Attached files
| file | filename |
|---|---|
| 8-K - 8-K - MEDIVATION, INC. | d217665d8k.htm |
  Talazoparib (MDV3800):
A Potentially Best-in-Class PARP Inhibitor
July 2016 Exhibit 99.1 WELCOME TO MEDIVATION |
 2 Forward-Looking Statements Forward-looking statements are made throughout this presentation. The forward-looking statements in this presentation include, but are not limited to, estimates and other speculative statements regarding the potential for the PARP inhibitor class in general and talazoparib in particular and how it will drive growth for Medivation; potential clinical trial design, initiation, enrollment and results; potential regulatory approvals; and talazoparib’s addressable market in terms of indications, patients and product sales and are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward-looking statements may also be identified by words such as “expect,” believe,” “should,” “potential,” “suggest” or similar expressions. All forward-looking statements are subject to risks and uncertainties which may cause actual results to differ significantly from those expressed or implied by such forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to, the risk that clinical trials of talazoparib will be unsuccessful, the inherent uncertainty associated with the regulatory approval process and the related risk of failure to obtain marketing approval for talazoparib for its target (or any) patient populations and indications; the risk that competitive, regulatory or other factors could negatively impact the pricing of talazoparib, if approved; the risk that unexpected adverse events could impact sales of talazoparib, if approved; competition from other approved or generic products similar to talazoparib, if approved; failure to cost- effectively manufacture talazoparib, if approved; general economic conditions; and other risks detailed in Medivation’s filings with the Securities and Exchange Commission, or SEC, including its annual report on Form 10-K for the year ended December 31, 2015, which was filed on February 26, 2016, and its latest Quarterly Report on Form 10-Q. You are cautioned not to place undue reliance on the forward-looking statements, which speak only as of the date of this presentation. Medivation disclaims any obligation or undertaking to update, supplement or revise any forward-looking statements contained in this presentation. |
 Talazoparib: A Potentially Best-in-Class PARP Inhibitor
(PARPi) 3 Significantly greater potency in vitro and lowest clinical dose relative
to other PARPi’s in clinical development
Superior PARP trapping
ability Differentiated Mechanism of Action Compelling Clinical Data High & differentiated response rates observed in patients with ovarian and breast cancers Potential beyond HRD (homologous
recombination deficiency) cancers Phase 3 Data Approaching Phase 3 EMBRACA trial in BRCA-mutated breast cancer expected to complete enrollment in 2016,
top-line data expected 1H 2017 Blockbuster Potential DNA repair and PARP trapping relevant
to multiple tumor types Numerous potential opportunities as a
single-agent, in combination with low-dose chemotherapy or
radiation, or other combinations High Probability of
Success PARPi class validated through FDA approval (despite historical failure of iniparib incorrectly believed to be a PARPi) Clinical efficacy appears to correlate better with PARP trapping than PARP enzyme inhibition and talazoparib is the best PARP trapper in development |
 Most
potent PARP inhibitor reported Strongest tumor cell killing
ability Highest efficiency at PARP trapping
Highly selective for PARP1 and PARP2
Clinical proof-of-concept established
as a monotherapy and combination
therapy in multiple indications
Lowest clinical dose with most
convenient dosing regimen (1mg daily
dosing vs. 300-1,200mg given daily to
twice daily dosing for others)
Competitive safety profile
Talazoparib is Distinguished from Other PARP Inhibitors by
Several Important Characteristics
Talazoparib Appears to be the Most Optimized PARP Inhibitor in Development
4 |
 Introduction to Talazoparib |
 PARP
Inhibitors Could be the Next Major Class of Therapeutics in
Oncology Recent Equity Research Highlight the Excitement of PARP
Inhibitors 6
“PARP inhibitors may have broad applicability in cancer therapies beyond the initial indications in a subset of high- grade ovarian cancers.” - William Blair 04/11/16 “Given the potential breadth of action, we estimate that PARP inhibitors could have a large addressable market of c$20bn across ovarian, breast, pancreatic and prostate cancers.”
- BofA Merrill Lynch 06/10/16 “Upside
PARP Potential in Other Cancers or Combinations: We believe the PARP
class of drugs in cancer has promise in
more than just ovarian…. PARP market is one of the next big categories in
cancer.” - Credit Suisse 05/12/16 “Data suggests high potential
for Talazoparib” - BofA Merrill Lynch 06/29/16 “Tesaro announced positive phase 3 top-line data for niraparib – further validating the “PARP” class, a novel DNA pathway repair inhibitor, which includes Medivation’s
talazoparib.” - Leerink 06/29/16 We think the |
 PARP
Inhibitors Act Through Dual Cytotoxic Mechanisms National Cancer
Institute Research (Yves Pommier) 7
Source: Murai et al. Cancer Res 2012, 72(21) 5588-99; and Medivation. Mechanism 1: PARP Enzyme Inhibition Mechanism 2: PARP Trapping Cell death in DNA damage repair deficient cells (e.g. BRCA-mutation) DNA Damage (Single-Strand) DNA Damage (Double-Strand) Trapping of PARP on DNA Enhanced cell death in any tumor cell with single-strand DNA damage Talazoparib PARP Enzyme |
 Mechanism 1: Inhibition of PARP Enzyme
PARP Inhibitors Prevent DNA Damage Repair Leading to Tumor Cell Kill
8 Cancer cells replicate frequently and make many errors in DNA that require repair for survival PARP is the major DNA repair enzyme Tumors have many DNA repair defects, such as BRCA-mutations and other HRD (homologous recombination deficiency) Tumors with HRD struggle to repair and replicate DNA When HRD is combined with PARP inhibition, DNA repair is so compromised that tumors can no longer survive Source: Sonnenblick et al. Nature
Reviews Clinical Oncology 2015; 12:
27–41. |
 9 Source: Shen et al. Clin Cancer Res 2013; 19:5003–15. Note: PARP inhibition represents relative concentration for 50% inhibition in PARP1 enzyme assay.
Most Potent PARP Enzyme Inhibitor (~3x–8x) Relative Potency for PARP Enzyme Inhibition Mechanism 1: Talazoparib is the Most Potent Inhibitor of PARP Enzyme in Clinical Development All PARP inhibitors inhibit PARP enzyme… |
 10 Source: Shen et al. Clin Cancer Res 2013; 19:5003–15. Note: PARP inhibition represents relative concentration for 50% inhibition in PARP1 enzyme assay; antitumor activity represents relative
concentration for 50% Capan-1 cell survival reduction in
single-agent cytotoxicity assay. Most Potent
PARP Enzyme Inhibitor (~3x–8x) Most Potent Antitumor Activity (~50x–2,000x+) Relative Potency for PARP Enzyme Inhibition Relative Potency for Cytotoxicity If Talazoparib is 3X – 8X More Potent than Other PARP Inhibitors in PARP Inhibition, Why is it 2,000X Better at Killing Cancer Cells in Vitro? All PARP inhibitors inhibit PARP enzyme… …but potency of PARP enzyme inhibition does not correlate with antitumor activity… |
 And
Why is Talazoparib Orders of Magnitude More Potent than Olaparib in
Cancer Xenograft Models? BRCA-Mutated Breast Cancer Xenograft Mouse
Model 11
Talazoparib 0.1 mg/kg
Talazoparib 0.33 mg/kg
Olaparib 100 mg/kg Vehicle Source: Shen et al. Clin Cancer Res 2013; 19:5003–15.
