Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Valeritas Holdings Inc. | a8-k31218.htm |

Valeritas
Corporate Presentation
NASDAQ: VLRX
March 2018
©2018 Valeritas, Inc..
Improving health and simplifying life for people with diabetes

©2018 Valeritas, Inc.. 2
NASDAQ: VLRX
John Timberlake, CEO
Improving health and simplifying life for people with diabetes

Forward Looking Statements
This presentation shall not be deemed an offer to sell securities nor a solicitation of an offer to purchase securities. Any sale by the company shall be made pursuant to a
definitive purchase agreement. Unless otherwise stated in this presentation, references to “Valeritas,” “we,” “us,” “our” or “our company” refer to Valeritas
Holdings, Inc.. and its subsidiaries.
This presentation contains estimates, projections and forward-looking statements. Our estimates, projections and forward-looking statements are based on our
management’s current assumptions and expectations of future events and trends, which affect or may affect our business, strategy, operations or financial
performance, including but not limited to our revenue, gross margin and cash-flow break-even projections. Although we believe that these estimates,
projections and forward-looking statements are based upon reasonable assumptions and expectations, they are subject to numerous known and unknown risks
and uncertainties and are made in light of information currently available to us. Many important factors may adversely and materially affect our results as
indicated in forward-looking statements. All statements other than statements of historical fact are forward-looking statements. The words “believe,” “may,”
“might,” “could, “would,” “will,” “aim,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “plan” and similar words are intended to identify estimates,
projections and forward-looking statements. Estimates, projections and forward-looking statements speak only as of the date they are made, and, except to the
extent required by law, we undertake no obligation to update or review any estimate, projection or forward-looking statement because of new information,
future events or other factors.
Our estimates, projections and forward-looking statements may be influenced by one or more of the following factors:
• our history of operating losses and uncertainty regarding our ability to achieve profitability;
• our reliance on V-Go® Wearable Insulin Delivery device, or V-Go, to generate all of our revenue;
• our inability to retain a high percentage of our patient customer base or our significant wholesale customers;
• the failure of V-Go to achieve and maintain market acceptance;
• our inability to operate in a highly competitive industry and to compete successfully against competitors with greater resources;
• competitive products and other technological breakthroughs that may render V-Go obsolete or less desirable;
• our inability to maintain or expand our sales and marketing infrastructure;
• any inaccuracies in our assumptions about the insulin-dependent diabetes market;
• manufacturing risks, including risks related to manufacturing in Southern China, damage to facilities or equipment and failure to efficiently increase
production to meet demand;
• our dependence on limited source suppliers and our inability to obtain components for our product;
• our failure to secure or retain adequate coverage or reimbursement for V-Go by third-party payers;
• our inability to enhance and broaden our product offering, including through the successful commercialization of the V-Go® SIMTM and V-Go® pre-fill;
• our inability to protect our intellectual property and proprietary technology;
• our failure to comply with the applicable governmental regulations to which our product and operations are subject;
• our ability to operate as a going concern; and
• our liquidity.
©2018 Valeritas, Inc.. 3

