Attached files
| file | filename |
|---|---|
| EX-23.1 - EXHIBIT 23.1 - ARGOS THERAPEUTICS INC | exh_231.htm |
| EX-31.2 - EXHIBIT 31.2 - ARGOS THERAPEUTICS INC | exh_312.htm |
| EX-32.1 - EXHIBIT 32.1 - ARGOS THERAPEUTICS INC | exh_321.htm |
| EX-31.1 - EXHIBIT 31.1 - ARGOS THERAPEUTICS INC | exh_311.htm |
| EX-10.31 - EXHIBIT 10.31 - ARGOS THERAPEUTICS INC | exh_1031.htm |
| EX-10.30 - EXHIBIT 10.30 - ARGOS THERAPEUTICS INC | exh_1030.htm |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2015
or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission File Number 001-35443
ARGOS THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
| Delaware | 56-2110007 |
|
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
|
4233 Technology Drive Durham, North Carolina |
27704 |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: (919) 287-6300
Securities registered pursuant to Section 12(b) of the Act:
|
Title of Each Class |
Name of Each Exchange on Which Registered |
| Common Stock, par value $0.001 per share | NASDAQ Global Market |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ¨ | Accelerated filer | ¨ |
| Non-accelerated filer | x (Do not check if a smaller reporting company) | Smaller reporting company | ¨ |
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
As of June 30, 2015 (the last business day of the registrant’s most recently completed second fiscal quarter), the aggregate market value of the registrant’s common stock held by non-affiliates was approximately $65.0 million based upon the closing price for shares of the registrant’s common stock of $6.85 as reported by the NASDAQ Global Market on that date. For purposes of this calculation, the registrant has assumed that its directors, executive officers and holders of 10% or more of the outstanding common stock are affiliates.
As of March 23, 2016, there were 25,330,229 shares outstanding of the registrant’s common stock, par value $0.001 per share.
ARGOS THERAPEUTICS, INC.
ANNUAL REPORT ON FORM 10-K
For the Year Ended December 31, 2015
Table of Contents
Argos Therapeutics®, Argos® and Arcelis™, the Argos Therapeutics logo and other trademarks or service marks of Argos appearing in this Annual Report on Form 10-K are the property of Argos Therapeutics, Inc. The other trademarks, trade names and service marks appearing in this Annual Report on Form 10-K are the property of their respective owners.
| 1 |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K, including “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations,” contains forward-looking statements that involve substantial risks and uncertainties. All statements, other than statements of historical facts, contained in this Annual Report on Form 10-K, including statements regarding our strategy, future operations, future financial position, future revenues, projected costs, prospects, plans and objectives of management, are forward-looking statements. The words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “predict,” “project,” “target,” “potential,” “will,” “would,” “could,” “should,” “continue,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. The forward-looking statements in this Annual Report on Form 10-K include, among other things, statements about:
| • | the progress and timing of our development and commercialization activities; |
| • | the timing and conduct of our ongoing Phase 3 clinical trial of AGS-003 for the treatment of metastatic renal cell carcinoma, or mRCC, and the ongoing and planned investigator-initiated Phase 2 clinical trials of AGS-003, including the timing of the initiation, enrollment and completion of the trials and the availability of data from the trials; |
| • | the timing and conduct of the ongoing investigator-initiated Phase 2 clinical trial of AGS-004 for HIV eradication and the planned investigator-initiated Phase 2 clinical trial of AGS-004 for long-term viral control in pediatric patients, including the timing of the initiation, enrollment and the completion of the trials and the availability of data from the trials; |
| • | our ability to obtain U.S. and foreign marketing approval for AGS-003 for the treatment of mRCC and for AGS-004 for the treatment of HIV, and the ability of these product candidates to meet existing or future regulatory standards; |
| • | the potential benefits of our Arcelis platform and our Arcelis-based product candidates; |
| • | our ability to build out and equip a North American commercial manufacturing facility and supply on a commercial scale our Arcelis-based products or, alternatively, to enter into a contract manufacturing arrangement to supply on a commercial scale our Arcelis-based products; |
| • | our intellectual property position and strategy; |
| • | our expectations related to the sufficiency of our cash, cash equivalents and short-term investments; |
| • | the accuracy of our estimates of the size and characteristics of the markets that may be addressed by our product candidates; |
| • | our ability to establish and maintain collaborations for the development and commercialization of our product candidates; |
| • | developments relating to our competitors and our industry; and |
| • | the impact of government laws and regulations. |
We have based these forward-looking statements largely on our current plans, intentions, expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements we make. We have included important factors in the cautionary statements included in this Annual Report on Form 10-K, particularly in “Item 1A. Risk Factors,” that could cause actual results or events to differ materially from the forward-looking statements that we make. Our forward-looking statements do not reflect the potential impact of any future acquisitions, mergers, dispositions, collaborations, joint ventures or investments that we may make or enter into.
You should read this Annual Report on Form 10-K with the understanding that our actual future results may be materially different from what we expect. The forward-looking statements contained in this Annual Report on Form 10-K are made as of the date of filing of this Annual Report on Form 10-K, and we do not assume any obligation to update any forward-looking statements, except as required by applicable law.
This Annual Report on Form 10-K includes statistical and other industry and market data that we obtained from industry publications and research, surveys and studies conducted by third parties. Industry publications and third party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. This Annual Report on Form 10-K also includes data based on our own internal estimates. While we believe these industry publications and third party research, surveys and studies are reliable, we have not independently verified such data.
| 2 |
We are an immuno-oncology company focused on the development and commercialization of truly individualized immunotherapies for the treatment of cancer and infectious diseases based on our proprietary technology platform called Arcelis.
Our most advanced product candidate is AGS-003, which we are developing for the treatment of metastatic renal cell carcinoma, or mRCC, and other cancers. We are currently conducting a pivotal Phase 3 clinical trial of AGS-003 plus sunitinib or another targeted therapy for the treatment of newly diagnosed mRCC under a special protocol assessment, or SPA, with the Food and Drug Administration, or FDA. We refer to this trial as the ADAPT trial. We initiated the ADAPT trial in January 2013, dosed the first patient in May 2013 and completed enrollment of the trial in July 2015. Based upon the actual rate of enrollment and projected event rate as defined in the protocol, we anticipate having a sufficient number of events to permit the primary analysis and assessment of overall survival to occur in the first half of 2017. The independent data monitoring committee for the ADAPT trial has twice recommended the continuation of the ADAPT trial based on results of its data analyses for safety and futility. We expect the independent data monitoring committee to meet again in June 2016 and approximately six months later in 2016 to conduct additional data analyses. We are also supporting investigator-initiated Phase 2 trials in patients with early stage RCC and non-small cell lung cancer and plan to support investigator-initiated trials of AGS-003 in mRCC and bladder cancer.
We are developing AGS-004, our second most advanced Arcelis-based product candidate, for the treatment of HIV. We have completed Phase 1 and Phase 2 trials funded by government grants and a Phase 2b trial that was funded in full by the National Institutes of Health, or NIH, and the National Institute of Allergy and Infectious Diseases, or NIAID, under a $39.8 million agreement. We are currently supporting an ongoing investigator-initiated Phase 2 clinical trial of AGS-004 in adult HIV patients evaluating the use of AGS-004 in combination with a latency reversing drug for HIV eradication, and plan to support a second investigator-initiated Phase 2 clinical trial of AGS-004 evaluating AGS-004 for long-term viral control in pediatric patients.
Our Arcelis Platform
Our proprietary Arcelis technology platform utilizes biological components from a patient’s own cancer cells or virus to generate truly individualized immunotherapies. These immunotherapies employ specialized white blood cells called dendritic cells to activate an immune response specific to the patient’s own disease. Arcelis is based on the work of Dr. Ralph Steinman, winner of the 2011 Nobel Prize in medicine for the discovery of the role of dendritic cells in the immune system. We believe our Arcelis-based immunotherapies are applicable to a wide range of cancers and infectious diseases and have the following attributes that we consider critical to a successful immunotherapy:
| • | target a patient’s disease-specific antigens, including mutated antigens, or neoantigens, to elicit a potent immune response that is specific to the patient’s own disease; |
| • | overcome the immune suppression that exists in cancer and infectious disease patients; |
| • | induce memory T-cells, a specialized type of immune cell that is known to correlate with improved clinical outcomes for cancer and HIV patients; |
| • | have minimal toxicity; and |
| • | can be produced using a centralized manufacturing process at a cost that will be comparable to biologics. |
We believe that our immunotherapies combine the advantages of other approaches to immunotherapy, including antigen-based approaches and pathway-based approaches such as checkpoint inhibition, while addressing limitations that they present.
| 3 |
Our Development Programs
The following table summarizes our development programs for AGS-003 and AGS-004.
| Product Candidate | Primary Indication | Status | |||
| AGS-003 | mRCC | • | Ongoing ADAPT trial; enrollment completed in July 2015; final data expected in the first half of 2017 | ||
| • | Investigator-initiated Phase 2 clinical trial, in combination with a PD-1 inhibitor, expected to begin in the second half of 2016 | ||||
| Early stage RCC (neoadjuvant) | • | Ongoing investigator-initiated Phase 2 clinical trial; initial data expected in fourth quarter of 2016 | |||
| Advanced solid tumors | • | Ongoing investigator-initiated Phase 2 clinical trial in non-small cell lung cancer initiated in the first quarter of 2016 | |||
| • | Investigator-initiated Phase 2 clinical trial in muscle invasive bladder cancer expected to begin in mid-2016 | ||||
| AGS-004 | HIV | • | Ongoing second stage of investigator-initiated Phase 2 clinical trial for HIV eradication | ||
| • | Investigator-initiated Phase 2 clinical trial for long-term viral control in pediatric patients expected to begin in 2017 | ||||
We hold all commercial rights to AGS-003 and AGS-004 in all geographies other than rights to AGS-003 in Russia and the other states comprising the Commonwealth of Independent States, which we exclusively licensed to Pharmstandard International S.A., or Pharmstandard, rights to AGS-003 for the treatment of mRCC, in South Korea, which we exclusively licensed to Green Cross Corp., or Green Cross, and rights to AGS-003 in China, Hong Kong, Taiwan and Macau, which we exclusively licensed to Lummy (Hong Kong) Co. Ltd., or Lummy HK. We have granted to MEDcell Co., Ltd., a wholly-owned subsidiary of Medinet Co. Ltd., hereinafter referred to together as “Medinet,” an exclusive license to manufacture in Japan AGS-003 for the treatment of mRCC and an option to acquire a non-exclusive license to sell in Japan AGS-003 for the treatment of mRCC. We do not expect Medinet to exercise this option before its expiration on March 31, 2016.
AGS-003
We are developing AGS-003 for the treatment of mRCC and other cancers. We are currently conducting the ADAPT trial of AGS-003 plus sunitinib / targeted therapy for the treatment of newly diagnosed mRCC under an SPA with the FDA. We initiated the ADAPT trial in January 2013 and dosed the first patient in May 2013. In July 2015 we completed enrollment in the trial, enrolling 462 patients with the goal of generating 290 events for the primary endpoint of overall survival. We enrolled these patients at 127 clinical sites in North America, Europe and Israel. Under the trial protocol, these patients were randomized between the AGS-003 plus sunitinib / targeted therapy combination arm and the sunitinib / targeted therapy alone control arm on a two-to-one basis. Based upon the actual rate of enrollment and projected event rate as defined in the protocol, we anticipate having a sufficient number of events to permit the primary analysis and assessment of overall survival to occur in the first half of 2017.
In addition, in mRCC we plan to support an investigator-initiated clinical trial of AGS-003 in combination with a PD-1 inhibitor that we expect will begin in the second half of 2016.
We are also supporting an ongoing investigator-initiated Phase 2 clinical trial designed to evaluate treatment with AGS-003 in patients with early stage RCC prior to nephrectomy. This trial was open for enrollment in late 2014 and 4 patients were enrolled as of March 15, 2016. We expect that a total of 10 patients will be enrolled in this trial.
We believe that AGS-003 may be capable of treating a wide range of cancers and are planning to evaluate AGS-003 in clinical trials in additional cancer indications. We are supporting an investigator-initiated Phase 2 clinical trial of AGS-003 in patients with non-small cell lung cancer, or NSCLC, that was initiated in the first quarter of 2016. In the trial, the safety, efficacy and immunologic effects of AGS-003 when combined with platinum-based chemotherapy and radiation will be evaluated in approximately 20 stage 3 NSCLC patients. In the trial, AGS-003 will be administered either concurrently with chemotherapy and with or without radiation or sequentially with chemotherapy and with or without radiation, according to the patient’s assigned treatment arm. The rationale for this trial is two-fold. First, NSCLC is a tumor type reported to have a high number of mutated targets for the immune system, which could make it more susceptible to AGS-003’s mechanism of action. Secondly, platinum-based chemotherapy has immunomodulatory effects by downregulating immunosuppressive regulatory T cells which may lead to an additive or synergistic effect with AGS-003. We also plan to support an additional investigator-initiated Phase 2 clinical trial of AGS-003 in muscle invasive bladder cancer, which we expect may begin in mid-2016. The trial has two phases: a pre-treatment phase and a treatment phase. In the pre-treatment phase, tumor tissue will be obtained via a transurethral resection of the bladder tumor, which will then be used to extract RNA for the manufacture of AGS-003. In the treatment phase, AGS-003 will be given before tumor resection and combined with standard-of-care cytotoxic chemotherapy. Booster doses of AGS-003 will continue after tumor resection. As with the neoadjuvant renal cancer trial, we have the unique opportunity to observe any meaningful impact of AGS-003 on the immune response in the peripheral blood and immune responses infiltrating the primary tumor.
| 4 |
AGS-004
We are developing AGS-004 for the treatment of HIV and are focusing this program on the use of AGS-004 in combination with other therapies for the eradication of HIV. We believe that by combining AGS-004 with therapies that are being developed to expose the virus in latently infected cells to the immune system, we can potentially eradicate the virus. The current standard of care, antiretroviral drug therapy, or ART, can reduce levels of HIV in a patient’s blood, increase the patient’s life expectancy and improve the patient’s quality of life. However, ART cannot eliminate the virus, which persists in latently infected cells, remains undetectable by the immune system and can recur. In addition, ART requires daily, life-long treatment and can have significant side effects.
We are supporting an investigator-initiated Phase 2 clinical trial of AGS-004 in up to 12 adult HIV patients to evaluate the use of AGS-004 in combination with one of these latency reversing therapies for the eradication of HIV at the University of North Carolina. This trial is being conducted in two stages. Stage 1 of this trial has been completed and was designed to study immune response kinetics to AGS-004 in patients on continuous ART. These data were used to better define the optimal dosing strategy in combination with a latency reversing therapy in the ongoing Stage 2. We expect that some patients in Stage 1 will rollover into Stage 2, which is studying AGS-004 in combination with one of the latency reversing drugs. The patient clinical costs for the first stage of this trial were funded by Collaboratory of AIDS Researchers for Eradication, or CARE. The NIH Division of AIDS has approved $6.6 million in funding for the second stage of this trial.
We also plan to explore the use of AGS-004 monotherapy to provide long-term control of HIV viral load in otherwise immunologically healthy patients and eliminate their need for ART. Accordingly, we plan to support an investigator-initiated Phase 2 clinical trial of AGS-004 monotherapy in pediatric patients infected with HIV who have otherwise healthy immune systems and have been treated with ART since birth or shortly thereafter and, as a result, are lacking the antiviral memory T-cells to combat the virus. The commencement of this trial is subject to supportive data obtained from the adult eradication trial and approval of the protocol by the principal investigator(s), institutional review boards, the IMPAACT Network leadership and the FDA and to the agreement by the NIH to fund the trial costs not related to AGS-004 manufacturing. Assuming the supportive data and the necessary approvals are obtained, we expect this trial to initiate in 2017.
Strategy
Our goal is to become a leading biopharmaceutical company focused on discovering, developing and commercializing truly individualized immunotherapies for the treatment of a wide range of cancers. Key elements of our strategy are:
| • | complete clinical development and seek marketing approvals of AGS-003 for the treatment of mRCC; |
| • | expand clinical development of AGS-003 in other cancers, including early stage RCC and advanced solid tumors; |
| • | commercialize AGS-003 in North America independently and with third parties outside North America; |
| • | build out and equip a North American commercial manufacturing facility to manufacture and supply on a commercial scale our Arcelis-based products or, alternatively, enter into a contract manufacturing arrangement with a third party to manufacture and supply on a commercial scale our Arcelis-based products; |
| • | continue clinical development of AGS-004 for the treatment of HIV, potentially through government funding or other third party funding, and collaborate with third parties for commercialization on a worldwide basis; and |
| • | pursue expansion of our broad intellectual property protection for our Arcelis technology platform, product candidates and proprietary manufacturing processes through U.S. and international patent filings and maintenance of trade secret confidentiality. |
Immunotherapy to Treat Cancer and Infectious Diseases
Cancer cells occur frequently in the human body, yet are effectively controlled by T-cells in the immune system, which recognize proteins produced by the cancer cells, known as antigens, as abnormal and kill the associated cancer cells. Two specific types of T- cells are necessary for an effective anti-cancer immune response: CD8+ T-cells, which kill cancer cells, and CD4+ T-cells, which provide a “help” signal that activates and directs the CD8+ T-cell response.
| 5 |
Cancer cells utilize several strategies to escape detection by the immune system and T-cells. For example, cancer cells secrete factors that act systemically to prevent T-cells from responding to activation signals, resulting in the inability of T-cells to carry out their role of killing cancer cells. Chronic viral infections such as HIV or hepatitis C present the same challenges to the immune system as cancer because the immune system must overcome this disease-induced immune suppression to recognize and respond to virus-infected cells.
Immunotherapy is intended to stimulate and enhance the body’s natural mechanism for recognizing and killing cancer cells and virus-infected cells. Current immunotherapeutic approaches to treat cancer can generally be separated into two different mechanisms of action: antigen-based approaches that target one or more specific antigens and pathway-based approaches that target specific immunologic pathways.
