Attached files
| file | filename |
|---|---|
| EX-23.1 - EXHIBIT 23.1 - QUEST DIAGNOSTICS INC | dgx12312015ex231.htm |
| EX-10.13 - EXHIBIT 10.13 - QUEST DIAGNOSTICS INC | dgx12312015ex1013.htm |
| EX-10.11 - EXHIBIT 10.11 - QUEST DIAGNOSTICS INC | dgx12312015ex1011.htm |
| EX-10.3 - EXHIBIT 10.3 - QUEST DIAGNOSTICS INC | dgx12312015ex103.htm |
| EX-31.2 - EXHIBIT 31.2 - QUEST DIAGNOSTICS INC | dgx12312015ex312.htm |
| EX-32.1 - EXHIBIT 32.1 - QUEST DIAGNOSTICS INC | dgx12312015ex321.htm |
| EX-32.2 - EXHIBIT 32.2 - QUEST DIAGNOSTICS INC | dgx12312015ex322.htm |
| EX-31.1 - EXHIBIT 31.1 - QUEST DIAGNOSTICS INC | dgx12312015ex311.htm |
| EX-21.1 - EXHIBIT 21.1 - QUEST DIAGNOSTICS INC | dgx12312015ex211.htm |
| EX-10.15 - EXHIBIT 10.15 - QUEST DIAGNOSTICS INC | dgx12312015ex1015.htm |
| EX-10.12 - EXHIBIT 10.12 - QUEST DIAGNOSTICS INC | dgx12312015ex1012.htm |
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 10-K
Annual Report Pursuant to Section 13 or 15(d) of
the Securities Exchange Act of 1934
For the Fiscal Year Ended December 31, 2015
Commission File Number 001-12215
Quest Diagnostics Incorporated
3 Giralda Farms
Madison, New Jersey 07940
(973) 520-2700
Delaware
(State of Incorporation)
16-1387862
(I.R.S. Employer Identification Number)
Securities registered pursuant to Section 12(b) of the Act: | |
Title of Each Class | Name of Each Exchange on Which Registered |
Common Stock, $.01 par value per share | New York Stock Exchange |
Securities registered pursuant to Section 12(g) of the Act: | None |
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes X No
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act.
Yes No X
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Yes X No
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files).
Yes X No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. [ ]
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer X Accelerated filer Non-accelerated filer (do not check if a smaller reporting company)
Smaller reporting company
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes No X
As of June 30, 2015, the aggregate market value of the approximately 143 million shares of voting and non-voting common equity held by non-affiliates of the registrant was approximately $10.4 billion, based on the closing price on such date of the registrant's Common Stock on the New York Stock Exchange.
As of January 31, 2016, there were outstanding 142,758,584 shares of the registrant’s common stock, $.01 par value.
Documents Incorporated by Reference | Part of Form 10-K into which incorporated |
Document | |
Portions of the registrant's Proxy Statement to be filed by April 29, 2016 | Part III |
Such Proxy Statement, except for the portions thereof which have been specifically incorporated by reference, shall not be deemed “filed” as part of this report on Form 10-K.
TABLE OF CONTENTS
Item | Page | |
Item 1. | ||
Item 1A. | ||
Item 1B. | ||
Item 2. | ||
Item 3. | ||
Item 4. | ||
Item 5. | ||
Item 6. | ||
Item 7. | ||
Item 7A. | ||
Item 8. | ||
Item 9. | ||
Item 9A. | ||
Item 9B. | ||
Item 10. | ||
Item 11. | ||
Item 12. | ||
Item 13. | ||
Item 14. | ||
Item 15. | ||
i
Item 1. Business
Quest Diagnostics Incorporated is the world's leading provider of diagnostic information services. We empower people to take action to improve health outcomes. Derived from the world's largest database of clinical lab results, our diagnostic insights reveal new avenues to identify and treat disease, inspire healthy behaviors and improve health care management. In the right hands and with the right context, our diagnostic insights can inspire actions that transform lives.
Quest Diagnostics was incorporated in Delaware in 1990; its predecessor companies date back to 1967. We conduct business through our headquarters in Madison, New Jersey, and our laboratories, patient service centers, offices and other facilities around the United States and in selected locations outside the United States. Unless the context otherwise requires, the terms “Quest Diagnostics,” the “Company,” “we” and “our” mean Quest Diagnostics Incorporated and its consolidated subsidiaries.
During 2015, we generated net revenues of $7.5 billion. Additional financial information concerning Quest Diagnostics, including our consolidated subsidiaries and businesses, for each of the years ended December 31, 2015, 2014 and 2013 is included in the consolidated financial statements and notes thereto in “Financial Statements and Supplementary Data” in Part II, Item 8.
The discussion below includes several tables. The index below is a guide to those tables.
Index to Tables |
Table 1 - Vision, Goals and Values |
Table 2 - Five Point Business Strategy |
Table 3 - Restore Growth |
Table 4 - Clinical Franchises |
Table 5 - Key Professional Lab Services Offerings |
Table 6 - Invigorate Cost Excellence Program |
Table 7 - Invigorate Cost Excellence Program - Savings |
Table 8 - Acquisition Highlights |
Table 9 - Positioned to Grow and Lead |
Table 10 - Assets and Capabilities |
Table 11 - New or Enhanced Disease Area Solutions |
Table 12 - Sample Collaborations |
Table 13 - 2015 Scientific and Medical |
Table 14 - 2015 QuanumTM Health Information Technology Solutions Introduced |
Table 15 - 2015 Net Revenues |
Table 16 - Sample Esoteric Test Fields |
Table 17 - U.S. Clinical Testing industry |
Table 18 - Key Trends |
Table 19 - Customers |
Table 20 - Sample Third Party Payers |
Table 21 - Factors Traditionally Considered When Selecting a Diagnostic Information Services Provider |
Table 22 - 2015 Medicare and Medicaid Revenues |
Table 23 - Key Regulatory Schemes |
Table 24 - Information Available at Our Corporate Governance Webpage |
Table 25 - Executive Officers |
1
OUR STRATEGY AND STRENGTHS
In 2012, Quest Diagnostics launched a new vision, goals and strategy.
Table 1 - Vision, Goals and Values | |
Vision | Empowering Better Health with Diagnostic Insights |
Three Aspirational Goals | A healthier world Build a valuable company Create an inspiring workplace |
Values | Quality Integrity Accountability Innovation Collaboration Leadership |
Our Strategy
In November 2012, we introduced a five-point business strategy to achieve our vision and our goals.
Table 2 - Five Point Business Strategy |
1. Restore growth |
2. Drive operational excellence |
3. Simplify the organization |
4. Refocus on diagnostic information services |
5. Deliver disciplined capital deployment |
Since the end of 2013, restoring growth has been our top priority. At our Investor Day in November 2014, we shared progress on our strategy and commitments, reaffirmed our strategy and outlined our path forward. In 2015, we continued to execute on this strategy, consistent with our stated path forward. The discussion below focuses on our five-point strategy and our path forward.
1. Restore growth. We are pursuing seven opportunities to restore growth. Three opportunities have a near-term focus; four are longer-term.
Table 3 - Restore Growth | |
Near term focus | Sales and marketing excellence |
Grow esoteric testing through a disease focus | |
Provide professional lab services to hospitals and IDNs | |
Longer-term focus | Lead in precision medicine |
Create value from information assets | |
Leverage capabilities into extended care settings | |
Succeed internationally | |
In 2013, we launched a multi-year initiative, Project Restore, to identify, prioritize, resource and implement a wide range of activities designed to create consistent, profitable growth. The program leverages center-led planning, analytics, project management and best practice teams to improve sales and marketing effectiveness, reduce attrition, launch new solutions, enhance key account relationships, instill pricing discipline, develop impactful marketing and bring lessons learned to local teams across the enterprise. The investments that we have made and our efforts to restore growth have yielded positive results: we have restored growth. Our goal is to achieve and ultimately exceed market growth rates.
2
Our efforts to foster sales and marketing excellence have progressed. We maintain one commercial organization in our Diagnostic Information Services business, centrally led and focused on local customer needs. We employ world-class management discipline around processes, tools and measurement. We are investing in talent, providing industry-leading training and development, focusing on physician specialty opportunities, and fostering a customer-focused, performance-driven culture.
We plan to grow esoteric testing revenues by creating value through scientific and product innovation and delivering comprehensive solutions for major clinical opportunities. We are more than just a laboratory; we seek to offer solutions using data information services and strategies that enable our customers to deliver the most effective healthcare to the right populations and individuals. Starting with a clinical focus on a specific disease state or clinical problem, we pursue opportunities to create value by providing holistic solutions centered on evidence-supported standards of care, and to combine routine, guideline mandated testing with esoteric solutions. We take advantage of advanced technology for more precise, comprehensive and actionable information, and integrate our extensive clinical data to help manage populations and target health care solutions.
Our clinical franchise organizations, working with our research and development team, focus on these opportunities and coordinate with our commercial organization to deliver new and improved solutions. Our franchises are designed to enable us to act like a boutique service provider while maintaining the advantages of our scale, and identify and tap growing market segments so that we more wisely deploy our resources and target opportunities.
Table 4 - Clinical Franchises | |
Cardiovascular, Metabolic and Endocrinology | Oncology |
General Health and Wellness | Prescription Drug Monitoring and Toxicology |
Infectious Diseases and Immunology | Sports Science and Human Performance |
Neurology | Women’s and Reproductive Health |
Our 2015 introduction of companion and complementary test services for new anti-PD-1 therapies for metastatic non-small cell lung cancer, the 2015 launch of our LeukoVantageTM solution for myeloid neoplasms and the continued growth of our prescription drug monitoring and infectious diseases and immunology businesses are recent examples of the power of our clinical franchises to deliver new solutions and grow organically.
We also plan to grow by pursuing strategic partnerships with hospitals and independent delivery networks ("IDNs"). We believe that continued price transparency, cost and utilization pressure, and evolving healthcare payment models will drive demand for our expertise. Our key professional laboratory services offerings are highlighted in Table 5 below. We have announced seven new professional lab services relationships over the past 24 months, including most recently our relationship with Barnabas Health, the largest health care system in New Jersey.
Table 5 - Key Professional Lab Services Offerings | |
Lab management outsourcing | Data diagnostics, consolidation and insights |
Joint venture | Reference testing |
Outreach acquisition | Supply chain management |
Test menu management | Programs enabling effective patient care management |
2. Drive operational excellence. We strive to enhance operational excellence and improve our quality and efficiency across every portion of our value chain, from the time that we receive an order until the time we receive payment, including in our supporting operations. Improving our operations will yield many benefits, including: enhancing customer satisfaction, employee engagement and shareholder value; improving our quality and competitiveness; and strengthening our foundation for growth.
We have made strong progress driving operational excellence, improving our quality and efficiency and improving our overall customer experience. For example, in 2015 increased adoption of our customer self-service tools reduced demand for
3
transactional phone calls. We also deployed new automation and technology platforms in revenue services, laboratory operations and logistics services, improving the customer experience and reducing labor costs.
Our cost excellence program, Invigorate, includes structured plans to drive savings and improve performance across the end-to-end value chain. We believe that many of these efforts also strengthen our foundation for growth. For example, in 2015 we improved reagent utilization and enhanced our approach to service and maintenance, in each case improving efficiency and reducing cost.
Table 6 - Invigorate Cost Excellence Program | |
Flagship Programs | |
Revenue services | Organizational design |
Information technology | Procurement |
Laboratory | Field and customer service |
Additional Key Opportunities Identified in November 2014 | |
Standardize our processes, information technology systems and business platforms | |
Delivering services electronically | |
Enhancing cash collection | |
The following chart provides information regarding Invigorate program savings.
Table 7 - Invigorate Cost Excellence Program - Savings | |
2012 Goal for run-rate savings exiting 2014 | $600 Million |
2013 Revised goal for run-rate savings exiting 2014 | $700 Million |
2014 Year-end run-rate savings | Over $700 million |
2014 Goal for additional run-rate savings exiting 2017 | $600 million (cumulative savings total: $1.3 billion) |
2015 Realized savings | Over $200 million |
3. Simplify and strengthen the organization to enable growth and productivity. Starting in 2012, we revised our senior management team and restructured our organization to eliminate silos in our core business, provide leadership in defined geographies and eliminate three unnecessary management layers. Our organization is designed to align around future growth opportunities, to coordinate upstream and downstream units in our business for seamless execution and to leverage our company-wide infrastructure to gain more capability, value and efficiency. We continue to enhance our structure and capabilities to support our strategy.
We adopted the Quest Management System to manage our Company. This system provides a foundation for day-to-day management, and includes best-in-class business performance tools to help us develop new capabilities to improve our Company. The system, which enables us to run the Company with a common language, approach and philosophy, supports our efforts as we build a high-performance culture, with employees focused on behaviors to make us more agile, transparent, customer-focused, collaborative and performance oriented.
In 2015, we launched our new brand - Action from Insight - recommitting to a superior customer experience. We also introduced our Everyday Excellence program, which includes guiding principles to support a superior customer experience, to inspire our employees to be their best every day, with every person and with every customer interaction. We continue to simplify and strengthen the organization, and our processes, to better focus on our customers, speed decision-making and to empower employees.
4. Refocus on diagnostic information services. We have a sharp focus on diagnostic information services. In 2015, we contributed our business of central laboratory testing for clinical trials to a joint venture, Q2 Solutions. Since 2012, we also have sold our OralDNA salivary diagnostics business, our HemoCue and Enterix diagnostic products businesses and ibrutinib royalty rights, generating approximately $800 million of proceeds.
5. Deliver disciplined capital deployment and strategically aligned accretive acquisitions. We are focused on increasing shareholder returns and returns on invested capital (“ROIC”) through a framework that encompasses improving operating performance and disciplined capital deployment.
4
Our disciplined capital deployment framework includes dividends, share repurchases and investment in our business and is intended to improve ROIC. The framework is grounded in maintaining an investment grade credit rating. We expect to return a majority of our free cash flow to investors through a combination of dividends and share repurchases. In 2015, we returned 80% of our free cash flow to investors.
Consistent with that expectation, in January 2016 we announced that we increased our quarterly common stock dividend by 5%, from $0.38 per common share to $0.40 per common share. This represents our fifth increase in the dividend since 2011. We also believe that opportunities may arise to return incremental capital to shareholders from free cash flow as a result of portfolio actions. In December 2015, we announced a $500 million increase in repurchase authority under our common stock repurchase program. Since the beginning of 2012, we have returned more than $1.5 billion to stockholders through repurchases of our common stock.
We will continue to invest in our business in a disciplined manner. We have established a solid foundation of strategic assets and capabilities. We expect to generate 1 to 2 percent revenue growth per year through value-creating, strategically-aligned acquisitions using disciplined investment criteria. We screen potential acquisitions using guidelines that assess strategic fit and financial considerations, including value creation, ROIC and impact on our earnings.
Table 8 - Acquisition Highlights | |
2015 Closed Acquisitions | • MemorialCare Health System outreach laboratory testing business |
• Superior Mobile Medics, a national provider of paramedical and health data collection services to the life insurance and employer health and wellness industries | |
2015 Announced Acquisitions | • Clinical Laboratory Partners (subsidiary of Hartford HealthCare) outreach laboratory testing business |
Our additional near-term investments in growth are likely to focus on investments in innovation in the form of licensing, collaborations and internal development to grow esoteric testing, tools to support commercial excellence and Project Restore. We also expect to make investments to improve operational excellence, with systems standardization and automation, footprint optimization and our Invigorate cost excellence program as potential examples.
Our Strengths
We offer high value diagnostic information services and diagnostic solutions, including those grounded in pathology and gene-based and esoteric testing, that are attractive to our customers (discussed under the heading Customers on page 18). We believe that our customers prefer providers that offer a comprehensive and innovative range of tests and services and convenient access to those services. We believe that, by offering such services, we strengthen our market offering, market position and reputation. Table 9 summarizes our strengths, which are discussed in greater detail below.
Table 9 - Positioned to Grow and Continue to Lead | |
• Unmatched size, scale and capabilities | • High value, low cost provider experience |
• Leader in providing innovative solutions and diagnostic insights | • Strong focus on quality and providing a superior customer experience |
• Strong collaborator | • Medical and scientific expertise |
Our assets and capabilities. We are the world's leading provider of diagnostic information services. We estimate that we have access to more than 20 billion patient data points from tests over the past decade. We are the leading provider in the United States of routine and gene-based and esoteric testing services, including anatomic pathology. We serve approximately one-third of the adult population of the United States annually, and approximately one-half of the adult population in the United States over a three-year period. We estimate that annually we serve approximately half of the physicians and half of the hospitals in the United States.
5
Table 10 - Assets and Capabilities | |
• Provide healthcare connectivity solutions to >250,000 clinician and hospital accounts | • 157 million test requisitions processed in 2015 |
• Strong logistics capabilities • make > 7,500 stops daily • approximately 3,500 courier vehicles • > 25 aircraft serving the U.S. | • One of the largest medical and scientific staffs in the industry to provide interpretive consultation • > 650 M.D.s and Ph.D.s, many of whom are recognized leaders in their field • genetic counselors |
• Mobile teams to support different business initiatives • paramedical examiners • health and wellness professionals, including nursing professionals | • National access to patient testing, with most extensive network in the U.S.: • >2,300 of our own patient service centers • >4,200 phlebotomists in physician offices |
• Industry-leading test menu | • Access to >20 billion patient data points from test results delivered over past decade |
Innovation. We are a leading innovator in diagnostic information services. We continue to introduce new tests and services, including many with a focus on personalized and targeted medicine. Our capabilities include discovery, technology development and clinical validation of diagnostic tests. We develop tests at our laboratories, such as Quest Diagnostics Nichols Institute and Athena Diagnostics.
We successfully transfer technical innovations to the market through our in-house expertise and our relationships with technology developers, including the academic community, pharmaceutical and biotechnology firms, emerging medical technology companies and others that develop and commercialize novel diagnostics, pharmaceutical and device technologies. We search for new opportunities and continue to build a robust pipeline of new solutions. Through our strengths in assay development and the commercialization of test services, we believe that we are the partner of choice for developers of new technologies and tests to introduce their products to the marketplace.
We seek innovations and solutions that help healthcare providers care for their patients through better testing for predisposition, screening, monitoring, diagnosis, prognosis and treatment choices. We seek to develop innovations and solutions that help to determine a patient's genotype or gene expression profile relative to a particular disease and its potential therapies, because they can help healthcare providers to determine a patient's susceptibility to disease or to tailor medical care to an individual's needs - such as determining if a medication might be an optimum choice for a particular person, or tailoring the right dosage once the proper medicine is prescribed. In addition, we aim to develop holistic solutions responsive to challenges that healthcare providers and patients face, by developing solutions of multiple tests, information and services focused on specific clinical challenges, and taking advantage of the latest informatics capabilities. We also look for innovations and solutions that are less invasive than currently available options, to increase the choices that healthcare providers and patients have for the collection of diagnostic samples.
We have expertise with laboratory developed tests for companion and complementary diagnostics, and can offer an array of assets and services to support the development of companion diagnostics, including our robust collection of laboratory data and access to the company's patient service centers.
6
With these priorities in mind, during 2015 we introduced over 20 new or enhanced disease area solutions, including those discussed below.
Table 11 - New or Enhanced Disease Area Solutions | |
Cardiovascular, Metabolic and Endocrinology | We continued to expand our CardioIQ® advanced cardiovascular testing menu with the addition of MPO (myeloperoxidase), an inflammation marker. MPO complements the other inflammation markers that are part of the CardioIQ® suite, providing additional insights into disease activity in the artery wall. We introduced a new risk panel (an advanced lipid panel with inflammation) that combines 6 important biomarkers (lipid panel, lipid subfractionation by ion mobility, apoB, Lp(a), hsCRP and Lp-PLA2) to provide a more comprehensive risk assessment for heart attack and stroke. We developed and released a test for Zinc transporter 8 (ZnT8) Antibody. This test is primarily used for children and adolescents to confirm a diagnosis of type 1 diabetes. We developed and released a test for Intact Insulin by mass spectrometry. This test is useful for measuring fasting insulin levels and can be used to assess insulin resistance in diabetic patients. |
General Health and Wellness | We made our innovative, proprietary Wellness Engine application available to our customers. The Wellness Engine provides access to our wellness screening, reporting and incentive management solutions and offers: • Greater customization of program elements to meet the needs of diverse populations; • Simpler, intuitive participant experience that promotes greater engagement; and • Fully integrated data, regardless of collection site or type, to provide more powerful, actionable insights. |
Infectious Diseases and Immunology | We launched automatic performance of molecular testing on all patient specimens whose antibody screening results indicate hepatitis C virus infection, and removed standalone antibody screening as a test option. This change eliminated the prospect that a patient may receive a positive screening result but fail to undergo additional molecular testing, as recommended by medical guidelines, to help confirm a diagnosis of active hepatitis C infection, the cause of chronic hepatitis C. We developed and introduced a test for HIV-1 RNA, Quantitative Real-Time PCR with reflex to Coreceptor Tropism. This test may be used to determine whether a patient's viral HIV-1 load is sufficient to perform the HIV-1 RNA tropism test. We developed the Helicobacter pylori, Culture with Reflex to Susceptibility assay. Helicobacter pylori has been associated with peptic ulcer disease and cancers of the gastrointestinal tract. We developed a rapid identification test for Leptospirois, an infection caused by a waterborne spirochete of the genus Leptospira. |
7
Neurology | We introduced the first personalized genetic diagnostic test of the entire human genome in partnership with Personalis, Inc., to specifically help diagnose neurological disorders. We launched a series of new advanced sequencing molecular tests to more reliably and quickly diagnose hundreds of clinically relevant genetic causes of muscular dystrophies and myopathies in adults and children. These tests help avoid painful muscle biopsies that, in the past, were the only means to diagnose patients with muscle weakness. We launched advanced sequencing molecular tests to test for hundreds of genetic causes of rare, progressive movement disorders called hereditary ataxias, which affect movement of the legs and hands, and speech and eye movements. Patients benefit from a molecular diagnosis by eliminating the need for further testing and identifying a basis for therapies to extend mobility and life. We also launched advanced sequencing molecular tests to test for hundreds of genetic causes of rare, progressive movement disorders called hereditary spastic paraplegias, which are characterized by a slowly progressive stiffness and weakness of the legs. The diagnosis guides physicians in the use of medications to reduce muscle tightness. |
Oncology | We introduced LeukoVantageTM, a genomic test service that aids in the diagnosis, prognosis monitoring and selection of treatment for myeloid neoplasms, a group of hematologic malignancies that include acute myeloid leukemia. We introduced access to a companion test service for a new anti-PD-1 therapy for metastatic non-small cell lung cancer developed by Merck and a complementary test service for a new anti-PD-1 therapy for metastatic non-small cell lung cancer developed by Bristol-Myers Squibb. We introduced a prognostic panel including new probes to aid physicians in treating patients with plasma cell myeloma. |
Prescription Drug Monitoring and Toxicology | We introduced a quantitative test for buprenorphine and naloxone. Patients treated with buprenorphine-naloxone co-formulation will demonstrate both naloxone and buprenorphine/norbuprenorphine drug analysis. Naloxone is not detected with buprenorphine-only use. |
Sports Science and Human Performance | The New York Football Giants began using Blueprint for AthletesTM as a major factor in the Giants' training and preparation for National Football League games. We conducted beta programs at select elite endurance events using the Blueprint for AthletesTM service to empower athletes to track their progress from the onset of training through the event. |
Women’s and Reproductive Health | We introduced QNatal AdvancedTM, a new noninvasive prenatal screening service for detecting chromosomal abnormalities in certain high-risk pregnancies. We enhanced our HPV offerings with five new tests. One assay is used in connection with cytology to assess the presence or absence of high risk HPV genotypes 16, 18 and/or 45. This information together with physician's assessment of cytology history, other risk factors and professional guidelines may be used to guide patient treatment. The second assay is used in conjunction with cytology to evaluate anal dysplasia. The other three are used in combination with cytology, cytology history and other risk factors in aiding the diagnosis of vaginal cancer. |
Collaboration. We collaborate with partners that can help us to achieve our vision of empowering better health through diagnostic insights. Through our relationships, we believe that we are a leader in bringing innovation to the market. As the industry leader with the largest and broadest U.S. network and a presence outside the United States, we believe we are the distribution channel of choice for developers of new solutions, including large commercial manufacturers, academic medical centers and pharmaceutical and biotechnology firms, to introduce their products to the marketplace. We maintain relationships with advisers and consultants who are leaders in key fields of science and medicine. We work with key groups and organizations, including world class healthcare leaders, to foster important advances in healthcare, including in precision medicine. Some examples of our collaborations include:
8
Table 12 - Sample Collaborations | |
Collaborator | Collaboration |
Inovalon | Data DiagnosticsTM, a tool that provides real-time patient-specific data analysis that clinicians can order at the point of care to identify and address gaps in quality, risk, utilization and medical history insights. |
Inserm, the French National Institute of Health and Medical Research | BRCA ShareTM a novel data share initiative to provide scientists and laboratory organizations around the world with open access to BRCA1 and BRCA2 genetic data. |
Perinatal Quality Foundation | The national initiative to advance clinically appropriate noninvasive prenatal screening. |
University of California, San Francisco, the nation's leading university focused exclusively on health | To accelerate the translation of biomedical research into advanced diagnostics in the field of precision medicine. This collaboration has the overarching aim of enabling holistic and integrated diagnostic solutions that close gaps in care or enable new clinical value, with initial focus areas including autism, oncology, neurology and women's health. |
U.S. Centers for Disease Control and Prevention ("CDC") | To improve public health analysis of hepatitis C screening, diagnosis and treatment, based on analysis of our database of national hepatitis C virus ("HCV") diagnostic information. |
National Institutes of Health | We participate in studies they sponsor (e.g., NIH National Children Study). |
Memorial Sloan Kettering Cancer Center | Our OncoVantageTM solid tumor mutation testing services. |
Quintiles Transnational Holdings Inc. | Joint venture to provide central laboratory testing services for clinical trials. |
Medical and Scientific Expertise. Our medical and scientific experts publish research that demonstrates the clinical value and importance of diagnostic testing, including in connection with our research and development efforts. Our Quest Diagnostics Drug Testing IndexTM, which is a periodic report of trends derived from our aggregate drug testing results, is cited by employers, the federal government and the media to help identify and quantify drug abuse among the nation's workforce. The table below provides a further sample of the activities of our scientific and medical experts.
Table 13 - 2015 Medical and Scientific | |
Authored more than 150 publications, including approximately 100 articles in peer-reviewed journals | • Insights into diagnostic testing; introduce novel diagnostic approaches; provide latest thinking in lab testing and disease diagnosis - Addressed such topics as cervical cancer screening, ion mobility testing and cardiovascular disease, genetic testing for breast cancer risk and non-invasive prenatal testing. |
Authored textbooks or chapters | • Used by academic institutions to train healthcare providers |
Participated on scientific committees determining guidelines for diagnostic usage | • Fields include HIV, HCV and testosterone testing |
Published Quest Diagnostics Health TrendsTM reports | • Identify trends in disease and wellness. - Recent reports focused on prescription drug misuse, cervical disease and diabetes |
Health Information Technology Solutions. We have a history of providing leading information technology for diagnostic information services, including solutions that help healthcare organizations and clinicians enter, share and access clinical information without costly information technology implementation or significant workflow disruption, and technology solutions to enable patients to manage their healthcare and medical information. Examples of this include our Care360® products, national Care 360® healthcare provider network and MyQuest® by Care360® patient healthcare portal, which allows
9
patients to, among other things, use their smartphone or computer to receive and archive their Quest Diagnostics test results, manage their personal health information, find a Quest Diagnostics location and schedule appointments. We were the first national diagnostic information services provider to offer patient appointment scheduling and a patient mobile application.
We also have significant information assets, including many years of test result data. Our QuanumTM health information technology solutions leverage the power of our information assets, and our technology prowess, to help our customers empower better health through diagnostic insights. In 2015 we unveiled several new solutions, including:
Table 14 - 2015 QuanumTM Health Information Technology Solutions Introduced | |
Data DiagnosticsTM | A tool, introduced with Inovalon, that provides real-time patient-specific data analysis that clinicians can order at the point of care to identify and address gaps in quality, risk, utilization and medical history insights. |
Interactive InsightsTM by Care360® | A web-based reporting solution for clinicians and patients that provides health insights based on a patient's lab data to improve decision-making by both the clinician and the patient. |
IntelliTest AnalyticsTM | A web-based secure portal that provides hospitals, IDNs and clinician practices with data-driven insights about test utilization patterns and business analysis of clinically appropriate testing norms, payer type and costs. |
Care360 Revenue Cycle Management | A holistic medical billing solution that aids the management of billing claims with a focus on increasing patient and payer collections. The latest offering in the Care360 product portfolio. |
Quality. We strive to provide the highest quality possible and, to meet that goal, we have adopted the Quest Diagnostics Quality Program. This program includes policies and procedures that document, measure and monitor the effectiveness of our laboratory operations in providing and improving quality and meeting the requirements of the agencies that regulate the U.S. clinical laboratory testing industry.
We use the Quest Management System, including standard frameworks and methodologies for project and change management, to manage our Company. This system not only provides a foundation for day-to-day management, including best-in-class business performance tools, but also helps us to develop the capabilities that we need to manage the Company. We have a culture of continuous improvement. Employing root cause analysis, process improvements and rigorous tracking and measuring, we seek to enhance quality, continuously reduce defects, streamline processes, further increase the efficacy and efficiency of our operations and processes, eliminate waste and help standardize operations across our Company. We use Hoshin management principles in our efforts to achieve breakthrough performance. We use customer insights in our solutions development, listening to the voice of internal and external customers in all our business processes.
Customer focus. The customer is at the center of everything we do. Customers have a choice when it comes to selecting a healthcare provider and we strive to give them reason to put their trust in us. Focusing on a thorough understanding of customer needs and requirements, we seek to identify and implement processes that will result in a superior customer experience. We strive to provide a superior customer experience for our customers, because we believe that this will drive customer loyalty. In 2015, we launched our new brand - Action from Insight - recommitting to a superior customer experience. We also introduced our Everyday Excellence program, which includes guiding principles to support a superior customer experience, to inspire our employees to be their best every day, with every person and with every customer interaction.
BUSINESS OPERATIONS
The Company is made up of two businesses: Diagnostic Information Services and Diagnostic Solutions. The Company provides insights that empower and enable a broad range of customers, including patients, clinicians, hospitals, IDNs, health plans, employers and accountable care organizations ("ACOs").
10
Our Diagnostic Information Services business develops and delivers diagnostic testing information and services. The value creation side of the business is organized by clinical franchise and focuses on customer solutions for the marketplace, including new test development, diagnostic insights and product marketing. The value delivery side includes sales, marketing, routine and esoteric laboratory operations, field operations, logistics and client services. Diagnostic Solutions includes our risk assessment services, diagnostic products and healthcare information technology businesses and, prior to its July 1, 2015 contribution to our joint venture, Q2 Solutions, our business providing central laboratory testing for clinical trials.
Our Diagnostics Information Services business is the leading provider of diagnostic information services, which includes providing clinical testing services such as routine (including drugs-of-abuse) testing, gene-based and esoteric testing, and anatomic pathology services, as well as related services and insights. We offer the broadest access in the United States to diagnostic information services through our nationwide network of laboratories, Company-owned patient service centers, phlebotomists in physician offices. We provide interpretive consultation through one of the largest medical and scientific staffs in the industry.
In our Diagnostic Solutions group, we offer a variety of solutions for insurers and healthcare providers. We are the leading provider of risk assessment services for the life insurance industry. In addition, we offer healthcare organizations and clinicians robust health information technology solutions and diagnostic products.
We leverage our capabilities and assets to serve multiple customer bases. Most of our services are provided in the United States. For each of the years ended December 31, 2015, 2014 and 2013, we derived approximately 2% of our net revenues from foreign operations. For the years ended December 31, 2015, 2014 and 2013, approximately 1% of our long-lived assets were held outside the United States. The following table shows the percentage of our 2015 net revenues generated by the activities identified.
Table 15 - 2015 Net Revenues | |||
Activity | Approximate Percentage of 2015 Net Revenues | ||
Diagnostic information services | 93 | ||
Routine clinical testing services | 61 | ||
Gene-based, esoteric and anatomic pathology testing services | 32 | ||
Diagnostic Solutions: Healthcare information technology, clinical trials testing, risk assessment services and diagnostic products* | 7 | ||
*Includes clinical trials testing revenue until July 1, 2015, when we contributed our clinical trials business to Q2 Solutions.
Diagnostic Information Services
Background - clinical testing.
Clinical testing is an essential element in the delivery of healthcare services. Clinicians use clinical testing for predisposition, screening, monitoring, diagnosis, prognosis and treatment choices of diseases and other medical conditions. Clinical testing is generally categorized as clinical laboratory testing and anatomic pathology services.
Clinical laboratory testing generally is performed on whole blood, serum, plasma and other body fluids, such as urine, and specimens such as microbiology samples. Clinical laboratory tests which can be performed by most clinical laboratories are considered routine. Routine testing measures various important bodily health parameters such as the functions of the kidney, heart, liver, thyroid and other organs. Commonly ordered tests include blood chemistries, urinalysis, allergy tests and complete blood cell counts.
Esoteric tests are more advanced clinical laboratory tests that are not routine. Esoteric tests include procedures in the areas of molecular diagnostics (including next-generation sequencing), protein chemistry, cellular immunology and advanced microbiology. These tests may require professional “hands-on” attention from highly-skilled technical personnel, generally require more sophisticated technology, equipment or materials and may be performed less frequently than routine tests. Consequently, esoteric tests generally are reimbursed at higher levels than routine tests. It is not practical, from a cost-effectiveness or infrastructure perspective, for most hospitals, IDNs, ACOs, commercial laboratories or physician office
11
laboratories to develop and perform a broad menu of esoteric tests, or to perform low-volume esoteric testing in-house. Such tests generally are outsourced to an esoteric clinical testing laboratory, which specializes in performing these complex tests. Commonly ordered esoteric tests include viral and bacterial detection tests, drug therapy monitoring tests, gene-based tests, autoimmune panels and complex cancer evaluations. Esoteric tests increasingly are ordered by physicians to assist them in the diagnostic process, to establish a prognosis and to choose or monitor a therapeutic regimen.
Anatomic pathology services are performed on tissues, such as biopsies, and other samples, such as human cells. Anatomic pathology involves the diagnosis of cancer and other diseases and medical conditions through examination of tissue and cell samples taken from patients.
Our services.
We are the world's largest provider of diagnostic information services. We provide information and insights based on clinical testing, including routine, esoteric, gene-based and anatomic pathology testing, and related services. Increasingly, we are focused on providing solutions and insights to our customers, based on the testing that we perform and our extensive information and connectivity assets.
We are the leading provider of routine, esoteric and gene-based and anatomic pathology testing in the world, and offer customers the broadest access to the most extensive test menu. We are a leading provider of infectious disease diagnostic information services and strive to be the first to provide diagnostic solutions for emerging infectious diseases, including our Focus Diagnostics® offerings for West Nile Virus, SARS and Influenza A H1N1. We have leading positions in the neurology diagnostics market, in advanced cardiovascular diagnostic information services, including our CardioIQ® offering, and in cancer diagnostics, including the BRCAVantage®, OncoVantage® and ColoVantage® offerings. We are a leader in providing testing for the detection of employee use of drugs of abuse, offering a full range of solutions, including urine, hair, blood and oral fluid tests.
We also are a leading provider of biometric wellness screenings, flu shots and related preventative services that leverage clinical data to improve population health outcomes and reduce healthcare spend. We offer biometric wellness screenings to employer populations through our patient service centers and on-site through our extensive network of mobile examiners and nurses. In addition to a wide range of screening options, we also offer Blueprint for Wellness® and private label reporting, analytics and incentive management services. These services are sold directly to employers and through reseller partnerships with many health plans
We have significant information assets, including many years of test result data, and we are focused on ways to leverage the full power of these assets to help our customers. We also have a history of providing leading information technology for diagnostic information services. To help our customers empower better health through diagnostic insights by taking advantage of our information assets and technology prowess, we offer QuanumTM health information technology solutions. We believe that our QuanumTM solutions, including those introduced in 2015 and listed in Table 14 on page 10, promote our strategy, enhance the value we provide to our customers and will result in increased customer loyalty. We are working on health information technology solutions designed to:
• | enhance the customer experience, including ease of use and patient and provider engagement; |
• | deliver more precise, comprehensive solutions and actionable information; |
• | provide increased and interactive insights and analytics to patients and providers; |
• | foster greater adherence to clinical and reimbursement guidelines; |
• | promote population health solutions; |
• | tap the potential of large amounts of clinical information; and |
• | advance the development of precision medicine. |
We believe that offering services, solutions and insights based on a full range of tests and information assets will strengthen our market offering, market position and reputation. Our experienced medical staff has a passion for providing the highest quality service to our customers. Our in-house experts, including medical directors, scientific directors, genetic counselors and board certified geneticists, provide medical and scientific consultation regarding our tests and test results, and help clinicians and others best utilize these tests to improve patient outcomes and enhance patient satisfaction. Our approach fosters personalized patient care.
We have built advanced testing capabilities, including gene-based testing services for the predisposition, diagnosis, treatment and monitoring of cancers and other diseases. We provide integrated, comprehensive diagnostic information services
12
that include both anatomic pathology and clinical pathology testing, enabling our pathologists to offer patients and clinicians a complete analysis. We offer hundreds of esoteric tests, including but not limited to the following fields:
Table 16 - Sample Esoteric Test Fields | |
Field | Study/Focus Area |
Endocrinology and metabolism | Hormones and their effects on body growth and metabolism |
Genetics | Chromosomes, genes and their expression |
Hematology | Blood and bone marrow cells |
Coagulation | Process of blood clotting |
Neurology | Nervous system, its structure and its diseases |
Immunology | Immune system, including antibodies, cytokines, immune system cells and their effect, receptor systems and autoimmune diseases |
Microbiology and infectious diseases | Microscopic forms of life, including parasites, bacteria, viruses, fungi and other infectious agents |
Oncology | Abnormal cell growth, including pre-cancerous conditions and cancer |
Serology | Body fluids and their analysis, including antibodies, proteins and other characteristics |
Toxicology | Chemicals and drugs and their intended and adverse effects on the body |
We provide our services through our nationwide network of major laboratories, anatomic pathology laboratories and rapid response laboratories. Rapid response laboratories are smaller facilities where we can quickly perform an abbreviated menu of routine tests for customers that require rapid turnaround times. We conduct esoteric testing, including molecular diagnostics, in our world renowned Quest Diagnostics Nichols Institute laboratory facilities and in other facilities, including Focus Diagnostics and Athena Diagnostics. We operate 24 hours a day, 365 days a year. We also provide routine testing services, and inpatient anatomic pathology and medical director services, at hospital laboratories.
Our services primarily are provided under the Quest Diagnostics brand, but we also provide services under the AmeriPath,® Dermpath Diagnostics,® Focus Diagnostics,® Athena Diagnostics,® ExamOne,® Solstas® and Summit Health® brands.
International.
We provide diagnostic information services in multiple markets outside the United States. We have laboratory facilities in Gurgaon, India; Mexico City, Mexico; and San Juan, Puerto Rico. We see opportunities to bring our experience and expertise in diagnostic information services to markets outside the United States, including by leveraging existing facilities to serve new markets.
Diagnostic Solutions
Risk Assessment Services.
ExamOne is the largest provider of risk assessment services to the life insurance industry in North America. We also provide risk assessment services for insurance companies operating outside North America.
Our risk assessment services comprise underwriting support services, including data gathering, paramedical examinations and clinical laboratory testing and analytics, designed to assist life insurance companies objectively to evaluate the mortality risks of applicants. Most specimen collections and paramedical examinations are performed by our network of paramedical examiners at the applicant's home or workplace, but they also are offered at approximately 600 company patient service centers in the United States and approximately 120 additional locations in North America. We also contract with third parties to coordinate providing these exams at more than 350 additional locations outside North America. In 2015, ExamOne enhanced its service offering when it acquired the assets of Superior Mobile Medics, a national provider of paramedical and health data collection services to the life insurance, employer health and wellness industries.
13
Diagnostic Products.
Our diagnostic products business develops, manufactures and markets diagnostic products which can be used on a variety of instrument platforms that enable healthcare professionals to make healthcare diagnoses. We offer these products to a broad spectrum of customers in the United States and, through distributors, in other countries.
Healthcare Information Technology.
Our Care360® EHR product allows clinicians to generate a complete record of a clinical patient encounter, automates and streamlines the clinician's workflow, and allows for rapid deployment and implementation with minimal workflow disruption. The solution allows doctors to electronically create, manage and distribute patient encounter notes and allows for patient communication via a patient portal. It captures lab and radiology results, provides clinical decision support tools and allows doctors to send secure messages and clinical information to other practitioners and secure laboratory results to patients' personal health records.
ChartMaxx® is our enterprise content management system for hospitals. Clients have contracted for its use at over 250 sites in North America to enable clinical and business workflows.
Interest in Joint Venture
In 2015, we contributed our business of central laboratory testing for clinical trials to a joint venture, Q2 Solutions. Q2 Solutions is the second largest central laboratory services company in the world and provides services to customers across all segments of the biopharmaceutical industry. Clinical laboratory services are central to advances in genomics, precision medicine and drug development.
Non-Commercial, Development State Drug Asset
The Company has an interest in a non-commercial, development state drug asset: an agreement with Merck & Co., Inc. under which Merck has a license to our intellectual property for the development of, among other things, small molecule inhibitors of cathepsin K. We do not control the development activities conducted by Merck. Merck may not successfully develop or commercialize any compounds covered by the agreement, may not obtain needed regulatory approvals, and may not sell any products that would generate royalty payments to us, and we may not receive any further payments under this agreement. We are entitled to receive future milestone payments based on development progress for each potential product under the agreement. We are also entitled to receive single digit royalty payments from the sale of drugs, if any, resulting from the program. The obligation to pay royalties generally coincides with the life of the underlying patents. The Company is evaluating options with respect to this asset.
THE UNITED STATES CLINICAL TESTING INDUSTRY
The U.S. clinical testing industry consists of two segments. The following table discusses how we believe the industry is structured.
Table 17 - U.S. Clinical Testing Industry | ||
Testing | Approximate % of Total Industry | |
• Hospital inpatient and outpatient testing | 40% | |
• Testing of persons who are not hospital patients, including testing done in commercial clinical laboratories, physician-office laboratories and other locations, as well as hospital outreach (non-hospital patients) testing | 60% Consists of approximately: 35% - hospital-affiliated laboratories 53% - commercial clinical laboratories Balance - physician-office laboratories and other locations | |
14
Key Trends
The healthcare system in the United States is evolving; significant change is taking place in the system. We expect that the evolution of the healthcare industry will continue, and that industry change is likely to be extensive. There are a number of key trends that are having, and that we expect will continue to have, a significant impact on the diagnostic information services business in the United States and on our business. These trends present both opportunities and risks. However, because diagnostic information service is an essential healthcare service and because of the key trends discussed below, we believe that the industry will continue to grow over the long term and that we are well positioned to benefit from the long-term growth expected in the industry.
