Attached files
As filed with the Securities and Exchange Commission on
February, 22 2018
Registration No. 333-222148
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
S-1/A
(Amendment
No.2)
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
Arrestage International, Inc.
(Exact name of Registrant as specified in its charter)
|
Nevada
|
|
3841
|
|
45-2552289
|
|
(State or other jurisdiction of
incorporation or organization)
|
|
(Primary Standard Industrial
Classification Code Number)
|
|
(I.R.S. Employer
Identification Number)
|
20343 N. Hayden Road, Suite 105
Scottsdale, Arizona 85255
(480)
710.2229
(Address, including zip code, and telephone number, including area
code, of Registrant’s principal executive
offices)
Gary Croft
President and Chief Executive Officer
Arrestage International, Inc.
20343 N. Hayden Road, Suite 105
Scottsdale, Arizona 85255
(480)
710.2229
(Name, address, including zip code, and telephone number, including
area code, of agent for service)
|
Schumacher and Associates, Inc.
7931 S. Broadway St. #314
Littleton, CO 80122
(303) 480-5037
|
|
Gary Croft
Chief Executive Officer
Arrestage International,
Inc.
20343 N. Hayden Road, Suite
105
Scottsdale, Arizona 85255
(480)
710.2229
|
|
Erica L. Jozwiak, Esq.
45 Davis Blvd.
STE. 25
Tampa, Florida 33606
(727)743.6143
|
Approximate date of
commencement of proposed sale to the
public: As soon as
practicable after this Registration Statement becomes
effective.
If
any of the securities being registered on this Form are to be
offered on a delayed or continuous basis pursuant to Rule 415 under
the Securities Act of 1933 check the following
box:
If this Form is filed to register additional
securities for an offering pursuant to Rule 462(b) under the
Securities Act, please check the following box and list the
Securities Act registration statement number of the earlier
effective registration statement for the same
offering. ☐z
If this Form is a post-effective amendment filed
pursuant to Rule 462(c) under the Securities Act, check the
following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same
offering. ☐
If this Form is a post-effective amendment filed
pursuant to Rule 462(d) under the Securities Act, check the
following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same
offering. ☐
Indicate
by check mark whether the registrant is a large accelerated filer,
an accelerated filer, a non-accelerated filer, smaller reporting
company, or an emerging growth company. See the definitions of
“large accelerated filer,” “accelerated
filer”, “smaller reporting company” and "emerging
growth company" in Rule 12b-2 of the Exchange Act. (Check
one):
|
Large accelerated filer
|
☐
|
Accelerated filer
|
☐
|
|
Non-accelerated
filer
|
☐ (Do not check if a smaller reporting
company)
|
Smaller reporting company
|
☒
|
|
|
|
Emerging
growth company
|
☐
|
If an
emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided
pursuant to Section 13(a) of the Exchange Act. ☐
CALCULATION OF REGISTRATION FEE
|
Title of Each
Class of Securities to be Registered
|
Proposed Maximum
Aggregate
Offering
Price (1)
|
Amount
of
Registration
Fee (2)
|
|
Common Stock, par
value $0.001 per share
|
$4,000,000
|
$ 498
|
|
Total
|
$ 4,000,000
|
$ 498
|
|
|
(1)
|
Estimated
solely for the purpose of calculating the amount of the
registration fee pursuant to Rule 457(o) under the Securities Act
of 1933, as amended. Includes offering price of shares that the
underwriters have the option to purchase to cover over-allotments,
if any. Pursuant to Rule 416 under the Securities Act of 1933, as
amended, the shares of common stock registered hereby also include
an indeterminate number of additional shares of common stock as may
from time to time become issuable by reason of stock splits, stock
dividends, recapitalizations or other similar
transactions.
|
|
|
(2)
|
Calculated
pursuant to Rule 457(o) under the Securities Act based on an
estimate of the proposed maximum aggregate offering
price.
|
|
|
(3)
|
Estimated
solely for the purposes of calculating the registration fee
pursuant to Rule 457(g) under the Securities Act.
|
The
Registrant hereby amends this Registration Statement on such date
or dates as may be necessary to delay its effective date until the
Registrant shall file a further amendment which specifically states
that this Registration Statement shall thereafter become effective
in accordance with Section 8(a) of the Securities Act of 1933 or
until the Registration Statement shall become effective on such
date as the Commission, acting pursuant to said Section 8(a), may
determine.
2
EXPLANATORY NOTE
3
This prospectus is not an offer to sell these securities and is not
soliciting an offer to buy these securities in any state or
jurisdiction where the offer or sale is not permitted.
Shares
of Common Stock
ARRESTAGE®
International,
Inc.
Shares of Common Stock
Two Million (2,000,000) Shares - $2.00 per Share
This is
a Best Efforts initial public offering of shares of
common stock of Arrestage International, Inc. No public market
currently exists for our shares. We anticipate that the initial
public offering price of our shares of common stock will be between
$1 and $3 per share.
We will
apply to list our shares of common stock for trading on the OTCQB
under the symbol “_______.” No assurance can be
given that our application will be approved. We are an
“emerging growth company” as defined in the Jumpstart
Our Business Startups Act of 2012 and, as such, we have elected to
comply with certain reduced public company reporting requirements
for this prospectus and future filings.
Investing in our common stock involves a high degree of risk. See
“Risk Factors” beginning on page 19 of this
prospectus for a discussion of information that should be
considered in connection with an investment in our common
stock.
Neither the Securities and Exchange Commission nor any state
securities commission has approved or disapproved of these
securities or determined if this prospectus is truthful or
complete. Any representation to the contrary is a criminal
offense.
|
|
Per
Share
|
Total
|
|
Public offering
price
|
$2.00
|
$4,000,000
|
|
Underwriting
discounts and commissions (1)
|
$0.20
|
$400,000
|
|
Proceeds to
Arrestage International, Inc., before expenses
|
$1.80
|
$3,600,000
|
(1)
No commissions have
been paid to date in exchange for selling or underwriting our
shares.
4
ARRESTAGE®
International,
Inc.

Arrestage Skin Care Products
Arrestage has licensed a line of skin care products that combines
the expertise of herbalists, chemists, and dermatologists to create
products that are customized for each client’s skincare
concerns.
Arrestage only uses all-natural ingredients and components in their
skin care products. All products are monitored closely, with high
freshness, purity, and potency that result from Arrestage’s
micro-batching processes. Over time, as the original customized
formula no longer has the desired results, Arrestage will provide
the client with custom “boosters” to enhance the
original formula’s results.
5
TABLE OF CONTENTS
|
|
|
|
||
|
PROSPECTUS
SUMMARY
|
|
|
9
|
|
|
RISK
FACTORS
|
|
|
17
|
|
|
USE
OF PROCEEDS
|
|
|
42
|
|
|
CAPITALIZATION
|
|
|
45
|
|
|
DILUTION
|
|
|
46
|
|
|
PRINCIPAL
STOCKHOLDERS
|
|
|
48
|
|
|
SHARES
ELIGIBLE FOR FUTURE SALE
|
|
|
52
|
|
|
MATERIAL
U.S. FEDERAL INCOME TAX CONSEQUENCES TO NON-U.S.
HOLDERS
|
|
|
55
|
|
|
LEGAL
MATTERS
|
|
|
60
|
|
|
EXPERTS
|
|
|
60
|
|
|
WHERE
YOU CAN FIND MORE INFORMATION
|
|
|
61
|
|
|
DIVIDEND
POLICY
|
|
|
65
|
|
|
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS
|
|
|
66
|
|
|
BUSINESS
|
|
|
79
|
|
|
MANAGEMENT
|
|
|
84
|
|
|
EXECUTIVE
AND DIRECTOR COMPENSATION
|
|
|
90
|
|
|
CERTAIN
RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
|
|
|
94
|
|
|
UNDERWRITING
|
|
|
102
|
|
|
FINANCIAL
STATEMENTS OF ARRESTAGE INTERNATIONAL, INC.
|
|
|
F-1
|
|
6
We have
not authorized anyone to provide you with any information other
than that contained in this prospectus, any amendment or supplement
to this prospectus or in any free writing prospectus we may
authorize to be delivered or made available to you. The
underwriters and we take no responsibility for, and can provide no
assurance as to the reliability of, any other information that
others may give you. We are offering to sell shares of our common
stock and seeking offers to buy shares of our common stock only in
jurisdictions where offers and sales are permitted. The information
in this prospectus is complete and accurate only as of the date on
the front cover of this prospectus, regardless of the time of
delivery of this prospectus or any sale of shares of our common
stock.
For investors outside the United
States: We have not, and the underwriters
have not, done anything that would permit this offering or
possession or distribution of this prospectus in any jurisdiction
where action for that purpose is required, other than in the U.S.
Persons outside the U.S. who come into possession of this
prospectus must inform themselves about, and observe any
restrictions relating to, the offering of the shares of common
stock and the distribution of this prospectus outside the
U.S.
7
MARKET DATA AND FORECASTS
Unless
otherwise indicated, information in this prospectus concerning
economic conditions, our industry, our markets and our competitive
position is based on a variety of sources, including information
from independent industry analysts and publications, as well as our
own estimates and research. Our estimates are derived from industry
and general publications, studies and surveys conducted by
third-parties, as well as data from our own internal research.
Industry publications, studies and surveys generally state that
they have been obtained from sources believed to be
reliable.
ORGANIZATIONAL FACTS
On June
15, 2011, we formed Arrestage International, Inc. in the state of
Nevada for the purpose of conducting business within the
nutraceutical and medical sectors in perpetuity. The first
organizational meeting was held on June 20, 2011 where the articles
of incorporation and bylaws were adopted. In the same meeting the
stock specimen certificate and the annual meeting set for June
6th each
fiscal year. The registered agent is Nevada Commercial Registered
Agents, LC with offices located at 4231 Dant Blvd, Reno, NV
89509.
FINANCIAL STATEMENT PRESENTATION
The
financial statements as of December 31, 2015 and 2016, are those of
Arrestage International, Inc. and will be presented below in
tabular form with corresponding notes. They have been
professionally prepared by the Company and audited by Schumacher
and Associates, Inc.
TRADEMARKS AND TRADENAMES
This
prospectus includes our trademarks such as Arrestage International,
Inc. ARRESTAGE®
International, Inc. which are each protected under
applicable intellectual property laws and are the property of
Arrestage International, Inc. Solely for convenience, trademarks,
service marks and tradenames referred to in this prospectus may
appear without the®, TM or SM symbols,
but such references are not intended to indicate, in any way, that
we will not assert, to the fullest extent under applicable law, our
rights or the right of the applicable licensor to these trademarks,
service marks and tradenames. This prospectus may also contain
trademarks, service marks, tradenames and copyrights of other
companies, which are the property of their respective owners. The
most recent renewals of registration to the marks to Arrestage
International, Inc.’s trademarks were filed on December of
2017 with the United State’s Patent and Trademark
Office.
ABOUT THIS PROSPECTUS
Except
where the context otherwise requires or where otherwise indicated,
the terms “Arrestage,” “Arrestage
International,” “AII,” “we,”
“us,” “our,” “our company” and
“our business” refer to Arrestage International,
Inc.
8
PROSPECTUS SUMMARY
This summary highlights certain information about us and this
offering contained elsewhere in this prospectus. Because it is only
a summary, it does not contain all the information that you should
consider before investing in shares of our common stock and it is
qualified in its entirety by, and should be read in conjunction
with, the more detailed information appearing elsewhere in this
prospectus. Before you decide to invest in our common stock, you
should read the entire prospectus carefully, including “Risk
Factors” beginning in our prospectus and our financial
statements and the accompanying notes included in this
prospectus.
Market for Securities
Currently
these securities are not offered on a stock exchange or any
exchange traded entity.
The company has a limited operating history, is not generating
revenue, and the auditor has raised substantial concern about the
business as a going concern.
The
Company has a limited operating history and faces all the risks and
uncertainties associated with an unproven history. The Company is
not currently generating revenues and does not expect to generate
revenue in this quarter. The Company has debt from its previous
operating history. The audit firm Schumacher and Associates has
expressed substantial doubt about the Company’s ability to
remain a going concern. Investors should understand that there can
be no assurance that the Company will ever achieve revenues or
profitability. The likelihood of our success must be considered in
light of the problems, expenses, difficulties, complications and
delays frequently encountered in connection with the formation of a
pre-revenue business.
Company Overview
We are
a Company within the healthcare space. Specifically, we are in the
Nutraceutical business that holds formulations on skin care
products as well as brand formulas, and other intellectual
property. In addition, we are seeking to perform roll-up
transactions, to include a finance company that purchases and
securitizes medical devises for periodic rental to end users.
Arrestage is in the sector of research and developement of
nutraceutical products. Arrestage plans to acquire and
maintain secured UCC-1 on the machines it purchases and provides
full service plans and warranty options, and other companies within
the wellness sector.
ARRESTAGE INTERNATIONAL, INC.
(“AII” or the “Company”) a Nevada
corporation, plans to develop an international licensing program
whereby we plan to provide turnkey assistance with operations and
support for distributors in the Skin Care and Anti-Aging arena,
using our brand, custom formulas, trademark, internet presence and
proprietary marketing protocols. In addition to this, we plan to
continue to develop these aspects of the business and potentially
acquire complimentary products and services. Arrestage
International intends to create a significant brand in the
aesthetic skincare marketplace. The officers and directors of AII
all have created, or participated in the marketing and development
of branded products in this space. AII will generate a licensing
fee of 5-8% (or a higher rate the market may bear) of all revenues
generated by future licensees.
AII
plans to license full use of all intellectual property and formulas
of its licensed products, in all geographical regions and channels
of distribution. Arrestage is working on Research and Development
of future products by working towards other possible acquisitions.
AII plans to set itself apart from its competitors by providing
clinically based sales materials that attract a more sophisticated
consumer which may enable the licensee to increase their sales
revenues with higher margins. Licensees will have access to the
luminary panel, clinical and marketing training protocols, and
collateral materials. Arrestage International plans to address an
ever-increasing world demand for western culturally influenced
aesthetic skin topical products, in addition to the increasing
demand in US domestic and other western markets. AII seeks to
license distribution organizations, in the US and Internationally,
to market its proprietary brand of aesthetic dermal formulations
and additional products that it may potentially
acquire.
9
The
initial products have gone through clinical and behavioral testing
and several reformulations to achieve the high acceptance status
that they now hold. The size of the aesthetic topical world market
is approximately $7.4 billion. Morningstar estimates a continued
growth rate of 11% CAGR from 2016-2020. Due to the high margins,
the major aesthetic device makers are attempting to enter this
market as distributors. This may be a beneficial circumstance as it
may enhance the valuation of AII and widen future exit
possibilities.
Medical Device Rental Division
The
Company plans to establish a medical device rental program that
purchases or secures medical devices such as MRI systems and
various other diagnostic equipment. Our target market will be
physician groups, clinics, rural hospitals, and insurance
companies. Arrestage intends to obtain residual income from such
rentals and would maintain a fully secured UCC-1 position on each
piece of equipment. To grow this division, Arrestage plans to
aggressively market such services in this coming year. This
function will help meet the expanding demand of medical systems due
to the current healthcare trends. The United States market, the
largest in the world, has reached a value of $133 billion by 2016,
and is expected to reach 278 billion by 2021 worldwide. (2A) To
date the Company has accumulated a strong market presence within
this lucrative sector of secured structured medical
lending.
Market Opportunity
Medical Equipment Rental is
an industry that is expanding due to many factors in the health
care industry. Professionally, many smaller businesses have found
it much more cost effective to rent expensive equipment that they
would normally have to refer patients to a larger facility to use.
Rentals are creating greater accessibility to medical equipment in
many places where there are not larger facilities close by. Rural
areas that only have local access to small healthcare facilities
could see specialized equipment that would not usually be
available. Many medical professions that deal with varying, rare
health issues, such as emergency clinics, pediatrics, physical
therapists, and oncologists, in small or medium-sized facilities
could have much more reliable access to this specialized equipment.
Also, it is more convenient for patients to receive care and
testing all in one location, and it streamlines the process of
receiving test results when equipment can be found in house.
Lastly, for larger medical facilities renting is beneficial for
very specialized equipment that can be too expensive to purchase.
Rentals allow the use of specialized equipment without undue
burdens on a facility’s budget. Due to the constantly
changing advances in medical technology many places do not have
consistent access to the most helpful equipment. If rented,
adjustments in technology would be much more feasible for tight
facility budgets, and more locations could use the latest
technology in the healthcare market.
●
The global medical
equipment rental market was recorded at US$33417.4 million in 2013
and, at a CAGR of 5.8% during the forecast period of 2014 to 2020,
will reach a value of US$49,112.8 million by 2020
(1A).
10
As
demonstrated in the graphics below, the need for long-term medical
equipment is rising. Geriatric and diabetic populations are two
areas where our services could be used, and that need is
rising.
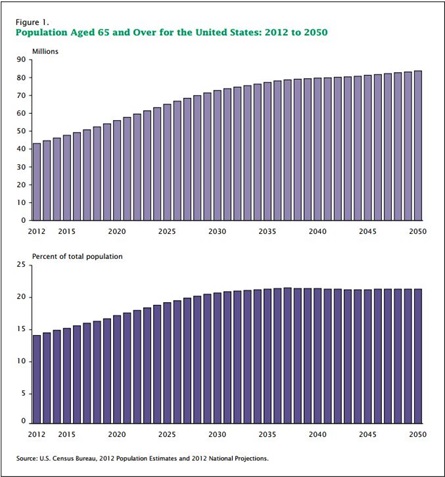
11
Nutraceuticals is an industry that is
rapidly growing, and it is unique because it requires the
overlapping of two industries that are highly relied upon in
today’s society: food and pharmaceuticals. Nutraceuticals are
natural, bioactive chemical compounds that have health-promoting,
disease-preventing or general medicinal properties. This category
encompasses vitamins, minerals, herbal supplements, and certain
animal products. This industry is becoming more popular to
consumers both because greater self-awareness and diet education,
but also because it incorporates preventative and curative care
into everyday life through food, beverages, and supplements. The
governmental support for this industry is positive worldwide, and
in the United States some health insurances reimburse consumers for
medical foods. This industry is largely dominated by a few
companies, with the 5 top companies holding over 50% of the market
as of 2014. North America is expected to continue to lead this
industry over this period (North America held 39.6% market share in
2014). The Asia Pacific region held the second largest market share
in 2014 with a significant growth rate. (source 2A)
●
The global
nutraceuticals market was valued at US$165.62 billion in 2014 and
is expected to reach US$278.96 billion by 2021, growing at a CAGR
of 7.3% from 2015 to 2021. (2A)
●
Revenue-functional
food segment held the largest share in the global nutraceuticals
market, accounting for 31.0% market share in 2014 and is expected
to grow at a CAGR of 7.1% from 2015 to 2021. Functional beverage is
2nd and
expected to grow at a CAGR of 7.4 % from 2015-2021.
(2A)
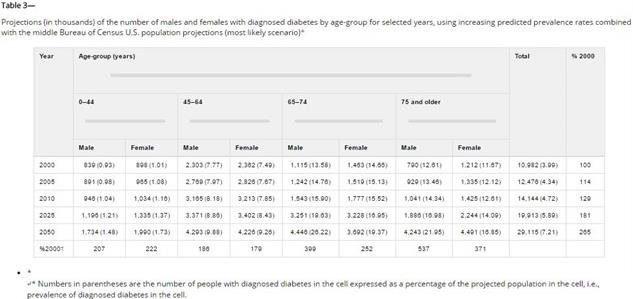
12
●
Top companies are
Cargill, Archer Daniels Midland Company, Royal DSM N.V, BASF SE,
Nestle S.A., General Mills, Inc., etc. (2A)
●
The WHO predicts
that by 2020 over 65 million people will have dementia and that the
prevalence of other neurodegenerative brain diseases, like
Alzheimer’s, is also increasing. (2B)
●
It is estimated
that over 100 million people globally are effected by ‘gut
health’ problems, and the prevalence is rising.
(2B)
●
Cardiovascular
disease (CVD) is significant. It is the number one cause of death
globally. The risk of contracting CVD is associated with lifestyle
choices including a poor diet. (2B)
●
The WHO estimate
that 347 million people worldwide have diabetes. (2B)
●
Sales are predicted
to reach US$250 billion by 2018. (2B)
SOURCES
(1A)-
http://www.transparencymarketresearch.com/medical-equipment-rental.html
(1B)-see
scan attached in email
(1C)-
https://www.census.gov/prod/2014pubs/p25-1140.pdf
(1D)-
http://care.diabetesjournals.org/content/24/11/1936#T3
(2A)-
http://www.prnewswire.com/news-releases/nutraceuticals-market-to-surpass-us278-billion-globally-by-2021-propelled-by-soaring-demand-for-functional-foods-and-beverages-transparency-market-research-531577541.html
(2B)- https://www.kpmg.com/ID/en/industry/CM/Documents/neutraceuticals-the-future-of-intelligent-food.pdf
Forward looking Statements: This prospectus contains
statements that express our opinions, expectations, beliefs, plans,
objectives, assumptions or projections regarding future events or
future results, in contrast with statements that reflect historical
facts. Many of these statements are contained under the headings
“Prospectus Summary,” “Management’s
Discussion and Analysis of Financial Condition and Results of
Operations” and “Business.” In some cases, we
have identified such forward-looking statements with typical
conditional words such as “anticipate,”
“intend,” “believe,”
“estimate,” “plan,” “seek,”
“project” or “expect,” “may,”
“will,” “would,” “could” or
“should,” the negative of these terms or other
comparable terminology.
Important factors
related to forward-looking statements may include, among others,
assumptions regarding:
●
our
ability to achieve and sustain profitability;
●
market
acceptance of our product line;
●
our
ability to successfully commercialize our products;
●
our
ability to compete effectively in selling our products and
services;
13
●
our
ability to expand, manage and maintain our direct sales and
marketing organizations;
●
our
actual financial results may vary significantly from forecasts and
from period to period;
●
our
ability to successfully develop new products, improve or enhance
existing products or acquire complementary products, technologies,
services or businesses;
●
our
ability to obtain and maintain intellectual property of sufficient
scope to adequately protect our products;
●
market
risks regarding consolidation in the healthcare and nutraceutical
industry;
●
our
ability to effectively manage our anticipated growth;
●
the
regulatory requirements applicable to us and our
competitors;
●
our
reliance on third party manufacturers and sole- or single-source
suppliers;
●
our
ability to efficiently manage our supply chain
processes;
●
the
regulatory and legal risks, and certain operating risks, that our
international operations subject us to;
●
the
fact that product quality issues or product defects may harm our
business; and
●
any
product liability claims.
Forward-looking
statements are not a guarantee of performance, and you should not
place undue reliance on such statements. We have based
forward-looking statements largely on our current expectations and
projections about future events. Forward-looking statements are
subject to many uncertainties and other variable circumstances,
including those discussed elsewhere in this prospectus under the
headings “Risk Factors” and “Management’s
Discussion and Analysis of Financial Condition and Results of
Operations,” many of which are outside of our control, that
could cause our actual results and experience to differ materially
from any forward-looking statement. Given these risks and
uncertainties, you are cautioned not to place undue reliance on
these forward-looking statements. The forward-looking statements
included in this prospectus are made only as of the date hereof. We
do not undertake, and specifically decline, any obligation to
update any such statements or to publicly announce the results of
any revisions to any of such statements to reflect future events or
developments, except as required by law.
14
Nutraceutical Division Strengths
Our
Strategy
Our
strategy is to promote, market and sell our existing product line,
and expand by development and acquisition, a more inclusive product
line. By maintaining our cost-effective structure and leveraging
our vast network of in-house and freelance notable experts in the
field we feel we will be able to penetrate the market place in an
expedited manner. Our partnerships are far reaching within the
medical and entertainment fields. By leveraging those contacts we
will be able to showcase products with endorsements from known
persons and entities.
Exclusive focus on a large,
growing market. The size of the aesthetic
topical world market is rapidly growing. Morningstar estimates a
continued growth rate of 11% CAGR from 2016-2020. Due to the high
margins, the major aesthetic device makers are attempting to enter
this market as distributors. This may be a beneficial circumstance
as it may enhance the valuation of AII and widen future exit
possibilities.
Pursue opportunities to
enhance our product offerings. We intend to
continue to expand applications of our nutraceutical products and
vigorously protect those innovations through patent enforcement and
applications. We may also opportunistically pursue the licensing or
acquisition of complementary products and technologies to
strengthen our market position or improve product
margins.
Expand our sales
organization to support growth. We intend
to expand our highly-trained direct sales organization and broaden
our relationships with distributor partners to increase sales and
drive revenues. We intend to expand this sales initiative to new
regional markets and then worldwide distribution and product
placement.
Acquire new product
line. We are exploring the possibility of
expanding our product line through acquisitions of new products
that will allow additional market share. We may also perform
research and development functions in-house, which could include
the development or acquisition of an entirely new product line or
expansion of the current line. The target products are on the
cutting edge of health applications and import new techniques
integrating Allopathic medicine, Naturopathic medicine, and some
New Age features. Because of the relationship we have built within
our research and development planning and due diligence, we are in
a prime position to view and explore potential products that fit
within our Company. Further, we have the personnel to determine
viability of the marketing and sales of such products. If such
products fit within the Company framework, we can quickly add such
by acquisition.
Price effective ownership
of current product lines. We have been able to acquire a
developed line of formulas that have previously been produced.
Thereby, the cost to bring these products to market is much less
than full development, formulation, and production of a line of
skin care nutraceuticals.
Highly experienced
management and medical advisory team. We
have assembled a senior management team and medical advisory board
with significant experience in the healthcare industry. Our
leadership team has a long track record in introducing our products
to the healthcare market in the field nutraceuticals. Members of
our management team also have experience in product development,
launching new products into the healthcare market and securitizing
and renting medical devices and technology to hospitals and private
healthcare practices through direct sales organizations,
distributors and manufacturers. We also collaborate with a network
of leading medical advisors in the design and use of our
products.
15
Extensive product support
network. In addition to the product line we
offer, we also provide a support network for clinicians and
therapists, which includes site planning and preparation, system
deployment and installation, a national and global network of
medical physicists for system commissioning and calibration, a
dedicated service network, a dedicated clinical applications and
education network and service, and live customer support. We
believe that by offering these dedicated and tailored services we
have enhanced our brand and gained market presence.
Relationships with the
medical community. The board members,
acting on behalf of Arrestage, are all actively involved in
scientific, medical, and commercial organizations and communities.
We have on our management team and Board of Directors, well
respected doctors and entrepreneurs with a medical background. We
anticipate that we will be able to leverage our involvement in this
community to increase awareness of the benefits of radiation
therapy and increase sales of our products.
Medical Device Rental Division- Strengths
Our Strategy
Our
goal is to be a leading medical device procurement and finance
company providing real time solutions for the hospitals, insurance
companies, health care clinics, and medical malpractice law firms.
The key elements of our strategy include:
Leveraging pre-existing
relationships, we will be able to scale the business quickly via
new business acquisition along with factoring commercial paper from
our partners. Arrestage will seek to acquire commercial
paper from firms we currently have a relationship. In addition, we
will grow our existing rental program with expansion to new markets
and additional facilities. Since our principals have a long track
record of procuring such transactions, the length of time to scale
such business will be minimal.
Drive adoption and
awareness of our Companies products to specialists, physicians, and
administrators. We intend to educate
specialists, physicians, on the need for having our devices in
house as opposed to outsourcing these services. We believe that
increased awareness of the benefits having these services in house
will maintain patient loyalty and drive revenue. Additionally, we
believe that our products will allow specialists to treat patients
without having to refer them to specialists for treatment and will
allow for faster in-house diagnosis of a patients
issues.
Provide new technology
products and services. Since acquiring our
current technology, we have developed optional add-on technology
products and service options which have enhanced the operational
capabilities of our desirable device technologies. We believe
continued product offerings will keep us relevant to all of our
clients’ needs.
Expand our sales
organization to support growth. We intend
to expand our highly-trained direct sales organization and broaden
our relationships with distributor partners to increase sales and
drive revenues.
Residuals of our currently
owned products. We intend to hold a secured
position on our technology, thus will be able to maintain a secured
payment stream. In addition, the residual value of our products
will provide back end liquidity and additional rental opportunities
after the current client has relinquished the
machines.
16
Risk Factors - Company
Investing in our
common stock involves substantial risk, and our ability to
successfully operate our business is subject to numerous risks,
including those that are generally associated with our industry.
Any of the risks set forth in this prospectus under the heading
“Risk Factors” may limit our ability to successfully
execute our business strategy. You should carefully consider all
the information set forth in this prospectus and, in particular,
should evaluate the specific risks set forth in this prospectus
under the heading “Risk Factors” in deciding whether to
invest in our common stock. The following is a summary of some of
the principal risks we face:
Undercapitalization; Need for Additional Funding
AII
believes the proceeds from offering contemplated herein, assuming
Two Million “(2,000,000)” Shares are sold in this
Offering “($4,000,000)”, will be sufficient to attain
AII's financing goals for the next twelve months. Additionally,
executive management has not nor will in the near term draw a
salary or executive compensation package. Without realizing all or
a significant portion of the maximum Offering proceeds, AII may not
be able to implement its business plan and may have to seek
alternative sources of funding. AII can also give no assurance
that, even if the shares are completely sold, no additional capital
will be needed eventually. Further, no assurance can be given as to
how much additional working capital will be required or that
additional financing can be obtained, or if obtainable, that the
terms will be satisfactory to AII, or that such financing would not
result in a substantial dilution of shareholder's
interest.
Participation is Subject to Risks of Investing in Micro
Capitalization Companies
AII
believes that certain micro capitalization companies have
significant potential for growth, although such companies generally
have limited product lines, markets, market shares and financial
resources. The securities of such companies, if traded in the
public market, may trade less frequently and in more limited volume
than those of more established companies. Additionally, in recent
years, the stock market has experienced a high degree of price and
volume volatility for the securities of micro capitalization
companies. In particular, micro capitalization companies that trade
in the over-the-counter markets have experienced wide price
fluctuations not necessarily related to the operating performance
of such companies.
No exchange
listing currently exists for investors to sell their securities if
securities become listed. Investors in the common Stock may
not be able to realize the listed Market Price due to a potential
lack of liquidity within the Market on which AII will be eventually
offered.
17
Specific Risk Factors- Nutraceutical Division
High Production of Products will create a considerable amount of
inventory.
In
order to produce our products at affordable prices, we will have to
produce through high volume automated processes. We do not know
whether we will be able to contract efficient, automated, low-cost
assembly capability and processes that will enable us to meet the
quality, price, and production standards, or production volumes
required to successfully mass market our product. Even if we are
successful in developing our high-volume production capabilities
and processes, we do not know whether we will do so in time to meet
our product commercialization schedules or to satisfy the
requirements of customers. Our failure to develop such processes
and capabilities could have a material adverse effect on our
business, results of operations and financial
condition.
Underproduction could lead to undercapitalization of Market
demand.
We may
not meet our product development and commercialization milestones.
We have several development programs that are in the pre-commercial
stage. The success of each formulation development program is
highly dependent on our correct interpretation of commercial market
requirements, and our translation of those requirements into
applicable product specifications and appropriate development
milestones. If we have misinterpreted market requirements, or if
the requirements of the market change, we may develop a product
that does not meet the cost and performance requirements for a
successful commercial product. In addition, if we do not meet the
required development milestones, our commercialization schedules
could be delayed, which could result in potential adverse effects
on our business.
Competitive
Landscape.
Significant
competition for topical aesthetic products may develop or may
develop more slowly than we anticipate which would significantly
harm our revenues and may cause us to be unable to recover the
losses we have incurred and expect to incur in the development of
our products. Significant markets may never develop our product
technology or they may develop more slowly than we anticipate. Any
such delay or failure would significantly harm our revenues and we
may be unable to recover the losses we have incurred and expect to
continue to incur in the development and marketing of our products.
If this were to occur, we may never achieve profitability and our
business could fail. Topical products represent an emerging market,
and whether or not end-users will want to use them may be affected
by many factors, some of which are beyond our control,
including:
●
the emergence of
more competitive technologies and products, including other
products that could render our products obsolete;
●
the future cost of
components that may affect our delivery systems;
●
the regulatory
requirements of agencies, including the development of uniform
codes and standards that may result in higher costs;
●
government support
for preventive medicine may wane;
●
the assembling and
supply costs may escalate to levels that would restrict our
marketing efforts;
●
the perceptions of
consumers regarding the safety of our products;
●
the willingness of
consumers to try new technologies;
●
the continued
development and improvement of existing technologies that we may
not be able to access or license; and
●
the future cost of
components used in existing products.
18
Production could be stifled if production technology
changes
Base
components used in our formulation may eventually not be readily
available on a cost-effective basis, in which case our products may
be unable to compete with existing topical and cosmetic products
and our revenues and results of operations would be materially
adversely affected. If our customers, including retail distributors
are not able to purchase our products at a cost-effective basis or
are unable to purchase them at locations convenient to them, our
products may be unable to compete with existing products and our
revenues and results of operations would be materially adversely
affected. Significant growth in the use of topical types of
products, may require the development of a larger than expected
infrastructure to deliver products to our distributors. There is no
guarantee that such an infrastructure will be developed on a timely
basis or at all.
Even if
our products are readily available, if the effective price is such
that it costs more to use our products than to use products
provided through other manufacturers, we may be unable to compete
successfully with our competition.
Governmental
Policies Could Affect our Revenue or Profitability
Changes
in government policies and regulations could hurt the market for
our products. The OTC topical
and cosmetic market is in a phase where it is currently subject to
limited industry-specific government regulations in the United
States relating to matters such as design, storage, testing of
these products. However, given that their production is subject to
government regulation, we may expect to encounter more industry
specific government regulations in the future in the jurisdictions
and markets in which we operate. To the extent that there may be
implementation of further regulations, delays in gaining such
regulatory approval, may hinder our development and our growth may
be constrained.
