Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Recro Pharma, Inc. | d474247d8k.htm |
 October
2017 Relieving pain…….Improving lives
Exhibit 99.1 |
 Special
Note Regarding Forward-Looking Statements
This presentation includes forward-looking statements within the meaning of Section
27A of the Securities Act of 1933 and Section 21E of the Securities
Exchange Act of 1934. These statements, among other things, relate
to our business strategy, goals and expectations concerning our product
candidates, future operations, prospects, plans and objectives of
management. The words "anticipate", "believe", "could", "estimate", "expect", "intend", "may", "plan", "predict", "project", "will" and
similar terms and phrases are used to identify forward-looking
statements in this presentation. Our operations involve risks and
uncertainties, many of which are outside our control, and any one of
which, or a combination of which, could materially affect our results of
operations and whether the forward-looking statements ultimately prove to be
correct. This presentation is intended to be nonpromotional and for
investor discussion purposes only. The information provided herein
contains references to investigational products and use of these products
has not been approved by the FDA. The safety and efficacy of the
investigational use of these development products has not been
determined. There is no guarantee that the investigational use listed will be
filed with and/or approved for marketing by any regulatory agency.
These forward-looking statements should be considered together with
the risks and uncertainties that may affect our business and future
results included in our filings with the Securities and Exchange
Commission at www.sec.gov. These forward-looking statements are based on information currently available to us, and we assume no obligation to update any
forward-looking statements except as required by applicable law.
2 |
 Company
Highlights 3
• Specialty pharmaceutical company focused on hospital and related settings with late stage investigational
product, IV Meloxicam, targeting management of moderate to severe pain
• Filed New Drug Application for IV Meloxicam in July 2017; Received FDA acceptance for review in September
2017 (PDUFA Date-
May 26, 2018) – Reported results for pivotal Phase 3 trial in patients following abdominoplasty surgery
– Reported results for pivotal Phase 3 trial in patients following bunionectomy surgery
– Reported results for Phase 3 safety study demonstrating solid safety and tolerability profile
– Completed renal impairment and ECG studies • Multiple therapeutics in clinical development for hospital and related settings
– Non-opioid, alpha 2 agonist for peri-procedural use; – Recent acquisition of Neuromuscular Blockers (NMB) and related reversal agents from Cornell University
• Used for rapid induction and reversal of neuromuscular blockade • Revenue and cash flow positive contract development and manufacturing (CDMO) business
– Raised Revenue and EBITDA Guidance, August 2017 • Solid cash position- $50.2 million cash @ 6/30/17 • Experienced management team with significant development, regulatory and commercial experience |
 Experienced
Management and Board •
Gerri Henwood – President and CEO Founded Recro Pharma (REPH, NASDAQ), Auxilium Pharmaceuticals (AUXL, NASDAQ) and IBAH (former NASDAQ Co. – acquired 1998); GSK • Michael Celano – CFO Over 35 years of financial leadership experience – Kensey Nash, BioRexis, Orasure, Arthur Andersen/KPMG • Randy Mack – SVP, Development Over 25 years of clinical development experience – Adolor, Auxilium, Abbott Labs and Harris Labs • Stewart McCallum, MD – CMO Over 9 years of GSK Clinical experience – Development experience; past clinical Investigator, KOL and Stanford U. • Fred Graff – CCO Over 30 years of successful commercial experience, including sales and marketing leadership roles at Sepracor, RPR, and MAP Pharmaceuticals • Scott Rizzo- General Manager, Gainesville CDMO Over 25 years of operations experience – Roche Pharmaceuticals, Barrier Therapeutics, PwC, E.I. DuPont Board of Directors Wayne B. Weisman – Chairman SCP VitaLife Partners Alfred Altomari CEO, Agile Therapeutics; former J&J