We know diabetes is a serious problem!
©2018 Valeritas, Inc.. 4
Insulin is the most potent
glucose-lowering agent2
Do we really need
another new insulin?
1. (1) American Diabetes Association Diabetes Fast Facts 2015. Available at
https://www.professional.diabetes.org/content/fast-facts-data-and-statistics-about-diabetes. Accessed March 10, 2018., .
2. ACE/ACE Consensus Statement, ENDOCRINE PRACTICE Vol 23 No. 2 Feb. 2017.
~ 5.6 Million
Patients in U.S. with
Type 2 Diabetes take insulin3
Do we need another
new diabetes drug?
“Drugs don’t work in people who
don’t take them.”
– C. Everett Koop, M.D. (4)
We need to help the 5.6 Million
T2 patients ALREADY on insulin to
ACTUALLY take their insulin
$ 245 Billion
Cost of Diagnosed
Diabetes in U.S. in 20121
3. U.S. Roper Diabetes Market Study provided by GfK Customer Research LLC.
4. Eur J Heart Fail. 2017 Nov;19(11):1412-1413.
1
©2018 Valeritas, Inc.. 5
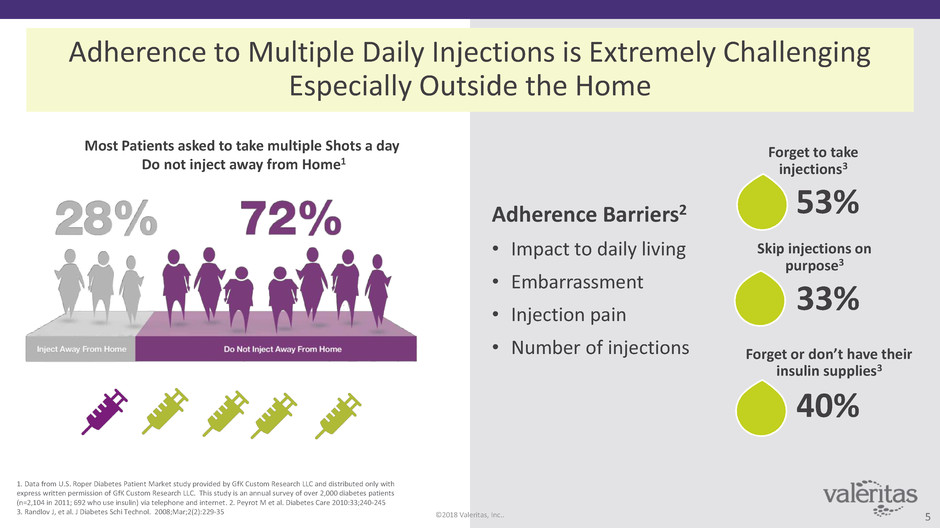
Adherence to Multiple Daily Injections is Extremely Challenging
Especially Outside the Home
Adherence Barriers2
• Impact to daily living
• Embarrassment
• Injection pain
• Number of injections
Forget to take
injections3
53%
Forget or don’t have their
insulin supplies3
40%
Skip injections on
purpose3
33%
1. Data from U.S. Roper Diabetes Patient Market study provided by GfK Custom Research LLC and distributed only with
express written permission of GfK Custom Research LLC. This study is an annual survey of over 2,000 diabetes patients
(n=2,104 in 2011; 692 who use insulin) via telephone and internet. 2. Peyrot M et al. Diabetes Care 2010:33;240-245
3. Randlov J, et al. J Diabetes Schi Technol. 2008;Mar;2(2):229-35
Most Patients asked to take multiple Shots a day
Do not inject away from Home1

©2018 Valeritas, Inc.. 6
Most Patients with Type 2 Diabetes
on Insulin are NOT at Goal1
6
20%
AT A1c Goal
80%
NOT AT A1c Goal
Impact of Improving
Glucose Control2
Deaths from Diabetes
21%
Microvascular Complications
37%
Heart Attacks
14%
Peripheral Vascular Disease
43%
1. Grabner M, et al. Clinicoecon Outcomes Res. 2013;5:471-479 2. Stratton IM et al. BMJ. 2000;321:405-412.
For every 1% reduction in A1C (blood glucose/sugar)
there is a decrease in:
©2018 Valeritas, Inc..
Valeritas’ Mission is for V-Go® to be the Preferred
Insulin Delivery Method for Patients with Type 2 Diabetes
7
Achieving this mission will ELIMINATE the need for Multiple Daily Injections of Insulin

©2018 Valeritas, Inc..
8
The V-Go® Solution
Breakfast
Lunch
Snack
Dinner
Bedtime
Insulin delivery that is worn like a patch
Only FDA & E.U. approved 24-hour, single-use,
fully disposable insulin delivery device
• that provides basal (background) and
bolus (mealtime) insulin delivery
• No batteries, programming or alarms
Designed for the patient with type 2 diabetes
Convenient drug-like fulfilment at the
pharmacy model
Reimbursed by Medicare Part D and most
Commercial Insurance

Valeritas - Entering Growth Stage with Significant Market Opportunity
©2018 Valeritas, Inc.. 9
Entering Growth Stage
• 3rd Consecutive quarter of
growth
• 40% of sales reps were hired
in 2017
• Planned introduction of
Bluetooth V-Go SIMTM
Partnering
• Executed distribution agreements
in Italy & Puerto Rico, and looking
at other International markets
• Evaluating cloud based diabetes
management companies to
partner with V-Go SIMTM
• Exploring insulin providers to
develop V-Go Prefill
Commercial Stage
• $20.2M revenue in U.S. in 2017
• 45% Gross Margin in Q4
• Established reimbursement: Cost
neutral/benefit to patients & payers
• V-Go® proven to deliver strong
clinical and economic benefits
Guidance
• 2018
• $26-$28 Million in
revenue
• 50% gross margin in Q4
* 4.5M patients x $6.50 per V-Go x 360 days x 15% share = approx. $1.6 Billion
Significant Market Opportunity: A 15% market share of U.S. patients with type 2 on insulin but not at their glucose goal would represent $1.6B in revenue*

