Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Integrity Applications, Inc. | zk1313771.htm |
Exhibit 99.1

1
Your track to health!...™
Avner Gal
President & CEO
October 29, 2013
(OTCQB: IGAP)
AMIZINO Fall 2013 Small Cap /
Micro Cap Investment Conference
Seattle, WA

This presentation includes forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995 and various provisions of the U.S. Securities Act
of 1933, as amended, and the U.S. Securities Exchange Act of 1934, as amended. All
statements, other than statements of historical facts, included in this presentation are
forward-looking statements. Forward-looking statements included in this presentation
address, among other things, our future product development activities, strategies and
timing of seeking regulatory approval to market our current product candidate. These
statements are based on certain historical trends, current conditions and expected future
developments as well as other factors the Company believes are appropriate in the
circumstances. In addition to statements which explicitly describe these risks and
uncertainties, readers are urged to consider statements labeled with the terms “expects”,
“anticipates” and other similar words and phrases to be uncertain and forward-looking. All
of the forward-looking statements made in this presentation are qualified by these
cautionary statements and there can be no assurance that the actual results anticipated by the
Company will be realized or, even if substantially realized, that they will have the expected
consequences to or effects on the Company or its business or operations. Whether actual
results will conform to the Company’s expectations and predictions is subject to a number
of risks and uncertainties that may cause actual results to differ materially. See the risks
described in our reports filed with the Securities and Exchange Commission,
including under the heading "Risk Factors" of our Annual Report on Form 10-K for the year
ended December 31, 2012.
Securities Litigation Reform Act of 1995 and various provisions of the U.S. Securities Act
of 1933, as amended, and the U.S. Securities Exchange Act of 1934, as amended. All
statements, other than statements of historical facts, included in this presentation are
forward-looking statements. Forward-looking statements included in this presentation
address, among other things, our future product development activities, strategies and
timing of seeking regulatory approval to market our current product candidate. These
statements are based on certain historical trends, current conditions and expected future
developments as well as other factors the Company believes are appropriate in the
circumstances. In addition to statements which explicitly describe these risks and
uncertainties, readers are urged to consider statements labeled with the terms “expects”,
“anticipates” and other similar words and phrases to be uncertain and forward-looking. All
of the forward-looking statements made in this presentation are qualified by these
cautionary statements and there can be no assurance that the actual results anticipated by the
Company will be realized or, even if substantially realized, that they will have the expected
consequences to or effects on the Company or its business or operations. Whether actual
results will conform to the Company’s expectations and predictions is subject to a number
of risks and uncertainties that may cause actual results to differ materially. See the risks
described in our reports filed with the Securities and Exchange Commission,
including under the heading "Risk Factors" of our Annual Report on Form 10-K for the year
ended December 31, 2012.
2
Disclaimer

3
About Integrity Applications (OTCQB: IGAP)
Developing a family of non-
invasive glucose monitors for
diabetics, to measure their
blood glucose level without
the pain of drawing blood,
and without incremental cost
per measurement
invasive glucose monitors for
diabetics, to measure their
blood glucose level without
the pain of drawing blood,
and without incremental cost
per measurement
R&D began in 2001 (Israel);
Initial prototype completed in late 2003;
Methodology patent received October 2005;
Incorporated in Delaware in 2010;
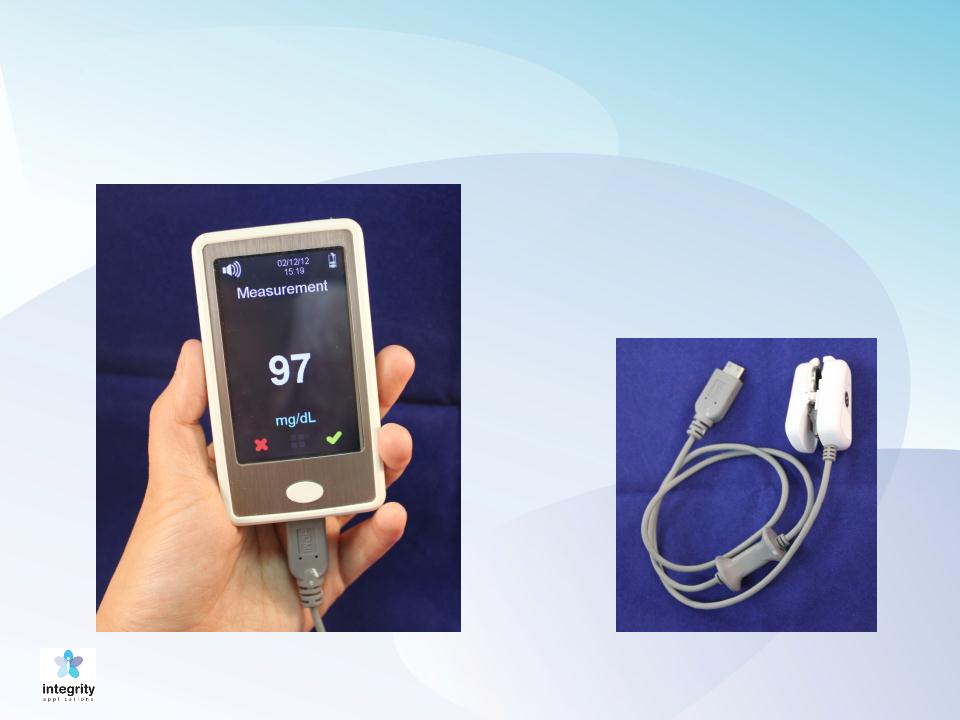
GlucoTrack® Model DF-F
4
Main Unit (MU)
Personal Ear Clip
(PEC)
(PEC)

