Attached files
| file | filename |
|---|---|
| EX-99.1 - PRESS RELEASE - LUMOS PHARMA, INC. | nlnk-20160729x8kxex991.htm |
| 8-K - 8-K - LUMOS PHARMA, INC. | nlnk-20160729x8k.htm |

NewLink Genetics Corporation
Nasdaq: NLNK
July 29, 2016
Second Quarter 2016 Financial Results
Agenda
2
Introduction
Jack Henneman, Executive Vice President & CFO
2016 Ongoing Priorities
Charles J. Link, Jr., M.D., Chairman, CEO & CSO
Clinical Updates
Nicholas N. Vahanian, M.D., President & CMO
Restructuring & Second Quarter 2016 Financial Results
Mr. Henneman

Cautionary Note Regarding
Forward-Looking Statements
This presentation contains forward-looking statements of NewLink Genetics that involve substantial
risks and uncertainties. All statements, other than statements of historical facts, contained in this
presentation are forward-looking statements, within the meaning of The Private Securities Litigation
Reform Act of 1995. The words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,”
“target,” “potential,” “will,” “could,” “should,” “seek” or the negative of these terms or other similar
expressions are intended to identify forward-looking statements, although not all forward-looking
statements contain these identifying words. These forward-looking statements include, among
others, statements about NewLink Genetics’ financial guidance for 2016; results of its clinical trials
for product candidates; NewLink Genetics’ future financial performance, results of operations, cash
position and sufficiency of capital resources to fund its operating requirements; and any other
statements other than statements of historical fact. Actual results or events could differ materially
from the plans, intentions and expectations disclosed in the forward-looking statements that
NewLink Genetics makes due to a number of important factors, including those risks discussed in
“Risk Factors” and elsewhere in NewLink Genetics’ Annual Report on Form 10-K for the year
ended December 31, 2015 and other reports filed with the U.S. Securities and Exchange
Commission (SEC). The forward-looking statements in this presentation represent NewLink'
Genetics’ views as of the date of this presentation. NewLink Genetics anticipates that subsequent
events and developments will cause its views to change. However, while it may elect to update
these forward-looking statements at some point in the future, it specifically disclaims any obligation
to do so. You should, therefore, not rely on these forward-looking statements as representing
NewLink Genetics’ views as of any date subsequent to the date of this presentation.
NASDAQ: NLNK
3

4
Oncology Focused Pipeline
Two IDO pathway inhibitors in clinical development (indoximod and GDC-0919)
Research collaboration with Genentech for IDO and TDO inhibitors
Small molecule program focused on additional immuno-oncology targets
Strong Scientific and Business Leadership
Executing on vision to bring immunotherapies to patients
Strategic collaborations with Genentech/Roche and Merck
Strong cash position
Substantial Near-term News Flow
Multiple opportunities to validate IDO pathway inhibitor programs
Clinical updates on the indoximod program
Clinical advances from partnership with Genentech/Roche for GDC-0919
NewLink Genetics
Building a Leading Immuno-Oncology Company

5
Progress and accelerate the development of indoximod
Continue Genentech and Merck alliances
Advance PTEN and Zika
Evaluate external opportunities for pipeline expansion
Ongoing Priorities
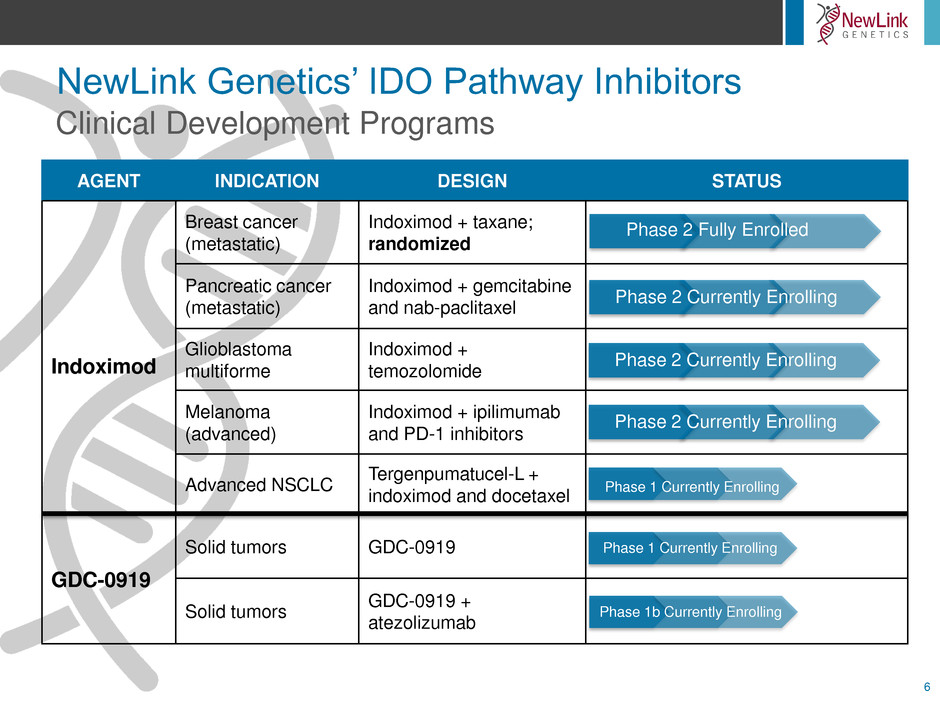
NewLink Genetics’ IDO Pathway Inhibitors
6
AGENT INDICATION DESIGN STATUS
Indoximod
Breast cancer
(metastatic)
Indoximod + taxane;
randomized
Pancreatic cancer
(metastatic)
Indoximod + gemcitabine
and nab-paclitaxel
Glioblastoma
multiforme
Indoximod +
temozolomide
Melanoma
(advanced)
Indoximod + ipilimumab
and PD-1 inhibitors
Advanced NSCLC
Tergenpumatucel-L +
indoximod and docetaxel
GDC-0919
Solid tumors GDC-0919
Solid tumors
GDC-0919 +
atezolizumab
Clinical Development Programs
Phase 2 Fully Enrolled
Phase 2 Currently Enrolling
Phase 2 Currently Enrolling
Phase 2 Currently Enrolling
Phase 1 Currently Enrolling
Phase 1 Currently Enrolling
Phase 1b Currently Enrolling

