Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Cardiff Oncology, Inc. | a14-14563_18k.htm |
| EX-99.2 - EX-99.2 - Cardiff Oncology, Inc. | a14-14563_1ex99d2.htm |
Exhibit 99.1
|
|
Stephen Zaniboni | Chief Financial Officer Jefferies 2014 Global Healthcare Conference June 2, 2014 Better Cancer Monitoring with Cell-Free DNA in Urine |
|
|
Forward-Looking Statements Statements in this presentation about the Company's expectations, applications of its technology, markets, launch of tests and other statements that are not historical facts are "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934 and are based on management's current beliefs, assumptions, estimates and projections. Actual results may differ materially from those projected in the forward-looking statements for various reasons, including risks associated with product and test development, test transfer to contracting labs, government regulation, market acceptance, limited commercial experience, dependence on key personnel, obtaining financing and other factors discussed in the Company's periodic reports filed with the Securities and Exchange Commission. DISCLAIMER |
|
|
Our Goal: Improve Treatment Outcomes For Cancer Patients INTRODUCTION Trovagene’s technology detects and quantitates oncogene mutations from cell-free DNA in the urine of cancer patients for improved disease management Our diagnostic platform provides important clinical information beyond the current standard of care |
|
|
Key Assets Precision cancer monitoring technology addressing a high unmet clinical need Enables more frequent, non-invasive monitoring of oncogene mutation status, disease progression, and recurrence Numerous clinical collaborations with leading cancer centers Expanding clinical evidence to support broad market adoption Generating health economic data necessary for appropriate reimbursement CLIA certified, CAP accredited, high complexity lab to offer diagnostic services Significant patent portfolio around cell-free DNA in urine proprietary methods of extracting, purifying, preparing, and detecting cell-free DNA and RNA mutations INTRODUCTION NASDAQ: TROV $113M market cap* Molecular Diagnostic Specialist founded in 1999 NASDAQ listing 2012 $55M invested in R&D and IP ~$22.9M cash on hand (3/31/14) Focused on cancer management *as of 30 May 2014 Company Overview |
|
|
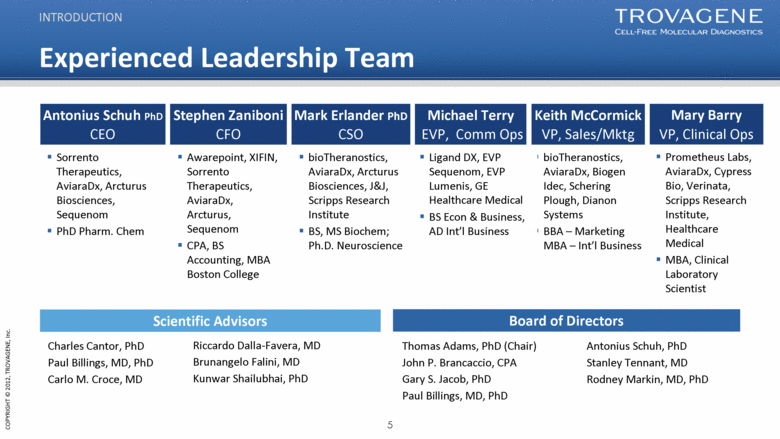
Experienced Leadership Team INTRODUCTION Thomas Adams, PhD (Chair) John P. Brancaccio, CPA Gary S. Jacob, PhD Paul Billings, MD, PhD Charles Cantor, PhD Paul Billings, MD, PhD Carlo M. Croce, MD Antonius Schuh, PhD Stanley Tennant, MD Rodney Markin, MD, PhD Scientific Advisors Antonius Schuh PhD CEO Sorrento Therapeutics, AviaraDx, Arcturus Biosciences, Sequenom PhD Pharm. Chem Stephen Zaniboni CFO Mark Erlander PhD CSO Michael Terry EVP, Comm Ops Keith McCormick VP, Sales/Mktg Awarepoint, XIFIN, Sorrento Therapeutics, AviaraDx, Arcturus, Sequenom CPA, BS Accounting, MBA Boston College bioTheranostics, AviaraDx, Arcturus Biosciences, J&J, Scripps Research Institute BS, MS Biochem; Ph.D. Neuroscience bioTheranostics, AviaraDx, Biogen Idec, Schering Plough, Dianon Systems BBA – Marketing MBA – Int’l Business Ligand DX, EVP Sequenom, EVP Lumenis, GE Healthcare Medical BS Econ & Business, AD Int’l Business Mary Barry VP, Clinical Ops Prometheus Labs, AviaraDx, Cypress Bio, Verinata, Scripps Research Institute, Healthcare Medical MBA, Clinical Laboratory Scientist Board of Directors Riccardo Dalla-Favera, MD Brunangelo Falini, MD Kunwar Shailubhai, PhD |
