Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K RE INVESTOR PRESENTATION 07.27.10 - JUNIPER PHARMACEUTICALS INC | form8k.htm |

1
Corporate Presentation July 2010
Frank Condella, President and CEO
Larry Gyenes, CFO

2
This presentation contains forward-looking statements, which statements are indicated by the words
“may,” “will,” “plans,” “believes,” “expects,” “anticipates,” “potential,” and similar expressions. Such
forward-looking statements involve known and unknown risks, uncertainties, and other factors that may
cause actual results to differ materially from those projected in the forward-looking statements.
“may,” “will,” “plans,” “believes,” “expects,” “anticipates,” “potential,” and similar expressions. Such
forward-looking statements involve known and unknown risks, uncertainties, and other factors that may
cause actual results to differ materially from those projected in the forward-looking statements.
Factors that might cause future results to differ include, but are not limited to, the following: the
successful marketing of CRINONE® (progesterone gel) by Watson Pharmaceuticals, Inc., in the United
States; the successful marketing of CRINONE by Merck Serono outside the United States; the timely
and successful completion of the ongoing Phase III PREGNANT (PROCHIEVE® (progesterone gel)
Extending Gestation A New Therapy) Study of PROCHIEVE 8% to reduce the risk of preterm birth in
women with a short cervix at mid-pregnancy; successful development of a next-generation vaginal
progesterone product; success in obtaining acceptance and approval of new products and new
indications for current products by the United States Food and Drug Administration and international
regulatory agencies; the impact of competitive products and pricing; the timely and successful
negotiation of partnerships or other transactions; the strength of the United States dollar relative to
international currencies, particularly the euro; competitive economic and regulatory factors in the
pharmaceutical and healthcare industry; general economic conditions; and other risks and
uncertainties that may be detailed, from time-to-time, in Columbia’s reports filed with the SEC.
successful marketing of CRINONE® (progesterone gel) by Watson Pharmaceuticals, Inc., in the United
States; the successful marketing of CRINONE by Merck Serono outside the United States; the timely
and successful completion of the ongoing Phase III PREGNANT (PROCHIEVE® (progesterone gel)
Extending Gestation A New Therapy) Study of PROCHIEVE 8% to reduce the risk of preterm birth in
women with a short cervix at mid-pregnancy; successful development of a next-generation vaginal
progesterone product; success in obtaining acceptance and approval of new products and new
indications for current products by the United States Food and Drug Administration and international
regulatory agencies; the impact of competitive products and pricing; the timely and successful
negotiation of partnerships or other transactions; the strength of the United States dollar relative to
international currencies, particularly the euro; competitive economic and regulatory factors in the
pharmaceutical and healthcare industry; general economic conditions; and other risks and
uncertainties that may be detailed, from time-to-time, in Columbia’s reports filed with the SEC.
All forward-looking statements contained herein are neither promises nor guarantees. Columbia does
not undertake any responsibility to revise or update any forward-looking statements.
not undertake any responsibility to revise or update any forward-looking statements.

3
} Frank C. Condella, Jr. - President and CEO, Director
◦ Chairman of SkyePharma
◦ Formerly CEO of SkyePharma; European President of IVAX Corporation;
CEO of Faulding Pharmaceutical Co.; Vice President, Roche Laboratories
CEO of Faulding Pharmaceutical Co.; Vice President, Roche Laboratories
} Lawrence A. Gyenes - SVP, CFO & Treasurer
◦ Formerly CFO at Acusphere, Zila, Savient & Reliant; 15 years at Searle
} Michael McGrane - SVP, General Counsel and Secretary
◦ Formerly General Counsel, The Liposome Co.; Novartis Consumer Health
} George W. Creasy, MD - VP, Clinical Research & Development
• Fellow of the American College of Obstetricians and Gynecologists
• Formerly spent 16 years Johnson & Johnson

