Attached files
| file | filename |
|---|---|
| 8-K - 8-K - FIBROGEN INC | d339420d8k.htm |
Exhibit 99.1
FibroGen’s Roxadustat (FG-4592) Meets Primary Endpoints in Two Phase 3 Anemia Studies in China
Roxadustat: effective in dialysis (hemodialysis and peritoneal dialysis) and
non-dialysis chronic kidney disease (CKD) patients
SAN FRANCISCO - January 30, 2017 - FibroGen, Inc. (NASDAQ: FGEN), a science-based biopharmaceutical company, and its subsidiary FibroGen China Medical
Technology Development Co., Ltd. (FibroGen China), today reported positive topline results from the two Phase 3 clinical studies of roxadustat (FG-4592 or
 1) designed to support new drug application submission in China. Roxadustat is a
first-in-class, orally administered small molecule for treatment of anemia in dialysis-dependent CKD (DD-CKD) and non-dialysis-dependent CKD (NDD-CKD) patients. Both of the Phase 3 studies met their primary efficacy endpoints, which were evaluated in the comparator-controlled portions of
the studies.
1) designed to support new drug application submission in China. Roxadustat is a
first-in-class, orally administered small molecule for treatment of anemia in dialysis-dependent CKD (DD-CKD) and non-dialysis-dependent CKD (NDD-CKD) patients. Both of the Phase 3 studies met their primary efficacy endpoints, which were evaluated in the comparator-controlled portions of
the studies.
In the double-blind, placebo-controlled eight-week portion of the 26-week NDD-CKD trial, 151 anemia patients were randomized 2:1 to receive roxadustat (n=101) or placebo (n=50). Roxadustat met its primary efficacy endpoint of correcting anemia, by achieving a statistically significant increase in hemoglobin (Hb) levels compared to placebo over eight weeks. Furthermore, the secondary endpoint of Hb response was met as Hb response was achieved by a higher proportion of patients in the roxadustat arm than in the placebo arm.
| • | Roxadustat-treated patients achieved a mean Hb increase of 1.9g/dL from baseline (8.9g/dL) over eight weeks of treatment vs. a mean change in Hb of -0.4g/dL (from 8.9 g/dL baseline) in the placebo arm (p<0.00001). |
| • | A significantly higher proportion of roxadustat patients achieved Hb response (an increase ³1g/dL from baseline) after eight weeks vs. placebo patients, 84.2% compared to 0.0% (p<0.00001). |
In the dialysis study, 304 patients (271 hemodialysis and 33 peritoneal dialysis patients) previously on
epoetin alfa were randomized to and treated with roxadustat (n=204) or epoetin alfa (n=100) for 26 weeks. All the patients randomized to the active comparator arm were treated with
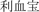 ® (“Li Xue Bao”) epoetin alfa, manufactured in Japan and marketed in China by Kyowa Hakko Kirin China Pharmaceutical Co., Ltd. (Kirin EPO). The
primary endpoint was Hb change from baseline to the Hb level averaged during the final five weeks of the 26-week treatment period.
® (“Li Xue Bao”) epoetin alfa, manufactured in Japan and marketed in China by Kyowa Hakko Kirin China Pharmaceutical Co., Ltd. (Kirin EPO). The
primary endpoint was Hb change from baseline to the Hb level averaged during the final five weeks of the 26-week treatment period.
| • | Roxadustat met the predefined non-inferiority criterion for its primary endpoint in comparison to Kirin EPO in both full analysis set (FAS) and per protocol set (PPS) analyses. |
| • | Furthermore, on superiority testing of the primary endpoint, the mean Hb increase observed in the roxadustat arm was higher than in the Kirin EPO arm, 0.75g/dL vs. 0.46g/dL (p=0.037) in PPS analysis. |
| 1 | China Approved Drug Name or China generic name for “roxadustat” |
Initial findings suggest that adverse events were consistent with previous clinical trials of roxadustat in the CKD patient population with no new or unexpected safety signals identified. The 52-week safety assessment in at least 100 patients is ongoing with expected study completion in June 2017. Full results from these Phase 3 trials are expected to be reported in due course.
