UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Date of report (Date of earliest event reported): May 1, 2017
Commission File Number: 001-37544
AmpliPhi Biosciences Corporation
(Exact name of Registrant as specified in its charter)
| Washington | 91-1549568 |
| (State or other jurisdiction of incorporation or organization) |
(IRS Employer Identification No.) |
3579 Valley Centre Drive, Suite 100
San Diego, California 92130
(Address of principal executive offices)
(858) 829-0829
(Registrant’s Telephone number)
N/A
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the Registrant under any of the following provisions (see General Instruction A.2. below):
| ¨ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company x
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. x
In this report, “AmpliPhi Biosciences,” “AmpliPhi,” “Company,” “we,” “us” and “our” refer to AmpliPhi Biosciences Corporation and its wholly owned subsidiaries, unless the context otherwise provides.
Item 8.01 Other Events.
We are filing the following information with the SEC for the purpose of updating certain aspects of our publicly disclosed descriptions of our business and risk factors.
Company Overview
We are a biotechnology company focused on the discovery, development and commercialization of novel phage therapeutics. Phage therapeutics use bacteriophages, a family of viruses, to kill pathogenic bacteria. Phages have powerful and highly selective mechanisms of action that permit them to target and kill specific bacteria. We believe that phages represent a promising means to treat bacterial infections, especially those that have developed resistance to current therapies, including the so-called multi-drug-resistant or “superbug” strains of bacteria.
The extensive use of antibiotics since the beginning of the modern antibiotics era in the 1940s has resulted in drug resistance among many disease-causing bacteria. According to the U.S. Centers for Disease Control and Prevention, or CDC, resistance to antibiotics threatens to reverse many of the key medical advances of the last half-century. Examples of clinically important microbes that are rapidly developing resistance to available antimicrobials include bacteria that cause skin, bone, lung and bloodstream infections (e.g., Staphylococcus aureus, or S. aureus and methicillin-resistant S. aureus, or MRSA), pneumonia and lung infections in both community and hospital settings and cystic fibrosis, or CF, patients (e.g., A. baumanii, P. aeruginosa, and K. pneumoniae), meningitis (e.g., S. pneumonia ), urinary tract and gastrointestinal infections (e.g., E. coli and C. difficile ). As phages kill bacteria in ways entirely unlike the mechanisms used by traditional antibiotics, we believe that most multi-drug resistant bacteria will be susceptible to phage therapy. Furthermore, should resistant bacteria emerge or evolve, we believe it will remain possible to identify phages that can effectively kill these resistant bacteria.
Our goal is to be the leading developer of phage therapeutics. We are combining our expertise in the manufacture of drug-quality bacteriophages and our proprietary approach and expertise in identifying, characterizing and developing naturally occurring bacteriophages with that of collaboration partners in bacteriophage biology, synthetic biology and manufacturing, to develop state-of-the-art bacteriophage products. We are developing phage products to combat multi- or pan-drug-resistant bacterial pathogens, leveraging advances in sequencing and molecular biology. We have developed certain phage combinations that we believe maximize efficacy and minimize phage resistance. We currently have product candidates for the treatment of S. aureus infections, including MRSA, P. aeruginosa infections, and C. difficile infections.
Our lead product candidate is AB-SA01, for the treatment of S. aureus infections, including MRSA. We also have another product candidate in earlier stage development, AB-PA01 for the treatment of P. aeruginosa infections, and an additional discovery program, AB-CD01 for the treatment of C. difficile infections.
We are developing our phage product candidates using a proprietary discovery and development platform, which is designed for rapid identification, characterization and manufacturing of multiple phage therapeutics, allowing for real-time manufacturing of personalized phage cocktails following isolate screen and phage selection. Each product candidate combines several carefully chosen phages, which target a specific disease-causing bacteria such as S. aureus, Salmonella, P. aeruginosa, Enterococcus, E. coli, Klebsiella, Enterobacter, Acinetobacter, Streptococcus, and C. difficile. We believe that the combination of our platform, our manufacturing capability, our understanding of the regulatory and development requirements of bacteriophage therapeutics, and the clinical and scientific expertise of our collaboration partners may enable the rapid advancement of phage therapeutics through the clinic and the regulatory approval process.
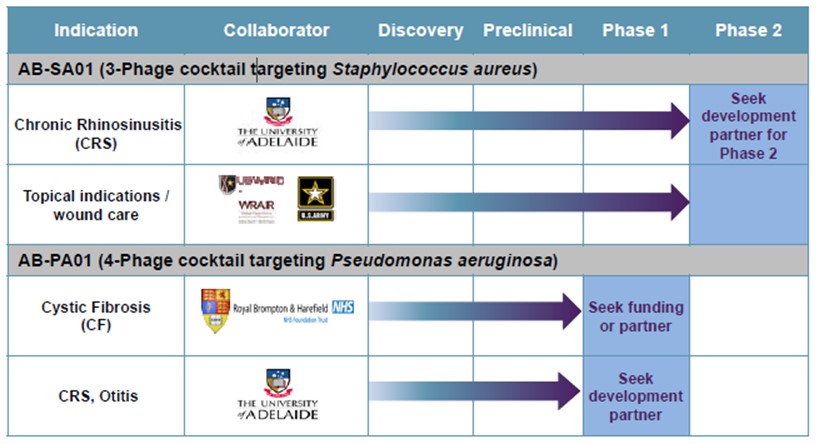
In November 2015, our Australian subsidiary, AmpliPhi Australia Pty Ltd, entered into a clinical trial research agreement with the University of Adelaide and the Queen Elizabeth Hospital, both in Adelaide, SA, Australia, to conduct a Phase 1 clinical trial titled “A Phase 1 Investigator Initiated Study to Evaluate the Safety, Tolerability and Preliminary Effectiveness of AB-SA01 in Patients with Chronic Rhinosinusitis Associated with S. aureus infection”. The University of Adelaide sponsored the clinical trial while we supplied AB-SA01 and controlled the trial protocol. This clinical trial primarily measured the safety and tolerability of AB-SA01 and secondarily examined the presence of S. aureus and symptoms assessed by the patient as well as by the physician using standard questionnaires used by physicians to assess treatment efficacy. We enrolled nine patients in the trial, divided into three cohorts. The first cohort received a twice daily dose of AB-SA01 for seven days. The second cohort received the same dose twice daily for 14 days. The third cohort received a higher dose of AB-SA01 twice daily for 14 days. Patients were monitored an additional 30 days following their last day of treatment. In October 2016, we reported topline safety and tolerability results which demonstrated that AB-SA01 was well tolerated with no drug-related serious adverse events. In December 2016, we reported the final results from the Phase 1 trial. AB-SA01 met the trial’s primary endpoints of safety and tolerability and all nine patients enrolled in the study experienced a reduction in the quantity of S. aureus infecting their sinuses, with some patients showing complete eradication of the bacterial infection. A comparison of pre- and post-treatment endoscopic images showed symptomatic improvement, including reductions in mucosal edema, discharge and polyps. In February 2017, we held a telephonic meeting with the FDA during which we received positive feedback from the FDA regarding our previously submitted proposal to commence a Phase 2 clinical trial of AB-SA01 in chronic rhinosinusitis, or CRS, patients. We are evaluating whether or when to pursue the initiation of such Phase 2 trial, but we believe AB-SA01 is well-positioned for study in a Phase 2 trial in patients with CRS. In the official minutes from the meeting, the FDA acknowledged that phage therapy is an exciting approach to treatment of multidrug-resistant organisms and expressed a commitment to addressing the unique regulatory challenges that might arise during product development. The FDA further acknowledged that the clinical safety and effectiveness data collected during development, including from emergency case studies, could inform future discussions for clinical development and ultimately, the regulatory pathway to approval. The FDA indicated they were open for continued discussions and suggested various potential next steps.
In June 2013, we entered into a cooperative research and development agreement, or Research and Development Agreement, with the United States Army Medical Research and Materiel Command focusing on developing bacteriophage therapeutics to treat S. aureus, E. coli and P. aeruginosa infections. In 2016, under this Research and Development Agreement, we completed enrollment of a Phase 1 safety study of AB-SA01 for the treatment of wounds infected with S. aureus and we reported safety and tolerability results which demonstrated that AB-SA01 was well tolerated throughout the trial. No subject had a treatment-emergent adverse event, or TEAE, which was considered definitely or probably related to AB-SA01, and there were no severe or higher grade TEAEs, serious adverse events or discontinuation of treatment due to TEAEs. Additionally, all laboratory values and vital sign parameters were within normal ranges. Overall, treatment with AB-SA01 was well tolerated when administered topically to the intact skin of healthy adults.
Personalized Precision Medicine Applications
We believe our bacteriophage technology may have unique application in the area of personalized medicine. In particular, we believe our bacteriophage technology can be used to develop personalized, targeted therapies for patients who suffer from serious or life-threatening antibiotic-resistant bacterial infections and who have limited or no other satisfactory treatment options. Moreover, we believe our ability to customize phage therapies for antibiotic-resistant infections, combined with the ability of bacteriophage to re-sensitize drug-resistant populations to antibiotics, represents what could be a powerful tool against the growing challenge of antibiotic-resistant infections. We have commenced a focused effort to develop precisely targeted and personalized bacteriophage therapies aimed at addressing the unmet medical need of serious or life-threatening antibiotic-resistant infections.