Three independent (non-optical) technologies
Attempts (by others) to develop non-invasive glucose monitors have mostly
been based upon optical technologies, and have not been successful
been based upon optical technologies, and have not been successful
Patented combination of technologies (simultaneously):
ü Ultrasound
ü Electromagnetic (Conductivity)
ü Thermal (Heat Capacity)
Infrequent re-calibrations (*), by simple process
Current model, DF-F: Spot measurement device
5
GlucoTrack Unique Approach
ü Unique algorithms

GlucoTrack Features & Benefits
6
Easy to operate; User friendly (Just clip it! ™)
• Clear and simple instructions
• Color touch screen
• Large digits and Audible result within a minute
History graph and list of past 1000 readings
Average glucose level calculated displayed
Glucose average within desired points of time
Calculated HbA1c level (estimated)
Easy to download data to external storage device
Main Unit supports up to 3 users (individually calibrated)
Measurement alerts (pre-set by users)

“…Like an iPod!” (Prof. Steven Edelman)
Just Clip it! ™
7
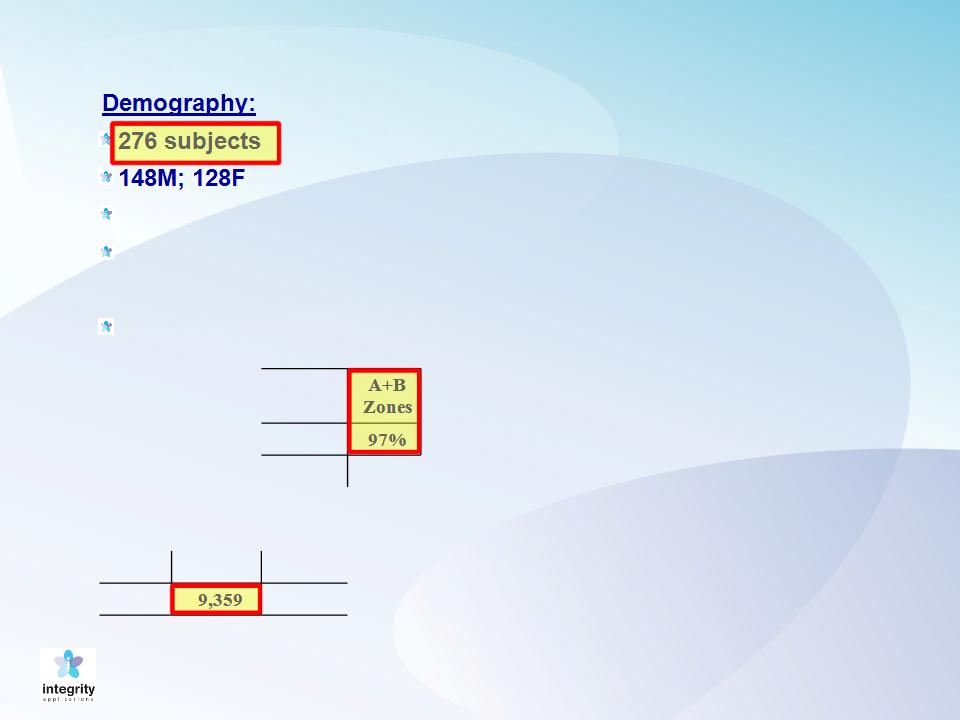
GlucoTrack Results
|
CEG
