Attached files
p
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
|
☑ |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2017
OR
|
☐ |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File No. 001-36276
Ultragenyx Pharmaceutical Inc.
(Exact name of registrant as specified in its charter)
|
Delaware |
27-2546083 |
|
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
|
60 Leveroni Court Novato, California |
94949 |
|
(Address of principal executive offices) |
(Zip Code) |
(415) 483-8800
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
|
Title of Each Class |
|
Name of Each Exchange on Which Registered |
|
Common Stock, $0.001 par value |
|
The Nasdaq Global Select Market |
Securities registered pursuant to Section 12(g) of the Act: None.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. YES ☑ NO ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. YES ☐ NO ☑
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. YES ☑ NO ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). YES ☑ NO ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ☑
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
Large accelerated filer ☑Accelerated filer ☐
Non- accelerated filer ☐ (Do not check if a smaller reporting company) Smaller reporting company ☐
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). YES ☐ NO ☑
The aggregate market value of the voting and non-voting common equity held by non-affiliates of the Company as of June 30, 2017 was approximately $2.6 billion, based upon the closing price on The Nasdaq Global Select Market reported for such date. Shares of common stock held by each executive officer and director and by each person who is known to own 10% or more of the outstanding common stock have been excluded in that such persons may be deemed to be affiliates of the Company. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of February 15, 2018, the Company had 49,605,852 shares of common stock issued and outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive proxy statement relating to its 2018 Annual Meeting of Stockholders, to be held on or about June 19, 2018, are incorporated by reference into Part III of this Annual Report on Form 10-K where indicated. Such proxy statement will be filed with the U.S. Securities and Exchange Commission within 120 days after the end of the fiscal year to which this report relates.
|
|
|
|
Page |
|
|
|
|
|
|
Item 1. |
|
4 |
|
|
Item 1A. |
|
28 |
|
|
Item 1B. |
|
60 |
|
|
Item 2. |
|
60 |
|
|
Item 3. |
|
61 |
|
|
Item 4. |
|
61 |
|
|
|
|
|
|
|
Item 5. |
|
62 |
|
|
Item 6. |
|
63 |
|
|
Item 7. |
|
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
64 |
|
Item 7A. |
|
76 |
|
|
Item 8. |
|
76 |
|
|
Item 9. |
|
Changes in and Disagreements With Accountants on Accounting and Financial Disclosure |
76 |
|
Item 9A. |
|
77 |
|
|
Item 9B. |
|
78 |
|
|
|
|
|
|
|
Item 10. |
|
79 |
|
|
Item 11. |
|
79 |
|
|
Item 12. |
|
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
79 |
|
Item 13. |
|
Certain Relationships and Related Transactions, and Director Independence |
79 |
|
Item 14. |
|
79 |
|
|
|
|
|
|
|
Item 15. |
|
80 |
|
|
Item 16. |
|
85 |
|
|
|
86 |
||
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K, or Annual Report, contains forward-looking statements that involve risks and uncertainties. We make such forward-looking statements pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. All statements other than statements of historical facts contained in this Annual Report are forward-looking statements. In some cases, you can identify forward-looking statements by words such as “anticipate,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “seek,” “should,” “target,” “will,” “would,” or the negative of these words, or other comparable terminology. These forward-looking statements include, but are not limited to, statements about:
|
|
• |
our commercialization, marketing, and manufacturing capabilities and strategy; |
|
|
• |
our expectations regarding the timing of clinical study commencements and reporting results from same; |
|
|
• |
the timing and likelihood of regulatory approvals for our product candidates; |
|
|
• |
the anticipated indications for our product candidates, if approved; |
|
|
• |
the potential market opportunities for commercializing our product and product candidates; |
|
|
• |
our expectations regarding the potential market size and the size of the patient populations for our product and product candidates, if approved for commercial use; |
|
|
• |
estimates of our expenses, future revenue, capital requirements, and our needs for additional financing; |
|
|
• |
our ability to develop, acquire, and advance product candidates into, and successfully complete, clinical studies; |
|
|
• |
the implementation of our business model and strategic plans for our business, product and product candidates and the integration and performance of any acquired businesses; |
|
|
• |
the initiation, timing, progress, and results of ongoing and future preclinical and clinical studies, and our research and development programs; |
|
|
• |
the scope of protection we are able to establish and maintain for intellectual property rights covering our product and product candidates; |
|
|
• |
our ability to maintain and establish collaborations or strategic relationships or obtain additional funding; |
|
|
• |
our ability to maintain and establish relationships with third parties, such as contract research organizations, suppliers, and distributors; |
|
|
• |
our financial performance and the expansion of our organization; |
|
|
• |
our ability to obtain supply of our product and product candidates; |
|
|
• |
the scalability and commercial viability of our manufacturing methods and processes; |
|
|
• |
developments and projections relating to our competitors and our industry; and |
|
|
• |
other risks and uncertainties, including those listed under Part I, Item 1A. Risk Factors. |
Any forward-looking statements in this Annual Report reflect our current views with respect to future events or to our future financial performance and involve known and unknown risks, uncertainties, and other factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by these forward-looking statements. Factors that may cause actual results to differ materially from current expectations include, among other things, those discussed under Part I, Item 1A. Risk Factors and discussed elsewhere in this Annual Report. Given these uncertainties, you should not place undue reliance on these forward-looking statements. Except as required by law, we assume no obligation to update or revise these forward-looking statements for any reason, even if new information becomes available in the future.
This Annual Report also contains estimates, projections, and other information concerning our industry, our business, and the markets for certain diseases, including data regarding the estimated size of those markets, and the incidence and prevalence of certain medical conditions. Information that is based on estimates, forecasts, projections, market research, or similar methodologies is inherently subject to uncertainties and actual events or circumstances may differ materially from events and circumstances reflected in this information. Unless otherwise expressly stated, we obtained this industry, business, market, and other data from reports, research surveys, studies, and similar data prepared by market research firms and other third parties, industry, medical and general publications, government data, and similar sources.
As used in this Annual Report, “Ultragenyx,” “we,” “our,” and similar terms include Ultragenyx Pharmaceutical Inc. and its subsidiaries, unless the context indicates otherwise.
3
Overview
We are a biopharmaceutical company focused on the identification, acquisition, development, and commercialization of novel products for the treatment of serious rare and ultra-rare diseases, with a focus on serious, debilitating genetic diseases. We target diseases for which the unmet medical need is high, the biology for treatment is clear, and for which there are no currently approved therapies. Since our inception in 2010, we have in-licensed potential treatments for multiple rare genetic disorders. Our strategy, which is predicated upon time- and cost-efficient drug development, allows us to pursue multiple programs in parallel with the goal of delivering safe and effective therapies to patients with the utmost urgency.
Our approved product and current product candidate pipeline have been either in-licensed from academic institutions or derived from partnerships with other pharmaceutical companies. Our strategy is to acquire and retain global commercialization rights to our products to maximize long-term value, where possible. We have built our own commercial organization, which is highly targeted due to the relatively small number of specialists who treat patients with rare and ultra-rare diseases.
The patients we seek to treat have diseases with limited or no treatment options, and we recognize that their lives and well-being are dependent upon our efforts to develop new therapies. For this reason, we are passionate about developing these therapies with the utmost urgency and care.
We were founded in April 2010 by our current President and Chief Executive Officer, Emil Kakkis, M.D., Ph.D. We have assembled an experienced team with extensive rare disease drug development and commercialization capabilities. Dr. Kakkis and the team at Ultragenyx have been previously involved at other companies in the development and/or commercialization of many therapies approved or in development for rare genetic diseases.
On November 7, 2017, we completed our acquisition of Dimension Therapeutics, Inc., a Delaware corporation, which became our wholly-owned subsidiary. Upon the closing of the acquisition, we paid aggregate consideration of approximately $152.3 million, not including related transaction fees and expenses, using available cash and investments. For additional information regarding the acquisition, see Note 3 to our consolidated financial statements.
Our Strategy
Our strategy is to identify, acquire, develop, and commercialize novel products for the treatment of rare and ultra-rare diseases in the United States, or U.S., the European Union, or EU, and select international markets, with the goal of becoming a leading rare disease biotechnology company. The critical components of our business strategy include the following:
|
|
• |
Focus on rare and ultra-rare diseases with significant unmet medical need and clear biology. There are numerous rare and ultra-rare genetic diseases that currently have no approved drug therapy and for which no therapies are currently in development. Patients suffering from these diseases often have a high unmet medical need with significant morbidity and/or mortality. We are focused on developing and commercializing therapies for multiple such indications with the utmost urgency. We also focus on diseases that have biology that is well understood. For example, MepseviiTM is a replacement therapy for a single deficient enzyme and several of our product candidates are replacement therapies for a single deficient substrate in the body. We believe that developing drugs that directly impact known disease pathways will increase the probability of success of our development programs. Our four modalities of small molecules, biologics, mRNA and AAV gene therapy provide us with what we believe is an optimal set of options to treat metabolic genetic diseases by selecting the best treatment strategy available for each disease. |
|
|
• |
Leverage our experience and relationships to in-license promising product candidates; retain global commercialization rights to product candidates. Our management team seeks to develop and maintain strong relationships with key opinion leaders in the genetic field and leverage our success in the development and commercialization of therapies for rare and ultra-rare genetic diseases. All of our current clinical product candidates are in-licensed from academic institutions or derived from partnerships with other pharmaceutical companies. We believe parties agree to license product candidates to us because they are confident in our team’s drug development capabilities and our plans and execution thus far in bringing rare disease therapies to market. We intend to seek and retain global commercialization rights to our product and product candidates whenever possible to maximize the potential value of our product portfolio. Because we typically in-license product candidates that require translational or clinical research, at this time we do not intend to invest significant capital in basic research, which can be expensive and time-consuming. |
4
|
|
• |
Establish global commercial organization. We have established our own unique commercial organization in major pharmaceutical markets and developed a network of third-party distributors in smaller markets and expect to expand these efforts. We believe our commercial organization is highly targeted, in part as a result of the relatively small number of specialists who typically treat patients with the diseases to be addressed by our product and product candidates. |
Product - MepseviiTM
On November 15, 2017, the U.S. Food and Drug Administration, or FDA, approved our first product, Mepsevii (vestronidase alfa), the first medicine approved for the treatment of children and adults with MPS VII, also known as Sly syndrome. Mepsevii is available to patients in the U.S. In order to support patients, we launched UltraCare™, a comprehensive support service that provides ongoing support to patients and caregivers. UltraCare will help patients obtain coverage and assist with financial support for both medication and administration of medication. With this approval, the FDA issued a Rare Pediatric Disease Priority Review Voucher, or PRV, which confers priority review to a subsequent drug application that would not otherwise qualify for priority review. We completed the sale of the PRV in January 2018 for $130.0 million.
Mepsevii is an intravenous, or IV, enzyme replacement therapy for the treatment of MPS VII. MPS VII is caused by a deficiency of the lysosomal enzyme beta-glucuronidase, which is required for the breakdown of certain complex carbohydrates known as glycosaminoglycans, or GAGs. The inability to properly break down GAGs leads to their accumulation in many tissues, resulting in a serious multi-system disease. Mepsevii is designed to replace the deficient lysosomal enzyme beta-glucuronidase in MPS VII patients. Patients with MPS VII suffer from severe cellular and organ dysfunction that typically leads to death in the teens or early adulthood and may have abnormal coarsened facial features, enlargement of the liver and spleen, airway obstruction, lung disease, cardiovascular complications, joint stiffness, short stature, and a skeletal disease known as dysostosis multiplex. In addition, many patients experience progressive lung problems as a result of airway obstruction and mucous production, often leading to sleep apnea and pulmonary insufficiency, and eventually requiring tracheostomy. Mepsevii is the only FDA-approved drug therapy for MPS VII.
In Europe, the European Medicines Agency, or EMA, is currently reviewing the Marketing Authorization Application, or MAA, for vestronidase alfa, and an opinion from the Committee for Medicinal Products for Human Use, or CHMP, is expected in the first half of 2018. The EMA has agreed that approval under exceptional circumstances could be possible based upon a single positive placebo-controlled pivotal study in approximately 12 patients using urinary GAG levels as a surrogate primary endpoint, provided the data was strongly supportive of a favorable benefit/risk ratio and some evidence or trend in improvement in clinical endpoints was observed to support the primary endpoint. The EMA recognized that a statistically significant result on clinical endpoints was unlikely given the small number of patients expected to be enrolled in the study.
We are also supplying vestronidase alfa to investigators who are treating patients under emergency investigational new drug, or eIND, applications and other expanded access programs.
Please see “—License and Collaboration Agreements—Approved Product—Saint Louis University” for a description of our license agreement with Saint Louis University.
Clinical Product Candidates
Our current clinical-stage pipeline consists of three product categories: biologics, small-molecule substrate replacement therapies, and gene therapy. Enzymes are proteins that the body uses to process materials needed for normal cellular function, and substrates are the materials upon which enzymes act. When enzymes or substrates are missing, the body is unable to perform its normal cellular functions, often leading to significant clinical disease. Several of our therapies are intended to replace deficient enzymes or substrates. Gene therapy is a therapeutic approach in which an isolated gene sequence or segment of DNA is administered to a patient, most commonly for the purpose of treating a genetic disease that is caused by mutations. Gene therapy aims to address the disease-causing effects of absent or dysfunctional genes by delivering functional copies of the gene to the patient’s cells, offering the potential for durable therapeutic benefit.
5
The following table summarizes our advanced product candidate pipeline:
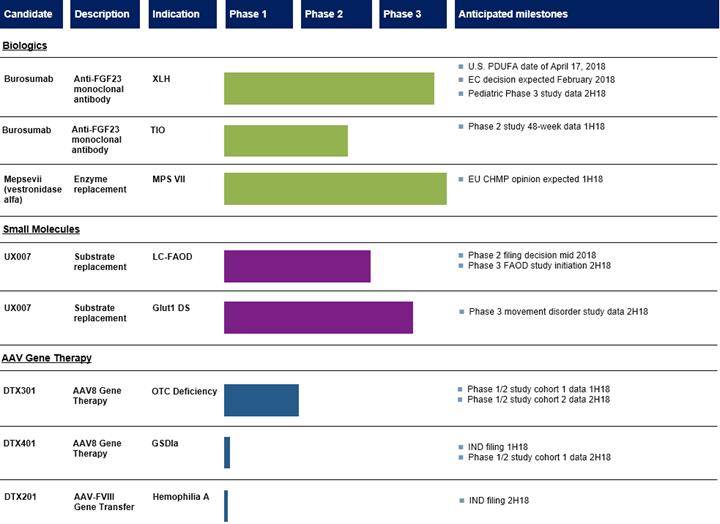
Burosumab for the treatment of XLH
Burosumab is a fully human monoclonal antibody administered via subcutaneous injection that is designed to bind and reduce the biological activity of fibroblast growth factor 23, or FGF23, to increase abnormally low phosphate levels in patients with XLH. Patients with XLH have low serum phosphate levels due to excessive phosphate loss into the urine, which is directly caused by the effect on kidney function of excess FGF23 production in bone cells. Low phosphate levels lead to poor bone mineralization and a variety of clinical manifestations, including rickets leading to bowing and other skeletal deformities, short stature, bone pain and fractures, and muscle weakness. There is no approved drug therapy or treatment for the underlying cause of XLH. Most patients are managed using frequently dosed oral phosphate replacement and vitamin D therapy, which can lead to significant side effects. Oral phosphate/vitamin D replacement therapy requires extremely close monitoring due to the potential for excessive phosphate levels and secondary increases in calcium, which can result in severe damage to the kidneys from excess calcium phosphate deposits and other complications. Additionally, some patients are unable to tolerate the regimen due to the chalky stool that results from taking large amounts of oral phosphate or the high frequency of dosing required.
In April 2017, we announced positive 64-week data from a 52-patient pediatric Phase 2 randomized, multicenter, open-label, dose-finding study of burosumab for the treatment of XLH in children aged five to 12 years of age. Patients demonstrated increases in mean serum phosphorus, renal phosphate reabsorption (TmP/GFR) and serum 1,25 dihydroxy vitamin D levels through 64 weeks of treatment. Rickets severity was assessed using the RSS scoring system. There was a statistically significant improvement in rickets scores in all groups at 64 weeks, with the greatest improvements in patients with higher baseline rickets scores (RSS ≥1.5) who received bi-weekly dosing of burosumab. Overall, patients (n=52) had a 51% reduction in RSS score (p < 0.0001). Patients with higher baseline rickets scores (n=34) had a 59% reduction in RSS score (p < 0.0001). Patients who were dosed bi-weekly (n=26) had a 58% reduction in RSS score (p < 0.0001). Patients with higher baseline rickets scores who were dosed bi-weekly (n=17) had a 62% reduction in RSS score (p < 0.0001). The change in the severity of rickets was assessed by the RGI-C score. Data show significant improvement in rickets in all groups at 64 weeks. Overall, all patients (n=52) experienced a mean improvement in RGI-C score of
6
+1.57 (p< 0. 0001) and those patients with higher baseline rickets scores (n=34) experienced a mean improvement of +1.98 (p< 0.0001). Within the higher severity subset, 77% (26/34) experienced substantial healing (score > 2). Overall, all patients who were dosed bi-weekly (n=26) experienced a mean improvement in RGI-C score of +1.62 (p< 0.0001). Patients with higher baseline rickets scores who were dosed bi-weekly (n=17) showed a mean improvement of +2.08 (p< 0.0001) (substantial healing), and 82% experienced substantial healing (score > 2). Patients with higher baseline rickets scores showed more growth impairment (baseline height percentile= 5.84), and these patients demonstrated greater improvement in growth. Among all patients (n=52), growth velocity improved by a mean of +0.55 cm/year (p=0.0376), and there was 0.15 change in height z-score (p<0.0001). Patients with higher baseline rickets scores had a +0.86 cm/year improvement in growth velocity (p=0.0175) and a 0.17 change in height z-score (p=0.0016). Patients who were dosed bi-weekly (n=26) experienced a +0.73 cm/year change in growth velocity (p=0.0160) and a 0.18 change in height z-score (p=0.0002). Patients with higher baseline rickets scores who were dosed bi-weekly (n=17) had a +1.11 cm/year change in growth velocity (p=0.0076) and a 0.18 change in height z-score (p=0.0063). Approximately 65% of patients had injection site reactions, all of which were considered mild. There was one previously reported serious adverse event considered possibly treatment-related. This was a patient with fever and muscle pain who improved without complication and is still in the study. There have been no deaths or discontinuations from the study. No clinically meaningful changes were observed in mean serum calcium, urinary calcium and in serum intact parathyroid hormone. None of the patients had serum phosphorus levels above the upper limit of normal at any time point. No clinically significant changes were observed in renal ultrasounds pre- and post-treatment.
In September 2017 we announced 40-week data from the 64-week Phase 2 study in children less than five years old (mean age 2.9 years). Burosumab increased mean serum phosphorus levels by 1.2 mg/dL into the low normal range after one week of treatment and these levels were maintained through week 40 with 77% of children achieving normal serum phosphorus levels at week 40. Serum 1,25 dihydroxy vitamin D levels were also increased from baseline to week 40. Rickets severity was assessed using the RSS scoring system. The mean total RSS score improved significantly (59% reduction) at week 40 (p<0.0001). The change in rickets severity was also assessed at week 40 by the RGI-C score which showed substantial healing (RGIC score > 2) in all patients (p<0.0001). Burosumab treatment also resulted in significantly improved bowing as determined by RGI-C lower limb deformity (p<0.0001). Additionally, mean levels of alkaline phosphatase were significantly reduced (-39%, p<0.0001) in these patients at week 40. All patients experienced one or more adverse events. There was one serious adverse event of a tooth abscess that was considered unrelated to burosumab treatment. All other events were assessed as mild or moderate in severity except for a Grade 3 food allergy that was considered unrelated to burosumab treatment. Three patients had injection site reactions and four patients experienced hypersensitivity events that were all mild and considered unrelated to burosumab treatment. No clinically meaningful changes were observed in mean serum calcium, urinary calcium and in serum intact parathyroid hormone. There have been no events of hyperphosphatemia and there have been no deaths or discontinuations from the study.
In February 2018, we reported continued improvement in rickets and bowing in 64 week data from this Phase 2 study. This longer term data from this study demonstrated that treatment with burosumab was consistent with and further improved from what was seen at 40 weeks. These included sustained improvements in serum phosphorus levels, and a progressive reduction into the normal range of alkaline phosphatase. There were continued improvements in bowing and rickets scores at 64 weeks. The safety profile observed in this study was consistent with other burosumab studies.
In December 2017, we and our partners Kyowa Kirin International PLC, a wholly owned subsidiary of KHK, announced that the Committee for Medicinal Products for Human Use (CHMP), the European Medicines Agency's (EMA) scientific committee, adopted a Positive Opinion recommending the conditional marketing authorization of burosumab for the treatment of XLH with radiographic evidence of bone disease in children one year of age and older and adolescents with growing skeletons. The CHMP's recommendation has been referred to the European Commission (EC), which is expected to render its final decision in February of 2018.
We have an ongoing Phase 3 randomized open-label clinical study comparing the efficacy and safety of burosumab to oral phosphate and active vitamin D therapy in approximately 60 pediatric patients with XLH. The study is evaluating changes in rickets, growth velocity and height, pharmacodynamic assessments, walking ability, patient reported outcomes assessing pain, fatigue and physical function, and safety. We expect data from this study in the second half of 2018. This study will not be required to support a US approval and will serve as a confirmatory study in Europe.
We are also continuing to develop burosumab in adults with XLH. In April 2017, we announced positive 24-week data from the randomized, double-blind, placebo-controlled Phase 3 study of burosumab in adults with X-linked hypophosphatemia (XLH). The study enrolled 134 patients, randomized 1:1 to burosumab at a dose of 1 mg/kg or placebo every four weeks for 24 weeks. The study met the primary endpoint of increasing serum phosphorus levels as 94% of patients treated with burosumab (n=68) achieved serum phosphorus levels above the lower limit of normal and maintained levels in the low normal range through 24 weeks, compared to 8% in the placebo arm (n=66; p<0.0001). There were three pre-specified key secondary endpoints, including stiffness and physical function, both measured by the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC®), and pain measured by the Brief Pain Inventory Question 3 (BPI Q3; pain at its worst in the last 24 hours). At week 24, stiffness improved by a mean score of 7.87 points for patients treated with burosumab compared to a 0.25 point worsening among patients in the placebo group (mean difference of 8.12; p=0.0122). Physical function improved by 3.11 points for patients treated with burosumab compared to a 1.79 point worsening among patients in the placebo group (mean difference of 4.90 points; p=0.0478). Pain score improved by 0.79 for patients treated with burosumab compared to a 0.32 improvement among patients in the placebo group (mean score difference of 0.46 points;
7
p=0.0919). Results were directionally consistent towards improvement across all three key secondary endpoints. After pre-planned multiplicity adjustment, the improvement in stiffness among patients treated with burosumab remained statistically significant at the less than the 0.0167 threshold, while physical function and pain scores demonstrated strong trends.
In December 2017, we announced additional positive 48-week data from the study. From 24 to 48 weeks of treatment, 84% of patients who had received burosumab since the beginning of the study (n=68) achieved and maintained serum phosphorus levels above the lower limit of normal (2.5 mg/dL). 89% of patients who crossed over from placebo to burosumab after 24 weeks (n=66) achieved and maintained serum phosphorus levels above the lower limit of normal. Patients treated with burosumab showed continued improvement in stiffness and physical function as measured by WOMAC. For patients treated with burosumab, stiffness further improved from a mean change of 7.42 points at 24 weeks to 16.03 points at 48 weeks. Patients who crossed over from placebo to burosumab treatment had a mean change of 15.82 points from 24 to 48 weeks. Physical function also further improved from a mean change of 2.78 points at 24 weeks to 7.76 points at 48 weeks. For patients in the crossover group, physical function improved by a mean change of 8.18 points from 24 to 48 weeks. Burosumab was associated with a reduction in pain measured by BPI Q3, as well as a reduction in the use of pain medication. For patients treated with burosumab, pain scores further improved from a mean change of 0.81 points at 24 weeks to 1.09 points at 48 weeks. Patients who crossed over from placebo to burosumab treatment had a mean change of 1.18 points from 24 to 48 weeks. The patient frequency of reported opioid use decreased by 76% from 17 patients (25%) at baseline to four patients (6%) at week 48 in the burosumab group, and by 70% from 13 patients (20%) to four patients (6%) in the crossover group. The patient frequency of reported nonsteroidal anti-inflammatory drugs (NSAIDs) use decreased by 72% from 47 patients (69%) at baseline to 13 patients (19%) at week 48 in the burosumab group, and by 74% from 43 patients (65%) to 11 patients (17%) in the crossover group. Burosumab treatment resulted in increased healing of fractures (active fractures and pseudofractures) compared to placebo at week 24, and this improvement continued through 48 weeks. When evaluating follow-up X-rays in the 52% of patients with identified fractures or pseudofractures at baseline, the 43% rate of fracture healing observed at 24 weeks on burosumab increased to 63% at 48 weeks. In the crossover group which had an 8% rate of fracture healing at 24 weeks, the rate increased to 35% at week 48. The crossover patient group fracture healing result was consistent with the effect observed in the first 24 weeks of the burosumab group treatment.
There was no difference in the overall frequency of treatment emergent serious and non-serious adverse events, treatment related adverse events and serious adverse events between the group who received burosumab for the 48-week period compared to the group who received placebo for the 24-week double-blind period and then crossed over to burosumab. The safety profile at 48 weeks was generally similar to that observed at 24 weeks. The most common adverse events in patients during treatment with burosumab (>10%) were arthralgia (24%), nasopharyngitis (22%), headache (20%), back pain (16%), tooth abscess (13%), fatigue (13%), restless leg syndrome (11%), pain in extremity (11%), pain (11%), toothache (11%), vitamin D deficiency (10%), and musculoskeletal pain (10%). Eleven percent of patients who received burosumab experienced clinical symptoms compatible with hypersensitivity. There were 15 patients who experienced serious adverse events (SAEs) during treatment with burosumab, but none of these SAEs were considered treatment-related. No meaningful changes were observed in serum intact parathyroid hormone levels or ectopic mineralization as assessed by renal ultrasounds or echocardiograms. Of the 134 patients enrolled in the study, one patient in the burosumab arm discontinued treatment during the 24-week double-blind treatment period, as previously reported. During the open-label period, seven patients discontinued treatment. No discontinuations were related to adverse events or tolerability. There has been one non-treatment related death due to a car accident that was reported after the Week 48 data cutoff date.
In February 2018, we reported that bone biopsy data from adult patients in the bone quality study demonstrated continued improvement in osteomalacia. At 48 weeks, all ten patients with evaluable paired bone biopsies demonstrated meaningful improvements from baseline in mean osteoid volume/bone volume. The mean decrease from 26.1% to 11.2% among these patients represents a 57% improvement from baseline in mean osteoid volume/bone volume which is the gold standard for the evaluation of osteomalacia. The patients also demonstrated mean improvements of 32% and 26% in osteoid thickness and osteoid surface/bone surface parameters respectively. These patients also experienced a meaningful improvement in mineralization lag time. These results, including safety, are consistent with the data provided to the FDA in the first 6 of these 10 patients showing a substantial reduction in osteomalacia.
In October 2017, we announced that the FDA accepted the biologics license application, or BLA, for burosumab to treat pediatric and adult patients with XLH and has granted Priority Review status. The Prescription Drug User Fee Act, or PDUFA, action date for the BLA is April 17, 2018. The Agency has indicated that it is not currently planning to hold an advisory committee meeting to discuss the BLA. The FDA previously granted Fast Track Designation to the burosumab program for the treatment of XLH, and Breakthrough Therapy Designation for pediatric patients one year of age or older. The FDA also designated burosumab as a drug for a "rare pediatric disease," enabling issuance of a priority review voucher if burosumab is approved.
8
Burosumab for the treatment of tumor-induced osteomalacia, or TIO
We are also developing burosumab for the treatment of TIO. TIO results from typically benign tumors that produce excess levels of FGF23, which can lead to severe hypophosphatemia, osteomalacia, bone fractures, fatigue, bone and muscle pain, and muscle weakness. There are cases in which resection of the tumor is not feasible or recurrence of the tumor occurs after resection. In patients for whom the tumor is inoperable, the current standard of care consists of oral phosphate and/or vitamin D replacement. The efficacy of this treatment is often limited, as it does not treat the underlying disease and its benefits must be balanced with monitoring for potential risks such as nephrocalcinosis, hypercalciuria, and hyperparathyroidism.
In September 2017, interim data from the open-label Phase 2 study of burosumab in 17 adult patients with TIO was presented. In 16 patients with baseline and week 24 data, mean serum phosphorus, renal phosphate reabsorption (TmP/GFR) and serum 1,25 dihydroxy vitamin D levels increased after the first dose and over 24 weeks of treatment. The mean serum phosphorus level entered the normal range within two weeks of treatment, and was maintained in the low normal range through week 24 of treatment. At week 24 there was a statistically significant increase in mean percent change from baseline levels (51% and 38% respectively) of the bone turnover markers, Procollagen type 1 N-terminal propeptide (P1NP) and collagen type 1 cross-linked C-telopeptide of type I collagen (CTx). All patients had moderate to severe osteomalacia at baseline as assessed by histomorphometric indices of osteomalacia. Four patients who completed 48 weeks of treatment had bone biopsy data. In three of these patients burosumab treatment was associated with improvements in histomorphometric indices of osteomalacia. One patient did not receive burosumab consistently. Burosumab demonstrated a clinically meaningful improvement in patient reported outcomes. At 24 weeks, patients experienced a statistically significant reduction in all four fatigue parameters as assessed by the Brief Fatigue Inventory, or BFI. Burosumab also demonstrated a statistically significant increase in lower limb strength as seen with the increase in repetitions at 24 weeks in the Sit-to-Stand test (p<0.01). Adverse events occurred in all patients (n=16). Treatment-related adverse events were observed in seven patients (44%), and included, as previously disclosed, Vitamin D deficiency and rash, and dysgeusia, all mild in grade. Most adverse events were grade 1 or 2 and included two patients with injection site reactions and two patients with restless leg syndrome that were previously disclosed. Three patients had a serious adverse event (previously disclosed tumor progression, thoracic epidural tumor compression, and a mesenchymal tumor progression). None of the serious adverse events were considered treatment related and all of these patients had a history of tumor progression at baseline and one patient discontinued to treat their tumor progression. No clinically meaningful changes were observed in mean serum calcium, urinary calcium and in serum intact parathyroid hormone. We expect 48-week data from this study in the first half of 2018.
Please see “—License and Collaboration Agreements—Clinical Product Candidates—Kyowa Hakko Kirin” for a description of our collaboration and license agreement with KHK.
UX007 for the treatment of LC-FAOD
We are developing UX007 for oral administration intended as a substrate replacement therapy for patients with LC-FAOD. UX007 is a purified, pharmaceutical-grade form of triheptanoin, a specially designed synthetic triglyceride compound, created via a multi-step chemical process. Triheptanoin has been studied clinically for over a decade in more than a hundred human subjects affected by a variety of diseases. UX007 is a medium odd-chain triglyceride of seven-carbon fatty acids designed to provide substrate replacement for fatty acid metabolism and restore production of energy. Patients with LC-FAOD have a deficiency that impairs the ability to produce energy from fat, which can lead to depletion of glucose in the body, and severe liver, muscle, and heart disease, as well as death. There are currently no approved drugs or treatments specifically for LC-FAOD. The current standard of care for LC-FAOD includes diligent prevention of fasting combined with the use of low-fat/high-carbohydrate diets, carnitine supplementation in some cases, and medium even-chain triglyceride oil supplementation. Despite treatment with the current standard of care, many patients continue to suffer significant morbidity and mortality.
In November 2016, we reported positive 78-week data from the Phase 2 study in LC-FAOD patients. The study was single-arm open-label and evaluated 29 pediatric and adult patients across three main symptom groups (musculoskeletal, liver/hypoglycemia, and cardiac). Patients needed to have moderate to severe LC-FAOD with significant disease in at least one of these domains or a frequent medical events history in order to enroll. The study began with a four-week run-in period to assess baseline data while on the standard of care therapy including MCT oil, if applicable. Patients on MCT oil then discontinued it and UX007 was titrated to a target dose of 25-35% of total daily caloric intake. Patients were followed to evaluate the acute effects of UX007 treatment over 24 weeks on several endpoints, including cycle ergometry performance, 12-minute walk test, liver disease/hypoglycemia, cardiac disease, and quality of life. Patients who opted to continue were treated for a total of 78 weeks, and rates of major medical events, such as rhabdomyolysis, hypoglycemia and cardiac events, were monitored and compared to rates for the two years prior to treatment with UX007. The study evaluated the safety and tolerability of UX007. The data showed a 48.1% reduction in the mean annualized rate of MCEs (Major Clinical Events) and a 50.6% reduction in the median annualized rate of MCEs after 78 weeks of treatment, compared to the annualized rates in the 18 months prior to treatment with UX007. There was a 50.3% reduction in the mean annualized duration of all MCEs and a 76.7% reduction in the median annualized duration of MCEs following 78 weeks of UX007 treatment. The safety profile was consistent with what has been previously observed with UX007.
9
In January 2018, we announced an update to our development plan for UX007 in LC-FAOD. Following an end-of-phase 2 meeting, we are working to provide additional information to submit to FDA for consideration of an early filing based on the results from the Phase 2 study.
The clinical effect observed was considered important, but it was not clear if there were dietary or other changes in the regimen as each patient crossed over onto UX007 that might have accounted for the improvement. We are working to provide additional information to FDA to support that the improvement demonstrated was likely due to UX007 and not any other changes. After this information is submitted and evaluated by FDA, we plan to determine with the FDA whether an early submission could be pursued. We are simultaneously finalizing a full protocol for a Phase 3, randomized, controlled study examining major clinical events as the primary endpoint as discussed with the FDA. This study would provide additional information that would be important in utilization and reimbursement long-term for UX007. If the FDA agrees to an early submission based on the Phase 2 study, the Phase 3 study would serve as a post-approval commitment for label expansion. Alternatively, the Phase 3 study could serve as a registrational study if an early filing is not possible.
Please see “—License and Collaboration Agreements—Clinical Product Candidates—Baylor Research Institute” for a description of our license agreement with Baylor Research Institute.
UX007 for the treatment of Glut1 DS
We are also developing UX007 for patients with Glut1 DS. Glut1 DS is caused by a mutation affecting the gene that codes for Glut1, which is a protein that transports glucose from the blood into the brain. Because glucose is the primary source of energy for the brain, Glut1 DS results in a chronic state of brain energy deficiency and is characterized by seizures, developmental delay, and movement disorder. There are currently no approved drugs specific to Glut1 DS. The current standard of care for Glut1 DS is the ketogenic diet, an extreme high-fat (70-80% of daily calories as fat)/low-carbohydrate diet, which generates ketone bodies as an alternative energy source to glucose, and one or more antiepileptic drugs. The ketogenic diet can be effective in reducing seizures but compliance can be difficult, and the effectiveness of the diet in the treatment of developmental delay and movement disorders has not been confirmed. In addition, ketogenic diet can lead to side effects including renal stones. In general, Glut1 DS patients are considered relatively refractory to antiepileptic drugs with only approximately 8% achieving seizure control on antiepileptic drugs alone. There are currently no antiepileptic drugs approved specifically for patients with Glut1 DS.UX007 is intended as a substrate replacement therapy to provide an alternative source of energy to the brain in Glut1 DS patients.
In March 2017, we announced topline data from the Phase 2 global, randomized, double-blind, placebo-controlled, parallel-group clinical study of UX007 for the treatment of Glut1DS patients with seizures. The study enrolled 36 patients who were not fully compliant with ketogenic diet and continued to have seizures. Patients treated with UX007 (n=25) demonstrated a reduction of 13.4% in overall seizure frequency (p=0.41) relative to placebo (n=11), which did not meet the primary endpoint of reducing the frequency of total number of observable and absence seizures among patients treated from baseline to Week 8 with UX007 compared to placebo. Two of the 36 enrolled patients discontinued treatment during the eight-week placebo-controlled period, and 12 patients have discontinued during the extension period to date. Two patients discontinued due to adverse events, four patients due to tolerability reasons, and eight patients due to compliance or study burden issues. There were no deaths, and no treatment-related serious adverse events. During the placebo-controlled period, 18 patients (72%) in the UX007 arm had treatment-related adverse events (AEs) and five patients (45%) in the placebo arm had treatment-related AEs. Most AEs were mild-to-moderate GI events including vomiting, diarrhea, and abdominal pain.
In April 2017, we screened the first patient in the Phase 3 study of UX007 for the treatment of Glut1 DS patients with the movement disorder phenotype. The study is a randomized, double-blind, placebo-controlled, cross-over study designed to assess the efficacy and safety of UX007 in approximately 40 patients who are experiencing disabling paroxysmal movement disorders associated with Glut1 DS. Movement disorder events are defined as disabling if they affect or limit a patient's ability to perform activities of daily living. Eligible patients are randomized in a 1:1 ratio to one of two treatment sequences. Patients in the first group will begin a two-week titration period followed by an eight-week treatment period on UX007. Patients will then begin a 2-week washout period, followed by a 2-week titration period and 8-week period on placebo. Patients in the second group will follow the same schedule but will start with placebo and then cross over to UX007. The primary endpoint compares the frequency of disabling paroxysmal movement disorder events during the 8-week treatment period with UX007, to the frequency of disabling movement disorder events during the 8-week placebo treatment period as recorded by a daily electronic diary. Secondary endpoints include the duration of disabling paroxysmal movement disorder events; walking capacity and endurance measured by the 12-minute walk test; patient-reported health-related quality of life assessments of physical function, mobility, upper extremity function, fatigue and pain; cognitive function and safety. Following the 22-week blinded crossover study period, patients may roll into the open-label extension period to continue on UX007 treatment. We expect data from this study in the second half of 2018.
DTX301 for the treatment of OTC deficiency
We are developing DTX301 as an adeno-associated virus 8, orAAV8, gene therapy product candidate designed for patients with ornithine transcarbamylase, or OTC, deficiency. OTC is part of the urea cycle, an enzymatic pathway in the liver that converts excess nitrogen, in the form of ammonia, to urea for excretion. OTC deficiency is the most common urea cycle disorder and leads to
10
increased levels of ammonia. Patients with OTC deficiency suffer from acute hyperammonemic episodes that can lead to hospitalization, adverse cognitive and neurological effects, and death. We estimate that there are approximately 10,000 patients worldwide with OTC deficiency, of which we estimate approximately 80% are classified as late-onset, our target population. We initiated patient dosing in our Phase 1/2 open-label clinical trial of DTX301 in August 2017. DTX301 has received Orphan Drug Designation in both the United States and Europe and Fast Track Designation in the United States.
In January 2018, we announced positive interim safety and efficacy data from the first dose cohort of the Phase 1/2 study of DTX301 in OTC deficiency. All three patients in the first, lowest-dose cohort received a single DTX301 dose of 2.0 × 10^12 GC/kg. As of the December 22, 2017 data cutoff date, two of the three patients had been followed for at least 12 weeks, the pre-defined endpoint for efficacy evaluation, and the third patient had been followed for 6 weeks. As of the cutoff date, there were no infusion-related adverse events and no serious adverse events reported. All adverse events were Grade 1 or 2 and resolved. The only treatment-related adverse events were mild, clinically asymptomatic and manageable elevations in alanine aminotransferase (ALT) in two patients, peaking at 45 (Patient 1) and 118 IU/L (Patient 2). These ALT elevations were mild and similar to what has been observed in other programs using AAV gene therapy. Both patients completed a standard tapering course of corticosteroids to treat the ALT elevations, and as of the data cutoff date, their ALT levels were in the normal range (below 40 U/L). The third patient had ALTs that remained in the normal range through six weeks. The first patient's rate of ureagenesis was normalized and maintained over 12 weeks. Their rate of ureagenesis at baseline was 200 umol/kg/hr (67% of normal, defined as 300 umol/kg/hr). At 6 weeks, their rate of ureagenesis increased to 335 umol/kg/hr (67% increase from baseline, 112% of normal). At 12 weeks, their rate of ureagenesis was 261 umol/kg/hr (30% increase from baseline, 87% of normal). The second patient did not show a clinically meaningful change in rate of ureagenesis over the 12-week period. The third patient showed a modest increase in ureagenesis from baseline over the first six weeks of treatment. This patient has not yet reached the 12-week post-dosing point. Full 12-week data from the first cohort is expected in March 2018. We expect to be able to move to the higher-dose second cohort (6.0 x 10^12 GC/kg) pending the data monitoring committee's review of the 12-week safety data for all three patients in this cohort. Data from this second cohort should be available in the second half of 2018.
Please see “—License and Collaboration Agreements—Clinical Product Candidates—REGENXBIO Inc.” for a description of our license agreement with REGENXBIO Inc.
Other Development
In August 2017, we announced that a Phase 3 study evaluating aceneuramic acid extended release, or Ace-ER, in patients with GNE Myopathy did not achieve its primary endpoint of demonstrating a statistically significant difference in the upper extremity muscle strength composite score compared to placebo. The study also did not meet its key secondary endpoints. Adverse events were generally balanced between Ace-ER and placebo and safety was consistent with previously released Ace-ER data. We are discontinuing further clinical development of Ace-ER.
Preclinical Pipeline
DTX401 for the treatment of GSDIa
DTX401 is our AAV8 gene therapy program for the treatment of patients with GSDIa, a disease that arises from a defect in G6Pase, an essential enzyme in glycogen and glucose metabolism. GSDIa is the most common glycogen storage disease and we estimate there are approximately 6,000 patients worldwide. We expect to submit an IND in the first half of 2018 and expect data from the first cohort of the Phase 1/2 study in the second half of 2018. DTX401 has been granted Orphan Drug Designation in the United States and Europe.
DTX201 for the treatment of Hemophilia A
DTX201 is our FVIII gene therapy program for the treatment of hemophilia A that we are developing in collaboration with Bayer Healthcare LLC, or Bayer. Hemophilia A is the most common form of hemophilia with approximately 144,000 patients worldwide. We expect to submit an IND for DTX201 in the second half of 2018.
rhPPCA (UX004) for the treatment of galactosialidosis
Recombinant human protective protein cathepsin-A, or rhPPCA, which we in-licensed from St. Jude Children’s Research Hospital, or St. Jude, in September 2012, is in preclinical development as an enzyme replacement therapy for galactosialidosis, a rare lysosomal storage disease for which there are no currently approved drug therapies. Similar to MPS patients, patients with galactosialidosis present with both soft tissue storage in the liver, spleen, and other tissues, as well as connective tissue (bone and cartilage) related disease. As with MPS VII, an enzyme deficiency results in accumulation of substrates in the lysosomes, causing skeletal and organ dysfunction, and death. We are continuing preclinical development of rhPPCA. Please see “—License and Collaboration Agreements—Preclinical Product Candidates—St. Jude Children’s Research Hospital” for a description of our license agreement with St. Jude.
11
UX068 for the treatment of creatine transporter deficiency (CTD)
UX068 is in preclinical development for the treatment of CTD, an X-linked recessive disorder due to mutations in the SLC6A8 gene. Patients with CTD can suffer from CNS deficits, seizures, progressive intellectual disability, autism, speech/language/gross motor delays, and muscle hypotonia and hypotrophy. CTD affects approximately 10,000 to 50,000 patients in developed world.
Collaboration with Arcturus Therapeutics, Inc. for mRNA therapeutics including UX053 for the treatment of glycogen storage disease type III (GSDIII)
We signed a research collaboration and license agreement with Arcturus Therapeutics, Inc. to develop mRNA therapeutics for select rare disease targets in October 2015. The Arcturus collaboration may help us address a wider range of rare diseases than possible with current approaches. As part of the collaboration, Arcturus will utilize its UNA Oligomer™ chemistry and LUNAR™ nanoparticle delivery platform to initially design and optimize mRNA therapeutics for two targets selected by us; we also have the option to add up to eight additional targets during the collaborative research period.
The collaboration includes preclinical candidate UX053 for the treatment of GSDIII, a disease caused by a glycogen debranching enzyme (AGL) deficiency that results in glycogen accumulation in the liver and muscle. GSDIII can cause hepatomegaly, hypoglycemia, hyperlipidemia, some progressive liver cirrhosis, and muscle disease later in life, and affects more than 10,000 patients worldwide.
Collaboration with Takeda Pharmaceutical Company Limited
We entered into a strategic partnership with Takeda Pharmaceutical Company Limited, or Takeda, to develop and commercialize therapies to treat rare genetic diseases in June 2016. As part of the collaboration, we received an exclusive license to one preclinical Takeda product candidate in a pre-determined field of use. We discontinued the development efforts on the pre-clinical compound in the pre-determined field of use. We have also established a five-year research collaboration with Takeda in which we will have the option to license up to five additional Takeda product candidates for rare diseases; accordingly, we continue to evaluate additional product candidates for potential addition to the collaboration. Please see “—License and Collaboration Agreements—Preclinical Product Candidates—Takeda Pharmaceutical Company Limited” for a description of our license agreement with Takeda.
Other preclinical programs
We continue to work on other compounds in various preclinical stages of development.
Competition
The commercialization of new drugs is competitive, and we may face worldwide competition from individual investigators, major pharmaceutical companies, specialty pharmaceutical companies, biotechnology companies, nutraceutical companies, and ultimately biosimilar and generic companies. Our competitors may develop or market therapies that are more effective, safer, or less costly than any that may be commercialized by us, or may obtain regulatory approval for their therapies more rapidly than we may obtain approval for ours.
The acquisition or licensing of pharmaceutical products is also very competitive, and a number of more established companies, which have acknowledged strategies to license or acquire products, may have competitive advantages as may other emerging companies taking similar or different approaches to product acquisitions. These established companies may have a competitive advantage over us due to their size, cash flows, and institutional experience.
With respect to burosumab, although we are not aware of any other products currently in clinical development for the treatment of XLH, it is possible that competitors may produce, develop, and commercialize therapeutics, or utilize other approaches such as gene therapy, to treat XLH. Most pediatric patients with XLH are managed using oral phosphate replacement and/or vitamin D therapy, which is relatively inexpensive and therefore may adversely affect our ability to commercialize burosumab, if approved, in some countries.
With respect to Mepsevii, we are not aware of any other compounds currently in clinical development for MPS VII, but it is possible that other companies may produce, develop, and commercialize compounds that might treat these diseases. Additionally, gene therapy and other therapeutic approaches may emerge for the treatment of lysosomal diseases. Bone marrow or stem cell transplants have also been used in MPS VII and in other lysosomal storage diseases and represent a potential competing therapy. Stem cell transplants have been effective in treating soft tissue storage and in having an impact on brain disease, but have not to date proven effective in treating bone and connective tissue disease. Typically, enzyme replacement therapy has had an impact on bone and connective tissue disease in other disorders when patients were treated early.
12
With respect to UX007/triheptanoin, there are currently no approved drugs or treatments for patients with LC-FAOD or Glut1 DS. LC-FAOD is commonly treated with diet therapy and MCT oil, and UX007 would compete with MCT oil. Glut1 DS is commonly treated with ketogenic diet and antiepileptic drugs. UX007 may compete with these approaches. Although we believe that UX007 should be considered a drug and will be regulated that way, it is possible that other companies or individuals may attempt to produce triheptanoin for use in LC-FAOD, Glut1 DS, and other patients by attempting to sell the product via a nutritional supplement or medical food pathway. Investigators are testing triheptanoin in clinical studies across multiple indications, including LC-FAOD and Glut1 DS. For example, B. Braun Medical Inc., or B. Braun, has applied for and received orphan drug designation for triheptanoin for the treatment of certain types of LC-FAOD in Europe; however, we are not aware of any ongoing clinical development activities by B. Braun. It is also possible that other companies may produce, develop, and commercialize other medium odd-chain fatty acids, or completely different compounds, to treat LC-FAOD and Glut1 DS. Other companies may also utilize other approaches, such as gene therapy, to treat LC-FAOD and Glut1 DS.
With respect to DTX301, the current treatments for patients with OTC deficiency are nitrogen scavenging drugs and severe limitations in dietary protein. Drug therapy includes sodium phenylbutyrate (Buphenyl) and glycerol phenylbutyrate (Ravicti), both nitrogen scavengers that help eliminate excess nitrogen, in the form of ammonia, by facilitating its excretion. During a metabolic crisis, patients routinely receive carbohydrate and lipid rich nutrition, including overnight feeding through a nasogastric tube, to limit bodily protein breakdown and ammonia production. In acute cases, ammonia must be removed by dialysis or hemofiltration. Liver transplant may also be a solution for OTC deficiency.
Many of our competitors have substantially greater financial, technical, and human resources than we have. Additional mergers and acquisitions in the pharmaceutical industry may result in even more resources being concentrated in our competitors. Competition may increase further as a result of advances made in the commercial applicability of technologies and greater availability of capital for investment in these fields. Our success will be based in part on our ability to build and actively manage a portfolio of drugs that addresses unmet medical needs and creates value in patient therapy.
License and Collaboration Agreements
Our product and current product candidate pipeline have been either in-licensed from academic institutions or derived from partnerships with other pharmaceutical companies. Following is a description of our significant license and collaboration agreements.
Approved Product
Saint Louis University
In November 2010, we entered into a license agreement with Saint Louis University, or SLU, wherein SLU granted us certain exclusive rights to intellectual property related to GUS. Under the terms of the license agreement, SLU granted us an exclusive worldwide license to make, have made, use, import, offer for sale, and sell therapeutics related to SLU’s beta-glucuronidase product, such as our rhGUS product candidate, for use in the treatment of human diseases. Under this agreement, we agreed to use best efforts to develop and commercialize a licensed product as soon as practicable consistent with sound and reasonable business practices and judgment.
