Attached files
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM 10-K
x ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
For the
fiscal year ended March 31, 2010
OR
o TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF
1934
For the
transition period from
to .
Commission
file number 000-50601
SYNUTRA
INTERNATIONAL, INC.
|
DELAWARE
|
13-4306188
|
|
|
(State
or Other Jurisdiction of
Incorporation
or Organization)
|
I.R.S.
Employer
Identification
No.
|
|
|
2275
Research Blvd., Suite 500
Rockville,
Maryland 20850
|
||
|
(Address
of Principal Executive Offices, Zip Code)
|
||
|
(301)
840-3888
|
||
|
(Registrant’s
Telephone Number, Including Area
Code)
|
||
Securities
registered pursuant to Section 12(b) of the Act:
|
Title of Each Class
|
Name of Each Exchange on Which
Registered
|
|
Common
Stock $0.0001 Par Value
|
NASDAQ
Global Select Market
|
Securities
registered pursuant to Section 12(g) of the Act: None
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act. Yes oNo x
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Exchange Act. Yes oNo x
Indicate
by check mark whether the registrant (1) has filed all reports required to be
filed by Section 13 or 15(d) of the Securities Exchange Act 1934 during the
preceding 12 months (or for such shorter period that the registrant was required
to file such reports) and (2) has been subject to such filing requirements for
the past 90 days. Yes x
No o
Indicate
by check mark whether the registrant has submitted electronically and posted on
its corporate Web site, if any, every Interactive Data File required to be
submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this
chapter) during the preceding 12 months (or for such shorter period that the
registrant was required to submit and post such
files). Yes o
No o
Indicate
by check mark if disclosure of delinquent filers in response to Item 405 of
Regulation S-K is not contained herein, and will not be contained, to the best
of registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this
Form 10-K x
Indicate
by check mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting company. See
the definitions of “large accelerated filer”, “accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
|
Large
accelerated filer o
|
Accelerated
filer x
|
Non-accelerated
filer o
|
Smaller
reporting company o
|
Indicate
by check mark whether the registrant is a shell company (as defined in Rule
12b-2 of the Exchange Act). Yes oNo x
The
aggregate market value of the voting and non-voting common equity held by
non-affiliates of the registrant based on the closing sales price of the
registrant’s common stock on September 30, 2009 (the last business day of the
registrant’s most recently completed second fiscal quarter), as reported on the
NASDAQ Global Select Market, was $246.8 million. For purposes of this
disclosure, shares of common stock held by persons who hold more than 10% of the
outstanding shares of common stock and shares held by officers and directors of
the registrant have been excluded in that such persons may be deemed to be
affiliates. This determination is not necessarily conclusive for other
purposes.
As of June
1, 2010, there were 54,000,713 shares of the registrant’s common stock
outstanding.
TABLE
OF CONTENTS
Page
PART
I
|
Item
1. Business
|
2
|
|
Item
1A. Risk Factors
|
16
|
|
Item
1B. Unresolved Staff Comments
|
38
|
|
Item
2. Properties
|
38
|
|
Item
3. Legal Proceedings
|
39
|
|
Item
4. [Removed And Reserved]
|
40
|
|
PART
II
|
|
|
Item
5. Market for Registrant’s Common Equity, Related Stockholders Matters and
Issuer Purchases of Equity Securities
|
40
|
|
Item
6. Selected Financial Data
|
41
|
|
Item
7. Management’s Discussion and Analysis of Financial Condition and Results
of Operations
|
42
|
|
Item
7A. Quantitative and Qualitative Disclosures About Market
Risk
|
63
|
|
Item
8. Financial Statements and Supplementary Data
|
64
|
|
Item
9. Changes in and Disagreements With Accountants on Accounting and
Financial Disclosure
|
98
|
|
Item
9A. Controls and Procedures
|
98
|
|
Item
9B. Other Information
|
100
|
|
PART
III
|
|
|
Item
10. Directors, Executive Officers and Corporate Governance
|
101
|
|
Item
11. Executive Compensation
|
105
|
|
Item
12. Security Ownership of Certain Beneficial Owners and Management and
Related Stockholder Matters
|
108
|
|
Item
13. Certain Relationships and Related Transactions, and Director
Independence
|
110
|
|
Item
14. Principal Accounting Fees and Services
|
113
|
|
PART
IV
|
|
|
Item
15. Exhibits and Financial Statement Schedules
|
113
|
|
Signatures
|
114
|
i
CONVENTIONS
THAT APPLY TO THIS ANNUAL REPORT ON FORM 10-K
Except
where the context otherwise requires and for purposes of this Annual Report on
Form 10-K only:
|
·
|
“we,”
“us,” “our company,” “our,” and “Synutra” refer to Synutra International,
Inc., and its consolidated
subsidiaries;
|
|
·
|
“China”
or “PRC” refers to the People’s Republic of China, excluding Taiwan and
the Special Administrative Regions of Hong Kong and
Macau;
|
|
·
|
all
references to “ton” or “tons” are to “tonne” or “metric
ton”;
|
|
·
|
all
references to “Renminbi” or “RMB” are to the legal currency of China;
and
|
|
·
|
all
references to “U.S. dollars,” “dollars,” or “$” are to the legal currency
of the United States.
|
Amounts
may not always add to the totals due to rounding.
Unless
otherwise noted, all translations from Renminbi to U.S. dollars were made at the
mid rate published by the People’s Bank of China, or the mid rate, as of March
31, 2010, which was RMB6.8263 to $1.00. We make no representation that the
Renminbi amounts referred to in this Annual Report on Form 10-K could have been
or could be converted into U.S. dollars at any particular rate or at all. On
June 1, 2010, the mid rate was RMB 6.8279 to $1.00.
PART
I
This
Annual Report on Form 10-K, or Form 10-K, contains “forward-looking statements”
within the meaning of the Private Securities Litigation Reform Act of 1995, that
are based on our current expectations, assumptions, estimates and projections
about us and our industry. All statements other than statements of historical
fact in this Form 10-K are forward-looking statements. In some cases, these
forward-looking statements can be identified by words or phrases such as
“anticipate,” “believe,” “continue,” “estimate,” “expect,” “intend,” “is/are
likely to,” “may,” “plan,” “should,” “will,” “aim,” “potential,” “continue,” or
other similar expressions. The forward-looking statements included in this Form
10-K relate to, among others:
|
·
|
our
goals and strategies;
|
|
·
|
our
future business development, financial condition and results of
operations;
|
|
·
|
the
expected growth of the nutritional products and infant formula markets in
China;
|
|
·
|
market
acceptance of our products;
|
|
·
|
adverse
effects associated with the melamine contamination
incident;
|
|
·
|
our
expectations regarding demand for our
products;
|
|
·
|
our
ability to stay abreast of market trends and technological
advances;
|
|
·
|
competition
in the infant formula industry in
China;
|
|
·
|
PRC
governmental policies and regulations relating to the nutritional products
and infant formula industries; and
|
|
·
|
general
economic and business conditions in
China.
|
These
forward-looking statements involve various risks and uncertainties. Although we
believe that our expectations expressed in these forward-looking statements are
reasonable, our expectations may turn out to be
1
incorrect.
Our actual results could be materially different from our expectations.
Important risks and factors that could cause our actual results to be materially
different from our expectations are generally set forth in the “Item 1.
Business,” “Item 1A. Risk Factors,” “Item 7. Management’s Discussion and
Analysis of Financial Condition and Results of Operations,” and other sections
in this Form 10-K.
The
forward-looking statements are made as of the date of this Form 10-K. We
undertake no obligation to update any forward-looking statements to reflect
events or circumstances after the date on which the statements are made or to
reflect the occurrence of unanticipated events.
ITEM
1. BUSINESS
General
Development and Narrative Description of Business
We are a
leading infant formula company in China. We principally produce, market, and
sell our products under the “Shengyuan” or “Synutra” name, together with other
complementary brands. Our strategy is focused on selling premium infant formula
products, as well as more affordable infant formulas targeting the mass market
and other nutritional products and ingredients. We sell our products through an
extensive nationwide sales and distribution network, including independent
distributors, covering 30 provinces and provincial-level municipalities in
China. As of March 31, 2010, this network comprised over 540 independent
distributors and over 1,000 independent sub-distributors who sell our products
in over 71,000 retail outlets.
We
currently have three reportable segments which are:
|
·
|
the
powdered formula segment, which includes powdered infant and adult formula
products sold under our Super, U-Smart, Mingshan and Helanruniu
sub-brands;
|
|
·
|
the
baby food segment, which includes prepared foods for babies and children
sold under our Huiliduo sub-brand;
and
|
|
·
|
the
nutritional ingredients and supplements segment, which includes the
production and sale of nutritional ingredients and supplements such as
chondroitin sulfate, microencapsulated Docosahexanoic Acid (“DHA”) and
Arachidonic Acid (“ARA”).
|
Our other
business includes non-core operations such as toll packaging, toll drying
service and sales of ingredients and materials to industrial customers. A major
portion of other business for the fiscal year ended March 31, 2010 consist of
sales of surplus industrial milk powder which generated immediate cash flow for
us. Sales from our other business comprised approximately 32.6% of our net sales
for the fiscal year ended March 31, 2010.
On
September 16, 2008, we announced a compulsory recall on certain lots of U-Smart
products and a voluntary recall of other products that were contaminated or
suspected to be contaminated by melamine, a substance not approved for use in
food that is linked to illnesses among infants and children in China. The cost
of this recall during the fiscal year ended March 31, 2009 was $101.5 million,
including the cost of product replacement of $48.1 million in cost of sales, the
write-down and write-off of affected inventory of $48.5 million in cost of
sales, the net amount of $2.3 million to a compensation fund set up by China
Dairy Industry Association to settle existing and potential claims arising in
China from families of infants affected by melamine contamination in general and
administrative expenses, and freight charges of $2.6 million in selling and
distribution expenses, of which $4.5 million was recorded as a product recall
provision in the consolidated balance sheet as of March 31, 2009. During the
fiscal year ended March 31, 2010, we reversed recall expense of $0.9 million,
mostly for overestimated product replacement cost. We believe that the product
recall has been substantially completed and based on current information, we
believe that there should be no further material product recall costs incurred
relating to the melamine contamination incident. We believe that our fast
response to the melamine crisis, as well as recall efforts, including
instituting a voluntary recall were well recognized by the public and helped us
to maintain our reputation, brand recognition and relationships with our
distributors and suppliers.
There
have been certain legal proceedings brought against us in connection with the
melamine contamination incident, which may have an adverse effect on our results
of operations, see Part I - Item 1A. Risk Factors—Risks
2
Related
to Our Business—Product
liability claims against us could result in adverse publicity and potential
significant monetary damages. Although management is not aware of any
additional significant issues associated with the melamine contamination
incident, there can be no assurance that additional issues will not be
identified in the future and this may have an adverse effect on our results of
operations. See Part I - Item 1A. Risk Factors—Risks Related to Our
Business—We are highly
dependent upon consumers’ perception of the safety and quality of our products.
Any ill effects, product liability claims, recalls, adverse publicity or
negative public perception regarding particular ingredients or products or our
industry in general could harm our reputation and damage our brand and adversely
affect our results of operations.
Due to
the lingering impact of the product recall carried out in late calendar year
2008 and a greater proportion of rebates and discounts to distributors, our net
sales for the fiscal year ended March 31, 2010 decreased by 6.6% to $291.9
million from $312.5 million for the prior fiscal year. Our gross profit for the
fiscal year ended March 31, 2010 increased by 56.1% to $83.4 million from $53.4
million for the prior fiscal year. Our net loss attributable to Synutra
International, Inc. common shareholders for the fiscal year ended March 31, 2010
was $24.6 million, as compared to net loss attributable to Synutra
International, Inc. common shareholders of $100.5 million for the prior fiscal
year.
Our
Corporate Structure and History
Synutra
International, Inc is a Delaware holding company that conducts its business
through its operating subsidiaries in China. It owns all or majority of the
equity interests in its operating subsidiaries, directly or indirectly, through
Synutra, Inc., or Synutra Illinois, an intermediate holding company, and Synutra
International Company Limited. Synutra Illinois was incorporated in Illinois in
2000 and has no other significant assets and operations of its own. Our
corporate structure reflects common practice for companies with operations in
the PRC where separate legal entities are often used for tax or administrative
reasons.
On July
15, 2005, Synutra Illinois completed a reverse acquisition transaction with
Vorsatech Ventures, Inc., or Vorsatech. Upon the consummation of this share
exchange transaction, Vorsatech’s total issued and outstanding common stock
equaled 50,000,713 shares, including 48,879,500 shares issued pursuant to the
reverse acquisition transaction and 1,121,213 shares owned by Vorsatech’s
existing stockholders. Thereafter, Synutra Illinois became Vorsatech’s wholly
owned subsidiary and Vorsatech became the reporting entity for our business. We
subsequently changed the name of the reporting entity to Synutra International,
Inc.
On May
24, 2007, we entered into a Common Stock Purchase Agreement with Warburg Pincus
Private Equity IX, L.P., or Warburg, pursuant to which we sold 4,000,000 shares
of our common stock for an aggregate purchase price of $66 million. The closing
of the transaction took place on June 15, 2007.
The
following is a brief description of our major operating subsidiaries in
China.
|
·
|
Shengyuan
Nutritional Food Co., Ltd., or Shengyuan Nutrition, formerly known as
Qingdao St. George Dairy Co., Ltd., located in Qingdao, Shandong, China,
was established by Synutra Illinois in September 2001 and is engaged in
the dry-blending, packaging, shipping and distribution of all of our
powdered formula products; in addition, it plans to provide diagnostic
services for pregnant women pursuant to a series of control agreements and
an entrustment agreement with related
parties.
|
|
·
|
Heilongjiang
Mingshan Dairy Co., Ltd., or Mingshan, formerly known as Luobei Shengyuan
Dairy Co., Ltd., located in Luobei, Heilongjiang, China, was established
in April 2001 and is engaged in raw milk processing and the production of
powdered formula. Synutra Illinois acquired 67% and 33% of the ownership
interest in Mingshan from Sheng Zhi Da Dairy Group Corporation (“Sheng Zhi
Da”) and Xiuqing Meng, the wife of Liang Zhang, our chairman and chief
executive officer, respectively, in January
2005.
|
|
·
|
Zhangjiakou
Shengyuan Dairy Co., Ltd., or
Zhangjiakou, located in Zhangjiakou, Hebei, China, was established in
March 2004 with Synutra Illinois and Sheng Zhi Da holding 40% and 60%,
respectively, of its equity interests and is engaged in raw milk
processing and the production of powdered formula. Synutra Illinois
acquired the remaining 60% ownership interest in Zhangjiakou from Sheng
Zhi Da in April 2005.
|
3
|
·
|
Inner
Mongolia Shengyuan Food Co., Ltd., or Inner Mongolia Shengyuan, located in
Zhenglanqi, Inner Mongolia, China, was established in September 2006 and
has been constructing its production facilities since its
establishment.
|
|
·
|
Inner
Mongolia Mengyuan Food Co., Ltd., or Mengyuan, located in Fengzhen, Inner
Mongolia, China, commenced operations in July 2007 and is engaged in raw
milk processing. Mengyuan was acquired by Zhangjiakou from its then
shareholders in November 2006.
|
|
·
|
Meitek
Technology (Qingdao) Co., Ltd., or Meitek, formerly known as Mei Tai
Technology (Qingdao) Co, Ltd., located in Qingdao, Shandong, China, was
established in November 2006 to produce certain nutritional supplements
and ingredients. Meitek began operations in October
2008.
|
|
·
|
Heilongjiang
Baoquanling Shengyuan Dairy Co., Ltd., or Baoquanling, located in
Junchuan, Heilongjiang, China, is engaged in raw milk processing and the
production of powdered formula. On September 9, 2009, we sold the two milk
processing factories of Baoquanling and three dairy farms of Heilongjiang
Baoquanling Shengyuan Dairy Cow Breeding Co., Ltd. (“Cow Breeding”), a
wholly owned subsidiary of Baoquanling, to Heilongjiang Wondersun Dairy
Co., Ltd. (“Wondersun”).
|
|
·
|
Harbin
Shengyuan Dairy Co., Ltd., or Harbin, located in Harbin, Heilongjiang,
China, is acquired in July 2008 and has been constructing its production
facilities since the acquisition.
|
|
·
|
Beijing
Shengyuan Huiliduo Food Technology Co., Ltd., or Huiliduo, located in
Beijing, China, was established in July 2008 to produce prepared baby
food. Huiliduo began operations in March
2009.
|
|
·
|
Beijing
Shengyuan Huimin Technology Service Co., Ltd., or Huimin, a variable
interest entity which was incorporated on July 10, 2008, plans to provide
diagnostic services for pregnant women through medical institutions.
Huimin was still in the pre-operation stage as of March 31,
2010.
|
|
·
|
Global
Food Trading (Shanghai) Co., Ltd., or Global Food, was acquired in June
2009 and is engaged in the sales and distribution of Helanruniu brand
products.
|
The
following chart reflects our organizational structure as of March 31,
2010.
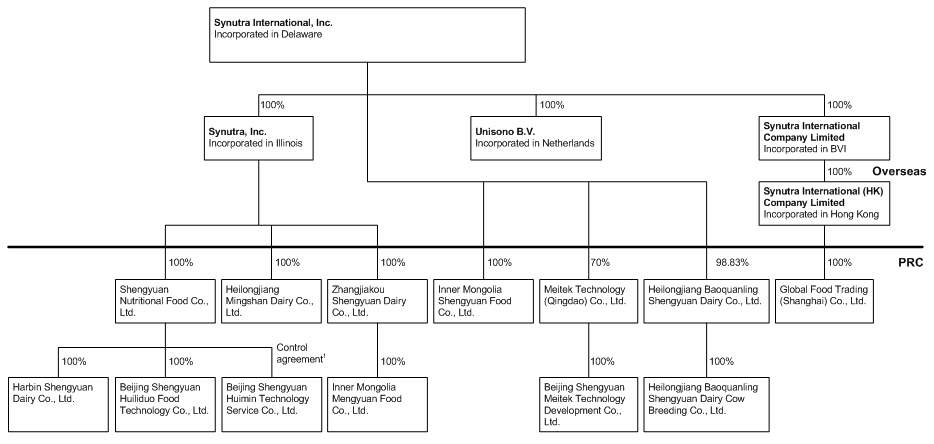
4
The
following chart shows the structure of our control agreements and the affiliated
entities consolidated into our group consolidated financial results as a result
of the control agreements:
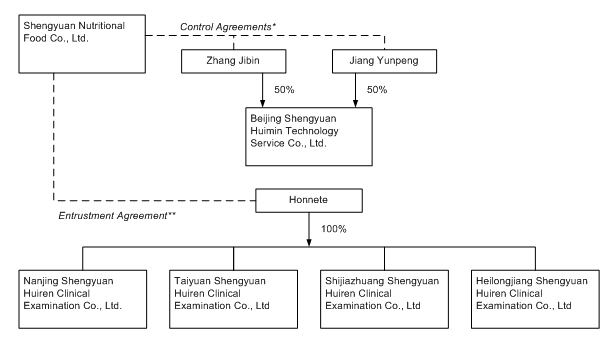
|
|
*
|
Control
Agreements include:
|
|
|
(a)
|
Exclusive
Consulting and Service Agreement entered into by and between Nutritional
and Huimin;
|
|
|
(b)
|
Business
Operating Agreement entered into by and among Nutritional, Huimin, Zhang
Jibin (who is our Director of Loans) and Jiang Yunpeng (who is our
Director of Strategic
Acquisitions);
|
|
|
(c)
|
Call
Option Agreement entered into by and among Nutritional, Huimin, Zhang
Jibin and Jiang Yunpeng;
|
|
|
(d)
|
Pledge
Agreement entered into by and among Nutritional, Zhang Jibin and Jiang
Yunpeng; and
|
|
|
(e)
|
Entrustment
Agreement entered into by and among Nutritional, Zhang Jibin and Jiang
Yunpeng.
|
|
|
**
|
Entrustment
Agreement entered into by and among Nutritional, Zhang Jibin, Jiang
Yunpeng and Honnete.
|
For a
more detailed description of the control agreements, see “Item 13. Certain
Relationships and Related Transactions, and Director Independence.”
Our
Brands
We
primarily market our products under the Synutra, or Shengyuan, name which has
been associated with infant formula products in China for more than 10 years. In
addition to the Synutra, or Shengyuan name, our products are marketed in China
under brands that we have developed through our national sales and marketing
efforts.
Synutra
Family of Brands
The
Synutra family of brands includes several of China’s leading infant formula and
children’s nutrition brands, including Super and U-Smart. We have positioned the
Synutra family of brands as high quality brands, which provide unique,
clinically supported health and developmental benefits. The Synutra family of
brands features products that include DHA and ARA, which support brain, visual
and nervous system development of infants. Building upon the strength of our
brand equity, we are extending the Synutra family of brands into the fast
growing children’s nutrition market, such as prepared baby foods.
5
Complementary
Brands
In
addition to the Synutra family of brands, we market several other brands
targeted at various consumer segments and designed to meet the nutritional needs
of the broader consumer population in China. These brands include the Mingshan
(powdered formula), Helanruniu or Holsteina (adult formula), Meitek (nutritional
supplements), and Huiliduo (prepared baby foods).
Our
Products
Our
nutritional products are grouped by category of production process and usage as
well as internal resources allocation: (1) powdered formula, (2) baby foods, (3)
nutritional ingredients and supplements, and (4) other business. Sales of
powdered formula, baby foods, nutritional ingredients and supplements comprised
approximately 66.6%, 0.3%, 0.5% of our net sales for the fiscal year ended March
31, 2010. Sales from our other business comprised approximately 32.6% of our net
sales for the fiscal year ended March 31, 2010. A major portion of other
business for the fiscal year ended March 31, 2010 consist of sales of surplus
industrial milk powder.
Powdered
Formula Products
Powdered
formula segment covers the sale of powdered infant and adult formula products.
It includes the brands of Super, U-Smart, Mingshan which was launched in October
2008 and Helanruniu, or Holsteina, which was launched in December 2008. Infant
formula is our primary product line in the powdered formula segment, accounting
for 92.8%, 85.9% and 84.8% of our total net sales in the powdered formula
segment for the fiscal years ended March 31, 2010, 2009, and 2008,
respectively.
Each of
our Super, U-Smart and Mingshan product lines has multiple formulations designed
to meet nutritional requirements and help promote a baby or child’s healthy
growth at each developmental stage. We endeavor to bring our infant formula
products closer to the quality of breast milk. We have devoted resources to
extensively adjust our product portfolio, upgrade our product lines, and add new
products or line extensions to respond to market needs and target a wider group
of consumers. To meet consumer expectations, we also periodically upgrade our
product concepts, packaging, and pricing of our products.
We
supplement our powdered infant formula products with other nutritional products
for both adults and children. Our products are targeted at, and come in
formulations that are developed to address specific types of consumer profiles,
such as middle-aged and elderly consumers with cardiologic health issues,
diabetic conditions, and calcium deficiency. Furthermore, we have developed a
product specially designed for young adults to address their calcium and other
nutrient fortification needs. Our products for women and young adults have also
undergone product extensions and upgrades to further clarify the health and
nutritional message and product image we intend to convey.
We
continue to improve our rice cereal products as supplemental and functional
foods to our powdered infant and children formula products. These improvements
included upgrades to packaging as well as product extensions with new
functionalities, new tastes and flavors, and new protein sources such as fish
and chicken.
Baby
Food Products
Baby food
segment covers the sale of prepared baby food for babies and children. It
includes the brand of Huiliduo which was launched in the quarter ended March 31,
2009. These products are designed to be part of a child’s healthy diet with
enhanced nutrition value at different stages of development.
Nutritional
Ingredients and Supplements
Nutritional
ingredients and supplements segment covers the production and sale of
nutritional ingredients and supplements such as chondroitin sulfate,
microencapsulated DHA and ARA. In the past, we had sourced and exported
chondroitin sulfate, a nutrient for joint health, to U.S. industrial customers
through our exclusive third-party agent. With the completion of our Meitek
facilities in October 2008, we are now able to produce chondroitin sulfate
ourselves. In addition, our Meitek facilities can produce microencapsulated DHA
and ARA powders and other nutritional ingredients and supplements for our own
use and for external industrial customers.
6
Production
Powdered
Formula Processing
In the
fiscal year ended March 31, 2010, all of our milk powder used for our Super,
U-Smart and Holsteina brands is imported from Fonterra Co-operative Group
(“Fonterra”) in New Zealand.
Currently,
milk powder produced at our own facilities is primarily used for commercial
resale for our other business. Only the Mingshan series of products continues to
use locally produced raw milk. Raw milk is collected from dairy farmers. Local
dairy farmers bring their dairy cattle to collection stations owned by us where
raw milk is automatically received using fully enclosed, stainless-steel vacuum
milking machines. These collection stations collect and transport the raw milk
to our production facilities which are located within 100 kilometers of these
milk collection stations, except for our Qingdao facility. Although raw milk can
remain fresh for up to 72 hours, we normally process it within 24
hours. Once received, the raw milk is processed with refrigeration
equipment that cools the raw milk to approximately four degrees Celsius. The raw
milk is then stored in air-tight tanks in preparation for advanced processes,
which include milk fat separation, sterilization and spray-drying. At our
Mingshan facilities, sterilized raw milk is mixed with whey protein powder and
other nutrients to the specifications of product formula through a wet mixing
method. The resulting mixture is then spray dried into milk powder and
transported to our Qingdao facility for final packaging.
At the
Qingdao facility, dried milk powder is mixed in large automated mechanical
mixers with whey protein powder and other additives in a method known as
dry-blending. Our dry-blending equipment can automatically adjust the level of
ingredients to achieve the complex formulations required by our premium
products. The resulting milk powder is then checked to ensure proper granule
size before packaging and distribution.
Packaging
The bulk
of our powdered formula and other nutritional products come in three types of
retail packaging: tin canisters, standup/display pouches, or sealed packages in
a box. All packaging labels carry product information, nutritional profile, user
instructions, product tracing data and shelf life date, product certification
status, quality control and assurance remarks, manufacturer contact information,
as well as customer service information that comply with PRC labeling
requirements. Selected products are also retail-packaged in single-use sizes.
Before any product leaves our packaging facility to distributors, we generally
engage in an extensive testing and inspection of the final product.
Production
and Packaging Facilities
Our
processing and packaging facilities, which are all owned by us, are located in
various locations in China, including Beijing, Qingdao, Luobei, Zhangjiakou,
Fengzhen and Zhenglanqi. These facilities encompass approximately 77,990 square
meters of office, plant, and warehouse space. Our distribution center located in
Qingdao includes over 14,116 square meters of owned office space. All of our
production facilities are built based on the GMP standard, with equipment
imported from Europe and all of our facilities that have commenced operation
have ISO9000 and HACCP series qualifications with some also being ISO14000
certified.
We
currently own and operate four processing facilities and one packaging facility
for our powdered formula production. As of March 31, 2010, we had raw milk
processing capacity of 32,600 tons per year, packaging capacity of 82,000 tons
per year and dry-blending processing capacity of 73,000 tons per
year.
Our
Qingdao facility serves as our dry-blending and packaging plant. Various
ingredients, such as milk powder, whey protein powder and nutritional additives
arrive at our Qingdao facility from our production facilities and our suppliers,
and are mixed using the dry-blending method. Qingdao facility repackages the
mixed ingredients into retail-size tin canisters or stand up/display pouches or
sealed packages in boxes. This packaging facility also provides inventory
control and logistics management, product quality monitoring and product
development assistance.
7
Our
production facility for prepared baby foods is located in Beijing and
Zhenglanqi. As of March 31, 2010, the Beijing facility had a processing capacity
of 4.8 million jars (approximately 540 tons) per year, and the Zhenglanqi
facility is planned to have a processing capacity of 18,000 tons per year upon
completion.
Our
production facility for nutritional ingredients and supplements is located in
Qingdao. As of March 31, 2010, this facility had a processing capacity of 700
tons per year for chondroitin sulfate, 1,000 tons per year for collagen protein,
and 700 tons for microencapsulated DHA and ARA powders and other nutritional
ingredients.
For
information with respect to the installed capacity, location and function of our
processing and packaging facilities, see “Item 2.
Properties”.
Raw
Materials and Suppliers
Raw
Materials
Our
business depends on maintaining a regular and adequate supply of high-quality
raw materials. In the aftermath of the melamine contamination incident, we
decided to use imported milk powder for the production of our higher end
powdered formula products. We currently source approximately 90% of milk powder
used in our production from New Zealand. We also pay market prices or premium
prices in certain regions in China for our raw milk. Our milk suppliers are
primarily dairy farmers located throughout Heilongjiang and Hebei provinces and
in Inner Mongolia.
Whey
protein powder is the other key ingredient used in the production of our
powdered infant formula products and our other dairy-based products. Like all
powdered milk producers, we use whey protein powder as the active ingredient to
help reconstituted dairy-based formula to mimic the consistency of breast milk,
which can constitute approximately 55% of the final powdered infant formula
product by weight. Whey protein powder is a byproduct of cheese-making
processes, and is difficult and costly to produce as a stand-alone product.
Since China is not a large consumer or producer of cheese and cheese products,
we and other domestic producers typically obtain whey protein powder in volume
from overseas sources, such as France.
Based on
our experience, prices of milk powder and whey protein powder can fluctuate over
relatively short periods of time depending on market conditions. Our sourcing
team monitors price movements and makes major purchases at times when prices are
attractive, subject to projected customer order flow and other
factors.
Some of
our powdered milk products, including our powdered infant formulas, also include
additives such as DHA and ARA fatty acids and other nutritional additives. DHA
and ARA fatty acids are long-chain poly-unsaturated fatty acids found in breast
milk that are believed to aid in the development of an infant’s brain, eyes and
nervous system. Studies have suggested that DHA and ARA fortification can
replicate some of the nutritional benefits of breast milk in infant formulas.
Currently we are producing microencapsulated DHA and ARA powders at our Meitek
facility which began operations in October 2008 for both internal use and
external sales.
We use
vegetable oils in our dry-spraying powder infant formula production processes as
a binder for the dry ingredients, helping diminish the occurrence of “lumpiness”
or uneven texture when reconstituting powdered infant formula.
We
purchase animal cartilage from third-party suppliers, including overseas
slaughtering houses, for the production of chondroitin sulfate, a substance that
provides nutrients for joints, tendon, ligaments and bones, in our Meitek
facility.
Suppliers
and Supplier Arrangements
Prior to
September 2008, we were able to meet our milk powder production needs by
purchasing raw milk on the open market in established dairy regions in northern
and northeastern China. We generally negotiate the purchase price of raw milk
with many dairy farmers and cooperatives.
8
In the
fiscal year ended March 31, 2010, we have been purchasing approximately 90% of
our milk powder from Fonterra in New Zealand. We generally negotiate the prices
for each separate purchase on spot and do not sign long term contracts with our
suppliers.
Prior to
June 2007, we obtained our supply of whey protein powder from Honnete, a large
volume importer of processed dairy products in China. Honnete, a company
controlled by Liang Zhang, our chairman and chief executive officer, is a major
supplier of China’s whey protein powder. Beginning in June 2007, we began
sourcing our whey protein powder directly from Eurosérum S.A.S (“Eurosérum”),
Honnete’s supplier in France.
Sales
and Distribution
Sales
We
generally sell our products directly to distributors and in limited
circumstances directly to retailers. Our recent marketing efforts for our
nutritional products have focused on extending retail coverage in terms of
geography and market sectors. Our sales and marketing approach combines
advertising, brand-building and store-level promotions. Our sales team of more
than 300 employees use our customer relations management, or CRM, database in
order to acquire, process, and manage targeted customer
information.
We have
built a sales network that currently covers 30 provinces and provincial-level
municipalities. Our sales group is divided into multiple sub-sales regions. Each
sub-sales region covers between eight to 20 urban sales areas which acts as an
independent operating unit, while each urban sales area covers three to 20
county sales areas. As of March 31, 2010, we had a sales and marketing force of
more than 3,300 employees, complemented by more than 18,000 commissioned field
nutrition consultants or retail site promoters employed by our distributors and
sub-distributors to promote and sell our products.
Although
we sell primarily to our distributors and a few resellers, our sales teams work
directly with each retail outlet to manage the sales process and to collect
customer and purchasing related data. We use multiple criteria to select our
distributors, including reviewing each potential distributor’s financial
condition. We intend to expand our sales organization into additional cities and
municipalities that we do not currently serve. City managers are rotated
periodically among various cities. We have recently set up a sales budget
management team to manage our sales expenses and to supervise the execution of
our budgeting plan. This team reports directly to the president of marketing and
sales.
We
compensate our sales personnel through a combination of fixed salaries and
bonuses based on sales growth. Our targeted sales incentive programs compensate
our sales personnel on a product-specific level, thereby enabling us to
incentivize our sales personnel to focus their sales and promotion efforts on
certain product lines, such as our premium product lines or larger product
packages.
Distribution
We
primarily work directly with over 540 independent distributors, who in turn work
with over 1,000 independent sub-distributors, and more than 71,000 retail
outlets. Our packaging subsidiary, Shengyuan Nutrition, also serves as our
national distribution center for our distributors in China. From the beginning
of 2007, prior to the melamine contamination incident, we only offered credit
for our products to a few selected distributors. In light of the financial
difficulties experienced by our distributors as a result of the melamine
contamination incident, we extended credit to more distributors and increased
the amount of credit we granted to distributors whom we had previously extended
credit to. We ask our distributors to provide monthly inventory reports which
allows us to monitor their inventory levels. Our sales personnel also regularly
inspect distributors’ inventories to identify and control any potential
inventory buildup by our distributors. We employ trucking companies locally and
nationally to distribute retail packaged products to various regional and
provincial distributors.
Distributors
normally have exclusive distribution rights in their respective regions and
cities to distribute our products, and are also responsible for developing the
sub-distributors in their own region and cities. We typically enter into a
contract with each of our distributors that establishes the range of sales
obligations and their respective pricing ranges. However, our obligation to sell
and the distributor’s obligation to purchase arise only at the time
a
9
purchase
order is accepted. We seek to carefully manage our distributors through an
evaluation system that monitors and grades each distributor with respect to
performance criteria such as monthly sales and investment in promotional
activities. We seek to incentivize well-performing distributors by providing
discounts, larger sales territory and other incentives. While we do not directly
manage our sub-distributors, we do track sub-distributor performance through
coordinated efforts between our own sales personnel in the field and
distributors. Our distributors generally have the right to return products due
to package damage.
We
currently distribute our nutritional products across China. Our logistics center
in our Qingdao facilities occupies an area of 14,000 square meters. This
logistics center can currently dispatch 6,900 tons of our products for shipment
to our distributors per month. Our Qingdao facility also has the capability to
respond to urgent requests for product shipments within an average of five
days.
We
currently work with approximately 22 transportation companies that transport our
goods directly from our Qingdao facilities to distributors in a timely and
efficient manner.
We have
an enterprise resource planning system, or ERP system, which is a financial
information system with an inventory module that manages and records inventory
transactions.
Seasonality
Our
business experiences some seasonal fluctuations. Summer time is typically a slow
time for the infant formula market because the population generally consumes
less food during the summer. Furthermore, Chinese parents tend to choose the
summer time to switch from milk feeding to more concrete food for their babies.
As a result, we generally experience weaker sales in our first and second fiscal
quarters.
Marketing,
Advertising and Promotion
Advertising
We
advertise through various media, including television, print media and the
Internet. Additionally, we conduct promotional activities with supermarket
chains and entertainment companies in order to reach our target
market.
We
started nationwide television advertising coverage in September 2006. In certain
cases, we supplement our nationwide television coverage with local television
coverage. We also pursue advertising over the Internet. Our advertising spending
was $16.7 million, $72.8 million and $30.3 million for the fiscal years ended
March 31, 2010, 2009 and 2008, respectively. Our advertising spending has
enabled us to secure prime-time placements with China Central Television and
other premium regional or satellite television stations. After an aggressive
advertising and promotional campaign from October 2008 to March 2009 to regain
market share in the aftermath of the melamine contamination incident, we slowed
down our advertising and promotional expenses in the fiscal year ended March 31,
2010 to better utilize our resources. We plan to resume more active advertising
activities in the near future and we expect our advertising expenditure will
increase in the fiscal year ending March 31, 2011.
Marketing
and Promotion
As part
of our sales and marketing approach, our sales force works with more than 14,000
healthcare facilities across China to provide maternity, infant nutrition and
health education programs. We have also established a national customer service
call center providing live assistance and a toll-free line to provide consumers
with prenatal, nursing, baby care education, product information, and address
complaints and dispute resolution.
We
provide displays, posters and other promotional print to retail outlets and
sales consultants employed by our distributors at each point of sale. We also
pay entry fees to various retail outlets to place our products within such
outlets. We collect customer information through surveys voluntarily provided by
each customer via the point of sale or via mailed forms provided to our
customers in each product package. We also have promotional activities with
supermarket chains and entertainment companies in order to reach our target
market.
10
Quality
Control
We place
primary importance on quality. We have established quality control and food
safety management systems for the purchase of raw materials, raw milk checks,
raw milk processing, packaging, storage and transportation. We use commercial
strength 25KG poly kraft bags for packaging before shipping the formula products
to our retail packaging and distribution facilities. Additionally, we maintain
cold storage areas at each of our three raw milk processing facilities to store
fluid milk. All of our processing facilities are equipped with in-house
laboratories for quality assurance and quality control purposes. Our laboratory
in Qingdao has been qualified as a National Standard Laboratory by the China
National Accreditation Service for Conformity Assessment.
In order
to ensure the quality and safety of our ingredients and products, we have also
installed testing equipment and have implemented control procedures at each
stage of production, including at the initial raw material purchase stage. There
are over 1,100 quality control points throughout the entire production process,
including 24 quality control points at the milk collection stations. We employ
strict internal procedures and monitoring by highly trained employees during
production, transportation and storage. Additionally, we have been increasing
our investment in quality control equipment and training. All policies relating
to quality control are subject to PRC laws and regulations.
Highlights
of our quality control procedures are summarized below, organized by the main
stages of production:
Imported
Milk Powder and Whey Protein:
|
·
|
Procurement
staff inspects the Certificate of Analysis to ensure the products are
manufactured and tested according to production countries’ national
standard;
|
|
·
|
Entry-Exit
Inspection and Quarantine of the People’s Republic of China performs
quality test to ensure the products are up to national standard and issue
a Sanitary Certificate; and
|
|
·
|
Central
Lab staff of the Company performs detailed test on quality and nutritional
ingredients of the products before using them in
production.
|
Purchase
of Raw Milk:
|
·
|
Raw
milk procurement manager conducts pre-purchase assessment of dairy farmers
and requests issuance of clean bill of health for dairy
cows;
|
|
·
|
Procurement
staff conducts on-site inspection in compliance with our quality standards
and rejects nonconforming supply;
|
|
·
|
Inspection
of specimen—sampling in the process of raw milk collection for inspection
at our facilities pursuant to national standards;
and
|
|
·
|
Sterilization
of equipment for raw milk
collection.
|
Milk
Powder Production:
|
·
|
Compliance
with production process control procedure, HACCP Plan implemented at all
plants;
|
|
·
|
All
raw materials are subject to prior
inspection;
|
|
·
|
Detailed
process designed for all parts of the production process including
pretreatment, vaporization, drying, powder receiving, cooling and
packaging;
|
|
·
|
Maintain
hygiene standards for staff, equipment, environment and any other object;
and
|
|
·
|
Inspection
conducted throughout the production
process.
|
11
Packaging,
Storage and Transport:
|
·
|
Establishment
and practice of total process management with respect to product
identification and traceability;
|
|
·
|
Inspection
before warehousing of products;
|
|
·
|
Maintain
hygiene standards in the course of transport and storage;
and
|
|
·
|
Products
must be positioned according to their category during transport and
storage.
|
In the
fiscal year ended March 31, 2010, we have been importing approximately 90% of
our milk powder from New Zealand. There are three steps of quality control for
imported milk powder: (1) the exporters conduct their own quality control before
they ship the milk powder; (2) all of our milk powder imports are inspected by
China’s import-export inspection and quarantine authorities at landing, pursuant
to a national standard of inspection, and (3) our Qingdao laboratory tests each
batch of imported milk powder using strict standards for quality
assurance.
Research
and Development
Our
research and development activities focus on new product formulation, new
ingredient development, creation of new methods to incorporate certain nutrients
in our products, and improvement in product tastes and ingredient shelf
stabilities. We engage in regular product refinement and new product development
for our dairy-based formula products, as well as other forms of foods and
nutritional supplements.
We
utilize our research and development facilities to engage in the development of
bringing our infant formula products closer to the quality of breast milk and
promote our brand image. We also engage third-party research institutions to
research and develop such trial products for us.
We seek
to leverage our research and development resources in order to extend our new
product pipeline. We believe we can accomplish this goal with new formulations
and product concepts in dairy-based formula products as well as other
nutritional food products and supplements.
In
addition to new formulations and products, we have also developed a variety of
delivery systems such as orally delivered supplements in a pill format and
single use packages which can provide the formula to the end-user in convenient
single packets instead of bulky large canisters.
We also
plan to open a new research and development facility in Beijing, which we plan
to be operational by late calendar year 2010.
During
each of the fiscal years ended March 31, 2010, 2009 and 2008, we spent
approximately 0.2% of net sales per year on research and
development.
Competition
The
infant formula industry in China is highly competitive. We generally compete
with both multinational and domestic infant formula producers. Competitive
factors include brand recognition, distribution network, quality, advertising,
formulation, packaging and price. Many of our competitors have significant
market share in the markets we compete in. Our principal competitors can be
classified generally into the following two groups:
Multinational
Producers
|
·
|
Abbot
Laboratories’ Ross Products Division, a U.S. producer and distributor of
infant formulas marketed under the brand names of Similac and Enfalac
family of formulas;
|
|
·
|
Mead
Johnson Nutrition Co., or Mead Johnson, formerly a Bristol-Myers Squibb
Company Division, a U.S. producer and distributor of the Enfamil family of
formulas;
|
12
|
·
|
Groupe
Danone SA’s Numico division, or Numico, a Dutch producer of baby foods,
which sells and markets infant formula products in China under the Dumex
brand;
|
|
·
|
Nestlé
Suisse SA, or Nestlé, a Swiss producer and distributor of starter and
follow-up formulas, milk, cereals, oral supplements and performance foods
marketed under Nestlé brands such as Carnation;
and
|
|
·
|
Wyeth,
a U.S. producer and distributor of infant formula sold under private label
brands.
|
Domestic
Producers
|
·
|
Inner
Mongolia Yili Industrial Group Co., Ltd., or Yili, a PRC producer and
distributor of liquid and powdered milk under their Yili
brand;
|
|
·
|
Beingmate
Group Company Limited, or Beingmate, a PRC producer and distributor of
infant formula products under their Beingmate
brand;
|
|
·
|
Guangdong
Yashili Group Co., Ltd., or Yashili, a PRC consumer brand marketer which
sells a line of infant formula products under their Yashili brand;
and
|
|
·
|
American
Dairy, Inc., a PRC producer and distributor of milk formula products under
their Feihe brand.
|
According
to data collected by the PRC National Commercial Information Center, or CIC, an
entity affiliated with the PRC General Chamber of Commerce responsible for
collecting retail sales data, the top ten brands accounted for 83.0% of total
infant formulas sold in China in calendar year 2009.
Intellectual
Property
All of
our product formulations have been developed in-house and are proprietary. We
have not registered or applied for protections in China for most of our
intellectual property or proprietary technologies relating to the formulations
of our powdered infant formula. See Item 1A. Risk factors—Risks Related to Our
Business—Failure to adequately protect our intellectual property rights may
undermine our competitive position, and litigation to protect our intellectual
property rights may be costly. Although we believe that, as of today,
patents and copyrights have not been essential to maintaining our competitive
market position, we intend to assess in the future whether to seek patent and
copyright protections for those aspects of our business that provide significant
competitive advantages.
As of
March 31, 2010, we had 175 registered trademarks in China, one registered
trademark in Hong Kong, one registered trademark in the United States, and 3
registered trademarks in France. Additionally, we had 38 trademark applications
pending approval in China.
We rely
on trade secret protection and confidentiality agreements to protect our
proprietary information and know-how. Our management and each of our research
and development personnel have entered into annual employment contracts, each of
which includes a confidentiality clause and a clause acknowledging that all
inventions, designs, trade secrets, works of authorship, developments and other
processes generated by them on our behalf are our property, and assigning to us
any ownership rights that they may claim in those works. Despite our
precautions, it may be possible for third parties to obtain and use, without our
consent, intellectual property that we own or are licensed to use. Unauthorized
use of our intellectual property by third parties, and the expenses incurred in
protecting our intellectual property rights, may adversely affect our business.
See Item 1A. Risk factors—Risks Related to Our Business—Failure to
adequately protect our intellectual property rights may undermine our
competitive position, and litigation to protect our intellectual property rights
may be costly.
Environmental
Matters
Our
manufacturing facilities are subject to various pollution control regulations in
China with respect to noise, water and air pollution and the disposal of waste
and hazardous materials. We are also subject to periodic inspections by local
environmental protection authorities in China. We are not currently subject to
any pending actions alleging any violations of applicable PRC environmental
laws.
13
Our
Employees
As of
March 31, 2010, we employed approximately 5,800 employees in all of our
facilities, with approximately 140 head office management staff and research and
development employees, approximately 2,300 production employees, and
approximately 3,300 sales and marketing employees. Our employees are not
represented by a labor organization or covered by a collective bargaining
agreement. We have not experienced any work stoppages.
We offer
our employees both a base salary and a profit sharing program composed of
performance bonuses and rewards for exceptional performance. As required by PRC
regulations, we participate in various employee benefit plans that are organized
by municipal and provincial governments, including pension, work-related injury
benefits, maternity insurance, medical and unemployment benefit plans. We are
required under PRC law to make contributions to the employee benefit plans at
specified percentages of the salaries, bonuses and certain allowances of our
employees, up to a maximum amount specified by the local government from time to
time. Members of the retirement plan are entitled to a pension equal to a fixed
proportion of the member’s salary amount at the member’s retirement
date.
Regulation
The food
industry, of which nutritional and infant formula products form a part, and
medical institutions, are subject to extensive regulations in China. This
section summarizes the most significant PRC regulations governing our business
in China.