Table 18 - Key Trends | |
Demographics | As the population continues to grow and age, the burden of chronic diseases and unmet diagnostic needs may increase the demand for diagnostic information services. |
Prevention and wellness | We believe that the value of detection, prevention, wellness and personalized care now is well recognized. Consumers, employers, ACOs, IDNs, health plans and government agencies increasingly focus on helping the healthy stay healthy, detecting symptoms among those at risk and providing preventive insight and care that helps avoid disease. Health care providers increasingly rely on diagnostic information services to help identify risk for a disease, to detect the symptoms of disease earlier, to aid in the choice of therapeutic regimen, to monitor patient compliance and to evaluate treatment results. There is increased focus on a disease-oriented approach to diagnostics, treatment and management. Health care providers, consumers and payers increasingly recognize the value of diagnostic information services as a means to improve health and reduce the overall cost of healthcare through early detection, prevention and treatment. |
Medical innovation | Medical advances allow for more accurate and earlier diagnosis and treatment of diseases. Continuing advances in genomics and proteomics are expected to yield new, more sophisticated and specialized diagnostic tests. These advances also are spurring interest in and demand for precision medicine, which relies on diagnostic and prognostic testing and in which data information services and strategies are used to deliver the most effective healthcare to the right populations and individuals. Pharmacogenomic testing increasingly is used as a parameter to help speed drug approval processes and to better focus therapy based on patient and tumor-specific genetic markers. Demand also is growing toward comprehensive care management solutions that serve patients, payers and health care providers by improving clinical decision support, access to patient data, increasing patient participation in care management, population health management and improving clinical outcomes. There is increasing focus on access to patient data and data-driven insights. |
15
Customers and payers | Our customers and payers, including clinicians, health plans, IDNs, ACOs, employers and others, have been consolidating, converging and diversifying. For example, an increased number of hospital systems are considering establishing or have established health insurance plans, and health insurance plans increasingly are considering providing or are providing healthcare services. Consolidation is increasing pricing transparency and bargaining power, and encouraging internalization of clinical testing. Physicians increasingly are employed by hospital systems, IDNs, ACOs or large group practices integrated with healthcare systems, instead of organizing physician-owned practices, which is changing the dynamics for whether clinical testing is performed in or outside of a hospital. Physicians and other clinicians also increasingly are being employed by health plans or their affiliates. Value-based reimbursement is contributing to changes in the healthcare system. ACOs and patient-centered medical homes are growing as a means to deliver patient care. Health care services increasingly are being provided by non-traditional providers (e.g., physician assistants), in non-traditional venues (e.g., retail medical clinics, urgent care centers) and using new technologies (e.g., telemedicine). In addition, federal healthcare reform legislation adopted in 2010 is resulting in changes in the way that some healthcare services are purchased and delivered in the United States. Patients are also our customers. Increasingly, patients are engaged in their own healthcare and are bearing responsibility for payment for the services that we provide to them. |
Pricing transparency | There has been a trend toward greater pricing transparency in the healthcare marketplace. This transparency, combined with increased patient financial responsibility for medical care, is enhancing purchasing sophistication and changes in behavior in the health care marketplace. |
Competition | The clinical testing industry remains fragmented, is highly competitive and is subject to new competition. Competition is growing from non-traditional competitors. Increased hospital acquisitions of physician practices enhance clinician ties to hospital-affiliated laboratories and may strengthen their competitive position. New industry entrants with extensive resources may make acquisitions or expand into our traditional areas of operations. |
Reimbursement pressure | There is a strong focus in the United States on controlling the overall cost of healthcare. Healthcare market participants, including governments, are focusing on controlling costs, including potentially by changing reimbursement for healthcare services (including but not limited to a shift from fee for service to capitation), changing medical coverage policies (e.g., healthcare benefits design), pre-authorization of lab testing, requiring co-pays, introducing lab spend management utilities and payment and patient care innovations such as ACOs and patient-centered medical homes. In light of continued pressure to reduce systemic healthcare costs, hospitals may change their approach to providing clinical testing services. While pressure to control healthcare costs poses a risk to our Company, it also creates opportunities, such as an opportunity for increased proper utilization of testing as an efficient means to manage the total cost of healthcare. We believe that it also creates greater opportunities for high value, low-cost providers, like our Company, as compared to other providers. |
16
Healthcare utilization | In the past few years, healthcare utilization in the United States has fluctuated based on a number of factors. These factors include, without limitation, the economy, healthcare benefits design, patients delaying medical care and increased patient financial responsibility for medical care. However, federal healthcare reform legislation adopted in 2010 contained provisions eliminating patient cost-sharing for preventive services, and additional provisions that we believe have increased the number of patients that have health insurance, including Medicaid, and thus better access to diagnostic testing. |
Legislative, regulatory and policy environment | Government oversight of and attention to the healthcare industry in the United States is significant and increasing; healthcare payment reform is a top issue. In 2015, the President of the United States launched the Precision Medicine Initiative to improve health and treat disease. The Initiative is expected to pioneer a new model of patient-powered research that promises to accelerate biomedical discoveries and provide clinicians with new tools, knowledge and therapies to select which treatments will work best for patients. Pursuant to the federal Protecting Access to Medicare Act of 2014, it is expected that the Centers for Medicare and Medicaid Services (“CMS”) will revise reimbursement schedules for clinical laboratory testing services provided under Medicare. The FDA has announced guidance initiatives that may impact the clinical laboratory testing business, including by increasing regulation of laboratory-developed tests ("LDTs"). Federal healthcare reform legislation adopted in 2010 has created significant uncertainty as healthcare markets react to changes. For example, more than half of the states have opted in to Medicaid expansion and employers may discontinue offering group health insurance to their employees, shifting more people to exchange products. |
Globalization | There is a growing demand for healthcare services in emerging market countries. Opportunities are arising to participate in the restructuring or growth of the healthcare systems outside the United States. Demographic changes globally also may create opportunities. |
Informatics | The increased availability of healthcare data, including data made available as a result of next generation DNA sequencing, and the increased ability to effectively analyze that data at population and patient levels, is impacting healthcare practices. It is anticipated that the increased use of data in healthcare will help to improve patient outcomes and reduce overall healthcare costs. Informatics, including integrated diagnostic and decision support solutions, use of population data and healthcare information technology, is spurring advances in precision medicine, including medical decision making and value, for populations and individuals. Healthcare market participants, including pharmaceutical companies, health plans, clinicians, ACOs and hospitals, are striving to leverage interoperability, informatics and analytics to positively influence the health of patient populations. |
17
Customers
We provide diagnostic information services to a broad range of customers, including those discussed below.
Table 19 - Customers | |
Health plans including managed care organizations and other health insurance providers | These customers typically reimburse us as a contracted provider on behalf of their members. In certain locations, health plans may delegate to independent physician associations (“IPAs”) or other alternative delivery systems (e.g., physician hospital organizations, ACOs, patient-centered medical homes) the ability to negotiate for diagnostic information services on behalf of certain members. Health plans and IPAs often require that diagnostic information services providers accept discounted fee structures or assume all or a portion of the financial risk associated with providing such services through capitated payment arrangements. Under capitated payment arrangements, we provide services at a predetermined monthly reimbursement rate for each covered member, generally regardless of the number or cost of services provided by us. Reimbursement under programs that do not provide for capitated payments is typically negotiated on a fee-for-service basis. Reimbursement from our five largest health plans totaled less than 20%, and no one health plan accounted for 10%, of our consolidated net revenues in 2015. Health plans typically negotiate directly or indirectly with a number of diagnostic information services providers, and represent approximately one-half of our total clinical testing volumes and one-half of our net revenues from diagnostic information services. There has been a trend of consolidation among health plans. Health plans also are narrowing their provider networks. We are also sometimes a member of a “complementary network.” A complementary network generally is a set of contractual arrangements that a third party will maintain with various providers that provide discounted fees for the benefit of its customers. A member of a health plan may choose to access a non-contracted provider that is a member of a complementary network; if so, the provider will be reimbursed at a rate negotiated by the complementary network. We attempt to strengthen our relationships with health plans and increase the volume of our services for their members by offering to health plans services and programs that leverage our Company's expertise and resources, including our superior access, extensive test menu, medical staff, data, IT solutions, and wellness and disease management capabilities. |
Clinicians | Clinicians, including both primary care physicians and specialists, requiring diagnostic information services for patients are the primary referral source of our services. Clinicians determine which laboratory to recommend or use based on a variety of factors, including those set forth in Table 21 on page 21. |
18
Hospitals | We believe that we are the industry's leader in servicing hospitals. We provide services to hospitals throughout the United States, including esoteric testing services, in some cases helping manage their laboratories and serving as the medical directors of the hospital's histology or clinical laboratory, including through our Professional Laboratory Services offerings. Hospitals generally maintain an on-site laboratory to perform the significant majority of clinical testing for their patients and refer esoteric testing to outside service providers, which typically charge the hospitals on a negotiated fee-for-service basis. Fee schedules for hospital reference testing services often are negotiated on behalf of hospitals by group purchasing organizations. Hospitals also provide outreach testing, and historically were able to negotiate higher reimbursement rates with health plans than commercial clinical laboratories for comparable services. They may seek to leverage their relationships with community clinicians by encouraging the clinicians to send their outreach testing to the hospital's laboratory. In addition, hospitals that own physician practices may require the practices to refer outreach testing to the hospital's affiliated laboratory. In recent years, there has been a trend of hospitals acquiring physician practices, and as a result, an increased percentage of physician practices are owned by hospitals. Increased hospital acquisitions of physician practices enhance clinician ties to hospital-affiliated laboratories and may strengthen their competitive position. We also have joint venture arrangements with leading hospitals or IDNs in several metropolitan areas. These joint venture arrangements, which provide diagnostic information services for affiliated hospitals as well as for unaffiliated clinicians and other local healthcare providers, serve as our principal facilities in their service areas. Typically, we have either a majority ownership interest in, or day-to-day management responsibilities for, our joint venture relationships. In light of continued pressure to reduce systemic healthcare costs, hospitals may change their approach to providing clinical testing services, including by seeking ways to improve profitability or to better utilize their laboratory capacity. We believe that our combination of services positions us to be an attractive partner for hospitals, offering a full range of strategic relationships. |
ACOs and IDNs | An ACO is a network of providers and facilities that share financial risk in providing or arranging for the provision of healthcare. An IDN is a network of providers and facilities working together in providing or arranging for the provision of healthcare. ACOs and IDNs are increasing in number and becoming more important constituents in delivering healthcare services; their impact on the provision of healthcare services to date has varied. ACOs and IDNs may exercise operational and financial control over providers across the continuum of care, and may function as a payer. Thus, they may be able to manage the health of a population group within a defined geography, and also may be able to influence the cost and quality of healthcare delivery, for example through owned entities and through ancillary services. ACOs may be encouraged to consider exclusive arrangements with healthcare providers that become part of the ACO, or to limit service providers to the ACO, since members of the ACO share financial risk. We are actively engaging with ACOs and IDNs to demonstrate the value of our services. |
Employers | Employers use tests for drugs of abuse to determine an individual's employability and his or her “fitness for duty.” Companies with high employee turnover, safety conscious environments or regulatory testing requirements provide the highest volumes of testing. Factors such as the general economy and job market can impact the utilization of drugs-of-abuse testing. Employers also are investing in health and wellness services. We meet their needs by providing nationwide access to our customizable biometric wellness testing, reporting and analytics, incentive management and flu shot services, directly and through health plan and health improvement providers. These services help employers, employees and others manage healthcare costs and capitalize on trends in personalized health. We seek to grow our employer business through offering new and innovative programs to help them with their goal of maintaining a safe and productive workplace. |
19
Patients | In the current environment, patients are taking increased interest in and responsibility for their healthcare. In addition, patients often are bearing increased financial responsibility for their healthcare (e.g., high deductible health plans). Patients are paying greater attention to their healthcare, are increasing their demands of healthcare providers, have increased expectations regarding their healthcare experiences and are becoming more sophisticated regarding healthcare. For example, in our experience, patients are more focused on transparency, ease of doing business and understanding diagnostics information services than they have been in the past. The changing expectations of patients about their healthcare and their healthcare transactions is influencing the way that we think about our business and the services that we provide. We are well positioned to provide information and insights to patients to help them take actions to improve their healthcare, and increasingly we are providing patients with tools to do this. We strive to give patients reasons to choose us, including a superior patient experience. |
Emerging Retail Healthcare Providers | In recent years, as the healthcare sector changes, retail providers of healthcare services have emerged and are growing. These providers include "big-box" retailers, pharmacy chains, supermarkets, urgent care centers and Internet-based service providers. |
Other Laboratories and Other Customers | We also provide services on a fee-for-service basis to federal, state and local governmental agencies and to other commercial clinical laboratories |
In many cases, the customer that orders the services is not responsible to pay for them. Depending on the billing arrangement and applicable law, the payer may be the patient or a third party. Increasingly, even if a third party is primarily responsible for payment, patients may bear responsibility for a portion of the payment. The following table provides examples of third-party payers.
Table 20 - Sample Third Party Payers | |
• Health plans | • Patient-centered medical homes |
• Self-insured employer benefit funds | • Traditional Medicare or Medicaid program |
• ACOs | • Physicians |
• IDNs | • Others (e.g., hospital, laboratory or employer) |
In light of health care reform, there is increased market activity regarding alternative payment models, including bundled payment models. Increasingly, patients are bearing responsibility for some portion of the payment for the services we provide to them.
GENERAL
Competition. While there has been significant consolidation in the diagnostic information services industry in recent years, our industry remains fragmented and highly competitive. We primarily compete with three types of clinical testing providers: commercial clinical laboratories, hospital-affiliated laboratories and physician-office laboratories. We also compete with other providers, including anatomic pathology practices and large physician group practices. In recent years, competition from hospital-affiliated laboratories has increased. Our largest commercial clinical laboratory competitor is Laboratory Corporation of America Holdings, Inc. In addition, we compete with many smaller regional and local commercial clinical laboratories and specialized esoteric laboratories, as well as manufacturers of in vitro diagnostic products. In anatomic pathology, additional competitors include anatomic pathology practices, including those in academic institutions. There also has been a trend among specialty physician practices to establish their own histology laboratory capabilities and/or bring pathologists into their practices, thereby reducing referrals from these practices.
We believe that healthcare providers traditionally consider a number of factors when selecting a diagnostic information services provider. Those factors include:
20
Table 21 - Factors Traditionally Considered When Selecting a Diagnostic Information Services Provider | |
• Service capability and quality | • Reputation in the medical community |
• Accuracy, timeliness and consistency in reporting test results | • Healthcare information technology solutions, including connectivity options |
• Access to medical/scientific thought leaders for consultation | • Patient access, including the number, convenience and geographic coverage of patient service centers |
• Patient insurance coverage and experience | • Ability to develop new and useful tests and services |
• Number and type of tests performed | • Qualifications of its staff |
• Pricing and overall value | |
We believe that providing the most attractive service offering in the industry, including the most comprehensive test menu, innovative test offerings, a positive customer experience, a staff including medical and scientific experts, strong quality, unparalleled access and distribution, and data-powered integrated information technology solutions provide us with a competitive advantage.
We believe that large diagnostic information services providers may be able to increase their share of the overall diagnostic information services industry due to their large networks and lower cost structures. These advantages should enable larger providers to more effectively serve customers. In addition, we believe that consolidation in the diagnostic information services industry will continue. However, a significant portion of clinical testing is likely to continue to be performed by hospitals, which generally have affiliations with community clinicians and may have more, or more convenient, locations in a market. As a result, we compete against hospital-affiliated laboratories primarily on the basis of service capability, quality and pricing. In addition, market activity may increase the competitive environment. For example, health plan actions to exclude large national providers from contracts may enhance the relative competitive position of regional providers. In addition, increased hospital acquisitions of physician practices enhance the ties of the clinicians to hospital-affiliated laboratories, enhancing the competitive position of hospital-affiliated laboratories. The formation of ACOs and IDNs, and their approach to contracts with healthcare providers, in addition to the impact of informatics, also may impact competition to provide diagnostic information services.
The diagnostic information services industry is faced with changing technology and new product introductions. Competitors may compete using advanced technology, including technology that enables more convenient or cost-effective testing. Competitors also may offer testing to be performed outside of a commercial clinical laboratory, such as (1) point-of-care testing that can be performed by physicians in their offices; (2) complex testing that can be performed by hospitals in their own laboratories; and (3) home testing that can be carried out without requiring the services of outside providers.
The diagnostic products, risk assessment and healthcare information technology industries are highly competitive. We have many competitors, some of which have much more extensive experience in these industries and some of which have greater resources. We compete in the diagnostic products industry through unique and differentiated products. We compete in the risk assessment business by seeking to provide a superior applicant experience, faster services completion and a wider array of quality, integrated services than our competitors. We compete in the healthcare information technology industry by offering solutions that foster better patient care and improve performance for healthcare providers, including smaller and medium sized physician practices.
Sales and Marketing. Our Diagnostic Information Services business has a unified commercial organization focused on the sale and marketing of most of our services. It coordinates closely with our clinical franchise organizations, which are responsible for product marketing. The commercial organization is centrally led, and is organized regionally, in conjunction with our operations organization, to ensure aligned delivery for our customers. We maintain a separate sales and marketing organization for our employer drugs-of-abuse testing services.
In Diagnostic Solutions, we maintain separate sales organizations that focus on selling diagnostic products, healthcare information technology solutions and risk assessment services.
Information Technology. We use information systems extensively in virtually all aspects of our business, including clinical testing, test ordering and reporting, billing, customer service, logistics and management of medical data. We endeavor to establish systems that create value and efficiencies for our Company and customers. The successful delivery of our services
21
depends, in part, on the continued and uninterrupted performance of our information technology systems. We have taken precautionary measures to prevent problems that could affect our information technology systems.
Some of our historic growth has come through acquisitions and, as a result, we continue to use multiple information systems. We have implemented some common systems, and are planning to implement more common laboratory information and billing systems across our operations to standardize our processes. We expect implementation will take several more years to complete, and will result in significantly more centralized systems, improved operating efficiency, more timely and comprehensive information for management and enhanced control over our operational environment.
Quality Assurance. In our Diagnostic Information Services business, our goal is to continually improve the processes for collection, handling, storage and transportation of patient specimens, as well as the precision and accuracy of analysis and result reporting. The Quest Diagnostics Quality Program includes policies and procedures to document, measure and monitor the effectiveness of our laboratory operations in providing and improving quality and meeting the requirements of the agencies that regulate the U.S. clinical laboratory testing industry. The Quality Program is designed so that the quality of laboratory services is monitored objectively and evaluated systematically to proactively identify opportunities to improve patient care and resolve identified problems. We track, and seek to improve on, medical quality and service metrics.
Our quality assurance efforts focus on pre-analytic, analytic and post-analytic processes, including positive patient identification of specimens, specimen tracking, report accuracy, proficiency testing, reference range relevance, process audits, statistical process control and personnel training for all of our laboratories and patient service centers. We also focus on the licensing, credentialing, training and competence of our professional and technical staff. To help achieve our goal of becoming recognized as the undisputed quality leader in the diagnostics information services industry, we continue to implement initiatives to enhance our quality and standardization, using our best-in-class business performance tools. In addition, some of our laboratories have achieved International Organization for Standardization, or ISO, certification for their quality management systems.
As part of our comprehensive quality assurance program, we utilize internal proficiency testing, extensive quality control and rigorous process audits for our diagnostic information services. For most clinical laboratory tests, quality control samples are processed in parallel with the analysis of patient specimens. The results of tests on these quality control samples are monitored to identify trends, biases or imprecision in our analytical processes.
We participate in external proficiency testing and have accreditation or licenses for our clinical laboratory operations from various regulatory agencies or accrediting organizations, such as CMS, the College of American Pathologists (“CAP”) and certain states. All of our laboratories participate in various external quality surveillance programs. They include, but are not limited to, proficiency testing programs administered by CAP, as well as some state agencies. CAP is an independent, nongovernmental organization of board-certified pathologists approved by CMS to inspect clinical laboratories to determine compliance with the standards required by the Clinical Laboratory Improvement Act ("CLIA"). CAP offers an accreditation program to which clinical laboratories may voluntarily subscribe. All of our major regional and esoteric laboratories, including our laboratories outside the U.S., and most of our rapid response laboratories, are accredited by CAP. Accreditation includes on-site inspections and participation in the CAP (or equivalent) proficiency testing program. Also, our cytotechnologists and pathologists participate in an internal peer-review evaluation and one or more external individual proficiency testing programs.
Our diagnostic products business maintains extensive quality assurance programs focused on ensuring that our products are safe and effective and that we comply with applicable regulatory requirements in the United States and other countries. It is regulated by the FDA and is required to be in compliance with the Quality Systems Regulations, 21 CFR part 820, and with applicable standards outside the United States. In addition, our manufacturing sites are certified in accordance with ISO 13485: 2003 standards. We endeavor to design and manufacture our diagnostics products in compliance with Quality Systems Regulations.
Intellectual Property Rights. We own significant intellectual property, including patents, patent applications, technology, trade secrets, know-how, copyrights and trademarks in the United States and other countries. From time to time, we also license U.S. and non-U.S. patents, patent applications, technology, trade secrets, know-how, copyrights or trademarks owned by others. In the aggregate, these intellectual property assets and licenses are of material importance to our business. We believe, however, that no single patent, technology, trademark, intellectual property asset or license is material to our business as a whole.
Our approach is to manage our intellectual property assets, to safeguard them and to maximize their value to our enterprise. We actively defend our important intellectual property assets and pursue protection of our products, processes and other intellectual property where possible.
22
Our success in remaining a leading innovator in the diagnostic information services industry by continuing to introduce new tests, technology and services will depend, in part, on our ability to license new and improved technologies on favorable terms. Other companies or individuals, including our competitors, may obtain patents or other property rights on tests or processes that we may be performing, particularly in such emerging areas as gene-based testing and other specialty testing, that could prevent, limit or interfere with our ability to develop, perform or sell our tests or operate our business.
Employees. At December 31, 2015, we employed approximately 44,000 people. This total excludes employees of the joint ventures where we do not have a majority ownership interest. We have no collective bargaining agreements with unions covering employees in the United States, and we believe that our overall relations with our employees are good.
BILLING AND REIMBURSEMENT
Billing. We generally bill for diagnostic information services on a fee-for-service basis under one of two types of fee schedules. These fees may be negotiated or discounted. The types of fee schedules are:
• | “Client” fees charged to physicians, hospitals and institutions for which services are performed on a wholesale basis and which are billed on a monthly basis. |
• | “Patient” fees charged to individual patients and certain third-party payers on a claim-by-claim basis. |
Billing for diagnostic information services is very complicated, and we maintain compliance policies and procedures for our billing. Patients, insurance companies, Medicare, Medicaid, physicians, hospitals, IDNs and employer groups all have different billing requirements. Some billing arrangements require us to bill multiple payers, and there are several other factors that complicate billing (e.g., disparity in coverage and information requirements among various payers; and incomplete or inaccurate billing information provided by ordering clinicians). We incur additional costs as a result of our participation in Medicare and Medicaid programs because diagnostic testing services are subject to complex, stringent and frequently ambiguous federal and state laws and regulations, including those relating to coverage, billing and reimbursement. Additionally, auditing for compliance with applicable laws and regulations as well as internal compliance policies and procedures adds further cost and complexity to the billing process. Changes in laws and regulations could further complicate our billing and increase our billing costs. CMS establishes procedures and continuously evaluates and implements changes to the reimbursement process and requirements for coverage.
As an integral part of our billing compliance program, we investigate reported failures or suspected failures to comply with federal and state healthcare reimbursement requirements. Any Medicare or Medicaid overpayments resulting from non-compliance are reimbursed by us. As a result of these efforts, we have periodically identified and reported overpayments, reimbursed the payers for overpayments and taken appropriate corrective action.
Our bad debt expense is primarily the result of the failure of patients to pay the portion of the receivable that is their responsibility. In recent years, increased patient responsibility has adversely impacted our bad debt expense. To the extent that health plans and other programs require greater levels of patient cost-sharing, this could negatively impact our bad debt expense. We are taking, and plan to continue to take, steps to improve our patient collection experience.
The remainder of our bad debt expense is primarily due to missing or incorrect billing information on requisitions and Advance Beneficiary Notices received from healthcare providers. In general, due to the nature of our business, historically we have performed the requested testing and reported test results regardless of whether the billing information is correct or complete. We subsequently attempt to contact the healthcare provider or patient to obtain any missing information and to rectify incorrect billing information. Missing or incorrect information on requisitions complicates and slows down the billing process, creates backlogs of unbilled requisitions and generally increases the aging of accounts receivable and bad debt expense. The increased use of electronic ordering reduces the incidence of missing or incorrect information.
Government Coverage and Reimbursements. Government payers, such as Medicare and Medicaid, have taken steps and are expected to continue to take steps to control the cost, utilization and delivery of healthcare services, including clinical test services. Historically, most Medicare and Medicaid beneficiaries were covered under the traditional Medicare and Medicaid programs administered by the federal government. Over the last several years, the federal government has continued to expand its contracts with private health insurance plans for Medicare beneficiaries and has encouraged such beneficiaries to switch from the traditional programs to the private programs, called “Medicare Advantage” programs. There has been growth of health insurance providers offering Medicare Advantage programs and of beneficiary enrollment in these programs. In recent years, in an effort to control costs, states also have mandated that Medicaid beneficiaries enroll in private managed care arrangements.
23
With regard to the clinical testing services performed on behalf of Medicare beneficiaries, we must bill the Medicare program directly and must accept the local Medicare carrier's fee schedule amount for covered services as payment in full. In addition, state Medicaid programs are prohibited from paying more (and in most instances, pay significantly less) than Medicare. Currently, Medicare does not require the beneficiary to pay a co-payment for diagnostic information services reimbursed under the Clinical Laboratory Fee Schedule, but generally does require a patient deductible for anatomic pathology services. Certain Medicaid programs require Medicaid recipients to pay co-payment amounts for diagnostic information services.
Part B of the Medicare program contains fee schedule payment methodologies for clinical testing services performed for covered patients, including a national ceiling on the amount that carriers could pay under their local Medicare clinical testing fee schedules. Historically, the Medicare Clinical Laboratory Fee Schedule and the Medicare Physician Fee Schedule established under that program have been subject to change, including each year. For 2016, each schedule is changing and reimbursement under each schedule will be different in 2016 than 2015 levels. The following table sets forth the percentage of our consolidated net revenues reimbursed under Medicare and Medicaid in 2015.
Table 22 - 2015 Medicare and Medicaid Revenues | |||
% of 2015 Consolidated Net Revenues | |||
Medicare Clinical Laboratory Fee Schedule | 12 | ||
Medicare Physician Fee Schedule | 2 | ||
Total Medicare | 14 | ||
Medicaid Programs | 3 | ||
Total Traditional Medicare and Medicaid | 17 | ||
Violations of laws relating to billing government healthcare programs or federal and state fraud and abuse laws may result in: exclusion from participation in Medicare/Medicaid programs; civil and criminal fines and penalties; and the loss of various licenses, certificates and authorizations necessary to operate our business. Certain violations of these laws may also provide the basis for a civil remedy under the federal False Claims Act, including fines and damages of up to three times the amount claimed.
REGULATION
Our businesses are subject to or impacted by extensive and frequently changing laws and regulations in the United States (at both the federal and state levels) and the other jurisdictions in which we conduct business, including some particular to our business and others relating to conducting business generally (e.g., export controls laws, U.S. Foreign Corrupt Practices Act). We also are subject to inspections and audits by governmental agencies. The table below highlights the key regulatory schemes applicable to our businesses.
Table 23 - Key Regulatory Schemes | |
CLIA and State Clinical Laboratory Licensing | CLIA regulates the operations of virtually all clinical laboratories, requiring that they be certified by the federal government and that they comply with various technical, operational, personnel and quality requirements intended to ensure that the services provided are accurate, reliable and timely. State laws may require additional personnel qualifications or licenses, quality control, record maintenance, proficiency testing or detailed review of our scientific method validations and technical procedures for certain tests. |
24
Fraud and Abuse | Anti-kickback laws and regulations prohibit making payments or furnishing other benefits to influence the referral of tests billed to Medicare, Medicaid or certain other federal or state healthcare programs. In addition, federal and state anti-self-referral laws generally prohibit Medicare and Medicaid payments for clinical tests referred by physicians who have an ownership or investment interest in, or a compensation arrangement with, the testing laboratory. Some states have similar laws that are not limited to Medicare and Medicaid referrals and could also affect other tests referred by clinicians with investment or compensation arrangements with the testing laboratory. Violations of these laws and regulations may result in monetary fines, criminal and civil penalties and/or suspension or exclusion from participation in Medicare, Medicaid and other federal or state healthcare programs. |
FDA | The FDA has regulatory responsibility over, among other areas, instruments, software, test kits, reagents and other devices used by clinical laboratories to perform diagnostic testing in the United States. The FDA also regulates drugs-of-abuse testing for employers and insurers, testing for blood bank purposes and testing of donors of human cells for purposes such as in vitro fertilization. A number of esoteric tests we develop internally are offered as LDTs. The FDA has claimed regulatory authority over all LDTs, but has stated that it exercised enforcement discretion with regard to most LDTs performed by high complexity CLIA-certified laboratories. The FDA has announced guidance initiatives that may significantly impact the clinical laboratory testing business, including by increasing regulation of LDTs. Our diagnostic products business is subject to regulation by the FDA, as well as by foreign governmental agencies, under laws and regulations that govern the development, testing, manufacturing, labeling, advertising, marketing, distribution and post-market surveillance of diagnostic products. These regulators possess the authority to take various administrative and legal actions against us for non-compliance, such as fines, product suspensions, submission of warning letters, recalls, product seizures, injunctions and other civil and criminal sanctions. |
Environmental, Health and Safety | We are subject to laws and regulations related to the protection of the environment, the health and safety of employees and the handling, transportation and disposal of medical specimens, infectious and hazardous waste and radioactive materials. For example, the U.S. Occupational Safety and Health Administration has established extensive requirements relating specifically to workplace safety for healthcare employers in the U.S. This includes requirements to develop and implement multi-faceted programs to protect workers from exposure to blood-borne pathogens, including preventing or minimizing any exposure through needle stick injuries. For purposes of transportation, some biological materials and laboratory supplies are classified as hazardous materials and are subject to regulation by one or more of the following agencies: the U.S. Department of Transportation, the U.S. Public Health Service, the U.S. Postal Service and the International Air Transport Association. |
25
Physicians | Our pathologists are required to hold a valid license to practice medicine in the jurisdiction in which they practice. Many of our pathologists enter into an employment agreement. These agreements have varying terms, but generally can be terminated at any time, upon advance notice. Most of the agreements contain covenants generally limiting the activities of the pathologist within a defined geographic area for a limited period of time after termination of employment; the enforceability of these covenants may be limited under state law. Several jurisdictions, including some in which our businesses are located, prohibit business corporations from engaging in the practice of medicine. In certain jurisdictions, business corporations are prohibited from employing licensed healthcare professionals to provide services on behalf of the corporation; these laws vary. In some jurisdictions, anatomic pathology services are delivered through physician-owned entities that employ the practicing pathologists. The manner in which licensed physicians can be organized to perform medical services may be governed by the laws of the jurisdictions in which medical services are provided and by the medical boards or other entities authorized by these jurisdictions to oversee the practice of medicine. |
Privacy and Security of Health and Personal Information | We are subject to laws and regulations regarding protecting the security and privacy of certain healthcare and personal information, including the federal Health Insurance Portability and Accountability Act and the regulations thereunder, which establish (i) a complex regulatory framework including requirements for safeguarding protected health information and (ii) comprehensive federal standards regarding the uses and disclosures of protected health information. A healthcare provider may be required to notify individuals or the government if the provider discovers certain breaches of personal information or protected health information. |
Drug Testing; Controlled Substances | All U.S. laboratories that perform drug testing for certain public sector employees and employees of certain federally regulated businesses are required to be certified as meeting the detailed performance and quality standards of the Substance Abuse and Mental Health Services Administration. To obtain access to controlled substances used to perform drugs-of-abuse testing in the United States, laboratories must be licensed by the Drug Enforcement Administration. |
Compliance. We strive to conduct our business in compliance with all applicable laws and regulations. All of our laboratories and, where applicable, patient service centers, are licensed and accredited as required by the appropriate federal and state agencies. Many of the laws and regulations applicable to us, however, including many of those relating to billing, reimbursement for tests and relationships with clinicians and hospitals, are vague or indefinite or have not been interpreted by the courts. They may be interpreted or applied by a prosecutorial, regulatory or judicial authority in a manner that could require us to make changes in our operations, including our pricing and/or billing practices. The applicability or interpretation of laws and regulations also may not be clear in light of emerging changes in clinical testing science, healthcare technology and healthcare organizations. Such occurrences, regardless of their outcome, could, among other things:
• | increase our administrative, billing or other operating costs; |
• | decrease the amount of reimbursement related to diagnostic information services performed; |
• | damage our reputation; or |
• | adversely affect important business relationships with third parties. |
If we fail to comply with applicable laws and regulations, we could suffer civil and criminal penalties, fines, exclusion from participation in governmental healthcare programs and the loss of various licenses, certificates and authorizations necessary to operate our business, as well as incur additional liabilities from third-party claims, all of which could have a material adverse effect on our business. Certain federal and state statutes, regulations and other laws, including the qui tam provisions of federal and state false claims acts, allow private individuals to bring lawsuits against healthcare companies on behalf of government payers, private payers and/or patients alleging inappropriate billing practices.
The federal or state governments may bring claims based on our current practices, which we believe are lawful. The federal and state governments have substantial leverage in negotiating settlements since the amount of potential damages far exceeds the rates at which we are reimbursed, and the government has the remedy of excluding a non-compliant provider from participation in the Medicare and Medicaid programs. We believe that, based on our experience with settlements and public announcements by various government officials, federal and state governments continue to strengthen their enforcement efforts against perceived healthcare fraud. In addition, in recent years legislative provisions relating to healthcare fraud and abuse
26
provide government enforcement personnel substantially increased funding, powers, penalties and remedies to pursue suspected cases of fraud and abuse.
We have a long-standing and well-established compliance program. The Quality, Safety & Compliance Committee of our Board of Directors oversees our compliance program and requires periodic management reports regarding our compliance program. Our program includes detailed policies and procedures and training programs intended to ensure the strict implementation and observance of all applicable laws, regulations and Company policies. Further, we conduct in-depth reviews of procedures and facilities to assure regulatory compliance throughout our operations. We conduct annual training of our employees on these compliance policies and procedures.
AVAILABLE INFORMATION
We file annual, quarterly and current reports, proxy statements and other information with the Securities and Exchange Commission (the “SEC”). You may read and copy any document that we file with the SEC at the SEC's public reference room at 100 F Street, NE, Washington, DC 20549 on official business days. Please call the SEC at 1-800-SEC-0330 for information regarding the public reference room. The SEC maintains an internet site that contains annual, quarterly and current reports, proxy and information statements and other information that issuers (including the Company) file electronically with the SEC. Our electronic SEC filings are available to the public at the SEC's internet site, www.sec.gov.
Our internet site is www.QuestDiagnostics.com. You can access our Investor Relations webpage at www.QuestDiagnostics.com/investor. The information on our website is not incorporated by reference into this Report. We make available free of charge, on or through our Investor Relations webpage, our proxy statements, Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and any amendments to those reports filed or furnished pursuant to the Securities Exchange Act of 1934, as amended (the “Exchange Act”), as soon as reasonably practical after such material is filed with, or furnished to, the SEC.
We also have a corporate governance webpage, www.QuestDiagnostics.com/governance. You can access information regarding our corporate governance, including the information set forth below, at that page.
Table 24 - Information Available at Our Corporate Governance Webpage | |
• Directors | • Corporate Governance Guidelines |
• Composition of the committees of our Board of Directors | • Code of Business Ethics |
• Management | • Certificate of Incorporation |
• Charters for the standing committees of our Board of Directors | • Bylaws |
• Integrity Commitment | • Corporate Political Contributions Policy |
• Statements of beneficial ownership of our equity securities filed by our directors, officers and others under Section 16 of the Exchange Act | • Values |
27
EXECUTIVE OFFICERS OF THE COMPANY
The following persons serve as executive officers of the Company.
Table 25 - Executive Officers | |
Name, Age, Title | Background |
Stephen H. Rusckowski (58) President and Chief Executive Officer | Prior to joining the Company in May 2012, since October 2006, he was Chief Executive Officer of Philips Healthcare, the largest unit of Royal Philips Electronics, and a member of the Board of Management of Royal Philips Electronics and its Executive Committee. Previously, he was CEO of the Imaging Systems business of Royal Phillips Electronics. Before joining Philips in 2001, Mr. Rusckowski held numerous management positions with the healthcare division of Hewlett-Packard/Agilent Technologies. Mr. Rusckowski has been a director of the Company since May 2012. He has been a director of Xerox Corporation since February 2015. Mr. Rusckowski is the Chairman of the American Clinical Laboratory Association. |
Jon R. Cohen, M.D. (61) Senior Vice President and Group Executive - Diagnostic Solutions | He joined the company in March 2009 as Chief Medical Officer. From May 2011 to January 2013, Dr. Cohen also had responsibility for Hospital Services. In January 2013, he assumed responsibility for cancer diagnostics, pathology services, sports science and human performance and professional laboratory services. From February 2014 to July 2015, he had responsibility for our clinical trials business. Dr. Cohen served as the Senior Adviser to New York Governor David Patterson from 2008 to 2009, where he was responsible for all policy and strategic planning. Previously, Dr. Cohen was a managing director, health industries advisory services, at PricewaterhouseCoopers LLP, and spent 21 years with North Shore-Long Island Jewish Health System, one of the nation's largest not-for-profit health systems, including serving as its Chief Medical Officer from 2000 to 2006. |
Everett V. Cunningham (49) Senior Vice President, Commercial | He is responsible for the commercial organization for the Company's Diagnostic Information Services business. Prior to joining the Company in October 2012, Mr. Cunningham spent 21 years with Pfizer, Inc., where he served from June 2011 to October 2012 as Regional President, Established Products, Asia. From 2009 to 2011, Mr. Cunningham served as Regional President, West Business Unit, Primary Care. From 2007 to 2009, he served as Vice President, Human Resources, Corporate Groups. Before that Mr. Cunningham served Pfizer in a series of sales and leadership and general management roles. |
28
James E. Davis (53) Senior Vice President and Group Executive - Regional Businesses | In January 2015, he assumed responsibility for the general management of the Company's regional Diagnostic Information Services businesses. Mr. Davis also remains responsible for our products business; he has been responsible for this business since February 2014. From February 2014 to January 2015, he was responsible for operations for the Company's Diagnostic Information Services business. He joined Quest Diagnostics in April 2013 as Senior Vice President, Diagnostics Solutions, with responsibility for the healthcare information technology, risk assessment, clinical trials, diagnostic products and employer solutions businesses. Prior to joining Quest Diagnostics, from March 2012 to April 2013, Mr. Davis served as Lead Director, and then as Chief Executive Officer, of InSightec, Inc., a medical device company that designs and develops ultrasound ablation devices that are guided by magnetic resonance imaging systems. Previously, Mr. Davis held a number of senior positions in General Electric’s healthcare business, including from 2007 to 2012 as Vice President and General Manager of GE Healthcare’s magnetic resonance imaging business. Prior to joining GE Healthcare, Mr. Davis held leadership positions in GE’s aviation business and led the development of strategic and operational improvement initiatives for clients of McKinsey & Company, Inc. |
Catherine T. Doherty (53) Senior Vice President and Group Executive - Clinical Franchise Solutions | She is responsible for overseeing the development of clinical franchise solutions in the areas of cardiovascular, infectious disease and immunology, neurology, prescription drug monitoring and toxicology, women's health and general wellness, as well as enterprise-wide strategic marketing and business development. In February 2014, Ms. Doherty assumed responsibility for the employer solutions, healthcare information technology and risk assessment businesses. From May 2011 to December 2012, she served as Senior Vice President, Physician Services. From 2008 through May 2011, Ms. Doherty served as Vice President, Hospital Services. Prior to 2008, Ms. Doherty held a variety of positions of increasing responsibility since joining the Company in 1990, including Vice President, Office of the Chairman; Vice President, Finance and Administration for the Hospital business; Vice President, Communications and Investor Relations; and Chief Accounting Officer. |
Mark J. Guinan (54) Senior Vice President and Chief Financial Officer | He joined the Company in July 2013. From 2010 until joining Quest Diagnostics in 2013, Mr. Guinan served as Chief Financial Officer for Hill-Rom Holdings Inc., a manufacturer and provider of medical technologies and related services for the health care industry. Previously, he had served in a number of finance and operations roles in a long career at Johnson & Johnson including 2009 to 2010 as Vice President, Chief Procurement Officer, and 2005 to 2009 as Vice President, Group Finance Pharmaceuticals. Before joining Johnson & Johnson in 1997, he held a number of financial roles at Procter & Gamble. |
29
Michael E. Prevoznik (54) Senior Vice President and General Counsel | Mr. Prevoznik joined the Company as Vice President and General Counsel in August 1999. In 2003, he assumed responsibility for governmental affairs. From 1999 until April 2009, Mr. Prevoznik also had responsibility for the Company's Compliance Department. Since April 2011, in addition to serving as General Counsel, Mr. Prevoznik has had management responsibility for the Company's diagnostic information services activities outside the U.S. In addition, from April 2011 to January 2013, Mr. Prevoznik had management responsibility for the Company's clinical trials business. Prior to joining the Company, Mr. Prevoznik served in positions of increasing responsibility within the compliance organization at SmithKline Beecham, most recently as Vice President, Compliance, with responsibility for coordinating all SmithKline Beecham compliance activities worldwide. |
Item 1A. Risk Factors
You should carefully consider all of the information set forth in this Report, including the following risk factors, before deciding to invest in any of our securities. The risks below are not the only ones that we face. Additional risks not presently known to us, or that we presently deem immaterial, may also negatively impact us. Our business, consolidated financial condition, revenues, results of operations, profitability, reputation or cash flows could be materially impacted by any of these factors.
This Report also includes forward-looking statements that involve risks or uncertainties. Our results could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including the risks we face described below and elsewhere. See “Cautionary Factors that May Affect Future Results” on page 37. |
The U.S. healthcare system is evolving, and our business could be adversely impacted if we fail to adapt.
The U.S. healthcare system is evolving, in part in response to the passage of the Affordable Care Act ("ACA") in 2010. The law provided for reductions in the Medicare clinical laboratory fee schedule of 1.75% for five years beginning in 2011 and also included a productivity adjustment that reduced the CPI market basket update since 2011. The law imposes an excise tax on the seller for the sale of certain medical devices in the U.S., including those purchased and used by laboratories; effective January 2016, Congress imposed a two-year moratorium on the device tax. The law established the Independent Payment Advisory Board, which is responsible to submit annually proposals aimed at reducing Medicare cost growth while preserving quality. These proposals automatically will be implemented unless Congress enacts alternative proposals that achieve the same savings targets. Further, the ACA established the Center for Medicare and Medicaid Innovation to examine alternative payment methodologies and conduct demonstration programs. The law provided for extensive health insurance reforms, including the elimination of pre-existing condition exclusions and other limitations on coverage, fixed percentages on medical loss ratios, expansion in Medicaid and other programs, employer mandates, individual mandates, creation of state and regional health insurance exchanges, and tax subsidies for individuals to help cover the cost of individual insurance coverage. The law also permits the establishment of ACOs.
Significant change is taking place in the healthcare system, including as discussed above under the heading The United States Clinical Testing Industry, beginning on page 14. For example, ACOs and patient-centered medical homes are growing as a means to deliver patient care. Value-based reimbursement is increasing; CMS has set goals for value-based reimbursement to be achieved in coming years. Healthcare industry participants are consolidating. Healthcare services increasingly are being provided by non-traditional providers (e.g., physician assistants), in non-traditional venues (e.g., retail medical clinics, urgent care centers) and using new technologies (e.g., telemedicine). We expect that the evolution of the healthcare industry will continue, and that industry change is likely to be extensive.
30
The clinical testing business is highly competitive, and if we fail to provide an appropriately priced level of service or otherwise fail to compete effectively it could have a material adverse effect on our revenues and profitability.
The clinical testing business remains a fragmented and highly competitive industry. We primarily compete with three types of clinical testing providers: other commercial clinical laboratories, hospital-affiliated laboratories and physician-office laboratories. We also compete with other providers, including anatomic pathology practices and large physician group practices. Hospitals generally maintain on-site laboratories to perform testing on their patients (inpatient or outpatient). In addition, many hospitals compete with commercial clinical laboratories for outreach (non-hospital patients) testing. Hospitals may seek to leverage their relationships with community clinicians and encourage the clinicians to send their outreach testing to the hospital's laboratory. In addition, hospitals that own physician practices may require the practices to refer testing to the hospital's laboratory. In recent years, there has been a trend of hospitals acquiring physician practices, and as a result, an increased percentage of physician practices are owned by hospitals. As a result of this affiliation between hospitals and community clinicians, we compete against hospital-affiliated laboratories primarily based on quality and scope of service as well as pricing. Increased hospital acquisitions of physician practices enhance clinician ties to hospital-affiliated laboratories and may strengthen their competitive position. The formation of ACOs and IDNs, and their approach to contracts with healthcare providers, in addition to the impact of informatics, also may increase competition to provide diagnostic information services.