Antigen-Based Approaches
Cancer immunotherapies that use an antigen-based approach are designed to stimulate an immune response against one or more tumor-associated antigens. In most cases, the tumor-associated antigens that are being targeted are non-mutated, or normal, antigens, which are usually well tolerated by the immune system. In the context of cancer, these normal antigens are either produced at abnormally high levels or predominantly in tumor cells, or both. The goal of antigen-based immunotherapies is to activate the patient’s own immune system to seek out and kill the cancer cells that carry the targeted antigen. Dendreon Corporation’s Provenge (sipuleucel-T) for metastatic castrate-resistant prostate cancer is the only antigen-based immunotherapy that has been approved by the FDA. Because these immunotherapies are designed to target specific antigens, they are less likely to have toxicity. However, antigen-based immunotherapies may have limited efficacy because they are only able to capture one or a limited number of antigens, which may or may not be present in the patient’s cancer cells, and do not capture mutated antigens.
Pathway-Based Approaches
Immunotherapies that rely on the pathway approach are designed to overcome immunosuppression in patients by blocking signaling pathways that prevent T-cell activation and function. The class of monoclonal antibody-based immunotherapies known as checkpoint inhibitors are being developed on the basis of this approach. For example, Bristol-Myers Squibb’s immunotherapy Yervoy (ipilimumab), an FDA-approved treatment for patients with unresectable or metastatic melanoma, is designed to act by blocking the function of a protein expressed in activated T-cells called CTLA4, which acts as a T-cell “off” switch. By blocking the function of CTLA4, the patient’s T-cells can become activated, resulting in an immune response against tumors. Another pathway that immunotherapies are being developed to address is the PD-1/PD-L1 pathway. In this pathway, activated T-cells expressing the protein PD-1 are disabled when binding occurs between PD-1 and its ligand, PD-L1, which is expressed on tumor cells. Immunotherapies are being developed to interrupt this pathway by binding to the PD-1 protein or the PD-L1 ligand to prevent them from binding with each other. Two of these anti-PD-1 checkpoint inhibitors were recently approved by FDA: Merck’s Keytruda for NSCLC and advanced melanoma and Bristol-Myers Squibb’s Opdivo for NSCLC, advanced melanoma and mRCC. Immunotherapies that use a pathway approach have demonstrated the ability to effectively overcome immunosuppression and enable T-cells to function against tumor cells and potentially virus-infected cells. . However, pathway-based immunotherapies are limited because they act systemically to enable T-cells to function and do not specifically target a patient’s tumor or the associated antigens. This lack of specificity can negatively impact healthy tissue and generate unwanted toxicity.
Designing Immunotherapies Using Our Arcelis Platform
We believe that our proprietary Arcelis platform enables us to produce truly individualized immunotherapies that combine the advantages of the antigen-based and pathway-based approaches to immunotherapy while addressing the limitations and disadvantages of these approaches. We have designed our Arcelis platform to create product candidates which have attributes that we believe are critical to a successful immunotherapy:
| • | Target disease-specific antigens, including mutated antigens. The immunotherapy should target antigens, including unique mutated antigens, associated with the patient’s disease. We believe that immunotherapies that target only non-mutated, or normal, tumor-associated antigens will be limited in terms of efficacy as non-mutated antigens are generally poor at stimulating immune responses. Our Arcelis platform uses messenger RNA, or mRNA, from the patient’s own cancer or virus to yield a truly individualized immunotherapy that contains the patient’s disease-specific antigens, including mutated antigens, and is designed to elicit a potent immune response specific to the patient’s own disease. |
| • | Overcome disease-induced immune suppression. The immunotherapy must be able to generate an effective immune response in patients whose immune systems are compromised by their disease. Both tumors and HIV are known to impair the functionality of CD4+ T helper cells, which aid their escape from CD8+ T-cell attack. Our Arcelis-based immunotherapies do not require CD4+ helper T-cells to mount an immune response with effective anti-tumor or anti- viral activity as we add the protein known as CD40 ligand, or CD40L, to provide the signaling that the CD4+ helper T-cells would otherwise provide. |
| 6 |
| • | Induce memory T-cells. The immunotherapy should be able to induce specific T-cells, such as CD8+CD28+ memory T-cells, which are known to correlate with improved clinical outcomes for cancer and HIV patients. These memory T-cells are long lived and necessary for a durable immune response. Our Arcelis process produces dendritic cells that secrete IL-12, which is necessary to induce and expand patient-specific CD8+CD28+ memory T-cells. These memory T-cells are able to seek out and kill cancer or virus-infected cells that express the antigens identical to those displayed on the surface of the dendritic cells. In addition, because these newly generated memory T-cells do not express PD-1, they are not subject to inhibition by the PD-1/PD-L1 pathway. |
| • | Have minimal toxicity. The immunotherapy should have minimal toxicity, which would potentially enable it to be combined with other therapies for cancer and infectious diseases. The mechanism of action of Arcelis-based products induces patient- and disease-specific memory T-cells. This target customization and specificity is less likely to impact healthy tissue and cause toxicity. Our Arcelis-based product candidates have been well tolerated in clinical trials in more than 375 patients with no serious adverse events attributed to our immunotherapies. |
Our Arcelis platform is focused on dendritic cells which present antigens to the attention of the human immune system, including, in particular, T-cells, and are critical to the immune system’s recognition of proteins derived from cancer cells or virus-infected cells. Dendritic cells are capable of internalizing cancer or virus protein antigens and displaying fragments of these protein antigens on their surface as small peptides. The dendritic cells then present these peptide antigens to T-cells. This allows the T-cells to bind to these peptide antigens and, in the case of cancer, kill cancer cells and, in the case of infectious disease, kill virus-infected cells to control the spread of infectious pathogens.
The following graphic illustrates the processes comprising our Arcelis platform:
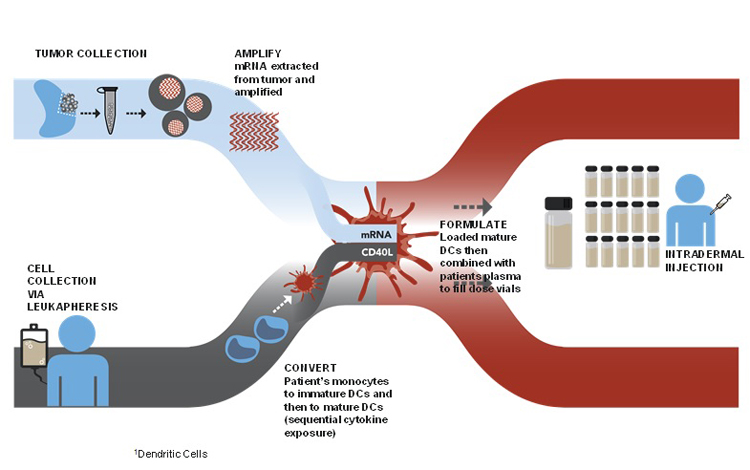
At the clinical site. As shown in the graphic above, the manufacture of our Arcelis-based immunotherapies requires two components derived from the patient:
| • | A disease sample: In the case of cancer, the sample consists of tumor cells, and in the case of infectious disease, the sample consists of blood containing the virus. The disease sample is generally collected at the time of diagnosis or initial treatment. |
| • | Monocytes: Monocytes are a type of white blood cell, which are obtained through a laboratory procedure called leukapheresis that occurs after diagnosis and at least three weeks prior to initiating treatment with our immunotherapy. |
| 7 |
At our centralized manufacturing facility. The tumor cells or the blood sample and the leukapheresis product are shipped to our centralized manufacturing facility following collection at the clinical site. After receipt of these components at our facility, we take the following steps:
| • | We isolate the patient’s disease mRNA, which carries the genetic information to recreate the patient’s disease antigens, from the disease sample and amplify the mRNA so that only a small disease sample is required to manufacture the immunotherapy. |
| • | Separately, we extract the monocytes from the leukapheresis product and culture them using a proprietary process to produce matured dendritic cells. |
| • | We then combine the matured dendritic cells with a solution of the patient’s isolated mRNA and a proprietary synthetic CD40L RNA. We apply a brief electric pulse to the solution in a process referred to as electroporation, which enables the patient’s mRNA and the CD40L RNA to pass into, or load, the dendritic cells. The dendritic cells process the CD40L RNA into CD40L protein, enabling the dendritic cells to secrete IL-12, a cytokine required to induce and expand CD8+CD28+ memory T-cells. |
| • | We then further culture the mRNA-loaded dendritic cells so that these cells allow for antigen expression from the patient’s mRNA and presentation in the form of peptides on the surface of the dendritic cells. These mature, loaded dendritic cells are formulated using the patient’s plasma that was collected during the leukapheresis to become the Arcelis-based product. Typically, several years of doses are produced for each patient. |
| • | After verifying the quality of the product, we vial, cryogenically freeze and then ship individual patient doses to the clinic, where each is thawed and administered by intradermal injection. |
Patient treatment. Upon injection into the skin of the patient, the mature, loaded dendritic cells migrate to the lymph nodes near the site of the injection. It is at these lymph nodes that the dendritic cells come into contact with T-cells. This interaction with the loaded dendritic cells is intended to cause a measurable increase in patient- and disease-specific memory T-cells.
We believe that our Arcelis platform allows us to create truly individualized immunotherapies capable of treating a wide range of cancers and infectious diseases using a manufacturing process that we expect will be at a cost that is comparable to other biologics. Specifically, our Arcelis platform typically allows us to:
| • | produce several years of customized therapy for a patient from a small disease sample and a single leukapheresis from that patient; |
| • | produce additional years of therapy for a patient at a later date without requiring an additional disease sample from the patient; |
| • | use a centralized manufacturing facility for North America, which is possible because our Arcelis process can utilize monocytes obtained through leukapheresis within four days of the procedure, and doses of our immunotherapies can be shipped frozen in a cryoshipper that can maintain the target temperature for at least ten days; |
| • | cryopreserve the multiple doses generated from the single manufacturing process for each patient in a direct injectable formulation that allows the doses to remain stable and usable for up to five years; and |
| • | produce immunotherapies that can be administered by intradermal injection in an outpatient procedure. |
AGS-003 for the Treatment of Metastatic Renal Cell Carcinoma and Other Cancers
We are initially developing AGS-003 for use in combination with sunitinib and other targeted therapies for the treatment of mRCC. Sunitinib is an oral small molecule drug sold under the trade name Sutent and is the current standard of care for initial treatment, or first-line treatment, of mRCC following diagnosis. In April 2012, the FDA notified us that we have obtained fast track designation for AGS-003 for the treatment of mRCC.
We are currently conducting the ADAPT trial of AGS-003 plus sunitinib / targeted therapy compared to sunitinib/targeted therapy monotherapy for the treatment of newly diagnosed mRCC under an SPA with the FDA. In July 2015 we completed enrollment in the trial, enrolling 462 patients with the goal of generating 290 events for the primary endpoint of overall survival. Based upon the actual rate of enrollment and projected event rate as defined in the protocol, we anticipate having a sufficient number of events to permit the primary analysis and assessment of overall survival to occur in the first half of 2017. The independent data monitoring committee for the ADAPT trial has twice recommended the continuation of the ADAPT trial based on results of its data analyses for safety and futility. We expect the independent data monitoring committee to conduct additional data analyses of the trial in June 2016 and approximately six months later in 2016.
| 8 |
We are supporting investigator-initiated Phase 2 clinical trials designed to evaluate treatment with AGS-003 in patients with early stage RCC prior to nephrectomy and in patients with NSCLC and plan to support investigator-initiated Phase 2 clinical trials of AGS-003 in mRCC and muscle invasive bladder cancer.
Renal Cell Carcinoma
RCC is the most common type of kidney cancer. The American Cancer Society, or ACS estimates that there will be approximately 63,000 new cases of kidney cancer and approximately 14,000 deaths from this disease in the United States in 2016. The National Comprehensive Cancer Network, or NCCN estimates that 90% of kidney cancer cases are RCC. For patients with RCC that had metastasized by the time RCC was first diagnosed, a condition referred to as newly diagnosed mRCC, the five-year survival rate has historically been approximately 12%.
ACS statistics indicate that approximately 25% of newly diagnosed RCC patients present with mRCC annually in the United States. Additional patients who were initially diagnosed with earlier stage RCC may also progress to mRCC as these patients suffer relapses. The NCCN estimates between 20% to 30% of patients with early stage RCC will relapse within three years of surgical excision of the primary tumor. Although the National Cancer Institute, or NCI does not provide prevalence of RCC by stage, based on the NCCN’s three-year relapse rate, we estimate that there may be up to an additional 10,000 to 15,000 cases of mRCC identified annually in the United States. Combining newly diagnosed mRCC patients with patients who relapse, we estimate that there may be between 20,000 to 25,000 new cases of mRCC in the United States each year. We estimate, based on publicly available information, including 2013 quarterly and annual reports of companies that market targeted therapies approved for mRCC, that the current worldwide mRCC market for these targeted therapies exceeds $2 billion.
Physicians generally diagnose mRCC by examining a tumor biopsy under a microscope. Upon evaluation of the visual appearance of the tumor cells, a pathologist will classify the mRCC into clear cell or non-clear cell types. According to the NCCN, approximately 80% of all RCC diagnoses are clear cell RCC. Because clear cell types are the most common type of tumor cell, most of the more recently approved therapies for mRCC have limited their clinical trials to patients with the clear cell type of tumor cell. However, the FDA has not limited the approval of these therapies to clear cell types of mRCC, so they may be used for both clear cell and non-clear cell types.
mRCC Patient Classification
Upon diagnosis, the prognosis for patients with mRCC is classified into three overall disease risk profiles — favorable, intermediate and poor — using objective prognostic risk factors. These risk factors were originally developed by researchers at Memorial Sloane Kettering Cancer Center and subsequently revised by Dr. Daniel Heng from the University of Calgary’s Baker Cancer Center and contributors from the International Metastatic Renal Cell Carcinoma Database Consortium, or the Consortium based on clinical data from patients treated with sunitinib and other targeted therapies. These risk factors, which we refer to as the Heng risk factors, have been correlated to adverse overall survival in mRCC and include:
| • | time from diagnosis to the initiation of systemic therapeutic treatment of less than one year, which is indicative of more aggressive disease. We refer to this risk factor as the less than one year to treatment risk factor; |
| • | low levels of hemoglobin, a protein in the blood that carries oxygen; |
| • | elevated corrected calcium levels; |
| • | diminished overall patient performance status or physical functioning; |
| • | elevated levels of neutrophils, a type of white blood cell; and |
| • | elevated platelet count. |
Patients exhibiting zero risk factors at the time of treatment are included in the favorable risk group; patients exhibiting one or two risk factors are included in the intermediate risk group; and patients exhibiting three or more risk factors are included in the poor risk group. Even when treated with standard of care therapies such as sunitinib, patients in the intermediate risk group have an expected survival of less than two years, and patients in the poor risk group have an expected survival of less than one year. In January 2013, Dr. Heng published in Lancet Oncology the following data from the Consortium database regarding overall survival of mRCC patients in these three risk groups treated with sunitinib and other targeted therapies:
| • | in 157 favorable risk patients, the median overall survival was 43.2 months; |
| 9 |
| • | in 440 intermediate risk patients, the median overall survival was 22.5 months; and |
| • | in 252 poor risk patients, the median overall survival was 7.8 months. |
Current Treatment
The initial treatment for most mRCC patients when the primary tumor is intact is surgical removal of the tumor, usually requiring partial or complete removal of the affected kidney, referred to as nephrectomy. The NCCN generally recommends systemic treatment with approved therapies for mRCC patients following nephrectomy for patients whose tumors have metastasized or for patients who present with mRCC upon diagnosis or as a result of a relapse from an earlier stage of RCC.
Historically, mRCC has been treated with non-specific, cytokine-based immunotherapies such as interferona and IL-2, which have demonstrated a clinical benefit in a small number of mRCC patients. However, these therapies lack specificity and have been demonstrated to have severe toxicities, which can lead to cardiopulmonary, neuropsychiatric, dermatologic, renal, hepatic and hematologic side effects and limits their use. For example, although high-dose IL-2 is the only therapy to have demonstrated durable complete mRCC remissions, its toxicity restricts its use to a small minority of patients and for a short duration.
Several targeted therapies, such as Sutent (sunitinib), Votrient (pazopanib), Torisel (temsirolimus), Nexavar (sorafenib), Avastin (bevacizumab) plus interferon-a, Afinitor (everolimus) and Inlyta (axitinib), are approved for the treatment of mRCC. While most of these targeted therapies have been evaluated in first-line treatment of mRCC, Sutent demonstrated a higher rate of progression free survival and overall survival in its pivotal Phase 3 clinical trial than that shown by the other targeted therapies in their pivotal Phase 3 clinical trials. According to an independent market research survey conducted during the second half of 2014 of 87 US-based medical oncologists and new prescription data (IMS), Sutent is still the first-line drug of choice for approximately half of newly treated advanced RCC patients. In addition, the data showed that the use of Votrient has been increasing as initial therapy for advanced RCC.
Although most of these targeted therapies have demonstrated prolonged progression free survival as compared to interferon-a, they are rarely associated with durable remissions or enhanced long-term survival, particularly in patients who are classified as intermediate or poor risk at the time of treatment. In addition, each of these targeted therapies has shortcomings that limit their use in the treatment of mRCC, including significant toxicities, such as neutropenia and other hematologic toxicities, fatigue, diarrhea, hand-foot syndrome, hypertension and other cardiovascular effects. The overlapping and combined toxicities of the targeted therapies have prevented their use in combination therapies. For instance, researchers conducting a Phase 1 clinical trial of the combination of sunitinib and temsirolimus discontinued the trial due to toxicities. We believe that the inability to date to combine these therapies without additive toxicity and the absence of durable remissions and prolonged survival in patients with intermediate and poor risk disease indicates there is an unmet need for novel therapeutic approaches for mRCC that can improve efficacy without adding any appreciable toxicity.