Under the license agreement, upon reaching a certain level of worldwide sales of the product, we will pay to SLU a low single-digit royalty on net sales of the licensed products in any country or region, subject to certain potential deductions. Our obligation to pay royalties to SLU continues on a country-by-country basis until the expiration of the last-to-expire licensed patent covering the product in such country or, in the United States, Japan, and the EU, until the later expiration of any orphan drug exclusivity. We may terminate the agreement for convenience at any time and SLU may terminate the agreement for our material breach, bankruptcy, or challenge of the licensed patents or technology, and SLU may terminate the agreement or render our license non-exclusive if we fail to meet our diligence obligations. Unless terminated as set forth above, this license agreement continues in full force and effect until the latest of expiration of the last patent based on technology licensed under the agreement, at which point our license becomes fully paid.
13
Kyowa Hakko Kirin
In August 2013, we entered into a collaboration and license agreement with KHK. Under the terms of this collaboration and license agreement, as amended, we and KHK will collaborate on the development and commercialization of certain products containing burosumab in the field of orphan diseases in the United States and Canada, or the “profit-share territory”, and in the European Union and Switzerland, or the European territory, and we will have the right to develop and commercialize such products in the field of orphan diseases in Mexico and Central and South America, or Latin America. In the field of orphan diseases, and except for ongoing studies being conducted by KHK, we will be the lead party for development activities in the profit-share territory and in the European territory until the applicable transition date; we will also be the lead party for core development activities conducted in Japan and Korea for which the core development plan is limited to clinical trials mutually agreed to by us and KHK. We will share the costs for development activities in the profit share territory and European territory conducted pursuant to the development plan before the applicable transition date equally with KHK and KHK shall be responsible for 100% of the costs for development activities in Japan and Korea. On the applicable transition date in the profit-share territory and the European territory, KHK will become the lead party and be responsible for the costs of the development activities. However, we will continue to share the costs of the studies commenced prior to the applicable transition date equally with KHK. We have the primary responsibility for conducting certain research and development activities. We are obligated to provide assistance in accordance with the agreed upon development plan as well as participate on various committees. If burosumab is approved, we and KHK will share commercial responsibilities and profits in the profit share territory until the applicable transition date, KHK will commercialize burosumab in the European territory, and we will develop and commercialize burosumab in Latin America. KHK will manufacture and supply burosumab for clinical use globally and will manufacture and supply burosumab for commercial use in the profit share territory and Latin America.
In the profit share territory, KHK will book sales of products and we will have the sole right to promote the products for a specified period of time, with KHK increasingly participating in the promotion of the products until five years from commercial launch, after which KHK will have the sole right to promote the products, subject to a limited promotion right retained by us. In the European territory, KHK will book sales of products and have the sole right to promote and sell the products. In Latin America, we will book sales of products and have the sole right to promote and sell the products.
KHK will supply all quantities of product for clinical studies. KHK will also supply all quantities of product for commercial sales in the profit-share territory and in Latin America. The supply price to us for commercial sales in the profit-share territory and in Latin America will be determined based on a fixed double-digit percentage of net sales.
The remaining profit or loss from commercializing products in the profit-share territory, until the applicable transition date, will be shared between us and KHK on a 50/50 basis. Thereafter, we will be entitled to receive a tiered double-digit revenue share in the mid-to-high 20% range in the profit share territory, intended to approximate the profit share. We will also be entitled to receive a royalty of up to 10% on net sales in the European territory. In Latin America, we will pay to KHK a low single-digit royalty on net sales. Our and KHK’s obligations to pay royalties will continue on a country-by-country basis for so long as we or KHK, as applicable, are selling products in such country.
In May 2017, we signed an agreement with a wholly-owned subsidiary of KHK pursuant to which we were granted the right to commercialize burosumab in Turkey. KHK’s subsidiary has the option to assume responsibility for commercialization efforts from us, after a certain minimum period.
The collaboration and license agreement will continue for as long as products in the field of orphan diseases are sold in the profit-share territory, European territory, Turkey, or Latin America, unless the agreement is terminated in accordance with its terms.
KHK may terminate the agreement in certain countries or territories based upon our failure to meet certain milestones. Specifically, if we do not obtain U.S. or European marketing approval of burosumab for the treatment of XLH by a certain date, or make a first commercial sale, on a country-by-country basis, in Latin America by certain deadlines, KHK may terminate the agreement only with respect to the applicable territory or country in which the milestone was not timely met. In certain circumstances, we have the right to obtain an extension of the applicable deadline by making a payment to KHK in the low single-digit to low double-digit millions of dollars, depending on the milestone. Also, in the event of the occurrence of certain excusable delays, the deadline for meeting the applicable milestone above is extended to account for the period of the delay. Furthermore, either party may terminate the agreement for the material breach or bankruptcy of the other party. In any event of termination by KHK, unless such termination is the result of KHK’s termination for certain types of breach of the agreement by us, we may receive low single-digit to low double-digit royalties on net post-termination sales by KHK in one or more countries or territories, the amount of which varies depending on the timing of, and reason for, such termination. In any event of termination, our rights to burosumab under the agreement and our obligations to share development costs will cease, and the program will revert to KHK, worldwide if the agreement is terminated as a whole or solely in the terminated countries if the agreement is terminated solely with respect to certain countries.
14
In September 2012, we entered into a license agreement with Baylor Research Institute, or BRI, under which we exclusively licensed certain intellectual property related to triheptanoin. The license includes patents, patent applications, know-how, and intellectual property related to the composition and formulation of triheptanoin as well as its use in treating a number of orphan diseases, including LC-FAOD. The license grant includes the sole right to develop, manufacture, and commercialize licensed products for all human and animal uses. Under the license agreement, we are obligated to use commercially reasonable efforts to develop and commercialize licensed products in select orphan indications. If we fail to meet our diligence obligations with respect to a specified orphan indication or set of orphan indications, BRI may convert our license to a non-exclusive license with respect to such orphan indication or set of orphan indications until we receive regulatory approval for licensed products in the applicable orphan indication or set of orphan indications. We are also obligated to pay a mid-single digit royalty on net sales to BRI, subject to certain reductions and offsets. Our obligation to pay royalties to BRI continues on a licensed product-by-licensed product and country-by-country basis until the later of the expiration of the first regulatory exclusivity granted with respect to such product in such country or the expiration of the last-to-expire licensed patent claiming such product in such country, in each case in connection with approval in such country for LC-FAOD or an orphan disease covered by our license from BRI. We may make future payments of up to $10.5 million contingent upon attainment of certain development milestones and $7.5 million if certain sales milestones are achieved. We may terminate the agreement for convenience at any time and either we or BRI may terminate the agreement for the material breach or bankruptcy of the other party. If we terminate for BRI’s breach or bankruptcy, our license from BRI will remain in effect, subject to our continued payment of reduced milestones and royalties. Unless terminated by its terms, this license agreement continues in full force and effect, on a product-by-product and country-by-country basis, until our royalty obligations expire, at which point our license from BRI with respect to such product in such country becomes irrevocable, perpetual, fully paid and royalty-free.
REGENXBIO Inc.
In October 2013, we entered into an exclusive license agreement with REGENXBIO Inc., or REGENX, under which we are developing products to treat hemophilia B, hemophilia A, OTC deficiency and GSD1a. Under the 2013 license agreement, REGENX granted us an exclusive worldwide license to make, have made, use, import, sell, and offer for sale licensed products with respect to such disease indications, subject to certain exclusions. We do not have the right to control prosecution of the in-licensed patent applications, and our rights to enforce the in-licensed patents are subject to certain limitations. Under the 2013 license agreement, we pay or will pay REGENX an annual maintenance fee and certain milestone fees per disease indication, low to mid single-digit royalty percentages on net sales of licensed products, and milestone and sublicense fees, if any, owed by REGENX to its licensors as a result of our activities under the 2013 license agreement. We are required to develop licensed products in accordance with certain milestones. In the event that we fail to meet a particular milestone within established deadlines, we can extend the relevant deadline by providing a separate payment to REGENX. The 2013 license agreement will expire upon the expiration, lapse, abandonment, or invalidation of the last claim of the licensed intellectual property to expire, lapse, or become abandoned or unenforceable in all the countries of the world. Upon expiration, our know-how license will become non-exclusive, perpetual, irrevocable and royalty-free with respect to licensed know-how that REGENX owns in the field and will continue with respect to all of REGENX’s other know-how in the field under certain of its licenses for so long as its rights from those licensors continue. Subject to certain obligations to Bayer, we may terminate the 2013 license agreement upon prior written notice or for a material breach. REGENX may terminate the license agreement if we or our controlling affiliate become insolvent, are late in paying money due, commence certain actions relating to the licensed patents or materially breach the agreement. If the 2013 license agreement is terminated with respect to an indication, we grant certain rights to REGENX, including transferring ownership of any applicable regulatory approvals and granting an exclusive license under certain of our intellectual property for use with respect to products covered by the intellectual property we had licensed from REGENX in that indication.
15
In March 2015, we entered into an option and license agreement with REGENX under which we are developing product candidates to treat PKU, citrullinemia type 1 and Wilson disease and have an option for another disease indication. The 2015 option and license agreement grants us an exclusive worldwide license to make, have made, use, import, sell, and offer for sale licensed products with respect to such disease indications, subject to certain exclusions. The exercise of our remaining option is subject to availability. We do not have the right to control prosecution of the in-licensed patent applications, and our rights to enforce the in-licensed patents are subject to certain limitations. Under the 2015 option and license agreement, we pay or will pay REGENX an annual maintenance fee and certain milestone fees per disease indication, mid to high single-digit royalty percentages on net sales of licensed products, and mid-single to low double-digit percentages of any sublicense fees we receive from sublicenses for the licensed intellectual property rights. We are obligated to pay REGENX an upfront fee if we exercise our remaining option. We are required to develop licensed products in accordance with certain milestones. In the event that we fail to meet a particular milestone within established deadlines, we can extend the relevant deadline by providing a separate payment to REGENX. The 2015 option and license agreement will expire upon the expiration of the royalty obligations with respect to all licensed products for all licensed indications under all licenses granted under all exercised commercial options. Our remaining option terminates in 2019, unless we extend the date by paying an additional fee. Upon expiration, our know-how license will become non-exclusive, perpetual, irrevocable and royalty-free with respect to licensed know-how that REGENX owns in the field and will continue with respect to all of REGENX’s other know-how in the field under certain of its licenses for so long as its rights from those licensors continue. We may terminate the 2015 option and license agreement upon prior written notice or for a material breach. REGENX may terminate the 2015 option and license agreement if we or our controlling affiliate become insolvent, are late in paying money due, commence certain actions relating to the licensed patents or materially breach the agreement. If the 2015 option and license agreement is terminated with respect to an indication, we grant certain rights to REGENX, including transferring ownership of any applicable regulatory approvals and granting an exclusive license under certain of our intellectual property for use with respect to products covered by the intellectual property we had licensed from REGENX in that indication.
Preclinical Product Candidates
St. Jude Children’s Research Hospital
In September 2012, we entered into a license agreement with St. Jude wherein St. Jude granted us certain exclusive rights to intellectual property related to rhPPCA. Under the terms of the license agreement, St. Jude granted us an exclusive license under certain know-how to research, develop, make, use, offer to sell, import, and otherwise commercialize and exploit certain PPCA protein products to treat, prevent, and/or diagnose galactosialidosis and other monogenetic diseases. We agreed to make commercially reasonable efforts to develop and commercialize at least one licensed product.
Under the license agreement, we will pay to St. Jude a royalty of less than 1% on net sales of these products for so long as such products retain orphan drug exclusivity, on a country-by-country basis. We may terminate the agreement for convenience at any time and St. Jude may terminate the agreement for our material breach of the agreement. Unless terminated by its terms, this license agreement continues in full force and effect, until our royalty obligations expire, at which point our license becomes irrevocable, perpetual, fully paid, and royalty-free.
Takeda Pharmaceutical Company Limited
In June 2016, we entered into a collaboration and license agreement with Takeda. Under the terms of the license agreement, we obtained, among other things, an exclusive license for a pre-clinical compound from Takeda in a pre-determined field of use, which includes an option to an additional field of use for this product. We are responsible for the development costs for the pre-clinical compound pursuant to an initial development plan.
As part of the agreement, we established a five-year research collaboration with Takeda whereby the parties may mutually agree to add additional option products candidates to the collaboration, in which case we will bear the cost of the development activities, with certain exceptions, and terms to be negotiated
We also granted Takeda an exclusive option for Asian rights, for a limited period, to any licensed products and any additional products resulting from the collaboration, as well as an option to exclusively license one of our products for development and commercialization in Japan. If Takeda exercises any of its option rights to license a product pursuant to the agreement, Takeda will pay for the development costs within the licensed territory, will share in a portion of the global development costs, and will make a milestone payment upon regulatory approval. Takeda will also owe royalties on net sales in the licensed territory for any licensed product, depending on the development stage when the product is licensed as well as sales levels. The royalties related to the option to license our product, as well as the additional product are subject to future good faith negotiations at the time that the option is exercised.
We discontinued the development efforts on the pre-clinical compound in the pre-determined field of use. We continue to evaluate additional product candidates for potential addition to the collaboration.
16
In June 2014, we entered into an agreement with Bayer to research, develop and commercialize AAV gene therapy products for treatment of hemophilia A. Under this agreement, we granted Bayer an exclusive license to develop and commercialize one or more novel gene therapies for hemophilia A. We are responsible for the development of DTX201 through a proof-of-concept clinical trial, with reimbursement from Bayer for project costs. Bayer is responsible operationally, including for conducting the proof-of-concept clinical trial, and will incur the costs of the conduct of the trial. Upon the successful demonstration of clinical proof of concept, Bayer agreed to use commercially reasonable efforts to manage and fund any subsequent clinical trials and commercialization of gene therapy products for treatment of hemophilia A. Bayer will have worldwide rights to commercialize the potential future product.
Under the agreement, Bayer paid us an upfront cash payment and will pay us development and commercialization milestone payments, and tiered royalties based on product sales. The agreement expires on a licensed treatment-by-licensed treatment and country-by-country basis until the later of ten years from the date of first commercial sale or when patent claims have expired, lapsed, been abandoned, or been invalidated in such country. Either party may terminate the agreement for an uncured material breach by the other party. Bayer may terminate the agreement upon prior notice to us, either in its entirety or with respect to certain territories subject to the agreement. Bayer may also terminate the agreement upon notice of a product’s failure to meet certain criteria or after the successful completion of certain Phase I trials in the event Bayer makes a good faith determination that there is a material safety issue with respect to such product. Either party may terminate the agreement upon bankruptcy or insolvency of the other party, and we may terminate the agreement if Bayer institutes certain actions. Under certain termination circumstances, we would have worldwide rights to the terminated program(s).
University of Pennsylvania
In January 2015, we entered into an agreement with the University of Pennsylvania to sponsor certain research of Dr. Wilson at University of Pennsylvania School of Medicine related to liver gene therapy and hemophilia. Under the agreement, the University of Pennsylvania granted us an option to obtain a worldwide, non-exclusive or exclusive, royalty-bearing license, with the right to sublicense, under certain patent rights conceived, created or reduced to practice in the conduct of the research. We are required to reimburse the University of Pennsylvania for filing, prosecuting and maintaining such patent rights unless and until we decline to exercise our option. The University is required to provide us with task-based, scientific reports of progress and results of the research, and granted us a royalty-free, nontransferable, non-exclusive right to copy and distribute any research reports furnished to us for any reasonable purpose, provided the results are not made publicly available until certain conditions are met, and the right to use, disclose and otherwise exploit the research results for any reasonable purpose, subject to similar restrictions on our public disclosure of the research results.
This agreement expired on December 31, 2017, but ongoing mutual commitments extend into 2018. The agreement may be extended further, or renewed, by mutual agreement. If extended or renewed, then either party may terminate the agreement if Dr. Wilson becomes unavailable and an acceptable substitute is not found within a certain period of time, or if we fail to mutually agree on an acceptable work plan and budget for the sponsored research. We may also terminate the sponsored research agreement upon written notice, as long as we have met all of our payment and performance obligations. Either party may terminate this agreement for an uncured material breach. In the event of termination, we shall pay University of Pennsylvania the amount needed to cover costs through the effective termination date as well as allowable commitments extending beyond the termination date (up to one-fourth of the total budget).
In May 2016, we entered into a research, collaboration and license agreement with the University of Pennsylvania under which we are collaborating on the pre-clinical development of gene therapy products for the treatment of citrullinemia type I, phenylketonuria, and Wilson disease, each, a Subfield. Under the agreement, we were granted an exclusive, worldwide, royalty-bearing right and license to certain patent rights arising out of the research program, and a non-exclusive, worldwide, royalty-bearing right and license to certain University of Pennsylvania intellectual property, in each case to research, develop, make, have made, use, sell, offer for sale, commercialize and import licensed products in each Subfield for the term of the agreement. We will fund the cost of the research program and will be responsible for clinical development, manufacturing and commercialization of each Subfield. In addition, we will be required to make milestone payments (up to a maximum of $5 million per Subfield) if certain development milestones are achieved over time, and to pay low to mid single-digit royalties on net sales of each Subfield’s licensed products. We will also make milestone payments per approved product if certain commercial milestones are achieved.
17
Patents and Proprietary Rights
The proprietary nature of, and protection for, our product, product candidates, processes, and know-how are important to our business. Our success depends in part on our ability to protect the proprietary nature of our product, product candidates, technology, and know-how, to operate without infringing on the proprietary rights of others, and to prevent others from infringing our proprietary rights. We seek patent protection in the United States and internationally for our product, product candidates, and other technology. Our policy is to patent or in-license the technology, inventions and improvements that we consider important to the development of our business. In addition to patent protection, we intend to use other means to protect our proprietary rights, including pursuing marketing or data exclusivity periods, orphan drug status, and similar rights that are available under regulatory provisions in certain countries, including the United States, Europe, Japan, and China. See “Government Regulation—U.S. Government Regulation — Orphan Designation and Exclusivity,” “Government Regulation—U.S. Government Regulation — Pediatric Studies and Exclusivity,” “Government Regulation—U.S. Government Regulation — Patent Term Restoration,” “Government Regulation—U.S. Government Regulation — Biosimilars and Exclusivity,” “Government Regulation—U.S. Government Regulation — Abbreviated New Drug Applications for Generic Drugs and New Chemical Entity Exclusivity,” and “Government Regulation—European Union Regulation — Orphan Designation and Exclusivity” below for additional information.
We also rely on trade secrets, know-how, and continuing innovation to develop and maintain our competitive position. We cannot be certain that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents granted to us in the future will be commercially useful in protecting our technology.
We seek regulatory approval for our product candidates in disease areas with high unmet medical need, significant market potential, and where we expect to have a proprietary position through patents covering various aspects of our product candidates, such as composition, dosage, formulation, use, and manufacturing process, among others. Our success depends in part on an intellectual property portfolio that supports our future revenue streams and erects barriers to our competitors. We are maintaining and building our patent portfolio by filing new patent applications, prosecuting existing applications, and licensing and acquiring new patents and patent applications.
Despite these measures, any of our intellectual property and proprietary rights could be challenged, invalidated, circumvented, infringed or misappropriated, or such intellectual property and proprietary rights may not be sufficient to achieve or maintain market exclusivity or otherwise to provide competitive advantages. For more information, please see “Risks Related to Our Intellectual Property.”
We own or license a number of patents in the U.S. and foreign countries that cover our product, product candidates, or methods of their use. With respect to our owned or in-licensed issued patents in the U.S. and Europe, we may be entitled to obtain a patent term extension to extend the patent expiration date. For example, in the U.S., we can apply for a patent term extension of up to five years for one of the patents covering a product once the product is approved by the FDA, although the restoration period may not exceed 14 years following FDA approval of the product candidate. The exact duration of the extension depends on the time we spend in clinical studies as well as getting marketing approval from the FDA. The exclusivity positions for our commercial product, Mepsevii, and our clinical-stage product candidates as of December 31, 2017 are summarized below.
Mepsevii (Vestronidase Alfa) Exclusivity
We own three issued U.S. patents, one pending U.S. patent application, and corresponding foreign patent applications relating to the vestronidase alfa composition of matter and its use in the treatment of lysosomal storage disorders such as MPS VII. The patents in the U.S. expire in 2035. Any patents issuing from the pending application in the U.S. and the corresponding foreign pending patent applications would be projected to also expire in 2035. Mepsevii is additionally protected in the U.S. by regulatory data exclusivity until 2029 and by orphan drug exclusivity for treating MPS VII until 2024. Outside the U.S., we intend to pursue marketing and orphan drug exclusivity periods that are available under regulatory provisions in certain countries. In Europe, vestronidase alfa has received orphan drug designation for the treatment of MPS VII.
Burosumab Exclusivity
We have in-licensed rights from Kyowa Hakko Kirin Co., Ltd., or KHK, to patents and patent applications relating to burosumab and its use for the treatment of XLH and various other hypophosphatemic conditions. Pursuant to this license, we have rights to a number of issued patents and pending applications, including four U.S. patents and one pending U.S. application, as well as patents and applications in other jurisdictions covering generic and specific antibodies against FGF23 as well as their use for the treatment of XLH and related conditions. The patent terms for the issued patents in the U.S. are from 2022 to 2029 (without patent term extension), while the issued patents outside the U.S. expire between 2021 and 2028 (without patent extension). We also jointly own with KHK a pending application in the U.S. and corresponding foreign patent applications relating to dosing regimens for administration of anti-FGF23 antibodies, including burosumab. Any patents issuing from these jointly-owned applications would be projected to expire in 2035. We intend to pursue marketing and orphan drug exclusivity periods that are available to us under regulatory provisions in certain countries. Burosumab has received orphan drug designation in the U.S. and Europe for the treatment of XLH.
18
We are the licensee or owner of patents and patent applications relating to UX007 and its use for a number of diseases including FAOD and Glut1 DS. We have an exclusive license from Baylor Research Institute, or BRI, to issued U.S. patents and pending U.S. patent applications, as well as corresponding foreign patents and applications, relating to the UX007 composition and its uses. In the U.S., the in-licensed BRI patent portfolio includes issued patents with claims covering the UX007 composition that expire between 2020 and 2025 (without patent term extension). The in-licensed BRI portfolio additionally includes an issued U.S. patent with claims covering the use of UX007 for the treatment of FAOD that expires in 2020 (without patent term extension) and an issued U.S. patent with claims covering the use of UX007 for the treatment of various movement disorders associated with Glut1 DS that expires in 2033 (without patent term extension). Outside the U.S., the in-licensed BRI portfolio includes issued patents with claims covering the use of UX007 for the treatment of FAOD that expire in 2020 and pending patent applications with claims covering the use of UX007 for the treatment of Glut1 DS that would expire in 2033 if issued. We also own a pending U.S. patent application and corresponding foreign patent applications relating to our pharmaceutical-grade UX007 composition. Any patents issuing from these owned applications would be projected to expire in 2034. We intend to pursue marketing and orphan drug exclusivity periods that are available under regulatory provisions in certain countries. UX007 has received orphan designation in the U.S. for FAOD and in Europe for various subtypes of FAOD, while UX007 for the treatment of Glut1 DS has received orphan drug designation in the U.S. and Europe.
DTX301 Exclusivity
We have in-licensed patents and patent applications owned by the University of Pennsylvania relating to various adeno-associated viruses and vectors utilizing the capsids of those viruses. These patents and patent applications are licensed or sublicensed to REGENXBIO and sublicensed to us. Our product candidate DTX301 utilizes an AAV8 capsid and a codon-optimized version of the OTC gene. The in-licensed patents relevant to the AAV8 capsid expire between 2022 and 2024 in the U.S., and 2022 elsewhere. Our in-license also includes a pending application in the U.S. and corresponding foreign patent applications directed to the codon-optimized version of the OTC gene used in DTX301. Any patents issuing from these applications relating to the codon-optimized OTC gene would be projected to expire in 2035. We intend to pursue marketing and orphan drug exclusivity periods that are available under regulatory provisions in certain countries. DTX301 for the treatment of OTC deficiency has received orphan drug designation in the U.S. and Europe.
Trademarks
We have registered trademarks covering the Ultragenyx word mark in the U.S. and multiple other jurisdictions. In addition, we have a registered trademark in the U.S. covering a stylized design of our Ultragenyx Pharmaceutical logo. We also have pending trademark applications in the U.S. and multiple other jurisdictions relating to our Mepsevii brand name for vestronidase alfa.
Other
We rely upon unpatented trade secrets, know-how, and continuing technological innovation to develop and maintain our competitive position. We seek to protect our ownership of know-how and trade secrets through an active program of legal mechanisms including assignments, confidentiality agreements, material transfer agreements, research collaborations, and licenses.
Manufacturing
We currently contract with third parties for the manufacturing and testing of our product and product candidates for use in preclinical, clinical, and commercial applications and intend to do so in the future. We do not own or operate manufacturing facilities for the cGMP production of clinical or commercial quantities of our product candidates. We do, however, have process and analytical development capabilities focused on the gene therapy technologies. The use of contracted manufacturing and reliance on collaboration partners is relatively cost-efficient and has minimized the need for our direct investment in manufacturing facilities and additional staff early in development. Although we rely on contract manufacturers, we have personnel with extensive manufacturing experience to oversee our contract manufacturers. All of our third-party manufacturers will be subject to periodic audits to confirm compliance with applicable regulations and must pass inspection before we can manufacture our drugs for commercial sales.
To date, our third-party manufacturers have met our manufacturing requirements. To meet our projected needs for commercial manufacturing, third parties with whom we currently work might need to increase their scale of production or we will need to secure alternate suppliers. We believe that there are alternate sources of supply that can satisfy our clinical and commercial requirements, although we cannot be certain that identifying and establishing relationships with such sources, if necessary, would not result in significant delay or material additional costs.
19
The Mepsevii drug substance and drug product are manufactured by Rentschler Biopharma SE, or Rentschler, under non-exclusive commercial supply and services agreements effective December 2017 and January 2018, respectively. The drug substance agreement has an initial term of five years, which will be automatically extended for another five years following the initial term, and will continue in full force and effect for its term unless earlier terminated. Following the initial term, we and Rentschler can withdraw from the agreement without cause upon prior notice for specified periods. In addition, either party may terminate the agreement if the other party breaches a material provision of the agreement and such breach remains uncured for a specified period following receipt by the breaching party of written notice of such breach. The drug product agreement expires on December 31, 2025 and will continue in full force and effect for its term unless earlier terminated. Either party may terminate the agreements with immediate effect if the other party violates or breaches certain obligations set forth in the agreement, undergoes a material change in control, or infringes its intellectual property rights. We can also terminate the agreements if Rentschler loses the right to operate under the agreement. Either party can also terminate the agreements if Rentschler is unable to deliver its agreed upon services for a certain period in the case of a force majeure event. The cell line to produce Mepsevii is specific for this product and is in our control and stored in multiple secure locations. All other raw materials are commercially available. Under the drug product agreement, the last product will be produced no later than June 30, 2019, unless this date is extended in accordance with the agreement. We intend to transfer the drug product manufacturing to a new site as the Rentschler drug product manufacturing in Laupheim, Germany is being discontinued.
Product Candidates
Burosumab
The drug substance and drug product for burosumab are made by KHK in Japan under the collaboration and license agreement with KHK. The cell line to produce burosumab is specific for this product and is in KHK’s control. All other raw materials are commercially available.
UX007
The pharmaceutical-grade drug substance for UX007 is manufactured by IOI Oleo GmbH, or IOI Oleo, in Germany under an exclusive worldwide supply agreement, subject to certain limitations, executed in 2012 with an initial term of three years. The agreement automatically renews for two-year periods at the end of each then current term unless either party notifies the other party of its intention not to renew in writing at least three calendar months before the expiration of the then current term. Additionally, if a party materially breaches an obligation under the agreement and does not cure such breach within 60 days of receiving notice of the breach from the non-breaching party, the non-breaching party may terminate the agreement immediately upon written notice to the breaching party. Multiple parties have manufactured the UX007 drug product for us, which is not considered a very specialized task.
DTX301
The drug substance and drug product for DTX301, our AAV product candidate, are manufactured on a non-exclusive basis by a contract manufacturing organization, or CMO, pursuant to cGMP requirements. Since the beginning of 2015, this CMO has successfully completed two cGMP drug substance batches and two drug product batches of DTX301.
DTX 301 is currently manufactured using HEK293 adherent mammalian cells. Adherent and suspension HEK293 cells are straightforward to grow and transfect readily, and as a result, are widely used in the biotechnology industry to produce therapeutic proteins and viral vectors for gene therapy on a small scale. Vectors produced using HEK293 cells have been, or are being, used safely in multiple clinical trials, including trials conducted in the United States and European Union by other biopharmaceutical companies and academic government institutions. A key advantage of the HEK293 cell manufacturing system is flexibility and the relative speed with which AAV vectors can be manufactured for early Phase 1/2 clinical trials, allowing the establishment of early indications of therapeutic benefit in patients. As we advance and scale up our processes for Phase 3 clinical and commercial scale manufacturing, we intend to transition from the HEK293 cell manufacturing scale used for our DTX301 Phase 1/2 programs to a cell-based suspension bioreactor format.
Commercialization and Product Support
We have built our own commercial organizations in North America, Europe and Latin America to effectively support the commercialization of our product and product candidates and we expect to expand these efforts. We may elect to utilize strategic partners, distributors, or contract organizations to assist in the commercialization of our products. The commercial infrastructure for rare disease products typically consists of a targeted, specialty field organization that calls on a limited and focused group of physicians supported by field management, UltraCare liaisons (our commercial field force), internal support, and distribution support. One challenge, unique to commercializing therapies for rare diseases, is the difficulty in identifying eligible patients due to the very small and sometimes heterogeneous disease populations. Our management team will focus on maximizing patient identification for both clinical development and commercialization purposes in rare diseases.
20
Additional capabilities important to the rare disease marketplace include the management of key accounts such as managed care organizations, group-purchasing organizations, specialty pharmacies, and government accounts. To develop the appropriate commercial infrastructure, we will have to invest significant amounts of financial and management resources, some of which will be committed prior to regulatory approval of the products that they are intended to support.
We continue to build a medical affairs organization and multiple capabilities across North America, Europe and Latin America to meet the scientific needs of the healthcare providers and patients in the rare disease community, focusing on providing accurate and balanced disease state and product information across our portfolio for appropriate management of patients with rare disorders.
Medical affairs is comprised of the following capabilities in support of our mission: medical information, patient advocacy, patient diagnosis, medical scientific liaisons, research and educational grants. Medical affairs will engage as early as Phase I and will continue work throughout the lifecycle of each product and product candidate as dictated by the specific scientific needs in each therapeutic area.
Government Regulation
Government authorities in the United States (including federal, state, and local authorities) and in other countries, extensively regulate, among other things, the manufacturing, research and clinical development, marketing, labeling and packaging, storage, distribution, post-approval monitoring and reporting, advertising and promotion, pricing, and export and import of pharmaceutical products, such as those we are developing. We must obtain the requisite approvals from regulatory authorities in the United States and foreign countries prior to the commencement of clinical studies or marketing of the product in those countries. Accordingly, our operations are and will be subject to a variety of regulations and other requirements, which vary from country to country. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local, and foreign statutes and regulations require the expenditure of substantial time and financial resources.
Global Regulation of Clinical Studies
Clinical studies involve the administration of an investigational medicinal product to human subjects under the supervision of qualified investigators in accordance with protocols, GCP, the ethical principles that have their origin in the Declaration of Helsinki and applicable regulatory requirements. A protocol for each clinical study and any subsequent protocol amendments are typically submitted to the FDA or other applicable regulatory authorities as part of an IND or clinical trial application, or CTA. Additionally, approval must also be obtained from each clinical study site’s institutional review board, or IRB, or Ethics Committee, or EC, before the studies may be initiated, and the IRB or EC must monitor the study until completed. There are also requirements governing the reporting of ongoing clinical studies and clinical study results to public registries.
The clinical investigation of a drug is generally divided into three or four phases. Although the phases are usually conducted sequentially, they may overlap or be combined.
|
|
• |
Phase 1. The drug is initially introduced into healthy human subjects or patients with the target disease or condition. These studies are designed to evaluate the safety, dosage tolerance, pharmacokinetics and pharmacologic actions of the investigational new drug in humans, and if possible, to gain early evidence on effectiveness. |
|
|
• |
Phase 2. The drug is administered to a limited patient population to evaluate dosage tolerance and optimal dosage, identify possible adverse side effects and safety risks, and preliminarily evaluate efficacy. |
|
|
• |
Phase 3. The drug is administered to an expanded patient population, generally at geographically dispersed clinical study sites to generate enough data to statistically evaluate dosage, clinical effectiveness and safety, to establish the overall benefit-risk relationship of the investigational new drug product, and to provide an adequate basis for product approval. |
|
|
• |
Phase 4. In some cases, additional studies and patient follow-up are conducted to gain experience from the treatment of patients in the intended therapeutic indication. Regulatory authorities may condition approval of a marketing application for a product candidate on the sponsor’s agreement to conduct additional clinical studies after approval. In other cases, a sponsor may voluntarily conduct additional clinical studies after approval to gain more information about the drug. Such post-approval studies are typically referred to as Phase 4 clinical studies. |
A pivotal study is a clinical study that adequately meets regulatory authority requirements for the evaluation of a drug candidate’s efficacy and safety such that it can be used to justify the approval of the product. Generally, pivotal studies are Phase 3 studies, but regulatory authorities may accept results from Phase 2 studies if the study design provides a well-controlled and reliable assessment of clinical benefit, particularly in situations where there is an unmet medical need and the results are sufficiently robust.
U.S. Government Regulation
In the United States, the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act, or FDCA, and its implementing regulations, and biologics under the FDCA and the Public Health Service Act, or PHSA, and its implementing regulations. FDA approval is required before any new drug or dosage form, including a new use of a previously approved drug, can be marketed in the United States. Drugs and biologics are also subject to other federal, state, and local statutes and regulations.
21
The process required by the FDA before product candidates may be marketed or sold in the United States generally involves the following:
|
|
• |
completion of extensive preclinical laboratory tests and preclinical animal studies performed in accordance with the Good Laboratory Practices, or GLP, regulations; |
|
|
• |
submission to the FDA of an IND, which must become effective before human clinical studies may begin and must be updated annually; |
|
|
• |
conducting adequate and well-controlled human clinical studies to establish the safety and efficacy of the product candidate for each proposed indication under an active IND and approved by an independent IRB representing each clinical site; |
|
|
• |
preparation of and submission to the FDA of a new drug application, or NDA, or BLA, after completion of all pivotal clinical studies; |
|
|
• |
potential review of the product application by an FDA advisory committee, where appropriate and if applicable; |
|
|
• |
satisfactory completion of an FDA pre-approval inspection of the manufacturing facilities where the proposed drug substance and drug product are produced to assess compliance with Good Manufacturing Practices, or GMP; |
|
|
• |
FDA inspection of one or more clinical sites to assure compliance with Good Clinical Practices, or GCP; and |
|
|
• |
FDA review and approval of an NDA or BLA. |
Submission of an NDA or BLA to the FDA
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, detailed investigational new drug product information is submitted to the FDA in the form of an NDA or BLA requesting approval to market the product for one or more indications. Under federal law, the submission of most NDAs and BLAs is subject to a significant application user fee, unless waived.
Once an NDA or BLA has been submitted, the FDA’s goal is to review the application within ten months after it accepts the application for filing, or, if the application relates to an unmet medical need in the treatment of a serious or life-threatening condition, six months after the FDA accepts the application for filing. The review process can be significantly extended by FDA requests for additional information or clarification.
The FDA’s Decision on an NDA or BLA
The FDA may issue an approval letter or a Complete Response Letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA or BLA approval, the FDA may impose additional requirements, such as post-marketing studies and/or a risk evaluation and mitigation strategy (REMS) to help ensure that the benefits of the drug outweigh the potential risks. A REMS can include medication guides, communication plans for healthcare professionals, and elements to assure safe use. A Complete Response Letter indicates that the review cycle of the application is complete and the application is not ready for approval. A Complete Response Letter may require additional clinical data and/or an additional pivotal Phase 3 clinical study(ies), and/or other significant, expensive and time-consuming requirements related to clinical studies, preclinical studies or manufacturing.
Expedited Review and Accelerated Approval Programs
A sponsor may seek approval of its product candidate under programs designed to accelerate FDA’s review and approval of NDAs and BLAs. For example, Fast Track Designation may be granted to a drug intended for treatment of a serious or life-threatening disease or condition and data demonstrate its potential to address unmet medical needs for the disease or condition. The key benefits of fast track designation are the eligibility for priority review, rolling review (submission of portions of an application before the complete marketing application is submitted), and accelerated approval, if relevant criteria are met. The FDA may grant the NDA or BLA a priority review designation, which sets the target date for FDA action on the application at six months after the FDA accepts the application for filing. Priority review is granted where there is evidence that the proposed product would be a significant improvement in the safety or effectiveness of the treatment, diagnosis, or prevention of a serious condition. Priority review designation does not change the scientific/medical standard for approval or the quality of evidence necessary to support approval.
The FDA may approve an NDA or BLA under the accelerated approval program if the drug treats a serious condition, provides a meaningful advantage over available therapies, and demonstrates an effect on either (1) a surrogate endpoint that is reasonably likely to predict clinical benefit, or (2) on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. Post-marketing studies or completion of ongoing studies after marketing approval are generally required to verify the drug’s clinical benefit in relationship to the surrogate endpoint or ultimate outcome in relationship to the clinical benefit.
22
In addition, the Food and Drug Administration Safety and Innovation Act, or FDASIA, established the new Breakthrough Therapy designation. A sponsor may seek FDA designation of its product candidate as a breakthrough therapy if the drug is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. If a drug is designated as breakthrough therapy, FDA will provide more intensive guidance on the drug development program and expedite its review.
Orphan Designation and Exclusivity
The FDA may grant orphan drug designation to drugs intended to treat a rare disease or condition that affects fewer than 200,000 individuals in the United States, or if it affects more than 200,000 individuals in the United States and there is no reasonable expectation that the cost of developing and making the drug for this type of disease or condition will be recovered from sales in the United States.
Orphan drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical study costs, tax advantages, and user-fee waivers. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. In addition, the first NDA or BLA applicant to receive orphan drug designation for a particular drug is entitled to orphan drug exclusivity, which means the FDA may not approve any other application to market the same drug for the same indication for a period of seven years in the United States, except in limited circumstances. Orphan drug exclusivity does not prevent the FDA from approving a different drug for the same disease or condition, or the same drug for a different disease or condition.
Pediatric Studies and Exclusivity
NDAs and BLAs must contain data to assess the safety and effectiveness of an investigational new drug product for the claimed indications in all relevant pediatric populations in order to support dosing and administration for each pediatric subpopulation for which the drug is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults or full or partial waivers if certain criteria are met. Discussions about pediatric development plans can be discussed with the FDA at any time, but usually occur any time between the end-of-Phase II meeting and submission of the NDA or BLA. Unless otherwise required by regulation, the requirements for pediatric data do not apply to any drug for an indication for which orphan designation has been granted.
Pediatric exclusivity is another type of non-patent exclusivity in the United States that may be granted if certain FDA requirements are met, such as FDA’s determination that information relating to the use of a new drug in the pediatric population may produce health benefits, and the applicant agrees to perform and report on FDA-requested studies within a certain time frame. Pediatric exclusivity adds a period of six months of exclusivity to the end of all existing marketing exclusivity and patents held by the sponsor for that active moiety. This is not a patent term extension, but it effectively extends the regulatory period during which the FDA cannot accept or approve another application relying on the NDA or BLA sponsor’s data.
Biosimilars and Exclusivity
The Patient Protection and Affordable Care Act of 2010, or Affordable Care Act, includes a subtitle called the Biologics Price Competition and Innovation Act of 2009, or BPCI Act, which created an abbreviated approval pathway for biological products shown to be similar to, or interchangeable with, an FDA-licensed reference biological product.
A reference biologic is granted twelve years of exclusivity from the time of first licensure of the reference product. The first biologic product submitted under the abbreviated approval pathway that is determined to be interchangeable with the reference product has exclusivity against other biologics submitting under the abbreviated approval pathway for the lesser of (i) one year after the first commercial marketing, (ii) eighteen months after approval if there is no legal challenge, (iii) eighteen months after the resolution in the applicant’s favor of a lawsuit challenging the biologics’ patents if an application has been submitted, or (iv) 42 months after the application has been approved if a lawsuit is ongoing within the 42-month period.
Abbreviated New Drug Applications for Generic Drugs and New Chemical Entity Exclusivity
The Hatch-Waxman Amendments authorized the FDA to approve generic drugs that are bioequivalent (i.e. identical) to previously approved branded drugs. To obtain approval of a generic drug, an applicant must submit an abbreviated new drug application, or ANDA, to the FDA. In support of such applications, a generic manufacturer may rely on the preclinical and clinical testing conducted for a drug product previously approved under an NDA, known as the reference listed drug, or RLD.
Specifically, in order for an ANDA to be approved, the FDA must find that the generic version is bioequivalent to the RLD with respect to the active ingredients, the route of administration, the dosage form, quality and performance characteristics, the strength of the drug, and intended use.
23
The FDCA provides a period of five years of non-patent exclusivity for a new drug containing a new chemical entity. In cases where such exclusivity has been granted, an ANDA may not be filed with the FDA until the expiration of five years unless the submission is accompanied by a Paragraph IV certification, in which case the applicant may submit its application four years following the original product approval. The FDCA also provides for a period of three years of exclusivity if an NDA or supplement includes reports of one or more new clinical investigations, other than bioavailability or bioequivalence studies, that were conducted by or for the applicant and are essential to the approval of the application. This three-year exclusivity period often protects changes to a previously approved drug product, such as a new dosage form, route of administration, combination or indication.
When an ANDA applicant files its application with the FDA, it must certify, among other things, that the new product will not infringe the already approved product’s listed patents or that such patents are invalid or unenforceable, which is called a Paragraph IV certification. If the applicant does not challenge the listed patents or indicates that it is not seeking approval of a patented method of use, the ANDA application will not be approved until all the listed patents claiming the referenced product have expired. If the ANDA applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the ANDA has been accepted for filing by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days after the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA until the earlier of 30 months after the receipt of the Paragraph IV notice, expiration of the patent, or a decision in the infringement case that is favorable to the ANDA applicant.
Patent Term Restoration
Some of our U.S. patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, commonly referred to as the Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one-half the time between the effective date of an IND and the submission date of an NDA or BLA, plus the time between the submission date and the approval of that application. Only one patent applicable to an approved product is eligible for the extension and the application for the extension must be submitted prior to the expiration of the patent. The U.S. Patent and Trademark Office, or USPTO, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. Thus, for each approved product, we may apply for restoration of patent term for one of our related owned or licensed patents to add patent life beyond the original expiration date, depending on the expected length of the clinical studies and other factors involved in the filing of the relevant NDA or BLA.
European Union Regulation
In the EU, to obtain regulatory approval of an investigational medicinal product, we must submit an MAA. The content of the MAA is similar to that of an NDA or BLA filed in the United States, with the exception of, among other things, country-specific document requirements.
Authorization Procedures
Medicines can be authorized by using the centralized authorization procedure or national authorization procedures. The centralized authorization procedure results in a single marketing authorization issued by the EMA that is valid across the European Economic Area, or EEA, which is comprised of the 28 member states of the EU plus Norway, Iceland, and Lichtenstein. The centralized procedure is compulsory for human medicines that are derived from biotechnology processes, such as genetic engineering; contain a new active substance indicated for the treatment of certain diseases, such as HIV/AIDS, cancer, diabetes, neurodegenerative disorders or autoimmune diseases and other immune dysfunctions; and officially designated orphan medicines. Medicines that fall outside the mandatory scope of the centralized procedure have three routes to authorization: (i) they can be authorized under the centralized procedure if they concern a significant therapeutic, scientific or technical innovation, or if their authorization would be in the interest of public health; (ii) they can be authorized under a decentralized procedure where an applicant applies for simultaneous authorization in more than one EU country; or (iii) they can be authorized in a EU member state in accordance with that state’s national procedures and then be authorized in other EU countries by a procedure whereby the countries concerned agree to recognize the validity of the original, national marketing authorization (mutual recognition procedure).
A Pediatric Investigation Plan, or PIP, and/or a request for waiver or deferral, is required for submission prior to submitting a marketing authorization application. A PIP describes, among other things, proposed pediatric studies and their timing relative to clinical studies in adults and an MAA must comply with the PIP to be validated.
24
MAA Review and Approval Timeframe and Accelerated Assessment
Under the centralized procedure in the EU, the maximum timeframe for the evaluation of a marketing authorization application that has been validated is 210 days, excluding time taken by an applicant to respond to questions. A favorable opinion on the application by the CHMP will typically result in the granting of the marketing authorization within 67 days of receipt of the opinion. Generally, the entire review process takes approximately one year. Accelerated evaluation might be granted by the CHMP in exceptional cases, when a medicinal product is expected to be of a major public health interest, particularly from the point of view of therapeutic innovation. In this circumstance, EMA ensures that the opinion of the CHMP is given within 150 days, excluding time taken by an applicant to respond to questions.
Exceptional Circumstances/Conditional Approval
Orphan drugs or drugs with unmet medical needs may be eligible for EU approval under exceptional circumstances or with conditional approval. Approval under exceptional circumstances is applicable to orphan products and is used when an applicant is unable to provide comprehensive data on the efficacy and safety under normal conditions of use because the indication for which the product is intended is encountered so rarely that the applicant cannot reasonably be expected to provide comprehensive evidence, when the present state of scientific knowledge does not allow comprehensive information to be provided, or when it is medically unethical to collect such information. Conditional marketing authorization is applicable to orphan medicinal products, medicinal products for seriously debilitating or life-threatening diseases, or medicinal products to be used in emergency situations in response to recognized public threats. Conditional marketing authorization can be granted on the basis of less complete data than is normally required in order to meet unmet medical needs and in the interest of public health, provided the risk-benefit balance is positive, it is likely that the applicant will be able to provide the comprehensive clinical data, and unmet medical needs will be fulfilled. Conditional marketing authorization is subject to certain specific obligations to be reviewed annually.
PRIME Program
PRIME is a program launched by the EMA to enhance support for the development of medicines that target an unmet medical need. The program focuses on medicines that may offer a major therapeutic advantage over existing treatments, or benefit patients without treatment options. These medicines are considered priority medicines by EMA. To be accepted for PRIME, a medicine has to show its potential to benefit patients with unmet medical needs based on early clinical data. Through PRIME, the EMA offers early and proactive support to medicine developers to optimize development plans and the generation of robust data on a medicine’s benefits and risks and enables accelerated assessment of medicines applications.
Orphan Designation and Exclusivity
As in the United States, we may apply for designation of a product as an orphan drug for the treatment of a specific indication in the EU before the application for marketing authorization is made. The EMA’s Committee for Orphan Medicinal Products, or COMP, grants orphan drug designation to promote the development of products that are intended for the diagnosis, prevention or treatment of life-threatening or chronically debilitating conditions affecting not more than 5 in 10,000 persons in the EU Community and for which no satisfactory method of diagnosis, prevention, or treatment has been authorized (or the product would be a significant benefit to those affected). Additionally, designation is granted for products intended for the diagnosis, prevention, or treatment of a life-threatening, seriously debilitating or serious and chronic condition when, without incentives, it is unlikely that sales of the drug in the EU would be sufficient to justify the necessary investment in developing the medicinal product. Orphan drug designation entitles a party to financial incentives such as reduction of fees or fee waivers and 10 years of market exclusivity is granted following medicinal product approval. This period may be reduced to six years if the orphan drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process.
New Chemical Entity Exclusivity
In the EU, new chemical entities, sometimes referred to as new active substances, qualify for eight years of data exclusivity upon marketing authorization and an additional two years of market exclusivity. This data exclusivity, if granted, prevents regulatory authorities in the EU from referencing the innovator’s data to assess a generic (abbreviated) application for eight years, after which generic marketing authorization can be submitted, and the innovator’s data may be referenced, but not approved for two years. The overall ten-year period will be extended to a maximum of eleven years if, during the first eight of those ten years, the marketing authorization holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies.
25
Drugs manufactured or distributed pursuant to regulatory approvals are subject to pervasive and continuing regulation by the regulatory authorities, including, among other things, requirements relating to formal commitments for post approval clinical trials and studies, manufacturing, recordkeeping, periodic reporting, product sampling and distribution, marketing, labeling, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior regulatory authority review and approval.
Drug manufacturers are subject to periodic unannounced inspections by regulatory authorities and country or state agencies for compliance with GMP and other requirements. Changes to the manufacturing process are strictly regulated, and, depending on the significance of the change, may require prior regulatory approval before being implemented. Regulations also require investigation and correction of any deviations from GMP and impose reporting and documentation requirements upon us and any third-party manufacturers that we may decide to use. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain compliance with GMP and other aspects of regulatory compliance.
Pharmaceutical Coverage, Pricing and Reimbursement
In the United States and markets in other countries, sales of any products for which we receive regulatory approval for commercial sale will depend in part on the availability of coverage and reimbursement from third-party payors. Third-party payors include government authorities, managed care providers, private health insurers and other organizations. The process for determining whether a payor will provide coverage for a drug product may be separate from the process for setting the reimbursement rate that the payor will pay for the drug product. Third-party payors may limit coverage to specific drug products on an approved list, or formulary, which might not include all of the approved drugs for a particular indication. Moreover, a payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Adequate third-party reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development.