Note: MX-1 tumor xenograft model. MX-1 is a human mammary carcinoma cell
line that harbors BRCA1 deletion events and is BRCA1 deficient. |
 Because PARP Enzyme Inhibition is Not Key: Olaparib Maximally Inhibits
PARP Enzyme Even at 40 mg, a Dose That is Clinically
Ineffective Current Olaparib Clinical Dose is 300-400mg
BID 12 Source: Fong et al. NEJM 2009; 361: 123-134. Maximum PARP enzyme inhibition evidenced by virtual elimination of PAR in human tumors |
 Because PARP Enzyme Inhibition is Not Key: Rucaparib Maximally Inhibits
PARP Enzyme Even at 92 mg, a Dose That is Clinically
Ineffective Current Rucaparib Clinical Dose is 600mg BID
13 Phase 2 multi-center trial investigating intermittent and continuous dosing schedules of rucaparib in germline BRCA- mutation carriers with advanced ovarian and breast cancer. Scatter plot showing % PARP enzyme inhibition levels relative to pretreatment levels in patients’ peripheral blood lymphocytes following treatment with rucaparib. Samples were analyzed using a validated assay that uses quantitative immunologic detection of ex vivo poly(ADPribose) formation. Source: Drew et al. Br J Cancer. 2016;114:723-30. |
 Source: Shen et al. Clin Cancer Res 2013;
19:5003–15. Note: PARP inhibition represents relative concentration
for 50% inhibition in PARP1 enzyme assay; antitumor activity represents relative concentration for 50% Capan-1 cell survival reduction in single-agent cytotoxicity assay; and temozolomide potentiation represents the relative
concentration that, when combined with 200 µmol/L of temozolomide,
resulted in 50% growth inhibition of LoVo cells in a temozolomide potentiation assay. All PARP inhibitors inhibit PARP enzyme… …but potency of PARP enzyme inhibition does not correlate with antitumor activity… …while PARP trapping ability is better correlated to antitumor activity Most Potent PARP Enzyme Inhibition (~3x–8x) Most Potent Antitumor Activity (~50x–2,000x+) Most Effective PARP Trapping (~50x–2,000x) Relative Potency for PARP Enzyme Inhibition Relative Potency for Cytotoxicity Relative Potentiation of Chemotherapy (Temozolomide) Because Cancer Cell Cytotoxicity Correlates Better with PARP Trapping than PARP Enzyme Inhibition 14 |
 Mechanism 2: Talazoparib, When Bound to PARP, Traps
PARP Protein on Single-Strand DNA Breaks
15 PARP PARP PARP PARP Temozolomide Radiation
Single-Strand DNA Breaks
PARP Trapping Enhanced PARP-dependent cell killing Talazoparib Alkylation chemotherapies and radiation create single-strand DNA breaks which become sites for PARP trapping Source: Medivation. |
 Publications Support PARP Trapping as a Better Correlate
with Tumor Cell Killing than PARP Inhibition
PARP Inhibitors Differ Markedly in PARP Trapping Ability
16 “Trapped PARP–DNA complexes were more cytotoxic than…PARP inactivation” “a new mechanistic foundation for the rational application of PARP inhibitors in cancer therapy” “potency in trapping PARP differed markedly among inhibitors…, a pattern not correlated with the catalytic inhibitory properties for each drug.” “[Talazoparib] is the most potent clinical PARP inhibitor tested to date with the highest efficiency at trapping PARP-DNA complexes” Source: Murai et al. Cancer Res 2012, 72(21) 5588-99; and Murai et al.
Molecular Cancer Ther 2014, 13 (2): 433–443. |
 Talazoparib is the Most Potent and Niraparib is the Second
Most Potent PARP Trapper in Clinical Development
Slide 20, Yves Pommier, 13th International Congress on Targeted Anticancer Therapies,
March 2015 17
Source: Pommier,13th International Congress on Targeted Anticancer Therapies (TAT
2015), Paris, March 2015, Slide 20. |
 Talazoparib is the Most Potent and Niraparib is the Second
Most Potent PARP Trapper in Clinical Development
Source: Murai et al, Cancer Res 2012; Murai et
al, Molecular Cancer Ther 2014.
18 Niraparib is 2x more potent at PARP trapping than Olaparib Talazoparib is 100x more potent at PARP trapping than Olaparib By inference, Talazoparib is 50 fold more potent at PARP trapping than Niraparib |
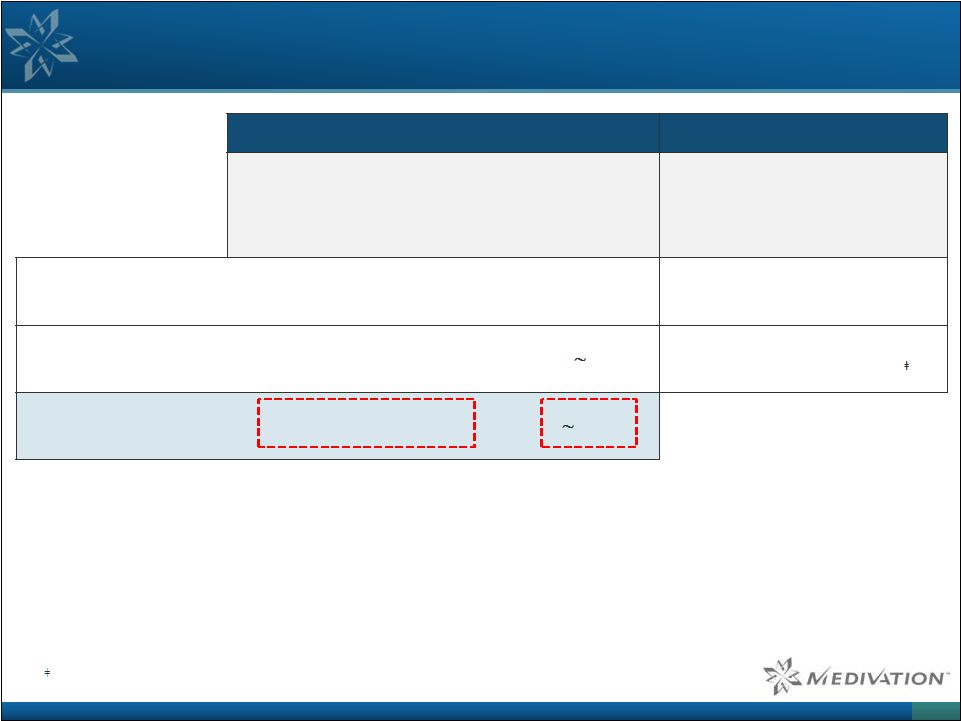 PARP
Trapping Potency Appears to Correlate Better with Clinical Outcomes than
PARP Enzyme Inhibition Potency PARP Assays
1 Ovarian PFS 2 PARP-1 Enzyme Inhibition (IC50, nM) Cytotoxicity (IC90, nM) Relative PARP Trapping Activity BRCA- Mutated Patients Non-BRCA Mutated Patients Olaparib 6 4500 1 11.2 vs. 4.3 months 7.4 vs. 5.5 months Niraparib 60 2300 2 21.0 vs 5.5 months † 9.3 vs 3.9 months Talazoparib 4 40 100 1 Data from: Pommier,13th International Congress on Targeted Anticancer Therapies (TAT 2015), Paris, March 2015.
Cytotoxicity with DT40 cells.
2 Data from: Ledermann et al, Lancet Oncol. 2014 Jul;15(8):852-61.
Company filings and press releases.
† Only includes
gBRCA-mutated
patients. Only excludes gBRCA-mutated patients.
