Attached files
| file | filename |
|---|---|
| 8-K - 8-K - Lipocine Inc. | v446370_8k.htm |

Annual Canaccord Genuity Growth Conference 2016 Presentation August 2016 Exhibit 99.1

Forward Looking Statements 2 This presentation contains forward - looking statements about Lipocine Inc. (the “Company”). These forward - looking statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward - looking stat ements relate to the Company’s product candidates, clinical and regulatory processes and objectives, potential benefits of the Compa ny’ s product candidates, intellectual property and related matters, all of which involve known and unknown risks and uncertainties . Actual results may differ materially from the forward - looking statements discussed in this presentation . Accordingly, the Company cautions investors not to place undue reliance on the forward - looking statements contained in, or made in connection with, this presentation . Several factors may affect the initiation and completion of clinical trials, the potential advantages of the Company’s product candidates and the Company’s capital needs. Among other things, the projected commencement and completion of the Company’s clinical trials may be affected by difficulties or delays. In addition, the Company’s results ma y b e affected by its ability to manage its financial resources, difficulties or delays in developing manufacturing processes for its produc t c andidates, preclinical and toxicology testing and regulatory developments. Delays in clinical programs, whether caused by competitive developments, adverse events, patient enrollment rates, regulatory issues or other factors, could adversely affect the Compan y’s financial position and prospects. Prior clinical trial program designs and results are not necessarily predictive of future cli nical trial designs or results. If the Company’s product candidates do not meet safety or efficacy endpoints in clinical evaluations, th ey will not receive regulatory approval and the Company will not be able to market them. The Company may not be able to enter into any strategic partnership agreements. Operating expense and cash flow projections involve a high degree of uncertainty, including variances in future spending rates due to changes in corporate priorities, the timing and outcomes of clinical trials, compet iti ve developments and the impact on expenditures and available capital from licensing and strategic collaboration opportunities. If the Company is unable to raise additional capital when required or on acceptable terms, it may have to significantly delay, scale ba ck or discontinue one or more of its drug development or discovery research programs. The Company is at an early stage of developm ent and may not ever have any products that generate significant revenue. The forward - looking statements contained in this presentation are further qualified by the detailed discussion of risks and uncertainties set forth in the documents filed by the Company w ith the Securities and Exchange Commission, all of which can be obtained on the Company’s website at www.lipocine.com or on the SEC website at www.sec.gov . The forward - looking statements contained in this document represent the Company’s estimates and assumptions only as of the date of this presentation and the Company undertakes no duty or obligation to update or revise publicly any forward - looking statements contained in this presentation as a result of new information, future events or changes in the Company’s expectations.

Lipocine Investment Highlights 3 ▪ Differentiated product targeting ~$2.0 Billion established US TRT market ▪ Targets unmet need with first entrant advantage ▪ Robust clinical data with branded market leader as active control ▪ LPCN 1111 – Next generation oral TRT option currently in Phase 2b with results expected 3Q 2016 ▪ LPCN 1107 Orphan designated oral alternative for the prevention of preterm birth – Potential to optimize clinical outcomes – Avoids painful injections and injection site reaction – EOP2 meeting scheduled for August 2016 LPCN 1021: Potential first oral TRT option Pipeline assets advancing towards “Phase 3 ready” status

LPCN: Focused on Innovative Products for Men’s and Women’s Health 4 “Transformative“ Oral Testosterone Franchise First Oral Alternative for the Prevention of Pre - Term Birth PRODUCT (Indication) RESEARCH / PRECLINICAL PHASE 1 PHASE 2 PHASE 3 NDA MEN'S HEALTH LPCN 1021 (Oral Testosterone Replacement Therapy) LPCN 1111 (Next Generation Oral T) WOMEN'S HEALTH LPCN 1107 (Prevention of Preterm Birth) End of Review Meeting with FDA – 2H 2106

LPCN 1021: Committed to Bringing LPCN 1021 to Patients ▪ LPCN 1021 received a Complete Response Letter (“CRL”) from the U.S. Food and Drug Administration (“FDA”) on June 28, 2016 – Deficiency identified relates to the dosing algorithm for the label ▪ The proposed titration scheme in the NDA for clinical practice was significantly different from the titration scheme used in the Phase 3 trial leading to discordance in titration decisions between the Phase 3 trial and real - world clinical practice ▪ CRL did not identify any other deficiencies ▪ Phase 3 study design with intensive 24 hour sampling previously used in FDA approved testosterone product ▪ Approvability dependent upon deriving a practical dosing algorithm for the label for safe and effective “real world” use ▪ Next step: Request a meeting with the FDA in 3Q 2016 – Understand more fully the issues raised – Agreement on a path forward for approval 5

