Attached files
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON,
D.C. 20549
FORM
10-K
[X] Annual
Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of
1934
For the
Fiscal Year Ended December 31, 2009
[ ] Transition
Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of
1934
Commission
File No. 0-11550
Pharmos
Corporation
(Exact
name of registrant as specified in its charter)
|
Nevada
|
36-3207413
|
|
(State
or other jurisdiction of
|
(IRS
Employer Id. No.)
|
|
incorporation
or organization)
|
|
99
Wood Avenue South, Suite 311
Iselin,
NJ 08830
(Address
of principal executive offices) (zip code)
Registrant’s
telephone number, including area code: (732) 452-9556
Securities
registered pursuant to Section 12(b) of the Act:
None
(Title of
Class)
Securities
registered pursuant to Section 12(g) of the Act:
Common
Stock, $.03 par value
(Title of
Class)
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in
Rule 405 of the Securities Act. [ ]
Yes [X] No
Indicate
by check mark if the registrant is not required to file reports pursuant to
Section 13 or Section 15(d) of the Act. [ ]
Yes [X] No
Indicate
by check mark whether the registrant (1) has filed all reports required to be
filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the
preceding 12 months (or for such shorter period that the registrant was required
to file such reports), and (2) has been subject to such filing requirements for
the past 90 days. [X] Yes [ ]
No
Indicate
by check mark whether the registrant has submitted electronically and posted on
its corporate website, if any, every Interactive Data File required to be
submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding
12 months (or for such shorter period that the registrant was required to submit
and post such files). [ ]
Yes [ ] No
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of
Regulation S-K is not contained herein, and will not be contained, to the best
of registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this
Form 10-K. [X]
Indicate
by check mark whether registrant is a large accelerated filer, an accelerated
filer, a non-accelerated filer or a smaller reporting company (as defined in
Rule 12b-2 of the Act).
Large accelerated filer
[ ] Accelerated
filer [ ]
Non-accelerated filer
[ ] Smaller
reporting company [X]
Indicate
by check mark whether the registrant is a shell company (as defined in Rule
12b-2 of the Act).
[ ]
Yes [X] No
The
aggregate market value of the registrant’s Common Stock at June 30, 2009 held by
those persons deemed to be non-affiliates was approximately
$5,317,592.
As of
February 22, 2010, the Registrant had outstanding 59,438,213 shares of its $.03
par value Common Stock.
PART
I
This
report contains information that may constitute "forward-looking
statements." The use of words such as "believe," "expect," "intend,"
"estimate," "anticipate," "project," "will" and similar expressions identify
forward-looking statements, which generally are not historical in
nature. All statements that address operating performance, events or
developments that we expect or anticipate will occur in the future are
forward-looking statements. As and when made, we believe that these
forward-looking statements are reasonable. However, caution should be
taken not to place undue reliance on any such forward-looking statements because
such statements speak only as of the date when made. We undertake no
obligation to publicly update or revise any forward-looking statements, whether
as a result of new information, future events or otherwise. In
addition, forward-looking statements are subject to certain risks and
uncertainties that could cause actual results to differ materially from our
company's historical experience and our present expectations or
projections. These risks and uncertainties include, but are not
limited to, those described in Part I, "Item 1A. Risk Factors" and
elsewhere in this report and those described from time to time in our future
reports filed with the Securities and Exchange Commission.
We
do not undertake to discuss matters relating to our ongoing clinical trials or
our regulatory strategies beyond those which have already been made public or
discussed herein.
Item
1. Business
OVERVIEW
OF BUSINESS
1. The
Company
Pharmos
Corporation (the Company or Pharmos) is a biopharmaceutical company that
discovers and develops novel therapeutics to treat a range of diseases of the
nervous system, including disorders of the brain-gut axis (e.g., Irritable Bowel
Syndrome), with a focus on pain/inflammation, and autoimmune disorders. The
Company's most advanced product is Dextofisopam for the treatment of irritable
bowel syndrome (IBS). IBS is a chronic and sometimes debilitating condition that
affects roughly 10%-15% of U.S. adults and is two to three times more prevalent
in women than in men. With an absence of safe and effective therapies,
Dextofisopam’s novel non-serotonergic activity holds the potential for a unique
and innovative treatment approach to IBS with diarrhea predominant and
alternating patients.
On
September 14, 2009, the Company announced the results of its Phase 2b
Dextofisopam clinical trial to evaluate safety and efficacy of the compound in
irritable bowel syndrome.
Although
the primary efficacy variable (% of weeks responding for adequate overall relief
of IBS symptoms) did not reach statistical significance, the percentage
responding for the Dextofisopam 200 mg group was higher than that observed for
the Phase 2a trial. However, the placebo response rate was also higher than
expected compared to the Phase 2a placebo response.
This
result was similarly demonstrated across all other secondary efficacy variables
associated with the adequate overall relief question. In all cases except in the
first month, the response rates for the Dextofisopam 200 mg group were
essentially the same as or in most cases better than the response rates observed
for the Phase 2a trial.
Also,
secondary response variables of adequate relief of abdominal pain and discomfort
and overall IBS symptoms ratings showed statistical significance and trends
favoring the Dextofisopam 200 mg group compared to placebo.
1
The Phase
2b double-blind, randomized, placebo-controlled study evaluated the clinical
safety, tolerability and efficacy of multiple doses of Dextofisopam. Female
outpatients with diarrhea predominant and alternating diarrhea and constipation
IBS (according to Rome III Criteria for IBS) were randomized into each of four
treatment groups: 100mg, 200mg, and 300mg twice a day (BID) Dextofisopam or
placebo. A total of 324 patients were enrolled using approximately 70 US
centers. The patients participated for up to 19 weeks, including a screening
period, a 12-week treatment period, and a 28-day post treatment period. The
primary endpoint was “adequate overall relief” of IBS symptoms during the
12-week treatment period.
In the
Phase 2b study, of the 324 patients randomized, 311 (96%) were evaluated in the
Intent to Treat (ITT) analysis. Patients who completed the full study numbered
225 (69%), a similar percentage to the Phase 2a study, and 99 (31%) patients
discontinued the study. The majority of patients who discontinued did so due to
withdrawal of consent or a non serious adverse event.
Demographics
and baseline calculations were comparable among the four treatment groups. The
mean age was between 44 and 45 years of age. The duration of IBS disease
symptoms ranged from 9 to 11 years, and the majority of patients were
white.
The most
bothersome IBS symptom reported at baseline was abdominal pain and discomfort,
followed by a sense of urgency and stool frequency.
This
Phase 2b study followed a successful 141-patient Phase 2a study in which
Dextofisopam with a dose of 200 mg BID demonstrated a statistically significant
improvement over placebo on the primary endpoint of adequate overall relief (
p=0.033). In the Phase 2a trial, Dextofisopam was well tolerated and did not
cause significant constipation. Dextofisopam also provided benefit on a variety
of secondary endpoints. Importantly, the beneficial effects on stool frequency
were observable after two days and maintained for 12 weeks of treatment.
Overall, similar rates of adverse events were seen with Dextofisopam and
placebo. Diarrhea and constipation as adverse events occurred at a very low
rate.
Additionally,
Dextofisopam has been evaluated in three randomized, double-blind,
placebo-controlled Phase 1 trials comprising healthy volunteers. Clinical data
from the Phase 1 studies demonstrated that Dextofisopam appears to be safe and
well tolerated at doses up to 600 mg BID.
The
Company’s strategy is to seek a pharmaceutical partner with the appropriate GI
clinical and scientific expertise for further development of
Dextofisopam. On October 21, 2009 the Company announced that it had
engaged Cowen and Company as advisors to assist with accelerating a partnership
arrangement for Dextofisopam.
The
Company’s primary focus is on the development and partnering of Dextofisopam and
to that end cash resources are being conserved and targeted for that program. On
August 29, 2008 the Company announced that effective October 31, 2008 it would
cease operations in Rehovot, Israel, and manage those activities out of the
Company’s US headquarters in Iselin, New Jersey. The Company’s subsidiary in
Israel, Pharmos Ltd. is being voluntarily liquidated. The Company
expects this to be completed by March 31, 2010.
Pharmos’
cannabinoid research was geared toward development of selective and specific CB2
receptor agonists. By activating CB2 receptors, CB2 agonists inhibit autoimmune
and inflammatory processes, and are likely to be useful for treating pain,
autoimmune, inflammatory and degenerative disorders. Although
progress has been made, the early stage of this work and resource limitations
have resulted in termination of these programs. Pharmos’ strategy is
to sell or out license the technology developed around the cannabinoid research.
Pharmos
had developed these compounds in preclinical testing for neuropathic
pain.
2
In
November 2009 the Company entered into a Material Transfer Agreement whereby a
European company will perform certain experiments with samples of our synthetic
selective cannabinoid receptor CB2 agonist.
Tianeptine,
a potential follow-on product to Dextofisopam, has completed late-preclinical
development for the treatment of irritable bowel syndrome (IBS). Tianeptine, a
racemic molecule, has been marketed outside the United States since 1988 for the
treatment of depression. Preclinical studies support the potential utility of
Tianeptine for the treatment of functional gastrointestinal disorders and, in
particular, IBS. Pharmos has established patent rights for the use of Tianeptine
and its enantiomers for the treatment of IBS and functional dyspepsia.
Tianeptine is available for out-licensing.
The
Company owns the rights to both R and S Tofisopam. Dextofisopam is the R
enantiomer of racemic tofisopam and is being developed for IBS as described
above. In December 2009 the Company entered into a Material Transfer Agreement
with a US based company to perform certain experiments with
S-Tofisopam.
On March
13, 2009 the Company was officially delisted from the Nasdaq Capital Market, and
is currently trading on the OTCBB pink sheets under the symbol “PARS.PK”. The
Company was not in compliance with the minimum $2,500,000 stockholders’ equity
requirement for continued listing and was unable to comply during the grace
period extended by NASDAQ. As a result of trading on the OTCBB pink sheets,
liquidity for the Company’s common stock may be significantly decreased which
could reduce trading price and increase the transaction costs of trading shares
of the Company’s common stock.
Except
for 2001, the Company has experienced operating losses every year since
inception in funding the research, development and clinical testing of our drug
candidates. The Company had an accumulated deficit of $209.8 million as of
December 31, 2009 and expects to continue to incur losses going forward. Such
losses have resulted principally from costs incurred in research and development
and from general and administrative expenses. Previously the Company had
financed its operations with public and private offerings of securities,
advances and other funding pursuant to an earlier marketing agreement with
Bausch & Lomb, grants from the Office of the Chief Scientist of Israel,
research contracts, the sale of a portion of its New Jersey net operating loss
carryforwards (NOL’s), and interest income. Although the Company
received $3.9 million in December 2009 from the sale of their NOL’s and had
approximately $4.6 million of cash and cash equivalents at December 31, 2009,
the Company is largely dependent upon achieving a collaboration with a
pharmaceutical partner or raising additional capital to either advance its lead
compound, Dextofisopam, for the treatment of IBS or to advance other earlier
stage intellectual property assets. During the year, the
Company engaged an investment advisor to assist with these strategies but has
not been successful to date. The Company has in the past pursued
various funding and financing options; however management believes that future
funding or financing options may be challenging because of the current
environment.
Also, the
Company does not currently have the finances and resources to complete full
clinical trials for its lead compound, Dextofisopam. As a result it may decide
to embark on smaller clinical trials for Dextofisopam or commence pre-clinical
development on its other intellectual property assets which could further reduce
the company’s current resources.
The
Company has corporate offices in Iselin, New Jersey.
3
STRATEGY
Pharmos
is currently developing only one compound, Dextofisopam, which has
completed a double-blind, placebo-controlled diarrhea-predominant or alternating
IBS Phase 2a study with positive effect on primary efficacy endpoint (n=141,
p=0.033). In this study, Dextofisopam was well-tolerated and demonstrated
significant improvement over placebo, suggesting that Dextofisopam has the
potential to become a novel firstline treatment for IBS. Pharmos initiated a
Phase 2b trial in February 2007 and in June 2007 the Company announced patient
screening had commenced. Dextofisopam is the R-enantiomer of racemic tofisopam,
a molecule marketed and used safely outside the United States for over three
decades for multiple indications including IBS. Unlike the two 5-HT3 or 5-HT4
IBS therapies recently introduced into the market, and subsequently withdrawn
because of safety concerns, Dextofisopam’s novel non-serotonergic activity
offers a unique and innovative approach to IBS treatment.
The
results of a Phase 2b study were announced in September 2009. The primary
endpoint of overall adequate relief was not met due to a high placebo response,
but drug activity was observed in all drug cohorts, especially the 200 mg
dose.
The
Company now is seeking a pharmaceutical partner with both scientific and
financial capabilities to further develop Dextofisopam.
In
research efforts over the past decade, the Company has developed a significant
expertise in cannabinoid biology and chemistry, and has generated significant
know-how and an intellectual property estate pertaining to multiple areas of
cannabinoid biology. With the decision to focus resources on
Dextofisopam and with the closure of the Company’s operations in Israel
effective October 31, 2008, no further development work is being performed on
the cannabinoid assets and the focus is to partner or sell these
assets.
In
November 2009 the Company entered into a Material Transfer Agreement whereby a
European company will perform certain experiments with samples of our synthetic
selective cannabinoid receptor CB2 agonist.
The
Company continues to seek, sell or license other CB2 assets, including
Cannabinor which was the only CB2 asset to enter human clinical
trials.
The
Company also maintains a commitment to out-license proprietary technologies and
products not consistent with our primary corporate focus. Assets involved are
Tianeptine to treat IBS or functional dyspepsia and S-Tofisopam.
The
Company owns the rights to both R and S Tofisopam. Dextofisopam is the R
enantiomer of racemic tofisopam and is being developed for IBS as described
above. In December 2009 the Company entered into a Material Transfer Agreement
with a US based company to perform certain experiments with
S-Tofisopam.
COMPOUND
IN CLINICAL DEVELOPMENT
Pharmos
currently has only one compound in clinical development –
Dextofisopam. Dextofisopam, a non-serotonergic agent currently being
evaluated for the treatment of irritable bowel syndrome (IBS), is the
R-enantiomer of racemic tofisopam, a molecule marketed and used safely outside
the United States for over three decades for multiple indications including IBS.
Dextofisopam represents a novel, first-in-class opportunity with a positive
proof-of-concept study in an arena where there are few compounds with unique
approaches or positive efficacy results. By structure, Dextofisopam is a member
of the homophthalazine class; Dextofisopam binds to specific receptors in areas
of the brain affecting autonomic function, including gastrointestinal (GI)
function. Unlike the two 5-HT3 or 5-HT4 mediated IBS therapies recently introduced into
the market, and subsequently withdrawn because of safety concerns,
Dextofisopam novel non-serotonergic activity offers a unique and innovative
approach to IBS treatment.
4
A
double-blind, placebo-controlled diarrhea-predominant or alternating IBS Phase
2a study has been completed with positive effect on primary efficacy endpoint
(n=141, p=0.033). In this study, Dextofisopam was well-tolerated and
demonstrated significant improvement over placebo suggesting that Dextofisopam
has the potential to become a novel firstline treatment for IBS.
On
September 14, 2009, the Company announced the results of its Phase 2b
Dextofisopam clinical trial to evaluate safety and efficacy of the compound in
irritable bowel syndrome.
Although
the primary efficacy variable (% of weeks responding for adequate overall relief
of IBS symptoms) did not reach statistical significance, the percentage
responding for the Dextofisopam 200 mg group was higher than that observed for
the Phase 2a trial. However, the placebo response rate was also higher than
expected compared to the Phase 2a placebo response.
An
extensive patent estate is in place with Dextofisopam. This consists of an
issued composition-of-matter patent, an issued manufacturing patent, and
numerous additional pending patent applications in both the United States
(including use of Dextofisopam for inflammatory disorders and immunomodulation)
in both the United States and foreign counterparts.
Potential pharmaceutical
market for Dextofisopam
The
development of Dextofisopam for the treatment of Irritable Bowel Syndrome (IBS)
is a major goal of Pharmos’ development activity. Direct medical costs
associated with IBS have been estimated at $8 billion annually in the U.S. It
has been estimated that indirect costs for IBS in the U.S. exceed $20 billion.
The few recent products introduced in this market have been limited by poor
safety profiles.
Irritable
Bowel Syndrome: Disease Description and Diagnosis
Irritable
bowel syndrome is defined by a constellation of symptoms that includes abdominal
pain or discomfort accompanied by diarrhea, constipation, or an alternation
between the two. IBS is classified as a functional disorder; it is diagnosed by
symptom-based criteria following exclusion of organic diseases that may produce
abdominal pain and altered bowel function.
Although
the etiology of IBS is not completely understood, five factors have been
proposed as playing a role in the development of IBS:
|
|
1.
|
Psychosocial
factors
|
|
|
a.
|
impact
of stress on motor function of the GI
tract
|
|
|
b.
|
approximately
60% of patients seen at referral centers have psychiatric
symptoms.
|
|
|
2.
|
Visceral
hypersensitivity - lower threshold for abdominal
pain
|
|
|
3.
|
Altered
bowel motility - abnormal motility of the small
intestine
|
|
|
4.
|
Infection
and inflammation
|
|
|
5.
|
Autonomic
nervous system dysfunction
|
|
|
a.
|
studies
show that IBS patients have aberrant autonomic nervous system
activity
|
|
|
b.
|
altering
autonomic nervous system activity in volunteers produces symptoms of
IBS.
|
This
perspective on the pathophysiology of IBS suggests that a drug that improves
altered bowel motility, decreases visceral hypersensitivity reduces
stress-related impact on GI function, normalizes autonomic dysfunction, and
possesses anti-inflammatory properties may provide a superior, broad-spectrum
approach to the treatment of IBS. In preclinical studies, Dextofisopam exhibits
all of these properties.
The
diagnostic criteria for IBS have evolved over the years. Today, diagnosis for
clinical trials and regulatory purposes is defined by the Rome III Criteria.
Diagnosis is based on the patient having a) recurrent abdominal pain or
discomfort for at least 6 months, and b) at least two of the following
three features on at least 3 days per month over the past 3
months:
5
|
|
1.
|
Improvement
with defecation
|
|
|
2.
|
Onset
associated with a change in frequency of
stool
|
|
|
3.
|
Onset
associated with a change in form (appearance) of
stool
|
Based on
the clustering of symptoms, IBS patients are usually categorized as either
“diarrhea-predominant,” “constipation-predominant,” or “alternating” (also
termed “mixed”). For a diagnosis of IBS, specific “red flag” symptoms
are usually excluded. These “red flag” symptoms may indicate the presence of
organic disease, such as colon cancer (especially if onset is rapid or occurs
over the age of 40), ulcerative colitis (rectal bleeding), or Crohn’s disease
(weight loss, fever). For the diagnosis of IBS to be made, normal results must
have been obtained for blood and stool tests, x-rays, endoscopy, and
biopsies.
While not
a life-threatening disease, IBS can have a large negative impact on the quality
of life of patients. Even mild cases can be life-altering, and severe cases are
often debilitating, with the frequency and severity of episodes seriously
affecting work, school, and social schedules.
IBS is a
leading cause of physician visits, accounting for approximately 3,000,000 visits
annually in the U.S., representing 4% of all visits to office-based physicians
and 49% of visits to office-based gastroenterologists. The need to eliminate
other possible diagnoses (colon cancer, inflammatory bowel disease, and other GI
diseases) necessitates expensive in-office procedures. As noted earlier, the
annual direct medical costs for IBS in the U.S. have been estimated at
$8 billion.
Prevalence
of IBS
IBS is a
very common disorder, with studies indicating prevalence in the range of 6-15%
for North America, Europe, and Japan. Based on a prevalence rate of 12.5%, approximately
36 million individuals in the United States meet diagnostic criteria for
IBS. Prevalence rates are similar in the major European markets
(France, Germany, Italy, Spain, and the United Kingdom). While prevalence rates
for other countries are not well established, published studies support the
existence and recognition of IBS throughout the world, including China and
India.
Irritable
bowel syndrome, or IBS, is a chronic, recurring condition with symptoms that
affect more often women than men. IBS is characterized by multiple
symptoms that include bowel dysmotility—diarrhea, constipation, or alternating
diarrhea and constipation—and abdominal discomfort. Studies have
shown that diarrhea-predominant IBS appears to be the most common
subtype. For patients with diarrhea-predominant and alternating-type
IBS, there are no recently approved treatments for any but the most severely
affected women, and none for men.
The
prevalence of IBS varies with gender and age. Higher prevalence rates are
consistently reported for women than for men (two to three times greater). While
IBS is observed in all age groups, including pediatric and geriatric
populations, it is more common in the age range of 20 to 60 years for both
genders
Current
and Recent Therapies
Even with
the introduction of two new therapies - Lotronex®
(alosetron), a 5-HT3
antagonist, and Zelnorm®
(tegaserod), a 5-HT4 partial
agonist - the IBS market is still largely served by older products, with
questionable efficacy and poor tolerability. These older products include
antispasmodics, laxatives, and antidiarrheal agents. While antispasmodics at
high doses may provide relief of specific symptoms, these drugs are poorly
tolerated at those doses. The estimated sales of IBS drugs in the U.S. market
totaled $348 M in 2009.
6
Lotronex® was
introduced into the U.S. IBS market in March 2000, and was withdrawn from the
market in November 2000 due to safety issues. Lotronex® was
subsequently reintroduced in 2003 with a restricted marketing program (Physician
Prescribing Program). The utility of Lotronex® is
severely hampered both by its narrow indication (only for women with severe
diarrhea-predominant IBS who have failed to respond to conventional therapy) and
a major safety issue (the risk of potentially fatal ischemic colitis). Since its
reintroduction, Lotronex® has
had minimal prescription volume.
Zelnorm® was
introduced into the U.S. IBS market in July 2002 for short-term use in women
with constipation-predominant IBS. In April 2004, a precautionary statement was
added to Zelnorm®
labeling regarding post-marketing cases of ischemic colitis and a warning for
severe diarrhea. Despite these changes to the package insert, the strong
marketing and educational efforts supporting Zelnorm®
appeared to have increased awareness and expanded utilization. Zelnorm®
achieved U.S. sales of $26 M in 2002, $132 M in 2003, $249 M in 2004, $357 M in
2005, and $488 M in 2006, with 2006 worldwide sales of $561 M. Zelnorm® was
approved in 30 countries, though not in major EU markets or in Japan.
Zelnorm® was
voluntarily withdrawn from the market in March 2007 because of severe side
effects.
Market
Potential (United States and Rest of World)
The
market potential for IBS is very large. IBS is a prevalent disorder
for which there are currently no safe and broadly effective
treatments. Investment research analysts estimate a U.S. market worth
approximately $2.5 B. Market research reports project that the
current full worldwide (WW) market potential for IBS therapies could be as high
as $15 B (over $6 B in the United States alone). Based on its current clinical
profile, Dextofisopam may have the potential to capture a significant portion of
the IBS market.
COMPOUNDS AVAILABLE FOR
LICENSING OR SALE
Pharmos
is not currently performing any research or discovery work. The focus of the
Company is to secure a pharmaceutical partnership arrangement for
Dextofisopam.
Dextofisopam
As
described above the Company has completed both a Phase 2a and Phase 2b trials of
Dextofisopam for IBS. The results of the Phase 2b trial were reported in
September 2009 and in October 2009 the Company engaged Cowen and Company to help
accelerate a partnership arrangement with a pharmaceutical company possessing
greater scientific and financial capabilities than Pharmos.
CB2-selective
cannabinoids
Pharmos’
novel CB2-selective cannabinoids are synthetic compounds which belong to the
class of nonclassical cannabinoids. Compounds in this class have been
demonstrated to possess immunomodulatory and analgesic activities. Importantly,
the CB2-selective cannabinoids display fewer of the undesired psychotropic and
cardiovascular side-effects seen with some natural cannabinoids because they
bind with high affinity to the peripheral cannabinoid type two (CB2) receptor
and with lower affinity to the cannabinoid type one (CB1) receptor, located in
the central nervous system. In contrast to CB1 receptors, CB2 receptors are
expressed mainly outside of the central nervous system, on immune and
inflammatory cells. CB2 activation also inhibits the release of
pro-nociceptive peptide from the periphery which may prevent activation of
primary afferent neurons. CB2 activation also modulates T-cell
activity, skewing the T-helper cell type 1 (Th1) responses
to the T-helper cell type 2 (Th2)
activity. Most autoimmune diseases and models of autoimmunity in
which susceptibility is associated with the expression of specific MHC class II
allotypes appear to be of the Th1 type. Thus
considerable emphasis has been placed on developing means of altering the course
of the autoimmune Th1 response
to become that of a Th2 response,
with the goal of down regulating the autoimmune pathogenesis.
7
Several
candidates from Pharmos’ CB2-selective cannabinoid library have demonstrated
promise in animal models for autoimmune inflammatory disorders, such as multiple
sclerosis and inflammatory bowel disease. These compounds have also demonstrated
efficacy in animal models of neuropathic, inflammatory and chronic nociceptive
pain. In selected preclinical models, these compounds have demonstrated
analgesic activity equivalent or better than Gabapentin.
Pharmos’
chemical library consists of several chemically distinct classes of cannabinoid
compounds. The Company primary focused on development of families of
CB2-selective compounds which were cannabinoid receptor agonists that bind
preferentially to CB2 receptors. These receptors were found primarily
in peripheral neurons and immune cells. The compounds possess
advantages such as a simple synthesis, increased potency and improved
drugability. Pharmos recognized the potential therapeutic promise of
CB2 activation in such diverse disease entities as pain, inflammation,
autoimmunity, osteoporosis and atherosclerosis.
Pharmos
closed its operations in Israel in late 2008 and has ceased any discovery or
development work on CB2 selective cannabinoid program. However these assets are
available for licensing and / or sale. There has been limited interest to date.
In November 2009 Pharmos entered into a Material Transfer Agreement whereby a
European company will perform certain experiments with samples of our synthetic
selective cannabinoid receptor agonist.
COMPETITION
The
pharmaceutical industry is highly competitive. Pharmos competes with a number of
pharmaceutical companies that have financial, technical and marketing resources
that are significantly greater than those of Pharmos. Some companies
with established positions in the pharmaceutical industry may be better equipped
than Pharmos to develop market and distribute products in the global markets
that Pharmos is seeking to enter. A significant amount of pharmaceutical
research is also being carried out at universities and other not-for-profit
research organizations. These institutions are becoming increasingly aware of
the commercial value of their findings and are becoming more active in seeking
patent protection and licensing arrangements to collect royalties for the use of
technology they have developed. They may also market competitive commercial
products on their own or through joint ventures and will compete with Pharmos in
recruiting highly qualified scientific personnel. Further, these institutions
will compete with Pharmos in recruiting qualified patients for enrollment in
their trials.
Pharmos
is pursuing areas of product development in which there is a potential for
extensive technological innovation. Pharmos’ competitors may succeed in
developing products that are more effective than those of Pharmos. Rapid
technological change or developments by others may result in Pharmos’ potential
products becoming obsolete or non-competitive.
We know
of a number of programs in various stages that compete in the
area of IBS.
Salix
Pharmaceuticals is developing rifaximin, an antibiotic marketed for the
treatment of traveler’s diarrhea. Salix is conducting clinical development
studies to assess the utility of rifaximin for the treatment of irritable bowel
disease. Ocera Therapeutics is developing AST-120 for a number of conditions,
including IBS; Lexicon Genetics is developing LX1031, which has completed a
Phase 2 clinical trial. LX1031 is an orally-delivered small molecule designed to
regulate gastrointestinal (GI) function by reducing the amount of serotonin
available for receptor activitation in the GI tract without affecting serotonin
levels in the brain. Serotonin is a key regulator of GI function, and is
associated with the most common symptoms of IBS: problems of bowel motility and
GI discomfort. Several other compounds are reported to be in development for the
treatment of IBS. However, it is not clear if these compounds are
intended for a specific type of IBS, e.g., diarrhea-predominant,
constipation-predominant, or alternating-type IBS. Such compounds
include a glucagon-like peptide-1 agonist (Gastrotech) and asimadoline, a kappa
opiate agonist (Tioga), both reportedly in Phase 2. Several other
compounds are reported to be in development for the treatment of
constipation-predominant IBS. While such compounds may not directly
compete with Dextofisopam, it is possible that they may eventually be developed
for diarrhea-predominant or alternating-type IBS, or that Dextofisopam may
eventually be developed
for constipation-predominant IBS. Such compounds include MD-1100, a
guanylate cyclase C agonist reportedly in Phase 3
(Ironwood).
8
Dextofisopam
is currently targeted for the treatment of diarrhea-predominant IBS and/or
alternating-type IBS. To the best of our knowledge, there are no competing
compounds in development specifically for the treatment of alternating-type
IBS. However, the alternating results have been mixed between the Phase 2a
and Phase 2b trials and the numbers of patients were small. Several
compounds are reported to be in development for the potential treatment of
diarrhea-predominant IBS.
The IBS
area remains a difficult development area. During the past year some of our
potential competition has reported disappointing or failed clinical trials.
However, as IBS represents a large unmet medical need, we expect other
competition to emerge.
Collaborative
Relationships
The
Company’s principal focus is to seek a pharmaceutical partner with the
appropriate GI clinical and scientific expertise for further development of
Dextofisopam. On October 21, 2009 the Company announced that it had
engaged Cowen and Company as advisors to assist with accelerating a partnership
arrangement for Dextofisopam.
Depending
on the availability of financial, marketing and scientific resources, among
other factors, Pharmos may license its technology or products to others and
retain profit sharing, royalty, manufacturing, co-marketing, co-promotion or
similar rights. Any such arrangements could limit Pharmos’ flexibility in
pursuing alternatives for the commercialization of its products. Due to the
often unpredictable nature of the collaborative process, Pharmos cannot be
certain that it will be able to establish any additional collaborative
arrangements or that, if established, any of these relationships will be
successful.
Patents and Proprietary
Rights
Proprietary
protection generally has been important in the pharmaceutical industry, and the
commercial success of products incorporating Pharmos' technologies may depend,
in part, upon the ability to obtain strong patent protection.
Pharmos
generally maintains, at its expense, U.S. and foreign patent rights with respect
to both the licensed technology and its own technology and files and/or
prosecutes the relevant patent applications in the U.S. and foreign countries.
