Attached files
| file | filename |
|---|---|
| 8-K - 8-K - LANDAUER INC | ldr-20150810x8k.htm |
| EX-99.1 - EX-99.1 - LANDAUER INC | ldr-20150810ex9913ed77d.htm |
nvestor Presentation August 2015 Safe Harbor Statement Some of the information shared here (including, in particular, the section titled “Fiscal 2015 Outlook”) constitutes forward-looking statements that are based on assumptions and involve certain risks and uncertainties. These include the following, without limitation: assumptions, risks and uncertainties associated with the Company’s future performance; the Company’s development and introduction of new technologies in general; the ability to protect and utilize the Company’s intellectual property; continued customer acceptance of the InLighttechnology; the adaptability of optically stimulated luminescence (OSL) technology to new platforms and formats; military and other government funding for the purchase of certain of the Company’s equipment and services; the impact on sales and pricing of certain customer group purchasing arrangements; changes in spending or reimbursement for medical products or services; the costs associated with the Company’s research and business development efforts; the usefulness of older technologies and related licenses and intellectual property; the effectiveness of and costs associated with the Company’s IT platform enhancements; the anticipated results of operations of the Company and its subsidiaries or ventures; valuation of the Company’s long-lived assets or business units relative to future cash flows; changes in pricing of services and products; changes in postal and delivery practices; the Company’s business plans; anticipated revenue and cost growth; the ability to integrate the operations of acquired businesses and to realize the expected benefits of acquisitions; the risks associated with conductingbusiness internationally; costs incurred for potential acquisitions or similar transactions; other anticipated financial events; the effects of changing economic and competitive conditions, including instability in capital markets which could impact availability of short and long-term financing; the timing and extent of changes in interest rates; the level of borrowings; foreign exchange rates; government regulations; accreditation requirements; changes in the trading market that affect the costs of obligations under the Company’s benefit plans; and pending accounting pronouncements. These assumptions may not materialize to the extent assumed, and risks and uncertainties may cause actual results to be different from what is anticipated today. These risks and uncertainties also may result in changes to the Company’s businessplans and prospects, and could create the need from time to time to write down the value of assets or otherwise cause the Company to incurunanticipated expenses. Additional information may be obtained by reviewing the information set forth in Item 1A “Risk Factors”and Item 7A “Quantitative and Qualitative Disclosures about Market Risk” and information contained in the Company's Annual Report on Form10-K for the year ended September 30, 2014 and other reports filed by the Company, from time to time, with the Securities and Exchange Commission. The Company does not undertake, and expressly disclaims, any duty to update any forward-looking statement whether as a result ofnew information, future events or changes in the Company’s expectations, except as required by law.
Safe Harbor Statement Some of the information shared here (including, in particular, the section titled “Fiscal 2015 Outlook”) constitutes forward-looking statements that are based on assumptions and involve certain risks and uncertainties. These include the following, without limitation: assumptions, risks and uncertainties associated with the Company’s future performance; the Company’s development and introduction of new technologies in general; the ability to protect and utilize the Company’s intellectual property; continued customer acceptance of the InLighttechnology; the adaptability of optically stimulated luminescence (OSL) technology to new platforms and formats; military and other government