Food
Hygiene and Safety Laws and Regulations
As a
producer of nutritional products, and particularly dairy-based infant formula
products, in China, we are subject to a number of PRC laws and regulations
governing the manufacturing (including composition of ingredients), labeling,
packaging, safety and hygiene of food products:
|
·
|
the
PRC Product Quality Law;
|
|
·
|
the
PRC Food Safety Law;
|
|
·
|
the
Implementation Rules on the PRC Food Safety
Law;
|
|
·
|
the
Dairy Product Industrial Policies (2009
Version);
|
|
·
|
the
Regulation on the Supervision and Administration of the Quality and Safety
of Dairy Products;
|
|
·
|
The
Outlines of the Rectification and Revival of the Dairy
Industry;
|
|
·
|
the
Measures of the Administration on the New
Food-Additives;
|
|
·
|
the
Measures of the Filing of the Enterprise Standard of the Food
Safety;
|
|
·
|
the
Implementation Rules on the Administration and Supervision of Quality and
Safety in Food Producing and Processing
Enterprises;
|
|
·
|
the
Regulation on the Administration of Production Licenses for Industrial
Products;
|
|
·
|
the
General Standards for the Labeling of Prepackaged
Foods;
|
|
·
|
the
Implementation Measures on Examination of Dairy Product Production
Permits;
|
|
·
|
the
Standardization Law;
|
|
·
|
the
Raw Milk Collection Standard;
|
14
|
·
|
the
Whole Milk Powder, Skimmed Milk Powder, Sweetened Whole Milk Powder and
Flavored Milk Powder Standards; and
|
|
·
|
the
General Technical Requirements for Infant Formula Powder and Supplementary
Cereal for Infants and Children.
|
These
laws and regulations set out safety and hygiene standards and requirements for
various aspects of food production, such as the use of additives, production,
packaging, handling, labeling and storage, as well as facilities and equipment.
Failure to comply with these laws and regulations may result in confiscation of
our products and proceeds from the sales of non-compliant products, destruction
of our products and inventory, fines, suspension of production and operation,
product recalls, revocation of licenses, and, in extreme cases, criminal
liability.
As a
result of the melamine contamination incident, the PRC government authorities
have conducted extensive dairy industry inspections. In addition to the initial
22 companies implicated in the incident, these subsequent government inspections
have identified other companies with unacceptable contamination in their
products. On October 7, 2008, the State General Administration of Quality
Supervision, Inspection and Quarantine (“AQSIQ”) issued a national standard on
the detection of melamine in raw milk and dairy based products. On October 9,
2008, the State Council promulgated with immediate effect a Regulation for the
Quality and Safety Supervision of Dairy Based Products, which, among other
things, imposes more stringent requirements for inspection, production,
packaging, labeling and product recall on dairy product producers. This
regulation also established a “Black-List” system to ensure that illegal
business operators in the dairy production chain are timely disclosed and
severely punished.
Environmental
Regulations
We are
subject to various governmental regulations related to environmental protection.
The major environmental regulations applicable to us include:
|
·
|
the
Environmental Protection Law of the
PRC;
|
|
·
|
the
Law of PRC on the Prevention and Control of Water
Pollution;
|
|
·
|
Implementation
Rules of the Law of PRC on the Prevention and Control of Water
Pollution;
|
|
·
|
the
Law of PRC on the Prevention and Control of Air
Pollution;
|
|
·
|
Implementation
Rules of the Law of PRC on the Prevention and Control of Air
Pollution;
|
|
·
|
the
Law of PRC on the Prevention and Control of Solid Waste Pollution;
and
|
|
·
|
the
Law of PRC on the Prevention and Control of Noise
Pollution.
|
We are
periodically inspected by local environmental protection authorities. Our
operating subsidiaries have received certifications from the relevant PRC
government agencies in charge of environmental protection indicating that their
business operations are in compliance with the relevant PRC environmental laws
and regulations.
Dairy
Industry Access Conditions and Policies
In June
2009, the PRC National Development and Reform Commission, or the NDRC, and the
Ministry of Industry and Information Technology, or the MIIT, jointly
promulgated and issued Dairy Industry Policies (2009 Version), or the Policies.
The Policies set forth the conditions an entity must satisfy in order to engage,
or continue to engage, in the dairy products processing business, including
technique and equipment, product quality, energy and water consumption,
sanitation and environmental protection, as well as production safety. Any new
or continuing dairy products processing projects or enterprises will be required
to meet all the conditions and requirements set forth in the Policies. For
projects or enterprises that already commenced operations before the
promulgation of the Policies, improvements or rectification actions may need to
be taken in order to have such projects or enterprises meet the conditions
before the end of 2010.
15
The
Policies also set forth some requirements relating to the location, processing
capacity and raw milk source for any new or continuing dairy products processing
project or enterprise. Any new or continuing dairy products processing projects
or enterprises that fail to meet the requirements will not be able to procure
land, license, permits, loan facility and electricity necessary for the
processing of dairy products, and those projects or enterprises already in
operation before the promulgation of the Policies will be deregistered and
ordered to shut down if they fail to meet the conditions before the end of 2010.
We believe that all of our existing entities and facilities meet the
requirements under the Access Conditions. See Item 1A. Risk Factors—Risks
Associated with Doing Business in China—Changes in the regulatory
environment for dairy and infant nutrition products in China could negatively
impact our business.
Medical
Institutions
On
February 26, 1994, the State Council promulgated the Regulations of
Administration on Medical Institutions which established the regulations for
establishing, managing and supervision of medical institutions. In particular,
the regulations required a medical institution to be approved by and register
with the applicable administrative department of public health prior to
establishment. On December 14, 2009, the Ministry of Public Health promulgated
the Standards of Medical Inspection Laboratory which set forth the standards for
establishing and managing medical inspection laboratories.
Financial
Information About Segments and Geographic Areas
We have
three reportable segments, which are powdered formula, baby food and nutritional
ingredients and supplements. In addition, in the fiscal year ended March 31,
2010, sales from our other business which includes non-core operations accounted
for 32.6% of our net sales. Please refer to Note 18 to the Consolidated
Financial Statements for further discussion about segments and geographic
areas.
Available
Information
Our
Internet website address is www.synutra.com. We make
available at this address, free of charge, our Annual Reports on Form 10-K,
Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, and all amendments
to those reports as soon as reasonably practicable after such material is
electronically filed with or furnished to the United States Securities and
Exchange Commission, or SEC. Information available on our website is not
incorporated by reference in and is not deemed a part of this Form 10-K. The
public may read and copy any materials we file with the SEC at the SEC’s Public
Reference Room at 100 F. Street, N.E., Washington, DC, 20549. The public may
obtain information on the operation of the Public Reference Room by calling the
SEC at 1-800-SEC-0330. The SEC maintains an Internet site that contains reports,
proxy and information statements, and other information regarding issues that
file electronically with the SEC at www.sec.gov.
ITEM
1A. RISK FACTORS
Because
of the following factors, as well as other factors affecting our financial
condition and operating results, past financial performance should not be
considered to be a reliable indicator of future performance, and investors
should not use historical trends to anticipate results or trends in future
periods.
You
should carefully consider the following risks and other information in this Form
10-K before making an investment decision with respect to our common stock. The
following risks and uncertainties could materially and adversely affect our
business, results of operations and financial condition. The risks described
below are not the only ones we face. Additional risks that we are not presently
aware of or that we currently believe are immaterial may also impair our
business operations.
16
Risks
Related to Our Business
We
are highly dependent upon consumers’ perception of the safety and quality of our
products. Any ill effects, product liability claims, recalls, adverse publicity
or negative public perception regarding particular ingredients or products or
our industry in general, could harm our reputation and damage our brand, and
adversely affect our results of operations.
We sell
products for human consumption, which involves risks such as product
contamination, spoilage and tampering. We may be subject to liability if the
consumption of any of our products causes injury, illness or death. Adverse
publicity or negative public perception regarding particular ingredients, our
products, our actions relating to our products, or our industry in general could
result in a substantial drop in demand for our products. This negative public
perception may include publicity regarding the safety or quality of particular
ingredients or products in general, of other companies or of our products or
ingredients specifically. Negative public perception may also arise from
regulatory investigations or product liability claims, regardless of whether
those investigations involve us or whether any product liability claim is
successful against us.
On
September 16, 2008, China’s Administration of Quality Supervision, Inspection
and Quarantine, or China AQSIQ, announced its finding that the formula products
of 22 Chinese formula producers, including certain lots of our U-Smart products,
were contaminated by melamine, a substance not approved for use in food and
linked to the illness and deaths of infants and children in China. To date,
there have been six reported deaths and approximately 300,000 children have
suffered kidney-related illnesses due to the contaminated infant formula of one
of our competitors. This contamination incident has resulted in significant
negative publicity for the entire domestic dairy and formula industries in China
and demand for domestically-produced dairy and formula products, including our
products, has declined significantly since September 2008 until late 2009. We
recalled our affected U-Smart products as well as all other products produced at
the same facilities in the Hebei and Inner Mongolia regions of China, where we
believe the contaminated milk supplies originated. We also suspended production
at our facilities in Qingdao, Hebei and Inner Mongolia for two weeks pending
government and internal investigations. The total cost of this action was $100.6
million which was recognized as a charge to cost of sales, selling and
distribution expenses and general and administrative expenses in our
consolidated statement of income for the two fiscal years ended March 31,
2010.
Although
we have not confirmed any cases of kidney-related or other illnesses caused by
our products, we cannot assure you that such cases will not surface in the
future. The Chinese government has provided medical screening, treatment, and
care for consumers affected by melamine contamination in infant formula
products. We have contributed a net amount of $2.3 million to a compensation
fund set up by China Dairy Industry Association to settle existing and potential
claims arising in China from families of infants affected by melamine
contamination. We cannot assure you that the Chinese government will not seek
further reimbursement from dairy and formula product manufacturers, including
us.
We
believe the melamine contamination incident negatively impacted our brand and
reputation in China. It also affected investor confidence in us as reflected by
the significant decrease in our stock price after September 16, 2008. We cannot
predict the long-term effect this recall and the negative publicity associated
with the melamine contamination incident will have on our reputation among our
customers, consumers and investors. Our results of operations and financial
position, may, in the future, continue to be severely impacted if our customers
and consumers cease to purchase our products as a result of lingering concerns
from the melamine contamination incident.
In the
past, there have also been occurrences of counterfeiting and imitation of
products in China that have been widely publicized. We cannot guarantee that
contamination or counterfeiting or imitation of our or similar products will not
occur in the future or that we will be able to detect it and deal with it
effectively. Any occurrence of contamination or counterfeiting or imitation
could negatively impact our corporate and brand image or consumers’ perception
of our products or similar nutritional products generally, particularly if the
counterfeit or imitation products cause injury or death to consumers. For
example, in April 2004, sales of counterfeit and substandard infant formula in
Anhui, China caused the deaths of 13 infants as well as harming many others.
Although this incident did not involve the counterfeiting of our products, it
caused significant negative publicity for the entire infant formula industry in
China. The mere publication of information asserting that infant formula
ingredients or products may be
17
harmful
could have a material adverse effect on us, regardless of whether these reports
are scientifically supported or concern our products or the raw materials used
in our products.
We
believe that the melamine contamination incident and any other adverse news
related to formula products in China will also result in increased regulatory
scrutiny of our industry, which may result in increased costs and reduce our
margins and profitability. The government has enhanced its regulations on the
industry aimed to ensure the safety and quality of dairy products, including but
not limited to compulsory batch by batch inspection. This is likely to increase
our operating costs and capital expenditure.
In
addition, we are currently subject to claims and litigation stemming from the
melamine contamination incident. See Item 3, Legal Proceedings. An adverse
judgment in any of these cases could materially and negatively affect our
results of operations or further damage our reputation and brand. Moreover, even
if these claims are not successful, to the extent certain plaintiffs continue to
pursue such claims and litigation, we may suffer continuing harm to our
reputation and brand, which could negatively impact our market share and ability
to compete. In the event any of these claims is successful, other potential
claimants may be more likely to bring similar claims against us in the
future.
If
we fail to obtain raw materials in the quantity and the quality we need, and at
commercially acceptable prices, our results of operations, financial condition
and business prospects would be materially and adversely affected.
Our
business requires certain key raw materials, such as raw milk, milk powder and
whey protein powder. We may experience a shortage in the supply of certain raw
materials in the future, which could materially and adversely affect our
production and results of operations. We do not have guaranteed supply contracts
with any of our raw material suppliers, and some of our suppliers may, without
notice or penalty, terminate their relationship with us at any time. We also
rely on a small number of suppliers for some of our raw materials, such as whey
protein powder and imported milk powder. After the melamine contamination
incident, we began importing milk powder from New Zealand for our U-Smart, Super
and Helanruniu, or Holsteina products as consumers have less confidence in
domestically-produced milk powder. If any supplier is unwilling or unable to
provide us with high quality raw materials in required quantities and at
acceptable prices, we may be unable to find alternative sources or at
commercially acceptable prices, on satisfactory terms, in a timely manner, or at
all. Our inability to find or develop alternative sources could result in delays
or reductions in production, product shipments or a reduction in our profit
margins. Moreover, these suppliers may delay material shipments or supply us
with inferior quality raw materials that may adversely impact the timely
delivery or the quality of our products. If any of these events were to occur,
our product quality, competitive position, reputation and business could
suffer.
In
addition, most of the raw materials used in our business are imported, such as
whey protein powder and milk powder. For example, approximately 90% of the milk
powder used in our production is now imported from New Zealand. Our imported raw
materials are subject to various PRC governmental permit requirements, approval
procedures and import duties, and may also, from time to time, be subject to
export controls and other legal restrictions imposed by foreign countries.
Should the PRC government refuse to issue the necessary permits or approvals to
us or our suppliers, or take any administrative actions to limit imports of
certain raw materials, or if we or our suppliers fail to pay any required import
duties, or if governmental agencies or laws of foreign countries prevent the
timely export of certain raw materials we require to China, our ability to
produce and sell our products in China could be materially and adversely
affected. In addition, import duties increase the cost of our products and may
make them less competitive.
Finally,
certain suppliers of raw materials within our supply chain may contaminate our
raw material supplies or provide us with substandard raw material supplies that
adversely impact the quality of our products exposing our customers to health
risks and damaging our reputation, brand and financial condition. For a more
detailed description of this risk, and in particular the impact of the melamine
contamination incident in China, see Part 1 - Item 1A. Risk Factors — We are highly dependent upon
consumers’ perception of the safety and quality of our products. Any ill
effects, product liability claims, recalls, adverse publicity or negative public
perception regarding particular ingredients or products or our industry in
general, could harm our reputation and damage our brand, and adversely affect
our results of operations.
18
Any
interruption in our supply of milk powder and raw milk could materially and
adversely affect our results of operations, financial condition and business
prospects.
We
currently import approximately 90% of the milk powder used in our production
from New Zealand. The continuity of the milk powder supplies is of critical
importance to our business. The importation of milk powder is influenced by
numerous factors beyond our control, including, among other: (1) export control
policy in the originating countries, (2) China’s government policy and
regulation on milk powder importation as well as China’s custom inspection
standards and (3) acts of God such as wars and natural disasters. Any
interruption in our milk powder supplies could have a material adverse effect on
our results of operations, financial condition and business prospects. In
addition, we currently source substantially all of our milk powder from one
supplier, Fonterra. If Fonterra fails to deliver the milk powder we need on the
terms we have agreed, we may not be able to find an alternative source at a
comparable price or on other favorable terms, and any delays in securing an
alternative source could result in production delays and late shipments of our
products to distribution and end customers.
We
purchase most of our raw milk from individual dairy farmers and cooperatives
without long-term contractual arrangements. Our raw milk supply is limited by
the ability of the individual dairy farmers and cooperatives to provide raw milk
in the amount and quality to meet our requirements. Raw milk production is, in
turn, influenced by numerous factors beyond our control such as: (1) seasonal
factors, with dairy cows generally producing more milk in temperate weather as
opposed to cold or hot weather and extended unseasonably cold or hot weather
potentially leading to lower than expected production; (2) environmental
factors, with the volume and quality of milk produced by dairy cows closely
linked to the quality of the nourishment provided by the environment around
them; and (3) impact of governmental agricultural and environmental policy, with
government grants, subsidies, provision of land, technical assistance and other
agricultural and environmental policies having a direct effect on the viability
of individual dairy farmers and dairy cooperatives, and the numbers of dairy
cows and quantities of milk they are able to produce. We cannot assure you that
we will be able to establish relationships with additional milk collection
centers or that there will be sufficient supplies of raw milk from individual
dairy farmers and cooperatives to be provided to any milk collection centers.
Any interruption in our supply of raw milk could materially and adversely affect
our ability to produce products that rely on raw milk.
Our
results of operations may be affected by fluctuations in availability and price
of raw materials.
The raw
materials we use are subject to price fluctuations due to various factors beyond
our control, including increasing market demand, inflation, severe climatic and
environmental conditions, commodity price fluctuations, currency fluctuations,
changes in governmental and agricultural regulations and programs and other
factors. We also expect that our raw material prices will continue to fluctuate
and be affected by inflation in the future. Changes to our raw materials prices
may result in increases in production and packaging costs, and we may be unable
to raise the prices of our products to offset these increased costs in the
short-term or at all. As a result, our results of operations may be materially
and adversely affected.
We
might face inventory write-down if milk powder inventory continues to increase
and milk powder prices continue to decline. We maintain inventories of raw
materials and finished products, and our inventories may spoil.
In the
fiscal year ended March 31, 2010, all of milk powder used for the production of
our Super, U-Smart, and Helanruniu series of products is imported. However, our
domestic production facilities continued to purchase raw milk locally to produce
milk powder for commercial resale and for our Mingshan series of products. As a
result of the melamine contamination incident, there has been a decline in the
consumption of dairy based products in the PRC, and a significant increase in
milk powder imports. This has caused a nationwide inventory build up of
domestically produced milk powder in the PRC. According to the Dairy Industry
Association of China, as of March 31, 2009, surplus milk powder inventory in the
PRC was estimated at 300,000 tons. Such inventory build up has caused a
significant decline in domestically produced milk powder prices. For the fiscal
year ended March 31, 2010, we took a write-down of our inventory of industrial
milk powder of $6.7 million. If milk powder inventory continues to rise and the
milk powder prices continue to fall, we might face significant inventory
write-down which will adversely affect our financial results.
19
Most of
our finished products have an average shelf life of 18 to 24 months before the
product is opened. Our raw materials, excluding raw milk, have an average shelf
life of 12 months. Our inventory levels are based, in part, on our expectations
regarding future sales. While we do not currently maintain large inventory
levels for long periods, we may in future periods experience inventory buildup
if our sales slow for any reason. Any significant shortfall in sales may result
in higher inventory levels of raw materials and finished products than we
require, thereby increasing our risk of inventory spoilage and corresponding
inventory write-downs and write-offs, which may materially and adversely affect
our results of operations.
Any
major outbreak of illness or disease relating to cows in China and in the
regions in which we import milk powder could lead to significant shortfalls in
the supply of our raw milk and milk powder, and could result in consumers
avoiding dairy products, which could result in substantial declines in our sales
and possibly substantial losses.
A major
outbreak of any illness or disease in cows in China and globally could lead to a
serious loss of consumer confidence in, and demand for, dairy products. A major
outbreak of mad cow disease (bovine spongiform encephalopathy), bovine
tuberculosis, or bovine TB, or other serious disease in the principal regions
supplying our raw milk and milk powder could lead to significant shortfalls in
the supply of our raw milk and milk powder. Limited cases of bovine TB have
occurred in several parts of China in the past. Furthermore, adverse publicity
about these types of concerns, whether or not valid, may discourage consumers
from buying dairy products or cause production and delivery disruptions. If
consumers generally were to avoid our products, our sales would decline
substantially and we could suffer substantial losses.
We
may experience problems with product quality or product performance, or the
perception of such problems, which could materially and adversely affect our
reputation or result in a decrease in customers and revenue, unexpected expenses
and loss of market share.
Our
operating results depend, in part, on our ability to deliver high quality
products on a timely and cost-effective manner. Our quality control and food
safety management systems are complex. For example, there are over 1,100 quality
control points throughout the whole production process. If the quality of any of
our products deteriorated, it could result in delays in shipments, cancellations
of orders or customer returns and complaints, loss of goodwill, and harm to our
brand and reputation. In addition, following the melamine contamination
incident, we purchase all of the milk powder used for our U-Smart, Super and
Helanruniu, or Holsteina products from New Zealand. We may be unable to exercise
the same degree of quality control over this overseas supplier as we can over
our own facilities. Any quality problems associated with the milk powder
produced by this supplier would also affect our products’ quality and lead to
negative publicity against us, materially and adversely affecting our reputation
and brand, and causing a decrease in sales of our products and a loss of market
share. For example, the melamine contamination incident in China has resulted in
certain of our products being contaminated, impacting our brand and
reputation.
Product
liability claims against us could result in adverse publicity and potentially
significant monetary damages.
As with
other infant formula producers, we are also exposed to risks associated with
product liability claims if the consumption of infant formula products we sell
results in injury or death. We cannot predict what impact such product liability
claims or resulting negative publicity would have on our business or on our
brand image. The successful assertion of product liability claims against us
could result in potentially significant monetary damages, diversion of
management resources and require us to make significant payments and incur
substantial legal expenses. We do not have product liability insurance and have
not made provisions for potential product liability claims. Therefore, we may
not have adequate resources to satisfy a judgment if a successful claim is
brought against us. Even if a product liability claim is not successfully
pursued to judgment by a claimant, we may still incur substantial legal expenses
defending against such a claim and our brand image and reputation would suffer.
Finally, serious product quality concerns could result in governmental actions
against us, which, among other things, could result in the suspension of
production or distribution of our products, loss of certain licenses, or other
governmental penalties.
20
For
example, the melamine contamination incident in China in 2008 resulted in
certain of our products being contaminated. As a result, lawsuits have been
filed against us in both China and the U.S. by Chinese families alleged to have
been affected by melamine contamination, seeking compensatory and punitive
damages. See “Item 3 - Legal Proceedings.” We may incur significant legal
expenses and be subject to significant monetary damages in connection with such
claims, which may adversely affect our results of operations and further harm
our reputation and damage our brand. Further, we cannot assure you that we will
not become subject to future product liability claims in connection with the
melamine contamination incident. See Part 1 - Item 1A. Risk Factors — We are highly dependent upon
consumers’ perception of the safety and quality of our products. Any ill
effects, product liability claims, recalls, adverse publicity or negative public
perception regarding particular ingredients or products or our industry in
general could harm our reputation and damage our brand and adversely affect our
results of operations.”
Our
sales, results of operations, brand image and reputation could be materially and
adversely affected if we fail to efficiently manage our operations without
interruption, or fail to ensure that our products are delivered on
time.
Our
business requires successful coordination of several sequential and complex
processes, the disruption of any of which could interrupt our operations and
materially and adversely affect our relationships with our distributors,
sub-distributors and end-customers, our brand name and reputation, and our
financial performance. Our operations involve the coordination of raw material
sourcing from third parties, internal production processes and external
distribution processes. We may face difficulties in coordinating the various
aspects of our production processes, resulting in downtime and
delays.
In
addition, we may encounter interruptions in our production processes due to a
catastrophic loss or events beyond our control, such as fires, explosions, labor
disturbances, earthquakes or other natural disasters. If there is a stoppage in
production at any of our facilities, even if only temporary, or delays in
deliveries to our customers, our business and reputation could be materially and
adversely affected. Along with many other producers of dairy and consumer
products in China, we generally rely on third-party logistics companies and
distributors for the delivery of our products. Delivery may be disrupted for
various reasons, many of which are beyond our control, including natural
disasters, weather conditions or social unrest and strikes, which could lead to
delayed or lost deliveries. In addition, transportation and related
infrastructure conditions are often generally under-developed in some of the
regions where we sell our products. We currently do not have business
interruption insurance to offset these potential losses, delays and risks, so a
material interruption of our business operations could materially damage our
business.
We
rely primarily on third-party distributors and cannot assure you that their
marketing and distribution of our products will be effective or will not harm
our brand and reputation. Moreover, if we fail to timely identify and appoint
additional or replacement distributors as needed, or are unable to successfully
manage our distribution network, our operating results could
suffer.
We do not
sell our products directly to our end customers. Instead, we primarily rely on
third-party distributors and sub-distributors for the distribution and sales of
our products. We sell our products through an extensive nationwide distribution
and sales network covering 30 provinces and provincial-level municipalities in
China. As of March 31, 2010, this network comprised over 540 independent
distributors and over 1,000 independent sub-distributors who sell our products
in over 71,000 retail outlets. Our distributors normally have exclusive
distribution rights in their respective regions, and are also responsible for
developing the sub-distributors located in their own regions. In addition, our
distributors are not required to exclusively distribute our products. We
typically do not enter into long-term agreements with distributors and have no
control over their everyday business activities. Consequently, our distributors
may engage in activities that are prohibited under our arrangements with them,
that violate PRC laws and regulations governing the dairy industry or other PRC
laws and regulations generally, or that are otherwise harmful to our business or
our reputation. Due to our dependence on distributors for the sale and
distribution of our products to retail outlets, any one of the following events
could cause material fluctuations or declines in our revenue and have a material
adverse effect on our financial condition and results of
operations:
|
·
|
reduction,
delay or cancellation of orders from one or more of our
distributors;
|
21
|
·
|
selection
or increased sales by our distributors of our competitors’ products;
and
|
|
·
|
our
failure to timely identify and appoint additional or replacement
distributors upon the loss of one or more of our
distributors.
|
The
competition for distributors is intense in our industry in China and many of our
competitors are expanding their distribution networks in China. We may not be
able to compete successfully against the larger and better-funded sales and
marketing operations of some of our current or future competitors, especially if
these competitors provide more favorable arrangements for distributors. As a
result, we may lose some of our distributors to our competitors, which may cause
us to lose some or all of our favorable arrangements with such distributors and
may even result in the termination of our relationships with some of our
distributors. While we do not believe we are substantially dependent upon any
individual distributor, finding replacement distributors could be time-consuming
and any resulting delay may be disruptive and costly to our business. In
addition, we may not be able to successfully manage our distributors and the
cost of any consolidation or further expansion of our distribution network may
exceed the revenue generated from these efforts. The occurrence of any of these
factors could result in a significant decrease in the sales volume of our
products and therefore materially harm our financial condition and results of
operations.
Our
results of operations and business prospects may be impaired by changing
consumer preferences if we do not develop and offer products to meet changing
preferences.
Consumer
preferences evolve over time and the success of our products depends on our
ability to identify the tastes and nutritional needs of our customers and to
offer products that appeal to their preferences. We introduce new products and
improved products from time to time and incur significant development and
marketing costs. If our products fail to meet consumer preferences, then our
strategy to grow sales and profits with new products will be less
successful.
More
mothers may breastfeed their babies rather than use our products, resulting in
reduced demand for our products and adversely affecting our
revenues.
Our
results of operations are affected by the number of mothers who choose to use
our products rather than breastfeeding their babies. Much publicly available
data suggests that breastfeeding has many health benefits for the baby that
cannot be replicated by dairy-based infant formula products. Additionally,
popular literature, cultural pressure, government policies and medical advice in
China generally promote the benefits of breastfeeding. For example, on August 1,
2007, China’s Ministry of Health issued an Infant Feeding Strategy which
promoted breastfeeding and requested all local relevant departments to publicize
the benefits of breastfeeding through radio broadcasting, television and
newspapers during World Breastfeeding Week, which took place in early August
2007. Thus, to the extent that private, public and government sources
increasingly promote the benefits of breastfeeding, there could be a reduced
demand for our products and our revenues could be adversely
affected.
In
addition, we believe the melamine contamination incident has deteriorated
customer confidence in the safety and quality of infant formula products made in
China and the number of mothers (and future mothers) who choose to breastfeed
their babies may significantly increase. The overall market demand for infant
formula products has slowed and may continue to decline and reduced demand for
our products will negatively impact our revenues and growth
prospects.
The
disruptions in the overall economy and the financial markets that started in
late calendar year 2007 may adversely impact our business and results of
operations and may limit our access to additional financing.
Financial
markets in the United States, Europe and Asia had experienced extreme
disruptions that started in late calendar year 2007, including, among other
things, extreme volatility in security prices, the failure and near failure of a
number of large financial services companies, severely diminished liquidity and
credit availability, rating downgrades of certain investments and declining
valuations of others.
The
infant formula industry can be affected by macro economic factors, including
changes in national, regional, and local economic conditions, employment levels
and consumer spending patterns. Though the disruptions in
the
22
overall
economy and financial markets was less severe in China than in the U.S., it
could reduce consumer confidence in the economy and negatively affect consumers’
spending, which could be harmful to our financial position and results of
operations.
In
addition, if the capital and credit markets continue to experience volatility
and the availability of funds remains limited, we will incur increased costs
associated with equity and/or debt financing. It is possible that our ability to
access the capital and credit markets may be limited by these or other factors
at a time when we would like, or need, to do so, which could have an adverse
impact on our ability to refinance maturing debt and/or react to changing
economic and business conditions.
Failure
to execute our expansion plan could adversely affect our financial condition and
results of operations.
We may
increase our annual production capacity in the future to meet any expected
increase in demand for our products. Our decision to increase our production
capacity is based in part on our projections of increases in our sales volume
and growth in the size of the infant formula product market in China. If actual
customer demand does not meet our projections, we will likely suffer
overcapacity problems and have idle capacity, which may materially and adversely
affect our financial condition and results of operations. Our future success
depends on our ability to expand our business to address expected growth in
demand for our current and future products. Our ability to add production
capacity and increase output is subject to significant risks and uncertainties,
including:
|
·
|
the
availability and cost of additional funding to expand our production
capacity, build new processing and packaging facilities, make additional
investments in our subsidiaries, acquire additional businesses or
production facilities, purchase additional fixed assets and purchase raw
materials on favorable terms or at
all;
|
|
·
|
delays
and cost overruns as a result of a number of factors, many of which may be
beyond our control, such as problems with equipment vendors and suppliers
of raw materials;
|
|
·
|
failure
to maintain high quality control
standards;
|
|
·
|
global
or local shortage of raw materials, such as raw milk or whey protein
powder;
|
|
·
|
our
inability to obtain, or delays in obtaining, required approvals by
relevant government authorities;
|
|
·
|
diversion
of significant management attention and other resources;
and
|
|
·
|
failure
to execute our expansion plan
effectively.
|
As our
business grows, we will need to implement a variety of new and upgraded
operational and financial systems, procedures and controls, including
improvements to our accounting and other internal management systems by
dedicating additional resources to our reporting and accounting functions, and
improvements to our record keeping and contract tracking system. We will need to
respond to competitive market conditions and continue to enhance existing
products and develop new products, and retain existing customers and attract new
customers. We will also need to recruit more personnel and train and manage our
growing employee base. Furthermore, we will need to maintain and expand our
relationships with our current and future customers, suppliers, distributors and
other third parties, and there is no guarantee that we will
succeed.
If we
encounter any of the risks described above, or are otherwise unable to establish
or successfully operate additional production capacity or to increase production
output, we may be unable to grow our business and revenues, reduce our operating
costs, maintain our competitiveness or improve our profitability, and our
business, financial condition, results of operations and prospects may be
adversely affected.
23
Any
future expansion into many of China’s major urban centers may be costly,
time-consuming and difficult. If we do not successfully expand into such
markets, our results of operations and prospects may be materially and adversely
affected.
Our
future success may depend upon our ability to successfully expand into many of
China’s major urban centers. To further promote our brand and generate demand
for our products in such markets, we may increase our spending on marketing and
promotion. We may be unable to attract a sufficient number of distributors with
the experience and ability to penetrate these markets, and our selected
distributors may not be suitable for selling our products in these markets for
other reasons. We may also fail to attract new customers in such markets who may
have less familiarity with our brand and products. Furthermore, we may fail to
anticipate and address competitive conditions in these new markets that are
different from those in our existing primary markets. These competitive
conditions may make it difficult or impossible for us to effectively operate in
these markets. If our expansion efforts in existing and new markets are
unsuccessful, our results of operations and prospects may be materially and
adversely affected.
Part
of our strategy involves the development of new products and new business
segments, and if we fail to timely develop new products or we incorrectly gauge
the potential market for new products, our financial results would be adversely
affected.
We plan
to utilize our in-house research and development capabilities to develop new
products that could become new sources of revenue for us in the future and help
us to diversify our revenue base. We have also entered into new business
segments such as the prepared baby food segment, and the nutritional ingredients
and supplements segment. Our future research and development efforts will focus
on further expanding our product offerings beyond dairy-based nutritional
products. If we fail to timely develop these and other new products or if we
incorrectly gauge market demand for such new products, we may not be able to
grow our sales revenue at expected growth rates and may incur expenses and
capital expenditure costs relating to the development of new products that are
not offset by the sales they generate.
We
operate in a competitive environment, which may lead to declining revenue growth
or other circumstances that would negatively affect our results of
operations.
The
market for pediatric nutritional products is competitive, and we believe that
competition in this market will continue to intensify. We believe that the
principal competitive factors in our markets are brand recognition, quality,
advertising, formulation, packaging, and price. We face significant competition
from a number of competitors, including multinational companies, such as Abbot
Laboratories’ Ross Products Division, Mead Johnson, Nestle, Numico and Wyeth,
and domestic companies, such as Beingmate, Yashili, Feihe and Yili. See “Item 1.
Business—Competition”. Many of our competitors have longer operating histories,
greater name recognition, significantly larger market shares, access to larger
customer bases and significantly greater financial resources and economies of
scale in financial, sales and marketing, production, distribution, technical and
other resources than we do. Some of these competitors have used, and we expect
will continue to use, more aggressive pricing strategies, greater amounts of
incentives and subsidies for distributors, retailers and customers and more
advanced processes and technologies. Furthermore, consolidation among industry
participants in China may potentially result in stronger domestic competitors
that are better able to compete as end-to-end suppliers as well as competitors
who are more specialized in particular areas and geographic markets. This could
have a material adverse effect on our business, financial condition, results of
operations and prospects. In addition, as a result of the melamine contamination
incident, customers have lost confidence in infant formula produced by domestic
companies for the time being, which gives multinational infant formula companies
an advantage over us.
In order
to compete successfully in our markets, we will need to continue to restore
customer confidence in our brand and products, develop new products and enhance
our product offerings while maintaining price competitiveness. Even if we
successfully restore customer confidence, if and to the extent we fail to
develop new products that differentiate us from our competitors, we may need to
compete largely on price, which may cause our operating margins to decline. Our
inability to compete successfully against competitors and pricing pressures
could result in lost customers, loss of market share and reduced operating
margins, which would adversely impact our results of operations.
24
If
we grant employee stock options and other stock-based compensation in the
future, we will be required to recognize stock-based compensation expense, which
would adversely affect our results of operations.
As of the
date of this Form 10-K, we have not granted any stock-based compensation and
thus have not been required to reflect any such expenses in our results of
operations. We adopted a stock -based compensation plan in June 2008 and intend
to grant certain employees and directors stock-based compensation, which we
believe will be important to attract and retain key personnel. We will be
required to account for compensation costs for all stock options, including
stock options granted to our directors and employees, using the fair value
method and recognize the expense in our consolidated statement of operations in
accordance with Accounting Standards Codification (ASC) 718-10, “Compensation –
Stock Compensation – Overall” (previously Statement of Financial Accounting
Standards (SFAS) No. 123-R, “Share-Based Payment”) which
may have a material adverse effect on our results of operations.
We
may be exposed to liabilities under the Foreign Corrupt Practices Act, and any
determination that we violated the Foreign Corrupt Practices Act could have a
material adverse effect on our business.
We are
subject to the Foreign Corrupt Practice Act, or the FCPA, and other laws that
prohibit improper payments or offers of payments to foreign governments and
their officials and political parties by U.S. persons and issuers as defined by
the statute for the purpose of obtaining or retaining business. We have
operations, agreements with third parties and make sales in China, which may
experience corruption. Our activities in China create the risk of unauthorized
payments or offers of payments by one of the employees, consultants, sales
agents or distributors of our company, because these parties are not always
subject to our control. It is our policy to implement safeguards to discourage
these practices by our employees. However, our existing safeguards and any
future improvements may prove to be less than effective, and the employees,
consultants, sales agents or distributors of our Company may engage in conduct
for which we might be held responsible. Violations of the FCPA may result in
severe criminal or civil sanctions, and we may be subject to other liabilities,
which could negatively affect our business, operating results and financial
condition. In additions, the government may seek to hold our Company liable for
successor liability of FCPA violations committed by companies in which we invest
or that we acquire.
We
have previously experienced certain material weaknesses and deficiencies with
our internal control over financial reporting. Any further failure to accurately
report our financial results, including any resulting restatement, could result
in harm to our business, loss of investor confidence in our financial reporting
and a lower trading price of our common stock, or possibly lead to the delisting
of our common stock by the NASDAQ Global Select Market.
In
connection with the review of our financial statements for the fiscal year ended
March 31, 2008, our independent registered public accounting firm and our
management identified certain material weaknesses and significant deficiencies.
A material weakness is defined by the Public Company Accounting Oversight Board,
or PCAOB, as a deficiency, or a combination of deficiencies, in internal control
over financial reporting, such that there is a reasonable possibility that a
material misstatement of our annual or interim financial statements will not be
prevented or detected on a timely basis. A significant deficiency is defined by
the PCAOB as a deficiency, or combination of deficiencies, in internal control
over financial reporting that is less severe than a material weakness, yet
important enough to merit attention by those responsible for oversight of our
financial reporting.
Due to
the material weaknesses and significant deficiencies in our internal control
over financial reporting for the fiscal year ended March 31, 2008—as evidenced
by the significant number and magnitude of out-of-period adjustments identified
during the year-end and period-end closing process—we previously concluded in
our prior SEC filings that our disclosure controls and procedures and our
internal control over financial reporting were not effective at the reasonable
assurance level, and, prior to the restatement of certain prior periods, that
investors should not rely on our prior financial statements. We have taken steps
to remediate our material weaknesses and deficiencies in our internal control
over financial reporting, and we believe that the material weakness identified
as of March 31, 2008 has been fully remediated, see “Part II. Item 9A. Controls
and Procedures.” of 10-K for the fiscal year ended March 31, 2009. There can be
no assurance that any current or future deficiencies will not contribute to, or
cause, possible material weaknesses in the future or, that we will be able to
implement effectively new or improved controls or that our management or our
independent registered public accounting firm will determine that our disclosure
controls and procedures or our internal control over financial reporting will be
effective in the future.
25
Moreover,
a lack of effective internal control over financial reporting in the future
could cause us to fail to provide accurate financial statements or fail to meet
our reporting obligations, either of which could cause investors to lose
confidence in our reported financial information, and have a negative effect on
the trading price of our common stock. Our failure to meet our reporting
obligations in a timely fashion may also lead to the delisting of our common
stock by the NASDAQ Global Select Market.
Our
success depends to a substantial degree upon our senior management and key
personnel, and our business operations may be significantly and negatively
affected if we fail to attract and retain highly competent senior management and
key personnel.
Our
future success depends substantially on the continued services of our key
personnel including, particularly, Liang Zhang, our chairman and chief executive
officer. We rely substantially on their experience in the dairy and nutritional
products industry, and on their relationships and ability to work with our
suppliers and distributors and other customers. If we lose the services of one
or more of these key personnel, we may not be able to replace them readily, if
at all, with suitable or qualified candidates, and may incur additional expenses
to recruit and retain new officers and key personnel, which could severely
disrupt our business and growth. We do not maintain key-man life insurance for
any of our key personnel.
In
addition, if any of these key personnel joins a competitor or forms a competing
company, we may lose some of our distributors. We have entered into employment
agreements with each of these key personnel, which contain confidentiality and
non-competition provisions. However, if any disputes were to arise between these
key personnel and us, it is not clear, in light of uncertainties associated with
the PRC legal system, what the court decisions would be and the extent to which
these court decisions could be enforced in China, where most of these key
personnel reside and hold some of their assets. See “Item 1A. Risk Factors—Risks Associated
with Doing Business in China—Uncertainties with
respect to the PRC legal system could limit the legal protections available to
you and us.” Furthermore, as we expect to continue to expand our
operations and develop new products, we will need to continue attracting and
retaining experienced management and key research and development
personnel.
Competition
for experienced management and research and development personnel in China is
intense, and the availability of experienced, suitable and qualified candidates
is limited. Competition for these individuals may require us to pay higher
compensation and other benefits in order to attract and retain them, which could
have a material adverse effect on our financial condition and results of
operations. We may also be unable to attract or retain the personnel necessary
to achieve our business objectives, and any failure in this regard could
severely disrupt our business and growth.
Liang
Zhang’s association with other businesses could impede his ability to devote
sufficient time to our business and could present potential conflicts of
interest.
Liang
Zhang, our founder, chairman, chief executive officer and principal beneficial
stockholder, controls several other companies, including Honnete, a supplier of
whey protein powder in China. Liang Zhang devotes most of his time to our
affairs and the remainder of his time to the affairs of the other companies.
Liang Zhang’s decision-making responsibilities for these other companies are
similar in the areas of public relations, risk management and strategic
planning. As a result, conflicts of interest may arise from time to time and we
cannot assure you that they will be resolved in our favor. Additionally, even
though Liang Zhang devotes most of his time to our affairs and is further
accountable to us and our stockholders as a fiduciary, which requires that he
exercise good faith and due care in handling our affairs, we cannot assure you
this will always be the case and his existing responsibilities to other
businesses and entities may limit the amount of time he can spend on our
affairs.
Our
chairman and chief executive officer, Liang Zhang, beneficially owns a
substantial amount of our common stock and will have significant influence over
our corporate affairs.
Our
founder, chairman and chief executive officer, Liang Zhang, beneficially owns
approximately 66.67% of our outstanding common stock through a company owned by
his wife as of March 31, 2010. Accordingly, Liang Zhang will be able to direct
the outcome of matters requiring approval of the stockholders, including the
election of our directors and other corporate actions such as:
26
|
·
|
our
merger with or into another
company;
|
|
·
|
a
sale of our assets; and
|
|
·
|
amendments
to our certificate of
incorporation.
|
The
decisions of Liang Zhang may conflict with our interests or the interests of our
other stockholders.
We
may not succeed in identifying suitable acquisition targets, which could
adversely affect our ability to expand our operations and service offerings and
enhance our competitiveness.
Our
strategy contemplates growth through acquisitions. We have pursued and may in
the future pursue strategic acquisition opportunities to increase our scale and
geographic presence and expand the number of our product offerings. However, we
may not be able to identify suitable acquisition or investment candidates, or,
even if we do identify suitable candidates, we may not be able to complete those
transactions on terms commercially favorable to us or at all, which could
adversely affect our competitiveness and our growth prospects.
If we
acquire other companies in the future, we could face the following
risks:
|
·
|
difficulty
in integrating the target company’s personnel, operations, products,
services and technology into our
operations;
|
|
·
|
the
presence of unforeseen or unrecorded
liabilities;
|
|
·
|
entry
into unfamiliar markets;
|
|
·
|
inability
to generate sufficient revenues to offset acquisition costs;
and
|
|
·
|
tax
and accounting issues.
|
Many
recently joined employees may decide not to work with us or to leave shortly
after joining our company. These difficulties could disrupt our ongoing
business, distract our management and current employees and increase our
expenses, including write-offs or impairment charges. Acquired companies also
may not perform to our expectations for various reasons, including the loss of
key personnel, key distributors, key suppliers or key customers, and our
strategic focus may change. As a result, we may not realize the benefits we
anticipated. If we fail to integrate acquired businesses or realize the expected
benefits, we may lose the return on the investment in these acquisitions or
incur additional transaction costs and our operations may be negatively impacted
as a result. Further, any acquisition or investment that we attempt, whether or
not completed, or any media reports or rumors with respect to any such
transactions, may adversely affect our competitiveness, our growth prospects,
and the value of our common stock.
Our
business is capital intensive and our growth strategy may require additional
capital that may not be available on favorable terms or at all.
We have,
in the past, obtained loans and sold our common stock to raise additional
capital. Our business requires significant capital and although we believe that
our current cash, and cash flow from operations will be sufficient to meet our
present and reasonably anticipated cash needs, we may, in the future, require
additional cash resources due to changed business conditions, implementation of
our strategy to expand our production capacity or other investments or
acquisitions we may decide to pursue. If our own financial resources are
insufficient to satisfy our capital requirements, we may seek to sell additional
equity or debt securities or obtain additional credit facilities. For example,
the melamine contamination incident has significantly impacted our liquidity due
to our product recall and required us to obtain additional funding through
short-term loans for working capital purposes after the incident. In addition to
these short-term loans, we may need to obtain additional private or public
financing including debt or equity financing and there can be no assurance that
such financing will be available as needed or, if available, on terms favorable
to us. Any additional equity financing may be dilutive to stockholders and such
additional equity securities may have rights, preferences or privileges that are
senior to those of our existing common stock. The incurrence of indebtedness
would result in increased debt service obligations and could require us to agree
to
27
operating
and financial covenants that would restrict our operations. If we are unable to
generate sufficient cash flow from operating activities or obtain funds for
required payments of interest and principal on such additional indebtedness, or
if we fail to comply with our debt covenants, we will be in default. In
addition, changes in our debt rating due to the melamine contamination incident
could have a material adverse effect on our interest costs and financing
sources. Financing may not be available in amounts or on terms acceptable to us,
if at all. Any failure by us to raise additional funds on terms favorable to us,
or at all, could limit our ability to expand our business operations and could
harm our overall business prospects.
We
have recently incurred operating losses; this may have a harmful effect on our
business and the value of our common stock.
As a
result of the melamine contamination incident, we have incurred losses for the
quarters ended September 30, 2008, December 31, 2008, March 31, 2009, June 30,
2009, September 30, 2009 and December 31, 2009 from the lingering impact of the
product recall and only returned to profitability in the quarter ended March 31,
2010. Though we expect this to be a short-term financial challenge for us, we
believe the melamine contamination incident has negatively impacted our brand
and reputation in China and may impact our future revenues. We have incurred
expenses relating to the melamine contamination incident and we may incur more
expenses in the future relating to expanding our production capacity or other
investments or acquisitions we may decide to pursue. If our revenue does not
increase or if we fail to maintain our expenses at an amount less than our
projected revenues, we will not be able to achieve or sustain operating
profitability on a consistent basis. Given our planned operating and capital
expenditures as well as our overall business plan, for the foreseeable future we
expect our results of operations to fluctuate, and during this period we may
continue to incur losses. Our lack of profitability may have an adverse effect
on the market value of our common stock and on our cash flow and
liquidity.
We
may face liquidity challenges to meet our debt obligations and may require
additional funding in the future.
As a
result of the melamine contamination incident, we had negative cash flow from
operations of $109.5 million for the fiscal year ended March 31, 2009. In
addition, at March 31, 2009, we had short-term debt of $224.6 million,
consisting of short-term debt from banks in the amount of $182.6 million, an
additional $7.5 million in short-terms loans from related parties, and $34.5
million under our loan agreement (the “RBS Loan”) with The Royal Bank of
Scotland, N.V. (“RBS”) which has been reclassified as a current liability since
we did not meet our financial covenants under the loan as of March 31, 2009. We
amended the RBS Loan in the fiscal year ended March 31, 2010 and our results
improved in the fiscal year ended March 31, 2010. However, we still had negative
cash flow from operations of $11.8 million for the fiscal year ended March 31,
2010 and as of March 31, 2010, we had short-term debt and long-term debt due
within one year of $159.3 million. Our ability to meet our debt obligations will
depend on our future performance, which will be affected by financial, business,
domestic and foreign economic conditions and other factors, many of which we are
unable to control. As a result, we cannot be assured that our revenues will
provide cash flow in excess of our cash needs, and we therefore may have
negative cash flow in the future. If our cash flow is not sufficient to service
our debt, we may be required to obtain additional financing in the future, and
such additional financing may not be available at times, in amounts or on terms
acceptable to us or at all, which would have a material adverse effect on our
business. See “Item 7. Management’s Discussion and Analysis of Financial
Condition and Results of Operations—Liquidity and Capital
Resources.”