Pharmos also relies upon trade secrets, know-how, continuing technological
innovations and licensing opportunities to develop its competitive position.
Pharmos' policy is to protect its technology by, among other things, filing, or
requiring the applicable licensor to file, patent applications for technology
that it considers important to the development of its business. Pharmos intends
to file additional patent applications, when appropriate, relating to its
technology, improvements to its technology and to specific products it
develops.
The
patent positions of pharmaceutical firms, including Pharmos, are uncertain and
involve complex factual questions. In addition, the coverage claimed in a patent
application can be significantly reduced before or after the patent is issued.
Consequently, Pharmos does not know whether any of the pending patent
applications underlying the licensed technology will result in the issuance of
patents or, if any patents are issued, whether they will provide significant
proprietary protection or will be circumvented or invalidated. Since patent
applications in the U.S. and elsewhere publish only 18 months after priority
date, and since publication of discoveries in the scientific or patent
literature often lag behind actual discoveries, Pharmos cannot be certain that
it or its licensors, as the case may be, were the first creators of inventions
covered by pending and issued patents or that it or its licensors, as the case
may be, were the first to file patent applications for such inventions.
Moreover, it may be necessary for Pharmos to participate in interference
proceedings declared by the U.S. Patent and Trademark Office in order to
determine priority of invention. Involvement in these proceedings could result
in substantial cost to Pharmos, even if the eventual outcomes are favorable to
Pharmos. Because the results of the judicial process are often uncertain, we
cannot be certain that a court of competent jurisdiction will uphold the
patents, if issued, relating to the licensed technology, or that a competitor's
product will be found to infringe those patents.
9
Other
pharmaceutical and drug delivery companies and research and academic
institutions may have filed patent applications or received patents in Pharmos'
fields. If patents are issued to other companies that contain competitive or
conflicting claims and those claims are ultimately determined to be valid, it is
possible that Pharmos would not be able to obtain licenses to these patents at a
reasonable cost or be able to develop or obtain alternative
technology.
Pharmos
also relies upon trade secret protection for its confidential and proprietary
information. It is always possible that others will independently
develop substantially equivalent proprietary information and techniques or
otherwise gain access to Pharmos' trade secrets.
It is
Pharmos' policy to require its employees, consultants, outside scientific
collaborators, sponsored researchers and other advisors to execute
confidentiality agreements upon the commencement of employment or consulting or
advisory relationships with Pharmos. These agreements generally
provide that all confidential information developed or made known to the
individual during the course of the individual's relationship with Pharmos is to
be kept confidential and not disclosed to third parties except in specific
circumstances. Further, these agreements provide for the maintenance of
confidentiality following the termination of the individual’s relationship with
Pharmos. In the case of employees and certain consultants, the agreements
provide that all inventions conceived by the individual in the course of their
employment or consulting relationship shall be the exclusive property of
Pharmos. Due to the vital nature of trade secrets and the often uncertain
results of the judicial process, we cannot be sure, however, that these
agreements will provide meaningful protection or adequate remedies for Pharmos'
trade secrets in the event of unauthorized use or disclosure of such
information. Pharmos' patents and licenses underlying its potential products
described herein are summarized below.
Dextofisopam
to Treat IBS. Dextofisopam is a novel
non-serotonergic agent in development for the treatment of
IBS. Dextofisopam is the R-enantiomer of racemic
tofisopam. Pharmos holds an issued composition-of-matter patent in
the United States on Dextofisopam which expires in 2019. Pharmos owns
certain counterpart foreign patents and patent applications, as
well. Pharmos has filed additional patent applications for the
treatment of IBS in the United States, and abroad.
Tianeptine
to Treat IBS or Functional Dyspepsia. Tianeptine is a
racemic molecule marketed and used outside the United States for the treatment
of depression. Pharmos’ patent protection for tianeptine consists of
a United States method-of-use patent covering tianeptine and its two enantiomers
for the treatment of IBS and non-ulcer dyspepsia. This patent expires
in 2024. Additional foreign patents related to this United States
patent are pending.
FDA
and Comparable Authorities in Other Countries
Regulation
by governmental authorities in the U.S. and other countries is a significant
factor in our ongoing research and development activities and in the production
and marketing of our products. Pharmaceutical products intended for
therapeutic use in humans are governed in the U.S. by the Federal Food, Drug and
Cosmetic Act (21 U.S.C. § 321 et seq.) and by FDA
regulations and by comparable agency regulations in other
countries. Specifically, in order to undertake clinical tests, and to
produce and market products for human therapeutic or diagnostic use, mandatory
procedures and safety standards established by the FDA and Department of Health
and Human Services in the U.S. and comparable agencies in other countries must
be implemented and followed. These standards include protection of
human research subjects.
The
following is an overview of the steps that must be followed before a drug
product may be marketed lawfully in the U.S.:
10
|
|
(i)
|
Preclinical
studies including pharmacology, laboratory evaluation and animal studies
to test for initial safety and
efficacy;
|
|
|
(ii)
|
Submission
to the FDA of an Investigational New Drug (IND) Application, which must
become effective before human clinical trials may
commence;
|
|
|
(iii)
|
Adequate
and well-controlled human clinical trials to establish the safety and
efficacy of the drug in its intended
application;
|
|
|
(iv)
|
Submission
to the FDA of a New Drug Application (NDA), which application is not
automatically accepted by the FDA for consideration;
and
|
|
|
(v)
|
FDA
approval of the New Drug Application prior to any commercial sale or
shipment of the drug.
|
In
addition to obtaining FDA approval for each product, each drug-manufacturing
establishment must be registered or licensed by the FDA for each product sold
within the US that is manufactured at that facility. Manufacturing
establishments are subject to inspections by the FDA and by other national and
local agencies and must comply with current Good Manufacturing Practices (cGMPs)
requirements that are applicable to the manufacture of pharmaceutical drug
products and their components.
Preclinical
studies include laboratory evaluation of product chemistry and animal studies to
assess the potential safety and efficacy of the product and its
formulation. The results of the preclinical studies are submitted to
the FDA as part of an IND, and unless the FDA objects, the application will
become effective 30 days following its receipt by the FDA. If the potential of
addiction is found in the animal tests, then additional regulatory requirements
may be imposed by the FDA and DEA.
Clinical
trials involve the administration of the drug to healthy volunteers as well as
to patients under the supervision of a qualified “principal investigator,” who
is a medical doctor. Clinical trials in humans are necessary because
effectiveness in humans may not always be gleaned from findings of effectiveness
in animals. They are conducted in accordance with protocols that detail the
objectives of the study, the parameters to be used to monitor safety and the
efficacy criteria to be evaluated. Each protocol is submitted to the
FDA as part of the application. Each clinical study is approved and monitored by
an independent Institutional Review Board (IRB) (Ethics Committee) at each
clinical site. The IRB must consider, among other things, the process of
obtaining the informed consents of each study subject, the safety of human
subjects, the possible liability of the institution conducting a clinical study,
as well as various ethical factors.
Clinical
trials typically are conducted in three sequential phases, although the phases
may overlap. In Phase I, the initial introduction of the drug to humans, the
drug is tested in a small group of healthy volunteers for safety and clinical
pharmacology such as metabolism and tolerance. Phase I trials may also yield
preliminary information about the product’s effectiveness and dosage levels.
Phase II involves detailed evaluation of safety and efficacy of the drug in
patients with the disease or condition being studied. It also involves a
determination of optimal dosage and identification of possible side effects in a
larger patient group. Phase III trials consist of larger scale evaluation of
safety and efficacy and usually require greater patient numbers and multiple
clinical trial sites, depending on the clinical indications for which marketing
approval is sought.
The
process of completing clinical testing and obtaining FDA approval for a new
product is likely to take a number of years and requires the expenditure of
substantial resources. The FDA may grant an unconditional approval of a drug for
a particular indication or may grant approval conditioned on further
post-marketing testing. The FDA also may conclude that the submission is not
adequate to support an approval and may require further clinical and preclinical
testing, re-submission of the New Drug Application, and further
review. Even after initial FDA approval has been obtained, further
studies may be required to provide additional data on safety or to gain approval
for the use of a product for clinical indications other than those for which the
product was approved initially. This could delay the NDA approval
process.
11
The 1962
amendments to the Federal Food, Drug and Cosmetic Act required for the first
time that drug effectiveness be proven by adequate and well-controlled clinical
trials. The FDA interpretation of that requirement is that at least two such
trials are necessary to demonstrate effectiveness for approval of an NDA. This
interpretation is based on the scientific need for independent substantiation of
study results. However, Section 115 of FDAMA revised Section 505 of the Act to
read, in pertinent part that “based on relevant science, data from one adequate
and well-controlled clinical investigation and confirmatory evidence … are
sufficient to establish effectiveness.” The FDA has not issued
comprehensive standards of testing conditions for pivotal trials. The
FDA maintains a preference for at least two adequate and well-controlled
clinical trials.
Pharmos’
products will be subject to foreign regulatory approval before they may be
marketed abroad. Marketing beyond the US is subject to regulatory requirements
that vary widely from country to country. In the European Union, the general
trend has been towards coordination of the common standards for clinical testing
of new drugs. Centralized approval in the European Union is coordinated through
the European Medicines Evaluation Agency (EMEA). The time required to obtain
regulatory approval from comparable regulatory agencies in each country may be
longer or shorter than that required for FDA or EMEA approval. Further, in
certain markets, reimbursement may be subject to governmentally mandated
prices.
Corporate
History
Pharmos
Corporation, (formerly known as Pharmatec, Inc.) a Nevada corporation, was
incorporated under the laws of the State of Nevada on December 20,
1982. On October 29, 1992, Pharmatec, the Nevada Corporation,
completed a merger with a privately held New York corporation known as Pharmos
Corporation founded by Dr. Haim Aviv (the name of the post-merger Nevada
Corporation was changed to Pharmos Corporation).
Human
Resources
As of
December 31, 2009, Pharmos had 4 full-time employees in the U.S.
Pharmos’
employees are not covered by a collective bargaining agreement. To date, Pharmos
has not experienced employment-related work stoppages and considers its employee
relations to be excellent.
Public
Funding and Grants
Pharmos’
subsidiary, Pharmos Ltd., has received certain funding from the Chief Scientist
of the Israel Ministry of Industry and Trade (the Chief Scientist) for: (1)
research and development of dexanabinol; (2) SubMicron Emulsion technology for
injection and nutrition; (3) research relating to pilocarpine, dexamethasone and
ophthalmic formulations for dry eyes; (4) research and development of CB2,
including cannabinor. As of December 31, 2009, the total amounts received under
such grants amounted to $17,897,830. Under the terms of the grant agreements,
aggregate future royalty payments related to sales of products developed, if
any, as a result of the grants are limited to $16,408,890 based on grants
received through December 31, 2009. Pharmos will be required to pay royalties to
the Chief Scientist ranging from 3% to 5% of product sales, if any, as a result
of the research activities conducted with such funds. Aggregate
royalty payments per product are limited to the amount of funding received to
develop that product and interest. Additionally, funding by the Chief
Scientist places certain legal restrictions on the transfer of know-how and the
manufacture of resulting products outside of Israel. With the closure
of the Israel location, the Company will not be eligible for further OCS grants
and received none in 2008 or 2009.
The
Company closed its operations in Israel effective August 31, 2008 and has
subsequently reconciled and repaid a Chief Scientist grant that was overpaid.
All obligations of the Company have been settled except the requirement to pay
royalties should any of the related technologies be licensed out and further
developed.
12
All
reports filed by the Company with the SEC are available free of charge via EDGAR
through the SEC website at www.sec.gov. In
addition, the public may read and copy materials filed by the Company with the
SEC at the SEC’s public reference room located at 100 F Street NE, Washington,
D.C., 20549. The company also provides copies of its Forms 8-K, 10-K, 10-Q,
Proxy and Annual Report at no charge available through its website at http://investors.pharmoscorp.com/sec.cfm
as soon as reasonably practicable after filing electronically such material with
the SEC. Copies are also available, without charge, from Pharmos Corporation, 99
Wood Avenue South, Suite 311, Iselin, NJ, 08830.
Item
1A. Risk Factors
We
are very largely dependent upon the success of our lead drug candidate,
Dextofiosopam, currently being developed for Irritable Bowel Syndrome, for
diarrhea predominant patients.
We
currently have one product candidate, Dextofisopam that has completed Phase 2b
clinical trials. Our other drug candidate assets are in earlier stages of
development and are not currently being developed. Our primary focus is on the
development and partnering of Dextofisopam. We engaged Cowen and Company in late
2009 to help us accelerate and secure a partnership arrangement for Dextofisopam
with a pharmaceutical company with greater scientific and financial resources
than we have. If we fail to secure a partnership arrangement, or raise
additional capital, our operations will need to be scaled back or
discontinued.
We closed
our operations in Israel in late 2008 and those earlier stage assets are
available for licensing.
We
are at an early stage of development.
We are at
an early stage of development. Our sole product candidate, Dextofisopam, has
completed a Phase 2b trial for the treatment of Irritable Bowel Syndrome
(IBS). While the trial did not meet the primary endpoint, the drug
treatment group at 200 mg demonstrated activity and secondary response variables
of adequate relief of abdominal pain and discomfort, and overall IBS symptoms
ratings showed statistical significance and trends favoring the Dextofisopam
group compared to placebo. Although the primary efficacy variable did
not reach statistical significance, the percentage responding for the
Dextofisopam 200 mg group was higher than that observed in the successful Phase
2a trial. However, the placebo was also higher than the Phase 2a
trial. We plan to seek a pharmaceutical partner with the appropriate
GI clinical and scientific expertise for further development of Dextofisopam;
however, there can be no assurance that we will be able to find such a
partner. We have no other products in development.
We
will need to raise additional capital.
Our
ability to operate as a going concern is dependent upon raising adequate
financing or securing a partnership arrangement for Dextofisopam.
Except for 2001, the Company has experienced operating losses every year since
inception in funding the research, development and clinical testing of our drug
candidates. The Company had an accumulated deficit of $209.8 million as of
December 31, 2009 and expects to continue to incur losses going forward. Such
losses have resulted principally from costs incurred in research and development
and from general and administrative expenses. Previously the Company had
financed its operations with public and private offerings of securities,
advances and other funding pursuant to an earlier marketing agreement with
Bausch & Lomb, grants from the Office of the Chief Scientist of Israel,
research contracts, the sale of a portion of its New Jersey net operating loss
carryforwards (NOL’s), and interest income. Although the Company
received $3.9 million in December 2009 from the sale of their NOL’s and had
approximately $4.6 million of cash and cash equivalents at December 31, 2009,
the Company is largely dependent upon achieving a collaboration with a
pharmaceutical partner or raising additional capital to either advance its lead
compound, Dextofisopam, for the treatment of IBS or to advance other earlier
stage intellectual property assets. During the year, the
Company engaged an investment advisor to assist with these strategies but has
not been successful to date. The Company has in the past pursued
various funding and financing options; however management believes that future
funding or financing options may be challenging because of the current
environment.
13
Also, the
Company does not currently have the finances and resources to complete full
clinical trials for its lead compound, Dextofisopam. As a result it may decide
to embark on smaller clinical trials for Dextofisopam or commence pre-clinical
development on its other intellectual property assets which could further reduce
the company’s current resources.
We
have been delisted from Nasdaq.
On March
13, 2009, the Company was officially delisted from the Nasdaq Capital Market,
and is currently trading on the OTCBB pink sheets. The Company was not in
compliance with the minimum $2,500,000 stockholders’ requirement for continued
listing and was unable to comply during the grace period extended by Nasdaq. As
a result of trading on the OTCBB pink sheets, liquidity for our common stock may
be significantly decreased which could reduce trading price and increase the
transaction costs of trading shares of the company’s common stock.
The
price of our Common Stock may experience volatility.
The
trading price of our Common Stock could be subject to wide fluctuations in
response to variations in our quarterly operating results, the failure of trial
results, the failure to bring products to market, conditions in the industry,
and the outlook for the industry as a whole or general market or economic
conditions. In addition, in recent years, the stock market has experienced
extreme price and volume fluctuations. These fluctuations have had a substantial
effect on the market prices for many companies, often unrelated to the operating
performance of the specific companies. Such market fluctuations could have a
material adverse effect on the market price for our securities.
We
have certain obligations to indemnify our officers and directors.
We
have certain obligations to indemnify our officers and directors and to advance
expenses to such officers and directors. Although we have purchased liability
insurance for our directors and officers, if our insurance carriers should deny
coverage, or if the indemnification costs exceed the insurance coverage, we may
be forced to bear some or all of these indemnification costs directly, which
could be substantial and may have an adverse effect on our business, financial
condition, results of operations and cash flows. If the cost of our liability
insurance increases significantly, or if this insurance becomes unavailable, we
may not be able to maintain or increase our levels of insurance coverage for our
directors and officers, which could make it difficult to attract or retain
qualified directors and officers.
We
have a history of operating losses and expect to sustain losses in the
future.
We have
experienced significant operating losses since our inception. As of December 31,
2009, we had an accumulated deficit of approximately $210 million. We expect to
incur operating losses over the next several years as our research and
development efforts and preclinical and clinical testing activities continue.
Our ability to generate revenues and achieve profitability depends in part upon
our ability, alone or with others, to successfully complete development of our
proposed products, to obtain required regulatory approvals and to manufacture
and market our products.
14
Our
product candidates may not successfully complete clinical trials required for
commercialization, and as a result our business may never achieve
profitability.
To obtain
regulatory approvals needed for the sale of our drug candidates, we must
demonstrate through testing and clinical trials that each drug candidate is both
safe and effective for the human population that it was intended to treat. In
general, two successful Phase III clinical trials are required. The clinical
trial process is complex and the regulatory environment varies widely from
country to country. Positive results from testing and early clinical trials do
not ensure positive results in the Phase III human clinical trials. Many
companies in our industry have suffered significant setbacks in Phase III,
potentially pivotal clinical trials, even after promising results in earlier
trials. The results from our trials, if any, may show that our drug candidates
produce undesirable side effects in humans or that our drug candidates are not
safe or effective or not safe or effective enough to compete in the marketplace.
Such results could cause us or regulatory authorities to interrupt, delay or
halt clinical trials of a drug candidate. Moreover, we, the FDA, or foreign
regulatory authorities may suspend or terminate clinical trials at any time if
we or they believe the trial participants face unacceptable health risks or that
our drug candidates are not safe or effective enough. Clinical trials are
lengthy and expensive. They require adequate supplies of drug substance and
sufficient patient enrollment. Patient enrollment is a function of many factors,
including:
|
·
|
the
size of the patient population,
|
|
·
|
the
nature of the protocol (i.e., how the drug is given, and the size and
frequency of the dose and use of placebo
control),
|
|
·
|
the
proximity of patients to clinical sites,
and
|
|
·
|
the
eligibility criteria for the clinical trial (i.e., age group, level of
symptoms, concomitant diseases or medications
etc.).
|
Delays in
patient enrollment or negative trial outcomes can result in increased costs and
longer development times. Even if we successfully complete clinical trials, we
may not be able to file any required regulatory submissions in a timely manner
and we may not receive regulatory approval for the particular drug candidate
that was tested.
In
addition, if the FDA or foreign regulatory authorities require additional
clinical trials, we could face increased costs and significant development
delays. Changes in regulatory policy or additional regulations adopted during
product development and regulatory review of information we submit could also
result in delays or rejections.
Our
clinical trials depend on third party investigators who are outside our
control.
We depend
upon the personnel of third party independent investigators to conduct our
clinical trials. Such personnel are not our employees, and we cannot control the
amount of time or resources that they devote to our programs. They may not
assign as great a priority to our programs or pursue them as diligently as we
would if we were undertaking such programs ourselves. If such third-party
personnel fail to devote sufficient time and resources to our clinical trials,
or if their performance is substandard, the approval of our FDA applications, if
any, and our introduction of new drugs, if any, will be delayed. Such
third-party investigators may also have relationships with other commercial
entities that compete with us. If they assist our competitors at our expense,
our competitive position would be harmed.
15
We
face extensive governmental regulation and any failure to adequately comply
could prevent or delay product approval or cause the disallowance of our
products after approval.
The FDA
and comparable agencies in foreign countries impose many requirements on the
introduction of new drugs through lengthy and detailed clinical testing
procedures, and other costly and time consuming compliance procedures. These
requirements make it difficult to estimate when any of our products in
development will be available commercially, if at all. In addition, the FDA or
other comparable agencies in foreign countries may impose additional
requirements in the future that could further delay or even stop the
commercialization of our products in development.
Our
proprietary compounds in development require substantial clinical trials and FDA
review as new drugs. Even if we successfully enroll patients in our clinical
trials, patients may not respond to our potential drug products. We think it is
prudent to expect setbacks and possible product failures. Failure to comply with
the regulations applicable to such testing may delay, suspend or cancel our
clinical trials, or the FDA might not accept the test results. The FDA, or any
comparable regulatory agency in another country, may suspend clinical trials at
any time if it concludes that the trials expose subjects participating in such
trials to unacceptable health risks. Further, human clinical testing may not
show any current or future product candidate to be safe and effective to the
satisfaction of the FDA or comparable regulatory agencies or the data derived
there from may be unsuitable for submission to the FDA or other regulatory
agencies.
We cannot
predict with certainty when we might submit any of our proposed products
currently under development for regulatory review. Once we submit a proposed
product for review, the FDA or other regulatory agencies may not issue their
approvals on a timely basis, if at all. If we are delayed or fail to obtain such
approvals, our business may be damaged due to the resulting inability to
generate revenues from the sale of such product. If we fail to comply with
regulatory requirements, either prior to approval or in marketing our products
after approval, we could be subject to regulatory or judicial enforcement
actions. These actions could result in:
|
·
|
injunctions;
|
|
·
|
criminal
prosecution;
|
|
·
|
refusals
to approve new products and withdrawal of existing approvals;
and
|
|
·
|
enhanced
exposure to product liabilities.
|
We
need to find collaborative partners.
Our
strategy for the development, clinical testing, manufacture, marketing and
commercialization of our products includes the use of collaborations with
corporate partners, licensors, licensees and others.
Due to
the often unpredictable nature of the collaboration process, we cannot be sure
that any present or future collaborative agreements will be successful. To the
extent we choose not to or are not able to establish such arrangements, we would
experience increased capital requirements. In addition, we may encounter
significant delays in introducing our products currently under development into
certain markets or find that the development, manufacture, or sale of those
products is hindered by the absence of collaborative agreements due to the
relatively small size of our company as compared with that of some of our
potential competitors.
16
The
value of our research could diminish if we cannot protect or enforce our
intellectual property rights adequately.
We
actively pursue both domestic and foreign patent protection for our proprietary
products and technologies. We have filed for patent protection for our
technologies in all markets we believe to be important for the development and
commercialization of our drug products; however, our patents may not protect us
against our competitors. We may have to file suit to protect our patents or to
defend our use of our patents against infringement claims brought by others.
Because we have limited cash resources, we may not be able to afford to pursue
or defend against litigation in order to protect our patent rights. As a result,
while we currently have no specific concerns about gaps in our intellectual
property portfolio, we recognize that for companies like ours, where
intellectual property constitutes a key asset, there is always a risk that a
third party could assert a patent infringement claim or commence a patent
interference action. Defending against any such claims or actions could be very
costly to us, even if they were without merit.
We also
rely on trade secret protection for our unpatented proprietary technology.
However, trade secrets are difficult to protect. While we enter into proprietary
information agreements with our employees and consultants, these agreements may
not successfully protect our trade secrets or other proprietary
information.
We
face large competitors and our limited financial and research resources may
limit our ability to develop and market new products.
The
pharmaceutical industry is highly competitive. We compete with a number of
pharmaceutical companies that have financial, technical and marketing resources
that are significantly greater than ours. Some companies with established
positions in the pharmaceutical industry may be better equipped than we are to
develop, market and distribute products in the global markets we seek to enter.
A significant amount of pharmaceutical research is also being carried out at
universities and other not-for-profit research organizations. These institutions
are becoming increasingly aware of the commercial value of their findings and
are becoming more active in seeking patent protection and licensing arrangements
to collect royalties for the use of technology they have developed. They may
also market competitive commercial products on their own or through joint
ventures and may compete with us in recruiting highly qualified scientific
personnel. Further, these institutions may compete with us in recruiting
qualified patients for enrollment in their trials.
We are
pursuing areas of product development in which there is a potential for
extensive technological innovation. Our competitors may succeed in developing
products that are more effective than those we develop. Rapid technological
change or developments by others may result in our potential products becoming
obsolete or non-competitive.
We
lack manufacturing capability.
Other
than for the production of clinical trial material, we currently do not have
manufacturing facilities. Should any of our products receive approval for
marketing, we would likely need to find third party manufacturers to assist in
their production. If we should be unable to find such manufacturers with which
to work on commercially reasonable terms, it could delay or restrict any
potential revenues from such products.
We
use hazardous materials in our research.
As with
most other pharmaceutical companies, our research and development involves the
controlled use of hazardous materials. Our laboratories store and/or produce
carbon monoxide, nitric acid and ammonia. Although we believe that our safety
procedures for handling and disposing of these hazardous materials comply in all
material respects with the standards prescribed by government regulations, the
risk of accidental contamination or injury from these materials cannot be
completely eliminated. In the event of an accident, we could be held liable for
any resulting damages which may or may not be covered by insurance.
17
We
have certain anti-takeover provisions and are also subject to certain Nevada
anti-takeover provisions that may make it difficult for a third party to acquire
us or for stockholders to replace or remove current
management. Further, the board controls a majority of the outstanding
shares.
After the
equity financing in April 2009, the current board represents 61% of the
outstanding shares and on a fully diluted basis a total of 67% as of December
31, 2009.
Additionally,
we have adopted a stockholder rights plan that imposes a significant penalty
upon any person or group that acquires 15% or more of our outstanding common
stock without the approval of our board. Moreover, certain provisions of the
Nevada General Corporation Law that limit our ability to enter into “business
combinations” with certain “interested shareholders” and limit the voting rights
of those stockholders who obtain “control shares” may also act to inhibit a
hostile acquisition of our company. All of these provisions described above are
likely to discourage potential acquisition proposals and delay or prevent a
transaction resulting in a change in control.
18
Item
1B. Unresolved Staff Comments
None.
Item
2. Properties
Pharmos
is headquartered in Iselin, New Jersey, where it leases its executive offices
from which the Company is managed. The New Jersey lease expired on
December 2009. The Company renewed the lease on a short term basis through June
30, 2010 with a provision that the landlord can recapture the space upon giving
the Company thirty days notice.
In the
opinion of management, the New Jersey facilities are sufficient to meet the
current requirements of Pharmos. In addition, management believes
that because of the high level of available commercial office space it has
sufficient ability to renew its present New Jersey lease or obtain suitable
replacement facilities.
Previously,
Pharmos also leased facilities used in the operation of its research,
development and administrative activities in Rehovot, Israel. The
Rehovot lease was terminated effective January 31, 2009 through the exercise of
an early termination clause in the lease. However, the landlord contested that
proper notice had not been given. This matter has been settled with the landlord
in Israel for the release of $38,000 from a restricted cash escrow
account.
Item
3. Legal Proceedings
None
Item
4. Submission of Matters to a Vote of Security Holders
None
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters
and Issuer Purchases of Equity Securities
The
Company’s Common Stock is traded on the Over the Counter Pink Sheets ( OTCBB)
under the symbol “PARS.PK”. The Company was delisted from the Nasdaq Capital
Market in March 2009 as it did not meet the requirements for continued
listing.
The
following table sets forth the range of high and low sales prices per share for
the Common Stock as reported on Nasdaq and OTCBB respectively during the periods
indicated.
|
Year ended December 31,
2009
|
HIGH
|
LOW
|
||||||
|
4th
Quarter
|
$ | .17 | $ | .06 | ||||
|
3rd
Quarter
|
.48 | .15 | ||||||
|
2nd
Quarter
|
.30 | .06 | ||||||
|
1st
Quarter
|
.14 | .03 | ||||||
|
Year ended December 31,
2008
|
HIGH
|
LOW
|
||||||
|
4th
Quarter
|
$ | .20 | $ | .08 | ||||
|
3rd
Quarter
|
.39 | .16 | ||||||
|
2nd
Quarter
|
.53 | .36 | ||||||
|
1st
Quarter
|
.94 | .32 | ||||||
19
The high
and low sales prices for the Common Stock from January 1, 2010 through February
11, 2010 were $.11 and $.07, respectively. The closing price on
February 11, 2010 was $.09.
On
February 11, 2009, there were approximately 296 record holders of the Common
Stock of the Company and approximately 10,401 beneficial owners of the Common
Stock of the Company, based upon the number of shares of Common Stock held in
“street name”.
The
Company has paid no dividends on its Common Stock and does not expect to pay
cash dividends in the foreseeable future. The Company is not under any
contractual restriction as to its present or future ability to pay dividends.
The Company currently intends to retain any future earnings to finance the
growth and development of its business.
PERFORMANCE
GRAPH
The
following graph compares the Company's cumulative stockholder's return for the
five year period ended December 31, 2009 with the cumulative total return of the
Nasdaq Equity Market Index and the Nasdaq Pharmaceuticals Index over the same
period.