funding for the purchase of certain of the Company’s equipment and services; the impact on sales and pricing of certain customer group purchasing arrangements; changes in spending or reimbursement for medical products or services; the costs associated with the Company’s research and business development efforts; the usefulness of older technologies and related licenses and intellectual property; the effectiveness of and costs associated with the Company’s IT platform enhancements; the anticipated results of operations of the Company and its subsidiaries or ventures; valuation of the Company’s long-lived assets or business units relative to future cash flows; changes in pricing of services and products; changes in postal and delivery practices; the Company’s business plans; anticipated revenue and cost growth; the ability to integrate the operations of acquired businesses and to realize the expected benefits of acquisitions; the risks associated with conductingbusiness internationally; costs incurred for potential acquisitions or similar transactions; other anticipated financial events; the effects of changing economic and competitive conditions, including instability in capital markets which could impact availability of short and long-term financing; the timing and extent of changes in interest rates; the level of borrowings; foreign exchange rates; government regulations; accreditation requirements; changes in the trading market that affect the costs of obligations under the Company’s benefit plans; and pending accounting pronouncements. These assumptions may not materialize to the extent assumed, and risks and uncertainties may cause actual results to be different from what is anticipated today. These risks and uncertainties also may result in changes to the Company’s businessplans and prospects, and could create the need from time to time to write down the value of assets or otherwise cause the Company to incurunanticipated expenses. Additional information may be obtained by reviewing the information set forth in Item 1A “Risk Factors”and Item 7A “Quantitative and Qualitative Disclosures about Market Risk” and information contained in the Company's Annual Report on Form10-K for the year ended September 30, 2014 and other reports filed by the Company, from time to time, with the Securities and Exchange Commission. The Company does not undertake, and expressly disclaims, any duty to update any forward-looking statement whether as a result ofnew information, future events or changes in the Company’s expectations, except as required by law.
 Compelling Investment Opportunity Industry leading position, compelling market trends and strong financial profile drives long-term shareholder value •The Joint Commission standards effective in July 2015 in the United States -Impacts 7,100 of the 20,500 U.S. healthcare organizations and programs, including hospitals, imaging clinics and certain ambulatory care centers -Estimated incremental cost of ~$30,000 annually for a hospital to comply with new standards •International standards becoming more restrictive •Next generation Verifii™ platform on target for 2016 launch •Well positioned to meet emerging need for broader solution •Differentiated position creates higher barrier to entry for competition •Strong margins and limited capital requirements deliver compelling free cash flow •Defined capital allocation plan supports investment in growth initiatives and dividend •Industry-leading technology; proprietary Optically Stimulated Luminescence (OSL) crystal manufacturing process •Installed based of over 73,000 customers; 1.8 million individuals served worldwide •90% recurring revenue; 94% annual customer retention •Ability to provide enterprise radiation management or product only model Global Leader in Occupational Radiation Products and Services Increased Regulation Drives Demand Compelling Growth and Expansion Opportunities Strong Financial Profile
