Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - SEELOS THERAPEUTICS, INC. | d850362d8k.htm |
| EX-99.1 - EX-99.1 - SEELOS THERAPEUTICS, INC. | d850362dex991.htm |
| Exhibit 99.2 |

Biotech Showcase 2015 NASDAQ: APRI January 12, 2015

2” Forward-Looking Statements This corporate presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act, as amended. Statements in this presentation that are not purely historical are forward-looking statements. Such forward-looking statements include, among other things: references to the timing of initial and re-order shipments of Vitaros® and royalty revenues from the sale of Vitaros in various countries by Apricus’ commercial partners; the planned launch of Vitaros in France, Spain, Italy and other countries; the planned production of the required 12-month stability data and the expected timing of approval in Europe for the Vitaros room temperature device; the ongoing enrollment in the RayVa™ Phase 2a clinical trial of patients with Raynaud’s phenomenon secondary to scleroderma; the planned commencement of clinical trials for fispemifene and an approved pathway for RayVa and fispemifene; the size of the commercial opportunity for Vitaros, RayVa and fispemifene and the potential for such products to achieve commercial success; the expected funding under the credit facility of a second term loan subject to initiation of a Phase 2b trial for fispemifene; the planned outlicense of Femprox®, Vitaros and RayVa; and Apricus’ 2014 financial outlook, including cash projections. Actual results could differ from those projected in any forward-looking statements due to a variety of reasons that are outside the control of Apricus, including, but not limited to: its ability to further develop its product Vitaros for the treatment of erectile dysfunction, such as the room temperature version of Vitaros, and its product candidates RayVa for the treatment of Raynaud’s phenomenon and fispemifene for the treatment of secondary hypogonadism, chronic prostatitis and lower urinary tract symptoms in men, as well as the timing of such events; Apricus’ ability to carry out clinical studies for RayVa and fispemifene, as well as the timing and success of the results of such studies; Apricus’ dependence on its commercial partners to carry out the commercial launch or grow sales of Vitaros in various territories, such as Takeda in the United Kingdom, Sandoz in Sweden, Germany and Belgium, and Majorelle in France, and the potential for delays in the timing of commercial launches in additional countries; competition in the erectile dysfunction market and other markets in which Apricus and its partners operate; Apricus’ ability to obtain and maintain intellectual property protection for Vitaros, RayVa, fispemifene or any other product candidates; Apricus’ ability to raise additional funding that it may need to continue to pursue its commercial and business development plans; Apricus’ ability to draw the second term loan under the credit facility when expected, or at all, including Apricus’ failure to meet the conditions required to draw under the loan and security agreement; Apricus’ ability to remain in compliance with the terms and restrictions under the credit facility; Apricus’ ability to access additional capital under the equity facility; Apricus’ ability to obtain the requisite governmental approval for the room temperature version of Vitaros, RayVa, fispemifene and Femprox; and market conditions. These forward-looking statements are made as of the date of this presentation, and Apricus assumes no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements. Readers are urged to read the risk factors set forth in Apricus’ most recent annual report on Form 10-K, subsequent quarterly reports filed on Form 10- Q, and other filings made with the SEC. Copies of these reports are available from the SEC’s website at www.sec.gov or without charge from Apricus.

3” Apricus Overview Advancing Innovations in Specialty Urology and Rheumatology Markets — Headquarters: San Diego, CA — NASDAQ: APRI

