Attached files
| file | filename |
|---|---|
| EX-10.1 - EX-10.1 - Kindred Biosciences, Inc. | severanceandreleaseagr.htm |
| 8-K - 8-K - Kindred Biosciences, Inc. | kin-20200805.htm |
Exhibit 99.1
Kindred Biosciences Announces Second Quarter 2020 Financial Results
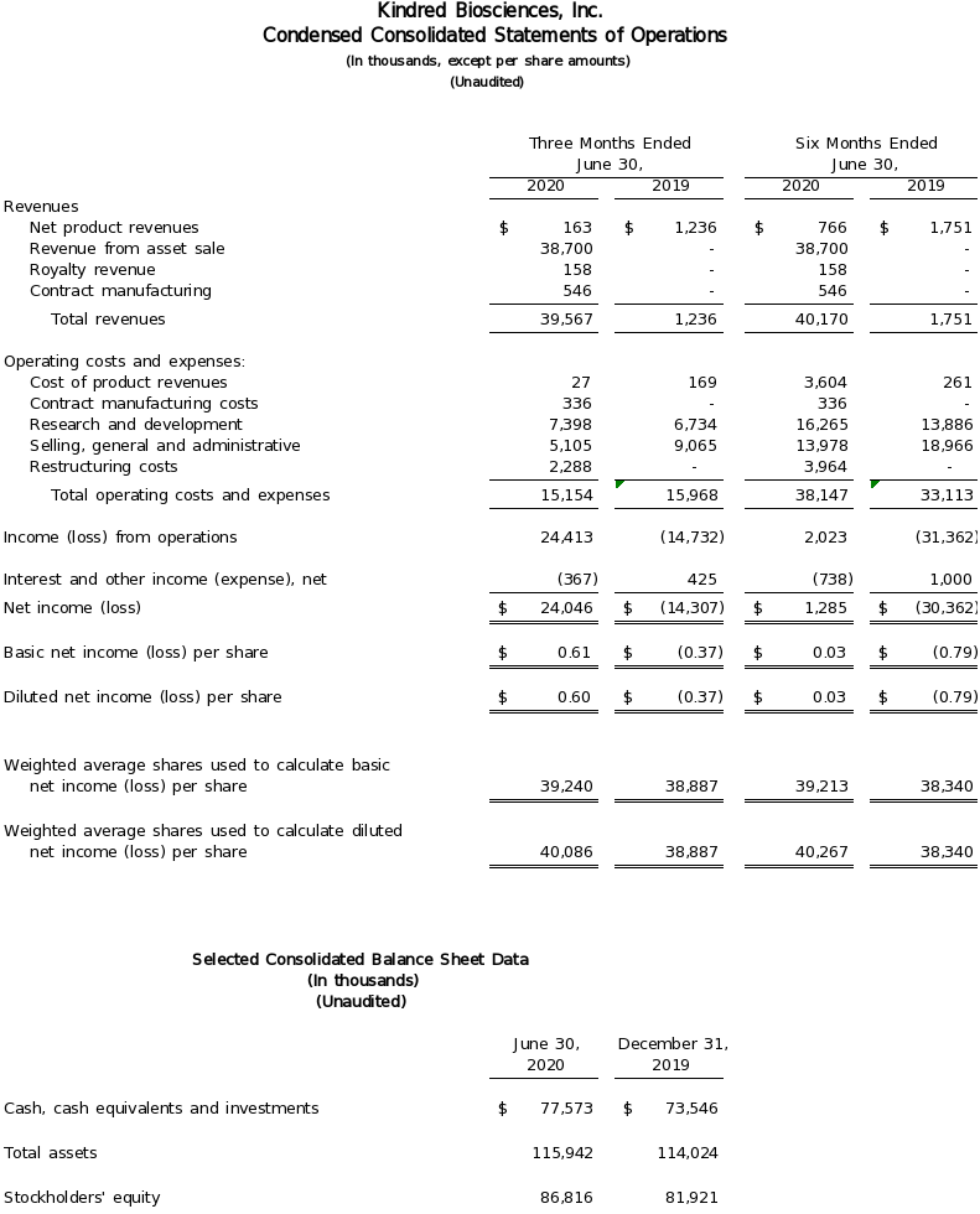
San Francisco, California (August 5, 2020) - Kindred Biosciences, Inc. (NASDAQ: KIN), a biopharmaceutical company focused on saving and improving the lives of pets, today announced financial results for the second quarter ended June 30, 2020 and provided updates on its programs. For the second quarter of 2020, KindredBio reported net revenues of $39.6 million and net income of $24.0 million, or $0.60 per share (diluted), which includes a non-recurring charge of $2.3 million.