We
have limited insurance coverage and do not carry any business interruption
insurance, third-party liability insurance for our production facilities or
insurance that covers the risk of loss of our products in shipment.
Operation
of our facilities involves many risks, including equipment failures, natural
disasters, industrial accidents, power outages, labor disturbances and other
business interruptions. Furthermore, if any of our products are faulty or
contaminated, then we may become subject to product liability claims or we may
have to engage in a product recall. For details about our recall of contaminated
products in connection with the melamine contamination incident, please see
“Item 7. Management’s Discussion and Analysis of Financial Condition and Results
of Operations — Overview.”
We do not
carry any business interruption insurance, product recall or third-party
liability insurance for our production facilities or with respect to our
products to cover claims pertaining to personal injury or property
or
28
environmental
damage arising from defects in our products, product recalls, accidents on our
property or damage relating to our operations. Therefore, our existing insurance
coverage may not be sufficient to cover all risks associated with our business.
As a result, we may be required to pay for financial and other losses, damages
and liabilities, including those caused by natural disasters and other events
beyond our control, out of our own funds, which could have a material adverse
effect on our business, financial condition and results of operations. For
example, all of the costs we have incurred to date and may incur in the future
that are related to our product recall in connection with the melamine
contamination incident is not covered by our existing insurance
policies.
Failure
to adequately protect our intellectual property rights may undermine our
competitive position, and litigation to protect our intellectual property rights
may be costly.
We rely
primarily on trade secrets and other contractual restrictions to protect our
intellectual property. We have not registered or applied for protections in
China for most of our intellectual property or proprietary technologies relating
to the formulations of powdered infant formula that we produce. In order to
protect our proprietary technology and processes, we also rely in part on
nondisclosure agreements with our employees, licensing partners, third-party
producers, consultants, agents and other organizations to which we disclose our
proprietary information. The actions we have taken to protect our intellectual
property rights may not be adequate to provide us with meaningful protection or
commercial advantage. As a result, third parties may use the intellectual
property or proprietary technologies that we have developed and compete with us,
which could have a material adverse effect on our business, financial condition
and operating results.
PRC
intellectual property-related laws and their implementation are still under
development. Accordingly, intellectual property rights and confidentiality
protections in China may not be as effective as in the United States or many
other countries. In addition, policing unauthorized use of proprietary
technology can be difficult and expensive. Litigation may be necessary to
enforce our intellectual property rights and the outcome of any such litigation
may not be in our favor. Given the relative unpredictability of China’s legal
system and potential difficulties enforcing a court judgment in China, there is
no guarantee that we would be able to halt the unauthorized use of our
intellectual property through litigation in a timely manner or at all.
Furthermore, any such litigation may be costly and may divert management
attention away from our business and cause us to expend significant resources.
An adverse determination in any such litigation will impair our intellectual
property rights and may harm our business, prospects and reputation. In
addition, we have no insurance coverage against litigation costs and would have
to bear all costs arising from such litigation to the extent we are unable to
recover them from other parties. The occurrence of any of the foregoing could
have a material adverse impact on our business, financial condition and results
of operations.
We
may be exposed to infringement or misappropriation claims by third parties,
which, if determined adversely against us, could disrupt our business and
subject us to significant liability to third parties.
Our
success largely depends on our ability to use and develop our know-how and
product formulations without infringing upon the intellectual property rights of
third parties. We may be subject to litigation involving claims of infringement
or violation of other intellectual property rights of third parties. The holders
of patents and other intellectual property rights potentially relevant to our
product offerings may be unknown to us or may otherwise make it difficult for us
to acquire a license on commercially acceptable terms.
There may
also be technologies licensed to and relied on by us that are subject to
infringement or other corresponding allegations or claims by others which could
damage our ability to rely on such technologies. In addition, although we
endeavor to ensure that companies that work with us possess appropriate
intellectual property rights or licenses, we cannot fully avoid the risks of
intellectual property rights infringement created by suppliers of raw materials
used in our products, our third-party producers, or by companies with which we
work in cooperative research and development activities. Our current or
potential competitors, many of which have substantial resources and have made
substantial investments in competing technologies, may have obtained or may
obtain patents that will prevent, limit or interfere with our ability to make,
use or sell our products in China or other countries. The defense of
intellectual property claims, including patent infringement suits, and related
legal and administrative proceedings can be both costly and time-consuming, and
may significantly divert the efforts and resources of our technical and
management personnel. Furthermore, an adverse determination in any such
litigation or proceeding to which we may become a party could cause us
to:
29
|
·
|
pay
damage awards;
|
|
·
|
seek
licenses from third parties, which may not be available on reasonable
terms or at all;
|
|
·
|
pay
additional ongoing royalties, which could decrease our profit
margins;
|
|
·
|
redesign
our products, which may be costly, if possible at all;
or
|
|
·
|
be
restricted by injunctions.
|
These
factors could effectively prevent us from pursuing some or all of our business
and result in our customers or potential customers deferring, canceling or
limiting their purchase or use of our products, which could have a material
adverse effect on our financial condition and results of
operations.
Our
Independent Registered Public Accounting Firm issued a clean opinion to our
consolidated financial statements for the fiscal year ended March 31, 2009;
however, it contained an emphasis paragraph raising substantial doubt about our
ability to continue as a going concern.
Our
financial statements for the fiscal year ended March 31, 2009 were prepared
assuming that we would continue as a going concern. By discussing the
difficulties we were experiencing after the melamine contamination incident, we
raised substantial doubt about our ability to continue as a going concern. Our
independent registered public accounting firm issued a clean opinion on our
consolidated financial statements, however, they included an emphasis paragraph
in its report on our fiscal year ended March 31, 2009 financial statements with
a statement raising substantial doubt regarding our ability to continue as a
going concern. In the fiscal year ended March 31, 2010, we had
restructured our debt and our liquidity position had improved, as a result, our
independent registered public accounting firm’s report on our financial
statements no longer contains a reference to our ability to continue as a going
concern. However, we cannot assure you that our liquidity position will not
deteriorate in the future and that there will not be renewed doubt about our
ability to continue as a going concern, which could materially and adversely
affect our relationships with suppliers or customers.
Risks
Associated With Doing Business in China
Adverse
changes in political and economic policies of the PRC government could impede
the overall economic growth of China, which could reduce the demand for our
products and damage our business.
We
conduct our operations and generate all of our revenues in China. Accordingly,
our business, financial condition, results of operations and prospects are
affected significantly by economic, political and legal developments in China.
The PRC economy differs from the economies of most developed countries in many
respects, including:
|
·
|
the
higher level of government involvement and
regulation;
|
|
·
|
the
early stage of development of the market-oriented sector of the
economy;
|
|
·
|
the
rapid growth rate;
|
|
·
|
the
higher rate of inflation;
|
|
·
|
the
higher level of control over foreign exchange;
and
|
|
·
|
government
control over the allocation of many
resources.
|
As the
PRC economy has been transitioning from a planned economy to a more
market-oriented economy, the PRC government has implemented various measures to
encourage economic growth and guide the allocation of resources. While these
measures may benefit the overall PRC economy, they may also have a negative
effect on us.
Although
the PRC government has in recent years implemented measures emphasizing the
utilization of market forces for economic reform, the PRC government continues
to exercise substantial control over virtually every
30
sector of
the PRC economy through the allocation of resources, controlling payment of
foreign currency-denominated obligations, setting monetary policy and imposing
policies that impact particular industries or companies in different ways. Our
ability to operate in China may be harmed by changes in PRC laws and
regulations, including those relating to how we conduct our business, taxation,
import and export tariffs, environmental regulations, land use rights, property
and other matters. We believe that our operations in China are in material
compliance with all applicable legal and regulatory requirements. However, the
central or local governments of the jurisdictions in which we operate may impose
new, stricter regulations or interpretations of existing regulations that would
require additional expenditures and efforts on our part to ensure our compliance
with such regulations or interpretations.
Accordingly,
government actions in the future, including any decision not to continue to
support recent economic reforms and to return to a more centrally planned
economy or regional or local variations in the implementation of economic
policies, could have a significant adverse effect on economic conditions in
China or particular regions thereof and could require us to divest ourselves of
any interest we then hold in PRC properties or joint ventures.
PRC
food hygiene and safety laws may become more onerous, which may adversely affect
our operations and financial performance and lead to an increase in our costs
which we may be unable to pass on to our customers.
Operators
within the PRC dairy industry and infant formula sector are subject to
compliance with PRC food hygiene and safety laws and regulations. Such laws and
regulations require all enterprises engaged in the production of dairy and
infant formula products to obtain a hygiene license. They also set out hygiene
standards with respect to food and food additives, packaging and containers, and
labeling on packaging as well as hygiene requirements for food production and
sites, facilities and equipment used for the transportation and the sale of
food. Failure to comply with PRC food hygiene and safety laws may result in
fines, suspension of operations, loss of hygiene license and, in certain cases,
criminal proceedings against an enterprise and its management. Although we are
in compliance with current PRC food hygiene and safety laws and regulations, in
the event that such laws and regulations become more stringent or widen in
scope, we may fail to comply with such laws, or if we comply, our production and
distribution costs may increase, and we may be unable to pass these additional
costs on to our customers. For example, in response to the melamine
contamination incident, the PRC State Council abolished the regulations on
inspection exemptions for food on September 18, 2008 so that our products were
to be subject to batch by batch inspection going forward. In addition, the PRC
State Council promulgated with immediate effect the Regulation on Supervision
and Administration of Quality and Safety of Dairy Products on October 9, 2008
which, among other things, imposes more stringent requirements for inspection,
production, packaging, labeling and product recall on dairy product producers.
These measures have increased our costs in the past and are likely to continue
to contribute to our costs in the future, which costs we may be unable to pass
on to our customers.
Changes
in the regulatory environment for dairy and infant nutrition products in China
could negatively impact our business.
The dairy
and infant nutrition product industries in China are regulated and regulatory
changes may affect both our customers and us. Any changes in regulations that
impose additional requirements for construction of new production lines and
facilities or expansion of existing facilities will require us to secure
additional government approvals for our current production expansion projects.
Similarly, additional safety and quality control regulations could impact our
costs of production. For example, on June 26, 2009, NDRC and MIIT jointly,
promulgated the Dairy Industry Policies (2009 Version), or the Policies. The
Policies impose new conditions that an entity must satisfy in order to engage,
or continue to engage, in the dairy products processing business. For a more
detailed description of these requirements, see “Item 1. Business—Regulation”.
Although we believe our existing entities and facilities meet the Policies
requirements, it is possible that our future expansion plans or the
establishment of new entities may fail to meet one or more of the requirements
under the Policies in the future. Failure to comply with these or any other
changes in regulations affecting our business could have a material adverse
effect on our business and our results of operations. In addition, the indirect
impact of any such changes could adversely affect our business even if the
specific regulations do not directly apply to our products or us.
31
Uncertainties
with respect to the PRC legal system could limit the legal protections available
to you and us.
We
conduct our business through our operating subsidiaries in China. Our operating
subsidiaries are generally subject to laws and regulations applicable to foreign
investments in China and, in particular, laws applicable to foreign-invested
enterprises. The PRC legal system is based on written statutes, and prior court
decisions may be cited for reference, but have limited precedential value. Over
the past several decades, a series of new PRC laws and regulations have
significantly enhanced the protections afforded to various forms of foreign
investments in China. However, since the PRC legal system continues to rapidly
evolve, the interpretations of many laws, regulations and rules are not always
uniform and enforcement of these laws, regulations and rules involve
uncertainties, which may limit legal protections available to you and us. In
addition, any litigation in China may be protracted and result in substantial
costs and diversion of resources and management attention.
If
we are found to have failed to comply with applicable laws, we may incur
additional expenditures or be subject to significant fines and
penalties.
Our
operations are subject to PRC laws and regulations applicable to us. However,
many PRC laws and regulations are uncertain in their scope, and the
implementation of such laws and regulations in different localities could have
significant differences. In certain instances, local implementation rules and/or
the actual implementation are not necessarily consistent with the regulations at
the national level. Although we strive to comply with all the applicable PRC
laws and regulations, we cannot assure you that the relevant PRC government
authorities will not later determine that we have not been in compliance with
certain laws or regulations. For example, there is a PRC regulation requiring
all employees to make contributions to a housing fund based on their salary
level and their employers to match such contributions. However, many employees
are reluctant to make such contributions as they do not perceive such
contribution will benefit them, and this regulation has generally not been
rigorously enforced. We have allowed our employees to make contributions on a
voluntary basis and then match their contributions. However, we cannot assure
you that in the future the PRC government will not start to enforce this
regulation more rigorously. Although there are no material penalties stipulated
in this regulation, if we are found not to be in compliance, we may be required
to bring current any past deficiencies, which could adversely affect our
financial condition and results of operations.
In
addition, our facilities and products are subject to many laws and regulations
administered by the PRC State Administration for Industry and Commerce, the PRC
State Administration of Taxation, the PRC Ministry of Health and Hygiene
Permitting Office, the PRC General Administration of Quality Supervision,
Inspection and Quarantine, and the PRC State Food and Drug Administration Bureau
relating to the processing, packaging, storage, distribution, advertising,
labeling, quality, and safety of food products. Our failure to comply with these
and other applicable laws and regulations in China could subject us to
administrative penalties and injunctive relief, as well as civil remedies,
including fines, injunctions and recalls of our products. It is possible that
changes to such laws, more rigorous enforcement of such laws or with respect to
our current or past practices, could have a material adverse effect on our
business, operating results and financial condition. Further, additional
environmental, health or safety issues relating to matters that are not
currently known to management may result in unanticipated liabilities and
expenditures.
Restrictions
on currency exchange may limit our ability to receive and use our sales revenue
effectively.
All of
our sales revenue and expenses are denominated in Renminbi. Under PRC law, the
Renminbi is currently convertible under the “current account,” which includes
dividends and trade and service-related foreign exchange transactions, but not
under the “capital account,” which includes foreign direct investment and loans.
Currently, our PRC operating subsidiaries may purchase foreign currencies for
settlement of current account transactions, including payments of dividends to
us, without the approval of the PRC State Administration of Foreign Exchange, or
SAFE, by complying with certain procedural requirements. However, the relevant
PRC government authorities may limit or eliminate our ability to purchase
foreign currencies in the future.
Foreign
exchange transactions by PRC operating subsidiaries under the capital account
continue to be subject to significant foreign exchange controls and require the
approval of or registration with PRC government authorities, including SAFE. In
particular, if our PRC operating subsidiaries borrow foreign currency through
loans from us or other foreign lenders, these loans must be registered with
SAFE, and if we finance the subsidiaries by means of
32
additional
capital contributions, these capital contributions must be approved by certain
government authorities, including the PRC Ministry of Commerce, or their
respective local branches. These limitations could affect our PRC operating
subsidiaries’ ability to obtain foreign exchange through debt or equity
financing.
Recent
PRC regulations relating to investment activities by, and holdings in entities
outside of China of, PRC residents and citizens, may subject our PRC resident
and citizen stockholders to personal liability and limit our ability to acquire
PRC companies or to inject capital into our PRC subsidiaries, limit our PRC
subsidiaries’ ability to distribute profits to us or otherwise materially and
adversely affect us.
In
October 2005, SAFE issued a public notice, the Notice on Relevant Issues in the
Foreign Exchange Control over Financing and Return Investment Through Special
Purpose Companies by Residents Inside China, or Notice 75. In May 2007, SAFE
issued the Notice of the State Administration of Foreign Exchange on Operating
Procedures Concerning Issues Relating to the Administration of Foreign Exchange
in Fund Raising and Return Investment Activities of Domestic Residents Conducted
via Offshore Special Purpose Companies, or Notice 106. According to Notice 75
and Notice 106, prior registration with the local SAFE branch is required for
PRC residents to establish or to control an offshore company for the purposes of
financing that offshore company with assets or equity interests in an onshore
enterprise located in the PRC. An amendment to registration or filing with the
local SAFE branch by such PRC resident is also required for the injection of
equity interests or assets of an onshore enterprise in the offshore company or
overseas funds raised by such offshore company, or any other material change
involving a change in the capital of the offshore company. Moreover, Notice 75
and Notice 106 apply retroactively. As a result, PRC residents who have
established or acquired control of offshore companies that have made onshore
investments in the PRC in the past were required to complete the relevant
registration with the local SAFE branch. If any PRC stockholder of any offshore
special purpose company fails to make the required SAFE registration and
amendment, the PRC subsidiaries of that offshore special purpose company may be
prohibited from distributing their profits and the proceeds from any reduction
in capital, share transfer or liquidation to the offshore special purpose
company. Moreover, failure to comply with the SAFE registration and amendment
requirements described above could result in liability under PRC laws for
evasion of applicable foreign exchange restrictions.
Many of
the terms and provisions in Notice 75 and Notice 106 remain unclear and
implementation by central SAFE and local SAFE branches of Notice 75 and Notice
106 have been inconsistent since their adoption. Based on the advice of our PRC
counsel, De Heng Law Offices, and after consultation with relevant SAFE
officials, our PRC resident stockholders and the PRC resident beneficial
stockholders of Meitek may be required to complete their respective SAFE
registrations pursuant to Notice 75 and Notice 106. The local SAFE branch may
not accept their applications for SAFE registration until more detailed rules or
announcements concerning the penalties for those who failed to make their SAFE
registrations are implemented. Moreover, because of uncertainty over how Notice
75 and Notice 106 will be interpreted and implemented, and how or whether SAFE
will apply it to us, we cannot predict how it will affect our business
operations or future strategies. For example, our present and prospective PRC
subsidiaries’ ability to conduct foreign exchange activities, such as the
remittance of dividends and foreign currency-denominated borrowings, may be
subject to compliance with Notice 75 and Notice 106 by our PRC resident
beneficial holders. In addition, such PRC residents may not always be able to
complete the necessary registration procedures required by Notice 75 and Notice
106. We also have little control over either our present or prospective direct
or indirect stockholders, the PRC resident beneficial stockholders of Meitek, or
the outcome of such registration procedures. A failure by our PRC resident
beneficial holders or future PRC resident stockholders to comply with the SAFE
notice, if SAFE requires it, could subject us to fines or legal sanctions,
restrict our overseas or cross-border investment activities, limit our
subsidiary’s ability to make distributions or pay dividends or affect our
ownership structure, which could adversely affect our business and
prospects.
Fluctuations
in exchange rates could adversely affect our business and the value of our
common stock.
The value
of our common stock will be indirectly affected by the foreign exchange rate
between the U.S. dollar and the Renminbi and between those currencies and other
currencies in which our sales may be denominated. Because our earnings and cash
assets are denominated in Renminbi, appreciation or depreciation in the value of
the Renminbi relative to the U.S. dollar would affect our financial results
reported in U.S. dollar terms without giving effect to any underlying change in
our business or results of operations. Fluctuations in the exchange rate will
also affect the relative value of any dividend we issue that will be exchanged
into U.S. dollars and earnings from, and the value of, any U.S.
dollar-denominated investments we make in the future.
33
Since
July 2005, the Renminbi has no longer been pegged to the U.S. dollar. Although
the People’s Bank of China, or PBOC, regularly intervenes in the foreign
exchange market to prevent significant short-term fluctuations in the exchange
rate, the Renminbi may appreciate or depreciate significantly in value against
the U.S. dollar in the medium to long-term. Moreover, it is possible that in the
future the PRC authorities may lift restrictions on fluctuations in the Renminbi
exchange rate and lessen intervention in the foreign exchange
market.
Very
limited hedging transactions are available in China to reduce our exposure to
exchange rate fluctuations. To date, we have not entered into any hedging
transactions in an effort to reduce our exposure to foreign currency exchange
risk. While we may enter into hedging transactions in the future, the
availability and effectiveness of these transactions may be limited, and we may
not be able to successfully hedge our exposure at all. In addition, our foreign
currency exchange losses may be magnified by PRC exchange control regulations
that restrict our ability to convert Renminbi into foreign
currencies.
Currently,
some of our raw materials and major equipment are imported. In the event that
the U.S. dollar appreciated against the Renminbi, our costs may increase. If we
cannot pass the resulting cost increases on to our customers, our profitability
and operating results will suffer. In addition, to the extent we enter markets
outside China in the future, we may be increasingly subject to the risk of
foreign currency fluctuations.
Restrictions
under PRC law on our PRC subsidiaries’ ability to make dividends and other
distributions could materially and adversely affect our ability to grow, make
investments or acquisitions that could benefit our business, pay dividends to
our shareholders, and otherwise fund and conduct our businesses.
Our
revenues are earned by our PRC subsidiaries. However, PRC regulations restrict
the ability of our PRC subsidiaries to make dividends and other payments to
their offshore parent company. PRC legal restrictions permit payments of
dividend by our PRC subsidiaries only out of their accumulated after-tax
profits, if any, determined in accordance with PRC accounting standards and
regulations. Each of our PRC subsidiaries is also required under PRC laws and
regulations to allocate at least 10% of our annual after-tax profits determined
in accordance with PRC GAAP to a statutory general reserve fund until the
amounts in such fund reaches 50% of our registered capital. Allocations to these
statutory reserve funds can only be used for specific purposes and are not
transferable to us in the form of loans, advances or cash dividends. As of March
31, 2010, the amount of our restricted net assets was $52.3 million. Any
limitations on the ability of our PRC subsidiaries to transfer funds to us could
materially and adversely limit our ability to grow, make investments or
acquisitions that could be beneficial to our business, pay dividends and
otherwise fund and conduct our business.
Under
China’s New Enterprise Income Tax Law (“EIT Law”), we may be classified as a
“resident enterprise” of China. This classification could result in unfavorable
tax consequences to us and our non-PRC shareholders.
The New
EIT Law provides that enterprises established outside of China whose “de facto
management bodies” are located in China are considered “resident enterprises”
and are generally subject to the uniform 25% enterprise income tax rate on their
worldwide income. In addition, a circular issued by the State Administration of
Taxation regarding the standards used to classify certain Chinese-invested
enterprises established outside of China as “resident enterprises” clarified
that dividends and other income paid by such “resident enterprises” will be
considered to be PRC source income, subject to PRC withholding tax, currently at
a rate of 10%, when recognized by non-PRC shareholders. This circular also
subjects such “resident enterprises” to various reporting requirements with the
PRC tax authorities. Under the implementation regulations to the enterprise
income tax, a “de facto management body” is defined as a body that has material
and overall management and control over the manufacturing and business
operations, personnel and human resources, finances and treasury, and
acquisition and disposition of properties and other assets of an enterprise. In
addition, the circular mentioned above details that certain Chinese-invested
enterprises will be classified as “resident enterprises” if the following are
located or reside in China: senior management personnel and departments that are
responsible for daily production, operation and management; financial and
personnel decision making bodies; key properties, accounting books, company
seal, and minutes of board meetings and shareholders’ meetings; and half or more
of the directors or senior management having voting rights. If the PRC tax
authorities determine that Synutra, Inc. or Synutra International, Inc. are
“resident enterprises,” we may be subject to enterprise income tax at the rate
of 25% on our worldwide income and dividends paid by us to our non-PRC
shareholders and, while less clear, capital gains recognized by them
with
34
respect
to the sale of our stock, may be subject to a PRC withholding tax. This will
have an impact on our effective tax rate, a material adverse effect on our net
income and results of operations, and may require us to withhold tax on our
non-PRC shareholders. We are actively monitoring the “resident enterprise”
classification rules and are evaluating appropriate organizational changes to
avoid this treatment, to the extent possible.
If we
were treated as a “resident enterprise” by PRC tax authorities, we would be
subject to tax in both the U.S. and China, and our PRC tax may not be creditable
against our U.S. tax.
The
M&A Rule establishes more complex procedures for some acquisitions of
Chinese companies by foreign investors, which could make it more difficult for
us to pursue growth through acquisitions in China.
On August
8, 2006, six PRC regulatory agencies, including the China Securities Regulatory
Commission, or CSRC, promulgated the Provisions Regarding Mergers and
Acquisitions of Domestic Enterprises by Foreign Investors, or the M&A Rule,
which became effective on September 8, 2006. The M&A Rule
establishes additional procedures and requirements that could make some
acquisitions of Chinese companies by foreign investors more time-consuming and
complex, including requirements in some instances that the PRC Ministry of
Commerce be notified in advance of any change-of-control transaction and in some
situations, require approval of the PRC Ministry of Commerce when a foreign
investor takes control of a Chinese domestic enterprise. In the future, we may
grow our business in part by acquiring complementary businesses, although we do
not have any plans to do so at this time. The M&A Rule also requires PRC
Ministry of Commerce anti-trust review of any change-of-control transactions
involving certain types of foreign acquirers. Complying with the requirements of
the M&A Rule to complete such transactions could be time-consuming, and any
required approval processes, including obtaining approval from the PRC Ministry
of Commerce, may delay or inhibit our ability to complete such transactions,
which could adversely affect our ability to expand our business or maintain our
market share.
Our
shareholders may have difficulty enforcing judgments against us.
We are a
Delaware holding company and most of our assets are located outside of the
United States. All of our current operations are conducted in the PRC. In
addition, most of our directors and officers are nationals and residents of
countries other than the United States. A substantial portion of the assets of
these persons is located outside the United States. As a result, it may be
difficult for our shareholders to effect service of process within the United
States upon these persons. It may also be difficult for our shareholders to
enforce in U.S. courts judgments on the civil liability provisions of the U.S.
federal securities laws against us and our officers and directors. In addition,
there is uncertainty as to whether the courts of the PRC would recognize or
enforce judgments of U.S. courts. DeHeng Law Offices, our counsel as to PRC law,
has advised us that the recognition and enforcement of foreign judgments are
provided for under the PRC Civil Procedures Law. Courts in China may recognize
and enforce foreign judgments in accordance with the requirements of the PRC
Civil Procedures Law based on treaties between China and the country where the
judgment is made or on reciprocity between jurisdictions. China does not have
any treaties or other arrangements that provide for the reciprocal recognition
and enforcement of foreign judgments with the United States. In addition,
according to the PRC Civil Procedures Law, courts in the PRC will not enforce a
foreign judgment against us or our directors and officers if they decide that
the judgment violates basic principles of PRC law or national sovereignty,
security or public interest. So it is uncertain whether a PRC court would
enforce a judgment rendered by a court in the United States.
The
PRC government’s recent measures to curb inflation rates could adversely affect
our future results of operations.
In recent
years, the Chinese economy has experienced periods of rapid expansion and high
rates of inflation. Rapid economic growth can lead to growth in the money supply
and rising inflation. In April 2008, the change in China’s Consumer Price Index
increased to 8.5% according to the National Bureau of Statistics of China, or
the NBS. If prices for our products rise at a rate that is insufficient to
compensate for the rise in the costs of supplies, it may have an adverse effect
on our profitability. These factors have led to the adoption by the Chinese
government, from time to time, of various corrective measures designed to
restrict the availability of credit or regulate growth and constrain inflation.
High inflation may in the future cause the Chinese government to impose controls
on credit and/or prices, or to take other actions, which could inhibit economic
activity in China, and thereby harm the market for our products.
35
Recently,
the government of China undertook various measures to alleviate the effects of
inflation, particularly on key commodities. On January 16, 2008, the PRC
National Development and Reform Commission imposed national price controls on
various products, including milk. Similarly, the government of China may
conclude that the prices of infant formula or our other products are too high
and may institute price controls that would limit our ability to set prices for
our products. The government of China has also encouraged local governments to
institute price controls on similar products. Such price controls could
adversely affect our future results of operations and the price of our common
stock.
Our
labor costs are likely to increase as a result of changes in Chinese labor
laws.
We expect
to experience an increase in our cost of labor due to changes in Chinese labor
laws which are likely to increase costs further and impose restrictions on our
relationship with our employees. In June 2007, the National People’s Congress of
the PRC enacted new labor law legislation called the Labor Contract Law and more
strictly enforced existing labor laws. The new law, which became effective on
January 1, 2008, amended and formalized workers’ rights concerning overtime
hours, pensions, layoffs, employment contracts and the role of trade unions. As
a result of the new law, we have had to reduce the number of hours of overtime
its employees can work, increase the salaries of its employees, provide
additional benefits to its employees, and revise certain of its other labor
practices. The increase in labor cost has increased our operating costs, which
we have not always been able to pass through to its customers. In addition,
under the new law, employees who have worked for us for at least 10 years or who
have had two consecutive fixed-term contracts must be given an “open-ended
employment contract” that, in effect, constitutes a lifetime, permanent
contract, which is terminable only in the event the employee materially breaches
our rules and regulations or is in serious dereliction of his or her duty. Such
non-cancelable employment contracts will substantially increase its employment
related risks and limit our ability to downsize its workforce in the event of an
economic downturn. No assurance can be given that we will not in the future be
subject to labor strikes or that we will not have to make other payments to
resolve future labor issues caused by the new laws. Furthermore, there can be no
assurance that the labor laws will not change further or that their
interpretation and implementation will vary, which may have a negative effect
upon our business and results of operations.
The
PRC government may deem the control agreements between us and certain of our
consolidated affiliated entities to be non-compliant with restrictions on
foreign investment and as a result we may be subject to penalties or required to
perform a costly restructuring of these entities.
PRC laws and regulations currently
restrict foreign entities from establishing clinical business in China. Foreign
entities that wish to establish medical clinical businesses in China must have
domestic entities as partners. In order to comply with PRC law and avoid
restrictions on foreign investment in medical clinical operations, we operate
our medical treatment services (mostly pre-natal diagnostics services) through four entities -- Nanjing Shengyuan Huiren
Clinical Examination Co., Ltd., Taiyuan Shengyuan Huiren Clinical Examination
Co., Ltd, Shijiazhuang Shengyuan Huiren Clinical Examination Co., Ltd and
Heilongjiang Shengyuan Huiren Clinical Examination Co., Ltd (the “Four
Entities”) that are not directly owned by us. We control and consolidate these
entities into our group consolidated results through a series of contractual
arrangements. See Item 1, Business—Our Corporate Structure and History and “Item
13. Certain Relationships and Related Transactions, and Director
Independence.”
The contractual arrangements we have in
place are governed by PRC law. There are substantial uncertainties
regarding the interpretation and application of current and future PRC laws and
regulations on these contractual agreements and it is possible that the PRC
authorities may in the future find these arrangements unlawful. If these
arrangements were to be deemed unlawful, the PRC authorities have broad
discretion to penalize us, including:
|
·
|
revoking the business and operating licenses of our PRC subsidiaries and affiliates; |
|
·
|
discontinuing or restricting our PRC subsidiaries’ and affiliates’ operations; |
|
·
|
imposing fines or confiscating the income of our PRC subsidiaries or affiliates; |
|
·
|
requiring us or our PRC subsidiaries and affiliates to restructure the relevant ownership structure or operations; or |
36
|
·
|
taking other regulatory or enforcement actions that could be harmful to our clinic examination business. |
Furthermore,
we have no assurance that Zhang Jibin and Jiang Yunpeng, parties to these
control agreements, will continue to cooperate with us and honor these control
agreements. There is a risk that they will not always act in our best
interests. If we cannot resolve any conflicts of interest or dispute
that may arise between them and us, we may have to take legal action to compel
them to fulfill their contractual obligations and there is substantial
uncertainty as to the outcome of any such legal proceedings. It may
be difficult for us to change our corporate structure or to bring claims against
the Four Entities if they do not perform their obligations under these
contractual arrangements.
Risks
Related to Our Common Stock
The
market price of our common stock is volatile, and its value may be depressed at
a time when our stockholders want to sell their holdings.
The
market price of our common stock is volatile, and this volatility may continue.
For instance, between July 1, 2008 and March 31, 2010, the price of our common
stock, as reported on the NASDAQ on which our common stock has traded, ranged
between $4.60 and $52.24. Though we believe this dramatic fluctuation resulted
mainly from the melamine contamination incident and the worldwide market
disruption, numerous factors, many of which are beyond our control, may cause
the market price of our common stock to fluctuate significantly.
In
addition to market and industry factors, the price and trading volume for our
common stock may be highly volatile for specific business reasons. Factors such
as variations in our revenues, earnings and cash flow, announcements of new
investments, cooperation arrangements or acquisitions, and fluctuations in
market prices for our products could cause the market price for our shares to
change substantially.
Securities
class action litigation is often instituted against companies following periods
of volatility in their stock price. This type of litigation could result in
substantial costs to us and divert our management’s attention and
resources.
The
trading market for our common stock will also be influenced by research or
reports that industry or securities analysts publish about us or our business.
If one or more analysts who cover us downgrade our common stock, the market
price for our common stock would likely decline. If one or more of these
analysts cease coverage of us or fail to regularly publish reports on us, we
could lose visibility in the financial markets, which, in turn, could cause the
market price for our common stock or trading volume to decline.
Furthermore,
securities markets may from time to time experience significant price and volume
fluctuations for reasons unrelated to operating performance of particular
companies. These market fluctuations may adversely affect the price of our
common stock and other interests in our company at a time when our stockholders
want to sell their interest in us.
Although
publicly traded, the trading market in our common stock has been substantially
less liquid than the common stock of many companies quoted on the NASDAQ Global
Select Market, and this low trading volume may adversely affect the price of our
common stock.
Although
our common stock is traded on the NASDAQ Global Select Market, the trading
volume of our common stock has generally been very low. Reported average daily
trading volume in our common stock for the three month period ended June 1, 2010
was approximately 33,800 shares. Limited trading volume will subject our shares
of common stock to greater price volatility and may make it difficult for our
stockholders to sell their shares of common stock at a price that is attractive
to them.
37
Provisions
in our charter documents and under Delaware law could discourage a
change-of-control that our stockholders may consider favorable.
Provisions
in our amended and restated certificate of incorporation and bylaws may have the
effect of delaying or preventing a change of control or changes in our
management. These provisions include the following:
|
·
|
our
board of directors is divided into three classes, with approximately
one-third of our directors elected each
year;
|
|
·
|
our
board of directors has the right to elect directors to fill a vacancy
created by the expansion of the board of directors or the resignation,
death or removal of a director, which prevents stockholders from being
able to fill vacancies on our board of
directors;
|
|
·
|
our
stockholders must provide timely notice for any stockholder proposals and
director nominations;
|
|
·
|
we
have adopted provisions that eliminate the personal liability of directors
for monetary damages for actions taken as a director, with certain
exceptions; and
|
|
·
|
our
board of directors may issue, without stockholder approval, shares of
undesignated preferred stock, which makes it possible for our board of
directors to issue preferred stock with voting or other rights or
preferences that could impede the success of any attempt to acquire
us.
|
As a
Delaware corporation, we are also subject to certain Delaware anti-takeover
provisions. Under Delaware law, a corporation may not engage in a business
combination with any holder of 15% or more of its capital stock unless the
holder has held the stock for three years or, among other things, the board of
directors has approved the transaction. Our board of directors could rely on
Delaware law to prevent or delay an acquisition of us.
ITEM
1B. UNRESOLVED STAFF COMMENTS
None.
ITEM
2. PROPERTIES
Our
headquarters are located in Beijing with around 6,400 square meters of leased
office space. Synutra Illinois leases an executive office in Rockville,
Maryland, USA. Our processing and packaging facilities are located in various
locations in China, including Beijing, Qingdao, Luobei, Zhangjiakou, Fengzhen
and Zhenglanqi. These facilities encompass approximately 77,990 square meters of
office, plant, and warehouse space. Our distribution center located in Qingdao
includes over 14,116 square meters of owned office space. All of our production
facilities are built based on the GMP standard, with equipment imported from
Europe and all of our facilities that have commenced operation have ISO9000 and
HACCP series qualifications with some also being ISO14000
certified.
We
currently own and operate four processing facilities and one packaging facility
for our powder formula products. As of March 31, 2010, we had raw milk
processing capacity of 32,600 tons per year, packaging capacity of 82,000 tons
per year and dry-blending processing capacity of 73,000 tons per
year.
The
following table sets forth certain information with respect to our production,
processing and packaging facilities.
|
Facility
|
Province/Region
|
Installed
Capacity as
of
March 31, 2010
|
Description
|
Property
Right
|
|
(tons
per year)
|
||||
|
Zhangjiakou
facility
|
Hebei
|
22,000
|
Raw
milk processing
|
Land
Use Right
|
|
Luobei
facility
|
Heilongjiang
|
3,600
|
Raw
milk processing
|
Land
Use Right
|
|
Fengzhen
facility
|
Inner
Mongolia
|
7,000
|
Raw
milk processing
|
Land
Use Right
|
38
|
Facility
|
Province/Region
|
Installed
Capacity as
of
March 31, 2010
|
Description
|
Property
Right
|
|
(tons
per year)
|
||||
|
Qingdao
facility
|
Shandong
|
73,000
|
Dry-mixing
of all of our powdered formula products
|
Land
Use Right
|
|
82,000
|
Packaging
of all of our products
|
Land
Use Right
|
||
|
Beijing
facility
|
Beijing
|
540
|
Production
of prepared baby foods
|
Land
Use Right
|
|
Zhenglanqi
facility
|
Inner
Mongolia
|
18,000
|
Production
and processing of prepared baby food and other non-core
products
|
Land
Use Right
|
|
Qingdao
Meitek facility
|
Shandong
|
2,400
|
Production
and processing of nutritional ingredients and supplement and other
non-core products
|
Land
Use Right
|
There is
no private land ownership in China. Individuals and companies are permitted to
acquire land use rights for specific purposes and for limited periods. Each
period may be renewed at the expiration of the initial and any subsequent terms.
Granted land use rights are transferable and may be used as security for
borrowings and other obligations.
We
believe that our facilities are adequate for our current operations and any
increase in production in the near term will not require additional
space.
ITEM
3. LEGAL PROCEEDINGS
As of
March 31, 2010, the end of the period covered by this report, the Company was
subject to various legal proceedings and claims discussed below, as
well as certain other legal proceedings and claims that have not been fully
resolved and that have arisen in the ordinary course of business. Other than as
discussed below, in the opinion of management, the Company does not have a
potential liability related to any current legal proceedings and claims that
would individually or in the aggregate have a material adverse effect on its
financial condition or operating results. However, the results of legal
proceedings cannot be predicted with certainty. The Company intends to contest
each lawsuit vigorously but should the Company fail to prevail in any of these
legal matters or should several of these legal matters be resolved against the
Company in the same reporting period, the operating results of a particular
reporting period could be materially and adversely affected.
On March
29, 2010, U.S. District Judge Deborah Chasanow for the District of Maryland
ordered the dismissal of a complaint filed January 15, 2009 on behalf of 54
Chinese families alleged to be affected by melamine contamination, against
Synutra International, Inc. and Synutra Inc. (Jiali Tang, et al vs. Synutra
International, Inc., et al.), alleging negligent or intentional infliction of
personal injury, negligent or intentional infliction of emotional distress,
battery, breach of warranty, fraudulent or negligent misrepresentation, seeking
compensation for punitive damages in the amount of US$500 million, together with
any compensatory damages. In an opinion issued the same date of the order above,
the court sided with the Company’s positions and granted the motion to dismiss
on the grounds of forum
non conveniens. The court also granted the motion to file under seal a
response to a Notice of Recent Development filed by the Plaintiffs. In
considering the motion to dismiss on the grounds of forum
non conveniens, the court examined both the availability and adequacy of
the alternative forum in China as well as how public and private interests favor
the choice of forum. In addition, taking into account that an “alternative
compensation plan is undisputedly available to Plaintiffs,” the court ruled that
“a conditional dismissal will not be employed to protect the Plaintiffs’ rights
to pursue a judicial remedy in the alternative forum.” On April 27, 2010, the
plaintiffs filed a notice of appeal of the dismissal
order. Plaintiffs’ opening brief is due June 28,
2010.
Management
continues to evaluate the lawsuits discussed above and based on the stage of
these proceedings, management is unable to reasonably estimate the likelihood of
any loss or the amount or range of any potential loss that could result from the
litigation. Therefore, no accrual has been established for any potential loss in
connection with these lawsuits.
39
On March
2, 2009, a lawsuit was filed in China on behalf of 54 Chinese families alleged
to be affected by melamine contamination against our subsidiary, Shengyuan
Nutritional Food Co., Ltd. at Qingdao Intermediate People’s Court, seeking
compensation for damages, including medical and other expenses, emotional harm
and punitive damages, for an aggregate amount over US$1.0 million (RMB6.9
million). Further, on April 14, 2009, a lawsuit was filed on behalf of 73
families (including the original 54 families) against our subsidiary, Shengyuan
Nutritional Food Co., Ltd. at Shandong Higher People’s Court on April 14, 2009,
seeking compensation for damages of over US$1.7 million (RMB11.3 million). The
PRC Civil Procedure Law provides that, generally, a court shall make a decision
as to whether it will accept a case within seven days after a lawsuit is
filed. To date, we have not received any notice from such PRC courts
that a lawsuit has been accepted against us in connection with the melamine
contamination incident. Therefore, no accrual has been established for any
potential loss in connection with these lawsuits. It is also possible that these
plaintiffs may bring other similar suits against us in the same court or in a
new court.
ITEM
4. [REMOVED AND RESERVED]
PART
II
ITEM
5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDERS
MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Price
Range of our Common Stock
Our
common stock has been trading on the NASDAQ Global Select Market under the
symbol “SYUT” since November 8, 2007. Our common stock was previously quoted on
the Over-The-Counter Bulletin Board, or OTCBB, under the trading symbol
“SYUT.OB” until April 11, 2007. On April 12, 2007, our common stock was listed
on the NASDAQ Global Market and subsequently approved for listing on the NASDAQ
Global Select Market on November 8, 2007.
The high
and low bid prices reflect inter-dealer prices, without retail mark-up,
mark-down or commission, and may not represent actual transactions.
|
The
NASDAQ Global/Global Select Market Price per Share
|
||||||||
|
High
|
Low
|
|||||||
|
Fiscal
Year Ending March 31, 2011
|
||||||||
|
First
Quarter (through June 1, 2010)
|
$ | 24.99 | $ | 18.59 | ||||
|
Fiscal
Year Ended March 31, 2010
|
||||||||
|
Fourth
Quarter
|
$ | 23.47 | $ | 12.8 | ||||
|
Third
Quarter
|
14.50 | 10.79 | ||||||
|
Second
Quarter
|
16.60 | 9.92 | ||||||
|
First
Quarter
|
13.37 | 6.87 | ||||||
|
Fiscal
Year Ended March 31, 2009
|
||||||||
|
Fourth
Quarter
|
$ | 11.97 | $ | 4.33 | ||||
|
Third
Quarter
|
23.58 | 7.30 | ||||||
|
Second
Quarter
|
52.24 | 11.83 | ||||||
|
First
Quarter
|
34.00 | 29.30 | ||||||
As of
June 1, 2010, we had 36 registered stockholders of our common stock on record.
This number does not include shares held by brokerage clearing houses,
depositories or otherwise in unregistered form or shares held by a custodian for
the benefit of our employees.
40
Dividend
Policy
We have
never declared or paid any dividends on shares of our common stock. We intend to
retain any future earnings to fund the development and growth of our business,
and we do not anticipate paying any dividends in the foreseeable
future.
The RBS
Loan restricts our ability to pay any dividends that, when taken in the
aggregate with other payments by us within a 12-month period, exceed 30.0% of
our consolidated net income for such period. For a description of the RBS Loan,
see Item 7. Management’s Discussion and Analysis of Financial Condition and
Results of Operations—Liquidity and Capital Resources—Outstanding Indebtedness.
We may enter into agreements in the future that restrict our ability to make
distributions to stockholders.
Our
revenues are earned by our PRC subsidiaries. However, PRC regulations restrict
the ability of our PRC subsidiaries to make dividends and other payments to
their offshore parent company. PRC legal restrictions permit payments of
dividend by our PRC subsidiaries only out of their accumulated after-tax
profits, if any, determined in accordance with PRC accounting standards and
regulations. Each of our PRC subsidiaries is also required under PRC laws and
regulations to allocate at least 10% of our annual after-tax profits determined
in accordance with PRC GAAP to a statutory general reserve fund until the
amounts in such fund reaches 50% of our registered capital. Allocations to these
statutory reserve funds can only be used for specific purposes and are not
transferable to us in the form of loans, advances or cash dividends. In
addition, there are restrictions on the distribution of share capital from the
Company’s PRC subsidiaries. As of March 31, 2010, the amount of our restricted
net assets was $52.3 million.
Recent
Sales of Unregistered Securities
None.
ITEM
6. SELECTED FINANCIAL DATA
The
following selected consolidated financial data for the fiscal years ended March
31, 2010, 2009 and 2008 are derived from our audited consolidated financial
statements included elsewhere in this Form 10-K. The financial data for the
years ended March 31, 2007 and 2006 are derived from audited consolidated
financial statements which are not included in this Form 10-K. The results of
operations for past accounting periods are not necessarily indicative of the
results to be expected for any future accounting period.
