Attached files
| file | filename |
|---|---|
| EX-10.42 - EXHIBIT 10-42 - Enumeral Biomedical Holdings, Inc. | s105622_ex10-42.htm |
| EX-32.2 - EXHIBIT 32-2 - Enumeral Biomedical Holdings, Inc. | s105622_ex32-2.htm |
| EX-32.1 - EXHIBIT 32-1 - Enumeral Biomedical Holdings, Inc. | s105622_ex32-1.htm |
| EX-31.2 - EXHIBIT 31-2 - Enumeral Biomedical Holdings, Inc. | s105622_ex31-2.htm |
| EX-31.1 - EXHIBIT 31-1 - Enumeral Biomedical Holdings, Inc. | s105622_ex31-1.htm |
| EX-23.1 - EXHIBIT 23-1 - Enumeral Biomedical Holdings, Inc. | s105622_ex23-1.htm |
| EX-21.1 - EXHIBIT 21-1 - Enumeral Biomedical Holdings, Inc. | s105622_ex21-1.htm |
| EX-10.7 - EXHIBIT 10-7 - Enumeral Biomedical Holdings, Inc. | s105622_ex10-7.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
| þ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2016
or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from __________________ to __________________________
Commission file number: 000-55415
ENUMERAL BIOMEDICAL HOLDINGS, INC.
(Exact name of registrant as specified in its charter)
| Delaware | 99-0376434 |
| (State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
| 200 CambridgePark Drive, Suite 2000 Cambridge, Massachusetts |
02140 |
| (Address of principal executive offices) | (Zip Code) |
| Registrant's telephone number, including area code: | (617) 945-9146 |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Name of each exchange on which registered |
| None | Not applicable |
Securities registered pursuant to Section 12(g) of the Act:
Common Stock, $0.001 Par Value Per Share
(Title of class)
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. ¨ Yes þ No
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. ¨ Yes þ No
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. þYes ¨ No
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Website, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). þYes ¨ No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of the registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. þ Yes ¨ No
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company:
| Large accelerated filer | ¨ | Accelerated filer | ¨ |
| Non-accelerated filer | ¨ | Smaller reporting company | þ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act) ¨ Yes þ No
State the aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day of the registrant's most recently completed second fiscal quarter:
The aggregate market value of the registrant’s common stock, $0.001 par value per share (“Common Stock”), held by non-affiliates of the registrant, based on the last reported sale price of the Common Stock on the OTCQB at the close of business on June 30, 2016, was approximately $9,389,712. For purposes hereof, shares of Common Stock held by each executive officer and director of the registrant and entities affiliated with such executive officers and directors have been excluded from the foregoing calculation because such persons and entities may be deemed to be affiliates of the registrant. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
Indicate the number of shares outstanding of each of the registrant's classes of Common Stock, as of the latest practicable date: 128,409,788 shares of Common Stock are issued and outstanding as of March 24, 2017.
DOCUMENTS INCORPORATED BY REFERENCE
None.
TABLE OF CONTENTS
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING INFORMATION
This Annual Report on Form 10-K contains forward-looking statements, including, without limitation, within the sections captioned “Item 1. - Business,” “Item 1A. - Risk Factors,” and “Item 7. - Management’s Discussion and Analysis of Financial Condition and Results of Operations.” Any and all statements contained in this Annual Report on Form 10-K that are not statements of historical fact may be deemed forward-looking statements. Terms such as “may,” “might,” “would,” “should,” “could,” “project,” “estimate,” “pro-forma,” “predict,” “potential,” “strategy,” “anticipate,” “attempt,” “develop,” “plan,” “help,” “believe,” “continue,” “intend,” “expect,” “future,” and terms of similar import (including the negative of any of the foregoing) may be intended to identify forward-looking statements, however, not all forward-looking statements may contain one or more of these identifying terms. Forward-looking statements in this Annual Report on Form 10-K may include, without limitation, statements regarding (i) the plans and objectives of management for future operations, including plans or objectives relating to the development of commercially viable pharmaceuticals, (ii) a projection of income (including income/loss), earnings (including earnings/loss) per share, capital expenditures, dividends, capital structure or other financial items, (iii) our future financial performance, including any such statement contained in a discussion and analysis of financial condition by management or in the results of operations included pursuant to the rules and regulations of the Securities and Exchange Commission (the “SEC”), and (iv) the assumptions underlying or relating to any statement described in points (i), (ii) or (iii) above.
The forward-looking statements are not meant to predict or guarantee actual results, performance, events or circumstances and may not be realized because they are based upon our current projections, plans, objectives, beliefs, expectations, estimates and assumptions and are subject to a number of risks and uncertainties and other influences, many of which we have no control over. Actual results and the timing of certain events and circumstances may differ materially from those described by the forward-looking statements as a result of these risks and uncertainties. Factors that may influence or contribute to the inaccuracy of the forward-looking statements or cause actual results to differ materially from expected or desired results may include, without limitation, our inability to obtain adequate financing, the significant length of time associated with drug development and related insufficient cash flows and resulting illiquidity, our inability to expand our business, significant government regulation of pharmaceuticals and the healthcare industry, lack of product diversification, volatility in the price of our raw materials, existing or increased competition, results of arbitration and litigation, stock volatility and illiquidity of our securities, and our failure to implement effectively our business plans or strategies. A description of some of the risks and uncertainties that could cause our actual results to differ materially from those described by the forward-looking statements in this Annual Report on Form 10-K appears in the section captioned “Risk Factors” and elsewhere in this Annual Report on Form 10-K.
Readers are cautioned not to place undue reliance on forward-looking statements because of the risks and uncertainties related to them and to the risk factors. We disclaim any obligation to update the forward-looking statements contained in this Annual Report on Form 10-K to reflect any new information or future events or circumstances or otherwise, except as required by law.
Readers should read this Annual Report on Form 10-K in conjunction with the discussion under the caption “Risk Factors,” our consolidated financial statements and the related notes thereto in this Annual Report on Form 10-K, and other documents which we may file from time to time with the SEC.
OTHER PERTINENT INFORMATION
Unless the context indicates otherwise, all references in this Annual Report on Form 10-K to “Enumeral Biomedical,” the “Company,” “we,” “us” and “our” refer to Enumeral Biomedical Holdings, Inc., and its wholly-owned subsidiaries, Enumeral Biomedical Corp. and Enumeral Securities Corporation; and references to “Enumeral” refer to Enumeral Biomedical Corp.
Unless specifically set forth to the contrary, the information which appears on our website at www.enumeral.com is not part of this Annual Report on Form 10-K.
Description of Business
Overview
We are a biopharmaceutical company focused on discovering and developing novel antibody immunotherapies that help the immune system fight cancer and other diseases. We utilize a proprietary platform technology that facilitates the rapid high resolution measurement of immune cell function within small tissue biopsy samples. Our initial focus is on the development of a pipeline of next generation monoclonal antibody drugs targeting established and novel immuno-modulatory receptors.
The concept of stimulating the immune system to fight cancer was first advanced more than a century ago, but it is only recently that the field of immuno-oncology has seen clinical success, with marketing approvals being granted for antibodies that block CTLA-4 (Yervoy® (ipilimumab)) and PD-1 (Keytruda® (pembrolizumab) and Opdivo® (nivolumab)), and PD-L1 (Tecentriq® (atezolizumab)). Use of these drugs has established that durable anti-tumor responses can be elicited in some patients by blocking the checkpoints that normally suppress the human immune response against cancer cells. The success of these drugs suggests that immuno-oncology may fundamentally alter the course of cancer treatment.
In our lead antibody program, we have characterized certain anti-PD-1 antibodies, or simply “PD-1 antibodies,” using patient biopsy samples, in an effort to identify next generation PD-1 antagonists with enhanced selectivity for the immune effector cells that carry out anti-tumor functions. We have identified two antagonist PD-1 antibodies that inhibit PD-1 activity in different ways. The distinction is that one of the antibodies (ENUM 388D4) blocks binding of the ligand PD-L1 to PD-1, while the other antibody (ENUM 244C8) does not inhibit PD-L1 binding. However, both display activity in various biological assays. In addition to our PD-1 antibody program, we are developing antibody drug candidates for a number of immunomodulatory protein targets, including TIM-3, CD39, and TIGIT. We are also pursuing several antibody programs for which we have not yet publicly disclosed the targets.
Our proprietary platform technology, exclusively licensed from the Massachusetts Institute of Technology, or MIT, is a microwell array technology that detects secreted molecules (such as antibodies and cytokines) and cell surface markers, at the level of single, live cells – and enables recovery of single, live cells of interest. The platform technology can be used to achieve at least three separate, but complementary, objectives. First, we use the platform to rapidly produce antibody libraries with high diversity. Second, the platform has the potential to guide rational selection of lead candidates derived from these libraries, through characterization of immune function at the level of single cells from human biopsy samples. Third, it has the potential to identify patients more likely than others to benefit from treatment with a given therapeutic antibody. Thus, our platform is a multipurpose tool that is valuable for activities ranging from antibody discovery to target discovery to patient stratification in clinical development. The platform yields multidimensional, functional read-outs from single live cells, such as tumor infiltrating lymphocytes, or TILs, from human tumor biopsy samples, and it enables us to examine the responses of different classes of human immune cells to treatment with immuno-modulators in the context of human disease, as opposed to animal models of disease.
To date, our proof-of-concept corporate collaborations have provided minimal revenue. However, our business has not generated (nor do we anticipate that in the foreseeable future it will generate) the cash necessary to finance our operations. We expect to continue to incur losses and negative cash flows from operations for the foreseeable future, and we will require additional capital to continue our operations beyond May 2017.
On December 12, 2016, we consummated an offer to amend and exercise certain outstanding warrants to purchase an aggregate of 21,549,510 shares of our common stock originally issued to investors who participated in our July 31, 2014 private placement financing (the “Warrant Tender Offer”). Pursuant to the Warrant Tender Offer, an aggregate of 6,863,000 warrants were tendered by their holders, and we received gross proceeds in the amount of $3,431,500. As a result, we issued 27,452,000 shares of our common stock to the holders who amended and exercised their PPO Warrants in the Warrant Tender Offer. For additional information on the Warrant Tender Offer, please refer to “Merger and Related Transactions - Warrant Tender Offer” below.
| 1 |
As of December 31, 2016 we had cash and cash equivalents totaling $3,162,400, excluding restricted cash.
As of the date of this filing, we believe that we only have sufficient liquidity to fund operations into May 2017. We are currently exploring a range of potential transactions, which may include public or private equity offerings, debt financings, collaborations and licensing arrangements, and/or other strategic alternatives, including a merger, sale of assets or other similar transactions. If we are unable to raise additional capital through one or more of the means listed above prior to the end of May 2017, we will face substantial liquidity problems and will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets.
No assurance can be given that additional financing or strategic alliances and licensing arrangements will be available when needed or that, if available, such financing could be obtained on terms favorable to us or our stockholders. See Item 1A. - “Risk Factors.”
Our Strategy
In general, our strategy is to exploit our proprietary platform technology and build a pipeline of next generation therapeutics targeting the immune system for the treatment of cancer and other diseases through in-house discovery and corporate development. Key elements of our strategy include:
| · | Platform Technology. We seek to translate biological complexity into next generation therapeutics using our proprietary single cell microengraving platform technology. Our platform technology enables us to examine rare cells in human biopsies to understand what different cells are secreting and how they respond to different stimuli. Eventually, our platform may enable us to identify which patients are more likely to benefit from such treatments. We are currently pursuing this approach against a number of immunomodulatory protein targets, including PD-1, TIM-3, CD39, and others. We are also currently exploring the potential of profiling human tissue to identify novel monoclonal antibody therapeutics. |
| · | Internal Pipeline Development. We are advancing our internally developed pipeline and characterizing molecules utilizing our proprietary platform technology to enhance the value of our assets at an early stage of development for potential partnering or licensing. |
| · | Pipeline Enhancement. We are exploring potential arrangements to enhance our pipeline of next-generation therapeutics, including the acquisition or licensing of complementary assets that are at a more advanced stage of development than our internally developed pipeline. |
| · | Collaborations. We believe our proprietary technology platform positions us for collaborations with other biotechnology companies as well as academic institutions, which will further validate our science and business model and may also provide non-dilutive sources of capital for our company. |
| 2 |
Our Programs
Anti-PD-1 Immunotherapy
Our lead program involves antibodies against the “Programmed Cell Death” receptor, commonly known as “PD-1.” Our PD-1 program has yielded twenty-five families of PD-1 antibodies. We are conducting functional characterization of our lead antibodies, using patient biopsy samples, in an effort to identify next generation PD-1 antagonists with enhanced selectivity for the immune effector cells that have anti-tumor functions.
We have employed ex vivo methods to examine how TILs derived from human lung biopsies respond to various PD-1 antibodies. Measurements include production of interferon gamma (IFNγ), which is a cytokine indicative of T cell activation. We have found that approximately half of assayed biopsies harbored TILs responsive to different PD-1 antibodies. We have also found that TILs isolated from lung tumor biopsies that display elevated TIM-3 expression levels have lower responsiveness to the effects of PD-1 antibodies in ex vivo assays.
Our two lead PD-1 antibodies, 388D4 and 244C8, both enhance T cell function through reversal of PD-1-dependent immunosuppression, but they appear to act on PD-1 through different mechanisms. Antibody 388D4 blocks binding of PD-L1 to PD-1 (as do the currently-marketed PD-1 antibodies), while 244C8 does not block binding of PD-L1 to PD-1. Using TILs derived from human lung biopsy samples, we have performed experiments comparing the activities of 244C8 and a currently marketed PD-1 antibody, in reversing T cell exhaustion. In some experiments, 244C8 has appeared to restore T cell function to a higher level than did a currently-marketed PD-1 antibody, which we used as a comparator. We have also presented research results in which 244C8 has appeared to elicit cytokine secretion from cell types associated with innate immunity. Results from other experiments are consistent with the observed pattern of cytokine secretion being dependent on the activity of CD40L, a receptor involved in T cell engagement with dendritic cells, which are involved in anti-tumor immunity.
Despite the clinical breakthroughs achieved with the currently marketed PD-1 and PD-L1 antibody drugs, Opdivo® (nivolumab), Keytruda® (pembrolizumab), and Tecentriq® (atezolizumab), those drugs are not effective in all patients, or in all cancers. We hope to develop anti-PD-1 treatments for certain solid tumor indications, where effective therapies do not exist.
Anti-TIM-3 Immunotherapy
In early 2015, we initiated screening for antibodies against the “T Cell Immunoglobulin and Mucin Protein 3,” commonly known as TIM-3. This program (designated internally as ENUM005) has yielded 42 families of TIM-3 antibodies, which represent high diversity with respect to sequence relatedness. Our lead selection work in this program is ongoing.
Our future plans for our PD-1 and TIM-3 programs, as well as our other antibody programs described below, will depend on our ability to raise additional capital through public or private equity offerings, debt financings, or strategic collaborations and licensing arrangements, and/or our success in promptly establishing a strategic alternative that is in our stockholders’ best interests.
Other Antibody Programs
In addition to our PD-1 program and TIM-3 program, we have several other antibody programs, including CD39 (ENUM008) and TIGIT (ENUM009). We are also pursuing several antibody programs for which we have not yet publicly disclosed the targets.
| · | CD39 (ENUM008). CD39 is an ectonucleotidase involved, along with CD73, in the generation of adenosine from adenosine triphosphate (ATP). A reduction of free adenosine would be expected to enhance immune response in cancer by reducing adenosine mediated suppression of immune cells expressing the immunosuppressive adenosine A2A receptor. |
| · | TIGIT (ENUM009). TIGIT is believed to be a negative immunomodulatory receptor expressed on lymphoid cells. Our objective for the ENUM009 program is to generate therapeutic antibody candidates that block the interaction of TIGIT (on NK or T cells) with cognate ligand on accessory cells. We have initiated immunization with an optimized murine immune system model. |
| 3 |
Our Proprietary Platform Technology
In our drug discovery programs, our platform enables us to use primary cells from fully intact immune systems in various strains of mice (or other species) for antibody screening. As discussed in the Antibody Discovery section below, using primary cells from a fully intact immune system provides a significant advantage over established technologies such as phage display and mouse strains genetically engineered to produce human antibodies, because those technologies place certain limits on recovery of functional antibody diversity.
Our platform is built around a proprietary microwell array chip, which contains 84,672 spatially addressable sub-nanoliter microwells. Our microwell array chips are manufactured exclusively for us by a third-party provider, using custom molds and industrialized processes. In conjunction with the microwell array chips, we employ commercial laboratory equipment and proprietary software to create an integrated system.
Using this system, we can analyze cells from various sources, including human peripheral blood mononuclear cells (PBMCs), bone marrow, tumor biopsy, mucosal biopsy, and cerebrospinal fluid (CSF). Figures 1A and 1B below illustrate the three major elements of our platform:
| 1) | Cell culture or cell handling, which includes preparation (or dissociation) of cells from primary tissue samples. Our system generally avoids complex cell handling processes. This provides for rapid measurements involving fragile, individual live cells (element 2, below), which can be difficult with technologies other than ours; |
| 2) | Measurement of cellular functioning at the level of single cells, including independent identification of cell surface markers and secreted proteins, such as antibodies and cytokines; and |
| 3) | Retrieval of individual cells, after which they can be subjected to molecular analyses such as reverse transcription polymerase chain reaction, or RT-PCR, RNA sequencing, and other molecular biological methods. These data link cell type (surface markers), and cell function (identified by secretion), to underlying genomic information. We believe that without our platform, this linkage of information at the single cell level would be difficult or impossible to achieve with cell-limited biopsy material from human patients. |
The microwells containing individual cells allow for rapid and sensitive detection of secreted factors, and eliminate the need for common procedures such as cell proliferation (for example, Enzyme-Linked ImmunoSpot Assay, or ELISPOT), cell fusion (hybridoma techniques), and cell sorting and enrichment (such as fluorescence activated cell sorting, or FACS, and B cell cloning), all of which can bias results.
Cell Loading
 |
 |
 |
| Simple pipetting step achieves desired cell density, e.g. single cell occupancy | Secreted proteins bind to capture antibodies on cover slide | Spatially-registered custom protein microarray allows identification of cells with desired functionality for recovery |
| 4 |
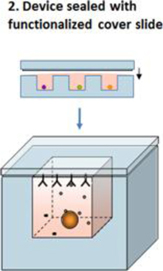
Figure 1A: Platform Overview
Cells are loaded directly on the microwell array without manipulations that could lead to loss of cellular function or viability. This process effectively preserves the entire original population of cells for analysis. Loading is rapid, which is critical for analysis of live primary cells derived from limited biopsy materials. A glass cover slide treated with commercially available capture reagents specific to secreted proteins of interest is sealed across the top of the chip. Multiple capture reagents can be used at the same time. We routinely analyze four secreted factors in parallel, across the entire cell population. The sealed chip is incubated (usually between 2 and 6 hours). Proteins secreted in each microwell during the incubation period are captured on the slide. This yields a spatially registered protein array corresponding to each position (microwell) on the chip.
Analysis & Recovery

Figure 1B: Platform Overview
The cover slide is treated with conventional fluorescently-labeled detection reagents, and array scanners are used to identify proteins secreted from individual cells. In parallel, the cells are labeled with reagents specific for cell surface protein markers and examined through high-speed multiplexed imaging cytometry to determine cell lineage. Data are integrated to produce independent, unbiased measurements of lineage and function for the cells within each microwell. Based on this information, cells are retrieved by automated micromanipulation.
Antibody Discovery
Our antibody discovery programs exploit our proprietary, high diversity antibody libraries by screening primary cells from murine tissues such as splenocytes or bone marrow cells, following target-specific immunizations that are performed for us by a third party. In contrast to the limited diversity of human antibodies from transgenic mouse platforms, we employ multiple mouse strains with intact, fully diverse murine immune systems, and we use multiple adjuvants during our immunization campaigns. This avoids loss in the natural diversity of the antibodies produced in the mouse for antibody screening and selection. Our platform enables direct screening of antibody-secreting cells without any requirement for intervening cell fusion, enrichment or sorting. This direct screening enables the exceptional degree of antibody diversity provided by our system.
| 5 |
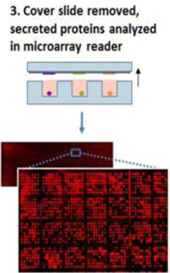
In our screening process, after secretion and capture of the antibodies, we measure the isotype, amount secreted, target specificity, and relative target affinity. This information is used to derive a priority list for retrieval of individual cells that secreted the antibodies. Cells are retrieved using an automated commercial system customized to our platform and the genes encoding the secreted antibodies are cloned through single cell RT-PCR protocols which we have optimized. Our standard procedures can result in approximately 80% recovery of paired heavy-chain and light-chain genes. Figure 2 illustrates the binding of a secreted antibody to a functionalized cover slide in the context of antibody discovery.

Figure 2: Detection of antibody secretion events from single cells.
The design of our proprietary microwell array provides for spatial registration of the secreted antibodies with the microwell containing the secreting cells. This enables retrieval of specific cells of interest. After cloning and sequencing of the antibody genes, bioinformatics analysis enables classification of the antibodies into “families.”
Cellular Response Profiling
Cellular response profiling is what we call our analysis of human tissue samples to measure specific functions of individual cells, and to elucidate how cells in the human immune system respond to a given set of circumstances. As illustrated in Figure 3, we can measure the frequency and functional status of tumor-infiltrating lymphocytes to generate a functional immune “signature.” Such signatures might be generated, for example, using biopsies from patients that have responded to a particular treatment, and compared to signatures generated using biopsies from non-responders. This primary biopsy-based approach also can be used to compare among a series of antibody drug candidates, to profile the immune responses they elicit ex vivo, and to facilitate selection of those with the best immune modulation properties. Single-cell functional profiling enables simultaneous and unbiased analysis of cellular parameters from individual cells within the same population of cells, including antibodies, cytokines, other proteins, and cell lineage.
| 6 |

Figure 3: Profiling of human tissue samples for immunological signatures.
We believe that there are measurable differences in how cells in the human immune system respond to different antibodies, even when those antibodies bind the same target (e.g., PD-1). A composite of those differences, as measured at the level of single cells, is what we call a cellular response profile. The differences in cellular response profiles may translate into differences in how certain patients respond to certain therapies.
Cellular response profiling is a versatile method that can be employed in various ways. Cellular response profiling complements our antibody programs. Once a desired cellular response profile has been defined, testing of human tissue samples on our platform can support either (or both) of two different courses of action: (1) screening for patients with tumors that display a cellular response profile indicating suitability for treatment with a particular drug; or (2) screening for antibodies that elicit a desired cellular response profile in samples from different patient subpopulations, i.e., screening for an antibody that produces higher initial response rates. Whether screening antibodies or screening patients, the concept and principles are the same. Accumulated data from cellular response profiling may lead to valuable insight into details of the mechanisms of action of different antibodies in different tissues or disease indications. In turn, this may guide selection of particular drug combinations for testing in clinical trials. These points are illustrated by our studies using ex vivo TIL analysis in our PD-1 and TIM-3 programs, as described above.
Licenses and Collaborations
Pieris
In June 2016, we entered into a Definitive License and Transfer Agreement (the “Definitive Agreement”) with Pieris Pharmaceuticals, Inc. and Pieris Pharmaceuticals GmbH (collectively, “Pieris”). Pursuant to the terms and conditions of the Definitive Agreement, Pieris is licensing from us specified intellectual property related to our anti-PD-1 antibody program 388D4 for the potential development and commercialization by Pieris of novel multispecific therapeutic proteins comprising fusion proteins based on Pieris’ Anticalins® class of therapeutic proteins and our antibodies in the field of oncology. We had previously entered into a License and Transfer Agreement (the “License Agreement”) with Pieris in April 2016, which the Definitive Agreement superseded. Pieris paid us an upfront license fee in the amount of $250,000 in connection with execution of the License Agreement, and paid us a $750,000 license maintenance fee to continue the licensing arrangements under the License Agreement.
Under the Definitive Agreement, we have granted Pieris an option until May 31, 2017 to license specified patent rights and know-how of ours covering two additional undisclosed antibody programs on the same terms and conditions as for our 388D4 anti-PD-1 antibody (each, a “Subsequent Option”). Pieris may exercise the Subsequent Options separately and on different dates during the option period. Pieris will pay us additional license fees in the event that Pieris exercises one or both Subsequent Options.
| 7 |
Merck
In December 2014, we entered into a collaboration for a term of up to 24 months with Merck Sharp & Dohme Corp., or Merck. Pursuant to our study agreement with Merck, we conducted a specified research program using our platform technology to identify functional response of single cell types in cancer in the presence or absence of Merck’s proprietary immunomodulatory receptor (IMR) modulators. In February 2016, we and Merck subsequently amended the work plan under the study agreement to include non-small cell lung cancer tissue samples.
In this collaboration, Merck reimbursed us for the cost of performing the work specified in the agreement, for up to a specified number of full time employees, at an agreed annual rate. In addition, the study agreement provides that Merck will make certain milestone payments to us upon the completion of specified objectives set forth in the Merck agreement and related work plan. In September 2015, we announced the achievement of the first milestone. Merck retains ownership of the results generated from the studies on the IMR modulators identified by Merck, and we retain a royalty-free, non-exclusive, non-sublicensable license to use the study results for our own internal research purposes.
National Cancer Institute (NCI)
In September 2014, we were awarded a Phase II Small Business Innovation Research contract from the NCI for up to $999,967 over two years. As part of this Phase II contract, we have developed and deployed an automated prototype system for human tissue immuno-oncology profiling at the Ragon Institute of Massachusetts General Hospital, MIT and Harvard University and also at Memorial Sloan Kettering Cancer Center. We are collaborating with the laboratory of Douglas S. Kwon, M.D., Ph.D. at the Ragon Institute, and we are collaborating with the laboratory of Jedd D. Wolchok, M.D., Ph.D. at Sloan-Kettering.
In September 2016, we entered to an amendment to our NCI contract to extend the period of performance under the contract to March 15, 2017. We do not anticipate any further amendments to the contract.
M.D. Anderson
In January 2016, we entered into a collaborative research and development agreement with The University of Texas M.D. Anderson Cancer Center, or MDACC. Under this agreement, we and MDACC plan to collaborate on the discovery and development of novel monoclonal antibodies against selected targets in immuno-oncology, utilizing our antibody discovery and immune profiling platform and MDACC’s preclinical and development expertise and infrastructure.
We and MDACC will share the costs of research and development activities necessary to take development candidates through successful completion of a Phase I clinical trial. The agreement provides for a structure whereby we and MDACC are each granted the right to receive a percentage of the net income from product sales or any payments associated with licensing or otherwise partnering a program with a third party.
The agreement contemplates that, in conjunction with the research and development activities, either we or MDACC will seek to enter into licensing transactions with third parties to engage in development or commercialization activities involving the identified antibodies, subject to approval by the other party. We and MDACC also have the option of continuing collaboration efforts into advanced development and commercialization, with one party taking the lead, and both parties sharing costs equally.
We and MDACC each has the right to opt out and cease further funding of future collaboration activities with respect to a collaboration antibody at specified stages, provided that a party which elects to opt out may continue to be responsible for certain expenses incurred prior to such election. A party that exercises its opt out right will also have its percentage of net income from product sales or payments associated with such collaboration antibody program adjusted in accordance with the terms of the agreement.
To date, we have not yet commenced work under the MDACC agreement.
| 8 |
Intellectual Property
From MIT, we have in-licensed a patent portfolio (which includes patents owned by Harvard or co-owned by MIT and The Whitehead Institute, or MIT and Massachusetts General Hospital) that we refer to as the Platform Portfolio. It broadly covers our platform technology in the United States, and also covers certain aspects of it in certain foreign countries. The license, referred to as the Platform License, gives us exclusive worldwide rights under patents and patent applications covering the platform technology. The Platform License provides us with worldwide rights under the Platform Portfolio, in all fields of use. This enables us to use the platform in drug discovery and development (including partnered programs), and the right to commercialize its application for purposes such as diagnostics. The Platform License obligates us to achieve certain diligence milestones within certain time frames.
The Platform Portfolio protects our platform technology through eight issued U.S. patents that cover various aspects of the platform technology, including the microarray apparatus, microarrays with micro-channels, a method of capturing products secreted from single cells, a method of making a microarray of secreted antibodies, microarray methods for screening antibodies, single cell cytotoxicity assays, methods of immuno-profiling, and methods of performing RT-PCR from single cells. There are also corresponding issued patents or pending applications in certain foreign jurisdictions. The basic patent coverage of our platform technology in the United States will expire in 2027. The U.S. patent on microarrays with micro-channels will expire in 2029.
We have begun to build our own patent portfolio to protect our novel antibodies and other inventions. In December 2014, we filed two provisional patent applications of which we are the sole owners. One application covers a method of using our platform for rapid identification of compounds that elicit specific, desired cellular response profiles. The second application covers novel PD-1 antibodies (and their therapeutic use), which were identified using our platform, and tested in cell-based assays. The application covers 26 individual heavy chain variable sequences and 27 individual light chain variable sequences, as well as matched pairs of heavy and light chains. Both of these provisional applications were updated and re-filed as non-provisional applications in December 2015. In addition, in March 2017 we filed a new provisional patent application that covers our TIM-3 antibodies. As we discover additional antibodies against selected therapeutics targets, we plan to file patent applications covering those antibodies and their therapeutic uses. We also plan to file patent applications that cover novel research tools and methods that we discover, and any improvements that we make in the platform technology. For example, in March 2016, we filed a new patent application covering certain small accessories that we designed to facilitate processing and imaging of microwell arrays. In addition to filing and prosecuting patent applications in the United States, we plan to file counterpart applications in Australia, Canada, Europe, Japan, and additional jurisdictions, in cases where we think such filings are likely to be cost-effective and important to our business objectives.
For some aspects of our proprietary technology, trade secret protection is more suitable than patent protection. For example, certain proprietary bioinformatics software, methods and databases, which enable us to store, analyze and interpret the large volume of data generated from our platform technology, are protected as trade secrets.
Many pharmaceutical companies, biotechnology companies and academic institutions are competing with us in the field of immunotherapy and oncology, and they have obtained, or may obtain in the future, patents potentially relevant to our business. In order to identify and mitigate the risk of third party intellectual property conflicts, we conduct freedom-to-operate studies, and where appropriate, we obtain opinion of counsel, as an ongoing part of our business operations. We are aware of certain third party patents that contain broad claims potentially relevant to certain therapeutic uses of our PD-1 antibodies. We also are aware of certain third party patents that contain claims potentially relevant to other antibodies (against other targets) in our pipeline, and certain uses of those antibodies. Based on our analyses, if any claims in these patents were asserted against us, we do not believe our activities would be found to infringe any valid claim.
From time to time, we may find it necessary or prudent to obtain licenses from third party patent owners. Where licenses are available at reasonable cost, such licenses are considered a normal cost of doing business. In other instances, we may use the results of our freedom-to-operate studies to guide our early stage research away from areas where we are likely to encounter obstacles in the form of third party intellectual property. We strive to identify potential third party intellectual property issues in the early stages of research in our programs in order to minimize the cost and disruption of resolving such issues. In some cases, otherwise potentially relevant third party patents will expire before marketing approval of our drug candidates by the U.S. Food and Drug Administration is likely to have been granted. In such instances, under current U.S. law, our pre-clinical and clinical activities are exempt from claims of patent infringement. This exemption is sometimes called the “safe harbor” provision of U.S. law.
| 9 |
The Merger and Related Transactions
Overview
We were incorporated in Nevada as Cerulean Group, Inc. on February 27, 2012, and converted to a Delaware corporation on July 10, 2014. Prior to the Merger (as defined below), our Board of Directors determined to discontinue operations in our original business area to seek a new strategic opportunity. As a result of the Merger, we acquired the business of Enumeral and changed our name to Enumeral Biomedical Holdings, Inc.
Our authorized capital stock currently consists of 300,000,000 shares of common stock, par value $0.001 (the “Common Stock”), and 10,000,000 shares of “blank check” preferred stock, par value $0.001. Our Common Stock is quoted on the OTC Markets (OTCQB) under the symbol “ENUM.”
Enumeral was incorporated on December 11, 2009 under the laws of the State of Delaware.
On July 25, 2014, we completed a 4.62-for-1 forward split of our common stock in the form of a dividend, with the result that the 6,190,000 shares of common stock outstanding immediately prior to the stock split became 28,597,804 shares of Common Stock outstanding immediately thereafter. All share and per share numbers in this Annual Report on Form 10-K relating to our common stock have been adjusted to give effect to this stock split, unless otherwise stated.
On July 31, 2014 (the “Merger Closing Date”), our wholly owned subsidiary, Enumeral Acquisition Corp. (“Acquisition Sub”) merged with and into Enumeral (the “Merger”). Enumeral was the surviving corporation in the Merger and became our wholly owned subsidiary. All of the outstanding Enumeral stock was converted into shares of our Common Stock, as described in more detail below.
Upon the closing of the Merger and under the terms of a split-off agreement and a general release agreement (the “Split-Off Agreement”), we transferred all of our pre-Merger operating assets and liabilities to our wholly-owned special-purpose subsidiary, Cerulean Operating Corp. (“Split-Off Subsidiary”). Thereafter, pursuant to the Split-Off Agreement, we transferred all of the outstanding shares of capital stock of Split-Off Subsidiary to our pre-Merger majority stockholder, and our former sole officer and director (the “Split-Off”), in consideration of and in exchange for (i) the surrender and cancellation of an aggregate of 23,100,000 shares of our Common Stock held by such stockholder (which were cancelled and resumed the status of authorized but unissued shares of our Common Stock) and (ii) certain representations, covenants and indemnities.
As a result of the Merger and transactions effected pursuant to the Split-Off Agreement, we discontinued our pre-Merger business and acquired the business of Enumeral, and will continue the existing business operations of Enumeral as a publicly-traded company under the name Enumeral Biomedical Holdings, Inc. On the Merger Closing Date, we changed our fiscal year from a fiscal year ending on October 31 of each year to one ending on December 31 of each year, which is the fiscal year end of Enumeral.
Also on the Merger Closing Date, we closed a private placement offering (the “PPO”) of 21,549,510 Units of our securities, at a purchase price of $1.00 per Unit, each Unit consisting of one share of our Common Stock and a warrant to purchase one share of Common Stock at an exercise price of $2.00 per share with a term of five years (the “PPO Warrants”). Additional information concerning the PPO and PPO Warrants is presented below under “—The Merger and Related Transactions—the PPO” in this Section.
Merger Agreement
On the Merger Closing Date, we, Acquisition Sub and Enumeral entered into an Agreement and Plan of Merger and Reorganization (the “Merger Agreement”), which closed on the same date. Pursuant to the terms of the Merger Agreement, Acquisition Sub merged with and into Enumeral, which was the surviving corporation and thus became our wholly-owned subsidiary.
Pursuant to the Merger, we acquired the business of Enumeral to discover and develop novel therapeutics known as immunomodulators or immunotherapies that help the human immune system attack diseased cells. (See “Description of Business” above.)
| 10 |
At the closing of the Merger, (a) each share of Enumeral’s common stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.102121 shares of our Common Stock, (b) each share of Enumeral’s Series A Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.598075 shares of our Common Stock, (c) each share of Enumeral’s Series A-1 Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.790947 shares of our Common Stock, (d) each share of Enumeral’s Series A-2 Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.997594 shares of our Common Stock, (e) each share of Enumeral’s Series B Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 2.927509 shares of our Common Stock, and (f) a convertible note was converted into 3,230,869 shares of our Common Stock. As a result, an aggregate of 22,700,645 shares of our Common Stock were issued to the holders of Enumeral’s stock.
In addition, pursuant to the Merger Agreement: (a) warrants to purchase 694,443 shares of Enumeral’s common stock issued and outstanding immediately prior to the closing of the Merger were converted into warrants to purchase shares of our Common Stock at a conversion ratio of 1.102121 for one; (b) warrants to purchase 41,659 shares of Enumeral’s Series A Preferred Stock issued and outstanding immediately prior to the closing of the Merger were converted into warrants to purchase shares of our Common Stock at a conversion ratio of 1.598075 for one; (c) warrants to purchase 144,140 shares of Enumeral’s Series B Preferred Stock issued and outstanding immediately prior to the closing of the Merger were converted into warrants to purchase shares of our Common Stock at a conversion ratio of 2.927509 for one; and (d) options to purchase 948,567 shares of Enumeral’s common stock issued and outstanding immediately prior to the closing of the Merger were converted into options to purchase shares of our Common Stock at a conversion ratio of 1.102121 for one. As a result, warrants to purchase an aggregate of 1,253,899 shares of our Common Stock and options to purchase an aggregate of 1,045,419 shares of our Common Stock were issued in connection with the Merger.
The Merger Agreement provided certain anti-dilution protection to our Common Stock holders immediately prior to the Merger (after giving effect to the Split-Off), in the event that the aggregate number of Units sold in the PPO after the final closing thereof were to exceed 15,000,000. Accordingly, based on the final amount of gross proceeds raised in the PPO, we issued 1,690,658 additional shares of Common Stock to our Common Stock holders immediately prior to the Merger.
The Merger Agreement contained customary representations and warranties and pre- and post-closing covenants of each party and customary closing conditions. Breaches of the representations and warranties were subject to indemnification provisions. Each of the stockholders of Enumeral as of the date of the Merger initially received in the Merger 98% of the shares to which each such stockholder is entitled, with the remaining 2% of such shares held in escrow for 18 months to satisfy post-closing claims for indemnification by us (“Indemnity Shares”). The indemnification period expired on January 31, 2016 without any claims being made and all of the Indemnity Shares were distributed to the pre-Merger stockholders of Enumeral on a pro rata basis. The Merger Agreement also contained a provision providing for a post-Merger share adjustment as a means for which claims for indemnity could have been made by the pre-Merger stockholders of Enumeral. Pursuant to this provision, up to 500,000 additional shares (“R&W Shares”) of Common Stock could have been issued to the pre-Merger stockholders of Enumeral, pro rata, during the 18-month period following the Merger for breaches of representations and warranties by us. The indemnification period expired on January 31, 2016 with no claims being made.
The Merger was treated as a recapitalization for financial accounting purposes. Enumeral was considered the acquirer for accounting purposes, and our historical financial statements prior to the Merger have been replaced with the historical financial statements of Enumeral prior to the Merger in all filings with the SEC subsequent to the Merger. The Merger is intended to be treated as a tax-free reorganization under Section 368 of the Internal Revenue Code of 1986, as amended.
The Merger Agreement is filed as Exhibit 2.1 to this Annual Report on Form 10-K. All descriptions of the Merger Agreement herein are qualified in their entirety by reference to the text thereof filed as an exhibit hereto, which is incorporated herein by reference.
| 11 |
The PPO
Concurrently with the closing of the Merger and in contemplation of the Merger, we closed our PPO in which we sold 21,549,510 Units of our securities, at a purchase price of $1.00 per Unit, each Unit consisting of one share of our Common Stock and a PPO Warrant to purchase one share of Common Stock at an exercise price of $2.00 per share with a term of five years. The aggregate gross proceeds of the PPO were $21,549,510 (before deducting placement agent fees and expenses of approximately $3,294,000).
The investors in the PPO had anti-dilution protection on the shares of Common Stock included in Units purchased in the PPO in the event that within two years after the closing of the PPO we issued Common Stock or securities convertible into or exercisable for shares of Common Stock at a price lower than the Unit purchase price. The anti-dilution protection expired on July 31, 2016, without being triggered.
In addition, the PPO Warrants had anti-dilution protection in the event that prior to the warrant expiration date we issue Common Stock or securities convertible into or exercisable for shares of Common Stock at a price lower than the warrant exercise price, subject to exceptions for certain issuances. The PPO Warrants were amended in connection with the Warrant Tender Offer on December 12, 2016 to remove the anti-dilution provisions.
In connection with the PPO, we paid our placement agents, EDI Financial, Inc. and Katalyst Securities LLC (the “Placement Agents”), a commission equal to 10% of the gross proceeds raised from investors in the PPO. In addition, the Placement Agents collectively received warrants to purchase 10% of the number of shares of Common Stock included in the Units sold in the PPO, with a term of five (5) years and an exercise price of $1.00 per share (the “PPO Agent Warrants”); provided, however, that the Placement Agents were not entitled to any warrants on the sale of Units in excess of 20,000,000. We also agreed to pay to the Placement Agents a cash fee on the amount that any person or entity contacted by the Placement Agents, in connection with the Offering, invested in us at any time prior to the date that was eighteen (18) months after the closing of the PPO. No such fee was earned or paid.
As a result of the foregoing, the Placement Agents and their respective sub-agents were collectively paid an aggregate commission of $2,154,951 and were issued PPO Agent Warrants to purchase an aggregate of 2,000,000 shares of our Common Stock. We were also required to reimburse the Placement Agents up to $30,000 of legal expenses incurred in connection with the PPO, in the aggregate. The PPO Agent Warrants were amended in connection with the Warrant Tender Offer on December 12, 2016 to remove the anti-dilution protection provisions and to reduce the exercise price of such PPO Agent Warrants from $1.00 to $0.125 per share.
We agreed to indemnify the Placement Agents and their respective sub-agents to the fullest extent permitted by law, against certain liabilities that may be incurred in connection with the PPO, including certain civil liabilities under the Securities Act, and, where such indemnification is not available, to contribute to the payments the Placement Agents and their respective sub-agents may be required to make in respect of such liabilities.
All descriptions of the PPO Warrants and the PPO Agent Warrants herein are qualified in their entirety by reference to the text of the forms of such documents filed as Exhibits 10.40 and 10.41 to this Annual Report on Form 10-K.
Composition of the Board; Voting Agreement
In connection with the Merger, certain of our stockholders (holding a majority of our common stock), including all of the investors in the PPO, all of our pre-Merger stockholders and certain of the Enumeral stockholders (including all officers and directors and certain principal stockholders), entered into a Voting Agreement in which they agreed to vote their Enumeral Biomedical stock to maintain a specific composition of our Board of Directors. The Voting Agreement expired on July 31, 2016 and is therefore no longer in effect.