3“4” Richard“Pascoe” CEO%&%Director% Brian“Dorsey” SVP,%Chief%Development%Officer% Barbara“Troupin,“MD” SVP,%Chief%Medical%Officer% Catherine“Bovenizer” VP,%Finance%&%Chief%Accoun@ng%Officer% Neil“Morton” VP,%Business%Development% Kleanthis“G.“Xanthopoulos,“Ph.D.” Chairman%% Rusty“Ray” Director% Deirdre“Y.“Gillespie,“M.D.” Director% Paul“V.“Maier” Director%% Wendell“Wierenga,“Ph.D.” Director%% Sandford“Smith” Director%% % Management(Team( Board(of(Directors( Experienced team leading life science companies Apricus Executive Management & Board of Directors
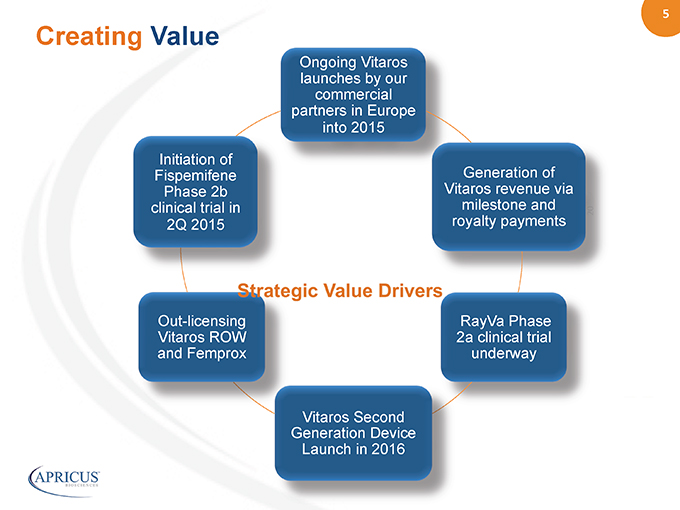
5” %%20% Ongoing Vitaros launches by our commercial partners in Europe into 2015 Generation of Vitaros revenue via milestone and royalty payments RayVa Phase 2a clinical trial underway Vitaros Second Generation Device Launch in 2016 Out-licensing Vitaros ROW and Femprox Initiation of Fispemifene Phase 2b clinical trial in 2Q 2015 Creating Value

6” First-in-class topical cream treatment for erectile dysfunction

7” Vitaros—Alprostadil/DDAIP.HCl — Only topically delivered cream approved for ED — Approved in Canada and Europe — Rapid onset (generally 5-15 minutes) — Significant efficacy and safety profile— including difficult to treat populations and those with greatest need: • Diabetics • Hypertensives • Post- prostatectomy • Patients on nitrates/alpha blockers • PDE-5 (e.g. Viagra®) failures Vitaros®: Treatment for Erectile Dysfunction (ED)
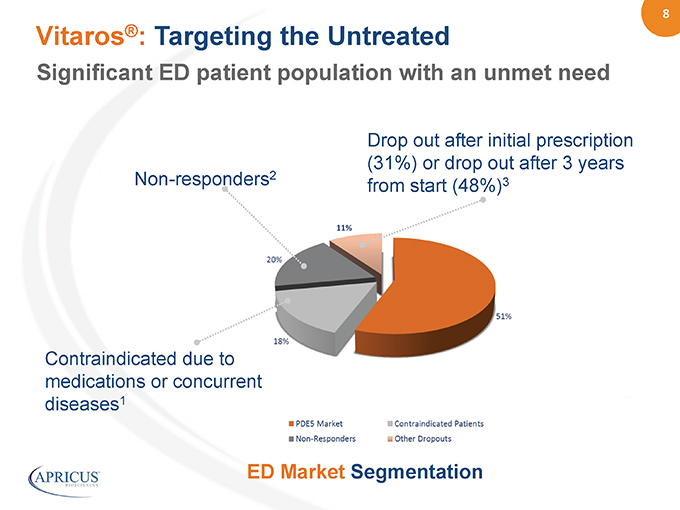
8” Vitaros®: Targeting the Untreated Drop out after initial prescription (31%) or drop out after 3 years Non-responders2 from start (48%)3 Contraindicated due to medications or concurrent diseases1 ED Market Segmentation Significant ED patient population with an unmet need

9” Vitaros®: Attractive Market Opportunity — Large ED market—$5.5B worldwide with $1.3B in Europe4 — Unique profile has potential to double the number of treatable patients1,2,3 — Launched in United Kingdom, Germany, Sweden and Belgium — Additional launches expected in 2015 in DCP approved countries — Royalty and milestone revenues expected through 2032 — Out-licensed in Canada, Europe, Africa, Australia, Israel and Middle East — Exploring options to advance the US clinical development program%

10” Second Manufacturing Site Operational — Technical transfer successfully completed at Groupe Parima in Canada — Commercial product orders placed with Parima by EU partners Second Generation Dispenser — Room Temperature Device (RTD): No refrigeration required — Targeting 24+ month shelf-life — On-track to generate stability data in 2015 — Approval targeted for 2016 — Key driver of Vitaros global market growth and expansion in 2016 and beyond Vitaros®: Treatment for Erectile Dysfunction (ED)