7
Conserve cash
Wind-down HyperAcute Cellular Immunotherapy trials and
manufacturing activities
Carefully planned company-wide headcount reductions to ~130
Consolidate/scale-back facilities footprint
Eliminate commercialization spending
Capital spending reduced to primarily supporting R&D needs
2016-2018 Cash Conservation Initiatives
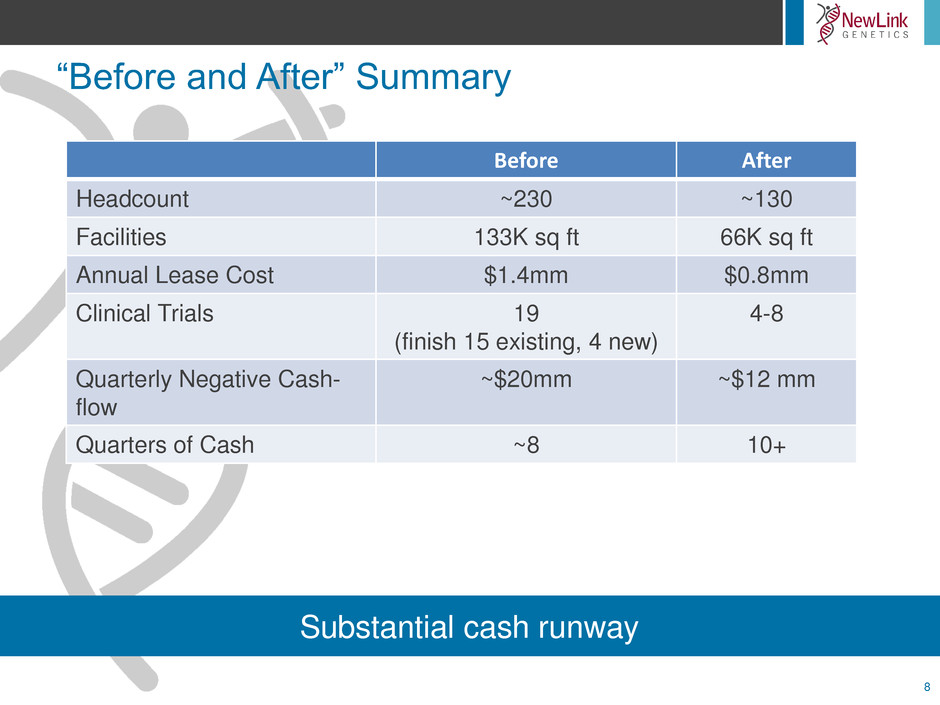
8
Substantial cash runway
Before After
Headcount ~230 ~130
Facilities 133K sq ft 66K sq ft
Annual Lease Cost $1.4mm $0.8mm
Clinical Trials 19
(finish 15 existing, 4 new)
4-8
Quarterly Negative Cash-
flow
~$20mm ~$12 mm
Quarters of Cash ~8 10+
“Before and After” Summary

Second Quarter 2016 Financial Results
9
Jack Henneman, EVP and Chief Financial Officer
Strong Capital Position
Stable and reliable cash position - $160.5M at Q2 2016 / $197.8M at YE 2015
Goal and expectation to finish 2016 with two years of cash-on-hand
Multiple Value Drivers
Capacity for Incremental Investments
Managing spending carefully
Advantage of partnered programs
Potential for combinations of IDO pathway inhibitors
Proprietary and infectious disease initiatives
Opportunity for potentially synergistic therapies

10
Q & A
