Attached files
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended: December 31, 2010
Or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 001-35060
PACIRA PHARMACEUTICALS, INC.
(Exact Name of Registrant as Specified in Its Charter)
| Delaware | 51-0619477 | |
| (State or Other Jurisdiction of Incorporation or Organization) |
(I.R.S. Employer Identification No.) |
5 Sylvan Way, Suite 125
Parsippany, New Jersey 070504
(Address of Principal Executive Offices) (zip code)
Registrant’s telephone number, including area code (973) 254-3560
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Name of each exchange on which registered | |
| Common Stock, $0.001 par value | The NASDAQ Global Market |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ¨ No x
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ¨ No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See definitions of “large accelerated filer,” “accelerated filer,” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ¨ | Accelerated filer | ¨ | |||
| Non-accelerated filer | x (Do not check if a smaller reporting company) | Smaller reporting company | ¨ | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of voting Common Stock held by non-affiliates of the registrant as of February 8, 2011, the date of the closing of the registrant’s initial public offering, was $36,871,827, computed by reference to the last reported sale price of the registrant’s Common Stock on the NASDAQ Global Market on that date.
The number of shares outstanding of the registrant’s Common Stock as of February 28, 2011: 17,232,875
Documents incorporated by reference:
None.
Table of Contents
| Page No. | ||||||
| 1 | ||||||
| Item 1. |
1 | |||||
| Item 1A. |
27 | |||||
| Item 1B. |
54 | |||||
| Item 2. |
54 | |||||
| Item 3. |
55 | |||||
| Item 4. |
55 | |||||
| 56 | ||||||
| Item 5. |
56 | |||||
| Item 6. |
59 | |||||
| Item 7. |
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
61 | ||||
| Item 7A. |
78 | |||||
| Item 8. |
78 | |||||
| Item 9. |
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
78 | ||||
| Item 9A. |
78 | |||||
| Item 9B. |
79 | |||||
| 80 | ||||||
| Item 10. |
80 | |||||
| Item 11. |
85 | |||||
| Item 12. |
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
100 | ||||
| Item 13. |
Certain Relationships and Related Transactions, and Director Independence |
103 | ||||
| Item 14. |
108 | |||||
| 110 | ||||||
| Item 15. |
110 | |||||
i
Table of Contents
References
Pacira Pharmaceuticals, Inc. is the holding company for our California operating subsidiary of the same name, which we refer to as PPI-California. In March 2007, we acquired PPI-California from SkyePharma Holding, Inc. (referred to in this Annual Report on Form 10-K as the “Acquisition”). Unless the context requires otherwise, references to “Pacira,” “we,” the “company,” “us” and “our” in this Annual Report on Form 10-K refers to Pacira Pharmaceuticals, Inc., and its subsidiaries. In addition, references in this Annual Report on Form 10-K to DepoCyt(e) mean DepoCyt when discussed in the context of the United States and Canada and DepoCyte when discussed in the context of Europe.
Forward-Looking Statements
This Annual Report on Form 10-K and certain other communications made by us contain forward-looking statements within the meaning of Section 21E of the Securities Exchange of 1934 (the “Exchange Act”), including statements about our growth and future operating results, discovery and development of products, strategic alliances and intellectual property. For this purpose, any statement that is not a statement of historical fact should be considered a forward-looking statement. We often use the words “believe,” “anticipate,” “plan,” “expect,” “intend,” “may,” “will” and similar expressions to help identify forward-looking statements. We cannot assure you that our assumptions and expectations will prove to have been correct. These forward-looking statements include, among others, statements about: the company’s plans to develop and commercialize EXPAREL; the Company’s plans to continue to manufacture and provide support services for its commercial partners who have licensed DepoCyt(e) and DepoDur; the timing of, and the Company’s ability to obtain, regulatory approval of EXPAREL; the timing of the Company’s anticipated commercial launch of EXPAREL; the rate and degree of market acceptance of EXPAREL; the size and growth of the potential markets for EXPAREL and the Company’s ability to serve those markets; the Company’s plans to expand the indications of EXPAREL to include nerve block and epidural administration; and our commercialization and marketing capabilities. Important factors could cause our actual results to differ materially from those indicated or implied by forward-looking statements, including those discussed below. We undertake no intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise and readers should not rely on the forward looking statements as representing the company’s views as of any date subsequent to the date of the filing of this Annual Report on Form 10-K.
| Item 1. | Business |
Overview
We are an emerging specialty pharmaceutical company focused on the development, commercialization and manufacture of proprietary pharmaceutical products, based on our proprietary DepoFoam drug delivery technology, for use in hospitals and ambulatory surgery centers. We filed a New Drug Application, or NDA, for our lead product candidate, EXPAREL, a long-acting bupivacaine (anesthetic/analgesic) product for postsurgical pain management with the United States Food and Drug Administration, or FDA, which was accepted by the FDA for review on December 10, 2010. Our clinical data demonstrates that EXPAREL provides analgesia for up to 72 hours post-surgery, compared with seven hours or less for bupivacaine. We believe EXPAREL will address a significant unmet medical need for a long-acting non-opioid postsurgical analgesic, resulting in simplified postsurgical pain management and reduced opioid consumption, leading to improved patient outcomes and enhanced hospital economics. We estimate there are approximately 39 million opportunities annually in the United States for EXPAREL to be used. EXPAREL will be launched by certain members of our management team who have successfully launched multiple products in the hospital market.
EXPAREL consists of bupivacaine encapsulated in DepoFoam, both of which are used in FDA-approved products. DepoFoam, our extended release drug delivery technology, is the basis for our two FDA-approved commercial products: DepoCyt(e) and DepoDur, which we manufacture for our commercial partners. DepoFoam-based products have been manufactured for over a decade and have an extensive safety record and regulatory approvals in the United States, European countries and other territories. Bupivacaine, a well-characterized, FDA-approved anesthetic/analgesic, has an established safety profile and over 20 years of use in the United States.
1
Table of Contents
EXPAREL has demonstrated efficacy and safety in two multicenter, randomized, double-blind, placebo-controlled, pivotal Phase 3 clinical trials in patients undergoing soft tissue surgery (hemorrhoidectomy) and orthopedic surgery (bunionectomy). Overall, EXPAREL has demonstrated safety in over 1,300 subjects. In September 2010, we filed an NDA for EXPAREL with the FDA, using a 505(b)(2) application. We are initially seeking approval for postsurgical analgesia by local administration into the surgical wound, or infiltration, a procedure commonly employing bupivacaine. Under the Prescription Drug User Fee Act, or PDUFA, guidelines, the FDA has a goal of ten months from the date of NDA filing to make a decision regarding the approval of our filing. The PDUFA goal date for our NDA is July 28, 2011. We also plan to expand the indications of EXPAREL to include nerve block and epidural administration, markets where bupivacaine is also used routinely.
Our current product portfolio and product candidate pipeline is summarized in the table below:
| Product(s)/ |
Primary Indication(s) |
Status |
Commercialization Rights | |||
| EXPAREL |
Postsurgical analgesia by infiltration | PDUFA goal date: July 28, 2011 |
Pacira (worldwide) | |||
| Postsurgical analgesia by nerve block | Phase 2/3 (planning | Pacira (worldwide) | ||||
| Postsurgical analgesia by epidural administration | Phase 1 (completed) | Pacira (worldwide) | ||||
| DepoCyt(e) |
Lymphomatous meningitis | Marketed | Sigma-Tau Pharmaceuticals Mundipharma International | |||
| DepoDur |
Post-operative pain | Marketed | EKR Therapeutics Flynn Pharmaceuticals | |||
| DepoNSAID |
Acute pain | Preclinical | Pacira (worldwide) | |||
| DepoMethotrexate |
Rheumatoid arthritis Oncology | Preclinical | Pacira (worldwide) Pacira (worldwide) | |||
Our Strategy
Our goal is to be a leading specialty pharmaceutical company focused on the development, commercialization and manufacture of proprietary pharmaceutical products principally for use in hospitals and ambulatory surgery centers. We plan to achieve this by:
| • | obtaining FDA approval for EXPAREL in the United States for postsurgical analgesia by local infiltration; |
| • | building a streamlined commercial organization concentrating on major hospitals and ambulatory surgery centers in the United States and targeting surgeons, anesthesiologists, pharmacists and nurses; |
| • | working directly with managed care payers, quality improvement organizations, key opinion leaders, or KOLs, in the field of postsurgical pain management and leading influence hospitals with registry programs to demonstrate the economic benefits of EXPAREL; |
| • | securing commercial partnerships for EXPAREL in regions outside of the United States; |
| • | obtaining FDA approval for nerve block and epidural administration indications for EXPAREL; |
2
Table of Contents
| • | manufacturing all our DepoFoam-based products, including EXPAREL, DepoCyt(e) and DepoDur, in our current Good Manufacturing Practices, or cGMP, compliant facilities; and |
| • | continuing to expand our marketed product portfolio through development of additional DepoFoam-based hospital products utilizing a 505(b)(2) strategy, which permits us to rely upon the FDA’s previous findings of safety and effectiveness for an approved product. A 505(b)(2) strategy may not succeed if there are successful challenges to the FDA’s interpretation of Section 505(b)(2). |
Postsurgical Pain Market Overview
According to Thomson Reuters, roughly 45 million surgical procedures were performed in the United States during the twelve months ending in October 2007. We estimate there are approximately 39 million opportunities annually in the United States for EXPAREL to be used to improve patient outcomes and enhance hospital economics. Postsurgical pain is a response to tissue damage during surgery that stimulates peripheral nerves, which signal the brain to produce a sensory and psychological response. Numerous studies reveal that the incidence and severity of postsurgical pain is primarily determined by the type of surgery, duration of surgery and the pain treatment choice following surgery. Postsurgical pain is usually greatest the first few days after the completion of a surgical procedure.
Limitations of Current Therapies for Postsurgical Pain
Substantially all surgical patients experience postsurgical pain, with approximately 50% reporting inadequate pain relief according to epidemiological studies. Unrelieved acute pain causes patient suffering and can lead to other health problems, which delays recovery from surgery and may result in higher healthcare costs. According to the Agency for Healthcare Research and Quality, aggressive prevention of pain is better than treatment of pain because, once established, pain is more difficult to suppress. Current multimodal therapy for postsurgical pain includes wound infiltration with local anesthetics combined with the systemic administration of opioid and non-steroidal anti-inflammatory drug, or NSAID, analgesics.
Local Analgesics
Treatment of postsurgical pain typically begins at the end of surgery, with local analgesics, such as bupivacaine, administered by local infiltration. Though this infiltration provides a base platform of postsurgical pain management for the patient, efficacy of conventional bupivacaine and other available local analgesics is limited, lasting seven hours or less. As local infiltration is not practical after the surgery is complete, and as surgical pain is greatest in the first few days after surgery, additional therapeutics are required to manage postsurgical pain.
Opioids
Opioids, such as morphine, are the mainstay of postsurgical pain management but are associated with a variety of unwanted and potentially severe side effects, leading healthcare practitioners to seek opioid-sparing strategies for their patients. Opioid side effects include sedation, nausea, vomiting, urinary retention, headache, itching, constipation, cognitive impairment, respiratory depression and death. Side effects from opioids have been demonstrated to reduce the patient’s quality of life and result in suboptimal pain relief. These side effects may require additional medications or treatments and prolong a patient’s stay in the post-anesthesia care unit and the hospital or ambulatory surgery center, thereby increasing costs significantly.
PCA and Elastomeric Bag Systems
Opioids are often administered intravenously through patient controlled analgesia, or PCA, systems in the immediate postsurgical period. The total cost of PCA postsurgical pain management for three days can be up to $500, not including the costs of treating opioid complications. In an attempt to reduce opioid usage, many hospitals employ elastomeric bag systems designed to deliver bupivacaine to the surgical area through a catheter over a period of time. This effectively extends the duration of bupivacaine in the postsurgical site but has significant shortcomings.
3
Table of Contents
PCA systems and elastomeric bag systems are clumsy and difficult to use, which may delay patient ambulation and introduce catheter-related issues, including infection. In addition, PCA systems and elastomeric bags require significant hospital resources to implement and monitor.
NSAIDs
NSAIDs are considered to be useful alternatives to opioids for the relief of acute pain since they do not produce respiratory depression or constipation. Despite these advantages, the use of injectable NSAIDs, such as ketorolac and ibuprofen, is severely limited in the postsurgical period because they increase the risk of bleeding and gastrointestinal and renal complications.
Our Solution—EXPAREL
Based on our clinical trial data, EXPAREL provides continuous and extended postsurgical analgesia for up to 72 hours and reduces the consumption of supplemental opioid medications. We believe this will simplify postsurgical pain management, minimize breakthrough episodes of pain and result in improved patient outcomes and enhanced hospital economics.
Our EXPAREL strategy has four principal elements:
Replace the use of bupivacaine in postsurgical infiltration. We believe EXPAREL:
| • | extends postsurgical analgesia for up to 72 hours, from seven hours or less; |
| • | utilizes existing postsurgical infiltration administration techniques; |
| • | dilutes easily with saline to reach desired volume; |
| • | is a ready-to-use formulation; and |
| • | facilitates treatment of both small and large surgical wounds. |
Become the foundation of a postsurgical pain management regimen in order to reduce and delay opioid usage. We believe EXPAREL:
| • | significantly delays and reduces opioid usage while improving postsurgical pain management as demonstrated in our Phase 3 hemorrhoidectomy trial, in which EXPAREL demonstrated the following: |
| • | delayed first opioid usage to approximately 14 hours post-surgery, compared to approximately one hour for placebo; |
| • | significantly increased percentage of patients requiring no opioid rescue medication through 72 hours post-surgery, to 28% compared to 10% for placebo; |
| • | 45% less opioid usage at 72 hours post-surgery compared to placebo; and |
| • | increased percentage of patients who are pain free at 24 hours post-surgery compared to placebo; and |
| • | may reduce hospital cost and staff monitoring of PCA systems. |
Improve patient satisfaction. We believe EXPAREL:
| • | reduces the need for patients to be constrained by elastomeric bags and PCA systems, which are clumsy, difficult to use and may introduce catheter-related issues, including infection; |
| • | promotes maintenance of early postsurgical pain management, thereby reducing the time spent in the intensive care unit; and |
4
Table of Contents
| • | promotes early ambulation, which potentially reduces the risk of life-threatening blood clots, and allows quicker return of bowel function, thereby leading to a faster switch to oral nutrition and medicine, and thus a faster discharge from the hospital. |
Develop and seek approval of EXPAREL for nerve block and epidural administration. We believe these additional indications for EXPAREL:
| • | present a low-risk, low-cost opportunity for clinical development; and |
| • | will enable us to fully leverage our manufacturing and sales infrastructure. |
EXPAREL Development Program
EXPAREL has demonstrated efficacy and safety in two multicenter, randomized, double-blind, placebo-controlled, pivotal Phase 3 clinical trials in patients undergoing soft tissue surgery (hemorrhoidectomy) and orthopedic surgery (bunionectomy). At a pre-NDA meeting in February 2010, the FDA acknowledged that the two pivotal Phase 3 clinical trials conducted by us, in patients undergoing hemorrhoidectomy and bunionectomy surgeries, appeared to be appropriately designed to evaluate the safety and efficacy of EXPAREL. Both trials met their primary efficacy endpoints in demonstrating statistically significant analgesia through 72 hours for the hemorrhoidectomy trial and 24 hours for the bunionectomy trial. Both trials also met multiple secondary endpoints, including decreased opioid use and delayed time to first opioid use. These two pivotal Phase 3 clinical trials formed the basis of the evidence for efficacy in the NDA for EXPAREL.
The safety of EXPAREL has been demonstrated in 21 clinical trials consisting of nine Phase 1 trials, seven Phase 2 trials and five Phase 3 trials. EXPAREL was administered to over 1,300 human patients at doses ranging from 10 mg to 750 mg administered by local infiltration into the surgical wound, and by subcutaneous, perineural, epidural and intraarticular administration. In all 21 clinical trials, EXPAREL was well tolerated. The most common treatment emergent adverse events in the EXPAREL and placebo groups were nausea and vomiting and occurred with similar frequency across the EXPAREL and placebo groups. No signal of any of the central nervous system or cardiovascular system adverse events typically observed with high doses of bupivacaine has been observed with EXPAREL. We conducted two thorough QTc studies that demonstrated that EXPAREL did not cause significant QTc prolongation (a measure of cardiac safety mandated by the FDA for all new products) even at the highest dose evaluated. No events of destruction of articular cartilage, or chondrolysis, have been reported in any of the EXPAREL trials. EXPAREL did not require dose adjustment in patients with mild to moderate liver impairment.
Pivotal Phase 3 Clinical Trials
Hemorrhoidectomy. Our pivotal Phase 3 hemorrhoidectomy clinical trial was a multicenter, randomized, double-blind, placebo-controlled trial conducted in 189 patients at 14 sites in Europe. The study enrolled patients 18 years of age or older undergoing a two or three column excisional hemorrhoidectomy under general anesthesia using the Milligan-Morgan technique, a commonly used method for surgically removing hemorrhoids. We studied a 300 mg dose of EXPAREL with a primary endpoint of pain control for up to 72 hours with morphine rescue medication available to both trial groups. Additional endpoints included the proportion of pain-free patients, proportion of patients requiring opioid rescue medication, total opioid usage, time to first use of opioid rescue medication and patient satisfaction.
The 300 mg dose of EXPAREL provided a statistically significant 30% reduction in pain (p<0.0001), as measured by the area under the curve, or AUC, of the NRS-R pain scores at 72 hours and all additional time points measured up to 72 hours. The numeric rating scale at rest score, or the NRS-R, is a commonly used patient reported measurement of pain. Under the NRS-R, severity of pain is measured on a scale from 0 to 10, with 10 representing the worst possible pain. The AUC of the NRS-R pain score represents a sum of the patient’s pain measured at several time points using the NRS-R, from time of surgery to the specified endpoint. A lower number indicates less cumulative pain. The p-value is a measure of probability that the difference between the placebo group and the EXPAREL group is due to chance (e.g., p = 0.01 means that there is a 1% (0.01 = 1.0%) chance that the difference between the placebo group and EXPAREL group is the result of random chance as opposed to the EXPAREL treatment). A p-value less than or equal to 0.05 (0.05 = 5%) is commonly used as a criterion for statistical significance.
5
Table of Contents
Phase 3 Hemorrhoidectomy Clinical Trial: AUC of NRS-R Pain Intensity Scores from Initial Infiltration
Timepoint, EXPAREL Compared to Placebo
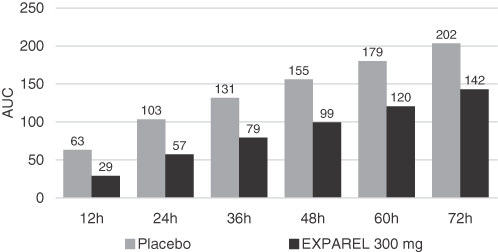
Note: Differences between study groups were statistically significant at 72 hours (p<0.0001), the primary endpoint, and all
additional time points measured (p<0.0001).
In secondary endpoints, EXPAREL demonstrated efficacy in reducing the use of opioid rescue medication, which was available to both the EXPAREL treatment group and the placebo treatment group. Approximately three times the number of patients in the EXPAREL treatment group avoided opioid rescue medication altogether, and patients in the EXPAREL treatment group showed 45% less opioid usage compared to the placebo treatment group at 72 hours. Opioid related secondary endpoints included:
| • | Total avoidance of opioid rescue medication. 28% of patients treated with EXPAREL received no postsurgical opioid rescue pain medication through 72 hours post-dose. By contrast only 10% of placebo treated patients avoided all opioid rescue medication through 72 hours, and this difference was statistically significant (p=0.0007); |
| • | Reduced total consumption of opioid rescue medication. The adjusted mean total postsurgical consumption of supplemental opioid pain medication was 45% lower in patients treated with EXPAREL compared to the placebo treatment group through 72 hours (p=0.0006) post-dose; and |
| • | Delayed use of opioid rescue medication. EXPAREL delayed the median time to first opioid use from approximately one hour in the placebo treatment group to approximately 14 hours in the EXPAREL treatment group and this difference was statistically significant (p<0.0001). At 14 hours post-surgery compared to one hour post-surgery, patients have substantially recovered from the effects of surgical anesthesia and are able to tolerate oral opioids and require less intensive monitoring. |
In addition to the reduced usage of opioids compared to patients receiving placebo, secondary endpoints also demonstrated that patients in the EXPAREL treatment group had higher satisfaction scores and more were pain free compared to those in the placebo treatment group.
| • | More pain free patients. A greater percentage of patients treated with EXPAREL were pain free compared to the placebo treatment group, and the difference reached statistical significance at all times up to and through 24 hours post-dose (p=0.0448); and |
6
Table of Contents
| • | Improved patient satisfaction. A greater percentage of patients treated with EXPAREL were “extremely satisfied” compared to the placebo treatment group, and the difference was statistically significant (p=0.0007) at 24 and 72 hours post-dose. |
We believe that this combination of reduced opioid usage and continuous and extended postsurgical pain management highlights the efficacy of EXPAREL and its ability to be used as a part of a multimodal, opioid sparing postsurgical pain management strategy.
Bunionectomy. Our pivotal Phase 3 bunionectomy clinical trial was a multicenter, randomized, double-blind, placebo-controlled trial conducted in 193 patients at four sites in the United States. The study enrolled patients 18 years of age or older undergoing a bunionectomy. We studied a 120 mg dose of EXPAREL with a primary endpoint of pain control at 24 hours, the critical period for postsurgical pain management in bunionectomy, with opioid rescue medication available to both trial groups. EXPAREL provided a statistically significant reduction in pain, as measured by the AUC of the NRS-R pain scores at 24 hours (p=0.0005). This reduction was also statistically significant at 36 hours.
EXPAREL also achieved statistical significance in secondary endpoints related to pain measurement and the use of opioid rescue medication, which was available to both patients in the EXPAREL treatment group and the placebo treatment group, including:
| • | Total avoidance of opioid rescue medication. The difference between treatment groups in the percentage of patients who received opioid rescue pain medication was statistically significant, favoring the group treated with EXPAREL compared to the placebo treatment group through 12 hours (p=0.0003) and 24 hours (p=0.0404); |
| • | Delayed use of opioid rescue medication. EXPAREL delayed the median time before first opioid use compared to the placebo treatment group and this difference was statistically significant (p<0.0001); and |
| • | More pain free patients. A statistically significant increase in the percentage of pain free patients was observed between treatment groups, favoring the group treated with EXPAREL compared to the placebo treatment group at 2 hours (p=0.0019), 4 hours (p=0.0002), 8 hours (p=0.0078) and 48 hours (p=0.0153) post-dose. The difference between groups was not statistically significant at 24 hours post-dose. |
Other Clinical Trials
In 2009, we completed two Phase 3 clinical trials comprising 223 patients who received EXPAREL, comparing them to patients who received bupivacaine in a multimodal setting where patients received additional concomitant analgesics. One of these Phase 3 clinical trials was for total knee arthroplasty and the other was for hemorrhoidectomy. Although EXPAREL performed as expected and continued to demonstrate its safety and tolerability, due to the unexpectedly positive results in the control arm, these trials did not meet their primary endpoint. The results of these studies influenced some of the inclusion and exclusion criteria and protocol specified measures used in our successful pivotal Phase 3 clinical trials described above.
Based on the outcome of these two trials, in 2009, we discontinued a Phase 3 clinical trial in breast augmentation early. At the time of discontinuation, we had only enrolled approximately half of the number of patients required to demonstrate statistical significance. EXPAREL demonstrated a positive trend and safety, but did not meet the primary efficacy endpoint. We have collected data on all patients for whom data was available and expect to publish this data in a peer reviewed medical journal.
We have completed seven Phase 2 clinical trials, five of which were in wound infiltration. A total of 452 patients received various doses of EXPAREL and/or bupivacaine in various surgical settings including hernia repair, total knee arthroplasty, hemorrhoidectomy, and breast augmentation. The data from these Phase 2 clinical trials guided the dose selection for our successful pivotal Phase 3 clinical trials, which formed the basis of our NDA.
7
Table of Contents
The EXPAREL wound infiltration program encompassed 21 dosing comparisons (a dose of EXPAREL compared to a control) throughout a total of ten clinical trials; nine of these were randomized parallel-group clinical trials, seven of which had a bupivacaine control and two of which had a placebo control. When a program-wide primary endpoint of the area under the curve of the numeric rating scale score for pain at rest from 0 through 72 hours was applied to the 19 doses in the randomized parallel-group clinical trials, 16 favored EXPAREL.
EXPAREL Health Economic Benefits
In addition to being efficacious and safe, we believe that EXPAREL provides health economic benefits that play an important role in formulary decision making and these health economic benefits are an often over-looked factor in planning for the commercial success of a pharmaceutical product. Several members of our management team have extensive experience applying health economic outcomes research to support the launch of successful commercial products. Our strategy is to work directly with managed care payers, quality improvement organizations, KOLs in the field of postsurgical pain management and leading influence hospitals with registry programs to demonstrate the economic benefits of EXPAREL.
EXPAREL is designed as a single postsurgical injection intended to replace the current use of clumsy and expensive PCA systems and elastomeric bag systems, reduce the consumption of opioids, and their related side effects, and reduce the length of stay in the hospital, all factors that negatively impact patient outcomes and hospital economics. For example, in our Phase 2 hemorrhoidectomy trial, 300 mg of EXPAREL reduced pain by 47%, as measured by the AUC of the NRS-R pain scores, with a 66% reduction in opioid consumption and a corresponding 89% reduction in opioid related adverse events through 72 hours, compared to the standard 75 mg dose of bupivacaine.
We intend to expand upon the results of this Phase 2 hemorrhoidectomy trial with commercial registry programs designed to confirm that the administration of EXPAREL in the surgical setting improves patient outcomes while consuming fewer resources. We intend to develop publications, abstracts, clinical pharmacology newsletters and meeting presentations that demonstrate the value of EXPAREL as the foundation for effective multimodal postsurgical pain management. In addition, we plan to develop new treatment protocols for postsurgical pain management overall and in specific patient populations.
Reimbursement for surgical procedures is typically capitated, or fixed by third-party payers based on the specific surgical procedure performed regardless of the cost or amount of treatments provided. However, many patients, including those who are elderly, obese, suffer from sleep apnea or are opioid tolerant, are likely to have a high incidence of opioid-related adverse events, increasing the length of stay and the cost relative to the capitated reimbursement. We intend to conduct commercial registry studies to demonstrate reduced opioid use, reduced opioid-related adverse effects, lower total resource consumption, reduced length of stay and greater patient satisfaction. Furthermore, the use of EXPAREL to reduce opioid consumption may also present the opportunity to move selected hospital procedures to the ambulatory setting.
EXPAREL Regulatory Plan
In September 2010, we filed an NDA for EXPAREL with the FDA, which was accepted by the FDA for review on December 10, 2010, using a 505(b)(2) application. We are initially seeking FDA approval of EXPAREL for postsurgical analgesia by local administration into the surgical wound, or infiltration, a procedure commonly employing bupivacaine. Under the PDUFA guidelines, the FDA has a goal of ten months from the date of an NDA filing to make a decision regarding the approval of our filing. Section 505(b)(2) of the Federal Food, Drug and Cosmetic Act, or the FFDCA, permits the submission of an NDA where at least some of the information required for approval comes from studies not conducted by or for the applicant, and for which the applicant has not obtained a right of reference. Supportive information may also include scientific literature and publicly available information contained in the labeling of other medications.
EXPAREL consists of bupivacaine encapsulated in DepoFoam, both of which are used in FDA-approved products:
8
Table of Contents
| • | Bupivacaine, a well-characterized generic anesthetic/analgesic, has an established safety profile and over 20 years of use in the United States. |
| • | DepoFoam, modified to meet the requirements of each product, is used to extend the release of the active drug substances in the marketed products DepoCyt(e) and DepoDur. |
We have requested a clinical trial waiver for children under two years of age. We have also requested and currently expect to receive a deferral for patients 2-18 years of age until patients in these groups can be studied in an appropriate step-wise manner. Three Phase 2/3 trials are planned, first in children 12-18 years old, then 6-11 years old, then 2-5 years old. The waiver and deferral, if granted, will allow us to conduct these trials after the approval of our NDA.
Additional Indications
We are pursuing several additional indications for EXPAREL and expect to submit a supplemental NDA, or sNDA, for nerve block and epidural administration. We believe that these additional indications for EXPAREL present a low-risk, low-cost opportunity for clinical development and will allow us to fully leverage our manufacturing and commercial infrastructure.
Nerve Block. Nerve block is a general term used to refer to the injection of local anesthetic onto or near nerves for control of pain. Nerve blocks can be single injections but have limited duration of action. When extended pain management is required, a catheter is used to deliver bupivacaine continuously using an external pump. According to Thomson Data over eight million nerve block procedures were conducted in the United States in 2008, with over four million of these procedures utilizing bupivacaine. EXPAREL is designed to provide extended pain management with a single injection utilizing a narrow gauge needle.
We have completed two Phase 2 clinical trials in which 40 patients received EXPAREL for nerve block. EXPAREL demonstrated efficacy and was safe and well tolerated in these clinical trials. We expect to conduct additional clinical trials in this indication.
Epidural Administration. An epidural is a form of regional anesthesia involving injection of anesthetic drugs into the outermost part of the spinal canal, or the epidural space. Epidurals can be single injections but have limited duration of action. When extended pain management is required, a catheter is placed into the epidural space and the anesthetic drug is delivered continuously using an external pump. According to IMS and Thomson Data, over six million epidural procedures were conducted in the United States in 2007, with over 590,000 of these procedures utilizing local anesthetics, including bupivacaine. EXPAREL is designed to provide extended pain management with a single injection utilizing a narrow gauge needle.
We have completed one Phase 1 clinical trial in which 24 subjects received EXPAREL by epidural administration that demonstrated proof of concept for this indication. EXPAREL was safe and well tolerated in this clinical trial. We expect to conduct additional clinical trials in this indication.
Sales and Marketing
We currently intend to develop and commercialize EXPAREL and our other product candidates in the United States while out-licensing commercialization rights for other territories. Our goal is to retain significant control over the development process and commercial execution for our product candidates, while participating in a meaningful way in the economics of all drugs that we bring to the market.
The members of our management team who will lead the commercialization of EXPAREL, if it is approved, have successfully commercialized multiple products in the hospital market, including Rocephin, Versed, Zantac IV and Angiomax. We are currently developing our commercialization strategy, with the input of KOLs in the field of postsurgical pain management as well as healthcare practitioner and quality improvement organizations. We continue to expand our pre-commercialization activities including EXPAREL positioning and messaging, publication strategy, Phase 3b/4 clinical trials and registry trials, initiatives with payer organizations, and distribution and national accounts strategies.
9
Table of Contents
If EXPAREL is approved, we intend to hire our own dedicated field sales force, consisting of approximately 60 representatives at the time of the commercial launch, to commercialize the product. Within three years of launch we expect to have approximately 100 representatives, which we estimate can effectively cover our hospital and ambulatory surgery customers in the United States. We believe a typical sales representative focused on office-based healthcare practitioners can effectively reach five to seven healthcare practitioners per day; whereas, a typical hospital-focused sales representative can reach many more healthcare practitioners. Notably, a hospital-focused sales representative faces significantly less travel time between sales calls and less wait time in healthcare practitioner offices as a large number of prescribers can be found in a single location. Our sales force will be supported by marketing as well as several teams of healthcare professionals who will support our formulary approval and customer education initiatives.
The target audience for EXPAREL is healthcare practitioners who influence pain management decisions, including surgeons, anesthesiologists, pharmacists and nurses. Our commercial sales force will focus on reaching the top 1,000 U.S. hospitals performing surgical procedures (based on Thomson Reuters benchmark obstetrician and gynecological, general and orthopedic surgical procedures performed within these hospitals), which represent approximately 70% of the market opportunity for EXPAREL. If we obtain regulatory approvals for additional indications for EXPAREL and our other product candidates, our targeted audience may change to reflect new market opportunities.
DepoFoam—Our Proprietary Drug Delivery Technology
Our current product development activities utilize our proprietary DepoFoam drug delivery technology. DepoFoam consists of microscopic spherical particles composed of a honeycomb-like structure of numerous internal aqueous chambers containing an active drug ingredient. Each chamber is separated from adjacent chambers by lipid membranes. Following injection, the DepoFoam particles release drug over an extended period of time by erosion and/or reorganization of the particles’ lipid membranes. Release rates are determined by the choice and relative amounts of lipids in the formulation.
Our DepoFoam formulation provides several technical, regulatory and commercial advantages over competitive technologies, including:
| • | Convenience. Our DepoFoam products are ready to use and do not require reconstitution or mixing with another solution, and can be used with patient friendly narrow gauge needles and pen systems; |
| • | Multiple regulatory precedents. Our DepoFoam products, DepoCyt(e) and DepoDur, have been approved in the United States and Europe, making regulatory authorities familiar with our DepoFoam technology; |
| • | Extensive safety history. Our DepoFoam products have over ten years of safety data as DepoCyt(e) has been sold in the United States since 1999; |
| • | Administration into privileged sites. Our DepoFoam products are approved for epidural administration (DepoDur) and intrathecal injection (DepoCyt(e)) and may potentially be used for intraocular and intratumoral administration; |
| • | Proven manufacturing capabilities. We continue to make DepoFoam-based products in our cGMP facilities on a daily basis as we prepare for the launch of EXPAREL; |
| • | Flexible time release. Encapsulated drug releases over a desired period of time, from 1 to 30 days; |
| • | Favorable pharmacokinetics. Decrease in adverse events associated with high peak blood levels, thereby improving the utility of the product; |
10
Table of Contents
| • | Shortened development timeline. Does not alter the native molecule potentially enabling the filing of a 505(b)(2) application; and |
| • | Aseptic manufacturing and filling. Enables use with proteins, peptides, nucleic acids, vaccines and small molecules. |
Other Products
Depocyt(e)
DepoCyt(e) is a sustained-release liposomal formulation of the chemotherapeutic agent cytarabine utilizing our DepoFoam technology. Depocyt(e) is indicated for the intrathecal treatment of lymphomatous meningitis, a life-threatening complication of lymphoma, a cancer of the immune system. Lymphomatous meningitis can be controlled with conventional cytarabine, but because of the drug’s short half-life, a spinal injection is required twice per week, whereas DepoCyt(e) is dosed once every two weeks in an outpatient setting. DepoCyt(e) was granted accelerated approval by the FDA in 1999 and full approval in 2007. We received revenue from DepoCyt(e) of $10.3 million from our commercial partners in 2010.
DepoDur
DepoDur is an extended-release injectable formulation of morphine utilizing our DepoFoam technology. DepoDur is indicated for epidural administration for the treatment of pain following major surgery. DepoDur is designed to provide effective pain relief of up to 48 hours and has demonstrated improved patient mobility and freedom from indwelling catheters. DepoDur was approved by the FDA in 2004. We received revenue from DepoDur of $1.1 million from our commercial partners in 2010.
Other Product Candidates
DepoNSAID
Our preclinical product candidates, extended release formulations of NSAIDs, are designed to provide the benefits of injectable NSAIDs with a prolonged duration of action in order to improve patient care and ease of use in the acute pain environment. Currently available injectable products provide a four to six hour duration of action. We believe that there is an unmet medical need for a product which could provide a longer duration of action, especially for postsurgical pain management as part of a multimodal pain regimen. Prolonged intra-articular delivery of NSAIDs is also being evaluated for acute pain in major joints due to injury or arthritis. We have DepoFoam formulations for several NSAIDs, and we expect to select a lead product candidate in 2011.
DepoMethotrexate
Our preclinical product candidate, an extended release formulation of methotrexate, is designed to improve the market utility of methotrexate, the most commonly used disease modifying anti-rheumatic drug currently being prescribed for over 500,000 patients globally. While methotrexate is the established standard of care for first line therapy in rheumatoid arthritis, this agent is commonly associated with nausea, vomiting and drowsiness due to high peak blood levels immediately following traditional administration. Our product candidate is designed to address the medical need for a patient friendly and cost effective formulation which can be utilized to improve patient compliance and the ability to tolerate methotrexate therapy. We believe DepoMethotrexate will also allow healthcare providers to treat these patients more aggressively, improve efficacy outcomes and avoid the progression to more expensive alternatives such as biologic therapies. We currently have one year of stability data for our desired product formulation.
11
Table of Contents
Commercial Partners and Agreements
SkyePharma
In connection with the stock purchase agreement related to the Acquisition, we agreed to pay SkyePharma Holdings, Inc., or SPHI, a specified contingent milestone payment related to EXPAREL sales. Additionally, we agreed to pay to SPHI a 3% royalty of our sales of EXPAREL in the United States, Japan, the United Kingdom, France, Germany, Italy and Spain. Such obligations to make contingent milestone payments and royalties will continue for the term in which such sales related to EXPAREL are covered by a valid claim in certain patent rights related to EXPAREL and other biologics products.
We have the right to cease paying royalties in the event that SPHI breaches certain covenants not to compete contained in the stock purchase agreement. In the event that we cease to sell EXPAREL and begin marketing a similar replacement product for EXPAREL, we would no longer be obligated to make royalty payments, but we may be required to make certain milestone payments upon reaching certain sales milestones.
Research Development Foundation
Pursuant to an agreement with one of our stockholders, the Research Development Foundation, or RDF, we are required to pay RDF a low single-digit royalty on our gross revenues, as defined in our agreement with RDF, from our DepoFoam-based products, for as long as certain patents assigned to us under the agreement remain valid. RDF has the right to terminate the agreement for an uncured material breach by us, in connection with our bankruptcy or insolvency or if we directly or indirectly oppose or dispute the validity of the assigned patent rights.
Sigma-Tau Pharmaceuticals
In December 2002, we entered into a supply and distribution agreement with Enzon Pharmaceuticals Inc. regarding the sale of DepoCyt. Pursuant to the agreement, Enzon was appointed the exclusive distributor of DepoCyt in the United States and Canada for a ten year term. In January 2010, Sigma-Tau Pharmaceuticals, Inc., or Sigma-Tau, acquired the rights to sell DepoCyt from Enzon Pharmaceuticals for the United States and Canada. Under the supply and distribution agreement, we supply unlabeled DepoCyt vials to Sigma-Tau for finished packaging. Under these agreements, we receive a fixed payment for manufacturing the vials of DepoCyt and a double-digit royalty on sales by Sigma-Tau in the United States and Canada.
We and Sigma-Tau have the right to terminate the agreement for an uncured material breach by the other party or in the event that a generic pharmaceutical product that is therapeutically equivalent to DepoCyt is commercialized. We may terminate the agreement if certain minimum sales targets are not met by Sigma-Tau. Sigma-Tau may terminate the agreement if, as a result of a settlement or a final court or regulatory action, the manufacture, use or sale of DepoCyt in the United States is prohibited.
Mundipharma International Holdings Limited
In June 2003, we entered into an agreement granting Mundipharma International Holdings Limited, or Mundipharma, exclusive marketing and distribution rights to DepoCyte in the European Union and certain other European countries. This agreement continues in force for 15 years, and after that term expires, continues year to year unless terminated by us or by Mundipharma upon no less than 12 months written notice.
Under the agreement, as amended, and a separate supply agreement, we receive a fixed payment for manufacturing the vials of DepoCyte, as well as a royalty comprised of a fixed sum per vial supplied to Mundipharma, an additional sum payable if Mundipharma’s quarterly net sales exceed a certain amount, and a mid single-digit royalty on all sales exceeding a certain amount. We are also entitled to receive up to €10 million in milestone payments from Mundipharma upon the achievement by Mundipharma of certain milestone events, of which we have already received €2.5 million and we do not expect to receive the remaining €7.5 million.
12
Table of Contents
We and Mundipharma have the right to terminate the agreement for an uncured material breach by the other party, in connection with the other party’s bankruptcy or insolvency or the repossession of all or any material part of the other party’s business or assets. Mundipharma has the right to terminate the agreement if its marketing authorization is cancelled or withdrawn for a certain period, or if it is prevented from selling DepoCyte in any three countries in the territory covered in the agreement by a final non-appealable judgment in respect of infringement by DepoCyte of any third party intellectual property rights.
EKR Therapeutics Inc.
In August 2007, we entered into a licensing, distribution and marketing agreement with EKR Therapeutics, Inc., or EKR, granting them exclusive distribution rights to DepoDur in North America, South America and Central America. Under this agreement, as amended, we received nonrefundable license fees of $5.0 million upon execution of the agreement in August 2007, $5.0 million in 2008 and $5.0 million in 2009. At the time we entered into the agreement we had the right to receive aggregate milestone payments of up to $20 million, but we do not expect any additional milestone payments under the agreement. This agreement continues in force for the longer of 15 years from the first commercial sale of DepoDur in the territory covered by the agreement or until the expiration of the last valid claim in our patents covering DepoDur in such territory. After that term, the agreement continues for consecutive periods of two years, unless terminated earlier by EKR.
Under this agreement, as amended, we receive a fixed payment for manufacturing the vials of DepoDur and a royalty comprised of a fixed amount per vial, a single-digit royalty on any incremental price increase implemented by EKR over the base price specified in the agreement and a fixed advanced royalty payment that was made within three days of the agreement date, which is offset against EKR’s future payment obligations.
We and EKR have the right to terminate the agreement for an uncured material breach by the other party, an uncured material misrepresentation in any representation or warranty made in the agreement, in connection with the other party’s bankruptcy or insolvency, in connection with the threat of or actual cessation of all or any material part of the other party’s business, if the other Party is prevented from performing any of its material obligations by any law, governmental or other action for a period of 120 days, or if force majeure prevents other party from performing any of its material obligations for six months. We have the right to terminate the agreement if EKR fails to make its first commercial sale of DepoDur within a fixed period from the receipt of marketing authorization for any country in the territory covered by the agreement, or if we terminate the supply agreement upon written notice to EKR and all royalties paid by EKR to us in any one year period following the date of such termination are less than a certain amount, unless the difference between that amount and the actual royalties paid by EKR is paid to us within 30 days of notice of such termination. EKR has the right to terminate the agreement at any time without cause upon written notice to us within a specified timeframe. EKR has the right to terminate the agreement as to any country if DepoDur is withdrawn from the market in such country as a result of regulatory action by FDA or other governmental entities or there are significant adverse reactions from use of DepoDur.
Flynn Pharma Limited
In September 2007, we entered into a marketing agreement with Flynn Pharma Limited, or Flynn, granting them exclusive distribution rights to DepoDur in the European Union, certain other European countries, South Africa and the Middle East. This agreement continues in force for the longer of five years from first commercial sale of DepoDur in the territory covered by the agreement or until the expiration of the last valid claim in our patents covering DepoDur for a maximum term of 15 years from the date of first commercial sale in such territory.
Under this agreement and a separate supply agreement with Flynn, we provide DepoDur manufacturing supply of finished product for sale in the territories licensed by Flynn, and we receive a fixed payment for manufacturing the vials and if net sales of DepoDur in the territory covered by the agreement exceed a certain amount, an additional payment. We are also entitled to receive milestone payments from Flynn upon the achievement by Flynn of certain milestone events.
We and Flynn have the right to terminate the agreement for an uncured material breach by the other party, in connection with the other party’s bankruptcy or insolvency or the repossession of all or any material part of the other party’s business or assets, or if force majeure prevents other party from performing any of its material
13
Table of Contents
obligations for 180 days. We have the right to terminate the agreement if Flynn fails to make its first commercial sale of DepoDur in specified countries covered by the agreement by one year from the later of Flynn’s receipt of marketing authorization or pricing approval for DepoDur, or if first commercial sale has not been made within 18 months of Flynn’s receipt of marketing authorization or pricing approval for DepoDur.
Novo Nordisk
In January 2011, we entered into an agreement with Novo Nordisk A/S, or Novo, pursuant to which we granted non-exclusive rights to Novo under certain of our patents and know-how to develop, manufacture and commercialize formulations of a Novo proprietary drug using our DepoFoam drug delivery technology. Under this agreement, we agreed to undertake specified development and technology transfer activities and to manufacture pre-clinical and certain clinical supplies of such DepoFoam formulated Novo product until the completion of such technology transfer activities. Novo is obligated to pay for all costs incurred by us in conducting such development, manufacturing and technology transfer activities. We received a one-time upfront payment of $1.5 million from Novo. We are also entitled to receive single-digit royalties on sales of such Novo product for up to twelve years following the first commercial sale of such Novo product. In addition, we are entitled to receive up to $24 million in milestone payments based on achievement of specified development events, and up to an additional $20 million in milestone payments based on sales of such Novo product exceeding specified amounts. Each party has the right to terminate the agreement for an uncured material breach by the other party or in connection with the other party’s bankruptcy or similar event. In addition, Novo has the right to terminate the agreement for convenience at any time upon sixty (60) days notice prior to commercialization of such Novo product and upon ninety (90) days notice thereafter, subject to Novo’s payment of a specified termination fee if, after initiation of the technology transfer but prior to commercialization, Novo terminates the agreement other than for certain specified reasons. We also have the right to terminate the agreement if (1) Novo decides to discontinue or terminate the development or commercialization of such Novo product, (2) such Novo product no longer has regulatory approval in any market, or (3) Novo or any of its affiliates or sublicensees of such Novo product challenges the validity or enforceability of any of the licensed patents.
Paul Capital
On March 23, 2007, we entered into an amended and restated royalty interests assignment agreement with Paul Capital, pursuant to which we assigned to Paul Capital the right to receive a portion of our royalty payments from DepoCyt(e) and DepoDur. The original agreement was entered into prior to the Acquisition by the Predecessor in order to monetize certain royalty payments from DepoCyt(e) and DepoDur. In connection with the Acquisition, the original agreement with Paul Capital was amended and restated and the responsibility to pay the royalty interest in product sales of DepoCyt(e) and DepoDur was transferred to us and we were required to make payments to Paul Capital upon the occurrence of certain events. For additional information, see “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Liquidity and Capital Resources—Royalty Interests Assignment Agreement” and “Risk Factors—Risks Related to Our Financial Condition and Capital Requirements.” Under our financing arrangement with Paul Capital, upon the occurrence of certain events, Paul Capital may require us to repurchase the right to receive royalty payments that we assigned to it, or may foreclose on certain assets that secure our obligations to Paul Capital. Any exercise by Paul Capital of its right to cause us to repurchase the assigned right or any foreclosure by Paul Capital would adversely affect our results of operations and our financial condition.
Feasibility Agreements with Third Parties
In the ordinary course of our business activities, we enter into feasibility agreements with third parties who desire access to our proprietary DepoFoam technology to conduct research, feasibility and formulation work. Under these agreements, we are compensated to perform feasibility testing on a third-party product to determine the likelihood of developing a successful formulation of that product using our proprietary DepoFoam technology. If successful in the feasibility stage, these programs can advance to a full development contract.
14
Table of Contents
Manufacturing
We manufacture DepoCyt(e) and DepoDur for our various commercial partners. We also manufacture all of our clinical supplies of EXPAREL. We manufacture our products in two manufacturing facilities. These facilities are designated as Building 1 and Building 6 and are located within two miles of each other on two separate and distinct sites in San Diego, California. Both of our facilities are inspected regularly and approved for pharmaceutical manufacturing by the FDA, the European Medicines Agency, or the EMA, the Medicines and Healthcare Products Regulatory Agency, or the MHRA, the Drug Enforcement Administration, or the DEA, and the Environmental Protection Agency, or the EPA.
We provide DepoCyt(e) and DepoDur to our commercial partners on a set cost basis as established by each specific licensing contract. All manufacturing of products, initial product release and stability testing are conducted by us in accordance with cGMP.
Building 1 is an approximately 80,000 square foot concrete structure located on a five acre site. It was custom built as a pharmaceutical R&D and manufacturing facility in August 1995. Activities in this facility include the manufacture of EXPAREL bulk pharmaceutical product candidate in a dedicated production line and its fill/finish into vials, the manufacture of the DepoDur bulk commercial pharmaceutical product, microbiological and quality control testing, product storage, development of analytical methods, research and development, the coordination of clinical and regulatory functions, and general administrative functions. We have renovated the dedicated EXPAREL production line to expand its capacity and it will be available for the FDA’s pre-approval inspection in 2011. This production line is designed to meet forecasted market demands after initial launch of EXPAREL, if it is approved. We have current plans to further expand our manufacturing capacity to meet future demand.
Building 6 is located in a 17-acre pharmaceutical industrial park. It is a two story concrete masonry structure built in 1977 that we and our predecessors have leased since August 1993. We occupy approximately 22,000 square feet of the first floor. Building 6 houses the current manufacturing process for DepoCyt(e), the fill/finish of DepoCyt(e) and DepoDur into vials, a pilot plant suite for new product development and early stage clinical product production, a microbiology laboratory and miscellaneous support and maintenance areas.
Distribution of our DepoFoam products, including EXPAREL, requires cold-chain distribution, whereby a product must be maintained between specified temperatures. We have validated processes for continuous monitoring of temperature from manufacturing through delivery to the end-user. We and our partners have utilized similar cold-chain processes for DepoCyt(e) and DepoDur.
Intellectual Property and Exclusivity
We seek to protect our product candidates and our technology through a combination of patents, trade secrets, proprietary know-how, regulatory exclusivity and contractual restrictions on disclosure.
Patents and Patent Applications
We seek to protect the proprietary position of our product candidates by, among other methods, filing U.S. and foreign patent applications related to our proprietary technology, inventions and improvements that are important to the development of our business. As of December 31, 2010, there are over 15 families of patents and patent applications relating to various aspects of the DepoFoam delivery technology. Patents have been issued in numerous countries, with an emphasis on the North American, European and Japanese markets. These patents generally have a term of 20 years from the date of the nonprovisional filing unless referring to an earlier filed application. Some of our U.S. patents have a term from 17 years from the grant date. Our issued patents expire at various dates in the future, with the last currently issued patent expiring in 2019. All of these patent families are assigned solely to us, with the exception of one family relating to DepoFoam formulations of insulin-like growth factor I, which is jointly assigned to us and Novartis Vaccines and Diagnostics, Inc. (formerly Chiron Corporation). In addition, two provisional patents have been filed within the last year relating to either DepoFoam-based products or processes for making DepoFoam.
15
Table of Contents
In regard to patents providing protection for EXPAREL, issued patents in the United States relating to the composition of the product candidate and methods for modifying the rate of drug release of the product candidate expire in November 2013 and January 2017, respectively. Pending U.S. applications relating to the composition of the product candidate and the process for making the product candidate, if granted, would expire in September 2018 and November 2018, respectively. In Europe, granted patents related to the composition of the product candidate expire in November 2014 and September 2018. Pending applications in Europe relating to methods of modifying the rate of drug release of the product candidate and the process for making the product candidate, if granted, would expire in January 2018 and November 2018, respectively. Recently, a provisional patent was filed relating to a new process to manufacture EXPAREL and other DepoFoam-based products. The process offers many advantages to the current process, including larger scale production and lower manufacturing costs. This new processing is in the process of being pursued as a non-provisional application. The provisional patent, if granted, could prevent others from using this process until 2031. Furthermore, a non-exclusively licensed patent of ours relating to EXPAREL was allowed in Europe with an expiration date in October 2021 and was extended in the United States until October 2023.
Trade Secrets and Proprietary Information
Trade secrets play an important role in protecting DepoFoam-based products and provide protection beyond patents and regulatory exclusivity. The scale-up and commercial manufacture of DepoFoam products involves processes, custom equipment, and in-process and release analytical techniques that we believe are unique to us. The expertise and knowledge required to understand the critical aspects of DepoFoam manufacturing steps requires knowledge of both traditional and non-traditional emulsion processing and traditional pharmaceutical production, overlaid with all of the challenges presented by aseptic manufacturing. We seek to protect our proprietary information, including our trade secrets and proprietary know-how, by requiring our employees, consultants and other advisors to execute proprietary information and confidentiality agreements upon the commencement of their employment or engagement. These agreements generally provide that all confidential information developed or made known during the course of the relationship with us be kept confidential and not be disclosed to third parties except in specific circumstances. In the case of our employees, the agreements also typically provide that all inventions resulting from work performed for us, utilizing our property or relating to our business and conceived or completed during employment shall be our exclusive property to the extent permitted by law. Where appropriate, agreements we obtain with our consultants also typically contain similar assignment of invention obligations. Further, we require confidentiality agreements from entities that receive our confidential data or materials.
Competition
The pharmaceutical and biotechnology industries are intensely competitive and subject to rapid and significant technological change. Our competitors include organizations such as major multinational pharmaceutical companies, established biotechnology companies, specialty pharmaceutical companies and generic drug companies. Many of our competitors have greater financial and other resources than we have, such as more commercial resources, larger research and development staffs and more extensive marketing and manufacturing organizations. As a result, these companies may obtain marketing approval more rapidly than we are able and may be more effective in selling and marketing their products. Smaller or early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies.
Our competitors may succeed in developing, acquiring or licensing on an exclusive basis technologies and drug products that are more effective or less costly than EXPAREL or any other products that we are currently selling through partners or developing or that we may develop, which could render our products obsolete and noncompetitive. We expect any products that we develop and commercialize to compete on the basis of, among other things, efficacy, safety, convenience of administration and delivery, price and the availability of reimbursement from government and other third-party payers. We also expect to face competition in our efforts to identify appropriate collaborators or partners to help commercialize our product candidates in our target commercial markets.
16
Table of Contents
We anticipate EXPAREL will compete with currently marketed bupivacaine and opioid analgesics such as morphine. We also expect to compete with an extended release bupivacaine product in development by Durect Corporation which has been licensed to Hospira in North America (Posidur) and to Nycomed for Europe (Optesia).
We also anticipate that EXPAREL will compete with elastomeric bag/catheter devices intended to provide bupivacaine over several days. I-FLOW Corporation (acquired by Kimberly-Clark Corporation in 2009) has marketed these medical devices in the United States since 2004.
Government Regulation
Federal Food, Drug and Cosmetic Act
Prescription drug products are subject to extensive pre- and post-market regulation by the FDA, including regulations that govern the testing, manufacturing, distribution, safety, efficacy, approval, labeling, storage, record keeping, reporting, advertising and promotion of such products under the FDCA, and its implementing regulations, and by comparable agencies and laws in foreign countries. Failure to comply with applicable FDA or other regulatory requirements may result in, among other things, warning letters, clinical holds, civil or criminal penalties, recall or seizure of products, injunction, debarment, partial or total suspension of production or withdrawal of the product from the market. The FDA must approve any new drug, including a new dosage form or new use of a previously approved drug, prior to marketing in the United States. All applications for FDA approval must contain, among other things, information relating to safety and efficacy, pharmaceutical formulation, stability, manufacturing, processing, packaging, labeling and quality control.