William L. Ashton
Harrison Consulting Group; frmly Amgen Michael Berelowitz, M.D. Former SVP, Specialty Care Business Unit, Pfizer Winston J. Churchill SCP VitaLife Partners Karen Flynn Senior Vice President and Chief Commercial Officer of West Pharmaceutical Services, Inc. Gerri Henwood – CEO Bryan Reasons SVP and CFO, Impax Laboratories 4 |
      Recro Pharma: An Acute Care Company (REPH:NASDAQ) 5 |
 Contract Manufacturing
Acute Care Division
Our Two Divisions 6 6 |
 Acute Care
Clinical Stage Pipeline Investigational Product
PC I II III Rights Meloxicam WW IV formulation Acute post operative pain Filed NDA IM formulation Acute pain Dexmedetomidine
(“Dex”) WW, exc. Europe, Turkey, CIS Dex-IN (intranasal) Peri-procedural pain Phase 2 Cancer breakthrough pain Neuromuscular Blockers (NMB) (Anesthesia) WW IV Intermed – action (RP1000) IV Ultra-short action (RP2000) 7 NMB Reversal (Anesthesia) WW RP3000 |
 Post Op
Pain Market Underserved •
$5.9 billion market (1) • Predominantly opioid use (1) • Significant side effects / issues associated with opioids (2) • Dearth of non-opioid drugs in development Inpatient procedures Total procedures (2009) 47.9M Addressable >25M Ambulatory procedures Total procedures (2006) 53.3M Addressable >25M Note: Addressable includes procedures expected to utilize pain medication. Source: National Center for Health Statistics and management estimates. (1) GBI Research, 2010 sales. (2) Benyamin et al. 2008. “Opioid Complications and Side Effects.” Pain Physician 11 (SPEC. ISS. 2):
S105–20
8 |
 Limited
Acute Pain Relief Options for Patients *Pain severity based upon market
research / physician feedback Pain
Severity* Class Compounds Advantages Disadvantages Mild Acetaminophen Antipyretic properties; Oral; no opioid AEs Only effective for mild pain; short acting NSAIDs Ketorolac, ibuprofen, aspirin Mild to moderate analgesia; oral; no opioid AEs Bleeding risk; GI and renal complications; short acting Moderate Sodium channel blockers Bupivacaine, lidocaine Use directly at pain site; mostly peri-operative Limited duration of action; some are concerned about local tissue impact Alpha 2 agonists Dexmedetomidine (Recro Pharma) Target pain relief; anxiolytic properties; no respiratory depression, impaired GI or addictive properties In development – potential to be approved for peri-procedural pain; blood pressure decrease Moderate to Severe Non-opioid, once a day preferential COX-2 IV/IM meloxicam (Recro Pharma) q24 hour dosing; Both Ph. 3 pivotal studies met primary endpoints; MOA- no impact on GI motility; well tolerated Class effects: Bleeding risk; GI and renal complications. Opioids Morphine, hydrocodone, oxycodone, fentanyl Good pain relief Respiratory depression, impaired GI motility after even one dose; frequent nausea and vomiting; abuse/addiction potential 9 |
 IV
Meloxicam |
 IV
Meloxicam Overview •
FDA approved as an oral preferential COX-2 inhibitor and used in a number
of indications
• Proprietary non-opioid, once daily injectable form for management of moderate to severe pain – Incorporates Alkermes’ NanoCrystal ® technology • IV Meloxicam – once daily preferential COX-2 inhibitor for moderate to severe acute pain – Multiple Phase 2 studies met primary endpoints in acute pain models – Phase 3 efficacy trial in abdominoplasty met the primary endpoint – Phase 3 efficacy trial in bunionectomy met the primary endpoint – Phase 3 safety study demonstrated solid safety and tolerability profile – Completed renal impairment and ECG studies – Filed New Drug Application in July 2017; Received FDA acceptance for review in
September 2017 (PDUFA Date-
May 26, 2018) • Formulation IP issued through 2022 and additional methods of preparation IP issued through May 2030 NanoCrystal ® is a registered trademark of APIL 11 |
 Phase 3
Safety Study Reported Q2 2017 •
Randomized, Double-blind, Placebo-controlled Safety