Zone |
Data
Points |
Percent
|
|
|
A
|
4,309
|
46%
|
|
|
B
|
4,730
|
51%
|
|
|
C
|
180
|
2%
|
|
|
D
|
135
|
1%
|
|
|
E
|
5
|
0%
|
|
|
Total
|
100%
|
|
36 T1DM; 240 T2DM
Body Mass Index (BMI):
33.1+/- 14.2 Kg/m2
33.1+/- 14.2 Kg/m2
Age: 49.5 +/- 31.5 years
8
Clinical trials, conducted by
third party, covered virtually
the entire demography,
including skin colors and
levels of education.
third party, covered virtually
the entire demography,
including skin colors and
levels of education.
97% of the results fall into the
clinically acceptable A&B
zones of the Clarke Error
Grid (CEG).
clinically acceptable A&B
zones of the Clarke Error
Grid (CEG).
GlucoTrack model DF-F has CE Mark approval
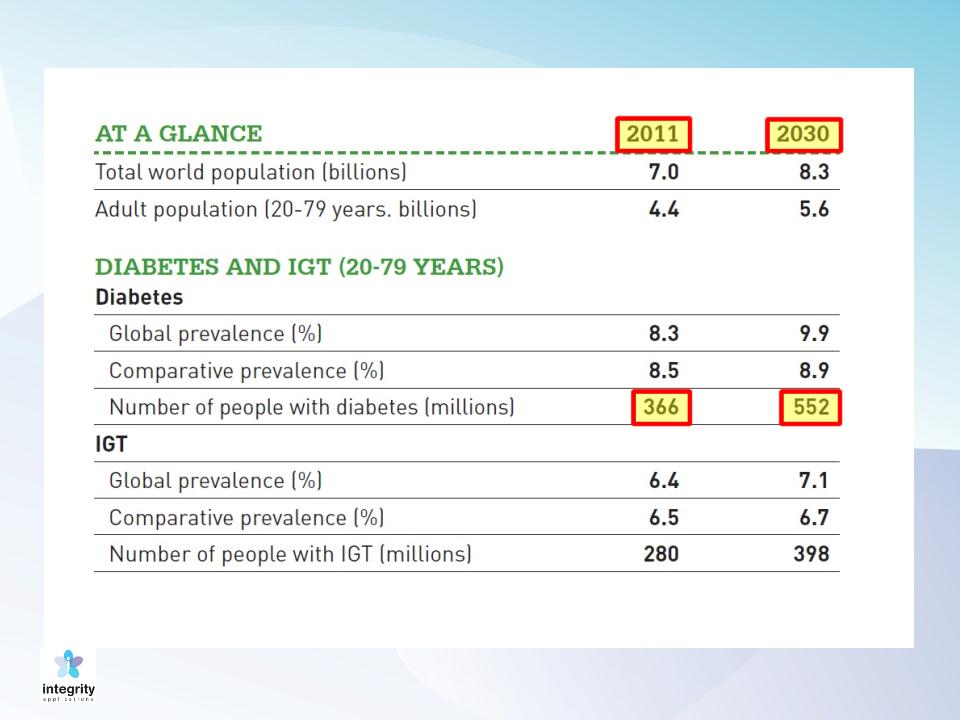
9
Prevalence of Diabetes: 20-79 Years Old
Source: IDF Diabetes Atlas, 5th Edition, 2011
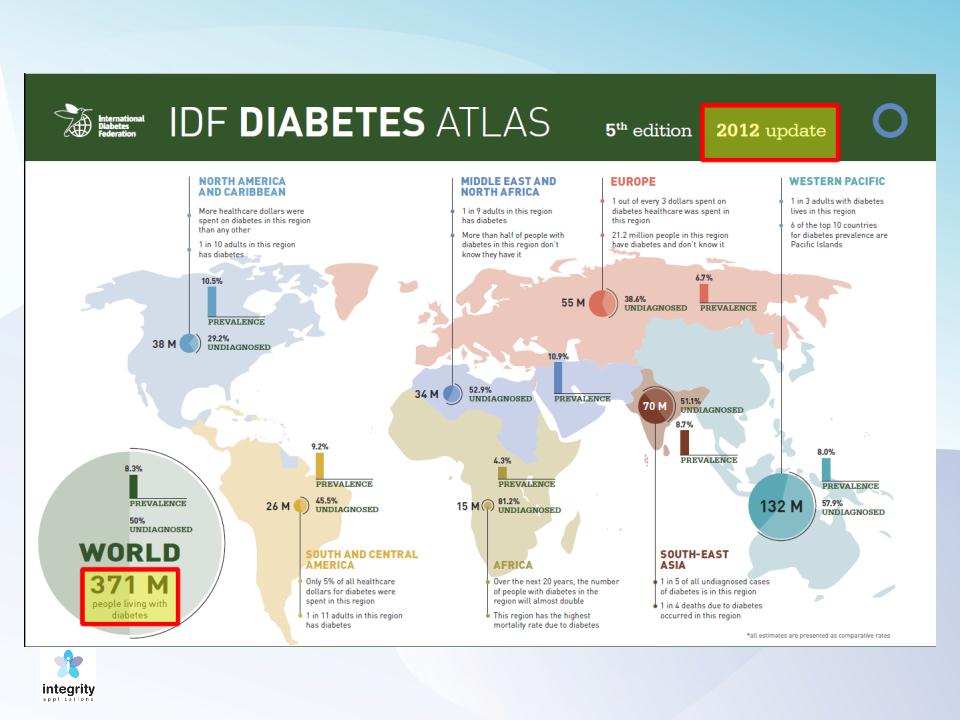
Prevalence of Diabetes: 20-79 Years Old (ctnd.)
10
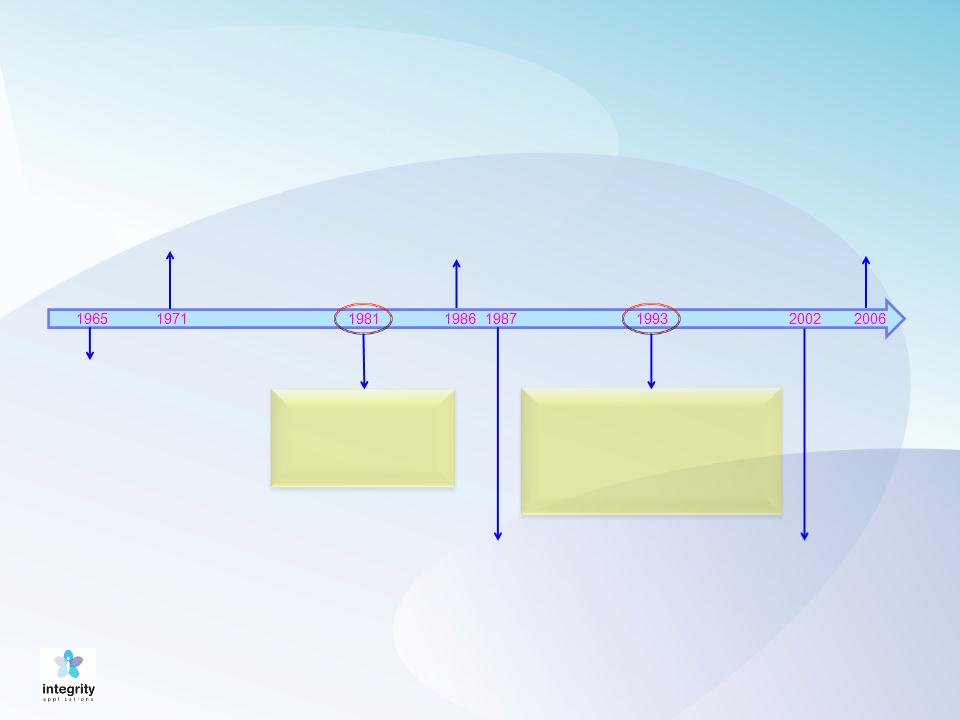
Advances in Blood Glucose Monitoring
11
A strip of paper
changes its
color according
to the patient’s
glucose level
changes its
color according
to the patient’s
glucose level
Meter interprets more
accurately the light
reflected by a color on
the meter into a
readable number
accurately the light
reflected by a color on
the meter into a
readable number
Glucometer (Bayer)
becomes the first
home glucose
monitoring device
becomes the first
home glucose
monitoring device
FDA, CDC and
ADA discuss
SMBG: key tool in
diabetes treatment
ADA discuss
SMBG: key tool in
diabetes treatment
Industry begins to
produce lower-cost
home glucose meters,
allowing better access to
more patients
produce lower-cost
home glucose meters,
allowing better access to
more patients
Study shows that patients
who conduct SMBG reduce
chances for complications
(76% for eye disease, 50% for
kidney disease and 60% for
nerve disease)
who conduct SMBG reduce
chances for complications
(76% for eye disease, 50% for
kidney disease and 60% for
nerve disease)
First continuous
glucose
monitoring via a
device implanted
under the skin is
released
glucose
monitoring via a
device implanted
under the skin is
released
Cygnus introduces
GlucoWatch. Causes
skin irritation, thus
cannot be called “non-
invasive”
GlucoWatch. Causes
skin irritation, thus
cannot be called “non-
invasive”