New Drug Applications
Generally, the FDA must approve any new drug before marketing of the drug occurs in the United States. This process generally involves:
| • | completion of preclinical laboratory and animal testing and formulation studies in compliance with the FDA’s Good Laboratory Practice, or GLP, regulations; |
| • | submission to the FDA of an IND application for human clinical testing, which must become effective before human clinical trials may begin in the United States; |
| • | approval by an independent institutional review board, or IRB, at each clinical trial site before each trial may be initiated; |
| • | performance of human clinical trials, including adequate and well-controlled clinical trials in accordance with good clinical practices, or GCP, to establish the safety and efficacy of the proposed drug product for each intended use; |
| • | submission of an NDA to the FDA; |
| • | satisfactory completion of an FDA pre-approval inspection of the product’s manufacturing facility or facilities to assess compliance with the FDA’s cGMP regulations, and to ensure that the facilities, methods and controls are adequate to preserve the drug’s identity, quality and purity; |
| • | satisfactory completion of an FDA advisory committee review, if applicable; and |
| • | approval by the FDA of the NDA. |
The preclinical and clinical testing and approval process requires substantial time, effort and financial resources, and we cannot be certain that the FDA will grant approvals for any of our product candidates on a timely basis, if at all. Preclinical tests include laboratory evaluation of product chemistry, formulation and stability, as well as studies to evaluate toxicity in animals. The results of preclinical tests, together with manufacturing information,
17
Table of Contents
analytical data and a proposed clinical trial protocol and other information, are submitted as part of an IND application to the FDA. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, places the trial on a clinical hold because of, among other things, concerns about the conduct of the clinical trial or about exposure of human research subjects to unreasonable health risks. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. Our submission of an IND may not result in FDA authorization to commence a clinical trial. In addition, the FDA requires sponsors to amend an existing IND for each successive clinical trial conducted during product development. Further, an independent institutional review board, or IRB, covering each medical center proposing to conduct the clinical trial must review and approve the plan for any clinical trial and informed consent information for subjects before the clinical trial commences at that center, and it must monitor the clinical trial until completed. The FDA, the IRB or the sponsor may suspend a clinical trial at any time, or from time to time, on various grounds, including a finding that the subjects or patients are being exposed to an unacceptable health risk.
Clinical trials involve the administration of the investigational new drug to human subjects under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research subjects provide their informed consent for their participation in any clinical trial. For purposes of an NDA submission and approval, typically, the conduct of human clinical trials occurs in the following three pre-market sequential phases, which may overlap:
| • | Phase 1: sponsors initially conduct clinical trials in a limited population to test the product candidate for safety, dose tolerance, absorption, metabolism, distribution and excretion in healthy humans or, on occasion, in patients, such as cancer patients. |
| • | Phase 2: sponsors conduct clinical trials generally in a limited patient population to identify possible adverse effects and safety risks, to determine the efficacy of the product for specific targeted indications and to determine dose tolerance and optimal dosage. Sponsors may conduct multiple Phase 2 clinical trials to obtain information prior to beginning larger and more extensive Phase 3 clinical trials. |
| • | Phase 3: these include expanded controlled and uncontrolled trials, including pivotal clinical trials. When Phase 2 evaluations suggest the effectiveness of a dose range of the product and acceptability of such product’s safety profile, sponsors undertake Phase 3 clinical trials in larger patient populations to obtain additional information needed to evaluate the overall benefit and risk balance of the drug and to provide an adequate basis to develop labeling. |
In addition, sponsors may elect to conduct, or be required by the FDA to conduct, Phase 4 clinical trials to further assess the drug’s safety or effectiveness after NDA approval. Such post approval trials are typically referred to as Phase 4 clinical trials.
Sponsors submit the results of product development, preclinical studies and clinical trials to the FDA as part of an NDA. NDAs must also contain extensive information relating to the product’s pharmacology, chemistry, manufacture, controls and proposed labeling, among other things. In addition, 505(b)(2) applications must contain a patent certification for each patent listed in FDA’s “Orange Book” that covers the drug referenced in the application and upon which the third-party studies were conducted. For some drugs, the FDA may require risk evaluation and mitigation strategies, or REMS, which could include medication guides, physician communication plans, or restrictions on distribution and use, such as limitations on who may prescribe the drug or where it may be dispensed or administered. Upon receipt, the FDA has 60 days to determine whether the NDA is sufficiently complete to initiate a substantive review. If the FDA identifies deficiencies that would preclude substantive review, the FDA will refuse to accept the NDA and will inform the sponsor of the deficiencies that must be corrected prior to resubmission. If the FDA accepts the submission for substantive review, the FDA typically reviews the NDA in accordance with established timeframes. Under PDUFA, the FDA agrees to specific goals for NDA review time through a two-tiered classification system, Priority Review and Standard Review. A Priority Review designation is given to drugs that offer major advances in treatment, or provide a treatment where no adequate therapy exists. For a Priority Review application, the FDA aims to complete the initial review cycle in six months. Standard Review applies to all applications that are not eligible for Priority Review. The FDA aims to complete Standard Review NDAs within a ten-month timeframe. We anticipate that the FDA will grant our product candidate a Standard
18
Table of Contents
Review. Review processes often extend significantly beyond anticipated completion dates due to FDA requests for additional information or clarification, difficulties scheduling an advisory committee meeting, negotiations regarding REMS, or FDA workload issues. The FDA may refer the application to an advisory committee for review, evaluation and recommendation as to the application’s approval. The recommendations of an advisory committee do not bind the FDA, but the FDA generally follows such recommendations.
Under PDUFA, NDA applicants must pay significant NDA user fees upon submission. In addition, manufacturers of approved prescription drug products must pay annual establishment and product user fees.
Before approving an NDA, the FDA may inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and are adequate to ensure consistent production of the product within required specifications. Additionally, the FDA will typically inspect one or more clinical sites to ensure compliance with GCP before approving an NDA.
After the FDA evaluates the NDA and the manufacturing facilities, it may issue an approval letter or a Complete Response Letter, or CRL, to indicate that the review cycle for an application is complete and that the application is not ready for approval. CRLs generally outline the deficiencies in the submission and may require substantial additional testing or information in order for the FDA to reconsider the application. Even if such additional information is submitted, the FDA may ultimately decide that the NDA does not satisfy the criteria for approval. Data from clinical trials are not always conclusive and the FDA may interpret data differently than we do. The FDA could also require a REMS plan to mitigate risks, which could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA also may condition approval on, among other things, changes to proposed labeling, a commitment to conduct one or more post-market studies or clinical trials and the correction of identified manufacturing deficiencies, including the development of adequate controls and specifications. If and when the deficiencies have been addressed to the FDA’s satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Section 505(b)(2) New Drug Applications
As an alternate path to FDA approval, particularly for modifications to drug products previously approved by the FDA, an applicant may submit an NDA under Section 505(b)(2) of the FDCA. Section 505(b)(2) was enacted as part of the Drug Price Competition and Patent Term Restoration Act of 1984, also known as the Hatch-Waxman Act, and permits the submission of an NDA where at least some of the information required for approval comes from clinical trials not conducted by or for the applicant and for which the applicant has not obtained a right of reference. The FDA interprets Section 505(b)(2) of the FDCA to permit the applicant to rely upon the FDA’s previous findings of safety and effectiveness for an approved product. The FDA may also require companies to perform additional clinical trials or measurements to support any change from the previously approved product. The FDA may then approve the new product candidate for all or some of the label indications for which the referenced product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant.
Section 505(b)(2) applications are subject to any non-patent exclusivity period applicable to the referenced product, which may delay approval of the 505(b)(2) application even if FDA has completed its substantive review and determined the drug should be approved. In addition, 505(b)(2) applications must include patent certifications to any patents listed in the Orange Book as covering the referenced product. If the 505(b)(2) applicant seeks to obtain approval before the expiration of an applicable listed patent, the 505(b)(2) applicant must provide notice to the patent owner and NDA holder of the referenced product. If the patent owner or NDA holder bring a patent infringement lawsuit within 45 days of such notice, the 505(b)(2) application cannot be approved for 30 months or until the 505(b)(2) applicant prevails, whichever is sooner. If the 505(b)(2) applicant loses the patent infringement suit, FDA may not approve the 505(b)(2) application until the patent expires, plus any period of pediatric exclusivity.
19
Table of Contents
In the NDA submissions for our product candidates, we intend to follow the development and approval pathway permitted under the FDCA that we believe will maximize the commercial opportunities for these product candidates.
Post-Approval Requirements
After approval, the NDA sponsor must comply with comprehensive requirements governing, among other things, drug listing, recordkeeping, manufacturing, marketing activities, product sampling and distribution, annual reporting and adverse event reporting. There are also extensive U.S. Drug Enforcement Agency, or DEA, regulations applicable to marketed controlled substances.
If new safety issues are identified following approval, the FDA can require the NDA sponsor to revise the approved labeling to reflect the new safety information; conduct post-market studies or clinical trials to assess the new safety information; and implement a REMS program to mitigate newly-identified risks. The FDA may also require post-approval testing, including Phase 4 studies, and surveillance programs to monitor the effect of approved products which have been commercialized, and the FDA has the authority to prevent or limit further marketing of a product based on the results of these post-marketing programs. Drugs may be marketed only for approved indications and in accordance with the provisions of the approved label. Further, if we modify a drug, including any changes in indications, labeling or manufacturing processes or facilities, the FDA may require us to submit and obtain FDA approval of a new or supplemental NDA, which may require us to develop additional data or conduct additional preclinical studies and clinical trials.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies, and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP and impose reporting and documentation requirements upon us and any third-party manufacturers that we may decide to use.
If after approval the FDA determines that the product does not meet applicable regulatory requirements or poses unacceptable safety risks, the FDA may take other regulatory actions, including initiating suspension or withdrawal of the NDA approval. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
| • | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| • | fines, warning letters or holds on post-approval clinical trials; |
| • | refusal of the FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals; |
| • | product seizure or detention, or refusal to permit the import or export of products; or |
| • | injunctions or the imposition of civil or criminal penalties. |
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. These regulations include standards and restrictions for direct-to-consumer advertising, industry-sponsored scientific and educational activities, promotional activities involving the internet, and off-label promotion. While physicians may prescribe for off label uses, manufacturers may only promote for the approved indications and in accordance with the provisions of the approved label. The FDA has very broad enforcement authority under the FDCA, and failure to abide by these regulations can result in penalties, including the issuance of a warning letter directing entities to correct deviations from FDA standards, a requirement that future advertising and promotional materials be pre-cleared by the FDA, and state and federal civil and criminal investigations and prosecutions.
20
Table of Contents
In addition, the distribution of prescription pharmaceutical products is subject to the Prescription Drug Marketing Act, or PDMA, which regulates the distribution of drugs and drug samples at the federal level, and sets minimum standards for the registration and regulation of drug distributors by the states. Both the PDMA and state laws limit the distribution of prescription pharmaceutical product samples and impose requirements to ensure accountability in distribution, including a drug pedigree which tracks the distribution of prescription drugs.
DEA Regulation
One of our marketed products, DepoDur, is regulated as a “controlled substance” as defined in the Controlled Substances Act of 1970, or CSA, which establishes registration, security, recordkeeping, reporting, storage, distribution and other requirements administered by the DEA. The DEA is concerned with the control of handlers of controlled substances, and with the equipment and raw materials used in their manufacture and packaging, in order to prevent loss and diversion into illicit channels of commerce.
The DEA regulates controlled substances as Schedule I, II, III, IV or V substances. Schedule I substances by definition have no established medicinal use, and may not be marketed or sold in the United States. A pharmaceutical product may be listed as Schedule II, III, IV or V, with Schedule II substances considered to present the highest risk of abuse and Schedule V substances the lowest relative risk of abuse among such substances. DepoDur, a sustained-release injectable morphine sulfate, is listed as a Schedule II controlled substance under the CSA. Consequently, its manufacture, shipment, storage, sale and use is subject to a high degree of regulation. For example, generally, all Schedule II drug prescriptions must be signed by a physician, physically presented to a pharmacist and may not be refilled without a new prescription.
Annual registration is required for any facility that manufactures, tests, distributes, dispenses, imports or exports any controlled substance. Except for certain defined co-incident activities, each registration is specific to the particular location, activity and controlled substance schedule. For example, separate registrations are needed for import and manufacturing, and each registration must specify which schedules of controlled substances are authorized.
The DEA typically inspects a facility to review its security measures prior to issuing a registration and, thereafter, on a periodic basis. Security requirements vary by controlled substance schedule, with the most stringent requirements applying to Schedule I and Schedule II substances. Required security measures include background checks on employees and physical control of inventory through measures such as vaults, cages, surveillance cameras and inventory reconciliations. Records must be maintained for the handling of all controlled substances, and periodic reports made to the DEA, for example distribution reports for Schedule I and II controlled substances, Schedule III substances that are narcotics, and other designated substances. Reports must also be made for thefts or significant losses of any controlled substance, and to obtain authorization to destroy any controlled substance. In addition, special authorization, notification and permit requirements apply to imports and exports.
In addition, a DEA quota system controls and limits the availability and production of controlled substances in Schedule I or II. Distributions of any Schedule I or II controlled substance must also be accomplished using special order forms, with copies provided to the DEA. Because DepoDur, a sustained-release injectable morphine sulfate, is regulated as a Schedule II controlled substance, it is subject to the DEA’s production and procurement quota scheme. The DEA establishes annually an aggregate quota for how much morphine may be produced in total in the United States based on the DEA’s estimate of the quantity needed to meet legitimate scientific and medicinal needs. This limited aggregate amount of morphine that the DEA allows to be produced in the United States each year is allocated among individual companies, who must submit applications annually to the DEA for individual production and procurement quotas. We must receive an annual quota from the DEA in order to produce or procure any Schedule I or Schedule II substance, including morphine sulfate for use in manufacturing DepoDur. The DEA may adjust aggregate production quotas and individual production and procurement quotas from time to time during the year, although the DEA has substantial discretion in whether or not to make such adjustments. Our quota of an active ingredient may not be sufficient to meet commercial demand or complete the manufacture or purchase of material required for clinical trials. Any delay or refusal by the DEA in establishing our quota for controlled substances could delay or stop our clinical trials or product launches, or interrupt commercial sales of our products which could have a material adverse effect on our business, financial position and results of operations.
21
Table of Contents
The DEA conducts periodic inspections of registered establishments that handle controlled substances. Failure to maintain compliance with applicable requirements, particularly as manifested in loss or diversion, can result in enforcement action that could have a material adverse effect on our business, results of operations and financial condition. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate proceedings to revoke those registrations. In certain circumstances, violations could eventuate in criminal proceedings.
Individual states also regulate controlled substances, and we are subject to such regulation by several states with respect to the manufacture and distribution of these products.
International Regulation
In addition to regulations in the United States, we are subject to a variety of foreign regulations governing clinical trials and the commercial sales and distribution of our products. Whether or not we obtain FDA approval for a product, we must obtain approval by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval. The requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from country to country.
For example, in the EEA (which is comprised of the 27 Member States of the EU plus Norway, Iceland and Liechtenstein), medicinal products can only be commercialized after obtaining a Marketing Authorization (MA). There are two types of marketing authorizations:
| • | The Community MA, which is issued by the European Commission through the Centralized Procedure, based on the opinion of the Committee for Medicinal Products for Human Use (CHMP) of the EMA, and which is valid throughout the entire territory of the EEA. The Centralized Procedure is mandatory for certain types of products, such as biotechnology medicinal products, orphan medicinal products, and medicinal products containing a new active substance indicated for the treatment of AIDS, cancer, neurodegenerative disorders, diabetes, auto-immune and viral diseases. The Centralized Procedure is optional for products containing a new active substance not yet authorized in the EEA, or for products that constitute a significant therapeutic, scientific or technical innovation or which are in the interest of public health in the EU. |
| • | National MAs, which are issued by the competent authorities of the Member States of the EEA and only cover their respective territory, are available for products not falling within the mandatory scope of the Centralized Procedure. Where a product has already been authorized for marketing in a Member State of the EEA (the Reference Member State or RMS), this National MA can be recognized in other Member States (the Concerned Member States or CMS) through the Mutual Recognition Procedure. If the product has not received a National MA in any Member State at the time of application, it can be approved simultaneously in various Member States through the Decentralized Procedure. Under the Decentralized Procedure, an identical dossier is submitted to the competent authorities of each of the Member States in which the MA is sought, one of which is selected by the applicant as the RMS. The competent authority of the RMS prepares a draft assessment report, a draft summary of the product characteristics, or SPC, and a draft of the labeling and package leaflet, which are sent to the CMS for their approval. If the CMS raise no objections, based on a potential serious risk to public health, to the assessment, SPC, labeling, or packaging proposed by the RMS, the product is subsequently granted a national MA in all the Member States (i.e. in the RMS and the CMS). If one or more CMS raise objections based on a potential serious risk to public health, the application is referred to the Coordination group for mutual recognition and decentralized procedure for human medicinal products (the CMDh), which is composed of representatives of the EEA Member States. If a consensus cannot be reached within the CMDh the matters is referred for arbitration to the CHMP, which can reach a final decision binding on all EEA Member States. A similar process applies to disputes between the RMS and the CMS in the Mutual Recognition Procedure. |
As with FDA approval we may not be able to secure regulatory approvals in Europe in a timely manner, if at all. Additionally, as in the United States, post-approval regulatory requirements, such as those regarding product manufacture, marketing, or distribution, would apply to any product that is approved in Europe, and failure to comply with such obligations could have a material adverse effect on our ability to successfully commercialize any product.
22
Table of Contents
The conduct of clinical trials in the EU is governed by the EU Clinical Trials Directive (Directive 2001/20/EC of 4 April 2001, of the European Parliament and of the Council on the approximation of the laws, regulations and administrative provisions of the Member States relating to implementation of good clinical practice in the conduct of clinical trials on medicinal products for human use). The provisions of the EU Clinical Trials Directive were required to be implemented and applied by the EEA Member States before May 2004. The EU Clinical Trials Directive harmonizes the regulatory requirements of the Member States of the EEA for the conduct of clinical trials in their respective territories. The EU Clinical Trials Directive requires sponsors of clinical trials to submit formal applications to, and to obtain the approval of, national ethics committees and regulatory authorities prior to the initiation of clinical trials.
In addition to regulations in Europe and the United States, we will be subject to a variety of foreign regulations governing clinical trials and commercial distribution of any future products.
Third Party Payer Coverage and Reimbursement
The commercial success of our product candidates will depend, in part, upon the availability of coverage and reimbursement from third-party payers at the federal, state and private levels. Government payer programs, including Medicare and Medicaid, private health care insurance companies and managed care plans may deny coverage or reimbursement for a product or therapy in whole or in part if they determine that the product or therapy is not medically appropriate or necessary. Also, third-party payers have attempted to control costs by limiting coverage and the amount of reimbursement for particular procedures or drug treatments. The United States Congress and state legislatures from time to time propose and adopt initiatives aimed at cost containment, which could impact our ability to sell our products profitably.
For example, in March 2010, President Obama signed into law the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, which we refer to collectively as the Health Care Reform Law, a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. Effective October 1, 2010, the Health Care Reform Law revises the definition of “average manufacturer price” for reporting purposes, which could increase the amount of Medicaid drug rebates owed to states by pharmaceutical manufacturers. The Health Reform Law also establishes a new Medicare Part D coverage gap discount program, in which drug manufacturers must provide 50% point-of-sale discounts on products covered under Part D beginning in 2011. Further, also beginning in 2011, the new law imposes a significant annual, nondeductible fee on companies that manufacture or import branded prescription drug products. Substantial new provisions affecting compliance have also been enacted, which may require us to modify our business practices with healthcare practitioners. We will not know the full effects of the Health Care Reform Law until applicable federal and state agencies issue regulations or guidance under the new law. Although it is too early to determine the effect of the Health Care Reform Law, the new law appears likely to continue the pressure on pharmaceutical pricing, especially under the Medicare program, and may also increase our regulatory burdens and operating costs. Moreover, in the coming years, additional changes could be made to governmental healthcare programs that could significantly impact the success of our products.
The cost of pharmaceuticals continues to generate substantial governmental and third-party payer interest. We expect that the pharmaceutical industry will experience pricing pressures due to the trend toward managed healthcare, the increasing influence of managed care organizations and additional legislative proposals. Our results of operations could be adversely affected by current and future healthcare reforms.
Some third-party payers also require pre-approval of coverage for new or innovative devices or drug therapies before they will reimburse healthcare providers that use such therapies. While we cannot predict whether any proposed cost-containment measures will be adopted or otherwise implemented in the future, the announcement or adoption of these proposals could have a material adverse effect on our ability to obtain adequate prices for our product candidates and to operate profitably.
23
Table of Contents
In international markets, reimbursement and healthcare payment systems vary significantly by country, and many countries have instituted price ceilings on specific products and therapies. There can be no assurance that our products will be considered medically reasonable and necessary for a specific indication, that our products will be considered cost-effective by third-party payers, that an adequate level or reimbursement will be available so that the third-party payers’ reimbursement policies will not adversely affect our ability to sell our products profitably.
Marketing/Data Exclusivity
The FDA may grant three or five years of marketing exclusivity in the United States for the approval of new or supplemental NDAs, including Section 505(b)(2) NDAs, for, among other things, new indications, dosages or dosage forms of an existing drug, if new clinical investigations that were conducted or sponsored by the applicant are essential to the approval of the application. Additionally, six months of marketing exclusivity in the United States is available under Section 505A of the FDCA if, in response to a written request from the FDA, a sponsor submits and the agency accepts requested information relating to the use of the approved drug in the pediatric population. This six month pediatric exclusivity period is not a standalone exclusivity period, but rather is added to any existing patent or non-patent exclusivity period for which the drug product is eligible. Based on our clinical trial program for EXPAREL, we plan to seek at least three years of marketing exclusivity upon receipt of FDA approval for EXPAREL (anticipated exclusivity through at least the third quarter of 2014). We may also seek an additional period of six months exclusivity from the FDA if the FDA requests, and we successfully complete, pediatric clinical trials for EXPAREL.
Manufacturing Requirements
We must comply with applicable FDA regulations relating to FDA’s cGMP regulations. The cGMP regulations include requirements relating to organization of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, packaging and labeling controls, holding and distribution, laboratory controls, records and reports, and returned or salvaged products. The manufacturing facilities for our products must meet cGMP requirements to the satisfaction of the FDA pursuant to a pre-approval inspection before we can use them to manufacture our products. We and any third-party manufacturers are also subject to periodic inspections of facilities by the FDA and other authorities, including procedures and operations used in the testing and manufacture of our products to assess our compliance with applicable regulations. Failure to comply with these and other statutory and regulatory requirements subjects a manufacturer to possible legal or regulatory action, including warning letters, the seizure or recall of products, injunctions, consent decrees placing significant restrictions on or suspending manufacturing operations and civil and criminal penalties. Adverse experiences with the product must be reported to the FDA and could result in the imposition of market restrictions through labeling changes or in product removal. Product approvals may be withdrawn if compliance with regulatory requirements is not maintained or if problems concerning safety or efficacy of the product occur following approval.
Healthcare Fraud and Abuse Laws
We are subject to various federal, state and local laws targeting fraud and abuse in the healthcare industry. For example, in the United States, there are federal and state anti-kickback laws that prohibit the payment or receipt of kickbacks, bribes or other remuneration intended to induce the purchase or recommendation of healthcare products and services or reward past purchases or recommendations. Violations of these laws can lead to civil and criminal penalties, including fines, imprisonment and exclusion from participation in federal healthcare programs. These laws are potentially applicable to manufacturers of products regulated by the FDA, such as us, and hospitals, physicians and other potential purchasers of such products.
In particular, the federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, receiving, offering or providing remuneration, directly or indirectly, to induce either the referral of an individual, or the furnishing, recommending, or arranging for a good or service, for which payment may be made under a federal healthcare program such as the Medicare and Medicaid programs. The term “remuneration” is not defined in the federal Anti-Kickback Statute and has been broadly interpreted to include anything of value, including for example,
24
Table of Contents
gifts, discounts, the furnishing of supplies or equipment, credit arrangements, payments of cash, waivers of payments, ownership interests and providing anything at less than its fair market value. In addition, the recently enacted Health Care Reform Law, among other things, amends the intent requirement of the federal Anti-Kickback Statute and the applicable criminal healthcare fraud statutes contained within 42 U.S.C. § 1320a-7b effective March 23, 2010. Pursuant to the statutory amendment, a person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation. In addition, the PPACA provides that the government may assert that a claim including items or services resulting from a violation of 42 U.S.C. § 1320a-7b constitutes a false or fraudulent claim for purposes of the civil False Claims Act (discussed below) or the civil monetary penalties statute, which imposes a penalty of $5000 against any person who is determined to have presented or caused to be presented claims to a federal health care program that the person knows or should know is for an item or service that was not provided as claimed or is false or fraudulent. Moreover, the lack of uniform court interpretation of the Anti-Kickback Statute makes compliance with the law difficult.
Recognizing that the Anti-Kickback Statute is broad and may technically prohibit many innocuous or beneficial arrangements within the healthcare industry, the U.S. Department of Health and Human Services’ Office of Inspector General, or OIG, issued regulations in July of 1991, and periodically since that time, which the OIG refers to as “safe harbors.” These safe harbor regulations set forth certain provisions which, if met in form and substance, will assure pharmaceutical companies, healthcare providers and other parties that they will not be prosecuted under the federal Anti-Kickback Statute. Additional safe harbor provisions providing similar protections have been published intermittently since 1991. Although full compliance with these provisions ensures against prosecution under the federal Anti-Kickback Statute, the failure of a transaction or arrangement to fit within a specific safe harbor does not necessarily mean that the transaction or arrangement is illegal or that prosecution under the federal Anti-Kickback Statute will be pursued. However, conduct and business arrangements that do not fully satisfy each applicable safe harbor may result in increased scrutiny by government enforcement authorities, such as the OIG or federal prosecutors. Additionally, there are certain statutory exceptions to the federal Anti-Kickback Statute, one or more of which could be used to protect a business arrangement, although we understand that OIG is of the view that an arrangement that does not meet the requirements of a safe harbor cannot satisfy the corresponding statutory exception, if any, under the federal Anti-Kickback Statute.
Additionally, many states have adopted laws similar to the federal Anti-Kickback Statute. Some of these state prohibitions apply to referral of patients for healthcare items or services reimbursed by any third-party payer, not only the Medicare and Medicaid programs, and do not contain identical safe harbors. Government officials have focused their enforcement efforts on marketing of healthcare services and products, among other activities, and have brought cases against numerous pharmaceutical and medical device companies, and certain sales and marketing personnel for allegedly offering unlawful inducements to potential or existing customers in an attempt to procure their business or reward past purchases or recommendations.
Another development affecting the healthcare industry is the increased use of the federal civil False Claims Act and, in particular, actions brought pursuant to the False Claims Act’s “whistleblower” or “qui tam” provisions. The civil False Claims Act imposes liability on any person or entity who, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal healthcare program. The qui tam provisions of the False Claims Act allow a private individual to bring civil actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government, and to share in any monetary recovery. In recent years, the number of suits brought by private individuals has increased dramatically. In addition, various states have enacted false claim laws analogous to the False Claims Act. Many of these state laws apply where a claim is submitted to any third-party payer and not merely a federal healthcare program. When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties of $5,500 to $11,000 for each separate false claim. There are many potential bases for liability under the False Claims Act. Liability arises, primarily, when an entity knowingly submits, or causes another to submit, a false claim for reimbursement to the federal government. The False Claims Act has been used to assert liability on the basis of inadequate care, kickbacks and other improper referrals, improperly reported government pricing metrics such as Best Price or Average Manufacturer Price, improper use of Medicare numbers when detailing the provider of services, improper promotion of off-label uses (i.e., uses not expressly approved by FDA in a drug’s label), and allegations as to misrepresentations with respect to the services rendered. Our activities relating to the reporting of discount and rebate information and other information affecting federal, state and third-party reimbursement of our products, and the sale and marketing of our
25
Table of Contents
products and our service arrangements or data purchases, among other activities, may be subject to scrutiny under these laws. We are unable to predict whether we would be subject to actions under the False Claims Act or a similar state law, or the impact of such actions. However, the cost of defending such claims, as well as any sanctions imposed, could adversely affect our financial performance.
Also, the Health Insurance Portability and Accountability Act of 1996, or HIPAA, created several new federal crimes, including health care fraud, and false statements relating to health care matters. The health care fraud statute prohibits knowingly and willfully executing a scheme to defraud any health care benefit program, including private third-party payers. The false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services.
In addition, under California law, pharmaceutical companies must adopt a comprehensive compliance program that is in accordance with both the April 2003 Office of Inspector General Compliance Program Guidance for Pharmaceutical Manufacturers, or OIG Guidance, and the Pharmaceutical Research and Manufacturers of America Code on Interactions with Healthcare Professionals, or the PhRMA Code. The PhRMA Code seeks to promote transparency in relationships between health care professionals and the pharmaceutical industry and to ensure that pharmaceutical marketing activities comport with the highest ethical standards. The PhRMA Code contains strict limitations on certain interactions between health care professionals and the pharmaceutical industry relating to gifts, meals, entertainment and speaker programs, among others. Also, certain states, such as Massachusetts and Minnesota, have imposed restrictions on the types of interactions that pharmaceutical and medical device companies or their agents (e.g., sales representatives) may have with health care professionals, including bans or strict limitations on the provision of meals, entertainment, hospitality, travel and lodging expenses, and other financial support, including funding for continuing medical education activities.
Healthcare Privacy and Security Laws
We may be subject to, or our marketing activities may be limited by, HIPAA, and its implementing regulations, which established uniform standards for certain “covered entities” (healthcare providers, health plans and healthcare clearinghouses) governing the conduct of certain electronic healthcare transactions and protecting the security and privacy of protected health information. The American Recovery and Reinvestment Act of 2009, commonly referred to as the economic stimulus package, included sweeping expansion of HIPAA’s privacy and security standards called the Health Information Technology for Economic and Clinical Health Act, or HITECH, which became effective on February 17, 2010. Among other things, the new law makes HIPAA’s privacy and security standards directly applicable to “business associates”—independent contractors or agents of covered entities that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also increased the civil and criminal penalties that may be imposed against covered entities, business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorney’s fees and costs associated with pursuing federal civil actions.
Research and Development Expenses
From the Acquisition through December 31, 2010, we incurred research and development expenses of $98.7 million, of which $95.1 million is related to the development of EXPAREL. We incurred research and development expenses of $18.6 million for the year ended December 31, 2010, $26.2 million for the year ended December 31, 2009 and $33.2 million for the year ended December 31, 2008.
Employees
As of December 31, 2010, we employed 83 employees, with 8 in research and development, 56 in operations, and 19 in general and administrative. All of our employees are located in the United States. None of our employees are represented by a labor union, and we consider our current employee relations to be good.
26
Table of Contents
Available Information
We file reports and other information with the SEC as required by the Securities Exchange Act of 1934, as amended, which we refer to as the Exchange Act. You can find, copy and inspect information we file at the SEC’s public reference room, which is located at 100 F Street, N.E., Room 1580, Washington, DC 20549. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the SEC’s public reference room. You can review our electronically filed reports and other information that we file with the SEC on the SEC’s web site at http://www.sec.gov.
We were incorporated in Delaware under the name Blue Acquisition Corp. in December 2006 and changed our name to Pacira, Inc. in June 2007. In October 2010, we changed our name to Pacira Pharmaceuticals, Inc. Our principal executive offices are located at 5 Sylvan Way, Suite 125, Parsippany, New Jersey 07054, and our telephone number is (973) 254-3560. Our Internet website is http://www.pacira.com. We make available free of charge through our website our annual report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Exchange Act. We make these reports available through our website as soon as reasonably practicable after we electronically file such reports with, or furnish such reports to, the SEC. In addition, we regularly use our website to post information regarding our business, product development programs and governance, and we encourage investors to use our website, particularly the information in the section entitled “Investors & Media,” as a source of information about us.
The foregoing references to our website are not intended to, nor shall they be deemed to, incorporate information on our website into this report by reference.
| Item 1A. | Risk Factors |
In addition to the other information in this Annual Report on Form 10-K, any of the factors set forth below could significantly and negatively affect our business, financial condition, results of operations or prospects. The trading price of our common stock may decline due to these risks. This sections contains forward-looking statements. You should refer to the explanation of the qualifications and limitations on forward-looking statements beginning on page 1.
Risks Related to the Development and Commercialization of our Product Candidates
We are dependent on the success of our lead product candidate, EXPAREL, and cannot guarantee that this product candidate will receive regulatory approval or be successfully commercialized.
We have invested a significant portion of our efforts and financial resources in the development of our most advanced product candidate, EXPAREL. Our ability to generate revenues in the near term is substantially dependent on our ability to develop and commercialize EXPAREL. In September 2010, we submitted a new drug application, or NDA, with the U.S. Food and Drug Administration, or FDA, which was accepted by the FDA for review on December 10, 2010, seeking approval to commercialize EXPAREL for treatment of postsurgical pain. We cannot commercialize EXPAREL prior to obtaining FDA approval. Even though EXPAREL has completed two pivotal Phase 3 clinical trials with positive results, EXPAREL is still, nonetheless, susceptible to the risks of failure inherent at any stage of product development, including the appearance of unexpected adverse events, the FDA’s determination that EXPAREL is not approvable or failure to achieve its primary endpoints in subsequent clinical trials. For example, in 2009, we completed two Phase 3 clinical trials of EXPAREL that did not meet their primary endpoints.
If we do not receive FDA approval for, and commercialize, EXPAREL, we will not be able to generate revenue from EXPAREL in the foreseeable future, or at all. Any significant delays in obtaining approval for and commercializing EXPAREL will have a substantial adverse impact on our business and financial condition.
If approved, our ability to generate revenues from EXPAREL will depend on our ability to:
27
Table of Contents
| • | create market demand for EXPAREL through our own marketing and sales activities, and any other arrangements to promote this product candidate we may later establish; |
| • | hire, train and deploy a sales force to commercialize EXPAREL in the United States; |
| • | manufacture EXPAREL in sufficient quantities and at an acceptable quality and at an acceptable manufacturing cost to meet commercial demand at launch and thereafter; |
| • | establish and maintain agreements with wholesalers, distributors and group purchasing organizations on commercially reasonable terms; |
| • | create partnerships with, or offer licenses to, third parties to promote and sell EXPAREL outside the United States; and |
| • | maintain patent and trade secret protection and regulatory exclusivity for EXPAREL. |
We face significant competition from other pharmaceutical and biotechnology companies. Our operating results will suffer if we fail to compete effectively.
The pharmaceutical and biotechnology industries are intensely competitive and subject to rapid and significant technological change. Our major competitors include organizations such as major multinational pharmaceutical companies, established biotechnology companies and specialty pharmaceutical and generic drug companies. Many of our competitors have greater financial and other resources than we have, such as larger research and development staff, more extensive marketing, distribution, sales and manufacturing organizations and experience, more extensive clinical trial and regulatory experience, expertise in prosecution of intellectual property rights and access to development resources like personnel generally and technology. As a result, these companies may obtain regulatory approval more rapidly than we are able to and may be more effective in selling and marketing their products. Smaller or early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies. Our competitors may succeed in developing, acquiring or licensing on an exclusive basis technologies and drug products that are more effective or less costly than EXPAREL or any other product candidate that we are currently developing or that we may develop, which could render our products obsolete and noncompetitive or significantly harm the commercial opportunity for EXPAREL.
As a result of these factors, our competitors may obtain regulatory approval of their products more rapidly than we are able to or may obtain patent protection or other intellectual property rights that limit our ability to develop or commercialize EXPAREL. Our competitors may also develop drugs that are more effective, useful or less costly than ours and may be more successful than us in manufacturing and marketing their products.
EXPAREL will compete with well-established products with similar indications. Competing products available for postsurgical pain management include opioids such as morphine, fentanyl, meperidine and hydromorphone, each of which is available generically from several manufacturers, and several of which are available as proprietary products using novel delivery systems. Ketorolac, an injectable non-steroidal anti-inflammatory drug, or NSAID, is also available generically in the United States from several manufacturers, and Caldolor (ibuprofen for injection), an NSAID, has been approved by the FDA for pain management and fever in adults. In addition, EXPAREL will compete with non-opioid products such as bupivacaine, Marcaine, ropivacaine and other anesthetics/analgesics, all of which are also used in the treatment of postsurgical pain and are available as either oral tablets, injectable dosage forms or administered using novel delivery systems. Additional products may be developed for the treatment of acute pain, including new injectable NSAIDs, novel opioids, new formulations of currently available opioids and NSAIDS, long-acting local anesthetics and new chemical entities as well as alternative delivery forms of various opioids and NSAIDs.
We also expect to compete with an extended release bupivacaine product in development by Durect Corporation which has been licensed to Hospira in North America (Posidur) and to Nycomed for Europe (Optesia).
28
Table of Contents
We also anticipate that EXPAREL will compete with elastomeric bag/catheter devices intended to provide bupivacaine over several days. I-FLOW Corporation (acquired by Kimberly-Clark Corporation in 2009) has marketed these medical devices in the United States since 2004.
If we are unable to establish effective marketing and sales capabilities or enter into agreements with third parties to market and sell our product candidates, if they are approved, we may be unable to generate product revenues.
We currently do not have a commercial infrastructure for the marketing, sale and distribution of pharmaceutical products. In order to commercialize our products, we must build our marketing, sales and distribution capabilities or make arrangements with third parties to perform these services. If EXPAREL is approved by the FDA, we plan to build a commercial infrastructure to launch EXPAREL in the United States, including a specialty sales force of approximately 100 people within three years from launch. We may seek to further penetrate the U.S. market in the future by expanding our sales force or through collaborations with other pharmaceutical or biotechnology companies or third-party manufacturing and sales organizations. We may also seek to commercialize EXPAREL outside the United States, although we currently plan to do so with a marketing and sales collaborator and not with our own sales force.
The establishment and development of our own sales force and related compliance plans to market any products we may develop will be expensive and time consuming and could delay any product launch, and we may not be able to successfully develop this capability. We, or our future collaborators, will have to compete with other pharmaceutical and biotechnology companies to recruit, hire, train and retain marketing and sales personnel. In the event we are unable to develop a marketing and sales infrastructure, we would not be able to commercialize EXPAREL or any other product candidates that we develop, which would limit our ability to generate product revenues.
Although our current plan is to hire most of our sales and marketing personnel only if EXPAREL is approved by the FDA, we will incur expenses prior to product launch in recruiting this sales force and developing a marketing and sales infrastructure. If the commercial launch of EXPAREL is delayed as a result of FDA requirements or other reasons, we would incur these expenses prior to being able to realize any revenue from sales of EXPAREL. Even if we are able to effectively hire a sales force and develop a marketing and sales infrastructure, our sales force and marketing teams may not be successful in commercializing EXPAREL or any other product candidates that we may develop.
To the extent we rely on third parties to commercialize any products for which we obtain regulatory approval, we may receive less revenues than if we commercialized these products ourselves. In addition, we would have less control over the sales efforts of any other third parties involved in our commercialization efforts. In the event we are unable to collaborate with a third-party marketing and sales organization, our ability to generate product revenues may be limited either in the United States or internationally.
If EXPAREL does not achieve broad market acceptance, the revenues that we generate from its sales will be limited.
Other than DepoCyt(e) and DepoDur, we have never commercialized a product candidate for any indication. Even if EXPAREL is approved by the appropriate regulatory authorities for marketing and sale, it may not gain acceptance among physicians, hospitals, patients and third-party payers. If our products for which we obtain regulatory approval do not gain an adequate level of acceptance, we may not generate significant additional product revenues or become profitable. Market acceptance of EXPAREL, and any other product candidates that we develop, license or acquire, by physicians, hospitals, patients and third-party payers will depend on a number of factors, some of which are beyond our control. The degree of market acceptance of EXPAREL will depend on a number of factors, including:
| • | limitations or warnings contained in the product’s FDA-approved labeling, including potential limitations or warnings for EXPAREL that may be more restrictive than other pain management products; |
29
Table of Contents
| • | changes in the standard of care for the targeted indications for EXPAREL, which could reduce the marketing impact of any claims that we could make following FDA approval, if obtained; |
| • | the relative convenience and ease of administration of EXPAREL; |
| • | the prevalence and severity of adverse events associated with EXPAREL; |
| • | cost of treatment versus economic and clinical benefit in relation to alternative treatments; |
| • | the availability of adequate coverage or reimbursement by third parties, such as insurance companies and other healthcare payers, and by government healthcare programs, including Medicare and Medicaid; |
| • | the extent and strength of our marketing and distribution of EXPAREL; |
| • | the safety, efficacy and other potential advantages over, and availability of, alternative treatments, including, in the case of EXPAREL, a number of products already used to treat pain in the hospital setting; and |
| • | distribution and use restrictions imposed by the FDA or to which we agree as part of a mandatory risk evaluation and mitigation strategy or voluntary risk management plan. |
Our ability to effectively promote and sell EXPAREL and any other product candidates that we may develop, license or acquire in the hospital marketplace will also depend on pricing and cost effectiveness, including our ability to produce a product at a competitive price and achieve acceptance of the product onto hospital formularies, and our ability to obtain sufficient third-party coverage or reimbursement. Since many hospitals are members of group purchasing organizations, which leverage the purchasing power of a group of entities to obtain discounts based on the collective buying power of the group, our ability to attract customers in the hospital marketplace will also depend on our ability to effectively promote our product candidates to group purchasing organizations. We will also need to demonstrate acceptable evidence of safety and efficacy, as well as relative convenience and ease of administration. Market acceptance could be further limited depending on the prevalence and severity of any expected or unexpected adverse side effects associated with our product candidates. If our product candidates are approved but do not achieve an adequate level of acceptance by physicians, health care payers and patients, we may not generate sufficient revenue from these products, and we may not become or remain profitable. In addition, our efforts to educate the medical community and third-party payers on the benefits of our product candidates may require significant resources and may never be successful.
In addition, even if the medical community accepts that EXPAREL is safe and effective for its approved indications, physicians and patients may not immediately be receptive to EXPAREL and may be slow to adopt it as an accepted treatment of postsurgical pain. It is unlikely that any labeling approved by the FDA will contain claims that EXPAREL is safer or more effective than competitive products or will permit us to promote EXPAREL as being superior to competing products. Further, the availability of inexpensive generic forms of postsurgical pain management products may also limit acceptance of EXPAREL among physicians, patients and third-party payers. If EXPAREL is approved but does not achieve an adequate level of acceptance among physicians, patients and third-party payers, we may not generate meaningful revenues from EXPAREL and we may not become profitable.
We may rely on third parties to perform many essential services for any products that we commercialize, including services related to warehousing and inventory control, distribution, customer service, accounts receivable management, cash collection and adverse event reporting. If these third parties fail to perform as expected or to comply with legal and regulatory requirements, our ability to commercialize EXPAREL will be significantly impacted and we may be subject to regulatory sanctions.
We may retain third-party service providers to perform a variety of functions related to the sale and distribution of EXPAREL, key aspects of which will be out of our direct control. These service providers may provide key services related to warehousing and inventory control, distribution, customer service, accounts receivable management and cash collection, and, as a result, most of our inventory may be stored at a single
30
Table of Contents
warehouse maintained by one such service provider. If we retain this provider, we would substantially rely on them as well as other third-party providers that perform services for us, including entrusting our inventories of products to their care and handling. If these third-party service providers fail to comply with applicable laws and regulations, fail to meet expected deadlines, or otherwise do not carry out their contractual duties to us, or encounter physical or natural damage at their facilities, our ability to deliver product to meet commercial demand would be significantly impaired. In addition, we may engage third parties to perform various other services for us relating to adverse event reporting, safety database management, fulfillment of requests for medical information regarding our product candidates and related services. If the quality or accuracy of the data maintained by these service providers is insufficient, we could be subject to regulatory sanctions.
Distribution of our DepoFoam-based products requires cold-chain distribution provided by third parties, whereby a product must be maintained between specified temperatures. We and our partners have utilized similar cold-chain processes for DepoCyt(e) and DepoDur. If a problem occurs in our cold-chain distribution processes, whether through our failure to maintain our products or product candidates between specified temperatures or because of a failure of one of our distributors or partners to maintain the temperature of the products or product candidates, the product or product candidate could be adulterated and rendered unusable. This could have a material adverse effect on our business, financial condition, results of operations and reputation.
We will need to increase the size of our organization, and we may experience difficulties in managing growth.
As of December 31, 2010, we had 83 employees. We will need to substantially expand our managerial, commercial, financial, manufacturing and other personnel resources in order to manage our operations and prepare for the commercialization of EXPAREL, if approved. Our management, personnel, systems and facilities currently in place may not be adequate to support this future growth. In addition, we may not be able to recruit and retain qualified personnel in the future, particularly for sales and marketing positions, due to competition for personnel among pharmaceutical businesses, and the failure to do so could have a significant negative impact on our future product revenues and business results. Our need to effectively manage our operations, growth and various projects requires that we:
| • | continue the hiring and training of an effective commercial organization in anticipation of the potential approval of EXPAREL, and establish appropriate systems, policies and infrastructure to support that organization; |
| • | ensure that our consultants and other service providers successfully carry out their contractual obligations, provide high quality results, and meet expected deadlines; |
| • | continue to carry out our own contractual obligations to our licensors and other third parties; and |
| • | continue to improve our operational, financial and management controls, reporting systems and procedures. |
We may be unable to successfully implement these tasks on a larger scale and, accordingly, may not achieve our development and commercialization goals.
We may not be able to manage our business effectively if we are unable to attract and retain key personnel.
We may not be able to attract or retain qualified management and commercial, scientific and clinical personnel due to the intense competition for qualified personnel among biotechnology, pharmaceutical and other businesses, particularly in the San Diego, California and Northern New Jersey areas. If we are not able to attract and retain necessary personnel to accomplish our business objectives, we may experience constraints that will significantly impede the achievement of our development objectives, our ability to raise additional capital and our ability to implement our business strategy.
Our industry has experienced a high rate of turnover of management personnel in recent years. We are highly dependent on the development and manufacturing expertise for our DepoFoam delivery technology and the commercialization expertise of certain members of our senior management. In particular, we are highly dependent
31
Table of Contents
on the skills and leadership of our management team, including David Stack, our president and chief executive officer. If we lose one or more of these key employees, our ability to successfully implement our business strategy could be seriously harmed. Replacing key employees may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to develop, gain regulatory approval of and commercialize products successfully. Competition to hire from this limited pool is intense, and we may be unable to hire, train, retain or motivate additional key personnel.
Mr. Stack, our chief executive officer, is also a managing director at MPM Capital and a managing partner of Stack Pharmaceuticals, Inc. Although Mr. Stack has devoted substantially all of his time to our company over the past 12 months, Mr. Stack’s responsibilities at MPM Capital and Stack Pharmaceuticals, Inc. might require that he spend less than all his time managing our company in the future.
Under our consulting agreement with Gary Patou, M.D., our chief medical officer, he is not required to devote all of his time to our company. We cannot assure you that Dr. Patou’s time commitment to us will be sufficient to perform the duties of our chief medical officer.
We face potential product liability exposure, and if successful claims are brought against us, we may incur substantial liability for DepoCyt(e), DepoDur, EXPAREL or other product candidates that we may develop and may have to limit their commercialization.
The use of DepoCyt(e), DepoDur, EXPAREL and any other product candidates that we may develop, license or acquire in clinical trials and the sale of any products for which we obtain regulatory approval expose us to the risk of product liability claims. Product liability claims might be brought against us by consumers, health care providers or others using, administering or selling our products. If we cannot successfully defend ourselves against these claims, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
| • | loss of revenue from decreased demand for our products and/or product candidates; |
| • | impairment of our business reputation or financial stability; |
| • | costs of related litigation; |
| • | substantial monetary awards to patients or other claimants; |
| • | diversion of management attention; |
| • | loss of revenues; |
| • | withdrawal of clinical trial participants and potential termination of clinical trial sites or entire clinical programs; and |
| • | the inability to commercialize our product candidates. |
We have obtained limited product liability insurance coverage for our products and our clinical trials with a $10.0 million annual aggregate coverage limit. However, our insurance coverage may not reimburse us or may not be sufficient to reimburse us for any expenses or losses we may suffer. Moreover, insurance coverage is becoming increasingly expensive, and, in the future, we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability. We intend to expand our insurance coverage to include the sale of additional commercial products if we obtain FDA approval for our product candidates in development, but we may be unable to obtain commercially reasonable product liability insurance for any products approved for marketing, or at all. On occasion, large judgments have been awarded in class action lawsuits based on drugs that had unanticipated side effects. A successful product liability claim or series of claims brought against us could cause our stock price to fall and, if judgments exceed our insurance coverage, could decrease our cash and adversely affect our business.
32
Table of Contents
We are the sole manufacturer of DepoCyt(e) and DepoDur and we only have two FDA approved manufacturing facilities. Our inability to continue manufacturing adequate supplies of DepoCyt(e) and DepoDur could result in a disruption in the supply of DepoCyt(e) and DepoDur to our partners.
We are the sole manufacturer of DepoCyt(e) and DepoDur. We develop and manufacture DepoCyt(e) and DepoDur at our facilities in San Diego, California, which are the only FDA approved sites for manufacturing DepoCyt(e) and DepoDur in the world. Our San Diego facilities are subject to the risks of a natural or man-made disaster, including earthquakes and fires, or other business disruption. There can be no assurance that we would be able to meet our requirements for DepoCyt(e) and DepoDur if there were a catastrophic event or failure of our current manufacturing system. If we are required to change or add a new manufacturer or supplier, the process would likely require prior FDA and/or equivalent foreign regulatory authority approval, and would be very time consuming. An inability to continue manufacturing adequate supplies of DepoCyt(e) and DepoDur at our facility in San Diego, California could result in a disruption in the supply of DepoCyt(e) and DepoDur to our partners and breach of our contractual obligations.
If we fail to manufacture DepoCyt(e) and DepoDur we will lose revenues and be in breach of our licensing obligations.
We have licensed the commercial rights in specified territories of the world to market and sell our products, DepoCyt(e) and DepoDur. Under those licenses we have obligations to manufacture commercial product for our commercial partners. If we are unable to timely fill the orders placed with us by our commercial partners, we will potentially lose revenue and be in breach of our licensing obligations under the agreements. In addition, we would be in breach of our obligations to comply with our supply and distribution agreements for DepoCyt(e) and DepoDur, which would in turn be a breach of our obligations under our amended and restated royalty interests assignment agreement, or the Amended and Restated Royalty Interests Assignment Agreement, with Royalty Securitization Trust I, an affiliate of Paul Capital Advisors, LLC, or Paul Capital. See “Risk Factors—Risks Related to Our Financial Condition and Capital Requirements—Under our financing arrangement with Paul Capital, upon the occurrence of certain events, Paul Capital may require us to repurchase the right to receive royalty payments that we assigned to it, or may foreclose on certain assets that secure our obligations to Paul Capital. Any exercise by Paul Capital of its right to cause us to repurchase the assigned right or any foreclosure by Paul Capital would adversely affect our results of operations and our financial condition.”
We rely on third parties for the timely supply of specified raw materials and equipment for the manufacture of DepoCyt(e) and DepoDur. Although we actively manage these third-party relationships to provide continuity and quality, some events which are beyond our control could result in the complete or partial failure of these goods and services. Any such failure could have a material adverse effect on our financial condition and operations.
The manufacture of pharmaceutical products requires significant expertise and capital investment, including the development of advanced manufacturing techniques and process controls, and the use of specialized processing equipment. We must comply with federal, state and foreign regulations, including current Good Manufacturing Practices, or cGMP, regulations and in the case of the manufacturing of DepoDur required government licenses regarding the storage and use of controlled substances. Any failure to comply with applicable regulations may result in fines and civil penalties, suspension of production, suspension or delay in product approval for sale, product seizure or recall, or withdrawal of product approval, and would limit the availability of our product. Any manufacturing defect or error discovered after products have been produced and distributed could result in even more significant consequences, including costly recall procedures, re-stocking costs, damage to our reputation, product liability claims and litigation.
Our future growth depends on our ability to identify, develop, acquire or in-license products and if we do not successfully identify develop, acquire or in-license related product candidates or integrate them into our operations, we may have limited growth opportunities.
An important part of our business strategy is to continue to develop a pipeline of product candidates by developing, acquiring or in-licensing products, businesses or technologies that we believe are a strategic fit with our focus on the hospital marketplace. However, these business activities may entail numerous operational and financial risks, including:
33
Table of Contents
| • | difficulty or inability to secure financing to fund development activities for such development, acquisition or in-licensed products or technologies; |
| • | incurrence of substantial debt or dilutive issuances of securities to pay for development, acquisition or in-licensing of new products; |
| • | disruption of our business and diversion of our management’s time and attention; |
| • | higher than expected development, acquisition or in-license and integration costs; |
| • | exposure to unknown liabilities; |
| • | difficulty and cost in combining the operations and personnel of any acquired businesses with our operations and personnel; |
| • | inability to retain key employees of any acquired businesses; |
| • | difficulty in managing multiple product development programs; and |
| • | inability to successfully develop new products or clinical failure. |
We have limited resources to identify and execute the development, acquisition or in-licensing of products, businesses and technologies and integrate them into our current infrastructure. We may compete with larger pharmaceutical companies and other competitors in our efforts to establish new collaborations and in-licensing opportunities. These competitors likely will have access to greater financial resources than us and may have greater expertise in identifying and evaluating new opportunities. Moreover, we may devote resources to potential development, acquisitions or in-licensing opportunities that are never completed, or we may fail to realize the anticipated benefits of such efforts.
Our business involves the use of hazardous materials and we must comply with environmental laws and regulations, which can be expensive and restrict how we do business.
Our manufacturing activities involve the controlled storage, use and disposal of hazardous materials, including the components of our products, product candidates and other hazardous compounds. We are subject to federal, state and local laws and regulations governing the use, manufacture, storage, handling, release and disposal of, and exposure to, these hazardous materials. Violation of these laws and regulations could lead to substantial fines and penalties. Although we believe that our safety procedures for handling and disposing of these materials comply with the standards prescribed by these laws and regulations, we cannot eliminate the risk of accidental contamination or injury from these materials. In the event of an accident, state or federal authorities may curtail our use of these materials and interrupt our business operations. In addition, we could become subject to potentially material liabilities relating to the investigation and cleanup of any contamination, whether currently unknown or caused by future releases.
Our business and operations would suffer in the event of system failures.
Despite the implementation of security measures, our internal computer systems are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. Any system failure, accident or security breach that causes interruptions in our operations could result in a material disruption of our product development programs. For example, the loss of clinical trial data from completed clinical trials for EXPAREL could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we may incur liability and the further development of our product candidates may be delayed.
34
Table of Contents
Any collaboration arrangements that we may enter into in the future may not be successful, which could adversely affect our ability to develop and commercialize our product candidates.
Our business model is to commercialize our product candidates in the United States and generally to seek collaboration arrangements with pharmaceutical or biotechnology companies for the development or commercialization of our product candidates in the rest of the world. Accordingly, we may enter into collaboration arrangements in the future on a selective basis. Any future collaboration arrangements that we enter into may not be successful. The success of our collaboration arrangements will depend heavily on the efforts and activities of our collaborators. Collaborators generally have significant discretion in determining the efforts and resources that they will apply to these collaboration arrangements.
Disagreements between parties to a collaboration arrangement regarding clinical development and commercialization matters can lead to delays in the development process or commercializing the applicable product candidate and, in some cases, termination of the collaboration arrangement. These disagreements can be difficult to resolve if neither of the parties has final decision making authority.
Collaborations with pharmaceutical companies and other third parties often are terminated or allowed to expire by the other party. Any such termination or expiration would adversely affect us financially and could harm our business reputation.
Regulatory Risks
We may not receive regulatory approval for EXPAREL or any of our other product candidates, or the approval may be delayed for various reasons, including successful challenges to the FDA’s interpretation of Section 505(b)(2), which would have a material adverse effect on our business and financial condition.
We may experience delays in our efforts to obtain regulatory approval from the FDA for EXPAREL or any of our other product candidates, and there can be no assurance that such approval will not be delayed, or that the FDA will ultimately approve these product candidates.