The diagnostic information services industry also is faced with changing technology and new product introductions. Competitors may compete using advanced technology, including technology that enables more convenient or cost-effective testing. Competitors also may offer testing to be performed outside of a commercial clinical laboratory, such as (1) point-of-care testing that can be performed by physicians in their offices; (2) complex testing that can be performed by hospitals in their own laboratories; and (3) home testing that can be carried out without requiring the services of outside providers.
Government payers, such as Medicare and Medicaid, have taken steps to reduce the utilization and reimbursement of healthcare services, including clinical testing services.
We face efforts by government payers to reduce utilization of and reimbursement for diagnostic information services. We expect efforts to reduce reimbursements, to impose more stringent cost controls and to reduce utilization of clinical test services will continue.
From time to time, Congress has legislated reductions in, or frozen updates to, the Medicare Clinical Laboratory Fee Schedule. In addition, CMS has adopted policies limiting or excluding coverage for clinical tests that we perform. We also provide physician services which are reimbursed by Medicare under a physician fee schedule, which is subject to adjustment on an annual basis. In recent years, reductions in the Medicare Physician Fee Schedule for anatomic pathology services adversely impacted our business relative to the business of some of our competitors whose anatomic pathology business was not as sizable as ours. Medicaid reimbursement varies by state and is subject to administrative and billing requirements and budget pressures. The 2010 federal healthcare reform legislation includes further provisions that are designed to control utilization and payment levels.
In addition, over the last several years, the federal government has continued to expand its contracts with private health insurance plans for Medicare beneficiaries, called “Medicare Advantage” programs, and has encouraged such beneficiaries to switch from the traditional programs to the private programs. There has been continued growth of health insurance plans offering Medicare Advantage programs, and of beneficiary enrollment in these programs. Also in recent years, states have mandated that Medicaid beneficiaries enroll in private managed care arrangements. Recently, state budget pressures have encouraged states to consider several courses of action that may impact our business, such as delaying payments, reducing reimbursement, restricting coverage eligibility, denying claims and service coverage restrictions.
From time to time, the federal government has considered whether competitive bidding could be used to provide clinical testing services for Medicare beneficiaries at attractive rates while maintaining quality and access to care. Congress periodically considers cost-saving initiatives as part of its deficit reduction discussions. These initiatives have included coinsurance for clinical testing services, co-payments for clinical testing and further laboratory fee schedule reductions.
2014 U.S. federal legislation, the Protecting Access to Medicare Act of 2014, is impacting the clinical testing industry. Key parts of this legislation included provisions that provide for the establishment of an advisory panel and a market-based process to rebase the Clinical Laboratory Fee Schedule, developing a new fee schedule and limiting reductions in that fee schedule. If this process does not recognize the value that clinical testing services bring to the healthcare system, our business can be materially adversely impacted.
31
Health plans and other third parties have taken steps to reduce the utilization and reimbursement of health services, including clinical testing services.
We face efforts by non-governmental third-party payers, including health plans, to reduce utilization of and reimbursement for clinical testing services. For example, since the passage of ACA, there is increased market activity regarding alternative payment models, including bundled payment models. We expect continuing efforts by third-party payers, including in their rules, practices and policies, to reduce reimbursements, to impose more stringent cost controls and to reduce utilization of clinical testing services. ACOs and IDNs also may undertake efforts to reduce utilization of, or reimbursement for, diagnostic information services.
The healthcare industry has experienced a trend of consolidation among health insurance plans, resulting in fewer but larger insurance plans with significant bargaining power to negotiate fee arrangements with healthcare providers, including clinical testing providers. These health plans, and independent physician associations, may demand that clinical testing providers accept discounted fee structures or assume all or a portion of the financial risk associated with providing testing services to their members through capitated payment arrangements. In addition, some health plans have been willing to limit the PPO or POS laboratory network to only a single national laboratory to obtain improved fee-for-service pricing; we may cease to be a contracted provider to a health plan. Some health plans also are reviewing test coding, evaluating coverage decisions and considering steps such as requiring preauthorization of testing. There are also an increasing number of patients enrolling in consumer driven products and high deductible plans that involve greater patient cost-sharing.
The increased consolidation among health plans also has increased pricing transparency and bargaining power and the potential adverse impact of ceasing to be a contracted provider with any such insurer. The ACA included provisions, including ones regarding the creation of healthcare exchanges, that may encourage health insurance plans to increase exclusive contracting.
Government payers and third parties, including health plans, may not recognize the value of, or compensate or reimburse us for, new and innovative solutions.
Government payers and third parties, including health plans, are taking steps to reduce utilization of, and reimbursement for, some new and innovative healthcare solutions, including new tests and other solutions that we may offer. These steps discourage innovation and access to innovative solutions that we may offer.
Our business could be negatively affected if we are unable to continue to improve our efficiency.
It is important that we continue to improve our efficiency to enable us to mitigate the impact on our profitability of steps taken by government payers and health insurers to reduce the utilization and reimbursement of healthcare services, including diagnostic information services.
Business development activities are inherently risky, and integrating our operations with businesses we acquire may be difficult.
We plan selectively to enhance our business from time to time through business development activities, such as acquisitions, licensing, investments and alliances. However, these plans are subject to the availability of appropriate opportunities and competition from other companies seeking similar opportunities. Moreover, the success of any such effort may be affected by a number of factors, including our ability to properly assess and value the potential business opportunity, and to integrate it into our business. The success of our strategic alliances depends not only on our contributions and capabilities, but also on the property, resources, efforts and skills contributed by our strategic partners. Further, disputes may arise with strategic partners, due to conflicting priorities or conflicts of interests.
Each acquisition involves the integration of a separate company that has different systems, processes, policies and cultures. Integration of acquisitions involves a number of risks including the diversion of management's attention to the assimilation of the operations of businesses we have acquired, difficulties in the integration of operations and systems and the realization of potential operating synergies, the assimilation and retention of the personnel of the acquired companies, challenges in retaining the customers of the combined businesses, and potential adverse effects on operating results. The process of combining companies may be disruptive to our businesses and may cause an interruption of, or a loss of momentum in, such businesses as a result of the following difficulties, among others:
• | loss of key customers or employees; |
• | difficulty in standardizing information and other systems; |
32
• | difficulty in consolidating facilities and infrastructure; |
• | failure to maintain the quality or timeliness of services that our Company has historically provided; |
• | diversion of management's attention from the day-to-day business of our Company as a result of the need to deal with the foregoing disruptions and difficulties; and |
• | the added costs of dealing with such disruptions. |
If we are unable successfully to integrate strategic acquisitions in a timely manner, our business and our growth strategies could be negatively affected. Even if we are able to successfully complete the integration of the operations of other companies or businesses we may acquire in the future, we may not be able to realize all or any of the benefits that we expect to result from such integration, either in monetary terms or in a timely manner.
We are subject to numerous legal and regulatory requirements governing our activities, and we may face substantial fines and penalties, and our business activities may be impacted, if we fail to comply.
Our business is subject to or impacted by extensive and frequently changing laws and regulations in the United States (including at both the federal and state levels) and the other jurisdictions in which we engage in business. While we seek to conduct our business in compliance with all applicable laws, many of the laws and regulations applicable to us are vague or indefinite and have not been interpreted by the courts, including many of those relating to:
• | billing and reimbursement of clinical testing; |
• | certification or licensure of clinical laboratories; |
• | the anti-self-referral and anti-kickback laws and regulations; |
• | the laws and regulations administered by the FDA; |
• | the corporate practice of medicine; |
• | operational, personnel and quality requirements intended to ensure that clinical testing services are accurate, reliable and timely; |
• | physician fee splitting; |
• | relationships with physicians and hospitals; |
• | safety and health of laboratory employees; and |
• | handling, transportation and disposal of medical specimens, infectious and hazardous waste and radioactive materials. |
These laws and regulations may be interpreted or applied by a prosecutorial, regulatory or judicial authority in a manner that could require us to make changes in our operations, including our pricing and/or billing practices. We may not be able to maintain, renew or secure required permits, licenses or any other regulatory approvals needed to operate our business or commercialize our services. If we fail to comply with applicable laws and regulations, or if we fail to maintain, renew or obtain necessary permits, licenses and approvals, we could suffer civil and criminal penalties, fines, exclusion from participation in governmental healthcare programs and the loss of various licenses, certificates and authorizations necessary to operate our business, as well as incur additional liabilities from third-party claims. If any of the foregoing were to occur, our reputation could be damaged and important business relationships with third parties could be adversely affected.
We regularly receive requests for information, and occasionally subpoenas, from governmental authorities. We also are subject from time to time to qui tam claims brought by former employees or other “whistleblowers.” The federal and state governments continue to strengthen their scrutiny and enforcement efforts against perceived healthcare fraud. Legislative provisions relating to healthcare fraud and abuse provide government enforcement personnel substantially increased funding, powers, penalties and remedies to pursue suspected cases of fraud and abuse. In addition, the government has substantial leverage in negotiating settlements since the amount of potential damages far exceeds the rates at which we are reimbursed for our services, and the government has the remedy of excluding a non-compliant provider from participation in the Medicare and Medicaid programs. Regardless of merit or eventual outcome, these types of investigations and related litigation can result in:
• | diversion of management time and attention; |
• | expenditure of large amounts of cash on legal fees, costs and payment of damages; |
• | limitations on our ability to continue some of our operations; |
• | enforcement actions, fines and penalties or the assertion of private litigation claims and damages; |
• | decreased demand for our services ; and/or |
• | injury to our reputation. |
Although we believe that we are in compliance, in all material respects, with applicable laws and regulations, there can be no assurance that a regulatory agency or tribunal would not reach a different conclusion. Moreover, even when an
33
investigation is resolved favorably, the process may be time-consuming and the legal costs and diversion of management focus may be extensive.
Changes in applicable laws and regulations may result in existing practices becoming more restricted, or subject our existing or proposed services to additional costs, delay, modification, withdrawal or reconsideration. Such changes also could require us to modify our business objectives.
Our business could be adversely impacted by the FDA's approach to regulation.
The FDA has regulatory responsibility over, among other areas, instruments, test kits, reagents and other devices used by clinical laboratories to perform diagnostic testing in the U.S. A number of esoteric tests we develop internally are offered as LDTs. The FDA has claimed regulatory authority over all LDTs, but has stated that it exercised enforcement discretion with regard to most LDTs performed by high complexity CLIA-certified laboratories. The FDA has announced guidance initiatives that may impact the clinical testing business, including by increasing regulation of LDTs. These initiatives could have a significant impact on our business, including the application of medical device excise taxes to our business. The approach may hinder our ability to develop and market new services, cause an increase in the cost of our services, delay our ability to introduce new tests or hinder our ability to perform testing.
Failure to accurately bill for our services could have a material adverse effect on our business.
Billing for diagnostic information services is complex and subject to extensive and non-uniform rules and administrative requirements. Depending on the billing arrangement and applicable law, we bill various payers, such as patients, insurance companies, Medicare, Medicaid, clinicians, hospitals and employer groups. Failure to comply with applicable laws relating to billing government healthcare programs may result in various consequences, including: (1) exclusion from participation in Medicare/Medicaid programs; (2) asset forfeitures; (3) civil and criminal fines and penalties; and (4) the loss of various licenses, certificates and authorizations necessary to operate our business.
Attacks on our information technology systems, or failure in these systems, including failures resulting from our systems conversions, could disrupt our operations and cause the loss of confidential information, customers and business opportunities or otherwise adversely impact our business.
IT systems are used extensively in virtually all aspects of our business, including clinical testing, test reporting, billing, customer service, logistics and management of medical data. Our success depends, in part, on the continued and uninterrupted performance of our IT systems. IT systems may be vulnerable to damage, disruptions and shutdown from a variety of sources, including telecommunications or network failures, human acts and natural disasters. Unauthorized persons may seek to obtain intellectual property and other confidential information that we house on our IT systems. Moreover, despite the security measures we have implemented, our IT systems may be subject to physical or electronic intrusions, computer viruses, unauthorized tampering and similar disruptive problems. Our information technology systems from time to time have experienced minor attacks, minor viruses, attempted intrusions or similar problems, like other major companies, but each was mitigated, and none materially disrupted, interrupted, damaged or shutdown the Company's information technology systems, materially disrupted the Company's performance of its business or, to the Company's knowledge, resulted in material unauthorized access to data.
We have taken precautionary measures to prevent or minimize vulnerabilities in our IT systems, including the loss or theft of intellectual property and other confidential information that we house on our systems. In addition, we continue to strengthen precautionary measures to reduce the risk of, and to detect and respond to, future cyber threats. However, cyber threats are constantly evolving, thereby increasing the difficulty of detecting and successfully defending against them. Breaches of our network or data security could disrupt the security of our internal systems and business applications, impair our ability to provide services to our customers, compromise intellectual property or confidential information or otherwise adversely impact our business. There can be no assurances that our precautionary measures will prevent or successfully defend against cyber threats that could have a significant impact on our business.
We are planning to implement common laboratory information and billing systems, which will promote standardized processes. We expect that this effort will take several years to complete and may result in temporary disruptions in service.
34
Failure to develop, or acquire licenses for, new tests, technology and services could negatively impact our testing volume and revenues.
The clinical testing industry is faced with changing technology and new product introductions. Other companies or individuals, including our competitors, may obtain patents or other property rights that would prevent, limit or interfere with our ability to develop, perform or sell our solutions or operate our business or increase our costs. In addition, they could introduce new tests, technologies or services that may result in a decrease in the demand for our services or cause us to reduce the prices of our services. Our success in continuing to introduce new solutions, technology and services will depend, in part, on our ability to license new and improved technologies on favorable terms. We may be unable to develop or introduce new solutions or services. We also may be unable to continue to negotiate acceptable licensing arrangements, and arrangements that we do conclude may not yield commercially successful clinical tests. If we are unable to license these testing methods at competitive rates, our research and development costs may increase as a result. In addition, if we are unable to develop and introduce, or license, new solutions, technology and services to expand our esoteric testing business, our services may become outdated when compared with our competition.
We may be unable to obtain, maintain or enforce our intellectual property rights and may be subject to intellectual property litigation that could adversely impact our business.
We may be unable to obtain or maintain adequate patent or other proprietary rights for our solutions or services or to successfully enforce our proprietary rights. In addition, we may be subject to intellectual property litigation and we may be found to infringe on the proprietary rights of others, which could force us to do one or more of the following:
• | cease developing, performing or selling solutions or services that incorporate the challenged intellectual property; |
• | obtain and pay for licenses from the holder of the infringed intellectual property right; |
• | redesign or re-engineer our tests; |
• | change our business processes; or |
• | pay substantial damages, court costs and attorneys' fees, including potentially increased damages for any infringement held to be willful. |
The development of new, more cost-effective solutions that can be performed by our customers or by patients, and the continued internalization of testing by hospitals or physicians, could negatively impact our testing volume and revenues.
The diagnostic information services industry is faced with changing technology and new product introductions, including technology that enables more convenient or cost-effective testing. Competitors also may offer testing to be performed outside of a commercial clinical laboratory, such as (1) point-of-care testing that can be performed by physicians in their offices; (2) complex testing that can be performed by hospitals in their own laboratories; and (3) home testing that can be carried out without requiring the services of outside providers. Advances in technology also may lead to the need for less frequent testing. Further, diagnostic tests approved or cleared by the FDA for home use are automatically deemed to be “waived” tests under CLIA and may be performed by patients in their homes; test kit manufacturers could seek to increase sales to patients of such test kits.
Some traditional customers for anatomic pathology services, including specialty physicians that generate biopsies through surgical procedures, such as dermatologists, gastroenterologists, urologists and oncologists, have added in-office histology labs or have retained pathologists to read cases on site. Hospitals also are internalizing clinical laboratory testing, including some esoteric testing. Internalization of testing may reduce demand for services previously referred to outside service providers, such as the Company.
Our outstanding debt may impair our financial and operating flexibility.
As of December 31, 2015, we had approximately $3.7 billion of debt outstanding. Except for operating leases, we do not have any off-balance sheet financing arrangements in place or available. Our debt agreements contain various restrictive covenants. These restrictions could limit our ability to use operating cash flow in other areas of our business because we must use a portion of these funds to make principal and interest payments on our debt. We have obtained ratings on our debt from Standard and Poor's, Moody's Investor Services and Fitch Ratings. There can be no assurance that any rating so assigned will remain for any given period of time or that a rating will not be lowered or withdrawn entirely by a rating agency if in that rating agency's judgment future circumstances relating to the basis of the rating, such as adverse changes in our Company or our industry, so warrant. If such ratings are lowered, our borrowing costs could increase. Changes in our credit ratings, however, do not require repayment or acceleration of any of our debt.
35
We or our subsidiaries may incur additional indebtedness in the future. Our ability to make principal and interest payments will depend on our ability to generate cash in the future. If we incur additional debt, a greater portion of our cash flows may be needed to satisfy our debt service obligations and if we do not generate sufficient cash to meet our debt service requirements, we may need to seek additional financing. In that case, it may be more difficult, or we may be unable, to obtain financing on terms that are acceptable to us. As a result, we would be more vulnerable to general adverse economic, industry and capital markets conditions as well as the other risks associated with indebtedness.
Our ability to attract and retain qualified employees is critical to the success of our business and the failure to do so may materially adversely affect our performance.
Our people are a critical resource. The supply of qualified personnel may be limited and competition for qualified employees is strong. We may lose, or fail to attract and retain, key management personnel, or qualified skilled technical or professional employees (e.g., pathologists) at our clinical laboratories or research centers.
Failure to establish, and perform to, appropriate quality standards to assure that the highest level of quality is observed in the performance of our diagnostic information services could adversely affect the results of our operations and adversely impact our reputation.
The provision of diagnostic information services involves certain inherent risks. The services that we provide are intended to provide information for healthcare providers in providing patient care. Therefore, users of our services may have a greater sensitivity to errors than the users of services or products that are intended for other purposes.
Negligence in performing our services can lead to injury or other adverse events. We may be sued under physician liability or other liability law for acts or omissions by our pathologists, laboratory personnel and hospital employees who are under the supervision of our hospital-based pathologists. We are subject to the attendant risk of substantial damages awards and risk to our reputation.
Our operations and reputation may be impaired if we do not comply with privacy laws or information security policies.
In our business, we generate or maintain sensitive information, such as patient data and other personal information. If we do not adequately safeguard that information and it were to become available to persons or entities that should not have access to it, our business could be impaired, our reputation could suffer and we could be subject to fines, penalties and litigation.
We are subject to numerous political, legal, operational and other risks as a result of our international operations which could impact our business in many ways.
Although we conduct most of our business in the United States, our international operations increase our exposure to the inherent risks of doing business in international markets. Depending on the market, these risks include without limitation:
• | changes in the local economic environment; |
• | political instability; |
• | social changes; |
• | intellectual property legal protections and remedies; |
• | trade regulations; |
• | procedures and actions affecting approval, production, pricing, reimbursement and marketing of services; |
• | exchange controls; |
• | attracting and retaining qualified employees; |
• | local market practices; |
• | export and import controls; |
• | weak legal systems which may affect our ability to enforce contractual rights; |
• | changes in local laws or regulations; and |
• | potentially longer payment and collection cycles. |
International operations also require us to devote significant management resources to implement our controls and systems in new markets, to comply with the U.S. Foreign Corrupt Practices Act and similar anti-corruption laws in non-U.S. jurisdictions and to overcome challenges based on differing languages and cultures.
36
Our operations may be adversely impacted by the effects of natural disasters such as hurricanes and earthquakes, health pandemics, hostilities or acts of terrorism and other criminal activities.
Our operations may be adversely impacted by the effects of natural disasters such as hurricanes and earthquakes, health pandemics, hostilities or acts of terrorism or other criminal activities. Such events may result in a temporary decline in the number of patients who seek clinical testing services or in our employees' ability to perform their job duties. In addition, such events may temporarily interrupt our ability to transport specimens, to receive materials from our suppliers or otherwise to provide our services.
Our business could be adversely impacted by adoption of new coding for tests.
The American Medical Association CPT® Editorial Panel is continuing its process of establishing billing codes to replace codes that describe procedures used in performing molecular testing and toxicology testing. The adoption of these codes is allowing payers to better determine tests being performed. This has led, and could continue to lead, to limited coverage decisions, payment denials or new procedures or conditions for payment. Health plans, Medicare contractors and Medicaid programs continue to consider or implement the new codes and issue coverage and payment decisions. Payment levels for many new codes remain largely unresolved and healthcare providers continue to address implementation of the new codes.
Our business could be adversely impacted by CMS' adoption of the new coding set for diagnoses.
CMS has adopted a new coding set for diagnosis, commonly known as ICD-10, which significantly expands the coding set for diagnoses. The new coding set was implemented October 1, 2015. Clinicians may fail to provide appropriate codes for desired tests, and our claims for reimbursement using the new codes may be denied. Further, delays in billing historically have resulted in increased costs and decreased collection of payment.
Adverse results in material litigation could have an adverse financial impact and an adverse impact on our client base and reputation.
We are involved in various legal proceedings arising in the ordinary course of business including, among other things, disputes as to intellectual property, professional liability and employee-related matters, as well as inquiries from governmental agencies and Medicare or Medicaid carriers. Some of the proceedings against us involve claims that are substantial in amount and could divert management's attention from operations. The proceedings also may result in substantial monetary damages.
Our operations may be adversely impacted by the effect of trends in the U.S. healthcare system, including healthcare utilization and increased patient financial responsibility for services.
Our operations may be adversely impacted by the effects of trends in the utilization of the healthcare system in the United States. Trends in the utilization of the U.S. healthcare system can be influenced by such factors as under-employed workers, decisions to delay medical care and increased patient financial responsibility for medical care. Declining utilization of the U.S. healthcare system may result in a decline in the number of patients who seek clinical testing services.
In the current environment, patients are encouraged to take increased interest in and responsibility for, and often are bearing increased financial responsibility for, their healthcare. Our operations also may be adversely impacted by the recent trend to increased patient responsibility for payment for healthcare services, including diagnostic information services.
CAUTIONARY FACTORS THAT MAY AFFECT FUTURE RESULTS
Some statements and disclosures in this document are forward-looking statements. Forward-looking statements include all statements that do not relate solely to historical or current facts and can be identified by the use of words such as “may,” “believe,” “will,” “expect,” “project,” “estimate,” “anticipate,” “plan” or “continue.” These forward-looking statements are based on our current plans and expectations and are subject to a number of risks and uncertainties that could cause our plans and expectations, including actual results, to differ materially from the forward-looking statements. Investors are cautioned not to unduly rely on such forward-looking statements when evaluating the information presented in this document. The following important factors could cause our actual financial results to differ materially from those projected, forecasted or estimated by us in forward-looking statements:
(a) | Heightened competition from commercial clinical testing companies, hospitals, physicians and others. |
(b) | Increased pricing pressure from customers and payers. |
(c) | A decline in economic conditions. |
37
(d) | Impact of changes in payer mix, including any shift from fee-for-service to discounted or capitated fee arrangements. |
(e) | Adverse actions by government or other third-party payers, including healthcare reform that focuses on reducing healthcare costs but does not recognize the value and importance to healthcare of clinical testing, unilateral reduction of fee schedules payable to us, competitive bidding, and an increase in the practice of negotiating for exclusive arrangements that involve aggressively priced capitated or fee-for-service payments by health insurers or other payers. |
(f) | The impact upon our testing volume and collected revenue or general or administrative expenses resulting from our compliance with Medicare and Medicaid administrative policies and requirements of third-party payers. These include: |
(1) the requirements of Medicare carriers to provide diagnosis codes for many commonly ordered tests and the possibility that third-party payers will increasingly adopt similar requirements;
(2) | inability to obtain from patients a valid advance beneficiary notice form for tests that cannot be billed without prior receipt of the form; |
(3) | increased challenges in operating as a non-contracted provider with respect to health plans; |
(4) | the impact of additional or expanded limited coverage policies and limits on the allowable number of test units; and |
(5) | the impact of increased prior authorization programs for clinical testing. |
(g) | Adverse results from pending or future government investigations, lawsuits or private actions. These include, in particular, monetary damages, loss or suspension of licenses, and/or suspension or exclusion from the Medicare and Medicaid programs and/or criminal penalties. |
(h) | Failure to efficiently integrate acquired businesses and to manage the costs related to any such integration, or to retain key technical, professional or management personnel. |
(i) | Denial, suspension or revocation of CLIA certification or other licenses for any of our clinical laboratories under the CLIA standards, revocation or suspension of the right to bill the Medicare and Medicaid programs or other adverse regulatory actions by federal, state and local agencies. |
(j) | Changes in federal, state or local laws or regulations, including changes that result in new or increased federal or state regulation of commercial clinical laboratories, tests developed by commercial clinical laboratories or other products or services that we offer or activities in which we are engaged, including regulation by the FDA. |
(k) | Inability to achieve expected benefits from our acquisitions of other businesses. |
(l) | Inability to achieve additional benefits from our business performance tools and efficiency initiatives. |
(m) | Adverse publicity and news coverage about the clinical testing industry or us. |
(n) | Computer or other IT system failures that affect our ability to perform testing, report test results or properly bill customers, or result in the disclosure of confidential information, including potential failures resulting from implementing common IT systems and other system conversions, telecommunications failures, malicious human acts (such as electronic break-ins or computer viruses) or natural disasters. |
(o) | Development of technologies that substantially alter the practice of clinical testing, including technology changes that lead to the development of more convenient or cost-effective testing, or testing to be performed outside of a commercial clinical laboratory, such as (1) point-of-care testing that can be performed by physicians in their offices, (2) esoteric testing that can be performed by hospitals in their own laboratories or (3) home testing that can be carried out without requiring the services of clinical laboratories. |
(p) | Negative developments regarding intellectual property and other property rights that could prevent, limit or interfere with our ability to develop, perform or sell our tests or operate our business. These include: |
(1) | Issuance of patents or other property rights to our competitors or others; and |
(2) | Inability to obtain or maintain adequate patent or other proprietary rights for our products and services or to successfully enforce our proprietary rights. |
(q) | Development of tests by our competitors or others which we may not be able to license, or usage of our technology or similar technologies or our trade secrets or other intellectual property by competitors, any of which could negatively affect our competitive position. |
(r) | Regulatory delay or inability to commercialize newly developed or licensed tests or technologies or to obtain appropriate reimbursements for such tests. |
(s) | Inability to properly bill for our services or to obtain appropriate payments for services that we do bill. |
(t) | Changes in interest rates and changes in our credit ratings from Standard & Poor's, Moody's Investor Services or Fitch Ratings causing an unfavorable impact on our cost of and access to capital. |
(u) | Inability to hire and retain qualified personnel or the loss of the services of one or more of our key senior management personnel. |
(v) | Terrorist and other criminal activities, hurricanes, earthquakes or other natural disasters, and health pandemics, which could affect our customers, transportation or systems, or our facilities, and for which insurance may not adequately reimburse us. |
38
(w) | Difficulties and uncertainties in the discovery, development, regulatory environment and/or marketing of new services or solutions or new uses of existing tests. |
(x) | Failure to adapt to changes in the healthcare system and healthcare delivery, including those stemming from the 2010 federal healthcare reform legislation. |
(y) | Results and consequences of governmental inquiries. |
(z) Trends in utilization of the healthcare system.
(aa) Increased patient financial responsibility for services.
(bb) Difficulty in implementing, or lack of success with, our strategic plan.
(cc) | Inability to adapt to diverse and dynamic non-U.S. markets. |
(dd) The impact of informatics on our industry and the ability of our Company to adapt to that impact.
Item 1B. Unresolved Staff Comments
There are no unresolved SEC comments that require disclosure.
Item 2. Properties
Our executive offices are located in Madison, New Jersey. We maintain clinical testing laboratories throughout the continental United States; in several instances a joint venture of which we are a partner maintains the laboratory. We also maintain offices, data centers, billing centers, call centers, distribution centers and patient service centers at locations throughout the United States. In addition, we maintain offices, patient service centers and clinical laboratories in locations outside the United States, including in Puerto Rico, Mexico, India and Ireland. Our properties that are not owned are leased on terms and for durations that are reflective of commercial standards in the communities where these properties are located. We believe that, in general, our facilities are suitable and adequate for our current and anticipated future levels of operation and are adequately maintained. We believe that if we were unable to renew a lease on any of our facilities, we could find alternative space at competitive market rates and relocate our operations to such new location without material disruption to our business. Several of our principal facilities are highlighted below.
Location | Leased or Owned | |
Sacramento, California (laboratory) | Leased | |
West Hills, California (laboratory) | Leased | |
San Juan Capistrano, California (laboratory) | Owned | |
Tampa, Florida (laboratory) | Owned | |
Atlanta, Georgia (laboratory) | Owned | |
Chicago, Illinois (2) (laboratories) | One owned, one leased | |
Marlborough, Massachusetts (laboratories) | Leased | |
Baltimore, Maryland (laboratory) | Owned | |
Teterboro, New Jersey (laboratory) | Owned | |
Philadelphia, Pennsylvania (laboratory) | Leased | |
Norristown, Pennsylvania (offices) | Leased | |
Dallas, Texas (laboratory) | Leased | |
Chantilly, Virginia (laboratory) | Leased | |
Lenexa, Kansas (laboratory) | Owned | |
Greensboro, North Carolina (laboratory) | Leased | |
Item 3. Legal Proceedings
See Note 17 to the Consolidated Financial Statements (Part II, Item 8 of this Report) for information regarding legal proceedings in which we are involved.
Item 4. Mine Safety Disclosures
Not applicable.
39
PART II
Item 5. Market for Registrant's Common Stock, Related Stockholder Matters and Issuer Purchases of Equity Securities
Our common stock is listed and traded on the New York Stock Exchange under the symbol “DGX.” As of February 1, 2016, we had approximately 2,800 record holders of our common stock; we believe that the number of beneficial holders of our common stock exceeds the number of record holders. The following table sets forth, for the periods indicated, the high and low sales price per share as reported on the New York Stock Exchange Consolidated Tape and dividend information.
Common Stock Market Price | Dividends Declared | ||||||||||
High | Low | ||||||||||
2014 | |||||||||||
First Quarter | $ | 60.50 | $ | 50.46 | $ | 0.33 | |||||
Second Quarter | 62.42 | 54.90 | 0.33 | ||||||||
Third Quarter | 64.38 | 58.56 | 0.33 | ||||||||
Fourth Quarter | 68.51 | 56.27 | 0.33 | ||||||||
2015 | |||||||||||
First Quarter | $ | 78.33 | $ | 66.09 | $ | 0.38 | |||||
Second Quarter | 89.00 | 69.47 | 0.38 | ||||||||
Third Quarter | 75.25 | 60.07 | 0.38 | ||||||||
Fourth Quarter | 72.43 | 60.15 | 0.38 | ||||||||
We currently expect that comparable cash dividends will continue to be paid in the future.
In January 2016, we declared a common stock dividend of $0.40 per common share, payable in April 2016.
The table below sets forth the information with respect to purchases made by or on behalf of the Company of its common stock during the fourth quarter of 2015.
ISSUER PURCHASES OF EQUITY SECURITIES | ||||||||||||||
Period | Total Number of Shares Purchased | Average Price Paid per Share | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs | Approximate Dollar Value of Shares that May Yet Be Purchased Under the Plans or Programs (in thousands) | ||||||||||
October 1, 2015 – October 31, 2015 | ||||||||||||||
Share Repurchase Program (A) | — | $ | — | — | $ | 522,116 | ||||||||
Employee Transactions (B) | 809 | $ | 62.24 | N/A | N/A | |||||||||
November 1, 2015 – November 30, 2015 | ||||||||||||||
Share Repurchase Program (A) | 733,817 | $ | 68.14 | 733,817 | $ | 472,116 | ||||||||
Employee Transactions (B) | 431 | $ | 66.56 | N/A | N/A | |||||||||
December 1, 2015 – December 31, 2015 | ||||||||||||||
Share Repurchase Program (A) | — | $ | — | — | $ | 972,116 | ||||||||
Employee Transactions (B) | 4,121 | $ | 67.51 | N/A | N/A | |||||||||
Total | ||||||||||||||
Share Repurchase Program (A) | 733,817 | $ | 68.14 | 733,817 | $ | 972,116 | ||||||||
Employee Transactions (B) | 5,361 | $ | 66.64 | N/A | N/A | |||||||||
40
(A) | Since the share repurchase program's inception in May 2003, our Board of Directors has authorized $7.0 billion of share repurchases of our common stock through December 31, 2015. The share repurchase authority has no set expiration or termination date. |
(B) | Includes: (1) shares delivered or attested to in satisfaction of the exercise price and/or tax withholding obligations by holders of stock options (granted under the Company's Amended and Restated Employee Long-Term Incentive Plan) who exercised options; and (2) shares withheld (under the terms of grants under the Long-Term Incentive Plan) to offset tax withholding obligations that occur upon the delivery of outstanding common shares underlying restricted share units and performance share units. |
Performance Graph
Set forth below is a line graph comparing the cumulative total shareholder return on Quest Diagnostics' common stock since December 31, 2010 based on the market price of the Company's common stock and assuming reinvestment of dividends, with the cumulative total shareholder return of companies on the Standard & Poor's 500 Stock Index and the S&P 500 Healthcare Equipment & Services Index.
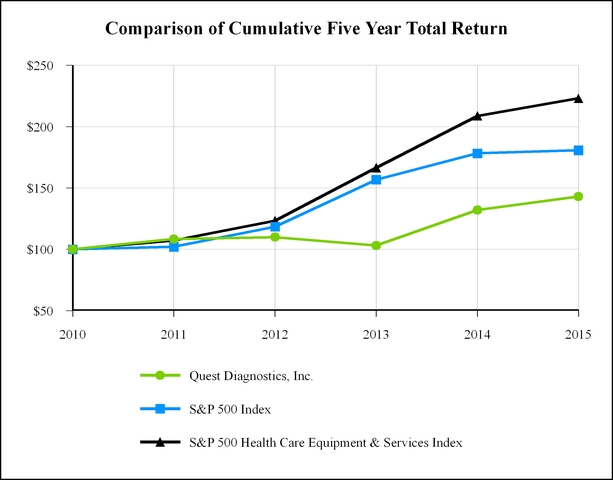
Closing DGX Price | Total Shareholder Return | Performance Graph Values | |||||||||||||||||||||
Date | DGX | S&P 500 | S&P 500 H.C. | DGX | S&P 500 | S&P 500 H.C. | |||||||||||||||||
12/31/2011 | $58.06 | 8.33 | % | 2.11 | % | 7.21 | % | $ | 108.33 | $ | 102.11 | $ | 107.21 | ||||||||||
12/30/2012 | $58.27 | 1.49 | % | 16.00 | % | 15.02 | % | $ | 109.94 | $ | 118.45 | $ | 123.31 | ||||||||||
12/31/2013 | $53.54 | (6.24 | )% | 32.39 | % | 35.05 | % | $ | 103.08 | $ | 156.82 | $ | 166.53 | ||||||||||
12/31/2014 | $67.06 | 28.06 | % | 13.69 | % | 25.34 | % | $ | 132.00 | $ | 178.28 | $ | 208.73 | ||||||||||
12/31/2015 | $71.14 | 8.35 | % | 1.38 | % | 6.89 | % | $ | 143.03 | $ | 180.75 | $ | 223.11 | ||||||||||
41
Item 6. Selected Financial Data
See page 48. |
Item 7. Management's Discussion and Analysis of Financial Condition and Results of Operations
See page 52. |
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
See Management's Discussion and Analysis of Financial Condition and Results of Operations.
Item 8. Financial Statements and Supplementary Data
See Item 15(a)1 and Item 15(a)2.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Conclusion Regarding Effectiveness of Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our Chief Executive Officer and our Chief Financial Officer, we have evaluated the effectiveness of our disclosure controls and procedures (as defined under Rules 13a-15(e) and 15d-15(e) of the Securities Exchange Act of 1934, as amended). Based upon that evaluation, our Chief Executive Officer and our Chief Financial Officer concluded that our disclosure controls and procedures were effective as of the end of the period covered by this annual report.
Management's Report on Internal Control Over Financial Reporting
See page 71. |
Changes in Internal Control
During the fourth quarter of 2015, there were no changes in our internal control over financial reporting (as defined in Rule 13a-15(f) under the Securities Exchange Act of 1934, as amended) that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
On November 5, 2014, the Company announced that it was developing a new multi-year program designed to drive operational excellence and reduce the Company’s cost structure by an additional $600 million in run rate savings as it exits 2017. The Company identified key opportunities to change how it operates, in order to meet this goal. These opportunities include: standardizing the Company's processes, information technology systems, equipment and data; enhancing electronic enabling services; and enhancing reimbursement for work the Company performs.
On January 26, 2015, the Company adopted a course of action related to this multi-year program. The Company has developed a high-level estimate of the pre-tax charges expected to be incurred in connection with the course of action for the program: $300 million.
During 2015, the cumulative charges recorded in connection with the 2015 course of action were $89 million. On February 26, 2016, the Company developed high-level estimates of the pre-tax charges expected to be incurred in connection with the course of action for 2016: $80 million to $100 million, consisting of up to $10 million of employee separation costs and $80 million to $90 million of systems conversion and integration costs. Principally all of the total estimated pre-tax charges expected to be
42
incurred in 2016 are anticipated to result in cash expenditures. The actual charges incurred in connection with the course of action in 2016 could be materially different from these estimates.
As detailed plans to implement the course of action are approved and executed, it will result in charges to earnings.
43
PART III
Item 10. Directors, Executive Officers and Corporate Governance
Our Code of Business Ethics applies to all employees, executive officers and directors, including our Chief Executive Officer, Chief Financial Officer and Corporate Controller. You can find our Code of Business Ethics on our corporate governance website, www.QuestDiagnostics.com/governance. We will post any amendments to the Code of Business Ethics, and any waivers that are required to be disclosed by the rules of either the SEC or the New York Stock Exchange, on our website.
Information regarding the Company's executive officers is contained in Part I, Item 1 of this Report under “Executive Officers of the Company.” Information regarding the directors and executive officers of the Company appearing in our Proxy Statement to be filed by April 29, 2016 (“Proxy Statement”) under the captions “Proposal No. 1 - Election of Directors,” “Information about our Corporate Governance - Director Independence,” “Information about our Corporate Governance - Board Committees,” and “Information about our Corporate Governance - Audit and Finance Committee” and "Additional Information Regarding Executive Compensation - Section 16(a) Beneficial Ownership Reporting Compliance" is incorporated by reference herein.
Item 11. Executive Compensation
Information appearing in our Proxy Statement under the captions “2015 Director Compensation Table,” “Compensation Discussion and Analysis,” “Information Regarding Executive Compensation” and “Report of the Compensation Committee” is incorporated by reference herein.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholders' Matters
Information regarding security ownership of certain beneficial owners and management appearing in our Proxy Statement under the captions “Stock Ownership Information” and "Information Regarding Executive Compensation - Equity Compensation Plan Information" is incorporated by reference herein.
Item 13. Certain Relationships and Related Transactions, and Director Independence
Information regarding certain relationships and related transactions appearing in our Proxy Statement under the captions “Information about our Corporate Governance - Related Person Transactions” and “Information about our Corporate Governance - Director Independence” is incorporated by reference herein.
Item 14. Principal Accounting Fees and Services
Information regarding principal accountant fees and services appearing in our Proxy Statement under the caption “Audit" (excluding the information under the subheading “Report of the Audit and Finance Committee”) is incorporated by reference herein.
44
PART IV
Item 15. Exhibits, Financial Statement Schedules
(a) | Documents filed as part of this Report. |
1. | Index to financial statements and supplementary data filed as part of this Report. |
Item | Page |
Financial Statements | |
2. | Financial Statement Schedule. |
3. | Exhibits |
An exhibit index has been filed as part of this Report beginning on page E-1 and is incorporated herein by reference.
(b) | Exhibits filed as part of this Report. |
An exhibit index has been filed as part of this Report beginning on page E-1 and is incorporated herein by reference.
(c) | None. |
45
Signatures
Pursuant to the requirements of Sections 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized, on February 26, 2016.
QUEST DIAGNOSTICS INCORPORATED | ||
(Registrant) | ||
By: | /s/Stephen H. Rusckowski | |
Stephen H. Rusckowski | ||
President and Chief Executive Officer | ||
Each individual whose signature appears below constitutes and appoints Michael E. Prevoznik and William J. O'Shaughnessy, Jr., and each of them singly, his or her true and lawful attorneys-in-fact and agents with full power of substitution, for him or her and in his or her name, place and stead, in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K filed with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all the said attorneys-in-fact and agents or any of them or their or his or her substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities indicated on February 26, 2016.
46
Signature | Capacity | |
/s/Stephen H. Rusckowski Stephen H. Rusckowski | Director, President and Chief Executive Officer (Principal Executive Officer) | |
/s/Mark J. Guinan Mark J. Guinan | Senior Vice President and Chief Financial Officer (Principal Financial Officer) | |
/s/Robert A. Klug Robert A. Klug | Vice President, Corporate Controller and Chief Accounting Officer (Principal Accounting Officer) | |
/s/John C. Baldwin, M.D. John C. Baldwin, M.D. | Director | |
/s/Jenne K. Britell, Ph.D. Jenne K. Britell, Ph.D. | Director | |
/s/Vicky B. Gregg Vicky B. Gregg | Director | |
/s/Jeffrey M. Leiden, M.D., Ph. D. Jeffrey M. Leiden, M.D., Ph. D. | Director | |
/s/Timothy L. Main Timothy L. Main | Director | |
/s/Gary M. Pfeiffer Gary M. Pfeiffer | Director | |
/s/Timothy M. Ring Timothy M. Ring | Director | |
/s/Daniel C. Stanzione, Ph.D. Daniel C. Stanzione, Ph.D. | Chairman of the Board | |
/s/Gail R. Wilensky, Ph.D. Gail R. Wilensky, Ph.D. | Director | |
/s/John B. Ziegler John B. Ziegler | Director | |
47
SELECTED HISTORICAL FINANCIAL DATA OF OUR COMPANY
The following table summarizes selected historical financial data of our Company and our subsidiaries at the dates and for each of the periods presented. We derived the selected historical financial data for the years 2011 through 2015 from the audited consolidated financial statements of our Company. During the fourth quarter of 2012, we sold our OralDNA salivary diagnostics business, and committed to a plan to sell our HemoCue diagnostic products business. The sale of HemoCue was completed in April 2013. During the third quarter of 2006, we completed the wind down of NID, a test kit manufacturing subsidiary. As a result, the operations for HemoCue, OralDNA and NID have been classified as discontinued operations for all periods presented. At December 31, 2012, the assets and liabilities of HemoCue were reported as held for sale. The selected historical financial data is only a summary and should be read together with the audited consolidated financial statements and related notes of our Company and management's discussion and analysis of financial condition and results of operations included elsewhere in this Annual Report on Form 10-K.