Study – Subjects requiring major surgery that was expected to result in inpatient hospitalization for at least 24-48 hours • 721 subjects at 31 centers in 4 countries treated with: – Placebo – IV Meloxicam 30 mg • Study drug administration – Added to existing pain management protocol after surgery (No minimum pain score required for randomization) – Once daily for up to 7 doses – Additional dose at discharge was at the discretion of subject and investigator. 12 |
 Phase 3
Safety Study- Endpoints
• Incidence of AEs and SAEs • Change from baseline in laboratory tests; incidence of abnormal clinical laboratory tests • Change from baseline in vital signs; incidence of clinically significant changes in vital signs • Incidence of clinically significant abnormal ECG findings • Incidence of abnormal wound healing • Opioid consumption over 48 hours 13 |
 Phase 3
Safety Study- Subject Demographics
Overall
Variable
(N=721)
Mean (Range) Age (yrs)
53 (18-80)
>=65 years: n (%)
163 (22.6)
Male: n
(%)
293 (40.6)
Mean (SD) BMI (kg/m
2
)
29.4 (4.93)
>=35 kg/m
2
: n (%)
111 (15.4)
Surgery: n (%)
Abdominoplasty
43 (6.0)
Gynecologic Surgery
89 (12.3)
GI Surgery
39 (5.4)
Soft Tissue Surgery
170 (23.6)
Total Ankle Replacement
1 (0.1)
Total Shoulder Replacement
8 (1.1)
Total Hip Replacement
68 (9.4)
Total Knee Replacement
156 (21.6)
Orthropedic Trauma
1 (0.1)
Complex Foot
71 (9.8)
Bunionectomy
53 (7.4)
Spinal
13 (1.8)
Head And Neck
1 (0.1)
14 |
 15 % of Subjects IV Meloxicam 30 mg Placebo Adverse Event N = 538 N = 183 Subjects with an SAE 2.6 5.5 Subjects with >=1 AE 63.0 65.0 Nausea 22.9 27.9 Constipation 9.5 9.3 Vomiting 5.0 7.7 Pruritis 3.9 5.5 GGT Increased 3.9 2.7 Headache 3.7 6.6 Anemia 3.3 2.2 Phase 3 Safety Study Adverse Events – 3% in the IV Meloxicam Group |
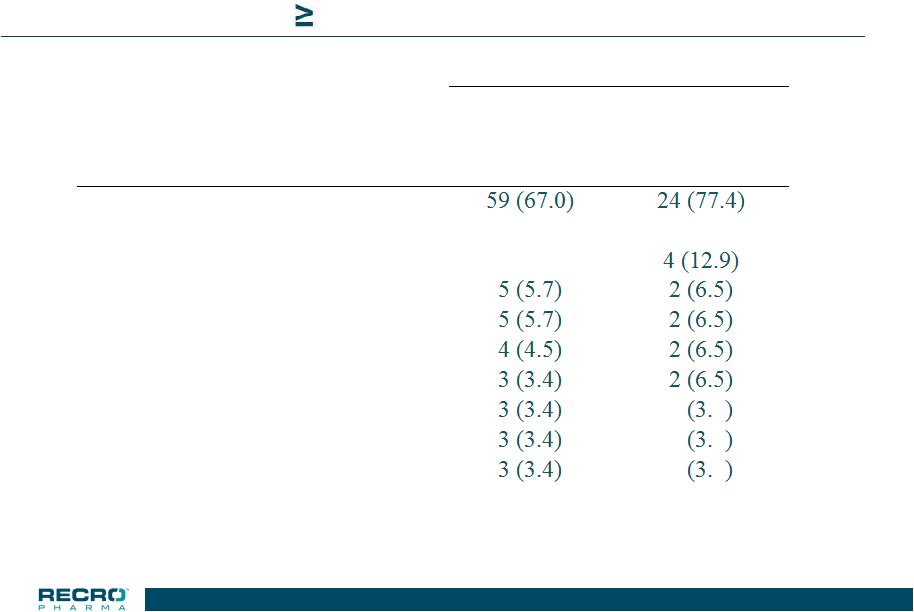 16 Phase 3 Safety Study >65 years & Mild Renal Impairment Adverse Events – 3% in the IV Meloxicam Group n (%) of Subjects IV Meloxicam 30 mg Placebo Adverse Event N = 88 N = 31 Subjects with >=1 AE Nausea 17 (19.3) 8 (25.8) Constipation 16 (18.2) Anemia Vomiting Pruritus GGT increased Pyrexia 1 2 Urinary Tract Infection 1 2 Postoperative Fever 1 2 16 |
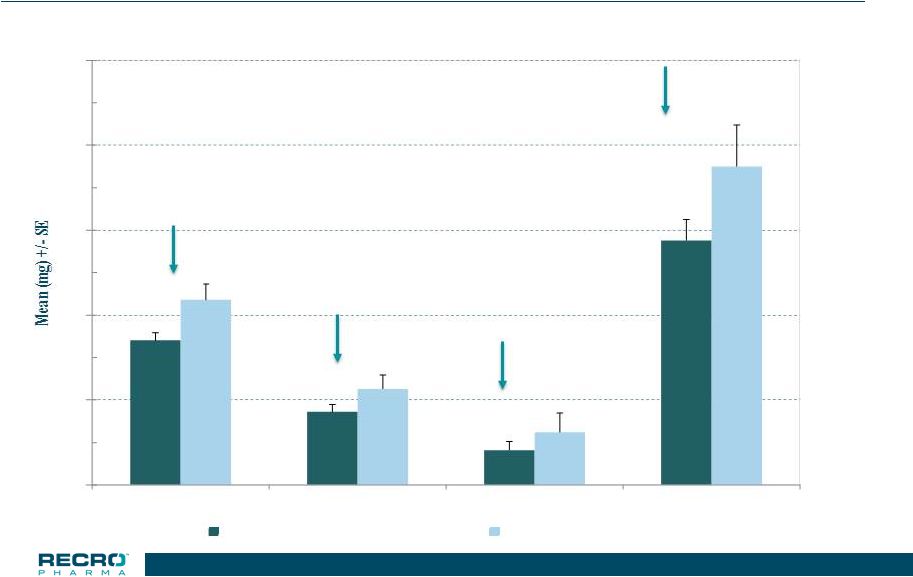 17 Phase 3 Safety Study – Total Population Total Opioid Used (in morphine equivalents) *p<0.05 22.0% * 23.9% 33.9% * 23.2% * 0 10 20 30 40 50 0-24 HR 24-48 HR 48-72 HR Treatment Period IV Meloxicam 30 mg (n=538) Placebo (n=183) 17 |
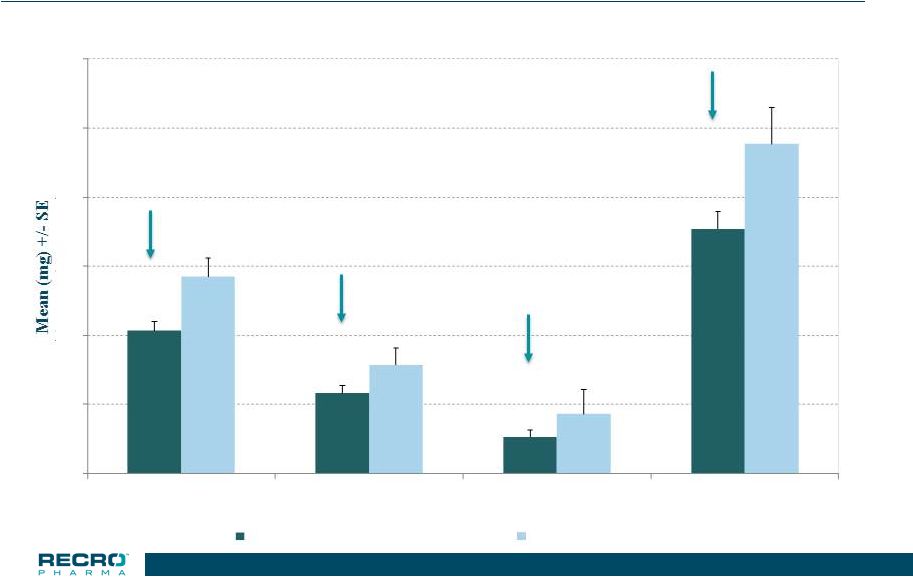 18 Phase 3 Safety Study – Orthopedic Surgeries Total Opioid Used (in morphine equivalents) *p<0.05 27.4% * 26.1% * 38.4% * 25.8% * 0 10 20 30 40 50 60 0-24 HR 24-48 HR 48-72 HR Treatment Period IV Meloxicam 30 mg (n=283) Placebo (n=96) |
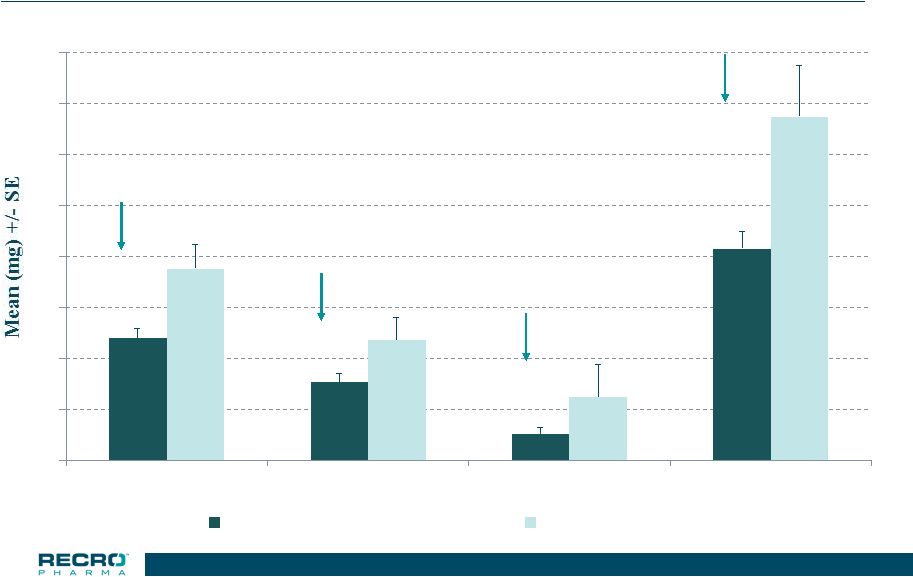 19 0 10 20 30 40 50 60 70 80 0-24 HR 24-48 HR 48-72 HR Treatment Period IV Meloxciam 30 mg (n=117) Placebo (n=39) Phase 3 Safety Study – Total Knee Replacement Total Opioid Used (in morphine equivalents) 36.4% 35.2% *p<0.01 * * * 58.9% 38.4% 19 |
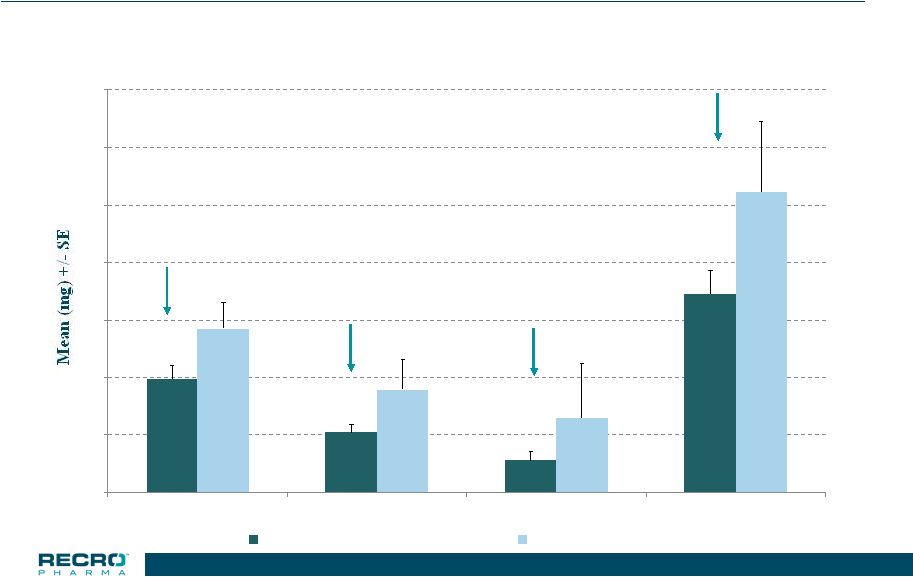 20 Phase 3 Safety Study >65 years & Mild Renal Impairment Total Opioid Used (in morphine equivalents) *p<0.05 0 10 20 30 40 50 60 70 0-24 HR 24-48 HR 48-72 HR Treatment Period IV Meloxicam 30 mg (n=88) Placebo (n=31) 30.5% 41.9% * 56.9% 33.8% 20 |
 Phase 3
Abdominoplasty Study •
Multicenter, Multi-dose, Randomized, Double-blind,
Placebo-controlled
• 219 subjects randomized to IV Meloxicam 30mg or Placebo – Study medication administered once daily up to 3 doses – 98% of subjects completed the 48 hour assessments • Standard analgesia design – Pain Intensity assessments (SPID24 = Primary Endpoint) – Use of rescue medication – Time to onset – Patient Global Assessment of Pain Control 21 |
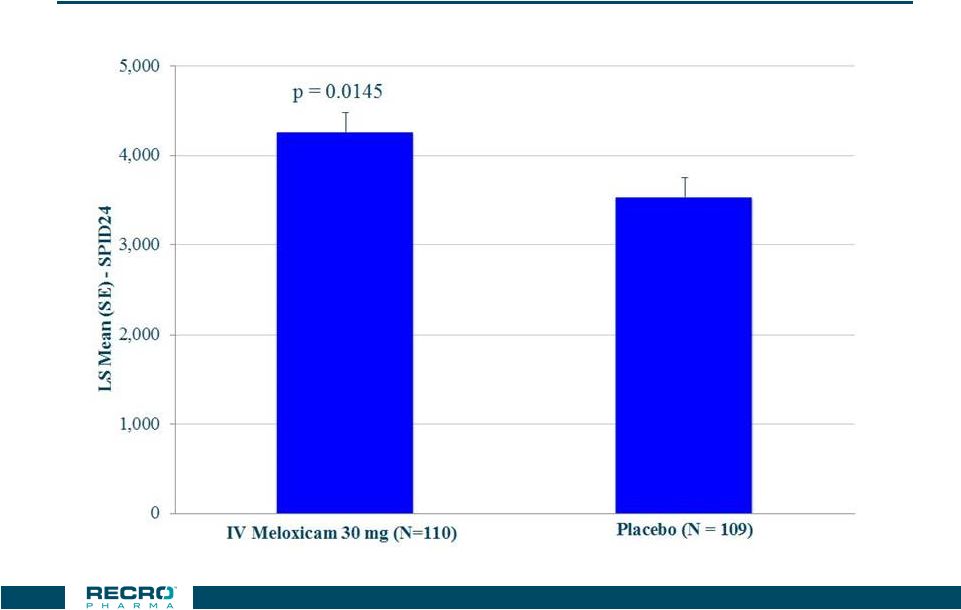 Phase 3
Abdominoplasty Study Primary Endpoint –
SPID24* 22 * W2LOCF |
 Phase 3
Abdominoplasty Study Summary of Secondary Endpoints
SPID6, Times to Meaningful Pain Relief and First
Rescue, Number of Subjects
rescued
0-24
and
0-48
hours,
%
Subjects
with
>30
and
>50%
Improvement
within
6
Hours
and
>50%
within
24
hours,
PGA
of
Pain
Control at 24 hours were not significantly different
between treatment groups.