~
4
.5
M
N
o
t
a
t
A
1c
G
o
a
l
5.6M Patients with Type 2
Diabetes On Insulin1
©2018 Valeritas, Inc.. 10
Significant Market Opportunity for Valeritas in the U.S.
U.S. Type 2 Diabetes
15%
Initial V-Go® Target Market
2M Type 2 on Multiple Daily Shots
Longer-Term V-Go Market
(with V-Go pipeline products)
15%
With National Sales Force
“IF” Market Share V-Go Revenue
15% $700 Million3
V-Go Potential Revenue
With Current Sales Force
“IF” Market Share V-Go Revenue
15% $75 Million2
2.5M on
1-2 Shots
2M on
MDI
1.5M at
A1c Goal
$1.6 Billion V-Go Potential Revenue4
(“IF” at 15% market share)
2. 221,500 Total Meal Time Insulin TRx’s written by Valeritas Reachable Customers x $6.50 x 360 x 15% = ~$75 million
3. 2M patients x $6.50 per V-Go x 360 days x 15% share = ~ $700 million
4. 4.5M patients x $6.50 per V-Go x 360 days x 15% share = ~$1.6B
1 Number of patients with T2DM on insulin are approximate and based on 2014 US Roper Diabetes Patient
Market Study provided by GfK Customer Research LLC and achievement of A1C goal based on Grabner et al.
ClinicoEconomics and Outcomes Research. 2013:5 471–479.
4.5M on
Insulin

©2018 Valeritas, Inc.. 11
Sales & Marketing Model
PATIENT PHYSICIAN
• Target high insulin volume prescribers
• High frequency of office contact
• High support & service for prescriber & patient
• Activate patients to seek and ask for V-Go
• Direct-to-Patient multi-channel marketing
• Patient education and support
WANT PRESCRIBE
EDUCATION AWARENESS
ASK FOR
©2018 Valeritas, Inc.. 12
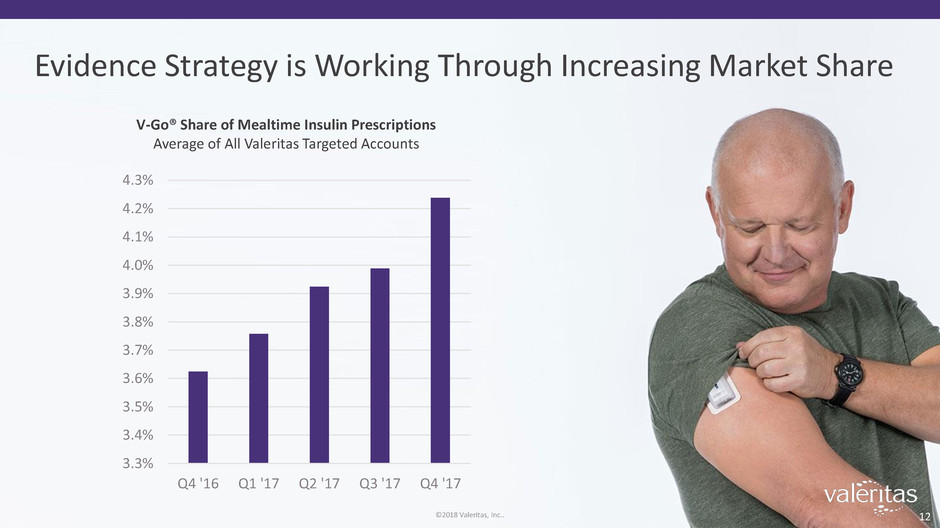
Evidence Strategy is Working Through Increasing Market Share
Q4 '16 Q1 '17 Q2 '17 Q3 '17 Q4 '17
3.3%
3.4%
3.5%
3.6%
3.7%
3.8%
3.9%
4.0%
4.1%
4.2%
4.3%
V-Go® Share of Mealtime Insulin Prescriptions
Average of All Valeritas Targeted Accounts

©2018 Valeritas, Inc.. 13
Targeted Accounts
(Monthly TRx's)
$2,000
$2,500
$3,000
$3,500
$4,000
$4,500
$5,000
$5,500
$6,000
$6,500
Q1'16 Q2'16 Q3'16 Q4'16 Q1'17 Q2'17 Q2'17 Q3'17 Q4'17
Quarterly Revenue
(2015-2017)
Strong Prescription Growth from Targeted Accounts
Driving Accelerating Revenue
Turning point for growth,
fueled by high touch /
higher service model
Turning point for growth,
fueled by high touch /
higher service model