The FDA may require additional data or information as part of its review of our NDA. If additional stability data or other manufacturing data is required, such data may not be available for a significant amount of time, which could further delay the approval of our NDA for EXPAREL and cause us to incur significant additional expenses. The FDA may also require us to study EXPAREL in pediatric patients. Although we have requested a waiver for patients under two years of age and a deferral for patients under 18 years of age, there can be no assurance that the FDA will grant our waiver or deferral and we may be required to perform these additional pediatric trials, which could be expensive and time consuming.
Our NDA approval is subject to a pre-approval inspection of our production facilities for manufacturing for EXPAREL. Our NDA approval for EXPAREL could be delayed if the FDA does not agree that the registration batches submitted in our NDA are fully representative of the manufacturing process and thus meet the requirements for batches that may be used to provide evidence of stability for this product candidate. In such an event, we would be required to potentially manufacture new batches in order to provide the necessary stability data which could delay FDA approval and cause us to incur significant additional expenses.
Additionally, our NDA for EXPAREL may not be approved, or approval may be delayed, as a result of changes in FDA policies for drug approval. For example, although many products have been approved by the FDA in recent years under Section 505(b)(2) under the Federal Food, Drug and Cosmetic Act, objections have been raised by certain brand-name pharmaceutical companies and others to the FDA’s interpretation of Section 505(b)(2). If challenges to the FDA’s interpretation of Section 505(b)(2) are successful, the agency may be required to change its interpretation, which could delay or prevent the approval of our NDAs for EXPAREL or any of our other product candidates.
35
Table of Contents
Any significant delay in re-submitting an NDA and obtaining FDA approval for EXPAREL, or a non-approval, could negatively impact our ability to ultimately obtain marketing authorization for this product candidate and would have a material adverse effect on our business and financial condition.
If EXPAREL is approved and we fail to manufacture the product in sufficient quantities and at acceptable quality and pricing levels, or to fully comply with cGMP regulations, we may face delays in the commercialization of this product candidate or be unable to meet market demand, and may lose potential revenues.
The manufacture of pharmaceutical products requires significant expertise and capital investment, including the development of advanced manufacturing techniques and process controls, and the use of specialized processing equipment. In order to meet anticipated demand for EXPAREL if this product candidate is approved, we plan to install additional specialized processing equipment to expand the manufacturing capacity for EXPAREL in our facilities. This processing equipment is designed based on our specifications and is not generally commercially available. If we are not able to expand our capacity to manufacture EXPAREL on time or at all, our ability to meet our customers’ product demands may be materially and adversely impacted.
We purchase raw materials and components from various suppliers in order to manufacture EXPAREL. If we are unable to source the required raw materials from our suppliers, we may experience delays in manufacturing EXPAREL and may not be able to meet our customers’ demands for EXPAREL.
In addition, we must comply with federal, state and foreign regulations, including cGMP requirements enforced by the FDA through its facilities inspection program. Any failure to comply with applicable regulations may result in fines and civil penalties, suspension of production, suspension or delay in product approval, product seizure or recall, or withdrawal of product approval, and would limit the availability of our product. Any manufacturing defect or error discovered after products have been produced and distributed could result in even more significant consequences, including costly recall procedures, re-stocking costs, damage to our reputation and potential for product liability claims.
If we are unable to produce the required commercial quantities of EXPAREL to meet market demand for EXPAREL on a timely basis or at all, or if we fail to comply with applicable laws for the manufacturing of EXPAREL, we will suffer damage to our reputation and commercial prospects and we will lose potential revenues.
The FDA may determine that EXPAREL or any of our other product candidates have undesirable side effects that could delay or prevent their regulatory approval or commercialization.
If concerns are raised regarding the safety of a new drug as a result of undesirable side effects identified during clinical testing, the FDA may decline to approve the drug at the end of the NDA review period or issue a letter requesting additional data or information prior to making a final decision regarding whether or not to approve the drug. The number of such requests for additional data or information issued by the FDA in recent years has increased, and resulted in substantial delays in the approval of several new drugs. Undesirable side effects caused by EXPAREL or any other product candidate could also result in the inclusion of unfavorable information in our product labeling, denial of regulatory approval by the FDA or other regulatory authorities for any or all targeted indications, and in turn prevent us from commercializing and generating revenues from the sale of EXPAREL or any other product candidate.
For example, the side effects observed in the EXPAREL clinical trials completed to date include nausea and vomiting. In addition, the class of drugs that EXPAREL belongs to has been associated with nervous system and cardiovascular toxicities at high doses. We cannot be certain that these side effects and others will not be observed in the future, or that the FDA will not require additional trials or impose more severe labeling restrictions due to these side effects or other concerns. The active component of EXPAREL is bupivacaine and bupivacaine infusions have been associated with the destruction of articular cartilage, or chondrolysis. Chondrolysis has not been observed in clinical trials of EXPAREL, but we cannot be certain that this side effect will not be observed in the future.
36
Table of Contents
If EXPAREL or any of our other product candidates receives regulatory approval and we or others later identify undesirable side effects caused by such products:
| • | regulatory authorities may require the addition of unfavorable labeling statements, specific warnings or a contraindication; |
| • | regulatory authorities may suspend or withdraw their approval of the product, or require it to be removed from the market; |
| • | regulatory authorities may impose restrictions on the distribution or use of the product; |
| • | we may be required to change the way the product is administered, conduct additional clinical trials or change the labeling of the product; |
| • | we may be subject to product liability claims and litigation; and |
| • | our reputation may suffer. |
Any of these events could prevent us from achieving or maintaining market acceptance of EXPAREL or any of our other product candidates and could substantially increase our commercialization costs and expenses, which in turn could delay or prevent us from generating significant revenues from its sale.
Regulatory approval for any approved product is limited by the FDA to those specific indications and conditions for which clinical safety and efficacy have been demonstrated.
Any regulatory approval is limited to those specific diseases and indications for which a product is deemed to be safe and effective by the FDA. In addition to the FDA approval required for new formulations, any new indication for an approved product also requires FDA approval. If we are not able to obtain FDA approval for any desired future indications for our products and product candidates, our ability to effectively market and sell our products may be reduced and our business may be adversely affected.
While physicians may choose to prescribe drugs for uses that are not described in the product’s labeling and for uses that differ from those tested in clinical studies and approved by the regulatory authorities, our ability to promote the products is limited to those indications that are specifically approved by the FDA. These “off-label” uses are common across medical specialties and may constitute an appropriate treatment for some patients in varied circumstances. Regulatory authorities in the United States generally do not regulate the behavior of physicians in their choice of treatments. Regulatory authorities do, however, restrict communications by pharmaceutical companies on the subject of off-label use. If our promotional activities fail to comply with these regulations or guidelines, we may be subject to warnings from, or enforcement action by, these authorities. In addition, our failure to follow FDA rules and guidelines relating to promotion and advertising may cause the FDA to issue warning letters or untitled letters, suspend or withdraw an approved product from the market, require a recall or institute fines or civil fines, or could result in disgorgement of money, operating restrictions, injunctions or criminal prosecution, any of which could harm our business.
If we are unable to complete pre-commercialization manufacturing development activities for EXPAREL on a timely basis or fail to comply with stringent regulatory requirements, we will face delays in our ability to obtain regulatory approval for, and to commercialize, this product candidate, and our costs will increase.
As part of the process for obtaining regulatory approval, we must demonstrate that the facilities, equipment and processes used to manufacture EXPAREL are capable of consistently producing a product that meets all applicable quality criteria, and that is comparable to the product that was used in our clinical trials. We must also provide the FDA with information regarding the validation of the manufacturing facilities, equipment and processes and data supporting the stability of our product candidate. If we are not in compliance with cGMP requirements, the approval of our NDA may be delayed, existing product batches may be compromised, and we may experience delays in the availability of this product candidate for commercial distribution.
37
Table of Contents
Even if EXPAREL receives regulatory approval, it and any other products we may market, including DepoCyt(e) and DepoDur, will remain subject to substantial regulatory scrutiny.
EXPAREL, DepoCyt(e) and DepoDur and any other product candidates that we may develop, license or acquire will also be subject to ongoing FDA requirements with respect to the manufacturing, labeling, packaging, storage, distribution, advertising, promotion, record-keeping and submission of safety and other post-market information on the drug. In addition, the subsequent discovery of previously unknown problems with a product may result in restrictions on the product, including withdrawal of the product from the market.
If EXPAREL, DepoCyt(e) and DepoDur or any other product that we may develop, license or acquire fails to comply with applicable regulatory requirements, such as cGMP regulations, a regulatory agency may:
| • | issue warning letters or untitled letters; |
| • | require us to enter into a consent decree, which can include imposition of various fines, reimbursements for inspection costs, required due dates for specific actions and penalties for noncompliance; |
| • | impose fines and other civil or criminal penalties; |
| • | suspend regulatory approval; |
| • | suspend any ongoing clinical trials; |
| • | refuse to approve pending applications or supplements to approved applications filed by us; |
| • | impose restrictions on operations, including costly new manufacturing requirements; or |
| • | seize or detain products or require a product recall. |
For example, the FDA informed us that certain adverse event reports related to DepoCyt(e) and DepoDur submitted to us during the previous two years were not submitted by us to the FDA within the required 15-day timeframe for reporting such events. In response to the FDA’s observations, we enhanced our reporting procedures and hired additional personnel to support our pharmacovigilance efforts.
If we fail to comply with federal and state healthcare laws, including fraud and abuse and health information privacy and security laws, we could face substantial penalties and our business, results of operations, financial condition and prospects could be adversely affected.
As a pharmaceutical company, even though we do not and will not control referrals of healthcare services or bill directly to Medicare, Medicaid or other third-party payers, certain federal and state healthcare laws and regulations pertaining to fraud and abuse and patients’ rights are and will be applicable to our business. We would be subject to healthcare fraud and abuse and patient privacy regulation by both the federal government and the states in which we conduct our business. The laws that may affect our ability to operate include:
| • | the federal Anti-Kickback Statute, which constrains our marketing practices, educational programs, pricing policies, and relationships with healthcare providers or other entities, by prohibiting, among other things, soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce, or in return for, the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
| • | federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payers that are false or fraudulent; |
38
Table of Contents
| • | the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created new federal criminal statutes that prohibit executing a scheme to defraud any healthcare benefit program and making false statements relating to healthcare matters; |
| • | federal physician self-referral laws, such as the Stark law, which prohibit a physician from making a referral to a provider of certain health services with which the physician or the physician’s family member has a financial interest, and prohibit submission of a claim for reimbursement pursuant to a prohibited referral; |
| • | HIPAA, as amended by the Health Information Technology and Clinical Health Act, or HITECH, and its implementing regulations, which imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information; and |
| • | state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws which may apply to items or services reimbursed by any third-party payer, including commercial insurers, and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. |
Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available under the U.S. federal Anti-Kickback Statute, it is possible that some of our business activities could be subject to challenge under one or more of such laws. Recently, several pharmaceutical and other healthcare companies have been prosecuted under the federal false claims laws for allegedly inflating drug prices they report to pricing services, which in turn are used by the government to set Medicare and Medicaid reimbursement rates, and for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. In addition, certain marketing practices, including off-label promotion, may also violate false claims laws. To the extent that any product we make is sold in a foreign country, we may be subject to similar foreign laws and regulations. If we or our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from participation in U.S. federal or state health care programs, and the curtailment or restructuring of our operations. Any penalties, damages, fines, curtailment or restructuring of our operations could materially adversely affect our ability to operate our business and our financial results. Although compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, the risks cannot be entirely eliminated. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. Moreover, achieving and sustaining compliance with applicable federal and state privacy, security and fraud laws may prove costly.
The design, development, manufacture, supply, and distribution of DepoCyt(e) and DepoDur is highly regulated and technically complex.
The design, development, manufacture, supply, and distribution of our products DepoCyt(e) and DepoDur is technically complex and highly regulated. We, along with our third-party providers, must comply with all applicable regulatory requirements of the FDA and foreign authorities. In addition, the facilities used to manufacture, store, and distribute our products are subject to inspection by regulatory authorities at any time to determine compliance with applicable regulations.
The manufacturing techniques and facilities used for the manufacture and supply of our products must be operated in conformity with cGMP. In complying with cGMP requirements, we, along with our suppliers, must continually expend time, money and effort in production, record keeping, and quality assurance and control to ensure that our products meet applicable specifications and other requirements for safety, efficacy and quality. In addition, we, along with our suppliers, are subject to unannounced inspections by the FDA and other regulatory authorities.
39
Table of Contents
Any failure to comply with regulatory and other legal requirements applicable to the manufacture, supply and distribution of our products could lead to remedial action (such as recalls), civil and criminal penalties and delays in manufacture, supply and distribution of our products. For instance, in connection with routine inspections of one of our manufacturing facilities in April and May 2008, the FDA issued a Form 483 Notice of Inspectional Observations identifying certain deficiencies with respect to our laboratory control system for Depocyt(e). As a result, we did not release new lots of Depocyt(e) for a limited time period as we validated a new assay. We also submitted the new assay to the FDA in July 2008 and in August 2008 we began releasing new lots of DepoCyt(e).
If we fail to comply with the extensive regulatory requirements to which we and our products DepoCyt(e) and DepoDur are subject, such products could be subject to restrictions or withdrawal from the market and we could be subject to penalties.
The testing, manufacturing, labeling, safety, advertising, promotion, storage, sales, distribution, export and marketing, among other things, of our products DepoCyt(e) and DepoDur are subject to extensive regulation by governmental authorities in the United States and elsewhere throughout the world. Quality control and manufacturing procedures regarding DepoCyt(e) and DepoDur must conform to cGMP. Regulatory authorities, including the FDA, periodically inspect manufacturing facilities to assess compliance with cGMP. Our failure or the failure of our contract manufacturers to comply with the laws administered by the FDA or other governmental authorities could result in, among other things, any of the following:
| • | product recall or seizure; |
| • | suspension or withdrawal of an approved product from the market; |
| • | interruption of production; |
| • | operating restrictions; |
| • | warning letters; |
| • | injunctions; |
| • | fines and other monetary penalties; |
| • | criminal prosecutions; and |
| • | unanticipated expenditures. |
If the government or third-party payers fail to provide coverage and adequate coverage and payment rates for DepoCyt(e), DepoDur, EXPAREL or any future products we may develop, license or acquire, if any, or if hospitals choose to use therapies that are less expensive, our revenue and prospects for profitability will be limited.
In both domestic and foreign markets, sales of our existing products and any future products will depend in part upon the availability of coverage and reimbursement from third-party payers. Such third-party payers include government health programs such as Medicare and Medicaid, managed care providers, private health insurers and other organizations. Coverage decisions may depend upon clinical and economic standards that disfavor new drug products when more established or lower cost therapeutic alternatives are already available or subsequently become available. Assuming coverage is approved, the resulting reimbursement payment rates might not be adequate. In particular, many U.S. hospitals receive a fixed reimbursement amount per procedure for certain surgeries and other treatment therapies they perform. Because this amount may not be based on the actual expenses the hospital incurs, hospitals may choose to use therapies which are less expensive when compared to our product candidates. Accordingly, DepoCyt(e), DepoDur, EXPAREL or any other product candidates that we may develop, in-license or acquire, if approved, will face competition from other therapies and drugs for these limited hospital financial resources. We may need to conduct post-marketing studies in order to demonstrate the cost-effectiveness of any
40
Table of Contents
future products to the satisfaction of hospitals, other target customers and their third-party payers. Such studies might require us to commit a significant amount of management time and financial and other resources. Our future products might not ultimately be considered cost-effective. Adequate third-party coverage and reimbursement might not be available to enable us to maintain price levels sufficient to realize an appropriate return on investment in product development.
Third party payers, whether foreign or domestic, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In addition, in the United States, no uniform policy of coverage and reimbursement for drug products exists among third-party payers. Therefore, coverage and reimbursement for drug products can differ significantly from payer to payer.
Further, we believe that future coverage and reimbursement will likely be subject to increased restrictions both in the United States and in international markets. Third-party coverage and reimbursement for our products or product candidates for which we receive regulatory approval may not be available or adequate in either the United States or international markets, which could have a negative effect on our business, results of operations, financial condition and prospects.
The FDA may not approve our proposed trade name, EXPAREL.
EXPAREL, or any other trade name that we intend to use for extended-release liposome injection of bupivacaine, must be approved by the FDA irrespective of whether we have secured a formal trademark registration from the U.S. Patent and Trademark Office. The FDA conducts a rigorous review of proposed product names, and may reject a product name if it believes that the name inappropriately implies medical claims or if it poses the potential for confusion with other product names. The FDA will not approve this trade name until the NDA for EXPAREL is approved. If the FDA determines that the trade names of other products that are approved prior to the approval of extended-release liposome injection of bupivacaine may present a risk of confusion with our proposed trade name, the FDA may not ultimately approve EXPAREL. If our trade name, EXPAREL, is rejected, we will lose the benefit of any brand equity that may already have been developed for this product candidate, as well as the benefit of our existing trademark applications for this trade name. If the FDA does not approve the EXPAREL trade name, we may be required to launch this product candidate without a brand name, and our efforts to build a successful brand identity for, and commercialize, this product candidate may be adversely impacted.
We are subject to new legislation, regulatory proposals and healthcare payer initiatives that may increase our costs of compliance and adversely affect our ability to market our products, obtain collaborators and raise capital.
In March 2010, the President signed the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, which we refer to collectively as the Health Care Reform Law. The Health Care Reform Law makes extensive changes to the delivery of health care in the United States. Among the provisions of the Health Care Reform Law of greatest importance to the pharmaceutical industry are the following:
| • | an annual, nondeductible fee on any entity that manufactures or imports certain branded prescription drugs and biologic agents, apportioned among these entities according to their market share in certain government healthcare programs, beginning in 2011; |
| • | an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program, retroactive to January 1, 2010, to 23.1% and 13% of the average manufacturer price for most branded and generic drugs, respectively; |
| • | a new Medicare Part D coverage gap discount program, in which manufacturers must agree to offer 50% point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D, beginning in 2011; |
41
Table of Contents
| • | extension of manufacturers’ Medicaid rebate liability to covered drugs dispensed to individuals who are enrolled in Medicaid managed care organizations, effective March 23, 2010; |
| • | expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to additional individuals beginning in April 2010 and by adding new mandatory eligibility categories for certain individuals with income at or below 133% of the Federal Poverty Level beginning in 2014, thereby potentially increasing both the volume of sales and manufacturers’ Medicaid rebate liability; |
| • | expansion of the entities eligible for discounts under the Public Health Service pharmaceutical pricing program, effective in January 2010; |
| • | new requirements to report certain financial arrangements with physicians and others, including reporting any “transfer of value” made or distributed to prescribers and other healthcare providers and reporting any investment interests held by physicians and their immediate family members during each calendar year beginning in 2012, with reporting starting in 2013; |
| • | a new requirement to annually report drug samples that manufacturers and distributors provide to physicians, effective April 1, 2012; |
| • | a licensure framework for follow-on biologic products; |
| • | a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research; |
| • | creation of the Independent Payment Advisory Board which, beginning in 2014, will have authority to recommend certain changes to the Medicare program that could result in reduced payments for prescription drugs and those recommendations could have the effect of law even if Congress does not act on the recommendations; and |
| • | establishment of a Center for Medicare Innovation at the Centers for Medicare & Medicaid Services to test innovative payment and service delivery models to lower Medicare and Medicaid spending, potentially including prescription drug spending, beginning by January 1, 2011. |
These measures could result in decreased net revenues from our pharmaceutical products and decrease potential returns from our development efforts. Many of the details regarding the implementation of the Health Care Reform Law are yet to be determined, and at this time, the full effect that the Health Care Reform Law would have on our business remains unclear.
In addition, there have been a number of other legislative and regulatory proposals aimed at changing the pharmaceutical industry. In particular, California has enacted legislation that requires development of an electronic pedigree to track and trace each prescription drug at the saleable unit level through the distribution system. California’s electronic pedigree requirement is scheduled to take effect in January 2015. Compliance with California and future federal or state electronic pedigree requirements may increase our operational expenses and impose significant administrative burdens. As a result of these and other new proposals, we may determine to change our current manner of operation, provide additional benefits or change our contract arrangements, any of which could have a material adverse effect on our business, financial condition and results of operations.
Public concern regarding the safety of drug products such as EXPAREL could delay or limit our ability to obtain regulatory approval, result in the inclusion of unfavorable information in our labeling, or require us to undertake other activities that may entail additional costs.
In light of widely publicized events concerning the safety risk of certain drug products, the FDA, members of Congress, the Government Accountability Office, medical professionals and the general public have raised concerns about potential drug safety issues. These events have resulted in the withdrawal of drug products, revisions
42
Table of Contents
to drug labeling that further limit use of the drug products and the establishment of risk management programs that may, for example, restrict distribution of drug products after approval. The Food and Drug Administration Amendments Act of 2007, or FDAAA, grants significant expanded authority to the FDA, much of which is aimed at improving the safety of drug products before and after approval. In particular, the FDAAA authorizes the FDA to, among other things, require post-approval studies and clinical trials, mandate changes to drug labeling to reflect new safety information and require risk evaluation and mitigation strategies for certain drugs, including certain currently approved drugs. It also significantly expands the federal government’s clinical trial registry and results databank, which we expect will result in significantly increased government oversight of clinical trials. Under the FDAAA, companies that violate these and other provisions of the new law are subject to substantial civil monetary penalties, among other regulatory, civil and criminal penalties. The increased attention to drug safety issues may result in a more cautious approach by the FDA in its review of data from our clinical trials. Data from clinical trials may receive greater scrutiny, particularly with respect to safety, which may make the FDA or other regulatory authorities more likely to require additional preclinical studies or clinical trials. If the FDA requires us to conduct additional preclinical studies or clinical trials prior to approving EXPAREL, our ability to obtain approval of this product candidate will be delayed. If the FDA requires us to provide additional clinical or preclinical data following the approval of EXPAREL, the indications for which this product candidate is approved may be limited or there may be specific warnings or limitations on dosing, and our efforts to commercialize EXPAREL may be otherwise adversely impacted.
Our product, DepoDur, is subject to regulation by the Drug Enforcement Agency and such regulation may affect the sale of DepoDur.
Products used to treat and manage pain, especially in the case of opioids, are from time to time subject to negative publicity, including illegal use, overdoses, abuse, diversion, serious injury and death. These events have led to heightened regulatory scrutiny. Controlled substances are classified by the DEA as Schedule I through V substances, with Schedule I substances being prohibited for sale in the United States, Schedule II substances considered to present the highest risk of abuse and Schedule V substances being considered to present the lowest relative risk of abuse. DepoDur contains morphine, and it is regulated as a Schedule II controlled substance. Despite the strict regulations on the marketing, prescribing and dispensing of such substances, illicit use and abuse of morphine does occur. Thus, the marketing of DepoDur by our partners may generate public controversy that may adversely affect sales of DepoDur and decrease the revenue we receive from the sale of DepoDur.
In addition, we and our contract manufacturers are subject to ongoing DEA regulatory obligations, including, among other things, annual registration renewal, security, recordkeeping, theft and loss reporting, periodic inspection and annual quota allotments for the raw material for commercial production of our products. The DEA, and some states, conduct periodic inspections of registered establishments that handle controlled substances. Facilities that conduct research, manufacture, store, distribute, import or export controlled substances must be registered to perform these activities and have the security, control and inventory mechanisms required by the DEA to prevent drug loss and diversion. Failure to maintain compliance, particularly non-compliance resulting in loss or diversion, can result in regulatory action that could have a material adverse effect on our business, results of operations, financial condition and prospects. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate proceedings to revoke those registrations. In certain circumstances, violations could lead to criminal proceedings.
Individual states also have controlled substances laws. Though state controlled substances laws often mirror federal law, because the states are separate jurisdictions, they may separately schedule drugs, as well. While some states automatically schedule a drug when the DEA does so, in other states there has to be rulemaking or a legislative action. State scheduling may delay commercial sale of any controlled substance drug product for which we obtain federal regulatory approval and adverse scheduling could have a material adverse effect on the commercial attractiveness of such product. We or our partners must also obtain separate state registrations in order to be able to obtain, handle, and distribute controlled substances for clinical trials or commercial sale, and failure to meet applicable regulatory requirements could lead to enforcement and sanctions from the states in addition to those from the DEA or otherwise arising under federal law.
43
Table of Contents
Risks Related to Intellectual Property
The patents and the patent applications that we have covering our products are limited to specific injectable formulations, processes and uses of drugs encapsulated in our DepoFoam drug delivery technology and our market opportunity for our product candidates may be limited by the lack of patent protection for the active ingredient itself and other formulations and delivery technology and systems that may be developed by competitors.
The active ingredients in EXPAREL, DepoCyt(e) and DepoDur are bupivacaine, cytarabine and morphine, respectively. Patent protection for the bupivacaine, cytarabine and morphine molecules themselves has expired and generic immediate-release products are available. As a result, competitors who obtain the requisite regulatory approval can offer products with the same active ingredients as EXPAREL, DepoCyt(e) and DepoDur so long as the competitors do not infringe any process, use or formulation patents that we have developed for these drugs encapsulated in our DepoFoam drug delivery technology.
For example, we are aware of at least one long acting injectable bupivacaine product in development which utilizes an alternative delivery system to EXPAREL. Such a product is similar to EXPAREL in that it also extends the duration of effect of bupivacaine, but achieves this clinical outcome using a completely different drug delivery system compared to our DepoFoam drug delivery technology.
The number of patents and patent applications covering products in the same field as EXPAREL indicates that competitors have sought to develop and may seek to market competing formulations that may not be covered by our patents and patent applications. The commercial opportunity for EXPAREL could be significantly harmed if competitors are able to develop and commercialize alternative formulations of bupivacaine that are long acting but outside the scope of our patents.
If EXPAREL is approved by the FDA, one or more third parties may challenge the patents covering this product, which could result in the invalidation or unenforceability of some or all of the relevant patent claims. For example, if a third party files an Abbreviated New Drug Application, or ANDA, for a generic drug product containing bupivacaine and relies in whole or in part on studies conducted by or for us, the third party will be required to certify to the FDA that either: (1) there is no patent information listed in the FDA’s Orange Book with respect to our NDA for EXPAREL; (2) the patents listed in the Orange Book have expired; (3) the listed patents have not expired, but will expire on a particular date and approval is sought after patent expiration; or (4) the listed patents are invalid or will not be infringed by the manufacture, use or sale of the third-party’s generic drug product. A certification that the new product will not infringe the Orange Book-listed patents for EXPAREL, or that such patents are invalid, is called a paragraph IV certification. If the third party submits a paragraph IV certification to the FDA, a notice of the paragraph IV certification must also be sent to us once the third-party’s ANDA is accepted for filing by the FDA. We may then initiate a lawsuit to defend the patents identified in the notice. The filing of a patent infringement lawsuit within 45 days of receipt of the notice automatically prevents the FDA from approving the third-party’s ANDA until the earliest of 30 months or the date on which the patent expires, the lawsuit is settled, or the court reaches a decision in the infringement lawsuit in favor of the third party. If we do not file a patent infringement lawsuit within the required 45-day period, the third-party’s ANDA will not be subject to the 30-month stay. Litigation or other proceedings to enforce or defend intellectual property rights are often very complex in nature, may be very expensive and time-consuming, may divert our management’s attention from our core business, and may result in unfavorable results that could adversely impact our ability to prevent third parties from competing with our products.
Because it is difficult and costly to protect our proprietary rights, we may not be able to ensure their protection and all patents will eventually expire.
Our commercial success will depend in part on obtaining and maintaining patent protection and trade secret protection for EXPAREL, DepoCyt(e), DepoDur, DepoFoam and for any other product candidates that we may develop, license or acquire and the methods we use to manufacture them, as well as successfully defending these patents and trade secrets against third-party challenges. We will only be able to protect our technologies from unauthorized use by third parties to the extent that valid and enforceable patents or trade secrets cover them.
44
Table of Contents
The patent positions of pharmaceutical and biotechnology companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. No consistent policy regarding the breadth of claims allowed in pharmaceutical or biotechnology patents has emerged to date in the United States. The patent situation outside the United States is even more uncertain. Changes in either the patent laws or in interpretations of patent laws in the United States and other countries may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in our patents or in third-party patents.
The degree of future protection for our proprietary rights is uncertain, because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
| • | we may not have been the first to make the inventions covered by each of our pending patent applications and issued patents; |
| • | we may not have been the first to file patent applications for these inventions; |
| • | others may independently develop similar or alternative technologies or duplicate any of our product candidates or technologies; |
| • | it is possible that none of the pending patent applications will result in issued patents; |
| • | the issued patents covering our product candidates may not provide a basis for commercially viable active products, may not provide us with any competitive advantages, or may be challenged by third parties; |
| • | we may not develop additional proprietary technologies that are patentable; or |
| • | patents of others may have an adverse effect on our business. |
Patent applications in the United States are maintained in confidence for at least 18 months after their earliest effective filing date. Consequently, we cannot be certain we were the first to invent or the first to file patent applications on EXPAREL, our DepoFoam drug delivery technology or any other product candidates that we may develop, license or acquire. In the event that a third party has also filed a U.S. patent application relating to our product candidates or a similar invention, we may have to participate in interference proceedings declared by the U.S. Patent and Trademark Office to determine priority of invention in the United States. The costs of these proceedings could be substantial and it is possible that our efforts would be unsuccessful, resulting in a material adverse effect on our U.S. patent position. Furthermore, we may not have identified all United States and foreign patents or published applications that affect our business either by blocking our ability to commercialize our drugs or by covering similar technologies that affect our drug market.
In addition, some countries, including many in Europe, do not grant patent claims directed to methods of treating humans, and in these countries patent protection may not be available at all to protect our product candidates. Even if patents issue, we cannot guarantee that the claims of those patents will be valid and enforceable or provide us with any significant protection against competitive products, or otherwise be commercially valuable to us.
Some of our older patents have already expired. In the cases of DepoCyt(e) and DepoDur, key patents providing protection in Europe have expired. In the case of EXPAREL, while pending patent applications, if granted, would provide protection for EXPAREL in Europe and the United States through November 2018, an existing formulation patent for EXPAREL will expire in November 2013. Once our patents covering EXPAREL have expired, we are more reliant on trade secrets to protect against generic competition.
We also rely on trade secrets to protect our technology, particularly where we do not believe patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. While we use reasonable efforts to protect our trade secrets, our licensors, employees, consultants, contractors, outside scientific collaborators
45
Table of Contents
and other advisors may unintentionally or willfully disclose our information to competitors. Enforcing a claim that a third party illegally obtained and is using our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, courts outside the United States are sometimes less willing to protect trade secrets. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how.
If we fail to obtain or maintain patent protection or trade secret protection for EXPAREL, DepoCyt(e), DepoDur, DepoFoam or any other product candidate that we may develop, license or acquire, third parties could use our proprietary information, which could impair our ability to compete in the market and adversely affect our ability to generate revenues and achieve profitability.
If we are sued for infringing intellectual property rights of third parties, it will be costly and time consuming, and an unfavorable outcome in any litigation would harm our business.
Our ability to develop, manufacture, market and sell EXPAREL, our DepoFoam drug delivery technology or any other product candidates that we may develop, license or acquire depends upon our ability to avoid infringing the proprietary rights of third parties. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the general fields of pain management and cancer treatment and cover the use of numerous compounds and formulations in our targeted markets. Because of the uncertainty inherent in any patent or other litigation involving proprietary rights, we and our licensors may not be successful in defending intellectual property claims by third parties, which could have a material adverse affect on our results of operations. Regardless of the outcome of any litigation, defending the litigation may be expensive, time-consuming and distracting to management. In addition, because patent applications can take many years to issue, there may be currently pending applications, unknown to us, which may later result in issued patents that EXPAREL, DepoCyt(e) or DepoDur may infringe. There could also be existing patents of which we are not aware that EXPAREL, DepoCyt(e) or DepoDur may inadvertently infringe.
There is a substantial amount of litigation involving patent and other intellectual property rights in the biotechnology and biopharmaceutical industries generally. If a third party claims that we infringe on their products or technology, we could face a number of issues, including:
| • | infringement and other intellectual property claims which, with or without merit, can be expensive and time consuming to litigate and can divert management’s attention from our core business; |
| • | substantial damages for past infringement which we may have to pay if a court decides that our product infringes on a competitor’s patent; |
| • | a court prohibiting us from selling or licensing our product unless the patent holder licenses the patent to us, which it would not be required to do; |
| • | if a license is available from a patent holder, we may have to pay substantial royalties or grant cross licenses to our patents; and |
| • | redesigning our processes so they do not infringe, which may not be possible or could require substantial funds and time. |
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
As is common in the biotechnology and pharmaceutical industry, we employ individuals who were previously employed at other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although no claims against us are currently pending, we may be subject to claims that these employees or we have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management.
46
Table of Contents
Risks Related to Our Financial Condition and Capital Requirements
We have incurred significant losses since our inception and anticipate that we will incur continued losses for the foreseeable future.
We are an emerging specialty pharmaceutical company with a limited operating history. We have focused primarily on developing EXPAREL with the goal of achieving regulatory approval. We have incurred losses in each year since our inception in December 2006, including net losses of $27.1 million, $31.7 million and $41.9 million for the years ended December 31, 2010, 2009 and 2008, respectively. As of December 31, 2010, we had an accumulated deficit of $136.9 million. These losses, among other things, have had, and will continue to have, an adverse effect on our stockholders’ equity (deficit) and working capital. We incurred increased pre-commercialization expenses during 2009 and 2010 as we prepared for the potential market launch of EXPAREL, and we expect to incur significant sales, marketing and manufacturing expenses, as well as continued development expenses related to the commercialization of EXPAREL, if approved by the FDA. As a result, we expect to continue to incur significant losses for the foreseeable future. Because of the numerous risks and uncertainties associated with developing pharmaceutical products, we are unable to predict the extent of any future losses or when we will become profitable, if at all.
We may never become profitable.
Our ability to become profitable depends upon our ability to generate revenue from EXPAREL and to continue to generate revenue from DepoCyt(e) and DepoDur. Our ability to generate revenue depends on a number of factors, including, but not limited to, our ability to:
| • | continue to manufacture DepoCyt(e) and DepoDur for sale by our commercial partners; |
| • | obtain regulatory approval for EXPAREL, or any other product candidates that we may develop, license or acquire; |
| • | manufacture commercial quantities of EXPAREL, if approved, at acceptable cost levels; and |
| • | develop a commercial organization and the supporting infrastructure required to successfully market and sell EXPAREL, if it is approved. |
If EXPAREL is approved for commercial sale, we anticipate incurring significant costs associated with its commercialization. We also do not anticipate that we will achieve profitability for a period of time after generating material revenues, if ever. If we are unable to generate revenues, we will not become profitable and may be unable to continue operations without continued funding.
We will need to raise additional financing to continue as a going concern and may be unable to raise capital when needed
We have incurred losses in each year since our inception in December 2006, including net losses of $27.1 million, $31.7 million and $41.9 million for the years ended December 31, 2010, 2009 and 2008, respectively and as of December 31, 2010, we had an accumulated deficit of $136.9 million. The Company has raised $42 million of gross proceeds, before offering costs, through an initial public offering completed on February 8, 2011. Although the offering and our cash resources provide the Company adequate funding for the next 12 months, the longer-term ability of the Company to continue as a going concern is dependent on improving the Company’s profitability and cash flow and securing additional financing. We may not be able to raise finances in time or on favorable terms when needed and our efforts to continue as a going concern may not prove successful.
Under our financing arrangement with Paul Capital, upon the occurrence of certain events, Paul Capital may require us to repurchase the right to receive royalty payments that we assigned to it, or may foreclose on certain assets that secure our obligations to Paul Capital. Any exercise by Paul Capital of its right to cause us to repurchase the assigned right or any foreclosure by Paul Capital would adversely affect our results of operations and our financial condition.
47
Table of Contents
On March 23, 2007, we entered into the Amended and Restated Royalty Interests Assignment Agreement with affiliates of Paul Capital, pursuant to which we assigned to Paul Capital the right to receive a portion of our royalty payments from DepoCyt(e) and DepoDur. To secure our obligations to Paul Capital, we granted Paul Capital a security interest in collateral which includes the royalty payments we are entitled to receive with respect to sales of DepoCyt(e) and DepoDur, as well as to bank accounts to which such payments are deposited. Under our arrangement with Paul Capital, upon the occurrence of certain events, or the put events, including if we experience a change of control, we or our subsidiary undergo certain bankruptcy events, transfer any or substantially all of our rights in DepoCyt(e) or DepoDur, transfer all or substantially all of our assets, breach certain of the covenants, representations or warranties under the Amended and Restated Royalty Interests Assignment Agreement, or sales of DepoCyt(e) or DepoDur are suspended due to an injunction or if we elect to suspend sales of DepoCyt(e) or DepoDur as a result of a lawsuit filed by certain third parties, Paul Capital may (i) require us to repurchase the rights we assigned to it at a cash price equal to (a) 50% of all cumulative payments made by us to Paul Capital under the Amended and Restated Royalty Interests Assignment Agreement during the preceding 24 months, multiplied by (b) the number of days from the date of Paul Capital’s exercise of such option until December 31, 2014, divided by 365. Any exercise by Paul Capital of its right to cause us to repurchase the assigned right or any foreclosure by Paul Capital would adversely affect our results of operations and our financial condition.
Our debt obligations expose us to risks that could adversely affect our business, operating results and financial condition.
We have a substantial level of debt. At December 31, 2010, after giving effect to the Hercules Credit Facility and the conversion of our other outstanding debt into common stock in connection with the completion of our initial public offering in February 2011, we had $26.25 million in aggregate principal amount of indebtedness outstanding, not including our obligation under the Amended and Restated Royalty Interests Assignment Agreement with Paul Capital. The level and nature of our indebtedness, among other things, could:
| • | make it difficult for us to make payments on our outstanding debt from time to time or to refinance it; |
| • | make it difficult for us to obtain any necessary financing in the future for working capital, capital expenditures, debt service, product and company acquisitions or general corporate purposes; |
| • | limit our flexibility in planning for or reacting to changes in our business including life cycle management; |
| • | reduce funds available for use in our operations; |
| • | impair our ability to incur additional debt because of financial and other restrictive covenants; |
| • | make us more vulnerable in the event of a downturn in our business; |
| • | place us at a possible competitive disadvantage relative to less leveraged competitors and competitors that have better access to capital resources; |
| • | restrict the operations of our business as a result of provisions in the Amended and Restated Royalty Interests Assignment Agreement with Paul Capital that restrict our ability to (i) amend, waive any rights under, or terminate any material agreements relating to DepoCyt(e) and DepoDur, (ii) enter into any new agreement or amend or fail to exercise any of our material rights under existing agreements that would materially adversely affect Paul Capital’s royalty interest, and (iii) sell any material assets related to DepoCyt(e) or DepoDur; or |
| • | impair our ability to merge or otherwise effect the sale of the Company due to the right of the holders of certain of our indebtedness to accelerate the maturity date of the indebtedness in the event of a change of control of the Company. |
48
Table of Contents
We will need to raise additional capital to pay our indebtedness as it comes due. If we are unable to obtain funds necessary to make required payments, or if we fail to comply with the various requirements of our indebtedness, we would be in default, which would permit the holders of our indebtedness to accelerate the maturity of the indebtedness and could cause defaults under any indebtedness we may incur in the future. Any default under our indebtedness would have a material adverse effect on our business, operating results and financial condition. If we are unable to refinance or repay our indebtedness as it becomes due, we may become insolvent and be unable to continue operations.
For example, our loan and security agreement governing the Hercules Credit Facility, contains a number of affirmative and restrictive covenants, including reporting requirements and other collateral limitations, certain limitations on liens and indebtedness, dispositions, mergers and acquisitions, restricted payments and investments, corporate changes and limitations on waivers and amendments to certain agreements, our organizational documents, and documents relating to debt that is subordinate to our obligations under the Hercules Credit Facility. Our failure to comply with the covenants in the loan and security agreement governing the Hercules Credit Facility could result in an event of default that, if not cured or waived, could result in the acceleration of all or a substantial portion of our debt and potential foreclosure on the assets pledged to secure the debt.
Our short operating history makes it difficult to evaluate our business and prospects.
We were incorporated in December 2006 and have only been conducting operations with respect to EXPAREL since March 2007. Our operations to date have been limited to organizing and staffing our company, conducting product development activities, including clinical trials and manufacturing development activities, for EXPAREL and manufacturing and related activities for DepoCyt(e) and DepoDur. Further, in 2010 we began to establish our commercial infrastructure for EXPAREL. We have not yet demonstrated an ability to obtain regulatory approval for or successfully commercialize a product candidate. Consequently, any predictions about our future performance may not be as accurate as they could be if we had a history of successfully developing and commercializing pharmaceutical products.
We will need additional funding and may be unable to raise capital when needed, which would force us to delay, reduce or eliminate our product development programs or commercialization efforts.
Developing products for use in the hospital setting, conducting clinical trials, establishing outsourced manufacturing relationships and successfully manufacturing and marketing drugs that we may develop is expensive. We will need to raise additional capital to:
| • | fund our operations and continue our efforts to hire additional personnel and build a commercial infrastructure to prepare for the commercialization of EXPAREL, if approved by the FDA; |
| • | qualify and outsource the commercial-scale manufacturing of our products under cGMP; and |
| • | in-license and develop additional product candidates. |
Throughout 2009 and 2010, we generated net proceeds of approximately $47.5 million through several private placements of secured and unsecured notes and proceeds of approximately $26.25 million under the Hercules Credit Facility. We believe that with our currently available cash and cash equivalent balance, along with the net proceeds from our initial public offering, we have sufficient funds to meet our projected operating requirements and service our indebtedness for at least the next 12 months. We have based this estimate on assumptions that may prove to be wrong and we could spend our available financial resources faster than we currently expect. Further, we may not have sufficient financial resources to meet all of our objectives if EXPAREL is approved, which could require us to postpone, scale back or eliminate some, or all, of these objectives, including our potential launch activities. Our future funding requirements will depend on many factors, including, but not limited to:
| • | the potential for delays in our efforts to seek regulatory approval for EXPAREL, and any costs associated with such delays; |
49
Table of Contents
| • | the costs of establishing a commercial organization to sell, market and distribute EXPAREL; |
| • | the rate of progress and costs of our efforts to prepare for the submission of an NDA for any product candidates that we may in-license or acquire in the future, and the potential that we may need to conduct additional clinical trials to support applications for regulatory approval; |
| • | the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights associated with our product candidates, including any such costs we may be required to expend if our licensors are unwilling or unable to do so; |
| • | the cost and timing of manufacturing sufficient supplies of EXPAREL in preparation for commercialization; |
| • | the effect of competing technological and market developments; |
| • | the terms and timing of any collaborative, licensing, co-promotion or other arrangements that we may establish; |
| • | if EXPAREL is approved, the potential that we may be required to file a lawsuit to defend our patent rights or regulatory exclusivities from challenges by companies seeking to market generic versions of extended-release liposome injection of bupivacaine; and |
| • | the success of the commercialization of EXPAREL. |
Future capital requirements will also depend on the extent to which we acquire or invest in additional complementary businesses, products and technologies.
Until we can generate a sufficient amount of product revenue, if ever, we expect to finance future cash needs through public or private equity offerings, debt financings, product supply revenue and royalties, corporate collaboration and licensing arrangements, as well as through interest income earned on cash and investment balances. We cannot be certain that additional funding will be available on acceptable terms, or at all. If adequate funds are not available, we may be required to delay, reduce the scope of, or eliminate, one or more of our development programs or our commercialization efforts.
Our quarterly operating results may fluctuate significantly.
We expect our operating results to be subject to quarterly fluctuations. Our net loss and other operating results will be affected by numerous factors, including:
| • | whether the FDA requires us to complete additional, unanticipated studies, tests or other activities prior to approving EXPAREL, which would likely further delay any such approval; |
| • | if EXPAREL is approved, our ability to establish the necessary commercial infrastructure to launch this product candidate without substantial delays, including hiring sales and marketing personnel and contracting with third parties for warehousing, distribution, cash collection and related commercial activities; |
| • | maintaining our existing manufacturing facilities and expanding our manufacturing capacity, including installing specialized processing equipment for the manufacturing of EXPAREL; |
| • | our execution of other collaborative, licensing or similar arrangements and the timing of payments we may make or receive under these arrangements; |
| • | variations in the level of expenses related to our future development programs; |
50
Table of Contents
| • | any product liability or intellectual property infringement lawsuit in which we may become involved; |
| • | regulatory developments affecting EXPAREL or the product candidates of our competitors; and |
| • | if EXPAREL receives regulatory approval, the level of underlying hospital demand for this product candidate and wholesaler buying patterns. |
If our quarterly or annual operating results fall below the expectations of investors or securities analysts, the price of our common stock could decline substantially. Furthermore, any quarterly or annual fluctuations in our operating results may, in turn, cause the price of our stock to fluctuate substantially. We believe that quarterly comparisons of our financial results are not necessarily meaningful and should not be relied upon as an indication of our future performance.
Raising additional funds by issuing securities may cause dilution to existing stockholders and raising funds through lending and licensing arrangements may restrict our operations or require us to relinquish proprietary rights.
To the extent that we raise additional capital by issuing equity securities, our existing stockholders’ ownership will be diluted. If we raise additional funds through licensing arrangements, it may be necessary to relinquish potentially valuable rights to our potential products or proprietary technologies, or grant licenses on terms that are not favorable to us. Any debt financing we enter into may involve covenants that restrict our operations. These restrictive covenants may include limitations on additional borrowing and specific restrictions on the use of our assets as well as prohibitions on our ability to create liens, pay dividends, redeem our stock or make investments.
We incur significant increased costs as a result of operating as a public company.
As a public company, we incur significant legal, accounting, insurance and other expenses that we did not incur as a private company, including costs associated with public company reporting requirements. We also have incurred and will incur costs associated with complying with the requirements of the Sarbanes-Oxley Act of 2002 and related rules implemented by the Securities and Exchange Commission and The NASDAQ Global Market. The expenses incurred by public companies generally for reporting and corporate governance purposes have been increasing. We expect these rules and regulations to increase our legal and financial compliance costs and to make some activities more time-consuming and costly, although we are currently unable to estimate these costs with any degree of certainty. These laws and regulations could also make it more difficult or costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. These laws and regulations could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, our board committees or as our executive officers. Furthermore, if we are unable to satisfy our obligations as a public company, we could be subject to delisting of our common stock, fines, sanctions and other regulatory action and potentially civil litigation.
Compliance with Section 404 of the Sarbanes-Oxley Act of 2002 will require our management to devote substantial time to new compliance initiatives.
Pursuant to Section 404 of the Sarbanes-Oxley Act of 2002, or Section 404, we are required to furnish a report by our management on the effectiveness of our internal control over financial reporting. The internal control report must contain (i) a statement of management’s responsibility for establishing and maintaining adequate internal control over financial reporting, (ii) a statement identifying the framework used by management to conduct the required evaluation of the effectiveness of our internal control over financial reporting, and (iii) management’s assessment of the effectiveness of our internal control over financial reporting as of the end of our most recent fiscal year, including a statement as to whether or not internal control over financial reporting is effective.
To achieve compliance with Section 404 within the prescribed period is both costly and challenging. In this regard, we will need to dedicate resources, potentially engage outside consultants and adopt a detailed work plan to
51
Table of Contents
(i) assess and document the adequacy of internal control over financial reporting, (ii) continue steps to improve control processes where appropriate, (iii) validate through testing that controls are functioning as documented, and (iv) implement a continuous reporting and improvement process for internal control over financial reporting.
The use of our net operating loss carryforwards and research tax credits may be limited.
Our net operating loss carryforwards and research and development tax credits may expire and not be used. As of December 31, 2010, we had federal and state net operating loss carryforwards of approximately $111.8 million and $97.7 million, respectively, and we also had federal and state research and development tax credit carryforwards of approximately $2.5 million and $1.1 million, respectively. Our net operating loss carryforwards will begin expiring in 2026 for federal purposes and 2020 for state purposes if we have not used them prior to that time, and our federal tax credits will begin expiring in 2028 unless previously used. Our state tax credits carryforward indefinitely. Additionally, our ability to use any net operating loss and credit carryforwards to offset taxable income or tax, respectively, in the future will be limited under Internal Revenue Code Sections 382 and 383 if we have a cumulative change in ownership of more than 50% within a three-year period. In addition, California and certain states have suspended use of net operating loss carryforwards for certain taxable years, and other states are considering similar measures. As a result, we may incur higher state income tax expense in the future. Depending on our future tax position, continued suspension of our ability to use net operating loss carryforwards in states in which we are subject to income tax could have an adverse impact on our results of operations and financial condition.
Our results of operations and liquidity needs could be materially negatively affected by market fluctuations and economic downturn.
Our results of operations could be materially negatively affected by economic conditions generally, both in the United States and elsewhere around the world. Continuing concerns over inflation, energy costs, geopolitical issues, the availability and cost of credit, the U.S. mortgage market and a declining residential real estate market in the United States have contributed to increased volatility and diminished expectations for the economy and the markets going forward. These factors, combined with volatile oil prices, declining business and consumer confidence and increased unemployment, have precipitated an economic recession and fears of a possible depression. Domestic and international equity markets continue to experience heightened volatility and turmoil. These events and the continuing market upheavals may have an adverse effect on us. In the event of a continuing market downturn, our results of operations could be adversely affected by those factors in many ways, including making it more difficult for us to raise funds if necessary, and our stock price may further decline.
Risks Related to Ownership of Our Common Stock
The market price of our common stock may be highly volatile.
The trading price of our common stock is likely to be volatile. Our stock price could be subject to wide fluctuations in response to a variety of factors, including the following:
| • | any delay in the FDA approving our NDA for EXPAREL; |
| • | the commercial success of EXPAREL, if approved by the FDA; |
| • | results of clinical trials of our product candidates or those of our competitors; |
| • | changes or developments in laws or regulations applicable to our product candidates; |
| • | introduction of competitive products or technologies; |
| • | failure to meet or exceed financial projections we provide to the public; |
| • | actual or anticipated variations in quarterly operating results; |
52
Table of Contents
| • | failure to meet or exceed the estimates and projections of the investment community; |
| • | the perception of the pharmaceutical industry by the public, legislatures, regulators and the investment community; |
| • | general economic and market conditions and overall fluctuations in U.S. equity markets; |
| • | developments concerning our sources of manufacturing supply; |
| • | disputes or other developments relating to patents or other proprietary rights; |
| • | additions or departures of key scientific or management personnel; |
| • | issuances of debt, equity or convertible securities; |
| • | changes in the market valuations of similar companies; and |
| • | the other factors described in this “Risk Factors” section. |
In addition, the stock market in general, and the market for small pharmaceutical and biotechnology companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our actual operating performance.
Our principal stockholders and management own a significant percentage of our stock and will be able to exert significant control over matters subject to stockholder approval.
Our executive officers, directors and 5% stockholders and their affiliates beneficially own approximately 69.2% of our outstanding common stock. As a result, these stockholders will have significant influence and may be able to determine all matters requiring stockholder approval. For example, these stockholders may be able to control elections of directors, amendments of our organizational documents, or approval of any merger, sale of assets, or other major corporate transaction. This concentration of ownership could delay or prevent any acquisition of our company on terms that other stockholders may desire.
Sales of a substantial number of shares of our common stock in the public market by our existing stockholders could cause our stock price to fall.
Sales of a substantial number of shares of our common stock in the public market or the perception that these sales might occur, could depress the market price of our common stock and could impair our ability to raise adequate capital through the sale of additional equity securities. We are unable to predict the effect that sales may have on the prevailing market price of our common stock.
Substantially all of our existing stockholders are subject to lock-up agreements that restrict the stockholders’ ability to transfer shares of our common stock until August 1, 2011, subject to certain exceptions. The lock-up agreements limit the number of shares of common stock that may be sold prior to August 1, 2011. Subject to certain limitations, 12,218,769 shares will become eligible for sale on August 1, 2011. In addition, shares issued or issuable upon exercise of options and warrants vested as of the expiration of the lock-up period will be eligible for sale at that time. Sales of stock by these stockholders could have a material adverse effect on the market price of our common stock.
Certain holders of shares of our common stock are entitled to rights with respect to the registration of their shares under the Securities Act of 1933, as amended, or the Securities Act, subject to the lock-up arrangement described above. Registration of these shares under the Securities Act would result in the shares becoming freely tradable without restriction under the Securities Act, except for shares held by our affiliates as defined in Rule 144 under the Securities Act. Any sales of securities by these stockholders could have a material adverse effect on the trading price of our common stock.
53
Table of Contents
Because we do not intend to pay dividends on our common stock, your returns will be limited to any increase in the value of our stock.
We have never declared or paid any cash dividends on our capital stock. We currently intend to retain all available funds and any future earnings to support our operations and finance the growth and development of our business and do not anticipate declaring or paying any cash dividends on our common stock for the foreseeable future. Any return to stockholders will therefore be limited to the appreciation of their stock, if any.
Some provisions of our charter documents and Delaware law may have anti-takeover effects that could discourage an acquisition of us by others, even if an acquisition would be beneficial to our stockholders, and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our restated certificate of incorporation and our bylaws, as well as provisions of the Delaware General Corporation Law, or DGCL, could make it more difficult for a third party to acquire us or increase the cost of acquiring us, even if doing so would benefit our stockholders, including transactions in which stockholders might otherwise receive a premium for their shares. These provisions include:
| • | authorizing the issuance of “blank check” preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval; |
| • | prohibiting stockholder action by written consent, thereby requiring all stockholder actions to be taken at a meeting of our stockholders; |
| • | eliminating the ability of stockholders to call a special meeting of stockholders; and |
| • | establishing advance notice requirements for nominations for election to the board of directors or for proposing matters that can be acted upon at stockholder meetings. |
These provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors, which is responsible for appointing the members of our management. In addition, we are subject to Section 203 of the DGCL, which generally prohibits a Delaware corporation from engaging in any of a broad range of business combinations with an interested stockholder for a period of three years following the date on which the stockholder became an interested stockholder, unless such transactions are approved by our board of directors. This provision could have the effect of delaying or preventing a change of control, whether or not it is desired by or beneficial to our stockholders.
| Item 1B. | Unresolved Staff Comments |
Not applicable.
| Item 2. | Properties |
Our research and development and manufacturing facilities are located in San Diego, California, where we occupy two facilities totaling approximately 106,000 square feet under leases expiring in July 2015. We use these facilities for research and development, manufacturing and general and administrative purposes. In addition, we maintain our executive offices and our commercial and business development facility in Parsippany, New Jersey.
We believe that our manufacturing facilities are sufficient for our current needs. We intend to add new facilities or expand existing facilities as we add employees or expand our geographic markets, and we believe that suitable additional or substitute space will be available as needed to accommodate any such expansion of our operations.
54
Table of Contents
| Item 3. | Legal Proceedings |
From time to time, we have been and may again become involved in legal proceedings arising in the ordinary course of our business. We are not presently a party to any material litigation and we are not aware of any pending or threatened litigation against us that could have a material adverse effect on our business, operating results, financial condition or cash flows.
| Item 4. | (Removed and Reserved) |
55
Table of Contents
| Item 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
Market Information
Our common stock has been publicly traded on the NASDAQ Global Market under the symbol “PCRX” since our initial public offering on February 3, 2011. Prior to that time, there was no public market for our common stock. As a result, we have not set forth quarterly information with respect to the high and low prices for our common stock for the two most recent fiscal years.
Holders of our Common stock
As of February 28, 2011, there were 36 stockholders of record of our common stock. The number of record holders is based upon the actual number of holders registered on the books of the company at such date and does not include holders of shares in “street names” or persons, partnerships, associations, corporations or other entities identified in security position listings maintained by depositories.