“We are pleased with the progress on our pipeline of late-stage biologics,” said KindredBio’s Chief Executive Officer, Richard Chin, M.D. “The commencement of the first parvovirus pivotal study is a key milestone for this program, and we look forward to the upcoming initiation of the tirnovetmab (IL-31 antibody) pivotal study.”
Development and Corporate Updates
Biologics Candidates
•The first of the pivotal studies for KIND-030, a monoclonal antibody targeting canine parvovirus, has commenced and remaining studies will be initiated in the coming months. Pivotal efficacy and safety studies remain on track to be completed by year-end 2020, with approval expected by early 2021. Regulatory approval and review timeline are subject to the typical risks inherent in such a process.
•The scale up process for tirnovetmab (KIND-016), a fully caninized, high-affinity monoclonal antibody targeting interleukin (IL)-31 for the treatment of atopic dermatitis in dogs, is proceeding as planned and the pivotal study is on track to start in the fourth quarter of 2020.
•On December 16, 2019, KindredBio unveiled positive results from its randomized, placebo-controlled laboratory pilot study of KIND-032, a fully caninized monoclonal antibody targeting IL-4R for the treatment of atopic dermatitis in dogs. A second pilot study to further assess efficacy and dosing is planned for the third quarter of 2020.
•The pivotal efficacy study for KindredBio’s feline recombinant erythropoietin was initiated in the fourth quarter. Those veterinary clinics that had suspended clinical trials due to COVID-19 have since resumed operations. KindredBio continues to implement practices consistent with guidance provided by the U.S. Food and Drug Administration (FDA) on studies conducted during the COVID-19 pandemic to minimize the impact on timelines.
•The pilot field effectiveness study for KindredBio's anti-TNF antibody for canine inflammatory bowel disease is underway. Those veterinary clinics that had suspended clinical trials due to COVID-19 have since resumed operations. KindredBio continues to implement practices consistent with guidance provided by the FDA on studies conducted during the COVID-19 pandemic to minimize the impact on timelines. Assuming enrollment continues as expected, completion is now anticipated by year-end 2020.
Mirataz
•The sale of Mirataz to Dechra Pharmaceuticals PLC for an upfront payment of $43 million and royalties on worldwide sales was completed on April 15, 2020, resulting in a partial quarter. Transfer of the asset went smoothly and Dechra recognized significant growth in second quarter Mirataz sales in the US market. Dechra plans to launch Mirataz in the UK and the European Union, and intends to conduct the necessary regulatory activities to achieve approvals in other key international markets. Royalties on future global sales of Mirataz by Dechra will be recorded by KindredBio as revenue.
KindredBio Equine
The strategic evaluation of the future direction of the equine franchise remains ongoing.
•KindredBio recorded Zimeta net product revenues of $7,000 in the second quarter, reflecting a downturn in equine events and transportation as a result of COVID-19. The FDA approved Zimeta for the control of pyrexia in horses in November 2019. The product also received Canadian approval in June 2020.
Second Quarter 2020 Financial Results
For the quarter ended June 30, 2020, KindredBio reported net income of $24.0 million or $0.60 per share (diluted), as compared to a net loss of $14.3 million or $0.37 per share for the same period in 2019. For the six months ended June 30, 2020, net income was $1.3 million or $0.03 per share (diluted), as compared to a net loss of $30.4 million or $0.79 per share for the same period in 2019.
The company recorded $39.6 million and $40.2 million in net revenues in the three and six months ended June 30, 2020, compared with $1.2 million and $1.8 million for the same periods of 2019. The increase in revenue was primarily due to $38.7 million from the sale of Mirataz to Dechra Pharmaceuticals, which was completed on April 15, 2020. Royalty revenue totaled $158,000 in the second quarter.
Substantially all of the product revenues recorded in the first half of 2020 were for Mirataz with $138,000 and $734,000 earned in the three and six months ended June 30, 2020, respectively. Product revenues for Zimeta were $7,000 and $14,000 for the same periods, reflecting a downturn in equine events and transportation as a result of COVID-19. In conjunction with Mirataz and Zimeta, the company also recorded $18,000 in revenue derived from the co-marketing of products for partners, namely Butterfly Networks and Astaria Global.