In the European Community, governments influence the price of pharmaceutical products through their pricing and reimbursement rules and control of national health care systems that fund a large part of the cost of those products to consumers. Some jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed to by the government. Other member states allow companies to fix their own prices for medicines, but monitor and control company profits. In addition, in some countries, cross-border imports from low-priced markets exert a commercial pressure on pricing within a country.
Other Healthcare Laws and Compliance Requirements
If we obtain regulatory approval for any of our product candidates, we may be subject to various laws targeting, among other things, fraud and abuse in the healthcare industry. These laws may impact, among other things, our proposed sales, marketing, and education programs. The laws that may affect our ability to operate include:
|
|
• |
the federal Anti-Kickback Statute, which prohibits, among other things, persons from knowingly and willfully soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce, or in return for, the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
|
|
• |
federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payers that are false or fraudulent; |
|
|
• |
the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, as amended, which prohibits executing a scheme to defraud any healthcare benefit program and making false statements relating to healthcare matters and imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information; |
|
|
• |
the EU General Data Protection Regulation (GDPR), replacing the EU Data Protection Directive (95/46/EC), effective May 25, 2018, which seeks to harmonize data privacy laws across Europe to ensure data subjects’ fundamental right to privacy in the EU in the digital age by imposing requirements and limitations relating to the processing, storage, purpose of collection, accuracy, security and transmission of personal data and the notification of regulation authorities about data breaches, accompanied by a strong sanctioning mechanism; |
|
|
• |
the 21st Century Cures Act, or the Cures Act, signed into law in December 2016, which introduced a wide range of reforms, such as broadening the types of data required to support drug approval, extending protections for generic competition, accelerating approval of breakthrough therapies, expanding the orphan drug product program, requiring disclosures about compassionate care programs, and clarifying how manufacturers communicate about their products; |
|
|
• |
the federal transparency laws, including the federal Physician Payment Sunshine Act, that requires drug manufacturers to disclose payments and other transfers of value provided to physicians and teaching hospitals; and |
26
Additional Regulation
The U.S. Foreign Corrupt Practices Act or FCPA, to which we are subject, prohibits corporations and individuals from engaging in certain activities to obtain or retain business or to influence a person working in an official capacity. It is illegal to pay, offer to pay or authorize the payment of anything of value to any foreign government official, government staff member, political party or political candidate in an attempt to obtain or retain business or to otherwise influence a person working in an official capacity. Similar laws exist in other countries, such as the United Kingdom, that restrict improper payments to public and private parties. Many countries have laws prohibiting these types of payments within the respective country. In addition to these anti-corruption laws, we are subject to import and export control laws, tariffs, trade barriers, economic sanctions and regulatory limitations on our ability to operate in certain foreign markets.
In addition, federal, state and foreign government bodies and agencies have adopted, are considering adopting, or may adopt laws and regulations regarding the collection, use, storage and disclosure of personally identifiable information or other information treated as confidential obtained from consumers and individuals.
We are also subject to regulation under the Occupational Safety and Health Act, the Environmental Protection Act, the Toxic Substances Control Act, the Resource Conservation and Recovery Act and other present and potential federal, state or local regulations. These and other laws govern our use, handling and disposal of various biological and chemical substances used in, and waste generated by, our operations.
Employees
As of December 31, 2017, we had 520 full-time employees. None of our employees is represented by labor unions or covered by collective bargaining agreements. We consider our relationship with our employees to be good.
Research and Development
We recognized $231.6 million, $183.2 million, and $114.7 million in research and development expense in the years ended December 31, 2017, 2016, and 2015, respectively.
Financial Information about Segments
We operate in a single accounting segment — the identification, acquisition, development and commercialization of novel products for the treatment of rare and ultra-rare diseases. Please refer to Note 1, “Organization and Basis of Presentation,” of the notes to our consolidated financial statements. For the years ended December 31, 2017, 2016 and 2015, our revenues were $2.6 million, $0.1 million and $0, respectively, and we incurred net losses of $302.1 million, $245.9 million and $145.6 million, respectively. As of December 31, 2017, 2016 and 2015, our total assets were $490.8 million, $540.6 million and $559.6 million, respectively.
Please refer to the discussion of risks related to our foreign operations in the section entitled “Item 1A. Risk Factors.”
General Information
We were incorporated in California in April 2010 and reincorporated in Delaware in June 2011. Our principal executive offices are located at 60 Leveroni Court, Novato, California 94949. Our telephone number is (415) 483-8800 and our e-mail address is info@ultragenyx.com. Our Internet website address is www.ultragenyx.com. No portion of our website is incorporated by reference into this Annual Report.
You are advised to read this Annual Report in conjunction with other reports and documents that we file from time to time with the Securities and Exchange Commission, or SEC. In particular, please read our definitive proxy statements, our Quarterly Reports on Form 10-Q and any Current Reports on Form 8-K that we may file from time to time. You may obtain copies of these reports after the date of this Annual Report directly from us or from the SEC at the SEC’s Public Reference Room at 100 F Street, N.E. Washington, D.C. 20549. In addition, the SEC maintains information for electronic filers (including Ultragenyx) at its website at www.sec.gov. The public may obtain information regarding the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. We make our periodic and current reports available on our internet website, free of charge, as soon as reasonably practicable after such material is electronically filed with, or furnished to, the SEC.
27
Investing in our common stock involves a high degree of risk. You should carefully consider the following risks, together with all the other information in this Annual Report, including our financial statements and notes thereto, before deciding to invest in our common stock. If any of the following risks actually materialize, our operating results, financial condition, and liquidity could be materially adversely affected. As a result, the trading price of our common stock could decline and you could lose part or all of your investment.
Risks Related to Our Financial Condition and Capital Requirements
We have a limited operating history on which to assess our business, have incurred significant losses since our inception, and anticipate that, excluding non-recurring events, we will continue to incur significant losses for the foreseeable future.
We are a biopharmaceutical company with a limited operating history. We have incurred net losses in each year since our inception in April 2010, including net losses of $302.1 million, $245.9 million, and $145.6 million for the years ended December 31, 2017, 2016, and 2015, respectively.
Biopharmaceutical product development is a highly speculative undertaking and involves a substantial degree of risk. We have devoted substantially all of our financial resources to identifying, acquiring, and developing our product and product candidates, including conducting clinical studies, developing manufacturing processes, manufacturing product candidates for clinical studies, and providing general and administrative support for these operations. To date, we have financed our operations primarily through the sale of equity securities. The amount of our future net losses will depend, in part, on non-recurring events, the rate of our future expenditures and our ability to obtain funding through equity or debt financings, strategic collaborations, or grants. Our future revenue will depend upon the size of any markets in which our product is approved and any of our product candidates may receive approval, and our ability to achieve sufficient market acceptance, pricing, reimbursement, and adequate market share for our product and product candidates in those markets. However, even if we obtain adequate market share for our product and product candidates, because the potential markets in which our product and product candidates may ultimately receive regulatory approval are very small, and our expenses may be greater than expected, we may never become profitable despite obtaining such market share and acceptance of our product and product candidates that may receive approval.
We expect to continue to incur significant expenses and increasing operating losses for the foreseeable future, excluding non-recurring events. We anticipate that our expenses will increase substantially if and as we:
|
|
• |
continue our research and nonclinical and clinical development of our product candidates; |
|
|
• |
expand the scope of our current clinical studies for our product candidates; |
|
|
• |
advance our programs into more expensive clinical studies; |
|
|
• |
initiate additional nonclinical, clinical, or other studies for our product candidates; |
|
|
• |
pursue preclinical and clinical development for additional indications for existing product candidates; |
|
|
• |
change or add additional manufacturers or suppliers; |
|
|
• |
seek regulatory and marketing approvals for our product candidates that successfully complete clinical studies; |
|
|
• |
establish Medical Affairs field teams to initiate relevant disease education; |
|
|
• |
establish a marketing and distribution infrastructure and field force to commercialize our product and any product candidates for which we may obtain marketing approval; |
|
|
• |
continue to establish our international subsidiaries; |
|
|
• |
continue to operate as a public company and comply with legal, accounting and other regulatory requirements; |
|
|
• |
seek to identify, assess, license, acquire, and/or develop other product candidates, technologies, and/or businesses; |
|
|
• |
make milestone or other payments under any license or other agreements; |
|
|
• |
seek to maintain, protect, and expand our intellectual property portfolio; |
|
|
• |
seek to attract and retain skilled personnel; |
|
|
• |
create additional infrastructure, including facilities and systems, to support the growth of our operations, our product development, and our planned future commercialization efforts; and |
|
|
• |
experience any delays or encounter issues with any of the above, including, but not limited to, failed studies, complex results, safety issues, inspection outcomes, or other regulatory challenges that require longer follow-up of existing studies, additional major studies, or additional supportive studies in order to pursue marketing approval. |
Further, the net losses we incur may fluctuate significantly from quarter to quarter and year to year, such that a period-to-period comparison of our results of operations may not be a good indication of our future performance.
28
We have not generated any significant revenue from product sales and we may never be profitable.
We have one product approved for commercialization but have not generated any significant revenue from product sales. Our ability to generate significant revenue and achieve profitability depends on our ability, alone or with strategic collaboration partners, to successfully commercialize our product and to complete the development of, and obtain the regulatory and marketing approvals necessary to commercialize, one or more of our product candidates. We do not anticipate generating significant revenue from product sales in the near future. Our ability to generate substantial future revenue from product sales, including named patient sales, and to potentially be profitable depends heavily on our success in many areas, including but not limited to:
|
|
• |
completing research and nonclinical and clinical development of our product candidates; |
|
|
• |
obtaining regulatory and marketing approvals for product candidates for which we complete clinical studies; |
|
|
• |
developing a sustainable and scalable manufacturing process for our product and any approved product candidates and establishing and maintaining supply and manufacturing relationships with third parties that can conduct the processes and provide adequate (in amount and quality) product supply to support clinical development and the market demand for our product and product candidates, if approved; |
|
|
• |
launching and commercializing our product and product candidates for which we obtain regulatory and marketing approval, either directly or with a collaborator or distributor; |
|
|
• |
obtaining market acceptance of our product and product candidates as viable treatment options; |
|
|
• |
obtaining adequate reimbursement and pricing for our product and product candidates; |
|
|
• |
our ability to sell our product and product candidates on a named patient basis or through an equivalent mechanism and the amount of revenue generated from such sales; |
|
|
• |
addressing any competing technological and market developments; |
|
|
• |
identifying, assessing, licensing, acquiring, and/or developing new product candidates, technologies, and/or businesses; |
|
|
• |
negotiating favorable terms in any collaboration, licensing, or other arrangements into which we may enter, any amendments thereto or extensions thereof; |
|
|
• |
maintaining, protecting, and expanding our portfolio of intellectual property rights, including patents, trade secrets, and know-how; and |
|
|
• |
attracting, hiring, and retaining qualified personnel. |
We anticipate incurring significant costs associated with commercializing our product and any approved product candidate. Our expenses could increase beyond expectations if we are required by the FDA, the EMA, or other regulatory agencies, domestic or foreign, to change our manufacturing processes or assays, or to perform clinical, nonclinical, or other types of studies in addition to those that we currently anticipate. In cases where we are successful in obtaining regulatory approvals to market one or more of our product candidates, our revenue will be dependent, in part, upon the size of the markets in the territories for which we gain regulatory approval, the accepted price for the product, the approved indication(s), the ability to obtain reimbursement at any price, and whether we own the commercial rights for that territory. If the number of our addressable rare disease patients is not as significant as we estimate, the indication approved by regulatory authorities is narrower than we expect, or the reasonably accepted population for treatment is narrowed by competition, physician choice, or treatment guidelines, we may not generate significant revenue from sales of our products, even if approved. For example, the development of burosumab and UX007 is an important part of our current business strategy; if we are unable to obtain regulatory approval for the target product profile, our business may suffer.
We expect we will need to raise additional capital to fund our activities. This additional financing may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit, or terminate our product development efforts or other activities.
As we continue to advance our product candidates through preclinical and clinical development and increase our commercialization efforts, we expect our expenses to increase substantially in connection with our ongoing activities.
As of December 31, 2017, our available cash, cash equivalents, and investments were $244.5 million. We will likely require additional capital to commercialize our product and to obtain regulatory approval for, and to commercialize, all of our product candidates. In addition, our operating plans may change as a result of many factors that may currently be unknown to us, and we may need to seek additional funds sooner than planned. Our future funding requirements will depend on many factors, including but not limited to:
|
|
• |
the scope, rate of progress, results, and cost of our clinical studies, nonclinical testing, and other related activities; |
|
|
• |
the cost of manufacturing clinical supplies, and establishing commercial supplies, of our product candidates and any products that we may develop; |
29
|
|
• |
the cost, timing, and outcomes of regulatory approvals; |
|
|
• |
the cost and timing of establishing and operating our international subsidiaries; |
|
|
• |
the cost and timing of establishing field forces, marketing, and distribution capabilities; |
|
|
• |
the cost and timing of other activities needed to commercialize our products; and |
|
|
• |
the terms and timing of any collaborative, licensing, acquisition, and other arrangements that we may establish, including any required milestone, royalty, and reimbursements or other payments thereunder. |
Any additional fundraising efforts may divert our management’s attention from their day-to-day activities, which may adversely affect our ability to develop and commercialize our product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. Moreover, the terms of any financing may adversely affect the holdings or the rights of our stockholders and the issuance of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our shares to decline. The sale of additional equity or convertible securities would dilute all of our stockholders. If we incur debt, it could result in increased fixed payment obligations and we may be required to agree to certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell, or license intellectual property rights, and other operating restrictions that could adversely impact our ability to conduct our business. If we are granted priority review vouchers in connection with regulatory approvals for our product candidates, we may be unable to sell the vouchers or, if we do sell the vouchers, we may have to sell them on unfavorable terms and at prices that are lower than expected. Regulatory authorities may also cease granting such vouchers in the future. We could also be required to seek funds through collaborative partnerships, strategic alliances, and licensing or other arrangements and we may be required to relinquish rights to some of our technologies or product candidates, future revenue streams, research programs, and other product candidates or otherwise agree to terms unfavorable to us, any of which may have a material adverse effect on our business, operating results, and prospects. Even if we believe we have sufficient funds for our current or future operating plans, we may seek additional capital if market conditions are favorable or if we have specific strategic considerations.
If we are unable to obtain funding on a timely basis or at all, we may be required to significantly curtail, delay, or discontinue one or more of our research or development programs or the commercialization of our product and any approved product candidates or be unable to expand our operations or otherwise capitalize on our business opportunities, as desired, which could materially affect our business, financial condition, and results of operations.
Risks Related to the Discovery and Development of Our Product Candidates
Clinical drug development involves a lengthy and expensive process with uncertain outcomes and the potential for substantial delays, and the results of earlier studies may not be predictive of future study results.
Before obtaining marketing approval from regulatory authorities for the sale of our product candidates, we must conduct extensive clinical studies to demonstrate the safety and efficacy of the product candidates in humans. Clinical testing is expensive, time consuming, and uncertain as to outcome. We cannot guarantee that any clinical studies will be conducted as planned or completed on schedule, if at all. A failure of one or more clinical studies can occur at any stage of testing, and our future clinical studies may not be successful. Product candidates that have shown promising results in early-stage clinical studies may still suffer significant setbacks in subsequent clinical studies. The safety or efficacy results generated to date in clinical studies for burosumab and UX007 do not ensure that later clinical studies will demonstrate similar results. For example, our Phase 3 study that evaluated Ace-ER in patients with GNE myopathy did not achieve its primary or secondary endpoints and efficacy results from our Phase 2 study of UX007 in Glut1 DS patients with seizures did not meet the primary endpoint. Results from investigator-sponsored studies or compassionate-use studies may not be confirmed in company-sponsored studies or may negatively impact the prospects for our programs. Additionally, given the nature of the rare diseases we are seeking to treat, we often have to devise newly-defined endpoints to be tested in our studies, which can lead to some subjectivity in interpreting study results and could result in regulatory agencies not agreeing with the validity of our endpoints, or our interpretation of the clinical data, and therefore denying approval. For example, for our Glut1 DS Phase 3 clinical trial, we have proposed utilizing a patient diary to track movement disorder events. Based on FDA feedback expressing concern about the clinical meaningfulness of all such events tracked, we modified the clinical endpoint. Even so, there is no guarantee that these modifications to the endpoint will be acceptable to the FDA. Given the illness of the subjects in our studies and the nature of their rare diseases, we may also be required or choose to conduct certain studies on an open-label basis. Additionally, we have in the past, and may in the future elect to review interim clinical data at multiple time points during the studies, which could introduce bias into the study results and potentially result in denial of approval.
In the biopharmaceutical industry, there is a high failure rate for drugs and biologics proceeding through clinical studies, and product candidates in later stages of clinical studies may fail to show the desired safety and efficacy despite having progressed through nonclinical studies and initial clinical studies. A number of companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical studies due to lack of efficacy or adverse safety profiles, notwithstanding promising results in earlier studies.
30
Scenarios that may prevent successful or timely completion of clinical development include but are not limited to:
|
|
• |
delays or failures in generating sufficient preclinical, toxicology, or other in vivo or in vitro data to support the initiation or continuation of human clinical studies or filings for regulatory approval; |
|
|
• |
failure to demonstrate a starting dose for our product candidates in the clinic that might be reasonably expected to result in a clinical benefit; |
|
|
• |
delays or failures in reaching a consensus with regulatory agencies on study design; |
|
|
• |
delays in reaching agreement on acceptable terms with contract research organizations, or CROs, clinical study sites, and other clinical trial-related vendors; |
|
|
• |
failure or delays in obtaining required regulatory agency approval and/or IRB or EC approval at each clinical study site or in certain countries; |
|
|
• |
failure to correctly design clinical studies which may result in those studies failing to meet their endpoints; |
|
|
• |
changes in clinical study design or development strategy resulting in delays related to obtaining approvals from IRBs or ECs and/or regulatory agencies to proceed with clinical studies; |
|
|
• |
imposition of a clinical hold by regulatory agencies after review of an IND application or amendment, another equivalent application or amendment, or an inspection of our clinical study operations or study sites; |
|
|
• |
delays in recruiting suitable patients to participate in our clinical studies; |
|
|
• |
difficulty collaborating with patient groups and investigators; |
|
|
• |
failure by our CROs, other third parties, or us to adhere to clinical study requirements; |
|
|
• |
failure to perform in accordance with the FDA’s and/or ICH’s good clinical practices requirements or applicable regulatory guidelines in other countries; |
|
|
• |
delays in patients’ completion of studies or their returns for post-treatment follow-up; |
|
|
• |
patients dropping out of a study; |
|
|
• |
adverse events associated with the product candidate occurring that are viewed to outweigh its potential benefits; |
|
|
• |
changes in regulatory requirements and guidance that require amending or submitting new clinical protocols; |
|
|
• |
greater than anticipated costs associated with clinical studies of our drug candidates; |
|
|
• |
clinical studies of our drug candidates producing negative or inconclusive results, which may result in us deciding, or regulators requiring us, to conduct additional clinical or nonclinical studies or to abandon drug development programs; |
|
|
• |
competing clinical studies of potential alternative product candidates or investigator-sponsored studies of our product candidates; and |
|
|
• |
delays in manufacturing, testing, releasing, validating, or importing/exporting sufficient stable quantities of our product candidates for use in clinical studies or the inability to do any of the foregoing. |
Any inability to successfully complete nonclinical and clinical development could result in additional costs to us or negatively impact our ability to generate revenue. In addition, if we make manufacturing or formulation changes to our product candidates, we may need to conduct additional studies to bridge our modified product candidates to earlier versions. Clinical study delays could also shorten any periods during which our products have commercial exclusivity and may allow our competitors to bring products to market before we do, which could negatively impact our ability to obtain orphan exclusivity and to successfully commercialize our product candidates and may harm our business and results of operations.
If we do not achieve our projected development goals in the timeframes we announce and expect, the commercialization of our products may be delayed and the credibility of our management may be adversely affected and, as a result, our stock price may decline.
For planning purposes, we estimate the timing of the accomplishment of various scientific, clinical, regulatory, and other product development goals, which we sometimes refer to as milestones. These milestones may include the commencement or completion of scientific studies and clinical trials, the submission of regulatory filings, and the potential approval of such regulatory filings. We periodically make public announcements about the expected timing of some of these milestones. All of these milestones are based on a variety of assumptions, but the actual timing of these milestones can vary dramatically from our estimates. If we do not meet these publicly announced milestones, the commercialization of our products may be delayed and the credibility of our management may be adversely affected and, as a result, our stock price may decline.
31
We are heavily dependent on the success of our product candidates, some of which are in the early stages of clinical development, which is a lengthy and expensive process with uncertain outcomes and the potential for substantial delays. We cannot give any assurance that any of our product candidates will receive regulatory approval, which is necessary before they can be commercialized.
Before obtaining marketing approval from regulatory authorities for the sale of our product candidates, we must conduct extensive clinical studies to demonstrate the safety and efficacy of the product candidates in humans. To date, we have focused substantially all of our efforts and financial resources on identifying, acquiring, and developing our product candidates, including conducting clinical studies and providing general and administrative support for these operations. We cannot be certain that any clinical studies will be conducted as planned or completed on schedule, if at all. Our inability to successfully complete nonclinical and clinical development could result in additional costs to us and negatively impact our ability to generate revenue. Our future success is dependent on our ability to successfully develop, obtain regulatory approval for, and then successfully commercialize our product candidates. We currently generate no significant revenue from sales of drugs, and we may never be able to develop or successfully commercialize an additional marketable drug.
Each of our product candidates is in development and will require additional clinical development; management of nonclinical, clinical, and manufacturing activities; regulatory approval; obtaining adequate manufacturing supply; building a commercial organization; significant marketing efforts; and reimbursement before we generate any significant revenue from commercial product sales, if ever. For burosumab, we filed for conditional marketing authorization in the EU in late 2016 based on Phase 1/2 and Phase 2 data, and we filed a BLA in the US in August 2017 based on Phase 2 data for the XLH pediatric indication and Phase 3 data for the adult XLH indication. For Mepsevii, which was approved in the US in November 2017, we filed a MAA in the EU, which was accepted for review in the first half of 2017, based on Phase 3 data. There can be no assurance that we will be able to secure approval with the current filings, for the specified indications, or within projected time periods. For example, in November 2016, we withdrew our first filing application for regulatory approval that sought conditional marketing authorization in the EU for Ace-ER, and in August 2017 we discontinued further clinical development of Ace-ER. Even if we obtain regulatory approval, it may be withdrawn under certain circumstances. In addition, confirmatory clinical studies could be required for a conditional marketing authorization and could fail to demonstrate sufficient safety and efficacy to obtain full approval.
Some of our product candidates are in the early-stage translational research phases of development. Such early-stage programs will require substantial investment to reach clinical studies and regulatory approval, and their risk of failure is high. For example, our collaboration with Arcturus focuses on an advanced but less established technology platform that will require significant effort and investment. A failure in that collaboration or our other early-stage programs may negatively affect our operational results.
We generally plan to seek regulatory approval to commercialize our product candidates in the United States, the EU, and in additional foreign countries where we have commercial rights. To obtain regulatory approval in other countries, we must comply with numerous and varying regulatory requirements of such other countries regarding safety, efficacy, chemistry, manufacturing, and controls, clinical studies, commercial sales, pricing, and distribution of our product candidates. Even if we are successful in obtaining approval in one jurisdiction, we cannot ensure that we will obtain approval in any other jurisdiction. If we are unable to obtain approval for our product candidates in multiple jurisdictions, our revenue and results of operations could be negatively affected.
We cannot be certain that any of our product candidates will be successful in clinical studies or receive regulatory approval. Further, our product candidates may not receive regulatory approval even if they are successful in clinical studies. If we do not receive regulatory approvals for our product candidates, we may not be able to continue our operations.
We may find it difficult to enroll patients in our clinical studies given the limited number of patients who have the diseases for which our product candidates are being studied. Difficulty in enrolling patients could delay or prevent clinical studies of our product candidates.
Identifying and qualifying patients to participate in clinical studies of our product candidates is critical to our success. The timing of our clinical studies depends in part on the speed at which we can recruit patients to participate in testing our product candidates, and we may experience delays in our clinical studies if we encounter difficulties in enrollment.
Each of the conditions for which we plan to evaluate our current product candidates is a rare genetic disease. Accordingly, there are limited patient pools from which to draw for clinical studies. For our current product candidates:
|
|
• |
we estimate that several thousand patients in the United States suffer from XLH, for which burosumab is being studied; |
|
|
• |
we estimate that several hundred patients in the United States suffer from TIO, for which burosumab is being studied; |
|
|
• |
we estimate that several thousand patients in the United States suffer from LC-FAOD, for which UX007 is being studied; |
|
|
• |
we estimate that several thousand patients in the United States suffer from Glut1 DS, for which UX007 is being studied; and |
|
|
• |
we estimate that approximately 8,000 patients in the developed world suffer from late-onset OTC deficiency, for which DTX301 is being studied, and these all may not be treatable if they are immune to the virus. |
32
In addition to the rarity of these diseases, the eligibility criteria of our clinical studies will further limit the pool of available study participants as we will require patients to have specific characteristics that we can measure or to assure their disease is either severe enough or not too advanced to include them in a study. For example, enrolling patients in the UX007 Glut1 DS Phase 3 movement disorder study could face delays if a higher than expected number of patients that we identify for the study are on the ketogenic diet. Additionally, the process of finding and diagnosing patients may prove costly, especially since the rare diseases we are studying are commonly under diagnosed. We also may not be able to identify, recruit, and enroll a sufficient number of patients to complete our clinical studies because of the perceived risks and benefits of the product candidate under study, the proximity and availability of clinical study sites for prospective patients, and the patient referral practices of physicians. The availability and efficacy of competing therapies and clinical studies can also adversely impact enrollment. If patients are unwilling to participate in our studies for any reason, the timeline for recruiting patients, conducting studies, and obtaining regulatory approval of potential products may be delayed, the commercial prospects of our product candidates will be harmed, and our ability to generate product revenue from any of these product candidates could be delayed or prevented. In addition, any delays in completing our clinical studies will increase our costs, slow down our product candidate development and approval process, and jeopardize our ability to commence product sales and generate revenue. Any of these occurrences may harm our business, financial condition, and prospects significantly. In addition, many of the factors that cause, or lead to, a delay in the commencement or completion of clinical studies may also ultimately lead to the denial of regulatory approval of our product candidates.
The regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time consuming, and inherently unpredictable. Even if we achieve positive results in our pre-clinical and clinical studies, if we are ultimately unable to obtain timely regulatory approval for our product candidates, our business will be substantially harmed.
Our future success is dependent on our ability to successfully commercialize our product and develop, obtain regulatory approval for, and then successfully commercialize one or more product candidates. We are not permitted to market or promote any of our product candidates before we receive regulatory approval from the FDA or comparable foreign regulatory authorities. We have only obtained regulatory approval for one product, and it is possible that none of our existing product candidates or any product candidates we may seek to develop in the future will ever obtain regulatory approval.
To obtain regulatory approval in the United States and other jurisdictions, we must comply with numerous and varying requirements regarding safety, efficacy, chemistry, manufacturing and controls, clinical studies (including good clinical practices), commercial sales, pricing, and distribution of our product candidates. Even if we are successful in obtaining approval in one jurisdiction, we cannot ensure that we will obtain approval in any other jurisdictions. In addition, approval policies, regulations, positions of the regulatory agencies on study design and/or endpoints, or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development, which may cause delays in the approval or the decision not to approve an application. Communications with the regulatory agencies during the approval process are also unpredictable; favorable communications early in the process do not ensure that approval will be obtained and unfavorable communications early on do not guarantee that approval will not be obtained. If we are unable to obtain approval for our product candidates in multiple jurisdictions, our revenue and results of operations could be negatively affected. Applications for our product candidates could fail to receive regulatory approval, or could be delayed in receiving regulatory approval, for many reasons, including but not limited to the following:
|
|
• |
the FDA or comparable foreign regulatory authorities may disagree with the design, implementation, or conduct of our clinical studies; |
|
|
• |
the FDA or other comparable foreign regulatory authorities may change their guidance or requirements for a development program for a product candidate; |
|
|
• |
the population studied in the clinical program may not be sufficiently broad or representative to assure efficacy and safety in the full population for which we seek approval; |
|
|
• |
the FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from nonclinical studies or clinical studies; |
|
|
• |
the data collected from clinical studies of our product candidates may not be sufficient to support the submission of an NDA, or biologics license application, or BLA, or other submission or to obtain regulatory approval in the United States or elsewhere; |
|
|
• |
we may be unable to demonstrate to the FDA or comparable foreign regulatory authorities that a product candidate’s risk-benefit ratio for its proposed indication is acceptable; |
|
|
• |
the FDA or comparable foreign regulatory authorities may fail to approve the manufacturing processes, test procedures and specifications, or facilities of third-party manufacturers with which we contract for clinical and commercial supplies; |
|
|
• |
failure of our nonclinical or clinical development to comply with an agreed upon Pediatric Investigational Plan (PIP), which details the designs and completion timelines for nonclinical and clinical studies and is a condition of marketing authorization in the EU; and |
33
|
|
• |
the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval. |
Furthermore, the disease states we are evaluating often will not have clear regulatory paths for approval and/or do not have validated outcome measures. In these circumstances, we work closely with the regulatory authorities to define the approval path and may have to qualify outcome measures as part of our development programs. For example, for patients with XLH there is no available regulatory precedent describing requirements for obtaining approval to treat this disease, and there are no validated patient-reported outcome measures that are specific to this disease. Additionally, many of the disease states we are targeting are highly heterogeneous in nature, which may impact our ability to determine the treatment benefit of our potential therapies. For example, patients with FAOD and Glut1 DS have a highly heterogeneous disease course, which may impact our ability to determine the true treatment benefit of our product candidates in these patients.
This lengthy and uncertain approval process, as well as the unpredictability of the clinical and nonclinical studies, may result in our failure to obtain regulatory approval to market any of our product candidates, or delayed regulatory approval, which would significantly harm our business, results of operations, and prospects.
FDA, the U.S. National Institutes of Health, or NIH, Health Canada, and the EMA have demonstrated caution in their regulation of gene therapy treatments, and ethical and legal concerns about gene therapy and genetic testing may result in additional regulations or restrictions on the development and commercialization of our gene therapy product candidates, which may be difficult to predict.
The clinical trial requirements of FDA, the NIH, Health Canada, the EMA and other regulatory authorities and the criteria these regulators use to determine the safety and efficacy of a product candidate vary substantially according to the type, complexity, novelty and intended use and market of the product candidate. The regulatory approval process for novel product candidates such as our gene therapy product candidates can be more expensive and take longer than for other, better known or more extensively studied product candidates. Only one AAV gene therapy product, Glybera from uniQure N.V., or uniQure, has received marketing authorization from the European Commission and one AAV gene therapy product, LUXTURNA™ (voretigene neparvovec) from Spark Therapeutics, has been approved in the United States. Different or additional preclinical studies or clinical trials may be required to support regulatory approval in each respective jurisdiction.
Additionally, FDA, the NIH, Health Canada, and the EMA have each expressed interest in further regulating biotechnology, including gene therapy and genetic testing. For example, the EMA advocates a risk-based approach to the development of a gene therapy product. Agencies at both the federal and state level in the United States, as well as U.S. congressional committees and foreign governments, have also expressed interest in further regulating the biotechnology industry. Such action may delay or prevent commercialization of some or all of our product candidates. For example, in 1999, a patient suffering from OTC deficiency died during a gene therapy clinical trial that utilized an adenovirus vector. It was discovered that adenoviruses could generate an extreme immune system reaction that can be life-threatening. Thereafter, in January 2000, FDA halted that trial and began investigating 69 other gene therapy trials underway in the United States. Eventually, 28 trials were reviewed, with 13 requiring remedial action. Subsequently, in 2003, FDA suspended 27 additional gene therapy trials involving several hundred patients after learning that a child treated in France had developed leukemia.
Regulatory requirements in the United States and abroad governing gene therapy products have changed frequently and may continue to change in the future. FDA has established the Office of Cellular, Tissue and Gene Therapies within its Center for Biologics Evaluation and Research to consolidate the review of gene therapy and related products, and has established the Cellular, Tissue and Gene Therapies Advisory Committee to advise this review. Prior to submitting an IND, our human gene therapy clinical trials are subject to review by the NIH Office of Biotechnology Activities’, or OBA’s, Recombinant DNA Advisory Committee, or the RAC. As of April 2016, the updated NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules, including gene therapy, provide the opportunity for one or more oversight bodies (institutional review board, or IRB, or the institutional biosafety committee, or IBC) to request a public RAC review based on their own review of the protocol and NIH requirements. Regardless of the request for public review, NIH RAC members make their own assessment as to whether the protocol would significantly benefit from a public RAC review. The RAC’s recommendations are shared with FDA and the oversight bodies. The RAC can delay the initiation of a clinical trial, even if FDA has reviewed the trial design and details and has not objected to its initiation or has notified the sponsor that the study may begin. Conversely, FDA can put an IND on a clinical hold even if the RAC has provided a favorable review or has recommended against an in-depth, public review. Moreover, under guidelines published by the NIH, patient enrollment in our gene therapy clinical trials cannot begin until, among other things, the investigator for that clinical trial has received a letter from the OBA indicating that the protocol registration process has been completed. Upon receipt of the letter from OBA confirming completion of protocol registration the investigator may obtain final approval from the oversight bodies and patient enrollment may begin if all other applicable regulatory authorizations have been obtained.
If there is a public RAC review, the receipt of the final recommendation letter concludes the protocol registration process and then oversight body, or bodies, approval can be issued. While the RAC completed its initial public review for DTX301 and DTX201, approving the protocols and issuing written recommendations, the RAC will continue to review DTX301 and DTX201, and may recommend additional public reviews in the future with respect to DTX301, DTX201, or any of our other product candidates. In
34
addition, adverse developments in clinical trials of gene therapy products conducted by others may cause FDA or other oversight bodies to change the requirements for approval of any of our product candidates. Similarly, the EMA governs the development of gene therapies in the EU and may issue new guidelines concerning the development and marketing authorization for gene therapy products and require that we comply with these new guidelines.
These regulatory review committees and advisory groups and the new guidelines they promulgate may lengthen the regulatory review process, require us to perform additional studies or trials, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of our product candidates or lead to significant post- approval limitations or restrictions. As we advance our product candidates, we will be required to consult with these regulatory and advisory groups and comply with applicable guidelines. If we fail to do so, we may be required to delay or discontinue development of such product candidates. These additional processes may result in a review and approval process that is longer than we otherwise would have expected. Delays as a result of an increased or lengthier regulatory approval process or further restrictions on the development of our product candidates can be costly and could negatively impact our or our collaborators’ ability to complete clinical trials and commercialize our current and future product candidates in a timely manner, if at all.
Our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, limit the commercial profile of an approved label, or result in significant negative consequences following marketing approval, if any.
Undesirable side effects caused by our product candidates could cause us or regulatory authorities to interrupt, delay, or halt clinical studies or further development, and could result in a more restrictive label, the delay or denial of regulatory approval by the FDA or other comparable foreign authorities, or a Risk Evaluation and Mitigation Strategy, or REMS, plan, which could include a medication guide outlining the risks of such side effects for distribution to patients, restricted distribution, a communication plan for healthcare providers, and/or other elements to assure safe use. Some of our product candidates are in the early stages of development and the safety profile has not been established. For example, in completed Phase 2 study, LC-FAOD patients treated with UX007 experienced treatment-related adverse events, the most common of which were diarrhea, abdominal/gastrointestinal pain and vomiting. There were no deaths, but there was one treatment-related serious adverse event of moderate gastroenteritis with vomiting. In a completed Phase 2 seizure study, Glut1 DS patients with seizures treated with UX007 experienced treatment-related adverse events, the most common of which were vomiting, diarrhea, and abdominal pain. There were no deaths, and no treatment-related serious adverse events. Gene therapy product candidates using AAV vectors, like DTX301, have been associated with immunologic reaction to the capsid protein or gene at early time points after administration. For example, in our recently discontinued Phase 1/2 clinical trial of DTX101 in hemophilia B, we observed elevated laboratory alanine transaminase levels, or ALTs. In previous clinical trials involving AAV viral vectors for gene therapy, some subjects experienced adverse events, including the development of a T-cell mediated immune response against the vector capsid proteins. In addition, theoretical side effects of AAV vectors include replication and spread of the virus to other parts of the body and insertional oncogenesis, which is the process whereby the insertion of a gene near a gene that is important in cell growth or division results in uncontrolled cell division, which could potentially enhance the risk of malignant transformation or cancer. Potential procedure-related events are similar to those associated with standard coronary diagnostic procedures, and may include vascular injury (e.g., damage to the femoral, radial or brachial arteries at the site of vascular access, or damage to the coronary arteries) or myocardial injury. Future product candidates may also cause these or similar side effects as development proceeds. Results of our studies or investigator-sponsored trials could reveal a high and unacceptable severity and prevalence of these or other side effects. In such an event, our studies could be suspended or terminated, and the FDA or comparable foreign regulatory authorities could order us to cease further development of or deny or withdraw approval of our product candidates for any or all targeted indications.
Drug-related side effects could affect patient recruitment and the ability of enrolled patients to complete a study. Such side effects could also result in potential product liability claims. We currently carry product liability insurance in the amount of $10.0 million per incident and $10.0 million in the aggregate, and we are required to maintain product liability insurance pursuant to certain of our agreements. We believe our product liability insurance coverage is sufficient in light of our current clinical programs; however, we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability, or losses may exceed the amount of insurance that we carry. A product liability claim or series of claims brought against us could cause our stock price to decline and, if judgments exceed our insurance coverage, could adversely affect our results of operations and business. In addition, regardless of merit or eventual outcome, product liability claims may result in impairment of our business reputation, withdrawal of clinical study participants, costs due to related litigation, distraction of management’s attention from our primary business, initiation of investigations by regulators, substantial monetary awards to patients or other claimants, the inability to commercialize our product candidates, and decreased demand for our product candidates, if approved for commercial sale.
Additionally, even though we received regulatory approval for Mepsevii and even if our product candidates receive marketing approval in the future, if we or others later identify undesirable side effects caused by such products, a number of potentially significant negative consequences could result, including but not limited to:
|
|
• |
regulatory authorities may withdraw approvals of such product; |
|
|
• |
regulatory authorities may require additional warnings on the product’s label or restrict the product’s approved use; |
35
|
|
• |
patients and physicians may elect not to use our products, or reimbursement authorities may elect not to reimburse for them; and |
|
|
• |
our reputation may suffer. |
Any of these events could prevent us from achieving or maintaining market acceptance of the particular product candidate, if approved, and could significantly harm our business, results of operations, and prospects.
If preclinical studies for DTX401 for GSDIa, DTX201 for hemophilia A, DTX701 for Wilson disease, DTX501 for PKU, and DTX601 for citrullinemia type I, or all of our future product candidates do not result in the determination of a minimally effective dose range, we may not obtain the regulatory approvals required to initiate clinical testing.
As with any systemically delivered adeno-associated virus, or AAV, gene therapy, it is important that we accurately determine a minimally effective dose in order to successfully execute our clinical trial. Exposure to the AAV virus has been shown to induce the production of neutralizing antibodies, which can reduce or eliminate the therapeutic effect of subsequently administered intravenous AAV therapies such as our product candidates. Because of the potential for immune response producing neutralizing antibodies making patients ineligible for a second dose of that vector, clinical trials are required to determine the minimum effective dose and the maximum safe dose. If our preclinical studies fail to demonstrate a starting dose in the clinic that might be reasonably expected to result in a clinical benefit, regulatory agencies may not approve the start of our clinical trials. In addition, even if we start our clinical program, we may not be able to recruit patients who will seek assurance of a clinical benefit following administration of our therapy.
Even if we obtain regulatory approval for our product candidates, our products will remain subject to regulatory scrutiny.
Our product and any product candidates that are approved will be subject to ongoing regulatory requirements for manufacturing, labeling, packaging, storage, distribution, advertising, promotion, sampling, record-keeping, conduct of post-marketing studies, and submission of safety, efficacy, and other post-market information, including both federal and state requirements in the United States and requirements of comparable foreign regulatory authorities.
Manufacturers and manufacturers’ facilities are required to comply with extensive FDA, and comparable foreign regulatory authority, requirements, including ensuring that quality control and manufacturing procedures conform to Good Manufacturing Practices (GMP) regulations. As such, we and our contract manufacturers will be subject to continual review and inspection to assess compliance with GMP and adherence to commitments made in any NDA, BLA, MAA, or other comparable application for approval in another jurisdiction. Accordingly, we and others with whom we work must continue to expend time, money, and effort in all areas of regulatory compliance, including manufacturing, production, and quality control.
Any regulatory approvals that we receive for our product candidates may be subject to limitations on the approved indicated uses for which the product may be marketed or other conditions of approval, or contain requirements for potentially costly post-marketing testing, including Phase IV clinical studies, and surveillance to monitor the safety and efficacy of the product candidate. We could also be asked to conduct post-marketing clinical studies to verify the safety and efficacy of our products in general or in specific patient subsets. If original marketing approval was obtained via the accelerated approval or conditional marketing authorization pathways, we would be required to conduct a successful post-marketing clinical study to confirm clinical benefit for our products. An unsuccessful post-marketing study or failure to complete such a study could result in the withdrawal of marketing approval. We will be required to report certain adverse events and manufacturing problems, if any, to the FDA and comparable foreign regulatory authorities. Any new legislation addressing drug safety issues could result in delays in product development or commercialization, or increased costs to assure compliance. We will have to comply with requirements concerning advertising and promotion for our products. Promotional communications with respect to prescription drugs are subject to a variety of legal and regulatory restrictions and must be consistent with the information in the product’s approved label. As such, we may promote our products only for indications or uses for which they have approval. The holder of an approved NDA, BLA, MAA, or other comparable application must submit new or supplemental applications and obtain approval for certain changes to the approved product, product labeling, or manufacturing process.
If we fail to comply with applicable regulatory requirements, or there are safety or efficacy problems with a product, a regulatory agency or enforcement authority may, among other things:
|
|
• |
issue warning or notice of violation letters; |
|
|
• |
impose civil or criminal penalties; |
|
|
• |
suspend or withdraw regulatory approval; |
|
|
• |
suspend any of our ongoing clinical studies; |
|
|
• |
refuse to approve pending applications or supplements to approved applications submitted by us; |
|
|
• |
impose restrictions on our operations, including closing our contract manufacturers’ facilities; |
36
|
|
• |
require entry into a consent decree. |
Any government investigation of alleged violations of law could require us to expend significant time and resources in response, and could generate negative publicity. Any failure to comply with ongoing regulatory requirements may significantly and adversely affect our ability to commercialize and generate revenue from our products. If regulatory sanctions are applied or if regulatory approval is withdrawn, the value of our company and our operating results will be adversely affected.
Our gene therapy approach utilizes vectors derived from viruses, which may be perceived as unsafe or may result in unforeseen adverse events. Negative public opinion and increased regulatory scrutiny of gene therapy may damage public perception of the safety of our product candidates and adversely affect our ability to conduct our business or obtain regulatory approvals for our product candidates.
Gene therapy remains a novel technology. Public perception may be influenced by claims that gene therapy is unsafe, and gene therapy may not gain the acceptance of the public or the medical community. For example, earlier gene therapy trials led to several well- publicized adverse events, including cases of leukemia and death seen in other trials using other vectors. The risk of cancer remains a concern for gene therapy and we cannot assure you that it will not occur in any of our planned or future clinical studies. In addition, there is the potential risk of delayed adverse events following exposure to gene therapy products due to persistent biological activity of the genetic material or other components of products used to carry the genetic material. Serious adverse events in our clinical trials, or other clinical trials involving gene therapy products, particularly AAV gene therapy products such as candidates based on the same capsid serotypes as our product candidates, or occurring during use of our competitors’ products, even if not ultimately attributable to the relevant product candidates, and the resulting publicity, could result in increased government regulation, unfavorable public perception, potential regulatory delays in the testing or approval of our gene therapy product candidates, stricter labeling requirements for those gene therapy product candidates that are approved and a decrease in demand for any such gene therapy product candidates, all of which would have an adverse effect on our business, financial condition, results of operations and prospects.
Our transition from HEK293 to a HeLa platform may require additional toxicology and comparability studies for gene therapy product candidates, which may result in delays to the approval process for our current or future programs and increased costs resulting from additional preclinical trials.
We have conducted some of our preclinical evaluations with viral vectors produced on adherent and suspension platforms utilizing human embryonic kidney 293, or HEK293, and HeLa cells, the latter an immortal cell line used in scientific research. HeLa is the oldest and most commonly used human cell line. We are conducting our Phase 1/2 trial of DTX301, and plan to conduct our Phase 1/2 trials of DTX401, and potentially other programs using viral vectors produced on the HEK293 platform. We plan to conduct our Phase 1/2 trial of DTX201 using viral vectors produced on the HeLa platform. For Phase 3 studies and commercial production of each of our gene therapy product candidates, we plan to use HeLa. Even if we successfully complete our planned preclinical studies and clinical trials using vectors produced on our adherent and suspension HEK293 and HeLa platforms, FDA or other regulatory authorities may require additional toxicology and/or a clinical bridge study, or comparability study, the latter showing comparability of vectors produced on the HeLa platform prior to commencing Phase 3 trials of DTX301, DTX401, DTX701, DTX501, and DTX601 delaying the development process. For example, while FDA has agreed to our plan, other regulators may require additional assessments of HeLa material during the manufacturing of our products as well as additional animal studies. In addition, if we make manufacturing or formulation changes to our product candidates in the future, we may need to conduct additional preclinical studies to bridge our modified product candidates to earlier versions.
If we are unable to identify, source, and develop effective predictive biomarkers, or our collaborators are unable to successfully develop and commercialize companion diagnostics for our product candidates, or experience significant delays in doing so, we may not realize the full commercial potential of our product candidates.
We currently anticipate that we will need companion diagnostics to determine whether or not we can dose a particular patient with each of our products. We expect to use predictive biomarkers to identify the right patients for certain of our product candidates. For example, to evaluate therapeutic response of DTX301, we plan to measure ammonia levels and other biomarkers, including 13C-acetate, which are established measures of OTC deficiency disease status and ureagenesis. We cannot assure you that 13C-acetate or any other future potential biomarker will in fact prove predictive, be reliably sourced, or be accepted by the FDA or other regulatory authorities. In addition, our success may depend, in part, on the development and commercialization of companion diagnostics. We also expect the FDA will require the development and regulatory approval of a companion diagnostic assay as a condition to approval of DTX301. There has been limited success to date industrywide in developing and commercializing these types of companion diagnostics. Development and manufacturing of companion diagnostics is complex and there are limited manufacturers with the necessary expertise and capability. Even if we are able to find a qualified collaborator, it may not be able to manufacture the companion diagnostics at a cost or in quantities or on timelines necessary for use with our product candidates. To be successful, we need to address a number of scientific, technical and logistical challenges. We have not yet initiated development and commercialization of companion diagnostics. We have little experience in the development and commercialization of diagnostics and
37
may not be successful in developing and commercializing appropriate diagnostics to pair with any of our product candidates that receive marketing approval. University of Pennsylvania School of Medicine currently conducts some of our clinical assays pursuant to a sponsored research agreement, one of which is required for our ongoing Phase 1/2 clinical trial. We intend to enter into agreements with third parties for the automation, characterization and validation, of our companion diagnostic and the manufacture of its critical reagents. However, we may be unable to enter into any such agreement on favorable terms, or at all.
Companion diagnostics are subject to regulation by FDA and similar regulatory authorities outside the United States as medical devices and require regulatory clearance or approval prior to commercialization. In the United States, companion diagnostics are cleared or approved through FDA’s 510(k) premarket notification or premarket approval, or PMA, process. Changes in marketing approval policies during the development period, changes in or the enactment of additional statutes or regulations, or changes in regulatory review for each submitted 510(k) premarket notification, PMA or equivalent application types in jurisdictions outside the United States, may cause delays in the approval, clearance or rejection of an application. Given our limited experience in developing and commercializing diagnostics, we expect to rely in part or in whole on third parties for companion diagnostic design and commercialization. We and our collaborators may encounter difficulties in developing and obtaining approval or clearance for the companion diagnostics, including issues relating to selectivity/specificity, analytical validation, reproducibility, or clinical validation. Any delay or failure by us or our collaborators to develop or obtain regulatory approval of the companion diagnostics could delay or prevent approval of our product candidates.
Risks Related to our Reliance on Third Parties
We rely on third parties to conduct our nonclinical and clinical studies and perform other tasks for us. If these third parties do not successfully carry out their contractual duties, meet expected deadlines, or comply with regulatory requirements, we may be exposed to sub-optimal quality and reputational harm, we may not be able to obtain regulatory approval for or commercialize our product candidates, and our business could be substantially harmed.