Compelling Investment Opportunity Industry leading position, compelling market trends and strong financial profile drives long-term shareholder value •The Joint Commission standards effective in July 2015 in the United States -Impacts 7,100 of the 20,500 U.S. healthcare organizations and programs, including hospitals, imaging clinics and certain ambulatory care centers -Estimated incremental cost of ~$30,000 annually for a hospital to comply with new standards •International standards becoming more restrictive •Next generation Verifii™ platform on target for 2016 launch •Well positioned to meet emerging need for broader solution •Differentiated position creates higher barrier to entry for competition •Strong margins and limited capital requirements deliver compelling free cash flow •Defined capital allocation plan supports investment in growth initiatives and dividend •Industry-leading technology; proprietary Optically Stimulated Luminescence (OSL) crystal manufacturing process •Installed based of over 73,000 customers; 1.8 million individuals served worldwide •90% recurring revenue; 94% annual customer retention •Ability to provide enterprise radiation management or product only model Global Leader in Occupational Radiation Products and Services Increased Regulation Drives Demand Compelling Growth and Expansion Opportunities Strong Financial Profile
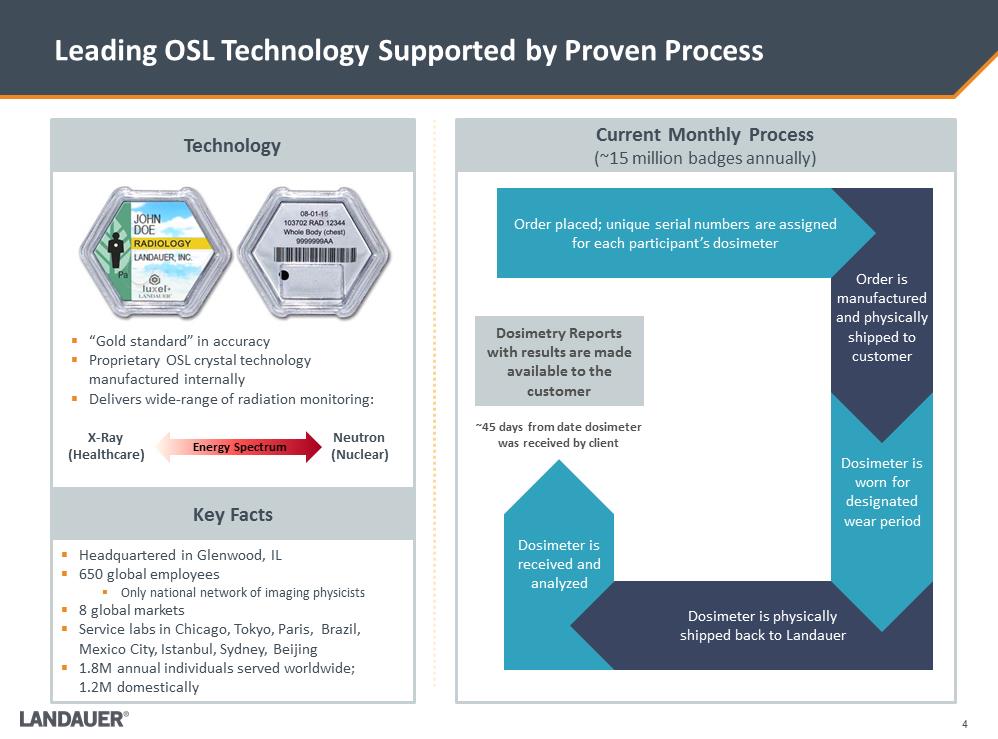 Dosimetry Reports with results are made available to the customer Leading OSL Technology Supported by Proven Process .“Gold standard” in accuracy .Proprietary OSL crystal technology manufactured internally .Delivers wide-range of radiation monitoring: .Headquartered in Glenwood, IL .650 global employees .Only national network of imaging physicists .8 global markets .Service labs in Chicago, Tokyo, Paris, Brazil, Mexico City, Istanbul, Sydney, Beijing .1.8M annual individuals served worldwide; 1.2M domestically Dosimeter is received and analyzed Dosimeter is physically shipped back to Landauer Dosimeter is worn for designated wear period Order is manufactured and physically shipped to customer Order placed; unique serial numbers are assigned for each participant’s dosimeter ~45 days from date dosimeter was received by client Neutron (Nuclear) X-Ray (Healthcare) Current Monthly Process (~15 million badges annually) Technology Key Facts Energy Spectrum
Dosimetry Reports with results are made available to the customer Leading OSL Technology Supported by Proven Process .“Gold standard” in accuracy .Proprietary OSL crystal technology manufactured internally .Delivers wide-range of radiation monitoring: .Headquartered in Glenwood, IL .650 global employees .Only national network of imaging physicists .8 global markets .Service labs in Chicago, Tokyo, Paris, Brazil, Mexico City, Istanbul, Sydney, Beijing .1.8M annual individuals served worldwide; 1.2M domestically Dosimeter is received and analyzed Dosimeter is physically shipped back to Landauer Dosimeter is worn for designated wear period Order is manufactured and physically shipped to customer Order placed; unique serial numbers are assigned for each participant’s dosimeter ~45 days from date dosimeter was received by client Neutron (Nuclear) X-Ray (Healthcare) Current Monthly Process (~15 million badges annually) Technology Key Facts Energy Spectrum
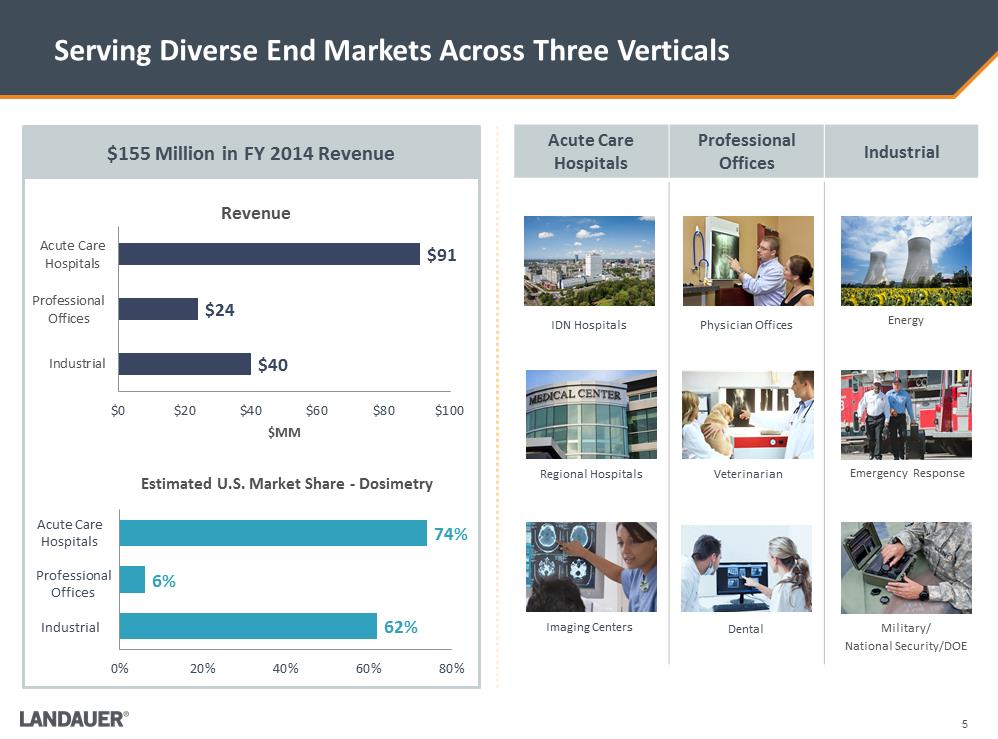 $40 $24 $91 Industrial ProfessionalOffices Acute CareHospitals $0 $20 $40 $60 $80 $100 $MM Revenue Serving Diverse End Markets Across Three Verticals Acute Care Hospitals Professional Offices Industrial Energy Emergency Response Military/ National Security/DOE W:\Landauer\Investors\Investor deck\Images\73470968_5.jpg W:\Landauer\Investors\Investor deck\Images\L41513.2429.jpg Physician Offices IDN Hospitals Veterinarian Dental Regional Hospitals Imaging Centers 62% 6% 74% 0% 20% 40% 60% 80% Estimated U.S. Market Share -Dosimetry $155 Million in FY 2014 Revenue Acute Care Hospitals Professional Offices Industrial
$40 $24 $91 Industrial ProfessionalOffices Acute CareHospitals $0 $20 $40 $60 $80 $100 $MM Revenue Serving Diverse End Markets Across Three Verticals Acute Care Hospitals Professional Offices Industrial Energy Emergency Response Military/ National Security/DOE W:\Landauer\Investors\Investor deck\Images\73470968_5.jpg W:\Landauer\Investors\Investor deck\Images\L41513.2429.jpg Physician Offices IDN Hospitals Veterinarian Dental Regional Hospitals Imaging Centers 62% 6% 74% 0% 20% 40% 60% 80% Estimated U.S. Market Share -Dosimetry $155 Million in FY 2014 Revenue Acute Care Hospitals Professional Offices Industrial