“We are grateful for the opportunity to advance development of treatment for anemia associated with CKD, a serious and potentially life-threating condition, for patients in China and worldwide,” said Tom Neff, Chief Executive Officer of FibroGen. “We are very encouraged by the topline results of these two pivotal Phase 3 studies for roxadustat, which bring us closer to the first oral anemia therapy that does not require injectable administration or IV iron supplementation. In addition, use of roxadustat is free of the burden of cold-chain storage and maintaining sterility, and costly distribution infrastructure. Data from these two studies will be used to complete our new drug application submission in China later this year.”
FibroGen and AstraZeneca (NYSE: AZN) are collaborating for the development and commercialization of roxadustat in China, the U.S., and other markets. In China, FibroGen is conducting all clinical trials and will hold all roxadustat regulatory licenses and permits to be issued by China regulatory authorities. After market approval, FibroGen China will manage manufacturing and medical affairs and AstraZeneca will manage launch and commercialization activities in China.
About the FGCL-4592-808 and FGCL-4592-806 Studies
FGCL-4592-808 Phase 3 Study in CKD patients not on dialysis in China: This is a double-blind, placebo-controlled multi-center study in 151 patients, randomized 2:1 to roxadustat or placebo for the first eight weeks, during which 101 patients received roxadustat (initial dose of 70 mg or 100 mg, based on body weight) and 50 patients received placebo three times weekly (TIW), followed by dose titration to Hb levels every four weeks as needed. After the initial eight-week period, placebo-treated patients are crossed over to receive 18 weeks of roxadustat treatment, while the active arm continues on roxadustat for the same period. Primary efficacy endpoint is Hb change from baseline at the end of Week 8. A subset of roxadustat-treated patients entered the ongoing open-label extension for safety assessment and will receive roxadustat for up to 52 weeks of continuous exposure.
FGCL-4592-806 Phase 3 Study in CKD patients on dialysis in China: This is a multi-center, randomized, epoetin alfa-controlled, open-label study in 304 patients over 26 weeks. CKD patients on chronic dialysis (whose anemia was previously treated with stable doses of one or more of nine brands of commercially available epoetin alfa) were randomized 2:1 to roxadustat or Kirin EPO, with 202 patients receiving roxadustat (initial dose of 100 mg or 120 mg TIW, based on body weight) and 100 patients receiving Kirin EPO, followed by dose titration to Hb levels every four weeks as needed. A subset of roxadustat-treated patients entered the ongoing open-label extension for safety assessment and will receive roxadustat for up to 52 weeks of continuous exposure.
About CKD Anemia in China
Anemia commonly develops in association with chronic kidney disease (CKD) and is linked to significant morbidity and mortality in both the dialysis and non-dialysis populations. CKD affects an estimated 119.5 million patients in China (Zhang et al. Lancet 2012; 379: 815–822). Although CKD may occur at any age, it is more common in aging populations, and its prevalence is increasing. CKD can be both a cause and a consequence of cardiovascular disease and is a critical healthcare issue. Currently, there is no treatment available that is curative or has the ability to stop kidney deterioration in patients with CKD with the exception of kidney transplantation.
The dialysis population in China, exceeding 400,000 patients, has been growing at a double-digit rate. The number of patients that require anemia therapy in China and other emerging markets is expected to increase steadily, as the CKD population continues to grow and the number of hemodialysis and peritoneal dialysis patients increases. We believe there is a significant opportunity for roxadustat to treat CKD anemia patients on dialysis (hemodialysis and peritoneal dialysis) and not on dialysis, as well as to address need in the large number of patients whose anemia remains undertreated or untreated in China.