PFS = progression-free survival.
19 PARP trapping also appears to be correlated with better responses beyond BRCA and HRD Lower IC50s & IC90s mean higher potency 50-100x better than Niraparib and Olaparib at PARP trapping |
 The
Multi-Log Superiority of Talazoparib Over Olaparib in PARP Trapping
Correlates with a Multi-Log Superiority in NON-HRD Cell Kill*
20 (1) Cosmic database, Wellcome Trust Sanger Institute; CCLE database, Broad Institute.
*These cell lines have NO detectable mutations in the most common HRD genes:
ATM, ATR, BRCA1, BRCA2, FANCA, FANCD2, MRE11A, NBN, PALB21
Prostate cancer PC-3 cells
Ovarian cancer OvCAR3 cells |
 Niraparib, a More Potent PARP Trapper than Olaparib,
Resulted in Longer PFS than Olaparib
21 NIRAPARIB (NOVA) OLAPARIB Sponsor Tesaro AstraZeneca Trial Phase 3 ovarian cancer trial Phase 2 ovarian cancer trial Setting Maintenance following response to platinum in recurrent ovarian cancer Maintenance following response to platinum in recurrent ovarian cancer Design Placebo-controlled N>500 (target 490) 2:1 randomization Two independent cohorts based on gBRCA status Placebo-controlled N=265 1:1 randomization One cohort including patients with BRCA mutation Dosing 300 mg QD (3 pills per day) 400 mg BID (16 pills per day) Primary Endpoint PFS PFS Drug vs. Placebo PFS by Patient Subgroup gBRCA: 21.0 vs 5.5
months g/sBRCA:
11.2 vs 4.3 months non-gBRCA: 9.3 vs 3.9 months non-g/sBRCA: 7.4 vs 5.5 months non-gBRCA HRD+: 12.9 vs 3.8
months Source: Ledermann et al,
Lancet Oncol. 2014 Jul;15(8):852-61. Company filings and press releases. Note: gBRCA = germline BRCA mutation; sBRCA = somatic BRCA mutation; PFS = progression-free survival
|
 22 Single-Strand DNA Breakers (e.g., Low-Dose Chemo) Can FURTHER Amplify Talazoparib’s Superior PARP Trapping Potentially Registrational Studies to Evaluate Combination to Begin by YE 2016 Source: Medivation. Note: NCI-H209 SCLC xenograft model. Talazoparib and temozolomide were administered orally, once daily for 4 days x 3 as indicated. Temozolomide dose was 3mg/kg and talazoparib dose was 0.25 mg/kg. Vehicle Temozolomide Talazoparib Talazoparib + Temozolomide |
 Single-Strand DNA Breakers (e.g., Low-Dose Radiation) Can
FURTHER Amplify Talazoparib’s Superior PARP Trapping
23 Source: Wong et al. Potent anti-tumor activity of talazoparib (BMN673) in combination with radiation for squamous cell carcinoma of the head and neck. AACR 2015. Note: DMSO: vehicle; IR only: radiation only: Enhancement of Head and Neck Cancer Cell Kill with Talazoparib in Combination with Low-Dose Radiation |
 Talazoparib is the Most Specific PARP Inhibitor for PARP1
and PARP2 in Clinical Development
PARP1 and PARP2 are the PARP Isoforms Used Most by Cancers
24 Source: Poster presented by Herwig Schuler at the 2014 PARP meeting. Wahlberg et al. Nature Biotech 2012;
30: 283-289. Specificity: Talazoparib > Veliparib > Olaparib
> Rucaparib Olaparib
Veliparib Rucaparib |
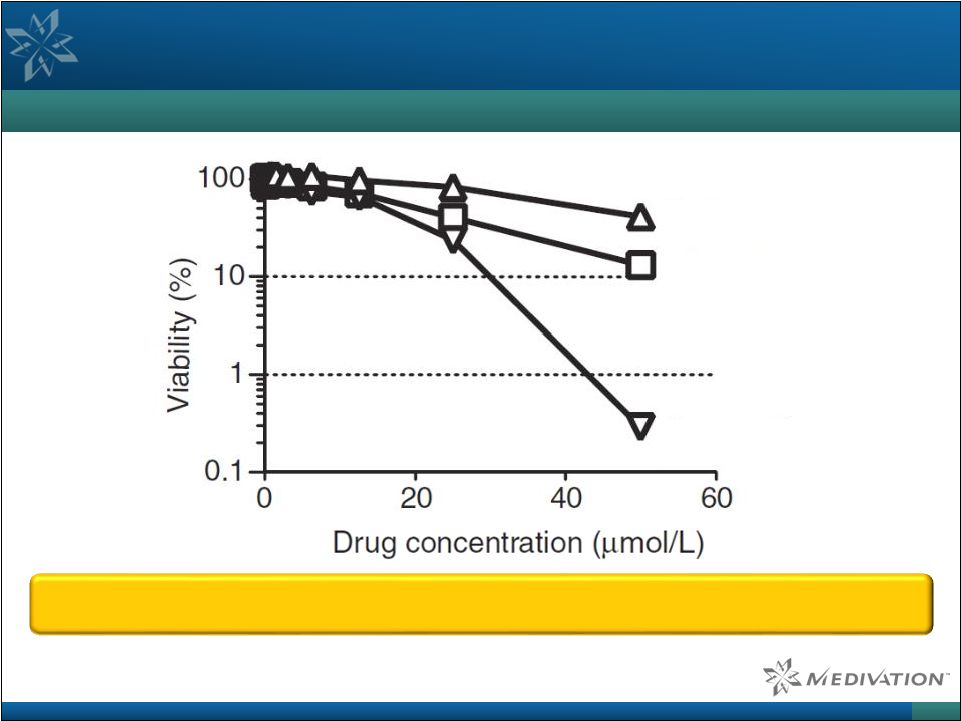 Talazoparib is a Highly Specific PARP Inhibitor and has
Lower Off-Target Cell Toxicity
Cell Viability Assay in PARP1/2 Deficient Cells
25 Source: Murai et al. Mol Cancer Ther 2014, 13 (2): 433–443. Note: DT40 PARP1-/- avian B lymphoblast cells used are equivalent to PARP1 and PARP2 double knockout cells and do not have detectable levels
of PARP activity. Talazoparib has less effect on PARP1 and PARP2 deficient cells than other PARP inhibitors in
clinical development, showcasing greatly reduced off-target toxicity
Talazoparib Olaparib Rucaparib |
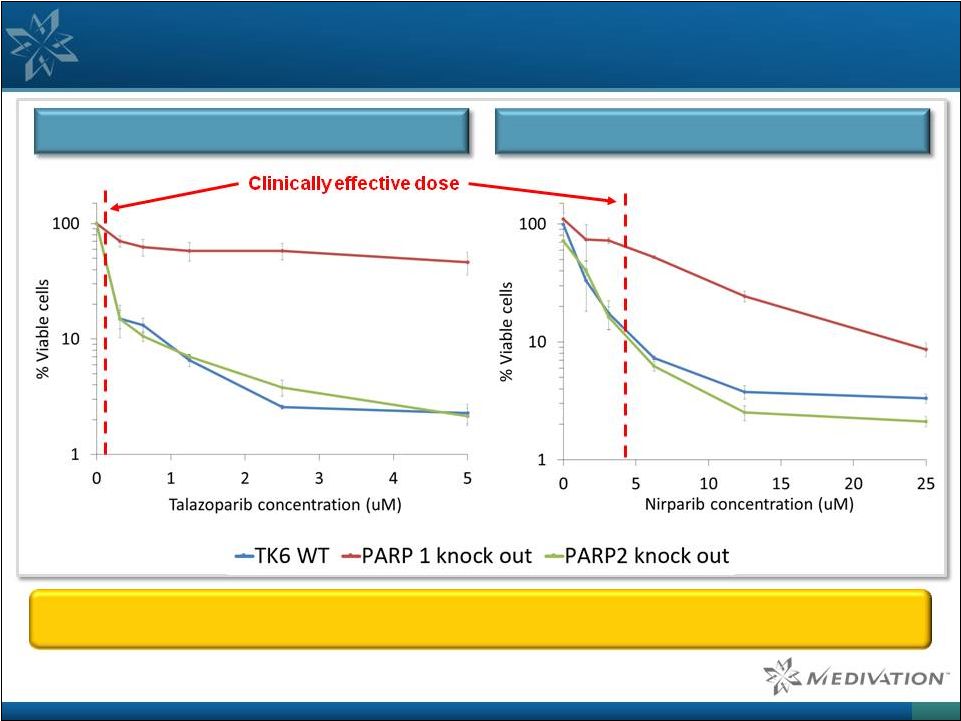 Talazoparib’s Cell Killing Effects are More PARP-Specific
than Niraparib in TK6 Human Lymphoblastoid Cells
26 TALAZOPARIB NIRAPARIB A cell line lacking PARP1 suffers little toxicity from Talazoparib at clinically effective doses
Source: Pommier lab, NCI, unpublished data. |
 Third-Party Comparison of Adverse Event Profile of
Talazoparib vs. Other PARP Inhibitors
Talazoparib: >10% pts
Niraparib: >10% pts
Olaparib: >15% pts
Rucaparib: >15% pts
Note: Compiled by BioMarin. Source: Wainberg et al. ASCO 2014 (1mg patients),