Under existing compassionate-use guidelines, we expect to provide personalized phage therapies to patients suffering from severe, multidrug-resistant, or MDR, infections who have failed prior therapies. We believe this strategic approach will not only provide potential benefit to patients to whom we are able to provide personalized phage therapies under the compassionate-use guidelines, but also provide the clinical data from these compassionate use cases that we expect to support the potential validation of the clinical utility of phage therapy and inform our future discussions with the FDA in 2018 or later on defining a potential path to market approval. We anticipate that we will initially make personalized phage therapies available in Australia, where we plan to collaborate with leading hospitals and key opinion leaders to identify and select eligible patients. We believe Australia has a favorable regulatory framework with respect to treating patients under compassionate use guidelines.
Our new emphasis on personalized medicine builds upon our prior successes using tailored bacteriophage therapies under emergency investigational new drug applications to treat individual patients battling life-threatening, MDR bacterial pathogens who had exhausted their treatment options. In March 2016 we collaborated with several academic institutions and a U.S. Navy laboratory to produce a personalized bacteriophage therapy that successfully treated a critically ill, comatose patient with an MDR Acinetobacter baumannii (A. baumannii) infection. Shortly after phage therapy was initiated, the patient emerged from the coma and continued to improve under an ongoing combination of phage and antibiotic therapies until the infection was cleared. To date, the infection has not returned. Additionally, in December 2007 our wholly owned subsidiary, Special Phage Services, was instrumental in developing a personalized phage therapy that, together with a course of antibiotics, eliminated a previously antibiotic-resistant Pseudomonas aeruginosa (P. aeruginosa) infection in the bladder of a female cancer patient.
We currently estimate that we may be able to leverage compassionate-use data to demonstrate the clinical utility of our bacteriophage therapies between 2017 and 2018. During this period, we anticipate optimizing bacteriophage therapeutic regimens, expanding our phage libraries for MDR pathogens and potentially publishing clinical evidence in collaboration with treating physicians and other researchers involved with the patient treatment.
The Need for New Anti-Infective Therapies
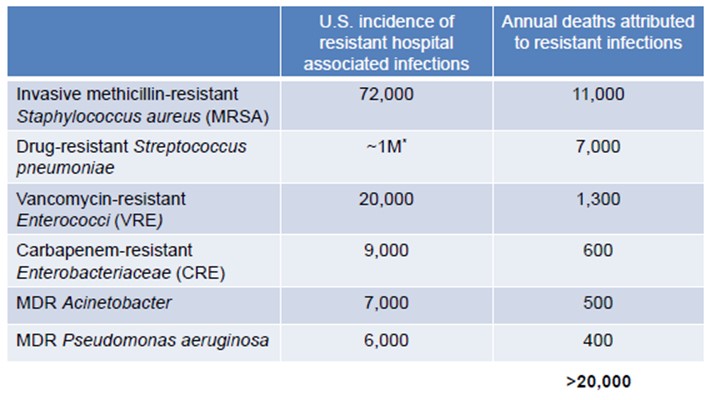
The rapid and continuous emergence of antibiotic-resistant bacteria has become a global crisis. Despite this crisis, the number of novel anti-infective therapies currently in development is at historically-low levels. The CDC estimates that more than two million people in the United States acquire an antibiotic-resistant infection each year and more than 23,000 of these prove fatal. In a reported filed in September 2016, a Reuters analysis found that nationwide, drug-resistant infections were mentioned as contributing or causing the death of more than 180,000 people, meaning drug-resistant infections now kill more patients every year than breast cancer. In a report commissioned by the U.K. government and published in May 2016, it is estimated that 700,000 people die yearly from drug-resistant infections worldwide and by 2050 that number could reach 10,000,000. It is estimated that 50% of hospital-acquired infections are resistant to first-line anti-infective therapies. The cumulative annual cost for treating resistant bacterial infections in the United States alone is estimated to be $20 billion, and it is further estimated that by 2050 the cumulative annual cost to global economic output could reach $100 trillion.
The CDC’s latest report on the matter, Antibiotic Resistance Threats in the United States, 2013, notes that there are “potentially catastrophic consequences of inaction.” Despite the potential market opportunity, only two New Drug Applications, or NDAs, for antibacterial drugs were approved by the FDA between 2010 and 2012 compared to 18 in the period between 1980 and 1984. One of the primary recommendations of the CDC is the development of new antimicrobials to diversify treatment options.
We believe bacteriophage technology is uniquely positioned to address the global health threat of antibiotic resistance, due to the ability of bacteriophage to precisely target bacterial infections and work synergistically with antibiotics by re-sensitizing multi-drug resistant bacterial infections to antibiotics.
Product Candidates
AB-SA01: Infections Caused by S. aureus
By screening our proprietary library of phages, we selected a phage product candidate mix that has demonstrated, in in vitro studies, greater than 92% activity against a global diversity panel that includes some of the most virulent isolates of S. aureus, including MRSA isolates. The three phage constituents of AB-SA01 were subsequently tested for their ability to infect clinically relevant bacterial isolates collected from around the world and were shown to have similar activity with maximal complementation. Complementation, defined as the percentage of S. aureus isolates susceptible to more than one phage, is emphasized in product selection to reduce risk of the emergence of bacterial resistance.
In connection with our Research and Development Agreement with the U.S. Army Medical Research and Materiel Command, we have been developing AB-SA01 to treat acute and chronic infections caused by S. aureus, including infections caused by MRSA strains of the same bacterium. MRSA infections are one of the most common causes of hospital-acquired (nosocomial) infections. The CDC estimates that more than 850,000 patients were treated for S. aureus infections of the skin or soft tissue in 2013 and, due to failure of first-line treatment, more than 50% of these patients required a second-line treatment and approximately 35% of them required a third-line treatment. Global Data estimates the market for MRSA infection treatments alone was more than $2.7 billion in 2007. This market is forecasted to grow to more than $3.5 billion by 2019. We initiated the Phase 1 clinical trial in May 2016 and completed enrollment in July 2016. In December 2016, we reported final results from this Phase 1 trial to evaluate the safety and tolerability of AB-SA01, our proprietary investigational phage cocktail targeting Staphylococcus aureus (S. aureus) infections. Overall, treatment with AB-SA01 was well tolerated when administered topically to the intact skin of healthy adults.
In December 2015, we initiated a Phase 1 trial at the University of Adelaide Queen Elizabeth Hospital to evaluate the safety and preliminary efficacy of AB-SA01 in CRS patients infected with S. aureus. In December 2016, we reported final results from this trial. AB-SA01 met the trial’s primary endpoints of safety and tolerability and all nine patients enrolled in the study experienced a reduction in the quantity of S. aureus infecting their sinuses, with some patients showing complete eradication of the bacterial infection.
AB-PA01: Lung Infections in Cystic Fibrosis (CF) Patients Caused by P. aeruginosa
We are initially developing AB-PA01, a cocktail comprised of four phages, for the treatment of P. aeruginosa, the most prevalent bacterial infection in cystic fibrosis (“CF”) patients. P. aeruginosa is the primary cause of lung infection in approximately 80% of CF patients ages 25 to 34, causing an estimated 450 deaths per year in the United States. To develop our product candidates, we have created a global diversity panel of relevant clinical isolates (bacteria isolated from patients) from clinics around the globe. These diversity panels have been screened against our phage libraries, which are isolated and characterized according to our set of proprietary discovery protocols. We have demonstrated, in in vitro and in vivo studies, that our proprietary phage mix is able to effectively kill targeted bacteria. Furthermore, our phage mixes are selected to exhibit a high degree of overlap, defined as the strains of bacteria targeted by more than one phage in the product. We believe that high overlap is an important factor in preventing bacteria from developing resistance to our phage product candidates.
Similar to work described above for S. aureus, we have tested over 400 clinical P. aeruginosa clinical isolates. As an example, initial host range testing was performed with a reference panel of 67 CF isolates. AB-PA01 showed an activity of 95.5% (64/67) with 87.5% (56/64) of the positives isolates hit by more than one phage in the mix.
In collaboration with Institut Pasteur (Paris, France) and also with the Brompton Hospital, Imperial College (London, United Kingdom), we have demonstrated in the preclinical studies that phages can effectively treat infections in animal models of acute P. aeruginosa lung infections. In one such study, we inoculated eight mice with P. aeruginosa and treated them with either PBS (control group), our phage mix, or with an antibiotic.
Bacterial counts and the number of bacteriophage infection units detected by assay, or phage titers, were measured in these animals after 24 hours, and the results demonstrated that our phage mix effectively lowered the bacterial counts, or CFU, in the mouse lung to levels comparable to antibiotic treatment (PBS vs. antibiotic, p=0.0003; PBS vs. bacteriophage, p=0.0003). A p-value is a statistical measure of the probability that the difference in two values could have occurred by chance. The smaller the p-value, the lower the likelihood is that the difference occurred by chance, or the greater our confidence is that the results are statistically significant. Furthermore, it was evident that phage replicated to high levels in the infected lung.
Another preclinical study conducted at the Institut Pasteur in mice (12 mice in each of the treatment and control groups) demonstrated the ability of our phage mix to reach the lung within two hours of being delivered by oral administration. The phage levels increased between two and six hours post-treatment, and the difference was statistically significant (p-value <0.001). These results demonstrate that when orally administered in mice, phages not only reached the lungs, but were also able to infect and multiply in target bacteria.