We have relied upon and plan to continue to rely upon third parties, including CROs, collaborative partners, and independent investigators to analyze, collect, monitor, and manage data for our ongoing nonclinical and clinical programs. We rely on third parties for execution of our nonclinical and clinical studies, and for estimates regarding costs and efforts completed, and we control only certain aspects of their activities. For example, we will rely on our partner Arcturus for the design and optimization of initial product candidates under our messenger RNA collaboration. Nevertheless, we are responsible for ensuring that each of our studies is conducted in accordance with the applicable protocol, legal, regulatory, and scientific standards, and our reliance on the CROs and other third parties does not relieve us of our regulatory responsibilities. We and our CROs and other vendors and partners are required to comply with GMP, GCP, and GLP, which are regulations and guidelines enforced by the FDA, the Competent Authorities of the Member States of the European Economic Area, and comparable foreign regulatory authorities for all of our product candidates in development. Regulatory authorities enforce these regulations through periodic inspections of study sponsors, principal investigators, study sites, and other contractors. If we or any of our CROs or other vendors and partners, including the sites at which clinical studies are conducted, fail to comply with applicable regulations, the data generated in our nonclinical and clinical studies may be deemed unreliable and the FDA, EMA, or comparable foreign regulatory authorities may deny approval and/or require us to perform additional nonclinical and clinical studies before approving our marketing applications, which would delay the approval process. We cannot make assurances that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical studies comply with GCP regulations or that nonclinical studies comply with GLP regulations. In addition, our clinical studies must be conducted with products produced under GMP regulations. If the regulatory authorities determine that we have failed to comply with GLP, GMP, or GCP regulations, they may deny approval of our product candidates and/or we may be required to repeat clinical or nonclinical studies, which would delay the regulatory approval process.
Our CROs and other vendors and partners are not our employees, and except for remedies available to us under our agreements with such third parties, we cannot control whether or not they devote sufficient time and resources to our on-going nonclinical and clinical programs. If our vendors and partners do not successfully carry out their contractual duties or obligations or meet expected deadlines, if they need to be replaced, or if the quality or accuracy of the data they obtain is compromised due to the failure to adhere to our protocols, regulatory requirements, or for other reasons, our clinical studies may be extended, delayed, or terminated, and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates. CROs and other vendors and partners may also generate higher costs than anticipated as a result of changes in scope of work or otherwise. As a result, our results of operations and the commercial prospects for our product candidates would be harmed, our costs could increase, and our ability to generate revenue could be delayed.
If any of our relationships with these third parties terminate, we may not be able to enter into arrangements with alternative vendors or do so on commercially reasonable terms. Switching or adding additional vendors involves additional cost and requires management time and focus. In addition, there is a natural transition period when a new vendor commences work. As a result, delays may occur, which can materially impact our ability to meet our desired clinical development timelines. Though we carefully manage our relationships with our vendors and partners, there can be no assurance that we will not encounter similar challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, financial condition, and business prospects.
38
We also rely on third parties in other ways, including efforts to support patient identification, to assist our finance and legal departments, and to provide other resources for our business. Use of these third parties could expose us to sub-optimal quality, missed deadlines, and non-compliance with applicable laws, all of which could result in reputational harm to us and negatively affect our business.
We are dependent on KHK for the clinical and commercial supply of burosumab for all major markets and for the development and commercialization of burosumab in certain major markets, and KHK’s failure to provide an adequate supply of burosumab or to commercialize burosumab in those markets could result in a material adverse effect on our business and operating results.
Under our agreement with KHK, KHK has the sole right to commercialize burosumab in Europe and, at a specified time, in the United States, Canada, and Turkey, subject to a limited promotion right we retained. Our development partnership with KHK may not be successful, and we may not realize the expected benefits from such partnership, due to a number of important factors, including but not limited to the following:
|
|
• |
KHK has no obligation under our agreement to use diligent efforts to commercialize burosumab in Europe. The timing and amount of any royalty payments we may receive under our agreement will depend on, among other things, the efforts, allocation of resources, and successful commercialization of burosumab by KHK in Europe. Additionally, if KHK were to decide not to commercialize burosumab in Europe, and we nevertheless wished to commercialize burosumab in Europe, we would need to renegotiate with KHK certain terms of our agreement, which we may be unable to do on reasonable terms in a timely manner, or at all; |
|
|
• |
the timing and amount of any royalty payments we may receive under our agreement with KHK will depend on, among other things, the efforts, allocation of resources, and successful commercialization of burosumab by KHK in the United States and Canada under our agreement; |
|
|
• |
KHK may change the focus of its commercialization efforts or pursue higher-priority programs; |
|
|
• |
KHK may make decisions regarding the indications for our product candidates in countries where it has the sole right to commercialize the product candidates that limit commercialization efforts in those countries or in countries where we have the right to commercialize our product candidates; |
|
|
• |
KHK may make decisions regarding market access and pricing in countries where it has the sole right to commercialize our product candidates which can negatively impact our commercialization efforts in countries where we have the right to commercialize our product candidates; |
|
|
• |
KHK may fail to manufacture or supply sufficient drug product of burosumab in compliance with applicable laws and regulations or otherwise for our development and clinical use, which could result in program delays; |
|
|
• |
KHK may fail to manufacture or supply sufficient drug product of burosumab in compliance with applicable laws and regulations or otherwise for our commercial use, if approved, which could result in lost revenue; |
|
|
• |
KHK may elect to develop and commercialize burosumab indications with a larger market than XLH and at a lower price, thereby reducing the profit margin on sales of burosumab for any orphan indications, including XLH; |
|
|
• |
if KHK were to breach or terminate the agreement with us, we would no longer have any rights to develop or commercialize burosumab or such rights would be limited to non-terminated countries; |
|
|
• |
KHK may terminate its agreement with us, adversely affecting our potential revenue from licensed products; and |
|
|
• |
the timing and amounts of expense reimbursement that we may receive are uncertain, and the total expenses for which we are obligated to reimburse KHK may be greater than anticipated. |
39
We rely on third parties to manufacture our product and most of our product candidates. Our business could be harmed if those third parties fail to provide us with sufficient quantities of drug product, or fail to do so at acceptable quality levels or prices.
We have limited infrastructure or capability internally to manufacture our product and product candidates, and we lack the resources and the capability to manufacture most of our product candidates on a clinical or commercial scale. We rely on our manufacturers to purchase from third-party suppliers the materials necessary to produce our product candidates for our clinical studies. There are a limited number of suppliers for raw materials that we use to manufacture our drugs, placebos, or active controls, and there may be a need to identify alternate suppliers to prevent a possible disruption of the manufacture of the materials necessary to produce our product and our product candidates for our clinical studies, and, if approved, ultimately for commercial sale. We do not have any control over the process or timing of the acquisition of these raw materials by our manufacturers. Although we generally do not begin a clinical study unless we believe we have a sufficient supply of a product candidate to complete such study, any significant delay or discontinuity in the supply of a product candidate, or the raw material components thereof, for an ongoing clinical study due to, among other things, the failure of a manufacturer to provide a drug substance or drug product of sufficient quantity or quality, or the need to replace a third-party manufacturer could considerably delay completion of our clinical studies, product testing, and potential regulatory approval of our product candidates, and could also impair named patient sale supply of our product candidates, which could harm our business and results of operations.
We expect our manufacturing strategy will involve the use of one or more CMOs as well as establishing our own capabilities and infrastructure, including at our Woburn, MA facility where we will support continued innovation in vector optimization and development of manufacturing processes required for IND-enabling studies and the reliable production of high quality AAV vectors at commercial scale. We expect that development of our own process development facility will provide us with enhanced control of material supply for both clinical trials and the commercial market, enable the more rapid implementation of process changes, and allow for better long-term margins. However, we have no experience as a company in developing a manufacturing facility and may never be successful in developing our own manufacturing facility or capability. Additionally, given that cGMP gene therapy manufacturing is a nascent industry, there are a small number of CMOs with the experience necessary to manufacture our gene therapy product candidates and we may have difficulty finding or maintaining relationships with such CMOs or hiring experts for internal manufacturing and accordingly, our production capacity may be limited. We may establish multiple manufacturing facilities as we expand our commercial footprint to multiple geographies, which may lead to regulatory delays or prove costly. Even if we are successful, our manufacturing capabilities could be affected by cost-overruns, unexpected delays, equipment failures, lack of capacity, labor shortages, natural disasters, power failures and numerous other factors that could prevent us from realizing the intended benefits of our manufacturing strategy and have a material adverse effect on our business.
Gene therapy products and product candidates are novel, complex, expensive and difficult to manufacture. We could experience manufacturing problems that result in delays in our gene therapy development or commercialization programs or otherwise harm our business.
The manufacturing process used to produce our gene therapy product candidates is complex, novel and has not been validated for commercial use. Several factors could cause production interruptions, including equipment malfunctions, regulatory inspections, facility contamination, raw material shortages or contamination, natural disasters, disruption in utility services, human error or disruptions in the operations of our suppliers.
Our gene therapy product candidates require processing steps that are more complex than those required for most small molecule drugs. Moreover, unlike small molecules, the physical and chemical properties of a biologic such as our gene therapy ones generally cannot be fully characterized. As a result, assays of the finished product candidate may not be sufficient to ensure that the product candidate is consistent from lot to lot or will perform in the intended manner. Accordingly, we employ multiple steps to control the manufacturing process to assure that the process works reproducibly and the product candidate is made strictly and consistently in compliance with the process. Problems with the manufacturing process, even minor deviations from the normal process, could result in product defects or manufacturing failures that result in lot failures, noncompliance with regulatory requirements, product recalls, product liability claims or insufficient inventory. We may encounter problems achieving adequate quantities and quality of clinical-grade materials that meet FDA, the EMA or other applicable standards or specifications with consistent and acceptable production yields and costs.
In addition, FDA, the EMA and other foreign regulatory authorities may require us to submit samples of any lot of any approved product together with the protocols showing the results of applicable tests at any time. Under some circumstances, FDA, the EMA or other foreign regulatory authorities may require that we not distribute a lot until the agency authorizes its release. Slight deviations in the manufacturing process, including those affecting quality attributes and stability, may result in unacceptable changes in the product that could result in lot failures or product recalls. Lot failures or product recalls could cause us to delay product launches or clinical trials, which could be costly to us and otherwise harm our business, financial condition, results of operations and prospects.
We also may encounter problems hiring and retaining the experienced scientific, quality-control and manufacturing personnel needed to operate our gene therapy manufacturing processes, which could result in delays in production or difficulties in maintaining compliance with applicable regulatory requirements. We also may not be able to complete scaling up of our facility in Woburn, MA,
40
and this facility may not enable the expansion of our internal manufacturing process discovery and development to the extent we anticipate, or at all.
We are subject to a multitude of manufacturing risks, any of which could substantially increase our costs and limit the supply of our product candidates.
The process of manufacturing our product and product candidates is complex, highly regulated, and subject to several risks, including but not limited to those listed below.
|
|
• |
The process of manufacturing our product and product candidates is extremely susceptible to product loss due to contamination, equipment failure or improper installation or operation of equipment, or vendor or operator error. Even minor deviations from normal manufacturing processes for our product and any of our product candidates could result in reduced production yields, product defects, and other supply disruptions. If microbial, viral, or other contaminations are discovered in our product and product candidates or in the manufacturing facilities in which our product and product candidates are made, such manufacturing facilities may need to be closed for an extended period of time to investigate and remedy the contamination. |
|
|
• |
The manufacturing facilities in which our product and product candidates are made could be adversely affected by equipment failures, labor shortages, raw material shortages, natural disasters, power failures, and numerous other factors. |
Any adverse developments affecting manufacturing operations for our product and product candidates may result in shipment delays, inventory shortages, lot failures, withdrawals or recalls, or other interruptions in the supply of our product and product candidates. Due to their stage of development, small volume requirements, and infrequency of batch production runs, we carry limited amounts of safety stock for our product and product candidates. We may also have to take inventory write-offs and incur other charges and expenses for product and product candidates that fail to meet specifications, undertake costly remediation efforts, or seek more costly manufacturing alternatives.
The drug substance and drug product for our product and most of our product candidates are currently acquired from single-source suppliers. The loss of these suppliers, or their failure to supply us with the necessary drug substance or drug product, could materially and adversely affect our business.
We acquire most of the drug substances and drug products for our product and product candidates from single sources. The drug substance and drug product for burosumab are made by KHK pursuant to our license and collaboration agreement with KHK. The drug substance and drug product for Mepsevii are manufactured by Rentschler under a commercial supply and services agreement and accompanying purchase orders. The pharmaceutical-grade drug substance for UX007 is manufactured by IOI Oleo pursuant to our supply agreement with IOI Oleo, and the drug product for UX007 is prepared by Haupt Pharma AG and CPM pursuant to purchase orders. For DTX301, a CMO manufactures clinical materials pursuant to cGMP requirements. We have not currently secured any other suppliers for the drug substance or drug product of our product and product candidates and, although we believe that there are alternate sources of supply that could satisfy our clinical and commercial requirements, we cannot provide assurance that identifying alternate sources and establishing relationships with such sources would not result in significant delay in the commercialization of our product or the development of our product candidates. Additionally, we may not be able to enter into supply arrangements with alternative suppliers on commercially reasonable terms or at all. A delay in the commercialization of our product or the development of our product candidates or having to enter into a new agreement with a different third-party on less favorable terms than we have with our current suppliers could have a material adverse impact upon on our business.
41
We and our collaborators and contract manufacturers are subject to significant regulation with respect to manufacturing our product candidates. The manufacturing facilities on which we rely may not continue to meet regulatory requirements or may not be able to meet supply demands.
All entities involved in the preparation of therapeutics for clinical studies or commercial sale, including our existing contract manufacturers and collaboration partners for our product candidates, are subject to extensive regulation. Components of a finished therapeutic product approved for commercial sale or used in clinical studies must be manufactured in accordance with GMP. These regulations govern manufacturing processes and procedures (including record keeping) and the implementation and operation of quality systems to control and assure the quality of investigational products and products approved for sale. Poor control of production processes can lead to the introduction of contaminants or to inadvertent changes in the properties or stability of our product candidates that may not be detectable in final product testing. We, our collaborators, or our contract manufacturers must supply all necessary documentation in support of an NDA, BLA, MAA, or other application for regulatory approval, on a timely basis and must adhere to GLP, GMP, and similar regulations enforced by the FDA and other regulatory agencies through their facilities inspection programs. Some of our contract manufacturers have never produced a commercially approved pharmaceutical product and therefore have not obtained the requisite regulatory authority approvals to do so. The facilities and quality systems of some or all of our collaborators and third-party contractors must pass a pre-approval inspection for compliance with the applicable regulations as a condition of regulatory approval of our product candidates or any of our other potential products. In addition, the regulatory authorities may, at any time, audit or inspect a manufacturing facility involved with the preparation of our product candidates or our other potential products or the associated quality systems for compliance with the regulations applicable to the activities being conducted. Although we oversee the contract manufacturers, we cannot control the manufacturing process of, and are substantially dependent on, our contract manufacturing partners for compliance with the regulatory requirements. If these facilities cannot schedule manufacturing to meet inspectional demands or do not pass a pre-approval plant inspection, regulatory approval of the products may not be granted or may be substantially delayed until any violations are corrected to the satisfaction of the regulatory authority, if ever.
The regulatory authorities also may, at any time following approval of a product for sale, audit the manufacturing facilities of our collaborators and third-party contractors. If any such inspection or audit identifies a failure to comply with applicable regulations or if a violation of our product specifications or applicable regulations occurs independent of such an inspection or audit, we or the relevant regulatory authority may require remedial measures that may be costly and/or time consuming for us or a third-party to implement, and that may include the temporary or permanent suspension of a clinical study or commercial sales or the temporary or permanent closure of a facility. Any such remedial measures imposed upon us or third parties with whom we contract could materially harm our business.
If we, our collaborators, or any of our third-party manufacturers fail to maintain regulatory compliance, the FDA or other applicable regulatory authority can impose regulatory sanctions including, among other things, refusal to approve a pending application for a new drug product or biologic product, withdrawal of an approval, or suspension of production. As a result, our business, financial condition, and results of operations may be materially harmed.
Additionally, if supply from one approved manufacturer is interrupted, an alternative manufacturer would need to be qualified through an NDA or BLA supplement or MAA variation, or equivalent foreign regulatory filing, which could result in further delay. The regulatory agencies may also require additional studies if a new manufacturer is relied upon for commercial production. Switching manufacturers may involve substantial costs and is likely to result in a delay in our desired clinical and commercial timelines.
These factors could cause us to incur higher costs and could cause the delay or termination of clinical studies, regulatory submissions, required approvals, or commercialization of our product candidates. Furthermore, if our suppliers fail to meet contractual requirements and we are unable to secure one or more replacement suppliers capable of production at a substantially equivalent cost, our clinical studies may be delayed or we could lose potential revenue.
The actions of distributors could affect our ability to sell or market products profitably. Fluctuations in buying or distribution patterns by such distributors could adversely affect our revenues, financial condition, or results of operations
We intend to rely on commercial distributors for a considerable portion of our product sales and we expect such sales to be concentrated within a small number of distributors. The financial failure of any of these distributors could adversely affect our revenues, financial condition or results of operations. Our revenues, financial condition or results of operations may also be affected by fluctuations in distributor buying or distribution patterns. These fluctuations may result from seasonality, pricing, wholesaler inventory objectives, or other factors.
42
Our reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them or that our trade secrets will be misappropriated or disclosed.
Because we rely on third parties in connection with the development and manufacture of our product candidates and will likely rely on third parties in connection with the commercialization of our approved products, we must, at times, share trade secrets with them. We seek to protect our proprietary technology in part by entering into confidentiality agreements and, if applicable, material transfer agreements, collaborative research agreements, consulting agreements, letters of engagement, or other similar agreements with our collaborators, advisors, employees, and consultants prior to beginning research or disclosing proprietary information. These agreements typically limit the rights of the third parties to use or disclose our confidential information, such as trade secrets. Despite the contractual provisions employed when working with third parties, the need to share trade secrets and other confidential information increases the risk that such trade secrets become known by our competitors, are inadvertently incorporated into the technology of others, or are disclosed or used in violation of these agreements. Given that our proprietary position is based, in part, on our know-how and trade secrets, a competitor’s discovery of our trade secrets or other unauthorized use or disclosure would impair our competitive position and may have a material adverse effect on our business.
Risks Related to Commercialization of Our Product Candidates
If the market opportunities for our product candidates are smaller than we believe they are, our revenue may be adversely affected, and our business may suffer. Because the target patient populations of our product candidates are small, and the addressable patient population potentially even smaller, we must be able to successfully identify patients and acquire a significant market share to achieve profitability and growth.
We focus our research and product development on treatments for rare and ultra-rare genetic diseases. Given the small number of patients who have the diseases that we are targeting, it is critical to our ability to grow and become profitable that we continue to successfully identify patients with these rare and ultra-rare genetic diseases. Some of our current clinical programs may be most appropriate for patients with more severe forms of their disease. For instance, our Phase 2 study of UX007 in LC-FAOD enrolled patients with more severe disease. In addition, while adults make up the majority of the XLH patients, they often have less severe disease that may reduce the penetration of burosumab in the adult population relative to the pediatric population. Given the overall rarity of the diseases we target, it is difficult to project the prevalence of the more severe forms, or the other subsets of patients that may be most suitable to address with our product candidates, which may further limit the addressable patient population to a small subset. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who have the potential to benefit from treatment with our product candidates, are based on our beliefs and estimates. These estimates have been derived from a variety of sources, including the scientific literature, surveys of clinics, patient foundations, or market research, and may prove to be incorrect. Further, new studies may change the estimated incidence or prevalence of these diseases. The number of patients may turn out to be lower than expected. The effort to identify patients with diseases we seek to treat is in early stages, and we cannot accurately predict the number of patients for whom treatment might be possible. Additionally, the potentially addressable patient population for each of our product candidates may be limited or may not be amenable to treatment with our product candidates, and new patients may become increasingly difficult to identify or access, which would adversely affect our results of operations and our business. Further, even if we obtain significant market share for our product candidates, because the potential target populations are very small we may never become or remain profitable nor generate sufficient revenue growth to sustain our business.
We intend to rely on third-party manufacturers to produce our product candidates. Additionally, these manufacturers do not have experience producing our product candidates at commercial levels and may not achieve the necessary regulatory approvals or produce our product candidates at the cost, quality, quantities, locations, and timing needed to support profitable commercialization.
We have not yet secured manufacturing capabilities for commercial quantities of our product candidates. Although we intend to rely on third-party manufacturers for commercialization, we have only entered into agreements with such manufacturers to support our clinical studies. We may be unable to negotiate binding agreements with the manufacturers to support our commercialization activities on commercially reasonable terms.
Manufacturers may not have the experience or ability to produce our product candidates at commercial levels. We may run into technical or scientific issues related to manufacturing or development that we may be unable to resolve in a timely manner or with available funds. We also have not completed all of the characterization and validation activities necessary for commercialization and regulatory approvals. If our manufacturing partners are not able to conduct all such necessary activities in accordance with applicable regulations, our commercialization efforts will be harmed.
Even if our third-party product manufacturers develop an acceptable manufacturing process, if such third-party manufacturers are unable to produce the necessary quantities of our product candidates, are unable to comply with GMP or other pertinent regulatory requirements, or are unable to produce our product candidates within our planned timeframe and cost parameters, the development and sales of our products, if approved, may be materially harmed.
43
Additionally, the cost to us for the supply of our product and product candidates manufactured by such third parties may be high and could limit our profitability, even if our third-party product manufacturers develop acceptable manufacturing processes that provide the necessary quantities of our product and product candidates in a compliant and timely manner. Furthermore, KHK is our sole supplier of commercial quantities of burosumab. The supply price to us for commercial sales of burosumab, which will be determined on a fixed double-digit percentage of net sales, will be higher than the typical cost of goods sold by companies focused on rare diseases.
We face intense competition and rapid technological change and the possibility that our competitors may develop therapies that are similar, more advanced, or more effective than ours, which may adversely affect our financial condition and our ability to successfully commercialize our product candidates.
The biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological change. We are currently aware of various existing therapies that may compete with our product candidates. For example, XLH is currently treated with oral phosphate and vitamin D therapy, which may compete with burosumab. Furthermore, B. Braun Medical Inc., or B. Braun, has received orphan drug designation for triheptanoin in Europe for certain LC-FAOD indications and we do not know if B. Braun is planning to initiate clinical development. Triheptanoin is also available in food-grade form, which may compete with our pharmaceutical-grade product. Investigator-sponsored trials evaluating triheptanoin in multiple indications are ongoing. LC-FAOD is currently treated with diet therapy and medium-chain triglyceride oil, which may compete with UX007. Glut1 DS is currently treated primarily with the ketogenic diet and anti-epileptic drugs, which may also compete with UX007. OTC deficiency is currently treated with nitrogen scavenging drugs and severe limitations in dietary protein, which may compete with DTX301. Gene therapy, gene correction, RNA-based therapies, and other approaches may also emerge for the treatment of any of the disease areas in which we focus.
We have competitors both in the United States and internationally, including major multinational pharmaceutical companies, specialty pharmaceutical companies, biotechnology companies, startups, academic research institutions, government agencies, and public and private research institutions. Many of our competitors have substantially greater financial, technical, and other resources, such as larger research and development staff and experienced marketing and manufacturing organizations. Additional mergers and acquisitions in the biotechnology and pharmaceutical industries may result in even more resources being concentrated in our competitors. As a result, these companies may obtain regulatory approval more rapidly than we are able to and may be more effective in selling and marketing their products as well. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large, established companies. Competition may increase further as a result of advances in the commercial applicability of technologies and greater availability of capital for investment in these industries. Our competitors may succeed in developing, acquiring, or licensing on an exclusive basis, products that are more effective or less costly than any product candidate that we may develop, or achieve earlier patent protection, regulatory approval, product commercialization, and market penetration than we do. Additionally, technologies developed by our competitors may render our potential product candidates uneconomical or obsolete, and we may not be successful in marketing our product candidates against competitors.
We continue to build and evolve an integrated commercial organization. If we are unable to expand our existing commercial infrastructure or enter into agreements with third parties to market and sell our product candidates, as needed, we may be unable to generate significant revenue.
In preparation to successfully commercialize Mepsevii as well as any additional products that may result from our development programs, we have begun to build commercial infrastructure in the United States, Europe and Latin America. This infrastructure consists of both office based as well as field teams with technical expertise, and will be expanded as we approach the potential approval dates of additional products that result from our development programs. This will be expensive and time consuming. Any failure or delay in the expansion of this infrastructure may adversely impact the commercialization of our approved products.
Although our employees may promoted other similar products in the past while employed at other companies, we, as a company, have limited, recent experience selling and marketing our product. Further, given our limited experience in marketing and selling biopharmaceutical products, our initial estimate of the size of the required field force may be materially more or less than the size of the field force actually required to effectively commercialize our product candidates. As such, we may be required to hire large teams to adequately support the commercialization of our product candidates or we may incur excess costs as a result of hiring more commercial personnel than necessary. With respect to certain geographical markets, we may enter into collaborations with other entities to utilize their local marketing and distribution capabilities, but we may be unable to enter into such agreements on favorable terms, if at all. If our future collaborators do not commit sufficient resources to commercialize our future products, if any, and we are unable to develop the necessary marketing capabilities on our own, we will be unable to generate sufficient product revenue to sustain our business. We may be competing with companies that currently have extensive and well-funded marketing and sales operations. Without an internal team or the support of a third-party to perform key commercial functions, we may be unable to compete successfully against these more established companies.
44
The commercial success of any current or future product candidate will depend upon the degree of market acceptance by physicians, patients, third-party payors, and others in the medical community.
Even with the requisite approvals from the FDA and comparable foreign regulatory authorities, the commercial success of our product candidates will depend in part on the medical community, patients, and payors accepting our product candidates as medically useful, cost-effective, and safe. Any product that we bring to the market may not gain market acceptance by physicians, patients, payors, and others in the medical community. The degree of market acceptance of any of our product candidates, if approved for commercial sale, will depend on a number of factors, including:
|
|
• |
the efficacy of the product as demonstrated in clinical studies and potential advantages over competing treatments; |
|
|
• |
the prevalence and severity of any side effects, including any limitations or warnings contained in a product’s approved labeling; |
|
|
• |
the clinical indications for which approval is granted; |
|
|
• |
relative convenience and ease of administration; |
|
|
• |
the cost of treatment, particularly in relation to competing treatments; |
|
|
• |
the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
|
|
• |
the effectiveness of our field forces and marketing efforts; |
|
|
• |
the strength of marketing and distribution support and timing of market introduction of competitive products; |
|
|
• |
publicity concerning our products or competing products and treatments; and |
|
|
• |
sufficient third-party insurance coverage and reimbursement. |
Even if a potential product displays a favorable efficacy and safety profile in nonclinical and clinical studies, market acceptance of the product will not be fully known until after it is launched. Our efforts to educate the medical community and payors on the benefits of the product candidates may require significant resources and may never be successful. If our product candidates are approved but fail to achieve an adequate level of acceptance by physicians, patients, payors, and others in the medical community, we will not be able to generate sufficient revenue to become or remain profitable.
The insurance coverage and reimbursement status of newly approved products is uncertain. Failure to obtain or maintain adequate coverage and reimbursement for new or current products could limit our ability to market those products and decrease our ability to generate revenue.
Our target patient populations are small, and accordingly the pricing, coverage, and reimbursement of our product candidates, if approved, must be adequate to support our commercial infrastructure. Our per-patient prices must be sufficient to recover our development and manufacturing costs and potentially achieve profitability. We expect the cost of a single administration of gene therapy products, such as those we are developing, to be substantial, when and if they achieve regulatory approval. Accordingly, the availability and adequacy of coverage and reimbursement by governmental and private payors are essential for most patients to afford expensive treatments such as ours, assuming approval. Sales of our product candidates will depend substantially, both domestically and abroad, on the extent to which the costs of our product candidates will be paid for by health maintenance, managed care, pharmacy benefit, and similar healthcare management organizations, or reimbursed by government authorities, private health insurers, and other payors. If coverage and reimbursement are not available, are available only to limited levels, or are not available on a timely basis, we may not be able to successfully commercialize our product candidates. Even if coverage is provided, the approved reimbursement amount may not be high enough to allow us to establish or maintain pricing sufficient to sustain our overall enterprise.
There is significant uncertainty related to the insurance coverage and reimbursement of newly approved products. In the United States, the Centers for Medicare & Medicaid Services, or CMS, an agency within the U.S. Department of Health and Human Services, decides whether and to what extent a new drug will be covered and reimbursed under Medicare. Private payors tend to follow the coverage reimbursement policies established by CMS to a substantial degree. It is difficult to predict what CMS or private payors will decide with respect to reimbursement for products such as ours, especially our gene therapy product candidates as there is a limited body of established practices and precedents for gene therapy products.
Outside the United States, international operations are generally subject to extensive governmental price controls and other market regulations, and we believe the increasing emphasis on cost-containment initiatives in Europe, Canada, and other countries will put pressure on the pricing and usage of our product candidates. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems. Other countries allow companies to fix their own prices for medicinal products, but monitor and control company profits. Additional foreign price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our product candidates. Accordingly, in markets outside the United States, the reimbursement for our products may be reduced compared with the United States and may be insufficient to generate commercially reasonable revenue and profits.
45
Moreover, increasing efforts by governmental and third-party payors in the United States and abroad to cap or reduce healthcare costs may cause such organizations to limit both coverage and the level of reimbursement for new products approved and, as a result, they may not cover or provide adequate payment for our product candidates. We expect to experience pricing pressures in connection with the sale of any of our product candidates due to the trend toward managed healthcare, the increasing influence of health maintenance organizations, and additional legislative changes. For example, recently, several states in the U.S. have introduced legislation to require pharmaceutical companies to disclose their costs to justify the prices of their products, and an “Affordable Drug Pricing Task-Force” has been formed in the U.S. House of Representatives with the goal of combating the increased costs of prescription drugs. The downward pressure on healthcare costs in general, and with respect to prescription drugs, surgical procedures, and other treatments in particular, has become very intense. As a result, increasingly high barriers are being erected to the entry of new products.
Risks Related to Our Intellectual Property
If we are unable to obtain and maintain effective patent rights for our product, product candidates, or any future product candidates, we may not be able to compete effectively in our markets.
We rely upon a combination of patents, trade secret protection, and confidentiality agreements to protect the intellectual property related to our technologies, our product, and our product candidates. Our success depends in large part on our and our licensors’ ability to obtain and maintain patent and other intellectual property protection in the United States and in other countries with respect to our proprietary technology, our product, and our product candidates.
We have sought to protect our proprietary position by filing patent applications in the United States and abroad related to our novel technologies and products that are important to our business. This process is expensive and time consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain and involves complex legal and factual questions for which legal principles remain unsolved. The patent applications that we own or in-license may fail to result in issued patents with claims that cover our product or product candidates in the United States or in other foreign countries. There is no assurance that all potentially relevant prior art relating to our patents and patent applications has been found, which can invalidate a patent or prevent a patent from issuing from a pending patent application. Even if patents do successfully issue, and even if such patents cover our product or product candidates, third parties may challenge their validity, enforceability, or scope, which may result in such patents being narrowed, found unenforceable, or invalidated. For instance, in June 2017, a third party filed an opposition with the European Patent Office challenging the validity of a European patent owned by the University of Pennsylvania and sub-licensed to us from REGENXBIO relating to the AAV8 capsid used in our DTX301 product candidate. This opposition is in its very early stages and we are unable to estimate the timing or outcome of this matter. Furthermore, even if the patents and patent applications we own or in-license are unchallenged, they may not adequately protect our intellectual property, provide exclusivity for our product or product candidates, or prevent others from designing around our claims. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
We, independently or together with our licensors, have filed several patent applications covering various aspects of our product or product candidates. We cannot offer any assurances about which, if any, patents will issue, the breadth of any such patent or whether any issued patents will be found invalid and unenforceable or will be threatened by third parties. Any successful opposition to these patents or any other patents owned by or licensed to us after patent issuance could deprive us of rights necessary for the successful commercialization of any product candidates that we may develop. Further, if we encounter delays in regulatory approvals, the period of time during which we could market a product candidate under patent protection could be reduced.
Although we have a number of patents or applications covering methods of use and certain compositions of matter, we do not have complete patent protection for our product and product candidates in all territories. For example, there are no issued patents covering the burosumab composition of matter in Latin America where we have rights to commercialize the compound. Therefore, a competitor could develop the same antibody or a similar antibody as well as other approaches that target FGF23 for potential commercialization in Latin America, subject to any intellectual property rights or regulatory exclusivities awarded to us. If we cannot obtain and maintain effective patent rights for our product or product candidates, we may not be able to compete effectively and our business and results of operations would be harmed.
We may not have sufficient patent terms to effectively protect our products and business.
Patents have a limited lifespan. In the United States, the natural expiration of a patent is generally 20 years after it is filed. Although various extensions may be available, the life of a patent, and the protection it affords, is limited. Even if patents covering our product candidates are obtained, once the patent life has expired for a product, we may be open to competition from generic medications.
46
While patent term extensions under the Hatch-Waxman Act in the United States and under supplementary protection certificates in Europe may be available to extend the patent exclusivity term for burosumab, vestronidase alfa, UX007, and DTX301, we cannot provide any assurances that any such patent term extension will be obtained and, if so, for how long. In addition, upon issuance in the United States, any patent term can be adjusted based on certain delays caused by the applicant(s) or the United States Patent and Trademark Office (USPTO). For example, a patent term can be reduced based on certain delays caused by the patent applicant during patent prosecution. If we do not have sufficient patent terms or regulatory exclusivity to protect our products, our business and results of operations will be adversely affected.
Patent policy and rule changes could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents.
Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents or narrow the scope of our patent protection. The laws of foreign countries may not protect our rights to the same extent as the laws of the United States. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. We therefore cannot be certain that we or our licensors were the first to make the invention claimed in our owned and licensed patents or pending applications, or that we or our licensor were the first to file for patent protection of such inventions. Assuming the other requirements for patentability are met, in the United States prior to March 15, 2013, the first to make the claimed invention is entitled to the patent, while outside the United States, the first to file a patent application is entitled to the patent. After March 15, 2013, under the Leahy-Smith America Invents Act, or the Leahy-Smith Act, enacted on September 16, 2011, the United States has moved to a first to file system. The Leahy-Smith Act also includes a number of significant changes that affect the way patent applications will be prosecuted and may also affect patent litigation. The effects of these changes are currently unclear as the courts have only begun to address these provisions. In general, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition.
The U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of protection available in certain circumstances or weakening the rights of patent owners in certain situations. For example, in Association for Molecular Pathology v. Myriad Genetics, Inc., the Supreme Court ruled that a “naturally occurring DNA segment is a product of nature and not patent eligible merely because it has been isolated,” invalidating Myriad Genetics’ patents on the BRCA1 and BRCA2 genes. Certain claims of our licensed U.S. patents covering DTX301 relate to isolated AAV8 vectors, capsid proteins, or nucleic acids. To the extent that such claims are deemed to be directed to natural products, or to lack an inventive concept above and beyond an isolated natural product, a court may decide the claims are invalid under Myriad. Additionally, there have been recent proposals for additional changes to the patent laws of the United States and other countries that, if adopted, could impact our ability to obtain patent protection for our proprietary technology or our ability to enforce our proprietary technology. Depending on future actions by the U.S. Congress, the U.S. courts, the USPTO and the relevant law-making bodies in other countries, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
If we are unable to maintain effective proprietary rights for our product, product candidates, or any future product candidates, we may not be able to compete effectively in our markets.
In addition to the protection afforded by patents, we rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes for which patents are difficult to enforce and any other elements of our product or product candidate discovery and development processes that involve proprietary know-how, information, or technology that is not covered by patents. However, trade secrets can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements with our employees, consultants, scientific advisors, and contractors. We also seek to preserve the integrity and confidentiality of our data and trade secrets by maintaining physical security of our premises and physical and electronic security of our information technology systems. While we have confidence in these individuals, organizations and systems, agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors.
Although we expect all of our employees and consultants to assign their inventions to us, and all of our employees, consultants, advisors, and any third parties who have access to our proprietary know-how, information, or technology to enter into confidentiality agreements, we cannot provide any assurances that all such agreements have been duly executed or that our trade secrets and other confidential proprietary information will not be disclosed or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. Misappropriation or unauthorized disclosure of our trade secrets could impair our competitive position and may have a material adverse effect on our business. Additionally, if the steps taken to maintain our trade secrets are deemed inadequate, we may have insufficient recourse against third parties for misappropriating the trade secret.
47
Claims of intellectual property infringement may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on our avoiding infringement of the patents and proprietary rights of others. There have been many lawsuits and other proceedings involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interferences, oppositions, and reexamination proceedings before the USPTO and corresponding foreign patent offices. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by other parties, exist in the fields in which we are developing product candidates. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our product or product candidates may be subject to claims of infringement of the patent rights of these other parties.
Other parties may assert that we are employing their proprietary technology without authorization. There may be patents or patent applications with claims to materials, formulations, methods of manufacture, or methods for treatment related to the use or manufacture of our product or product candidates. We have conducted freedom to operate analyses with respect only to our product and certain of our product candidates, and therefore we do not know whether there are any patents of other parties that would impair our ability to commercialize all of our product candidates. We also cannot guarantee that any of our analyses are complete and thorough, nor can we be sure that we have identified each and every patent and pending application in the United States and abroad that is relevant or necessary to the commercialization of our product or product candidates. Because patent applications can take many years to issue, there may be currently pending patent applications that may later result in issued patents that our product or product candidates may infringe.
We are aware of three third-party patent families that include issued U.S. patents with claims that, if valid and enforceable, could be construed to cover DTX301, if and when approved, or methods of its manufacture. We are also aware of an additional three third-party patent families that include issued European claims that, if valid and enforceable, could be construed to cover certain methods that may be used in the manufacture of DTX301. In addition, other parties may obtain patents in the future and claim that use of our technologies infringes upon these patents. If any of these patents were held by a court of competent jurisdiction to cover aspects of our formulations, the manufacturing process of our product or any of our product candidates, methods of use, any molecules formed during the manufacturing process or any final product itself, the holders of any such patents may be able to block our ability to commercialize our product or a product candidate unless we obtained a license under the applicable patents, or until such patents expire or are finally determined to be invalid or unenforceable. Such a license may not be available on commercially reasonable terms or at all.
Parties making claims against us may obtain injunctive or other equitable relief, which could effectively block our ability to continue commercialization of our product, or block our ability to develop and commercialize one or more of our product candidates. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of employee resources from our business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys’ fees for willful infringement, pay royalties, redesign our infringing products, or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure.
We may not be successful in obtaining or maintaining necessary rights to our product candidates through acquisitions and in-licenses.
We currently have rights to the intellectual property, through licenses from third parties and under patents that we own, to develop our product candidates. Because our programs may require the use of proprietary rights held by third parties, the growth of our business will likely depend in part on our ability to acquire, in-license, or use these proprietary rights. For example, our product candidates may require specific formulations to work effectively and efficiently and the rights to these formulations may be held by others. We may be unable to acquire or in-license any compositions, methods of use, processes, or other third-party intellectual property rights from third parties that we identify as necessary for our product candidates. The licensing and acquisition of third-party intellectual property rights is a competitive area, and a number of more established companies are also pursuing strategies to license or acquire third-party intellectual property rights that we may consider attractive. These established companies may have a competitive advantage over us due to their size, cash resources, and greater clinical development and commercialization capabilities. In addition, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. We also may be unable to license or acquire third-party intellectual property rights on terms that would allow us to make an appropriate return on our investment.
We sometimes collaborate with U.S. and foreign academic institutions to accelerate our preclinical research or development under written agreements with these institutions. Typically, these institutions provide us an option to negotiate a license to any of the institution’s rights in technology resulting from the collaboration. Regardless of such option, we may be unable to negotiate a license within the specified timeframe or under terms that are acceptable to us. If we are unable to do so, the institution may offer the intellectual property rights to other parties, potentially blocking our ability to pursue our program.
If we are unable to successfully obtain rights to required third-party intellectual property rights or maintain the existing intellectual property rights we have, we may have to abandon development of that program and our business and financial condition could suffer.
48
We may face competition from biosimilars, which may have a material adverse impact on the future commercial prospects of burosumab, vestronidase alfa, and DTX301.
Even if we are successful in achieving regulatory approval to commercialize a product candidate faster than our competitors, we may face competition from biosimilars with respect to burosumab, vestronidase alfa, and DTX301. In the United States, the Biologics Price Competition and Innovation Act of 2009, or BPCI Act, was included in the Affordable Care Act and created an abbreviated approval pathway for biological products that are demonstrated to be “highly similar,” or biosimilar, to or “interchangeable” with an FDA-approved biological product. The BPCI Act prohibits the FDA from approving a biosimilar or interchangeable product that references a brand biological product until 12 years after the licensure of the reference product, but permits submission of an application for a biosimilar or interchangeable product to the FDA four years after the reference product was first licensed. The BPCI Act does not prevent another company from developing a product that is highly similar to the innovative product, generating its own data, and seeking approval. The BPCI Act is complex and is only beginning to be interpreted and implemented by the FDA. Moreover, it is not known whether the BPCI Act will survive in whole or in part if the Affordable Care Act is repealed. As a result, its ultimate impact, implementation, meaning, and long-term existence are subject to uncertainty. Elimination or modification of the BPCI Act, or changes to the FDA’s interpretation or implementation of the BPCI Act, could have a material adverse effect on the future commercial prospects for burosumab, vestronidase alfa, and DTX301.
In Europe, the European Commission has granted marketing authorizations for several biosimilars pursuant to a set of general and product class-specific guidelines for biosimilar approvals issued over the past few years. In Europe, a competitor may reference data supporting approval of an innovative biological product, but will not be able to get on the market until 10 years after the time of approval of the innovative product. This 10-year marketing exclusivity period will be extended to 11 years if, during the first eight of those 10 years, the marketing authorization holder obtains an approval for one or more new therapeutic indications that bring significant clinical benefits compared with existing therapies. In addition, companies may be developing biosimilars in other countries that could compete with our products.
If competitors are able to obtain marketing approval for biosimilars referencing our products, our products may become subject to competition from such biosimilars, with the attendant competitive pressure and consequences.
Additional competitors could enter the market with generic versions of our small-molecule product candidates, which may result in a material decline in sales of UX007 or future small-molecule product candidates.
Under the Hatch-Waxman Act, a pharmaceutical manufacturer may file an abbreviated new drug application, or ANDA, seeking approval of a generic copy of an approved innovator product. Under the Hatch-Waxman Act, a manufacturer may also submit an NDA under section 505(b)(2) that references the FDA’s finding of safety and effectiveness of a previously approved drug. A 505(b)(2) NDA product may be for a new or improved version of the original innovator product. Innovative small molecule drugs may be eligible for certain periods of regulatory exclusivity (e.g., five years for new chemical entities, three years for changes to an approved drug requiring a new clinical study, and seven years for orphan drugs), which preclude FDA approval (or in some circumstances, FDA filing and review of) an ANDA or 505(b)(2) NDA relying on the FDA’s finding of safety and effectiveness for the innovative drug. In addition to the benefits of regulatory exclusivity, an innovator NDA holder may have patents claiming the active ingredient, product formulation or an approved use of the drug, which would be listed with the product in the “Orange Book.” If there are patents listed in the Orange Book, a generic applicant that seeks to market its product before expiration of the patents must include in the ANDA or 505(b)(2) what is known as a “Paragraph IV certification,” challenging the validity or enforceability of, or claiming non-infringement of, the listed patent or patents. Notice of the certification must be given to the innovator, too, and if within 45 days of receiving notice the innovator sues to protect its patents, approval of the ANDA is stayed for 30 months, or as lengthened or shortened by the court.
Accordingly, if UX007 is approved, competitors could file ANDAs for generic versions of UX007, or 505(b)(2) NDAs that reference UX007. If there are patents listed for UX007 in the Orange Book, those ANDAs and 505(b)(2) NDAs would be required to include a certification as to each listed patent indicating whether the ANDA applicant does or does not intend to challenge the patent. We cannot predict whether any patents issuing from our pending patent applications will be eligible for listing in the Orange Book, how any generic competitor would address such patents, whether we would sue on any such patents, or the outcome of any such suit.
We may not be successful in securing or maintaining proprietary patent protection for products and technologies we develop or license. Moreover, if any patents that are granted and listed in the Orange Book are successfully challenged by way of a Paragraph IV certification and subsequent litigation, the affected product could more immediately face generic competition and its sales would likely decline materially. Should sales decline, we may have to write off a portion or all of the intangible assets associated with the affected product and our results of operations and cash flows could be materially and adversely affected.
The patent protection and patent prosecution for some of our product candidates is dependent on third parties.
While we normally seek and gain the right to fully prosecute the patents relating to our product candidates, there may be times when patents relating to our product candidates are controlled by our licensors. This is the case with our agreement with KHK, who is primarily responsible for the prosecution of patents and patent applications licensed to us under the collaboration agreement.
49
In addition, we have in-licensed patents and patent applications owned by the University of Pennsylvania, relating to the AAV8 vector used in DTX301. These patents and patent applications are licensed or sublicensed by REGENXBIO and sublicensed to us. We do not have the right to control the prosecution of these patent applications, or the maintenance of any of these patents. In addition, under our agreement with REGENXBIO, we do not have the first right to enforce the licensed patents, and our enforcement rights are subject to certain limitations that may adversely impact our ability to use the licensed patents to exclude others from commercializing competitive products. Moreover, REGENXBIO and the University of Pennsylvania may have interests which differ from ours in determining whether and the manner in which to enforce such patents.
If KHK, the University of Pennsylvania, or any of our future licensing partners fail to appropriately prosecute, maintain, and enforce patent protection for the patents covering any of our product candidates, our ability to develop and commercialize those product candidates may be adversely affected and we may not be able to prevent competitors from making, using, and selling competing products. In addition, even where we now have the right to control patent prosecution of patents and patent applications we have licensed from third parties, we may still be adversely affected or prejudiced by actions or inactions of our licensors and their counsel that took place prior to us assuming control over patent prosecution.
If we fail to comply with our obligations in the agreements under which we license intellectual property and other rights from third parties or otherwise experience disruptions to our business relationships with our licensors, we could lose license rights that are important to our business.
We are a party to a number of intellectual property license agreements that are important to our business and expect to enter into additional license agreements in the future. Our existing license agreements impose, and we expect that future license agreements will impose, various diligence, milestone payment, royalty, and other obligations on us. If we fail to comply with our obligations under these agreements, or we are subject to a bankruptcy, we may be required to make certain payments to the licensor, we may lose the exclusivity of our license, or the licensor may have the right to terminate the license, in which event we would not be able to develop or market products covered by the license. Additionally, the milestone and other payments associated with these licenses will make it less profitable for us to develop our drug candidates. See “Business—License and Collaboration Agreements” in this Annual Report for a description of our license agreements with KHK, Baylor Research Institute, Saint Louis University, Bayer, REGENXBIO, and the University of Pennsylvania, which include descriptions of the termination provisions of these agreements.
In certain cases, we control the prosecution of patents resulting from licensed technology. In the event we breach any of our obligations related to such prosecution, we may incur significant liability to our licensing partners. Licensing of intellectual property is of critical importance to our business and involves complex legal, business, and scientific issues. Disputes may arise regarding intellectual property subject to a licensing agreement, including but not limited to:
|
|
• |
the scope of rights granted under the license agreement and other interpretation-related issues; |
|
|
• |
the extent to which our technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement; |
|
|
• |
the sublicensing of patent and other rights; |
|
|
• |
our diligence obligations under the license agreement and what activities satisfy those diligence obligations; |
|
|
• |
the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our collaborators; and |
|
|
• |
the priority of invention of patented technology. |
If disputes over intellectual property and other rights that we have licensed prevent or impair our ability to maintain our current licensing arrangements on acceptable terms, we may be unable to successfully develop and commercialize the affected product candidates.
Although we are not currently involved in any intellectual property litigation, we may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time consuming, and unsuccessful.
Competitors may infringe our patents or the patents of our licensors. Although we are not currently involved in any intellectual property litigation, if we or one of our licensing partners were to initiate legal proceedings against a third party to enforce a patent covering our product or one of our product candidates, the defendant could counterclaim that the patent covering our product candidate is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. The outcome following legal assertions of invalidity and unenforceability is unpredictable.
50
Interference proceedings or derivation proceedings now available under the Leahy-Smith Act provoked by third parties or brought by us or declared or instituted by the USPTO may be necessary to determine the priority of inventions with respect to our patents or patent applications or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms. In addition, the validity of our patents could be challenged in the USPTO by one of the new post grant proceedings (i.e., inter partes review or post grant review) now available under the Leahy-Smith Act. Our defense of litigation, interference proceedings, or post grant proceedings under the Leahy-Smith Act may fail and, even if successful, may result in substantial costs and distract our management and other employees. In addition, the uncertainties associated with litigation could have a material adverse effect on our ability to raise sufficient capital to continue our clinical studies, continue our research programs, license necessary technology from third parties, or enter into development partnerships that would help us bring our product candidates to market.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions, or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our common stock.
We may be subject to claims that our employees, consultants, or independent contractors have wrongfully used or disclosed confidential information of third parties or that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
We employ certain individuals who were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees, consultants, and independent contractors do not use the proprietary information or know-how of others in their work for us, and we are not currently subject to any claims that our employees, consultants, or independent contractors have wrongfully used or disclosed confidential information of third parties, we may in the future be subject to such claims. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel, which could adversely impact our business. Even if we are successful in defending against such claims, litigation could result in substantial costs and distract management and other employees.
We may be subject to claims challenging the inventorship of our patents and other intellectual property.
