Attached files
| file | filename |
|---|---|
| EX-99.1 - EXHIBIT 99.1 - Vyant Bio, Inc. | a2016q2exhibit991-pressrel.htm |
| 8-K - 8-K - Vyant Bio, Inc. | a2016q2pressrelease-form8xk.htm |

August 10, 2016
Q2 2016 Earnings Call Nasdaq (CGIX)
THE ONCOLOGY
DIAGNOSTICS
PARTNER FROM
BENCH TO BEDSIDE

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Forward-Looking Statements
2
These slides may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act
of 1995. All statements pertaining to future financial and/or operating results, future growth in research, technology,
clinical development and potential opportunities for Cancer Genetics, Inc. products and services, along with other
statements about the future expectations, beliefs, goals, plans, or prospects expressed by management constitute
forward-looking statements.
Any statements that are not historical fact (including, but not limited to, statements that contain words such as "will,"
"believes," "plans," "anticipates," "expects," "estimates") should also be considered to be forward-looking statements.
Forward-looking statements involve risks and uncertainties, including, without limitation, risks inherent in the
development and/or commercialization of potential products, risks of cancellation of customer contracts or
discontinuance of trials, risks that anticipated benefits from acquisitions will not be realized, uncertainty in the results of
clinical trials or regulatory approvals, need and ability to obtain future capital, maintenance of intellectual property rights
and other risks discussed in the Cancer Genetics, Inc. Forms 10-K for the year ended December 31, 2015 and 10-Q for
the quarter ended June 30, 2016 along with other filings with the Securities and Exchange Commission. These forward-
looking statements speak only as of the date hereof. Cancer Genetics, Inc. disclaims any obligation to update these
forward-looking statements.

EVERYTHING WE DO AT CGI IS
FOCUSED ON ONE KEY IDEA…
…DELIVER INNOVATION & PATIENT
VALUE BY PROVIDING THE MOST
COMPREHENSIVE CAPABILITIES IN
ONCOLOGY DIAGNOSTICS &
PRECISION MEDICINE
Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
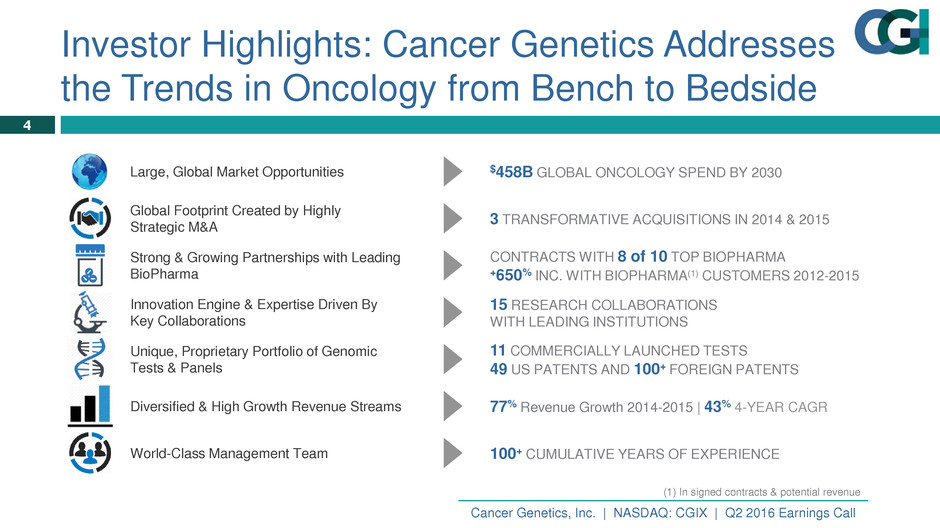
Large, Global Market Opportunities $458B GLOBAL ONCOLOGY SPEND BY 2030
Global Footprint Created by Highly
Strategic M&A 3 TRANSFORMATIVE ACQUISITIONS IN 2014 & 2015
Strong & Growing Partnerships with Leading
BioPharma
CONTRACTS WITH 8 of 10 TOP BIOPHARMA
+650% INC. WITH BIOPHARMA(1) CUSTOMERS 2012-2015
Innovation Engine & Expertise Driven By
Key Collaborations
15 RESEARCH COLLABORATIONS
WITH LEADING INSTITUTIONS
Unique, Proprietary Portfolio of Genomic
Tests & Panels
11 COMMERCIALLY LAUNCHED TESTS
49 US PATENTS AND 100+ FOREIGN PATENTS
Diversified & High Growth Revenue Streams 77% Revenue Growth 2014-2015 | 43% 4-YEAR CAGR
World-Class Management Team 100+ CUMULATIVE YEARS OF EXPERIENCE
Investor Highlights: Cancer Genetics Addresses
the Trends in Oncology from Bench to Bedside
(1) In signed contracts & potential revenue
4

