Attached files
| file | filename |
|---|---|
| 8-K - 8-K - PRESENTATION DECK AND PRESS RELEASE DATED 01-05-2017 - Ocera Therapeutics, Inc. | ocera-8xkrejpm.htm |
| EX-99.2 - EXHIBIT 99.2 - Ocera Therapeutics, Inc. | oceraannouncessuccessfulph.htm |

A Liver Disease Medicines Company
NASDAQ: OCRX
J A N UA R Y 2 0 1 7

2
Forward‐Looking Statements
Certain statements in this presentation constitute “forward‐looking statements” within the meaning of
the Securities Act of 1933, as amended (the “Securities Act”), and Securities Exchange Act of 1934, as
amended (“Exchange Act”), including, without limitation, all statements related to the OCR‐002 clinical
development program, including patient enrollment estimates, expected timing for the receipt of clinical
data, the potential success of OCR‐002 in clinical trials, the size of the potential market opportunity for
OCR‐002, as well as cash projections, and we intend these forward‐looking statements to be covered by
the safe harbor provisions for forward‐looking statements contained in the Securities Act and the
Exchange Act and are making this statement for purposes of complying with those safe harbor provisions.
These forward‐looking statements reflect our current views about our plans, intentions, expectations,
strategies and prospects, which are based on the information currently available to us and on
assumptions we have made. Although we believe that our plans, intentions, expectations, strategies and
prospects as reflected in or suggested by those forward‐looking statements are reasonable, we can give
no assurance that the plans, intentions, expectations or strategies will be attained or achieved.
Furthermore, actual results may differ materially from those described in the forward‐looking statements
and will be affected by a variety of risks and factors that are beyond our control, including those risks and
uncertainties discussed under “Risk Factors” in our Annual Report on form 10‐K for the year ended
December 31, 2015, as well as other risks detailed in our subsequent filings with the SEC. All information
in this presentation is as of the date of this presentation, and we undertake no duty to update this
information unless required by law.

3
Seeking to Provide Continuity of Care from Treatment to Prevention
Developing Therapeutics for
Liver Diseases
Lead Program OCR‐002
Ammonia Scavenger for Hepatic Encephalopathy (HE)
IV‐Phase 2b
ACUTE
Oral‐Phase 1
CHRONIC

4
Investment Highlights
OCR‐002 for Hepatic Encephalopathy (HE): ~$1.5‐2B potential U.S.
market opportunity
Novel ammonia scavenger; broad IP protection & WW rights
IV formulation for acute use and oral formulation for chronic use
Only direct ammonia scavenger in development for HE
DIFFERENTIATED
PRODUCT FOR
UNMET NEED
IV Phase 2b STOP‐HE study data
Oral Phase 2a multi‐dose study initiation in cirrhotic patients
ANTICIPATED
NEWS FLOW
NEXT TWO
QUARTERS
Cash as of September 30, 2016: $32.5M
Existing cash resources expected to fund development into Q1 2018
FINANCIAL
STRENGTH

5
LINDA S. GRAIS, MD, JD
President & Chief Executive Officer
STAN BUKOFZER, MD
Chief Medical Officer
MICHAEL BYRNES, MBA
Chief Financial Officer
Experienced Leadership
AND a Team with Deep Clinical and Development Experience

6
Disorientation
Impaired
Motor Skills
Personality
Changes
Stupor Coma Death
0 1 2 3 4
Hepatic Encephalopathy (HE):
Neurocognitive Disorder in Serious Liver Disease
Blood
Stream
Ammonia
Gut
Elevated Ammonia Levels Drive HE
Note: 0 to 4 as measured by West Haven Scale

7
OCR‐002 Mechanism of Action:
Clears Ammonia When the Liver Cannot
Ornithine
Contributes to
Glutamate, which
combines with Ammonia
to Create Glutamine
Gut
OCR-
002
Kidney
PAGN
Excretion
AmmoniaGlutamate
PAA Combines with
Glutamine to Form
Phenylacetylglutamine
(PAGN) Which Is Excreted
Through Kidneys
Phenylacetic
Acid (PAA) Non-Toxic Carrier of Ammonia,
Contributes to Muscle Building
Glutamine
PAGN
Note: Simplification of MOA