4
} Successful specialty pharmaceutical company
leveraging our bioadhesive drug delivery system and
clinical expertise to develop proprietary products
leveraging our bioadhesive drug delivery system and
clinical expertise to develop proprietary products
} Marketed products:
◦ CRINONE® 8% progesterone vaginal gel
– Sold worldwide for use in infertility treatments
– Marketed by Watson (US) and Merck Serono (RoW)
◦ STRIANT® testosterone buccal system
– Testosterone replacement for hypogonadal men
– Marketed by Columbia (US), The Urology Company (UK) &
Sandoz (Italy)
Sandoz (Italy)

5
} Transactions closed 7/2/2010
} Watson acquired:
◦ Substantially all of CBRX progesterone assets
◦ 11.2 million shares CBRX common stock
} Watson assumed responsibility for all US sales,
marketing & distribution activities for CRINONE
& PROCHIEVE
marketing & distribution activities for CRINONE
& PROCHIEVE
} All CBRX debt retired

6
} Watson paid CBRX:
◦ $62 million upfront for assets
} Watson will pay CBRX:
◦ Ongoing royalties escalating from 10 to 20%
◦ Up to $45.5 million upon CBRX achievement of five
milestones in the short cervix/preterm birth opportunity
milestones in the short cervix/preterm birth opportunity
} Watson will directly fund:
◦ Cost of PREGNANT Study and NDA filing above $7 million
from 1/1/10
from 1/1/10
◦ Cost of progesterone life-cycle management program
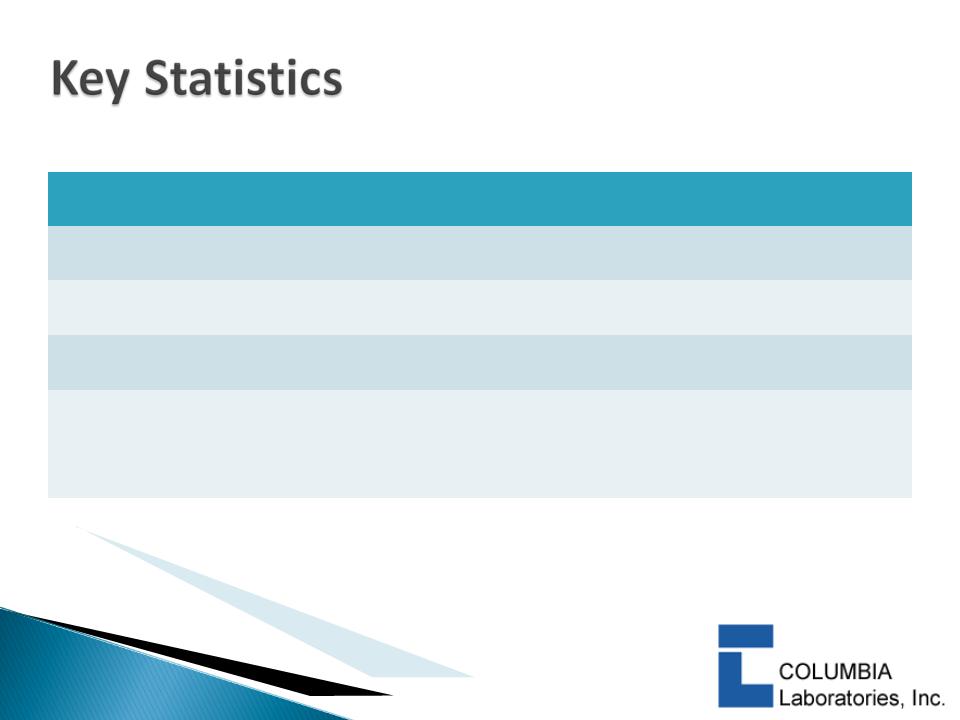
7
|
Nasdaq: CBRX
|
|
|
Recent market price (7/23/2010)
|
$0.94
|
|
Shares Outstanding
|
84.3 million
|
|
Market capitalization
|
$79.2 million
|
|
Cash and equivalents (7/2/2010)
|
$25.0+ million
|
|
Debt (7/2/2010)
|
$0.00
|