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
2nd Quarter 2016 Revenue Highlights
67% REVENUE GROWTHFROM Q2 2015
Q2 TOTAL REVENUE GREW FROM
$4.2M TO
2015 2016
$1.5
$4.2
$7.0
$0
$4
$7 3-Year
CAGR
67.1%
Q
2
201
4
Q
2
201
5
Q
2
201
6
5

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Revenues By Category
6
BIOPHARMA SERVICES
CLINICAL SERVICES
DISCOVERY SERVICES
64% 30%
6%
Q2
2015
$4.2 Mn
206% increase in clinical test volume over Q2
2015 due to enhanced portfolio in both
immuno-oncology testing and our solid tumor
center of excellence
Driven by support for pre-clinical and
translational initiatives by research
organizations
Launched 11 Immuno-Oncology clinical
trials and studies focused on Solid Tumor
and Blood Cancers
Over 50 new projects closed
$4.2 Mn
$2.5 Mn
$0.2 Mn
60%
36%
4%
Q2
2016
$7.0 Mn

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Q2 2016 Revenue Trends & Drivers
7
Negotiated and executed over 50 new contracts and
project extensions for biopharma testing throughout CGI.
$6+ million in biopharma contracts closed during Q1 2016;
contracts will deliver future revenue during 2016-18.
Total Q2 2016 clinical services test count reached over
7,100 tests, an increase of more than 200% over Q2 2015
and an 8% increase over Q1 2016.
CGI brought its total number of immuno-oncology trials
for testing and validation services to 20, which is up
significantly from 12 during Q1 2016.
3
12
20
Q 4 2 0 1 5 Q 1 2 0 1 6 Q 2 2 0 1 6
# of Immuno-Oncology Trials
37 41
47
Q 4 2 0 1 5 Q 1 2 0 1 6 Q 2 2 0 1 6
Biopharma Signed Contracts
(Expected Revenue $ in Millions)
$ $
$

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Q2 2016 Corporate Highlights
8
Launched a proprietary comprehensive next-generation sequencing panel
FOCUS::Lymphoma™ with CLIA validation to apply in both clinical care
and trials by biotech and pharmaceutical companies.
FOCUS::CLL™ became the first New York State-Approved NGS panel
targeting diagnosis and prognosis in chronic lymphocytic leukemia.
Announced offering of multiple FDA-approved PD-L1 tests, including those
by Dako and Ventana for lung cancer and urothelial cancer, respectively.
Announced the acceptance of a poster at USCAP highlighting the actionable,
clinical value of the FDA-cleared Tissue of Origin test.
Added healthcare industry-veteran John “Jay” Roberts as COO and EVP
of Finance

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Q2 2016 Collaboration Highlights
9
Biopharmaceutical company, H3 Biomedicine Inc. (H3), selected CGI to
provide clinical biomarker services for H3’s lead oncology drug candidate
H3B-8800, an anticancer therapeutic agent being developed for the potential
treatment of select hematologic (blood based) cancers.
CGI and Sayre Therapeutics entered into a distribution agreement to
commercialize CGI’s FDA-cleared Tissue of Origin test throughout India
and South Asia.
Announced results at ASCO with HTG, Keck School of Medicine at USC
and Japan Clinical Cancer Research Organization of using immune-
related genes to identify subtypes of patients associated with clinical
outcome in metastatic colon cancer.