8
Ammonia Lowering Reduced Risk of HE in
HALT‐HE Study
1 “Randomized, Double‐Blind, Controlled Study of Glycerol Phenylbutrate in Hepatic Encephalopathy” Rockey, et al. Hepatology 2014; 59:1073‐1083
2 “Fasting Ammonia (NH3) as a Predictor or Hepatic Encephalopathy (HE) Events” Vierling, et al. Journal of Hepatology 2013; 58: S63‐S227
• Studies have long observed an
association between ammonia and HE
• Causal link demonstrated by Ravicti®
HALT‐HE Study
– Significantly delayed time to next HE
episode (p<0.05)1
– Significantly reduced proportion of
patients who experienced HE episode
(p=0.02)1
– Patients with baseline fasting ammonia
>1.5x upper limit of normal had a six‐
fold elevation in rate of annualized HE
episodes compared to patients with
fasting ammonia between 0‐1.5x ULN
(p<0.01)2

9
OCR‐002 is the ONLY Direct Ammonia Scavenger in
Development for HE Treatment and Prevention
Ammonia
RIFAXIMIN
Non‐Absorbed Antibiotic
Kills Gut Bacteria
− Only approved for maintenance,
not acute treatment
− Does not directly clear circulating
ammonia
LACTULOSE
Laxative, Used for Decades
+ Inexpensive
− Hard to administer
− Diarrhea
− Poor compliance
Current
Treatments are
Indirect and
Limited to Oral
RAVICTI
Active Ingredient: Phenylacetate
+ Ammonia scavenger approved for UCD
+ Strong clinical efficacy for HE prevention
− HE development halted by Horizon
− Priced for genetic ultra‐orphan disease,
unsuitable for HE
AMMONUL
Sodium Phenylacetate and Sodium Benzoate
+ Potent in acute urea cycle disorders
− Requires large volumes
− Label cautions usage due to high sodium
load; electrolyte and fluid shifts may be
unsafe in HE patients
Other
Ammonia Scavengers
Not Approved for HE

10
OCR‐002 Designed to Treat HE
• IV easy to administer in acute care patients
– Peripheral IV line, low infusion volume and neutral pH
– Rapid onset
– Demonstrated favorable safety and tolerability profile in sick
population
– No sodium load or electrolyte disturbances
• Oral formulation for chronic care patients
– PAA validated ammonia scavenger in HALT‐HE Study for
prevention of HE
– Provides continuity of care for patients at home
– Provides opportunity for step‐down care post IV therapy