AGS-003 Opportunity
We believe, based on the clinical results of AGS-003, that the combination of AGS-003 with sunitinib or other targeted therapies has the potential to address this unmet need for the following reasons:
| • | We believe that because the mechanism of action of AGS-003 is unrelated to the mechanism of action of sunitinib or the other targeted therapies, combining AGS-003 with these therapies has the potential to have an additive efficacy benefit. |
| • | We believe that if a combination therapy with AGS-003 shows improved efficacy, the combination could be used as the standard of care for first-line treatment of mRCC in our targeted patient population. |
| • | We believe that the lack of significant toxicity of AGS-003 will enable it to be combined with sunitinib and the other targeted therapies at a full dose for both therapies without added toxicity. |
| • | We believe that, by following an initial cycle of sunitinib with AGS-003 in combination therapy, patients are likely to have a lower metastatic tumor burden, or at least a slowing of tumor progression, at the time of initiation of AGS-003 therapy, making it more likely that AGS-003 would have the opportunity to elicit immune responses and demonstrate an effect on the tumor. |
| • | We believe that continued dosing of sunitinib, as well as certain of the other targeted therapies, decreases regulatory T-cells and myeloid-derived suppressor cells, both of which are immunosuppressive cells known to expand during cancer and suppress T-cell responses. As a result, by combining with these therapies, we believe that AGS-003 may be able to generate more potent T-cell responses. |
| 10 |
Development Status
We are conducting an ongoing pivotal Phase 3 clinical trial of AGS-003. We have previously conducted three clinical trials of AGS-003 and its predecessor product, including a Phase 2 trial and 2 Phase 1 trials. To date, we have administered AGS-003 to over 300 patients in these trials. We submitted to the FDA an investigational new drug application, or IND, for AGS-003 in March 2003.
Pivotal Phase 3 Combination Therapy Clinical Trial. We are currently conducting a pivotal Phase 3 clinical trial of AGS-003 plus sunitinib / targeted therapy for the treatment of newly diagnosed mRCC under an SPA with the FDA. We refer to this trial as the ADAPT trial. We initiated the ADAPT trial in January 2013, dosed the first patient in May 2013 and completed enrollment of the trial in July 2015. Based upon the actual rate of enrollment and projected event rate as defined in the protocol, we anticipate having a sufficient number of events to permit the primary analysis and assessment of overall survival to occur in the first half of 2017.
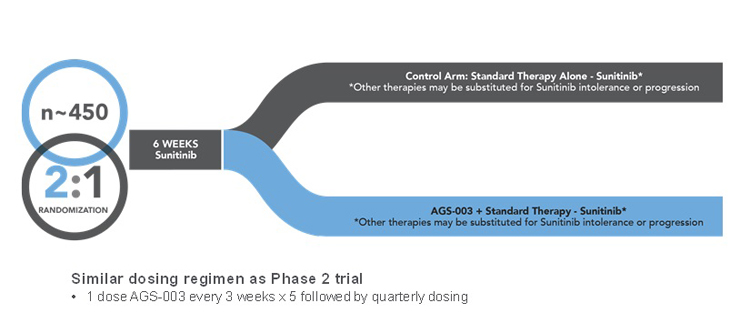
We have designed this trial to be a randomized, multicenter, open label trial of AGS-003 in combination with sunitinib / targeted therapy compared to sunitinib / targeted therapy monotherapy. We enrolled 462 patients with the goal of generating 290 events for the primary endpoint of overall survival. We enrolled these patients at 127 clinical sites in North America, Europe and Israel. Under the trial protocol, these patients were randomized between the AGS-003 plus sunitinib / targeted therapy combination arm and the sunitinib / targeted monotherapy control arm on a two-to-one basis. The independent data monitoring committee for the ADAPT trial twice has recommended the continuation of the ADAPT trial based on results of its interim data analyses for safety and futility. We expect the independent data monitoring committee to conduct additional data analyses in June 2016 and approximately six months later in 2016.
We have designed this trial with a primary endpoint of overall survival. Secondary endpoints include progression free survival, overall response rate and safety. In order to achieve the primary endpoint, results from the trial must demonstrate an increase of approximately six months in median overall survival for the AGS-003 plus sunitinib arm compared to the sunitinib monotherapy arm. Such a result would be statistically significant (p £ 0.05).
Our design for this trial requires adult patients who have been newly diagnosed with mRCC with primary tumor intact and metastatic disease following nephrectomy, who have predominantly clear cell RCC based upon the tumor collected at nephrectomy and who have not received any prior therapies for RCC. Participating patients must be suitable candidates for sunitinib therapy and possess poor risk or intermediate risk disease at presentation, with the less than one year to treatment risk factor and not more than four Heng risk factors in total. As part of the trial design, the two arms of the trial are balanced based upon known prognostic risk factors. Patients are stratified by number of risk factors (1, 2, 3 or 4) as well as whether they had measurable versus non-measurable metastatic disease following nephrectomy. The patient population in the pivotal Phase 3 clinical trial is generally comparable to the patient population treated in our Phase 2 combination therapy clinical trial. Approximately 73% of the patients enrolled in the trial are intermediate risk patients (1-2 risk factors) and 27% are poor risk (3-4 risk factors).
Under the trial protocol, patients in the AGS-003 plus sunitinib / targeted therapy arm are dosed with AGS-003 once every three weeks for five doses, followed by a booster dose every three months. In accordance with its label, sunitinib dosing is administered in six-week cycles, consisting of four weeks on drug and two weeks on drug holiday. AGS-003 dosing is initiated at the end of the initial six-week sunitinib cycle. The first dose of AGS-003 is administered prior to the start of sunitinib dosing in the second sunitinib cycle. This dosing regimen is identical to the dosing regimen used in our Phase 2 combination therapy clinical trial of AGS-003 and sunitinib, except that the start of the sixth dose is scheduled for week 24 to better provide patients the opportunity to receive a total of eight doses across 48 weeks. Patients in the sunitinib monotherapy control arm receive sunitinib on the same dosing schedule as patients receive sunitinib in the AGS-003-sunitinib combination arm.
| 11 |
Under the trial protocol, AGS-003 is administered for at least 48 weeks so that patients receive at least eight doses of AGS-003. Dosing will cease prior to 48 weeks if two events of disease progression or unacceptable toxicity occur or upon the joint decision of the patient and the investigator. If after 48 weeks of dosing of AGS-003, a patient has stable disease or is responding to treatment, dosing will continue once every three months until disease progression. If an investigator determines to discontinue sunitinib, either due to disease progression or toxicity, the investigator can, at any time during the trial after the first six week cycle of sunitinib, initiate second-line therapy with one of the other approved targeted therapies, including pazopanib, axitinib, everolimus or temsirolimus. In the event of discontinuation of sunitinib for patients in the combination therapy arm, such patients would continue with AGS-003 dosing in combination with the second-line therapy. In our Phase 2 combination therapy clinical trial, dosing ceased upon the first event of disease progression and second-line therapy was not permitted.
A graphic of the trial design is shown below.
Other Development Activities. We believe that AGS-003 may be capable of treating a wide range of cancers and are planning to evaluate AGS-003 in clinical trials in additional cancer indications.
| • | We plan to support an investigator-initiated Phase 2 clinical trial of AGS-003 in combination with a PD-1 inhibitor in mRCC that is expected to begin in the second half of 2016. |
| • | We are supporting an investigator-initiated Phase 2 clinical trial designed to evaluate treatment with AGS-003 in patients with early stage RCC prior to nephrectomy. This trial was opened for enrollment of patients in late 2014. Four patients have been enrolled in the trial as of March 15, 2016. We expect that a total of 10 patients will be enrolled in this trial and that initial data will be available in the fourth quarter of 2016. |
| • | We are supporting an investigator-initiated Phase 2 clinical trial of AGS-003 in NSCLC that was initiated in the first quarter of 2016. In the trial, the safety, efficacy and immunologic effects of AGS-003 when combined with platinum-based chemotherapy and radiation will be evaluated in approximately 20 stage 3 NSCLC patients. |
| • | We also plan to support an additional investigator-initiated Phase 2 clinical trial of AGS-003 in muscle invasive bladder cancer, which we expect may begin in mid-2016. |
Phase 2 Combination Therapy Clinical Trial. From July 2008 to October 2009, we enrolled 21 newly diagnosed mRCC patients in a single arm, multicenter, open label Phase 2 clinical trial of AGS-003 in combination with sunitinib. We conducted this clinical trial at nine clinical sites in the United States and Canada. Our design for the trial required adult patients with previously untreated mRCC, no prior nephrectomy or at least one accessible lesion for biopsy, a histologically confirmed predominantly clear cell tumor, and suitability for sunitinib therapy. The primary endpoint of the trial was complete response rate. Secondary endpoints included progression free survival, overall survival, safety, clinical benefit rate and immune response.
Patients in the trial generally received one initial six-week cycle of sunitinib, consisting of four weeks on drug and two weeks on drug holiday, prior to initiating the combined treatment with AGS-003. Patients then received a dose of AGS-003 every three weeks for a total of five doses, while also continuing three additional six-week cycles of sunitinib. This 24-week induction phase was followed by a booster phase during which patients received a dose of AGS-003 once every three months and continued to receive sunitinib in six-week cycles until disease progression.
| 12 |
The following table summarizes certain key data from the 11 intermediate risk and 10 poor risk patients enrolled in the Phase 2 combination therapy clinical trial.
| Outcome | (N=21) | |||
| Median OS (1) | 30.2 months | |||
| Median PFS (2) | 11.2 months | |||
| Complete response (3) | 0 patients | |||
| Partial response (4) | 9 patients | |||
| Stable disease (5) | 4 patients | |||
| Immune response | CD8+ CD28+ memory T-cells correlated with OS, PFS and reduced metastatic tumor burden; IL-2 and interferon- g (IFN- g ) recovery | |||
_________________
| (1) | Overall survival, or OS, is the length of time from the initiation of treatment to the patient’s death. |
| (2) | Progression free survival, or PFS, is the length of time from treatment initiation to the worsening of the patient’s disease or the patient’s death. |
| (3) | Complete response is the disappearance of all measurable target lesions and non-target lesions. |
| (4) | Partial response is the overall tumor regression based on a decrease of at least 30% in the overall amount of measurable tumor mass in the body and improvement or no change in non-target lesions. |
| (5) | Stable disease is neither sufficient decrease in tumor size to qualify as a partial response nor sufficient increase in tumor size to qualify as disease progression. |
| 13 |
Particular observations from these data and the trial, which have informed our further clinical development of AGS-003, include:
Efficacy Analysis
| • |
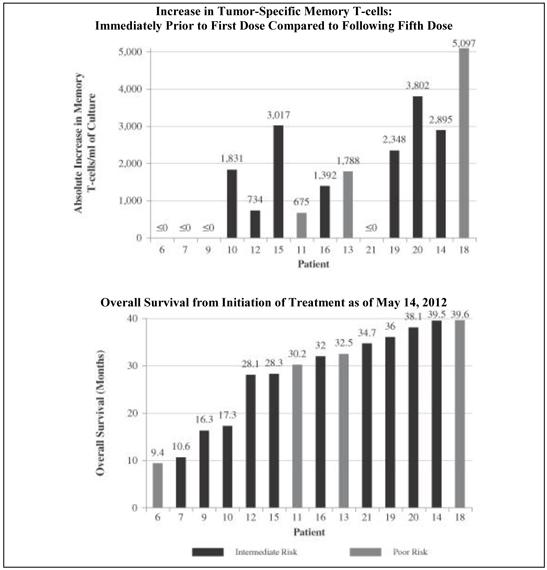
Seven patients survived for more than 4.5 years following enrollment in this trial. Two of these patients remained alive as of December 31, 2015 and both have had a sustained clinical response spanning nearly seven years and remain on AGS-003 in combination with continued targeted therapy. |
| • | Five poor risk patients did not receive five doses of AGS-003 due to early disease progression. Median overall survival in the 16 patients who received at least five doses of AGS-003 was 36.0 months. |
| • | Median overall survival in the 11 intermediate risk patients was 61.9 months. Median overall survival in the 10 poor risk patients was 9.1 months. |
| • | The following graphic shows data and follow-up as of December 31, 2015, the number of months that each patient in the Phase 2 clinical trial survived from the time of enrollment in the trial. |

| • | Of the nine patients who exhibited a partial response, four patients exhibited partial responses during the 24-week induction phase, including two patients who exhibited partial responses prior to initiation of treatment with AGS-003. The other three patients exhibited partial responses after prolonged dosing with AGS-003 during the booster phase. We do not believe that these late occurring partial responses have been observed in clinical trials of sunitinib alone. As a result, we believe that these late responses may relate to the immunologic effects of prolonged AGS-003 dosing and AGS-003’s effect on CD8+ CD28+ memory T-cells. |
| 14 |
| • | We observed a statistically significant correlation between increased progression free survival and prolonged survival (p<0.001). Statistical significance is determined by methods that establish the p-value of the results. Typically, results are considered statistically significant if they have a p-value of 0.05 or less, meaning that there is less than a one-in-20 likelihood that the observed results occurred by chance. |
Immune Response Analysis
| • | In the 14 patients in the trial who received at least five doses of AGS-003 and could be evaluated for memory T-cell response, we observed a statistically significant correlation between the increase in the number of CD8+ CD28+ memory T-cells over the initial five doses of AGS-003 and survival (p<0.002), progression free survival (p<0.031) and reduced metastatic tumor burden (p<0.045). We presented at the 2013 Annual Meeting of ASCO, Genitourinary Cancers Symposium results, as of May 14, 2012, showing the correlation with survival. The following graphics show, for each of these 14 patients, the increase in their tumor-specific memory T-cells that they exhibited as measured immediately prior to their first dose of AGS-003 and immediately following the patient’s fifth dose of AGS-003, or the absence of such increase, as compared to such patient’s survival. |
Phase 2 Combination Therapy Clinical Trial of AGS-003:
Correlation of Immune Response and Overall Survival
| • | AGS-003 was found to have positive impact on immune cell function and restoration of cellular immunity in a majority of patients, including an increase in levels of IL-2 and IFN- g . |
| 15 |
Safety
| • | The adverse events in this trial associated with AGS-003 were generally only mild injection site reactions, while the toxicities associated with sunitinib were consistent with those expected from treatment with sunitinib alone. |
The original design for the Phase 2 clinical trial called for the recruitment of 50 patients to generate 38 fully evaluable patients. However, in October 2009, we terminated enrollment in this trial early due to a lack of funding. As a result, only 21 patients were enrolled and received at least one dose of AGS-003. In addition, the trial was originally designed to enroll patients with favorable and intermediate risk disease profiles. Instead, the actual population enrolled consisted entirely of patients with intermediate or poor risk disease profiles who had the less than one year to treatment risk factor. Because the patient population had poorer prognoses when they entered the trial than we expected and we did not have a sufficient number of evaluable patients, we did not perform the statistical analysis to determine whether the primary endpoint of complete response rate was achieved. As a result, we expect the data from this trial to be considered by the FDA for the purpose of evaluating the safety and feasibility of AGS-003, but that it will only have a limited impact on the FDA’s ultimate assessment of the efficacy of AGS-003.
Based on our experience with the Phase 2 clinical trial, we concluded that the secondary endpoints in the trial, progression free survival and overall survival, along with immune response, were the appropriate endpoints to consider for measuring the efficacy of AGS-003 in combination with sunitinib in patients with mRCC in our pivotal Phase 3 clinical trial.
AGS-003 Phase 2 Combination Therapy Clinical Trial, as Compared to Independent Third Party mRCC Data. At ASCO in June 2013, Dr. Heng presented data from the Consortium database regarding overall survival and progression free survival for intermediate and poor risk patients treated with sunitinib and other targeted therapies, including data with respect to 1,189 intermediate and poor risk patients with the less than one year to treatment risk factor.
Using the overall survival data from the Consortium database presented in June 2013 and published in April 2014, a summary comparison of this data with our Phase 2 clinical trial of AGS-003 in combination with sunitinib is set forth in the graphic below. This graphic compares the median overall survival data from the Consortium intermediate and poor risk patients with the less than one year to treatment risk factor with the median overall survival data from the 21 patients in our Phase 2 clinical trial of AGS-003 in combination with sunitinib, all of whom had the less than one year to treatment risk factor. A majority of the Consortium patients and the patients in our Phase 2 clinical trial had one or more additional risk factors.
| 16 |

Progression free survival for intermediate and poor risk patients in the Consortium database with the less than one year to treatment risk factor was 5.6 months, as compared to the 11.2 months of median progression free survival that we observed in the 21 patients in our Phase 2 clinical trial of AGS-003 in combination with sunitinib.
Although we believe comparisons between our data and these collections of data are useful in evaluating the overall results of our Phase 2 clinical trial, the treatment of the Consortium patients was conducted at different sites, at different times and in different patient populations than the treatment in our Phase 2 combination therapy trial. The treatment also differed because certain of the Consortium patients received therapies other than sunitinib as first-line treatment. All of the patients in our Phase 2 clinical trial received sunitinib as first-line treatment. Our ongoing pivotal Phase 3 combination therapy clinical trial of AGS-003 is the first trial that we have conducted that directly compares AGS-003 and sunitinib or other targeted therapies as a combination therapy against sunitinib as monotherapy. Results of this head-to-head comparison in our phase 3 ADAPT trial may differ significantly from the comparisons presented above and elsewhere in this Annual Report on Form 10-K.
AGS-004 for the Treatment of Human Immunodeficiency Virus
We are developing AGS-004, our second Arcelis-based product candidate, for the treatment of HIV. We have completed three clinical trials of AGS-004. These include Phase 1 and Phase 2 clinical trials that were funded by government grants and a Phase 2b trial that was funded in full by the NIH.
Based on the clinical data that we have generated to date, we have determined to focus our development program on the use of AGS-004 in combination with other therapies to achieve complete virus eradication and the use of AGS-004 monotherapy to provide long-term control of HIV viral load in immunologically healthy patients and eliminate their need for ART.
| 17 |
Human Immunodeficiency Virus
HIV is characterized by a chronic viral infection and an associated deterioration of immune function. Specifically, the virus disables and kills crucial human immune cells called CD4+ T-cells. CD4+ T-cells are necessary to generate and maintain antiviral T-cells, including the CD8+CD28+ memory T cells that kill virus-infected cells. Over time, this viral impact on an infected person’s immune system outpaces the body’s natural ability to replace CD4+ T-cells and immunodeficiency results. As a result, the longer a person has been infected with the virus, the more functionally impaired these cells become.