©2018 Valeritas, Inc.. 14
V-Go® is Cost-effective for Both Payers & Patients
Product is Primarily Fulfilled at Retail Pharmacy
*What a Payor Pays (Net of Rebates & Co-pays when V-Go Contracted in Preferred Position)
$17 / day calculated based on published Wholesale Acquisition Prices less estimated rebates and sales deductions.
1.Avg. Nat’l Tier 2 or Tier 3 Co-pay for commercial plans is $31 and $53, respectively, The Kaiser Family Foundation and Health Research & Education Trust Employer Health Benefits 2016 Annual Survey.
Neutral Cost to Patients
(~$17 /day)
between Regimens*
$33
co-pay1
$33
co-pay1
$11
co-pay1
$2.50
Patient co-pay / day
$33
monthly co-pay1
$33 or $57
monthly co-pay1
$2 to $3
Patient co-pay / day
Basal/Bolus
Pen Therapy
V-Go® Therapy
Neutral Cost to Payers

©2018 Valeritas, Inc.. 15
Demonstrated Statistically
Significant Improvements in A1C1-6
Improved Quality of Life1
Lowered Total Daily Dose of Insulin
(Prescribed / Administered)1-6
11
48
Published
Clinical Papers
V-Go Patients
Studied
Presentations
at National
Conferences
1. Rosenfeld CR, et al. Endocr Pract. 2012; 18 (5):660-667. 2. Grunberger, G, et al. Poster presented at 73rd Scientific
Sessions of the ADA; 2013 June 21-25; Chicago, IL. 985-P. 3. Omer, A. et al. Poster presented at 73rd Scientific Sessions of
the ADA; 2013 June 21-25; Chicago, IL. 980-P. 4. Lajara, R, et al. Drugs-Real World Outcomes 2016 Jun 2;3(2):191-199. 5.
Lajara R, et al. Diabetes Ther. 2015;6 (4):531-545.; 6. Lajara R et al. Endocr Pract. 2016 June; 22 (6): 726-725.
Demonstrated Cost Reductions4
Strong Clinical Evidence
1K>

©2018 Valeritas, Inc.. 16
Use of a Simple Titration Approach with V-Go®
Significantly Lowered A1C (glucose)
Use of a weekly physician-driven mealtime dosing titration approach with patients with T2D uncontrolled on prior regimens
13%
Before V-Go
< 7%
< 8%
≤ 9%
On V-Go
47%
40% 87%
67% 100%
A1C Goal Achievement8.7
7.1*
A
1
C
(
%
)
144
60*
TD
D
(
Units
)
Before V-Go On V-Go (4 months)
Texas Health Resources. MOTIV (Managing Optimization and Titration of Insulin Delivery with V-Go) Retrospective Study
T2D=Type 2 diabetes, TDD=Total daily dose of insulin *Significant compared to baseline N=15
Mehta. S, et al. Presented as a poster at OMED, Philadelphia, PA 2017, DTM, Bethesda, MD 2017 and ATTD, Venice, AT, 2018
Hypoglycemia (very low blood glucose) incidence
decreased from 23% at baseline to 7% of patients.
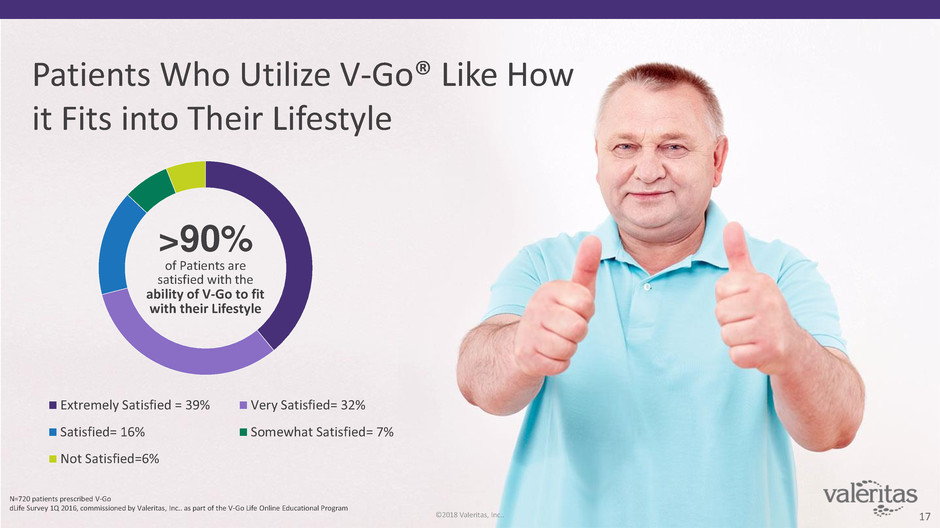
Patients Who Utilize V-Go® Like How
it Fits into Their Lifestyle
Extremely Satisfied = 39% Very Satisfied= 32%
Satisfied= 16% Somewhat Satisfied= 7%
Not Satisfied=6%
>90%
of Patients are
satisfied with the
ability of V-Go to fit
with their Lifestyle
N=720 patients prescribed V-Go
dLife Survey 1Q 2016, commissioned by Valeritas, Inc.. as part of the V-Go Life Online Educational Program
©2018 Valeritas, Inc.. 17