On May 20, 2020, KindredBio entered into an agreement with Vaxart, Inc. for the manufacture of Vaxart's oral vaccine candidate for COVID-19 and recorded contract manufacturing revenue of $546,000 based on the percentage completion of specific milestones for the quarter.
The cost of product sales totaled $27,000 in the second quarter and $3.6 million for the first six months of 2020, compared to $169,000 and $261,000 for the same periods in 2019. Cost of product sales in 2020 included a $3.5 million inventory write-off on Mirataz due to the transition to Dechra brand labelling. Contract manufacturing costs of $336,000 consisted primarily of the cost of direct materials, direct labor, and overhead costs.
Research and development expenses for the three and six months ended June 30, 2020 were $7.4 million and $16.3 million, respectively, compared to $6.7 million and $13.9 million for the same periods in 2019. Stock-based compensation expense included in research and development expense was $0.5 million and $1.1 million for the three and six months ended June 30, 2020, as compared to $0.5 million and $0.9 million for the same periods in 2019. The $2.4 million year-over-year increase in research and development expenses was primarily due to the inclusion of expenses from the Kansas facility as it began to manufacture clinical trial material. Prior to 2020, construction and commissioning expenditures associated with the Kansas facility had been categorized as general and administrative expenses.
Selling, general and administrative expenses were $5.1 million and $14.0 million for the three and six months ended June 30, 2020, compared to $9.1 million and $19.0 million for the same periods in 2019. The $5.0 million year-over-year decrease is the result of the re-categorization of Kansas plant expenditures as research and development expenses, and lower payroll and related expenses as a result of the elimination of KindredBio’s companion animal sales force. Stock based compensation expense was $1.4 million and $2.9 million for the three and six months in the first half of 2020, versus $1.4 million and $2.8 million in the year-ago period.
The company recorded restructuring charges of $2.3 million and $4.0 million for the three and six months ended June 30, 2020. Restructuring charges were the result of a strategic realignment to a biologics-only company, the prioritization of KindredBio’s late stage programs and an associated workforce reduction.
On April 15, 2020, KindredBio completed the sale of Mirataz to Dechra Pharmaceuticals for an upfront payment of $43 million, of which 10% shall be held in escrow for up to 18 months post closing.
As of June 30, 2020, KindredBio had $77.6 million in cash, cash equivalents and investments, compared with $73.5 million as of December 31, 2019. Net cash provided by operating activities for the first six months of 2020 was approximately $7.1 million, reflecting payment received for the Mirataz asset sale. The Company also invested approximately $2.9 million in capital expenditures for the purchase of associated lab and manufacturing equipment for the Kansas facility.
With respect to spending in 2020, the Company remains focused on advancing its core biologics pipeline and programs, including the commencement of multiple pivotal studies. KindredBio anticipates operating expenses of between $53 million and $55 million, excluding the impact of stock-based compensation expense and the impact of acquisitions, if any. The 2020 operating expense includes the beforementioned restructuring charges, first quarter expenditures that reflect a full organizational structure and second quarter expenditures that reflect various mid-stage development programs that have since been put on hold. Excluding first half expenditures, the annualized run rate for 2020 is expected to be between $41 million and $43 million. KindredBio also plans to invest $3.0 million to $4.0 million in capital expenditures on lab and manufacturing equipment for its biologics programs in 2020. KindredBio believes its existing cash, cash equivalents and investments, the net reduction in the company's workforce, proceeds from the Mirataz sale, and royalties and other revenues from anticipated partnerships will be sufficient to fund the current operating plan through mid-2022, excluding the drawdown of $30 million from its debt facility.
Webcast and Conference Call
KindredBio will host a conference call and webcast today at 4:30 p.m. Eastern time/1:30 p.m. Pacific time. Interested parties may access the call by dialing toll-free (888) 771-4371 from the U.S. or (847) 585-4405 internationally, and using conference ID 49852246. The call will be webcast live here, with a replay available at that link for 30 days.