Accounting Treatment; Change of Control
The Merger was accounted for as a “reverse merger,” and Enumeral is deemed to be the acquirer in the reverse merger. Consequently, the assets and liabilities and the historical operations that are reflected in the consolidated financial statements prior to the Merger are those of Enumeral and are recorded at the historical cost basis of Enumeral, and the consolidated financial statements after completion of the Merger will include the assets and liabilities of Enumeral, historical operations of Enumeral and operations our company and our subsidiaries from the closing date of the Merger. As a result of the issuance of the shares of our Common Stock pursuant to the Merger, a change in control of Enumeral Biomedical occurred as of the date of consummation of the Merger. Except as described herein, no arrangements or understandings exist among present or former controlling stockholders with respect to the election of members of our Board of Directors and, to our knowledge, no other arrangements exist that might result in a change of control of Enumeral Biomedical.
| 12 |
We continue to be a “smaller reporting company,” as defined under the Exchange Act, and an “emerging growth company” under the Jumpstart Our Business Startups (JOBS) Act of 2012. We believe that as a result of the Merger we have ceased to be a “shell company” (as such term is defined in Rule 12b-2 under the Exchange Act).
Convertible Note Offering
On July 29, 2016, we entered into a Subscription Agreement (the “Subscription Agreement”) with certain accredited investors (the “Buyers”), pursuant to which the Buyers purchased our 12% Senior Secured Promissory Notes (the “Notes”) in the aggregate principal amount of $3,038,256 (before deducting placement agent fees and expenses of approximately $385,337 and legal fees of $122,512 associated with the offering), which includes $38,256 pursuant to an over-allotment option (the “Note Offering”).
Our obligations under the Notes were secured, pursuant to the terms of an Intellectual Property Security Agreement (the “Security Agreement”), dated as of July 29, 2016, among the Grantors (as defined below), the Buyers and the collateral agent for the Buyers named therein, by a first priority security interest in all now owned or hereafter acquired intellectual property of us and Enumeral (collectively, the “Grantors”), except to the extent such intellectual property could not be assigned or the creation of a security interest would be prohibited by applicable law or contract. In connection with the conversion of the Notes (as discussed below), the security interest was terminated.
The Notes had an aggregate principal balance of $3,038,256, and a stated maturity date of 12 months from the date of issuance. The principal on the Notes bore interest at a rate of 12% per annum, payable monthly commencing on September 1, 2016. Interest was payable in shares (the “Repayment Shares”) of Common Stock, provided, however, that interest was not to be calculated or accrued in a manner that would have triggered the anti-dilution adjustment on the PPO Warrants. In the event that on an interest payment date, the PPO Warrants’ anti-dilution provision would be triggered by the payment of interest in Common Stock, interest payments on the Notes were to be paid in cash. The Notes ranked senior to all our existing indebtedness, except as otherwise set forth in the Notes.
The Notes provided that the maturity date of all of the outstanding principal amount of the Notes, together with accrued and unpaid interest due thereon, would accelerate to the date, on or after September 1, 2016, on which we completed and closed (i) an offering involving the sale of at least $5,000,0000 of equity securities or securities convertible into or exercisable for equity securities by us, or in the case of a solicitation of the exercise of outstanding July 2014 Warrants (a “Warrant Solicitation”), an offering in which we received at least $2,000,000 (both such offerings being an “Equity or Equity Equivalent Offering”), or (ii) an alternative financing transaction not constituting an Equity or Equity Equivalent Offering, including transactions involving a business combination, debt financing, asset sale or other financing transaction, which, directly or indirectly, increased our cash and cash equivalent assets by not less than $5,000,000 (an “Alternative Financing” and together with the Equity or Equity Equivalent Offering, a “Qualified Offering”).
Upon the closing of a transaction that would be deemed to be a Qualified Offering but for the fact that such transaction closed prior to September 1, 2016 (a “Non-Qualified Offering”), the maturity date of all of the outstanding principal amount of the Notes, together with accrued and unpaid interest due thereon, would have accelerated to September 1, 2016.
At the closing of a Qualified Offering the outstanding principal and interest then due on the Notes was payable, at our discretion, in cash or shares of Common Stock; provided, however, that cash could only be paid if, after taking into account such payments, we would have had a minimum of $10,000,000 in cash, cash equivalents and marketable securities. Payments made in Common Stock would have been based on a valuation per share equal to 50% of the price per share of the Common Stock sold in the Equity or Equity Equivalent Offering or the price at which such securities could have been converted into or exercised for Common Stock. In the event of a sale of our company during the term of the Notes (including a merger (whether or not we were the surviving entity), an acquisition, or tender offer for a majority of the shares of the outstanding Common Stock), each holder of a Note would have been entitled to receive an amount equal to one and a half times the principal amount of, and any accrued and unpaid interest on, such holder’s Note at the closing of such sale; provided, however, that such sale preference amount would have been paid either in cash or in equivalent amount of securities of the acquiring entity at the acquiring entity’s discretion. Pursuant to the terms of the Notes, a transaction that triggered this 1.5x sale preference would not also have triggered the acceleration and conversion features described above.
| 13 |
The Notes provided that if we failed to pay any principal amount or interest due under the Notes within 5 business days of the date such payment is due, or upon the occurrence of other event of default under the terms of the Notes (which such event of default continues beyond any applicable cure period), the entire unpaid principal balance of the Note, together with any accrued and unpaid interest thereon, would have become due and payable, without presentment, demand, protest or notice of any kind.
Pursuant to the terms of a placement agency agreement (the “Placement Agency Agreement”), dated as of June 21, 2016, between us and the placement agent for the Note Offering (the “Placement Agent”), in connection with the closing of the Note Offering, the Placement Agent was paid a commission equal to ten percent (10%) of the gross proceeds of the Offering. The Placement Agency Agreement also provided that, in the event that we completed a Qualified Offering or a Non-Qualified Offering during the term of the Notes, the Placement Agent, or its designees, would have received ten-year warrants to purchase a number of shares of Common Stock equal to 10% of the Repayment Shares issued at maturity in connection with such Qualified Offering or Non-Qualified Offering, as applicable, with an exercise price per share equal to the price per share at which payments in shares are made to the Buyers (the “Placement Agent Warrants”). To the extent that we completed a Warrant Solicitation constituting an Equity or Equity Equivalent Offering which involves a restructuring of the warrant exercise price and amount of shares exercisable, the exercise price of the Placement Agent Warrants would reflect the average exercise price. However, in the event that the Notes are repaid in cash in full prior to the closing of a Qualified Offering or Non-Qualified Offering, and under circumstances where payment in cash is permitted under the Subscription Agreement, the Placement Agent shall not be entitled to any warrants.
The Placement Agency Agreement also provides that if, within 12 months of the first closing of the Note Offering, we complete a financing or similar transaction with a party introduced to us by the Placement Agent in connection with the Note Offering (a “Fee Tail Party”), the Placement Agent shall be entitled to receive a cash commission from us in an amount equal to 10% of the gross amount of the financing transaction with the Fee Tail Party. In addition, if such financing transaction involves the issuance to the Fee Tail Party of equity securities or securities exercisable, convertible or exchangeable for equity securities, the Placement Agent shall also be entitled to receive a ten-year warrant to purchase the equivalent of 10% of the equity securities issued or issuable to the Fee Tail Party at the price per share paid or to be paid by the Fee Tail Party. Any Placement Agent Warrants shall only be issued at such time and to the extent that such issuance would not trigger an anti-dilution adjustment with respect to the PPO Warrants.
Pursuant to the terms of the Placement Agency Agreement, we agreed to pay the Placement Agent’s legal counsel’s fees in the amount of $50,000 and the Placement Agent’s legal counsel’s reasonable out of pocket expenses related to the Note Offering (not to exceed $5,000 in the aggregate). In addition, we paid the Placement Agent a $20,000 non-accountable expense allowance.
The Placement Agency Agreement also provides that we will indemnify the Placement Agent and its agents to the fullest extent permitted by law, against certain liabilities that may be incurred in connection with the Note Offering, including certain civil liabilities under the Securities Act of 1933, as amended (the “Securities Act”), and, where such indemnification is not available, to contribute to the payments the Placement Agents and their respective sub-agents may be required to make in respect of such liabilities.
We also granted registration rights to each Buyer with respect to the Repayment Shares, and to the Placement Agent with respect to the shares of Common Stock issuable upon exercise of the Placement Agent Warrants, in the event a transaction occurs pursuant to which Placement Agent Warrants are issued (the “Placement Agent Warrant Shares,” and, together with the Repayment Shares, the “Registrable Shares”). These registration rights shall, in each case, be on a pari passu basis with, and upon substantially the same terms as, the registration rights granted to the investors in a Qualified Offering or Non-Qualified Offering. In the event of a Warrant Solicitation involving a Warrant Restructuring (as described in the Placement Agency Agreement), we agreed, without any action by a holder of the Notes, to register the resale of the Registrable Shares, together with the shares underlying the July 2014 Warrants, as restructured. Such registration rights will be no less favorable than those contained in the registration rights agreement executed in connection with the issuance of the PPO Warrants.
| 14 |
The Notes automatically converted (the “Note Conversion”) into 48,806,545 shares of Common Stock on December 12, 2016 in connection with the consummation of the Warrant Tender Offer (described below). In connection with the Note Conversion and pursuant to the terms of the Placement Agent Agreement, we issued Placement Agent Warrants to purchase 4,880,655 shares of Common Stock at an exercise price of $0.125 per share to designees of the Placement Agent.
On March 21, 2017, we entered into an amendment with the holders of the Placement Agent Warrants to reduce the exercise price of such warrants from $0.125 per share to $0.0625 per share in consideration of their past efforts as well as future support and cooperation on our behalf.
We issued the Notes in a private placement in reliance on the exemption from registration provided by Rule 506 of Regulation D under the Securities Act of 1933, as amended (the “Securities Act”). In connection with such transaction, the purchasers of the Notes represented that they were “accredited investors.”
The foregoing descriptions of the Subscription Agreement, Security Agreement, Placement Agency Agreement, and Note do not purport to be complete and are subject to, and qualified in their entirety by, the full text of the documents, copies of which are filed as Exhibit 10.36, 10.37 and 10.35, respectively, to this Annual report on Form 10-K.
Warrant Tender Offer
On December 12, 2016, we consummated our offer to amend and exercise (the “Warrant Tender Offer”) the outstanding PPO Warrants to purchase an aggregate of 21,549,510 shares of Common Stock.
The PPO Warrants of holders who elected to participate in the Warrant Tender Offer were amended to (i) receive four shares of common stock for each warrant exercised rather than one, (ii) reduce the exercise price to $0.50 per warrant in cash (or $0.125 per share); (iii) shorten the exercise period so that it expired concurrently with the expiration of the Warrant Tender Offer at 5:00 p.m. (Eastern Time) on December 9, 2016, and (iv) delete any price-based anti-dilution provisions.
The Warrant Tender Offer expired at 5:00 p.m. Eastern time on December 9, 2016. Pursuant to the Warrant Tender Offer, an aggregate of 6,863,000 PPO Warrants were tendered by their holders and were amended and exercised in connection therewith for gross proceeds to us of $3,431,500. As a result, we issued 27,452,000 shares of our common stock to the holders who amended and exercised their PPO Warrants in the Warrant Tender Offer. Such tendered PPO Warrants represented approximately 31.8% of our outstanding PPO Warrants as of December 9, 2016.
The warrant agent for the Warrant Tender Offer (the “Warrant Agent”) was paid an aggregate commission of approximately $255,000.
We are using the net proceeds from the Warrant Tender Offer to fund our ongoing operations and for general working capital purposes. As of the date of this filing, and after giving effect to the net proceeds from the Warrant Tender Offer, we believe that we only have sufficient liquidity to fund operations into May 2017. We continue to explore a range of potential transactions, which may include public or private equity offerings, debt financings, collaborations and licensing arrangements, and/or other strategic alternatives, including a merger, sale of assets or other similar transactions.
We issued the PPO Warrants in a private placement in reliance on the exemption from registration provided by Rule 506 of Regulation D under the Securities Act of 1933, as amended (the “Securities Act”). In connection with such transaction, the holders of the PPO Warrants represented that they were “accredited investors.” Similarly, the issuance of the shares of our Common Stock upon the amendment and exercise of the 6,863,000 warrants was exempt from registration under the Securities Act pursuant to Rule 506 of Regulation D. In connection with the Warrant Tender Offer, all holders of tendered PPO Warrants represented that they were “accredited investors.” The issuance of the Placement Agent Warrants in connection with this transaction was exempt from registration under Section 4(a)(2) of the Securities Act as a transaction by an issuer not involving any public offering.
| 15 |
Amendment of Certain Warrants to Purchase Common Stock
Effective as of December 12, 2016, we and holders of a majority of the PPO Warrants approved an amendment to remove the price-based anti-dilution provisions in the PPO Warrants. As a result, the priced-based anti-dilution provisions contained in the PPO Warrants have been removed and are of no further force or effect as of December 12, 2016. Accordingly, those PPO Warrants that were not tendered in the Warrant Tender Offer remain outstanding with an exercise price of $2.00 per share, and have been amended such that they no longer include price-based anti-dilution provisions.
Also in connection with the consummation of the Warrant Tender Offer, on December 12, 2016 we and holders of a majority of the outstanding PPO Agent Warrants approved an amendment to remove the price-based anti-dilution provisions in the PPO Agent Warrants and to reduce the exercise price of the PPO Agent Warrants from $1.00 per share to $0.125 per share. None of the PPO Agent Warrants were exercised in connection with the Warrant Tender Offer and all PPO Agent Warrants remain outstanding as of December 31, 2016.
Registration Rights
On December 12, 2016, in connection with the consummation of the Warrant Tender Offer and the Note Conversion, we entered into a Registration Rights Agreement (the “Registration Rights Agreement”), pursuant to which we agreed that we will promptly, but no later than 90 calendar days from the effective date of the Registration Rights Agreement, file a registration statement with the SEC (the “Registration Statement”) to register the resale of the shares of Common Stock issued upon (a) exercise of the PPO Warrants in the Warrant Tender Offer, and (b) the Note Conversion, as well as the Placement Agent Warrants under the Securities Act (collectively, the “Registrable Shares”). We also agreed to use our commercially reasonable efforts to ensure that such Registration Statement is declared effective within 180 calendar days of the effective date of the Registration Rights Agreement. If we were late in filing the Registration Statement (which condition we satisfied, and therefore is not applicable) or if the Registration Statement is not declared effective within 180 days of the effective date of the Registration Rights Agreement, we will be required to pay the holders of Registrable Shares that have not been so registered, liquidated damages at a rate equal to 1.00% of the aggregate purchase price paid by such holder (x) for the Notes and (y) the aggregate exercise price paid by the holder with respect to the PPO Warrants tendered in the Warrant Tender Offer, as applicable, for each full thirty (30) days (i) we were late in filing the Registration Statement, (ii) the Registration Statement is late in being declared effective by the SEC, (iii) after the Registration Statement is declared effective, the Registration Statement ceases for any reason to remain continuously effective or the holders of the Registrable Shares are otherwise not permitted to utilize the prospectus therein to resell the Registrable Securities for a period of more than 30 consecutive trading days, or (iv) the Registrable Shares, if issued, are not listed or included for quotation on a national exchange or an over-the-counter market, or trading of the Common Stock is suspended or halted on such exchange or market, which at the time constitutes the principal market for the Common Stock, for more than three (3) full, consecutive trading days; provided, however, a Registration Event (as defined in the Registration Rights Agreement) shall not be deemed to occur if all or substantially all trading in equity securities (including the Common Stock) of ours is suspended or halted on the Approved Market (as defined in the Registration Rights Agreement) for any length of time; provided, however, that in no event shall the aggregate of any such liquidated damages exceed 8% of (x) the aggregate purchase price paid by a holder for the July 2016 Notes and (y) the aggregate exercise price paid by the holder with respect to the PPO Warrants tendered in the Warrant Tender Offer. No liquidated damages will accrue and accumulate with respect to (a) any Registrable Shares removed from the Registration Statement in response to a comment from the staff of the SEC limiting the number of shares of Common Stock which may be included in the Registration Statement (a “Cutback Comment”), or (b) after the shares may be resold under Rule 144 under the Securities Act or another exemption from registration under the Securities Act.
We are required to use our commercially reasonable efforts to keep the Registration Statement “evergreen” for 12 months from the date it is declared effective by the SEC or until Rule 144 is available to the holders of Registrable Shares who are not and have not been our affiliates with respect to all of their registrable shares, whichever is earlier.
The holders of Registrable Shares (including any shares of Common Stock removed from the Registration Statement as a result of a cutback comment from the SEC) will have two “piggyback” registration rights for such shares with respect to any registration statement filed by us following the effectiveness of the Registration Statement that would permit the inclusion of such shares, subject to customary cut-backs on a pro rata basis if the underwriter or we determine that marketing factors require a limitation on the number of shares of stock or other securities to be underwritten.
| 16 |
We have agreed to pay all expenses in connection with any registration obligation provided in the Registration Rights Agreement, including, without limitation, all registration, filing, stock exchange fees, printing expenses, all fees and expenses of complying with applicable securities laws, and the fees and disbursements of its counsel and of its independent accountants. Each investor will be responsible for his, hers, or its own sales commissions, if any, transfer taxes and the expenses of any attorney or other advisor that such investor decides to employ.
Because we were at one time designated as a “shell company” as defined in Rule 12b-2 under the Exchange Act, even if we otherwise meet the holding period and other requirements of Rule 144, our securities may nevertheless only be sold in reliance on Rule 144 if at the time of a proposed sale pursuant to Rule 144, we have filed all reports and other materials required to be filed by Section 13 or 15(d) of the Exchange Act, as applicable, during the preceding twelve (12) months, other than Form 8-K reports. As a result, the restrictive legends on certificates for our Common Stock and Warrants cannot be removed (a) except in connection with an actual sale meeting the foregoing requirements or (b) pursuant to an effective registration statement.
All descriptions of the Registration Rights Agreement herein are qualified in their entirety by reference to the text of the form of such document filed as Exhibit 10.38 to this Annual Report on Form 10-K.
Competition
The biotechnology industry, especially early-stage companies, is characterized by rapidly advancing technologies, intense competition, and a strong emphasis on intellectual property. While we believe our technology, experience and scientific resources provide us with certain competitive advantages, we face actual and potential competition from various sources, including large, established pharmaceutical companies, emerging pharmaceutical and biotechnology companies (including companies specializing in immuno-oncology), academic institutions, governmental agencies, and public and private research institutions.
Many of the companies against which we are competing, or against which we are likely to compete in the future, have significantly greater financial resources and greater expertise in research and development, preclinical testing, clinical trials, manufacturing, regulatory affairs, and marketing than we do. We also compete with small and early-stage companies, particularly in our attempts to secure collaborative arrangements with large and established companies, and in our attempts to acquire technologies complementary to our programs. These companies also compete with us in recruiting and retaining qualified scientific and management personnel. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated in some of our competitors.
Although we are an early-stage company focused on the discovery and development of novel therapeutics, and have not yet begun any clinical trials, we anticipate that any pharmaceutical products that we develop and commercialize in the future will face competition from existing therapies and from new therapies that become available. The key competitive factors affecting the success of those products, if developed and approved, are likely to be their efficacy, safety, convenience, price, the level of generic competition and the availability of reimbursement from government and other third-party payors. Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize drugs that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any drugs that we may develop. Our competitors also may obtain FDA or other regulatory approval for their drugs more rapidly than we obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market.
Government Regulation
Regulatory authorities in the United States and in other countries regulate numerous aspects of the pharmaceutical industry, including pre-clinical testing, human clinical trials, manufacturing, marketing approval, labeling, promotion, advertising, distribution, post-approval monitoring, and export and import of pharmaceutical products.
Marketing Approval of Drugs in the United States
In the United States, the Food and Drug Administration, or FDA, regulates drugs under the Federal Food, Drug, and Cosmetic Act and its implementing regulations. The steps ordinarily required before a new pharmaceutical product may be marketed in the United States include:
| 17 |
| · | preclinical laboratory studies involving animals; |
| · | submission of an Investigational New Drug, or IND, application for human clinical testing; |
| · | rigorous human clinical trials to establish the safety and efficacy of the drug, on an indication-by-indication basis; |
| · | FDA inspections of the manufacturing facility at which the product is manufactured; |
| · | FDA inspections of clinical trial sites; |
| · | submission of a New Drug Application, or NDA; |
| · | payment of user fees; and |
| · | FDA review and approval of the NDA. |
Preclinical Studies:
Preclinical studies include laboratory testing and animal studies. Preclinical studies must comply with federal regulations, which specify certain requirements such as compliance with good laboratory practices. Results of the preclinical studies, manufacturing information, analytical data and a proposed clinical trial protocol are submitted to the FDA as part of an IND, which is an exemption that allows an unapproved drug to be shipped in interstate commerce and administered to humans. The IND must become effective before clinical trials may be commenced.
Clinical Trials:
Clinical trials must be conducted under the supervision of a qualified medical professional acting as the principal investigator, in compliance with detailed protocols that have been reviewed and approved by the FDA and by an independent Institutional Review Board (known as an IRB) at, or affiliated with, the medical facility where the new drug is being administered.
Human clinical trials are typically conducted in three sequential (and sometimes overlapping) phases:
| · |
In Phase I, the drug is administered to a small population of healthy human subjects or patients having the indication for which the drug is being tested. The primary purpose of Phase I is initial assessment of safety, although preliminary indications of effectiveness are sometimes sought. |
| · |
In Phase II, the drug is administered to a somewhat larger, but limited, patient population. The primary purposes of Phase II are to obtain additional safety data, to assess effectiveness in patients with the relevant indication, and to obtain data concerning dosage and dosage regimen. |
| · | In Phase III, the drug is administered to an expanded patient population, generally at geographically dispersed clinical trial sites. The primary purposes of Phase III are to obtain additional data on safety and efficacy in a population large enough to permit rigorous statistical analysis of results, and to provide information such as a list of side effects, for use in drug labeling. |
NDA:
The results of preclinical studies and clinical trials, together with detailed information on the drug’s chemistry, pharmacology, manufacture, formulation, safety and effectiveness, are submitted to the FDA in an NDA requesting approval to market the drug. The cost of an NDA is substantial, both in terms of studies required to generate and compile the requisite data, as well as the mandatory user fees submitted with the application.
As part of its review, the FDA may refer the application to an outside expert advisory committee for review, evaluation and a non-binding recommendation as to whether the drug should be approved. If the FDA is satisfied that all requirements have been met, it issues an approval letter, which authorizes commercial marketing of the drug for specific indications. As a condition of approval, the FDA may require post-marketing testing and surveillance to monitor the drug’s safety or efficacy, or impose other conditions. Once granted, marketing approvals may be withdrawn if compliance with regulatory standards is not maintained or if other problems occur.
| 18 |
Hazardous Materials
Our research and development processes involve the use of certain hazardous materials. We are subject to federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposing of hazardous materials and waste products, including certain regulations promulgated by the U.S. Environmental Protection Agency.
Employees
As of March 24, 2017 we employed three business executives and seven scientists and other employees. None of our employees is represented by a collective bargaining agreement, nor have we experienced work stoppages. We believe that our relations with our employees are good.
Available Information
We file reports and other information with the Securities and Exchange Commission as required by the Securities Exchange Act of 1934, as amended, which we refer to as the Exchange Act. You can find, copy and inspect information we file at the SEC’s Public Reference Room, which is located at 100 F Street, N.E., Room 1580, Washington, DC 20549, on official business days during the hours of 10:00 a.m. to 3:00 p.m. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the SEC’s Public Reference Room. You can review our electronically filed reports and other information that we file with the SEC on the SEC’s web site at www.sec.gov.
We were incorporated under the laws of the State of Nevada as Cerulean Group, Inc. on February 27, 2012, and converted to a Delaware corporation on July 10, 2014. We subsequently changed our name to Enumeral Biomedical Holdings, Inc. Our principal executive offices are located at 200 CambridgePark Drive, Suite 2000, Cambridge, Massachusetts, 02140, and our telephone number is (617) 945-9146. Our Internet website is www.enumeral.com. We make available free of charge through our website our Annual Report on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Exchange Act. We make these reports available through our website as soon as reasonably practicable after we electronically file such reports with, or furnish such reports to, the SEC. In addition, we regularly use our website to post information regarding our business, research and development programs and governance, and we encourage investors to use our website, particularly the information in the section entitled “Investor Relations,” as a source of information about us.
The foregoing references to our website are not intended to, nor shall they be deemed to, incorporate information on our website into this annual report on Form 10-K by reference.
An investment in our securities is speculative and involves various risks that may affect our operations or financial results. Many of those risks are driven by factors and events that we cannot control or predict. Before investing in our securities you should carefully consider the following risks, together with the financial and other information contained herein.
This report contains certain statements relating to future events or the future financial performance of our company. Prospective investors are cautioned that such statements are only predictions and involve risks and uncertainties, and actual events or results may differ materially. In evaluating such statements, prospective investors should specifically consider the various factors identified herein, including the matters set forth below, which could cause actual results to differ materially from those indicated by such forward-looking statements.
If any of the following or other risks materialize, our business, financial condition, and results of operations could be materially adversely affected which could adversely affect the value of our common stock. In such a case, investors in our common stock could lose all or part of their investment.
Prospective investors should consider carefully whether an investment in our company is suitable for them in light of the information contained herein and the financial resources available to them. The risks described below do not purport to be all the risks to which we could be exposed. This section is a summary of certain risks and is not set out in any particular order of priority. They are the risks that we presently believe are material to our operations. Additional risks of which we are not presently aware or which we presently deem immaterial may also impair our business, financial condition or results of operations.
| 19 |
Risks Related to our Business and the Industry in Which We Operate
We face technological uncertainties.
To date, we have not developed or commercialized any products utilizing our proprietary platform technology. There can be no assurance that our approach will enable us to successfully discover and develop novel therapeutics that help the immune system attack diseased cells. The discovery and development of such novel therapeutics for use in the diagnosis and treatment of cancer, infectious, autoimmune and inflammatory diseases also will be subject to the risks of failure inherent in the development of products based on new technologies. These risks include the possibilities that products based on these technologies will be ineffective or toxic, or otherwise fail to receive necessary regulatory approvals; that the products, if safe and effective, will be difficult or uneconomical to manufacture on a large scale; that third party patent rights will preclude us or our partners from marketing products; or that third parties will market equivalent or superior competing products. As a result, there can be no assurance that our research and development activities will lead to any commercially viable products in a relevant timeframe.
Biotechnology and pharmaceutical technologies have undergone and are expected to continue to undergo rapid and significant change. Our future success will depend in large part on our ability to maintain a competitive position with respect to these technologies. Rapid technological developments by our company or others may result in products or processes becoming obsolete before we recover any expenses that we incur in connection with the development of such products.
We may not achieve profitability.
We have historically incurred operating losses and expect to continue to have negative cash flows from operations. As of December 31, 2016, we have accumulated losses of $26,265,207. Our future profitability will depend on our ability to increase our revenues, which is subject to a number of factors, including our ability to successfully enter into collaborations with third parties, the success of our core platform technology and research and development efforts, our ability to compete effectively in a crowded field, availability of public funding through grants and other federal funds, the time required to reach commercial revenue and profitability, if at all, and global economic and political conditions.
Our future profitability also depends on our expense levels, which are influenced by a number of factors, including the resources we devote to developing and supporting our projects and potential products, the continued progress of our research and development of potential products, our ability to improve research and development efficiencies, license fees or royalties we may be required to pay, and the potential need to acquire licenses to new technology, the availability of intellectual property for licensing or acquisition, or to use our technology in new markets, which could require us to pay unanticipated license fees and royalties in connection with these licenses. If we fail to grow our revenue and manage our expenses, we may never achieve profitability, which would adversely and materially affect our ability to provide a return to our investors.
We have incurred significant operating losses since our inception, and anticipate that we will continue to incur significant losses for the foreseeable future. In addition, we have limited cash resources, and may not be able to raise additional capital on favorable terms, if at all. These factors raise substantial doubt about our ability to continue as a going concern.
Our continuation as a going concern is dependent upon our company attaining and maintaining profitable operations, generating continued cash payments from partners under new or existing contracts and/or raising additional capital, such as through public or private equity offerings, debt financings, or strategic collaborations and licensing arrangements. As of December 31, 2016, we had cash and cash equivalents of $3,162,400.
Since our inception in 2009, we have incurred significant net losses. As of December 31, 2016, we had an accumulated deficit of $26,265,207. Our recurring losses from operations and negative cash flows from operations raise substantial doubt about our ability to continue as a going concern, and as a result our independent registered public accounting firm included an explanatory paragraph in its report on our consolidated financial statements for the fiscal year ended December 31, 2016.
| 20 |
Our liquidity is highly dependent on our ability to obtain additional capital in the future. Our failure to raise new capital would impair our ability to both continue our current collaborations and develop new collaborative partnerships and could result in our failure to continue to operate as a going concern. Substantial doubt about our ability to continue as a going concern may also create negative reactions to the price of our common stock, and we may not be able to obtain additional financing in the future.
As of the date of this filing we believe that we only have sufficient liquidity to fund operations into May 2017. We are currently exploring a range of potential transactions, which may include public or private equity offerings, debt financings, collaborations and licensing arrangements, and/or other strategic alternatives, including a merger, sale of assets or other similar transactions. If we are unable to raise additional capital on terms acceptable to us and on a timely basis, we will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets. Our consolidated financial statements do not include any adjustments related to the recovery and classification of asset carrying amounts or the amount and classification of liabilities that might result should we be unable to continue as a going concern. Should the going concern assumption not be appropriate and we are not able to realize our assets and settle our liabilities, commitments and contingencies in the normal course of operations, adjustments would be required to our audited consolidated financial statements to the amounts and classifications of assets and liabilities, and these adjustments could be significant. Additionally, we expect to incur significant expenses and operating losses for the foreseeable future, and our net losses may fluctuate significantly from quarter to quarter and from year to year. These factors also raise substantial doubt about our ability to continue as a going concern.
We need additional funding to continue our operations and meet our research and development, capital and general and administrative expenses. Such funding may not be available on favorable terms, if at all, and may be dilutive to our existing stockholders. Without modifications to our existing payment obligations or receipt of additional funding, our existing cash and other sources of liquidity may not be sufficient to fund our operations beyond May 2017. If additional capital is not available, we may have to further curtail our operations, or take other actions that could adversely affect our stockholders.
Our business has not generated (nor do we anticipate that in the foreseeable future it will generate) the cash necessary to finance our operations. We expect to continue to incur losses and negative cash flows from operations for the foreseeable future, and we will require additional capital to continue our operations beyond May 2017. As of December 31, 2016, we had cash and cash equivalents totaling $3,162,400, excluding restricted cash.
Our near-term viability depends on our ability to raise additional capital through public or private equity offerings, debt financings, or strategic collaborations and licensing arrangements, and/or our success in promptly establishing a strategic alternative that is in our stockholders’ best interests.
If we are unable to raise additional capital through one or more of the means listed above prior to the end of May 2017, we will face substantial liquidity problems and will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets.
We will require additional capital to support operations and business growth, and such capital might not be available when needed or on terms satisfactory to us.
Continued investment will be needed to support our operations and business growth. Our future capital requirements will depend on a number of factors, including, but not limited to:
| · | the size and complexity of, and continued scientific progress in, our research and development programs; |
| · | our entry into new partnerships, and the terms of such partnering agreements; |
| · | competing technological and market developments; |
| 21 |
| · | the time and expense of building and maintaining our patent portfolio and enforcing patent claims; and |
| · | the cost of conducting clinical development and commercialization activities and potentially in-licensing products, if it proves necessary to do so. |
We intend to continue to make investments to support operations and business growth and will require additional funds to respond to business challenges, which may include the need to develop new products, conduct clinical trials (on our own or with our partners), enhance our operating infrastructure, and acquire complementary businesses and technologies. To continue to make such investments, we will likely need to obtain equity or debt financing or consummate a collaboration or licensing arrangement, or some other strategic transaction. However, such financing or potential transaction might not be available when needed or, if available, might not be available on terms satisfactory to us. From time to time, capital markets may experience periods of disruption and instability, which can contribute to worsening economic conditions that materially adversely affect broader financial and credit markets and reduce the availability of debt and equity capital for the market as a whole and for small capitalization businesses such as ours in particular.
In addition, to the extent additional capital is raised through the sale of equity or convertible debt securities, the issuance of these securities could result in dilution to our shareholders. These securities may also be sold at a discount from the market price of our common stock.
Additionally, if we fail to meet the criteria set forth in SEC regulations, various requirements would be imposed by law on broker-dealers who sell our securities to persons other than established customers and accredited investors. Consequently, such regulations may deter broker-dealers from recommending or selling our common stock, which may make it more difficult for us to raise capital. If we are unable to obtain adequate financing or financing on terms satisfactory to us, our ability to continue to support our operations and business growth and to respond to business challenges could be significantly impaired and there would be a material adverse effect on our business and financial condition.
If we are unable to raise capital on terms acceptable to us and on a timely basis, we will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets.
We rely on relationships with third parties for product development and commercialization, and those third parties could fail to perform as expected.
We believe that our success depends on developing and maintaining scientific and business relationships with other entities, including companies, academic institutions, and other organizations. Relying on such collaborative relationships entails risk to our future success because, among other things:
| · | our partners may not devote sufficient resources to the success of our collaboration; |
| · | our partners may not obtain regulatory approvals necessary to continue the collaborations in a timely manner, if at all; |
| · | our partners may be acquired by another company and decide to terminate our collaborative partnership or cease doing business or become insolvent; |
| · | our partners may develop or license technologies or components competitive with our products; |
| · | disagreements with partners could result in litigation or termination of the relationship; |
| · | collaborators may not have sufficient capital resources; |
| · | our existing collaborations may preclude us from entering into additional future arrangements; and |
| · | we may not be able to negotiate future partnerships, or renew existing collaborative agreements, on acceptable terms, if at all. |
Because these and other factors may be beyond our control, the development or commercialization of our products may be delayed or otherwise adversely affected.
If we or any of our partners terminate a collaborative arrangement, we may be required to devote additional resources to product development and commercialization, or we may need to cancel some development programs, which could adversely affect our product pipeline and business.
| 22 |
Successful development of products is uncertain.
Our development of future product candidates is subject to the risks inherent in the development of new biotechnology products, which include:
| · | delays in research and development, clinical testing or manufacturing; |
| · | unplanned expenditures in product development, clinical testing or manufacturing; |
| · | failure in clinical trials or failure to receive and maintain regulatory approvals; |
| · | emergence of equivalent or superior products; |
| · | inability to manufacture product candidates on a commercial scale; |
| · | inability to market products due to third-party patent rights; |
| · | decisions by our partners not to pursue product development; and |
| · | failure to achieve market acceptance after regulatory approval. |
Because of these and other risks, our research and development efforts or those of our partners may not result in any commercially viable products. If a significant portion of these development efforts is not successfully completed, required regulatory approvals are not obtained, or any approved products are not commercially successful, our business, financial condition and results of operations may be materially adversely affected.
The regulatory approval process is lengthy, expensive and uncertain.
Prior to marketing, any new therapeutic product must undergo an extensive regulatory approval process in the United States (and in other countries, as applicable) to establish that the product meets minimum requirements for safety and efficacy. This regulatory process, which includes preclinical studies and clinical trials (and may also include post-marketing surveillance), can take many years to complete and require the expenditure of substantial resources. The commencement or completion of clinical trials may be delayed or halted for various reasons, including difficulty in patient accrual, inadequate drug supply, adverse medical events, lack of efficacy, and issues with evaluator institutional review boards. Data obtained from preclinical studies and clinical trials are susceptible to varying interpretations that could delay, limit or prevent regulatory approval. In addition, previously unknown problems associated with a product that come to light after marketing approval might lead to a requirement for additional clinical studies or withdrawal of the product from the market.
We have not submitted an investigational new drug, or IND, application for any product candidate, and no product candidate has been approved for commercialization in the United States or elsewhere. No assurance can be given that we or any of our partners will be able to identify a product candidate to submit for approval, conduct clinical testing, or obtain the necessary approvals from the U.S. Food and Drug Administration or other regulatory authorities for any products.
Our use of our platform technology depends on a third party license that could be terminated.
We utilize our platform technology under an exclusive patent license agreement with the Massachusetts Institute of Technology. The term of the license agreement remains in effect until the expiration or abandonment of all issued patents and filed patent applications within the patent rights set forth in the agreement. The license agreement contains certain diligence obligations relating to research and development milestones, clinical milestones, and commercialization milestones. If we fail to achieve those milestones, we could lose certain rights under the license, or even lose the license entirely, which would have a material adverse effect on our business.
We operate in a highly competitive industry, and if our competitors develop superior products and technologies, our competitive position could be compromised.
We face various types of actual and potential competition. Competition could come from both established and emerging pharmaceutical and biotechnology companies (including companies specializing in immunotherapy), academic institutions, governmental agencies, and public and private research institutions. Many of the companies against which we are likely to compete have significantly greater financial resources and expertise in research and development, preclinical testing, clinical trials, manufacturing, regulatory affairs, and marketing than we do. We also compete with small and early-stage companies, particularly with respect to collaborative arrangements with large and established companies and with respect to acquiring technologies that may be complementary to our programs. In addition, we face competition in recruiting and retaining qualified scientific and management personnel. If we are unable to compete effectively against these companies, our business, financial condition and results of operations could be materially adversely affected. Additional information concerning competition is set forth in the section entitled “Description of Business – Competition.”
| 23 |
Claims that our platform technology or our products infringe third party patents might result in costly litigation
We cannot be certain that our platform technology, our products, or their respective use, do not infringe third party patents. Third parties might allege that we are infringing their patent rights and resort to litigation against us. Although we have conducted freedom-to-operate studies, it is possible that we have failed to identify relevant patents or applications. New patent applications in the United States and elsewhere are published approximately 18 months after their initial filing. Therefore, it is possible that relevant third party patent applications have been filed, but are not yet publicly available. During the examination process, which is sometimes called patent prosecution, amendment of existing claims or introduction of new claims is permissible in the U.S. Patent and Trademark Office and in most foreign patent offices. Therefore, potentially problematic claims may be introduced into pending third party applications that do not currently contain such claims.
We are aware of third party patents that contain broad claims potentially relevant to certain therapeutic uses of our anti-PD-1 antibodies. If we were to challenge the validity of any claim in an issued U.S. patent in court, we would need to overcome a statutory presumption of validity that attaches to every U.S. patent. This means that in order to prevail, we would have to present clear and convincing evidence as to the invalidity of the claim(s) in question. There is no assurance that a court would find in our favor on questions of infringement or validity.
In order to avoid or to settle actual or potential patent infringement claims, we may seek a license from a third party and pay license fees, or royalties, or both. Necessary licenses might not be available on commercially reasonable terms, if at all. Ultimately, we could be prevented from commercializing a product, or forced to cease using our technology platform, as a result of patent infringement claims. Defending against claims of patent infringement or misappropriation of trade secrets could be costly and time consuming, even if we prevail in the litigation, which could materially adversely affect our business, financial condition and results of operations.
We might be unsuccessful in obtaining adequate patent protection for one or more of our potential products.
The patent position of biotechnology and pharmaceutical companies is often uncertain because it involves complex legal and factual considerations. The standards applied by the U.S. Patent and Trademark Office and foreign patent offices in granting patents are not always applied uniformly or predictably, and such standards are subject to change based on court decisions, such as recent U.S. Supreme Court cases involving patents. Consequently, patents might not issue from some or all of our patent applications. In the event that we are unsuccessful in obtaining adequate patent protection for one or more of our products in the future, our business could be adversely affected.
Our competitors might successfully evade our patent protection.
Notwithstanding valid patents that we may own or control through an exclusive license, our competitors may independently develop and market alternative products similar to ours, without infringing any of our patent rights. In addition, they may design around our patented platform technology. This could diminish our competitive advantage, particularly if the competitor is a company with resources greater than ours.
One or more of our patents might be found invalid or unenforceable if challenged in court.
If we were to initiate legal proceedings against a third party to enforce a patent covering our platform technology or one of our products, the defendant might argue successfully that the claims we are asserting are invalid or unenforceable. Even though all of our issued patents will have undergone examination by the U.S. Patent and Trademark Office or a foreign patent office, we cannot be certain that there is no invalidating prior art. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we could lose part or all of the patent protection on one or more of our products, or on our platform technology, which could have a materially adverse effect on our business.
| 24 |
In the course of litigation, we might be subject to unfavorable publicity.
During the course of any patent litigation, there could be public announcements regarding hearings, rulings on motions, or other interim developments. If securities analysts or investors perceive such announcements as negative, our business could be adversely affected and the market price of our common stock could decline.
If we fail to retain key members of our staff, or fail to attract and train skilled new employees, our ability to conduct and expand our business would be impaired.
The loss of any of our key employees could seriously harm our product development and commercialization efforts. Furthermore, as our company continues to grow, we will require additional highly trained technical and business personnel. The market for such individuals is highly competitive in the biotechnology industry, particularly in the greater Cambridge, Massachusetts area where our main office and laboratory is located. If we fail to hire, train and retain a sufficient number of qualified employees to match our growth, our ability to conduct and expand our business could be impaired. We have entered into employment agreements with certain executive officers of our company. However, the existence of an employment agreement does not guarantee retention of members of our management team and we may not be able to retain those individuals for the duration of or beyond the end of their respective terms.
If we become subject to claims relating to improper handling, storage or disposal of hazardous materials, we could expend significant resources to bring our company into compliance.