TLANDO - First Oral TRT Option 6 D IFFERENTIATION vs. MARKET LEADER ▪ Not prone to accidental T transference ▪ Starting dose is the right dose for most patients ▪ Patient preferred oral option ▪ LPCN 1021 met Primary endpoint: 87% response rate vs. FDA requirement of 75% ▪ Secondary endpoints generally consistent with approved products ▪ T levels not affected by food fat content EFFICACY SAFETY ▪ 52 week long term exposure data ▪ Well tolerated ▪ AE profile comparable to active control, including GI ▪ No cardiac, hepatic or drug related SAEs

2014 2015 Source: IMS Dec. 2015 TRT Market Monthly TRx Trend 350 375 400 425 450 475 500 525 550 575 Sep 14 Oct 14 Nov 14 Dec 14 Jan 15 Feb 15 Mar 15 Apr 15 May 15 Jun 15 Jul 15 Aug 15 Sep 15 Oct 15 Nov 15 Dec 15 Jan 16 Feb 16 Mar 16 Apr 16 May 16 Monthly TRx (000s) FDA Label Guidance 7 x No TRx impact of FDA TRT label change x Monthly TRx stable around 500,000/month 2016
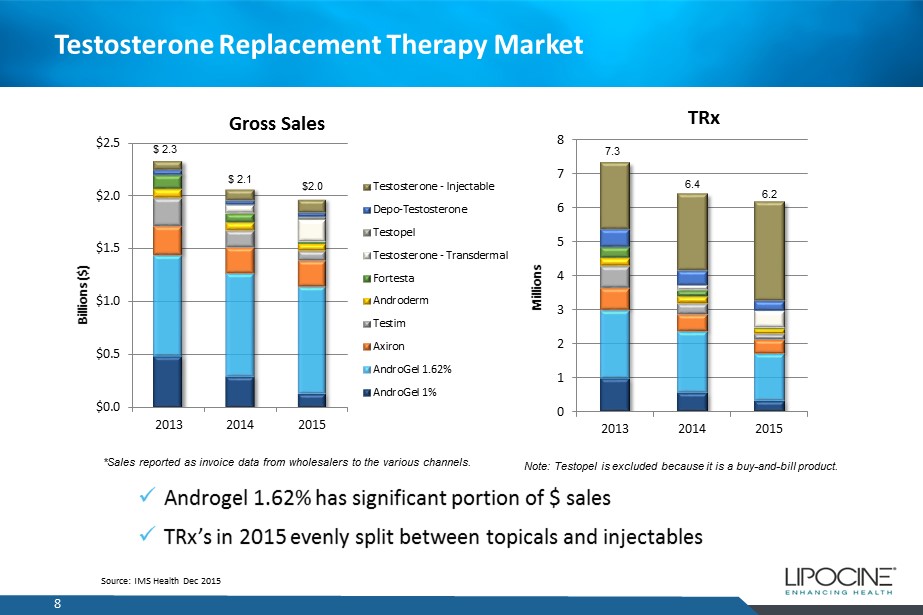
Testosterone Replacement Therapy Market 8 Source: IMS Health Dec 2015 $0.0 $0.5 $1.0 $1.5 $2.0 $2.5 2013 2014 2015 Billions ($) Gross Sales Testosterone - Injectable Depo-Testosterone Testopel Testosterone - Transdermal Fortesta Androderm Testim Axiron AndroGel 1.62% AndroGel 1% 0 1 2 3 4 5 6 7 8 2013 2014 2015 Millions TRx $ 2.3 $ 2.1 $2.0 *Sales reported as invoice data from wholesalers to the various channels. Note: Testopel is excluded because it is a buy - and - bill product. 7.3 6.4 6.2 x Androgel 1.62% has significant portion of $ sales x TRx’s in 2015 evenly split between topicals and injectables
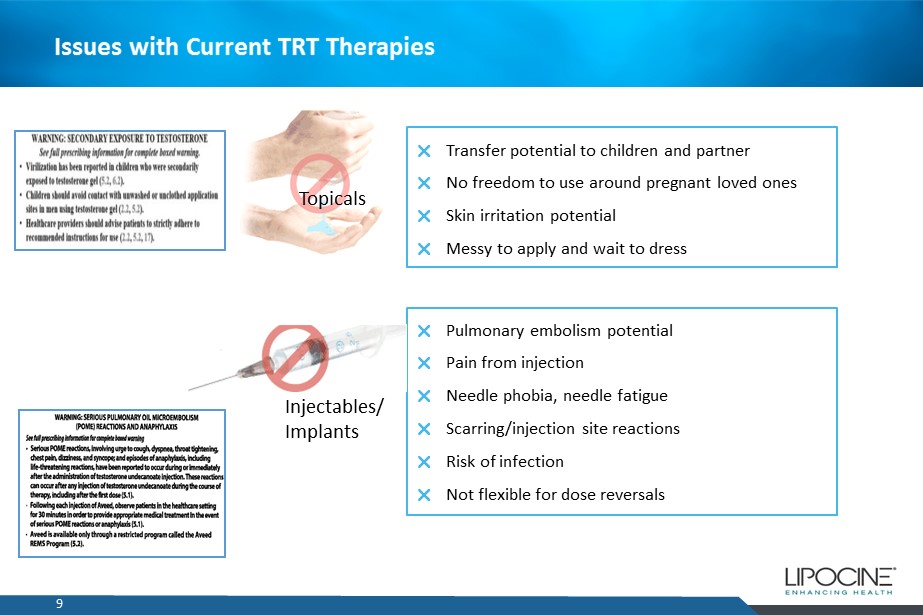
9 Issues with Current TRT Therapies Transfer potential to children and partner No freedom to use around pregnant loved ones Skin irritation potential Messy to apply and wait to dress Pulmonary embolism potential Pain from injection Needle phobia, needle fatigue Scarring/injection site reactions Risk of infection Not flexible for dose reversals Topicals Injectables / Implants