Parameter
Met criteria for Statistical
Significance:
SPID12
SPID48
SPID24-48
Number of Subjects Rescued 24-48 Hours
Number of Times Rescued 0-24 Hours
Number of Times Rescued 24-48 Hours
Number of Times Rescued 0-48 Hours
Time to Perceptible Pain Relief
%
Subjects
with
>30%
Improvement
-
24
Hours
PGA of Pain Control at 48 hours
23 |
 Phase 3
Abdominoplasty Study Adverse Events –
2% in the IV Meloxicam group
24 n (%) of Subjects IV Meloxicam 30 mg Placebo Preferred Term (N=110) (N=109) Subjects with >=1 TEAE 58 (52.7) 80 (73.4) Nausea 30 (27.3) 41 (37.6) Headache 13 (11.8) 18 (16.5) Vomiting 5 ( 4.5) 10 ( 9.2) Dizziness 4 ( 3.6) 10 ( 9.2) ** Four (4) subjects experienced Serious Adverse Events during this
study. Three subjects were randomized to placebo
and one to IV Meloxicam.
|
 Phase 3
Bunionectomy Study •
Multicenter, Multi-dose, Randomized, Double-blind,
Placebo-controlled
• 201 subjects randomized to either IV Meloxicam 30 mg or Placebo – Study medication administered once daily up to 3 doses – 95% of subjects completed the 48 hour assessments • Standard analgesia design – Pain Intensity assessments (SPID48 = Primary Endpoint) – Use of rescue medication – Time to onset – Patient Global Assessment of Pain Control 25 |
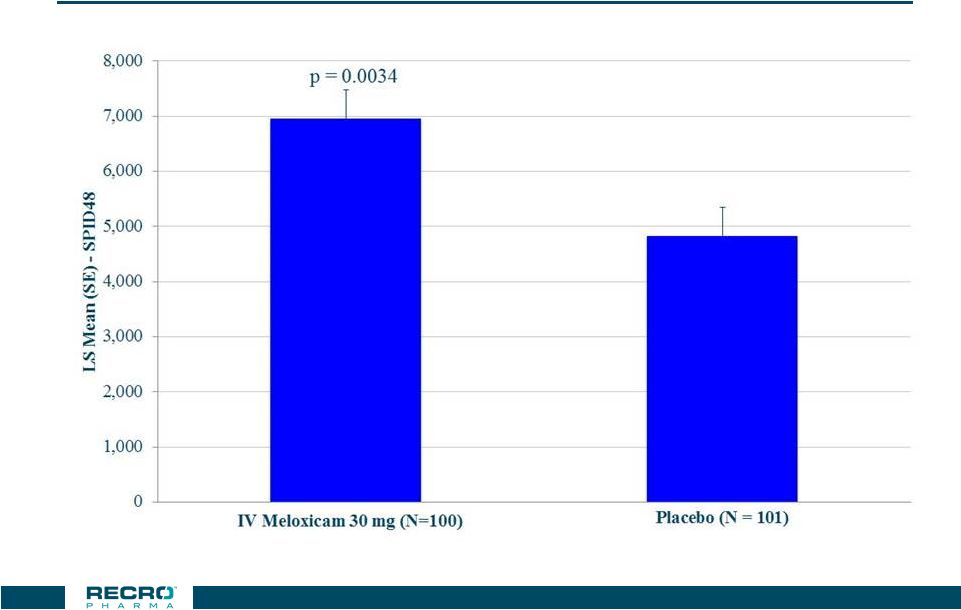 Phase 3
Bunionectomy Study Primary Endpoint –
SPID48* 26 * W2LOCF |
 Phase 3
Bunionectomy Study Summary of Secondary Endpoints
Parameter
Met criteria for Statistical
Significance:
SPID6
SPID12
SPID24
SPID24-48
Time to First Rescue Analgesia
Number of Subjects Rescued 0-24 Hours
Number of Subjects Rescued 24-48 Hours
Number of Subjects Rescued 0-48 Hours
Number of Times Rescued 0-24 Hours
Number of Times Rescued 24-48 Hours
Number of Times Rescued 0-48 Hours
%
Subjects
with
>30%
Improvement
-
6
Hours
%
Subjects
with
>30%
Improvement
-
24
Hours
%
Subjects
with
>50%
Improvement
-
24
Hours
PGA of Pain Control at 48 hours
Times to Perceptible and Meaningful Pain Relief, %
Subjects with >50% Improvement within
6 Hours, PGA of Pain Control at 24 hours
were not significantly different between treatment groups.