Our research and development processes involve the controlled storage, use and disposal of hazardous materials. We are subject to federal, state and local regulations governing the use, manufacture, storage, handling and disposal of hazardous materials and waste products. We may incur significant costs complying with both existing and future environmental laws and regulations, and we are unable to predict whether any agency will adopt regulations in the future that would have a material adverse effect on our operations. The risk of accidental contamination or injury from hazardous materials cannot be eliminated completely. In the event of an accident, we could be held liable for damages that result, and the associated liability could exceed the limits or fall outside our existing insurance coverage.
If a catastrophe strikes our research facilities, we might be unable to complete our projects and our business operations would suffer, including but not limited to lost revenue.
Our research and development facilities are located in Cambridge, Massachusetts. Although we have commercial insurance, our facilities and some pieces of equipment and biological samples are difficult to replace and could require substantial replacement lead-time. Various types of disasters, including earthquakes, fires, floods, and acts of terrorism, may affect our research and development facilities. In the event our existing research and development facilities or equipment is affected by man-made or natural disasters, we may be unable to continue research and development activities, and may fail to meet our internal objectives or our partner demands. If our research and development operations were curtailed or ceased, it would seriously harm our business. In the event of a prolonged or even short-term power failure, we may lose biological samples stored in our freezers or growing in our incubators that could affect our ability to complete projects for ourselves or partners.
We might face product liability claims related to the use or misuse of products employing our antibody technology.
The administration of drugs to humans, in clinical trials or after commercialization, may expose us to product liability claims. Consumers, healthcare producers or persons selling products derived from our research and development programs may be able to bring claims against us based on the use of those products in clinical trials or the sale of those products. Product liability claims may be expensive to defend and may result in significant judgments against us, which could exceed our insurance coverage.
| 25 |
Pharmaceutical pricing, reimbursement and related matters are uncertain.
Our business, financial condition and results of operations may be materially and adversely affected by the continuing efforts of the government and third party payors to contain or reduce the costs of healthcare. Federal and state proposals to implement government controls, as well as ongoing emphasis on managed care in the U.S. healthcare industry, may continue to put pressure on the pricing of pharmaceutical products and diagnostic tests. Cost control initiatives could decrease the price that we or our partners receive for products commercialized in the future, and also may have a material adverse effect on our business, financial condition and results of operations. To the extent that cost control initiatives have a material adverse effect on our partners, our ability to commercialize products and to realize royalties may be adversely affected.
Investment Risks
You could lose all of your investment.
An investment in our securities is speculative and involves a high degree of risk. Potential investors should be aware that the value of an investment in our securities may go down as well as up. In addition, there can be no certainty that the market value of an investment in our securities will fully reflect its underlying value. You could lose your entire investment.
Failure to develop or maintain a trading market could negatively affect the value of our common stock and make it difficult or impossible for you to sell your shares.
Our common stock is currently quoted on the OTCQB. The OTCQB is a thinly traded market and lacks the liquidity of certain other public markets with which some investors may have more experience. We may not be able to satisfy the requirements of a national securities exchange for our common stock to be listed on such an exchange, which is often a more widely-traded and liquid market. Among the factors which may delay or prevent the listing of our common stock on a more widely-traded and liquid market are the following: our stockholders’ equity may be insufficient; the market value of our outstanding securities may be too low; our net income from operations may be too low; our common stock may not be sufficiently widely held; we may not be able to secure market makers for our common stock; and we may fail to meet the rules and requirements mandated by the various national exchanges and markets to have our common stock listed. Should we fail to satisfy the listing standards of the national exchanges or if our common stock is otherwise rejected for listing, or if our common stock remains listed on the OTCQB or is suspended from the OTCQB, the trading price of our common stock could suffer. In addition, the trading market for our common stock may continue to be less liquid and our common stock price may be subject to increased volatility, making it difficult or impossible for stockholders to sell shares of our common stock. Further, an unestablished trading market for our common stock may also impair our ability to raise capital by selling additional equity in the future, and may impair our ability to enter into strategic partnerships or acquire companies or products by using shares of our common stock as consideration.
Our stock may be traded infrequently and in low volumes, so you may be unable to sell your shares at or near the quoted bid prices if you need to sell your shares.
Unless we are able to meet the listing requirements of a national securities exchange, such as the New York Stock Exchange or the Nasdaq Stock Market, we expect our common stock to remain eligible for quotation on the OTCQB, or on another over-the-counter quotation system, or in the “pink sheets.” In those venues, however, the shares of our common stock may trade infrequently and in low volumes, meaning that the number of persons interested in purchasing our common shares at or near bid prices at any given time may be relatively small or non-existent. An investor may find it difficult to obtain accurate quotations as to the market value of our common stock or to sell his, her or its shares at or near bid prices, if at all. In addition, if we fail to meet the criteria set forth in SEC regulations, various requirements would be imposed by law on broker-dealers who sell our securities to persons other than established customers and accredited investors. Consequently, such regulations may deter broker-dealers from recommending or selling our common stock, which may further affect the liquidity of our common stock. This would also make it more difficult for us to raise capital.
| 26 |
You may experience dilution of your ownership interests because of the future issuance of additional shares of our common or preferred stock or other securities that are convertible into or exercisable for our common or preferred stock.
In the future, we may issue our authorized but previously unissued equity securities, resulting in the dilution of the ownership interests of our present stockholders. We are authorized to issue an aggregate of 300,000,000 shares of our common stock and 10,000,000 shares of “blank check” preferred stock. We will need to raise additional capital in the near future to meet our working capital needs, and there can be no assurance that we will not be required to issue additional shares, warrants or other convertible securities in the future in conjunction with these capital raising efforts, including at a price (or exercise price) below the price you paid for your stock. We may issue additional shares of our common stock or other securities that are convertible into or exercisable for our common stock in connection with hiring, promoting or retaining employees, future acquisitions, future sales of our securities for capital raising purposes, or for other business purposes. The future issuance of any such additional shares of our common stock may create downward pressure on our stock’s trading price.
Our common stock is subject to the “penny stock” rules of the SEC and the trading market in the securities is limited, which makes transactions in the stock cumbersome and may reduce the value of an investment in the stock.
The Securities Exchange Act of 1934, as amended (the “Exchange Act”), establishes the definition of a “penny stock,” for the purposes relevant to us, as any equity security that has a market price of less than $5.00 per share or with an exercise price of less than $5.00 per share, subject to certain exceptions. For any transaction involving a penny stock, unless exempt, the rules require that a broker or dealer take a number of additional steps to approve a person’s account for transactions in penny stocks, including obtaining financial information and investment experience objectives of the person, making a reasonable determination that the transactions in penny stocks are suitable for that person, and making a reasonable determination that the person has sufficient knowledge and experience in financial matters to be capable of evaluating the risks of transactions in penny stocks. In addition, the rules require that the broker or dealer receive from the investor a written agreement to the transaction, setting forth the identity and quantity of the penny stock to be purchased. Due to the additional burdens imposed by these rules, brokers may be less willing to execute transactions in securities subject to the penny stock rules, and this may make it more difficult for investors to dispose of our common stock and consequently cause a decline in the market value of our common stock.
We do not anticipate paying dividends on our common stock, and investors may lose the entire amount of their investment.
Cash dividends have never been declared or paid on our common stock, and we do not anticipate such a declaration or payment for the foreseeable future. We expect to use future earnings, if any, to fund business growth. Therefore, stockholders will not receive any funds absent a sale of their shares of common stock. If we do not pay dividends, our common stock may be less valuable because a return on your investment will only occur if our stock price appreciates. We cannot assure stockholders of a positive return on their investment when they sell their shares, nor can we assure stockholders that they will not lose the entire amount of their investment.
Being a public company is expensive and administratively burdensome.
As a public reporting company, we are subject to the information and reporting requirements of the Securities Act, the Exchange Act, and other federal securities laws, rules and regulations related thereto, including compliance with the Sarbanes-Oxley Act. Complying with these laws, regulations and standards requires and will continue to require the time and attention of our board of directors and management, and increases our expenses. Among other things, we are required to:
| · | prepare and distribute periodic reports in compliance with our obligations under federal securities laws; |
| · | institute a comprehensive compliance function, including with respect to corporate governance; |
| · | maintain and evaluate a system of internal controls over financial reporting in compliance with the requirements of Section 404 of the Sarbanes-Oxley Act and the related rules and regulations of the SEC and the Public Company Accounting Oversight Board; |
| · | maintain policies relating to our disclosure controls and procedures; and |
| · | involve, to a greater degree, our outside legal counsel and accountants in the above activities. |
| 27 |
The costs of preparing and filing annual and quarterly reports, proxy statements and other information with the SEC and furnishing audited reports to stockholders are significant. Continued compliance with the rules and regulations applicable to publicly-traded companies may require us to hire additional financial reporting, internal controls and other finance personnel, and involve significant regulatory, legal and accounting expenses, investor relations costs, as well as the attention of management. There can be no assurance that we will be able to continue to comply with the applicable regulations in a timely manner, if at all. In addition, being a public company makes it more expensive for us to obtain director and officer liability insurance, and, in the future, we may be required to accept reduced coverage or incur substantially higher costs to obtain coverage. These factors could also make it more difficult for us to attract and retain qualified executives and members of our board of directors, particularly directors willing to serve on our audit committee.
As an emerging growth company, we may follow certain permitted corporate governance practices, and may delay adoption of new or revised accounting standards, which may make our stock less attractive and result in less protection than is accorded to investors in a non-emerging growth company.
As an emerging growth company, we are permitted to follow certain corporate governance practices and disclosure requirements that are less robust than those otherwise required by the SEC. This may provide our stockholders with less information and less protection than what is accorded to investors under a national stock exchange’s listing requirements applicable to non-emerging growth company issuers.
In addition, as an emerging growth company, we have elected to take advantage of an extended transition period for any new or revised accounting standards that may be issued by the Financial Accounting Standards Board or the SEC. This means that when a standard is issued or revised and it has different application dates for public or private companies, we can delay adoption of the standard until it applies to private companies. This may make a comparison of our financial statements with any other public company that is not an emerging growth company, or is an emerging growth company that has opted out of using the extended transition period, difficult, as different or revised standards may be used. As a result, investors may find our common stock less attractive and there may be a less active trading market for our common stock. Consequently, our common stock price may be more volatile and could decline.
Any failure to maintain effective internal control over our financial reporting could materially adversely affect us.
Section 404 of the Sarbanes-Oxley Act of 2002 requires us to include in our annual reports on Form 10-K an assessment by management of the effectiveness of our internal control over financial reporting. In addition, at such time, if any, as we are no longer a “smaller reporting company” or “emerging growth company,” our independent registered public accounting firm will have to attest to and report on management’s assessment of the effectiveness of such internal control over financial reporting. Our compliance with Section 404 may require that we incur substantial accounting expense and expend significant management efforts. We currently do not have an internal audit group, and we may need to retain the services of additional accounting and financial staff or consultants with appropriate public company experience and technical accounting knowledge to satisfy the ongoing requirements of Section 404.
While we intend to diligently and thoroughly document, review, test and improve our internal control over financial reporting in order to ensure compliance with Section 404, in the future management may not be able to conclude that our internal control over financial reporting is effective. Furthermore, even if management were to reach such a conclusion, if our independent registered public accounting firm is not satisfied with the adequacy of our internal control over financial reporting, or if the independent auditors interpret the requirements, rules or regulations differently than we do, then they may decline to attest to management’s assessment or may issue a report that is qualified. Any of these events could result in a loss of investor confidence in the reliability of our consolidated financial statements, which in turn could negatively affect the price of our common stock.
Provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable.
Certain provisions in our certificate of incorporation and by-laws may have the effect of delaying or preventing a change of control or changes in our management. These provisions include the following:
| · | authorize “blank check” preferred stock that could be issued by our board of directors to defend against a takeover attempt; |
| 28 |
| · | establish a classified board of directors, as a result of which the successors to the directors whose terms have expired will be elected to serve from the time of election and qualification until the third annual meeting following their election; |
| · | require that directors only be removed from office for cause and only upon a supermajority stockholder vote; |
| · | provide that vacancies on the board of directors, including newly created directorships, may be filled only by a majority vote of directors then in office rather than by stockholders; |
| · | prevent stockholders from calling special meetings; and |
| · | prohibit stockholder action by written consent, requiring all actions to be taken at a meeting of the stockholders. |
In addition, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which generally prohibits a Delaware corporation from engaging in a broad range of business combinations with any “interested” stockholder for a period of three years following the date on which the stockholder becomes an “interested” stockholder.
Our ability to issue additional stock may prevent or make more difficult certain transactions, including a sale or merger of our company.
Our board of directors may authorize our issuance of up to 10,000,000 shares of preferred stock with powers, rights and preferences designated by it. Shares of voting or convertible preferred stock could be issued, or rights to purchase such shares could be issued, to create voting impediments or to frustrate persons seeking to effect a takeover or otherwise gain control of our company. The ability of our board of directors to authorize the issuance of such additional shares of preferred stock, with rights and preferences it deems advisable, could discourage an attempt by a party to acquire control of our company by tender offer or other means. Such issuances could therefore deprive stockholders of benefits that could result from such an attempt, such as the realization of a premium over the market price for their shares or the temporary increase in market price that such an attempt could cause. Moreover, the issuance of such additional shares of preferred stock to persons friendly to our board of directors could make it more difficult to remove incumbent officers and directors from office even if such change were to be favorable to stockholders generally.
The sale of a significant number of registered shares of our common stock may cause our stock price to decline.
In March 2017, we filed a Registration Statement on Form S-1 and prospectus that relates to the public offering, which is not being underwritten, by the selling stockholders listed therein, of up to 133,699,598 shares of our common stock. Of the shares being offered, 110,945,108 are presently issued and outstanding, and 22,754,490 are issuable upon exercise of common stock purchase warrants. The sale of these shares of our common stock will lead to an increase in the public float of our common stock, and such increase may cause the market price of our common stock to decline or fluctuate significantly.
***
The risks above do not necessarily comprise all of those associated with an investment in our company. This Annual Report on Form 10-K contains forward looking statements that involve unknown risks, uncertainties and other factors that may cause the actual results, financial condition, performance or achievements of our company to be materially different from any future results, performance or achievements expressed or implied by such forward looking statements. Factors that might cause such a difference include, but are not limited to, those set out above.
| 29 |
ITEM 1B. UNRESOLVED STAFF COMMENTS.
Not applicable to a smaller reporting company.
In March 2015, we relocated our offices and research laboratories to 200 CambridgePark Drive in Cambridge, Massachusetts. We are leasing 16,825 square feet at this facility pursuant to a lease that we entered into in November 2014. The term of this lease is for five years, and the initial base rent is $42.50 per square foot, or approximately $715,062 on an annual basis. The base rent will increase incrementally over the term of the lease, reaching approximately $804,739 on an annual basis in the fifth year of the term. In addition, we are obligated to pay a proportionate share of the operating expenses and applicable taxes associated with the premises, as calculated pursuant to the terms of the lease. We have delivered a security deposit to the landlord in the amount of $529,699, in the form of an irrevocable letter of credit, which may be reduced to $411,988 following the second anniversary of our commencement date under the lease, provided that we meet certain financial conditions set forth in the lease.
We previously occupied offices and research laboratories in approximately 4,782 square feet of space at One Kendall Square in Cambridge, Massachusetts, at an annual rent of $248,664, which we refer to as the Kendall Lease. For the year ended December 31, 2015, we recorded an expense of $55,352, representing all exit costs associated with our move to new offices and research laboratories in March 2015. In June 2015, we entered into a lease termination agreement with the landlord for our former facility at One Kendall Square, pursuant to which the Kendall Lease was terminated as of June 17, 2015. In accordance with the terms of the lease termination agreement, we are not obligated to pay rent for the One Kendall Square facility after May 31, 2015. We had maintained a security deposit relating to the facility, recorded as restricted cash on the accompanying consolidated balance sheet, which was returned to us pursuant to the lease termination agreement.
In addition, we maintained a small corporate office at 1370 Broadway in New York, New York, at an annual rent of $23,100. Our lease for our New York office expired on December 31, 2016.
From time to time, we may become involved in various lawsuits and legal proceedings which arise in the ordinary course of business. However, litigation is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may harm our business.
We are currently not aware of any pending legal proceedings to which we are a party or of which any of our property is the subject, nor are we aware of any such proceedings that are contemplated by any governmental authority.
ITEM 4. MINE SAFETY DISCLOSURES.
Not applicable to our company.
| 30 |
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES.
Our common stock is currently eligible for quotation and trades on the OTCQB under the symbol “ENUM.” Prior to the Merger, there was very limited trading in the Common Stock, and since the Merger our common stock continues to be thinly traded. The following table sets forth the high and low closing sale prices for our common stock for the fiscal quarter indicated as reported on OTCQB. The quotations reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not represent actual transactions.
| Period | High | Low | ||||||
| Fiscal 2017 | ||||||||
| Quarter ending March 31, 2017 (through March 24, 2017) | $ | 0.19 | $ | 0.13 | ||||
| Fiscal 2016 | ||||||||
| Quarter ended December 31, 2016 | $ | 0.20 | $ | 0.13 | ||||
| Quarter ended September 30, 2016 | $ | 0.22 | $ | 0.15 | ||||
| Quarter ended June 30, 2016 | $ | 0.24 | $ | 0.13 | ||||
| Quarter ended March 31, 2016 | $ | 0.29 | $ | 0.16 | ||||
| Fiscal 2015 | ||||||||
| Quarter ended December 31, 2015 | $ | 0.39 | $ | 0.22 | ||||
| Quarter ended September 30, 2015 | $ | 0.65 | $ | 0.36 | ||||
| Quarter ended June 30, 2015 | $ | 0.85 | $ | 0.57 | ||||
| Quarter ended March 31, 2015 | $ | 1.05 | $ | 0.70 | ||||
Holders
As of March 24, 2017, we had 128,409,788 shares of our common stock issued and outstanding held by approximately 407 stockholders of record.
Securities Authorized for Issuance Under Equity Compensation Plans
See “Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Shareholder Matters.”
Dividend Policy
We have never paid any cash dividends on our capital stock and do not anticipate paying any cash dividends on our common stock in the foreseeable future. We intend to retain future earnings to fund ongoing operations and future capital requirements. Any future determination to pay cash dividends will be at the discretion of our Board of Directors and will be dependent upon financial condition, results of operations, capital requirements and such other factors as the Board of Directors deems relevant.
Recent Sales of Unregistered Securities
Except as otherwise disclosed in reports filed with the SEC, we have not sold any unregistered equity securities during the period covered by this Annual Report on Form 10-K.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
None.
| 31 |
ITEM 6. SELECTED FINANCIAL DATA.
Not applicable to a smaller reporting company.
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS.
The following discussion highlights our results of operations and the principal factors that have affected our financial condition as well as our liquidity and capital resources for the periods described, and provides information that management believes is relevant for an assessment and understanding of the statements of financial condition and results of operations presented herein. The following discussion and analysis are based on our audited consolidated financial statements contained in this Annual Report on Form 10-K, which we have prepared in accordance with generally accepted accounting principles in the United States, or GAAP. You should read the discussion and analysis together with such consolidated financial statements and the related notes thereto.
Principles of Consolidation and Presentation
The consolidated financial statements include the accounts of Enumeral Biomedical and its subsidiaries. In these consolidated financial statements, “subsidiaries” are companies that are wholly owned by us, the accounts of which are consolidated with ours. Significant intercompany transactions and balances are eliminated in consolidation.
Overview
We were incorporated in Nevada as Cerulean Group, Inc. on February 27, 2012, and converted to a Delaware corporation on July 10, 2014. Prior to the Merger, which is described in further detail below, our Board of Directors determined to discontinue operations in our original business area to seek a new strategic opportunity. As a result of the Merger, we acquired the business of Enumeral and changed our name to Enumeral Biomedical Holdings, Inc.
On July 25, 2014, we completed a 4.62-for-1 forward split of our common stock in the form of a dividend, with the result that the 6,190,000 shares of our common stock outstanding immediately prior to the stock split became 28,597,804 shares of common stock outstanding immediately thereafter. All share and per share numbers in this report relating to our common stock have been adjusted to give effect to this stock split, unless otherwise stated.
On July 31, 2014, our wholly owned subsidiary, Enumeral Acquisition Corp. merged with and into Enumeral (the “Merger”). Enumeral was the surviving corporation in the Merger and became our wholly owned subsidiary. All of the outstanding Enumeral stock was converted into shares of our common stock, as described in more detail below.
Upon the closing of the Merger and under the terms of a split-off agreement and a general release agreement (the “Split-Off Agreement”), we transferred all of our pre-Merger operating assets and liabilities to a wholly-owned special-purpose subsidiary, Cerulean Operating Corp. (the “Split-Off Subsidiary”). Thereafter, pursuant to the Split-Off Agreement, we transferred all of the outstanding shares of capital stock of the Split-Off Subsidiary to our pre-Merger majority stockholder, and our former sole officer and director (the “Split-Off”), in consideration of and in exchange for (i) the surrender and cancellation of an aggregate of 23,100,000 shares of our common stock held by such stockholder (which were cancelled and will resume the status of authorized but unissued shares of our common stock) and (ii) certain representations, covenants and indemnities.
As a result of the Merger and transactions effected pursuant to the Split-Off Agreement, we discontinued our pre-Merger business and acquired the business of Enumeral, and are continuing the existing business operations of Enumeral as a publicly-traded company under the name Enumeral Biomedical Holdings, Inc.
Also on July 31, 2014, we closed a private placement offering (the “PPO”) of 21,549,510 Units of our securities, at a purchase price of $1.00 per Unit, each Unit consisting of one share of our common stock and a warrant to purchase one share of common stock at an exercise price of $2.00 per share with a term of five years (the “PPO Warrants”).
| 32 |
Also on July 31, 2014, we changed our fiscal year from a fiscal year ending on October 31 of each year to one ending on December 31 of each year, which is the fiscal year end of Enumeral.
Enumeral was incorporated on December 11, 2009 and has devoted substantially all of its resources to the discovery of monoclonal antibodies and other novel biologics for use in the diagnosis and treatment of cancer, infectious and inflammatory diseases. To date, all of our revenue has resulted from payments from strategic partners and the NCI, and we have not received any revenue from the sale of products or services. As of December 31, 2016, we had total stockholders’ equity of $3,907,914, including an accumulated deficit of $26,265,207.
We are a biopharmaceutical company focused on discovering and developing novel antibody immunotherapies that help the immune system fight cancer and other diseases. We utilize a proprietary platform technology that facilitates the rapid high resolution measurement of immune cell function within small tissue biopsy samples. Our initial focus is on the development of a pipeline of next generation monoclonal antibody drugs targeting established and novel immuno-modulatory receptors.
The concept of stimulating the immune system to fight cancer was first advanced more than a century ago, but it is only recently that the field of immuno-oncology has seen clinical success, with marketing approvals being granted for antibodies that block CTLA-4 (Yervoy® (ipilimumab)) and PD-1 (Keytruda® (pembrolizumab) and Opdivo® (nivolumab)), and PD-L1 (Tecentriq® (atezolizumab)). Use of these drugs has established that durable anti-tumor responses can be elicited in some patients by blocking the checkpoints that normally suppress the human immune response against cancer cells. The success of these drugs suggests that immuno-oncology may fundamentally alter the course of cancer treatment.
In our lead antibody program, we have characterized certain anti-PD-1 antibodies, or simply “PD-1 antibodies,” using patient biopsy samples, in an effort to identify next generation PD-1 antagonists with enhanced selectivity for the immune effector cells that carry out anti-tumor functions. We have identified two antagonist PD-1 antibodies that inhibit PD-1 activity in different ways. The distinction is that one of the antibodies (ENUM 388D4) blocks binding of the ligand PD-L1 to PD-1, while the other antibody (ENUM 244C8) does not inhibit PD-L1 binding. However, both display activity in various biological assays. In addition to our PD-1 antibody program, we are developing antibody drug candidates for a number of other immunomodulatory protein targets, including TIM-3, CD39, and TIGIT. We are also pursuing several antibody programs for which we have not yet publicly disclosed the targets.
Our proprietary platform technology, exclusively licensed from the Massachusetts Institute of Technology, or MIT, is a microwell array technology that detects secreted molecules (such as antibodies and cytokines) and cell surface markers, at the level of single, live cells – and enables recovery of single, live cells of interest. The platform technology can be used to achieve at least three separate, but complementary, objectives. First, we use the platform to rapidly produce antibody libraries with high diversity. Second, the platform has the potential to guide rational selection of lead candidates derived from these libraries, through characterization of immune function at the level of single cells from human biopsy samples. Third, it has the potential to identify patients more likely than others to benefit from treatment with a given therapeutic antibody. Thus, our platform is a multipurpose tool that is valuable for activities ranging from antibody discovery to target discovery to patient stratification in clinical development. The platform yields multidimensional, functional read-outs from single live cells, such as tumor infiltrating lymphocytes, or TILs, from human tumor biopsy samples, and it enables us to examine the responses of different classes of human immune cells to treatment with immuno-modulators in the context of human disease, as opposed to animal models of disease.
On July 29, 2016, we entered into a Subscription Agreement with certain accredited investors (the “Buyers”), pursuant to which the Buyers purchased our 12% Senior Secured Promissory Notes (the “Notes”) in the aggregate principal amount of $3,038,256 (before deducting placement agent fees and expenses of $385,337 and legal fees of $122,512), which includes $38,256 pursuant to an over-allotment option (the “Note Offering”). For additional information on the Notes and the Note Offering, please refer to “The Merger and Related Transactions – Convertible Note Offering” above.
On December 12, 2016, we consummated an offer to amend and exercise certain outstanding warrants to purchase an aggregate of 21,549,510 shares of our common stock originally issued to investors who participated in our July 31, 2014 private placement financing (the “Warrant Tender Offer”). Pursuant to the Warrant Tender Offer, an aggregate of 6,863,000 warrants were tendered by their holders, and we received gross proceeds in the amount of $3,431,500 for the issuance of 27,452,000 shares of our common stock. In addition, pursuant to the Warrant Tender Offer, the full principal balance and accrued interest of the Notes was converted into 48,806,545 shares of our common stock. For additional information on the Warrant Tender Offer, please refer to “The Merger and Related Transactions - Warrant Tender Offer” above.
| 33 |
As of December 31, 2016, we had cash and cash equivalents totaling $3,162,400, excluding restricted cash.
To date, our proof-of-concept corporate collaborations and our contracts with the NCI have provided minimal revenue. However, our business has not generated (nor do we anticipate that in the foreseeable future it will generate) the cash necessary to finance our operations. We expect to continue to incur losses and negative cash flows from operations for the foreseeable future, and we will require additional capital to continue our operations beyond May 2017.
Our near-term viability depends on our ability to raise additional capital through public or private equity offerings, debt financings, or strategic collaborations and licensing arrangements, and/or our success in promptly establishing a strategic alternative that is in our stockholders’ best interests.
If we are unable to raise additional capital through one or more of the means listed above prior to the end of May 2017, we will face substantial liquidity problems and will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets.
No assurance can be given that additional financing or strategic alliances and licensing arrangements will be available when needed or that, if available, such financing could be obtained on terms favorable to us or our stockholders. See Item 1A. - “Risk Factors.”
Results of Operations
Year ended December 31, 2016 as compared to year ended December 31, 2015
| Year Ended December 31, | Increase | |||||||||||
| 2016 | 2015 | (Decrease) | ||||||||||
| Revenue: | ||||||||||||
| Collaboration and license revenue | $ | 1,999,666 | $ | 1,100,000 | $ | 899,666 | ||||||
| Grant revenue | 453,202 | 389,385 | 63,817 | |||||||||
| Total revenue | 2,452,868 | 1,489,385 | 963,483 | |||||||||
| Cost of revenue and expenses: | ||||||||||||
| Research and development | 4,865,056 | 6,493,859 | (1,628,803 | ) | ||||||||
| General and administrative | 4,561,076 | 5,695,932 | (1,134,856 | ) | ||||||||
| Total cost of revenue and expenses | 9,426,132 | 12,189,791 | (2,763,659 | ) | ||||||||
| Loss from operations | (6,973,264 | ) | (10,700,406 | ) | 3,727,142 | |||||||
| Other income (expense): | ||||||||||||
| Interest income (expense) | (4,913,545 | ) | 9,699 | (4,923,244 | ) | |||||||
| Loss on extinguishment of derivative liabilities | (1,577,896 | ) | - | (1,577,896 | ) | |||||||
| Change in fair value of derivative liabilities | 1,608,474 | 13,980,711 | (12,372,237 | ) | ||||||||
| Total other income (expense), net | (4,882,967 | ) | 13,990,410 | (18,873,377 | ) | |||||||
| Net income (loss) | $ | (11,856,231 | ) | $ | 3,290,004 | $ | (15,146,235 | ) | ||||
Collaboration and license revenue. Collaboration and license revenue increased by $899,666, to $1,999,666 for the year ended December 31, 2016, as compared to $1,100,000 for the year ended December 31, 2015. This increase is primarily attributable to our license agreement with Pieris, which we entered into in the second quarter of 2016.
| 34 |
Grant revenue. Grant revenue increased by $63,817 to $453,202 for the year ended December 31, 2016, as compared to $389,385 for the year ended December 31, 2015. This increase is attributable additional activities under our Phase II Small Business Innovation Research agreement with the National Cancer Institute, or NCI.
Research and development expenses. Research and development expenses decreased by $1,628,803 to $4,865,056 for the year ended December 31, 2016, as compared to $6,493,859 for the year ended December 31, 2015. This decrease was primarily attributable to a decrease in payroll and personnel expenses of $985,521, as a result of lower headcount, a decrease in laboratory expenses of $492,818, and a decrease in patent costs of $324,051, offset by an increase in facilities costs of $179,592.
General and administrative expenses. General and administrative expenses decreased by $1,134,856 to $4,561,076 for the year ended December 31, 2016, as compared to $5,695,932 for the year ended December 31, 2015. This decrease was primarily attributable to a decrease in payroll and personnel expenses of $544,527, as a result of lower headcount, a decrease in professional service fees of $395,324, and a decrease in investor relations costs of $185,087.
Interest income (expense). Interest income (expense) changed by $4,923,244 to ($4,913,545) for the year ended December 31, 2016, as compared to $9,699 the year ended December 31, 2015. This change is primarily attributable to the following: (i) the expense associated with the contingent beneficial conversion feature of the Notes of $4,253,558 that was recognized on December 12, 2016 when the contingency was resolved, (ii) the accretion of debt issuance costs associated with the Notes of $507,849, and (iii) the interest expense associated with the Notes of $136,722.
Loss on extinguishment of derivative liabilities. Loss on extinguishment of derivative liabilities was $1,577,896 for the year ended December 31, 2016. We had no such loss for the year ended December 31, 2015. In connection with the Warrant Tender Offer, all derivative liabilities outstanding were extinguished. As a result the Company recorded the following: (i) the expense associated with the issuance of agent warrants of $704,848, (ii) the expense related to the loss on conversion of the PPO Warrants of $686,300, and (iii) the expense related to the modification of the PPO Agent Warrants exercise price from $1.00 a share to $0.125 a share of $187,240.
Change in fair value of derivative liabilities. Change in fair value of derivative liabilities decreased $12,372,237 to $1,608,474 for the year ended December 31, 2016, as compared to $13,980,711 for the year ended December 31, 2015. In connection with the PPO, we issued warrants to purchase an aggregate of 23,549,510 shares of our common stock. We also previously issued warrants to purchase an aggregate of 66,574 shares of our common stock related to a prior financing transaction with Square 1 Bank. As of December 31, 2016, these warrants have either been exercised or amended to remove the anti-dilution protection as part of the Warrant Tender Offer. The fair value of these warrants was recorded in the liability section of the consolidated balance sheet and as of December 31, 2015 was estimated at $2,138,091. The fair value of the derivative liabilities was determined at issuance and the end of each reporting period, with the resulting gains and losses recorded as the change in fair value of derivative liabilities on the consolidated statements of operations and comprehensive income (loss). In connection with the Warrant Tender Offer, the change in fair value was determined for the period from January 1, 2016 through December 12, 2016, the date which the anti-dilution protection provision was removed. During this period, we realized a gain of $1,608,474, due to the change in the fair value of the derivative liabilities. This gain is principally a result of the decrease in fair value of our common stock price between December 31, 2015 and December 12, 2016. As of December 12, 2016 these warrants were no longer classified as a derivative liabilities on our consolidated balance sheet as the awards were exercised or amended and have been reclassified into equity.
Net income (loss). Net income (loss) changed by $15,146,235 to ($11,856,231) for the year ended December 31, 2016, as compared to $3,290,004 for the year ended December 31, 2015. This change in net income (loss) was primarily due to a change in other income (expense) of $18,873,377 as a result of various non-cash income (expenses), offset by a decrease of $2,763,659 in cost of revenue and expenses, resulting from cost cutting measures taken during the year ended December 31, 2016, and an increase in revenue of $963,483, associated with our contract with Pieris.
As of December 31, 2016, we had accumulated losses of $26,265,207 since inception and, therefore, have not paid any federal income taxes. Realization of deferred tax assets is dependent on future earnings, if any, the timing and amount of which are uncertain. Accordingly, valuation allowances in amounts equal to the deferred tax assets have been established to reflect these uncertainties. Utilization of the deferred tax asset, consisting of net operating loss and research and development credit carryforwards, may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986, as amended, due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of net operating loss and research and development credit carryforwards that can be utilized annually to offset future taxable income and tax, respectively.
| 35 |
Liquidity and Capital Resources
We have financed our operations since inception primarily through equity financings of both preferred and common stock, venture debt, and revenues from corporate collaborations and our contracts with NCI. We have raised gross proceeds of approximately $39.3 million, consisting of: (i) a $3.2 million Series A Preferred Stock financing completed in early 2011; (ii) a $2.7 million Series A-1 Preferred Stock financing completed in mid-2012; (iii) a $2.7 million Series A-2 Preferred Stock financing completed in mid-2013; (iv) a $2.0 million Series B Preferred Stock financing in April 2014; (v) a $750,000 bridge note financing in the first half of 2014; (vi) $21.5 million in the PPO in July 2014; (vii) a $3.0 million 12% Senior Note financing in July 2016; and (viii) the $3.4 million Warrant Tender Offer in December 2016. The securities issued in the above financing rounds prior to the PPO were converted into Enumeral Biomedical common stock in the Merger.
The consolidated financial statements have been prepared in conformity with generally accepted accounting principles in the United States which contemplate our continuation as a going concern. As of December 31, 2016, we had an accumulated deficit of $26,265,207 and have experienced negative cash flows from operations since inception. As of the date of this filing, we believe we have sufficient liquidity to fund operations into May 2017. If we are unable to raise additional capital on terms acceptable to us and on a timely basis, we will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets. The consolidated financial statements do not include any adjustments related to the recovery and classification of asset carrying amounts or the amount and classification of liabilities that might result should we be unable to continue as a going concern.
Cash Flows
The following table summarizes our sources and uses of cash for the years ended December 31, 2016 and 2015:
| Year Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Net cash used in operating activities | $ | (5,671,079 | ) | $ | (9,285,052 | ) | ||
| Net cash provided by investing activities | - | 1,977,923 | ||||||
| Net cash provided by financing activities | 5,237,217 | 443,274 | ||||||
| Net decrease in cash and cash equivalents | (433,862 | ) | (6,863,855 | ) | ||||
The change in the net decrease in cash for the year ended December 31, 2016 as compared to the year ended December 31, 2015 was $6,429,993, representing the aggregate of (i) a decrease in net cash used in operating activities of $3,613,973, (ii) a decrease in net cash provided by investing activities of $1,977,923, and (iii) an increase in net cash provided by financing activities of $4,793,943.
Operating Activities
Net cash used in operating activities was $5,671,079 for the year ended December 31, 2016, which primarily resulted from a net loss of $11,856,231 adjusted for non-cash items including: (i) interest expense of $4,265,711, (ii) change in fair value of derivative liabilities of $1,608,474, (iii) loss on extinguishment of derivative liabilities of $1,577,896, (iv) stock-based compensation expense of $932,325, (v) depreciation and amortization of $609,396, and (vi) accretion of debt discount of $507,849.
Net cash used in operating activities was $9,285,052 for the year ended December 31, 2015, which primarily resulted from the change in fair value of derivative liabilities of $13,980,711, offset by net income of $3,290,004.
| 36 |
Investing Activities
There was no net cash provided by investing activities for the year ended December 31, 2016.
Net cash provided by investing activities was $1,977,923 for the year ended December 31, 2015, which primarily resulted from net proceeds from the sale of marketable securities of $3,000,799, offset by purchases of property and equipment of $1,050,506.
Financing Activities
Net cash provided by financing activities was $5,237,217 for the year ended December 31, 2016, which consisted of net proceeds from the issuance of the Notes of $2,530,407 and net proceeds from the Warrant Tender Offer of $2,947,283, offset by payments on equipment lease financing of $240,473.
Net cash provided by financing activities was $443,274 for the year ended December 31, 2015, which consisted of net proceeds from the equipment lease financing of $413,599 and proceeds from the excise of stock options of $29,675.
Our business does not generate (nor do we anticipate that in the foreseeable future it will generate) the cash necessary to finance our operations. We expect to continue to incur losses and negative cash flows from operations for the foreseeable future, and we will require additional capital to continue our operations beyond May 2017. As of December 31, 2016, we had cash and cash equivalents totaling $3,162,400, excluding restricted cash.
Our near-term viability depends on our ability to raise additional capital through public or private equity offerings, debt financings, or strategic collaborations and licensing arrangements, and/or our success in promptly establishing a strategic alternative that is in our stockholders’ best interests. To the extent additional capital is raised through the sale of equity or convertible debt securities, such securities may be sold at a discount from the market price of our common stock. The issuance of these securities could also result in significant dilution to some or all of our stockholders, depending on the terms of the transaction.
If we are unable to raise additional capital through one or more of the means listed above prior to the end of May 2017, we will face substantial liquidity problems and will be required to downsize or wind down our operations through liquidation, bankruptcy, or a sale of our assets.
No assurance can be given that additional financing or strategic alliance and licensing arrangements will be available when needed or that, if available, such financing could be obtained on terms favorable to us or our stockholders. See Item 1A. “Risk Factors.”
Critical Accounting Policies, Estimates, and Judgments
Cash and Cash Equivalents
We consider all highly liquid investments with maturities of 90 days or less from the purchase date to be cash equivalents. Cash and cash equivalents are held in depository and money market accounts and are reported at fair value.
Marketable Securities
Marketable securities consist of U.S. treasury securities with maturities of more than 90 days. We have determined the appropriate balance sheet classification of the securities as current since they are available for use in current operating activities, regardless of actual maturity dates, and are recorded on the balance sheet at fair value, with the unrealized gains and losses reported in accumulated other comprehensive income (loss), which is a separate component of stockholders’ equity.
| 37 |
Fair Value of Financial Instruments
Fair values of financial instruments included in current assets and current liabilities are estimated to approximate their book values, due to the short maturity of such instruments. All debt is based on current rates at which we could borrow funds with similar remaining maturities and approximates fair value. Our assets and liabilities that are measured at fair value on a recurring basis are measured in accordance with the Financial Accounting Standards Board’s (“FASB”) Accounting Standards Codification 820, Fair Value Measurements and Disclosures, which establishes a three-level valuation hierarchy for measuring fair value and expands financial statement disclosures about fair value measurements. The valuation hierarchy is based upon the transparency of inputs to the valuation of an asset or liability as of the measurement date.
Revenue Recognition
Collaboration and License Revenue
Non-refundable license fees are recognized as revenue when we have a contractual right to receive such payment, the contract price is fixed or determinable, the collection of the resulting receivable is reasonably assured and we have no further performance obligations under the license agreement. Multiple element arrangements, such as license and development arrangements are analyzed to determine whether the deliverables, which often include license and performance obligations such as research and steering committee services, can be separated or whether they must be accounted for as a single unit of accounting in accordance with GAAP.
We recognize up-front license payments as revenue upon delivery of the license only if the license has stand-alone value and the fair value of the undelivered performance obligations, typically including research and/or steering committee services, can be determined. If the fair value of the undelivered performance obligations can be determined, such obligations would then be accounted for separately as performed. If the license is considered to either (i) not have stand-alone or (ii) have stand-alone value but the fair value of any of the undelivered performance obligations cannot be determined, the arrangement would then be accounted for as a single unit of accounting and the license payments and payments for performance obligations are recognized as revenue over the estimated period of when the performance obligations are performed.
Whenever we determine that an arrangement should be accounted for as a single unit of accounting, we must determine the period over which the performance obligations will be performed and revenue will be recognized. Revenue will be recognized using either a relative performance or straight-line method. We recognize revenue using the relative performance method provided that we can reasonably estimate the level of effort required to complete our performance obligations under an arrangement and such performance obligations are provided on a best-efforts basis. Direct labor hours or full-time equivalents are typically used as a measure of performance. Revenue recognized under the relative performance method would be determined by multiplying the total payments under the contract, excluding royalties and payments contingent upon achievement of substantive milestones, by the ratio of level of effort incurred to date to estimated total level of effort required to complete our performance obligations under the arrangement. Revenue is limited to the lesser of the cumulative amount of payments received or the cumulative amount of revenue earned, as determined using the relative performance method, as of each reporting period.
If we cannot reasonably estimate the level of effort required to complete our performance obligations under an arrangement, but we can reasonably estimate when the performance obligation ceases or the remaining obligations become inconsequential and perfunctory, then the total payments under the arrangement (excluding royalties and payments contingent upon achievement of substantive milestones) would be recognized as revenue on a straight-line basis over the performance period.
Revenue is limited to the lesser of the cumulative amount of payments received or the cumulative amount of revenue earned, as determined using the straight-line basis, as of the period ending date.
| 38 |
Grant Revenue
We recognize nonrefundable grant revenue that is earned in connection with our Small Business Innovation Research (“SBIR”) contracts with the NCI. In September 2012, we entered into a Phase I SBIR contract with the NCI, and in September 2014, we were awarded a Phase II SBIR contract from the NCI. Grant revenue consists of a portion of the funds received to date from the NCI, which allows us to conduct research on colon cancer tissues. Revenue is recognized as the related research services are performed in accordance with the terms of the contract.