|
|
THE CLINICAL UTILITY OF CELL-FREE DNA FROM URINE Treatment Response and Resistance Monitoring Metastatic cancer patients per year: ~525,000 Price Per Test: $1,000 Tests Per Year: 6 TAM patients: 3.2M TAM $: ~$3.2B Average patient receives 8-10 imaging studies during initial 12 month period post-diagnosis Over 170M cancer screening and diagnostic procedures performed annually in the U.S. Cancer survivors: ~11M Price Per Test: $1,000 Tests Per Year: 1 TAM patients: ~11M TAM $: ~$11B Recurrence Monitoring Large Addressable Market; Est. 45% Rise in U.S. Cancer Incidence by 2030* *Smith, B.D. et al. Future of Cancer Incidence in the United States: Burdens Upon Ageing, Changing Nation, Journal of Clinical Oncology, Vol. 27, No. 17, June 10, 2009. Cancer Facts and Figures 2013, American Cancer Society, p. 4 American Cancer Society Website; http://www.cancer.org/cancer/cancerbasics/cancer-prevalence. Table 2 in Dinan, et. Al. Changes in the Use and Costs of Diagnostic Imaging Among Medicare Beneficiaries With Cancer, 1999-2006, JAMA, April 2010. |
|
|
Clinical Dilemma In Cancer Treatment Large body of knowledge has been developed around the molecular basis of cancer Paradigm Shift: Cancers are driven by oncogene mutations resulting in the trend toward new targeted therapies Effectiveness requires monitoring Resistance develops Therapies are expensive and can have side effects THE CLINICAL UTILITY OF CELL-FREE DNA FROM URINE IMAGING Rough indication of tumor size No molecular information TISSUE BIOPSY Major procedures, not possible in some cases Limitations in access to serial biopsies Tumor heterogeneity; may not obtain the right tissue from the tumor sample CIRCULATING TUMOR CELLS Very low sensitivity Technically challenging, expensive Current monitoring tools are insufficient |
|
|
Cancer Monitoring Using cfDNA Is An Emerging Trend THE CLINICAL UTILITY OF CELL-FREE DNA FROM URINE “Circulating tumor DNA levels showed a greater dynamic range, and greater correlation with changes in tumor burden, than did CA 15-3 or circulating tumor cells. This proof-of-concept analysis showed that circulating tumor DNA is an informative, inherently specific, and highly sensitive biomarker of metastatic breast cancer”. Sarah-Jane Dawson et. al., Analysis of Circulating Tumor DNA to Monitor Metastatic Breast Cancer, NEJM, Vol. 368, No. 12, March 2013 “Cancer is a disease of DNA. It does make it look as if our means of defining cancer by what tissue it arose in is going to go by the boards pretty soon. Lung, breast, blood—it almost doesn't matter. What matters are the genes that are mutated, the mistakes in the instruction book. For the first time, we have a clear list of targets that are much more smart-bomb opportunities than carpet-bomb opportunities." Dr. Frances Collins, National Institutes of Health (NIH) Director, ‘Politics on The Frontier of Science’, Wall Street Journal, November 8, 2013 |
|
|