The
financial data set forth below should be read in conjunction with, and are
qualified in their entirety by reference to “Item 7. Management’s Discussion and
Analysis of Financial Condition and Results of Operations,” and our consolidated
financial statements and related notes included elsewhere in this Form
10-K.
|
Fiscal
Year Ended March 31,
|
||||||||||||||||||||
|
2010
|
2009
|
2008
|
2007
|
2006
|
||||||||||||||||
|
(in
thousands except earnings per share data)
|
||||||||||||||||||||
|
Selected
Consolidated Statement of Income Data:
|
||||||||||||||||||||
|
Net
sales
|
$ | 291,886 | $ | 312,528 | $ | 362,090 | $ | 216,605 | $ | 132,289 | ||||||||||
|
Cost
of sales
|
208,476 | 259,086 | 175,568 | 109,900 | 76,653 | |||||||||||||||
|
Gross
profit
|
83,410 | 53,442 | 186,522 | 106,705 | 55,636 | |||||||||||||||
|
Selling
and distribution expenses
|
43,989 | 44,178 | 34,449 | 25,561 | 15,494 | |||||||||||||||
|
Advertising
and promotion expenses
|
33,854 | 115,478 | 76,388 | 52,322 | 20,908 | |||||||||||||||
|
General
and administrative expenses
|
24,509 | 25,455 | 16,013 | 7,031 | 5,896 | |||||||||||||||
|
Impairment
loss
|
5,894 | — | — | — | — | |||||||||||||||
|
Other
operating income, net
|
894 | 5,790 | 1,492 | 1,109 | 734 | |||||||||||||||
|
Income
(loss) from operations
|
(23,942 | ) | (125,879 | ) | 61,164 | 22,900 | 14,072 | |||||||||||||
|
Interest
expense
|
8,603 | 4,857 | 6,354 | 1,896 | 1,784 | |||||||||||||||
|
Interest
income
|
1,850 | 341 | 1,801 | 356 | 238 | |||||||||||||||
41
|
Fiscal
Year Ended March 31,
|
||||||||||||||||||||
|
2010
|
2009
|
2008
|
2007
|
2006
|
||||||||||||||||
|
(in
thousands except earnings per share data)
|
||||||||||||||||||||
|
Other
income (expense), net
|
(1,081 | ) | (580 | ) | (3,084 | ) | 110 | (44 | ) | |||||||||||
|
Income
(loss) before income tax expense (benefit)
|
(31,776 | ) | (130,975 | ) | 53,527 | 21,470 | 12,482 | |||||||||||||
|
Income
tax expense (benefit)
|
(6,904 | ) | (30,386 | ) | 7,855 | 1,596 | 1,446 | |||||||||||||
|
Net
income (loss)
|
(24,872 | ) | (100,589 | ) | 45,672 | 19,874 | 11,036 | |||||||||||||
|
Net
income (loss) attributable to the noncontrolling interest
|
(257 | ) | (40 | ) | 11 | — | 1 | |||||||||||||
|
Net
income (loss) attributable to Synutra International, Inc. common
shareholders
|
$ | (24,615 | ) | $ | (100,549 | ) | $ | 45,661 | $ | 19,874 | $ | 11,035 | ||||||||
|
Earnings
per share-basic
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.86 | $ | 0.40 | $ | 0.23 | ||||||||
|
Earnings
per share-diluted
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.85 | $ | 0.40 | $ | 0.23 | ||||||||
|
March
31,
|
||||||||||||||||||||
|
2010
|
2009
|
2008
|
2007
|
2006
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Selected
Balance Sheets Data:
|
||||||||||||||||||||
|
Cash
and cash equivalents
|
$ | 48,693 | $ | 37,736 | $ | 97,425 | $ | 20,836 | $ | 5,677 | ||||||||||
|
Working
capital (deficit)
|
(27,593 | ) | (80,432 | ) | 111,230 | (8,281 | ) | (14,270 | ) | |||||||||||
|
Inventory
|
52,134 | 114,724 | 61,853 | 16,406 | 11,789 | |||||||||||||||
|
Total
assets
|
349,357 | 472,571 | 294,318 | 127,271 | 83,009 | |||||||||||||||
|
Short-term
debt
|
98,069 | 224,647 | 21,228 | 53,104 | 36,680 | |||||||||||||||
|
Long-term
debt due within one year
|
61,194 | — | 1,923 | — | — | |||||||||||||||
|
Long-term
debt
|
41,018 | 8,777 | 34,184 | — | — | |||||||||||||||
|
Capital
lease obligation
|
5,372 | 5,254 | — | — | — | |||||||||||||||
|
Total
long-term liabilities
|
52,497 | 20,468 | 39,993 | 4,138 | — | |||||||||||||||
|
Total
equity
|
$ | 52,931 | $ | 76,859 | $ | 171,259 | $ | 42,701 | $ | 20,951 | ||||||||||
ITEM
7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND
RESULTS OF OPERATIONS
The
following discussion and analysis of our financial condition and results of
operations should be read in conjunction with our consolidated financial
statements and related notes that appear elsewhere in this Form 10-K. In
addition to historical consolidated financial information, the following
discussion contains forward-looking statements that reflect our plans, estimates
and beliefs. Our actual results could differ materially from those discussed in
the forward-looking statements. Factors that could cause or contribute to these
differences include those discussed below and elsewhere in this Form 10-K,
particularly in “Item 1A. Risk Factors.”
Overview
We are a
leading infant formula company in China. We principally produce, market and sell
our products under the “Shengyuan,” or “Synutra,” name, together with other
complementary brands. We focus on selling premium infant formula products, which
are supplemented by more affordable infant formulas targeting the mass market as
well as other nutritional products and ingredients. We sell our products through
an extensive nationwide sales and distribution network covering 30 provinces and
provincial-level municipalities in China. As of March 31, 2010, this network
comprised over 540 independent distributors and over 1,000 independent
sub-distributors who sell our products in over 71,000 retail
outlets.
We
currently have three reportable segments which are:
42
|
o
|
Powdered
formula segment: Powdered formula segment covers the sale of powdered
infant and adult formula products. It includes the brands of Super,
U-Smart, Mingshan which was launched in October 2008 and Helanruniu which
was launched in December 2008;
|
|
o
|
Baby
food segment: Baby food segment covers the sale of prepared baby food for
babies and children. It includes the brand of Huiliduo which was launched
in March 2009;
|
|
o
|
Nutritional
ingredients and supplements segment: Nutritional ingredients and
supplements segment covers the production and sale of nutritional
ingredients and supplements such as chondroitin sulfate, and
microencapsulated Docosahexanoic Acid (“DHA”) and Arachidonic Acid (“ARA”)
which we launched in the fiscal quarter ended December 31,
2008.
|
Our
“Other” business includes non-core businesses such as toll packaging, toll
drying service and sales of ingredients and materials to industrial customers. A
major portion of our “Other” business for the fiscal year ended March 31, 2010
was sales of surplus industrial milk powder which generated immediate cash flow
for us. We do not anticipate sales of our other business to be as significant as
going forward.
On
September 16, 2008, we announced a compulsory recall on certain lots of U-Smart
products and a voluntary recall of other products that were contaminated or
suspected to be contaminated by melamine, a substance not approved for use in
food and linked to illnesses among infants and children in China. The cost of
this action during the year ended March 31, 2009 was $101.5 million, including
the cost of product replacement of $48.1 million in cost of sales, the
write-down and write-off of affected inventory of $48.5 million in cost of
sales, the net amount of $2.3 million to a compensation fund set up by China
Dairy Industry Association to settle existing and potential claims arising in
China from families of infants affected by melamine contamination in general and
administrative expenses, and freight charges of $2.6 million in selling and
distribution expenses, of which $4.5 million was recorded as a product recall
provision in the consolidated balance sheet as of March 31, 2009. During the
fiscal year ended March 31, 2010, we reversed recall expense of $0.9 million,
mostly being overestimated product replacement cost. We believes that the
product recall has been substantially completed and estimates that there should
be no further material product recall costs incurred. We believe that our
response to the melamine crisis, as well as recall efforts, including
instituting a voluntary recall were well recognized by the public and helped us
to maintain our reputation, brand recognition and relationships with our
distributors and suppliers.
Our net
sales for the fiscal year ended March 31, 2010 decreased by 6.6% to $291.9
million from $312.5 million for the prior fiscal year. Our gross profit for the
fiscal year ended March 31, 2010 increased by 56.1% to $83.4 million from $53.4
million for the prior fiscal year. Our net loss attributable to Synutra
International, Inc. common shareholders for the fiscal year ended March 31, 2010
was $24.6 million, as compared to $100.5 million for the prior fiscal
year.
The
decrease in our net sales reflected in part the lingering impact of the product
recall carried out in late calendar year 2008, as well as a greater proportion
of rebates to distributors and the additional product discounts we provided to
distributors beginning August 2009 to compensate distributors as they were
carrying out certain product promotion activities that were previously handled
by us. However, there have been strong signs of recovery in
end-market sales. According to data released by the Ministry of Commerce’s
Commercial Information Center (CIC), the Company’s market share for the fiscal
quarter ended March 31, 2010 was 7.0%, representing a significant increase from
the 3.4% reported in October 2008, the month immediately following the melamine
contamination incident. The Company’s powdered formula segment recorded a gross
profit of $96.2 million, or 49.5% from net sales of $194.4 million for the
fiscal year ended March 31, 2010, representing a significant improvement from
fiscal year when the product recall happened. The sales of Super series infant
formula accounted for about 57.2% of the volume of sales and 69.4% of the net
sales for the segment for the fiscal year ended March 31, 2010. The Company
believes that the improvement in sales of its Super series infant formula has
led the segment in market recovery and helped to stabilize the Company’s market
position.
Though
powdered formula sales have improved since the quarter ended December 31, 2008
that immediately followed the recall, the Company has not yet recovered its
pre-recall market share of approximately 10.5% in August 2008, according to
CIC. In order to regain its market leadership, the Company has
focused on fundamental approaches to procure new customers through its
integrated marketing platform, capitalizing on database resources
43
built
with a nationwide deployed team of more than 300 nutrition education specialists
who work directly with medical and healthcare professionals at maternity wards
or clinics throughout the country. The Company also made sales and
distribution system adjustments including a new control and monitoring mechanism
aimed at improving sales efficiency and managing the effectiveness of our
distribution channels which comprise of more than 540 independent distributors
across the market. The Company believes that these initiatives are generating
favorable results as reflected in the improved market share data for the fiscal
quarter ended March 31, 2010 compared to the fiscal quarter ended December 31,
2008 that immediately followed the recall.
We offer
rebates to distributors and supermarket retailers to sustain and increase our
market share. Pursuant to these rebate programs, distributors and supermarket
retailers receive a rebate after attaining certain performance parameters
relating to product purchases, formulary status and/or pre-established market
share milestones relative to competitors. We record rebates as a reduction of
revenues in the year in which these programs are offered. After the melamine
contamination incident of 2008, we increased the rate of these rebates. Such
measures were essential after the melamine contamination incident to stabilize
our relationship with distributors. More recently, as the infant formula market
in China gradually returned to normal, we have scaled back the rate of these
rebates.
After the
melamine contamination incident, the Company began to use imported milk powder
in the powdered formula products and decreased the consumption of domestically
produced milk powder. In light of the decrease in demand for domestically
produced milk powder as well as increasing competition from other domestic raw
milk suppliers, the Company decided to sell certain production facilities and
dairy farms. On September 9, 2009, two subsidiaries of Synutra International,
Inc., Baoquanling and Cow Breeding, entered into an asset purchase agreement
(the “Agreement”) with Wondersun, to sell three dairy farms and two milk
processing factories in China for approximately $28.9 million plus reimbursement
for certain qualified construction in progress assets acquired subsequent to the
valuation date, of which $4.4 million will be received in the fiscal year ending
March 31, 2012. The assets were classified as held for sale assets
and were written down to the fair value less estimated cost to sell. No
depreciation or amortization was recorded after September 9, 2009.
The
estimated impairment loss on the assets group for the fiscal year ended March
31, 2010 was $5.9 million. As of March 31, 2010, the Company has transferred a
majority of these assets to Wondersun. The Company believes the sale of these
facilities will streamline the Company’s operations and benefit the Company’s
long-term operating efficiency.
In the
fiscal year ended March 31, 2010, we imported all the milk powder used for the
production of our Super, U-Smart and Helanruniu series of products. In the
meantime, our domestic production facilities continue to purchase raw milk
locally to produce milk powder for commercial resale for our other business and
for our Mingshan series of products. We intend to continue relying on imported
milk powder for use in our Super, U-Smart and Helanruniu series of products in
the foreseeable future. Due to the subsequent decline in the consumption of
dairy based products in the PRC as a result of the melamine contamination
incident and the significant increase in milk powder imports, there has been a
nationwide inventory build-up of domestically produced milk powder in the PRC.
Such inventory build-up has caused a decline in milk powder prices and has
resulted in us taking a write-down of our inventory of industrial milk powder of
$6.7 million for the fiscal year ended March 31, 2010. Currently milk powder,
whether imported or domestically produced, accounts for approximately 37% of our
raw materials inventory. We managed to reduce our overall inventory level by
selling down significant portion of domestically produced raw milk powder at a
loss for the fiscal year ended March 31, 2010, and resulting in a corresponding
increase in our cost of goods sold of our “Other” business. We also sold some
surplus imported milk powder. Sales of this surplus milk powder generated
immediate cash flow, which was used in our operations. The inventory has
decreased to $52.1 million as of March 31, 2010 from $114.7 million as of March
31, 2009, which management believes is optimal for our normal operating needs
and close to the pre-recall inventory level.
The
Company’s main operations are located in mainland China. Nevertheless, the
current global economic disruption in the overall economy and financial markets
could lead to reduced consumer confidence in the economy and consumer spending,
which could negatively affect our financial position and results of operations.
See Part I - Item 1A. Risk Factors—Risks Related to Our Business—The disruptions in the overall
economy and the financial markets that started in late calendar year
2007 may adversely impact our business and results of operations and
may limit our access to additional financing.
44
Unless
otherwise noted, all translations from Renminbi to U.S. dollars were made at the
mid rate published by the People’s Bank of China, or the mid rate, as of March
31, 2010, which was RMB6.8263 to $1.00. We make no representation that the
Renminbi amounts referred to in this Annual Report on Form 10-K could have been
or could be converted into U.S. dollars at any particular rate or at all. On
June 1, 2010, the mid rate was RMB 6.8279 to $1.00.
Factors
Affecting Our Results of Operations
Our
operating results are primarily affected by the following factors:
Perceptions
of Product Quality and Safety
Rising
consumer wealth in China has contributed to a greater acceptance by consumers in
China of and desire for higher-priced products with perceived quality advantages
associated with such products. Thus, we believe that infant formula producers
with a reputation for quality and safety should be able to command higher
average selling prices and thereby generate higher gross margins than
competitors that do not possess the same perceived reputation for quality and
safety. Conversely, any decrease in consumer perceptions of quality and safety
could adversely impact such producers’ sales and gross margins. Moreover, a
decrease in the quality and safety of any particular product could trigger wider
negative perception of the decrease in the quality and safety of all producers,
thereby affecting the industry generally. For example, the melamine
contamination incident had resulted in a significant reduction in the sales of a
number of major dairy product companies in China, including us. If a future
market crisis involving any of our products should occur, especially if
management failed to respond to such crisis in a timely and effective manner,
our brand recognition and reputation could be severely damaged, which could
adversely affect our results of operations. See Part I - Item 1A. Risk
Factors—Risks Related to Our Business—We are highly dependent upon
consumers’ perception of the safety and quality of our products. Any ill
effects, product liability claims, recalls, adverse publicity or negative public
perception regarding particular ingredients or products or our industry in
general could harm our reputation and adversely affect our results of
operations.
Brand
Recognition and Customer Loyalty
In recent
years, there has been growing demand in China for premium infant formula
products due to increasing consumer awareness of brand image and nutritional
value of the products offered by leading producers. Although the market is still
highly competitive, we believe that companies with strong national brands and
customer loyalty will increasingly capture market share from regional brands
with less brand recognition. Moreover, we believe brand recognition and customer
loyalty are predominantly influenced by customer perceptions of the quality and
safety of branded products. We believe the melamine contamination incident
involving 22 infant formula producers has increased the importance of consumer
perception of quality and safety and the need to maintain and increase brand
recognition and customer loyalty.
Competition
and Market Position
While
China’s infant formula market is expected to grow significantly, competition is
intense. The market has become highly fragmented in recent years as an
increasing number of infant formula producers have entered the market. We face
significant competition from domestic and multinational producers. A small
number of multinational players enjoy significant market share in China,
particularly in the more affluent major urban areas, based on greater brand name
recognition among Chinese consumers. In addition, competition from domestic
producers has become more intense in recent years, especially from large
national milk companies, such as Yili, Yashili, Beingmate and Feihe, which have
entered the infant formula market.
We focus
on developing and marketing premium products for the infant formula market in
China. By leveraging our focused marketing strategy, our brand name and our
sales and marketing infrastructure, we have been able to sell infant formula
products to consumers in China’s small to mid-size cities and rural areas and
are perceived to deliver premium quality that justifies our premium prices. This
strategy has allowed us to maintain and improve our market share in our primary
markets.
45
Product
Offering and Pricing
Infant
formula has been, and is expected to remain, our primary product. Due to rising
economic affluence in China, infant formula products have become more
affordable, resulting in the rapid growth of the overall market for infant
formula in China. Despite the recent rapid growth, we believe much of the market
is still underserved with respect to infant formula. We believe this growth in
demand will help drive sales for many PRC infant formula producers, but
companies with strong brand loyalty and extensive distribution networks in China
will have greater ability to capitalize on such growth as well as to increase
prices and pass on higher raw material costs to customers. This can be
accomplished through launching higher-priced new infant formula product lines
(such as our Super infant formula products) or re-launching older product lines
with higher prices and improved product features (such as our U-Smart infant
formula products).
Raw
Material Supply and Prices
The per
unit costs of producing our infant formula are subject to the supply and price
volatility of raw milk and other raw materials, which are affected by the PRC
and global markets. For example, raw milk prices are affected by factors such as
geographic location, fluctuations in production and competition. Historically,
we have been able to meet our raw milk supply needs by building our processing
facilities close to our milk suppliers and by maintaining long-term business
relationships with milk collection stations. In the fiscal year ended March 31,
2010, all of the milk powder used for our Super, U-Smart and Helanruniu or
Holsteina series products is imported. This has led to a significant reduction
in our raw milk procurement.
Although
we have not used as much raw milk in the aftermath of the melamine contamination
incident, increases in the price of raw milk, milk powder and whey protein
powder would negatively impact our gross margins if we are not able to offset
such price increases through increases in our selling price or change in product
mix. See Part I - Item 1A. Risk Factors—Risks Related to Our Business—We might face inventory write-down
if milk powder inventory continues to increase and milk powder prices continue
to decline. We maintain
inventories of raw materials and finished products, and our inventories may
spoil.
Advertising
and Sales Promotion Costs
We have
historically relied on our extensive distribution network, our consumer
education programs and customer relation services to market and sell our
products. Following aggressive advertising and promotional campaigns in the
fiscal year ended March 31, 2009, we reduced the expenditures on advertising and
promotion, and have refocused our efforts to achieve effective market
pull-through with new customers by supporting increased activities on the
consumer-end and beyond the distribution channels by our field promoters in the
communities and our nutrition education professionals at the medical and
healthcare facilities. We expect to increase our advertising and promotion
expenditures going forward.
Taxation
We file
separate tax returns in the United States and China. Income taxes of our
subsidiaries are calculated in accordance with taxation principles currently
effective in the PRC. For Synutra Illinois and Synutra Delaware, applicable U.S.
tax laws are followed.
On March
16, 2007, the National People’s Congress of the PRC approved and promulgated a
new tax law, which took effect beginning January 1, 2008. The Company’s PRC
subsidiaries then measure and pay enterprise income tax pursuant to the new tax
law. Under the new tax law, foreign investment enterprise and domestic companies
are subject to a uniform income tax rate of 25%. The new tax law provides a
five-year transition period from its effective date for those enterprises which
were established before the promulgation date of the new tax law and which were
entitled to a preferential lower tax rate under the then effective tax laws or
regulations.
We
operate under tax holidays in PRC, which are effective through December 2012.
The impact of these tax holidays increased PRC taxes by $1.4 million for fiscal
years ended March 31, 2010, and decreased PRC taxes by $3.2 million and $14.0
million for fiscal years ended March 31, 2009 and 2008, respectively. The
benefit of the tax
46
holidays
on earnings per share was $(0.03), $0.06 and $0.26 for fiscal years ended March
31, 2010, 2009 and 2008, respectively.
Some of
the Company’s PRC subsidiaries are eligible under the transition rules to
continue enjoying tax holidays or reduced tax rate until expiration. The
following table illustrates the applicable tax rate and tax holidays of major
PRC subsidiaries under the new EIT Law:
|
Name
of Subsidiaries
|
Statutory
Tax Rate Beginning
January
1, 2008
|
Tax
Holiday (based on calendar year)
|
||
|
Shengyuan
Nutritional Food Co., Ltd.
|
25%
|
2
years tax free (2004, 2005); 12% (2006, 2007); 12.5%
(2008)
|
||
|
Heilongjiang
Mingshan Dairy Co., Ltd.
|
25%
|
2
years tax free (2006, 2007); 3 years tax at 12.5%
(2008-10)
|
||
|
Zhangjiakou
Shengyuan Dairy Co.,
Ltd.
|
25%
|
2
years tax free (2006, 2007); 3 years tax at 12.5%
(2008-10)
|
||
|
Inner
Mongolia Shengyuan Food Co., Ltd.
|
25%
|
2
years tax free (2008, 2009); 3 years tax at 12.5%
(2010-12)
|
||
|
Inner
Mongolia Mengyuan Food Co., Ltd.
|
25%
|
No
tax holiday
|
||
|
Meitek
Technology (Qingdao) Co., Ltd.
|
25%
|
2
years tax free (2008, 2009); 3 years tax at 12.5%
(2010-12)
|
||
|
Heilongjiang
Baoquanling Shengyuan Dairy Co., Ltd.
|
25%
|
No
tax holiday
|
||
|
Beijing
Shengyuan Huiliduo Food Technology Co., Ltd.
|
25%
|
No
tax holiday
|
||
|
Global
Food Trading (Shanghai) Co., Ltd.
|
25%
|
20%
(2009), 22% (2010), 24% (2011), 25%
(2012)
|
Our
income will be derived from dividends we receive from our PRC operating
subsidiaries described above. The New EIT Law and its implementing rules
generally provide that a 10% withholding tax applies to China-sourced income
derived by non-resident enterprises for PRC enterprise income tax purposes. We
expect that such 10% withholding tax will apply to dividends paid to us by our
PRC subsidiaries but this treatment will depend on our status as a non-resident
enterprise. For detailed discussion of PRC tax issues related to resident
enterprise status, see Part-I - Item 1A. Risk Factors—Risks Associated with
Doing Business in China—Under
China’s New Enterprise Income Tax Law (“EIT Law”), we may be classified as a
“resident enterprise” of China. This classification could result in unfavorable
tax consequences to us and our non-PRC shareholders.
Each of
our PRC subsidiaries files stand-alone tax returns and we do not file a
consolidated tax return.
Critical
Accounting Policies and Estimates
We
prepare our financial statements in accordance with US GAAP. The preparation of
these financial statements requires the use of estimates and assumptions that
affect the reported amounts of assets and liabilities and the disclosure of
contingent assets and liabilities at the date of the financial statements and
the reported amount of revenues and expenses during the reporting period.
Management periodically evaluates the estimates and judgments made. Management
bases its estimates and judgments on historical experience and on various
factors that are believed to be reasonable under the circumstances. Actual
results may differ materially from these estimates as a result of different
assumptions or conditions.
The
following critical accounting policies involve more significant judgments and
estimates used in the preparation of our financial statements.
Revenue
We
recognize revenue when title and risk and rewards for the products are
transferred to the customer, price is fixed and determinable and collectability
is reasonably assured. At the time of the sale, we also record estimates for a
variety of sales deductions, including value added taxes, rebates, discounts and
incentives, trade promotions and
47
product
returns. Sales deductions are reported as a reduction of revenue. Most of our
nutritional product sales are made through distributors. Under the distributor
arrangement, evidenced by purchase order together with advance payment, sales
revenue is realized and earned upon acceptance of delivery of products by the
distributors. We apply this revenue recognition policy uniformly to all
nutritional products, including all dairy-based pediatric and adult nutritional
products.
A small
fraction of our nutritional product sales are through supermarket retailers
directly. Our revenue arrangement with some of these retailers includes a right
of return clause. Our price to the supermarkets is fixed. The supermarkets’
obligation to us would not be changed in the event of theft or physical
destruction or damage of the product. We recognize revenue when the supermarkets
have paid us, or the supermarkets are obligated to pay us and the obligation is
not contingent on resale of the product. The amount of future returns are
estimated and recognized in the current period.
Our gross
sales are subject to various deductions, primarily comprised of rebates and
discounts to distributors and retailers. These deductions represent estimates of
the related obligations, requiring the use of judgment when estimating the
impact of these sales deductions on gross sales for a reporting period. We
report these adjustments as a reduction of gross sales to arrive at net
sales.
|
|
·
|
We
offer rebates to distributors and supermarket retailers to sustain and
increase our product market share. These rebate programs provide that
distributors and supermarket retailers receive a rebate after attaining
certain performance parameters relating to product purchases, formulary
status and/or pre-established market share milestones relative to
competitors. Since rebates are contractually agreed upon, we estimate
rebates based on the specific terms in each agreement, historical
experience, anticipated reimbursement channel mix and product growth
rates. We consider the sales performance of products subject to rebates
and other contract discounts and adjust the provision periodically to
reflect actual experience. Actual amounts may differ if the actual
performance vary from estimates. The Company records rebates as a
reduction of revenues in the year in which these programs are
offered.
|
|
|
·
|
We
offer product discounts to compensate distributors for the promotional
activities which were previously performed by us. Prior to August 2009,
promotional activities were generally jointly performed by distributors
and us, and each party was responsible for the pre-determined portion of
expense respectively. Effective August 2009, we established a monthly
budget for promotional expenses. Distributors are responsible for
organizing the promotional activities, and providing the documentation of
their expenses, which would be recorded as product discounts to offset
receivable from distributors in the applicable
month.
|
|
|
·
|
We
record a provision for estimated sales returns due to package damage and
termination of distributorships. Due to the melamine contamination
incident, the turnover rate of products in the distribution channel to end
customers was significantly increased. To avoid the potential loss the
distributors might suffer, we offered certain product exchange for those
products approaching the expiration dates. To respond to the exchange
program, since October 2009, we recorded estimated replacement costs for
future potential product exchange program. The sales return amount
represents management’s best estimates based on the available information
at the time of estimate.
|
|
|
·
|
For
product sales and promotions at supermarkets and shopping malls, certain
expenses in relation to shelf display, end-cap placement, bar-coding,
banner advertising, etc. are paid to supermarkets and shopping mall
operators. These expenses are deducted from revenues in accordance with
ASC 605, Revenue Recognition (previously EITF 01-9 “Accounting for Consideration
Given by a Vendor to a Customer (Including a Reseller of the Vendor’s
Product).”)
|
Allowance
for Doubtful Accounts
We
maintain allowances for doubtful accounts primarily based on the age of
receivables and factors surrounding the credit risk of specific customers. An
estimate for doubtful accounts is made when collection of the full amount is no
longer probable. We perform risk assessment for each customer, and provide
specific allowance
48
for those
deemed to have high risk of uncollectibility. We also record provision for other
customers without specific risks by review the aging of the receivables. Bad
debts are written off as incurred.
Inventories
Our
inventories are stated at the lower of cost or market. The valuation of
inventory requires us to estimate excess and slow moving inventory. The
determination of the value of excess and slow moving inventory is based upon
assumptions of future demands and market conditions. If actual market conditions
are less favorable than those projected by management, inventory write-downs may
be required. We routinely evaluate quantities and value of our inventories in
light of current market conditions and market trends, and record write-down
against the cost of inventories for a decline in market. Inventory write-down
charges establish a new cost basis for inventory. In estimating obsolescence, we
utilize our backlog information and project future demand. Market conditions are
subject to change and actual consumption of inventories could differ materially
from forecasted demand. Furthermore, the price of raw milk and milk powder are
subject to fluctuations based on global supply and demand. If actual market
conditions are less favorable or other factors arise that are significantly
different from those anticipated by management, additional inventory write-downs
or increases in obsolescence reserves may be required.
Income
Taxes
The
provision for income taxes has been determined using the asset and liability
approach of accounting for income taxes. Under this approach, deferred taxes
represent the future tax consequences expected to occur when the reported
amounts of assets and liabilities are recovered or paid. The provision for
income taxes represents income taxes paid or payable for the current year plus
the change in deferred taxes during the year. Deferred taxes result from
differences between the financial and tax bases of our assets and liabilities
and are adjusted for changes in tax rates and tax laws when changes are enacted.
Valuation allowances are recorded to reduce deferred tax assets when it is more
likely than not that a tax benefit will not be realized. The assessment of
whether or not a valuation allowance is required often requires significant
judgment including the long-range forecast of future taxable income and the
evaluation of tax planning initiatives. Adjustments to the deferred tax
valuation allowances are made to earnings in the period when such assessments
are made.
Our tax
rate is based on expected income, statutory tax rates and tax planning
opportunities available in the various jurisdictions in which we operate. For
interim financial reporting, we estimate the annual tax rate based on projected
taxable income for the full year and record a quarterly income tax provision in
accordance with the anticipated annual rate. As the year progresses, we refine
the estimates of the year’s taxable income as new information becomes available,
including year-to-date financial results. This continual estimation process
often results in a change to our expected effective tax rate for the year. When
this occurs, we adjust the income tax provision during the quarter in which the
change in estimate occurs so that the year-to-date provision reflects the
expected annual tax rate. Significant judgment is required in determining our
effective tax rate and in evaluating our tax positions.
In
accordance with ASC 740, “Income Taxes” (previously SFAS No. 109, “Accounting
for Income Taxes,”) we recognize deferred tax assets and liabilities based on
the differences between the financial statement carrying amounts and the tax
basis of assets and liabilities. Deferred tax assets represent items to be used
as a tax deduction or credit in future tax returns for which we have already
properly recorded the tax benefit in the income statement. At least quarterly,
we assess the likelihood that the deferred tax asset balance will be recovered
from future taxable income. We take into account such factors as prior earnings
history, expected future earnings, carry-back and carry-forward periods, and tax
strategies that could potentially enhance the likelihood of a realization of a
deferred tax asset. To the extent recovery is unlikely, a valuation allowance is
established against the deferred tax asset and increasing our income tax expense
in the year such determination is made.
ASC
740-30, “Income Taxes – Other Considerations or Special Areas” (previously APB
Opinion No. 23, “Accounting for Income Taxes, Special Areas,”) does not require
U.S. income taxes to be provided on foreign earnings when such earnings are
indefinitely reinvested offshore. We periodically evaluate our investment
strategies with respect to each foreign tax jurisdiction in which we operate to
determine whether foreign earnings will be indefinitely reinvested offshore and,
accordingly, whether U.S. income taxes should be provided when such earnings
49
are
recorded. As of March 31, 2010, we believed all earnings generated in China
would be permanently reinvested and as a result, we did not record any income
taxes on such earnings.
We
adopted ASC 740-10, “Income Taxes” (previously FASB Interpretation No. 48,
“Accounting for Uncertainty in Income Taxes, an Interpretation of FASB Statement
No. 109”) effective April 1, 2007. In accordance with ASC 740-10, we recognize a
tax benefit associated with an uncertain tax position when, in our judgment, it
is more likely than not that the position will be sustained upon examination by
a taxing authority. For a tax position that meets the more-likely-than-not
recognition threshold, we initially and subsequently measure the tax benefit as
the largest amount that we judge to have a greater than 50% likelihood of being
realized upon ultimate settlement with a taxing authority. Our liability
associated with unrecognized tax benefits is adjusted periodically due to
changing circumstances, such as the progress of tax audits, case law
developments and new or emerging legislation. Such adjustments are recognized
entirely in the period in which they are identified. Our effective tax rate
includes the net impact of changes in the liability for unrecognized tax
benefits and subsequent adjustments as considered appropriate by management. As
of March 31, 2010, we had recorded liabilities of $1.0 million for our PRC
subsidiaries.
We
classify interest and penalties recognized on the liability for unrecognized tax
benefits as income tax expense. For the fiscal year ended March 31, 2010, the
unrecognized tax benefit did not change significantly and the amount of interest
and penalties related to uncertain tax position is immaterial.
Product
Recall
We
establish a reserve for product recall on a product-specific basis when
circumstances giving rise to the recall become known. Facts and circumstances
related to the recall, including where the product affected by the recall or
withdrawal is located (e.g., with consumers, in customers’ inventory, or in the
Company’s inventory), the expected product return rates by our distributor and
end-customers, cost estimates for shipping and handling for returns and
estimated replacement costs are considered when establishing a product recall
reserve. These factors are updated and reevaluated each period and the related
reserves are adjusted when these factors indicate that the recall reserve is
either not sufficient to cover or exceeds the estimated product recall
expenses.
Impairment
of Goodwill and Indefinite Lived Intangible Assets
We
account for goodwill and intangible assets with indefinite lives in accordance
with Accounting Standards Codification (ASC) No. 350-10, “Intangibles-Goodwill
and other – Overall” (previously Statement of Financial Accounting Standards
(“SFAS”) No. 142, “Goodwill and Other Intangible Assets”) or ASC No.350-10. ASC
No.350-10 states that goodwill and intangible assets with indefinite lives are
not amortized, but are instead reviewed for impairment annually (or more
frequently if impairment indicators arise). We conduct our annual impairment
testing on March 31 to determine if we will be able to recover all or a portion
of the carrying value of goodwill and intangible assets with indefinite
lives.
The
application of the impairment test requires judgment, including the
identification of reporting units, assignments of assets and liabilities to
reporting units and the determination of the fair value of each reporting unit.
Further, the impairment test involves the use of accounting estimates and
assumptions related to future operating results. Consistent with the
requirements of ASC No.350-10, the fair values of our reporting units are
generally based on discounted cash flow projections that are believed to be
reasonable under current and forecasted circumstances, the results of which form
the basis for making judgments about carrying values of the reported net assets
of our reporting units.
Prior to
performing the goodwill impairment testing process for a reporting unit under
ASC No.350-10, if there is reason to believe that other non-goodwill related
intangible assets may be impaired, these other intangible assets must first be
tested for impairment under ASC No.350-10 or ASC No.360-10 -35, “Property, plant
and equipment – Overall – Subsequent measurement” (previously SFAS No. 144,
“Accounting for the Impairment or Disposal of Long-Lived Assets”), or ASC
No.360-10 -35. Assets governed by ASC No.360-10 -35 require a recoverability
test for impairment whereby the gross undiscounted cash flows are determined
specific to the asset. For non-goodwill related intangible assets with
indefinite lives, a fair value determination is made. If the carrying value of
the asset
50
exceeds
the fair value, then impairment occurs. The carrying values of these assets are
impaired as necessary to provide the appropriate carrying value for the goodwill
impairment calculation.
These
impairment tests also involve the use of accounting estimates and assumptions
that management believed to be reasonable, the results of which form the basis
for our conclusions. Significant changes to these estimates and assumptions
could adversely impact our conclusion to these impairment tests. We
will continue to closely monitor our financial performance and projections for
our reporting units and the economic conditions of the product end-markets. Any
significant change in market conditions and estimates or judgments could give
rise to impairment in the period that the change becomes known.
No
goodwill or indefinite lived intangible assets have been impaired during any of
the periods presented.
Results
of Operations
The
following table sets forth, for the periods indicated, our consolidated
statements of income and certain other information, each expressed as a
percentage of net sales.
|
Fiscal
Years Ended March 31,
|
||||||||||||||||||||||||
|
2010
|
2009
|
2008
|
||||||||||||||||||||||
|
Amount
|
%
of Net Sales
|
Amount
|
%
of Net Sales
|
Amount
|
%
of Net Sales
|
|||||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||||||
|
Net
sales
|
$ | 291,886 | 100.0 | % | $ | 312,528 | 100.0 | % | $ | 362,090 | 100.0 | % | ||||||||||||
|
Cost
of sales
|
208,476 | 71.4 | % | 259,086 | 82.9 | % | 175,568 | 48.5 | % | |||||||||||||||
|
Gross
profit
|
83,410 | 28.6 | % | 53,442 | 17.1 | % | 186,522 | 51.5 | % | |||||||||||||||
|
Selling
and distribution expenses
|
43,989 | 15.1 | % | 44,178 | 14.1 | % | 34,449 | 9.5 | % | |||||||||||||||
|
Advertising
and promotion expenses
|
33,854 | 11.6 | % | 115,478 | 36.9 | % | 76,388 | 21.1 | % | |||||||||||||||
|
General
and administrative expenses
|
24,509 | 8.4 | % | 25,455 | 8.1 | % | 16,013 | 4.4 | % | |||||||||||||||
|
Impairment
loss
|
5,894 | 2.0 | % | — | — | — | — | |||||||||||||||||
|
Other
operating income, net
|
894 | 0.3 | % | 5,790 | 1.9 | % | 1,492 | 0.4 | % | |||||||||||||||
|
Income
(loss) from operations
|
(23,942 | ) | -8.2 | % | (125,879 | ) | -40.3 | % | 61,164 | 16.9 | % | |||||||||||||
|
Interest
expense
|
8,603 | 2.9 | % | 4,857 | 1.6 | % | 6,354 | 1.8 | % | |||||||||||||||
|
Interest
income
|
1,850 | 0.6 | % | 341 | 0.1 | % | 1,801 | 0.5 | % | |||||||||||||||
|
Other
expense, net
|
1,081 | 0.4 | % | 580 | 0.2 | % | 3,084 | 0.9 | % | |||||||||||||||
|
Income
(loss) before income tax expense (benefit)
|
(31,776 | ) | -10.9 | % | (130,975 | ) | -41.9 | % | 53,527 | 14.8 | % | |||||||||||||
|
Income
tax expense (benefit)
|
(6,904 | ) | -2.4 | % | (30,386 | ) | -9.7 | % | 7,855 | 2.2 | % | |||||||||||||
|
Net
income (loss)
|
(24,872 | ) | -8.5 | % | (100,589 | ) | -32.2 | % | 45,672 | 12.6 | % | |||||||||||||
|
Net
income (loss) attributable to the noncontrolling interest
|
(257 | ) | -0.1 | % | (40 | ) | -0.0 | % | 11 | 0.0 | % | |||||||||||||
|
Net
income (loss) attributable to Synutra International, Inc. common
shareholders
|
$ | (24,615 | ) | -8.4 | % | $ | (100,549 | ) | -32.2 | % | $ | 45,661 | 12.6 | % | ||||||||||
Fiscal
Year Ended March 31, 2010 Compared to Fiscal Year Ended March 31,
2009
Net
Sales
Net sales
for the fiscal year ended March 31, 2010 decreased by 6.6% to $291.9 million
from $312.5 million for the prior fiscal year. This decrease in net sales was
mainly due to the lingering impact of melamine contamination incident on our
powdered formula segment, partially offset by an increase in the sales of
surplus industrial milk powder of our “Other” business.
51
Powdered
formula segment
Net sales
of our powdered formula products, including infant powdered formula and other
powdered formula products for children and adults under our Super, U-Smart,
Mingshan and Helanruniu brand names accounted for 66.6% of our total sales for
the fiscal year ended March 31, 2010. Net sales of our powdered formula products
for the fiscal year ended March 31, 2010 decreased by 31.8% to $194.4 million
from $284.8 million for the prior fiscal year, primarily as a result of the
following factors:
|
|
·
|
Sales
volume of powdered formula products decreased by 19.6% to 24,438 tons for
the fiscal year ended March 31, 2010 from 30,383 tons for the prior fiscal
year, due primarily to a slow-down in sales activities following the
melamine contamination incident.
|
|
|
·
|
The
average selling price of our powdered formula products for the fiscal year
ended March 31, 2010 decreased by 15.1% to $7,954 per ton from $9,374 per
ton for the prior fiscal year. While our quoted product prices have not
changed significantly from August 2008 to March 2010 and our revenue mix
is shifting to higher-priced products, our average selling prices has
remained below pre-recall levels, because management believes more
aggressive rebates and discounts to distributors, which are recorded as a
reduction to our net sales, are important incentives to motivate our sales
and distribution networks in regaining market share. Also, beginning in
August 2009, distributors are responsible for organizing local promotion
activities, which were previously jointly organized by the distributors
and us, to make quicker response to market trends and to improve
efficiency. The compensation to distributors for this change are recorded
as reduction of net sales, instead of marketing expense. As we recover
from the melamine contamination incident, we believe the average selling
price should gradually return to pre-recall levels and we expect to
decrease the amount of rebates to our distributors. For example, the
average selling price of our powdered formula products for the fiscal
quarter ended March 31, 2010 was $8,619 per ton, compared to the
pre-recall level of $9,036 per ton for the fiscal quarter ended June 30,
2008.
|
Baby
food segment
Net sales
of baby food segment for the fiscal year ended March 31, 2010 was $886,000,
representing sales of prepared baby food, such as cooked meat and vegetables,
which began its operation in March 2009.
Nutritional
ingredients and supplements segment
Net sales
of nutritional ingredients and supplements segment for the fiscal year ended
March 31, 2010 was $1.5 million, representing chondroitin sulfate sold to third
parties. There were also inter-segment sales of $11.3 million of nutritional
ingredients, such as microencapsulated DHA and ARA, which were used in the
production of powdered infant formula products, as compared to $3.4 million fro
the fiscal year ended March 31, 2009. We did not have any external sales in this
segment for the fiscal year ended March 31, 2009.
Other
Other
sales for the fiscal year ended March 31, 2010 was $95.2 million, which mainly
included sales of surplus milk powder to industrial customers of $80.0 million,
as compared to $27.4 million for the prior fiscal year. The
significant increase was due to the fact that after the melamine contamination
incident, actual sales of powdered formula segment was lower than what we had
initially estimated which led to a build up of milk powder, both imported and
self-produced. We sold the surplus portion of the milk powder to industrial
customers during the fiscal year ended March 31, 2010 and generated immediate
cash. We regard this as an one-off event caused by the melamine contamination
incident and do not anticipate our other business to be significant going
forward.
Cost
of Sales
Cost of
sales for the fiscal year ended March 31, 2010 decreased by 19.5% to $208.5
million from $259.1 million for the prior fiscal year. The decrease in the cost
of sales is due primarily to a decrease in the sales volume of our powdered
formula products in the fiscal year ended March 31, 2010, and the product recall
cost which was recorded in the fiscal year ended March 31, 2009, and partially
offset by an increase in cost of sales of surplus industrial milk powder in the
fiscal year ended March 31, 2010.
52
Powdered
formula segment
Cost of
sales for the powdered formula products for the fiscal year ended March 31, 2010
decreased by 58.1% to $98.2 million from $234.5 million for the prior fiscal
year. The decrease in the cost of sales is due primarily to the lingering impact
of the melamine contamination incident on the sales volume of our powdered
formula products for the fiscal year ended March 31, 2010, and the significant
product recall cost recorded in the fiscal year ended March 31,
2009.
Baby
food segment
Cost of
sales of baby food segment for the fiscal year ended March 31, 2010 was
$793,000, representing cost of sales of prepared baby food, such as cooked meat
and vegetables, which began its operation in March 2009.
Nutritional
ingredients and supplements segment
Cost of
sales of nutritional ingredients and supplements segment for the fiscal year
ended March 31, 2010 was $2.7 million, representing chondroitin sulfate sold to
third parties. There were also inter-segment sales of nutritional ingredients,
such as microencapsulated DHA and ARA, which were used in the production of
powdered infant formula products. We did not have external cost of sales in this
segment for the fiscal year ended March 31, 2009.
Other
Other
cost of sales for the fiscal year ended March 31, 2010 was $106.8 million, which
mainly included the cost of sales of surplus milk powder of $85.4 million and
write-down of industrial milk powder of $6.7 million due to a decline in selling
price as a result of lower-priced domestically produced milk powder, as compared
to $24.5 million for the prior fiscal year. We do not anticipate the cost of
sales going forward to be as significant.
Gross
Profit and Gross Margin
As a
result of the foregoing, gross profit for the fiscal year ended March 31, 2010
increased by 56.1% to $83.4 million from $53.4 million for the prior fiscal
year. Gross profit for our powdered formula products for the fiscal year ended
March 31, 2010 increased by 91.1% to $96.2 million from $50.3 million for the
prior fiscal year. Gross profit for our baby food segment for the fiscal year
ended March 31, 2010 was $93,000. Gross loss for our nutritional ingredients and
supplements segment for fiscal year ended March 31, 2010 was $1.2 million, as
the business is still at its early stage and we were in the process of
developing a new sales channel. Gross loss for other business for the fiscal
year ended March 31, 2010 was $11.7 million, primarily due to the loss from
sales of domestically produced milk powder and write-down of domestically
produced milk powder in inventory. In order to improve our working capital
situation, we sold part of milk powder which was surplus to our own requirement
for the fiscal year ended March 31, 2010.
Our
overall gross margin increased to 28.6% for the fiscal year ended March 31, 2010
from 17.1% for the prior fiscal year. Our gross margin for powdered formula
products was 49.5% for the fiscal year ended March 31, 2010, as compared to
17.7% for the prior fiscal year. Our gross margin for baby food segment for the
fiscal year ended March 31, 2010 was 10.5%. Gross margin for nutritional
ingredients and supplements segment for the fiscal year ended March 31, 2010 was
negative 84.2%. Gross margin for other business for the fiscal year ended March
31, 2010 was negative 12.3%.
Selling
and Distribution Expenses
Selling
and distribution expenses for the fiscal year ended March 31, 2010 decreased
slightly by 0.4% to $44.0 million from $44.2 million for the prior fiscal year.
This decrease was a combined result of a decrease in freight charges and sales
administration expense due to tightened budgetary control, partially offset by
an increase in compensation expenses for our sales force. Total compensation for
our sales force for the fiscal year ended March 31, 2010 increased by 16.8% to
$25.3 million from $21.7 million for the prior fiscal year due to a pay raise
for the sales staff in the fiscal quarter ended June 30, 2009, partially offset
by a slight decrease in the number of sales staff to 2,917 as of March 31, 2010
from 2,997 as of March 31, 2009. Freight charges for the fiscal year ended March
31,
53
2010
decreased by 36.1% to $4.7 million from $7.4 million for prior fiscal year, due
primarily to the freight charges accrual for product recall in prior fiscal year
and a decrease of sales volume.
Advertising
and Promotion Expenses
Advertising
and promotion expenses for the fiscal year ended March 31, 2010 decreased
significantly by 70.7% to $33.9 million from $115.5 million for the prior fiscal
year. Advertising expenses for the fiscal year ended March 31, 2010, which
accounted for 49.3% of total advertising and promotion expenses, decreased by
77.1% to $16.7 million from $72.8 million for the prior fiscal year. Promotion
expenses for the fiscal year ended March 31, 2010, which accounted for 50.7% of
total advertising and promotion expenses, decreased by 59.8% to $17.2 million
from $42.7 million for the prior fiscal year, as certain promotions were carried
out directly by distributors in exchange for us providing such distributors with
an increase in product discount which was recorded as reduction to net sales.
Following aggressive advertising and promotional campaigns in the fiscal quarter
ended March 31, 2009 to regain market share in the immediate aftermath of the
melamine contamination incident, we reduced our advertising and promotion
expenses, and have refocused our efforts in the subsequent quarters to achieve
effective market pull-through with new customers and to improve brand loyalty.
The significant decrease reflected a redeployment of resources to support
activities on the consumer-end and beyond the distribution channels by our field
promoters in the communities and our nutrition education professionals at the
medical and healthcare facilities. The reduction of advertising and promotion
expenses also helped to improve our liquidity position in early quarters of the
fiscal year ended March 31, 2010. As our operations gradually returned to their
normal condition, we resumed since March 2010 our advertisement in major Chinese
TV stations and we expect to increase our advertising and promotion expenses
going forward.
General
and Administrative Expenses
General
and administrative expenses for the fiscal year ended March 31, 2010 decreased
slightly by 3.7% to $24.5 million from $25.5 million for the prior fiscal year.