Research and Development Expenses
Research and development expenditures are charged to the consolidated statement of operations as incurred. Research and development expenses are comprised of costs incurred in performing research and development activities, including salaries and benefits, facilities costs, clinical supply costs, contract services, depreciation and amortization expense and other related costs. Costs associated with acquired technology, in the form of upfront fees or milestone payments, are charged to research and development expense as incurred. Legal fees incurred in connection with patent applications along with fees associated with the license to our core technology are expensed as research and development expense.
Accounting for Derivative Liabilities
Our derivative liabilities related to (a) warrants to purchase an aggregate of 23,549,510 shares of our common stock that were issued in connection with the July 2014 PPO and (b) warrants to purchase 41,659 shares of Enumeral Series A Preferred Stock that were issued in December 2011 and June 2012 pursuant to Enumeral’s debt financing arrangement with Square 1 Bank that were subsequently converted into warrants to purchase 66,574 shares of our common stock in connection with the July 2014 Merger.
Due to the price protection provision included in the original warrant agreements, the warrants were deemed to be derivative liabilities and, therefore, the fair value of the warrants was recorded in the current liability section of the consolidated balance sheet. The outstanding warrants, accounted for as derivative liabilities, were revalued each reporting period with the resulting gains and losses recorded as the change in fair value of derivative liabilities on the consolidated statement of operations. Upon exercise of the warrant or modification of the warrant agreements to remove the terms that required derivative liability treatment, the derivative liabilities are revalued on the date that such event occurs and the remaining value is reclassified to additional paid in capital. In connection with the Warrant Tender Offer, all derivative liabilities outstanding were extinguished and we applied the extinguishment of liabilities model to determine the loss on extinguishment of derivative liabilities. The consideration transferred to extinguish the derivative liabilities was recorded at fair value and compared to the cash received in connection with the tender offer to determine the loss on extinguishment of the derivative liabilities.
We used the Black-Scholes option-pricing model to estimate the fair value of the issued and outstanding warrants upon issuance and through December 12, 2016.
Stock-Based Compensation
We account for our stock-based compensation awards to employees and directors in accordance with ASC Topic 718, Compensation-Stock Compensation (“ASC 718”). ASC 718 requires all stock-based payments to employees, including grants of employee stock options and restricted stock, to be recognized in the audited consolidated statements of operations and comprehensive income (loss) based on their grant date fair values. Compensation expense related to awards to employees is recognized on a straight-line basis based on the grant date fair value over the associated service period of the award, which is generally the vesting term. The vesting of employee and director awards is generally time-based, but certain option awards for employees provide for vesting of all or a portion of the shares underlying such option upon the achievement of certain milestones or performance-based criteria. Share-based payments issued to non-employees are recorded at their fair values, and are periodically revalued as the equity instruments vest and are recognized as expense over the related service period in accordance with the provisions of ASC 718 and ASC Topic 505, Equity.
| 39 |
We estimate the fair value of our stock options using the Black-Scholes option pricing model, which requires the input of subjective assumptions, including (a) the expected stock price volatility, (b) the expected term of the award, (c) the risk-free interest rate, (d) expected dividends, and (e) the estimated fair value of our common stock on the measurement date. As of January 1, 2016, we began using a blended average of our historical volatility and the historical volatility of a group of similarly situated companies to calculate the expected volatility when valuing our stock options. For purposes of calculating this blended volatility, we selected companies with comparable characteristics to us, including enterprise value, risk profiles, position within the industry, and with historical share price information sufficient to meet the expected term of the stock-based awards. We compute historical volatility data using the daily closing prices for our company and the selected companies’ shares during the equivalent period of the calculated expected term of the stock-based awards. Prior to January 1, 2016, due to the lack of a public market for the trading of our common stock and a lack of company specific historical and implied volatility data, we have based our estimate of expected volatility only on the historical volatility of a group of similarly situated companies that were publicly traded. Due to the lack of our specific historical option activity, we have estimated the expected term of our employee stock options using the “simplified” method, whereby, the expected term equals the arithmetic average of the vesting term and the original contractual term of the option. The expected term for non-employee awards is the remaining contractual term of the option. The risk-free interest rates are based on the U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. We have never paid dividends, and do not expect to pay dividends in the foreseeable future.
Effective January 1, 2016, we have elected to account for forfeitures as they occur, as permitted by ASU No. 2016-09, Compensation-Stock Compensation (Topic 718), Improvements to Employee Share-Based Payment Accounting. See the Accounting Standards Adopted in the Period section below for further details.
Prior to the adoption of ASU No. 2016-09, we estimated the number stock-based awards that were expected to vest, and only recognized compensation expense for such awards. The estimation of stock awards that will ultimately vest required judgment, and to the extent actual results or updated estimates differed from current estimates, such amounts were recorded as a cumulative adjustment in the period estimates were revised. We considered many factors when estimating expected forfeitures, including type of awards granted, employee class, and historical experience.
Our common stock is publicly traded, and fair market value is determined based on the closing sales price of our common stock on the OTCQB.
Recent Accounting Pronouncements
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers (Topic 606). ASU No. 2014-09 provides for a single comprehensive model for use in accounting for revenue arising from contracts with customers and supersedes most current revenue recognition guidance. The accounting standard is effective for interim and annual periods beginning after December 15, 2016 with no early adoption permitted. In August 2015, the FASB issued ASU No. 2015-14, Revenue from Contracts with Customers (Topic 606): Deferral of the Effective Date, which deferred the effective date of ASU No. 2014-09 to annual periods beginning after December 15, 2017, along with an option to permit early adoption as of the original effective date. In April 2016, the FASB issued ASU No. 2016-10, Revenue from Contracts with Customers (Topic 606): Identifying Performance Obligations and Licensing. The ASU clarifies the following two aspects of Topic 606: (a) identifying performance obligations; and (b) the licensing implementation guidance. The ASU does not change the core principle of the guidance in Topic 606. In May 2016, the FASB issued ASU No. 2016-12, Revenue from Contracts with Customers (Topic 606): Narrow-Scope Improvements and Practical Expedients, related to disclosures of remaining performance obligations, as well as other amendments to guidance on collectability, non-cash consideration and the presentation of sales and other similar taxes collected from customers. We are required to adopt the amendments in ASU No. 2014-09 using one of two acceptable methods. The new revenue standard allows for either full retrospective or modified retrospective application. We anticipate using the modified retrospective approach to implement this standard. The effective date and transition requirements for the ASUs are the same as the effective date and transition requirements in Topic 606. Public entities should apply the ASUs for annual reporting periods beginning after December 15, 2017, including interim reporting periods therein (i.e., January 1, 2018, for a calendar year entity). Early application for public entities is permitted only as of annual reporting periods beginning after December 15, 2016, including interim reporting periods within that reporting period. We do not expect to record a material adjustment upon adoption as our current contracts with customers will be completed prior to adoption of the new standard using the modified retrospective application, which requires a cumulative effect of initially applying the standard to opening accumulated deficit as of January 1, 2018. We do anticipate changes in our revenue recognition policies for any revenue generating contracts we may enter into in the future.
| 40 |
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842). ASU No. 2016-02 is effective for annual periods beginning after December 15, 2018, and requires a lessee to recognize assets and liabilities for leases with a maximum possible term of more than 12 months. A lessee would recognize a liability to make lease payments (the lease liability) and a right-of-use asset representing its right to use the leased asset (the underlying asset) for the lease term. Early application is permitted. We are currently evaluating the impact the adoption of the accounting standard will have on our consolidated financial statements.
In November 2016, the FASB issued ASU No. 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash. ASU 2016-18 requires entities to reconcile and explain the period-over-period change in total cash, cash equivalents and restricted cash within its statements of cash flows. ASU 2016-18 is effective for fiscal years, and interim periods within, beginning after December 15, 2017. Early adoption is permitted. A reporting entity must apply the amendments in ASU 2016-18 using a full retrospective approach. We are currently evaluating the impact the adoption of the ASU will have on our consolidated financial statements.
Accounting Standards Adopted in the Period
In March 2016, the FASB issued ASU No. 2016-09, Compensation—Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting, which simplified several aspects of employee share-based payment accounting. In particular, ASU No. 2016-09 permits entities to make an accounting policy election to either estimate forfeitures on share-based payment awards, as previously required, or to recognize forfeitures as they occur. Effective January 1, 2016, we elected to recognize forfeitures as they occur. The impact of that change in accounting policy has been recorded as an $8,333 cumulative effect adjustment to accumulated deficit, as of December 31, 2015. We recognized slightly higher share-based payment expense for the remainder of 2016, relative to prior periods, as the effects of forfeitures were not to be recognized until they occurred, rather than being estimated at the time of grant and subsequently adjusted as and when necessary. The effects of adopting the remaining provisions in ASU No. 2016-09 affecting the income tax consequences of share-based payments, classification of awards as either equity or liabilities when an entity partially settles the award in cash in excess of the employer’s minimum statutory withholding requirements, and classification in the statement of cash flows did not have any impact on our financial position, results of operations or cash flows.
In April 2015, the FASB issued ASU No. 2015-03, Interest – Imputation of Interest (Subtopic 305-40): Simplifying the Presentation of Debt Issuance Costs. The new guidance requires the debt issuance costs related to a recognized debt liability be presented in the balance sheet as a direct deduction from the carrying amount of that debt liability. This guidance will be effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2015. Accordingly, the standard was effective for us on January 1, 2016.
In August 2014, the FASB issued ASU No. 2014-15, Presentation of Financial Statements-Going Concern (Subtopic 205-40). ASU No. 2014-15 requires management to evaluate for the existence of conditions or events that raise substantial doubt about the entity’s ability to continue as a going concern and to provide related footnote disclosures. The standard provides guidance to management, with principles and definitions that are intended to reduce diversity in the timing and content of disclosures that companies commonly provide in their footnotes. Under the new standard, management must decide whether there are conditions or events, considered in the aggregate, that raise substantial doubt about the company’s ability to continue as a going concern within one year after the issuance date of the financial statements. The accounting standard is effective for annual periods ending after December 15, 2016 and for interim periods thereafter. We adopted this in the fourth quarter of 2016.
Other accounting standards that have been issued by the FASB or other standards-setting bodies that do not require adoption until a future date are not expected to have a material impact on our consolidated financial statements upon adoption.
Off-Balance Sheet Arrangements
We did not engage in any “off-balance sheet arrangements” (as that term is defined in Item 303(a)(4)(ii) of Regulation S-K) as of December 31, 2016.
| 41 |
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK.
We are exposed to market risk related to changes in interest rates. As of December 31, 2016 and December 31, 2015, we had cash and cash equivalents of $3,162,400 and $3,596,262, respectively, consisting primarily of money market funds. The investments in these financial instruments are made in accordance with an investment policy approved by our board of directors, which specifies the categories, allocations and ratings of securities we may consider for investment. The primary objective of our investment activities is to preserve principal while at the same time maximizing the income we receive without significantly increasing risk. Some of the financial instruments in which we invest could be subject to market risk. This means that a change in prevailing interest rates may cause the value of the instruments to fluctuate. For example, if we purchase a security that was issued with a fixed interest rate and the prevailing interest rate later rises, the value of that security will probably decline. To minimize this risk, we intend to maintain a portfolio that may include cash, cash equivalents and investment securities available-for-sale in a variety of securities, which may include money market funds, government and non-government debt securities and commercial paper, all with various maturity dates. Based on our current investment portfolio, we do not believe that our results of operations or our financial position would be materially affected by an immediate change of 10% in interest rates.
We do not hold or issue derivatives, derivative commodity instruments or other financial instruments for speculative trading purposes. Further, we do not believe our cash equivalents and investment securities have significant risk of default or illiquidity. We made this determination based on discussions with our investment advisors and a review of our holdings. Although we believe our cash equivalents and investment securities do not contain excessive risk, we cannot provide absolute assurance that in the future our investments will not be subject to adverse changes in market value. All of our investments are recorded at fair value.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA.
Please see our Financial Statements beginning on page F-1 of this Annual Report on Form 10-K.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE.
None.
ITEM 9A. CONTROLS AND PROCEDURES.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our chief executive officer and our vice president of finance, chief accounting officer and treasurer (our principal executive officer and principal financial officer, respectively), evaluated the effectiveness of our disclosure controls and procedures as of December 31, 2016. The term “disclosure controls and procedures”, as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, or Exchange Act, means controls and other procedures of a company that are designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate, to allow timely decisions regarding required disclosure. Management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Based on the evaluation of our disclosure controls and procedures as of December 31, 2016, our principal executive officer and principal financial officer concluded that, as of such date, our disclosure controls and procedures were effective at the reasonable assurance level.
| 42 |
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Internal control over financial reporting, as defined in Rules 13a-15(f) and 15d-15(f) promulgated under the Exchange Act, is a process designed by, or under the supervision of, our principal executive officer and principal financial officer and effected by our management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP and includes those policies and procedures that:
| · | pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets; |
| · | provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with GAAP, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and |
| · | provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. In addition, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures included in such controls may deteriorate.
Our management assessed the effectiveness of our internal control over financial reporting as of December 31, 2016. In making this assessment, our management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control-Integrated Framework (2013). Based on our assessment, management concluded that, as of December 31, 2016, our internal control over financial reporting was effective based on those criteria.
Attestation Report of Registered Public Accounting Firm
This Annual Report on Form 10-K does not include an attestation report of our registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by our independent registered public accounting firm pursuant to rules of the SEC that permit us to provide only management’s report in this Annual Report.
Changes in Internal Control Over Financial Reporting
There was no change in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act) that occurred during the quarter ended December 31, 2016 that has materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
None.
| 43 |
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE.
Directors and Executive Officers
Below are the names of and certain information regarding our current executive officers and directors as of March 17, 2017:
| Name | Age | Position | Date Named to Board of Directors/as Executive Officer |
Director Class (if applicable) | ||||
| Wael Fayad | 50 | Chairman of the Board, President and Chief Executive Officer | September 21, 2016 | I | ||||
| Kevin G. Sarney | 52 | Vice President of Finance, Chief Accounting Officer and Treasurer | July 31, 2014 | - | ||||
| Barry Buckland, Ph.D. | 69 | Director | July 31, 2014 | II | ||||
| Robert J. Easton | 72 | Director | July 30, 2015 | III | ||||
| Allan P. Rothstein | 58 | Director | July 31, 2014 | II | ||||
| Paul J. Sekhri | 58 | Director | December 16, 2014 | III | ||||
| Robert L. Van Nostrand | 59 | Director | December 1, 2014 | III |
Our board of directors is divided into three classes, designated Class I, Class II and Class III. Each class consists, as nearly as may be possible, of one-third of the total number of directors constituting the entire board of directors. At our last annual meeting of stockholders, the Class I directors were elected for a three -year term which will expire at the 2018 annual meeting of stockholders. The Class II directors were elected initially for a two-year term which was due to expire at the 2016 annual meeting of stockholders (which has not yet been held and therefore such persons will continued to serve until their successors are duly elected and qualified in accordance with our By-Laws). The Class III directors were elected initially for a three-year term which will expire at the 2017 annual meeting of stockholders. At each succeeding annual meeting of stockholders, successors to the class of directors whose term expires at that annual meeting will be elected for a three-year term. However, we anticipate that the next annual meeting of stockholders will also include an election for Class II directors, whose term was initially expected to expire in 2016, and those directors will be elected to a two-year term. If the number of directors is changed, any increase or decrease will be apportioned among the classes so as to maintain the number of directors in each class as nearly equal as possible, and any additional director of any class will hold office for a term that will coincide with the remaining term of that class, but in no case will a decrease in the number of directors shorten the term of any incumbent director. As a result of the resignations of John J. Rydzewski and Arthur H. Tinkelenberg, we currently have an unequal number of directors in each class.
A majority of the authorized number of directors constitutes a quorum of the board of directors for the transaction of business. The directors must be present at the meeting to constitute a quorum. However, any action required or permitted to be taken by the board of directors may be taken without a meeting if all members of the board of directors individually or collectively consent in writing to the action.
| 44 |
Executive officers are appointed by the board of directors and serve at its pleasure.
The principal occupation and business experience during the past five years for our executive officers and directors is as follows:
Wael Fayad –Chairman of the Board of Directors, President and Chief Executive Officer
Mr. Fayad has served as the Chairman of our board of directors, President and Chief Executive Officer since September 2016. From January 2015 until September 2016, Mr. Fayad was engaged in a number of entrepreneurial ventures, including helping to launch Valentine & Cautreels, Inc., a start-up company focusing on improving healthcare outcomes and reducing the total cost of care, in October 2015. Mr. Fayad continues to serve on Valentine & Cautreels’ advisory board. From January 2001 until January 2015, Mr. Fayad served in roles of increasing responsibilities at Forest Laboratories, Inc., culminating in his service as Corporate Vice President, Global Business Development. In that role, Mr. Fayad was responsible for business development and alliance management, including the identification, assessment and negotiation of multiple business opportunities. Prior to Forest, Mr. Fayad served in positions of increasing responsibility at Schering-Plough and Novartis in the fields of sales, marketing, and new product development. Mr. Fayad holds a B.S. in biology from the American University of Beirut, and an M.B.A. from Concordia University. The board of directors has concluded that Mr. Fayad should serve on our board because of his significant experience as an executive in the life sciences industry.
Kevin G. Sarney – Vice President of Finance, Chief Accounting Officer and Treasurer
Kevin G. Sarney has served as our Vice President of Finance, Chief Accounting Officer and Treasurer since the Merger. From September 2013 to July 2014, Mr. Sarney served as Vice President, Finance and Administration for Avaxia Biologics, Inc., a privately-held clinical-stage biopharmaceutical company developing gut-targeted therapeutics. Prior to joining Avaxia, Mr. Sarney provided financial consulting services to various life sciences companies from May 2009 to August 2013, including Helicos BioSciences Corporation, a publicly traded medical device sequencing company, from December 2009 to May 2010 and Archemix Corp., a privately held aptamer therapeutics company, from June 2010 to February 2012. In addition, from February 2012 to August 2013, Mr. Sarney provided consulting services for various life sciences companies, including Tetraphase Pharmaceuticals, Inc., Tesaro Inc., Karyopharm Therapeutics Inc., and Genocea Biosciences, Inc. From March 2005 to April 2009, Mr. Sarney held positions of increasing responsibility at NitroMed, Inc., a publicly traded cardiovascular-focused pharmaceutical company, culminating as Corporate Controller and Principal Accounting Officer. Mr. Sarney earned a B.S. in business management from the University of Hartford, an M.B.A. from Boston University, and an M.A. in accounting from Suffolk University. Mr. Sarney is a certified public accountant in the Commonwealth of Massachusetts.
Barry Buckland, Ph.D. – Director, Co-Founder and Chairman of the Scientific Advisory Board
Dr. Buckland has been a member of our board of directors since the Merger and prior thereto served as member of the board of directors of Enumeral since December 2009. Dr. Buckland’s career in the pharmaceutical industry includes his leadership of Merck’s bioprocess Research and Development Group, which saw the market launch of major biological products, including Gardasil, Rotateq, and Zostavax. Since May 2009, Dr. Buckland has been the Chief Executive Officer of BiologicB LLC, a consulting firm working with biotechnology and vaccine products. Dr. Buckland is a member of the supervisory board of Mucosis BV, a privately held Netherlands company focused on developing a proprietary mucosal vaccine technology platform. Dr. Buckland also serves on the board of directors for Engineering Conferences International, a not-for-profit global engineering conferences program. Dr. Buckland is the recipient of numerous awards, among them the Donald Medal, UK Institute of Chemical Engineering in 2002, Prix Galien award as a member of the team that received the Vaccine Award for Gardasil in 2007, the Marvin Johnson award by ACS for lifetime contribution to Biotechnology in 2008, and the PhRMA Discoverer of the Year for development of the Merck HPV vaccine (along with Eliav Barr and Kathrin Jansen) in 2009. He has chaired multiple International Conferences related to Bioprocess Research and Development and Vaccine Technology and is an author on over 70 papers. He has been a Visiting Professor at University College London since 1995. Dr. Buckland earned his Ph.D. in Biochemical Engineering at University College London in 1974. The board of directors has concluded that Dr. Buckland should serve on our board because of his significant experience in the pharmaceutical and biotechnology industries.
| 45 |
Robert J. Easton – Director
Mr. Easton has been a member of our board of directors since 2015. He is a recognized leader in the life sciences and healthcare industries, having led strategy development and supervised opportunity assessments for hundreds of clients across the globe, including large and specialty pharmaceutical companies, early-stage and publicly traded biopharmaceutical companies, and diagnostics businesses. Mr. Easton currently serves as Co-chairman of Bionest Partners, Inc., a consulting firm he co-founded that specializes in strategic planning for pharmaceutical, medical device and diagnostic companies. Prior to co-founding Bionest Partners, Mr. Easton built and led two consulting firms, Easton Associates and The Wilkerson Group, which provided advisory services in the healthcare, pharmaceuticals and medical products industries. Previously, Mr. Easton spent 12 years in various management roles at Union Carbide and Union Carbide Europe, including in marketing and engineering in the clinical diagnostics business unit. From 2002 until November 2016, Mr. Easton served on the Board of Directors of Cepheid, a molecular diagnostics company, where he was a member of the Compensation Committee, the Audit Committee, and the Nominating and Governance Committee. He currently serves on the Board of Advisors of the Global Cardiovascular Innovation Center at the Cleveland Clinic, and is past chairman of the New York Biotechnology Association. Mr. Easton holds B.S. and B.A. degrees in chemical engineering from Rice University, and an M.B.A. from Harvard University. Mr. Easton serves as Chairman of our Audit Committee. The board of directors has concluded that Mr. Easton should serve on our board because of his significant experience in the life sciences and healthcare industries.
Allan P. Rothstein – Director
Mr. Rothstein has been a member of our board of directors since the Merger, and prior thereto served as member of the board of directors of Enumeral since September 2013. Since April 2004, Mr. Rothstein has owned and managed Hedge Capital Partners LLC, an investment vehicle for venture capital, portfolio management and asset management. Mr. Rothstein has also owned Shizoom LLC, a business funding company, since it was formed in March 2013. Mr. Rothstein has served as founder and president of Empire Spirits Project, Inc., a New York-based distillery, since 2013. Since April 2015, Mr. Rothstein has also served as a partner and Chairman of The Agency Worx LLC, which specializes in staffing, recruitment and relationship management. From September 2010 to August 2011, Mr. Rothstein was head of trading at Centurion Partners. Mr. Rothstein served as a principal of Healthcare Recovery Services LLC, which services financial aspects and enhances revenue cycles of hospitals and other healthcare service enterprises, from December 2011 through November 2014. From 2002 through 2007, Mr. Rothstein was co-founder and Chairman of NanoDynamics Inc. He has served as a member of the National Nanotechnology Business Alliance and the New York Nanotechnology Business Alliance advisory boards and the Dean’s Council of the Graduate School of SUNY Stonybrook. Mr. Rothstein has over 30 years’ experience in the financial industry, having started his career at Shearson Loeb Rhoades where he handled arbitrage lines for domestic and international customers. In 1982, he became a member of the New York Futures Exchange. In 1990 he joined Fahnestock & Company (presently, Oppenheimer & Company) as a Nasdaq market maker. In his role as Senior Vice President and co-head of Nasdaq Trading, Mr. Rothstein was responsible for trading in over 500 markets, oversight of all Nasdaq market making and trading systems construction and integration. Mr. Rothstein graduated as a Benjamin Franklin Scholar from The University of Pennsylvania, magna cum laude, in 1980. The board of directors has concluded that Mr. Rothstein should serve on our board because of his significant experience as an investor in, and advisor to, emerging technology companies.
| 46 |
Paul J. Sekhri – Director
Mr. Sekhri has been a member of our board of directors since December 2014. Since February 2015, Mr. Sekhri has served as President and Chief Executive Officer and a member of the board of directors of Lycera Corp., a biopharmaceutical company that is developing medicines to treat cancer and autoimmune disease. From April 2014 to February 2015, Mr. Sekhri was Senior Vice President, Integrated Care at Sanofi S.A., a multinational pharmaceutical company, where he led the company’s efforts to create innovative solutions and business models to meet patient needs. Prior to Sanofi, Mr. Sekhri served from June 2013 to March 2014 as Group Executive Vice President, Global Business Development and Chief Strategy Officer for Teva Pharmaceutical Industries, Ltd., a multinational pharmaceutical company. From January 2009 to May 2014, Mr. Sekhri served as Operating Partner and Head of the Biotechnology Operating Group at TPG Biotech, the life sciences venture capital arm of TPG Capital. From 2004 to 2009, Mr. Sekhri was Founder, President, and Chief Executive Officer of Cerimon Pharmaceuticals, Inc. Prior to founding Cerimon, Mr. Sekhri was President and Chief Business Officer of ARIAD Pharmaceuticals, Inc. from 2003 to 2004. From 2002 to 2003, Mr. Sekhri was a partner at New Leaf Ventures, where he focused on healthcare investments. From 1999 to 2003, Mr. Sekhri held various positions at Novartis Pharma AG, including Senior Vice President and Head of Global Search and Evaluation, Business Development and Licensing. Mr. Sekhri serves on the board of directors of Veeva Systems Inc., a publicly-held cloud-based software company focused on the life sciences industry, and Nivalis Therapeutics, Inc., a publicly held clinical stage pharmaceutical company that focuses on the discovery, development, and commercialization of drug candidates for people with cystic fibrosis. Mr. Sekhri also serves as Chairman of the Board of Supervisory Directors of Pharming N.V., a listed Netherlands biotechnology company focused on the development and production of human therapeutic proteins, and as a director of Petra Pharma Corporation, a privately held early stage biotechnology company. In addition, Mr. Sekhri serves on several non-profit boards, including the BioExec Institute, Inc., the TB Alliance, Young Concert Artists, Inc., the Caramoor Center for Music and the Arts, and the English Concert in America (TECA). Mr. Sekhri is also a member of the Patrons Council of Carnegie Hall, where he served as a member of the Board of Trustees from 2010 to 2012. Mr. Sekhri holds a B.S. in Zoology from the University of Maryland. The board of directors has concluded that Mr. Sekhri should serve on our board because of his significant experience as a senior executive and director in the life sciences industry.
Robert L. Van Nostrand – Director
Mr. Van Nostrand has been a member of our board of directors since December 2014. Since 2010, Mr. Van Nostrand has served as an advisor and board member to several biotechnology companies. From January 2010 to July 2010, Mr. Van Nostrand was Executive Vice President and Chief Financial Officer of Aureon Laboratories, Inc., a pathology life sciences company. Prior to joining Aureon Laboratories, Mr. Van Nostrand served as Executive Vice President and Chief Financial Officer of AGI Dermatics, Inc., a private biotechnology company, from July 2007 to September 2008, at which time the company was acquired. From May 2005 to July 2007, Mr. Van Nostrand served as the Senior Vice President and Chief Compliance Officer of OSI Pharmaceuticals, Inc. a biotechnology company, where he previously served as Vice President and Chief Financial Officer from December 1996 through May 2005, and as Vice President, Finance and Administration prior to that. He also served as OSI’s Treasurer from March 1992 to May 2005, and as Secretary from March 1995 to January 2004. Mr. Van Nostrand joined OSI as Controller and Chief Accounting Officer in September 1986. Prior to joining OSI, Mr. Van Nostrand served in a managerial position with the accounting firm of Touche Ross & Co. (now Deloitte). Mr. Van Nostrand serves on the board of directors of Achillion Pharmaceuticals, Inc., a publicly-held biotechnology company, where he is chairman of the audit committee and a member of the compensation committee. Mr. Van Nostrand also serves on the board of directors and is chairman of the audit committee of Intra-Cellular Therapies, Inc. and Yield10 Bioscience, Inc. (formerly known as Metabolix, Inc.), both publicly-held biotechnology companies. In addition, Mr. Van Nostrand is chairman of the board of directors at Yield10 Bioscience. Mr. Van Nostrand is on the board of the Biomedical Research Alliance of New York, a private company providing clinical trial services, as well as the Board of the New York Biotechnology Association, where he previously served as chairman. In addition, he is on the Foundation Board of Farmingdale University. Mr. Van Nostrand holds a B.S. in Accounting from Long Island University, New York, and he is a Certified Public Accountant. Mr. Van Nostrand has also completed advanced management studies at the Wharton School of the University of Pennsylvania. The board of directors has concluded that Mr. Van Nostrand should serve on our board because of his significant experience as a senior executive and director in the life sciences industry.
John J. Rydzewski resigned as Executive Chairman and a director, effective as of September 21, 2016. Arthur H. Tinkelenberg was terminated as President and Chief Executive Officer effective as of July 28, 2016 and he subsequently resigned as a director as of September 19, 2016. Anhco Nguyen resigned from his position as Vice President of Research and Development, effective as of August 17, 2016.
Director Relationships and Arrangements
Except for Messrs. Easton, Sekhri and Van Nostrand, none of our current directors or executive officers have served as a director of another public company within the past five years.
To our knowledge, there are no arrangements or understandings between any director, director nominee or executive officer and any other person pursuant to which any person was selected as a director, director nominee or executive officer, other than the Voting Agreement, which has expired. There are no family relationships between any of our directors, director nominees or executive officers. To our knowledge, there have been no material legal proceedings as described in Item 401(f) of Regulation S-K during the last ten years that are material to an evaluation of the ability or integrity of any of our directors, director nominees or executive officers. In addition, to our knowledge, there are no pending legal proceedings to which any of the directors or executive officers is a party adverse to or has a material interest that is adverse to our company.
| 47 |
Board of Directors and Corporate Governance
While we are currently quoted on the over-the-counter market, we have adopted the corporate governance standards of a listed company on the Nasdaq Stock Market, or Nasdaq. These standards require that a majority of the members of our board of directors be “independent,” as Nasdaq defines that term, and that our board make an affirmative determination as to the independence of each director. Consistent with these rules, our board of directors undertook its annual review of director independence in March 2017. During the review, our board considered relationships and transactions during 2016 and during the past three fiscal years between each director or any member of his or her immediate family, on the one hand, and our company and its subsidiaries and affiliates, on the other hand. The purpose of this review was to determine whether any such relationships or transactions were inconsistent with a determination that the director is independent. Based on this review, our board of directors determined that Dr. Buckland and Messrs. Easton, Rothstein, Sekhri, and Van Nostrand are independent under the criteria established by Nasdaq and by our board of directors.
Our board of directors held 20 meetings during 2016, and each incumbent director standing for election attended at least 75% of the meetings of the board and the meetings of those committees on which each incumbent director served, in each case during the period that such person was a director. The permanent committees established by our board of directors are the audit committee, the compensation committee, and the nominating and governance committee, descriptions of which are set forth in more detail below.
Each member of our board of directors is expected to participate, either in person or via teleconference, in meetings of the board and meetings of committees of which each director is a member, and to spend the time necessary to properly discharge such director’s respective duties and responsibilities. Although we do not have a written policy regarding directors’ attendance at annual meetings of stockholders, all directors are encouraged to attend. Four directors attended our annual meeting of stockholders in 2015. We did not hold an annual meeting of stockholders in 2016.
Board Leadership Structure
Our board of directors is currently chaired by Mr. Fayad. The Board Chairman has authority, among other things, to call and preside over board of directors meetings, to set meeting agendas and to determine materials to be distributed to the board. Following the resignations of Dr. Tinkelenberg and Mr. Rydzewski, we appointed Mr. Fayad to serve as both our Board Chairman and our President and Chief Executive Officer. In light of our financial resources, size and stage of business development, we believe that our leadership structure is appropriate.
The board has designated Mr. Van Nostrand as the lead independent director. We believe that the position of lead independent director reinforces the independence of the board in its oversight of our business and affairs. The lead independent director, among his other duties and responsibilities, serves as chairman at sessions of the independent and non-employee directors, and is the principal liaison between the independent and non-employee directors and the Board Chairman, President and Chief Executive Officer, and our senior management. The lead independent director consults with the Board Chairman regarding the schedule of board meetings, as well as the agenda and information to be presented at such meetings. When the Board Chairman is not present, the lead independent director acts as chairman of board meetings. Pursuant to the lead independent director charter, the individual holding that position must meet the independence requirements set forth in the applicable rules and listing standards of Nasdaq, or such other national securities exchange on which our securities are then listed.
Role of the Board in Risk Oversight
Our board of directors has an active role in overseeing the management of our risks, which the board administers directly as well as through various standing committees of the board that address risks inherent in their respective areas of oversight. In particular, our board of directors is responsible for monitoring and assessing strategic risk exposure, including information regarding our credit, liquidity and operations and the risks associated with each. Our primary risks are currently associated with the development of our platform technology, including our ability to raise additional capital to complete the development and potential commercialization of product candidates using our platform technology. The audit committee of the board of directors has the responsibility to consider and discuss our major financial risk exposures and the steps that our management has taken to monitor and control these exposures. The audit committee also monitors compliance with legal and regulatory requirements, in addition to oversight of the performance of our internal controls over financial reporting. The nominating and governance committee of the board of directors monitors the effectiveness of our corporate governance guidelines, and manages risks associated with the independence of the board of directors and potential conflicts of interest. The compensation committee of the board of directors assesses and monitors whether any of our compensation policies and programs has the potential to encourage excessive risk-taking. While each committee is responsible for evaluating certain risks and overseeing management of such risks, the entire board of directors is regularly informed through committee reports about such risks.
| 48 |
Communicating with the Board of Directors
Stockholders can mail communications to our board of directors to our Corporate Secretary, Enumeral Biomedical Holdings, Inc., 200 CambridgePark Drive, Suite 2000, Cambridge, Massachusetts 02140, who will forward the correspondence to each addressee.
Board Committees
Audit Committee
The audit committee was established on August 4, 2014 and currently consists of Mr. Easton, as chairman, and Messrs. Sekhri and Van Nostrand. The audit committee held four committee meetings during 2016.
The duties and responsibilities of the audit committee are set forth in the charter of the audit committee. A copy of the charter of the audit committee is available under the Investors—Corporate Governance section of our corporate website at www.enumeral.com. Among other things, the duties and responsibilities of the audit committee include reviewing and monitoring our consolidated financial statements and internal accounting procedures, the selection of our independent registered public accounting firm and consulting with and reviewing the services provided by our independent registered public accounting firm. The audit committee has sole discretion over the retention, compensation, evaluation and oversight of our independent registered public accounting firm.
The Unites States Securities and Exchange Commission, or SEC, and Nasdaq have established rules and regulations regarding the composition of audit committees and the qualifications of audit committee members. Our board of directors has examined the composition of the audit committee and the qualifications of the audit committee members in light of the current rules and regulations governing audit committees. Based upon this examination, our board of directors has determined that each member of the audit committee is independent and is otherwise qualified to be a member of the audit committee in accordance with the rules of the SEC and Nasdaq.
Additionally, the SEC requires that at least one member of the audit committee have a “heightened” level of financial and accounting sophistication. Such a person is known as the “audit committee financial expert” under the SEC’s rules. Our board of directors has determined that Mr. Van Nostrand satisfies the definition of “audit committee financial expert” in the rules and regulations of the SEC, and is an independent member of the board of directors and the audit committee.
Compensation Committee
The compensation committee was established on August 4, 2014 and currently consists of Mr. Sekhri, as chairman, Dr. Buckland and Mr. Van Nostrand. The compensation committee held three committee meetings during 2016.
The duties and responsibilities of the compensation committee are set forth in the charter of the compensation committee. A copy of the charter of the compensation committee is available under the Investors—Corporate Governance section of our corporate website at www.enumeral.com. As discussed in its charter, among other things, the duties and responsibilities of the compensation committee include evaluating the performance of our executive officers, determining the overall compensation of our executive officers and administering all executive compensation programs, including, but not limited to, our incentive and equity-based plans. The compensation committee evaluates the performance of our executive officers on an annual basis and reviews and approves on an annual basis all compensation programs and awards relating to such officers. The compensation committee applies discretion in the determination of individual executive compensation packages to ensure compliance with our compensation philosophy. Our chief executive officer makes recommendations to the compensation committee with respect to the compensation packages for officers other than himself.
| 49 |
Nasdaq has established rules and regulations regarding the composition of compensation committees and the qualifications of compensation committee members. Our board of directors has examined the composition of the compensation committee and the qualifications of the compensation committee members in light of the current rules and regulations governing compensation committees. Based upon this examination, our board of directors has determined that each member of the compensation committee is independent and is otherwise qualified to be a member of the compensation committee in accordance with such rules.
None of our executive officers has served as a member of the compensation committee (or other committee serving an equivalent function) of any other entity.
Nominating and Governance Committee
The nominating and governance committee was established in February 2015 and currently consists of Dr. Buckland, as chairman, and Messrs. Sekhri and Van Nostrand. The nominating and governance committee held one committee meeting during 2016.
The duties and responsibilities of the nominating and governance committee are set forth in the nominating and governance committee charter. A copy of the nominating and governance committee charter is available under the Investors—Corporate Governance section of our corporate website at www.enumeral.com. Among other things, the duties and responsibilities of the nominating and governance committee include identifying individuals qualified to become board members, recommending director nominees to the board for the next annual meeting of stockholders, evaluating the overall effectiveness of the board, developing, mentoring and evaluating our corporate governance practices, and performing such other duties as enumerated in and consistent with its charter.
The nominating and governance committee will also consider candidates recommended by stockholders for nomination to the board of directors. A stockholder who wishes to recommend a candidate for nomination to the board of directors must submit such recommendation to Enumeral Biomedical Holdings, Inc., 200 CambridgePark Drive, Suite 2000, Cambridge, Massachusetts 02140; Attention: Corporate Secretary. All stockholder recommendations of candidates for nomination for election to the board of directors must be in writing and must set forth the following: (i) the candidate’s name, age, business address, and other contact information, (ii) the number of shares of our common stock beneficially owned by the candidate, (iii) a complete description of the candidate’s qualifications, experience, background and affiliations, as would be required to be disclosed in the proxy statement pursuant to Schedule 14A under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), (iv) a sworn or certified statement by the candidate in which he or she consents to being named in the proxy statement as a nominee and to serve as director if elected, and (v) the name and address of the stockholder(s) of record making such a recommendation and the number of shares owned by the recommending stockholder(s).
We believe that our board of directors as a whole should encompass a range of talent, skill and expertise enabling it to provide sound guidance with respect to our operations and interests. The nominating and governance committee evaluates all candidates for the board of directors by reviewing their biographical information and qualifications. The nominating and governance committee then determines, based on the background information and the information obtained in the interviews, whether to recommend to the board of directors that the candidate be nominated for approval by the stockholders to fill a directorship. With respect to an incumbent director whom the nominating and governance committee is considering as a potential nominee for re-election, the nominating and governance committee reviews and considers the incumbent director’s service during his or her term, including the number of meetings attended, level of participation, and overall contribution to the board of directors. The manner in which the nominating and governance committee evaluates a potential nominee will not differ based on whether the candidate is recommended by the directors or stockholders.
Nasdaq has established rules and regulations regarding the composition of nominations committees and the qualifications of nominations committee members. The board of directors has examined the composition of the nominating and governance committee and the qualifications of the members of the nominating and governance committee in light of the current rules and regulations governing nominations committees. Based upon this examination, the board of directors has determined that each member of the nominating and governance committee is independent and is otherwise qualified to be a member of the nominating and governance committee in accordance with such rules.
| 50 |
We do not have a formal policy in place with regard to the consideration of diversity of candidates for the board of directors, but the board of directors strives to nominate candidates with a variety of complementary skills so that, as a group, the board of directors will possess the appropriate talent, skills and expertise to oversee our business.
Code of Ethics and Business Conduct
We have adopted a written code of ethics and business conduct that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. A current copy of the code of ethics and business conduct is available under the Investors—Corporate Governance section of our corporate website at www.enumeral.com. A copy of the code of ethics and business conduct will also be provided to any person, without charge, upon written request sent to Enumeral Biomedical Holdings, Inc., 200 CambridgePark Drive, Suite 2000, Cambridge, Massachusetts 02140; Attention: Corporate Secretary. Any amendments to or waivers of the code of ethics and business conduct will be promptly posted on our website at www.enumeral.com or in a Current Report on Form 8-K, as required by applicable laws.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Exchange Act requires our directors and officers and holders of more than 10% of our common stock to file with the SEC initial reports of ownership of our common stock and other equity securities on a Form 3 and reports of changes in such ownership on a Form 4 or Form 5. Directors and officers and holders of 10% of our common stock are required by SEC regulations to furnish us with copies of all Section 16(a) forms they file. We registered our common stock under Section 12(g) of the Exchange Act on April 3, 2015. To our knowledge, based solely on a review of our records and representations made by our directors and officers regarding their filing obligations, all Section 16(a) filing requirements were satisfied with respect to the applicable period in 2016.
Related Person Transaction Approval Policy
While we have no written policy regarding approval of transactions between us and a related person, our board of directors, as matter of appropriate corporate governance, reviews and approves all such transactions to the extent required by applicable rules and regulations. Generally, management would present any related person transactions proposed to be entered into by us to the board of directors for approval. The board may approve the transaction if it is deemed to be in the best interests of our stockholders and our company.
ITEM 11. EXECUTIVE COMPENSATION.
Executive Compensation Overview
Our compensation committee sets base salaries and bonuses and grants equity incentive awards to our executive officers. In setting base salaries and bonuses and granting equity incentive awards, our compensation committee considers compensation for comparable positions in the market, the historical compensation levels of our executives, individual and corporate performance as compared to our expectations and objectives, our desire to motivate our employees to achieve short- and long-term results that are in the best interests of our stockholders, and a long-term commitment to our company. The compensation committee also consults with the board of directors from time to time regarding compensation for our executive officers.