©2018 Valeritas, Inc.. 18
Illustration of Life with Type 2 Diabetes
Thoughts from Actual V-Go® patients like Debbie and Jim
• Living with type 2 diabetes was no longer manageable when my
doctor asked me to take multiple daily shots of insulin.
• Others would tell me it was my fault I had diabetes.
• All the needles and social embarrassment.
• I would rarely if ever take insulin out of my house.
• Constant planning and stress.
• I didn’t feel normal.
• Then I discovered V-Go® and a life without pens, syringes or
pumps.
• It’s simple. It’s private. I’m in control of my treatment.
To hear what patients have to say, visit
https://www.valeritas.com/investors/videos
Debbie
Jim

©2018 Valeritas, Inc.. 19
Valeritas Generated 45% Gross Margins at Significantly Lower Revenue
$5.8
$21.4
$27.0
$61.1
$-
$10
$20
$30
$40
$50
$60
$70
Valeritas Q4'17 Dexcom Q2'11 Tandem Q3'17 Insulet Q3'13
Quarterly Revenue
(1st Q Company reached 45% GM)*
*Respective Company Public Filings. Gross Margins from each
Company and Product may not be directly comparable
Valeritas Projected
Gross Margins
• 50% GM in Q4 2018
with $7M to $8M
quarterly revenue

©2018 Valeritas, Inc.. 20
V-Go® Line Extension Products Provide Path to the
Broader Diabetes & Insulin Market
V-Go® SIMTM
(Simple Insulin Management)2
Expected to:
• Provide connectivity to smart phones and data
aggregation glucose meters and other devices
• Permit tracking and reporting of basal rate and
bolus insulin usage
• Be used as diagnostic tool to make treatment
adjustments
• Increase patient adherence
V-Go®
• Current product
• Filled by patient by transferring insulin
from vial using V-Go EZ Fill
• Commercially available in U.S.
• Approved in E.U. with initial country
distribution agreement executed
V-Go® Prefill2
Expected to:
• Eliminate filling step as it would include a
prefilled insulin cartridge
• Eliminate need for & cost of V-Go EZ Fill
• Lower number of co-pays1
• Provide revenue from insulin
• Expand target population (1st line Insulin)
• Extend patent life to 2032
1. Assumes V-Go devices and insulin cartridges packaged in a single box under a single NDC thereby
potentially reducing the number of prescriptions and the number of co-pays required per patient.
2. Product currently under development.

©2018 Valeritas, Inc.. 21
V-Go® with V-Go® SIMTM Technology (Simple Insulin Management)*
The Future of Type 2 Diabetes Management
PATIENT PHYSICIAN INDUSTRY PAYERS
MANAGEMENT APPLICATIONS
CLOUD HOSTING
Partner
* Product currently under development.
©2018 Valeritas, Inc..
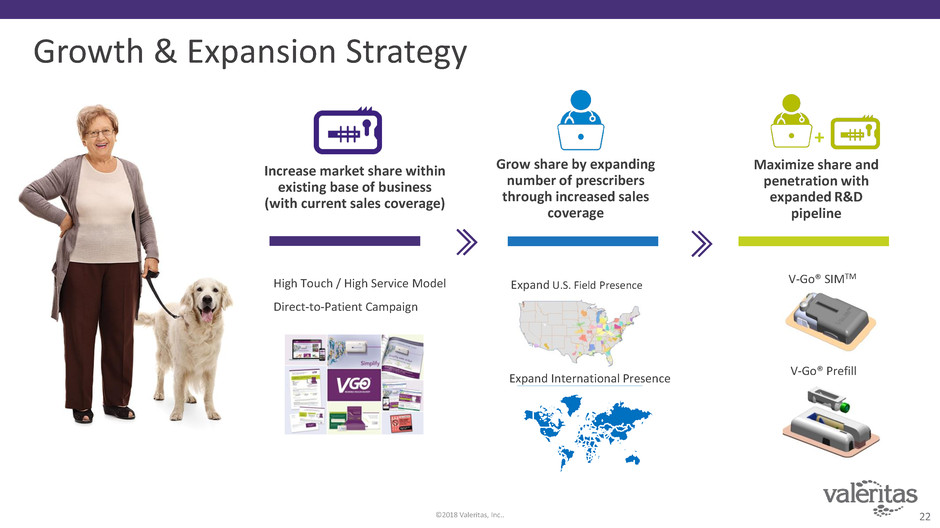
Growth & Expansion Strategy
22
Increase market share within
existing base of business
(with current sales coverage)
Grow share by expanding
number of prescribers
through increased sales
coverage
Maximize share and
penetration with
expanded R&D
pipeline
+
High Touch / High Service Model
Direct-to-Patient Campaign
Expand International Presence
V-Go® Prefill
V-Go® SIMTMExpand U.S. Field Presence

