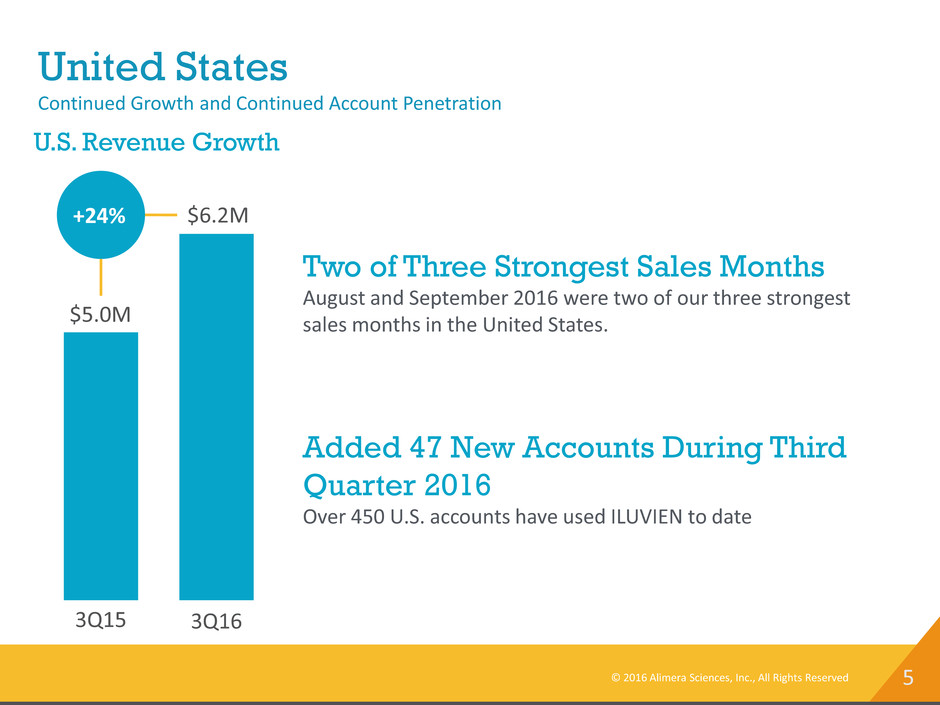
United States
Continued Growth and Continued Account Penetration
3Q15 3Q16
$6.2M
$5.0M
+24%
U.S. Revenue Growth
Two of Three Strongest Sales Months
August and September 2016 were two of our three strongest
sales months in the United States.
Added 47 New Accounts During Third
Quarter 2016
Over 450 U.S. accounts have used ILUVIEN to date

© 2016 Alimera Sciences, Inc., All Rights Reserved 6
Europe
Continued Progress
3Q15 3Q16
$2.1M
$1.9M
+11%
European Revenue
Growth
Germany
Sixth consecutive quarter of unit volume growth, a record high in
terms of ILUVIEN implants sold, despite seasonality
UK
Initiating discussions 4Q16 to amend reimbursement guidelines
Portugal
Strong growth year over year
EURETINA Conference
Strongest ever presence, 23 clinical presentations involving more
than 450 cases of ILUVIEN use

© 2016 Alimera Sciences, Inc., All Rights Reserved 7
Expanding International Footprint
Middle East
Recently announced first sales and usage of ILUVIEN under agreement with MEAgate.
Ireland
Pricing approved in July
Italy
Distribution partner SIFI is in active discussions regarding pricing anticipate a decision by year
end. If achieved, Italy launch anticipated during 2017

Financial Overview
Rick Eiswirth, President & CFO

© 2016 Alimera Sciences, Inc., All Rights Reserved 9
Select Income Statement Data
($000’s)
Three Months Ended
September 30, 2016
Unaudited Results
Three Months Ended
September 30, 2015
Unaudited Results
Revenue $8,298 $6,901
Gross Profit $7,812 $6,267
Gross Margin 94.1% 90.8%
Net (Loss) From
Operations
($7,243) ($8,444)
Net (Loss) Applicable to
Common Stockholders
($9,245) ($1,543)

© 2016 Alimera Sciences, Inc., All Rights Reserved 10
Select Balance Sheet Data
($000’s)
As of September 30, 2016
Unaudited Results
As of December 31, 2015
Cash & Cash Equivalents $33,853 $31,075
Total Current Assets $51,603 $45,159
Total Assets $74,980 $70,484
Total Current Liabilities $42,678 $39,933
Total Stockholder’s Equity $30,032 $26,320

Closing Remarks
Dan Myers, CEO

Third Quarter 2016 Results
Conference Call
Thursday, November 3, 2016