Year Ended December 31, | |||||||||||||||||||
2015 | 2014 | 2013 | 2012 | 2011 | |||||||||||||||
(dollars in millions, except per share data) | |||||||||||||||||||
Operations Data: | (a) (b) | (c) (d) | (e) (f) | (g) (h) | (i) (j) | ||||||||||||||
Net revenues | $ | 7,493 | $ | 7,435 | $ | 7,146 | $ | 7,383 | $ | 7,392 | |||||||||
Operating income | 1,399 | 983 | 1,475 | 1,201 | 987 | ||||||||||||||
Income from continuing operations | 753 | 587 | 848 | 666 | 494 | ||||||||||||||
Income (loss) from discontinued operations, net of taxes | — | 5 | 35 | (74 | ) | 12 | |||||||||||||
Net income | 753 | 592 | 883 | 592 | 506 | ||||||||||||||
Less: Net income attributable to noncontrolling interests | 44 | 36 | 34 | 36 | 35 | ||||||||||||||
Net income attributable to Quest Diagnostics | $ | 709 | $ | 556 | $ | 849 | $ | 556 | $ | 471 | |||||||||
Amounts attributable to Quest Diagnostics' stockholders: | |||||||||||||||||||
Income from continuing operations | $ | 709 | $ | 551 | $ | 814 | $ | 630 | $ | 459 | |||||||||
Income (loss) from discontinued operations, net of taxes | — | 5 | 35 | (74 | ) | 12 | |||||||||||||
Net income | $ | 709 | $ | 556 | $ | 849 | $ | 556 | $ | 471 | |||||||||
Earnings per share attributable to Quest Diagnostics' common stockholders - basic: | |||||||||||||||||||
Income from continuing operations | $ | 4.92 | $ | 3.80 | $ | 5.35 | $ | 3.96 | $ | 2.88 | |||||||||
Income (loss) from discontinued operations | — | 0.03 | 0.23 | (0.47 | ) | 0.07 | |||||||||||||
Net income | $ | 4.92 | $ | 3.83 | $ | 5.58 | $ | 3.49 | $ | 2.95 | |||||||||
Earnings per share attributable to Quest Diagnostics' common stockholders - diluted: | |||||||||||||||||||
Income from continuing operations | $ | 4.87 | $ | 3.78 | $ | 5.31 | $ | 3.92 | $ | 2.85 | |||||||||
Income (loss) from discontinued operations | — | 0.03 | 0.23 | (0.46 | ) | 0.07 | |||||||||||||
Net income | $ | 4.87 | $ | 3.81 | $ | 5.54 | $ | 3.46 | $ | 2.92 | |||||||||
Dividends per common share | $ | 1.52 | $ | 1.32 | $ | 1.20 | $ | 0.81 | $ | 0.47 | |||||||||
48
Year Ended December 31, | |||||||||||||||||||
2015 | 2014 | 2013 | 2012 | 2011 | |||||||||||||||
(dollars in millions) | |||||||||||||||||||
Balance Sheet Data (at end of year): | (a) (b) | (c) (d) | (e) (f) | (g) (h) | (i) (j) | ||||||||||||||
Cash and cash equivalents | $ | 133 | $ | 192 | $ | 187 | $ | 296 | $ | 165 | |||||||||
Total assets | 9,962 | 9,857 | 8,930 | 9,263 | 9,288 | ||||||||||||||
Long-term debt | 3,492 | 3,224 | 3,102 | 3,333 | 3,346 | ||||||||||||||
Total debt | 3,651 | 3,742 | 3,314 | 3,343 | 4,000 | ||||||||||||||
Other Data: | |||||||||||||||||||
Net cash provided by operating activities | $ | 810 | $ | 938 | $ | 652 | $ | 1,187 | $ | 895 | |||||||||
Net cash (used in) provided by investing activities | (362 | ) | (1,025 | ) | 328 | (217 | ) | (1,243 | ) | ||||||||||
Net cash (used in) provided by financing activities | (507 | ) | 92 | (1,106 | ) | (822 | ) | 64 | |||||||||||
Capital expenditures | 263 | 308 | 231 | 182 | 161 | ||||||||||||||
Purchases of treasury stock | 224 | 132 | 1,037 | 200 | 935 | ||||||||||||||
Dividends paid | 212 | 187 | 185 | 108 | 65 | ||||||||||||||
(a) | On August 3, 2015, we completed the acquisition of MemorialCare Health System's laboratory outreach business ("MemorialCare"). On November 16, 2015, the Company completed the acquisition of the business assets of Superior Mobile Medics, Inc. ("Superior Mobile Medics"). Consolidated operating results for 2015 include the results of operations of MemorialCare and Superior Mobile Medics subsequent to the closing of the applicable acquisition. In July 2015, we contributed our clinical trials testing business to a newly formed global clinical trials central laboratory services joint venture with Quintiles Transnational Holdings Inc., Q2 Solutions ("Clinical Trials Contribution"). For further details regarding our acquisitions and dispositions, see Note 5 and Note 6, respectively, to the consolidated financial statements. |
(b) | Operating income includes a pre-tax gain of $334 million associated with the Clinical Trials Contribution. In addition, operating income includes pre-tax charges of $105 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating our business. Operating income also includes net pre-tax charges of $27 million primarily associated with non-cash asset impairment charges associated with our Celera Products business and the winding down of a subsidiary, costs incurred related to legal matters and a pre-tax gain of $13 million associated with a decrease in the fair value of the contingent consideration accrual associated with our Summit Health acquisition. |
Income from continuing operations includes $144 million of pre-tax charges on retirement of debt associated with the March 2015 cash tender offer and the April 2015 redemption and the related income tax benefit of $57 million. In addition, income from continuing operations includes deferred tax expense of $145 million associated with the gain on the Clinical Trials Contribution, partially offset by the recognition of a $58 million deferred tax benefit associated with winding down a subsidiary. For further details regarding our retirement of debt, see Note 13 to the consolidated financial statements.
Net cash provided by operating activities includes $146 million of pre-tax cash charges, or $89 million after the related cash tax benefit, associated with the early retirement of debt. In addition, net cash provided by operating activities includes payments associated with an additional payroll cycle in 2015 and an income tax payment in the third quarter of 2015 associated with certain tax contingencies.
Net cash used in investing activities includes a $33 million investment in Q2 Solutions.
Net cash used in financing activities includes $51 million of deferred acquisition consideration payments, primarily to UMass Memorial Medical Center ("UMass"), related to the business acquisition in 2013, and $63 million of proceeds from the sale of a noncontrolling interest in a subsidiary to UMass.
(c) | On March 7, 2014, we completed the acquisition of Solstas Lab Partners Group ("Solstas"). On April 18, 2014, we completed the acquisition of Summit Health, Inc. ("Summit Health"). On April 16, 2014, we completed the acquisition of the outreach laboratory service operations of Steward Healthcare, LLC ("Steward"). Consolidated operating results for 2014 include the results of operations of Solstas, Summit Health and Steward subsequent to the closing of the applicable acquisition. For further details regarding our acquisitions, see Note 5 to the consolidated financial statements. |
49
(d) | Operating income includes pre-tax charges of $121 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating our business. In addition, operating income includes pre-tax charges of $24 million principally associated with costs related to legal matters, partially offset by a pre-tax gain of $9 million associated with a decrease in the fair value of the contingent consideration accrual associated with our Summit Health acquisition. |
Income from continuing operations includes discrete income tax benefits of $44 million associated with the favorable resolution of certain tax contingencies.
(e) | On January 2, 2013, we completed the acquisition of the clinical outreach and anatomic pathology businesses of UMass. On May 15, 2013, we completed the acquisition of the toxicology and clinical laboratory business of Advanced Toxicology Network ("ATN") from Concentra, a subsidiary of Humana Inc. On June 22, 2013, we completed the acquisition of certain lab-related clinical outreach service operations of Dignity Health ("Dignity"), a hospital system in California. On October 7, 2013, we completed the acquisition of ConVerge Diagnostic Services, LLC ("ConVerge"), a leading full-service laboratory providing clinical, cytology and anatomic pathology testing services to patients, physicians and hospitals in New England. Consolidated operating results for 2013 include the results of operations of UMass, ATN, Dignity and ConVerge subsequent to the closing of the applicable acquisition. For further details regarding our acquisitions, see Note 5 to the consolidated financial statements. |
(f) | Operating income includes pre-tax charges of $115 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating our business. In addition, operating income includes a pre-tax gain on sale of royalty rights of $474 million and a pre-tax loss of $40 million associated with the sale of the Enterix. For further details regarding the sale of royalty rights and Enterix, see Note 6 to the consolidated financial statements. |
Income (loss) from discontinued operations, net of taxes includes a gain of $14 million (including foreign currency translation adjustments, partially offset by income tax expense and transaction costs) associated with the sale of HemoCue. In addition, income (loss) from discontinued operations, net of taxes includes discrete tax benefits of $20 million associated with favorable resolution of certain tax contingencies related to our NID business. For further details regarding our discontinued operations, see Note 18 to the consolidated financial statements.
Net cash provided by operating activities includes income tax payments of $175 million associated with the sale of royalty rights. In addition, it includes approximately $70 million of income tax payments which were deferred from the fourth quarter of 2012 under a program offered to companies whose principal place of business was in states most affected by Hurricane Sandy.
Net cash provided by investing activities includes proceeds from the sale of the ibrutinib royalty rights of $474 million, net of transaction costs, as well as proceeds from the sales of HemoCue and Enterix of $296 million.
(g) | On January 6, 2012, we completed the acquisition of S.E.D. Medical Laboratories ("S.E.D.") from Lovelace Health System. Consolidated operating results for 2012 include the results of operations of S.E.D. subsequent to the closing of the acquisition. |
(h) | Operating income includes $106 million of pre-tax charges incurred in conjunction with further restructuring and integrating our business. Results for 2012 also include pre-tax charges of $10 million, principally representing severance and other separation benefits as well as accelerated vesting of certain equity awards in connection with the succession of our prior CEO. In addition, we estimate that the impact of severe weather during the fourth quarter of 2012 adversely affected operating income for 2012 by approximately $16 million. |
Income (loss) from discontinued operations, net of taxes includes charges for the asset impairment associated with HemoCue and the loss on sale associated with OralDNA totaling $86 million. Discontinued operations also includes a $8 million income tax expense related to the re-valuation of deferred tax assets associated with HemoCue and a $4 million income tax benefit related to the remeasurement of deferred taxes associated with HemoCue as a result of an enacted income tax rate change in Sweden. In February 2013, we entered into an agreement to sell HemoCue. The sale of HemoCue was completed in April 2013. For further details regarding our discontinued operations, see Note 18 to the consolidated financial statements.
50
Net cash provided by operating activities includes receipts of $72 million from the termination of certain interest rate swap agreements and the deferral of approximately $70 million of income tax payments into the first quarter of 2013, which was offered to companies whose principal place of business was in states most affected by Hurricane Sandy.
(i) | On April 4, 2011, we completed the acquisition of Athena Diagnostics (“Athena”). On May 17, 2011, we completed the acquisition of Celera Corporation (“Celera”). Consolidated operating results for 2011 include the results of operations of Athena and Celera subsequent to the closing of the applicable acquisition. |
(j) | Operating income includes a pre-tax charge to earnings in the first quarter of 2011 of $236 million which represented the cost to resolve a previously disclosed civil lawsuit brought by a California competitor in which the State of California intervened (the “California Lawsuit”). Results for 2011 also include $52 million of pre-tax charges incurred in conjunction with further restructuring and integrating our business, consisting of $42 million of pre-tax charges principally associated with workforce reductions, with the remainder principally professional fees. Results for 2011 also include $17 million of pre-tax transaction costs, primarily related to professional fees, associated with the acquisitions of Athena and Celera. In addition, operating income includes pre-tax charges of $6 million, principally representing severance and other separation benefits as well as accelerated vesting of certain equity awards in connection with the succession of our prior CEO. In addition, we estimate that the impact of severe weather during the first quarter of 2011 adversely affected operating income for 2011 by $19 million. |
Income from continuing operations includes $3 million of pre-tax financing related transaction costs associated with the acquisition of Celera, a $3 million pre-tax gain associated with the sale of an investment, and $18 million of discrete income tax benefits, primarily associated with certain state tax planning initiatives and the favorable resolution of certain tax contingencies.
Net cash provided by operating activities includes payments associated with the settlement of the California Lawsuit, restructuring and integration costs, and transaction costs associated with the acquisitions of Athena and Celera totaling $320 million, or $202 million net of an associated reduction in estimated tax payments.
51
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Overview
Our Company
Diagnostic Information Services
Quest Diagnostics empowers people to take action to improve health outcomes. We use our extensive database of clinical lab results to derive diagnostic insights that reveal new avenues to identify and treat disease, inspire healthy behaviors and improve healthcare management. Our diagnostic information services business ("DIS") provides insights through clinical testing and related services to patients, physicians, hospitals, accountable care organizations ("ACOs"), integrated delivery networks ("IDNs"), health plans, employers and others. We offer the broadest access in the United States to diagnostic information services through our nationwide network of laboratories, Company-owned patient service centers and phlebotomists in physician offices. We are the world's leading provider of diagnostic information services, which includes providing clinical testing services such as routine (including drugs-of-abuse) testing, gene-based and esoteric testing, and anatomic pathology services, as well as related services and insights. The Company provides interpretive consultation with one of the largest medical and scientific staffs in the industry. Our DIS business makes up over 90% of our consolidated net revenues.
The clinical testing that we perform is an essential element in the delivery of healthcare services. Clinicians use clinical testing for predisposition, screening, monitoring, diagnosis, prognosis and treatment choices of diseases and other medical conditions. The United States clinical testing industry consists of two segments. One segment, which we believe makes up approximately 40% of the total industry, includes hospital inpatient and outpatient testing. The second segment, which we believe makes up approximately 60% of the total industry, includes testing of persons who are not hospital patients, including testing done in commercial clinical laboratories, physician-office laboratories and other locations, as well as hospital outreach (non-hospital patients) testing. We believe that hospital-affiliated laboratories account for approximately 35% of the second segment, commercial clinical laboratories approximately 53% and physician-office laboratories and other locations account for the balance.
The clinical testing industry is subject to seasonal fluctuations in operating results and cash flows. Typically, testing volume declines during vacation and major holiday periods, reducing net revenues and operating cash flows below annual averages. Testing volume is also subject to declines due to severe weather or other events, which can deter patients from having testing performed and which can vary in duration and severity from year to year. Additionally, orders for clinical testing generated from physician offices, hospitals and employers can be affected by factors such as changes in the United States economy and regulatory environment, which affect the number of unemployed and uninsured, and design changes in healthcare plans, which affect the number of physician office and hospital visits.
Diagnostic Solutions
In our Diagnostic Solutions ("DS") businesses, which represents the balance of our consolidated net revenues, we offer a variety of solutions to insurers and healthcare providers. We are the leading provider of risk assessment services for the life insurance industry as well as a leading provider of central laboratory testing for clinical trials (see discussion below regarding the contribution of our clinical trials testing business to a newly formed joint venture effective July 1, 2015). In addition, we offer healthcare organizations and clinicians robust information technology solutions and diagnostic products.
2015 Highlights
Our total net revenues of $7.5 billion were 0.8% above the prior year. The contribution of our clinical trials testing business to the clinical trials central laboratory services joint venture, Q2 Solutions, negatively impacted total net revenues by 1.2%. DIS revenues of $7.0 billion were 1.3% above the prior year. DIS volume increased 1.2% compared to the prior year. DIS revenue per requisition increased 0.1% compared to the prior year. DS revenues decreased by 6.1% compared to the prior year. The contribution of our clinical trials testing business negatively impacted DS revenue by approximately 17%. Income from continuing operations attributable to Quest Diagnostics' stockholders was $709 million, or $4.87 per diluted share, which benefited from the gain on the contribution of our clinical trials testing business to Q2 Solutions and recognition of a deferred income tax benefit associated with winding down a subsidiary. These benefits were partially offset by charges on the retirement of debt and related refinancing charges, restructuring and integration charges and other costs.
52
Five-point Strategy
We made good progress on the execution of our five-point strategy during 2015 as follows:
• | We have seen continued growth in our gene-based and esoteric testing. |
• | We acquired MemorialCare Health System's laboratory outreach services business and the business assets of Superior Mobile Medics, Inc. ("Superior Mobile Medics"), which operated in the risk assessment business. In addition, we announced that we entered into a definitive agreement to acquire the outreach laboratory service business of Clinical Laboratory Partners. |
• | We entered into an agreement to provide professional lab services for Barnabas Health, New Jersey's largest not-for-profit integrated healthcare delivery system. |
• | We announced a partnership with Inovalon to deliver our Data Diagnostics Solutions offering, as part of our new Quanum solutions suite, which generates further value from our information assets by delivering real time insights to physicians at the point of service. |
• | Our cost excellence program, Invigorate, delivered realized savings of more than $200 million in 2015. |
• | We closed on our Q2 Solutions joint venture with Quintiles Transnational Holdings Inc. ("Quintiles"), which is an important element of our growth strategy along with our other equity method investees. |
• | We completed a $1.2 billion senior notes offering and used the proceeds to retire over $1.2 billion in outstanding senior notes resulting in reduced future interest expense and an improved maturity profile. |
• | We repurchased approximately $224 million of our common stock as part of our share repurchase program and paid dividends to our shareholders of $212 million. |
• | On January 28, 2016, we announced that our Board of Directors authorized a 5% increase in our quarterly dividend from $0.38 per share to $0.40 per share, or $1.60 annually, commencing with the dividend payable in April 2016. |
For additional information on our five-point strategy, see Item 1: "Our Strategy and Strengths."
Outlook and Trends
The healthcare system in the United States is evolving; significant change is taking place in the system. We expect that the evolution of the healthcare industry will continue, and that industry change is likely to be extensive. There are a number of key trends that are having, and that we expect will continue to have, a significant impact on the diagnostic information services business in the United States and on our business. These trends present both opportunities and risks. However, because diagnostic information services is an essential healthcare service and because of the key trends discussed below, we believe that the industry will continue to grow over the long term and that we are well positioned to benefit from the long-term growth expected in the industry.
We expect to see a stable business environment in 2016, and we expect reimbursement pressure for our DIS business will be moderate through 2017. However, healthcare market participants in the United States, including governments, are focused on controlling healthcare costs through means including but not limited to shifting from fee for service to capitation, changing medical coverage policies (e.g., healthcare benefit design), pre-authorization of lab testing, requiring co-pays, introducing lab spend management utilities and payment and patient care innovations such as ACOs and patient-centered medical homes. As health plans and government programs require greater levels of patient cost-sharing, our patient collections could be negatively impacted and adversely impact our bad debt expense. As previously mentioned, there could be a shift to capitation arrangements where we agree to a predetermined monthly reimbursement rate for each member enrolled in a restricted plan, generally regardless of the number or cost of services provided by us. In 2015 and 2014, we derived approximately 11% of our testing volume and 4% and 3%, respectively, of our DIS revenues from capitated payment arrangements.
Historically, the Medicare Clinical Laboratory Fee Schedule and the Medicare Physician Fee Schedule established under Part B of the Medicare program have been subject to change, including each year. In addition, pursuant to the federal Protecting Access to Medicare Act of 2014 ("PAMA"), it is expected that the Centers for Medicare and Medicaid Services will revise reimbursement schedules for clinical laboratory testing services provided under Medicare. We expect the revised reimbursement schedules as a result of PAMA to become effective as early as 2017. In 2015, approximately 12% of our consolidated net revenues were reimbursed by Medicare under the Clinical Laboratory Fee Schedule and approximately 2% were reimbursed by Medicare under the Physician Fee Schedule.
The FDA has announced guidance initiatives that may impact the clinical laboratory testing business, including by increasing regulation of laboratory-developed tests. In addition, the trend of consolidating, converging and diversifying among
53
our customers and payers has continued. Consolidation is increasing price transparency and bargaining power, and encouraging internalization of clinical testing.
For additional information on our key trends, see Item 1: "The United States Clinical Testing Industry."
Operational Excellence
Invigorate Program
The clinical testing industry is labor intensive. Employee compensation and benefits constitute approximately one-half of our total costs and expenses. In addition, performing clinical testing involves significant fixed costs for facilities and other infrastructure required to obtain, transport and test specimens. Therefore, relatively small changes in volume can have a significant impact on profitability in the short-term.
We are engaged in a multi-year program called Invigorate, which is designed to reduce our cost structure. We delivered more than $700 million in run-rate savings as we exited 2014. In November 2014, we announced our goal to deliver an additional $600 million in run-rate savings as we exit 2017. Achieving this goal would bring the total savings from the Invigorate program to $1.3 billion in run-rate savings, compared to 2011. In 2015, we delivered realized savings of more than $200 million and believe we are on track to achieve our $1.3 billion run-rate savings goal by the end of 2017.
Invigorate has consisted of several flagship programs, with structured plans in each, to drive savings and improve performance across the customer value chain. These flagship programs include: organization excellence; information technology excellence; procurement excellence; service excellence; lab excellence; and billing excellence. In addition to these programs, we identified new key opportunities to change how we operate in order to meet our goal of delivering the additional $600 million in run-rate savings as we exit 2017. These new key opportunities include: standardizing our processes, information technology systems, equipment and data; enhancing electronic enabling services; and enhancing reimbursement for work we perform. We believe that our efforts to standardize our information technology systems, equipment and data also will foster our efforts to restore growth and support the value creation initiatives of our clinical franchises by enhancing our operational flexibility, empowering and enhancing the customer experience, facilitating the delivery of actionable insights and bolstering our large data platform.
In January 2015, we adopted a course of action related to this multi-year program. We developed a high-level estimate of the total pre-tax charges expected to be incurred in 2015 through 2017 in connection with the course of action for the program: $300 million. In February 2016, we developed high-level estimates of the pre-tax charges expected to be incurred in connection with the course of action for 2016 totaling $80 million to $100 million, consisting of up to $10 million of employee separation costs and $80 million to $90 million of systems conversion and integration costs. As detailed plans to implement the course of action are approved and executed, it will result in charges to earnings. Principally all of the total estimated pre-tax charges expected to be incurred in 2016 are anticipated to result in cash expenditures. The actual charges incurred in connection with the course of action in 2016 could be materially different from these estimates.
From 2012 through 2014, the cumulative charges recorded in connection with the Invigorate program were $266 million, including $178 million of cumulative pre-tax employee separation costs and other restructuring related costs. During 2015, the cumulative charges recorded in connection with the 2015 course of action were $89 million, including $39 million of cumulative pre-tax employee separation costs and other restructuring related costs.
For further details regarding restructuring costs related to the Invigorate program, see Note 4 to the consolidated financial statements.
Recent Transactions
Acquisitions
In 2015, we acquired MemorialCare Health System's laboratory outreach business and the business assets of Superior Mobile Medics, a national provider of paramedical and health data collection services to the life insurance and employer health and wellness industries. For details regarding our acquisitions, see Note 5 to the consolidated financial statements.
54
In November 2015, we announced that we entered into a definitive agreement to acquire the outreach laboratory service business of Clinical Laboratory Partners ("CLP"), a wholly-owned subsidiary of Hartford HealthCare. CLP provides clinical lab testing to physicians in Connecticut. The acquisition is expected to be completed in the first quarter of 2016, subject to customary regulatory closing conditions.
Contribution of Clinical Trials Business
On July 1, 2015, we closed on our joint venture with Quintiles to form a global clinical trials central laboratory services joint venture, Q2 Solutions. In connection with the transaction, we contributed certain assets of our clinical trials testing business to the newly formed joint venture in exchange for a non-controlling, 40% ownership interest ("Clinical Trials Contribution"). As a result of the transaction, we recognized a non-cash pre-tax gain of $334 million. The Clinical Trials Contribution is consistent with our five-point strategy.
Consolidated net revenues and operating costs and expenses include the operating results of our clinical trials testing business prior to closing. Subsequent to closing, our ownership interest in the operating results of the joint venture is being accounted for under the equity method of accounting and recorded within a single line item, equity in earnings of equity method investees, net of taxes, on the consolidated statements of operations.
Assets Held for Sale
During the third quarter of 2015, certain non-core assets of our DS businesses were reclassified to assets held for sale.
For further details regarding our dispositions and assets held for sale, see Note 6 to the consolidated financial statements.
Senior Notes Offering
In March 2015, we completed a $1.2 billion senior notes offering (the "2015 Senior Notes") that was sold in three tranches: (a) $300 million aggregate principal amount of 2.50% senior notes due March 2020, issued at a discount of $1 million; (b) $600 million aggregate principal amount of 3.50% senior notes due March 2025; and (c) $300 million aggregate principal amount of 4.70% senior notes due March 2045. A portion of the proceeds from the 2015 Senior Notes were used to fund the cash tender offer (the "Tender Offer") in March 2015 in which we purchased $176 million of our 6.95% Senior Notes due July 2037 and $74 million of our 5.75% Senior Notes due January 2040. The remaining proceeds from the 2015 Senior Notes, together with borrowing under our existing credit facilities, were used in April 2015 to redeem all of our $500 million 5.45% Senior Notes due November 2015, $150 million, or 50%, of our 3.2% Senior Notes due April 2016 and all of our $375 million 6.4% Senior Notes due July 2017 (the "Redemption"). In connection with the Tender Offer and Redemption, we recorded a $79 million pre-tax loss on early retirement of debt in the first quarter of 2015 and a $65 million pre-tax loss on early retirement of debt in the second quarter of 2015, respectively.
For further details regarding our 2015 Senior Notes, Tender Offer and Redemption, see Note 13 to the consolidated financial statements.
Critical Accounting Policies
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires us to make estimates and assumptions and select accounting policies that affect our reported financial results and the disclosure of contingent assets and liabilities.
While many operational aspects of our business are subject to complex federal, state and local regulations, the accounting for most of our business is generally straightforward, with net revenues primarily recognized upon completion of the testing process. Our revenues are primarily comprised of a high volume of relatively low-dollar transactions, and about one-half of our total costs and expenses consist of employee compensation and benefits. Due to the nature of our business, several of our accounting policies involve significant estimates and judgments:
• | revenues and accounts receivable associated with DIS; |
• | reserves for general and professional liability claims; |
• | reserves for other legal proceedings; |
• | accounting for and recoverability of goodwill; and |
• | accounting for stock-based compensation expense. |
55
Revenues and accounts receivable associated with DIS
The process for estimating the ultimate collection of receivables associated with our DIS business involves significant assumptions and judgments. We primarily recognize revenue for services rendered upon completion of the testing process. Billings for services reimbursed by third-party payers, including Medicare and Medicaid, are generally recorded as revenues net of allowances for differences between amounts billed and the estimated receipts from such payers. Adjustments to the allowances, based on actual receipts from the third-party payers, are recorded upon settlement as an adjustment to net revenues.
We have a standardized approach to estimate and review the collectibility of our receivables based on a number of factors, including the period they have been outstanding, which results in increased allowances for doubtful accounts requirements as the aging of the related receivables increases. Historical collection and payer reimbursement experience is an integral part of the estimation process related to revenues and allowances for doubtful accounts. Changes to the allowances for doubtful accounts estimates are recorded as an adjustment to bad debt expense within selling, general and administrative expenses. Less than 5% of our net accounts receivable as of December 31, 2015 were outstanding more than 150 days.
We believe that the majority of our bad debt expense is primarily the result of the failure of patients to pay the portion of the receivable that is their responsibility; the remainder is primarily the result of missing or incorrect billing information on requisitions. In addition, we regularly assess the state of our billing operations in order to identify issues which may impact the collectibility of receivables or allowance estimates. We believe that the collectibility of our receivables is directly linked to the quality of our billing processes, most notably those related to obtaining the correct information in order to bill effectively for the services we provide. As such, we continue to implement “best practices” and increase the use of electronic ordering to reduce the number of requisitions that we receive from healthcare providers with missing or incorrect billing information. We believe that our collection and allowance estimation processes, along with our close monitoring of our billing operations, help to reduce the risk associated with material adjustments to reserve estimates.
The following table shows current estimates of the percentage of our total volume of requisitions and net revenues associated with our DIS business during 2015 applicable to each payer group:
% of | % of | ||
DIS | DIS | ||
Volume | Revenues | ||
Healthcare Insurers (including coinsurance and deductible responsibilities) | 44 - 48 | 48 - 52 | |
Government Payers | 14 - 18 | 17 - 21 | |
Client Payers | 34 - 38 | 26 - 30 | |
Patients | 1 - 3 | 1 - 3 | |
The following table shows net accounts receivable associated with our DIS business as of December 31, 2015 applicable to each payer group:
% of | |
Consolidated | |
Net Accounts | |
Receivable | |
Healthcare Insurers | 16 |
Government Payers | 16 |
Client Payers | 44 |
Patients (including coinsurance and deductible responsibilities) | 17 |
Total DIS | 93 |
Healthcare insurers
Reimbursements from healthcare insurers are based on negotiated fee-for-service schedules and on capitated payment rates.
56
Substantially all of the accounts receivable due from healthcare insurers represent amounts billed under negotiated fee-for-service arrangements. Collection of such receivables is normally a function of providing complete and correct billing information to the healthcare insurers within the various filing deadlines and typically occurs within 30 to 60 days of billing. Provided we have billed healthcare insurers accurately with complete information prior to the established filing deadline, there has historically been little to no collection risk. If there has been a delay in billing, we determine if the amounts in question will likely go past the filing deadline, and if so, we will reserve accordingly for the billing.
Approximately 4% of our DIS net revenues for the year ended December 31, 2015 are reimbursed under capitated payment arrangements, in which case the healthcare insurers typically reimburse us in the same month services are performed, essentially giving rise to no outstanding accounts receivable at month-end. If any capitated payments are not received on a timely basis, we determine the cause and make a separate determination as to whether or not the collection of the amount from the healthcare insurer is at risk and, if so, would reserve accordingly.
Government payers
Payments for diagnostic information services made by the government are based on fee schedules set by governmental authorities. Collection of such receivables is normally a function of providing the complete and correct billing information within the various filing deadlines. Collection typically occurs within 30 days of billing. Our processes for billing, collecting and estimating uncollectible amounts for receivables due from government payers, as well as the risk of non-collection, are similar to those for healthcare insurers under negotiated fee-for-service arrangements.
Client payers
Client payers include physicians, hospitals, ACOs, IDNs, employers, other commercial laboratories and institutions for which services are performed on a wholesale basis, and are billed based on a negotiated fee schedule. Credit risk and ability to pay are more of a consideration for these payers than healthcare insurers and government payers. Collection typically occurs within 60 to 90 days of billing. In addition to our standard approach to establishing allowances for doubtful accounts, our approach to client payer receivables also considers specific account reviews, historical collection experience and other factors.
Patients
Patients are billed based on established patient fee schedules, subject to any limitations on fees negotiated with healthcare insurers or physicians on behalf of their patients. Collection of receivables due from patients is subject to credit risk and ability of the patients to pay. In addition to our standard approach to establishing allowances for doubtful accounts, our approach to patient receivables also considers historical collection experience and other factors. Patient receivables are generally fully reserved for when the related billing reaches 210 days outstanding. Balances are automatically written off when they are sent to collection agencies. Reserves are adjusted for estimated recoveries of amounts sent to collection agencies based on historical collection experience, which is regularly monitored.
Reserves for general and professional liability claims
As a general matter, providers of diagnostic information services may be subject to lawsuits alleging negligence or other similar legal claims. These suits could involve claims for substantial damages. Any professional liability litigation could also have an adverse impact on our client base and reputation. We maintain various liability insurance coverages for claims that could result from providing, or failing to provide, diagnostic information services, including inaccurate testing results, and other exposures. Our insurance coverage limits our maximum exposure on individual claims; however, we are essentially self-insured for a significant portion of these claims. While the basis for claims reserves is actuarially determined losses based upon our historical and projected loss experience, the process of analyzing, assessing and establishing reserve estimates relative to these types of claims involves a high degree of judgment. Although we believe that our present reserves and insurance coverage are sufficient to cover currently estimated exposures, it is possible that we may incur liabilities in excess of our recorded reserves or insurance coverage. Changes in the facts and circumstances associated with claims could have a material impact on our results of operations (principally costs of services), cash flows and financial condition in the period that reserve estimates are adjusted or paid.
57
Reserves for other legal proceedings
Our businesses are subject to or impacted by extensive and frequently changing laws and regulations, including inspections and audits by governmental agencies, in the United States (at both the federal and state levels) and the other jurisdictions in which we conduct business. Although we believe that we are in compliance, in all material respects, with applicable laws and regulations, there can be no assurance that a regulatory agency would not reach a different conclusion. Any noncompliance by us with applicable laws and regulations could have a material adverse effect on our results of operations. In addition, these laws and regulations may be interpreted or applied by a prosecutorial, regulatory or judicial authority in a manner that could require us to make changes in our operations, including our pricing and/or billing practices. We have, in the past, entered into several settlement agreements with various government and private payers relating to industry-wide billing and marketing practices that had been substantially discontinued. The federal or state governments may bring claims based on our current practices, which we believe are lawful. In addition, certain federal and state statutes, including the qui tam provisions of federal and state false claims acts, allow private individuals to bring lawsuits against healthcare companies on behalf of government or private payers alleging inappropriate billing practices. We are aware of certain pending lawsuits including class action lawsuits, and have received several subpoenas related to billing practices. See Note 17 to the consolidated financial statements for a discussion of the various legal proceedings that involve the Company.
The process of analyzing, assessing and establishing reserve estimates relative to legal proceedings involves a high degree of judgment. Management has established reserves for legal proceedings in accordance with generally accepted accounting principles. Changes in facts and circumstances related to such proceedings could lead to significant adjustments to reserve estimates for such matters and could have a material impact on our results of operations, cash flows and financial condition in the period that reserve estimates are adjusted or paid.
Accounting for and recoverability of goodwill
We do not amortize goodwill, but evaluate the recoverability and measure the potential impairment of our goodwill annually, or more frequently, in the case of other events that indicate a potential impairment. We have identified the following reporting units for goodwill impairment testing:
• | DIS business; |
• | Diagnostic products business; |
• | Risk assessment services business; and |
• | Clinical trials testing business (disposed of on July 1, 2015 as a result of the Clinical Trials Contribution). |
Certain reporting units have components that have been aggregated into a single reporting unit because they have similar economic characteristics, including similarities in financial performance, nature of products or services, nature of production processes and types of customers.
The annual impairment test includes an option to perform a qualitative assessment of whether it is more-likely-than-not that a reporting unit's fair value is less than its carrying value prior to, or as an alternative to, performing the two-step quantitative goodwill impairment test. In evaluating whether it is more likely than not that the fair value of a reporting unit is less than its carrying value, we assess relevant events and circumstances such as: (a) macroeconomic conditions; (b) industry and market considerations; (c) cost factors; (d) overall financial performance; (e) other relevant entity-specific events; (f) events affecting a reporting unit; (g) a sustained decrease in share price; and (h) other relevant events and circumstances that affect the fair value or carrying value of a reporting unit. If, after assessing the totality of events or circumstances, we determine that it is more-likely-than-not that the fair value of a reporting unit is less than its carrying value, then we are required to perform the first step of the two-step goodwill impairment test. Otherwise, no further analysis is required.
The quantitative impairment test is a two-step process that begins with the estimation of the fair value of the reporting unit. The first step in the two-step process screens for potential impairment and the second step measures the amount of the impairment, if any. As part of the first step to assess potential impairment, we compare our estimate of fair value for the reporting unit to the carrying value of the reporting unit. If the carrying value is greater than our estimate of fair value, we would then proceed to the second step to measure the impairment, if any. The second step compares the implied fair value of goodwill with its carrying value. The implied fair value is determined by allocating the fair value of the reporting unit to all of the assets and liabilities of that unit as if the reporting unit had been acquired in a business combination and the fair value of the reporting unit was the purchase price paid to acquire the reporting unit. The excess of the fair value of the reporting unit over the amounts assigned to its assets and liabilities is the implied fair value of goodwill. If the carrying value of the reporting unit's goodwill is greater than its implied fair value, an impairment loss will be recognized in the amount of the excess.
58
We calculate the fair value of each reporting unit using either a discounted cash flows analysis that converts future cash flow amounts into a single discounted present value amount or a market approach. We assess the valuation methodology based upon the relevance and availability of the data at the time we perform the valuation. The discounted cash flows analysis includes several unobservable inputs related to our own assumptions. The assumptions and estimates used in the discounted cash flows model are based upon the best available information in the circumstances and include a forecast of expected future cash flows, long-term growth rates, discount rates that are commensurate with economic risks, assumed income tax rates and estimates of capital expenditures and working capital. The fair values of the reporting units could be different if, for example, forecasted revenue growth rates, economic conditions, government regulations or actions by payers to control utilization of or reimbursement for health care services, turn out to be different than our assumptions or estimates. Changes in the assumed discount rates due to changes in interest rates could also affect the estimated fair values of the reporting units. We use a discount rate that considers a weighted average cost of capital plus an appropriate risk premium based upon the reporting unit being valued. Our analysis also considers publicly available information regarding the market capitalization of our Company, as well as (i) the financial projections and future prospects of our business, including its growth opportunities and likely operational improvements, and (ii) comparable sales prices, if available. We believe our estimation methods are reasonable and reflect common valuation practices.
On a quarterly basis, we perform a review of our business to determine if events or changes in circumstances have occurred which could have a material adverse effect on the fair value of the Company and its goodwill. If such events or changes in circumstances were deemed to have occurred, we would perform an impairment test of goodwill as of the end of the quarter, consistent with the annual impairment test performed during the fourth quarter of our fiscal year ended December 31st, and record any noted impairment loss.
For year ended December 31, 2015, we performed the qualitative assessment for our DIS and risk assessment services reporting units. Based on the totality of information available for our DIS and risk assessment services reporting units, we concluded that it was more likely than not that the estimated fair values were greater than the carrying values of the reporting units, and as such, no further analysis was required. For our diagnostic products reporting unit, we performed step one of the goodwill impairment test and concluded that goodwill of the reporting unit was not impaired. Our most recent assessment indicates that no reporting units are currently at risk of impairment. For the year ended December 31, 2014, we elected to bypass the qualitative assessment, performed step one of the goodwill impairment test for all of our reporting units and concluded that goodwill of each reporting unit was not impaired.
Accounting for stock-based compensation expense
We record stock-based compensation as a charge to earnings, net of the estimated impact of forfeited awards. As such, we recognize stock-based compensation cost only for those stock-based awards that are estimated to ultimately vest over their requisite service period, based on the vesting provisions of the individual grants. The process of estimating the fair value of stock-based compensation awards and recognizing stock-based compensation cost over their requisite service periods involves significant assumptions and judgments.
Beginning in 2015, we changed the method for estimating the fair value of our stock option awards from a lattice-based option-valuation method to a Black-Scholes option-valuation model, which has been applied prospectively to stock option awards. The change did not have a significant effect on our stock-based compensation expense reported in our consolidated statements of operations because we grant stock option awards based on a prescribed dollar value.
Estimating the fair value of stock option awards on the date of grant using the Black-Scholes option-valuation model and lattice-based option-valuation model requires management to make certain assumptions regarding: (i) the expected volatility in the market price of our common stock; (ii) dividend yield; (iii) risk-free interest rates; and (iv) the period of time employees are expected to hold the award prior to exercise (referred to as the expected holding period). Under the Black-Scholes option-valuation model, the expected volatility is based on historical volatilities of our common stock. The dividend yield is based on the approved annual dividend rate in effect and current market price of the underlying common stock at the time of grant. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for bonds with maturities consistent with the expected holding period of the related award. The expected holding period of the awards granted is estimated using the historical exercise behavior of employees.
We estimate the expected impact of forfeited awards and recognize stock-based compensation cost only for those awards expected to vest. We use historical experience to estimate projected forfeitures. If actual forfeiture rates are materially different from our estimates, stock-based compensation expense could be significantly different from what we have recorded in the current period. We periodically review actual forfeiture experience and adjust our estimates as necessary. The cumulative
59
effect on current and prior periods of a change in the estimated forfeiture rate is recognized as compensation cost in earnings in the period of the change.
The terms of our performance share unit awards allow the recipients to earn a variable number of shares based on the achievement of the performance goals specified in the awards. Stock-based compensation expense associated with performance share units is recognized based on management's best estimates of the achievement of the performance goals specified in such awards and the resulting number of shares that will be earned. If the actual number of performance share units earned is different from our estimates, stock-based compensation could be significantly different from what we have recorded in the current period. The cumulative effect on current and prior periods of a change in the estimated number of performance share units expected to be earned is recognized as compensation cost in earnings in the period of the change. While the assumptions used to calculate and account for stock-based compensation awards represent management's best estimates, these estimates involve inherent uncertainties and the application of management's judgment. As a result, if changes are made to our assumptions and estimates, our stock-based compensation expense could vary significantly from period to period. In addition, the number of awards made under our equity compensation plans, changes in the design of those plans, the price of our shares and the performance of our Company can all cause stock-based compensation expense to vary from period to period.
Results of Operations
Basis of Presentation
Our DIS business currently represents our one reportable business segment. The DIS business for each of the three years ended December 31, 2015 accounted for more than 90% of our consolidated net revenues from continuing operations. Our other operating segments consist of our DS businesses. For further details regarding our business segment information, see Note 19 to the consolidated financial statements.
The results of operations for NID and HemoCue have been classified as discontinued operations in the accompanying consolidated statements of operations and related disclosures for all periods presented. In December 2012, we committed to a plan to sell HemoCue and completed the sale of HemoCue in April 2013. We began reporting NID, a test kit manufacturing subsidiary which was wound down in 2006, as a discontinued operation in 2006 and will continue to report NID as a discontinued operation until uncertain tax benefits associated with NID are resolved. For further details regarding our discontinued operations, see Note 18 to the consolidated financial statements.
