Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - Paratek Pharmaceuticals, Inc. | d8k.htm |
Exhibit 99.1
| The Nature and Prevalence of Middle-of-the-Night Hypnotic Use |
Thomas Roth, Ph.D.1; Patricia Berglund, M.B.A. 2; Victoria Shahly, Ph.D.3; Alicia C. Shillington, Ph.D.4; Judith J. Stephenson, S.M.5; Denise Cooke6; Nikhilesh N. Singh, Ph.D.6; Ronald C. Kessler, Ph.D.8
1. Sleep Disorders and Research Center, Henry Ford Health System, Detroit, Michigan; 2. Institute for Social Research, University of Michigan, Ann Arbor, MI; 3. Department of Health Care Policy, Harvard Medical School, Boston, MA; 4. Epi-Q, Inc., Oak Brook, IL; 5. HealthCore, Inc., Wilmington, Delaware; 6. Transcept Pharmaceuticals, Inc., Pt. Richmond, CA; 7. Department of Health Care Policy, Harvard Medical School, Boston, MA
| Abstract |
Background: Middle-of-the-night (MOTN) awakening with difficulty returning to sleep is common and associated with significant adverse consequences.1-3 Therefore, a study was undertaken to assess the prevalence and nature of off-label MOTN hypnotic use.
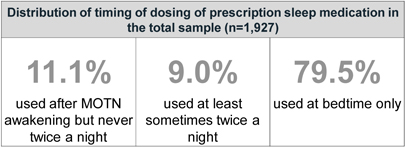
Methodology: 1,927 subjects, ages 18-64yrs, who received a hypnotic prescription in the past year, were sampled from 120,000 eligible members of a large commercial health plan (>34 million lives). Respondents with a history of MOTN hypnotic use who reported never using a hypnotic twice in the same night (n=209) plus a weighted sample of at-bedtime only users (n=303) were studied further.
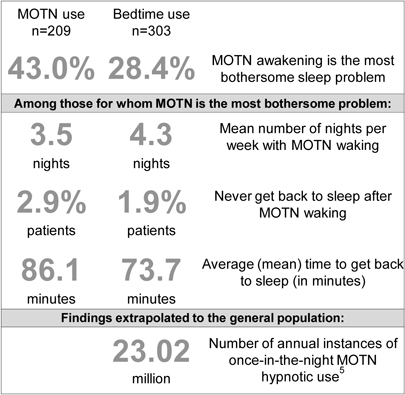
Results: 20.1% reported using bedtime hypnotics MOTN at least some of the time – 11.5% never used hypnotics twice in the same night and 9.0% did – and 79.5% used them at bedtime only. Of those studied further, 43.0% of MOTN and 28.0% of bedtime users reported MOTN insomnia as their biggest sleep problem. Of those reporting MOTN insomnia as their biggest sleep problem, 51.5% reported MOTN use. 81.7% used hypnotics MOTN on their own initiative, 12.1% under doctors’ direction, and 5.2% were not sure. 69.5% of MOTN users had a MOTN dosing rule involving when to take it: average MOTN use time was 6 hours before having to arise. Hypnotics taken MOTN and at-bedtime were essentially the same.
Conclusion: Despite there being no hypnotic approved for MOTN use, a number of patients with a prescription hypnotic use it off label in the middle of the night.
| Introduction |
| • | Epidemiology studies indicate that middle-of-the-night (MOTN) awakening with difficulty resuming sleep affects 15% of the general U.S. adult population.1-3 |
| • | Patients with MOTN awakening have significantly less total sleep time and suffer greater daytime impairment relative to patients with other insomnia symptoms.2 |
| • | Risk factors for MOTN insomnia include increasing age, psychiatric disorders and pain conditions.2 In addition, MOTN insomnia is more frequent in women than men.2 |
| • | All currently approved hypnotics are for bedtime use. |
| • | 14% of MOTN insomnia patients have been instructed by their physicians to dose at the time of MOTN awakening.4 |
| • | Therefore, a study was undertaken to assess the prevalence and nature of off-label MOTN hypnotic use. |
| Objectives |
The objectives of this survey were to measure:
| 1. | The prevalence of people taking a prescription hypnotic in the middle of the night (MOTN), and |
| 2. | The prevalence of people taking prescription hypnotics in the middle of the night who have MOTN awakenings and trouble resuming sleep. |
| Methodology |
From 120,000 eligible members of a large commercial health benefit organization, 23,616 were screened to yield 1,927 subjects, ages 18-64, who received a hypnotic prescription in the past year and completed the survey. Respondents with a history of MOTN hypnotic use who reported never using a hypnotic twice in the same night (n=209) plus a statistically weighted sample of at-bedtime only users (n=303) were studied further.
| Study population |
From the HealthCore Integrated Research Database (HIRDTM) of claims covering 34 million lives, approximately 120,000 Americans were sampled based upon the use of prescription hypnotic medication, inclusion and exclusion criteria.
| • | > 18 years of age at the time of sampling* |
| • | Current health plan member |
| • | At least 1 fill of an FDA approved sleep medication within the last year |
| * | Subjects 65+ years were not called because the sampling was too small and therefore not representative. |
| Results |
| 11.1% of patients with hypnotic Rx have used the hypnotic once after MOTN awakening at least once in the past year |
| MOTN awakening frequently reported as the most bothersome symptom by patients who use hypnotics. |
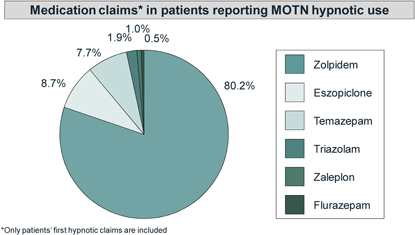
| Zolpidem is the most frequently used hypnotic after MOTN awakening. |
| Occasional once-nightly MOTN use frequent among those reporting MOTN awakening as the most bothersome symptom (n=178) |