Although we are not currently experiencing any claims challenging the inventorship of our patents or ownership of our intellectual property, we may in the future be subject to claims that former employees, collaborators, or other third parties have an interest in our patents or other intellectual property as an inventor or co-inventor. For example, we may have inventorship disputes arise from conflicting obligations of consultants or others who are involved in developing our product candidates. Litigation may be necessary to defend against these and other claims challenging inventorship. If we fail to successfully defend against such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, valuable intellectual property. Such an outcome could have a material adverse effect on our business. Even if we are successful in defending against such claims, litigation could result in substantial costs and distract management and other employees.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing our ability to protect our products.
As is the case with other biotechnology and pharmaceutical companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biotechnology and pharmaceutical industries involves both technological and legal complexity. Therefore, obtaining and enforcing such patents is costly, time consuming, and inherently uncertain. In addition, the United States has recently enacted and is currently implementing wide-ranging patent reform legislation. Recent U.S. Supreme Court rulings have narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in certain situations. In addition to increasing uncertainty about our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on future actions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents or to enforce our existing patents and patents that we might obtain in the future.
51
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting, and defending patents on our product or product candidates in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. Further, licensing partners such as KHK may not prosecute patents in certain jurisdictions in which we may obtain commercial rights, thereby precluding the possibility of later obtaining patent protection in these countries. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and may also export infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States. These products may compete with our products, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets, and other intellectual property protection, particularly those relating to biotechnology products, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly, could put our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
Risks Related to Our Business Operations
Our future success depends in part on our ability to retain our Founder, President, and Chief Executive Officer and to attract, retain, and motivate other qualified personnel.
We are dependent on Emil D. Kakkis, M.D., Ph.D., our Founder, President, and Chief Executive Officer, the loss of whose services may adversely impact the achievement of our objectives. Dr. Kakkis could leave our employment at any time, as he is an “at will” employee. Recruiting and retaining other qualified employees, consultants, and advisors for our business, including scientific and technical personnel, will also be critical to our success. There is currently a shortage of skilled personnel in our industry, which is likely to continue. As a result, competition for skilled personnel is intense and the turnover rate can be high. We may not be able to attract and retain personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for individuals with similar skill sets. In addition, failure to succeed in preclinical or clinical studies may make it more challenging to recruit and retain qualified personnel. The inability to recruit and retain qualified personnel, or the loss of the services of Dr. Kakkis, may impede the progress of our research, development, and commercialization objectives.
If we fail to obtain or maintain orphan drug exclusivity for our products, our competitors may sell products to treat the same conditions and our revenue will be reduced.
Our business strategy focuses on the development of drugs that are eligible for FDA and EU orphan drug designation. In the United States, orphan drug designation entitles a party to financial incentives such as opportunities for grant funding towards clinical study costs, tax advantages, and user-fee waivers. In addition, if a product receives the first FDA approval for the indication for which it has orphan designation, the product is entitled to orphan drug exclusivity, which means the FDA may not approve any other application to market the same drug for the same indication for a period of seven years, except in limited circumstances, such as a showing of clinical superiority over the product with orphan exclusivity or where the manufacturer is unable to assure sufficient product quantity. In the EU, orphan drug designation entitles a party to financial incentives such as reduction of fees or fee waivers and ten years of market exclusivity following drug or biological product approval. This period may be reduced to six years if the orphan drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable not to justify maintenance of market exclusivity.
Because the extent and scope of patent protection for our products may in some cases be limited, orphan drug designation is especially important for our products for which orphan drug designation may be available. For eligible drugs, we plan to rely on the exclusivity period under the Orphan Drug Act to maintain a competitive position. If we do not obtain orphan drug exclusivity for our drug products and biologic products that do not have broad patent protection, our competitors may then sell the same drug to treat the same condition sooner than if we had obtained orphan drug exclusivity, and our revenue will be reduced.
52
Even though we have orphan drug designation for UX007 for the treatment of fatty acid oxidation disorders in the United States and for various subtypes of FAOD in Europe, as well as for UX007 for the treatment of Glut1 DS, burosumab, Mepsevii, and DTX301 in the United States and Europe, we may not be the first to obtain marketing approval for any particular orphan indication due to the uncertainties associated with developing pharmaceutical products. Further, even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs with different active moieties can be approved for the same condition or the same drug can be approved for a different indication unless there are other exclusivities such as new chemical entity exclusivity preventing such approval. Even after an orphan drug is approved, the FDA or EMA can subsequently approve the same drug with the same active moiety for the same condition if the FDA or EMA concludes that the later drug is safer, more effective, or makes a major contribution to patient care. Orphan drug designation neither shortens the development time or regulatory review time of a drug nor gives the drug any advantage in the regulatory review or approval process.
We will need to expand our organization and we may experience difficulties in managing this growth, which could disrupt our operations.
As our development and commercialization plans and strategies develop, we expect to need additional managerial, operational, field forces, marketing, financial, legal, and other resources. Our management may need to divert a disproportionate amount of its attention away from our day-to-day activities and devote a substantial amount of time to managing these growth activities. We may not be able to effectively manage the expansion of our operations, which may result in weaknesses in our infrastructure, operational mistakes, loss of business opportunities, loss of employees, and reduced productivity among remaining employees. Our expected growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of additional product candidates. If our management is unable to effectively manage our growth, our expenses may increase more than expected, our ability to generate and/or grow revenue could be reduced, and we may not be able to implement our business strategy. Our future financial performance and our ability to commercialize product candidates and compete effectively will depend, in part, on our ability to effectively manage any future growth.
Our operating results would be adversely impacted if our intangible assets become impaired.
As a result of the accounting for our acquisition of Dimension Therapeutics, Inc. (Dimension) in November 2017, we have recorded on our balance sheet intangible assets for in-process research and development (“IPR&D”) of $129.0 million and an acquired contract asset of $12.5 million as of December 31, 2017. We test the intangible assets for impairment annually during the fourth quarter and more frequently if events or changes in circumstances indicate that it is more likely than not that the asset is impaired. If the associated research and development effort is abandoned, the related assets will be written-off and we will record a noncash impairment loss on our statement of operations. We have not recorded any impairments since inception.
We may not be successful in our efforts to identify, license, discover, develop, or commercialize additional product candidates.
Although a substantial amount of our effort will focus on the continued clinical testing, potential approval, and commercialization of our existing product candidates, the success of our business also depends upon our ability to identify, license, discover, develop, or commercialize additional product candidates. Research programs to identify and develop new product candidates, such as those under our collaboration with Arcturus, require substantial technical, financial, and human resources. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful. Our research programs or licensing efforts may fail to yield additional product candidates for clinical development and commercialization for a number of reasons, including but not limited to the following:
|
|
• |
our research or business development methodology or search criteria and process may be unsuccessful in identifying potential product candidates; |
|
|
• |
we may not be able or willing to assemble sufficient technical, financial or human resources to acquire or discover additional product candidates; |
|
|
• |
we may face competition in obtaining and/or developing additional product candidates; |
|
|
• |
our product candidates may not succeed in research, discovery, preclinical or clinical testing; |
|
|
• |
our potential product candidates may be shown to have harmful side effects or may have other characteristics that may make the products unmarketable or unlikely to receive marketing approval; |
|
|
• |
competitors may develop alternatives that render our product candidates obsolete or less attractive; |
|
|
• |
product candidates we develop may be covered by third parties’ patents or other exclusive rights; |
|
|
• |
the market for a product candidate may change during our program so that such a product may become unreasonable to continue to develop; |
53
|
|
• |
a product candidate may not be capable of being produced in commercial quantities at an acceptable cost or at all; and |
|
|
• |
a product candidate may not be accepted as safe and effective by regulatory authorities, patients, the medical community, or payors. |
If any of these events occur, we may be forced to abandon our development efforts for a program or programs, or we may not be able to identify, license, discover, develop, or commercialize additional product candidates, which would have a material adverse effect on our business and could potentially cause us to cease operations.
If we are unable to maintain and further develop effective internal control over financial reporting, investors may lose confidence in the accuracy and completeness of our financial reports and the market price of our stock may decrease.
The Sarbanes-Oxley Act requires, among other things, that we maintain effective internal controls for financial reporting and disclosure controls and procedures. In particular, we are required to perform system and process evaluation and testing of our internal controls over financial reporting to allow management to report on the effectiveness of our internal controls over financial reporting, as required by Section 404(a) of the Sarbanes-Oxley Act. We are also subject to the compliance requirements of Section 404(b) of the Sarbanes-Oxley Act, which results in us incurring substantial expenses and expending significant management efforts. We currently do not have an internal audit group. We may need to hire additional accounting and financial staff with appropriate public company experience and technical accounting knowledge. If we are not able to comply with the requirements of Section 404(b) or if we or our independent registered public accounting firm identify deficiencies in our internal controls over financial reporting that are deemed to be material weaknesses, investors may lose confidence in the accuracy and completeness of our financial reports, the market price of our stock could decline and we could be subject to sanctions or investigations by Nasdaq, the SEC, or other regulatory authorities, which would require additional financial and management resources.
Changes to healthcare and FDA laws, regulations, and policies may have a material adverse effect on our business and results of operations.
United States
In the United States, there have been and continue to be a number of legislative initiatives to contain healthcare costs and to modify the regulation of drug and biologic products. For example, the Affordable Care Act, as amended, substantially changed the way health care is financed by both governmental and private insurers, and significantly impacts the U.S. pharmaceutical industry. The Affordable Care Act, among other things, subjects biologic products to potential competition by lower-cost biosimilars, addresses a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted, or injected, increases the minimum Medicaid rebates owed by manufacturers under the Medicaid Drug Rebate Program and extends the rebate program to individuals enrolled in Medicaid managed care organizations, and establishes annual fees and taxes on manufacturers of certain branded prescription drugs. Implementation of the Affordable Care Act remains ongoing, and there remains uncertainty as to how the law’s various provisions will ultimately affect the industry and whether the law will remain in place.
Other legislative changes have been adopted in the United States, including the Cures Act and the Budget Control Act of 2011, or the Budget Act, signed into law on August 2, 2011. The Cures Act introduces a wide range of reforms and the Budget Act, among other things, required reductions in federal spending, which eventually triggered Medicare sequestration—the requirement to reduce Medicare payments to providers up to 2% per fiscal year. In 2013, the 2% Medicare payment reductions were applied to fee-for-service claims with dates of service or dates of discharge on or after April 1, 2013. Sequestration was initially set to expire in fiscal year 2021 but has been extended to 2025.
We expect that additional state and federal healthcare reform measures and regulations will be adopted in the future, including proposals to reduce the exclusivity protections provided to already approved biological products and to provide biosimilar and interchangeable biologic products an easier path to approval. Any of these measures and regulations could limit the amounts that federal and state governments will pay for healthcare products and services, result in reduced demand for our product candidates or additional pricing pressures and affect our product development, testing, marketing approvals and post-market activities.
European Union
In the EU, the European Commission has adopted detailed rules for the safety features appearing on the packaging of medicinal products for human use. The regulations set forth the rules for the features appearing on the packaging of these medicinal products, including, inter alia, the characteristics and technical specifications of the unique identifier that enables the authenticity of medicinal products to be verified and individual packs to be identified, the modalities for the verification of the safety features, and the list of medicinal products and product categories subject and not subject to prescription which shall not bear and bear (respectively) safety features.
54
The European Commission has also launched a series of public consultations that are aimed at the adoption of notices and guidelines which will serve the interpretation of currently applicable regulations and directives. For example, between August 2015 and December 2016, the European Commission launched public consultations which concerned good manufacturing practices, clinical trials for human medicinal products, and orphan medicinal products. The purpose of the consultation on orphan medicinal products (which will be replaced with a Notice) is to streamline the regulatory framework and to adapt the applicable regulations to technical progress. The consultation focuses on a variety of elements of Regulation (EC) No 141/2000, which include the encouragement of development of orphan medicinal products for communicable diseases and the simplification of the procedure for the reassessment of orphan criteria when two authorization application procedures are pending in parallel for two orphan medicinal products.
We may be subject, directly or indirectly, to federal and state healthcare fraud and abuse laws, false claims laws, and health information privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties.
If we obtain FDA approval for any of our product candidates and begin commercializing those products in the United States, our operations may be directly, or indirectly through our customers, subject to various federal and state fraud and abuse laws, including, without limitation, the federal Anti-Kickback Statute, the federal False Claims Act, and physician sunshine laws and regulations. These laws may impact, among other things, our proposed field marketing and education programs. In addition, we may be subject to patient privacy regulation by both the federal government and the states in which we conduct our business. The laws that may affect our ability to operate are described under “Business—Government Regulation” in this Annual Report. Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. If our operations are found to be in violation of any of the laws described above or any other governmental regulations that apply to us, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from participation in government health care programs, such as Medicare and Medicaid, imprisonment, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations.
International expansion of our business exposes us to business, regulatory, political, operational, financial, and economic risks associated with doing business outside of the United States.
We currently have limited international operations, but our business strategy incorporates significant international expansion, particularly in anticipation of approval of our product candidates. We currently conduct physician and patient association outreach activities, as well as clinical studies, outside of the United States and plan to maintain field forces representatives internationally in the future. Doing business internationally involves a number of risks, including but not limited to:
|
|
• |
multiple, conflicting, and changing laws and regulations such as privacy and data regulations, transparency regulations, tax laws, export and import restrictions, employment laws, regulatory requirements, and other governmental approvals, permits, and licenses; |
|
|
• |
introduction of new health authority requirements and/or changes in health authority expectations; |
|
|
• |
failure by us to obtain and maintain regulatory approvals for the use of our products in various countries; |
|
|
• |
additional potentially relevant third-party patent rights; |
|
|
• |
complexities and difficulties in obtaining protection for, and enforcing, our intellectual property; |
|
|
• |
difficulties in staffing and managing foreign operations; |
|
|
• |
complexities associated with managing multiple payor reimbursement regimes, government payors, or patient self-pay systems; |
|
|
• |
limits in our ability to penetrate international markets; |
|
|
• |
financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for our products, and exposure to foreign currency exchange rate fluctuations; |
|
|
• |
natural disasters and political and economic instability, including wars, terrorism, political unrest, outbreak of disease, boycotts, curtailment of trade, and other business restrictions; |
|
|
• |
certain expenses including, among others, expenses for travel, translation, and insurance; |
|
|
• |
regulatory and compliance risks that relate to maintaining accurate information and control over commercial operations and activities that may fall within the purview of the U.S. Foreign Corrupt Practices Act, or FCPA, its books and records provisions, or its anti-bribery provisions, including those under the U.K. Bribery Act and similar foreign laws and regulations; and |
|
|
• |
regulatory and compliance risks relating to doing business with any entity that is subject to sanctions administered by the Office of Foreign Assets Control of the U.S. Department of the Treasury. |
55
Any of these factors could significantly harm our future international expansion and operations and, consequently, our results of operations.
We may incur additional tax liabilities related to our operations.
We have a multinational tax structure and are subject to income tax in the United States and various foreign jurisdictions. Our effective tax rate is influenced by many factors including changes in our operating structure, changes in the mix of our earnings among countries, our allocation of profits and losses among our subsidiaries, our intercompany transfer pricing agreements and rules relating to transfer pricing, the availability of U.S. research and development tax credits, and future changes in tax laws and regulations in the U.S. and foreign countries. Significant judgment is required in determining our tax liabilities including management’s judgment for uncertain tax positions. The Internal Revenue Service, other domestic taxing authorities, or foreign taxing authorities may disagree with our interpretation of tax laws as applied to our operations. Our reported effective tax rate and after-tax cash flows may be materially and adversely affected by tax assessments in excess of amounts accrued for our financial statements. This could materially increase our future effective tax rate thereby reducing net income and adversely impacting our results of operations for future periods.
Failure to comply with laws and regulations could harm our business and our reputation.
Our business is subject to regulation by various federal, state, local and foreign governmental agencies, including agencies responsible for monitoring and enforcing employment and labor laws, workplace safety, and tax laws and regulations. In certain jurisdictions, these regulatory requirements may be more stringent than those in the United States, and in other circumstances these requirements may be more stringent in the United States. Noncompliance with applicable regulations or requirements could subject us to investigations, sanctions, mandatory recalls, enforcement actions, disgorgement of profits, fines, damages, civil and criminal penalties, or injunctions. If any governmental sanctions, fines or penalties are imposed, or if we do not prevail in any possible civil or criminal litigation, our business, operating results, financial condition and our reputation could be harmed. In addition, responding to any action will likely result in a significant diversion of management’s attention and resources and an increase in professional fees. Enforcement actions and sanctions could further harm our business, operating results, financial condition, and our reputation.
In particular, our research and development activities and our and our third-party manufacturers’ and suppliers’ activities involve the controlled storage, use, and disposal of hazardous materials, including the components of our product candidates, such as viruses, and other hazardous compounds, which subjects us to laws and regulations governing such activities. In some cases, these hazardous materials and various wastes resulting from their use are stored at our or our manufacturers’ facilities pending their use and disposal. We cannot eliminate the risk of contamination, which could cause an interruption of our commercialization efforts, research and development efforts, and business operations or environmental damage that could result in costly clean-up and liabilities under applicable laws and regulations governing the use, storage, handling, and disposal of these materials and specified waste products. We cannot guarantee that the safety procedures utilized by us and our third-party manufacturers for handling and disposing of these materials comply with the standards prescribed by these laws and regulations, or eliminate the risk of accidental contamination or injury from these materials. In such an event, we may be held liable for any resulting damages—and such liability could exceed our resources—and state or federal or other applicable authorities may curtail our use of certain materials and/or interrupt our business operations. Furthermore, environmental laws and regulations are complex, change frequently, and have tended to become more stringent. We cannot predict the impact of such changes and cannot be certain of our future compliance. We do not currently carry biological or hazardous waste insurance coverage.
Risks generally associated with a company-wide implementation of an enterprise resource planning (ERP) system may adversely affect our business and results of operations or the effectiveness of our internal controls over financial reporting.
We are in the process of implementing a company-wide ERP system to upgrade certain existing business, operational, and financial processes. Our ERP implementation is a complex and time-consuming project that we expect will require multiple years to complete. Our results of operations could be adversely affected if we experience time delays or cost overruns during the ERP implementation process, or if the ERP system or associated process changes do not give rise to the benefits that we expect. This project has required and may continue to require investment of capital and human resources, the re-engineering of processes of our business, and the attention of many employees who would otherwise be focused on other aspects of our business. Any deficiencies in the design and implementation of the new ERP system could result in potentially much higher costs than we had incurred and could adversely affect our ability to develop and launch solutions, provide services, fulfill contractual obligations, file reports with the SEC in a timely manner, operate our business or otherwise affect our controls environment. Any of these consequences could have an adverse effect on our results of operations and financial condition.
56
Our business and operations may be materially adversely affected in the event of computer system failures or security breaches.
Despite the implementation of security measures, our internal computer systems, and those of our CROs and other third parties on which we rely, are vulnerable to damage from computer viruses, unauthorized access, cyber-attacks, natural disasters, fire, terrorism, war, and telecommunication and electrical failures. If such an event were to occur and interrupt our operations, it could result in a material disruption of our drug development programs. For example, the loss of clinical trial data from ongoing or planned clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss of or damage to our data or applications, loss of trade secrets or inappropriate disclosure of confidential or proprietary information, including protected health information or personal data of employees or former employees, access to our clinical data, or disruption of the manufacturing process, we could incur liability and the further development of our drug candidates could be delayed. We may also be vulnerable to cyber-attacks by hackers or other malfeasance. This type of breach of our cybersecurity may compromise our confidential information and/or our financial information and adversely affect our business or result in legal proceedings.
We or the third parties upon whom we depend may be adversely affected by earthquakes or other natural disasters and our business continuity and disaster recovery plans may not adequately protect us from a serious disaster.
Our corporate headquarters and one of our laboratories are located in the San Francisco Bay Area, and our collaboration partner for burosumab, KHK, is located in Japan, which have both in the past experienced severe earthquakes and other natural disasters. We do not carry earthquake insurance. Earthquakes or other natural disasters could severely disrupt our operations or those of our collaborators, and have a material adverse effect on our business, results of operations, financial condition, and prospects. If a natural disaster, power outage, or other event occurred that prevented us from using all or a significant portion of our headquarters, that damaged critical infrastructure (such as the manufacturing facilities of our third-party contract manufacturers) or that otherwise disrupted operations, it may be difficult or, in certain cases, impossible for us to continue our business for a substantial period of time. The disaster recovery and business continuity plans we have in place currently are limited and are unlikely to prove adequate in the event of a serious disaster or similar event. We may incur substantial expenses as a result of the limited nature of our disaster recovery and business continuity plans, which, particularly when taken together with our lack of earthquake insurance, could have a material adverse effect on our business.
We may acquire other companies or products or engage in strategic transactions, which could divert our management’s attention and cause us to incur various costs and expenses.
We may acquire or invest in businesses or products that we believe could complement or expand our business or otherwise offer growth opportunities. For example, we acquired Dimension in November 2017. The pursuit of potential acquisitions or investments may divert the attention of management and may cause us to incur various costs and expenses in identifying, investigating, and pursuing them, whether or not they are consummated. We may not be able to identify desirable acquisitions or investments or be successful in entering into an agreement for such transactions.
In addition, we may receive inquiries relating to potential strategic transactions, including collaborations, licenses, and acquisitions. Such potential transactions may divert the attention of management and may cause us to incur various costs and expenses in investigating and evaluating such transactions, whether or not they are consummated.
Litigation may substantially increase our costs and harm our business.
We may become party to lawsuits in the future, including, without limitation, actions and proceedings in the ordinary course of business relating to our stockholders, intellectual property, and employment matters, which will cause us to incur legal fees and other costs related thereto, including potential expenses for the reimbursement of legal fees of officers and directors under indemnification obligations. The expense of defending against such litigation may be significant and there can be no assurance that we will be successful in any defense. Further, the amount of time that may be required to resolve such lawsuits is unpredictable, and these actions may divert management’s attention from the day-to-day operations of our business, which could adversely affect our business, results of operations, and cash flows. Litigation is subject to inherent uncertainties, and an adverse result in such matters that may arise from time to time could have a material adverse effect on our business, results of operations, and financial condition.
Risks Related to Ownership of Our Common Stock
The market price of our common stock may be highly volatile.
The market price of our common stock has been, and is likely to continue to be, volatile, including for reasons unrelated to changes in our business. Our stock price could be subject to wide fluctuations in response to a variety of factors, including but not limited to the following:
|
|
• |
adverse results or delays in preclinical or clinical studies; |
|
|
• |
any inability to obtain additional funding; |
57
|
|
• |
the perception of limited market sizes or pricing for our product and product candidates; |
|
|
• |
decisions by our collaboration partners with respect to the indications for our product candidates in countries where they have the right to commercialize the product candidates; |
|
|
• |
decisions by our collaboration partners regarding market access and pricing in countries where they have the right to commercialize our product candidates; |
|
|
• |
failure to successfully develop and commercialize our product candidates; |
|
|
• |
the level of any revenue we receive from named patient sales; |
|
|
• |
post-marketing safety issues; |
|
|
• |
failure to maintain our existing strategic collaborations or enter into new collaborations; |
|
|
• |
failure by us or our licensors and strategic collaboration partners to prosecute, maintain, or enforce our intellectual property rights; |
|
|
• |
changes in laws or regulations applicable to our products; |
|
|
• |
any inability to obtain adequate product supply for our product and product candidates or the inability to do so at acceptable prices; |
|
|
• |
adverse regulatory decisions; |
|
|
• |
introduction of new products, services, or technologies by our competitors; |
|
|
• |
failure to meet or exceed financial projections we may provide to the public; |
|
|
• |
failure to meet or exceed the financial projections of the investment community; |
|
|
• |
the perception of the pharmaceutical industry by the public, legislatures, regulators, and the investment community; |
|
|
• |
the perception of the pharmaceutical industry’s approach to drug pricing; |
|
|
• |
announcements of significant acquisitions, strategic partnerships, joint ventures, or capital commitments by us, our strategic collaboration partners, or our competitors; |
|
|
• |
the integration and performance of any businesses we acquire; |
|
|
• |
disputes or other developments relating to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technologies; |
|
|
• |
additions or departures of key scientific or management personnel; |
|
|
• |
significant lawsuits, including patent or stockholder litigation; |
|
|
• |
securities or industry analysts’ reports regarding our stock, or their failure to issue such reports; |
|
|
• |
changes in the market valuations of similar companies; |
|
|
• |
general market or macroeconomic conditions; |
|
|
• |
sales of our common stock by us or our stockholders in the future; and |
|
|
• |
trading volume of our common stock. |
In addition, biotechnology and biopharmaceutical companies in particular have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our actual operating performance.
Future sales and issuances of our common stock or rights to purchase common stock, including pursuant to our equity incentive plans, could result in additional dilution of the percentage ownership of our stockholders and could cause our stock price to fall.
We will need additional capital in the future to continue our planned operations. To the extent we raise additional capital by issuing equity securities, our stockholders may experience substantial dilution. We may sell common stock, convertible securities, or other equity securities in one or more transactions at prices and in a manner we determine from time to time. If we sell common stock, convertible securities, or other equity securities in more than one transaction, investors may be materially diluted by subsequent sales. These sales may also result in material dilution to our existing stockholders, and new investors could gain rights superior to our existing stockholders.
58
Pursuant to our 2014 Incentive Plan, or the 2014 Plan, our management is authorized to grant stock options and other equity-based awards to our employees, directors, and consultants. At December 31, 2017, 1,070,108 shares were available for future grants under the 2014 Plan. The number of shares available for future grant under the 2014 Plan will automatically increase on January 1 of each year by the lesser of 2,500,000 shares or 4% of all shares of our capital stock outstanding as of December 31 of the prior calendar year, subject to the ability of our compensation committee to take action to reduce the size of the increase in any given year.
Pursuant to our 2014 Employee Stock Purchase Plan, or 2014 ESPP, eligible employees can acquire shares of our common stock at a discount to the prevailing market price. At December 31, 2017, 1,782,441 shares were available for issuance under the 2014 ESPP. The number of shares available for issuance under the 2014 ESPP will automatically increase on January 1 of each year by the lesser of 1,200,000 shares or 1% of all shares of our capital stock outstanding as of December 31 of the prior calendar year, subject to the ability of our compensation committee to take action to reduce the size of the increase in any given year.
Currently we plan to register the increased number of shares available under the 2014 Plan and the 2014 ESPP each year. If our board of directors elects to increase the number of shares available for future grant under the 2014 Plan or the 2014 ESPP, our stockholders may experience additional dilution, which could cause our stock price to fall.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
We have incurred substantial losses during our history and do not expect to become profitable in the near future nor may we ever achieve profitability. To the extent that we continue to generate taxable losses, unused taxable losses will, subject to certain limitations, carry forward to offset future taxable income, if any, until such unused losses expire. Under Sections 382 and 383 of the Internal Revenue Code of 1986, as amended, or the IRC, if a corporation undergoes an “ownership change,” generally defined as a greater than 50% change (by value) in its equity ownership over a three-year period, the corporation’s ability to use its pre-change net operating loss carryforwards, or NOL carryforwards, and other pre-change tax attributes (such as research tax credits) to offset its post-change income may be limited. An analysis to determine limitations upon our NOL carryforwards and other pre-change tax attributes for ownership changes that have occurred previously has been performed, resulting in a permanent decrease of federal and state NOL carryforwards in the amount of $7.2 million and a permanent decrease in federal research tax credit carryforwards in the amount of $0.2 million. As a result of these decreases and others that may occur as a result of future ownership changes, our ability to use our pre-change NOL carryforwards and other tax attribute carryforwards to offset U.S. federal taxable income and tax liabilities is limited and may become subject to even greater limitations, which could potentially accelerate or permanently increase future federal tax liabilities for us. In addition, there may be periods during which the use of state income tax NOL carryforwards and other state tax attribute carryforwards (such as state research tax credits) are suspended or otherwise limited, which could potentially accelerate or permanently increase future state tax liabilities for us.
The recently enacted comprehensive tax reform legislation could adversely affect our business and financial condition.
On December 22, 2017, the U.S. government enacted comprehensive tax legislation commonly referred to as the Tax Cuts and Jobs Act (Tax Act). The Tax Act makes broad and complex changes to the U.S. tax code, including, but not limited to, (1) reducing the U.S. federal corporate tax rate from 35% to 21%; (2) requiring companies to pay a one-time transition tax on certain unrepatriated earnings of foreign subsidiaries; (3) generally eliminating U.S. federal income taxes on dividends from foreign subsidiaries; (4) requiring a current inclusion in U.S. federal taxable income of certain earnings of controlled foreign corporations; (5) eliminating the corporate alternative minimum tax (AMT) and changing how existing AMT credits can be realized; (6) creating the base erosion anti-abuse tax (BEAT), a new minimum tax; (7) creating a new limitation on deductible interest expense; and (8) changing rules related to uses and limitations of net operating loss carryforwards created in tax years beginning after December 31, 2017. Notwithstanding the reduction in the corporate income tax rate, the overall impact of the new federal tax law is uncertain and our business and financial condition could be adversely affected. In addition, it is uncertain if and to what extent various states will conform to the newly enacted federal tax law. The determination of the benefit from (provision for) income taxes requires complex estimations, significant judgments and significant knowledge and experience concerning the applicable tax laws. Given that we are still in the transition period for the accounting for income tax effects of the Tax Act, the current assessment on deferred tax assets (liabilities) is based on the currently available information and guidance. If in the future any element of the tax reform changes the related accounting guidance for income tax, it could affect our income tax position and we may need to adjust the benefit from (provision for) income taxes accordingly.
We do not intend to pay dividends on our common stock so any returns will be limited to the value of our stock.
We have never declared or paid any cash dividends on our common stock. We currently intend to retain all available funds and any future earnings, if any, for the development, operation, and expansion of our business and do not anticipate declaring or paying any cash dividends for the foreseeable future. Any return to stockholders will therefore be limited to the appreciation of their stock.
59
Provisions in our amended and restated certificate of incorporation and by-laws, as well as provisions of Delaware law, could make it more difficult for a third-party to acquire us or increase the cost of acquiring us, even if doing so would benefit our stockholders, or remove our current management.
Our amended and restated certificate of incorporation, amended and restated by-laws, and Delaware law contain provisions that may have the effect of delaying or preventing a change in control of us or changes in our management. Our amended and restated certificate of incorporation and by-laws include provisions that:
|
|
• |
authorize “blank check” preferred stock, which could be issued by our board of directors without stockholder approval and may contain voting, liquidation, dividend, and other rights superior to our common stock; |
|
|
• |
create a classified board of directors whose members serve staggered three-year terms; |
|
|
• |
specify that special meetings of our stockholders can be called only by our board of directors or the chairperson of our board of directors; |
|
|
• |
prohibit stockholder action by written consent; |
|
|
• |
establish an advance notice procedure for stockholder approvals to be brought before an annual meeting of our stockholders, including proposed nominations of persons for election to our board of directors; |
|
|
• |
provide that our directors may be removed only for cause; |
|
|
• |
provide that vacancies on our board of directors may be filled only by a resolution adopted by the board of directors; |
|
|
• |
expressly authorize our board of directors to modify, alter or repeal our amended and restated by-laws; and |
|
|
• |
require holders of 75% of our outstanding common stock to amend specified provisions of our amended and restated certificate of incorporation and amended and restated by-laws. |
These provisions, alone or together, could delay, deter, or prevent hostile takeovers and changes in control or changes in our management.
In addition, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which limits the ability of stockholders owning in excess of 15% of our outstanding voting stock to merge or combine with us. Further, no stockholder is permitted to cumulate votes at any election of directors because this right is not included in our amended and restated certificate of incorporation.
Any provision of our amended and restated certificate of incorporation or amended and restated by-laws or Delaware law that has the effect of delaying or deterring a change in control could limit the opportunity for our stockholders to receive a premium for their shares of our common stock, and could also affect the price that some investors are willing to pay for our common stock.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the sole and exclusive forum for substantially all disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the sole and exclusive forum for (1) any derivative action or proceeding brought on our behalf, (2) any action asserting a claim of breach of fiduciary duty owed by any of our directors, officers, or other employees to us or to our stockholders, (3) any action asserting a claim against us arising under the Delaware General Corporation Law or under our amended and restated certificate of incorporation or bylaws, or (4) any action against us asserting a claim governed by the internal affairs doctrine. The choice of forum provision may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers, or other employees, which may discourage such lawsuits against us and our directors, officers, and other employees. Alternatively, if a court were to find the choice of forum provision contained in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could harm our business, operating results and financial condition.
Item 1B. Unresolved Staff Comments
None.
60
Our primary operations are conducted at the leased facilities described below.
We lease approximately 129,500 square feet of office space in Novato, California used primarily for corporate, clinical, regulatory, quality, manufacturing administration, and commercial functions. The leases for approximately 74,000 square feet will expire in April 2019, the lease for approximately 42,500 square feet will expire in December 2020, and the lease for approximately 13,000 will expire in April 2022.
We also lease approximately 63,000 square feet of office space in Brisbane, California. The rental term for this space will expire in June 2026.
We also lease approximately 15,000 square feet of office and laboratory space in Cambridge, Massachusetts. This lease will expire in January 2020.
We also lease approximately 17,600 square feet of laboratory and office space in Woburn, Massachusetts. This lease will expire in March 2021.
We believe our facilities are adequate and suitable for our current needs, and that we will be able to obtain new or additional leased space in the future when necessary.
We are not currently a party to any material legal proceedings. We may, however, in the ordinary course of business face various claims brought by third parties and we may, from time to time, make claims or take legal actions to assert our rights, including intellectual property rights as well as claims relating to employment matters and the safety or efficacy of our products. Any of these claims could subject us to costly litigation and, while we generally believe that we have adequate insurance to cover many different types of liabilities, our insurance carriers may deny coverage, may be inadequately capitalized to pay on valid claims, or our policy limits may be inadequate to fully satisfy any damage awards or settlements. If this were to happen, the payment of any such awards could have a material adverse effect on our consolidated operations, cash flows and financial position. Additionally, any such claims, whether or not successful, could damage our reputation and business.
Item 4. Mine Safety Disclosures
Not applicable.
61
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Our common stock has been traded on The Nasdaq Global Select Market since January 31, 2014 under the symbol “RARE”. The following tables set forth, for the periods indicated, the intraday high and low sales prices of our common stock as reported by Nasdaq.
|
|
|
Fiscal 2016 |
|
|||||
|
|
|
High |
|
|
Low |
|
||
|
First Quarter |
|
$ |
110.06 |
|
|
$ |
49.00 |
|
|
Second Quarter |
|
$ |
78.13 |
|
|
$ |
46.52 |
|
|
Third Quarter |
|
$ |
81.40 |
|
|
$ |
48.33 |
|
|
Fourth Quarter |
|
$ |
86.77 |
|
|
$ |
52.60 |
|
|
|
|
Fiscal 2017 |
|
|||||
|
|
|
High |
|
|
Low |
|
||
|
First Quarter |
|
$ |
91.35 |
|
|
$ |
65.55 |
|
|
Second Quarter |
|
$ |
68.46 |
|
|
$ |
51.67 |
|
|
Third Quarter |
|
$ |
71.99 |
|
|
$ |
49.57 |
|
|
Fourth Quarter |
|
$ |
58.00 |
|
|
$ |
43.14 |
|
As of February 15, 2018, we had 4 holders of record of our common stock. Certain shares are held in “street” name and, accordingly, the number of beneficial owners of such shares is not known or included in the foregoing number.
STOCK PRICE PERFORMANCE GRAPH
The following stock performance graph compares our total stock return with the total return for (i) the Nasdaq Composite Index and the (ii) the Nasdaq Biotechnology Index for the period from January 31, 2014 (the date our common stock commenced trading on the Nasdaq Global Market) through December 31, 2017. The figures represented below assume an investment of $100 in our common stock at the closing price of $42.25 on January 31, 2014 and in the Nasdaq Composite Index and the Nasdaq Biotechnology Index on January 31, 2014 and the reinvestment of dividends into shares of common stock. The comparisons in the table are required by the SEC and are not intended to forecast or be indicative of possible future performance of our common stock. This graph shall not be deemed “soliciting material” or be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or the Exchange Act, or otherwise subject to the liabilities under that section, and shall not be deemed to be incorporated by reference into any of our filings under the Securities Act of 1933, as amended, or the Securities Act, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
|
$100 investment in stock or index |
|
Ticker |
|
January 31, 2014 |
|
|
December 31, 2014 |
|
|
December 31, 2015 |
|
|
December 31, 2016 |
|
|
December 31, 2017 |
|
|
|||||
|
Ultragenyx Pharmaceutical Inc. |
|
RARE |
|
$ |
100.00 |
|
|
$ |
103.86 |
|
|
$ |
265.51 |
|
|
$ |
166.41 |
|
|
$ |
109.78 |
|
|
|
Nasdaq Composite Index |
|
^IXIC |
|
$ |
100.00 |
|
|
$ |
115.40 |
|
|
$ |
122.02 |
|
|
$ |
131.17 |
|
|
$ |
168.22 |
|
|
|
Nasdaq Biotechnology Index |
|
^NBI |
|
$ |
100.00 |
|
|
$ |
123.70 |
|
|
$ |
137.83 |
|
|
$ |
107.94 |
|
|
$ |
130.67 |
|
|
Dividend Policy
We have never declared or paid cash dividends on our common stock. We currently intend to retain all available funds and any future earnings, if any, to fund the development, operation, and expansion of our business, and we do not anticipate paying any cash dividends on our common stock in the foreseeable future. Any future determination to pay dividends will be made at the discretion of our board of directors or any authorized committee thereof.
62
Unregistered Sales of Equity Securities
None
Issuer’s Purchases of Equity Securities
None
Item 6. Selected Financial Data
The information set forth below for the five years ended December 31, 2017 is not necessarily indicative of the results that may be expected in the future and interim results are not necessarily indicative of results to be expected for the full year. You should read the selected historical financial data below in conjunction with the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the consolidated financial statements and related notes included elsewhere in this Annual Report.
|
|
Year Ended December 31, |
|
|||||||||||||||||
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|
2014 |
|
|
2013 |
|
|||||
|
|
(in thousands, except share and per share amounts) |
|
|||||||||||||||||
|
Consolidated Statements of Operations Data: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Collaboration and license |
$ |
2,136 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
— |
|
|
Product sales |
|
476 |
|
|
|
133 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Total revenues |
|
2,612 |
|
|
|
133 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of sales |
|
1 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Research and development |
|
231,644 |
|
|
|
183,204 |
|
|
|
114,737 |
|
|
|
45,967 |
|
|
|
27,829 |
|
|
Selling, general and administrative |
|
99,909 |
|
|
|
64,936 |
|
|
|
33,001 |
|
|
|
10,811 |
|
|
|
4,451 |
|
|
Total operating expenses |
|
331,554 |
|
|
|
248,140 |
|
|
|
147,738 |
|
|
|
56,778 |
|
|
|
32,280 |
|
|
Loss from operations |
|
(328,942 |
) |
|
|
(248,007 |
) |
|
|
(147,738 |
) |
|
|
(56,778 |
) |
|
|
(32,280 |
) |
|
Interest income |
|
4,074 |
|
|
|
3,789 |
|
|
|
2,320 |
|
|
|
608 |
|
|
|
216 |
|
|
Other income (expense) |
|
6,530 |
|
|
|
(1,621 |
) |
|
|
(200 |
) |
|
|
(3,632 |
) |
|
|
(3,006 |
) |
|
Loss before income taxes |
$ |
(318,338 |
) |
|
$ |
(245,839 |
) |
|
$ |
(145,618 |
) |
|
$ |
(59,802 |
) |
|
$ |
(35,070 |
) |
|
Benefit from (provision for) income taxes |
|
16,199 |
|
|
|
(35 |
) |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
Net loss |
$ |
(302,139 |
) |
|
$ |
(245,874 |
) |
|
$ |
(145,618 |
) |
|
$ |
(59,802 |
) |
|
$ |
(35,070 |
) |
|
Net loss attributable to common stockholders(1) |
$ |
(302,139 |
) |
|
$ |
(245,874 |
) |
|
$ |
(145,618 |
) |
|
$ |
(64,610 |
) |
|
$ |
(50,289 |
) |
|
Net loss per share attributable to common stockholders, basic and diluted(1) |
$ |
(7.12 |
) |
|
$ |
(6.21 |
) |
|
$ |
(3.96 |
) |
|
$ |
(2.25 |
) |
|
$ |
(14.87 |
) |
|
Shares used to compute net loss per share attributable to common stockholders, basic and diluted(1) |
|
42,453,135 |
|
|
|
39,586,908 |
|
|
|
36,782,603 |
|
|
|
28,755,758 |
|
|
|
3,382,489 |
|
|
|
(1) |
See Notes 2 and 13 to our audited consolidated financial statements of this Annual Report for an explanation of the calculations of basic and diluted net loss per share attributable to common stockholders. |
|
|
As of December 31, |
|
|||||||||||||||||
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|
2014 |
|
|
2013 |
|
|||||
|
|
(in thousands) |
|
|||||||||||||||||
|
Consolidated Balance Sheets Data: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash, cash equivalents and investments |
$ |
244,468 |
|
|
$ |
498,111 |
|
|
$ |
536,256 |
|
|
$ |
187,487 |
|
|
$ |
53,377 |
|
|
Working capital |
|
198,569 |
|
|
|
341,436 |
|
|
|
422,289 |
|
|
|
180,899 |
|
|
|
49,304 |
|
|
Total assets |
|
490,753 |
|
|
|
540,626 |
|
|
|
559,569 |
|
|
|
197,967 |
|
|
|
59,649 |
|
|
Convertible preferred stock warrant liability |
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
3,419 |
|
|
Convertible preferred stock |
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
124,930 |
|
|
Total stockholders' equity (deficit) |
|
383,454 |
|
|
|
473,974 |
|
|
|
531,090 |
|
|
|
184,945 |
|
|
|
(74,821 |
) |
63
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
You should read the following discussion and analysis of our financial condition and results of operations together with the section of this Annual Report entitled “Selected Financial Data” and our consolidated financial statements and related notes included elsewhere in this Annual Report.
Overview
We are a biopharmaceutical company focused on the identification, acquisition, development, and commercialization of novel products for the treatment of serious rare and ultra-rare diseases, with a focus on serious, debilitating genetic diseases. We target diseases for which the unmet medical need is high, the biology for treatment is clear, and for which there are no currently approved therapies. Since our inception in 2010, we have in-licensed potential treatments for multiple rare genetic disorders. Our strategy, which is predicated upon time- and cost-efficient drug development, allows us to pursue multiple programs in parallel with the goal of delivering safe and effective therapies to patients with the utmost urgency.
Our current approved therapy and clinical-stage pipeline consist of three product categories: biologics, small-molecule substrate replacement therapies, and gene therapy product candidates. Enzymes are proteins that the body uses to process materials needed for normal cellular function, and substrates are the materials upon which enzymes act. When enzymes or substrates are missing, the body is unable to perform its normal cellular functions, often leading to significant clinical disease. Several of our therapies are intended to replace deficient enzymes or substrates. Gene therapy is a therapeutic approach in which an isolated gene sequence or segment of DNA is administered to a patient, most commonly for the purpose of treating a genetic disease that is caused by mutations. Gene therapy aims to address the disease-causing effects of absent or dysfunctional genes by delivering functional copies of the gene to the patient’s cells, offering the potential for durable therapeutic benefit.
Our first product, Mepsevii (vestronidase alfa), is approved by the FDA for the treatment of children and adults with Mucopolysaccharidosis VII.
|
|
• |
On November 15, 2017, the U.S. FDA approved Mepsevii, the first medicine approved for the treatment of children and adults with Mucopolysaccharidosis VII, also known as MPSVII or Sly syndrome. Mepsevii is an intravenous, or IV, enzyme replacement therapy for the treatment of MPS VII, a rare lysosomal storage disease that often leads to multi-organ dysfunction, pervasive skeletal disease, and death. In Europe, the EMA is currently reviewing the MAA, and an opinion from the CHMP is expected in the first half of 2018. |
Our biologics pipeline includes burosumab in clinical development for the treatment of two diseases:
|
|
• |
Burosumab (KRN23 or UX023) is an antibody targeting fibroblast growth factor 23, or FGF23, in development for the treatment of X-linked hypophosphatemia, or XLH, a rare genetic disease that impairs bone growth. We are developing burosumab pursuant to our collaboration with Kyowa Hakko Kirin Co., Ltd., or KHK. We announced positive 64-week data from a Phase 2 study in pediatric patients ages five through 12 in April 2017, and in February 2018 we announced 64-week Phase 2 study in children less than five years old. We have an ongoing Phase 3 pediatric study with data expected in the second half of 2018. We also announced positive 48-week data from a Phase 3 study in adult XLH patients in December 2017, and positive 48-week bone biopsy data from a separate bone quality study in February 2018. In the U.S., the Prescription Drug User Fee Act (PDUFA) goal date for the BLA for burosumab for the treatment of adult and pediatric patients is April 17, 2018. In Europe, the Committee for Medicinal Products for Human Use, or CHMP, adopted a Positive Opinion recommending the conditional marketing authorization of burosumab for the treatment of XLH with radiographic evidence of bone disease in children one year of age and older and adolescents with growing skeletons. The CHMP's recommendation has been referred to the European Commission (EC), which is expected to render its final decision in February 2018. A filing for adults in Europe is planned after a decision is first reached on the pediatric indication. |
|
|
• |
Burosumab is also being developed for the treatment of tumor-induced osteomalacia, or TIO. TIO results from typically benign tumors that produce excess levels of FGF23, which can lead to severe hypophosphatemia, osteomalacia, fractures, fatigue, bone and muscle pain, and muscle weakness. We announced positive interim data from the Phase 2 study of burosumab in TIO in September 2017, and we expect 48-week data from this study in the first half of 2018. |
Our substrate replacement therapy pipeline includes UX007 in clinical development for the treatment of two diseases:
64
|
|
2018. We are simultaneously finalizing a full protocol for a Phase 3, randomized, controlled study examining major clinical events as the primary endpoint as discussed with the FDA, and plan to initiate this Phase 3 study in the second half of 2018. |
|
|
• |
UX007 is also being studied for the treatment of glucose transporter type-1 deficiency syndrome, or Glut1 DS, a rare metabolic disease of brain energy deficiency that is characterized by seizures, developmental delay, and movement disorder. Topline data from the Phase 2 seizure study, which we announced in the first quarter of 2017, indicated that the study did not meet the primary endpoint of reducing the frequency of the total number of observable and absence seizures among patients treated from baseline to Week 8 with UX007 compared to placebo. We are enrolling patients in the 3 study in movement disorders, and expect to announce data from this study in the second half of 2018. If positive, the movement disorder study could serve as the basis for regulatory submissions. |
Our gene therapy pipeline includes DTX301 in clinical development for the treatment of OTC deficiency:
|
|
• |
DTX301 is an adeno-associated virus (AAV8) gene therapy product candidate designed for the treatment of patients with OTC deficiency, the most common urea cycle disorder. In January 2018, we announced positive interim safety and efficacy results from the first dose cohort of the Phase 1/2 open-label clinical trial of DTX301. Full 12-week data from the first cohort is expected in March 2018. We expect to be able to move to the higher-dose second cohort pending the data monitoring committee's review of the 12-week safety data for all three patients in cohort 1, and data from the second cohort should be available in the second half of 2018. |
Financial Operations Overview
We are a biopharmaceutical company with a limited operating history. To date, we have invested substantially all of our efforts and financial resources in identifying, acquiring, and developing our product and product candidates, including conducting clinical studies and providing general and administrative support for these operations. To date, we have funded our operations primarily from the sale of equity securities.
We have never been profitable and have incurred net losses in each year since inception. Our net losses were $302.1 million, $245.9 million and $145.6 million for the years ended December 31, 2017, 2016 and 2015. Substantially all of our net losses have resulted from costs incurred in connection with our research and development programs and from selling, general and administrative costs associated with our operations.
Revenue
On November 15, 2017, the U.S. FDA approved Mepsevii for the treatment of children and adults with MPS VII. For the year ended December 31, 2017, we recorded $0.5 million in product sales from the sales of Mepsevii, which includes named patient sales, and $2.1 million in collaboration and license revenue from providing certain research and development services under our collaboration and license arrangement with Bayer. For the year ended December 31, 2016, we recorded named patient sales of $0.1 million as product sales.
Research and Development Expenses
Research and development expenses consist primarily of costs incurred for the development of our product candidates, which include:
|
|
• |
expenses incurred under agreements with clinical study sites that conduct research and development activities on our behalf; |
|
|
• |
expenses incurred under license agreements with third parties; |
|
|
• |
employee and consultant-related expenses, which include salaries, benefits, travel, and stock-based compensation; |
|
|
• |
laboratory and vendor expenses related to the execution of preclinical, non-clinical, and clinical studies; |
|
|
• |
the cost of acquiring, developing, and manufacturing clinical study materials; and |
|
|
• |
facilities, depreciation, and other expenses, which include direct and allocated expenses for rent and maintenance of facilities, insurance, and other supply costs. |
65
We expense all research and development costs in the periods in which they are incurred. Costs for certain development activities are recognized based on an evaluation of the progress to completion of specific tasks using information and data provided to us by our vendors and clinical sites. Nonrefundable advance payments for goods or services to be received in future periods for use in research and development activities are deferred and capitalized. The capitalized amounts are then expensed as the related goods are delivered and the services are performed.
The largest component of our total operating expenses has historically been our investment in research and development activities, including the clinical development of our product candidates. We allocate research and development salaries, benefits, stock-based compensation, and indirect costs to our product candidates on a program-specific basis, and we include these costs in the program-specific expenses. We expect our research and development expenses will increase in absolute dollars in future periods as we continue to invest in research and development activities related to developing our product candidates, and as programs advance into later stages of development and we enter into larger clinical studies. The process of conducting the necessary clinical research to obtain FDA approval is costly and time consuming and the successful development of our product candidates is highly uncertain. As a result, we are unable to determine the duration and completion costs of our research and development projects or when and to what extent, if any, we will generate revenue from the commercialization and sale of any of our product candidates.