8

9
} Birth of an infant before 37 completed weeks of
gestation
gestation
} 1 in 8 babies were born preterm in the US in 20081
} #1 cause of neonatal mortality (<28 days) in US2
} #2 cause of infant mortality (<1 year) in US2
◦ #1 cause of infant mortality for non-Hispanic black infants in
US2
US2
1 Martin, JA, Osterman MJK. Are Preterm Births on the Decline in the United States? Recent Data from the
National Vital Statistics System. NCHS data brief, no. 39 Hyattsville, MD: National Center for Health Statistics,
2010.
National Vital Statistics System. NCHS data brief, no. 39 Hyattsville, MD: National Center for Health Statistics,
2010.
2 Mathews TJ, MacDorman MF. National Vital Statistics Report 2006; 54:1-29; National Center for Health
Statistics, 2003 period-linked birth/infant death data. Prepared by March of Dimes Perinatal Data Center, 2006.
Statistics, 2003 period-linked birth/infant death data. Prepared by March of Dimes Perinatal Data Center, 2006.

Behrman RE et al. in: Behrman RE, Butler AS, eds. Preterm Birth: Causes, Consequences,
and Prevention. Washington, DC: The National Academies Press; 2006:329-354.
and Prevention. Washington, DC: The National Academies Press; 2006:329-354.
Lost household and
market productivity
market productivity
$5.7 billion
Maternal delivery costs
$1.9 billion
Children’s early intervention
services
services
$611 million
Infant Costs
Special education services
$1.1 billion
Medical care services
$16.9 billion
~$51,600 per Preterm Infant

11
$1.7+ Billion Total US Market Opportunity
$225+ million market
potential
potential
4.3 Million Births Annually
>2.5 - 3.0 cm
(20%)
(20%)
$1.1+ billion
market potential
> 2.0 - 2.5 cm
(6%)
(6%)
1.0 - 2.0 cm
(4%)
(4%)
$340+ million market
potential
potential
16 weeks X $83.31 week =
$1,333 per patient (at current price levels)

12
} Studies have shown that “short cervix” is the most
powerful predictor of preterm birth
powerful predictor of preterm birth
◦ The risk of spontaneous preterm delivery increases as
cervical length (CL) decreases
cervical length (CL) decreases
} To assess risk, cervical length measurements must
be taken at mid-pregnancy (18-22 weeks)
be taken at mid-pregnancy (18-22 weeks)
◦ Transvaginal ultrasound
} A cervical length of <3.0cm is considered “short”
} The shorter the cervix at mid-pregnancy, the higher
the risk of PTB
the risk of PTB

13
} To & Nicolaides 2006: Short cervix is the most important single
predictor of PTB
predictor of PTB
} Fonseca & Nicolaides 2007: Women with a short cervix
responded to vaginal progesterone therapy
responded to vaginal progesterone therapy
} DeFranco 2007:
◦ PROCHIEVE 8% use was associated with a statistically significant
reduction in PTB in women with CL ≤ 3.0 cm & < 2.8 cm
reduction in PTB in women with CL ≤ 3.0 cm & < 2.8 cm
◦ PROCHIEVE 8% improved infant outcomes1 in women with
baseline CL < 2.8 cm
baseline CL < 2.8 cm
– Statistically significant reduction in Neonatal Intensive Care Unit
(NICU) admissions
(NICU) admissions
– Statistically significant reduction in average NICU days
1 First study to show this

14
progesterone
placebo
33 investigators
n=116 (58 Prochieve, 58 Placebo)
Baseline Cervical Length ≤ 3.0 cm
Baseline Cervical Length < 2.8 cm
progesterone
placebo
22 Investigators
N=46 (19 Prochieve, 27 Placebo)
Fishers Exact Test
at <32 weeks*:
at <32 weeks*:
(p = 0.014)
The Kaplan-Meier method was
used for calculation; (Wilcoxon
P = 0.043).
used for calculation; (Wilcoxon
P = 0.043).
DeFranco et al, Ultrasound Obstet
Gynecol. 2007; 30: 697-705.
Gynecol. 2007; 30: 697-705.