The future worldwide demand for our current products and our future
products is uncertain. Our current products and our future products
may not be accepted by hospitals, physicians or patients, and may
not become commercially successful.
Clinicians may not
perceive the benefits of our products and may be reluctant or
unwilling to adopt our products as a treatment option. While we
believe that our products are a better alternative to other
treatments of certain skin conditions, clinicians or physicians who
are accustomed to using other modalities to treat patients may be
reluctant to adopt broad use of our products.
We must
grow markets for our products through education and awareness
programs. While studies have been done, there may be more to
perform, and certain there will be with new projects. The process
of marketing the results of the studies is subject to a peer-review
process. Peer reviewers may not consider the results of studies of
our products and any future products sufficiently novel or worthy
of publication. Failure to have studies of our product accepted may
affect adoption of our products.
19
In Certain Cases, We May need to Artificially
Divide Market Share
In
order to develop certain market share, we may enter into agreements
with customers and partners that require us to provide shared
intellectual property rights in certain situations, and there can
be no assurance that any future relationships that we enter into
will not require us to share some of our intellectual property. Any
change in the strategies of a potential partner could have a
material adverse effect on our business and our future
prospects.
Finally, in some
cases, our relationships may be governed by a non-binding
memorandum of understanding or a letter of intent. We cannot assure
you that we will be able to successfully negotiate and execute
definitive agreements with any of these partners, and failure to do
so may effectively terminate the relevant relationship. We may also
enter into relationships with third-party distributors to supply us
with parts when we experience a shortfall in production or to sell
them our excess capacity, even though they also indirectly compete
with us. In addition, our third-party distributors may require us
to provide volume price discounts and other allowances, or
customize our products, either of which could reduce the potential
profitability of these relationships.
Reliance
on Third Party Suppliers Could be a Barrier
We rely
upon third party suppliers to supply key materials and components
for our products. A supplier’s
failure to supply materials or components in a timely manner, or to
supply materials and components that meet our quality, quantity or
cost requirements, or our inability to obtain substitute sources
for these materials and components in a timely manner or on terms
acceptable to us, may harm our ability to manufacture our products
cost-effectively or at all, and our revenues and gross margins
might suffer.
We may be unable to attract and retain highly qualified personnel,
which could adversely and materially affect our competitive
position.
Our
future success depends on our ability to attract and retain our
executive officers and other key employees. We may be unable to
attract or retain qualified management and other key personnel in
the future due to the intense competition for qualified personnel
among companies in the medical device business and related
industries, particularly in the Phoenix, Arizona area where we are
headquartered. The medical device and nutraceutical industries has
experienced a high rate of turnover of management personnel in
recent years. Consequently, we could have difficulty attracting or
retaining experienced personnel and may be required to spend
significant time and expend significant financial resources in our
employee recruitment and retention efforts. Many of the other
medical device companies with which we compete for qualified
personnel have greater financial and other resources and risk
profiles different from ours. They also may provide more diverse
opportunities and better chances for career advancement. Some of
these characteristics may be more appealing to high quality
candidates than that which we may offer. If we are unable to
attract and retain the necessary personnel to accomplish our
business objectives, we may have difficulty implementing our
business strategy and achieving our business
objectives.
20
If we fail to properly manage our anticipated growth, our business
could suffer.
Our
strategy involves substantial growth. If we experience periods of
rapid growth and expansion, our limited personnel, operational
infrastructure and other resources could be significantly strained.
In particular, the possible internalization of manufacturing, and
anticipated expansion of our direct sales force in the U.S. will
require significant management, financial and other supporting
resources. In addition, in order to manage expanding operations, we
will need to continue to improve our operational and management
controls, reporting and information technology systems and
financial internal control procedures. If we are unable to manage
our growth effectively, it may be difficult for us to execute our
business strategy and our operating results and business could
suffer. Any failure by us to manage our growth effectively could
have an adverse effect on our ability to achieve our
goals.
We may be unable to retain and develop our U.S. sales force and
non-U.S. distributors, which would adversely affect our ability to
meet our revenue targets and other goals. Further, Foreign Markets
May Create a Need for Separate Potentially Costly
Analysis
As we
launch products, increase current sales efforts and expand into new
geographies, we will need to retain, grow and develop our direct
sales personnel, distributors and agents. There is significant
competition for sales personnel experienced in relevant medical
device sales. Upon completion of training, sales representatives
typically require lead time in the field to develop or expand their
network of accounts and achieve the productivity levels we expect
them to reach in any individual territory. If we are unable to
attract, motivate, develop, and retain a sufficient number of
qualified sales personnel, or if the sales representatives do not
achieve the productivity levels expected, our revenue will not grow
as expected, and our financial performance will
suffer.
In
addition, we may not succeed in entering into and maintaining
productive arrangements with an adequate number of distributors
outside of the U.S. that are sufficiently committed to selling our
products in international markets. The establishment and
maintenance of a distribution network is expensive and time
consuming. Even if we engage and maintain suitable relationships
with an adequate number of distributors, they may not generate
revenue as quickly as we expect them to, commit the necessary
resources to effectively market and sell our products, or
ultimately succeed in selling our products. Moreover, if our sales
force and distributors are unable to attract and retain new
customers, we may be unable to achieve our expected growth, and our
business could suffer.
Furthermore, some
of our distributors may market or sell the products of our
competitors. In these cases, the competitors may have the ability
to influence the products that our distributors choose to market
and sell, for example, by offering higher commission payments, or
by convincing the distributors to terminate their relationships
with us, carry fewer of our products or reduce their sales and
marketing efforts for our products. Any of the foregoing would
hinder our ability to meet our revenue targets and other
goals.
21
Our future success depends on our ability to develop, receive
regulatory approval for, and introduce new products or product
enhancements that will be accepted by the market in a timely
manner, and if we do not do so, our results of operations will
suffer.
It is
important to our business that we continue to build a pipeline of
product offerings for the treatment skin conditions and cosmetic
improvements to remain competitive. Consequently, our success will
depend in part on our ability to develop or acquire, and introduce
new products. However, we may be unable to successfully maintain
our regulatory clearance for existing products, or develop, obtain
and maintain regulatory clearance or approval for product
enhancements, or new products, or these products may not be
accepted by clinicians who financially support many of the
procedures performed with our products.
If we
do not develop new products or product enhancements in time to meet
market demand, if there is insufficient demand for these products
or enhancements, or if competitors introduce new products with
enhanced functionalities that are superior to those of ours, then
our results of operations will suffer.
If we acquire other companies or
businesses, we will be subject to risks that could hurt our
operations in the future.
We may
in the future acquire complementary businesses, products or
technologies. Any acquisition may not produce the revenues,
earnings or business synergies anticipated, and any acquired
business, product or technology might not perform as expected. Our
management could spend a significant amount of time, effort and
money in identifying, pursuing and completing acquisitions. If we
complete an acquisition, we may encounter significant difficulties
and incur substantial expenses in integrating the operations and
personnel of the acquired company into our operations. In
particular, we may lose the services of key employees of the
acquired company, and we may make changes in management that impair
the acquired company’s relationships with employees, vendors
and customers. Additionally, we may acquire development-stage
companies that are not yet profitable and require continued
investment, which could decrease our future earnings or increase
our futures losses. Any of these outcomes could prevent us from
realizing the anticipated benefits of an acquisition.
To pay
for an acquisition, we might use equity or cash. Alternatively, we
might borrow money from a bank or other lender. If we use equity,
our stockholders would experience dilution of their ownership
interests. If we use cash or debt financing, our financial
liquidity would be reduced.
Any
acquisition could result in recording significant amounts of
goodwill or other intangible assets, some of which could result in
significant quarterly amortization expense. Moreover, if we
determine during annual reviews or otherwise that an intangible
asset has been impaired, we may need to write off some or all of
its carrying value, resulting in large charges to expense.
Amortization charges and write-downs or write-offs of intangibles
would decrease our future earnings or increase our future
losses.
22
Product liability claims could damage our reputation and adversely
affect our business.
The
design, manufacture and marketing of our products each carry an
inherent risk of product liability claims and other damage claims.
In addition to the exposure we may have for defective products,
clinicians may misuse our products or use improper techniques,
regardless of how well trained, potentially leading to injury and
an increased risk of product liability. A product liability or
other damages claim, product recall or product misuse could require
us to spend significant time and money in litigation, regardless of
the ultimate outcome, or to pay significant damages and could
seriously harm our business.
Future
Acquisitions Could Create Synergistic Issues
We
intend to acquire technologies or companies in the future, and
these acquisitions could disrupt our business and dilute our
shareholders’ interests. We intend to acquire other companies
(and may acquire additional technologies) in the future and we
cannot provide assurances that we will be able to successfully
integrate their operations or that the cost savings we anticipate
will be fully realized. Entering into an acquisition or investment
entails many risks, any of which could materially harm our
business, including:
●
diversion of
management’s attention from other business
concerns;
●
failure to
effectively assimilate the acquired technology, employees or other
assets of the company into our business;
●
the loss of key
employees from either our current business or the acquired
business;
●
assumption of
significant liabilities of the acquisitions.
If we
complete acquisitions, we may dilute the ownership of current
shareholders. In addition, achieving the expected returns and cost
savings from acquisitions will depend in part upon our ability to
integrate the products and services, technologies, research and
development programs, operations, sales and marketing functions,
finance, accounting and administrative functions, and other
personnel of these businesses into our business in an efficient and
effective manner. We cannot ensure that we will be able to do so or
that the acquired businesses will perform at anticipated levels. If
we are unable to successfully integrate acquired businesses, our
anticipated revenues may be lower and our operational costs may be
higher.
Reliance
on Outsourcing for Some Production Could Slow Growth
We have no capacity for producing our
product on a large-scale basis, instead Arrestage has a
co-branding licensing strategy it will implement. Currently there
are no manufacturing contracts for our products; if efforts to
co-brand our products do not materialize as planned, it could
negatively affect profitability. We are reliant on licensed
laboratories to contract manufacture them. Should they be unable to
achieve our required levels of production in a timely manner, then
profitability may be affected.
23
Delays
in Market Viability Could Prove Costly
Any
failures or delays in our testing of our products could negatively
affect our customer relationships and increase our assembling
costs. We intend to regularly test our products during the
manufacturing process and prior to initiating any type of marketing
campaign. Delays in our tests could harm our competitive position
and impair our ability to sell our newly developed or existing
product formulations. Our testing may encounter problems and delays
for a number of reasons, including the failure of our formulation,
the failure of the components of others, the failure to combine
these properly, operator error and the failure to maintain and
service the test prototypes properly. Many of these potential
problems and delays will be beyond our control. Such field test
failures may also negatively affect our relationships with
customers, requiring us to extend the formulation and testing
process longer than anticipated before undertaking commercial
sales.
Competitive
Landscape
The
world anti-aging products market stands enthused by the growing
need for appearance-enhancing and age-defying skin cosmetics among
the aging population according to a new report by Global Industry
Analysts. In addition to changes in lifestyles by modern consumers
to increase their chances of longevity, changing practices in
personal grooming is resulting in more time and money being spent
on external grooming to minimize visible effects of
aging.
This is
leading to increased demand for products, such as, skin lotions,
toners, wrinkle-removal creams, skin whiteners, luxury topical skin
care products, concealers, and cover-ups. The growing acceptance of
vitamins and antioxidants as effective anti-aging nutrients is
expected to prod the growth of anti-aging drugs and
pharmaceuticals. With younger consumers becoming more proactive
about their skin maintenance regime, women aged between 25 years to
30 years are increasingly beginning to use anti-aging creams,
thereby resulting in expanded market opportunities. Manufacturers
are additionally adding fuel to the trend by unveiling new
promotional mother-daughter packages, which are designed to
discreetly inculcate adherence to anti-aging treatment regimens as
early as possible. Growing wariness over harsh chemicals is
expected to lead to increased demand for anti-aging products with
organic, natural, herbal and botanical extracts as active
ingredients according to the report. Anti-aging products, which
include natural ingredients such as chamomile, copper, gold,
minerals and amino acids, are expected to score huge gains in the
marketplace in the upcoming years. Driven by the desire to stay
young and healthy, sales of dietary supplements, vitamins, and
minerals are forecast to rise in the upcoming years.
The
trend towards combining the use of both pharmaceuticals and dietary
supplements in the pursuit of well-being is resulting in these
products shifting image from being just healthcare enhancers to
beauty enhancers. As stated by the recent report published by
Global Industry Analysts, Inc., the United States, and Europe
dominate the world market for anti-aging products, with a combined
share of 62.8% estimated in the year 2007. Global growth is
expected to stem from the anti-aging cosmeceuticals market, has
grown at a CAGR of 9.8% over the years 2001 through 2010. Sales of
anti-aging cosmeceuticals in Asia-Pacific and Latin America, the
two fast growing markets worldwide, rose by U.S. $1.0 billion
between the period, 2007 to 2010. The U.S. dominates the world
anti-aging drugs & pharmaceuticals market, followed closely by
Europe. Global sales in this market, rose by a strong 30.9% between
the period, 2007 to 2010. Leading global, and regional players
operating in the industry include Chanel SA, Christian Dior, Avon,
L'Oréal SA, Revlon, Beiersdorf, Elizabeth Arden Inc., Estee
Lauder, GlaxoSmithKline, Johnson & Johnson, Mary Kay, Procter
& Gamble, Alberto Culver, Robanda International, Clarins,
Shiseido, Valeant Pharmaceuticals International, and Unilever,
among others. The report titled "Anti-Aging Products: A Global
Strategic Business Report" published by Global Industry Analysts,
Inc., provides a comprehensive review of market trends, drivers,
issues, challenges, consumer demographics, purchasing trends,
marketing strategies, competition, players, and products. Richly
annotated with authoritative, and unbiased commentaries, and
hard-to-find statistical facts, the report provides unequivocal
views on future potential while throwing light on the prevailing
climate in key regional markets.
24
Product
markets analyzed in the report include Anti-Aging Drugs &
Pharmaceuticals, Anti-Aging Vitamins, Supplements, & Minerals,
Anti-Aging Cosmeceuticals (Skin Care Cosmeceuticals (Face Care
Cosmeceuticals, Body Care Cosmeceuticals, and Sun Protection
Cosmeceuticals), Hair Care Cosmeceuticals, and Hair Growth
Cosmeceuticals). Latest demand patterns in product markets are
quantified across major geographic market verticals including the
United States, Canada, Japan, France, Germany, United Kingdom,
Italy, Spain, and Russia, Asia-Pacific, Middle East/Africa, and
Latin America. Also provided in the report is an enumeration of
recent mergers, acquisitions, and other strategic industry
activities.
The
Company anticipates that it will face additional competition from
new entrants that may offer significant performance, price,
creative or other advantages over those offered by the Company.
Many of these competitors have greater name recognition and
resources than the Company.
Additionally,
potential competitors with established market shares and greater
financial resources may introduce competing products. Thus, there
can be no assurance that AII will be able to compete successfully
in the future or that competition will not have a material adverse
effect on AII’s results of operations.
While
there is an expansive market that continues to grow to feed the
need of medical practices to access these medical devices, this
also opens up the risk of greater competition as other
company’s also look to build their services. This increase in
need is largely due to the speed at which technology develops and
the rate at which we are enhancing medical care and treatment
options.
Specific Risks - Medical Device Rental Division
If End Users of Our Rental Products do not make timely payments or
are unable to receive payment from third-party payors, our revenues
will be negatively impacted.
With any structured financial
business, especially the rental business, mandating timely
receivables is paramount to business success. Thereby, our ability
to make timely collections and the ability of our end users to make
timely payments is essential to our business success. If our end
users are not able to collect from third party payors, or unable to
utilize our technology in a fashion that is economically viable,
our business will take a substantial downturn and the service of
the payment for utilization of our technology will not be
sustainable without additional funding.
25
If third-party payors do not provide coverage and adequate
reimbursement for the use of our products, our revenue will be
negatively impacted.
In the
U.S., the commercial success of our rental of existing products and
any rental of future products rental will depend, in part, on the
extent to which governmental payors at the federal and state
levels, including Medicare and Medicaid, private health insurers
and other third-party payors provide coverage for and establish
adequate reimbursement levels for procedures using our products.
The existence of coverage and adequate reimbursement for our
products and related procedures by government and private payors is
critical to market acceptance of our existing and future products.
Neither hospitals nor physicians are likely to use our products if
they do not receive adequate reimbursement payments for the
procedures using our products.
Some
private payors in the U.S. may base their reimbursement policies on
the coverage decisions determined by the Centers for Medicare and
Medicaid Services, which administers the Medicare program. Others
may adopt different coverage or reimbursement policies for
procedures performed using our products, while some governmental
programs, such as Medicaid, have reimbursement policies that vary
from state to state, some of which may not pay for our products in
an amount that supports our selling price, if at all. A Medicare
national or local coverage decision denying coverage for any of the
procedures performed with our products could result in private and
other third-party payors also denying coverage. Medicare (part B)
and a number of private insurers in the U.S. currently cover and
pay for both non-melanoma skin cancer and keloid treatments using
the SRT-100. A withdrawal, or even contemplation of a withdrawal,
by Centers for Medicare and Medicaid Services, or CMS, Medicaid or
private payors of reimbursements, or any other unfavorable coverage
or reimbursement decisions by government programs or private
payors, could have a material adverse effect on our
business.
If
sufficient coverage and reimbursement are not available for our
current or future products the demand for our products and,
consequently, our revenues will be adversely affected.
We may be unable to retain and develop our sales force, which would
adversely affect our ability to meet our revenue targets and other
goals.
While
we have an organic sales and service team, as we plan to offer
rental products, increase current sales efforts and expand into new
geographies, we will need to retain, grow and develop our direct
sales personnel, distributors and agents. There is significant
competition for sales personnel experienced in relevant medical
device sales. In addition, the training process is lengthy because
it requires significant education for new sales representatives to
achieve an acceptable level of clinical competency with our
products. Upon completion of training, sales representatives
typically require lead time in the field to develop or expand their
network of accounts and achieve the productivity levels we expect
them to reach in any individual territory. If we are unable to
attract, motivate, develop, and retain a sufficient number of
qualified sales personnel, or if the sales representatives do not
achieve the productivity levels expected, our revenue will not grow
as expected, and our financial performance will
suffer.
26
Furthermore, some
of our distributors may market or sell the products of our
competitors. In these cases, the competitors may have the ability
to influence the products that our distributors choose to market
and sell, for example, by offering higher commission payments, or
by convincing the distributors to terminate their relationships
with us, carry fewer of our products or reduce their sales and
marketing efforts for our products. Any of the foregoing would
hinder our ability to meet our revenue targets and other
goals.
We
are in a highly competitive market segment, which is subject to
rapid technological change. If our competitors are able to offer
rental products and market those that are more effective, less
costly, easier to use or otherwise more attractive than any of our
products, our business will be adversely impacted.
The medical device industry is highly
competitive and subject to technological change. In the arena for
technology and products for use discovery and screening, there may
be technological change that makes current products less
attractive. While we believe our offered products currently have
certain competitive advantages over the products offered by these
competitors, our success depends, in part, upon our ability to
maintain this competitive position. Our competitive
advantages include access to participating physicians that have
contributed to research and development in the past. These
individuals which include our board members have decades of
experience of developing and delivering products to market
internationally. If these competitors offer new products, or
expand their operations, we may be unable to maintain our
competitive advantages over these
competitors.
Furthermore, new
competitors, including companies larger than us, may enter the
market in the future and may offer products with similar or
alternative functionalities. These companies may enjoy several
advantages relative to us, including:
●
greater financial
and human resources for product development, sales and
marketing;
●
greater name
recognition;
●
long-established
relationships with physicians and hospitals;
●
the
ability to offer rebates or bundle multiple product offerings to
offer greater discounts or incentives;
●
more
established distribution channels and sales and marketing
capabilities; and
●
greater experience
in and resources for conducting research and development, clinical
studies, manufacturing, preparing regulatory submissions, obtaining
regulatory clearance or approval for products and marketing cleared
products.
27
Hospitals,
physicians and investors may not view our products as competitive
with other products that are marketed and offered by competitors,
including much larger and more established companies. Our
competitors may offer competing products more rapidly than us or
develop more effective, more convenient or less expensive products
or technologies that render our technology or products less
competitive. If our existing or new competitors are more successful
than us in any of these matters, our business may be
harmed.
Our products may become obsolete prior to the end of their
anticipated useful lives, and we may be required to dispose of
inventory or write off the value or accelerate the depreciation of
these assets, each which would materially and adversely impact our
results of operations.
We will
focus on offering products, from our current formulations, that
have been widely accepted. While we believe this provides a
competitive edge, it also results in the risk that our products may
decay into a competitively weaker position prior to the end of
their anticipated useful lives. If we introduce new products or
next-generation products prior to the end of the useful life of a
prior generation, we may be required to write down the value of
these assets, each of which would materially and adversely impact
our results of operations.
If we are unable to make scheduled interest or principal payments
on our present or future debt obligations, our operations could be
adversely affected.
Our
ability to make scheduled payments on our debt obligations depends
on numerous factors, including the amount of cash balances and
actual and projected financial and operating performance. These
amounts and performance are subject to certain financial and
business factors, as well as prevailing economic and competitive
conditions, some of which may be beyond our control. We may be
unable to maintain a level of cash balances or cash flows from
operating activities sufficient to permit ourselves to pay the
principal and interest on existing or future indebtedness. If cash
flows and capital resources are insufficient to fund debt service
obligations, we may be forced to reduce or delay capital
expenditures, sell assets or operations, seek additional capital or
restructure or refinance our indebtedness. We may be unable to take
any of these actions, and even if able, these actions may be
insufficient to permit us to meet scheduled debt service
obligations.
We are subject to various federal, state and foreign healthcare
laws and regulations, and a finding of failure to comply with these
laws and regulations could have a material adverse effect on our
business.
Our
operations are, and will continue to be, directly and indirectly
affected by various federal and state healthcare laws. Many of the
specific regulatory risks have been discussed in depth below and
within this document including: Federal “Sunshine” (42
U.S. Code §1320a-7h) law and Federal Health Insurance
Portability and Accountability Act of 1996, or HIPAA. Any violation
or change in these regulatory schemes would affect operations,
potentially in a negative way.
28
We or our affiliates are required to comply with medical device
reporting requirements and must report certain malfunctions,
deaths, and serious injuries associated with our products, which
can result in voluntary corrective actions or agency enforcement
actions.
Under
the U.S. Food and Drug Administration medical device reporting
regulations (21 CFR 803), medical device manufacturers are required
to submit information to the U.S. Food and Drug Administration when
they receive a report or become aware that a device has or may have
caused or contributed to a death or serious injury or has or may
have a malfunction that would likely cause or contribute to death
or serious injury if the malfunction were to recur. All
manufacturers placing medical devices on the market in the European
Economic Area are legally bound to report any serious or
potentially serious incidents involving devices they produce or
sell (MEDDEV 2.12-1) to the Competent Authority in whose
jurisdiction the incident occurred through the European Vigilance
process.
If an
event subject to medical device reporting requirements occurs, we
or our affiliates will need to comply with the reporting
requirements, which would adversely affect our reputation and
subject us to actions by regulatory authorities, such as ordering
recalls, imposing fines, or seizing the affected products.
Furthermore, any corrective action, whether voluntary or
involuntary, will require the dedication of time and capital and
will distract management from operating our business. Any of the
foregoing would further harm our reputation and financial
results.
Corporate Management Risks
If we fail to develop or maintain an effective system of internal
controls, we may not be able to accurately report our financial
results or prevent fraud.
Effective internal
controls are necessary for us to provide reliable financial reports
and effectively prevent fraud. If we cannot provide reliable
financial reports or prevent fraud, our reputation and operating
results would be harmed. Our efforts to continue to develop and
maintain internal controls may not be successful, and we may be
unable to maintain adequate controls over our financial processes
and reporting in the future, including compliance with the
obligations under Section 404 of the Sarbanes-Oxley Act of 2002.
Any failure to develop or maintain effective controls, or
difficulties encountered in our implementation or other effective
improvement of our internal controls, could harm our operating
results.
Reliance upon Key Personnel and Necessity of Additional
Personnel
AII is
largely dependent upon the personal efforts and abilities of
existing management, especially Gary Croft (Chief Executive Officer
and Director), Kimberly Shapiro (Vice-Chairman and Director), and
Phillip Nuciola (President of Capital Markets). The success
of AII will also be largely dependent upon the ability of AII to
continue to attract quality management and employees to help
operate AII as its operations may grow.
29
Control by Existing Management
Currently,
officers, directors and founders of AII, as a group directly or
indirectly own approximately 3,451,000 restricted shares of Common
Stock in AII which is 95.86% of the 3,600,000 shares of Common
Stock issued pre-offering or approximately 62% of 5,600,000 shares
of common stock issued post-offering including the sale of all
2,000,000 Shares pursuant to this Offering (e.g. does not take into
account possible payments for acquisitions or other products,
services or intellectual property or processes.)
As a
result, officers, directors and founders of AII as a group could
exercise substantial control over all matters requiring stockholder
approval, including the election of directors and approval of
significant corporate transactions. This concentration of ownership
limits the power to exercise control by the minority shareholders
who will have purchased their stock in this Offering.
Specific Risks Related to our Regulatory Environment
We are subject to various federal, state and foreign healthcare
laws and regulations, and a finding of failure to comply with these
laws and regulations could have a material adverse effect on our
business.
Our
operations are, and will continue to be, directly and indirectly
affected by various federal, state and foreign healthcare laws,
including, but not limited to, those described below.
●
Federal
Anti-Kickback Statute (42 U.S. Code §1320a-7b), which
prohibits any person or entity from knowingly and willfully
offering, paying, soliciting or receiving any remuneration,
directly or indirectly, in cash or in kind, in return for or to
induce the referring, ordering, leasing, purchasing or arranging
for or recommending the referring, ordering, purchasing or leasing
of any good, facility, item or service, for which payment may be
made, in whole or in part, under federal healthcare programs, such
as the Medicare and Medicaid programs.
●
Federal
civil and criminal false claims laws and civil monetary penalty
laws, which prohibit, among other things, persons or entities from
knowingly presenting, or causing to be presented, a false or
fraudulent claim to, or the knowing use of false records or
statements to obtain payment from, or approval by, the federal
government. Suits filed under the False Claims Act, known as
“qui tam” actions, can be brought by any individual on
behalf of the government and such individuals, commonly known as
“whistleblowers,” may share in any amounts paid by the
entity to the government in fines or settlement. When an entity is
determined to have violated the False Claims Act (31 U.S. Code
§3729-3733), it may be required to pay up to three times the
actual damages sustained by the government, plus civil penalties
for each separate false claim.
●
Federal Health
Insurance Portability and Accountability Act of 1996, or HIPAA,
statute, which, among other things, created federal criminal laws
that prohibit knowingly and willfully executing, or attempting to
execute, a scheme or artifice to defraud any healthcare benefit
program and knowingly and willfully falsifying, concealing or
covering up a material fact or making any materially false,
fictitious or fraudulent statements in connection with the delivery
of or payment for healthcare benefits, items or services.
Additionally, HIPAA, as amended by the Health Information
Technology for Economic and Clinical Health Act of 2009, and
applicable implementing regulations, impose certain requirements
relating to the privacy, security and transmission of individually
identifiable health information without appropriate authorization
on entities subject to the law, such as health plans,
clearinghouses, and healthcare providers and their business
associates. Internationally, substantially every jurisdiction in
which we operate has established its own data security and privacy
legal framework with which we must comply, including the Data
Protection Directive 95/46/EC and national implementation of the
Directive in the member states of the European
Union.
30
The off-label use or misuse of our products may harm our image in
the marketplace, result in injuries that lead to costly product
liability suits, or result in costly investigations and regulatory
agency sanctions under certain circumstances.
If the
U.S. Food and Drug Administration determines that our promotional
materials or training constitute promotion of an off-label or other
improper use, it could request that we modify our training or
promotional materials, or subject us to regulatory or enforcement
actions, including the issuance of an untitled letter, a warning
letter, injunction, seizure, civil fine or criminal penalties. It
is also possible that other federal, state or foreign enforcement
authorities might take action if they consider our business
activities to constitute promotion of an off-label use, which could
result in significant penalties, including, but not limited to,
criminal, civil or administrative penalties, damages, fines,
disgorgement, exclusion from participation in government healthcare
programs, and the curtailment of our operations. Any of these
events could significantly harm our business and results of
operations.
These
regulations or codes may limit our ability to affectively market
our products, or we could run afoul of the requirements imposed by
these regulations, causing reputational harm, imposing potentially
substantial costs, and adversely affecting our operations as a
result.
Healthcare policy changes, including recent federal legislation to
reform the U.S. healthcare system, may have a material adverse
effect on our business.
In
March 2010, President Obama signed the Patient Protection and
Affordable Care Act, as amended by the Health Care and Education
Reconciliation Act. This Act includes, among other things, a
deductible 2.3% excise tax on any entity that manufactures or
imports medical devices offered for sale in the U.S., with limited
exceptions, effective January 1, 2013. This excise tax imposed a
significant increase in the tax burden on the medical device
industry, and if our efforts to offset the excise tax are
unsuccessful, the increased tax burden could have an adverse effect
on our results of operations and cash flows. However, this excise
tax has recently been suspended for 2016 and 2017. Other elements
of this Act, including comparative effectiveness research, an
independent payment advisory board, payment system reforms
including shared savings pilots and other provisions, may
significantly affect the payment for, and the availability of,
healthcare services and may result in fundamental changes to
federal healthcare reimbursement programs, any of which may
materially affect numerous aspects of our business.
31
Other
healthcare reform measures may result in more rigorous coverage
criteria and in additional downward pressure on the reimbursement
received for procedures utilizing our products. In addition, other
legislative changes have been proposed and adopted since the Act
discussed above was enacted that may adversely affect our revenues.
These changes include aggregate reductions to Medicare payments to
providers of up to 2% per fiscal year effective April 2013 which,
due to subsequent legislative amendments to the statute, will stay
in effect through 2024 unless additional Congressional action is
taken. Additionally, in January 2013, President Obama signed into
law the American Taxpayer Relief Act of 2012, which, among other
things, reduced Medicare payments to several providers, and
increased the statute of limitations period for the government to
recover overpayments to providers from three to five years. These
new laws may result in additional reductions in Medicare and other
healthcare funding, which could have a material adverse effect on
our business and financial operations. Any reduction in
reimbursement from Medicare or other government programs may result
in a similar reduction in payments from private payors. The
implementation of cost containment measures or other healthcare
reforms may cause our suppliers to sustain loses or make them
unable to provide new machines or products in a timely
manner.
Specific Risks Related to our Intellectual Property
If our anticipated patents and other intellectual property rights
do not adequately protect our products, we may lose market share to
competitors and be unable to operate our business
profitably.
Our
success depends significantly on our ability to protect our
proprietary rights to the technologies used in our products. We
rely on the formulations which we have acquired, as well as a
combination of copyright, trade secret and trademark laws, and
nondisclosure, confidentiality and other contractual restrictions
to protect our proprietary technology. However, these legal means
afford only limited protection and may not adequately protect our
rights or permit us to gain or keep any competitive advantage. For
example, any future pending applications may be unsuccessful. The
U.S. Patent and Trademark Office may deny or require significant
narrowing of claims in our pending patent applications or future
patent applications, and patents issued as a result of these patent
applications, if any, may not provide us with significant
commercial protection or be issued in a form that is advantageous
to us. We could also incur substantial costs in proceedings before
the U.S. Patent and Trademark Office. These proceedings could
result in adverse decisions as to the priority of our inventions
and the narrowing or invalidation of claims in our patents. Third
parties may successfully challenge our issued patents and those
that may be issued in the future, which would render these patents
invalidated or unenforceable, and which could limit our ability to
stop competitors from marketing and selling related products. In
addition, our pending patent applications include claims to aspects
of products and procedures that are not currently protected by
issued patents, and third parties may successfully patent those
aspects before us or otherwise challenge our rights to these
aspects.
32
In the
event a competitor infringes upon one of our patents or other
intellectual property rights, enforcing those patents and rights
may be difficult and time consuming. Even if successful, litigation
to defend our patents against challenges or to enforce our
intellectual property rights could be expensive and time consuming
and could divert management’s attention from managing our
business. Moreover, we may not have sufficient resources to defend
our patents against challenges or to enforce our intellectual
property rights, any of which would adversely affect our ability to
compete and our business operations as a result.
If our trademarks or trade names are not adequately protected, then
we may be unable to build name recognition in our markets of
interest and our business may be adversely affected.
Our
registered or unregistered trademarks or trade names may be
challenged, infringed, circumvented or declared generic or
determined to infringe other marks. We may be unable to protect our
rights to these trademarks and trade names, which we need to build
name recognition by potential partners or customers in markets of
interest. If our trademarks are challenged, infringed upon,
circumvented, or declared generic or infringing, or if we are
unable to establish name recognition based on our trademarks and
trade names, then we may be unable to compete effectively and our
business may be adversely affected.
Adverse outcomes in litigation or similar proceedings could
adversely impact our business.
We may
in the future be, named as a party to litigation or other similar
legal proceedings. Adverse outcomes in any or all of these
proceedings could result in monetary damages or injunctive relief
that could adversely affect our ability to continue conducting our
business. While our management has verified there are no
proceedings currently pending against us such matters are subject
to inherent uncertainties and management’s view of these
matters may change in the future. If an unfavorable final outcome
in any such matter becomes probable and reasonably estimable, our
financial condition could be materially and adversely
affected.
Risks Related to this Offering and Ownership of our Common
Stock
There has been no public market for our common stock prior to this
offering, and an active trading market for our common stock may not
develop after this offering. As a result, you may be unable to
resell your common stock at or above the price paid under this
offering, or at all.