Selling, General and Administrative Expenses
Selling, general and administrative expenses consist primarily of personnel costs, allocated facilities costs, and other expenses for outside professional services, including legal, human resources, audit, and accounting services and acquisition related costs. Personnel costs consist of salaries, benefits, and stock-based compensation. We expect that our selling, general and administrative expenses will increase in the future to support continued research and development activities, preparation for commercialization of our product and product candidates, and as a result of operating as a public company, including expenses related to compliance with the rules and regulations of the SEC and those of any national securities exchange on which our securities are traded, additional insurance expenses, investor relations activities, and other administration and professional services.
Interest Income
Interest income consists of interest earned on our cash, cash equivalents, and investments.
Other Income (Expense)
Other income (expense) primarily consists of foreign currency exchange gains and losses. Our foreign currency exchange gains and losses relate to transactions and asset and liability balances denominated in currencies other than the U.S. dollar.
Income Tax Benefit (Provision)
We recorded $47.4 million in non-current deferred tax liability resulting from the acquisition of Dimension Therapeutics, Inc. (Dimension), reflecting the tax impact of the difference between the book basis and tax basis of acquired in process research and development (IPR&D) assets. Subsequently, as a result of the reduction of the US corporate tax rate from 34% to 21% in December 2017, we recorded an income tax benefit of $16.2 million and reduced the deferred tax liability accordingly.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with GAAP. The preparation of these consolidated financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the reported expenses incurred during the reporting periods. Our estimates are based on our historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions. We periodically review our estimates as a result of changes in circumstances, facts and experience. The effects of material revisions in estimates are reflected in the financial statements prospectively from the date of the change in estimate. Our significant accounting policies are more fully described in Note 2 to our financial statements included elsewhere in this Annual Report.
We define our critical accounting policies as those accounting principles generally accepted in the United States of America that require us to make subjective estimates and judgments about matters that are uncertain and are likely to have a material impact on our financial condition and results of operations as well as the specific manner in which we apply those principles. We believe the critical accounting policies used in the preparation of our financial statements that require significant estimates and judgments are as follows:
66
Valuation of Goodwill and Acquired Intangible Assets
We have recorded goodwill and acquired intangible assets related to our acquisition of Dimension. When identifiable intangible assets, including IPR&D, are acquired, we determine the fair values of the assets as of the acquisition date. An income approach is used in these valuations and the models require the use of significant estimates and assumptions including but not limited to:
|
|
• |
timing and costs to complete the in-process projects; |
|
|
• |
timing and probability of success of clinical events or regulatory approvals; |
|
|
• |
estimated future cash flows from product sales resulting from completed products and in-process projects; and |
|
|
• |
discount rates commensurate with the stage of development of the projects and uncertainties in the economic estimates used in the projections. |
Intangible assets with definite useful lives are amortized over their estimated useful lives or other systematic basis and reviewed for impairment if certain events occur.
Intangible assets related to IPR&D projects are considered to be indefinite-lived until the completion or abandonment of the associated research and development efforts. During the period the assets are considered indefinite-lived, they will not be amortized but will be tested for impairment. Impairment testing is performed at least annually or when a triggering event occurs that could indicate a potential impairment. If and when development is complete, which generally occurs when regulatory approval to market a product is obtained, the associated assets are deemed finite-lived and are amortized over a period that best reflects the economic benefits provided by these assets.
If projects are not successfully developed, our sales and profitability may be adversely affected in future periods. Additionally, the value of the acquired intangible assets, including IPR&D, may become impaired if the underlying projects do not progress as we initially estimated. We believe that the assumptions used in developing our estimates of intangible asset values were reasonable at the time of the acquisition. However, the underlying assumptions used to estimate expected project sales, development costs, profitability, or the events associated with such projects, such as clinical results, may not occur as we estimated at the acquisition date.
Goodwill represents the excess of purchase price over fair value of net assets acquired in a business combination and is not amortized. Goodwill is subject to impairment testing at least annually or when a triggering event occurs that could indicate a potential impairment.
Accrued Research and Development, and Research and Development Expenses
As part of the process of preparing consolidated financial statements, we are required to estimate and accrue expenses, the largest of which is related to accrued research and development expenses. This process involves reviewing contracts and purchase orders, identifying services that have been performed on our behalf, and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of the actual costs.
We record accruals for estimated costs of research, preclinical and clinical studies, and manufacturing development. These costs are a significant component of our research and development expenses. A substantial portion of our ongoing research and development activities is conducted by third-party service providers. We accrue the costs incurred under our agreements with these third parties based on actual work completed in accordance with agreements established with these third parties. We determine the actual costs through discussions with internal personnel and external service providers as to the progress or stage of completion of the services and the agreed-upon fee to be paid for such services. We make significant judgments and estimates in determining the accrual balance in each reporting period. As actual costs become known, we adjust our accruals. Although we do not expect our estimates to be materially different from amounts actually incurred, our understanding of the status and timing of services performed relative to the actual status and timing of services performed may vary and could result in us reporting amounts that are too high or too low in any particular period. Our accrual is dependent, in part, upon the receipt of timely and accurate reporting from clinical research organizations and other third-party vendors.
Research and development costs are expensed as incurred and consist of salaries and benefits, stock-based compensation, lab supplies, materials and facility costs, as well as fees paid to other nonemployees and entities that conduct certain research and development activities on our behalf. Amounts incurred in connection with collaboration and license agreements are also included in research and development expense. Payments made prior to the receipt of goods or services to be used in research and development are capitalized until the goods or services are received.
To date, there have been no material differences from our accrued estimated expenses to the actual clinical trial expenses; however, due to the nature of estimates, we cannot assure you that we will not make changes to our estimates in the future as we become aware of additional information about the status or conduct of our clinical studies and other research activities.
67
Collaboration and License Revenue
We have certain collaboration and license arrangements in the scope of ASC 808, Collaborative Agreements. Funding received related to research and development services and pre-commercialization costs of such agreements is classified as a reduction of research and development expenses and selling, general and administrative expenses, respectively, in the consolidated statement of operations because the provision of such services for collaborative partners is not considered to be part of the our ongoing major or central operations.
We also receive royalty revenues under certain of our license or collaboration agreements in exchange for license of intellectual property. If we do not have any future performance obligations under these license or collaborations agreements, revenue is recorded as sales occur as part of collaboration and license revenue.
We have certain collaboration and license arrangements in the scope of ASC 606, Revenue from Contract with Customers. The terms of these agreements may contain multiple performance obligations, which may include licenses and research and development activities. Prior to recognizing revenue, we make estimates of the transaction price, including variable consideration that is subject to a constraint. Amounts of variable consideration are included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur and when the uncertainty associated with the variable consideration is subsequently resolved. Variable consideration may include nonrefundable upfront license fees, payments for research and development activities, reimbursement of certain third-party costs, payments based upon the achievement of specified milestones, and royalty payments based on product sales derived from the collaboration. If there are multiple distinct performance obligations, we allocate the transaction price to each distinct performance obligation based on its relative standalone selling price. The standalone selling price is generally determined based on the prices charged to customers or using expected cost plus margin. We recognize license and collaboration revenue for these arrangements by measuring the progress toward complete satisfaction of the performance obligations using an input measure.
Product sales
We sell Mepsevii through a limited number of distributors. Revenue from product sales is recognized at the point in time when the delivery is made and when title and risk of loss transfers to these distributors. We also recognize revenue from sales of Mepsevii on a “named patient” basis, which are allowed in certain countries prior to the commercial approval of the product in the territory. Prior to recognizing revenue, we make estimates of the transaction price, including variable consideration that is subject to a constraint. Amounts of variable consideration are included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. We estimate reserves for rebates payable under government mandated programs, chargebacks, distribution fees, estimated product returns and other deductions and record as a reduction of revenue at the time product revenues are recorded.
Inventories Produced in Preparation for Product Launches
We expense costs associated with the manufacture of our products prior to regulatory approval. Typically, capitalization of such inventory begins when we have received the regulatory approval of the product. Prior to the approval of Mepsevii by the FDA in November 2017, manufacturing and related costs were expensed; accordingly, these costs were not capitalized and as a result are not reflected in the costs of sales during the current period. If manufacturing and related costs were capitalized prior to the approval period, we expect that cost of sales for the year ended December 31, 2017 would have been approximately $39,000 for our commercial product sales. We expect cost of sales to increase in relation to product revenues as we deplete these previously expensed inventories and in turn, the inventory cost of Mepsevii will increase as we produce and then sell Mepsevii that has an inventory cost that reflects the full cost of manufacturing similar biologic products.
Stock-Based Compensation
Stock-based compensation costs related to equity awards granted to employees are measured at the date of grant based on the estimated fair value of the award, net of estimated forfeitures. We estimate the grant date fair value of options, and the resulting stock-based compensation expense, using the Black-Scholes option-pricing model. The grant date fair value of the stock-based awards is recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the respective awards. We expect to continue to grant stock options in the future, and to the extent that we do, our actual stock-based compensation will likely increase. The Black-Scholes option-pricing model requires the use of highly subjective assumptions which determine the estimated fair value of stock-based awards. These assumptions include:
|
|
• |
Expected term — The expected term represents the period that the stock-based awards are expected to be outstanding and is determined using the simplified method (based on the midpoint between the vesting date and the end of the contractual term). |
68
|
|
• |
Risk-free interest rate — The risk-free interest rate is based on the U.S. Treasury zero coupon issues in effect at the time of grant for periods corresponding with the expected term of option. |
|
|
• |
Expected dividend — We have never paid dividends on our common stock and have no plans to pay dividends on our common stock. Therefore, we used an expected dividend yield of zero. |
In addition to the assumptions used in the Black-Scholes option-pricing model, we also estimate a forfeiture rate to calculate the stock-based compensation for our awards. We will continue to use judgment in evaluating the expected volatility, expected terms, and forfeiture rates utilized for our stock-based compensation calculations on a prospective basis and will revise in subsequent periods, if actual forfeitures differ from those estimates.
For restricted stock units (RSUs) and performance stock units (PSUs), the fair value is based on the market value of our common stock on the date of grant. Stock-based compensation expense for RSUs is recognized on a straight-line basis over the requisite service period. As PSUs are subject to vest only if certain specified criteria are achieved and the employees’ continued service with us after achievement, compensation expense for PSUs is recognized on a straight-line basis between the period from the date on which the likelihood of the PSUs being earned is deemed probable and the expected vest date.
For the years ended December 31, 2017, 2016 and 2015 stock-based compensation expense was $68.0 million, $48.3 million and $24.9 million, respectively. As of December 31, 2017, we had $165.3 million of total unrecognized stock-based compensation costs, net of estimated forfeitures, which we expect to recognize over a weighted-average period of 2.52 years.
Income Taxes
We use the liability method of accounting for income taxes. Under this method, deferred tax assets and liabilities are determined based on the differences between the financial reporting and the tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. We assess the likelihood that the resulting deferred tax assets will be realized. A valuation allowance is provided when it is more likely than not that some portion or all of a deferred tax asset will not be realized.
In conjunction with the Dimension acquisition, we recorded a deferred tax liability reflecting the tax impact of the difference between the book basis and tax basis of acquired IPR&D. Such deferred income tax liability was not used to offset deferred tax assets when analyzing our valuation allowance as the acquired IPR&D is considered to have an indefinite life until we complete or abandon development of the acquired IPR&D.
We recognize benefits of uncertain tax positions if it is more likely than not that such positions will be sustained upon examination based solely on their technical merits, as the largest amount of benefit that is more likely than not to be realized upon the ultimate settlement. Our policy is to recognize interest and penalties related to the underpayment of income taxes as a component of income tax expense or benefit. To date, there have been no interest or penalties charged in relation to the unrecognized tax benefits.
As of December 31, 2017, our total deferred tax assets were $304.2 million, excluding the deferred tax liability generated from Dimension acquisition. Due to our lack of earnings history and uncertainties surrounding our ability to generate future taxable income, the net deferred tax assets have been fully offset by a valuation allowance. The deferred tax assets were primarily comprised of federal and state tax net operating losses and tax credit carryforwards. Utilization of the net operating loss and tax credit carryforwards may be subject to an annual limitation due to historical or future ownership percentage change rules provided by the Internal Revenue Code of 1986, and similar state provisions. The annual limitation may result in the expiration of certain net operating loss and tax credit carryforwards before their utilization.
69
Comparison of Years Ended December 31, 2017 and 2016
Revenues (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2017 |
|
|
2016 |
|
|
Change |
|
|
Change |
|
||||
|
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Collaboration and license |
$ |
2,136 |
|
|
$ |
— |
|
|
$ |
2,136 |
|
|
* |
|
|
|
Product sales |
|
476 |
|
|
|
133 |
|
|
|
343 |
|
|
|
258 |
% |
|
Total revenues |
$ |
2,612 |
|
|
$ |
133 |
|
|
$ |
2,479 |
|
|
* |
|
|
*not meaningful
We recognized revenue of $2.1 million in collaboration and license revenue for our research arrangement with Bayer and $0.5 million in product sales of Mepsevii for the year ended December 31, 2017 compared to $0.1 million in product sales for the year ended December 31, 2016. The increase in collaboration and license revenue is due to our acquisition of Dimension, and the increase in product sales is due to the approval of Mepsevii in November 2017.We expect our revenues to increase in the future as we meet milestones under our arrangement with Bayer and reflect product sales for full fiscal periods.
Research and Development Expenses (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2017 |
|
|
2016 |
|
|
Change |
|
|
Change |
|
||||
|
Development candidate: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
burosumab |
$ |
42,847 |
|
|
$ |
34,723 |
|
|
$ |
8,124 |
|
|
|
23 |
% |
|
vestronidase alfa |
|
33,472 |
|
|
|
29,707 |
|
|
|
3,765 |
|
|
|
13 |
% |
|
UX007 |
|
38,335 |
|
|
|
34,478 |
|
|
|
3,857 |
|
|
|
11 |
% |
|
Ace-ER |
|
34,630 |
|
|
|
32,532 |
|
|
|
2,098 |
|
|
|
6 |
% |
|
DTX301 |
|
472 |
|
|
|
— |
|
|
|
472 |
|
|
* |
|
|
|
Other research costs and preclinical costs |
|
81,888 |
|
|
|
51,764 |
|
|
|
30,124 |
|
|
|
58 |
% |
|
Total research and development expenses |
$ |
231,644 |
|
|
$ |
183,204 |
|
|
$ |
48,440 |
|
|
|
26 |
% |
Research and development expenses increased $48.4 million for the year ended December 31, 2017 compared to the same period in 2016. The increase in research and development expenses is primarily due to:
|
|
• |
for burosumab, an increase of $8.1 million related to the continued development of our clinical program, the enrollment of our Phase 3 adult and pediatric studies, regulatory filing preparation costs, patient identification efforts, and other development planning activities, net of KHK reimbursement; |
|
|
• |
for vestronidase alfa, an increase of $3.8 million related to regulatory filing preparation costs and the timing of manufacturing-related costs; |
|
|
• |
for UX007, an increase of $3.9 million primarily related to the initiation of our Phase 3 movement disorder study and support of investigator-sponsored studies across multiple diseases; |
|
|
• |
for Ace-ER, an increase of $2.1 million primarily related to our Phase 3 and extension studies and one-time manufacturing-related expenses incurred as a result of our decision to terminate the program; |
|
|
• |
for DTX301, an increase of expense of $0.5 million, related to the conduct of the Phase 1/2 study, as the program costs are reflected only after the acquisition of Dimension, and are primarily Phase 1 study costs; and |
|
|
• |
an increase of $30.1 million in other research and development costs including expenses in support of our clinical product candidate pipeline, expenses related to our existing research stage programs and research programs added with the Dimension acquisition, research collaborations, and certain cost allocations. |
We expect our research and development expenses to increase in the future as we advance our product candidates through clinical development. The timing and amount of expenses incurred will depend largely upon the outcomes of current or future clinical studies for our product candidates as well as the related regulatory requirements, manufacturing costs and any costs associated with the advancement of our preclinical programs.
70
Selling, General and Administrative Expenses (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2017 |
|
|
2016 |
|
|
Change |
|
|
Change |
|
||||
|
Selling, general and administrative |
$ |
99,909 |
|
|
$ |
64,936 |
|
|
$ |
34,973 |
|
|
|
54 |
% |
Selling, general and administrative expenses increased $35.0 million for the year ended December 31, 2017 compared to the same period in 2016. The increase in selling, general and administrative expenses was primarily due to increases in personnel costs resulting from an increase in the number of employees in support of our activities, stock-based compensation, acquisition-related costs, and commercial planning costs.
We expect selling, general and administrative expenses to increase to support our organizational growth and for our expected staged build out of our commercial organization over the next several years related to our approved product and multiple late-stage product candidates.
Interest Income (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2017 |
|
|
2016 |
|
|
Change |
|
|
Change |
|
||||
|
Interest income |
$ |
4,074 |
|
|
$ |
3,789 |
|
|
$ |
285 |
|
|
|
8 |
% |
Interest income increased $0.3 million for the year ended December 31, 2017 compared to the same period in 2016, primarily due to an increase in yields on our investment portfolio.
Other Income (Expense) (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2017 |
|
|
2016 |
|
|
Change |
|
|
Change |
|
||||
|
Other income (expense) |
$ |
6,530 |
|
|
$ |
(1,621 |
) |
|
$ |
8,151 |
|
|
|
-503 |
% |
Other income (expense) increased $8.2 million for the year ended December 31, 2017 compared to the same period in 2016. Other income (expense) primarily consists of gains (losses) resulting from the remeasurement of transactions denominated in foreign currencies. Our primary exposure to currency risk is related to intercompany balances with our foreign subsidiaries, resulting in the foreign currency gains and losses generated on the remeasurement of our intercompany balances with our foreign subsidiaries, which are reported in other income (expense). For the year ended December 31, 2017, we recorded a remeasurement gain of $10.0 million on intercompany balances which was offset by the transfer of foreign currency translation adjustment balance of $3.5 million as a result of the liquidation of a foreign entity due to the termination of the Ace-ER program. We expect other income to increase in the first quarter of 2018 as a result of the sale of our PRV to Novartis Pharma AG in January 2018.
Benefit from (provision for) income taxes
We recorded $47.4 million in non-current deferred tax liability resulting from the acquisition of Dimension, reflecting the tax impact of the difference between the book basis and tax basis of IPR&D assets. We recorded an income tax benefit of $16.2 million for the year ended December 31, 2017 due to the reduction of the US corporate tax rate from 34% to 21% and reduced the deferred tax liability accordingly. We also recorded nominal amounts in income tax provision for US states and certain foreign taxes.
Comparison of Years Ended December 31, 2016 and 2015
Revenue
We recognized revenue for $0.1 million of named patient sales of vestronidase alfa in Europe for the year ended December 31, 2016. We did not recognize any revenue for the year ended December 31, 2015.
71
Research and Development Expenses (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2016 |
|
|
2015 |
|
|
Change |
|
|
Change |
|
||||
|
Development candidate: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
burosumab |
$ |
34,723 |
|
|
$ |
12,886 |
|
|
$ |
21,837 |
|
|
|
169 |
% |
|
vestronidase alfa |
|
29,707 |
|
|
|
18,989 |
|
|
|
10,718 |
|
|
|
56 |
% |
|
UX007 |
|
34,478 |
|
|
|
19,952 |
|
|
|
14,526 |
|
|
|
73 |
% |
|
Ace-ER |
|
32,532 |
|
|
|
24,164 |
|
|
|
8,368 |
|
|
|
35 |
% |
|
Other research costs and preclinical costs |
|
51,764 |
|
|
|
38,746 |
|
|
|
13,018 |
|
|
|
34 |
% |
|
Total research and development expenses |
$ |
183,204 |
|
|
$ |
114,737 |
|
|
$ |
68,467 |
|
|
|
60 |
% |
Research and development expenses increased $68.5 million for the year ended December 31, 2016 compared to the same period in 2015. The increase in research and development expenses is primarily due to:
|
|
• |
for burosumab, an increase of $21.8 million related to the continued development of the XLH clinical program, including the enrollment of our Phase 3 adult study, the initiation of our Phase 3 pediatric study, the execution of our MAA filing, as well as continued clinical development and other regulatory activities for both the XLH and TIO clinical programs, net of KHK reimbursement; |
|
|
• |
for vestronidase alfa, an increase of $10.7 million related to our Phase 3 and Under 5 studies in addition to increases in manufacturing-related and quality activities; |
|
|
• |
for UX007, an increase of $14.5 million related to clinical manufacturing, the continued development of our clinical Phase 2 program for LC-FAOD, study start-up activities for our Phase 3 clinical study for Glut1 DS, as well as patient identification efforts and support of investigator-sponsored studies across multiple diseases; |
|
|
• |
for Ace-ER, an increase of $8.4 million related to the enrollment of our Phase 3 study, and manufacturing, quality, patient identification, and regulatory activities for this program; |
|
|
• |
an increase of $13.0 million in other research and development costs including expenses in support of our clinical product candidate pipeline, expenses related to our research stage programs and research collaborations, and certain cost allocations, including stock compensation. |
General and Administrative Expenses (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2016 |
|
|
2015 |
|
|
Change |
|
|
Change |
|
||||
|
General and administrative |
$ |
64,936 |
|
|
$ |
33,001 |
|
|
$ |
31,935 |
|
|
|
97 |
% |
General and administrative expenses increased $31.9 million for the year ended December 31, 2016 compared to the same period in 2015. The increase in general and administrative expenses was primarily due to increases in commercial planning costs, professional services costs, stock-based compensation, and personnel costs resulting from an increase in the number of employees in support of our activities
Interest Income (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2016 |
|
|
2015 |
|
|
Change |
|
|
Change |
|
||||
|
Interest income |
$ |
3,789 |
|
|
$ |
2,320 |
|
|
$ |
1,469 |
|
|
|
63 |
% |
Interest income increased $1.5 million for the year ended December 31, 2016 compared to the same period in 2015, primarily due to funds invested from our common stock offerings and increased yield on our investment portfolio.
Other Income (Expense) (dollars in thousands)
|
|
Year Ended December 31, |
|
|
Dollar |
|
|
% |
|
|||||||
|
|
2016 |
|
|
2015 |
|
|
Change |
|
|
Change |
|
||||
|
Other income (expense) |
$ |
(1,621 |
) |
|
$ |
(200 |
) |
|
$ |
(1,421 |
) |
|
|
711 |
% |
Other income (expense) decreased $1.4 million for the year ended December 31, 2016 compared to the same period in 2015. This was primarily due to the fluctuations of the volume of transactions and the foreign exchange rates used in the remeasurement of transactions denominated in foreign currencies.
72
Liquidity and Capital Resources
We have funded our operations primarily with net proceeds from our equity financings and equity sales in connection with a collaboration and license agreement. On July 1, 2016, we entered into an At-The-Market, or ATM, sales agreement with Cowen and Company, LLC, or Cowen, under which we may offer and sell from time to time common stock having aggregate gross proceeds of up to $150.0 million. During the year ended December 31, 2017, the proceeds from the offering were approximately $67.6 million, after commissions and other offering costs. On July 27, 2017, we entered into another ATM sales agreement with Cowen and during the year ended December 31, 2017, the proceeds from the offering were approximately $64.3 million, after commissions and other offering costs.
As of December 31, 2017, we had $244.5 million in available cash, cash equivalents, and investments. Our cash, cash equivalents and investments are held in a variety of deposit accounts, interest-bearing accounts, corporate bond securities, U.S government securities and money market funds. Cash in excess of immediate requirements is invested with a view toward liquidity and capital preservation, and we seek to minimize the potential effects of concentration and credit risk.
The following table summarizes our cash flows for the periods indicated (in thousands):
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Cash used in operating activities |
|
$ |
(253,843 |
) |
|
$ |
(160,975 |
) |
|
$ |
(105,977 |
) |
|
Cash provided by (used in) investing activities |
|
|
56,416 |
|
|
|
89,915 |
|
|
|
(292,351 |
) |
|
Cash provided by financing activities |
|
|
136,267 |
|
|
|
138,676 |
|
|
|
467,573 |
|
|
Effect of exchange rate changes on cash |
|
|
528 |
|
|
|
(65 |
) |
|
|
— |
|
|
Net increase (decrease) in cash and cash equivalents |
|
$ |
(60,632 |
) |
|
$ |
67,551 |
|
|
$ |
69,245 |
|
Cash Used in Operating Activities
Our primary use of cash is to fund operating expenses, which consist primarily of research and development expenditures. Due to our significant research and development expenditures, we have generated significant operating losses since our inception. Cash used to fund operating expenses is affected by the timing of when we pay these expenses, as reflected in the change in our outstanding accounts payable and accrued expenses.
Cash used in operating activities for the year ended December 31, 2017 was $253.8 million and reflected a net loss of $302.1 million, offset by non-cash charges of $68.0 million for stock-based compensation, $1.7 million for the amortization of premium paid on purchased investments, $5.8 million for depreciation and amortization and a decrease of $7.0 million for foreign currency remeasurement gain due to an increase in foreign entity transactions and fluctuations in the foreign exchange rate during the period. Cash used in operating activities also reflected a $3.3 million decrease in prepaid expenses and other current assets primarily due to an increase in accounts receivables under a collaboration agreement and other miscellaneous prepayments, a $4.6 million decrease in accrued expenses and other liabilities due to decrease in liabilities for Ace-ER related vendors and a reduction of the repayment liability for the Takeda collaboration agreement, and a $16.2 million decrease in deferred tax liabilities recorded in conjunction with the Dimension acquisition which were then revalued as a result of the reduction of the US corporate tax rate in December 2017 from 34% to 21%. These decreases were offset by a $0.5 million increase in non-current assets and a $3.5 million increase in accounts payable primarily due to increased spend and the timing of payments.
Cash used in operating activities for the year ended December 31, 2016 was $161.0 million and reflected a net loss of $245.9 million, offset by non-cash charges of $48.3 million for stock-based compensation, $4.8 million for the amortization of premium paid on purchased investments, $3.4 million for depreciation and amortization, $1.3 million for a foreign currency remeasurement loss due to an increase in foreign entity transactions and fluctuations in the foreign exchange rate during the period, and $0.7 million for the estimated fair value of a license fee in conjunction with the Takeda collaboration agreement. Cash used in operating activities also reflected a $7.1 million increase in prepaid expenses and other current assets primarily due to an increase in prepaid manufacturing and an increase in receivables related to a tenant improvement allowance, and a $1.2 million increase in non-current assets as result of an increase in upfront payments to contract research organizations and in clinical study costs. These increases were offset by a $32.2 million increase in accrued expenses and other liabilities as a result of an accrual for a liability under a collaboration agreement, increase in clinical study, manufacturing, and related costs as we continued to increase our research and development activities and employee compensation in bonuses and vacation due to higher headcount, and a $2.5 million increase in accounts payable primarily due to increased spend and the timing of payments.
73
Cash used in operating activities for the year ended December 31, 2015 was $106.0 million and reflected a net loss of $145.6 million, offset by non-cash charges of $5.6 million for the amortization of premium paid on purchased investments, $1.4 million for depreciation and amortization, and $24.9 million for stock-based compensation. Cash used in operating activities also reflected a $7.1 million increase in prepaid expenses and other current assets primarily due to an increase in CRO prepaid clinical costs, an increase in KHK receivable and an increase in interest receivable, and a $2.0 million decrease in accounts payable primarily due to the timing of payments. These increases were offset by a $0.2 million decrease in non-current assets as result of a decrease in manufacturing prepaid expenses and a $16.7 million increase in accrued expenses and other liabilities as a result of an increase in clinical study, manufacturing, related costs as we continued to increase our research and development activities and employee bonuses.
Cash Provided by (Used in) Investing Activities
Cash provided by investing activities for the year ended December 31, 2017 was $56.4 million and related to proceeds from maturities of investments of $273.6 million and the sale of investments of $157.9 million, and a decrease of $0.9 million in restricted cash related to line of credit reductions under our current lease agreements, offset by $142.8 million of cash used for the Dimension acquisition, purchases of property and equipment of $2.8 million, and purchases of investments of $230.5 million.
Cash provided by investing activities for the year ended December 31, 2016 was $89.9 million and related to purchases of investments of $442.5 million, purchases of property and equipment of $10.2 million and an increase of $1.2 million in restricted cash for the expansion of the space under our current lease, offset by proceeds from maturities of investments of $403.2 million and the sale of investments of $140.6 million.
Cash used in investing activities for the year ended December 31, 2015 was $292.4 million and related to purchases of investments of $624.2 million, purchases of property and equipment of $5.0 million and an increase of $1.5 million in restricted cash for the expansion of the space under our current lease, offset by proceeds from maturities of investments of $249.0 million and the sale of investments of $89.3 million.
Cash Flows Provided by Financing Activities
Cash provided by financing activities for the year ended December 31, 2017 was $136.3 million and was comprised of $132.0 million from the sale of common stock from our ATM offering and $9.3 million in net proceeds from the issuance of common stock upon the exercise of stock options and the vesting of restricted stock units, offset by payment of notes payable of $4.9 million.
Cash provided by financing activities for the year ended December 31, 2016 was $138.7 million and was comprised of $79.5 million from the sale of common stock from our ATM offering, $51.4 million from the sale of common stock to Takeda, and $7.8 million in net proceeds from the issuance of common stock upon the exercise of stock options and the vesting of restricted stock units.
Cash provided by financing activities for the year ended December 31, 2015 was $467.6 million and was comprised of $461.1 million from our underwritten public offerings and $6.5 million in net proceeds from the issuance of common stock upon the exercise of stock options and warrants and the vesting of restricted stock units.
Funding Requirements
In January 2018, we had $11.8 million in proceeds, net of commissions and other offering costs, from the offering related to the ATM sales agreement with Cowen. In January 2018, we completed the sale of our rare pediatric disease PRV for $130.0 million which we received in November 2017 from the FDA in connection with the approval of Mepsevii. Lastly, in January 2018, we also completed an underwritten public offering in which we sold 5,043,860 shares of common stock and received net proceeds of approximately $271.0 million.
We anticipate, excluding non-recurring items, that we will continue to generate annual losses for the foreseeable future, and we expect the losses to increase as we continue the development of, and seek regulatory approvals for, our product candidates, and begin to commercialize our approved product. Due to certain non-recurring or infrequent items like the sale of the PRV, we may have lower levels of losses or income in the near term in quarterly periods that may not be indicative of future periods or trends. We will likely require additional capital to fund our operations, complete our ongoing and planned clinical studies and commercialize our product, and funding may not be available to us on acceptable terms or at all.
If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may be required to delay, limit, reduce the scope of, or terminate one or more of our clinical studies, research and development programs, future commercialization efforts, or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
Our future funding requirements will depend on many factors, including the following:
|
|
• |
the scope, rate of progress, results and cost of our clinical studies, nonclinical testing, and other related activities; |
|
|
• |
the cost of manufacturing clinical supplies, and establishing commercial supplies, of our product candidates and any products that we may develop; |
74
|
|
• |
the cost, timing, and outcomes of regulatory approvals; |
|
|
• |
the cost and timing of establishing our commercial infrastructure, and distribution capabilities; and |
|
|
• |
the terms and timing of any collaborative, licensing, and other arrangements that we may establish, including any required upfront milestone and royalty payments thereunder. |
We expect to satisfy future cash needs through existing capital balances and through some combination of public or private equity offerings, debt financings, collaborations, strategic alliances, licensing arrangements, and other marketing and distribution arrangements. Please see “Risk Factors—Risks Related to Our Financial Condition and Capital Requirements.”
Contractual Obligations
We have contractual obligations from our operating leases, manufacturing and service contracts, licenses, royalties, development and collaboration arrangements, and other research and development activities. The following table summarizes our significant binding contractual obligations at December 31, 2017 (in thousands):
|
|
Payments due by period |
|
|||||||||||||||||
|
|
Less than 1 year |
|
|
1 to 3 years |
|
|
3 to 5 years |
|
|
More than 5 years |
|
|
Total |
|
|||||
|
Operating leases |
$ |
6,210 |
|
|
$ |
9,020 |
|
|
$ |
4,890 |
|
|
$ |
9,033 |
|
|
$ |
29,153 |
|
|
Manufacturing and service contracts |
|
7,542 |
|
|
|
1,286 |
|
|
$ |
- |
|
|
$ |
— |
|
|
$ |
8,828 |
|
|
Total |
$ |
13,752 |
|
|
$ |
10,306 |
|
|
$ |
4,890 |
|
|
$ |
9,033 |
|
|
$ |
37,981 |
|
The terms of certain of our license and collaboration agreements require us to pay potential future milestone payments based on product development success. The above table excludes milestone or contractual payment obligations as the amount and timing of such obligations are unknown or uncertain, which potential obligations are further described in Note 12 to the accompanying Consolidated Financial Statements.
Recent Accounting Pronouncements
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842), which requires an entity that is a lessee to record a right of use asset and a corresponding lease liability on the balance sheet for all leases with terms longer than 12 months. This guidance also requires disclosures about the amount, timing, and uncertainty of cash flows arising from leases. This guidance is effective for annual reporting periods beginning after December 15, 2018, and interim periods within those annual periods, using a modified retrospective approach, and early adoption is permitted. We are evaluating the effect that this guidance will have on our consolidated financial statements and related disclosures.
In October 2016, the FASB issued ASU 2016-16, Income Taxes - Intra-Entity Transfers of Assets Other Than Inventory, which requires entities to recognize the income tax consequences of an intra-entity transfer of an asset other than inventory when the transfer occurs. The standard is effective for fiscal years beginning after December 15, 2017, including interim periods within those fiscal years. Early adoption is permitted as of the beginning of a fiscal year. The new standard must be adopted using a modified retrospective transition method which is a cumulative-effective adjustment to retained earnings as of the beginning of the first effective reporting period. We are evaluating the effect that this guidance will have on our consolidated financial statements and related disclosures; however we expect the impact to be immaterial.
In November 2016, the FASB issued ASU 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash, which requires restricted cash to be presented with cash and cash equivalents on the statement of cash flows and disclosure of how the statement of cash flows reconciles to the balance sheet if restricted cash is shown separately from cash and cash equivalents on the balance sheet. The guidance is effective for interim and annual periods beginning after December 15, 2017, and early adoption is permitted. We are evaluating the effect that this guidance will have on our statement of cash flows and related disclosures; however we expect the impact to be immaterial.
In January 2017, the FASB issued ASU 2017-04, Intangibles – Goodwill and Other (Topic 350): Simplifying the Test for Goodwill Impairment, which will eliminate the requirement to calculate the implied fair value of goodwill, commonly referred to as “Step 2” in the current goodwill impairment test. An entity will still have the option to perform the qualitative assessment for a reporting unit to determine if the quantitative impairment test is necessary. This guidance will be effective for annual and interim impairment tests performed in annual reporting periods beginning after December 15, 2020, and early adoption is permitted for annual or interim impairment tests performed after January 1, 2017. We are evaluating the effect that this guidance will have on our consolidated financial statements and related disclosures.
75
Off-Balance Sheet Arrangements
Since our inception in April 2010, we have not engaged in any off-balance sheet arrangements, as defined in the rules and regulations of the SEC.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Our exposure to market risk for changes in interest rates relates primarily to interest earned on our cash equivalents and investments. The primary objective of our investment activities is to preserve our capital to fund operations. A secondary objective is to maximize income from our investments without assuming significant risk. Our investment policy provides for investments in low-risk, investment-grade debt instruments. As of December 31, 2017, we had cash, cash equivalents, and investments totaling $244.5 million which includes bank deposits, money market funds, U.S. government treasury and agency securities, and investment-grade corporate bond securities which are subject to default, changes in credit rating, and changes in market value. The securities in our investment portfolio are classified as available for sale and are subject to interest rate risk and will decrease in value if market interest rates increase. A hypothetical 100 basis point change in interest rates during any of the periods presented would not have had a material impact on our consolidated financial statements. To date, we have not experienced a loss of principal on any of our investments.
We face foreign exchange risk as a result of entering into transactions denominated in currencies other than U.S. dollars. Due to the uncertain timing of expected payments in foreign currencies, we do not utilize any forward exchange contracts. All foreign transactions settle on the applicable spot exchange basis at the time such payments are made. An adverse movement in foreign exchange rates could have a material effect on payments made to foreign suppliers and for license agreements. Our primary exposure to currency risk is related to intercompany balances with our foreign subsidiaries, resulting in the foreign currency gains and losses generated on the remeasurement of our intercompany balances with our foreign subsidiaries, which are reported in other income (expense). The intercompany amounts are largely offset by the translation gains (losses) reported in other comprehensive income (loss), resulting in immaterial impact on stockholders’ equity. A hypothetical 10% change in foreign exchange rates during any of the periods presented would not have had a material impact on our consolidated financial statements.
Item 8. Financial Statements and Supplementary Data
Our financial statements are annexed to this Annual Report beginning on page F-1 and are incorporated by reference into this Item 8.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
76
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
We carried out an evaluation, under the supervision and with the participation of our management, including our Chief Executive Officer and our Chief Financial Officer, of the effectiveness of our “disclosure controls and procedures” as of the end of the period covered by this Annual Report, pursuant to Rules 13a-15(b) and 15d-15(b) under the Exchange Act. In connection with that evaluation, our Chief Executive Officer and our Chief Financial Officer concluded that our disclosure controls and procedures were effective and designed to provide reasonable assurance that the information required to be disclosed is recorded, processed, summarized and reported within the time periods specified in the SEC rules and forms as of December 31, 2017. For the purpose of this review, disclosure controls and procedures means controls and procedures designed to ensure that information required to be disclosed by us in the reports that we file or submit is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. These disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in the reports that we file or submit is accumulated and communicated to management, including our principal executive officer and principal financial officer, as appropriate to allow timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, our management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and our management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Management’s Annual Report on Internal Control Over Financial Reporting
Management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act. Our management used the Committee of Sponsoring Organizations of the Treadway Commission Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), or the COSO framework, to evaluate the effectiveness of internal control over financial reporting. Management believes that the COSO framework is a suitable framework for its evaluation of financial reporting because it is free from bias, permits reasonably consistent qualitative and quantitative measurements of our internal control over financial reporting, is sufficiently complete so that those relevant factors that would alter a conclusion about the effectiveness of our internal control over financial reporting are not omitted and is relevant to an evaluation of internal control over financial reporting.
Management has assessed the effectiveness of our internal control over financial reporting as of December 31, 2017 and has concluded that such internal control over financial reporting is effective.
Management’s assessment of internal control over financial reporting as of December 31, 2017 excluded Dimension’s internal control over financial reporting because we acquired Dimension in a purchase business combination in November 2017. This exclusion is in accordance with the general guidance issued by the Staff of the SEC that an assessment of a recent business combination may be omitted from management’s report on internal control over financial reporting in the first year of consolidation.
Our independent registered public accounting firm, Ernst & Young LLP, has audited the financial statements included in this Annual Report and has issued a report on the effectiveness of our internal control over financial reporting. The report of Ernst & Young LLP is included below.
Changes in Internal Control over Financial Reporting
As discussed above, on November 7, 2017, we completed our acquisition of Dimension and Dimension became our wholly owned subsidiary. As a result of the Dimension acquisition, the internal control over financial reporting utilized by us prior to the acquisition became the internal control over financial reporting of Dimension, and we are currently in the process of evaluating and integrating Dimension’s historical internal control over financial reporting with ours.
Other than continuing changes to our internal control processes resulting from the Dimension acquisition as discussed above, there have been no changes in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) during our fourth quarter ended December 31, 2017, that have materially affected, or are reasonably likely to materially affect our internal control over financial reporting.
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of Ultragenyx Pharmaceutical Inc.:
Opinion on Internal Control over Financial Reporting
We have audited Ultragenyx Pharmaceutical, Inc.’s internal control over financial reporting as of December 31, 2017, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, Ultragenyx Pharmaceutical Inc. (the “Company”) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2017, based on the COSO criteria.
77
As indicated in the accompanying Management’s Annual Report on Internal Control Over Financial Reporting, management’s assessment of and conclusion on the effectiveness of internal control over financial reporting did not include the internal controls of Dimension Therapeutics, Inc., which is included in the 2017 consolidated financial statements of the Company and constituted 8% and 8% of total and net assets, respectively, as of December 31, 2017 and 81% and 2% of revenues and net income, respectively, for the year then ended. Our audit of internal control over financial reporting of the Company also did not include an evaluation of the internal control over financial reporting of Dimension Therapeutics, Inc.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2017 and 2016, the related consolidated statements of operations, comprehensive loss, stockholders’ equity, and cash flows, for each of the three years in the period ended December 31, 2017 and related notes and our report dated February 21, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management's Annual Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
San Jose, California
February 21, 2018
None.
78
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this Item is incorporated herein by reference to information in the proxy statement for our 2018 Annual Meeting of Stockholders, which we will file with the SEC within 120 days of the end of the fiscal year to which this Annual Report relates (the “2018 Proxy Statement”), including under the headings “Proposal No. 1—Election of Class II Directors,” “Executive Officers,” “Section 16(a) Beneficial Ownership Reporting Compliance,” “Corporate Governance—Code of Business Conduct and Ethics,” “Proposal No. 1—Election of Class II Directors—Nomination of Directors” and “Board of Directors and Committees.” We have adopted a code of ethics that applies to all of our directors, officers and employees, including our principal executive, principal financial and principal accounting officers, or persons performing similar functions, or Code of Ethics. Our Code of Ethics is posted on our corporate governance website located at www.ultragenyx.com. We intend to disclose future amendments to certain provisions of the Code of Ethics, and waivers of the Code of Ethics granted to executive officers and directors, on the website within four business days following the date of the amendment or waiver.
Item 11. Executive Compensation
The information required by this Item is incorporated herein by reference to information in the 2018 Proxy Statement, including under the headings “Executive Compensation,” “Director Compensation,” “Board of Directors and Committees—Compensation Committee Interlocks and Insider Participation,” “Executive Compensation—Risk Management and Mitigation,” and “Executive Compensation—Compensation Committee Report.”
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this Item is incorporated herein by reference to information in the 2018 Proxy Statement, including under the headings “Security Ownership of Certain Beneficial Owners and Management” and “Equity Compensation Plan Information.”
Item 13. Certain Relationships and Related Transactions, and Director Independence
The information required by this Item is incorporated herein by reference to information in the 2018 Proxy Statement, including under the headings “Certain Relationships and Related-Person Transactions,” “Corporate Governance,” and “Board of Directors and Committees.”
Item 14. Principal Accountant Fees and Services
The information required by this Item is incorporated herein by reference to information in the 2018 Proxy Statement, including under the heading “Proposal No. 2—Ratification of the Selection of Independent Registered Public Accounting Firm.”
79
Item 15. Exhibits and Financial Statement Schedules
|
|
(a) |
The following documents are filed as part of this Annual Report. |
|
|
(1) |
Consolidated Financial Statements |
|
|
|
Consolidated Financial Statements—See Index to Consolidated Financial Statements at page F-1 of this Annual Report. |
|
|
(2) |
Consolidated Financial Statement Schedules |
|
|
|
Consolidated Financial statement schedules have been omitted in this Annual Report because they are not applicable, not required under the instructions, or the information requested is set forth in the consolidated financial statements or related notes thereto. |
|
|
(b) |
Exhibits |
|
Exhibit |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed |
||||
|
|
|
Form |
|
Date |
|
Number |
|
Herewith |
||
|
2.1 |
|
|
|
8-K |
|
10/2/2017 |
|
2.1 |
|
|
|
2.2 |
|
|
|
|
|
|
|
|
|
X |
|
3.1 |
|
|
|
8-K |
|
2/5/2014 |
|
3.1 |
|
|
|
3.2 |
|
|
|
8-K |
|
2/5/2014 |
|
3.2 |
|
|
|
4.1 |
|
Reference is made to Exhibits 3.1 and 3.2 |
|
|
|
|
|
|
|
|
|
4.2 |
|
|
|
S-1 |
|
11/8/2013 |
|
4.2 |
|
|
|
4.3 |
|
Warrant, dated as of June 30, 2010, issued to Emil D. Kakkis, M.D., Ph.D. |
|
S-1 |
|
11/8/2013 |
|
4.3 |
|
|
|
4.4 |
|
Warrant, dated as of June 14, 2011, issued to Emil D. Kakkis, M.D., Ph.D. |
|
S-1 |
|
11/8/2013 |
|
4.6 |
|
|
|
4.5 |
|
Warrant, dated as of June 14, 2011, issued to Emil D. Kakkis, M.D., Ph.D. |
|
S-1 |
|
11/8/2013 |
|
4.7 |
|
|
|
10.1† |
|
|
|
S-1/A |
|
12/23/2013 |
|
10.1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.2 |
|
|
10-Q |
|
11/10/2015 |
|
10.2 |
|
|
|
|
10.3 |
|
|
|
|
|
|
|
|
X |
|
|
10.4* |
|
|
|
|
|
|
|
|
X |
|
|
10.5* |
|
|
|
|
|
|
|
|
X |
|
80
|
Exhibit |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed |
||||
|
|
|
Form |
|
Date |
|
Number |
|
Herewith |
||
|
10.6† |
|
|
|
S-1/A |
|
12/23/2013 |
|
10.3 |
|
|
|
10.7† |
|
|
|
S-1 |
|
11/8/2013 |
|
10.4 |
|
|
|
10.8† |
|
|
|
S-1/A |
|
12/23/2013 |
|
10.7 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.9 |
|
|
10-Q |
|
8/11/2014 |
|
10.1 |
|
|
|
|
10.10† |
|
|
|
S-1/A |
|
12/23/2013 |
|
10.8 |
|
|
|
10.11 |
|
|
|
|
|
|
|
|
|
X |
|
10.12† |
|
|
|
10-Q/A |
|
12/12/2016 |
|
10.1 |
|
|
|
10.13† |
|
|
|
|
|
|
|
|
|
X |
|
10.14† |
|
|
|
|
|
|
|
|
|
X |
|
10.15† |
|
|
|
|
|
|
|
|
|
X |
|
10.16† |
|
|
|
|
|
|
|
|
|
X |
|
10.17* |
|
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
10.18* |
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.19* |
|
|
|
|
|
|
|
|
X |
|
81
|
Exhibit |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed |
||||
|
|
|
Form |
|
Date |
|
Number |
|
Herewith |
||
|
10.20 |
|
|
|
10-Q |
|
7/28/2017 |
|
1.1 |
|
|
|
10.21# |
|
|
|
S-1 |
|
11/8/2013 |
|
10.11 |
|
|
|
10.22# |
|
|
|
S-1 |
|
11/8/2013 |
|
10.12 |
|
|
|
10.23# |
|
|
|
10-K |
|
2/17/2017 |
|
10.20 |
|
|
|
10.24# |
|
|
|
S-1/A |
|
1/17/2014 |
|
10.14 |
|
|
|
10.25# |
|
|
|
S-1/A |
|
1/17/2014 |
|
10.15 |
|
|
|
10.26# |
|
Form of Non Statutory Stock Option Agreement (Employees)(ex-U.S.) |
|
10-Q |
|
5/10/2016 |
|
10.3 |
|
|
|
10.27# |
|
|
|
S-1/A |
|
1/17/2014 |
|
10.16 |
|
|
|
10.28# |
|
|
|
10-Q |
|
5/10/2016 |
|
10.1 |
|
|
|
10.29# |
|
Form of Restricted Stock Unit Agreement (Employees)(ex-U.S.) |
|
10-Q |
|
5/10/2016 |
|
10.2 |
|
|
|
10.30# |
|
|
|
S-1/A |
|
1/17/2014 |
|
10.18 |
|
|
|
10.31# |
|
Form of Performance Stock Unit Agreement (Current Employees) |
|
|
|
|
|
|
|
X |
|
10.32# |
|
|
|
|
|
|
|
|
|
X |
|
10.33# |
|
|
|
10-K |
|
2/17/2017 |
|
10.28 |
|
|
|
10.34# |
|
|
|
S-1/A |
|
1/17/2014 |
|
10.27 |
|
|
|
10.35# |
|
|
|
S-1 |
|
11/8/2013 |
|
10.18 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.36# |
|
|
10-Q |
|
8/11/2014 |
|
10.2 |
|
|
|
|
10.37# |
|
|
|
S-1 |
|
11/8/2013 |
|
10.19 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.38# |
|
|
10-Q |
|
8/11/2014 |
|
10.3 |
|
|
|
|
10.39# |
|
Offer Letter, dated as of March 12, 2012, between Ultragenyx Pharmaceutical Inc. and Shalini Sharp |
|
S-1 |
|
11/8/2013 |
|
10.20 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.40# |
|
|
10-Q |
|
8/11/2014 |
|
10.4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.41# |
|
|
10-Q |
|
8/9/2016 |
|
10.3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
82
|
Exhibit |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed |
||||
|
|
|
Form |
|
Date |
|
Number |
|
Herewith |
||
|
|
Offer Letter, dated as of February 20, 2015, between Ultragenyx Pharmaceutical Inc. and Dennis Huang |
|
10-K |
|
2/17/2017 |
|
10.36 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.43# |
|
|
10-K |
|
2/17/2017 |
|
10.37 |
|
|
|
|
10.44# |
|
|
10-K |
|
2/17/2017 |
|
10.38 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.45# |
|
|
10-K |
|
2/17/2017 |
|
10.39 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.46# |
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.47# |
|
|
10-K |
|
3/24/2014 |
|
10.23 |
|
|
|
|
10.48 |
|
|
|
S-1 |
|
11/8/2013 |
|
10.22 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.49 |
|
|
10-K |
|
2/26/2016 |
|
10.34 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.50 |
|
|
10-K |
|
2/26/2016 |
|
10.35 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.51 |
|
|
8-K |
|
2/25/2014 |
|
10.1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.52 |
|
|
8-K |
|
3/13/2015 |
|
10.1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.53 |
|
|
10-K |
|
2/26/2016 |
|
10.38 |
|
|
|
|
10.54 |
|
|
|
S-1 |
|
11/8/2013 |
|
10.23 |
|
|
|
10.55 |
|
|
|
S-1 |
|
11/8/2013 |
|
10.24 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.56 |
|
|
10-Q |
|
11/10/2014 |
|
10.1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
83
|
Exhibit |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed |
||||
|
|
|
Form |
|
Date |
|
Number |
|
Herewith |
||
|
|
|
10-Q |
|
11/10/2015 |
|
10.3 |
|
|
||
|
10.58 |
|
|
10-K |
|
2/17/2017 |
|
10.51 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.59 |
|
|
10-K |
|
2/26/2016 |
|
10.43 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.60 |
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.61 |
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.62 |
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.63 |
|
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.64 |
|
|
|
|
|
|
|
|
X |
|
|
10.65 |
|
|
|
|
|
|
|
|
X |
|
|
10.66 |
|
|
|
|
|
|
|
|
X |
|
|
21.1 |
|
|
|
|
|
|
|
|
|
X |
|
23.1 |
|
|
|
|
|
|
|
|
|
X |
|
24.1 |
|
Power of Attorney (included on the signature page of this report) |
|
|
|
|
|
|
|
|
|
31.1 |
|
|
|
|
|
|
|
|
|
X |
84
|
Exhibit |
|
Exhibit Description |
|
Incorporated by Reference |
|
Filed |
||||
|
|
|
Form |
|
Date |
|
Number |
|
Herewith |
||
|
31.2 |
|
|
|
|
|
|
|
|
|
X |
|
32.1§ |
|
|
|
|
|
|
|
|
|
X |
|
101.INS |
|
XBRL Instance Document |
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
101.SCH |
|
XBRL Taxonomy Extension Schema Document |
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
101.CAL |
|
XBRL Taxonomy Extension Calculation Linkbase Document |
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
101.DEF |
|
XBRL Taxonomy Extension Definition Linkbase Document |
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
101.LAB |
|
XBRL Taxonomy Extension Labels Linkbase Document |
|
|
|
|
|
|
|
X |
|
|
|
|
|
|
|
|
|
|
|
|
|
101.PRE |
|
XBRL Taxonomy Extension Presentation Linkbase Document |
|
|
|
|
|
|
|
X |
|
|
|
† |
Confidential treatment has been granted with respect to certain portions (indicated by asterisks) of this exhibit. Omitted portions have been filed separately with the SEC. |
|
* |
Portions of this exhibit (indicated by asterisks) have been omitted pursuant to a request for confidential treatment. Omitted portions have been filed separately with the SEC |
|
# |
Indicates management contract or compensatory plan. |
|
§ |
The certification attached as Exhibit 32.1 that accompanies this Annual Report is not deemed filed with the SEC and is not to be incorporated by reference into any filing of Ultragenyx Pharmaceutical Inc. under the Securities Act or the Exchange Act, whether made before or after the date of this Annual Report, irrespective of any general incorporation language contained in such filing. |
None.