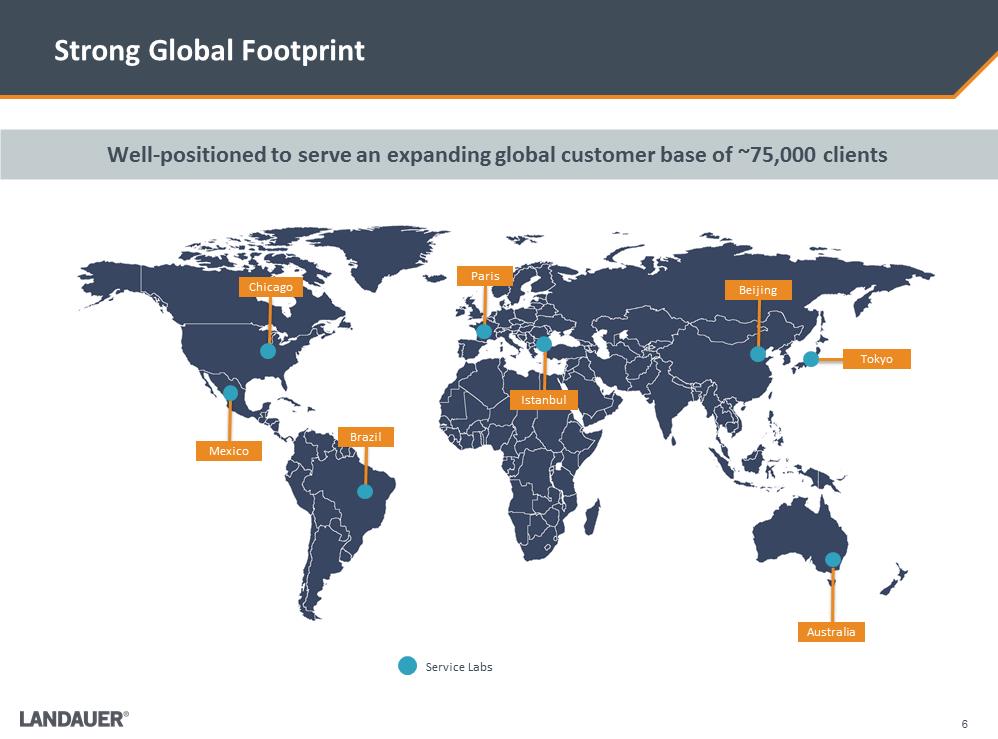 Strong Global Footprint Well-positioned to serve an expanding global customer base of ~75,000 clients Service Labs Chicago Mexico Brazil Paris Istanbul Beijing Tokyo Australia
Strong Global Footprint Well-positioned to serve an expanding global customer base of ~75,000 clients Service Labs Chicago Mexico Brazil Paris Istanbul Beijing Tokyo Australia
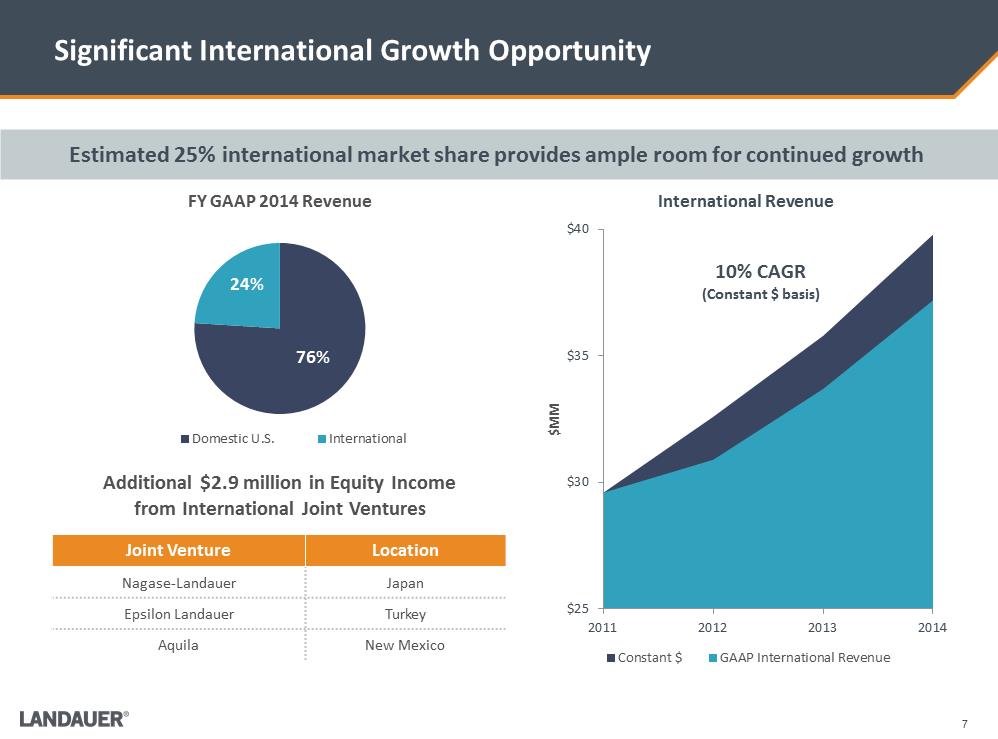 Significant International Growth Opportunity $25 $30 $35 $40 2011 2012 2013 2014 $MM International Revenue Constant $ GAAP International Revenue 76% 24% FY GAAP 2014 Revenue Domestic U.S. International Additional $2.9 million in Equity Income from International Joint Ventures Joint Venture Location Nagase-Landauer Japan Epsilon Landauer Turkey Aquila New Mexico 10% CAGR (Constant $ basis) Estimated 25% international market share provides ample room for continued growth
Significant International Growth Opportunity $25 $30 $35 $40 2011 2012 2013 2014 $MM International Revenue Constant $ GAAP International Revenue 76% 24% FY GAAP 2014 Revenue Domestic U.S. International Additional $2.9 million in Equity Income from International Joint Ventures Joint Venture Location Nagase-Landauer Japan Epsilon Landauer Turkey Aquila New Mexico 10% CAGR (Constant $ basis) Estimated 25% international market share provides ample room for continued growth
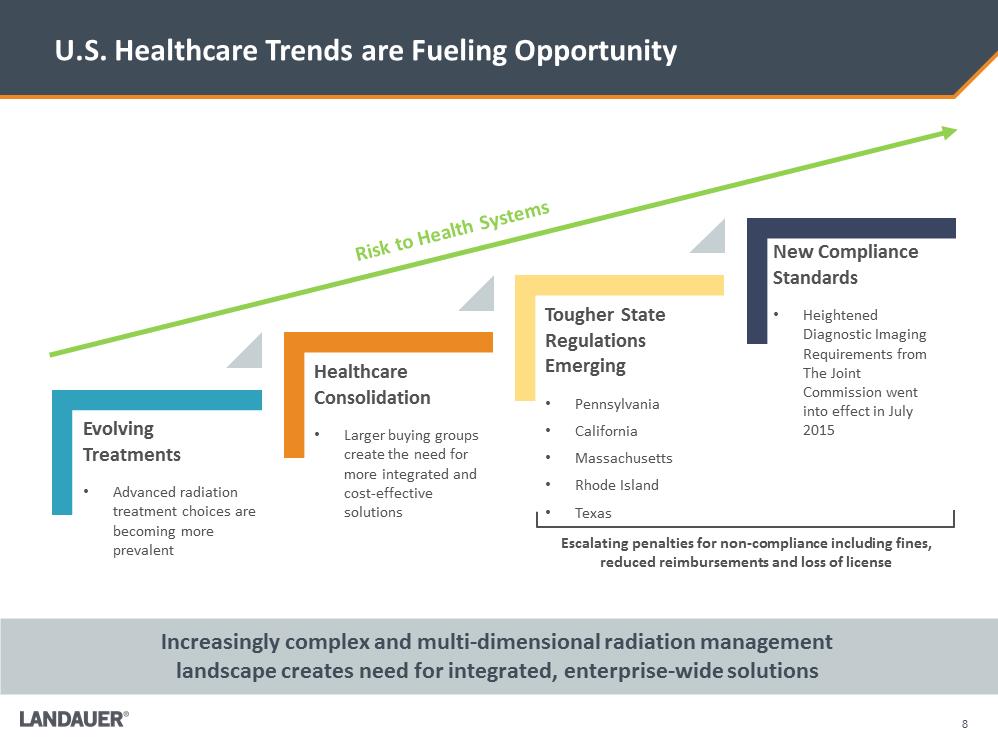 U.S. Healthcare Trends are Fueling Opportunity Increasingly complex and multi-dimensional radiation management landscape creates need for integrated, enterprise-wide solutions Risk to Health Systems Tougher State Regulations Emerging •Pennsylvania •California •Massachusetts •Rhode Island •Texas Healthcare Consolidation •Larger buying groups create the need for more integrated and cost-effective solutions Evolving Treatments •Advanced radiation treatment choices are becoming more prevalent New Compliance Standards •Heightened Diagnostic Imaging Requirements from The Joint Commission went into effect in July 2015 Escalating penalties for non-compliance including fines, reduced reimbursements and loss of license
U.S. Healthcare Trends are Fueling Opportunity Increasingly complex and multi-dimensional radiation management landscape creates need for integrated, enterprise-wide solutions Risk to Health Systems Tougher State Regulations Emerging •Pennsylvania •California •Massachusetts •Rhode Island •Texas Healthcare Consolidation •Larger buying groups create the need for more integrated and cost-effective solutions Evolving Treatments •Advanced radiation treatment choices are becoming more prevalent New Compliance Standards •Heightened Diagnostic Imaging Requirements from The Joint Commission went into effect in July 2015 Escalating penalties for non-compliance including fines, reduced reimbursements and loss of license
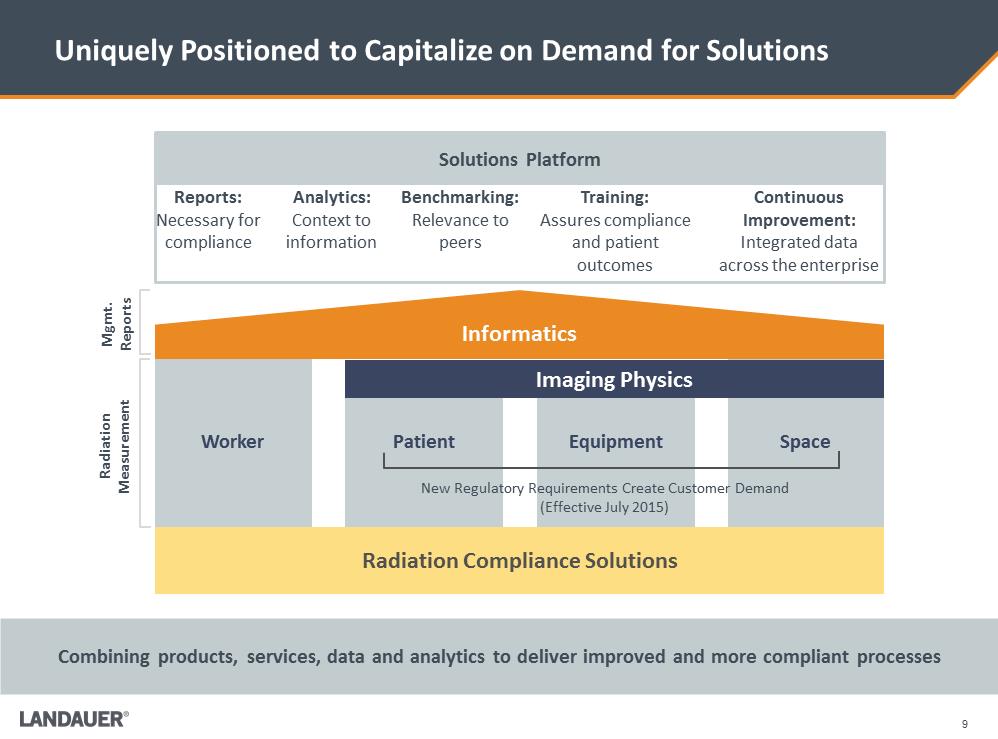 Reports: Necessary for compliance Analytics: Context to information Benchmarking: Relevance to peers Training: Assures compliance and patient outcomes Continuous Improvement: Integrated data across the enterprise Uniquely Positioned to Capitalize on Demand for Solutions Combining products, services, data and analytics to deliver improved and more compliant processes Mgmt. Reports Space Radiation Compliance Solutions Informatics Radiation Measurement Equipment Patient Worker Imaging Physics New Regulatory Requirements Create Customer Demand (Effective July 2015) Solutions Platform
Reports: Necessary for compliance Analytics: Context to information Benchmarking: Relevance to peers Training: Assures compliance and patient outcomes Continuous Improvement: Integrated data across the enterprise Uniquely Positioned to Capitalize on Demand for Solutions Combining products, services, data and analytics to deliver improved and more compliant processes Mgmt. Reports Space Radiation Compliance Solutions Informatics Radiation Measurement Equipment Patient Worker Imaging Physics New Regulatory Requirements Create Customer Demand (Effective July 2015) Solutions Platform