11” — 450,000 commercial doses manufactured for commercial use — Rx growth continues to exceed expectations — 432,000 additional doses on order at both manufacturing sites to be released in 1Q 2015 Vitaros is widely available in UK, Germany, Sweden and Belgium — 1H 2015 additional launches expected in France, Spain and Italy — Regulatory approval activities underway in non-DCP countries by existing partners Expansion of Commercialization Efforts in Europe — Vitaros currently promoted by Takeda and Sandoz with existing sales forces — Majorelle to launch in France with ~ 40 sales reps pending reimbursement decision — Commercial product orders placed by Bracco for Italy and Recordati for Spain Early results exceed our expectations in launched territories Vitaros®: Launch Update
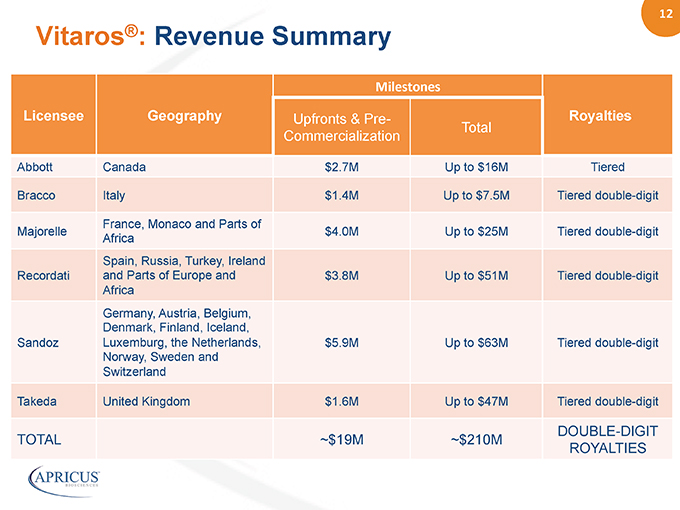
12” Licensee Geography Milestones” Upfronts & Pre- Royalties Commercialization Total Abbott Canada $2.7M Up to $16M Tiered Bracco Italy $1.4M Up to $7.5M Tiered double-digit Majorelle France, Monaco and Parts of Africa $4.0M Up to $25M Tiered double-digit Recordati Spain, Russia, Turkey, Ireland and Parts of Europe and Africa $3.8M Up to $51M Tiered double-digit Sandoz Germany, Austria, Belgium, Denmark, Finland, Iceland, Luxemburg, the Netherlands, Norway, Sweden and Switzerland $5.9M Up to $63M Tiered double-digit Takeda United Kingdom $1.6M Up to $47M Tiered double-digit TOTAL ~$19M ~$210M DOUBLE-DIGIT ROYALTIES Vitaros®: Revenue Summary 12” Licensee Geography Milestones”

13” %%20% Fispemifene Novel SERM for Urological Conditions in Men

14” Fispemifene: In-Licensing In October 2014, Apricus entered into a definitive agreement to license the exclusive U.S. development and commercialization rights for fispemifene from Forendo Pharma Ltd Deal Terms: — $10 Million venture debt transaction with Oxford Finance and Silicon Valley Bank payable over 4 years — Apricus in-license upfront fee valued at $12.5 million • $5 Million cash plus $7.5 Million in Apricus common stock valued based on the 360-day average stock price of $2.08/ share — Future regulatory, milestone and royalty payments will be due to Forendo