Advances in Blood Glucose Monitoring
12
GlucoTrack model DF-F,
a truly non-invasive
glucose monitor, has
gained CE Mark approval
glucose monitor, has
gained CE Mark approval
June 2013

Importance of SMBG is Recognized!
13
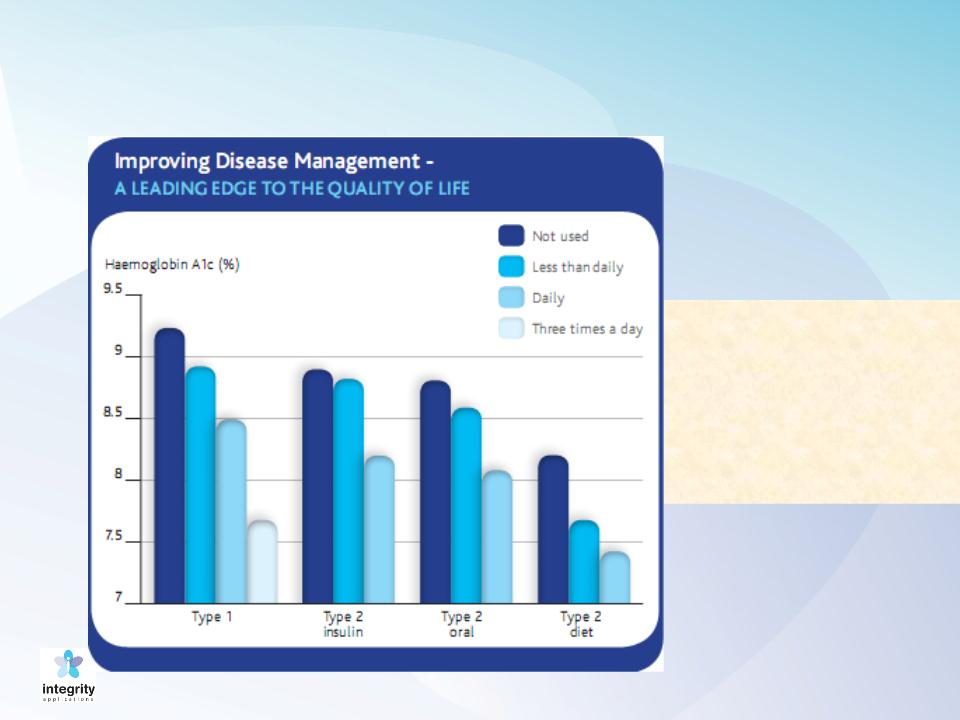
HbA1c as a Function of SMBG
HbA1c as a function of SMBG measurements per day (Source: Diabetes Atlas, 2nd edition, 2003)
14
ADA (June 2012):
Reducing HbA1c a
Little Less than 1
Point Reduces CV
Risk by 45%
Reducing HbA1c a
Little Less than 1
Point Reduces CV
Risk by 45%