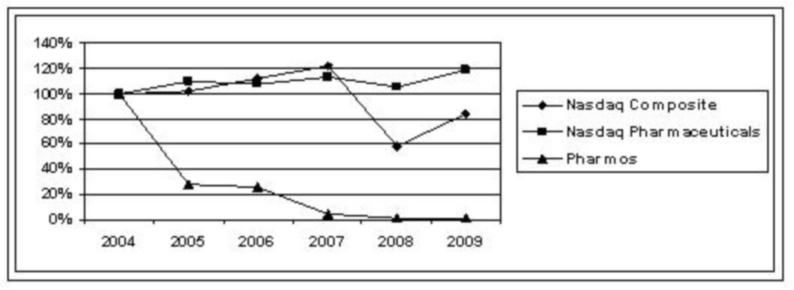
20
Item
6. Selected Financial Data
|
Year
Ended December 31,
|
||||||||||||||||||||
|
2009
|
2008
|
2007
|
2006
|
2005
|
||||||||||||||||
|
Revenues
|
—
|
—
|
—
|
—
|
—
|
|||||||||||||||
|
Operating
expenses
|
$
|
(6,147,384
|
)
|
$
|
(11,100,184
|
)
|
$
|
(17,579,259
|
)
|
$
|
(37,542,519
|
) (1)
|
$
|
(15,708,888
|
)
|
|||||
|
Other
income (expense), net
|
(821,431
|
)
|
(193,348
|
)
|
997,652
|
1,792,775
|
12,288,382
|
(2)
|
||||||||||||
|
Loss
before income taxes
|
(6,968,815
|
)
|
(11,293,532
|
)
|
(16,581,607
|
)
|
(35,749,744
|
)
|
(3,420,506
|
)
|
||||||||||
|
Net
loss
|
(3,054,334
|
)
|
(10,089,406
|
)
|
(15,625,825
|
)
|
(35,136,969
|
)
|
(2,929,872
|
)
|
||||||||||
|
Net
loss applicable to common shareholders
|
$
|
(3,054,334
|
)
(3)
|
$
|
(10,089,406
|
)
(3)
|
$
|
(15,625,825
|
) (3)
|
$
|
(35,136,969
|
)
(3)
|
$
|
(2,929,872
|
)
(3)
|
|||||
|
Net
loss per share applicable to common shareholders - basic and
diluted
|
$
|
(0.06
|
)
|
$
|
(0.39
|
)
|
$
|
(0.61
|
)
|
$
|
(1.74
|
)
|
$
|
(0.15
|
)
|
|||||
|
Total
assets
|
$
|
4,689,022
|
$
|
5,972,164
|
$
|
12,374,959
|
|
$
|
28,393,338
|
$
|
48,990,772
|
|||||||||
|
Long
term obligations
|
$
|
1,000,000
|
$
|
4,044,316
|
$
|
410,594
|
$
|
1,388,306
|
$
|
1,125,551
|
||||||||||
|
Cash
dividends declared
|
—
|
—
|
—
|
—
|
—
|
|||||||||||||||
|
Average
shares outstanding - basic and diluted
|
47,445,014
|
25,934,973
|
25,591,660
|
20,249,714
|
18,974,175
|
|||||||||||||||
|
|
1.
|
The
Company acquired in-process research and development in the Vela
acquisition in October 2006. Vela results are consolidated from October
26, 2006 forward.
|
|
|
2.
|
Includes
a $10.7 million milestone payment received in 2005 related to the sale of
the ophthalmic product line in October
2001.
|
|
|
3.
|
Includes
benefit of sales of Pharmos NJ Net Operating Loss in 2009, 2008, 2007,
2006 and 2005 of $3,914,481, $1,204,126, $955,782, $612,775 and $490,634,
respectively.
|
Item
7. Management’s Discussion and Analysis of Financial Condition and
Results of Operations
This
discussion and analysis of our financial condition and results of operations
contains forward-looking statements that involve risks and uncertainties. We
have based these forward-looking statements on our current expectations and
projections of future events. Such statements reflect our current views with
respect to future events and are subject to unknown risks, uncertainty and other
factors that may cause results to differ materially from those contemplated in
such forward looking statements. In addition, the following discussion should be
read in conjunction with the audited consolidated financial statements and the
related notes thereto included elsewhere in this report.
21
Executive
Summary
The
results for the year ended December 31, 2009 and 2008 were a net loss of $3.1
million and $10.1 million or a loss per share of $.06 and $.39,
respectively.
The
operating expenses for 2009 were $6.1 million, primarily comprised of $4.4
million research and development expenses primarily relating to the commencement
of the Dextofisopam Phase 2b clinical trial and $1.5 million of general and
administration expenses.
Additionally
$0.2 million was charged to in-process research and development as the value of
the 2 million shares issued in November 2009, in connection with a milestone
payment to former Vela shareholders.
In 2009,
the majority of the Company’s research and development expenditures were spent
on the advancement of the Dextofisopam Phase 2b clinical trial which concluded
in September of 2009.
Critical
Accounting Policies
The
preparation of our consolidated financial statements in conformity with
accounting principles generally accepted in the U.S. requires management to make
estimates and assumptions that affect the amounts reported in our consolidated
financial statements and accompanying notes. Future events and their effects
cannot be determined with absolute certainty. Therefore, the determination of
estimates requires the exercise of judgment. Actual results inevitably will
differ from those estimates, and such differences may be material to the
financial statements. The listing below is not intended to be a comprehensive
list of all of our accounting policies. The Company considers certain accounting
policies related to stock-based compensation and the tax valuation allowance to
be critical policies due to the estimation process involved in
each.
In the
quarter ended September 30, 2009, the Company adopted Statement of Financial
Accounting Standards (“SFAS”) No. 168, “The FASB Accounting Standards
Codification (ASC) and Hierarchy of Generally Accepted Accounting Principles”,
which is now the single source of authoritative US GAAP for interim and annual
periods. The adoption had no effects and the codification supersedes all
existing non-SEC standards; new references are included in this report on Form
10-K.
Going
Concern Considerations
The
financial statements have been prepared on a going concern
basis. Except for 2001, the Company has experienced operating losses
every year since inception in funding the research, development and clinical
testing of our drug candidates. The Company had an accumulated deficit of $209.8
million as of December 31, 2009 and expects to continue to incur losses going
forward. Such losses have resulted principally from costs incurred in research
and development and from general and administrative expenses. Previously the
Company had financed its operations with public and private offerings of
securities, advances and other funding pursuant to an earlier marketing
agreement with Bausch & Lomb, grants from the Office of the Chief Scientist
of Israel, research contracts, the sale of a portion of its New Jersey net
operating loss carryforwards (NOL’s), and interest income. Although
the Company received $3.9 million in December 2009 from the sale of their NOL’s
and had approximately $4.6 million of cash and cash equivalents at December 31,
2009, the Company is largely dependent upon achieving a collaboration with a
pharmaceutical partner or raising additional capital to either advance its lead
compound, Dextofisopam, for the treatment of IBS or to advance other earlier
stage intellectual property assets. During the year, the
Company engaged an investment advisor to assist with these strategies but has
not been successful to date. The Company has in the past pursued
various funding and financing options; however management believes that future
funding or financing options may be challenging because of the current
environment.
Also, the
Company does not currently have the finances and resources to complete full
clinical trials for its lead compound, Dextofisopam. As a result it may decide
to embark on smaller clinical trials for Dextofisopam or commence pre-clinical
development on its other intellectual property assets which could further reduce
the company’s current resources.
22
Accounting
for Advance Payments for Goods or Services to be Used in Future Research and
Development Activities.
FASB ASC
Topic 730 requires companies involved in research and development activities to
capitalize such non-refundable advance payments for goods and services pursuant
to an executory contractual arrangement because the right to receive those
services in the future represents a probable future economic benefit. Advance
payments are capitalized until the goods have been delivered or the related
services have been performed. This standard was effective January 1, 2007 under
the previously issued EITF 07-03. At December 31, 2009 and 2008 there
was $0 and $519,000, respectively, in capitalized prepayments.
Equity
based compensation
The
Company follows the provisions of FASB Topic 718 on stock based compensation.
Effective January 1, 2006, the Company adopted the provisions which established
the financial accounting and reporting standards for stock-based compensation
plans. This required the measurement and recognition of compensation expense for
all stock-based awards made to employees and directors, including employee stock
options and restricted stock units, and employee stock purchases related to the
ESPP. Under the provisions of ASC Topic 718, stock-based compensation cost is
measured at the grant date, based on the fair value of the award, and is
recognized as an expense over the requisite service period of the entire award
(generally the vesting period of the award). As of
December 31, 2009, the total compensation costs related to non-vested awards not
yet recognized is $460,000 which will be recognized over the next three and
one-half years.
Options
issued to non-employees other than directors are accounted for under the fair
value method in accordance with ASC Topic 718 “Accounting for Equity Instruments
that are Issued to Other than Employees for Acquiring, or in Conjunction with
Selling Goods or Services.” Under the fair value method, compensation cost is
measured at the grant date of the option based on the value of the award using
the Black-Scholes method. Compensation cost is periodically remeasured as the
underlying options vest in accordance with ASC Topic 718 and is recognized over
the service period.
Tax
Valuation Allowance
The
Company has assessed the likelihood of realizing future taxable income and has
determined that a 100% deferred tax valuation allowance is deemed
necessary. In the event the Company were to determine that it would
be able to realize its deferred tax asset, an adjustment to the valuation
allowance would increase income or decrease the loss in the period such
determination is made.
Sale
of New Jersey Net Operating Losses
The
Company participates in the State of New Jersey’s Technology Business Tax
Certificate Transfer Program (the “Program”) administered by the New Jersey
Economic Development Authority. The Program allows qualified technology and
biotechnology businesses located in New Jersey to sell unused amounts of net
operating loss carryforwards and defined research and development tax credits
for cash. In 2009 the Company received net proceeds of $3,914,481 from the sale
of New Jersey Net Operating Losses for the years 2004 through 2008 and there is
$6,890,533 left from 2008 of operating losses to sell in future years, which at
the 9% state tax rate equates to $620,148.
The
proceeds will enable the Company to continue operations with its main focus on
seeking a pharmaceutical partner to further the development of
Dextofisopam.
23
Subsequent
Events
In May
2009, the FASB established general standards of accounting for and disclosures
of events that occur after the balance sheet date but before financial
statements are issued or are available to be issued (ASC Topic 855). It requires
the disclosure of the date through which an entity has evaluated subsequent
events and the basis for selecting that date, that is, whether that date
represents the date the financial statements were issued or were available to be
issued. The Company's adoption of these standards had no material impact on its
financial position, results of operations and cash flows.
The
Company has evaluated events and transactions that occurred between December 31,
2009 and February 22, 2010, which is the date the financial statements were
issued, for possible disclosure and recognition in the financial
statements.
Results
of Operations
Years
Ended December 31, 2009 and 2008
The
Company recorded no product sales revenue and cost of sales during 2009 and
2008.
Research
and development efforts in 2009 and 2008 were focused primarily on the
Dextofisopam Phase IIb trial initiated in June 2007 with enrollment ongoing
through April of 2009. In July of 2009 the trial officially ended and in
September the results were released. The 2009 research and development expenses
exclude expenses that were incurred in Israel in 2008 as those operations were
closed in October 2008. Additionally the dextofisopam clinical trial costs
extended through all of 2008 and only through September 2009 when the results
were released. Consequently the research and development expenses in 2009 were
significantly less than in 2008.
The
Company considers major research & development projects to be those projects
that have reached at least Phase II level of clinical development. In June 2007
the Company announced that patient screening had commenced in the Phase 2b trial
for Dextofisopam for IBS. Through December 31, 2009 expenditures on this trial,
from inception, amounted to $15,905,060.
During
the year 2008 and 2009 the Company’s operations were considerably scaled back
from 2007. The main focus was the continued enrollment of the Dextofisopam Phase
2b trial for IBS, using about 70 sites in the US. The Company’s operations in
Israel were closed effective October 31, 2008. Prior to closure a limited amount
of development work was performed on the CB2 program, primarily focused on
enabling a license or sale. The Nano Emulsion Phase 2a clinical trial was
completed and did not meet its endpoints. Further work on the Nano Emulsion drug
delivery program was discontinued.
During
2008 and 2009 the Company lowered its general and administrative costs by
streamlining operations, focusing on reducing all non core costs.
Gross
expenses for other research and development projects in early stages of
development for the year ended December 31, 2009 and 2008 were $0 and
$1,385,284, respectively. Research & development (R&D)
expenses decreased by $4,606,105 or 51% from $9,028,705 in 2008 to $4,422,600 in
2009 due to the curtailment of research and development activities at the Israel
location and focusing the financial assets on the Dextofisopam Phase IIb
trial. The decline reflects decreases in virtually every research and
development category. The primary reductions include a $762,000 reduction in
payroll, a $411,000 reduction in professional fees and consulting, a $2,976,000
reduction in clinical fees and a reduction of $457,000 in various facility
related expenses.
24
In
process research and development costs which were related to the Vela milestone
increased by $180,000 from $0 in 2008 to $180,000 in 2009. On April 9, 2009 the
last patients were enrolled in the Phase 2b trial thus triggering the following
milestone: $1 million cash and 2 million shares of Pharmos common stock: Final
patient enrolled in Phase 2b trial. The expense of the milestone of $180,000 was
reflected in the Q1 2009 results while the payment of the cash portion of the
milestone was deferred under an amendment to the acquisition agreement. Under
the terms of the Vela acquisition agreement as amended, the 2 million shares
were issued on November 2, 2009. The cash portion was also expensed in Q1 2009
but was reversed in Q4 2009 since it is not probable that the amended terms will
be met in the foreseeable future, which includes receipt of at least $10 million
cash upfront payment. Since the trial results were not successful, no other
milestones have been achieved.
General
and administrative expenses decreased by $428,773 or 22%, from $1,965,243 in
2008 to $1,536,470 in 2009. The decrease in general and
administrative expenses is due to a reduction in expenses in every expense
category. Significant reductions occurred in employee compensation
($200,000), professional & consulting fees ($294,000), facility related
expenses $(90,000) and offset by an increase to the miscellaneous expenses of
$155,000 when comparing 2009 to 2008. The overall decline in employee
headcount from 14 employees at the beginning of 2008 to 4 employees at the end
of 2009 accounts for the decline in the employee compensation. Lower
professional & consulting fees in 2009 are due to reductions in the
utilization of outside counsel of $102,000, a change in auditors which resulted
in a $71,000 reduction and lower fees of $121,000 related to our Israel entity.
Finally we incurred an increase in miscellaneous expenses in 2009 over 2008 as
credits against miscellaneous expenses were greater in 2008 than in 2009. In
2008 we recorded gains of fixed assets and equipment lease income of $259,000
and in 2009 we reduced our expenses of $100,000 for an accrued marketing expense
that will not be paid.
Other
expense net, increased by $628,083 from an expense of $193,348 in 2008 to an
expense of $821,431 in 2009. Interest expense decreased by $265,202
from $490,537 in 2008 to $225,335 in 2009. The decrease in 2009 interest expense
results from the retirement of convertible debentures in April
2009. Interest income decreased by $247,339 from $255,751 in 2008 to
$8,412 in 2009 due to the utilization of invested balances and lower interest
rates in 2009. Debt conversion expense increased by $596,104 from $0 in 2008 to
$596,104 in 2009 is attributable to the convertible debentures retired in April
2009. The conversion of the debentures at a reduced conversion price resulted in
a debt conversion expense of $596,104 as the original conversion price of $0.70
was reduced to $0.275. Other income (expense) increased by $49,842 from income
of $41,438 in 2008 to an expense of (8,404) in 2009. The majority of income in
2008 was attributable to a $30,000 sale of material from our Israel facility and
the expenses in 2009 were attributable to translation losses on foreign
currency.
The
Company had an increase in income tax benefit by $2,710,355 from $1,204,126 in
2008 to $3,914,481 in 2009. The income tax benefit represents funds derived from
the sale of Pharmos’ New Jersey State net operating losses.
Years
Ended December 31, 2008 and 2007
The
Company recorded no product sales revenue and cost of sales during 2008 and
2007.
Research
efforts in 2008 and 2007 have been focused primarily on the Dextofisopam Phase
IIb trial initiated in June 2007 with enrollment ongoing throughout
2008. The major decrease in year-to-date operating expenses
year–over-year reflects the curtailment of in-house research & development
activities in 2008 and the manpower reduction initiatives taken in the second
half of 2007 and the 2008 year. The Company’s operations in Israel were closed
effective October 31, 2008 and all staff was terminated. Prior to that time in
2008 research and development activity was very limited.
25
The
Company considers major research & development projects to be those projects
that have reached at least Phase II level of clinical development. In
June 2007 the Company announced that patient screening had commenced in the
Phase 2b trial for Dextofisopam for IBS with a target of 480 patients. Through
December 31, 2008 expenditures on this trial, from inception, amounted to
$11,861,481.
During
the year 2008 the Company’s operations were considerably scaled back from 2007.
The main focus was the continued enrollment of the Dextofisopam Phase 2b trial
for IBS, using about 70 sites in the US. The Company’s operations in Israel were
closed effective October 31, 2008. Prior to closure a limited amount of
development work was performed on the CB2 program, primarily focused on enabling
a license or sale. The Nano Emulsion Phase 2a clinical trial was completed and
did not meet its endpoints. Further work on the Nano Emulsion drug delivery
program was discontinued.
During
2008 the Company lowered its general and administrative costs by streamlining
operations, focusing on reducing all non core costs. Included in the
2007 general and administrative costs are costs relating to the departure of
three senior executives. These payments were made under contractual agreements.
In 2008 aggregate general and administrative costs are lower than in 2007 due in
part to cost management efforts, several staff reduction initiatives, closing of
the Israel location and the scaling back of activities not core to the Company’s
research and development efforts.
Gross
expenses for other research and development projects in early stages of
development for the year ended December 31, 2008 and 2007 were $1,385,284 and
$4,350,348, respectively. Research & development (R&D) gross
expenses decreased by $2,428,861 or 21% from $11,457,566 in 2007 to $9,028,705
in 2008 due to the curtailment of research and development activities at the
Israel location and focusing the financial assets on the Dextofisopam Phase IIb
trial. The Company recorded research and development grants received
from the Office of the Chief Scientist of Israel’s Ministry of Industry and
Trade of $0 and $812,042 during 2008 and 2007, respectively, which reduced
research and development expenses. The decrease in grants is directly related to
the decrease in the underlying eligible activity in the Israel location for the
grants in 2008 over 2007 as the Company focused more research funds on the US
based Phase 2b
clinical trial of Dextofisopam. Total
research and development expenses, net of grants, decreased by $1,616,819 or 15%
from $10,645,524 in 2007 to $9,028,705 in 2008.
General
and administrative expenses decreased by $4,733,358 or 71%, from $6,698,601 in
2007 to $1,965,243 in 2008. The decrease in general and
administrative expenses is due to a reduction in expenses in every expense
category. Significant reductions were seen in employee compensation
($2,714,000) and professional & consulting fees ($953,000) when comparing
2008 to 2007. The decline in employee compensation reflects the
departure of several executives in 2007, their related severance packages paid
in 2007 and the overall decline in employee headcount from 51 employees at the
beginning of 2007 to 5 employees at the end of 2008. Lower
professional & consulting fees in 2008 are primarily due to 2007 non
recurring expenses for consulting agreements with a former employee of $410,000,
reductions in the utilization of outside counsel of $170,000 and a 2007
consulting cost of $99,000 related to an IRS Section 382 Federal Net Operating
Loss analysis.
Other
income (expense) net, decreased by $1,191,000 from income of $997,652 in 2007 to
expense of $193,348 in 2008. Interest expense increased by $490,537
from $0 in 2007 to $490,537 in 2008. The increase in 2008 interest expense is a
result of the convertible debentures issued in January 2008. Interest
income decreased by $682,561 from $938,312 in 2007 to $255,751 in 2008 due to
the utilization of invested balances and lower interest rates in
2008.
The
Company had an increase in income tax benefit by $248,344 from $955,782 in 2007
to $1,204,126 in 2008. The income tax benefit represents funds derived from the
sale of Pharmos’ New Jersey State net operating losses.
26
Liquidity
and Capital Resources
Except
for 2001, the Company has experienced operating losses every year since
inception in funding the research, development and clinical testing of our drug
candidates. The Company had an accumulated deficit of $209.8 million as of
December 31, 2009 and expects to continue to incur losses going forward. Such
losses have resulted principally from costs incurred in research and development
and from general and administrative expenses. Previously the Company had
financed its operations with public and private offerings of securities,
advances and other funding pursuant to an earlier marketing agreement with
Bausch & Lomb, grants from the Office of the Chief Scientist of Israel,
research contracts, the sale of a portion of its New Jersey net operating loss
carryforwards (NOL’s), and interest income. Although the Company
received $3.9 million in December 2009 from the sale of their NOL’s and had
approximately $4.6 million of cash and cash equivalents at December 31, 2009,
the Company is largely dependent upon achieving a collaboration with a
pharmaceutical partner or raising additional capital to either advance its lead
compound, Dextofisopam, for the treatment of IBS or to advance other earlier
stage intellectual property assets. During the year, the
Company engaged an investment advisor to assist with these strategies but has
not been successful to date. The Company has in the past pursued
various funding and financing options; however management believes that future
funding or financing options may be challenging because of the current
environment.
Also, the
Company does not currently have the finances and resources to complete full
clinical trials for its lead compound, Dextofisopam. As a result it may decide
to embark on smaller clinical trials for Dextofisopam or commence pre-clinical
development on its other intellectual property assets which could further reduce
the company’s current resources.
The
following table describes the Company’s liquidity and financial position on
December 31, 2009, and December 31, 2008:
|
December 31, 2009
|
December 31, 2008
|
|||||||
|
Working
capital
|
$ | 4,238,033 | $ | 4,232,549 | ||||
|
Cash
and cash equivalents
|
$ | 4,629,486 | $ | 4,730,282 | ||||
|
Convertible
debentures
|
$ | 1,000,000 | $ | 4,000,000 | ||||
Current
working capital position
As of
December 31, 2009, the Company had working capital of $4.2 million consisting of
current assets of $4.6 million and current liabilities of $0.4 million. This
represents no net change from its working capital of $4.2 million on current
assets of $5.8 million and current liabilities of $1.6 million as of December
31, 2008.
Current
and future liquidity position
At
December 31, 2009 the Company had approximately $4.6 million of cash and cash
equivalents, which at current operating levels should be sufficient to continue
operations at least through December 31, 2011. However, the Company’s success
will be dependent upon achieving a pharmaceutical partnership for the
advancement of its lead compound, Dextofisopam for the treatment of IBS.
Further, the Company may embark on smaller clinical trials, the expense of which
would reduce cash resources available.
The
Company has in the past pursued various funding options, including additional
equity offerings, strategic corporate alliances, and business combinations, as
well as grants and the sale of some of its New Jersey net operating loss carry
forwards. Future equity financings will be challenging because of the low market
capitalization of the Company and the move from the Nasdaq market to trading on
the over the counter Pink Sheets in March 2009.
27
Cash
At
December 31, 2009, cash and cash equivalents totaled $4.6 million. At December
31, 2008 cash and cash equivalents totaled $4.7 million. This net decrease in
cash of $0.1 million was due to the utilization of cash for operating activities
offset by the proceeds from the sale of the NJ NOL and the equity
financing. The cash and cash equivalents will be used to finance
future operating expenses.
Operating
activities
Net cash
used in operating activities for 2009 was $2.3 million compared to $11.0 million
for 2008. The decrease is primarily attributed to lower research and development
expenses and general and administrative expenses incurred in 2009. Research and
development expenses declined from $9.0 million in 2008 to $4.4 million in 2009
while general and administrative expenses declined from $2.0 million in 2008 to
$1.5 million in 2009.
Capital
expenditures
Our
capital expenditures for property, plant and equipment for 2009, 2008 and 2007
totaled approximately $0, $2,000 and $177,000 respectively for normal
replacements and improvements.
Investing
activities
In 2009
the only significant activity was the cash received related to the disposition
of the Israeli fixed assets.
Financing
activities
In April
2009 the proceeds from the issuance of common stock and warrants, net of
issuance costs, were recorded in the amount of $1,779,777.
Common
Stock Transactions
At the
closing of the Vela Pharmaceuticals Inc. acquisition on October 25, 2006, the
Company issued 6.5 million shares of common stock and paid $6 million to Vela
shareholders. Pharmos also agreed to reimburse Vela for $679,000 of
operating expenses from July 1, 2006 through closing. The amended
Merger Agreement also includes additional performance-based milestone payments
to the Vela stockholders related to the development of Dextofisopam, aggregating
up to an additional $8 million in cash and the issuance of up to an additional
13,500,000 shares of Pharmos common stock. In the event that such
shares or payments are issued or funded in future periods, a determination will
then be made as to whether the values are to be written off as in-process
research and development and charged to results of operations; any such future
charge could be material. None of the conditions requiring issuance
of these contingent shares or funding these payments had been met as of December
31, 2008 except for a $1.0 million milestone payment made by Pharmos due upon
the study’s commencement.
On
January 3, 2008, the Company entered into an Amendment to the Merger Agreement
relating to its acquisition of Vela. The Amendment defers the payment by the
Company of certain cash milestones payable by the Company to the former
stockholders of Vela upon (A) the enrollment of the final patient in the
Company’s current Phase 2b clinical trial for Dextofisopam ($1 million payment
obligation) and (B) the successful completion of such Phase 2b trial ($2 million
payment obligation). Payment of such cash milestones will be deferred until such
time as (X) the Company has successfully entered into a strategic collaboration
or licensing agreement with a third party for the development of Dextofisopam
resulting in an upfront cash fee of at least $10 million, or a financing with
net proceeds of at least such amount, and (Y) payment of one or both of the cash
milestones would still leave the Company with at least one year’s operating
cash. Additionally, the Company’s obligations to issue to the former Vela
stockholders 2 million shares of Common Stock after final patient enrollment in
the Dextofisopam Phase 2b clinical trial and was deferred until November 2009,
and the 2 million patient enrollment milestone shares were issued in November
2009. Finally the 2.25 million shares of Common
Stock after a successful Phase 2b trial milestone was not met as the Phase 2b
dextofisopam trial did not meet its primary endpoints, as it did not meet the
definition of a successful trial. Therefore the 2.25 million shares related to
this milestone are not payable.
28
Other
In 2009,
2008, and 2007, the Company sold $49,707,690, $15,290,510 and $12,136,911,
respectively, of its State Net Operating Loss carryforwards under the State of
New Jersey’s Technology Business Tax Certificate Transfer Program (the Program).
The Program allows qualified technology and biotechnology businesses in New
Jersey to sell unused amounts of net operating loss carryforwards and defined
research and development tax credits for cash. The proceeds from the sale in
2009, 2008, and 2007 were $3,914,481, $1,204,128 and $955,782, respectively and
such amounts were recorded as a tax benefit in the consolidated statements of
operations. The New Jersey State Governor must approve the Program each year on
July 1st within the approval of the annual state budget. The maximum
amount of benefits a participant can apply to sell is capped at $10,000,000
annually per company. The remaining NOL’s which qualify under the program
through 2008 amount to $6,890,541. When the 2009 tax returns are finalized they
too will be included in the Company’s application to sell losses. We cannot be
certain if we will be able to sell any of our remaining or future carryforwards
under the Program.
Under
Internal Revenue Code Section 382 rules, a change in ownership can occur
whenever there is a shift in ownership by more than 50 percentage points by one
or more five-percent shareholders within a three-year period. When a
change of ownership is triggered, the Company’s net operating losses (NOL) asset
may be impaired. The Company believes that substantially all of its
Federal NOL is subject to restrictions as to future use in accordance with IRS
Code Section 382.
Commitments
and Long Term Obligations
The
table below sets out our current contractual obligations.
|
Payments
Due by Period
|
||||||||||||||||||||||||
|
Total
|
Less
than 1 Year
|
1 –
3
Years
|
3 –
5
Years
|
More
Than
5 Years
|
Undetermined
|
|||||||||||||||||||
|
Operating
Leases
|
$ | 45,945 | $ | 45,945 | $ | - | $ | - | $ | - | $ | - | ||||||||||||
|
Convertible
Debenture Interest
|
283,333 | 100,000 | 183,333 | |||||||||||||||||||||
|
Convertible
Debenture
|
1,000,000 | - | 1,000,000 | - | - | - | ||||||||||||||||||
|
Total
|
$ | 1,329,278 | $ | 145,945 | $ | 1,183,333 | $ | - | $ | - | $ | - | ||||||||||||
In
connection with the acquisition of Vela Pharmaceuticals which closed on October
25, 2006 the Company is obligated to pay certain performance based milestones
connected to the development of Dextofisopam.
The
remaining potential future milestones are as follows:
|
|
·
|
$1
million cash + 2 million shares of Pharmos common stock: Final patient
enrolled in Phase 2b trial (1)
|
|
|
·
|
$2
million cash + 2.25 million shares: Successful completion of Phase 2b
(milestone not met)
|
|
|
·
|
$2
million + 2 million shares: NDA
submission
|
|
|
·
|
$2
million cash +2.25 million shares: FDA
approval
|
|
|
·
|
1
million shares: Approval to market in Europe or
Japan
|
|
|
·
|
4
million shares: $100 million sales of Dextofisopam, when and if approved,
in any 12-month period
|
29
(1) The milestone related the
final patient was enrolled in the Dextofisopam Phase 2b trial was booked in the
first quarter of 2009 as all probability criteria were met. The milestone had
two components, a cash portion of $1,000,000 and a share portion of 2,000,000
shares valued at $180,000. The total charge was $1,180,000. The
shares were issued in November 2009. The payment of the cash portion was
deferred until such time as 1) the Company successfully entered into a strategic
collaboration or licensing agreement with a third party for the development of
Dextofisopam resulting in an upfront cash fee of at least $10 million, and 2)
payment of the cash milestone would still leave the Company with one year’s
operating cash.
The
Company recorded the milestone in the first quarter as it met the accounting
requirements of under ACS 450. The results of the Phase 2b trial were announced
in September 2009 and reported that while there was clearly drug activity, the
trial did not achieve its primary endpoints. Cowen and Company were subsequently
engaged to help the Company achieve a pharmaceutical partnership. Under the
terms of the Vela acquisition agreement as amended, the 2 million shares were
issued on November 2, 2009. The cash portion was also expensed in Q1 2009 but
was reversed in Q4 2009 since it is not probable that the amended terms will be
met in the foreseeable future, which includes receipt of at least $10 million
cash upfront payment. Since the trial results were not successful, no other
milestones have been achieved.
Management
believes that the current cash and cash equivalents, totaling approximately $4.6
million as of December 31, 2009, will be sufficient to support the Company’s
currently planned continuing operations through at least December 31,
2011.
The
Company’s strategic focus is to seek a pharmaceutical partnership for its lead
compound Dextofisopam for IBS-D. The Company may pursue strategic corporate
alliances, and / or business combinations to advance the development of
Dextofisopam.