85
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
ULTRAGENYX PHARMACEUTICAL Inc. |
||
|
By: |
|
/s/ Emil D. Kakkis |
|
|
|
Emil D. Kakkis, M.D., Ph.D. |
|
|
|
President and Chief Executive Officer |
Date: February 21, 2018
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Emil D. Kakkis, M.D., Ph.D. and Shalini Sharp, and each of them, as his or her true and lawful attorneys-in-fact and agents, with full power of substitution for him or her, and in his or her name in any and all capacities, to sign any and all amendments to this Annual Report, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done therewith, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, and any of them, his or her substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
|
|
Signature |
|
|
|
Title |
|
|
|
Date |
|
|
/s/ Emil D. Kakkis
Emil D. Kakkis, M.D., Ph.D. |
|
|
|
|
|
|
|
|
||
|
|
President and Chief Executive Officer and Director (Principal Executive Officer) |
|
February 21, 2018 |
|||||||
|
/s/ Shalini Sharp
Shalini Sharp |
|
Executive Vice President and Chief Financial Officer (Principal Financial Officer) |
|
February 21, 2018 |
||||||
|
/s/ Theodore A. Huizenga
Theodore A. Huizenga |
|
Vice President, Corporate Controller (Principal Accounting Officer) |
|
February 21, 2018 |
||||||
|
/s/ Daniel G. Welch
Daniel G. Welch. |
|
Chairman of the Board |
|
February 21, 2018 |
||||||
|
/s/ William Aliski
William Aliski |
|
Director |
|
February 21, 2018 |
||||||
|
/s/ Deborah Dunsire
Deborah Dunsire, M.D. |
|
Director |
|
February 21, 2018 |
||||||
|
/s/ Lars Ekman
Lars Ekman, M.D., Ph.D. |
|
Director |
|
February 21, 2018 |
||||||
|
/s/ Matthew K. Fust
Matthew K. Fust |
|
Director |
|
February 21, 2018 |
||||||
|
/s/ Michael Narachi
Michael Narachi |
|
Director |
|
February 21, 2018 |
||||||
|
/s/ Clay B. Siegall
Clay B. Siegall, Ph.D. |
|
Director |
|
February 21, 2018 |
||||||
86
Ultragenyx Pharmaceutical Inc.
INDEX TO FINANCIAL STATEMENTS
|
|
Page |
|
F-2 |
|
|
Consolidated Financial Statements: |
|
|
F-3 |
|
|
F-4 |
|
|
F-5 |
|
|
F-6 |
|
|
F-7 |
|
|
F-8 |
F-1
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of Ultragenyx Pharmaceutical Inc.:
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Ultragenyx Pharmaceutical Inc. (the “Company”) as of December 31, 2017 and 2016, the related consolidated statements of operations, comprehensive loss, stockholders’ equity, and cash flows, for each of the three years in the period ended December 31, 2017, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2017 and 2016, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2017, in conformity with US generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company’s internal control over financial reporting as of December 31, 2017, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 Framework) and our report dated February 21, 2018 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the US federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures include examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2010.
San Jose, California
February 21, 2018
F-2
ULTRAGENYX PHARMACEUTICAL INC.
(In thousands, except share and per share amounts)
|
|
|
December 31, |
|
|||||
|
|
|
2017 |
|
|
2016 |
|
||
|
Assets |
|
|
|
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
$ |
100,488 |
|
|
$ |
161,120 |
|
|
Short-term investments |
|
|
134,005 |
|
|
|
219,028 |
|
|
Accounts receivable |
|
|
5,172 |
|
|
|
— |
|
|
Inventory |
|
|
757 |
|
|
|
— |
|
|
Restricted cash |
|
|
461 |
|
|
|
1,411 |
|
|
Prepaid expenses and other current assets |
|
|
28,700 |
|
|
|
20,136 |
|
|
Total current assets |
|
|
269,583 |
|
|
|
401,695 |
|
|
Property and equipment, net |
|
|
21,837 |
|
|
|
17,055 |
|
|
Restricted cash |
|
|
2,092 |
|
|
|
2,076 |
|
|
Long-term investments |
|
|
9,975 |
|
|
|
117,963 |
|
|
Intangible assets, net |
|
|
141,545 |
|
|
|
— |
|
|
Goodwill |
|
|
44,406 |
|
|
|
— |
|
|
Other assets |
|
|
1,315 |
|
|
|
1,837 |
|
|
Total assets |
|
$ |
490,753 |
|
|
$ |
540,626 |
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Stockholders’ Equity |
|
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
|
Accounts payable |
|
$ |
8,886 |
|
|
$ |
5,364 |
|
|
Accrued liabilities |
|
|
61,427 |
|
|
|
54,554 |
|
|
Deferred rent—current portion |
|
|
701 |
|
|
|
341 |
|
|
Total current liabilities |
|
|
71,014 |
|
|
|
60,259 |
|
|
|
|
|
|
|
|
|
|
|
|
Deferred tax liabilities |
|
|
31,166 |
|
|
|
— |
|
|
Other liabilities |
|
|
5,119 |
|
|
|
6,393 |
|
|
Total liabilities |
|
|
107,299 |
|
|
|
66,652 |
|
|
|
|
|
|
|
|
|
|
|
|
Commitments and contingencies (Note 12) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stockholders’ equity: |
|
|
|
|
|
|
|
|
|
Preferred stock, par value of $0.001 per share—25,000,000 shares authorized; nil outstanding as of December 31, 2017 and 2016 |
|
|
— |
|
|
|
— |
|
|
Common stock, par value of $0.001 per share—250,000,000 shares authorized; 44,167,071 and 41,240,230 shares issued and outstanding as of December 31, 2017 and 2016, respectively |
|
|
44 |
|
|
|
41 |
|
|
Additional paid-in capital |
|
|
1,221,762 |
|
|
|
1,003,561 |
|
|
Accumulated other comprehensive income (loss) |
|
|
(5,680 |
) |
|
|
905 |
|
|
Accumulated deficit |
|
|
(832,672 |
) |
|
|
(530,533 |
) |
|
Total stockholders’ equity |
|
|
383,454 |
|
|
|
473,974 |
|
|
Total liabilities and stockholders’ equity |
|
$ |
490,753 |
|
|
$ |
540,626 |
|
See accompanying notes.
F-3
ULTRAGENYX PHARMACEUTICAL INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(In thousands, except share and per share amounts)
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Revenues: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Collaboration and license |
|
$ |
2,136 |
|
|
$ |
— |
|
|
$ |
— |
|
|
Product sales |
|
|
476 |
|
|
|
133 |
|
|
|
— |
|
|
Total revenues |
|
|
2,612 |
|
|
|
133 |
|
|
|
— |
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of sales |
|
|
1 |
|
|
|
— |
|
|
|
— |
|
|
Research and development |
|
|
231,644 |
|
|
|
183,204 |
|
|
|
114,737 |
|
|
Selling, general and administrative |
|
|
99,909 |
|
|
|
64,936 |
|
|
|
33,001 |
|
|
Total operating expenses |
|
|
331,554 |
|
|
|
248,140 |
|
|
|
147,738 |
|
|
Loss from operations |
|
|
(328,942 |
) |
|
|
(248,007 |
) |
|
|
(147,738 |
) |
|
Interest income |
|
|
4,074 |
|
|
|
3,789 |
|
|
|
2,320 |
|
|
Other income (expense) |
|
|
6,530 |
|
|
|
(1,621 |
) |
|
|
(200 |
) |
|
Loss before income taxes |
|
|
(318,338 |
) |
|
|
(245,839 |
) |
|
|
(145,618 |
) |
|
Benefit from (provision for) income taxes |
|
|
16,199 |
|
|
|
(35 |
) |
|
|
— |
|
|
Net loss |
|
$ |
(302,139 |
) |
|
$ |
(245,874 |
) |
|
$ |
(145,618 |
) |
|
Net loss per share, basic and diluted |
|
$ |
(7.12 |
) |
|
$ |
(6.21 |
) |
|
$ |
(3.96 |
) |
|
Shares used in computing net loss per share, basic and diluted |
|
|
42,453,135 |
|
|
|
39,586,908 |
|
|
|
36,782,603 |
|
See accompanying notes.
F-4
ULTRAGENYX PHARMACEUTICAL INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(In thousands)
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Net loss |
|
$ |
(302,139 |
) |
|
$ |
(245,874 |
) |
|
$ |
(145,618 |
) |
|
Other comprehensive income (loss): |
|
|
|
|
|
|
|
|
|
|
|
|
|
Foreign currency translation adjustments |
|
|
(10,110 |
) |
|
|
1,322 |
|
|
|
— |
|
|
Transfer of currency translation adjustments balance to other income for the |
|
|
|
|
|
|
|
|
|
|
|
|
|
liquidation of a foreign entity |
|
|
3,490 |
|
|
|
— |
|
|
|
— |
|
|
Unrealized gain (loss) on available-for-sale securities |
|
|
35 |
|
|
|
451 |
|
|
|
(694 |
) |
|
Other comprehensive income (loss): |
|
|
(6,585 |
) |
|
|
1,773 |
|
|
|
(694 |
) |
|
Total comprehensive loss |
|
$ |
(308,724 |
) |
|
$ |
(244,101 |
) |
|
$ |
(146,312 |
) |
See accompanying notes.
F-5
ULTRAGENYX PHARMACEUTICAL INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(In thousands, except share amounts)
|
|
|
Common Stock |
|
|
Additional Paid-In |
|
|
Accumulated Other Comprehensive |
|
|
Accumulated |
|
|
Total Stockholders' |
|
|||||||||
|
|
|
Shares |
|
|
Amount |
|
|
Capital |
|
|
Income (Loss) |
|
|
Deficit |
|
|
Equity |
|
||||||
|
Balance as of December 31, 2014 |
|
|
31,934,682 |
|
|
$ |
32 |
|
|
$ |
324,128 |
|
|
$ |
(174 |
) |
|
$ |
(139,041 |
) |
|
$ |
184,945 |
|
|
Issuance of common stock in connection with underwritten public offering, net of issuance costs |
|
|
5,980,000 |
|
|
|
6 |
|
|
|
461,130 |
|
|
|
— |
|
|
|
— |
|
|
|
461,136 |
|
|
Employee stock-based compensation |
|
|
— |
|
|
|
— |
|
|
|
24,884 |
|
|
|
— |
|
|
|
— |
|
|
|
24,884 |
|
|
Issuance of common stock under equity plan awards, net of tax |
|
|
967,712 |
|
|
|
1 |
|
|
|
6,436 |
|
|
|
— |
|
|
|
— |
|
|
|
6,437 |
|
|
Other comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(694 |
) |
|
|
— |
|
|
|
(694 |
) |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(145,618 |
) |
|
|
(145,618 |
) |
|
Balance as of December 31, 2015 |
|
|
38,882,394 |
|
|
|
39 |
|
|
|
816,578 |
|
|
|
(868 |
) |
|
|
(284,659 |
) |
|
|
531,090 |
|
|
Issuance of common stock in connection with at-the-market offering, net of issuance costs |
|
|
1,159,415 |
|
|
|
1 |
|
|
|
79,485 |
|
|
|
— |
|
|
|
— |
|
|
|
79,486 |
|
|
Issuance of common stock in connection with collaboration agreement, net of issuance costs |
|
|
727,120 |
|
|
|
1 |
|
|
|
52,271 |
|
|
|
— |
|
|
|
— |
|
|
|
52,272 |
|
|
Put option grant in connection with collaboration agreement |
|
|
— |
|
|
|
— |
|
|
|
(916 |
) |
|
|
— |
|
|
|
— |
|
|
|
(916 |
) |
|
Employee stock-based compensation |
|
|
— |
|
|
|
— |
|
|
|
48,309 |
|
|
|
— |
|
|
|
— |
|
|
|
48,309 |
|
|
Issuance of common stock under equity plan awards, net of tax |
|
|
471,301 |
|
|
|
— |
|
|
|
7,834 |
|
|
|
— |
|
|
|
— |
|
|
|
7,834 |
|
|
Other comprehensive income |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,773 |
|
|
|
— |
|
|
|
1,773 |
|
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(245,874 |
) |
|
|
(245,874 |
) |
|
Balance as of December 31, 2016 |
|
|
41,240,230 |
|
|
|
41 |
|
|
|
1,003,561 |
|
|
|
905 |
|
|
|
(530,533 |
) |
|
|
473,974 |
|
|
Issuance of common stock in connection with at-the-market offering, net of issuance costs |
|
|
2,251,217 |
|
|
|
2 |
|
|
|
131,958 |
|
|
|
— |
|
|
|
— |
|
|
|
131,960 |
|
|
Fair value of vested stock options assumed from acquisition |
|
|
— |
|
|
|
— |
|
|
|
8,979 |
|
|
|
— |
|
|
|
— |
|
|
|
8,979 |
|
|
Employee stock-based compensation |
|
|
— |
|
|
|
— |
|
|
|
68,014 |
|
|
|
— |
|
|
|
— |
|
|
|
68,014 |
|
|
Issuance of common stock under equity plan awards, net of tax |
|
|
675,624 |
|
|
|
1 |
|
|
|
9,250 |
|
|
|
— |
|
|
|
— |
|
|
|
9,251 |
|
|
Other comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(6,585 |
) |
|
|
— |
|
|
|
(6,585 |
) |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(302,139 |
) |
|
|
(302,139 |
) |
|
Balance as of December 31, 2017 |
|
|
44,167,071 |
|
|
$ |
44 |
|
|
$ |
1,221,762 |
|
|
$ |
(5,680 |
) |
|
$ |
(832,672 |
) |
|
$ |
383,454 |
|
See accompanying notes.
F-6
ULTRAGENYX PHARMACEUTICAL INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands)
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Operating activities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
$ |
(302,139 |
) |
|
$ |
(245,874 |
) |
|
$ |
(145,618 |
) |
|
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation |
|
|
68,014 |
|
|
|
48,309 |
|
|
|
24,884 |
|
|
Amortization of premium on investment securities, net |
|
|
1,706 |
|
|
|
4,842 |
|
|
|
5,637 |
|
|
Depreciation and amortization |
|
|
5,825 |
|
|
|
3,424 |
|
|
|
1,384 |
|
|
Foreign currency remeasurement (gain) loss |
|
|
(7,018 |
) |
|
|
1,322 |
|
|
|
— |
|
|
Non-cash license fee from collaboration arrangement |
|
|
— |
|
|
|
700 |
|
|
|
— |
|
|
Changes in operating assets and liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Prepaid expenses and other current assets |
|
|
(3,338 |
) |
|
|
(7,147 |
) |
|
|
(7,131 |
) |
|
Other assets |
|
|
522 |
|
|
|
(1,242 |
) |
|
|
179 |
|
|
Accounts payable |
|
|
3,459 |
|
|
|
2,502 |
|
|
|
(1,982 |
) |
|
Accrued liabilities and other liabilities |
|
|
(4,628 |
) |
|
|
32,189 |
|
|
|
16,670 |
|
|
Deferred tax liabilities |
|
|
(16,246 |
) |
|
|
— |
|
|
|
— |
|
|
Net cash used in operating activities |
|
|
(253,843 |
) |
|
|
(160,975 |
) |
|
|
(105,977 |
) |
|
Investing activities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition, net of cash acquired |
|
|
(142,804 |
) |
|
|
— |
|
|
|
— |
|
|
Purchase of property and equipment |
|
|
(2,793 |
) |
|
|
(10,188 |
) |
|
|
(4,955 |
) |
|
Purchase of investments |
|
|
(230,487 |
) |
|
|
(442,490 |
) |
|
|
(624,226 |
) |
|
Proceeds from sale of investments |
|
|
157,934 |
|
|
|
140,556 |
|
|
|
89,321 |
|
|
Proceeds from maturities of investments |
|
|
273,632 |
|
|
|
403,239 |
|
|
|
249,050 |
|
|
Decrease (increase) in restricted cash |
|
|
934 |
|
|
|
(1,202 |
) |
|
|
(1,541 |
) |
|
Net cash provided by (used in) investing activities |
|
|
56,416 |
|
|
|
89,915 |
|
|
|
(292,351 |
) |
|
Financing activities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from the issuance of common stock in connection with underwritten public offerings, net |
|
|
— |
|
|
|
— |
|
|
|
461,136 |
|
|
Proceeds from the issuance of common stock in connection with at-the-market offering, net |
|
|
131,960 |
|
|
|
79,486 |
|
|
|
— |
|
|
Proceeds from the issuance of common stock in connection with collaboration agreement, net |
|
|
— |
|
|
|
51,356 |
|
|
|
— |
|
|
Proceeds from the issuance of common stock under equity plan awards, net of tax |
|
|
9,251 |
|
|
|
7,834 |
|
|
|
6,437 |
|
|
Repayment of note payable |
|
|
(4,944 |
) |
|
|
— |
|
|
|
— |
|
|
Net cash provided by financing activities |
|
|
136,267 |
|
|
|
138,676 |
|
|
|
467,573 |
|
|
Effect of exchange rate changes on cash |
|
|
528 |
|
|
|
(65 |
) |
|
|
— |
|
|
Net increase (decrease) in cash and cash equivalents |
|
|
(60,632 |
) |
|
|
67,551 |
|
|
|
69,245 |
|
|
Cash and cash equivalents at beginning of year |
|
|
161,120 |
|
|
|
93,569 |
|
|
|
24,324 |
|
|
Cash and cash equivalents at end of year |
|
$ |
100,488 |
|
|
$ |
161,120 |
|
|
$ |
93,569 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental disclosures of non-cash investing and financing information: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Fair value of vested stock options assumed in acquisition |
|
$ |
8,979 |
|
|
$ |
— |
|
|
$ |
— |
|
|
Costs of property and equipment included in accounts payable and accrued liabilities |
|
$ |
400 |
|
|
$ |
147 |
|
|
$ |
769 |
|
|
Tenant improvement allowance |
|
$ |
— |
|
|
$ |
3,467 |
|
|
$ |
— |
|
See accompanying notes.
F-7
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements
1. Organization and Basis of Presentation
Ultragenyx Pharmaceutical Inc. (the Company) is a biopharmaceutical company and was incorporated in California on April 22, 2010. The Company subsequently reincorporated in the state of Delaware in June 2011.
The Company is focused on the identification, acquisition, development, and commercialization of novel products for the treatment of rare and ultra-rare diseases, with a focus on serious, debilitating genetic diseases. On November 15, 2017, the U.S. Food and Drug Administration (FDA), approved the Company’s first product, Mepsevii (vestronidase alfa), the first medicine approved for the treatment of children and adults with MPS VII, also known as Sly syndrome. Mepsevii is an intravenous enzyme replacement therapy for the treatment of MPS VII, a rare lysosomal storage disease that often leads to multi-organ dysfunction, pervasive skeletal disease, and death.
The Company is conducting Phase 2 and Phase 3 studies of burosumab (KRN23 or UX023), an antibody targeting fibroblast growth factor 23 (FGF23), in pediatric and adult patients with X-linked hypophosphatemia (XLH) and a Phase 2 study in tumor induced osteomalacia (TIO), both rare diseases that impair bone mineralization; a Phase 3 study for UX007 in patients with glucose transporter type-1 deficiency syndrome (Glut1 DS), a brain energy deficiency, who are experiencing movement disorders; a Phase 2 clinical study of UX007 in patients severely affected by long-chain fatty acid oxidation disorders (LC-FAOD), a genetic disorder in which the body is unable to convert long chain fatty acids into energy and a Phase 1/2 open label clinical study of DTX 301, an adeno-associated virus (AAV8) gene therapy product candidate designed for the treatment of patients with ornithine transcarbamylase (OTC) deficiency, the most common urea cycle disorder. The Company operates as one reportable segment.
In the course of its research activities, the Company has sustained operating losses and expects such annual losses to continue over the next several years. The Company’s ultimate success depends on the outcome of its research and development activities. Management expects to incur additional losses in the future to conduct product research and development and recognizes the need to raise additional capital to fully implement its business plan. Through December 31, 2017, the Company has relied primarily on the proceeds from equity offerings to finance its operations.
The Company intends to raise additional capital through the issuance of equity, borrowings, or strategic alliances with partner companies. However, if such financing is not available at adequate levels, the Company will need to reevaluate its operating plans.
2. Summary of Significant Accounting Policies
Basis of Consolidation
The consolidated financial statements include the accounts of Ultragenyx Pharmaceutical Inc. and our wholly-owned subsidiaries. All intercompany balances and transactions have been eliminated.
Use of Estimates
The accompanying consolidated financial statements have been prepared in accordance with U.S. generally accepted accounting principles (“GAAP”). The preparation of the consolidated financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, disclosure of contingent liabilities and the reported amounts of expenses in the consolidated financial statements and the accompanying notes. On an ongoing basis, management evaluates its estimates, including those related to clinical trial accruals, fair value of assets and liabilities, income taxes, and stock-based compensation. Management bases its estimates on historical experience and on various other market-specific and relevant assumptions that management believes to be reasonable under the circumstances. Actual results could differ from those estimates.
Cash and Cash Equivalents
The Company considers all highly liquid investments with original maturities of three months or less from the date of purchase to be cash equivalents. Cash equivalents consist primarily of amounts invested in money market accounts.
Investments
All investments have been classified as “available-for-sale” and are carried at estimated fair value as determined based upon quoted market prices or pricing models for similar securities. Management determines the appropriate classification of its investments at the time of purchase and reevaluates such designation as of each balance sheet date. Investments with a maturity of one year or less from the balance sheet date are reported as short-term investments and investments with a maturity of greater than one year from the balance sheet date are reported as long-term investments. Unrealized gains and losses are excluded from earnings and are reported as a component of comprehensive loss. Realized gains and losses and declines in fair value judged to be other than temporary, if any, on available-for-sale securities are included in other income (expense). The cost of securities sold is based on the specific-identification method. Interest on investments is included in interest income.
F-8
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
Concentration of Credit Risk and Other Risks and Uncertainties
Financial instruments that potentially subject the Company to a concentration of credit risk consist of cash, cash equivalents, and investments. The Company’s cash, cash equivalents, and investments are held by financial institutions that management believes are of high credit quality. The Company’s investment policy limits investments to fixed income securities denominated and payable in U.S. dollars such as U.S. government obligations, money market instruments and funds, corporate bonds, and asset-backed securities and places restrictions on maturities and concentrations by type and issuer. Such deposits may, at times, exceed federally insured limits. The Company has not experienced any losses on its deposits of cash and cash equivalents and its accounts are monitored by management to mitigate risk. The Company is exposed to credit risk in the event of default by the financial institutions holding its cash and cash equivalents, corporate bond issuers and other financial instruments to the extent recorded in the balance sheets.
Concentrations on credit risk with respect to accounts receivable are limited due to the Company’s limited number of customers.
The Company is dependent on third-party manufacturers to supply products for research and development activities in its programs. In particular, the Company relies and expects to continue to rely on a small number of manufacturers to supply it with its requirements for the active pharmaceutical ingredients and formulated drugs related to these programs. These programs could be adversely affected by a significant interruption in the supply of active pharmaceutical ingredients and formulated drugs.
Inventory
The Company values inventory at the lower of cost and net realizable value and determines the cost of inventory using the average-cost method. Inventories consist of currently approved product.
The Company periodically reviews its inventories for excess amounts or obsolescence and writes down obsolete or otherwise unmarketable inventory to its estimated net realizable value.
Restricted Cash
Restricted cash primarily consists of money market accounts as collateral for its obligations under its facility leases of the Company’s corporate headquarters in Novato, California and for its facilities in Brisbane, California and Woburn and Cambridge, Massachusetts.
Property and Equipment
Property and equipment are stated at cost, less accumulated depreciation and amortization. Depreciation and amortization is computed using the straight-line method over the estimated useful lives of the respective assets. Depreciation and amortization begins at the time the asset is placed in service. Maintenance and repairs are charged to operations as incurred. Upon sale or retirement of assets, the cost and related accumulated depreciation are removed from the balance sheet and the resulting gain or loss, if any, is reflected in operations.
The useful lives of the property and equipment are as follows:
|
Research and development equipment |
|
5 years |
|
Furniture and office equipment |
|
5 years |
|
Computer equipment |
|
3 years |
|
Software |
|
3-5 years |
|
Leasehold improvements |
|
Shorter of lease term or estimated useful life |
Intangible Assets
The Company recognizes an acquired intangible apart from goodwill whenever the intangible arises from contractual or other legal rights, or whenever it can be separated or divided from the acquired entity and sold, transferred, licensed, rented or exchanged, either individually or in combination with a related contract, asset or liability. The Company’s intangible assets consist of acquired in-process research and development (IPR&D) and an acquired contract asset.
IPR&D assets represent capitalized incomplete research projects that the Company acquired through business combinations. Such assets are initially measured at their acquisition date fair values and are tested for impairment, until the completion or abandonment of the associated research and development efforts. If and when development is complete, which generally occurs when regulatory approval to market a product is obtained, the associated assets will be deemed finite-lived and will be amortized over a period that best reflects the economic benefits provided by these assets. The acquired contract asset was initially recorded at fair value and is amortized over estimated useful live.
F-9
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The Company tests its definite and indefinite-lived intangible assets for impairment annually during the fourth quarter and more frequently if events or changes in circumstances indicate that it is more likely than not that the asset is impaired. The Company also reviews the useful lives of its assets periodically to determine whether events and circumstances warrant a revision to the remaining useful life. Changes in the useful life are adjusted prospectively by revising the remaining period over which the asset is amortized. The Company has not recorded any impairments nor made any adjustments to the useful lives of its assets since inception.
Goodwill
Goodwill represents the excess of purchase price over fair value of net assets acquired in a business combination and is not amortized. Goodwill is subject to impairment testing at least annually during the fourth quarter or when a triggering event occurs that could indicate a potential impairment. The Company has not recorded any impairments since inception.
Impairment of Long-Lived Assets
The Company evaluates its long-lived assets, including property and equipment, for impairment whenever events or changes in circumstances indicate that the carrying value of these assets may not be recoverable. Recoverability of these assets is measured by comparison of the carrying amount of each asset to the future undiscounted cash flows expected to result from the use of the asset and its eventual disposition. If the asset is considered to be impaired, the amount of any impairment is measured as the difference between the carrying value and the fair value of the impaired asset. The Company has not recorded impairment of any long-lived assets since inception.
Accruals of Research and Development Costs
The Company records accruals for estimated costs of research, preclinical and clinical studies and manufacturing development. These costs are a significant component of the Company’s research and development expenses. A substantial portion of the Company’s ongoing research and development activities are conducted by third-party service providers, including contract research organizations. The Company accrues the costs incurred under its agreements with these third parties based on actual work completed in accordance with agreements established with these third parties. The Company determines the actual costs through discussions with internal personnel and external service providers as to the progress or stage of completion of the services and the agreed-upon fee to be paid for such services.
Revenue Recognition
In May 2014, the Financial Accounting Standards Board (FASB), issued Accounting Standards Update (ASU) 2014-09, Revenue from Contracts with Customers (ASC 606), to supersede nearly all existing revenue recognition guidance under GAAP. The core principle of ASU 2014-09 is to recognize revenues when promised goods or services are transferred to customers in an amount that reflects the consideration that is expected to be received for those goods or services. ASU 2014-09 defines a five-step process to achieve this core principle and, in doing so, it is possible more judgment and estimates may be required within the revenue recognition process than are required under existing GAAP, including identifying performance obligations in a contract, estimating the amount of variable consideration to include in the transaction price and allocating the transaction price to each separate performance obligation. The Company has early adopted the new revenue standard as of January 1, 2017 using a full retrospective application to each prior reporting period presented. Through January 1, 2017, the Company had recorded a cumulative inception to date total of $0.1 million of revenues. The adoption did not have an effect on the Consolidated Financial Statements on the adoption date and no adjustment to prior year consolidated financial statements was required.
Product sales
The Company sells Mepsevii through a limited number of distributors. Under ASC 606, revenue from product sales is recognized at the point in time when the delivery is made and when title and risk of loss transfers to these distributors. The Company also recognizes revenue from sales of Mepsevii on a “named patient” basis, which are allowed in certain countries prior to the commercial approval of the product in the territory. Prior to recognizing revenue, the Company makes estimates of the transaction price, including variable consideration that is subject to a constraint. Amounts of variable consideration are included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. Product sales are recorded net of estimated government-mandated rebates and chargebacks, distribution fees, estimated product returns and other deductions.
F-10
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
Collaboration and license revenue
The Company has certain license and collaboration agreements that are within the scope of ASC 808, Collaborative Agreements, which provides guidance on the presentation and disclosure of collaborative arrangements. Funding received related to research and development services and pre-commercialization costs are classified as a reduction of research and development expenses and selling, general and administrative expenses, respectively in the consolidated statement of operations because the provision of such services for collaborative partners are not considered to be part of the Company’s ongoing major or central operations.
The Company also receives royalty revenues under certain of the Company’s license or collaboration agreements in exchange for license of intellectual property. If the Company does not have any future performance obligations under these license or collaborations agreements, royalty revenue is recorded as the underlying sales occur.
The Company also recognizes collaboration and license revenue under certain of the Company’s license or collaboration agreements that are within the scope of ASC 606. The terms of these agreements may contain multiple performance obligations, which may include licenses and research and development activities. The Company evaluates these agreements under ASC 606 to determine the distinct performance obligations.
Prior to recognizing revenue, the Company makes estimates of the transaction price, including variable consideration that is subject to a constraint. Amounts of variable consideration are included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur and when the uncertainty associated with the variable consideration is subsequently resolved. Variable consideration may include nonrefundable upfront license fees, payments for research and development activities, reimbursement of certain third-party costs, payments based upon the achievement of specified milestones, and royalty payments based on product sales derived from the collaboration.
If there are multiple distinct performance obligations, the Company allocates the transaction price to each distinct performance obligation based on its relative standalone selling price. The standalone selling price is generally determined based on the prices charged to customers or using expected cost plus margin. Revenue is recognized by measuring the progress toward complete satisfaction of the performance obligations using an input measure.
Leases
The Company enters into lease agreements for its office and laboratory facilities. These leases are classified as operating leases. Rent expense is recognized on a straight-line basis over the term of the lease and, accordingly, the Company records the difference between cash rent payments and the recognition of rent expense as a deferred rent liability. Incentives granted under the Company’s facilities leases, including allowances to fund leasehold improvements, are deferred and are recognized as adjustments to rental expense on a straight-line basis over the term of the lease.
Comprehensive Loss
Comprehensive loss is the change in stockholders’ equity from transactions and other events and circumstances other than those resulting from investments by stockholders and distributions to stockholders. The Company’s other comprehensive loss is comprised of unrealized gains and losses on investments in available-for-sale securities and foreign currency translation adjustments.
Research and Development
Research and development costs are expensed as incurred and consist of salaries and benefits, stock-based compensation expense, lab supplies and facility costs, as well as fees paid to other nonemployees and entities that conduct certain research and development activities on the Company’s behalf. Amounts incurred in connection with license agreements are also included in research and development expense. Nonrefundable advance payments for goods or services to be received in the future for use in research and development activities are deferred and capitalized. The capitalized amounts are expensed as the related goods are delivered or the services are performed.
Stock-Based Compensation
Stock-based awards issued to employees, including stock options, restricted stock units (RSUs), and performance stock units (PSUs) are recorded at fair value as of the grant date and recognized as expense on a straight-line basis over the employee’s requisite service period (generally the vesting period). PSUs vest only if certain specified criteria are achieved and the employees’ continued service requirements are met; therefore, the expense recognition occurs when the likelihood of the PSUs being earned is deemed probable. Noncash stock compensation expense on awards expected to vest are recognized net of estimated forfeitures.
F-11
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
In March 2016, the FASB issued ASU 2016-09, Compensation — Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting, which simplifies several aspects of the accounting for employee share-based payments, including income tax consequences, application of award forfeitures to expense, classification on the statement of cash flows, and classification of awards as either equity or liabilities. The Company adopted ASU 2016-09 as of January 1, 2017. On January 1, 2017, there was $19.7 million of cumulative unrecognized excess tax benefits which was fully offset by a corresponding increase in the valuation allowance. The adoption did not have any other impact on the Consolidated Financial Statements on the adoption date.
Income Taxes
The Company uses the liability method of accounting for income taxes. Under this method, deferred tax assets and liabilities are determined based on the differences between the financial reporting and the tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. The Company must then assess the likelihood that the resulting deferred tax assets will be realized. A valuation allowance is provided when it is more likely than not that some portion or all of a deferred tax asset will not be realized. Due to the Company’s lack of earnings history, the net deferred tax assets have been fully offset by a valuation allowance.
In conjunction with Dimension acquisition, a deferred tax liability was recorded reflecting the tax impact of the difference between the book basis and tax basis of acquired IPR&D. Such deferred income tax liability is not used to offset deferred tax assets when analyzing the Company’s valuation allowance as the acquired IPR&D is considered to have an indefinite life until the Company completes or abandons development of the acquired IPR&D.
The Company recognizes benefits of uncertain tax positions if it is more likely than not that such positions will be sustained upon examination based solely on their technical merits, as the largest amount of benefit that is more likely than not to be realized upon the ultimate settlement. The Company’s policy is to recognize interest and penalties related to the underpayment of income taxes as a component of income tax expense or benefit. To date, there have been no interest or penalties charged in relation to the unrecognized tax benefits.
Foreign Currency
Assets and liabilities of non-U.S. subsidiaries that operate in a local currency environment, where the local currency is the functional currency, are translated to U.S. dollars at exchange rates in effect at the balance sheet date, with the resulting translation adjustments directly recorded to a separate component of accumulated other comprehensive loss. Income and expense accounts are translated at average exchange rates for the period. Transactions which are not in the functional currency of the entity are remeasured into the functional currency and gains or losses resulting from the remeasurement recorded in other income (expense).
Net Loss per Share
Basic net loss per share is calculated by dividing the net loss by the weighted-average number of shares of common stock outstanding during the period, without consideration for common stock equivalents. Diluted net loss per share is the same as basic net loss per share, since the effects of potentially dilutive securities are antidilutive. In periods when we have incurred a net loss, options and warrants to purchase common stock are considered common stock equivalents, but have been excluded from the calculation of diluted net loss per share, as their effect is antidilutive.
Business Combinations
The Company applies the provisions of ASC Topic 805, “Business Combinations” (Topic 805)”, in the accounting for acquisitions. The Company allocates the purchase price of acquired businesses to the tangible and intangible assets acquired and liabilities assumed based upon their estimated fair values on the acquisition date. The purchase price allocation process requires management to make significant estimates and assumptions, especially at the acquisition date with respect to intangible assets which includes IPR&D.
Recent Accounting Pronouncements
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842), which requires an entity that is a lessee to record a right of use asset and a corresponding lease liability on the balance sheet for all leases with terms longer than 12 months. This guidance also requires disclosures about the amount, timing, and uncertainty of cash flows arising from leases. This guidance is effective for annual reporting periods beginning after December 15, 2018, and interim periods within those annual periods, using a modified retrospective approach, and early adoption is permitted. The Company is evaluating the effect that this guidance will have on its Consolidated Financial Statements and related disclosures.
In October 2016, the FASB issued ASU 2016-16, Income Taxes - Intra-Entity Transfers of Assets Other Than Inventory, which requires entities to recognize the income tax consequences of an intra-entity transfer of an asset other than inventory when the transfer occurs. The standard is effective for fiscal years beginning after December 15, 2017, including interim periods within those fiscal
F-12
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
years. Early adoption is permitted as of the beginning of a fiscal year. The new standard must be adopted using a modified retrospective transition method which is a cumulative-effective adjustment to retained earnings as of the beginning of the first effective reporting period. The Company is evaluating the effect that this guidance will have on its Consolidated Financial Statements and related disclosures; however, the impact is expected to be immaterial.
In November 2016, the FASB issued ASU 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash, which requires restricted cash to be presented with cash and cash equivalents on the statement of cash flows and disclosure of how the statement of cash flows reconciles to the balance sheet if restricted cash is shown separately from cash and cash equivalents on the balance sheet. The guidance is effective for interim and annual periods beginning after December 15, 2017, and early adoption is permitted. The Company is evaluating the effect that this guidance will have on our statement of cash flows and related disclosures; however the impact is expected to be immaterial.
In January 2017, the FASB issued ASU 2017-04, Intangibles – Goodwill and Other (Topic 350): Simplifying the Test for Goodwill Impairment, which will eliminate the requirement to calculate the implied fair value of goodwill, commonly referred to as “Step 2” in the current goodwill impairment test. An entity will still have the option to perform the qualitative assessment for a reporting unit to determine if the quantitative impairment test is necessary. This guidance will be effective for annual and interim impairment tests performed in annual reporting periods beginning after December 15, 2020, and early adoption is permitted for annual or interim impairment tests performed after January 1, 2017. The Company is evaluating the effect that this guidance will have on its Consolidated Financial Statements and related disclosures.
3. Acquisition
On November 7, 2017, the Company acquired all of the issued and outstanding share capital of Dimension Therapeutics, Inc. (Dimension), headquartered in Cambridge, Massachusetts for a purchase price of $6.00 per share or $152.3 million in cash. Dimension is developing new therapeutic adeno-associated virus (AAV) gene therapies for those living with rare metabolic diseases. In connection with the acquisition, the Company also paid a $2.9 million termination fee to REGENXBIO Inc. (REGENX), as a result of a previously existing merger agreement between REGENX and Dimension and assumed all the outstanding equity awards of Dimension at the date of the acquisition. The assumed equity awards were valued at $15.4 million using Black-Scholes option pricing model on the acquisition date. The equity awards assumed were allocated $9.0 million to the purchase consideration relating to the vested portion of stock options assumed, $2.2 million for the acceleration of certain awards that were recognized immediately as expense in the post-combination financial statements and $4.2 million is expected to be recognized as expense prospectively over the employee’s remaining service period. The acquisition date fair value of the consideration transferred for Dimension was approximately $164.1 million, which consisted of the following (in thousands):
|
Cash payments |
$ |
152,292 |
|
|
Fair value of vested stock options assumed |
|
8,979 |
|
|
REGENX termination fee |
|
2,850 |
|
|
Fair value of total consideration |
$ |
164,121 |
|
The following table summarizes the fair values of assets acquired and liabilities assumed as of the date of acquisition (in thousands):
|
Cash and cash equivalents |
$ |
12,338 |
|
|
|
Short-term investments |
|
9,737 |
|
|
|
Other current assets |
|
11,155 |
|
|
|
Property and equipment |
|
6,580 |
|
|
|
In-process research and development |
|
129,000 |
|
|
|
Bayer collaboration agreement |
|
13,526 |
|
|
|
Accounts payable and accrued liabilities |
|
(10,265 |
) |
|
|
Notes payable |
|
(4,944 |
) |
|
|
Deferred tax liabilities |
|
(47,412 |
) |
|
|
Net identifiable net assets acquired |
|
119,715 |
|
|
|
Goodwill |
|
44,406 |
|
|
|
Net assets acquired |
$ |
164,121 |
|
|
The transaction was accounted for as a business combination under the acquisition method of accounting as outlined in ASC 805, Business Combinations. The excess of purchase consideration over the fair value of net tangible and identifiable intangible assets acquired was recorded as goodwill. The fair values assigned to tangible and identifiable intangible assets acquired and liabilities assumed were based on management’s estimates and assumptions based on the information that was available as of the date of the acquisition. See also Note 6 “Intangible Assets, net” for a description of the intangible assets.
F-13
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The Company recorded $47.4 million in non-current deferred tax liability resulting from the acquisition reflecting the tax impact of the difference between the book basis and tax basis of acquired IPR&D. Such deferred income tax liability is not used to offset deferred tax assets when analyzing the Company’s valuation allowance as the acquired IPR&D is considered to have an indefinite life until the Company completes or abandons development of the acquired IPR&D.
The goodwill balance is primarily attributed to the deferred tax liabilities arising on the temporary differences on IPR&D assets between book and tax basis as well as the relating to the assembled workforce and expanded market opportunities when integrating Dimension’s research with the Company. The goodwill balance is not deductible for U.S. income tax purposes.
The assumed notes payable of $4.9 million, along with any outstanding interest was repaid in December 2017. In connection with the acquisition, the Company recognized transaction costs of $6.0 million as selling, general and administrative expense.
Pro Forma Financial Information
The Company's consolidated statement of operations from November 7, 2017 through December 31, 2017 includes Dimension total revenue of $2.1 million and a net loss of $7.5 million.
The following supplemental unaudited pro forma information presents the financial results as if the acquisition had occurred on January 1, 2016 (in thousands):
|
|
For the year ended December 31, |
|
|||||
|
|
2017 |
|
|
2016 |
|
||
|
Total revenues |
$ |
18,528 |
|
|
$ |
12,684 |
|
|
Net loss |
|
341,737 |
|
|
|
306,596 |
|
The unaudited pro forma financial information include pro forma adjustments that assume the acquisition occurred on January 1, 2016. These items include adjustments to remove the impact of transaction costs related to the acquisition of $9.6 million for the year ended December 31, 2017 and to record the amortization of definite-lived intangible assets of $1.8 million and $11.1 million for the years ended December 31, 2017 and 2016, respectively. Other adjustments include reduction of interest income, amounts related to severance of certain employees, acceleration of certain equity awards, and adjustments to conform to the Company’s accounting policies on revenue. These unaudited pro forma results are presented for informational purposes only and are not necessarily indicative of what the actual results of operations of the combined company would have been if the acquisition had occurred at the beginning of the period presented, nor are they indicative of future results of operations.
4. Fair Value Measurements
Financial assets and liabilities are recorded at fair value. The carrying amount of certain financial instruments, including cash and cash equivalents, accounts receivable, accounts payable and accrued liabilities approximate fair value due to their relatively short maturities. Assets and liabilities recorded at fair value on a recurring basis in the balance sheets are categorized based upon the level of judgment associated with the inputs used to measure their fair values. Fair value is defined as the exchange price that would be received for an asset or an exit price that would be paid to transfer a liability in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. The authoritative guidance on fair value measurements establishes a three-tier fair value hierarchy for disclosure of fair value measurements as follows:
Level 1—Inputs are unadjusted, quoted prices in active markets for identical assets or liabilities at the measurement date;
Level 2—Inputs are observable, unadjusted quoted prices in active markets for similar assets or liabilities, unadjusted quoted prices for identical or similar assets or liabilities in markets that are not active, or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the related assets or liabilities; and
Level 3—Unobservable inputs that are significant to the measurement of the fair value of the assets or liabilities that are supported by little or no market data.
The Company’s financial instruments consist of Level 1 and Level 2 assets. Where quoted prices are available in an active market, securities are classified as Level 1. Money market funds are classified as Level 1. Level 2 assets consist primarily of corporate bonds, asset backed securities, commercial paper and U.S. Government agency securities based upon quoted market prices for similar movements in active markets, quoted prices for identical or similar instruments in markets that are not active and model-based valuation techniques for which all significant inputs are observable in the market or can be corroborated by observable market data for substantially the full term of the assets. Where applicable these models project future cash flows and discount the future amounts to a present value using market-based observable inputs obtained from various third party data providers, including but not limited to, benchmark yields, interest rate curves, reported trades, broker/dealer quotes and reference data.
F-14
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The following table sets forth the fair value of the Company’s financial assets and liabilities remeasured on a recurring basis based on the three-tier fair value hierarchy (in thousands):
|
|
December 31, 2017 |
|
|||||||||||||
|
|
Level 1 |
|
|
Level 2 |
|
|
Level 3 |
|
|
Total |
|
||||
|
Financial Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds |
$ |
79,670 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
79,670 |
|
|
Corporate bonds |
|
— |
|
|
|
39,240 |
|
|
|
— |
|
|
|
39,240 |
|
|
U.S. Government Treasury and agency securities |
|
— |
|
|
|
104,739 |
|
|
|
— |
|
|
|
104,739 |
|
|
Total financial assets |
$ |
79,670 |
|
|
$ |
143,979 |
|
|
$ |
— |
|
|
$ |
223,649 |
|
|
|
December 31, 2016 |
|
|||||||||||||
|
|
Level 1 |
|
|
Level 2 |
|
|
Level 3 |
|
|
Total |
|
||||
|
Financial Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds |
$ |
123,536 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
123,536 |
|
|
Corporate bonds |
|
— |
|
|
|
207,726 |
|
|
|
— |
|
|
|
207,726 |
|
|
Commercial paper |
|
— |
|
|
|
11,970 |
|
|
|
— |
|
|
|
11,970 |
|
|
Asset-backed securities |
|
— |
|
|
|
27,713 |
|
|
|
— |
|
|
|
27,713 |
|
|
U.S. Government Treasury and agency securities |
|
— |
|
|
|
111,931 |
|
|
|
— |
|
|
|
111,931 |
|
|
Total financial assets |
$ |
123,536 |
|
|
$ |
359,340 |
|
|
$ |
— |
|
|
$ |
482,876 |
|
5. Balance Sheet Components
Cash Equivalents and Investments
The fair values of cash equivalents, short-term investments, and long-term investments classified as available-for-sale securities, consisted of the following (in thousands):
|
|
December 31, 2017 |
|
|||||||||||||
|
|
|
|
|
|
Gross Unrealized |
|
|
|
|
|
|||||
|
|
Amortized Cost |
|
|
Gains |
|
|
Losses |
|
|
Estimated Fair Value |
|
||||
|
Money market funds |
$ |
79,670 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
79,670 |
|
|
Corporate bonds |
|
39,330 |
|
|
|
— |
|
|
|
(90 |
) |
|
|
39,240 |
|
|
U.S. Government Treasury and agency securities |
|
105,029 |
|
|
|
— |
|
|
|
(290 |
) |
|
|
104,739 |
|
|
Total |
$ |
224,029 |
|
|
$ |
— |
|
|
$ |
(380 |
) |
|
$ |
223,649 |
|
|
|
December 31, 2016 |
|
|||||||||||||
|
|
|
|
|
|
Gross Unrealized |
|
|
|
|
|
|||||
|
|
Amortized Cost |
|
|
Gains |
|
|
Losses |
|
|
Estimated Fair Value |
|
||||
|
Money market funds |
$ |
123,536 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
123,536 |
|
|
Corporate bonds |
|
207,909 |
|
|
|
14 |
|
|
|
(197 |
) |
|
|
207,726 |
|
|
Commercial paper |
|
11,970 |
|
|
|
— |
|
|
|
— |
|
|
|
11,970 |
|
|
Asset-backed securities |
|
27,712 |
|
|
|
3 |
|
|
|
(2 |
) |
|
|
27,713 |
|
|
U.S. Government Treasury and agency securities |
|
112,166 |
|
|
|
10 |
|
|
|
(245 |
) |
|
|
111,931 |
|
|
Total |
$ |
483,293 |
|
|
$ |
27 |
|
|
$ |
(444 |
) |
|
$ |
482,876 |
|
At December 31, 2017, the remaining contractual maturities of available-for-sale securities were less than two years. There have been no significant realized gains or losses on available-for-sale securities for the periods presented. All marketable securities with unrealized losses at December 31, 2017 have been in a loss position for less than twelve months or the loss is not material and were temporary in nature. We do not intend to sell the investments that are in an unrealized loss position before recovery of their amortized cost basis.