About Roxadustat
Roxadustat (FG-4592) is a first-in-class, orally administered small molecule currently in global Phase 3 clinical development as a potential therapy for anemia associated with CKD. Roxadustat is a hypoxia-inducible factor prolyl hydroxylase inhibitor (HIF-PHI) that promotes erythropoiesis through increasing endogenous erythropoietin, improving iron regulation, and reducing hepcidin. Administration of roxadustat has been shown to induce coordinated erythropoiesis – increasing red blood cell count while maintaining plasma erythropoietin levels within or near normal physiologic range in multiple subpopulations of CKD patients – including in the presence of inflammation and without a need for supplemental intravenous iron.
Roxadustat is currently advancing through Phase 3 clinical trials worldwide, supported by extensive Phase 2 clinical data demonstrating correction and maintenance of hemoglobin levels in multiple subpopulations of CKD anemia patients. To date, roxadustat has been evaluated in Phase 1 and Phase 2 studies, involving more than 1,400 subjects. Globally, a total of 15 Phase 3 studies, with target enrollment of about 10,000 patients worldwide, are currently being conducted to support independent regulatory approvals of roxadustat in both NDD-CKD and DD-CKD patients in the U.S., Europe, Japan, and China.
AstraZeneca and FibroGen are collaborating for the development and commercialization of roxadustat in the U.S., China, and other markets. In China, FibroGen China is conducting all clinical trials and will hold all roxadustat regulatory licenses and permits to be issued by China regulatory authorities. After market approval, FibroGen China will manage manufacturing and medical affairs and AstraZeneca will manage launch and commercialization activities in China.
Astellas and FibroGen are collaborating for the development and commercialization of roxadustat in Europe, Japan, the Commonwealth of Independent States, the Middle East, and Africa.
Roxadustat is also entering a Phase 3 clinical trial in the U.S. for treatment of anemia in patients with myelodysplastic syndrome (MDS). In China, a roxadustat MDS Phase 2/3 clinical trial application (CTA) is currently under review by the China Food and Drug Administration (CFDA).
For information about roxadustat studies currently recruiting patients, please visit clinicaltrials.gov at this link:
https://clinicaltrials.gov/ct2/results?term=FG-4592&Search=Search
About FibroGen
FibroGen, Inc., headquartered in San Francisco with subsidiary offices in Beijing and Shanghai, is a leading science-based biopharmaceutical company discovering and developing a pipeline of first-in-class therapeutics. The company applies its pioneering expertise in fibrosis and hypoxia-inducible factor (HIF) biology and clinical development to advance innovative medicines for the treatment of anemia, fibrotic disease, and cancer. Roxadustat (FG-4592), the company’s most advanced product candidate, is an oral small molecule inhibitor of HIF prolyl hydroxylase activity in Phase 3 clinical development for the treatment of anemia in CKD. Pamrevlumab (FG-3019), a fully-human monoclonal antibody that inhibits the activity of connective tissue growth factor (CTGF), is in Phase 2 clinical development for the treatment of idiopathic pulmonary fibrosis (IPF), pancreatic cancer, and Duchenne’s muscular dystrophy (DMD). FibroGen is also developing a biosynthetic cornea in China. For more information, please visit www.fibrogen.com.
Forward-Looking Statements
This release contains forward-looking statements, including statements regarding the potential ability of roxadustat to treat anemia in CKD patients, and the potential commercial market for roxadustat worldwide, including in China and emerging markets, timelines for study completion, reporting data, and clinical and regulatory developments for roxadustat. Our actual results may differ materially from these early data and any forward-looking statements due to risks and uncertainties that are described in our Annual Report on Form 10-K and our quarterly reports on Form 10-Q filed with the Securities and Exchange Commission, including the risk factors set forth therein. Investors are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this release and we undertake no obligation to update any forward-looking statement in this press release, except as required by law.
# # #
Contact
FibroGen, Inc.
Karen L. Bergman
VP, Investor Relations and Corporate Communications
+1.415.978.1433
kbergman@fibrogen.com