27 |
 Phase 3
Bunionectomy Study Adverse Events –
3% in either group 28 n (%) of Subjects IV Meloxicam 30 mg Placebo Preferred Term (N=100) (N=101) Subjects with 1 TEAE 44 (44.0) 54 (53.5) Nausea 20 (20.0) 26 (25.7) Headache 8 (8.0) 12 (11.9) Vomiting 3 (3.0) 9 (8.9) Pruritus 8 (8.0) 3 (3.0) Decreased appetite 2 (2.0) 7 (6.9) Constipation 4 (4.0) 5 (5.0) Abdominal pain -- 6 (5.9) Dizziness 3 (3.0) 4 (4.0) Flushing 3 (3.0) 1 (1.0) Somnolence 3 (3.0) 2 (2.0) ALT increased -- 3 (3.0) **Two (2) subjects experienced Serious Adverse Events during this
study. Both subjects were randomized to
placebo. |
 Next Steps
for IV Meloxicam •
Filed NDA for IV Meloxicam in July 2017; Received FDA acceptance for
review in September 2017 (PDUFA Date-
May 26, 2018) • Total across NDA: approximately 1400 meloxicam patients – Largest Phase 3 double blind placebo controlled non-opioid study evaluating post operative IV pain product in a post-operative setting • Finalize and initiate Phase 3B studies – Orthopedic surgery (total knee)- opioid consumption/pain intensity – Colorectal surgery-opioid consumption; length of stay • Execute Commercialization and Launch Plan – Strategic Segmentation, Prioritization and Targeting to Accelerate Formulary Adoption 29 |
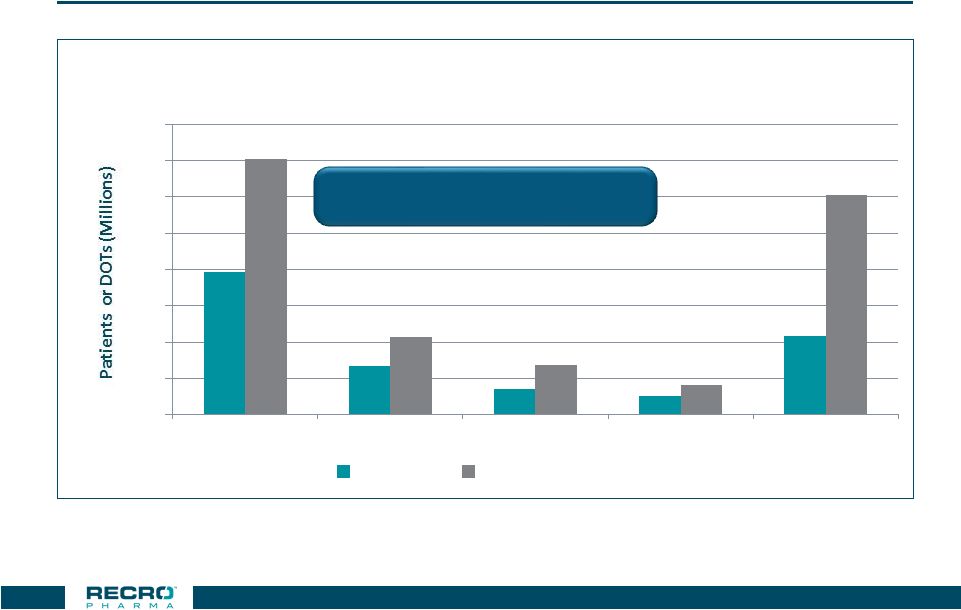 Opioids
Dominate the IV Pain Market 3.9
1.3 0.7 0.5 2.2 7.1 2.1 1.4 0.8 6.1 0 1 2 3 4 5 6 7 8 Opioids Local Anesthetics Ketorolac Ofirmev Any Drug Number of Inpatients Treated & DOT For Top 10 Surgical Procedures Treated
With Selected IV Analgesics
Patients Days Of Therapy 30 Note: Patients may be treated with more than one analgesic during a given DRG
Source: Truven Health Analytics 2014
Demonstrate clinical and economic
value versus IV opioids |
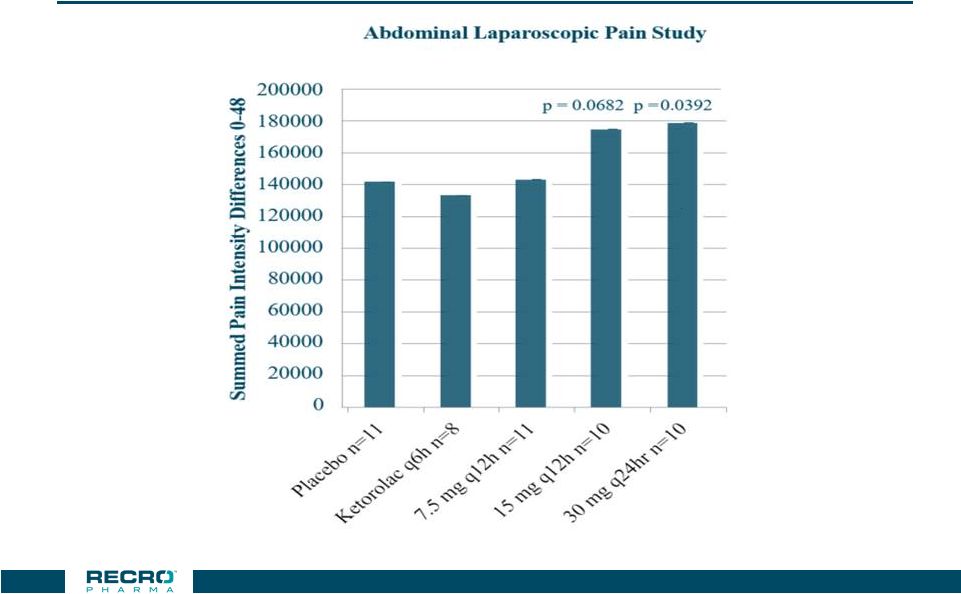 Efficacy
in Double Blind Laparoscopic Abdominal Surgery compared to Ketorolac and to
Placebo *** p < 0.001 vs.
Placebo
CONFIDENTIAL
31 |
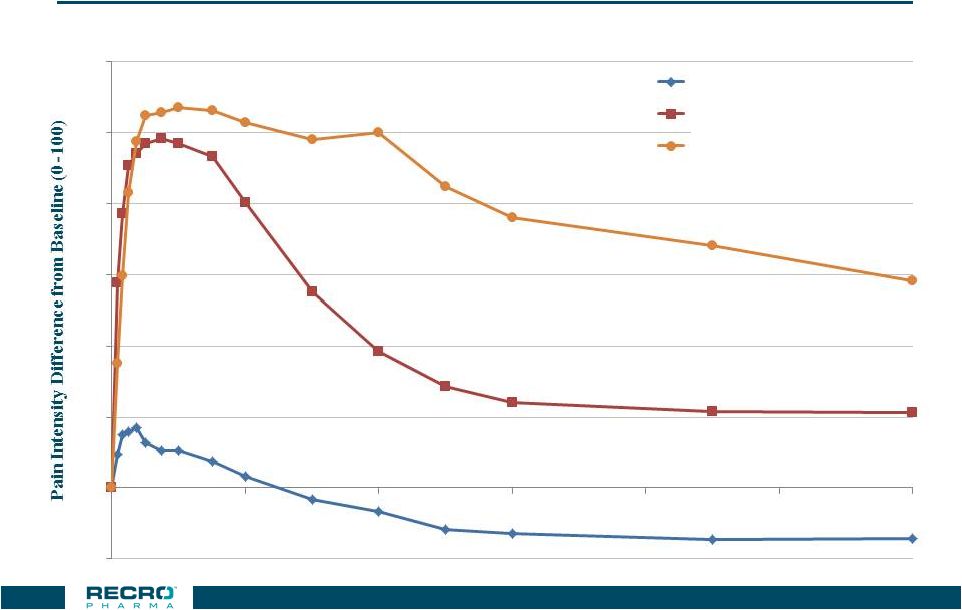 Peak
Analgesic Effects of IV Meloxicam and Morphine
32 Single 30mg Dose Performance over 24 hrs (Abdominal Hysterectomy Trial – IV Meloxicam vs IV Morphine) Baseline Pain Level ~60 -10 0 10 20 30 40 50 60 0 4 8 12 16 20 24 Time (Hours) Placebo n=64 Morphine n=62 IV Meloxicam 30 mg n=60 |
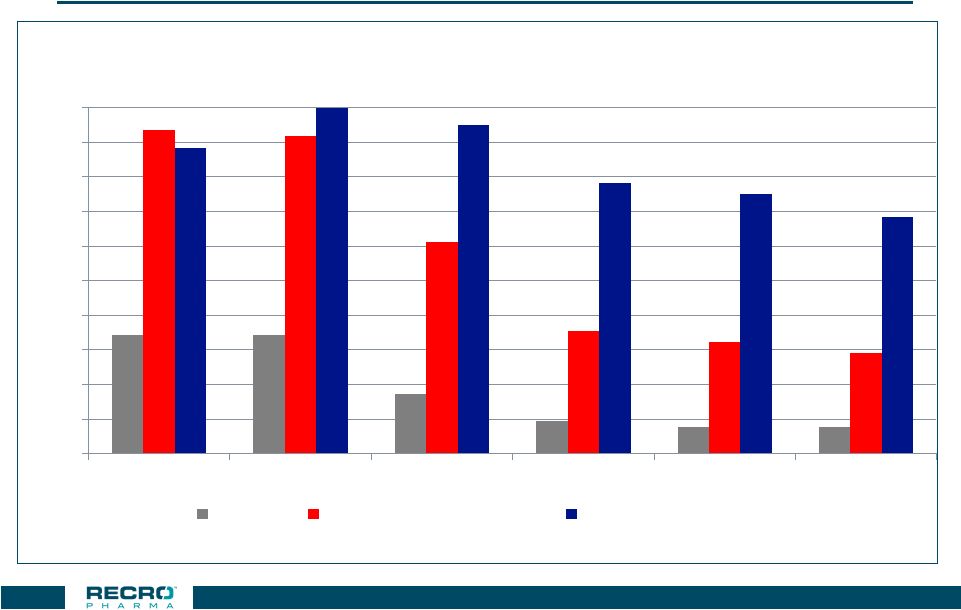 Length of
Analgesic Effects of IV Meloxicam and Morphine
33 34% 34% 17% 9% 8% 8% 94% 92% 61% 36% 32% 29% 88% 100% 95% 78% 75% 68% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% 0.5 hour 1 hour 6 hour 12 hour 18 hour 24 hour Placebo IV Morphine 0.15 mg/kg IV Meloxicam 30 mg Responder rate based on >30% improvement in pain intensity scores in moderate/severe patients
following open hysterectomy