The decrease in general and administrative expenses was a combined result of a
net contribution of $2.3 million made to the compensation fund set up by China
Dairy Industry Association for the settlement of existing and potential claims
in China from families of infants affected by melamine contamination and $1.8
million write-off of legal and professional fees incurred for the terminated
equity capital markets transaction for the prior fiscal year, partially offset
by an increase of $2.3 million in bad debt allowance in the fiscal year ended
March 31, 2010 due to extended credit term to help the recovery of distributors
and termination of certain distributorship after the melamine contamination
incident.
Impairment
Loss
We
recorded impairment loss of $5.9 million for the fiscal year ended March 31,
2010 which represented the difference between the estimated fair value and
carrying value of the assets sold by our subsidiaries, Baoquanling and Cow
Breeding.
Other
operating income, net
Other
operating income for the fiscal year ended March 31, 2010 decreased to $894,000
from $5.8 million for the prior fiscal year. Other operating income for the
fiscal year ended March 31, 2010 represents a general purpose government subsidy
from a local government.
Interest
Expense
Interest
expense for the fiscal year ended March 31, 2010 increased to $8.6 million from
$4.9 million for the prior fiscal year, due primarily to the significant
increase in bank borrowings after the melamine contamination
incident.
Interest
Income
Interest
income for the fiscal year ended March 31, 2010 increased to $1.9 million from
$0.3 million for the prior fiscal year, due primarily to the increased cash
deposit pledged for short-term loans, which had a higher interest rate than cash
equivalents.
54
Income
Tax Benefit
As a
result of the net loss, we recorded an income tax benefit of $6.9 million for
the fiscal year ended March 31, 2010, as compared to $30.4 million for prior
year. Our effective tax rate decreased to 21.7% for the fiscal year ended March
31, 2010 from 23.2% for the prior fiscal year. The decrease in the
effective tax rate was driven primarily by the valuation allowance for net
operating loss carryforward of certain subsidiaries and tax credits carryforward
for PRC equipment purchasing, partially offset by the effect of tax refund which
represented refund of overpaid income tax prior to the melamine contamination
incident.
Net
Loss Attributable to Synutra International, Inc. Common
Shareholders
As a
result of the foregoing, net loss attributable to Synutra International, Inc.
for the fiscal year ended March 31, 2010 was $24.6 million, as compared to net
loss of $100.5 million for the prior fiscal year.
Fiscal
Year Ended March 31, 2009 Compared to Fiscal Year Ended March 31,
2008
Net
Sales
Net sales
for the fiscal year ended March 31, 2009 decreased by 13.7% to $312.5 million
from $362.1 million for the prior fiscal year. This decrease in net sales was
mainly due to the significant product recall and decrease in sales which
resulted from the melamine contamination incident, partially offset by the
increases in volume and average selling price before the melamine contamination
incident.
Powdered
formula segment
Net sales
of our powdered formula products, including infant milk formula and other
powdered formula products for children and adults under our Super, U-Smart,
Mingshan and Helanruniu brand names, accounted for 91.1% of our total sales for
the fiscal year ended March 31, 2009. Net sales of our powdered formula products
for the fiscal year ended March 31, 2009 decreased by 11.3% to $284.8 million
from $321.1 million for the prior fiscal year, primarily as a result of the
following factors:
|
|
·
|
Sales
volume of powdered formula products decreased by 26.5% to 30,383 tons for
the fiscal year ended March 31, 2009 from 41,359 tons for the prior fiscal
year, due primarily to a slow-down in sales activities in the second half
of September 2008 to March 2009 following the melamine contamination
incident, partially offset by significant business growth experienced in
the months from April to August in
2008.
|
|
|
·
|
The
average selling price of our powdered formula products for the fiscal year
ended March 31, 2009 increased by 20.7% to $9,374 per ton from $7,764 per
ton for the prior fiscal year. This increase in average selling price was
a combined result of a greater proportion of higher-priced products in our
product mix and an increase of our sales price in August
2008.
|
Baby
food segment
We
acquired the prepared baby food business from Huilian in October 2008. The
business began to generate sales for us in March 2009, and we are currently in
the process of providing shipments of prepared baby food to be placed in
stores.
Nutritional
ingredients and supplements segment
There
were no external sales of nutritional ingredients and supplements segment for
the fiscal year ended March 31, 2009. There were inter-segment sales of
nutritional ingredients, such as microencapsulated DHA and ARA, which were used
in the production of powdered infant formula products. We did not have any net
sales in this segment in the fiscal year ended March 31, 2008.
55
Cost
of Sales
Cost of
sales for the fiscal year ended March 31, 2009 increased by 47.6% to $259.1
million from $175.6 million for the prior fiscal year. The increase in the cost
of sales is due primarily to the increase of product recall related cost,
partially offset by a decrease in the sales volume of our powdered formula
products.
Powdered
formula segment
Cost of
sales for the powdered formula products for the fiscal year ended March 31, 2009
increased by 64.7% to $234.5 million from $142.4 million for the prior fiscal
year. The increase in the cost of sales is due primarily to increase of product
recall related cost, partially offset by a decrease in the sales volume of our
powdered formula products. The portion of the product recall cost, which has
been recognized in cost of sales for the fiscal year ended March 31, 2009, was
$96.6 million, reflecting the cost of recalled products of $48.1 million and the
write-down and write-off of affected inventory of $48.5 million. The sales
volume of powdered formula products sold for the fiscal year ended March 31,
2009 decreased by 10,976 tons as compared to the prior fiscal year.
Baby
food segment
We
acquired the prepared baby food business from Huilian in October 2008. The
business began to generate sales for us in March 2009.
Nutritional
ingredients and supplements segment
There
were no external cost of sales of nutritional ingredients and supplements
segment for the fiscal year ended March 31, 2009. There were inter-segment cost
of sales of nutritional ingredients, such as microencapsulated DHA and ARA,
which were used in the production of powdered infant formula products. We did
not have any cost of sales for this segment in the fiscal year ended March 31,
2008.
Gross
Profit and Gross Margin
As a
result of the foregoing, gross profit for the fiscal year ended March 31, 2009
decreased by 71.4% to $53.4 million from $186.5 million for the prior fiscal
year. Gross profit for our powdered formula products for the fiscal year ended
March 31, 2009 decreased by 71.9% to $50.3 million from $178.7 million for the
prior fiscal year due primarily to the significant costs in relation to product
recall including the cost of recalled products and the write-down and write-off
of affected inventory, and due also to decreased sales in the last three fiscal
quarters of the fiscal year ended March 31, 2009 following the melamine
contamination incident. Gross profit for our baby food segment and nutritional
ingredients and supplements segment for the fiscal year ended March 31, 2009
were $0.2 million and nil, respectively.
Our
overall gross margin decreased to 17.1% for the fiscal year ended March 31, 2009
from 51.5% for the prior fiscal year. Our gross margin for powdered formula
products was 17.7% for the fiscal year ended March 31, 2009, as compared to
55.7% for the prior fiscal year. The decrease in our gross margin for powdered
formula products was primarily due to the significant cost of the product recall
and increased free products offered to our customers in the aftermath of the
melamine contamination incident, partially offset by an increase in the
proportion of sales of our higher margin infant formula products. Our gross
margin for baby food segment and nutritional ingredients and supplements segment
for the fiscal year ended March 31, 2009 were 5.8% and nil,
respectively.
Selling
and Distribution Expenses
Selling
and distribution expenses for the fiscal year ended March 31, 2009 increased by
28.5% to $44.2 million from $34.4 million for the prior fiscal year. This
increase was primarily due to an increase in compensation expenses for our sales
force, an increase in travel and communication expenses and the freight charges
relating to product recall and replacement. Total compensation for our sales
force for the fiscal year ended March 31, 2009 increased by 49.7% to $21.7
million from $14.5 million for the prior fiscal year. This increase was
primarily due to the compensation paid to the field promoters (nutrition
consultants), who were previously paid by our distributors on a commission
basis, during the months of October to December 2008 following the melamine
contamination incident. We assumed such expenses in order to retain the services
of these field promoters as their commission-based salaries
56
decreased
significantly during this period. In addition, the increase in the number of
sales staff to 2,997 as of March 31, 2009 from 2,780 as of March 31, 2008 also
contributed to the increase in compensation expense relating to our sales force.
Travel and communication expenses for the fiscal year ended March 31, 2009
increased by 69.0% to $7.1 million from $4.2 million for the prior fiscal year,
due mainly to the expansion of our business. The freight charges of $2.6 million
relating to product recall and replacement also contributed to the increase in
selling and distribution expenses.
Advertising
and Promotion Expenses
Advertising
and promotion expenses for the fiscal year ended March 31, 2009 increased by
51.2% to $115.5 million from $76.4 million for the prior fiscal year.
Advertising expenses for the fiscal year ended March 31, 2009, which accounted
for 63.0% of total advertising and promotion expenses, increased by 140.3% to
$72.8 million from $30.3 million for the prior fiscal year, due primarily to our
intensified efforts to improve our corporate image and brand name and to recover
our lost market share following the melamine contamination incident. Promotion
expenses for the fiscal year ended March 31, 2009, which accounted for 37.0% of
total advertising and promotion expenses, decreased by 7.4% to $42.7 million
from $46.1 million for the prior fiscal year.
General
and Administrative Expenses
General
and administrative expenses for the fiscal year ended March 31, 2009 increased
by 59.4% to $25.5 million from $16.0 million for the prior fiscal year. The
increase in general and administrative expenses was primarily due to a net
contribution of $2.3 million made to the compensation fund set up by China Dairy
Industry Association for the settlement of existing and potential claims in
China from families of infants affected by melamine contamination, an increase
of $2.6 million in salary and social insurance as result of the increased
headcount, an increase of $1.7 million in rental expenses for new office space,
and an increase of $2.2 million in bad debt expense due to increased accounts
receivable balance and risk caused by melamine contamination
incident.
Other
operating income, net
Other
operating income for the fiscal year ended March 31, 2009 increased by 286.7% to
$5.8 million from $1.5 million for the prior fiscal year. The increase in other
operating income was primarily due to a grant received from local government in
recognition of our significant contribution to the local economy.
Interest
Expense
Interest
expense for the fiscal year ended March 31, 2009 decreased to $4.9 million from
$6.4 million for the prior fiscal year, due primarily to the amortization of
debt discount associated with the issuance of warrants to RBS which amounted to
$2.6 million in the fiscal year ended March 31, 2008, which was offset by the
increased interest expenses due to significant borrowing after the melamine
contamination incident in the fiscal year ended March 31, 2009.
Interest
Income
Interest
income for the fiscal year ended March 31, 2009 decreased to $0.3 million from
$1.8 million for the prior fiscal year due to significant decrease in our cash
and cash equivalent balances.
Income
Tax Benefit (Expense)
As a
result of the net loss arising primarily from the cost of the product recall, we
recorded an income tax benefit of $30.4 million for the fiscal year ended March
31, 2009, as compared to an income tax expense of $7.9 million for the prior
fiscal year. Our effective tax rate increased to 23.2% for the fiscal year ended
March 31, 2009 from 14.7% for the prior fiscal year. This increase in our
effective income tax rate was primarily due to the expirations of tax holidays
enjoyed by certain subsidiaries in China.
57
Net
Loss Attributable to Synutra International, Inc. Common
Shareholders
As a
result of the foregoing, net loss attributable to Synutra International, Inc.
common shareholders for the fiscal year ended March 31, 2009 was $100.5 million,
as compared to net income of $45.7 million for the prior fiscal
year.
Liquidity
and Capital Resources
The
consolidated financial statements of the Company have been prepared in
accordance with accounting principles generally accepted in the United States of
America (“US GAAP”). The accompanying consolidated financial statements have
been prepared assuming the Company will continue as a going concern. Under that
assumption, it is expected that assets will be realized and liabilities will be
satisfied in the normal course of business. As a result of the melamine
contamination incident and subsequent loss of sales, the Company experienced
significant operating losses, negative cash flows from operations, a working
capital deficit and non-compliance with certain covenants of its loan agreement
with RBS, and the Company included a statement raising substantial doubt about
its ability to continue as a going concern in Note 2 of its financial statements
included in its annual report on form 10-K for the fiscal year ended March 31,
2009.
The
melamine contamination incident has significantly impacted our liquidity. Since
the damage to our reputation caused by the melamine contamination incident will
take time to recover, the net sales of our powdered formula products were
negatively impacted for the fiscal years ended March 31, 2010 and 2009, and we
incurred substantial cash outflow for the purchase of raw materials and for
operating expenses during those fiscal years.
The
Company’s status has improved in the fiscal year ended March 31, 2010. Although
the full year operating result was still a loss and the full year cash flows
generated from operating activities was still negative, the fourth quarter
income from operations was a profit of $11.8 million and the third and fourth
quarter cash flows from operations was a net inflow of $19.4 million and $17.4
million, respectively. Other than the profit and operating cash generated, the
Company took other measurements to overcome liquidity difficulties, including
the following:
|
|
·
|
Compliance
with debt covenants: In conjunction with the negotiation with RBS, the
Company was able to resolve the debt covenant noncompliance issue and has
been and is expected to be in full compliance with the renegotiated debt
covenant through the debt maturity.
|
|
|
·
|
Availability
of credit facilities: The Company has full and unrestricted access to
committed and available bank standby credit facilities of $96.8 million as
of March 31, 2010.
|
|
|
·
|
Restructure
of indebtedness: Part of the Company’s debt has been restructured
subsequent to the balance sheet date. From April 1, 2010 to June 1, 2010,
in order to further improve working capital condition, the Company prepaid
short-term and long-term borrowings from PRC banks in the amount of $49.8
million, with original maturity date ranging from September 2010 to April
2011 and with weighted average interest rate of 5.5%, and borrowed
long-term loan from the same PRC banks in the same amount, with maturity
date ranging from August 2011 to May 2012 and with weighted average
interest rate of 5.4%.
|
|
|
·
|
Disposal
of assets: The Company sold assets of Baoquanling and Cow Breeding, at the
price of $28.9 million, to Wondersun. By the end of fiscal year 2010, the
Company had collected the proceeds of $20.2
million.
|
|
|
·
|
Sales
of surplus industrial milk powder: during the fiscal year ended March 31,
2010, the Company sold surplus milk powder to industrial customers for
$80.0 million, most of which has generated operating cash flow for the
Company.
|
As a
result, the working capital deficit had narrowed to $27.6 million as of March
31, 2010 from $80.4 million as of March 31, 2009.
Prior to
the melamine contamination incident, cash generated from our operating
activities was sufficient for normal operating needs, and financing from banks
was used to fund investing activities such as the expansion of our manufacturing
plant.
58
We
believe that the product recall has been substantially completed as of March 31,
2010 and do not expect further material cash outflows in connection
with the product recall.
Our cash
and cash equivalent balance increased to $48.7 million at March 31, 2010, as
compared to $37.7 million at March 31, 2009.
The
following table sets forth, for the periods indicated, certain information
relating to our cash flows:
|
Fiscal
Year Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands)
|
||||||||||||
|
Net
cash provided by (used in) operating activities
|
$ | (11,824 | ) | $ | (109,488 | ) | $ | 38,229 | ||||
|
Net
cash provided by (used in) investing activities
|
56,529 | (127,287 | ) | (33,096 | ) | |||||||
|
Net
cash provided by (used in) financing activities
|
(33,804 | ) | 174,871 | 64,731 | ||||||||
|
Effect
of foreign currency translation on cash and cash
equivalents
|
56 | 2,215 | 6,725 | |||||||||
|
Net
cash flow
|
$ | 10,957 | $ | (59,689 | ) | $ | 76,589 | |||||
Cash
Flows from Operating Activities
Net cash
used in operating activities was $11.8 million for the fiscal year ended March
31, 2010, as compared to $109.5 million for the prior fiscal year. Net cash used
in operating activities for the fiscal year ended March 31, 2010 included net
loss of $24.9 million, non-cash items not affecting cash flows of $18.2 million,
and a $5.2 million increase in working capital. The changes in working capital
for the fiscal year ended March 31, 2010, were primarily related to a $61.3
million decrease in inventories due to sales of surplus industrial milk powder,
and a $57.2 million decrease in accounts payable due to the settlement of
certain accumulated extended liabilities. In the fiscal year ended March 31,
2010, we spent $217.7 million to purchase raw materials and other production
materials, $38.1 million in staff compensation and social welfare, $33.2 million
in other taxes, $86.4 million in selling and distribution, advertising and
promotion, and general and administrative expenses, and received $374.7 million
from our customers.
Net cash
used in operating activities was $109.5 million for the fiscal year ended March
31, 2009, as compared to net cash provided by operating activities of $38.2
million for the prior fiscal year. Net cash used in operating activities for the
fiscal year ended March 31, 2009 included net loss of $100.5 million, non-cash
items not affecting cash flows of $20.0 million, and a $11.1 million increase in
working capital. The changes in working capital for the fiscal year ended March
31, 2009, were primarily related to a $50.9 million increase in inventories due
to the increase in goods-in-transit for imported milk powder, a $77.0 million
increase in accounts payable due to a credit term extension from our suppliers
resulting from our tightened liquidity position and payable for goods-in-transit
for imported milk powder, a $13.4 million increase in accounts receivable due to
our extended credit term to certain distributors, a $5.6 million increase in
income tax receivable due to prepaid income tax before the melamine
contamination incident, and a $4.5 million increase in product recall provision.
For the fiscal year ended March 31, 2009, we spent $238.8 million to purchase
raw materials and other production materials, $33.0 million in staff
compensation and social welfare, $33.8 million in other taxes, $197.1 million in
selling and distribution, advertising and promotion, and general and
administrative expenses, and received $403.8 million from our
customers.
Net cash
provided by operating activities was $38.2 million for the fiscal year ended
March 31, 2008. Net cash provided by operating activities for the fiscal year
ended March 31, 2008 included net income of $45.7 million, non-cash items not
affecting cash flows of $5.6 million, partially offset by $13.0 million of
negative changes in working capital. The changes in working capital for the year
ended March 31, 2008, were primarily related to $39.1 million increase in
inventory, $5.7 million increase in prepaid expense and other current assets,
$4.1 million increase in accounts receivable, and partially offset by $9.4
million decrease in due from related parties, $6.8 million increase in accounts
payable, and $8.1 million increase in other liabilities. The increase in
inventory was due to several factors: our increased production and sales, the
acquisition of Baoquanling and its inventory, and direct whey protein purchase
from Eurosérum in France. Due to the distance of Eurosérum and the longer
delivery time, we kept more whey protein inventory in our warehouse and had more
whey protein in-transit as our inventory. In the fiscal year ended March 31,
2008, we spent $198.4 million to purchase raw materials and other production
materials, $21.7
59
million
in staff compensation and social welfare, $19.7 million in other taxes, $143.5
million in selling and distribution, advertising and promotion, and general and
administrative expenses, and received $431.5 million from our
customers.
Cash
Flows from Investing Activities
Net cash
provided by investing activities was $56.5 million for the fiscal year ended
March 31, 2010, as compared to net cash used in investing activities of $127.3
million for the prior fiscal year. Cash invested in purchases of property and
equipment was $13.3 million and $44.9 million for the fiscal year ended March
31, 2010 and 2009, respectively. The decrease is mainly due to the suspension of
major investing projects in the aftermath of the melamine contamination
incident. We expect the suspension will slow down our expansion into the baby
food business. Cash inflow from restricted cash was $51.1 million for the fiscal
year ended March 31, 2010 due to repayment of certain bank borrowings which was
secured by cash, as compared to cash outflow for restricted cash of $73.9
million for the prior fiscal year. Restricted cash represents cash deposited
with banks as security against the issuance of letters of credit for the import
of machinery and raw materials and as pledges for certain short-term
borrowings.
Net cash
used in investing activities was $127.3 million for the fiscal year ended March
31, 2009, as compared to $33.1 million for the prior fiscal year. Cash invested
in purchases of property and equipment was $44.9 million and $36.5 million for
the fiscal year ended March 31, 2009 and 2008, respectively. This increase in
net cash used in investing activities is primarily due to our plant expansion to
increase our production capacity prior to the melamine contamination incident,
partially offset by the suspension of major investing projects in the aftermath
of such event. We expect the suspension will slow down our expansion into the
baby food business. Cash outflow for restricted cash was $73.9 million for the
fiscal year ended March 31, 2009, as compared to cash inflow from restricted
cash of $4.2 million for the prior fiscal year due to a significant increase in
certain bank borrowings which was pledged by cash. Restricted cash represents
cash deposited with banks as security against the issuance of letters of credit
for the import of machinery and raw materials and as pledges for certain
short-term borrowings.
Net cash
used in investing activities was $33.1 million for the fiscal year ended March
31, 2008. This was due primarily to our plant expansion to increase our
production capacities. Cash invested in purchases of property and equipment was
$36.5 million for the fiscal years ended March 31, 2008. Cash inflow from
restricted cash was $4.2 million for the fiscal year ended March 31, 2008.
Restricted cash represents cash deposited with banks as security against the
issuance of letters of credit.
Cash
Flows from Financing Activities
Net cash
used in financing activities was $33.8 million for the fiscal year ended March
31, 2010. Cash provided by financing activities during the fiscal year ended
March 31, 2010 was primarily related to $337.7 million in short-term loans from
PRC banks in China and a company controlled by our Chairman and Chief Executive
Officer, $32.2 million in long-term loans from domestic banks in China, offset
by $403.7 million repayment of short-term loans to banks in China.
Net cash
provided by financing activities was $174.9 million for the fiscal year ended
March 31, 2009. Cash provided by financing activities during the fiscal year
ended March 31, 2009 was primarily related to $238.2 million in short-term loans
from banks in China, $7.5 million loan from related parties, $8.8 million in
long-term loans from banks in China, offset by $77.3 million repayment of
short-term loans from PRC banks and $1.9 million repayment of long-term loans
from PRC banks.
Net cash
provided by financing activities was $64.7 million for the fiscal year ended
March 31, 2008. Cash provided by financing activities during the fiscal year
ended March 31, 2008 was primarily related to $65.8 million in the proceeds from
the issuance of common stock to Warburg, $60.2 million in short-term loans from
banks in China and RBS, and a $35.0 million in long-term loan from RBS, offset
by the repayment of $96.2 million in short-term borrowings from banks in China
and RBS.
60
Outstanding
Indebtedness
On April
19, 2007, the Company entered into a bridge loan agreement with ABN AMRO Bank
N.V., Hong Kong branch (now known as The Royal Bank of Scotland N.V. (“RBS”), in
the amount of $35.0 million (the “Bridge Loan Agreement”). Among other terms,
pursuant to a USD facility side letter and warrant agreement dated April 19,
2007 between the Company and RBS, the Company was obligated to issue warrants to
RBS to purchase up to 400,000 shares of the Company’s common stock. On April 19,
2007 and October 11, 2007, respectively, the Company issued warrants to purchase
200,000 shares of its common stock at $8.84 per share and the remaining 200,000
shares at the same price. All of the warrants may be exercised up to the third
anniversary of the completion of a “Qualified Public Offering”, as defined in
the warrant agreement.
On
October 11, 2007, RBS and another lender provided a three year term loan (the
“Original Loan”), to the Company in the aggregate amount of $35.0 million. The
principal amount, and any unpaid accrued interest on the Original Loan, will be
due on October 11, 2010 and may be prepaid without penalty. The Original Loan
bears interest at LIBOR for deposits in U.S. dollars plus 3.5% with interest
payable on the last day of each three-month period. The loan is secured by a
pledge of 10,000 shares of common stock, or 100% of the outstanding common stock
of Synutra Illinois.
The loan
agreement for the Original Loan (the “Original Loan Agreement”) provides for
mandatory prepayment upon the occurrence of certain events, and contains
customary covenants for financings of this type, including restrictions on the
incurrence of liens, payment of dividends, and disposition of properties. The
Original Loan Agreement also contains certain financial covenants, including a
requirement to maintain specified leverage ratio, interest coverage ratio,
tangible net worth, and indebtedness to tangible net worth ratio. The Company
has performed an analysis of the relevant ratios and found that due to the net
loss which resulted from the significant costs of the product recall, including
inventory write-down and write-off and subsequent loss of sales, the Company has
not been able to meet two of the financial covenants since September 30, 2008
and all four financial covenants since December 31, 2008. The Company requested
the lenders to waive these breaches and to reset the financial covenants for the
following quarters, and placed $5.0 million as cash deposit. On February 26,
2010, the Company entered into an amendment (the “Amendment”) to its Original
Loan Agreement with RBS as administrative agent, as collateral agent and as
arranger, and certain lenders party thereto (the “Lenders”). Pursuant to the
Amendment, the Lenders and RBS agreed to waive their rights and remedies of the
Company’s breaches of certain covenants under the Original Loan Agreement in
exchange for a waiver fee equal to 0.5% of the aggregate principal amount
outstanding under the Original Loan Agreement as of February 26, 2010, or
approximately $175,000. In addition, the Amendment amends the Original Loan
Agreement, among other things, to increase the interest rate of the loan from
LIBOR plus 3.5% to LIBOR plus 4.5%, to place additional cash deposits under
certain circumstances, to increase the cash deposit to an aggregate amount not
in excess of $1 0.0 million, and replace the financial covenants with a single
requirement to maintain consolidated earnings before interest, taxes,
depreciation and amortization (EBITDA) that is no lower than the minimum
Consolidated EBITDA specified in the Original Loan Agreement, as amended. The
Company has performed an analysis on the consolidated EBITDA and confirmed that
the financial covenant was satisfied as of March 31, 2010.
In
addition to the RBS Loans, as of March 31, 2010 and 2009, the Company had
short-term debt from PRC banks in the amount of $94.2 million and $186.2
million, respectively. The maturity dates of the short-term debt
outstanding range from April 2010 to March 2011. The weighted average interest
rate on short-term debt from PRC banks outstanding at March 31, 2010 and 2009
was 4.7% and 4.1%, respectively. The loans at March 31, 2010 and 2009 were
secured by the pledge of certain fixed assets held by the Company of $33.4
million and $34.2 million, respectively; the pledge of the Company’s land use
rights of $2.2 million and $3.4 million, respectively; and the pledge of cash
deposits of $11.7 million and $66.1 million, respectively.
As of
March 31, 2010 and 2009, the Company had long-term debt, including current
portion, from PRC banks in the amount of $67.4 million and $8.8 million,
respectively. The maturity date of the long-term loans and long term loans due
within one year outstanding from PRC banks at March 31, 2010 is from November
2010 to January 2013. The weighted average interest rate of outstanding
long-term loans at March 31, 2010 and 2009 was 5.4% and 5.4%, respectively. The
Company had $0.9 million and nil of loans at March 31, 2010 and 2009,
respectively, that were secured by the pledge of the Company’s land use
right.
61
As of
March 31, 2010, the Company had unused credit facility of $96.8 million from
certain PRC banks. The interest rate will be negotiated by both parties upon
future drawdown.
Apart
from borrowings from banks, the Company had short term loans from related
parties in the amount of $3.9 million and $7.5 million as of March 31, 2010 and
2009, respectively. The maturity dates of related party loans at March 31, 2010
are from October 2010 to January 2011, and are extendable on the same terms upon
maturity.
Tabular
Disclosure of Contractual Obligations
Our cash
flows from operations are dependent on a number of factors, including
fluctuations in our operating results, accounts receivable collections,
inventory management, and the timing and amount of tax and other payments. We
plan for and measure our liquidity and capital resources through an annual
budgeting process.
Below is
a table setting forth our contractual obligations as of March 31,
2010:
|
Total
|
Less
than
1
year
|
1-3
years
|
3-5
years
|
More
than
5
years
|
||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||
|
Long-term
debt and related interest payment obligations
|
$ | 108,827 | $ | 66,011 | $ | 42,816 | $ | — | $ | — | ||||||||||
|
Capital
lease obligations
|
17,784 | 473 | 946 | 946 | 15,419 | |||||||||||||||
|
Operating
lease obligations
|
66,400 | 1,590 | 3,683 | 3,539 | 57,588 | |||||||||||||||
|
Advertising
commitments
|
18,910 | 18,910 | — | — | — | |||||||||||||||
|
Capital
expenditure commitments
|
2,547 | 2,494 | 53 | — | — | |||||||||||||||
|
Total
|
$ | 214,468 | $ | 89,478 | $ | 47,498 | $ | 4,485 | $ | 73,007 | ||||||||||
We
computed the long-term debt-related interest based on the interest rate as of
March 31, 2010.
As of
March 31, 2010, our uncertain income tax liability was $1.0 million. We are
unable to reasonably estimate the timing of the effective settlement of this tax
position.
Capital
Expenditures
Our
capital expenditures were $13.3 million, $44.8 million and $36.5 million for the
fiscal years ended March 31, 2010, 2009 and 2008, respectively. Our planned
capital expenditures for the fiscal year ending March 31, 2011 will be for
expanding production facilities and building research and development center.
However, the actual use of the capital expenditure may differ depending on our
cash flow status.
Off-Balance
Sheet Arrangements
We do not
have off-balance sheet arrangements that have or are reasonably likely to have a
current or future effect on our financial condition, changes in financial
condition, revenues or expenses, results of operations, liquidity, capital
expenditures or capital resources that is material to investors.
Recent
Accounting Pronouncements
In
January 2010, the FASB issued ASU 2010-06, Improving Disclosures about Fair
Value Measurements. The ASU amends ASC 820 (formerly Statement No. 157, Fair
Value Measurements) to add new requirements for disclosures about transfers into
and out of Levels 1 and 2 and separate disclosures about purchases, sales,
issuances, and settlements relating to Level 3 measurement on a gross basis
rather than as a net basis as currently required. ASU 2010-06 also
clarifies existing fair value disclosures about the level of disaggregation and
about inputs and valuation techniques used to measure fair value. ASU
2010-06 is effective for annual and interim periods beginning after December 15,
2009, except for the requirement to provide the level 3 activity of purchases,
sales, issuances, and settlements on a gross basis, which will be effective for
annual and interim periods beginning after December 15, 2010. Early
application is permitted and in the period of initial adoption, entities are not
required to provide the amended disclosures for any previous periods presented
for comparative purposes. We do not expect significant impact of adoption
on its consolidated financial statements.
62
ITEM
7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET
RISK
The
objective of our policies is to mitigate potential income statement, cash flow
and fair value exposures resulting from possible future adverse fluctuations in
interest rates and foreign exchange rates. We evaluate our exposure to market
risk by assessing the anticipated near-term and long-term fluctuations in
interest rates and foreign exchange rates. This evaluation includes the review
of leading market indicators, discussions with financial analysts and banks
regarding current and future economic conditions and the review of market
projections.
Foreign
Exchange Risk
The value
of the Renminbi against the U.S. dollar and other currencies is affected by,
among other things, changes in China’s political and economic conditions. Since
July 2005, the Renminbi has no longer been pegged to the U.S. dollar. Although
the PBOC regularly intervenes in the foreign exchange market to prevent
significant short-term fluctuations in the exchange rate, the Renminbi may
appreciate or depreciate significantly in value against the U.S. dollar in the
medium to long term. Moreover, it is possible that in the future, PRC
authorities may lift restrictions on fluctuations in the Renminbi exchange rate
and lessen intervention in the foreign exchange market.
Because
our earnings and cash assets are denominated in Renminbi, appreciation or
depreciation in the value of the Renminbi relative to the U.S. dollar would
affect our financial results reported in U.S. dollar terms without giving effect
to any underlying change in our business or results of operations. As a result,
we face exposure to adverse movements in currency exchange rates as the
financial results of our Chinese operations are translated from local currency
into U.S. dollars upon consolidation. If the U.S. dollar weakens against the
Renminbi, the translation of our foreign-currency-denominated balances will
result in increased net assets, net revenues, operating expenses, and net income
or loss. Similarly, our net assets, net revenues, operating expenses, and net
income or loss will decrease if the U.S. dollar strengthens against the
Renminbi. Additionally, foreign exchange rate fluctuations on transactions
denominated in Renminbi other than the functional currency results in gains and
losses that are reflected in our Consolidated Statement of Income. Our
operations are subject to risks typical of international business, including,
but not limited to, differing economic conditions, changes in political climate,
differing tax structures, other regulations and restrictions, and foreign
exchange rate volatility.
Considering
the RMB balance of our cash and cash equivalents as of March 31, 2010, which
amounted to $48.7 million, a 1.0% change in the exchange rates between the
Renminbi and the U.S. dollar would result in an increase or decrease of
approximately $0.5 million of the balance.
To date,
we have not entered into any hedging transactions in an effort to reduce our
exposure to foreign currency exchange risk as very limited hedging transactions
are available in China to reduce our exposure to exchange rate fluctuations.
While we may enter into hedging transactions in the future, the availability and
effectiveness of these transactions may be limited, and we may not be able to
successfully hedge our exposure at all. In addition, our foreign currency
exchange losses may be magnified by PRC exchange control regulations that
restrict our ability to convert Renminbi into foreign currencies.
Inflation
Inflationary
factors, such as increases in the cost of our products and overhead costs, could
impair our operating results. Although we do not believe that inflation has had
a material impact on our financial position or results of operations to date, a
high rate of inflation in the future may have an adverse effect on our ability
to maintain current levels of gross margin and selling, general and
administrative expenses as a percentage of sales revenue if the selling prices
of our products do not increase with these increased costs.
Interest
Rate Risk
We did
not experience any material changes in interest rate exposures during the fiscal
year ended March 31, 2010. We currently do not use interest rate swaps to manage
exposure to interest rate changes.
Currently,
we are exposed to interest rate risk primarily associated with: the RBS Loan, a
variable-rate debt obligation based on LIBOR, with the carrying value of $35.0
million which approximates its fair value at March 31,
63
2010. A
1.0% change in the LIBOR would result in an increase or decrease of
approximately $350,000 in our annual interest expense.
Concentration
of Credit Risk
We are
subject to concentrations of credit risk consisting primarily of accounts
receivable. We perform ongoing credit evaluations with respect to the financial
condition of our debtors, but do not require collateral. In order to determine
the value of our accounts receivable, we record a provision for doubtful
accounts to cover probable credit losses. Our management reviews and adjusts
this allowance periodically based on historical experience and its evaluation of
the collectibility of outstanding accounts receivable.
Commodities
Risk
Raw milk,
imported milk powder and whey protein are the principal raw materials in the
Company’s business, accounting for over 70% of the Company’s production costs.
Domestically produced milk powder and other dairy based ancillary products are
also the Company’s products for commercial sales. As such, the Company is
exposed to the fluctuations in the price of raw milk, milk powder and whey
protein. During the past two fiscal years, the rise and fall in the prices of
these commodities have affected the Company’s business operations and
profitability. A significant rise in the prices of these raw materials will
adversely affect the gross margin of the Company’s powdered formula products,
but may also adversely affect the gross margin of domestically produced milk
powder and other dairy based ancillary products for commercial
sales.
ITEM
8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
INDEX
TO CONSOLIDATED FINANCIAL STATEMENTS
|
Report
of Independent Registered Public Accounting Firms
|
64
|
|
Consolidated
Balance Sheets as of March 31, 2010 and 2009
|
66
|
|
Consolidated
Statements of Income for the fiscal years ended March 31, 2010, 2009 and
2008
|
67
|
|
Consolidated
Statements of Comprehensive Income (Loss) for the fiscal years ended March
31, 2010, 2009 and 2008
|
68
|
|
Consolidated
Statements of Equity for the fiscal years ended March 31, 2010, 2009 and
2008
|
69
|
|
Consolidated
Statements of Cash Flows for the fiscal years ended March 31, 2010, 2009
and 2008
|
70
|
|
Notes
to Consolidated Financial Statements
|
72
|
|
Financial
Statement Schedules
|
94
|
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the
Board of Directors and Stockholders of
Synutra
International, Inc.
Rockville,
Maryland
We have
audited the accompanying consolidated balance sheets of Synutra International,
Inc. and subsidiaries (the “Company”) as of March 31, 2010 and 2009, and the
related consolidated statements of income, comprehensive income
(loss), equity, and cash flows for each of the three years in the period ended
March 31, 2010 and related financial statement schedules. These financial
statements and financial statement schedules are the responsibility of the
Company’s management. Our responsibility is to express an opinion on these
financial statements and financial statement schedules based on our
audits.
64
We
conducted our audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether the financial
statements are free of material misstatement. An audit includes examining, on a
test basis, evidence supporting the amounts and disclosures in the financial
statements. An audit also includes assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall
financial statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
In our
opinion, such consolidated financial statements present fairly, in all material
respects, the financial position of Synutra International, Inc. and subsidiaries
at March 31, 2010 and 2009, and the results of their operations and their cash
flows for each of the three years in the period ended March 31, 2010, in
conformity with accounting principles generally accepted in the United States of
America. Also, in our opinion, such financial statement schedules, when
considered in relation to the basic consolidated financial statements taken as a
whole, present fairly, in all material respect, the information set forth
therein.
As
discussed in Note 2 to the consolidated financial statements, the Company
changed its method of accounting for the adoption of new accounting standard
related to the presentation and disclosure requirements for noncontrolling
interests.
We have
also audited, in accordance with the standards of the Public Company Accounting
Oversight Board (United States), the Company’s internal control over financial
reporting as of March 31, 2010, based on the criteria established in Internal Control — Integrated
Framework issued by the Committee of Sponsoring Organizations of the
Treadway Commission and our report dated June 9, 2010 expressed an unqualified
opinion on the Company’s internal control over financial reporting.
/s/
Deloitte Touche Tohmatsu CPA Ltd.
Shanghai,
China
June 9,
2010
65
SYNUTRA
INTERNATIONAL, INC.
CONSOLIDATED
BALANCE SHEETS
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(in
thousands,
except
share par value)
|
||||||||
|
ASSETS
|
||||||||
|
Current
Assets:
|
||||||||
|
Cash
and cash equivalents
|
$ | 48,693 | $ | 37,736 | ||||
|
Restricted
cash
|
33,384 | 84,338 | ||||||
|
Accounts
receivable, net of allowance of $4,355 and $1,452,
respectively
|
26,013 | 23,826 | ||||||
|
Inventories
|
52,134 | 114,724 | ||||||
|
Due
from related parties
|
8,111 | 2,463 | ||||||
|
Income
tax receivable
|
523 | 1,476 | ||||||
|
Receivable
from assets disposal
|
5,879 | — | ||||||
|
Prepaid
expenses and other current assets
|
8,209 | 13,436 | ||||||
|
Deferred
tax assets
|
33,390 | 16,276 | ||||||
|
Total
current assets
|
216,336 | 294,275 | ||||||
|
Property,
plant and equipment, net
|
110,037 | 144,481 | ||||||
|
Land
use rights, net
|
5,996 | 6,374 | ||||||
|
Intangible
assets, net
|
3,394 | 3,136 | ||||||
|
Goodwill
|
1,437 | 1,435 | ||||||
|
Receivable
from assets disposal
|
4,404 | — | ||||||
|
Other
assets
|
3,575 | 4,406 | ||||||
|
Deferred
tax assets
|
4,178 | 18,464 | ||||||
|
TOTAL
ASSETS
|
$ | 349,357 | $ | 472,571 | ||||
|
LIABILITIES
AND EQUITY
|
||||||||
|
Current
Liabilities:
|
||||||||
|
Short-term
debt
|
$ | 98,069 | $ | 224,647 | ||||
|
Long-term
debt due within one year
|
61,194 | — | ||||||
|
Accounts
payable
|
49,947 | 112,968 | ||||||
|
Due
to related parties
|
2,670 | 5,172 | ||||||
|
Advances
from customers
|
9,375 | 5,448 | ||||||
|
Product
recall provision
|
— | 4,547 | ||||||
|
Other
current liabilities
|
22,674 | 21,925 | ||||||
|
Total
current liabilities
|
243,929 | 374,707 | ||||||
|
Long-term
debt
|
41,018 | 8,777 | ||||||
|
Deferred
revenue
|
4,688 | 4,681 | ||||||
|
Capital
lease obligations
|
5,372 | 5,254 | ||||||
|
Other
long-term liabilities
|
1,419 | 1,756 | ||||||
|
Total
liabilities
|
296,426 | 395,175 | ||||||
|
Equity:
|
||||||||
|
Synutra
International, Inc., shareholders’ equity
|
||||||||
|
Common
stock, $.0001 par value: 250,000 authorized; 54,001 and 54,001 issued and
outstanding at March 31, 2010 and 2009, respectively
|
5 | 5 | ||||||
|
Additional
paid-in capital
|
76,607 | 76,607 | ||||||
|
Accumulated
deficit
|
(48,289 | ) | (23,674 | ) | ||||
|
Accumulated
other comprehensive income
|
24,015 | 23,921 | ||||||
|
Total
Synutra common shareholders’ equity
|
52,338 | 76,859 | ||||||
|
Noncontrolling
interest
|
593 | 537 | ||||||
|
Total
equity
|
52,931 | 77,396 | ||||||
|
TOTAL
LIABILITIES AND EQUITY
|
$ | 349,357 | $ | 472,571 | ||||
The
accompanying notes are an integral part of the consolidated financial
statements.
66
SYNUTRA
INTERNATIONAL, INC.
CONSOLIDATED
STATEMENTS OF INCOME
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands except earnings per share data)
|
||||||||||||
|
Net
sales
|
$ | 291,886 | $ | 312,528 | $ | 362,090 | ||||||
|
Cost
of sales
|
208,476 | 259,086 | 175,568 | |||||||||
|
Gross
profit
|
83,410 | 53,442 | 186,522 | |||||||||
|
Selling
and distribution expenses
|
43,989 | 44,178 | 34,449 | |||||||||
|
Advertising
and promotion expenses
|
33,854 | 115,478 | 76,388 | |||||||||
|
General
and administrative expenses
|
24,509 | 25,455 | 16,013 | |||||||||
|
Impairment
loss from assets disposal
|
5,894 | — | — | |||||||||
|
Other
operating income, net
|
894 | 5,790 | 1,492 | |||||||||
|
Income
(loss) from operations
|
(23,942 | ) | (125,879 | ) | 61,164 | |||||||
|
Interest
expense
|
8,603 | 4,857 | 6,354 | |||||||||
|
Interest
income
|
1,850 | 341 | 1,801 | |||||||||
|
Other
expense, net
|
1,081 | 580 | 3,084 | |||||||||
|
Income
(loss) before income tax expense (benefit)
|
(31,776 | ) | (130,975 | ) | 53,527 | |||||||
|
Income
tax expense (benefit)
|
(6,904 | ) | (30,386 | ) | 7,855 | |||||||
|
Net
income (loss)
|
(24,872 | ) | (100,589 | ) | 45,672 | |||||||
|
Net
income (loss) attributable to the noncontrolling interest
|
(257 | ) | (40 | ) | 11 | |||||||
|
Net
income (loss) attributable to Synutra International, Inc. common
shareholders
|
$ | (24,615 | ) | $ | (100,549 | ) | $ | 45,661 | ||||
|
Earnings
(loss) per share – basic
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.86 | ||||
|
Earnings
(loss) per share – diluted
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.85 | ||||
|
Weighted
average common share outstanding – basic
|
54,001 | 54,001 | 53,170 | |||||||||
|
Weighted
average common share outstanding – diluted
|
54,001 | 54,001 | 53,476 | |||||||||
The
accompanying notes are an integral part of the consolidated financial
statement.
67
SYNUTRA
INTERNATIONAL, INC.
CONSOLIDATED
STATEMENTS OF COMPREHENSIVE INCOME (LOSS)
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands)
|
||||||||||||
|
Net
income (loss)
|
$ | (24,872 | ) | $ | (100,589 | ) | $ | 45,672 | ||||
|
Currency
translation adjustment
|
95 | 6,149 | 14,851 | |||||||||
|
Total
comprehensive income (loss)
|
(24,777 | ) | (94,440 | ) | 60,524 | |||||||
|
Less:
Comprehensive income (loss) attributable to noncontrolling
interest
|
(256 | ) | (40 | ) | 22 | |||||||
|
Comprehensive
income (loss) attributable to Synutra International, Inc. common
shareholders
|
$ | (24,521 | ) | $ | (94,400 | ) | $ | 60,501 | ||||
The
accompanying notes are an integral part of the unaudited condensed consolidated
financial statements.
68
SYNUTRA
INTERNATIONAL, INC.
CONSOLIDATED
STATEMENTS OF EQUITY
|
Common
Stock
|
||||||||||||||||||||||||||||
|
Shares
|
Amount
|
Additional
paid-in capital
|
Retained
earnings (accumulated deficit)
|
Accumulated
other comprehensive income
|
Noncontrolling
Interest
|
Total
equity
|
||||||||||||||||||||||
|
(in
thousands)
|
||||||||||||||||||||||||||||
|
Balance,
March 31, 2007
|
50,001 | $ | 5 | $ | 8,226 | $ | 31,538 | $ | 2,932 | $ | — | $ | 42,701 | |||||||||||||||
|
Issuance
of common stock, net of issuance costs of $206
|
4,000 | — | 65,794 | — | — | — | 65,794 | |||||||||||||||||||||
|
Issuance
of warrants
|
— | — | 2,587 | — | — | — | 2,587 | |||||||||||||||||||||
|
Adoption
of FIN 48
|
— | — | — | (375 | ) | — | — | (375 | ) | |||||||||||||||||||
|
Acquisition
of a subsidiary
|
— | — | — | 51 | — | 363 | 414 | |||||||||||||||||||||
|
Net
income
|
— | — | — | 45,661 | — | 11 | 45,672 | |||||||||||||||||||||
|
Currency
translation adjustment
|
— | — | — | — | 14,840 | 11 | 14,851 | |||||||||||||||||||||
|
Balance,
March 31, 2008
|
54,001 | 5 | 76,607 | 76,875 | 17,772 | 385 | 171,644 | |||||||||||||||||||||
|
Net
loss
|
— | — | — | (100,549 | ) | — | (40 | ) | (100,589 | ) | ||||||||||||||||||
|
Currency
translation adjustment
|
— | — | — | — | 6,149 | — | 6,149 | |||||||||||||||||||||
|
Other
|
— | — | — | — | — | 192 | 192 | |||||||||||||||||||||
|
Balance,
March 31, 2009
|
54,001 | 5 | 76,607 | (23,674 | ) | 23,921 | 537 | 77,396 | ||||||||||||||||||||
|
Net
loss
|
— | — | — | (24,615 | ) | — | (257 | ) | (24,872 | ) | ||||||||||||||||||
|
Currency
translation adjustment
|
— | — | — | — | 94 | 1 | 95 | |||||||||||||||||||||
|
Other
|
— | — | — | — | — | 312 | 312 | |||||||||||||||||||||
|
Balance,
March 31, 2010
|
54,001 | $ | 5 | $ | 76,607 | $ | (48,289 | ) | $ | 24,015 | $ | 593 | $ | 52,931 | ||||||||||||||
The
accompanying notes are an integral part of the consolidated financial
statements.
69
SYNUTRA
INTERNATIONAL, INC.