15
p=0.016
p=0.077
p=0.16
DeFranco et al, Ultrasound Obstet
Gynecol. 2007; 30: 697-705.
Gynecol. 2007; 30: 697-705.
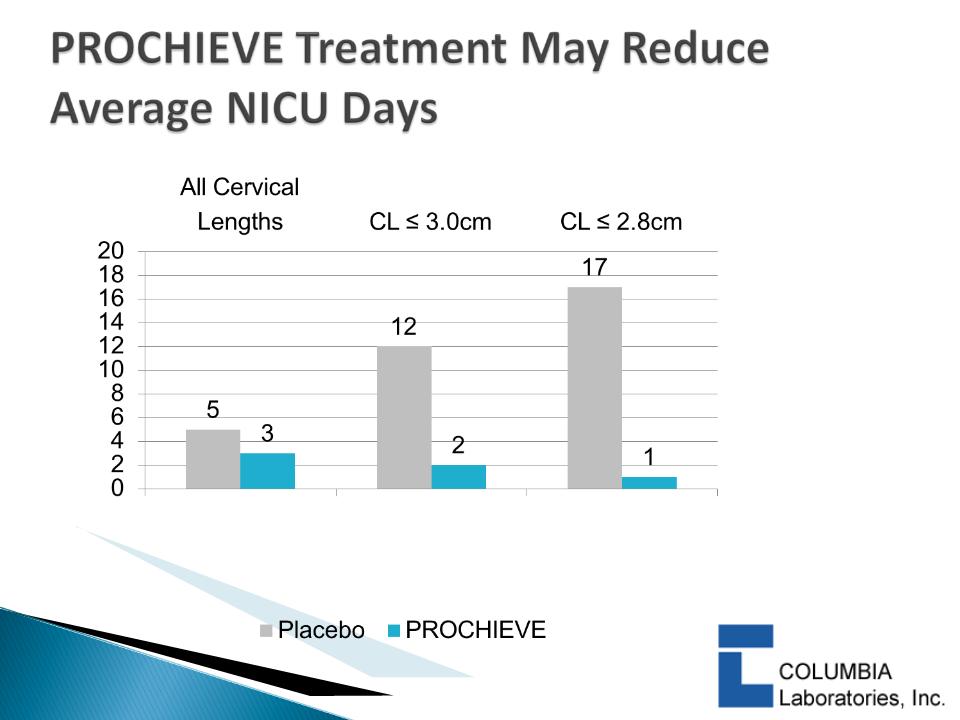
16
p=0.013
p=0.026
p=0.05
DeFranco et al, Ultrasound Obstet
Gynecol. 2007; 30: 697-705.
Gynecol. 2007; 30: 697-705.

17
} Common interventions (bed rest, tocolytics,
antibiotics, and cervical cerclage) have proven to be
of little or no benefit1,2
antibiotics, and cervical cerclage) have proven to be
of little or no benefit1,2
} PROCHIEVE is the most promising treatment to
reduce PTB and improve infant outcomes in women
with a short cervix
reduce PTB and improve infant outcomes in women
with a short cervix
} PROCHIEVE 8% progesterone vaginal gel has a long
history of safety and convenience
history of safety and convenience
1 Nicolaides et al, The Lancet. 2004; 363:1849-1853.
2 Smith et al, Eur J Obstet Gynecol Reprod Biol. 2009; 142:3-11.

18
} Phase III clinical trial with PROCHIEVE 8%
◦ Double-blind, placebo controlled
◦ Enrolled 465 pregnant women with cervical length 1.0-2.0cm
◦ 40+ centers (US & abroad)
◦ Primary endpoint: a reduction in preterm births at ≤32 6/7
weeks vs. placebo
weeks vs. placebo
◦ Improved infant outcomes important secondary endpoint
} NIH co-sponsor
◦ Validates science and design of trial

19
} Enrollment complete June 2010
} Last infant is born Q4 2010
} Report study outcomes Around Y/E 2010
} File with FDA* 2011
} FDA decision* 2011/2012**
*Assuming positive outcome
**PDUFA limits review time to 10 months;
could shorten to 6 months if granted
priority review
could shorten to 6 months if granted
priority review