At the same time, HIV infection causes the immune cells in HIV patients, including CD4+ T-cells and CD8+ T-cells that are not killed by the virus to be in a chronic state of activation. The persistent state of immune activation in HIV patients results in chronic inflammation. We believe that this inflammation plays a role in the elevated rates of age-related comorbidities, including malignancies and cardiovascular disease observed in HIV patients. In addition, the activation of the CD4+ T-cells supports virus replication which leads to the production of new virus and increased viral load.
HIV is a persistent virus that can rapidly adapt to its environment by mutating and creating HIV variants that are drug resistant and can evade immune attack. As a result, there are a large number of mutated variants of HIV existing in any one infected individual and no two individuals have identical viral sequences.
According to the World Health Organization, the number of people living with HIV in the world was approximately 35 million in 2013. The Centers for Disease Control and Prevention estimates that more than 1.2 million people are currently living with HIV in the United States and the number of new cases of HIV infection in the United States is expected to remain constant at approximately 50,000 cases per year.
Current treatments for HIV. In 1996, a combination of oral medications known as ART was introduced to treat patients with HIV. Since then, the introduction of new drug classes of ART and combination drug treatment strategies has enhanced treatment for HIV.
ART in HIV-infected patients can decrease levels of HIV in the blood to below the limits of detection, increase life expectancy and improve quality of life. However, there continues to be an unmet need for HIV therapies for the following reasons:
| • | ART can have significant side effects. The most recent U.S. guidelines on ART treatment contain a number of tables of adverse effects of combination regimens and how to manage them. Some combinations present potentially life-threatening complications and other complications that are chronic, cumulative and overlapping, and sometimes irreversible. |
| • | ART requires life-long daily treatment. The risks of long-term daily administration of ART remain unknown but are potentially significant. In addition, the requirement for life-long daily treatment has made strict adherence to the treatment regime difficult. Poor compliance has led to the development of drug resistant HIV variants that are ineffectively controlled by the available armamentarium of ART. |
| • | ART cannot eradicate the virus and, therefore, does not cure HIV-infected patients. For example, up to 20% of patients receiving ART fail to achieve normal CD4+ T-cell counts, resulting in a continued weakened immune system. In addition, certain patients are not able to achieve effective control of the virus using current treatment regimens. ART cannot eradicate the virus because the virus persists in latently infected cells. These cells, which constitute the HIV latent reservoir, do not express HIV antigens and are therefore invisible to the immune system. Instead, these cells serve as the source for virus replication and viral rebound in the absence of ART. Following discontinuation of treatment with ART, HIV viral levels return to levels observed prior to treatment with ART within 12 weeks of treatment interruption. |
AGS-004 Opportunity
We believe, based on the mechanism of action of AGS-004 and the clinical data that we have generated, that AGS-004 has the potential to address this unmet need for the following reasons:
| • | Potential to Eradicate HIV in Combination with Latency Reversing Drugs. A number of companies and academic groups are evaluating drugs that can potentially activate the latently infected cells to increase viral antigen expression and make the cells vulnerable to elimination by the immune system. We believe that treating HIV-infected patients, who are being successfully treated with ART, with a combination of AGS-004 and one of these latency reversing drugs could lead to activation of antigen expression from the latently infected cells along with a potent memory T-cell response that is specific to the patient’s own unique viral antigens. We believe that this approach could potentially result in complete eradication of the patient’s virus. |
| 18 |
| • | Long-Term Viral Load Control in Immunologically Healthy Patients. We believe that AGS-004 may allow for long-term virus control and eliminate the need for life-long treatment with ART in infected patients who have minimal immune suppression but no T-cell response against their virus. We have designed AGS-004 to induce CD8+ CD28+ memory T-cells that are specific to the patient’s own unique viral antigens, do not require CD4+ T-cell help to kill viral cells and do not result in CD4+ T-cell activation which typically increases viral replication and viral load. As reported in Clinical & Experimental Immunology, researchers have demonstrated that elevated levels of CD8+CD28+ memory T-cells in the blood are a statistically significant predictor of long-term non-progression in HIV-infected patients not treated with ART drugs. As a result, we believe that inducing these memory T-cells may lead to viral control. Patients with minimal immune suppression and no T-cell response include pediatric patients who have been successfully treated with ART drugs since birth or shortly thereafter and have generally healthy immune systems. |
| • | Minimal Toxicity. AGS-004 has been well tolerated in clinical trials with no serious adverse events being attributed to it. As a result, we believe we can combine AGS-004 with other HIV therapies without additional toxicities. |
| • | Lack of Chronic Inflammation. We have designed AGS-004 to elicit a patient-specific and disease-specific immune response that does not cause any additional inflammation. In our clinical trials of AGS-004, AGS-004 has not induced changes in markers that are associated with chronic inflammation in HIV patients. |
Description and Development Status
AGS-004 is a truly individualized immunotherapy based on our Arcelis platform. It is produced by electroporating dendritic cells with mRNA encoding for patient-specific HIV antigens that have been derived from a patient’s virus-infected blood and with RNA that encodes the CD40L protein. The process for producing AGS-004 is the same process as is used to produce AGS-003, with the one key difference being that AGS-003 contains all of the antigens from a patient’s tumor cells while AGS-004 contains potentially all variants unique to each individual patient of four selected HIV antigens (Gag, Nef, Vpr and Rev). We designed AGS-004 to include these antigens because immunity to them has been observed in long-term non-progressors and elite controllers, two groups of rare patients able to control virus replication without ART. Because no two patients share identical HIV antigen sequences and there are a large number of mutated variants of HIV existing in each infected patient, by using mRNA that is specific to the patient’s virus and that captures potentially all of the unique patient-specific variants of each antigen, we believe our immunotherapy maximizes the relevance of the immune responses induced in each patient.
We have conducted three clinical trials of AGS-004, which include:
| · | a phase 2b clinical trial of AGS-004; |
| · | a phase 2a clinical trial of AGS-004; and |
| · | a phase 1 clinical trial of AGS-004. |
We submitted to the FDA an IND for AGS-004 in August 2008.
We are focusing our development program for AGS-004 on the use of AGS-004 in combination with latency reversing therapies to achieve complete virus eradication. Latently infected cells differ from other infected cells in that the HIV genome is permanently integrated into the chromosomal DNA of the latently infected cells. These latently infected cells persist long-term and constitute the HIV latent reservoir, which serves as a source for low level virus replication and viral rebound in the absence of antiretroviral therapy. As a result, demonstration that latently infected cells can be targeted by immune responses induced by AGS-004 is essential to our development strategy pertaining to virus eradication.
Adult Eradication Trial. We are supporting an investigator-initiated Phase 2 clinical trial of AGS-004 in adult HIV patients who are being treated with ART to evaluate the use of AGS-004 to eradicate the virus. The trial is being conducted by co-investigator Dr. David Margolis, Professor of Medicine at the University of North Carolina. Dr. Margolis is the leader of the Collaboratory of AIDS Researchers for Eradication, or CARE, and has been a pioneer in the research of HIV latent reservoir reversing treatments. The trial is being conducted in two stages. In the first stage, HIV-infected patients received AGS-004 while remaining on ART and the kinetics of the anti-HIV CD8+ CD28+ memory T-cell responses were evaluated. In the second stage, which Dr. Margolis initiated in the first quarter of 2016, the use of AGS-004 in combination with the latency reversing drug Vorinostat will be evaluated. CARE funded all patient clinical costs for the first stage of this Phase 2 clinical trial, except for the associated manufacturing costs for which we were responsible. The NIH Division of AIDS has approved $6.6 million in funding for the second stage of this trial.
Planned Pediatric Functional Cure Trial. We believe that a patient population that could benefit from AGS-004 monotherapy consists of 14+ year old, HIV-infected individuals who have been treated with ART since birth or shortly thereafter. These individuals are characterized by having very small HIV latent reservoirs and otherwise healthy immune systems, while lacking antiviral CD8+ CD28+ memory T-cell responses. We believe that successfully inducing antiviral CD8+ CD28+ memory T-cell responses in these patients could allow for long-term viral load control and eliminate the need for life-long antiretroviral therapy. We plan to support an investigator-initiated Phase 2 clinical trial of AGS-004 in pediatric HIV patients to evaluate the use of AGS-004 monotherapy to allow for long-term control of viral load and eliminate the need for ART. We are currently developing the clinical protocol for this trial to immunize pediatric HIV patients who were infected at birth and treated with antiretroviral therapy at or near birth. We are developing this clinical protocol in collaboration with Drs. Katherine Luzuriaga, University of Massachusetts, and Deborah Persaud, John Hopkins Medical Center, both specializing in pediatric virology. The commencement of this trial is subject to supportive data obtained from the adult eradication trial and approval of the protocol by the principal investigator(s), institutional review boards, the IMPAACT Network leadership and the FDA and to the agreement by the NIH to fund the trial costs not related to AGS-004 manufacturing. Assuming the supportive data and the necessary approvals are obtained, we expect this trial to initiate in 2017.
| 19 |
Phase 2b Clinical Trial. In January 2015, we completed a randomized, placebo controlled, double blind Phase 2b clinical trial of AGS-004 in chronically infected patients on ART that we initiated in July 2010. We designed this trial to confirm the data obtained in an earlier Phase 2a clinical trial in which AGS-004 led to a reduction in virus replication. We initially planned to enroll 42 chronically infected patients in the Phase 2b trial at nine clinical sites in the United States and Canada with the intent to generate 36 events for the primary endpoint analysis. However, due to a higher than anticipated dropout rate by patients who were unable to complete the full 12 week treatment interruption period provided for by the trial, we needed to enroll 53 patients in the trial to generate 36 events for the primary endpoint analysis. These patients were randomized between AGS-004 treatment and a placebo control on a two-to-one basis.
HIV infection is classified as “chronic” or “acute” based on how long the patient has been infected prior to starting ART. Patients with chronic HIV infection are patients who have initiated ART after at least six months from the time of initial infection. Patients with acute HIV infection are patients who have initiated ART less than 45 days after initial infection. This trial enrolled adult patients with chronic HIV-1 infection and undetectable viral loads as a result of treatment with ART. Patients also had to have adequate CD4+ T-cell counts and a pre-ART plasma viral sample to be used to manufacture AGS-004.
In this trial, patients first received intradermal doses of AGS-004 or placebo every four weeks for a total of four doses, together with their ART. Following the fourth dose of AGS-004 or placebo, patients discontinued their ART but continued to receive AGS-004 or placebo every four weeks for 12 weeks. We refer to this period as the treatment interruption period. Patients who demonstrated control of viral replication under 10,000 copies/ml and maintained CD4+ T-cell counts above 350 cells/mm3 could remain off ART and continue their treatment interruption past 12 weeks. Following the end of treatment interruption, all patients were eligible for continued treatment with the combination of AGS-004 and ART. A schematic of the trial design is shown below.
Phase 2b Study Design for the Chronically Infected Cohort

The primary endpoint of the trial was a comparison of the median viral load in the AGS-004-treated patients with the median viral load in patients receiving placebo after 12 weeks of ART treatment interruption. Under this protocol, the primary endpoint required that there was a ³1.1 log 10 difference in median viral load between the AGS-004-treated cohort compared to the placebo-treated cohort. A 1.1 log 10 reduction means a 92% lower virus concentration in the AGS-004-treated cohort compared to the placebo-treated cohort. Secondary endpoints included comparisons between AGS-004-treated patients and the patients receiving placebo with respect to change in viral load from pre-ART to the end of 12 weeks of treatment interruption, duration of treatment interruption, changes in CD4+ T-cell counts and safety.
In September 2011, we added to the trial a single arm, open-label, unblinded cohort of up to 12 patients with acute HIV-1 infection and undetectable viral loads as a result of treatment with ART. We evaluated AGS-004 in this patient population to assess AGS-004 in patients who initiated ART during the acute phase of infection and as a result may have sustained less immune damage. Patients in this cohort were dosed in the same manner as patients in the chronically infected arm of the clinical trial. However, in this cohort, patients had to demonstrate a positive CD8+ CD28+ anti-HIV memory T-cell response in order to become eligible to enter the 12 week treatment interruption period. The primary endpoints for this cohort included the time to detectable viral load during the ART interruption period and comparison of changes in CD4+ T-cell counts during the ART interruption period between the acute cohort and the chronic cohort. Six patients were enrolled in this cohort. All six patients demonstrated a positive CD8+ CD28+ memory T-cell response and initiated treatment interruption. For the five of six patients that re-initiated ART after treatment interruption, there were no significant declines in CD4+ T cells between the interruption date and the re-initiation date. All six patients experienced viral rebound during treatment interruption with the times to detectable viral load ranging from two to eight weeks and the duration of treatment interruption for those patients who reinitiated ART ranged from approximately one month to approximately nine months. In addition, three of six patients had a decrease in circulating CD4+ T cells containing HIV DNA of 25%, 47% and 63%, respectively, when measured after three doses of AGS-004 while on ART.
| 20 |
In the Phase 2b trial, 54 patients received the full four doses of AGS-004 or placebo during the first four weeks together with their ART. Of these patients, 36 patients continued on AGS-004 or placebo for the full 12-week treatment interruption period, 23 of whom received AGS-004.
In January 2015, we announced top-line results from the trial. The primary endpoint of the trial was not achieved.
However, we believe that data from the trial provided evidence of the ability of AGS-004 to induce memory T-cell responses which may have directly impacted the latent viral reservoir. Of the evaluated 22 patients who received AGS-004 and completed the 12-week treatment interruption period, 15 patients or approximately 70 percent, had positive antiviral memory T-cell responses prior to beginning the treatment interruption versus zero percent of placebo patients. Within the AGS-004 treatment group, those patients that had antiviral memory T-cell responses had significantly fewer CD4+ T-cells with integrated HIV DNA when compared to non-responders. These findings relate directly to the utilization of AGS-004 in our ongoing adult eradication study and our planned pediatric study, where one of the key objectives is to decrease the latent HIV reservoir.
Safety analysis
In this trial, AGS-004 was safe and well tolerated. No AGS-004-related serious adverse events were reported. The most common adverse event was mild injection site reactions. During the antiretroviral treatment interruption, no notable differences in incidence of adverse events occurred compared to when patients were receiving AGS-004 in combination with antiretroviral drug therapy.
NIH and NIAID Contract. Our development of AGS-004 has received significant funding from the U.S. federal government. In September 2006, we entered into a multi-year research contract with the NIH and the National Institute of Allergy and Infectious Diseases, or NIAID, to design, develop and clinically test an autologous HIV immunotherapy capable of eliciting therapeutic immune responses. We are using funds from this contract to develop AGS-004. Under this contract, as it has been amended, the NIH and the NIAID have committed to fund up to $39.8 million, including reimbursement of our direct expenses and allocated overhead and general and administrative expenses of up to $38.4 million and payment of specified amounts totaling up to $1.4 million upon our achievement of specified development milestones. We have recorded total revenue of $37.1 million through December 31, 2015 under the NIH agreement. As of December 31, 2015, there was up to $2.7 million of potential revenue remaining to be earned under the agreement. This commitment extends until July 2016.
We have agreed to a statement of work under the contract, and are obligated to furnish all the services, qualified personnel, material, equipment, and facilities, not otherwise provided by the U.S. government, needed to perform the statement of work. In accordance with the laws applicable to government intellectual property rights under federal contracts, we have a right under our contract with the NIH to elect to retain title to inventions conceived or first reduced to practice under the NIH and NIAID contract, subject to the right of the U.S. government to a royalty-free license to practice or have practiced for or on behalf of the United States the subject invention throughout the world. The government also has special statutory “march-in” rights to license or to require us to license such inventions to third parties under limited circumstances. In addition, we may not grant to any person the exclusive right to use or sell any such inventions in the United States unless such person agrees that any products embodying the subject invention or produced through the use of the subject invention will be manufactured substantially in the United States.
Manufacturing
We currently manufacture our Arcelis-based products, AGS-003 and AGS-004, for use in our clinical trials of those product candidates at our facility in Durham, North Carolina. Our facility includes manufacturing suites for the production of products using our Arcelis technology platform. We have designed these suites to comply with the FDA’s current good manufacturing practice, or cGMP, requirements. We have manufactured the product for our development and clinical trial activities associated with AGS-003 and AGS-004 to date, and are manufacturing the product for the ADAPT trial using our current processes at our current facility.
| 21 |
In August 2014, in connection with our plan to build out and equip a commercial manufacturing facility, we entered into a lease agreement with the developer, TKC LXXII, LLC, or TKC. Under the lease agreement, we agreed to lease certain land and an approximately 124,000 square-foot building to be constructed in Durham County, North Carolina. This facility would house our corporate headquarters and commercial manufacturing. The shell of the new facility was constructed on a build-to-suit basis in accordance with agreed upon specifications and plans and was completed in June 2015. A portion of the build-out and equipping of the interior of the facility began in the second half of 2015.
Under the lease agreement, we had an option to purchase the property. In February 2015, we exercised this purchase option and entered into a Purchase and Sale Agreement with TKC. The purchase price to be paid by us is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by us, for which we have paid $1.7 million as of December 31, 2015, and the amount of any improvement allowances advanced to us by TKC prior to the closing. Under the terms of the Purchase and Sale Agreement, we had until February 16, 2016 to consummate the purchase of the property. We are currently in discussions with TKC to extend the period for purchasing the property as we will need to obtain additional funding beyond the $60 million that we may receive under the March 2016 securities purchase agreement to consummate the purchase of the property. If we purchase the property, upon the closing, the lease agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to us.
Due to our limited resources and the costs and challenges of building and equipping a new facility, we are exploring alternative plans for the commercial supply of our products, including building out and equipping our current facility for commercial manufacture, leasing and equipping an existing manufacturing space that would be built out for commercial manufacture, and engaging a contract manufacturer to supply AGS-003 on a commercial scale on our behalf. We believe that building out and equipping our current facility would only be a temporary solution from a capacity standpoint and would only cover commercial manufacturing for a limited number of years.