Yet Significant Barriers Exist
Prick the fingertip (ouch…)
Draw blood, infuse drop onto a test strip
Painful; Expensive
15
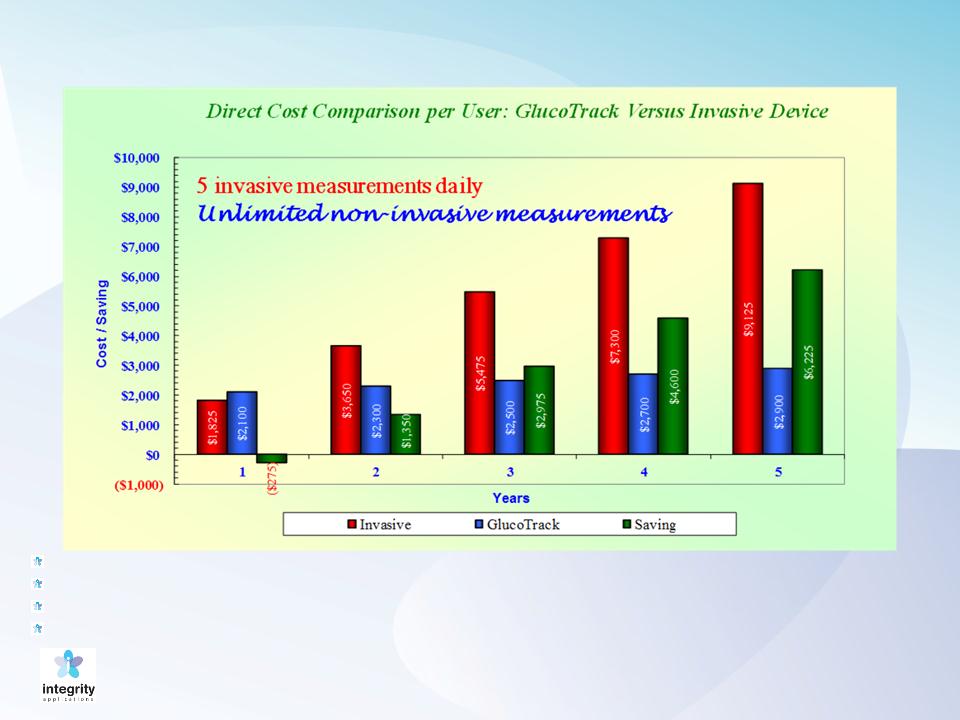
16
Compelling Economics
Assumptions:
Invasive measurement costs $1.00 each;
Retail price for GlucoTrack DF-F: $2,000 (including one PEC);
PEC costs $100; replaced every 6 months;