In a separate in vivo study of acute P. aeruginosa infection of the mouse lung conducted at the Brompton Clinic, results demonstrated that our phage mix reduced CFU levels upon simultaneous intranasal administration (six mice in each of the treatment and control groups) and also when administered 24 hours post-bacterial infection (seven mice in the treatment group and eight mice in the control group) using a standard strain of P. aeruginosa.
We were granted an advisory meeting with the MHRA in the first quarter of 2014 to discuss our plans and intend to move the AB-PA01 compound into additional preclinical testing in preparation for a Phase 1/2 clinical trial in CF patients. We also sought advice on the acceptability of CMC plans. The MHRA concurred with our approach and plans as presented, including a first-in-man dose ranging clinical trial in CF patients. We have completed product candidate selection and have conducted manufacturing process development and scale-up. We estimate that we may be in a position to initiate a Phase 1 clinical study of AB-PA01 for the treatment of P. aeruginosa infections in CF patients in as early as approximately nine months after electing to move forward with such study.
We have also begun an evaluation of our P. aeruginosa phages in preclinical animal models of CRS in collaboration with the University of Adelaide.
If we achieve successful proof of concept studies, we may consider developing this compound for the treatment of other acute and chronic lung infections, such as ventilator associated bacterial pneumonia, or VABP, and chronic obstructive pulmonary disease, or COPD and chronic suppurative otitis media. P. aeruginosa is the predominant pathogen in these indications.
AB-CD01: Gastrointestinal (GI) Infection Caused by C. difficile, or CDI
From 2000 through 2007, deaths in the United States from infections caused by C. difficile, or CDI, increased over 400%. Over 90% of such deaths occur in hospitalized or confined patients over the age of 65. Global Data estimates that the major European Union and United States markets for CDI therapies grew to more than $314 million in 2011 and they are expected to grow to more than $500 million by 2019.
According to the CDC almost 250,000 people each year require hospitalization for CDI and at least 14,000 people die each year in the United States from CDI. The CDC also estimates that 20 – 40% of CDI recurs with standard antibiotic treatment. We believe that orally delivered phages are well suited to treat CDI. Researchers at the University of Leicester have discovered phages that have been shown to be effective in vitro and in vivo against clinically-relevant strains of C. difficile isolated from around the world. These same researchers have also shown phage cocktails to be effective in preventing C. difficile biofilm formation in vitro. While current pathogenic strains of C. difficile are not yet antibiotic-resistant, the CDC has categorized C. difficile as an urgent threat and has stated that CDI requires urgent and aggressive action. We believe that there may be a significant market opportunity for a phage therapy in treating this infection. We have conducted preclinical studies to select and optimize our phage cocktail and manufacturing strains as well as evaluate their efficacy in animal models. Data published in 2016 by our collaborators (Nale et al) in Frontiers in Microbiology suggest that the phages significantly reduced C. difficile biofilms in vitro, and in vivo prevented bacterial colonization in a wax model (G. mellonella) when phages were used alone or in combination with vancomycin and antibiotic commonly used to treat CDI.
Prior Clinical Development
In 2010, our wholly owned subsidiary, Biocontrol Ltd, reported a double-blind placebo-controlled, randomized Phase 1/2 clinical trial targeting chronic ear infections (otitis) caused by P. aeruginosa . To our knowledge, this was the first randomized placebo-controlled efficacy trial of bacteriophage therapy. Results were published demonstrating decreasing levels of P. aeruginosa in the ear and improvement of clinical condition with a single input dose of 2.4 nanograms of bacteriophage preparation. While this was a small trial (n=24), changes from baseline at the end of the trial in the test group (n=12) were statistically significant for both clinical condition (p=0.001) and bacterial load (p=0.016). No significant changes were seen in the control group (n=12) compared to baseline at the end of the trial. Difference between test and control groups was statistically significant by analysis by covariance on day 21 for bacterial count (p=0.0365). These results will need to be validated in larger well-controlled trials.
Anti-Infective Therapeutics Market
The market opportunity for antibiotics is large, with the market estimated to reach $44.7 billion in annual sales globally in 2020. Almost one in every five deaths worldwide occurs as a result of infection and, according to the World Health Organization, or WHO, many bacterial infections will become difficult or impossible to cure as the efficacy of current antibiotic drugs wanes. Despite the advances in antimicrobial and vaccine development, infectious diseases still remain as the third-leading cause of death in the United States and the second-leading cause of death worldwide.
The number of new antibiotics approved by the FDA and other global regulatory authorities has declined consistently over the last two decades. According to the PEW Charitable Trusts report, as of December 2016 there were an estimated 40 new antibiotics in clinical development for the U.S. market. Historically, the success rate from Phase 1 to marketing approval is only one in five for infectious disease products. We therefore believe there is a need for new approaches to treat serious bacterial infections. Hospital-acquired (nosocomial) infections are a major healthcare problem throughout the world, affecting developed countries as well as resource-poor countries. The WHO reports that hospital-acquired infections are among the major causes of death and increased morbidity among hospitalized patients and estimates that more than 1.4 million people per year worldwide suffer from infectious complications from a hospital stay.
A recent CDC report also cites that in the United States, between 5 and 10% of all patients admitted to a hospital will be affected by a hospital-acquired infection during their stay, typically requiring extended stays and additional care. There is also a significant risk of death from such infections. In the United States, the CDC estimates that approximately 99,000 people die from hospital-acquired infections each year. The Cystic Fibrosis Foundation estimates that P. aeruginosa accounts for 10% of all hospital-acquired infections.
Compounding the above situations is the alarming and continuing rise in the prevalence of antibiotic-resistant bacterial infections. This, coupled with the lack of new antibiotics in current discovery and development pipelines, has generated a significant clinical management problem worldwide, leading to increases in morbidity and mortality due to these antibiotic-resistant bacteria as well as increases in healthcare costs.
The first of these antibiotic-resistant infections to reach epidemic proportions was caused by the Gram-positive bacterium S. aureus. S. aureus resistance to a broad range of antibiotics has necessitated the use of expensive and potentially toxic “drugs of last resort”, most notably vancomycin. Antibiotic-resistant forms of S. aureus, usually termed MRSA, VISA (vancomycin-intermediate S. aureus), or VRSA (vancomycin-resistant S. aureus ), can be extremely challenging to treat. Although several antibiotics targeting S. aureus have been developed, rapidly developing bacterial resistance has been noted for all of these including linezolid, daptomycin and tigecycline. On the basis of historical evidence, resistance to these existing products is likely to increase over time, and this picture is further complicated by the reduced efficacy of conventional antibiotics against Staphylococcus biofilms.
Typically, S. aureus infection causes a variety of suppurative (pus-forming) infections and toxinoses (lesions) in humans. It causes superficial skin lesions such as boils, styes and furuncles; more serious infections such as pneumonia, mastitis, phlebitis, meningitis and urinary tract infections; and deep-seated infections, such as osteomyelitis and endocarditis. S. aureus is the leading cause of wound infections, in particular, hospital-acquired (nosocomial) infection of surgical wounds and infections associated with indwelling medical devices. S. aureus is the leading pathogen in healthcare-associated infections in the United States as a whole, accounting for 30.4% of surgical site infections, or SSI, and 15.6% of such infections overall.
Infections also occur in patients with CF, which is a genetic disease affecting primarily Caucasians of northern European descent. According to the Cystic Fibrosis Foundation, there are approximately 50,000 cases of CF in North America and Europe. P. aeruginosa opportunistically infects the mucous membranes, primarily the lungs, of CF patients and quickly grows out of control, resulting in pneumonia. P. aeruginosa infections are notoriously resistant to known antibiotics, and treatment may be further complicated by the formation of biofilms. Biofilms are organized structures of microorganisms growing on solid surfaces (such as lung tissue) and often limit access of antibiotics to the covered tissues. Since phages attack bacteria in a manner independent of chemical antibiotic resistance mechanisms and can infect bacteria growing in biofilms, we believe that P. aeruginosa infection among CF patients represents a compelling indication to pursue. The availability of Pseudomonas-specific phages along with validated animal models of P. aeruginosa lung infections has contributed to the development of our bacteriophage program in CF.
Anti-Infective Treatments with Bacteriophages
Background
The dramatic rise in antibiotic resistance, the appearance of an increasing number of new “superbugs” and the lack of new antibiotics in the pipeline has prompted calls to action from many of the world’s major health bodies such as the CDC and the WHO, who warn of an “antibiotic cliff” and a “post-antibiotic era.” In 2009, the European Antimicrobial Resistance Surveillance System, or EARSS, concluded that “the loss of effective antimicrobial therapy increasingly threatens the delivery of crucial health services in hospitals and in the community.” This conclusion was reinforced by The Antimicrobial Availability Task Force, or AATF, of the Infectious Diseases Society of America, or IDSA, and the European Centre for Disease Prevention and Control, or ECDC, in conjunction with the European Medicine Agency, or EMA. We therefore believe there is a pressing need to find alternative antibacterial therapies.