New
accounting pronouncements
Recently
Issued Accounting Standards
In August
2009, the FASB issued Accounting Standards Update No. 2009-05, Measuring Liabilities at Fair
Value ("ASU 2009-05"). ASU 2009-05 amends Accounting Standards
Codification Topic 820, Fair
Value Measurements. Specifically, ASU 2009-05 provides clarification that
in circumstances in which a quoted price in an active market for the identical
liability is not available, a reporting entity is required to measure fair value
using one or more of the following methods: (1) a valuation technique that
uses (a) the quoted price of the identical liability when traded as an
asset or (b) quoted prices for similar liabilities or similar liabilities
when traded as assets and/or (2) a valuation technique that is consistent
with the principles of Topic 820 of the Codification (e.g. an income
approach or market approach). ASU 2009-05 also clarifies that when estimating
the fair value of a liability, a reporting entity is not required to adjust to
include inputs relating to the existence of transfer restrictions on that
liability. The adoption of this standard did not have an impact on the Company's
financial statements other than the requisite disclosures.
In
October 2009, the FASB issued ASU No. 2009-13, Multiple-Deliverable Revenue
Arrangements ("ASU 2009-13"). ASU 2009-13, amends existing revenue
recognition accounting pronouncements that are currently within the scope of
Codification Subtopic 605-25 (previously included within EITF 00-21, Revenue Arrangements with Multiple
Deliverables ("EITF 00-21"). The consensus to ASU 2009-13 provides
accounting principles and application guidance on whether multiple deliverables
exist, how the arrangement should be separated, and the consideration allocated.
This guidance eliminates the requirement to establish the fair value of
undelivered products and services and instead provides for separate revenue
recognition based upon management's estimate of the selling price for an
undelivered item when there is no other means to determine the fair value of
that undelivered item. EITF 00-21 previously required that the fair value
of the undelivered item be the price of the item either sold in a separate
transaction between unrelated third parties or the price charged for each item
when the item is sold separately by the vendor. Under EITF 00-21, if the
fair value of all of the elements in the arrangement was not determinable, then
revenue was deferred until all of the items were delivered or fair value was
determined. This new approach is effective prospectively for revenue
arrangements entered into or materially modified in fiscal years beginning on or
after June 15, 2010 and
30
allows
for retroactive application. The Company is currently evaluating the potential
impact of this standard on its financial position and results of operations.
In May
2008, the FASB issued standards relating to "Accounting for Convertible Debt
Instruments That May Be Settled in Cash upon Conversion (Including Partial Cash
Settlement)" These are covered by ASC Topic 470 (formerly FSP APB 14-1"). Topic
470 requires the issuer of certain convertible debt instruments that may be
settled in cash (or other assets) on conversion to separately account for the
liability (debt) and equity (conversion option) components of the instrument in
a manner that reflects the issuer's non-convertible debt borrowing rate. The
standard was effective for fiscal years beginning after December 15, 2008 on a
retroactive basis. The Company adopted the standard in the first quarter of
2009. The adoption
of this standard did not have an impact on the Company's financial position or
results of operations.
Item
7A. Quantitative and Qualitative Disclosures About Market Risk
We
assessed our vulnerability to certain market risks, including interest rate risk
associated with financial instruments included in cash and cash equivalents,
short term investments and restricted cash. Due to the short-term nature of the
cash and cash equivalents, short term investments and restricted cash, we have
determined that the risks associated with interest rate fluctuations related to
these financial instruments do not pose a material risk to us.
Item
8. Financial Statements and Supplementary Data
The
information called for by this Item 8 is included following the “Index to
Consolidated Financial Statements” contained in this Annual Report on Form
10-K.
Item
9. Changes in and Disagreements with Accountants on Accounting and
Financial Disclosure
On
October 19, 2009, Pharmos Corporation dismissed its independent registered
public accounting firm, PricewaterhouseCoopers LLP. The dismissal was
approved by the Pharmos Audit Committee. During the two most recent
fiscal years and through October 19, 2009, there have been no disagreements
between Pharmos and PricewaterhouseCoopers LLP on any matter of accounting
principles or practices, financial statement disclosure, or auditing scope or
procedure, which disagreements if not resolved to the satisfaction of
PricewaterhouseCoopers LLP would have caused them to make reference thereto in
their reports on the financial statements for such years. During the
two most recent fiscal years and through October 19, 2009, there were no
reportable events (as defined in Item 304(a)(1)(v)) of Regulation
S-K. Neither of the reports of PricewaterhouseCoopers LLP for the
past two years contained an adverse opinion or disclaimer of opinion, or was
qualified or modified as to uncertainty, audit scope or accounting principles,
except that their report dated February 25, 2009 contained an explanatory
paragraph expressing substantial doubt about Pharmos' ability to continue as a
going concern. On October 20, 2009, Pharmos Corporation engaged
Friedman LLP as its new independent accounting firm.
31
Item
9A(T). Controls and Procedures
Evaluation
of Disclosure Controls and Procedures
Our
management, with the participation of our principal executive officer and
principal financial officer, has evaluated the effectiveness of the design and
operation of our disclosure controls and procedures (as defined in Rule
13a-15(e) under the Securities Exchange Act of 1934, as amended (the “Act”)) as
of the end of the period covered by this annual report on Form 10-K. Based on
this evaluation, our principal executive officer and principal financial officer
concluded that these disclosure controls and procedures were effective as of
such date, at a reasonable level of assurance, in ensuring that the information
required to be disclosed by our company in the reports we file or submit under
the Act is (i) accumulated and communicated to our management (including the
principal executive officer and principal financial officer) in a timely manner,
and (ii) recorded, processed, summarized and reported within the time periods
specified in the SEC’s rules and forms.
Internal
Control Over Financial Reporting
Management
is responsible for establishing and maintaining adequate internal control over
financial reporting, as such term is defined in Exchange Act Rules 13a-15(f).
Under the supervision and with the participation of our management, including
our Chief Executive Officer and Chief Financial Officer, we conducted an
evaluation of the effectiveness of Pharmos’ internal control over financial
reporting based on the criteria in Internal Control – Integrated Framework
issued by the Committee of Sponsoring Organizations of the Treadway Commission.
Based on our evaluation, management has concluded that Pharmos’ internal control
over financial reporting was effective as of December 31, 2009. This
annual report does not include an attestation report of our registered public
accounting firm regarding internal control over financial reporting pursuant to
temporary rules of the Securities and Exchange Commission.
Changes
in Internal Control Over Financial Reporting
There was
no change in our internal control over financial reporting (as defined in Rule
13a-15(f) under the Securities Exchange Act of 1934, as amended) during our last
fiscal quarter that has materially affected, or is reasonably likely to
materially affect, our internal control over financial reporting.
Item
9B. Other Information
None.
32
PART
III
Item
10. Directors, Executive Officers and Corporate
Governance
The
directors, officers and key employees of the Company are as
follows:
|
Name
|
Age
|
Position
|
|
Robert
F. Johnston
|
73
|
Executive
Chairman of the Board of Directors
|
|
S.
Colin Neill
|
63
|
President,
Chief Financial Officer, Secretary and Treasurer
|
|
Srinivas
Akkaraju, MD, Ph.D*
|
41
|
Director
|
|
Anthony
B. Evnin, Ph.D*
|
68
|
Director
|
|
Charles
W. Newhall III
|
65
|
Director
|
|
*
|
Members
of the Audit Committee
|
Robert F. Johnston became
Executive Chairman of the Board of Directors of Pharmos in January 2008. Mr.
Johnston, a venture capitalist, is President of Johnston Associates which he
founded in 1968 to provide financing for emerging companies in the biotechnology
and healthcare fields. Mr. Johnston was a founder and Chairman of Vela
Pharmaceuticals, Inc., which merged into Pharmos in late 2006, and has founded
numerous public companies including Sepracor, Cytogen, I-STAT (sold to Abbott),
Ecogen, Genex and Envirogen (sold to Shaw Environmental). He also played an
active and key role in the early formations of private companies such as
Sonomed, Immunicon (sold to J&J), PharmaStem (formerly Biocyte), ExSAR and
Targent. Mr. Johnston served as CEO of Cytogen from July 1988 to April 1989. He
is also a member of the Advisory Council of the Department of Molecular Biology
at Princeton University. Mr. Johnston is a founder and President of
Educational Ventures, a foundation focused on funding improvements in the
educational system; and Vice-Chairman of Center for Education Reform (CER) an
advocate for charter schools. Mr. Johnston received his B.A from Princeton
University and his M.B.A. from New York University.
S. Colin Neill became
President of Pharmos in January 2008, and has served as Chief Financial Officer,
Secretary, and Treasurer of Pharmos since October 2006. Prior to becoming
President, he also served as Senior Vice President from October 2006 to January
2008. From September 2003 to October 2006, Mr. Neill served as Chief Financial
Officer, Treasurer and Secretary of Axonyx Inc., a biopharmaceutical company
that developed products and technologies to treat Alzheimer's disease and other
central nervous system disorders, where he played an integral role in the merger
between Axonyx and TorreyPines Therapeutics Inc., a privately-held
biopharmaceutical company. Mr. Neill served as Senior Vice President, Chief
Financial Officer, Secretary and Treasurer of ClinTrials Research Inc., a $100
million publicly traded global contract research organization in the drug
development business, from 1998 to its successful sale in 2001. Following that
sale from April 2001 to September 2003 Mr. Neill served as an independent
consultant assisting small start-up and development stage companies in raising
capital. Earlier experience was gained as Vice President Finance and Chief
Financial Officer of BTR Inc., a $3.5 billion US subsidiary of BTR plc, a
British diversified manufacturing company, and Vice President Financial Services
of The BOC Group Inc., a $2.5 billion British owned industrial gas company with
substantial operations in the health care field. Mr. Neill served four years
with American Express Travel Related Services, first as chief internal auditor
for worldwide operations and then as head of business planning and financial
analysis. Mr. Neill began his career in public accounting with Arthur Andersen
LLP in Ireland and later with Price Waterhouse LLP as a senior manager in New
York City. He also served with Price Waterhouse for two years in Paris, France.
Mr. Neill graduated from Trinity College, Dublin with a first class honors
degree in Business/Economics and he holds a masters degree in Accounting and
Finance from the London School of Economics. He is a Certified Public Accountant
in New York State and a Chartered Accountant in Ireland. Mr. Neill serves on the
board of Pro Pharmaceuticals, Inc. and from April 2004 to June 2008 on the board
of OXIS International, Inc.
33
Srinivas Akkaraju, M.D.,
Ph.D., a director since October 2006, is a Managing Director of New Leaf
Venture Partners. Dr. Akkaraju joined New Leaf in January 2009. Previously
he was a managing director of Panorama Capital, LLC, a private equity firm
founded by the former venture capital investment team of J.P. Morgan Partners,
LLC, and a private equity division of JPMorgan Chase & Co. Panorama Capital
is advising J.P. Morgan Partners as to its investment in the Company. Prior to
August 1, 2006, Dr. Akkaraju was a Partner with J.P. Morgan Partners, LLC which
he joined in April 2001. Prior to JPMorgan Partners, LLC, from October 1998 to
April 2001, Dr. Akkaraju was in the Business and Corporate Development group at
Genentech, Inc. where he served in various capacities, most recently as Senior
Manager and project team leader for one of Genentech’s clinical development
products. Dr. Akkaraju is currently a member of the Board of Directors of
Seattle Genetics, Inc., and several private biotechnology companies. Dr.
Akkaraju received his undergraduate degrees in Biochemistry and Computer Science
from Rice University and his M.D. and Ph.D. in Immunology from Stanford
University.
Anthony B. Evnin, Ph.D., a
director since October 2006, is a Partner of Venrock, a venture capital firm,
where he has been a Partner since 1975. He is currently a member of the Board of
Directors of Icagen, Inc. and Infinity Pharmaceuticals, Inc. as well as being on
the Board of Directors of a number of private companies. Dr. Evnin is a Trustee
of The Rockefeller University, a Trustee Emeritus of Princeton University, and a
Member of the Board of Overseers of Memorial Sloan-Kettering Cancer Center. He
received an A.B. in Chemistry from Princeton University and a Ph.D. in Chemistry
from the Massachusetts Institute of Technology.
Charles W. Newhall, III, a
director since October 2006, co-founded New Enterprise Associates (NEA). Founded
in 1977, Baltimore-based NEA is one of the largest Venture Capital firms in the
United States. To date Mr. Newhall has served as a director of over 40 venture
backed companies. Many have gone public and have been acquired. Several of these
companies achieved market capitalizations in excess of $1 billion. He also
started several healthcare information technology companies like PatientKeeper,
TargetRx, and LifeMetrix. Some of his current board memberships include Vitae
Pharmaceuticals, Supernus Pharmaceuticals, Bravo Health, TargetRx, Sensors for
Medicine and Science, and BrainCells Inc. In 1986 he founded the Mid-Atlantic
Venture Capital Association (MAVA), which now has over 80 venture capital firms
that are members, and is one of the most active regional venture associations in
the country. He is Chairman Emeritus of MAVA. Before NEA, Mr. Newhall was a Vice
President of T. Rowe Price. He served in Vietnam commanding an independent
platoon including an initial reconnaissance of Hamburger Hill. His decorations
include the Silver Star and Bronze Star V (1st OLC). He received an MBA from
Harvard Business School, and an Honors Degree in English from the University of
Pennsylvania.
Role
of the Board; Corporate Governance Matters
It is the
paramount duty of the Board of Directors to oversee the Chief Executive Officer
and other senior management in the competent and ethical operation of the
Company on a day-to-day basis and to assure that the long-term interests of the
shareholders are being served. To satisfy this duty, the directors set standards
to ensure that the Company is committed to business success through maintenance
of the highest standards of responsibility and ethics.
Members
of the Board bring to the Company a wide range of experience, knowledge and
judgment. The governance structure in the Company is designed to be a working
structure for principled actions, effective decision-making and appropriate
monitoring of both compliance and performance. The key practices and procedures
of the Board are outlined in the Company’s Code of Ethics and Business Conduct,
which is available on the Company’s website at www.pharmoscorp.com. Click
“Investors,” and then “Corporate Governance.”
34
Board
Committees
The Board
has a standing Compensation Committee, Governance and Nominating Committee and
Audit Committee.
The
Compensation Committee is primarily responsible for reviewing the compensation
arrangements for the Company’s executive officers, including the Chief Executive
Officer, and for administering the Company’s stock option
plans. Members of the Compensation Committee are Messrs. Newhall and
Evnin.
The
Governance and Nominating Committee, created by the Board in February 2004,
assists the Board in identifying qualified individuals to become directors,
determines the composition of the Board and its committees, monitors the process
to assess Board effectiveness and helps develop and implement the Company’s
corporate governance guidelines. Members of the Governance and Nominating
Committee are Messrs. Newhall and Evnin.
The Audit
Committee is primarily responsible for overseeing the services performed by the
Company’s independent registered public accounting firm and evaluating the
Company’s accounting policies and its system of internal
controls. The Audit Committee is comprised of two members: Messrs.
Evnin and Akkaraju, both of whom are independent directors. Mr. Evnin
is the designated “audit committee financial expert” under Item 407(d)(5) of
Regulation S-K. Mr. Evnin is considered “independent”.
The Audit
Committee, Compensation Committee and Governance and Nominating Committee each
operate under written charters adopted by the Board. These charters
are available on the Company’s website at www.pharmoscorp.com. Click
“Investors,” and then “Corporate Governance.”
Code
of Ethics
As part
of our system of corporate governance, our Board of Directors has adopted a Code
of Ethics and Business Conduct that is applicable to all employees and
specifically applicable to our chief executive officer, president, chief
financial officer and controllers. The Code of Ethics and Business
Conduct is available on the Company’s website at
www.pharmoscorp.com. Click “Investors,” and then “Corporate
Governance.” We intend to disclose any changes in or waivers from our Code of
Ethics and Business Conduct by filing a Form 8-K or by posting such information
on our website.
Section
16(a) Beneficial Ownership Reporting Compliance
No person
who, during the fiscal year ended December 31, 2009, was a “Reporting Person”
defined as a director, officer or beneficial owner of more than ten percent of
the Company’s Common Stock which is the only class of securities of the Company
registered under Section 12 of the Securities Exchange Act of 1934 (the “Act”),
failed to file on a timely basis, reports required by Section 16 of the Act
during the most recent fiscal year. The foregoing is based solely
upon a review by the Company of Forms 3 and 4 during the most recent fiscal year
as furnished to the Company under Rule 16a-3(d) under the Act, and Forms 5 and
amendments thereto furnished to the Company with respect to its most recent
fiscal year, and any representation received by the Company from any reporting
person that no Form 5 is required.
35
Item
11. Executive Compensation
COMPENSATION
DISCUSSION AND ANALYSIS
General
Executive Compensation Policy
The
Compensation and Stock Option Committee of the Board of Directors establishes
the general compensation policies of the Company, the compensation plans and
specific compensation levels for executive officers, and administers the
Company’s 2001 Employee Stock Purchase Plan, as well as the 2000, 1997 and 1992
Incentive and Non-Qualified Stock Option Plans and the 2009 Incentive
Compensation Plan. The Compensation and Stock Option Committee is composed of
two independent, non-employee Directors who have no interlocking relationships
as defined by the Securities and Exchange Commission.
The
Compensation and Stock Option Committee, being responsible for overseeing and
approving executive compensation and grants of stock options, is in a position
to appropriately balance the current cash compensation considerations with the
longer-range incentive-oriented growth outlook associated with stock options.
The main objectives of the Company’s compensation structure include rewarding
individuals for their respective contributions to the Company’s performance,
establishing executive officers with a stake in the long-term success of the
Company and providing compensation policies that will attract and retain
qualified executive personnel.
The
Compensation and Stock Option Committee uses no set formulas and may accord
different weight to different factors for each executive. The Committee looks
toward the progress of the Company’s research and development programs and its
clinical programs, its ability to gain support for those programs, either
internally or externally, its ability to attract, motivate and retain talented
employees and its ability to secure capital sufficient for its product
development to achieve rapid and effective commercialization as may be
practicable.
The
Compensation and Stock Option Committee believes that officers’ compensation
should be heavily influenced by Company performance. The Committee
establishes base salaries that are within the range of salaries for persons
holding positions of similar responsibility at other companies. In
addition, the Committee considers factors such as relative Company performance,
the executive’s past performance and future potential in establishing the base
salaries of executive officers.
The
number of options granted to officers is determined by the subjective evaluation
of the executive’s ability to influence the Company’s long-term growth. All
options are granted at no less than the current market price. Since the value of
an option bears a direct relationship to the Company’s stock price, it is an
effective incentive for managers to create value for
shareholders. The Committee therefore views stock options as an
important component of its long-term, performance-based compensation
philosophy.
Compensation
Determinations for 2008
Cash
Compensation. In January, 2008, the Compensation and Stock
Option Committee increased Colin Neill’s annual base compensation 13%, effective
January 1, 2008, from $265,000 for 2007 to $300,000 for 2008 reflecting his
increased responsibilities and duties as President and Chief Financial
Officer.
Equity
Compensation. Seeking to base a significant part of their
respective compensation on future performance, the Committee in January 2008
awarded 350,000 ten-year stock options to Mr. Johnston, Executive Chairman and
130,000 ten-year stock options to Mr. Neill all under the Company’s 2000 Stock
Plan. Mr. Johnston was also to be eligible for an additional grant of 100,000
ten-year stock options on October 1, 2008 if certain performance-based
milestones had been achieved prior to that date in the areas of corporate
development and clinical product trials. Those milestones were
not achieved and the options were not granted.
36
January
2008 Officer Transitions
On
January 3, 2008, the Company announced that Elkan Gamzu, Ph.D., who had been
Chief Executive Officer, retired from that position (Dr. Gamzu remained on the
Board through December 30, 2008). Robert F. Johnston was named as the Company’s
new Executive Chairman of the Board of Directors, and Colin Neill was named to
serve as President in addition to his responsibilities as Chief Financial
Officer, Treasurer and Secretary.
Bonuses
for 2008 and Compensation Determinations for 2009
Cash
Compensation. The Compensation and Stock Option Committee awarded no 2008
cash bonuses nor did the Committee award any 2009 salary increases due to the
Company’s limited capital resources.
Equity
Compensation. In May 2009, the Compensation and Stock Option
Committee awarded 1.2 million shares of restricted stock to Robert Johnston
under the 2000 Stock Option Plan. The restricted shares shall vest
(subject to accelerated vesting or risk of forfeiture in certain circumstances)
as follows: one-third of the restricted shares shall vest on the first
anniversary of the grant date, and the remaining restricted shares shall vest in
quarterly increments over a three-year period commencing on the first
anniversary of the grant date. In May 2009, the Committee also
awarded 600,000 ten-year stock options to Colin Neill under the 2000 Stock
Option Plan.
Bonuses
for 2009 and Compensation Determinations for 2010
In
January 2010, the Compensation and Stock Option Committee, taking into account
that no bonuses were awarded for 2008, awarded cash bonuses for 2009 performance
in the amount of $80,000 to each of Robert F. Johnston and S. Colin
Neill. The Committee determined to leave base salaries unchanged for
2010.
COMPENSATION
AND STOCK OPTION COMMITTEE REPORT
The
Compensation and Stock Option Committee of Pharmos Corporation has reviewed and
discussed the Compensation Discussion and Analysis required by Item 402(b) of
Regulation S-K with management and, based on such review and discussions, the
Compensation and Stock Option Committee has recommended to the Board that the
Compensation Discussion and Analysis be included in this Annual Report on Form
10-K.
THE
COMPENSATION AND
STOCK
OPTION COMMITTEE
Anthony
B. Evnin
Charles
W. Newhall, III
37
SUMMARY
COMPENSATION TABLE
The
following table summarizes the total compensation of the President, Chief
Executive Officer and Executive Chairman of the Company in 2009 and the two
previous years, as well as all other executive officers of the Company who
received compensation in excess of $100,000 for 2009.
|
Name/Principal Position
|
Year
|
Salary
|
Bonus
|
Option
Awards
|
All
Other
Compensation
|
Total
Compensation
|
||||||||||||||||||
|
S.
Colin Neill (1)
|
2009
|
$ | 300,000 | $ | 80,000 | $ | 56,383 | $ | 21,967 | (3) | $ | 458,350 | ||||||||||||
|
President,
|
2008
|
$ | 300,000 | - | $ | 38,426 | $ | 21,517 | (3) | $ | 359,943 | |||||||||||||
|
Chief
Financial Officer,
|
2007
|
$ | 265,000 | $ | 75,000 | (2) | $ | 31,036 | $ | 20,712 | (3) | $ | 391,748 | |||||||||||
|
Secretary
& Treasurer
|
||||||||||||||||||||||||
|
Elkan
R. Gamzu, Ph.D,
|
2009
|
- | - | - | - | - | ||||||||||||||||||
|
Former
Director and former
|
2008
|
- | - | - | - | - | ||||||||||||||||||
|
Chief
Executive Officer
|
2007
|
$ | 452,500 | (4) | $ | 18,346 | $ | 6,750 | (5) | $ | 477,596 | |||||||||||||
|
Robert
F. Johnston
|
2009
|
- | $ | 80,000 | $ | 61,685 | - | $ | 141,685 | |||||||||||||||
|
Executive
Chairman
|
2008
|
- | - | $ | 27,681 | - | $ | 27,681 | ||||||||||||||||
|
2007
|
- | - | - | - | - | |||||||||||||||||||
|
(1)
|
Mr.
Neill joined Pharmos Corporation in October 2006. He became President in
January 2008.
|
|
(2)
|
Consists
of $50,000 in cash and 75,000 shares of common stock valued at
$25,000.
|
|
(3)
|
In
2009, consists of $7,350 in 401k employer contribution, $5,617 in life
insurance and $9,000 in automobile allowance. In 2008, consists of $6,900
in 401k employer contribution, $5,617 in life insurance and $9,000 in
automobile allowance. In 2007, consists of $6,750 in 401k
employer contribution, $4,962 in life insurance and $9,000 in automobile
allowance.
|
|
(4)
|
Dr.
Gamzu served as Chief Executive Officer from March 2007 to January
2008. Dr. Gamzu’s compensation consists of $252,500 in salary
and $200,000 in employment contract severance
payments.
|
|
(5)
|
Consists
of 401k employer contribution.
|
|
|
GRANTS
OF PLAN-BASED AWARDS IN 2009
|
|
Name
|
Grant
Date
|
All
other Option Awards: Number of Securities Underlying Options (#)
|
Exercise
or Base Price of Option Awards
($/Sh)
|
Grant
Date Fair Value of Stock and Option Awards
($)
|
|
S.
Colin Neill (1)
|
5/11/2009
|
600,000
|
$0.22
|
$105,820
|
|
Robert
F. Johnston (2)
|
5/11/2009
|
1,200,000
|
$0.22
|
$264,000
|
(1) 25%
of the options granted to Mr. Neill vest upon the first anniversary of the grant
with the remaining 75% of the grant vesting ratably on a quarterly basis over
the three years following the first anniversary of the grant.
(2)
Robert F. Johnston, the executive chairman was granted a restricted stock award
of 1,200,000 shares on May 11, 2009.
All
option grants in 2009 were made under the Company’s 2000 Stock Option
Plan.
38
|
OPTION
EXERCISES AND STOCK VESTED IN 2009
|
|||||
|
Option Awards
|
Stock Awards
|
||||
|
Name
|
Number
of Shares Acquired on Exercise
(#)
|
Value
Realized on Exercise
($)
|
Number
of Shares Acquired on Vesting
(#)
|
Value
Realized on Vesting($)
|
|
|
None
|
-
|
$ -
|
-
|
-
|
|
|
OUTSTANDING
EQUITY AWARDS AT 2009 FISCAL YEAR-END
|
|||||||||||||||||||
|
Option Awards
|
Stock Awards
|
||||||||||||||||||
|
Name
|
Number
of securities underlying unexercised options (#) Exercisable
|
Number
of securities underlying unexercised options (#) Unexercisable
|
Equity
Incentive Plan awards: Number of securities underlying unexercised
Unearned Options (#)
|
Option
Exercise Price ($)
|
Option
Expiration Date
|
Number
of Shares or Units of Stock That Have Not Vested
|
Market
Value of Shares or Units of Stock That have not vested ($)
|
||||||||||||
|
S.
Colin Neill
|
0 | 600,000 | (1) | 600,000 | $ | 0.22 |
5/11/2019
|
||||||||||||
| 56,875 | 73,125 | (2) | 130,000 | $ | 0.35 |
1/23/2018
|
|||||||||||||
| 27,500 | 12,500 | (3) | 40,000 | $ | 1.84 |
1/17/2017
|
|||||||||||||
| 75,000 | 15,000 | (4) | 90,000 | $ | 1.75 |
10/5/2016
|
|||||||||||||
| 159,375 | 700,625 | 860,000 | |||||||||||||||||
|
Robert
F. Johnston (6)
|
194,444 | 155,556 | (5) | 350,000 | $ | 0.35 |
1/23/2018
|
1,200,000
|
$264,000
|
||||||||||
(1) 25%
of the options were scheduled to vest upon the first anniversary of the grant
date May 11, 2009 with the remaining 75% of the grant vesting ratably on a
quarterly basis over the three years following the first anniversary of the
grant.
(2) 25%
of the options were scheduled to vest upon the first anniversary of the grant
date January 23, 2008 with the remaining 75% of the grant vesting ratably on a
quarterly basis over the three years following the first anniversary of the
grant.
(3) 25%
of the options granted vested upon the first anniversary of the grant date of
January 17, 2007 with the remaining 75% of the grant vesting ratably on a
quarterly basis over the three years following the first anniversary of the
grant.
(4) The
remaining options are scheduled to vest in seven equal increments on a quarterly
basis beginning on January 5, 2009.
(5) One
third of the options granted to Mr. Johnston vested immediately with the
remaining two thirds of the grant vesting ratably on a quarterly basis over the
three years commencing on the first anniversary of the grant.
(6) On
May 11, 2009, Robert F. Johnston was granted a restrictive stock award of
1,200,000 shares vesting over four years.
39
Stock
Option Plans
It is
currently the Company’s policy that all full time key employees are considered
annually for the possible grant of stock options, depending upon employee
performance. The criteria for the awards are experience, uniqueness of
contribution to the Company and level of performance shown during the year.
Stock options are intended to generate greater loyalty to the Company and help
make each employee aware of the importance of the business success of the
Company.
As of
December 31, 2009, 3,647,155 options to purchase shares of the Company’s Common
Stock were outstanding under various option plans. During 2009, the Company
granted 1,050,000 options to purchase shares of its Common Stock to employees,
directors and consultants.
A summary
of the various established stock option plans is as follows:
1992
Plan. The maximum number of shares of the Company’s Common
Stock available for issuance under the 1992 Plan is 150,000 shares, subject to
adjustment in the event of stock splits, stock dividends, mergers,
consolidations and the like. Common Stock subject to options granted under the
1992 Plan that expire or terminate would again be available for options to be
issued under the 1992 Plan. As of December 31, 2009, there were no options
outstanding to purchase the Company’s Common Stock under this plan. The Company
does not plan to issue any additional options from the 1992 Plan.
1997 Plan and
2000 Plan. The 1997 Plan and the 2000 Plan are each
administered by a committee appointed by the Board of Directors (the
“Compensation Committee”). The Compensation Committee will designate
the persons to receive options, the number of shares subject to the options and
the terms of the options, including the option price and the duration of each
option, subject to certain limitations. All stock options grants during 2006
were made from the 2000 Plan. The Company does not plan to issue any additional
options from the 1997 Plan.
The
maximum number of shares of Common Stock available for issuance under the 1997
Plan is 300,000 shares, as amended, and under the 2000 Plan, as amended, is
4,700,000 shares. Each plan is subject to adjustment in the event of stock
splits, stock dividends, mergers, consolidations and the like. Common Stock
subject to options granted under the 1997 Plan and the 2000 Plan that expire or
terminate will again be available for options to be issued under each
Plan.
The price
at which shares of Common Stock may be purchased upon exercise of an incentive
stock option must be at least 100% of the fair market value of Common Stock on
the date the option is granted (or at least 110% of fair market value in the
case of a person holding more than 10% of the outstanding shares of Common Stock
(a “10% Stockholder”).