11
• Sample size of approximately 230 patients provides 80% power to detect the difference at study‐end that
was observed at the planned interim analysis
STOP‐HE Phase 2b: OCR‐002 IV for Overt HE
Enrollment Complete – Data Q1 2017
*As scored by a modified version of the West Haven Scale
Key Exclusion
• Kidney failure/dialysis
• Life expectancy < 2 weeks
HOSPITALIZED
CIRRHOTICS
Key Inclusion
• West Haven* Score ≥ 2
• Ammonia >ULN
• Standard of care (SOC)
treatment and observation
• Physician’s choice for SOC
• Patients must remain in
HE
• Weed out rapid
responders
MINIMUM 12‐HOUR
PRE‐SCREEN
• Randomized 1:1
− placebo + SOC
− drug + SOC
• Continuous infusion for up
to 5 days
• Patient assessed 2x/day
• Primary Endpoint: Time to
meaningful clinical
improvement in HE
symptoms
TREATMENT
12
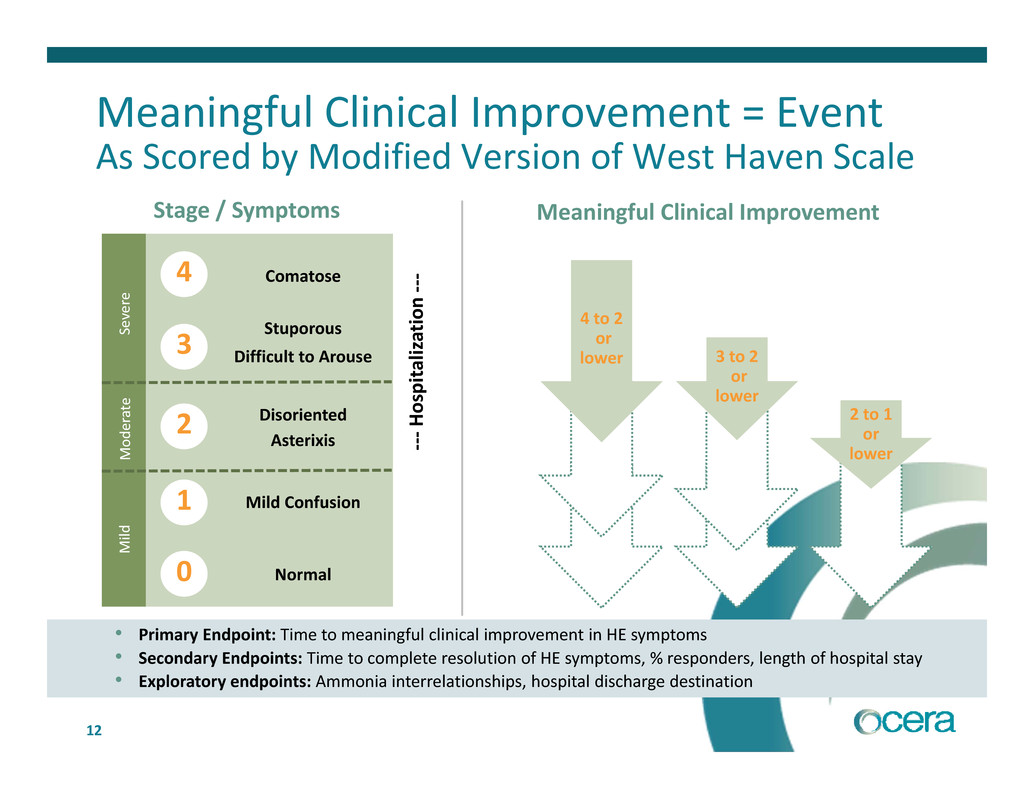
• Primary Endpoint: Time to meaningful clinical improvement in HE symptoms
• Secondary Endpoints: Time to complete resolution of HE symptoms, % responders, length of hospital stay
• Exploratory endpoints: Ammonia interrelationships, hospital discharge destination
Meaningful Clinical Improvement = Event
As Scored by Modified Version of West Haven Scale
Stage / Symptoms Meaningful Clinical Improvement
4 to 2
or
lower 3 to 2
or
lower
2 to 1
or
lower
0
1
2
3
4
‐
‐
‐
H
o
s
p
i
t
a
l
i
z
a
t
i
o
n
‐
‐
‐Comatose
Stuporous
Disoriented
Difficult to Arouse
Asterixis
Mild Confusion
Normal
M
o
d
e
r
a
t
e
M
i
l
d
S
e
v
e
r
e

13
STOP‐HE Study Powering Assumptions
Initial
Design
Following DMC
Recommendation
At Interim Analysis
PBO Active PBO Active
Percent Response thru
5 days (120 hrs)
45% 65% ? ?
Median Time to
Response
5.7 days
(139 hrs)
3.2 days
(79 hrs)
? ?
Treatment Benefit 2.5 days (60 hrs)* Observed by DMC
Total Patients 200 ~230
Power 80% 80%
• 200 patients is 80% powered to show treatment benefit of 60 hours
• 230 patients is 80% powered to show treatment benefit at the end of the study
that was observed by the DMC at the interim analysis
*Any Treatment Benefit in Excess of 24 hours is Considered Meaningful

14
STUDY
RAVICTI®
Phase 2 HALT‐HE
OCR‐002
STOP‐HE (Enrollment Complete)
Dose of Drug 6mL BID = 13.2g/day
10g/15g/20g/day
(depending on liver function, dose
adjustment to normalize PAA levels)
Mean PAA
Exposure
65‐150μg/mL1 Target: 75‐150μg/mL
Clinical
Results
Statistically significant reductions in
incidence of HE events and time to first
event2
Promising interim analysis; sample size
recommendation highly suggestive of
treatment benefit
Conclusion Dose sufficient for clinical benefit
Dosing strategy to achieve similar PAA
levels as RAVICTI® study
1 “Glycerol Phenylbutyrate in Patients With Cirrhosis and Episodic Hepatic Encephalopathy: A Pilot Study of Safety and Effect on Venous Ammonia
Concentration” Ghabril, et al. Clinical Pharmacology in Drug Development 2013; 2(3) 278‐284
2 “Randomized, Double‐Blind, Controlled Study of Glycerol Phenylbutrate in Hepatic Encephalopathy” Rockey, et al. Hepatology 2014; 59:1073‐1083
Dose Rationale for OCR‐002 Informed by HALT‐HE

15
Outlook for STOP‐HE
Favorable
Safety and
Tolerability
Profile
PROMISING FACTORS
Appropriate
Dosing Level
Clinically
Validated
Mechanism
Encouraging
Interim
Analysis
Enrollment Completed; Data Expected Q1 2017