TLANDO (First Oral TRT Option): Why No Oral Product To - Date Challenges of Oral T Product ▪ Native testosterone has poor oral bioavailability with a very short half life (~30 min) – Impractical daily doses would be required to obtain effective levels – Inconsistent and unpredictable performance ▪ Methyl testosterone – Liver toxicity – Unsafe for chronic use LPCN 1021 Advantage ▪ Novel product primarily directing Testosterone Undecanoate (TU) into the lymph – Maintains effective T blood levels in eugonadal range when dosed twice daily – Consistent and predictable performance – By - passes liver in first pass metabolism 10

11 TLANDO (First Oral TRT Option): Primary Efficacy Results in Phase 3 Measure FDA Targets Efficacy Population* 1,2 Full Analysis Set #1 Number of subjects 151 193 % subjects with C avg w ithin normal range (300 - 1140 ng/ dL ) ≥75% 87.4% 87.0% 95 % CI lower bound ≥ 65% 81.7% 82.0% x TLANDO met both the primary endpoint targets x C avg and overall variability comparable or better than marketed products Parameter Mean (CV) Mean (CV) C avg ( ng / dL ) 446 (38%) 471 (41%) * Subjects randomized into the study with at least one PK profile and no significant protocol deviations # Subjects randomized into the study with at least one post - baseline efficacy variable response 1 Missing data imputed by LOCF 2 3.3% of subjects were non - responders ( C avg <300 ng/ dL at highest (300 mg) dose)

TLANDO (First Oral TRT Option): Secondary Endpoints in Phase 3 ▪ Proportion of subjects achieving maximum serum total T concentrations (Cmax) in predefined Cmax range 12 Measure FDA Threshold Efficacy Population Number of subjects 151 C max < 1500 ng/ dL ≥ 85 % 82.8% 1800 ≤ C max ≤ 2500 ng/ dL ≤ 5 % 4.6% C max > 2500 ng/ dL None 2.0% x Results generally consistent with approved products

LPCN 1111 - Next Generation Oral TRT Option ▪ Novel prodrug of testosterone for oral delivery through proprietary drug delivery technology ▪ Once daily is expected to sustain and improve market share or oral T franchise ▪ Once daily feasibility established – Positive Phase 2a study results in hypogonadal men ▪ Single daily oral dose provides T levels in the eugonadal range ▪ No subject exceeded peak T levels of 1500 ng/ dL ▪ Development status – Phase 2b PK dose finding study – ongoing – Next step: Top - line results expected – 3Q 2016 13