In 2015, our compensation committee engaged J. Thelander Consulting as its independent compensation consultant to review our executive compensation and director compensation and assess such compensation relative to comparable companies. J. Thelander Consulting worked with our compensation committee to determine a peer group of comparable companies, and review comparable compensatory arrangements for our executive officers and our board of directors and the committees thereof. Following consultation with J. Thelander Consulting, our compensation committee made certain adjustments to the compensatory arrangements of executive officers and the board of directors in 2015. We did not re-engage J. Thelander Consulting during 2016.
| 51 |
The material terms of the elements of our executive compensation program for 2016 are described below.
Base Salary
Base salaries are used to recognize the experience, skills, knowledge and responsibilities required of all our employees, including our named executive officers. None of our named executive officers is currently party to an employment agreement or other agreement or arrangement that provides for automatic or scheduled increases in base salary.
Our compensation committee determined not to grant any salary increases to executive officers during 2016.
Performance Bonus
Each named executive officer is eligible for an annual performance cash bonus based upon the achievement of certain corporate and individual goals and objectives approved by our board or our compensation committee. Cash bonuses are based on the executive officer’s base salary.
All final bonus payments to our named executive officers are determined by our compensation committee, which retains full discretion to adjust individual target bonus awards. The actual bonuses, if any, awarded in a given year may vary from target, depending on individual performance and the achievement of corporate objectives and may also vary based on other factors at the discretion of our compensation committee.
Pursuant to their July 2014 Amended and Restated Employment Agreements, each of Dr. Tinkelenberg and Mr. Rydzewski was entitled to participate in bonus plans approved by our board or compensation committee, with a target incentive bonus percentage equal to 40% of their then-current annual base salary. Neither Dr. Tinkelenberg nor Mr. Rydzewski received a bonus in 2016.
In September 2016, our Board awarded Mr. Sarney a discretionary cash retention award in the amount of $50,000.
Our compensation committee did not establish formal corporate goals and objectives for the calendar year 2016.
Equity Incentives
Our equity incentive program is the primary vehicle for offering long-term incentives to our executives, although the compensation committee has the authority to award other forms of equity-based compensation under our stock incentive plan. We believe that grants of equity incentive awards provide our executives with a strong link to our long-term performance and create an ownership culture that helps align the interests of our executives and stockholders. In addition, the vesting feature of our equity incentive grants furthers our goal of executive retention because this feature provides an incentive to our executives to remain in our employ during the vesting period.
In March 2016, our compensation committee awarded each of Dr. Nguyen and Mr. Sarney options to purchase 275,000 shares of our common stock, respectively, at an exercise price of $0.20 per share. Fifty percent (50%) of the shares underlying these options vested immediately upon grant and the remaining fifty percent (50%) vested on June 30, 2016.
Summary Compensation Table
The following table sets forth information concerning the total compensation paid or accrued by our company and Enumeral during the last two fiscal years indicated to (i) all individuals that served as our or Enumeral’s principal executive officer or acted in a similar capacity for us or Enumeral at any time during the most recent fiscal year indicated; (ii) the two most highly compensated executive officers who were serving as executive officers for us or Enumeral at the end of the most recent fiscal year indicated; and (iii) up to two additional individuals for whom disclosure would have been provided pursuant to clause (ii) above but for the fact that the individual was not serving as an executive officer for us or Enumeral at the end of the most recent fiscal year indicated.
| 52 |
| Name & Principal Position | Fiscal Year Ended December 31, | Salary ($) | Bonus ($) | Option Awards ($)(1) | All Other Compensation ($) | Total ($) | ||||||||||||||||
| Wael Fayad (2) | ||||||||||||||||||||||
| Chairman, President and Chief Executive Officer | 2016 | 91,250 | — | 400,686 | (3) | — | 491,936 | |||||||||||||||
| Kevin G. Sarney | 2016 | 260,000 | 50,000 | (4) | 44,863 | (5) | — | 354,863 | ||||||||||||||
| Vice President of Finance, Chief Accounting Officer and Treasurer | 2015 | 237,731 | 21,000 | (6) | — | — | 258,731 | |||||||||||||||
| John J. Rydzewski (7) | 2016 | 189,976 | — | — | 16,659 | (8) | 206,635 | |||||||||||||||
| Former Executive Chairman | 2015 | 262,500 | 105,000 | (9) | 216,235 | (10) | — | 583,735 | ||||||||||||||
| Arthur H. Tinkelenberg, Ph.D. (11) | 2016 | 150,615 | — | — | 130,972 | (12) | 281,587 | |||||||||||||||
| Former President and Chief Executive Officer | 2015 | 262,500 | 105,000 | (9) | 264,287 | (13) | — | 631,787 | ||||||||||||||
| Anhco Nguyen, Ph.D. (14) | 2016 | 173,990 | — | 44,863 | (5) | 10,154 | (15) | 229,007 | ||||||||||||||
| Former Vice President of Research and Development | 2015 | 241,597 | 25,000 | (16) | 156,860 | (17) | — | 423,457 | ||||||||||||||
| (1) | These amounts represent the aggregate grant date fair value of stock option awards granted in fiscal year 2016 or 2015, as applicable. These amounts are computed in accordance with FASB ASC Topic 718. A description of the assumptions used for purposes of determining grant date fair value is set forth in the notes to our consolidated financial statements included in this Annual Report on Form 10-K for the year ended December 31, 2016. The named executive officers will only realize compensation to the extent the trading price of our common stock is greater than the exercise price of such stock options. |
| (2) | Mr. Fayad’s employment with our company commenced on September 21, 2016. |
| (3) | This amount reflects a September 21, 2016 grant of 2,600,000 options to purchase shares of our common stock, of which 850,000 stock options were granted under the 2014 Plan and 1,750,000 options were granted outside of the 2014 Plan. |
| (4) | This amount reflects a one-time cash bonus paid to Mr. Sarney in September 2016. |
| (5) | This amount reflects a March 24, 2016 grant of 275,000 options to purchase shares of our common stock. |
| (6) | This amount reflects a bonus paid in 2015 for service in calendar year 2014, representing a pro-rated portion of Mr. Sarney’s target bonus for that year. |
| (7) | Mr. Rydzewski resigned as our Executive Chairman and as a member of our board on September 21, 2016. |
| (8) | This amount reflects the accrued vacation paid to Mr. Rydzewski as part of his resignation. |
| (9) | These amounts reflect bonuses paid in 2015 for service in calendar year 2014, representing the target bonus of 40% of annual salary. |
| (10) | This amount reflects a September 28, 2015 grant of 720,000 options to purchase shares of our common stock. |
| (11) | Dr. Tinkelenberg was terminated as our President and Chief Executive Officer on July 28, 2016, and subsequently resigned as a director on September 19, 2016. |
| (12) | This amount reflects certain benefits that Dr. Tinkelenberg has received pursuant to the terms of his separation letter agreement, dated as of August 4, 2016 (the “Tinkelenberg Agreement”), including (a) $111,885 in salary continuation during his severance period (as defined in the Tinkelenberg Agreement), (b) a lump sum amount of $10,000, less applicable taxes, to be used for the purchase of life insurance, disability insurance, or other health coverage, or any other purpose Dr. Tinkelenberg may choose and (c) $9,087 in accrued vacation paid to Mr. Tinkelenberg as part of his termination. |
| (13) | This amount reflects a September 28, 2015 grant of 880,000 options to purchase shares of our common stock. |
| 53 |
| (14) | Dr. Nguyen resigned as our Vice President of Research and Development on August 17, 2016. |
| (15) | This amount reflects the accrued vacation paid to Dr. Nguyen as part of his resignation. |
| (16) | This amount reflects a bonus paid in 2015 for service in calendar year 2014, representing over-achievement of Dr. Nguyen’s then-target bonus. |
| (17) | This amount reflects a September 22, 2015 grant of 400,000 options to purchase shares of our common stock. |
We have no plans in place and have never maintained any plans that provide for the payment of retirement benefits or benefits that will be paid primarily following retirement including, but not limited to, tax qualified deferred benefit plans, supplemental executive retirement plans, tax-qualified deferred contribution plans and nonqualified deferred contribution plans.
Except as indicated below, we have no contracts, agreements, plans or arrangements, whether written or unwritten, that provide for payments to the named executive officers listed above.
Outstanding Equity Awards at Fiscal Year-End
As of December 31, 2016, we have one compensation plan, the Enumeral Biomedical Holdings, Inc. 2014 Equity Incentive Plan, or the 2014 Plan. The 2014 Plan was approved by our stockholders on July 31, 2014 and provides for awards of stock options, stock appreciation rights, restricted stock, restricted stock units, performance shares, and performance units. In addition, we have granted equity awards outside of the 2014 Plan. The table below provides information with respect to the outstanding equity awards of our named executive officers as of December 31, 2016. Dr. Nguyen resigned from our company on August 17, 2016, and in accordance with the terms of the 2014 Plan his unvested equity awards expired on his resignation date, and his vested equity awards expired 90 days thereafter. None of Dr. Nguyen’s equity awards remained outstanding as of December 31, 2016.
| Option Awards | Stock Awards | |||||||||||||||||||||||||
| Name | Number of Securities Underlying Unexercised Options Exercisable (#) | Number of Securities Underlying Unexercised Options Unexercisable (#) | Number of Securities Underlying Unexercised Unearned Options (#) | Option Exercise Price ($) | Option Expiration Date | Number
of Shares or Units of Stock That Have Not Vested (#) | Market Value of Shares or Units of Stock That Have Not Vested ($) | |||||||||||||||||||
| Wael Fayad | 100,000 | (1) | 2,500,000 | (1) | 0.19 | 09/20/2026 | — | — | ||||||||||||||||||
| Kevin G. Sarney | 275,000 | (2) | — | — | 0.20 | 03/23/2026 | — | — | ||||||||||||||||||
| 175,000 | (3) | 125,000 | (3) | 50,000 | (4) | 1.00 | 07/30/2024 | — | — | |||||||||||||||||
| John J. Rydzewski | 720,000 | (5) | — | — | 0.36 | 09/28/2021 | — | — | ||||||||||||||||||
| 300,000 | (5) | — | — | 1.00 | 09/28/2021 | — | — | |||||||||||||||||||
| Arthur H. Tinkelenberg, Ph.D. | 357,505 | (6) | 302,495 | (6) | 220,000 | (6)(7) | 0.36 | 07/28/2022 | — | — | ||||||||||||||||
| 62,506 | (6) | 37,494 | (6) | 200,000 | (6)(8) | 1.00 | 07/28/2022 | — | — | |||||||||||||||||
| (1) | Consists of stock options granted on September 21, 2016 for (a) 850,000 shares of our common stock granted under the 2014 Plan, and (b) 1,750,000 shares of common stock granted outside of the 2014 Plan. 100,000 of the shares granted under the 2014 Plan vested in full immediately upon grant, and the remainder of the options granted both under and outside of the 2014 Plan vest upon satisfactory achievement of performance-based objectives. These objectives have not yet been met. |
| (2) | Consists of stock options granted on March 24, 2016 for 275,000 shares of our common stock, of which 137,500 shares vested immediately upon grant and 137,500 shares vested on June 30, 2016. |
| 54 |
| (3) | Consists of stock options granted on July 31, 2014 for 300,000 shares of our common stock, of which 75,000 shares vested on July 31, 2015, and the remaining 225,000 shares vest monthly over three years in 36 monthly installments beginning on August 31, 2015. |
| (4) | Consists of stock options granted on July 31, 2014 for 50,000 shares of our common stock which vest upon satisfactory achievement of performance-based objectives. These objectives have not yet been met. |
| (5) | Pursuant to the terms of Mr. Rydzewski’s separation letter agreement (the “Rydzewski Agreement”), dated as of September 21, 2016, all of Mr. Rydzewski’s options to purchase shares of our common stock and restricted stock grants that were unvested as of Mr. Rydzewski’s separation date vested in full as of the effective date of the Rydzewski Agreement. In addition, the Rydzewski Agreement provides that the period for exercising all such options to purchase shares of our common stock is extended to the date that is five years following the effective date of the Rydzewski Agreement. |
| (6) | Pursuant to the terms of Dr. Tinkelenberg’s separation letter agreement (the “Tinkelenberg Agreement”), dated as of August 4, 2016, all of Dr. Tinkelenberg’s options to purchase shares of our common stock and restricted stock grants that were unvested as of Dr. Tinkelenberg’s separation date shall continue to vest during Dr. Tinkelenberg’s 12-month severance period pursuant to the vesting schedules set forth in such option agreements and restricted stock agreements, as applicable. Absent a breach of the Tinkelenberg Agreement by Dr. Tinkelenberg during the severance period, all remaining unvested options and shares of restricted stock shall fully vest and become exercisable on the expiration of the severance period, and the period for exercising all such options to purchase shares of our common stock shall be extended to the date that is five years following the expiration of the severance period. |
| (7) | Consists of stock options granted on September 28, 2015 that vest and become exercisable upon satisfactory achievement of performance-based objectives. Certain objectives were met in 2016, and certain other objectives have not yet been met. |
| (8) | Consists of stock options granted on July 31, 2014 that vest and become exercisable upon satisfactory achievement of performance-based objectives. These objectives have not yet been met. |
The following table sets forth certain information with respect to grants of stock options to the Named Executive Officers during fiscal year 2016:
| GRANTS OF PLAN BASED AWARDS TABLE | ||||||||||||||
| Name | Grant Date | All Other Option Awards: Number of Securities Underlying Options (#) | Exercise Price of Option Awards ($/Share) | Grant Date Fair Value ($)(1) | ||||||||||
| Wael Fayad | 09/21/2016 | 850,000 | (2) | 0.19 | 130,994 | |||||||||
| Kevin G. Sarney | 03/24/2016 | 275,000 | 0.20 | 44,863 | ||||||||||
| Anhco Nguyen, Ph.D.(3) | 03/24/2016 | 275,000 | 0.20 | 44,863 | ||||||||||
| (1) | These amounts represent the aggregate grant date fair value of stock option awards granted in fiscal year 2016 under our 2014 Equity Incentive Plan. These amounts are computed in accordance with FASB ASC Topic 718. A description of the assumptions used for purposes of determining grant date fair value is set forth in the notes to our consolidated financial statements included in this Annual Report on Form 10-K for the year ended December 31, 2016. The named executive officers will only realize compensation to the extent the trading price of our common stock is greater than the exercise price of such stock options. |
| 55 |
| (2) | In addition to granting Mr. Fayad 850,000 stock options under the 2014 Plan, we also granted Mr. Fayad 1,750,000 stock options outside of the 2014 Plan. All grants were made on September 21, 2016, and all options have an exercise price of $0.19 per share. |
| (3) | Dr. Nguyen resigned as our Vice President of Research and Development on August 17, 2016. |
Employment Agreements
Wael Fayad
On September 21, 2016, we entered into an offer letter with Mr. Fayad (the “Fayad Agreement”), which sets forth the terms pursuant to which Mr. Fayad serves as our Chairman of the Board, President and Chief Executive Officer. The Fayad Agreement provides that Mr. Fayad receives a base salary at the rate of $325,000 per annum. Mr. Fayad is also eligible to earn a target bonus of up to 50% of the base salary, payable in cash, based upon achievement of corporate objectives, individual objectives, and our financial condition, all as determined and at the discretion of the independent members of the Board or the Board’s Compensation Committee.
Pursuant to the terms of the Fayad Agreement, the Board approved the following grants to Mr. Fayad:
| (I) | options under the 2014 Plan to purchase 100,000 shares of our Common Stock, at an exercise price of $0.19 per share, fully vested and exercisable upon grant, to be treated as incentive stock options to the maximum extent permitted by applicable federal tax laws; |
| (II) | options under the 2014 Plan to purchase 750,000 shares of our Common Stock at an exercise price of $0.19 per share, to be treated as incentive stock options to the maximum extent permitted by applicable federal tax laws, and which shall vest and become exercisable at such time as we enter into a collaboration and/or licensing agreement with a corporate partner on terms acceptable to the Board that provides we will receive at least a specified amount in upfront fees upon signing (the “Tranche A Options”); and |
| (III) | non-qualified stock options granted outside of the 2014 Plan to purchase an aggregate of 1,750,000 shares of our Common Stock at an exercise price of $0.19 per share, which such options shall vest and become exercisable on the following schedule: |
| (a) | 750,000 shares of Common Stock underlying the options shall vest and become exercisable at such time as we enter into a second (i.e., in addition to the transaction described above) collaboration and/or licensing agreement with a corporate partner on terms acceptable to the Board that provides that we will receive at least a specified amount in upfront fees upon signing (the “Tranche B Options”); provided that, in the event that we enter into collaboration and/or licensing agreement(s) with one or more corporate partners on terms acceptable to the Board that collectively provide we will receive at least an aggregate of a specified amount in upfront fees upon signing, all of the Tranche A Options and Tranche B Options shall vest and become exercisable; and |
| (b) | the remaining 1,000,000 shares of Common Stock underlying the options shall vest and become exercisable upon the closing of an equity financing transaction on terms acceptable to the Board with gross proceeds to us of at least a specified amount, which may include proceeds from the exercise of some or all of the warrants issued in connection with our July 2014 private placement offering (the “Tranche C Options”); provided that, in the event we raise at least a certain specified amount in one or more transactions (including proceeds from the exercise of some or all of the warrants issued in connection with our July 2014 private placement offering), with at least a certain specified portion raised through non-dilutive collaboration and/or licensing agreements, all of the Tranche A Options, Tranche B Options, and Tranche C Options shall vest and become exercisable. |
As of the date of this filing, none of the Tranche A Options, Tranche B Options, or Tranche C Options have vested.
| 56 |
The Fayad Agreement also includes an Obligations Agreement which contains, among other provisions, non-competition and non-solicitation provisions applicable to Mr. Fayad in certain circumstances specified therein.
Kevin G. Sarney
Mr. Sarney was hired as our Vice President of Finance and Chief Accounting Officer pursuant to an offer letter dated July 25, 2014. On March 24, 2016, we entered into a new employment letter with Mr. Sarney (the “Sarney Agreement”), which stated our desire to continue to employ Mr. Sarney in his position and confirmed the terms of his employment including, among other things, certain benefits in the event his employment with our company was terminated. The Sarney Agreement provides that (i) Mr. Sarney will continue to serve as our Vice President of Finance, Chief Accounting Officer and Treasurer, reporting to our President and Chief Executive Officer, (ii) receive a base salary at the rate of $260,000 per annum, and (iii) be eligible to earn a target bonus up to 25% of his base salary, based on the achievement of corporate objectives as determined by our board or our compensation committee.
Pursuant to Mr. Sarney’s original July 2014 offer letter, Mr. Sarney received an incentive stock option to purchase 350,000 shares of our common stock, of which 50,000 shares vest upon the satisfactory achievement of performance-based objectives, with the remainder of shares vesting on a time-based schedule.
John J. Rydzewski
On October 1, 2011, we entered into an employment agreement with Mr. Rydzewski (the “2011 Agreement”), pursuant to which Mr. Rydzewski was employed as Enumeral’s Executive Chairman. On July 21, 2014, Enumeral entered into an Amended and Restated Employment Agreement with Mr. Rydzewski (the “Rydzewski Amended Agreement”), which we assumed on July 31, 2014 and which amended and restated the 2011 Agreement. Pursuant to the terms of the Rydzewski Amended Agreement, Mr. Rydzewski (i) received a base salary of $262,500, (ii) was entitled to participate in bonus plans which are approved by the board for Enumeral employees generally, with a target incentive bonus percentage equal to 40% of his then-current annual salary, (iii) received an incentive stock option award to purchase 100,000 shares of Enumeral’s or, if applicable, our common stock with an exercise price equal to the fair market value of Enumeral’s common stock as determined by Enumeral’s board of directors and vesting in 48 equal monthly installments over 4 years beginning on July 31, 2014, (iv) received an incentive stock option award to purchase 200,000 shares of Enumeral’s or, if applicable, our common stock with an exercise price equal to the fair market value of Enumeral’s common stock as determined by Enumeral’s board of directors and vesting subject to performance-based criteria set forth in the employment agreement, and (v) received a warrant to purchase 47,058 shares of Enumeral’s Series B Preferred Stock subject to vesting conditions set forth in the employment agreement and exercisable at $2.125 per share (which was subsequently converted in connection with the Merger into a warrant to purchase 137,762 shares of our common stock at an exercise price of $0.726 per share).
Pursuant to the October 1, 2011 employment agreement, Mr. Rydzewski received (a) a restricted stock grant of 100,000 shares and (b) a stock option award to purchase 50,000 shares of Enumeral’s common stock, in each case subject to the vesting conditions set forth in the employment agreement and with an exercise price equal to the fair market value of Enumeral’s common stock as determined by Enumeral’s board of directors. Mr. Rydzewski exercised his option to purchase 50,000 options on July 25, 2012. Additionally, on January 2, 2013, Mr. Rydzewski received a restricted stock grant of 324,000 shares of Enumeral common stock, of which 13,500 shares were immediately vested upon grant and the balance vests in 46 equal monthly installments beginning on January 26, 2013.
Contemporaneous with the closing of the Merger and PPO, Mr. Rydzewski’s warrants and options to purchase shares of Enumeral common stock, as well as restricted grants of Enumeral common stock, were converted into warrants and options to purchase shares of our common stock, as well as restricted grants of our common stock, respectively.
In connection with Mr. Rydzewski’s resignation as Executive Chairman of our company, we entered into a separation letter agreement with Mr. Rydzewski (the “Rydzewski Separation Agreement”), dated September 21, 2016 (the “Rydzewski Separation Date”).
| 57 |
Pursuant to the terms of the Rydzewski Separation Agreement, we will continue to pay one hundred percent (100%) of the cost for Mr. Rydzewski’s continuing medical, dental, and prescription benefits to which he was entitled during his employment with us until the earlier of (i) eighteen (18) months following the Rydzewski Separation Date pursuant to Section 4980B of the Internal Revenue Code of 1986, as amended (“COBRA”), or (ii) such time as Mr. Rydzewski becomes eligible for similar benefits from another employer.
In addition, pursuant to the terms of the Rydzewski Separation Agreement, all options to purchase shares of our Common Stock and our restricted stock grants held by Mr. Rydzewski or in a trust established by Mr. Rydzewski for the benefit of his spouse, children or heirs that were unvested as of the Separation Date shall vest in full immediately upon the effective date of the Rydzewski Separation Agreement (as defined in paragraph 15(e) thereof). In addition, the period for exercising all such options was extended to the date that is five (5) years following the effective date of the Rydzewski Separation Agreement.
The Rydzewski Separation Agreement also contains non-solicitation provisions applicable to Mr. Rydzewski during the twelve (12) month period following the Separation Date, confidentiality and non-disparagement provisions, and a full release of claims he may have against us, subject to certain exceptions.
Arthur H. Tinkelenberg, Ph.D.
On July 1, 2010, Enumeral entered into an employment agreement with Dr. Tinkelenberg (the “2010 Agreement”), pursuant to which Dr. Tinkelenberg was employed as Enumeral’s President and Chief Executive Officer. On July 21, 2014, Enumeral entered into an Amended and Restated Employment Agreement with Dr. Tinkelenberg (the “Tinkelenberg Amended Agreement”), which we assumed on July 31, 2014 and which amended and restated the 2010 Agreement. Pursuant to the terms of the Tinkelenberg Amended Agreement, Dr. Tinkelenberg (i) received a base salary of $262,500, (ii) was entitled to participate in bonus plans which are approved by the Board for Enumeral employees generally, (iii) received an incentive stock option award to purchase 100,000 shares of Enumeral’s or, if applicable, our common stock with an exercise price equal to the fair market value of Enumeral’s common stock as determined by Enumeral’s board of directors and vesting in 48 equal monthly installments over 4 years beginning on July 31, 2014, (iv) received an incentive stock option award to purchase 200,000 shares of Enumeral’s or, if applicable, our common stock with an exercise price equal to the fair market value of Enumeral’s common stock as determined by Enumeral’s board of directors and vesting subject to performance-based criteria set forth in the employment agreement, and (v) received a warrant to purchase 58,823 shares of Enumeral’s Series B Preferred Stock subject to vesting conditions set forth in the employment agreement and exercisable at $2.125 per share (which was subsequently converted in connection with the Merger into a warrant to purchase 172,204 shares of our common stock at an exercise price of $0.726 per share).
Pursuant to the July 1, 2010 employment agreement, Dr. Tinkelenberg received (a) a restricted stock grant of 393,750 shares and (b) a stock option award to purchase 51,576 shares of Enumeral’s common stock, in each case subject to the vesting conditions set forth in the employment agreement (and which were fully vested prior to the closing of the Merger) and with an exercise price equal to the fair market value of Enumeral’s common stock as determined by Enumeral’s board of directors. Dr. Tinkelenberg exercised his 51,576 options on July 27, 2012. Additionally, on January 2, 2013, Dr. Tinkelenberg received a restricted stock grant of 324,000 shares of Enumeral common stock, of which 13,500 shares were immediately vested upon grant and the balance vests in 46 equal monthly installments beginning on January 26, 2013.
Contemporaneous with the closing of the Merger and PPO, Dr. Tinkelenberg’s warrants and options to purchase shares of Enumeral common stock, as well as restricted grants of Enumeral common stock, were converted into warrants and options to purchase shares of our common stock, as well as restricted grants of our common stock, respectively.
In connection with Dr. Tinkelenberg’s termination of employment with us on July 28, 2016 (the “Tinkelenberg Separation Date”), we entered into a separation letter agreement with Dr. Tinkelenberg, dated August 4, 2016 (the “Tinkelenberg Separation Agreement”).
Pursuant to the terms of the Tinkelenberg Separation Agreement, we will pay Dr. Tinkelenberg his annual base salary of $262,500, less applicable tax withholdings (the “Tinkelenberg Severance Payment”), for a period of twelve months from July 28, 2016 (the “Tinkelenberg Severance Period”). In addition, we paid Dr. Tinkelenberg a lump sum amount of $10,000, less applicable tax withholdings, to be used for the purchase of health coverage, life insurance, disability insurance, or any other purpose Dr. Tinkelenberg chooses. In the event a Change of Control of our company (as such term is defined in the Tinkelenberg Amended Agreement) occurs during the Tinkelenberg Severance Period, the remaining then-unpaid portion of the Tinkelenberg Severance Payment shall be paid in a lump sum in lieu of any further salary continuation payments.
| 58 |
The Tinkelenberg Separation Agreement also provides that all of Dr. Tinkelenberg’s options to purchase shares of our common stock and our restricted stock grants that were unvested as of the Tinkelenberg Separation Date shall continue to vest during the Tinkelenberg Severance Period pursuant to the vesting schedules set forth in such option agreements and restricted stock agreements, as applicable. In the event that Dr. Tinkelenberg breaches the terms of the Tinkelenberg Separation Agreement during the Tinkelenberg Severance Period, all unvested options and restricted stock awards as of the date of notice by us of such breach (subject to any cure periods) shall be immediately cancelled and forfeited. In the absence of such breach, all remaining unvested options and shares of restricted stock shall fully vest and become exercisable on the expiration of the Tinkelenberg Severance Period, and the period for exercising all such options to purchase our common stock shall be extended to the date that is five (5) years following the expiration of the Tinkelenberg Severance Period. In addition, if there occurs a Change of Control (as such term is defined in the Tinkelenberg Amended Agreement) during the Tinkelenberg Severance Period, then all of Dr. Tinkelenberg’s unvested shares of restricted stock and options to purchase our common stock shall immediately accelerate and become fully vested and exercisable.
The Tinkelenberg Separation Agreement also contains non-competition and non-solicitation provisions applicable to Dr. Tinkelenberg during the Tinkelenberg Severance Period, confidentiality and non-disparagement provisions, and a full release of claims he may have against us, subject to certain exceptions.
Anhco Nguyen, Ph.D.
Dr. Nguyen was initially hired as our Research Director pursuant to an offer letter dated December 17, 2011. Dr. Nguyen was subsequently promoted to increasing roles of responsibility within our company, eventually serving as our Vice President of Research and Development. On March 24, 2016, we entered into a new employment letter with Dr. Nguyen (the “Nguyen Agreement”), which stated our desire to continue to employ Dr. Nguyen in his position and confirmed the terms of his employment including, among other things, certain benefits in the event his employment with our company was terminated. The Nguyen Agreement provided that (i) Dr. Nguyen would continue to serve as our Vice President of Research and Development, reporting to our President and Chief Executive Officer, (ii) receive a base salary at the rate of $275,000 per annum, and (iii) would have been eligible to earn a target bonus up to 30% of his base salary, based on the achievement of corporate objectives as determined by our board or our compensation committee.
Pursuant to Dr. Nguyen’s original December 2011 offer letter, Dr. Nguyen received an incentive stock option to purchase 32,500 shares of Enumeral common stock, which were converted into 35,819 shares of our common stock in connection with the Merger. With respect to this option grant, 9,000 of the original Enumeral shares granted were to vest upon the satisfactory achievement of performance-based objectives, with the remainder of shares vesting on a time-based schedule.
As Dr. Nguyen progressed in our company, he was awarded additional equity grants. Contemporaneous with the closing of the Merger and PPO, Dr. Nguyen’s options to purchase shares of Enumeral common stock were converted into options to purchase shares of our common stock.
Dr. Nguyen resigned from his position as Vice President of Research and Development, effective as of August 17, 2016. In accordance with the terms of the 2014 Plan, all of Dr. Nguyen’s equity grants expired 90 days after his effective resignation date.
Potential Payments upon Termination or Change in Control
Employment Letter with Mr. Fayad
The Fayad Agreement provides that if, while Mr. Fayad is employed as Chairman, President and Chief Executive Officer, we achieve the milestones listed in both (II) and (III)(a) in the above description of Mr. Fayad’s equity grants pursuant to the Fayad Agreement, or, if earlier, upon the achievement of the milestone listed in (III)(b) in the above description, and following such funding achievement Mr. Fayad’s employment with us is subsequently involuntarily terminated without Cause (as defined in the Fayad Agreement), then we will pay Mr. Fayad his base salary for a period of twelve (12) months following the date of the notice of termination (the “Severance Benefits”), provided that Mr. Fayad continues to comply with and not breach the terms set forth in the Fayad Agreement and enters into a termination and release agreement in a form acceptable to us.
| 59 |
Pursuant to the terms of the Fayad Agreement, in the event a Change of Control (as defined in the Fayad Agreement) occurs within the first six (6) months following the effective date of the Fayad Agreement, and Mr. Fayad’s employment with us is terminated in connection with such Change of Control (and provided that Mr. Fayad is not at that time entitled to the Severance Benefits set forth above), then we will pay Mr. Fayad his base salary for a period of six (6) months following the date of the notice of termination, provided that Mr. Fayad continues to comply with and not breach the terms set forth in the Fayad Agreement and enters into a termination and release agreement in a form acceptable to us.
Employment Agreements of Mr. Rydzewski and Dr. Tinkelenberg
If Mr. Rydzewski’s or Dr. Tinkelenberg’s employment were terminated for cause, death, disability or if they resign for any reason (other than resignation for Good Reason, as such term is defined below), the Rydzewski Amended Agreement and the Tinkelenberg Amended Agreement provided that such executive would have received the following:
| · | any accrued but unpaid salary through the date of termination plus any accrued vacation; |
| · | any earned and declared but unpaid bonus for the most recently completed year; |
| · | reimbursement of any unreimbursed expenses; and |
| · | benefits in accordance with the terms of our applicable plans and programs. |
In addition, all of the executive’s unvested restricted stock or options would have immediately been forfeited and be cancelled.
If Mr. Rydzewski’s or Dr. Tinkelenberg’s employment were terminated by us other than for Cause or by Mr. Rydzewski or Dr. Tinkelenberg for Good Reason where a Change in Control has not occurred (as such terms are defined below), they would have been entitled to the following:
| · | base salary for the greater of twelve months or the remaining time until two years from the date of the closing of the Merger and PPO (the “Severance Period”); |
| · | all earned and declared but unpaid bonuses which are due and payable, including a pro rata portion of the current year’s potential bonus, calculated based on the then-current target bonus percentage (but no less than 40%), payable in lump sum, based upon achievement of then-current target milestones through the termination date of employment as determined in good faith by our compensation committee or board of directors, as applicable; |
| · | continued coverage for the Severance Period of all our group insurance (including life, health, accident and disability insurance) and, to the extent permitted by applicable law, all other employee benefit plans, programs or arrangements in which the executive was participating at any time during the six month period preceding the termination of employment, provided that we will no longer be obligated to fund the cost of premiums for such plans and benefits in the event that the executive becomes eligible for similar insurance or other employee benefits from a new employer during the Severance Period; |
| 60 |
| · | continued vesting of any non-vested stock options and restricted stock granted pursuant to the employment agreement for the Severance Period pursuant to the vesting schedule set forth therein, as well as extension of the period to exercise vested stock options until five years following the expiration of the Severance Period, provided that if the executive breaches the terms of his employment agreement, all of the executive’s unvested options and shares of restricted stock will be immediately forfeited; and |
| · | payment for all accrued but unused vacation on the termination date of employment. |
In addition, if a Change in Control occurred during the Severance Period, all non-vested stock options and shares of restricted stock would have immediately accelerated and become exercisable or non-forfeitable, as the case may be.
If Mr. Rydzewski’s or Dr. Tinkelenberg’s employment was terminated, or Mr. Rydzewski or Dr. Tinkelenberg resigned for Good Reason where a Change in Control has occurred, they would have received:
| · | an amount equal to one year of base salary, payable immediately in one lump sum; |
| · | all bonuses which are due and payable, including the full amount of the current year’s target incentive bonus as if the then-current target milestones were achieved for the applicable year; |
| · | continued coverage for the Severance Period of all our group insurance (including life, health, accident and disability insurance) and, to the extent permitted by applicable law, all other employee benefit plans, programs or arrangements in which the executive was participating at any time during the six month period preceding the termination of employment; |
| · | all non-vested stock options or restricted stock shall vest immediately and become exercisable or non-forfeitable and shall be exchanged for cash or freely tradable stock of the acquiring company, provided that in the event it is not possible for such stock options or restricted stock to be exchanged for cash or freely tradable stock of the acquiring company, the period to exercise vested stock options shall be extended until five years following the date such shares vested; and |
| · | payment for all accrued but unused vacation on the termination date of employment. |
“Cause” was defined in the Rydzewski Amended Agreement and the Tinkelenberg Amended Agreement as:
| · | the executive’s conviction by a court of competent jurisdiction of any felony involving dishonesty, breach of trust or misappropriation or entry of a plea of nolo contendere thereto; |
| · | the commission of an act of fraud upon, breaching the duty of loyalty to, us or any of our subsidiaries; |
| · | a conviction for willful violation of any law, rule or regulation governing operation of our company or any of its subsidiaries that is punishable by six months or more imprisonment; |
| · | the substantial and continuing failure or refusal of the executive, after seven days’ written notice thereof, to perform his or her job duties and responsibilities which failure or refusal is committed in bad faith (other than failure or refusal resulting from incapacity due to physical disability or mental illness); |
| · | the executive’s breach of his employment agreement that continues for more than seven days after written notice has been given to the executive; or |
| · | the deliberate and willful disregard for our rules or policies that results in a material and substantial loss, damage or injury to our company. |
“Good Reason” was defined in the Rydzewski Amended Agreement and the Tinkelenberg Amended Agreement as:
| · | a reduction in the executive’s then-current base salary or target bonus percentage; |
| 61 |
| · | any failure to offer to the executive at least the same level of benefits offered to similarly situated employees; |
| · | a significant diminution in the executive’s managerial authority, duties and responsibilities following a Change in Control of our company; |
| · | the relocation of the executive’s primary business location to a location outside of the New York metropolitan area; |
| · | failure to pay to the executive any portion of his current base salary, bonus or benefits within 20 days of when such compensation is due, based upon the payment terms currently in effect, unless such payment is prohibited by law, regulation or rule; or |
| · | our failure to obtain a reasonably satisfactory agreement from any successor to our company to assume and agree to perform the executive’s employment agreement. |
“Change in Control” was defined in the Rydzewski Amended Agreement and the Tinkelenberg Amended Agreement as:
| · | the merger or consolidation of our company with another entity, where, immediately after the transaction: |
| · | our stockholders immediately prior to the merger or consolidation beneficially own, directly or indirectly, less than 50% of the voting shares of the surviving entity in the consolidation or merger (or of its ultimate parent corporation, if any); or |
| · | persons who constitute our board of directors prior to the transaction cease to constitute at least a majority of the board of directors of the surviving entity in the consolidation or merger (or of its ultimate parent corporation, if any); |
| · | sale, lease or other transfer of all or substantially all of our assets or the exclusive license of all or substantially all of our patents, provisional patent applications and patent applications for substantially all uses to a third party; |
| · | any person (other than our company, any of its subsidiaries, or any trustee, fiduciary or other person or entity holding securities under any employee benefit plan), together with all affiliates and associates of such person, becomes the beneficial owner of our securities representing 50% or more of the combined voting power of our then outstanding securities having the right to vote in an election of our board of directors. |
Tinkelenberg Separation Agreement
As noted above, on August 4, 2016 we entered into the Tinkelenberg Separation Agreement with Dr. Tinkelenberg in connection with his termination as our President and Chief Executive Officer. Please see the description of the terms of the Tinkelenberg Separation Agreement set forth above for details of the benefits that Dr. Tinkelenberg received in connection with the termination of his employment (including benefits that Dr. Tinkelenberg may receive in the event that a Change of Control (as such term is defined in the Tinkelenberg Amended Agreement) occurs during the Tinkelenberg Severance Period.
Employment Letters of Mr. Sarney and Dr. Nguyen
Pursuant to the terms of the Sarney Agreement and the Nguyen Agreement, in the event that Mr. Sarney’s or Dr. Nguyen’s employment with our company were terminated without Cause (as defined below) or if Mr. Sarney or Dr. Nguyen resigned for Good Reason (as defined below), Mr. Sarney and Dr. Nguyen would have been entitled to the following severance benefits:
| 62 |
| · | The continued receipt of his base salary at the then-current rate for a period of six (6) months following the date of his termination of employment with our company, provided that he continued to comply with and not breach the terms of the separation and release agreement and, as applicable, any other agreement that he has previously entered into with us; and |
| · | The receipt of all earned and declared but unpaid bonuses which were due and payable, including without limitation a pro rata portion of the current year’s potential bonus calculated based on the executive’s then-current target bonus percentage, payable in a lump sum, based on the number of days in any given year that he was employed through the date of termination, and based upon achievement of then-current corporate objectives as determined in good faith by our President and Chief Executive Officer. |
The Sarney Agreement and the Nguyen Agreement also provided that upon a Change of Control (as defined in our 2014 Equity Incentive Plan, as amended), Mr. Sarney would be, and Dr. Nguyen would have been, entitled, in addition to the severance benefits set forth above, to immediate and full vesting of the shares underlying his equity awards which remained unvested as of the date of such Change in Control.
For purposes of the Sarney Agreement and the Nguyen Agreement, “Cause” means any of the following, as determined by our President and Chief Executive Officer, acting in good faith: (a) any failure of the executive to take or refrain from taking any corporate action consistent with the executive’s duties as specified in written directions of our President and Chief Executive Officer or our board of directors; (b) the executive’s willful engagement in illegal conduct or gross misconduct that is injurious to our company; (c) the conviction of the executive of, or the entry of a pleading of guilty or nolo contendere by the executive to, any crime involving moral turpitude or any felony; (d) fraud upon our company including, without limitation, falsification of company records or financial information; or (e) the executive’s breach of any of the non-compete, non-solicitation, and proprietary information provisions of the Sarney Agreement or the Nguyen Agreement, as applicable (including the terms of any obligations agreement entered into between the executive and our company).
In addition, for purposes of the Sarney Agreement and the Nguyen Agreement, “Good Reason” means the occurrence, without the executive’s prior written consent, of any of these events or circumstances:
| · | a material diminution in the executive’s rate of base salary; |
| · | a material diminution in the executive’s authority, duties, or responsibilities; or |
| · | a material breach by us of the Sarney Agreement or the Nguyen Agreement, as applicable; |
provided that any of these events described above shall constitute Good Reason only if we fail to cure such event within thirty (30) days after receipt from the executive of written notice of the event which constitutes Good Reason; and provided further that Good Reason shall cease to exist for an event on the sixtieth (60th) day following its occurrence, unless the executive has given us written notice thereof prior to such date.
For purposes of the Sarney Agreement and the Nguyen Agreement, “Change in Control” has the meaning ascribed to such term in the 2014 Plan. The 2014 Plan provides that a Change in Control shall be deemed to have occurred as of the first day that any one or more of the following conditions shall have been satisfied:
| · | the beneficial ownership of securities (as defined in Rule 13d-3 under the Exchange Act) representing more than thirty-three percent (33%) of the combined voting power of our company is acquired by any person (other than our company, any trustee or other fiduciary holding securities under an employee benefit plan of our company, or any corporation owned, directly or indirectly, by the stockholders of our company in substantially the same proportions as their ownership of stock of our company); or |
| · | the consummation of a definitive agreement to merge or consolidate our company with or into another corporation or to sell or otherwise dispose of all or substantially all of its assets, or adopt a plan of liquidation; or |
| 63 |
| · | during any period of three consecutive years, individuals who at the beginning of such period were members of our board cease for any reason to constitute at least a majority thereof (unless the election, or the nomination for election by our stockholders, of each new director was approved by a vote of at least a majority of the directors then still in office who were directors at the beginning of such period or whose election or nomination was previously so approved). |
The 2014 Plan also details additional triggers for a Change in Control applicable to equity awards subject to Section 409A of the Internal Revenue Code of 1986, as amended, including threshold changes of ownership, changes in effective control, and changes in ownership of substantial assets. The full definition of “Change in Control” set forth in the 2014 Plan is incorporated herein by reference.