F-15
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
Inventory consists of the following (in thousands):
|
|
|
December 31, |
|
|
|||||
|
|
|
2017 |
|
|
2016 |
|
|
||
|
Work-in-process |
|
$ |
737 |
|
|
$ |
— |
|
|
|
Finished goods |
|
|
20 |
|
|
|
— |
|
|
|
Total inventory |
|
$ |
757 |
|
|
$ |
— |
|
|
Property and Equipment, net
Property and equipment, net consists of the following (in thousands):
|
|
|
December 31, |
|
|||||
|
|
|
2017 |
|
|
2016 |
|
||
|
Research and development equipment |
|
$ |
7,696 |
|
|
$ |
2,154 |
|
|
Furniture and office equipment |
|
|
2,873 |
|
|
|
2,595 |
|
|
Computer equipment |
|
|
2,563 |
|
|
|
2,013 |
|
|
Software |
|
|
4,182 |
|
|
|
3,931 |
|
|
Leasehold improvements |
|
|
15,085 |
|
|
|
12,486 |
|
|
Construction-in-progress |
|
|
429 |
|
|
|
37 |
|
|
Property and equipment, gross |
|
|
32,828 |
|
|
|
23,216 |
|
|
Less accumulated depreciation |
|
|
(10,991 |
) |
|
|
(6,161 |
) |
|
Property and equipment, net |
|
$ |
21,837 |
|
|
$ |
17,055 |
|
Depreciation expense for the years ended December 31, 2017, 2016 and 2015 was $4.8 million, $3.4 million and $1.4 million respectively. Amortization of leasehold improvements and software is included in depreciation expense.
Accrued Liabilities
Accrued liabilities consists of the following (in thousands):
|
|
|
December 31, |
|
|||||
|
|
|
2017 |
|
|
2016 |
|
||
|
Research and clinical study expenses |
|
$ |
17,141 |
|
|
$ |
18,593 |
|
|
Payroll and related expenses |
|
|
26,527 |
|
|
|
17,226 |
|
|
Repayment liability under collaboration agreement |
|
|
3,681 |
|
|
|
13,650 |
|
|
Contract liability |
|
|
5,986 |
|
|
|
— |
|
|
Other |
|
|
8,092 |
|
|
|
5,085 |
|
|
Total accrued liabilities |
|
$ |
61,427 |
|
|
$ |
54,554 |
|
6. Intangible Assets, net
In connection with the acquisition as described in Note 3 “Acquisition" the Company recognized IPR&D assets of $129.0 million and a contract asset of $13.5 million. The estimated fair value of these intangible assets was measured using Level 3 inputs as of the acquisition date.
IPR&D assets represent the fair value of acquired programs to develop an AAV gene therapy for OTC deficiency and to develop an AAV gene therapy for glycogen storage disease type Ia. The fair value of IPR&D assets acquired was determined based on the discounted present value of each research project’s projected cash flows using an income approach, including the application of probability factors related to the likelihood of success of the program reaching final development and commercialization. Additionally, the projections consider the relevant market sizes and growth factors, estimated future cash flows from product sales resulting from completed products and in-process projects and timing and costs to complete the in-process projects. The rates utilized to discount the net cash flows to their present value are commensurate with the stage of development of the projects and uncertainties in the economic estimates used in the projections. IPR&D assets are considered to be indefinite-live until the completion or abandonment of the associated research and development efforts.
F-16
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The contract asset represents the fair value of the agreement with Bayer HealthCare LLC to research, develop, and commercialize AAV gene therapy products for treatment of hemophilia A. The fair value of the contract asset was determined based on the discounted present value of the estimated net future income and would be amortized to research and development expense over the research term which is expected to be complete in 2019. The Company recorded research and development expense of $1.0 million for the period from November 7, 2017 through December 31, 2017 related to the amortization of the asset. As of December 31, 2017, the estimated future amortization expense associated with the contract asset is $10.5 million in 2018 and $2.0 million in 2019.
7. License and Research Agreements
Saint Louis University License Agreement
In November 2010, the Company entered into a license agreement with Saint Louis University (SLU). Under the terms of this license agreement, SLU granted the Company an exclusive worldwide license to make, have made, use, import, offer for sale, and sell therapeutics related to SLU’s beta-glucuronidase product for use in the treatment of human diseases.
The Company made a milestone payment of $0.1 million upon approval of Mepsevii for treatment of MPS 7. The Company is required to pay to SLU a low single-digit royalty on net sales of the licensed products in any country or region, upon reaching a certain level of cumulative worldwide sales of the product, if such product sales are ever achieved.
St. Jude Children’s Research Hospital License Agreement
In September 2012, the Company entered into a license agreement with St. Jude Children’s Research Hospital (St. Jude). Under the terms of this license agreement, St. Jude granted the Company an exclusive license under certain know-how to research, develop, make, use, offer to sell, import, and otherwise commercialize and exploit St. Jude’s protective protein, cathepsin, a protein product to treat, prevent, and/or diagnose galactosialidosis and other monogenetic diseases.
The Company will pay to St. Jude a royalty of less than 1% on net sales of the licensed products in the licensed territories, if such product sales are ever achieved.
Baylor Research Institute License Agreement
In September 2012, the Company entered into a license agreement with Baylor Research Institute (BRI). Under the terms of this license agreement, BRI exclusively licensed to the Company its territories for certain intellectual property related to triheptanoin.
The Company may make future payments of up to $10.5 million contingent upon attainment of various development milestones and $7.5 million contingent upon attainment of various sales milestones. Additionally, the Company will pay to BRI a mid-single digit royalty on net sales of the licensed product in the licensed territories, if such product sales are ever achieved.
Kyowa Hakko Kirin Collaboration and License Agreement
In August 2013, the Company entered into a collaboration and license agreement with Kyowa Hakko Kirin Co., Ltd. (KHK). Under the terms of this collaboration and license agreement, as amended, the Company and KHK will collaborate on the development and commercialization of certain products containing burosumab in the field of orphan diseases in the United States and Canada, or the profit share territory, and in the European Union and Switzerland, or the European territory, and the Company will have the right to develop and commercialize such products in the field of orphan diseases in Mexico and Central and South America, or Latin America. In the field of orphan diseases, and except for ongoing studies being conducted by KHK, the Company will be the lead party for development activities in the profit share territory and in the European territory until the applicable transition date; the Company will also be the lead party for core development activities conducted in Japan and Korea, for which the core development plan is limited to clinical trials mutually agreed to by the Company and KHK. The Company will share the costs for development activities in the profit share territory and the European territory conducted pursuant to the development plan before the applicable transition date equally with KHK, and KHK shall be responsible for 100% of the costs for development activities in Japan and Korea. On the applicable transition date in the profit share territory and the European territory, KHK will become the lead party and be responsible for the costs of the development activities. However, the Company will continue to share the costs of the studies commenced prior to the applicable transition date equally with KHK. The Company has the primary responsibility for conducting certain research and development services. If burosumab is approved, the Company and KHK will share commercial responsibilities and profits in the profit share territory until the applicable transition date and KHK will commercialize burosumab in the European territory. The Company will develop and commercialize burosumab in Latin America.
KHK will manufacture and supply burosumab for clinical use globally and will manufacture and supply burosumab for commercial use in the profit share territory and Latin America. The remaining profit or loss from commercializing products in the profit-share territory, until the applicable transition date, will be shared between the Company and KHK on a 50/50 basis. Thereafter, the Company will be entitled to receive a tiered double-digit revenue share in the mid-to-high 20% range in the profit share territory. Net proceeds from the anticipated sale of the priority review voucher, if received, will be shared equally with KHK. The Company
F-17
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
will also be entitled to receive a royalty of up to 10% on net sales in the European territory. In Latin America, the Company will pay to KHK a low single-digit royalty on net sales.
In May 2017, the Company signed an agreement with a wholly-owned subsidiary of KHK pursuant to which the Company was granted the right to commercialize burosumab in Turkey. KHK’s subsidiary has the option to assume responsibility for commercialization efforts from the Company, after a certain minimum period.
The Company is accounting for the agreement as a collaboration arrangement as defined in ASC 808, Collaborative Agreements. The Company’s expenses were reduced by $31.2 million and $25.3 million for the years ended December 31, 2017 and 2016, respectively, for its share of the costs as research and development and were reduced by $4.5 million and $1.5 million for the years ended December 31, 2017 and 2016, respectively, for its share of the selling, general and administrative expenses. As of December 31, 2017 and 2016, the Company had receivables in the amount of $10.3 million and $8.6 million, respectively, for this collaboration arrangement.
Arcturus Research Collaboration and License Agreement
In October 2015, the Company entered into a Research Collaboration and License Agreement with Arcturus Therapeutics, Inc. (Arcturus). The Company and Arcturus are collaborating on the research and development of therapies for select rare diseases. As consideration for entering into the arrangement, the Company paid Arcturus an upfront fee of $10.0 million. Arcturus has the primary responsibility for conducting certain research services, funded by the Company, and the Company will be responsible for development and commercialization costs.
Takeda License and Collaboration and Purchase Agreements
In June 2016, the Company executed a collaboration and license agreement with Takeda Pharmaceutical Company Limited (Takeda). Pursuant to the agreement, which became effective in July 2016, the Company obtained an exclusive license for a pre-clinical compound from Takeda in a pre-determined field of use. The Company is responsible for the development costs for the pre-clinical compound and the identified option product pursuant to an initial development plan. Because the license to the pre-clinical compound has no alternative future use, the estimated fair value of $0.7 million was recorded as a research and development expense upon acquisition. Any products resulting from the pre-clinical compound or the identified option product is referred to in this report as the “licensed product.” The Company discontinued the development efforts on the pre-clinical compound in the pre-determined field of use and the identified option product.
As part of the agreement, the Company and Takeda established a five-year research collaboration whereby the parties may mutually agree to add additional option products candidates to the collaboration, in which case the Company will bear the cost of the development activities, with certain exceptions.
In July 2016, the Company consummated a common stock purchase agreement, executed in conjunction with the collaboration and license agreement, whereby Takeda purchased 374,590 shares of the Company’s common stock for $40.0 million in cash. The fair market value of the common stock issued to Takeda was $27.3 million, based on the closing stock price of $72.95 on the date of issuance, resulting in a $12.7 million premium paid to the Company. The Company also received a put option to require Takeda to purchase an additional $25.0 million in shares of the Company’s common stock which was exercised in October 2016, whereby Takeda purchased 352,530 shares of the Company’s common stock for $25.0 million in cash. Takeda is subject to a five-year standstill (subject to customary exceptions or release). The Company estimated the fair value of the put options to be $0.9 million and recorded the put options in additional paid-in capital.
The Company also granted Takeda an exclusive option for Asian rights, for a limited period, to any licensed products and any additional products resulting from the collaboration, as well as an option to exclusively license one of the Company’s products for development and commercialization in Japan. If Takeda exercises any of its option rights to license a product pursuant to the agreement, Takeda will pay for the development costs within the licensed territory, will share in a portion of the global development costs, and will make a milestone payment upon regulatory approval. Takeda will also owe royalties on net sales in the licensed territory for any licensed product, depending on the development stage when the product is licensed as well as sales levels. The royalties related to the option to license the Company’s product, as well as the additional product are subject to future good faith negotiations at the time that the option is exercised.
The research and license agreement and the stock purchase agreement are being accounted for as one arrangement because they were entered into at the same time with interrelated financial terms. The Company analogized to Topic 606 for the accounting for the arrangements. The Company concluded that there are multiple promised goods and services under the collaboration agreement, including obligations related to research and development services with respect to licensed products as well as committee participation, which were determined to represent distinct performance obligations. The total consideration received from Takeda was $14.3 million and was comprised of the $12.7 million premium on the sale of the common stock, the $0.9 million estimated fair value of the put options, and the $0.7 million estimated fair value of the pre-clinical compound.
F-18
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The Company is responsible for the costs under the initial development plan. A significant portion of this work is performed by Takeda which has an estimated cost of approximately $10.0 million to $11.2 million and is subject to changes as development activities are adjusted and cost estimates are refined. The Company concluded that the payment to Takeda is not in return for a distinct service that Takeda transfers to the Company, therefore, the payment made to Takeda is accounted for as a reduction in the transaction price. As of December 31, 2017, the Company concluded that $3.1 million of the estimated transaction price should not be constrained because it is probable that a significant reversal in the amount to be recognized will not occur. The unconstrained transaction price was allocated to the distinct performance obligations on a relative standalone selling price basis. The Company recorded $2.5 million for the year ended December 31, 2017, respectively, as a reduction of research and development expenses by measuring the progress toward complete satisfaction of the individual performance obligation using an input measure.
The Company concluded that the remaining transaction price should continue to be constrained as of December 31, 2017. The Company will continue to re-evaluate the application of the constraint to the transaction price at each reporting period end date.
Costs incurred by the Company associated with co-development activities performed under this collaboration are included in research and development expense in the accompanying consolidated statements of operations. As of December 31, 2017 and 2016, the Company had a repayment liability in the amount of $3.7 million and $14.3 million, respectively, and a contract liability in the amount of $0.6 million as of December 31, 2017 and none as of December 31, 2016.
University of Pennsylvania
The Company has an agreement with University of Pennsylvania School of Medicine (Penn) to sponsor certain research related to liver and hemophilia gene therapy. In consideration for funding such research, Penn granted the Company an option to obtain a worldwide, non-exclusive or exclusive, royalty-bearing license, with the right to sublicense, under certain patent rights conceived, created or reduced to practice in the conduct of the research. The Company is required to reimburse Penn for filing, prosecuting and maintaining such patent rights unless and until the Company declines to exercise its option. Penn provides the Company with task-based, scientific reports of progress and results of the research, and granted the Company a royalty-free, nontransferable, non-exclusive right to copy and distribute any research reports furnished to the Company for any reasonable purpose, provided the results are not made publicly available until certain conditions are met, and the right to use, disclose and otherwise exploit the research results for any reasonable purpose, subject to similar restrictions on our public disclosure of the research results. Otherwise, the sponsored research agreement contains customary confidentiality provisions.
The Company also has a research, collaboration, and license agreement with Penn, which provides the terms for the Company and Penn to collaborate with respect to the pre-clinical development of gene therapy products for the treatment of certain indications. Under the agreement, Penn granted the Company an exclusive, worldwide license to certain patent rights arising out of the research program, subject to certain retained rights, and a non-exclusive, worldwide license to certain Penn intellectual property, in each case to research, develop, make, have made, use, sell, offer for sale, commercialize and import licensed products in each indication for the term of the agreement. The Company will fund the cost of the research program in accordance with a mutually agreed-upon research budget and will be responsible for clinical development, manufacturing and commercialization of each indication. The Company will make milestone payments of up to $5.0 million for each indication, if certain development milestones are achieved over time, as well as low to mid-single digit royalties on net sales of each licensed product. The Company will also make milestone payments per approved product if certain commercial milestones are achieved.
REGENXBIO, Inc.
The Company has a license agreement with REGENX, for an exclusive, sublicensable, worldwide commercial license under certain intellectual property for preclinical and clinical research and development, and commercialization of drug therapies using REGENX 's licensed patents for the treatment of hemophilia A, hemophilia B, OTC deficiency and GSD1a. The Company will pay an annual fee and certain milestone fees per disease indication, low to mid single-digit royalty percentages on net sales of licensed products, and milestone and sublicense fees owed by REGENX to its licensors, contingent upon the attainment of certain development activities as outlined in the agreement.
The Company also has an option and license agreement with REGENX under which the Company has an exclusive, sublicensable, worldwide license to make, have made, use, import, sell, and offer for sale licensed products with respect to three disease indications, subject to certain exclusions and has an option for another disease indication. The exercise of the remaining option will require payment of a $1.0 million option fee, is subject to availability and if not exercised, will automatically terminate in 2019 (which may be extended for additional time for a fee). Each exercised option carries an annual maintenance fees of $0.1 million. In addition, for each option exercised, the Company is obligated to pay up to $9.0 million upon achievement of various milestones, as well as mid to high single-digit royalties on net sales of licensed products and mid single-digit to low double-digit percentage sublicenses fees, if any.
F-19
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The Company has an agreement with Bayer Healthcare LLC (Bayer) to research, develop and commercialize AAV gene therapy products for treatment of hemophilia A (DTX 201). Under this agreement, Bayer has been granted an exclusive license to develop and commercialize one or more novel gene therapies for hemophilia A. The Company is responsible for the development of DTX201 under the agreement through a proof-of-concept (POC) clinical trial, in accordance with the mutually agreed upon research budget. Upon the successful demonstration of clinical POC, the agreement requires that Bayer use commercially reasonable efforts to manage and fund any subsequent clinical trials and commercialization of gene therapy products for treatment of hemophilia A. Bayer will have worldwide rights to commercialize the potential future product.
Bayer is responsible to fund certain research and development services performed by the Company in the performance of its obligations under the annual research plan and budget. Under the terms of the agreement with Bayer, the Company is eligible to receive development and commercialization milestone payments of up to $232.0 million, as well as, royalty payments ranging in the high single-digit to low double-digit percentages, not exceeding the mid-teens, of net sales of licensed products. In December 2017, the first milestone was achieved and the Company invoiced Bayer $5.0 million for the milestone.
As of the acquisition date of November 7, 2017, the Company valued the contract under ASC 805 Business Combinations and recorded an intangible asset of $13.5 million. The intangible asset will be amortized to research and development expense over the remainder of the research term which is expected to be complete in 2019. The Company recorded a research and development expense of $1.0 million for the period from November 7, 2017 through December 31, 2017 related to the amortization of the intangible asset.
The Company also evaluated the agreement under ASC 606 Revenue from Contracts with Customers, and recorded a contract liability as of November 7, 2017 of $2.5 million. It was determined that the performance obligations under the agreement includes (i) research and development services to be provided over the research term, (ii) a development and commercialization license and (iii) the Company’s participation in certain committees. It was determined that these performance obligations are not distinct in the context of the contract and therefore are a single performance obligation. The Company calculated the transaction price by including the unconstrained milestones along with the estimated payments for research and development services. $2.1 million was recorded as collaboration and license revenue for the period from November 7, 2017 through December 31, 2017 by measuring the progress toward complete satisfaction of the performance obligation using an input measure and $5.4 million of contract liability as of December 31, 2017. The performance obligation under the contract is estimated to be substantially complete by end of 2019.
8. Equity
At-the-Market Offerings
In July 2016, the Company entered into an At-The-Market (ATM) sales agreement with Cowen and Company, LLC (Cowen), whereby the Company sold $150.0 million in aggregate proceeds of common stock, through Cowen as our sales agent. During the years ended December 31, 2017 and 2016, the Company sold 912,351 and 1,159,415 shares of common stock, resulting in net proceeds of approximately $67.6 million and $79.5 million, respectively, after commissions and other offering costs.
In July 2017, the Company entered into an additional ATM sales agreement with Cowen whereby the Company may sell up to $150.0 million in aggregate proceeds of common stock from time to time, through Cowen as its sales agent. During the year ended December 31, 2017, the Company sold an additional 1,338,866 shares of common stock, resulting in net proceeds of approximately $64.3 million, after commissions and other offering costs.
Common Stock Warrants
As of December 31, 2017 and 2016, there was an aggregate of 149,700 of common stock warrants outstanding that were issued in June 2010 and June 2011 with a contractual term of 10 years and an exercise price of $3.01.
F-20
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
Equity Plan Awards
In 2011, the Company adopted the 2011 Equity Incentive Plan (the 2011 Plan). The 2011 Plan provides for the granting of stock-based awards to employees, directors, and consultants under terms and provisions established by the board of directors. In 2014, the Company adopted the 2014 Incentive Plan (the 2014 Plan). The 2014 Plan had 2,250,000 shares of common stock available for future issuance at the time of its inception, which included 655,038 shares available under the 2011 Plan, which were transferred to the 2014 Plan upon adoption. No further grants subsequent to the IPO were made under the 2011 Plan. The 2014 Plan provides for automatic annual increases in shares available for grant, beginning on January 1, 2015 through January 1, 2024. Under the terms of the 2014 Plan, awards may be granted at an exercise price not less than fair market value. For employees holding more than 10% of the voting rights of all classes of stock, the exercise prices for awards must be at least 110% of fair market of the common stock on the grant date, as determined by the board of directors. The term of an award granted under the 2014 Plan may not exceed ten years. Typically, the vesting schedule for option grants to the employees provides that 1/4 of the grant vests upon the first anniversary of the date of grant, with the remainder of the shares vesting monthly thereafter at a rate of 1/48 of the total shares subject to the option. The vesting schedule for RSU grants provides that 1/4 of the grant vests upon the annual anniversary of the date of grant over the period of four years.
As part of the acquisition of Dimension (discussed in Note 3 “Acquisition”), the Company assumed an equivalent 639,897 options to purchase shares of common stock of the Company from the equity plans of Dimension. No further grants subsequent to the acquisition are available under these equity plans.
As of December 31, 2017, an aggregate of 9,732,069 shares of common stock have been authorized for issuance under all equity award plans.
Stock Option Activity
The following table summarizes activity under the Company’s stock option plans, including the 2011 Plan, the 2014 Plan, the assumed equity awards from the Dimension plans and related information:
|
|
|
Options Outstanding |
|
|||||||||||||
|
|
|
Number of Options |
|
|
Weighted- Average Exercise Price |
|
|
Weighted-Average Remaining Contractual Term (Years) |
|
|
Aggregate Intrinsic Value |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(In thousands) |
|
|
|
Outstanding — December 31, 2014 |
|
|
2,739,475 |
|
|
$ |
16.15 |
|
|
|
8.43 |
|
|
$ |
79,840 |
|
|
Options granted |
|
|
2,013,350 |
|
|
|
90.60 |
|
|
|
|
|
|
|
|
|
|
Options exercised |
|
|
(795,825 |
) |
|
|
8.65 |
|
|
|
|
|
|
|
|
|
|
Options cancelled |
|
|
(130,037 |
) |
|
|
31.49 |
|
|
|
|
|
|
|
|
|
|
Outstanding — December 31, 2015 |
|
|
3,826,963 |
|
|
$ |
56.36 |
|
|
|
8.58 |
|
|
$ |
217,386 |
|
|
Options granted |
|
|
1,435,995 |
|
|
|
67.00 |
|
|
|
|
|
|
|
|
|
|
Options exercised |
|
|
(425,922 |
) |
|
|
21.21 |
|
|
|
|
|
|
|
|
|
|
Options cancelled |
|
|
(407,564 |
) |
|
|
70.87 |
|
|
|
|
|
|
|
|
|
|
Outstanding — December 31, 2016 |
|
|
4,429,472 |
|
|
$ |
61.85 |
|
|
|
8.22 |
|
|
$ |
79,135 |
|
|
Options granted |
|
|
1,328,860 |
|
|
|
71.99 |
|
|
|
|
|
|
|
|
|
|
Options assumed |
|
|
639,897 |
|
|
|
27.97 |
|
|
|
|
|
|
|
|
|
|
Options exercised |
|
|
(478,470 |
) |
|
|
16.25 |
|
|
|
|
|
|
|
|
|
|
Options cancelled |
|
|
(520,349 |
) |
|
|
78.70 |
|
|
|
|
|
|
|
|
|
|
Outstanding — December 31, 2017 |
|
|
5,399,410 |
|
|
$ |
62.75 |
|
|
|
7.40 |
|
|
$ |
37,687 |
|
|
Vested and exercisable — December 31, 2017 |
|
|
2,709,510 |
|
|
$ |
53.53 |
|
|
|
6.22 |
|
|
$ |
34,558 |
|
|
Vested and expected to vest — December 31, 2017 |
|
|
5,216,110 |
|
|
$ |
62.50 |
|
|
|
7.35 |
|
|
$ |
37,528 |
|
The aggregate intrinsic values of options outstanding, vested and exercisable, and vested and expected to vest were calculated as the difference between the exercise price of the options and the fair value of the Company’s common stock. The total intrinsic value of options exercised during the years ended December 31, 2017, 2016, and 2015 was $20.4 million, $22.2 million and $59.0 million, respectively. Cash received from the exercise of options was $7.8 million, $9.0 million, and $6.9 million as of December 31, 2017, 2016, and 2015, respectively.
F-21
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The weighted-average estimated fair value of stock options granted was $44.20, $40.49 and $54.90 per share of the Company’s common stock during the years ended December 31, 2017, 2016, and 2015, respectively. The total estimated fair value of options vested during the years ended December 31, 2017, 2016, and 2015 was $49.1 million, $43.8 million, and $10.7 million, respectively.
Restricted Stock Units
The following table summarizes activity under the Company’s Restricted Stock Units (RSUs) from the 2014 Plan and related information:
|
|
RSUs Outstanding |
|
|||||
|
|
Number of Shares |
|
|
Weighted- Average Grant Date Fair Value |
|
||
|
Unvested — December 31, 2014 |
|
31,000 |
|
|
$ |
53.23 |
|
|
RSUs granted |
|
187,260 |
|
|
|
89.67 |
|
|
RSUs released |
|
(18,209 |
) |
|
|
54.72 |
|
|
RSUs cancelled |
|
(2,900 |
) |
|
|
84.89 |
|
|
Unvested — December 31, 2015 |
|
197,151 |
|
|
$ |
87.24 |
|
|
RSUs granted |
|
477,816 |
|
|
|
66.83 |
|
|
RSUs released |
|
(52,273 |
) |
|
|
82.59 |
|
|
RSUs cancelled |
|
(48,950 |
) |
|
|
78.04 |
|
|
Unvested — December 31, 2016 |
|
573,744 |
|
|
$ |
71.45 |
|
|
RSUs granted |
|
516,161 |
|
|
|
71.58 |
|
|
RSUs released |
|
(156,021 |
) |
|
|
71.93 |
|
|
RSUs cancelled |
|
(112,324 |
) |
|
|
76.78 |
|
|
Unvested — December 31, 2017 |
|
821,560 |
|
|
$ |
70.71 |
|
The fair value of the RSUs is determined on the grant date based on the fair value of the Company’s common stock. The fair value of the RSUs is recognized as expense ratably over the vesting period of one to four years. The total fair value of the 156,021 shares vested during 2017 was approximately $11.2 million with an aggregate intrinsic value of the shares of $9.2 million.
Performance Stock Units
In December 2017, the Company granted 508,850 performance stock units (PSUs) to certain employees at a grant date fair value of $48.03 per share of the Company’s common stock, of which no shares vested during 2017. These PSUs are subject to vest only if certain specified criteria are achieved and the employees’ continued service with the Company after achievement of the specified criteria. Stock-based compensation for these PSUs is recognized over the service period beginning in the period the Company determines it is probable that the performance criteria will be achieved. In December 2017, the Company began recognizing stock compensation expense on one of the criteria as the outcome was deemed probable.
Employee Stock Purchase Plan
In January 2014, the Company adopted the 2014 Employee Stock Purchase Plan (ESPP) and had reserved a total of 600,000 shares of common stock for issuance under the ESPP. The ESPP provides for automatic annual increases in shares available for grant, beginning on January 1, 2015 through January 1, 2024. Eligible employees may purchase common stock at 85% of the lesser of the fair market value of common stock on the offering date or the purchase date with a six-month look-back feature. ESPP purchases are settled with common stock from the ESPP’s previously authorized and available pool of shares. During the year ended December 31, 2017, the Company issued 62,412 shares of common stock under the ESPP.
Stock-Based Compensation Expense
Total stock-based compensation recognized was as follows (in thousands):
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Research and development |
|
$ |
38,212 |
|
|
$ |
29,412 |
|
|
$ |
17,100 |
|
|
Selling, general and administrative |
|
|
29,802 |
|
|
|
18,897 |
|
|
|
7,784 |
|
|
Total stock-based compensation expense |
|
$ |
68,014 |
|
|
$ |
48,309 |
|
|
$ |
24,884 |
|
F-22
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
As of December 31, 2017, the total unrecognized compensation expense related to unvested equity awards, net of estimated forfeitures, was $165.3 million, which the Company expects to recognize over an estimated weighted-average period of 2.52 years. In determining the estimated fair value of the stock options and ESPP, the Company uses the Black-Scholes option-pricing model and assumptions discussed below. Each of these inputs is subjective and generally requires significant judgment to determine.
Expected Term—The Company’s expected term represents the period that the Company’s stock-based awards are expected to be outstanding and is determined using the simplified method (based on the mid-point between the vesting date and the end of the contractual term).
Expected Volatility—As the Company does not have sufficient historical stock price information from the Company to meet the expected life of the stock-based awards, our approach to estimating expected volatility is to phase in our own common stock trading history and supplement the remaining historical information with a blended volatility from the trading history from the common stock of the set of comparable publicly traded biopharmaceutical companies. When selecting comparable publicly traded biopharmaceutical companies on which to base the expected stock price volatility, we selected companies with comparable characteristics to it, including enterprise value, risk profiles, position within the industry, and with historical share price information sufficient to meet the expected life of the stock-based awards. The historical volatility data was computed using the daily closing prices for the selected companies’ shares during the equivalent period of the calculated expected term of the stock-based awards. The average volatility for comparable publicly traded biopharmaceutical companies over a period equal to the expected term of the stock option grants is used to supplement the Company’s historical volatility. We will continue to apply this process until a sufficient amount of historical information regarding the volatility of our own stock price becomes available.
Risk-Free Interest Rate—The risk-free interest rate is based on the U.S. Treasury zero coupon issues in effect at the time of grant for periods corresponding with the expected term of option.
Expected Dividend—The Company has never paid dividends on its common stock and has no plans to pay dividends on its common stock. Therefore, the Company used an expected dividend yield of zero.
The fair value of stock option awards granted was estimated at the date of grant using a Black-Scholes option-pricing model with the following weighted-average assumptions:
|
|
|
Year Ended December 31, |
|
||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|
Expected term (years) |
|
6.23 |
|
|
6.23 |
|
|
6.23 |
|
|
Expected volatility |
|
65% |
|
|
65% |
|
|
65% |
|
|
Risk-free interest rate |
|
2.1% |
|
|
1.5% |
|
|
1.8% |
|
|
Expected dividend rate |
|
0.0% |
|
|
0.0% |
|
|
0.0% |
|
10. Defined Contribution Plan
Since 2013, the Company has sponsored a 401(k) retirement plan, in which substantially all of its full-time employees are eligible to participate. Eligible participants may contribute a percentage of their annual compensation to this plan, subject to statutory limitations. Prior to 2015, the Company had not provided any contributions to the plan. The Company recorded $2.1 million, $1.5 million, and $0.6 million as contribution expenses for the years ended December 31, 2017, 2016, and 2015, respectively.
11. Income Taxes
The components of the Company’s loss before income taxes were as follows (in thousands):
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Domestic |
|
$ |
250,917 |
|
|
$ |
192,287 |
|
|
$ |
145,618 |
|
|
Foreign |
|
|
67,421 |
|
|
|
53,552 |
|
|
|
— |
|
|
Total loss before income taxes |
|
$ |
318,338 |
|
|
$ |
245,839 |
|
|
$ |
145,618 |
|
F-23
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The components of the Company’s income tax provision were as follows (in thousands):
|
|
|
Year Ended December 31, |
|
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|
|||
|
Current provision for income taxes: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Federal |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
— |
|
|
|
State |
|
|
5 |
|
|
|
— |
|
|
|
— |
|
|
|
International |
|
|
42 |
|
|
|
35 |
|
|
|
— |
|
|
|
Total current tax provision |
|
|
47 |
|
|
|
35 |
|
|
|
— |
|
|
|
Deferred tax benefit: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Federal |
|
|
(16,243 |
) |
|
|
— |
|
|
|
— |
|
|
|
State |
|
|
(3 |
) |
|
|
— |
|
|
|
— |
|
|
|
International |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
Total deferred tax benefit |
|
|
(16,246 |
) |
|
|
— |
|
|
|
— |
|
|
|
Total (benefit from) provision for income taxes |
|
$ |
(16,199 |
) |
|
$ |
35 |
|
|
$ |
— |
|
|
The Company has incurred net operating losses since inception. The Company has not reflected any benefit of such net operating loss carryforwards in the accompanying financial statements. The Company has established a full valuation allowance against its deferred tax assets due to the uncertainty surrounding the realization of such assets.
The effective tax rate of our provision for income taxes differs from the federal statutory rate as follows:
|
|
|
Year Ended December 31, |
|
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|
|||
|
Federal statutory income tax rate |
|
|
34.0 |
|
% |
|
34.0 |
|
% |
|
34.0 |
|
% |
|
State income taxes, net of federal benefit |
|
|
— |
|
|
|
1.3 |
|
|
|
7.5 |
|
|
|
Federal tax credits |
|
|
9.0 |
|
|
|
13.7 |
|
|
|
5.9 |
|
|
|
Other |
|
|
(0.9 |
) |
|
|
(0.3 |
) |
|
|
0.8 |
|
|
|
Nondeductible permanent items |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
Stock-based compensation |
|
|
(0.6 |
) |
|
|
(1.4 |
) |
|
|
(1.3 |
) |
|
|
Uncertain tax positions |
|
|
(1.8 |
) |
|
|
2.0 |
|
|
|
(7.7 |
) |
|
|
Change in valuation allowance |
|
|
(32.5 |
) |
|
|
(41.9 |
) |
|
|
(39.2 |
) |
|
|
Foreign rate differential |
|
|
(7.2 |
) |
|
|
(7.4 |
) |
|
|
— |
|
|
|
Change in federal tax rate |
|
|
5.1 |
|
|
|
— |
|
|
|
— |
|
|
|
Provision for income taxes |
|
|
5.1 |
|
|
|
— |
|
|
|
— |
|
|
The tax effect of temporary differences that give rise to significant portions of the deferred tax assets is presented below (in thousands):
|
|
|
Year Ended December 31, |
|
|
|||||
|
|
|
2017 |
|
|
2016 |
|
|
||
|
Deferred tax assets: |
|
|
|
|
|
|
|
|
|
|
Loss carryforwards |
|
$ |
154,949 |
|
|
$ |
102,960 |
|
|
|
Tax credits |
|
|
119,542 |
|
|
|
78,324 |
|
|
|
Stock options |
|
|
21,336 |
|
|
|
16,084 |
|
|
|
Accruals and reserves |
|
|
5,939 |
|
|
|
10,039 |
|
|
|
Fixed assets and intangibles |
|
|
1,194 |
|
|
|
3,014 |
|
|
|
Other |
|
|
1,286 |
|
|
|
1,950 |
|
|
|
Gross deferred tax assets |
|
|
304,246 |
|
|
|
212,371 |
|
|
|
Valuation allowance |
|
|
(304,246 |
) |
|
|
(212,371 |
) |
|
|
Total deferred tax assets |
|
|
— |
|
|
|
— |
|
|
|
Deferred tax liabilities: |
|
|
|
|
|
|
|
|
|
|
In-process research and development |
|
|
(31,166 |
) |
|
|
— |
|
|
|
Net deferred tax assets (liabilities) |
|
$ |
(31,166 |
) |
|
$ |
— |
|
|
F-24
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
As of December 31, 2017 and 2016, the Company had $422.3 million and $315.5 million of federal net operating loss carryforwards available to reduce future taxable income that will begin to expire in 2030. As of December 31, 2017 and 2016, the Company had $457.7 million and $259.8 million of state net operating loss carryforwards available to reduce future taxable income that will begin to expire in 2030.
As of December 31, 2017 and 2016, the Company had federal research tax credit carryforwards of $4.5 million and $2.5 million available to reduce future tax liabilities that will begin to expire in 2030. As of December 31, 2017 and 2016, the Company had state research credit carryforwards of $12.4 million and $6.7 million available to reduce future tax liabilities that will be carried forward indefinitely.
As of December 31, 2017 and 2016, the Company had federal Orphan Drug Credits of $124.6 million and $84.3 million available to reduce future tax liabilities that will begin to expire in 2031.
The Company’s ability to use net operating loss and tax credit carryforwards to reduce future taxable income and liabilities may be subject to annual limitations pursuant to Internal Revenue Code Sections 382 and 383 as a result of ownership changes in the past and future. As a result of ownership changes in 2012 and 2011, $3.6 million of federal net operating loss carryforwards, $3.6 million of state net operating loss carryforwards, and $0.2 million of federal tax credits are permanently limited. Deferred tax assets for net operating losses and tax credits have been reduced and a corresponding adjustment to the valuation allowance has been recorded.
On November 7, 2017, the Company acquired Dimension (see Note 3 “Acquisition”). The Company recorded a $47.4 million deferred tax liability relating to the tax impact of future GAAP amortization or potential impairments associated with the identified intangible assets acquired, which are indefinitely lived assets and are not currently deductible for tax purposes. Due to the reduction of the US corporate tax rate to 21% in the period subsequent to the acquisition, the Company recorded a net decrease to the deferred tax liability of $16.2 million with a corresponding benefit from income taxes of $16.2 million for the year ended December 31, 2017.
The valuation allowance increased by $91.9 million and $102.9 million during the year ended December 31, 2017 and 2016, respectively.
On December 22, 2017, the U.S. government enacted comprehensive tax legislation commonly referred to as the Tax Cuts and Jobs Act (Tax Act). The Company has calculated its best estimate of the impact of the Tax Act in its year end income tax provision in accordance with its understanding of the Tax Act and guidance available as of the date of this filing. The provisional amount related to the remeasurement of certain deferred tax assets and liabilities based on the rates at which they are expected to reverse in the future was a net decrease related to deferred tax assets and deferred tax liabilities of $70.5 million, with a corresponding net adjustment to benefit from income taxes of $16.1 million and offsetting change in valuation allowance of $86.6 million for the year ended December 31, 2017. The Company does not expect a material impact related to the one-time transition tax on the mandatory deemed repatriation of foreign earnings.
On December 22, 2017, Staff Accounting Bulletin No. 118 (SAB 118) was issued to address the application of GAAP in situations when a registrant does not have the necessary information available, prepared, or analyzed (including computations) in reasonable detail to complete the accounting for certain income tax effects of the Act. In accordance with SAB 118, the Company has determined that the adjustment to deferred taxes was a provisional amount and a reasonable estimate at December 31, 2017. The determination of the benefit from (provision for) income taxes requires complex estimations, significant judgments and significant knowledge and experience concerning the applicable tax laws. Given that the Company is still in the transition period for the accounting for income tax effects of the Tax Act, the current assessment on deferred tax assets (liabilities) is based on the currently available information and guidance. If in the future any element of the tax reform changes the related accounting guidance for income tax, it could affect the Company’s income tax position and the Company may need to adjust the benefit from (provision for) income taxes accordingly.
The Company recorded unrecognized tax benefits for uncertainties in income taxes. A reconciliation of the Company’s unrecognized tax benefits for the years ended December 31, 2017, 2016, and 2015 is as follows (in thousands):
|
|
|
December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Balance at beginning of year |
|
$ |
13,505 |
|
|
$ |
24,010 |
|
|
$ |
7,275 |
|
|
Additions based on tax positions related to current year |
|
|
9,338 |
|
|
|
6,777 |
|
|
|
15,628 |
|
|
Additions for tax positions of prior years |
|
|
5,534 |
|
|
|
877 |
|
|
|
5,505 |
|
|
Reductions for tax positions of prior years |
|
|
— |
|
|
|
(18,159 |
) |
|
|
(4,398 |
) |
|
Balance at end of year |
|
$ |
28,377 |
|
|
$ |
13,505 |
|
|
$ |
24,010 |
|
The entire amount of the unrecognized tax benefits would not impact the Company’s effective tax rate if recognized. The Company has elected to include interest and penalties as a component of tax expense. During the years ended December 31, 2017 and 2016, the Company did not recognize accrued interest and penalties related to unrecognized tax benefits. The Company does not anticipate that the amount of existing unrecognized tax benefits will significantly increase or decrease during the next year.
F-25
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
It is our intention to reinvest the earnings of our non-U.S. subsidiaries in their operations. As of December 31, 2017, the Company had not made a provision for U.S. income taxes or foreign withholding taxes on a nominal amount of the excess of the amount of net income for financial reporting over the tax basis of investments in foreign subsidiaries that are essentially permanent in duration. Generally, such amounts become subject to U.S. taxation upon repatriation as dividends and under certain other circumstances. If these earnings were repatriated to the U.S., the deferred tax liability associated with these temporary differences would result in a nominal amount.
The Company files income tax returns in the U.S. federal, California, and other state tax jurisdictions. The federal and state income tax returns from inception to December 31, 2017 remain subject to examination.
12. Commitments and Contingencies
Facilities
The Company leases office space and research, testing and manufacturing laboratory space in various facilities in Novato and Brisbane, California and in Cambridge and Woburn, Massachusetts, under operating agreements expiring at various dates through 2026. Certain of the leases provide for options by the Company to extend the lease for multiple five-year renewal periods and also provide for annual minimum increases in rent, usually based on a consumer price index or annual minimum increases.
The Company recognizes rent expense on a straight-line basis over the noncancelable term of its operating lease. Rent expense was $4.5 million, $3.3 million, and $1.4 million during the years ended December 31, 2017, 2016 and 2015, respectively.
Other Commitments
The Company has various manufacturing, clinical, research, and other contracts with vendors in the conduct of the normal course of its business. Other than as noted below, contracts are terminable, with varying provisions regarding termination. If a contract with a specific vendor were to be terminated, the Company would only be obligated for the products or services that the Company had received at the time the termination became effective.
As of December 31, 2017, the aggregate future minimum lease payments under the noncancelable operating lease arrangements and future payments under contractually binding manufacturing and service agreements are as follows (in thousands):
|
Year Ending December 31, |
|
Leases |
|
|
Manufacturing and Services |
|
||
|
2018 |
|
$ |
6,210 |
|
|
$ |
7,542 |
|
|
2019 |
|
|
5,293 |
|
|
|
1,224 |
|
|
2020 |
|
|
3,727 |
|
|
|
62 |
|
|
2021 |
|
|
2,477 |
|
|
|
— |
|
|
2022 |
|
|
2,413 |
|
|
|
— |
|
|
Thereafter |
|
|
9,033 |
|
|
|
— |
|
|
|
|
$ |
29,153 |
|
|
$ |
8,828 |
|
Contingencies
While there are no material legal proceedings the Company is aware of, the Company may become party to various claims and complaints arising in the ordinary course of business. Management does not believe that any ultimate liability resulting from any of these claims will have a material adverse effect on its results of operations, financial position, or liquidity. However, management cannot give any assurance regarding the ultimate outcome of these claims, and their resolution could be material to operating results for any particular period, depending upon the level of income for the period.
Guarantees and Indemnifications
The Company indemnifies each of its directors and officers for certain events or occurrences, subject to certain limits, while the director is or was serving at the Company’s request in such capacity, as permitted under Delaware law and in accordance with its certificate of incorporation and bylaws. The term of the indemnification period lasts as long as a director may be subject to any proceeding arising out of acts or omissions of such director in such capacity. The maximum amount of potential future indemnification is unlimited; however, the Company currently holds director liability insurance. This insurance allows the transfer of risk associated with the Company’s exposure and may enable it to recover a portion of any future amounts paid. The Company believes that the fair value of these indemnification obligations is minimal. Accordingly, it has not recognized any liabilities relating to these obligations for any period presented.
F-26
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
The following table sets forth the computation of the basic and diluted net loss per share during the years ended December 31, 2017, 2016 and 2015 (in thousands, except share and per share data):
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Numerator: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss |
|
$ |
(302,139 |
) |
|
$ |
(245,874 |
) |
|
$ |
(145,618 |
) |
|
Denominator: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted-average shares used to compute net loss per share, basic and diluted |
|
|
42,453,135 |
|
|
|
39,586,908 |
|
|
|
36,782,603 |
|
|
Net loss per share, basic and diluted |
|
$ |
(7.12 |
) |
|
$ |
(6.21 |
) |
|
$ |
(3.96 |
) |
The following weighted-average outstanding common stock equivalents were excluded from the computation of diluted net loss per share for the periods presented because including them would have been antidilutive:
|
|
|
Year Ended December 31, |
|
|||||||||
|
|
|
2017 |
|
|
2016 |
|
|
2015 |
|
|||
|
Options to purchase common stock, RSUs, and PSUs |
|
|
5,862,784 |
|
|
|
4,699,111 |
|
|
|
3,368,507 |
|
|
Employee stock purchase plan |
|
|
2,728 |
|
|
|
7,933 |
|
|
|
— |
|
|
Common stock warrants |
|
|
149,700 |
|
|
|
149,700 |
|
|
|
195,762 |
|
|
|
|
|
6,015,212 |
|
|
|
4,856,744 |
|
|
|
3,564,269 |
|
14. Accumulated Other Comprehensive Income (Loss)
Total accumulated other comprehensive income (loss) consisted of the following (in thousands):
|
|
|
Unrealized Gain (Loss) on Securities Available-for-Sale |
|
|
Foreign Currency Translation Adjustments |
|
|
Accumulated Other Comprehensive Income (Loss) |
|
|||
|
December 31, 2014 |
|
$ |
(174 |
) |
|
$ |
— |
|
|
$ |
(174 |
) |
|
Current period other comprehensive loss |
|
|
(694 |
) |
|
|
— |
|
|
|
(694 |
) |
|
December 31, 2015 |
|
|
(868 |
) |
|
|
— |
|
|
|
(868 |
) |
|
Current period other comprehensive income |
|
|
451 |
|
|
|
1,322 |
|
|
|
1,773 |
|
|
December 31, 2016 |
|
|
(417 |
) |
|
|
1,322 |
|
|
|
905 |
|
|
Current period other comprehensive income (loss) |
|
|
35 |
|
|
|
(10,110 |
) |
|
|
(10,075 |
) |
|
Reclassification from accumulated other comprehensive income |
|
|
— |
|
|
|
3,490 |
|
|
|
3,490 |
|
|
December 31, 2017 |
|
$ |
(382 |
) |
|
$ |
(5,298 |
) |
|
$ |
(5,680 |
) |
15. Quarterly Financial Data (unaudited)
The following table presents certain unaudited quarterly financial information. This information has been prepared on the same basis as the audited financial statements and includes all adjustments (consisting only of normal recurring adjustments) necessary to present fairly the unaudited quarterly results of operations set forth herein (in thousands, except share and per share data):
|
|
|
2017 |
|
|||||||||||||
|
|
|
March 31, |
|
|
June 30, |
|
|
September 30, |
|
|
December 31, |
|
||||
|
Revenue |
|
$ |
— |
|
|
$ |
— |
|
|
$ |
198 |
|
|
$ |
2,414 |
|
|
Operating expenses |
|
|
69,954 |
|
|
|
78,441 |
|
|
|
83,911 |
|
|
|
99,248 |
|
|
Net loss |
|
|
(68,290 |
) |
|
|
(72,891 |
) |
|
|
(79,227 |
) |
|
|
(81,731 |
) |
|
Net loss per share, basic and diluted |
|
$ |
(1.63 |
) |
|
$ |
(1.72 |
) |
|
$ |
(1.87 |
) |
|
$ |
(1.89 |
) |
F-27
ULTRAGENYX PHARMACEUTICAL INC.
Notes to Consolidated Financial Statements (continued)
|
|
|
2016 |
|
|||||||||||||
|
|
|
March 31, |
|
|
June 30, |
|
|
September 30, |
|
|
December 31, |
|
||||
|
Revenue |
|
$ |
— |
|
|
$ |
17 |
|
|
$ |
111 |
|
|
$ |
5 |
|
|
Operating expenses |
|
|
53,622 |
|
|
|
58,070 |
|
|
|
65,894 |
|
|
|
70,554 |
|
|
Net loss |
|
|
(52,757 |
) |
|
|
(56,923 |
) |
|
|
(64,907 |
) |
|
|
(71,287 |
) |
|
Net loss per share, basic and diluted |
|
$ |
(1.35 |
) |
|
$ |
(1.46 |
) |
|
$ |
(1.64 |
) |
|
$ |
(1.75 |
) |
16. Subsequent Events
ATM Offering
During the period of January 1, 2018 through February 21, 2018, the Company sold an additional 240,417 shares of common stock, resulting in net proceeds of approximately $11.8 million, after commissions and other offering costs.
Priority Review Voucher sale
On January 10, 2018, the Company completed the sale of a PRV to Novartis Pharma AG for $130.0 million. The Company had received the PRV from the FDA in connection with the approval of Mepsevii.
Underwritten Public Offering
In January 2018, the Company completed an underwritten public offering in which 5,043,860 shares of common stock were sold, which included 657,895 shares purchased by the underwriters pursuant to an option granted to them in connection with the offering, at a public offering price of $57.00 per share. The total proceeds that the Company received from the offering were approximately $271.0 million, net of underwriting discounts and commissions.