The
aggregate fair market value (determined at the time the option is granted) of
Common Stock with respect to which incentive stock options are exercisable for
the first time in any calendar year by an optionee under the 1997 Plan, the 2000
Plan or any other plan of the Company or a subsidiary, shall not exceed
$100,000. The Compensation Committee will fix the time or times when,
and the extent to which, an option is exercisable, provided that no option for
annual option grants will be exercisable earlier than one year or later than ten
years after the date of grant (or five years in the case of a 10%
Stockholder). The option price is payable in cash or by check to the
Company. However, the Board of Directors may grant a loan to an
employee, other than an executive officer, pursuant to the loan provision of the
1997 Plan or the 2000 Plan, for the purpose of exercising an option or may
permit the option price to be paid in shares of Common Stock at the then current
fair market value, as defined in the 1997 Plan or the 2000 Plan.
40
Under the
1997 Plan, upon termination of an optionee’s employment or consultancy, all
options held by such optionee will terminate, except that any option that was
exercisable on the date employment or consultancy terminated may, to the extent
then exercisable, be exercised within three months thereafter (or one year
thereafter if the termination is the result of permanent and total disability of
the holder), and except such three month period may be extended by the
Compensation Committee in its discretion. If an optionee dies while
he is an employee or a consultant or during such three-month period, the option
may be exercised within one year after death by the decedent’s estate or his
legatees or distributees, but only to the extent exercisable at the time of
death. The 2000 Plan provides that the Compensation Committee may in
its discretion determine when any particular stock option shall
expire. A stock option agreement may provide for expiration prior to
the end of its term in the event of the termination of the optionee’s service to
the Company or death or any other circumstances.
The 1997
Plan and the 2000 Plan each provides that outstanding options shall vest and
become immediately exercisable in the event of a “sale” of the Company,
including (i) the sale of more than 75% of the voting power of the Company in a
single transaction or a series of transactions, (ii) the sale of substantially
all assets of the Company, (iii) approval by the stockholders of a
reorganization, merger or consolidation, as a result of which the stockholders
of the Company will own less than 50% of the voting power of the reorganized,
merged or consolidated company.
The Board
of Directors may amend, suspend or discontinue the 1997 Plan, but it must obtain
stockholder approval to (i) increase the number of shares subject to the 1997
Plan, (ii) change the designation of the class of persons eligible to receive
options, (iii) decrease the price at which options may be granted, except that
the Board may, without stockholder approval accept the surrender of outstanding
options and authorize the granting of new options in substitution therefore
specifying a lower exercise price that is not less than the fair market value of
Common Stock on the date the new option is granted, (iv) remove the
administration of the 1997 Plan from the Compensation Committee, (v) render any
member of the Compensation Committee eligible to receive an option under the
1997 Plan while serving thereon, or (vi) amend the 1997 Plan in such a manner
that options issued under it intend to be incentive stock options, fail to meet
the requirements of Incentive Stock Options as defined in Section 422 of the
Code.
The Board
of Directors may amend, suspend or discontinue the 2000 Plan, but it must obtain
stockholder approval to (i) increase the number of shares subject to the 2000
Plan or (ii) change the designation of the class of persons eligible to receive
options.
Under
current federal income tax law, the grant of incentive stock options under the
1997 Plan or the 2000 Plan will not result in any taxable income to the optionee
or any deduction for the Company at the time the options are
granted. The optionee recognizes no gain upon the exercise of an
option. However the amount by which the fair market value of Common
Stock at the time the option is exercised exceeds the option price is an “item
of tax preference” of the optionee, which may cause the optionee to be subject
to the alternative minimum tax. If the optionee holds the shares of
Common Stock received on exercise of the option at least one year from the date
of exercise and two years from the date of grant, he will be taxed at the time
of sale at long-term capital gains rates, if any, on the amount by which the
proceeds of the sale exceed the option price. If the optionee
disposes of the Common Stock before the required holding period is satisfied,
ordinary income will generally be recognized in an amount equal to the excess of
the fair market value of the shares of Common Stock at the date of exercise over
the option price, or, if the disposition is a taxable sale or exchange, the
amount of gain realized on such sale or exchange if that is less. If,
as permitted by the 1997 Plan or the 2000 Plan, the Board of Directors permits
an optionee to exercise an option by delivering already owned shares of Common
Stock valued at fair market value) the optionee will not recognize gain as a
result of the payment of the option price with such already owned
shares. However, if such shares were acquired pursuant to the
previous exercise of an option, and were held less than one year after
acquisition or less than two years from the date of grant, the exchange will
constitute a disqualifying disposition resulting in immediate taxation of the
gain on the already owned shares as ordinary income. It is not clear
how the gain will be computed on the disposition of shares acquired by payment
with already owned shares.
41
2001 Employee
Stock Purchase Plan. The 2001 Plan is intended to qualify as
an employee stock purchase plan under Section 423 of the Code. All
employees of the Company, its Pharmos Ltd. subsidiary or any other subsidiaries
or affiliated entities who have completed 180 consecutive days of employment and
who customarily work at least 20 hours per week will be eligible to participate
in the 2001 Plan, except for any employee who owns five percent or more of the
total combined voting power or value of all classes of stock of the Company or
any subsidiary on the date a grant of a right to purchase shares under the 2001
Plan (Right) is made. There currently are no such employees with such
large holdings. Participation by officers in the 2001 Plan will be on
the same basis as that of any other employee. No employee will be granted a
Right which permits such employee to purchase shares under the 2001 Plan at a
rate which exceeds $25,000 of fair market value of such shares (determined at
the time such Right is granted) for each calendar year in which such Right is
outstanding. Each Right will expire if not exercised by the date specified in
the grant, which date will not exceed 27 months from the date of the grant.
Rights will not be assignable or transferable by a participating employee, other
than in accordance with certain qualified domestic relations orders, as defined
in the Code, or by will or the laws of descent and distribution.
The total
number of shares reserved for issuance under the 2001 Plan is 100,000
shares. Under the 2001 Plan, for any given calendar year, a
participating employee can only be granted Rights to purchase that number of
shares which, when multiplied by the exercise price of the Rights, does not
exceed more than 10% of the employee’s base pay. To date, the Company has issued
12,560 shares of its common stock under the 2001 Plan. The Company did not issue
any shares under the 2001 Plan in 2009 and 2008.
From time
to time, the Board of Directors may fix a date or a series of dates on which the
Company will grant Rights to purchase shares of Common Stock under the 2001 Plan
at prices not less than 85% of the lesser of (i) the fair market value of the
shares on the date of grant of such Right or (ii) the fair market value of the
shares on the date such Right is exercised.
The 2001
Plan also provides that any shares of Common Stock purchased upon the exercise
of Rights cannot be sold for at least six months following exercise, to avoid
potential violations of the “short swing” trading provisions of Section 16 of
the Securities Exchange Act of 1934, as amended.
The Board
of Directors or a committee to which it delegates its authority under the 2001
Plan will administer, interpret and apply all provisions of the 2001 Plan. The
Board has delegated such authority to the Compensation and Stock Option
Committee.
The Board
of Directors may amend, modify or terminate the 2001 Plan at any time without
notice, provided that no such amendment, modification or termination may
adversely affect any existing Rights of any participating employee, except that
in the case of a participating employee of a foreign subsidiary of the Company,
the 2001 Plan may be varied to conform with local laws. In addition, subject to
certain appropriate adjustments to give effect to relevant changes in the
Company’s capital stock, no amendments to the 2001 Plan may be made without
stockholder approval if such amendment would increase the total number of shares
offered under the 2001 Plan or would render Rights “unqualified” for special tax
treatment under the Code.
No
taxable income will be recognized by a participant either at the time a Right is
granted under the 2001 Plan or at the time the shares are purchased. Instead,
tax consequences are generally deferred until a participant disposes of the
shares (e.g., by sale or gift). The federal income tax consequences of a sale of
shares purchased under the 2001 Plan will depend on the length of time the
shares are held after the relevant date of grant and date of exercise, as
described below.
42
If shares
purchased under the 2001 Plan are held for more than one year after the date of
purchase and more than two years from the date of grant, the participant
generally will have taxable ordinary income on a sale or gift of the shares to
the extent of the lesser of: (i) the amount (if any) by which the fair market
value of the stock at the date of grant exceeds the exercise price paid by the
participant; or (ii) the amount by which the fair market value of the shares on
the date of sale or gift exceeds the exercise price paid by the participant for
the shares. In the case of a sale, any additional gain will be
treated as long-term capital gain. If the shares are sold for less
than the purchase price, there will be no ordinary income, and the participant
will have a long-term capital loss for the difference between the purchase price
and the sale price.
If the
stock is sold or gifted within either one year after the date of purchase or two
years after the date of grant (a “disqualifying disposition”), the participant
generally will have taxable ordinary income at the time of the sale or gift to
the extent that the fair market value of the stock at the date of exercise was
greater than the exercise price. This amount will be taxable in the
year of sale or disposition even if no gain is realized on the sale, and the
Company would be entitled to a corresponding deduction. A capital
gain would be realized upon the sale of the shares to the extent the sale
proceeds exceed the fair market value of those shares on the date of
purchase. A capital loss would be realized to the extent the sales
price of the shares disposed of is less than the fair market value of such
shares on the date of purchase. Special tax consequences may follow from
dispositions other than a sale or gift.
2009 Incentive Compensation
Plan.
Under the
2009 Plan, the total number of shares of common stock of our company reserved
and available for delivery is 8,000,000 shares. The foregoing limit shall be
increased by the number of shares of common stock with respect to which awards
previously granted under the 2009 Plan are forfeited, expire or otherwise
terminate without issuance of shares, or that are settled for cash or otherwise
do not result in the issuance of shares, and the number of shares that are
tendered (either actually or by attestation) or withheld upon exercise of an
award to pay the exercise price or any tax withholding requirements. Awards
issued in substitution for awards previously granted by a company acquired by
our company or a related entity, or with which our company or any related entity
combines, do not reduce the limit on grants of awards under the 2009 Plan. The
2009 Plan imposes individual limitations on the amount of certain awards in part
to comply with Code Section 162(m).
The
persons eligible to receive awards under the 2009 Plan are the officers,
directors, employees, consultants and other persons who provide services to our
company or any related entity. An employee on leave of absence may be considered
as still in the employ of our company or a related entity for purposes of
eligibility for participation in the 2009 Plan.
The 2009
Plan is to be administered by the Compensation Committee, provided, however,
that except as otherwise expressly provided in the 2009 Plan, our Board of
Directors may exercise any power or authority granted to the Compensation
Committee under the 2009 Plan. Subject to the terms of the 2009 Plan, the
Compensation Committee is authorized to select eligible persons to receive
awards, determine the type, number and other terms and conditions of, and all
other matters relating to, awards, prescribe award agreements (which need not be
identical for each participant), and the rules and regulations for the
administration of the 2009 Plan, construe and interpret the 2009 Plan and award
agreements, correct defects, supply omissions or reconcile inconsistencies
therein, and make all other decisions and determinations as the Compensation
Committee may deem necessary or advisable for the administration of the 2009
Plan.
43
The
Compensation Committee is authorized to grant stock options, including both
incentive stock options (“ISOs”), which can result in potentially favorable tax
treatment to the participant, and non-qualified stock options, and stock
appreciation rights entitling the participant to receive the amount by which the
fair market value of a share of common stock on the date of exercise exceeds the
grant price of the stock appreciation right. The exercise price per share
subject to an option and the grant price of a stock appreciation right are
determined by the Compensation Committee, but must not be less than the fair
market value of a share of common stock on the date of grant. For purposes of
the 2009 Plan, the term “fair market value” means the fair market value of
common stock, awards or other property as determined by the Compensation
Committee or under procedures established by the Compensation Committee. Unless
otherwise determined by the Compensation Committee, the fair market value of
common stock as of any given date shall be the closing sales price per share of
common stock as reported on the principal stock exchange or market on which
common stock is traded on the date as of which such value is being determined
or, if there is no sale on that date, then on the last previous day on which a
sale was reported or the average of the closing bid and asked prices for the
common stock quoted by an established quotation service for over-the-counter
securities, if the common stock is not then traded on a national securities
exchange, or, in the discretion of the Committee, the last sale price or the
closing price. The maximum term of each option or stock appreciation right, the
times at which each option or stock appreciation right will be exercisable, and
provisions requiring forfeiture of unexercised options or stock appreciation
rights at or following termination of employment generally are fixed by the
Compensation Committee, except that no option or stock appreciation right may
have a term exceeding ten years. Methods of exercise and settlement and other
terms of the stock appreciation right are determined by the Compensation
Committee. The Compensation Committee, thus, may permit the exercise price of
options awarded under the 2009 Plan to be paid in cash, shares, other awards or
other property (including loans to participants). Options may be exercised by
payment of the exercise price in cash, shares of common stock, outstanding
awards or other property having a fair market value equal to the exercise price,
as the Compensation Committee may determine from time to time.
The
Compensation Committee is authorized to grant restricted stock and deferred
stock. Restricted stock is a grant of shares of common stock which may not be
sold or disposed of, and which shall be subject to such risks of forfeiture and
other restrictions as the Compensation Committee may impose. A participant
granted restricted stock generally has all of the rights of a stockholder of our
company, unless otherwise determined by the Compensation Committee. An award of
deferred stock confers upon a participant the right to receive shares of common
stock at the end of a specified deferral period, subject to such risks of
forfeiture and other restrictions as the Compensation Committee may impose.
Prior to settlement, an award of deferred stock carries no voting or dividend
rights or other rights associated with share ownership, although dividend
equivalents may be granted, as discussed below.
The
Compensation Committee is authorized to grant dividend equivalents conferring on
participants the right to receive, currently or on a deferred basis, cash,
shares of common stock, other awards or other property equal in value to
dividends paid on a specific number of shares of common stock or other periodic
payments. Dividend equivalents may be granted alone or in connection with
another award, may be paid currently or on a deferred basis and, if deferred,
may be deemed to have been reinvested in additional shares of common stock,
awards or otherwise as specified by the Compensation Committee.
The
Compensation Committee is authorized to grant shares of common stock as a bonus
free of restrictions, or to grant shares of common stock or other awards in lieu
of company obligations to pay cash under the 2009 Plan or other plans or
compensatory arrangements, subject to such terms as the Compensation Committee
may specify.
The
Compensation Committee or our Board of Directors is authorized to grant awards
that are denominated or payable in, valued by reference to, or otherwise based
on or related to shares of common stock. The Compensation Committee determines
the terms and conditions of such awards.
44
The
Compensation Committee is authorized to grant performance awards to participants
on terms and conditions established by the Compensation Committee. The
performance criteria to be achieved during any performance period and the length
of the performance period is determined by the Compensation Committee upon the
grant of the performance award; provided however, that a performance period
cannot be shorter than 12 months or longer than 5 years. Performance awards may
be valued by reference to a designated number of shares (in which case they are
referred to as performance shares) or by reference to a designated amount of
property including cash (in which case they are referred to as performance
units). Performance awards may be settled by delivery of cash, shares or other
property, or any combination thereof, as determined by the Compensation
Committee.
Awards
may be settled in the form of cash, shares of common stock, other awards or
other property, in the discretion of the Compensation Committee. The
Compensation Committee may require or permit participants to defer the
settlement of all or part of an award in accordance with such terms and
conditions as the Compensation Committee may establish, including payment or
crediting of interest or dividend equivalents on deferred amounts, and the
crediting of earnings, gains and losses based on deemed investment of deferred
amounts in specified investment vehicles. The Compensation Committee is
authorized to place cash, shares of common stock or other property in trusts or
make other arrangements to provide for payment of our company’s obligations
under the 2009 Plan. The Compensation Committee may condition any payment
relating to an award on the withholding of taxes and may provide that a portion
of any shares of common stock or other property to be distributed will be
withheld (or previously acquired shares of common stock or other property be
surrendered by the participant) to satisfy withholding and other tax
obligations. awards granted under the 2009 Plan generally may not be pledged or
otherwise encumbered and are not transferable except by will or by the laws of
descent and distribution, or to a designated beneficiary upon the participant’s
death, except that the Compensation Committee may, in its discretion, permit
transfers for estate planning or other purposes subject to any applicable
restrictions under Rule 16b-3.
Awards
under the 2009 Plan are generally granted without a requirement that the
participant pay consideration in the form of cash or property for the grant (as
distinguished from the exercise), except to the extent required by law. The
Compensation Committee may, however, grant awards in exchange for other awards
under the 2009 Plan, awards under other company plans, or other rights to
payment from our company, and may grant awards in addition to and in tandem with
such other awards, rights or other awards.
45
Employment
Contracts
S. Colin Neill. In October
2006, the Compensation and Stock Option Committees of the Board of Directors
recommended, and the Board approved, a one year employment agreement for Mr.
Neill as full time Senior Vice President, Chief Financial Officer, Secretary and
Treasurer of the Company. Mr. Neill’s initial base compensation was
$265,000. The other provisions of Mr. Neill’s employment agreement relate to
benefits, severance arrangements, automatic renewal and confidentiality and
non-competition obligations. Mr. Neill received a sign-on bonus of $20,000 in
October 2006. Mr. Neill was named President on January 3, 2008 and
retained all previous titles and positions.
Compensation
of Directors
Our
Director Compensation policy is as follows:
|
|
1.
|
At
the election of each Director, either (i) 20,000 fully vested ten-year
stock options are granted in January and 20,000 fully vested ten-year
stock options are granted on July 1, or (ii) a cash payment of $6,000 is
made in January and $6,000 on July 1;
and
|
|
|
2.
|
The
Chairman of the Audit Committee will be granted an additional 5,000 fully
vested ten-year stock options in January and 5,000 fully vested ten-year
stock options on July 1; and the Chairmen of the Compensation and the
Governance and Nominating Committees will each be granted an additional
2,500 fully vested ten-year stock options in January and 2,500 fully
vested ten-year stock options on July
1.
|
46
DIRECTOR
COMPENSATION FOR 2009
The table
below summarizes the compensation paid by the Company to non-employee Directors
for the fiscal year ended December 31, 2009:
|
Name
|
Fees
Earned or Paid in Cash($)
|
Option
Awards ($) (1)
|
Total
($)
|
Number
of Shares
|
|||||||||
|
Srinivas
Akkaraju, M.D., Ph.D.
|
0
|
$
|
15,728
|
(2)
|
$
|
15,728
|
140,000
|
||||||
|
Anthony
B. Evnin, Ph.D.
|
0
|
$
|
15,071
|
$
|
15,071
|
52,500
|
|||||||
|
Charles
W. Newhall, III
|
0
|
$
|
14,060
|
$
|
14,060
|
47,500
|
|||||||
|
(1)
|
Reflects
the dollar amount recognized for financial statement reporting purposes
for the fiscal year ended December 31, 2009 in accordance with FAS ASC
Topic 718, and thus includes amounts from awards granted in and prior to
2009. All options awarded to Directors in 2009 remained
outstanding at fiscal year-end.
|
|
|
(2)
|
In
addition to his regular compensation for service on the Board of
Directors, Dr Akkaraju was granted 100,000 ten-year options in May 2009
for providing special service and advice to the Board in his capacity as a
Director.
|
At
December 31, 2009 the aggregate number of director options was 430,000 which
consisted of the following breakdown (Akkaraju 200,000, Evnin 117,500 and
Newhall 112,500).
The fair
value of the director options awarded during 2009 was as follows: Akkaraju
$23,761, Evnin $8,406 and Newhall $7,395.
The
Executive Chairman, Robert F. Johnston, is not an independent director. At
December 31, 2009 he had a total of 350,000 shares of options
outstanding.
Director
Retirements and New Director Appointment in 2009
None
Compensation
Committee Interlocks and Insider Participation
The
members of the Compensation and Stock Option Committee in 2009 were Anthony B.
Evnin and Charles W. Newhall, III. There were no interlocks on the
Compensation and Stock Option Committee in 2009.
47
Item 12. Security Ownership of Certain
Beneficial Owners and
Management and Related Stockholder Matters
EQUITY
COMPENSATION PLAN INFORMATION
The table
below provides certain information concerning our equity compensation plans as
of December 31, 2009.
|
Plan
Category
|
Number
of securities to be issued upon exercise of outstanding options, warrants
and rights
(a)
|
Weighted
average exercise price of outstanding options, warrants and
rights
(b)
|
Number
of securities remaining available for future issuance under equity
compensation plans (excluding securities reflected in column
(a)
(c)
|
|||||||||
|
Equity
compensation plans approved by security holders
|
3,685,130 | $ | 2.42 | 8,872,568 | ||||||||
|
Equity
compensation plans not approved by security holders
|
N/A | N/A | N/A | |||||||||
|
Total
|
3,685,130 | $ | 2.42 | 8,872,568 | ||||||||
48
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS
The
following table sets forth certain information with respect to the beneficial
ownership of the Company’s Common Stock as of February 22, 2010, except as set
forth in the footnotes, by (i) each person who was known by the Company to own
beneficially more than 5% of any class of the Company’s Stock, (ii) each of the
Company’s executive officers and Directors, and (iii) all current Directors and
executive officers of the Company as a group. Except as otherwise noted, each
person listed below has sole voting and dispositive power with respect to the
shares listed next to such person’s name.
|
Name and Address of Beneficial
Owner
|
Amount
of Beneficial Ownership
|
|
Percentage
of Total (1)
|
||||||
|
Robert
F. Johnston
c/o
Pharmos Corporation
99
Wood Avenue South, Suite 311, Iselin, NJ 08830
|
18,025,118 | (2 | ) | 27.5 | % | ||||
|
Srinivas
Akkaraju, M.D., Ph.D.
c/o
Panorama Management, LLC
2440
Sand Hill Road, Suite 302, Menlo Park, CA 94025
|
116,250 | (3 | ) | * | |||||
|
Anthony
B. Evnin
c/o
Venrock Associates
530
Fifth Avenue, 22nd
Floor, New York, NY 10036
|
13,656,886 | (4 | ) | 21.5 | % | ||||
|
Charles
W. Newhall, III
c/o
New Enterprise Associates
1119
St. Paul Street, Baltimore, MD 21202
|
22,422,341 | (5 | ) | 33.2 | % | ||||
|
S.
Colin Neill
c/o
Pharmos Corporation
99
Wood Avenue South, Suite 311, Iselin, NJ 08830
|
257,499 | (6 | ) | * | |||||
|
All
Current Directors and
Executive
Officers as a group (five persons)
|
54,478,094 | (7 | ) | 69.6 | % | ||||
|
New
Enterprise Associates 10, LP
1119
St. Paul Street, Baltimore, MD 21202
|
22,288,591 | (8 | ) | 33.1 | % | ||||
|
Venrock
Assoicates
Venrock Associates III
LP
Venrock Entrepreneurs Fund
III LP
530 Fifth Avenue, 22nd
Floor, New York, NY 10036
|
13,518,136 | (9 | ) | 21.3 | % | ||||
* Less
than 1%.
|
(1)
|
Based
on 59,438,213 shares of common stock outstanding as of February 22, 2010,
plus each individual’s warrants or options which are either currently
exercisable or will be exercisable within 60 days of the date set forth
above. Assumes that no other individual will exercise any
warrants and/or options.
|
|
(2)
|
Consists
of 5,811,230 outstanding shares, 213,888 shares issuable upon exercise of
currently exercisable options, and 6,000,000 outstanding shares and
6,000,000 currently exercisable warrants held by Demeter
Trust.
|
|
(3)
|
Consists
of 116,250 shares issuable upon exercise of currently exercisable
options.
|
|
(4)
|
Consists
of shares beneficially owned by Venrock Associates, Venrock Associates III
LP and Venrock Entrepreneurs Fund III LP and 138,750 shares issuable upon
exercise of currently exercisable
options.
|
|
(5)
|
Consists
of shares beneficially owned by New Enterprise Associates 10, LP and
133,750 shares issuable upon exercise of currently exercisable
options.
|
|
(6)
|
Consists
of 75,000 outstanding shares and 182,499 shares issuable upon exercise of
currently exercisable options.
|
|
(7)
|
Consists
of 35,692,957 outstanding shares, 785,137 shares issuable upon exercise of
currently exercisable options and 18,000,000 shares issuable upon exercise
of currently exercisable warrants.
|
|
(8)
|
Consists
of 14,288,591 outstanding shares and 8,000,000 shares issuable upon
exercise of currently exercisable
warrants.
|
|
(9)
|
Consists
of 9,518,136 outstanding shares and 4,000,000 shares issuable upon
exercise of currently exercisable
warrants.
|
49
Item
13. Certain Relationships, Related Transactions and Director
Independence
Director
Independence. The Company has determined that three of the
four Directors serving at December 31, 2009 (Srinivas Akkaraju, Anthony B.
Evnin, and Charles W. Newhall, III), were independent.
Item
14. Principal Accounting Fees and Services
On
October 19, 2009, Pharmos Corporation dismissed its independent registered
public accounting firm, PricewaterhouseCoopers LLP. The dismissal was
approved by the Pharmos Audit Committee. During the two most recent
fiscal years and through October 19, 2009, there have been no disagreements
between Pharmos and PricewaterhouseCoopers LLP on any matter of accounting
principles or practices, financial statement disclosure, or auditing scope or
procedure, which disagreements if not resolved to the satisfaction of
PricewaterhouseCoopers LLP would have caused them to make reference thereto in
their reports on the financial statements for such years. During the
two most recent fiscal years and through October 19, 2009, there were no
reportable events (as defined in Item 304(a)(1)(v)) of Regulation
S-K. Neither of the reports of PricewaterhouseCoopers LLP for the
past two years contained an adverse opinion or disclaimer of opinion, or was
qualified or modified as to uncertainty, audit scope or accounting principles,
except that their report dated February 25, 2009 contained an explanatory
paragraph expressing substantial doubt about Pharmos' ability to continue as a
going concern. On October 20, 2009, Pharmos Corporation engaged
Friedman LLP as its new independent accounting firm.
Audit
fees
For 2009
the aggregate fees billed by Friedman LLP for professional services rendered for
the audit of the Company’s annual financial statements for the year ending
December 31, 2009 and for the review of the financial statements included in the
Company’s quarterly reports on Form 10-Q for the quarter ended September 30,
2009 was $36,000.
For 2009
the aggregate fees billed by PricewaterhouseCoopers LLP for professional
services rendered for the review of the financial statements included in the
Company’s quarterly reports on Form 10Q for the quarters ended March 31, 2009
and June 30, 2009 were $40,000. PricewaterhouseCoopers LLP also billed $7,700
for the reissuance of the 2008 audit opinion. The Pricewaterhouse Israel fee for
the statutory audit of the Pharmos Limited financial statements was
$4,000.
For 2008
the aggregate fees billed by PricewaterhouseCoopers LLP for professional
services rendered for the audit of the Company’s annual financial statements for
the year ended December 31, 2008 and for the reviews of the financial statements
included in the Company’s quarterly reports on Form 10-Q for the same fiscal
year was $185,000. The fee charged by Pricewaterhouse Israel for the statutory
audit of the Pharmos Limited financial statements was $23,000.
Audit-related
fees
There
were no audit—related fees in 2009 and 2008.
Tax
fees
Aggregate
fees for professional services rendered by PricewaterhouseCoopers LLP in
connection with its income tax compliance and related tax services for the years
ended December 31, 2009 and 2008 were $0 and $0, respectively.
50
All
other fees
A fee of
$1,500 was incurred in each of 2009 and 2008 in relation to subscription
services for accounting related topics. The Company also licenses Automated
Disclosure Checklist - Client Assist from PricewaterhouseCoopers LLP at no
cost.
Policy
on Audit Committee Pre-Approval of Audit and Non-Audit Services of Independent
Auditor
The
charter of the Audit Committee requires that the Committee review and
pre-approve all audit, review or attest engagements of, and non-audit services
to be provided by, the independent registered public accounting firm (other than
with respect to the de minimis exception permitted by the Sarbanes-Oxley Act of
2002 and the SEC rules promulgated there under). The Audit Committee
pre-approved all auditing services and permitted non-audit services rendered by
PricewaterhouseCoopers LLP and Friedman LLP in 2009.
The
pre-approval duty may be delegated to one or more designated members of the
Audit Committee, with any such pre-approval reported to the Committee at its
next regularly scheduled meeting. Any such designated member(s) of
the Committee shall also have the authority to approve non-audit services
already commenced by the independent registered public accounting firm if (i)
the aggregate amount of all such services provided constitutes no more than five
percent (5%) of the total amount of revenues paid by the Company to the
independent registered public accounting firm during the fiscal year in which
the services are provided, (ii) such services were not recognized by the Company
at the time of the engagement to be non-audit services, and (iii) such services
are promptly brought to the attention of the Committee and approved by such
designated member(s) prior to the completion of the audit.
51
PART
IV
Item
15. Exhibits and Financial Statement Schedules
|
(a)
|
Financial
Statements and Exhibits
|
|
(1)
|
FINANCIAL
STATEMENTS
|
|
Reports
of Independent Registered Public Accounting Firms
|
|
|
Consolidated
balance sheets as of December 31, 2009 and 2008
|
|
|
Consolidated
statements of operations for the years ended
|
|
|
December 31, 2009, 2008 and
2007
|
|
|
Consolidated
statements of changes in shareholders’ equity
|
|
|
for the years ended December
31, 2009, 2008 and 2007
|
|
|
Consolidated
statements of cash flows for the years ended
|
|
|
December 31, 2009, 2008, and
2007
|
|
|
Notes
to consolidated financial statements
|
|
(2)
|
FINANCIAL
STATEMENT SCHEDULES
|
All
schedules have been omitted because the information required to be set forth
therein is not applicable or is shown in the financial statements or notes
thereto.
|
(3)
|
EXHIBITS
|
|
2
|
Plan
of purchase, sale, reorganization, arrangement, liquidation, or
succession
|
|
|
2(a)
|
Agreement
and Plan of Merger by and among Pharmos Corporation, Vela Acquisition
Corporation and Vela Pharmaceuticals Inc. dated March 14, 2006
(Incorporated by reference to Exhibit 2.1 to the Company’s Current Report
on Form 8-K filed March 15, 2006).
|
|
|
2(b)
|
Amendment
to Agreement and Plan of Merger by and among Pharmos Corporation, Vela
Acquisition Corporation and Vela Pharmaceuticals Inc. dated August 31,
2006 (incorporated by reference to Exhibit 2.1 of the registrant’s Current
Report on Form 8-K filed September 5,
2006).
|
|
|
2(c)
|
Amendment
No.2 to Agreement and Plan of Merger by and among Pharmos Corporation,
Vela Acquisition Corporation, Vela Acquisition No.2 Corporation and Vela
Pharmaceuticals Inc. dated September 29, 2006 (incorporated by reference
to Exhibit 2.1 of the registrant’s Current Report on Form 8-K filed
October 5, 2006).
|
|
|
2(d)
|
Amendment
No. 3 to Agreement and Plan of Merger dated as of January 3, 2008 by and
among Pharmos Corporation and the Representatives named therein (incorporated by
reference to Exhibit 4.5 of the registrant’s Current Report on Form 8-K
filed January 4, 2008).
|
|
3
|
Articles
of Incorporation and By-Laws
|
|
|
3(a)
|
Restated
Articles of Incorporation (Incorporated by reference to Appendix E to the
Joint Proxy Statement/Prospectus included in the Form S-4 Registration
Statement of the Company dated September 28, 1992 (No.