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Our Three Pillars of Innovation
I. UNIQUE CONTENT
VIA PARTNERSHIPS
& COLLABORATIONS
II. IMMUNO-ONCOLOGY
CAPABILITIES THAT
ARE INDUSTRY LEADING
III. LIQUID BIOPSY
HIGH SENSITIVITY
CELL-FREE ANALYSIS
• Mayo Clinic
Oncospire Genomics joint venture targeting hematological cancers
• Columbia University
Genomic signatures for myelodysplastic syndromes and AML
• Huntsman Cancer Center
Patient response to kidney cancer frontline therapies
• HTG Collaboration
Expression of genes implicated in patient immune responses to tumors
• Dako / Ventana CDx
Dako 22C3 and Ventana SP263 CDx test for KEYTRUDA®
• Unique Immunophentotyping Panel
Commercialization of Comprehensive IO Panel via FLOW Cytometry
• Portfolio Updates
Anticipated launch of focused, multi-gene liquid biopsy tests for lung and
renal cancer in upcoming quarters
• Omcomine™Lung cfDNA Assay
Detection levels down to 0.1% with 90% sensitivity and >98% specificity
for point mutations and indels using only a single blood sample
10

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
Summary Statement of Operations
11
Income Statement Item
($ in Thousands)
Q2 2015 Q1 2016 Q2 2016
Revenue $4,185 $6,068 $7,000
Gross Profit 1,088 1,965 2,716
Gross Margin (%) 26% 32% 39%
Research & Development (R&D) 1,256 1,532 1,680
Sales & Marketing (S&M) 1,184 1,298 1,379
General & Administrative (G&A) 3,062 4,318 3,658
Operating Profit (Loss) (4,414) (5,183) (4,001)
Net Income (Loss) (4,980) (5,254) (4,028)
Balance Sheet Information ($ in Thousands) Actual 06/30/16
Cash $10,552
Stockholders’ Equity 29,159

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
12
Revenues were $7.0 million, a 67% increase over
Q2 2015 revenues of $4.2 million, and 15%
sequential growth over Q1 2016.
Revenue from Biopharma Services grew 26% and
Clinical Services increased by 4% sequentially
over Q1 2016.
Gross profit margins improved to 39% an
improvement from 26% in Q2 2015 and a sequential
increase from 32% during Q1 2016.
Total cash was $10.6 million.
Q2 2016 Financial Highlights:
Revenue
$4.0
$5.5
$6.1
$7.0
Q3 2015 Q4 2015 Q1 2016 Q2 2016
Trailing 12-Month Revenue Growth
($ in Millions)

Cancer Genetics, Inc. | NASDAQ: CGIX | Q2 2016 Earnings Call
13
Expenses for the quarter were $11.0 million, a 2.2% improvement over Q1
2016 expenses of $11.3 million.
COGS was $4.3 million, a modest 4% increase over Q1 2016 while revenue
grew 15%.
G&A expenses were $3.7 million, a 15% improvement over Q1 2016.
S&M expenses were $1.4 million.
R&D expenses were $1.7 million, a 10% increase over Q1 2016 as a result
of developing the immuno-oncology offering and validation of the new NGS
panels.
One-time costs during Q2, including severance-related items, were ~$400 K.
Operating loss for Q2 2016 was $4.0 million, and net loss was $4.03
million, a sequential improvement of $1.2 or ~23% over operating and net
losses in Q1 2016.
Q2 2016 Financial Highlights:
Expenses
$-
$1.0
$2.0
$3.0
$4.0
$5.0
$6.0
Q2 2015 Q1 2016 Q2 2016
Operating Loss Net Loss
Operating Loss and Net Loss

THE ONCOLOGY
DIAGNOSTICS
PARTNER FROM
BENCH TO BEDSIDE
WWW.CGIX.COM
WWW.CANCERGENETICS.COM
CGI Headquarters
201 Route 17 North
Rutherford, NJ 07070
Phone: +1 201-528-9200
Fax: +1 201-528-9235
RUTHERFORD, NJ
Research Triangle Park
133 Southcenter Court
Morrisville, NC 27569
Phone: +1 919-465-0100
Fax: +1 919-465-0554
RALEIGH, NC LOS ANGELES, CA
1640 Marengo Street
Fourth Floor
Los Angeles, CA 90033
Phone: +1 323-224-3900
Fax: +1 323-224-3096
#3-1-135/1A CNR Complex
Mallapur Main Road, R.R. Dst.
Hyderabad – 500 076, Telangana
Toll-free: +91 040-2717-8178
Fax: +91 040-2717-8176
HYDERABAD, INDIA
781 Cai Lun Road, Room 803
Shanghai 201203
P.R. China
Toll-free: +91 040-2717-8178
Fax: +91 040-2717-8176
SHANGHAI, CHINA