Each medication given as a single dose
LOCF Analysis
Phase 2 Open Hysterectomy Study
|
 IV
Meloxicam Target Opportunity Intra-abdominal Procedures
• Surgeons have keen interest in: – Avoidance of opioids – Avoidance of troughs in existing non-opioid pain med options • If approved, could answer needs through: – Relief of moderate to severe pain over 24 hours – Reducing LOS 34 Orthopedic Procedures • Surgeons have keen interest in: – Long lasting serious pain relief to get the patient through the trip home and first post-op day/night – Many surgeons concerned about opioid use and potential for addiction • If approved, could answer needs through: – Relief of moderate to severe pain over 24 hours – No addiction properties |
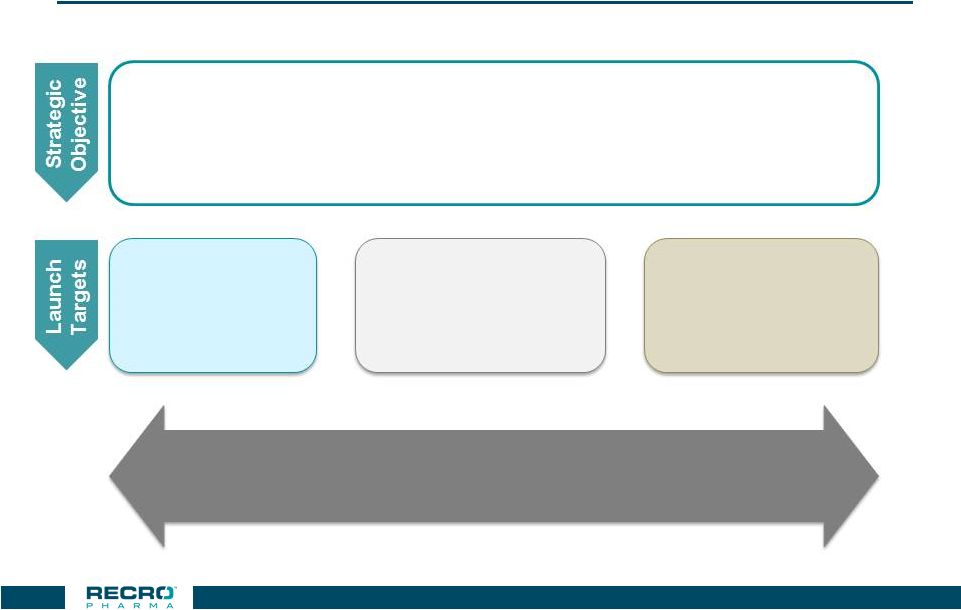 Ambulatory
Surgical Centers Offer Opportunity for High Volume Surgeons To Gain Early
Meloxicam Experience Experience with IV Meloxicam:
Build market experience with IV Meloxicam at settings that
have lower barriers to adoption and appetite for
experimentation Hospital Outpatient Settings Hospital Inpatient Settings Ambulatory Surgical Center Setting High Performing Surgeons That Practice at All Locations Early trials may carry back and drive adoption at Community and Academic Hospitals 35 CONFIDENTIAL |
 Long-Lasting Opioid Sparing Option is Perceived
Important Across All Settings of Care
36 Segment Size Summary ASC ~2.2M Patients Opioid sparing option reduces physician risk, facilitates same day discharge with improved patient experience and 24 hour pain control. Expand surgery repertoire by enabling more complex surgeries to be performed and discharged on the same day at ASCs. Swift formulary decision-making Healthy margin, limited by level of reimbursement per case HOPD ~9M Patients Opioid sparing option reduces physician risk, facilitates on-time or early discharge with improved patient experience and 24 hour pain control. Healthy margin on average, burdened by hospital-wide pharmacy budget
Slow formulary decision-making, but shared across multiple
settings Inpatient
~11M Patients Opioid sparing option with 24 hour efficacy remains appealing Reduction in length of stay remains important but less compelling because benefit masked by operational efficiencies of the staff Slow formulary decision-making, but shared across multiple settings
Greater institutional inertia to impact change
Source: Primary market research with customer interviews and
analysis |
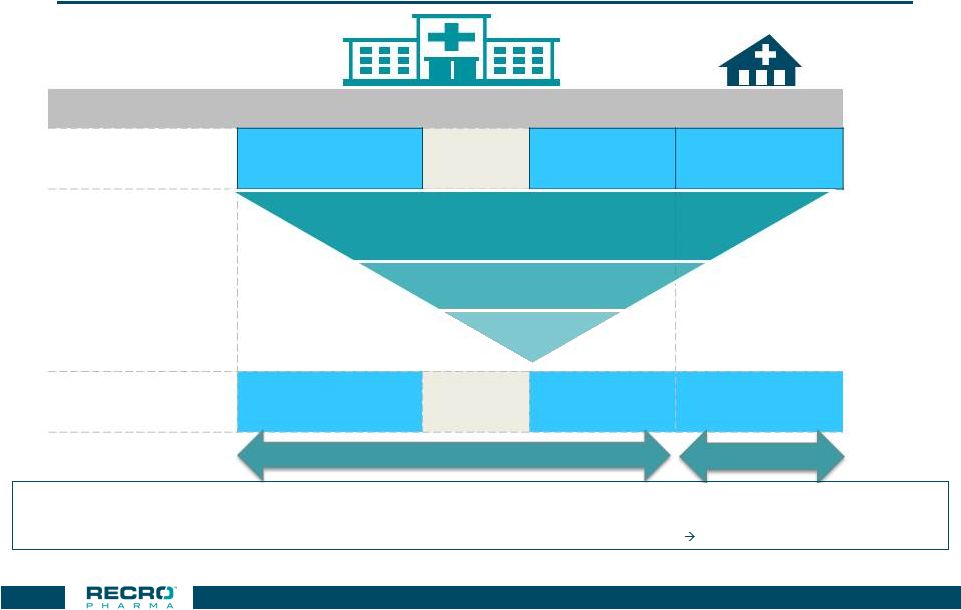 Targeted
Accounts Cover ~12.6M Patients Across All Settings Of Care
37 Hospital Inpatient ER HOPD ASC Total Addressable Procedures 11m 6.9m 9m 2.2m ~29m Core Target Procedures Targeted Accounts 3.7m 0.5m 7.2m 1.2m ~12.6m Patient opportunity by setting of care Source: Definitive HC (DRG and CPT#) –