16
Oral OCR‐002 Unique Opportunity
Develop orally‐administered formulation of OCR‐002 in concert
with IV formulation to provide continuity of care for HE patients
GOAL OF OCR‐002
ORAL PROGRAM
Phase 1 program showed favorable single‐dose PK data
Phase 2a study with new optimized tablet formulation planned
for H1 2017
STATUS AND
NEXT STEPS
1.5‐2M cirrhotics at risk of hepatic encephalopathy in the U.S.
Less than 10% of at risk population represents $900M ‐ $1.2B
potential revenue opportunity in the U.S. alone
High re‐hospitalization rate for those discharged
MARKET
OPPORTUNITY

17
Proof of Principle Achieved in Phase 1 Program
Phase 1: Cirrhotic Patient Study – Completed Q1 17
Single Dose Orally‐Administered OCR‐002
Findings include:
Single IV Dose of OCR‐002VS
• Absolute bioavailability > 95% in the fasted state
• Pharmacokinetics supporting convenient dosing
Extended‐Release Formulations of OCR‐002
Glycerol Phenylbutyrate (RAVICTI®)
Immediate‐Release Oral Solution of OCR‐002
VS
Phase 1: Healthy Volunteer Study – Completed Q4 15
Findings include:
• Rapid onset of adequate plasma PAA concentrations with all OCR‐002 formulations
• Sustained higher drug levels and PAGN than Ravicti
All doses and treatment arms of OCR‐002 observed were safe and well‐tolerated

18
Oral OCR‐002 Phase 2a Study; Initiation H1 2017
Multi‐Dose Orally‐Administered OCR‐002 Glycerol Phenylbutyrate (RAVICTI®)VS
Phase 2a: Cirrhotic Patients
Evaluation of:
• New optimized tablet formulation of OCR‐002
• Steady state pharmacokinetics
• Pharmacodynamics
• OCR‐002 vs. Clinically‐validated ammonia scavenger

19
Ammonia
Brain
Swelling3
Portal
Hypertension2
NASH4
Protein
Catabolism5
Neuronal
Cell Death1
Elevated Ammonia Correlated with Other Complications
of Cirrhosis; OCR‐002 Potentially Beneficial
1 Oral Presentation; Clément et al (AASLD 2015)
2 Jalan, et al “Ammonia produces pathological changes in human hepatic
stellate cells and is a target for therapy of portal hypertension” J Hepatology
2016 p. 823‐233
3 Oral Presentation; Ventura‐Cots et al (AASLD 2015)
4 Poster #908; Thomsen et al (AASLD 2015)
5 Poster #1523; Bosoi et al (AASLD 2015)
20
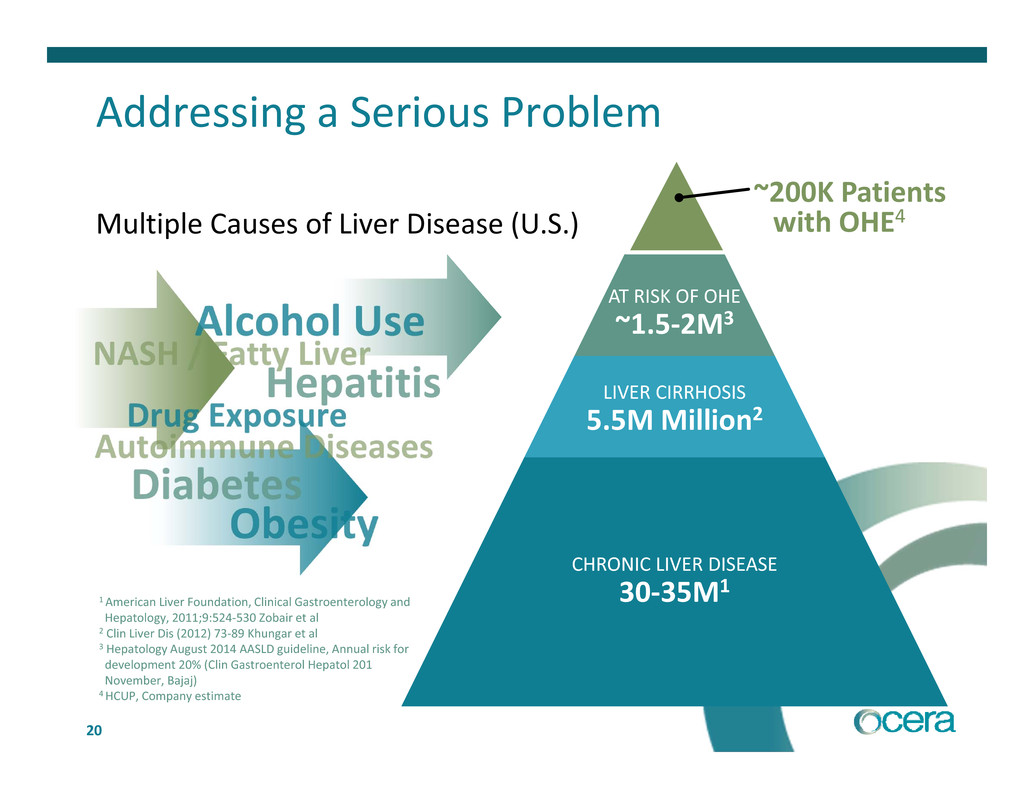
Addressing a Serious Problem
Multiple Causes of Liver Disease (U.S.)
CHRONIC LIVER DISEASE
30‐35M1
LIVER CIRRHOSIS
5.5M Million2
AT RISK OF OHE
~1.5‐2M3
~200K Patients
with OHE4
1 American Liver Foundation, Clinical Gastroenterology and
Hepatology, 2011;9:524‐530 Zobair et al
2 Clin Liver Dis (2012) 73‐89 Khungar et al
3 Hepatology August 2014 AASLD guideline, Annual risk for
development 20% (Clin Gastroenterol Hepatol 201
November, Bajaj)
4 HCUP, Company estimate