Cell-free DNA Filters Into The Urine THE CLINICAL UTILITY OF CELL-FREE DNA FROM URINE Enabling Benefits Large sample volume Hours of continuous cfDNA collection Cell-free DNA stable in urine High frequency of collection possible Logistics/Convenience Truly non-invasive Patient can self-sample at home or clinic No medical professional required for specimen No refrigeration required No infection risk Lower cost Urine sample Tracking-cell free DNA Cells in the body die continuously; cancer cells die at an accelerated rate DNA is released into the bloodstream, which is then broken down into smaller segments and filtered by the kidney Small, stable DNA fragments collect in urine where Trovagene technology can identify and quantify mutations of interest |
|
|
A Decade of Scientific Work Validates Our Novel Approach 15 peer reviewed publications covering all nucleic acid types in urine DNA RNA miRNA Methylation Fields of Study Oncology Prenatal Infectious Disease Transplant TROVAGENE PLATFORM See appendix for details |
|
|
Core IP Extends Into Assay Detection & Development Original TrNA Patent Family Term to approx. 2018 US & EP Viral and Pathogen TrNA Families Term to approx. 2026 US, EP, JP, China, Australia, Canada, India 20-50 bp TrNA Family Term to approx. 2029 US & EP 51-110 bp TrNA Family Term to approx. 2034 US provisional app filed Transrenal miRNA Detection Family Term to approx. 2028 US, EP, JP, China, Australia, Canada, India Original Anion Exchange Purification Family Term to approx. 2027 US, EP, Canada Purification with Chaotropic Salt Family Term to approx. 2034 US provisional app filed PROPRIETARY ADVANTAGE |
|
|
Collaborations To Validate & Integrate Our PCM Platform CLINICAL PROGRAM |
|
|
Improved Patient Outcomes Strategy To Drive Clinical Adoption & Reimbursement CLINICAL PROGRAM STAGE 1 Demonstrate concordance of oncogene mutation status between urine and tumor tissue. Clinical Utility: Determine mutational status of actionable biomarkers in urine when a biopsy is not an option. Ex. BRAF, KRAS Diagnostic $100-$200M Revenue STAGE 2 Expand mutational coverage of urine-based actionable oncongene mutations for our PCM platform Clinical Utility: Quantitatively assess mutational status in urine longitudinally as an indicator of responsiveness to therapy, disease status & emergence of resistant mutations QUANTITATIVE Validation QUALITATIVE Validation Drug-Diagnostic STAGE 3 Demonstrate in multi-institutional trials that results from urine-based monitoring of actionable mutations increases patient progression-free survival (PFS) & overall survival (OS). Demonstrate health economic benefits of non-invasive oncogene mutation monitoring Clinical Utility: Quantitatively assess patient mutational status in urine longitudinally for mutational status and early detection of resistance to therapy as a decision tool for therapy selection $500+M Revenue Clinical Standard of Care Multi $B Revenue |
|
|