60
Results of Operations
The following table sets forth certain results of operations data for the periods presented:
2015 | 2014 | 2013 | 2015 vs. 2014 Increase (Decrease) | 2014 vs. 2013 Increase (Decrease) | 2015 vs. 2014 % Increase (Decrease) | 2014 vs. 2013 % Increase (Decrease) | |||||||||||||||||||
(dollars in millions, except per share amounts) | |||||||||||||||||||||||||
Net revenues: | |||||||||||||||||||||||||
DIS business | $ | 6,965 | $ | 6,873 | $ | 6,587 | $ | 92 | $ | 286 | 1.3 | % | 4.3 | % | |||||||||||
DS businesses | 528 | 562 | 559 | (34 | ) | 3 | (6.1 | ) | 0.5 | ||||||||||||||||
Total net revenues | $ | 7,493 | $ | 7,435 | $ | 7,146 | $ | 58 | $ | 289 | 0.8 | % | 4.0 | % | |||||||||||
Operating costs, expenses and other income: | |||||||||||||||||||||||||
Cost of services | $ | 4,657 | $ | 4,637 | $ | 4,326 | $ | 20 | $ | 311 | 0.4 | % | 7.2 | % | |||||||||||
Selling, general and administrative | 1,679 | 1,728 | 1,704 | (49 | ) | 24 | (2.8 | ) | 1.4 | ||||||||||||||||
Amortization of intangible assets | 81 | 94 | 79 | (13 | ) | 15 | (14.1 | ) | 18.5 | ||||||||||||||||
Gain on contribution of business to joint venture | (334 | ) | — | — | (334 | ) | — | NM | — | ||||||||||||||||
Gain on sale of royalty rights | — | — | (474 | ) | — | 474 | — | NM | |||||||||||||||||
Other operating expense (income), net | 11 | (7 | ) | 36 | 18 | (43 | ) | NM | NM | ||||||||||||||||
Total operating costs, expenses and other income, net | $ | 6,094 | $ | 6,452 | $ | 5,671 | $ | (358 | ) | $ | 781 | (5.5 | )% | 13.8 | % | ||||||||||
Operating income | $ | 1,399 | $ | 983 | $ | 1,475 | $ | 416 | $ | (492 | ) | 42.3 | % | (33.3 | )% | ||||||||||
Other income (expense): | |||||||||||||||||||||||||
Interest expense, net | $ | (153 | ) | $ | (164 | ) | $ | (159 | ) | $ | (11 | ) | $ | 5 | (6.7 | )% | 2.8 | % | |||||||
Other (expense) income, net | (143 | ) | 4 | 8 | 147 | (4 | ) | NM | NM | ||||||||||||||||
Total non-operating expenses, net | $ | (296 | ) | $ | (160 | ) | $ | (151 | ) | $ | 136 | $ | 9 | 85.8 | % | 5.5 | % | ||||||||
Income tax expense | $ | (373 | ) | $ | (262 | ) | $ | (500 | ) | $ | 111 | $ | (238 | ) | 41.9 | % | (47.5 | )% | |||||||
Effective income tax rate | 33.8 | % | 31.8 | % | 37.8 | % | 2.0 | % | (6.0 | )% | NM | NM | |||||||||||||
Equity in earnings of equity method investees, net of taxes | $ | 23 | $ | 26 | $ | 24 | $ | (3 | ) | $ | 2 | (10.8 | )% | 7.7 | % | ||||||||||
Income from discontinued operations, net of taxes | $ | — | $ | 5 | $ | 35 | $ | (5 | ) | $ | (30 | ) | NM | NM | |||||||||||
Income from continuing operations attributable to Quest Diagnostics' stockholders | $ | 709 | $ | 551 | $ | 814 | $ | 158 | $ | (263 | ) | 28.7 | % | (32.3 | )% | ||||||||||
Diluted earnings per common share from continuing operations attributable to Quest Diagnostics’ common stockholders | $ | 4.87 | $ | 3.78 | $ | 5.31 | $ | 1.09 | $ | (1.53 | ) | 28.8 | % | (28.8 | )% | ||||||||||
NM - Not Meaningful
61
The following table sets forth certain results of continuing operations data as a percentage of net revenues for the periods presented:
2015 | 2014 | 2013 | ||||||
Net revenues: | ||||||||
DIS business | 93.0 | % | 92.4 | % | 92.2 | % | ||
DS businesses | 7.0 | % | 7.6 | % | 7.8 | % | ||
Total net revenues | 100.0 | % | 100.0 | % | 100.0 | % | ||
Operating costs, expenses and other income: | ||||||||
Cost of services | 62.1 | % | 62.4 | % | 60.5 | % | ||
Selling, general and administrative | 22.4 | 23.2 | 23.8 | |||||
Amortization of intangible assets | 1.1 | 1.3 | 1.1 | |||||
Gain on contribution of business to joint venture | (4.4 | ) | — | — | ||||
Gain on sale of royalty rights | — | — | (6.6 | ) | ||||
Other operating expense (income), net | 0.1 | (0.1 | ) | 0.6 | ||||
Total operating costs, expenses and other income, net | 81.3 | % | 86.8 | % | 79.4 | % | ||
Operating income | 18.7 | % | 13.2 | % | 20.6 | % | ||
Bad debt | 4.0 | % | 4.0 | % | 3.8 | % | ||
Continuing Operations
Results for the year ended December 31, 2015 were affected by certain items that benefited earnings per diluted share by a net $0.48 as follows:
• | a pre-tax gain of $334 million, or $1.30 per diluted share, related to the Clinical Trials Contribution; |
• | pre-tax charges of $150 million, or $0.62 per diluted share, related to the loss on early retirement of debt and related refinancing charges in connection with the Tender Offer and Redemption ($6 million in interest expense, net and $144 million in other (expense) income, net); |
• | pre-tax charges of $105 million, or $0.44 per diluted share, related to restructuring costs primarily associated with workforce reductions, integration costs associated with acquisitions and professional fees associated with the further restructuring and integrating our business ($63 million in cost of services and $42 million in selling, general and administrative expenses); |
• | a deferred income tax benefit of $58 million, or $0.40 per diluted share, associated with winding down a subsidiary; |
• | net pre-tax costs of $33 million, or $0.14 per diluted share, primarily associated with non-cash asset impairment charges and other costs associated with Celera Products and winding down a subsidiary as well as costs incurred related to legal matters, partially offset by a pre-tax gain of $13 million associated with a decrease in the fair value of the contingent consideration accrual associated with our Summit Health acquisition ($2 million of costs in cost of services, $21 million of costs in selling, general and administrative expenses and $10 million of net costs in other operating expense (income), net); and |
• | pre-tax charges of $5 million, or $0.02 per diluted share, in equity in earnings of equity method investees, net of taxes consisting of restructuring and integration charges incurred by Q2 Solutions. |
Results for the year ended December 31, 2014 were affected by certain items that reduced earnings per diluted share by a net $0.32 as follows:
• | pre-tax charges of $121 million, or $0.53 per diluted share, related to restructuring costs primarily associated with workforce reductions, integration costs associated with acquisitions and professional fees associated with the further restructuring and integrating our business ($50 million in cost of services, $69 million in selling, general and administrative expenses and $2 million in other operating expense (income), net); |
• | a discrete tax benefit of $44 million, or $0.30 per diluted share, associated with the favorable resolution of certain tax contingencies; and |
• | net pre-tax costs of $15 million, or $0.09 per diluted share, primarily associated with costs related to legal matters, partially offset by a pre-tax gain of $9 million associated with a decrease in the fair value of the contingent |
62
consideration accrual associated with our Summit Health acquisition ($24 million of costs in selling, general and administrative expenses and a pre-tax gain of $9 million in other operating expense (income), net).
Results for the year ended December 31, 2013 were affected by certain items that benefited earnings per diluted share by a net $1.31 as follows:
• | a pre-tax gain of $474 million, or $1.95 per diluted share, associated with the sale of future royalty rights of ibrutinib ("Ibrutinib Sale"); |
• | pre-tax charges of $115 million, or $0.47 per diluted share, related to restructuring costs primarily associated with workforce reductions, integration costs and professional fees associated with further restructuring and integrating our business ($43 million in cost of services and $72 million in selling, general and administrative expenses); and |
• | a pre-tax loss of $40 million, or $0.17 per diluted share, in other operating expense (income), net associated with the sale of Enterix. |
Net Revenues
Net revenues for the year ended December 31, 2015 were 0.8% above the prior year level. The Clinical Trials Contribution negatively impacted net revenues by 1.2%.
DIS revenues increased by 1.3% for the year ended December 31, 2015 compared to the prior year. Acquisitions contributed nearly 1.3% to DIS revenue growth. DIS volume, measured by the number of requisitions, increased 1.2% for the year ended December 31, 2015 compared to the prior year. Acquisitions contributed approximately 1.5% to DIS volume in the period. Revenue per requisition for the year ended December 31, 2015 increased 0.1% compared to the prior year, reflecting growth in gene-based and esoteric testing as well as strong growth in wellness and prescription drug monitoring, substantially offset by moderate reimbursement pressure.
For the year ended December 31, 2015, combined revenues in our DS businesses decreased by 6.1% compared to the prior year. The Clinical Trials Contribution negatively impacted DS revenues by approximately 17%.
Net revenues for the year ended December 31, 2014 were 4.0% above the prior year level.
DIS revenues increased by 4.3% for the year ended December 31, 2014 compared to the prior year. Acquisitions contributed approximately 6% to DIS revenue growth. In addition, six new professional lab services agreements executed in late 2013 and 2014 contributed to DIS revenue growth. These benefits were partially offset by our decision to not renew certain business due to strategic reasons during the year. DIS volume, measured by the number of requisitions, increased 6.3% for the year end December 31, 2014 compared to the prior year. Acquisitions contributed approximately 7% to the DIS volume for the year ended December 31, 2014. Organic volume decreased approximately 1% primarily due to the harsh winter and our decision to not renew certain business due to strategic reasons during the year. Revenue per requisition for the year ended December 31, 2014 decreased 1.8% compared to the prior year. Acquisitions reduced revenue per requisition by approximately 1% during the year as a result of test mix associated with those acquisitions.
For the year ended December 31, 2014, combined revenues in our DS businesses increased by 0.5% compared to the prior year.
Cost of Services
Cost of services consists principally of costs for obtaining, transporting and testing specimens as well as facility costs used for the delivery of our services.
Cost of services increased $20 million for the year ended December 31, 2015 compared to the prior year. This increase was primarily driven by the additional operating costs associated with our acquisitions and, to a lesser extent, higher compensation and benefits and higher restructuring and integration charges, which were partially offset by the impact of our disposition (the Clinical Trials Contribution) on operating costs and net cost reductions under the Invigorate program.
63
Cost of services increased $311 million for the year ended December 31, 2014, as compared to the year ended December 31, 2013. This increase was primarily driven by additional operating costs associated with our acquisitions and higher performance-based compensation costs. These increases were partially offset by cost reductions under the Invigorate program and lower restructuring and integration costs compared to the prior year. In addition, cost of services includes a $56 million increase in the current year, that was principally due to the allocation of certain facility costs between cost of services and selling, general and administrative expenses for those facilities that support both service delivery and administrative functions, in order to reflect our current operations.
Selling, General and Administrative Expenses ("SG&A")
SG&A consist principally of the costs associated with our sales and marketing efforts, billing operations, bad debt expense and general management and administrative support as well as administrative facility costs.
SG&A decreased $49 million for the year ended December 31, 2015 compared to the prior year. The decrease in SG&A was primarily driven by lower compensation and benefits as a result of cost reductions under the Invigorate program, lower restructuring and integration charges and the impact of our disposition (the Clinical Trials Contribution) on operating costs, partially offset by additional operating costs associated with our acquisitions.
SG&A increased $24 million for the year ended December 31, 2014 compared to the prior year. This increase was due to additional operating costs associated with our acquisitions, higher performance-based compensation costs, costs related to legal matters and higher bad debt expense. This increase was partially offset by a $56 million reduction to SG&A, that was principally due to the allocation of certain facility costs between cost of services and SG&A for those facilities that support both service delivery and administrative functions, in order to reflect our current operations. In addition, this increase was also partially offset by lower overall compensation and benefit costs, resulting from reduced headcount under the Invigorate program, and lower restructuring and integration related costs compared to the prior year.
Amortization of Intangible Assets
The $13 million decrease in amortization of intangible assets for the year ended December 31, 2015 compared to the prior year was primarily a result of intangible assets that became fully amortized in 2014 and early 2015.
The $15 million increase in amortization of intangible assets for the year ended December 31, 2014 compared to the prior year primarily reflects the impact of amortization of intangible assets acquired as part of our Solstas Lab Partners Group ("Solstas"), Summit Health, Inc. ("Summit Health") and Steward Health Care Systems, LLC outreach laboratory service ("Steward") acquisitions.
Gain on Contribution of Business to Joint Venture
Gain on contribution of business to joint venture for the year ended December 31, 2015 is the non-cash gain resulting from the Clinical Trials Contribution.
Gain on Sale of Royalty Rights
For the year ended December 31, 2013, gain on sale of royalty rights includes the gain associated with the Ibrutinib Sale.
Other Operating Expense (Income), net
Other operating expense (income), net includes miscellaneous income and expense items and other charges related to operating activities.
For the year ended December 31, 2015, other operating expense (income), net includes non-cash asset impairment charges primarily associated with our Celera Products business and another subsidiary, partially offset by a gain of $13 million associated with a decrease in the fair value of the contingent consideration accrual associated with our Summit Health acquisition. For the year ended December 31, 2014, other operating expense (income), net includes a gain of $9 million associated with a decrease in the fair value of the contingent consideration accrual associated with our Summit Health acquisition. For the year ended December 31, 2013 other operating expense (income), net includes the loss on sale of Enterix of $40 million.
64
Interest Expense, net
Interest expense, net for the year ended December 31, 2015 decreased by $11 million compared to the prior year. The decrease in interest expense, net was primarily a result of lower outstanding debt balances compared to the prior year and lower interest rates associated with our 2015 Senior Notes.
Interest expense, net for the year ended December 31, 2014 increased by $5 million compared to the prior year, primarily as a result of higher outstanding debt balances compared to the prior year.
Other (Expense) Income, net
For the years ended December 31, 2015, 2014 and 2013, other (expense) income, net includes (losses) gains of $(1) million, $4 million and $10 million, respectively, associated with investments held in trusts pursuant to our supplemental deferred compensation plans. In addition, other (expense) income, net for the year ended December 31, 2015, includes the loss on early retirement of debt of $144 million associated with the Tender Offer and Redemption.
Income Tax Expense
The $111 million increase in income tax expense for the year ended December 31, 2015, compared to the prior year, was primarily a result of deferred tax expense of $145 million associated with the gain on the Clinical Trials Contribution, partially offset by the recognition of a $58 million deferred tax benefit associated with winding down a subsidiary as well as a $57 million benefit associated with the loss on early retirement of debt in connection with the Tender Offer and Redemption. The increase was further driven by a $44 million tax benefit recognized in 2014 related to the favorable resolution of a tax contingency.
Our effective income tax rate for the year ended December 31, 2015, was negatively impacted by the higher tax rate associated with the gain on the Clinical Trials Contribution.
The $238 million decrease in income tax expense for the year ended December 31, 2014 compared to the prior year was primarily due to expense associated with the Ibrutinib Sale in 2013. Additionally, income tax expense and our effective income tax rate for the year ended December 31, 2014, were positively impacted by a $44 million benefit related to the favorable resolution of a tax contingency.
Discontinued Operations
Discontinued operations includes HemoCue, which was sold in April 2013, and NID, a test kit manufacturing subsidiary discontinued in 2006. The results of operations for HemoCue and NID have been classified as discontinued operations for all periods presented. For further details regarding our discontinued operations, see Note 18 to the consolidated financial statements.
The following table summarizes our income from discontinued operations, net of taxes:
2015 vs. 2014 Increase (Decrease) | 2014 vs. 2013 Increase (Decrease) | ||||||||||||||||||
2015 | 2014 | 2013 | |||||||||||||||||
(dollars in millions) | |||||||||||||||||||
Net revenues | $ | — | $ | — | $ | 28 | $ | — | $ | (28 | ) | ||||||||
Income from discontinued operations before taxes | — | 1 | 25 | (1 | ) | (24 | ) | ||||||||||||
Income tax benefit | — | (4 | ) | (10 | ) | (4 | ) | (6 | ) | ||||||||||
Income from discontinued operations, net of taxes | $ | — | $ | 5 | $ | 35 | $ | (5 | ) | $ | (30 | ) | |||||||
Income from discontinued operations, net of taxes for the year ended December 31, 2013 includes a gain of $14 million (including foreign currency translation adjustments, partially offset by income tax expense and transaction costs) associated with the sale of HemoCue. In addition, income from discontinued operations, net of taxes for the year ended December 31, 2013 includes discrete tax benefits of $20 million associated with favorable resolution of certain tax contingencies related to NID.
65
Quantitative and Qualitative Disclosures About Market Risk
We address our exposure to market risks, principally the market risk of changes in interest rates, through a controlled program of risk management that includes the use of derivative financial instruments. We do not hold or issue derivative financial instruments for speculative purposes. We seek to mitigate the variability in cash outflows that result from changes in interest rates by maintaining a balanced mix of fixed-rate and variable-rate debt obligations. In order to achieve this objective, we have entered into interest rate swaps. Interest rate swaps involve the periodic exchange of payments without the exchange of underlying principal or notional amounts. Net settlements are recognized as an adjustment to interest expense. We believe that our exposures to foreign exchange impacts and changes in commodity prices are not material to our consolidated financial condition or results of operations.
At December 31, 2015 and 2014, the fair value of our debt was estimated at approximately $3.7 billion and $4.2 billion, respectively, using quoted active market prices and yields for the same or similar types of borrowings, taking into account the underlying terms of the debt instruments. At December 31, 2015 and 2014, the estimated fair value exceeded the carrying value of the debt by $82 million and $416 million, respectively. A hypothetical 10% increase in interest rates (representing 39 basis points and 45 basis points at December 31, 2015 and 2014, respectively) would potentially reduce the estimated fair value of our debt by approximately $112 million and $108 million at December 31, 2015 and 2014, respectively.
Borrowings under our secured receivables credit facility and our senior unsecured revolving credit facility are subject to variable interest rates. Interest on our secured receivables credit facility is based on rates that are intended to approximate commercial paper rates for highly rated issuers plus a spread. Interest on our senior unsecured revolving credit facility is subject to a pricing schedule that can fluctuate based on changes in our credit ratings. As such, our borrowing cost under this credit arrangement will be subject to both fluctuations in interest rates and changes in our credit ratings. At December 31, 2015, the borrowing rates under these debt instruments were: for our secured receivables credit facility, 0.96%; and for our senior unsecured revolving credit facility, LIBOR plus 1.125%. At December 31, 2015, the weighted average LIBOR was 0.4%. As of December 31, 2015, there were no borrowings outstanding under our $600 million secured receivables credit facility or under our $750 million senior unsecured revolving credit facility.
The notional amount of fixed-to-variable interest rate swaps at both December 31, 2015 and 2014 was $1.2 billion. The aggregate fair value of the fixed-to-variable interest rate swaps that are in an asset position was $23 million at December 31, 2015. The aggregate fair value of the fixed-to-variable interest rate swaps that are in a liability position was $6 million at December 31, 2015. There were no forward starting interest rate swaps outstanding at December 31, 2015. The notional amount of forward starting interest rate swaps at December 31, 2014 was $150 million.
Based on our net exposure to interest rate changes, a hypothetical 10% change to the variable interest rate component of our variable rate indebtedness (representing 3 basis points) would not impact annual interest expense materially, assuming no changes to the debt outstanding at December 31, 2015. A hypothetical 10% change in the forward one-month LIBOR curve (representing a 18 basis point change in the weighted average yield) would potentially change the fair value of our derivative assets by $8 million. A hypothetical 10% change in the forward one-month LIBOR curve (representing a 15 basis point change in the weighted average yield) would potentially change the fair value of our derivative liabilities by $4 million.
For further details regarding our outstanding debt and our financial instruments and hedging activities, see Notes 13 and 14, respectively, to the consolidated financial statements.
Risk Associated with Investment Portfolio
Our investment portfolio includes equity investments comprised primarily of strategic equity holdings in privately and publicly held companies. These securities are exposed to price fluctuations and are generally concentrated in the life sciences industry. The carrying value of our equity investments (excluding investments accounted for under the equity method) was $14 million at December 31, 2015.
We regularly evaluate the fair value measurements of our equity investments to determine if losses in value are other than temporary and if an impairment loss has been incurred. The evaluation considers whether the security has the ability to recover and, if so, the estimated recovery period. Other factors that are considered in this evaluation include the amount of the other-than-temporary decline and its duration, the issuer’s financial condition and short-term prospects and whether the market decline was caused by overall economic conditions or conditions specific to the individual security.
We do not hedge our equity price risk. The impact of an adverse movement in equity prices on our holdings in privately held companies cannot be easily quantified, as our ability to realize returns on investments depends on, among other
66
things, the enterprises’ ability to raise additional capital or derive cash inflows from continuing operations or through liquidity events such as initial public offerings, mergers or private sales.
Liquidity and Capital Resources
2015 | 2014 | 2013 | |||||||||
(dollars in millions) | |||||||||||
Net cash provided by operating activities | $ | 810 | $ | 938 | $ | 652 | |||||
Net cash (used in) provided by investing activities | (362 | ) | (1,025 | ) | 328 | ||||||
Net cash (used in) provided by financing activities | (507 | ) | 92 | (1,106 | ) | ||||||
Net change in cash and cash equivalents | $ | (59 | ) | $ | 5 | $ | (126 | ) | |||
Cash and Cash Equivalents
Cash and cash equivalents consist of cash and highly liquid short-term investments. Cash and cash equivalents at December 31, 2015, 2014 and 2013 totaled $133 million, $192 million and $187 million, respectively.
Cash Flows from Operating Activities
Net cash provided by operating activities for the year ended December 31, 2015 was $810 million, compared to $938 million for the year ended December 31, 2014. This $128 million decrease was primarily a result of $146 million of pre-tax cash charges associated with the early retirement of debt ($89 million after the related cash tax benefit), payments associated with an additional payroll cycle in 2015, an income tax payment in the third quarter of 2015 associated with certain tax contingencies and higher performance-based compensation payments, which were partially offset by improved operating performance and improved working capital through management of days sales outstanding and days payable outstanding.
Net cash provided by operating activities for the year ended December 31, 2014 was $938 million, compared to $652 million for the year ended December 31, 2013. The $286 million increase was primarily a result of a $175 million income tax payment in 2013 associated with the Ibrutinib Sale and $70 million of income tax payments which were deferred from the fourth quarter of 2012 into the first quarter of 2013 under a program offered to companies whose principal place of business was in states most affected by Hurricane Sandy as well as a third quarter of 2013 income tax payment of $28 million related to the resolution of certain audit matters.
Days sales outstanding, a measure of billing and collection efficiency, was 47 days, 48 days and 47 days at December 31, 2015, 2014 and 2013, respectively.
Cash Flows from Investing Activities
Net cash used in investing activities for the year ended December 31, 2015 was $362 million, compared to $1.0 billion for the year ended December 31, 2014. This $663 million decrease was a result of a $661 million decrease in business acquisitions in 2015 compared to 2014 and a $45 million decrease in capital expenditures, which were partially offset by $33 million of cash included in our Clinical Trials Contribution.
Net cash (used in) provided by investing activities for the year ended December 31, 2014 was $(1.0) billion, compared to $328 million for the year ended December 31, 2013. The $1.4 billion increase in cash used in investing activities was a result of a $515 million increase in business acquisitions, primarily associated with the Solstas acquisition, and a $77 million increase in capital expenditures, primarily a result of investments to support our Invigorate program, our new clinical testing laboratory in Marlborough, Massachusetts and continued investments to integrate our acquisitions. The increase in cash used in investing activities for the year ended December 31, 2014, as compared to the prior year, was also a result of proceeds from the Ibrutinib Sale of $474 million, net of transaction costs, in 2013, and proceeds from the sales of HemoCue and Enterix of $296 million, net of transaction costs, in 2013.
The acquisition of Solstas in 2014 was funded using borrowings under our secured receivables credit facility and our senior unsecured revolving credit facility.
67
Cash Flows from Financing Activities
Net cash (used in) provided by financing activities for the year ended December 31, 2015 was $(507) million, compared to $92 million for the year ended December 31, 2014. This $599 million increase in cash used in financing activities was primarily a result of a $455 million decrease in net borrowings (proceeds from borrowings less repayments of debt), a $92 million increase in repurchases of our common stock (discussed in "Share Repurchases"), a $25 million increase in dividends paid as a result of higher dividend rates in 2015 (discussed in "Dividends") and an $18 million decrease in proceeds from the exercise of stock options. Net cash used in financing activities for the year ended December 31, 2015, also includes $51 million of deferred acquisition consideration payments, primarily to UMass Memorial Medical Center ("UMass") related to the business acquisition in 2013, and $63 million of proceeds from the sale of a noncontrolling interest in a subsidiary to UMass.
Net cash provided by (used in) financing activities for the year ended December 31, 2014 was $92 million, compared to $(1.1) billion for the year ended December 31, 2013. The $1.2 billion increase in cash provided by financing activities was primarily a result of a $375 million net increase in debt, primarily a result of borrowings of $598 million from our Senior Notes Offering in March 2014, partially offset by the $200 million repayment of our floating rate senior notes due March 2014, and a $904 million decrease in repurchases of our common stock (discussed in "Share Repurchases"). These increases in cash provided by financing activities were partially offset by a $60 million decrease in proceeds from the exercise of stock options.
In 2015, we borrowed $1.3 billion under our secured receivables credit facility, all of which was repaid in 2015. In 2014, we borrowed $1.2 billion under our secured receivables credit facility and $200 million under our senior unsecured revolving credit facility, all of which was repaid in 2014.
For details regarding our 2015 Senior Notes Offering, see "Recent Transactions: Senior Notes Offering" and Note 13 to the consolidated financial statements. For details regarding our 2014 Senior Notes Offering, see Note 13 to the consolidated financial statements
Dividends
During each of the quarters of 2015, our Board of Directors declared a quarterly cash dividend of $0.38 per common share. During each of the quarters of 2014, our Board of Directors declared a quarterly cash dividend of $0.33 per common share. During each of the quarters in 2013, our Board of Directors declared a quarterly cash dividend of $0.30 per common share. We expect to fund future dividend payments with cash flows from operations, and do not expect the dividend to have a material impact on our ability to finance future growth.
On January 28, 2016, we announced that our Board of Directors authorized a 5% increase in our quarterly dividend from $0.38 per share to $0.40 per share, or $1.60 annually, commencing with the dividend payable in April 2016.
Share Repurchases
In December 2015, our Board of Directors authorized us to repurchase an additional $500 million of our common stock. In August 2013, our Board of Directors authorized us to repurchase an additional $1 billion of our common stock. At December 31, 2015, $972 million remained available under the share repurchase authorization.
For the year ended December 31, 2015, we repurchased 3.2 million shares of our common stock for $224 million. For the year ended December 31, 2014, we repurchased 2.2 million shares of our common stock for $132 million. For the year ended December 31, 2013, we repurchased 17.4 million shares of our common stock for $1.0 billion, including 13.3 million shares repurchased under accelerated share repurchase agreements for $800 million. For further details regarding our share repurchases, see Note 15 to the consolidated financial statements.
68
Contractual Obligations and Commitments
The following table summarizes certain of our contractual obligations as of December 31, 2015 (dollars in millions):
Payments due by period | ||||||||||||||||||||
Contractual Obligations | Total | Less than 1 year | 1-3 years | 3-5 years | After 5 years | |||||||||||||||
Outstanding debt | $ | 3,625 | $ | 150 | $ | — | $ | 1,100 | $ | 2,375 | ||||||||||
Capital lease obligations | 22 | 9 | 10 | 3 | — | |||||||||||||||
Interest payments on outstanding debt | 1,820 | 137 | 268 | 241 | 1,174 | |||||||||||||||
Operating leases | 677 | 182 | 232 | 111 | 152 | |||||||||||||||
Purchase obligations | 206 | 96 | 84 | 6 | 20 | |||||||||||||||
Merger consideration obligation | 4 | 2 | 2 | — | — | |||||||||||||||
Total contractual obligations | $ | 6,354 | $ | 576 | $ | 596 | $ | 1,461 | $ | 3,721 | ||||||||||
Interest payments on our long-term debt have been calculated after giving effect to our interest rate swap agreements, using the interest rates as of December 31, 2015 applied to the December 31, 2015 balances, which are assumed to remain outstanding through their maturity dates.
A full description of the terms of our indebtedness and related debt service requirements and our future payments under certain of our contractual obligations is contained in Note 13 to the consolidated financial statements. A full discussion and analysis regarding our minimum rental commitments under noncancelable operating leases and noncancelable commitments to purchase product or services at December 31, 2015 is contained in Note 17 to the consolidated financial statements. Merger consideration obligation includes consideration owed, including any contingent consideration at maximum payout, on the Steward acquisition. A full discussion and analysis regarding our acquisitions and the related merger consideration obligations as of December 31, 2015 is contained in Note 5 to the consolidated financial statements. A full discussion regarding the fair value of the contingent consideration associated with our acquisitions is discussed in Note 7 to the consolidated financial statements.
As of December 31, 2015, our total liabilities associated with unrecognized tax benefits were approximately $91 million, which were excluded from the table above. We believe it is reasonably possible that these liabilities may decrease by less than $5 million within the next twelve months, primarily as a result of payments, settlements and/or the conclusion of tax examinations on certain tax positions. For the remainder, we cannot make reasonably reliable estimates of the timing of the future payments of these liabilities. Additionally, it is reasonably possible that within the next 12 months, as result of ongoing negotiations with tax authorities and the expiration of statutes of limitations, our total liabilities associated with unrecognized tax benefits will further decrease and beneficially impact the effective tax rate for continuing operations. However, due to the inherent uncertainty of the negotiations and the resulting outcomes, we are not able to estimate the effective tax rate impact at this time. For further details regarding the contingent tax liability reserves, see Note 8 to the consolidated financial statements.
In connection with the sale of an 18.9% noncontrolling interest in a subsidiary to UMass, we granted UMass the right to require us to purchase all of its interest in the subsidiary at fair value commencing July 1, 2020. In connection with the transaction, we received consideration of $68 million, including $50 million associated with the call option exercise price. As of December 31, 2015, the fair value of the redeemable noncontrolling interest on the consolidated balance sheet was $70 million, which was excluded from the table above. Since the redemption of the noncontrolling interest is outside of our control, we cannot make a reasonably reliable estimate of the timing of the future payment, if any, of the redeemable noncontrolling interest. For further details regarding the redeemable noncontrolling interest, see Note 15 to the consolidated financial statements.
Our credit agreements contain various covenants and conditions, including the maintenance of certain financial ratios, that could impact our ability to, among other things, incur additional indebtedness. As of December 31, 2015, we were in compliance with the various financial covenants included in our credit agreements and we do not expect these covenants to adversely impact our ability to execute our growth strategy or conduct normal business operations.
69
Equity Method Investees
Our equity method investees primarily consist of our clinical trials central laboratory services joint venture and our diagnostic information services joint ventures, which are accounted for under the equity method of accounting. We believe that our transactions with our equity method investees are conducted at arm’s length, reflecting current market conditions and pricing. Our investment in equity method investees equals less than 5% of our consolidated total assets. Our proportionate share of income before income taxes associated with our equity method investees equals less than 5% of our consolidated income before income taxes and equity in earnings of equity method investees. We have no material unconditional obligations or guarantees to, or in support of, our equity method investees and their operations.
Requirements and Capital Resources
We estimate that we will invest approximately $250 million to $300 million during 2016 for capital expenditures, to support and grow our existing operations, principally related to investments in information technology, laboratory equipment and facilities, including specific initiatives associated with our Invigorate and other programs.
We believe our cash on hand, cash from operations and borrowing capacity under our credit facilities are sufficient to fund the repayment of the current portion of our long-term debt and the CLP acquisition (discussed in "Recent Transactions: Acquisitions").
In October 2015, we amended and restated the $525 million secured receivables credit facility, increasing the borrowing capacity to $600 million. The amended and restated secured receivables credit facility matures in October 2017. Under the credit facility, we can issue letters of credit totaling $100 million. Issued letters of credit reduce the available borrowing capacity under the facility. For further details regarding the outstanding letters of credit, see Note 17 to the consolidated financial statements.
As of December 31, 2015, $1.3 billion of borrowing capacity was available under our secured receivables credit facility and senior unsecured revolving credit facility. Should one or several banks no longer participate in either of our credit facilities, we would not expect it to impact our ability to fund operations. We expect that we will be able to replace our existing credit facilities with alternative arrangements prior to their expiration. For further details regarding the credit facilities, see Note 13 to the consolidated financial statements.
At December 31, 2015, approximately 37% of our $133 million of consolidated cash and cash equivalents were held outside of the United States. These funds are considered indefinitely reinvested to be used to expand operations either organically or through acquisitions outside the United States. Further, our current plans do not demonstrate a need to repatriate foreign funds in order to fund operations in the United States. If the foreign cash and cash items are needed for operations in the United States, or we otherwise elect to repatriate the funds, we may be required to accrue and pay United States taxes on a significant portion of these amounts.
We believe that cash and cash equivalents on-hand and cash from operations, together with our borrowing capacity under our credit facilities, will provide sufficient financial flexibility to fund seasonal and other working capital requirements, capital expenditures, debt service requirements and other obligations, cash dividends on common shares, share repurchases and additional growth opportunities for the foreseeable future. We believe that our credit profile should provide us with access to additional financing to refinance upcoming debt maturities and, if necessary, to fund growth opportunities that cannot be funded from existing sources.
Inflation
We believe that inflation generally does not have a material adverse effect on our results of operations or financial condition.
Impact of New Accounting Standards
The impacts of recent accounting pronouncements not yet effective on our consolidated financial statements are discussed in Note 2 to the consolidated financial statements.
70
REPORT OF MANAGEMENT ON INTERNAL CONTROL OVER FINANCIAL REPORTING
The management of the Company, including its Chief Executive Officer and Chief Financial Officer, is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Securities Exchange Act of 1934, as amended. Management assessed the effectiveness of the Company's internal control over financial reporting as of December 31, 2015 based on criteria for effective internal control over financial reporting described in “Internal Control - Integrated Framework (2013)” issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on this assessment, management has determined that the Company's internal control over financial reporting as of December 31, 2015 is effective.
The Company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America. Internal control over financial reporting includes policies and procedures that: (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with accounting principles generally accepted in the United States of America, and that receipts and expenditures of the Company are being made only in accordance with authorization of management and directors of the Company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of assets that could have a material effect on the consolidated financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
PricewaterhouseCoopers LLP, the independent registered public accounting firm that audited the financial statements included in this annual report, audited the Company's internal control over financial reporting as of December 31, 2015 and issued their audit report on the Company's internal control over financial reporting included herein.
71
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders
of Quest Diagnostics Incorporated
In our opinion, the consolidated financial statements listed in the index appearing under Item 15(a)(1) present fairly, in all material respects, the financial position of Quest Diagnostics Incorporated and its subsidiaries at December 31, 2015 and 2014, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2015 in conformity with accounting principles generally accepted in the United States of America. In addition, in our opinion, the financial statement schedule listed in the index appearing under Item 15(a)(2) presents fairly, in all material respects, the information set forth therein when read in conjunction with the related consolidated financial statements. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2015, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The Company's management is responsible for these financial statements and the financial statement schedule, for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Report of Management on Internal Control over Financial Reporting. Our responsibility is to express opinions on these financial statements, on the financial statement schedule, and on the Company's internal control over financial reporting based on our integrated audits. We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement and whether effective internal control over financial reporting was maintained in all material respects. Our audits of the financial statements included examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
As discussed in Note 8 to the consolidated financial statements, the Company changed the manner in which it classifies deferred taxes in 2015 due to the adoption of Accounting Standards Update 2015-17, Balance Sheet Classification of Deferred Taxes.
A company's internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company's internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company's assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ | PricewaterhouseCoopers LLP |
Florham Park, New Jersey | |
February 26, 2016 | |
F- 1
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
DECEMBER 31, 2015 AND 2014
(in millions, except per share data)
2015 | 2014 | ||||||
Assets | |||||||
Current assets: | |||||||
Cash and cash equivalents | $ | 133 | $ | 192 | |||
Accounts receivable, net of allowance for doubtful accounts of $254 and $250 at December 31, 2015 and 2014, respectively | 901 | 932 | |||||
Inventories | 84 | 110 | |||||
Deferred income taxes | — | 169 | |||||
Prepaid expenses and other current assets | 207 | 186 | |||||
Assets held for sale | 176 | 14 | |||||
Total current assets | 1,501 | 1,603 | |||||
Property, plant and equipment, net | 925 | 933 | |||||
Goodwill | 5,905 | 6,032 | |||||
Intangible assets, net | 984 | 1,071 | |||||
Investment in equity method investees | 473 | 46 | |||||
Other assets | 174 | 172 | |||||
Total assets | $ | 9,962 | $ | 9,857 | |||
Liabilities and Stockholders’ Equity | |||||||
Current liabilities: | |||||||
Accounts payable and accrued expenses | $ | 1,014 | $ | 1,191 | |||
Current portion of long-term debt | 159 | 518 | |||||
Total current liabilities | 1,173 | 1,709 | |||||
Long-term debt | 3,492 | 3,224 | |||||
Other liabilities | 514 | 594 | |||||
Commitments and contingencies | |||||||
Redeemable noncontrolling interest | 70 | — | |||||
Stockholders’ equity: | |||||||
Quest Diagnostics stockholders’ equity: | |||||||
Common stock, par value $0.01 per share; 600 shares authorized at both December 31, 2015 and 2014; 216 shares and 215 shares issued at December 31, 2015 and 2014, respectively | 2 | 2 | |||||
Additional paid-in capital | 2,481 | 2,418 | |||||
Retained earnings | 6,199 | 5,723 | |||||
Accumulated other comprehensive loss | (38 | ) | (27 | ) | |||
Treasury stock, at cost; 73 shares and 71 shares at December 31, 2015 and 2014, respectively | (3,960 | ) | (3,815 | ) | |||
Total Quest Diagnostics stockholders’ equity | 4,684 | 4,301 | |||||
Noncontrolling interests | 29 | 29 | |||||
Total stockholders’ equity | 4,713 | 4,330 | |||||
Total liabilities and stockholders’ equity | $ | 9,962 | $ | 9,857 | |||
The accompanying notes are an integral part of these statements.
F- 2
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS
FOR THE YEARS ENDED DECEMBER 31, 2015, 2014 AND 2013
(in millions, except per share data)
2015 | 2014 | 2013 | |||||||||
Net revenues | $ | 7,493 | $ | 7,435 | $ | 7,146 | |||||
Operating costs, expenses and other income: | |||||||||||
Cost of services | 4,657 | 4,637 | 4,326 | ||||||||
Selling, general and administrative | 1,679 | 1,728 | 1,704 | ||||||||
Amortization of intangible assets | 81 | 94 | 79 | ||||||||
Gain on contribution of business to joint venture | (334 | ) | — | — | |||||||
Gain on sale of royalty rights | — | — | (474 | ) | |||||||
Other operating expense (income), net | 11 | (7 | ) | 36 | |||||||
Total operating costs, expenses and other income, net | 6,094 | 6,452 | 5,671 | ||||||||
Operating income | 1,399 | 983 | 1,475 | ||||||||
Other income (expense): | |||||||||||
Interest expense, net | (153 | ) | (164 | ) | (159 | ) | |||||
Other (expense) income, net | (143 | ) | 4 | 8 | |||||||
Total non-operating expenses, net | (296 | ) | (160 | ) | (151 | ) | |||||
Income from continuing operations before income taxes and equity in earnings of equity method investees | 1,103 | 823 | 1,324 | ||||||||
Income tax expense | (373 | ) | (262 | ) | (500 | ) | |||||
Equity in earnings of equity method investees, net of taxes | 23 | 26 | 24 | ||||||||
Income from continuing operations | 753 | 587 | 848 | ||||||||
Income from discontinued operations, net of taxes | — | 5 | 35 | ||||||||
Net income | 753 | 592 | 883 | ||||||||
Less: Net income attributable to noncontrolling interests | 44 | 36 | 34 | ||||||||
Net income attributable to Quest Diagnostics | $ | 709 | $ | 556 | $ | 849 | |||||
Amounts attributable to Quest Diagnostics’ stockholders: | |||||||||||
Income from continuing operations | $ | 709 | $ | 551 | $ | 814 | |||||
Income from discontinued operations, net of taxes | — | 5 | 35 | ||||||||
Net income | $ | 709 | $ | 556 | $ | 849 | |||||
Earnings per share attributable to Quest Diagnostics’ common stockholders - basic: | |||||||||||
Income from continuing operations | $ | 4.92 | $ | 3.80 | $ | 5.35 | |||||
Income from discontinued operations | — | 0.03 | 0.23 | ||||||||
Net income | $ | 4.92 | $ | 3.83 | $ | 5.58 | |||||
Earnings per share attributable to Quest Diagnostics’ common stockholders - diluted: | |||||||||||
Income from continuing operations | $ | 4.87 | $ | 3.78 | $ | 5.31 | |||||
Income from discontinued operations | — | 0.03 | 0.23 | ||||||||
Net income | $ | 4.87 | $ | 3.81 | $ | 5.54 | |||||
Dividends per common share | $ | 1.52 | $ | 1.32 | $ | 1.20 | |||||
The accompanying notes are an integral part of these statements.
F- 3
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME
FOR THE YEARS ENDED DECEMBER 31, 2015, 2014 AND 2013
(in millions)
2015 | 2014 | 2013 | |||||||||
Net income | $ | 753 | $ | 592 | $ | 883 | |||||
Other comprehensive (loss) income: | |||||||||||
Currency translation | (15 | ) | (7 | ) | (27 | ) | |||||
Market valuation, net of tax | — | (1 | ) | (1 | ) | ||||||
Net deferred loss on cash flow hedges, net of tax | 3 | (10 | ) | 2 | |||||||
Other | 1 | (1 | ) | 4 | |||||||
Other comprehensive loss | (11 | ) | (19 | ) | (22 | ) | |||||
Comprehensive income | 742 | 573 | 861 | ||||||||
Less: Comprehensive income attributable to noncontrolling interests | 44 | 36 | 34 | ||||||||
Comprehensive income attributable to Quest Diagnostics | $ | 698 | $ | 537 | $ | 827 | |||||
The accompanying notes are an integral part of these statements.
F- 4
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE YEARS ENDED DECEMBER 31, 2015, 2014 AND 2013
(in millions)
2015 | 2014 | 2013 | |||||||||
Cash flows from operating activities: | |||||||||||
Net income | $ | 753 | $ | 592 | $ | 883 | |||||
Adjustments to reconcile net income to net cash provided by operating activities: | |||||||||||
Depreciation and amortization | 304 | 314 | 283 | ||||||||
Provision for doubtful accounts | 297 | 296 | 270 | ||||||||
Deferred income tax provision | 112 | 23 | 19 | ||||||||
Stock-based compensation expense | 52 | 51 | 28 | ||||||||
Excess tax benefits from stock-based compensation arrangements | (5 | ) | — | (4 | ) | ||||||
Gain on contribution of business to joint venture | (334 | ) | — | — | |||||||
Gain on sale of royalty rights | — | — | (474 | ) | |||||||
Other, net | 6 | (12 | ) | 19 | |||||||
Changes in operating assets and liabilities: | |||||||||||
Accounts receivable | (262 | ) | (312 | ) | (247 | ) | |||||
Accounts payable and accrued expenses | (31 | ) | 68 | (21 | ) | ||||||
Income taxes payable | (41 | ) | (84 | ) | (93 | ) | |||||
Other assets and liabilities, net | (41 | ) | 2 | (11 | ) | ||||||
Net cash provided by operating activities | 810 | 938 | 652 | ||||||||
Cash flows from investing activities: | |||||||||||
Business acquisitions, net of cash acquired | (67 | ) | (728 | ) | (213 | ) | |||||
Proceeds from sale of businesses | — | — | 296 | ||||||||
Proceeds from sale of royalty rights | — | — | 474 | ||||||||
Capital expenditures | (263 | ) | (308 | ) | (231 | ) | |||||
Investment in equity method investee | (33 | ) | — | — | |||||||
Decrease in investments and other assets | 1 | 11 | 2 | ||||||||
Net cash (used in) provided by investing activities | (362 | ) | (1,025 | ) | 328 | ||||||
Cash flows from financing activities: | |||||||||||
Proceeds from borrowings | 2,453 | 2,018 | 896 | ||||||||
Repayments of debt | (2,537 | ) | (1,647 | ) | (900 | ) | |||||
Purchases of treasury stock | (224 | ) | (132 | ) | (1,037 | ) | |||||
Exercise of stock options | 60 | 78 | 138 | ||||||||
Excess tax benefits from stock-based compensation arrangements | 5 | — | 4 | ||||||||
Dividends paid | (212 | ) | (187 | ) | (185 | ) | |||||
Distributions to noncontrolling interests | (42 | ) | (31 | ) | (32 | ) | |||||
Sale of noncontrolling interest in subsidiary | 63 | — | — | ||||||||
Payment of deferred business acquisition consideration | (51 | ) | — | — | |||||||
Other financing activities, net | (22 | ) | (7 | ) | 10 | ||||||
Net cash (used in) provided by financing activities | (507 | ) | 92 | (1,106 | ) | ||||||
Net change in cash and cash equivalents | (59 | ) | 5 | (126 | ) | ||||||
Change in cash and cash equivalents included in assets held for sale | — | — | 17 | ||||||||
Cash and cash equivalents, beginning of year | 192 | 187 | 296 | ||||||||
Cash and cash equivalents, end of year | $ | 133 | $ | 192 | $ | 187 | |||||
The accompanying notes are an integral part of these statements.