We plan to initiate commercial manufacturing using our current manual manufacturing process and have decided to complete the development and implementation of our automated manufacturing process after commercial launch. Prior to implementing commercial manufacturing of AGS-003, we would be required to demonstrate that the commercial manufacturing facility is constructed and operated in accordance with current good manufacturing practice. We would also be required to show the comparability between AGS-003 that we produce using the manual processes in our current facility and AGS-003 produced using the manual process in the commercial manufacturing facility.
If we transition to automated manufacturing processes, we expect our automated manufacturing processes will be based on existing functioning prototypes of automated devices for the production of commercial quantities of our Arcelis-based product candidates. These devices can be used to perform substantially all steps required for the manufacture of our Arcelis-based product candidates.
We plan to use the automated equipment to perform Arcelis product processing in closed, single-use disposable containers. A patient’s disease sample would only be in contact with these single use disposable containers and not with any component of our manufacturing equipment. Because our equipment would never be in direct contact with patient material, we believe that the time required to prepare the equipment between batches would be minimal. We also believe that automated processing of material in disposable containers would reduce the complexity and size of the facility by reducing the amount of required labor.
We have granted exclusive manufacturing rights for AGS-003 to Pharmstandard in Russia and the other states comprising the Commonwealth of Independent States, to Green Cross in South Korea, to Medinet in Japan and to Lummy HK in China, Hong Kong, Taiwan and Macau. We have also agreed to enter into an agreement with Pharmstandard for the manufacture of AGS-003 in the European market.
Sales and Marketing
We hold exclusive commercial rights to all of our product candidates in all geographies other than rights to AGS-003 in Russia and the other states comprising the Commonwealth of Independent States, which are held by Pharmstandard, rights to AGS-003 for the treatment of mRCC in South Korea, which are held by Green Cross and rights to AGS-003 in China, Hong Kong, Taiwan and Macau, which are held by Lummy HK.. We have granted to Medinet an exclusive license to manufacture in Japan AGS-003 for the treatment of mRCC and an option to acquire a non-exclusive license to sell in Japan AGS-003 for the treatment of mRCC. We do not expect Medinet to exercise this option before its expiration on March 31, 2016.
| 22 |
We currently intend to retain North American marketing rights for AGS-003 and any future oncology products that we may develop. To maximize the value of these rights, we would expect to build a commercial infrastructure for such products comprised of medical, marketing and sales teams, a customer service function to manage patient access and logistics partners associated with AGS-003 production and distribution. Our commercial infrastructure would also include personnel who manage reimbursement activities with third party payors, such as managed care organizations, group purchasing organizations, oncology group networks and government accounts. We plan to hire additional personnel to fill key positions in advance of the approval of AGS-003. We currently have limited commercial capabilities and few in-house personnel specializing in these functions. Outside North America, we plan to seek to enter into collaboration agreements with other pharmaceutical or biotechnology firms to commercialize AGS-003.
For AGS-004, we plan to seek to enter into collaboration agreements with other pharmaceutical or biotechnology firms to commercialize this product candidate on a worldwide basis.
Competition
The biotechnology and pharmaceutical industries are highly competitive. There are many pharmaceutical companies, biotechnology companies, public and private universities and research organizations actively engaged in the research and development of products that may be similar to or competitive with our products. There are a number of multinational pharmaceutical companies and large biotechnology companies currently marketing or pursuing the development of products or product candidates targeting the same indications as our product candidates. It is probable that the number of companies seeking to develop products and therapies for the treatment of unmet needs in these indications will increase. Some of these competitive products and therapies are based on scientific approaches that are the same as or similar to our approaches, and others are based on entirely different approaches.
Many of our competitors, either alone or with their strategic partners, have substantially greater financial, technical and human resources than we do and significantly greater experience in the discovery and development of product candidates, obtaining FDA and other regulatory approvals of products and the commercialization of those products. Our competitors’ drugs may be more effective, or more effectively marketed and sold, than any drug we may commercialize and may render our product candidates obsolete or non-competitive. We anticipate that we will face intense and increasing competition as new drugs enter the market and advanced technologies become available. We expect any products that we develop and commercialize to compete on the basis of, among other things, efficacy, safety, convenience of administration and delivery, price, the level of generic competition and the availability of reimbursement from government and other third party payors.
mRCC
Historically, mRCC was treated with chemotherapy, radiation and hormonal therapies, as well as cytokine-based therapies such as interferon-µ and IL-2. More recently, the FDA has approved several targeted therapies as monotherapies for mRCC, including Nexavar (sorafenib), marketed by Bayer Healthcare Pharmaceuticals, Inc. and Onyx Pharmaceuticals, Inc.; Sutent (sunitinib) and Inlyta (axitinib), marketed by Pfizer, Inc.; Avastin (bevacizumab), marketed by Genentech, Inc., a member of the Roche Group; Votrient (pazopanib) and Afinitor (everolimus), marketed by Novartis Pharmaceuticals Corporation; Torisel (temsirolimus), marketed by Pfizer and most recently, Opdivo (nivolumab) for second-line RCC, marketed by Bristol-Myers Squibb. In addition, we estimate that there are numerous therapies for mRCC in clinical development by many public and private biotechnology and pharmaceutical companies targeting numerous different cancer types and stages. A number of these are in late stage development. Opdivo (nivolumab) plus Yervoy (ipilimumab) in combination for first-line mRCC is being developed by Bristol-Myers Squibb and is currently being compared in a Phase 3 trial to sunitinib. Cometriq (cabozantinib) is being developed by Exelixis has recently reported positive results from its Phase 3 trial and is expected to receive FDA approval for second-line mRCC in 2016. In addition, if a standalone therapy for mRCC were developed that demonstrated improved efficacy over currently marketed first-line therapies with a favorable safety profile and without the need for combination therapy, such a therapy might pose a significant competitive threat to AGS-003.
We estimate that there are numerous other cancer immunotherapy products in clinical development by many public and private biotechnology and pharmaceutical companies targeting numerous different cancer types. A number of these product candidates are in late-stage clinical development or have recently been approved in different cancer types including two recently approved checkpoint inhibitor-based immunotherapies, Nivolumab which is marketed by Bristol-Myers Squibb and Pembrolizumab which is marketed by Merck.
HIV
There are numerous FDA-approved treatments for HIV, primarily antiretroviral therapies, marketed by large pharmaceutical companies. In addition, generic competition has recently developed as patent exclusivity periods for older drugs have expired, with more than 15 generic bioequivalents currently on the market. The presence of these generic drugs is resulting in price pressure in the HIV therapeutics market. Currently, there are no approved therapies for the eradication of HIV. We expect that major pharmaceutical companies that currently market antiretroviral therapy products or other companies that are developing HIV product candidates may seek to develop products for the eradication of HIV.
| 23 |
Intellectual Property
Our success depends in part on our ability to obtain and maintain proprietary protection for our products and product candidates, technology and know-how, to operate without infringing the proprietary rights of others and to prevent others from infringing our proprietary rights. We are seeking a range of patent and other protections for our product candidates and platform technology. We also rely on trade secrets, know-how, continuing technological innovation and in-licensing opportunities to develop and maintain our proprietary position.
Patents
We own or exclusively license 16 U.S. patents and seven U.S. patent applications, as well as approximately 60 foreign counterparts, covering our Arcelis technology platform and Arcelis-based product candidates.
We use our Arcelis technology platform to generate truly individualized mRNA-loaded dendritic cell immunotherapies. As described above, the process of obtaining a disease sample and dendritic cells from a patient, using those materials to manufacture a truly individualized drug product and shipping the drug product to the clinical site for use in the treatment of the patient involves many important steps. These steps include:
| • | amplifying mRNA from a disease sample obtained from the patient; |
| • | differentiating dendritic cell precursors (monocytes) isolated from the patient into immature dendritic cells; |
| • | maturing the immature dendritic cells in culture and loading the mature dendritic cells with the amplified mRNA and CD40L protein; and |
| • | formulating the matured, loaded dendritic cells in the patient’s plasma with cryoprotectants to protect the cells in the resulting drug product when the drug product is frozen and thawed. |
We have sought to protect these steps or the equipment related to carrying out one or more of these steps through patents or trade secrets. We have also sought to protect the resultant drug product through patents.
These patents and patent applications are directed to one or more aspects of our Arcelis technology platform or Arcelis-based products. Specifically, these patents and patent applications are collectively directed to:
| • | Arcelis-based compositions of matter and products; |
| • | methods of manufacturing Arcelis-based products; |
| • | methods of using Arcelis-based products for treatment of tumors; |
| • | compositions that we use in the manufacture of Arcelis-based AGS-004 products; and |
| • | equipment that we intend to use for assisting the automated manufacture of Arcelis-based products. |
We believe that all of the above aspects of our Arcelis technology platform are required to successfully and efficiently produce our Arcelis-based product candidates and are covered by a combination of our patents, patent applications, trade secrets and know-how. The U.S. patents expire between 2016 and 2029, and the U.S. patent applications, if issued, would expire between 2025 and 2029, the counterpart patents in Europe and Japan expire between 2017 and 2027, and the counterpart patent applications in Europe and Japan, if issued, would expire between 2025 and 2027. Included in these patents and patent applications are:
| • | six U.S. patents that are collectively directed toward the composition of matter of Arcelis-based products (dendritic cells loaded with RNA from tumors or pathogens), methods of manufacture of these products and methods of using these products to treat tumors. Five of these U.S. patents encompass the AGS-003 composition of matter. Three of the six U.S. patents encompass the AGS-004 composition of matter. The U.S. patents expire in 2016. Two corresponding patents in Europe and two corresponding patents in Japan, expire in 2017. |
| • | four U.S. patents, one U.S. patent application and corresponding patent application in Europe and patent in Japan collectively directed towards an automated apparatus for the manipulation of nucleic acids in a closed container, components thereof and related methods of use. The U.S. and Japanese patents expire in 2027, and the patent applications in the United States and Europe, if issued, would expire in 2027. |
| 24 |
| • | one U.S. patent and corresponding European and Japanese patents collectively directed towards cryoconserved dendritic cells and related methods of manufacture. The U.S., European and Japanese patents expire in 2021. |
| • | two U.S. patents and two U.S. patent applications, two corresponding European patents, two corresponding Japanese patents and a corresponding patent application in Europe collectively directed towards methods of maturing dendritic cells and the composition of matter of dendritic cells that have undergone this maturation process. The U.S. patents expire in 2026 and the U.S. applications, if issued, would expire in 2025, the European patents expire in 2025 and 2027, the Japanese patents expire in 2025 and 2027 and the patent application in Europe, if issued, would expire in 2025. |
| • | one U.S. patent application, one U.S. reissue patent application and corresponding U.S patent submitted for reissue, and corresponding patent application in Europe and patent in Japan collectively directed towards methods of manufacture of dendritic cells from monocytes stored for more than six hours and up to four days without freezing and the composition of matter of dendritic cells that have been manufactured from these monocytes. The U.S. patent application, if issued, will expire in 2026, the U.S. reissue patent application, if issued, will expire in 2029, the Japanese patent will expire in 2026 and the patent application in Europe, if issued, would expire in 2026. |
| • | one U.S. patent and one U.S. patent application, one patent and one patent application in Europe and one patent in Japan are collectively directed towards the composition of matter of AGS-004 and related methods of manufacture. The U.S. patent expires in 2026. The U.S. patent application and the patent application in Europe, if issued, would expire in 2025. The European and Japanese patents will expire in 2025. |
| • | one U.S. patent and one U.S. patent application are directed towards the composition of matter and related methods of use of some of the primers that we use in the manufacture of AGS-004. The U.S. patent and U.S. patent application, if issued, will expire in 2028. |
In addition, if the use of Arcelis-based products for the treatment of RCC and HIV are approved by the FDA, then, depending upon factors such as the timing and duration of FDA review and the timing and conditions of FDA approval, as well as factors such as patent claim scope, some of our issued U.S. patents (or patents that may issue from our pending U.S. patent applications) may be eligible for limited patent term extension under the Hatch-Waxman Act.
Trade Secrets
In addition to patents, we rely on trade secrets and know-how to develop and maintain our competitive position. For example, significant aspects of the process by which we manufacture or plan to automate manufacturing of our Arcelis-based drug product candidates are based on unpatented trade secrets and know-how. Trade secrets and know-how can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by confidentiality agreements and invention assignment agreements with our employees, consultants, scientific advisors, contractors and commercial partners. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with a third party. We also seek to preserve the integrity and confidentiality of our data, trade secrets and know-how by maintaining physical security of our premises and physical and electronic security of our information technology systems. Although we have confidence in these individuals, organizations and systems, agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our consultants, contractors or collaborators use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Key Licenses
We are party to a number of license agreements that are important to our business.
Duke University. Pursuant to a 2000 agreement with Duke University, we hold an exclusive worldwide license to specified patents, patent applications and know-how owned or otherwise controlled by Duke, including for use in the development, manufacture and commercialization of dendritic cells loaded with tumor or pathogen RNA. Under the agreement, we:
| • | must pay all costs of prosecution and maintenance of the licensed patent rights; |
| • | must pay an annual minimum royalty to Duke beginning with the calendar year beginning the second January 1 after first approval of a licensed product approved by the FDA or a comparable regulatory authority in a foreign country or any sale of a licensed product that does not require regulatory approval; and |
| • | must pay low single-digit percentage royalties, subject to reduction in specified circumstances, to Duke on net sales of licensed products, which are creditable against the annual minimum royalty. |
| 25 |
We are required to use reasonable commercial diligence to research, develop and market licensed products, to develop manufacturing capabilities, and to sublicense those patent rights for applications which we are not pursuing. If we fail to satisfy these obligations and do not cure such failure after receiving written notice from Duke, Duke may terminate the agreement or convert it to a nonexclusive license.
We may terminate our agreement with Duke at any time upon three months’ written notice. The agreement will terminate upon expiration of the last to expire of the patent rights licensed under the agreement. The U.S. patents licensed under the agreement expire in April 2016 and the patents licensed under the agreement in Europe and Japan expire in 2017. Either party may terminate the agreement upon written notice for fraud, willful misconduct or illegal conduct of the other party that materially adversely affects the terminating party. If either party fails to fulfill any of its material obligations under the agreement, subject to a cure process specified in the agreement, the non-breaching party may terminate the agreement. A party’s ability to cure a breach will only apply to the first two breaches. In addition, the agreement will terminate if we become insolvent, bankrupt or placed in the hands of a receiver or trustee.
Celldex Therapeutics, Inc. In July 2011, we entered into an agreement with Celldex Therapeutics, Inc., or Celldex, pursuant to which Celldex granted us a non-exclusive license to specified patents and patent applications directed to compositions and methods for processing dendritic cells. Upon the execution of the agreement, we paid Celldex $50,000 of a $100,000 upfront license fee. We paid the balance of this fee on January 31, 2012. Under this agreement, we must pay:
| • | a $75,000 annual license fee; |
| • | a specified milestone payment based on the achievement of a specified regulatory milestone; and |
| • | a specified dollar amount per dose of AGS-003 we sell. |
We may terminate our agreement with Celldex at any time upon notice to Celldex. We or Celldex may terminate the agreement, subject to a cure period specified in the agreement, upon a material breach of the other party by providing written notice and waiting a specified period. The agreement will terminate upon the expiration of the last to expire of the patent rights licensed under the agreement on country-by-country basis. The latest date of expiration of the licensed Celldex patents is 2016.
Development and Commercialization Agreements
An important part of our business strategy is to enter into arrangements with third parties for the development and commercialization of our product candidates.
Pharmstandard. In August 2013, in connection with the purchase of shares of our series E preferred stock by Pharmstandard , we entered into an exclusive royalty-bearing license agreement with Pharmstandard. Under this license agreement, we granted Pharmstandard and its affiliates a license, with the right to sublicense, to develop, manufacture and commercialize AGS-003 and other products for the treatment of human diseases, which are developed by Pharmstandard using our individualized immunotherapy platform, in the Russian Federation, Armenia, Azerbaijan, Belarus, Georgia, Kazakhstan, Kyrgyzstan, Moldova, Tajikistan, Turkmenistan, Ukraine and Uzbekistan, which we refer to as the Pharmstandard Territory. We also provided Pharmstandard with a right of first negotiation for development and commercialization rights in the Pharmstandard Territory to specified additional products we may develop.
Under the terms of the license agreement, Pharmstandard licensed us rights to clinical data generated by Pharmstandard under the agreement and granted us an option to obtain an exclusive license outside of the Pharmstandard Territory to develop and commercialize improvements to our Arcelis technology generated by Pharmstandard under the agreement, a non-exclusive worldwide royalty-free license to Pharmstandard improvements to manufacture products using our Arcelis technology and a license to specified follow-on licensed products generated by Pharmstandard outside of the Pharmstandard Territory, each on terms to be negotiated upon our request for a license. In addition, Pharmstandard agreed to pay us pass-through royalties on net sales of all licensed products in the low single digits until it has generated a specified amount of aggregate net sales. Once the net sales threshold is achieved, Pharmstandard will pay us royalties on net sales of specified licensed products, including AGS-003, in the low double digits below 20%. These royalty obligations last until the later of the expiration of specified licensed patent rights in a country or the twelfth anniversary of the first commercial sale in such country on a country by country basis and no further royalties on specified other licensed products. After the net sales threshold is achieved, Pharmstandard has the right to offset a portion of the royalties Pharmstandard pays to third parties for licenses to necessary third party intellectual property against the royalties that Pharmstandard pays to us.
| 26 |
The agreement will terminate upon expiration of the royalty term, upon which all licenses will become fully paid up perpetual exclusive licenses. Either party may terminate the agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy and we may terminate the agreement if Pharmstandard challenges or assists a third party in challenging specified patent rights of ours. If Pharmstandard terminates the agreement upon our material breach or bankruptcy, Pharmstandard is entitled to terminate our licenses to improvements generated by Pharmstandard, upon which we may come to rely for the development and commercialization of AGS-003 and other licensed products outside of the Pharmstandard Territory, and Pharmstandard is entitled to retain its licenses from us and to pay us substantially reduced royalty payments following such termination.