Kaufman et al. ASCO 2013 (400mg patients), Sandu et al. Lancet Oncology 2013 (290-300mg patients), and Swisher et al. SGO 2015 (600mg patients). 12% 4% 5% 33% 30% 23% 25% 16% 10% 2% Anemia Thrombocytopenia Neutropenia Lymphopenia Nausea Vomiting Constipation Anorexia Diarrhea Dyspepsia Dysgeusia Fatigue Insomnia Alopecia ALT/AST Increased 0% 10% 20% 30% 40% 50% 60% 70% Talazoparib Gr 1–2 Talazoparib Gr 3–4 16% 59% 35% 21% 26% 17% 16% 53% 17% 2% 1% 6% Anemia Thrombocytopenia Neutropenia Lymphopenia Nausea Vomiting Constipation Anorexia Diarrhea Dyspepsia Dysgeusia Fatigue Insomnia Alopecia ALT/AST Increased 0% 10% 20% 30% 40% 50% 60% 70% 47% 25% 24% 15% 56% 25% 36% 27% 49% 13% 11% 15% 5% 2% 2% 2% 5% Anemia Thrombocytopenia Neutropenia Lymphopenia Nausea Vomiting Constipation Anorexia Diarrhea Dyspepsia Dysgeusia Fatigue Insomnia Alopecia ALT/AST Increased 0% 10% 20% 30% 40% 50% 60% 70% Niraparib Gr 1–2 Niraparib Gr 3–4 10% 59% 20% 28% 26% 16% 36% 43% 19% 4% 2% 1% 2% 1% 6% Anemia Thrombocytopenia Neutropenia Lymphopenia Nausea Vomiting Constipation Anorexia Diarrhea Dyspepsia Dysgeusia Fatigue Insomnia Alopecia ALT/AST Increased 0% 10% 20% 30% 40% 50% 60% 70% Rucaparib Gr 1–2 Rucaparib Gr 3–4 30% 7% Olaparib Gr 1–2 Olaparib Gr 3–4 27 |
    Clinical
Data and Development Plans |
 Talazoparib (MDV3800) Talazoparib Will Be Tested Across Multiple Solid Tumor Indications Current Talazoparib Clinical Development Plan 29 Phase 1 Phase 2 Phase 3 Breast Cancer (Germline BRCA-Mutation) (EMBRACA) Potential Addressable Patient Population 1 10K Reviewed with FDA Potentially registrational Prostate Cancer (Monotherapy, Genetically-selected) Select optimal regimen for Phase 3 Prostate Cancer (Monotherapy & Low-dose Combo Chemo, All-comers) 140K Small Cell Lung Cancer (Monotherapy & Low-dose Combo Chemo, All-comers) Reviewed with FDA Registrational 80K Select optimal regimen for Phase 3 Breast Cancer (Monotherapy & Low-Dose Combo Chemo, All-comers) 140K Potentially registrational Ovarian Cancer (PARP-failures & TBD) 100K Note: Light blue shading indicates trials expected to be initiated in 2016. Further
clinical development plans under review. Last updated July 6, 2016. 1 Addressable patient population defined as all lines of treatable metastatic patients in U.S. and Europe in 2016. Source: © 2016 DR/Decision
Resources, LLC. All rights reserved. Reproduction,
distribution, transmission or publication is prohibited. Reprinted with permission. |
       Talazoparib: The Leading Breast Cancer PARPi Program
30 Note: gBRCA = germline BRCA; BRCAm = BRCA-mutated; ORR = objective response rate; CBR = clinical benefit response
RATIONALE
DESIGN
OUR
OPPORTUNITY
EMBRACA BREAST CANCER BEYOND gBRCA High ORR / CBR in phase 1 in gBRCA-mutated patients, as monotherapy Phase 3 randomized (2:1) trial vs. physician’s choice in gBRCA-mutated locally advanced and/or metastatic breast cancer Powered to show overall survival (OS) benefit Potentially superior PFS prolongation relative to other PARP inhibitors Largest BRCAm breast cancer trial, potentially best-powered to show an OS advantage Preclinical data supporting activity in non-gBRCA- mutated patients Multi-arm Phase 2 study, testing talazoparib monotherapy and in combination with low-dose chemotherapy, with the primary endpoint of PFS Study in a broad breast cancer population (non- genetically selected) Only combination with chemotherapy study in this indication |
 Talazoparib was Studied in a Phase 1 Trial as a
Monotherapy in BRCA-Mutated Cancer Patients
Evaluated 46 Germline BRCA-Mutated Breast and Ovarian Cancer Patients
31 Note: Asterisk indicates Talazoparib. Safety and Antitumor Activity of the PARP inhibitor BMN 673 *
in a Phase 1 Trial Recruiting Metastatic Small-Cell Lung Cancer (SCLC) and Germline BRCA
Mutation Carrier Cancer
Patients
1 David Geffen School of Medicine at University of California, Los Angeles, CA, 2 Institute of Cancer Research, Royal Marsden NHS Foundation Trust, Sutton, United Kingdom, 3 Virginia G. Piper Cancer Center at Scottsdale Healthcare/TGen, Scottsdale, AZ, 4 Indiana University, Indianapolis, IN, 5 University of Texas MD Anderson Cancer Center, Houston, TX, 6 University of Michigan, Ann Arbor, MI, 7 BioMarin Pharmaceutical, Novato, CA Zev A. Wainberg 1 , Saeed Rafii 2 , Ramesh K. Ramanathan 3 , Lida A. Mina 4 , Lauren Averett Byers 5 , Rashmi Chugh 6 , Jonathan W. Goldman 1 , Jasgit Sachdev 3 , Daniela Matei 4 , Jennifer J. Wheler 5 , Joshua W. Henshaw 7 , Charlie Zhang 7 , Gilles J.A. Gallant 7 , Johann S. de Bono 2 |
 Talazoparib Demonstrated Compelling Activity in BRCA-
Mutated Breast Cancer Patients
RECIST Response in Germline BRCA-Mutated Breast Cancer
32 At the 1 mg/day Phase 3 dose: 7/14 had an objective response (ORR = 50%)
12/14 had a clinical benefit response (CBR =
86%) Source: Wainberg et al. ASCO 2014.