Dividends
We have never declared or paid any cash dividends on our common stock. We currently intend to retain all available funds and any future earnings to support our operations and finance the growth and development of our business and do not anticipate declaring or paying any cash dividends on our common stock for the foreseeable future. Our ability to pay dividends on our common stock is limited by the covenants of our loan and security agreement governing the Hercules Credit Facility and may be further restricted by the terms of any of our future indebtedness.
Purchases and Sales of Unregistered Securities
We did not purchase any of our registered equity securities during the period covered by this Annual Report on Form 10-K. The following sets forth information regarding all unregistered securities sold during the last three fiscal years. Within the last three years, we issued and sold the following securities:
Issuances of Promissory Notes
In January 2009, we issued convertible promissory notes to the Foundation for Research, HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures. The aggregate principal amount of the notes issued was $10,625,000 and the notes had an annual interest rate of 5%. The convertible promissory notes converted into shares of our common stock upon the completion of our initial public offering.
In August, September and October 2009, we issued secured promissory notes to the Foundation for Research, HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures. The aggregate principal amount of the notes issued was $10,625,000 and the notes had an annual interest rate of 12%. The secured promissory notes converted into shares of our common stock upon the completion of our initial public offering.
In March, June and September 2010, we issued secured promissory notes to HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures. The aggregate principal amount of the notes issued was $15,000,000 and the notes had an annual interest rate of 5%. The secured promissory notes converted into shares of our common stock upon the completion of our initial public offering.
56
Table of Contents
In April, June and September 2010, we issued subordinated secured promissory notes to HBM BioVentures (Cayman) Ltd. The aggregate principal amount of the notes issued was $3,750,000 and the notes had annual interest rates between 9.05% and 9.24%. The subordinated secured promissory notes converted into shares of our common stock upon the completion of our initial public offering.
In April 2010, we issued a secured promissory note to General Electric Capital Corporation, or GECC. The principal amount of the note issued was $11,250,000 and the note had an annual interest rate of 9.24%. In November 2010, all borrowings under this note were repaid in full from proceeds of a credit facility with Hercules Technology Growth Capital, Inc.
In November 2010, we issued a secured promissory note to Hercules Technology Growth Capital, Inc. and Hercules Technology III, L.P. The principal amount of the note issued is $26,250,000 and the note has a variable interest rate.
In December 2010, we entered into an agreement for the issuance of convertible promissory notes to HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures. On December 29, 2010, we issued notes for an aggregate principal amount of $7,500,000. The notes have an annual interest rate of 5%. The convertible promissory notes converted into shares of our common stock upon the completion of our initial public offering.
No underwriters were involved in the issuances of promissory notes described above. The promissory notes described above were issued to investors in reliance upon the exemption from the registration requirements of the Securities Act, as set forth in Section 4(2) under the Securities Act and, in certain cases, Regulation D promulgated thereunder, relative to transactions by an issuer not involving any public offering, to the extent an exemption from such registration was required.
Issuances of Common Stock and Preferred Stock
In March 2007, in connection with the Acquisition, we issued a total of 464,900 shares of common stock at a price per share of $0.11 to HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures, for an aggregate purchase price of $50,000.
In March 2007, February 2008, July 2008 and October 2008, we issued a total of 6,322,640 shares of Series A convertible preferred stock at a price per share of $13.44 to HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures, for an aggregate purchase price of $85.0 million.
Stock Option Grants
Since inception, we have issued options to certain directors, employees and consultants to purchase an aggregate of 3,043,198 shares of common stock as of December 31, 2010. As of December 31, 2010, options to purchase 110,196 shares of common stock had been exercised and options to purchase 2,073,700 shares of common stock remained outstanding at a weighted average exercise price of $2.69 per share.
Issuances of Warrants
In January 2009, we issued to HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures warrants to purchase 158,061 shares of common stock in connection with the issuance of convertible promissory notes also in January 2009. The common stock warrants have an exercise price of $2.69 per share.
In July 2009, we issued warrants for an aggregate of 23,244 shares of Series A convertible preferred stock to our landlord in connection with rent deferral. The warrants had an exercise price of $13.44 per share.
57
Table of Contents
In November 2010, we issued to Hercules Technology Growth Capital, Inc. and Hercules Technology III, L.P. a warrant to purchase 178,986 shares of Series A preferred stock in connection with the Hercules Credit Facility. The preferred stock warrant has an exercise price of $13.44 per share, which expires upon the earlier to occur of (i) November 24, 2020 or (ii) five years following the initial public offering.
In December 2010, we issued to HBM BioVentures (Cayman) Ltd., entities affiliated with MPM Capital, entities affiliated with Orbimed Advisors and entities affiliated with Sanderling Ventures warrants to purchase an aggregate of 167,361 shares of common stock in connection with the issuance of certain convertible promissory notes. The common stock warrants have an exercise price of $13.44 per share.
The sales and issuances of restricted securities in the transactions described above were deemed to be exempt from registration under the Securities Act in reliance upon Rule 701 promulgated under Section 3(b) of the Securities Act, as transactions pursuant to a written compensation benefit plan and contracts relating to compensation as provided under Rule 701 and Section 4(2) of the Securities Act, or Rule 506 of Regulation D promulgated thereunder, as transactions by an issuer not involving any public offering. The recipients of securities in the transaction represented their intentions to acquire the securities for investment only and not with a view to or for sale in connection with any distribution thereof and appropriate legends were affixed to the securities issued in such transactions. The sales of these securities were made without general solicitation or advertising. All recipients were accredited investors or had adequate access, through their relationship with us, to information about us. There were no underwritten offerings employed in connection with any of the transactions set forth above. Each share of our convertible preferred stock described above converted into one share of our common stock upon the completion of our initial public offering.
In February 2011, we completed the initial public offering of our common stock pursuant to a registration statement on Form S-1, as amended (File No. 333-170245) that was declared effective on February 2, 2011. Under the registration statement, we registered the offering and sale of an aggregate of 6,900,000 shares of our common stock. An aggregate of 6,000,000 shares of common stock registered under the registration statement were sold at a price to the public of $7.00 per share. Barclays Capital Inc. and Piper Jaffray and Co. acted as joint book running managers of the offering and as representatives of the underwriters. The offering commenced on February 3, 2011 and closed on February 8, 2011. The over-allotment option was not exercised by the underwriters. As a result of our IPO, we raised a total of $42.0 million in gross proceeds, and approximately $37.0 million in net proceeds after deducting approximately $5.0 million in underwriting discounts and commissions and estimated offering expenses. There has been no material change in our planned use of proceeds from the initial public offering from that described in the final prospectus filed with the SEC on February 3, 2011.
Securities Authorized for Issuance Under Equity Compensation Plans
The following table contains information about our equity compensation plans as of December 31, 2010.
Equity Compensation Plan Information
| Plan category |
Number of securities to be issued upon exercise of outstanding options, warrants and rights |
Weighted-average exercise price of outstanding options, warrants and rights |
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) |
|||||||||
| (a) | (b) | (c) | ||||||||||
| Equity compensation plans approved by security holders |
2,073,700 | $ | 2.69 | 363,814 | ||||||||
| Equity compensation plans not approved by security holders |
— | — | — | |||||||||
| Total |
2,073,700 | $ | 2.69 | 363,814 | (1) | |||||||
| (1) | Our 2011 stock incentive plan, or the 2011 Plan, contains an “evergreen” provision, which allows for an increase in the number of shares available for issuance under the 2011 Plan on the first day of each calendar year from 2012 through 2015. The annual increase in the number of shares shall be equal to the lesser of (i) 557,880 shares of our common stock; (ii) a number of shares equal to 3% of our outstanding shares as of such date; or (iii) an amount determined by our board of directors. |
58
Table of Contents
| Item 6. | Selected Consolidated Financial Data |
The following selected consolidated financial data should be read together with our consolidated financial statements and accompanying notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included elsewhere in this Annual Report on Form 10-K. The selected consolidated financial data in this section is not intended to replace our consolidated financial statements and the accompanying notes. The selected consolidated financial data as of December 31, 2010 and 2009, and for the years ended December 31, 2010, 2009 and 2008, and the consolidated balance sheet data as of December 31, 2010 and 2009, have been derived from our consolidated financial statements included elsewhere in this Annual Report on Form 10-K, which have been audited by J.H. Cohn LLP, an independent registered public accounting firm. Our historical results are not necessary indicative of the results to be expected in any future period.
| • | The selected consolidated financial data as of and for the years ended December 31, 2008 and 2007 have been derived from our audited consolidated financial statements not contained herein. |
| • | The selected consolidated financial data as of December 31, 2006, and for the year ended December 31, 2006, and for the period from January 1, 2007 through March 23, 2007, have been derived from unaudited consolidated financial statements of the Predecessor, SkyePharma, Inc., not included in this Annual Report on Form 10-K. |
The term Predecessor refers to SkyePharma, Inc. prior to March 24, 2007, or the Acquisition Date, and the term Successor refers to Pacira Pharmaceuticals, Inc. and its consolidated subsidiaries. Our results of operations for the year ended December 31, 2007, while representing a full year for Pacira Pharmaceuticals, Inc., do not reflect the operations of PPI-California until March 24, 2007, after the Acquisition Date. We have presented the Predecessor for the period from January 1, 2007 through March 23, 2007, as we believe it best presents the continuity of operations of the Successor prior to the Acquisition.
59
Table of Contents
| Predecessor | Successor | |||||||||||||||||||||||
| Year Ended
December 31 2006 |
January 1, 2007 to March 23, 2007 |
Year Ended December 31 |
||||||||||||||||||||||
| 2007 | 2008 | 2009 | 2010 | |||||||||||||||||||||
| (unaudited) | (audited) | |||||||||||||||||||||||
| (in thousands, except share and per share data) | ||||||||||||||||||||||||
| Consolidated Statement of Operations Data: |
||||||||||||||||||||||||
| Revenues: |
||||||||||||||||||||||||
| Supply revenue |
$ | 5,800 | $ | 684 | $ | 5,444 | $ | 6,852 | $ | 6,324 | $ | 7,640 | ||||||||||||
| Royalties |
2,784 | 500 | 2,388 | 3,648 | 4,044 | 3,705 | ||||||||||||||||||
| Collaborative licensing and development revenue |
3,088 | 204 | 509 | 3,425 | 4,638 | 3,217 | ||||||||||||||||||
| Revenue from SkyePharma PLC |
702 | 39 | — | — | — | — | ||||||||||||||||||
| Total revenues |
12,374 | 1,427 | 8,341 | 13,925 | 15,006 | 14,562 | ||||||||||||||||||
| Operating expenses: |
||||||||||||||||||||||||
| Cost of revenues |
15,782 | 2,825 | 9,492 | 17,463 | 12,301 | 12,276 | ||||||||||||||||||
| Research and development |
16,060 | 3,251 | 20,665 | 33,214 | 26,233 | 18,628 | ||||||||||||||||||
| Selling, general and administrative |
8,685 | 2,632 | 4,170 | 8,611 | 5,020 | 6,030 | ||||||||||||||||||
| Acquired in-process research and development |
— | — | 12,400 | — | — | — | ||||||||||||||||||
| Total operating expenses |
40,527 | 8,708 | 46,727 | 59,288 | 43,554 | 36,934 | ||||||||||||||||||
| Loss from operations |
(28,153 | ) | (7,281 | ) | (38,386 | ) | (45,363 | ) | (28,548 | ) | (22,372 | ) | ||||||||||||
| Other income (expense) |
(2,713 | ) | (13 | ) | 16 | (224 | ) | 367 | 150 | |||||||||||||||
| Loss on early extinguishment of debt |
— | — | — | — | — | (184 | ) | |||||||||||||||||
| Interest: |
||||||||||||||||||||||||
| Interest income |
60 | 4 | 491 | 235 | 77 | 146 | ||||||||||||||||||
| Interest expense |
(11,221 | ) | (2,265 | ) | — | — | (1,723 | ) | (3,959 | ) | ||||||||||||||
| Royalty interest obligation |
4,694 | (1,486 | ) | 1,686 | 3,490 | (1,880 | ) | (930 | ) | |||||||||||||||
| Net loss |
$ | (37,333 | ) | $ | (11,041 | ) | $ | (36,193 | ) | $ | (41,862 | ) | $ | (31,707 | ) | $ | (27,149 | ) | ||||||
| Net loss per share applicable to common stockholders—basic and diluted |
$ | (77.85 | ) | $ | (79.23 | ) | $ | (55.32 | ) | $ | (47.29 | ) | ||||||||||||
| Weighted average number of common shares used in net loss per share calculation |
434,900 | 528,357 | 573,118 | 574,072 | ||||||||||||||||||||
| Pro forma net loss per share—basic and diluted (unaudited)(1) |
$ | (3.60 | ) | $ | (2.20 | ) | ||||||||||||||||||
| Shares used in computing pro forma loss per share—basic and diluted (unaudited) |
8,545,094 | 11,232,917 | ||||||||||||||||||||||
| (1) | Pro forma basic and diluted net loss per share is calculated assuming the conversion of all of our outstanding shares of Series A convertible preferred stock and our secured and unsecured notes (including the notes issued upon the first closing of the December 2010 Convertible Notes) and accrued interest thereon into common stock at the beginning of the period or at the original date of issuance, if later. The net losses for the years ended December 31, 2009 and 2010 were adjusted to reflect the elimination of interest expense associated with the assumed conversion at the beginning of each period of the convertible and secured notes in the amounts of $0.9 million and $2.5 million, respectively. |
60
Table of Contents
| Predecessor | Successor | |||||||||||||||||||||||||||
| December
31, 2006 |
December 31, | December 31, 2010 | ||||||||||||||||||||||||||
| 2007 | 2008 | 2009 | Actual | Pro forma(1) |
Pro
forma as adjusted(2) |
|||||||||||||||||||||||
| (unaudited) | (unaudited) | (audited) | ||||||||||||||||||||||||||
| (in thousands) | (in thousands) | |||||||||||||||||||||||||||
| Consolidated Balance Sheet Data: |
||||||||||||||||||||||||||||
| Cash and cash equivalents |
$ | 627 | $ | 7,240 | $ | 12,386 | $ | 7,077 | $ | 26,133 | $ | 26,133 | $ | 64,568 | ||||||||||||||
| Working capital (deficit) |
27,010 | 2,354 | 2,341 | (1,868 | ) | 14,817 | 14,817 | 53,252 | ||||||||||||||||||||
| Total assets |
63,188 | 39,157 | 50,541 | 43,954 | 66,562 | 66,562 | 103,590 | |||||||||||||||||||||
| Long-term debt |
21,648 | 8,241 | 3,618 | 25,820 | 74,660 | 24,865 | 24,865 | |||||||||||||||||||||
| Convertible preferred stock, par value |
— | 3 | 6 | 6 | 6 | |||||||||||||||||||||||
| Common stock, par value |
— | 1 | 1 | 1 | 1 | 11 | 17 | |||||||||||||||||||||
| Accumulated deficit |
(319,756 | ) | (36,193 | ) | (78,055 | ) | (109,762 | ) | (136,911 | ) | (136,911 | ) | (136,911 | ) | ||||||||||||||
| Total stockholders’ equity (deficit) |
$ | (221,541 | ) | $ | 8,937 | $ | 7,490 | $ | (22,949 | ) | $ | (48,383 | ) | $ | 1,412 | $ | 38,440 | |||||||||||
| (1) | Pro forma includes the impact of the conversion of all of our outstanding shares of Series A convertible preferred stock and our secured and unsecured notes (including the notes issued upon the first closing of the December 2010 Convertible Notes) and accrued interest thereon into common stock. |
| (2) | Pro forma as adjusted includes the proceeds from initial public offering, completed in February 2011, net of underwriters’ discounts and commissions and estimated offering expenses. |
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
The following discussion of our financial condition and results of operations should be read in conjunction with our financial statements and the notes to those financial statements appearing elsewhere in this Annual Report on Form 10-K. This discussion contains forward-looking statements that involve significant risks and uncertainties. As a result of many factors, such as those set forth under “Risk Factors” in Item 1A of this Annual Report on Form 10-K, our actual results may differ materially from those anticipated in these forward-looking statements.
Overview
We are an emerging specialty pharmaceutical company focused on the development, commercialization and manufacture of proprietary pharmaceutical products, based on our proprietary DepoFoam drug delivery technology, for use in hospitals and ambulatory surgery centers. In September 2010, we filed an NDA for EXPAREL with the United States Food and Drug Administration, or FDA, which was accepted by the FDA for review on December 10, 2010, using a 505(b)(2) application. Our clinical data demonstrates that EXPAREL provides analgesia for up to 72 hours post-surgery, compared with seven hours or less for bupivacaine. We are initially seeking approval for postsurgical analgesia by local administration into the surgical wound, or infiltration, a procedure commonly employed using bupivacaine. Under the Prescription Drug User Fee Act, or PDUFA, guidelines, the FDA has a goal of ten months from the date of NDA filing to make a decision regarding the approval of our filing. The PDUFA goal date for our NDA is July 28, 2011. We are also pursuing several additional indications for EXPAREL and expect to submit a supplemental NDA, or sNDA, for nerve block and epidural administration. We currently intend to develop and commercialize EXPAREL and our other product candidates in the United States while out-licensing commercialization rights for other territories.
We were incorporated in Delaware under the name Blue Acquisition Corp. in December 2006 and changed our name to Pacira, Inc. in June 2007. In October 2010, we changed our name to Pacira Pharmaceuticals, Inc. Pacira Pharmaceuticals, Inc. is the holding company for our California operating subsidiary of the same name, which we refer to as PPI-California. On March 24, 2007, or the Acquisition Date, MPM Capital, Sanderling Ventures, OrbiMed Advisors, HBM BioVentures, the Foundation for Research and their co-investors, through Pacira Pharmaceuticals, Inc., acquired PPI-California, from SkyePharma Holding, Inc., which we refer to as the Acquisition. PPI-California was known as SkyePharma, Inc. prior to the Acquisition.
61
Table of Contents
Our two marketed products, DepoCyt(e) and DepoDur, and our proprietary DepoFoam extended release drug delivery technology were acquired as part of the Acquisition. DepoCyt(e) is a sustained release liposomal formulation of the chemotherapeutic agent cytarabine and is indicated for the intrathecal treatment of lymphomatous meningitis. DepoCyt(e) was granted accelerated approval by the FDA in 1999 and full approval in 2007. DepoDur is an extended release injectable formulation of morphine indicated for epidural administration for the treatment of pain following major surgery. DepoDur was approved by the FDA in 2004.
Since inception, we have incurred significant operating losses. Our net loss was $27.1 million for the year ended December 31, 2010, including research and development expenses of $18.6 million. Our net loss was $31.7 million for the year ended December 31, 2009, including research and development expenses of $26.2 million. We do not expect our currently marketed products to generate revenue that is sufficient for us to achieve profitability because we expect to continue to incur significant expenses as we advance the development of EXPAREL and our other product candidates, seek FDA approval for our product candidates that successfully complete clinical trials and develop our sales force and marketing capabilities to prepare for their commercial launch. We also expect to incur additional expenses to add operational, financial and management information systems and personnel, including personnel to support our product development efforts and our obligations as a public reporting company. For us to become and remain profitable, we believe that we must succeed in commercializing EXPAREL or other product candidates with significant market potential.
Financial Operations Overview
Revenues
Our revenue derived from DepoCyt(e) and DepoDur, our products manufactured by us and sold by our commercial partners, is comprised of two components: supply revenue and royalties. Supply revenue is derived from a contractual supply price paid to us by our commercial partners. Royalties are recognized as the product is sold by our commercial partners and is typically calculated as a percentage of the net selling price, which is net of discounts, returns, and allowances incurred by our commercial partners. Accordingly, the primary factors that determine our revenues derived from DepoCyt(e) and DepoDur are:
| • | the level of orders submitted by our commercial partners; |
| • | the level of prescription and institutional demand for our products; |
| • | unit sales prices; |
| • | the amount of gross-to-net sales adjustments realized by our commercial partners; and |
| • | exchange rates on European sales, denominated in euros, that are repatriated in dollars. |
We also generate collaborative licensing and development revenue from our collaborations with third parties who seek to use our DepoFoam technology to develop extended release formulations of their products and product candidates.
The following table sets forth a summary of our supply revenue, royalties and collaborative licensing and development revenue for the years ended December 31, 2008, 2009 and 2010:
62
Table of Contents
| Year Ended December 31, | ||||||||||||
| 2008 | 2009 | 2010 | ||||||||||
| (in thousands) | ||||||||||||
| DepoCyt(e)(1) |
||||||||||||
| Supply revenue |
$ | 5,912 | $ | 5,882 | $ | 6,843 | ||||||
| Royalties |
3,195 | 3,708 | 3,411 | |||||||||
| 9,107 | 9,590 | 10,254 | ||||||||||
| DepoDur(1) |
||||||||||||
| Supply revenue |
940 | 442 | 797 | |||||||||
| Royalties |
453 | 336 | 294 | |||||||||
| 1,393 | 778 | 1,091 | ||||||||||
| Total DepoCyt(e) and DepoDur revenue(1) |
10,500 | 10,368 | 11,345 | |||||||||
| Collaborative licensing and development revenue |
3,425 | 4,638 | 3,217 | |||||||||
| Total revenue |
$ | 13,925 | $ | 15,006 | $ | 14,562 | ||||||
| (1) | Total DepoCyt(e) and DepoDur revenue does not include collaborative licensing and development revenue related to DepoCyt(e) and DepoDur. |
Cost of Revenues
Cost of revenues consists of the costs associated with producing our products for our commercial partners and providing research and development services to our collaboration partners. In particular, our cost of revenues includes:
| • | manufacturing overhead and fixed costs associated with running two cGMP manufacturing facilities, including salaries and related costs of personnel involved with our manufacturing activities; |
| • | allocated overhead, personnel conducting research and development, as well as research and development performed by outside contractors or consultants for our collaborative licensing and development activities; |
| • | royalties due to third parties on our revenues; |
| • | packaging, testing, freight and shipping; |
| • | the cost of active pharmaceutical ingredients; and |
| • | overhead costs associated with excess manufacturing capacity are charged to cost of revenue as incurred. Manufacturing, labor and overhead costs are capitalized only to the extent of actual capacity utilized. The cost of excess capacity was $10.1 million, $5.5 million and $6.0 million for the years ended December 31, 2008, 2009 and 2010, respectively. Gross margins from supply revenue were -110%, -55% and -49% for the years ended December 31, 2008, 2009 and 2010, respectively. Our negative margin is primarily due to excess capacity. Excluding the cost of excess capacity, as described above, gross margin from supply revenue was 36%, 31%, and 30% for the years ended December 31, 2008, 2009 and 2010, respectively. |
Research and Development Expenses
Our research and development expenses consist of expenses incurred in developing, testing, manufacturing and seeking regulatory approval of our product candidates, including:
63
Table of Contents
| • | expenses associated with regulatory submissions, clinical trials and manufacturing, including additional expenses to prepare for the commercial manufacture of EXPAREL, such as the hiring and training of additional personnel; |
| • | payments to third-party contract research organizations, contract laboratories and independent contractors; |
| • | payments made to consultants who perform research and development on our behalf and assist us in the preparation of regulatory filings; |
| • | payments made to third-party investigators who perform research and development on our behalf and clinical sites where such research and development is conducted; |
| • | personnel related expenses, such as salaries, benefits, travel and other related expenses, including stock-based compensation; |
| • | expenses incurred to maintain technology licenses; and |
| • | facility, maintenance, and allocated rent, utilities, and depreciation and amortization, and other related expenses. |
Clinical trial expenses for our product candidates are a significant component of our current research and development expenses. Product candidates in later stage clinical development, such as EXPAREL, generally have higher research and development expenses than those in earlier stages of development, primarily due to the increased size and duration of the clinical trials. We coordinate clinical trials through a number of contracted investigational sites and recognize the associated expense based on a number of factors, including actual and estimated subject enrollment and visits, direct pass-through costs and other clinical site fees.
From the Acquisition Date through December 31, 2010, we incurred research and development expenses of $98.7 million, of which $95.1 million is related to the development of EXPAREL. We incurred research and development expenses associated with the development of EXPAREL of $18.4 million for the year ended December 31, 2010, $25.2 million for the year ended December 31, 2009 and $31.9 million for the year ended December 31, 2008.
We expect to incur additional research and development expenses as we accelerate the development of EXPAREL in additional indications. These expenditures are subject to numerous uncertainties regarding timing and cost to completion. Completion of clinical trials may take several years or more and the length of time generally varies according to the type, complexity, novelty and intended use of a product candidate. We are currently unable to determine our future research and development expenses related to EXPAREL because the timing and outcome of the FDA’s review of the NDA for EXPAREL is not currently known and the requirements of any additional clinical trials of EXPAREL for additional indications has yet to be determined. The cost of clinical development may vary significantly due to factors such as the scope, rate of progress, expense and outcome of our clinical trials and other development activities.
Selling, General and Administrative Expenses
Selling, general and administrative expenses consist primarily of salaries, benefits and other related costs, including stock-based compensation, for personnel serving in our executive, finance, accounting, legal, human resource, and sales and marketing functions. Our selling, general and administrative expenses also include facility and related costs not included in research and development expenses and cost of revenues, professional fees for legal, consulting, tax and accounting services, insurance, depreciation and general corporate expenses. We expect that our selling, general and administrative expenses will increase with the continued development and potential commercialization of our product candidates and increased expenses associated with us becoming a public company. Additionally, we plan to build a commercial infrastructure for the anticipated launch of EXPAREL and we currently plan to hire most of our sales force only if EXPAREL is approved by the FDA.
64
Table of Contents
Interest Income (Expense)
Interest income (expense) consists of interest income, interest expense, and royalty interest obligation. Interest income consists of interest earned on our cash and cash equivalents, and amortization of discount on a note receivable from one of our commercial partners. Interest expense consists primarily of cash and non-cash interest costs related to our credit facility, our secured and unsecured notes issued to certain of our investors that converted into common stock upon completion of our initial public offering, and negotiated rent deferral payments. Royalty interest obligation consists of our royalty payments made in connection with the amended and restated royalty interests assignment agreement, or the Amended and Restated Royalty Interests Assignment Agreement, with Royalty Securitization Trust I, an affiliate of Paul Capital Advisors, LLC, or Paul Capital.
We record our royalty interest obligation as a liability in our consolidated balance sheets in accordance with ASC 470-10-25, Sales of Future Revenues. We impute interest expense associated with this liability using the effective interest rate method. The effective interest rate may vary during the term of the agreement depending on a number of factors including the actual sales of DepoCyt(e) and DepoDur and a significant estimation, performed quarterly, of certain of our future cash flows related to these products during the remaining term of the Amended and Restated Royalty Interests Assignment Agreement which terminates on December 31, 2014. The effect of the change in the estimates is reflected in our consolidated statements of operations as interest income (expense). In addition, such cash flows are subject to foreign exchange movements related to sales of DepoCyt(e) and DepoDur denominated in currencies other than U.S. dollars.
Critical Accounting Policies and Use of Estimates
We have based our management’s discussion and analysis of our financial condition and results of operations on our financial statements that have been prepared in accordance with generally accepted accounting principles, or GAAP, in the United States. The preparation of these financial statements requires us to make estimates that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements as well as the reported revenues and expenses during the reporting periods. On an ongoing basis, we evaluate our estimates and judgments, including those related to clinical trial expenses and share-based compensation. We base our estimates on historical experience and on various other factors we believe to be appropriate under the circumstances. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully discussed in Note 2 to our audited consolidated financial statements included in this filing, we believe that the following accounting policies are critical to the process of making significant judgments and estimates in the preparation of our consolidated financial statements. We have reviewed these critical accounting policies and estimates with the audit committee of our board of directors.
Revenue Recognition
We recognize revenue in accordance with SEC Staff Accounting Bulletin, or SAB, No. 104, Revenue Recognition, and Statement of Financial Accounting Standards, or ASC 605, Revenue Recognition.
Supply revenue. We recognize supply revenue from products manufactured and supplied to our commercial partners, when the following four basic revenue recognition criteria under the related accounting guidance are met: (1) persuasive evidence of an arrangement exists; (2) delivery has occurred or services have been rendered; (3) the fee is fixed or determinable; and (4) collectability is reasonably assured. Prior to the shipment of our manufactured products, we conduct initial product release and stability testing in accordance with cGMP. Our commercial partners can return the products within contracted specified timeframes if the products do not meet the applicable inspection tests. We estimate our return reserves based on our experience with historical return rates. Historically, our product returns have not been material.
Royalties. We recognize revenue from royalties based on our commercial partners’ net sales of products. Royalties are recognized as earned in accordance with contract terms when they can be reasonably estimated and
65
Table of Contents
collectability is reasonably assured. Our commercial partners are obligated to report their net product sales and the resulting royalty due to us within 60 days from the end of each quarter. Based on historical product sales, royalty receipts and other relevant information, we accrue royalty revenue each quarter and subsequently true-up when we receive royalty reports from our commercial partners.
Collaborative licensing and development revenue. We recognize revenue from reimbursement received in connection with feasibility studies and development work for third parties who desire to utilize our DepoFoam extended release drug delivery technology for their products, when our contractual services are performed, provided collectability is reasonably assured. Our principal costs under these agreements include our personnel conducting research and development, and our allocated overhead, as well as research and development performed by outside contractors or consultants.
We recognize revenues from non-refundable up-front license fees received under collaboration agreements ratably over the performance period as determined under the collaboration agreement (estimated development period in the case of development agreements, and contract period or longest patent life in the case of supply and distribution agreements). If the estimated performance period is subsequently modified, we will modify the period over which the up-front license fee is recognized accordingly on a prospective basis. Upon termination of a collaboration agreement, any remaining non-refundable license fees received by us, which had been deferred, are generally recognized in full. All such recognized revenues are included in collaborative licensing and development revenue in our consolidated statements of operations.
We recognize revenue from milestone payments received under collaboration agreements when earned, provided that the milestone event is substantive, its achievability was not reasonably assured at the inception of the agreement, we have no further performance obligations relating to the event, and collectability is reasonably assured. If these criteria are not met, we recognize milestone payments ratably over the remaining period of our performance obligations under the collaboration agreement.
Research and Development Expenses
We expense all research and development costs as incurred. We rely on third parties to conduct our preclinical and clinical studies and to provide services, including data management, statistical analysis and electronic compilation for our clinical trials. We track and record information regarding third-party research and development expenses for each study or trial that we conduct and recognize these expenses based on the estimated progress towards completion at the end of each reporting period. Factors we consider in preparing these estimates include the number of subjects enrolled in studies, milestones achieved and other criteria related to the efforts of our vendors. Historically, any adjustments we have made to these assumptions have not been material. Depending on the timing of payments to vendors and estimated services provided, we may record net prepaid or accrued expenses related to these costs.
We expense the manufacturing costs (labor and overhead) of our clinical supplies as incurred. Through December 31, 2010, these expenses have not been material. Unused raw material for manufacturing clinical supplies is included in inventory and expensed when used.
Share-Based Compensation
We have adopted the fair value recognition provisions of Financial Accounting Standards Board Accounting Standards Codification, or ASC, 718 “Accounting for Stock Based Compensation” (formerly Statement of Financial Accounting Standards No. 123(R), Share-Based Payments), which we refer to as ASC 718, using the modified prospective transition method. The modified prospective transition method applies the provisions of ASC 718 to new awards and to awards modified, repurchased or cancelled after the adoption date. Additionally, compensation cost for the portion of the awards for which the requisite service has not been rendered that are outstanding as of the adoption date is recognized in the Statement of Operations over the remaining service period after the adoption date based on the award’s original estimate of fair value. All stock-based awards granted to non-employees are accounted for at their fair value in accordance with ASC 718, and ASC 505, “Accounting for Equity Instruments that are Issued to Other Than Employees for Acquiring, or in Conjunction with Selling, Goods or Services,” under which compensation expense is generally recognized over the vesting period of the award. Determining the amount of stock-based compensation to be recorded requires us to develop estimates of fair values of stock options as of the grant date.
66
Table of Contents
For the years ended December 31, 2008, 2009 and 2010, we recognized employee stock-based compensation expense of $242,000, $524,000 and $23,000, respectively. The intrinsic value of all outstanding vested and non-vested stock-based compensation arrangements, based on our initial public offering price of $7.00 per share, was $8.9 million, based on 2,073,700 shares of our common stock issuable upon exercise of stock-based compensation arrangements outstanding at December 31, 2010 at a weighted average exercise price of $2.69 per share.
We account for stock-based compensation by measuring and recognizing compensation expense for all stock-based payments made to employees and directors based on estimated grant date fair values. We use the straight-line method to allocate compensation cost to reporting periods over each optionee’s requisite service period, which is generally the vesting period. We estimate the fair value of our stock-based awards to employees and directors using the Black-Scholes option valuation model, or Black-Scholes model. The Black-Scholes model requires the input of subjective assumptions, including the expected stock price volatility, the calculation of expected term and the fair value of the underlying common stock on the date of grant, among other inputs.
Results of Operations
Comparison of Years Ended December 31, 2010 and 2009
| Year Ended December 31, | Increase/ (Decrease) |
%
Increase/ (Decrease) | ||||||
| 2009 | 2010 | |||||||
| (dollars in thousands) | ||||||||
| Revenues |
$15,006 | $14,562 | ($444) | -3% | ||||
| Cost of revenues |
12,301 | 12,276 | (25) | 0% | ||||
| Research and development |
26,233 | 18,628 | (7,605) | -29% | ||||
| Selling, general and administrative |
5,020 | 6,030 | 1,010 | 20% | ||||
| Loss on extinguishment of debt |
— | 184 | 184 | N.M. | ||||
| Other income |
367 | 150 | (217) | -59% | ||||
| Interest expense |
($3,526) | ($4,743) | ($1,217) | 35% | ||||
Revenues. Revenues decreased by $0.4 million, or 3%, to $14.6 million in the year ended December 31, 2010 as compared to $15.0 million in the year ended December 31, 2009. The decrease was primarily due to a decrease in collaborative licensing and development revenue of $1.4 million and a decrease in royalties revenue of $0.3 million, offset by an increase in supply revenue of $1.3 million. The decrease in collaborative licensing and development revenue reflected a reduction in contract development activities for Amylin, for the year ended December 31, 2010, as well as a one-time purchase of equipment for which we were reimbursed by Amylin in the year ended December 31, 2009. The decrease in royalties in 2010 reflected a decrease in end user sales of DepoCyt(e) and DepoDur and foreign exchange rate impact on sales in Europe. The increase in supply revenue in 2010 was primarily due to higher sales of DepoCyt(e) to our European partner, driven by fulfillment of an order backlog at the end of 2009.
Cost of Revenues. Cost of revenues of $12.3 million, remained unchanged from the prior year level. Cost of collaborative licensing and development revenue decreased, as our personnel were re-assigned subsequent to the reduction in contract development activities for Amylin. The reduction was offset by an increase in cost of supply revenue due to higher volume of supply sales and higher cost of maintenance activities.
67
Table of Contents
Research and Development Expenses. Research and development expenses decreased by $7.6 million, or 29%, to $18.6 million in the year ended December 31, 2010 from $26.2 million in the year ended December 31, 2009. This decrease resulted primarily from a $6.7 million decrease in third party clinical trials costs, to $2.0 million in 2010 from $8.7 million in 2009, when we completed our pivotal Phase 3 placebo controlled studies.
In the years ended December 31, 2010 and 2009, research and development expenses attributable to EXPAREL were $18.4 million, or 99%, and $25.2 million, or 96% of total research and development expenses, respectively. The remaining research and development expenses related to our other product candidate initiatives.
Selling, General and Administrative Expenses. Selling, general and administrative expenses increased by $1.0 million, or 20%, to $6.0 million in the year ended December 31, 2010 as compared to $5.0 million in the year ended December 31, 2009. Selling expenses increased by $0.1 million, or 15%, to $0.9 million in the year ended December 31, 2010 as compared to $0.8 million in the year ended December 31, 2009, due to hiring of our commercial personnel in November 2010. General and administrative expenses increased by $0.9 million, or 20%, to $5.1 million in the year ended December 31, 2010 as compared to $4.2 million in the year ended December 31, 2009. The increase was primarily due to higher costs associated with completing the previous three years of audits and tax filings.
Loss on extinguishment of debt. The Company recorded $0.2 million loss on extinguishment of a $11.25 million credit facility, established with GE Capital Corporation in April 2010. Although the facility was established originally for a period of 3 years, the Company elected to repay the debt in full in November 2010, from proceeds of a new term loan, established with Hercules Technology Growth Capital, Inc in November 2010. The amount represents the final payment fees and the balance of deferred financing cost which were written off when the debt was paid off.
Other Income (Expense). Other income decreased by $0.2 million to $0.2 million in the year ended December 31, 2010 as compared to $0.4 million in the year ended December 31, 2009. The Company had entered into trade settlement agreements with its trade creditors in 2009. The decrease was primarily due to a lower amount of gain realized on settlements with trade creditors in 2010 compared to 2009, as a result of lower proportionate settlement payments.
Interest Income (Expense). Interest expense increased by $1.2 million in the year ended December 31, 2010, or 35%, to $4.7 million, as compared to interest expense of $3.5 million in the year ended December 31, 2009. The increase was primarily due to a $2.2 million increase in interest expenses resulting from our debt financing activities, offset by $1.0 million decrease in interest expense, attributable to the royalty interest obligation under the Amended and Restated Royalty Interests Assignment Agreement. The interest expense relating to the obligations under the Amended and Restated Royalty Interests Assignment Agreement is composed of (1) the difference in the revaluation of our obligations between each reporting period and (2) the actual royalty interest payments payable pursuant to the Amended and Restated Royalty Interests Assignment Agreement for such reporting period. In determining the amount of the royalty interest obligation, we employ estimates of future cash flows derived from our royalties payable to Paul Capital based on end user sales of DepoCyt(e) and DepoDur, discounted at a rate that reflects an estimate of the cost of capital under the Amended and Restated Royalty Interests Assignment Agreement. At December 31, 2010, our estimate of future end user sales of DepoCyt(e) sales in the US was lower than the estimate as of December 31, 2009. This lower estimate resulted in a decrease of the royalty interest obligation valuation at December 31, 2010 and, as a result, $0.6 million of the royalty interest obligation was recorded as interest income in the year ended December 31, 2010. In comparison, the valuation of the royalty interest obligation at December 31, 2009 was slightly higher than the valuation at December 31, 2008, which resulted in a $0.2 million interest expense in the year ended December 31, 2009.
68
Table of Contents
Comparison of Years Ended December 31, 2009 and 2008
| Year Ended December 31, | Increase/ (Decrease) |
%
Increase/ (Decrease) |
||||||||||||||
| 2008 | 2009 | |||||||||||||||
| (dollars in thousands) | ||||||||||||||||
| Revenues |
$ | 13,925 | $ | 15,006 | $ | 1,081 | 8% | |||||||||
| Cost of revenues |
17,463 | 12,301 | (5,162 | ) | (30)% | |||||||||||
| Research and development |
33,214 | 26,233 | (6,981 | ) | (21)% | |||||||||||
| Selling, general and administrative |
8,611 | 5,020 | (3,591 | ) | (42)% | |||||||||||
| Other income (expense) |
(224 | ) | 367 | 591 | N.M | . | ||||||||||
| Interest income (expense) |
$ | 3,725 | $ | (3,526 | ) | $ | (7,251 | ) | N.M | . | ||||||
Revenues. Revenues increased by $1.1 million, or 8%, to $15.0 million in the year ended December 31, 2009 as compared to $13.9 million in the year ended December 31, 2008. The increase was primarily due to increases of collaborative licensing and development revenue of $1.2 million and royalties of $0.4 million, offset by a decrease in supply revenue of $0.5 million. The increase in collaborative licensing and development revenue reflected in part a $1.0 million increase in contract development activities for Amylin in 2009, and an increase in 2009 milestone revenue resulting from a milestone payment from our U.S. DepoDur commercial partner, EKR, paid at the end of 2008. The increase in royalties in 2009 reflected an increase in end user sales of DepoCyt(e) in 2009, offset by a decline in DepoDur royalties. The decrease in supply revenue in 2009 was primarily due to EKR gradually selling down excess inventory accumulated in 2008.
Cost of Revenues. Cost of revenues decreased by $5.2 million, or 30%, to $12.3 million in the year ended December 31, 2009 as compared to $17.5 million in the year ended December 31, 2008. The decrease was primarily due to reduction in cost of supply revenue, driven by cost control measures initiated in December 2008 and April 2009, including a reduction in force of manufacturing and support personnel, decreased reliance on outsourced providers to support our manufacturing activities, and elimination of non-essential activities.
Research and Development Expenses. Research and development expenses decreased by $7.0 million, or 21%, to $26.2 million in the year ended December 31, 2009 from $33.2 million in the year ended December 31, 2008. This decrease resulted primarily from a $6.1 million decrease in clinical trials costs, to $8.7 million in 2009 from $14.8 million in 2008. In 2009, we completed our pivotal Phase 3 placebo controlled studies, as compared to in 2008 when we incurred most of the expenses for three Phase 3 comparator studies as well as three Phase 2 studies.
In the years ended December 31, 2009 and 2008, research and development expenses attributable to EXPAREL were $25.2 million, or 96%, and $31.9 million, or 96% of total research and development expenses, respectively. The remaining research and development expenses related to our other product candidate initiatives.
Selling, General and Administrative Expenses. Selling, general and administrative expenses decreased by $3.6 million, or 42%, to $5.0 million in the year ended December 31, 2009 from $8.6 million in the year ended December 31, 2008. Selling expenses were $1.6 million lower in 2009 as compared to 2008, as we curtailed our pre-commercial efforts in early 2009, resulting in $1.0 million decrease in outside services and $0.3 million decrease in compensation expenses. General and administrative expenses decreased by $2.0 million in the year ended December 31, 2009 as compared to 2008, primarily due to a $0.8 million decrease in salary expenses and a $0.7 million decrease in severance and recruiting expenses.
Other Income (Expense). Other income increased by $0.6 million, to $0.4 million in the year ended December 31, 2009 as compared to $0.2 million in other expense in the year ended December 31, 2008. The increase was primarily due to a gain realized on settlement with trade creditors in 2009.
Interest Income (Expense). Interest expense increased by $7.3 million in the year ended December 31, 2009, to $3.5 million, as compared to interest income of $3.7 million in the year ended December 31, 2008. $5.4 million of the increase in interest expense was primarily attributable to the royalty interest obligation under the Amended and Restated Royalty Interests Assignment Agreement and $1.7 million was due to our debt financing activities in 2009. The interest income (expense) relating to the obligations under the Amended and Restated Royalty Interests Assignment Agreement is composed of (1) the difference in the revaluation of our obligations under the Amended and Restated Royalty Interests Assignment Agreement between each reporting period and (2)
69
Table of Contents
the actual royalty interest payments payable pursuant to the Amended and Restated Royalty Interests Assignment Agreement for such reporting period. In determining the amount of the royalty interest obligation, we employ estimates of future cash flows derived from our royalties payable to Paul Capital based on end user sales of DepoCyt(e) and DepoDur, discounted at a rate that reflects an estimate of the cost of capital under the Amended and Restated Royalty Interests Assignment Agreement. At December 31, 2008, our estimate of future end user sales of DepoCyt(e) and DepoDur was considerably lower than the estimate as of December 31, 2007. This lower estimate resulted in a decrease of the royalty interest obligation valuation of $10.2 million at December 31, 2007 to $5.0 million at December 31, 2008. As a result, $5.2 million of the royalty interest obligation was recorded as interest income in the year ended December 31, 2008. In comparison, the valuation of the royalty interest obligation of $5.2 million at December 31, 2009 was slightly higher than the valuation of $5.0 million at December 31, 2008, which resulted in a $0.2 million interest expense in the year ended December 31, 2009.
Liquidity and Capital Resources
Since our inception in 2007, we have devoted most of our cash resources to research and development and general and administrative activities primarily related to the development of EXPAREL. We have financed our operations primarily with the proceeds of the sale of convertible preferred stock, secured and unsecured notes and borrowings under debt facilities, supply revenue, royalties and collaborative licensing and development revenue. To date, we have generated limited supply revenue and royalties, and we do not anticipate generating any revenues from the sale of EXPAREL, if approved, until at least the fourth quarter of 2011. We have incurred losses and generated negative cash flows from operations since inception. As of December 31, 2010, we had an accumulated deficit of $136.9 million, cash and cash equivalents of $26.1 million and working capital of $14.8 million.
The following table summarizes our cash flows from operating, investing and financing activities for the years ended December 31, 2008, 2009 and 2010:
| Year Ended December 31, | ||||||||||||
| 2008 | 2009 | 2010 | ||||||||||
| (in thousands) | ||||||||||||
| Consolidated Statement of Cash Flows Data: |
||||||||||||
| Net cash provided by (used in) |
||||||||||||
| Operating activities |
$ | (29,189 | ) | $ | (20,838 | ) | $ | (24,880 | ) | |||
| Investing activities |
(5,838 | ) | (5,509 | ) | (6,769 | ) | ||||||
| Financing activities |
40,173 | 21,038 | 50,705 | |||||||||
| Net increase (decrease) in cash and cash equivalents |
$ | 5,146 | $ | (5,309 | ) | $ | 19,056 | |||||
Operating Activities
For the years ended December 31, 2010, 2009 and 2008, our net cash used in operating activities was $24.9 million, $20.8 million and $29.2 million, respectively. The increase in net cash used in operating activities in 2010 resulted from an absence of milestone receipts in 2010 compared to 2009, when the Company received $5.0 million license fees from one of our commercial partners, and higher interest expense on the Company’s credit facilities, offset by lower expenses on research and development. The decrease in net cash used in operating activities in 2009 resulted from lower research and development and selling expenses and a $3.8 million increase in the deferred revenue balance, primarily due to receipt of license fees from one of our commercial partners, offset by a decrease in accounts payable of $4.4 million.
Investing Activities
For the years ended December 31, 2010, 2009 and 2008, our net cash used in investing activities was $6.8 million, $5.5 million and $5.8 million, respectively. The net cash used in investing activities in 2010 and 2009 and 2008 was primarily for the purchases of fixed assets of $6.8 million, $5.5 million, and $5.8 million, respectively as the Company constructed its manufacturing capability to produce EXPAREL.
70
Table of Contents
Financing Activities
For the years ended December 31, 2010, 2009 and 2008, our net cash provided by financing activities was $50.7 million, $21.0 million and 40.2 million, respectively. The net cash provided by financing activities in 2010 was primarily due to the Hercules Credit Facility for net proceeds of $25.8 million, sale and issuance of secured notes for net proceeds of $18.6 million, and sale and issuance of convertible notes to certain of our existing investors for net proceeds of $7.5 million. The net cash provided by financing activities in 2009 was primarily due to the sale and issuance of notes payable, for total net proceeds of $21.0 million. The cash provided by financing activities in 2008 was due primarily to the sale and issuance of our Series A convertible preferred stock, for total net proceeds of $40.0 million.
Equity Financings
From inception through December 31, 2010, we have received net proceeds of $85 million from the sale of our Series A convertible preferred stock. The various issuances of our Series A convertible preferred stock are described in more detail under “Related Person Transactions—Preferred Stock Issuances.”
Common Stock
In connection with our formation, we issued in March 2007 an aggregate of 464,900 shares of common stock for total aggregate consideration of $50,000.
Series A Convertible Preferred Stock
In March 2007, we entered into a Series A Preferred Stock Purchase Agreement pursuant to which we issued and sold an aggregate of 6,322,640 shares of Series A convertible preferred stock in four separate closings held in March 2007, February 2008, July 2008 and October 2008, at a purchase price of $13.44 per share. The aggregate consideration for the shares of Series A convertible preferred stock was $85 million in cash.
Warrants
On January 22, 2009, we issued warrants in connection with the issuance of the 2009 Convertible Notes (see “Investor Notes Converted into Common Stock”). The warrants are convertible into an aggregate of 158,061 of shares of our common stock at an exercise price of $2.69 per share and will expire on January 21, 2014.
On July 2, 2009, we issued warrants to the landlord of our two San Diego facilities in connection with amendments to respective lease agreements that deferred minimum annual rental obligations. The warrants are exercisable for an aggregate of 23,244 shares of Series A convertible preferred stock at a price of $13.44 per share and will expire on the earlier of July 1, 2016 or the fifth anniversary of the consummation of our initial public offering.
On November 24, 2010, we issued warrants in connection with the Hercules Credit Facility to the lenders to purchase 178,986 shares of our Series A convertible preferred stock (see “Debt Facilities”). The warrants are exercisable at a price of $13.44 per share and shall be valid from the date of issuance until the earlier to occur of ten (10) years from the date of issuance or five (5) years following the effective date of the registration statement for an initial public offering.
On December 29, 2010, we issued warrants in connection with the December 2010 Convertible Notes (see “Investor Notes Converted into Common Stock”). The warrants are convertible into an aggregate of 167,361 of shares of our common stock with an exercise price of $13.44 per share and will expire on December 29, 2017.
Debt Facilities
As of December 31, 2010, after giving effect to the Hercules Credit Facility and the issuance and sale of the December 2010 Convertible Notes and the application of the proceeds therefrom, we had $26.25 million of
71
Table of Contents
indebtedness under the Hercules Credit Facility. Pursuant to an agreement entered into in October 2010 between us and the holders of the convertible and secured notes, all principal and accrued interest on the convertible and secured notes (other than the December 2010 Convertible Notes) converted into 3,264,777 shares of our common stock upon completion of our initial public offering at a conversion price of $13.44, in accordance with the terms of the October 2010 agreement and the December 2010 Convertible Notes were converted into 1,071,428 shares of our common stock at a conversion price equal to our initial public offering price of $7.00 per share. The table below shows the principal amount of our indebtedness and the number of shares of our common stock that our indebtedness was converted into.
| Debt Issue |
Principal Amount | Conversion Shares | ||||||
| Hercules Credit Facility |
$ | 26.25 million | — | |||||
| 2009 Convertible Notes |
10.63 million | 871,635 | ||||||
| 2009 Secured Notes |
10.63 million | 927,881 | ||||||
| 2010 Secured Notes |
15.00 million | 1,156,606 | ||||||
| HBM Secured Notes |
3.75 million | 308,655 | ||||||
| December 2010 Convertible Notes |
7.50 million | 1,071,428 | (1) | |||||
| (1) | The December 2010 Convertible Notes were converted into shares of our common stock at a conversion price equal to the initial public offering price of $7.00 per share. |
Hercules Credit Facility. On November 24, 2010, we entered into a $26.25 million credit facility with Hercules Technology Growth Capital, Inc. and Hercules Technology III, L.P., as lenders. At the closing of the Hercules Credit Facility, we entered into a term loan in the aggregate principal amount of $26.25 million, which was the full amount available under the Hercules Credit Facility. As of December 31, 2010, the entire term loan of $26.25 million was outstanding. The term loan under the Hercules Credit Facility is comprised of two tranches, Tranche A and Tranche B. The Tranche A portion of the term loan is comprised of $11.25 million in principal and carries a floating per annum interest rate equal to 10.25% plus the amount, if any, by which the prime rate exceeds 4.00%. Upon the release of the investors’ guaranty (described below), the interest rate on the Tranche A portion of the term loan will increase to a floating per annum interest rate equal to 11.00% plus the amount, if any, by which the prime rate exceeds 4.00%. The Tranche B portion of the term loan is comprised of $15.0 million in principal and carries a floating per annum interest rate equal to 12.65% plus the amount, if any, by which the prime rate exceeds 4.00%. As of December 31, 2010, the interest rate on the Tranche A portion was 10.25% and on the Tranche B portion was 12.65%. Interest on the term loan is payable monthly. If there is an event of default under the Hercules Credit Facility, we will be obligated to pay interest at a higher default rate. The proceeds of the term loan under the Hercules Credit Facility have been used to repay the GECC Credit Facility in full and the remainder will be used for other general corporate purposes.
As further consideration to the lenders to provide the term loan to us under the Hercules Credit Facility, we issued to the lenders a warrant to purchase 178,986 shares of our common stock. The exercise price for the shares to be issued under the warrant is equal to $13.44 per share. The warrant is valid from the date of issuance until the earlier to occur of ten (10) years from the date of issuance or five (5) years following the effective date of the registration statement for our initial public offering.
The Hercules Credit Facility provides for an “interest only period” when no principal amounts are due and payable. The interest only period runs initially from November 24, 2010 through August 31, 2011, but can be extended, at our request, to either November 30, 2011 or February 28, 2012 if certain conditions are satisfied. Following the end of the interest only period, the term loan is to be repaid in 33 equal monthly installments of principal and interest beginning on the first business day after the month in which the interest only period ends. Amounts repaid may not be re-borrowed. We can, at any time, prepay all or any part of the term loan provided that so long as the investors’ guaranty (as described below) is in effect, we cannot prepay any part of the Tranche A portion of the term loan without the lenders’ consent if any of the Tranche B portion is outstanding. If the investors’ guaranty is not in effect, then any prepayments are to be applied pro rata across the outstanding balance of both portions of the term loan. In connection with any prepayments of the term loan under the Hercules Credit Facility,
72
Table of Contents
we are required to pay, in addition to all principal and accrued and unpaid interest on such term loan, a prepayment charge equal to 1.25% of the principal amount being prepaid. In addition, there is an end of term charge that is payable to the lenders upon the earliest to occur of the maturity date, the prepayment in full of our obligations under the Hercules Credit Facility and the acceleration of our obligations under the Hercules Credit Facility.
The Hercules Credit Facility is secured by a first priority lien on all of our assets other than the assets that secure our obligations under Amended and Restated Royalty Interests Assignment Agreement (as described below). In addition, the Hercules Credit Facility is guaranteed by certain of our investors (other than the entities affiliated with HBM) on a several and not joint basis which guarantee is limited to each investor’s pro rata portion of the outstanding principal and accrued and unpaid interest under the Hercules Credit Facility, but in no event exceeding $11.25 million in the aggregate. The Hercules loan agreement provides that, upon the occurrence of certain circumstances and upon our request, the investors’ guarantee may be terminated and released.
The Hercules loan and security agreement contains events of default including payment default, default arising from the breach of the provisions of the Hercules loan and security agreement and related documents (including the occurrence of certain changes in control, including if our chief executive officer ceases under certain conditions to be involved in the daily operations or management of the business, or if certain holders of our capital stock cease to retain, after the consummation of certain corporate transactions, shares representing more than 50% of the surviving entity after such transactions (provided that our initial public offering shall not constitute such a change in control)) or the inaccuracy of representations and warranties contained in the loan and security agreement, attachment default, bankruptcy and insolvency, cross-default with respect to certain other indebtedness (including certain events under the Amended and Restated Royalty Interests Assignment Agreement), breach of the terms of any guarantee (including the investors’ guarantee) of the Hercules Credit Facility, the occurrence of a material adverse effect (as defined in the Hercules loan and security agreement).
The occurrence of an event of default under the Hercules Credit Facility could trigger the acceleration of our obligations under the Hercules Credit Facility or allow the lenders to exercise other rights and remedies, including rights against our assets that secure the Hercules Credit Facility and rights under guarantees provided to support the obligations under the Hercules Credit Facility.
The Hercules loan and security agreement contains a number of affirmative and restrictive covenants, including reporting requirements and other collateral limitations, certain limitations on liens and indebtedness, dispositions, mergers and acquisitions, restricted payments and investments, corporate changes and waivers and amendments to certain agreements, our organizational documents, and documents relating to debt that is subordinate to our obligations under the Hercules Credit Facility.