Bacteriophage therapy has the potential to be an alternative method of treating bacterial infection. Phages are ubiquitous environmental viruses that grow only within bacteria, but are among the most abundant and diverse organisms on the planet. The name “bacteriophage” translates as “eaters of bacteria” and reflects the fact that as they grow, phages kill the bacterial host by multiplying inside and then bursting through the cell membrane in order to release the next generation of phages. Phages can differ substantially in morphology and each phage is active against a specific range of a given bacterial species. Phages were first discovered in 1915 at the Institut Pasteur and were shown to kill bacteria taken from patients suffering from dysentery. Furthermore, it was noted that phage numbers rose as patients recovered from infection, suggesting a direct association.
Life Cycle of a Bacteriophage
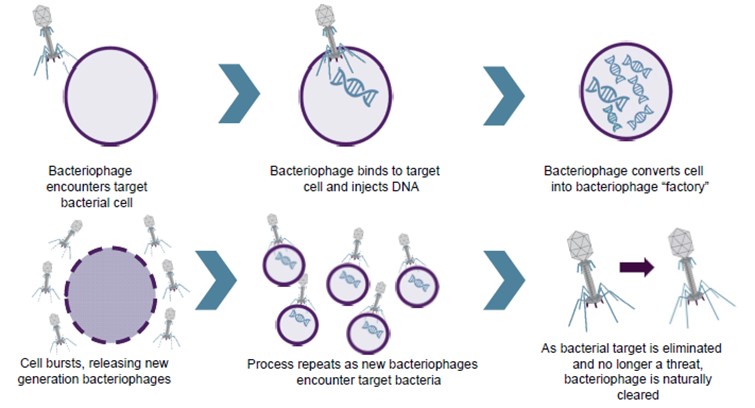
Until the discovery of effective antibiotics, phages were used as an effective means of combating bacterial infection. When broad-spectrum antibiotics came into common use in the early 1940s, phages were considered unnecessary, with antibiotics being seen for many years as the answer to bacterial disease. This attitude persisted until the development of the wide-ranging, and in some cases total, resistance to antibiotics seen within the last 10 years.
Phages have the potential to provide both an alternative to, and a synergistic approach with, antibiotic therapy. Since they use different mechanisms of action, phages are unaffected by resistance to conventional antibiotics. Phages containing certain enzymes also have the ability to disrupt bacterial biofilms, thus potentiating the effect of chemical antibiotics when used in combination with them.
Our Strategy
Our strategy is to use techniques of modern biotechnology and current state-of-the-art practices for drug development in concert with existing regulatory guidance to develop a pipeline of bacteriophage products that will destroy bacteria such as MRSA, which are resistant to antibiotics. We intend to leverage advances in sequencing and molecular biology to build upon the demonstrated ability of using phages therapeutically to successfully treat bacterial infections.
As discussed in greater detail above, we also plan to use our bacteriophage technology to develop personalized targeted therapies for patients who suffer from serious or life-threatening antibiotic-resistant bacterial infections and who have few or no other satisfactory treatment options. Our long-term strategy is to become the world leader in treating drug-resistant bacterial infections.
We supplement our internal resources with world-class scientific and medical collaborations throughout the world. For example, through a collaboration with The University of Adelaide in Australia and the University Hospital Ghent in Belgium, we conducted preclinical studies showing the ability of S. aureus phage preparations to kill over 140 clinical isolates from CRS patients demonstrating activity of greater than 90%. Furthermore, a S. aureus mixture was shown to be safe and efficacious in a preclinical sheep model of CRS. A Phase 1 clinical trial for this program was conducted at the University of Adelaide’s Queen Elizabeth Hospital for the treatment of patients suffering from CRS associated with S. aureus infection. In December 2016, we reported final results from the Phase 1 trial of AB-SA01 in patients with CRS. AB-SA01 met the trial’s primary endpoints of safety and tolerability and all nine patients enrolled in the study experienced a reduction in the quantity of S. aureus infecting their sinuses, with some patients showing complete eradication of the bacterial infection. In August 2016, we tested AB-SA01 against 90 S. aureus clinical isolates from CRS patients located in Belgium and showed similar activity to isolates obtained from Australian patients, highlighting the diverse geographic activity of our phage cocktail.
In collaboration with the U.S. Army, we completed a Phase 1 safety study under an IND that we believe will support the further development of a treatment for S. aureus infections for wound and skin infections. In December 2016, we reported final results from the Phase 1 trial to evaluate the safety and tolerability of AB-SA01. Overall, treatment with AB-SA01 was well tolerated when administered topically to the intact skin of healthy adults.
We collaborate with the Royal Brompton Hospital in London where we have demonstrated that a phage product candidate can survive nebulization, was effective in killing over 83% of recent clinical P. aeruginosa isolates, and in preclinical mouse models demonstrated that a phage mixture dose-dependently clears P. aeruginosa infection from the lung and reduced inflammation. We have completed selection of the phages for drug product selection for AB-PA01, and we estimate that we may be in a position to initiate a Phase 1 clinical study of AB-PA01 for the treatment of P. aeruginosa infections in CF patients in as early as approximately nine months after electing to move forward with such study.
Strategic Alliances and Research Agreements
Global R&D Agreement with U.S. Army
In June 2013, we entered into a Research and Development Agreement with the U.S. Army Medical Research and Materiel Command. The Research and Development Agreement focuses on developing bacteriophage therapeutics to treat S. aureus, E. coli and P. aeruginosa infections, with the initial therapeutic development focus being wounds and skin infections from S. aureus, which is the leading pathogen in healthcare-associated infections in the United States as a whole, accounting for 30.4% of surgical site infections.
We retain global regulatory ownership and commercial rights to all products developed by us under the Research and Development Agreement. The U.S. Army Medical Research and Materiel Command will have the right to retain a non-exclusive license to use any products developed by or on behalf of the U.S. Government for non-commercial uses. We also have the rights to exclusively license any intellectual property developed by the U.S. Army Medical Research and Materiel Command under the collaboration on terms to be agreed upon.
The Research and Development Agreement expires in June 2018 and can be terminated by either the U.S. Army Medical Research and Materiel Command or us upon 60 days’ written notice to the other party at any time.
University of Leicester License Agreement
In September 2013, we entered into a license agreement with the University of Leicester which provides us with exclusive rights to certain patents and materials owned by the University of Leicester, as well as non-exclusive licenses to related know-how, to research, develop, manufacture, use and sell one or more phage therapy products for treating C. difficile infection or carriage in humans or animals.
Under the license agreement, we have paid an up-front fee and have agreed to pay the University of Leicester royalties based on product sales and make certain milestone payments based on product development. The license agreement expires on the later of the expiration of the licensed patents or September 2028, and is terminable by us at any time upon 60 days’ notice, by the University of Leicester (a) if we legally challenge the validity or ownership of any of the licensed patents, (b) if we fail to pay the fees, milestones or royalties due under the license agreement or (c) if we fail to make substantial commercial progress in developing the licensed technology and, following good faith discussions, the parties are unable to identify feasible next steps to remedy any such failure to make substantial commercial progress. The license agreement is also terminable by either party upon the material breach by the other party (subject to a 30-day cure period) or upon the other party’s bankruptcy or insolvency.
In April 2017, the University of Leicester provided us with notice that it intends to terminate the license agreement as a result of its determination that we have not continued to make substantial commercial progress in relation to the technology licensed to us under the agreement. Under the license agreement, we have the right to enter in good faith discussions with the University of Leicester to identify feasible next steps to remedy the perceived lack of commercial progress prior to a termination of the license agreement on such basis. Although we intend to engage in such discussions with the University of Leicester, there can be no assurance that the parties will be able to identify or agree upon feasible next steps to remedy the purported lack of commercial progress, or that we will otherwise be able to resolve the matter in a manner that results in our retaining the rights licensed to us on the original terms of the agreement, on other favorable terms, or at all. The licensed rights relate to bacteriophage therapeutic products for the treatment of C. difficile, which is a program we are not actively developing at present, but may choose to develop in the future pending the rights to do so.
License Agreement with United Kingdom Secretary of State for the Department of Health
In January 2011, upon completion of our acquisition of Biocontrol Ltd., we assumed a license agreement entered into in March 2007 between Biocontrol Ltd. and the Health Protection Agency, Centre for Emergency Preparedness and Response, to use certain intellectual property rights to develop treatments for bacterial biofilm infections. The agreement was subsequently assigned to the United Kingdom Secretary of State for the Department of Health, or DoH.
Under the license agreement, we have obtained exclusive rights to a patent portfolio related to the use of bacteriophages combined with biofilm-disrupting agents in treating biofilm infections. In consideration for the exclusive license, we may be required to pay to the DoH certain milestone payments in the aggregate of up to £10,000 per product, as well as single digit percentage royalty on net sales of products incorporating licensed intellectual property.
The license agreement shall remain in full force and effect until the expiration of the last patent exclusively licensed under the license agreement. If we default on any milestone or royalty payments, or upon breach by us of certain other terms of the license agreement, the DoH may either terminate the license agreement immediately upon written notice or modify the license to be non-exclusive upon 30 days’ written notice.
Intellectual Property
General
Our goal is to obtain, maintain and enforce patent protection for our product candidates, formulations, processes, methods and any other proprietary technologies, preserve our trade secrets and operate without infringing on the proprietary rights of other parties, both in the United States and in other countries. Our policy is to actively seek to obtain, where appropriate, the broadest intellectual property protection possible for our current product candidates and any future product candidates, proprietary information and proprietary technology through a combination of contractual arrangements and patents, both in the United States and abroad. However, patent protection may not afford us with complete protection against competitors who seek to circumvent our patents.