LPCN 1111 Phase 2a Study - Responder Analysis At Day 28 x Once daily dosing feasibility established 14 Measure 550mg QD 770mg QD Typical FDA targets for approval of TRT C avg w ithin normal range 67% 88% ≥ 75% C max < 1500 ng / dL 100% 100% ≥ 85% 1800 ≤ C max ≤ 2500 ng / dL 0% 0% ≤ 5% C max > 2500 ng / dL 0% 0% 0%

Preterm Birth (PTB) Represents a Significant Unmet Medical Need ▪ 11.7% of all US pregnancies 2 result in PTB (< 37 weeks) - a leading cause of neonatal mortality and morbidity 3 ▪ ~10x more first year medical costs are for PTB infants than for full term infants 4 ▪ ≥ $26 billion economic impact: 4 $1 billion market opportunity 5 15 1 Pediatric Research (2006) 60, 775 – 776 2 CDC (2010) 3 J . Maternal - Fetal and Neonatal Medicine, Dec. 2006, 19(12), 773 – 782 4 Institute of Medicine of the National Academies. Jul.2006 5 AMAG Pharmaceuticals presentation 09/29/2014 One preterm infant per minute in the U.S. 1

LPCN 1107 (First Oral PTB Candidate): “Addresses Unmet Need” 16 ▪ Potential to be the first oral standard - of care therapy – Elimination of 18 - 22 injections – Potential for optimizing clinical outcomes ▪ Orphan drug designation – A major contribution to patient care ▪ Development status – Pivotal Phase 3 study starting dose/rationale identified – Next step: End of Phase 2 meeting – August 2016 • Primary objective: x Agreement on development plan for sub part H approval x Discuss pivotal study design

LPCN 1107 (First Oral PTB Candidate): Competitive Landscape 17 ▪ Makena® IM injection - only approved product – Viscous oily vehicle – 21 gauge needle – Injection takes up to 1 min – ~35% of patients experienced injection site pain during clinical trial – ~17% of patients reported site swelling - much greater than placebo during clinical trial – AMAG 2016 guidance for Makena: $310 - $340 million ▪ Limited competitive pipeline

LPCN 1107 (First Oral PTB Candidate): Potential to Optimize Clinical Outcomes Limitations of Current Injectable Therapy Potential Advantages of LPCN 1107 18 ▪ Several weeks to get to steady state HPC levels ▪ “One size fits all” fixed single dosing option ▪ Weekly visits to care giver ▪ Steady state levels in seven days ▪ Amenable to customize dose – Enables sooner desired HPC levels in patients – Maintain patient at desired levels through dose titration independent of pre - pregnancy BMI, age, race, etc. – No lingering HPC levels post delivery

Multiple Near Term Value Drivers 19 Event Expected Timing LPCN 1021: Request End of Review Meeting 3Q16 LPCN 1107: End of Phase 2 Meeting 3Q16 LPCN 1111: Top - line Results from Phase 2b Study 3Q16 LPCN 1021: End of Review Meeting 2H16 LPCN 1111: End of Phase 2 Meeting 4Q16

20 Lipocine is a Compelling Value Proposition Stock Exchange NASDAQ Capital Markets Ticker Symbol LPCN Closing Stock Price (8/4/16) $3.70/share Market Capitalization (8/4/16) $67.5 million Fully Diluted Shares Outstanding (6/30/16) 20,504,298 Cash Balance (6/30/16) $32.9 million Debt None

Lipocine Investment Highlights 21 ▪ Differentiated product targeting ~$2.0 Billion established US TRT market ▪ Targets unmet need with first entrant advantage ▪ Robust clinical data with branded market leader as active control ▪ LPCN 1111 – Next generation oral TRT option currently in Phase 2b with results expected 3Q 2016 ▪ LPCN 1107 Orphan designated oral alternative for the prevention of preterm birth – Potential to optimize clinical outcomes – Avoids painful injections and injection site reaction – EOP2 meeting scheduled for August 2016 LPCN 1021: Potential first oral TRT option Pipeline assets advancing towards “Phase 3 ready” status