F- 5
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
FOR THE YEARS ENDED DECEMBER 31, 2015, 2014 AND 2013
(in millions)
Quest Diagnostics Stockholders’ Equity | ||||||||||||||||||||||||||||
Shares of Common Stock Out- standing | Common Stock | Additional Paid-In Capital | Retained Earnings | Accumulated Other Comprehensive Income (Loss) | Treasury Stock, at Cost | Non- controlling Interests | Total Stock-holders’ Equity | Redeemable Non-controlling Interest | ||||||||||||||||||||
Balance, December 31, 2012 | 158 | $ | 2 | $ | 2,371 | $ | 4,690 | $ | 14 | $ | (2,914 | ) | $ | 23 | $ | 4,186 | $ | — | ||||||||||
Net income | 849 | 34 | 883 | |||||||||||||||||||||||||
Other comprehensive loss, net of tax | (22 | ) | (22 | ) | ||||||||||||||||||||||||
Dividends declared | (181 | ) | (181 | ) | ||||||||||||||||||||||||
Distributions to noncontrolling interests | (32 | ) | (32 | ) | ||||||||||||||||||||||||
Issuance of common stock under benefit plans | 1 | 3 | 17 | 20 | ||||||||||||||||||||||||
Stock-based compensation expense | 24 | 4 | 28 | |||||||||||||||||||||||||
Exercise of stock options | 3 | (9 | ) | 147 | 138 | |||||||||||||||||||||||
Shares to cover employee payroll tax withholdings on stock issued under benefit plans | (1 | ) | (11 | ) | (11 | ) | ||||||||||||||||||||||
Tax benefits associated with stock-based compensation plans | 1 | 1 | ||||||||||||||||||||||||||
Purchases of treasury stock | (17 | ) | (1,037 | ) | (1,037 | ) | ||||||||||||||||||||||
Balance, December 31, 2013 | 144 | $ | 2 | $ | 2,379 | $ | 5,358 | $ | (8 | ) | $ | (3,783 | ) | $ | 25 | $ | 3,973 | $ | — | |||||||||
Net income | 556 | 36 | 592 | |||||||||||||||||||||||||
Other comprehensive loss, net of tax | (19 | ) | (19 | ) | ||||||||||||||||||||||||
Dividends declared | (191 | ) | (191 | ) | ||||||||||||||||||||||||
Distributions to noncontrolling interests | (31 | ) | (31 | ) | ||||||||||||||||||||||||
Issuance of common stock under benefit plans | 1 | 2 | 17 | 19 | ||||||||||||||||||||||||
Stock-based compensation expense | 48 | 3 | 51 | |||||||||||||||||||||||||
Exercise of stock options | 1 | (2 | ) | 80 | 78 | |||||||||||||||||||||||
Shares to cover employee payroll tax withholdings on stock issued under benefit plans | (6 | ) | (6 | ) | ||||||||||||||||||||||||
Tax benefits associated with stock-based compensation plans | (3 | ) | (3 | ) | ||||||||||||||||||||||||
Purchases of treasury stock | (2 | ) | (132 | ) | (132 | ) | ||||||||||||||||||||||
Other | (1 | ) | (1 | ) | ||||||||||||||||||||||||
Balance, December 31, 2014 | 144 | $ | 2 | $ | 2,418 | $ | 5,723 | $ | (27 | ) | $ | (3,815 | ) | $ | 29 | $ | 4,330 | $ | — | |||||||||
Net income | 709 | 42 | 751 | 2 | ||||||||||||||||||||||||
Other comprehensive loss, net of tax | (11 | ) | (11 | ) | ||||||||||||||||||||||||
Dividends declared | (219 | ) | (219 | ) | ||||||||||||||||||||||||
Distributions to noncontrolling interests | (42 | ) | (42 | ) | ||||||||||||||||||||||||
Issuance of common stock under benefit plans | 1 | 6 | 15 | 21 | ||||||||||||||||||||||||
Stock-based compensation expense | 48 | 4 | 52 | |||||||||||||||||||||||||
Exercise of stock options | 1 | 60 | 60 | |||||||||||||||||||||||||
Shares to cover employee payroll tax withholdings on stock issued under benefit plans | (7 | ) | (7 | ) | ||||||||||||||||||||||||
Tax benefits associated with stock-based compensation plans | 5 | 5 | ||||||||||||||||||||||||||
Purchases of treasury stock | (3 | ) | (224 | ) | (224 | ) | ||||||||||||||||||||||
Sale of redeemable noncontrolling interest | 11 | 11 | 54 | |||||||||||||||||||||||||
Adjustment to fair value | (14 | ) | (14 | ) | 14 | |||||||||||||||||||||||
Balance, December 31, 2015 | 143 | $ | 2 | $ | 2,481 | $ | 6,199 | $ | (38 | ) | $ | (3,960 | ) | $ | 29 | $ | 4,713 | $ | 70 | |||||||||
The accompanying notes are an integral part of these statements.
F- 6
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in millions unless otherwise indicated)
1. DESCRIPTION OF BUSINESS
Background
Quest Diagnostics Incorporated and its subsidiaries ("Quest Diagnostics" or the "Company") empower people to take action to improve health outcomes. The Company uses its extensive database of clinical lab results to derive diagnostic insights that reveal new avenues to identify and treat disease, inspire healthy behaviors and improve healthcare management. The Company's diagnostic information services business ("DIS") provides insights through clinical testing and related services to patients, physicians, hospitals, accountable care organizations ("ACOs"), integrated delivery networks ("IDNs"), health plans, employers and others. The Company offers the broadest access in the United States to diagnostic information services through its nationwide network of laboratories, Company-owned patient service centers and phlebotomists in physician offices. The Company is the world's leading provider of diagnostic information services, which includes providing clinical testing services such as routine (including drugs-of-abuse) testing, gene-based and esoteric testing, and anatomic pathology services, as well as related services and insights. The Company provides interpretive consultation with one of the largest medical and scientific staffs in the industry and hundreds of M.D.s and Ph.D.s, many of whom are recognized leaders in their fields. The Company's Diagnostic Solutions ("DS") businesses offer a variety of solutions for life insurers, healthcare providers and others. The Company is the leading provider of risk assessment services for the life insurance industry as well as a leading provider of central laboratory testing for clinical trials (see Note 6 regarding the contribution of the clinical trials testing business to a newly formed joint venture effective July 1, 2015). The Company's diagnostics products business manufactures and markets diagnostic products. In addition, the Company offers healthcare organizations, clinicians and patients robust information technology solutions.
During 2015, Quest Diagnostics processed approximately 157 million test requisitions through its extensive laboratory network.
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Principles of Consolidation
The consolidated financial statements include the accounts of all entities controlled by the Company through its direct or indirect ownership of a majority voting interest and the accounts of any variable interest entities ("VIEs") where the Company is subject to a majority of the risk of loss from the variable interest entity's activities, or entitled to receive a majority of the entity's residual returns, or both. The Company assesses the requirements related to the consolidation of VIEs, including a qualitative assessment of power and economics that considers which entity has the power to direct the activities that “most significantly impact” the VIEs economic performance and has the obligation to absorb losses of, or the right to receive benefits that could be potentially significant to, the VIE. The Company did not have any VIEs at both December 31, 2015 and 2014. All significant intercompany accounts and transactions are eliminated in consolidation.
Equity Method Investments
Investments in entities which the Company does not control, but in which it has a substantial ownership interest (generally between 20% and 49%) and can exercise significant influence, are accounted for using the equity method of accounting. These investments are classified as investment in equity method investees in the consolidated balance sheets. The Company records its pro rata share of the earnings, adjusted for accretion of basis difference, of these investments in equity in earnings of equity method investees, net of taxes in the consolidated statements of operations. The Company reviews its investments in equity method investees for impairment whenever events or changes in circumstances indicate that the carrying amounts may not be recoverable.
Basis of Presentation
In December 2012, the Company committed to a plan to sell its HemoCue diagnostics products business ("HemoCue"). In April 2013, the Company completed the sale of HemoCue. During the third quarter of 2006, the Company completed the wind-down of NID, a test kit manufacturing subsidiary. The accompanying consolidated statements of operations and related disclosures report the results of HemoCue and NID as discontinued operations for all periods presented. See Note 18 for a further discussion of discontinued operations.
F- 7
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Reclassifications
Prior to the Company's clinical trials central laboratory services joint venture, Q2 Solutions, the earnings of the Company's equity method investees consisted of earnings that were not directly taxable to the investees, in which case it was appropriate to present equity in earnings of equity method investees before income tax expense on the consolidated statements of operations. The earnings of Q2 Solutions, which closed on July 1, 2015, includes earnings that are directly taxable to the joint venture. As a result of the Q2 Solutions transaction, the current period presentation of equity in earnings of equity method investees is required to be presented below income tax expense on the consolidated statements of operations. The Company's equity in earnings of equity method investees on the consolidated statements of operations for the years ended December 31, 2014 and 2013 have been reclassified to conform with the current period presentation. In addition as a result of the current period presentation of investment in equity method investees on the consolidated balance sheet, the Company's investment in equity method investees as of December 31, 2014 has been reclassified to conform with the current period presentation. For further details regarding the Company's investment in Q2 Solutions, see Note 6.
As a result of the classification of certain non-core assets as held for sale on the consolidated balance sheet in the current period, current assets held for sale on the consolidated balance sheet at December 31, 2014 were reclassified to conform with the current period presentation. For further details regarding assets held for sale, see Note 6.
As a result of the early adoption of the accounting standard update (“ASU”) associated with classification of debt issuance costs, certain reclassifications have been made to the prior period financial statements to conform with the current period presentation. For further details regarding the impact of the ASU, see New Accounting Standards.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States (“GAAP”) requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Revenue Recognition
The Company primarily recognizes revenue for services rendered upon completion of the testing process. Billings for services reimbursed by third-party payers, including Medicare and Medicaid, are recorded as revenues net of allowances for differences between amounts billed and the estimated receipts from such payers. Adjustments to the allowances, based on actual receipts from the third-party payers, are recorded upon settlement. Billings to the Medicare and Medicaid programs were approximately 17%, 17% and 18% of the Company's consolidated net revenues for the years ended December 31, 2015, 2014 and 2013, respectively. Under capitated arrangements with healthcare insurers, the Company recognizes revenue based on a predetermined monthly reimbursement rate for each member of an insurer's health plan regardless of the number or cost of services provided by the Company.
Revenues from the Company's risk assessment services, clinical trials testing (see Note 6 regarding the contribution of the clinical trials testing business to a newly formed joint venture effective July 1, 2015), healthcare information technology
and diagnostics products businesses are recognized when persuasive evidence of a final agreement exists; delivery has occurred or services have been rendered; the price of the product or service is fixed or determinable; and collectibility from the customer is reasonably assured.
F- 8
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Taxes on Income
The provision for income taxes represents income taxes paid or payable for the current year plus the change in deferred taxes during the year. Current and deferred income taxes are measured based on the tax laws that are enacted as of the balance sheet date of the relevant reporting period. Deferred tax assets and liabilities are recognized for the expected future tax consequences of differences between the carrying amounts of assets and liabilities and their respective tax bases using tax rates in effect for the year in which the differences are expected to reverse. A valuation allowance is provided when it is more likely than not that some portion or all of the deferred tax assets will not be realized. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period when the change is enacted. Tax benefits from uncertain tax positions are recognized only if the tax position is more likely than not to be sustained upon examination by taxing authorities based on the technical merits of the position.
Earnings Per Share
The Company's unvested restricted stock units that contain non-forfeitable rights to dividends are participating securities and, therefore, are included in the earnings allocation in computing earnings per share using the two-class method. Basic earnings per common share is calculated by dividing net income, adjusted for earnings allocated to participating securities, by the weighted average number of common shares outstanding. Diluted earnings per common share is calculated by dividing net income, adjusted for earnings allocated to participating securities, by the weighted average number of common shares outstanding after giving effect to all potentially dilutive common shares outstanding during the period. Potentially dilutive common shares include the dilutive effect of outstanding stock options and performance share units granted under the Company's Amended and Restated Employee Long-Term Incentive Plan (“ELTIP”) and its Amended and Restated Non-Employee Director Long-Term Incentive Plan (“DLTIP”). Earnings allocable to participating securities include the portion of dividends declared as well as the portion of undistributed earnings during the period allocable to participating securities.
Stock-Based Compensation
The Company records stock-based compensation as a charge to earnings net of the estimated impact of forfeited awards. As such, the Company recognizes stock-based compensation cost only for those stock-based awards that are estimated to ultimately vest over their requisite service period, based on the vesting provisions of the individual grants. The cumulative effect on current and prior periods of a change in the estimated forfeiture rate is recognized as compensation cost in earnings in the period of the change. The terms of the Company's performance share unit awards allow the recipients of such awards to earn a variable number of shares based on the achievement of the performance goals specified in the awards. Stock-based compensation expense associated with performance share units is recognized based on management's best estimates of the achievement of the performance goals specified in such awards and the resulting number of shares that will be earned. The cumulative effect on current and prior periods of a change in the estimated number of performance share units expected to be earned is recognized as compensation cost in earnings in the period of the change. The Company recognizes stock-based compensation expense related to the Company's Amended Employee Stock Purchase Plan (“ESPP”) based on the 15% discount at purchase. For further details regarding stock-based compensation, see Note 16.
Fair Value Measurements
The Company determines fair value measurements used in its consolidated financial statements based upon the exit price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants exclusive of any transaction costs, as determined by either the principal market or the most advantageous market.
Inputs used in the valuation techniques to derive fair values are classified based on a three-level hierarchy. The basis for fair value measurements for each level within the hierarchy is described below with Level 1 having the highest priority and Level 3 having the lowest.
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Quoted prices for similar assets or liabilities in active markets; quoted prices for identical or similar instruments in markets that are not active; and model-derived valuations in which all significant inputs are observable in active markets.
Level 3: Valuations derived from valuation techniques in which one or more significant inputs are unobservable.
F- 9
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Foreign Currency
The Company predominately uses the U.S. dollar as its functional currency. The functional currency of the Company's foreign operating subsidiaries generally is the applicable local currency. Assets and liabilities denominated in non-U.S. dollars are translated into U.S. dollars at exchange rates as of the end of the reporting period. Income and expense items are translated at average exchange rates prevailing during the year. The translation adjustments are recorded as a component of accumulated other comprehensive loss within stockholders' equity. Gains and losses from foreign currency transactions are included within other operating expense (income), net in the consolidated statements of operations. Transaction gains and losses have historically not been material. The Company may be exposed to market risk for changes in foreign exchange rates primarily under certain intercompany receivables and payables. From time to time, the Company uses foreign exchange forward contracts to mitigate the exposure of the eventual net cash inflows or outflows resulting from these intercompany transactions. As a result of the HemoCue disposition, this foreign currency risk has largely been eliminated. The Company's remaining foreign exchange exposure is not material to the Company's consolidated financial condition. The Company does not hedge its net investment in non-U.S. subsidiaries because it views those investments as long-term in nature.
Cash and Cash Equivalents
Cash and cash equivalents include all highly-liquid investments with original maturities, at the time acquired by the Company, of three months or less.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk are principally cash, cash equivalents, short-term investments, accounts receivable and derivative financial instruments. The Company's policy is to place its cash, cash equivalents and short-term investments in highly-rated financial instruments and institutions. Concentration of credit risk with respect to accounts receivable is mitigated by the diversity of the Company's payers and their dispersion across many different geographic regions, and is limited to certain payers who are large buyers of the Company's services. To reduce risk, the Company routinely assesses the financial strength of these payers and, consequently, believes that its accounts receivable credit risk exposure, with respect to these payers, is limited. While the Company has receivables due from federal and state governmental agencies, the Company does not believe that such receivables represent a credit risk since the related healthcare programs are funded by federal and state governments, and payment is primarily dependent on submitting appropriate documentation. At December 31, 2015 and 2014, receivables due from government payers under the Medicare and Medicaid programs represent approximately 16% and 14%, respectively, of the Company's consolidated net accounts receivable. The portion of the Company's accounts receivable due from patients comprises the largest portion of credit risk. At both December 31, 2015 and 2014, receivables due from patients represent approximately 17% of the Company's consolidated net accounts receivable. The Company applies assumptions and judgments including historical collection experience for assessing collectibility and determining allowances for doubtful accounts for accounts receivable from patients.
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable are reported at realizable value, net of allowances for doubtful accounts, which is estimated and recorded in the period the related revenue is recorded. The Company has a standardized approach to estimate and review the collectibility of its receivables based on a number of factors, including the period they have been outstanding. Historical collection and payer reimbursement experience is an integral part of the estimation process related to allowances for doubtful accounts. In addition, the Company regularly assesses the state of its billing operations in order to identify issues which may impact the collectibility of these receivables or reserve estimates. Changes to the allowances for doubtful accounts estimates are recorded as an adjustment to bad debt expense within selling, general and administrative expenses in the consolidated statements of operations. Receivables deemed to be uncollectible are charged against the allowance for doubtful accounts at the time such receivables are written-off. Recoveries of receivables previously written-off are recorded as credits to the allowance for doubtful accounts.
Inventories
Inventories, which consist principally of testing supplies and reagents, are valued at the lower of cost (first in, first out method) or market.
F- 10
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Property, Plant and Equipment
Property, plant and equipment is recorded at cost. Major renewals and improvements are capitalized, while maintenance and repairs are expensed as incurred. Costs incurred for computer software developed or obtained for internal use are capitalized for application development activities and expensed as incurred for preliminary project activities and post-implementation activities. Capitalized costs include external direct costs of materials and services consumed in developing or obtaining internal-use software, payroll and payroll-related costs for employees who are directly associated with the internal-use software project, and interest costs incurred, when material, while developing internal-use software. Capitalization of such costs ceases when the project is substantially complete and ready for its intended purpose. Costs for maintenance and training are expensed as incurred. The Company capitalizes interest on borrowings during the active construction period of major capital projects. Capitalized interest is added to the cost of the underlying assets and is amortized over the expected useful lives of the assets. Depreciation and amortization are provided on the straight-line method over expected useful asset lives as follows: buildings and improvements, ranging up to thirty-one and a half years; laboratory equipment and furniture and fixtures, ranging from three to seven years; leasehold improvements, the lesser of the useful life of the improvement or the remaining life of the building or lease, as applicable; and computer software developed or obtained for internal use, ranging from three to five years.
Goodwill
Goodwill represents the excess of the fair value of the acquiree (including the fair value of non-controlling interests) over the recognized bases of the net identifiable assets acquired and includes the future economic benefits from other assets that could not be individually identified and separately recognized. Goodwill is not amortized, but instead is periodically reviewed for impairment and an impairment charge is recorded in the periods in which the recorded carrying value of goodwill is more than its implied fair value. The goodwill test is performed at least annually, or more frequently, in the case of other events that indicate a potential impairment.
The annual impairment test includes an option to perform a qualitative assessment of whether it is more-likely-than-not that a reporting unit's fair value is less than its carrying value prior to, or as an alternative to, performing the two-step quantitative goodwill impairment test. If, after assessing the totality of events or circumstances, the Company determines that it is more-likely-than-not that the fair value of a reporting unit is less than its carrying value, then it is required to perform the first step of the two-step goodwill impairment test. Otherwise, no further analysis is required.
The quantitative impairment test is a two-step process that begins with the estimation of the fair value of the reporting unit. The first step screens for potential impairment and the second step measures the amount of the impairment, if any. The fair value of the reporting unit is based upon either a discounted cash flows analysis that converts future cash flow amounts into a single discounted present value amount or a market approach. The Company assesses the valuation methodology based upon the relevance and availability of the data at the time it performs the valuation. As part of the first step to assess potential impairment, management compares the estimate of fair value for the reporting unit to the carrying value of the reporting unit. If the carrying value is greater than the estimate of fair value, the Company would then proceed to the second step to measure the impairment, if any. The second step compares the implied fair value of goodwill with its carrying value. The implied fair value is determined by allocating the fair value of the reporting unit to all of the assets and liabilities of that unit as if the reporting unit had been acquired in a business combination and the fair value of the reporting unit was the purchase price paid to acquire the reporting unit. The excess of the fair value of the reporting unit over the amounts assigned to its assets and liabilities is the implied fair value of goodwill. If the carrying amount of the reporting unit's goodwill is greater than its implied fair value, an impairment loss will be recognized in the amount of the excess.
On a quarterly basis, management performs a review of the Company's business to determine if events or changes in circumstances have occurred which could have a material adverse effect on the fair value of the Company and its goodwill. If such events or changes in circumstances were deemed to have occurred, the Company would perform an impairment test of goodwill as of the end of the quarter, consistent with the annual impairment test performed during the fourth quarter of the fiscal year ended December 31st, and record any noted impairment loss.
For year ended December 31, 2015, the Company performed the qualitative assessment for its DIS and risk assessment services reporting units. Based on the totality of information available for the DIS and risk assessment services reporting units, the Company concluded that it was more likely than not that the estimated fair values were greater than the carrying values of the reporting units, and as such, no further analysis was required. For the diagnostic products reporting unit, the Company
F- 11
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
performed step one of the goodwill impairment test and concluded that goodwill of the reporting unit was not impaired. For the year ended December 31, 2014, the Company elected to bypass the qualitative assessment, performed step one of the goodwill impairment test for all of its reporting units and concluded that goodwill of each reporting unit was not impaired.
Intangible Assets
Intangible assets are recognized at fair value, as an asset apart from goodwill if the asset arises from contractual or other legal rights, or if it is separable. Intangible assets, principally representing the cost of customer-related intangibles, non-competition agreements and technology acquired, are capitalized and amortized on the straight-line method over their expected useful life, which generally ranges from five to twenty years. Intangible assets with indefinite useful lives, consisting principally of acquired tradenames, are not amortized, but instead are periodically reviewed for impairment.
The Company reviews indefinite-lived intangible assets periodically for impairment and an impairment charge is recorded in the periods in which the recorded carrying value of indefinite-lived intangibles is more than its estimated fair value. The indefinite-lived intangible asset impairment test is performed at least annually, or more frequently in the case of other events that indicate a potential impairment.
Based upon the Company’s most recent annual impairment tests completed during the fourth quarter of the years ended December 31, 2015 and 2014, the Company concluded that indefinite-lived intangible assets were not impaired.
The Company reviews the recoverability of its long-lived assets (including amortizable intangible assets), other than goodwill and indefinite-lived intangible assets, when events or changes in circumstances occur that indicate that the carrying value of the asset may not be recoverable. Evaluation of possible impairment is based on the Company's ability to recover the asset from the expected future pre-tax cash flows (undiscounted and without interest charges) of the related operations. If the expected undiscounted pre-tax cash flows are less than the carrying amount of such asset, an impairment loss is recognized for the difference between the estimated fair value and carrying amount of the asset.
Investments
The Company's investments, which are included in other assets in the consolidated balance sheets, are comprised of trading securities, available-for-sale securities and other investments. The classification of an investment depends on our intent and ability to hold the investment.
• | Trading securities represent participant-directed investments of deferred employee compensation and related Company matching contributions held in trusts pursuant to the Company's supplemental deferred compensation plans (see Note 16). Trading securities are carried at fair value with both realized and unrealized gains and losses recorded currently in earnings as a component of non-operating expenses within other (expense) income, net in the consolidated statements of operations. For the years ended December 31, 2015, 2014 and 2013, gains from trading equity securities totaled $0 million, $3 million, and $7 million, respectively. |
• | Available-for-sale equity securities consists of an investment in registered shares of a public corporation. Available-for-sale equity securities are carried at fair value with unrealized gains and losses, net of tax, recorded as a component of accumulated other comprehensive loss within stockholders' equity and realized gains and losses recorded in other (expense) income, net in the consolidated statements of operations. At December 31, 2015, the Company had gross unrealized losses from available-for-sale equity securities of $2 million. |
• | Other investments do not have readily determinable fair values and consist of investments in preferred and common shares of privately held companies and are accounted for under the cost method. |
Gains and losses on securities sold are based on the average cost method. The Company periodically reviews its investments to determine whether a decline in fair value below the cost basis is other-than-temporary. The primary factors considered in the determination are: the length of time that the fair value of the investment is below carrying value; the financial condition, operating performance and near-term prospects of the investee; and the Company's intent and ability to hold the investment for a period of time sufficient to allow for a recovery in fair value. If the decline in fair value is deemed to be other-than-temporary, the cost basis of the security is written down to fair value.
F- 12
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Investments at December 31, 2015 and 2014 consisted of the following:
2015 | 2014 | ||||||
Available-for-sale equity securities | $ | 6 | $ | 9 | |||
Trading equity securities | 49 | 49 | |||||
Other investments | 8 | 8 | |||||
Total | $ | 63 | $ | 66 | |||
Derivative Financial Instruments
The Company uses derivative financial instruments to manage its exposure to market risks for changes in interest rates and, from time to time, foreign currencies. This strategy includes the use of interest rate swap agreements, forward starting interest rate swap agreements, interest rate lock agreements and foreign currency forward contracts to manage its exposure to movements in interest and currency rates. The Company has established policies and procedures for risk assessment and the approval, reporting and monitoring of derivative financial instrument activities. These policies prohibit holding or issuing derivative financial instruments for speculative purposes. The Company does not enter into derivative financial instruments that contain credit-risk-related contingent features or requirements to post collateral.
Interest Rate Risk
The Company is exposed to interest rate risk on its cash and cash equivalents and its debt obligations. Interest income earned on cash and cash equivalents may fluctuate as interest rates change; however, due to their relatively short maturities, the Company does not hedge these assets or their investment cash flows and the impact of interest rate risk is not material. The Company's debt obligations consist of fixed-rate and variable-rate debt instruments. The Company's primary objective is to achieve the lowest overall cost of funding while managing the variability in cash outflows within an acceptable range. In order to achieve this objective, the Company has entered into interest rate swaps. Interest rate swaps involve the periodic exchange of payments without the exchange of underlying principal or notional amounts. Net settlements between the counterparties are recognized as an adjustment to interest expense.
The Company accounts for these derivatives as either an asset or liability measured at its fair value. The fair value is based upon model-derived valuations in which all significant inputs are observable in active markets and includes an adjustment for the credit risk of the obligor's non-performance. For a derivative instrument that has been formally designated as a fair value hedge, fair value gains or losses on the derivative instrument are reported in earnings, together with offsetting fair value gains or losses on the hedged item that are attributable to the risk being hedged. For derivatives that have been formally designated as a cash flow hedge, the effective portion of changes in the fair value of the derivatives is recorded in accumulated other comprehensive loss and the ineffective portion is recorded in earnings. Upon maturity or early termination of an effective interest rate swap designated as a cash flow hedge, unrealized gains or losses are deferred in stockholders' equity, as a component of accumulated other comprehensive loss, and are amortized as an adjustment to interest expense over the period during which the hedged forecasted transaction affects earnings, which is when the Company recognizes interest expense on the hedged cash flows. At inception and quarterly thereafter, the Company formally assesses whether the derivatives that are used in hedging transactions are highly effective in offsetting changes in the fair value or cash flows of the hedged item. All components of each derivative financial instrument's gain or loss are included in the assessment of hedge effectiveness. If it is determined that a derivative ceases to be a highly effective hedge, the Company discontinues hedge accounting and any deferred gains or losses related to a discontinued cash flow hedge shall continue to be reported in accumulated other comprehensive loss, unless it is probable that the forecasted transaction will not occur. If it is probable that the forecasted transaction will not occur by the originally specified time period, the Company discontinues hedge accounting, and any deferred gains or losses reported in accumulated other comprehensive loss are classified into earnings immediately.
Comprehensive Income (Loss)
Comprehensive income (loss) encompasses all changes in stockholders' equity (except those arising from transactions with stockholders) and includes net income, net unrealized gains or losses on available-for-sale securities, foreign currency translation adjustments and deferred gains and losses related to certain derivative financial instruments (see Note 15).
F- 13
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
New Accounting Standards
In May 2014, the FASB issued an ASU on revenue recognition. This ASU outlines a single comprehensive model to use in accounting for revenue arising from contracts with customers. This standard supersedes existing revenue recognition requirements and eliminates most industry-specific guidance from GAAP. The core principle of the revenue recognition standard is to require an entity to recognize as revenue the amount that reflects the consideration to which it expects to be entitled in exchange for goods or services as it transfers control to its customers. The standard requires additional disclosures including those that are qualitative and quantitative disclosures about the nature, amount, timing and uncertainty of revenue and cash flows arising from contracts with customers. In August 2015, the FASB deferred the effective date of this ASU to the first quarter of 2018, with early adoption permitted beginning in the first quarter of 2017. The ASU can be applied using a full retrospective method or a modified retrospective method of adoption. The Company is currently assessing the impact of the adoption of this ASU on the Company’s results of operations, financial position and cash flows.
In February 2015, the FASB issued an ASU which makes targeted amendments to the current consolidation guidance for variable interest entities and limited partnerships and similar entities. The ASU is effective for the Company in the first quarter of 2016 with the option of using a full retrospective or a modified retrospective method. The Company does not expect the adoption of this standard to have a material impact on its results of operations, financial position and cash flows.
In April 2015, the FASB issued an ASU which requires that debt issuance costs be presented in the balance sheet as a direct reduction to the carrying amount of the associated debt liability, consistent with debt discounts. The Company adopted this standard in the second quarter of 2015 and applied the standard retrospectively. Adoption of this standard has resulted in the reclassification of $20 million of unamortized debt issuance costs from other assets to a reduction of long-term debt on the consolidated balance sheet at December 31, 2014.
In April 2015, the FASB issued an ASU which provides guidance in determining whether a cloud computing arrangement includes a software license. If it is determined that a cloud computing arrangement does include a software license, the software element of the arrangement should be accounted for consistent with the acquisition of other software licenses. If the arrangement does not include a software license, it should be accounted for as a service contract. The ASU is effective for the Company in the first quarter of 2016 and can be applied prospectively to all arrangements entered into or materially modified after the effective date or retrospectively. The Company does not expect the adoption of this ASU to have a material impact on its results of operations, financial position and cash flows.
In September 2015, the FASB issued an ASU which requires that an acquirer recognize adjustments to provisional amounts in a business combination that are identified during the measurement period in the reporting period in which the adjustment amounts are determined, including the cumulative effect of the change in provisional amount as if the accounting had been completed at the acquisition date. The ASU is effective for the Company in the first quarter of 2016 and will be applied prospectively to adjustments to provisional amounts after the effective date. The Company does not expect the adoption of this ASU to have a material impact on its results of operations, financial position and cash flows.
In November 2015, the FASB issued an ASU which requires that all deferred income tax assets and liabilities be classified as non-current in the balance sheet. The current requirement that deferred tax assets and liabilities of a tax-paying component of an entity be offset and presented as a single amount is not affected by the new guidance. The Company adopted this standard in the fourth quarter of 2015 and elected to apply the standard prospectively. As such, prior periods were not retrospectively adjusted. Adoption of this standard resulted in $168 million of deferred taxes associated with current assets and liabilities being classified as non-current deferred tax assets of $26 million and as a reduction of non-current deferred tax liabilities of $142 million on the consolidated balance sheet as of December 31, 2015.
In January 2016, the FASB issued an ASU on the recognition and measurement of financial assets and financial liabilities. This ASU requires that all equity investments (except those accounted for under the equity method of accounting or those that result in consolidation of the investee) be measured at fair value with changes in fair value recognized in net income. However, companies may elect to measure equity investments that do not have readily determinable fair values at cost minus impairment, if any, plus or minus changes resulting from observable price changes in orderly transactions for the identical or a similar investment of the same issuer. In addition, the ASU eliminates the requirement to disclose the method and significant assumptions used to estimate the fair value for financial instruments measured at amortized cost on the balance sheet. The ASU is effective for the Company in the first quarter of 2018. The Company does not expect the adoption of this ASU to have a material impact on its results of operations, financial position and cash flows.
F- 14
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
3. EARNINGS PER SHARE
The computation of basic and diluted earnings per common share is as follows (in millions, except per share data):
2015 | 2014 | 2013 | |||||||||
Amounts attributable to Quest Diagnostics’ stockholders: | |||||||||||
Income from continuing operations | $ | 709 | $ | 551 | $ | 814 | |||||
Income from discontinued operations, net of taxes | — | 5 | 35 | ||||||||
Net income attributable to Quest Diagnostics’ common stockholders | $ | 709 | $ | 556 | $ | 849 | |||||
Income from continuing operations | $ | 709 | $ | 551 | $ | 814 | |||||
Less: Earnings allocated to participating securities | 3 | 2 | 3 | ||||||||
Earnings available to Quest Diagnostics’ common stockholders – basic and diluted | $ | 706 | $ | 549 | $ | 811 | |||||
Weighted average common shares outstanding – basic | 144 | 145 | 152 | ||||||||
Effect of dilutive securities: | |||||||||||
Stock options and performance share units | 1 | — | 1 | ||||||||
Weighted average common shares outstanding – diluted | 145 | 145 | 153 | ||||||||
Earnings per share attributable to Quest Diagnostics’ common stockholders – basic: | |||||||||||
Income from continuing operations | $ | 4.92 | $ | 3.80 | $ | 5.35 | |||||
Income from discontinued operations | — | 0.03 | 0.23 | ||||||||
Net income | $ | 4.92 | $ | 3.83 | $ | 5.58 | |||||
Earnings per share attributable to Quest Diagnostics’ common stockholders – diluted: | |||||||||||
Income from continuing operations | $ | 4.87 | $ | 3.78 | $ | 5.31 | |||||
Income from discontinued operations | — | 0.03 | 0.23 | ||||||||
Net income | $ | 4.87 | $ | 3.81 | $ | 5.54 | |||||
The following securities were not included in the calculation of diluted earnings per share due to their antidilutive effect:
2015 | 2014 | 2013 | ||||||
Stock options and performance share units | 2 | 2 | 1 | |||||
4. RESTRUCTURING ACTIVITIES
Invigorate Program
During 2012, the Company committed to a course of action related to a multi-year program called Invigorate which is
designed to reduce its cost structure. Invigorate has consisted of several flagship programs, with structured plans in each, to drive savings and improve performance across the customer value chain. These flagship programs include: organization excellence; information technology excellence; procurement excellence; service excellence; lab excellence; and billing excellence. From 2012 through 2014, the Invigorate program was intended to partially offset reimbursement pressures and labor and benefit cost increases; free up additional resources to invest in science, innovation and other growth initiatives; and enable us to improve service quality and operating profitability.
F- 15
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
In January 2015, the Company adopted a course of action related to its multi-year Invigorate program to further reduce its cost structure through 2017. This multi-year course of action continues to focus on the flagship program opportunities and new key opportunities such as: standardizing processes, information technology systems, equipment and data; enhancing electronic enabling services; and enhancing reimbursement for work performed.
The following table provides a summary of the Company's pre-tax restructuring charges associated with its Invigorate program and other restructuring activities:
2015 | 2014 | 2013 | |||||||||
Employee separation costs | $ | 38 | $ | 31 | $ | 69 | |||||
Facility-related costs | 1 | 12 | 6 | ||||||||
Asset impairment charges | — | 1 | — | ||||||||
Accelerated vesting of stock-based compensation | — | — | 1 | ||||||||
Total restructuring charges | $ | 39 | $ | 44 | $ | 76 | |||||
Total restructuring charges incurred in the year ended December 31, 2015 are primarily associated with various workforce reduction initiatives as the Company continued to simplify and restructure its organization. Of the total $39 million in restructuring charges incurred during the year ended December 31, 2015, $32 million and $7 million were recorded in cost of services and selling, general and administrative expenses, respectively.
Total restructuring charges incurred in the year ended December 31, 2014 are primarily associated with various workforce reduction initiatives as the Company continued to simplify and restructure its organization. Of the total $44 million in restructuring charges incurred during the year ended December 31, 2014, $21 million and $23 million were recorded in cost of services and selling, general and administrative expenses, respectively.
Total restructuring charges incurred during the year ended December 31, 2013 included $29 million of employee separation costs associated with various workforce reduction initiatives aimed at centralizing certain support functions, $20 million associated with the Company's management layer reduction initiative, $16 million associated with the outsourcing of certain aspects of the Company's support functions and $4 million associated with the Company's voluntary retirement program. Of the total $76 million in restructuring charges incurred during the year ended December 31, 2013, $27 million and $49 million were recorded in cost of services and selling, general and administrative expenses, respectively.
Charges for all periods presented were primarily recorded in the Company's DIS business.
The following table summarizes the activity of the restructuring liability as of December 31, 2015 and 2014, which is included in accrued expenses in Note 12:
Employee Separation Costs | Facility-Related Costs | Total | |||||||||
Balance, December 31, 2013 | $ | 31 | $ | 5 | $ | 36 | |||||
Income statement expense | 31 | 12 | 43 | ||||||||
Cash payments | (44 | ) | (6 | ) | (50 | ) | |||||
Balance, December 31, 2014 | 18 | 11 | 29 | ||||||||
Income statement expense | 38 | 1 | 39 | ||||||||
Cash payments | (40 | ) | (6 | ) | (46 | ) | |||||
Other / adjustments | — | (3 | ) | (3 | ) | ||||||
Balance, December 31, 2015 | $ | 16 | $ | 3 | $ | 19 | |||||
F- 16
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
5. BUSINESS ACQUISITIONS
2015 Acquisitions
Acquisition of MemorialCare Health System's Laboratory Outreach Business
On August 3, 2015, the Company completed the acquisition of MemorialCare Health System's laboratory outreach business ("MemorialCare") in an all-cash transaction valued at $35 million. The assets acquired primarily represent tax deductible goodwill and intangible assets, principally comprised of customer-related intangibles.
Acquisition of the Business Assets of Superior Mobile Medics, Inc.
On November 16, 2015, the Company completed the acquisition of the business assets of Superior Mobile Medics, Inc. ("Superior Mobile Medics"), a national provider of paramedical and health data collection services to the life insurance and employer health and wellness industries, in an all-cash transaction valued at $27 million. The assets acquired primarily represent accounts receivable, tax deductible goodwill and intangible assets, principally comprised of customer-related intangibles.
2014 Acquisitions
Acquisition of Solstas Lab Partners Group
On March 7, 2014, the Company completed its acquisition of Solstas Lab Partners Group and its subsidiaries ("Solstas") in an all-cash transaction valued at $572 million, or $563 million net of cash acquired. The Company financed the acquisition with borrowings under its secured receivables credit facility and senior unsecured revolving credit facility. The final consideration paid is subject to post closing adjustments related to working capital and other items. Through the acquisition, the Company acquired all of Solstas' operations. Solstas is a full-service commercial laboratory based in Greensboro, North Carolina and operates in nine states throughout the southeastern United States, including the Carolinas, Virginia, Tennessee, Georgia and Alabama.
For the year ended December 31, 2014, Solstas contributed $300 million to the Company's consolidated net revenues and $294 million to operating expenses which included approximately $17 million of restructuring, integration and transaction related costs. Of the $17 million of restructuring, integration and transaction related costs recorded for the year ended December 31, 2014, $4 million and $13 million were in cost of services and selling, general and administrative expenses, respectively.
Acquisition of Summit Health, Inc.
On April 18, 2014, the Company completed its acquisition of Summit Health, Inc. ("Summit Health") for $152 million, which consisted of cash consideration of $125 million (which included $10 million of working capital adjustments), or $124 million net of cash acquired, estimated contingent consideration of $22 million and $5 million associated with certain transaction related costs due to the sellers of Summit Health. The contingent consideration arrangement was dependent on the achievement of certain revenue targets in 2015, with a maximum payment of $25 million in 2016. Based on actual 2015 revenue results for Summit Health compared to the earn-out target included in the contingent consideration arrangement, no payment is required, and therefore, the contingent consideration accrual was $0 at December 31, 2015. At December 31, 2014, the contingent consideration accrual was $13 million. Through the acquisition, the Company acquired all of Summit Health's operations. Summit is a provider of on-site prevention and wellness programs. For further details regarding the fair value of the estimated contingent consideration associated with the Summit Health acquisition, see Note 7.
F- 17
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Acquisition of Steward Health Care Systems, LLC's Laboratory Outreach Business
On April 16, 2014, the Company completed the acquisition of the laboratory outreach service operations of Steward Health Care Systems, LLC ("Steward") for $34 million, which consisted of cash consideration of $30 million and contingent consideration of $4 million. The assets acquired primarily represent goodwill and intangible assets, principally comprised of customer-related intangibles. The contingent consideration arrangement secures the seller's compliance with a non-compete agreement under which the Company will pay up to $5 million, ratably through 2018, provided the non-compete agreement is not violated. For further details regarding the fair value of the estimated contingent consideration associated with the Steward acquisition, see Note 7.
2013 Acquisitions
During 2013, the Company completed four acquisitions for a total purchase price of $264 million, or $213 million net of cash acquired and deferred consideration associated with the UMass acquisition.
Acquisition of UMass Memorial Medical Center's Laboratory Outreach Business
On January 2, 2013, the Company completed the acquisition of the clinical outreach and anatomic pathology businesses of UMass Memorial Medical Center ("UMass"). The purchase price included $50 million of deferred consideration that is included in accounts payable and accrued expenses at December 31, 2014 and was paid in 2015. This purchase was the first step in a series of transactions between the parties whereby the two organizations expected to eventually have a financial stake in a new entity performing diagnostic information testing services in a defined territory within the state of Massachusetts (see Redeemable Noncontrolling Interest section of Note 15). The assets acquired at the acquisition date primarily represent goodwill and intangible assets, principally comprised of customer-related intangibles. In addition the Company granted to UMass a call option and UMass granted to the Company a put option for UMass to acquire an 18.90% equity interest in a newly formed entity. For further details regarding the put and call options, see Note 7.
Acquisition of Advanced Toxicology Network
On May 15, 2013, the Company completed the acquisition of the toxicology and clinical laboratory business of Advanced Toxicology Network ("ATN") from Concentra, a subsidiary of Humana Inc. The assets acquired at the acquisition date primarily represent goodwill and intangible assets, principally comprised of customer-related intangibles.
Acquisition of Dignity Health's Laboratory Outreach Business
On June 22, 2013, the Company completed the acquisition of certain lab-related clinical outreach service operations of Dignity Health ("Dignity"), a hospital system in California. The assets acquired at the acquisition date primarily represent goodwill and intangible assets, principally comprised of customer-related intangibles.
Acquisition of ConVerge Diagnostics, LLC
On October 7, 2013, the Company completed the acquisition of ConVerge Diagnostic Services, LLC ("ConVerge"). ConVerge is a leading full-service laboratory providing clinical, cytology and anatomic pathology testing services to patients, physicians and hospitals in New England. The assets acquired at the acquisition date primarily represent goodwill and intangible assets, principally comprised of customer-related intangibles.
Total Consideration, Assets Acquired and Liabilities Assumed
The acquisitions described above were accounted for under the acquisition method of accounting. As such, the assets acquired and liabilities assumed are recorded based on their estimated fair values as of the closing date. The consolidated financial statements include the results of operations of the acquisitions subsequent to the closing of each acquisition. All of the goodwill acquired in connection with the MemorialCare, Solstas, Summit Health, Steward, UMass, ATN, Dignity and ConVerge acquisitions has been allocated to the Company's DIS business. The goodwill associated with the Superior Mobile Medics acquisition has been allocated to the Company's risk assessment services business. The goodwill recorded as part of the acquisitions includes the expected synergies resulting from combining the operations of the acquired business with those of the Company and the value associated with an assembled workforce that has a historical track record of identifying opportunities.
F- 18
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
The following tables summarize the total consideration and the amounts of assets acquired and liabilities assumed for Solstas and Summit Health. The amounts of assets acquired and liabilities assumed for all other acquisitions have not been included in the tables below as these acquisitions were not material to the Company’s consolidated financial statements.
Solstas | Summit Health | ||||||||||
Cash | $ | 572 | $ | 125 | |||||||
Estimated fair value of contingent consideration | — | 22 | |||||||||
Transaction related costs due to sellers | — | 5 | |||||||||
Total consideration | $ | 572 | $ | 152 | |||||||
Solstas | Summit Health | ||||||||||
Fair Value | Weighted Average Useful Life (in years) | Fair Value | Weighted Average Useful Life (in years) | ||||||||
Allocation of purchase price: | |||||||||||
Cash and cash equivalents | $ | 9 | $ | 1 | |||||||
Accounts receivable, net | 48 | 9 | |||||||||
Current deferred income taxes | 7 | — | |||||||||
Other current assets | 12 | 16 | |||||||||
Property, plant and equipment, net | 49 | 6 | |||||||||
Goodwill | 271 | 92 | |||||||||
Intangible assets: | |||||||||||
Customer relationships | 203 | 20 | 33 | 15 | |||||||
Tradename | 7 | 2 | 2 | 1 | |||||||
Software | — | 3 | 4 | ||||||||
Total intangible assets | 210 | 38 | |||||||||
Non-current deferred income taxes | 42 | — | |||||||||
Total assets acquired | 648 | 162 | |||||||||
Current liabilities | 64 | 10 | |||||||||
Non-current deferred income taxes | 4 | — | |||||||||
Other non-current liabilities | 8 | — | |||||||||
Total liabilities assumed | 76 | 10 | |||||||||
Net assets acquired | $ | 572 | $ | 152 | |||||||
F- 19
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Pro Forma Combined Financial Information
The following unaudited pro forma combined financial information reflects the consolidated statement of operations of the Company as if the acquisitions of Solstas and Summit Health had occurred as of January 1, 2013. The unaudited pro forma information includes adjustments primarily related to the amortization of intangible assets acquired, interest expense associated with debt extinguished prior to the acquisitions, and transaction costs related to the Solstas and Summit Health acquisitions. The unaudited pro forma combined financial information does not include the estimated annual synergies expected to be realized upon completion of the integration of Solstas and Summit Health and is not indicative of the results of operations as they would have been had the transaction been effected on the assumed date. Pre-acquisition financial information for all other acquisitions has not been included in the table below as these acquisitions were not material to the Company’s consolidated financial statements.