GECC Credit Facility. On April 30, 2010, we entered into an $11.25 million credit facility with General Electric Capital Corporation, as both agent and the sole lender, or the GECC Credit Facility. We borrowed the full $11.25 million under the GECC Credit Facility. On November 24, 2010, all borrowings under the GECC Credit Facility were repaid in full from proceeds of the Hercules Credit Facility, and the GECC Credit Facility was terminated and any and all liens in favor of the lenders under the GECC Credit Facility were released.
Investor Notes Converted into Common Stock.
2009 Convertible Notes. In January 2009, we sold $10.63 million in aggregate principal amount of convertible promissory notes, or the 2009 Convertible Notes, to certain of our existing investors. In connection with the issuance of the 2009 Convertible Notes, we issued warrants to purchase an aggregate of 158,061 shares of our common stock with an exercise price of $2.69 per share. The 2009 Convertible Notes have an interest rate of 5% per year and all principal and accrued and unpaid interest on the 2009 Convertible Notes was due on December 31, 2010. In connection with entering into the Hercules Credit Facility, the maturity date of the 2009 Convertible Notes was extended to the earliest of (1) a sale of us, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. In connection with entering into the Hercules Credit Facility, the holders of the 2009 Convertible Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the 2009 Convertible Notes were subordinated to the Hercules Credit Facility. The
73
Table of Contents
holders of the 2009 Convertible Notes previously entered into a separate intercreditor agreement with the holders of the 2009 Secured Notes (as described below) and the 2010 Secured Notes (as described below) pursuant to which the 2009 Convertible Notes were subordinated to the 2009 Secured Notes and the 2010 Secured Notes, and the holders of the 2009 Secured Notes agreed to share payments pro rata with the holders of the 2010 Secured Notes. As of December 31, 2010, $11.67 million aggregate principal and accrued and unpaid interest was outstanding under the 2009 Convertible Notes. All principal and interest due on the 2009 Convertible Notes was converted into 871,635 shares of our common stock upon the completion of our initial public offering.
2009 Secured Notes. In June 2009, we entered into an agreement with certain of our existing investors to issue $10.63 million in aggregate principal amount of secured notes, or the 2009 Secured Notes. To secure the performance of our obligations under the purchase agreement for the 2009 Secured Notes, we granted a security interest in substantially all of our assets, including our intellectual property assets, except the assets that secure our obligations under our agreement with Paul Capital. In connection with entering into the Hercules Credit Facility, the holders of the 2009 Secured Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the 2009 Secured Notes were subordinated to the Hercules Credit Facility.
The 2009 Secured Notes had an interest rate of 12% per year and all principal and accrued and unpaid interest on the 2009 Convertible Notes was due on December 31, 2010. In connection with entering into the Hercules Credit Facility, the maturity date of the 2009 Secured Notes was extended to the earliest of (1) a sale of us, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. As of December 31, 2010, $12.32 million aggregate principal and accrued and unpaid interest was outstanding under the 2009 Secured Notes. All principal and interest due on the 2009 Secured Notes was converted into 927,881 shares of our common stock upon the completion of our initial public offering.
2010 Secured Notes. In March 2010, we entered into an agreement with certain of our existing investors to issue $15.0 million in aggregate principal amount of secured notes and the investors purchased the entire $15.0 million of 2010 Secured Notes. To secure the performance of our obligations under the purchase agreement for the 2010 Secured Notes, we granted a subordinated security interest in substantially all of our assets, including our intellectual property assets, except the assets that secure our obligations under the Amended and Restated Royalty Interests Agreement. In connection with entering into the Hercules Credit Facility, the holders of the 2010 Secured Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the 2010 Secured Notes were subordinated to the Hercules Credit Facility.
The 2010 Secured Notes have an interest rate of 5% per year and all principal and accrued and unpaid interest on the 2010 Secured Notes is due on December 31, 2010. In connection with entering into the Hercules Credit Facility, the maturity date of the 2010 Secured Notes was extended to the earliest of (1) a sale of us, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. As of December 31, 2010, $15.46 million in aggregate principal and accrued and unpaid interest was outstanding pursuant to the 2010 Secured Notes. All principal and interest due on the 2010 Secured Notes was converted into 1,156,606 shares of our common stock upon the completion of our initial public offering.
HBM Term Loan. On April 30, 2010, we entered into a subordinated secured note purchase agreement with entities affiliated with HBM BioVentures, or HBM, to issue $3.75 million in aggregate principal amount of secured notes, or the HBM Secured Notes. To secure the performance of our obligations under the purchase agreement for the HBM Secured Notes, we granted a subordinated security interest in substantially all of our assets, including our intellectual property assets, other than the assets that secure our obligations under the Amended and Restated Royalty Interests Agreement. The HBM Secured Notes carry an interest rate of approximately 10% per year. In addition, the HBM Secured Notes require a final payment fee if they are prepaid prior to the maturity date. The maturity date of the HBM Secured Notes is the earliest of (1) a sale of us, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are
74
Table of Contents
paid in full and the Hercules Credit Facility is terminated. In connection with entering into the Hercules Credit Facility, the holders of the HBM Secured Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the HBM Secured Notes were subordinated to the Hercules Credit Facility. As of December 31, 2010, $3.94 million in aggregate principal and accrued and unpaid interest was outstanding pursuant to the HBM Secured Notes. All principal and interest due on the HBM Secured Notes was converted into 308,655 shares of our common stock upon the completion of our initial public offering.
December 2010 Convertible Notes. On December 29, 2010, we sold $7.5 million in aggregate principal amount of convertible promissory notes, or the December 2010 Convertible Notes, in a private placement to certain of our existing investors. In connection with the issuance and sale of the December 2010 Convertible Notes, we issued warrants to the holders of the December 2010 Convertible Notes to purchase an aggregate of 167,361 shares of our common stock with an exercise price of $13.44 per share. The December 2010 Convertible Notes will had an interest rate of 5% per year from and after March 31, 2011 and all principal and accrued and unpaid interest on the December 2010 Convertible Notes was due and payable upon the earliest of: (1) sale of us, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. The December 2010 Convertible Notes were converted into 1,071,428 shares of our common stock at a conversion price equal to our initial public offering price of $7.00 per share.
Royalty Interests Assignment Agreement
On March 23, 2007, we entered into the Amended and Restated Royalty Interests Assignment Agreement with Paul Capital, pursuant to which we assigned to Paul Capital the right to receive up to approximately 20% of our royalty payments from DepoCyt(e) and DepoDur. The original agreement was entered into prior to the Acquisition by the Predecessor in order to monetize certain royalty payments from DepoCyt(e) and DepoDur. In connection with the Acquisition, the original agreement with Paul Capital was amended and restated and the responsibility to pay the royalty interest in product sales of DepoCyt(e) and DepoDur was transferred to us and we were required to make payments to Paul Capital upon the occurrence of certain events. To secure our obligations to Paul Capital, we granted Paul Capital a security interest in collateral which includes the royalty payments we are entitled to receive with respect to sales of DepoCyt(e) and DepoDur, as well as to bank accounts to which such payments are deposited. Under our arrangement with Paul Capital, upon the occurrence of certain events, including if we experience a change of control, undergo certain bankruptcy events of us or our subsidiary, transfer any or substantially all of our rights in DepoCyt(e) and/or DepoDur, transfer all or substantially all of our assets, breach certain of the covenants, representations or warranties under the Amended and Restated Royalty Interests Assignment Agreement, or sales of DepoCyt(e) and/or DepoDur are suspended due to an injunction or if we elect to suspend sales of DepoCyt(e) and/or DepoDur as a result of a lawsuit filed by certain third parties, Paul Capital may require us to repurchase the rights we assigned to it at a cash price equal to (a) 50% of all cumulative payments made by us to Paul Capital under the Amended and Restated Royalty Interests Assignment Agreement during the preceding 24 months, multiplied by (b) the number of days from the date of Paul Capital’s exercise of such option until December 31, 2014, divided by 365. Under the terms of the Amended and Restated Royalty Interests Assignment Agreement, our initial public offering did not constitute a change of control.
Future Capital Requirements
In February 2011, we completed an initial public offering of our common stock pursuant to a registration statement on Form S-1, as amended (File No. 333-170245) that was declared effective on February 2, 2011. Under the registration statement, we registered the offering and sale of an aggregate of 6,900,000 shares of our common stock. An aggregate of 6,000,000 shares of common stock registered under the registration statement were sold at a price to the public of $7.00 per share. Barclays Capital Inc. and Piper Jaffray and Co. acted as joint book running managers of the offering and as representatives of the underwriters. The offering commenced on February 3, 2011 and closed on February 8, 2011. The over-allotment option was not exercised by the underwriters. As a result of our IPO, we raised a total of $42.0 million in gross proceeds, and approximately $37.0 million in net proceeds after deducting approximately $5.0 million in underwriting discounts and commissions and estimated offering expenses.
75
Table of Contents
We believe that the net proceeds from our initial public offering, together with our existing cash and cash equivalents and revenue from product sales, will be sufficient to enable us to fund our operating expenses, capital expenditure requirements and service our indebtedness for at least the next 12 months. However, no assurance can be given that this will be the case, and we may require additional debt or equity financing to meet our working capital requirements. We expect that the net proceeds from our initial public offering will be sufficient for our planned manufacture and commercialization of EXPAREL in the United States. Our need for additional external sources of funds will depend significantly on the level and timing of our sales of EXPAREL. Moreover, changing circumstances may cause us to expend cash significantly faster than we currently anticipate, and we may need to spend more cash than currently expected because of circumstances beyond our control.
Our expectations regarding future cash requirements do not reflect the potential impact of any future acquisitions, mergers, dispositions, joint ventures or investments that we make in the future. We have no current understandings, agreements or commitments for any material acquisitions or licenses of any products, businesses or technologies. We may need to raise substantial additional capital in order to engage in any of these types of transactions.
We expect to continue to incur substantial additional operating losses as we seek FDA approval for and commercialize EXPAREL and develop and seek regulatory approval for our other product candidates. If we obtain FDA approval for EXPAREL, we will incur significant sales and marketing and manufacturing expenses. In addition, we expect to incur additional expenses to add operational, financial and information systems and personnel, including personnel to support our planned product commercialization efforts. We also expect to incur significant costs to comply with corporate governance, internal controls and similar requirements applicable to us as a public company following the closing of our initial public offering.
Our future use of operating cash and capital requirements will depend on many forward-looking factors, including the following:
| • | the timing and outcome of the FDA’s review of the NDA for EXPAREL; |
| • | the extent to which the FDA may require us to perform additional clinical trials for EXPAREL; |
| • | the costs of our commercialization activities for EXPAREL, if it is approved by the FDA; |
| • | the cost and timing of expanding our manufacturing facilities and purchasing manufacturing and other capital equipment for EXPAREL and our other product candidates; |
| • | the scope, progress, results and costs of development for additional indications for EXPAREL and for our other product candidates; |
| • | the cost, timing and outcome of regulatory review of our other product candidates; |
| • | the extent to which we acquire or invest in products, businesses and technologies; |
| • | the extent to which we choose to establish collaboration, co-promotion, distribution or other similar agreements for our product candidates; and |
| • | the costs of preparing, submitting and prosecuting patent applications and maintaining, enforcing and defending intellectual property claims. |
To the extent that our capital resources are insufficient to meet our future operating and capital requirements, we will need to finance our cash needs through public or private equity offerings, debt financings, corporate collaboration and licensing arrangements or other financing alternatives. The covenants under the Hercules Credit Facility and the Amended and Restated Royalty Interests Assignment Agreement and the pledge of
76
Table of Contents
our assets as collateral limit our ability to obtain additional debt financing. We have no committed external sources of funds. Additional equity or debt financing or corporate collaboration and licensing arrangements may not be available on acceptable terms, if at all.
If we raise additional funds by issuing equity securities, our stockholders will experience dilution. Debt financing, if available, would result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. Any debt financing or additional equity that we raise may contain terms, such as liquidation and other preferences, that are not favorable to us or our stockholders. If we raise additional funds through collaboration and licensing arrangements with third parties, it may be necessary to relinquish valuable rights to our technologies, future revenue streams or product candidates or to grant licenses on terms that may not be favorable to us.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements, except for operating leases, or relationships with unconsolidated entities or financial partnerships, such as entities often referred to as structured finance or special purpose entities.
Contractual Obligations
The following table summarizes our contractual obligations as of December 31, 2010:
| Payments Due by Period | ||||||||||||||||
| Total | 2011 | 2012 and 2013 |
2014 and 2015 |
|||||||||||||
| (in thousands) | ||||||||||||||||
| Contractual Obligations(1): |
||||||||||||||||
| Debt Obligations(2) |
$ | 26,250 | $ | 3,182 | $ | 19,091 | $ | 3,977 | ||||||||
| Interest payments on debt(2) |
6,684 | 3,064 | 3,456 | 164 | ||||||||||||
| Operating lease obligations(3) |
23,821 | 5,827 | 9,786 | 8,208 | ||||||||||||
| $ | 56,755 | $ | 12,073 | $ | 32,333 | $ | 12,349 | |||||||||
| (1) | This table does not include (i) royalties payable to Paul Capital (through 2014 pursuant to the Amended and Restated Royalty Interest Assignment Agreement described in “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Liquidity and Capital Resources—Royalty Interests Assignment Agreement”) and pursuant to the Assignment Agreement with Research Development Foundation; (ii) contingent milestone payments of up to $62 million related to EXPAREL due to SkyePharma PLC, including $10 million due upon the first commercial sale of EXPAREL to end users in the United States. |
| (2) | Debt obligations and interest payments includes payments under the Hercules Credit Facility entered into on November 24, 2010, and exclude the secured and unsecured notes (including the December 2010 Convertible Notes) and accrued interest thereon that were converted into common stock in our initial public offering. |
| (3) | Includes building and equipment leases. |
Recent Accounting Pronouncements
In October 2009, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update No. 2009-13, “Multiple-Deliverable Revenue Arrangements” (“ASU 2009-13”). ASU 2009-13, amends existing revenue recognition accounting pronouncements that are currently within the scope of ASC Subtopic 605-25. This authoritative guidance provides principles for allocation of consideration among its multiple-elements, allowing more flexibility in identifying and accounting for separate deliverables under an arrangement. ASU 2009-13 introduces an estimated selling price method for valuing the elements of a bundled arrangement if vendor-
77
Table of Contents
specific objective evidence or third-party evidence of selling price is not available, and significantly expands related disclosure requirements. This guidance is effective on a prospective basis for revenue arrangements entered into or materially modified in fiscal years beginning on or after June 15, 2010. Alternatively, adoption may be on a retrospective basis, and early application is permitted. The adoption of this guidance is not expected to have any impact on our consolidated financial statements.
In April 2010, the FASB issued Accounting Standards Update No. 2010-17, “Milestone Method of Revenue Recognition (Topic 605)” (“ASU 2010-17”). This update provides guidance on defining a milestone and determining when it may be appropriate to apply the milestone method of revenue recognition for research or development transactions. Authoritative guidance on the use of the milestone method did not previously exist. This guidance is effective on a prospective basis for milestones achieved in fiscal years, and interim periods within those years, beginning on or after June 15, 2010. Alternatively, retrospective adoption is permitted for all prior periods. The adoption of this guidance is not expected to have any impact on our consolidated financial statements.
Other pronouncements issued by the FASB or other authoritative accounting standards groups with future effective dates are either not applicable or not significant to the consolidated financial statements of the Company.
| Item 7A. | Quantitative and Qualitative Disclosures about Market Risk |
The primary objective of our investment activities is to preserve our capital to fund operations. We also seek to maximize income from our investments without assuming significant risk. Our exposure to market risk is confined to our cash and cash equivalents. As of December 31, 2010, we had cash and cash equivalents of $26.1 million. We do not engage in any hedging activities against changes in interest rates. Because of the short-term maturities of our cash and cash equivalents, we do not believe that an increase in market rates would have any significant impact on the realized value of our investments, but may increase the interest expense associated with our debt.
We have commercial partners for DepoCyte and DepoDur who sell our products in the EU. Under these agreements, we provide finished goods to our commercial partners in exchange for euro-denominated supply revenue, and we also receive euro-denominated royalties on market sales when the products are sold to end users. Under these agreements, we received $7.6 million in the year ended December 31, 2010, $7.2 million in the year ended December 31, 2009 and $7.3 million in the year ended December 31, 2008 from these commercial partners.
Because of these agreements, we are subject to fluctuations in exchange rates, specifically in the relative values of the U.S. dollar and the euro. We estimate that an unfavorable fluctuation in exchange rates of 10% would have an impact of approximately $0.7 million on our annual revenue. Between January 2008 and December 2010 the exchange rate between the U.S. dollar and the Euro ranged between $1.58 per Euro and $1.22 per Euro.
| Item 8. | Financial Statements and Supplementary Data |
Our consolidated financial statements, together with the report of our independent registered public accounting firm, appear on pages F-1 through F-35, of this Annual Report on Form 10-K.
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
There have been no disagreements with our independent accountants on accounting and financial disclosure matters.
| Item 9A. | Controls and Procedures |
Our management, with the participation of our chief executive officer and chief financial officer, evaluated the effectiveness of our disclosure controls and procedures as of December 31, 2010. The term “disclosure controls and procedures,” as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act, means controls and other procedures of a company that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported, within the
78
Table of Contents
time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to the company’s management, including its principal executive and principal financial officers, as appropriate to allow timely decisions regarding required disclosure. Management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on the evaluation of our disclosure controls and procedures as of December 31, 2010, our chief executive officer and chief financial officer concluded that, as of such date, our disclosure controls and procedures were effective.
This Form 10-K does not include a report of management’s assessment regarding internal control over financial reporting due to a transition period established by rules of the Securities and Exchange Commission for newly public companies.
No change in our internal control over financial reporting occurred during the fiscal quarter ended December 31, 2010 that has materially affected, or is reasonably likely to materially affect, the Company’s internal control over financial reporting.
| Item 9B. | Other Information |
None.
79
Table of Contents
PART III
| Item 10. | Directors, Executive Officers and Corporate Governance |
Executive Officers and Directors
Our executive officers and directors, their current positions and their ages as of March 15, 2011 are set forth below:
| Name |
Age | Position(s) | ||||
| David Stack |
59 | President and Chief Executive Officer, Director | ||||
| James Scibetta |
46 | Chief Financial Officer | ||||
| Gary Patou, M.D |
52 | Chief Medical Officer | ||||
| William Lambert, Ph.D. |
52 | Senior Vice President, Pharmaceutical Development | ||||
| Mark Walters |
55 | Senior Vice President, Technical Operations | ||||
| Fred Middleton(2) |
61 | Chairman of the Board of Directors | ||||
| Luke Evnin, Ph.D.(2) |
47 | Director | ||||
| Carl Gordon, Ph.D.(1) |
46 | Director | ||||
| John Longenecker, Ph.D.(1)(2)(3) |
63 | Director | ||||
| Gary Pace, Ph.D.(1)(3) |
63 | Director | ||||
| Andreas Wicki, Ph.D. |
52 | Director | ||||
| (1) | Member of audit committee. |
| (2) | Member of compensation committee. |
| (3) | Member of nominating and corporate governance committee. |
David Stack has served as our president and chief executive officer and as a director since November 2007. Mr. Stack has been a managing director of MPM Capital since 2005 and a managing partner of Stack Pharmaceuticals, Inc. since 1998. From 2001 to 2004, he was president and chief executive officer of The Medicines Company (NASDAQ: MDCO). Previously, Mr. Stack was president and general manager at Innovex, Inc. He was vice president, business development/marketing at Immunomedics from 1993 until 1995. Prior to that, he was with Roche Laboratories in positions of increasing responsibility from 1981 until 1993, including therapeutic world leader in infectious disease and director, business development and planning, infectious disease, oncology, and virology. He currently serves as a member of the board of directors of PepTx, Inc., and Molecular Insight Pharmaceuticals, Inc. (NASDAQ: MIPI). Mr. Stack holds a B.S. in pharmacy from Albany College of Pharmacy and a B.S. in Biology from Siena College. We believe Mr. Stack’s qualifications to sit on our board of directors include his extensive experience with pharmaceutical companies, his financial expertise and his years of experience providing strategic and financial advisory services to pharmaceutical and biotechnology organizations, including evaluating business plans involving clinical trials.
James Scibetta has served as our chief financial officer since August 2008. Prior to that, Mr. Scibetta was chief financial officer of Bioenvision, Inc. (NASDAQ: BIVN) from 2006 until its acquisition by Genzyme, Inc. in 2007. From 2001 to 2006, Mr. Scibetta was executive vice president and chief financial officer of Merrimack Pharmaceuticals, Inc., and he was a member of the board of directors of Merrimack from 1998 to 2004. Mr. Scibetta formerly served as a senior investment banker at Shattuck Hammond Partners, LLC and PaineWebber Inc., providing capital acquisition, merger and acquisition, and strategic advisory services to healthcare companies. He currently serves as chairman of the board and audit committee of Nephros, Inc. (NASDAQ: NEPH). Mr. Scibetta holds a B.S. in physics from Wake Forest University, and an M.B.A. in finance from the University of Michigan. He completed executive education studies in the Harvard Business School Leadership & Strategy in Pharmaceuticals and Biotechnology program.
Gary Patou, M.D. has served as our chief medical officer since March 2009. Dr. Patou has been a managing director of MPM Capital since 2005. He has served as chief medical officer of the following MPM Capital portfolio companies: Peplin, Ltd. (ASX: PLI), from July 2006 to April 2007 and from June 2008 to May
80
Table of Contents
2009, Cerimon Pharmaceuticals, Inc., from June 2005 to June 2006, and Oscient Pharmaceuticals, Inc., from February 2004 to April 2005. Dr. Patou currently spends part of his time as the acting chief executive officer of Cerimon Pharmaceuticals, Inc. From 2001 to 2004, he was president of Genesoft and from 1995 to 2000, Dr. Patou worked at SmithKline Beecham Pharmaceuticals, now a unit of GlaxoSmithKline (LSE: GSK), where he held positions of increasing responsibility including senior vice president and director, project and portfolio management. From 1991 to 1995, he held increasing senior, director level positions at Vernalis (LSE:VER), formerly British Biotechnology. He currently serves as a member of the board of directors of Xenon Pharmaceuticals, Inc. Dr. Patou has held a number of academic appointments at University College & Middlesex School of Medicine in London and holds an M.D. from University College, London.
William Lambert, Ph.D. has served as our senior vice president, pharmaceutical development since the Acquisition in March 2007. Dr. Lambert has served as senior vice president pharmaceutical development since he joined SkyePharma, Inc. in January 2006. From July 1997 until January 2006, Dr. Lambert held director positions at Eisai Inc., in drug delivery technology and pharmaceutical and analytical research and development, for almost ten years. Prior to Eisai, Dr. Lambert worked at Pfizer Inc. (NYSE: PFE) and The Upjohn Company (now Pfizer Inc.) as a research investigator with increasing levels of responsibility. Dr. Lambert is on the advisory council for the National Institute for Pharmaceutical Technology and Education, a U.S. Pharmacopeia Expert Committee, and on the advisory boards of the Journal of Pharmaceutical Sciences and the Handbook of Pharmaceutical Excipients. Dr. Lambert received his Ph.D. in pharmaceutics from the University of Utah and his B.S. in pharmacy from the University of Rhode Island.
Mark Walters has served as our senior vice president, technical operations since February 2008, and served as our vice president, business and commercial development since the Acquisition in March 2007. From January 2001 until March 2007, Mr. Walters was with SkyePharma, Inc. (LSE: SKP) serving as the vice president of business and commercial development and as director of both strategic sourcing and product management. From 1989 until 2001 Mr. Walters served in the positions of program director, project director and product director in the imaging and liquid ventilation areas for Alliance Pharmaceutical Corp. Mr. Walters received his B.A. in biology from Hamilton College.
Fred Middleton has served as our director since our inception in December 2006. Since 1987, he has been a general partner/managing director of Sanderling Ventures, a firm specializing in biomedical venture capital. From 1984 through 1986, he was the managing general partner of Morgan Stanley Ventures, an affiliate of Morgan Stanley & Co. Earlier in his career, Mr. Middleton was part of the of the founding management team at Genentech, Inc., a biotechnology company, serving there from 1978 through 1984 as vice president of finance and corporate development, and chief financial officer. During the last 30 years, he has participated in active management roles and as an investor and director in over 20 start-up biomedical companies. He currently serves as chairman of the board of Stereotaxis, Inc. (NASDAQ: STXS), a medical device company that markets magnetically guided robotic surgery systems in cardiology. He also currently serves as a board member of Cardionet, Inc. (NASDAQ: BEAT), a company that markets devices and services for wireless 24/7 real time monitoring of patients. He also serves as a director of seven other privately-held biomedical companies, engaged in the development of therapeutic and diagnostic products in healthcare. Mr. Middleton received a B.S. degree in chemistry from the Massachusetts Institute of Technology and an M.B.A. from Harvard Business School. We believe Mr. Middleton’s qualifications to sit on our board of directors include his extensive experience with biopharmaceutical and biotechnology companies, his financial expertise and his years of experience providing strategic advisory services to diverse companies.
Luke Evnin, Ph.D. has served as our director since our inception in December 2006. Dr. Evnin has served as a general partner or managing director at MPM Capital since co-founding the firm in 1998. Prior to joining MPM, Dr. Evnin was at Accel Partners from 1990 to 1997 serving as general partner from 1994 to 1997. Dr. Evnin has served as director of several public companies, including Epix Medical, Inc. (NASDAQ: EPIX), Metabasis Therapeutics, Inc., Oscient Pharmaceuticals Company, Restore Medical, Inc., Otix Global, Inc. (NASDAQ: OTIX), formerly known as Sonic Innovations, Inc. and Signal Pharmaceuticals, Inc. and is currently or has been a director of several private healthcare companies in both the medical device and biopharmaceutical sectors. Dr. Evnin earned his Ph.D. in biochemistry from the University of California, San Francisco and his A.B. in molecular biology from Princeton University. We believe Dr. Evnin’s qualifications to sit on our board of directors include his extensive experience with biopharmaceutical and biotechnology companies, his financial expertise and his years of experience providing strategic advisory services to diverse companies.
81
Table of Contents
Carl Gordon, Ph.D. has served as our director since our inception in December 2006. Dr. Gordon is a founding general partner of OrbiMed Advisors. Dr. Gordon is active in both private equity and small-capitalization public equity investments. Prior to founding OrbiMed Advisors in January 1998, Dr. Gordon was a senior biotechnology analyst at Mehta & Isaly from 1995 to 1997. He was a fellow at The Rockefeller University from 1993 to 1995. Dr. Gordon received a Ph.D. in molecular biology from the Massachusetts Institute of Technology where his doctoral work involved studies of protein folding and assembly. He received a B.S. from Harvard College. We believe Dr. Gordon’s qualifications to sit on our board of directors include his extensive experience with biopharmaceutical and biotechnology companies, his financial expertise and his years of experience providing strategic advisory services to diverse companies.
John Longenecker, Ph.D. has served as our director since July 2007. Dr. Longenecker currently serves as the president, chief executive officer and a director of HemaQuest Pharmaceuticals, Inc. From February 2002 to January 2009, Dr. Longenecker was the president and chief executive officer of Favrille, Inc. In 1992, Dr. Longenecker joined DepoTech as senior vice president of research, development and operations and then served as president and chief operating officer from February 1998 to March 1999. Under Dr. Longenecker’s leadership, DepoTech took its lead product, DepoCyt(e), from early pre-clinical research and development through to commercial launch. Following SkyePharma PLC’s acquisition of DepoTech in 1999, Dr. Longenecker served as president for the U.S. operations of SkyePharma, Inc. and as a member of the executive committee for SkyePharma PLC. From 1982 to 1992, Dr. Longenecker was at Scios Inc. (Cal Bio), a biotechnology company where he served as vice-president and director of development. Dr. Longenecker was also a director of a number of Cal Bio subsidiaries during this period including Meta Bio and Karo Bio. Dr. Longenecker holds a B.S. in chemistry from Purdue University and a Ph.D. in biochemistry from The Australian National University. He was a post doctoral fellow at Stanford University from 1980 to 1982. Dr. Longenecker’s experience as the chief executive officer of a public company, demonstrates his leadership capability and extensive knowledge of complex financial and operational issues that public companies face and a thorough understanding of our business and industry and business acumen to our board of directors. We believe Dr. Longenecker’s extensive experience in the pharmaceutical and biotechnology industries provides valuable background and insight to our board of directors.
Gary Pace, Ph.D. has served as our director since June 2008. He is currently founder and chairman of the privately held Sova Pharmaceuticals Inc., founded in 2010, founder, director and consultant to QRxPharma Ltd. (ASX:QRX) founded in 2001, a Director of ResMed (NYSE:RMD) since 1994 and Transition Therapeutics Inc. (CDNX:TTH) since 2002. From 2002 to 2007, Dr. Pace was founder, chairman and chief executive officer of QRxPharma Ltd. and from 1995 to 2001, he was president and chief executive officer of RTP Pharma and from 2000 to 2002, Dr. Pace was chairman and chief executive officer of Waratah Pharmaceuticals Inc., a spin-off company from RTP Pharma. From 1993 to 1994, he was the founding president and chief executive officer of Transcend Therapeutics Inc. (formerly Free Radical Sciences Inc.), a biopharmaceutical company. From 1989 to 1993, he was senior vice president of Clintec International, Inc., a Baxter/Nestle joint venture and manufacturer of clinical nutritional products. Dr. Pace holds a B.S. with honors from the University of New South Wales and a Ph.D. from Massachusetts Institute of Technology. We believe Dr. Pace’s qualifications to sit on our board of directors include his financial expertise and his years of experience providing strategic advisory services to complex organizations, including as a public company director.
Andreas Wicki, Ph.D. has served as our director since our inception in December 2006. Dr. Wicki is a life sciences entrepreneur and investor with over 16 years of experience in the pharmaceutical and biotechnology industries. Dr. Wicki has been chief executive officer of HBM Partners AG and HBM BioVentures AG since 2001. From 1998 to 2001, Dr. Wicki was the senior vice president of the European Analytical Operations at MDS Inc. From 1990 to 1998, he was co-owner and chief executive officer of ANAWA Laboratorien AG and Clinserve AG, two life sciences contract research companies. Dr. Wicki holds an M.Sc. and Ph.D. in chemistry and biochemistry from the University of Bern, Switzerland. He currently serves on the board of directors of Buchler GmbH, HBM BioPharma India Ltd., HBM BioVentures (Cayman) Ltd., HBM Partners Ltd. and PharmaSwiss SA. We believe Dr. Wicki’s qualifications to sit on our board of directors include his extensive experience with pharmaceutical companies, his financial expertise and his years of experience providing strategic and advisory services to pharmaceutical and biotechnology organizations.
82
Table of Contents
Family Relationships
There are no family relationships among any of our directors or executive officers.
Code of Business Conduct and Ethics
We have adopted a written code of business conduct and ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. A current copy of the code is posted on the Corporate Governance section of our website, which is located at www.pacira.com. If we make any substantive amendments to, or grant any waivers from, the code of business conduct and ethics for any officer or director, we will disclose the nature of such amendment or waiver on our website or in a current report on Form 8-K.
Board Composition
Our board of directors currently consists of seven members, six of whom are non-employee members. Each director holds office until his or her successor is duly elected and qualified or until his or her death, resignation or removal.
In accordance with the terms of our restated certificate of incorporation and bylaws, our board of directors will be divided into three classes, class I, class II and class III, with each class serving staggered three-year terms. Upon the expiration of the term of a class of directors, directors in that class will be eligible to be elected for a new three-year term at the annual meeting of stockholders in the year in which their term expires. The members of the classes are divided as follows:
| • | Class I: Luke Evnin and Carl Gordon, and their term expires at the annual meeting of stockholders to be held in 2012 |
| • | Class II: John Longenecker and Andreas Wicki, and their term expires at the annual meeting of stockholders to be held in 2013 |
| • | Class II: Fred Middleton, Gary Pace and David Stack, and their term expires at the annual meeting of stockholders to be held in 2014 |
Our restated certificate of incorporation and amended and restated bylaws provide that the authorized number of directors may be changed only by resolution of the board of directors. Our restated certificate of incorporation and amended and restated bylaws also provide that our directors may be removed only for cause by the affirmative vote of the holders of at least 75% of the votes that all our stockholders would be entitled to cast in an annual election of directors, and that any vacancy on our board of directors, including a vacancy resulting from an enlargement of our board of directors, may be filled only by vote of a majority of our directors then in office.
We have no formal policy regarding board diversity. Our priority in selection of board members is identification of members who will further the interests of our stockholders through his or her established record of professional accomplishment, the ability to contribute positively to the collaborative culture among board members, knowledge of our business and understanding of the competitive landscape.
Procedures for Nominations to the Registrant’s Board of Directors
No changes have been made to the procedures by which security holders may recommend nominees to our board of directors.
Board Committees
Our board of directors has established an audit committee, a governance and nominating committee and a compensation committee. Each committee operates under a charter that has been approved by our board.
83
Table of Contents
Audit Committee. The members of our audit committee are John Longenecker, Gary Pace and Carl Gordon, and Dr. Gordon is chair the audit committee. Our board of directors has determined that Dr. Longenecker and Dr. Pace, two of the three directors serving on our audit committee, are independent within the meaning of The NASDAQ Marketplace Rules and Rule 10A-3 under the Securities Exchange Act of 1934, as amended, or the Exchange Act. In addition, our board of directors has determined that Dr. Gordon qualifies as an audit committee financial expert within the meaning of SEC regulations and The NASDAQ Marketplace Rules. In making this determination, our board has considered the formal education and nature and scope of his previous experience, coupled with past and present service on various audit committees. Our audit committee assists our board of directors in its oversight of our accounting and financial reporting process and the audits of our financial statements. Our audit committee’s responsibilities include:
| • | appointing, evaluating, retaining and, when necessary, terminating the engagement of our independent registered public accounting firm; |
| • | overseeing the independence of our independent registered public accounting firm, including obtaining and reviewing reports from the firm; |
| • | setting the compensation of our independent registered public accounting firm; |
| • | overseeing the work of our independent registered public accounting firm, including receiving and considering reports made by our independent registered public accounting firm regarding accounting policies and procedures, financial reporting and disclosure controls; |
| • | reviewing and discussing with management and our independent registered public accounting firm our audited financial statements and related disclosures; |
| • | preparing the annual audit committee report required by SEC rules; |
| • | coordinating internal control over financial reporting, disclosure controls and procedures and code of conduct; |
| • | reviewing our policies with respect to risk assessment and risk management; |
| • | establishing procedures related to the receipt, retention and treatment of complaints regarding accounting, internal accounting controls or auditing matters, and the confidential, anonymous submission by employees of concerns regarding accounting or auditing matters; |
| • | reviewing our policies and procedures for reviewing and approving or ratifying related person transactions, including our related person transaction policy; and |
| • | meeting independently with management and our independent registered public accounting firm. |
All audit services to be provided to us and all non-audit services, other than de minimis non-audit services, to be provided to us by our independent registered public accounting firm must be approved in advance by our audit committee.
Compensation Committee. The members of our compensation committee are Luke Evnin, John Longenecker and Fred Middleton, and Dr. Longenecker is the chair of the compensation committee. Our compensation committee assists our board of directors in the discharge of its responsibilities relating to the compensation of our executive officers. Our compensation committee’s responsibilities include:
| • | reviewing and recommending to the board of directors our chief executive officer’s compensation, and approving the compensation of our other executive officers reporting directly to our chief executive officer; |
| • | overseeing the evaluation of our senior executives; |
| • | overseeing, administering, reviewing and making recommendations to the board of directors with respect to our incentive compensation and equity-based plans; |
84
Table of Contents
| • | reviewing and making recommendations to the board of directors with respect to director compensation; |
| • | reviewing and discussing with management the compensation discussion and analysis required by SEC rules; and |
| • | preparing the annual compensation committee report required by SEC rules. |
Nominating and Corporate Governance Committee. The members of our nominating and corporate governance committee are John Longenecker and Gary Pace, and Dr. Pace is the chair of the nominating and corporate governance committee. The nominating and corporate governance committee’s responsibilities include:
| • | recommending to the board of directors the persons to be nominated for election as directors or to fill any vacancies on the board of directors, and to be appointed to each of the board’s committees; |
| • | developing and recommending to the board of directors corporate governance guidelines; and |
| • | overseeing an annual self-evaluation of the board of directors. |
Section 16(a) Beneficial Ownership Reporting Compliance
Our directors, executive officers and beneficial owners of more than 10% of our common stock are required under Section 16(a) of the Securities Exchange Act of 1934, as amended, to file reports of ownership and changes in ownership of our securities with the SEC. We completed the initial public offering of our common stock on February 8, 2011, and accordingly, we did not have a class of securities registered pursuant to Section 12 of the Exchange Act in 2010.
| Item 11. | Executive Compensation |
Compensation Discussion and Analysis
This section discusses the material elements of our executive compensation policies and decisions and the most important factors relevant to an analysis of these policies and decisions. It provides qualitative information regarding the manner and context in which compensation is awarded to and earned by our executive officers named in the “Summary Compensation Table,” or our “named executive officers,” and is intended to place in perspective the data presented in the tables and the narrative that follows.
In preparing to become a public company, we conducted a thorough review of all elements of our executive compensation program, including the function and design of our equity incentive programs. We have begun, and we expect to continue in the coming months, to evaluate the need for revisions to our executive compensation program to ensure our program is competitive with the companies with which we compete for executive talent and is appropriate for a public company.
Overview of our Executive Compensation Process
Roles of Our Board, Chief Executive Officer and Compensation Committee in Compensation Decisions. As a private company, our chief executive officer and compensation committee have historically overseen our executive compensation program. Our compensation committee, either as a committee or together with the other independent directors, makes all compensation decisions regarding our chief executive officer. Our chief executive officer may make recommendations to the compensation committee regarding the compensation of our executive officers other than the chief executive officer, but the compensation committee either makes all compensation decisions regarding our other executive officers or makes recommendations concerning executive compensation to our board of directors, with the independent directors making such decisions. In his role, our chief executive officer has reviewed all compensation decisions relating to our executive officers other than himself. He has annually reviewed the performance of each of our other executive officers, and, based on these reviews, has made recommendations to our compensation committee regarding salary adjustments, annual incentive bonus payments and equity incentive awards for our executive officers.
85
Table of Contents
Competitive Market Data and Use of Compensation Consultants. Historically, we have not formally benchmarked our executive compensation against compensation data of a peer group of companies, but rather have relied on the business judgment and experience in the pharmaceutical industry of our chief executive officer and members of our compensation committee. We have developed substantial information about compensation practices and levels at comparable companies through extensive recruiting, networking and industry research. Our compensation committee may in the future elect to engage an independent compensation consulting firm to provide advice regarding our executive compensation program and general information regarding executive compensation practices in our industry. Although the compensation committee would consider such a compensation consulting firm’s advice in establishing and approving the various elements of our executive compensation program, our chief executive officer and the compensation committee would ultimately make their own decisions, or make recommendations to our board of directors, about these matters.
Objectives and Philosophy of Our Executive Compensation Program. Our primary objective with respect to executive compensation is to attract, retain and motivate highly talented individuals who have the skills and experience to successfully execute our business strategy. Our executive compensation program is designed to:
| • | reward the achievement of our annual and long-term operating and strategic goals; |
| • | recognize individual contributions; |
| • | align the interests of our executives with those of our stockholders by rewarding performance that meets or exceeds established goals, with the ultimate objective of increasing stockholder value; and |
| • | retain and build our executive management team. |
To achieve these objectives, our executive compensation program ties a portion of each executive’s overall compensation to key corporate financial goals and to individual goals. We have also provided a portion of our executive compensation in the form of option awards that vest over time, which we believe helps to retain our executive officers and aligns their interests with those of our stockholders by allowing them to participate in our long-term performance as reflected in the trading price of shares of our common stock.
Elements of Our Executive Compensation Program. The primary elements of our executive compensation program are:
| • | base salaries; |
| • | annual incentive bonuses; |
| • | company sale bonus plan; |
| • | equity incentive awards; and |
| • | other employee benefits. |
We have not adopted any formal or informal policies or guidelines for allocating compensation among these elements.
Base Salaries. We use competitive base salaries to attract and retain qualified candidates to help us achieve our growth and performance goals. Base salaries are intended to recognize an executive officer’s immediate contribution to our organization, as well as his or her experience, knowledge and responsibilities.
Historically, our chief executive officer (with respect to executive officers other than himself) and our vice president, human resources have annually evaluated and recommended adjustments to executive officer base salary levels to our compensation committee or board of directors based on factors determined to be relevant, including:
86
Table of Contents
| • | the executive officer’s skills and experience; |
| • | the particular importance of the executive officer’s position to us; |
| • | the executive officer’s individual performance; |
| • | the executive officer’s growth in his or her position; and |
| • | base salaries for comparable positions within our company and at other companies. |
Our chief executive officer’s base salary has been determined by the non-management members of our board of directors, taking into account these same factors.
We have historically made annual base salary adjustments at the end of each year, with the adjustments taking effect at the beginning of the following year. In 2010, we made no adjustments to the base salaries for our chief executive officer or any of our other named executive officers.
Our compensation committee performs such annual evaluations, and we expect that it will consider similar factors, as well as perhaps the input of a compensation consulting firm and peer group benchmarking data, in making any adjustments to executive officer base salary levels.
Annual Incentive Bonuses. In addition to the corporate goals described below, members of management, including each of our executive officers, were assigned personal achievement goals near the beginning of fiscal 2007. For our executive officers other than our chief executive officer, these individual goals were set by our chief executive officer in collaboration with our executive management team and the individual goals for our chief executive officer were set by our board of directors, taking into account discussions with our chief executive officer.
We do not currently have a formal annual incentive bonus program. The company did pay cash bonuses based on the achievement of approved operational milestones in 2007. The 2007 bonus program was targeted at 75% based on the achievement of corporate goals and 25% based on personal achievement goals. A total pool of $57,570 was shared equally between six executives. The compensation committee did not establish a formal annual incentive bonus program in 2009 or 2010 and we have not paid any bonuses based on corporate goals or personal achievement goals in 2009 or 2010. Although our 2009 and 2010 corporate goals were informal, they were focused on the achievement of certain objectives. In 2009, the objectives were (1) successful completion of additional Phase 3 clinical trials of EXPAREL and (2) obtaining additional financing. In 2010, the objectives were (1) filing our NDA for EXPAREL, (2) obtaining additional financing, (3) converting our current clinical manufacturing suite to a commercial manufacturing suite and (4) filing this registration statement. For 2009 and 2010, our compensation committee made the decision not to pay annual bonuses based on the need to manage expenses and allocate resources to our clinical development programs, and did not formally evaluate whether our 2009 or 2010 corporate goals had been achieved. We did not have additional individual performance goals for our executive officers in 2009 or 2010. Our compensation committee has the authority to award discretionary performance-based cash bonuses to our executive officers and certain non-executive employees. Our compensation committee considers awarding such discretionary bonuses in the event of extraordinary short-term efforts and achievements by our executives and employees, as recommended by management. No such discretionary bonuses were awarded in 2009 or 2010. We do expect that our compensation committee will establish a formal cash incentive program in the future, and that our executive officers will participate in that program.
Company Sale Bonus Plan. In March 2009, we adopted a company sale bonus plan, amended and restated in March 2010, that provided for a potential cash bonus payment to specified employees and consultants, including our executive officers, and our non-employee directors, in the event of a sale of our company. The purpose of the company sale bonus plan was to provide these employees, consultants and directors with an additional incentive in connection with a transaction that would have been in our and our stockholders’ best interests, but which may have otherwise created personal uncertainties. Under the company sale bonus plan, upon the closing of a sale transaction that satisfied specified criteria, each participant in the company sale plan would receive either a bonus in an amount equal to a portion of the sale proceeds multiplied by a specified percentage for that participant or a fixed bonus
87
Table of Contents
payment. As a condition to becoming participants under the plan, most of the participants, including all of our executive officers and non-employee directors, agreed to have their existing option grants cancelled in March 2009. The participants in the bonus plan were determined by our board of directors. This bonus plan terminated upon the completion of our initial public offering.
Equity Incentive Compensation. We believe that our long-term performance is enhanced through equity awards. Equity awards reward executives and employees for maximizing stockholder value over time and align the interests of our employees and management with those of the stockholders. We granted stock options to our employees, including our named executive officers, in connection with their initial employment with us. In connection with the adoption of our company sale bonus plan, most of the participants under the plan, including all of our executive officers and non-employee directors, agreed to have their existing option grants cancelled. Subsequent to the cancellation, in September 2010, our board of directors granted new options to all of our employees, including our executive officers, and our non-employee directors, including options to purchase an aggregate of 809,390 shares of common stock to our named executive officers. The following table sets forth the number of shares underlying stock options granted to our named executive officers in September 2010:
| Name |
Number of Shares of Common Stock Underlying Stock Option | |
| David Stack, Chief Executive Officer |
441,655 | |
| James Scibetta, Chief Financial Officer |
147,373 | |
| Gary Patou, Chief Medical Officer |
118,084 | |
| Mark Walters, Senior Vice President |
51,139 | |
| William Lambert, Senior Vice President |
51,139 |
In December 2010, our board of directors granted options to all of our employees, including our named executive officers and our non-employee directors. Options to purchase an aggregate of 290,407 shares of common stock were granted to our named executive officers. The following table sets forth the number of shares underlying stock options granted to our named executive officers in December 2010:
| Name |
Number of Shares of Common Stock Underlying Stock Option | |
| David Stack, Chief Executive Officer |
158,466 | |
| James Scibetta, Chief Financial Officer |
52,877 | |
| Gary Patou, Chief Medical Officer |
42,368 | |
| Mark Walters, Senior Vice President |
18,348 | |
| William Lambert, Senior Vice President |
18,348 |
Equity Incentive Awards. Our equity incentive award program is the primary vehicle for offering long-term incentives to our executive officers. To date, equity incentive awards to our executive officers have been made in the form of stock options. We believe that equity incentive awards:
| • | provide our executive officers with a strong link to our long-term performance by enhancing their accountability for long-term decision making; |
| • | create an ownership culture by aligning the interests of our executive officers with the creation of value for our stockholders; and |
| • | further our goal of executive retention. |
88
Table of Contents
Employees who are considered important to our long-term success are eligible to receive equity incentive awards. Equity incentive awards have been granted to all of our current employees and certain of our non-employee directors. On September 2, 2010, we granted options to purchase an aggregate of 809,390 shares of common stock to our named executive officers. On December 29, 2010, we granted options to purchase an aggregate of 290,407 shares of common stock to our named executive officers.
Historically, all equity incentive awards granted to our executive officers have been approved by our board of directors, with input from our chief executive officer, our executive management team and our compensation committee. In determining the size of equity incentive awards to executive officers, our board and chief executive officer have generally considered the executive’s experience, skills, level and scope of responsibilities, existing equity holdings, and comparisons to comparable positions in our company.
Our compensation committee has the authority to make equity awards to our executive officers and to administer our equity incentive plans.
We do not have any equity ownership guidelines or requirements for our executive officers.
Other Employee Benefits. We maintain broad-based benefits that are provided to all employees, including our 401(k) retirement plan, flexible spending accounts, medical and dental care plans, life insurance, short- and long-term disability policies, vacation and company holidays. Our executive officers are eligible to participate in each of these programs on the same terms as non-executive employees; however, employees at the director level and above are eligible for life insurance coverage equal to three times (rather than twice) their annual base salary.
Severance and Change of Control Arrangements. We have entered into employment agreements with David Stack, our chief executive officer, James Scibetta, our chief financial officer, Gary Patou, our chief medical officer, Mark Walters, our senior vice president, technical operations and William Lambert, our senior vice president, pharmaceutical development. Each of these agreements provides the executive officer with certain severance benefits in connection with certain terminations of the executive’s employment or, in the case of Dr. Patou, consulting arrangement, both before and after a change of control of us. See “Executive Compensation—Employment Agreements, Severance and Change in Control Arrangements” below.
Risk Considerations in our Compensation Program. We have reviewed and evaluated the standards on which our compensation plans have been developed and implemented across our company. It is our belief that our compensation programs do not encourage inappropriate actions by our executive officers. Specifically, we believe that our compensation policies and practices avoid:
| • | a compensation mix overly weighted toward annual bonus awards; |
| • | an excessive focus on stock option awards that would cause behavior to drive short-term stock price gains in lieu of long-term value creation; and |
| • | unreasonable financial goals or thresholds that would encourage efforts to generate near-term revenue with an adverse impact on long-term success. |
We believe that our current business process and planning cycle fosters the following behaviors and controls that would mitigate the potential for adverse risk caused by the action of our executives.
| • | Annual review of corporate and individual objectives of the executive officers to align these goals with our annual operating and strategic plans and do not encourage unnecessary or excessive risk taking. |
| • | Incentive awards are based on a review of a variety of indicators, including both financial performance and strategic achievements, reducing the potential to concentrate on one indicator as the basis of an annual incentive award. |
89
Table of Contents
| • | The mixes between fixed and variable and cash and equity compensation are designed to encourage strategies and actions that are in our long-term best interests. |
| • | Discretionary authority by the compensation committee to adjust annual bonus funding and payments reduces business risk associated with our cash bonus program. |
| • | Stock option awards vest over a period of time. As a result of the longer time horizon to receive the value of a stock option award, the prospect of short-term or risky behavior is mitigated. |
As a result, we do not believe that any risks arising from our employee compensation policies and practices are reasonably likely to have a material adverse effect on us. In addition, we do not believe that the mix and design of the components of our executive compensation program encourage management to assume excessive risks.
Tax Considerations. Section 162(m) of the U.S. Internal Revenue Code of 1986, as amended, which we refer to as the Code, generally disallows a tax deduction for compensation in excess of $1.0 million paid by a public company to its chief executive officer and to each other officer (other than its chief financial officer) whose compensation is required to be reported to stockholders by reason of being among the three other most highly paid executive officers. Qualifying performance-based compensation is not subject to the deduction limitation if specified requirements are met. We will periodically review the potential consequences of Section 162(m) on the various elements of our executive compensation program, and we generally intend to structure the equity incentives component of our executive compensation program, where feasible, to comply with exemptions in Section 162(m) so that the compensation remains tax deductible to us. However, our board of directors or compensation committee may, in its judgment, authorize compensation payments that do not comply with the exemptions in Section 162(m) when it believes that such payments are appropriate to attract and retain executive talent.
Section 409A of the Code applies to plans, agreements and arrangements that provide for the deferral of compensation, and imposes penalty taxes on employees if those plans, agreements and arrangements do not comply with Section 409A. We have sought to structure our executive compensation arrangements to be exempt from, or comply with, Section 409A.
Summary Compensation Table
The following table sets forth information regarding compensation earned by our chief executive officer, our chief financial officer and each of our three other most highly compensated executive officers during our fiscal years ended December 31, 2009 and 2010. We refer to these individuals as our named executive officers.
| Name and Principal Position |
Year | Salary ($) |
Bonus(1) ($) |
Option Awards(2) ($) |
All
Other Compensation(3) ($) |
Total ($) |
||||||||||||||||||
| David Stack |
2010 | 400,000 | — | 1,112,323 | 1,504 | 1,513,827 | ||||||||||||||||||
| Chief Executive Officer |
2009 | 400,000 | — | — | 1,504 | 401,504 | ||||||||||||||||||
| James Scibetta |
2010 | 270,000 | — | 370,735 | 1,504 | 642,239 | ||||||||||||||||||
| Chief Financial Officer |
2009 | 270,000 | — | — | 1,504 | 271,504 | ||||||||||||||||||
| Gary Patou |
2010 | 336,660 | 300,000 | 295,018 | — | 931,678 | ||||||||||||||||||
| Chief Medical Officer(4) |
2009 | 280,550 | — | — | — | 280,550 | ||||||||||||||||||
| Mark Walters |
2010 | 250,000 | — | 127,595 | 1,600 | 379,195 | ||||||||||||||||||
| Senior Vice President |
2009 | 250,000 | — | — | 1,600 | 251,600 | ||||||||||||||||||
| William Lambert |
2010 | 220,000 | — | 127,595 | 1,487 | 349,082 | ||||||||||||||||||
| Senior Vice President |
2009 | 220,000 | — | — | 1,483 | 221,483 | ||||||||||||||||||
| (1) | Represents a bonus paid to Dr. Patou upon the successful completion of the NDA submission for EXPAREL pursuant to the Services Agreement with MPM Asset Management LLC, or MPM AM, and Dr. Patou. |
90
Table of Contents
| (2) | Represents the grant date fair value of option awards granted in 2010 in accordance with ASC 718. Our named executive officers will only realize compensation to the extent the fair value of our common stock is greater than the exercise price of such stock options. For information regarding assumptions underlying the valuation of equity awards, see note 11 to our financial statements included elsewhere in this annual report. |
| (3) | Amounts represent the value of perquisites and other personal benefits which are further detailed in the table below: |
| Name |
2009 Group Life Insurance ($) |
2010 Group Life Insurance ($) |
||||||
| David Stack |
1,504 | 1,504 | ||||||
| James Scibetta |
1,504 | 1,504 | ||||||
| Gary Patou |
— | — | ||||||
| Mark Walters |
1,600 | 1,600 | ||||||
| William Lambert |
1,483 | 1,487 | ||||||
| (4) | Dr. Patou, a managing director at MPM, is a consultant to us and provided the services customarily expected of a chief medical officer. Pursuant to the Services Agreement with MPM AM and Dr. Patou, we paid a service fee of $26,467 per month to MPM AM for the services provided by Dr. Patou and MPM AM. For more information, see “Executive Compensation—Services Agreement with MPM and Gary Patou.” |
Grants of Plan-Based Awards in 2010
The following table sets forth information for the year ended December 31, 2010 regarding grants of stock options made during 2010 to our named executive officers.
| Name |
Grant Date | All other Option Awards: Number of Securities Underlying Options (#) |
Exercise or Base Price of Option Awards ($/Sh) |
Grant Date Fair Value of Stock and Option Awards(1) |
||||||||||||
| David Stack |
9/02/10 | 441,655 | $ | 1.61 | $ | 495,195 | ||||||||||
| 12/29/10 | 158,466 | 5.49 | 617,128 | |||||||||||||
| James Scibetta |
9/02/10 | 147,373 | 1.61 | 164,809 | ||||||||||||
| 12/29/10 | 52,877 | 5.49 | 205,926 | |||||||||||||
| Gary Patou |
9/02/10 | 118,084 | 1.61 | 130,018 | ||||||||||||
| 12/29/10 | 42,368 | 5.49 | 165,001 | |||||||||||||
| Mark Walters |
9/02/10 | 51,139 | 1.61 | 56,138 | ||||||||||||
| 12/29/10 | 18,348 | 5.49 | 71,457 | |||||||||||||
| William Lambert |
9/02/10 | 51,139 | 1.61 | 56,138 | ||||||||||||
| 12/29/10 | 18,348 | 5.49 | 71,457 | |||||||||||||
| (1) | Represents the grant date fair value of option awards granted in 2010 in accordance with ASC 718. |
Outstanding Equity Awards at Year End
The following table sets forth certain information with respect to outstanding options held by our named executive officers at December 31, 2010.