20

21
} Solid track record in development of women’s health
products:
products:
◦ Bioadhesive products
◦ Vaginal delivery systems
} Unique expertise in clinical development in PTB
} Proven ability to design and successfully manage
complex global development projects, including
collaboration with NIH
complex global development projects, including
collaboration with NIH
} Expertise in utilizing technology platform in multiple
therapeutic areas
therapeutic areas

22
} Progesterone delivery (2013)*
} Progressive hydration for extended release (2019)
} First Uterine Pass Effect (2018)
} pH buffering to prevent preterm labor (2018)
} Treatment of endometriosis/infertility with β-adrenergic agonists (2020)
} Carbamide peroxide for bacterial vaginosis (2022)
} Local anesthetic for chronic pelvic pain (2022) pending
} Extended release ionic treating agents (2023) pending
} Progesterone for treatment of preterm (2028) pending*
*Progesterone-specific patents were transferred to Watson
following the close of the Watson Transactions
following the close of the Watson Transactions

23
Polycarbophil:
hydrogen bonding
with the cell surface
hydrogen bonding
with the cell surface
Active entrapped in
the cross-linked polymer
polycarbophil
Bioadhesive gel,
tablet or system
with active
tablet or system
with active
Adheres to mucosal surface
Discharged upon normal cell
turnover:
turnover:
Oral mucosa up to 24
hours
hours
Vaginal epithelium 72+
hours
hours
Subcutaneous
tissue
tissue

Results in drug delivery
directly to the endometrium
with minimal systemic levels
directly to the endometrium
with minimal systemic levels
CRINONE® 8% (progesterone gel) (90 mg)
IM progesterone (50 mg)
Cicinelli E et al. Obstet Gynecol. 2000; 95: 403-406.

25
|
Product
|
Indication
|
Clinical Stage
|
Market
Opportunity |
|
Crinone/Prochieve 8%
Vaginal Gel
|
Reduce risk of preterm birth in
women with a short cervix in mid -pregnancy |
Pivotal Phase III
|
$1.7 billion (US)
|
|
Crinone/Prochieve 8%
Next generation
|
Lifecycle management program:
ART (Infertility) |
Phase I underway
|
$300+ million
(US) |
|
Crinone/Prochieve 8%
Next generation
|
Lifecycle management program:
Preterm Birth |
Phase I underway
|
$1.7 billion (US)
|
|
COL-2401
Vaginal Tablet
|
Treatment and prevention of
bacterial vaginosis (BV) |
Preclinical work
in-process |
$200+ million
(US) |
|
COL-1777
Vaginal delivery |
Fibroid reduction
|
Phase I work
underway |
$1.2 billion (US)
|
|
COL-1077
Vaginal Gel
|
Treatment of chronic pelvic pain
(CPP) |
Pilot clinical work
complete |
$1.5 billion (US)
|

26
} Most common bacterial infection for women of
childbearing years
childbearing years
◦ 7.4 million new cases annually (US est.)
} Involves an imbalance in the vaginal bacterial ecosystem,
such that natural flora are diminished
such that natural flora are diminished
} Current treatments: antibiotics given orally or intravaginally
◦ Significant side effects from oral forms
◦ Patients report greater satisfaction with intravaginal forms
} Risk factor for preterm birth
} High recurrence rate

27
} Facilitates restoration of normal, protective bacteria
} Maintains normal pH to prevent growth of harmful
bacteria
bacteria
} Vaginally administered
} Natural bacteriostatic agent
} $200+ million US market opportunity
} Patent protection to 2022

28
} Symptoms include excessive vaginal bleeding, pelvic
pressure and anemia
pressure and anemia
◦ Increased risk of infertility and pregnancy complications
} Affects 30 million women (up to 20%) in US
} Most common indication for hysterectomy
} Health care costs >$2 billion in 1997 for surgical
management of uterine fibroids
management of uterine fibroids
} $1.2 billion market opportunity