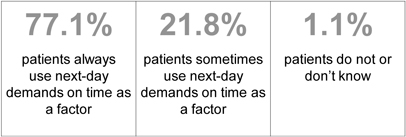
| Most MOTN hypnotic users use next-day demands on time as a factor for MOTN dosing (n=213) |
| Majority of patients who took hypnotic only after MOTN awakening did so on own volition (n=213) |

| Most patients who took hypnotic after MOTN awakening on own had a rule about hypnotic dosing after MOTN awakening (n=214) |
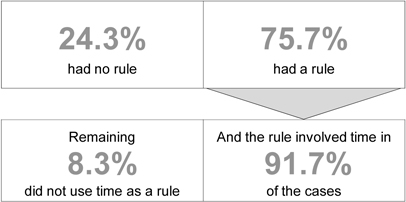
| Average time between hypnotic use after MOTN awakening and time to get up the next morning (n=213) |

| Conclusion |
Although hypnotics are not approved for MOTN use, a number of patients use prescription hypnotics off label to treat middle of the night insomnia.
References: 1. Ohayon MM. Difficulty in resuming or inability to resume sleep and the links to daytime impairment: definition, prevalence and comorbidity. J Psychiatr Res. 2009;43(10):934-940. 2. Ohayon MM. Nocturnal awakenings and comorbid disorders in the American general population. J Psychiatric Res. 2009;43:48-54. 3. Ohayon MM, Krystal A, Roehrs TA, Roth T, Vitiello MV. Using difficulty resuming sleep to define nocturnal awakenings. Sleep Med. 2010;11(3):236-241. 4. Roth T, Singh NN, Cooke D. Treatment of Middle of the Night Insomnia: Current Therapeutic Approaches. Poster presented at: 10th International Forum on Mood and Anxiety Disorders; November 2010; Vienna, Austria. 5. Kessler RC, Shillington A, Stephenson JJ, Weindorf M, Roth T. Middle of the night (MOTN) hypnotic use among insured Americans with hypnotic prescriptions. Study Report 70-1029-001, 30 November 2010.
Acknowledgement: Funded by Transcept Pharmaceuticals, Inc., Pt. Richmond, CA, USA