CONSOLIDATED
STATEMENTS OF CASH FLOWS
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands)
|
||||||||||||
|
Operating
activities:
|
||||||||||||
|
Net
income (loss)
|
$ | (24,872 | ) | $ | (100,589 | ) | $ | 45,672 | ||||
|
Adjustments
to reconcile net income (loss) to net cash provided by (used in) operating
activities:
|
||||||||||||
|
Amortization
of debt discount
|
— | — | 2,733 | |||||||||
|
Amortization
of debt issuance costs
|
321 | 321 | 539 | |||||||||
|
Depreciation
and amortization
|
10,098 | 7,649 | 4,646 | |||||||||
|
Bad
debt expense (reversal)
|
4,404 | 2,138 | (47 | ) | ||||||||
|
Loss
(gain) on disposal of property, plant and equipment
|
10 | (20 | ) | 249 | ||||||||
|
Impairment
loss
|
5,894 | 880 | 175 | |||||||||
|
Deferred
income tax
|
(2,805 | ) | (31,124 | ) | (2,695 | ) | ||||||
|
Other
compensation expense
|
312 | 192 | — | |||||||||
|
Changes
in assets and liabilities:
|
||||||||||||
|
Accounts
receivable
|
(4,538 | ) | (13,399 | ) | (4,149 | ) | ||||||
|
Inventories
|
61,336 | (50,940 | ) | (39,066 | ) | |||||||
|
Due
from related parties
|
(5,699 | ) | (3,557 | ) | 9,400 | |||||||
|
Prepaid
expenses and other current assets
|
1,150 | (2,691 | ) | (5,676 | ) | |||||||
|
Accounts
payable
|
(57,159 | ) | 76,975 | 6,831 | ||||||||
|
Due
to related parties
|
(2,432 | ) | 4,880 | 1,909 | ||||||||
|
Advances
from customers
|
3,917 | (4,248 | ) | 4,606 | ||||||||
|
Income
tax receivable
|
955 | (5,636 | ) | — | ||||||||
|
Income
tax payable
|
— | — | 5,037 | |||||||||
|
Deferred
revenue
|
(992 | ) | 986 | — | ||||||||
|
Product
recall provision
|
(4,547 | ) | 4,547 | — | ||||||||
|
Other
liabilities
|
2,823 | 4,148 | 8,065 | |||||||||
|
Net
cash provided by (used in) operating activities
|
(11,824 | ) | (109,488 | ) | 38,229 | |||||||
|
Investing
activities:
|
||||||||||||
|
Acquisition
of property, plant and equipment
|
(13,264 | ) | (44,827 | ) | (36,453 | ) | ||||||
|
Acquisition
of intangible assets
|
— | (2,474 | ) | — | ||||||||
|
Disposal
of investment in a subsidiary
|
— | — | (1,046 | ) | ||||||||
|
Change
in restricted cash
|
51,059 | (73,934 | ) | 4,213 | ||||||||
|
Payment
for business acquisitions
|
(1,468 | ) | (6,052 | ) | 190 | |||||||
|
Proceeds
from assets disposal
|
20,202 | — | — | |||||||||
|
Advance
to related companies
|
— | — | (3,324 | ) | ||||||||
|
Repayment
of advance to related companies
|
— | — | 3,324 | |||||||||
|
Net
cash provided by (used in) investing activities
|
56,529 | (127,287 | ) | (33,096 | ) | |||||||
|
Financing
activities:
|
||||||||||||
|
Proceeds
from short-term debt
|
337,659 | 245,710 | 60,179 | |||||||||
|
Repayment
of short-term debt
|
(403,704 | ) | (77,314 | ) | (96,242 | ) | ||||||
|
Proceeds
from long-term debt
|
32,241 | 8,777 | 35,000 | |||||||||
|
Repayment
of long-term debt
|
— | (1,923 | ) | — | ||||||||
|
Payment
on capital lease obligations
|
— | (379 | ) | — | ||||||||
|
Proceeds
from issuance of common stock
|
— | — | 66,000 | |||||||||
|
Issuance
costs for common stock issuance
|
— | — |
(206
|
) | ||||||||
|
Net
cash provided by (used in) financing activities
|
(33,804 | ) | 174,871 | 64,731 | ||||||||
70
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands)
|
||||||||||||
|
Effect
of exchange rate changes on cash and cash equivalents
|
56 | 2,215 | 6,725 | |||||||||
|
Net
change in cash and cash equivalents
|
10,957 | (59,689 | ) | 76,589 | ||||||||
|
Cash
and cash equivalents, beginning of year
|
37,736 | 97,425 | 20,836 | |||||||||
|
Cash
and cash equivalents, end of year
|
$ | 48,693 | $ | 37,736 | $ | 97,425 | ||||||
|
Supplemental
cash flow information:
|
||||||||||||
|
Interest
paid
|
$ | 10,742 | $ | 5,718 | $ | 3,769 | ||||||
|
Income
tax paid
|
$ | 762 | $ | 6,443 | $ | 6,486 | ||||||
|
Non-cash
investing and financing activities:
|
||||||||||||
|
Purchase
of property, plant and equipment by accounts payable
|
$ | — | $ | 7,501 | $ | 4,149 | ||||||
|
Issuance
of warrant in relation to the long-term debt
|
$ | — | $ | — | $ | 2,733 | ||||||
|
Assets
acquired under capital lease
|
$ | — | $ | 5,336 | $ | — | ||||||
|
Acquisition
of subsidiary by accounts payable
|
$ | — | $ | 1,521 | $ | — | ||||||
|
Disposal
of property by due from related parties
|
$ | — | $ | 1,726 | $ | — | ||||||
|
Assets
disposal by other receivable
|
$ | 5,879 | $ | — | $ | — | ||||||
The
accompanying notes are an integral part of the consolidated financial
statements.
71
SYNUTRA
INTERNATIONAL, INC.
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
1. ORGANIZATION
AND PRINCIPAL ACTIVITIES
Directly
or through its wholly owned subsidiary, Synutra Inc., an Illinois corporation
(“Synutra Illinois”), Synutra International, Inc. (collectively with its
subsidiaries, the “Company” or “Synutra”) owns all or majority of the equity
interests of the entities in the People’s Republic of China (“China” or “PRC”)
that are principally engaged in the production, marketing and distribution of
dairy based nutritional products under the Company’s own brands in China. The
Company is a leader in sales of infant formula products in China.
The
Company produces, markets and sells nutritional products under the “Shengyuan”
or “Synutra” name, together with other complementary brands. The Company focuses
on selling premium infant formula products, which are supplemented by more
affordable infant formula products targeting the mass market as well as other
nutritional products, such as adult powdered formula and prepared baby food, and
certain nutritional ingredients and supplements.
2. SUMMARY
OF SIGNIFICANT ACCOUNTING POLICIES
A. Basis
of presentation
The
consolidated financial statements of the Company have been prepared in
accordance with accounting principles generally accepted in the United States of
America (“US GAAP”). Although the Company experienced a net loss and negative
cash flows from operations in the year ended March 31, 2010 and had a working
capital deficit as of that date, the Company has full and unrestricted access to
unused committed and available bank standby credit facilities at year end and is
in full compliance with renegotiated debt covenants. In addition, part of the
Company’s debt has been restructured subsequent to the balance sheet date and
improved operating profits are included within net profit recorded in the fourth
quarter of fiscal year 2010 and positive operating cash flows were generated in
the second, third and fourth quarter of fiscal year 2010. The Company will be
able to realize its assets and satisfy its liabilities in the normal course of
business. As a result, the accompanying consolidated financial statements have
been prepared assuming the Company will continue as a going
concern.
B. Basis
of consolidation
The
consolidated financial statements include the financial statements of Synutra
International, Inc. and its subsidiaries, its variable interest entity, Beijing
Shengyuan Huimin Technology Service Co., Ltd., and its subsidiaries. All
significant inter-company accounts and transactions have been eliminated in
consolidation. The variable interest entity and its subsidiaries have not
commenced their planned operations as of March 31, 2010.
C. Use
of estimates
The
preparation of financial statements in conformity with generally accepted
accounting principles in the United States of America requires management to
make estimates and assumptions that affect the reported amounts of assets and
liabilities and disclosure of contingent assets and liabilities at the date of
the financial statements, and the reported amounts of revenue and expenses
during the reporting period. Actual results may differ from those
estimates. Significant accounting estimates reflected in the
Company’s consolidated financial statements include allowance for doubtful
accounts, inventory valuation, assumptions related to the valuation of assets
held for sale, accrued replacement provision, the useful lives of and impairment
for property and equipment and intangible assets, valuation allowance of
deferred tax assets, and impairment of goodwill.
D. Cash
and cash equivalents
Cash and
cash equivalents consist of cash on hand and highly liquid investments which are
unrestricted as to withdrawal or use, and which have original maturities of
three months or less when purchased.
72
E. Restricted
cash
Restricted
cash are bank demand deposits used as security against letter of credits and
short-term borrowings. This is used by the Company as a short-term instrument to
reduce financing cost.
F. Account
receivables
Trade
receivables are recognized and carried at the original invoice amount less
allowance for any uncollectible amounts. An estimate for doubtful accounts is
made when collection of the full amount is no longer probable. The Company
performs risk assessment for each customer, and provides specific allowance for
those deemed to have high risk of uncollectibility. The Company also records
provision for other customers without specific risks by reviewing the aging of
the receivables. Bad debts are written off as incurred.
G. Inventories
Inventories
are stated at the lower of cost or market. Cost is calculated on the
moving-average basis and includes all costs to acquire and other costs incurred
in bringing the inventories to their present location and condition. The Company
evaluates market of its inventories on a regular basis and records a provision
for loss to reduce the computed weighted-average cost if it exceeds market.
Potential losses from obsolete and slow-moving inventories are recorded when
identified. Market is current replacement cost, which does not exceed the net
realizable value, that is, estimated selling price in the ordinary course of
business less reasonable predictable cost of completion and disposal, and is not
less than net realizable value reduced by an allowance for normal profit
margin.
H. Property,
plant and equipment, net
Property,
plant and equipment are carried at cost. The cost of repairs and maintenance is
expensed as incurred; major replacements and improvements are
capitalized.
When
assets are retired or disposed of, the cost and accumulated depreciation are
removed from the accounts, and any resulting gains or losses are included in
income in the year of disposition.
Depreciation
is calculated on a straight-line basis over the estimated useful lives of the
assets or, when applicable, the life of the lease, whichever is shorter. The
useful lives for property, plant and equipment are as follows:
|
Buildings
|
20
- 40 years
|
|
Plant
and machinery
|
5 -
10 years
|
|
Office
equipment and furnishings
|
3 -
5 years
|
|
Motor
vehicles
|
5
years
|
|
Others
|
5
years
|
I. Leases
Leases
are classified as capital or operating leases. A lease that transfers to the
lessee substantially all the benefits and risks incidental to ownership is
classified as a capital lease. At inception, a capital lease is recorded at
present value of minimum lease payments or the fair value of the asset,
whichever is less. Assets under capital leases are amortized on a basis
consistent with that of similar fixed assets or the lease term, whichever is
less. Operating lease costs are recognized on a straight-line basis over the
lease term.
J. Construction
in progress
Construction
in progress represents direct costs of construction or acquisition and design
fees incurred. Capitalization of these costs ceases and the construction in
progress is transferred to plant and equipment when substantially all the
activities necessary to prepare the assets for their intended use are completed.
No depreciation is provided until it is completed and ready for intended
use.
The
capitalized interest recognized for the fiscal years ended March 31, 2010, 2009
and 2008 was $346,000, $1.4 million and $848,000 respectively, associated with
construction in progress.
73
K. Impairment
of long-lived assets
The
Company evaluates its long-lived assets for impairment whenever events or
changes in circumstances indicate that the carrying amount of an asset may not
be recoverable. When these events occur, the Company measures impairment by
comparing the carrying amount of the assets to future undiscounted net cash
flows expected to result from the use of the assets and their eventual
disposition. If the sum of the expected undiscounted cash flow is less than the
carrying amount of the assets, the Company would recognize an impairment loss
based on the fair value of the assets.
L. Impairment
of goodwill and indefinite lived intangible assets
Goodwill
is tested for impairment using a two-step process on an annual basis or when
current facts or circumstances indicate that a potential impairment may exist.
The first step compares the fair value of each reporting unit to its carrying
amount, including goodwill and indefinite lived intangible assets. If the fair
value of each reporting unit exceeds its carrying amount, goodwill is not
considered to be impaired and the second step will not be required. If the
carrying amount of a reporting unit exceeds its fair value, the second step
compares the implied fair value of goodwill to the carrying value of a reporting
unit’s goodwill. The implied fair value of goodwill is determined in a manner
similar to accounting for a business combination with the allocation of the
assessed fair value determined in the first step to the assets and liabilities
of the reporting unit. The excess of the fair value of the reporting unit over
the amounts assigned to the assets and liabilities is the implied fair value of
goodwill. This allocation process is only performed for purposes of evaluating
goodwill impairment and does not result in an entry to adjust the value of any
assets or liabilities. An impairment loss is recognized for any excess in the
carrying value of goodwill over the implied fair value of goodwill.
Intangible
assets with an indefinite life are tested for impairment annually or more
frequently if events or changes in circumstances indicate that the asset might
be impaired. The impairment test consists of a comparison of the fair value of
the intangible asset to its carrying amount. If the carrying amount exceeds the
fair value, an impairment loss is recognized equal in amount to that
excess.
Management
performs its annual impairment test for goodwill and intangible assets with
indefinite life on March 31.
M.
Income taxes
The
Company accounts for income taxes in accordance with ASC 740, Income Taxes
(previously SFAS No. 109, “Accounting for Income Taxes,”) which requires
recognition of deferred tax liabilities and assets for the expected future tax
consequences of events that have been included in the financial statements or
tax returns. Under this method, deferred tax liabilities and assets are
determined based on the temporary difference between the financial statement and
tax basis of assets and liabilities using presently enacted tax rates in effect.
Valuation allowances are established when necessary to reduce deferred tax
assets to the amounts expected to be realized.
N. Foreign
currency translation
The
functional currency and reporting currency of Synutra International, Inc. and
Synutra Illinois are United States Dollar (“US Dollar”). Monetary assets and
liabilities denominated in currencies other than the US dollar are translated
into US dollar at the rates of exchange ruling at the balance sheet date.
Transactions in currencies other than the US dollar during the year are
converted into the US dollar at the applicable rates of exchange prevailing on
the day transactions occurred. Transaction gains and losses are recognized in
the statements of income.
The
financial records of the Company’s PRC subsidiaries are maintained in Renminbi
(“RMB”) which are their functional currency. Assets and liabilities are
translated at the exchange rates at the balance sheet date, equity accounts are
translated at historical exchange rates and revenues, expenses, gains and losses
are translated using the average rate for the year. Translation adjustments are
reported as cumulative translation adjustments and are shown as a separate
component of accumulated other comprehensive income.
74
O. Revenue
recognition
The
Company recognizes revenue when title and risk and rewards for the products are
transferred to the customer, price is fixed or determinable, and collectibility
is reasonably assured. At the time of sale, the Company also records estimates
for a variety of sales deductions, including value added taxes, rebates,
discounts and incentives, trade promotions and product returns. Sales deductions
are reported as a reduction of revenue. Most of the Company’s nutritional
product sales are made through distributors. Under the distributor arrangement,
evidenced by purchase order together with advance payment, sales revenue is
realized and earned upon acceptance of delivery of products by the distributors.
The Company applies this revenue recognition policy uniformly to all nutritional
products, including all dairy-based pediatric and adult nutritional
products.
A small
fraction of the Company’s nutritional product sales are through supermarket
retailers directly. The Company’s revenue arrangement with some of these
retailers includes right of return clause. The Company’s price to the
supermarkets is fixed. The supermarkets’ obligation to the Company would not be
changed in the event of theft or physical destruction or damage of the product.
The Company recognizes revenue when the supermarkets have paid the Company, or
the supermarkets are obligated to pay the Company and the obligation is not
contingent on resale of the product. The amount of future returns are estimated
and recognized in the current period.
The
Company’s gross sales are subject to various deductions, primarily comprised of
rebates and discounts to distributors and retailers. These deductions represent
estimates of the related obligations, requiring the use of judgment when
estimating the impact of these sales deductions on gross sales for a reporting
period. The Company reports these adjustments as a reduction of gross sales to
arrive at net sales.
|
|
·
|
The
Company offers rebates to distributors and supermarket retailers to
sustain and increase the Company’s product market share. These rebate
programs provide that distributors and supermarket retailers receive a
rebate after attaining certain performance parameters relating to product
purchases, formulary status and/or pre-established market share milestones
relative to competitors. Since rebates are contractually agreed upon, the
Company estimate rebates based on the specific terms in each agreement,
historical experience, anticipated reimbursement channel mix and product
growth rates. The Company considers the sales performance of products
subject to rebates and other contract discounts and adjusts the provision
periodically to reflect actual experience. Actual amount may differ if
actual performance vary from estimates. The Company records rebates as a
reduction of revenues in the year in which these programs are
offered.
|
|
|
·
|
The
Company offers product discounts to compensate distributors for the
promotional activities which were previously performed by the Company.
Prior to August 2009, promotional activities were generally jointly
performed by distributors and the Company, and each party was responsible
for the pre-determined portion of expenses respectively. Effective August
2009, the Company establishes a monthly budget for promotional expenses.
Distributors are responsible for organizing the promotional activities,
and providing us with documentation of their expenses, which would be
recorded as product discounts to offset receivable from distributors in
the applicable month.
|
|
|
·
|
The
Company records a provision for estimated sales returns due to package
damage and termination of distributorships. Due to the melamine
contamination incident, the turnover rate of products in the distribution
channel to the end customers was significantly increased. To avoid the
potential loss the distributors might suffer, the Company offered certain
product exchange for those products approaching the expiration dates. To
respond to the exchange program, since October 2009, the Company recorded
estimated replacement costs for future potential product exchange program.
The sales return amount represents management’s best estimates based on
the available information at the time of
estimate.
|
|
|
·
|
For
product sales and promotions at supermarkets and shopping malls, certain
expenses in relation to shelf display, end-cap placement, bar-coding,
banner advertising, etc. are paid to supermarkets and shopping mall
operators. These expenses are deducted from revenues in accordance with
ASC 605, Revenue Recognition (previously EITF 01-9 “Accounting for
Consideration Given by a Vendor to a Customer (Including a Reseller of the
Vendor’s Product)”).
|
75
P. Shipping
and handling costs
Shipping
and handling costs are expensed as incurred and outbound freight is not billed
to customers. Shipping and handling costs are included in selling and
distribution expenses. The expenses were $4.7 million, $7.4 million and $6.7
million for the fiscal years ended March 31, 2010, 2009 and 2008,
respectively.
Q. Advertising
and promotion expenses
Advertising
and promotion expenses are expensed as incurred.
R. Government
subsidies
Government
grants for revenue and/or expenses should be recognized in income when the
related revenue and/or expense are recorded. Government grants related to
property, plant, and equipment should be netted against the depreciation expense
of the related assets over the useful lives of these assets. Government
subsidies relating to specific borrowings are recorded as an offset to the
interest expense over the term of these borrowings. Unrestricted government
subsidies from local governmental agencies allowing the Company full discretion
in the fund utilization were $894,000, $5.8 million and $1.5 million for the
fiscal years ended March 31, 2010, 2009 and 2008, respectively, which was
recorded in other operating income in the consolidated statements of income. In
addition, the Company received $4.6 million (RMB 32.0 million) of government
subsidy in fiscal year ended March 31, 2007 which was directly related to the
manufacturing facility construction. Such subsidy was recorded in deferred
revenue and will be amortized to offset the depreciation expenses associated
with the underlining property.
S. Employee’s
benefits
Mandatory
contributions are made to the Government’s health, retirement benefit and
unemployment schemes at the statutory rates in force during the period, based on
gross salary payments. The cost of these payments is charged to the statements
of income in the same period as the related salary cost.
T. Comprehensive
income (loss)
The
Company has adopted ASC 220, Comprehensive Income (previously SFAS No. 130,
“Reporting Comprehensive Income”). ASC 220 establishes standards for the
reporting and display of comprehensive income (loss), its components and
accumulated balances in a full set of general-purpose financial statements. ASC
220 defines comprehensive income (loss) to include all changes in equity except
those resulting from investments by owners and distributions to owners,
including adjustments to accumulated foreign currency translation.
U. Concentration
of credit risk
Financial
instruments that potentially subject the Company to significant concentrations
of credit risk consist primarily of trade accounts receivable. The Company
performs ongoing credit evaluations with respect to the financial condition of
its creditors, but does not require collateral. In order to determine the value
of the Company’s accounts receivable, the Company records a provision for
doubtful accounts to cover probable credit losses. Management reviews and
adjusts this allowance periodically based on historical experience and its
evaluation of the collectibility of outstanding accounts
receivable.
V. Noncontrolling
interest
The
noncontrolling interest represents the Company’s equity interest in Baoquanling
and Meitek. In accordance with ASC No. 810-10-65, Consolidation (previously
SFAS No. 160, “Noncontrolling
Interests in Consolidated Financial Statements”), the Company
reclassified the ownership interest in the consolidated entity to noncontrolling
interest and reported as equity in the consolidated financial statements, it
also reported the consolidated net income at amounts that include the amounts
attributable to both the parent and the noncontrolling interest on the face of
the consolidated statements of income and consolidated statements of
comprehensive income (loss).
76
W. Recently
issued accounting pronouncements
In
January 2010, the FASB issued ASU 2010-06, Improving Disclosures about Fair
Value Measurements. The ASU amends ASC 820 (formerly Statement No. 157, Fair
Value Measurements) to add new requirements for disclosures about transfers into
and out of Levels 1 and 2 and separate disclosures about purchases, sales,
issuances, and settlements relating to Level 3 measurement on a gross basis
rather than on a net basis as currently required. ASU 2010-06 also
clarifies existing fair value disclosures about the level of disaggregation and
the inputs and valuation techniques used to measure fair value. ASU
2010-06 is effective for annual and interim periods beginning after December 15,
2009, except for the requirement to provide the level 3 activity of purchases,
sales, issuances, and settlements on a gross basis, which will be effective for
annual and interim periods beginning December 15, 2010. Early
application is permitted and in the period of initial adoption, entities are not
required to provide the amended disclosures for any previous periods presented
for comparative purposes. The Company is currently evaluating the
impact of the adoption on its consolidated financial statements.
3. PRODUCT
RECALL
On
September 16, 2008, the Company announced a compulsory recall on certain lots of
U-Smart products and a voluntary recall of other products that were contaminated
or suspected to be contaminated by melamine, a substance not approved for use in
food and linked to illnesses among infants and children in China. The cost of
this action during the year ended March 31, 2009 was $101.5 million, including
the cost of product replacement of $48.1 million in cost of sales, the
write-down and write-off of affected inventory of $48.5 million in cost of
sales, the net amount of $2.3 million to a compensation fund set up by China
Dairy Industry Association to settle existing and potential claims arising in
China from families of infants affected by melamine contamination in general and
administrative expenses, and freight charges of $2.6 million in selling and
distribution expenses, of which $4.5 million was recorded as a product recall
provision in the consolidated balance sheet as of March 31, 2009. In the fiscal
year ended March 31, 2010, the Company reversed recall expenses of $0.9 million,
mostly being overestimated product replacement costs. The Company believes that
the product recall has been substantially completed as of March 31, 2010 and
estimates that there should be no further material product recall costs to
incur. The following is a summary of the liabilities related to the product
recall that were recorded during the fiscal year ended March 31, 2010 and
2009:
|
(In
thousands)
|
||||
|
Balance
at April 1, 2008
|
$ | — | ||
|
Product
recall expenses
|
53,038 | |||
|
Replacement
of products
|
(44,002 | ) | ||
|
Payment
of cash, net
|
(4,395 | ) | ||
|
Foreign
currency translation
|
(94 | ) | ||
|
Balance
at March 31, 2009
|
4,547 | |||
|
Product
recall expenses (reversal)
|
(886 | ) | ||
|
Replacement
of products
|
(3,421 | ) | ||
|
Payment
of cash, net
|
(243 | ) | ||
|
Foreign
currency translation
|
3 | |||
|
Balance
at March 31, 2010
|
$ | — | ||
4. IMPAIRMENT
LOSS AND ASSETS DISPOSAL
After the
melamine contamination incident, the Company began to use imported milk powder
in the powdered formula products and decreased the consumption of domestically
produced milk powder, hence the Company decided to sell certain production
facilities and dairy farms. On September 9, 2009, two subsidiaries of Synutra
International, Inc., Heilongjiang Baoquanling Shengyuan Dairy Co., Ltd.
(“Baoquanling”) and Heilongjiang Baoquanling Shengyuan Dairy Cow Breeding Co.,
Ltd. (“Cow Breeding”) entered into an asset purchase agreement (the “Agreement”)
with Heilongjiang Wondersun Dairy Co., Ltd. (“Wondersun”), to sell three dairy
farms and two milk processing factories in China for approximately $28.9
million, plus reimbursement for certain qualified construction in progress
assets acquired subsequent to the valuation date, of which $4.4 million will be
received in the fiscal year ending March 31, 2012. The assets were classified as
held for sale assets on September 9, 2009 and
77
were
written down to the fair value less estimated cost to sell. No depreciation or
amortization was recorded after September 9, 2009. The estimated impairment loss
on the assets group for the fiscal year ended March 31, 2010 was $5.9 million.
The previous operating results of these assets was not reported as discontinued
operation, as the assets group is part of a larger cash-flow-generating group,
and on its own is not a component of the entity, and therefore the conditions in
paragraph 205-20-45-1, Reporting Discontinued
Operations, for reporting in discontinued operations the operations
(losses) of the assets group would not be met. As of March 31, 2010, the Company
has transferred majority of these assets to Wondersun.
5. FAIR
VALUE MEASUREMENTS
The carrying value of financial
instruments including cash, receivables, accounts payable, accrued expenses,
short-term debt and long-term debt due within one year, approximates their fair
value at March 31, 2010 and 2009 due to the relatively short-term nature of
these instruments. The carrying value of long-term debt approximates its fair
value as their interest rates are at the same level of the current market yield
for comparable loans. The fair value of the long-term receivable from the assets
disposal was approximately $4.4 million as of March 31, 2010, and was estimated
using the discounted cash flow method with prevailing market interest rate on
the valuation date as the discount rate.
On April
1, 2008, the Company adopted ASC No.820, Fair Value Measurements and
Disclosures, which was not delayed by ASC No. 820-10-65-1, Transition related to FASB Staff
Position Financial Accounting Standards (“FAS”) 157-2, Effective Date of FASB
statement No. 157 (pre-codification reference as FASB Staff Position FAS
157-2). ASC 820-10-65-1 delays the effective date of ASC No.820 as it applies to
non-financial assets and liabilities that are not required to be measured at
fair value on a recurring (at least annual) basis. As a result of the delay, ASC
No.820 was applied to the Company’s non-financial assets and liabilities
effective on April 1, 2009. ASC No.820 defines fair value as the price that
would be received to sell an asset or paid to transfer a liability in an orderly
transaction between market participants at the measurement date (also referred
to as an exit price). ASC No.820 also establishes a three-level fair value
hierarchy for classifying financial instruments that is based on whether the
inputs to the valuation techniques used to measure fair value are observable or
unobservable. The three levels of the ASC No.820 fair value hierarchy are
described below:
|
Level
1:
|
Quoted
prices (unadjusted) in active markets for identical assets or
liabilities.
|
|
Level
2:
|
Observable
market-based inputs, other than quoted prices in active markets for
identical assets or liabilities.
|
|
Level
3:
|
Unobservable
inputs.
|
The
following table sets forth by level, within the fair value hierarchy, the
Company’s assets and liabilities accounted for at fair value on a nonrecurring
basis as of March 31, 2010. The Company did not have any items recorded at fair
value on a recurring basis subsequent to initial recognition as of March 31,
2010 and 2009.
|
Description
|
For
the fiscal year ended March 31, 2010
|
Quoted
Prices in Active Markets for Identical Assets
(Level
1)
|
Significant
Other
Observable
Inputs
(Level
2)
|
Significant
Unobservable
Inputs
(Level
3)
|
Total
Loss
|
|||||||||||||||
|
(In
thousands)
|
||||||||||||||||||||
|
Assets
held for sale
|
$ | 30,211 | — | $ | 30,211 | — | $ | 5,894 | ||||||||||||
In
accordance with the provisions of ASC No.360, Property, Plant and
Equipment, assets held for sale with a carrying amount of $35.1 million
were written down to their fair value of $30.2 million, less cost to sell of
$1.0 million, resulting in a loss of $5.9 million, which was included in
impairment loss for the fiscal year ended March 31, 2010. The fair value of the
assets held for sale was determined by executed transaction price with a third
party. The Company has categorized this as a significant other
observable input (level 2).
78
6. INVENTORIES
The
Company’s inventories at March 31, 2010 and 2009 are summarized as
follows:
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Raw
materials
|
$ | 35,667 | $ | 96,361 | ||||
|
Work-in-progress
|
10,893 | 11,739 | ||||||
|
Finished
goods
|
5,574 | 6,624 | ||||||
|
Total
Inventories
|
$ | 52,134 | $ | 114,724 | ||||
The value
of goods-in-transit included in raw materials was $9.6 million and $43.5 million
as of March 31, 2010 and 2009, respectively, which mainly represented the
overseas purchase of milk powder and whey protein.
7. DUE
FROM (TO) RELATED PARTIES AND RELATED PARTY TRANSACTIONS
Parties
are considered to be related if one party has the ability, directly or
indirectly, to control the other party or exercise significant influence over
the other party in making financial and operational decisions. Parties are also
considered to be related if they are subject to common control or common
significant influence. Related parties may be individuals or corporate
entities.
The
following entities are considered to be related parties to the Company because
they are affiliates of the Company under the common control of the Company’s
major shareholder. These related parties act only as the Company’s suppliers or
distributors and there are no other relationships wherein the Company has the
ability to exercise significant influence over the operating and financial
policies of these parties. The Company is not obligated to provide any type of
financial support to these related parties. The related parties were excluded
from consolidation due to equity structure and other business reasons. The
economic substance of each entity apart from its relationship with the Company
is discussed in the table below:
|
Related
Party
|
Economic
Substance
|
|
| Sheng Zhi Da Dairy Group Corporation | ||
|
(“Sheng
Zhi
Da”)
|
Engages
in buying and selling packaging materials, vitamin and mineral pre-mixes,
and other food ingredients.
|
|
|
Beijing
Kelqin Dairy Co. Ltd. (“Kelqin”)
|
Produces
and distributes retail-packaged yogurt products in
Beijing.
|
|
| St. Angel (Beijing) Business Service Co., Ltd. | ||
|
(“St.
Angel (Beijing) Business
Service”)
|
Publishes
catalogues and engages in advertising and direct marketing of consumer
products featured in catalogues.
|
|
| Beijing St. Angel Cultural Communication Co., Ltd. | ||
|
(“St.
Angel Cultural
Communication”)
|
Engages
in television designing and programming.
|
|
|
Beijing
Honnete Dairy Co., Ltd. (“Honnete”)
|
Engages
in importing and distributing whey protein products to commercial
customers.
|
|
| Beijing Luding Xueyuan Trading Co., Ltd. | ||
|
(“Luding
Xueyuan”)
|
Operates
retail shelf spaces in supermarkets in Beijing. Luding Xueyuan became a
third party since January 2008.
|
|
| Qingdao Lvyin Waste Disposal | ||
|
Investment
Management Co., Ltd. (“Luyin”)
|
Engages
in waste disposal and sewage treatment
activities.
|
79
A. Classification
of related party balances by name
a. Due
from related parties
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Sheng
Zhi Da Dairy Group Corporation
|
$ | 1,728 | $ | 1,726 | ||||
|
Beijing
Honnete Dairy Co., Ltd.
|
5,476 | 737 | ||||||
|
St.
Angel (Beijing) Business Service Co. Ltd..
|
907 | — | ||||||
|
Total
due from related parties
|
$ | 8,111 | $ | 2,463 | ||||
In June
2008, the Company sold two commercial buildings to Sheng Zhi Da Dairy Group
Corporation, an entity 100% controlled by the Company’s CEO, at the carrying
value of $1.7 million. The amount due from Sheng Zhi Da Dairy Group Corporation
as of March 31, 2010 and 2009 represents the receivable for the sales of the two
buildings.
b. Due
to related parties
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Sheng
Zhi Da Dairy Group Corporation
|
$ | 2,098 | $ | 2,113 | ||||
|
Beijing
Honnete Dairy Co., Ltd.
|
568 | 3,057 | ||||||
|
St.
Angel (Beijing) Business Service Co., Ltd.
|
— | 2 | ||||||
|
Beijing
St. Angel Cultural Communication Co., Ltd.
|
4 | — | ||||||
|
Total
due to related parties
|
$ | 2,670 | $ | 5,172 | ||||
The
Company had certain related party borrowings which were recorded in short-term
debt. See Note 12. Except for the related party borrowings, the amount due to
and due from related parties were unsecured and interest free.
B. Sales
to related parties
In the
fiscal years ended March 31, 2010, 2009 and 2008, the Company’s sales to the
related parties included whey protein to Honnete, milk fat and non-fat dry milk
to Kelqin, industrial milk powder to Luding Xueyuan, and powdered formula
products to St. Angel.
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
Beijing
Honnete Dairy Co., Ltd.
|
$ | 8,830 | $ | 2,255 | $ | — | ||||||
|
Beijing
Kelqin Dairy Co., Ltd.
|
— | — | 143 | |||||||||
|
Beijing
Luding Xueyuan Trading Co., Ltd.
|
— | — | 1,497 | |||||||||
|
St.
Angel (Beijing) Business Service Co., Ltd.
|
824 | — | — | |||||||||
|
Total
|
$ | 9,654 | $ | 2,255 | $ | 1,640 | ||||||
C. Purchases
from related parties
In the
fiscal year ended March 31, 2010, 2009 and 2008, the Company’s purchases from
related parties included whey protein powders from Kelqin and Honnete, and
catalogues, brochures, and marketing materials from St. Angel.
80
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
Beijing
Honnete Dairy Co., Ltd.
|
$ | 98 | $ | 10,376 | $ | 17,862 | ||||||
|
Beijing
Kelqin Dairy Co., Ltd.
|
— | 154 | 3,286 | |||||||||
|
Beijing
St. Angel Cultural Communication Co. Ltd..
|
470 | 1,639 | 988 | |||||||||
|
Total
|
$ | 568 | $ | 12,169 | $ | 22,136 | ||||||
8. PREPAID
EXPENSES AND OTHER CURRENT ASSETS
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Advance
to suppliers
|
$ | 301 | $ | 584 | ||||
|
Prepaid
advertising expense
|
1,232 | 535 | ||||||
|
Other
tax receivable
|
2,287 | 3,148 | ||||||
|
Subsidy
receivable
|
1,465 | 5,060 | ||||||
|
Others
|
2,924 | 4,109 | ||||||
|
Total
|
$ | 8,209 | $ | 13,436 | ||||
9. PROPERTY,
PLANT AND EQUIPMENT, NET
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Property,
plant and equipment, cost:
|
||||||||
|
Buildings
|
$ | 49,351 | $ | 51,693 | ||||
|
Plant
and machinery
|
63,547 | 65,020 | ||||||
|
Office
equipment and furnishings
|
3,314 | 3,076 | ||||||
|
Motor
vehicles
|
2,590 | 2,963 | ||||||
|
Others
|
392 | 543 | ||||||
|
Total
cost
|
$ | 119,194 | $ | 123,295 | ||||
|
Less:
Accumulated depreciation:
|
||||||||
|
Buildings
|
6,353 | 4,472 | ||||||
|
Plant
and machinery
|
17,065 | 11,804 | ||||||
|
Office
equipment and furnishings
|
1,634 | 1,183 | ||||||
|
Motor
vehicles
|
1,099 | 885 | ||||||
|
Others
|
329 | 273 | ||||||
|
Total
accumulated depreciation
|
26,480 | 18,617 | ||||||
|
Construction
in progress
|
17,323 | 39,803 | ||||||
|
Property,
plant and equipment, net
|
$ | 110,037 | $ | 144,481 | ||||
Construction
in progress primarily represents the construction of manufacturing facilities
and related equipment, and administrative buildings.
The
Company recorded depreciation expense of $9.9 million, $7.5 million and $4.6
million for the fiscal years ended March 31, 2010, 2009 and 2008,
respectively.
10.
LAND USE RIGHTS, NET
There is
no private land ownership in China. Companies or individuals are authorized to
possess and use the land only through land use rights granted by the PRC
government. Land use rights are amortized using the straight-line method over
the lease term of 20 to 50 years.
81
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Land
use rights, cost
|
$ | 6,511 | $ | 6,757 | ||||
|
Less:
Accumulated amortization
|
515 | 383 | ||||||
|
Land
use rights, net
|
$ | 5,996 | $ | 6,374 | ||||
The
Company recorded amortization expense of $141,000, $128,000 and $80,000 for the
fiscal years ended March 31, 2010, 2009 and 2008, respectively.
11.
INTANGIBLE ASSETS, NET
|
March
31, 2010
|
||||||||||||||||
|
Gross
Carrying Amount
|
Accumulated
Amortization
|
Write-off
|
Net
Carrying Amount
|
|||||||||||||
|
(In
thousands)
|
||||||||||||||||
|
Know-how
(5-year useful life)
|
$ | 399 | $ | 120 | $ | — | $ | 279 | ||||||||
|
Registered
trademark (indefinite life)
|
3,115 | — | — | 3,115 | ||||||||||||
| $ | 3,514 | $ | 120 | $ | — | $ | 3,394 | |||||||||
|
March
31, 2009
|
||||||||||||||||
|
Gross
Carrying Amount
|
Accumulated
Amortization
|
Write-off
|
Net
Carrying Amount
|
|||||||||||||
|
(In
thousands)
|
||||||||||||||||
|
Know-how
(5-year useful life)
|
$ | 398 | $ | 40 | $ | — | $ | 358 | ||||||||
|
Customer
relationship
|
880 | — | 880 | — | ||||||||||||
|
Registered
trademark (indefinite life)
|
2,778 | — | — | 2,778 | ||||||||||||
| $ | 4,056 | $ | 40 | $ | 880 | $ | 3,136 | |||||||||
The
intangible assets acquired in the fiscal year ended March 31, 2009 included the
know-how, customer relationship and registered trademarks arising from the
acquisition of Huilian’s prepared baby food business, and the registered
trademarks of Helanruniu, or Holsteina, that are being used for the Company’s
powdered adult formula products.
Amortization
expense was $80,000 and $40,000 for the fiscal year ended March 31, 2010 and
2009, respectively.
12.
DEBT AND WARRANTS
On April
19, 2007, the Company entered into a bridge loan agreement with ABN AMRO Bank
N.V., Hong Kong branch (now known as The Royal Bank of Scotland N.V. (“RBS”)),
in the amount of $35.0 million (the “Bridge Loan Agreement”). Among
other terms, pursuant to a USD facility side letter and warrant agreement dated
April 19, 2007 between the Company and RBS, the Company was obligated to issue
warrants to RBS to purchase up to 400,000 shares of the Company’s common
stock. On April 19, 2007 and October 11, 2007, respectively, the
Company issued warrants to purchase 200,000 shares of its common stock at $8.84
per share and the remaining 200,000 shares at the same price. All of
the warrants may be exercised up to the third anniversary of the completion of a
“Qualified Public Offering”, as defined in the warrant agreement.
On
October 11, 2007, RBS and another lender provided a three year term loan (the
“Original Loan”), to the Company in the aggregate amount of $35.0 million. The
principal amount, and any unpaid accrued interest on the Original Loan, will be
due on October 11, 2010 and may be prepaid without penalty. The Original Loan
bears interest at LIBOR for deposits in U.S. dollars plus 3.5% with interest
payable on the last day of each three-month
82
period.
The loan is secured by a pledge of 10,000 shares of common stock, or 100% of the
outstanding common stock of Synutra Illinois.
The loan
agreement for the Original Loan (the “Original Loan Agreement”) provides for
mandatory prepayment upon the occurrence of certain events, and contains
customary covenants for financings of this type, including restrictions on the
incurrence of liens, payment of dividends, and disposition of properties. The
Original Loan Agreement also contains certain financial covenants, including a
requirement to maintain specified leverage ratio, interest coverage ratio,
tangible net worth, and indebtedness to tangible net worth ratio. The Company
has performed an analysis of the relevant ratios and found that due to the net
loss which resulted from the significant costs of the product recall, including
inventory write-down and write-off and subsequent loss of sales, the Company has
not been able to meet two of the financial covenants since September 30, 2008
and all four financial covenants since December 31, 2008. The Company
requested the lenders to waive these breaches and to reset the financial
covenants for the following quarters, and placed $5.0 million as cash deposit.
On February 26, 2010, the Company entered into an amendment (the “Amendment”) to
its Original Loan Agreement with RBS as administrative agent, as collateral
agent and as arranger, and certain lenders party thereto (the “Lenders”).
Pursuant to the Amendment, the Lenders and RBS agreed to waive their rights and
remedies of the Company’s breaches of certain covenants under the Original Loan
Agreement in exchange for a waiver fee equal to 0.5% of the aggregate principal
amount outstanding under the Original Loan Agreement as of February 26, 2010, or
approximately $175,000. In addition, the Amendment amends the Original Loan
Agreement, among other things, to increase the interest rate of the loan from
LIBOR plus 3.5% to LIBOR plus 4.5%, to place additional cash deposits under
certain circumstances, to increase the cash deposit to an aggregate amount not
in excess of $10.0 million, and replace the financial covenants with a single
requirement to maintain consolidated earnings before interest, taxes,
depreciation and amortization (EBITDA) that is no lower than the minimum
Consolidated EBITDA specified in the Original Loan Agreement, as amended. The
Company has performed an analysis on the consolidated EBITDA and confirmed that
the financial covenant was satisfied as of March 31, 2010.
In
addition to the RBS Loan, as of March 31, 2010 and 2009, the Company had
short-term debt from PRC banks in the amount of $94.2 million and $186.2
million, respectively. The maturity dates of the short-term debt
outstanding range from April 2010 to March 2011. The weighted average interest
rate on short-term debt from PRC banks outstanding at March 31, 2010 and 2009
was 4.7% and 4.1%, respectively. The loans at March 31, 2010 and 2009 were
secured by the pledge of certain fixed assets held by the Company of $33.4
million and $34.2 million, respectively; the pledge of the Company’s land use
right of $2.2 million and $3.4 million, respectively; and the pledge of cash
deposits of $11.7 million and $66.1 million, respectively.
As of
March 31, 2010 and 2009, the Company had long-term debt, including current
portion, from PRC banks in the amount of $67.4 million and $8.8 million,
respectively. The maturity date of the long-term loans and long term loans due
within one year outstanding from PRC banks at March 31, 2010 is from November
2010 to January 2013. The weighted average interest rate of outstanding
long-term loans at March 31, 2010 and 2009 was 5.4% and 5.4%, respectively. The
Company had $0.9 million and nil of loans at March 31, 2010 and 2009,
respectively, that were secured by the pledge of the Company’s land use
right.
As of
March 31, 2010, the Company had unused credit facility of $96.8 million from
certain PRC banks. The interest rate will be negotiated by both parties upon
future drawdown.
Apart
from borrowings from banks, the Company had short term loans from related
parties in the amount of $3.9 million and $7.5 million as of March 31, 2010 and
2009, respectively. The maturity dates of these related party loans at March 31,
2010 range from October 2010 to January 2011, and are extendable on the same
terms upon maturity.
83
13. OTHER
CURRENT LIABILITIES
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Accrued
rebate and slotting fee
|
$ | 1,256 | $ | 844 | ||||
|
Payroll
and bonus payables
|
4,102 | 3,615 | ||||||
|
Accrued
selling and marketing expenses
|
1,098 | 4,118 | ||||||
|
Accrued
advertising and promotion expenses
|
6,116 | 5,984 | ||||||
|
Accrued
interest
|
1,028 | 1,004 | ||||||
|
Accrued
sales return
|
3,960 | 593 | ||||||
|
Accrued
rental
|
1,930 | 38 | ||||||
|
Others
|
3,184 | 5,729 | ||||||
|
Total
|
$ | 22,674 | $ | 21,925 | ||||
14. OBLIGATIONS
UNDER CAPITAL LEASES
Future
minimum capital lease payments at March 31, 2010 are as follows:
|
Year
Ending March 31,
|
||||
|
(In
thousands)
|
||||
|
2011
|
$ | 473 | ||
|
2012
|
473 | |||
|
2013
|
473 | |||
|
2014
|
473 | |||
|
2015
|
473 | |||
|
2016
and thereafter
|
15,419 | |||
|
Total
minimum lease payments
|
$ | 17,784 | ||
|
Less: Amount
representing interest
|
12,412 | |||
|
Present
value of minimum lease payments
|
5,372 | |||
|
Current
|
25 | |||
|
Long-term
|
5,347 | |||
|
Total
|
5,372 | |||
The
interest rate associated with the capital leases is 7.8% per annum.
15.
INCOME TAXES
A. Tax
law of each tax jurisdictions
United
States
Under the
federal and state income tax laws of United States, the Company is subject to
tax on its income or capital gains. As at March 31, 2010, the Company’s
subsidiary in United States does not have any assessable profit and accordingly,
no provision for federal and state income taxes have been provided
thereon.
China
On March
16, 2007, the National People’s Congress of the PRC approved and promulgated the
new Enterprise Income Tax Law (“new EIT Law”), which took effect beginning
January 1, 2008. Under the new EIT law, foreign investment enterprises and
domestic companies are subject to a uniform tax rate of 25%. The new tax law
provides a five-year transition period from its effective date for certain
qualifying enterprises which were established before the promulgation date of
the new tax law and which were entitled to a preferential lower tax rate or tax
holiday under the then effective tax laws or regulations.