Prior
to this offering, there has been no public market for our common
stock, and an active trading market for our common stock may not
develop or be sustained after this offering. Also, the initial
public offering price for our common stock will be determined by
management and may bear no relationship to the market price for our
common stock after the offering. Furthermore, the market price of
our common stock may decline below the initial public offering
price. As a result of any of the foregoing, you may be unable to
resell your common stock at or above the price you paid under this
offering, or at all, and you may lose part or all of your
investment in our common stock.
33
We have never declared or paid cash dividends on our common stock
and do not anticipate paying dividends in the foreseeable future.
As a result, you must rely on price appreciation of our common
stock for a return on your investment in the foreseeable
future.
We have
never declared or paid cash dividends on our common stock. We
currently expect to retain our funds and future earnings to support
the operation, growth and development of our business. We do not
anticipate paying any cash dividends on our common stock in the
foreseeable future, except for those related to the corporate
conversion. As a result, a return on your investment in the near
future will occur only if our share price appreciates. Our share
price may not appreciate in value after this offering or maintain
the price at which you purchased our common stock pursuant to this
offering, and in either case, you would not realize a return on
investment or could lose all or part of your investment in our
common stock.
Furthermore, any
future determination to declare cash dividends will be made at the
discretion of our board of directors and will be subject to
compliance with applicable laws and covenants under any future
credit facilities, which may restrict or limit our ability to pay
dividends.
The price of our common stock may be volatile, and you could lose
all or part of your investment.
Stock
markets have experienced extreme volatility that has often been
unrelated to the operating performance of particular companies.
These broad market fluctuations may adversely affect the trading
price of our common stock. In addition, limited trading volume of
our stock may contribute to its future volatility. Price declines
in our common stock could result from general market and economic
conditions, some of which are beyond our control, and a variety of
other factors, including any of the risk factors described in this
prospectus. These broad market and industry factors may harm the
market price of our common stock, regardless of our operating
performance, and could cause you to lose all or part of your
investment in our common stock since you might be unable to sell
your shares at or above the price you paid in this offering.
Factors that could cause fluctuations in the market price of our
common stock include the following:
●
price
and volume fluctuations in the overall stock market from time to
time;
●
volatility in the
market prices and trading volumes of medical device company
stocks;
●
changes in
operating performance and stock market valuations of other medical
device companies generally, or those in our industry in
particular;
●
sales
of shares of our common stock by us or our
stockholders;
34
●
failure of
securities analysts to initiate or maintain coverage of us, changes
in financial estimates by securities analysts who follow our
company, or our failure to meet these estimates or the expectations
of investors;
●
the
financial projections we may provide to the public, any changes in
those projections or our failure to meet those
projections;
●
rumors
and market speculation involving us or other companies in our
industry;
●
actual
or anticipated changes in our results of operations or fluctuations
in our results of operations;
●
actual
or anticipated developments in our business, our competitors’
businesses or the competitive landscape generally;
●
litigation
involving us, our industry or both, or investigations by regulators
into our operations or those of our competitors;
●
developments or
disputes concerning our intellectual property or other proprietary
rights;
●
announced or
completed acquisitions of businesses or technologies by us or our
competitors;
●
new
laws or regulations or new interpretations of existing laws or
regulations applicable to our business;
●
changes in
accounting standards, policies, guidelines, interpretations or
principles;
●
any
significant change in our management; and
●
general economic
conditions and slow or negative growth of our markets.
35
In
addition, in the past, following periods of volatility in the
overall market and the market price of a particular company’s
securities, securities class action litigation has often been
instituted against these companies. This litigation, if instituted
against us, could result in substantial costs and a diversion of
our management’s attention and resources.
Substantial future sales of our common stock in the public market,
or the perception that these sales may occur, could cause the price
of our common stock to decline, even if our business is doing
well.
Sales
of our common stock in the public market after this offering, or
the perception that these sales may occur, could cause the market
price of our common stock to decline, even if our business is doing
well. All common stock sold in this offering, other than shares
acquired by our affiliates, will be freely transferable without
restriction or additional registration under the Securities Act of
1933. All of the remaining common stock outstanding after this
offering will be available for sale upon the expiration of the
360-day lock-up period pursuant to Rule 144 and Rule 701 under the
Securities Act. See “Shares eligible for future sale”
and “Underwriting” for a detailed description of the
lock-up and Securities Act of 1933 restrictions. Any or all of our
common stock may be released prior to expiration of the lock-up
period at the discretion of the underwriter. To the extent this
common stock is released before the expiration of the lock-up
period and sold into the market, the market price of our common
stock could decline.
Our executive officers, directors and principal stockholders may
exert control over us and may exercise influence over matters
subject to stockholder approval.
Our
executive officers and directors, and significant shareholders
together with their respective affiliates, beneficially owned
approximately 95.86% of our outstanding common stock as of December
31, 2016, and upon completion of this offering, that same group
will beneficially own approximately 61.36% of our outstanding
Common stock after all 2 million shares offered herein are sold.
Accordingly, these stockholders, if they act together, may exercise
substantial influence over matters requiring stockholder approval,
including the election of directors and approval of corporate
transactions, such as a merger. This concentration of ownership
could have the effect of delaying or preventing a change in control
or otherwise discourage a potential acquirer from attempting to
obtain control over us, which in turn could have a material adverse
effect on the market value of our common stock. For information
regarding the ownership of our common stock by our executive
officers and directors and their affiliates, please see the section
entitled “Security ownership of certain beneficial owners and
management.”
Our management will have broad discretion over the use and
investment of the net proceeds received in this offering and might
not apply the proceeds in ways that increase the value of your
investment in our common stock.
Our
management will have broad discretion over the use and investment
of the net proceeds received from this offering, and you will be
relying on, and may not agree with, the judgment of management
regarding the application of these net proceeds. Management intends
to use the net proceeds received from this offering as described in
the section entitled “Use of Proceeds.” The failure by
management to apply these funds effectively may result in financial
losses that could have a material adverse effect on our business
and cause the price of our common stock to decline. Management may
invest the net proceeds received from this offering in a manner
that does not produce income or increase value, which could have a
material adverse effect on our business and cause the price of our
common stock to decline.
36
If securities or industry analysts do not publish research or
publish unfavorable or inaccurate research about our business, our
common stock share price and trading volume could
decline.
The
trading market for our common stock will depend, in part, on the
research and reports that securities or industry analysts publish
about us or our business. We may be unable to attract or sustain
coverage by well-regarded securities and industry analysts. If
either none or only a limited number of securities or industry
analysts cover us or our business, or if these securities or
industry analysts are not widely respected within the general
investment community, the trading price for our common stock would
be materially and negatively impacted. In the event we obtain
securities or industry analyst coverage, if one or more of the
analysts who cover us or our business downgrade our common stock or
publish inaccurate or unfavorable research about us or our
business, the price of our common stock would likely decline. If
one or more of these analysts cease coverage of us or our business,
or fail to publish reports on us or our business regularly, demand
for our common stock could decrease, which might cause the price of
our common stock and trading volume to decline.
Public company requirements may strain our resources and divert
management’s attention, which could adversely impact our
ability to execute our strategy and harm operating
results.
As a
public company, we will be subject to the reporting requirements of
the Securities Exchange Act of 1934, the Sarbanes-Oxley Act, the
Dodd-Frank Act, the listing requirements of the NYSE MKT and other
applicable securities rules and regulations, although the company
plans to list on the OTCQB, it will adhere to the NYSE market
guidelines for future growth possibilities. Despite recent reforms
made possible by the Jumpstart Our Business Startups Act,
compliance with these rules and regulations will nonetheless
increase our legal and financial compliance costs, make some
activities more difficult, time-consuming or costly and increase
demand on our systems and resources, particularly after we are no
longer an “emerging growth company.” The Securities
Exchange Act of 1934 requires, among other things, that we file
annual, quarterly, and current reports with respect to our business
and operating results.
While
the members of our board of directors and our executive officers
have substantial experience relevant to our business and experience
with operations as a public company upon which you can base your
prediction of our future success or failure in complying with
public company requirements, the compliance costs and requirements
may stretch the management team’s ability to focus on current
business operations as opposed to a private company.
Furthermore, as a
result of disclosure of information in this prospectus and in
filings required of a public company, our business and financial
condition will become more visible, which may result in threatened
or actual litigation, including by competitors and other third
parties. If these claims are successful, our business and operating
results could be harmed, and even if the claims do not result in
litigation or are resolved in our favor, these claims, and the time
and resources necessary to resolve them, could divert the resources
of management and adversely affect our business, brand and
reputation and results of operations.
37
Our new
public company status and these new rules and regulations will make
it more expensive for us to obtain director and officer liability
insurance, and we may be required to accept reduced coverage or
incur substantially higher costs to obtain coverage. These factors
could also make it more difficult for us to attract and retain
qualified members of the board of directors, particularly to serve
on the audit committee and compensation committee, and qualified
executive officers.
Our certificate of incorporation, our bylaws and Nevada law contain
provisions that could discourage another company from acquiring us
and may prevent attempts by our stockholders to replace or remove
our current management.
Provisions of
Nevada law (where we are incorporated), our certificate of
incorporation and bylaws may discourage, delay or prevent a merger
or acquisition that stockholders may consider favorable, including
transactions in which you might otherwise receive a premium for
your stock. In addition, these provisions may frustrate or prevent
any attempts by our stockholders to replace or remove our current
management by making it more difficult for stockholders to replace
or remove our board of directors.
If we fail to maintain proper and effective internal controls, our
ability to produce accurate and timely financial statements could
be impaired and investors’ views of us or our business could
be harmed, resulting in the decrease in value of our common
stock.
As a
public company, we will be required to maintain internal control
over financial reporting and to report any material weaknesses in
our internal controls. In addition, beginning with our annual
report on Form 10-K for our fiscal year ending December 31, 2017 to
be filed in 2018, we will be required to furnish a report by
management on the effectiveness of our internal control over
financial reporting pursuant to Section 404 of the Sarbanes-Oxley
Act. We are in the process of designing, implementing and testing
the internal control over financial reporting required to comply
with this obligation, which process is time-consuming, costly and
complicated. In addition, our independent registered public
accounting firm will be required to attest to the effectiveness of
our internal control over financial reporting beginning with our
annual report on Form 10-K following the date on which we are no
longer an emerging growth company, which may be up to five full
years following the date of this offering. Our compliance with
Section 404 of the Sarbanes-Oxley Act will require us to incur
substantial accounting expense and expend significant management
efforts. If we are unable to comply with the requirements of
Section 404 in a timely manner, or we or our independent registered
public accounting firm identify deficiencies in our internal
control over financial reporting that are deemed to be material
weaknesses, the market price of our common stock could decline and
we could be subject to sanctions.
38
Our
ability to implement our business plan successfully and comply with
Section 404 requires us to be able to prepare timely and accurate
financial statements. We expect that we will need to continue to
improve existing, and implement new, operational and financial
systems, procedures and controls to manage our business
effectively. Any delay in the implementation of, or disruption in
the transition to, new or enhanced systems, procedures or controls,
may cause our operations to suffer and we may be unable to conclude
that our internal control over financial reporting is effective and
to obtain an unqualified report on internal controls from our
auditors when required under Section 404 of the Sarbanes-Oxley Act.
Moreover, we may not implement and maintain adequate controls over
our financial processes and reporting in the future. Even if we
were to conclude, and, when required, our auditors were to concur,
that our internal control over financial reporting provided
reasonable assurance regarding the reliability of financial
reporting and the preparation of financial statements for external
purposes in accordance with generally accepted accounting
principles, because of our inherent limitations, internal control
over financial reporting may not prevent or detect fraud or
misstatements or omissions.
FINRA (“Financial Industry Regulatory Authority”) sales
practice requirements may limit a stockholder’s ability to
buy and sell our stock
In
addition to the "penny stock" rules described below, FINRA has
adopted rules that require that in recommending an investment to a
customer, a broker-dealer must have reasonable grounds for
believing that the investment is suitable for that customer. Prior
to recommending speculative low priced securities to their
non-institutional customers, broker-dealers must make reasonable
efforts to obtain information about the customer's financial
status, tax status, investment objectives and other information.
Under interpretations of these rules, FINRA believes that there is
a high probability that speculative low priced securities will not
be suitable for at least some customers. FINRA requirements make it
more difficult for broker-dealers to recommend that their customers
buy our common stock, which may have the effect of reducing the
level of trading activity in our common stock. As a result, fewer
broker-dealers may be willing to make a market in our common stock,
reducing a stockholder's ability to resell shares of our common
stock.
Our common stock is a penny stock. Trading of our common stock may
be restricted by SEC penny stock regulations and FINRA sales
practice requirements, which may limit a stockholder's ability to
buy and sell our stock.
If and
until a liquid trading market does develop for our common stock, it
is likely we will be subject to the regulations applicable to
"Penny Stock." The regulations of the SEC promulgated under the
Exchange Act that require additional disclosure relating to the
market for penny stocks in connection with trades in any stock
defined as a penny stock. The SEC regulations define penny stocks
to be any non-NASDAQ equity security that has a market price of
less than $5.00 per share, subject to certain exceptions. Unless an
exception is available, those regulations require the broker-dealer
to deliver, prior to any transaction involving a penny stock, a
standardized risk disclosure schedule prepared by the SEC, to
provide the customer with current bid and offer quotations for the
penny stock, the compensation of the broker-dealer and its
salesperson in the transaction, monthly account statements showing
the market value of each penny stock held in the customer’s
account, to make a special written determination that the penny
stock is a suitable investment for the purchaser and receive the
purchaser's written agreement to the transaction. These disclosure
requirements may have the effect of reducing the level of trading
activity, if any, in the secondary market for a stock that becomes
subject to the penny stock rules. Consequently, these penny stock
rules may affect the ability of broker-dealers to trade our
securities. We believe that the penny stock rules discourage market
investor interest in and limit the marketability of our common
stock.
39
In
addition to the "penny stock" rules promulgated by the Securities
and Exchange Commission, FINRA has adopted rules that require that
in recommending an investment to a customer, a broker-dealer must
have reasonable grounds for believing that the investment is
suitable for that customer. Prior to recommending speculative low
priced securities to their non-institutional customers,
broker-dealers must make reasonable efforts to obtain information
about the customer's financial status, tax status, investment
objectives and other information. Under interpretations of these
rules, FINRA believes that there is a high probability that
speculative low priced securities will not be suitable for at least
some customers. FINRA requirements make it more difficult for
broker-dealers to recommend that their customers buy our common
stock, which may limit your ability to buy and sell our common
stock.
New
Regulation Could Affect Our Business Negatively
Our
business may become subject to additional future product
certification regulations, which may impair our ability to market
our products. We cannot assure you that our products will continue
to meet potentially changing standards. The failure to comply with
changing requirements could result in the recall of our products or
in civil or criminal penalties.
Shareholder
Mandates Would Alter the Company Status
In the
future our Articles of Incorporation and corporate structure could
be changed by the vote of a majority of our shareholders. This may
also impact the taxation of the company as well as the tax of any
dividends or stock payments authorized to shareholders of record.
Further, the company may become a target of a takeover and the
results of which may not be predictable and as beneficial as other
alternatives to our current shareholders.
Emerging Growth Company Status
We are
an “emerging growth company” as defined in the
Jumpstart Our Business Startups Act of 2012, or the JOBS Act, which
permits us to elect not to be subject to certain disclosure and
other requirements that otherwise would have been applicable to us
had we not been an “emerging growth company.” These
provisions include:
●
reduced disclosure
about our executive compensation arrangements;
●
no
non-binding advisory votes on executive compensation or golden
parachute arrangements; and
●
exemption from the
auditor attestation requirement in the assessment of our internal
control over financial reporting.
40
We may
take advantage of these exemptions for up to five years or such
earlier time as we are no longer an “Emerging Growth Company.” We will
qualify as an “Emerging
Growth Company” until the earliest of (1) the last day
of our fiscal year following the fifth anniversary of the date of
completion of this offering, (2) the last day of our fiscal year in
which we have annual gross revenue of $1.0 billion or more, (3) the
date on which we have, during the previous three-year period,
issued more than $1.0 billion in non-convertible debt and (4) the
last day of the fiscal year in which we become a “large
accelerated filer” as defined in Rule 12b-2 under the
Securities Exchange Act of 1934, as amended, or the Exchange
Act.
In
addition, the JOBS Act provides that an emerging growth company can
take advantage of an extended transition period for complying with
new or revised accounting standards. This allows an emerging growth
company to delay the adoption of certain accounting standards until
those standards would otherwise apply to private companies. We have
irrevocably elected not to avail ourselves of this exemption from
new or revised accounting standards and, therefore, we will be
subject to the same new or revised accounting standards as other
public companies that are not emerging growth
companies.
Proceeds Applied to General Corporate Purposes – Management
Discretion
The
proceeds of this Offering are for working capital and general
corporate purposes. Therefore, the application of the net proceeds
of this offering is substantially within the discretion of the
management. Investors will be relying on AII's management and
business judgment based solely on limited information. No assurance
can be given that the application of the net proceeds of this
Offering will result in AII achieving its financial and strategic
objectives.
41
USE OF PROCEEDS
We
estimate, based upon an assumed initial public offering price of
$2.00 per share, which is the price range per share estimate set
forth on the cover page of this prospectus, we will receive net
proceeds from this offering of approximately $3,600,000 (2,000,000
shares sold), after deducting estimated underwriting discounts and
commissions and estimated offering expenses payable by
us.
The
principal purposes of this offering are to obtain additional
capital to support our operations, create a public market for our
common stock and to facilitate future access to the public equity
markets. We currently expect to use the net proceeds of this
offering primarily to fund the commercialization and continued
development of the products we currently hold in the Nutraceuticals
markets and to acquire additional products that have been
researched. We have not yet determined which if any acquisitions
will be made. In addition, we will use the proceeds to build our
sales team within that market. Further, we plan on using the
proceeds to acquire commercial paper within our medical device
division. Below are some of the ways we may utilize the
funds:
●
Expansion of our
sales and marketing activities, including hiring new sales
representatives and a director of marketing within both
divisions;
●
Research and
development for new products and improvements to existing
products;
●
Potential
acquisition within our nutraceutical division; Potential
acquisition of commercial paper and new machines within our medical
device division;
●
The
remainder for working capital and other general corporate
purposes.
The
allocations set forth in the table below are the estimates of
management as to how the net proceeds of this Offering will
generally be allocated. There is no assurance that the estimates
set forth below will correspond with the actual expenditures of AII
during the next twelve months or thereafter, or that the results of
this Offering and the allocations set forth below will be
sufficient to maintain AII's operations following this
Offering.
|
USE OF PROCEEDS
|
|
|
Corporate
Development
|
792,000
|
|
Sales
and Marketing
|
900,000
|
|
Research
and Development
|
1,188,000
|
|
General
Working Capital
|
720,000
|
|
|
|
|
Total
Use of Proceeds
|
3,600,000
|
In
addition, we may also use a portion of our net proceeds to acquire
and invest in complementary products, technologies, services or
businesses. The Company is exploring the possibility of reducing
its reliance on third party manufacturers by bringing certain
manufacturing, service and research and development functions
in-house, which could include the acquisition of equipment and
other fixed assets or the acquisition or lease of a manufacturing
facility. However, we currently have no agreements or commitments
to complete any such transactions nor are we involved in
negotiations to do so.
Our
expected use of net proceeds from this offering represents our
current intentions based upon our plans and business condition. As
of the date of this prospectus, we cannot predict with certainty
all of the particular uses for the net proceeds to be received upon
the completion of this offering or the amounts that we will
actually spend on the uses set forth above. The amounts and timing
of our actual use of the net proceeds will vary depending on
numerous factors, including the factors described under the heading
“Risk Factors” in this prospectus. As a result,
management will have broad discretion in its application of the net
proceeds, and investors will be relying on our judgment in such
application.
42
Pending
use of the net proceeds from this offering, we may invest in short-
and intermediate-term interest-bearing obligations,
investment-grade instruments, certificates of deposit or direct or
guaranteed obligations of the U.S. government.
Net proceeds from this Offering
In the
offering contemplated herein, if all 2,000,000 shares are sold,
proceeds of $4,000,000 dollars will be raised. The costs identified
herein are estimated to be approximately Ten Percent (10%) of the
total offering, or $400,000, resulting in net proceeds to the
Company of approximately $3,600,000.
|
OFFERING AND USE OF PROCEEDS
|
|
|
|
|
|
|
Offering at $2.00 per
share
|
|
Total
Proceeds
|
4,000,000
|
|
|
|
|
Offering
Expenses
|
(400,000)
|
|
|
|
|
Net
Proceeds to the Company
|
3,600,000
|
|
|
|
43
Determination of Offering Price
We intend to register the Shares underlying the Class A
common stock shares offered herein and
register our company as a fully reporting issuer, however, we may
fail to do so.
After
we complete this filing we intend to hire transfer agents,
accountants, and consultants to assist us in the process of
registering the common stock and potentially offer warrants, with
the OTC marketplace. This process is time consuming, complicated
and expensive. We may not be able to secure effectiveness before
the Commission. If we do not successfully gain effectiveness for
this Registration Statement, the common stock may not become liquid
until such time as Rule 144 of the Securities Act is applicable. As
such, investors may find it difficult to liquidate their investment
or resell their securities. Additionally, if these shares are not
successfully registered, no public market would exist for the
shares.
Arbitrary Offering Price
The
offering price of $2.00 per Unit was arbitrarily determined by AII
and is unrelated to specific investment criteria, such as the
assets or past results of AII’s operations. In determining
the Offering price, AII considered such factors as the prospects,
if any, of similar companies, the previous experience of
management, AII's anticipated results of operations, and the
likelihood of acceptance of this Offering. Please review any
financial or other information contained in this offering with
qualified persons to determine its suitability as an investment
before purchasing any shares in this offering.
Best Efforts Offering
This
is a Best Efforts Offering. Arrestage is currently in the process
of engaging an underwriter. Underwriters are not required to sell
any specific number or dollar amount of securities but will use
their best efforts to sell the securities offered. Since no
arrangements with underwriter currently exist there are currently
no minimum purchase requirements.
The
information in this prospect is not complete and may be changed. We
may not sell these securities until the registration statement
filed with securities and Exchange Commission is effective. These
prospects are not an offer to sell these securities and it is not
soliciting an offer to buy these securities in any state where the
offer or sale is not permitted.
The
common stock outstanding prior to the offering is based on
3,600,000 shares outstanding as of December 31, 2016, and excludes
the following as of such date:
44
Capitalization
The
following table sets forth our cash and cash equivalents and our
capitalization as of September 30, 2017 and December
31, 2016:
●
on an actual basis;
and
●
on a pro forma as
adjusted basis to additionally give effect to issuance of 2,000,000
shares of our common stock in this offering at an assumed initial
public offering price of $2.00 per share (the offering price range
per share estimate set forth on the cover of this prospectus after
deducting estimated underwriting discounts and commissions and
estimated offering expenses payable by us.
You
should read the following information together with the information
contained under the “Management’s Discussion and
Analysis of Financial Condition and Results of Operations”
and our financial statements and the accompanying notes appearing
elsewhere in this prospectus.
Summary Balance Sheets
|
|
As of
September 30, 2017 (unaudited)
|
As of
December 31, 2016
|
||
|
|
Actual
|
Pro
Forma- 2,000,000 shares
|
Actual
|
Pro
Forma- 2,000,000 shares
|
|
Balance sheet data:
|
|
|
|
|
|
Cash
and cash equivalents
|
1,199
|
3,601,199
|
6,568
|
3,606,568
|
|
Working capital (deficit) (2)
|
-62,630
|
3,537,370
|
-41,880
|
3,558,120
|
|
Total
assets
|
13,199
|
3,613,199
|
15,068
|
3,615,068
|
|
Total
liabilities
|
63,828
|
63,828
|
48,448
|
48,448
|
|
Stockholders
(deficit)
|
|
|
|
|
|
Common
stock, $0.001 par value – 30,000,000 shares
authorized, 3,600,000 shares issued and outstanding at December 31,
2016.
|
3,600
|
5,600
|
3,600
|
5,600
|
|
Preferred Stock, $0.001 par value, 5,000,000
shares of preferred shares authorized, no shares of preferred stock
issued and outstanding
|
0
|
0
|
0
|
0
|
|
Additional
Paid In Capital
|
148,851
|
4,146,851
|
148,851
|
4,146,851
|
|
Accumulated
Deficit
|
-203,080
|
-603,080
|
-185,831
|
-585,831
|
|
Total
stockholders’ (deficit)
|
-33,380
|
3,543,771
|
-33,380
|
3,566,620
|
|
Total
stockholders’ (deficit) and Liabilities
|
13,199
|
3,615,068
|
15,068
|
3,615,068
|
45
The
table set forth above is based on 3,600,000 shares of our common
stock outstanding as of September 30, 2017 and
December 31, 2016.
Each
$1.00 increase (decrease) in the assumed initial public offering
price of $2.00 per share, which is the estimated price set forth on
the cover of this prospectus, would increase (decrease) each of
cash and cash equivalents, working capital, total assets and total
stockholders’ (deficit) equity by approximately $1,800,000
assuming the shares offered by us are sold as set forth on the
cover of this prospectus, remains the same, and after deducting
underwriting discounts and commissions and estimated offering
expenses payable by us.
We may
also increase or decrease the number of shares we are offering.
Each increase (decrease) of 1.0 million shares in the number of
shares offered by us would increase (decrease) each of cash and
cash equivalents, working capital, total assets and total
stockholders’ (deficit) equity by approximately $1.8 million,
assuming that the assumed initial public offering price remains the
same, and after deducting underwriting discounts and commissions
and estimated offering expenses payable by us. The pro forma as
adjusted information discussed above is illustrative only and will
adjust based on the actual initial public offering price and other
terms of this offering determined at pricing.
Items 6 and 7
Dilution
In many
cases, the present owners of each of the Company’s issued and
outstanding securities acquired such securities at a cost less than
that which the investors in this offering will pay. Upon the sale
of the shares offered hereby, the investors in this offering will
experience an immediate and substantial dilution. Therefore, the
investors in this offering will bear a substantial portion of the
risk of loss. Additionally, sales of securities of the Company or
use of stock for payment in the future could result in further
dilution.
If
you invest in our common stock, your ownership interest will be
diluted to the extent of the difference between the initial public
offering price in this offering per share of our common stock and
the pro forma as adjusted net tangible book value per share of our
common stock after the consummation of this offering. Net tangible
book value per share represents the book value of our total
tangible assets less the book value of our total liabilities
divided by the number of shares of common stock then issued and
outstanding. As of December 31, 2016, our net tangible book value
was $(41,880) or $(0.02) per share of common stock, which
represents the amount of our total tangible assets less total
liabilities, divided by 3,600,000 shares outstanding at December
31, 2016. As of September 30, 2017, our net tangible book
value was $(62,630) or $(0.02) per share of common stock, which
represents the amount of our total tangible assets less total
liabilities, divided by 3,600,000 shares outstanding at September
30, 2017.
46
After
giving effect to our sale of common stock in this offering at the
initial public offering price of $2.00 per share (the midpoint of
the price range set forth on the cover page of this prospectus),
and after deducting estimated underwriting discounts and
commissions and estimated offering expenses payable by us, our pro
forma as adjusted net tangible book value as of December 31, 2016
would have been $3,558,120, or $0.64 per share based 5,600,000
shares of common stock issued and outstanding after the Offering.
This represents an immediate and substantial dilution of $1.36 per
share, or 68.23%, to new investors purchasing common stock in this
offering. The following table illustrates this dilution per
share:
|
Net Tangible Book Value
|
||||
|
|
As
of September 30, 2017 (unaudited)
|
Pro
Forma September 30, 2017 (assuming 2,000,000 shares
sold)
|
As
of December 31, 2016
|
Pro
Forma December 31, 2016 (assuming 2,000,000 shares
sold)
|
|
Tangible
Assets
|
1,199
|
3,606,568
|
6,568
|
3,606,568
|
|
Liabilities
|
63,828
|
48,448
|
48,448
|
48,448
|
|
Net
Tangible Book Value
|
(62,630)
|
3,558,120
|
(41,880)
|
3,558,120
|
|
Shares
Outstanding
|
3,600,000
|
5,600,000
|
3,600,000
|
5,600,000
|
|
Per
Share value
|
(0.02)
|
$ 0.64
|
(0.01)
|
$0.64
|
|
Offering
Price
|
|
$ 2.00
|
|
$2.00
|
|
Immediate
Dilution
|
|
$ 1.36
|
|
$1.36
|
|
Dilution
as a Percentage
|
|
68.23%
|
|
68.23%
|
Each
$1.00 increase (decrease) in the assumed initial public offering
price of $2.00 per share, which is the estimated price set forth on
the cover of this prospectus, would increase (decrease) each of
cash and cash equivalents, working capital, total assets and total
stockholders’ (deficit) equity by approximately $1,800,000
assuming the shares offered by us are sold as set forth on the
cover of this prospectus, remains the same, and after deducting
underwriting discounts and commissions and estimated offering
expenses payable by us.
We may
also increase or decrease the number of shares we are offering.
Each increase (decrease) of 1.0 million shares in the number of
shares offered by us would increase (decrease) each of cash and
cash equivalents, working capital, total assets and total
stockholders’ (deficit) equity by approximately $1.8 million,
assuming that the assumed initial public offering price remains the
same, and after deducting underwriting discounts and commissions
and estimated offering expenses payable by us. The pro forma as
adjusted information discussed above is illustrative only and will
adjust based on the actual initial public offering price and other
terms of this offering determined at pricing.
The
following table summarizes, on a pro forma as adjusted basis as of
December 31, 2016, the differences between the number of shares of
common stock purchased from us, the total price and the average
price per share paid by existing stockholders and by the new
investors in this offering, before deducting estimated underwriting
discounts and commissions and estimated offering expenses payable
by us, at an assumed initial public offering price of $2.00 per
share (the midpoint of the price range set forth on the cover page
of this prospectus).
|
|
Shares Purchased
|
Total Consideration
|
|
||
|
|
Number
|
As a %
|
Amount
|
as a %
|
Avg. Price per share
|
|
Existing
Shareholders
|
3,600,000
|
64.29%
|
152,451
|
3.59%
|
$0.041
|
|
New
Investors in this Offering
|
2,000,000
|
35.71%
|
4,000,000
|
96.41%
|
$2.000
|
|
Total
|
5,600,000
|
100.00%
|
4,148,750
|
100.00%
|
$0.741
|
The
discussion and tables above are based on 3,600,000 shares of our
common stock outstanding as of December 31, 2016
47
PRINCIPAL STOCKHOLDERS
The
following table sets forth information as of December 31, 2016
regarding the beneficial ownership of our common stock, giving
effect to the Offering contemplated herein:
●
each
person or group known by us to beneficially own our outstanding
shares of common stock;
●
each
of our named executive officers; with special notation to anyone
owning over 5% of our common stock;
●
each
of our directors; and
●
all of
our current executive officers and directors as a
group.
Beneficial
ownership for the purposes of the following table is determined in
accordance with the rules and regulations of the SEC. These rules
generally provide that a person is the beneficial owner of
securities if such person has or shares the power to vote or direct
the voting of securities, or to dispose or direct the disposition
of securities or has the right to acquire such powers within 60
days. For purposes of calculating each person’s percentage
ownership, common stock issuable pursuant to options exercisable
within 60 days are included as outstanding and beneficially owned
for that person or group, but are not deemed outstanding for the
purposes of computing the percentage ownership of any other person.
Except as disclosed in the footnotes to this table and subject to
applicable community property laws, we believe that each beneficial
owner identified in the table possesses sole voting and investment
power over all common stock shown as beneficially owned by the
beneficial owner.
The
percentage of beneficial ownership is based on 3,600,000 shares of
common stock outstanding prior to this offering after giving effect
to our private placement offering, shares of common stock to be
outstanding after the completion of this offering, assuming no
exercise of the underwriters’ option to purchase additional
shares of our common stock and shares of common stock to be
outstanding after the completion of this offering, assuming
exercise of the underwriters’ option to purchase additional
shares of our common stock in full.
The
address for each beneficial owner is c/o Arrestage International,
Inc., Arrestage International, Inc.,
20343 N. Hayden Road, Suite 101, Scottsdale, Arizona 85255,
(480) 710.2229.
48
Principal Shareholders
|
|
Prior to Offering
|
After this offering (1)
|
||
|
|
Number
|
|
Number
|
|
|
Name
|
of shares
|
AS a %
|
of shares
|
AS a %
|
|
5% Shareholders
|
|
|
|
|
|
The Shapiro Trust (2)(5)
|
1,443,000
|
40.08%
|
1,443,000
|
25.77%
|
|
Crestrock Advisors (3)(6)
|
1,143,000
|
31.75%
|
1,143,000
|
20.41%
|
|
The Demitra Trust (4)
|
300,000
|
8.33%
|
300,000
|
5.36%
|
|
Plum Mountain Trust (5)
|
300,000
|
8.33%
|
300,000
|
5.36%
|
|
Total 5% Shareholders
|
3,186,000
|
88.50%
|
3,186,000
|
56.89%
|
|
Officers and Directors
|
|
|
|
|
|
Gary Croft
|
100,000
|
2.78%
|
100,000
|
1.79%
|
|
Greg Washington
|
50,000
|
1.39%
|
50,000
|
0.89%
|
|
Kimberly Shapiro
|
50,000
|
1.39%
|
50,000
|
0.89%
|
|
Phillip Weisman
|
50,000
|
1.39%
|
50,000
|
0.89%
|
|
Total Officers and Directors
|
250,000
|
6.94%
|
250,000
|
4.46%
|
|
Total for 5% Shareholders and Officers and Directors
|
3,436,000
|
95.44%
|
3,436,000
|
61.36%
|
|
All Other Shareholders
|
164,000
|
4.56%
|
2,164,000
|
38.64%
|
|
Total Shareholders
|
3,600,000
|
100.00%
|
5,600,000
|
100.00%
|
(1)
after the Offering contemplated herein
(2)
Roy Shapiro, as director, is also a beneficiary of The Shapiro
Trust
(3)
Directors Roy Shapiro and Phillip Nuciola, are principals of
CrestRock Advisors
(4)
Denise Weisman, daughter of Director Phillip Weisman,
is the beneficiary of The Demetra
Trust.