Note: CR = complete response, PR = partial response, SD = stable disease and PD =
progressive disease. Genetic Status
N CR PR SD <24 weeks or PD 7 11 0 1 2 5 1 1 4 4 Deleterious BRCA1 mutation Deleterious BRCA2 mutation Total 18 1 (6%) 7 (39%) 5 (28%) RECIST Response 5 (28%) SD 24 weeks |
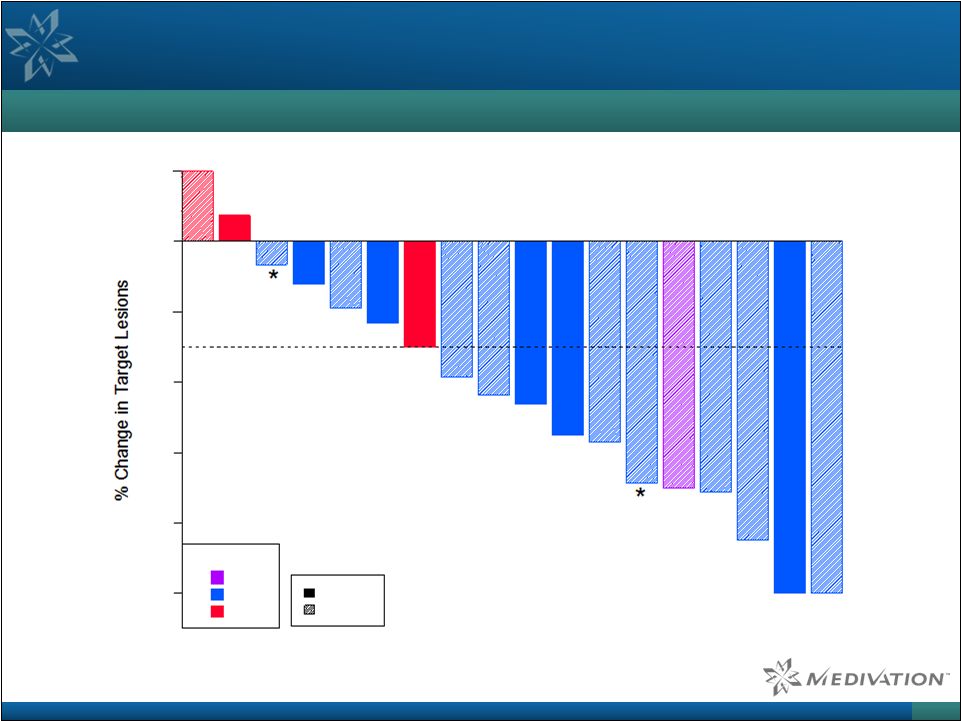 Kaplan-Meier Estimates:
Median duration of response: 32.0 weeks
95% C.I.: 19.9 –
40.3 weeks Median progression-free survival: 32.1 weeks 95% C.I. 13.1 – 43.4 weeks Talazoparib Demonstrated Strong Objective Responses in BRCA-Mutated Breast Cancer Patients BRCA-Mutated Breast Cancer RECIST Response Waterfall Plot 33 Source: Wainberg et al. ASCO 2014. Note: Asterisks indicate treatment ongoing at the time of data cutoff (April 18, 2014).
-100 -80 -60 -40 -20 0 20 Dose (mg/day) 0.9 1.0 1.1 BRCA 1 BRCA 2 |
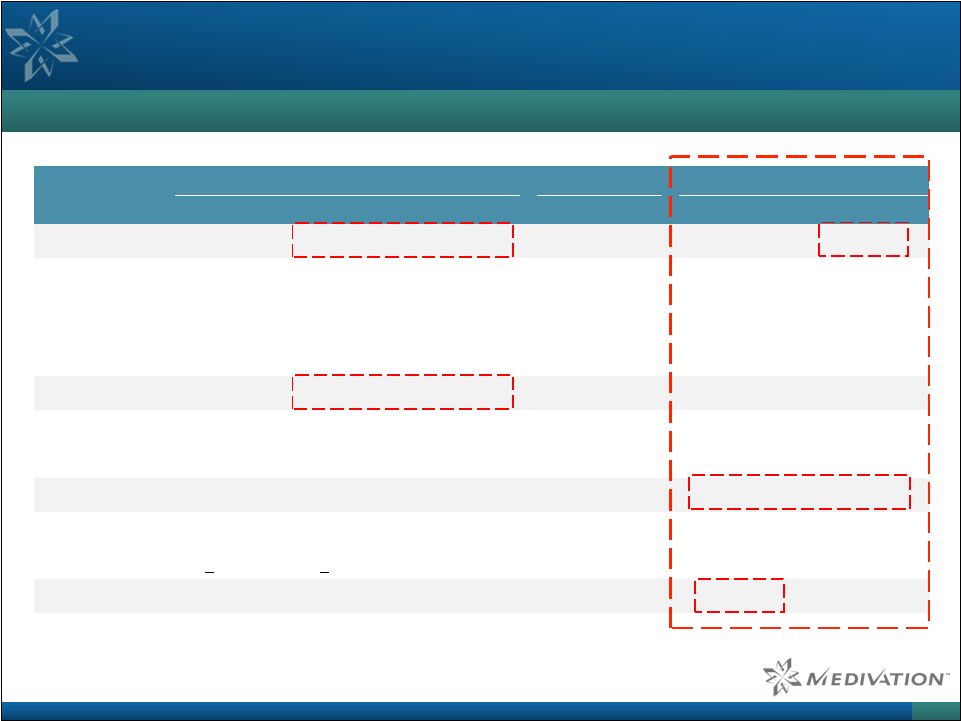 Third-Party Analysis of PARP Inhibitors in BRCAm Breast
Cancer Supports Talazoparib’s Best-In-Class Potential
Comparison of Single-Agent Activity in BRCA-Mutated Breast Cancer
34 Source: Barclays Research; Kaye et al. J Clin Oncol 2012 Feb 1; 30 (4): 372; company reports. Olaparib Niraparib Talazoparib Phase 2 Breast POC Phase 2 Phase 1/2 Phase 1 Dosing 100mg BID 400mg BID 400mg BID Various 0.9–1.1mg QD 1mg QD Size, n 27 27 62 4 18 14 Prior Regimens, median 3 3 4.6 5 3 Platinum 22% 30% 33% ORR, % 22% 41% 13% 50% 44% 50% CR – 4% – 6% 7% PR 22% 37% 13% 50% 39% 43% CBR, % 67% 85% 60% 72% 86% SD 44% 44% 47% 28% 36% (>23wks) (>23wks) (>8wks) (>24wks) (>24wks) PFS (months) 3.8 5.7 8.0 |
 EMBRACA: Talazoparib Phase 3 Study in Germline BRCA-
Mutated Metastatic Breast Cancer is Ongoing
Note: Enrollment Initiated in October 2013.