In November 2013, we entered into an agreement with Pharmstandard under which Pharmstandard purchased additional shares of our series E preferred stock. Under this agreement, we agreed to enter into a manufacturing rights agreement for the European market with Pharmstandard and that the manufacturing rights agreement would provide for the issuance of warrants to Pharmstandard to purchase 499,788 shares of our common stock at an exercise price of $5.82 per share. As of March 23, 2016, we had not entered into this manufacturing rights agreement or issued the warrants.
Green Cross. In July 2013, in connection with the purchase of our series E preferred stock by Green Cross, we entered into an exclusive royalty-bearing license agreement with Green Cross. Under this agreement we granted Green Cross a license to develop, manufacture and commercialize AGS-003 for mRCC in South Korea. We also provided Green Cross with a right of first negotiation for development and commercialization rights in South Korea to specified additional products we may develop.
Under the terms of the license, Green Cross has agreed to pay us $500,000 upon the initial submission of an application for regulatory approval of a licensed product in South Korea, $500,000 upon the initial regulatory approval of a licensed product in South Korea and royalties ranging from the mid-single digits to low double digits below 20% on net sales until the fifteenth anniversary of the first commercial sale in South Korea. In addition, Green Cross has granted us an exclusive royalty free license to develop and commercialize all Green Cross improvements to our licensed intellectual property in the rest of the world, excluding South Korea, except that, as to such improvements for which Green Cross makes a significant financial investment and that generate significant commercial benefit in the rest of the world, we are required to negotiate in good faith a reasonable royalty that we will be obligated to pay to Green Cross for such license. Under the terms of the agreement, we are required to continue to develop and to use commercially reasonable efforts to obtain regulatory approval for AGS-003 in the United States
The agreement will terminate upon expiration of the royalty term, which is 15 years from the first commercial sale, upon which all licenses will become fully paid up perpetual non-exclusive licenses. Either party may terminate the agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy and we may terminate the agreement if Green Cross challenges or assists a third party in challenging specified patent rights of ours. If Green Cross terminates the agreement upon our material breach or bankruptcy, Green Cross is entitled to terminate our licenses to improvements and retain its licenses from us and to pay us substantially reduced milestone and royalty payments following such termination.
Medinet. In December 2013, we entered into a license agreement with Medinet. Under this agreement, we granted Medinet an exclusive, royalty-free license to manufacture in Japan AGS-003 and other products using our Arcelis technology solely for the purpose of the development and commercialization of AGS-003 and these other products for the treatment of mRCC. We refer to this license as the manufacturing license. In addition, under this agreement, we granted Medinet an option to acquire a nonexclusive, royalty-bearing license under our Arcelis technology to sell in Japan AGS-003 and other products for the treatment of mRCC. We refer to the option as the sale option and the license as the sale license.
We do not expect Medinet to exercise the sale option before its expiration on March 31, 2016. If Medinet does not exercise the sale option, Medinet may only manufacture AGS-003 and these other products for us or our designee. In addition, if Medinet does not exercise the sale option, we and Medinet have agreed to negotiate in good faith a supply agreement under which Medinet would supply us or our designee with AGS-003 and these other products for development and sale for the treatment of mRCC in Japan. If Medinet exercises the sale option, it may only manufacture AGS-003 and these other products for itself, its related parties and its sublicensees. During the term of the manufacturing license, we may not manufacture AGS-003 or these other products for us or any designee for development or sale for the treatment of mRCC in Japan.
In consideration for the manufacturing license, Medinet paid us $1.0 million. Medinet also loaned us $9.0 million in connection with us entering into the agreement. We have agreed to use these funds in the development and manufacturing of AGS-003 and the other products. Medinet also agreed to pay us milestone payments of up to a total of $9.0 million upon the achievement of developmental and regulatory milestones and $5.0 million upon the achievement of a sales milestone related to AGS-003 and these products. If Medinet exercises the sale option, it will pay us $1.0 million, as well as royalties on net sales at a rate to be negotiated until the later of the expiration of the licensed patent rights in Japan and the twelfth anniversary of the first commercial sale in Japan. If we cannot agree on the royalty rate, we have agreed to submit the matter to arbitration.
| 27 |
We borrowed the $9.0 million pursuant to an unsecured promissory note that bears interest at a rate of 3.0 % per annum. The principal and interest under the note are due and payable on December 31, 2018. Under the terms of the note and the license agreement, any milestone payments related to the developmental and regulatory milestones that become due will be applied first to the repayment of the loan. We have the right to prepay the loan at any time. If we have not repaid the loan by December 31, 2018, then we have agreed to grant to Medinet a non-exclusive, royalty-bearing license to make and sell Arcelis products in Japan for the treatment of cancer. In such event, the amounts owing under the loan as of December 31, 2018 will constitute pre-paid royalties under the license and will not be otherwise due and payable. Royalties under this license would be paid until the expiration of the licensed patent rights in Japan at a rate to be negotiated. If we cannot agree on the royalty rate, we have agreed to submit the matter to arbitration.
Medinet has also granted us a royalty-free, sublicensable, transferable, exclusive license under Medinet improvements to our intellectual property in the rest of the world and for uses other than for mRCC in Japan.
Under the agreement, we have the right to revoke both the manufacturing license and the sale license to be granted to Medinet or the sale license only. If we exercise this right, we will be obligated to make a one-time payment to Medinet calculated based on the nonroyalty payments made to us by Medinet under the agreement, repay the outstanding amount due under the loan and assume certain obligations of Medinet, and Medinet will be obligated to assist us in transitioning the relevant rights in Japan to us or a party that we designate. If we exercise our revocation right with respect to the sale license only, the one-time payment will equal the total amount of nonroyalty payments. If we exercise our revocation right with respect to the manufacturing license and the sale license, the one-time payment will equal 150% or 200% of the nonroyalty payments depending on the timing of the exercise of the revocation right.
The agreement will terminate upon expiration of the royalty term, upon which all licenses will become fully paid up, perpetual non-exclusive licenses. Either party may terminate the agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy, and we may terminate the agreement if Medinet challenges or assists a third party in challenging specified patent rights of ours. If Medinet terminates the agreement upon our material breach or bankruptcy, Medinet is entitled to terminate our licenses to improvements and retain its royalty-bearing licenses from us.
Lummy. On April 7, 2015, we and Lummy HK entered into a license agreement pursuant to which we granted to Lummy HK an exclusive license under the Arcelis technology, including patents, know-how and improvements to manufacture, develop and commercialize products for the treatment of cancer in China, Hong Kong, Taiwan and Macau. Lummy HK also has a right of first negotiation with respect to a license under the Arcelis technology for the treatment of infectious diseases in China, Hong Kong, Taiwan and Macau.
Under the terms of the license agreement, the parties will share relevant data, and we will have a right to reference Lummy HK data for purposes of its development programs under the Arcelis technology. In addition, Lummy HK has granted to us an exclusive, royalty-free license under and to any and all Lummy HK improvements to the Arcelis technology conceived or reduced to practice by Lummy HK and Lummy HK data to develop and/or commercialize products outside China, Hong Kong, Taiwan and Macau, an exclusive, royalty-free license under and to any and all INDs and other regulatory approvals and Lummy HK trademarks used for an Arcelis-Based Product to develop and/or commercialize an Arcelis-Based Product outside China, Hong Kong, Taiwan and Macau and a non-exclusive, worldwide, royalty-free license under any Lummy HK improvements and Lummy HK data to manufacture Arcelis-Based Products anywhere in the world. Lummy HK has the right to reference our data, INDs and other regulatory filings and submissions for the purpose of developing and obtaining regulatory approval of licensed products in China, Hong Kong, Taiwan and Macau.
Pursuant to the license agreement, Lummy HK will pay us royalties on net sales and up to an aggregate of $20.0 million upon the achievement of manufacturing, regulatory and commercial milestones. The license agreement will terminate upon expiration of the last to expire royalty term for all Arcelis-Based Products, with each royalty term being the longer of the expiration of the last valid patent claim covering the applicable Arcelis-Based Product and 10 years from the first commercial sale of such Arcelis-Based Product. Either party may terminate the license agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy. We may terminate the license agreement if Lummy HK challenges or assists a third party in challenging specified patent rights of ours. If Lummy HK terminates the license agreement upon our material breach or bankruptcy, Lummy HK is entitled to terminate the licenses it granted to us and retain its licenses from us with respect to Arcelis-Based Products then in development or being commercialized, subject to Lummy HK’s continued obligation to pay royalties and milestones with respect to such Arcelis-Based Products.
| 28 |
Invetech. On October, 29, 2014, we entered into a development agreement with Invetech Pty Ltd, or Invetech. The development agreement supersedes and replaces the development agreement entered into by the parties as of July 20, 2005. Under the development agreement, Invetech agreed to continue to develop and provide prototypes of the automated production system to be used for the manufacture of our Arcelis-based products, or the Production Systems. Development services will be performed on a proposal by proposal basis. Invetech has agreed to defer 30% of its fees, but such deferral may not exceed $5,000,000. Deferred fees (plus interest of 7% per annum) would become payable either, at our option, in a lump sum within 90 days of the “Sunset Date Trigger Event” or pursuant to an installment plan (either in four installments payable within the first year or eight installments payable within the first two years after the “Sunset Date Trigger Event”). Under the current terms of the agreement, the “Sunset Date Trigger Event” is December 31, 2016 or is June 30, 2016 if prior to such date our ongoing Phase 3 ADAPT clinical trial is closed early following the interim review at 50% or 75% of events, due to a positive efficacy outcome in favor of the active treatment arm that contains AGS-003. Invetech is entitled to a 10% bonus payment if this clinical trial is closed early indicating positive efficacy and Invetech has timely completed all activities up to the time of such early closure. We are planning to renegotiate the terms of the development agreement related to the deferred fees and bonus payment.
The development agreement requires the parties to discuss in good faith Invetech’s supply of Production Systems for use in manufacturing commercial product. We have an obligation to purchase $25.0 million worth of Production Systems, components, subsystems and spare parts for commercial use. Once that obligation has been satisfied, we have the right to have a third party supply Production Systems for use in manufacturing commercial product, provided that Invetech has a right of first refusal with respect to any offer by a third party and we may not accept an offer from a third party unless that offer is at a price that is less than that offered by Invetech and otherwise under substantially the same or better terms. We will own all intellectual property arising from the development services (with the exception of existing Invetech intellectual property incorporated therein-under which we will have a license). The term of the development agreement will continue until the completion of the development of the Production Systems. The development agreement can be terminated early by either party because of a technical failure or by us without cause.
Saint-Gobain. In January 2015, we entered into a development agreement with Saint-Gobain Performance Plastics Corporation, or Saint-Gobain. Under the agreement, Saint-Gobain will develop a range of disposables for use in our automated production systems to be used for the manufacture of our Arcelis-based products, which we refer to as the Disposables. We do not expect the fees and expenses incurred under the Saint-Gobain Agreement to exceed $6,000,000. Saint-Gobain has agreed to defer one half of the fees and expenses incurred until the earlier of (i) the date upon which we have raised an additional $60.0 million in capital, and (ii) September 30, 2016. The Saint-Gobain agreement required the parties to execute a commercial supply agreement under which Saint-Gobain would become the exclusive supplier of Disposables for the manufacture of our products treating solid tumors for no less than fifteen years by December 31, 2015. The parties are in discussions to extend this date and to finalize the commercial supply agreement. The Saint-Gobain agreement will continue until December 31, 2016, but can be terminated earlier by written agreement of the parties because of a material default, including the failure to execute the commercial supply agreement, or a failure to achieve a performance milestone.
Cellscript. In December 2015, we entered into a development and supply agreement with Cellscript, LL. Under the agreement, Cellscript has agreed to develop cGMP processes for the manufacture and production of CD40L RNA, a ribonucleic acid used in the production of our Arcelis -based products, and to manufacture and produce CD40L RNA.
In consideration for these development and production services, we have agreed to pay Cellscript total fees of $4,600,000. Upon the execution of the agreement, we made an initial payment to Cellscript of $2,000,000 through the issuance to Cellscript of 906,194 shares of our common stock. The balance of these fees are payable to Cellscript, at our option, in cash, common stock or a combination of cash and common stock upon the achievement of development milestones. Any shares of common stock issued pursuant to the agreement are subject to a lock-up period of 180 days from the date of issuance of such shares to Cellscript.
Under the terms of the agreement, Cellscript shall be the sole and exclusive manufacturer and supplier to us of CD40L RNA, and we will make agreed upon cash payments to Cellscript for CD40L RNA produced for us during the term of the Agreement. Under the agreement, Cellscript shall also be our sole and exclusive supplier of enzymes and various kits comprising enzymes for transcription, capping and/or polyadenylation of RNA. We will make agreed upon cash payments to Cellscript amounts for each kit that is purchased under the agreement.
The agreement will continue until the earlier of (i) December 31, 2017 or (ii) the effective date of a commercial supply agreement negotiated in good faith by the parties, but can be earlier terminated by either party due to a material breach or upon bankruptcy of the other party.
| 29 |
Government Regulation
Government authorities in the United States, at the federal, state and local level, and in other countries extensively regulate, among other things, the research, development, testing, manufacture, including any manufacturing changes, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, import and export of pharmaceutical and biological products such as those we are developing and may market. The processes for obtaining regulatory approvals in the United States and in foreign countries, along with subsequent compliance with applicable statutes and regulations, require the expenditure of substantial time and financial resources.
U.S. Drug and Biological Product Approval Process
In the United States, the FDA regulates drugs and biological products under the federal Food, Drug, and Cosmetic Act, or FDCA, the Public Health Service Act, or PHSA, and implementing regulations. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local and foreign statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements at any time during the product development process, approval process or after approval, may subject an applicant to a variety of administrative or judicial sanctions, such as the FDA’s refusal to approve pending applications, withdrawal of an approval, imposition of a clinical hold, issuance of warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement or civil or criminal penalties.
The process required by the FDA before a drug or biological product may be marketed in the United States generally involves the following:
| • | completion of preclinical laboratory tests, animal studies and formulation studies in compliance with the FDA’s good laboratory practice, or GLP, regulations; |
| • | submission to the FDA of an IND which must become effective before human clinical trials may begin; |
| • | approval by an independent institutional review board, or IRB, at each clinical site before each trial may be initiated; |
| • | performance of adequate and well-controlled human clinical trials in accordance with good clinical practices, or GCP, to establish the safety and efficacy of the proposed drug or biological product for each indication; |
| • | submission to the FDA of a new drug application, or NDA, or a biologics license application, or BLA; |
| • | satisfactory completion of an FDA advisory committee review, if applicable; |
| • | satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP, and to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality and purity; and |
| • | FDA review and approval of the NDA or BLA. |
Preclinical Studies. Preclinical studies include laboratory evaluation of product chemistry, toxicity and formulation, as well as animal studies to assess its potential safety and efficacy. An IND sponsor must submit the results of the preclinical tests, together with manufacturing information, analytical data and any available clinical data or literature, among other things, to the FDA as part of an IND. Some preclinical testing may continue even after the IND is submitted. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions related to one or more proposed clinical trials and places the clinical trial on a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, submission of an IND may not result in the FDA allowing clinical trials to commence.
Clinical Trials. Clinical trials involve the administration of the investigational new drug to human subjects under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research subjects provide their informed consent in writing for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives of the study, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND. In addition, an IRB at each institution participating in the clinical trial must review and approve the plan for any clinical trial before it commences at that institution. Information about certain clinical trials must be submitted within specific timeframes to the NIH for public dissemination on their ClinicalTrials.gov website.
| 30 |
Human clinical trials are typically conducted in three sequential phases, which may overlap or be combined:
| • | Phase 1: The drug or biological product is initially introduced into healthy human subjects or patients with the target disease or condition and tested for safety, dosage tolerance, absorption, metabolism, distribution, excretion and, if possible, to gain an early indication of its effectiveness. |
| • | Phase 2: The drug or biological product is administered to a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance and optimal dosage. |
| • | Phase 3: The drug or biological product is administered to an expanded patient population, generally at geographically dispersed clinical trial sites, in well-controlled clinical trials to generate enough data to statistically evaluate the efficacy and safety of the product for approval, to establish the overall risk-benefit profile of the product, and to provide adequate information for the labeling of the product. |
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently if serious adverse events occur. Phase 1, Phase 2 and Phase 3 clinical trials may not be completed successfully within any specified period, or at all. Furthermore, the FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research subjects are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the drug has been associated with unexpected serious harm to patients.
Special Protocol Assessment. The SPA process is designed to facilitate the FDA’s review and approval of drug and biological products by allowing the FDA to evaluate the proposed design and size of Phase 3 clinical trials that are intended to form the primary basis for determining a drug or biological product’s efficacy. Upon specific request by a clinical trial sponsor, the FDA will evaluate the trial protocol and respond to a sponsor’s questions regarding, among other things, primary efficacy endpoints, trial conduct and data analysis, within 45 days of receipt of the request. The FDA ultimately assesses whether the trial protocol design and planned analysis of the trial adequately address objectives in support of a regulatory submission. All agreements and disagreements between the FDA and the sponsor regarding an SPA must be clearly documented in an SPA letter or the minutes of a meeting between the sponsor and the FDA.
Even if the FDA agrees to the design, execution and analyses proposed in protocols reviewed under an SPA, the FDA may revoke or alter its agreement under the following circumstances:
| • | public health concerns emerge that were unrecognized at the time of the protocol assessment; |
| • | a sponsor fails to follow a protocol that was agreed upon with the FDA; |
| • | the relevant data, assumptions, or information provided by the sponsor in a request for SPA change are found to be false statements or misstatements or are found to omit relevant facts; or |
| • | the FDA and the sponsor agree in writing to modify the trial protocol and such modification is intended to improve the study. |
Marketing Approval. Assuming successful completion of the required clinical testing, the results of the preclinical and clinical studies, together with detailed information relating to the product’s chemistry, manufacture, controls and proposed labeling, among other things, are submitted to the FDA as part of an NDA or BLA requesting approval to market the product for one or more indications. In most cases, the submission of an NDA or BLA is subject to a substantial application user fee.