(5)
Phillip
Nucoila, President of Capital Markets, is the beneficiary of the
Plum Mountain Trust.
49
Item 8. Plan of Distribution/ Underwriters
PLAN OF DISTRIBUTION
Arestage
plans on utilizing the OTCQB market to list shares to the public.
No exchange traded calls, puts, or warrants will be available at
the outset of the offering.
Arrestage
has not been previously required to file reports pursuant to
Section 13(a) or 15(d) of the Exchange Act. The prospects
accompanying these securities defined and highlight all material
risks relating to its issue.
Securities
may be offered through the selling best efforts of
brokers or dealers, however, the company has no current plan of
distribution or any such agreement currently in place. Thereby, the
brokers or dealers participating in the offering, (if any), are
currently unknown. Arrestage
will sell shares for the duration of this offering at a fixed
price.
No
commissions or finder’s fees of any kind have been paid to
any person or entity, and although no such arrangement is expected,
any such future arrangement will be disclosed by
amendment.
Rather,
Arrestage plans to offer securities to the public on the OTCQB
exchange marketplace, and utilize passive market making
transactions as permitted by Rule 103, of Regulation M. Such
passive market making will express the direction of the
company’s basic plan for grants, highlight basic goals
attained, and explain the growth potential within the Sector in
general. Arrestage will create share demand by generally expressing
its corporate narrative and promised Board members involvement in
its personal network.
As and
if Arrestage determines it to be in the best long term interests of
the Company and its shareholders to engage Brokers or Dealers, such
information will be immediately updated via amendment as
required.
Item 9.
Description of Securities to be Registered.
50
NRS 78.138 Directors and officers: Exercise of
powers; performance of duties; presumptions and considerations;
liability to corporation and stockholders.
1. Directors
and officers shall exercise their powers in good faith and with a
view to the interests of the corporation.
2. In
performing their respective duties, directors and officers are
entitled to rely on information, opinions, reports, books of
account or statements, including financial statements and other
financial data, that are prepared or presented by:
(a) One or more directors, officers
or employees of the corporation reasonably believed to be reliable
and competent in the matters prepared or presented;
(b) Counsel, public accountants,
financial advisers, valuation advisers, investment bankers or other
persons as to matters reasonably believed to be within the
preparer’s or presenter’s professional or expert
competence; or
(c) A committee on which the director or
officer relying thereon does not serve, established in accordance
with NRS
78.125, as to matters within
the committee’s designated authority and matters on which the
committee is reasonably believed to merit confidence, but a
director or officer is not entitled to rely on such information,
opinions, reports, books of account or statements if the director
or officer has knowledge concerning the matter in question that
would cause reliance thereon to be unwarranted.
3. Directors
and officers, in deciding upon matters of business, are presumed to
act in good faith, on an informed basis and with a view to the
interests of the corporation.
4. Directors
and officers, in exercising their respective powers with a view to
the interests of the corporation, may consider:
(a) The interests of the
corporation’s employees, suppliers, creditors and
customers;
(b) The economy of the State
and Nation;
(c) The interests of the
community and of society; and
(d) The long-term as well as
short-term interests of the corporation and its stockholders,
including the possibility that these interests may be best served
by the continued independence of the corporation.
5. Directors
and officers are not required to consider the effect of a proposed
corporate action upon any particular group having an interest in
the corporation as a dominant factor.
6. The
provisions of subsections 4 and 5 do not create or authorize any
causes of action against the corporation or its directors or
officers.
51
7. Except
as otherwise provided in:
NRS 35.230, 90.660, 91.250, 452.200, 452.270, 668.045 and 694A.030,
or unless the articles of
incorporation or an amendment thereto, in each case filed on or
after October 1, 2003, provide for greater individual liability, a
director or officer is not individually liable to the corporation
or its stockholders or creditors for any damages as a result of any
act or failure to act in his or her capacity as a director or
officer unless it is proven that:
(a) The director’s or
officer’s act or failure to act constituted a breach of his
or her fiduciary duties as a director or officer; and
(b) The breach of those duties
involved intentional misconduct, fraud or a knowing violation of
law.
(Added to NRS
by 1991, 1184;
A 1993,
951; 1999,
1580; 2001,
3171; 2003,
3084)
Listing
We have
not applied to list our common stock on any exchange yet; however,
plan on registering shares on the OTCQB market place, during the
time our S-1 filing is taking place.
Transfer agent and registrar
The
transfer agent and registrar for our common stock will be V Stock
Transfer Agent, 18 Lafayette Place, Woodmere, NY
11598.
SHARES ELIGIBLE FOR FUTURE SALE
Based
upon the number of shares outstanding as of December 31, 2016 we
will have 5,600,000 shares of common stock outstanding upon the
closing of the offering contemplated herein. All the shares of our
common stock sold in this offering are freely tradable without
restriction or further registration under the Securities Act,
except for any such shares which may be held or acquired by our
“affiliates,” as that term is defined in Rule 144
promulgated under the Securities Act, which shares will be subject
to the volume limitations and other restrictions of Rule 144
described below. The remaining shares of common stock will be
“restricted securities,” as that term is defined in
Rule 144. These restricted securities will be eligible for public
sale only if they are registered under the Securities Act, or if
they qualify for an exemption from registration, for example, under
Rule 144.
52
Rule 144
In
general, under Rule 144 as in effect on the date of this
prospectus, a person who is not one of our affiliates at any time
during the three months preceding a sale, and who has beneficially
owned shares of our common stock for at least six months, would be
entitled to sell an unlimited number of shares of our common stock
provided current public information about us is available and,
after owning such shares for at least one year, would be entitled
to sell an unlimited number of shares of our common stock without
restriction. Our affiliates who have beneficially owned restricted
securities within the meaning of Rule 144 for at least six months
would be entitled to sell within any three-month period a number of
shares that does not exceed the greater of:
|
●
|
1% of
the number of shares of our common stock then outstanding;
or
|
|
●
|
the
average weekly trading volume of our common stock on the during the
four calendar weeks preceding the filing of a notice on Form 144
with respect to the sale.
|
Sales
under Rule 144 by our affiliates are also subject to manner of sale
provisions and notice requirements and to the availability of
current public information about us.
Rule 701
Rule
701 generally allows a member who acquired shares of our common
stock pursuant to a written compensatory plan or contract and who
is not deemed to have been an affiliate of our company during the
immediately preceding 90 days to sell these shares in reliance upon
Rule 144, but without being required to comply with the public
information, holding period, volume limitation or notice provisions
of Rule 144. Rule 701 also permits affiliates of our company to
sell their Rule 701 shares under Rule 144 without complying with
the holding period requirements of Rule 144. All holders of Rule
701 shares, however, are required by that rule to wait until 90
days after the date of this prospectus before selling those shares
pursuant to Rule 701.
Options
Following the date
of this prospectus, we may file one or more registration statements
on Form S-8 under the Securities Act to register the issuance of
additional shares of common stock under any stock plans approved in
the future. These registration statements will become effective
upon filing. All of the shares issued or to be issued upon the
exercise of stock options or settlement of other awards under our
stock plans are or will be eligible for resale in the public market
without restrictions, subject to Rule 144 limitations applicable to
affiliates and the lock-up agreements described below.
Lock-up agreements
Notwithstanding the foregoing, all of the holders of equity prior
to this offering are obligated, subject to limited exceptions, not
to dispose of or hedge any of their common stock or securities
convertible into or exchangeable for shares of common stock during
the 360-day lock-up period after the date of this prospectus
without the prior written consent of the Board and disclosure to
all shareholders in accord with this offering and shareholder
rights provisions of Nevada law.
53
Warrants
As of
the date of this Offering Document there are no outstanding
warrants or Stock Incentive Plan warrants outstanding nor
planned.
Options
As of
the date of this Offering Document there are no outstanding options
or Stock Incentive Plan options outstanding nor
planned.
Registration Rights
As of
the date of this Offering Document we have no registration rights
agreements pending or planned.
Anti-Takeover Provisions
Certificate of Incorporation and Amended and Restated
Bylaws
Because
our stockholders do not have cumulative voting rights, our
stockholders holding a majority of the voting power of our shares
of common stock outstanding will be able to elect all of our
directors. Our amended and restated certificate of incorporation
and amended and restated bylaws to be effective upon the closing of
this offering will provide that all stockholder actions must be
effected at a duly called meeting of stockholders and not by
consent in writing. A special meeting of stockholders may be called
only by a majority of our board of directors, the chair of our
board of directors, or our chief executive officer.
Further, our
amended and restated certificate of incorporation will provide
that, unless we consent in writing to the selection of an
alternative forum, the Court of Chancery of the State of Nevada
will be the sole and exclusive forum for: (i) any derivative
action or proceeding brought on behalf of us; (ii) any action
asserting a claim of breach of a fiduciary duty owed by any of our
directors, officers or other employees or agents to us or our
stockholders; (iii) any action asserting a claim against us
arising pursuant to any provision of the Nevada Corporation Law or
our amended and restated certificate of incorporation or amended
and restated bylaws; or (iv) any action asserting a claim
against us governed by the internal affairs doctrine. The
enforceability of similar choice of forum provisions in other
companies’ certificates of incorporation has been challenged
in legal proceedings, and it is possible that, in connection with
any action, a court could find the choice of forum provisions
contained in our amended and restated certificate of incorporation
to be inapplicable or unenforceable in such action.
54
The
foregoing provisions will make it more difficult for our existing
stockholders to replace our board of directors as well as for
another party to obtain control of our Company by replacing our
board of directors. Since our board of directors has the power to
retain and discharge our officers, these provisions could also make
it more difficult for existing stockholders or another party to
effect a change in management. In addition, the authorization of
undesignated preferred stock makes it possible for our board of
directors to issue preferred stock with voting or other rights or
preferences that could impede the success of any attempt to change
the control of our Company.
These
provisions are intended to enhance the likelihood of continued
stability in the composition of our board of directors and its
policies and to discourage certain types of transactions that may
involve an actual or threatened acquisition of our Company. These
provisions are also designed to reduce our vulnerability to an
unsolicited acquisition proposal and to discourage certain tactics
that may be used in proxy rights. However, these provisions could
have the effect of discouraging others from making tender offers
for our shares and may have the effect of deterring hostile
takeovers or delaying changes in control of our Company or our
management. As a consequence, these provisions also may inhibit
fluctuations in the market price of our stock that could result
from actual or rumored takeover attempts.
MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES TO NON-U.S.
HOLDERS
The
following discussion is a summary of the material U.S. federal
income tax consequences to Non-U.S. Holders (as defined below) of
the purchase, ownership and disposition of our common stock issued
pursuant to this offering, but does not purport to be a complete
analysis of all potential tax effects. The effects of other U.S.
federal tax laws, such as estate and gift tax laws, and any
applicable state, local or non-U.S. tax laws are not discussed.
This discussion is based on the U.S. Internal Revenue Code of 1986,
as amended, or the Code, Treasury Regulations promulgated
thereunder, judicial decisions, and published rulings and
administrative pronouncements of the U.S. Internal Revenue Service,
or the IRS, in each case in effect as of the date hereof. These
authorities may change or be subject to differing interpretations.
Any such change or differing interpretation may be applied
retroactively in a manner that could adversely affect a Non-U.S.
Holder of our common stock. We have not sought and will not seek
any rulings from the IRS regarding the matters discussed below.
There can be no assurance the IRS or a court will not take a
contrary position to that discussed below regarding the tax
consequences of the purchase, ownership and disposition of our
common stock.
This
discussion is limited to Non-U.S. Holders that hold our common
stock as a “capital asset” within the meaning of
Section 1221 of the Code (generally, property held for
investment). This discussion does not address all U.S. federal
income tax consequences relevant to a Non-U.S. Holder’s
particular circumstances, including the impact of the Medicare
contribution tax on net investment income. In addition, it does not
address consequences relevant to Non-U.S. Holders subject to
special rules, including, without limitation:
|
●
|
U.S.
expatriates and former citizens or long-term residents of the
United States;
|
|
●
|
persons
subject to the alternative minimum tax;
|
55
|
●
|
persons
holding our common stock as part of a hedge, straddle or other risk
reduction strategy or as part of a conversion transaction or other
integrated investment;
|
|
●
|
banks,
insurance companies, and other financial institutions;
|
|
●
|
brokers,
dealers or traders in securities;
|
|
●
|
“controlled
foreign corporations,” “passive foreign investment
companies,” and corporations that accumulate earnings to
avoid U.S. federal income tax;
|
|
●
|
partnerships
or other entities or arrangements treated as partnerships for U.S.
federal income tax purposes (and investors therein);
|
|
●
|
tax-exempt
organizations or governmental organizations;
|
|
●
|
persons
deemed to sell our common stock under the constructive sale
provisions of the Code;
|
|
●
|
persons
who hold or receive our common stock pursuant to the exercise of
any employee stock option or otherwise as compensation;
and
|
|
●
|
tax-qualified
retirement plans.
|
If an
entity treated as a partnership for U.S. federal income tax
purposes holds our common stock, the tax treatment of a partner in
the partnership will depend on the status of the partner, the
activities of the partnership and certain determinations made at
the partner level. Accordingly, partnerships holding our common
stock and the partners in such partnerships should consult their
tax advisors regarding the U.S. federal income tax consequences to
them.
THIS
DISCUSSION IS FOR INFORMATIONAL PURPOSES ONLY AND IS NOT TAX
ADVICE. INVESTORS SHOULD CONSULT THEIR TAX ADVISORS WITH RESPECT TO
THE APPLICATION OF THE U.S. FEDERAL INCOME TAX LAWS TO THEIR
PARTICULAR SITUATIONS AS WELL AS ANY TAX CONSEQUENCES OF THE
PURCHASE, OWNERSHIP AND DISPOSITION OF OUR COMMON STOCK ARISING
UNDER THE U.S. FEDERAL ESTATE OR GIFT TAX LAWS OR UNDER THE LAWS OF
ANY STATE, LOCAL OR NON-U.S. TAXING JURISDICTION OR UNDER ANY
APPLICABLE INCOME TAX TREATY.
Definition of a Non-U.S. Holder
For
purposes of this discussion, a “Non-U.S. Holder” is any
beneficial owner of our common stock that is neither a “U.S.
person” nor an entity treated as a partnership for U.S.
federal income tax purposes. A U.S. person is any person that, for
U.S. federal income tax purposes, is or is treated as any of the
following:
56
|
●
|
an
individual who is a citizen or resident of the United
States;
|
|
●
|
a
corporation created or organized under the laws of the United
States, any state thereof, or the District of
Columbia;
|
|
●
|
an
estate, the income of which is subject to U.S. federal income tax
regardless of its source; or
|
|
●
|
a trust
that (1) is subject to the primary supervision of a U.S. court
and the control of one or more “United States persons”
(within the meaning of Section 7701(a)(30) of the Code), or
(2) has a valid election in effect to be treated as a United
States person for U.S. federal income tax purposes.
|
Distributions
As
described in the section of this prospectus entitled
“Dividend Policy,” we do not anticipate declaring or
paying dividends to holders of our common stock in the foreseeable
future. However, if we do make distributions of cash or property on
our common stock, such distributions will constitute dividends for
U.S. federal income tax purposes to the extent paid from our
current or accumulated earnings and profits, as determined under
U.S. federal income tax principles. Amounts not treated as
dividends for U.S. federal income tax purposes will constitute a
return of capital and first be applied against and reduce a
Non-U.S. Holder’s adjusted tax basis in its common stock, but
not below zero. Any excess will be treated as capital gain and will
be treated as described below under “—Sale or Other
Taxable Disposition.”
Subject
to the discussion below on effectively connected income, dividends
paid to a Non-U.S. Holder of our common stock will be subject to
U.S. federal withholding tax at a rate of 30% of the gross amount
of the dividends (or such lower rate specified by an applicable
income tax treaty, provided the Non-U.S. Holder furnishes a valid
IRS Form W-8BEN or W-8BEN-E (or other applicable documentation)
certifying qualification for the lower treaty rate). If a Non-U.S.
Holder holds the stock through a financial institution or other
intermediary, the Non-U.S. Holder will be required to provide
appropriate documentation to the intermediary, which then will be
required to provide certification to the applicable withholding
agent, either directly or through other intermediaries. A Non-U.S.
Holder that does not timely furnish the required documentation, but
that qualifies for a reduced treaty rate, may obtain a refund of
any excess amounts withheld by timely filing an appropriate claim
for refund with the IRS. Non-U.S. Holders should consult their tax
advisors regarding their entitlement to benefits under any
applicable income tax treaty.
If
dividends paid to a Non-U.S. Holder are effectively connected with
the Non-U.S. Holder’s conduct of a trade or business within
the United States (and, if required by an applicable income tax
treaty, the Non-U.S. Holder maintains a permanent establishment in
the United States to which such dividends are attributable), the
Non-U.S. Holder will be exempt from the U.S. federal withholding
tax described above. To claim the exemption, the Non-U.S. Holder
must furnish to the applicable withholding agent a valid IRS Form
W-8ECI, certifying that the dividends are effectively connected
with the Non-U.S. Holder’s conduct of a trade or business
within the United States.
57
Any
such effectively connected dividends will be subject to U.S.
federal income tax on a net income basis at the regular graduated
rates. A Non-U.S. Holder that is a corporation also may be subject
to a branch profits tax at a rate of 30% (or such lower rate
specified by an applicable income tax treaty) on such effectively
connected dividends, as adjusted for certain items. Non-U.S.
Holders should consult their tax advisors regarding any applicable
tax treaties that may provide for different rules.
Sale or Other Taxable Disposition
A
Non-U.S. Holder will not be subject to U.S. federal income tax on
any gain realized upon the sale or other taxable disposition of our
common stock unless:
|
●
|
the
gain is effectively connected with the Non-U.S. Holder’s
conduct of a trade or business within the United States (and, if
required by an applicable income tax treaty, the Non-U.S. Holder
maintains a permanent establishment in the United States to which
such gain is attributable);
|
|
●
|
the
Non-U.S. Holder is a nonresident alien individual present in the
United States for 183 days or more during the taxable year of the
disposition and certain other requirements are met; or
|
|
●
|
our
common stock constitutes a U.S. real property interest, or USRPI,
by reason of our status as a U.S. real property holding
corporation, or USRPHC, for U.S. federal income tax
purposes.
|
Gain
described in the first bullet point above generally will be subject
to U.S. federal income tax on a net income basis at the regular
graduated rates. A Non-U.S. Holder that is a corporation also may
be subject to a branch profits tax at a rate of 30% (or such lower
rate specified by an applicable income tax treaty) on such
effectively connected gain, as adjusted for certain
items.
Gain
described in the second bullet point above will be subject to U.S.
federal income tax at a rate of 30% (or such lower rate specified
by an applicable income tax treaty), which may be offset by U.S.
source capital losses of the Non-U.S. Holder (even though the
individual is not considered a resident of the United States),
provided the Non-U.S. Holder has timely filed U.S. federal income
tax returns with respect to such losses.
58
With
respect to the third bullet point above, we believe we currently
are not, and do not anticipate becoming, a USRPHC. Because the
determination of whether we are a USRPHC depends, however, on the
fair market value of our USRPIs relative to the fair market value
of our non-U.S. real property interests and our other business
assets, there can be no assurance we currently are not a USRPHC or
will not become one in the future. Even if we are or were to become
a USRPHC, gain arising from the sale or other taxable disposition
by a Non-U.S. Holder of our common stock will not be subject to
U.S. federal income tax if our common stock is “regularly
traded,” as defined by applicable Treasury Regulations, on an
established securities market, and such Non-U.S. Holder owned,
actually or constructively, 5% or less of our common stock
throughout the shorter of the five-year period ending on the date
of the sale or other taxable disposition or the Non-U.S.
Holder’s holding period.
Non-U.S. Holders
should consult their tax advisors regarding potentially applicable
income tax treaties that may provide for different
rules.
Information Reporting and Backup Withholding
Payments of
dividends on our common stock will not be subject to backup
withholding, provided the applicable withholding agent does not
have actual knowledge or reason to know the holder is a United
States person and the holder either certifies its non-U.S. status,
such as by furnishing a valid IRS Form
W-8BEN, W-8BEN-E or W-8ECI, or otherwise establishes an
exemption. However, information returns are required to be filed
with the IRS in connection with any dividends on our common stock
paid to the Non-U.S. Holder, regardless of whether any tax was
actually withheld. In addition, proceeds of the sale or other
taxable disposition of our common stock within the United States or
conducted through certain U.S.-related brokers generally will not
be subject to backup withholding or information reporting, if the
applicable withholding agent receives the certification described
above and does not have actual knowledge or reason to know that
such holder is a United States person, or the holder otherwise
establishes an exemption. Proceeds of a disposition of our common
stock conducted through a non-U.S. office of a non-U.S. broker
generally will not be subject to backup withholding or information
reporting.
Copies
of information returns that are filed with the IRS may also be made
available under the provisions of an applicable treaty or agreement
to the tax authorities of the country in which the Non-U.S. Holder
resides or is established.
Backup
withholding is not an additional tax. Any amounts withheld under
the backup withholding rules may be allowed as a refund or a credit
against a Non-U.S. Holder’s U.S. federal income tax
liability, provided the required information is timely furnished to
the IRS.
59
Additional Withholding Tax on Payments Made to Foreign
Accounts
Withholding taxes
may be imposed under Sections 1471 to 1474 of the Code (such
Sections commonly referred to as the Foreign Account Tax Compliance
Act, or FATCA) on certain types of payments made to non-U.S.
financial institutions and certain other non-U.S. entities.
Specifically, a 30% withholding tax may be imposed on dividends on,
or gross proceeds from the sale or other disposition of, our common
stock paid to a “foreign financial institution” or a
“non-financial foreign entity” (each as defined in the
Code), unless (1) the foreign financial institution undertakes
certain diligence and reporting obligations, (2) the
non-financial foreign entity either certifies it does not have any
“substantial United States owners” (as defined in the
Code) or furnishes identifying information regarding each
substantial United States owner, or (3) the foreign financial
institution or non-financial foreign entity otherwise qualifies for
an exemption from these rules. Foreign financial institutions
located in jurisdictions that have an intergovernmental agreement
with the United States governing FATCA may be subject to different
rules.
Under
the applicable Treasury Regulations, withholding under FATCA
generally applies to payments of dividends on our common stock and
will apply to payments of gross proceeds from the sale or other
disposition of such stock on or after January 1,
2017.
Prospective
investors should consult their tax advisors regarding the potential
application of withholding under FATCA to their investment in our
common stock.
Item 10. Interest of Named Experts and Counsel
LEGAL MATTERS
The
validity of the issuance of our common stock offered in this
prospectus will be passed upon for us by the law office of Erica
Loren Jozwiak, Esq. in connection with this offering.
EXPERTS
The
audited financial statements in this prospectus and elsewhere in
the registration statement have been so included in reliance upon
the Audit report of Schumacher & Associates, Inc. CPA,
7931 S Broadway #314 Littleton, Colorado a member of the
Public Company Accounting Oversight Board (PCAOB), upon the
authority of said firm as experts in accounting and
auditing.

60
WHERE YOU CAN FIND MORE INFORMATION
We have
filed with the SEC a registration statement on Form S-1 under
the Securities Act with respect to the common stock offered hereby.
This prospectus, which constitutes a part of the registration
statement, does not contain all of the information set forth in the
registration statement or the exhibits and schedules filed
therewith. For further information with respect to Arrestage
International, Inc. and the shares of common stock offered hereby,
reference is made to the registration statement and the exhibits
and schedules filed therewith. Statements contained in this
prospectus regarding the contents of any contract or any other
document that is filed as an exhibit to the registration statement
are not necessarily complete, and each such statement is qualified
in all respects by reference to the full text of such contract or
other document filed as an exhibit to the registration statement. A
copy of the registration statement and the exhibits and schedules
filed therewith may be inspected without charge at the public
reference room maintained by the SEC, located at 100 F
Street N.E., Room 1580, Washington, D.C. 20549, and
copies of all or any part of the registration statement may be
obtained from such offices upon the payment of the fees prescribed
by the SEC. Please call the SEC at 1-800-SEC-0330 for further
information about the public reference room. The SEC also maintains
a website that contains reports, proxy and information statements
and other information regarding registrants that file
electronically with the SEC. The address is
www.sec.gov.
Corporate Information
Arrestage
International, Inc., a Nevada Corporation, was formed on June 15,
2011, to develop an international licensing program whereby we will
provide turnkey assistance with operations and support for
distributors in the Skin Care and Anti-Aging arena, using our
brand, custom formulas, trademark, Internet presence and
proprietary marketing protocols. In addition to this, we plan to
continue to develop these aspects of the business and potentially
acquire complimentary products and services. Arrestage
International intends to create a significant brand in the
aesthetic skincare marketplace. The officers and directors of AII
all have created, or participated in the marketing and development
of branded products in this space.
The
company intends to include a medical device rental program that
plans to be Rolled up in to Arrestage. Arrestage plans to utilize
this division purchase or secure medical devices such as MRI
systems and various other diagnostic equipment. We plan our target
market to be physician groups, clinics, rural hospitals, and
insurance companies. Arrestage intends to obtain residual income
from such rentals and would maintain a fully secured UCC-1 position
on each piece of equipment.
Our
principal executive offices are located at 20343 N. Hayden Road,
STE 101, Scottsdale, Arizona 85255 and our telephone number is
(480) 710.2229. Our website is located at www.arrestageinternational.com .
Our website, and the information on our website, is neither part of
this prospectus nor incorporated by reference herein.
You may
also request a copy of these filings, at no cost to you, by writing
us at the following address:
Arrestage International, Inc.
20343 N. Hayden Road, Suite 101
Scottsdale, Arizona 85255
61
Summary historical financial data
The
tables below summarize our financial information for the periods
indicated. We derived the financial information for the years ended
December 31, 2016 and 2015 from our audited financial statements
included elsewhere in this prospectus. You should read the
following information together with the more detailed information
contained in “Management’s Discussion and Analysis of
Financial Condition and Results of Operations” and our
financial statements and the accompanying notes. Our historical
results are not necessarily indicative of the results to be
expected in any future period.
Balance Sheets
|
|
September 30,
|
December 31,
|
December 31,
|
|
|
2017 (unaudited)
|
2016
|
2015
|
|
ASSETS
|
|||
|
|
|
|
|
|
Current
Assets
|
|
|
|
|
Cash
in Bank
|
$1,199
|
$6,568
|
$290
|
|
|
|
|
|
|
Total
Current Assets
|
1,199
|
6,568
|
290
|
|
|
|
|
|
|
Other
Assets
|
|
|
|
|
Prepaid
Expenses
|
12,000
|
8,500
|
-
|
|
Trademark
License
|
50,000
|
50,000
|
50,000
|
|
Impairment-Trademark
License
|
(50,000)
|
(50,000)
|
(50,000)
|
|
|
|
|
|
|
Total
Other Assets
|
12,000
|
8,500
|
-
|
|
|
|
|
|
|
Total
Assets
|
$13,199
|
$15,068
|
$290
|
|
|
|
|
|
|
LIABILITIES
AND STOCKHOLDERS' (DEFICIT)
|
|||
|
|
|
|
|
|
Current
Liabilities
|
|
|
|
|
|
|
|
|
|
Total
Current Liabilities
|
|
|
|
|
Note
Payable-Related Party
|
$32,331
|
$18,390
|
$12,830
|
|
Accrued
Interest Payable
|
5,707
|
4,268
|
3,006
|
|
Licensee
Fee Payable
|
25,790
|
25,790
|
25,790
|
|
Total
Current Liabilities
|
63,828
|
48,448
|
41,626
|
|
|
|
|
|
|
Total
Liabilities
|
63,828
|
48,448
|
41,626
|
|
|
|
|
|
|
Stockholders'
(Deficit)
|
|
|
|
|
Preferred
Stock, $0.001 par value, 5,000,000 shares of preferred shares
authorized, no shares of preferred stock issued and
outstanding
|
-
|
-
|
-
|
|
Common
Stock, $0.001 par value, 30,000,000 shares of common stock
authorized, 3,600,000 shares issued and outstanding as of September
30, 2017 and December 31, 2016, and 3,561,250 issued and
outstanding as of December 31, 2015.
|
3,600
|
3,600
|
3,561
|
|
Additional
Paid-in Capital
|
148,851
|
148,851
|
110,140
|
|
Accumulated
(Deficit)
|
(203,080)
|
(185,831)
|
(155,037)
|
|
|
|
|
|
|
Total
Stockholders' (Deficit)
|
(50,629)
|
(33,380)
|
(41,336)
|
|
|
|
|
|
|
Total
Liabilities & Stockholders' (Deficit)
|
$13,199
|
$15,068
|
$290
|
62
Statements of Operations
|
|
Year Ended
December 31, 2016
|
Year Ended
December 31, 2015
|
|
|
|
|
|
Revenues
|
$-
|
$-
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Legal
and Professional Fees
|
26,000
|
-
|
|
Stock
Based Compensation
|
-
|
10,000
|
|
Accounting/Bookkeeping
|
2,500
|
-
|
|
Other
Expense
|
1,032
|
1,136
|
|
Total
Operating Expenses
|
29,532
|
11,136
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Interest
Expense
|
1,262
|
1,163
|
|
|
|
|
|
Total
Expenses
|
30,794
|
12,299
|
|
|
|
|
|
Net
(Loss)
|
$(30,794)
|
$(12,299)
|
63
|
|
Nine Months Ended
September 30, 2017
(unaudited)
|
Nine Months Ended
September 30, 2016 (unaudited)
|
|
|
|
|
|
Revenues
|
$-
|
$-
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Legal
and Professional Fees
|
14,138
|
20,000
|
|
Accounting/Bookkeeping
|
1,535
|
2,500
|
|
Other
Expenses
|
137
|
793
|
|
Total
Operating Expenses
|
15,810
|
23,293
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Interest
Expense
|
1,439
|
945
|
|
|
|
|
|
Total
Expenses
|
17,249
|
24,238
|
|
|
|
|
|
Net
(Loss)
|
$(17,249)
|
$(24,238)
|
|
|
|
|
|
Net Tangible Book Value
|
||
|
|
As of December 31, 2016
|
Pro Forma December 31, 2016 (assuming 2,000,000 shares
sold)
|
|
Tangible
Assets
|
6,568
|
3,606,568
|
|
Liabilities
|
48,448
|
48,448
|
|
Net
Tangible Book Value
|
(41,880)
|
3,558,120
|
|
Shares
Outstanding
|
3,600,000
|
5,600,000
|
|
Per
Share value
|
(0.01)
|
$0.64
|
|
Offering
Price
|
|
$2.00
|
|
Immediate
Dilution
|
|
$1.36
|
|
Dilution
as a Percentage
|
|
68.23%
|
64
Each
$1.00 increase (decrease) in the assumed initial public offering
price of $2.00 per share, which is the estimated price set forth on
the cover of this prospectus, would increase (decrease) each of
cash and cash equivalents, working capital, total assets and total
stockholders’ (deficit) equity by approximately $1,800,000
assuming the shares offered by us are sold as set forth on the
cover of this prospectus, remains the same, and after deducting
underwriting discounts and commissions and estimated offering
expenses payable by us.
We may
also increase or decrease the number of shares we are offering.
Each increase (decrease) of 1.0 million shares in the number of
shares offered by us would increase (decrease) each of cash and
cash equivalents, working capital, total assets and total
stockholders’ (deficit) equity by approximately $1.8 million,
assuming that the assumed initial public offering price remains the
same, and after deducting underwriting discounts and commissions
and estimated offering expenses payable by us. The pro forma as
adjusted information discussed above is illustrative only and will
adjust based on the actual initial public offering price and other
terms of this offering determined at pricing.
|
(1)
|
Pro
forma and pro forma as adjusted information discussed above are
unaudited and illustrative only.
|
|
(2)
|
Represents
current assets less current liabilities.
|
DIVIDEND POLICY
AII has
not declared or paid any cash dividends on its capital stock since
inception. AII intends to retain any future earnings to finance the
operation and expansion of its business and does not anticipate
paying any cash dividends in the foreseeable future. As a result,
stockholders will need to sell the Shares purchased in this
Offering in order to realize a return on their investment, if
any.
See
“Risk Factors — We do not expect to pay any
dividends on our common stock for the foreseeable future.”
Any determination to pay dividends in the future will be at the
discretion of our Board of Directors and will depend upon, among
other factors, our results of operations, financial condition,
capital requirements and covenants in our existing financing
arrangements and any future financing arrangements.
65
Items 11and 12
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis of our financial condition
and results of operations should be read in conjunction with our
financial statements and the accompanying notes and other financial
information appearing elsewhere in this prospectus. In addition to
historical information, this discussion contains forward-looking
statements that involve risks, uncertainties and assumptions that
could cause actual results to differ materially from
management’s expectations. Factors that could cause such
differences include, but are not limited to, those identified below
and those described under the heading “Risk Factors”
appearing elsewhere in this prospectus.
Overview
We are
a Company within the healthcare space. Specifically, we are in the
Nutraceutical business that plans to distribute and holds
formulations on skin care products as well as brand formulas, and
other intellectual property. In addition, we plan to have a
division that is a finance company that purchases and securitizes
medical devises for periodic rental to end users. We plan to
maintain a UCC-1 on the machines we purchase and provide full
service plans and warranty options.