91
Table of Contents
| Name |
Number of Securities Underlying Unexercised Options Exercisable |
Number of Securities Underlying Unexercised Options Unexercisable |
Option Exercise Price ($) |
Option Expiration Date |
||||||||||||
| David Stack |
104,602 | 81,358 | (1) | $ | 1.61 | 9/2/2020 | ||||||||||
| — | 255,695 | (2) | 1.61 | 9/2/2020 | ||||||||||||
| — | 158,466 | (3) | 5.49 | 12/29/2020 | ||||||||||||
| James Scibetta |
41,841 | 32,543 | (1) | 1.61 | 9/2/2020 | |||||||||||
| — | 72,989 | (2) | 1.61 | 9/2/2020 | ||||||||||||
| — | 52,877 | (3) | 5.49 | 12/29/2020 | ||||||||||||
| Gary Patou |
33,211 | 25,831 | (1) | 1.61 | 9/2/2020 | |||||||||||
| — | 59,042 | (2) | 1.61 | 9/2/2020 | ||||||||||||
| — | 42,368 | (3) | 5.49 | 12/29/2020 | ||||||||||||
| Mark Walters |
30,218 | 6,974 | (4) | 1.61 | 9/2/2020 | |||||||||||
| — | 13,947 | (2) | 1.61 | 9/2/2020 | ||||||||||||
| — | 18,348 | (3) | 5.49 | 12/29/2020 | ||||||||||||
| William Lambert |
30,218 | 6,974 | (4) | 1.61 | 9/2/2020 | |||||||||||
| — | 13,947 | (2) | 1.61 | 9/2/2020 | ||||||||||||
| — | 18,348 | (3) | 5.49 | 12/29/2020 | ||||||||||||
| (1) | This option vested with respect to 50% of the shares subject to the option on September 2, 2010 and with respect to the remaining shares in approximately equal successive monthly installments over the next 24 months provided that the named executive officer continues to provide services to us over such period. |
| (2) | This option vests with respect to 25% of the shares subject to the option on September 2, 2011 and will vest in approximately equal successive monthly installments over the next 36 months provided that the named executive officer continues to provide services to us over such period. |
| (3) | This option vests with respect to 25% of the shares subject to the option on December 29, 2011 and will vest in approximately equal successive monthly installments over the next 36 months provided that the named executive officer continues to provide services to us over such period. |
| (4) | This option vested with respect to 75% of the shares subject to the option on September 2, 2010 and with respect to the remaining shares in approximately equal successive monthly installments over the next 12 months provided that the named executive officer continues to provide services to us over such period. |
Option Exercises and Stock Vested
None of our named executive officers exercised any options during the year ended December 31, 2010.
Potential Payments Upon Termination or Change of Control
The tables below summarize the potential payments to each of our named executive officers if he were to be terminated without cause or resigned for good reason on December 31, 2010, the last business day of the fiscal year ended December 31, 2010, under the following circumstances.
92
Table of Contents
| Not in Connection with a Change of Control | ||||||||||||||||
| Name |
Cash Severance Payments ($) |
Value of Continuation of Benefits ($) |
Value of Stock Vesting Upon Termination ($)(1) |
Total ($) | ||||||||||||
| David Stack |
400,000 | 9,305 | 894,375 | 1,303,680 | ||||||||||||
| James Scibetta |
202,500 | 6,979 | 286,650 | 496,129 | ||||||||||||
| Gary Patou |
238,206 | (2) | — | 228,600 | (3) | 466,806 | ||||||||||
| Mark Walters |
187,500 | 4,637 | 160,875 | 353,012 | ||||||||||||
| William Lambert |
165,000 | 6,979 | 160,875 | 332,854 | ||||||||||||
| (1) | This amount is equal to (i) the number of option shares that would vest as a direct result of the employment termination without cause or for good reason, assuming a December 31, 2010 employment termination, multiplied by (ii) the excess of fair market value of our common stock as of December 31, 2010, over the exercise price of the option. For a discussion of our methodology for determining the fair market value of our common stock, see the “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Critical Accounting Policies and Use of Estimates—Share-Based Compensation.” |
| (2) | Pursuant to the Services Agreement with MPM AM and Dr. Patou, we are required to make certain payments to MPM in the case of a termination of the agreement. For more information, see “Executive Compensation—Services Agreement with MPM and Gary Patou.” |
| (3) | Pursuant to the Services Agreement with MPM AM and Dr. Patou, Dr. Patou is entitled to accelerated vesting of his options in the case of a termination of the agreement. For more information, see “Executive Compensation—Services Agreement with MPM and Gary Patou.” |
| 30 days Prior to, or One Year After, a Change of Control |
||||||||||||||||
| Name |
Cash Severance Payments ($) |
Value of Continuation of Benefits ($) |
Value of Stock Vesting Upon Termination ($)(1) |
Total ($) | ||||||||||||
| David Stack |
400,000 | 9,305 | 1,710,000 | 2,119,305 | ||||||||||||
| James Scibetta |
202,500 | 6,979 | 570,600 | 780,079 | ||||||||||||
| Gary Patou |
238,206 | (2) | — | 457,200 | (3) | 695,406 | ||||||||||
| Mark Walters |
187,500 | 4,637 | 198,000 | 390,137 | ||||||||||||
| William Lambert |
165,000 | 6,979 | 198,000 | 369,979 | ||||||||||||
| (1) | This amount is equal to (i) the number of option shares that would vest as a direct result of the employment termination without cause or for good reason in connection with a change in control, assuming a December 31, 2010 employment termination, multiplied by (ii) the excess of fair market value of our common stock as of December 31, 2010, over the exercise price of the option. For a discussion of our methodology for determining the fair market value of our common stock, see the “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Critical Accounting Policies and Use of Estimates—Share-Based Compensation.” |
| (2) | Pursuant to the Services Agreement with MPM AM and Dr. Patou, we are required to make certain payments to MPM in the case of a termination of the agreement. For more information, see “Executive Compensation—Services Agreement with MPM and Gary Patou.” |
93
Table of Contents
| (3) | Pursuant to the Services Agreement with MPM AM and Dr. Patou, Dr. Patou is entitled to accelerated vesting of his options in the case of a termination of the agreement. For more information, see “Executive Compensation—Services Agreement with MPM and Gary Patou.” |
In addition, each of our named executive officers would be entitled to payments under our company sale bonus plan. See “Executive Compensation—Overview of our Executive Compensation Process—Company Sale Bonus Plan” below.
Employment Agreements, Severance and Change in Control Arrangements
We entered into employment agreements with each of our named executive officers other than Gary Patou. The agreements with each of our named executive officers provide for “at will” employment which means we or the executive can terminate his or her employment at any time, with or without cause. Pursuant to the agreements, each of our named executive officers will be entitled to a base salary and certain benefits as previously described.
If any of our named executive officers, other than our chief executive officer, (i) is terminated for any reason other than for “cause,” or (ii) terminates his or her employment for “good reason,” then such executive officer will be entitled to:
| • | earned and accrued base salary, bonus, vacation time and other benefits; |
| • | monthly salary continuation payments for a period of nine months from the effective date of the release required to be provided as a condition to receiving these payments; |
| • | health insurance coverage, subject to cost sharing, for nine months following the effective date of the release required to be provided as a condition to receiving this coverage; and |
| • | immediate vesting of the portion of the unvested options granted to him or her in connection with the agreement that would have become vested during the nine month period following the date of termination. |
If our chief executive officer (i) is terminated for any reason other than for “cause,” or (ii) terminates his employment for “good reason,” then he will be entitled to:
| • | earned and accrued base salary, bonus, vacation time and other benefits; |
| • | monthly salary continuation payments for a period of 12 months from the effective date of the release required to be provided as a condition to receiving these payments; |
| • | health insurance coverage, subject to cost sharing, for 12 months following the effective date of the release required to be provided as a condition to receiving this coverage; and |
| • | immediate vesting of the portion of the unvested options granted to him in connection with the agreement that would have become vested during the 12 month period. |
If, within 30 days prior to, or 12 months following, a “change in control,” any of our named executive officers, including our chief executive officer, (i) is terminated for any reason other than for “cause,” or (ii) terminates his or her employment during the agreement term for “good reason,” then, in addition to the severance payments described above, such executive officer will also be entitled to immediate vesting of the entire unvested portion of all equity compensation granted to him or her.
Our obligation to make the severance payments described above will be conditioned upon the executive officer’s continued compliance with the non-competition and confidentiality obligations set forth in his or her employment agreement and the executive officer’s execution of a general release of claims against us.
Under the employment agreements, “cause” means: (i) failure to substantially perform the duties owed to us after receiving written notice that sets forth in detail the specific respects in which our board of directors believes
94
Table of Contents
that the duties have not been substantially performed, and failure to correct the failure within 30 days after receiving a demand for substantial performance and opportunity to cure; (ii) fraud, misconduct, dishonesty, gross negligence or other acts either injurious to us or conducted with intentional disregard for our best interests; (iii) failure to follow reasonable and lawful instructions from our board of directors and failure to cure such failure after receiving 20 days advance written notice; (iv) material breach of the terms of the employment agreement or our employee proprietary information and inventions assignment agreement or any other similar agreement that may be in effect from time to time; or (v) conviction of, or pleading guilty or nolo contendere to, any misdemeanor involving dishonesty or moral turpitude or related to our business, or any felony.
Under the employment agreements, “good reason” means, without the executive officer’s prior written consent: (i) any material reduction of the executive officer’s then effective base salary that is not in accordance with his employment agreement or related to a cross-executive team salary reduction; (ii) any material breach by us of the executive officer’s employment agreement; or (iii) a material reduction in the executive officer’s responsibilities or duties, not including a mere reassignment following a change of control to a position that is substantially similar to the position held prior to the change of control; provided, however, that no such event or condition shall constitute good reason unless (x) the executive officer gives us a written notice of termination for good reason not more than 90 days after the initial existence of the condition, (y) the grounds for termination (if susceptible to correction) are not corrected by us within 30 days of our receipt of such notice and (z) the termination date occurs within one (1) year following our receipt of such notice.
Under the employment agreements, a “change of control” means (i) a merger or consolidation of either us or PPI-California into another entity in which the stockholders of us or PPI-California (as applicable) do not control 50% or more of the total voting power of the surviving entity (other than a reincorporation merger); (ii) the sale, transfer or other disposition of all or substantially all of our assets in a liquidation or dissolution; or (iii) the sale or transfer of more than 50% of our outstanding voting stock. In the case of each of the foregoing clauses (i), (ii) and (iii), a change of control as a result of a financing transaction entered into by us or PPI-California shall not constitute a change of control for purposes of these agreements.
Services Agreement with MPM and Gary Patou
In March 2009, we entered into a services agreement with Dr. Patou and MPM Asset Management LLC, or MPM AM. Pursuant to the services agreement, Dr. Gary Patou provided the services to us customarily expected of a chief medical officer. Mr. Patou’s principal duties were to manage and lead our clinical team as well as oversee development of protocols and clinical trials designed to provide a path for regulatory approval of EXPAREL. In March 2010, we amended and restated the services agreement to, among other things, extend the term of the services until the deadline for filing the NDA for EXPAREL to October 15, 2010 or until either party gives 10 days prior written notice. In consideration of the services performed under the services agreement, we paid a service fee of $26,467 per month to MPM AM. In addition, we paid a bonus to Dr. Patou upon the successful completion of an NDA submission for EXPAREL.
In October 2010, we entered into a new services agreement with Dr. Patou and MPM AM. Pursuant to this services agreement, Dr. Gary Patou continues to provide the services to us customarily expected of a chief medical officer. Dr. Patou’s principal duties include obtaining approval for the EXPAREL NDA in the United States, filing the EXPAREL dossier in the European Union, developing additional clinical indications for EXPAREL and assisting with our product pipeline development. Under the new services agreement, we pay a service fee of $26,467 per month to MPM AM which is adjusted based on the total amount of time Dr. Patou devotes to us during the term of the services agreement. If we terminate our consulting relationship with Dr. Patou and MPM AM other than for “cause” or the consulting relationship is terminated by Dr. Patou and MPM AM for “good reason”, then MPM AM will be entitled to continuation of the then effective monthly service fee for a period of nine months following the date of termination and Dr. Patou will be entitled to immediate vesting of the portion of the unvested options that would have vested during the nine month period following the date of termination, provided that the options granted to Dr. Patou in December 2010 are subject to additional vesting. In addition, if within 30 days prior to, or 12 months following, a “change of control,” the consulting relationship is terminated other than for “cause” or for “good reason”, then in addition to the service payments above, Dr. Patou will also be entitled to immediate vesting of the entire unvested portion of his stock options.
95
Table of Contents
Director Compensation
Non-Employee Director Compensation Policy
Our board of directors has approved a compensation policy for our non-employee directors that became effective upon the completion of our initial public offering. This policy provides for the following compensation to our non-employee directors following the completion of our initial public offering:
| • | each non-employee director is entitled to receive an annual fee from us of $35,000 and an additional $25,000 fee if the non-employee director is the chairman of our board of directors; |
| • | the chair of our audit committee will receive an annual fee from us of $15,000 and other members of our audit committee will receive $7,500; |
| • | the chair of our compensation committee will receive an annual fee from us of $15,000 and other members of our compensation committee will receive $7,500; |
| • | the chair of our nominating and corporate governance committee will receive an annual fee from us of $10,000 and other members will receive $5,000; and |
| • | each non-employee director will be entitled to an annual grant of options to purchase 2,325 shares of our common stock under our 2011 Stock Incentive Plan, or the 2011 Plan, or any other equity incentive plan we may adopt in the future. |
In addition, certain of our non-employee directors received option grants to purchase 6,043 shares of our common stock and certain of our non-employee directors received option grants to purchase 4,649 shares of our common stock, each of which began vesting upon the effective date of the registration statement for our initial public offering. Fifty percent of the shares underlying these options will vest on each anniversary of the completion of our initial public offering, such that all of the shares underlying such options will have vested on the second anniversary of the completion of our initial public offering. Upon a change in control of us, as defined in the 2007 Plan, 100% of the shares underlying these options shall become vested and exercisable immediately prior to such change in control.
Each non-employee director that joins our board of directors in the future will also receive an initial option grant to purchase 6,043 shares of our common stock. Fifty percent of the shares underlying each of these options will vest each year on the anniversary of the grant date, such that all of the shares underlying such options will have vested on the second anniversary of the grant date. Upon a change in control of us, as defined in the 2007 Plan, 100% of the shares underlying these options shall become vested and exercisable immediately prior to such change in control.
All fees under the director compensation policy will be paid on a rolling annual basis and no per meeting fees will be paid. We will also reimburse non-employee directors for reasonable expenses incurred in connection with attending board of director and committee meetings.
Director Compensation Table
The following table sets forth a summary of the compensation earned by our directors for the year ended December 31, 2010, with the exception of Mr. Stack, whose compensation is included in the “Summary Compensation Table” below.
| Name |
Option Awards(1) ($) |
Total ($) |
||||||
| Fred Middleton |
10,464 | 10,464 | ||||||
| Luke Evnin, Ph.D. |
10,464 | 10,464 | ||||||
| Carl Gordon, Ph.D. |
10,464 | 10,464 | ||||||
| John Longenecker, Ph.D. |
18,614 | 18,614 | ||||||
| Gary Pace, Ph.D. |
18,745 | 18,745 | ||||||
| Andreas Wicki, Ph.D. |
— | — | ||||||
| (1) | Represents the grant date fair value of option awards granted in 2010 in accordance with ASC Topic 718, or ASC 718, formerly Statement of Financial Accounting Standards No. 123(R). Our directors will only realize compensation to the extent the fair value of our common stock is greater than the exercise price of such stock options. For information regarding assumptions underlying the valuation of equity awards, see note 11 to our financial statements included elsewhere in this annual report. |
96
Table of Contents
Stock Option and Other Compensation Plans
2007 Stock Option/Stock Issuance Plan
In January 2007, our board of directors approved our 2007 Stock Option/Stock Issuance Plan, or the 2007 Plan. The 2007 Plan was approved by our stockholders in June 2007.
We initially reserved 650,860 shares of our common stock for issuance under the 2007 Plan. In April 2008, our board of directors amended the 2007 Plan to, among other things, increase the number of authorized plan shares from 650,860 to 1,066,946 shares of our common stock. This increase was approved by our stockholders in May 2008. In September 2010, our board of directors further amended the 2007 Plan to increase the number of authorized plan shares from 1,066,946 to 1,729,498 shares of our common stock. This increase was approved by our stockholders in October 2010. In December 2010, our board of directors further amended the 2007 Plan to increase the number of authorized plan shares from 1,729,498 to 2,546,657 shares of our common stock. This increase was approved by our stockholders in December 2010.
The material terms of the 2007 Plan are summarized below. The 2007 Plan was filed as an exhibit to the registration statement in connection with our initial public offering.
Administration. Our board of directors (or a committee of the board of directors) administers the 2007 Plan. Subject to the terms and conditions of the 2007 Plan, the administrator has the authority to select the persons to whom awards are to be made, to determine the type or types of awards to be granted to each person, determine the number of awards to grant, determine the number of shares to be subject to such awards, and the terms and conditions of such awards, and make all other determinations and decisions and to take all other actions necessary or advisable for the administration of the 2007 Plan. The plan administrator is also authorized to establish, adopt, amend or revise rules relating to administration of the 2007 Plan, subject to certain restrictions.
Eligibility. Options and restricted stock may be granted under the 2007 Plan to individuals who are then our employees, consultants or members of our board of directors or our subsidiaries. Only employees may be granted incentive stock options, or ISOs.
Awards. The 2007 Plan provides that our administrator may grant or issue stock options and restricted stock. The administrator considers each award grant subjectively, considering factors such as the individual performance of the recipient and the anticipated contribution of the recipient to the attainment of our long-term goals. Each award is set forth in a separate agreement with the person receiving the award and indicates the type, terms and conditions of the award.
| • | Non-qualified stock options, or NQSOs, provide for the right to purchase shares of our common stock at a specified price which may not be less than 85% of the fair market value of a share of stock on the date of grant, and usually will become exercisable (at the discretion of our compensation committee or the board of directors, in the case of awards to non-employee directors) in one or more installments after the grant date, subject to the participant’s continued employment or service with us and/or subject to the satisfaction of performance targets established by our compensation committee (or the board of directors, in the case of awards to non-employee directors). NQSOs may be granted for any term specified by our compensation committee (or the board of directors, in the case of awards to non-employee directors), but the term may not exceed ten years. |
| • | Incentive stock options, or ISOs, are designed to comply with the provisions of the Internal Revenue Code and are subject to specified restrictions contained in the Internal Revenue Code applicable to ISOs. Among such restrictions, ISOs must have an exercise price of not less than the fair market value of a share of common stock on the date of grant, may only be granted to employees, must expire within a specified period of time following the optionee’s termination of employment, and must be exercised within ten years after the date of grant. In the case of an ISO granted to an individual who owns (or is deemed to own) more than 10% of the total combined voting power of all classes of our capital stock on the date of grant, the 2007 Plan provides that the exercise price must be more than 110% of the fair market value of a share of common stock on the date of grant and the ISO must expire on the fifth anniversary of the date of its grant. |
97
Table of Contents
| • | Restricted stock may be granted to participants and made subject to such restrictions as may be determined by the administrator. Restricted stock may be repurchased by us at the original purchase price or, if no cash consideration was paid for such stock, forfeited for no consideration if the conditions or restrictions are not met, and the restricted stock may not be sold or otherwise transferred to third parties until restrictions are removed or expire. Recipients of restricted stock, unlike recipients of options, may have voting rights and may receive dividends, if any, prior to when the restrictions lapse. |
Corporate Transactions. In the event of a change of control where the acquiror does not assume awards granted under the 2007 Plan, awards issued under the 2007 Plan may be subject to accelerated vesting (at the discretion of the plan administrator) such that 100% of the awards will become vested and exercisable or payable, as applicable, immediately prior to a change in control. Under the 2007 Plan, a change of control is generally defined as:
| • | a merger, consolidation or other reorganization approved by our stockholders, unless securities representing more than 50% of the total combined voting power of the voting securities of the successor corporation are immediately thereafter beneficially owned, directly or indirectly and in substantially the same proportion, by the persons who beneficially owned our outstanding voting securities immediately prior to such transaction; |
| • | the acquisition, directly or indirectly by any person or related group of persons (other than us, our subsidiaries, or a person or entity that, prior to such transaction, directly or indirectly controls, is controlled by, or is under common control with, us), of beneficial ownership (within the meaning of Rule 13d-3 under the Exchange Act) of securities possessing more than 50% of the total combined voting power of our outstanding securities pursuant to a tender or exchange offer made directly to our stockholders; or |
| • | a stockholder-approved sale, transfer or other disposition of all or substantially all our assets in a complete liquidation or dissolution. |
Amendment of the 2007 Plan. Our board of directors may amend or modify the 2007 Plan in any and all respects. However, stockholder approval of any amendment to the 2007 Plan must be obtained to the extent necessary and desirable to comply with any applicable law, regulation or stock exchange rule, or for any amendment to the 2007 Plan that increases the number of shares available under the 2007 Plan. The administrator may, with the consent of the affected option holders, cancel any or all outstanding awards under the 2007 Plan and grant new awards in substitution. The 2007 Plan will terminate on the tenth anniversary of the date of its initial approval by our board of directors. All future awards will be granted pursuant to the terms of the 2011 stock incentive plan.
2011 Stock Incentive Plan
Our 2011 stock incentive plan, or the 2011 Plan was adopted by our board of directors and approved by our stockholders in December 2010. The 2011 Plan provides for the grant of incentive stock options, non-statutory stock options, restricted stock awards and other stock-based awards. Upon effectiveness, the sum of (up to 2,546,657 shares) (x) the number of shares of our common stock reserved for issuance under the 2007 Plan at such time, and (y) the number of shares of our common stock subject to awards granted under the 2007 Plan that expire, terminate or are otherwise surrendered, cancelled, forfeited or repurchased by us pursuant to a contractual repurchase right, will be reserved for issuance under the 2011 Plan. In addition, the 2011 Plan contains an “evergreen” provision, which allows for an increase in the number of shares available for issuance under the 2011 Plan on the first day of each calendar year from 2012 through 2015. The annual increase in the number of shares shall be equal to the lesser of:
| • | 557,880 shares of our common stock; |
| • | a number of shares equal to 3% of our outstanding shares as of such date; or |
98
Table of Contents
| • | an amount determined by our board of directors. |
Our employees, officers, directors, consultants and advisors are eligible to receive awards under our 2011 Plan. The 2011 Plan permits the grant of options, stock appreciation rights (SARs), restricted stock, restricted stock units and other stock-based awards. The exercise price of all stock options granted under the 2011 Plan cannot be less than 100% of the fair market value of the common stock on the date of grant. In general, stock options granted under the 2011 Plan will have a term of up to ten years. The measurement (base) price of SARs granted under the 2011 Plan cannot be less than 100% of the fair market value of the common stock on the date of grant. SARs will have a term of up to ten years.
The 2011 Plan is administered by the board of directors or another committee designated by the board of directors. Subject to limitations specified in the plan, the board or applicable committee to whom authority is delegated will select the recipients of awards and determine:
| • | the number of shares of common stock covered by options and the dates upon which the options become exercisable; |
| • | the exercise price of options; |
| • | the duration of the options; and |
| • | the number of shares of common stock subject to any SARs, restricted stock or other stock-based awards and the terms and conditions of such awards, including conditions for repurchase, issue price and repurchase price. |
Upon a merger or other reorganization event, our board of directors, may, in its sole discretion, take any one or more of the following actions pursuant to the 2011 Plan, as to some or all outstanding awards other than restricted stock awards:
| • | provide that all outstanding awards shall be assumed or substituted by the successor corporation; |
| • | upon written notice to a participant, provide that the participant’s unexercised options or awards will terminate immediately prior to the consummation of such transaction unless exercised by the participant; |
| • | provide that outstanding awards will become exercisable, realizable or deliverable, or restrictions applicable to an award will lapse, in whole or in part, prior to or upon the reorganization event; |
| • | in the event of a reorganization event pursuant to which holders of our common stock will receive a cash payment for each share surrendered in the reorganization event, make or provide for a cash payment to the participants equal to the excess, if any, of the acquisition price times the number of shares of our common stock subject to such outstanding awards (to the extent then exercisable (after giving effect to any acceleration of vesting) at prices not in excess of the acquisition price), over the aggregate exercise price of all such outstanding awards and any applicable tax withholdings, in exchange for the termination of such awards; and |
| • | provide that, in connection with a liquidation or dissolution, awards convert into the right to receive liquidation proceeds. |
Upon the occurrence of a reorganization event other than a liquidation or dissolution, the repurchase and other rights under each outstanding restricted stock award will continue for the benefit of the successor company and will, unless the board of directors may otherwise determine, apply to the cash, securities or other property into which our common stock is converted pursuant to the reorganization event. Upon the occurrence of a reorganization event involving a liquidation or dissolution, all conditions on each outstanding restricted stock award will automatically be deemed terminated or satisfied, unless otherwise provided in the agreement evidencing the restricted stock award.
99
Table of Contents
No award may be granted under the 2011 Plan after December 29, 2020. Our board of directors may amend, suspend or terminate the 2011 Plan at any time, subject to stockholder approval to the extent required by applicable law or stock market requirements.
401(k) Retirement Plan
We maintain a 401(k) retirement plan that is intended to be a tax-qualified defined contribution plan under Section 401(k) of the Internal Revenue Code. In general, all of our employees are eligible to participate, beginning on the first day of the month following commencement of their employment. The 401(k) plan includes a salary deferral arrangement pursuant to which participants may elect to reduce their current compensation by up to the statutorily prescribed limit, equal to $16,500 in 2010 and 2009, and have the amount of the reduction contributed to the 401(k) plan.
Compensation Committee Interlocks and Insider Participation
None of our executive officers serves, or in the past has served, as a member of the board of directors or compensation committee, or other committee serving an equivalent function, of any entity that has one or more executive officers who serve as members of our board of directors or our compensation committee. None of the members of our compensation committee is an officer or employee of our company. Other than John Longenecker, who was the president and chief operating officer of DepoTech, the predecessor to PPI-California, none of the members of our compensation committee have ever been an officer or employee of our company.
Compensation Committee Report
The Compensation Committee has reviewed and discussed the Compensation Discussion and Analysis required by Item 402(b) of Regulation S-K with our management. Based on this review and discussion, the Compensation Committee recommended to our board of directors that the Compensation Discussion and Analysis be included in this annual report.
By the Compensation Committee of the Board of Directors,
John Longenecker (chair)
Luke Evnin
Fred Middleton
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
Securities Authorized for Issuance Under Equity Compensation Plans
See “Securities Authorized for Issuance Under Equity Compensation Plans” in Item 5.
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth information regarding the beneficial ownership of our common stock as of February 28, 2011, by:
| • | each of our directors; |
| • | each of our named executive officers; |
| • | each person, or group of affiliated persons, who is known by us to beneficially own more than 5% of our common stock; and |
100
Table of Contents
| • | all of our directors and executive officers as a group. |
Beneficial ownership is determined in accordance with SEC rules. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities and include shares of common stock issuable upon the exercise of stock options and warrants that are immediately exercisable or exercisable within 60 days after February 28, 2011. Except as otherwise indicated, all of the shares reflected in the table are shares of common stock and all persons listed below have sole voting and investment power with respect to the shares beneficially owned by them, subject to applicable community property laws. The information is not necessarily indicative of beneficial ownership for any other purpose.
Percentage ownership calculations for beneficial ownership are based on 17,232,875 shares outstanding as of February 28, 2011. Except as otherwise indicated in the table below, addresses of named beneficial owners are in care of Pacira Pharmaceuticals, Inc., 5 Sylvan Way, Suite 125, Parsippany, New Jersey 07054.
In computing the number of shares of common stock beneficially owned by a person and the percentage ownership of that person, we deemed shares of common stock subject to options and warrants held by that person that are currently exercisable or exercisable within 60 days of February 28, 2011 to be outstanding. We did not deem these shares outstanding, however, for the purpose of computing the percentage ownership of any other person. Beneficial ownership representing less than 1% is denoted with an asterisk (*).
| Name and Address of Beneficial Owner |
Number of Shares Beneficially Owned |
Percentage of Total Voting Power(1) |
||||||
| 5% Stockholders |
||||||||
| HBM BioVentures (Cayman) Ltd.(2) |
3,216,630 | 18.6 | % | |||||
| MPM Capital and its affiliates(3) |
2,907,971 | 16.8 | % | |||||
| OrbiMed Advisors and its affiliates(4) |
2,907,972 | 16.8 | % | |||||
| Sanderling Ventures and its affiliates(5) |
3,000,951 | 17.3 | % | |||||
| Officers and Directors |
||||||||
| David Stack(6) |
143,695 | * | ||||||
| James Scibetta(7) |
53,039 | * | ||||||
| Gary Patou(8) |
38,131 | * | ||||||
| William Lambert(9) |
33,317 | * | ||||||
| Mark Walters(10) |
33,317 | * | ||||||
| Luke Evnin(11) |
2,907,971 | 16.8 | % | |||||
| Carl Gordon(12) |
2,907,972 | 16.8 | % | |||||
| John Longenecker(13) |
6,663 | * | ||||||
| Fred Middleton(14) |
3,000,951 | 17.3 | % | |||||
| Gary Pace(15) |
4,803 | * | ||||||
| Andreas Wicki(16) |
3,216,630 | 18.6 | % | |||||
| All current executive officers and directors as a group (11 persons) |
12,346,489 | 69.2 | % | |||||
| (1) | Percentage total voting power represents voting power with respect to all shares of our common stock on matters in which holders of our common stock are entitled to one vote per share. Each share of common stock has one vote per share. |
| (2) | The address for HBM BioVentures (Cayman) Ltd. is Centennial Towers, Suite 305, 2454 West Bay Road, Grand Cayman, Cayman Islands, B.V.I. Consists of (i) 3,137,597 shares of common stock held by HBM BioVentures (Cayman) Ltd., and (ii) 79,033 shares of common stock issuable upon exercise of warrants held by HBM BioVentures (Cayman) Ltd. The board of directors of HBM BioVentures (Cayman) Ltd. has sole voting and investment power with respect to the shares held by such entity and acts by majority vote. The board of directors of HBM BioVentures (Cayman) Ltd. is comprised of John Arnold, Richard Coles, Sophia Harris, Dr. Andreas Wicki and John Urquhart, none of whom has individual voting or investment power with respect to such shares. |
| (3) | The address for funds managed by MPM Capital is 200 Clarendon St., 54th Floor, Boston, MA 02116. Consists of (i) 2,651,400 shares of common stock held by MPM BioVentures IV-QP, L.P., (ii) 102,147 shares of common stock held by MPM BioVentures IV GmbH & Co. Beteiligungs KG, (iii) 75,394 shares of common stock held by MPM Asset Management Investors BV4 LLC, (iv) 74,072 shares of common stock issuable upon exercise of warrants held by MPM BioVentures IV-QP, L.P., (v) 2,852 shares of common stock issuable upon exercise of warrants held by MPM BioVentures IV GmbH & Co. Beteiligungs KG, (vi) 2,106 shares of common stock issuable upon exercise of warrants held by MPM Asset Management Investors BV4 LLC. Dr. Patou is a Managing Director of MPM Asset Management |
101
Table of Contents
| LLC. MPM Asset Management LLC is the Management Company of MPM BioVentures IV LLC. MPM BioVentures IV LLC is the Managing Member of MPM BioVentures IV GP LLC, which is the General Partner of MPM BioVentures IV-QP, LP. and the Managing Limited Partner of MPM BioVentures IV GmbH & Co. Beteiligungs KG. MPM BioVentures IV LLC is the Manager of MPM Asset Management Investors BV4 LLC. Dr. Evnin is a Member of MPM BioVentures IV LLC. Dr. Evnin has a shared power to vote, acquire, hold and dispose of all shares and warrants. Dr. Evnin disclaims beneficial ownership of the securities except to the extent of his pecuniary interest therein. |
| (4) | The address for funds managed by OrbiMed Advisors is 767 3rd Avenue, 30th Floor, New York, NY 10017. Consists of (i) 2,802,254 shares of common stock held by OrbiMed Private Investments III, LP, (ii) 26,687 shares of common stock held by OrbiMed Associates III, LP, (iii) 78,287 shares of common stock issuable upon exercise of warrants held by OrbiMed Private Investments III, LP, (iv) 744 shares of common stock issuable upon exercise of warrants held by OrbiMed Associates III, LP. OrbiMed Capital GP III LLC is the general partner of OrbiMed Private Investments III, LP and OrbiMed Advisors LLC is the managing member of OrbiMed Capital GP III LLC. OrbiMed Advisors LLC is also the general partner of OrbiMed Associates III, LP. Samuel D. Isaly is the managing member of and owner of a controlling interest in OrbiMed Advisors LLC and may be deemed to have voting and investment power over the shares held by OrbiMed Private Investments III, LP and OrbiMed Associates III, LP noted above. Mr. Isaly disclaims beneficial ownership of such shares, except to the extent of his pecuniary interest therein. Dr. Gordon, a member of our board of directors, is an affiliate of the above-mentioned funds. |
| (5) | The address for funds managed by Sanderling Ventures is 400 South El Camino Real, Suite 1200, San Mateo, California 94402. Consists of (i) 1,382,562 shares of common stock held by Sanderling Venture Partners VI, L.P., (ii) 47,754 shares of common stock held by Sanderling VI Beteiligungs GmbH & Co. KG, (iii) 56,896 shares of common stock held by Sanderling VI Limited Partnership, (iv) 1,336,113 shares of common stock held by Sanderling Venture Partners VI Co-Investment Fund, L.P., (v) 98,596 shares of common stock held by Sanderling Ventures Management VI, (vi) 38,193 shares of common stock issuable upon exercise of warrants held by Sanderling Venture Partners VI, L.P., (vii) 1,337 shares of common stock issuable upon exercise of warrants held by Sanderling VI Beteiligungs GmbH & Co. KG, (viii) 1,592 shares of common stock issuable upon exercise of warrants held by Sanderling VI Limited Partnership, (ix) 37,908 shares of common stock issuable upon exercise of warrants held by Sanderling Venture Partners VI Co-Investment Fund, L.P. Mr. Middleton is a managing director of Middleton, McNeil, Mills & Associates VI, LLC, which has the ultimate voting and investment power over shares held of record by Sanderling Venture Partners VI, L.P., Sanderling VI Beteiligungs GmbH & Co. KG, Sanderling VI Limited Partnership and Sanderling Venture Partners VI Co-Investment Fund, L.P. and he may be deemed to have voting and investment power over shares held of record by Sanderling Venture Partners VI, L.P., Sanderling VI Beteiligungs GmbH & Co. KG, Sanderling VI Limited Partnership and Sanderling Venture Partners VI Co-Investment Fund, L.P. Mr. Middleton is the owner of Sanderling Ventures Management VI and he may be deemed to have voting and investment power over shares held of record by Sanderling Ventures Management VI. Mr. Middleton disclaims beneficial ownership over the shares held by Sanderling Ventures and its affiliates, except to the extent of his pecuniary interest therein. |
| (6) | Consists of (i) 5,000 shares of common stock held by Mr. Stack, (ii) 18,596 shares of common stock held by Stack Schroon Mohawk FLP and (iii) 120,099 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. Mr. Stack is the general partner of Stack Schroon Mohawk FLP. |
| (7) | Consists of (i) 5,000 shares of common stock held by Mr. Scibetta and (ii) 48,039 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. |
| (8) | Consists of 38,131 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. |
| (9) | Includes 33,317 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. |
| (10) | Includes 33,317 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. |
| (11) | The address for funds managed by OrbiMed Advisors is 767 3rd Avenue, 30th Floor, New York, NY 10017. Consists of (i) 2,802,254 shares of common stock held by OrbiMed Private Investments III, LP, (ii) 26,687 shares of common stock held by OrbiMed Associates III, LP, (iii) 78,287 shares of common stock issuable upon exercise of warrants held by OrbiMed Private Investments III, LP, (iv) 744 shares of common stock issuable upon exercise of warrants held by OrbiMed Associates III, LP. OrbiMed Capital GP III LLC is the general partner of OrbiMed Private Investments III, LP and OrbiMed Advisors LLC is the managing member of OrbiMed Capital GP III LLC. OrbiMed Advisors LLC is also the general partner of OrbiMed Associates III, LP. Samuel D. Isaly is the managing member of and owner of a controlling interest in OrbiMed Advisors LLC and may be deemed to have voting and investment power over the shares held by OrbiMed Private Investments III, LP and OrbiMed Associates III, LP noted above. Mr. Isaly disclaims beneficial ownership of such shares, except to the extent of his pecuniary interest therein. Dr. Gordon, a member of our board of directors, is an affiliate of the above-mentioned funds. |
| (12) | Consists of the shares described in Note (4) above. Dr. Gordon disclaims beneficial ownership of the shares described in Note (3), except to the extent of his pecuniary interest therein. |
| (13) | Consists of 6,663 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. |
| (14) | The address for funds managed by Sanderling Ventures is 400 South El Camino Real, Suite 1200, San Mateo, California 94402. Consists of (i) 1,382,562 shares of common stock held by Sanderling Venture Partners VI, L.P., (ii) 47,754 shares of common stock held by Sanderling VI Beteiligungs GmbH & Co. KG, (iii) 56,896 shares of common stock held by Sanderling VI Limited Partnership, (iv) 1,336,113 shares of common stock held by Sanderling Venture Partners VI Co-Investment Fund, L.P., (v) 98,596 shares of common stock held by Sanderling Ventures Management VI, (vi) 38,193 shares of common stock issuable upon exercise of warrants held by Sanderling |
102
Table of Contents
| Venture Partners VI, L.P., (vii) 1,337 shares of common stock issuable upon exercise of warrants held by Sanderling VI Beteiligungs GmbH & Co. KG, (viii) 1,592 shares of common stock issuable upon exercise of warrants held by Sanderling VI Limited Partnership, (ix) 37,908 shares of common stock issuable upon exercise of warrants held by Sanderling Venture Partners VI Co-Investment Fund, L.P. Mr. Middleton is a managing director of Middleton, McNeil, Mills & Associates VI, LLC, which has the ultimate voting and investment power over shares held of record by Sanderling Venture Partners VI, L.P., Sanderling VI Beteiligungs GmbH & Co. KG, Sanderling VI Limited Partnership and Sanderling Venture Partners VI Co-Investment Fund, L.P. and he may be deemed to have voting and investment power over shares held of record by Sanderling Venture Partners VI, L.P., Sanderling VI Beteiligungs GmbH & Co. KG, Sanderling VI Limited Partnership and Sanderling Venture Partners VI Co-Investment Fund, L.P. Mr. Middleton is the owner of Sanderling Ventures Management VI and he may be deemed to have voting and investment power over shares held of record by Sanderling Ventures Management VI. Mr. Middleton disclaims beneficial ownership over the shares held by Sanderling Ventures and its affiliates, except to the extent of his pecuniary interest therein. |
| (15) | Consists of 4,803 shares of common stock issuable upon exercise of stock options within 60 days of February 28, 2011, including options that became exercisable upon completion of our initial public offering. |
| (16) | The address for HBM BioVentures (Cayman) Ltd. is Centennial Towers, Suite 305, 2454 West Bay Road, Grand Cayman, Cayman Islands, B.V.I. Consists of (i) 3,137,597 shares of common stock held by HBM BioVentures (Cayman) Ltd., and (ii) 79,033 shares of common stock issuable upon exercise of warrants held by HBM BioVentures (Cayman) Ltd. The board of directors of HBM BioVentures (Cayman) Ltd. has sole voting and investment power with respect to the shares held by such entity and acts by majority vote. The board of directors of HBM BioVentures (Cayman) Ltd. is comprised of John Arnold, Richard Coles, Sophia Harris, Dr. Andreas Wicki and John Urquhart, none of whom has individual voting or investment power with respect to such shares. |
| Item 13. | Certain Relationships and Related Transactions, and Director Independence |
The following is a description of transactions since January 1, 2010 to which we have been a party, in which the amount involved in the transaction exceeds $120,000, and in which any of our directors, executive officers or beneficial owners of more than 5% of our voting securities, or affiliates or immediate family members of any of our directors, executive officers or beneficial owners of more than 5% of our voting securities, had or will have a direct or indirect material interest. We believe the terms obtained or consideration that we paid or received, as applicable, in connection with the transactions described below were comparable to terms available or the amounts that would be paid or received, as applicable, from unrelated third parties.
Debt Financings
2010 Secured Debt Financing
In March 2010, we entered into an agreement with certain of our existing investors to issue $15.0 million in aggregate principal amount of secured notes, or the 2010 Secured Notes, in a private placement and the investors purchased the entire $15.0 million of 2010 Secured Notes. To secure the performance of our obligations under the purchase agreement for the 2010 Secured Notes, we granted a subordinated security interest in substantially all of our assets, including our intellectual property assets, to the investors. In connection with entering into the Hercules Credit Facility, the holders of the 2010 Secured Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the 2010 Secured Notes were subordinated to the Hercules Credit Facility. The holders of the 2010 Secured Notes previously entered into a separate intercreditor agreement with the holders of certain other debt securities of the Company. The 2010 Secured Notes had an interest rate of 5% per year and all principal and accrued and unpaid interest on the 2010 Secured Notes was due on December 31, 2010. In connection with entering into the Hercules Credit Facility, the maturity date was further extended to the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated.
All principal and interest due under the 2010 Secured Notes was converted into an aggregate of 1,156,606 shares of our common stock upon completion of our initial public offering. Purchasers of the 2010 Secured Notes included certain holders of more than 5% of our capital stock, or entities affiliated with them. The following table sets forth the amount of notes purchased by each such holder and the date of purchase.
103
Table of Contents
| Date of Purchase |
Purchaser |
Aggregate Principal Amount of Notes Purchased on Such Date |
||||
| March 10, 2010 |
Entities affiliated with HBM BioVentures | $ | 1,875,000 | |||
| Entities affiliated with MPM Capital | 1,875,000 | |||||
| Entities affiliated with OrbiMed Advisors | 1,875,000 | |||||
| Entities affiliated with Sanderling Ventures | 1,875,000 | |||||
| June 30, 2010 |
Entities affiliated with HBM BioVentures | 937,500 | ||||
| Entities affiliated with MPM Capital | 937,500 | |||||
| Entities affiliated with OrbiMed Advisors | 937,500 | |||||
| Entities affiliated with Sanderling Ventures | 937,500 | |||||
| September 1, 2010 |
Entities affiliated with HBM BioVentures | 937,500 | ||||
| Entities affiliated with MPM Capital | 937,500 | |||||
| Entities affiliated with OrbiMed Advisors | 937,500 | |||||
| Entities affiliated with Sanderling Ventures | 937,500 | |||||
HBM Term Loan
On April 30, 2010, we entered into a subordinated secured note purchase agreement with entities affiliated with HBM BioVentures, or HBM, to issue $3.75 million in aggregate principal amount of secured notes, or the HBM Secured Notes, in a private placement. HBM purchased the entire $3.75 million of the HBM Secured Notes. To secure the performance of our obligations under the purchase agreement for the HBM Secured Notes, we granted a subordinated security interest in substantially all of our assets, including our intellectual property assets, other than the assets that secure our obligations under the Amended and Restated Royalty Interests Assignment Agreement. The HBM Secured Notes carry an interest rate of approximately 10% per year. In addition, the HBM Secured Notes require a final payment fee if they are prepaid prior to the maturity date. The maturity date of the HBM Secured Notes is the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. In connection with entering into the Hercules Credit Facility, the holders of the HBM Secured Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the HBM Secured Notes were subordinated to the Hercules Credit Facility.
All principal and interest due under the HBM Secured Notes was converted into an aggregate of 308,655 shares of our common stock upon completion of our initial public offering. Purchasers of the HBM Secured Notes included certain holders of more than 5% of our capital stock, or entities affiliated with them.
December 2010 Convertible Notes
On December 29, 2010, we sold $7.5 million in aggregate principal amount of convertible promissory notes, or the December 2010 Convertible Notes, in a private placement to certain of our existing investors. In connection with the issuance and sale of the December 2010 Convertible Notes, we issued warrants to the holders of the December 2010 Convertible Notes to purchase an aggregate of 167,361 shares of our common stock with an exercise price of $13.44 per share. The December 2010 Convertible Notes will had an interest rate of 5% per year from and after March 31, 2011 and all principal and accrued and unpaid interest on the December 2010 Convertible Notes is due and payable upon the earliest of: (1) a sale of us, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated.
All principal and interest due under the December 2010 Convertible Notes was converted into an aggregate of 1,071,428 shares of our common stock at a conversion price equal to $7.00 price per share, the price per share of our common stock sold in our initial public offering. Purchasers of the December 2010 Convertible Notes included certain holders of more than 5% of our capital stock, or entities affiliated with them.
The following table sets forth the aggregate principal amount of December 2010 Convertible Notes purchased by each such holder and the warrants received in connection with the purchase of the December 2010 Convertible Notes, assuming that a second closing is not consummated.
104
Table of Contents
| Purchaser |
Aggregate Principal Amount of Notes |
Number of Warrant Shares |
||||||
| HBM BioVentures |
$ | 1,875,000 | 41,841 | |||||
| Entities affiliated with MPM Capital |
$ | 1,875,000 | 41,840 | |||||
| Entities affiliated with OrbiMed Advisors |
$ | 1,875,000 | 41,840 | |||||
| Entities affiliated with Sanderling Ventures |
$ | 1,875,000 | 41,840 | |||||
Stockholder Guarantee under Hercules Credit Facility
On November 24, 2010, we entered into a $26.25 million credit facility with Hercules Technology Growth Capital, Inc. and Hercules Technology III, L.P., as lenders, or the Hercules Credit Facility. We borrowed under the Hercules Credit Facility an aggregate principal amount of $26.25 million.
The Hercules Credit Facility is guaranteed by MPM Capital, Sanderling Ventures and Orbimed Advisors, and entities affiliated with them, which are holders of more than 5% of our voting securities, on a several and not joint basis, which guarantee is limited to each such stockholder’s pro rata portion of the outstanding principal and accrued and unpaid interest under the Hercules Credit Facility, but in no event to exceed $11.25 million in the aggregate. The obligations of these stockholders under the guarantee is not triggered until the earlier to occur of (i) 30 days after written notice from the agent that our obligations under the Hercules Credit Facility have been accelerated, and (ii) the occurrence of a bankruptcy or insolvency event with respect to the borrower under the Hercules Credit Facility, us or any of the guarantors. The guarantee by these stockholders of the Hercules Credit Facility also includes covenants that require each such investor to maintain at all times unfunded commitments from its fund investors in an amount equal to at least one and one-half times the maximum amount that the investor may be obligated for under the stockholder guarantee, and also includes certain control requirements with respect to such stockholders. The guarantee by these stockholders of the Hercules Credit Facility replaced the guarantee under the GECC Credit Facility which was terminated in November 2010.
Registration Rights
MPM Capital, Sanderling Ventures and Orbimed Advisors, and entities affiliated with them, which are holders of more than 5% of our voting securities, have registration rights with respect to shares of common stock and shares of our common stock issuable upon exercise of outstanding warrants held by them pursuant to an investors’ rights agreement, or the Investors’ Rights Agreement. These stockholders have the right to require us to register these shares under the Securities Act, under specified circumstances as more fully described below. After registration pursuant to these rights, these shares will become freely tradable without restriction under the Securities Act. These registration rights will terminate upon the earlier of (i) the date that is five years following the completion of our initial public offering or, (ii) for any particular holder with registration rights, at such time following our initial public offering when all of our securities held by that stockholder may be sold pursuant to Rule 144 under the Securities Act.
Demand and Form S-3 Registration Rights. Subject to specified limitations, the holders of at least thirty percent of our previously issued Series A convertible preferred stock having registration rights may demand that we register all or a portion of their registrable shares under the Securities Act. We are not obligated to file a registration statement pursuant to this provision:
| • | until 180 days after the completion of our initial public offering; and |
| • | on more than three occasions. |
In addition, the holders of our registrable shares may demand that we register on Form S-3 all or a portion of the registrable shares held by them. We are not obligated to file a Form S-3 pursuant to this provision on more than two occasions in any 12-month period.
105
Table of Contents
Incidental Registration Rights. If at any time we propose to file a registration statement to register any of our securities under the Securities Act, either for our own account or for the account of any of our stockholders, the holders of our registrable shares are entitled to notice of registration and are entitled to include their shares of common stock in the registration.
Limitations and Expenses. In the event that any registration in which the holders of registrable shares participate pursuant to the Investors’ Rights Agreement is an underwritten public offering, the number of registrable shares to be included may, in specified circumstances, be limited due to market conditions. Pursuant to the Investors’ Rights Agreement, we are required to pay all registration expenses, including the fees and expenses of one counsel to represent the selling holders, other than any underwriting discounts, selling commissions and similar discounts relating to underwriters or commissions related to sales, related to any demand or incidental registration. We are also required to indemnify each participating holder with respect to each registration of registrable shares that is effected.
Employment Agreements
We entered into employment agreements with the following executive officers and key employees: David Stack, our chief executive officer, James Scibetta, our chief financial officer, Mark Walters, our senior vice president, technical operations, William Lambert, our senior vice president, pharmaceutical development. For further information, see “Executive Compensation—Employment Agreements, Severance and Change in Control Arrangements.”
Services Agreements
We entered into a services agreement with Gary Patou, our chief medical officer, and MPM AM. For further information, see “Executive Compensation—Services Agreement with MPM and Gary Patou.”
In addition to the amounts paid to Gary Patou, MPM AM provides clinical management and subscription services to us. During the period from January 1, 2009 to December 31, 2010, we paid an aggregate of $34,000 to MPM AM for these services.
In February 2008, we entered into a services agreement with Stack Pharmaceuticals, Inc., or SPI, an entity controlled by David Stack, our chief executive officer. Pursuant to the agreement, SPI provided us with the use of SPI’s office facilities which included the use of office space for our employees, office furnishings, phone system, internet connections, printers and other related office amenities such as conference rooms. The office facilities are located at 5 Sylvan Way, Parsippany, New Jersey. Pursuant to the agreement, we paid SPI amounts ranging from $10,500 to $18,250 per month during the term of the services agreement. The term of the agreement was one year and was renewable upon consent of both parties and the agreement may be cancelled with 60 days written notice by either party. In February 2009, we renewed the agreement on a month-to-month basis.
In August 2010, we entered into a new services agreement with SPI that replaced the agreement that we entered into in February 2008. Pursuant to the new agreement, SPI provides us with the use of SPI’s office facilities which includes the use of office space for our employees, office furnishings, phone system, internet connections, printers and other related office amenities such as conference rooms. In addition, SPI provides consulting services and commercial leadership related to EXPAREL regarding the development of strategic plans and analyses for the commercialization of EXPAREL, support in the development of documents, data and materials for investor and commercial partner presentations and documents, and commercial leadership in support of our website. SPI provides these services from time to time as we request from August 2010 through December 2010. We pay SPI $2,500 for each day of services provided by SPI up to a maximum of five days per week. We also reimburse SPI for travel expenses incurred by SPI personnel.
In addition, during 2008, 2009 and 2010, upon our request, SPI performed various projects, all of which have been completed by SPI. These projects included a business analysis and commercial recommendation for our DepoDur product, a market research project related to the development of a DepoMethotrexate product, market research and forecasting in support of clinical development of EXPAREL for the potential additional indications of nerve block and epidural administration and reimbursement for access to Datamonitor reports for commercial analysis and partnering discussions regarding EXPAREL.
106
Table of Contents
During the period from January 1, 2009, through December 31, 2010, we have paid SPI an aggregate of $534,000 for the above services provided by SPI.
In April 2010, we signed a statement of work for a feasibility study with Rhythm Pharmaceuticals, Inc. We earned contract revenue of approximately $290,000 from this statement of work during the period from April 2010 through December 31, 2010. MPM Capital and its affiliates are holders of more than 5% of our capital stock. We have been informed that MPM Capital and its affiliates are holders of more than 10% of the capital stock of Rhythm Pharmaceuticals, Inc. and a managing director of MPM Capital is a member of the board of directors of Rhythm Pharmaceuticals, Inc.
Indemnification Agreements
We have entered into indemnification agreements with each of our directors and certain of our officers. These agreements require us to indemnify the individuals to the fullest extent permitted under Delaware law against liabilities that may arise by reason of their service to us, and to advance expenses incurred as a result of any proceeding against them as to which they could be indemnified. We intend to enter into indemnification agreements with our future directors and executive officer.
Policies and Procedures for Related Person Transactions
Our board of directors has adopted a written related person transaction policy to set forth the policies and procedures for the review and approval or ratification of related person transactions. This policy covers any transaction, arrangement or relationship, or any series of similar transactions, arrangements or relationships in which we were or are to be a participant, the amount involved exceeds $120,000, and a related person had or will have a direct or indirect material interest, including, without limitation, purchases of goods or services by or from the related person or entities in which the related person has a material interest, indebtedness, guarantees of indebtedness, and employment by us of a related person.
Any related person transaction proposed to be entered into by us is be required to be reported to our chief financial officer and is reviewed and approved by the audit committee in accordance with the terms of the policy, prior to effectiveness or consummation of the transaction, whenever practicable. If our chief financial officer determines that advance approval of a related person transaction is not practicable under the circumstances, the audit committee will review and, in its discretion, may ratify the related person transaction at the next meeting of the audit committee, or at the next meeting following the date that the related person transaction comes to the attention of our chief financial officer. Our chief financial officer, however, may present a related person transaction arising in the time period between meetings of the audit committee to the chair of the audit committee, who will review and may approve the related person transaction, subject to ratification by the audit committee at the next meeting of the audit committee.
In addition, any related person transaction previously approved by the audit committee or otherwise already existing that is ongoing in nature will be reviewed by the audit committee annually to ensure that such related person transaction has been conducted in accordance with the previous approval granted by the audit committee, if any, and that all required disclosures regarding the related person transaction are made.
Transactions involving compensation of executive officers will be reviewed and approved by the compensation committee in the manner specified in the charter of the compensation committee.
A related person transaction reviewed under this policy will be considered approved or ratified if it is authorized by the audit committee in accordance with the standards set forth in our related person transaction policy after full disclosure of the related person’s interests in the transaction. As appropriate for the circumstances, the audit committee will review and consider:
107
Table of Contents
| • | the related person’s interest in the related person transaction; |
| • | the approximate dollar value of the amount involved in the related person transaction; |
| • | the approximate dollar value of the amount of the related person’s interest in the transaction without regard to the amount of any profit or loss; |
| • | whether the transaction was undertaken in the ordinary course of business; |
| • | whether the transaction with the related person is proposed to be, or was, entered into on terms no less favorable to us than terms that could have been reached with an unrelated third party; |
| • | the purpose of, and the potential benefits to us of, the transaction; and |
| • | any other information regarding the related person transaction or the related person in the context of the proposed transaction that would be material to stockholders in light of the circumstances of the particular transaction. |
The audit committee reviews all relevant information available to it about the related person transaction. The audit committee may approve or ratify the related person transaction only if the audit committee determines that, under all of the circumstances, the transaction is in, or is not inconsistent with, our best interests. The audit committee may, in its sole discretion, impose conditions as it deems appropriate on us or the related person in connection with approval of the related person transaction.