Financial Summary
($ in millions)
4th Quarter Summary 2H Summary
4Q17 4Q16 Change 2H-2017 2H-2016 Change
Revenue $5.8 $4.8 +21% $10.8 $9.7 +12.3%
Gross Profit $2.6 $1.8 +47% $4.6 $3.5 +32.0%
Gross Margin 45.2% 37.1% +810 bp 42.7% 36.3% +640 bp
Net Loss $15.5 $9.8 +57% $25.5 $19.0 +34.2%
Cash Used in Operations $15.7 $10.7 +46.1%
Total Debt1 $36.0 $59.0 -38.9%
Cash and Cash Equivalents* $26.0 $9.9 +163.1%
©2018 Valeritas, Inc.. 23
Valeritas: Financial Summary – December 31, 2017
* Balances as of 12/31/17

Investor Highlights
Strong quarterly growth that will accelerate
in 2018 and beyond
Established reimbursement:
Cost neutral / benefit to patients & payers
V-Go® delivers clinically-relevant glucose
lowering with less insulin
50% Gross Margin in Q4 2018 with ability
to increase with additional sales volume
Substantial market potential with current
V-Go in U.S. and Internationally
Significant market expansion opportunities
with R&D pipeline
©2018 Valeritas, Inc.. 24

©2018 Valeritas, Inc.. 25
Improving health and simplifying life for people with diabetes
NASDAQ: VLRX
March 2018
ART-1236 Rev A 03/2018
Valeritas
Corporate Presentation

APPENDIX
©2018 Valeritas, Inc.. 26

Significant Number of People with Diabetes
Worldwide (2017 and 2045 est.)
©2018 Valeritas, Inc.. 27
IDF Diabetes Atlas, Eighth Edition, 2017
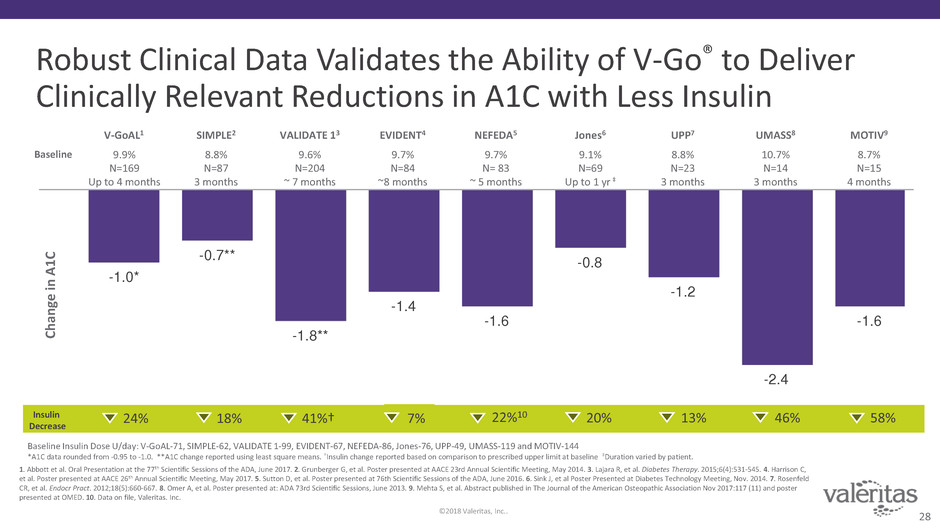
Robust Clinical Data Validates the Ability of V-Go® to Deliver
Clinically Relevant Reductions in A1C with Less Insulin
1. Abbott et al. Oral Presentation at the 77th Scientific Sessions of the ADA, June 2017. 2. Grunberger G, et al. Poster presented at AACE 23rd Annual Scientific Meeting, May 2014. 3. Lajara R, et al. Diabetes Therapy. 2015;6(4):531-545. 4. Harrison C,
et al. Poster presented at AACE 26th Annual Scientific Meeting, May 2017. 5. Sutton D, et al. Poster presented at 76th Scientific Sessions of the ADA, June 2016. 6. Sink J, et al Poster Presented at Diabetes Technology Meeting, Nov. 2014. 7. Rosenfeld
CR, et al. Endocr Pract. 2012;18(5):660-667. 8. Omer A, et al. Poster presented at: ADA 73rd Scientific Sessions, June 2013. 9. Mehta S, et al. Abstract published in The Journal of the American Osteopathic Association Nov 2017:117 (11) and poster
presented at OMED. 10. Data on file, Valeritas. Inc.
-1.0*
-0.7**
-1.8**
-1.4
-1.6
-0.8
-1.2
-2.4
-1.6
Baseline Insulin Dose U/day: V-GoAL-71, SIMPLE-62, VALIDATE 1-99, EVIDENT-67, NEFEDA-86, Jones-76, UPP-49, UMASS-119 and MOTIV-144
*A1C data rounded from -0.95 to -1.0. **A1C change reported using least square means. †Insulin change reported based on comparison to prescribed upper limit at baseline ‡Duration varied by patient.
Insulin
Decrease
18% 41%† 22%10 20% 13% 46% 58%
UPP7 UMASS8
8.8%
N=23
3 months
10.7%
N=14
3 months
9.7%
N= 83
~ 5 months
VALIDATE 13
9.6%
N=204
~ 7 months
Ch
an
ge
in
A1
C
NEFEDA5
9.1%
N=69
Up to 1 yr ‡
Jones6
8.7%
N=15
4 months
MOTIV9SIMPLE2
8.8%
N=87
3 months
V-GoAL1
9.9%
N=169
Up to 4 months
EVIDENT4
9.7%
N=84
~8 months
24% 7%
Baseline
©2018 Valeritas, Inc..
28