Medicare; Projected to US All Payer @ 3.5X for ED, 4X for HOPD & ASC;
Claims / Patient – LexisNexis: ASC 3X; HOPD/ED 1.2
Notes:
• Addressable DRGs exclude DRGs where Meloxicam may be inappropriate (e.g. ObGyn, cardiovascular); Core Target DRGs are high volume Ortho,
GI, General Surgery procedures •
ER procedures include many medical uses (e.g. back pain) where there is no
surgical procedure; at launch ER utilization is considered spillover from other hospital settings • Addressable HOPD/ASC procedures exclude colonoscopies, eye procedures and focus on Ortho, GI and General Surgery procedures
nearly all addressable procedures are targeted
1,475 Hospitals
1,475 Hospitals
~570 ASCs ~570 ASCs Orthopedic (Hip/Knee, Spine, other) General Surgery GI/Colorectal |
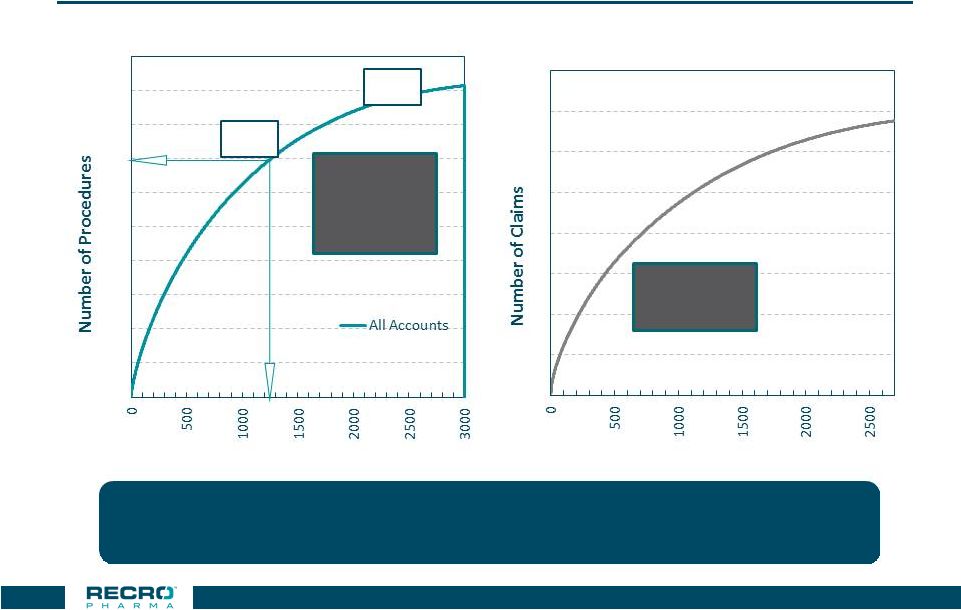 38 SF Size Of 80-100 Reps Provides 80% Coverage Of Key Procedures, Institutions, Key Surgeons & Top ASCs Source: Definitive HC (2015-20161H) 80% 95% 1,475 Institutions 3.8M Procedures ~570 ASC ~3.5m claims ~1.2m patients Further ASC & IDN Data analysis in addition to pre-launch account profiling activity will
lead to refinements in ASC targeting
0 0.5 1 1.5 2 2.5 3 3.5 4 4.5 5 Concentration Curve For US Hospitals, Procedure Volume 0 1 2 3 4 5 6 7 8 Concentration Curve For ASC, Estimated All Payer Claim Volume Number of Hospitals Number of ASCs |
 NMB Compounds from Cornell - Background • NMBs stands for a Neuromuscular Blocking
Agents: (Used as an “anesthetic” agent to facilitate intubation and surgery) • NMBs are classified by their onset time and duration of activity * Predicted based on currently available data
**supplemental pre-clinical
work required before next clinical trial at higher dose Compound Onset Time Duration of Activity Status Intermediate Acting* RP1000 Rapid* Intermediate Acting* Phase I** Ultra-Short Acting* RP2000 Rapid* Ultra-short acting*
Pre IND • Reversal agent specific to licensed compounds; acts quickly by a chemical
reaction to reverse blockade
• Potential for same dose of reversal agent to be used independent of time of
administration
• Reversal agent may require further refinement formulation 39 |
 NMB
– What is the clinical need?
• Novel NMBs and related agents with the potential for rapid onset with ultra-
short and intermediate durations of action, along with the potential to be
rapidly reversed
• Clinical potential: reducing the time required for induction of anesthesia and
the time needed to recover from NMB that patients require post procedure
(in the OR and in the PACU) while potentially enhancing patient safety.