33-52398)
|
|
|
3(b)
|
Certificate
of Amendment of Restated Articles of Incorporation dated January 30, 1995
(Incorporated by reference to Annual Report on Form 10-K for the year
ended December 31, 1994).
|
|
|
3(c)
|
Certificate
of Amendment of Restated Articles of Incorporation dated January 16, 1998
(Incorporated by reference to the Company’s Current Report on Form 8-K,
dated February 6, 1998).
|
52
|
|
3(d)
|
Certificate
of Amendment of Restated Articles of Incorporation dated October 21, 1999
(Incorporated by reference to exhibit 4(e) to the Form S-3 Registration
Statement of the Company filed September 28, 2000 (No.
333-46818)).
|
|
|
3(e)
|
Certificate
of Amendment of Restated Articles of Incorporation dated July 19, 2002
(Incorporated by reference to Exhibit 3 to the Company’s Report on Form
10-Q for the quarter ended June 30,
2002).
|
|
|
3(f)
|
Certificate
of Amendment of Restated Articles of Incorporation dated July 7, 2004
(Incorporated by reference to Exhibit 3.1 to the Company’s Report on Form
10-Q for the quarter ended June 30,
2004).
|
|
|
3(g)
|
Certificate
of Amendment to Articles of Incorporation dated September 23, 2005
(Incorporated by reference to exhibit 3.1 to the Company’s Quarterly
Report on Form 10-Q for the quarter ended September 30,
2005).
|
|
|
3(h)
|
Certificate
of Amendment to Articles of Incorporation dated August 5, 2009
(Incorporated by reference to exhibit 3.8 to the Company’s Quarterly
Report on Form 10-Q for the quarter ended June 30,
2009).
|
|
|
3(i)
|
Amended
and Restated By-Laws (Incorporated by reference to Exhibit 3.1 to the
Company’s Quarterly Report on Form 10-Q for the quarter ended September
30, 2007.
|
|
4
|
Instruments
defining the rights of security holders, including
indentures
|
|
|
4(a)
|
Form
of Employee Warrant Agreement, dated April 11, 1995, between the Company
and Oculon Corporation (Incorporated by reference to the Company’s Current
Report on Form 8-K, dated April 11, 1995, as
amended).
|
|
|
4(b)
|
Form
of Warrant Agreement dated as of April 30, 1995 between the Company and
Charles Stolper (Incorporated by reference to Form S-3 Registration
Statement of the Company dated November 14, 1995, as amended [No.
33-64289]).
|
|
|
4
(c)
|
Form
of Stock Purchase Warrant dated as of March 31, 1997 between the Company
and the Investor (Incorporated by reference to Form S-3 Registration
Statement of the Company dated March 5, 1998 [No.
333-47359]).
|
|
|
4(d)
|
Form
of Common Stock Purchase Warrant exercisable until September 1, 2005
(Incorporated by reference to Exhibit 4.4 to the Company’s Current Report
on Form 8-K filed on September 11,
2000).
|
|
|
4(e)
|
Form
of placement agent warrant with Ladenburg Thalmann & Co. Inc.
(Incorporated by reference to Form S-3 Registration Statement of the
Company dated September 28, 2000 (No.
333-46818).
|
|
|
4(f)
|
Form
of placement agent warrant with SmallCaps OnLine LLC (Incorporated by
reference to Form S-3 Registration Statement of the Company dated
September 28, 2000 (No. 333-46818).
|
|
|
4(g)
|
Form
of consulting warrant with SmallCaps OnLine LLC (Incorporated by reference
to Form S-3 Registration Statement of the Company dated September 28, 2000
(No. 333-46818).
|
|
|
4(h)
|
Certificate
of Designation, Rights Preferences and Privileges of Series D Preferred
Stock of the Company (Incorporated by reference to Exhibit 99.3 to the
Company’s Current Report on Form 8-K filed on October 24,
2002).
|
|
|
4(i)
|
Rights
Agreement dated as of October 23, 2002 between the Company and American
Stock Transfer & Trust Company, as Rights Agent (Incorporated by
reference to Exhibit 99.2 to the Company’s Current Report on Form 8-K
filed on October 24, 2002).
|
53
|
|
4(j)
|
Form
of Investor Warrant dated March 4, 2003 (Incorporated by reference to
Exhibit 99.2 to the Company’s Current Report on Form 8-K filed on March 4,
2003).
|
|
|
4(k)
|
Form
of Placement Agent’s Warrant dated March 4, 2003 (Incorporated by
reference to Exhibit 99.1 to the Company’s Current Report on Form 8-K
filed on March 4, 2003).
|
|
|
4(l)
|
Registration
Rights Agreement dated as of May 30, 2003 between the Company and the
purchasers. (Incorporated by reference to Exhibit 4.2 to the Company’s
Current Report on Form 8-K filed on June 3,
2003).
|
|
|
4(m)
|
Form
of Investor Warrant dated June 2, 2003 (Incorporated by reference to
Exhibit 4.3 to the Company’s Current Report on Form 8-K filed on June 3,
2003).
|
|
|
4(n)
|
Securities
Purchase Agreement dated as of September 26, 2003 between the Company and
the purchasers identified on the signature pages thereto 2003
(Incorporated by reference to Exhibit 4.1 to the Company’s Current Report
on Form 8-K filed on September 30,
2003).
|
|
|
4(o)
|
Form
of 4% convertible debenture due March 31, 2005 (Incorporated by reference
to Exhibit 4.2 to the Company’s Current Report on Form 8-K filed on
September 30, 2003).
|
|
|
4(p)
|
Registration
Rights Agreement dated as of September 26, 2003 between the Company and
the purchasers signatory thereto (Incorporated by reference to Exhibit 4.3
to the Company’s Current Report on Form 8-K filed on September 30,
2003).
|
|
|
4(q)
|
Form
of Common Stock Purchase Warrant (Incorporated by reference to Exhibit 4.4
to the Company’s Current Report on Form 8-K filed on September 30,
2003).
|
|
|
4(r)
|
Escrow
Agreement dated as of September 26, 2003 between the Company, the
purchasers signatory thereto and Feldman Weinstein LLP (Incorporated by
reference to Exhibit 4.5 to the Company’s Current Report on Form 8-K filed
on September 30, 2003).
|
|
|
4(s)
|
Form
of Placement Agent Common Stock Purchase Warrant (Incorporated by
reference to Exhibit 4.6 to the Company’s Current Report on Form 8-K filed
on September 30, 2003).
|
|
|
4(t)
|
Rights
Agreement Amendment, dated October 23, 2006, between Pharmos Corporation
and American Stock Transfer & Trust Co. (Incorporated by reference to
Exhibit 4.1 to the Company’s Current Report on Form 8-K filed on October
27, 2006).
|
|
|
4(u)
|
Registration
Rights Agreement, dated as of October 25, 2006, by and among Pharmos
Corporation and the Representatives named therein (Incorporated by
reference to Exhibit 4.1 to the Company’s Current Report on Form 8-K filed
on October 31, 2006).
|
|
|
4(v)
|
10% Convertible
Debenture dated as of January 3, 2008 of Pharmos Corporation (Incorporated
by reference to Exhibit 4.3 to the Company’s Current Report on Form 8-K
filed on January 4, 2008).
|
|
|
4(w)
|
Registration
Rights Agreement dated as of January 3, 2008 by and among Pharmos
Corporation and the Purchasers named therein (Incorporated by reference to
Exhibit 4.4 to the Company’s Current Report on Form 8-K filed on January
4, 2008).
|
|
|
4(x)
|
Amendment
No. 2 dated as of January 3, 2008 to the Rights Agreement, dated as of
September 5, 2002, as amended on October 23, 2006 (the ÒRights
AgreementÓ), between Pharmos Corporation and American Stock Transfer &
Trust Co., as Rights Agent (Incorporated by
reference to Exhibit 4.6 to the Company’s Current Report on Form 8-K filed
on January 4, 2008).
|
|
|
4(y)
|
Amendment
No. 1 to Registration Rights Agreement dated as of April 25, 2008 by and
among Pharmos Corporation and the Purchasers named therein (Incorporated by
reference to Exhibit 4.1 to the Company’s Current Report on Form 8-K filed
on May 1, 2008).
|
54
|
|
4(z)
|
Amendment No. 2 to
Registration Rights Agreement by and among Pharmos Corporation and the
Purchasers named therein (Incorporated by reference to Exhibit 4.2
to the Company’s Quarterly Report on Form 10-Q for the quarter ended June
30, 2008).
|
|
|
4(aa)
|
Form of waiver under
Pharmos Corporation 10% Convertible Debentures Due November 1, 2012
(Incorporated by reference to exhibit 4.3 to the Company’s
Quarterly Report on Form 10-Q for the quarter ended June 30, 2008).
|
|
|
4(bb)
|
Form
of waiver, dated January 15, 2009, under Pharmos Corporation 10%
Convertible Debentures Due November 1, 2012 (incorporated by reference to
exhibit 4.1 to the Company’s Current Report on Form 8-K filed January 22,
2009)
|
|
|
4(cc)
|
Securities
Purchase Agreement dated as of April 21, 2009 by and among Pharmos
Corporation and the Purchasers named therein (incorporated by reference to
exhibit 4.1 to the Company’s Current Report on Form 8-K filed April 23,
2009).
|
|
|
4(dd)
|
Form
of Stock Purchase Warrant dated April 21, 2009 (incorporated by reference
to exhibit 4.2 to the Company’s Current Report on Form 8-K filed April 23,
2009).
|
|
|
4(ee)
|
Registration
Rights Agreement dated as of April 21, 2009 by and among Pharmos
Corporation and the Purchasers named therein (incorporated by reference to
exhibit 4.3 to the Company’s Current Report on Form 8-K filed April 23,
2009).
|
|
|
4(ff)
|
Debenture
Amendment Agreement dated April 21, 2009 among Pharmos Corporation, New
Enterprise Associates 10, Limited Partnership, Venrock Associates, Venrock
Associates III, L.P., Venrock Entrepreneurs Fund III, L.P. and Robert F.
Johnston (incorporated by reference to exhibit 4.4 to the Company’s
Current Report on Form 8-K filed April 23,
2009).
|
|
|
4(gg)
|
Amendment
No. 3 dated as of April 21, 2009 to the Rights Agreement, dated as of
October 23, 2002, as amended on October 23, 2006 and on January 3, 2008
(the “Rights Agreement”), between Pharmos Corporation and American Stock
Transfer & Trust Company, LLC, as Rights Agent (incorporated by
reference to exhibit 4.5 to the Company’s Current Report on Form 8-K filed
April 23, 2009).
|
|
10
|
Material
Contracts
|
|
|
10(a)
|
Employment
Agreement dated as of April 2, 2001, between Pharmos Corporation and Haim
Aviv (Incorporated by reference to Exhibit 10(n) to the Company’s Annual
Report on Form 10-K for the year ended December 31,
2001).**
|
|
|
10(b)
|
Amendment
of Employment Agreement with Haim Aviv, dated as of January 25, 2006
(Incorporated by reference to Exhibit 10.1 to the Company’s Quarterly
Report on Form 10-Q for the quarter ended March 31,
2006).**
|
|
|
10(c)
|
Employment
Agreement dated as of April 2, 2001, between Pharmos Corporation and Gad
Riesenfeld (Incorporated by reference to Exhibit 10(o) to the Company’s
Annual Report on Form 10-K for the year ended December 31,
2001).**
|
|
|
10(d)
|
Amendment
of Employment Agreement dated as of April 23, 2001, between Pharmos
Corporation and Gad Riesenfeld (Incorporated by reference to Exhibit 10(p)
to the Company’s Annual Report on Form 10-K for the year ended December
31, 2001).**
|
|
|
10(e)
|
Amendment
of Employment Agreement dated as of February 16, 2005 between Pharmos
Corporation and Gad Riesenfeld (Incorporated by reference to Exhibit 10(w)
to the Company’s Annual Report on Form 10-K for the year ended December
31, 2004).**
|
|
|
10(f)
|
Employment
Agreement dated as of July 19, 2004 between Pharmos Corporation and James
A. Meer (Incorporated by reference to Exhibit 10.1 to the Company’s
Quarterly Report on Form 10-Q for the quarter ended September 30,
2004).**
|
55
|
|
10(g)
|
Employment
Agreement dated as of November 7, 2005, between Pharmos Corporation and
Alan L. Rubino (Incorporated by reference to exhibit 10.1 to the Company’s
Quarterly Report on Form 10-Q for the quarter ended September 30,
2005).**
|
|
|
10(h)
|
Employment
Agreement between Pharmos Corporation and S. Colin Neill, dated as of
October 5, 2006 (Incorporated by reference to Exhibit 10.1 to the
Company’s Current Report on Form 8-K filed on October 6,
2006).**
|
|
|
10(i)
|
Retention
Award Agreement dated as of September 6, 2004 between Pharmos Corporation
and Dr. Haim Aviv (Incorporated by reference to Exhibit 10.1 to the
Company’s Current Report on Form 8-K filed on September 10,
2004).**
|
|
|
10(j)
|
Retention
Award Agreement dated as of September 6, 2004 between Pharmos Corporation
and Dr. Gad Riesenfeld (Incorporated by reference to Exhibit 10.2 to the
Company’s Current Report on Form 8-K filed on September 10,
2004).**
|
|
|
10(k)
|
Consulting
Agreement between Pharmos Corporation and Dr. Georges Anthony Marcel,
dated October 17, 2006 (Incorporated by reference to Exhibit 10.1 to the
Company’s Current Report on Form 8-K filed on October 20,
2006).**
|
|
|
10(l)
|
Consulting
Agreement between Pharmos Corporation and Dr. Lawrence F. Marshall, dated
October 17, 2006 (Incorporated by reference to Exhibit 10.2 to the
Company’s Current Report on Form 8-K filed on October 20,
2006).**
|
|
|
10(m)
|
1997
Incentive and Non-Qualified Stock Option Plan (Annexed as Appendix B to
the Proxy Statement on Form 14A filed November 5,
1997).**
|
|
|
10(n)
|
Amended
and Restated 2000 Incentive and Non-Qualified Stock Option
Plan.**
|
|
|
10(o)
|
Amendment
to the 2000 Stock Option Plan (incorporated by reference to Appendix D to
the Company’s Definitive Proxy Statement on Schedule 14A filed on
September 10, 2007).**
|
|
|
10(p)
|
2001
Employee Stock Purchase Plan (Incorporated by reference to Exhibit B to
the Company’s Definitive Proxy Statement on Form 14A filed on June 6,
2001).**
|
|
|
10(q)(1)
|
Agreement
between Avitek Ltd. (“Avitek”) and Yissum Research Development Company of
the Hebrew University of Jerusalem (“Yissum”) dated November 20, 1986
(Incorporated by reference to Annual Report on Form 10-K, as amended by
Form10-K/A, for year ended December 31,
1992). (1)
|
|
|
10(q)(2)
|
Supplement
to Agreement (Incorporated by reference to Annual Report on Form 10-K, as
amended by Form 10-K/A, for year ended December 31,
1992). (1)
|
|
|
10(q)(3)
|
Hebrew
language original executed version of Agreement (Incorporated by reference
to Annual Report on Form 10-K, as amended by Form 10-K/A, for year ended
December 31, 1992). (1)
|
|
|
10(r)(1)
|
Agreement
between Avitek and Yissum dated January 25, 1987 (Incorporated by
reference to Annual Report on Form 10-K, as amended by Form 10-K/A, for
year ended December 31,
1992). (1)
|
|
|
10(r)(2)
|
Schedules
and Appendixes to Agreement (Incorporated by reference to Annual Report on
Form 10-K, as amended by Form 10-K/A, for year ended December 31,
1992). (1)
|
|
|
10(r)(3)
|
Hebrew
language original executed version of Agreement (Incorporated by reference
to Annual Report on Form 10-K, as amended by Form 10-K/A, for year ended
December 31, 1992). (1)
|
|
|
10(s)(1)
|
Research,
Development and License Agreement between Pharmos Ltd., Pharmos
Corporation (“Old Pharmos”) and Yissum dated February 5, 1991
(Incorporated by reference to Annual Report on Form 10-K, as amended by
Form 10-K/A, for year ended December 31,
1992). (1)
|
56
|
|
10(s)(2)
|
Schedules
and Appendixes to Agreement (Incorporated by reference to Annual Report on
Form 10-K, as amended by Form 10-K/A, for year ended December 31,
1992). (1)
|
|
|
10(s)(3)
|
Amendment
No.1 to Research, Development and License Agreement between Pharmos
Corporation, Pharmos Ltd. and Yissum Research Development Company of the
Hebrew University of Jerusalem, dated May 31, 2006 (incorporated by
reference to exhibit 99.1 to the registrant’s Current Report on Form 8-K
filed June 6, 2006).
|
|
|
10(t)
|
License
Assignment and Amendment Agreement dated as of October 9, 2001 by and
among Dr. Nicholas S. Bodor, Pharmos Corporation and Bausch & Lomb
Incorporated (Incorporated by reference to Exhibit 2.2 to the Company’s
Current Report on Form 8-K filed on October 16,
2001).
|
|
|
10(u)
|
Asset
Purchase Agreement between Bausch & Lomb Incorporated and Pharmos
Corporation dated October 9, 2001 (Incorporated by reference to Exhibit
2.1 to the Company’s Current Report on Form 8-K filed on October 16,
2001).
|
|
|
10(v)
|
Amendment
No. 1 to Asset Purchase Agreement dated as of December 28, 2001 between
Bausch & Lomb Incorporated and Pharmos Corporation (Incorporated by
reference to Exhibit 10(v) to the Company’s Annual Report on Form 10-K for
the year ended December 31, 2001).
|
|
|
10(w)
|
Amendment
No. 2 to Asset Purchase Agreement dated as of December 30, 2004 between
Bausch & Lomb Incorporated and Pharmos Corporation (Incorporated by
reference to Exhibit 10(v) to the Company’s Annual Report on Form 10-K for
the year ended December 31, 2004).
|
|
|
10(x)
|
License
Agreement dated as of December 18, 2001 between Pharmos Ltd. and Herbamed
Ltd. (Incorporated by reference to Exhibit 10(p) to the Annual Report on
Form 10-K for year ended December 31,
2002).
|
|
|
10(y)
|
Amendment
No. 1, dated as of June 30, 2005, to the License Agreement by and between
Pharmos Ltd. and Herbamed Ltd., dated as of December 18, 2001
(Incorporated by reference to exhibit 99.1 to the Company’s Quarterly
Report on Form 10-Q for the quarter ended June 30,
2005).
|
|
|
10(z)
|
Settlement
Agreement between the Company and Lloyd I. Miller, III dated August 31,
2006 (incorporated by reference to Exhibit 10.1 of the registrant’s
Current Report on Form 8-K filed September 5,
2006).
|
|
|
10(aa)
|
Voting
Agreement and Waiver by and among the Company, Lloyd I. Miller, III, Trust
A-4 - Lloyd I. Miller, Milfam II L.P. and Milfam LLC dated August 31, 2006
(incorporated by reference to Exhibit 10.2 of the registrant’s Current
Report on Form 8-K filed September 5,
2006).
|
|
|
10(bb)
|
Employment
Agreement between Pharmos Corporation and Elkan R. Gamzu, dated as of
March 20, 2007 (incorporated by reference to Exhibit 10.1 of the
registrant’s Current Report on Form 8-K filed March 26,
2007).
|
|
|
10(cc)
|
Letter
of Agreement between Pharmos Corporation and Haim Aviv, dated March 20,
2007 (incorporated by reference to Exhitit 10.2 of the registrant’s
Current Report on Form 8-K filed March 26,
2007.
|
57
|
|
10(dd)
|
Securities
Purchase Agreement dated as of January 3, 2008 by and among Pharmos
Corporation and the Purchasers named therein (incorporated by reference to
Exhibit 4.1 of the registrant’s Current Report on Form 8-K filed January
4, 2008).
|
|
|
10(ee)
|
Letter
Agreement dated January 3, 2008 regarding Additional Debenture Investment
among Pharmos Corporation, New Enterprise Associates 10, Limited
Partnership, Lloyd I. Miller, III and Robert F. Johnston (incorporated by
reference to Exhibit 4.2 of the registrant’s Current Report on Form 8-K
filed January 4, 2008).
|
|
|
10(ff)
|
Agreement dated
January 3, 2008 between Pharmos Corporation and Mony Ben Dor
(incorporated by reference to Exhibit 10.1 of the registrant’s
Current Report on Form 8-K filed January 4,
2008).
|
|
|
10(gg)
|
Agreement dated
January 3, 2008 between Pharmos Corporation and David Schlachet
(incorporated by reference to Exhibit 10.2 of the registrant’s
Current Report on Form 8-K filed January 4,
2008).
|
|
|
10(hh)
|
Agreement dated
January 3, 2008 between Pharmos Corporation and Haim Aviv (incorporated
by reference to Exhibit 10.3 of the registrant’s Current Report on Form
8-K filed January 4, 2008).
|
|
|
10(ii)
|
Asset
Purchase Agreement, dated as of February 11, 2009, by and between Pharmos
Corporation, Pharmos Ltd. and Reperio Pharmaceuticals Ltd. (Incorporated
by reference to exhibit 10.1 to the Company’s Quarterly Report on Form
10-Q for the quarter ended March 31,
2009)
|
|
|
10(jj)
|
Form
of Pharmos Corporation Indemnification Agreement dated as of April 21,
2009 (incorporated by reference to exhibit 10.1 to the Company’s Current
Report on Form 8-K filed April 23,
2009).
|
|
|
10(kk)
|
2009
Incentive Compensation Plan (incorporated by reference to Appendix A to
the Registrant’s Definitive Proxy Statement on Schedule 14A filed on June
18, 2009).**
|
|
21
|
Subsidiaries
of the Registrant
|
|
|
21(a)
|
Subsidiaries
of the Registrant (Incorporated by reference to Annual Report on Form
10-K,
as amended by Form 10-K/A, for year ended December 31,
1992).
|
|
23
|
Consents
of Experts and Counsel
|
|
|
23(a)***
|
Consent
of PricewaterhouseCoopers
LLP
|
|
|
23(b)***
|
Consent
of Friedman LLP
|
|
31
|
Rule
13a-14(a)/15d-14(a) Certifications
|
|
|
31(a)***
|
Rule
13a-14(a)/15d-14(a) Certification of Principal Executive
Officer
|
|
|
31(b)***
|
Rule
13a-14(a)/15d-14(a) Certification of Chief Financial
Officer
|
|
32
|
Section
1350 Certifications
|
|
|
32(a)***
|
Section
1350 Certification of Principal Executive
Officer and Chief Financial
Officer
|
|
(1)
|
Confidential
information is omitted and identified by a * and filed separately with the
SEC.
|
|
(**)
|
This
document is a management contract or compensatory plan or
arrangement.
|
|
(***)
|
Filed
herewith
|
58
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of
1934, the Registrant has duly caused this report to be signed on its behalf by
the undersigned, thereunto duly authorized.
PHARMOS
CORPORATION
By: /s/
Robert F. Johnston
__________________________________________
Robert F.
Johnston
Executive
Chairman
(Principal
Executive Officer)
Date:
February 22, 2010
Pursuant
to the requirements of the Securities Exchange Act of 1934, this report has been
signed below by the following persons on behalf of the Registrant and in the
capacities and on the dates indicated:
|
Signature
|
Title
|
Date
|
|
|
|
|
|
/s/ S. Colin Neill
|
||
|
S.
Colin Neill
|
President
and Chief Financial Officer
|
February
22, 2010
|
|
(Principal
Financial and Accounting Officer)
|
||
|
/s/ Srinivas Akkaraju
|
||
|
Srinivas
Akkaraju, MD, Ph.D
|
Director
|
February
22, 2010
|
|
/s/ Anthony B. Evnin
|
||
|
Anthony
B. Evnin, Ph.D
|
Director
|
February
22, 2010
|
|
/s/ Charles W. Newhall, III
|
||
|
Charles
W. Newhall, III
|
Director
|
February
22, 2010
|
59
Pharmos
Corporation
Index
to Consolidated Financial Statements
|
Reports
of Independent Registered Public Accounting Firms
|
F-2
|
|
|
Consolidated
balance sheets as of December 31, 2009 and 2008
|
F-4
|
|
|
Consolidated
statements of operations for the years ended
|
||
|
December
31, 2009, 2008 and 2007
|
F-5
|
|
|
Consolidated
statements of changes in shareholders’ equity
|
||
|
for
the years ended December 31, 2009, 2008 and 2007
|
F-6
|
|
|
Consolidated
statements of cash flows for the years ended
|
||
|
December
31, 2009, 2008, and 2007
|
F-7
|
|
|
Notes
to consolidated financial statements
|
F-8
|
F-1
REPORT OF INDEPENDENT
REGISTERED PUBLIC ACCOUNTING FIRM
The Board
of Directors and
Shareholders
of Pharmos Corporation
Iselin,
New Jersey
We have
audited the accompanying consolidated balance sheet of Pharmos Corporation and
its subsidiaries (the “Company”) as of December 31, 2009, and the related
consolidated statements of operations, changes in shareholders’ equity, and cash
flows for the year ended December 31, 2009. These consolidated financial
statements are the responsibility of the Company’s management. Our
responsibility is to express an opinion on these financial statements based on
our audit.
We
conducted our audit in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require that we plan
and perform the audit to obtain reasonable assurance about whether the financial
statements are free of material misstatement. The Company is not
required to have, nor were we engaged to perform, an audit of their internal
control over financial reporting. Our audit included consideration of internal
control over financial reporting as a basis for designing audit procedures that
are appropriate in the circumstances, but not for the purpose of expressing an
opinion of the effectiveness of the Company’s internal control over financial
reporting. Accordingly, we express no such opinion. An audit includes examining,
on a test basis, evidence supporting the amounts and disclosures in the
financial statements, assessing the accounting principles used and significant
estimates made by management, and evaluating the overall financial statement
presentation. We believe that our audit provides a reasonable basis for our
opinion.
In our
opinion, the consolidated financial statements referred to above present fairly,
in all material respects, the financial position of Pharmos Corporation and
subsidiaries as of December 31, 2009, and the results of their operations
and their cash flows for the year ended December 31, 2009, in conformity
with accounting principles generally accepted in the United States of
America.
As
discussed in Note 2 to the financial statements, the Company is largely
dependent upon ultimately achieving a collaboration with a pharmaceutical
partner or raising additional capital to either advance its lead compound or to
advance other earlier stage compounds. The accompanying financial statements
have been prepared assuming that the Company will continue as a going
concern.
Friedman
LLP
East
Hanover, NJ
February
22, 2010
F-2
Report
of Independent Registered Public Accounting Firm
To The
Board of Directors and
Shareholders
of Pharmos Corporation
In our
opinion, the consolidated balance sheet as of December 31, 2008 and the related
consolidated statements of operations, changes in shareholders' equity and cash
flows for each of two years in the period ended December 31, 2008 present
fairly, in all material respects, the financial position of Pharmos Corporation
and its subsidiaries at December 31, 2008 and the results of their operations
and their cash flows for each of the two years in the period ended December 31,
2008 in conformity with accounting principles generally accepted in the United
States of America. These financial statements are the responsibility of the
Company's management. Our responsibility is to express an opinion on
these financial statements based on our audits. We conducted our
audits of these statements in accordance with the standards of the Public
Company Accounting Oversight Board (United States). Those standards
require that we plan and perform the audit to obtain reasonable assurance about
whether the financial statements are free of material
misstatement. An audit includes examining, on a test basis, evidence
supporting the amounts and disclosures in the financial statements, assessing
the accounting principles used and significant estimates made by management, and
evaluating the overall financial statement presentation. We believe
that our audits provide a reasonable basis for our opinion.
As
discussed in Note 3 to the consolidated financial statements, the Company
changed the manner in which it accounts for advance payments for research and
development activities in 2008, for share-based compensation in 2006, and for
uncertain tax positions in 2007.
The
accompanying financial statements have been prepared assuming that the Company
will continue as a going concern. As discussed in Note 2 to the financial
statements, the Company has suffered recurring losses from operations and has an
accumulated deficit that raise substantial doubt about its ability to continue
as a going concern. Management’s plans in regard to these matters are also
described in Note 2. The financial statements do not include any adjustments
that might result from the outcome of this uncertainty.