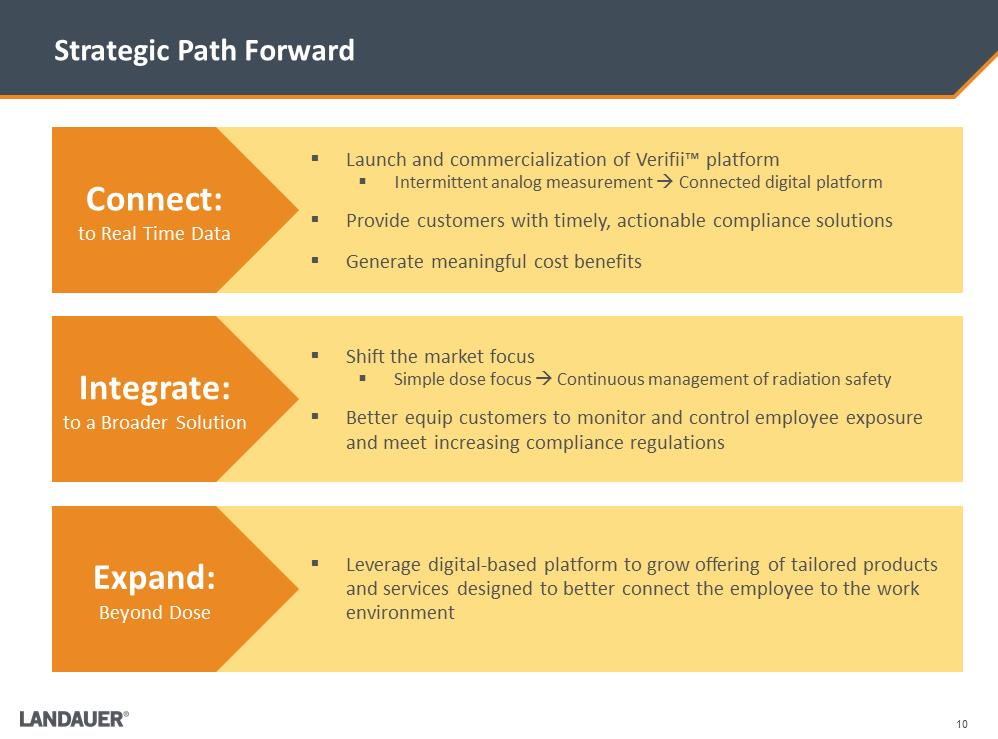 Strategic Path Forward .Shift the market focus .Simple dose focus .Continuous management of radiation safety .Better equip customers to monitor and control employee exposure and meet increasing compliance regulations .Leverage digital-based platform to grow offering of tailored products and services designed to better connect the employee to the work environment .Launch and commercialization of Verifii™ platform .Intermittent analog measurement .Connected digital platform .Provide customers with timely, actionable compliance solutions .Generate meaningful cost benefits Connect: to Real Time Data Integrate: to a Broader Solution Expand: Beyond Dose
Strategic Path Forward .Shift the market focus .Simple dose focus .Continuous management of radiation safety .Better equip customers to monitor and control employee exposure and meet increasing compliance regulations .Leverage digital-based platform to grow offering of tailored products and services designed to better connect the employee to the work environment .Launch and commercialization of Verifii™ platform .Intermittent analog measurement .Connected digital platform .Provide customers with timely, actionable compliance solutions .Generate meaningful cost benefits Connect: to Real Time Data Integrate: to a Broader Solution Expand: Beyond Dose
 Verifii™: Redefining Our Core Technology .Provides customers with a scalable, digital platform that connects employees to their environment .Eliminates fragmented manual processes enabling focus on building a robust radiation management solution .Timely radiation data adds value to customers by allowing them to take targeted actions for practical and measurable process improvements .Expect launch in 2016 A truly differentiated product and platform for incremental growth
Verifii™: Redefining Our Core Technology .Provides customers with a scalable, digital platform that connects employees to their environment .Eliminates fragmented manual processes enabling focus on building a robust radiation management solution .Timely radiation data adds value to customers by allowing them to take targeted actions for practical and measurable process improvements .Expect launch in 2016 A truly differentiated product and platform for incremental growth
 Creating a New Standard with Digital Dosimetry Real-time and historic data & analytics are available to client via web & mobile apps Data is processed and stored in the cloud Dosimeter connects via Bluetooth LE to docking base station Dosimeter is assigned to individual onsite; establishing chain of custody ~30 million incidents measured annually (~1% physically handled) Allows customers to track trends, monitor event specific dose levels and enact behavioral changes to better manage long-term exposure
Creating a New Standard with Digital Dosimetry Real-time and historic data & analytics are available to client via web & mobile apps Data is processed and stored in the cloud Dosimeter connects via Bluetooth LE to docking base station Dosimeter is assigned to individual onsite; establishing chain of custody ~30 million incidents measured annually (~1% physically handled) Allows customers to track trends, monitor event specific dose levels and enact behavioral changes to better manage long-term exposure
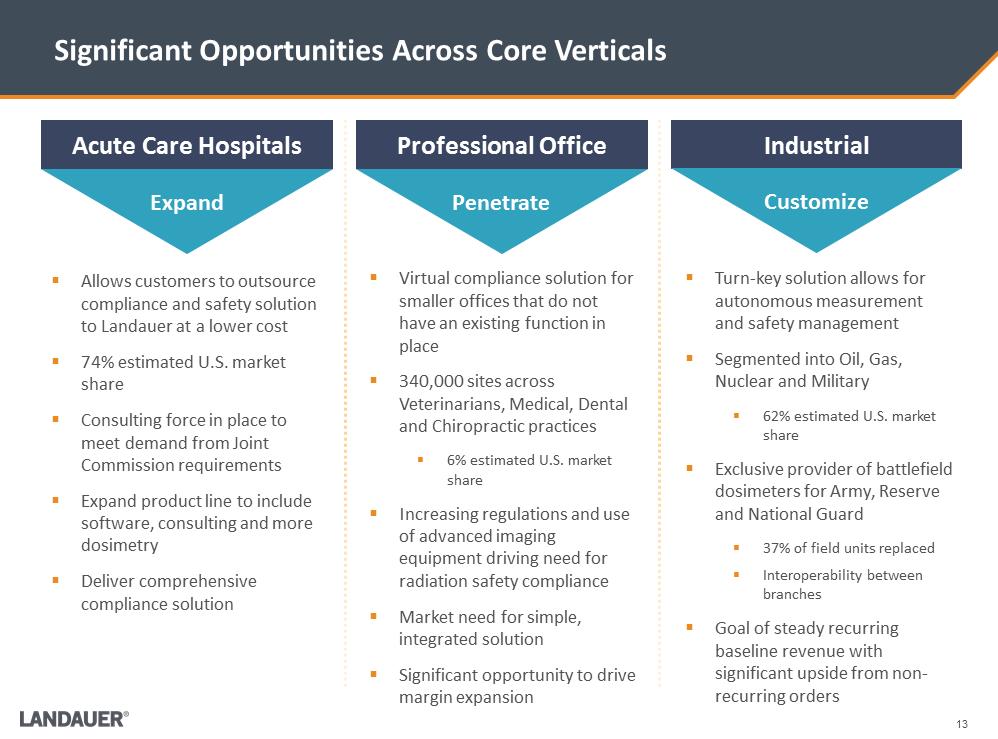 Significant Opportunities Across Core Verticals .Virtual compliance solution for smaller offices that do not have an existing function in place .340,000 sites across Veterinarians, Medical, Dental and Chiropractic practices .6% estimated U.S. market share .Increasing regulations and use of advanced imaging equipment driving need for radiation safety compliance .Market need for simple, integrated solution .Significant opportunity to drive margin expansion Acute Care Hospitals Professional Office Industrial .Allows customers to outsource compliance and safety solution to Landauerat a lower cost .74% estimated U.S. market share .Consulting force in place to meet demand from Joint Commission requirements .Expand product line to include software, consulting and more dosimetry .Deliver comprehensive compliance solution .Turn-key solution allows for autonomous measurement and safety management .Segmented into Oil, Gas, Nuclear and Military .62% estimated U.S. market share .Exclusive provider of battlefield dosimeters for Army, Reserve and National Guard .37% of field units replaced .Interoperability between branches .Goal of steady recurring baseline revenue with significant upside from non- recurring orders Expand Penetrate Customize