ARRESTAGE INTERNATIONAL, INC.
(“AII” or the “Company”) a Nevada
corporation, plans to develop an international licensing program
whereby we plan to provide turnkey assistance with operations and
support for distributors in the Skin Care and Anti-Aging arena,
using our brand, custom formulas, trademark, internet presence and
proprietary marketing protocols. In addition to this, we plan to
continue to develop these aspects of the business and potentially
acquire complimentary products and services. Arrestage
International intends to create a significant brand in the
aesthetic skincare marketplace. The officers and directors of AII
all have created, or participated in the marketing and development
of branded products in this space. AII plans to generate a
licensing fee of 5-8% (or a higher rate the market may bear) of all
revenues generated by future licensees.
AII
plans to license full use of all intellectual property and formulas
it currently possesses in all geographical regions and channels of
distribution. AII plans to set itself apart from its competitors by
providing clinically based sales materials that attract a more
sophisticated consumer which may enable the licensee to increase
their sales revenues with higher margins. Licensees will have
access to the luminary panel, clinical and marketing training
protocols, and collateral materials. Arrestage International plans
to address an ever-increasing world demand for western culturally
influenced aesthetic skin topical products, in addition to the
increasing demand in US domestic and other western markets. AII
seeks to license distribution organizations, in the US and
Internationally, to market its proprietary brand of aesthetic
dermal formulations and additional products that it may potentially
acquire.
66
The
initial products have gone through clinical and behavioral testing
and several reformulations as they strive to achieve a high
acceptance status. This was obtained through market research and in
house due diligence. The size of the aesthetic topical world market
is approximately $7.4 billion. (KPMG market report 2016) Morningstar
estimates a continued growth rate of 11% CAGR from 2016-2020. Due
to the high margins, the major aesthetic device makers are
attempting to enter this market as distributors. This may be a
beneficial circumstance as it may enhance the valuation of AII and
widen future exit possibilities.
Medical Device Rental Division
The
Company plans to establish a medical device rental program that
purchases or secures medical devices such as MRI systems and
various other diagnostic equipment. Our target market will be
physician groups, clinics, rural hospitals, and insurance
companies. Arrestage intends to obtain residual income from such
rentals and would maintain a fully secured UCC-1 position on each
piece of equipment. To grow this division, Arrestage plans to
aggressively market such services in this coming year. This
function will help meet the expanding demand of medical systems due
to the current healthcare trends. The United States market, the
largest in the world, is expected to reach a value of $133 billion
by 2016. Based on performance of the rental program sought to be
rolled-into Arrestage, The Company anticipates a strong market
presence within this lucrative sector of secured structured medical
lending.
Acquisition of a Licensing Agreement
Effective
June 22, 2011 Arrestage entered into an agreement to acquire
an exclusive licensing agreement from Ann Shapiro. Ms.
Ann Shapiro is the wife of one of the BOD members Dr. Roy Shapiro,
so he had direct insight on this product line. This transaction was
completed as a related party transaction with Arrestage. As part of
the license agreement Arrestage agreed to pay $50,000 in
three installments of unspecified amounts and on unspecified
dates in exchange for consideration received. The
license agreement expires in 2025.
“GRANT
OF LICENSE. ALS owns Registered Trademark of Arrestage and cosmetic
formulas ("ARRESTAGE MARK & FORMULAS"). In accordance with this
Agreement, ALS grants All an exclusive license to sell the
ARRESTAGE MARK & FORMULAS. ALS retains title and ownership of
the ARRESTAGE MARK & FORMULAS.”
All
rights other than those specifically granted herein to Licensee are
reserved to Licensor, including, without limitation, Licensors
right to continue to use the Licensed Property in any form, manner,
and medium.
Components of our results of operations
We plan
to manage our business globally within two reportable segments,
Nutraceutical Division and the Medical Device Rental Division,
which is consistent with how our management reviews our business,
prioritizes investment and resource allocation decisions and
assesses operating performance.
67
Revenue
We
anticipate that our revenues will relate to the sale and licensing
of products and rental fees. Revenues for service contracts will be
recognized in accordance with current accounting
guidance.
Cost of sales
We
anticipate that a significant portion of our costs of sales will be
reliant on third-party manufacturers for the production and
manufacturing of our main product. Cost of sales would likely
consist primarily of manufacturing, procurement and shipping,
overhead costs, direct material costs, direct labor, depreciation
and amortization. We anticipate that a significant portion of our
cost of sales will consist of costs paid to our third-party
manufacturer.
Gross profit
We
calculate gross profit as net revenue less cost of sales. Our gross
profit will be affected by a variety of factors, licensing fees and
charges, including production volumes, manufacturing costs, product
reliability and the implementation over time of cost-reduction
strategies. Our gross profit will likely fluctuate from quarter to
quarter.
Selling and marketing
We plan
to focus on two primary markets, private dermatology practices and
products and rental revenue of our future device division in both
private and hospital settings. We plan to employ a multi-tier sales
strategy in an attempt to optimize geographic coverage and focus on
what we perceive to be our key markets. This multi-tier sales model
would use a direct sales force and would eventually add
international dealers and distributors, and, to a lesser degree,
regional independent sales representatives compensated on a
commission-only basis.
General and administrative
General
and administrative expense, or G&A, will consist primarily of
salaries, employee benefits, bonuses, and related costs for
personnel who support our general operations such as executive
management, information technology, finance, accounting and human
resource functions, as well as legal and other professional fees.
These fees are currently extremely low and we expect them to
increase as operations ramp up. We expect the amount of G&A
expenses to continue to increase for the foreseeable future as we
will employ additional personnel and incur additional legal,
accounting, insurance and other professional service fees
associated with being a public company. We expect G&A expenses
to continue to decrease as a percentage of net revenue if we are
successful in growing the sales of our products, licensing, and
rental of equipment.
Research and development
We
expect the amount of our research and development, or R&D,
expense to increase as we continue to innovate and introduce new
products and technologies. However, we anticipate that our R&D
costs will decrease as a percentage of net revenues over time if we
are successful growing the sales of our products.
68
Income taxes
We
account for income taxes under the asset and liability method,
which requires the recognition of deferred tax assets and
liabilities for the expected future tax consequences of events that
have been included in the financial statements. Under this method,
we determine deferred tax assets and liabilities on the basis of
the differences between the financial statement and tax basis of
assets and liabilities by using enacted tax rates in effect for the
year in which the differences are expected to reverse. The effect
of a change in tax rates on deferred tax assets and liabilities is
recognized in income in the period that includes the enactment
date.
We
recognize deferred tax assets to the extent that we believe that
these assets are more likely than not to be realized. In making
such a determination, we consider all available positive and
negative evidence, including future reversals of existing taxable
temporary differences, projected future taxable income,
tax-planning strategies, and results of recent operations. If we
determine that we would be able to realize our deferred tax assets
in the future in excess of their net recorded amount, we would make
an adjustment to the deferred tax asset valuation allowance, which
would reduce the provision for income taxes.
We
record uncertain tax positions in accordance with ASC 740 on the
basis of a two-step process in which (1) we determine whether it is
more likely than not that the tax positions will be sustained on
the basis of the technical merits of the position and (2) for those
tax positions that meet the more-likely-than-not recognition
threshold, we recognize the largest amount of tax benefit that is
more than 50 percent likely to be realized upon ultimate settlement
with the related tax authority. The
Company accounted for income taxes using the asset and liability
method which required the Company to record deferred tax assets and
liabilities for the differences between the financial statement and
tax basis of assets and liabilities using the expected applicable
future tax.
69
Results of operations
STATEMENTS OF OPERATIONS
The
accompanying footnotes are an integral part of these financial
statements.
|
|
Year Ended
December 31, 2016
|
Year Ended
December 31, 2015
|
|
|
|
|
|
Revenues
|
$-
|
$-
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Legal
and Professional Fees
|
26,000
|
-
|
|
Stock
Based Compensation
|
-
|
10,000
|
|
Accounting/Bookkeeping
|
2,500
|
-
|
|
Other
Expense
|
1,032
|
1,136
|
|
Total
Operating Expenses
|
29,532
|
11,136
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Interest
Expense
|
1,262
|
1,163
|
|
|
|
|
|
Total
Expenses
|
30,794
|
12,299
|
|
|
|
|
|
Net
(Loss)
|
$(30,794)
|
$(12,299)
|
70
|
|
|||
|
Statements of Operations
|
|||
|
(Unaudited)
|
|
|
Nine Months Ended
September 30, 2017
|
Nine Months Ended
September 30, 2016
|
|
|
|
|
|
Revenues
|
$-
|
$-
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Legal
and Professional Fees
|
14,138
|
20,000
|
|
Accounting/Bookkeeping
|
1,535
|
2,500
|
|
Other
Expenses
|
137
|
793
|
|
Total
Operating Expenses
|
15,810
|
23,293
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Interest
Expense
|
1,439
|
945
|
|
|
|
|
|
Total
Expenses
|
17,249
|
24,238
|
|
|
|
|
|
Net
(Loss)
|
$(17,249)
|
$(24,238)
|
Seasonality
We do
not believe our business to be seasonal in nature.
Liquidity and capital resources
Overview
As of
December 31, 2016 we had cash and cash equivalents of $6,568 and an
accumulated deficit of $185,831 as compared to cash and cash
equivalents of $290 and an accumulated deficit of $155,037 as of
December 31, 2015.
71
Our
liquidity position and capital requirements may be impacted by a
number of factors, including the following:
|
●
|
our
ability to generate and increase revenue;
|
|
●
|
fluctuations
in gross margins, operating expenses and net loss; and
|
|
●
|
fluctuations
in working capital.
|
Our
primary short-term capital needs, which are subject to change,
include expenditures related to:
|
●
|
Anticipated
sales, marketing and distribution activities;
|
|
●
|
Anticipated
expansion of our research and development activities;
|
|
●
|
Anticipated
rental income
|
|
●
|
Maintenance
of consistent ratios of costs to revenue
|
We
regularly evaluate our cash requirements for current operations,
commitments, capital requirements and business development
transactions, and we may elect to raise additional funds for these
purposes in the future.
We may
need to raise additional funds to finance future cash needs through
public or private equity offerings, debt financings, receivables or
royalty financings or corporate collaboration and licensing
arrangements. The covenants under our credit facilities limit our
ability to obtain additional debt financing. We cannot be certain
that additional funding will be available on acceptable terms, or
at all. Any failure to raise capital in the future could have a
negative impact on our financial condition and our ability to
pursue our business strategies.
If we
raise additional funds by issuing equity securities or convertible
debt, our stockholders will experience dilution. Debt financing, if
available, would result in increased fixed payment obligations and
may involve agreements that include covenants limiting or
restricting our ability to take specific actions, such as incurring
additional debt, making capital expenditures or declaring
dividends. Any debt financing or additional equity that we raise
may contain terms, such as liquidation and other preferences, which
are not favorable to us or our stockholders. If we raise additional
funds through collaboration and licensing arrangements with third
parties, it may be necessary to relinquish valuable rights to our
products, future revenue streams or product candidates or to grant
licenses on terms that may not be favorable to us.
72
Cash flows
The
following table provides a summary of our cash flows for the
periods indicated:
|
|
For the Years Ended December 31,
|
|
|
|
2016
|
2015
|
|
|
|
|
|
Net
Cash provided by (Used in):
|
|
|
|
Operating
activities
|
(38,032)
|
(1,136)
|
|
Investing
Activities
|
-
|
-
|
|
Financing
Activities
|
44,310
|
1,253
|
Indebtedness
The
following summary table sets forth all indebtedness of the Company,
which consists of a Demand Note from a related party and a Royalty
Fee Payable:
|
Note Payable
|
|
|
|
|
|
|
|
The
Following is a summary of The Note Payable-Current
|
|
|
|
|
||
|
|
2016
|
2015
|
||||
|
|
Principal
|
Interest payable
|
Total Due Upon Demand
|
Principal
|
Interest payable
|
Total Due Upon Demand
|
|
|
|
|
|
|
|
|
|
Unsecured
Demand Note Payable to The Shapiro Trust, due upon demand. The Note
Payable is a multiple advance demand Note with a stated interest
rate of 8%.
|
18,390
|
4,268
|
22,658
|
12,830
|
3,006
|
15,836
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Royalty Fee Payable
|
|
|
|
|
|
|
|
The following is a summary of the Royalty
Fee Payable
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
A
Trademark Licenses Agreement was signed June 22, 2011 which calls
for Royalty Payments of $50,000 to be paid in three installments,
with no interest, on or before the 15th day of the month for which
the Royalty is due.
|
25,790
|
-
|
25,790
|
25,790
|
-
|
25,790
|
73
Off-balance sheet arrangements
We do
not have off-balance sheet financing arrangements that would have a
material impact on our financial statements.
Critical accounting policies
This
summary of significant accounting policies of Arrestage
International, Inc. (the ”Company”) is presented to
assist in understanding the Company’s financial statements.
The financial statements and footnotes are representations of the
Company’s management, who is responsible for their integrity
and objectivity. These accounting policies conform to generally
accepted accounting principles in the United States of America and
have been consistently applied in the preparation of the financial
statements.
Use of Estimates by Management
The
preparation of financial statements in conformity with accounting
principles generally accepted in the United States requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities and disclosure of
contingent assets and liabilities at the date of the financial
statements and the reported amounts of revenues and expenses during
the reporting period. Actual results could differ from those
estimates.
Cash and Cash Equivalents
Cash
and cash equivalents includes cash in banks and highly liquid
investments with original maturities of three months or less that
are readily convertible into cash and are not subject to
significant risk from fluctuations in interest rates. As a result,
the carrying amount of cash and cash equivalents approximates fair
value.
Inventories
The
Company had no inventory as of December 31, 2015 nor December 31,
2016, but may have inventories in the future. Inventories will
consist of finished products held for re-sale.
74
Property and Equipment
The
Company did not own any Property or Equipment as of December 31,
2015 nor December 31, 2016.
Intangible Assets
Goodwill and Intangible Assets—Intangible assets with finite
useful lives are amortized while intangible assets with indefinite
useful lives are not amortized. Amortizable intangible assets are
reviewed for impairment when events or changes in circumstances
indicate the carrying value may not be recoverable.
Indefinite-lived intangible assets are tested annually for
impairment and when events or changes in circumstances indicate the
carrying value may not be recoverable. The appropriateness of the
indefinite-life classification of non-amortizable intangible assets
is also reviewed as part of the annual testing or when
circumstances warrant a change to a finite life. The Company
performs its annual impairment testing as of December 31 each year,
which is the last day of the Company's fiscal year.
Intangible assets with indefinite useful lives (The Trademark
License) are tested for impairment at the individual asset level by
comparing the fair value of the indefinite-lived intangible asset
to its carrying amount. If the carrying amount of an
indefinite-lived intangible asset exceeds its fair value, an
impairment charge is recognized to reduce the carrying amount to
fair value. Fair values of indefinite-lived intangible assets are
estimated using discounted cash flow (“DCF”)
models.
In using a DCF method of establishing a fair value, management must
make certain assumptions as to the likelihood of having cash flow
from operations in the future based on past performance and
prospects of sales in the future. The Company has no history of
sales and no orders pending. Therefore, it is not possible to
determine an appropriate level of cash receipts from operations
into the future. Based on this assumption, during 2014 management
determined that the fair value of the Trademark License under
Generally Accepted Accounting Principles was zero. Accordingly, the
Company has chosen to write the carrying value of the Trademark
license to zero as of December 31, 2014.
Fair Value of Financial Instruments.
The
Company’s financial instruments include cash, accounts
receivable, employee advances, due from related parties, prepaid
expenses, development costs, deposits, accounts payable, credit
cards payable, accrued expenses, and due to related parties. The
estimated fair value of these instruments approximates its carrying
amount due to the short maturity of these instruments. The carrying
value of short and long-term debts approximates fair value because
those financial instruments bear interest at rates that approximate
current market rates for loans with similar maturities and credit
quality.
Recent Accounting Pronouncements
The Company does not believe that any recently issued, but not yet
effective accounting pronouncements, if adopted, would have a
material effect on the accompanying financial
statements.
75
Basis of Presentation-Going Concern
The accompanying financial statements have been prepared in
conformity with generally accepted accounting principles in the
United States of America, which contemplates continuation of the
Company as a going concern. However, as reflected in the
accompanying financial statements, the Company has no operations, a
net loss of ($30,794) for the year ended December 31, 2016, and
($17,249) for the nine months ended September 30, 2017, an
accumulated deficit of ($185,831) as of December 31, 2016 and
($203,080) as of September 30, 2017, and a working capital
deficiency of ($41,880) and ($62,629) at December 31, 2016 and
September 30, 2017 respectively. This raises substantial doubt
about its ability to continue as a going concern. The ability of
the Company to continue as a going concern is dependent on the
Company's ability to raise additional capital and implement its
business plan. The financial statements do not include any
adjustments that might be necessary if the Company is unable to
continue as a going concern.
These
financial statements are prepared on a going concern basis because
the Directors, officers and significant shareholders have
undertaken to provide continuing financial support so that the
Company is able to pay its debts as and when they fall due. In
addition, Management’s plans to
ensure the Company continues as a going concern include the
pay-down of outstanding debt of the Company and funding of future
operations using proceeds from the revenues expected to be
generated from the sale of products and services within acquired
companies.
While the Company looks to acquire targeted acquisitions, adverse
changes in market conditions or limits on the Company’s
ability to obtain financing could limit the Company’s
acquisition of such targets and the production, marketing and sale
of products and services of the targeted acquisitions. The impact
of such eventualities could influence future operations of the
Company.
Revenue Recognition
Revenue will be recognized when the following criteria are met:
(1) persuasive evidence of an arrangement exists; (2) the
product has been shipped and the customer takes ownership and
assumes the risk of loss; (3) the selling price is fixed or
determinable; and (4) collection of the resulting receivable
is reasonably assured.
Arrestage is reviewing new revenue recognition policy guidance to
determine appropriate revenue recognition for the
future.
Accounts Receivable
The
Company will do business and extend credit to customers based on an
evaluation of the customers’ financial condition, generally
without requiring collateral. Exposure to losses on receivables is
expected to vary by customer due to the financial condition of each
customer. The Company will monitor exposure to credit losses and
maintain allowances for anticipated losses considered necessary
under the circumstances.
76
Income Taxes
The
Company records deferred taxes in accordance with Statement of
Financial Accounting Standards (SFAS) ASC 740, “Accounting
for Income Taxes.” The statement requires recognition of
deferred tax assets and liabilities for temporary differences
between the tax basis of assets and liabilities and the amounts at
which they are carried in the financial statements, the effect of
net operating losses, based upon the enacted tax rates in effect
for the year in which the differences are expected to reverse. A
valuation allowance is established when necessary to reduce
deferred tax assets to the amount expected to be
realized.
Deferred
income taxes arise from temporary timing differences in the
recognition of income and expenses for financial reporting and tax
purposes. The Company’s deferred tax assets consist entirely
of the benefit from net operating loss (NOL) carry forwards. The
net operating loss carry forward if not used, will expire in
various years through 2037, and is severely restricted as per the
Internal Revenue code if there is a change in ownership. The
Company’s deferred tax assets are offset by a valuation
allowance due to the uncertainty of the realization of the net
operating loss carry forwards. Net operating loss carry forwards
may be further limited by other provisions of the tax laws. With
few exceptions, our income tax returns are no longer subject to
Federal tax examinations by tax authorities for years before
December 31, 2011.
The
Company is subject to federal and state income tax. No provision
for tax has been made at December 31, 2016, and 2015 as the Company
had no taxable income for both periods. The following table
reconciles the Company’s statutory tax rate to its effective
tax rate as a percentage of income before income
taxes:
|
|
For the Years
Ended December 31,
|
|
|
|
2016
|
2015
|
|
U.S. statutory
rate
|
34.0%
|
34.0%
|
|
Valuation
allowance
|
-34.0%
|
-34.0%
|
|
Effective tax
rate
|
0.0%
|
0.0%
|
The
components of the Company’s deferred income tax assets are
set forth below:
|
Year Ending
|
Estimated NOL Carry-forward
|
NOL Expires
|
Estimated Tax Benefit from NOL
|
Valuation Allowance
|
Change in Valuation Allowance
|
Net Tax Benefit
|
|
|
|
|
|
|
|
|
|
December 31, 2016
|
$ 129,147
|
Various
|
$ 43,910
|
$ 43,910
|
$ 13,448
|
$ -
|
|
December 31, 2015
|
$ 89,595
|
Various
|
$ 30,462
|
$ 30,462
|
$ 14,443
|
$ -
|
Earnings per Share
Basic
net income (loss) per share attributable to common stockholders is
calculated by dividing the net income (loss) attributable to common
stockholders by the weighted-average number of common shares
outstanding. There were no options or warrants outstanding at
December 31, 2016 nor December 31, 2015. Diluted earnings per share
is not shown for the periods in which the Company incurs a loss
because it would be anti-dilutive.
77
Equity-Based Compensation
Pursuant to
accounting guidance related to accounting for equity-based
compensation, the Company is required to recognize all share-based
payments to non-employees and employees in the financial statements
based on fair values on the grant date. The Company has accounted
for issuance of shares in accordance with the guidance, which
requires the recognition of expense, based on grant-date fair
values, over the service period.
Advertising Costs
Advertising and
promotion expenses will be charged to expense as incurred. There
was no Advertising and promotion expense included in selling
expenses in the accompanying statements of operations.
Operating Leases
The
Company had no operating leases for the periods ending December 31,
2015 and December 31, 2016.
JOBS Act
We
qualify as an “emerging growth company” pursuant to the
provisions of the JOBS Act. For as long as we are an
“emerging growth company,” we may take advantage of
certain exemptions from various reporting requirements that are
applicable to other public companies that are not “emerging
growth companies,” including, but not limited to, not being
required to comply with the auditor attestation requirements of
Section 404(b) of the Sarbanes-Oxley Act, reduced disclosure
obligations regarding executive compensation in our periodic
reports and proxy statements, reduced disclosure obligations
relating to the presentation of financial statements in
Management’s Discussion and Analysis of Financial Condition
and Results of Operations, exemptions from the requirements of
holding advisory “say-on-pay” votes on executive
compensation and shareholder advisory votes on golden parachute
compensation. We have availed ourselves of the reduced reporting
obligations and executive compensation disclosure in this
prospectus, and expect to continue to avail ourselves of the
reduced reporting obligations available to emerging growth
companies in future filings. We have established the OSX rules
“say or pay” within out charter attached per exhibit
hereto.
In
addition, an emerging growth company can delay its adoption of
certain accounting standards until those standards would otherwise
apply to private companies. However, we are choosing to “opt
out” of such extended transition period, and as a result, we
plan to comply with any new or revised accounting standards on the
relevant dates on which non-emerging growth companies must adopt
such standards. Section 107 of the JOBS Act provides that our
decision to opt out of the extended transition period for complying
with new or revised accounting standards is
irrevocable.
Recently issued accounting pronouncements
See our
audited financial statements included elsewhere in this prospectus
for a description of recently issued accounting
pronouncements.
78
BUSINESS
Overview
Arrestage International, Inc. is a
Nevada Company within the nutraceutical and medical device rental
space that plans to develop an international licensing program
whereby we will provide turnkey assistance with operations and
support for distributors in the Skin Care and Anti-Aging arena,
using our brand, custom formulas, trademark, Internet presence and
proprietary marketing protocols. In addition to this, we plan to
continue to develop these aspects of the business and potentially
acquire complementary products and services. Arrestage
International intends to create a significant brand in the
aesthetic skin care marketplace. The officers and directors of AII
all have created, or participated in the marketing and development
of branded products in this space. AII plans to generate a
licensing fee of 5-8% (or a higher rate the market may bear) of all
revenues generated by future licensees.
Arrestage
international has a total of eight employees, two of which are full
time. As the company's operations grow, it may add ational full
time employees within the next six months.
AII
will license full use of all intellectual property and formulas of
Arrestage Laboratories Corporation (“Arrestage
Labs”), in all geographical regions and channels of
distribution. Arrestage Labs was the company that formulated
the products Arrestage International now licenses. Arrestage Labs
is an affiliated company as the principal, Kimberly Shapiro, is a
current board member of Arrestage International. Also, Anne Shapiro
is the wife of Roy Shapiro (a current board member of
Arrestage). AII plans to set itself apart from its
competitors by providing clinically based sales materials that
attract a more sophisticated consumer which may enable the licensee
to increase their sales revenues with higher margins. Licensees
will have access to the luminary panel clinical and marketing
training protocols and collateral materials. Arrestage
International plans to address an ever-increasing world demand for
western culturally influenced aesthetic skin topical products, in
addition to the increasing demand in US domestic and other western
markets. AII seeks to license distribution organizations, in the US
and Internationally, to market its proprietary brand of aesthetic
dermal formulations and additional products that it may potentially
acquire.
The
initial products have gone through clinical and behavioral testing
and several reformulations to achieve the high acceptance status
that they now hold. Due to the high margins, the major aesthetic
device makers are attempting to enter this market as distributors.
This may be a beneficial circumstance as it may enhance the
valuation of AII and widen future exit
possibilities.
The
Company plans to establish a medical device rental program that
purchases or secures medical devices such as MRI systems and
various other diagnostic equipment. Our target market will be
physician groups, clinics, rural hospitals, and insurance
companies. Arrestage intends to obtain residual income from such
rentals and would maintain a fully secured UCC-1 position on each
piece of equipment. To grow this division, Arrestage plans to
aggressively market such services in this coming year to help meet
the expanding demand of medical systems due to the current
healthcare trends. The United States market, the largest in the
world, has reached a value of $133 billion in 2016. To date the
group that the Company is planning to include in its business has
accumulated a strong market presence within this lucrative sector
of secured structured medical lending.
79
Industry overview
Nutraceutical Industry
Nutraceutical Classification is an
industry that is rapidly growing, and it is unique because it
requires the overlapping of two industries that are highly relied
upon in today’s society: food and pharmaceuticals.
Nutraceuticals are natural, bioactive chemical compounds that have
health-promoting, disease-preventing or general medicinal
properties. This category encompasses vitamins, minerals, herbal
supplements, and certain animal products. This industry is becoming
more popular to consumers both because greater self-awareness and
diet education, but also because it incorporates preventative and
curative care into everyday life through food, beverages, and
supplements. The governmental support for this industry is positive
worldwide, and in the United States some health insurances
reimburse consumers for medical foods. This industry is largely
dominated by a few companies, with the 5 top companies held over
50% of the market as of 2014. North America is expected to continue
to lead this industry over this period (NA held 39.6% market share
in 2014. Asia Pacific region held the second largest market share
in 2014 with a significant growth rate. (source 2A)
●
The global
nutraceuticals market was valued at US$165.62 billion in 2014 and
is expected to reach US$278.96 billion by 2021, growing at a CAGR
of 7.3% from 2015 to 2021. (2A)
●
Revenue-functional
food segment held the largest share in the global nutraceuticals
market, accounting for 31.0% market share in 2014 and is expected
to grow at a CAGR of 7.1% from 2015 to 2021. Functional beverage is
2nd and
expected to grow at a CAGR of 7.4 % from 2015-2021.
(2A)
●
Top
companies=Cargill, Archer Daniels Midland Company, Royal DSM N.V,
BASF SE, Nestle S.A., General Mills, Inc., etc. (2A)
●
The WHO predicts
that by 2020 over 65 million people will have dementia and that the
prevalence of other neurodegenerative brain diseases, like
Alzheimer’s, is also increasing. (2B)
●
It is estimated
that over 100 million people globally are effected by ‘gut
health’ problems, and the prevalence is rising.
(2B)
●
Cardiovascular
disease (CVD) is significant. It is the number one cause of death
globally. The risk of contracting CVD is associated with lifestyle
choices including a poor diet. (2B)
80
●
The WHO estimate
that 347 million people worldwide have diabetes. (2B)
●
Sales are predicted
to reach US$250 billion by 2018. (2B)
Medical Device Rental Industry
Medical Equipment Rental is an industry that is expanding
due to many factors in the health care industry. Professionally,
many smaller businesses have found it much more cost effective to
rent expensive equipment that they would normally have to refer
patients to a larger facility to use. Rentals are creating greater
accessibility to medical equipment in many places where there are
not larger facilities close by. Rural areas that only have local
access to small healthcare facilities could see specialized
equipment that would not usually be available. Many medical
professions that deal with varying, rare health issues, such as
emergency clinics, pediatrics, physical therapists, and
oncologists, in small or medium-sized facilities could have much
more reliable access to this specialized equipment. Also, it is
more convenient for patients to receive care and testing all in one
location, and it streamlines the process of receiving test results
when equipment can be found in house. Lastly, for larger medical
facilities renting is beneficial for very specialized equipment
that can be too expensive to purchase. Rentals allow the use of
specialized equipment without undue burdens on a facility’s
budget. Due to the constantly changing advances in medical
technology many places do not have consistent access to the most
helpful equipment. If rented, adjustments in technology would be
much more feasible for tight facility budgets, and more locations
could use the latest technology in the healthcare
market.
Our products and services
The
services of the company which Arrestage plans to include, entails
seeking viable markets for future machine rentals, specifically,
pairing purchased machines with end users in the medical
industries.
We plan to implement a growth strategy which anticipates seeking to
generate our revenues through both internal organic growth and
acquisition. With plans to developing in-house nutraceutical
products and producing formulas, we anticipate we will be able to
bring high quality products to market. As we anticipate building
organic formulations, we also plan to aggressively partner with
other product development companies to create, co-brand, and
license products that are in high demand, such as topical pain
management formulas.
81
In
our medical device plan we intend on partnering with a medical
device company in order to purchase and license scanning technology
such as MRI, PET/CT scanners, and MRI machines. In such case, we
will hold first position UCC-1 liens on such equipment and rent the
machines to end users such as medical clinics, and rural
hospitals.
Strategic Relationship
Our target roll-up has established a number of strategic
relationships with insurance companies, providers, equipment
manufacturers, and sales personnel to deepen and complement our
platform and applications. These relationships include health care
payers, consulting and implementation services provider and broader
health care partners.
Sales and Marketing
Market I
Marketing the
nutraceutical products will require that we utilize both our
in-house personnel and our manufacturing partners (as the companies
that have previously manufactured our products) and marketing
agents. By continuing to grow and produce products that are in high
demand, the potential for licensing deals is eminent. We have
developed a full launch plan for our first two products that we
will roll out, including editorial coverage, social media
marketing, partnership with distribution centers, and sale of
co-branding initiatives.
Market II
Marketing for the
Medical device sector will largely focus on third-party
arrangements for direct sales and placement of machines. Principals
of Arrestage have long standing relationships that have proven
beneficial marketing this sector. As we grow and these
multi-faceted relationships strengthen, capital availability and
placement are anticipated to potentially scale quickly. These
relationships and long-term knowledge of the industry could
potentially create high barriers of entry for our
competition.
82
Healthcare fraud and abuse
Management is
familiar through management experience (as doctors and past
positions as pharmaceutical sales), and well read on the issues
dealing with Healthcare fraud and abuse. The US Department of
Health and Human Services issued a new Health care fraud and abuse
control program. The management of Arrestage has attended seminars
that explain the new control program. Specifically, efforts to combat fraud
were consolidated and strengthened under Public Law 104-191, the
Health Insurance Portability and Accountability Act of 1996
(HIPAA). The Act established a comprehensive program to combat
fraud committed against all health plans, both public and private.
The legislation required the establishment of a national Health
Care Fraud and Abuse Control Program (HCFAC), under the joint
direction of the Attorney General and the Secretary of the
Department of Health and Human Services (HHS) acting through the
Department's Inspector General (HHS/OIG).
Health information privacy
Arrestage
takes all privacy matters very seriously. Although, it would be
very rare and unanticipated for the Company to possess actual
patient documentation or reports, management is educated on the
Health Insurance Portability and Accountability Act, better known
as HIPAA.
Facilities
Arrestage International has its headquarters in Scottsdale,
Arizona, where we occupy an office that we also plan to use as a
distribution center.
Legal proceedings
There
are currently no pending or anticipated legal proceedings. We do
not believe that there are any pending legal proceedings that are
likely to have a material effect on our business, financial
condition or results of operations.
Seasonality
We do
not believe our business to be seasonal in nature.
83
MANAGEMENT
The
following table sets forth the name, age, and position of the
individuals who currently serve as executive officers and directors
of Arrestage International, Inc. The following also includes
certain information regarding our directors’ and
officers’ individual experience, qualifications, attributes
and skills and brief statements of those aspects of our
directors’ backgrounds that led us to conclude that they are
qualified to serve as directors.
|
Name
|
Age
|
Position
|
Period of Service
|
|
Kimberly
Shapiro
|
27
|
Vice
Chairman and Director
|
Since
Inception
|
|
Gary
Croft
|
57
|
Chief
Executive Officer and Director
|
Since
Inception
|
|
Philip
Nuciola
|
45
|
President of
Capital Markets
|
August
1, 2014
|
|
Greg
Van Washington
|
75
|
Executive Vice
President/ Director of Marketing
|
Since
Inception
|
|
Philip
Weisman
|
76
|
Director
|
Since
Inception
|
|
Roy
Shapiro
|
63
|
Director
|
Since
Inception
|
Kimberly Shapiro- Chairman and Director
Ms.
Shapiro has been in the Staffing and Recruiting industry for the
past three years. She previously had focused on a career in
financial services and investments with Northwest Mutual Life. She
worked in the Cosmetic and Beauty industry with various positions
in retail, wholesale as well as Salon services businesses before
graduating from California State University in Long Beach. After
obtaining her Bachelor of Arts degree in Communications and
Business Marketing, Ms. Shapiro was involved in the early
developmental stages and clinical trials of Arrestage
Laboratories’ formulations branding model.