29
} Current treatment options
◦ Medications such as GnRH agonists
– Side effects: menopausal symptoms & bone loss
– Short-term therapy only
◦ Surgical hysterectomy or myomectomy
– Hysterectomy not an option for women who want to preserve
childbearing capacity
childbearing capacity
} COL-1777 could
◦ Avoid menopausal side effects caused by GnRH agonists
◦ Avoid costs and side effects of surgery
◦ Preserve childbearing ability
} Patent protection to 2019

30
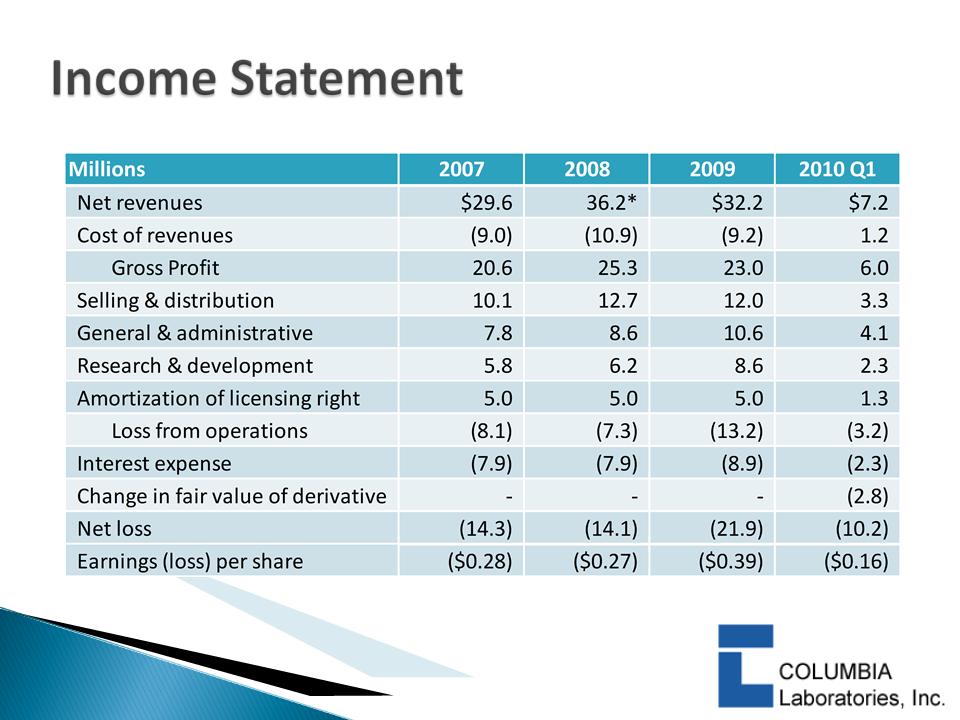
31
*2008 net revenues include $2.9 million in previously deferred revenue for
STRIANT licensing fees from Ardana as a results of its bankruptcy in Q2 2008.
STRIANT licensing fees from Ardana as a results of its bankruptcy in Q2 2008.

32
• A modest 12% increase in product sales to $17 million
would match the 2009 product contribution margin
would match the 2009 product contribution margin
• The above excludes Merck Serono revenues, which
contributed another $9 million in revenues and $6 million in
contribution margin in 2009
contributed another $9 million in revenues and $6 million in
contribution margin in 2009

33
} Strong cash balance; debt free
} Ongoing royalty revenues from Watson & Merck
Serono
Serono
} STRIANT sales
} Potential milestone payments
} Significantly lower operating expenses
} Cash burn rate ~ $1 million/quarter through YE 2010
} With successful PREGNANT Study outcome CBRX
will be cash flow & earnings positive on an annual
basis from that point forward
will be cash flow & earnings positive on an annual
basis from that point forward

34
|
Investor Relations Contacts
|
|
|
Larry Gyenes
Chief Financial Officer,
Columbia Laboratories, Inc.
|
Seth Lewis
Vice President,
The Trout Group
|
|
T: (973) 486-8860
|
T: (646) 378-2952
|
|
lgyenes@columbialabs.com
|
slewis@troutgroup.com
|
|
www.columbialabs.com
|
www.cbrxir.com
|