We also depend upon the skills, knowledge, experience and know-how of our management and research and development personnel, as well as that of our advisors, consultants and other contractors. To help protect our proprietary know-how, which is not patentable, and for inventions for which patents may be difficult to enforce, we currently and will in the future rely on trade secret protection and confidentiality agreements to protect our interests. To this end, we require all of our employees, consultants, advisors and other contractors to enter into confidentiality agreements that prohibit the disclosure of confidential information and, where applicable, require disclosure and assignment to us of the ideas, developments, discoveries and inventions important to our business.
As of December 31, 2016, we owned or had exclusive license rights to a total of 65 patents and applications: five U.S. patents, seven U.S. patent applications, 40 foreign patents, and 13 foreign patent applications, expiring on various dates between 2024 and 2036. These patents and applications cover our lead phage-therapeutic programs and use thereof, the sequential use of bacteriophages in combination with conventional antibiotics, genetic sequence variations, biofilm disrupting agents, methods to reduce antibiotic resistance, methods to design therapeutic combination panels of bacteriophage, disinfection methods using bacteriophages, and bacteriophage mutants having increased bacterial host spectra.
US 7758856 and national patents within the EU deriving from PCT WO2004062677; Bacteriophage for the treatment of bacterial biofilms
Under an existing license from the United Kingdom Secretary of State for the Department of Health (DoH), we have exclusive rights to a patent portfolio related to the use of bacteriophages combined with biofilm-disrupting agents in treating biofilm infections. This portfolio includes one issued patent in the United States and a patent granted in Europe (EP1587520 is validated in France, Germany, Netherlands, Switzerland, Liechtenstein and the United Kingdom). Claims issued in these patents include those directed to compositions and methods related to agents that are able to facilitate the penetration of biofilms, and their combination with therapeutic bacteriophage preparations. The U.S. patent is expected to expire in December 2026 (absent any extensions). The foreign patents are expected to expire in January 2024 (absent any extensions).
US 7807149, US 8105579, US 8388946, continuation application and national filings deriving from PCT WO2005009451; Bacteriophage containing therapeutic agents
Through our wholly owned subsidiary, Biocontrol Ltd, we own three granted U.S. patents and one pending U.S. continuation patent application with claims directed generally to bacteriophage compositions, therapeutic methods of using bacteriophages, and methods of treating bacterial infections by sequentially administering bacteriophages in combination with conventional antibiotics. The pending U.S. continuation application (US 13/757655) relates generally to panels of bacteriophages with different strain specificities for bacterial infections. Corresponding patents have been granted in Australia (AU2004258731), Europe (EP1663265 and EP2570130 — both patents are validated in the United Kingdom, Switzerland, Liechtenstein, Germany, Spain, France, Italy and the Netherlands), Japan (JP5731727 and JP5856556) and Canada (CA2533352). Claims issued in these patents include those directed to therapeutic and non-therapeutic applications of bacteriophage and the sequential use of antibiotics to treat bacterial infections. U.S. patents are expected to expire from July 2024 to March 2027 (absent any extensions). The foreign patents are expected to expire in July 2024 to March 2027 (absent any extensions).
US 8475787, continuation application and national filings deriving from PCT WO2008110840; Beneficial effects of bacteriophage treatment
Through our wholly owned subsidiary, Biocontrol Ltd, we own one granted U.S. patent (8475787), and one pending continuation application (14/625049). This patent family broadly relates to bacteriophage-induced induction of antibiotic sensitivity in a bacterial target, such as P. aeruginosa. The granted U.S. patent is expected to expire in July 2029 (absent any extensions). Corresponding patents have been granted in Australia (AU2008224651), Europe (EP2136826 — validated in the United Kingdom, Switzerland/Liechtenstein, Germany, Spain, France, Italy and the Netherlands), and Japan (JP5988417 and JP6004543). A related Canadian application (CA2680108) has been allowed and will be officially granted upon the payment of the issuance fee due July 4, 2017. Foreign patents in this family are expected to expire in March 2028 (absent any extensions).
PCT WO2013/164640 (United Kingdom priority filing 1207910.9); Therapeutic bacteriophage compositions
Through our wholly owned subsidiary, Biocontrol Ltd, we own a Patent Cooperation Treaty, or PCT, application relating to the design of effective bacteriophage combinations and elimination of antagonistic effects between said bacteriophage. The PCT application published on November 7, 2013, and following International Preliminary Examination a positive patentability opinion issued. National/regional phase applications are currently pending in the U.S. (US14/398384), Canada (CA2871986), Europe (EP2874635), Japan (JP2015/523850), and Australia (AU2013255583). Patents issuing from this PCT, if any, are expected to expire in May 2032 (absent any extensions).
PCT WO2009/044163 (United Kingdom priority filing 0719438.4); Anti-bacterial compositions
Pursuant to the terms of the Asset Purchase Agreement with Novolytics Ltd., we acquired and currently own one U.S. continuation application (14/686315) relating to methods for killing/treating Staphylococcus and MRSA, among other bacteria, using a combined bacteriophage K and bacteriophage P68 composition. A corresponding patent has been granted in Australia (AU2008306626), and China (CN101835384), and Japan (JP6053727), while European application (EP2197284) has been allowed. Related applications are pending in Australia (AU2015264918) and Canada (CA2700646). The granted foreign patents are expected to expire October 2028 (absent any extensions).
PCT WO2013/068743 (United Kingdom priority filing 1119167.3); Novel bacteriophages
Pursuant to the terms of the Asset Purchase Agreement with Novolytics Ltd., we acquired and currently own a U.S. patent application (14/356869) relating to Staphylococcus aureus and MRSA therapeutics, and in particular Phage K mutants capable of targeting an increased number of Staphylococcus aureus strains when compared to wild-type Phage K, as well as uses of said mutant. U.S. patent application 14/356869 has been allowed. Related applications are also pending in Australia (AU2012335397), Canada (CA2890450), Japan (JP 2014/533943) and Europe (EP2776559). Any granted patents will expire in November 2033.
US 15/237496 (converted from United States provisional filing 62/204915); Therapeutic bacteriophage compositions
We own U.S. patent application 15/237496, which is directed to our AB-SA01 bacteriophage panel, mutants thereof, and methods of treating Staphylococcus aureus infections (including MRSA) comprising the use of same. Corresponding foreign applications are being pursued by way of a parallel PCT application. Any granted US patent is expected to expire in August 2036 (absent extensions). Corresponding foreign applications are being pursued by way of a parallel PCT application.
Our success in preserving market exclusivity for our product candidates relies on patent protection, including extensions to this where appropriate, and on data exclusivity relating to an approved biologic. This may be extended by orphan drug and/or pediatric use protection where appropriate. Once any regulatory period of data exclusivity expires, depending on the status of our patent coverage, we may not be able to prevent others from marketing and selling biosimilar versions of our product candidates. We are also dependent upon the diligence of our appointed agents in national jurisdictions, acting for and on our behalf, which manage the prosecution of pending domestic and foreign patent applications and maintain granted domestic and foreign patents.
Competition
We operate in highly competitive segments of the biotechnology and biopharmaceutical markets. We face competition from many different sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies and private and public research institutions all seeking to develop novel treatment modalities for bacterial infections. Many of our competitors have significantly greater financial, product development, manufacturing and marketing resources than we do. Large pharmaceutical companies have extensive experience in clinical development and obtaining regulatory approval for drugs. In addition, many universities and private and public research institutes are active in antibacterial research, some in direct competition with us. We also may compete with these organizations to recruit scientists and clinical development personnel.
There are a handful of small biotechnology companies developing bacteriophage products to treat human diseases. Other than our ongoing clinical trials there is, to our knowledge, one corporate-sponsored clinical trial currently enrolling. A French biotechnology company, Pherecydes Pharma, is acting as clinical trial sponsor of a Phase 1/2 clinical trial in Europe of a phage therapy for the treatment of burn wounds infected with either E. coli and P. aeruginosa , referred to as PhagoBurn. This clinical trial is a randomized, multi-center open label study to assess tolerance and efficacy of local treatment with a bacteriophage cocktail. A multi-center clinical trial also sponsored by Pherecydes Pharma evaluating a bacteriophage cocktail versus placebo for diabetic foot ulcers, is listed on clinicaltrials.gov as active but not yet enrolling. To our knowledge, a small number of biotechnology companies, including Synthetic Genomics and LytPhage, Inc., as well as academic institutions, have earlier stage discovery programs utilizing synthetic biology approaches to genetically modify bacteriophages to remove or input genes to improve therapeutic properties such as increases to the bacterial host range to infect a larger number of bacterial strains and decrease the need for using multiple phages in a product.
A related approach to treating Staphylococcus infections is being pursued by Contrafect Corporation using a bacteriophage lysin (a hydrolytic enzyme produced by bacteriophages) to treat S. aureus bacteremia (infection in the blood). Contrafect has recently completed a Phase 1 intravenous single dose escalation study in healthy volunteers.