During the past five years, Ms. Shapiro has been in management for
Randstat in Irvine, California. Prior to that, Ms. Shapiro focused
on financial services management at Northwest Mutual Life. Neither
Randstat nor Northwest Mutual Life is affiliated with Arrestage in
any way.
Dr. Roy Shapiro- Director
Dr. Shapiro has worked in diagnostic radiology, and is an expert in
aesthetic procedures using lasers and intense light
sources. He is also
the co-founder of Hi-Tech
Medical Lasers, one of the first Carbon Dioxide Laser companies to
manufacture and market hand held, air-cooled lasers which was sold
to Pfizer. Subsequently, Dr. Shapiro formed Lakes Imaging
Centers, which built and acquired 15 Diagnostic Imaging Centers
operating in eight states and sold them to MedPartners and a
private equity firm. He has also been acting in an advisory
capacity in medical technology to Whartin Private Equity Fund,
Westwood Advisors, ShapiroGroup Aesthetics LLC and CLRS Technology,
and has served on the Board of Directors of HealthMed Services LTD,
a public company. Dr. Shapiro currently sits on the board of an
investment firm that invests in technology transfers to developing
countries.
He has over 20 years of experience creating financial models and
managing transition teams. Dr. Shapiro has overseen strategic
planning and sales expansion of the medical/healthcare businesses
and has played a role in a wide range of executive management
experiences in private and public sectors. In 1997 he constructed
one of the earliest platforms for a Medical Diagnostic Service
Enterprise and helped launch an HIPAA compliance and Image
storage/forward software company. Dr. Shapiro also
specializes in: aesthetic medicine, medical devices, clinic
management, imaging center development, healthcare investing,
anti-aging telemedicine/teleradiology, venture capital &
investment banking. Mr. Shapirno is also a collaborator and officer
of CrestRock Advisors, Inc. (“CrestRock”). CrestRock is
located at 20843 N. Hayden Road, Suite 105-193 in Scottsdale,
Arizona 85255. CrestRock is a structured finance consulting firm.
CrestRock is an affiliate of the offering due to the fact it owns
shares of Arrestage.
84
Gary Croft- Chief
Executive Officer and Director
Mr.
Croft brings over 26 years of experience in dermatology and
aesthetics commercialization to Arrestage International, as well as
worldwide business development and leadership skills. Before
joining Arrestage International, he was Vice President of
Aesthetics at Merz Pharmaceuticals and more recently, President at
The Aesthetics Group, an aesthetics and dermatology consulting
company focused on business planning and marketing services. Mr.
Croft led the Sales organization for OrthoNeutrogena Aesthetics
(Johnson and Johnson) in the U.S. and Canada and has held a variety
of positions in Sales, Marketing, New Product Development and
Business Development with Medicis, GlaxoSmithkline and Aventis from
1985 through 2004. Mr. Croft holds a Bachelor of Arts in Economics
from the University of East Anglia. Mr.
Croft is the Principal, President, and CEO of Yorkshire Square
Craft Brewery Based in Los Angeles, California since 2015.
Yorkshire Square Craft Brewery is located at 1109 Van Ness Ave., in
Torrance, California 90501.
Mr.
Croft is also the Principal of a Los Angeles based Healthcare
consulting firm called The Aesthetics Group that specializes in
aesthetics and skincare sales, marketing and business development.
The Aesthetics Group started in January of 2099 and still operates
today. Neither Yorkshire Square or The Aesthetics Group is
affiliated with Arrestage International in any
way.
Phillip Nuciola- President of Capital Markets
As
Director Product Development Mr. Nuciola is responsible for
determining which projects make it to final review for inclusion at
CrestRock Advisors Portfolio Projects. Phil also serves as Chairman
of CrestRock Advisors Portfolio Projects selection
committee.
Before
becoming the Director of Product Development for CrestRock
Advisors. Phil served as Chairman of the Board CRMI, a publicly
traded Energy company. During Mr. Nuciola’s tenure the
company grew from 1.5 million in assets and 4 million in debt to 14
million in assets and 2 million of debt. Mr. Nuciola has organized
resources, structured and financed numerous Initial Public
Offerings and private placements over his 18 year career as a
Stockbroker holding a series 7 & 63 licensees. Mr. Nuciola
maintained senior positions at Cornerstone Partners, Kingsman
Capital and later with American Freedom Securities. His total
assets under management approached half a billion dollars. Mr.
Nuciola was born and raised in New York City; he attended the
University of Rochester.
Mr. Nuciola is a product development advisor for CrestRock
Advisors, Inc. CrestRock Advisors is a structure financial
consultant firm and is affiliated with Arrestage by owning shares
in the company. Prior to his tenure at CrestRock Mr. nuciola was
Chairman of the Board and President of capital markets for Core
Resource Management, Inc. Core Resource Management, Inc. is a oil
and gas finance company located in Scottsdale,
Arizona.
Greg Washington- Executive Vice President/ Director of
Marketing
Mr.
Washington brings over 40 years of experience in cosmetic surgery
marketing to Arrestage International, as well as international
consumer marketing expertise. Before joining Arrestage
International, he was President and Chief Executive Officer of
Patients Unlimited Marketing Consultants, a full-service agency
specializing in international cosmetic surgery practice marketing
and consultation closure techniques. He has been a requested
speaker and frequent writer for the Societies of Plastic Surgeons,
Facial Plastic Surgeons, Dermatologists and Ophthalmologists in the
United States and Europe for almost 25 years. Over
the past five years and prior to joining Arrestage, Mr. Washington
was Chief Executive Officer of Patients Unlimited Marketing
Consultants as a Cosmetic Surgery Consultant.
Patients
Cosmetic Surgery Consultant is a company that specializes in
consulting on new plastic surgery techniques and products. Patients
Cosmetic Surgery Consultants is not affiliated with Arrestage in
any way.
85
Philip Weisman- Director
Mr.
Weisman has served on Board of Directors for five years and was
Chairman of the Board for two years at Goodwill Industries. Philip
was President of Fieldstone Investment Inc. a Real Estate Developer
and Corporate Broker where he built and operated three profitable
Self-Storage Facilities. Mr. Weisman was President of Greystone
Mortgage Inc. who managed and operated offices located in Nevada,
Colorado, Montana and Oregon. At the Riviera Hotel and Casino in
Las Vegas, Nevada, Mr. Weisman was Vice President and held the Key
Man Gaming license for the property. As President of Cellular City
he was the reseller of cellular minutes to conventioneers in
Chicago and Las Vegas. The Company held exclusive cellular phone
rental agreements. While President of World Wide Communications,
the company achieved revenues of over $20,000,000 in consulting and
resale of AT&T long distance network. From 1975 to 1986 he was
President of World Wide Computer and Communications a $15,000,000
company responsible for Consulting and facilities management for
major clients such as Schenley Industries, Culligan and other Rapid
American subsidiaries of a multi-billion-dollar
conglomerate. During
the past five years in addition to being on the Board of Directors
for Arrestage, Mr. Weisman has been the President of A Nevada based
real estate development firm, Fieldstone Investment, Inc.
Fieldstone has no affiliation with Arrestage
International.
Summary
Compensation Table
The following table
sets forth, for the fiscal years ended December 31, 2017, and
December 31, 2016, certain information regarding the compensation
earned by the Company’s named executive officers. Information
for December 31, 2017 is unaudited.
|
Summary Compensation Table
|
|||
|
Name and Principal Position
|
Fiscal Year Ended December 31,
|
Salary ($)
|
Total Compensation
|
|
|
|||
|
Gary
Croft, CEO
|
2017
|
0
|
0
|
|
|
2016
|
0
|
0
|
|
Kimberly
Shapiro, Vice Chairman
|
2017
|
0
|
0
|
|
|
2016
|
0
|
0
|
|
Phillip
Nuciola, President of Capital Markets
|
2017
|
0
|
0
|
|
|
2016
|
0
|
0
|
|
Greg
Van Washington
|
2017
|
0
|
0
|
|
|
2016
|
0
|
0
|
Employment
Agreements
The Company has no
employment agreements with its executive officers., and no salaries
are being accrued.
Board
of directors and committees
It is
anticipated that salaries for the Board of Directors will be, if
any, deminimis, and have not yet been determined. Currently there
is no direct compensation for participating on the Company board.
The Company will not pay bonuses to Board Members solely for their
roles on the Board. No bonuses have been paid to date. In addition,
management’s base salaries can be increased by our Board of
Directors based on the attainment of financial and other
performance guidelines set by the management of the
Company.
Our
Board of Directors will serve until the next annual meeting of the
stockholders and until successors are duly elected and qualified,
unless earlier removed as provided in the Company’s Corporate
Bylaws. Executive officers serve at the pleasure of the Board of
Directors.
Audit committee
The
Company will grow its Audit committee from its current members of
Dr. Roy Shapiro- Chairman and Mr. Larry Barken. Each member of the
audit committee will meet the definition of “independent
director” for purposes of the listed market rules (we will
add specifics when determined our listing) and the independence
requirements of Rule 10A-3 under the Exchange Act. We also believe
that Mr. Philip Weisman qualifies as an “audit committee
financial expert” under SEC rules.
86
Our
audit committee will be responsible for, among other
matters:
●
appointing,
compensating, retaining, evaluating, terminating and overseeing our
independent registered public accounting firm;
●
discussing with
our independent registered public accounting firm their
independence from management;
●
reviewing with our
independent registered public accounting firm the scope and results
of their audit;
●
approving all
audit and permissible non-audit services to be performed by our
independent registered public accounting firm;
●
overseeing the
financial reporting process and discussing with management and our
independent registered public accounting firm the interim and
annual financial statements that we file with the SEC;
●
reviewing and
monitoring our accounting principles, accounting policies,
financial and accounting controls and compliance with legal and
regulatory requirements;
●
establishing
procedures for the confidential anonymous submission of concerns
regarding questionable accounting, internal control or auditing
matters; and
●
reviewing and
approving related person transactions.
Our
board of directors anticipates that it will adopt a new written
charter for the audit committee to become effective upon listing on
the exchange. Such information will be available on our
website.
Compensation committee
The
Company will grow its Compensation committee from its current
member Mr. Nuciola. Our board of directors has affirmatively
determined that it will elect two members of the compensation
committee that will meet the heightened definition of
“independent director” for purposes of the rules
applicable to members of the compensation committee, and the
definition of “non-employee director” for purposes of
Section 16 of the Exchange Act.
The
compensation committee will be responsible for, among other
matters:
●
annually reviewing
and approving our goals and objectives for executive
compensation;
●
annually reviewing
and approving for the chief executive officer and other executive
officers (1) the annual base salary level, (2) the annual cash
incentive opportunity level, (3) the long-term incentive
opportunity level, and (4) any special or supplemental benefits or
perquisites;
87
●
reviewing and
approving employment agreements, severance arrangements and change
of control agreements for the chief executive officer and other
executive officers, as appropriate;
●
making
recommendations and reports to the board of directors concerning
matters of executive compensation;
●
reviewing
compensation plans, programs and policies;
●
handling such
other matters that are specifically delegated to the compensation
committee by the board of directors from time to time.
Nominating and corporate governance committee
Mr.
Gary Croft currently heads our nominating and corporate governance
committee. Members of such committee will be listed on our web site
before the Company becomes exchange traded. Currently the charter
for such committee calls for responsibilities listed
below.
The
nominating and corporate governance committee will be responsible
for, among other matters:
●
identifying the
requisite skills and characteristics to be found in individuals
qualified to serve as members of the board of
directors;
●
conducting
inquiries into the background and qualifications of possible
candidates;
●
recruiting of
qualified candidates for membership on the board of
directors;
●
recommending for
selection by the board of directors, (1) nominees to the board of
directors and (2) committee members for each committee of the board
of directors;
●
overseeing the
corporate governance of the company;
●
evaluating the
performance of the committee and its charter on an annual
basis;
●
handling such
other matters that are specifically delegated to the nominating and
corporate governance committee by the board of directors from time
to time.
88
Our
board of directors adopted a new written charter for the nominating
and corporate governance committee effective prior to the closing
of this offering, which will be available on our
website.
Role of the board in risk oversight
One of
the key functions of our board of directors is informed oversight
of our risk management process. The board of directors does not
have a standing risk management committee, but rather administers
this oversight function directly through the board of directors as
a whole, as well as through various standing committees of our
board of directors that address risks inherent in their respective
areas of oversight. In particular, our board of directors is
responsible for monitoring and assessing strategic risk exposure
and our audit committee has the responsibility to consider and
discuss our major financial risk exposures and the steps our
management has taken to monitor and control these exposures,
including guidelines and policies to govern the process by which
risk assessment and management is undertaken. The nominating and
corporate governance committee monitors compliance with legal and
regulatory requirements and the effectiveness of our corporate
governance practices, including whether they are successful in
preventing illegal or improper liability-creating conduct. Our
nominating and corporate governance committee is also responsible
for overseeing our risk management efforts generally, including the
allocation of risk management functions among our board of
directors and its committees. Our compensation committee assesses
and monitors whether any of our compensation policies and programs
has the potential to encourage excessive risk-taking. Our audit
committee periodically reviews the general process for the
oversight of risk management by our board of
directors.
89
EXECUTIVE AND DIRECTOR COMPENSATION
Introduction
This
section provides an overview of our executive compensation program,
including a narrative description of the material factors necessary
to understand the information disclosed in the summary compensation
table below. Since 2015, our named executive officers, or NEOs,
were:
Mr.
Phillip Nuciola III, President of Capital Markets;
Mr.
Gary Croft, Chief Executive Officer and Director.
Mr.
Phillip Weisman, Director
Ms.
Kimberly Shapiro, Vice-chairman and Director
Dr Roy
Shapiro, Director
Dr.
Greg Washington, Executive Vice President, Director of
Marketing
Currently,
no members of Arrestage International collect salary or are
anticipated to do so in the near future. However, after the
nearterm when Arrestage decides to provide salaries, the
compensation committee will be responsible for determining the
compensation of the executive officers. As details become
available as to the eventual hiring process, the Company will make
a material disclosure and amend any prior filing, if necessary. The
objective of our compensation program is to provide a total
compensation package to each named executive officer that will
enable us to attract, motivate and retain outstanding individuals,
reward named executive officers for performance and align the
financial interests of each named executive officer with the
interests of our stockholders to encourage each named executive
officer to contribute to our long-term performance and
success.
The
compensation program for our named executive officers consists of
the following elements: base salary; performance-based
discretionary cash bonus; equity-based incentive compensation; and
severance and change-in-control benefits.
Our
compensation committee, with input from the board, determines the
compensation for our named executive officers. Upon completion of
this offering, we will have an independent compensation committee
that meets the enhanced independence standards applicable to
compensation committees and that will be responsible for
determining the compensation for our named executive officers and
administering our equity compensation plans and
awards.
Employment arrangements and agreements
We have
not entered into written employment agreements with the named
executive officers. Eventually, as hired, these agreements will be
negotiated on an arms-length basis and establish the key elements
of compensation.
90
Performance-based cash bonus compensation
It is
anticipated that our executive compensation program will include an
annual performance-based discretionary cash bonus. Our board of
directors along with the compensation committee will approve the
terms and conditions of these awards on an annual
basis.
Benefits and Perquisites
We will
offer health and welfare benefits and life insurance to our named
executive officers on the same basis that these benefits are
offered to our other eligible employees, except that we pay the
employee contribution toward the cost of health insurance for our
named executive officers and we will provide our named executive
officers the opportunity for an executive physical. We will offer a
401(k) plan to all eligible employees.
Summary compensation table
The
financial statement sets forth information concerning the total
compensation awarded to, earned by or paid to the named executive
officers for the fiscal year ended December 31, 2016 calculated in
accordance with SEC rules and regulations.
Since
there has been no compensation for any executives, no disclosure is
necessary at this time.
Outstanding equity awards at fiscal year-end
We have
not entered into written employment agreements with the named
executive officers. Eventually, as hired, these agreements will be
negotiated on an arms-length basis and establish the key elements
of compensation.
Director compensation
In 2015
and 2016, we did not pay any cash fees or grant any equity or
equity-based awards to our directors in connection with their
service on our board of directors and currently do not pay for such
service. We are considering adopting a small compensation program
for non-employee directors following the closing of our initial
public offering.
91
PRINCIPAL STOCKHOLDERS
The
following table sets forth information as of September, 2017
regarding the beneficial ownership of our common
stock:
●
each
person or group known by us to beneficially owns more than 5% of
our outstanding shares of common stock;
●
each
of our named executive officers;
●
each
of our directors; and
●
all of
our current executive officers and directors as a
group.
Beneficial
ownership for the purposes of the following table is determined in
accordance with the rules and regulations of the SEC. These rules
generally provide that a person is the beneficial owner of
securities if such person has or shares the power to vote or direct
the voting of securities, or to dispose or direct the disposition
of securities or has the right to acquire such powers within 60
days. For purposes of calculating each person’s percentage
ownership, common stock issuable pursuant to options exercisable
within 60 days are included as outstanding and beneficially owned
for that person or group, but are not deemed outstanding for the
purposes of computing the percentage ownership of any other person.
Except as disclosed in the footnotes to this table and subject to
applicable community property laws, we believe that each beneficial
owner identified in the table possesses sole voting and investment
power over all common stock shown as beneficially owned by the
beneficial owner.
The
percentage of beneficial ownership is based on 3,600,000 shares of
common stock outstanding prior to this offering, shares of common
stock to be outstanding after the completion of this offering,
assuming no exercise of the underwriters’ option to purchase
additional shares of our common stock and shares of common stock to
be outstanding after the completion of this offering, assuming
exercise of the underwriters’ option to purchase additional
shares of our common stock in full.
The
address for each beneficial owner is
c/o
Arrestage International, Inc., 20343
N. Hayden Road, Suite 101
Scottsdale, Arizona 85255
The rest of this page is intentionally left blank
92
Principal Stockholders
|
|
Prior to Offering
|
After this offering (1)
|
||
|
|
Number
|
|
Number
|
|
|
Name
|
of shares
|
AS a %
|
of shares
|
AS a %
|
|
5% Shareholders
|
|
|
|
|
|
The Shapiro Trust (2)
|
1,443,000
|
40.08%
|
1,443,000
|
25.77%
|
|
CrestRock Advisors (3)
|
1,143,000
|
31.75%
|
1,143,000
|
20.41%
|
|
The Demitra Trust (4)
|
300,000
|
8.33%
|
300,000
|
5.36%
|
|
Plum Mountain Trust (5)
|
300,000
|
8.33%
|
300,000
|
5.36%
|
|
Total 5% Shareholders
|
3,186,000
|
88.50%
|
3,186,000
|
56.89%
|
|
Officers and Directors
|
|
|
|
|
|
Gary Croft
|
100,000
|
2.78%
|
100,000
|
1.79%
|
|
Greg Washington
|
50,000
|
1.39%
|
50,000
|
0.89%
|
|
Kimberly Shapiro
|
50,000
|
1.39%
|
50,000
|
0.89%
|
|
Phillip Weisman
|
50,000
|
1.39%
|
50,000
|
0.89%
|
|
Total Officers and Directors
|
250,000
|
6.94%
|
250,000
|
4.46%
|
|
Total for 5% Shareholders and Officers and Directors
|
3,436,000
|
95.44%
|
3,436,000
|
61.36%
|
|
All Other Shareholders
|
164,000
|
4.56%
|
2,164,000
|
38.64%
|
|
Total Shareholders
|
3,600,000
|
100.00%
|
5,600,000
|
100.00%
|
|
(1)
|
After the Offering contemplated herein
|
|
(2)
|
Roy Shapiro, as director, is also a beneficiary of The Shapiro
Trust
|
|
(3)
|
Roy Shapiro and Phillip Nuciola, directors, are principals of
CrestRock Advisors
|
|
(4)
|
Denise
Weisman, daughter of Director Phillip Weisman, is the beneficiary
of The Demetra Trust
|
|
(5)
|
Phillip
Nuciola, President of Capital Markets, is the beneficiary of the
Plum MountainTrust
|
93
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Prior
to the completion of this offering, we expect to adopt a written
policy on transactions with related persons. Under SEC rules, a
related person is an officer, director, nominee for director or
beneficial owner of more than 5% of any class of our voting
securities (our 5% Security Holders) or an immediate family member
of any of the foregoing. Pursuant to our related party transaction
written policy, directors (including director nominees), executive
officers and employees will be required to report any transactions
or circumstances that may create or appear to create a conflict
between the personal interests of the individual and our interests,
regardless of the amount involved.
The
audit committee of the board of directors is responsible for
evaluating each related party transaction and making a
recommendation to the disinterested members of the board of
directors as to whether the transaction at issue is fair,
reasonable and within our policy and whether it should be ratified
and approved. In the course of its review and approval of related
party transactions, our audit committee will consider the relevant
facts and circumstances to decide whether to approve such
transactions. Our audit committee will approve only those
transactions that it determines are in our best interest. In
particular, our policy on related party transactions will require
our audit committee to consider, among other factors it deems
appropriate:
●
whether the
related party transaction is on terms no less favorable than terms
generally available to an unaffiliated third party under the same
or similar circumstances; and
●
the
extent of the related party’s interest in the
transaction.
Pursuant to our
policy on related party transactions, our audit committee will
identify the following categories of transactions as deemed to be
preapproved by the audit committee, even if the aggregate amount
involved is less than (i) $120,000 or (ii) one percent of the
Company’s average assets as of the last day of the
Company’s two most recent fiscal years:
●
our
employment of any executive officer or compensation paid by us to
any executive officer if our compensation committee approved (or
recommended that our board of directors approve) such
compensation;
●
any
compensation paid to a director if the compensation is required to
be reported in our proxy statement under Item 402 of the
SEC’s compensation disclosure requirements;
●
any
transaction with another company at which a related person’s
only relationship is as an employee (other than an executive
officer), director or beneficial owner of less than 10% of that
company’s shares, if the aggregate amount involved does not
exceed the greater of $100,000, or 2% of that company’s total
annual net product revenues;
●
any
charitable contribution, grant or endowment made by us to a
charitable organization, foundation or university at which a
related person’s only relationship is as an employee (other
than an executive officer) or a director, if the aggregate amount
involved does not exceed the lesser of $100,000, or 2% of the
charitable organization’s total annual receipts;
94
●
any
transaction where the related person’s interest arises solely
from the ownership of our common stock and all holders of our
common stock received the same benefit on a pro rata
basis;
●
any
transaction involving a related person where the rates or charges
involved are determined by competitive bids;
●
any
transaction with a related person involving the rendering of
services as a common or contract carrier, or public utility, at
rates or charges fixed in conformity with law or governmental
authority; and
●
any
transaction with a related person involving services as a bank
depositary of funds, transfer agent, registrar, trustee under a
trust indenture, or similar services.
In
addition, our code of business conduct and ethics, which will
become effective upon our corporate conversion, requires that each
of our employees and directors inform his or her superior or the
chairman of the audit committee, respectively, of any material
transaction or relationship that comes to their attention that
could reasonably be expected to create a conflict of interest.
Further, at least annually, each director and executive officer
will complete a detailed questionnaire that asks questions about
any business relationship that may give rise to a conflict of
interest and all transactions in which we are involved and in which
the executive officer, a director or a related person has a direct
or indirect material interest.
Other
than compensation agreements and other arrangements which are
described under “Executive and Director Compensation”
and the transactions described below, since inception there has not
been, and there is not currently proposed, any transaction or
series of similar transactions to which we were or will be a party
in which the amount involved exceeded or will exceed $82,890 (our
current threshold for reporting related party transactions) and in
which any related person had or will have a direct or indirect
material interest.
Limitation of liability and indemnification
As
permitted by Nevada law, we adopted provisions in our certificate
of incorporation, which will be effective as of the closing date of
this offering, that limit or eliminate the personal liability of
our directors. Our certificate of incorporation limits the
liability of directors to the maximum extent permitted by Nevada
law. Nevada law provides that directors of a corporation will not
be personally liable for monetary damages for breaches of their
fiduciary duties as directors, except liability for:
95
●
any
breach of the director’s duty of loyalty to us or our
stockholders;
●
any
act or omission not in good faith or that involves intentional
misconduct or a knowing violation of law;
●
any
unlawful payments related to dividends or unlawful stock
repurchases, redemptions or other distributions; or
●
any
transaction from which the director derived an improper personal
benefit.
These
limitations do not apply to liabilities arising under federal
securities laws and do not affect the availability of equitable
remedies, including injunctive relief or rescission. If Nevada law
is amended to authorize the further elimination or limiting of a
director, then the liability of our directors will be eliminated or
limited to the fullest extent permitted by Nevada law as so
amended.
As
permitted by Nevada law, our certificate of incorporation or
bylaws, as applicable, that will be effective as of the closing
date of this offering also provide that:
●
we
will indemnify our directors and officers to the fullest extent
permitted by law; and
●
we
will advance expenses to our directors and officers in connection
with legal proceedings in connection with a legal proceeding for
which indemnification is required.
Pursuant to Nevada
law, we may, by action of our board of directors, also indemnify
our other employees and other agents to the same extent that we may
indemnify our officers and directors.
We
anticipate entering into indemnification agreements with our
directors and officers to provide such officers and directors with
additional contractual assurances regarding the scope of their
indemnification. We expect that each of these indemnification
agreements will provide that we will indemnify the director or
officer to the fullest extent permitted by law for claims arising
in his capacity as a director or officer, provided that he acted in
good faith and in a manner that he reasonably believed to be in, or
not opposed to, our best interests and, with respect to any
criminal proceeding, had no reasonable cause to believe that his
conduct was unlawful. We expect that each of these indemnification
agreements will provide that in the event that we do not assume the
defense of a claim against a director or officer, we will be
required to advance his expenses in connection with his defense,
provided that he undertakes to repay all amounts advanced if it is
ultimately determined that he is not entitled to be indemnified by
us.
We also
intend to purchase and maintain insurance on behalf of any person
who is or was a director or officer against any loss arising from
any claim asserted against him or her and incurred by him or her in
any such capacity, subject to certain exclusions.
We
believe that these provisions and agreements are necessary to
attract and retain qualified persons as directors and officers.
Insofar as indemnification for liabilities arising under the
Securities Act may be permitted to directors, officers or persons
controlling our company pursuant to the foregoing provisions, we
understand that in the opinion of the SEC such indemnification is
against public policy as expressed in the Securities Act and is
therefore unenforceable.
See Exhibit 10.5.
96
DESCRIPTION OF CAPITAL STOCK
The following description summarizes important terms of our capital
stock. For a complete description, you should refer to our
certificate of incorporation and bylaws, copies of which have been
filed as exhibits to the registration statement of which this
prospectus is a part, as well as the relevant portions of the
Nevada, “NRS”.
Description of Capital Stock
The Company has two classes of authorized capital stock: Common
Stock and Preferred Stock. Both were authorized June 22, 2011and
both have a par value of $001. The holders of Common Stock are
entitled to one vote per share. Rights and Preferences for the
Preferred Stock will be determined by the Board of Directors prior
to issuance of any Preferred Stock.
Common stock
All right fully defined in corporate bylaws attached hereto as
exhibit.
General. Our certificate of
incorporation authorizes the issuance of up to 30,000,000 shares of
our common stock and 5,000,000 preferred shares, and there were
3,600,000 shares of our common stock outstanding, as of
December 31, 2016. Following the issuance of
shares contemplated herein 5,600,000 shares of common stock
will be outstanding.
Preferred
stock
Our certificate of
incorporation authorizes the issuance of up to 5,000,000 shares of
our Preferred Stock, and as of December 31, 2016, no shares of
preferred stock were outstanding. Our certificate of incorporation
permits our board of directors to issue preferred stock from time
to time in one or more classes or series. The board also may fix
the relative rights and preferences of those shares, including
dividend rights, conversion rights, voting rights, redemption
rights, terms of sinking funds, liquidation preferences, the number
of shares constituting any class or series and the designation of
the class or series. Terms selected by our board of directors in
the future could decrease the amount of earnings and assets
available for distribution to holders of common stock or adversely
affect the rights and powers, including voting rights, of the
holders of common stock without any further vote or action by the
stockholders. As a result, the rights of holders of our common
stock will be subject to, and may be adversely affected by, the
rights of the holders of any preferred stock that may be issued by
us in the future, which could have the effect of decreasing the
market price of our common stock.
Voting rights
(a) Except as
otherwise provided by law, the Articles of Incorporation, or the
Corporate Bylaws, any corporate action, the affirmative vote of the
majority of shares entitled to vote on that matter and represented
either in person or by proxy at a meeting of Stockholders at which
a quorum is present, shall be the act of the Stockholders of the
Corporation.
(b)
Except as otherwise provided by statute, the Certificate of
Incorporation, or the Corporate Bylaws, at each meeting of
Stockholders, each Stockholder of the Corporation is entitled to
vote thereat, shall be the act of the Stockholders of the
Corporation.
(c)
Where appropriate communication facilities are reasonably
available, any or all Stockholders shall have the right to
participate in any Stockholders’ meeting, by means of
conference telephone or any means of communications by which all
persons participating in the meeting are able to hear each
other.
Dividend rights
Dividends
may be declared and paid out of any funds available therefore, as
often, in such amounts, and at such time or times as the Board of
Directors may determine and shares may be issued pro rata and
without consideration to the Corporation’s Stockholders or to
the Stockholders of one or more classes or series.
97
Other matters
Upon
our liquidation, dissolution or winding up, the holders of common
stock are entitled to share ratably in the net assets legally
available for distribution to stockholders after the payment of all
of our debts and other liabilities, subject to any liquidation
preference granted to holders of any outstanding preferred stock.
Holders of common stock will have no preemptive or conversion
rights or other subscription rights, and no redemption or sinking
fund provisions will be applicable to our common stock. All
outstanding shares of common stock are, and the shares of common
stock to be outstanding upon completion of this offering will be,
fully paid and non-assessable.
Annual Meetings
The
annual meeting of the Stockholders of the Corporation shall be held
at the time fixed, from time to time, by the
Directors.
Special Meetings
Special
meetings of the Stockholders may be called by the Board of
Directors or such person or persons authorized by the Board of
Directors and shall be held within or without the State of
Nevada.
Election, qualification,
(a)
The first Board of
Directors and all subsequent Boards of the Corporation shall
consist of not less than 1 nor more than 10, unless and until
otherwise determined by vote of a majority of the entire Board of
Directors. The Board of Directors or Stockholders all have the
power, in the interim between annual and special meetings of the
Stockholders, to increase or decrease the number of Directors of
the Corporation. A Director need not be a Stockholder of the
Corporation unless the Certificate of Incorporation of the
Corporation or these Bylaws so require.
(b)
Except as may
otherwise be provided herein or in the Articles of Incorporation,
the members of the Board of Directors of the Corporation shall be
elected at the first annual Stockholders’ meeting and at each
annual meeting thereafter, unless their terms are staggered in the
Articles of Incorporation of the Corporation or these Bylaws, by a
plurality of the votes cast at a meeting of Stockholders, by the
holders of shares entitled to vote in the election.
(c)
The first Board of
Directors shall hold office until the first meeting of Stockholders
and until their successors have been duly elected and qualified or
until there is a decrease in the number of Directors. Thereinafter,
Directors will be elected at the annual meeting of Stockholders and
shall hold office until the annual meeting of the Stockholders next
succeeding his election, unless their terms are staggered in the
Articles of Incorporation of the Corporation (so long as at least
one – fourth in number of Directors of the Corporation are
elected at each annual Stockholders’ meeting) or these
Bylaws, or until his prior death, resignation or removal. Any
Director may resign at any time upon written or verbal notice of
such resignation of the Corporation.
98
(d)
All Directors of
the Corporation shall have equal voting power unless the Articles
of Incorporation of the Corporation provide that the voting power
of the individual Directors of classes of Directors are greater
than or less than that of any other individual Directors or classes
of Directors, and the different voting powers may be stated in the
Articles of Incorporation or may be dependent upon any fact or
event that may be ascertained outside the Articles of Incorporation
if the manner in which the fact or event may operate on those
voting powers is stated in the Articles of Incorporation. If the
Articles of Incorporation provide that any Directors have voting
power greater than or less than other Directors of the Corporation,
every reference in these Bylaws to a majority or other proportion
of Directors shall be deemed to refer to a majority or other
proportion of the voting power of all the Directors of classes of
Directors, as may be required by the Articles of
Incorporation.
Removal
Unless
otherwise provided for by the Articles of Incorporation, one or
more or all the Directors of the Corporation may be removed with or
without cause at any time by a vote of two-thirds of the
Stockholders entitled to vote thereon, at a special meeting of the
Stockholders called for that purpose, unless the Articles of
Incorporation provide that Directors may only be removed for cause,
provided however, such Director shall not be removed if the
Corporation states in its Articles of Incorporation that its
Directors shall be elected by cumulative voting and there are a
sufficient number of shares cast against his or her removal, which
if cumulatively voted at an election of Directors would be
sufficient to elect him or her. If a Director was elected by a
voting group of Stockholders, only the Stockholders of that voting
group may participate in the vote to remove that
Director.
Other matters.
Upon
our liquidation, dissolution or winding up, the holders of common
stock are entitled to share ratably in the net assets legally
available for distribution to stockholders after the payment of all
of our debts and other liabilities, subject to any liquidation
preference granted to holders of any outstanding preferred stock.
Holders of common stock will have no preemptive or conversion
rights or other subscription rights, and no redemption or sinking
fund provisions will be applicable to our common stock. All
outstanding shares of common stock are, and the shares of common
stock to be outstanding upon completion of this offering will be,
fully paid and nonassessable.