84
Some of
the Company’s PRC subsidiaries are eligible under the transition rules to
continue enjoying tax holidays or reduced tax rate until expiration. The
following table illustrates the applicable tax rate and tax holidays of major
PRC subsidiaries under the new EIT Law:
|
Name
of Subsidiaries
|
Statutory
Tax Rate Beginning January 1, 2008
|
Tax
Holiday (based on calendar year)
|
||
|
Shengyuan
Nutritional Food Co., Ltd.
|
25%
|
2
years tax free (2004, 2005); 12% (2006, 2007); 12.5%
(2008)
|
||
|
Heilongjiang
Mingshan Dairy Co., Ltd.
|
25%
|
2
years tax free (2006, 2007); 3 years tax at 12.5%
(2008-10)
|
||
|
Zhangjiakou
Shengyuan Dairy Co., Ltd.
|
25%
|
2
years tax free (2006, 2007); 3 years tax at 12.5%
(2008-10)
|
||
|
Inner
Mongolia Shengyuan Food Co., Ltd.
|
25%
|
2
years tax free (2008, 2009); 3 years tax at 12.5%
(2010-12)
|
||
|
Inner
Mongolia Mengyuan Food Co., Ltd.
|
25%
|
No
tax holiday
|
||
|
Meitek
Technology (Qingdao) Co., Ltd.
|
25%
|
2
years tax free (2008, 2009); 3 years tax at 12.5%
(2010-12)
|
||
|
Heilongjiang
Baoquanling Shengyuan Dairy Co., Ltd.
|
25%
|
No
tax holiday
|
||
|
Beijing
Shengyuan Huiliduo Food Technology Co., Ltd.
|
25%
|
No
tax holiday
|
||
|
Global
Food Trading (Shanghai) Co., Ltd.
|
25%
|
20%
(2009), 22% (2010), 24% (2011), 25%
(2012)
|
According to the PRC Tax Administration
and Collection Law, the statute of limitations is three years if the
underpayment of income taxes is due to computational errors made by the
taxpayer. The statute of limitations may be extended to five years
under special circumstances, which are not clearly defined, but an underpayment
of income tax liability exceeding RMB 100,000 is specifically listed as a
special circumstance. In the case of a transfer pricing related adjustment, the
statute of limitations is ten years. There is no statute of limitations in the
case of tax evasion. The Company's PRC subsidiaries are therefore
subject to examination by the PRC tax authorities from 2005 through 2009 on
non-transfer pricing matters, and from 2000 through 2009 on transfer pricing
matters.
|
B.
|
Components
of income (loss) before income tax expense
(benefit)
|
For
financial reporting purposes, income (loss) before income tax expense (benefit)
includes the following components:
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
United
States
|
$ | (2,745 | ) | $ | (3,320 | ) | $ | (7,235 | ) | |||
|
PRC
|
(29,031 | ) | (127,655 | ) | 60,762 | |||||||
| $ | (31,776 | ) | $ | (130,975 | ) | $ | 53,527 | |||||
|
C.
|
Reconciliation
from income tax at statutory rate to reported amount of income tax
expense
|
The
income tax expense reconciled to the tax expense computed at the US statutory
rate was approximately as follows for the years ended March 31, 2010, 2009 and
2008:
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
Income
tax provision (benefit) at US federal statutory rate of
34%
|
$ | (10,804 | ) | $ | (44,531 | ) | $ | 18,199 | ||||
|
Foreign
tax rate differential
|
2,447 | 11,489 | (2,393 | ) | ||||||||
85
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
PRC
tax holiday effect
|
1,411 | (3,228 | ) | (13,978 | ) | |||||||
|
Change
in valuation allowance
|
4,092 | 3,072 | 2,135 | |||||||||
|
FIN
48 unrecognized tax benefits
|
78 | 81 | 508 | |||||||||
|
Effect
of tax refund
|
(4,318 | ) | — | — | ||||||||
|
Effect
of change in tax rate
|
(898 | ) | — | — | ||||||||
|
Nondeductible
(nontaxable) items
|
1,088 | 2,731 | 3,384 | |||||||||
|
Total
income tax expense (benefit)
|
$ | (6,904 | ) | $ | (30,386 | ) | $ | 7,855 | ||||
Certain
Company’s PRC subsidiaries are eligible for tax holidays. The impact of these
tax holidays increased PRC taxes by $1.4 million for the fiscal year ended March
31, 2010 and decreased PRC taxes by $3.2 million and $14.0 million for the
fiscal year ended March 31, 2009 and 2008, respectively. The benefit of the tax
holidays on earnings (loss) per share was negative $0.03, $0.06 and $0.26 for
the fiscal years ended March 31, 2010, 2009 and 2008, respectively.
|
D.
|
Deferred
tax assets by type of temporary difference, credits and change in
valuation allowance
|
Deferred
income taxes reflect the net tax effects of temporary differences between the
carrying amounts of assets and liabilities for financial reporting purposes and
the amounts used for income tax purposes. Significant components of the
Company’s net deferred tax assets are as follows:
|
March
31,
2010
|
March
31,
2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Deferred
tax assets – current
|
||||||||
|
Inventory
write-down
|
$ | 5,166 | $ | 8,005 | ||||
|
Promotional
expenses carryforward
|
25 | 7,367 | ||||||
|
Net
operating loss carryforward
|
29,770 | 151 | ||||||
|
Accrued
expenses
|
50 | 88 | ||||||
|
Bad
debt provision
|
1,188 | 362 | ||||||
|
Others
|
428 | 547 | ||||||
|
Subtotal
|
36,627 | 16,520 | ||||||
|
Less:
valuation allowance
|
(3,237 | ) | (244 | ) | ||||
|
Total
current deferred tax assets
|
$ | 33,390 | $ | 16,276 | ||||
|
Deferred
tax assets – non current
|
||||||||
|
Net
operating loss carryforward
|
$ | 7,732 | $ | 20,988 | ||||
|
Tax
credits carryforward for PRC equipment purchasing
|
2,086 | 2,083 | ||||||
|
Fixed
assets
|
254 | 278 | ||||||
|
Others
|
168 | 78 | ||||||
|
Subtotal
|
10,240 | 23,427 | ||||||
|
Less:
valuation allowance
|
(6,062 | ) | (4,963 | ) | ||||
|
Total
non-current deferred tax assets
|
$ | 4,178 | $ | 18,464 | ||||
The net
change during the year in the total valuation allowance is as
follows:
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Balance
at beginning of year
|
$ | 5,207 | $ | 2,135 | ||||
|
Additions
during the year
|
4,092 | 3,072 | ||||||
|
Balance
at end of year
|
$ | 9,299 | $ | 5,207 | ||||
86
The Company considers positive and
negative evidence to determine whether some portion or all of the deferred tax
assets will more likely than not be realized. This assessment considers, among
other matters, the nature, frequency and severity of recent losses, forecasts of
future profitability, the duration of statutory carryforward periods, the
Company’s experience with tax attributes expiring unused and tax planning
alternatives. Valuation allowances have been established for deferred tax assets
based on a more likely than not threshold. The Company’s ability to realize
deferred tax assets depends on its ability to generate sufficient taxable income
within the carryforward periods provided for under the tax law. The Company
considers the following possible sources of taxable income when assessing the
realization of deferred tax assets:
- future reversals of existing taxable
temporary differences;
- further
taxable income exclusive of reversing temporary differences and
carryforwards;
- future
taxable income arising from implementing tax planning strategies.
At March 31, 2010, the net operating
loss carryforward of Synutra Illinois and Synutra International, Inc., which
file a consolidated tax return, is approximately $16.6 million, which expires in
2030. Subject to the federal income tax rate of 34%, the tax benefit is $5.7
million as of March 31, 2010. Synutra Illinois and Synutra International, Inc.
do not have enough profit in the foreseeable future to realize the tax benefit.
At March 31, 2010, the net operating loss carryforward of certain PRC
subsidiaries is $6.2 million, which expires in 2015, and the tax benefit is $1.6
million. The Company believes that these PRC subsidiaries do not have
enough profit in the foreseeable future to realize the tax
benefit. At March 31, 2010, deductible tax credits carryforward for
PRC equipment purchasing was $2.1 million, which expires in 2012. The Company
believes that relative PRC subsidiaries do not have enough profit in the
foreseeable future to realize the tax benefit. As a result, the Company believes
that it is more likely than not that the benefit from the net operating loss
carryforward of Synutra Illinois and Synutra International, Inc., the net
operating loss carryforward of certain PRC subsidiaries and the benefit from the
tax credits carryforward for PRC equipment purchasing will not be realized. In
recognizing this risk, the Company has fully provided a valuation allowance on
the deferred tax assets relating to these operating loss carryforward of Synutra
Illinois and Synutra International, Inc., the net operating loss carryforward of
certain PRC subsidiaries and these tax credits carryforward for PRC equipment
purchasing. If or when recognized, the tax benefits relating to any reversal of
the valuation allowance on deferred tax assets at March 31, 2010 will be
recognized as a reduction of income tax expense.
|
E.
|
Significant
components of income tax expense
|
Income
taxes for the PRC subsidiaries are calculated on a separate entity basis. Each
of the Company’s PRC subsidiaries files stand-alone tax returns. The income tax
expense for the fiscal years ended March 31, 2010, 2009 and 2008 are summarized
as follows:
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
Current
tax
|
||||||||||||
|
US
|
$ | — | $ | — | $ | — | ||||||
|
PRC
|
(4,101 | ) | 738 | 10,550 | ||||||||
| (4,101 | ) | 738 | 10,550 | |||||||||
|
Deferred
tax
|
||||||||||||
|
Deferred
tax-benefit-US
|
— | — | 218 | |||||||||
|
Deferred
tax-benefit-PRC
|
(2,803 | ) | (31,124 | ) | (2,913 | ) | ||||||
| (2,803 | ) | (31,124 | ) | (2,695 | ) | |||||||
|
Total
|
$ | (6,904 | ) | $ | (30,386 | ) | $ | 7,855 | ||||
|
F.
|
Uncertainty
in income tax
|
Effective
on January 1, 2007, the Company adopted the provisions of ASC 740-10, “Income
Taxes” (previously FASB Interpretation No. 48, “Accounting for Uncertainty in
Income Taxes—an Interpretation of SFAS No. 109”).
87
The
Company made its assessment of the level of authority for each tax position
(including the potential application of interests and penalties) based on the
technical merits, and has measured the unrecognized benefits associated with the
tax positions.
The
adoption of FIN 48 has reduced the retained earnings as of April 1, 2007, by
$375,000, including interest and penalties amounting to $55,000, with a
corresponding increase in the liability for uncertain tax positions. The above
mentioned liability is recorded in other long term liabilities in the
consolidated balance sheet. The Company recognizes interest and penalties
related to unrecognized tax benefits within the income tax expense line in the
accompanying consolidated statement of operations. Accrued interest and
penalties are included within the related tax liability line in the consolidated
balance sheet. The Company does not anticipate any significant increases or
decreases to its liability for unrecognized tax benefits within the next 12
months.
The
following is a tabular reconciliation of the total amounts of unrecognized tax
benefits for the years presented:
|
March
31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
Balance
at beginning of year
|
$ | 964 | $ | 883 | $ | 375 | ||||||
|
Additions
based on tax positions related to the current year
|
78 | 343 | 508 | |||||||||
|
Reductions
for tax positions of prior years
|
— | (262 | ) | — | ||||||||
|
Balance
at end of year
|
$ | 1,042 | $ | 964 | $ | 883 | ||||||
The
balance of unrecognized tax benefits at March 31, 2010, if recognized, would
affect the effective tax rate.
According
to PRC Tax Administration and Collection Law, the statute of limitations is
three years if the underpayment of taxes is due to computational errors made by
the taxpayer or withholding agent. The statute of limitations will be extended
to five years under special circumstances, which are not clearly defined (but an
underpayment of tax liability exceeding RMB100,000 is specifically listed as a
special circumstance). In the case of a related party transaction, the statute
of limitations is 10 years. There is no statute of limitations in the case of
tax evasion. The Company is subject to taxation in the US and the PRC. The
statute of limitations in the US is three years.
For the
fiscal year ended March 31, 2010, the Company’s foreign subsidiaries did not
have any distributable earnings.
|
16.
|
EARNINGS
(LOSS) PER SHARE
|
For
purposes of calculating basic and diluted earnings per share, the Company used
the following weighted average common shares outstanding:
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands except for per share data)
|
||||||||||||
|
Net
income (loss) attributable to common shareholders
|
$ | (24,615 | ) | $ | (100,549 | ) | $ | 45,661 | ||||
|
Basic
weighted average common shares outstanding
|
54,001 | 54,001 | 53,170 | |||||||||
|
Dilutive
potential common shares from warrants
|
— | — | 306 | |||||||||
|
Diluted
weighted average shares outstanding
|
54,001 | 54,001 | 53,476 | |||||||||
|
Earnings
(loss) per share-basic
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.86 | ||||
|
Earnings
(loss) per share-diluted
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.85 | ||||
The
warrants to purchase 400,000 shares of common stock granted to RBS in connection
with long-term financing (Note 12) the Bridge Loan were excluded from the
computation of diluted earnings per share for the fiscal
88
year
ended March 31, 2010 and 2009 as they would be anti-dilutive. There were no
anti-dilutive warrants excluded from the computation of diluted earnings per
share for the fiscal year ended March 31, 2008.
|
17.
|
COMMITMENTS
AND CONTINGENCIES
|
|
A.
|
Purchase
commitments
|
As of
March 31, 2010, the Company had outstanding commitments in the amount of $18.9
million for advertising purchase within the next twelve months.
|
B.
|
Capital
commitments
|
As of
March 31, 2010, the Company’s capital commitments amounted to $4.6 million in
relation to asset improvement and plant expansion within the next twelve
months.
|
C.
|
Operating
lease commitments
|
The
Company leases certain office facilities and certain warehouses under
non-cancellable operating leases. The operating lease commitments of the Company
at March 31, 2010 are as follows:
|
Year
Ending
March
31,
(In
thousands)
|
||||
|
2011
|
$ | 1,590 | ||
|
2012
|
1,841 | |||
|
2013
|
1,841 | |||
|
2014
|
1,788 | |||
|
2015
|
1,751 | |||
|
2016
and thereafter
|
57,589 | |||
|
Total
|
$ | 66,400 | ||
|
D.
|
Legal
proceedings
|
As of
March 31, 2010, the end of the period covered by this report, the Company was
subject to various legal proceedings and claims discussed below, as
well as certain other legal proceedings and claims that have not been fully
resolved and that have arisen in the ordinary course of business. Other than as
discussed below, in the opinion of management, the Company does not have a
potential liability related to any current legal proceedings and claims that
would individually or in the aggregate have a material adverse effect on its
financial condition or operating results. However, the results of legal
proceedings cannot be predicted with certainty. The Company intends to contest
each lawsuit vigorously but should the Company fail to prevail in any of these
legal matters or should several of these legal matters be resolved against the
Company in the same reporting period, the operating results of a particular
reporting period could be materially adversely affected.
On March
29, 2010, U.S. District Judge Deborah Chasanow for the District of Maryland
ordered the dismissal of a complaint filed January 15, 2009 on behalf of 54
Chinese families alleged to be affected by melamine contamination, against
Synutra International, Inc. and Synutra Inc. (Jiali Tang, et al vs. Synutra
International, Inc., et al.), alleging negligent or intentional infliction of
personal injury, negligent or intentional infliction of emotional distress,
battery, breach of warranty, fraudulent or negligent misrepresentation, seeking
compensation for punitive damages in the amount of US$500 million, together with
any compensatory damages. In an opinion issued the same date of the order above,
the court sided with the Company’s positions and granted the motion to dismiss
on the grounds of forum
non conveniens. The court also granted the motion to file under seal a
response to a Notice of Recent Development filed by the Plaintiffs. In
considering the motion to dismiss on the grounds of forum
non conveniens, the court examined both the availability and adequacy of
the alternative forum in China as well as how public and private interests favor
the choice of forum. In addition, taking into account that an “alternative
compensation plan is undisputedly available to Plaintiffs,” the court ruled that
“a conditional dismissal will not be employed to protect the Plaintiffs’ rights
to pursue a judicial remedy in the alternative forum.” On April 27, 2010, the
plaintiffs filed a notice of appeal of the dismissal
order. Plaintiffs’ opening brief is due June 28,
2010.
Management
continues to evaluate the lawsuits discussed above and based on the stage of
these proceedings, management is unable to reasonably estimate the likelihood of
any loss or the amount or range of any potential loss that could result from the
litigation. Therefore, no accrual has been established for any potential loss in
connection with these lawsuits.
89
On March
2, 2009, a lawsuit was filed in China on behalf of 54 Chinese families alleged
to be affected by melamine contamination against our subsidiary, Shengyuan
Nutritional Food Co., Ltd. at Qingdao Intermediate People’s Court, seeking
compensation for damages, including medical and other expenses, emotional harm
and punitive damages, for an aggregate amount over US$1.0 million (RMB6.9
million). Further, on April 14, 2009, a lawsuit was filed on behalf of 73
families (including the original 54 families) against our subsidiary, Shengyuan
Nutritional Food Co., Ltd. at Shandong Higher People’s Court on April 14, 2009,
seeking compensation for damages of over US$1.7 million (RMB11.3 million). The
PRC Civil Procedure Law provides that, generally, a court shall make a decision
as to whether it will accept a case within seven days after a lawsuit is
filed. To date, we have not received any notice from such PRC courts
that a lawsuit has been accepted against us in connection with the melamine
contamination incident. Therefore, no accrual has been established for any
potential loss in connection with these lawsuits. It is also possible that these
plaintiffs may bring other similar suits against us in the same court or in a
new court.
|
E.
|
Guarantees
|
As at
March 31, 2010, the Company had an outstanding guarantee issued to the Zhangbei
Branch of the Agricultural Bank of China related to various bank loans totaling
$1.1 million to 104 dairy farmers in the Zhangbei Area (the “Guarantee”). Such
loans matured on December 25, 2007, but as of March 31, 2010, none of the loans
had been repaid and the total outstanding loan amount remained $1.1 million. The
Company has prepared its best estimate of possible losses related to these loans
based on a weighed average range of likely probabilities and potential payments
that would be required under the Guarantee, and estimated a potential loss of
$367,000 related to the Guarantee.
|
18.
|
SEGMENT
REPORTING AND GEOGRAPHIC
INFORMATION
|
The
Company focuses on selling premium infant formula products, which are
supplemented by more affordable infant formula products targeting the mass
market as well as other nutritional products, such as adult powdered formula and
prepared baby food, and certain nutritional ingredients and supplements. The
activities of each segment are as follows:
Powdered Formula - Sales of
powdered infant and adult formula products.
Baby Food - Sales of prepared
baby food for babies and children.
Nutritional Ingredients and
Supplements - Sales of nutritional ingredients and supplements such as
chondroitin sulfate, and microencapsulated Docosahexanoic Acid (“DHA”) and
Arachidonic Acid (“ARA”).
“All
Other” includes non-core businesses such as toll packaging, toll drying service
and sales of ingredients and materials to industrial customers.
The
Company’s underlying accounting records are maintained on a legal entity basis
for government and public reporting requirements. Segment disclosures are on a
performance basis consistent with internal management reporting.
90
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(In
thousands)
|
||||||||||||
|
NET
SALES TO EXTERNAL CUSTOMERS
|
||||||||||||
|
Powdered
formula
|
$ | 194,383 | $ | 284,818 | $ | 321,130 | ||||||
|
Baby
food
|
886 | 271 | — | |||||||||
|
Nutritional
ingredients and supplements
|
1,451 | — | — | |||||||||
|
All
other
|
95,166 | 27,439 | 40,960 | |||||||||
|
Net
sales
|
$ | 291,886 | $ | 312,528 | $ | 362,090 | ||||||
|
INTERSEGMENT
SALES
|
||||||||||||
|
Powdered
formula
|
$ | 7 | $ | — | $ | — | ||||||
|
Baby
food
|
535 | — | — | |||||||||
|
Nutritional
ingredients and supplements
|
11,284 | 3,367 | — | |||||||||
|
All
other
|
1,778 | — | — | |||||||||
|
Intersegment
sales
|
$ | 13,604 | $ | 3,367 | $ | — | ||||||
|
GROSS
PROFIT
|
||||||||||||
|
Powdered
formula
|
$ | 96,207 | $ | 50,336 | $ | 178,728 | ||||||
|
Baby
food
|
93 | 150 | — | |||||||||
|
Nutritional
ingredients and supplements
|
(1,222 | ) | — | — | ||||||||
|
All
other
|
(11,668 | ) | 2,956 | 7,794 | ||||||||
|
Gross
profit
|
$ | 83,410 | $ | 53,442 | $ | 186,522 | ||||||
|
Selling
and distribution expenses
|
43,989 | 44,178 | 34,449 | |||||||||
|
Advertising
and promotion expenses
|
33,854 | 115,478 | 76,388 | |||||||||
|
General
and administrative expenses
|
24,509 | 25,455 | 16,013 | |||||||||
|
Impairment
loss
|
5,894 | — | — | |||||||||
|
Other
operating income, net
|
894 | 5,790 | 1,492 | |||||||||
|
Income
(loss) from operations
|
(23,942 | ) | (125,879 | ) | 61,164 | |||||||
|
Interest
expense
|
8,603 | 4,857 | 6,354 | |||||||||
|
Interest
income
|
1,850 | 341 | 1,801 | |||||||||
|
Other
income (expense), net
|
(1,081 | ) | (580 | ) | (3,084 | ) | ||||||
|
Income
(loss) before income tax expense (benefit)
|
$ | (31,776 | ) | $ | (130,975 | ) | $ | 53,527 | ||||
|
CAPITAL
EXPENDITURE
|
||||||||||||
|
Powdered
formula
|
$ | 3,961 | $ | 45,492 | $ | 24,567 | ||||||
|
Baby
food
|
771 | 4,389 | 6,088 | |||||||||
|
Nutritional
ingredients and supplements
|
460 | 8,929 | 10,853 | |||||||||
|
All
other
|
1,243 | 3,231 | 1 | |||||||||
|
Total
|
$ | 6,435 | $ | 62,041 | $ | 41,509 | ||||||
|
DEPRECIATION
AND AMORTIZATION
|
||||||||||||
|
Powdered
formula
|
$ | 7,282 | $ | 6,686 | $ | 4,558 | ||||||
|
Baby
food
|
920 | 351 | 61 | |||||||||
|
Nutritional
ingredients and supplements
|
1,746 | 608 | 26 | |||||||||
|
All
other
|
150 | 4 | 1 | |||||||||
|
Total
|
$ | 10,098 | $ | 7,649 | $ | 4,646 | ||||||
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
TOTAL
ASSETS
|
||||||||
|
Powdered
formula
|
$ | 333,975 | $ | 443,946 | ||||
|
Baby
food
|
35,115 | 30,291 | ||||||
|
Nutritional
ingredients and supplements
|
37,879 | 49,611 | ||||||
|
All
other
|
115,787 | 115,923 | ||||||
|
Intersegment
elimination
|
(173,399 | ) | (167,200 | ) | ||||
|
Total
|
$ | 349,357 | $ | 472,571 | ||||
91
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(In
thousands)
|
||||||||
|
LONG
LIVED ASSETS
|
||||||||
|
Powdered
formula
|
$ | 71,581 | $ | 106,726 | ||||
|
Baby
food
|
21,338 | 21,223 | ||||||
|
Nutritional
ingredients and supplements
|
21,653 | 22,630 | ||||||
|
All
other
|
1,461 | 276 | ||||||
|
Total
|
$ | 116,033 | $ | 150,855 | ||||
Consolidated
revenue is generated from sales in the following areas:
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
Sales
in Mainland China
|
99.7% | 98.9% | 94.8% | |||||||||
|
Sales
to United States
|
0.3% | 1.1% | 5.2% | |||||||||
Only one
customer in the U.S. accounted for more than 5% of the Company’s total sales in
fiscal year ended March 31, 2008.
All of
the Company’s long-lived assets are located in China.
|
19.
|
MAINLAND
CHINA CONTRIBUTION PLAN AND RESTRICTED NET
ASSETS
|
|
A.
|
China
Contribution Plan
|
Full time
employees of the Company in the PRC participate in a government-mandated
multi-employer defined contribution plan pursuant to which certain pension
benefits, medical care, unemployment insurance and other welfare benefits are
provided to employees. PRC labor regulations require the Company to accrue for
these benefits based on certain percentage of the employees’ salaries. The total
contribution for such employee benefits were $6.1 million, $4.9 million and $2.7
million for the fiscal year ended March 31, 2010, 2009 and 2008,
respectively.
|
B.
|
Restricted
Net Assets
|
Relevant
PRC statutory laws and regulations permit payments of dividends by the Company’s
PRC subsidiaries only out of their retained earnings, if any, as determined in
accordance with PRC accounting standards and regulations. PRC laws and
regulations require that annual appropriations of 10% of after-tax income should
be set aside prior to payment of dividends as a general reserve fund. In
addition, there are restrictions on the distribution of share capital from the
Company’s PRC subsidiaries. As a result of these PRC laws and regulations, the
Company’s PRC subsidiaries and PRC affiliates are restricted in their ability to
transfer a portion of their net assets to the Company in the form of dividends,
loans or advances. Such restricted portions amounted to approximately $52.3
million as of March 31, 2010.
|
20.
|
QUARTERLY
FINANCIAL DATA (UNAUDITED)
|
Summarized
quarterly financial information in fiscal years ended March 31, 2010 and 2009 is
as follows:
|
Fiscal
Year 2010
|
Fiscal
Year 2009
|
|||||||||||||||||||||||||||||||
|
First
|
Second
|
Third
|
Fourth
|
First
|
Second
|
Third
|
Fourth
|
|||||||||||||||||||||||||
|
(In
thousands except per share data)
|
||||||||||||||||||||||||||||||||
|
Net
sales
|
$ | 47,350 | $ | 65,330 | $ | 96,795 | $ | 82,411 | $ | 127,380 | $ | 94,791 | $ | 17,658 | $ | 72,699 | ||||||||||||||||
|
Gross
profit (loss)
|
19,572 | 12,279 | 12,620 | 38,939 | 66,895 | (26,723 | ) | (9,760 | ) | 23,030 | ||||||||||||||||||||||
|
Net
income (loss) attributable to Synutra International, Inc. common
shareholders
|
(9,946 | ) | (14,019 | ) | (9,728 | ) | 9,078 | 15,644 | (49,680 | ) | (49,342 | ) | (17,171 | ) | ||||||||||||||||||
|
Weighted
average common share outstanding – basic
|
54,001 | 54,001 | 54,001 | 54,001 | 54,001 | 54,001 | 54,001 | 54,001 | ||||||||||||||||||||||||
|
Weighted
average common share outstanding – diluted
|
54,001 | 54,001 | 54,001 | 54,187 | 54,291 | 54,001 | 54,001 | 54,001 | ||||||||||||||||||||||||
|
Earnings
per share, basic
|
$ | (0.18 | ) | $ | (0.26 | ) | $ | (0.18 | ) | $ | 0.17 | $ | 0.29 | $ | (0.92 | ) | $ | (0.91 | ) | $ | (0.32 | ) | ||||||||||
|
Earnings
per share, diluted
|
$ | (0.18 | ) | $ | (0.26 | ) | $ | (0.18 | ) | $ | 0.17 | $ | 0.29 | $ | (0.92 | ) | $ | (0.91 | ) | $ | (0.32 | ) | ||||||||||
92
|
21.
|
SUBSEQUENT
EVENTS
|
From
April 1, 2010 to June 1, 2010, in order to further improve the working capital
condition, the Company prepaid short-term and long-term borrowings from PRC
banks in the amount of $49.8 million, with original maturity date ranging from
September 2010 to April 2011 and with weighted average interest rate of 5.5%,
and borrowed long-term loan from the same PRC banks in the same amount, with
maturity date ranging from August 2011 to May 2012 and with weighted average
interest rate of 5.4%.
93
SYNUTRA
INTERNATIONAL, INC.
ADDITIONAL
INFORMATION - FINANCIAL STATEMENT SCHEDULE I
FINANCIAL
INFORMATION OF PARENT COMPANY
BALANCE
SHEETS
|
March
31, 2010
|
March
31, 2009
|
|||||||
|
(in
thousands, except share par value)
|
||||||||
|
ASSETS
|
||||||||
|
Current
Assets:
|
||||||||
|
Cash
and cash equivalents
|
$ | 421 | $ | 441 | ||||
|
Prepaid
expenses and other current assets
|
1 | 1 | ||||||
|
Total
current assets
|
422 | 442 | ||||||
|
Due
from subsidiaries
|
38,627 | 39,540 | ||||||
|
Investments
in subsidiaries
|
50,215 | 73,098 | ||||||
|
Other
assets
|
3,179 | 4,153 | ||||||
|
TOTAL
ASSETS
|
$ | 92,443 | $ | 117,233 | ||||
|
LIABILITIES
AND EQUITY
|
||||||||
|
Current
Liabilities:
|
||||||||
|
Short-term
loans
|
38,685 | 38,364 | ||||||
|
Other
current liabilities
|
884 | 1,166 | ||||||
|
Total
current liabilities
|
39,569 | 39,530 | ||||||
|
Due
to subsidiaries
|
536 | 844 | ||||||
|
Total
liabilities
|
40,105 | 40,374 | ||||||
|
Equity:
|
||||||||
|
Synutra
International, Inc., shareholders’ equity
|
||||||||
|
Common
stock, $.0001 par value: 250,000 authorized; 54,001 and 54,001 issued and
outstanding at March 31, 2010 and 2009, respectively
|
5 | 5 | ||||||
|
Additional
paid-in capital
|
76,607 | 76,607 | ||||||
|
Accumulated
deficit
|
(48,289 | ) | (23,674 | ) | ||||
|
Accumulated
other comprehensive income
|
24,015 | 23,921 | ||||||
|
Total
equity
|
52,338 | 76,859 | ||||||
|
TOTAL
LIABILITIES AND EQUITY
|
$ | 92,443 | $ | 117,233 | ||||
94
FINANCIAL
INFORMATION OF PARENT COMPANY
STATEMENTS
OF INCOME
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands except earnings per share data)
|
||||||||||||
|
Operating
expenses:
|
||||||||||||
|
General
and administrative expenses
|
$ | 3,163 | $ | 2,605 | $ | 3,073 | ||||||
|
Loss
from operations
|
3,163 | 2,605 | 3,073 | |||||||||
|
Interest
expense
|
1,236 | 1,342 | 4,466 | |||||||||
|
Interest
income
|
4 | 13 | 1,064 | |||||||||
|
Other
income, net
|
2,600 | 1,000 | — | |||||||||
|
Equity
in earnings (losses) of subsidiaries
|
(22,820 | ) | (97,615 | ) | 52,136 | |||||||
|
Net
income (loss) attributable to Synutra International, Inc. common
shareholders
|
$ | (24,615 | ) | $ | (100,549 | ) | $ | 45,661 | ||||
|
Earnings
(loss) per share-basic
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.86 | ||||
|
Earnings
(loss) per share-diluted
|
$ | (0.46 | ) | $ | (1.86 | ) | $ | 0.85 | ||||
|
Weighted
average common share outstanding-basic
|
54,001 | 54,001 | 53,170 | |||||||||
|
Weighted
average common share outstanding-diluted
|
54,001 | 54,001 | 53,476 | |||||||||
95
FINANCIAL
INFORMATION OF PARENT COMPANY
STATEMENTS
OF CASH FLOWS
|
Year
Ended March 31,
|
||||||||||||
|
2010
|
2009
|
2008
|
||||||||||
|
(in
thousands)
|
||||||||||||
|
Cash
flow from operating activities:
|
||||||||||||
|
Net
income (loss)
|
$ | (24,615 | ) | $ | (100,549 | ) | $ | 45,661 | ||||
|
Adjustments
to reconcile net income to net cash provided by operating
activities:
|
||||||||||||
|
Equity
in (earnings) losses of subsidiaries
|
22,820 | 97,615 | (52,136 | ) | ||||||||
|
Amortization
of debt discount
|
— | — | 2,733 | |||||||||
|
Amortization
of debt issuance costs
|
321 | 321 | 539 | |||||||||
|
Changes
in assets and liabilities:
|
||||||||||||
|
Due
from subsidiaries
|
913 | 778 | (40,318 | ) | ||||||||
|
Prepaid
expenses and other current assets
|
450 | (1,855 | ) | (710 | ) | |||||||
|
Due
to subsidiaries
|
(152 | ) | 110 | 172 | ||||||||
|
Other
current liabilities
|
(281 | ) | 3,730 | 799 | ||||||||
|
Other
assets
|
524 | (1,658 | ) | (788 | ) | |||||||
|
Net
cash provided by (used in) operating activities
|
(20 | ) | (1,508 | ) | (44,048 | ) | ||||||
|
Cash
flow from investing activities:
|
||||||||||||
|
Capital
injection at subsidiaries
|
— | (225 | ) | (54,981 | ) | |||||||
|
Net
cash used in investing activities
|
— | (225 | ) | (54,981 | ) | |||||||
|
Cash
flow from financing activities:
|
||||||||||||
|
Proceeds
from bridge loan
|
— | — | 35,000 | |||||||||
|
Repayment
of bridge loan
|
— | — | (35,000 | ) | ||||||||
|
Proceeds
from long-term loan
|
— | — | 35,000 | |||||||||
|
Proceeds
from issuance of common stock
|
— | — | 66,000 | |||||||||
|
Issuance
costs for common stock issuance
|
(206 | ) | ||||||||||
|
Net
cash provided by financing activities
|
— | — | 100,794 | |||||||||
|
Net
change in cash and cash equivalents
|
(20 | ) | (1,733 | ) | 1,765 | |||||||
|
Cash
and cash equivalents, beginning of year
|
441 | 2,174 | 409 | |||||||||
|
Cash
and cash equivalents, end of year
|
$ | 421 | $ | 441 | $ | 2,174 | ||||||
96
Note
to Schedule I
The
parent company financial statements have been prepared using the same accounting
principles and policies described in the notes to the consolidated financial
statements, with the only exception being that the company accounts for its
subsidiaries using the equity method. Please refer to the notes to the
consolidated financial statements presented above for additional information and
disclosures with respect to these financial statements.
Schedule
I has been provided pursuant to the requirements of Rule 12-04(a) and 5-04(c) of
Regulation S-X, which require condensed financial information as to the
financial position, changes in financial position and results of operations of a
parent company as of the same dates and for the same periods for which audited
consolidated financial statements have been presented when the restricted net
assets of consolidated subsidiaries exceed 25 percent of consolidated net assets
as of the end of the most recently completed fiscal year.
97
SYNUTRA
INTERNATIONAL, INC.
SCHEDULE
II
VALUATION
AND QUALIFYING ACCOUNTS
|
Description
|
Balance
at Beginning
of
Year
|
Charged
to
Costs
and Expenses
|
Write-off
|
Balance
at End
of
Year
|
||||||||||||||
|
(In
thousands)
|
||||||||||||||||||
|
Allowance
for accounts receivable
|
||||||||||||||||||
| - 2010 | $ | 1,452 | $ | 2,903 | $ | — | $ | 4,355 | ||||||||||
| - 2009 | $ | 185 | $ | 1,267 | $ | — | $ | 1,452 | ||||||||||
| - 2008 | $ | 241 | $ | (56 | ) | $ | — | $ | 185 | |||||||||
|
Allowance
for other receivables
|
||||||||||||||||||
| - 2010 | $ | 1,472 | $ | 1,501 | $ | 875 | $ | 2,098 | ||||||||||
| - 2009 | $ | 585 | $ | 887 | $ | — | $ | 1,472 | ||||||||||
| - 2008 | $ | 576 | $ | 9 | $ | — | $ | 585 | ||||||||||
|
Allowance
for deferred tax assets
|
||||||||||||||||||
| - 2010 | $ | 5,207 | $ | 4,092 | $ | — | $ | 9,299 | ||||||||||
| - 2009 | $ | 2,135 | $ | 3,072 | $ | — | $ | 5,207 | ||||||||||
| - 2008 | $ | — | $ | 2,135 | $ | — | $ | 2,135 | ||||||||||
ITEM
9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND
FINANCIAL DISCLOSURE
None.
ITEM
9A. CONTROLS AND PROCEDURES
Conclusion
Regarding Effectiveness of Disclosure Controls and Procedures
The
Company’s management, with the participation of the Company’s Chief Executive
Officer and Chief Financial Officer, has evaluated the effectiveness of the
Company’s disclosure controls and procedures (as such term is defined in Rules
13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended,
or the Exchange Act) as of the end of the period covered by this
report.
Based on
such evaluation, the Company’s Chief Executive Officer and Chief Financial
Officer have concluded that, as of March 31, 2010, the Company’s disclosure
controls and procedures were effective to ensure that the information required
to be disclosed by the Company in the reports that it files or submits under the
Exchange Act is (a) recorded, processed, summarized, and reported within the
time periods specified in the SEC’s rules and forms and (b) accumulated and
communicated to the Company’s management, including its Chief Executive Officer
and Chief Financial Officer, or persons performing similar functions, as
appropriate to allow timely decisions regarding required
disclosure.
Management’s
Report on Internal Control Over Financial Reporting
The
Company’s management is responsible for establishing and maintaining adequate
internal control over financial reporting (as defined in Rule 13a-15(f) under
the Exchange Act). The Company’s internal control system was designed to provide
reasonable assurance regarding the reliability of financial reporting and the
preparation of its consolidated financial statements for external purposes in
accordance with generally accepted accounting principles. The Company’s
management assessed the effectiveness of the Company’s internal control over
financial reporting as of March 31, 2010. In making this assessment, the
Company’s management used the criteria set forth by the Committee of Sponsoring
Organizations of the Treadway Commission (“COSO”) in Internal Control —
98
Integrated
Framework. The Company’s management concluded that, as of March 31, 2010, the
Company’s internal control over financial reporting was effective based on these
criteria.
Our
independent registered public accounting firm, Deloitte Touche Tohmatsu CPA
Ltd., has audited the effectiveness of our internal control over financial
reporting as of March 31, 2010 and issued its report, which is included
herein.
Changes
in Internal Control over Financial Reporting
There
were no changes in the Company’s internal control over financial reporting
during the fiscal quarter ended March 31, 2010, which were identified in
connection with management’s evaluation required by paragraph (d) of Rules
13a-15 and 15d-15 under the Exchange Act, that have materially affected, or are
reasonably likely to materially affect the Company’s internal control over
financial reporting.
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the
Board of Directors and Stockholders of
Synutra
International Inc.
Rockville,
Maryland
We have
audited the internal control over financial reporting of Synutra International
Inc. (the “Company”) and subsidiaries as of March 31, 2010, based on the
criteria established in Internal Control — Integrated
Framework issued by the Committee of Sponsoring Organizations of the
Treadway Commission. The Company’s management is responsible for maintaining
effective internal control over financial reporting and for its assessment of
the effectiveness of internal control over financial reporting, included in the
accompanying Management's Report on Internal Control Over Financial Reporting.
Our responsibility is to express an opinion on the Company’s internal control
over financial reporting based on our audit.
We
conducted our audit in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether effective
internal control over financial reporting was maintained in all material
respects. Our audit included obtaining an understanding of internal control over
financial reporting, assessing the risk that a material weakness exists, testing
and evaluating the design and operating effectiveness of internal control based
on that risk, and performing such other procedures as we considered necessary in
the circumstances. We believe that our audit provides a reasonable basis for our
opinion.
A
company’s internal control over financial reporting is a process designed by, or
under the supervision of, the company’s principal executive and principal
financial officers, or persons performing similar functions, and effected by the
company’s board of directors, management, and other personnel to provide
reasonable assurance regarding the reliability of financial reporting and the
preparation of financial statements for external purposes in accordance with
generally accepted accounting principles. A company’s internal control over
financial reporting includes those policies and procedures that (1) pertain to
the maintenance of records that, in reasonable detail, accurately and fairly
reflect the transactions and dispositions of the assets of the company; (2)
provide reasonable assurance that transactions are recorded as necessary to
permit preparation of financial statements in accordance with generally accepted
accounting principles, and that receipts and expenditures of the company are
being made only in accordance with authorizations of management and directors of
the company; and (3) provide reasonable assurance regarding prevention or timely
detection of unauthorized acquisition, use, or disposition of the company’s
assets that could have a material effect on the financial
statements.
Because
of the inherent limitations of internal control over financial reporting,
including the possibility of collusion or improper management override of
controls, material misstatements due to error or fraud may not be prevented or
detected on a timely basis. Also, projections of any evaluation of the
effectiveness of the internal control over financial reporting to future periods
are subject to the risk that the controls may become inadequate because of
changes in conditions, or that the degree of compliance with the policies or
procedures may deteriorate.
99
In our
opinion, the Company maintained, in all material respects, effective internal
control over financial reporting as of March 31, 2010, based on the criteria
established in Internal
Control — Integrated Framework issued by the Committee of Sponsoring
Organizations of the Treadway Commission.
We have
also audited, in accordance with the standards of the Public Company Accounting
Oversight Board (United States), the consolidated financial statements and
financial statement schedules as of and for the year ended March 31, 2010, of
the Company and our report dated June 9, 2010 expressed an unqualified opinion
on those financial statements and financial statement schedules and included
explanatory paragraph regarding the adoption of new accounting standard related
to the presentation and disclosure requirement for noncontrolling
interests.
Deloitte
Touche Tohmatsu CPA Ltd.
Shanghai,
China
June 9,
2010
ITEM
9B. OTHER INFORMATION
None.
100
PART
III
ITEM
10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE
GOVERNANCE
EXECUTIVE
OFFICERS, DIRECTORS AND SIGNIFICANT EMPLOYEES
The
following sets forth information about our directors and executive officers as
of June 1, 2010:
|
Name
|
Age
|
Position
|
||
|
Directors
and Executive Officers
|
||||
|
Liang
Zhang
|
50
|
Chairman
of the Board of Directors and Chief Executive Officer
|
||
|
Joseph
Chow
|
48
|
Director
and Interim Chief Financial Officer
|
||
|
David
Hui Li
|
42
|
Director
|
||
|
Jinrong
Chen
|
51
|
Director(1)
|
||
|
Yiu-Chun
Chan
|
58
|
Director(1)
|
||
|
Lei
Lin
|
43
|
Director(1)
|
||
|
Ka-Keung
Yeung
|
51
|
Director(1)
|
||
|
Weiguo
Zhang
|
53
|
President
|
||
|
Donghao
Yang
|
38
|
Chief
Financial Officer
|
|
(1)
|
Member
of Audit Committee, Compensation Committee and Nominating
Committee
|
Liang Zhang. Liang Zhang is
our founder and has served as the chairman of our board of directors and Chief
Executive officer since we became a public company in 2005. Prior to that, Mr.
Zhang served as chief executive officer of Synutra Illinois since 2000. Mr.
Zhang has worked in the food ingredients industry since the 1980s and founded
his first entrepreneurial venture, Honnete, in the early 1990s. Honnete has
since become the dominant supplier of whey protein products in China. Mr. Zhang
has been recognized as a business leader in the dairy industry in China, serving
as vice chairman of the China Dairy Industry Association. Mr. Zhang’s experience
as founder of Honnet gives him valuable insight into marketing strategies in our
industry, and his service as vice chairman of the China Dairy Industry
Association is invaluable to our Board’s discussions of regulatory issues
pertaining to the dairy industry. Mr. Zhang received a bachelor’s degree in
French language and literature from Nanjing International Relations Institute of
China.
Joseph Chow. Joseph Chow has
been our interim Chief Financial Officer since November 10, 2009. Mr. Chow has
served as a director of our board of directors since May 4, 2010 and continues
to serve as a member of our management team. Mr. Chow has over 17 years of
experience in corporate finance, financial advisory and management and has held
senior executive and managerial positions in various public and private
companies. Prior to joining us, Mr. Chow was a managing director of Goldman
Sachs (Asia) LLP from 2008 to 2009. Prior to that, he served as an independent
financial consultant from 2006 to 2008, as chief financial officer of Harbor
Networks Limited from 2005 to 2006, and as chief financial officer of China
Netcom (Holdings) Company Limited from 2001 to 2004. Prior to that Mr. Chow also
served as the director of strategic planning of Bombardier Capital, Inc., as
vice president of international operations of Citigroup and as the corporate
auditor of GE Capital. Mr. Chow currently sits on the board as an independent
non-executive director for Kasen International Holdings Limited and for Intime
Department Store (Group) Co., Ltd. Mr. Chow has extensive knowledge of finance
and accounting issues from his experience as Chief Financial Officer at Harbor
Networks Limited and China Netcom (Holdings) Company Limited. Mr. Chow obtained
a Bachelor of Arts degree in political science from Nanjing Institute of
International Relations and an MBA from the University of Maryland at College
Park.
David Hui Li. David Hui Li
has served as a director of our board of directors since February 8, 2010. Mr.
Li is a managing director of Warburg Pincus Asia LLC, a leading global private
equity and venture capital firm. Mr. Li has been with Warburg Pincus since 2002.
Before joining Warburg Pincus, Mr. Li was an Executive Director of the
investment banking division of Goldman Sachs (Asia) LLC and a Vice President and
an Associate of Morgan Stanley’s investment banking division in Hong Kong and
New York. Mr. Li has been a director of Kasen International Holdings Limited
from May 30, 2006 to October 1, 2008. Mr. Li is also a director of Intime
101
Department
Store (Group) Company Ltd., Tulip Media (International) Limited and RCS Group.,
Ltd. Mr. Li’s experience as Executive Director at Goldman Sachs provides him
with valuable knowledge of capital markets and his experience with Tulip Media
(International) gives the Board perspective with respect to marketing and brand
building. Mr. Li received his B.S. degree in Economics from Renmin University of
China and an MBA from Yale University’s School of Management.
Jinrong Chen. Jinrong Chen
has served as a director of our board of directors since June 27, 2006. Ms. Chen
has served as associate professor at the School of Economics and Management of
Tsinghua University in Beijing since 2001, specializing in corporate finance
management, securities analysis, financial operations, corporate governance and
controls. In addition to her academic career with top business schools in China,
Ms. Chen also advises or sits on the board and audit committee of Bosun Tools
Co., Ltd. and Citic Development – Shenyang Commercial Building (Group) Company
Limited which are listed in China, and certain private businesses in China. Ms.
Chen has extensive knowledge of finance and accounting issues from her
experience as professor at the School of Economics and Management of Tsinghua
University, and her experience with Bosun Tools Co., Ltd. and Citic Development
– Shenyang Commercial Building (Group) Company Limited gives the Board
perspective with respect to corporate governance and brand building. Ms. Chen
received her bachelor’s degree in accounting from Beijing Institute of
Electronics & Information and her MBA degree from Renmin University of
China.
Yiu-Chun Chan. Yiu-Chun Chan
has served as a director of our board of directors since December 3, 2006. Mr.
Chan has over 30 years of experience in marketing agricultural and food products
in the Greater China area. Since 1996 Mr. Chan has been the chief executive
officer of P R Consultants Limited, a company he founded in 1984 with two other
partners. Previously, he served as a partner of Times Direct Marketing Asia—the
largest privately held direct marketing company in South East Asia since 1989
and Executive Director of Lintas Hong Kong Limited from 1987 to 1988. Mr. Chan
began his career marketing agricultural products at Sunkist Growers, where he
was responsible for the advertising and promotion activities in the Hong Kong
market. Mr. Chan’s experience in the agricultural and food products provides him
with valuable knowledge of the dairy industry. Mr. Chan received his Diploma in
communications from Hong Kong Baptist University.
Lei Lin. Lei Lin has served
as a director of our board of directors since October 1, 2007. Since 1992 Mr.
Lin has been president and co-chief executive officer of Sinotrust, a leading
consulting company in China which Mr. Lin founded in 1992. Mr. Lin’s experience
as the founder and co-chief executive officer of Sinotrust provides him with
deep insight into all aspects of business management. Mr. Lin received his
bachelor’s degree in applied economic mathematics from Renmin University of
China.