Stage 1 & 2: Summary of Ongoing Collaboration Programs MD Anderson Cancer Center KRAS and BRAF Mutational Analysis of Cell-free DNA from Urine in Patients with Advanced Cancers Multi-institutional Collaboration Includes Memorial Sloan-Kettering Cancer Center Detecting BRAF Mutations in Histiocytic Diseases Memorial Sloan-Kettering Cancer Center Detecting EGFR T790M Resistance Mutations in Lung Cancer USC Norris Cancer Center Monitoring and Emergence of KRAS Resistant Mutations in Colorectal Cancer US Oncology Detecting and Monitoring KRAS Mutations in Pancreatic Cancer Catholic Health Initiatives Multiple Clinician-Run Trials to Validate Clinical & Economic Benefits of TROV Platform; Potential Adoption Into CHI Network Pharmaceutical Collaboration Correlation of EGFR T790M Mutation Resistance vs. Drug Candidate Responsiveness in Lung Cancer Patients CLINICAL PROGRAM Stage 1 Determine mutational status of actionable biomarkers in urine when a biopsy is not an option Stage 2 Quantitatively assess patient mutational status in urine longitudinally as indicator of responsiveness to therapy and disease status of patient |
|
|
Cumulative # of urine and blood samples received by Trovagene to date Increasing Volume of Clinical Samples Received CLINICAL PROGRAM |
|
|
Clinical Results Presented & Published To Date Data presentation at AACR 2013/14 MD Anderson “All Comers” study, Mutational status correlates with response to therapy Publication in Journal Oncotarget Monitoring of patients with BRAF V600E malignancy 100% concordance with tissue biopsy Trovagene PCM technology identified BRAF V600E mutation when tissue biopsy was insufficient Clinical Guidelines Published in Journal Blood; References TROV Technology Alternative to tissue biopsy and other methods to diagnose and treat patients with Erdheim-Chester disease (ECD) CLINICAL PROGRAM Poster & Oral Presentation at ASCO 2014 Larger data set for MD Anderson/Mem. Sloan Kettering for histiocytosis (ECD) patients Longitudinal mutation load correlates with clinical response Data Publication at ASCO 2014 Additional data set from MD Anderson “All Comers” study Robust longitudinal monitoring results in patients with various advanced cancers Stat. significant correlation between mutation monitoring in urine & treatment response |
|
|
Clinical Guidelines Reference Trovagene’s PCM Technology Guidelines for The Diagnosis & Treatment of Erdheim-Chester Disease (ECD) Published in Blood – May 2014 Mention in clinical guidelines provides early evidence of the utility of Trovagene’s precision cancer monitoring technology ECD serves as a model disease and validates the use of our PCM platform when tissue biopsy presents a challenge Ability to monitor the BRAF V600E oncogene mutation in patients over time to determine treatment response was also demonstrated Roughly half of ECD Patients Have the BRAF V600E mutation Biopsy presents significant challenges in 60% of patients Insufficient tumor tissue Tumor heterogeneity CORPORATE SUMMARY |
|
|
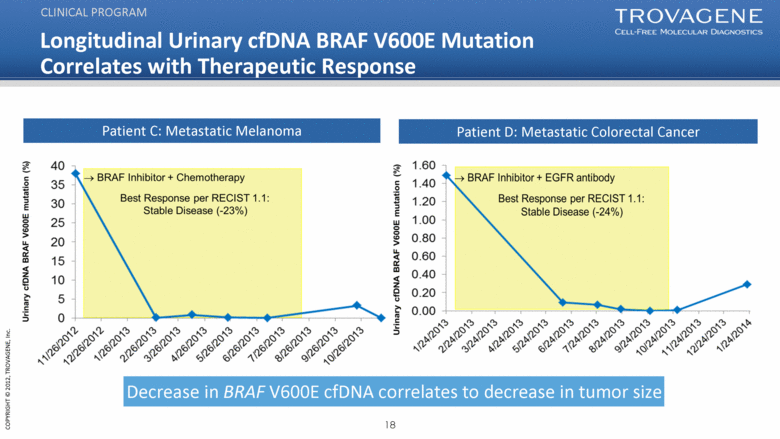
Patient D: Metastatic Colorectal Cancer Longitudinal Urinary cfDNA BRAF V600E Mutation Correlates with Therapeutic Response CLINICAL PROGRAM Patient C: Metastatic Melanoma Decrease in BRAF V600E cfDNA correlates to decrease in tumor size |
|
|
Data-Driven Commercialization Program To Begin in late 2014 Clinical Data Presentations AACR 2013/14 ASCO 2014 Published Peer-Reviewed Manuscripts Oncotarget May 2014 Publication in Second Journal in 2014 Possible Publication in Third Journal in 2014 Expand Sales & Marketing Headcount & Programs Broad Oncogene Mutation Coverage & Tumor Types Melanoma – BRAF V600E (available in CLIA lab) Colorectal Cancer – KRAS 7-Mutation (available) Pancreatic Cancer – KRAS 7-Mutation (available) Lung Cancer – T790M (expected in 4Q 2014) Collaborate with Integrated Healthcare Networks & NCCN Cancer Centers to Integrate PCM Technology Into Cancer Management Strategies COMMERCIALIZATION PROGRAM |