21
1 HCUP Database
2 Clinical Gastroenterology and Hepatology 2012;10:1034–1041
3 Annualized based off of actual reported revenues from Q1‐16 through Q3‐16
HE: Large and Growing Healthcare Burden
Rising Hospitalizations1
(000s)
• Total national charges
related to HE: $7 Billion2
• HE hospitalizations
continue to grow despite
Rifaximin launch in 20101
• Rifaximin annual revenues:
$900+ Million3
• HE patient demographics
show increase in severity
of illness, elderly
population, and obesity as
comorbidities2
105 107 106 110 120 133 141 148
156
83 104
180 196
252
275
331
380
436
2006 2007 2008 2009 2010 2011 2012 2013 2014
Hepatic Coma
Encephalopathy
Rifaximin
approved for
Prevention of
HE April 2010

22
Long Patient Stays1
HE Patient Distribution
60% >4days
>14 days
15%
5‐14 days
45%
1‐4 days
40%
Faster Improvement in HE Can Reduce Length of Stay
1 Ocera market research (CMS Medicare U.S. data 2011)
2 Becker’s Review / Hospital Review / May 2015
3 HCUP Hospital Intensive Care Units 2011
• ~One third of HE patients are treated in ICU1
‐ Average in‐patient hospital bed “cost” is
$1,500‐$2,000/Day2
‐ ICU cost approximately ~2.5X3
• 45‐50% of hospitalized HE patients are on
Medicare1
‐ Hospitals start losing money around day
four
Hospital Cost Drivers
Hospital stay/ICU stay and Medicare
HE: Longer Hospitalization Drives Cost

23
• Seeks to provide continuity of
care with IV
• Goal is to prevent re‐
hospitalization and enable
patients to remain stable at
home
ORAL
• Up to 600K annual
hospitalizations1
• Potentially favorable
reimbursement
• $1,300 / day for OCR‐0022
• Presumed standard of care
• Protocol in most institutions
IV
Significant Commercial Opportunity for
IV and Oral OCR‐002
$600 ‐
800MM
$900 ‐
1.2MM
~200K
PAT I E N T S A NNUA L LY
~$1.5‐2B
U. S . O P PORTUN I T Y
1 HCUP Database (includes ICD‐9 codes 572.2/hepatic coma and 348.3/ encephalopathy NOS)
2 Company research

24
Valuable Commercial Estate
BROAD PATENTS
Composition
of Matter
to 2030 (not including
Hatch‐Waxman
extension)
ORPHAN STATUS
IN US
WORLDWIDE
RIGHTS

25
HIGH HE Burden
UNMET Medical Need
ONLY Direct Ammonia Scavenger for HE
PROMISING Clinical Programs
ATTRACTIVE Commercial Opportunity
NEAR TERM MILESTONES
• STOP‐HE P2b Data
• Oral P2a Multi‐dose Study Initiation
V11022016