Excludes re-calibration costs, which are expected to be approximately $70, in total, over a five year period.
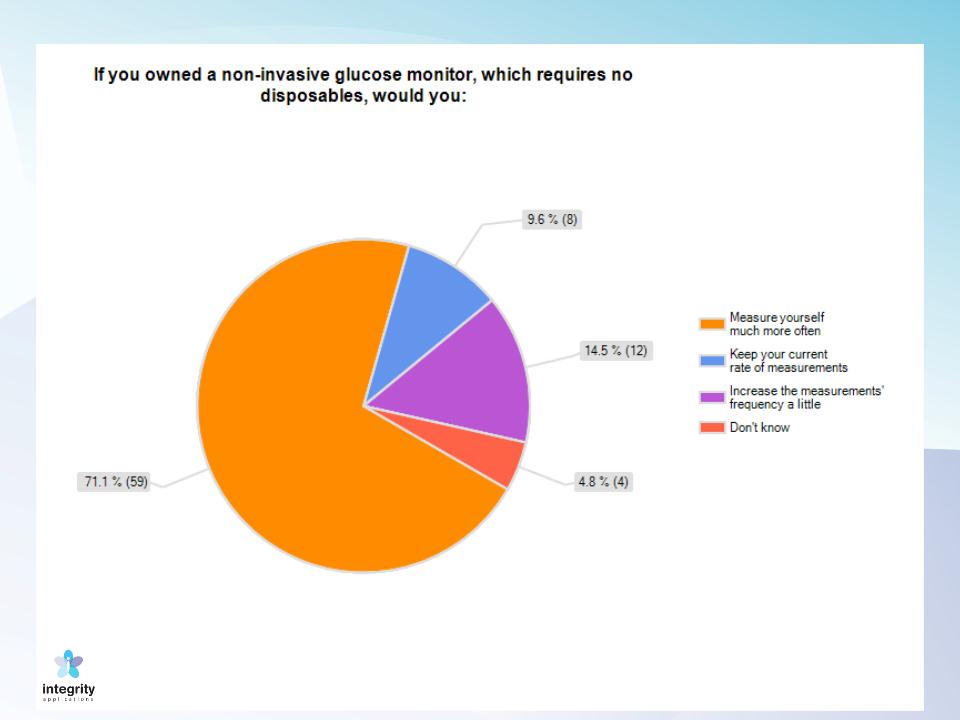
17
Survey results: T1DM & T2DM
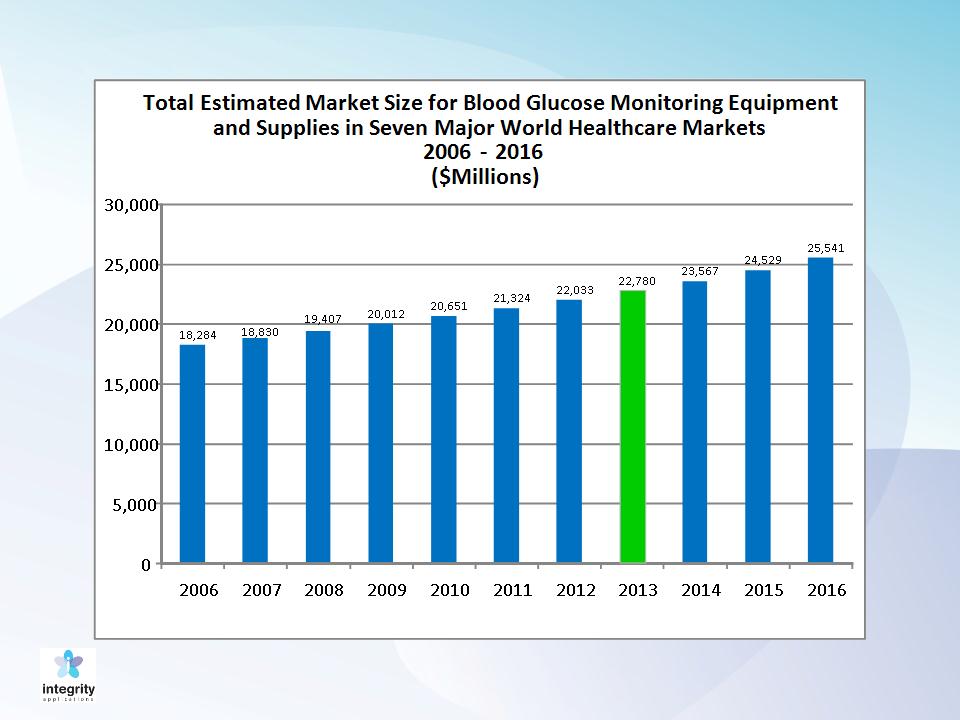
Market volume
18
Source:
Kalorama Information Diabetes and Diabetic Complications March 2007
Self Monitoring of Blood Glucose Market was estimated as $8B In 2010
Source: Pharmalive.com, May 2010
Source: Pharmalive.com, May 2010