Director Compensation
Non-employee directors’ compensation generally is determined and awarded by our board of directors, or the compensation committee thereof. Our board is responsible for, among other things, reviewing, evaluating and designing a director compensation package of a reasonable total value, typically based on comparisons with similar firms, and aligned with long-term interests of our stockholders, and reviewing director compensation levels and practices and considering, from time to time, changes in such compensation levels and practices. These matters also include making equity awards to non-employee directors from time to time under our equity-based plans. As part of these responsibilities, our board may request that our management provide the board with recommendations on non-employee director compensation and/or common director compensation practices, although the board retains its ultimate authority to take compensatory actions.
In September 2015, our compensation committee approved a revised director compensation program for non-employee directors, which we refer to as the 2015 Program. Under the 2015 Program, newly elected non-employee board members will continue to receive an option to purchase 20,000 shares of our common stock that will be fully vested upon their election to the board. Non-employee directors are also entitled to receive an annual grant of options to purchase common stock, as determined by the compensation committee. In 2016, the compensation committee did not make any equity awards to directors, other than in connection with the annual retainer grants described below. The 2015 Program also provides that non-employee directors are entitled to an annual retainer in the amount of $35,000, which individual directors may elect to take in the form of cash, restricted stock or options to purchase common stock. Compensation taken in the form of restricted stock or options to purchase our common stock shall vest over one year in four equal quarterly amounts on the first day of each fiscal quarter, provided the individual continues to serve as a director. In 2016, two of our directors elected to receive their annual retainer in the form of options, two directors elected to receive a portion of the retainer in cash and a portion in restricted stock, and one elected to receive the retainer in cash.
Pursuant to the 2015 Program, directors who serve as members of the audit committee are entitled to additional compensation at the rate of $10,000 per annum, with the chairman of the audit committee being compensated at the rate of $20,000 per annum. Directors who serve as members of the compensation committee are entitled to additional compensation at the rate of $7,500 per annum, with the chairman of the compensation committee being compensated at the rate of $15,000 per annum. Directors who serve as members of the nominating and governance committee are entitled to additional compensation at the rate of $5,000 per annum, with the chairman of the nominating and governance committee being compensated at the rate of $10,000 per annum. In addition, the 2015 Program provides that a director who serves in the position of lead independent director shall be entitled to compensation at the rate of $30,000 per annum. Pursuant to the terms of the 2015 Program, all such board and committee fees shall be paid quarterly in advance.
In February 2016, the Board also established a committee to advise management and assist in the review and negotiation of certain transactions. Robert Van Nostrand and Allan Rothstein, who were the non-employee directors on this committee, were compensated at the rate of $20,000 per quarter for their service. The Board determined to suspend the committee effective as of September 30, 2016.
The following table sets forth compensation paid to our non-employee directors during fiscal year 2016:
| 64 |
| Name | Fees earned or paid in cash ($)(1) | Stock awards ($)(2) | Option awards ($)(2) | Total ($) | ||||||||||||
| Barry Buckland, Ph.D. | 31,500 | (3) | — | 28,604 | 60,104 | |||||||||||
| Robert J. Easton | 20,000 | — | 28,604 | 48,604 | ||||||||||||
| Allan P. Rothstein | 65,429 | 21,000 | — | 86,429 | ||||||||||||
| Paul J. Sekhri | 65,000 | — | — | 65,000 | ||||||||||||
| Robert L. Van Nostrand | 128,929 | 10,000 | — | 138,929 | ||||||||||||
| (1) | Except as otherwise specifically noted, the amounts set forth in this column represent fees paid for service on our board of directors and committees thereof. |
| (2) | These amounts represent the aggregate grant date fair value of stock option awards and restricted stock awards granted in fiscal year 2016 under the 2014 Plan, computed in accordance with FASB ASC Topic 718. These amounts do not represent the actual amounts paid to or realized by the directors during fiscal year 2016, and there can be no assurance that the Topic 718 amount will ever be realized. A description of the assumptions we used for purposes of determining grant date fair value is set forth in the notes to our consolidated financial statements included in this Annual Report on Form 10-K for the year ended December 31, 2016. |
| (3) | Represents the aggregate of (i) $14,000 paid to Dr. Buckland pursuant to his service on our Scientific Advisory Board (“SAB”) and (ii) $17,500 paid to Dr. Buckland for service on our board of directors and committees thereof. For additional information on Dr. Buckland’s SAB agreement, please refer to “Certain Relationships and Related Transactions, and Director Independence.” |
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS.
Beneficial ownership is determined in accordance with the rules of the SEC and generally includes voting or investment power with respect to securities. In accordance with SEC rules, shares of our common stock which may be acquired upon exercise of stock options or warrants which are currently exercisable or which become exercisable within 60 days of the date of the table below are deemed beneficially owned by the holders of such options and warrants and are deemed outstanding for the purpose of computing the percentage of ownership of such person, but are not treated as outstanding for the purpose of computing the percentage of ownership of any other person. Subject to community property laws, where applicable, the persons or entities named in the tables below have sole voting and investment power with respect to all shares of our common stock indicated as beneficially owned by them.
The following table sets forth information with respect to the beneficial ownership of our common stock as of March 17, 2017 (the “Determination Date”), by (i) each stockholder known by us to be the beneficial owner of more than 5% of our common stock (our only classes of voting securities), (ii) each of our directors and executive officers, and (iii) all of our directors and executive officers as a group. To the best of our knowledge, except as otherwise indicated, each of the persons named in the table has sole voting and investment power with respect to the shares of our common stock beneficially owned by such person, except to the extent such power may be shared with a spouse. To our knowledge, none of the shares listed below are held under a voting trust or similar agreement, except as noted. In addition, to our knowledge there is no arrangement, including any pledge by any person of securities of our company or any of its parents, the operation of which may at a subsequent date result in a change in control of our company.
| 65 |
| Title of class | Name and address of beneficial owner (1) | Amount and nature of beneficial ownership | Percent of class (2) | |||||||
| Common Stock | E. Jeffrey Peierls (3) 73 South Holman Way Golden, CO 80401 | 11,430,080 | 8.9 | % | ||||||
| Common Stock | Brian Eliot Peierls (4) 3017 McCurdy St. Austin, TX 78723 | 11,149,120 | 8.7 | % | ||||||
| Common Stock | Harris & Harris Group, Inc. (5) 1450 Broadway, 24th Floor New York, NY 10018 | 9,221,488 | 7.1 | % | ||||||
| Common Stock | A.G. Family L.P. (6) 571 McDonald Road Rockwall, Texas 75032 | 6,419,200 | 5.0 | % | ||||||
| Common Stock | Wael Fayad (7) | 398,000 | * | |||||||
| Common Stock | Barry Buckland, Ph.D. (8) | 617,386 | * | |||||||
| Common Stock | Robert J. Easton (9) | 851,758 | * | |||||||
| Common Stock | Allan P. Rothstein (10) | 2,494,469 | 1.9 | % | ||||||
| Common Stock | Paul J. Sekhri (11) | 319,338 | * | |||||||
| Common Stock | Robert L. Van Nostrand (12) | 412,459 | * | |||||||
| Common Stock | Kevin G. Sarney (13) | 516,250 | * | |||||||
| Common Stock | All of our directors and executive officers as a group (7 persons) | 5,609,660 | 4.3 | % | ||||||
| * | Less than 1% |
| (1) | Unless otherwise set forth above, the address for each of the persons or entities listed above is c/o Enumeral Biomedical Holdings, Inc., 200 CambridgePark Drive, Suite 2000, Cambridge, MA 02140. |
| (2) | Applicable percentage ownership is based on 128,409,788 shares of common stock outstanding as of the Determination Date, together with securities exercisable or convertible into shares of common stock within 60 days after the Determination Date, for each shareholder. Beneficial ownership is determined in accordance with the rules of the SEC and generally includes voting or investment power with respect to securities. |
| (3) | Consists of 10,890,080 shares of common stock and 540,000 shares of common stock issuable upon exercise of PPO Warrants that are exercisable within 60 days of the Determination Date in each case owned by E. Jeffrey Peierls individually or by various trusts or The Peierls Foundation, Inc. of which he is a fiduciary. Mr. Peierls disclaims beneficial ownership of 5,666,360 shares of common stock and PPO Warrants to purchase 341,000 shares of common stock owned by The Peierls Foundation, Inc. Information is based on a Schedule 13G filed by E. Jeffrey Peierls and Brian Eliot Peierls filed with the SEC on February 10, 2017. Mr. Peierls does not serve as an officer or director of our company. |
| (4) | Consists of 10,629,120 shares of common stock and 520,000 shares of common stock issuable upon exercise of PPO Warrants that are exercisable within 60 days of the Determination Date in each case owned by Brian Elliot Peierls individually or by various trusts or The Peierls Foundation, Inc. of which he is a fiduciary. Mr. Peierls disclaims beneficial ownership of 5,666,360 shares of common stock and PPO Warrants to purchase 341,000 shares of common stock owned by The Peierls Foundation, Inc. Information is based on a Schedule 13G filed by E. Jeffrey Peierls and Brian Eliot Peierls filed with the SEC on February 10, 2017. Mr. Peierls does not serve as an officer or director of our company. |
| (5) | Consists of 7,966,368 shares of common stock owned directly by Harris & Harris Group, Inc. (“Harris”), 255,120 shares that Harris has the right to acquire upon the exercise of warrants that are exercisable within 60 days of the Determination Date, and 1,000,000 shares that Harris has the right to acquire upon the exercise of PPO Warrants that are exercisable within 60 days of the Determination Date. |
| (6) | Consists of 6,419,200 shares of common stock owned directly by A.G. Family L.P. Thomas A. Satterfield has a limited power of attorney for voting and disposition purposes, and as such may be deemed to have shared voting and dispositive power over the share of common stock beneficially owned by A.G. Family L.P. Information is based on a Schedule 13G filed by A.G. Family L.P. and Mr. Satterfield filed with the SEC on January 23, 2017. Mr. Satterfield does not serve as an officer or director of our company. |
| (7) | Consists of 298,000 shares of common stock owned directly by Mr. Fayad and 100,000 shares that Mr. Fayal has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. Terms of certain options are described in such option agreements. Does not include shares that Mr. Fayad has the right to acquire upon exercise of options subject to performance based criteria, which criteria has not yet been achieved. |
| 66 |
| (8) | Consists of 120,882 shares of common stock owned directly by Dr. Buckland, 175,286 shares of common stock owned by BiologicB, LLC, a company wholly-owned by Dr. Buckland, and 321,218 shares that Dr. Buckland has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. |
| (9) | Consists of 480,000 shares of common stock owned directly by Mr. Easton and 371,758 shares that Mr. Easton has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. |
| (10) | Consists of 2,024,415 shares of common stock owned directly by Mr. Rothstein, 270,427 shares that Mr. Rothstein has the right to acquire upon the exercise of warrants that are exercisable within 60 days of the Determination Date, and 199,627 shares that Mr. Rothstein has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. |
| (11) | Consists of 175,000 shares of common stock owned directly by Mr. Sekhri and 144,338 shares that Mr. Sekhri has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. |
| (12) | Consists of 268,121 shares of common stock owned directly by Mr. Van Nostrand and 144,338 shares that Mr. Van Nostrand has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. |
| (13) | Consists of 35,000 shares of common stock owned directly by Mr. Sarney and 481,250 shares that Mr. Sarney has the right to acquire upon the exercise of options that are exercisable within 60 days of the Determination Date. Terms of certain options are described in such option agreements. Does not include shares that Mr. Sarney has the right to acquire upon exercise of options subject to performance based criteria, which criteria has not yet been achieved. |
Equity Compensation Plan Information
The following table provides information with respect to the 2014 Plan, as well as grants outside of the 2014 Plan, as of December 31, 2016:
| Equity Compensation Plan Information | ||||||||||||
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights | Weighted- average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) | |||||||||
| Equity compensation plans approved by security holders | 5,920,823 | (1) | $ | 0.47 | 1,264,018 | |||||||
| Equity compensation plans not approved by security holders | 2,059,966 | (2) | $ | 0.27 | — | |||||||
| Total | 7,980,789 | $ | 0.42 | 1,264,018 | ||||||||
| (1) | A total of 8,100,000 shares of our common stock were authorized for issuance pursuant to awards under the 2014 Plan as of December 31, 2016, of which 5,920,823 stock options have been awarded to participants under the 2014 Plan and are outstanding as of December 31, 2016. The amount listed above does not include 790,253 shares of restricted stock granted under the 2014 Plan for which restrictions have lapsed as of December 31, 2016. |
| 67 |
| (2) | On April 15, 2014, John Rydzewski and Arthur Tinkelenberg, our former Executive Chairman and President and Chief Executive Officer, respectively, received warrants to purchase 47,058 and 58,823 shares of Enumeral Series B Preferred Stock, respectively, in relation to the executives agreeing to take a temporary salary reduction. In connection with the Merger, on July 31, 2014 Mr. Rydzewski’s and Dr. Tinkelenberg’s warrants to purchase Enumeral Series B Preferred Stock were converted into warrants to purchase 137,762 and 172,204 shares of our common stock, respectively. These warrants were granted outside of our 2014 Plan. |
On September 21, 2016, Wael Fayad was granted non-qualified stock options outside of the 2014 Plan to purchase 1,750,000 shares of Common Stock at an exercise price of $0.19 per share in connection with Mr. Fayad’s appointment as our Chairman, Chief Executive Officer and President. See “Employment Agreements – Wael Fayad” below.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE.
SEC rules require us to disclose any transaction or currently proposed transaction in which we are a participant and in which any related person has or will have a direct or indirect material interest involving the lesser of $120,000 or one percent (1%) of the average of our total assets as of the end of last two completed fiscal years. A related person is any executive officer, director, nominee for director, or holder of 5% or more of our common stock, or an immediate family member of any of those persons.
Pursuant to our Warrant Tender Offer which we launched on October 28, 2016 (as subsequently amended, the "Offering"), the following executive officers, directors, and holders of 5% or more of our common stock elected to participate and exercise (at the reduced exercise price of $0.50 per warrant) all or a portion of the warrants held by such person and received four shares for each Warrant tendered: Mr. Sekhri exercised 43,750 warrants with an aggregate exercise price of $21,875 and received 175,000 shares of common stock; Mr. Sarney exercised 8,750 warrants with an aggregate exercise price of $4,375 and received 35,000 shares of common stock; Mr. Rothstein exercised 50,000 warrants with an aggregate exercise price of $25,000 and received 200,000 shares of common stock; Mr. Easton exercised 120,000 warrants with an aggregate exercise price of $60,000 and received 480,000 shares of common stock; Mr. Fayad exercised 74,500 warrants with an aggregate exercise price of $37,250 and received 298,000 shares of common stock; Mr. Buckland exercised 13,000 warrants with an aggregate exercise price of $6,500 and received 52,000 shares of common stock; and Mr. Van Nostrand exercised 39,000 warrants with an aggregate exercise price of $19,500 and received 156,000 shares of common stock.
In September 2014, we and Dr. Buckland entered into a Scientific Advisory Board Agreement (the “SAB Agreement”) pursuant to which Dr. Buckland serves as chairman of our Scientific Advisory Board. In September 2016, we and Dr. Buckland entered into an amendment to the SAB Agreement to extend the term thereof to September 2017. Pursuant to the terms of the SAB Agreement, Dr. Buckland will receive compensation on an hourly or per diem basis, either in cash or, at Dr. Buckland’s election, in options to purchase our common stock, provided that any such compensation will not exceed $100,000 in any continuous twelve month period. The SAB Agreement has a term of two years. During 2016 and 2015, Mr. Buckland received aggregate compensation of $14,000 and $38,000, respectively.
Director Independence
While we are currently quoted on the over-the-counter market, we have adopted the corporate governance standards of a listed company on the Nasdaq Stock Market, or Nasdaq. These standards require that a majority of the members of our board of directors be “independent,” as Nasdaq defines that term, and that our board make an affirmative determination as to the independence of each director. Consistent with these rules, our board of directors undertook its annual review of director independence in March 2017. During the review, our board considered relationships and transactions during 2016 and during the past three fiscal years between each director or any member of his or her immediate family, on the one hand, and our company and its subsidiaries and affiliates, on the other hand. The purpose of this review was to determine whether any such relationships or transactions were inconsistent with a determination that the director is independent. Based on this review, our board of directors determined that Dr. Buckland and Messrs. Easton, Rothstein, Sekhri, and Van Nostrand are independent under the criteria established by Nasdaq and by our board of directors.
| 68 |
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES.
As a matter of corporate governance, we will not engage our independent registered public accounting firm to render audit or non-audit services unless the service is specifically approved in advance by the audit committee.
The following table shows the fees billed by Friedman LLP for audit and other services provided for the fiscal years ended December 31, 2016 and 2015, respectively. All of the services described in the following fee table were approved in conformity with the audit committee’s pre-approval process.
| Type of Fee | 2016 | 2015 | ||||||
| Audit Fees (1) | $ | 155,780 | $ | 156,500 | ||||
| Audit Related Fees (2) | — | — | ||||||
| Tax Fees (3) | — | — | ||||||
| All Other Fees (4) | — | — | ||||||
| Total | $ | 155,780 | $ | 156,500 | ||||
| (1) | Audit Fees – This category includes the audit of our annual consolidated financial statements and services that are normally provided by independent auditors in connection with the engagement for fiscal years. |
| (2) | Audit Related Services – This category consists of fees reasonably related to the performance of the audit or review of our consolidated financial statements that are not reported as “Audit Fees.” |
| (3) | Tax Fees – This category consists of tax compliance, tax advice and tax planning work. |
| (4) | All Other Fees – This category consists of fees for other miscellaneous items. |
| 69 |
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES.
| (a)(1) | Financial statements. |
| · | Report of Independent Registered Public Accounting Firm |
| · | Consolidated Balance Sheets as of December 31, 2016 and 2015 |
| · | Consolidated Statements of Operations and Comprehensive Income (Loss) for the years ended December 31, 2016 and 2015 |
| · | Consolidated Statements of Changes in Stockholders’ Equity (Deficiency) for the years ended December 31, 2016 and 2015 |
| · | Consolidated Statements of Cash Flows for the years ended December 31, 2016 and 2015 |
| · | Notes to Consolidated Financial Statements |
| (b) | Exhibits. |
| Exhibit Number |
Description | |
| 2.1 | Agreement and Plan of Merger and Reorganization, dated as of July 31, 2014, by and among the Company, Acquisition Sub and Enumeral Biomedical Corp. (Incorporated by reference to Exhibit 2.1 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 3.1 | Amended and Restated Certificate of Incorporation of the Company. (Incorporated by reference to Exhibit 3.1 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 3.2 | Amended and Restated By-Laws of the Registrant. (Incorporated by reference to Exhibit 3.2 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 3.3 | Certificate of Merger of Enumeral Biomedical Corp., with and into Acquisition Sub, filed July 31, 2014. (Incorporated by reference to Exhibit 3.3 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 4.1 | Form of 12% Senior Secured Promissory Note issued by Enumeral Biomedical Holdings, Inc. in July 29, 2016 Notes Offering (Incorporated by reference to Exhibit 4.1 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.1§ | Exclusive Patent License Agreement, dated as of April 15, 2011, between Enumeral Biomedical Corp. and the Massachusetts Institute of Technology. (Incorporated by reference to Exhibit 10.22 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on October 31, 2014 (SEC File No. 333-185891)). | |
| 10.2§ | First Amendment to Exclusive Patent License Agreement, effective as of March 8, 2013, between Enumeral Biomedical Corp. and the Massachusetts Institute of Technology. (Incorporated by reference to Exhibit 10.23 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on October 31, 2014 (SEC File No. 333-185891)). | |
| 10.3 | Second Amendment to Exclusive Patent License Agreement, effective as of July 16, 2014, between Enumeral Biomedical Corp. and the Massachusetts Institute of Technology. (Incorporated by reference to Exhibit 10.24 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.4§ | Third Amendment to Exclusive Patent License Agreement, effective as of April 15, 2015, between Enumeral Biomedical Corp. and the Massachusetts Institute of Technology. (Incorporated by reference to Exhibit 10.1 of the Form 10-Q for the period ended March 31, 2015 as filed with the Securities and Exchange Commission on May 15, 2015 (SEC File No. 000-55415)). |
| 70 |
| 10.5§ | Fourth Amendment to Exclusive Patent License Agreement, effective as of April 14, 2016, between Enumeral Biomedical Corp. and the Massachusetts Institute of Technology. (Incorporated by reference to Exhibit 10.1 of the Form 10-Q for the period ended March 31, 2016 as filed with the Securities and Exchange Commission on May 13, 2016 (SEC File No. 000-55415)). | |
| 10.6§ | Definitive License and Transfer Agreement, dated as of June 6, 2016, between Enumeral Biomedical Holdings, Inc., Pieris Pharmaceuticals, Inc., and Pieris Pharmaceuticals GmbH. (Incorporated by reference to Exhibit 10.1 of the Form 10-Q for the period ended June 30, 2016 as filed with the Securities and Exchange Commission on August 12, 2016 (SEC File No. 000-55415)). | |
| 10.7* | Amendment No. 1 to Definitive License and Transfer Agreement, effective as of January 3, 2017, between Enumeral Biomedical Holdings, Inc., Pieris Pharmaceuticals, Inc., and Pieris Pharmaceuticals GmbH. | |
| 10.8§ | Collaborative Research and Development Agreement, dated as of January 11, 2016, by and between Enumeral Biomedical Holdings, Inc. and the University of Texas M.D. Anderson Cancer Center. (Incorporated by reference to Exhibit 10.31 of the Form 10-K for the year ended December 31, 2015 as filed with the Securities and Exchange Commission on March 30, 2016 (SEC File No. 000-55415)). | |
| 10.9§ | Award/Contract, dated as of September 10, 2014, between Enumeral Biomedical Corp. and National Cancer Institute. (Incorporated by reference to Exhibit 10.2 of the Form 10-Q for the quarterly period ended September 30, 2014 as filed with the Securities and Exchange Commission on November 14, 2014 (SEC File No. 333-185891)). | |
| 10.10 | Amendment of Solicitation/Modification of Contract, dated September 13, 2016, to Award/Contract, dated September 10, 2014, between Enumeral Biomedical Corp. and National Cancer Institute. (Incorporated by reference to Exhibit 10.11 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.11§ | Study Agreement, dated as of December 17, 2014, between Enumeral Biomedical Holdings, Inc. and Merck Sharp & Dohme Corp. (Incorporated by reference to Exhibit 10.36 of the Form 10-K for the year ended December 31, 2014 as filed with the Securities and Exchange Commission on March 19, 2015 (SEC File No. 333-185891)). | |
| 10.12§ | Amendment No. 1 to Study Agreement, dated as of February 16, 2016, between Enumeral Biomedical Holdings, Inc. and Merck Sharp & Dohme Corp. (Incorporated by reference to Exhibit 10.35 of the Form 10-K for the year ended December 31, 2015 as filed with the Securities and Exchange Commission on March 30, 2016 (SEC File No. 000-55415)). | |
| 10.13 | Indenture of Lease, dated as of November 7, 2014, between King 200 CPD LLC and Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.1 of the Form 10-Q for the quarterly period ended September 30, 2014 as filed with the Securities and Exchange Commission on November 14, 2014 (SEC File No. 333-185891)). | |
| 10.14† | 2014 Equity Incentive Plan of Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.11 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.15† | Amendment No. 1 to Enumeral Biomedical Holdings, Inc.’s 2014 Equity Incentive Plan, adopted on December 1, 2014. (Incorporated by reference to Exhibit 10.3 of the Form 8-K for the event occurring on December 1, 2014 as filed with the Securities and Exchange Commission on December 4, 2014 (SEC File No. 333-185891)). | |
| 10.16† | Amendment No. 2 to Enumeral Biomedical Holdings, Inc.’s 2014 Equity Incentive Plan, adopted on May 21, 2015. (Incorporated by reference to Exhibit 10.1 of the Form 8-K for the event occurring on May 21, 2015 as filed with the Securities and Exchange Commission on May 22, 2015 (SEC File No. 000-55415)). | |
| 10.17† | Form of Non-Qualified Stock Option Agreement under 2014 Equity Incentive Plan. (Incorporated by reference to Exhibit 10.12 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.18† | Form of Stock Appreciation Right Grant Agreement under 2014 Equity Incentive Plan. (Incorporated by reference to Exhibit 10.13 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). |
| 71 |
| 10.19† | Form of Restricted Stock Agreement under 2014 Equity Incentive Plan. (Incorporated by reference to Exhibit 10.14 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.20† | Form of Incentive Stock Option Agreement under 2014 Equity Incentive Plan. (Incorporated by reference to Exhibit 10.15 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.21†§ | Employment Offer Letter, dated September 21, 2016, between Enumeral Biomedical Holdings, Inc. and Wael Fayad. (Incorporated by reference to Exhibit 10.8 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.22†§ | Incentive Stock Option Agreement between Enumeral Biomedical Holdings, Inc. and Wael Fayad, dated September 21, 2016. (Incorporated by reference to Exhibit 10.9 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.23†§ | Non-Qualified Stock Option Agreement between Enumeral Biomedical Holdings, Inc. and Wael Fayad, dated September 21, 2016. (Incorporated by reference to Exhibit 10.10 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.24† | Employment Letter, dated March 24, 2016, between Enumeral Biomedical Holdings, Inc. and Kevin G. Sarney. (Incorporated by reference to Exhibit 10.37 of the Form 10-K for the year ended December 31, 2015 as filed with the Securities and Exchange Commission on March 30, 2016 (SEC File No. 000-55415)). | |
| 10.25† | Amended and Restated Employment Agreement, dated as of July 21, 2014 between Enumeral Biomedical Corp. and Arthur Tinkelenberg, as assumed by Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.16 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.26† | Amended and Restated Employment Agreement, dated as of July 21, 2014 between Enumeral Biomedical Corp. and John Rydzewski, as assumed by Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.17 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.27†§ | Incentive Stock Option Agreement between Enumeral Biomedical Holdings, Inc. and Arthur H. Tinkelenberg, dated September 28, 2015. (Incorporated by reference to Exhibit 10.1 of the Form 10-Q for the quarterly period ended September 30, 2015 as filed with the Securities and Exchange Commission on November 10, 2015 (SEC File No. 000-55415)). | |
| 10.28†§ | Incentive Stock Option Agreement between Enumeral Biomedical Holdings, Inc. and John J. Rydzewski, dated September 28, 2015. (Incorporated by reference to Exhibit 10.2 of the Form 10-Q for the quarterly period ended September 30, 2015 as filed with the Securities and Exchange Commission on November 10, 2015 (SEC File No. 000-55415)). | |
| 10.29† | Separation Letter Agreement, dated as of August 4, 2016, by and between Enumeral Biomedical Holdings, Inc. and Arthur H. Tinkelenberg, Ph.D. (Incorporated by reference to Exhibit 10.1 of the Current Report on Form 8-K, as filed with the Securities and Exchange Commission on August 10, 2016 (File No. 000-55415)). | |
| 10.30† | Separation Agreement, dated as of September 21, 2016, between John J. Rydzewski and Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.1 of the Current Report on Form 8-K, as filed with the Securities and Exchange Commission on September 23, 2016 (File No. 000-55415)). | |
| 10.31† | Employment Letter, dated March 24, 2016, between Enumeral Biomedical Holdings, Inc. and Anhco Nguyen, Ph.D. (Incorporated by reference to Exhibit 10.38 of the Form 10-K for the year ended December 31, 2015 as filed with the Securities and Exchange Commission on March 30, 2016 (SEC File No. 000-55415)). | |
| 10.32† | Scientific Advisory Board Agreement, dated as of September 14, 2014, between Enumeral Biomedical Holdings, Inc. and Barry Buckland, Ph.D. (Incorporated by reference to Exhibit 10.3 of the Form 10-Q for the quarterly period ended September 30, 2014 as filed with the Securities and Exchange Commission on November 14, 2014 (SEC File No. 333-185891)). |
| 72 |
| 10.33† | Amendment No. 1 to Scientific Advisory Board Agreement, dated as of September 21, 2016, between Barry Buckland, Ph.D. and Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.2 of the Current Report on Form 8-K, as filed with the Securities and Exchange Commission on September 23, 2016 (File No. 000-55415)). | |
| 10.34 | Form of Indemnification Agreement between Enumeral Biomedical Holdings, Inc. and each of its directors and officers (Incorporated by reference to Exhibit 10.45 of the Form 8-K/A for the event occurring on July 31, 2014 as filed with the Securities and Exchange Commission on August 8, 2014 (SEC File No. 333-185891)). | |
| 10.35 | Placement Agency Agreement, dated as of June 21, 2016, by and between Enumeral Biomedical Holdings, Inc. and Katalyst Securities LLC, as amended. (Incorporated by reference to Exhibit 10.1 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.36 | Form of Subscription Agreement entered into between Enumeral Biomedical Holdings, Inc. and Investors in July 29, 2016 Notes Offering. (Incorporated by reference to Exhibit 10.2 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.37 | Intellectual Property Security Agreement, dated as of July 29, 2016, by and among Enumeral Biomedical Holdings, Inc., Enumeral Biomedical Corp., the Buyers named therein, and Intuitive Venture Partners, LLC, in its capacity as the Collateral Agent. (Incorporated by reference to Exhibit 10.4 of the Form 10-Q for the period ended September 30, 2016 as filed with the Securities and Exchange Commission on November 10, 2016 (SEC File No. 000-55415)). | |
| 10.38 | Form of Registration Rights Agreement, (Incorporated by reference to Exhibit 99(d)(2) of the tender offer statement on Schedule TO, as filed with the Securities and Exchange Commission on October 28, 2016 (SEC File No. 005-88781)). | |
| 10.39 | Warrant Agent Agreement, dated as of October 26, 2016, between Katalyst Securities LLC, as warrant agent and Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit (99)(d)(1) of the tender offer statement on Schedule TO, as filed with the Securities and Exchange Commission on October 28, 2016 (SEC File No. 005-88781)). | |
| 10.40 | Form of Amended PPO Warrant for Common Stock of Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.39 of the Registration Statement on Form S-1 as filed with the Securities and Exchange Commission on March 8, 2017 (SEC File No. 333-216533)). | |
| 10.41 | Form of Amended Agent Warrant in the PPO for Common Stock of Enumeral Biomedical Holdings, Inc. (Incorporated by reference to Exhibit 10.40 of the Registration Statement on Form S-1 as filed with the Securities and Exchange Commission on March 8, 2017 (SEC File No. 333-216533)). | |
| 10.42* | Form of Amended Placement Agent Warrant for Common Stock of Enumeral Biomedical Holdings, Inc. issued in connection with conversion of July 2016 Promissory Notes. | |
| 10.43 | Form of Replacement Warrant issued by Enumeral Biomedical Holdings, Inc. to holders of warrants of Enumeral Biomedical Corp issued in connection with the Bridge Notes offering. (Incorporated by reference to Exhibit 10.37 of the Form 10-K for the year ended December 31, 2014 as filed with the Securities and Exchange Commission on March 19, 2015 (SEC File No. 333-185891)). | |
| 21.1* | Subsidiaries of Enumeral Biomedical Holdings, Inc. | |
| 23.1* | Consent of Friedman LLP | |
| 31.1* | Principal Executive Officer Certification required by Rule 13a-14(a) or Rule 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 31.2* | Principal Financial and Accounting Officer Certification required by Rule 13a-14(a) or Rule 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |
| 32.1** | Principal Executive Officer Certification pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. | |
| 32.2** | Principal Financial and Accounting Officer Certification pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. |
| 73 |
| 101* | Interactive Data Files of Financial Statements and Notes. | |
| 101.INS * | XBRL Instance Document. | |
| 101.SCH* | XBRL Taxonomy Extension Schema Document. | |
| 101.CAL* | XBRL Calculation Linkbase Document. | |
| 101.DEF* | XBRL Taxonomy Extension Definition Linkbase Document. | |
| 101.LAB* | XBRL Label Linkbase Document. | |
| 101.PRE* | XBRL Taxonomy Presentation Linkbase Document. |
| * | Filed herewith. |
| ** | Furnished herewith. |
| † | Management contract or compensatory plan or arrangement. |
| § | Confidential treatment requested as to certain portions, which portions have been filed separately with the Securities and Exchange Commission. |
| 74 |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| ENUMERAL BIOMEDICAL HOLDINGS, INC. | ||
| March 28, 2017 | By: | /s/ Wael Fayad |
| Name: | Wael Fayad | |
| Title: |
Chairman, President and Chief Executive Officer (principal executive officer) | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant in the capacities and on the dates indicated.
| Name | Positions | Date | ||
| /s/ Wael Fayad | Chairman, President and Chief Executive Officer | March 28, 2017 | ||
| Wael Fayad | (principal executive officer) | |||
| /s/ Barry Buckland, Ph.D. | Director | March 28, 2017 | ||
| Barry Buckland, Ph.D. | ||||
| /s/ Robert J. Easton | Director | March 28, 2017 | ||
| Robert J. Easton | ||||
| /s/ Allan P. Rothstein | Director | March 28, 2017 | ||
| Allan P. Rothstein | ||||
| /s/ Paul J. Sekhri | Director | March 28, 2017 | ||
| Paul J. Sekhri | ||||
| /s/ Robert L. Van Nostrand | Director | March 28, 2017 | ||
| Robert L. Van Nostrand | ||||
| /s/ Kevin G. Sarney | Vice President of Finance, Chief Accounting Officer and | March 28, 2017 | ||
| Kevin G. Sarney | Treasurer (principal financial officer and principal accounting officer) |
| 75 |
Enumeral Biomedical Holdings, Inc.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| Page Number | |
| Report of Independent Registered Public Accounting Firm | F-2 |
| Consolidated Financial Statements for the Years Ended December 31, 2016 and 2015 | |
| Consolidated Balance Sheets | F-3 |
| Consolidated Statements of Operations and Comprehensive Income (Loss) | F-4 |
| Consolidated Statements of Stockholders’ Equity (Deficiency) | F-5 |
| Consolidated Statements of Cash Flows | F-6 |
| Notes to Consolidated Financial Statements | F-7 |
| F-1 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of
Enumeral Biomedical Holdings, Inc.
We have audited the accompanying consolidated balance sheets of Enumeral Biomedical Holdings, Inc. and subsidiaries as of December 31, 2016 and 2015, and the related consolidated statements of operations and comprehensive income (loss), stockholders’ equity (deficiency), and cash flows for each of the years in the two-year period ended December 31, 2016. Enumeral Biomedical Holdings, Inc.’s management is responsible for these consolidated financial statements. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Enumeral Biomedical Holdings, Inc. as of December 31, 2016 and 2015, and the results of its operations and its cash flows for each of the years in the two-year period ended December 31, 2016 in conformity with accounting principles generally accepted in the United States of America.
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the consolidated financial statements, the Company has recurring operating losses and negative cash flows from operations. These conditions raise substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 2. The consolidated financial statements do not include any adjustments relating to the recoverability and classification of assets carrying amounts or the amount and classification of liabilities that might result should the Company be unable to continue as a going concern.
| /s/ FRIEDMAN LLP |
| New York, New York |
| March 28, 2017 |
| F-2 |
Enumeral Biomedical Holdings, Inc.
Consolidated Balance Sheets
| As of December 31, | ||||||||
| 2016 | 2015 | |||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 3,162,400 | $ | 3,596,262 | ||||
| Accounts receivable | 189,068 | 306,012 | ||||||
| Prepaid expenses and other current assets | 47,317 | 280,479 | ||||||
| Total current assets | 3,398,785 | 4,182,753 | ||||||
| Property and equipment, net | 902,097 | 1,511,493 | ||||||
| Other assets: | ||||||||
| Restricted cash | 534,780 | 534,780 | ||||||
| Other assets | 111,556 | 114,572 | ||||||
| Total assets | $ | 4,947,218 | $ | 6,343,598 | ||||
| LIABILITIES AND STOCKHOLDERS' EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 308,857 | $ | 343,736 | ||||
| Accrued expenses and other current liabilities | 386,355 | 714,384 | ||||||
| Deferred revenue | 14,505 | 130,539 | ||||||
| Equipment lease financing | 251,631 | 240,473 | ||||||
| Derivative liabilities | - | 2,138,091 | ||||||
| Total current liabilities | 961,348 | 3,567,223 | ||||||
| Deferred rent, net of current portion | 63,116 | 36,847 | ||||||
| Long-term equipment lease financing | 14,840 | 266,471 | ||||||
| Total liabilities | 1,039,304 | 3,870,541 | ||||||
| Commitments and contingencies | ||||||||
| Stockholders' equity: | ||||||||
| Preferred stock, $0.001 par value; 10,000,000 shares authorized: -0- shares issued and outstanding as of December 31, 2016 and 2015, respectively | - | - | ||||||
| Common stock, $0.001 par value; 300,000,000 shares authorized: 128,343,122 and 51,932,571 shares issued and outstanding as of December 31, 2016 and 2015, respectively | 128,343 | 51,933 | ||||||
| Additional paid-in-capital | 30,044,778 | 16,830,100 | ||||||
| Accumulated deficit | (26,265,207 | ) | (14,408,976 | ) | ||||
| Total stockholders’ equity | 3,907,914 | 2,473,057 | ||||||
| Total liabilities and stockholders’ equity | $ | 4,947,218 | $ | 6,343,598 | ||||
The accompanying notes are an integral part of the consolidated financial statements
| F-3 |
Enumeral Biomedical Holdings, Inc.
Consolidated Statements of Operations and Comprehensive Income (Loss)
| Year Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Revenue: | ||||||||
| Collaboration and license revenue | $ | 1,999,666 | $ | 1,100,000 | ||||
| Grant revenue | 453,202 | 389,385 | ||||||
| Total revenue | 2,452,868 | 1,489,385 | ||||||
| Cost of revenue and expenses: | ||||||||
| Research and development | 4,865,056 | 6,493,859 | ||||||
| General and administrative | 4,561,076 | 5,695,932 | ||||||
| Total cost of revenue and expenses | 9,426,132 | 12,189,791 | ||||||
| Loss from operations | (6,973,264 | ) | (10,700,406 | ) | ||||
| Other income (expense): | ||||||||
| Interest income (expense) | (4,913,545 | ) | 9,699 | |||||
| Loss on extinguishment of derivative liabilities | (1,577,896 | ) | - | |||||
| Change in fair value of derivative liabilities | 1,608,474 | 13,980,711 | ||||||
| Total other income (expense), net | (4,882,967 | ) | 13,990,410 | |||||
| Net income (loss) before income taxes | (11,856,231 | ) | 3,290,004 | |||||
| Provision for income taxes | - | - | ||||||
| Net income (loss) | $ | (11,856,231 | ) | $ | 3,290,004 | |||
| Other comprehensive income (loss): | ||||||||
| Reclassification of loss included in net income | - | 19,097 | ||||||
| Net unrealized holding losses on available-for-sale securities arising during the period | - | (9,320 | ) | |||||
| Comprehensive income (loss) | $ | (11,856,231 | ) | $ | 3,299,781 | |||
| Basic net income (loss) per share | $ | (0.21 | ) | $ | 0.06 | |||
| Diluted net income (loss) per share | $ | (0.21 | ) | $ | 0.06 | |||
| Weighted-average number of common shares outstanding - basic | 56,188,284 | 51,679,230 | ||||||
| Weighted-average number of common shares oustanding - diluted | 56,188,284 | 52,365,494 | ||||||
The accompanying notes are an integral part of the consolidated financial statements
| F-4 |
Enumeral Biomedical Holdings, Inc.
Consolidated Statements of Stockholders’ Equity (Deficiency)
| Common Stock | Additional Paid-In | Accumulated
Other Comprehensive | Accumulated | Total Stockholders' Equity | ||||||||||||||||||||
| Shares | Amount | Capital | Loss | Deficit | (Deficiency) | |||||||||||||||||||
| Balance as of December 31, 2014 | 51,588,617 | $ | 51,589 | $ | 15,965,252 | $ | (9,777 | ) | $ | (17,690,647 | ) | $ | (1,683,583 | ) | ||||||||||
| Stock-based compensation expense | - | - | 827,184 | - | - | 827,184 | ||||||||||||||||||
| Reclassification of loss included in net income | - | - | - | 19,097 | - | 19,097 | ||||||||||||||||||
| Unrealized holding losses on available-for-sale securities | - | - | - | (9,320 | ) | - | (9,320 | ) | ||||||||||||||||
| Exercise of stock options | 124,906 | 125 | 29,550 | - | - | 29,675 | ||||||||||||||||||
| Issuance of restricted stock | 219,048 | 219 | (219 | ) | - | - | - | |||||||||||||||||
| Cumulative effect of change in accounting principle (Note 3) | - | - | 8,333 | - | (8,333 | ) | - | |||||||||||||||||
| Net income | - | - | - | - | 3,290,004 | 3,290,004 | ||||||||||||||||||
| Balance as of December 31, 2015 | 51,932,571 | $ | 51,933 | $ | 16,830,100 | $ | - | $ | (14,408,976 | ) | $ | 2,473,057 | ||||||||||||
| Stock-based compensation expense | - | - | 932,325 | - | - | 932,325 | ||||||||||||||||||
| Issuance of restricted stock | 140,910 | 141 | (141 | ) | - | - | - | |||||||||||||||||
| Warrant Tender Offer and extinguishment of derivative liabilities (Note 10) | 27,463,096 | 27,463 | 5,027,333 | - | - | 5,054,796 | ||||||||||||||||||
| Conversion of the Notes as a result of the Warrant Tender Offer (Note 10) | 48,806,545 | 48,806 | 7,255,161 | - | - | 7,303,967 | ||||||||||||||||||
| Net loss | - | - | - | - | (11,856,231 | ) | (11,856,231 | ) | ||||||||||||||||
| Balance as of December 31, 2016 | 128,343,122 | $ | 128,343 | $ | 30,044,778 | $ | - | $ | (26,265,207 | ) | $ | 3,907,914 | ||||||||||||
The accompanying notes are an integral part of the consolidated financial statements
| F-5 |
Enumeral Biomedical Holdings, Inc.