Our bacteriophage programs may compete with or be synergistic with currently approved antibiotics, and experimental approaches such as novel antibiotics, antimicrobial peptides, antimicrobial vaccines, metals, antisense, monoclonal antibodies and possibly microbiome manipulation. For example, Seres Therapeutics is developing a single-dose capsule (SER-109) consisting of bacterial spores to treat recurrent CDI (Clostridium difficile infection). SER-109, or similar products that may be in development by third parties, could prove to be competitive to or used in conjunction with a bacteriophage therapeutic approach.
Manufacturing and Supply
We have developed our own manufacturing capabilities at a facility in Ljubljana, Slovenia that is leased by our wholly owned subsidiary, AmpliPhi, Biotehnološke Raziskave in Razvoj, d.o.o. We believe that our facility complies with applicable cGMP regulations, which require, among other things, quality control and quality assurance as well as the corresponding maintenance of records and documentation. Pharmaceutical product manufacturers and other entities involved in the manufacture and distribution of approved pharmaceutical products are required to register their establishments with the FDA, and certain state agencies, including the applicable government agency where the facility is located, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP and other laws.
After conducting a global search, we elected to proceed with establishing a wholly owned cGMP compliant manufacturing facility in Ljubljana, Slovenia. Upon final product selection, we plan to manufacture each of our product candidates in this facility. We have been able to access and hire highly skilled process development and phage manufacturing expertise and believe that we have control of our proprietary platform from phage identification through final product fill and finish. Our facility is comprised of approximately 6,000 sq. ft. of laboratory and office space, where we produce cGMP clinical trial supplies in our 40-liter bioreactor for our current and planned clinical trials. We believe this facility will be sufficient to meet our manufacturing needs through initial Phase 3 clinical trials. Our current formulation for AB-SA01 is intended for sinonasal or topical delivery via a nasal wash solution or dressed bandage. We plan to further optimize future formulations of our product candidates.
Our facility in Ljubljana, Slovenia is subject to inspection and regulation by JAZMP, the Slovenian agency that regulates and supervises pharmaceutical products in Slovenia. Discovery of problems with a product after approval may result in restrictions on a product, manufacturer or holder of an approved New Drug Application/Biologics License Application, including withdrawal of the product from the market. In addition, changes to the manufacturing process generally require prior regulatory approval before being implemented and other types of changes to the approved product, such as adding new indications and additional labeling claims, are also subject to further regulatory review and approval, including approval by the FDA.
Commercialization and Marketing
We have full worldwide commercial rights to all of our phage-based product candidates to treat drug-resistant bacterial infections, including our product candidates: AB-PA01 for the treatment of CF patients with P. aeruginosa lung infections; AB-SA01, for the treatment of S. aureus infections; and AB-CD01 for the prevention or treatment of C. difficile infections. We believe we can maximize the value of our company by retaining substantial global commercialization rights to these product candidates and, where appropriate, entering into partnerships to develop and commercialize our other product candidates. We plan to build a successful commercial enterprise using a sales team in the United States and possibly other major markets and with partners in other territories.
We have not yet established a sales, marketing or product distribution infrastructure because our lead candidates are still in early clinical development. We generally expect to retain commercialization and co-commercialization rights in the United States for all of our product candidates for which we receive marketing approvals. Subject to receiving marketing approvals, we intend to explore building the necessary marketing and sales infrastructure to market and sell our current product candidates. We also intend to explore the use of a variety of distribution agreements and commercial partnerships in those territories where we do not establish a sales force for any of our product candidates that obtain marketing approval.
Government Regulation and Product Approval
Government authorities in the United States, at the federal, state and local level, and other countries extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, post-approval monitoring and reporting, marketing and export and import of products such as those we are developing.
United States Product Development Process
In the United States, the FDA regulates biological products under the Federal Food, Drug and Cosmetic Act, or FDCA, and the Public Health Service Act, or the PHS Act, and related regulations. Biological products are also subject to other federal, state and local statutes and regulations. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local and foreign statutes and regulations require the expenditure of substantial time and financial resources. Failure to comply with the applicable United States requirements at any time during the product development process or approval process, or after approval, may subject an applicant to administrative or judicial sanctions. FDA sanctions could include refusal to approve pending applications, withdrawal of an approval, a clinical hold, warning letters, product recalls, product seizures, total or partial suspension of production or distribution injunctions, fines, refusals of government contracts, restitution, disgorgement or civil or criminal penalties. Any agency or judicial enforcement action could have a material adverse effect on us. The process required by the FDA before a biological product may be marketed in the United States generally includes the following:
| · | completion of preclinical laboratory tests, animal studies and formulation studies according to good laboratory practice requirements, or GLP, or other applicable regulations; |
| · | submission to the FDA of an IND, which must become effective before human clinical trials may begin in the United States; |
| · | performance of adequate and well-controlled human clinical trials according to the FDA’s regulations commonly referred to as good clinical practices, or GCPs, and any additional requirements for the protection of human research subjects and their health information, to establish the safety and efficacy of the proposed biological product for its intended use or uses; |
| · | submission to the FDA of a Biologics License Application, or BLA, for a new biological product; |
| · | satisfactory completion of an FDA inspection of the manufacturing facility or facilities where the biological product is produced to assess compliance with the FDA’s cGMP regulations, to assure that the facilities, methods and controls are adequate to preserve the biological product’s identity, strength, quality and purity; |
| · | potential FDA audit of the nonclinical study sites and clinical trial sites that generated the data in support of the BLA; and |
| · | FDA review and approval, or licensure, of the BLA which must occur before a biological product can be marketed or sold. |
The lengthy process of seeking required approvals and the continuing need for compliance with applicable statutes and regulations require the expenditure of substantial resources even when approvals are inherently uncertain.
The strategies, nature, and technologies of bacteriophage products are different from the conventional antibiotic therapy products. From the regulatory requirements established to ensure the safety, efficacy and quality of bacteriophage preparations, there are several major points to consider during the development, manufacturing, characterization, preclinical study and clinical trial of bacteriophage. The major issues include:
| · | bacteriophage preparation design (single agent versus phage mixes and wild-type phage versus genetically engineered phage); |
| · | proof of concept in development of bacteriophage products; |
| · | selectivity of bacteriophage replication and targeting to specific species of bacteria; |
| · | relevant animal models in preclinical studies; and |
| · | clinical safety and efficacy. |
Before testing any compounds with potential therapeutic value in humans, the biological product candidate enters the preclinical testing stage. Preclinical tests include laboratory evaluations of product biology, toxicity and formulation, as well as animal studies to assess the potential safety and activity of the biological product candidate. The conduct of the preclinical tests must comply with federal regulations and requirements including GLP. The sponsor must submit the results of the preclinical tests, together with manufacturing information, analytical data, any available clinical data or literature and a proposed clinical protocol, to the FDA as part of the IND. The IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA places the IND on a clinical hold within that 30 day time period. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. The FDA may also impose clinical holds on a product candidate at any time before or during clinical trials due to safety concerns or non-compliance. Accordingly, we cannot be certain that submission of an IND will result in the FDA allowing clinical trials to begin, or that, once begun, issues will not arise that suspend or terminate such clinical trial.
Clinical trials involve the administration of the product candidate to healthy volunteers or patients under the supervision of qualified investigators, generally physicians not employed by the sponsor. Clinical trials are conducted under protocols detailing, among other things, the objectives of the clinical trial, dosing procedures, subject inclusion and exclusion criteria and the parameters to be used to monitor subject safety. Each protocol must be submitted to the FDA. Clinical trials must be conducted in accordance with GCP requirements. Further, each clinical trial must be reviewed and approved by an independent institutional review board, or IRB, or ethics committee if conducted outside of the U.S., at or servicing each institution at which the clinical trial will be conducted. An IRB or ethics committee is charged with protecting the welfare and rights of trial participants and considers such items as whether the risks to individuals participating in the clinical trials are minimized and are reasonable in relation to anticipated benefits. The IRB or ethics committee also approves the informed consent form that must be provided to each clinical trial subject or his or her legal representative and must monitor the clinical trial until completed. We intend to use third-party Clinical Research Organizations, or CROs, to administer and conduct our planned clinical trials and will rely upon such CROs, as well as medical institutions, clinical investigators and consultants, to conduct our trials in accordance with our clinical protocols. The failure by any of such third parties to meet expected timelines, adhere to our protocols or meet regulatory standards could adversely impact the subject product development program and we remain legally responsible for compliance with applicable laws and regulations governing the conduct of these clinical trials.
Human clinical trials are typically conducted in three sequential phases that may overlap or be combined:
| · | Phase 1: The product candidate is initially introduced into healthy human subjects and tested primarily for safety and dosage tolerance. Absorption, metabolism, distribution and excretion may also be tested. |
| · | Phase 2: The product candidate is evaluated in a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product candidate for specific targeted diseases and to determine dosage tolerance, optimal dosage and dosing schedule. |
| · | Phase 3: Clinical trials are undertaken to further evaluate dosage, clinical efficacy and safety in an expanded patient population at geographically dispersed clinical trial sites. |
These clinical trials are intended to establish the overall risk/benefit ratio of the product and provide an adequate basis for product labeling. Generally, two adequate and well-controlled Phase 3 clinical trials are required by the FDA and other regulatory authorities for approval of a marketing application.
Post-approval studies, or Phase 4 clinical trials, may be requested by the FDA as a condition of approval and are conducted after initial marketing approval. These studies are used to gain additional experience from the treatment of patients in the intended therapeutic indication.