2014 | 2013 | ||||||
(unaudited) | |||||||
Pro forma net revenues | $ | 7,520 | $ | 7,622 | |||
Pro forma income from continuing operations | $ | 585 | $ | 855 | |||
Earnings per share attributable to Quest Diagnostics’ common stockholders - basic: | |||||||
Pro forma income from continuing operations | $ | 3.79 | $ | 5.40 | |||
Earnings per share attributable to Quest Diagnostics’ common stockholders - diluted: | |||||||
Pro forma income from continuing operations | $ | 3.77 | $ | 5.36 | |||
6. DISPOSITIONS AND HELD FOR SALE
Dispositions
Contribution of Clinical Trials Business
On March 30, 2015, the Company entered into a definitive agreement with Quintiles Transnational Holdings Inc. to form a global clinical trials central laboratory services joint venture, Q2 Solutions. The transaction closed on July 1, 2015. In connection with the transaction, the Company contributed certain assets of its clinical trials testing business ("Clinical Trials") and a net $33 million of cash to the newly formed joint venture in exchange for a non-controlling, 40% ownership interest. The assets of Clinical Trials contributed to the joint venture, principally consisting of property, plant and equipment and goodwill, were classified as assets held for sale in the first quarter of 2015 and were contributed to Q2 Solutions upon closing of the transaction. Subsequent to closing, the Company's ownership interest in the joint venture is being accounted for under the equity method of accounting. At December 31, 2015, the investment in Q2 Solutions had a carrying value of $416 million.
During the third quarter of 2015, the Company recognized a pre-tax gain of $334 million based on the difference between the fair value of the Company's equity interest in the newly formed joint venture over the carrying value of the assets contributed. The fair value of the Company's equity interest was determined using discounted cash flows. In connection with the gain, the Company recorded a deferred tax liability of $145 million. Upon formation, the Company's investment in Q2 Solutions exceeded its equity in the underlying net assets by approximately $219 million. This basis difference is attributable to finite-lived assets, indefinite-lived intangible assets and goodwill of the joint venture. The basis difference associated with the finite-lived assets of $75 million is being amortized over a weighted average useful life of 8 years as a reduction to the carrying value of the investment in equity method investees and corresponding reduction in equity in earnings of equity method investees, net of taxes.
Clinical Trials, prior to July 1, 2015, is included in all other operating segments and has not been classified as discontinued operations. For further details regarding business segment information, see Note 19.
F- 20
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Sale of Royalty Rights
As part of its acquisition of Celera in 2011, the Company gained rights to receive royalties on ibrutinib, an experimental cancer therapy. In July 2013, the Company sold its right to receive royalties related to the commercialization of ibrutinib for $485 million in cash. The Company has accounted for this transaction as a sale of royalty rights and recognized a pre-tax gain of $474 million, net of transaction costs, associated with this sale.
Sale of Enterix
In September 2013, the Company completed the sale of Enterix and recorded a pre-tax loss of approximately $40 million associated with the sale, which is included in other operating expense (income), net. The Enterix business was not reclassified to discontinued operations due to the level of continuing involvement in the Enterix business subsequent to its sale. The continuing involvement relates to a minimum purchase agreement between the acquirer of the Enterix business and the Company.
Sale of HemoCue
In April 2013, the Company completed the sale of HemoCue, which was included in discontinued operations since 2012. For further details regarding the sale of HemoCue, see Note 18.
Held for Sale
Non-core Assets Held for Sale
During the third quarter of 2015, the Company classified certain non-core assets as held for sale. The assets consist of $113 million of goodwill, with the remainder consisting of property, plant and equipment, inventories and intangible assets and are classified as current assets held for sale in the consolidated balance sheet as of December 31, 2015.
The non-core assets held for sale are included in all other operating segments and have not been classified as discontinued operations. For further details regarding business segment information, see Note 19.
F- 21
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
7. FAIR VALUE MEASUREMENTS
The following table provides a summary of the recognized assets and liabilities that are measured at fair value on a recurring basis:
Basis of Fair Value Measurements | |||||||||||||||
Total | Level 1 | Level 2 | Level 3 | ||||||||||||
December 31, 2015 | |||||||||||||||
Assets: | |||||||||||||||
Trading securities | $ | 49 | $ | 49 | $ | — | $ | — | |||||||
Cash surrender value of life insurance policies | 29 | — | 29 | — | |||||||||||
Interest rate swaps | 23 | — | 23 | — | |||||||||||
Available-for-sale equity securities | 6 | 6 | — | — | |||||||||||
Total | $ | 107 | $ | 55 | $ | 52 | $ | — | |||||||
Liabilities: | |||||||||||||||
Deferred compensation liabilities | $ | 85 | $ | — | $ | 85 | $ | — | |||||||
Interest rate swaps | 6 | — | 6 | — | |||||||||||
Contingent consideration | 3 | — | — | 3 | |||||||||||
Total | $ | 94 | $ | — | $ | 91 | $ | 3 | |||||||
December 31, 2014 | |||||||||||||||
Assets: | |||||||||||||||
Trading securities | $ | 49 | $ | 49 | $ | — | $ | — | |||||||
Cash surrender value of life insurance policies | 30 | — | 30 | — | |||||||||||
Interest rate swaps | 17 | — | 17 | — | |||||||||||
Available-for-sale equity securities | 9 | 9 | — | — | |||||||||||
Put option | 1 | — | — | 1 | |||||||||||
Total | $ | 106 | $ | 58 | $ | 47 | $ | 1 | |||||||
Liabilities: | |||||||||||||||
Deferred compensation liabilities | $ | 85 | $ | — | $ | 85 | $ | — | |||||||
Contingent consideration | 17 | — | — | 17 | |||||||||||
Forward starting interest rate swaps | 15 | — | 15 | — | |||||||||||
Interest rate swaps | 13 | — | 13 | — | |||||||||||
Call option | 5 | — | — | 5 | |||||||||||
Total | $ | 135 | $ | — | $ | 113 | $ | 22 | |||||||
The Company offers certain employees the opportunity to participate in non-qualified supplemental deferred compensation plans. A participant's deferrals, together with Company matching credits, are invested in a variety of participant-directed stock and bond mutual funds that are classified as trading securities. Changes in the fair value of these securities are measured using quoted prices in active markets based on the market price per unit multiplied by the number of units held exclusive of any transaction costs. A corresponding adjustment for changes in fair value of the trading securities is also reflected in the changes in fair value of the deferred compensation obligation. The deferred compensation liabilities are classified within Level 2 because their inputs are derived principally from observable market data by correlation to the trading securities.
The Company offers certain employees the opportunity to participate in a non-qualified deferred compensation program. A participant's deferrals, together with Company matching credits, are “invested” at the direction of the employee in a hypothetical portfolio of investments which are tracked by an administrator. The Company purchases life insurance policies, with the Company named as beneficiary of the policies, for the purpose of funding the program's liability. Changes in the cash
F- 22
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
surrender value of the life insurance policies are based upon earnings and changes in the value of the underlying investments. Changes in the fair value of the deferred compensation obligation are derived using quoted prices in active markets based on the market price per unit multiplied by the number of units. The cash surrender value and the deferred compensation obligations are classified within Level 2 because their inputs are derived principally from observable market data by correlation to the hypothetical investments.
The fair value measurements of the Company's interest rate swaps and forward starting swaps are model-derived valuations as of a given date in which all significant inputs are observable in active markets including certain financial information and certain assumptions regarding past, present and future market conditions.
Investment in available-for-sale equity securities represents an investment in registered shares of a publicly-held company listed on the Euronext Paris exchange. The Company's investment in available-for-sale equity securities is classified within Level 1 of the fair value hierarchy because the fair value is obtained from quoted prices in an active market.
In connection with the acquisition of certain business assets of UMass (see Note 5), the Company granted to UMass a call option and UMass granted to the Company a put option for UMass to acquire an 18.90% equity interest in a newly formed entity. The put and call options were derivative instruments whose fair values were measured using a combination of discounted cash flows and the Black-Scholes-Merton option pricing model. On July 1, 2015, UMass exercised its call option, acquiring an 18.90% noncontrolling interest in a subsidiary of the Company that performs diagnostic information services in a defined territory within the state of Massachusetts. In connection with the transaction, the Company paid the $50 million deferred consideration associated with the January 2, 2013 acquisition of the Massachusetts-based clinical outreach and anatomic pathology businesses from UMass and received $50 million associated with the call option exercise price. The put option expired unexercised.
In April 2014, the Company completed the acquisitions of Summit Health and Steward (see Note 5). In connection with these acquisitions the Company initially recorded an aggregate contingent consideration liability of $26 million. The contingent consideration liability was classified within Level 3 measured at fair value using a probability weighted and discounted cash flow method. These measurements are based on externally obtained inputs and management's probability assessments of the occurrence of triggering events, appropriately discounted considering the uncertainties associated with the obligations, as well as the likelihood of achieving financial targets. The initial probability estimate of the occurrence of such triggering events associated with the amounts the Company could be obligated to pay in future periods for both Summit Health and Steward was between 5% and 95%. The probability-weighted cash flows were then discounted using a discount rate of 1.5% to 2.8%. The estimated fair value of the contingent consideration associated with Summit Health was reduced to $13 million in the fourth quarter of 2014 and $0 in the second quarter of 2015. These reductions were a result of updated revenue forecasts and actual results for 2015 compared to the earn-out target included in the contingent consideration arrangement. As a result, other operating expense (income), net for the years ended December 31, 2015 and 2014 include gains of $13 million and $9 million, respectively. The remaining contingent consideration associated with Steward is projected to be paid out in three equal annual installments, with a maximum payout of $4 million.
The following table provides a reconciliation of the beginning and ending balances of assets using significant unobservable inputs (Level 3):
Put Option Derivative Asset | |||
Balance, December 31, 2013 | $ | 4 | |
Total gains (losses) - realized/ unrealized: | |||
Included in earnings | (3 | ) | |
Balance, December 31, 2014 | 1 | ||
Total gains (losses) - realized/ unrealized: | |||
Included in earnings | (1 | ) | |
Balance, December 31, 2015 | $ | — | |
F- 23
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
The following table provides a reconciliation of the beginning and ending balances of liabilities using significant unobservable inputs (Level 3):
Contingent Consideration | Call Option Derivative Liability | Total | |||||||||
Balance, December 31, 2013 | $ | — | $ | 8 | $ | 8 | |||||
Purchases, additions and issuances | 26 | — | 26 | ||||||||
Total (gains) losses - realized/ unrealized: | |||||||||||
Included in earnings | (9 | ) | (3 | ) | (12 | ) | |||||
Balance, December 31, 2014 | 17 | 5 | 22 | ||||||||
Settlements | (1 | ) | (4 | ) | (5 | ) | |||||
Total (gains) losses - realized/ unrealized: | |||||||||||
Included in earnings | (13 | ) | (1 | ) | (14 | ) | |||||
Balance, December 31, 2015 | $ | 3 | $ | — | $ | 3 | |||||
The unrealized gains and losses associated with the change in fair value of the put option derivative asset and call option derivative liability included in earnings for the years ended December 31, 2015 and 2014 are reported in other (expense) income, net.
The carrying amounts of cash and cash equivalents, accounts receivable, accounts payable and accrued expenses approximate fair value based on the short maturities of these instruments. At December 31, 2015 and 2014, the fair value of the Company's debt was estimated at $3.7 billion and $4.2 billion, respectively. Principally all of the Company's debt is classified within Level 1 of the fair value hierarchy because the fair value of the debt is estimated based on rates currently offered to the Company with identical terms and maturities, using quoted active market prices and yields, taking into account the underlying terms of the debt instruments.
8. TAXES ON INCOME
The Company's pre-tax income from continuing operations before equity in earnings of equity method investees consisted of $1.1 billion, $810 million and $1.3 billion from U.S. operations and $11 million, $13 million and $19 million from foreign operations for the years ended December 31, 2015, 2014 and 2013, respectively.
For the year ended December 31, 2015, the Company recognized $145 million deferred tax expense associated with the financial reporting and tax basis difference resulting from the contribution of the Clinical Trials business to the Q2 Solutions joint venture. This was partially offset by a $58 million deferred tax benefit resulting from the future tax effects of winding down a subsidiary.
For the year ended December 31, 2013, pre-tax income from continuing operations in the U.S., income tax expense and the effective tax rate, including the state and local income tax rate, net of federal benefit, were impacted by the gain on sale of royalty rights. For further details regarding the sale of royalty rights, see Note 6.
The components of income tax expense from continuing operations for 2015, 2014 and 2013 were as follows:
2015 | 2014 | 2013 | |||||||||
Current: | |||||||||||
Federal | $ | 231 | $ | 204 | $ | 417 | |||||
State and local | 27 | 34 | 59 | ||||||||
Foreign | 3 | 3 | 4 | ||||||||
Deferred: | |||||||||||
Federal | 104 | 28 | 27 | ||||||||
State and local | 7 | (6 | ) | (6 | ) | ||||||
Foreign | 1 | (1 | ) | (1 | ) | ||||||
Total | $ | 373 | $ | 262 | $ | 500 | |||||
A reconciliation of the federal statutory rate to the Company's effective tax rate for 2015, 2014 and 2013 was as follows:
2015 | 2014 | 2013 | ||||||
Tax provision at statutory rate | 35.0 | % | 35.0 | % | 35.0 | % | ||
State and local income taxes, net of federal benefit | 2.6 | 3.2 | 2.8 | |||||
Impact of foreign operations | — | (0.2 | ) | (0.3 | ) | |||
Tax credits | (0.5 | ) | (0.8 | ) | (0.4 | ) | ||
Deferred taxes on gains and losses | (2.7 | ) | — | — | ||||
Adjustments to unrecognized tax positions (the net benefit mainly results from favorable resolution of certain tax contingencies) | (0.4 | ) | (5.1 | ) | 1.5 | |||
Non-deductible expenses, primarily meals and entertainment expenses | 0.5 | 0.4 | 0.3 | |||||
Impact of noncontrolling interests | (1.6 | ) | (1.7 | ) | (1.0 | ) | ||
Impact of equity earnings | 0.7 | 1.1 | 0.6 | |||||
Other, net | 0.2 | (0.1 | ) | (0.7 | ) | |||
Effective tax rate | 33.8 | % | 31.8 | % | 37.8 | % | ||
The tax effects of temporary differences that give rise to significant portions of the deferred tax assets (liabilities) at December 31, 2015 and 2014 were as follows:
2015 | 2014 | ||||||
Current deferred tax assets: | |||||||
Accounts receivable reserves | $ | — | $ | 91 | |||
Liabilities not currently deductible | — | 78 | |||||
Total current deferred tax assets | $ | — | $ | 169 | |||
Non-current deferred tax assets (liabilities): | |||||||
Accounts receivable reserves | $ | 95 | $ | — | |||
Liabilities not currently deductible | 202 | 138 | |||||
Stock-based compensation | 49 | 42 | |||||
Capitalized R&D expense | 1 | 3 | |||||
Basis differences in investments, joint ventures and subsidiaries | (90 | ) | — | ||||
Net operating loss carryforwards, net of valuation allowance | 140 | 165 | |||||
Depreciation and amortization | (517 | ) | (519 | ) | |||
Total non-current deferred tax liabilities, net | $ | (120 | ) | $ | (171 | ) | |
F- 24
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
During the fourth quarter of 2015, the Company early adopted a new accounting standard which requires that all deferred income tax assets and liabilities be classified as non-current in the balance sheet (see Note 2). As the Company elected to apply the standard prospectively, prior periods were not retrospectively adjusted. Adoption of this standard resulted in $168 million of deferred taxes associated with current assets and liabilities being classified as non-current deferred tax assets of $26 million and as a reduction of non-current deferred tax liabilities of $142 million on the consolidated balance sheet as of December 31, 2015.
At December 31, 2015 and 2014, non-current deferred tax assets of $37 million and $33 million, respectively, are recorded in other long-term assets in the consolidated balance sheets. At December 31, 2015 and 2014, non-current deferred tax liabilities of $157 million and $204 million, respectively, are included in other long-term liabilities in the consolidated balance sheets.
As of December 31, 2015, the Company had estimated net operating loss carryforwards for federal and state income tax purposes of $268 million and $1.4 billion, respectively, which expire at various dates through 2035. Estimated net operating loss carryforwards for foreign income tax purposes are $48 million at December 31, 2015, some of which can be carried forward indefinitely while others expire at various dates through 2025. As of December 31, 2015, 2014 and 2013, deferred tax assets associated with net operating loss carryforwards of $222 million, $242 million and $140 million, respectively, have each been reduced by valuation allowances of $54 million, $60 million and $34 million, respectively. The increase in the valuation allowance as of December 31, 2014 was a result of a $14 million valuation allowance recorded on net operating loss carryforwards acquired in a business combination, with the remainder primarily associated with additional net operating losses generated during 2014.
The Company has not provided U.S. federal income and foreign tax withholdings on undistributed earnings from certain non-U.S. subsidiaries for which the Company intends to reinvest such earnings indefinitely outside the U.S. Determination of the amount of unrecognized deferred tax liability related to these earnings is not practicable.
Income taxes payable, including those classified in other long-term liabilities in the consolidated balance sheets at December 31, 2015 and 2014, were $50 million and $110 million, respectively. Prepaid income taxes were $41 million and $44 million at December 31, 2015 and 2014, respectively, and were included in prepaid expenses and other current assets in the consolidated balance sheets.
The total amount of unrecognized tax benefits as of and for the years ended December 31, 2015, 2014 and 2013 consisted of the following:
2015 | 2014 | 2013 | |||||||||
Balance, beginning of year | $ | 122 | $ | 168 | $ | 199 | |||||
Additions: | |||||||||||
For tax positions of current year | 5 | 17 | 11 | ||||||||
For tax positions of prior years | 5 | 1 | 12 | ||||||||
Reductions: | |||||||||||
Changes in judgment | (11 | ) | (56 | ) | (23 | ) | |||||
Expirations of statutes of limitations | (3 | ) | (6 | ) | (2 | ) | |||||
Settlements | (27 | ) | (2 | ) | (29 | ) | |||||
Balance, end of year | $ | 91 | $ | 122 | $ | 168 | |||||
The contingent liabilities for tax positions primarily relate to uncertainties associated with the realization of tax benefits derived from the allocation of income and expense among state jurisdictions, the characterization and timing of certain tax deductions associated with business combinations, income and expenses associated with certain intercompany licensing arrangements, certain tax credits and the deductibility of certain settlement payments.
The total amount of unrecognized tax benefits as of December 31, 2015, that, if recognized, would affect the effective income tax rate from continuing operations is $38 million. Based upon the expiration of statutes of limitations, settlements and/or the conclusion of tax examinations, the Company believes it is reasonably possible that the total amount of unrecognized tax benefits may decrease by up to $4 million within the next twelve months.
F- 25
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Accruals for interest expense on contingent tax liabilities are classified in income tax expense in the consolidated statements of operations. Accruals for penalties have historically been immaterial. Interest expense (income) included in income tax expense in each of the years ended December 31, 2015, 2014 and 2013 was approximately $0 million, $(1) million and $3 million respectively. As of December 31, 2015 and 2014, the Company has approximately $9 million and $12 million, respectively, accrued, net of the benefit of a federal and state deduction, for the payment of interest on uncertain tax positions.
The recognition and measurement of certain tax benefits includes estimates and judgment by management and inherently involves subjectivity. Changes in estimates may create volatility in the Company's effective tax rate in future periods and may be due to settlements with various tax authorities (either favorable or unfavorable), the expiration of the statute of limitations on some tax positions and obtaining new information about particular tax positions that may cause management to change its estimates.
In the regular course of business, various federal, state, local and foreign tax authorities conduct examinations of the Company's income tax filings and the Company generally remains subject to examination until the statute of limitations expires for the respective jurisdiction. The Internal Revenue Service (“IRS”) has completed its examinations of the Company's consolidated federal income tax returns up through and including the 2011 tax year; however, the Company plans to pursue all alternatives for settlement, including litigation, for certain tax adjustments related to its 2009 tax year. At this time, the Company does not believe that there will be any material additional payments beyond its recorded contingent liability reserves that may be required as a result of these tax audits. As of December 31, 2015, a summary of the tax years that remain subject to examination, or that are under appeal, for the Company's major jurisdictions are:
United States - federal 2012 - 2015
United States - various states 2006 - 2015
9. SUPPLEMENTAL CASH FLOW & OTHER DATA
Supplemental cash flow data for the years ended December 31, 2015, 2014 and 2013 was as follows:
2015 | 2014 | 2013 | |||||||||
Depreciation expense | $ | 223 | $ | 220 | $ | 204 | |||||
Amortization expense | 81 | 94 | 79 | ||||||||
Depreciation and amortization expense | $ | 304 | $ | 314 | $ | 283 | |||||
Interest paid | $ | 172 | $ | 170 | $ | 167 | |||||
Income taxes paid | 319 | 327 | 568 | ||||||||
Assets acquired under capital leases | 3 | 12 | 13 | ||||||||
Accounts payable associated with capital expenditures | 15 | 26 | 28 | ||||||||
Dividend payable | $ | 55 | $ | 48 | $ | 43 | |||||
Businesses acquired: | |||||||||||
Fair value of assets acquired | $ | 63 | $ | 853 | $ | 280 | |||||
Fair value of liabilities assumed | — | 85 | 16 | ||||||||
Fair value of net assets acquired | 63 | 768 | 264 | ||||||||
Merger consideration paid (payable), net | 4 | (30 | ) | (50 | ) | ||||||
Cash paid for business acquisitions | 67 | 738 | 214 | ||||||||
Less: Cash acquired | — | 10 | 1 | ||||||||
Business acquisitions, net of cash acquired | $ | 67 | $ | 728 | $ | 213 | |||||
F- 26
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Supplemental continuing operations data for the statement of operations for the years ended December 31, 2015, 2014 and 2013 was as follows:
2015 | 2014 | 2013 | |||||||||
Depreciation expense | $ | 223 | $ | 220 | $ | 203 | |||||
Interest expense | (154 | ) | (167 | ) | (162 | ) | |||||
Interest income | 1 | 3 | 3 | ||||||||
Interest expense, net | $ | (153 | ) | $ | (164 | ) | $ | (159 | ) | ||
10. PROPERTY, PLANT AND EQUIPMENT
Property, plant and equipment at December 31, 2015 and 2014 consisted of the following:
2015 | 2014 | ||||||
Land | $ | 28 | $ | 28 | |||
Buildings and improvements | 370 | 367 | |||||
Laboratory equipment, furniture and fixtures | 1,407 | 1,386 | |||||
Leasehold improvements | 535 | 538 | |||||
Computer software developed or obtained for internal use | 756 | 675 | |||||
Construction-in-progress | 136 | 132 | |||||
3,232 | 3,126 | ||||||
Less: Accumulated depreciation and amortization | (2,307 | ) | (2,193 | ) | |||
Total | $ | 925 | $ | 933 | |||
11. GOODWILL AND INTANGIBLE ASSETS
The changes in goodwill for the years ended December 31, 2015 and 2014 were as follows:
2015 | 2014 | ||||||
Balance, beginning of year | $ | 6,032 | $ | 5,649 | |||
Goodwill acquired during the year | 33 | 383 | |||||
Reclassification to assets held for sale | (160 | ) | — | ||||
Balance, end of year | $ | 5,905 | $ | 6,032 | |||
Principally all of the Company’s goodwill as of December 31, 2015 and 2014 was associated with its DIS business.
For the year ended December 31, 2015, goodwill acquired during the period was principally associated with the MemorialCare and Superior Mobile Medics acquisitions (see Note 5), of which $32 million is deductible for tax purposes. The reclassification to assets held for sale was principally associated with Clinical Trials and non-core assets (see Note 6). Acquisitions during the year also resulted in $26 million of intangible assets, principally comprised of customer-related intangibles and tradenames.
For the year ended December 31, 2014, goodwill acquired was principally associated with the Solstas, Summit Health and Steward acquisitions (see Note 5), of which $90 million is deductible for tax purposes. Acquisitions during the year also resulted in $270 million of intangible assets, principally comprised of customer-related intangibles and tradenames.
F- 27
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Intangible assets at December 31, 2015 and 2014 consisted of the following:
Weighted Average Amort-ization Period (Years) | December 31, 2015 | December 31, 2014 | |||||||||||||||||||||||
Cost | Accumulated Amortization | Net | Cost | Accumulated Amortization | Net | ||||||||||||||||||||
Amortizing intangible assets: | |||||||||||||||||||||||||
Customer-related intangibles | 19 | $ | 936 | $ | (296 | ) | $ | 640 | $ | 929 | $ | (259 | ) | $ | 670 | ||||||||||
Non-compete agreements | 6 | 6 | (3 | ) | 3 | 43 | (37 | ) | 6 | ||||||||||||||||
Technology | 16 | 93 | (35 | ) | 58 | 118 | (38 | ) | 80 | ||||||||||||||||
Other | 9 | 106 | (59 | ) | 47 | 152 | (82 | ) | 70 | ||||||||||||||||
Total | 17 | 1,141 | (393 | ) | 748 | 1,242 | (416 | ) | 826 | ||||||||||||||||
Intangible assets not subject to amortization: | |||||||||||||||||||||||||
Tradenames | 235 | — | 235 | 244 | — | 244 | |||||||||||||||||||
Other | 1 | — | 1 | 1 | — | 1 | |||||||||||||||||||
Total intangible assets | $ | 1,377 | $ | (393 | ) | $ | 984 | $ | 1,487 | $ | (416 | ) | $ | 1,071 | |||||||||||
For the year ended December 31, 2015, intangible assets with a net book value of $16 million (original cost of $48 million and accumulated amortization of $32 million), principally associated with Clinical Trials and non-core assets (see Note 6), were reclassified to assets held for sale. During the second quarter of 2015, a fully amortized contractual non-compete agreement (original cost of $37 million and accumulated amortization of $37 million) that expired was written-off. Additionally, the Company recognized impairment charges associated with intangible assets of $16 million associated with Celera Products and winding down a subsidiary, which have been included in other operating expense (income), net for the year ended December 31, 2015.
Amortization expense related to intangible assets was $81 million, $94 million and $79 million for the years ended December 31, 2015, 2014 and 2013, respectively.
The estimated amortization expense related to amortizable intangible assets for each of the five succeeding fiscal years and thereafter as of December 31, 2015 is as follows:
Year Ending December 31, | |||
2016 | $ | 70 | |
2017 | 66 | ||
2018 | 62 | ||
2019 | 62 | ||
2020 | 62 | ||
Thereafter | 426 | ||
Total | $ | 748 | |
F- 28
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
12. ACCOUNTS PAYABLE AND ACCRUED EXPENSES
Accounts payable and accrued expenses at December 31, 2015 and 2014 consisted of the following:
2015 | 2014 | ||||||
Trade accounts payable | $ | 279 | $ | 257 | |||
Accrued wages and benefits (including incentive compensation) | 305 | 364 | |||||
Income taxes payable | 4 | 63 | |||||
Accrued interest | 52 | 67 | |||||
Accrued insurance | 60 | 59 | |||||
Merger consideration payable | 2 | 56 | |||||
Dividend payable | 55 | 48 | |||||
Accrued expenses | 257 | 277 | |||||
Total | $ | 1,014 | $ | 1,191 | |||
13. DEBT
Long-term debt at December 31, 2015 and 2014 consisted of the following:
2015 | 2014 | ||||||
5.45% Senior Notes due November 2015 | $ | — | $ | 500 | |||
3.20% Senior Notes due April 2016 | 150 | 304 | |||||
6.40% Senior Notes due July 2017 | — | 375 | |||||
2.70% Senior Notes due April 2019 | 300 | 300 | |||||
4.75% Senior Notes due January 2020 | 522 | 524 | |||||
2.50% Senior Notes due March 2020 | 299 | — | |||||
4.70% Senior Notes due April 2021 | 554 | 549 | |||||
4.25% Senior Notes due April 2024 | 313 | 311 | |||||
3.50% Senior Notes due March 2025 | 601 | — | |||||
6.95% Senior Notes due July 2037 | 247 | 421 | |||||
5.75% Senior Notes due January 2040 | 368 | 439 | |||||
4.70% Senior Notes due March 2045 | 300 | — | |||||
Other | 22 | 39 | |||||
Debt issuance costs | (25 | ) | (20 | ) | |||
Total long-term debt | 3,651 | 3,742 | |||||
Less: Current portion of long-term debt | 159 | 518 | |||||
Total long-term debt, net of current portion | $ | 3,492 | $ | 3,224 | |||
Secured Receivables Credit Facility
In October 2015, the Company amended and restated the agreement for the $525 million secured receivables credit facility (the “Secured Receivables Credit Facility”) entered into in December 2014, increasing the borrowing capacity under the facility to $600 million. The amended and restated Secured Receivables Credit Facility matures in October 2017. Under the Secured Receivables Credit Facility, the Company can issue letters of credit totaling $100 million (see Note 17). Issued letters of credit reduce the available borrowing capacity under the facility. Interest on the borrowings under the Secured Receivables Credit Facility is based on rates that are intended to approximate commercial paper rates for highly-rated issuers plus a spread. At December 31, 2015 and 2014, the Company's borrowing rate under the Secured Receivables Credit Facility was 0.96% and 0.86%, respectively. Borrowings under the Secured Receivables Credit Facility are collateralized by certain domestic receivables. At both December 31, 2015 and 2014, there were no outstanding borrowings under the Secured Receivables Credit Facility.
F- 29
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Senior Unsecured Revolving Credit Facility
In April 2014, the Company amended and restated the agreement for the $750 million senior unsecured revolving credit facility (the “Credit Facility” or "Senior Unsecured Revolving Credit Facility") entered into in September 2011. The amended and restated Credit Facility matures in April 2019. Under the Credit Facility, the Company can issue letters of credit totaling $150 million (see Note 17). Issued letters of credit reduce the available borrowing capacity under the facility. Interest on the Credit Facility is based on certain published rates plus an applicable margin that will vary over a range from 75 basis points to 163 basis points based on changes in the Company's public debt ratings. At the option of the Company, it may elect to lock into LIBOR-based interest rates for periods up to six months. Interest on any outstanding amounts not covered under LIBOR-based interest rate contracts is based on an alternate base rate, which is calculated by reference to the prime rate, the federal funds rate or an adjusted LIBOR rate. At both December 31, 2015 and 2014, the Company's borrowing rate for LIBOR-based loans under the Credit Facility was LIBOR plus 1.125%. The Credit Facility contains various covenants, including the maintenance of certain financial ratios, which could impact the Company's ability to, among other things, incur additional indebtedness. At both December 31, 2015 and 2014, there were no outstanding borrowings under the Credit Facility.
Senior Notes Offerings
In March 2015, the Company completed a $1.2 billion senior notes offering (the “2015 Senior Notes”) that was sold in three tranches: (a) $300 million aggregate principal amount of 2.50% senior notes due March 2020, issued at a discount of $1 million; (b) $600 million aggregate principal amount of 3.50% senior notes due March 2025; and (c) $300 million aggregate principal amount of 4.70% senior notes due March 2045. The Company incurred $11 million of costs associated with the 2015 Senior Notes, which is included as a reduction to the carrying amount of long-term debt and is being amortized over the term of the related debt.
In March 2014, the Company completed a $600 million senior notes offering (the “2014 Senior Notes”) that was sold in two tranches: (a) $300 million aggregate principal amount of 2.70% senior notes due April 2019; and (b) $300 million aggregate principal amount of 4.25% senior notes due April 2024, issued at a discount of $1 million. The Company incurred $5 million of costs associated with the 2014 Senior Notes, which is included as a reduction to the carrying amount of long-term debt and is being amortized over the term of the related debt.
All of the senior notes are unsecured obligations of the Company and rank equally with the Company's other senior unsecured obligations. None of the Company's senior notes have a sinking fund requirement.
Retirement of Debt
In March 2015, the Company commenced a cash tender offer to purchase up to $250 million aggregate principal amount of its 6.95% Senior Notes due July 2037 and 5.75% Senior Notes due January 2040 using a portion of the proceeds from the 2015 Senior Notes. The Company repurchased $176 million of its 6.95% Senior Notes due July 2037 and $74 million of its 5.75% Senior Notes due January 2040. In April 2015, the Company redeemed all of the 5.45% Senior Notes due November 2015, $150 million of the 3.2% Senior Notes due April 2016 and all of the 6.4% Senior Notes due July 2017 with the remaining proceeds from the 2015 Senior Notes. For the year ended December 31, 2015, the Company recorded a loss on retirement of debt in other (expense) income, net of $144 million, principally comprised of premiums paid in connection with these transactions.
F- 30
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Maturities of Long-Term Debt
As of December 31, 2015, long-term debt matures as follows:
Year Ending December 31, | |||
2016 | $ | 159 | |
2017 | 6 | ||
2018 | 4 | ||
2019 | 302 | ||
2020 | 801 | ||
Thereafter | 2,375 | ||
Total maturities of long-term debt | 3,647 | ||
Unamortized discount | (16 | ) | |
Debt issuance costs | (25 | ) | |
Fair value basis adjustments attributable to hedged debt | 45 | ||
Total long-term debt | 3,651 | ||
Less: Current portion of long-term debt | 159 | ||
Total long-term debt, net of current portion | $ | 3,492 | |
14. FINANCIAL INSTRUMENTS
Interest Rate Derivatives – Cash Flow Hedges
From time to time, the Company has entered into various interest rate lock agreements and forward starting interest rate swap agreements to hedge part of the Company's interest rate exposure associated with the variability in future cash flows attributable to changes in interest rates.
During the fourth quarter of 2013 and first quarter of 2014, the Company entered into various forward starting interest rate swap agreements for an aggregate notional amount of $150 million which were accounted for as cash flow hedges. In connection with the issuance of the 2015 Senior Notes, all of these agreements were settled and the Company paid $17 million. These losses are deferred in stockholders’ equity, net of income taxes, as a component of accumulated other comprehensive loss, and amortized as an adjustment to interest expense, net over the term of the Senior Notes due 2025.
In March 2015, the Company entered into interest rate lock agreements with several financial institutions for a total notional amount of $350 million which were accounted for as cash flow hedges. These agreements were entered into to hedge a portion of the Company’s interest rate exposure associated with variability in future cash flows attributable to changes in the five-year, ten-year and thirty-year treasury rates related to the planned issuance of the 2015 Senior Notes. In connection with the issuance of the 2015 Senior Notes, these agreements were settled and the Company received $3 million. These gains are deferred in stockholders’ equity, net of income taxes, as a component of accumulated other comprehensive loss, and amortized as an adjustment to interest expense, net over the term of the respective senior notes.
The total net loss, net of taxes, recognized in accumulated other comprehensive loss, related to the Company's cash flow hedges as of December 31, 2015 and 2014 was $12 million and $15 million, respectively. The loss recognized on the Company's cash flow hedges for the years ended December 31, 2015, 2014 and 2013, as a result of ineffectiveness, was not material. The net amount of deferred losses on cash flow hedges that is expected to be reclassified from accumulated other comprehensive loss into earnings within the next twelve months is $3 million.
F- 31
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Interest Rate Derivatives – Fair Value Hedges
The Company maintains various fixed-to-variable interest rate swaps to convert a portion of the Company's long-term debt into variable interest rate debt. A summary of the notional amounts of interest rate derivatives – fair value hedges as of December 31, 2015 and 2014 is as follows:
Floating Rate | Notional Amount | |||||||||
Debt Instrument | Paid by the Company | 2015 | 2014 | |||||||
3.20% Senior Notes due April 2016 | Six-month LIBOR plus a 2.3% spread | $ | — | $ | 200 | |||||
4.75% Senior Notes due January 2020 | One-month LIBOR plus a 3.6% spread | 350 | 350 | |||||||
4.70% Senior Notes due April 2021 | One-month LIBOR plus a 2.45% to 3.39% spread | 400 | 400 | |||||||
4.25% Senior Notes due April 2024 | One-month LIBOR plus a 1.54% to 1.59% spread | 250 | 250 | |||||||
3.50% Senior Notes due March 2025 | One-month LIBOR plus a 1.44% spread | 200 | — | |||||||
$ | 1,200 | $ | 1,200 | |||||||
In April 2015, the Company terminated the outstanding interest rate swaps associated with the Senior Notes due 2016. The value of these interest rate swaps at the date of termination was not material. The net amount of losses on fair value hedges related to the Senior Notes due 2016 reclassified into earnings upon redemption for the year ended December 31, 2015 was not material.
In prior years, the Company entered into various fixed-to-variable interest rate swap agreements that were accounted for as fair value hedges of a portion of the Senior Notes due 2016 and a portion of the Senior Notes due 2020. In July 2012, the Company monetized the value of these interest rate swap assets by terminating the hedging instruments. The asset value, including accrued interest through the date of termination, was $72 million and the amount to be amortized as a reduction of interest expense over the remaining terms of the hedged debt instruments was $65 million, of which the remaining unamortized balance as of December 31, 2015 was $28 million.
Since inception, the fair value hedges have been effective or highly effective; therefore, there is no impact on earnings for the years ended December 31, 2015, 2014 and 2013 as a result of hedge ineffectiveness.
Interest Rate Derivatives - Economic Hedges
In March 2015, in connection with the retirement of debt (see Note 13), the Company entered into reverse interest rate lock agreements with several financial institutions which were not designated for hedge accounting. The Company entered into these agreements to hedge the variability in cash flows associated with $280 million of the $1.3 billion principal amount of debt that was retired in the first and second quarters of 2015. These agreements were settled during the first and second quarters of 2015 which resulted in a gain of $3 million which was recognized in other (expense) income, net.
F- 32
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
A summary of the fair values of derivative instruments in the consolidated balance sheets is stated in the table below:
December 31, 2015 | December 31, 2014 | ||||||||||
Balance Sheet Classification | Fair Value | Balance Sheet Classification | Fair Value | ||||||||
Derivatives Designated as Hedging Instruments | |||||||||||
Asset Derivatives: | |||||||||||
Interest rate swaps | Other assets | $ | 23 | Other assets | $ | 17 | |||||
Liability Derivatives: | |||||||||||
Interest rate swaps | Other liabilities | 6 | Other liabilities | 13 | |||||||
Forward starting interest rate swaps | — | Other liabilities | 15 | ||||||||
Total Liability Derivatives | 6 | 28 | |||||||||
Derivatives Not Designated as Hedging Instruments | |||||||||||
Asset Derivatives: | |||||||||||
Put option | — | Prepaid expenses and other current assets | 1 | ||||||||
Liability Derivatives: | |||||||||||
Call option | — | Accounts payable and accrued expenses | 5 | ||||||||
Total Net Derivatives Assets (Liabilities) | $ | 17 | $ | (15 | ) | ||||||
15. STOCKHOLDERS’ EQUITY AND REDEEMABLE NONCONTROLLING INTEREST
Stockholders' Equity
Series Preferred Stock
Quest Diagnostics is authorized to issue up to 10 million shares of Series Preferred Stock, par value $1.00 per share. The Company's Board of Directors has the authority to issue such shares without stockholder approval and to determine the designations, preferences, rights and restrictions of such shares. No shares are currently outstanding.
Common Stock
On May 4, 2006, the Company's Restated Certificate of Incorporation was amended to increase the number of authorized shares of common stock, par value $0.01 per share, from 300 million shares to 600 million shares.
Changes in Accumulated Other Comprehensive Income (Loss) by Component
The market value adjustments represent unrealized holding gains (losses) on available-for-sale securities, net of taxes. The net deferred loss on cash flow hedges represents deferred losses, net of taxes on the Company’s interest rate related derivative financial instruments designated as cash flow hedges, net of amounts reclassified to interest expense (see Note 14). For the years ended December 31, 2015, 2014 and 2013, the tax effects related to the market valuation adjustments, deferred losses and other were not material. Foreign currency translation adjustments are not adjusted for income taxes since they relate to indefinite investments in non-U.S. subsidiaries.
F- 33
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
The changes in accumulated other comprehensive income (loss) by component for 2015, 2014 and 2013 were as follows:
Foreign Currency Translation Adjustment | Market Value Adjustment | Net Deferred Loss on Cash Flow Hedges | Other | Accumulated Other Comprehensive Income (Loss) | |||||||||||||||
Balance, December 31, 2012 | $ | 25 | $ | 1 | $ | (7 | ) | $ | (5 | ) | $ | 14 | |||||||
Other comprehensive income (loss) before reclassifications | 2 | (1 | ) | 1 | 1 | 3 | |||||||||||||
Amounts reclassified from accumulated other comprehensive income (loss) | (29 | ) | — | 1 | 3 | (25 | ) | ||||||||||||
Net current period other comprehensive income (loss) | (27 | ) | (1 | ) | 2 | 4 | (22 | ) | |||||||||||
Balance, December 31, 2013 | (2 | ) | — | (5 | ) | (1 | ) | (8 | ) | ||||||||||
Other comprehensive income (loss) before reclassifications | (7 | ) | (1 | ) | (11 | ) | (1 | ) | (20 | ) | |||||||||
Amounts reclassified from accumulated other comprehensive income (loss) | — | — | 1 | — | 1 | ||||||||||||||
Net current period other comprehensive income (loss) | (7 | ) | (1 | ) | (10 | ) | (1 | ) | (19 | ) | |||||||||
Balance, December 31, 2014 | (9 | ) | (1 | ) | (15 | ) | (2 | ) | (27 | ) | |||||||||
Other comprehensive income (loss) before reclassifications | (15 | ) | — | — | 1 | (14 | ) | ||||||||||||
Amounts reclassified from accumulated other comprehensive income (loss) | — | — | 3 | — | 3 | ||||||||||||||
Net current period other comprehensive income (loss) | (15 | ) | — | 3 | 1 | (11 | ) | ||||||||||||
Balance, December 31, 2015 | $ | (24 | ) | $ | (1 | ) | $ | (12 | ) | $ | (1 | ) | $ | (38 | ) | ||||
For the years ended December 31, 2015, 2014 and 2013, the gross deferred losses on cash flow hedges were reclassified from accumulated other comprehensive loss to interest expense, net.
For the year ended December 31, 2013, the gross foreign currency adjustment was reclassified from accumulated other comprehensive loss to income from discontinued operations, net of taxes due to the completed sale of HemoCue.
Dividends
During each of the quarters of 2015, the Company's Board of Directors declared a quarterly cash dividend of $0.38 per common share. During each of the quarters of 2014, the Company's Board of Directors declared a quarterly cash dividend of $0.33 per common share. During each of the quarters in 2013, the Company's Board of Directors declared a quarterly cash dividend of $0.30 per common share.
Share Repurchase Plan
In December 2015, the Company's Board of Directors authorized the Company to repurchase an additional $500 million of the Company's common stock. In August 2013, the Company’s Board of Directors authorized the Company to repurchase an additional $1 billion of the Company’s common stock.
At December 31, 2015, $972 million remained available under the Company’s share repurchase authorization. The share repurchase authorization has no set expiration or termination date.
F- 34
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Share Repurchases
For the year ended December 31, 2015, the Company repurchased 3.2 million shares of its common stock for $224 million.
For the year ended December 31, 2014, the Company repurchased 2.2 million shares of its common stock for $132 million.
On April 19, 2013 and September 4, 2013, the Company entered in accelerated share repurchase agreements ("ASR") with financial institutions to repurchase $450 million and $350 million, respectively, of the Company’s common stock as part of the Company’s Common Stock repurchase program. Each ASR was structured as a combination of two transactions: (1) a treasury stock repurchase and (2) a forward contract which permitted the Company to purchase shares immediately with the final purchase price of those shares determined by the volume weighted average price of the Company's common stock during the purchase period, less a fixed discount. Pursuant to these ASRs, the Company received 7.6 million shares of common stock at a final price of $59.46 per share and 5.8 million shares of common stock at a final price of $60.73 per share, respectively. In addition to the ASRs, the Company repurchased shares of its common stock on the open market in 2013. For the year ended December 31, 2013, the Company also repurchased 4.1 million shares of its common stock for $237 million on the open market.
For the years ended December 31, 2015, 2014 and 2013 the Company reissued 1 million shares, 2 million shares and 3 million shares, respectively, for employee benefit plans.
Redeemable Noncontrolling Interest
On July 1, 2015, UMass exercised its call option, acquiring an 18.9% noncontrolling interest in a subsidiary of the Company that performs diagnostic information services in a defined territory within the state of Massachusetts. In connection with the transaction, the Company received consideration of $68 million, including $50 million associated with the call option exercise price. Under the terms of the transaction, UMass has the right to require the Company to purchase all of its interest in the subsidiary at fair value commencing July 1, 2020. Since the redemption of the noncontrolling interest is outside of the Company's control, it has been presented outside of stockholders' equity at the greater of its carrying amount or its fair value. The Company will record changes in the fair value of the noncontrolling interest immediately as they occur. At December 31, 2015, the redeemable noncontrolling interest was $70 million and was presented at its fair value.