Consolidated Statements of Cash Flows
| Year Ended December 31, | ||||||||
| 2016 | 2015 | |||||||
| Cash flows from operating activities: | ||||||||
| Net income (loss) | $ | (11,856,231 | ) | $ | 3,290,004 | |||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 609,396 | 616,523 | ||||||
| Exit costs associated with write-off of net carrying value of leasehold improvements | - | 22,962 | ||||||
| Stock-based compensation | 932,325 | 827,184 | ||||||
| Change in fair value of derivative liabilities | (1,608,474 | ) | (13,980,711 | ) | ||||
| Loss on extinguishment of derivative liabilities | 1,577,896 | - | ||||||
| Realized loss on marketable securities | - | 19,097 | ||||||
| Non-cash interest expense | 4,265,711 | - | ||||||
| Accretion of debt discount | 507,849 | - | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | 116,944 | (21,611 | ) | |||||
| Prepaid expenses and other assets | 236,178 | (184,255 | ) | |||||
| Accounts payable | (34,879 | ) | (270,370 | ) | ||||
| Accrued expenses and other liabilities | (328,029 | ) | 497,243 | |||||
| Deferred rent | 26,269 | 4,431 | ||||||
| Deferred revenue | (116,034 | ) | (105,549 | ) | ||||
| Net cash used in operating activities | (5,671,079 | ) | (9,285,052 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Purchases of property and equipment | - | (1,050,506 | ) | |||||
| Proceeds from sale of marketable securities | - | 3,000,799 | ||||||
| Receipt of security deposit | - | 27,630 | ||||||
| Net cash provided by investing activities | - | 1,977,923 | ||||||
| Cash flows from financing activities: | ||||||||
| Proceeds from the issuance of promissory notes, net of issuance costs | 2,530,407 | - | ||||||
| Proceeds from the Warrant Tender Offer, net of issuance costs | 2,947,283 | - | ||||||
| Proceeds from the exercise of stock options | - | 29,675 | ||||||
| Proceeds from the equipment lease financing | - | 413,599 | ||||||
| Payments on equipment lease financing | (240,473 | ) | - | |||||
| Net cash provided by financing activities | 5,237,217 | 443,274 | ||||||
| Net decrease in cash and cash equivalents | (433,862 | ) | (6,863,855 | ) | ||||
| Cash and cash equivalents, beginning of period | 3,596,262 | 10,460,117 | ||||||
| Cash and cash equivalents, end of period | $ | 3,162,400 | $ | 3,596,262 | ||||
| Supplemental cash flow disclosures: | ||||||||
| Cash paid for interest | $ | 142,637 | $ | 6,464 | ||||
| Non-cash equipment lease financing | $ | - | $ | 93,345 | ||||
| Conversion of the Notes into common stock | $ | 3,038,256 | $ | - | ||||
| Reclassification of derivative liabilities to equity | $ | 529,617 | $ | - | ||||
The accompanying notes are an integral part of the consolidated financial statements
| F-6 |
ENUMERAL BIOMEDICAL HOLDINGS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1 - NATURE OF BUSINESS
Enumeral Biomedical Corp. (“Enumeral”) was founded in 2009 in the state of Delaware as Enumeral Technologies, Inc. The name was later changed to Enumeral Biomedical Corp.
On July 31, 2014, Enumeral entered into an Agreement and Plan of Merger and Reorganization (the “Merger Agreement”) with Enumeral Biomedical Holdings, Inc., which was formerly known as Cerulean Group, Inc. (“Enumeral Biomedical” or the “Company”), and Enumeral Acquisition Corp., a wholly owned subsidiary of Enumeral Biomedical (“Acquisition Sub”), pursuant to which the Acquisition Sub merged with and into Enumeral (the “Merger”). Enumeral was the surviving corporation in the Merger and became a wholly owned subsidiary of the Company.
As a result of the Merger, all issued and outstanding common and preferred shares of Enumeral were exchanged for common shares of Enumeral Biomedical Holdings, Inc. The Merger is considered to be a recapitalization of the Company which has been retrospectively applied to these financial statements for all periods presented.
Upon the closing of the Merger and under the terms of a split-off agreement and a general release agreement (the “Split-Off Agreement”), the Company transferred all of its pre-Merger operating assets and liabilities to its wholly-owned special-purpose subsidiary, Cerulean Operating Corp. (the “Split-Off Subsidiary”). Thereafter, pursuant to the Split-Off Agreement, the Company transferred all of the outstanding shares of capital stock of the Split-Off Subsidiary to the pre-Merger majority stockholder of the Company, and the former sole officer and director of the Company (the “Split-Off”), in consideration of and in exchange for (i) the surrender and cancellation of an aggregate of 23,100,000 shares of the Company’s common stock held by such stockholder (which were cancelled and will resume the status of authorized but unissued shares of the Company’s common stock) and (ii) certain representations, covenants and indemnities.
As a result of the Merger and the Split-Off, the Company discontinued its pre-Merger business and acquired the business of Enumeral, and is continuing the existing business operations of Enumeral as a publicly-traded company under the name Enumeral Biomedical Holdings, Inc.
Also on July 31, 2014, the Company closed a private placement offering (the “PPO”) of 21,549,510 Units (the “Units”) of its securities, at a purchase price of $1.00 per Unit, each Unit consisting of one share of the Company’s common stock and a warrant to purchase one share of the Company’s common stock at an exercise price of $2.00 per share and with a term of five years (the “PPO Warrants”). Additional information concerning the PPO and PPO Warrants is presented below in Note 10.
Also on July 31, 2014, the Company changed its fiscal year from a fiscal year ending on October 31 of each year to one ending on December 31 of each year, which is the fiscal year end of Enumeral.
Following the Merger, the Company has continued Enumeral’s business of discovering and developing novel antibody immunotherapies that help the immune system fight cancer and other diseases. The Company utilizes a proprietary platform technology that facilitates the rapid high resolution measurement of immune cell function within small tissue biopsy samples. The Company’s initial focus is on the development of a pipeline of next generation monoclonal antibody drugs targeting established and novel immune-modulatory receptors.
The concept of stimulating the immune system to fight cancer was first advanced more than a century ago, but it is only recently that the field of immuno-oncology has seen clinical success, with marketing approvals being granted for antibodies that block CTLA-4 (Yervoy® (ipilimumab)) and PD-1 (Keytruda® (pembrolizumab) and Opdivo® (nivolumab)), and PD-L1 (Tecentriq® (atezolizumab)). Use of these drugs has established that durable anti-tumor responses can be elicited in some patients by blocking the checkpoints that normally suppress the human immune response against cancer cells. The success of these drugs suggests that immuno-oncology may fundamentally alter the course of cancer treatment.
| F-7 |
In the Company’s lead antibody program, the Company has characterized certain anti-PD-1 antibodies, or simply “PD-1 antibodies,” using patient biopsy samples, in an effort to identify next generation PD-1 antagonists with enhanced selectivity for the immune effector cells that carry out anti-tumor functions. The Company has identified two antagonist PD-1 antibodies that inhibit PD-1 activity in different ways. The distinction is that one of the antibodies (ENUM 388D4) blocks binding of the ligand PD-L1 to PD-1, while the other antibody (ENUM 244C8) does not inhibit PD-L1 binding. However, both display activity in various biological assays. In addition to the Company’s PD-1 antibody program, the Company is developing antibody drug candidates for a number of other immunomodulatory protein targets, including TIM-3, CD39, and TIGIT. The Company is also pursuing several antibody programs for which the Company has not yet publicly disclosed the targets.
The Company’s proprietary platform technology, exclusively licensed from the Massachusetts Institute of Technology, or MIT, is a microwell array technology that detects secreted molecules (such as antibodies and cytokines) and cell surface markers, at the level of single, live cells – and enables recovery of single, live cells of interest. The platform technology can be used to achieve at least three separate, but complementary, objectives. First, the Company uses the platform to rapidly produce antibody libraries with high diversity. Second, the platform has the potential to guide rational selection of lead candidates derived from these libraries, through characterization of immune function at the level of single cells from human biopsy samples. Third, it has the potential to identify patients more likely than others to benefit from treatment with a given therapeutic antibody. Thus, the Company’s platform is a multipurpose tool that is valuable for activities ranging from antibody discovery to target discovery to patient stratification in clinical development. The platform yields multidimensional, functional read-outs from single live cells, such as tumor infiltrating lymphocytes, or TILs, from human tumor biopsy samples, and it enables the Company to examine the responses of different classes of human immune cells to treatment with immuno-modulators in the context of human disease, as opposed to animal models of disease.
To date, the Company’s proof-of-concept corporate collaborations have provided minimal revenue. However, the Company’s business has not generated (nor does the Company anticipate that in the foreseeable future it will generate) the cash necessary to finance its operations. The Company expects to continue to incur losses and negative cash flows from operations for the foreseeable future, and will require additional capital to continue its operations beyond May 2017.
The Company continues to be a “smaller reporting company,” as defined under the Exchange Act, following the Merger. The Company believes that as a result of the Merger, it has ceased to be a “shell company” (as such term is defined in Rule 12b-2 under the Securities and Exchange Act of 1934, as amended (the “Exchange Act”)).
2 - GOING CONCERN
The Company’s consolidated financial statements have been prepared in conformity with generally accepted accounting principles in the United States which contemplate the Company’s continuation as a going concern. As of December 31, 2016, the Company had cash and cash equivalents of $3,162,400. The Company’s continuation as a going concern is dependent upon the Company attaining profitable operations, generating continued cash payments from partners under new or existing contracts and raising additional capital, through public or private equity offerings, debt financings, or strategic collaborations and licensing arrangements.
Since the Company’s inception in 2009, it has incurred significant net losses and negative cash flows from operations. As of December 31, 2016, the Company had an accumulated deficit of $26,265,207. The Company’s recurring losses and negative cash flows from operations raise substantial doubt about its ability to continue as a going concern, and as a result the Company’s independent registered public accounting firm included an explanatory paragraph in its report on the Company’s consolidated financial statements for the fiscal year ended December 31, 2016.
The Company’s liquidity is highly dependent on its ability to obtain additional capital in the near future. The Company’s failure to raise new capital would impair its ability to both continue its current collaborations and develop new collaborative partnerships and could result in its failure to continue to operate as a going concern. Substantial doubt about its ability to continue as a going concern may also create negative reactions to the price of the Company’s common stock, and the Company may not be able to obtain additional financing in the future.
| F-8 |
As of the date of this filing the Company believes it has sufficient liquidity to fund operations into May 2017. The Company is currently exploring a range of potential transactions, which may include public or private equity offerings, debt financings, collaborations and licensing arrangements, and/or other strategic alternatives, including a merger, sale of assets or other similar transactions. If the Company is unable to raise additional capital on terms acceptable to the Company and on a timely basis, the Company will be required to downsize or wind down its operations through liquidation, bankruptcy, or a sale of its assets. In addition, to the extent additional capital is raised through the sale of equity or convertible debt securities, such securities may be sold at a discount from the market price of the Company’s common stock. The issuance of these securities could also result in significant dilution to some or all of the Company’s stockholders, depending on the terms of the transaction.
The consolidated financial statements do not include any adjustments related to the recovery and classification of asset carrying amounts or the amount and classification of liabilities that might result should the Company be unable to continue as a going concern. The Company expects to incur significant expenses and operating losses for the foreseeable future, and the Company’s net losses may fluctuate significantly from quarter to quarter and from year to year. These factors raise substantial doubt about the Company’s ability to continue as a going concern.
3 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accompanying consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“GAAP”). Any reference in these notes to applicable guidance is meant to refer to the authoritative United States generally accepted accounting principles as found in the Accounting Standards Codifications (“ASC”) and Accounting Standards Updates (“ASU”) of the Financial Accounting Standards Board (“FASB”).
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and accompanying notes. On an ongoing basis, the Company’s management evaluates its estimates, which include, but are not limited to, estimates related to accruals, stock-based compensation expense, warrants to purchase securities, and reported amounts of revenue and expenses during the reported period. The Company bases its estimates on historical experience and other market-specific or other relevant assumptions that it believes to be reasonable under the circumstances. Actual results may differ from those estimates or assumptions.
Principles of Consolidation and Presentation
The consolidated financial statements include the accounts of the Company and its subsidiaries. In these consolidated financial statements, “subsidiaries” are companies that are wholly owned, the accounts of which are consolidated with those of the Company. Significant intercompany transactions and balances are eliminated in consolidation.
Segment Information
Operating segments are defined as components of an enterprise about which separate discrete information is available for evaluation by the chief operating decision maker, or decision-making group, in deciding how to allocate resources and in assessing performance. The Company views its operations and manages its business in one operating segment, which is the business of discovering and developing novel antibody immunotherapies that help the immune system fight cancer and other diseases. The Company operates in only one geographic segment.
| F-9 |
Cash and Cash Equivalents
The Company considers all highly liquid investments with maturities of 90 days or less from the purchase date to be cash equivalents. Cash and cash equivalents are held in depository and money market accounts and are reported at fair value.
Marketable Securities
Marketable securities consist of U.S. treasury securities with maturities of more than 90 days. The Company has determined the appropriate balance sheet classification of the securities as current since they are available for use in current operating activities, regardless of actual maturity dates, and are recorded on the balance sheet at fair value, with the unrealized gains and losses reported in accumulated other comprehensive income (loss), which is a separate component of stockholders’ equity. When securities are sold, the unrealized gains and losses are reclassified to net earnings.
Concentration of Credit Risk
The Company has no significant off-balance sheet concentrations of credit risk such as foreign currency exchange contracts, option contracts or other hedging arrangements. Financial instruments that subject the Company to credit risk consist primarily of cash and cash equivalents. The Company generally invests its cash in money market funds, U.S. Treasury securities and U.S. Agency securities that are subject to minimal credit and market risk. Management has established guidelines relative to credit ratings and maturities intended to safeguard principal balances and maintain liquidity. At times, the Company’s cash balances may exceed the current insured amounts under the Federal Deposit Insurance Corporation.
Fair Value of Financial Instruments
Fair value of financial instruments included in current assets and current liabilities are estimated to approximate their book values, due to the short maturity of such instruments. All debt is based on current rates at which the Company could borrow funds with similar remaining maturities and approximates fair value. The Company’s assets and liabilities that are measured at fair value on a recurring basis are measured in accordance with FASB’s Accounting Standards Codification (“ASC”) Topic 820, Fair Value Measurements and Disclosures, which establishes a three-level valuation hierarchy for measuring fair value and expands financial statement disclosures about fair value measurements. The valuation hierarchy is based upon the transparency of inputs to the valuation of an asset or liability as of the measurement date.
The three levels are defined as follows:
| · | Level 1: Inputs to the valuation methodology are quoted prices (unadjusted) for identical assets in active markets. |
| · | Level 2: Inputs to the valuation methodology included quoted prices for similar assets and liabilities in active markets, and inputs that are observable for the asset or liability, either directly or indirectly, for substantially the full term of the financial instrument. |
| · | Level 3: Inputs to the valuation methodology are unobservable and significant to the fair value measurement. |
The Company’s cash equivalents carried at fair value are primarily comprised of federal agency backed money market funds. The valuation of the Company’s derivative liabilities is discussed below and in Note 11. The following table presents information about the Company’s financial assets and liabilities measured at a fair value on a recurring basis as of December 31, 2016 and 2015:
| December 31, 2016 | Quoted Prices in Active Markets (Level 1) | Observable Inputs (Level 2) | Unobservable Inputs (Level 3) | |||||||||||||
| Assets | ||||||||||||||||
| Cash | $ | 1,137,633 | $ | 1,137,633 | $ | - | $ | - | ||||||||
| Money market funds, included in cash equivalents | $ | 2,024,767 | $ | 2,024,767 | $ | - | $ | - | ||||||||
| F-10 |
| December 31, 2015 | Quoted Prices in Active Markets (Level 1) | Observable Inputs (Level 2) | Unobservable Inputs (Level 3) | |||||||||||||
| Assets | ||||||||||||||||
| Cash | $ | 815,890 | $ | 815,890 | $ | - | $ | - | ||||||||
| Money market funds, included in cash equivalents | $ | 2,780,372 | $ | 2,780,372 | $ | - | $ | - | ||||||||
| Liabilities | ||||||||||||||||
| Derivative liabilities | $ | 2,138,091 | $ | - | $ | - | $ | 2,138,091 | ||||||||
The following table provides a roll forward of the fair value of the Company’s derivative liabilities, using Level 3 inputs:
| Balance as of December 31, 2015 | $ | 2,138,091 | ||
| Change in fair value | (1,608,474 | ) | ||
| Extinguishment of derivative liabilities | (529,617 | ) | ||
| Balance as of December 31, 2016 | $ | - |
Accounts Receivable and Allowance for Doubtful Accounts
Trade receivables are recorded at the invoiced amount. The Company maintains allowances for doubtful accounts, if needed, for estimated losses resulting from the inability of customers to make required payments. This allowance is based on specific customer account reviews and historical collections experience. There was no allowance for doubtful accounts as of December 31, 2016 or December 31, 2015.
Property and Equipment
Property and equipment are recorded at cost. Expenditures for maintenance and repairs are charged to expense as incurred, whereas major betterments are capitalized as additions to property and equipment. Depreciation is recorded using the straight-line method over the estimated useful lives of the assets as follows:
| Lab equipment | 3-5 years | |
| Computer equipment and software | 3 years | |
| Furniture | 3 years | |
| Leasehold improvements | Shorter of useful life or life of the lease |
Impairment of Long-Lived Assets
Long-lived assets are reviewed for impairment annually or whenever events or changes in circumstances indicate that the carrying amount of the asset may not be recoverable. When such events occur, the Company compares the carrying amounts of the assets to their undiscounted expected future cash flows. If this comparison indicated that there is impairment, the amount of the impairment is calculated as the difference between the carrying value and fair value. There have been no impairments recognized during the years ended December 31, 2016 and 2015, respectively.
| F-11 |
Revenue Recognition
Collaboration and License Revenue
Non-refundable license fees are recognized as revenue when the Company has a contractual right to receive such payment, the contract price is fixed or determinable, the collection of the resulting receivable is reasonably assured and the Company has no further performance obligations under the license agreement. Multiple element arrangements, such as license and development arrangements are analyzed to determine whether the deliverables, which often include license and performance obligations such as research and steering committee services, can be separated or whether they must be accounted for as a single unit of accounting in accordance with GAAP. The Company recognizes up-front license payments as revenue upon delivery of the license only if the license has stand-alone value and the fair value of the undelivered performance obligations, typically including research and/or steering committee services, can be determined. If the fair value of the undelivered performance obligations can be determined, such obligations would then be accounted for separately as performed. If the license is considered to either (i) not have stand-alone or (ii) have stand-alone value but the fair value of any of the undelivered performance obligations cannot be determined, the arrangement would then be accounted for as a single unit of accounting and the license payments and payments for performance obligations are recognized as revenue over the estimated period of when the performance obligations are performed.
Whenever the Company determines that an arrangement should be accounted for as a single unit of accounting, it must determine the period over which the performance obligations will be performed and revenue will be recognized. Revenue will be recognized using either a relative performance or straight-line method. The Company recognizes revenue using the relative performance method provided that the Company can reasonably estimate the level of effort required to complete its performance obligations under an arrangement and such performance obligations are provided on a best-efforts basis. Direct labor hours or full-time equivalents are typically used as a measure of performance. Revenue recognized under the relative performance method would be determined by multiplying the total payments under the contract, excluding royalties and payments contingent upon achievement of substantive milestones, by the ratio of level of effort incurred to date to estimated total level of effort required to complete the Company’s performance obligations under the arrangement. Revenue is limited to the lesser of the cumulative amount of payments received or the cumulative amount of revenue earned, as determined using the relative performance method, as of each reporting period.
If the Company cannot reasonably estimate the level of effort required to complete its performance obligations under an arrangement, but the Company can reasonably estimate when the performance obligation ceases or the remaining obligations become inconsequential and perfunctory, then the total payments under the arrangement (excluding royalties and payments contingent upon achievement of substantive milestones) would be recognized as revenue on a straight-line basis over the performance period.
In December 2014, the Company entered into a study agreement for a term of up to 24 months with Merck Sharp & Dohme Corp., or Merck (the “Merck Agreement”). In February 2016, the Company and Merck subsequently amended the work plan under the Merck Agreement to also include non-small cell lung cancer tissues. Pursuant to the Merck Agreement, the Company is conducting a specified research program using its platform technology to identify functional response of single cell types in colorectal cancer and non-small cell lung cancer in the presence or absence of immunomodulatory receptor modulators identified by Merck. In this collaboration, Merck has reimbursed the Company for the cost of performing the work plan set forth in the Merck Agreement, for up to a specified number of full-time employees at a pre-determined annual rate. In addition, Merck will make certain milestone payments to the Company upon the completion of specified objectives set forth in the Merck Agreement and related work plan. In September 2015, the Company announced the achievement of the first milestone under the Merck Agreement.
In January 2016, the Company and The University of Texas M.D. Anderson Cancer Center (“MDACC”) entered into a Collaborative Research and Development Agreement (the “MDACC Agreement”). Under the MDACC Agreement, the Company and MDACC plan to collaborate on the discovery and development of novel monoclonal antibodies against selected targets in immune-oncology, utilizing the Company’s antibody discovery and immune profiling platform and MDACC’s preclinical and development expertise and infrastructure. Pursuant to the terms of the MDACC Agreement, the Company and MDACC will share the costs of research and development activities necessary to take development candidates through successful completion of a Phase I clinical trial. The MDACC Agreement provides for a structure whereby the Company and MDACC are each granted the right to receive a percentage of the net income from product sales or any payments associated with licensing or otherwise partnering a program with a third party. To date, the Company has not yet commenced work under the MDACC agreement.
| F-12 |
In June 2016, the Company entered into a Definitive License and Transfer Agreement (the “Definitive Agreement”) with Pieris Pharmaceuticals, Inc. and Pieris Pharmaceuticals GmbH (collectively, “Pieris”). Pursuant to the terms and conditions of the Definitive Agreement, Pieris is licensing from the Company specified intellectual property related to the Company’s anti-PD-1 antibody program ENUM 388D4 for the potential development and commercialization by Pieris of novel multispecific therapeutic proteins comprising fusion proteins based on Pieris’ Anticalins® class of therapeutic proteins and the Company’s antibodies in the field of oncology. The Company had previously entered into a License and Transfer Agreement (the “License Agreement”) with Pieris in April 2016, which the Definitive Agreement superseded. Pieris paid the Company an upfront license fee in the amount of $250,000 in connection with execution of the License Agreement, and paid the Company a $750,000 license maintenance fee, to continue the licensing arrangements under the License Agreement.
Under the Definitive Agreement, the Company has granted Pieris an option until May 31, 2017 to license specified patent rights and know-how covering two additional undisclosed antibody programs on the same terms and conditions as for the Company’s 388D4 anti-PD-1 antibody (each, a “Subsequent Option”). Pieris may exercise the Subsequent Options separately and on different dates during the option period. Pieris will pay the Company additional license fees in the event that Pieris exercises one or both Subsequent Options.
Grant Revenue
In September 2014, the Company was awarded a Phase II Small Business Innovation Research contract from the National Cancer Institute (“NCI”) for up to $999,967 over two years. In September 2016, the Company entered to an amendment to its NCI contract to extend the period of performance under the contract to March 15, 2017. We do not anticipate any further amendments to the NCI contract. Grant revenue consists of a portion of the funds received to date from the NCI. Revenue is recognized as the related research services are performed in accordance with the terms of the agreement.
Research and Development Expenses
Research and development expenditures are charged to the consolidated statements of operations and comprehensive income (loss) as incurred. Research and development expenses are comprised of costs incurred in performing research and development activities, including salaries and benefits, facilities costs, clinical supply costs, contract services, depreciation and amortization expense and other related costs. Costs associated with acquired technology, in the form of upfront fees or milestone payments, are charged to research and development expense as incurred. Legal fees incurred in connection with patent applications, along with fees associated with the license to the Company’s core technology, are expensed as research and development expense.
Derivative Liabilities
The Company’s derivative liabilities related to (a) warrants to purchase an aggregate of 23,549,510 shares of our common stock that were issued in connection with the July 2014 PPO and (b) warrants to purchase 41,659 shares of Enumeral Series A Preferred Stock that were issued in December 2011 and June 2012 pursuant to Enumeral’s debt financing arrangement with Square 1 Bank that were subsequently converted into warrants to purchase 66,574 shares of the Company’s common stock in connection with the July 2014 Merger.
Due to the price protection provision included in the original warrant agreements, the warrants were deemed to be derivative liabilities and, therefore, the fair value of the warrants was recorded in the current liability section of the consolidated balance sheet. As such, the outstanding warrants, accounted for as derivative liabilities, were revalued each reporting period with the resulting gains and losses recorded as the change in fair value of derivative liabilities on the consolidated statement of operations. Upon exercise of the warrant or modification of the warrant agreements to remove the terms that required derivative liability treatment, the derivative liabilities are revalued on the date that such event occurs and the remaining value is reclassified to additional paid in capital. In connection with the Warrant Tender Offer, all derivative liabilities outstanding were extinguished and the Company applied the extinguishment of liabilities model to determine the loss on extinguishment of derivative liabilities. The consideration transferred to extinguish the derivative liabilities was recorded at fair value and compared to the cash received in connection with the tender offer to determine the loss on extinguishment of the derivative liabilities.
| F-13 |
As a result of the December 12, 2016 Warrant Tender Offer these warrants were no longer classified as derivative liabilities on the consolidated balance sheet. Additional detail regarding these warrants can be found in Note 11 below.
The Company used the Black-Scholes option-pricing model to estimate the fair values of the issued and outstanding warrants.
Comprehensive Income (Loss)
Other comprehensive income (loss) is comprised of unrealized holding gains and losses arising during the period on available-for-sale securities that are not other-than-temporarily impaired. The unrealized gains and losses are reported in accumulated other comprehensive income (loss), until the securities are sold or mature, at which time they are reclassified to earnings. The Company made no reclassifications out of accumulated other comprehensive loss to net income (loss) during the year ended December 31, 2016. The Company reclassified $19,097 out of accumulated other comprehensive loss to net income (loss) during the year ended December 31, 2015.
Stock-Based Compensation
The Company accounts for stock-based compensation awards to employees and directors in accordance with ASC Topic 718, Compensation-Stock Compensation (“ASC 718”). ASC 718 requires all stock-based payments to employees, including grants of employee stock options and restricted stock, to be recognized in the audited consolidated statements of operations and comprehensive income (loss) based on their grant date fair values. Compensation expense related to awards to employees is recognized on a straight-line basis over the associated service period of the award, which is generally the vesting term. The vesting of employee and director awards is generally time-based, but certain option awards for employees provide for vesting of all or a portion of the shares underlying such option upon the achievement of certain milestones or performance-based criteria. Share-based payments issued to non-employees are recorded at their fair values, and are periodically revalued as the equity instruments vest and are recognized as expense over the related service period in accordance with the provisions of ASC 718 and ASC Topic 505.
The Company estimates the fair value of its common stock options using the Black-Scholes option pricing model, which requires the input of subjective assumptions, including (a) the expected stock price volatility, (b) the expected term of the award, (c) the risk-free interest rate, (d) expected dividends, and (e) the estimated fair value of the Company’s common stock on the measurement date. As of January 1, 2016, the Company began using a blended average of its historical volatility and the historical volatility of a group of similarly situated companies to calculate the expected volatility when valuing the Company’s stock options. For purposes of calculating this blended volatility, the Company selected companies with comparable characteristics, including enterprise value, risk profiles, position within the industry, and with historical share price information sufficient to meet the expected term of the stock-based awards. The Company computes historical volatility data using the daily closing prices for the Company and the selected companies’ shares during the equivalent period of the calculated expected term of the stock-based awards. Prior to January 1, 2016, due to the lack of a public market for the trading of the Company’s common stock and a lack of company specific historical and implied volatility data, the Company had based its estimate of expected volatility only on the historical volatility of a group of similarly situated companies that were publicly traded. Due to the lack of specific historical option activity, the Company has primarily estimated the expected term of its employee stock options using the “simplified” method, whereby, the expected term equals the arithmetic average of the vesting term and the original contractual term of the option. The expected term for non-employee awards is the remaining contractual term of the option. The risk-free interest rates are based on the U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. The Company has never paid dividends, and does not expect to pay dividends in the foreseeable future.
The Company’s common stock is publicly traded, and fair market value is determined based on the closing sales price of the Company’s common stock on the OTCQB.
| F-14 |
Effective January 1, 2016, the Company has elected to account for forfeitures as they occur, as permitted by ASU No. 2016-09, Compensation-Stock Compensation (Topic 718), Improvements to Employee Share-Based Payment Accounting. Prior to the adoption of ASU No. 2016-09, the Company estimated the number stock-based awards that were expected to vest, and only recognized compensation expense for such awards. The estimation of stock awards that will ultimately vest required judgment, and to the extent actual results or updated estimates differed from current estimates, such amounts were recorded as a cumulative adjustment in the period estimates were revised. The Company considered many factors when estimating expected forfeitures, including type of awards granted, employee class, and historical experience.
Effective January 1, 2016, the Company elected to recognize forfeitures as they occur. The impact of that change in accounting policy has been recorded as an $8,333 cumulative effect adjustment to accumulated deficit, as of December 31, 2015.
During the year ended December 31, 2014, the Company engaged a third party to develop a binomial lattice model to estimate the fair value of options to purchase a total of 450,000 shares with vesting based on the future performance of a share of the Company’s common stock.
Earnings (Loss) Per Share
Basic earnings (loss) per common share amounts are based on the weighted average number of common shares outstanding. Diluted earnings per share amounts are based on the weighted average number of common shares outstanding, plus the incremental shares that would have been outstanding upon the assumed exercise of all potentially dilutive stock options, warrants and convertible debt, subject to anti-dilution limitations. All such potentially dilutive instruments were anti-dilutive as of December 31, 2016. As of December 31, 2016 and 2015, the number of shares underlying options and warrants that were anti-dilutive were approximately 30.4 million and 26.8 million, respectively.
Income Taxes
Income taxes are recorded in accordance with FASB ASC Topic 740, Income Taxes (“ACS 740”), which provides for deferred taxes using an asset and liability approach. Under this method, deferred tax assets and liabilities are determined based on the difference between the financial reporting and the tax reporting basis of assets and liabilities and are measured using the enacted tax rates and laws that are expected to be in effect when the differences are expected to reverse. The Company provides a valuation allowance against net deferred tax assets unless, based upon the available evidence, it is more likely than not that the deferred tax assets will be realized. The Company has evaluated available evidence and concluded that the Company may not realize the benefit of its deferred tax assets; therefore a valuation allowance has been established for the full amount of the deferred tax assets. The Company accounts for uncertain tax positions in accordance with the provisions of ASC 740. When uncertain tax positions exist, the Company recognizes the tax benefit of tax positions to the extent that the benefit will more likely than not be realized. The determination as to whether the tax benefit will more likely than not be realized is based upon the technical merits of the tax position as well as consideration of the available facts and circumstances.
The Company had no uncertain tax liabilities as of December 31, 2016 or December 31, 2015. The guidance requires the Company to determine whether it is more likely than not that a tax position will be sustained upon examination by the appropriate taxing authority. If a tax position meets the more likely than not recognition criteria, the guidance requires the tax position be measured at the largest amount of benefit greater than 50% likely of being realized upon ultimate settlement.
As of December 31, 2016, the Company had accumulated losses of $26,265,207 since inception and, therefore, has not paid any federal income taxes. Realization of deferred tax assets is dependent on future earnings, if any, the timing and amount of which are uncertain. Accordingly, valuation allowances in amounts equal to the deferred tax assets have been established to reflect these uncertainties. Utilization of the deferred tax asset, consisting of net operating loss and research and development credit carryforwards, may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986, as amended, due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of net operating loss and research and development credit carryforwards that can be utilized annually to offset future taxable income and tax, respectively.
| F-15 |
Recent Accounting Pronouncements
In May 2014, the FASB issued ASU No. 2014-09, Revenue from Contracts with Customers (Topic 606). ASU No. 2014-09 provides for a single comprehensive model for use in accounting for revenue arising from contracts with customers and supersedes most current revenue recognition guidance. The accounting standard is effective for interim and annual periods beginning after December 15, 2016 with no early adoption permitted. In August 2015, the FASB issued ASU No. 2015-14, Revenue from Contracts with Customers (Topic 606): Deferral of the Effective Date, which deferred the effective date of ASU No. 2014-09 to annual periods beginning after December 15, 2017, along with an option to permit early adoption as of the original effective date. The Company is required to adopt the amendments in ASU No. 2014-09 using one of two acceptable methods. In April 2016, the FASB issued ASU No. 2016-10, Revenue from Contracts with Customers (Topic 606): Identifying Performance Obligations and Licensing. The ASU clarifies the following two aspects of Topic 606: (a) identifying performance obligations; and (b) the licensing implementation guidance. The ASU does not change the core principle of the guidance in Topic 606. In May 2016, the FASB issued ASU No. 2016-12, Revenue from Contracts with Customers (Topic 606): Narrow-Scope Improvements and Practical Expedients, related to disclosures of remaining performance obligations, as well as other amendments to guidance on collectability, non-cash consideration and the presentation of sales and other similar taxes collected from customers. The new revenue standard allows for either full retrospective or modified retrospective application. The Company anticipates using the modified retrospective approach to implement this standard. The effective date and transition requirements for the ASUs are the same as the effective date and transition requirements in Topic 606. Public entities should apply the ASUs for annual reporting periods beginning after December 15, 2017, including interim reporting periods therein (i.e., January 1, 2018, for a calendar year entity). Early application for public entities is permitted only as of annual reporting periods beginning after December 15, 2016, including interim reporting periods within that reporting period. The Company does not expect to record a material adjustment upon adoption as the Company’s current contracts with customers will be completed prior to adoption of the new standard using the modified retrospective application, which requires a cumulative effect of initially applying the standard to opening accumulated deficit as of January 1, 2018. The Company does anticipate changes in its revenue recognition policies for revenue generating contracts the Company may enter into in the future.
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842). ASU No. 2016-02 is effective for annual periods beginning after December 15, 2018, and requires a lessee to recognize assets and liabilities for leases with a maximum possible term of more than 12 months. A lessee would recognize a liability to make lease payments (the lease liability) and a right-of-use asset representing its right to use the leased asset (the underlying asset) for the lease term. Early application is permitted. The Company is currently evaluating the impact the adoption of the accounting standard will have on its consolidated financial statements.
In November 2016, the FASB issued ASU No. 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash. ASU 2016-18 requires entities to reconcile and explain the period-over-period change in total cash, cash equivalents and restricted cash within its statements of cash flows. ASU 2016-18 is effective for fiscal years, and interim periods within, beginning after December 15, 2017. Early adoption is permitted. A reporting entity must apply the amendments in ASU 2016-18 using a full retrospective approach. The Company is currently evaluating the impact the adoption of the ASU will have on its consolidated financial statements.
Accounting Standards Adopted in the Period
In March 2016, the FASB issued ASU No. 2016-09, Compensation—Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting, which simplified several aspects of employee share-based payment accounting. In particular, the ASU No. 2016-09 permits entities to make an accounting policy election to either estimate forfeitures on share-based payment awards, as previously required, or to recognize forfeitures as they occur. Effective January 1, 2016, the Company elected to recognize forfeitures as they occur. The impact of that change in accounting policy has been recorded as an $8,333 cumulative effect adjustment to accumulated deficit, as of December 31, 2015. The Company recognized slightly higher share-based payment expense for 2016, relative to prior periods, as the effects of forfeitures were not to be recognized until they occurred, rather than being estimated at the time of grant and subsequently adjusted as and when necessary. The effects of adopting the remaining provisions in ASU No. 2016-09 affecting the income tax consequences of share-based payments, classification of awards as either equity or liabilities when an entity partially settles the award in cash in excess of the employer’s minimum statutory withholding requirements and classification in the consolidated statements of cash flows did not have any impact on the Company’s financial position, results of operations or cash flows.
| F-16 |
In April 2015, the FASB issued ASU No. 2015-03, Interest – Imputation of Interest (Subtopic 305-40): Simplifying the Presentation of Debt Issuance Costs. The new guidance requires the debt issuance costs related to a recognized debt liability be presented on the balance sheet as a direct deduction from the carrying amount of that debt liability. This guidance was effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2015. Accordingly, the standard was effective for the Company on January 1, 2016.
In August 2014, the FASB issued ASU No. 2014-15, Presentation of Financial Statements-Going Concern (Subtopic 205-40). ASU No. 2014-15 requires management to evaluate for the existence of conditions or events that raise substantial doubt about the entity’s ability to continue as a going concern and to provide related footnote disclosures. The standard provides guidance to management, with principles and definitions that are intended to reduce diversity in the timing and content of disclosures that companies commonly provide in their footnotes. Under the new standard, management must decide whether there are conditions or events, considered in the aggregate, that raise substantial doubt about the Company’s ability to continue as a going concern within one year after the issuance date of the Company’s financial statements. The accounting standard is effective for annual periods ending after December 15, 2016 and for interim periods thereafter. The Company adopted ASU No. 2014-15 in the fourth quarter of 2016.
Other accounting standards that have been issued by the FASB or other standards-setting bodies that do not require adoption until a future date are not expected to have a material impact on the Company’s consolidated financial statements.
4 - PROPERTY AND EQUIPMENT, NET
Property and equipment, net consist of the following:
| December 31, | ||||||||
| 2016 | 2015 | |||||||
| Laboratory equipment | $ | 2,398,685 | $ | 2,559,986 | ||||
| Computer/office equipment and software | 115,885 | 187,337 | ||||||
| Furniture, fixtures and office equipment | 73,734 | 73,734 | ||||||
| Leasehold improvements | 75,262 | 75,262 | ||||||
| 2,663,566 | 2,896,319 | |||||||
| Less - Accumulated depreciation and amortization | (1,761,469 | ) | (1,384,826 | ) | ||||
| Total property and equipment, net | $ | 902,097 | $ | 1,511,493 | ||||
The Company recognized depreciation and amortization expense of $609,396 and $616,523 for the years ended December 31, 2016 and 2015, respectively. During the year ended December 31, 2016, the Company retired property and equipment with a gross cost of $232,753 which had no remaining carrying value. During the year ended December 31, 2015, the Company retired leasehold improvements with a gross cost of $112,507 and expensed the remaining net carrying value of $22,962 associated with the write-down of leasehold improvements due to a relocation of the Company’s corporate office and research laboratories in March 2015 (see Note 8).
| F-17 |
5 - RESTRICTED CASH
The Company held restricted cash of $534,780 as of December 31, 2016 and December 31, 2015, respectively. The balances are primarily held on deposit with a bank to collateralize a standby letter of credit in the name of the Company’s facility lessor in accordance with the Company’s facility lease agreement.
6 - ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
The Company’s accrued expenses and other current liabilities consist of the following:
| As of December 31, | ||||||||
| 2016 | 2015 | |||||||
| Accrued wages and benefits | $ | 222,141 | $ | 447,769 | ||||
| Accrued professional fees | 130,350 | 213,475 | ||||||
| Accrued other | 33,864 | 53,140 | ||||||
| Total accrued expenses and other current liabilities | $ | 386,355 | $ | 714,384 | ||||
7 - DEBT
Promissory Notes
On July 29, 2016, the Company entered into a Subscription Agreement (the “Subscription Agreement”) with certain accredited investors (the “Buyers”), pursuant to which the Buyers purchased the Company’s 12% Senior Secured Promissory Notes (the “Notes”) in the aggregate principal amount of $3,038,256 (before deducting placement agent fees and expenses of $385,337), which includes $38,256 pursuant to an over-allotment option (the “Note Offering”). The Company incurred additional legal fees of $122,512 associated with the Notes. The Company recorded the Notes net of loan issuance fees of $507,849, which were accreted to interest expense over the term of the loan using the effective interest method.
The Notes had an aggregate principal balance of $3,038,256 and a stated maturity date of 12 months from the date of issuance. The principal on the Notes bore interest at a rate of 12% per annum, payable monthly commencing on September 1, 2016. Interest was payable in shares (the “Repayment Shares”) of the Company’s common stock; provided, however, that interest would not be calculated or accrued in a manner that triggered an anti-dilution adjustment on the PPO Warrants. In the event that on an interest payment date, the PPO Warrants’ anti-dilution provision would be triggered by the payment of interest in shares of the Company’s common stock, interest payments on the Notes could be paid in cash. The Notes ranked senior to all of the Company’s existing indebtedness, except as otherwise set forth in the Notes.
The maturity date of the outstanding principal amount of the Notes, together with accrued and unpaid interest due thereon, would accelerate to the date (on or after September 1, 2016) on which the Company completed and closed certain financing transactions that achieved minimum thresholds, as specified in the Notes. In such specified transactions, the Notes would convert at a valuation per share equal to 50% of the price per share of securities sold in that financing transaction. In addition, in the event of a sale of the Company during the term of the Notes, Note Holders would have been entitled to receive 1.5x of the principal amount of the Notes plus accrued interest, paid in either cash or securities of acquiring entity at the acquiring entity’s discretion.
The Company’s obligations under the Notes were secured, pursuant to the terms of an Intellectual Property Security Agreement (the “Security Agreement”), dated as of the July 29, 2016, among the Company, Enumeral, the Buyers and the collateral agent for the Buyers named therein, by a first priority security interest in all now owned or hereafter acquired intellectual property of the Company and Enumeral, except to the extent such intellectual property cannot be assigned or the creation of a security interest would be prohibited by applicable law or contract.