• Current alternatives when patients need “reversing” from neuromuscular
blockade: i. encapsulating/binding reversal agents (Sugammadex) ii. enzymatic reversal agents (Neostigmine) – Both pathways may require higher doses and therefore incur greater expense if used earlier in a procedure compared to late in a procedure • (i.e., when more NMB agent is remaining in the patient’s system) • However, the proposed reversal agent used to reverse RP1000 and RP2000, may more quickly reverse neuromuscular blockade via a chemical reaction – This outcome could be beneficial to the patient and result in meaningful cost savings for the hospital/ASC 40 |
 Contract Development and Manufacturing (CDMO)
Business Overview Gainesville |
 42 Gainesville CDMO Facility Gainesville, GA, Facility – Located on 148 acres of land, the facility has approximately 97,000 square feet of space – Plant began operations early 90’s – Includes 288 pallet vault built in accordance with DEA specifications – Operates under cGMP standards – Two level facility – FDA/EMA/DEA Licensed – Development, Scale-up and full scale manufacturing – Manufactures product for U.S., European and Asian markets – Solid oral-dosage forms capacity – Capsule capacity- 500-700 million capsules – Tablet capacity- 1.5 billion tablets 42 |
 CDMO
Overview CDMO facility
• 97,000 + sq. ft. solid oral dosage manufacturing cGMP • DEA licensed; ~185 employees • Revenues include product sales, royalties and profit sharing • Positive cash flow providing debt service and non-dilutive financing source for Company development and operating activities Service capabilities • Formulation, process development and optimization • Process scale-up • Clinical supply and validation • Commercial supply Ritalin LA • Once daily ADHD treatment marketed by Novartis Focalin XR • ADHD treatment marketed by Novartis Verapamil/Verelan • CV/High blood pressure treatment marketed by Teva and Lannett Zohydro ER • Extended release hydrocodone marketed by Pernix • Launched in 2014 • Abuse deterrent form launched 43 |
 Solid
Oral Dosage Form Capabilities 44
• Site currently configured to produce capsules at commercial scale • Two commercial MG2 encapsulators each offering capacity of 125mm doses • Capsule range of fill sizing 00 to 3 • ~400K/Hour Fette tablet press • Coater in process Commercial-scale Oral Dosage Capabilities Development-scale Oral Dosage Capabilities • Capability to supply capsules and tablets at development- and clinical-scale manufacturing • Bosch GKF-400 encapsulation machine • Korsch XL100 Pro tablet press |
 2017
Additional Capabilities-Commercial Tablet Press and Film Coating
Equipment Fette
FE55 LBB Bohle BTC 100 45 |
 Packaging
Operations 46
• One line operation with annual maximum capacity of 2.5 million bottles per shift • Capability of packaging various size capsules and tablets • Can handle round or square bottles 40cc – 500cc, and counts of up to 500 units/bottle • Met serialization requirements guidance early |
 Strong
CDMO Business Performance •
Revenues include product sales, royalties and profit sharing
– 2016 revenues included one-time revenues; customer ordering patterns are not always predictable
– 2017 Guidance- Revenues of approximately $60-$63M, and EBITDA* of approximately $20-$23M
• Additional capacity for new product opportunities • Positive cash flow for debt service obligations, as well as cash flow to contribute to the funding of
Company operating activities, including product development, and
commercialization. *EBITDA and Excess Cash Flow are non-GAAP financial measures
(see reconciliation on last page of presentation) CDMO
Segment ($millions)
Year Ended December 31, 2016 (unaudited) Six Months Ended June 30, 2017 (unaudited) Revenues $69.3 $35.7 CDMO Segment Operating Income $24.2 $10.3 EBITDA* $31.8 $14.0 Excess Cash Flow (before interest & principal payments) * $30.5 $11.9 47 |
 Company
Highlights 48
• Specialty pharmaceutical company focused on hospital and related settings with late stage investigational
product, IV Meloxicam, targeting management of moderate to severe pain
• Filed New Drug Application for IV Meloxicam in July 2017; Received FDA acceptance for review in September
2017 (PDUFA Date-
May 26, 2018) – Reported results for pivotal Phase 3 trial in patients following abdominoplasty surgery
– Reported results for pivotal Phase 3 trial in patients following bunionectomy surgery
– Reported results for Phase 3 safety study demonstrating solid safety and tolerability profile
– Completed renal impairment and ECG studies • Multiple therapeutics in clinical development for hospital and related settings
– Non-opioid, alpha 2 agonist for peri-procedural use; – Recent acquisition of Neuromuscular Blockers (NMB) and related reversal agents from Cornell University
• Used for rapid induction and reversal of neuromuscular blockade • Revenue and cash flow positive contract development and manufacturing (CDMO) business
– Raised Revenue and EBITDA Guidance, August 2017 • Solid cash position- $50.2 million cash @ 6/30/17 • Experienced management team with significant development, regulatory and commercial experience |
 Reconciliation of Non-GAAP Financial Measures
(unaudited) CDMO Segment
($’s in millions)
2016 2017 Six Months 2017 Estimate Operating Income $24.2 $10.3 $12.6 - $15.6 Depreciation $5.0 $2.4 $4.8 Amortization of intangible assets $2.6 $1.3 $2.6 EBITDA $31.8 $14.0 $20.0 - $23.0 Capital expenditures ($4.3) ($2.5) ($4.8) Change in working capital $2.2 ($0.1) $1.8 Stock-based compensation expense $0.8 $0.5 $1.0 Excess Cash Flow (before interest & principal payments) $30.5 $11.9 $18.0 - $21.0 To supplement our financial results determined by U.S. generally accepted accounting principles ("GAAP"), we have also
disclosed in the table below the following non-GAAP information for our Contract
Development and Manufacturing division (‘CDMO segment”):
earnings before interest, taxes, depreciation and amortization ("EBITDA") and Excess Cash Flow. We believe these non-GAAP financial measures are helpful in understanding our CDMO segment as such useful
to investors in allowing for greater transparency of supplemental information used by
management. EBITDA is used by investors, as well as management in
assessing our performance. Under the credit agreement with our lender (OrbiMed), Excess Cash Flow after interest paid on the OrbiMed debt, is one of the metrics used to determine the amount that OrbiMed may require the Company to prepay on the outstanding principal on our loan on a quarterly basis (see Notes to
Financial Statements for description of the credit agreement). Excess Cash Flow is a
useful measure of cash flow available to not just service debt, but also
use for other Company operating activities, including product development and commercialization. Non-GAAP financial measures should be considered in addition to, but not as a substitute for,
reported GAAP results. Further, Non-GAAP financial measures, even if similarly
titled, may not be calculated in the same manner by all companies, and
therefore should not be compared. 49
|
 Appendix |
 Dosing
Profile Attribute
Meloxicam Ketorolac Caldolor (ibuprofen) Ofirmev (APAP) Route IV/IM IV/IM IV IV Onset of pain relief ~10 min* 30 min N/A N/A Time to peak analgesic effect ~40 min* 1-2 hrs N/A N/A Duration of pain relief 18-24 hrs 4-6 hrs 4-6 hrs 4-6 hrs Admin. IV bolus and eventual pre- filled syringe Ready to use IV Dilution required, 30 min infusion Ready to use, 15 min infusion 51 * IV Meloxicam clinical data from Study-04. |
 Multiple
IV Phase 2 Trials Trial
Design Outcome Phase 2 Study - 02 Acute pain following dental surgery (N = 230) Statistically significant differences for meloxicam doses compared to placebo were seen in SPID24, pain relief and onset of pain relief Phase 2 Study - 04 Acute pain following open abdominal hysterectomy surgery (N = 486) Statistically significant differences for meloxicam doses compared to placebo were seen in multiple efficacy analyses, including SPID24. meloxicam 30 mg and 60 mg produced the greatest response with no difference between doses Phase 2 Study - 05 Acute pain following laparoscopic abdominal surgery (N =50) Study stopped early (planned N = 250) for business reasons. However, statistically significant differences in SPID48 observed for 30mg QD dose despite small sample size Phase 2 Study -014 Safety, Efficacy and PK in Post-op bunionectomy (N=59) No serious AEs, no bleeding events Efficacy-Statistically significantly reductions in pain intensity as measured by SPID48 for 30 and 60 mg QD vs placebo 52 |