Compare PFS of patients treated with monotherapy
talazoparib relative to those treated with protocol-specified
physician’s choice single-agent chemotherapy
Up to 430 women with gBRCA-mutated locally advanced
and/or metastatic breast cancer
Powered for hazard ratio = 0.67
Two-arm study: Talazoparib 1 mg per day vs protocol-
specified physician’s choice of chemotherapy
Primary endpoint: Progression-free survival (PFS)
Radiographic progression determined by blinded independent
central radiology review
35 GOAL |
 Phase
3 EMBRACA is Similar to Competitors’ Trials, but More Conservatively
Designed and Higher-Powered Comparison of Phase 3 BRCA-Mutated
Breast Cancer Trial Designs 36
BRAVO (Niraparib) OlympiAD (Olaparib) EMBRACA (Talazoparib) Sponsor Tesaro AstraZeneca Medivation Phase 3 Dose Same as Phase 2 Dose? Yes No Yes Clinical Dose 300mg QD 300mg BID 1mg QD Target Hazard Ratio (control / active) 0.5 NA 0.67 Target Enrollment 306 310 429 Accrual Enrolling through 2016 Enrollment complete Complete enrollment in 2016 Option to read out early Reduce target number of events Source: Medivation, company filings and presentations, ClinicalTrials.gov, and Balmana et al. SABC 2015 Poster OT1-03-05.
|
 Talazoparib is Being Studied in a Phase 2 Trial for the
Treatment of Breast Cancer Beyond BRCA-Mutation
37 Source: Medivation and ISPYTrials.org. I-SPY 2 trial sponsored by QuantumLeap Healthcare Collaborative. Talazoparib arm of Phase 2 trial activated in May 2016.
"The I-SPY 2 TRIAL is designed to rapidly test promising agents to reduce the cost, time and number of patients needed to bring new therapies to breast cancer patients who urgently need new treatment options.” “We are very excited to initiate a new trial arm of talazoparib that is designed to optimize our understanding of this investigational drug and avoid some of the complications of paclitaxel, which is part of the current standard treatment." Laura J. Esserman, M.D. Principal Investigator of I-SPY 2 I-SPY 2 Trial Talazoparib + Irinotecan in Neoadjuvant Breast Cancer GOAL Compare pCR rates in women with early breast
cancer when treated in the neoadjuvant setting with
either talazoparib + irinotecan or paclitaxel followed by
standard chemotherapy Neoadjuvant setting in patients with newly-diagnosed, locally-advanced breast cancer Up to 75 patients with HER-2 negative breast cancer will be enrolled in the talazoparib arm Talazoparib arm: 1 mg talazoparib once-daily + low-dose irinotecan followed by standard chemotherapy Talazoparib arm activated in May 2016 Control arm: paclitaxel followed by standard chemotherapy Primary endpoint: Pathologic complete response Therapies found effective in I-SPY 2 can move onto a more focused Phase 3 registration trial (I-SPY 3) |
 Talazoparib: The Leading Prostate Cancer PARPi Program
MONO-
THERAPY STUDY Single-arm study in post- NHT / post-chemotherapy mCRPC patients Patients selected for DNA repair defects Alignment on trial design with FDA reached, potential for accelerated approval CHEMO- COMBINATION STUDY Monotherapy and in combination with low-dose chemotherapy in mCRPC non-genetically defined patients Phase 2 trial with data in 2017 DESIGN Potential to be the first PARPi approved for prostate cancer Leverages existing development and commercial expertise and capabilities OUR OPPORTUNITY Study in a broad prostate cancer population (non- genetically selected) Compelling response rates were observed with a PARPi in heavily pre- treated mCRPC patients Johnson & Johnson in- licensing of a PARPi for prostate cancer provides further validation (up to $500m in payments plus royalties) Strong synergistic effect with temozolomide demonstrated in preclinical prostate cancer model RATIONALE 38 Note: mCRPC = metastatic castration-resistant prostate cancer; NHT = novel hormonal therapy. |
 Investigator-Initiated Study of a PARPi in Advanced Prostate Cancer
39 Source: Mateo et al. N Engl J Med 2015 Oct 29; 373(18):1697-708. Study: A Phase 2 Trial of Olaparib in Patients With Advanced Castration Resistant Prostate Cancer (TOPARP) Sponsor: Institute of Cancer Research, United Kingdom Collaborator: Royal Marsden NHS Foundation Trust Principal Investigator: Johann de Bono Enrollment: 50 patients Data Presentation: AACR 2015 Publication: NEJM October 2015 FDA Breakthrough Therapy: Granted in January 2016 The TOPARP Study Demonstrated the Potential of PARP Inhibitors in the Treatment of Prostate Cancer |
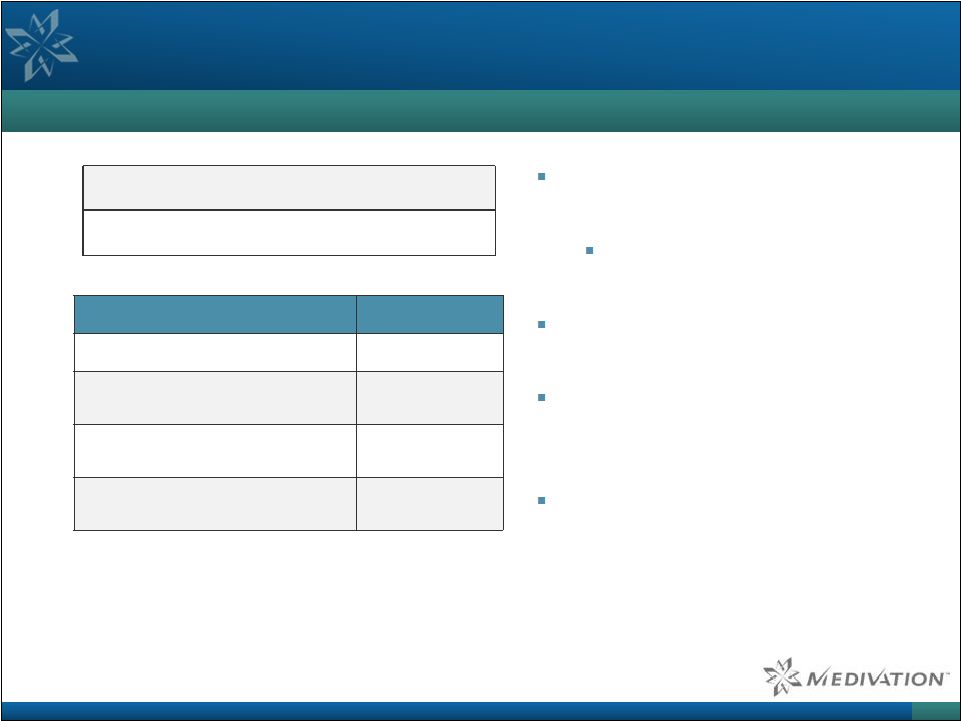 Data
from TOPARP Study with PARPi in Advanced Prostate Cancer Patients
dosed 50 Evaluable for response 49 Prior lines of treatment n (%) Docetaxel 50 (100%) Cabazitaxel 29 (58%) Zytiga (abiraterone) 48 (96%) Xtandi (enzalutamide) 14 (28%) 40 Heavily pre-treated patient population Prior Zytiga and/or Xtandi 49/50 (98%) Response rate: 33% (16/49 patients) 13/16 responders dropped circulating tumor cells (CTCs) to zero Median duration of treatment in responders was 40 weeks Striking Effects were Observed in Heavily Pre-treated, Advanced Metastatic Prostate Cancer Patients Source: Mateo et al. N Engl J Med 2015 Oct 29; 373(18):1697-708.