15” Fispemifene: Overview — Substantial $1 billion plus opportunity in $2 billion “low T” market6 — A novel selective estrogen receptor modulator (SERM) — New chemical entity (NCE) — Oral once-daily dosing — First tissue-specific SERM designed specifically for men — Potential to treat male secondary hypogonadism (low testosterone), chronic prostatitis and lower urinary tract symptoms — Composition of matter patent expires in the latter part of 2025, including an expected patent term extension, along with additional issues and pending applications that are expected to extend to 2028 and beyond
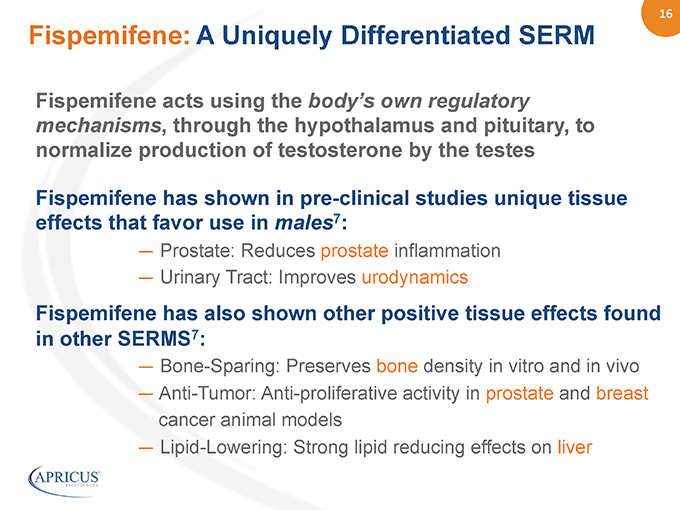
16” Fispemifene acts using the body’s own regulatory mechanisms, through the hypothalamus and pituitary, to normalize production of testosterone by the testes Fispemifene has shown in pre-clinical studies unique tissue effects that favor use in males7: — Prostate: Reduces prostate inflammation — Urinary Tract: Improves urodynamics Fispemifene has also shown other positive tissue effects found in other SERMS7: — Bone-Sparing: Preserves bone density in vitro and in vivo — Anti-Tumor: Anti-proliferative activity in prostate and breast cancer animal models — Lipid-Lowering: Strong lipid reducing effects on liver Fispemifene: A Uniquely Differentiated SERM
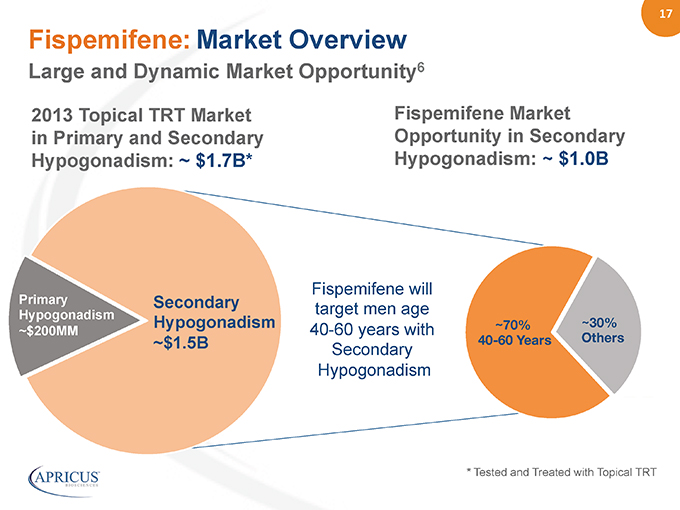
17” Fispemifene: Market Overview Large and Dynamic Market Opportunity6 Primary Hypogonadism ~$200MM” “ Fispemifene will target men age 40-60 years with Secondary Hypogonadism Secondary Hypogonadism ~$1.5B 2013 Topical TRT Market in Primary and Secondary Hypogonadism: ~ $1.7B*% * Tested and Treated with Topical TRT ~30% Others ~70% 40-60 Years Fispemifene Market Opportunity in Secondary Hypogonadism: ~ $1.0B%