Ka-Keung Yeung. Ka-Keung
Yeung has served as a director of our Board of Directors since January 8, 2010.
Mr. Yeung is the executive vice president and chief financial officer of Phoenix
Satellite Television Holdings Limited in charge of corporate finance, human
resources and administration. He is also a Qualified Accountant and Company
Secretary of Phoenix Satellite Television Holdings Limited. Mr. Yeung has held
these positions since he joined Phoenix in March 1996 and has been in charge of
all of Phoenix’s internal and external financial management and arrangements as
well as the supervision of administration and personnel matters. Mr. Yeung is a
director of The 9 Limited and Little Sheep Group Limited. Mr. Yeung has
extensive knowledge of finance and accounting issues from his experience as
chief financial officer at Phoenix Satellite Television Holdings Limited, and
his experience with The 9 Limited and Little Sheep Group Limited. These
experiences give the Board perspective with respect to corporate governance and
brand building. Mr. Yeung graduated from the University of Birmingham and
remained in the United Kingdom until 1992 after obtaining his qualification as a
chartered accountant. Upon returning to Hong Kong, he worked at Hutchison
Telecommunications and Satellite Television Asian Region, a subsidiary of News
Corporation, in the fields of finance and business development.
Weiguo Zhang. Weiguo Zhang
has been our president since 2005 and is primarily responsible for our financial
market operations, including investor relations, corporate development, and
international strategic development. Mr. Zhang first joined us as president of
Synutra Illinois in 2001 to oversee our U.S. operations, including information
support in research and technologies and business development. Prior to joining
us, Mr. Zhang was the managing director of Bambridge International, Ltd., which
he founded in 1995. Mr.
Zhang is also a director and a member of the compensation committee and audit
committee of Rino International Corporation. Mr. Zhang received a bachelor’s degree in English
language and literature from the Nanjing International Relations Institute and
a
102
master’s
degree in international economics and American foreign policy from the School of
Advanced International Studies from John Hopkins University.
Donghao Yang. Donghao Yang
has served as our Chief Financial Officer since May 4, 2010. Mr. Yang has over
15 years of experience in corporate finance, commodity trading and international
business development. Mr. Yang has held senior executive and managerial
positions in various public and private companies, including serving as Chief
Financial Officer of Greater China of Tyson Foods, Inc. (NYSE: TSN) from March
2007 to April 2010, as Finance Director Asia Pacific of Valmont Industries, Inc.
(NYSE: VMI) from October 2003 to March 2007, and as Director in China Minmetals
Brazil Holding Limited from January 1999 to April 2001. Mr. Yang earned a
Bachelor of Arts degree in Economics from Nankai University in 1993 and a Master
of Business Administration degree from Harvard Business School in
2003.
Our
directors have a term of office expiring at the next annual general meeting,
unless re-elected or earlier vacated in accordance with our bylaws. Our officers
are appointed by and serve at the discretion of the board of directors. All
officers have a term of office lasting until their removal or replacement by our
board of directors. There are no family relationships among our directors or
officers.
Board
of Directors
Our board
of directors currently has seven directors, consisting of four independent
directors, all of whom are independent as defined by the applicable listing
requirements of the NASDAQ Global Select Market.
On June
11, 2008, our board of directors and stockholders approved our amended and
restated certificate of incorporation, which became effective on October 17,
2008. Our amended and restated certificate of incorporation provides that our
board of directors shall be divided into three classes of directors. The three
classes, which are required to be as nearly equal in number as possible, will be
designated class I, class II and class III, and serve staggered three-year terms
and hold office until their term of office expires or until such time as they
are removed from office by resolution of our stockholders. To effect such
staggered terms, our amended and restated bylaws provides that the directors
first elected to class I shall serve for a term ending on the annual meeting
date following the end of 2008, the directors first elected to class II shall
serve for a term ending on the second annual meeting date following the end of
2008, and the directors first elected to class III shall serve for a term ending
on the third annual meeting date following the end of 2008. Our board of
directors has designated Liang Zhang and William Wu as class I directors,
Jinrong Chen, Yiu-Chun Chan and David Hui Li as class II directors and Lei Lin
and Ka-Keung Yeung as a class III director. At the stockholder’s annual meeting
on Feb 8, 2010, Jinrong Chen and Yiu-Chun Chan were re-elected as class II
directors and David Hui Li was elected as class II director. Upon the
resignation of William Wu as a director on May 4, 2010, Joseph Chow has served
as a class I director.
Board
Committees
Our board
has established the committees described below and may establish others from
time to time.
Audit
Committee
Our audit
committee consists of Jinrong Chen, Lei Lin, Yiu-Chun Chan and Ka-Keung Yeung,
all of whom are independent as defined by the applicable listing requirements of
the NASDAQ Global Select Market. Ms. Chen is the chairperson of our audit
committee and serves as the financial expert of the committee. The audit
committee oversees our accounting and financial reporting processes and the
audits of our financial statements. The audit committee is responsible for,
among other things:
|
·
|
reviewing
and discussing with management and the independent registered public
accounting firm our annual and quarterly financial statements and related
disclosures;
|
|
·
|
coordinating
the oversight and reviewing the adequacy of our internal control over
financial reporting with both management and the independent registered
public accounting firm;
|
|
·
|
establishing
policies and procedures for the receipt and retention of accounting
related complaints and concerns, including a confidential, anonymous
mechanism for the submission of concerns by
employees;
|
103
|
·
|
periodically
reviewing legal compliance matters, including securities trading policies,
periodically reviewing significant accounting and other risks or exposures
to our company, reviewing and, if appropriate, approving all transactions
between our company or its subsidiaries and any related party (as
described in Item 404 of Regulation S-K) and periodically reviewing
business expenses of our chief executive officer;
and
|
|
·
|
establishing
policies for the hiring of employees and former employees of the
independent registered public accounting
firm.
|
Compensation
Committee
Our
compensation committee consists of Jinrong Chen, Yiu-Chun Chan, Lei Lin and
Ka-Keung Yeung. Ms. Chen is the chairperson of our compensation committee. The
purpose of our compensation committee is to discharge the responsibilities of
our board of directors relating to compensation of our executive officers.
Specific responsibilities of our compensation committee include:
|
·
|
reviewing
and recommending approval of compensation of our executive
officers;
|
|
·
|
administering
our stock incentive and employee stock purchase plans;
and
|
|
·
|
reviewing
and making recommendations to our board with respect to incentive
compensation and equity plans.
|
Nominating
Committee
Our
nominating committee consists of Yiu-Chun Chan, Jinrong Chen, Lei Lin and
Ka-Keung Yeung. Mr. Chan is the chairman of our nominating committee. The
purpose of our nominating committee is to be primarily responsible for
identifying individuals qualified to serve as members of the board of directors
and recommending to the board the persons to be nominated by the board as
nominees for director at each annual meeting of our stockholders. Specific
responsibilities of our nominating committee include:
|
·
|
developing
and recommending to the board of directors criteria for board of directors
and committee membership;
|
|
·
|
identifying
individuals qualified to become board of directors
members;
|
|
·
|
recommending
to the board of directors the persons to be nominated for election as
directors and to each of the board of directors’
committees;
|
|
·
|
developing
and recommending to the board of directors a code of ethical conduct and a
set of corporate governance policies and practices;
and
|
|
·
|
monitoring
and evaluating the performance of the board of directors and leading the
board in an annual self-assessment of its practices and
effectiveness.
|
Section
16(a) Beneficial Ownership Reporting Requirements
Section
16(a) of the Securities Exchange Act of 1934 requires our executive officers,
directors and persons who own beneficially more than ten percent of a registered
class of our equity securities to file reports of ownership and changes in
ownership with the Securities and Exchange Commission and the NASDAQ Global
Select Market. The Securities and Exchange Commission has established specific
due dates for these reports, and we must disclose in this 10-K any late filings
during the fiscal year ended March 31, 2010. These persons are required by
regulation of the SEC to furnish copies of all Section 16(a) forms to the
Company. Based solely upon our review of the copies of such forms received by
the Company, we believe that during the year ended March 31, 2010 our officers,
directors and greater than ten percent beneficial owners complied with the
Section 16(a) filing requirements.
104
Code
of Ethics
The
Company has a code of ethics that applies to all of the Company’s employees,
including its principal executive officer, principal financial officer and
principal accounting officer, and the Board of Directors. A copy of this code is
available on the Company’s website at www.synutra.com. The Company intends to
disclose any changes in or waivers from its code of ethics by posting such
information on its website or by filing a Form 8-K.
ITEM
11. EXECUTIVE COMPENSATION
COMPENSATION
DISCUSSION AND ANALYSIS
This
section describes the material elements of compensation earned by our executive
officers during the fiscal year ended March 31, 2010. Our executive compensation
programs are determined and approved by the compensation committee of our board
of directors.
Liang
Zhang and Joseph Chow, who serve as our chief executive officer and our chief
financial officer, respectively, and William W. Wu were our “named executive
officers” for the fiscal year ended March 31, 2010, and the compensation for
these officers is reported in the “Summary Compensation Table” below. No other
executive officer received compensation in excess of $100,000 for the fiscal
year ended March 31, 2010. Lawrence Lee, who served as our chief financial
officer before Joseph Chow, resigned from that position effective November 10,
2009. His compensation is also reported in the table titled “Summary
Compensation Table—Fiscal Years Ended March 31, 2010, 2009 and 2008” below.
Unless otherwise noted, the amounts reported in this Form 10-K have been
converted from Renminbi to U.S. dollars based on the conversion rate as of March
31, 2010 of RMB6.8263 to $1.00.
Overview
of Executive Compensation Program
The
compensation committee has responsibility for establishing, implementing and
monitoring our executive compensation program philosophy and practices. The
compensation committee seeks to ensure that the total compensation paid to our
executive officers is fair, reasonable and competitive.
Compensation
Philosophy and Objectives
The
compensation committee believes that an effective executive compensation program
should provide base annual compensation that is reasonable in relation to the
individual executive’s job responsibilities and reward the achievement of both
annual and long-term strategic goals of our company. Because of the size of our
company, the small number of executive officers in our company, and our
company’s financial priorities, the compensation committee has decided not to
implement or offer any retirement plans, deferred compensation plans or other
similar plans for our executive officers. Accordingly, for the fiscal year ended
March 31, 2010, the components of our executive compensation program consisted
of cash salary only. On June 11, 2008, the Company adopted the 2008 Stock
Incentive Plan (the “Plan”). The Plan provides the Company with the ability to
grant stock-based awards to all employees, officers and directors. Although the
compensation committee currently has not granted any stock-based awards, the
compensation committee may in future years reassess the levels of equity and
cash compensation offered to our executives in light of our profitability and
other performance factors.
Role
of Executive Officers in Compensation Decisions
Our
compensation committee and chief executive officer annually review the
performance of each executive officer (other than the chief executive officer,
whose performance is reviewed only by the compensation committee). The
compensation committee makes all decisions with respect to compensation for the
chief executive officer and will approve grants of equity awards to all of our
executive officers. Decisions regarding the non-equity compensation of our
executive officers other than our chief executive officer are made by our chief
executive officer.
105
Setting
Executive Compensation
In making
its compensation decisions, neither the Company nor the compensation committee
retain outside compensation consultants. Instead, the compensation committee
reviews compensation data for executives of other listed companies located in
China but the Company does not use this data to benchmark the compensation of
its executives. The compensation committee utilizes this data to set
compensation for our executive officers at levels targeted at or around the
average of the compensation amounts provided to similarly situated executives at
comparable local companies considering, for each executive, their individual
experience level and the responsibilities of their position with us. There is no
pre-established policy or target for the allocation between cash and non-cash
incentive compensation.
Employment
Agreements
We have
entered into standard employment agreements with each of our executive officers,
including the named executive officers. The terms and conditions of these
employment agreements are determined via negotiations between the employee
parties and us, and the framework and structure of the agreements are intended
to comply with applicable PRC labor and employment laws.
Liang
Zhang’s employment agreement was automatically extended and has an indefinite
term and provides for an annual base salary of approximately $175,790. Joseph
Chow’s employment agreement has an indefinite term and provides for an annual
base salary of approximately $175,790.
Fiscal
Year 2010 Executive Compensation Components
For the
fiscal year ended March 31, 2010, the principal component of compensation for
our executive officers was their base salary. We provide executive officers with
a base salary to compensate them for services rendered during the fiscal year.
Base salary ranges for the executive officers are determined for each executive
based on his or her position and responsibility.
During
its review of base salaries for executives, the compensation committee primarily
considers:
|
·
|
the
negotiated terms of each executive’s employment
agreement;
|
|
·
|
internal
review of the executive’s compensation, both individually and relative to
other executive officers; and
|
|
·
|
individual
performance of the executive.
|
Salary
levels are typically considered annually as part of our performance review
process, as well as upon a change in job responsibility. Merit-based increases
to salaries are based on the compensation committee’s assessment of the
individual’s performance.
Our
executive officers did not receive any annual bonus or equity-based compensation
for the fiscal year ended March 31, 2010.
Compensation
Committee Report
The
Compensation Committee has reviewed and discussed with management the
“Compensation Discussion and Analysis” required by Item 402(b) of Regulation S-K
and, based on such review and discussions, has recommended to our board of
directors that the foregoing “Compensation Discussion and Analysis” be included
in this Annual Report.
Compensation Committee of
the Board of Directors
Jinrong
Chen (Chairperson)
Yiu-Chun
Chan
Lei
Lin
Ka-Keung
Yeung
106
Summary
Compensation Table — Fiscal Years Ended March 31, 2010, 2009 and
2008
The
following table presents information regarding compensation of our named
executive officers for services rendered during the fiscal years ended March 31,
2010, 2009 and 2008. The amounts reported in this table have been converted from
Renminbi to U.S. dollars based on the March 31, 2010 conversion rate of RMB
6.8263 to $1.00.
|
Name
and Principal Position
|
Year
ended March 31
|
Salary
($)
|
All
Other Compensation ($)
|
Total
($)
|
|||||||||||
|
Liang
Zhang
|
2010
|
175,791 | — | 175,791 | |||||||||||
|
Chairman
of the Board and
|
2009
|
175,544 | — | 175,544 | |||||||||||
|
Chief
Executive Officer
|
2008
|
170,964 | — | 170,964 | |||||||||||
|
Joseph Chow(1)
|
2010
|
73,246 | — | 73,246 | |||||||||||
|
Director
and Interim Chief
|
2009
|
— | — | — | |||||||||||
|
Financial
Officer
|
2008
|
— | — | — | |||||||||||
|
Lawrence Lee(2)
|
2010
|
117,194 | — | 117,194 | |||||||||||
|
Former
Chief Financial
|
2009
|
110,593 | — | 110,593 | |||||||||||
|
Officer
|
2008
|
56,988 | 10,407 | 67,395 | |||||||||||
|
Weiguo
Zhang
|
2010
|
60,000 | — | 60,000 | |||||||||||
|
President
|
2009
|
60,000 | — | 60,000 | |||||||||||
|
2008
|
60,000 | — | 60,000 | ||||||||||||
|
(1)
|
Joseph
Chow served as our interim chief financial officer from November 10, 2009
and will resign from this position on June 10, 2010. He has served as a
member of our Board of Directors since May 4, 2010. The amount reflects
the base salary paid to Joseph Chow from November 10, 2009 to March 31,
2010.
|
|
(2)
|
Lawrence
Lee was appointed our chief financial officer effective October 1, 2007
and resigned effective November 10, 2009. The amounts reported in column
“All Other Compensation” reflect retainer fees for his service on our
board of directors prior to his appointment as our chief financial
officer.
|
Plan-Based
Awards — Fiscal Year Ended March 31, 2010
None of
our named executive officers received any grants of options or other stock-based
awards during the fiscal year ended March 31, 2010. Additionally, none of our
named executive officers held any outstanding options or other stock-based
awards as of the last day of the fiscal year ended March 31, 2010, nor did any
of our named executive officers exercise any options or hold any other stock
awards that vested during the fiscal year ended March 31, 2010.
Potential
Payments upon Termination of Employment or Change of Control
We have
not entered into any arrangements with our executive officers to provide
severance or change of control benefits. Upon a termination of employment by us,
an employee is generally entitled under PRC labor law to one month’s severance
pay for each full year he or she has been employed with us (with a minimum of
one month’s severance pay and a maximum of 12 months’ severance
pay).
Director
Compensation
The
following table presents information regarding the compensation for the fiscal
year ended March 31, 2010 to members of our board of directors who were not also
employed by us (referred to as our “non-employee directors”) during the fiscal
year. None of our non-employee directors held any outstanding options or other
stock-based awards as of the last day of the fiscal year ended March 31, 2010.
The compensation paid to Liang Zhang and Joseph Chow, each of whom is employed
by us, is presented above in the table titled “Summary Compensation Table—Fiscal
Years Ended March 31, 2010, 2009 and 2008” and the related explanatory
notes.
107
|
Name
|
Fees
Earned or Paid in Cash ($)
|
Total
($)
|
||
|
Jinrong
Chen
|
40,000
|
40,000
|
||
|
Yiu-Chun
Chan
|
40,000
|
40,000
|
||
|
Lei
Lin
|
40,000
|
40,000
|
||
|
Ka-Keung
Yeung(1)
|
10,000
|
10,000
|
|
(1)
|
Ka-Keung
Yeung was elected to our board of directors on January 8,
2010.
|
Non-Employee
Director Compensation
Currently,
each of our non-employee directors is entitled to receive an annual cash
retainer of $40,000 for his or her services as a director. In addition, our
non-employee directors are reimbursed for travel, lodging and other reasonable
out-of-pocket expenses incurred in attending meetings of the board and board
committees. Our non-employee directors do not receive any equity-based awards or
other compensation for their service as directors.
Compensation
Committee Interlocks and Insider Participation
Jinrong
Chen, Yiu-Chun Chan, Lei Lin and Ka-Keung Yeung each served on the compensation
committee during the fiscal year ended March 31, 2010. None of these directors
is or was an executive officer of our company or had any relationships requiring
disclosure by us under the SEC’s rules requiring disclosure of certain
relationships and related-party transactions. None of our executive officers
served as a director or a member of a compensation committee (or other committee
serving an equivalent function) of any other entity, the executive officers of
which served as a director or member of our compensation committee during the
fiscal year ended March 31, 2010.
ITEM
12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
AND RELATED STOCKHOLDER MATTERS
Security
Ownership of Certain Beneficial Owners
The
following table sets forth information, as of June 1, 2010, regarding the
beneficial ownership of our common stock by:
|
·
|
each
person known by us to be a beneficial owner of more than five percent of
our outstanding common stock;
|
|
·
|
each
of our directors and director
nominees;
|
|
·
|
each
of our named executive officers;
and
|
|
·
|
all
directors and executive officers as a
group.
|
The
amounts and percentage of common stock beneficially owned are reported on the
basis of regulations of the SEC governing the determination of beneficial
ownership of securities. Under the rules of the SEC, a person is deemed to be a
“beneficial owner” of a security if that person has or shares “voting power,”
which includes the power to vote or to direct the voting of such security, or
“investment power,” which includes the power to dispose of or to direct the
disposition of such security. A person is also deemed to be a beneficial owner
of any securities of which that person has a right to acquire beneficial
ownership within 60 days after June 1, 2010. Under these rules, more than one
person may be deemed a beneficial owner of the same securities and a person may
be deemed a beneficial owner of securities as to which he has no economic
interest. Except as indicated by footnote, the persons named in the table below
have sole voting and investment power with respect to all shares of common stock
shown as beneficially owned by them.
Percentage
of class is based on 54,000,713 shares of common stock outstanding as of June 1,
2010. Unless otherwise noted below, the address of the persons listed on the
table is 2275 Research Blvd., Suite 500, Rockville, Maryland 20850.
108
|
Shares
Beneficially Owned
|
|||||||||
|
Name
|
Number
|
Percent
|
|||||||
|
OFFICERS
AND DIRECTORS
|
|||||||||
|
Liang
Zhang, Director and Chief Executive Officer(1)
|
36,000,000 | 66.67% | |||||||
|
Joseph
Chow, Interim Chief Financial Officer and Director
|
— | — | |||||||
|
Lawrence
Lee, Former Chief Financial Officer
|
— | — | |||||||
|
Weiguo
Zhang, President and Chief Operating Officer
|
2,000 | * | |||||||
|
Donghao
Yang, Chief Financial Officer
|
— | — | |||||||
|
David
Hui Li, Director(2)
|
4,000,000 | 7.41% | |||||||
|
Jinrong
Chen, Director
|
— | — | |||||||
|
Yiu-Chun
Chan, Director
|
— | — | |||||||
|
Lei
Lin, Director
|
— | — | |||||||
|
Ka-Keung
Yeung, Director
|
— | — | |||||||
|
All
Officers and Directors as a Group
|
40,002,000 | 74.08% | |||||||
|
PRINCIPAL
STOCKHOLDER
|
|||||||||
|
Warburg
Pincus Private Equity IX, L.P.(3)
|
4,000,000 | 7.41% | |||||||
|
*
|
Less
than 1%
|
|
(1)
|
This
amount includes 36,000,000 shares owned by Beams Power Investment Limited,
or Beams, a British Virgin Islands company. In addition, Beams had issued
a senior convertible note dated April 23, 2008, to the Warburg Pincus
Entities, as defined below, convertible into up to 1,000,000 shares of
common stock held by Beams. Liang Zhang has dispositive and voting power
over investments by Beams. Liang Zhang’s wife, Xiuqing Meng, is the sole
shareholder and director of Beams. Of the shares held by Beams, 5,967,000
shares were pledged to Warburg (as defined below) pursuant to a share
pledge agreement, dated April 23, 2008 and an additional 8,000,000 shares
were pledged to Warburg on December 4, 2008, see “Item 13. Certain
Relationships and Related Transactions, and Director
Independence.”
|
|
(2)
|
Mr.
David Hui Li is a Managing Director and Member of WP LLC. All shares
indicated as owned by Mr. Li are included because of his affiliation with
the Warburg Pincus entities. Joseph P. Landy and Charles R. Kaye are
Managing General Partners of WP and Managing Members and Co-Presidents of
WP LLC and may be deemed to control the Warburg Pincus entities.
Messrs. Kaye, Landy, and Li disclaim beneficial ownership of all shares
held by the Warburg Pincus
entities.
|
|
(3)
|
According
to a Schedule 13D/A filed by Warburg Pincus Private Equity IX, L.P., or
Warburg, with the SEC on September 1, 2008, Warburg Pincus IX LLC, or WP
IX LLC, is the sole general partner of Warburg. Warburg Pincus Partners
LLC, or WP Partners, is the sole managing member of WP IX LLC, and Warburg
Pincus & Co., or WP, is the sole managing member of WP Partners.
Charles R. Kaye and Joseph P. Landy are general partners of WP and
managing members and co-presidents of Warburg Pincus LLC, or WP LLC, which
manages Warburg. WP, WP Partners, WP IX LLC, Warburg and WP LLC are
collectively referred to as the “Warburg Pincus Entities.” Messrs. Kaye
and Landy may be deemed to indirectly beneficially own the shares held by
Warburg because of their affiliation with the Warburg Pincus Entities.
Messrs. Kaye and Landy disclaim beneficial ownership of the shares held by
Warburg except to the extent of their pecuniary interest therein. The
address of the Warburg Pincus Entities is 466 Lexington Avenue, New York,
New York, 10017. This amount represents the shares of common stock held
directly by the Warburg Pincus Entities. In addition, there are 1,000,000
shares of common stock issuable to the Warburg Pincus Entities pursuant to
the senior convertible note, dated April 23, 2008, issued by Beams Power
Investment Limited.
|
Securities
Authorized for Issuance Under Equity Compensation Plans
We
currently have a 2008 Stock Incentive Plan which has been approved by our
shareholders. The plan will terminate on June 10, 2018. To date, we have not
issued any awards under the Plan.
109
The
following table sets forth the number of common shares remaining available for
future issuance under the plan as of March 31, 2010.
|
Plan
Category
|
Number
of Common Shares to Be Issued Upon Exercise of Outstanding Options,
Warrants
and
Rights
|
Weighted-Average
Exercise Price of Outstanding Options, Warrants and
Rights
|
Number
of Common Shares Remaining Available for Future Issuance Under Equity
Compensation Plans (Excluding Shares Reflected in
the
First Column)
|
|||||||||
|
Equity
compensation plans approved by shareholders
|
— | — | 12,500,000 | |||||||||
|
Equity
compensation plans not approved by shareholders
|
— | — | — | |||||||||
|
Total
|
— | — | 12,500,000 | |||||||||
ITEM
13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR
INDEPENDENCE
Transactions
with Affiliate Companies controlled by Liang Zhang
Our
chairman, chief executive officer and principal stockholder, Liang Zhang,
controls several other companies in China. Among these companies, we sold whey
protein to Honnete and powdered formula products to St. Angel (Beijing) Business
Service, and we also purchased whey protein powders from Kelqin and Honnete, and
catalogues, brochures, and marketing materials from St. Angel Cultural
Communications for the fiscal years ended March 31, 2010 and 2009.
For the
fiscal year ended March 31, 2010, we purchased $98,000 of whey protein powder
from Honnete.
The
following table sets forth the value of our sales to our related parties for the
fiscal year ended March 31, 2010 and 2009:
|
Year
Ended March 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Beijing
Honnete Dairy Co., Ltd.
|
$ | 8,830 | $ | 2,255 | ||||
|
St.
Angel (Beijing) Business Service Co., Ltd.
|
824 | — | ||||||
|
Total
|
$ | 9,654 | $ | 2,255 | ||||
The
following table sets forth the value of our purchases from our related parties
for the fiscal year ended March 31, 2010 and 2009:
|
Year
Ended March 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Beijing
Honnete Dairy Co., Ltd.
|
$ | 98 | $ | 10,376 | ||||
|
Beijing
Kelqin Dairy Co., Ltd.
|
— | 154 | ||||||
|
Beijing
St. Angel Cultural Communication Co. Ltd..
|
470 | 1,639 | ||||||
|
Total
|
$ | 568 | $ | 12,169 | ||||
In June
2008, we sold two commercial buildings to Sheng Zhi Da Dairy Group Corporation,
an entity 100% controlled by the Liang Zhang, at the carrying value of $1.7
million. The amount due from Sheng Zhi Da Dairy Group Corporation as of March
31, 2010 and 2009 represents the receivable for the sales of the two
buildings.
110
We had
indebtedness from related parties controlled by Liang Zhang. The following table
sets forth the amount of indebtedness principal outstanding during the fiscal
years ended March 31, 2010 and 2009:
|
Year
Ended March 31,
|
||||||||
|
2010
|
2009
|
|||||||
|
(In
thousands)
|
||||||||
|
Balance
at beginning of the year.
|
$ | 7,523 | $ | — | ||||
|
Borrowing
from related parties
|
4,798 | 8,255 | ||||||
|
Repayment
to related parties
|
(8,461 | ) | (732 | ) | ||||
|
Balance
at end of the year
|
$ | 3,860 | $ | 7,523 | ||||
As of
June 1, 2010, the short term loan from related parties is $3.9 million. The
interest paid for the fiscal year ended March 31, 2010 and 2009 was $197,000 and
nil respectively. The interest rate for the loan outstanding at March 31, 2010
was 10.0%. The interest payable at March 31, 2010 was $512,000.
Registration
Rights Agreement with Warburg
On June
15, 2007, we issued 4,000,000 shares of common stock to Warburg for $66.0
million and entered into a registration rights agreement with Warburg. Pursuant
to this registration rights agreement, we granted Warburg certain customary
registration rights, including demand, piggyback and Form S-3 registration
rights. David Li, who is one of our directors, is a managing director of
Warburg. Subject to the provisions of the registration rights agreement and the
restrictions of its lock-up agreement, Warburg will be entitled to require us to
register the resale of its shares under the Securities Act.
On April
23, 2008, Beams Power Investment Limited, or Beams, a British Virgin Islands
limited liability company and our majority stockholder, and Warburg entered into
a note purchase agreement. Pursuant to this note purchase agreement, Beams
issued Warburg a senior convertible note in an aggregate principal amount of $30
million, which is convertible into up to 1,000,000 shares of our common stock
held by Beams. In addition, Beams and Warburg entered into a share pledge
agreement, dated April 23, 2008, pursuant to which Beams agreed to pledge an
aggregate of 5,967,000 of shares of our common stock held by Beams as initial
collateral for the loan covered by the note. On December 4, 2008, Beams agreed
to pledge an additional 8,000,000 shares of our common stock held by Beams as
collateral for the loan covered by the note.
In
connection with the issuance of the note and the share pledge agreement, we
entered into a registration rights agreement, dated April 23, 2008, with Beams
and Warburg, pursuant to which we granted Warburg certain customary registration
rights, including demand, piggyback and Form S-3 registration rights, with
respect to the shares of our common stock Warburg acquires or may acquire
pursuant to the note purchase agreement, the note and the share pledge
agreement.
Control
Agreements with Affiliated Entities
In order
to comply with PRC law and avoid restrictions on foreign investment in medical
clinical operations, we operate our medical treatment services (mostly pre-natal
diagnostics services) through four entities -- Nanjing Shengyuan Huiren Clinical
Examination Co., Ltd., Taiyuan Shengyuan Huiren Clinical Examination Co., Ltd,
Shijiazhuang Shengyuan Huiren Clinical Examination Co., Ltd and Heilongjiang
Shengyuan Huiren Clinical Examination Co., Ltd (the “Four Entities”) that are
not directly owned by us. We control and consolidate these entities into our
group consolidated results through a series of contractual arrangements which
are summarized below.
(a) Exclusive
Consulting and Service Agreement entered into by and between Shengyuan
Nutritional Food Co., Ltd. (“Nutritional”) and Beijing Shengyuan Huimin
Technology Service Co., Ltd. (“Huimin”). Pursuant to this agreement, Nutritional
agrees to provide to Huimin certain consulting services for which Huimin shall
pay Nutritional a service fee in an amount equal to 10%-50% of Huimin’s monthly
net sale, the exact percentage to be determined by the two parties. The term of
the agreement is 10 years unless Nutritional terminates it before its expiration
and Nutritional shall have the right to extend the Agreement before it
expires.
111
(b) Business
Operating Agreement entered into by and among Nutritional, Huimin, Zhang Jibin
(who is our Director of Loans) and Jiang Yunpeng (who is our Director of
Strategic Acquisitions). Pursuant to this agreement, Huimin agrees to conduct
its business in a way that is consistent with Nutritional’s direction. In
addition, Zhang Jibin and Jiang Yunpeng authorize Nutritional to exercise all of
their respective shareholders’ rights in Humin.
(c) Call
Option Agreement entered into by and among Nutritional, Huimin, Zhang Jibin and
Jiang Yunpeng. Pursuant to this agreement, within a ten-year period and any
extended period as requested by Nutritional, Nutritional is entitled to an
irrevocable and exclusive right to purchase or authorize any third party to
purchase the shares of Huimin held by Zhang Jibin and Jiang Yunpeng at the
lowest feasible price according to PRC laws and regulations. In addition,
Nutritional is entitled to call part or all of the option right at any time
during the effective period of this agreement and there is no limit on the
number of calls until Nutritional hold 100% of Huimin’s shares.
(d) Pledge
Agreement entered into by and among Nutritional, Zhang Jibin and Jiang Yunpeng.
To ensure the performance of the Exclusive Consulting and Service Agreement,
Call Option Agreement and Business Operating Agreement, Zhang Jibin and Jiang
Yunpeng, as pledgers under this agreement, pledge all of their shares and
corresponding interests and rights in Huimin to Nutritional, as pledgee under
this agreement.
(e) Entrustment
Agreement entered into by and among Nutritional, Zhang Jibin and Jiang Yunpeng.
Pursuant to this agreement, Nutritional loans Zhang Jibin and Jiang Yunpeng each
RMB15,000,000 to establish companies for pre-natal diagnostic services in China.
Zhang Jibin and Jiang Yunpeng are the nominal shareholders of these companies
and Nutritional is the actual shareholder of these companies.
(f) Entrustment
Agreement entered into by and among Nutritional, Zhang Jibin, Jiang Yunpeng and
Honnete. Nutritional agrees to entrust Zhang Jibin and Jiang Yunpeng arty A) to
establish a limited liability company (the “future company”) and Zhang Jibin and
Jiang Yunpeng will hold all of the shares of the future company in entrustment.
Zhang Jibin and Jiang Yunpeng have established Nanjing Huiren, Taiyuan Huiren,
Shijiazhuang Huiren and Haerbin Huiren (the “established companies”). Upon the
fulfillment of the registration of the said future company, Zhang Jibin and
Jiang Yunpeng will transfer their shares in the established companies to the
future company for free. Upon approval from Nutritional, Zhang Jibin and Jiang
Yunpeng will entrust Honnete to hold the shares of the established
companies.
Review,
Approval or Ratification of Transactions with Related Parties
Audit
committee must approve any new class of transactions involving the Company in
which any of our directors, director nominees, executive officers, greater than
five percent beneficial owners and their respective immediate family members has
a direct or indirect material interest. In doing so, the Board takes into
account, among other factors it deems appropriate:
|
·
|
The
related person’s interest in the
transaction;
|
|
·
|
The
approximate dollar value of the amount involved in the
transaction;
|
|
·
|
The
approximate dollar value of the amount of the related person’s interest in
the transaction without regard to the amount of any profit or
loss;
|
|
·
|
Whether
the transaction was undertaken in the ordinary course of our
business;
|
|
·
|
Whether
the transaction with the related person is proposed to be, or was, entered
into on terms no less favorable to us than terms that could have been
reached with an unrelated third
party;
|
|
·
|
The
purpose of, and the potential benefits to us of, the transaction;
and
|
|
·
|
Any
other information regarding the transaction or the related person in the
context of the proposed transaction that would be material to investors in
light of the circumstances of the particular
transaction.
|
112
Director
Independence
The Board
has determined all Board members, excluding Liang Zhang, Joseph Chow and David
Hui Li, are independent under the applicable NASDAQ rules. The Board has also
determined the members of each committee of the Board are independent under the
listing standards of the NASDAQ Global Select Market. In making these
determinations, the Board considered, among other things, the types and amounts
of the commercial dealings between the Company and the companies and
organizations with which the directors are affiliated.
ITEM
14. PRINCIPAL ACCOUNTING FEES AND SERVICES
On July
27, 2007, we engaged Deloitte Touche Tohmatsu CPA Ltd. (“DTTC”) as our new
independent auditors. The engagement was approved by the Audit Committee of our
Board of Directors. No relationship existed in any manner between DTTC and us
prior to the date we engaged DTTC.
Audit
Fees
DTTC was
paid aggregate fees of approximately $797,812 and $1,202,520 for the fiscal year
ended March 31, 2010 and 2009, respectively, for professional services rendered
for the audit of our annual financial statements and for the reviews of the
financial statements included in our quarterly reports on Form
10-Q.
Audit-related
Fees
DTTC was
paid aggregate fees of approximately $60,000 and nil for the fiscal year ended
March 31, 2010 and 2009, respectively, for assurance and related services
reasonably related to the performance of the audit or review of the Registrant’s
financial statements.
Tax
Fees
DTTC was
paid aggregate fees of approximately $nil and $13,000 for the fiscal year ended
March 31, 2010 and 2009, respectively, for professional services rendered for
tax compliance, tax advice and tax planning.
All
Other Fees
DTTC was
paid no other fees for any other services rendered to the Company for the fiscal
year ended March 31, 2010 and 2009.
Audit
Committee Pre-approval Policies and Procedures
Our Audit
Committee’s policy is to pre-approve all audit and permissible non-audit
services provided by our independent auditors. These services may include audit
services, audit-related services, tax services and other services. Pre-approval
is generally provided for up to one year and any pre-approval is detailed as to
the particular service or category of services. The independent auditor and
management are required to periodically report to the Audit Committee regarding
the extent of services provided by the independent auditor in accordance with
this pre-approval.
PART
IV
ITEM
15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a) List
of Documents Filed as a Part of this Annual Report on Form 10-K:
1. See
Index to Consolidated Financial Statements in Item 8 of this
Report.
2. See
Index to Consolidated Financial Statements in Item 8 of this Report, and the
Consolidated Financial Schedules listed on such index filed as part of this
Report are filed as part of this Report.
113
3. The
exhibits listed on the exhibit index filed as part of this Report are filed or
incorporated by reference as part of this Report
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the Registrant has
duly caused this report to be signed on its behalf by the undersigned thereunto
duly authorized.
|
SYNUTRA
INTERNATIONAL, INC.
|
||||||
|
Date:
|
June
9, 2010
|
By:
|
/s/
Liang Zhang
|
|||
|
Name:
|
Liang
Zhang
|
|||||
|
Title:
|
Chief
Executive Officer and Chairman
|
|||||
Each
person whose signature appears below appoints each of Liang Zhang and Joseph
Chow, severally and not jointly, to be his true and lawful attorney-in-fact and
agent, with full power of substitution and resubstitution, for him and in his
name, place and stead, in any and all capacities, to sign any amendments to our
Annual Report on Form 10-K for the fiscal year ended March 31, 2010; granting
unto said attorney-in-fact and agent, full power and authority to do and perform
each and every act and thing requisite and necessary to be done, as fully for
all intents and purposes as he might or could do in person, hereby ratifying and
confirming all that said attorney-in-fact and agent, or his substitute or
substitutes, shall lawfully do or cause to be done by virtue
hereof.
Pursuant
to the requirements of the Securities Exchange Act of 1934, this Annual Report
on Form 10-K has been signed below by the following persons on behalf of the
Registrant and in the capacities.
|
Name
|
Position
|
Date
|
||
|
/s/
Liang Zhang
|
||||
|
Liang
Zhang
|
Chief
Executive Officer, and Chairman (Principal Executive
Officer)
|
June
9, 2010
|
||
|
/s/
Joseph Chow
|
||||
|
Joseph
Chow
|
Interim
Chief Financial Officer and Director (Principal Financial and Accounting
Officer)
|
June
9, 2010
|
||
|
/s/
David Hui Li
|
||||
|
David
Hui Li
|
Director
|
June
9, 2010
|
||
|
/s/
Jinrong Chen
|
||||
|
Jinrong
Chen
|
Director
|
June
9, 2010
|
||
|
/s/
Yiu-Chun Chan
|
||||
|
Yiu-Chun
Chan
|
Director
|
June
9, 2010
|
||
|
/s/
Lei Lin
|
||||
|
Lei
Lin
|
Director
|
June
9, 2010
|
||
|
/s/
Ka-Keung Yeung
|
||||
|
Ka-Keung
Yeung
|
Director
|
June
9, 2010
|
114
EXHIBITS
INDEX
|
Exhibit
Number
|
Description
|
|
|
3.1
|
Amended
and Restated Certificate of Incorporation of Synutra International, Inc.
(incorporated by reference to Exhibit 3.1 to the Registrant’s Quarterly
Report on Form 10-Q filed on November 11, 2008)
|
|
|
3.2
|
Amended
and Restated Bylaws of Synutra International, Inc. (incorporated by
reference to Exhibit 3.2 to the Registrant’s Quarterly Report on Form 10-Q
filed on November 11, 2008)
|
|
|
4.1
|
Specimen
common stock certificate (incorporated by reference to Exhibit 4.1 to the
Registrant’s Annual Report on Form 10-K filed on June 16,
2008)
|
|
|
10.1
|
Employment
Agreement between Shengyuan Nutritional Food and Liang Zhang, dated
December 21, 2008
|
|
|
10.2
|
Employment
Agreement between Shengyuan Nutritional and Weiguo Zhang, dated June 4,
2010
|
|
|
10.3
|
Employment
Agreement between the Shengyuan Nutritional Food Co., Ltd. and Joseph
Chow, dated November 10, 2009.
|
|
|
10.4
|
Employment
Agreement between Sheng Yuan Nutritional Food Co., Ltd., Beijing R&D
Center and Donghao Yang, dated May 4, 2010
|
|
|
10.5
|
Warrant
Agreement among Beams Power Investment Limited, Synutra International,
Inc., the Bank of New York as Warrant Agent, and ABN AMRO N.V., Hong Kong
Branch, dated April 19, 2007 (incorporated by reference to Exhibit 4.1 to
the Registrant’s Current Report on Form 8-K filed on April 24,
2007)
|
|
|
10.6
|
Registration
Rights Agreement between Synutra International, Inc. and ABN AMRO N.V.,
Hong Kong Branch, dated April 19, 2007 (incorporated by reference to
Exhibit 4.3 to the Registrant’s Current Report on Form 8-K filed on April
24, 2007)
|
|
|
10.7
|
Common
Stock Purchase Agreement between Synutra International, Inc. and Warburg
Pincus Private Equity IX, L.P. , dated May 24, 2007 (incorporated by
reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K/A
filed on June 1, 2007)
|
|
|
10.8
|
Registration
Rights Agreement between Synutra International, Inc. and Warburg Pincus
Private Equity IX, L.P., dated June 15, 2007 (incorporated by reference to
Exhibit 10.1 to the Registrant’s Current Report on Form 8-K/A filed on
June 1, 2007)
|
|
|
10.9
|
Voting
and Co-Sale Agreement among Synutra International, Inc., Beams Power
Investment Limited, and Warburg Pincus Private Equity IX, L.P., dated June
15, 2007 (incorporated by reference to Exhibit 10.1 to the Registrant’s
Current Report on Form 8-K/A filed on June 1, 2007)
|
|
|
10.10
|
Registration
Rights Agreement among Synutra International, Inc., Beams Power Investment
Limited and Warburg Pincus Private Equity IX, L.P., dated April 23, 2008,
(incorporated by reference to Exhibit 4.1 to the Registrant’s Current
Report on Form 8-K filed on April 24, 2008)
|
|
|
10.11
|
Amended
Loan Agreement among Synutra International, Inc., ABN AMRO N.V. and
certain lenders party thereto, dated February 26, 2010 (incorporated by
reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K
filed on March 4, 2010)
|
|
|
10.12
|
Collateral
Agreement among Synutra International, Inc., ABN AMRO N.V. and Synutra,
Inc., dated October 11, 2007 (incorporated by reference to Exhibit 10.2 to
the Registrant’s Current Report on Form 8-K filed on October 17,
2007).
|
|
|
10.13
|
US
Dollar Facility Fee Letter Agreement between Synutra International, Inc.
and The Royal Bank of Scotland N.V., dated October 11, 2007 (incorporated
by reference to Exhibit 10.3 to the Registrant’s Current Report on Form
8-K filed on October 17, 2007).
|
|
|
10.14
|
Synutra
International, Inc. 2008 Stock Incentive Plan (incorporated by reference
to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed on
June 17, 2008)
|
115
|
Exhibit
Number
|
Description
|
|
|
10.15A
|
Form
of Incentive Stock Option Agreement under Synutra International, Inc. 2008
Stock Incentive Plan (incorporated by reference to Exhibit 10.16A to the
Registrant’s Annual Report on Form 10-K filed on June 15,
2009)
|
|
|
10.15B
|
Form
of Nonqualified Stock Option Agreement under Synutra International, Inc.,
2008 Stock Incentive Plan (incorporated by reference to Exhibit 10.16B to
the Registrant’s Annual Report on Form 10-K filed on June 15,
2009)
|
|
|
10.16
|
Asset
Purchase Agreement, dated July 14, 2008, between Sheng Yuan Nutritional
Food Co., Ltd. and Beijing Huilian Food Co, Ltd. (incorporated by
reference to Exhibit 2.1 from the Registrant’s Current Report on Form 8-K
filed on July 18, 2008)
|
|
|
10.17
|
Exclusive
Consulting and Service Agreement entered into by and between Shengyuan
Nutritional Food Co., Ltd. and Beijing Shengyuan Huimin Technology Service
Co., Ltd. dated July 20, 2008
|
|
|
10.18
|
Business
Operating Agreement entered into by and among Shengyuan Nutritional Food
Co., Ltd., Beijing Shengyuan Huimin Technology Service Co., Ltd. and Zhang
Jibin and Jiang Yunpeng dated July 20, 2008
|
|
|
10.19
|
Equity
Disposal Agreement entered into by and among Shengyuan Nutritional
Food Co., Ltd., Beijing Shengyuan Huimin Technology Service Co., Ltd. and
Zhang Jibin and Jiang Yunpeng dated July 20, 2008
|
|
|
10.20
|
Share
Pledge Agreement entered into by and among Shengyuan Nutritional Food Co.,
Ltd. and Zhang Jibin and Jiang Yunpeng dated July 20,
2008
|
|
|
10.21
|
Entrustment
Agreement entered into by and among Shengyuan Nutritional Food Co., Ltd.
and Zhang Jibin and Jiang Yunpeng dated July 20, 2008
|
|
|
10.22
|
Entrustment
Agreement entered into by and among Shengyuan Nutritional Food Co., Ltd.,
Zhang Jibin and Jiang Yunpeng and Beijing Honnete Dairy Co., Ltd. dated
July 20, 2008
|
|
|
10.23
|
Asset
Purchase Agreement dated as of September 9, 2009 among Heilongjiang
Baoquanling Shengyuan Dairy Co., Ltd., Heilongjiang Baoquanling Shengyuan
Dairy Cow Breeding Co., Ltd. and Heilongjiang Wondersun Dairy Co., Ltd.
(incorporated by reference to Exhibit 2.1 from the Registrant’s Current
Report on Form 8-K filed on September 15, 2009)
|
|
|
12.1
|
Statements
re computation of ratios
|
|
|
14.1
|
Code
of Ethics for Chief Executive Officer and Senior Financial Officers
(incorporated by reference to Exhibit 14. 1 to the Registrant’s Annual
Report on Form 10-K filed on June 16, 2008)
|
|
|
16.1
|
Letter
from Rotenberg & Co to the Securities and Exchange Commission dated
July 24, 2007 (incorporated by reference to Exhibit 16.1 to the
Registrant’s Current Report on Form 8-K filed on July 27,
2007)
|
|
|
21.1
|
List
of Subsidiaries
|
|
|
23.1
|
Consent
of Deloitte Touche Tohmatsu CPA Ltd.
|
|
|
24.1
|
Power
of Attorney (included on the Signature Page of this Annual Report on Form
10-K)
|
|
|
31.1
|
Certification
of Principal Executive Officer pursuant to Rule 13a-14(a) and Rule
15d-14(a), promulgated under the Securities and Exchange Act of 1934, as
amended
|
|
|
31.2
|
Certification
of Principal Financial Officer pursuant to Rule 13a-14(a) and Rule
15d-14(a), promulgated under the Securities and Exchange Act of 1934, as
amended
|
|
|
32.1
|
Certification
pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of
the Sarbanes-Oxley Act of 2002 (Chief Executive
Officer)
|
|
|
32.2
|
Certification
pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of
the Sarbanes-Oxley Act of 2002 (Chief Financial
Officer)
|
116