|
|
STAGE 3 Obtain Reimbursement Value Commensurate With Clinical Performance Concordance with standard of care (qualitative mutational status) Tier 1 Molecular Diagnostic codes with defined clinical utility and reimbursement values Press for value with payers using appeals process Early adoption within niche markets Novel clinical utility claims with ability to quantify mutational status “NOC“ (Not Otherwise Classified) codes Supported by sufficient clinical evidence of novel medical utility to successfully pass technology assessments by payers Broader adoption at key academic centers and community networks STAGE 2 STAGE 1 Demonstrated improvement in clinical outcomes & health economic benefits Apply to AMA for unique CPT code Supported by robust clinical adoption and appropriate valuation by payers based on evidence of significant clinical value Integration into cancer care (NCCN) guidelines and standard of care COMMERCIALIZATION PROGRAM Appeal *”Usual and Customary” designation by payer **May include patient financial responsibility. |
|
|
Completed assays will be transferred to Trovagene’s CLIA laboratory in 2014 2014 Development Program For Cancer Monitoring Applications ASSAY DEVELOPMENT PROGRAM EGFR L858R BRAF Quant HER-2 |
|
|
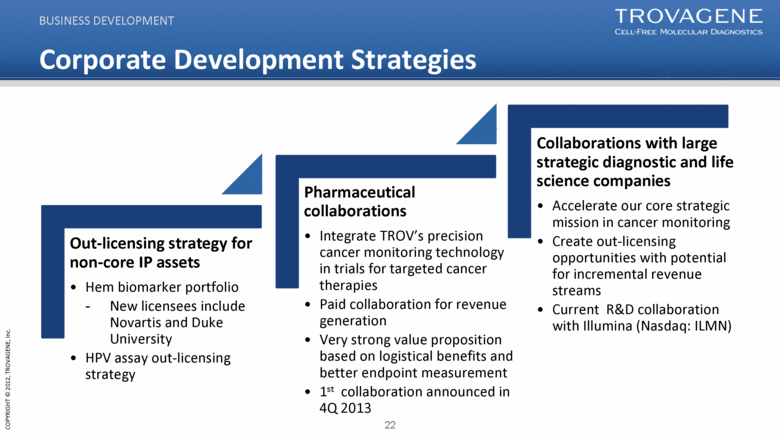
Corporate Development Strategies Collaborations with large strategic diagnostic and life science companies Accelerate our core strategic mission in cancer monitoring Create out-licensing opportunities with potential for incremental revenue streams Current R&D collaboration with Illumina (Nasdaq: ILMN) Pharmaceutical collaborations Integrate TROV’s precision cancer monitoring technology in trials for targeted cancer therapies Paid collaboration for revenue generation Very strong value proposition based on logistical benefits and better endpoint measurement 1st collaboration announced in 4Q 2013 Out-licensing strategy for non-core IP assets Hem biomarker portfolio New licensees include Novartis and Duke University HPV assay out-licensing strategy BUSINESS DEVELOPMENT |
|
|
2014 Goals & Expected Milestones Conduct additional clinical studies at major oncology centers and through collaborations with integrated healthcare networks Develop a health economic model supporting appropriate value and reimbursement levels for Trovagene’s PCM technology Present and publish clinical results for studies using Trovagene’s PCM technology as they become available Complete CLIA development and release of additional urine-based assays for the detection and monitoring of multiple clinically actionable oncogene mutations across a broad range of cancer types Enter into additional R&D collaborations with pharmaceutical companies as a future revenue source Expand and enter into new partnerships with strategic diagnostic and life science companies CORPORATE SUMMARY |
|
|
Please contact: Stephen Zaniboni, CFO szaniboni@trovagene.com For further information Antonius Schuh, CEO aschuh@trovagene.com |