Director Independence
Under The NASDAQ Marketplace Rules, a director will only qualify as an “independent director” if, in the opinion of our board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
Our board of directors has determined that each of our directors, with the exception of David Stack, is an “independent director” as defined under Rule 5605(a)(2) of The NASDAQ Marketplace Rules. In making such independence determination, the board of directors considered the relationships that each such non-employee director has with us and all other facts and circumstances that the board of directors deemed relevant in determining their independence, including the beneficial ownership of our capital stock by each non-employee director. In considering the independence of the directors listed above, our board of directors considered the association of our directors with the holders of more than 5% of our common stock.
| Item 14. | Principal Accountant Fees and Services |
The following table summarizes the fees of J.H. Cohn LLP, our independent registered public accounting firm, billed to us for each of the last two fiscal years:
| Purchaser |
Fiscal Year ended December 31, | |||||||
| 2009 | 2010 | |||||||
| Audit Fees(1) |
$ | — | $ | 568,000 | ||||
| Audit-Related Fees(2) |
— | 8,000 | ||||||
| Tax Fees(3) |
— | 1,000 | ||||||
| All Other Fees(4) |
3,000 | — | ||||||
| Total Fee |
$ | 3,000 | $ | 577,000 | ||||
108
Table of Contents
| (1) | Represents fees for services rendered for three years of audits and certain reviews of our financial statements. Includes fees for services associated with documents filed with the SEC, such as our S-1 registration statement, including documents issued in connection with those filings such as consents and comfort letters. |
| (2) | Represents fees associated with consultations relating to accounting consequences of certain proposed transactions. |
| (3) | Represents fees associated with tax consultations. |
| (4) | Represents fees associated with consultations relating to certain proposed transactions, prior to their engagement as the Company’s independent registered public accounting firm in 2010. |
Audit Committee Pre-Approval Policies and Procedures
The audit committee has adopted policies and procedures relating to the approval of all audit and non-audit services that are to be performed by our independent registered public accounting firm. This policy generally provides that we will not engage our independent registered public accounting firm to render audit or non-audit services unless the service is specifically approved in advance by the audit committee or the engagement is entered into pursuant to the pre-approval procedures described below.
From time to time, the audit committee may pre-approve specified types of services that are expected to be provided to us by our independent registered public accounting firm during the next 12 months. Any such pre-approval is detailed as to the particular service or type of services to be provided and is also generally subject to a maximum dollar amount.
The audit committee has delegated authority to the chair of the audit committee to pre-approve any audit or non-audit service to be provided to us by our independent registered public accounting firm. Any approval of services by the chair of the audit committee pursuant to this delegated authority must be reported on at the next meeting of the audit committee.
During our 2010 fiscal year, no services were provided to us by J.H. Cohn LLP or any other accounting firm other than in accordance with the pre-approval policies and procedures described above.
109
Table of Contents
| Item 15. | Exhibits and Financial Statement Schedules |
| (a) | Documents filed as part of Form 10-K. |
| (1) | Financial Statements |
Report of Independent Registered Public Accounting Firm
Consolidated Balance Sheets
Consolidated Statements of Operations
Consolidated Statements of Stockholder’s Equity (Deficit)
Consolidated Statements of Cash Flows
Notes to Consolidated Financial Statements
| (2) | Schedules |
Schedules have been omitted as all required information has been disclosed in the financial statements and related footnotes.
| (3) | Exhibits |
The Exhibits listed in the Exhibit Index are filed as a part of this Form 10-K.
110
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| PACIRA PHARMACEUTICALS, INC. | ||||
| Date: March 31, 2011 |
By: | /s/ David Stack David Stack President and Chief Executive Officer | ||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signature |
Title |
Date | ||
| /s/ David Stack David Stack |
Director, President and Chief Executive Officer (Principal Executive Officer) |
March 31, 2011 | ||
| /s/ James Scibetta James Scibetta |
Chief Financial Officer (Principal Financial and Accounting Officer) |
March 31, 2011 | ||
| /s/ Fred Middleton Fred Middleton |
Chairman | March 31, 2011 | ||
| /s/ Luke Evnin Luke Evnin |
Director | March 31, 2011 | ||
| /s/ Carl Gordon Carl Gordon |
Director | March 31, 2011 | ||
| /s/ John Longenecker John Longenecker |
Director | March 31, 2011 | ||
| /s/ Gary Pace Gary Pace |
Director | March 31, 2011 | ||
| /s/ Andreas Wicki Andreas Wicki |
Director | March 31, 2011 | ||
Table of Contents
Pacira Pharmaceuticals, Inc.
Index to Consolidated Financial Statements
F-1
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders
Pacira Pharmaceuticals, Inc.
We have audited the consolidated balance sheets of Pacira Pharmaceuticals, Inc. and Subsidiaries as of December 31, 2010 and 2009, and the related consolidated statements of operations, stockholders’ equity (deficit) and cash flows for each of the three years in the period ended December 31, 2010. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Pacira Pharmaceuticals, Inc. and Subsidiaries as of December 31, 2010 and 2009, and their results of operations and cash flows for each of the three years in the period ended December 31, 2010, in conformity with accounting principles generally accepted in the United States of America.
/s/ J.H. Cohn LLP
Roseland, New Jersey
March 31, 2011
F-2
Table of Contents
Consolidated Balance Sheets
as of December 31, 2010 and 2009
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (In thousands, except share and per share amounts) |
||||||||
| ASSETS | ||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 26,133 | $ | 7,077 | ||||
| Restricted cash |
1,314 | 1,216 | ||||||
| Trade accounts receivable |
1,191 | 1,455 | ||||||
| Inventories |
1,605 | 1,729 | ||||||
| Prepaid expenses and other current assets |
812 | 1,072 | ||||||
| Total current assets |
31,055 | 12,549 | ||||||
| Fixed assets, net |
23,950 | 19,560 | ||||||
| Intangibles, net |
8,912 | 11,178 | ||||||
| Other assets, net |
2,645 | 667 | ||||||
| Total assets |
$ | 66,562 | $ | 43,954 | ||||
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | ||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 6,038 | $ | 6,994 | ||||
| Accrued expenses |
3,260 | 3,478 | ||||||
| Current portion of royalty interest obligation |
1,575 | 1,599 | ||||||
| Current portion of deferred revenue |
2,267 | 2,346 | ||||||
| Current portion of long-term debt |
3,182 | — | ||||||
| Total current liabilities |
16,322 | 14,417 | ||||||
| Related party debt, including accrued interest |
49,795 | 22,173 | ||||||
| Long-term debt |
21,869 | — | ||||||
| Royalty interest obligation, excluding current potion |
2,996 | 3,647 | ||||||
| Deferred revenue, excluding current portion |
18,138 | 20,387 | ||||||
| Contingent purchase liability |
2,042 | 2,042 | ||||||
| Deferred rent |
1,331 | 1,177 | ||||||
| Other long-term liabilities |
2,452 | 3,060 | ||||||
| Total liabilities |
114,945 | 66,903 | ||||||
| Commitments and contingencies |
||||||||
| Stockholders’ deficit: |
||||||||
| Preferred stock, par value $0.001, 88,000,000 shares authorized, 6,322,640 issued and outstanding at December 31, 2010 and 2009 (liquidation preference of $85,000,000) |
6 | 6 | ||||||
| Common stock, par value $0.001, 120,000,000 shares authorized, 575,095 shares issued and 574,030 shares outstanding at December 31, 2010; 573,920 shares issued and outstanding at December 31, 2009 |
1 | 1 | ||||||
| Additional paid-in capital |
88,523 | 86,806 | ||||||
| Accumulated deficit |
(136,911 | ) | (109,762 | ) | ||||
| (48,381 | ) | (22,949 | ) | |||||
| Less: treasury stock, 1,065 shares at cost |
(2 | ) | — | |||||
| Total stockholders’ deficit |
(48,383 | ) | (22,949 | ) | ||||
| Total liabilities and stockholders’ deficit |
$ | 66,562 | $ | 43,954 | ||||
See accompanying notes to consolidated financial statements
F-3
Table of Contents
Consolidated Statements of Operations
Years Ended December 31, 2010, 2009 and 2008
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (In thousands, except share and per share data) |
||||||||||||
| Revenues: |
||||||||||||
| Supply revenue |
$ | 7,640 | $ | 6,324 | $ | 6,852 | ||||||
| Royalties |
3,705 | 4,044 | 3,648 | |||||||||
| Collaborative licensing and development revenue |
3,217 | 4,638 | 3,425 | |||||||||
| Total revenues |
14,562 | 15,006 | 13,925 | |||||||||
| Operating expenses: |
||||||||||||
| Cost of revenues |
12,276 | 12,301 | 17,463 | |||||||||
| Research and development |
18,628 | 26,233 | 33,214 | |||||||||
| Selling, general and administrative |
6,030 | 5,020 | 8,611 | |||||||||
| Total operating expenses |
36,934 | 43,554 | 59,288 | |||||||||
| Loss from operations |
(22,372 | ) | (28,548 | ) | (45,363 | ) | ||||||
| Other income |
150 | 367 | (224 | ) | ||||||||
| Loss on early extinguishment of debt |
(184 | ) | — | — | ||||||||
| Interest: |
||||||||||||
| Interest income |
146 | 77 | 235 | |||||||||
| Interest expense |
(3,959 | ) | (1,723 | ) | — | |||||||
| Royalty interest obligation |
(930 | ) | (1,880 | ) | 3,490 | |||||||
| Net loss |
$ | (27,149 | ) | $ | (31,707 | ) | $ | (41,862 | ) | |||
| Basic and diluted net loss per common share |
$ | (47.29 | ) | $ | (55.32 | ) | $ | (79.23 | ) | |||
| Weighted average common shares outstanding—basic and diluted |
574,072 | 573,118 | 528,357 | |||||||||
See accompanying notes to consolidated financial statements
F-4
Table of Contents
Consolidated Statements of Stockholders’ Equity (Deficit)
Years Ended December 31, 2010, 2009 and 2008
| Preferred Stock | Common Stock | Additional Paid-In Capital |
Accumulated Deficit |
Treasury Stock |
Total | |||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||||||||||
| Balances, January 1, 2008 |
3,347 | $ | 3 | 465 | $ | 1 | $ | 45,126 | $ | (36,193 | ) | $ | — | $ | 8,937 | |||||||||||||||||
| Issuance of preferred stock |
2,975 | 3 | 39,997 | 40,000 | ||||||||||||||||||||||||||||
| Exercise of stock options |
107 | — | 173 | 173 | ||||||||||||||||||||||||||||
| Share-based compensation |
242 | 242 | ||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | (41,862 | ) | — | (41,862 | ) | ||||||||||||||||||||||
| Balances, December 31, 2008 |
6,322 | 6 | 572 | 1 | 85,538 | (78,055 | ) | — | 7,490 | |||||||||||||||||||||||
| Exercise of stock options |
2 | — | 3 | 3 | ||||||||||||||||||||||||||||
| Share-based compensation |
524 | 524 | ||||||||||||||||||||||||||||||
| Issue of warrants to landlord |
204 | 204 | ||||||||||||||||||||||||||||||
| Debt discount from beneficial conversion features and issuance of warrants with convertible notes |
537 | 537 | ||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | (31,707 | ) | — | (31,707 | ) | ||||||||||||||||||||||
| Balances, December 31, 2009 |
6,322 | 6 | 574 | 1 | 86,806 | (109,762 | ) | — | (22,949 | ) | ||||||||||||||||||||||
| Exercise of stock options |
1 | — | 2 | 2 | ||||||||||||||||||||||||||||
| Share-based compensation |
23 | 23 | ||||||||||||||||||||||||||||||
| Purchase of treasury stock |
(2 | ) | (2 | ) | ||||||||||||||||||||||||||||
| Value of warrants issued with debt and beneficial conversion feature |
1,692 | 1,692 | ||||||||||||||||||||||||||||||
| Net loss |
— | — | — | — | — | (27,149 | ) | — | (27,149 | ) | ||||||||||||||||||||||
| Balances, December 31, 2010 |
6,322 | $ | 6 | 575 | $ | 1 | $ | 88,523 | $ | (136,911 | ) | $ | (2 | ) | $ | (48,383 | ) | |||||||||||||||
See accompanying notes to consolidated financial statements
F-5
Table of Contents
Consolidated Statements of Cash Flows
Years Ended December 31, 2010, 2009 and 2008
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (In thousands) | ||||||||||||
| Operating activities: |
||||||||||||
| Net loss |
$ | (27,149 | ) | $ | (31,707 | ) | $ | (41,862 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||||||
| Depreciation and amortization |
4,071 | 4,146 | 4,227 | |||||||||
| Amortization of other assets and unfavorable lease obligation |
35 | (314 | ) | (396 | ) | |||||||
| Amortization of note discounts and warrants |
146 | 600 | — | |||||||||
| Impairment loss |
— | — | 125 | |||||||||
| Loss on disposal of fixed assets |
11 | 1,707 | 301 | |||||||||
| Share-based compensation |
23 | 524 | 242 | |||||||||
| Change in royalty interest obligation |
(675 | ) | 185 | (5,183 | ) | |||||||
| Changes in operating assets and liabilities: |
||||||||||||
| Restricted cash |
(98 | ) | (34 | ) | 248 | |||||||
| Trade accounts receivable |
264 | 1,130 | (1,562 | ) | ||||||||
| Inventories |
124 | 299 | 277 | |||||||||
| Other current assets |
146 | 244 | (40 | ) | ||||||||
| Accounts payable |
(612 | ) | (4,438 | ) | 4,807 | |||||||
| Other liabilities |
1,009 | 2,724 | (2,122 | ) | ||||||||
| Deferred revenue |
(2,329 | ) | 3,793 | 11,303 | ||||||||
| Deferred rent |
154 | 303 | 446 | |||||||||
| Net cash used in operating activities |
(24,880 | ) | (20,838 | ) | (29,189 | ) | ||||||
| Investing activities: |
||||||||||||
| Purchase of fixed assets |
(6,770 | ) | (5,509 | ) | (5,840 | ) | ||||||
| Proceeds from sale of fixed assets |
1 | — | 2 | |||||||||
| Net cash used in investing activities |
(6,769 | ) | (5,509 | ) | (5,838 | ) | ||||||
| Financing activities: |
||||||||||||
| Proceeds from issuance of preferred stock |
— | — | 40,000 | |||||||||
| Proceeds from exercise of stock options and issuance of common stock |
2 | 3 | 173 | |||||||||
| Purchase of treasury stock |
(2 | ) | — | — | ||||||||
| Proceeds from convertible notes |
7,500 | 10,625 | — | |||||||||
| Proceeds from secured promissory notes and credit facility |
56,250 | 10,625 | — | |||||||||
| Payoff of credit facility |
(11,250 | ) | — | — | ||||||||
| Financing costs |
(1,795 | ) | (215 | ) | — | |||||||
| Net cash provided by financing activities |
50,705 | 21,038 | 40,173 | |||||||||
| Net increase (decrease) in cash and cash equivalents |
19,056 | (5,309 | ) | 5,146 | ||||||||
| Cash and cash equivalents, beginning of year |
7,077 | 12,386 | 7,240 | |||||||||
| Cash and cash equivalents, end of year |
$ | 26,133 | $ | 7,077 | $ | 12,386 | ||||||
| Supplemental cash flow information |
||||||||||||
| Cash paid for interest |
$ | 2,097 | $ | 1,714 | $ | 1,692 | ||||||
| Non cash investing and financing activities: |
||||||||||||
| Accrual for repurchase of intangibles |
$ | — | $ | 323 | $ | 294 | ||||||
| Accrued fixed asset purchases |
$ | — | $ | 2,254 | $ | 3,682 | ||||||
| Value of warrants issued with debt and beneficial conversion feature |
$ | 1,692 | $ | 537 | $ | — | ||||||
| Value of warrants issued to landlord |
$ | — | $ | 204 | $ | — | ||||||
| Accrued financing cost |
$ | 500 | $ | — | $ | — | ||||||
See accompanying notes to consolidated financial statements
F-6
Table of Contents
Notes to Consolidated Financial Statements
1. BUSINESS
Pacira Pharmaceuticals, Inc., and its subsidiaries (collectively, the “Company” or “Pacira”) is an emerging specialty pharmaceutical company focused on the development, commercialization and manufacture of proprietary pharmaceutical products, based on its proprietary DepoFoam drug delivery technology, for use in hospitals and ambulatory surgery centers.
The Company was incorporated in Delaware under the name Blue Acquisition Corp. in December 2006 and changed its name to Pacira, Inc. in June 2007. In October 2010, the Company changed its name to Pacira Pharmaceuticals, Inc. Pacira Pharmaceuticals, Inc. is the holding company for the Company’s California operating subsidiary of the same name, which we refer to as PPI-California.
As further discussed in Note 4, on March 24, 2007, or the Acquisition Date, MPM Capital, Sanderling Ventures, OrbiMed Advisors, HBM BioVentures, the Foundation for Research and their co-investors, or the Investors, through Pacira Pharmaceuticals, Inc., acquired PPI-California, from SkyePharma Holding, Inc., which we refer to as the Acquisition. PPI-California was known as SkyePharma, Inc. prior to the Acquisition.
Risks and Uncertainties
The Company is subject to risks common to companies in similar industries and stages of development, including, but not limited to, competition from larger companies, reliance on revenue from few customers and products, new technological innovations, dependence on key personnel, reliance on third-party service providers and vendors, protection of proprietary technology, and compliance with government regulations.
F-7
Table of Contents
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation and Principles of Consolidation
The consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries PPI-California and Pacira Limited. Pacira Limited was incorporated in the United Kingdom and its functional currency is the U.S. dollar. Intercompany accounts and transactions have been eliminated in consolidation. The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company has incurred losses and negative operating cash flows since inception and future losses are anticipated. As described in Note 18, the Company has raised $42 million of gross proceeds, before offering costs, through an initial public offering completed on February 8, 2011. Although the offering and our cash resources provide the Company adequate funding for the next 12 months, the longer-term ability of the Company to continue as a going concern is dependent on improving the Company’s profitability and cash flows and securing additional financing.
Reverse Stock Split
On January 12, 2011, the board of directors of the Company approved, and on January 12, 2011 the stockholders of the Company approved, a one-for-10.755 reverse stock split of the Company’s outstanding common stock, which was effected on January 12, 2011. Stockholders entitled to fractional shares as a result of the reverse stock split will receive a cash payment for such fractional shares within 180 days following the effective date of the reverse stock split in lieu of receiving fractional shares. The reverse stock split affected all holders of the Company’s preferred stock and common stock uniformly. Shares of common stock underlying outstanding stock options were proportionately reduced and the respective exercise prices of the stock options were proportionately increased in accordance with the terms of the agreements governing such securities. Shares of common stock reserved for issuance upon the conversion of the Company’s series A preferred stock and convertible notes were proportionately reduced and the respective conversion prices were proportionately increased. All references to preferred and common stock and per share information, except par value and authorized shares, in these consolidated financial statements and notes have been adjusted to reflect the effects of the reverse stock split.
Use of Estimates
The preparation of consolidated financial statements in conformity with accounting principles generally accepted in the United States of America requires the use of estimates and assumptions that affect the reported amounts of assets and liabilities, including disclosure of contingent assets and contingent liabilities, at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting period. The Company’s critical accounting policies are those that are both most important to the Company’s consolidated financial condition and results of operations and require the most difficult, subjective or complex judgments on the part of management in their application, often as a result of the need to make estimates about the effect of matters that are inherently uncertain. Because of the uncertainty of factors surrounding the estimates or judgments used in the preparation of the consolidated financial statements, actual results may materially vary from these estimates.
Cash and Cash Equivalents
All highly-liquid investments with maturities of 90 days or less when purchased are considered cash equivalents.
Restricted Cash
As further discussed in Note 10, the Company has entered into a financing agreement with Royalty Securitization Trust I (“RST”) for the sale of a royalty interest in its DepoCyt(e) and DepoDur supply revenue and royalties. As part of this financing agreement, the Company and RST maintain a lockbox, where all DepoCyt(e) and DepoDur supply revenue and royalties are received. The Company has no minimum payment obligations under this agreement. Commencing on April 1 of every year, the first $2.5 million received in the lockbox is restricted and will
F-8
Table of Contents
be used to make quarterly payments due to RST, if any, under the agreement during the subsequent 12 month period. On March 31 of the subsequent year, the balance of cash in the lockbox, if any, is remitted to Pacira. The RST agreement terminates on December 31, 2014. The royalty interest agreement pertains only to DepoCyt(e) and DepoDur, and does not include revenue related to EXPAREL or any other product candidates.
Credit Risk
Financial instruments which potentially subject the Company to concentrations of credit risk consist primarily of cash and cash equivalents and accounts receivable. The Company maintains its cash and cash equivalents with high-credit quality financial institutions. At times, such amounts may exceed Federal insured limits. At December 31, 2010, the Company had cash and cash equivalent balances in excess of Federally insured limits in the amount of approximately $1.3 million. The Company performs ongoing credit evaluations of its customers, as warranted, and generally does not require collateral. Revenues from the supply of manufactured product for the Company’s commercial partners, royalties, contractual services provided to its collaboration partners and licensing and development fees are primarily derived from major pharmaceutical companies that generally have significant cash resources. Allowances for doubtful accounts receivable are maintained based on historical payment patterns, aging of accounts receivable and actual write-off history. As of December 31, 2010 and 2009, no allowances for doubtful accounts were deemed necessary by the Company on its trade accounts receivable.
Concentration of Major Customers
The Company’s customers are its commercial and collaborative and licensing partners. For the year ended December 31, 2010, the Company’s three largest customers accounted for 49%, 21% and 13%, respectively, of the Company’s net revenues. For the year ended December 31, 2009, the Company’s three largest customers accounted for 44%, 23%, 20%, respectively, of the Company’s net revenues. For the year ended December 31, 2008, the Company’s four largest customers accounted for 46%, 20%, 16% and 12%, respectively, of the Company’s net revenues. No other individual customers accounted for more than 10% of net revenues. As of December 31, 2010, the Company’s three largest customers accounted for 66%, 17% and 11%, respectively, of the Company’s trade accounts receivables. As of December 31, 2009, the Company’s three largest customers accounted for 56%, 26% and 13%, respectively, of the Company’s trade accounts receivables. The Company is dependent on these commercial partners to market and sell DepoCyt(e) and DepoDur, from which a substantial portion of its revenues is derived; therefore, the Company’s future revenues from these products are highly dependent on these collaboration and distribution arrangements.
Domestic net revenues for the years ended December 31, 2010, 2009 and 2008 accounted for 48%, 52% and 48% of the Company’s net revenues, respectively. Export revenues for the years ended December 31, 2010, 2009 and 2008 accounted for 52%, 48% and 52% of the Company’s net revenues, respectively.
Inventories
Inventories consist of finished goods held for sale and distribution, raw materials and work in process, and are stated at the lower of cost, which includes amounts related to material, labor and overhead, and is determined using the first-in, first-out (“FIFO”) method, or market (net realizable) value. The Company periodically reviews its inventory to identify obsolete, slow-moving or otherwise unsalable inventories, and establishes allowances for situations in which the cost of the inventory is not expected to be recovered. Overhead costs associated with excess manufacturing capacity are charged to cost of revenue, as incurred.
Fixed Assets
Fixed assets are recorded at cost, net of accumulated depreciation and amortization. The Company reviews its property, plant and equipment assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable.
F-9
Table of Contents
Depreciation of fixed assets is provided over their estimated useful lives on a straight-line basis. Leasehold improvements are amortized on a straight-line basis over the shorter of their estimated useful lives or the related lease terms. Useful lives by asset category are as follows:
| Asset Category |
Years | |
| Manufacturing and laboratory equipment | 5 to 10 years | |
| Computer equipment and software | 1 to 3 years | |
| Office furniture and equipment | 5 years | |
| Leasehold improvements | 1 to 9 years (up to the lease term) |
Impairment of Long-Lived Assets
Intangible assets are recorded at cost, net of accumulated amortization. Amortization of intangible assets is provided over their estimated useful lives on a straight-line basis. Management reviews long-lived assets, including fixed assets, for impairment whenever events or changes in circumstances indicate the carrying amount of an asset may not be recoverable. Recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to future undiscounted net cash flows expected to be generated by the asset. If such assets are considered to be impaired, the impairment to be recognized is measured as the amount by which the carrying amount of the assets exceeds the fair value of the assets. Fair value for the Company’s long-lived assets is determined using the expected cash flows discounted at a rate commensurate with the risk involved.
Settlement of Trade Payables
During April 2009, the Company initiated a payables settlement program with its trade creditors using various settlement arrangements. As of April 30, 2009, total outstanding unsecured trade payables subject to these settlement arrangements were $14.3 million. These creditors agreed to settle their outstanding balances for an aggregate of $12.5 million resulting in reduction in payables of $1.8 million. The Company has recorded a $1.3 million reduction to the carrying amount of fixed assets and included a $0.4 million gain in other income on the Company’s consolidated statement of operations for the year ended December 31, 2009 and $0.1 million gain in other income on the Company’s consolidated statement of operations for the year ended December 31, 2010. As of December 31, 2010, $3.3 million related to these settlement arrangements remained outstanding and was included in accounts payable in the Company’s consolidated balance sheet.
Foreign Currencies
Realized gains and losses from foreign currency transactions are reflected in the consolidated statements of operations and were not significant in any period in the years ended December 31, 2010, 2009 or 2008. All foreign currency receivables and payables are measured at the applicable exchange rate at the end of the reporting period.
Income Taxes
The Company uses the asset and liability method of accounting for income taxes. Deferred tax assets and liabilities are recognized for the estimated future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. Valuation allowances are established when necessary to reduce deferred tax assets to the amount expected to be realized. As of December 31, 2010 and 2009, all deferred tax assets were fully offset by a valuation allowance.
The Company accrues interest and penalties, if any, on underpayment of income taxes related to unrecognized tax benefits as a component of income tax expense in its consolidated statements of operations.
F-10
Table of Contents
Revenue Recognition
Supply Revenue—The Company recognizes revenue from products manufactured and supplied to its commercial partners, when the following four basic revenue recognition criteria under the related accounting guidance are met: (1) persuasive evidence of an arrangement exists; (2) delivery has occurred or services have been rendered; (3) the fee is fixed or determinable; and (4) collectability is reasonably assured. The product can be returned within contracted specified time frames if it does not meet the applicable inspection tests. The Company estimates its return reserves based on its experience of historical return rates.
Royalties—The Company recognizes revenue from royalties based on sales of its products into the marketplace by its commercial partners. Royalties are recognized as earned in accordance with contract terms when they can be reasonably estimated and collectability is reasonably assured. The Company’s commercial partners are obligated to report their net product sales and the resulting royalty due to the Company within 60 days from the end of each quarter. Based on historical product sales, royalty receipts and other relevant information, the Company accrues royalty revenue each quarter.
Collaborative licensing and development revenue—The Company recognizes revenue from reimbursements received in connection with feasibility studies and development work for third parties who desire to utilize its DepoFoam extended release drug delivery technology for their products, when the Company’s contractual services are performed, provided collectability is reasonably assured. The Company’s principal costs under these agreements include its personnel conducting research and development, and its allocated overhead, as well as research and development performed by outside contractors or consultants.
The Company recognizes revenues from non-refundable up-front license fees received under collaboration agreements ratably over the performance period as determined under the collaboration agreement (estimated development period in the case of development agreements, and contract period or longest patent life in the case of supply and distribution agreements). If the estimated performance period is subsequently modified, the Company will modify the period over which the up-front license fee is recognized accordingly on a prospective basis. Upon termination of a collaboration agreement, any remaining non-refundable license fees received by the Company, which had been deferred, are generally recognized in full. All such recognized revenues are included in collaborative licensing and development revenue in the Company’s consolidated statements of operations.
The Company recognizes revenue from milestone payments received under collaboration agreements when earned, provided that the milestone event is substantive, its achievability was not reasonably assured at the inception of the agreement, the Company has no further performance obligations relating to the event, and collectability is reasonably assured. If these criteria are not met, the Company recognizes milestone payments ratably over the remaining period of the Company’s performance obligations under the collaboration agreement. All such recognized revenues are included in collaborative licensing and development revenue in the Company’s consolidated statements of operations.
Research and Development Expenses
Research and development expenses consist of costs associated with products being developed internally, and include related personnel expenses, laboratory supplies, active pharmaceutical ingredients, manufacturing supplies, facilities costs, preclinical and clinical trial costs, and other outside service fees. The Company expenses research and development costs as incurred. A significant portion of the Company’s development activities are outsourced to third parties, including contract research organizations. In such cases, the Company may be required to make estimates of related service fees to be accrued.
Per Share Data
Net loss per share is determined in accordance with the two-class method. This method is used for computing basic net loss per share when companies have issued securities other than common stock that contractually entitle the holder to participate in dividends and earnings of the Company. Under the two-class method, net loss is allocated between common shares and other participating securities based on their participation rights in both distributed and undistributed earnings. The Company’s Series A convertible preferred stock are participating securities, since the stockholders are entitled to share in dividends declared by the board of directors with the common stock based on their equivalent common shares.
F-11
Table of Contents
Basic net loss per share is computed by dividing net loss available (attributable) to common stockholders by the weighted average number of shares of common stock outstanding during the period. Because the holders of the Series A Convertible Preferred Stock are not contractually required to share in the Company’s losses, in applying the two-class method to compute basic net loss per common share no allocation to preferred stock was made for the years ended December 31, 2010, 2009 and 2008.
Diluted net loss per share is calculated by dividing net loss available (attributable) to common stockholders as adjusted for the effect of dilutive securities, if any, by the weighted average number of common stock and dilutive common stock outstanding during the period. Potential common shares include the shares of common stock issuable upon the exercise of outstanding stock options and a warrant (using the treasury stock method) and the conversion of the shares of Series A convertible preferred stock (using the more dilutive of the (a) as converted method or (b) the two-class method). Potential common shares in the diluted net loss per share computation are excluded to the extent that they would be anti-dilutive. No potentially dilutive securities are included in the computation of any diluted per share amounts as the Company reported a net loss for all periods presented. Potentially dilutive securities that would be issued upon the conversion of convertible notes, conversion of Series A convertible preferred stock and the exercise of outstanding warrants and stock options, were 8.9 million, 7.2 million and 6.6 million as of December 31, 2010, 2009 and 2008, respectively.
Share-Based Compensation
The Company’s share-based compensation programs include grants of stock options to employees, consultants and non-employee directors. The expense associated with these programs is recognized in the Company’s consolidated statements of operations based on their fair values as they are earned by the employees, consultants and non-employee directors under the applicable vesting terms.
The valuation of stock options is an inherently subjective process, since market values are generally not available for long-term, non-transferable stock options. Accordingly, the Company uses an option pricing model to derive an estimated fair value. In calculating the estimated fair value of stock options granted, the Company uses the Black-Scholes option pricing model which requires the consideration of the following variables for purposes of estimating fair value:
| • | the stock option exercise price; |
| • | the expected term of the option; |
| • | the grant date fair value of the Company’s common stock, which is issuable upon exercise of the option; |
| • | the expected volatility of the Company’s common stock; |
| • | expected dividends on the Company’s common stock; and |
| • | the risk-free interest rate for the expected option term. |
Of the variables above, the Company believes that the selection of an expected term and expected stock price volatility are the most subjective. The Company’s historical stock option exercise experience does not provide a reasonable basis upon which to estimate expected term. Accordingly, the Company uses a term based on a simplified method, pursuant to SEC Staff Accounting Bulletin No. 107, Share-based Payment, for “plain vanilla” options. For calculating stock price volatility, the Company utilizes historical stock prices of publicly traded companies that are similar to Pacira.
The Company estimates the level of award forfeitures expected to occur, and records compensation cost only for those awards that are ultimately expected to vest.
F-12
Table of Contents
Segment Reporting
The Company currently operates in a single operating segment. The Company generates revenue from various sources that result primarily from its revenue from DepoCyt(e) and DepoDur underlying research and development activities. In addition, financial results are prepared and reviewed by management as a single operating segment.
3. RECENT ACCOUNTING PRONOUNCEMENTS
In October 2009, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update No. 2009-13, “Multiple-Deliverable Revenue Arrangements” (“ASU 2009-13”). ASU 2009-13, amends existing revenue recognition accounting pronouncements that are currently within the scope of ASC Subtopic 605-25. This authoritative guidance provides principles for allocation of consideration among its multiple-elements, allowing more flexibility in identifying and accounting for separate deliverables under an arrangement. ASU 2009-13 introduces an estimated selling price method for valuing the elements of a bundled arrangement if vendor-specific objective evidence or third-party evidence of selling price is not available, and significantly expands related disclosure requirements. This guidance is effective on a prospective basis for revenue arrangements entered into or materially modified in fiscal years beginning on or after June 15, 2010. Alternatively, adoption may be on a retrospective basis, and early application is permitted. The adoption of this guidance is not expected to have any impact on our consolidated financial statements.
In April 2010, the FASB issued Accounting Standards Update No. 2010-17, “Milestone Method of Revenue Recognition (Topic 605)” (“ASU 2010-17”). This update provides guidance on defining a milestone and determining when it may be appropriate to apply the milestone method of revenue recognition for research or development transactions. Authoritative guidance on the use of the milestone method did not previously exist. This guidance is effective on a prospective basis for milestones achieved in fiscal years, and interim periods within those years, beginning on or after June 15, 2010. Alternatively, retrospective adoption is permitted for all prior periods. The adoption of this guidance is not expected to have any impact on our consolidated financial statements.
Other pronouncements issued by the FASB or other authoritative accounting standards groups with future effective dates are either not applicable or not significant to the consolidated financial statements of the Company.
4. ACQUISITION OF SKYEPHARMA, INC.
Pacira Pharmaceuticals, Inc., a Delaware corporation, is the holding company for a California operating subsidiary of the same name, which we refer to as PPI-California. On the Acquisition Date, MPM Capital, Sanderling Ventures, OrbiMed Advisors, HBM Bioventures, the Foundation for Research and their co-investors, through Pacira Pharmaceuticals, Inc., acquired PPI-California, from SkyePharma Holding, Inc. for an initial purchase price of $19.6 million.
At the Acquisition Date, the Company determined that the lease rates associated with the assumed facilities leases were above market rates resulting in a $3.3 million unfavorable lease accrual as of the Acquisition Date. The unfavorable lease accrual, which is recorded in other long-term liabilities in the Company’s consolidated balance sheets, is amortized over the remaining terms of the leases.
In addition to the initial $19.6 million purchase price, the Company entered into an earn-out agreement with SkyePharma Holding, Inc. as additional purchase price which was based on Pacira reaching certain revenue milestones following the Acquisition. According to this agreement, Pacira would pay SkyePharma Holding, Inc. royalty payments based on the net revenues of EXPAREL and certain other products from the future yet-to-be-developed biologics product line and milestone payments of up to an aggregate of $62 million upon the occurrence of the following events: a) first commercial sale in the United States; b) first commercial sale in a major EU country (UK, France, Germany, Italy, or Spain); c) annual net sales reach $100 million; d) annual net sales reach $250 million and e) annual net sales reach $500 million. Additionally, the Company agreed to pay to SkyePharma Holding, Inc. a 3% royalty of its sales of EXPAREL in the United States, Japan, the United Kingdom, France, Germany, Italy and Spain. The fair value of this contingent obligation was $13.9 million on the Acquisition Date. For business combinations involving contingent consideration (that is, a combination that might result in the acquiring enterprise recognizing additional purchase price in a future period (also referred to as “contingent consideration”)), the acquiring enterprise is required to recognize, as if it were a liability, an amount equal to the
F-13
Table of Contents
lesser of: (1) the maximum amount of contingent consideration issuable, and (2) the total amount of negative goodwill. Accordingly, even though the fair value of the contingent consideration was $13.9 million, the Company recognized only $2.0 million as a contingent purchase liability as of the Acquisition Date. The carrying amount of the contingent purchase liability to SkyePharma Holding, Inc. was $2.0 million as of December 31, 2010 and 2009. The Company has not paid any earn-out to SkyePharma Holding, Inc. for the years ended December 31, 2010, 2009 and 2008.
5. FAIR VALUE MEASUREMENTS
Financial assets and financial liabilities are required to be measured and reported on a fair value basis using the following three categories for classification and disclosure purposes:
Level 1: Quoted prices (unadjusted) in active markets that are accessible at the measurement date for assets or liabilities. The fair value hierarchy gives the highest priority to Level 1 inputs.
Level 2: Observable prices that are based on inputs not quoted on active markets, but corroborated by market data.
Level 3: Unobservable inputs that are used when little or no market data is available. The fair value hierarchy gives the lowest priority to Level 3 inputs.
In determining fair value, the Company utilizes valuation techniques that maximize the use of observable inputs and minimize the use of unobservable inputs to the extent possible. The Company also considers counterparty credit risk in its assessment of fair value.
The carrying value of financial instruments including cash and cash equivalents, restricted cash, accounts receivable, note receivable, and accounts payable approximate their respective fair values due to the short-term maturities of these instruments and debts. The fair value of the Company’s convertible notes (see Note 10) and promissory notes (see Note 10) cannot be practicably determined due to their related party nature. The carrying value of the long-term debt approximates its fair value as of December 31, 2010, due to the timing of its closing which occurred on November 24, 2010 as described further in Note 10.
6. INVENTORIES
The components of inventories were as follows:
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (In thousands) | ||||||||
| Raw materials |
$ | 1,108 | $ | 716 | ||||
| Work-in-process |
10 | 48 | ||||||
| Finished goods |
487 | 965 | ||||||
| Total |
$ | 1,605 | $ | 1,729 | ||||
7. FIXED ASSETS
Fixed assets, at cost, summarized by major category, consist of the following:
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (in thousands) | ||||||||
| Machinery and laboratory equipment |
$ | 7,002 | $ | 7,124 | ||||
| Computer equipment and software |
765 | 765 | ||||||
F-14
Table of Contents
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (in thousands) | ||||||||
| Office furniture and equipment |
167 | 167 | ||||||
| Leasehold improvements |
3,938 | 3,809 | ||||||
| Construction in progress |
18,144 | 12,289 | ||||||
| Total |
30,016 | 24,154 | ||||||
| Less accumulated depreciation |
(6,066 | ) | (4,594 | ) | ||||
| Fixed assets, net |
$ | 23,950 | $ | 19,560 | ||||
Depreciation expense for the years ended December 31, 2010, 2009 and 2008 was $1.8 million, $1.9 million and $2.0 million, respectively. Depreciation expenses associated with the Company’s commercial products are included in cost of revenue. Depreciation expense associated with the Company’s products in development are included in research and development expenses. Depreciation expense associated with general and administrative activities are included in selling, general and administrative expenses.
8. INTANGIBLE ASSETS
Intangible assets consist of core technology, developed technology, DepoDur rights, and trademarks and trade names acquired in the acquisition of SkyePharma, Inc. (see Note 4). Intangible assets are recorded at cost, net of accumulated amortization. Amortization of intangible assets is provided over their estimated useful lives on a straight-line basis.
Intangible assets are summarized as follows:
| December 31, | Estimated Useful Life |
|||||||||||
| 2010 | 2009 | |||||||||||
| (in thousands) | ||||||||||||
| Core Technology |
||||||||||||
| Gross amount |
$ | 2,900 | $ | 2,900 | 9 years | |||||||
| Accumulated amortization |
(1,208 | ) | (886 | ) | ||||||||
| Net |
1,692 | 2,014 | ||||||||||
| Developed Technology |
||||||||||||
| Gross amount |
11,700 | 11,700 | 7 years | |||||||||
| Accumulated amortization |
(6,268 | ) | (4,596 | ) | ||||||||
| Net |
5,432 | 7,104 | ||||||||||
| Trademarks and trade names |
||||||||||||
| Gross amount |
500 | 500 | 7 years | |||||||||
| Accumulated amortization |
(253 | ) | (176 | ) | ||||||||
| Net |
247 | 324 | ||||||||||
| DepoDur Rights |
||||||||||||
| Gross amount |
2,058 | 2,058 | |
Remaining patent life ending November 2018 |
| |||||||
| Accumulated amortization |
(517 | ) | (322 | ) | ||||||||
| Net |
1,541 | 1,736 | ||||||||||
| Intangible assets, net |
$ | 8,912 | $ | 11,178 | ||||||||
Annual amortization expense for intangibles was $2.3 million, $2.2 million and $2.2 million for the years ended December 31, 2010, 2009 and 2008, respectively. Amortization expenses associated with the Company’s commercial products and developed technology are included in cost of revenue. Amortization expenses associated with the Company’s products in development are included in research and development expenses.
F-15
Table of Contents
The approximate amortization expense for intangibles subject to amortization is as follows (in thousands):
| Core Technology |
Developed Technology |
Trademarks and Tradenames |
DepoDur Rights |
Total | ||||||||||||||||
| 2011 |
$ | 322 | $ | 1,671 | $ | 76 | $ | 196 | $ | 2,265 | ||||||||||
| 2012 |
322 | 1,671 | 76 | 196 | 2,265 | |||||||||||||||
| 2013 |
322 | 1,671 | 76 | 196 | 2,265 | |||||||||||||||
| 2014 |
322 | 419 | 19 | 196 | 956 | |||||||||||||||
| 2015 |
322 | — | — | 196 | 518 | |||||||||||||||
| Thereafter |
82 | — | — | 561 | 643 | |||||||||||||||
| Total |
$ | 1,692 | $ | 5,432 | $ | 247 | $ | 1,541 | $ | 8,912 | ||||||||||
Intangibles are evaluated for potential impairment whenever events or circumstances indicate that the carrying amount may not be recoverable. An impairment loss is recorded to the extent the asset’s carrying value is in excess of the fair value of the asset. When fair values are not readily available, the Company estimates fair values using expected discounted future cash flows. During 2008, the Company recorded an impairment loss of $125,000, primarily related to the Company’s DepoDur trademark. The DepoDur trademark was determined to be impaired because the Company’s revised estimates of future sales were significantly lower than its prior estimates. Such impairment losses are reflected in costs of revenue in the Company’s consolidated statements of operations. No impairment loss was recorded in 2010 and 2009.
9. OTHER BALANCE SHEET DETAILS
Prepaid expenses and other current assets consist of the following:
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (in thousands) | ||||||||
| Prepaid expenses |
$ | 569 | $ | 761 | ||||
| Other |
243 | 311 | ||||||
| Total |
$ | 812 | $ | 1,072 | ||||
Other assets consist of the following:
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (In thousands) | ||||||||
| Deferred financing costs, net |
$ | 590 | $ | 133 | ||||
| Deferred IPO costs |
1,407 | — | ||||||
| Note receivable—EKR |
583 | 470 | ||||||
| Other |
65 | 64 | ||||||
| Total |
$ | 2,645 | $ | 667 | ||||
Accrued expenses consist of the following:
F-16
Table of Contents
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (in thousands) | ||||||||
| Compensation and benefits |
$ | 643 | $ | 518 | ||||
| Lease rent deferral—current portion |
1,125 | 1,705 | ||||||
| Other |
1,492 | 1,255 | ||||||
| Total |
$ | 3,260 | $ | 3,478 | ||||
10. DEBT
The composition of the Company’s debt and financing obligations, including accrued interest, is as follows:
| December 31, | ||||||||
| 2010 | 2009 | |||||||
| (In thousands) | ||||||||
| Related party debt, including accrued interest: |
||||||||
| 2009 Convertible notes |
$ | 11,655 | $ | 11,124 | ||||
| 2009 Secured notes |
12,324 | 11,049 | ||||||
| 2010 Secured notes |
15,462 | — | ||||||
| 2010 HBM Secured note |
3,945 | — | ||||||
| 2010 Convertible note, net of debt discount |
6,409 | — | ||||||
| 49,795 | 22,173 | |||||||
| Financing obligations: |
||||||||
| Hercules note, current portion |
3,182 | — | ||||||
| Hercules note, long-term portion, net of debt discount |
21,869 | — | ||||||
| Royalty interest obligation, current portion |
1,575 | 1,599 | ||||||
| Royalty interest obligation, long-term portion |
2,996 | 3,647 | ||||||
| 29,622 | 5,246 | |||||||
| Total debt and financing obligations |
$ | 79,417 | $ | 27,419 | ||||
CONVERTIBLE NOTES PAYABLE
2009 Convertible Notes
In January 2009, the Company sold $10.63 million aggregate principal amount of unsecured convertible promissory notes, or the 2009 Convertible Notes. The 2009 Convertible Notes were issued with detachable warrants to purchase an aggregate of 158,061 shares of the Company’s common stock at an exercise price of $2.69 per share. In recording the transaction, the Company allocated the proceeds of the 2009 Convertible Notes and the warrants based on their relative fair values. Fair value of the warrants was determined using the Black-Scholes valuation model and allocated to additional paid-in capital. It was also determined that the 2009 Convertible Notes contained a beneficial conversion feature since the fair value of the common stock issuable upon the conversion of the notes exceeded the value allocated to the notes. The Company recognized and measured the embedded beneficial conversion feature of each of the 2009 Convertible Notes by allocating a portion of the proceeds equal to the intrinsic value of that feature to additional paid-in capital. The intrinsic value of the beneficial conversion feature was calculated at the commitment date as the difference between the conversion price and the fair value of the securities into which the convertible instruments were convertible.
The 2009 Convertible Notes accrue interest at an annual rate of 5% payable at maturity or at the time of conversion. In connection with entering into the Hercules Credit Facility, as described further below, the maturity dates of the 2009 Convertible Notes was extended to the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated.
F-17
Table of Contents
Also in connection with entering into the Hercules Credit Facility, the holders of the 2009 Convertible Notes entered into a subordination agreement with Hercules pursuant to which the 2009 Convertible Notes were subordinated to the Hercules Credit Facility. Previously, in connection with GECC Credit Facility, as further described below, holders of the 2009 Convertible Notes had entered into an inter-creditor agreement with the holders of the 2009 Secured Notes and the 2010 Secured Notes whereby the 2009 Convertible Notes were subordinated to the 2009 Secured Notes and the 2010 Secured Notes, and the holders of the 2009 Secured Notes agreed to share payments pro rata with the holders of the 2010 Secured Notes.
Upon the closing of a Qualified Financing (as defined below), unless the holders of a majority of the aggregate principal amount of all 2009 Convertible Notes have elected Optional Conversion of the 2009 Convertible Notes as described below, all outstanding principal and accrued interest under the 2009 Convertible Notes will automatically convert into shares of the same class and series of capital stock of the Company issued to investors in the Qualified Financing at a conversion price per share equal to the price per share paid by other investors in the Qualified Financing. A “Qualified Financing” means the next sale of preferred stock of the Company (i) with gross proceeds to the Company (including proceeds from any indebtedness of the Company that converts into equity in such financing) of at least $10 million or (ii) that is designated as a “Qualified Financing” by the holders of a majority of the aggregate principal amount of all 2009 Convertible Notes. Additionally, the 2009 Convertible Notes and any unpaid interest may be converted to Series A convertible preferred stock upon the election by the holders of a majority of the aggregate principal amount of all 2009 Convertible Notes with a conversion price paid per share equal to the price per share of Series A convertible preferred stock at the time of conversion (“Optional Conversion”). The warrants have an exercise price per share of $2.69 and will expire on January 21, 2014.
In the event of the completion of a merger or consolidation, sale of all the Company’s assets or common stock or voluntary or involuntary liquidation, prior to full payment of the 2009 Convertible Notes or prior to the time when the 2009 Convertible Notes may be converted, the 2009 Convertible Notes will be due and payable with a control premium and the then outstanding principal and unpaid accrued interest and will be senior to all payments of Company common stock and Series A convertible preferred stock. Additionally, the 2009 Convertible Notes are due on demand in the event of default, litigation that threatens to materially and adversely affect the Company’s business, operations, assets or results of operations, or bankruptcy by the Company.
The value of the warrants has been recorded as a discount to the 2009 Convertible Notes and amortized as a component of interest expense over the original term of the notes. For the year ended December 31, 2009, the amortization of the discount was $269,000 resulting in no remaining balance as of December 31, 2010 and 2009.
The value of the beneficial conversion feature has been recorded as a discount to the 2009 Convertible Notes and amortized as a component of interest expense over the original term of the notes. For the year ended December 31, 2009, the amortization of the discount was $269,000 resulting in no remaining balance as of December 31, 2010 and 2009.
The outstanding principal and accrued interest on the 2009 Convertible Notes was $11.7 million and $11.1 million as of December 31, 2010 and 2009, respectively, and annual interest expense associated with these notes was $0.5 million for the years ended December 31, 2010 and 2009. As further discussed in Note 18, the 2009 Convertible Notes converted into an aggregate of 871,635 shares of common stock upon the Company’s initial public offering in February 2011.
December 2010 Convertible Notes
On December 29, 2010, the Company sold $15.0 million in aggregate principal amount of convertible promissory notes, or the December 2010 Convertible Notes, in a private placement to certain of its existing investors. 50% of the principal amount was funded on December 29, 2010. The remaining 50% of the principal amount will be funded in a second closing to occur upon written request of holders of at least 75% of the outstanding principal amount of the December 2010 Convertible Notes on or before the earlier of the completion of the Company’s initial public offering or March 31, 2011. In connection with the issuance and sale of the December
F-18
Table of Contents
2010 Convertible Notes, the Company issued warrants to the holders of the December 2010 Convertible Notes to purchase an aggregate of 167,361 shares of its common stock with an exercise price of $13.44 per share. Pursuant to the terms of the agreement for the issuance and sale of the December 2010 Convertible Notes, in the event a second closing of the issuance and sale of the December 2010 Convertible Notes occurs, the Company will issue warrants to the holders of the December 2010 Convertible Notes to purchase an additional 167,361 shares of its common stock with an exercise price of $13.44 per share. The Company’s existing investors have indicated they will not purchase the additional $7.5 million of December 2010 Convertible Notes in the second closing. The December 2010 Convertible Notes will have an interest rate of 5% per year from and after March 31, 2011 and all principal and accrued and unpaid interest on the December 2010 Convertible Notes is due and payable upon the earliest of: (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated.
Upon completion of the Company’s proposed initial public offering (see Note 18), all principal and interest due under the December 2010 Convertible Notes will be converted into shares of the Company’s common stock at a conversion price equal to the price per share of common stock sold in the Company’s initial public offering. Purchasers of the December 2010 Convertible Notes included certain holders of more than 5% of the Company’s capital stock, or entities affiliated with them.
The fair value of the warrants granted on December 29, 2010 is $0.5 million and the fair value of the beneficial conversion feature is a corresponding $0.5 million. The value of the warrants and the beneficial conversion feature was recorded as a discount to the December 2010 Convertible Notes and amortized as a component of interest expense over the original term of the December 2010 Convertible Notes. Upon the completion of the Company’s initial public offering, when the December 2010 Convertible Notes are converted into common stock, any unamortized balance will be recognized in full on the date of such event.
The outstanding principal and accrued interest on the 2010 Convertible Notes was $7.5 million as of December 31, 2010. As further discussed in Note 18, the 2010 Convertible Notes converted into an aggregate of 1,071,428 shares of common stock upon the Company’s initial public offering in February 2011.
SECURED PROMISSORY NOTES
2009 Secured Notes
In June 2009, the Company entered into an agreement with certain of its existing investors to issue $10.63 million in aggregate principal amount of secured notes, or the 2009 Secured Notes. To secure the performance of the Company’s obligations under purchase agreement for the 2009 Secured Notes, the Company granted a security interest in all of its assets except the assets that secure the Company’s obligations under its agreement with Paul Capital to the investors. In connection with entering into the Hercules Credit Facility, the holders of the 2009 Secured Notes entered into a subordination agreement with Hercules pursuant to which the 2009 Secured Notes were subordinated to the Hercules Credit Facility. Previously, in connection with GECC Credit Facility, as further described below, 2009 Secured Noteholders had entered into an inter-creditor agreement with the holders of the 2009 Convertible Notes and the 2010 Secured Notes whereby the 2009 Convertible Notes were subordinated to the 2009 Secured Notes and the 2010 Secured Notes, and the holders of the 2009 Secured Notes agreed to share payments pro rata with the holders of the 2010 Secured Notes.
The 2009 Secured Notes have an interest rate of 12% per year and all principal and accrued and unpaid interest on the 2009 Secured Notes is due on December 31, 2010. In connection with entering into the Hercules Credit Facility, the maturity dates of the 2009 Secured Notes was extended to the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated.
The outstanding principal and accrued interest on the 2009 Secured Notes was $12.3 million and $11.0 million as of December 31, 2010 and 2009, respectively, and interest expense associated with these promissory notes was $1.3 million and $0.4 million for the years ended December 31, 2010 and 2009, respectively.
F-19
Table of Contents
2010 Secured Notes
In March 2010, the Company entered into an agreement with certain of its existing investors to issue $15 million in aggregate principal amount of secured notes, or the 2010 Secured Notes. To secure the performance of its obligations under the purchase agreement for the 2010 Secured Notes, the Company granted a subordinated security interest in substantially all of its assets, including its intellectual property assets, to the investors. The investors purchased the entire $15 million of 2010 Secured Notes in three closings in March, June and September 2010.
The 2010 Secured Notes have an interest rate of 5% per year and all principal and accrued and unpaid interest is due on December 31, 2010. In connection with entering into the Hercules Credit Facility, as described further below, the maturity dates of the 2009 Secured Notes was extended to the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. Also in connection with entering into the Hercules Credit Facility, the holders of the 2010 Secured Notes entered into a subordination agreement with Hercules pursuant to which the 2010 Secured Notes were subordinated to the Hercules Credit Facility. Previously, in connection with GECC Credit Facility, as further described below, 2010 Secured Noteholders had entered into an inter-creditor agreement with the holders of the 2009 Convertible Notes and the 2009 Secured Notes whereby the 2009 Convertible Notes were subordinated to the 2009 Secured Notes and the 2010 Secured Notes, and the holders of the 2009 Secured Notes agreed to share payments pro rata with the holders of the 2010 Secured Notes.
The outstanding principal and accrued interest on the 2010 Secured Notes was $15.5 million as of December 31, 2010 and interest expense associated with these notes was $0.5 million for the year ended December 31, 2010.
HBM Term Loan
On April 30, 2010, the Company entered into a subordinated secured note purchase agreement with entities affiliated with HBM BioVentures, or HBM, to issue $3.75 million in aggregate principal amount of secured notes, or the HBM Secured Notes, in a private placement. Pursuant to the purchase agreement for the HBM Secured Notes, upon written notice delivered to HBM prior to September 30, 2010, HBM purchased an amount of secured notes set forth in the notice. HBM purchased the entire $3.75 million of the HBM Secured Notes in three closings in April, June and September 2010. To secure the performance of its obligations under the purchase agreement for the HBM Secured Notes, the Company granted a subordinated security interest in substantially all of its assets, including its intellectual property assets, other than the assets that secure its obligations under its agreement with Paul Capital. The HBM Secured Notes carry an interest rate of approximately 10% per year. In addition, the HBM Secured Notes require a final payment fee if they are prepaid prior to the maturity date. The maturity date of the HBM Secured Notes is the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated. On November 24, 2010, the holders of the HBM Secured Notes entered into a subordination agreement with Hercules pursuant to which the HBM Secured Notes were subordinated to the Hercules Credit Facility.
The outstanding principal and accrued interest on the HBM Secured Notes was $3.9 million as of December 31, 2010 and interest expense associated with these notes was $0.2 million for the year ended December 31, 2010.
CREDIT FACILITIES
GECC Credit Facility
In April 2010, The Company entered into a credit facility with General Electric Capital Corporation (the “GECC Credit Facility”), with $11.25 million available for borrowing. The Company borrowed an aggregate principal amount of $5.62 million at the closing, $2.81 million on July 1, 2010 and the remaining $2.81 million on
F-20
Table of Contents
September 2, 2010. Each of the term loans under the GECC Credit Facility carried a fixed interest rate of approximately 10% that was payable monthly. The GECC Credit Facility required no payment of principal for the first year, and then equal principal payments over 24 months until the maturity dates of 3 years from the funding dates. The GECC Credit Facility was secured by a first priority lien on all of the Company’s assets other than the assets that secure its obligations under its agreement with Paul Capital, and was guaranteed in full by certain majority investors of the Company (the “guarantors”).