Significant Opportunities Across Core Verticals .Virtual compliance solution for smaller offices that do not have an existing function in place .340,000 sites across Veterinarians, Medical, Dental and Chiropractic practices .6% estimated U.S. market share .Increasing regulations and use of advanced imaging equipment driving need for radiation safety compliance .Market need for simple, integrated solution .Significant opportunity to drive margin expansion Acute Care Hospitals Professional Office Industrial .Allows customers to outsource compliance and safety solution to Landauerat a lower cost .74% estimated U.S. market share .Consulting force in place to meet demand from Joint Commission requirements .Expand product line to include software, consulting and more dosimetry .Deliver comprehensive compliance solution .Turn-key solution allows for autonomous measurement and safety management .Segmented into Oil, Gas, Nuclear and Military .62% estimated U.S. market share .Exclusive provider of battlefield dosimeters for Army, Reserve and National Guard .37% of field units replaced .Interoperability between branches .Goal of steady recurring baseline revenue with significant upside from non- recurring orders Expand Penetrate Customize
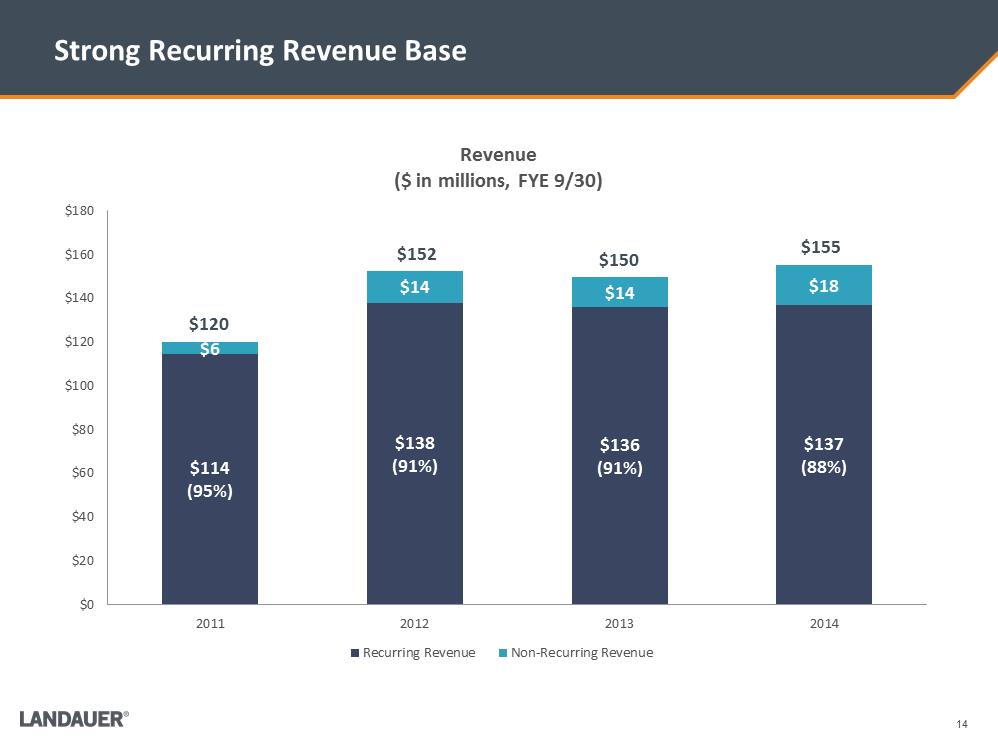 Strong Recurring Revenue Base $114 (95%) $138 (91%) $136 (91%) $137 (88%) $6 $14 $14 $18 $0 $20 $40 $60 $80 $100 $120 $140 $160 $180 2011 2012 2013 2014 Revenue ($ in millions, FYE 9/30) Recurring Revenue Non-Recurring Revenue $120 $152 $150 $155
Strong Recurring Revenue Base $114 (95%) $138 (91%) $136 (91%) $137 (88%) $6 $14 $14 $18 $0 $20 $40 $60 $80 $100 $120 $140 $160 $180 2011 2012 2013 2014 Revenue ($ in millions, FYE 9/30) Recurring Revenue Non-Recurring Revenue $120 $152 $150 $155
 .New IT systems implemented in FY12 negatively impacted working capital in FY13 by $11.0 million and positively impacted working capital in FY14 by $10.6 million .System implementation impacted timing of customer collections and vendor payments Driving Strong Cash Flows $18.3 $21.9 $16.2 $32.5 $0.0 $5.0 $10.0 $15.0 $20.0 $25.0 $30.0 $35.0 2011 2012 2013 2014 Free Cash Flow ($ in millions, FYE 9/30)
.New IT systems implemented in FY12 negatively impacted working capital in FY13 by $11.0 million and positively impacted working capital in FY14 by $10.6 million .System implementation impacted timing of customer collections and vendor payments Driving Strong Cash Flows $18.3 $21.9 $16.2 $32.5 $0.0 $5.0 $10.0 $15.0 $20.0 $25.0 $30.0 $35.0 2011 2012 2013 2014 Free Cash Flow ($ in millions, FYE 9/30)
 Balanced Approach to Capital Allocation .Core business investments focused on top and bottom line growth .Emphasis on higher margin, annuity-style opportunities .Commercialization of next generation digital dosimetry platform while transitioning from current OSL crystal-based platform .Management compensation tied to ROIC .Long-term target of 2X –2.5X Debt/EBITDA leverage ratio .Currently over 3% dividend yield .Long history of paying competitive dividend for over 20 years Fund Competitive Dividend Invest in the Business Pay Down Debt
Balanced Approach to Capital Allocation .Core business investments focused on top and bottom line growth .Emphasis on higher margin, annuity-style opportunities .Commercialization of next generation digital dosimetry platform while transitioning from current OSL crystal-based platform .Management compensation tied to ROIC .Long-term target of 2X –2.5X Debt/EBITDA leverage ratio .Currently over 3% dividend yield .Long history of paying competitive dividend for over 20 years Fund Competitive Dividend Invest in the Business Pay Down Debt