18” Fispemifene vs. Testosterone Replacement Therapies (TRT) Fispemifene Testosterone Replacement Dosing Oral, simple once daily Injectable or topical administration Bone Beneficial effects Limited effects Prostate Expected to be neutral or positive anti-inflammatory (improved urodynamics) Enlargement risk Testes Beneficial effects: maintains fertility and spermatogenesis Testicular atrophy: impaired spermatogenesis / infertility Cardiovascular Lack of supra-physiological T levels associated with increased risk Increases hematocrit and blood pressure. Concern with supraphysiological T levels Partner risks None Topical exposure risk to female and children; label warnings / precautions Abuse potential Low – endogenous stimulation preserves natural feedback mechanism Prone to abuse, over treatment and unpredictable peak / trough effects; controlled substance
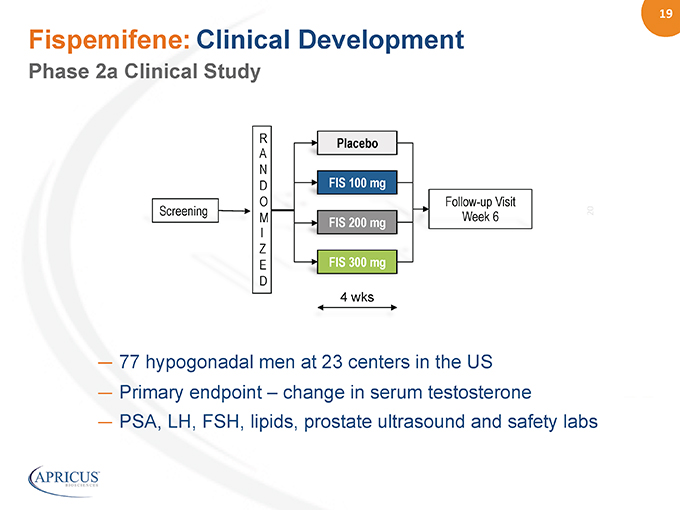
19” %%20% Fispemifene: Clinical Development Follow-up Visit Week 6 FIS 100 mg FIS 300 mg FIS 200 mg 4 wks RA ND O M I Z E D Screening Placebo — 77 hypogonadal men at 23 centers in the US — Primary endpoint – change in serum testosterone — PSA, LH, FSH, lipids, prostate ultrasound and safety labs Phase 2a Clinical Study
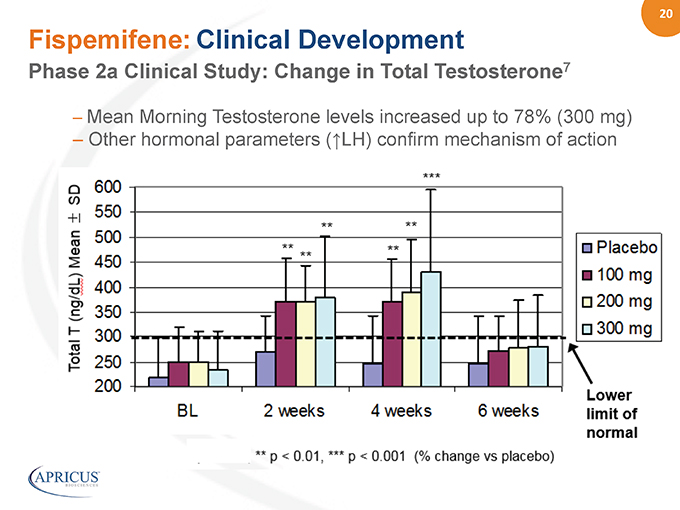
20” %%20% Fispemifene: Clinical Development – Mean Morning Testosterone levels increased up to 78% (300 mg) – Other hormonal parameters (?LH) confirm mechanism of action Phase 2a Clinical Study: Change in Total Testosterone7

21” Fispemifene: Clinical Development Fispemifene has been safe and well tolerated at all dose levels No clinically significant changes were found in: – Serum biochemistry – Hematology or urinalysis parameters – Vital signs, physical examination or ECG – Prostate health (PSA and prostate volume) examined in Phase 2 clinical trials – Obstructive symptoms, or urinary QoL Most common adverse reactions (191 subjects treated so far): – Upper respiratory tract infection (6 subjects) – Nausea, dizziness (5 subjects each) – Abdominal pain, diarrhea, headache and spontaneous penile erection (2 subjects each) Summary of Clinical Safety

22” %%20% Fispemifene: Clinical Development ? Clinical proof of concept demonstrated in secondary hypogonadism in two U.S. based Phase 2a trials ? Pre-clinical studies demonstrated a beneficial effect of fispemifene on urodynamics and a reduction in estrogen-induced prostatic inflammation 1H 2015: Commence Phase 2b dose ranging/confirmation trial (12 week treatment) in secondary hypogonadism • Evaluation to assess symptomatic response to treatment% • Evaluation of effects on lower urinary tract symptoms 2016: Target start of Phase 3 studies in approximately 1000 patients 2018: Target NDA submission “Phase 2b ready” asset

23” Potential first-in-class topical cream treatment for Raynaud’s Phenomenon Secondary to Scleroderma (alprostadil cream)

24” RayVa™: Treatment for Raynaud’s Phenomenon Secondary to Scleroderma (SSc) RayVa – Alprostadil/DDAIP.HCl — Topical, on-demand route of administration — RayVa—increased blood flow observed in preclinical models of Raynaud’s phenomenon with RayVa, using a cold challenge test Clear and Defined Pathway to Approval — Enrollment ongoing in Phase 2a proof-of-concept trial in approximately 45 patients — Working with nationally recognized Raynaud’s and scleroderma experts and academic sites (University of Michigan, Cleveland Clinic and University of Pittsburgh) — Phase 2b trial in approximately 80 patients expected following establishment of proof-of-concept — Two pivotal trials, using validated endpoints accepted by FDA, expected to begin following End-of-Phase 2 meeting — May qualify for Priority Review following NDA submission, which could occur as early as 2017