Switching from Multiple Daily Injections to V-Go® Results in
Significant Reductions in Total Insulin and A1C (glucose)
©2018 Valeritas, Inc.. 29
117
102
88
61*
M
e
a
n
T
o
ta
l
Da
il
y
D
o
s
e
o
f
In
s
ulin
(
U
/d
a
y
)
Patient
Reported
Prescribed
-1.2*
C
h
a
nge
in
A
1
C
(
%
)
n=86
*p<0.0001 compared to baseline
Lajara R, et al. AACE 24th Annual Scientific and Clinical Congress. May 2015; Nashville, TN
VALIDATE evaluated patients switched to V-Go when Glycemic Control was Suboptimal on Prior Regimens
Multiple Daily Injections (MDI) On V-Go
On V-Go

©2018 Valeritas, Inc.. 30
Prospective Study Conducted by HealthCore, Inc.1 Demonstrated
V-Go® Improved A1C (lowered glucose) with Less Insulin
-0.95*
-0.46*
Chan
ge
in
A
1
C
1 HealthCore, Inc.., an outcomes research subsidiary of Anthem, Inc..
*Significant compared to baseline, Significant between groups
Baseline A1C (%): V-Go 9.88 and STO 9.34 Baseline total daily insulin dose (u/day): V-Go 71 and STO 72
2 Cost includes the WAC cost for all diabetes treatments and medications and based on per patient/day (PPPD) at study end.
The cost is calculated as the sum of published price of insulin, device and concomitant medications.
Abbott, S, et al. Presented as an oral presentation at the 77th ADA Scientific Sessions, San Diego, CA 2017
29%
Less Insulin
with V-Go
vs SOC
at 4 months
(54 vs 72 u/day)
P<0.001
N= 415 patients across 52 Sites with duration of up to 4 months
V-Go was compared to Standard Treatment Optimization (STO) in a
Prospective Pragmatic Clinical Study and Demonstrated Real-World Clinical and Economic Benefits
V-Go proved to be
more cost-effective than STO
V-Go $24.48 vs STO $39.95 per patient per day
for each drop in 1% A1c 2
P = 0.0018
STO
n=246
V-Go
n=169

V-Go® with V-Go SIMTM Technology (Simple Insulin Management)*
The Future of Type 2 Diabetes Management
PATIENT PHYSICIAN INDUSTRY PAYERS
V-Go SIM sends data one-way to
Smart Device. SIM app provides
data on V-Go insulin dosages/timing.
SIM app transfers data to Cloud.
Cloud and other Apps can provide
add’l data to patient. Message by
email and/or secure login.
V-Go SIM will enable simple and
timely access to data that can be
used to improve clinical decision
making and deliver individualized
care. Partnerships will allow the
integration of glucose and insulin
dosing information.
Adherence, utilization and other
health data can improve success
and effectiveness of therapies.
The data can inform clinical trial
development and business
decision making.
Evidence of treatment
effectiveness, adherence
and other data to bolster
payer relationships
MANAGEMENT APPLICATIONS
CLOUD HOSTING
Partner
©2018 Valeritas, Inc.. 31* Product currently under development.