|
 Strong Activity Occurred in Most Patients with DNA Repair Defects
41 16/49 (33%) of patients were identified as having DNA-repair defects Of these 16 patients, 14 patients had a response (88%) All 8 patients with a BRCA1 or BRCA2 loss had a response PARP Inhibition Even More Effective in Patients with HRD in TOPARP Source: Mateo et al. N Engl J Med 2015 Oct 29; 373(18):1697-708. |
 Radiologic Progression-Free Survival: Biomarker+ vs. Biomarker-
42 PARP Inhibition Even More Effective in Patients with HRD in TOPARP Hazard Ratio: 0.24 (95% CI, 0.11 – 0.50) Source: Mateo et al. N Engl J Med 2015 Oct 29; 373(18):1697-708. |
 Selected Talazoparib Preclinical Prostate Cancer Data
43 Talazoparib’s Potency and PARP Trapping Ability Could Make It Best in Class for the Treatment of Prostate Cancer Source: Medivation, unpublished data. Note: Monotherapy study in LNCaP cells, which are patient-derived prostate cancer cell line with pathogenic mutations in DNA repair genes
including ATM, CHEK2, ERCC2, ERCC3, FANCA, and HDAC2.
Talazoparib Appears Significantly More Potent than Olaparib
as a Monotherapy in Prostate Cancer Cells |
 Low-Dose Temozolomide Potentiates the Anti-tumor Effects of
Sub-Maximal Talazoparib Doses in Prostate Cancer Cells
Talazoparib Single-Agent
Low-Dose Talazoparib with Temozolomide
Concentration tested for temozolomide potentiation
Source: Medivation, unpublished data.
Note: Temozolomide combination study in PC-3 prostate cancer cells.
44 |
 Talazoparib: The Leading Small Cell Lung Cancer (SCLC)
PARPi Program 1L MAINTENANCE STUDY Phase 3 maintenance study after first-line chemotherapy Monotherapy and in combination with low-dose temozolomide Alignment with FDA reached DESIGN Potentially first registrational study in a broad lung cancer population (all-comers, non-genetically selected) OUR OPPORTUNITY 10% ORR 1 observed with talazoparib as monotherapy in phase 1 study Significant synergy observed between talazoparib and low-dose temozolomide in preclinical models Temozolomide is used in SCLC PR observed in IST evaluating talazoparib + irinotecan in SCLC patient RATIONALE 45 1 Calculated as the ORR of evaluable patients. Source: Wainberg et al. ASCO 2014, and Medivation. Note: ORR = objective response rate; PR = partial response; IST = investigator-sponsored trial |
 46 Talazoparib Demonstrated Single-Agent Activity in Patients with Advanced Previously-Treated Small Cell Lung Cancer Source: Wainberg et al. ASCO 2014, and Medivation. -75 -50 -25 0 25 50 75 Dose (mg/day) 1.0 Small Cell Lung Cancer RECIST Response Waterfall Plot Kaplan-Meier Estimates: Median duration of response: 13.6 weeks 95% C.I.: 12.0 – 15.3 weeks Median progression-free survival: 11.1 weeks 95% C.I. 4.3 – 13.0 weeks |
 Talazoparib’s PARP Trapping Activity Synergizes Effectively
with Low-Dose Temozolomide in SCLC Animal Model
47 Vehicle Temozolomide Talazoparib Talazoparib + Temozolomide Source: Medivation. Note: NCI-H209 SCLC xenograft model. Talazoparib and temozolomide were administered orally, once daily for 4 days x 3 as
indicated. Temozolomide dose was 3mg/kg and talazoparib dose
was 0.25 mg/kg. |
 Talazoparib: Exploiting PARP Trapping in Ovarian Cancer
PARP- FAILURE & PARP- SENSITIVE STUDY Potentially registrational study with multiple arms testing monotherapy and combination low-dose chemotherapy in both PARP-sensitive and PARP-failure patients DESIGN Potentially first registrational study to show activity in PARP- failures Potentially rapid path to approval OUR OPPORTUNITY 48% ORR observed in heavily treated patients in Phase 1 (n=12) 57% ORR (4/7) observed in non-gBRCA ovarian patients in IST (+ low-dose chemotherapy) 2 ovarian olaparib failures experienced 6+ months SD (ASCO,
2016) Responses observed in
both platinum-sensitive
and platinum-resistant
patients RATIONALE 48 Source: Wainberg et al. Oral presentation, AACR 2016, and Medivation. Note: ORR = objective response rate; SD = stable disease; IST = investigator-sponsored trial. |
 Talazoparib Demonstrated Compelling Activity in BRCA-
Mutated Ovarian Cancer Patients
RECIST Response in Germline BRCA-Mutated Ovarian Cancer
49 Clinical benefit response rate (CR, PR, SD 24 weeks): 23/28 (CBR = 82%) Source: Wainberg et al. ASCO 2014. Note: ORR = objective response rate, CR = complete response, PR = partial response, SD = stable disease and PD = progressive
disease. (1)
25 of 28 patients measurable by RECIST.
(2) 27 of 28 patients evaluable for CA-125. Deleterious Mutation N Response CR/PR SD > 24 weeks SD < 24 weeks/PD RECIST (1) CA-125 (2) BRCA1 20 8 13 4 3 BRCA2 8 4 6 0 2 Total 28* 12 (48% ORR) 19 (70% ORR) 4 (14%) 5 (18%) |
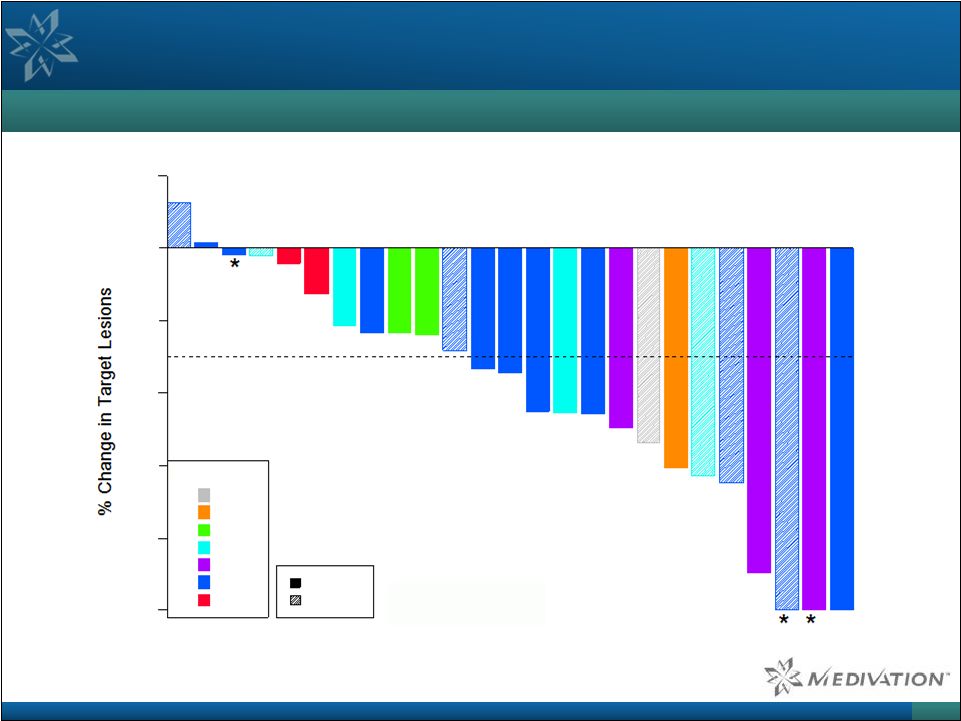 Talazoparib Demonstrated Compelling Activity in BRCA-
Mutated Ovarian Cancer Patients (continued) BRCA-Mutated Ovarian Cancer RECIST Response Waterfall Plot 50 Source: Wainberg et al. ASCO 2014. Note: Asterisks indicate treatment ongoing at the time of data cutoff (April 18, 2014).