In addition, under the Pediatric Research Equity Act of 2003, or PREA, as amended and reauthorized, an NDA, BLA or supplement to an NDA or BLA for certain types of new drug or biological products must contain data that are adequate to assess the safety and effectiveness of the drug or biological product for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements. Unless otherwise required by regulation, the pediatric data requirements do not apply to products with orphan designation.
The Food and Drug Administration Safety and Innovation Act, or FDASIA, amended the FDCA and requires that a sponsor who is planning to submit a marketing application for a drug or biological product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration submit an initial Pediatric Study Plan, or PSP, within sixty days of an end-of-phase 2 meeting or as may be agreed between the sponsor and FDA. The initial PSP must include an outline of the pediatric study or studies that the sponsor plans to conduct, including study objectives and design, age groups, relevant endpoints and statistical approach, or a justification for not including such detailed information, and any request for a deferral of pediatric assessments or a full or partial waiver of the requirement to provide data from pediatric studies along with supporting information. FDA and the sponsor must reach agreement on the PSP. A sponsor can submit amendments to an agreed-upon initial PSP at any time if changes to the pediatric plan need to be considered based on data collected from nonclinical studies, early phase clinical trials, or other clinical development programs.
| 31 |
The FDA also could require submission of a risk evaluation and mitigation strategy, or REMS, plan to mitigate any identified or suspected serious risks. The REMS plan could include medication guides, physician communication plans, assessment plans, and elements to assure safe use, such as restricted distribution methods, patient registries, or other risk minimization tools.
The FDA conducts a preliminary review of all NDAs and BLAs within the first 60 days after submission before accepting them for filing to determine whether they are sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an NDA or BLA for filing. In this event, the application must be resubmitted with the additional information. The resubmitted application is also subject to review before the FDA accepts it for filing. Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews an NDA or BLA to determine, among other things, whether the product is safe and effective (described as safe, pure and potent for BLAs) and the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product’s continued safety, purity and potency. The FDA is required to refer an application for a novel drug or biological product to an advisory committee or explain why such referral was not made. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Before approving an NDA or BLA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving an NDA or BLA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP and integrity of the clinical data submitted.
The testing and approval process requires substantial time, effort and financial resources, and each may take several years to complete. Data obtained from clinical activities are not always conclusive and may be susceptible to varying interpretations, which could delay, limit or prevent regulatory approval. The FDA may not grant approval on a timely basis, or at all. We may encounter difficulties or unanticipated costs in our efforts to develop our product candidates and secure necessary governmental approvals, which could delay or preclude us from marketing our products.
If the FDA’s evaluation of the NDA or BLA and inspection of the manufacturing facilities and clinical trial sites are favorable, the FDA may issue an approval letter, or, in some cases, a complete response letter. A complete response letter generally contains a statement of specific conditions that must be met in order to secure final approval of the NDA or BLA and may require additional clinical or preclinical testing in order for FDA to reconsider the application. If and when those conditions have been met to the FDA’s satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug or biological product with specific prescribing information for specific indications. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
Even if the FDA approves a product, it may limit the approved indications for use for the product, require that contraindications, warnings or precautions be included in the product labeling, require that post-approval studies, including Phase 4 clinical trials, be conducted to further assess a drug’s safety after approval, require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution restrictions or other risk management mechanisms, which can materially affect the potential market and profitability of the product. The FDA may prevent or limit further marketing of a product based on the results of post-marketing studies or surveillance programs. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval.
Special FDA Expedited Review and Approval Programs. The FDA has various programs, including fast track designation, accelerated approval and priority review, that are intended to expedite or simplify the process for the development and FDA review of drug and biological products that are intended for the treatment of serious or life threatening diseases or conditions and demonstrate the potential to address unmet medical needs. The purpose of these programs is to provide important new drugs to patients earlier than under standard FDA review procedures.
| 32 |
To be eligible for a fast track designation, the FDA must determine, based on the request of a sponsor, that a product is intended to treat a serious aspect of a serious or life threatening disease or condition and will fill an unmet medical need. The FDA will determine that a product will fill an unmet medical need if it will provide a therapy where none exists or provide a therapy that may be potentially superior to existing therapy based on efficacy or safety factors.
In addition, the FDA may give a priority review designation to drugs or biological products that provide safe and effective therapy where no satisfactory alternative exists or a significant improvement compared to marketed products in the treatment, diagnosis or prevention of a disease. For products regulated by the Center for Biologics Evaluation and Research, or CBER, the product must be intended to treat a serious or life- threatening disease or condition. A priority review means that the targeted time for the FDA to review an application is six months, rather than ten months. Most products that are eligible for fast track designation are also likely to be considered appropriate to receive a priority review.
Under the provisions of the Food and Drug Administration Safety and Innovation Act, or FDASIA, enacted in 2012, a sponsor also can request designation of a product candidate as a “breakthrough therapy.” A breakthrough therapy is defined as a drug or biological product that is intended, alone or in combination with one or more other drugs or biological products, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. Products designated as breakthrough therapies are also eligible for accelerated approval. The FDA must take certain actions, such as holding timely meetings and providing advice, intended to expedite the development and review of an application for approval of a breakthrough therapy.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.
Post-Approval Requirements. Any drug or biological products manufactured or distributed by us pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior FDA review and approval. There also are continuing, annual user fee requirements for any marketed products and the establishments at which such products are manufactured, as well as new application fees for supplemental applications with clinical data.
The FDA may impose a number of post-approval requirements as a condition of approval of an NDA or BLA. For example, the FDA may require post-marketing testing, including Phase 4 clinical trials, and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization. Regulatory approval of oncology products often requires that patients in clinical trials be followed for long periods to determine the overall survival benefit of the drug or biologic.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs and biological products are required to register their establishments with the FDA and state agencies, and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP and impose reporting and documentation requirements upon us and any third party manufacturers that we may decide to use. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance.
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, among other things:
| • | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| • | fines, warning letters or holds on post-approval clinical trials; |
| • | refusal of the FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals; |
| 33 |
| • | product seizure or detention, or refusal to permit the import or export of products; or |
| • | injunctions or the imposition of civil or criminal penalties. |
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off label uses, and a company that is found to have improperly promoted off label uses may be subject to significant liability.
In addition, the distribution of prescription pharmaceutical and biological products is subject to the Prescription Drug Marketing Act, or PDMA, which regulates the distribution of drugs and drug samples at the federal level, and sets minimum standards for the registration and regulation of drug distributors by the states. Both the PDMA and state laws limit the distribution of prescription pharmaceutical product samples and impose requirements to ensure accountability in distribution.
Exclusivity and Approval of Competing Products
Non-Patent Exclusivity. Under the Patient Protection and Affordable Care Act, or PPACA, newly- approved biological products may benefit from statutory periods of non-patent data and marketing exclusivity. The PPACA, among other things, permits the FDA to approve biosimilar or interchangeable versions of biological products through an abbreviated approval pathway following periods of data and marketing exclusivity. Biological products that are considered to be “reference products” are granted two overlapping periods of data and marketing exclusivity: a four-year period during which no abbreviated biologics license application, or abbreviated BLA, relying upon the reference product may be submitted to the FDA, and a twelve-year period during which no abbreviated BLA relying upon the reference product may be approved by FDA. For purposes of the PPACA, a reference product is defined as the single biological product licensed under a full BLA against which a biological product is evaluated in an application submitted under an abbreviated BLA.
We believe that our investigational products, if approved via full BLAs, will be considered “reference products” that are entitled to both four-year and twelve-year exclusivity under the PPACA. The FDA, however, has not issued any regulations or final guidance explaining how it will implement the PPACA, including the exclusivity provisions for reference products. Since February 2012, the FDA has issued six draft guidance documents that provide its preliminary thoughts on how to interpret and implement the abbreviated BLA provisions of the PPACA. The FDA has requested public comments on these draft guidance documents, including the proper interpretation of PPACA exclusivity provisions. It is thus possible that the FDA will decide to interpret the PPACA in such a way that our products are not considered to be reference products for purposes of the PPACA or be entitled to any period of data or marketing exclusivity. Even if our products are considered to be reference products and obtain exclusivity under the PPACA, another company nevertheless could also market a competing version of any of our biological products if such company can complete, and the FDA permits the submission of and approves, a full BLA. Although protection under PPACA will not prevent the submission or approval of another “full” BLA, the applicant would be required to conduct its own preclinical and adequate and well-controlled clinical trials to demonstrate safety, purity, and potency (i.e., effectiveness).
Pediatric Exclusivity. Pediatric exclusivity is another type of non-patent marketing exclusivity in the United States and, if granted, provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory exclusivity, including the four- and 12-year non-patent exclusivity periods described above. This six-month exclusivity may be granted based on the voluntary completion of a pediatric study or studies in accordance with an FDA-issued “Written Request” for such a study or studies.
Orphan Drug Designation and Exclusivity. Under the Orphan Drug Act, the FDA may grant orphan drug designation to a drug (including a biologic) intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making available in the United States a drug for this type of disease or condition will be recovered from sales in the United States for that drug. Orphan drug designation must be requested before submitting an NDA or BLA. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA.
If a product that has orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan product exclusivity, which means that the FDA may not approve any other applications, including a full NDA or full BLA, to market the same drug for the same indication for seven years. For purposes of small molecule drugs, the FDA defines “same drug” as a drug that contains the same active moiety and is intended for the same use as the previously approved orphan drug. For purposes of large molecule drugs, the FDA defines “same drug” as a drug that contains the same principal molecular structural features, but not necessarily all of the same structural features, and is intended for the same use as the drug in question. Notwithstanding the above definitions, a drug that is clinically superior to an orphan drug will not be considered the “same drug” and thus will not be blocked by orphan drug exclusivity.
| 34 |
A designated orphan drug may not receive orphan drug exclusivity if it is approved for a use that is broader than the indication for which it received orphan designation. In addition, orphan drug exclusive marketing rights in the United States may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the drug to meet the needs of patients with the rare disease or condition.
The FDA also administers a clinical research grants program, whereby researchers may compete for funding to conduct clinical trials to support the approval of drugs, biologics, medical devices, and medical foods for rare diseases and conditions. An application for an orphan grant should propose one discrete clinical trial to facilitate FDA approval of the product for a rare disease or condition. The study may address an unapproved new product or an unapproved new use for a product already on the market.
Foreign Regulation
Although we do not currently market any of our products outside the United States and have no current plans to engage in product commercialization outside the United States, we may decide to do so in the future. In order to market any product outside of the United States, we would need to comply with numerous and varying regulatory requirements of other countries regarding safety and efficacy and governing, among other things, clinical trials, marketing authorization, commercial sales and distribution of our products. Whether or not we obtain FDA approval for a product, we would need to obtain the necessary approvals by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country and can involve additional product testing and additional administrative review periods, and may be otherwise complicated by some of our products and product candidates being controlled substances. The time required to obtain approval in other countries might differ from and be longer than that required to obtain FDA approval. Regulatory approval in one country does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country may negatively impact the regulatory process in others.
Pharmaceutical Coverage, Pricing and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any drug products for which we obtain regulatory approval. Sales of any of our product candidates, if approved, will depend, in part, on the extent to which the costs of the products will be covered by third party payors, including government health programs such as Medicare and Medicaid, commercial health insurers and managed care organizations. The process for determining whether a payor will provide coverage for a drug product may be separate from the process for setting the price or reimbursement rate that the payor will pay for the drug product once coverage is approved. Third party payors may limit coverage to specific drug products on an approved list, or formulary, which might not include all of the approved drugs for a particular indication.
In order to secure coverage and reimbursement for any product that might be approved for sale, we may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost- effectiveness of the product, in addition to the costs required to obtain FDA or other comparable regulatory approvals. Our product candidates may not be considered medically necessary or cost-effective. A payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Third party reimbursement may not be sufficient to enable us to maintain price levels high enough to realize an appropriate return on our investment in product development.
The containment of healthcare costs has become a priority of federal, state and foreign governments, and the prices of drugs have been a focus in this effort. Third party payors are increasingly challenging the prices charged for medical products and services and examining the medical necessity and cost-effectiveness of medical products and services, in addition to their safety and efficacy. If these third party payors do not consider our products to be cost-effective compared to other available therapies, they may not cover our products after approval as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell our products at an appropriate return on investment. The U.S. government, state legislatures and foreign governments have shown significant interest in implementing cost-containment programs to limit the growth of government-paid health care costs, including price controls, restrictions on reimbursement and requirements for substitution of generic products for branded prescription drugs. Adoption of such controls and measures, and tightening of restrictive policies in jurisdictions with existing controls and measures, could limit payments for pharmaceuticals such as the drug product candidates that we are developing and could adversely affect our net revenue and results.
| 35 |
Pricing and reimbursement schemes vary widely from country to country. Some countries provide that drug products may be marketed only after a reimbursement price has been agreed. Some countries may require the completion of additional studies that compare the cost-effectiveness of a particular product candidate to currently available therapies. For example, the European Union provides options for its member states to restrict the range of drug products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. European Union member states may approve a specific price for a drug product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the drug product on the market. Other member states allow companies to fix their own prices for drug products, but monitor and control company profits. The downward pressure on health care costs in general, particularly prescription drugs, has become intense. As a result, increasingly high barriers are being erected to the entry of new products. In addition, in some countries, cross-border imports from low-priced markets exert competitive pressure that may reduce pricing within a country. There can be no assurance that any country that has price controls or reimbursement limitations for drug products will allow favorable reimbursement and pricing arrangements for any of our products.
The marketability of any products for which we receive regulatory approval for commercial sale may suffer if the government and third party payors fail to provide adequate coverage and reimbursement. In addition, emphasis on managed care in the United States has increased and we expect will continue to increase the pressure on drug pricing. Coverage policies, third party reimbursement rates and drug pricing regulation may change at any time. In particular, the PPACA and a related reconciliation bill, which we collectively refer to as the Affordable Care Act or ACA, contain provisions that may reduce the profitability of drug products, including, for example, increased rebates for covered outpatient drugs sold to Medicaid programs, extension of Medicaid rebates to Medicaid managed care plans, mandatory discounts for certain Medicare Part D beneficiaries, and annual fees based on pharmaceutical companies’ share of sales to federal health care programs. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
New Legislation and Regulations
From time to time, legislation is drafted, introduced and passed in Congress that could significantly change the statutory provisions governing the testing, approval, manufacturing and marketing of products regulated by the FDA. For example, the FDASIA and PPACA provisions discussed above were enacted in 2012 and 2010, respectively. In addition to new legislation, FDA regulations and policies are often revised or interpreted by the agency in ways that may significantly affect our business and our products. It is impossible to predict whether further legislative changes will be enacted, or FDA regulations, guidance, policies or interpretations changed or what the impact of such changes, if any, may be.
Segment and Geographic Information
Operating segments are defined as components of an enterprise engaging in business activities from which it may earn revenues and incur expenses, for which discrete financial information is available and whose operating results are regularly reviewed by the chief operating decision maker in deciding how to allocate resources and in assessing performance. We view our operations and manage our business in one operating segment and all of our operations are in North America.
Employees
As of February 29, 2016, we had 136 employees, including 20 in research and development, 13 in clinical development, 84 in manufacturing and 19 in general and administrative functions. None of our employees is subject to a collective bargaining agreement or represented by a labor or trade union. We believe that our relations with our employees are good.
Corporate Information
We were incorporated in the State of Delaware on May 8, 1997. Our principal executive offices are located at 4233 Technology Drive, Durham, North Carolina 27704, and our telephone number is (919) 287-6300.
Available Information
We file with the Securities and Exchange Commission, or SEC, annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy and information statements and amendments to reports filed or furnished pursuant to Sections 13(a), 14 and 15(d) of the Securities Exchange Act of 1934, as amended. The public may obtain these filings at the SEC’s Public Reference Room at 100 F Street, NE, Washington, DC 20549 or by calling the SEC at 1-800-SEC-0330. The SEC also maintains a website at http://www.sec.gov that contains reports, proxy and information statements and other information regarding Argos and other companies that file materials with the SEC electronically. As soon as practicable after filing with the SEC, copies of our reports on Form 10-K, Forms 10-Q and Forms 8-K may also be obtained, free of charge, electronically through the investor relations portion of our web site, www.argostherapeutics.com/investor-relations/sec-filings/default.aspx.
| 36 |
We webcast our earnings calls on our investor relations website. Additionally, we provide notifications of news or announcements regarding our financial performance, including SEC filings, investor events and press and earnings releases, on the investor relations portion of our website. Further corporate governance information, including our corporate governance guidelines, board committee charters, Code of Business Conduct and Ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, or persons performing similar functions, is also available on our investor relations website under the heading “Corporate Governance.” The contents of our website are not intended to be incorporated by reference into this Annual Report on Form 10-K or in any other report or document we file with the SEC.
| 37 |
We operate in a dynamic and rapidly changing business environment that involves multiple risks and substantial uncertainty. The following discussion addresses risks and uncertainties that could cause, or contribute to causing, actual results to differ from expectations in material ways. In evaluating our business, investors should pay particular attention to the risks and uncertainties described below and in other sections of this Annual Report on Form 10-K and in our subsequent filings with the SEC. These risks and uncertainties, or other events that we do not currently anticipate or that we currently deem immaterial also may affect our results of operations, cash flows and financial condition. The trading price of our common stock could also decline due to any of these risks, and you could lose all or part of your investment.
Risks Related to the Development and Regulatory Approval of Our Product Candidates
We depend heavily on the success of our two product candidates, AGS-003 and AGS-004, both of which are still in clinical development. Clinical trials of our product candidates may not be successful. If we are unable to commercialize our product candidates or experience significant delays in doing so, our business will be materially harmed.