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and written safety reports must be submitted to the FDA and the investigators for serious and unexpected adverse events or any finding from tests in laboratory animals that suggest that there may be a significant risk for human subjects. The FDA or the sponsor or, if used, its data safety monitoring board may suspend a clinical trial at any time on various grounds, including a finding that the research subjects or patients are being exposed to an unacceptable health risk. Similarly, an IRB or ethics committee can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s or ethics committee’s requirements or if the pharmaceutical product has been associated with unexpected serious harm to patients. Suspension of a clinical trial due to safety risks attributed to the investigational product will result in termination of the trial and possibly others that are underway.
Concurrently with clinical trials, companies usually complete additional animal studies and must also develop additional information about the physical characteristics of the product candidate as well as finalize a process for manufacturing the product candidate in commercial quantities in accordance with cGMP requirements. To help reduce the risk of the introduction of adventitious agents or other impurities with the use of biological products, the PHS Act emphasizes the importance of manufacturing control for products whose attributes cannot be precisely defined. The manufacturing process must be capable of consistently producing quality batches of the product candidate and, among other things, the sponsor must develop methods for testing the identity, strength, quality, potency, and purity of the final biological product. Additionally, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that the biological product candidate does not undergo unacceptable deterioration over its shelf life.
United States Review and Approval Processes
In order to obtain approval to market a biological product in the United States, a BLA that provides data establishing to the FDA’s satisfaction the safety and effectiveness of the investigational product candidate for the proposed indication must be submitted to the FDA. The application includes all data available from nonclinical studies and clinical trials, including negative or ambiguous results as well as positive findings, together with detailed information relating to the product’s manufacture and composition, and proposed labeling, among other things. The testing and approval processes require substantial time and effort and there can be no assurance that the FDA will accept the BLA for filing and, even if filed, that any approval will be granted on a timely basis, if at all.
Each BLA must be accompanied by a significant user fee. The FDA adjusts the user fees on an annual basis. Fee waivers or reductions are available in certain circumstances, including a waiver of the application fee for the first application filed by a small business. Additionally, no user fees are assessed on BLAs for products designated as orphan drugs, unless the product also includes a non-orphan indication.
The FDA has 60 days from its receipt of a BLA to determine whether the application will be accepted for filing based on the agency’s threshold determination that the application is sufficiently complete to permit substantive review. The FDA may refuse to file any BLA that it deems incomplete or not properly reviewable at the time of submission and may request additional information. In this event, the BLA must be resubmitted with the additional information. The resubmitted application also is subject to review before the FDA accepts it for filing. After the BLA is accepted for filing, the FDA reviews it to determine, among other things, whether the proposed product is safe and effective for its intended use, has an acceptable purity profile, and whether the product is being manufactured in accordance with cGMP to assure and preserve the product’s identity, safety, strength, quality, potency, and purity. The FDA may refer applications for novel product candidates or those that present difficult questions of safety or efficacy to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved and, if so, under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions. The FDA may ultimately decide that the BLA does not satisfy the criteria for approval. If a product receives regulatory approval, the approval may be significantly limited to specific diseases and dosages or the indications for use may otherwise be limited, which could restrict the commercial value of the product. Further, the FDA may require that certain contraindications, warnings or precautions be included in the product labeling.
Special FDA Expedited Review and Approval Programs
The FDA has various programs, including Fast Track designation, accelerated approval and priority review, that are intended to expedite the process for the development and FDA review of drugs that are intended for the treatment of serious or life threatening diseases or conditions and demonstrate the potential to address unmet medical needs. The purpose of these programs is to provide important new drugs and biological products to patients earlier than under standard FDA review procedures.
To be eligible for a Fast Track designation, the FDA must determine, based on the request of a sponsor, that a product is intended to treat a serious or life threatening disease or condition and demonstrates the potential to address an unmet medical need, or if the drug or biological product qualifies as a qualified infectious disease product under the Generating Antibiotic Incentives Now Act, or GAIN Act. The FDA will determine that a product will fill an unmet medical need if it will provide a therapy where none exists or provide a therapy that may be potentially superior to existing therapy based on efficacy or safety factors. We intend to request Fast Track designation for our product candidates if applicable.
Fast Track designation applies to the combination of the product and the specific indication for which it is being studied. The sponsor of a new drug or biological may request the FDA to designate the drug or biologic as a Fast Track product at any time during the clinical development of the product. Unique to a Fast Track product, the FDA may consider for review sections of the marketing application on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the application, the FDA agrees to accept sections of the application and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the application.
Any product submitted to the FDA for marketing, including under a Fast Track program, may be eligible for other types of FDA programs intended to expedite development and review, such as priority review and accelerated approval. Any product is eligible for priority review if it has the potential to provide safe and effective therapy where no satisfactory alternative therapy exists or a significant improvement in the treatment, diagnosis or prevention of a disease compared to marketed products. The FDA will attempt to direct additional resources to the evaluation of an application for a new drug or biological product designated for priority review in an effort to facilitate the review. Additionally, a product may be eligible for accelerated approval. Drug or biological products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval, which means that they may be approved on the basis of adequate and well-controlled clinical trials establishing that the product has an effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit, or on the basis of an effect on a clinical endpoint other than survival or irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments.
As a condition of approval, the FDA may require a sponsor of a drug or biological product receiving accelerated approval to perform post-marketing studies to verify and describe the predicted effect on irreversible morbidity or mortality or other clinical endpoint, and the drug or biological product may be subject to accelerated withdrawal procedures. In addition, the FDA currently requires as a condition for accelerated approval pre-approval of promotional materials, which could adversely impact the timing of the commercial launch of the product. Fast Track designation, priority review and accelerated approval do not change the standards for approval but may expedite the development or approval process.
A sponsor can also request designation of a product candidate as a “breakthrough therapy.” A breakthrough therapy is defined as a drug or biological product that is intended, alone or in combination with one or more other drugs or biological products, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the biological product or drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. Drugs or biological products designated as breakthrough therapies are also eligible for accelerated approval. The FDA must take certain actions, such as holding timely meetings and providing advice, intended to expedite the development and review of an application for approval of a breakthrough therapy. We intend to request “breakthrough therapy” designation for our product candidates if applicable.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.
Patent Term Extension and Biosimilars
Depending upon the timing, duration and specifics of FDA approval of our drugs, some of our U.S. patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch Waxman Amendments. The Hatch Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one half the time between the effective date of an IND, and the submission date of an NDA or BLA, plus the time between the submission date of an NDA or BLA and the approval of that application. Only one patent applicable to an approved drug is eligible for the extension, and the extension must be applied for prior to expiration of the patent. The United States Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration.
Pediatric exclusivity is a type of marketing exclusivity available in the U.S. under the Best Pharmaceuticals for Children Act, or BPCA, which provides for an additional six months of marketing exclusivity may be available if a sponsor conducts clinical trials in children in response to a written request from the FDA, or a Written Request. If the Written Request does not include clinical trials in neonates, the FDA is required to include its rationale for not requesting those clinical trials. The FDA may request studies on approved or unapproved indications in separate Written Requests. The issuance of a Written Request does not require the sponsor to undertake the described clinical trials.
Biologics Price Competition and Innovation Act of 2009
The Biologics Price Competition and Innovation Act of 2009, or BPCIA, amended the PHSA to create an abbreviated approval pathway for two types of “generic” biologics — biosimilars and interchangeable biologic products, and provides for a twelve year data exclusivity period for the first approved biological product, or reference product, against which a biosimilar or interchangeable application is evaluated; however if pediatric clinical trials are performed and accepted by the FDA, the twelve year data exclusivity period will be extended for an additional six months. A biosimilar product is defined as one that is highly similar to a reference product notwithstanding minor differences in clinically inactive components and for which there are no clinically meaningful differences between the biological product and the reference product in terms of the safety, purity and potency of the product. An interchangeable product is a biosimilar product that may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product.
The biosimilar applicant must demonstrate that the product is biosimilar based on data from (1) analytical studies showing that the biosimilar product is highly similar to the reference product; (2) animal studies (including toxicity); and (3) one or more clinical trials to demonstrate safety, purity and potency in one or more appropriate conditions of use for which the reference product is approved. In addition, the applicant must show that the biosimilar and reference products have the same mechanism of action for the conditions of use on the label, route of administration, dosage and strength, and the production facility must meet standards designed to assure product safety, purity and potency.
An application for a biosimilar product may not be submitted until four years after the date on which the reference product was first approved. The first approved interchangeable biologic product will be granted an exclusivity period of up to one year after it is first commercially marketed, but the exclusivity period may be shortened under certain circumstances.