99
Anti-takeover effects of provisions of our certificate of
incorporation and bylaws and Nevada Law
The
provisions of the NRS and our certificate of incorporation and
bylaws could have the effect of discouraging others from attempting
an unsolicited offer to acquire our company. Such provisions may
also have the effect of preventing changes in our management. It is
possible that these provisions could make it more difficult to
accomplish transactions that stockholders may otherwise deem to be
in their best interests. According to the NRS, Directors
may resist a change or potential
change in control of the corporation if the directors by a majority
vote of a quorum determine that the change or potential change is
opposed to or not in the best interest of the
corporation:
(a) Upon consideration of the
interests of the corporation’s stockholders or any of the
matters set forth in subsection 4 of NRS 78.138; or
(b) Because the amount or nature of
the indebtedness and other obligations to which the corporation or
any successor to the property of either may become subject, in
connection with the change or potential change, provides reasonable
grounds to believe that, within a reasonable time to make a
determination.
Limitation of liability and indemnification
Our
certificate of incorporation provides that no director will be
personally liable for monetary damages for breach of any fiduciary
duty as a director, except with respect to liability:
●
for
any breach of the director’s duty of loyalty to us or our
stockholders;
●
for
acts or omissions not in good faith or that involve intentional
misconduct or a knowing violation of law;
●
Sections under the
NRS that account for liability due to intentional misconduct, per
the regulations listed below.
●
for
any transaction from which the director derived any improper
personal benefit.
Listing
We are
applying to list our common stock on the OTCQB market.
Transfer agent and registrar
The
transfer agent and registrar for our common stock will be V Stock
Transfer Agent, 18 Lafayette Place, Woodmere, NY
11598.
100
THIS PAGE IS LEFT INTENTIONALLY BLANK
101
UNDERWRITING
Arestage
plans on utilizing the OTCQB market to list shares to the public.
No exchange traded calls, puts, or warrants will be available at
the outset of the offering.
Arrestage
has not been previously required to file reports pursuant to
Section 13(a) or 15(d) of the Exchange Act. The prospects
accompanying these securities defined and highlight all material
risks relating to its issue.
Securities
may be offered through the selling efforts of brokers or dealers;
however, the company has no current plan of distribution or any
such agreement currently in place. Thereby, the brokers or dealers
participating in the offering, (if any), are currently
unknown.
No
commissions or finders’ fees of any kind have been paid to
any person or entity, and although no such arrangement is expected,
any such future arrangement will be disclosed by
amendment.
Rather,
Arrestage plans to offer securities to the public on the OTCQB
exchange marketplace, and utilize passive market making
transactions as permitted by Rule 103, of Regulation M. Such
passive market making will express the direction of the
company’s basic plan for grants, highlight basic goals
attained, and explain the growth potential within the Sector in
general. Arrestage will create share demand by generally expressing
its corporate narrative and promised Board members involvement in
its personal network.
As and if Arrestage determines it to in in the best long-term
interests of the Company and its shareholders to engage Brokers or
Dealers, such information will be immediately updated via amendment
as required.
|
Name of
Underwriter
|
Number of
Shares
|
|
NAME OF PERSON OR
FIRM
N/A at this
time
Description of
terms
|
|
102
Discounts and Commissions
No Discounts or
Commissions have been promised or allocated.
The
following table shows the public offering price, underwriting
discounts and commissions and proceeds, before expenses, to us. The
information assumes either no exercise or full exercise by the
underwriters of their over-allotment option.
|
|
|
Total
|
|
|
|
Per
Share
|
Without Over-Allotment
|
With Over-Allotment
|
|
Public offering
price
|
$2.00
|
|
|
|
Underwriting
discounts and commissions
|
$
|
|
|
|
Non-accountable
expense allowance (1)
|
$
|
|
|
|
Proceeds, before
expenses, to us
|
$
|
|
|
Lock-Up Agreements
Notwithstanding the
foregoing, all of the holders of equity prior to this offering have
agreed, subject to limited exceptions, not to dispose of or hedge
any of their common stock or securities convertible into or
exchangeable for shares of common stock during the 360-day lock-up
period after the date of this prospectus without the prior written
consent of the Board and disclosure to all shareholders in accord
with this offering and shareholder rights provisions of Nevada
law.
Passive market making
In
connection with this offering, we may allow underwriters and the
selling group members to engage in passive market making
transactions in our common stock in accordance with Rule 103 of
Regulation M under the Exchange Act, during a period before the
commencement of offers or sales of the shares and extending through
the completion of the distribution. A passive market maker must
display its bid at a price not in excess of the highest independent
bid of that security. However, if all independent bids are lowered
below the passive market maker’s bid, then that bid must then
be lowered when specified purchase limits are
exceeded.
103
Other Relationships
No
other underwriting relationships exist at this time
Offer Restrictions Outside the U.S.
Other
than in the U.S., no action has been taken by us or any of our
affiliates that would permit a public offering of the securities
offered by this prospectus in any jurisdiction where action for
that purpose is required. The securities offered by this prospectus
may not be offered or sold, directly or indirectly, nor may this
prospectus or any other offering material or advertisements in
connection with the offer and sale of any such securities be
distributed or published in any jurisdiction, except under
circumstances that will result in compliance with the applicable
rules and regulations of that jurisdiction. Persons into whose
possession this prospectus comes are advised to inform themselves
about and to observe any restrictions relating to the offering and
the distribution of this prospectus. This prospectus does not
constitute an offer to sell or a solicitation of an offer to buy
any securities offered by this prospectus in any jurisdiction in
which such an offer or a solicitation is unlawful.
Disclosure necessary for Foreign Jurisdictional security
sales:
Canada
The
securities may be sold in Canada only to purchasers purchasing, or
deemed to be purchasing, as principal that are accredited
investors, as defined in National Instrument 45-106 Prospectus
Exemptions or subsection 73.3(1) of the Securities Act (Ontario),
and are permitted clients, as defined in National Instrument 31-103
Registration Requirements, Exemptions and Ongoing Registrant
Obligations. Any resale of the securities must be made in
accordance with an exemption from, or in a transaction not subject
to, the prospectus requirements of applicable securities
laws.
Securities
legislation in certain provinces or territories of Canada may
provide a purchaser with remedies for rescission or damages if this
prospectus (including any amendment thereto) contains a
misrepresentation, provided that the remedies for rescission or
damages are exercised by the purchaser within the time limit
prescribed by the securities legislation of the purchaser’s
province or territory. The purchaser should refer to any applicable
provisions of the securities legislation of the purchaser’s
province or territory for particulars of these rights or consult
with a legal advisor.
Pursuant to section
3A.3 of National Instrument 33-105 Underwriting Conflicts (NI
33-105), the underwriters are not required to comply with the
disclosure requirements of NI 33-105 regarding underwriter
conflicts of interest in connection with this
offering.
104
Australia
This
prospectus is not a disclosure document under Chapter 6D of the
Australian Corporations Act, has not been lodged with the
Australian Securities and Investments Commission and does not
purport to include the information required of a disclosure
document under Chapter 6D of the Australian Corporations Act.
Accordingly, (i) the offer of the common stock under this
prospectus is only made to persons to whom it is lawful to offer
the common stock without disclosure under Chapter 6D of the
Australian Corporations Act under one or more exemptions set out in
section 708 of the Australian Corporations Act, (ii) this
prospectus is made available in Australia only to those persons as
set forth in clause (i) above, and (iii) the offeree must be sent a
notice stating in substance that by accepting this offer, the
offeree represents that the offeree is such a person as set forth
in clause (i) above, and, unless permitted under the Australian
Corporations Act, agrees not to sell or offer for sale within
Australia any of the common stock sold to the offeree within 12
months after its transfer to the offeree under this
prospectus.
China
The
information in this document does not constitute a public offer of
the common stock, whether by way of sale or subscription, in the
People’s Republic of China (excluding, for purposes of this
paragraph, Hong Kong Special Administrative Region, Macau Special
Administrative Region and Taiwan). The common stock may not be
offered or sold directly or indirectly in the PRC to legal or
natural persons other than directly to “qualified domestic
institutional investors.”
European Economic Area — Belgium, Germany,
Luxembourg and Netherlands
The
information in this document has been prepared on the basis that
all offers of common stock will be made pursuant to an exemption
under the Directive 2003/71/EC (“Prospectus
Directive”), as implemented in Member States of the European
Economic Area (each, a “Relevant Member State”), from
the requirement to produce a prospectus for offers of
securities.
An
offer to the public of common stock has not been made, and may not
be made, in a Relevant Member State except pursuant to one of the
following exemptions under the Prospectus Directive as implemented
in that Relevant Member State:
●
to
legal entities that are authorized or regulated to operate in the
financial markets or, if not so authorized or regulated, whose
corporate purpose is solely to invest in securities;
●
to any
legal entity that has two or more of (i) an average of at least 250
employees during its last fiscal year; (ii) a total balance sheet
of more than €43,000,000 (as shown on its last annual
unconsolidated or consolidated financial statements) and (iii) an
annual net turnover of more than €50,000,000 (as shown on its
last annual unconsolidated or consolidated financial
statements);
●
to
fewer than 100 natural or legal persons (other than qualified
investors within the meaning of Article 2(1)(e) of the Prospectus
Directive) subject to obtaining the prior consent of Arrestage
International, Inc. or any underwriter for any such offer;
or
●
in any
other circumstances falling within Article 3(2) of the Prospectus
Directive, provided that no such offer of common stock shall result
in a requirement for the publication by Arrestage International,
Inc. of a prospectus pursuant to Article 3 of the Prospectus
Directive.
105
France
This
document is not being distributed in the context of a public
offering of financial securities (offre au public de titres
financiers) in France within the meaning of Article L.411-1 of the
French Monetary and Financial Code (Code monétaire et
financier) and Articles 211-1 et seq. of the General Regulation of
the French Autorité des marchés financiers
(“AMF”). The common stock have not been offered or sold
and will not be offered or sold, directly or indirectly, to the
public in France.
This
document and any other offering material relating to the common
stock have not been, and will not be, submitted to the AMF for
approval in France and, accordingly, may not be distributed or
caused to distributed, directly or indirectly, to the public in
France.
Such
offers, sales and distributions have been and shall only be made in
France to (i) qualified investors (investisseurs qualifiés)
acting for their own account, as defined in and in accordance with
Articles L.411-2-II-2° and D.411-1 to D.411-3, D. 744-1,
D.754-1 and D.764-1 of the French Monetary and Financial Code and
any implementing regulation or (ii) a restricted number of
non-qualified investors (cercle restreint d’investisseurs)
acting for their own account, as defined in and in accordance with
Articles L.411-2-II-2° and D.411-4, D.744-1, D.754-1 and
D.764-1 of the French Monetary and Financial Code and any
implementing regulation.
Pursuant to Article
211-3 of the General Regulation of the AMF, investors in France are
informed that the common stock cannot be distributed (directly or
indirectly) to the public by the investors otherwise than in
accordance with Articles L.411-1, L.411-2, L.412-1 and L.621-8 to
L.621-8-3 of the French Monetary and Financial Code.
Ireland
The
information in this document does not constitute a prospectus under
any Irish laws or regulations and this document has not been filed
with or approved by any Irish regulatory authority as the
information has not been prepared in the context of a public
offering of securities in Ireland within the meaning of the Irish
Prospectus (Directive 2003/71/EC) Regulations 2005 (the
“Prospectus Regulations”). The common stock have not
been offered or sold, and will not be offered, sold or delivered
directly or indirectly in Ireland by way of a public offering,
except to (i) qualified investors as defined in Regulation 2(l) of
the Prospectus Regulations and (ii) fewer than 100 natural or legal
persons who are not qualified investors.
Israel
The
common stock offered by this prospectus have not been approved or
disapproved by the Israeli Securities Authority, or ISA, nor have
such common stock been registered for sale in Israel. The shares
may not be offered or sold, directly or indirectly, to the public
in Israel, absent the publication of a prospectus. The ISA has not
issued permits, approvals or licenses in connection with the
offering or publishing the prospectus; nor has it authenticated the
details included herein, confirmed their reliability or
completeness, or rendered an opinion as to the quality of the
common stock being offered. Any resale in Israel, directly or
indirectly, to the public of the common stock offered by this
prospectus is subject to restrictions on transferability and must
be effected only in compliance with the Israeli securities laws and
regulations.
106
Italy
The
offering of the common stock in the Republic of Italy has not been
authorized by the Italian Securities and Exchange Commission
(Commissione Nazionale per le Società e la Borsa,
“CONSOB”) pursuant to the Italian securities
legislation and, accordingly, no offering material relating to the
common stock may be distributed in Italy and such securities may
not be offered or sold in Italy in a public offer within the
meaning of Article 1.1(t) of Legislative Decree No. 58 of 24
February 1998 (“Decree No. 58”), other
than:
●
to
Italian qualified investors, as defined in Article 100 of Decree
no. 58 by reference to Article 34-ter of CONSOB Regulation no.
11971 of 14 May 1999 (“Regulation no. 1197l”) as
amended (“Qualified Investors”); and
●
in
other circumstances that are exempt from the rules on public offer
pursuant to Article 100 of Decree No. 58 and Article 34-ter of
Regulation No. 11971 as amended.
Any
offer, sale or delivery of the common stock or distribution of any
offer document relating to the common stock in Italy (excluding
placements where a Qualified Investor solicits an offer from the
issuer) under the paragraphs above must be:
●
made
by investment firms, banks or financial intermediaries permitted to
conduct such activities in Italy in accordance with Legislative
Decree No. 385 of 1 September 1993 (as amended), Decree No. 58,
CONSOB Regulation No. 16190 of 29 October 2007 and any other
applicable laws; and
●
in
compliance with all relevant Italian securities, tax and exchange
controls and any other applicable laws.
Any
subsequent distribution of the common stock in Italy must be made
in compliance with the public offer and prospectus requirement
rules provided under Decree No. 58 and the Regulation No. 11971 as
amended, unless an exception from those rules applies. Failure to
comply with such rules may result in the sale of such common stock
being declared null and void and in the liability of the entity
transferring the common stock for any damages suffered by the
investors.
Japan
The
common stock have not been and will not be registered under Article
4, paragraph 1 of the Financial Instruments and Exchange Law of
Japan (Law No. 25 of 1948), as amended (the “FIEL”)
pursuant to an exemption from the registration requirements
applicable to a private placement of securities to Qualified
Institutional Investors (as defined in and in accordance with
Article 2, paragraph 3 of the FIEL and the regulations promulgated
thereunder). Accordingly, the common stock may not be offered or
sold, directly or indirectly, in Japan or to, or for the benefit
of, any resident of Japan other than Qualified Institutional
Investors. Any Qualified Institutional Investor who acquires common
stock may not resell them to any person in Japan that is not a
Qualified Institutional Investor, and acquisition by any such
person of common stock is conditional upon the execution of an
agreement to that effect.
107
Portugal
This
document is not being distributed in the context of a public offer
of financial securities (oferta pública de valores
mobiliários) in Portugal, within the meaning of Article 109 of
the Portuguese Securities Code (Código dos Valores
Mobiliários). The common stock have not been offered or sold
and will not be offered or sold, directly or indirectly, to the
public in Portugal. This document and any other offering material
relating to the common stock have not been, and will not be,
submitted to the Portuguese Securities Market Commission (Comissao
do Mercado de Valores Mobiliários) for approval in Portugal
and, accordingly, may not be distributed or caused to distributed,
directly or indirectly, to the public in Portugal, other than under
circumstances that are deemed not to qualify as a public offer
under the Portuguese Securities Code. Such offers, sales and
distributions of common stock in Portugal are limited to persons
who are “qualified investors” (as defined in the
Portuguese Securities Code). Only such investors may receive this
document and they may not distribute it or the information
contained in it to any other person.
Sweden
This
document has not been, and will not be, registered with or approved
by Finansinspektionen (the Swedish Financial Supervisory
Authority). Accordingly, this document may not be made available,
nor may the common stock be offered for sale in Sweden, other than
under circumstances that are deemed not to require a prospectus
under the Swedish Financial Instruments Trading Act (1991:980) (Sw.
lag (1991:980) om handel med finansiella instrument). Any offering
of common stock in Sweden is limited to persons who are
“qualified investors” (as defined in the Financial
Instruments Trading Act). Only such investors may receive this
document and they may not distribute it or the information
contained in it to any other person.
Switzerland
The
common stock may not be publicly offered in Switzerland and will
not be listed on the SIX Swiss Exchange (“SIX”) or on
any other stock exchange or regulated trading facility in
Switzerland. This document has been prepared without regard to the
disclosure standards for issuance prospectuses under art. 652a or
art. 1156 of the Swiss Code of Obligations or the disclosure
standards for listing prospectuses under art. 27 ff. of the SIX
Listing Rules or the listing rules of any other stock exchange or
regulated trading facility in Switzerland. Neither this document
nor any other offering material relating to the common stock may be
publicly distributed or otherwise made publicly available in
Switzerland.
Neither
this document nor any other offering material relating to the
common stock have been or will be filed with or approved by any
Swiss regulatory authority. In particular, this document will not
be filed with, and the offer of common stock will not be supervised
by, the Swiss Financial Market Supervisory Authority
(“FINMA”).
This
document is personal to the recipient only and not for general
circulation in Switzerland.
108
United Arab Emirates
Neither
this document nor the common stock have been approved, disapproved
or passed on in any way by the Central Bank of the United Arab
Emirates or any other governmental authority in the United Arab
Emirates, nor has Arrestage International, Inc. received
authorization or licensing from the Central Bank of the United Arab
Emirates or any other governmental authority in the United Arab
Emirates to market or sell the common stock within the United Arab
Emirates. This document does not constitute and may not be used for
the purpose of an offer or invitation. No services relating to the
common stock, including the receipt of applications or the
allotment or redemption of such shares, may be rendered within the
United Arab Emirates by Arrestage International, Inc.
No
offer or invitation to subscribe for common stock is valid or
permitted in the Dubai International Financial Centre.
United Kingdom
Neither
the information in this document nor any other document relating to
the offer has been delivered for approval to the Financial Services
Authority in the United Kingdom and no prospectus (within the
meaning of section 85 of the Financial Services and Markets Act
2000, as amended (“FSMA”)) has been published or is
intended to be published in respect of the common stock. This
document is issued on a confidential basis to “qualified
investors” (within the meaning of section 86(7) of FSMA) in
the United Kingdom, and the common stock may not be offered or sold
in the United Kingdom by means of this document, any accompanying
letter or any other document, except in circumstances which do not
require the publication of a prospectus pursuant to section 86(1)
FSMA. This document should not be distributed, published or
reproduced, in whole or in part, nor may its contents be disclosed
by recipients to any other person in the United
Kingdom.
Any
invitation or inducement to engage in investment activity (within
the meaning of section 21 of FSMA) received in connection with the
issue or sale of the common stock has only been communicated or
caused to be communicated and will only be communicated or caused
to be communicated in the United Kingdom in circumstances in which
section 21(1) of FSMA does not apply to Arrestage International,
Inc.
In the
United Kingdom, this document is being distributed only to, and is
directed at, persons (i) who have professional experience in
matters relating to investments falling within Article 19(5)
(investment professionals) of the Financial Services and Markets
Act 2000 (Financial Promotions) Order 2005 (“FPO”),
(ii) who fall within the categories of persons referred to in
Article 49(2)(a) to (d) (high net worth companies, unincorporated
associations, etc.) of the FPO or (iii) to whom it may otherwise be
lawfully communicated (together “relevant persons”).
The investments to which this document relates are available only
to, and any invitation, offer or agreement to purchase will be
engaged in only with, relevant persons. Any person who is not a
relevant person should not act or rely on this document or any of
its contents.
109
LEGAL MATTERS
There
are no outstanding or pending material legal matters in which the
Company is a Plaintiff or Defendant at this time.
The law
office of Erica Loren Jozwiak Esq. have reviewed this offering and
performed diligence of any potential legal threats.
110
EXPERTS
The
financial statements of Arrestage International, Inc. as of
December 31, 2016 and 2015, and for each of the years in the
two-year period ended December 31, 2016, have been included herein
and in the registration statement in reliance upon the report of
Schumacher & Associates, Inc. CPA, an independent registered
public accounting firm, appearing elsewhere herein, and upon the
authority of said firm as experts in accounting and auditing.
The balance sheet as of September 30,
2017, the statements of operations and cash flows for the nine
month periods ended September 30, 2017 and 2016, and the statement
of stockholders' (deficit) for the nine month period ended
September 30, 2017, have been prepared by the Company without
audit, pursuant to the rules and regulations of the Securities and
Exchange Commission.
111
WHERE YOU CAN FIND MORE INFORMATION
In addition,
to this registration statement and as a result of this offering, we
will become subject to the information and periodic reporting
requirements of the Exchange Act, and will file annual, quarterly
and current reports and other information with the SEC. Our SEC
filings, including the registration statement on Form S-1 and all
filed exhibits and schedules thereto, are available to the public
on the SEC’s website at http://www.sec.gov . To receive
copies of public records not posted to the SEC’s website at
prescribed rates, you may complete an online form
at http://www.sec.gov , send a fax to
(202) 772-9337 or submit a written request to the SEC, Office of
FOIA/PA Operations, 100 F Street, N.E., Washington, D.C. 20549.
Please call the SEC at 1-800-SEC-0330 for further
information.
112
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM
Board
of Directors
Arrestage
International, Inc.
We have
audited the accompanying balance sheets of Arrestage International,
Inc., as of December 31, 2016 and 2015, and the related statements
of operations, stockholders’ equity (deficit), and cash flows
for each of the two years in the period ended December 31, 2016.
These financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our
audit.
We
conducted our audit in accordance with the standards of the Public
Company Accounting Oversight Board (United States of America).
Those standards require that we plan and perform the audit to
obtain reasonable assurance about whether the financial statements
are free of material misstatement. The Company is not required to
have, nor were we engaged to perform an audit of its internal
control over financial reporting. Our audit included consideration
of internal control over financial reporting as a basis for
designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion on
the effectiveness of the Company’s internal control over
financial reporting. Accordingly, we express no such opinion. An
audit includes examining, on a test basis, evidence supporting the
amounts and disclosures in the financial statements. An audit also
includes assessing the accounting principles used and significant
estimates made by management, as well as evaluating the overall
financial statement presentation. We believe our audit provides a
reasonable basis for our opinion.
In our
opinion, the financial statements referred to above present fairly,
in all material respects, the financial position of Arrestage
International, Inc. as of December 31, 2016 and 2015, and the
results of its operations and its cash flows for each of the two
years in the period ended December 31, 2016, in conformity with
accounting principles generally accepted in the United States of
America.
The
accompanying financial statements have been prepared assuming that
the Company will continue as a going concern. As described in Note
1, the Company has no operations, has recurring losses, and has
negative working capital and stockholders’ deficits at
December 31, 2016, which raise substantial doubt about its ability
to continue as a going concern. Management’s plans in regard
to this matter are also discussed in Note 1. The financial
statements do not include any adjustments that might result from
the outcome of this uncertainty.
SCHUMACHER
& ASSOCIATES, INC.
Littleton,
Colorado
December
12, 2017
113
INDEX TO THE FINANCIAL STATEMENTS OF ARRESTAGE INTERNATIONAL,
INC.
|
Report of Independent Registered Public Accounting
Firm
|
|
|
F-2
|
|
|
Financial Statements
|
|
|
|
|
|
Balance
Sheets as of
September 30, 2017 (Unaudited) and December 31, 2016 and
2015
|
|
|
F-3
|
|
|
Statements
of Operations for the years ended December 31, 2016,
2015
|
|
|
F-4
|
|
|
Statements
of Stockholders’ Equity for the years ended December 31,
2016, 2015
|
|
|
F-6
|
|
|
Statements
of Cash Flows for the years ended December 31, 2016,
2015
|
|
|
F-7
|
|
|
Notes to Financial Statements
|
|
|
F-9
|
|
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM
Board
of Directors
Arrestage
International, Inc.
We have
audited the accompanying balance sheets of Arrestage International,
Inc., as of December 31, 2016 and 2015, and the related statements
of operations, stockholders’ equity (deficit), and cash flows
for each of the two years in the period ended December 31, 2016.
These financial statements are the responsibility of the
Company’s management. Our responsibility is to express an
opinion on these financial statements based on our
audit.
We
conducted our audit in accordance with the standards of the Public
Company Accounting Oversight Board (United States of America).
Those standards require that we plan and perform the audit to
obtain reasonable assurance about whether the financial statements
are free of material misstatement. The Company is not required to
have, nor were we engaged to perform an audit of its internal
control over financial reporting. Our audit included consideration
of internal control over financial reporting as a basis for
designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion on
the effectiveness of the Company’s internal control over
financial reporting. Accordingly, we express no such opinion. An
audit includes examining, on a test basis, evidence supporting the
amounts and disclosures in the financial statements. An audit also
includes assessing the accounting principles used and significant
estimates made by management, as well as evaluating the overall
financial statement presentation. We believe our audit provides a
reasonable basis for our opinion.
In our
opinion, the financial statements referred to above present fairly,
in all material respects, the financial position of Arrestage
International, Inc. as of December 31, 2016 and 2015, and the
results of its operations and its cash flows for each of the two
years in the period ended December 31, 2016, in conformity with
accounting principles generally accepted in the United States of
America.
The
accompanying financial statements have been prepared assuming that
the Company will continue as a going concern. As described in Note
1, the Company has no operations, has recurring losses, and has
negative working capital and stockholders’ deficits at
December 31, 2016, which raise substantial doubt about its ability
to continue as a going concern. Management’s plans in regard
to this matter are also discussed in Note 1. The financial
statements do not include any adjustments that might result from
the outcome of this uncertainty.
SCHUMACHER
& ASSOCIATES, INC.
Littleton,
Colorado
December
12, 2017
F-2
|
ARRESTAGE INTERNATIONAL, INC.
|
|||
|
BALANCE SHEETS
|
|||
|
As of December 31, 2016 and December 31, 2015
|
|||
|
and September 30, 2017 (unaudited)
|
|
|
September 30,
|
December 31,
|
December 31,
|
|
|
2017
(unaudited)
|
2016
|
2015
|
|
ASSETS
|
|||
|
|
|
|
|
|
Current
Assets
|
|
|
|
|
Cash
in Bank
|
$1,199
|
$6,568
|
$290
|
|
|
|
|
|
|
Total
Current Assets
|
1,199
|
6,568
|
290
|
|
|
|
|
|
|
Other
Assets
|
|
|
|
|
Prepaid
Expenses
|
12,000
|
8,500
|
-
|
|
Trademark
License
|
50,000
|
50,000
|
50,000
|
|
Impairment-Trademark
License
|
(50,000)
|
(50,000)
|
(50,000)
|
|
|
|
|
|
|
Total
Other Assets
|
12,000
|
8,500
|
-
|
|
|
|
|
|
|
Total
Assets
|
$13,199
|
$15,068
|
$290
|
|
|
|
|
|
|
LIABILITIES
AND STOCKHOLDERS' (DEFICIT)
|
|||
|
|
|
|
|
|
Current
Liabilities
|
|
|
|
|
|
|
|
|
|
Total
Current Liabilities
|
|
|
|
|
Note
Payable-Related Party
|
$32,331
|
$18,390
|
$12,830
|
|
Accrued
Interest Payable
|
5,707
|
4,268
|
3,006
|
|
Licensee
Fee Payable
|
25,790
|
25,790
|
25,790
|
|
Total
Current Liabilities
|
63,828
|
48,448
|
41,626
|
|
|
|
|
|
|
Total
Liabilities
|
63,828
|
48,448
|
41,626
|
|
|
|
|
|
|
Stockholders'
(Deficit)
|
|
|
|
|
Preferred
Stock, $0.001 par value, 5,000,000 shares of preferred shares
authorized, no shares of preferred stock issued and
outstanding
|
-
|
-
|
-
|
|
Common
Stock, $0.001 par value, 30,000,000 shares of common stock
authorized, 3,600,000 shares issued and outstanding as of September
30, 2017 and December 31, 2016, and 3,561,250 issued and
outstanding December 31, 2015.
|
3,600
|
3,600
|
3,561
|
|
Additional
Paid-in Capital
|
148,851
|
148,851
|
110,140
|
|
Accumulated
(Deficit)
|
(203,080)
|
(185,831)
|
(155,037)
|
|
|
|
|
|
|
Total
Stockholders' (Deficit)
|
(50,629)
|
(33,380)
|
(41,336)
|
|
|
|
|
|
|
Total
Liabilities & Stockholders' (Deficit)
|
$13,199
|
$15,068
|
$290
|
The accompanying notes are an integral part of these financial
statements.
F-3
|
ARRESTAGE INTERNATIONAL, INC.
|
|||
|
Statements of Operations
|
|
|
Year Ended
December 31, 2016
|
Year Ended
December 31, 2015
|
|
|
|
|
|
Revenues
|
$-
|
$-
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Legal
and Professional Fees
|
26,000
|
-
|
|
Stock
Based Compensation
|
-
|
10,000
|
|
Accounting/Bookkeeping
|
2,500
|
-
|
|
Other
Expense
|
1,032
|
1,136
|
|
Total
Operating Expenses
|
29,532
|
11,136
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Interest
Expense
|
1,262
|
1,163
|
|
|
|
|
|
Total
Expenses
|
30,794
|
12,299
|
|
|
|
|
|
Net
(Loss)
|
$(30,794)
|
$(12,299)
|
|
|
|
|
|
Per
Share
|
$(0.01)
|
$
nil
|
|
Weighted
average number of shares outstanding
|
3,580,313
|
3,553,750
|
The
accompanying notes are an integral part of these financial
statements.
F-4
|
ARRESTAGE INTERNATIONAL, INC.
|
|||
|
Statements of Operations
(Unaudited)
|
|
|
Nine Months Ended
September 30, 2017
|
Nine Months Ended
September 30, 2016
|
|
|
|
|
|
Revenues
|
$-
|
$-
|
|
|
|
|
|
Operating
Expenses
|
|
|
|
Legal
and Professional Fees
|
14,138
|
20,000
|
|
Accounting/Bookkeeping
|
1,535
|
2,500
|
|
Other
Expenses
|
137
|
793
|
|
Total
Operating Expenses
|
15,810
|
23,293
|
|
|
|
|
|
Other
Expenses
|
|
|
|
Interest
Expense
|
1,439
|
945
|
|
|
|
|
|
Total
Expenses
|
17,249
|
24,238
|
|
|
|
|
|
Net
(Loss)
|
$(17,249)
|
$(24,238)
|
|
|
|
|
|
Per
Share
|
$ nil
|
$(0.01)
|
|
Weighted
average number of shares outstanding
|
3,580,313
|
3,580,313
|
The accompanying notes are an integral part of these financial
statements.
F-5
|
ARRESTAGE INTERNATIONAL, INC.
|
||||||
|
STATEMENT OF CHANGES IN STOCKHOLDER'S (DEFICIT)
|
||||||
|
For The Period from January 1, 2015 through December 31,
2016
|
||||||
|
and the nine months ended September 30, 2017
|
|
|
Common Stock
|
|
|
|
|
|
|
Shares
|
Amount (Par Value $0.001)
|
Additional Paid In Capital
|
Accumulated (Deficit)
|
Total
|
|
Balance January 1, 2015
|
3,551,250
|
$3,551
|
$100,150
|
$(142,738)
|
$(39,037)
|
|
Stock
issued for services
|
10,000
|
10
|
9,990
|
-
|
10,000
|
|
Net
(Loss)
|
-
|
-
|
-
|
(12,299)
|
(12,299)
|
|
Balance December 31, 2015
|
3,561,250
|
3,561
|
110,140
|
(155,037)
|
(41,336)
|
|
Issuance
of Stock for cash
|
38,750
|
39
|
38,711
|
-
|
38,750
|
|
Net
(Loss)
|
-
|
-
|
-
|
(30,794)
|
(30,794)
|
|
Balance December 31, 2016
|
3,600,000
|
3,600
|
148,851
|
(185,831)
|
(33,380)
|
|
Net
(Loss)
|
-
|
-
|
-
|
(17,249)
|
(17,249)
|
|
Balance at September 30, 2017 (unaudited)
|
3,600,000
|
$3,600
|
$148,851
|
$(203,080)
|
$(50,629)
|
The accompanying notes are an integral part of these
financial statements.
F-6
|
ARRESTAGE INTERNATIONAL, INC.
|
|||
|
STATEMENTS OF CASH FLOWS
|
|
|
For the Years Ended
|
|
|
|
December 31,
|
|
|
|
2016
|
2015
|
|
|
|
|
|
Cash
Flows from operating activities:
|
|
|
|
Net
(Loss)
|
$(30,794)
|
$(12,299)
|
|
|
|
|
|
Adjustments
to reconcile net (loss) to net cash used in operating
activities:
|
|
|
|
Increase
in accrued interest payable
|
1,262
|
1,163
|
|
Stock
based compensation
|
-
|
10,000
|
|
(Increase)
in prepaid offering costs
|
(8,500)
|
-
|
|
|
|
|
|
Total
Adjustments
|
(7,238)
|
11,163
|
|
|
|
|
|
Net
Cash (used in) Operating Activities
|
(38,032)
|
(1,136)
|
|
|
|
|
|
Cash
Flows from financing activities:
|
|
|
|
Proceeds
from note payable-related party
|
5,560
|
1,253
|
|
Proceeds
from sale of common stock
|
38,750
|
-
|
|
|
|
|
|
Net
cash provided by financing activities:
|
44,310
|
1,253
|
|
|
|
|
|
Net
increase in cash
|
6,278
|
117
|
|
|
|
|
|
|
|
|
|
Cash
Balance at Beginning of Period
|
290
|
173
|
|
Cash
Balance at End of Period
|
$6,568
|
$290
|
|
|
|
|
|
Interest
Paid
|
$-
|
$-
|
|
Income
Taxes Paid
|
$-
|
$-
|
The accompanying notes are an integral part of these
financial statements.