 Compelling Investment Opportunity Industry leading position, compelling market trends and strong financial profile drives long-term shareholder value •The Joint Commission standards effective in July 2015 in the United States -Impacts 7,100 of the 20,500 U.S. healthcare organizations and programs, including hospitals, imaging clinics and certain ambulatory care centers -Estimated incremental cost of ~$30,000 annually for a hospital to comply with new standards •International standards becoming more restrictive •Next generation Verifii™ platform on target for 2016 launch •Well positioned to meet emerging need for broader solution •Differentiated position creates higher barrier to entry for competition •Strong margins and limited capital requirements deliver compelling free cash flow •Defined capital allocation plan supports investment in growth initiatives and dividend •Industry-leading technology; proprietary Optically Stimulated Luminescence (OSL) crystal manufacturing process •Installed based of over 73,000 customers; 1.8 million individuals served worldwide •90% recurring revenue; 94% annual customer retention •Ability to provide enterprise radiation management or product only model Global Leader in Occupational Radiation Products and Services Increased Regulation Drives Demand Compelling Growth and Expansion Opportunities Strong Financial Profile
Compelling Investment Opportunity Industry leading position, compelling market trends and strong financial profile drives long-term shareholder value •The Joint Commission standards effective in July 2015 in the United States -Impacts 7,100 of the 20,500 U.S. healthcare organizations and programs, including hospitals, imaging clinics and certain ambulatory care centers -Estimated incremental cost of ~$30,000 annually for a hospital to comply with new standards •International standards becoming more restrictive •Next generation Verifii™ platform on target for 2016 launch •Well positioned to meet emerging need for broader solution •Differentiated position creates higher barrier to entry for competition •Strong margins and limited capital requirements deliver compelling free cash flow •Defined capital allocation plan supports investment in growth initiatives and dividend •Industry-leading technology; proprietary Optically Stimulated Luminescence (OSL) crystal manufacturing process •Installed based of over 73,000 customers; 1.8 million individuals served worldwide •90% recurring revenue; 94% annual customer retention •Ability to provide enterprise radiation management or product only model Global Leader in Occupational Radiation Products and Services Increased Regulation Drives Demand Compelling Growth and Expansion Opportunities Strong Financial Profile
 Financial Overview
Financial Overview
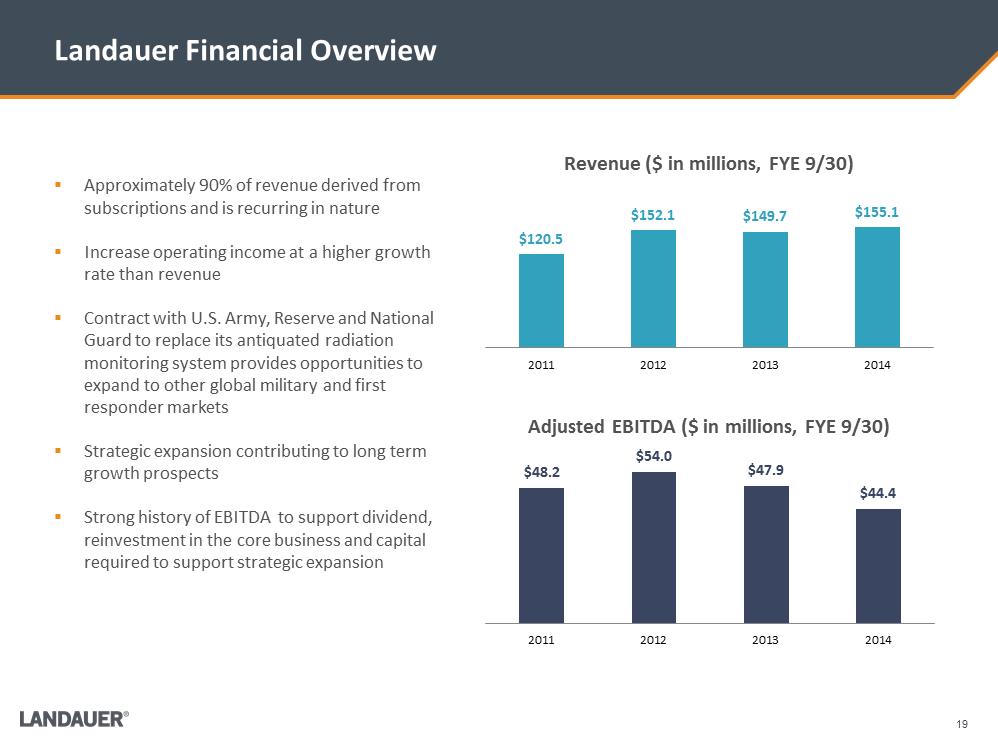
Landauer Financial Overview $120.5 $152.1 $149.7 $155.1 2011 2012 2013 2014 Revenue ($ in millions, FYE 9/30) $48.2 $54.0 $47.9 $44.4 2011 2012 2013 2014 Adjusted EBITDA ($ in millions, FYE 9/30) .Approximately 90% of revenue derived from subscriptions and is recurring in nature .Increase operating income at a higher growth rate than revenue .Contract with U.S. Army, Reserve and National Guard to replace its antiquated radiation monitoring system provides opportunities to expand to other global military and first responder markets .Strategic expansion contributing to long term growth prospects .Strong history of EBITDA to support dividend, reinvestment in the core business and capital required to support strategic expansion
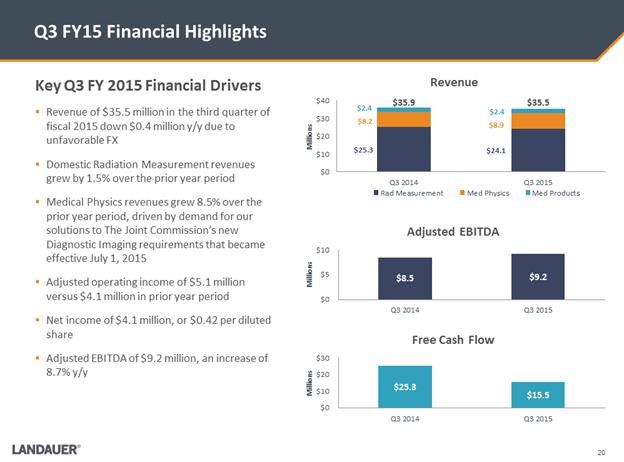
Key Q3 FY 2015 Financial Drivers .Revenue of $35.5 million in the third quarter of fiscal 2015 down $0.4 million y/y due to unfavorable FX .Domestic Radiation Measurement revenues grew by 1.5% over the prior year period .Medical Physics revenues grew 8.5% over the prior year period, driven by demand for our solutions to The Joint Commission’s new Diagnostic Imaging requirements that became effective July 1, 2015 .Adjusted operating income of $5.1 million versus $4.1 million in prior year period .Net income of $4.1 million, or $0.42 per diluted share .Adjusted EBITDA of $9.2 million, an increase of 8.7% y/y Q3 FY15 Financial Highlights $25.3 $24.1 $8.2 $8.9 $2.4 $2.4 $0 $10 $20 $30 $40 Q3 2014 Q3 2015 Millions Revenue Rad Measurement Med Physics Med Products $35.9 $35.5 $8.5 $9.2 $0 $5 $10 Q3 2014 Q3 2015 Millions Adjusted EBITDA $25.3 $15.5 $0 $10 $20 $30 Q3 2014 Q3 2015 Millions Free Cash Flow