PricewaterhouseCoopers
LLP
New York,
New York
February
25, 2009
F-3
|
PHARMOS
CORPORATION
|
||||||||
|
Consolidated
Balance Sheets
|
||||||||
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Assets
|
||||||||
|
Current
Assets
|
||||||||
|
Cash
and cash equivalents
|
$ | 4,629,486 | $ | 4,730,282 | ||||
|
Restricted
cash
|
- | 38,998 | ||||||
|
Prepaid
expenses and other current assets
|
25,678 | 1,049,898 | ||||||
|
Total
current assets
|
4,655,164 | 5,819,178 | ||||||
|
Fixed
assets, net
|
1,379 | 9,692 | ||||||
|
Other
assets
|
32,479 | 143,294 | ||||||
|
Total
assets
|
$ | 4,689,022 | $ | 5,972,164 | ||||
|
Liabilities
and Shareholders’ Equity
|
||||||||
|
Current
liabilities
|
||||||||
|
Accounts
payable
|
$ | 73,717 | $ | 883,966 | ||||
|
Accrued
expenses
|
148,414 | 615,663 | ||||||
|
Accrued
wages and other compensation
|
195,000 | 87,000 | ||||||
|
Total
current liabilities
|
417,131 | 1,586,629 | ||||||
|
Other
liability
|
- | 44,316 | ||||||
|
Convertible
debentures
|
1,000,000 | 4,000,000 | ||||||
|
Total
liabilities
|
1,417,131 | 5,630,945 | ||||||
|
Shareholders’
Equity
|
||||||||
|
Preferred
stock, $.03 par value, 1,250,000 shares authorized, none issued and
outstanding
|
- | - | ||||||
|
Common
stock, $.03 par value; 120,000,000 and 60,000,000 shares
authorized, 59,291,154 and 26,210,290 issued in 2009
and 2008, respectively
|
1,778,735 | 786,307 | ||||||
|
Paid-in
capital in excess of par
|
211,301,675 | 206,309,096 | ||||||
|
Accumulated
deficit
|
(209,808,093 | ) | (206,753,758 | ) | ||||
|
Treasury
stock, at cost, 2,838 shares
|
(426 | ) | (426 | ) | ||||
|
Total
shareholders' equity
|
3,271,891 | 341,219 | ||||||
|
Total
liabilities and shareholders' equity
|
$ | 4,689,022 | $ | 5,972,164 | ||||
|
The
accompanying notes are an integral part of these consolidated financial
statements.
|
||||||||
F-4
PHARMOS
CORPORATION
Consolidated
Statements of Operations
|
Year
ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Expenses
|
||||||||||||
|
Research
and development, gross
|
$ | 4,422,600 | $ | 9,028,705 | $ | 11,457,566 | ||||||
|
Grants
|
- | - | (812,042 | ) | ||||||||
|
Research
and development, net of grants
|
4,422,600 | 9,028,705 | 10,645,524 | |||||||||
|
In-process
acquired research and development
|
180,000 | - | - | |||||||||
|
General
and administrative
|
1,536,470 | 1,965,243 | 6,698,601 | |||||||||
|
Depreciation
and amortization
|
8,314 | 106,236 | 235,134 | |||||||||
|
Total
operating expenses
|
6,147,384 | 11,100,184 | 17,579,259 | |||||||||
|
Loss
from operations
|
(6,147,384 | ) | (11,100,184 | ) | (17,579,259 | ) | ||||||
|
Other
income (expense)
|
||||||||||||
|
Debt
conversion expense
|
(596,104 | ) | - | - | ||||||||
|
Interest
income
|
8,412 | 255,751 | 938,312 | |||||||||
|
Interest
expense
|
(225,335 | ) | (490,537 | ) | - | |||||||
|
Change
in value of warrants
|
- | - | 11,435 | |||||||||
|
Other
income (expense)
|
(8,404 | ) | 41,438 | 47,905 | ||||||||
|
Other
(expense) income, net
|
(821,431 | ) | (193,348 | ) | 997,652 | |||||||
|
Loss
before income taxes
|
$ | (6,968,815 | ) | $ | (11,293,532 | ) | $ | (16,581,607 | ) | |||
|
Income
tax benefit
|
(3,914,481 | ) | (1,204,126 | ) | (955,782 | ) | ||||||
|
Net
loss
|
$ | (3,054,334 | ) | $ | (10,089,406 | ) | $ | (15,625,825 | ) | |||
|
Net
loss per share
|
||||||||||||
|
-
basic and diluted
|
$ | (0.06 | ) | $ | (0.39 | ) | $ | (0.61 | ) | |||
|
Weighted
average shares outstanding
|
||||||||||||
|
-
basic and diluted
|
47,445,014 | 25,934,973 | 25,591,660 | |||||||||
The
accompanying notes are an integral part of these consolidated financial
statements.
F-5
Pharmos
Corporation
Consolidated
Statements of Changes in Shareholders’ Equity
For
the Years ended December 31, 2009, 2008 and 2007
|
Common
Stock
|
|
Treasury
Stock
|
||||||||||||||||||||||||||
|
Shares
|
Amount
|
Paid-in
Capital in Excess of
Par
|
Accumulated
Deficit
|
Shares
|
Amount
|
Total
Stockholders’ Equity
|
||||||||||||||||||||||
|
December
31, 2006
|
25,565,784 | $ | 766,973 | $ | 204,700,030 | $ | (181,038,527 | ) | 2,838 | $ | (426 | ) | $ | 24,428,050 | ||||||||||||||
|
Stock
and option issuance for employee compensation and amortization of
retention award
|
1,182,440 | 1,182,440 | ||||||||||||||||||||||||||
|
Issuance
of Retention Award Shares
|
37,975 | 1,139 | (1,139 | ) | ||||||||||||||||||||||||
|
Net
Loss
|
(15,625,825 | ) | (15,625,825 | ) | ||||||||||||||||||||||||
|
December
31, 2007
|
25,603,759 | 768,112 | 205,881,331 | (196,664,352 | ) | 2,838 | (426 | ) | 9,984,665 | |||||||||||||||||||
|
Stock
and option issuance for employee compensation
|
160,716 | 4,821 | 281,561 | 286,382 | ||||||||||||||||||||||||
|
Issuance
of Common Stock for Debenture Interest payment
|
445,815 | 13,374 | 146,204 | 159,578 | ||||||||||||||||||||||||
|
Net
Loss
|
(10,089,406 | ) | (10,089,406 | ) | ||||||||||||||||||||||||
|
December
31, 2008
|
26,210,290 | 786,307 | 206,309,096 | (206,753,758 | ) | 2,838 | (426 | ) | 341,219 | |||||||||||||||||||
|
Stock
and option issuance for employee compensation
|
175,438 | 175,438 | ||||||||||||||||||||||||||
|
April
2009 financing net
|
18,000,000 | 540,000 | 1,239,777 | 1,779,777 | ||||||||||||||||||||||||
|
Debenture
converted to equity
|
10,909,091 | 327,275 | 3,268,829 | 3,596,104 | ||||||||||||||||||||||||
|
Reversal
of deferred financing fees
|
(78,382 | ) | (78,382 | ) | ||||||||||||||||||||||||
|
Vela
Milestone
|
2,000,000 | 60,000 | 120,000 | 180,000 | ||||||||||||||||||||||||
|
Restricted
stock grant
|
1,200,000 | 36,000 | (36,000 | ) | - | |||||||||||||||||||||||
|
Issuance
of Common Stock for Debenture Interest payment
|
971,773 | 29,153 | 302,917 | 332,070 | ||||||||||||||||||||||||
|
Net
Loss
|
(3,054,335 | ) | (3,054,335 | ) | ||||||||||||||||||||||||
|
December
31, 2009
|
59,291,154 | $ | 1,778,735 | $ | 211,301,675 | $ | (209,808,093 | ) | 2,838 | $ | (426 | ) | $ | 3,271,891 | ||||||||||||||
The
accompanying notes are an integral part of these consolidated financial
statements.
F-6
Pharmos
Corporation
Consolidated
Statements of Cash Flows
|
Year
Ended December 31,
|
||||||||||||
|
2009
|
2008
|
2007
|
||||||||||
|
Cash
flows from operating activities
|
||||||||||||
|
Net
loss
|
$ | (3,054,334 | ) | $ | (10,089,406 | ) | $ | (15,625,825 | ) | |||
|
Adjustments
to reconcile net loss to net cash used in operating
activities:
|
||||||||||||
|
Depreciation
and amortization
|
8,314 | 106,236 | 235,134 | |||||||||
|
Provision
for severance pay
|
- | (358,706 | ) | (961,918 | ) | |||||||
|
Change
in the value of warrants
|
- | - | (11,435 | ) | ||||||||
|
Amortization
of deferred financing fees
|
32,432 | - | - | |||||||||
|
Non
cash debt conversion expense
|
596,104 | - | - | |||||||||
|
Share-based
compensation
|
175,438 | 286,382 | 1,182,441 | |||||||||
|
Debenture
interest paid in common stock
|
130,403 | 159,578 | - | |||||||||
|
Milestone
paid in common shares
|
180,000 | - | - | |||||||||
|
Gain
on disposition of fixed assets
|
- | (213,677 | ) | (12,383 | ) | |||||||
|
Changes
in operating assets and liabilities:
|
||||||||||||
|
Research
and development grants receivable
|
- | 3,859 | 294,006 | |||||||||
|
Prepaid
expenses and other current assets
|
656,228 | (454,713 | ) | 199,473 | ||||||||
|
Other
assets
|
- | (6,806 | ) | (111,474 | ) | |||||||
|
Accounts
payable
|
(810,249 | ) | 115,198 | 105,576 | ||||||||
|
Accrued
expenses
|
(265,585 | ) | 210,726 | (484,846 | ) | |||||||
|
Accrued
wages and other compensation
|
108,000 | (718,995 | ) | (196,577 | ) | |||||||
|
Other
liabilities
|
(44,316 | ) | (7,572 | ) | (25,794 | ) | ||||||
|
Net
cash used in operating activities
|
(2,287,565 | ) | (10,967,896 | ) | (15,413,622 | ) | ||||||
|
Cash
flows from investing activities
|
||||||||||||
|
Purchases
of fixed assets
|
||||||||||||
|
Proceeds
from disposition of fixed assets
|
- | (1,730 | ) | (176,592 | ) | |||||||
|
Purchase
of short-term investments
|
367,994 | 153,937 | 122,839 | |||||||||
|
Proceeds
from sale of short-term investments
|
- | - | (6,933,488 | ) | ||||||||
|
Severance
pay funding
|
- | 3,686,568 | 16,419,593 | |||||||||
|
Decrease
(increase) in restricted cash
|
- | 264,934 | 710,876 | |||||||||
|
Net
cash provided by investing activities
|
38,998 | 112,728 | (4,878 | ) | ||||||||
| 406,992 | 4,216,437 | 10,138,350 | ||||||||||
|
Cash
flows from financing activities
|
||||||||||||
|
Proceeds
from issuance of common stock and warrants net
|
1,779,777 | - | - | |||||||||
|
Proceeds
from issuance of convertible debentures
|
- | 4,000,000 | - | |||||||||
|
Net
cash provided by financing activities
|
1,779,777 | 4,000,000 | - | |||||||||
|
Net
decrease in cash and cash equivalents
|
(100,796 | ) | (2,751,459 | ) | (5,275,272 | ) | ||||||
|
Cash
and cash equivalents at beginning of year
|
4,730,282 | 7,481,741 | 12,757,013 | |||||||||
|
Cash
and cash equivalents at end of year
|
$ | 4,629,486 | $ | 4,730,282 | $ | 7,481,741 | ||||||
|
Supplemental
information:
|
||||||||||||
|
Interest
paid
|
- | $ | 53,201 | - | ||||||||
|
Supplemental
disclosure of non-cash investing and financing activities:
|
||||||||||||
|
Common
shares issued for accrued interest as of December 2008
|
$ | 201,667 | - | - | ||||||||
|
Common
stock issued for 2009 debenture interest
|
$ | 130,403 | - | - | ||||||||
|
Write
off of deferred financing fees with conversion of
debentures
|
$ | 78,382 | - | - | ||||||||
|
Conversion
of debentures to common stock
|
$ | 3,000,000 | - | - | ||||||||
The
accompanying notes are an integral part of these consolidated financial
statements.
F-7
Pharmos
Corporation
Notes
to Consolidated Financial Statements
|
1.
|
The
Company
|
Pharmos
Corporation (the “Company” or “Pharmos”) is a bio-pharmaceutical company that is
focused on the development and execution of a clinical trial for Dextofisopam
for the treatment of Irritable Bowel Syndrome (IBS). Earlier work performed in
the Pharmos Israel facilities covering the discovery and development of novel
therapeutic drugs to treat a range of indications including pain, inflammatory,
autoimmune and select CNS disorders is available for licensing or
sale.
The
Company has executive offices in Iselin, New Jersey. The Company’s operations in
Israel were closed effective October 31, 2008.
|
2.
|
Liquidity
and Business Risks
|
Except
for 2001, the Company has experienced operating losses every year since
inception in funding the research, development and clinical testing of our drug
candidates. The Company had an accumulated deficit of $209.8 million as of
December 31, 2009 and expects to continue to incur losses going forward. Such
losses have resulted principally from costs incurred in research and development
and from general and administrative expenses. Previously the Company had
financed its operations with public and private offerings of securities,
advances and other funding pursuant to an earlier marketing agreement with
Bausch & Lomb, grants from the Office of the Chief Scientist of Israel,
research contracts, the sale of a portion of its New Jersey net operating loss
carryforwards (NOL’s), and interest income. Although the Company
received $3.9 million in December 2009 from the sale of their NOL’s and had
approximately $4.6 million of cash and cash equivalents at December 31, 2009,
the Company is largely dependent upon achieving a collaboration with a
pharmaceutical partner or raising additional capital to either advance its lead
compound, Dextofisopam, for the treatment of IBS or to advance other earlier
stage intellectual property assets. During the year, the
Company engaged an investment advisor to assist with these strategies but has
not been successful to date. The Company has in the past pursued
various funding and financing options; however management believes that future
funding or financing options may be challenging because of the current
environment.
Also, the
Company does not currently have the finances and resources to complete full
clinical trials for its lead compound, Dextofisopam. As a result it may decide
to embark on smaller clinical trials for Dextofisopam or commence pre-clinical
development on its other intellectual property assets which could further reduce
the company’s current resources.
|
3.
|
Significant
Accounting Policies
|
Basis
of consolidation
The
accompanying consolidated financial statements include the accounts of the
Company and its wholly owned subsidiaries: Pharmos Ltd. and Vela
Pharmaceuticals. All significant intercompany balances and
transactions are eliminated in consolidation. The functional currency
for Pharmos Ltd. is the US dollar. Vela Acquisition Corp. is dormant
and was used as the vehicle to acquire Vela Pharmaceuticals Inc. in October
2006. The Israel operations, research and development activities
ceased effective October 31, 2008 and the Company is voluntarily liquidating
Pharmos Ltd.
Subsequent
Events
In May
2009, the FASB established general standards of accounting for and disclosures
of events that occur after the balance sheet date but before financial
statements are issued or are available to be issued. It requires the disclosure
of the date through which an entity has evaluated subsequent events and the
basis for selecting that date, that is, whether that date represents the date
the financial statements were issued or were available to be issued. The
Company's adoption of these standards had no material impact on its financial
position, results of operations and cash flows.
F-8
Pharmos
Corporation
Notes
to Consolidated Financial Statements
The
Company has evaluated events and transactions that occurred between December 31,
2009 and February 22, 2010, which is the date the financial statements were
issued, for possible disclosure and recognition in the financial
statements.
Use
of estimates
The
preparation of the consolidated financial statements in conformity with
generally accepted accounting principles requires the Company to make estimates
and assumptions that affect the reported amounts of assets and liabilities,
disclosure of contingent assets and liabilities at the date of the financial
statements and the reported amounts of revenues, costs and expenses during the
reporting period. The most significant estimates and assumptions related to
stock based compensation and estimates of commitments and contingencies, and the
tax valuation allowance. Actual results could differ from those
estimates.
Net
loss per common share
Basic and
diluted net loss per common share was computed by dividing the net loss for the
period by the weighted average number of shares of common stock issued and
outstanding. In accordance with the requirements of FASB ASC Topic 260 covering
earnings per share, potential shares of common stock have been excluded from the
calculation of diluted net loss per common share, as their inclusion would be
antidilutive.
The
following table summarized the equivalent number of potential common shares
assuming the related securities that were outstanding as of December 31, 2009
and 2008 had been converted.
|
2009
|
2008
|
|||||||
|
Stock
options
|
3,647,155 | 2,737,106 | ||||||
|
Restricted
stock
|
1,200,000 | - | ||||||
|
Convertible
debenture
|
1,428,571 | 5,714,286 | ||||||
|
Warrants
|
18,000,000 | - | ||||||
|
Total
potential dilutive securities not included in loss per
share
|
24,275,726 | 8,451,392 | ||||||
Cash
and cash equivalents
The
Company considers all highly liquid instruments purchased with an original
maturity of three months or less to be cash equivalents. Cash equivalents
primarily consist of US short term government obligations at December 31, 2009
and 2008.
Concentrations
of Credit Risk
Financial
instruments that subject the Company to credit risk primarily consist of cash
and cash equivalents. The Company maintains its cash and cash equivalent
balances with a high quality financial institution who invests the Company’s
funds in Government short-term instruments. Consequently the Company believes
that such funds are subject to minimal credit risk.
F-9
Pharmos
Corporation
Notes
to Consolidated Financial Statements
Revenue
recognition
The
Company’s policy with respect to license fees is to recognize revenue when all
performance obligations are completed. The Company had no product sales revenue
during 2009, 2008, or 2007 and does not expect product sale revenues for the
next few years and may never have such sales if products currently under
development fail to be commercialized.
Research
and development costs
All
research and development costs are expensed when incurred. The Company accounts
for reimbursements of research and development costs as a reduction of research
and development expenses as the underlying expenses are incurred.
Accounting
for Advance Payments for Goods or Services to be Used in Future Research and
Development Activities.
Effective
January 1, 2008 the Company adopted the provisions of FASB ASC Topic 730 which
requires companies involved in research and development activities to capitalize
such non-refundable advance payments for goods and services pursuant to an
executory contractual arrangement because the right to receive those services in
the future represents a probable future economic benefit. Advance payments are
capitalized until the goods have been delivered or the related services have
been performed. At December 31, 2009 and 2008 there was $0 and
$519,000, respectively, in capitalized prepayments.
Restricted
cash
The
Company has a lease agreement for the premises it occupies in New Jersey. The
lease agreement expired in December 2009. The lease agreement was secured by a
letter of credit of $65,031 which was released in 2009 and included in our cash
and cash equivalent balances in 2008.
A deposit
of $38,998, relating to Pharmos Ltd., was included in restricted cash at
December 31, 2008. During 2009 a settlement on this amount was resolved and at
December 31, 2009 the balance was $0.
Fixed
assets
Fixed
assets are recorded at cost and are depreciated on a straight-line basis over
their estimated useful lives. The Company uses the following estimated useful
lives:
|
Laboratory,
pilot plant and other equipment
|
7
years to 14 years
|
|
Leasehold
improvements
|
5
years to 14 years
|
|
Office
furniture and fixtures
|
3
years to 17 years
|
|
Computer
equipment
|
3
years to 4 years
|
|
Vehicles
|
5
years to 7 years
|
Leasehold
improvements are amortized on a straight-line basis over the shorter of the
lease term or the estimated lives of the related assets. Maintenance
and repairs are expensed as incurred.
At
December 31, 2009 there were minimal fixed assets remaining in the financial
statements.
F-10
Pharmos
Corporation
Notes
to Consolidated Financial Statements
Severance
pay
The
Company’s liability for severance pay is calculated pursuant to Israel’s
Severance Pay Law on the most recent salary of the employees multiplied by the
number of years of employment, as of the balance sheet date. Employees are
entitled to one month’s salary for each year of employment or a portion thereof.
The Company’s liability for all of its Israeli employees is fully provided by
monthly deposits with insurance policies and by an accrual. The value of these
policies is recorded as an asset in the Company’s balance sheet. At
December 31, 2008, the Company no longer has any employees in
Israel. All Israel severance pay obligations have been settled as of
December 31, 2008 with the corresponding reduction of the value of these
policies and the related severance liability.
Severance
expenses in Israel for the years ended December 31, 2009, 2008 and 2007 amounted
to $0, $51,102 and $267,879, respectively, and have been included in the
appropriate R&D and G&A expense categories.
Income
taxes
The
Company accounts for income taxes in accordance with the provisions of FASB ASC
Topic 740. “Accounting for Income Taxes” (“ASC – Topic 740”). Under
the asset and liability method of Topic 740, deferred tax assets and liabilities
are recognized for the future tax consequences attributable to differences
between the financial statement carrying amounts of existing assets and
liabilities and their respective tax bases and operating loss and tax credit
carryforwards. Deferred tax assets and liabilities, if any, are
measured using enacted tax rates expected to apply to taxable income in the
years in which those temporary differences are expected to be recovered or
settled. Under Topic 740, the effect on deferred tax assets and
liabilities of a change in tax rates is recognized in income in the period that
includes the enactment date.
Effective
January 1, 2007, the Company adopted, the Financial Accounting Standards Board
interpretation in ASC Topic 740 which covers, Accounting for Uncertainty in Income
Taxes This Interpretation clarifies the accounting for uncertainty in
income taxes recognized in an entity’s financial statements and prescribes a
recognition threshold of more-likely-than-not to be sustained upon examination.
Measurement of the tax uncertainty occurs if the recognition threshold has been
met. This Interpretation also provides guidance on derecognition,
classification, interest and penalties, accounting in interim periods,
disclosure, and transition. Pharmos conducts business in the US and Israel and,
as a result, files US, New Jersey and Israeli income tax returns. In the normal
course of business the Company is subject to examination by taxing authorities.
At present, there are no ongoing audits or unresolved disputes with the various
tax authorities that the Company files with. Given the Company’s substantial net
operating loss carryforwards (“NOLs”), which are subject to a full valuation
allowance as well as the historical operating losses, the adoption of the
standard on January 1, 2007 did not have any effect on our financial position,
results of operations or cash flows.
Foreign
exchange
The
Company’s foreign operations are principally conducted in U.S. dollars. Any
transactions or balances in currencies other than U.S. dollars are remeasured
and any resultant gains and losses are included in other income
(expense). To date, such gains and losses have been
insignificant.
Fair
value of financial instruments
The
carrying amounts of the Company’s financial instruments, including cash and cash
equivalents, investments, other receivables, other assets, accounts payable and
accrued liabilities approximate fair value due to their short term
maturities.
F-11
Pharmos
Corporation
Notes
to Consolidated Financial Statements
Share
based compensation
The
Company has stock-based compensation plans under which employees and outside
directors receive stock options and other equity-based awards. The plans provide
for the granting of stock options, restricted stock awards, and other stock unit
awards. The maximum number of shares of Common Stock available for issuance
under the 2000 Plan, as amended, is 4,700,000 shares. At December 31, 2009,
awards relating to 3,647,155 shares were outstanding. Pharmos stock options are
granted with an exercise price equal to 100% of the market value of a share of
common stock on the date of the grant, generally have 10 year terms, and vest no
later than four years from the date of grant.
Under the
terms of the Pharmos Employee Stock Purchase Plan (ESPP), all full-time and
part-time employees of the Company who have completed a minimum of 6 months of
employment are eligible to participate. The price of the Common Stock is
calculated at 85% of the lower of either the mean between the highest and lowest
prices at which Pharmos common stock trades on the first business day of the
month, or the mean between the highest and lowest trading prices on the day of
exercise (the last day of the month). A participant can purchase shares not to
exceed 10% of one’s annualized base pay; $25,000; or 5% or more of shares
outstanding. The total number of shares reserved for issuance under the 2001
Plan is 100,000 shares. During the years ended December 31, 2009
& 2008, no shares were purchased under the ESPP and 87,440 shares remain for
issuance under the 2001 Plan.
As of
December 31, 2009, the total compensation costs related to non-vested awards not
yet recognized is $460,000 which will be recognized over the next three and
one-half years.
Options
issued to non-employees other than directors are accounted for under the fair
value method in accordance with ASC Topic 718 “Accounting for Equity Instruments
that are Issued to Other than Employees for Acquiring, or in Conjunction with
Selling Goods or Services.” Under the fair value method, compensation cost is
measured at the grant date of the option based on the value of the award using
the Black-Scholes method. Compensation cost is periodically remeasured as the
underlying options vest in accordance with ASC Topic 718 and is recognized over
the service period.
Recently
Issued Accounting Standards
In August
2009, the FASB issued Accounting Standards Update No. 2009-05, Measuring Liabilities at Fair
Value ("ASU 2009-05"). ASU 2009-05 amends Accounting Standards
Codification Topic 820, Fair
Value Measurements. Specifically, ASU 2009-05 provides clarification that
in circumstances in which a quoted price in an active market for the identical
liability is not available, a reporting entity is required to measure fair value
using one or more of the following methods: (1) a valuation technique that
uses (a) the quoted price of the identical liability when traded as an
asset or (b) quoted prices for similar liabilities or similar liabilities
when traded as assets and/or (2) a valuation technique that is consistent
with the principles of Topic 820 of the Codification (e.g. an income
approach or market approach). ASU 2009-05 also clarifies that when estimating
the fair value of a liability, a reporting entity is not required to adjust to
include inputs relating to the existence of transfer restrictions on that
liability. The adoption of this standard did not have an impact on the Company's
financial position or results of operations.
In
October 2009, the FASB issued ASU No. 2009-13, Multiple-Deliverable Revenue
Arrangements ("ASU 2009-13"). ASU 2009-13, amends existing revenue
recognition accounting pronouncements that are currently within the scope of
Codification Subtopic 605-25 (previously included within EITF 00-21, Revenue Arrangements with Multiple
Deliverables ("EITF 00-21"). The consensus to ASU 2009-13 provides
accounting principles and application guidance on whether multiple deliverables
exist, how the arrangement should be separated, and the consideration allocated.
This guidance eliminates the requirement to establish the fair value of
undelivered products and services and instead provides for separate revenue
recognition based upon management's estimate of the selling price for an
undelivered item when there is no other means to determine the fair value of
that undelivered item. EITF 00-21 previously required that the fair value
of the undelivered item be the price of the item either sold in a
F-12
Pharmos
Corporation
Notes
to Consolidated Financial Statements
separate
transaction between unrelated third parties or the price charged for each item
when the item is sold separately by the vendor. Under EITF 00-21, if the
fair value of all of the elements in the arrangement was not determinable, then
revenue was deferred until all of the items were delivered or fair value was
determined. This new approach is effective prospectively for revenue
arrangements entered into or materially modified in fiscal years beginning on or
after June 15, 2010 and allows for retroactive application. The Company is
currently evaluating the potential impact of this standard on its financial
position and results of operations.
In May
2008, the FASB issued standards relating to "Accounting for Convertible Debt
Instruments That May Be Settled in Cash upon Conversion (Including Partial Cash
Settlement)" These are covered by ASC Topic 470 (formerly FSP APB 14-1"). Topic
470 requires the issuer of certain convertible debt instruments
that may be settled in cash (or other assets) on conversion to separately
account for the liability (debt) and equity (conversion option) components of
the instrument in a manner that reflects the issuer's non-convertible debt
borrowing rate. The standard was effective for fiscal years beginning after
December 15, 2008 on a retroactive basis. The Company adopted the standard in
the first quarter 2009. The adoption of this
standard did not have an impact on the Company's financial position or results
of operations.
|
4.
|
Convertible
Debentures
|
On January 3, 2008, Pharmos Corporation completed a private placement of its 10% Convertible Debentures due November 2012. At the closing the Company issued $4,000,000 principal amount of the Debentures, at par, and received gross proceeds in the same amount.
The
purchasers consisted of certain existing investors in the Company, namely
Venrock Associates (which is affiliated with Anthony B. Evnin), New Enterprise
Associates (which is affiliated with Charles W. Newhall, III), Lloyd I. Miller,
III and Robert F. Johnston.
The
Debentures mature the earlier of November 1, 2012 or the sale of the Company.
The Debentures, together with all accrued and unpaid interest thereon, may be
repaid, without premium or penalty, commencing on November 1, 2011. Starting on
November 1, 2009 (or earlier sale of the Company), any outstanding Debenture may
be converted into common shares at the option of the holder. The conversion
price is fixed equal to $0.70 per share. The Debentures bear interest at the
rate of 10% per annum, payable semi-annually either in cash or common stock of
the Company at the option of the Company, provided that an effective
registration statement is in effect.
The
closing price on the date of the first interest payment date was $0.34 which
means that, under the payment terms of the Convertible Debentures, up to an
additional 970,126 shares of common stock could be issued as interest over the
remaining life of the Convertible Debentures that remain outstanding. Should the
price be greater than $0.34 at the payment date, fewer shares would be
issued.
The
Company elected to pay the interest on its 10% Convertible Debentures due
November 2012 incurred through the first interest payment date, July 15, 2008,
in common stock and received waivers from three of the four holders of the
convertible debentures to pay the interest in common stock notwithstanding the
absence of a registration statement. The interest conversion rate is
defined as the greater of (i) the average of the five closing prices immediately
prior to the applicable interest payment date, (ii) the closing price on the
date of a second closing (which did not occur), and (iii) the closing price on
the date of the first closing (which was $0.34). The average of the
five closing prices prior to July 15, 2008 was $0.358. The dollar amount of
interest incurred from January 3, 2008 (the debenture inception) to July 14,
2008 to be paid in stock amounted to $159,602 which, converted at $0.358 per
share, resulted in an aggregate of 445,815 shares issued to the debenture
holders who agreed to receive interest in the form of common
stock. In addition, the Company made a cash interest payment of
$53,201 to the fourth debenture holder.
F-13
Pharmos
Corporation
Notes
to Consolidated Financial Statements
A
registration statement covering the resale of the shares underlying the
debenture held by one of the four debenture holders, Lloyd I. Miller, III, was
declared effective in December 2008.
In the
first quarter of 2009, the Company elected to pay the interest on its 10%
Convertible Debentures due November 2012 incurred through the second interest
payment date, January 15, 2009, in common stock and received waivers from the
three holders of the convertible debentures for whom there is no resale
registration statement to pay the interest in common stock notwithstanding the
absence of a registration statement. The dollar amount of interest
incurred from July 15, 2008 to January 15, 2009 for all four debenture holders
to be paid in stock amounted to $201,667 which, converted at $0.34 per share,
resulted in an aggregate of 588,236 shares issued to the debenture
holders.
On April
21, 2009, Venrock Associates (which is affiliated with Anthony B. Evnin, a
Director of the Company), New Enterprise Associates (which is affiliated with
Charles W. Newhall, III, a Director of the Company) and Robert F. Johnston, the
Company’s Executive Chairman of the Board of Directors, agreed to convert as of
such date the Company’s 10% Convertible Debentures due November 1, 2012 held by
them, comprising an aggregate of $3,000,000 in principal amount, at a conversion
price of $0.275 per share. Accrued but unpaid interest on their
debentures, aggregating $80,403, was also converted on such date, at a
conversion price of $0.34 per share. An aggregate of 11,145,570
shares was issued upon conversion of the principal and accrued but unpaid
interest on the debentures.