F-7
|
ARRESTAGE INTERNATIONAL, INC.
|
|||
|
STATEMENTS OF CASH FLOWS
(Unaudited)
|
|
|
For the Nine Months Ended
|
|
|
|
September 30,
|
|
|
|
2017 (unaudited)
|
2016 (unaudited)
|
|
|
|
|
|
Cash
Flows from operating activities:
|
|
|
|
Net
(Loss)
|
$(17,249)
|
$(24,238)
|
|
|
|
|
|
Adjustments
to reconcile net (loss) to net cash used in operating
activities:
|
|
|
|
(Increase)
in prepaid offering expenses
|
(3,500)
|
(8,500)
|
|
Increase
in accrued interest payable
|
1,439
|
945
|
|
Total
Adjustments
|
(2,061)
|
(7,555)
|
|
|
|
|
|
Net
Cash (used in) Operating Activities
|
(19,310)
|
(31,793)
|
|
|
|
|
|
Cash
Flows from financing activities:
|
|
|
|
Proceeds
from (payments on) note payable-related party
|
13,941
|
(1,134)
|
|
Proceeds
from sale of common stock
|
-
|
38,750
|
|
|
|
|
|
Net
cash provided by financing activities
|
13,941
|
37,616
|
|
|
|
|
|
Net
increase (decrease) in cash
|
(5,369)
|
5,823
|
|
|
|
|
|
Cash
Balance at Beginning of Period
|
6,568
|
290
|
|
Cash
Balance at End of Period
|
$1,199
|
$6,113
|
|
Interest
Paid
|
|
|
|
Income
taxes paid
|
$-
|
$-
|
The accompanying notes are an integral part of these
financial statements.
F-8
ARRESTAGE INTERNATIONAL, INC.
NOTES TO THE FINANCIAL STATEMENTS
December 31, 2016 and 2015
Note
1. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
This
summary of significant accounting policies of Arrestage
International, Inc. (the ”Company”) is presented to
assist in understanding the Company’s financial statements.
The financial statements and footnotes are representations of the
Company’s management, who is responsible for their integrity
and objectivity. These accounting policies conform to generally
accepted accounting principles in the United States of America and
have been consistently applied in the preparation of the financial
statements.Financial statements for September 30, 2017 and/or
September 20, 2016 are unaudited)
Organization
and Description of the Business
The
Company was incorporated under the laws of the State of Nevada on
June 15, 2011 and is in the Nutraceutical business and holds
formulas on skin care products as well as brand formulas, and other
intellectual property.
Use
of Estimates by Management
The
preparation of financial statements in conformity with accounting
principles generally accepted in the United States requires
management to make estimates and assumptions that affect the
reported amounts of assets and liabilities and disclosure of
contingent assets and liabilities at the date of the financial
statements and the reported amounts of revenues and expenses during
the reporting period. Actual results could differ from those
estimates.
Cash and Cash Equivalents
Cash
and cash equivalents includes cash in banks and highly liquid
investments with original maturities of three months or less that
are readily convertible into cash and are not subject to
significant risk from fluctuations in interest rates. As a result,
the carrying amount of cash and cash equivalents approximates fair
value.
Concentrations of Credit Risk
The Company places its cash and cash equivalents
with major financial institutions. At December 31, 2016 and
2015, the Company did not have any cash balances on deposit with
banks which exceeded the balance insured by the FDIC, and does not
believe that it is subject to
any unusual financial risk beyond the normal risk associated with
commercial banking relationships.
Intangible
Assets
Goodwill
and Intangible Assets—Intangible assets with finite useful
lives are amortized while intangible assets with indefinite useful
lives are not amortized. Amortizable intangible assets are reviewed
for impairment when events or changes in circumstances indicate the
carrying value may not be recoverable. Indefinite-lived intangible
assets are tested annually for impairment and when events or
changes in circumstances indicate the carrying value may not be
recoverable. The appropriateness of the indefinite-life
classification of non-amortizable intangible assets is also
reviewed as part of the annual testing or when circumstances
warrant a change to a finite life. The Company performs its annual
impairment testing as of December 31 each year, which is the last
day of the Company's fiscal year.
Intangible assets with indefinite useful lives (The Trademark
License) are tested for impairment at the individual asset level by
comparing the fair value of the indefinite-lived intangible asset
to its carrying amount. If the carrying amount of an
indefinite-lived intangible asset exceeds its fair value, an
impairment charge is recognized to reduce the carrying amount to
fair value. Fair values of indefinite-lived intangible assets are
estimated using discounted cash flow (“DCF”)
models.
F-9
In using a DCF method of establishing a fair value, management must
make certain assumptions as to the likelihood of having cash flow
from operations in the future based on past performance and
prospects of sales in the future. The Company has no history of
sales and no orders pending. Therefore, it is not possible to
determine an appropriate level of cash receipts from operations
into the future. Based on this assumption, during 2014 management
determined that the fair value of the Trademark License under
Generally Accepted Accounting Principles was zero. Accordingly, the
Company has chosen to write the carrying value of the Trademark
license to zero as of December 31, 2014.
Fair Value of Financial Instruments. The Company’s financial
instruments include cash, accounts receivable, employee advances,
due from related parties, prepaid expenses, development costs,
deposits, accounts payable, credit cards payable, accrued expenses,
and due to related parties. The estimated fair value of these
instruments approximates its carrying amount due to the short
maturity of these instruments. The carrying value of short and
long-term debts approximates fair value because those financial
instruments bear interest at rates that approximate current market
rates for loans with similar maturities and credit
quality.
Recent Accounting Pronouncements
The Company does not believe that any recently issued, but not yet
effective accounting pronouncements, if adopted, would have a
material effect on the accompanying financial
statements.
Ricks and Uncertainties
The
Company is subject to substantial business risks and uncertainties
inherent in starting a new business. There is no assurance that the
Company will be able to complete a business
combination.
Basis of Presentation-Going Concern
The accompanying financial statements have been prepared in
conformity with generally accepted accounting principles in the
United States of America, which contemplates continuation of the
Company as a going concern. However, as reflected in the
accompanying financial statements, the Company has no operations, a
net loss of ($30,794) for the year ended December 31, 2016,
and ($17,249) for the nine months
ended September 30, 2017, an accumulated deficit of ($185,831) as
of December 31, 2016 and
($203,080) as of September 30, 2017, and a working capital deficiency of ($41,880) and
($62,629) at December 31, 2016 and September 30, 2017 respectively.
This raises substantial doubt about its ability to continue as a
going concern. The ability of the Company to continue as a going
concern is dependent on the Company's ability to raise additional
capital and implement its business plan. The financial statements
do not include any adjustments that might be necessary if the
Company is unable to continue as a going
concern.
These
financial statements are prepared on a going concern basis because
the Directors, officers and significant shareholders have
undertaken to provide continuing financial support so that the
Company is able to pay its debts as and when they fall due. In
addition, Management’s plans to
ensure the Company continues as a going concern include the
pay-down of outstanding debt of the Company and funding of future
operations using proceeds from the revenues expected to be
generated from the sale of products and services within acquired
companies.
While the Company looks to acquire targeted acquisitions, adverse
changes in market conditions or limits on the Company’s
ability to obtain financing could limit the Company’s
acquisition of such targets and the production, marketing and sale
of products and services of the targeted acquisitions. The impact
of such eventualities could influence future operations of the
Company.
F-10
Income Taxes
The
Company records deferred taxes in accordance with Statement of
Financial Accounting Standards (SFAS) ASC 740, “Accounting
for Income Taxes.” The statement requires recognition of
deferred tax assets and liabilities for temporary differences
between the tax basis of assets and liabilities and the amounts at
which they are carried in the financial statements, the effect of
net operating losses, based upon the enacted tax rates in effect
for the year in which the differences are expected to reverse. A
valuation allowance is established when necessary to reduce
deferred tax assets to the amount expected to be
realized.
Unaudited Financial Statements
The balance sheet as of September 30, 2017, the statements of
operations and cash flows for the nine month periods ended
September 30, 2017 and 2016, and the statement of stockholders'
(deficit) for the nine month period ended September 30, 2017, have
been prepared by the Company without audit, pursuant to the rules
and regulations of the Securities and Exchange Commission. Certain
information and footnote disclosures, normally included in the
financial statements prepared in accordance with accounting
principles generally accepted in the United States of America, have
been condensed or omitted as allowed by such rules and regulations,
and the Company believes that the disclosures are adequate to make
the information presented not misleading. The results of operations
for the nine months ended September 30, 2017 are not necessarily
indicative of results expected for the full year ending December
31, 2017. In the opinion of management, all adjustments (which
include only normal recurring adjustments) necessary to present
fairly the financial position, results of operations and changes in
financial position at September 30, 2017 and for all periods
presented, have been made.
Earnings per Share
Basic
net income (loss) per share attributable to common stockholders is
calculated by dividing the net income (loss) attributable to common
stockholders by the weighted-average number of common shares
outstanding. There were no options or warrants outstanding at
December 31, 2016 nor December 31, 2015. Diluted earnings per share
is not shown for the periods in which the Company incurs a loss
because it would be anti-dilutive.
Equity-Based Compensation
Pursuant
to accounting guidance related to accounting for equity-based
compensation, the Company is required to recognize all share-based
payments to non-employees and employees in the financial statements
based on fair values on the grant date. The Company has accounted
for issuance of shares, options, and warrants in accordance with
the guidance, which requires the recognition of expense, based on
grant-date fair values, over the service period.
Other
The Company has selected December 31 as its fiscal year
end.
The Company has paid no dividends.
No advertising expense has been incurred.
The Company consists of one reportable business
segment.
The Company has not entered into any leases.
NOTE 2-Income Taxes
Deferred
income taxes arise from temporary timing differences in the
recognition of income and expenses for financial reporting and tax
purposes. The Company’s deferred tax assets consist entirely
of the benefit from net operating loss (NOL) carry forwards. The
net operating loss carry forward if not used, will expire in
various years through 2037, and is severely restricted as per the
Internal Revenue code if there is a change in ownership. The
Company’s deferred tax assets are offset by a valuation
allowance due to the uncertainty of the realization of the net
operating loss carry forwards. Net operating loss carry forwards
may be further limited by other provisions of the tax laws. With
few exceptions, our income tax returns are no longer subject to
Federal tax examinations by tax authorities for years before
December 31, 2011.
The
Company is subject to federal and state income tax. No provision
for tax has been made at December 31, 2016, and 2015 as the Company
had no taxable income for both periods. The following table
reconciles the Company’s statutory tax rate to its effective
tax rate as a percentage of income before income
taxes:
F-11
|
|
For the Years
Ended December 31,
|
|
|
|
2016
|
2015
|
|
U.S. statutory
rate
|
34.0%
|
34.0%
|
|
Valuation
allowance
|
-34.0%
|
-34.0%
|
|
Effective tax
rate
|
0.0%
|
0.0%
|
The
components of the Company’s deferred income tax assets are
set forth below:
|
Year Ending
|
Estimated NOL Carry-forward
|
NOL Expires
|
Estimated Tax Benefit from NOL
|
Valuation Allowance
|
Change in Valuation Allowance
|
Net Tax Benefit
|
|
|
|
|
|
|
|
|
|
December 31, 2016
|
$ 129,147
|
Various
|
$ 43,910
|
$ 43,910
|
$ 13,448
|
$ -
|
|
December 31, 2015
|
$ 89,595
|
Various
|
$ 30,462
|
$ 30,462
|
$ 14,443
|
$ -
|
NOTE 3-Related Party Transactions
On
June 27, 2011 the Company entered into a Multiple Advance Demand
Note with The Shapiro Trust, owner of 40% of the outstanding equity
in the Company. The Note is due upon Demand and bears an interest
rate of 8% per annum. As of December 31, 2016 and December 31, 2015
the principal balance on the Note was $18,390 and $12,830,
respectively, and $32,331 as of September 30, 2017. As of the
December 31, 2016 and December 31, 2015 the accrued interest on the
Note was $4,268 and $3,006, respectively and $5,707 as of September
30, 2017. The total principal and interest due The Shapiro Trust as
of December 31, 2016 and December 31, 2015 was $22,658 and $15,836,
respectively, and $38,038 as of September 30, 2017. See Note
4.
On
June 22, 2011 the Company entered into a Trademark License
Agreement with Ann Shapiro, a beneficiary of the Shapiro Trust and
wife of one of the Company’s directors. Ms. Shapiro owns the
Registered Trademark and cosmetic formulas. Under the terms of the
Agreement the Company was granted an exclusive license to sell
“Arrestage Mark and Formula” in exchange for a Royalty
of $50,000 to be paid in three installments. Ms. Shapiro retains
title and ownership of the Mark and Formulas. The Termination Date
of the License is 2025. See Note 3.
NOTE 4-TRADEMARK LICENSE
On June 22, 2011 the Company entered into a Trademark License
Agreement with Ann Shapiro. Ms. Shapiro owns the Registered
Trademark and cosmetic formulas. Under the terms of the Agreement
the Company was granted an exclusive license to sell
“Arrestage Mark and Formula” in exchange for a Royalty
of $50,000 to be paid in three installments. Ms. Shapiro retains
title and ownership of the Mark and Formulas. The Termination Date
of the License is 2025.
F-12
The Trademark License was originally recorded at a cost of $50,000
and tested for impairment in December 2014, at which time it was
determined that the fair value of the License was zero and an
impairment allowance of $50,000 was recorded. See Footnote
1-Intangible Assets.
|
|
2016
|
2015
|
||
|
|
Gross Carrying Amount
|
Impairment Allowance
|
Gross Carrying Amount
|
Impairment Allowance
|
|
|
|
|
|
|
|
Trademark
License
|
50,000
|
(50,000)
|
50,000
|
(50,000)
|
NOTE 5-FINANCING ACTIVITIES
Note Payable-Shapiro
The following is a summary of The Note Payable
|
|
As of September 30, 2017
|
As of December 31, 2016
|
As of December 31, 2015
|
||||||
|
|
Principal
|
Interest payable
|
Total Due Upon Demand
|
Principal
|
Interest payable
|
Total Due Upon Demand
|
Principal
|
Interest payable
|
Total Due Upon Demand
|
|
|
|
|
|
|
|
|
|
|
|
|
Unsecured
Demand Note Payable to The Shapiro Trust, due upon demand. The Note
Payable is a multiple advance demand Note with a stated interest
rate of 8%.
|
32,331
|
5,707
|
38,038
|
18,390
|
4,268
|
22,658
|
12,830
|
3,006
|
15,836
|
NOTE 6. CAPITAL STOCK and STOCKHOLDERS’ EQUITY
Description of Capital Stock--The Company has two classes of
authorized capital stock: Common Stock and Preferred Stock. Both
were authorized June 22, 2011 and both have a par value of $001.
The holders of Common Stock are entitled to one vote per share.
Rights and Preferences for the Preferred Stock will be determined
by the Board of Directors prior to issuance of any Preferred
Stock.
There are 30,000,000 shares of Common Stock authorized. As of
December 31, 2016, and 2015 there were 3,600,000 and 3,561,250
shares of Common Stock issued and outstanding,
respectively.
During the year ended December 31, 2016, the Company issued 38,750
shares of common stock at $1.00 per share for a total of $38,750
cash. During the year ended December 31, 2015, the Company issued
10,000 shares of common stock at $1.00 for services valued at
$10,000.
There are 5,000,000 shares of Preferred Stock authorized and zero
outstanding as of December 31, 2016 and 2015.
NOTE 7-COMMITMENTS AND CONTINGENCIES
In
March, 2017 the Board of Directors of the Company authorized
management to pursue certain merger and acquisition targets and to
pay certain legal, consulting, and diligence fees on behalf of
these entities. Approximately $9,000 has been paid for these legal
fees and due diligence through September 30, 2017. Fees are to be
paid as a sunk cost and expensed by the Company, with no
requirement for, or expectation of, reimbursement. There are no
limits set on the amounts or timing of such payments. A contingency
exists with respect to this matter, the ultimate resolution of
which cannot presently be determined.
NOTE 8 -SUBSEQUENT EVENTS
The Company has evaluated events subsequent to December 31, 2016
and the date the financial statements were available to be issued,
to assess the need for potential recognition or disclosure. No
subsequent events were noted that require recognition or disclosure
in the financial statements.
F-13
THIS PAGE IS INTENTIONALLY LEFT BLANK
PART II: INFORMATION NOT REQUIRED IN THE PROSPECTUS
Item 13. Other expenses of issuance and distribution.
The
following table sets forth all fees and expenses, other than
underwriting discounts and commissions, payable solely by the
registrant in connection with the offer and sale of the securities
being registered. All amounts shown are estimated except for the
registration fee of the SEC, the FINRA filing fee and the OTC
listing fee.
|
SEC registration
fee
|
$3,477
|
|
FINRA filing
fee
|
$N/A
|
|
OTC MKT listing
fee
|
$2,500
|
|
Accounting fees and
expenses
|
$8,500
|
|
Legal fees and
expenses
|
$32,000
|
|
Printing fees and
expenses
|
N/A
|
|
Transfer agent and
registrar fees and expenses
|
$2,000
|
|
Blue sky fees and
expenses
|
$4,000
|
|
Miscellaneous
|
|
|
Total
|
$
|
Item 14. Indemnification of directors and officers.
Chapter 78 of
the Nevada Restated Statutory langue (the “NRS”)
provides that a Nevada corporation may indemnify any person who was
or is a party or is threatened to be made a party to any
threatened, pending or completed action, suit or proceeding,
whether civil, criminal, administrative or investigative, other
than an action by or in the right of the corporation, by reason of
the fact that such person is or was a director, officer, employee
or agent of the corporation, or is or was serving at the request of
the corporation as a director, officer, employee or agent of
another corporation, partnership, joint venture, trust or other
enterprise, against expenses (including attorneys’ fees),
judgments, fines and amounts paid in settlement actually and
reasonably incurred by the person in connection with such action,
suit or proceeding if the person acted in good faith and in a
manner the person reasonably believed to be in or not opposed to
the best interests of the corporation, and, with respect to any
criminal action or proceeding, had no reasonable cause to believe
the person’s conduct was unlawful. Except as other wise
provided in NRS, except as:
35.23,
90.660, 91.250, 452.200, 452.270, 668.045 and 694A.030,
or unless the articles of
incorporation or an amendment thereto, in each case filed on or
after October 1, 2003, provide for greater individual liability, a
director or officer is not individually liable to the corporation
or its stockholders or creditors for any damages as a result of any
act or failure to act in his or her capacity as a director or
officer unless it is proven that:
II-1
(a) The director’s or
officer’s act or failure to act constituted a breach of his
or her fiduciary duties as a director or officer; and
(b) The breach of those duties
involved intentional misconduct, fraud or a knowing violation of
law.
(Added to NRS by 1991, 1184;
A 1993,
951; 1999,
1580; 2001,
3171; 2003,
3084)
NRS
provides that a Nevada corporation may indemnify any person who was
or is a party or is threatened to be made a party to any
threatened, pending or completed action or suit by or in the right
of the corporation to procure a judgment in its favor by reason of
the fact that such person acted in any of the capacities set forth
above, against expenses (including attorneys’ fees) actually
and reasonably incurred by such person in connection with the
defense or settlement of such action or suit if the person acted in
good faith and in a manner the person reasonably believed to be in
or not opposed to the best interests of the corporation, except
that no indemnification shall be made in respect of any claim,
issue or matter as to which such person shall have been adjudged to
be liable to the corporation, unless and only to the extent that
the court in which such action or suit was brought shall determine
upon application that, despite the adjudication of liability but in
view of all the circumstances of the case, such person is fairly
and reasonably entitled to indemnity for such expenses which the
court shall deem proper.
The NRS
makes provision for the indemnification of officers and directors
in terms sufficiently broad to indemnify officers and directors of
each of the registrants incorporated in Nevada under certain
circumstances from liabilities (including reimbursement for
expenses incurred) arising under the Securities Act of 1933, as
amended (the “Act”). Arrestage International, Inc. may,
in its discretion, similarly indemnify its employees and
agents.
The
certificate of incorporation and bylaws of Arrestage International,
Inc. provide that, to the fullest extent and under the
circumstances permitted by chapter 78 of the NRS, Arrestage
International, Inc. will indemnify directors and officers from and
against any and all of the expenses, liabilities or other maters as
adopted by the NRS. In addition, the certificate of incorporation
of Arrestage International, Inc. relieves its directors from
monetary damages to it or its stockholders for breach of such
director’s fiduciary duty as a director to the fullest extent
permitted by the NRS.
Under
Chapter 78 of the NRS the directors and officers of the Company
shall be indemnified pursuant to rules and regulations set for in
the revised statute:
NRS 78.138 Directors and officers: Exercise of
powers; performance of duties; presumptions and considerations;
liability to corporation and stockholders.
1. Directors and
officers shall exercise their powers in good faith and with a view
to the interests of the corporation.
2. In performing their
respective duties, directors and officers are entitled to rely on
information, opinions, reports, books of account or statements,
including financial statements and other financial data, that are
prepared or presented by:
II-2
(a) One or more directors,
officers or employees of the corporation reasonably believed to be
reliable and competent in the matters prepared or
presented;
(b) Counsel, public
accountants, financial advisers, valuation advisers, investment
bankers or other persons as to matters reasonably believed to be
within the preparer’s or presenter’s professional or
expert competence; or
(c) A committee on which the
director or officer relying thereon does not serve, established in
accordance with NRS
78.125, as to matters within
the committee’s designated authority and matters on which the
committee is reasonably believed to merit
confidence,
but a director or officer is not entitled to rely
on such information, opinions, reports, books of account or
statements if the director or officer has knowledge concerning the
matter in question that would cause reliance thereon to be
unwarranted.
3. Directors and
officers, in deciding upon matters of business, are presumed to act
in good faith, on an informed basis and with a view to the
interests of the corporation.
4. Directors and
officers, in exercising their respective powers with a view to the
interests of the corporation, may consider:
(a) The interests of the
corporation’s employees, suppliers, creditors and
customers;
(b) The economy of the State
and Nation;
(c) The interests of the
community and of society; and
(d) The long-term as well as
short-term interests of the corporation and its stockholders,
including the possibility that these interests may be best served
by the continued independence of the corporation.
5. Directors and
officers are not required to consider the effect of a proposed
corporate action upon any particular group having an interest in
the corporation as a dominant factor.
6. The provisions of
subsections 4 and 5 do not create or authorize any causes of action
against the corporation or its directors or officers.
7. Except as otherwise
provided in NRS
35.230, 90.660, 91.250, 452.200, 452.270, 668.045 and 694A.030,
or unless the articles of
incorporation or an amendment thereto, in each case filed on or
after October 1, 2003, provide for greater individual liability, a
director or officer is not individually liable to the corporation
or its stockholders or creditors for any damages as a result of any
act or failure to act in his or her capacity as a director or
officer unless it is proven that:
(a) The director’s or
officer’s act or failure to act constituted a breach of his
or her fiduciary duties as a director or officer; and
(b) The breach of those duties
involved intentional misconduct, fraud or a knowing violation of
law.
(Added to NRS
by 1991, 1184;
A 1993,
951; 1999,
1580; 2001,
3171; 2003,
3084)
Arrestage
International, Inc. also intends to purchase and maintain insurance
on behalf of any person who is or was a director or officer against
any loss arising from any claim asserted against him or her and
incurred by him or her in any such capacity, subject to certain
exclusions.
II-3
Item 15. Recent sales of unregistered securities.
The
following list sets forth information as to all securities we have
sold or exchanged since June 1, 2012, which were not registered
under the Securities Act.
Since
June, 2012, we have issued Shares in the following
transactions:
(2)
Issuance of the following shares
were made pursuant to a private placement dated June, 2016 (in
depth description of the offering.)
|
|
|
|
After this offering
(1)
|
|||
|
|
Prior to this offering
|
Assuming underwriters' option to purchase additional shares is
not exercised
|
Assuming
underwriters' option to purchase additional shares is exercised in
full
|
|||
|
|
Number of shares beneficially owned
|
Number of shares beneficially owned
|
Number of shares beneficially owned
|
|||
|
Name
|
Number of
shares
|
Percentage of shares
|
Number of
shares
|
Percentage of shares
|
Number of
shares
|
Percentage of shares
|
|
Plum Mt
Trust
|
1500
|
%
|
|
%
|
|
%
|
|
Tod
Wright
|
1500
|
%
|
|
%
|
|
%
|
|
Randy
Willard
|
2500
|
|
|
|
|
|
|
Nicolas
White
|
1500
|
%
|
|
%
|
|
%
|
|
Chad
Mekay
|
500
|
%
|
|
%
|
|
%
|
|
Darcy
Huish
|
1500
|
%
|
|
%
|
|
%
|
|
Roy
Shapiro
|
500
|
*
|
|
%
|
|
%
|
|
Daniel
Shapiro
|
500
|
%
|
|
%
|
|
%
|
|
M Brent
Stapley
|
1500
|
%
|
|
%
|
|
%
|
|
Karl
Huish
|
1500
|
|
|
|
|
|
|
Nina
Richardon
|
1500
|
|
|
|
|
|
|
David
Richardon
|
1500
|
|
|
|
|
|
|
Robert Chris
Newsome
|
500
|
|
|
|
|
|
|
Charley
Ingrao
|
750
|
|
|
|
|
|
|
Doug
Ingrao
|
500
|
|
|
|
|
|
|
Nathan
Ingrao
|
1500
|
|
|
|
|
|
|
Dennis
Miller
|
1500
|
|
|
|
|
|
|
Catherine
Miller
|
1500
|
|
|
|
|
|
|
Megan
Miller
|
500
|
|
|
|
|
|
|
Caitilin
Miller
|
500
|
|
|
|
|
|
|
Tarran
Merrill
|
2500
|
|
|
|
|
|
|
Ivy L
Kushner
|
1500
|
|
|
|
|
|
|
Alex
Campbell
|
1500
|
|
|
|
|
|
|
Krista
Finn
|
1500
|
|
|
|
|
|
|
Chris
Finn
|
1500
|
|
|
|
|
|
|
Joshua
Chapman
|
500
|
|
|
|
|
|
|
Neville Living
Trust
|
500
|
|
|
|
|
|
|
Robert
Neville
|
500
|
|
|
|
|
|
|
Roger
Mentch
|
1500
|
|
|
|
|
|
|
Andrew
Mentch
|
1500
|
|
|
|
|
|
|
Allison& Joshua
Lumadue
|
1000
|
|
|
|
|
|
|
Demetri
Panayi
|
500
|
|
|
|
|
|
|
Lucy
Panayi
|
500
|
|
|
|
|
|
|
Janae
Panayi
|
500
|
|
|
|
|
|
|
Sophia
Panayi
|
500
|
|
|
|
|
|
|
Veronica
Baker
|
250
|
|
|
|
|
|
|
Gary
Baker
|
25000
|
|
|
|
|
|
|
Richard W
Clement
|
25000
|
|
|
|
|
|
|
Emmett
Jobe
|
30000
|
|
|
|
|
|
|
Larry
Barken
|
25000
|
|
|
|
|
|
II-4
(2)
Above
table shows since June 1, 2012, we have issued Shares in the
following transactions to certain of our employee.
We
claimed exemption from registration under the Securities Act for
the sale and issuance of securities in the transactions described
in paragraph (1) by virtue of Section 4(a)(2) and Regulation D
promulgated thereunder as transactions not involving any public
offering. All of the purchasers of unregistered securities for
which we relied on Section 4(a)(2) and Regulation D represented
that they were accredited investors as defined under the Securities
Act. We claimed such exemption on the basis that (a) the purchasers
in each case represented that they intended to acquire the
securities for investment only and not with a view to the
distribution thereof and that they either received adequate
information about the registrant or had access, through employment
or other relationships, to such information and (b) appropriate
legends were affixed to the unit certificates issued in such
transactions.
We
claimed exemption from registration under the Securities Act for
the sales and issuances of securities in the transactions described
in paragraph (2) above under Section 4(a)(2) of the Securities Act
in that such sales and issuances did not involve a public offering
or under Rule 701 promulgated under the Securities Act, in that
they were offered and sold either pursuant to written compensatory
plans or pursuant to a written contract relating to compensation,
as provided by Rule 701.
II-5
Item 16. Exhibits and financial statement schedules.
(a) The
exhibits listed below in the “Index to exhibits” are
part of this Registration Statement on Form S-1 and are numbered in
accordance with Item 601 of Regulation S-K.
(b)
Financial statement schedules. Schedules have been omitted because
the information required to be set forth therein is not applicable
or is shown in the financial statements or notes
thereto.
Item 17. Undertakings.
The
undersigned registrant hereby undertakes that:
(1) For
purposes of determining any liability under the Securities Act of
1933, the information omitted from the form of prospectus filed as
part of this Registration Statement in reliance upon Rule 430A and
contained in a form of prospectus filed by the Registrant pursuant
to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall
be deemed to be part of this Registration Statement as of the time
it was declared effective.
(2) For
the purpose of determining any liability under the Securities Act
of 1933, each post-effective amendment that contains a form of
prospectus shall be deemed to be a new Registration Statement
relating to the securities offered therein, and the offering of
such securities at that time shall be deemed to be the initial
bona fide offering
thereof.
(3) The
undersigned will provide to the underwriters, at the closing
specified in the underwriting agreement, certificates in such
denominations and registered in such names as required by the
underwriters to permit prompt delivery to each
purchaser.
Insofar
as indemnification for liabilities arising under the Securities Act
of 1933 may be permitted to directors, officers and controlling
persons of the registrant pursuant to the foregoing provisions, or
otherwise, the registrant has been advised that in the opinion of
the SEC such indemnification is against public policy as expressed
in the Act and is, therefore, unenforceable. In the event that a
claim for indemnification against such liabilities (other than the
payment by the registrant of expenses incurred or paid by a
director, officer or controlling person of the registrant in the
successful defense of any action, suit or proceeding) is asserted
by such director, officer or controlling person in connection with
the securities being registered, the registrant will, unless in the
opinion of its counsel the matter has been settled by controlling
precedent, submit to a court of appropriate jurisdiction the
question whether such indemnification by it is against public
policy as expressed in the Act and will be governed by the final
adjudication of such issue.
II-6
SIGNATURES
Pursuant to the
requirements of the Securities Act of 1933, the registrant has duly
caused this Registration Statement to be signed on its behalf by
the undersigned, thereunto duly authorized, in the City of Phoenix,
State of Arizona, February 22,
2018.
|
|
Arrestage International, Inc. |
|
|
|
|
|
|
|
|
|
By:
|
/s/ Mr. Gary
Croft
|
|
|
|
|
Mr. Gary
Croft
|
|
|
|
|
Chief Executive Officer
|
|
POWER OF ATTORNEY
Each of
the undersigned officers and directors of Arrestage International,
Inc. hereby constitutes and appoints Mr. Gary Croft and Dr. Roy
Shapiro, each of them, his true and lawful attorneys-in-fact and
agents, each with full power of substitution and re-substitution,
for him and in his name, place and stead, in any and all
capacities, to sign the Registration Statement of Arrestage
International, Inc. on Form S-1, and any other registration
statement relating to the same offering (including any registration
statement, or amendment thereto, that is to become effective upon
filing pursuant to Rule 462(b) under the Securities Act of 1933),
and any and all amendments thereto (including post-effective
amendments), and to file the same, with all exhibits thereto, and
other documents in connection therewith, with the Securities and
Exchange Commission, granting unto said attorneys-in-fact and
agents, and each of them, full power and authority to do and
perform each and every act and thing requisite or necessary to be
done in connection therewith, as fully to all intents and purposes
as he might or could do in person, hereby ratifying and confirming
all that each of said attorneys-in-fact and agents, or his
substitute or substitutes, may lawfully do or cause to be done by
virtue hereof.
Pursuant to the
requirements of the Securities Act of 1933, this Registration
Statement has been signed by the following persons in the
capacities set forth opposite their names and on the date indicated
above.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/ Mr.
Gary Croft
|
|
Chief
Executive Officer and Director (Principal Executive
Officer)
|
|
February
22, 2018
|
|
|
|
|
|
|
|
/s/ Mr.
Rick Gean
|
|
Chief
Financial Officer (Principal Financial Officer and Principal
Accounting Officer)
|
|
February 22,
2018
|
|
|
|
|
|
|
|
/s/ Dr.
Roy Shapiro
|
|
Director
|
|
February 22,
2018
|
|
|
|
|
|
|
| /s/ Mr. Phillip Weisman |
|
Director |
|
February 22, 2018 |
|
|
|
|||
| /s/ Mr. Philip Nuciola III |
|
Director |
|
February 22, 2018 |
|
|
|
|||
|
/s/ Ms.
Kimberly Shapiro
|
|
Director
|
|
February 22,
2018
|
II-7
INDEX TO EXHIBITS
|
Exhibit
No.
|
|
Description
|
|
|
Form of
Amended and Restated Certificate of Incorporation of Arrestage
International, Inc.
|
|
|
|
Certificate
of Designation of Class Preferred Shares
(5,000,000)
|
|
|
|
Bylaws
of Arrestage International, Inc., effective as of June 12,
2012.
|
|
|
|
Amendment
to Bylaws by Board Resolution Approving New Class of Preferred
Shares
|
|
|
|
Form of
Common Stock Certificate of Arrestage International,
Inc.
|
|
|
|
Consent
of Counsel: Erica Loren Jozwiak, Esq.
|
|
|
|
Form of
Indemnification Agreement.
|
|
|
|
Arrestage
Internation. Inc/ Arrestage Laboratory Corporation Exclusive
Licensing Agreement
|
|
|
|
Arrestage International, Inc. Code of Ethics Charter. | |
|
|
Arrestage International, Inc. Compensation Committee Charter | |
|
|
Arrestage
International, Inc. Audit Committee Charter
|
|
Exhibit
No.
|
|
Description
|
|
|
Consent
of Schumacher and Associates, Inc.
|
|
|
|
Consent
of Attorney; Erica Loren Jozwiak, Esq. (included in exhibit
5)
|
|
|
|
Power
of Attorney — included on the signature page
hereto.
|
*
To be
filed by amendment
+
Indicates a
management contract or compensatory plan