©2018 Valeritas, Inc.. 32
V
A
L
U
E
Scalable Business Model to Drive Shareholder Value
CONTINUED GROTH IN U.S. WITH HIGH TOUCH / HIGH SERVICE MODEL
SHORT-TERM 6 – 18 months MID-TERM 1. – 3 Years LONGER-TERM > 3 years
Advance
V-Go Next
Gen / Line
Extensions
Expand
Sales Force
prudently in
U.S.
Begin to
Commercialize
V-Go® Int’l with
Distrib. Partners
Launch
V-Go SIMTM
(U.S.)
Collaborate
with h-Patch
Technology
Launch
V-Go Prefill
Product
Core Technology= h-patch platform OUS= Outside the United States Line Extensions= V-Go® Link, V-Go® Prefill and future product developments
Expand
Sales Force
in U.S.
Increase
Share in U.S.
Targeted
Accounts
Launch
V-Go SIM
(Int’l)
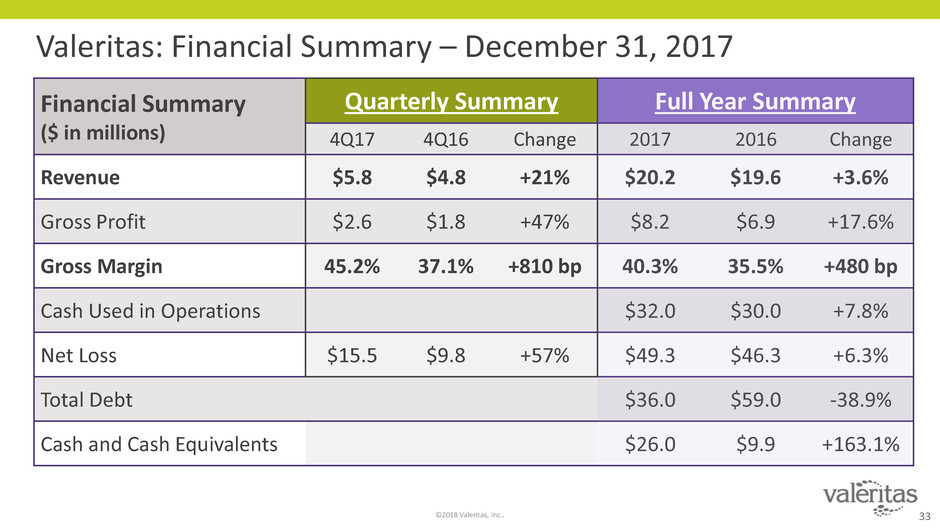
Valeritas: Financial Summary – December 31, 2017
Financial Summary
($ in millions)
Quarterly Summary Full Year Summary
4Q17 4Q16 Change 2017 2016 Change
Revenue $5.8 $4.8 +21% $20.2 $19.6 +3.6%
Gross Profit $2.6 $1.8 +47% $8.2 $6.9 +17.6%
Gross Margin 45.2% 37.1% +810 bp 40.3% 35.5% +480 bp
Cash Used in Operations $32.0 $30.0 +7.8%
Net Loss $15.5 $9.8 +57% $49.3 $46.3 +6.3%
Total Debt $36.0 $59.0 -38.9%
Cash and Cash Equivalents $26.0 $9.9 +163.1%
©2018 Valeritas, Inc.. 33

• Simple filling does not require calculations,
measuring or needles
• Prevents accidental needle sticks
Bolus Chamber
Insulin
Fluid
Fluid
Basal Chamber
Piston
Needle
24 hour Basal rate begins
with the push of a button
4.6 mm, 30 gauge
“Floating needle”
On-demand bolus
function is
manually activated
in 2-step process
Basal rate
flow
restrictor
Basal rate is
spring-driven
V-Go® is Designed for Patient Ease of Use with High Quality Commercial Scale Manufacturing
©2018 Valeritas, Inc.. 34
V-Go Device Overview V-Go® EZ FILLTM
Robust IP with 95 patents issued and 42 pending