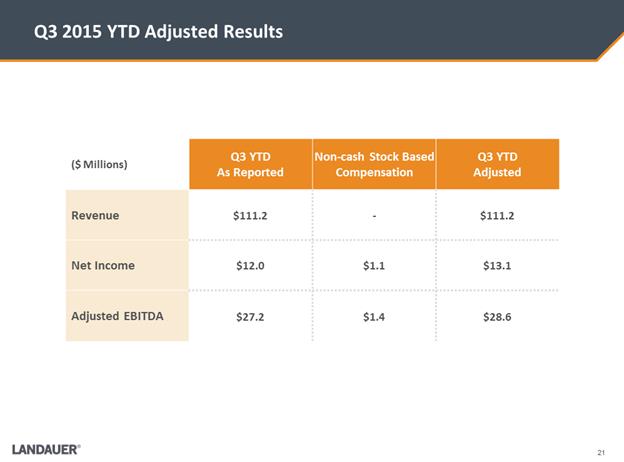
Q3 2015 YTD
Adjusted Results ($ Millions) Q3 YTD As Reported Non-cash Stock Based Compensation Q3 YTD Adjusted Revenue $111.2 - $111.2 Net Income $12.0 $1.1 $13.1 Adjusted EBITDA $27.2 $1.4 $28.6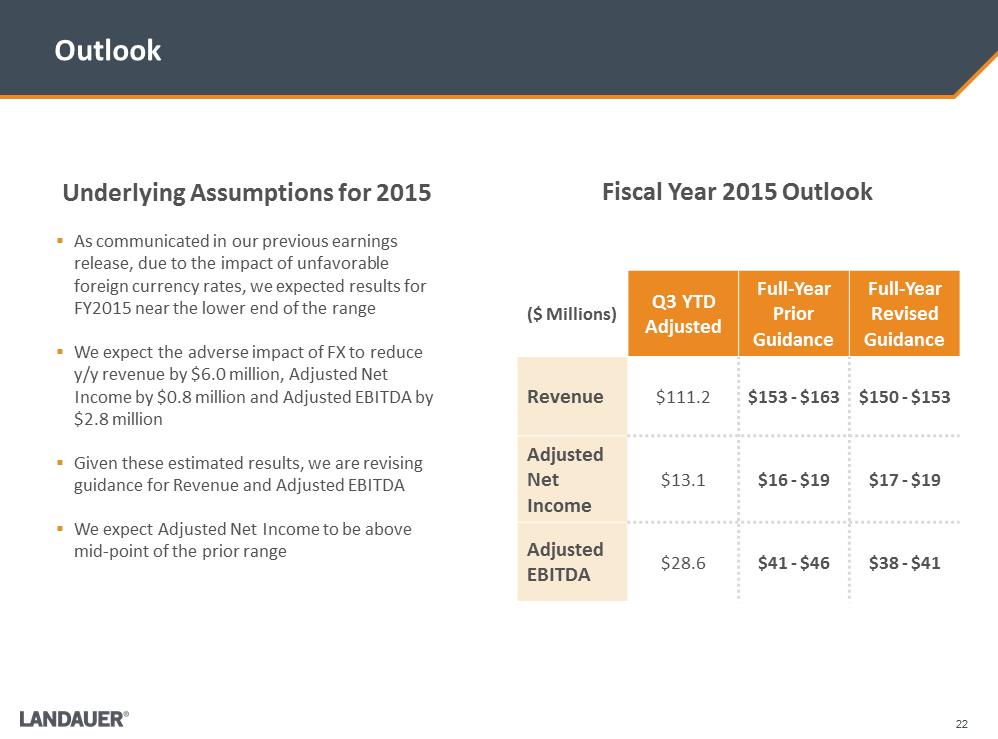 Underlying Assumptions for 2015 .As communicated in our previous earnings release, due to the impact of unfavorable foreign currency rates, we expected results for FY2015 near the lower end of the range .We expect the adverse impact of FX to reduce y/y revenue by $6.0 million, Adjusted Net Income by $0.8 million and Adjusted EBITDA by $2.8 million .Given these estimated results, we are revising guidance for Revenue and Adjusted EBITDA .We expect Adjusted Net Income to be above mid-point of the prior range Outlook Fiscal Year 2015 Outlook ($ Millions) Q3 YTD Adjusted Full-Year Prior Guidance Full-Year Revised Guidance Revenue $111.2 $153 -$163 $150 -$153 Adjusted Net Income $13.1 $16 -$19 $17 -$19 Adjusted EBITDA $28.6 $41 -$46 $38 -$41
Underlying Assumptions for 2015 .As communicated in our previous earnings release, due to the impact of unfavorable foreign currency rates, we expected results for FY2015 near the lower end of the range .We expect the adverse impact of FX to reduce y/y revenue by $6.0 million, Adjusted Net Income by $0.8 million and Adjusted EBITDA by $2.8 million .Given these estimated results, we are revising guidance for Revenue and Adjusted EBITDA .We expect Adjusted Net Income to be above mid-point of the prior range Outlook Fiscal Year 2015 Outlook ($ Millions) Q3 YTD Adjusted Full-Year Prior Guidance Full-Year Revised Guidance Revenue $111.2 $153 -$163 $150 -$153 Adjusted Net Income $13.1 $16 -$19 $17 -$19 Adjusted EBITDA $28.6 $41 -$46 $38 -$41
 Appendix
Appendix
 Business Facts Proven Technology Platform Fiscal 2014 Financial Highlights Diverse Markets .$155 Million in Annual Revenue .$37 Million in International Sales .~ 90% Recurring Revenues .$44.4 Million in Adjusted EBITDA .Adjusted EBITDA margin 28.6% .Headquartered in Glenwood, IL .Founded in 1954 .Approximately 650 employees .8 global markets .73,000 annual customers served; 1.8M annual individuals served .94%+ Customer retention rates inlight_clip_holder_front_L.png .Industry standard OSL technology .Passive measurement of wide range of radiation from low energy X-Ray to high energy neutron .Development of new digital dosimetry platform: Verifii™ Landauer at a Glance C:\Users\mdegraff\AppData\Local\Microsoft\Windows\Temporary Internet Files\Content.Outlook\EQH1N7PW\Dosimeter.2.png IDN Hospitals Imaging Centers Regional Hospitals Physician Offices Veterinarian Dental Energy Emergency Response Military/National Security/DOE W:\Landauer\Investors\Investor deck\Images\73470968_5.jpg W:\Landauer\Investors\Investor deck\Images\L41513.2429.jpg Acute Care Hospitals Professional Offices Industrial
Business Facts Proven Technology Platform Fiscal 2014 Financial Highlights Diverse Markets .$155 Million in Annual Revenue .$37 Million in International Sales .~ 90% Recurring Revenues .$44.4 Million in Adjusted EBITDA .Adjusted EBITDA margin 28.6% .Headquartered in Glenwood, IL .Founded in 1954 .Approximately 650 employees .8 global markets .73,000 annual customers served; 1.8M annual individuals served .94%+ Customer retention rates inlight_clip_holder_front_L.png .Industry standard OSL technology .Passive measurement of wide range of radiation from low energy X-Ray to high energy neutron .Development of new digital dosimetry platform: Verifii™ Landauer at a Glance C:\Users\mdegraff\AppData\Local\Microsoft\Windows\Temporary Internet Files\Content.Outlook\EQH1N7PW\Dosimeter.2.png IDN Hospitals Imaging Centers Regional Hospitals Physician Offices Veterinarian Dental Energy Emergency Response Military/National Security/DOE W:\Landauer\Investors\Investor deck\Images\73470968_5.jpg W:\Landauer\Investors\Investor deck\Images\L41513.2429.jpg Acute Care Hospitals Professional Offices Industrial

 I
I