Kaplan-Meier Estimates:
Median duration of response: 26.9 weeks
95% C.I.: 15.9 – 28.1 weeks Median progression-free survival: 32.9 weeks 95% C.I. 28.1 – 40.4 weeks -100 -80 -60 -40 -20 0 20 Dose (mg/day) 0.1 0.2 0.4 0.6 0.9 1.0 1.1 BRCA 1 BRCA 2 |
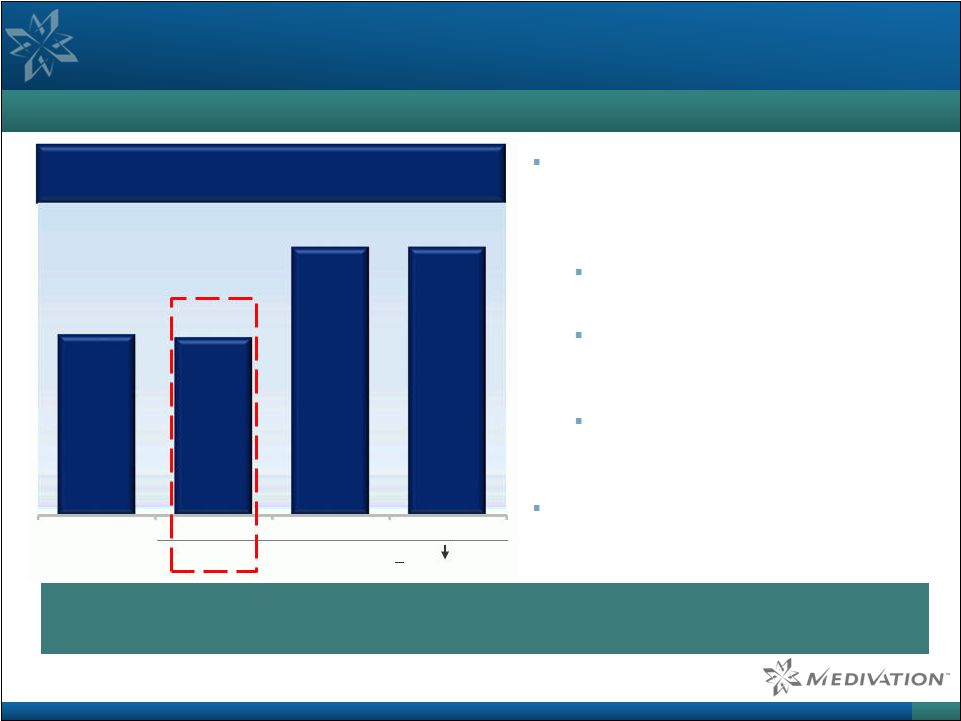 Talazoparib Plus Low-Dose Chemotherapy Clinical Data (AACR 2016)
51 Superior PARP Trapping May Lead to Improved Clinical Outcomes in Broad Patient Populations as Seen in Ovarian Cancer Phase 1 data from a 40 patient study of talazoparib in combination with low-dose chemotherapy in heavily pre-treated advanced malignancies 4/7 objective responses in non- BRCA mutated ovarian cancer patients 1 responder did not even have HRD suggesting possibilities of treating patients without genetic selection Additional responses in Ewing’s sarcoma, cervical adenocarcinoma, SCLC and TNBC Olaparib was approved based on
34% objective response rate (ORR) in BRCA- mutated ovarian cancer Talazoparib is active in tumors with defects in DNA repair genes beyond BRCA-mutations
(larger market than BRCA) and possibly even without HRD (largest market)
All (CBR) Ovarian Cancer, non-BRCA-mutated (ORR) (CBR) (>50% CA-125) Source: Wainberg et al. Oral presentation, AACR 2016, and FDA product label for olaparib.
Note: Chemotherapy regimens included temozolomide and irinotecan. HRD =
homologous recombination deficiency and CBR = clinical benefit rate. Phase
1 Response Data (talazoparib with low-dose chemotherapy)
58% 57% 86% 86% |
 Talazoparib Investigator-Sponsored Trials
52 Talazoparib is Being Further Explored Across Indications through Several Investigator-Sponsored Studies Investigator Institution Phase Indication Other Wainberg UCLA Phase 1 Advanced solid tumors Combination with irinotecan or temozolomide Piha-Paul MD Anderson Phase 2 Advanced cancers Somatic BRCA-mutations and deletions HRD positive PTEN deficiency gBRCAm (not breast or ovarian) Munster UCSF Phase 1 Triple negative breast cancer Combination with carboplatin Triple negative breast cancer with BRCAness Other tumors with BRCAm Chen NCI Pilot Advanced solid tumors Deleterious BRCAm Lee NCI Phase 2 Advanced solid tumors Deleterious BRCAm Prior PARP inhibitor treatment Litton MD Anderson Pilot Neoadjuvant breast cancer Invasive breast cancer with deleterious BRCAm Federico St. Jude Phase 1 Pediatric solid tumors Combination with irinotecan +/- temozolomide Telli Stanford Phase 2 Triple negative breast cancer HER2-negative breast cancer HRD positive Wisinski University of Wisconsin Phase 1 Advanced solid tumors Combination with carboplatin and paclitaxel Schafer Baylor Phase 1/2 Pediatric malignancies Combination with temozolomide Esserman Multisite Phase 2 Neoadjuvant breast cancer Combination with irinotecan (I-SPY 2) Owonikoko Multisite Phase 2/3 Non-small cell lung cancer 2nd-line, HRD positive patients (LUNG MAP) Note: Includes collaborative/cooperative-group studies (I-SPY 2 and LUNG MAP). HRD = homologous recombination deficiency. |
 Multiple Indications to Pursue as Monotherapy or Combination Therapy
53 Talazoparib Holds the Potential to Address Multiple Tumor Types and a Large Commercial Opportunity Potential Addressable Patient Population (U.S. and Europe) Phase Potential Launch 2022-23 2022-23 P2 ready P3 ready 2020-21 P2/3 ready 2019-22 P3 2018 ~760k P2 ready 2021-22 P2 ready 2022-23 Addressable Market (U.S. and Europe) Talazoparib Opportunity >$30bn Note: Addressable patient population defined as all lines of treatable metastatic patients in 2016. Dollar figures reflect current PARP inhibitor pricing. Source: Medivation; Peshkin et al. Breast Dis 2010; TCGA, Nature 2012; Mateo et al. N Engl J Med 2015; and TCGA Nature. 2011; and © 2016 DR/Decision Resources, LLC. All rights reserved. Reproduction, distribution, transmission or publication is
prohibited. Reprinted with permission. Breast
(gBRCAm) Prostate SCLC Ovarian P2 ready NSCLC 10k 140k 80k 100k 140k 60k 230k Current Patient Opportunity Breast (Beyond BRCAm) Glioblastoma |