25” — Raynaud’s phenomenon is an episodic vasoconstriction of the distal extremities affecting an estimated 3-5% of the US population8,9 — Increased incidence in woman (approx. 80% of scleroderma patients)10 — Triggers include cold, emotional stress and vibration Symptoms: — Pain, tingling, numbness, and coldness — Affected areas show at least two color changes: ? White, (pallor), Blue (cyanosis), and Red (hyperemia) ? Brittle and ridged nails Raynaud’s Phenomenon

26” Condition is classified as either primary or secondary: • Primary (7M-12M) 8,9,11 – Not associated with an underlying disease state (such as scleroderma or lupus) – Often goes undiagnosed and untreated Raynaud’s Phenomenon • Secondary (~500K) 9,11 Secondary to Scleroderma (~100,000)12 – Secondary Raynaud’s Phenomenon is driven by an underlying medical condition, such as scleroderma, lupus or rheumatoid arthritis Attractive Commercial Opportunity — Currently no approved treatment options for Raynaud’s Phenomenon in the United States, representing an unmet medical need — Targeted call point—over 4,500 rheumatologists in the US currently treating secondary Raynaud’s patients13 — Broad IP position with potential exclusivity to 2032

27” %%20% Financial Overview

28” Financial Dashboard NASDAQ: APRI Cash on-hand combined with access to additional capital through the Aspire Capital common stock purchase facility provides sufficient capital to support the current operating plan through 2015 44.4M† 55.1M† ~$70M† $16.1M‡ Shares outstanding Shares fully-diluted Market Cap Cash position † As of January 8, 2015 ‡ As of September 30, 2014 — Vitaros revenue expected to off-set operating expenses in 2015 and beyond — $5MM available under existing Oxford/SVB debt facility to fund fispemifene Phase 2b clinical trial

29” 2015 Milestones: Advancing the Pipeline Vitaros — Additional launches in major EU markets expected in 1H 2015 — Expected cash flows from milestone and royalty payments — Pursue additional ex-US commercial partnerships — Generate room temperature device stability data to support 2016 approval in Europe Fispemifene — Confirm regulatory pathway in US for secondary hypogonadism in 1Q 2015 — Initiation of Phase 2b clinical trial in 2Q 2015 — Completion of Phase 2b trial enrollment expected in 4Q 2015 — Initiate Phase 3 clinical program preparations in 2H 2015 — Initiate required long-term toxicity studies in 2H 2015 RayVa — Establish proof of concept through ongoing Phase 2a trial in 1H 2015 — Seek additional US regulatory guidance for Phase 3 program in 2H 2015 — Pursue potential ex-US development and commercialization partner(s) in 2H 2015

Contacts InsStuSonal“Investors:”” Angeli“Kolhatkar” angeli@areciaadvisors.com% Arecia%Advisors,%LLC% (917)%387P4770%% %% Retail“Investors:”” Chris“Eddy,“David“Collins” apri@catalystPir.com%% Catalyst%Global% (212)%924P9800%

Biotech Showcase 2015 NASDAQ: APRI January 12, 2015

32” 1 D2 Market Research, June 2007. 2 J Sex Med 2012; 9: 2361–2369. 3 International Journal of Urology 2007; 14: 339-342. 4 IMS Health 2013. 5 Analyst estimates (Cantor Fitzgerald, August 22, 2013). 6 According to data on file with the Company, certain published data and information presented at the September 17, 2014 Joint Meeting of the Bone, Reproductive and Urologic Drugs Advisory Committee and the Drug Safety and Risk Management Advisory Committee. 7 Data on file. 8 N Engl J Med 2002; 347: 1001–1008. 9 Drugs 2007; 67: 517-525. 10 Medicine 2013; 92: 191-205. 11 2012 U.S. Census Bureau: State and County QuickFacts (http://quickfacts.census.gov/qfd/states/ 00000.html). 12 Curr Opin Rheumatol 2012; 24: 165–170; American College of Rheumatology ( http://www.rheumatology.org/Practice/Clinical/Patients/Diseases_And_Conditions/Scleroderma). 13 American Medical Association 2011. References