Due to the Warrant Tender Offer (as discussed below in Note 10), the Notes were converted into 48,806,545 shares of the Company’s common stock. Through the date of the Warrant Tender offer, the Company had accreted $84,642 of the loan issuance fees to interest expense. As a result, the Company expensed the remaining loan issuance fees of $423,207 to interest expense for the year ended December 31, 2016. During the term of the Note, the Company incurred interest expense of $136,722, of which $124,569 was paid in cash to the Note Holders and $12,153 was accrued and converted into shares of the Company’s common stock issued to the Note Holders.
In addition, pursuant to the placement agent agreement for the Notes, the placement agent received warrants exercisable for a period of ten years to purchase shares of the Company’s common stock, equal to 10% percent of the number of shares issued upon the conversion of the Notes, resulting in warrants to purchase 4,880,655 shares of the Company’s common stock issued to the placement agent. As the conversion of the Notes was an integrated transaction that occurred due to the completion of the Warrant Tender Offer, the Company included the placement agent warrants issued as part of the loss on extinguishment of derivative liabilities.
| F-18 |
The Notes contained a contingent beneficial conversion feature as they were convertible upon the occurrence of certain equity financings. Upon the completion of the Warrant Tender Offer, the contingency was resolved and the Company recognized $4,253,558 of interest expense based on the intrinsic value of the beneficial conversion feature.
Equipment Lease Financing
In December 2015, the Company and Fountain Leasing 2013 LP (“Fountain”) entered into a master lease agreement and related transaction documents, pursuant to which Fountain provided the Company with $506,944 for the purchase of research and development lab equipment (the “Fountain Lease”). Fountain’s security under the Fountain Lease is the equipment purchased and a security deposit in the amount of $101,389. The initial term of the Fountain Lease is 36 months, with payments of $21,545 per month for the first 24 months and then $1,267 for the 12 months thereafter. Pursuant to the terms of the Fountain Lease, the Company has an option at the end of the initial term to purchase the equipment for the greater of $25,347 or current fair market value, provided that such amount shall not be in excess of $152,083. In addition, the Company also has the option to extend the Fountain Lease for an additional 12 month period at a rate of $8,872 per month with the right at the end of such extension term to purchase the equipment for fair value or to return the equipment to Fountain. The Fountain Lease has a lease rate factor of 4.25% per month for the first 24 months and 0.25% per month for the final 12 months of the initial term.
The Company has recorded current equipment lease financing of $251,631 and long-term equipment lease financing of $14,840 as of December 31, 2016. The equipment has been included in property and equipment on the Company’s consolidated balance sheets.
Future principal payments on the equipment lease financing are as follows:
| For the twelve months ended December 31, | Amount | |||
| 2017 | $ | 258,542 | ||
| 2018 | 15,208 | |||
| Total equipment lease financing payments | $ | 273,750 | ||
| As of December 31, 2016 | Amount | |||
| Current equipment lease financing payments | $ | 258,542 | ||
| Less: Amount representing interest | (6,911 | ) | ||
| Current equipment lease financing, net | $ | 251,631 | ||
| Long-term equipment lease financing payments | $ | 15,208 | ||
| Less: Amount representing interest | (368 | ) | ||
| Long-term equipment lease financing, net | $ | 14,840 | ||
8 - COMMITMENTS
Operating Leases
In March 2015, the Company relocated its offices and research laboratories to 200 CambridgePark Drive in Cambridge, Massachusetts. The Company is leasing 16,825 square feet at this facility pursuant to Indenture of Lease (the “CambridgePark Lease”) that the Company entered into in November 2014. The term of the CambridgePark Lease is for five years, and the initial base rent is $42.50 per square foot, or approximately $715,062 on an annual basis. The base rent will increase incrementally over the term of the CambridgePark Lease, reaching approximately $804,739 on an annual basis in the fifth year of the term. In addition, the Company is obligated to pay a proportionate share of the operating expenses and applicable taxes associated with the premises, as calculated pursuant to the terms of the CambridgePark Lease. Pursuant to the terms of the CambridgePark Lease, the Company has delivered a security deposit to the landlord in the amount of $529,699, which may be reduced to $411,988 following the second anniversary of the commencement date, provided that the Company meets certain financial conditions set forth in the CambridgePark Lease. The Company has recorded deferred rent in connection with the CambridgePark Lease of $63,116 and $36,847 as of December 31, 2016 and 2015, respectively. This amount has been recorded as a long-term liability on the Company’s consolidated balance sheets.
| F-19 |
The Company previously occupied offices and research laboratories in approximately 4,782 square feet of space at One Kendall Square in Cambridge, Massachusetts, at an annual rent of $248,664 (the “Kendall Lease”). For the year ended December 31, 2015, the Company recorded an expense of $55,352, representing all exit costs associated with its move to new offices and research laboratories in March 2015. In June 2015, Enumeral entered into a lease termination agreement with the landlord for Enumeral’s former facility at One Kendall Square, pursuant to which the Kendall Lease was terminated as of June 17, 2015. In accordance with the terms of the lease termination agreement, Enumeral is not obligated to pay rent for the One Kendall Square facility after May 31, 2015. Enumeral had maintained a security deposit relating to the facility, recorded as restricted cash on the consolidated balance sheet. This security deposit was returned to Enumeral pursuant to the lease termination agreement.
In addition, the Company maintained a small corporate office at 1370 Broadway in New York, New York, at an annual rent of $23,100. The lease for the Company’s New York office expired on December 31, 2016.
Rent expense was $1,211,567 and $1,079,753 for the years ended December 31, 2016 and 2015, respectively.
Future operating lease commitments as of December 31, 2016 are as follows:
| Years Ending December 31, | Amount | |||
| 2017 | 754,966 | |||
| 2018 | 777,568 | |||
| 2019 | 800,842 | |||
| Thereafter | 134,123 | |||
| $ | 2,467,499 | |||
Employment Agreements
The Company has employment letter agreements with members of management which contain minimum annual salaries and, in certain cases, severance benefits. In conjunction with the closing of the Note Offering, Arthur H. Tinkelenberg, Ph.D. was terminated by the Company from his position as President and Chief Executive Officer, effective July 28, 2016. In connection with Dr. Tinkelenberg’s termination, the Company entered into a separation letter agreement with Dr. Tinkelenberg, dated August 4, 2016 (the “Tinkelenberg Separation Agreement”). Pursuant to the terms of the Tinkelenberg Separation Agreement, all of Dr. Tinkelenberg’s options to purchase shares of the Company’s common stock and restricted stock grants that were unvested as of Dr. Tinkelenberg’s separation date shall continue to vest during Dr. Tinkelenberg’s 12-month severance period pursuant to the vesting schedules set forth in such option. Absent a breach of the Tinkelenberg Separation Agreement by Dr. Tinkelenberg during the severance period, all remaining unvested options and shares of restricted stock shall fully vest and become exercisable on the expiration of the severance period, and the period for exercising all such options to purchase shares of the Company’s common stock shall be extended to the date that is five years following the expiration of the severance period. As a result of the modification to Dr. Tinkelenberg’s options, the Company incurred a one-time stock-based compensation charge of $15,580 during the year ended December 31, 2016. During the year ended December 31, 2016, the Company recorded charges related to severance and benefits owed to Dr. Tinkelenberg as a result of his termination. Dr. Tinkelenberg resigned as a director of the Company on September 19, 2016.
On September 21, 2016, John J. Rydzewski resigned as Executive Chairman of the Company, and also resigned from the Board of Directors. In connection with Mr. Rydzewski’s resignation, the Company entered into a separation letter agreement with Mr. Rydzewski, dated September 21, 2016. As part of Mr. Rydzewski’s separation letter agreement, 703,326 options became fully vested and the Company incurred a one-time stock-based compensation charge of $83,361 during the year ended December 31, 2016. In addition, the terms of Mr. Rydzewski’s separation letter agreement provide that the Company will continue to pay 100% of the cost of Mr. Rydzewski’s continuation of health and dental benefits through COBRA, until the earlier of 18 months from Mr. Rydzewski’s separation date from the Company or such time as Mr. Rydzewski becomes eligible for similar benefits from another employer.
Effective as of September 21, 2016 the Company appointed Wael Fayad to serve as President and Chief Executive Officer of the Company, and, in connection therewith, appointed Mr. Fayad as a director of the Company and Chairman of the Board of Directors. In connection with Mr. Fayad’s appointment, the Board of Directors designated Mr. Fayad as the Company’s “Principal Executive Officer” for U.S. Securities and Exchange Commission reporting purposes, effective as of September 21, 2016.
| F-20 |
In addition, the Company entered into an offer letter with Mr. Fayad, dated September 21, 2016 (the “Letter Agreement”), which sets forth the terms pursuant to which Mr. Fayad shall serve as the Company’s Chairman of the Board, President and Chief Executive Officer. The Letter Agreement provides that Mr. Fayad will receive a base salary at the rate of $325,000 per annum. Mr. Fayad will also be eligible to earn a target bonus of up to 50% of the base salary, payable in cash, based upon achievement of corporate objectives, individual objectives, and the Company’s finances, all as determined and at the discretion of the independent members of the Board or the Board’s Compensation Committee. Mr. Fayad was granted 2,600,000 options to purchase shares of the Company’s common stock in connection with the Letter Agreement. The Company granted Mr. Fayad 850,000 options under the Company’s 2014 Equity Incentive Plan (the “2014 Plan”) and 1,750,000 options outside of the 2014 Plan. Of these, 100,000 options vested immediately upon grant, and the remaining 2,500,000 options vest upon achievement of certain performance-based milestones.
9 - LICENSE AGREEMENT AND RELATED-PARTY TRANSACTIONS
License Agreement
In April 2011, Enumeral licensed certain intellectual property from the Massachusetts Institute of Technology (“MIT”), then a related party (as one of Enumeral’s scientific co-founders was an employee of MIT), pursuant to an Exclusive License Agreement (the “License Agreement”), in exchange for the payment of upfront license fees and a commitment to pay annual license fees, patent costs, milestone payments, royalties on sublicense income and, upon product commercialization, royalties on the sales of products covered by the licenses or income from corporate partners, and the issuance of 66,303 shares of Enumeral common stock. This intellectual property portfolio includes patents owned by Harvard University or co-owned by MIT and The Whitehead Institute, or MIT and Massachusetts General Hospital.
In March 2013, Enumeral and MIT entered into a first amendment to the License Agreement to clarify how equity issuances were to be made thereunder. In July 2014, Enumeral and MIT entered into a second amendment to the License Agreement, pursuant to which MIT’s participation rights and anti-dilution rights under the license agreement were terminated. In April 2015, Enumeral and MIT entered into a third amendment to the License Agreement, which revised the timetable for Enumeral to complete certain diligence obligations relating to the initiation of clinical studies in support of obtaining regulatory approval of a Diagnostic Product (as such term is defined in the License Agreement), as well as the timetable by which Enumeral is required to make the first commercial sale of a Diagnostic Product. In April 2016, Enumeral and MIT entered into a fourth amendment to the License Agreement, which revised the timetable for Enumeral to complete certain diligence obligations relating to the establishment of sublicenses and/or corporate partner agreements for the development of Licensed Products and/or Diagnostic Products, as well as the timetable by which an Investigational New Drug Application shall be filed on a Therapeutic Product (as such terms are defined in the License Agreement).
All amounts paid related to the license fees have been expensed as research and development by Enumeral as incurred. The Company incurred $40,000 and $30,000 in the years ended December 31, 2016 and 2015, respectively.
In addition to potential future royalty and milestone payments that Enumeral may have to pay MIT per the terms of the License Agreement, Enumeral is obligated to pay an annual fee $50,000 every year unless the License Agreement is terminated. During the year ended December 31, 2016, the Company recorded expense of $100,000 related to a portion of third party license payments owed to MIT pursuant to the terms of the License Agreement. During the year ended December 31, 2015, the Company achieved the first milestone in the Merck Agreement and paid MIT the required percentage of the milestone payment pursuant to the terms of the License Agreement. No royalty payments have been payable as Enumeral has not commercialized any products as set forth in the License Agreement. The Company reimburses costs to MIT and Harvard University for the continued prosecution of the licensed patent estate. For the years ended December 31, 2016 and 2015, the Company reimbursed $183,359 and $325,945 to MIT and $21,042 and $23,637 to Harvard, respectively. As of December 31, 2016 and 2015, the Company had accounts payable and accrued expenses of $43,053 and $168,726, respectively, associated with the reimbursement of costs to MIT and Harvard.
| F-21 |
Consulting Agreements
In September 2014, the Company and Dr. Barry Buckland entered into a Scientific Advisory Board Agreement (the “SAB Agreement”), which replaced Dr. Buckland’s previous consulting agreement and pursuant to which Dr. Buckland serves as chairman of the Company’s Scientific Advisory Board. The SAB Agreement had a term of two years. Pursuant to the terms of the SAB Agreement, Dr. Buckland will receive compensation on an hourly or per diem basis, either in cash or, at Dr. Buckland’s election, in options to purchase the Company’s common stock. The SAB Agreement limits the total amount of compensation payable to Dr. Buckland at $100,000 over any rolling 12-month period. In September 2016, the Company and Dr. Buckland entered into an amendment to the SAB Agreement to extend the term of the agreement an additional year. During the year ended December 31, 2016 and 2015, the Company recognized $12,000 and $32,000 of expense related to the SAB Agreement, respectively.
Warrant Tender Offer
Pursuant to the Warrant Tender Offer, the following executive officers of the Company elected to participate and exercise all or a portion of the warrants held by such person and received four shares for each Warrant tendered, in accordance with the terms of the Warrant Tender Offer: Mr. Sekhri exercised 43,750 warrants with an aggregate exercise price of $21,875 and received 175,000 shares of Common Stock; Mr. Sarney exercised 8,750 warrants with an aggregate exercise price of $4,375 and received 35,000 shares of Common Stock; Mr. Rothstein exercised 50,000 warrants with an aggregate exercise price of $25,000 and received 200,000 shares of Common Stock; Mr. Easton exercised 120,000 warrants with an aggregate exercise price of $60,000 and received 480,000 shares of Common Stock; Mr. Fayad exercised 74,500 warrants with an aggregate exercise price of $37,250 and received 298,000 shares of Common Stock; Mr. Buckland exercised 13,000 warrants with an aggregate exercise price of $6,500 and received 52,000 shares of Common Stock; and Mr. Van Nostrand exercised 39,000 warrants with an aggregate exercise price of $19,500 and received 156,000 shares of Common Stock.
10 - EQUITY
Merger
On July 31, 2014, Enumeral entered into the Merger Agreement, pursuant to which Enumeral became a wholly owned subsidiary of the Company. The Company’s authorized capital stock currently consists of 300,000,000 shares of common stock, par value $0.001, and 10,000,000 shares of “blank check” preferred stock, par value $0.001.
As a result of the Merger, all issued and outstanding common and preferred shares of Enumeral were exchanged for common shares of the Company as follows: (a) each share of Enumeral’s common stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.102121 shares of the Company’s common stock for a total of 4,940,744 shares post-Merger, (b) each share of Enumeral’s Series A Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.598075 shares of the Company’s common stock for a total of 4,421,744 shares post-Merger, (c) each share of Enumeral’s Series A-1 Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.790947 shares of the Company’s common stock for a total of 3,666,428 shares post-Merger, (d) each share of Enumeral’s Series A-2 Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 1.997594 shares of the Company’s common stock for a total of 3,663,177 shares post-Merger, (e) each share of Enumeral’s Series B Preferred Stock issued and outstanding immediately prior to the closing of the Merger was converted into 2.927509 shares of the Company’s common stock for a total of 2,777,687 shares post-Merger and (f) a convertible note and accrued interest was converted into 3,230,869 shares of the Company’s common stock post-Merger.
As a result of the Merger and the Split-Off, the Company discontinued its pre-Merger business and acquired the business of Enumeral, and has continued the existing business operations of Enumeral as a publicly-traded company under the name Enumeral Biomedical Holdings, Inc. In accordance with “reverse merger” accounting treatment, historical financial statements for Enumeral Biomedical Holdings Inc. as of period ends, and for periods ended, prior to the Merger have been replaced with the historical financial statements of Enumeral prior to the Merger.
Private Placement
On July 31, 2014, the Company closed a private placement offering (the “PPO”) of 21,549,510 Units of securities, at a purchase price of $1.00 per Unit, each Unit consisting of one share of the Company’s common stock and a warrant to purchase one share of the Company’s common stock at an exercise price of $2.00 per share with a term of five years (the “PPO Warrants”). The net proceeds received from the PPO were $18,255,444.
The investors in the PPO had anti-dilution protection on the shares of common stock included in the Units purchased in the PPO in the event that within two years after the closing of the PPO the Company issued common stock or securities convertible into or exercisable for shares of the Company’s common stock at a price lower than the Unit purchase price. The anti-dilution protection expired on July 31, 2016, without being triggered.
| F-22 |
In addition, the PPO Warrants had anti-dilution protection in the event that prior to the warrant expiration date the Company issued common stock or securities convertible into or exercisable for shares of the Company’s common stock at a price lower than the warrant exercise price, subject to exceptions for certain issuances. The PPO Warrants were amended in connection with the Warrant Tender Offer to remove the anti-dilution provisions.
In connection with the PPO, the Company paid its placement agents, EDI Financial, Inc. and Katalyst Securities LLC (the “Placement Agents”), a commission equal to 10% of the gross proceeds raised from investors in the PPO. In addition, the Placement Agents collectively received warrants to purchase 10% of the number of shares of Common Stock included in the Units sold in the PPO, with a term of five (5) years and an exercise price of $1.00 per share (the “PPO Agent Warrants”); provided, however, that the Placement Agents were not entitled to any warrants on the sale of Units in excess of 20,000,000. The Company also agreed to pay to the Placement Agents a cash fee on the amount that any person or entity contacted by the Placement Agents, in connection with the Offering, invested in the Company at any time prior to the date that was eighteen (18) months after the closing of the PPO. No such fee was earned or paid.
As a result of the foregoing, the Placement Agents and their respective sub-agents were collectively paid an aggregate commission of $2,154,951 and were issued PPO Agent Warrants to purchase an aggregate of 2,000,000 shares of the Company’s common stock. The Company was also required to reimburse the Placement Agents up to $30,000 of legal expenses incurred in connection with the PPO, in the aggregate. The PPO Agent Warrants were amended in connection with the Warrant Tender Offer on December 12, 2016 to remove the anti-dilution protection provisions and to reduce the exercise price of such PPO Agent Warrants from $1.00 to $0.125 per share.
The Company incurred approximately $500,000 of expenses in connection with the offering outside of the placement agent commissions and issued the subagent to one of the placement agents 150,000 shares of the Company’s common stock.
In addition, the Merger Agreement provided certain anti-dilution protection to the holders of the Company’s common stock immediately prior to the Merger (after giving effect to the Split-Off), in the event that the aggregate number of units sold in the PPO after the final closing thereof were to exceed 15,000,000. Accordingly, based on the final amount of gross proceeds raised in the PPO, the Company issued 1,690,658 additional shares of the Company’s common stock to holders of the Company’s common stock immediately prior to the Merger.
Warrant Tender Offer
On December 12, 2016, the Company consummated its Warrant Tender Offer to amend and exercise the outstanding PPO Warrants to purchase an aggregate of 21,549,510 shares of the Company’s common stock. The PPO Warrants of holders who elected to participate in the Warrant Tender Offer were amended to (i) receive four shares of common stock for each warrant exercised rather than one, (ii) reduce the exercise price to $0.50 per warrant in cash (or $0.125 per share); (iii) shorten the exercise period so that it expired concurrently with the expiration of the Warrant Tender Offer at 5:00 p.m. (Eastern Time) on December 9, 2016, and (iv) delete any price-based anti-dilution provisions. Pursuant to the Warrant Tender Offer, an aggregate of 6,863,000 PPO Warrants were tendered by their holders and were amended and exercised in connection therewith for gross proceeds to the Company of $3,431,500. As a result, the Company issued 27,452,000 shares of its common stock to the holders who amended and exercised their PPO Warrants in the Warrant Tender Offer. This resulted in net cash of $2,947,283 after deducting transaction fees of $484,217, of which $254,520 was the Warrant Agent Fee. Per the Warrant Agent Agreement the Company was to pay the warrant agent 8% of the gross proceeds, exclusive of proceeds from the Company's officers and directors. The remaining warrants of holders who elected not to exercise were amended to remove the price-based anti-dilution provisions, thus extinguishing the associated derivative liabilities. In connection with the Warrant Tender Offer, the Company applied the liability extinguishment model to determine the loss on extinguishment of derivative liabilities.
| F-23 |
As a result of the Warrant Tender Offer, the Notes and the associated accrued interest converted into shares of the Company’s common stock per the Note agreement. The Company issued a total of 48,806,545 shares of the Company’s common stock to the Note Holders. Through the date of the Warrant Tender offer, the Company had accreted $84,642 of the loan issuance fees to interest expense. When the Notes converted, in concurrence with the Warrant Tender Offer, the Company expensed the remaining loan issuance fees of $423,207 to interest expense. During the term of the Note the Company incurred interest expense of $136,722, of which $124,569 was paid in cash to the Note Holders and $12,153 was accrued and converted into shares of the Company’s common stock issued to the Note Holders. The Notes contained a contingent beneficial conversion feature as they were convertible upon the occurrence of certain equity financings. Upon the completion of the Warrant Tender Offer, the contingency was resolved and the Company recognized $4,253,558 of interest expense based on the intrinsic value of the beneficial conversion feature. In addition, pursuant to the placement agent agreement for the Notes, the placement agent received warrants exercisable for a period of ten years to purchase shares of the Company’s common stock, equal to 10% percent of the number of shares issued upon the conversion of the Notes, resulting in warrants to purchase 4,880,665 shares of the Company’s common stock issued to the placement agent. As the conversion of the Notes was an integrated transaction that occurred due to the completion of the Warrant Tender Offer, the Company included the placement agent warrants issued as part of the loss on extinguishment of derivative liabilities discussed above. Refer to Note 11 below for further information regarding the placement agent warrants.
On December 12, 2016, in connection with the consummation of the Warrant Tender Offer, the Company and holders of a majority of the outstanding PPO Agent Warrants approved an amendment to remove the price-based anti-dilution provisions in the PPO Agent Warrants and to reduce the exercise price of the PPO Agent Warrants from $1.00 per share to $0.125 per share. The Company recorded a loss on extinguishment of derivative liabilities of $187,240 as a result of this amendment. None of the PPO Agent Warrants were exercised in connection with the Warrant Tender Offer, and all PPO Agent Warrants remain outstanding as of December 31, 2016. Refer to Note 11 below for further details.
On December 12, 2016, the Company and Square 1 Bank entered into an amendment in regards to the warrants issued by the Company to Square 1 on July 31, 2014 to change the expiration date to December 12, 2016, reduce the exercise price to $0.125 per share, and remove any anti-dilution provisions. Immediately following this amendment, Square 1 cashless exercised these warrants, and the Company issued 11,096 shares of its common stock. Refer to Note 11 below for further details.
The consideration transferred to extinguish the derivative liabilities was recorded at fair value and compared to the cash received in connection with the tender offer to determine the loss on extinguishment of the derivative liabilities. Included in the consideration transferred was the following: (i) the fair value of common shares issued in the Warrant Tender Offer, (ii) the expense associated with the issuance of agent warrants of $704,848, and (iii) the expense related to the modification of the PPO Agent Warrants exercise price from $1.00 a share to $0.125 a share of $187,239. See Note 11 below for additional discussion.
11 - STOCK OPTIONS, RESTRICTED STOCK AND WARRANTS
Stock Options
In December 2009, Enumeral adopted the 2009 Stock Incentive Plan (the “2009 Plan”). The 2009 Plan was terminated in July 2014 in connection with the Merger.
On July 31, 2014, the Company’s Board of Directors adopted, and the Company’s stockholders approved, the 2014 Equity Incentive Plan (the “2014 Plan”), which reserves a total of 8,100,000 shares of the Company’s common stock for incentive awards. In connection with the Merger, options to purchase 948,567 shares of Enumeral common stock previously granted under the 2009 Plan were converted into options to purchase 1,045,419 shares of the Company’s common stock under the 2014 Plan.
Generally, shares that are expired, terminated, surrendered or cancelled without having been fully exercised will be available for future awards. In addition, shares of common stock that are tendered to the Company by a participant to exercise an award are added to the number of shares of common stock available for the grant of awards.
| F-24 |
As of December 31, 2016, there were 1,264,018 shares available for issuance under the 2014 Plan to eligible employees, non-employee directors and consultants. This number is subject to adjustment in the event of a stock split, reverse stock split, stock dividend, or other change in the Company’s capitalization.
During the years ended December 31, 2016 and 2015, there were 4,191,182 and 3,444,000 stock options granted to employees, directors or consultants with weighted-average grant date fair values, using the Black-Scholes pricing model, of $0.16 and $0.31, respectively.
Pursuant to the terms of the Tinkelenberg Separation Agreement, all of Dr. Tinkelenberg’s options to purchase shares of the Company’s common stock and restricted stock grants that were unvested as of Dr. Tinkelenberg’s separation date shall continue to vest during Dr. Tinkelenberg’s 12-month severance period pursuant to the vesting schedules set forth in such option. As a result of Dr. Tinkelenberg’s separation from the Company, the Company incurred a one-time stock-based compensation charge of $15,580 during the year ended December 31, 2016 for the remaining shares the Company expects to vest.
In September 2016, Mr. Rydzewski resigned as Executive Chairman and director of the Company. In connection with his resignation, all of Mr. Rydzewski’s unvested options became fully vested. As a result, 703,326 options became fully vested and the Company incurred a one-time stock-based compensation charge of $83,361 during the year ended December 31, 2016.
In connection with Mr. Fayad’s appointment as the Company’s Chairman, Chief Executive Officer and President in September 2016, the Company granted Mr. Fayad 1,750,000 options to purchase Common Stock outside of the 2014 Plan, and 850,000 options to purchase Common Stock under the 2014 Plan.
The Company estimates the fair value of each stock award on the grant date using the Black-Scholes option-pricing model based on the following assumptions and the assumptions regarding the fair value of the underlying common stock on each measurement date:
| Year ended December 31, | ||||
| 2016 | 2015 | |||
| Expected Volatility | 116.0%-117.0% | 104.1%-136.0% | ||
| Risk-free interest rate | 1.20%-2.05% | 1.60%-2.22% | ||
| Expected term (in years) | 5.00 | 6.00-9.59 | ||
| Expected dividend yield | 0% | 0% | ||
Stock-based compensation expense for stock options was $852,775 and $725,799 for the years ended December 31, 2016 and 2015, respectively. The Company had an aggregate of $302,494 of unrecognized stock-based compensation expense for stock options as of December 31, 2016 to be amortized over a weighted average period of 1.5 years which excludes $385,275 of unrecognized stock-based compensation expense of certain awards that vest upon performance-based criteria which are not considered probable to occur as of December 31, 2016.
A summary of stock option activity for the year ended December 31, 2016, which includes stock options granted under the 2014 Plan as well as outside of the 2014 Plan, is as follows:
| Weighted- | ||||||||||||||||
| Weighted- | Average | |||||||||||||||
| Average | Remaining | Aggregate | ||||||||||||||
| Exercise | Contractual | Intrinsic | ||||||||||||||
| Shares | Price | Term (years) | Value | |||||||||||||
| Outstanding at December 31, 2015 | 5,926,654 | $ | 0.62 | 9.0 | $ | 8,848 | ||||||||||
| Granted | 4,191,182 | $ | 0.19 | |||||||||||||
| Canceled | (2,447,013 | ) | $ | 0.56 | ||||||||||||
| Outstanding at December 31, 2016 | 7,670,823 | $ | 0.41 | 8.8 | $ | 3,306 | ||||||||||
| Exercisable at December 31, 2016 | 3,876,506 | $ | 0.48 | 8.4 | $ | 3,306 | ||||||||||
| F-25 |
The aggregate intrinsic value was calculated as the difference between the exercise price of the stock options and the fair value of the underlying common stock as of the balance sheet date.
Restricted Stock
Stock-based compensation expense for restricted stock awards was $79,550 and $101,385 for the years ended December 31, 2016 and 2015, respectively.
A summary of restricted stock activity for the year ended December 31, 2016 is as follows:
| Weighted- | ||||||||
| Number of | Average Grant | |||||||
| Shares | Date Fair Value | |||||||
| Balance of unvested restricted stock at December 31, 2015 | 282,119 | $ | 0.24 | |||||
| Issuance of restricted stock | 140,910 | $ | 0.22 | |||||
| Restrictions lapsed | (423,029 | ) | $ | 0.23 | ||||
| Balance of unvested restricted stock at December 31, 2016 | - | |||||||
The Company had no unrecognized stock-based compensation expense for restricted stock awards as of December 31, 2016.
Warrants
As of December 31, 2016 and 2015, there were a total of 22,754,490 and 24,803,409 warrants outstanding to purchase shares of the Company’s common stock, respectively.
A summary of the warrant activity for the year ended December 31, 2016 is as follows:
| As of December 31, 2015 | As of December 31, 2016 | |||||||||||||||||||
| Exercise | Exercise | |||||||||||||||||||
| Warrant Type | Warrants | Price | Activity | Warrants | Price | |||||||||||||||
| PPO | 21,549,510 | $ | 2.00 | (6,863,000 | )(1) | 14,686,510 | (2) | $ | 2.00 | |||||||||||
| PPO Agent | 2,000,000 | $ | 1.00 | - | 2,000,000 | $ | 0.125 | (3) | ||||||||||||
| Enumeral Series B Financing | 421,968 | $ | 0.726 | - | 421,968 | $ | 0.726 | |||||||||||||
| Enumeral 2014 Convertible Promissory Note Financing | 765,357 | $ | 0.245 | - | 765,357 | $ | 0.245 | |||||||||||||
| Square 1 Financing | 66,574 | $ | 0.726 | (66,574 | )(4) | - | ||||||||||||||
| Agent Warrants | - | 4,880,655 | (5) | 4,880,655 | $ | 0.125 | ||||||||||||||
| Total | 24,803,409 | 22,754,490 | ||||||||||||||||||
| 1) | PPO Warrants exercised as part of the Warrant Tender Offer for $0.50 per warrant, or $0.125 per share when considering that warrant holders exercising received four shares for each underlying common share of the original warrant agreement. |
| 2) | PPO Warrants not exercised in the Warrant Tender Offer remain at a $2.00 exercise price. The anti-dilution protection provision has also been eliminated for these warrants in connection with the Warrant Tender Offer. |
| 3) | In connection with the closing of the Warrant Tender Offer, the PPO Agent Warrants were amended to reduce the exercise price from $1.00 to $0.125, and to remove the anti-dilution protection provisions contained therein. |
| F-26 |
| 4) | The cashless exercise of the Square 1 warrants concurrently with the closing of the Warrant Tender Offer. |
| 5) | Agent Warrants issued as a result of the conversion of the Notes in connection with the Warrant Tender Offer. |
Derivative Liability Warrants
PPO and PPO Agent Warrants
In July 2014, the Company issued warrants to purchase 23,549,510 shares of the Company’s common stock in connection with the PPO, of which warrants to purchase 21,549,510 shares of the Company’s common stock had an exercise price of $2.00 per share and were issued to the investors in the PPO, and warrants to purchase 2,000,000 shares of the Company’s common stock had an exercise price of $1.00 per share and were issued to the placement agents for the PPO (or their affiliates). Due to a price protection provision included in the warrant agreements, the warrants were deemed to be and were recorded as a derivative liability a liability. As such, the outstanding warrants are revalued each reporting period with the resulting gains and losses recorded as the change in fair value of derivative liabilities on the consolidated statement of operations and comprehensive income (loss). The estimated fair value of the warrants at December 31, 2015 was determined to be $2,130,822 using the Black-Scholes pricing model and the following assumptions: expected remaining term of 3.58 years, 109.4% volatility, risk-free rate of 1.44%, and no expected dividends.
The estimated fair value of the PPO Warrants and PPO Agent Warrants as of December 12, 2016 (the date of the Warrant Tender Offer) was determined to be $527,461 using the Black-Scholes pricing model and the following assumptions: expected remaining term of 2.6 years, 107.7% volatility, risk-free rate of 1.33%, and no expected dividends. The estimated fair value of the warrants of $527,461, on the date that the derivative liability was extinguished, was reclassified from derivative liabilities to equity.
Also in connection with the consummation of the Warrant Tender Offer, on December 12, 2016, the Company and holders of a majority of the outstanding PPO Agent Warrants approved an amendment to remove the price-based anti-dilution provisions in the PPO Agent Warrants and to reduce the exercise price of the PPO Agent Warrants from $1.00 per share to $0.125 per share. As a result, the Company recorded a loss on extinguishment of derivative liabilities of $187,240.
Square 1 Financing
In connection with the December 2011 Square 1 Financing, Enumeral issued to Square 1 Bank warrants to purchase an aggregate of 33,944 shares of Series A convertible preferred stock at an exercise price of $1.16 per share, exercisable for seven years. In July 2014, as part of the Merger, these warrants were converted into a warrant to purchase 54,245 shares of the Company’s common stock at an exercise price of $0.726 per share. The estimated fair value of the warrants as of December 31, 2015 was determined to be $5,777 using the Black-Scholes pricing model and the following assumptions: expected term of 2.9 years, 104.6% volatility, and a risk-free rate of 1.31%. As of December 31, 2015 these warrants were outstanding and expire on December 5, 2018.
On December 12, 2016, the warrants were exercised in connection with the Warrant Tender Offer. The estimated fair value of the December 2011 Square 1 Warrants as of December 12, 2016 was determined to be $1,649 using the Black-Scholes pricing model and the following assumptions: expected term of 2.0 years, 107.7% volatility, a risk-free rate of 1.15%, and no expected dividends. The estimated fair value of the warrants of $1,649 was reclassified from derivative liabilities to equity immediately after exercise.
As part of a June 12, 2012 amendment to the Loan and Security Agreement between Enumeral and Square 1 Bank, Enumeral issued warrants to Square 1 Bank to purchase an aggregate of 7,715 shares of Series A convertible preferred stock at an exercise price of $1.16 per share, exercisable for seven years. In July 2014, as part of the Merger, these warrants were converted into a warrant to purchase 12,329 shares of the Company’s common stock at an exercise price of $0.726 per share. The estimated fair value of these warrants as of December 31, 2015 was determined to be $1,492 using the Black-Scholes pricing model and the following assumptions: expected term of 3.5 years, 104.6% volatility, and a risk-free rate of 1.42%. The warrants are classified as derivative liabilities in the accompanying balance sheets and measured at fair value on a recurring basis. As such, the outstanding warrants are revalued each reporting period with the resulting gains and losses recorded as the change in fair value of warrant liability on the statement of operations. As of December 31, 2015, these warrants were outstanding and expire on June 12, 2019.
| F-27 |
On December 12, 2016, the warrants were exercised in connection with the Warrant Tender Offer. The estimated fair value of the June 2012 Square 1 Warrants as of December 12, 2016 was determined to be $507 using the Black-Scholes pricing model and the following assumptions: expected term of 2.5 years, 107.7% volatility, a risk-free rate of 1.30%, and no expected dividends. The estimated fair value of the warrants of $507 was reclassified from derivative liabilities to equity immediately after exercise.
Derivative Liability Re-Measurement
The Company used the Black-Scholes option-pricing model to estimate the fair values of the issued and outstanding warrants during the years ended December 31, 2016 and 2015, respectively. The Company recorded income of $1,608,474 and $13,980,711 for the years ended December 31, 2016 and 2015, respectively, due to the change in the fair value of the warrants. Outstanding warrants were revalued each reporting period with the resulting gains and losses recorded as the change in fair value of derivative liabilities on the consolidated statements of operations and comprehensive income (loss). Due to the Tender Offer, and removal of the anti-dilution provisions, the derivative liabilities were re-valued on December 12, 2016 and any remaining value was reclassified to equity upon extinguishment of the derivative liabilities.
Other Warrants
In connection with the conversion of the Notes, the Company issued warrants to purchase 4,880,655 shares of the Company’s common stock at an exercise price of $0.125 per share to the Warrant Agent, its sub-agent and their designees. The estimated fair value of the warrants at the time of issuance was determined to be $704,848 using the Black-Sholes pricing model and the following assumptions: expected term of ten years, exercise price of $0.125 per share, 126.0% volatility, a risk-free rate of 2.49%, and no expected dividends. As the conversion of the notes was an integrated transaction that occurred due to the completion of the Warrant Tender Offer, the Company included the $704,848 estimated fair value of the placement agent warrants issued as part of the loss on extinguishment of derivative liabilities for the year ended December 31, 2016.
On March 21, 2017, the Company entered into an amendment with the holders of the Placement Agent Warrants to reduce the exercise price of such warrants from $0.125 per share to $0.0625 per share in consideration of the past efforts as well as future support and cooperation of the agent and its designees on behalf of the Company.
12 - INCOME TAX
There is no provision for income taxes because the Company has historically incurred operating losses and maintains a full valuation allowance against its gross deferred tax assets. The reported amount of income tax expense differs from the amount that would result from applying domestic federal statutory tax rates to pretax losses primarily because of changes in the valuation allowance.
Significant components of the Company’s net deferred tax asset are as follows:
| As of December 31, | ||||||||
| 2016 | 2015 | |||||||
| Deferred tax assets: | ||||||||
| Net operating losses | $ | 6,750,034 | $ | 4,932,778 | ||||
| Accrued expenses | 208,657 | 482,130 | ||||||
| Stock options | 446,539 | 402,598 | ||||||
| Tax credit carryforwards | 913,326 | 748,255 | ||||||
| Depreciation and amortization | 21,751 | (23,311 | ) | |||||
| Capitalized R&D | 4,933,469 | 4,070,458 | ||||||
| Total gross deferred tax assets | 13,273,776 | 10,612,908 | ||||||
| Valuation allowance | (13,273,776 | ) | (10,612,908 | ) | ||||
| Total deferred tax assets | $ | - | $ | - | ||||
| F-28 |
The Company has evaluated the positive and negative evidence bearing upon the realizability of its deferred tax assets, which are comprised principally of net operating loss carryforwards and research and development credits. Under the applicable accounting standards, management has considered the Company’s history of losses and concluded that it is more likely than not that the Company will not recognize the benefits of federal and state deferred tax assets. Accordingly, a full valuation allowance of $13,273,776 and $10,612,908 has been established as of December 31, 2016 and 2015, respectively. The valuation allowance increased $2,660,868 and $4,292,178 during the years ended December 31, 2016 and 2015, respectively.
A reconciliation of income tax expense computed at the statutory federal income tax rate to income taxes as reflected in the consolidated financial statements is as follows:
| As of December 31, | ||||||||
| 2016 | 2015 | |||||||
| Income tax benefit computed at federal statutory tax rate | 34.0 | % | 34.0 | % | ||||
| State taxes, net of federal benefit | 2.8 | % | (14.8 | )% | ||||
| Non-deductible items | (14.9 | )% | (139.2 | )% | ||||
| General business credits and other credits | 1.2 | % | (10.2 | )% | ||||
| Change in valuation allowance | (22.5 | )% | 130.5 | % | ||||
| Other | (0.6 | )% | (0.3 | )% | ||||
| Effective tax rate | 0.0 | % | 0.0 | % | ||||
As of December 31, 2016, the Company had federal and state net operating loss carryforwards of $17,560,932 and $14,767,832, respectively, which begin to expire in 2030. As of December 31, 2016, the Company had federal and state research and development tax credit carryforwards of $584,846 and $428,982, respectively. These federal and state research and development tax credit carryforwards are available to reduce future income taxes payable and begin to expire in 2031 and 2026, respectively.
As of December 31, 2015, the Company had federal and state net operating loss carryforwards of $12,796,384 and $11,018,952, respectively, which begin to expire in 2030. As of December 31, 2015, the Company had federal and state research and development tax credit carryforwards of $466,599 and $358,038, respectively. These federal and state research and development tax credit carryforwards are available to reduce future income taxes payable and begin to expire in 2031 and 2026, respectively.
The Company adopted the authoritative guidance on accounting for and disclosure of uncertainty in tax positions on January 1, 2009, which required the Company to determine whether a tax position of the Company is more likely than not to be sustained upon examination, including resolution of any related appeals of litigation processes based on the technical merits of the position. For tax positions meeting the more likely than not threshold, the tax amount recognized in the consolidated financial statements is reduced by the largest benefit that has a greater than fifty percent likelihood of being realized upon the ultimate settlement with the relevant taxing authority. The Company determined that the adoption of this authoritative guidance did not have a material effect on the consolidated financial statements.
The Company files tax returns as prescribed by the tax laws of the jurisdictions in which it operates. In the normal course of business, the Company is subject to examination by federal and state jurisdictions, where applicable. There is currently no pending tax examination. The Company thus is still open under statute to potential examination from 2013 to the present. Earlier years may be examined to the extent that credit or loss-carry-forwards are used in it. The Company’s policy is to record interest and penalties related to income taxes as part of the tax provision.
| F-29 |
Utilization of the net operating loss and research and development credit carryforwards may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986, as amended, due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of net operating loss and research and development credit carryforwards that can be utilized annually to offset future taxable income and tax, respectively.
13 - CONCENTRATIONS
During the year ended December 31, 2016, the Company recorded revenue from three customers in excess of 10% of the Company’s revenue in the amounts of $1,000,000, $999,666 and $453,202, which represent 41%, 41% and 18% of the Company’s revenue for that period. During the year ended December 31, 2015, the Company recorded revenue from two customers in excess of 10% of the Company’s revenue in the amounts of $1,100,000 and $389,385, which represent 74% and 26% of the Company’s revenue for that period. As of December 31, 2016, accounts receivable consisted of amounts due from two customers which represented 64% and 36% of the outstanding accounts receivable balance. As of December 31, 2015, accounts receivable consisted of amounts due from two customers which represented 67% and 33% of the outstanding accounts receivable balance.
| F-30 |
EXHIBIT INDEX
The Company is filing as part of this Annual Report on Form 10-K the following exhibits:
| ** | Furnished herewith. |