Recent Achievements; Anticipated Actions
Improving the device
ü Smaller version of Personal Ear Clip (PEC) - enables use by people
with smaller ear lobes (awaiting clinical trials);
with smaller ear lobes (awaiting clinical trials);
ü PMCF: collecting data to enable algorithms’ improvement;
ü Embedding state of the art Anti-cloning & Encryption (H/W& S/W)
(implementation);
(implementation);
ü Using new connector to increase reliability of the PEC (Done);
ü Multi-lingual device (Hebrew; English; Russian; Arabic; French).
19

Recent Achievements; Anticipated Actions
Preparation for mass production manufacturing
ü Audited potential manufacturers (Israel, China, Taiwan,
Philippines);
Philippines);
ü Finalization of Main Unit (MU) mold (E/O December);
ü Design & build tools & testers for MU (E/O December);
ü Finalization of PEC mold (E/O November);
ü Improving assembling process of PEC sensors (E/O December);
ü Development of Self Test Aid (STA) (E/O November);
ü Functional testers & tools for PEC electrical & mechanical parts
(E/O December).
(E/O December).
20

Recent Achievements; Anticipated Actions
Marketing & Sales Activities
ü Booth in EASD Conference (September);
ü Table in Medica, Dusseldorf, Germany (November);
ü On going participation in conferences (posters; presentations);
ü Launched a redesigned website (October);
ü Meetings with Key Opinion Leaders in selected markets;
ü Discussions re: Distribution agreements (Europe; Middle East;
Asia; South East Asia; South America);
Asia; South East Asia; South America);
ü Training Field Engineers.
21

Recent Achievements; Anticipated Actions
Regulatory & Other Activities
ü Clinical pilot in South Africa (November);
ü Clinical trials for minors, 6-18 years old (E/O Q1/14);
ü Submission of 6 months trial’s results (Approval expected E/O Feb.);
ü Preparation for FDA clinical pathway (with NAMSA);
ü Development of DF-B Model (for developing countries);
ü Development of Web application (uploading data; analysis; links to
distributors websites; sales capabilities);
distributors websites; sales capabilities);
ü IP enhancing (patents applications);
ü Study regulatory pathways and requirements:
§ China CFDA; Taiwan TFDA; other countries;
§ Discussions; Workshops.
22

Products Road Map
DF-B: Basic model for use in developing countries
DF-C: Continuous monitoring of glucose level
DF-N: Provides Night time advanced warning of
hypoglycemic episodes
hypoglycemic episodes
DF-D: Warns Drivers of low glucose level
DF-I: System for pediatric Incubators
DF-P: Device for (IGT) Pre-diabetic patients
23

Competitive Landscape
|
#
|
Company
|
Product
|
Technology
|
Calibration
|
Measurement
|
Picture
|
Comments
|
|
1
|
Echo Therapeutics
(MA, USA) |
Symphony
|
Ultra Sound
(ISF)
|
12 hours
between calibrations
|
Continuous
|
|
For ICU;
In clinical
trials |
|
2
|
Grove Instruments
(MA, USA)
|
Gi-200
|
Optical Bridge
|
No data
|
Spot
|
|
In clinical
trials |
|
3
|
Cnoga Medical
(Israel)
|
TensorTip
CGM - Combo |
Optical; Bio-
parameters; Look-up table |
Long process
Many (>200) points |
Spot
|
|
CE Mark
Approved; “Selling”
|
|
4
|
Freedom Meditech
(CA, USA) |
I Sugar X
|
Optical (in front
of the eye) |
No data
|
Spot
|
Diagnostics
Convert to
monitoring |
|
|
5
|
AiMedics
(Australia)
|
HypoMon
|
Skin
Bio-sensors
|
No data
|
Continuous
|
|
Received CE
-Mark; Like DF-N
|
|
6
|
Cybiocare Optical
(Quebec, Canada) |
OHD
|
Optical
|
No data
|
Continuous
|
Like DF-N
|
24

Management
Avner Gal: President & CEO, Chairman (Co-Founder)
Retired Naval Commander, strong Engineering & Management background
David Malka: Executive VP & COO (Co-Founder)
Rich experience in Operations, including founding Operation Departments
Kobi Bar-Shalom: CFO
Rich experience in different size private & public companies
Eugene Naidis: Project Manager
Rich experience in Research & Development projects
Avner Avrahami: QA Manager
Long term experience in QA activities
25

Medical Advisory Board
26
Dr. Ilana Harman-Bohem, MD
Manager of Diabetic department at Soroka University Medical Center
Prof. Yariv Malimovka, MD
Senior consultant to Spanish medical centers in blood vessels & surgery
Adi Ickowicz / MedicSense (Regulatory affairs)
Rich experience in medical devices arena (FDA & CE Mark)
Adi Gluzberg / MedicSense (Regulatory affairs)
Rich experience in medical devices arena (FDA & CE Mark)

Capitalization
27
Approximately 5.14 million shares of common stock
outstanding (October 7, 2013);
outstanding (October 7, 2013);
Approximately 472,000 shares of the Company’s
common stock are issuable upon exercise of
outstanding stock options;
common stock are issuable upon exercise of
outstanding stock options;
Approximately 1.57 million shares of common stock
are issuable upon exercise of outstanding warrants;
are issuable upon exercise of outstanding warrants;
Approximately 1.48 million shares of common stock
are issuable upon conversion of outstanding shares
of convertible preferred stock.
are issuable upon conversion of outstanding shares
of convertible preferred stock.

28
Your track to health!...™
Thank You!
www.integrity-app.com