In connection with any prepayments of term loans under the GECC Credit Facility, the Company was required to pay, in addition to all principal and accrued and unpaid interest on such term loan, a final payment fee equal to (i) 0.45% of the original principal amount of such term loan if the prepayment was made or required before the one year anniversary of such term loan, (ii) 2.25% of the original principal amount of such term loan if the prepayment was made or required on or after the one year anniversary of such term loan but before the two year anniversary of such term loan, and (iii) 3.50% of the original principal amount of such term loan if the prepayment was made or required on or after the two year anniversary of such term loan.
The GECC Credit Facility was guaranteed by the Company and was secured by a first priority lien on all of the assets of both PPI-California and the guarantors, other than the assets that secure its obligations under its agreement with Paul Capital. In addition, the GECC Credit Facility was guaranteed by certain of the Company’s investors (other than HBM) on a several and not joint basis which guarantee was limited to each investor’s pro rata portion of the outstanding principal and accrued and unpaid interest under the GECC Credit Facility, but in no event to exceed $11.25 million in the aggregate. The obligations of the investors under the guarantee is not triggered until the earlier to occur of (i) thirty days after written notice from the agent that the obligations under the GECC Credit Facility have been accelerated, and (ii) the occurrence of a bankruptcy or insolvency event with respect to the borrower, the Company or any of the investor guarantors. The guarantee by the Company’s investors of the GECC Credit Facility also included covenants that required each such investor to maintain at all times unfunded commitments from its investors in an amount equal to at least one and one-half times the maximum amount that the investor would have been obligated for under the investor guarantee, and also included certain control requirements with respect to such investors.
The GECC Loan and Security Agreement contained events of default including payment default, default arising from the breach of the provisions of the GECC Loan and Security Agreement and related documents or the inaccuracy of representations and warranties, attachment default, judgment default, bankruptcy and insolvency, a cross-default provision with respect to other material indebtedness, default based on the unenforceability, invalidity or revocation of a the GECC Loan and Security Agreement or any other related documents (including any guarantee or applicable subordination agreement) or any security interests, the occurrence of a material adverse effect (as defined in the GECC Loan and Security Agreement) and certain changes in control, including if the chief executive officer or chief financial officer of the borrower cease to be involved in the daily operations or management of the business, if certain holders cease to own or control at least 51% of the outstanding capital stock of the Company, if the Company ceased to own or control all the economic and voting rights of the borrower and if the borrower ceased to own or control, directly or indirectly, all of the economic or voting rights of each of its subsidiaries.
The occurrence of an event of default under the GECC Credit Facility could have triggered the acceleration of the obligations under the GECC Credit Facility or allowed the agent or lenders to exercise other rights and remedies, including rights against the assets which secured the GECC Credit Facility and rights under guarantees provided to support the obligations under the GECC Credit Facility.
The GECC Loan and Security Agreement contained a number of affirmative and restrictive covenants including reporting requirements, compliance with laws, protection of intellectual property and other collateral covenants, and limitations, subject to certain exceptions set forth in the GECC Loan and Security Agreement, on liens and indebtedness, limitations on dispositions, limitations on mergers and acquisitions, limitations on restricted payments and investments, limitations on transactions with the Company’s affiliates, limitations on changes in business, limitations on amendments and waivers of certain agreements, and limitations on waivers and amendments to certain agreements, including certain portions of the Paul Capital agreements, the Company’s organizational documents, and documents relating to debt that is subordinate to the Company’s obligations under the GECC Credit Facility.
F-21
Table of Contents
On November 24, 2010, the GECC Credit Facility was repaid in full, from the proceeds of the term loan under the Hercules Credit Facility, as further described below. The Company incurred a loss on the extinguishment of debt of approximately $184,000. Interest expense associated with the facility was $0.3 million for the year ended December 31, 2010.
Hercules Credit Facility
On November 24, 2010, the Company entered into a $26.25 million credit facility with Hercules Technology Growth Capital, Inc. and Hercules Technology III, L.P., as lenders (the “Hercules Credit Facility”). At the closing of the Hercules Credit Facility, the Company entered into a term loan in the aggregate principal amount of $26.25 million, which was the full amount available under the Hercules Credit Facility. As of December 31, 2010, the entire term loan of $26.25 million was outstanding. The term loan under the Hercules Credit Facility is comprised of two tranches, Tranche A and Tranche B. The Tranche A portion of the term loan is comprised of $11.25 million in principal and carries a floating per annum interest rate equal to 10.25% plus the amount, if any, by which the prime rate exceeds 4.00%. Upon the release of the investors’ guaranty (described below), the interest rate on the Tranche A portion of the term loan will increase to a floating per annum interest rate equal to 11.00% plus the amount, if any, by which the prime rate exceeds 4.00%. The Tranche B portion of the term loan is comprised of $15.0 million in principal and carries a floating per annum interest rate equal to 12.65% plus the amount, if any, by which the prime rate exceeds 4.00%. As of December 31, 2010, the interest rate on the Tranche A portion was 10.25% and on the Tranche B portion was 12.65%. Interest on the term loan is payable monthly. If there is an event of default under the Hercules Credit Facility, the Company will be obligated to pay interest at a higher default rate. The proceeds of the term loan under the Hercules Credit Facility have been used to repay the GECC Credit Facility in full and will be used for other general corporate purposes.
As further consideration to the lenders to provide the term loan to the Company under the Hercules Credit Facility, the Company issued to the lenders a warrant to purchase 178,986 shares of the Company’s Series A convertible preferred stock. If after the closing date of the Hercules Credit Facility and prior to the completion of the Company’s initial public offering, the Company issues equity securities in a private placement then the lenders may, at their option, exercise the warrant for the same class and type of equity securities that the Company issues in such private placement in lieu of Series A convertible preferred stock. The exercise price for the shares to be issued under the warrant is equal to the lower of $13.44 per share or the price per share paid in the next private placement. The warrant is valid from the date of issuance until the earlier to occur of ten (10) years from the date of issuance or five (5) years following the effective date of a registration statement for an initial public offering. As further described in Note 18, the Company has completed an initial public offering on February 8, 2011 and no private placement occurred prior to its completion.
The Hercules Credit Facility provides for an “interest only period” when no principal amounts are due and payable. The interest only period runs initially from November 24, 2010 through August 31, 2011, but can be extended, at the Company’s request, to either November 30, 2011 or February 28, 2012 if certain conditions are satisfied. Following the end of the interest only period, the term loan is to be repaid in 33 equal monthly installments of principal and interest beginning on the first business day after the month in which the interest only period ends. Amounts repaid may not be re-borrowed. The Company can, at any time, prepay all or any part of the term loan provided that so long as the investors’ guaranty (as described below) is in effect, the Company cannot prepay any part of the Tranche A portion of the term loan without the lenders’ consent if any of the Tranche B portion is outstanding. If the investors’ guaranty is not in effect, then any prepayments are to be applied pro rata across the outstanding balance of both portions of the term loan. In connection with any prepayments of the term loan under the Hercules Credit Facility, the Company is required to pay, in addition to all principal and accrued and unpaid interest on such term loan, a prepayment charge equal to 1.25% of the principal amount being prepaid. In addition, there is an end of term charge that is payable to the lenders upon the earliest to occur of the maturity date, the prepayment in full of the Company’s obligations under the Hercules Credit Facility and the acceleration of the Company’s obligations under the Hercules Credit Facility.
The Hercules Credit Facility is secured by a first priority lien on all of the Company’s assets other than the assets that secure the Company’s obligations under the Amended and Restated Royalty Interests Assignment Agreement (as described below). In addition, the Hercules Credit Facility is guaranteed by certain of the Company’s investors (other than entities affiliated with HBM) on a several and not joint basis, which guarantee is limited to
F-22
Table of Contents
each investor’s pro rata portion of the outstanding principal and accrued and unpaid interest under the Hercules Credit Facility, but in no event exceeding $11.25 million in the aggregate. The Hercules loan agreement, provides that upon the occurrence of certain circumstances and upon the Company’s request, the investors’ guarantee may be terminated and released.
The Hercules loan and security agreement also contains a provision that entitles the lenders to, subject to applicable securities laws and regulatory requirements, a limited right to participate in any equity financings that occur between the closing date of the Hercules Credit Facility and the completion of the Company’s initial public offering.
The Hercules loan and security agreement contains events of default including payment default, default arising from the breach of the provisions of the Hercules loan and security agreement and related documents (including the occurrence of certain changes in control, including if the Company’s chief executive officer ceases under certain conditions to be involved in the daily operations or management of the business, or if certain holders of the Company’s capital stock cease to retain, after the consummation of certain corporate transactions, shares representing more than 50% of the surviving entity after such transactions (provided that the Company’s initial public offering shall not constitute such a change in control)) or the inaccuracy of representations and warranties contained in the loan and security agreement, attachment default, bankruptcy and insolvency, cross-default with respect to certain other indebtedness (including certain events under the Amended and Restated Royalty Interests Assignment Agreement), breach of the terms of any guarantee (including the investors’ guarantee) of the Hercules Credit Facility, the occurrence of a material adverse effect (as defined in the Hercules loan and security agreement).
The occurrence of an event of default under the Hercules Credit Facility could trigger the acceleration of the Company’s obligations under the Hercules Credit Facility or allow the lenders to exercise other rights and remedies, including rights against the Company’s assets that secure the Hercules Credit Facility and rights under guarantees provided to support the obligations under the Hercules Credit Facility.
The Hercules loan and security agreement contains a number of affirmative and restrictive covenants, including reporting requirements and other collateral limitations, certain limitations on liens and indebtedness, dispositions, mergers and acquisitions, restricted payments and investments, corporate changes and waivers and amendments to certain agreements, the Company’s organizational documents, and documents relating to debt that is subordinate to the Company’s obligations under the Hercules Credit Facility.
In connection with entering into the Hercules Credit Facility, the maturity dates of the 2009 Convertible Notes, the 2009 Secured Notes and the 2010 Secured Notes were extended to the earliest of (1) a sale of the Company, (2) the date which is 30 days after the last day of the month that is 33 months after the expiration of the “interest only period” under the Hercules Credit Facility (as described above) and (3) 91 days after the date that all obligations under the Hercules Credit Facility are paid in full and the Hercules Credit Facility is terminated.
In connection with entering into the Hercules Credit Facility, the holders of the 2010 Convertible Notes entered into a subordination and intercreditor agreement with the lenders under the Hercules Credit Facility pursuant to which the 2010 Convertible Notes were subordinated to the Hercules Credit Facility. The holders of the 2010 Convertible Notes previously entered into a separate intercreditor agreement with the holders of the 2010 Secured Notes and the 2010 Secured Notes pursuant to which the 2010 Convertible Notes were subordinated to the 2010 Secured Notes and the 2010 Secured Notes, and the holders of the 2010 Secured Notes agreed to share payments pro rata with the holders of the 2010 Secured Notes.
The end of term charge of approximately $0.6 million has been recorded in accrued expenses and as a discount to the Hercules Credit Facility and amortized as a component of interest expense over the original term. The warrants, valued at approximately $0.6 million, have also been recorded as a discount to the Hercules Credit Facility and amortized as a component of interest expense over the original term. For the year ended December 31, 2010, the combined amortization of the discount was $32,000. The financing costs of approximately $0.5 million were capitalized and are being amortized as a component of interest expense over the original term.
F-23
Table of Contents
The outstanding principal and accrued interest on the Hercules Credit Facility was $26.5 million as of December 31, 2010, and interest expense associated with the facility was $0.3 million for the year ended December 31, 2010.
Sale of Royalty Interests
In 2000, PPI-California and SkyePharma PLC entered into a Royalty Interests Assignment Agreement (“PLC Royalty Agreement”) with an affiliate of Paul Capital Advisors, LLC (“Paul Capital”) to raise $30 million. Under the PLC Royalty Agreement, Paul Capital had the right to receive a royalty interest in four of SkyePharma’s product sales including product sales of and other payments related to DepoCyt(e) and DepoDur. Payments began for product sales realized on or after January 1, 2003 and continue through December 31, 2014.
In connection with the Acquisition, the PLC Royalty Agreement was amended (“Amended and Restated Royalty Interests Assignment Agreement”). As part of this amendment the responsibility to pay the royalty interest in product sales of DepoCyt(e) and DepoDur were transferred to the Company and the payment to Paul
Capital in a “Purchase Option Event” of the Company, as described below, was defined. The net present value of royalties expected to be repaid to Paul Capital (the “royalty interest obligation”) was valued at $13.0 million.
The Company recorded the royalty interest obligation as a liability in the Company’s consolidated balance sheets in accordance with ASC 470-10-25, Sales of Future Revenues. The Company imputes interest expense associated with this liability using the effective interest rate method. The effective interest rate may vary during the term of the agreement depending on a number of factors including the actual sales of DepoCyt(e) and DepoDur and a significant estimation, performed quarterly, of certain of the Company’s future cash flows related to these products during the remaining term of the Royalty Interests Assignment Agreement which terminates on December 31, 2014. Any adjustment to the estimates is reflected in the Company’s consolidated statements of operations as interest income (expense). In addition, such cash flows are subject to foreign exchange movements related to sales of DepoCyt(e) and DepoDur denominated in currencies other than U.S. dollars.
The PLC Royalty Agreement also includes a provision for a “Purchase Option Event.” The events include: (1) any change of control, a direct or indirect consequence of which is a material abatement of efforts to develop, market or sell any of the products or reformulated products; or (2) the transfer by the parent of all or substantially all of the parent’s consolidated assets; or (3) the transfer by the Company of all or any part of their respective interests in the products or reformulated products, or (4) bankruptcy or other breach or default under the agreement.
In the event a Purchase Option Event occurs, Paul Capital shall have the right, but not the obligation, exercisable within 90 days, to require the Company to repurchase from Paul Capital the Royalty Interests Assignment, for a repurchase price equal to 50% of the cumulative amount of all payments made during the preceding 24 months (calculated from the date of the Purchaser’s receipt of the notice from the Company of the Purchase Option Event) multiplied by the number of days from the date of Paul Capital’s exercise of such option until December 31, 2014, divided by 365.
The Company has no minimum payment obligations under the PLC Royalty Agreement. However, the repayment of the Paul Capital liability is supported through a jointly controlled lockbox, where all DepoCyt(e) and DepoDur supply revenue and royalties are received. Commencing April 1 of every year, the first $2.5 million received in the lockbox is restricted and will be used to make quarterly payments due to Paul Capital, if any, under the agreement during the subsequent 12 month period. On March 31 of the subsequent year, the balance of cash in the lockbox, if any, is remitted to Pacira. The PLC Royalty Agreement terminates on December 31, 2014. The PLC Royalty Agreement pertains only to DepoCyt(e) and DepoDur, and does not include revenue related to EXPAREL or any other product candidates. $1.3 million and $1.2 million was in the lockbox and included in restricted cash in the Company’s consolidated balance sheets as of December 31, 2010 and 2009, respectively.
F-24
Table of Contents
11. STOCKHOLDERS’ EQUITY
Common Stock
In connection with its formation, the Company issued in March 2007 an aggregate of 464,900 shares of common stock for total aggregate consideration of $50,000.
Series A Convertible Preferred Stock
In March 2007, the Company entered into a Series A Preferred Stock Purchase Agreement pursuant to which the Company issued and sold an aggregate of 6,322,640 shares of Series A convertible preferred stock in four separate closings held in March 2007, February 2008, July 2008 and October 2008, at a purchase price of $13.44 per share. The aggregate consideration for the shares of Series A convertible preferred stock was $85 million in cash.
Each holder of Series A convertible preferred stock has the right, at the option of the holder at any time, to convert their shares of Series A convertible preferred stock into shares of common stock at a current conversion ratio of one-to-one, subject to adjustment for stock splits, certain capital reorganizations and dilutive stock issuances. Each share of the Company’s Series A convertible preferred stock will automatically convert into shares of the Company’s common stock, at the then effective applicable conversion ratio upon the earlier of: (i) the closing of the sale of the Company’s common stock pursuant to a firmly underwritten public offering in which the Company receives gross proceeds of at least $25 million or (ii) the consent of the holders of at least 66 2/3% of the then outstanding shares of Series A convertible preferred stock.
The holders of Series A convertible preferred stock are entitled to receive, when, as and if declared by the Company’s board of directors out of legally available funds, non-cumulative dividends in an amount equal to any dividends declared, paid or set aside on shares of the Company’s common stock. As of December 31, 2010, no dividends have been declared by the Company’s board of directors.
In the event of any liquidation, dissolution or winding up of the Company, the holders of the Series A convertible preferred stock will be entitled to receive in preference to the holders of the Company’s common stock, the amount of their original purchase price per share, plus declared and unpaid dividends, if any. If the assets and funds available to be distributed among the holders of the Series A convertible preferred stock are insufficient to permit the payment to such holders of the full preference, then the entire assets and funds legally available for distribution to such holders shall be distributed ratably based on the total due each holder of the Series A convertible preferred stock. Any remaining assets of the Company will be distributed ratably among the holders of its common stock.
Holders of the Series A convertible preferred stock are entitled to the number of votes they would have upon conversion of their Series A convertible preferred stock into common stock at the then-applicable conversion rate. The holders of Series A convertible preferred stock have been granted certain rights with regard to the election of board members and various other corporate actions.
Warrants
On January 22, 2009, the Company issued warrants in connection with the issuance of the 2009 Convertible Notes (see Note 10). The warrants are convertible into an aggregate of 158,061 of shares of the Company’s common stock at an exercise price of $2.69 per share and will expire on January 21, 2014. The value of the warrants has been recorded as a discount to the 2009 Convertible Notes and amortized as a component of interest expense over the original term of the 2009 Convertible Notes. For the year ended December 31, 2009, the amortization of the discount was $269,000 resulting in no remaining balance as of December 31, 2010 and 2009.
On July 2, 2009, the Company issued warrants to the landlord of the Company’s two San Diego facilities in connection with amendments to the respective lease agreements that deferred minimum annual rental obligations (see Note 13). The warrants are exercisable for an aggregate of 23,244 shares of Series A convertible preferred stock at a price of $13.44 per share and will expire on the earlier of July 1, 2016 or the fifth anniversary of the consummation of the Company’s initial public offering. The value of the warrants was recorded as prepaid interest and is being amortized as a component of interest expense over the deferred rental payment term. For the years ended December 31, 2010 and 2009, the amortization of the interest was $114,000 and $63,000, respectively, resulting in a balance of $27,000 and $141,000 as of December 31, 2010 and 2009, respectively.
F-25
Table of Contents
On November 24, 2010, the Company issued warrants in connection with the Hercules Credit Facility to the lenders to purchase 178,986 shares of the Company’s Series A convertible preferred stock (see Note 10). The warrants are exercisable at a price of $13.44 per share and shall be valid from the date of issuance until the earlier to occur of ten (10) years from the date of issuance or five (5) years following the effective date of the registration statement for an initial public offering. The warrants, valued at approximately $0.6 million, have been recorded as a discount to the Hercules Credit Facility and amortized as a component of interest expense over the original term. For the year ended December 31, 2010, the amortization of the discount was $17,000.
On December 29, 2010, the Company issued warrants in connection with the December 2010 Convertible Notes (see Note 10). The warrants are convertible into an aggregate of 167,361 of shares of the Company’s common stock with an exercise price of $13.44 per share and will expire on December 29, 2017. The warrants, valued at approximately $0.5 million, have been recorded as a discount to the 2010 Convertible Notes and will be amortized as a component of interest expense over the original term of the 2010 Convertible Notes. Upon the completion of the Company’s initial public offering, the December 2010 Convertible Notes will be converted into common stock, and any unamortized balance will be recognized in full on the date of such event (see Note 18).
Share-Based Compensation
The Company recognized share-based compensation in its consolidated statements of operations for the years ended December 31, 2010, 2009 and 2008 as follows:
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (in thousands) | ||||||||||||
| Selling, general and administrative |
$ | 1 | $ | 349 | $ | 126 | ||||||
| Research and development |
22 | 175 | 116 | |||||||||
| $ | 23 | $ | 524 | $ | 242 | |||||||
Pacira Stock Incentive Plan
Employees and directors have been granted options to purchase common shares under the 2007 Stock Option/Stock Issuance Plan (the “2007 Plan”). The original 2007 Plan provided for the grant of options to purchase up to 650,860 shares of the Company’s common stock. Options granted under the 2007 Plan generally expire no later than ten years from the date of grant. The exercise price of incentive stock options must be equal to at least the fair value of the Company’s common stock on the date of grant.
The 2007 Plan was amended in April 2008, to, among other things, increase the number of shares of common stock authorized for issuance under the 2007 Plan from 650,860 shares to 1,066,946 shares.
On September 2, 2010, the 2007 Plan was amended again to increase the number of authorized plan shares from 1,066,946 to 1,729,498 shares of common stock. Concurrent with the amendment of the 2007 Plan, in September 2010 the board of directors granted stock options to employees, non-employee board members and consultants for an aggregate of 1,448,301 shares of common stock. The stock options have an exercise price of $1.61 per share. In establishing the exercise price, the board of directors relied on a valuation that concluded as of August 31, 2010 the value of the Company’s common stock was $1.61 per share.
These stock options may be exercised only upon the completion of an initial public offering prior to December 31, 2012. If an initial public offering is not completed prior to December 31, 2012, then the options automatically cancel (see Note 18). The stock options have a 10-year term, and the option shares vest according to one of the following four schedules:
F-26
Table of Contents
(i) 75% of the option shares vest on the date of grant, and the remaining 25% of the option shares vest in equal successive monthly installments upon the optionee’s completion of each month of service over the 12 month period following the date of grant;
(ii) 50% of the option shares vest on the date of grant, and the remaining 50% of the option shares vest in equal successive monthly installments upon the optionee’s completion of each month of service over 24 month period following the date of grant;
(iii) 25% of the option shares vest upon optionee’s completion of one year of service to the Company measured from the date of grant, and the remaining 75% of the option shares vest in equal successive monthly installments upon the optionee’s completion of each month of service over the 36 month period following the first anniversary of the date of grant; or
(iv) 50% of the option shares vest on the first anniversary of the closing of the Company’s initial public offering provided that the optionee remains in service to the Company for such first year and, the remaining 50% of the option shares vest on the second anniversary of the closing of the Company’s initial public offering provided that the optionee remains in service to the Company over such second year. Upon a change in control of the Company, as defined in the 2007 Plan, 100% of the shares underlying each of these options shall become vested and exercisable immediately prior to such change in control.
In December 2010, the Company’s board of directors granted options to all of its employees, including its named executive officers, and its non-employee directors, for an aggregate of 571,300 shares of common stock. These stock options may be exercised only upon the completion of an initial public offering prior to December 31, 2012. If an initial public offering is not completed prior to December 31, 2012, then the options automatically cancel.
F-27
Table of Contents
The following table summarizes the Company’s stock option activity and related information for the period from January 1, 2008 to December 31, 2010:
| Shares | Weighted Average Exercise Price |
Weighted Average Remaining Term (years) |
Aggregate Intrinsic Value (in thousands) |
|||||||||||||
| Outstanding at January 1, 2008 |
558,141 | 1.61 | ||||||||||||||
| Granted |
454,110 | 1.96 | ||||||||||||||
| Exercised |
(107,264 | ) | 1.61 | |||||||||||||
| Forfeited |
(114,064 | ) | 1.64 | |||||||||||||
| Expired |
(1,546 | ) | 1.61 | |||||||||||||
| Outstanding at December 31, 2008 |
789,377 | 1.81 | ||||||||||||||
| Granted |
741 | 2.69 | ||||||||||||||
| Exercised |
(1,756 | ) | 1.61 | |||||||||||||
| Forfeited |
(655,350 | ) | 1.84 | |||||||||||||
| Expired |
(80,582 | ) | 1.61 | |||||||||||||
| Outstanding at December 31, 2009 |
52,430 | 1.79 | ||||||||||||||
| Granted |
2,028,158 | 2.71 | ||||||||||||||
| Exercised |
(1,177 | ) | 1.89 | $ | 1 | |||||||||||
| Forfeited |
(3,337 | ) | 1.89 | |||||||||||||
| Expired |
(2,374 | ) | 1.75 | |||||||||||||
| Outstanding at December 31, 2010 |
2,073,700 | $ | 2.69 | 9.7 | $ | 5,800 | ||||||||||
| Exercisable at December 31, 2010 |
46,982 | $ | 1.97 | 7.5 | $ | 165 | ||||||||||
| Vested and expected to vest at December 31, 2010 |
1,906,815 | $ | 2.64 | 9.7 | $ | 5,422 | ||||||||||
The weighted average fair value of options granted for the years ended December 31, 2010 and 2009 were $5.61 and $1.94 per share, respectively. The total fair value of options which vested during 2010 and 2009 was approximately $28,000 and $0.1 million, respectively.
As of December 31, 2010, 363,814 shares of common stock were reserved for future grant of stock options. As of December 31, 2010, $3.9 million of total unrecognized compensation cost related to non-vested stock options is expected to be recognized over the respective vesting terms of each award. The weighted average term of the unrecognized share-based compensation is 3.30 years. As further described in Note 15, unexercised options to purchase an aggregate of 477,820 shares of common stock options were cancelled during 2009, which resulted in share-based compensation of $0.5 million.
The fair values of each option grant in 2010, 2009 and 2008 were estimated using the Black-Scholes option pricing model with the following weighted average assumptions:
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| Expected dividend yield |
None | None | None | |||||||||
| Risk free interest rate |
1.6-3.4 | % | 2.1-2.7 | % | 1.9-3.8 | % | ||||||
| Expected volatility |
80.8 | % | 82.0 | % | 78.2 | % | ||||||
| Expected life of options |
6.25 years | 6.25 years | 6.25 years | |||||||||
F-28
Table of Contents
12. COST OF REVENUES
Cost of revenues consists of the following:
| Years Ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (In thousands) | ||||||||||||
| Cost of supply revenue |
$ | 11,031 | $ | 9,828 | $ | 14,467 | ||||||
| Cost of royalties |
343 | 401 | 567 | |||||||||
| Cost of collaborative licensing and development revenue |
902 | 2,072 | 2,429 | |||||||||
| Total cost of revenues |
$ | 12,276 | $ | 12,301 | $ | 17,463 | ||||||
Cost of supply revenue consists of the manufacturing and allocated overhead costs related to the Company’s supply of DepoCyt(e) and DepoDur to its commercial partners. Cost of royalties consists of payments to Research Development Foundation (“RDF”) for the use of DepoFoam technology. Cost of collaborative licensing and development revenue consists of the Company’s expenses related to feasibility studies and development work for third parties who desire to utilize the Company’s DepoFoam extended release drug delivery technology for their products.
13. COMMITMENTS AND CONTINGENCIES
Leases
The Company leases office, research and development, and manufacturing facilities in San Diego, California. The two facilities in San Diego are comprised of the Science Center location and the Torrey Pines location. The leases for both these facilities expire July 2015. Under these leases, the Company is required to pay certain maintenance expenses in addition to the monthly rent. Rent expense is recognized on a straight-line basis over the lease term for leases that have scheduled rent increases. During 2009, the Company entered into amendments to its real estate leases for the Science Center and Torrey Pines facilities. As part of the lease amendments, the property-owner agreed to defer a portion of the minimum annual rent obligation due from February 1, 2009 to March 31, 2010 in exchange for interest compounded at 10% per annum, and warrants to purchase 23,244 shares of Series A convertible preferred stock with values totaling $141,000 and $63,000 on the Science Center and Torrey Pines facilities, respectively. The total amount of rent deferred will be $438,414 and $2,109,101 for the Torrey Pines and Science Center facilities, respectively. The amounts are to be repaid from April 1, 2010 to September 1, 2011. The warrants are convertible into Series A convertible preferred stock with an exercise price of $13.44 per share and will expire on the earlier of July 1, 2016 or the fifth anniversary of the consummation of the Company’s initial public offering. The value of the warrants has been recorded as prepaid interest and is being amortized over the deferred rental payment term. As of December 31, 2010 and 2009, the balance of the related prepaid interest was $27,000 and $141,000, respectively. For the year ended December 31, 2010, the additional interest associated with the deferred payments and amortization of the warrants was $79,000 and $ 36,000, respectively.
The Company determined that its lease rates associated with the assumed the Torrey Pines and Science Center facilities’ leases were in excess of market rates resulting in a $3.3 million unfavorable lease accrual as of the Acquisition Date. The unfavorable lease accrual, which is recorded in other long-term liabilities in the Company’s consolidated balance sheets, is amortized over the remaining terms of the leases. The balance of the unfavorable lease accrual as of December 31, 2010 and 2009 was $1.8 million and $2.2 million, respectively. The annual amortization of the unfavorable lease accrual for 2010, 2009 and 2008 was $0.4 million.
As of December 31, 2010, annual minimum payments due under the Company’s office and equipment lease obligations are as follows (in thousands):
| 2011 |
$ | 5,827 | ||
| 2012 |
4,820 | |||
| 2013 |
4,968 | |||
| 2014 |
5,136 | |||
| 2015 |
3,072 | |||
| Total |
$ | 23,823 | ||
F-29
Table of Contents
Total rent expense, net of unfavorable lease obligation amortization, under all operating leases for years ended December 31, 2010, 2009 and 2008 was $4.5 million, $4.6 million and $4.6 million, respectively. Deferred rent at December 31, 2010 and 2009 was $1.3 million and $1.2 million, respectively.
Litigation
The Company periodically becomes subject to legal proceedings and claims arising in connection with its business. The ultimate legal and financial liability of the Company in respect to all claims, lawsuits and proceedings cannot be estimated with any certainty. Any outcome, either individually or in the aggregate, is not expected to be material to the Company’s consolidated financial position, results of operations, or cash flows.
14. INCOME TAXES
A reconciliation of income taxes at the U.S. Federal statutory rate to the provision for income taxes is as follows (in thousands):
| Year ended December 31, | ||||||||||||
| 2010 | 2009 | 2008 | ||||||||||
| (in thousands) | ||||||||||||
| Benefit at U.S. Federal statutory rate |
$ | (9,548 | ) | $ | (10,901 | ) | $ | (14,887 | ) | |||
| State taxes—deferred |
(2,115 | ) | (1,713 | ) | (1,844 | ) | ||||||
| Increase in valuation allowance |
12,213 | 12,916 | 17,417 | |||||||||
| Tax credits |
(50 | ) | (498 | ) | (1,319 | ) | ||||||
| Other |
(500 | ) | 196 | 633 | ||||||||
| Provision for income taxes |
$ | — | $ | — | $ | — | ||||||
Significant components of the Company’s deferred tax assets are as follows:
| Year ended December 31, | ||||||||
| 2010 | 2009 | |||||||
| (in thousands) | ||||||||
| Deferred tax assets: |
||||||||
| Federal and state net operating loss carry-forwards |
$ | 44,751 | $ | 32,321 | ||||
| Federal and state research credits |
2,828 | 2,778 | ||||||
| Depreciation and amortization |
2,563 | 1,090 | ||||||
| Accruals and reserves |
7,781 | 8,632 | ||||||
| Deferred revenue |
8,439 | 9,302 | ||||||
| Other |
306 | 332 | ||||||
| Total gross deferred tax assets |
66,668 | 54,455 | ||||||
| Less valuation allowance for deferred tax assets |
(66,668 | ) | (54,455 | ) | ||||
| Net deferred tax assets |
$ | — | $ | — | ||||
The valuation allowance for deferred tax assets increased by approximately $12.2 million, $12.9 million and $17.4 million during the years ended December 31, 2010, 2009 and 2008, respectively. Management believes the significant doubt regarding the realization of net deferred tax assets requires a full valuation allowance.
As of December 31, 2010, the Company had Federal and state net operating losses of approximately $111.8 and $97.7 million, respectively. The Company also had Federal and state research and development tax credit carry-forwards of approximately $2.5 and $1.1, respectively. The net operating loss carry-forwards and tax credits will expire at various dates, beginning in 2020, through 2031, if not utilized.
F-30
Table of Contents
The Tax Reform Act of 1986 limits the use of net operating loss and tax credit carry-forwards in certain situations where changes occur in the stock ownership of a company. In the event the Company has a change in ownership in the future, as defined by the tax law, utilization of the carry-forwards could be limited.
The Company follows new accounting principles on accounting for uncertain tax positions. Under these principles, tax positions are evaluated in a two-step process. The Company first determines whether it is more-likely-than-not that a tax position will be sustained upon examination. If a tax position meets the more-likely-than-not recognition threshold it is then measured to determine the amount of benefit to be recognized in the financial statements. The tax position is measured as the largest amount of benefit that has a greater than 50 percent likelihood of being realized upon ultimate settlement.
The Company did not have a liability related to unrecognized tax benefit as of December 31, 2010 and 2009 due to operating losses but has reduced its deferred tax assets by $476,000 and $420,000, respectively. Further, because the Company has recorded a full valuation allowance on its net deferred assets, the effect of implementing ASC 740 has been a reduction of the allowance by the amount above. A reconciliation of the beginning and ending amount of gross unrecognized tax benefit is as follows:
| Years Ended December 31, | ||||||||
| 2010 | 2009 | |||||||
| Balance at beginning of year |
$ | 420 | $ | 330 | ||||
| Increases related to tax positions taken during the current year |
56 | 90 | ||||||
| Increases related to tax positions taken during a prior period |
— | — | ||||||
| Balance at end of year |
$ | 476 | $ | 420 | ||||
No interest or penalties were accrued for 2010, 2009 or 2008.
The Company is currently open for audit by the United States Internal Revenue Service and state tax jurisdictions for 2006 through 2010.
15. RETIREMENT PLANS AND OTHER EMPLOYEE BENEFITS
Savings Plan
The Company sponsors a 401(k) savings plan. Under the plan, employees may make contributions to the plan, which are eligible for a discretionary percentage match as defined in the plan and determined by the board of directors. There was no compensation expense under the plan for years ended December 31, 2010, 2009 and 2008.
Incentive Bonus Plan
In March 2009, the Company adopted a company sale bonus plan and in March 2010 the Company amended and restated the company sale bonus plan. The company sale bonus plan provides for a potential cash bonus payment to specified employees and consultants, including executive officers, and non-employee directors, in the event of a sale of the Company. Under the company sale bonus plan, upon the closing of a sale transaction that satisfies specified criteria, each participant in the company sale bonus plan would receive either a bonus in an amount equal to a portion of the sale proceeds multiplied by a specified percentage for that participant or a fixed bonus payment. The plan terminates upon the completion of the Company’s initial public offering (see Note 18). As a condition to becoming participants under the plan, most of the participants, including all of the Company’s executive officers and non-employee directors, agreed to have their existing option grants cancelled. As a result, unexercised options for an aggregate of 477,820 shares of common stock were cancelled. In addition, certain
F-31
Table of Contents
employees were eligible to receive a retention bonus (equivalent to two weeks of base salary upon receipt of positive data on the EXPAREL Phase 3 clinical trials, or if the Company’s board of directors deemed related data to be positive) and a pre-determined percentage of salary in the event of a Company sale. In the fourth quarter of 2009, the Company received positive data on the EXPAREL Phase 3 clinical trials and, accordingly, recorded compensation expense and paid $0.1 million of retention bonuses.
In October 2010, the Company entered into employment agreements with its executive officers. Each of these agreements provides the executive officer with certain severance benefits in connection with certain terminations of the executive’s employment both before and after a change of control.
16. COMMERCIAL PARTNERS AND AGREEMENTS
Sigma -Tau
In December 2002, the Company entered into a supply and distribution agreement with Enzon Pharmaceuticals Inc. regarding the sale of DepoCyt. Pursuant to the agreement, Enzon was appointed the exclusive distributor of DepoCyt in the United States and Canada. In January 2010, Sigma-Tau Pharmaceuticals, Inc., or Sigma-Tau, acquired the rights to sell DepoCyt from Enzon Pharmaceuticals for the United States and Canada. Under the supply and distribution agreement, the Company supplies unlabeled DepoCyt vials to Sigma-Tau for finished packaging by Sigma-Tau. Under these agreements, the Company receives a fixed payment for manufacturing the vials of DepoCyt and a double-digit royalty on sales by Sigma-Tau in the United States and Canada.
Mundipharma International Holdings Limited
In June 2003, the Company entered into an agreement granting Mundipharma International Holdings Limited, or Mundipharma, exclusive marketing and distribution rights to DepoCyte in the European Union and certain other European countries. Under the agreement, as amended, and a separate supply agreement, the Company receives a fixed payment for manufacturing the vials of DepoCyte and a double-digit royalty on sales in the applicable territories by Mundipharma.
EKR Therapeutics Inc.
In August 2007, the Company entered into a licensing, distribution and marketing agreement with EKR Therapeutics, Inc., or EKR, granting them exclusive distribution rights to DepoDur in North America, South America and Central America. Under this agreement, as amended, the Company was entitled to receive non-refundable license fees of $5.0 million paid upon execution of the agreement in August 2007, $5.0 million paid at the end of 2008, and $5.0 million paid at the end of 2009. As noted above, the Company recognizes revenue from up-front license fees ratably over the performance period as determined under the agreement. The Company capitalized the up-front license fees into a deferred revenue liability, and amortizes the deferred revenue over a period of 15 years, which represents the contract period. Further, under the agreement, as amended, the Company receives a fixed payment for manufacturing the vials of DepoDur and a double-digit royalty on sales in the applicable territories by EKR.
Flynn Pharmaceuticals Limited
In September 2007, the Company entered into a marketing agreement with Flynn Pharma Limited, or Flynn, granting them exclusive distribution rights to DepoDur in the European Union, certain other European countries, South Africa and the Middle East. Under this agreement and a separate supply agreement with Flynn, the Company provides or procures DepoDur manufacturing supply of finished product for sale in the territories licensed by Flynn, and receives a fixed payment for manufacturing the vials of DepoDur and a double-digit royalty on sales in the applicable territories by Flynn.
F-32
Table of Contents
Amylin Pharmaceuticals Inc
In March 2008, the Company entered into a development and licensing agreement with Amylin Pharmaceuticals, Inc., or Amylin. Under the development and licensing agreement, the Company provides Amylin with access to its proprietary DepoFoam drug delivery technology to conduct research, feasibility and formulation work, and for the manufacturing of pre-clinical and clinical material for various Amylin products. The Company is entitled to payments from Amylin for its work on the formulation and development of compounds with the DepoFoam technology, its achievement of certain clinical development milestones, its achievement of certain worldwide sales and a tiered royalty based upon sales. In April 2008, the Company received a non-refundable up-front license fee of $8.0 million from Amylin. As noted above, the Company recognizes revenue from up-front license fees ratably over the performance period as determined under the agreement. The Company capitalized the up front license fee into a deferred revenue liability, and amortizes the deferred revenue over a period of approximately nine years. The development and licensing agreement with Amylin remains effective, however, neither party is currently performing any activities under the agreement.
Feasibility Study Agreements with Third Parties
In the ordinary course of its business activities, the Company enters into feasibility study agreements with third parties who desire access to its proprietary DepoFoam extended release drug delivery technology to conduct research, feasibility and formulation work. Under these agreements, the Company is compensated to perform feasibility testing on a third party product to determine the likelihood of developing a successful formulation of that product using its proprietary DepoFoam extended release drug delivery technology. If successful in the feasibility stage, these programs can advance to a full development contract.
17. RELATED PARTY TRANSACTIONS
During the years ended December 31, 2010 and 2009, the Company entered into 2009 Convertible Note Agreements, 2009 Secured Note Agreements, 2010 Secured Note Agreements and December 2010 Convertible Note Agreements, with certain investors in the Company (see Note 10). The composition of the balances due to these investors is $49.8 million, including accrued interest of $3.4 million, as of December 31, 2010.
In February 2008, the Company entered into a services agreement with Stack Pharmaceuticals Inc., or SPI, an entity controlled by David Stack, the Company’s chief executive officer. Pursuant to the agreement, SPI provides the Company with the use of SPI’s office facilities which include the use of office space for the Company’s employees, office furnishings, phone system, internet connections, printers and other related office amenities such as conference rooms. Pursuant to the agreement, the Company paid SPI amounts ranging from $10,500 to $18,250 per month during the term of the services agreement. The term of the agreement was one year and was renewable upon consent of both parties and the agreement may be cancelled with 60 days written notice by either party. In February 2009, we renewed the agreement on a month-to-month basis. In August 2010, we entered into a new services agreement with SPI that replaced the agreement that we entered into in February 2008. Pursuant to the new agreement, SPI provides us with the use of SPI’s office facilities which includes the use of office space for our employees, office furnishings, phone system, internet connections, printers and other related office amenities such as conference rooms. In addition, SPI provides consulting services and commercial leadership related to EXPAREL regarding the development of strategic plans and analyses for the commercialization of EXPAREL, support in the development of documents, data and materials for investor and commercial partner presentations and documents, and commercial leadership in support of our website. SPI provides these services from time to time as we request. We pay SPI $2,500 for each day of services provided by SPI up to a maximum of five days per week. We also reimburse SPI for travel expenses incurred by SPI personnel.
In addition, during 2009 and 2010, SPI performed various projects for the Company. These projects included a business analysis and commercial recommendation for the Company’s DepoDur product, a market research project related to the development of a DepoMethotrexate product, market research and forecasting in support of clinical development of EXPAREL for the potential additional indications of nerve block and epidural administration and reimbursement for access to Datamonitor reports for commercial analysis and partnering discussions regarding EXPAREL. The Company incurred expenses under the SPI agreement of $324,000, $210,000 and $258,000 for the years ended December 31, 2010, 2009, and 2008, respectively. As of December 31, 2010 and 2009, the Company had no outstanding balance payable to SPI.
F-33
Table of Contents
MPM Asset Management (“MPM”), an investor in the Company, provides clinical management and subscription services to the Company. The Company incurred expenses of $679,000, $316,000 and $30,000 for the years ended December 31, 2010, 2009 and 2008, respectively. $91,000 and $88,000 was payable to MPM as of December 31, 2010 and 2009, respectively.
In April 2010, the Company signed a statement of work for a feasibility study with Rhythm Pharmaceuticals, Inc. The Company earned $290,000 contract revenue from this statement of work during 2010. MPM and its affiliates are holders of the Company’s capital stock. MPM and its affiliates are holders of capital stock of Rhythm Pharmaceuticals, Inc. and a managing director of MPM is a member of the board of directors of Rhythm Pharmaceuticals, Inc.
18. SUBSEQUENT EVENTS
Initial Public Offering
In February 2011, we completed our initial public offering of our common stock pursuant to a registration statement on Form S-1, as amended (File No. 333-170245) that was declared effective on February 2, 2011. Under the registration statement, we registered the offering and sale of an aggregate of 6,900,000 shares of our common stock. An aggregate of 6,000,000 shares of common stock registered under the registration statement were sold at a price to the public of $7.00 per share. Barclays Capital Inc. and Piper Jaffray and Co. acted as joint book running managers of the offering and as representatives of the underwriters. The offering commenced on February 3, 2011 and closed on February 8, 2011. The over-allotment option was not exercised by the underwriters. As a result of our IPO, we raised a total of $42.0 million in gross proceeds, and approximately $37.0 million in net proceeds after deducting approximately $5.0 million in underwriting discounts and commissions and estimated offering expenses.
Upon the closing of the initial public offering, all outstanding Series A convertible preferred stock and the principal and accrued interest balance on the 2009 Convertible Notes, 2009 Secured Notes, 2010 Secured Notes, December 2010 Convertible Notes, and HBM Secured Notes were converted into 10,658,845 shares of common stock, as shown in the table below. Prior to the closing initial public offering, the second closing of the December 2010 Convertible Notes was not consummated.
| Conversion Shares | ||||
| Series A convertible preferred stock |
6,322,640 | |||
| 2009 Convertible Notes |
871,635 | |||
| 2009 Secured Notes |
927,881 | |||
| 2010 Secured Notes |
1,156,606 | |||
| HBM Secured Notes |
308,655 | |||
| December 2010 Convertible Notes |
1,071,428 | |||
2011 Plan
The Company’s 2011 stock incentive plan, or the 2011 Plan, which became effective immediately prior to the completion of the Company’s initial public offering, was adopted by its board of directors and approved by its stockholders in December 2010. The 2011 Plan provides for the grant of incentive stock options, non-statutory stock options, restricted stock awards and other stock-based awards. The sum of (up to 2,546,657 shares) (x) the number of shares of its common stock reserved for issuance under the 2007 Plan at such time, and (y) the number of shares of its common stock subject to awards granted under the 2007 Plan that expire, terminate or are otherwise surrendered, cancelled, forfeited or repurchased by the Company pursuant to a contractual repurchase right, are reserved for issuance under the 2011 Plan. In addition, the 2011 Plan contains an “evergreen” provision, which allows for an increase in the number of shares available for issuance under the 2011 Plan on the first day of each calendar year from 2012 through 2015.
F-34
Table of Contents
Novo Nordisk Development and License Agreement
In January 2011, the Company entered into an agreement with Novo Nordisk A/S, or Novo, pursuant to which it granted non-exclusive rights to Novo under certain of its patents and know-how to develop, manufacture and commercialize formulations of a Novo proprietary drug using the Company’s DepoFoam drug delivery technology. Under this agreement, the Company agreed to undertake specified development and technology transfer activities and to manufacture pre-clinical and certain clinical supplies of such DepoFoam formulated Novo product until the completion of such technology transfer activities. Novo is obligated to pay for all costs incurred by the Company in conducting such development, manufacturing and technology transfer activities. The Company received a one-time upfront payment of $1.5 million from Novo. The Company is also entitled to receive single-digit royalties on sales of such Novo product for up to twelve years following the first commercial sale of such Novo product. In addition, the Company is entitled to receive up to $24 million in milestone payments based on achievement of specified development events, and up to an additional $20 million in milestone payments based on sales of such Novo product exceeding specified amounts. Each party has the right to terminate the agreement for an uncured material breach by the other party or in connection with the other party’s bankruptcy or similar event. In addition, Novo has the right to terminate the agreement for convenience at any time upon sixty (60) days notice prior to commercialization of such Novo product and upon ninety (90) days notice thereafter, subject to Novo’s payment of a specified termination fee if, after initiation of the technology transfer but prior to commercialization, Novo terminates the agreement other than for certain specified reasons. The Company also has the right to terminate the agreement if (1) Novo decides to discontinue or terminate the development or commercialization of such Novo product, (2) such Novo product no longer has regulatory approval in any market, or (3) Novo or any of its affiliates or sublicensees of such Novo product challenges the validity or enforceability of any of the licensed patents.
F-35
Table of Contents
EXHIBIT INDEX
| Exhibit |
Exhibit Description |
Incorporated by Reference | Filed Herewith |
|||||||||||||||||||||
| Form | File Number |
Date of First Filing |
Exhibit Number |
|||||||||||||||||||||
| 3.1 | Amended and Restated Certificate of Incorporation of the Registrant | 8-K | 001-35060 | 02/11/2011 | 3.1 | |||||||||||||||||||
| 3.2 | Amended and Restated Bylaws of the Registrant | 8-K | 001-35060 | 02/11/2011 | 3.2 | |||||||||||||||||||
| 4.1 | Specimen Certificate evidencing shares of common stock | S-1/A | 333-170245 | 01/03/2011 | 4.1 | |||||||||||||||||||
| 10.1 | Second Amended and Restated 2007 Stock Option/Stock Issuance Plan | S-1 | 333-170245 | 11/01/2010 | 10.1 | |||||||||||||||||||
| 10.2 | Form of Stock Option Agreement under the Second Amended and Restated 2007 Stock Option/Stock Issuance Plan | S-1 | 333-170245 | 11/01/2010 | 10.2 | |||||||||||||||||||
| 10.3 | Investors’ Rights Agreement, dated March 23, 2007, among the Registrant and the parties named therein | S-1 | 333-170245 | 11/01/2010 | 10.3 | |||||||||||||||||||
| 10.4 | Assignment Agreement, dated February 9, 1994, amended April 15, 2004, between the Registrant and Research Development Foundation † | S-1/A | 333-170245 | 12/03/2010 | 10.4 | |||||||||||||||||||
| 10.5 | Stock Purchase Agreement, dated January 8, 2007, between SkyePharma, Inc. and the Registrant † | S-1/A | 333-170245 | 12/03/2010 | 10.5 | |||||||||||||||||||
| 10.6 | Amended and Restated Royalty Interests Assignment Agreement, dated March 23, 2007, as amended, between SkyePharma, Inc. and Royalty Securitization Trust I † | S-1/A | 333-170245 | 12/03/2010 | 10.6 | |||||||||||||||||||
| 10.7 | Amended and Restated Security Agreement (SKPI), dated March 23, 2007, between SkyePharma, Inc. and Royalty Securitization Trust I † | S-1/A | 333-170245 | 12/03/2010 | 10.7 | |||||||||||||||||||
| 10.8 | Supply Agreement, dated June 30, 2003, between SkyePharma, Inc. and Mundipharma Medical Company † | S-1/A | 333-170245 | 12/03/2010 | 10.8 | |||||||||||||||||||
| 10.9 | Distribution Agreement, dated June 30, 2003, between SkyePharma, Inc. and Mundipharma International Holdings Limited † | S-1/A | 333-170245 | 12/03/2010 | 10.9 | |||||||||||||||||||
| 10.10 | Distribution Agreement, dated July 27, 2005, between SkyePharma, Inc. and Mundipharma International Holdings Limited † | S-1/A | 333-170245 | 12/03/2010 | 10.10 | |||||||||||||||||||
| 10.11 | Co-development, Collaboration and License Agreement, dated January 2, 2003, among Enzon Pharmaceuticals, Inc., Jagotec, AG, SkyePharma, Inc. and SkyePharma PLC † | S-1/A | 333-170245 | 12/03/2010 | 10.11 | |||||||||||||||||||
| 10.12 | DepoCyt Supply and Distribution Agreement, dated December 31, 2002, between SkyePharma, Inc. and Enzon Pharmaceuticals, Inc. † | S-1/A | 333-170245 | 12/03/2010 | 10.12 | |||||||||||||||||||
| 10.13 | Amended and Restated Strategic Licensing, Distribution and Marketing Agreement, dated October 15, 2009, between the Registrant and EKR Therapeutics, Inc. † | S-1/A | 333-170245 | 12/03/2010 | 10.13 | |||||||||||||||||||
Table of Contents
| Exhibit |
Exhibit Description |
Incorporated by Reference | Filed Herewith |
|||||||||||||||||||||
| Form | File Number |
Date of First Filing |
Exhibit Number |
|||||||||||||||||||||
| 10.14 | Amended and Restated Supply Agreement, dated October 15, 2009, between the Registrant and EKR Therapeutics, Inc. † | S-1/A | 333-170245 | 12/03/2010 | 10.14 | |||||||||||||||||||
| 10.15 | Strategic Marketing Agreement, dated September 25, 2007, between the Registrant and Flynn Pharma Limited † | S-1/A | 333-170245 | 12/03/2010 | 10.15 | |||||||||||||||||||
| 10.16 | Supply Agreement, dated December 5, 2007, between the Registrant and Flynn Pharma Limited † | S-1/A | 333-170245 | 12/03/2010 | 10.16 | |||||||||||||||||||
| 10.17 | Lease Agreement, dated August 17, 1993, amended July 2, 2009, between Pacira Pharmaceuticals, Inc. and HCP TPSP, LLC | S-1 | 333-170245 | 11/01/2010 | 10.19 | |||||||||||||||||||
| 10.18 | Lease Agreement, dated December 8, 1994, amended July 2, 2009, between Pacira Pharmaceuticals, Inc. and LASDK Limited Partnership | S-1 | 333-170245 | 11/01/2010 | 10.20 | |||||||||||||||||||
| 10.19 | Services Agreement, dated October 28, 2010, between the Registrant, MPM Asset Management LLC and Gary Patou | S-1/A | 333-170245 | 12/03/2010 | 10.19 | |||||||||||||||||||
| 10.20 | Services Agreement, dated September 15, 2010, between Pacira Pharmaceuticals, Inc. and Stack Pharmaceuticals, Inc. | S-1/A | 333-170245 | 12/03/2010 | 10.20 | |||||||||||||||||||
| 10.21 | Employment Agreement between the Registrant and David Stack | S-1/A | 333-170245 | 12/03/2010 | 10.21 | |||||||||||||||||||
| 10.22 | Employment Agreement between the Registrant and James Scibetta | S-1/A | 333-170245 | 12/03/2010 | 10.22 | |||||||||||||||||||
| 10.23 | Employment Agreement between the Registrant and Mark Walters | S-1/A | 333-170245 | 12/03/2010 | 10.23 | |||||||||||||||||||
| 10.24 | Employment Agreement between the Registrant and William Lambert | S-1/A | 333-170245 | 12/03/2010 | 10.24 | |||||||||||||||||||
| 10.25 | Loan and Security Agreement, dated November 24, 2010, among the Registrant, Pacira Pharmaceuticals, Inc. (California), Hercules Technology Growth Capital, Inc. and Hercules Technology II, L.P. | S-1/A | 333-170245 | 12/03/2010 | 10.25 | |||||||||||||||||||
| 10.26 | Guaranty Agreement, dated November 24, 2010, between the Registrant, Hercules Technology Growth Capital, Inc., Hercules Technology II, L.P. and the parties named therein | S-1/A | 333-170245 | 12/03/2010 | 10.26 | |||||||||||||||||||
| 10.27 | Warrant to purchase preferred stock of the Registrant, dated November 24, 2010 | S-1/A | 333-170245 | 12/03/2010 | 10.27 | |||||||||||||||||||
| 10.28 | Form of Warrant to purchase Series A convertible preferred stock of the Registrant, dated July 2, 2009 | S-1 | 333-170245 | 11/01/2010 | 10.33 | |||||||||||||||||||
| 10.29 | Form of Warrant to purchase common stock of the Registrant, dated January 22, 2009 | S-1 | 333-170245 | 11/01/2010 | 10.34 | |||||||||||||||||||
| 10.30 | Form of Warrant to purchase common stock of the Registrant, dated December 29, 2010 | S-1/A | 333-170245 | 01/03/2011 | 10.30 | |||||||||||||||||||
| 10.31 | 2011 Stock Incentive Plan | S-1/A | 333-170245 | 01/03/2011 | 10.31 | |||||||||||||||||||
| 10.32 | Form of Indemnification Agreement between the Registrant and its directors and officers | S-1/A | 333-170245 | 01/13/2011 | 10.32 | |||||||||||||||||||
| 10.33 | Development and License Agreement, dated January 14, 2011, between the Registrant and Novo Nordisk A/S † | S-1/A | 333-170245 | 01/20/2011 | 10.33 | |||||||||||||||||||
| 21.1 | Subsidiaries of Registrant | X | ||||||||||||||||||||||
| 31.1 | Certification of President and Chief Executive Officer pursuant to Exchange Act Rule 13a-14(a) | X | ||||||||||||||||||||||
Table of Contents
| Exhibit |
Exhibit Description |
Incorporated by Reference | Filed Herewith |
|||||||||||||||||||||
| Form | File Number |
Date of First Filing |
Exhibit Number |
|||||||||||||||||||||
| 31.2 | Certification of Chief Financial Officer pursuant to Exchange Act Rule 13a-14(a) | X | ||||||||||||||||||||||
| 32.1 | Certification of President and Chief Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | X | ||||||||||||||||||||||
| 32.2 | Certification of Chief Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | X | ||||||||||||||||||||||
| † | Confidential treatment granted by the Securities and Exchange Commission as to certain portions. |