As
described above, $3,000,000 of the convertible debentures were converted on
April 21, 2009 resulting in an issuance of 10,909,091 shares of common stock.
The original conversion price of $0.70 was reduced to $0.275. The remaining
$1,000,000 debenture not converted retains the original $0.70 conversion price.
Conversion of this debenture at $0.70 would result in the issuance of 1,428,571
shares of common stock. The conversion of the debentures at a reduced conversion
price resulted in a non cash debt conversion expense of $596,104 which has been
reflected in the financial statements.
In the
third quarter of 2009, the Company elected to pay the interest on its 10%
Convertible Debentures due November 2012 incurred through the third interest
payment date, July 15, 2009, in common stock to the remaining debenture holder.
The dollar amount of interest incurred from January 15, 2009 to July 15, 2009 to
be paid in stock amounted to $50,000 which, converted at $0.34 per share,
resulted in an aggregate of 147,059 shares issued to the debenture holder. In
January 2010 the company paid in stock to the remaining debenture holder,
interest of $50,000, which was converted at $0.34 per share resulting in an
aggregate of 147,059 shares of common stock.
The
Company incurred $217,083 in financing costs which were capitalized and are
being amortized utilizing an effective interest rate of 7%. $32,432
in costs have been amortized in the twelve months ended December 31,
2009. These costs have been included in interest expense. Three
quarters of the deferred financing costs of $78,382 were written off to equity
on April 21, 2009 when the related debentures were converted. As of December 31,
2009, $13,534 of interest for the fourth debenture remains to be amortized over
the life of the debenture.
|
5.
|
Acquisition
of Vela Pharmaceuticals, Inc.
|
In March 2006, the Company announced an agreement to acquire Vela Pharmaceuticals, Inc. (“Vela”), which has a Phase II product candidate, Dextofisopam, in development to treat IBS. The Company has dedicated substantial resources to complete clinical development of this product candidate. The Vela acquisition also includes additional compounds in preclinical and/or clinical development for neuropathic pain, inflammation and sexual dysfunction. The final merger agreement, as amended, was announced on September 5, 2006 and approved by the Company’s shareholders on October 25, 2006.
F-14
Pharmos
Corporation
Notes
to Consolidated Financial Statements
Under the
amended Merger Agreement, the Company issued 6.5 million shares of common stock
and paid $6 million to Vela shareholders at closing. Pharmos also agreed to
reimburse Vela for $679,000 of operating expenses from July 1, 2006 through
closing. The amended Merger Agreement also includes additional performance-based
milestone payments to the Vela stockholders related to the development of
Dextofisopam, aggregating up to an additional $8 million in cash and the
issuance of up to an additional 13,500,000 shares of Pharmos common stock. In
the event that such shares or payments are issued or funded in future periods, a
determination will then be made as to whether the values are to be written off
as in - process research and development and charged to results of operations;
such future charge could be material. None of the conditions requiring issuance
of these contingent shares or funding these payments were met at December 31,
2008 except for a $1.0 million milestone payment made by Pharmos due upon the
study’s commencement which was paid in 2008.
The
remaining milestones are as follows:
|
|
·
|
$1
million cash + 2 million shares of Pharmos common stock: Final patient
enrolled in Phase 2b trial (1)
|
|
|
·
|
$2
million cash + 2.25 million shares: Successful completion of Phase 2b
(milestone not met)
|
|
|
·
|
$2
million + 2 million shares: NDA
submission
|
|
|
·
|
$2
million cash +2.25 million shares: FDA
approval
|
|
|
·
|
1
million shares: Approval to market in Europe or
Japan
|
|
|
·
|
4
million shares: $100 million sales of Dextofisopam, when and if approved,
in any 12-month period
|
(1) The milestone related the
final patient was enrolled in the Dextofisopam Phase 2b trial was booked in the
first quarter of 2009 as all probability criteria were met. The milestone had
two components, a cash portion of $1,000,000 and a share portion of 2,000,000
shares valued at $180,000. The total charge was $1,180,000. The
shares were issued in November 2009 under the terms of the Amendment #3 to the
agreement and plan of merger. The payment of the cash portion was deferred until
such time as 1) the Company successfully entered into a strategic collaboration
or licensing agreement with a third party for the development of Dextofisopam
resulting in an upfront cash fee of at least $10 million, and 2) payment of the
cash milestone would still leave the Company with one year’s operating cash. The
Company recorded the milestone in the first quarter as it met the accounting
requirements of ASC 450.
The
results of the Phase 2b trial were announced in September 2009 and reported that
while there was clearly drug activity, the trial did not achieve its primary
endpoints. Cowen and Company were subsequently engaged to help the Company
achieve a pharmaceutical partnership. Under the terms of the Vela acquisition
agreement as amended, the 2 million shares were issued on November 2, 2009. The
cash portion was also expensed in Q1 2009 but was reversed in Q4 2009 since it
is not probable that the amended terms will be met in the foreseeable future,
which includes receipt of at least $10 million cash upfront payment. Since the
trial results were not successful, no other milestones have been
achieved.
F-15
Pharmos
Corporation
Notes
to Consolidated Financial Statements
|
6.
|
Fixed
Assets
|
Fixed
assets consist of the following:
|
December
31,
|
||||||||
|
2009
|
2008
|
|||||||
|
Leasehold
improvements
|
$ |
158,023
|
$ |
158,023
|
||||
|
Office
furniture and fixtures
|
87,229
|
87,229
|
||||||
|
Computer
equipment
|
234,538
|
234,538
|
||||||
|
479,790
|
479,790
|
|||||||
|
Less
- Accumulated depreciation
|
(478,411
|
)
|
(470,098
|
)
|
||||
|
Fixed
Assets, net
|
$
|
1,379
|
$
|
9,692
|
||||
Depreciation
of fixed assets was $8,314, $106,236 and $235,134 in 2009, 2008 and 2007,
respectively.
|
7.
|
Grants
for Research and Development
|
During
2009, 2008 and 2007, gross research and development costs amounted to
$4,422,600, $9,028,705 and $11,457,566, respectively.
The
Company had entered into agreements with the State of Israel, which provide for
grants for research and development relating to certain
projects. Amounts received pursuant to these agreements have been
reflected as a reduction of research and development expense. Such
reductions amounted to $0, $0 and $812,042 during 2009, 2008 and 2007,
respectively. The agreements with agencies of the State of Israel place certain
legal restrictions on the transfer of the technology and manufacture of
resulting products outside Israel. The Company will be required to pay
royalties, at rates ranging from 3% to 5%, to such agencies from the sale of
products, if any, developed as a result of the research activities carried out
with the grant funds up to the amount received and interest.
As of
December 31, 2009, the total amounts received under such grants amounted to
$17,897,830. Aggregate future royalty payments related to sales of
products developed, if any, as a result of these grants are limited to
$16,408,890, exclusive of interest, based on grants received through December
31, 2009.
The
Company closed its operations in Israel effective August 31, 2008 and has
subsequently reconciled and repaid a grant in 2009 that was overpaid. All
obligations of the Company have been settled except the requirement to pay
royalties should any of the related technologies be licensed out and further
developed. The Company did not receive any grants in 2008 or 2009 and with the
closure of the Israel location, will not be eligible for further
grants.
|
8.
|
Shareholders
Equity Transactions
|
2009 Transactions
In the
third quarter of 2009, the Company elected to pay the interest on its 10%
Convertible Debentures due November 2012 incurred through the third interest
payment date, July 15, 2009, in common stock to the remaining debenture holder.
The dollar amount of interest incurred from January 15, 2009 to July 15, 2009 to
be paid in stock amounted to $50,000 which, converted at $0.34 per share,
resulted in an aggregate of 147,059 shares issued to the debenture
holder.
F-16
Pharmos
Corporation
Notes
to Consolidated Financial Statements
On May
11, 2009, Robert Johnston, the Executive Chairman, was awarded 1,200,000 shares
of restricted stock at an exercise price of $0.22 per share. 300,000 of such
shares become vested and free from a risk of forfeiture on the first anniversary
of the grant date, and the remaining 900,000 shares shall become vested and free
from a risk of forfeiture in quarterly increments over a three-year period
commencing on the first anniversary of the grant date. Over the four year
period, a total of $264,000 will be recorded as compensation
expense.
On April
21, 2009, the Company completed a private placement of common stock and
warrants. At the closing, the Company issued 18,000,000 shares of
common stock and warrants exercisable for an additional 18,000,000 shares of
common stock for an aggregate purchase price of $1,800,000. The exercise price
of the warrants, which have a five-year term, is $0.12 per share. The
direct costs relating to this offering totaled $20,223.
On April
21, 2009, Venrock Associates (which is affiliated with Anthony B. Evnin, a
Director of the Company), New Enterprise Associates (which is affiliated with
Charles W. Newhall, III, a Director of the Company) and Robert F. Johnston, the
Company’s Executive Chairman of the Board of Directors, agreed to convert as of
such date the Company’s 10% Convertible Debentures due November 1, 2012 held by
them, comprising an aggregate of $3,000,000 in principal amount, at a conversion
price of $0.275 per share. Accrued but unpaid interest on their
debentures, aggregating $80,403, was also converted on such date, at a
conversion price of $0.34 per share. An aggregate of 11,145,570
shares was issued upon conversion of the principal and accrued but unpaid
interest on the debentures.
The
purchase price and debenture conversion price were based on an offer from a
third party on similar financial terms based on certain conditions for a larger
proposed transaction that were not met.
In the
first quarter of 2009, the Company elected to pay the interest on its 10%
Convertible Debentures due November 2012 incurred through the second interest
payment date, January 15, 2009, in common stock and received waivers from the
three holders of the convertible debentures for whom there is no resale
registration statement to pay the interest in common stock notwithstanding the
absence of a registration statement. The dollar amount of interest
incurred from July 15, 2008 to January 15, 2009 to be paid in stock amounted to
$201,667 which, converted at $0.34 per share, resulted in an aggregate of
588,236 shares issued to the debenture holders.
For the
year ended December 31, 2009, there were no options exercised under the
Company’s Stock Option Plans. For the year ended December 31, 2009,
the Company incurred a non-cash charge of $175,438 for stock options to
employees and directors.
As of
December 31, 2009, the Company had reserved 3,647,155 common stock shares for
outstanding stock options. There were 18,000,000 outstanding warrants
as of December 31, 2009.
2008
Transactions
During
the first quarter of 2008, the Company incurred a non-cash charge of $56,250 for
the award of 28,572 shares of common stock each to three departing board members
in recognition for their service and 75,000 shares of common stock awarded to
the President/CFO as part of his annual bonus. These shares were valued at their
fair market value on the date of the awards.
F-17
Pharmos
Corporation
Notes
to Consolidated Financial Statements
In the
third quarter of 2008, the Company elected to pay the interest on its 10%
Convertible Debentures due November 2012 incurred through the first interest
payment date, July 15, 2008, in common stock and received waivers from three of
the four holders of the convertible debentures to pay the interest in common
stock notwithstanding the absence of a registration statement. The dollar amount
of interest incurred from January 3, 2008 (the debenture inception) to July 14,
2008 to be paid in stock amounted to $159,602 which, converted at $0.358 per
share, resulted in an aggregate of 445,815 shares issued to the debenture
holders who agreed to receive interest in the form of common stock. In addition,
the Company made a cash interest payment of $53,201 to the fourth debenture
holder.
|
9.
|
Warrants
|
Some of
the warrants issued in connection with various equity financing and related
transactions contain anti-dilution provisions requiring adjustment. The
following table summarizes the common shares issuable upon exercise of warrants
outstanding as adjusted for the events which have triggered anti-dilution
provisions contained in the respective warrant agreements:
|
Warrants
|
Weighted
Average Exercise Price
|
||||
|
Warrants
Outstanding at 12/31/06
|
424,769
|
$
|
7.02
|
||
|
Cancelled
|
(127,030
|
)
|
$
|
7.10
|
|
|
Warrants
Outstanding at 12/31/07
|
297,739
|
$
|
6.99
|
||
|
Cancelled
|
(297,739
|
)
|
$
|
7.10
|
|
|
Warrants
Outstanding at 12/31/08
|
-
|
-
|
|||
|
Issued
|
18,000,000
|
$ |
0.12
|
||
|
Cancelled
|
-
|
$
|
-
|
||
|
Warrants
Outstanding at 12/31/09
|
18,000,000
|
$
|
0.12
|
||
|
Warrants
Exercisable at 12/31/09
|
18,000,000
|
$
|
0.12
|
||
|
Warrants
Exercisable at 12/31/08
|
-
|
-
|
|||
|
Warrants
Exercisable at 12/31/07
|
297,739
|
$
|
6.99
|
||
|
10.
|
Stock
Option Plans
|
The Company’s shareholders have approved incentive stock option plans for officers and employees. The Company’s shareholders have approved nonqualified stock options for key employees, directors and certain non-employee consultants. Options granted are generally exercisable over a specified period, not less than one year from the date of grant, generally expire ten years from the date of grant and vest evenly over four years.
F-18
Pharmos
Corporation
Notes
to Consolidated Financial Statements
A summary
of the various established stock options plans are as follows:
1997 Plan
and 2000 Plan. The 1997 Plan was and the 2000 Plan is administered by
a committee appointed by the Board of Directors (the “Compensation
Committee”). The Compensation Committee will designate the persons to
receive options, the number of shares subject to the options and the terms of
the options, including the option price and the duration of each option, subject
to certain limitations.
The
maximum number of shares of Common Stock available for issuance under the 1997
Plan was 300,000 shares, as amended, and under the 2000 Plan is 4,700,000
shares, as amended. Each plan is subject to adjustment in the event of stock
splits, stock dividends, mergers, consolidations and the like. Common
Stock subject to options granted under the 1997 Plan and the 2000 Plan that
expire or terminate will again be available for options to be issued under each
Plan.
2009
Incentive Compensation Plan. The 2009 Plan approved at the annual
meeting of shareholders held on July 28, 2009 is administered by the
Compensation Committee, which is authorized to select eligible persons to
receive awards, determine the type, number and other terms and conditions of,
and all other matters relating to, awards and prescribe award
agreements. The total number of shares of common stock reserved and
available for delivery under the 2009 Plan is 8,000,000 shares. The
foregoing limit shall be increased by the number of shares of common stock with
respect to which awards previously granted under the 2009 Plan are forfeited,
expire or otherwise terminate without issuance of shares, or that are settled
for cash or otherwise do not result in the issuance of shares, and the number of
shares that are tendered (either actually or by attestation) or withheld upon
exercise of an award to pay the exercise price or any tax withholding
requirements.
All stock
option grants during 2009 were made from the Pharmos Corporation 2000 Incentive
and Non-Qualified Stock Option Plan.
The
following table summarizes activity in stock options approved by the Company’s
Board of Directors:
|
Option
|
Weighted
Average Exercise Price
|
|||||||
|
Options
Outstanding at 12/31/06
|
1,924,622 | $ | 5.62 | |||||
|
Granted
|
870,000 | $ | 1.70 | |||||
|
Cancelled
|
(374,236 | ) | $ | 7.06 | ||||
|
Options
Outstanding at 12/31/07
|
2,420,386 | $ | 3.99 | |||||
|
Granted
|
854,000 | $ | 0.36 | |||||
|
Cancelled
|
(537,280 | ) | $ | 2.76 | ||||
|
Options
Outstanding at 12/31/08
|
2,737,106 | $ | 3.10 | |||||
|
Granted
|
1,050,000 | $ | 0.22 | |||||
|
Cancelled
|
(139,951 | ) | $ | 2.63 | ||||
|
Options
Outstanding at 12/31/09
|
3,647,155 | $ | 2.29 | |||||
|
Options
exercisable at 12/31/09
|
2,441,814 | $ | 3.26 | |||||
|
Options
exercisable at 12/31/08
|
2,244,735 | $ | 3.63 | |||||
|
Options
exercisable at 12/31/07
|
1,606,763 | $ | 5.12 | |||||
F-19
Pharmos
Corporation
Notes
to Consolidated Financial Statements
Additional
information with respect to the outstanding stock options as of December 31,
2009 is as follows:
|
Options
Outstanding
|
Options
Exercisable
|
||||||||||||||||||
|
Range
of Exercise Prices
|
Options
|
Weighted
Average Remaining Contractual Life
|
Weighted
Average Exercise Price
|
Options
Exercisable
|
Weighted
Average Exercise Price
|
||||||||||||||
| $ | 0.14 - $0.39 | 1,801,000 |
8.5
years
|
$ | 0.27 | 647,693 | $ | 0.32 | |||||||||||
| $ | 1.46 - $2.01 | 526,000 |
5.0
years
|
$ | 1.83 | 474,123 | $ | 1.83 | |||||||||||
| $ | 2.15 - $3.95 | 1,000,510 |
4.9
years
|
$ | 2.41 | 1,000,353 | $ | 2.41 | |||||||||||
| $ | 5.10 - $8.75 | 65,127 |
3.1
years
|
$ | 5.10 | 65,127 | $ | 5.10 | |||||||||||
| $ | 9.38 - $21.20 | 254,518 |
2.8
years
|
$ | 16.26 | 254,518 | $ | 16.26 | |||||||||||
| 3,647,155 |
6.5
years
|
$ | 2.28 | 2,441,814 | $ | 3.26 | |||||||||||||
Fair value of
options:
The fair
value of each option grant was estimated on the date of grant using the
Black-Scholes option valuation model with the following weighted-average
assumptions:
|
2009
|
2008
|
2007
|
|||
|
Risk-free
interest rate
|
1.60-2.46%
|
2.48-3.30%
|
4.20-4.75%
|
||
|
Expected
lives (in years)
|
5.2
|
5.2
|
6.16
|
||
|
Dividend
yield
|
0
%
|
0
%
|
0
%
|
||
|
Expected
volatility
|
97
-121 %
|
80
-83 %
|
79
-80 %
|
||
|
Fair
value
|
$0.17
|
$0.36
|
$1.28
|
Expected Volatility. The
Company calculates the expected volatility of its stock options using historical
volatility of weekly stock prices.
Expected Term.
The expected term is based
on historical observations of employee exercise patterns during the Company’s
history.
Risk-Free Interest
Rate. The interest rate
used in valuing awards is based on the yield at the time of grant of a U.S.
Treasury security with an equivalent remaining term.
Dividend Yield. The Company
has never paid cash dividends, and does not currently intend to pay cash
dividends, and thus has assumed a 0% dividend yield.
Pre-Vesting
Forfeitures. Estimates of
pre-vesting option forfeitures are based on Company experience. The Company will
adjust its estimate of forfeitures over the requisite service period based on
the extent to which actual forfeitures differ, or are expected to differ, from
such estimates. Changes in estimated forfeitures will be recognized through a
cumulative catch-up adjustment in the period of change and will also impact the
amount of compensation expense to be recognized in future
periods.
During
the years ended December 31, 2009, 2008 and 2007, employees and outside
directors of the Company were granted stock options under the Pharmos 2000 Stock
Option Plan in the amount of 1,050,000 or a fair value of
$182,419, 854,000 or a fair value of $303,300, and 870,000 options,
or a fair value of $1,117,347, respectively. The fair value of options that
vested during the years ended December 31, 2009, 2008 and 2007, without
considering forfeitures, was $132,766, $232,435and $1,018,539,
respectively.
F-20
Pharmos
Corporation
Notes
to Consolidated Financial Statements
Total
compensation cost related to non-vested awards is approximately $460K which will
be recognized over a weighted average exercise period of three
years.
|
11.
|
Income
Taxes
|
For 2009, 2008 and 2007, the Company has not recorded a tax benefit on the operating losses generated by U.S and Israeli operations. After an assessment of all available evidence, including historical and forecasted operating results, management has concluded that realization of the Company’s net operating loss carryforwards (“NOLs”), which includes Vela’s historical NOLs and research tax credits generated through the acquisition date, and other deferred tax assets could not be considered more likely than not. Based on this assessment, the Company has increased (decreased) the valuation allowance established on deferred tax assets by approximately ($7,967,000), $4,197,000 and $3,177,000 in 2009, 2008 and 2007, respectively. The decrease in the valuation allowance for the year ended December 31, 2009 relates to reduced deferred tax assets available to the Company due to the expiration of Federal NOLs and tax credits, the liquidation of the Israeli operation (and the resulting loss of those NOLs), the sale of NJ NOLs (discussed in the following paragraph), and a remeasurement in the amount of future compensation deductions on expired/cancelled options and stock awards. A substantial portion of the 2008 and 2007 increases relates to the valuation allowances established on current years’ NOLs.
In 2009,
2008, and 2007, the Company sold $49,707,690, $15,290,510 and $12,136,911,
respectively, of its State Net Operating Loss carryforwards under the State of
New Jersey’s Technology Business Tax Certificate Transfer Program (the
“Program”). The Program allows qualified technology and biotechnology businesses
in New Jersey to sell unused amounts of net operating loss carryforwards and
defined research and development tax credits for cash. The proceeds from the
sale in 2009, 2008, and 2007, net of commissions, were $3,914,481, $1,204,126
and $955,782, respectively, and such amounts were recorded as a tax benefit in
the accompanying statements of operations. The State renews the Program annually
and currently limits the aggregate proceeds to $60,000,000. We cannot be certain
if we will be able to sell any of our remaining or future New Jersey loss
carryforwards or tax credits under the Program.
For 2009,
2008 and 2007, the Company’s recorded tax benefit differs from the benefit
calculated by applying the statutory U.S. Federal income tax rate due to the
valuation allowances established on deferred tax assets in those periods and
non-deductible charges offset by the aforementioned tax benefits from the sale
of New Jersey NOLs.
At
December 31, 2009 and 2008, the Company’s deferred tax assets are comprised of
the following:
|
2009
|
2008
|
|||||||
|
Domestic
NOLs
|
$ | 73,004,000 | $ | 79,876,000 | ||||
|
Israeli
NOLs
|
- | 827,000 | ||||||
|
Research
and Development Credit Carryforwards
|
7,380,000 | 6,990,000 | ||||||
|
Accrued
expenses, compensation and other
|
664,000 | 1,322,000 | ||||||
|
Net
Deferred Tax Assets
|
81,048,000 | 89,015,000 | ||||||
|
Valuation
allowance
|
(81,048,000 | ) | (89,015,000 | ) | ||||
| $ | - | $ | - | |||||
F-21
Pharmos
Corporation
Notes
to Consolidated Financial Statements
At
December 31, 2009 the Company had net operating losses of approximately $206
million and $52 million for U.S and New Jersey tax return purposes,
respectively. Of these amounts, approximately $60 million and $42 million,
respectively, relate to Vela’s results of operations prior to the acquisition.
Management believes that Vela’s State of New Jersey NOL will not qualify to be
available for sale under the Program. Net operating losses for Israel
tax return purposes approximated $ 0 and $3 million at December 31, 2009 and
2008, respectively, as the Company is currently in the final stages of a
voluntary liquidation of its operations in Israel. As a result of previous
business combinations and changes in its ownership, there is a substantial
amount of U.S. NOLs that may be subject to annual limitations on utilization.
The remaining U.S. NOLs begin to expire in 2010 and continue to expire through
2029.
The Vela
acquisition has been regarded as tax free reorganization, although no assurances
can be given to this treatment, within the meaning of Section 368(a) of the
Internal Revenue Code.
At
December 31, 2009 the Company had 2008, 2007 and 2006 tax returns that remain
open and are subject to audit.
|
12.
|
Commitments
and Contingencies
|
Leases
The
Company leases research and office facilities in Israel and New
Jersey. The facilities in Israel were used in the operation of the
Company’s research and development activities.
All of
the leases described above call for base rentals, payment of certain building
maintenance costs (where applicable) and future increases based on the consumer
price indices.
At
December 31, 2009, the future gross minimum lease commitments with respect to
non-cancelable operating leases (including office and equipment leases) with
initial terms in excess of one year are as follows:
|
Lease
Commitments
|
|||
| 2010 | $ | 45,945 | |
| 2011 | - | ||
| 2012 | - | ||
| 2013 | - | ||
| 2014 | - | ||
| $ | 45,945 | ||
Rent
expense during 2009, 2008 and 2007 amounted to $182,314, $252,469 and $447,316,
respectively. In 2009, 2008 and 2007, rent expense is net of sublease income of
$37,466, $140,347 and $100,713, respectively. A sublease agreement expired on
March 31, 2007 and was at an annual rate of $97,630. A second
sublease entered into March 2006 is for $37,466 annually and expired on December
2009.
Consulting
contracts and employment agreements
In the
normal course of business, the Company enters into annual employment and
consulting contracts with various employees and consultants.
F-22
Pharmos
Corporation
Notes
to Consolidated Financial Statements
|
13.
|
Fair
Value Measurements
|
The
Company has estimated the fair value of the $1,000,000 outstanding convertible
debenture due November 1, 2012 to be approximately $400,000 at December 31,
2009. In determining the fair value the Company used level 3 inputs
(unobservable) rate of 35%. Management used a discount rate they believe was
most relevant given the business risks described in Note 2 of the financial
statements and because they have been unable to raise third party financing
during the past several years.
|
14.
|
Employee
Benefit Plans
|
The
Company has a 401-K defined contribution profit-sharing plan covering its’ U.S.
employees. Contributions to the plan are based on employer
contributions as determined by the Company and allowable discretionary
contributions, as determined by the Company’s Board of Directors, subject to
certain limitations. Contributions by the Company to this plan amounted to
$16,251, $19,075 and $39,204 in 2009, 2008 and 2007, respectively.
|
15.
|
Segment
and Geographic Information
|
The
Company is active in one business segment: designing, developing, selling and
marketing pharmaceutical products. The Company maintained development operations
in the United States and Israel with the Israel locations activities terminated
effective October 31, 2008. Certain assets and liabilities were
maintained at the Israel location at December 31, 2008.
Geographic
information for the years ended December 31, 2009, 2008 and 2007 are as
follows:
|
2009
|
2008
|
2007
|
||||||||||
|
Net
loss
|
||||||||||||
|
United
States
|
$
|
(2,913,759
|
)
|
$
|
(10,286,538
|
)
|
$
|
(15,553,864
|
)
|
|||
|
Israel
|
(140,575
|
) |
197,132
|
|
(71,961
|
)
|
||||||
|
$
|
(3,054,334
|
)
|
$
|
(10,089,406
|
)
|
$
|
(15,625,825
|
)
|
||||
|
Total
assets
|
||||||||||||
|
United
States
|
$
|
4,669,681
|
$
|
5,417,338
|
$
|
10,827,747
|
||||||
|
Israel
|
19,341
|
554,826
|
1,547,212
|
|||||||||
|
$
|
4,689,022
|
$
|
5,972,164
|
$
|
12,374,959
|
|||||||
|
Long
lived assets, net
|
||||||||||||
|
United
States
|
$
|
1,379
|
$
|
9,692
|
$
|
18,280
|
||||||
|
Israel
|
-
|
-
|
406,178
|
|||||||||
|
$
|
1,379
|
$
|
9,692
|
$
|
424,458
|
|||||||
|
Capital
expenditures, net
|
||||||||||||
|
United
States
|
$
|
-
|
$
|
1,730
|
$
|
9,100
|
||||||
|
Israel
|
-
|
-
|
167,492
|
|||||||||
|
$
|
-
|
$
|
1,730
|
$
|
176,592
|
|||||||
F-23
Pharmos
Corporation
Notes
to Consolidated Financial Statements
|
16.
|
Quarterly
Information (Unaudited)
|
|
Year
ended
|
||||||||||||||||
|
December
31, 2009
|
1st
Quarter
|
2nd
Quarter
|
3rd
Quarter
|
4th
Quarter
|
||||||||||||
|
Operating
Expenses2
|
$
|
3,531,295
|
$
|
1,631,580
|
$
|
1,391,610
|
$
|
(407,101
|
)
|
|||||||
|
Gain
or (loss) from Operations
|
(3,531,295
|
)
|
(1,631,580
|
)
|
(1,391,610
|
)
|
407,101
|
|||||||||
|
Other
income (loss)
|
(147,157
|
)
|
(618,920
|
)
|
(25,697
|
)
|
(29,657
|
)
|
||||||||
|
Net
gain or (loss)1
|
$
|
(3,678,452
|
)
|
$
|
(2,250,500
|
)
|
$
|
(1,417,307
|
)
|
$
|
4,291,925
|
|||||
|
Net
gain or (loss) per share - basic and diluted*
|
$
|
(.14
|
)
|
$
|
(.05
|
)
|
$
|
(.02
|
)
|
$
|
.07
|
|||||
|
Year
ended
|
||||||||||||||||
|
December
31, 2008
|
1st
Quarter
|
2nd
Quarter
|
3rd
Quarter
|
4th
Quarter
|
||||||||||||
|
Operating
Expenses
|
$
|
3,609,710
|
$
|
2,613,967
|
$
|
2,760,493
|
$
|
2,116,014
|
||||||||
|
Loss
from Operations
|
(3,609,710
|
)
|
(2,613,967
|
)
|
(2,760,493
|
)
|
(2,116,014
|
)
|
||||||||
|
Other
income (loss)
|
14,827
|
(53,591
|
)
|
(83,859
|
)
|
(70,725
|
)
|
|||||||||
|
Net
loss1
|
$
|
(3,594,883
|
)
|
$
|
(2,667,558
|
)
|
$
|
(2,844,352
|
)
|
$
|
(982,613
|
)
|
||||
|
Net
loss per share - basic and diluted*
|
$
|
(.14
|
)
|
$
|
(.10
|
)
|
$
|
(.11
|
)
|
$
|
(.04
|
)
|
||||
*The
addition of earnings (loss) per share by quarter may not equal total earnings
(loss) per share for the year.
|
1.
|
Includes
the sale of the NJ Net Operating Losses in the fourth quarters of 2009 and
2008 of $3,914,481 and $1,204,126,
respectively.
|
|
2.
|
As
further described in Note 5, the Company recorded a charge for $1,000,000
relating to a Vela milestone in the first quarter of 2009. A reassessment
of this milestone in the fourth quarter determined that the milestone no
longer met the probability criteria to be recorded and the charge was
reversed in the fourth quarter 2009. There was no impact on the full
year.
|
F-24