16. STOCK OWNERSHIP AND COMPENSATION PLANS
Employee and Non-employee Directors Stock Ownership Programs
In 2005, the Company established the ELTIP to replace the Company's prior plan. The ELTIP provides for three types of awards: (a) stock options, (b) stock appreciation rights and (c) stock awards. The ELTIP provides for the grant to eligible employees of either non-qualified or incentive stock options, or both, to purchase shares of Company common stock at an exercise price no less than the fair market value of the Company's common stock on the date of grant. Grants of stock appreciation rights allow eligible employees to receive a payment based on the appreciation of Company common stock in cash, shares of Company common stock or a combination thereof. The stock appreciation rights are granted at an exercise price no less than the fair market value of the Company's common stock on the date of grant. Stock options and stock appreciation rights granted under the ELTIP expire on the date designated by the Board of Directors but in no event more than ten years from date of grant. No stock appreciation rights have been granted under the ELTIP. The stock options and shares are subject to forfeiture if employment terminates prior to the end of the vesting period prescribed by the Board of Directors. For all award types, the vesting period is generally over three years from the date of grant. For performance share unit awards, the actual amount of shares earned is based on the achievement of the performance goals specified in the awards. The maximum number of shares of Company common stock that may be optioned or granted under the ELTIP is approximately 71 million shares.
In 2005, the Company established the DLTIP, to replace the Company's prior plan. The DLTIP provides for the grant to non-employee directors of non-qualified stock options to purchase shares of Company common stock at an exercise price no less than the fair market value of the Company's common stock on the date of grant. The DLTIP also permits awards of restricted stock and restricted stock units to non-employee directors. Stock options granted under the DLTIP expire on the date
F- 35
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
designated by the Board of Directors but in no event more than ten years from date of grant, and generally become exercisable in three equal annual installments beginning on the first anniversary date of the grant of the option regardless of whether the optionee remains a director of the Company. The maximum number of shares that may be issued under the DLTIP is 2.4 million shares. For the years ended December 31, 2015, 2014 and 2013, grants under the DLTIP totaled 31 thousand shares, 32 thousand shares and 75 thousand shares, respectively.
The Company's practice has been to issue shares related to its stock-based compensation program from shares of its common stock held in treasury or by issuing new shares of its common stock. See Note 15 for further information regarding the Company's share repurchase program.
Beginning in 2015, the Company changed its method for estimating the fair value of its stock option awards from a lattice-based option-valuation method to a Black-Scholes option-valuation model which has been applied prospectively to stock option awards. The change did not have a significant effect on the stock-based compensation expense reported in the consolidated statements of operations for the year ended December 31, 2015, because stock option awards are granted based on a prescribed dollar value.
The expected volatility under the Black-Scholes option-valuation model was based on historical volatilities of the Company's common stock. The dividend yield was based on the approved annual dividend rate in effect and current market price of the underlying common stock at the time of grant. The risk-free interest rate was based on the U.S. Treasury yield curve in effect at the time of grant for bonds with maturities consistent with the expected holding period of the related award. The expected holding period was estimated using the historical exercise behavior of employees.
The weighted average assumptions used in valuing stock options granted in the periods presented were:
2015 | 2014 | 2013 | |||
Fair value at grant date | $11.57 | $10.99 | $12.64 | ||
Expected volatility | 21.0% | 25.1% | 25.8% | ||
Dividend yield | 2.1% | 2.1% | 1.4% | ||
Risk-free interest rate | 1.7% | 1.6% - 2.0% | 1.1% - 1.3% | ||
Expected holding period, in years | 5.3 | 5.5 - 6.6 | 5.5 - 6.7 | ||
The fair value of restricted stock awards, restricted stock units and performance share units is the average market price of the Company's common stock at the date of grant.
The following summarizes the activity relative to stock option awards for 2015:
Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term (in years) | Aggregate Intrinsic Value | |||||||||
Options outstanding, beginning of year | 6.6 | $ | 54.46 | |||||||||
Options granted | 2.5 | 71.14 | ||||||||||
Options exercised | (1.1 | ) | 53.79 | |||||||||
Options forfeited and canceled | (0.3 | ) | 61.99 | |||||||||
Options outstanding, end of year | 7.7 | $ | 59.65 | 7.3 | $ | 89 | ||||||
Exercisable, end of year | 3.5 | $ | 55.37 | 5.8 | $ | 56 | ||||||
Vested and expected to vest, end of year | 7.5 | $ | 59.43 | 7.3 | $ | 88 | ||||||
The aggregate intrinsic value in the table above represents the total pre-tax intrinsic value (the difference between the Company's closing common stock price on the last trading day of 2015 and the exercise price, multiplied by the number of in-the-money options) that would have been received by the option holders had all option holders exercised their options on
F- 36
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
December 31, 2015. This amount changes based on the fair market value of the Company's common stock. Total intrinsic value of options exercised in 2015, 2014 and 2013 was $21 million, $13 million and $32 million, respectively.
As of December 31, 2015, there was $15 million of unrecognized stock-based compensation cost related to nonvested stock options which is expected to be recognized over a weighted average period of 1.9 years.
The following summarizes the activity relative to stock awards, including restricted stock awards, restricted stock units and performance share units, for 2015, 2014 and 2013:
2015 | 2014 | 2013 | ||||||||||||||||||
Shares | Weighted Average Grant Date Fair Value | Shares | Weighted Average Grant Date Fair Value | Shares | Weighted Average Grant Date Fair Value | |||||||||||||||
Shares outstanding, beginning of year | 1.9 | $ | 55.50 | 1.9 | $ | 57.08 | 2.0 | $ | 56.77 | |||||||||||
Shares granted | 0.6 | 71.17 | 0.7 | 52.72 | 0.8 | 56.79 | ||||||||||||||
Shares vested | (0.3 | ) | 55.74 | (0.3 | ) | 57.14 | (0.5 | ) | 56.25 | |||||||||||
Shares forfeited and canceled | (0.5 | ) | 58.18 | (0.4 | ) | 56.44 | (0.4 | ) | 55.91 | |||||||||||
Shares outstanding, end of year | 1.7 | $ | 59.92 | 1.9 | $ | 55.50 | 1.9 | $ | 57.08 | |||||||||||
As of December 31, 2015, there was $29 million of unrecognized stock-based compensation cost related to nonvested stock awards, which is expected to be recognized over a weighted average period of 1.9 years. Total fair value of shares vested was $20 million, $17 million and $28 million for the years ended December 31, 2015, 2014 and 2013, respectively. The amount of unrecognized stock-based compensation cost is subject to change based on changes, if any, to management's best estimates of the achievement of the performance goals specified in such awards and the resulting number of shares that will be earned at the end of the performance periods.
For the years ended December 31, 2015, 2014 and 2013, stock-based compensation expense totaled $52 million, $51 million and $28 million, respectively. Income tax benefits related to stock-based compensation expense totaled $20 million, $20 million and $11 million for the years ended December 31, 2015, 2014 and 2013, respectively.
Employee Stock Purchase Plan
Under the Company's Employee Stock Purchase Plan (“ESPP”), substantially all employees can elect to have up to 10% of their annual wages withheld to purchase Quest Diagnostics common stock. The purchase price of the stock is 85% of the market price of the Company's common stock on the last business day of each calendar month. Under the ESPP, the maximum number of shares of Quest Diagnostics common stock which may be purchased by eligible employees is 5 million. Approximately 349 thousand, 392 thousand and 404 thousand shares of common stock were purchased by eligible employees in 2015, 2014 and 2013, respectively.
Defined Contribution Plans
The Company maintains qualified defined contribution plans covering substantially all of its employees. The maximum Company matching contribution is 5% of eligible employee compensation. The Company's expense for contributions to its defined contribution plans aggregated $77 million, $73 million and $71 million for 2015, 2014 and 2013, respectively.
F- 37
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Supplemental Deferred Compensation Plans
The Company has a supplemental deferred compensation plan that is an unfunded, non-qualified plan that provides for certain management and highly compensated employees to defer up to 50% of their salary in excess of their defined contribution plan limits and for certain eligible employees, up to 95% of their variable incentive compensation. The maximum Company matching contribution is 5% of eligible employee compensation. The compensation deferred under this plan, together with Company matching amounts, are credited with earnings or losses measured by the mirrored rate of return on investments elected by plan participants. Each plan participant is fully vested in all deferred compensation, Company match and earnings credited to their account. The amounts accrued under the Company's deferred compensation plans were $49 million at both December 31, 2015 and 2014. Although the Company is currently contributing all participant deferrals and matching amounts to trusts, the funds in these trusts, totaling $49 million at both December 31, 2015 and 2014, are general assets of the Company and are subject to any claims of the Company's creditors.
The Company also offers certain employees the opportunity to participate in a non-qualified deferred compensation program. Eligible participants are allowed to defer up to $20 thousand of eligible compensation per year. The Company matches employee contributions equal to 25%, up to a maximum of $5 thousand per plan year. A participant's deferrals, together with Company matching credits, are “invested” at the direction of the employee in a hypothetical portfolio of investments which are tracked by an administrator. Each participant is fully vested in their deferred compensation and vests in Company matching contributions over a four-year period at 25% per year. The amounts accrued under this plan were $36 million at both December 31, 2015 and 2014. The Company purchases life insurance policies, with the Company named as beneficiary of the policies, for the purpose of funding the program's liability. The cash surrender value of such life insurance policies was $29 million and $30 million at December 31, 2015 and 2014, respectively.
For the years ended December 31, 2015, 2014 and 2013, the Company's expense for matching contributions to these plans were not material.
17. COMMITMENTS AND CONTINGENCIES
Letters of Credit and Contractual Obligations
The Company can issue letters of credit under its Secured Receivables Credit Facility and Senior Unsecured Revolving Credit Facility (see Note 13). In support of its risk management program, to ensure the Company’s performance or payment to third parties, $67 million in letters of credit, principally issued under the Secured Receivables Credit Facility, were outstanding at December 31, 2015. The letters of credit primarily represent collateral for current and future automobile liability and workers’ compensation loss payments.
Minimum rental commitments under noncancelable operating leases, primarily real estate, in effect at December 31, 2015 are as follows:
Year Ending December 31, | |||
2016 | $ | 182 | |
2017 | 138 | ||
2018 | 94 | ||
2019 | 67 | ||
2020 | 44 | ||
2021 and thereafter | 152 | ||
Minimum lease payments | $ | 677 | |
Operating lease rental expense for 2015, 2014 and 2013 totaled $224 million, $242 million and $223 million, respectively. Rent expense associated with operating leases that include scheduled rent increases and tenant incentives, such as rent holidays and improvement allowances, is recorded on a straight-line basis over the term of the lease.
The Company has certain noncancelable commitments to purchase products or services from various suppliers, mainly for consulting and other service agreements, and standing orders to purchase reagents and other laboratory supplies.
F- 38
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
At December 31, 2015, the approximate total future purchase commitments are $206 million, of which $96 million are expected to be incurred in 2016, $84 million are expected to be incurred in 2017 through 2018 and the balance thereafter.
Contingent Lease Obligations
The Company remains subject to contingent obligations under certain real estate leases that were entered into by certain predecessor companies of a subsidiary prior to the Company's acquisition of the subsidiary. While over the course of many years, the title to the properties and interest in the subject leases have been transferred to third parties and the subject leases have been amended several times by such third parties, the lessors have not formally released the subsidiary predecessor companies from their original obligations under the leases and therefore remain contingently liable in the event of default. The remaining terms of the lease obligations and the Company's corresponding indemnifications range from 8 to 32 years. The lease payments under certain leases are subject to market value adjustments and contingent rental payments and therefore, the total contingent obligations under the leases cannot be precisely determined but are likely to total several hundred million dollars. A claim against the Company would be made only upon the current lessee's default and after a series of claims and corresponding defaults by third parties that precede the Company in the order of liability. The Company also has certain indemnification rights from other parties to recover losses in the event of default on the lease obligations. The Company believes that the likelihood of its performance under these contingent obligations is remote and no liability has been recorded for any potential payments under the contingent lease obligations.
Settlements
In 2010, a purported class action entitled In re Celera Corp. Securities Litigation was filed in the United States District Court for the Northern District of California against Celera Corporation and certain of its directors and current and former officers. An amended complaint filed in October 2010 alleges that from April 2008 through July 22, 2009, the defendants made false and misleading statements regarding Celera's business and financial results with an intent to defraud investors. The complaint was further amended in 2011 to add allegations regarding a financial restatement. Celera and the director and officer defendants have reached an agreement to settle this action; the court has given its final approval of the settlement. The settlement is fully covered by insurance.
Legal Matters
The Company is involved in various legal proceedings. Some of the proceedings against the Company involve claims that could be substantial in amount.
In addition to the matters described below, in the normal course of business, the Company has been named, from time to time, as a defendant in various legal actions, including arbitrations, class actions and other litigation, arising in connection with the Company's activities as a provider of diagnostic testing, information and services. These legal actions may include lawsuits alleging negligence or other similar legal claims. These actions could involve claims for substantial compensatory and/or punitive damages or claims for indeterminate amounts of damages, and could have an adverse impact on the Company's client base and reputation.
The Company is also involved, from time to time, in other reviews, investigations and proceedings by governmental agencies regarding the Company's business, including, among other matters, operational matters, which may result in adverse judgments, settlements, fines, penalties, injunctions or other relief. The number of these reviews, investigations and proceedings has increased in recent years with regard to many firms in the healthcare services industry, including the Company.
In June 2010, the Company received a subpoena from the Florida Attorney General's Office seeking documents relating to the Company's pricing and billing practices as they relate to Florida’s Medicaid program. The Company cooperated with the requests. In November 2013, the State of Florida intervened as a plaintiff in a civil lawsuit, Florida ex rel. Hunter Laboratories LLC v. Quest Diagnostics Incorporated, et al., filed in Florida Circuit Court. The suit, originally filed by a competitor laboratory, alleges that the Company overcharged Florida’s Medicaid program. The Company's motion to dismiss the state's amended complaint was denied.
In April 2015, a qui tam civil lawsuit entitled United States ex rel. Mayes v. Berkeley HeartLab, Inc., et al., filed in the U.S. District Court for the District of South Carolina, was unsealed. The complaint alleges that certain alleged business practices of the defendants violate the False Claims Act, and seeks monetary relief. The United States has intervened as a
F- 39
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
plaintiff as to Berkeley HeartLab, Inc., a subsidiary of the Company and filed a complaint in intervention; the United States did not intervene as a plaintiff as to Quest Diagnostics Incorporated.
The federal or state governments may bring claims based on the Company's current practices, which it believes are lawful. In addition, certain federal and state statutes, including the qui tam provisions of the federal False Claims Act, allow private individuals to bring lawsuits against healthcare companies on behalf of government or private payers. The Company is aware of lawsuits, and from time to time has received subpoenas, related to billing practices based on the qui tam provisions of the Civil False Claims Act or other federal and state statutes, regulations or other laws. The Company understands that there may be other pending qui tam claims brought by former employees or other "whistle blowers" as to which the Company cannot determine the extent of any potential liability.
Management cannot predict the outcome of such matters. Although management does not anticipate that the ultimate outcome of such matters will have a material adverse effect on the Company's financial condition, given the high degree of judgment involved in establishing loss estimates related to these types of matters, the outcome of such matters may be material to the Company's results of operations or cash flows in the period in which the impact of such matters is determined or paid.
These matters are in different stages. Some of these matters are in their early stages. Matters may involve responding to and cooperating with various government investigations and related subpoenas. As of December 31, 2015, the Company does not believe that any material losses related to the legal matters described above are probable. While the Company believes that a reasonable possibility exists that losses may have been incurred related to the legal matters described above for which an accrual has not been recorded, based on the nature and status of these matters, potential losses, if any, cannot be estimated.
Reserves for Legal Matters
Reserves for legal matters, other than those described above, totaled $9 million and $11 million at December 31, 2015 and 2014, respectively.
Reserves for General and Professional Liability Claims
As a general matter, providers of clinical testing services may be subject to lawsuits alleging negligence or other similar legal claims. These suits could involve claims for substantial damages. Any professional liability litigation could also have an adverse impact on the Company's client base and reputation. The Company maintains various liability insurance coverages for, among other things, claims that could result from providing, or failing to provide, clinical testing services, including inaccurate testing results, and other exposures. The Company's insurance coverage limits its maximum exposure on individual claims; however, the Company is essentially self-insured for a significant portion of these claims. Reserves for such matters, including those associated with both asserted and incurred but not reported claims, are established by considering actuarially determined losses based upon the Company's historical and projected loss experience. Such reserves totaled $124 million and $113 million as of December 31, 2015 and 2014, respectively. Management believes that established reserves and present insurance coverage are sufficient to cover currently estimated exposures. Management cannot predict the outcome of any claims made against the Company. Although management does not anticipate that the ultimate outcome of any such proceedings or claims will have a material adverse effect on the Company's financial condition, given the high degree of judgment involved in establishing accruals for loss estimates related to these types of matters, the outcome may be material to the Company's results of operations or cash flows in the period in which the impact of such claims is determined or paid.
18. DISCONTINUED OPERATIONS
During the fourth quarter of 2012, the Company committed to a plan to sell HemoCue. The Company sold HemoCue in April 2013 for $296 million (net of cash divested and transaction costs).
During the third quarter of 2006, the Company completed its wind down of NID and classified the operations of NID as discontinued operations. The Company will continue to report NID as a discontinued operation until uncertain tax benefits associated with NID are resolved.
The results of operations for NID and HemoCue have been classified as discontinued operations for all periods presented.
F- 40
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
Income from discontinued operations, net of taxes for the year ended December 31, 2013 includes a gain of $14 million (including foreign currency translation adjustments, partially offset by income tax expense and transaction costs) associated with the sale of HemoCue. In addition, income from discontinued operations, net of taxes for the year ended December 31, 2013, included discrete tax benefits of $20 million associated with favorable resolution of certain tax contingencies related to our NID business.
Summarized financial information for the discontinued operations is set forth below:
2015 | 2014 | 2013 | |||||||||
Net revenues | $ | — | $ | — | $ | 28 | |||||
Income from discontinued operations before taxes | — | 1 | 25 | ||||||||
Income tax benefit | — | (4 | ) | (10 | ) | ||||||
Income from discontinued operations, net of taxes | $ | — | $ | 5 | $ | 35 | |||||
The remaining balance sheet information related to NID was not material at December 31, 2015 and 2014.
19. BUSINESS SEGMENT INFORMATION
The Company's DIS business provides insights through clinical testing and related services to patients, physicians, hospitals, ACOs, IDNs, health plans, employers and others. The Company is the world's leading provider of diagnostic information services, which includes providing clinical testing services such as routine (including drugs-of-abuse) testing, gene-based and esoteric testing, and anatomic pathology services, as well as related services and insights. The DIS business accounted for greater than 90% of net revenues from continuing operations in 2015, 2014 and 2013.
All other operating segments include the Company's DS businesses, which consists of its risk assessment services, clinical trials testing, diagnostic products and healthcare information technology businesses (see Note 6 regarding the contribution of the clinical trials testing business to a newly formed joint venture effective July 1, 2015 and non-core assets held for sale). The Company's DS businesses offer a variety of solutions for life insurers, healthcare providers and others.
During 2015, the Company acquired certain operations of MemorialCare and Superior Mobile Medics (see Note 5). MemorialCare is included in the Company's DIS business. Superior Mobile Medics is included in all other operating segments.
During 2014, the Company acquired Solstas, Summit Health and Steward which are included in the Company's DIS business.
During 2013, the Company acquired certain operations of UMass, ATN, Dignity and ConVerge which are included in the Company's DIS business. In addition, the Company completed the sale of Enterix in the third quarter of 2013, which is included in all other operating segments.
On April 19, 2006, the Company decided to discontinue NID’s operations. The Company completed the sale of HemoCue in the second quarter of 2013. The results of operations for NID and HemoCue have been classified as discontinued operations for all periods presented (see Note 18).
At December 31, 2015, substantially all of the Company’s services are provided within the United States, and substantially all of the Company’s assets are located within the United States.
The following table is a summary of segment information for the years ended December 31, 2015, 2014 and 2013. Segment asset information is not presented since it is not used by the chief operating decision maker at the operating segment level. Operating earnings (loss) of each segment represents net revenues less directly identifiable expenses to arrive at operating income (loss) for the segment. General corporate activities included in the table below are comprised of general management and administrative corporate expenses, amortization and impairment of intangibles assets, other operating income and expenses net of certain general corporate activity costs that are allocated to the DIS and DS businesses, and the third quarter of 2015 pre-tax gain on contribution of business to joint venture (see Note 6), the third quarter of 2013 pre-tax gain on
F- 41
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
the sale of the ibrutinib royalty rights and the pre-tax loss on the sale of Enterix. The accounting policies of the segments are the same as those of the Company as set forth in Note 2.
2015 | 2014 | 2013 | |||||||||
Net revenues: | |||||||||||
DIS business | $ | 6,965 | $ | 6,873 | $ | 6,587 | |||||
All other operating segments | 528 | 562 | 559 | ||||||||
Total net revenues | $ | 7,493 | $ | 7,435 | $ | 7,146 | |||||
Operating earnings (loss): | |||||||||||
DIS business | $ | 1,118 | $ | 1,068 | $ | 1,201 | |||||
All other operating segments | 110 | 94 | 76 | ||||||||
General corporate activities | 171 | (179 | ) | 198 | |||||||
Total operating income | 1,399 | 983 | 1,475 | ||||||||
Non-operating expenses, net | (296 | ) | (160 | ) | (151 | ) | |||||
Income from continuing operations before income taxes and equity in earnings of equity method investees | 1,103 | 823 | 1,324 | ||||||||
Income tax expense | (373 | ) | (262 | ) | (500 | ) | |||||
Equity in earnings of equity method investees, net of taxes | 23 | 26 | 24 | ||||||||
Income from continuing operations | 753 | 587 | 848 | ||||||||
Income from discontinued operations, net of taxes | — | 5 | 35 | ||||||||
Net income | 753 | 592 | 883 | ||||||||
Less: Net income attributable to noncontrolling interests | 44 | 36 | 34 | ||||||||
Net income attributable to Quest Diagnostics | $ | 709 | $ | 556 | $ | 849 | |||||
2015 | 2014 | 2013 | |||||||||
Depreciation and amortization: | |||||||||||
DIS business | $ | 212 | $ | 206 | $ | 189 | |||||
All other operating segments | 10 | 13 | 12 | ||||||||
General corporate | 82 | 95 | 82 | ||||||||
Total depreciation and amortization | $ | 304 | $ | 314 | $ | 283 | |||||
Capital expenditures: | |||||||||||
DIS business | $ | 243 | $ | 283 | $ | 196 | |||||
All other operating segments | 16 | 17 | 26 | ||||||||
General corporate | 4 | 8 | 9 | ||||||||
Total capital expenditures | $ | 263 | $ | 308 | $ | 231 | |||||
20. RELATED PARTIES
The Company's equity method investees primarily consist of its clinical trials central laboratory services joint venture and its diagnostic information services joint ventures, which are accounted for under the equity method of accounting. During the year ended December 31, 2015, the Company recognized net revenues of $30 million associated with diagnostic information services provided to its equity method investees. As of December 31, 2015, there was $5 million of accounts receivable from equity method investees on the consolidated balance sheet related to such services.
During the year ended December 31, 2015, the Company recognized $31 million of income associated with the performance of certain corporate services, including transition services, for its equity method investees, classified within selling, general and administrative expenses. As of December 31, 2015, there was $32 million of other receivables from equity
F- 42
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS – CONTINUED
(in millions unless otherwise indicated)
method investees included in prepaid expenses and other current assets on the consolidated balance sheet related to these service agreements and other transition related items. In addition, accounts payable and accrued expenses on the balance sheet at December 31, 2015 included $9 million due to equity method investees.
21. SUBSEQUENT EVENTS
On January 28, 2016, the Company's Board of Directors authorized an increase in its quarterly dividend from $0.38 per share to $0.40 per share, commencing with the dividend payable in April 2016.
On February 26, 2016, the Company developed high-level estimates of the pre-tax charges expected to be incurred in connection with the course of action for 2016 under the Invigorate program. For further details regarding the Invigorate program, see Note 4.
F- 43
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
Quarterly Operating Results (unaudited)
(in millions, except per share data)
2015 (a) | First Quarter | Second Quarter | Third Quarter | Fourth Quarter | Total Year | ||||||||||||||
(b) | (c) | (d) | (e) | ||||||||||||||||
Net revenues | $ | 1,839 | $ | 1,925 | $ | 1,880 | $ | 1,849 | $ | 7,493 | |||||||||
Gross profit | 676 | 743 | 718 | 699 | 2,836 | ||||||||||||||
Income from continuing operations | 70 | 129 | 354 | 200 | 753 | ||||||||||||||
Income from discontinued operations, net of taxes | — | — | — | — | — | ||||||||||||||
Net income | 70 | 129 | 354 | 200 | 753 | ||||||||||||||
Less: Net income attributable to noncontrolling interests | 9 | 11 | 12 | 12 | 44 | ||||||||||||||
Net income attributable to Quest Diagnostics | $ | 61 | $ | 118 | $ | 342 | $ | 188 | $ | 709 | |||||||||
Amounts attributable to Quest Diagnostics' stockholders: | |||||||||||||||||||
Income from continuing operations | $ | 61 | $ | 118 | $ | 342 | $ | 188 | $ | 709 | |||||||||
Income from discontinued operations, net of taxes | — | — | — | — | — | ||||||||||||||
Net income | $ | 61 | $ | 118 | $ | 342 | $ | 188 | $ | 709 | |||||||||
Earnings per share attributable to Quest Diagnostics' stockholders - basic: | |||||||||||||||||||
Income from continuing operations | $ | 0.42 | $ | 0.82 | $ | 2.37 | $ | 1.31 | $ | 4.92 | |||||||||
Income from discontinued operations | — | — | — | — | — | ||||||||||||||
Net income | $ | 0.42 | $ | 0.82 | $ | 2.37 | $ | 1.31 | $ | 4.92 | |||||||||
Earnings per share attributable to Quest Diagnostics' stockholders - diluted: | |||||||||||||||||||
Income from continuing operations | $ | 0.42 | $ | 0.81 | $ | 2.35 | $ | 1.29 | $ | 4.87 | |||||||||
Income from discontinued operations | — | — | — | — | — | ||||||||||||||
Net income | $ | 0.42 | $ | 0.81 | $ | 2.35 | $ | 1.29 | $ | 4.87 | |||||||||
F- 44
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
Quarterly Operating Results (unaudited)
(in millions, except per share data)
2014 (a) | First Quarter | Second Quarter | Third Quarter | Fourth Quarter | Total Year | ||||||||||||||
(f) | (g) | (h) | (i) | ||||||||||||||||
Net revenues | $ | 1,746 | $ | 1,902 | $ | 1,904 | $ | 1,883 | $ | 7,435 | |||||||||
Gross profit | 645 | 728 | 726 | 699 | 2,798 | ||||||||||||||
Income from continuing operations | 111 | 142 | 139 | 195 | 587 | ||||||||||||||
Income from discontinued operations, net of taxes | — | — | — | 5 | 5 | ||||||||||||||
Net income | 111 | 142 | 139 | 200 | 592 | ||||||||||||||
Less: Net income attributable to noncontrolling interests | 7 | 9 | 10 | 10 | 36 | ||||||||||||||
Net income attributable to Quest Diagnostics | $ | 104 | $ | 133 | $ | 129 | $ | 190 | $ | 556 | |||||||||
Amounts attributable to Quest Diagnostics' stockholders: | |||||||||||||||||||
Income from continuing operations | $ | 104 | $ | 133 | $ | 129 | $ | 185 | $ | 551 | |||||||||
Income from discontinued operations, net of taxes | — | — | — | 5 | 5 | ||||||||||||||
Net income | $ | 104 | $ | 133 | $ | 129 | $ | 190 | $ | 556 | |||||||||
Earnings per share attributable to Quest Diagnostics' stockholders - basic: | |||||||||||||||||||
Income from continuing operations | $ | 0.72 | $ | 0.92 | $ | 0.89 | $ | 1.27 | $ | 3.80 | |||||||||
Income from discontinued operations | — | — | — | 0.03 | 0.03 | ||||||||||||||
Net income | $ | 0.72 | $ | 0.92 | $ | 0.89 | $ | 1.30 | $ | 3.83 | |||||||||
Earnings per share attributable to Quest Diagnostics' stockholders - diluted: | |||||||||||||||||||
Income from continuing operations | $ | 0.71 | $ | 0.92 | $ | 0.88 | $ | 1.26 | $ | 3.78 | |||||||||
Income from discontinued operations | — | — | — | 0.03 | 0.03 | ||||||||||||||
Net income | $ | 0.71 | $ | 0.92 | $ | 0.88 | $ | 1.29 | $ | 3.81 | |||||||||
(a) | In April 2013, the Company completed the sale of HemoCue. During the third quarter of 2006, the Company completed its wind down of NID and classified the operations of NID as discontinued operations. Results of operations have been prepared to report the results of HemoCue and NID as discontinued operations for all periods presented (see Note 18). In July 2015, the Company contributed its clinical trials testing business to a newly formed global clinical trials central laboratory services joint venture, Q2 Solutions. Subsequent to closing, the Company's ownership interest in the joint venture is being accounted for under the equity method of accounting. |
(b) | Includes pre-tax charges of $31 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($20 million in cost of services and $11 million in selling, general and administrative expenses); pre-tax charges of $8 million in other operating expense (income), net, representing non-cash asset impairment charges associated with our Celera Products business; pre-tax charges in selling, general and administrative expenses of $2 million, principally representing costs incurred related to legal matters; and pre-tax charges of $79 million on retirement of debt associated with the March 2015 cash tender offer in other (expense) income, net (see Note 13). |
(c) | Includes pre-tax charges of $23 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($11 million in cost of services and $12 million in selling, general and administrative expenses); a pre-tax gain included in other operating expense (income), net of $13 million associated with a decrease in the fair value of the contingent consideration accrual associated with our Summit Health acquisition (see Note 5 and Note 7); pre-tax charges in selling, general and administrative expenses of $5 million, principally representing costs incurred related to legal matters; pre-tax charges of $5 million in other operating expense |
F- 45
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
Quarterly Operating Results (unaudited)
(in millions, except per share data)
(income), net, representing non-cash asset impairment charges; and pre-tax charges of $65 million on retirement of debt associated with the April 2015 redemption in other (expense) income, net (see Note 13).
(d) | Includes a pre-tax gain of $334 million associated with the contribution of the Company's clinical trials testing business to Q2 Solutions (see Note 6); and pre-tax charges of $29 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($20 million in cost of services and $9 million in selling, general and administrative expenses). |
(e) | Includes pre-tax charges of $22 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($12 million in cost of services and $10 million in selling, general and administrative expenses); pre-tax charges of $11 million in other operating expense (income), net, representing non-cash asset impairment charges associated with winding down a subsidiary; and pre-tax charges in selling, general and administrative expenses of $10 million, principally representing costs incurred related to legal matters. Income from continuing operations includes a deferred income tax benefit of $58 million associated with winding down a subsidiary. |
(f) | Includes pre-tax charges of $24 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($12 million in cost of services and $12 million in selling, general and administrative expenses); and pre-tax charges in selling, general and administrative expenses of $4 million, principally representing costs incurred related to legal matters. |
(g) | Includes pre-tax charges of $27 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($11 million in cost of services and $16 million in selling, general and administrative expenses); and pre-tax charges in selling, general and administrative expenses of $7 million, principally representing costs incurred related to legal matters. |
(h) | Includes pre-tax charges of $40 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($14 million in cost of services, $25 million in selling, general and administrative expenses and $1 million in other operating expense (income), net); and pre-tax charges in selling, general and administrative expenses of $8 million, principally representing costs incurred related to legal matters. |
(i) | Includes pre-tax charges of $30 million, primarily associated with workforce reductions and professional fees incurred in connection with further restructuring and integrating the Company ($13 million in cost of services, $16 million in selling, general and administrative expenses and $1 million in other operating expense (income), net); pre-tax charges in selling, general and administrative expenses of $5 million, principally representing costs incurred related to legal matters; and a pre-tax gain included in other operating expense (income), net of $9 million associated with a decrease in the fair value of the contingent consideration accrual associated with the Summit Health acquisition (see Note 5 and Note 7). Income from continuing operations includes a discrete benefit of $44 million associated with the favorable resolution of certain tax contingencies. |
F- 46
QUEST DIAGNOSTICS INCORPORATED AND SUBSIDIARIES
SCHEDULE II - VALUATION ACCOUNTS AND RESERVES
(in millions)
Balance at Beginning of Year | Provision for Doubtful Accounts | Net Deductions and Other | Balance at End of Year | ||||||||||||
Year Ended December 31, 2015 | |||||||||||||||
Doubtful accounts and allowances | $ | 250 | $ | 297 | $ | 293 | (a) | $ | 254 | ||||||
Year Ended December 31, 2014 | |||||||||||||||
Doubtful accounts and allowances | $ | 236 | $ | 296 | $ | 282 | (a) | $ | 250 | ||||||
Year Ended December 31, 2013 | |||||||||||||||
Doubtful accounts and allowances | $ | 236 | $ | 270 | $ | 270 | (a) | $ | 236 | ||||||
(a) | Primarily represents the write-off of accounts receivable, net of recoveries. |
F- 47
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
EXHIBITS TO FORM 10-K
For the fiscal year ended December 31, 2015
Commission File No. 001-12215
QUEST DIAGNOSTICS INCORPORATED
Exhibit Number | Description |
3.1 | Restated Certificate of Incorporation (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: May 20, 2014) and incorporated herein by reference) (Commission File Number 001-12215) |
3.2 | Amended and Restated By-Laws of the Company (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: May 20, 2014) and incorporated herein by reference) (Commission File Number 001-12215) |
4.1 | Form of 6.95% Senior Note due 2037 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: June 19, 2007) and incorporated herein by reference) (Commission file Number 001-12215) |
4.2 | Form of 4.750% Senior Note due 2020 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: November 17, 2009) and incorporated herein by reference) (Commission file Number 001-12215) |
4.3 | Form of 5.750% Senior Note due 2040 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: November 17, 2009) and incorporated herein by reference) (Commission file Number 001-12215) |
4.4 | Form of 3.200% Senior Note due 2016 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 21, 2011) and incorporated herein by reference) (Commission File Number 001-12215) |
4.5 | Form of 4.700% Senior Note due 2021 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 21, 2011) and incorporated herein by reference) (Commission File Number 001-12215) |
4.6 | Form of 2.700% Senior Note due 2019 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 12, 2014) and incorporated herein by reference) (Commission File Number 001-12215) |
4.7 | Form of 4.250% Senior Note due 2024 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 12, 2014) and incorporated herein by reference) (Commission File Number 001-12215) |
4.8 | Form of 2.500% Senior Note due 2020 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 10, 2015) and incorporated herein by reference) (Commission File Number 001-12215) |
4.9 | Form of 3.500% Senior Note due 2025 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 10, 2015) and incorporated herein by reference) (Commission File Number 001-12215) |
4.10 | Form of 4.700% Senior Note due 2045 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 10, 2015) and incorporated herein by reference) (Commission File Number 001-12215) |
4.11 | Indenture dated as of June 27, 2001, among the Company, the Subsidiary Guarantors, and the Trustee (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: June 27, 2001) and incorporated herein by reference) (Commission File Number 001-12215) |
E - 1
4.12 | First Supplemental Indenture, dated as of June 27, 2001, among the Company, the Subsidiary Guarantors, and The Bank of New York (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: June 27, 2001) and incorporated herein by reference) (Commission File Number 001-12215) |
4.13 | Second Supplemental Indenture, dated as of November 26, 2001, among the Company, the Subsidiary Guarantors, and The Bank of New York (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: November 26, 2001) and incorporated herein by reference) (Commission File Number 001-12215) |
4.14 | Third Supplemental Indenture, dated as of April 4, 2002, among the Company, the Additional Subsidiary Guarantors, and The Bank of New York (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: April 1, 2002) and incorporated herein by reference) (Commission File Number 001-12215) |
4.15 | Fourth Supplemental Indenture dated as of March 19, 2003, among Unilab Corporation (f/k/a Quest Diagnostics Newco Incorporated), the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's quarterly report on Form 10-Q for the quarter ended March 31, 2003 and incorporated herein by reference) (Commission File Number 001-12215) |
4.16 | Fifth Supplemental Indenture dated as of April 16, 2004, among Unilab Acquisition Corporation (d/b/a FNA Clinics of America), the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's quarterly report on Form 10-Q for the quarter ended March 31, 2004 and incorporated herein by reference) (Commission File Number 001-12215) |
4.17 | Sixth Supplemental Indenture dated as of October 31, 2005, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: October 31, 2005) and incorporated herein by reference) (Commission File Number 001-12215) |
4.18 | Seventh Supplemental Indenture dated as of November 21, 2005, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: November 21, 2005) and incorporated herein by reference) (Commission File Number 001-12215) |
4.19 | Eighth Supplemental Indenture dated as of July 31, 2006, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: July 31, 2006) and incorporated herein by reference) (Commission File Number 001-12215) |
4.20 | Ninth Supplemental Indenture dated as of September 30, 2006, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: September 30, 2006) and incorporated herein by reference) (Commission File Number 001-12215) |
4.21 | Tenth Supplemental Indenture dated as of June 22, 2007, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: June 19, 2007) and incorporated herein by reference) (Commission File Number 001-12215) |
4.22 | Eleventh Supplemental Indenture dated as of June 22, 2007, among the Company, The Bank of New York, and the Additional Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: June 19, 2007) and incorporated herein by reference) (Commission File Number 001-12215) |
4.23 | Twelfth Supplemental Indenture dated as of June 25, 2007, among the Company, The Bank of New York, and the Additional Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: June 19, 2007) and incorporated herein by reference) (Commission File Number 001-12215) |
4.24 | Thirteenth Supplemental Indenture dated as of November 17, 2009, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: November 17, 2009) and incorporated herein by reference) (Commission File Number 001-12215) |
4.25 | Fourteenth Supplemental Indenture dated as of March 24, 2011, among the Company, The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: March 21, 2011) and incorporated herein by reference) (Commission File Number 001-12215) |
E - 2
4.26 | Fifteenth Supplemental Indenture dated as of November 30, 2011, among the Company, The Bank of New York Mellon Trust Company, N.A., as successor trustee to The Bank of New York, and the Subsidiary Guarantors (filed as an Exhibit to the Company's 2011 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
4.27 | Sixteenth Supplemental Indenture dated as of March 12, 2014, among the Company, The Bank of New York Mellon Trust Company, N.A., (filed as an Exhibit to the Company's current report on Form 8-K and incorporated herein by reference) (Commission File Number 001-12215) |
4.28 | Seventeenth Supplemental Indenture dated as of March 10, 2015, among the Company and The Bank of New York Mellon (filed as an Exhibit to the Company’s current report on Form 8-K (Date of Report: March 5, 2015) and incorporated herein by reference) (Commission File Number 001-12215) |
10.1‡ | Amended and Restated Employee Stock Purchase Plan (filed as an Exhibit to the Company's quarterly report on form 10-Q for the quarter ended June 30, 2015 and incorporated herein by reference) (Commission file number 001-12215) |
10.2‡ | Amended and Restated Quest Diagnostics Incorporated Employee Long-Term Incentive Plan as amended May 15, 2015 (filed as an exhibit to the Company's 2015 quarterly report on Form 10-Q for the quarter ended June 30, 2015 and incorporated herein by reference) (Commission file number 001-12215) |
10.3‡* | Form of Equity Award Agreement dated as of February 23, 2015 |
10.4‡ | Quest Diagnostics Supplemental Deferred Compensation Plan (Post 2004) amended December 22, 2008 (filed as an Exhibit to the Company's 2008 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
10.5‡ | Amendment No. 1 dated November 27, 2012 to Quest Diagnostics Supplemental Deferred Compensation Plan (Post 2004) amended December 22, 2008 (filed as an Exhibit to the Company's 2012 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
10.6‡ | Quest Diagnostics Supplemental Deferred Compensation Plan (Pre-2005) amended and restated November 27, 2012 (filed as an Exhibit to the Company's 2012 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
10.7‡ | Quest Diagnostics Incorporated Senior Management Incentive Plan (filed as Appendix A to the Company's Definitive Proxy Statement dated March 28, 2003 and incorporated herein by reference) (Commission File Number 001-12215) |
10.8‡ | Amended and Restated Quest Diagnostics Incorporated Executive Officer Severance Plan (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: May 20, 2014) and incorporated herein by reference) (Commission File Number 001-12215) |
10.9‡ | AmeriPath Group Holdings, Inc. 2006 Stock Option and Restricted Stock Purchase Plan (filed as an Exhibit to the Company's registration statement on Form S-8 (Registration No. 333-143889 filed with the Commission on June 19, 2007 and incorporated herein by reference) (Commission File Number 333-143889) |
10.10‡ | Amendment dated as of August 17, 2007 to the AmeriPath Group Holdings, Inc. 2006 Stock Option and Restricted Stock Purchase Plan (filed as an Exhibit to the Company's 2007 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
10.11‡* | The Profit Sharing Plan of Quest Diagnostics Incorporated (Amendment and Restatement, effective as of January 1, 2016) |
E - 3
10.12‡* | Quest Diagnostics Incorporated Amended and Restated Deferred Compensation Plan For Directors as amended effective January 1, 2016 |
10.13‡* | Amended and Restated Quest Diagnostics Incorporated Long-Term Incentive Plan for Non-Employee Directors as amended December 3, 2015 |
10.14‡ | Form of Non-Employee Director Equity Award Agreement (filed as an Exhibit to the Company's 2011 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
10.15‡* | Form of Non-Employee Director Equity Award Agreement dated May 15, 2015 |
10.16‡ | Form of Non-Employee Director Elective Option Award Agreement (filed as an Exhibit to the Company's 2011 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
10.17‡ | Employment Agreement between Stephen H. Rusckowski and Quest Diagnostics Incorporated, dated April 3, 2012 (filed as an Exhibit to the Company's current report on Form 8-K (Date of Report: April 9, 2012) and incorporated herein by reference) (Commission File Number 001-12215) |
10.18‡ | Amendment to Employment Agreement between Stephen H. Rusckowski and Quest Diagnostics Incorporated, dated June 11, 2015 (filed as an Exhibit to the Company’s current report on Form 8-K (Date of Report: June 11, 2015) and incorporated herein by reference) (Commission File Number 001-12215) |
10.19‡ | Aircraft Timesharing Agreement dated as of December 17, 2013 between Quest Diagnostics Incorporated and Stephen H. Rusckowski (filed as an Exhibit to the Company’s 2013 annual report on Form 10-K and incorporated herein by reference) (Commission File Number 001-12215) |
11.1 | Statement re: Computation of Earnings Per Common Share (the calculation of per share earnings is in Part II, Item 8, Note 3 to the consolidated financial statements (Earnings Per Share) and is omitted in accordance with Item 601(b)(11) of Regulation S-K) |
21.1* | Subsidiaries of Quest Diagnostics Incorporated |
23.1* | Consent of PricewaterhouseCoopers LLP |
24.1* | Power of Attorney (included on signature page) |
31.1* | Rule 13a-14(a) Certification of Chief Executive Officer |
31.2* | Rule 13a-14(a) Certification of Chief Financial Officer |
32.1** | Section 1350 Certification of Chief Executive Officer |
32.2** | Section 1350 Certification of Chief Financial Officer |
101.INS* | dgx-20151231.xml |
101.SCH* | dgx-20151231.xsd |
101.CAL* | dgx-20151231_cal.xml |
101.DEF* | dgx-20151231_def.xml |
E - 4
101.LAB* | dgx-20151231_lab.xml |
101.PRE* | dgx-20151231_pre.xml |
* | Filed herewith. |
** | Furnished herewith. |
‡ | Management contract or compensatory plan or arrangement required to be filed as an exhibit to this Form 10-K pursuant to Item 15(b) of Form 10-K. |
E - 5