Important Safety Information
ZimetaTM (dipyrone injection) should not be used more frequently than every 12 hours. For use in horses only. Do not use in horses with a hypersensitivity to dipyrone, horses intended for human consumption or any food producing animals, including lactating dairy animals. Not for use in humans, avoid contact with skin and keep out of reach of children. Take care to avoid accidental self-injection and use routine precautions when handling and using loaded syringes. Prior to use, horses should undergo a thorough history and physical examination by a veterinarian. Monitor for signs of abnormal bleeding and use caution in horses at risk for hemorrhage. Concurrent use with other NSAIDs, corticosteroids and drugs associated with kidney toxicity, should be avoided. As a class, NSAIDs may be associated with gastrointestinal, kidney, and liver toxicity. The most common adverse reactions observed during clinical trials were elevated glucose conversion enzymes, decreased blood protein, and gastric ulcers. Please see the full Prescribing Information.
About Kindred Biosciences
Kindred Biosciences is a biopharmaceutical company developing innovative biologics focused on saving and improving the lives of pets. Its mission is to bring to pets the same kinds of safe and effective medicines that human family members enjoy. The Company’s strategy is to identify targets that have already demonstrated safety and efficacy in humans and to develop therapeutics based on these validated targets for dogs and cats. KindredBio has a deep pipeline of novel biologics in development across many therapeutic classes, alongside state-of-the-art biologics manufacturing capabilities and a broad intellectual property portfolio.
For more information, visit: www.kindredbio.com
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including, but not limited to, statements regarding our expectations about the trials, regulatory approval, manufacturing, distribution and commercialization of our current and future product candidates, and statements regarding our anticipated revenues, expenses, margins, profits and use of cash.
These forward-looking statements are based on our current expectations. These statements are not promises or guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results to be materially different from any future results expressed or implied by the forward-looking statements. These risks include, but are not limited to, the following: our limited operating history and expectations of losses for the foreseeable future; the absence of significant revenue from our products and our product candidates for the foreseeable future; the likelihood that our revenue will vary from quarter to quarter; our potential inability to obtain any necessary additional financing; our substantial dependence on the success of our products and our lead product candidates which may not be successfully commercialized even if they are approved for marketing; the effect of competition; our potential inability to obtain regulatory approval for our existing or future product candidates; our dependence on third parties to conduct some of our development activities; our dependence upon third-party manufacturers for supplies of our products and our product candidates and the potential inability of these manufacturers to deliver a sufficient amount of supplies on a timely basis; the uncertain effect of the COVID-19 pandemic on our business, results of operations and financial condition; uncertainties regarding the outcomes of trials regarding our product candidates; our potential failure to attract and retain senior management and key scientific personnel; uncertainty about our ability to enter into satisfactory agreements with third-party licensees of our biologic products or to develop a satisfactory sales organization for our equine small molecule products; our significant costs of operating as a public company; potential cyber-attacks on our information technology systems or on our third-party providers’ information technology systems, which could disrupt our operations; our potential inability to repay the secured indebtedness that we have incurred from third-party lenders, and the restrictions on our business activities that are contained in our loan agreement with these lenders; the risk that our 2020 strategic realignment and restructuring plans will result in unanticipated costs or revenue shortfalls; uncertainty about the amount of royalties that we will receive from the sale of Mirataz® to Dechra Pharmaceuticals PLC; our potential inability to obtain and maintain patent protection and other intellectual property protection for our products and our product candidates; potential claims by third parties alleging our infringement of their patents and other intellectual property rights; our potential failure to comply with regulatory requirements, which are subject to change on an ongoing basis; the potential volatility of our stock price; and the significant control over our business by our principal stockholders and management.
For a further description of these risks and other risks that we face, please see the risk factors described in our filings with the U.S. Securities and Exchange Commission (the SEC), including the risk factors discussed under the caption “Risk Factors” in our Annual Report on Form 10-K and any subsequent updates that may be contained in our Quarterly Reports on Form 10-Q filed with the SEC. As a result of the risks described above and in our filings with the SEC, actual results may differ materially from those indicated by the forward-looking statements made in this press release. Forward-looking statements contained in this press release speak only as of the date of this press release and we undertake no obligation to update or revise these statements, except as may be required by law.
The results stated in this press release have not been reviewed by the Food and Drug Administration or the United States Department of Agriculture Center for Veterinary Biologics, as applicable.
Contacts
For investor inquiries:
Katja Buhrer
Katja.buhrer@kindredbio.com
(917) 969-3438