FDA Post-Approval Requirements
Maintaining substantial compliance with applicable federal, state, local, and foreign statutes and regulations requires the expenditure of substantial time and financial resources. Rigorous and extensive FDA regulation of new products continues after approval, particularly with respect to cGMP. We will rely on third parties for the production of commercial quantities of any products that we may commercialize. We and third party manufacturers of our products are required to comply with applicable requirements in the cGMPs, including quality control and quality assurance and maintenance of records and documentation. We cannot be certain that we or our present or future suppliers will be able to comply with the cGMP and other FDA requirements. Other post-approval requirements applicable to biological products include reporting of cGMP deviations that may affect the identity, potency, purity and overall safety of a distributed product, record-keeping requirements, reporting of adverse effects, reporting updated safety and efficacy information, and complying with electronic record and signature requirements. After a BLA is approved, the product also may be subject to official lot release. As part of the manufacturing process, the manufacturer is required to perform certain tests on each lot of the product before it is released for distribution. If the product is subject to official release by the FDA, the manufacturer submits samples of each lot of product to the FDA together with a release protocol showing a summary of the history of manufacture of the lot and the results of all of the manufacturer’s tests performed on the lot. The FDA also may perform certain confirmatory tests on lots of some products, such as viral vaccines, before releasing the lots for distribution by the manufacturer. In addition, the FDA conducts laboratory research related to the regulatory standards on the safety, purity, potency, and effectiveness of biological products.
Discovery of previously unknown problems or the failure to comply with the applicable regulatory requirements, by us or our suppliers, may result in restrictions on the marketing of a product or withdrawal of the product from the market as well as possible civil or criminal sanctions and adverse publicity. FDA sanctions could include refusal to approve pending applications, withdrawal of an approval, clinical hold, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, mandated corrective advertising or communications with doctors, debarment, restitution, disgorgement of profits, or civil or criminal penalties. Any agency or judicial enforcement action could have a material adverse effect on us.
Biological product manufacturers and other entities involved in the manufacture and distribution of approved products are required to register their facilities with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMPs and other laws. In addition, changes to the manufacturing process or facility generally require prior FDA approval before being implemented and other types of changes to the approved product, such as adding new indications and additional labeling claims, are also subject to further FDA review and approval.
Labeling, Marketing and Promotion
The FDA closely regulates the labeling, marketing and promotion of drugs and biological products, including direct-to-consumer advertising, promotional activities involving the internet, and industry-sponsored scientific and educational activities. While doctors are free to prescribe any product approved by the FDA for any use, a company can only make claims relating to safety and efficacy of a product that are consistent with FDA approval, and the company is allowed to actively market a product only for the particular use and treatment approved by the FDA. In addition, any claims we make for our products in advertising or promotion must be appropriately balanced with important safety information and otherwise be adequately substantiated. Failure to comply with these requirements can result in adverse publicity, warning letters, corrective advertising, injunctions and potential civil and criminal penalties.
Other Healthcare Laws and Compliance Requirements
In the United States, our activities are potentially subject to regulation by various federal, state and local authorities in addition to the FDA, including the Centers for Medicare and Medicaid Services, other divisions of the United States Department of Health and Human Services (e.g., the Office of Inspector General), the United States Department of Justice and individual United States Attorney offices within the Department of Justice and state and local governments.
International Regulation
In addition to regulations in the United States, we will be subject to a variety of foreign regulations governing clinical trials and commercial sales and distribution of our future products. Our manufacturing facility in Ljubljana, Slovenia is subject to inspection and regulation by JAZMP, the Slovenian agency that regulates and supervises pharmaceutical products in Slovenia. Whether or not we obtain FDA approval for a product, we must obtain approval of a product by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval. The requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from country to country.
Under European Union regulatory systems, marketing authorizations may be submitted either under a centralized or a mutual recognition procedure. The centralized procedure, which is compulsory for medicinal products produced by biotechnology or those medicinal products containing new active substances for specific indications such as the treatment of AIDS, cancer, neurodegenerative disorders, diabetes, viral diseases and designated orphan medicines and optional for other medicines which are highly innovative. Under the centralized procedure, a marketing application is submitted to the European Medicines Agency where it will be evaluated by the Committee for Medicinal Products for Human Use and a favorable opinion typically results in the grant by the European Commission of a single marketing authorization that is valid for all European Union member states within 67 days of receipt of the opinion. The initial marketing authorization is valid for five years, but once renewed is usually valid for an unlimited period.
Pricing and Reimbursement
Although none of our product candidates has been commercialized for any indication, if they are approved for marketing, commercial success of our product candidates will depend, in part, upon the availability of third-party reimbursement from payors at the federal, state and private levels. Third party payors include government healthcare programs, such as Medicare and Medicaid, private health insurers and managed-care plans. We anticipate third party payors will provide reimbursement for our products. However, these third party payors are increasingly challenging the price and examining the cost effectiveness of medical products and services. In addition, significant uncertainty exists as to the reimbursement status of newly approved healthcare products. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the cost effectiveness of our products. Our product candidates may not be considered cost effective. It is time consuming and expensive for us to seek reimbursement from third party payors. Reimbursement may not be available or sufficient to allow us to sell our products on a competitive and profitable basis.
We expect that there will continue to be a number of federal and state proposals to implement governmental pricing controls and limit the growth of healthcare costs, including the cost of prescription drugs.
In addition, in some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the European Union provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the European Union do not follow price structures of the U.S. and generally tend to be significantly lower.
RISK FACTORS
Investment in our stock involves a high degree of risk. You should consider carefully the risks described below, together with other information in this Current Report on Form 8-K and other public filings with the SEC, before making investment decisions regarding our stock. If any of the following events actually occur, our business, operating results, prospects or financial condition could be materially and adversely affected. This could cause the trading price of our common stock to decline and you may lose all or part of your investment. Additional risks facing our company are described under the heading “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2016, or Annual Report, as filed with the SEC. The risk factors below do not appear as separate risk factors in, or reflect changes to the similarly titled risk factors included in, our Annual Report. Moreover, the risks described below and in our Annual Report are not the only ones that we face. Additional risks not presently known to us or that we currently deem immaterial may also affect our business, operating results, prospects or financial condition.
Results from preclinical studies and Phase 1 or 2 clinical trials of our product candidates or from compassionate-use treatments may not be predictive of the results of later stage clinical trials.
Preclinical studies, including studies of our product candidates in animal disease models, may not accurately predict the result of human clinical trials of those product candidates. In particular, promising animal studies suggesting the efficacy of prototype phage products in the treatment of bacterial infections, such as P. aeruginosa and S. aureus, may not predict the ability of these products to treat similar infections in humans. Despite promising data in our completed Phase 1 clinical trials, our phage technology may be found not to be efficacious in treating bacterial infections alone or in combination with other agents, when studied in later-stage clinical trials.
In addition, we have used and plan to continue to use our bacteriophage technology in the area of personalized medicine under compassionate-use guidelines, which permit the use of phage therapy outside of clinical trials, beginning in Australia and then expanding to the United States and potentially other countries. Despite prior compassionate use successes, no assurance can be given that will have similar compassionate-use treatment successes in the future. Compassionate use is a term that is used to refer to the use of an investigational drug or therapy outside of a clinical trial to treat a patient with a serious or immediately life-threatening disease or condition who has no comparable or satisfactory alternative treatment options. Regulators often allow compassionate use on a case-by-case basis for an individual patient or for defined groups of patients with similar treatment needs. In some countries, such as Australia, the treating physician can administer treatment under compassionate-use guidelines without pre-approval from the applicable regulatory authority.
To satisfy FDA or foreign regulatory approval standards for the commercial sale of our product candidates, we must demonstrate in adequate and controlled clinical trials that our product candidates are safe and effective. Success in early clinical trials, including Phase 1 and Phase 2 trials, or in our compassionate-use program does not ensure that later clinical trials will be successful. Our initial results from early stage clinical trials or our compassionate-use program also may not be confirmed by later analysis or subsequent larger clinical trials. A number of companies in the pharmaceutical industry have suffered significant setbacks in advanced clinical trials, even after obtaining promising results in earlier clinical trials and most product candidates that commence clinical trials are never approved for commercial sale.
Our personalized phage therapies strategy may not be successful, which in turn could adversely affect our business.
Our personalized phage therapies strategy involves providing phage therapy under compassionate-use guidelines to patients outside of clinical trials with antibiotic-resistant infections who have few or no other therapeutic options. We believe this strategic approach will not only provide potential benefit to patients to whom we are able to provide personalized phage therapies under the compassionate-use guidelines, but also provide the clinical data from these compassionate-use cases that we expect to support the potential validation of the clinical utility of phage therapy and inform our future discussions with the FDA in 2018 or later on defining a potential path to market approval. However, this program is subject to numerous risks and uncertainties, including the following:
| · | We have not established a cost reimbursement structure or otherwise entered into an arrangement that would at least offset our manufacturing costs for our phage therapies that may be administered to patients under compassionate-use guidelines. Increasing demand for our phage therapies in compassionate-use cases could result in significant costs to us. |
| · | Responding to compassionate-use requests could divert attention of our personnel and use manufacturing resources that could otherwise be deployed in other development program activities. |
| · | Compassionate-use treatment data may not establish proof-of-concept, and the FDA or other regulatory authorities may not accept compassionate-use data as sufficient clinical validation in support of our regulatory approval efforts, which could materially delay and increase the costs of our product development and commercialization activities. |
| · | Patient access to phage therapy will be provided on an individual basis where physicians will make an application or post-treatment notification to the applicable regulatory authorities on a patient-by-patient basis. This can impose a significant administrative burden on participating physicians, who may be resistant to navigating a process with which they are unfamiliar. |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: May 1, 2017 | AmpliPhi Biosciences Corporation | ||
| By: | /s/ Steve R. Martin | ||
| Name: | Steve R. Martin | ||
| Title: | Chief Financial Officer | ||