We currently have no products approved for sale. We have invested a significant portion of our efforts and financial resources in the development of AGS-003 for the treatment of metastatic renal cell carcinoma, or mRCC, and other cancers and AGS-004 for the treatment of HIV. Our ability to generate product revenues, which we do not expect will occur for at least the next several years, if ever, will depend heavily on the successful development and eventual commercialization of these product candidates. The success of these product candidates will depend on several factors, including the following:
| • | successful completion of clinical trials, including clinical results that are statistically significant as well as clinically meaningful in the context of the indications for which we are developing our product candidates; |
| • | receipt of marketing approvals from the FDA and similar regulatory authorities outside the United States; |
| • | establishing commercial manufacturing capabilities by building out and equipping a commercial manufacturing facility for our Arcelis-based product candidates or, alternatively, entering into a commercial supply arrangement with a contract manufacturer; |
| • | maintaining patent and trade secret protection and regulatory exclusivity for our product candidates, both in the United States and internationally; |
| • | launching commercial sales of the products, if and when approved, whether alone or in collaboration with others; |
| • | commercial acceptance of our products, if and when approved, by patients, the medical community and third party payors; |
| • | obtaining and maintaining healthcare coverage and adequate reimbursement; |
| • | effectively competing with other therapies; and |
| • | a continued acceptable safety profile of the products following any marketing approval. |
If we do not achieve one or more of these factors in a timely manner or at all, we could experience significant delays or an inability to successfully commercialize our product candidates, which would materially harm our business.
If clinical trials of our product candidates, such as our ADAPT trial of AGS-003, fail to demonstrate safety and efficacy to the satisfaction of the FDA or similar regulatory authorities outside the United States or do not otherwise produce positive results, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of our product candidates.
Before obtaining regulatory approval for the sale of our product candidates, we must conduct extensive clinical trials to demonstrate the safety and efficacy of our product candidates in humans. Clinical testing is expensive, difficult to design and implement, can take many years to complete and is uncertain as to outcome. A failure of one or more of our clinical trials can occur at any stage of testing. The outcome of preclinical testing and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial do not necessarily predict final results. For instance, despite observing positive results in earlier clinical trials of AGS-004, we failed to achieve the primary endpoint of our Phase 2b clinical trial of AGS-004. Similarly, recommendations of the independent data monitoring committee that the ADAPT trial should be continued based on results of the committee’s interim data analyses of interim trial results may not be indicative as to results of the trial. Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical trials have nonetheless failed to obtain marketing approval of their products.
| 38 |
In particular, to date, we have not completed a clinical trial of AGS-003 against a placebo or a comparator therapy. While we believe comparisons to results from other reported clinical trials or from analyses of data from the International Metastatic Renal Cell Carcinoma Database Consortium, or the Consortium, can assist in evaluating the potential efficacy of our AGS-003 product candidate, there are many factors that affect the outcome for patients, some of which are not apparent in published reports. As a result, results from two different trials or between a trial and an analysis of a treatment database often cannot be reliably compared. Our ADAPT trial of AGS-003 is intended to compare directly the combination of AGS-003 and sunitinib or another targeted therapy to treatment with sunitinib or another targeted therapy monotherapy. Based on the design of the trial, the data from the trial will need to demonstrate an increase of approximately six months in median overall survival for the AGS-003 plus sunitinib / targeted therapy arm as compared to the sunitinib / targeted therapy monotherapy control arm in order to show statistical significance and achieve the primary endpoint of the trial. We will need to show this statistically significant benefit of the combined therapy as compared to treatment with the sunitinib / targeted therapy monotherapy as part of a submission for approval of AGS-003. However, demonstration of statistical significance and achievement of the primary endpoint of the trial do not assure approval by the FDA or similar regulatory authorities outside the United States.
Patients in our ADAPT trial who receive treatment with sunitinib / targeted therapy monotherapy may not have results similar to patients studied in clinical trials of sunitinib or to patients in the Consortium database who were treated with sunitinib. If the patients in our ADAPT trial who receive sunitinib / targeted therapy plus placebo have results which are better than the results that occurred in those other clinical trials or the results described in the Consortium database, we may not demonstrate a sufficient benefit from AGS-003 in combination with sunitinib to allow the FDA to approve AGS-003 for marketing. In addition, only 21 patients received the combination of AGS-003 and sunitinib in our Phase 2 clinical trial. If the patients in our ADAPT trial who receive the combination of AGS-003 and sunitinib / targeted therapy have results which are worse than the results that occurred in our Phase 2 clinical trial, we may not demonstrate a sufficient benefit from the combination therapy to allow the FDA to approve AGS-003 for marketing.
For drug and biological products, the FDA typically requires the successful completion of two adequate and well-controlled clinical trials to support marketing approval because a conclusion based on two such trials will be more reliable than a conclusion based on a single trial. In the case of AGS-003, which is intended for a life-threatening disease, we intend to seek approval based upon the results of a single pivotal Phase 3 clinical trial, our ADAPT trial. The FDA reviewed our plans to conduct our ADAPT trial under its special protocol assessment, or SPA, process. In February 2013, the FDA advised us in a letter that it had completed its review of our plans under the SPA process. The FDA also informed us that in order for a single trial to support approval of an indication, the trial must be well conducted, and the results of the trial must be internally consistent, clinically meaningful and statistically very persuasive. If the results for the primary endpoint are not robust, are subject to confounding factors, or are not adequately supported by other trial endpoints, the FDA may refuse to approve our biologics license application, or BLA based upon a single clinical trial. In addition, because only 21 patients received the combination of AGS-003 and sunitinib in our Phase 2 clinical trial, and as a result, we did not have enough evaluable patients to perform the statistical analysis to determine whether the primary endpoint of complete response rate was achieved in that trial, we expect that the data from our Phase 2 clinical trial will have only a limited impact on the FDA’s ultimate assessment of efficacy of AGS-003. Thus, there can be no guarantee that the FDA will not require additional pivotal clinical trials as a condition for approving AGS-003.
If we are required to conduct additional clinical trials or other testing of our product candidates beyond those that we currently contemplate, if we are unable to successfully complete clinical trials of our product candidates or other testing, if the results of these trials or tests are not positive or are only modestly positive or if there are safety concerns, we may:
| • | be delayed in obtaining marketing approval for our product candidates; |
| • | not obtain marketing approval at all; |
| • | obtain approval for indications or patient populations that are not as broad as intended or desired; |
| • | obtain approval with labeling that includes significant use restrictions or safety warnings, including boxed warnings; |
| • | be subject to additional post-marketing testing requirements; |
| • | be subject to restrictions on how the product is distributed or used; or |
| 39 |
| • | have the product removed from the market after obtaining marketing approval. |
If we experience any of a number of possible unforeseen events in connection with our clinical trials, potential marketing or commercialization of our product candidates could be delayed or prevented.
We may experience numerous unforeseen events during, or as a result of, clinical trials that could delay or prevent our ability to receive regulatory approval or commercialize our product candidates. For example, in September 2011, the FDA placed the original protocol for our ADAPT trial of AGS-003 in combination with sunitinib on partial clinical hold due to unresolved questions regarding the planned measurement of the secretion of the cytokine interleukin-12, or IL-12, as part of the specifications for the release of AGS-003. We subsequently reached an agreement with the FDA regarding the IL-12 release specifications and the FDA lifted the partial clinical hold.
Unforeseen events that could delay or prevent our ability to receive regulatory approval or commercialize our product candidates include:
| • | regulators or institutional review boards may not authorize us or our investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site; |
| • | we may have delays in reaching or fail to reach agreement on acceptable clinical trial contracts or clinical trial protocols with prospective trial sites; |
| • | clinical trials of our product candidates may produce negative or inconclusive results, and we may decide, or regulators may require us, to conduct additional clinical trials or abandon product development programs; |
| • | the number of patients required for clinical trials of our product candidates may be larger than we anticipate, enrollment in these clinical trials may be slower than we anticipate or participants may drop out of these clinical trials at a higher rate than we anticipate; for example, in our Phase 2b clinical trial of AGS-004, we experienced a higher dropout rate than we anticipated due to the higher than expected number of patients who did not complete the full 12 week antiretroviral treatment interruption required by the protocol for the trial; |
| • | our third party contractors may fail to comply with regulatory requirements or meet their contractual obligations to us in a timely manner, or at all; |
| • | we may decide, or regulators or institutional review boards may require us or our investigators to, suspend or terminate clinical research for various reasons, including noncompliance with regulatory requirements or a finding that the participants are being exposed to unacceptable health risks; |
| • | the cost of clinical trials of our product candidates may be greater than we anticipate; and |
| • | the supply or quality of our product candidates or other materials necessary to conduct clinical trials of our product candidates may be insufficient or inadequate. |
In addition, the patients recruited for clinical trials of our product candidates may have a disease profile or other characteristics that are different than we expect and different than the clinical trials were designed for, which could adversely impact the results of the clinical trials. For instance, our Phase 2 combination therapy clinical trial of AGS-003 in combination with sunitinib was originally designed to enroll patients with favorable disease risk profiles and intermediate disease risk profiles and with a primary endpoint of complete response rate. However, the actual trial population consisted entirely of patients with intermediate disease risk profiles and poor disease risk profiles. This is a population for which published research has shown that sunitinib alone, as well as other of the targeted therapies for mRCC, rarely if ever produce complete responses in mRCC, and in our Phase 2 clinical trial in this population the combination therapy of AGS-003 and sunitinib did not show a complete response rate that met the endpoint of the trial.
Our product development costs will also increase if we experience delays in testing or obtaining marketing approvals. We do not know whether any clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. For example, in response to our submission of an investigational new drug application, or IND, for AGS-004, the FDA raised safety concerns regarding the analytical treatment interruption contemplated by our protocol for our Phase 2 clinical trial of AGS-004, and required a one year safety follow-up after the final dose for each patient. This resulted in the need for an amendment to the trial protocol and a four month delay prior to initiating the Phase 2 clinical trial in the United States. In addition to additional costs, significant clinical trial delays also could shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before we do and impair our ability to commercialize our product candidates and may harm our business and results of operations.
| 40 |
The FDA has reviewed the protocol for our ADAPT trial of AGS-003 in combination with sunitinib / targeted therapy under the SPA process. However, agreement by the FDA with the protocol under the SPA process does not guarantee that the trial will be successful or that, if successful, AGS-003 will receive marketing approval.
The FDA has reviewed, under the SPA process, the protocol for our ADAPT trial of AGS-003 in combination with sunitinib / targeted therapy. The SPA process is designed to facilitate the FDA’s review and approval of drug and biological products by allowing the FDA to evaluate the proposed design and size of Phase 3 clinical trials that are intended to form the primary basis for determining a drug candidate’s efficacy. In February 2012, we received a letter from the FDA advising us that the FDA had completed its review of our protocol for the ADAPT trial under the SPA process. In the letter, the FDA stated that it had determined that the protocol sufficiently addressed the trial’s objectives and that the trial was adequately designed to provide the necessary data to support a submission for marketing approval.
An SPA does not guarantee that AGS-003 will receive marketing approval. The FDA may raise issues related to safety, trial conduct, bias, deviation from the protocol, statistical power, patient completion rates, changes in scientific or medical parameters or internal inconsistencies in the data prior to making its final decision. The FDA may also seek the guidance of an outside advisory committee prior to making its final decision. In addition, the combination of AGS-003 and sunitinib may not achieve the primary endpoint of the trial. Even if the primary endpoint in our ADAPT trial is achieved, AGS-003 may not be approved. Many companies which have been granted SPAs have ultimately failed to obtain final approval to market their products.
In its February 2012 letter, the FDA informed us that in order for a single trial to support approval of an indication, the trial must be well conducted, and the results of the trial must be internally consistent, clinically meaningful and statistically very persuasive. If the results for the primary endpoint are not robust, are subject to confounding factors, or are not adequately supported by other trial endpoints, the FDA may refuse to approve our BLA based upon a single clinical trial. There can be no guarantee that the FDA will not require additional pivotal clinical trials as a condition for approving AGS-003.
If we experience delays or difficulties in the enrollment of patients in our clinical trials, our receipt of necessary regulatory approvals could be delayed or prevented.
We may not be able to initiate or continue clinical trials for our product candidates if we are unable to locate and enroll a sufficient number of eligible patients to participate in these trials as required by the FDA or similar regulatory authorities outside the United States. Our competitors may have ongoing clinical trials for product candidates that could be competitive with our product candidates, and patients who would otherwise be eligible for our clinical trials may instead enroll in clinical trials of our competitors’ product candidates. For example, during the Phase 1/2 monotherapy clinical trial of AGS-003 that we conducted, our ability to enroll patients in the trial was adversely affected by the FDA’s approval of sorafenib and sunitinib, because patients did not want to receive, and physicians were reluctant to administer, AGS-003 as a monotherapy once new therapies that showed efficacy in clinical trials were introduced to the market and became widely available.
Patient enrollment is affected by other factors including:
| • | severity of the disease under investigation; |
| • | eligibility criteria for the trial in question; |
| • | perceived risks and benefits of the product candidate under study; |
| • | efforts to facilitate timely enrollment in clinical trials; |
| • | patient referral practices of physicians; |
| • | the ability to monitor patients adequately during and after treatment; and |
| • | proximity and availability of clinical trial sites for prospective patients. |
| 41 |
The actual amount of time for full enrollment of our clinical trials could be longer than planned. Enrollment delays in any of our clinical trials may result in increased development costs for our product candidates, which would cause the value of the company to decline and limit our ability to obtain additional financing. Our inability to enroll a sufficient number of patients for any of our other clinical trials would result in significant delays or may require us to abandon one or more clinical trials altogether.
We are developing AGS-004 for use with latency reversing drugs to eradicate HIV. If latency reversing drugs are not successfully developed for HIV on a timely basis or at all, we will be unable to develop AGS-004 for this use or will be delayed in doing so. In addition, because there are currently no products approved for HIV eradication, we cannot be certain of the clinical trials that we will need to conduct or the regulatory requirements that we will need to satisfy in order to obtain marketing approval of AGS-004 for this purpose.
We are focusing our development program for AGS-004 on the use of AGS-004 in combination with latency reversing drugs, including Vorinostat, to eradicate HIV. We plan to rely on these latency reversing drugs because we recognize that the ultimate objective of virus eradication is unlikely to be achieved with immunotherapy alone because the immune system is not able to recognize the HIV virus in latently infected cells with a low level or lack of expression of HIV antigens.
Several companies and academic groups are evaluating latency reversing drugs that can potentially activate latently infected cells to increase viral antigen expression and make the cells vulnerable to elimination by the immune system. We are not a party to any arrangements with these companies or academic groups. If these companies or academic groups determine not to develop latency reversing drugs for this purpose because the drugs do not sufficiently increase viral antigen expression or have unacceptable toxicities, or these companies or academic groups otherwise determine to collaborate with other developers of immunotherapies on a combination therapy for complete virus eradication, we will not be able to complete our AGS-004 development program. In addition, if these companies or academic groups do not proceed with such development on a timely basis, our AGS-004 program correspondingly would be delayed.
A number of the latency reversing drugs being evaluated for use in HIV patients are currently approved in the United States and elsewhere for use in the treatment of specified cancer indications. For instance Vorinostat is approved for cutaneous T cell lymphoma. If these drugs are not approved by the FDA or equivalent foreign regulatory authorities for use in HIV, the FDA and these other regulatory authorities may not approve AGS-004 without the latency reversing drug having received marketing approval for HIV. If the FDA and these other regulatory authorities approve AGS-004 without the approval of the latency reversing drug for HIV, the use of AGS-004 in combination with the latency reversing drug for virus eradication would require sales of the latency reversing drug for off-label use. In such event, the success of the combination of AGS-004 and the latency reversing drug would be subject to the willingness of physicians, patients, healthcare payors and others in the medical community to use the latency reversing drug for off-label use and of government authorities and third party payors to pay for the combination therapy. In addition, we would be limited in our ability to market the combination for its intended use if the latency reversing drug were to be used off-label.
Currently, there are no products approved for the eradication of HIV. As a result, we cannot be certain as to the clinical trials we will need to conduct or the regulatory requirements that we will need to satisfy in order to obtain marketing approval of AGS-004 for the eradication of HIV.
If serious adverse or inappropriate side effects are identified during the development of our product candidates, we may need to abandon or limit our development of some of our product candidates.
All of our product candidates are still in preclinical or clinical development and their risk of failure is high. It is impossible to predict when or if any of our product candidates will prove effective or safe in humans or will receive regulatory approval. If our product candidates are associated with undesirable side effects or have characteristics that are unexpected, we may need to abandon their development or limit development to certain uses or subpopulations in which the undesirable side effects or other characteristics are less prevalent, less severe or more acceptable from a risk-benefit perspective. In addition, such effects or characteristics could cause an institutional review board or regulatory authorities to interrupt, delay or halt clinical trials of one or more of our product candidates, require us to conduct additional clinical trials or other tests or studies, and could result in a more restrictive label, or the delay or denial of marketing approval by the FDA or comparable foreign regulatory authorities.
Our Arcelis-based product candidates are immunotherapies that are based on a novel technology utilizing a patient’s own tissue. This may raise development issues that we may not be able to resolve, regulatory issues that could delay or prevent approval or personnel issues that may prevent us from further developing and commercializing our product candidates.
AGS-003 and AGS-004 are based on our novel Arcelis technology platform. In the course of developing this platform and these product candidates, we have encountered difficulties in the development process. For example, we terminated the development of MB-002, the predecessor to AGS-003, when the results from the initial clinical trial of MB-002 indicated that the product candidate only corrected defects in the production of one of two critical cytokines required for effective immune response. There can be no assurance that additional development problems will not arise in the future which we may not be able to resolve or which may cause significant delays in development.
| 42 |
In addition, regulatory approval of novel product candidates such as our Arcelis-based product candidates manufactured using novel manufacturing processes such as ours can be more expensive and take longer than for other, more well-known or extensively studied pharmaceutical or biopharmaceutical products, due to our and regulatory agencies’ lack of experience with them. The FDA has only approved one individualized immunotherapy product to date. This lack of experience may lengthen the regulatory review process, require us to conduct additional studies or clinical trials, increase our development costs, lead to changes in regulatory positions and interpretations, delay or prevent approval and commercialization of these product candidates or lead to significant post-approval limitations or restrictions.
The novel nature of our product candidates also means that fewer people are trained in or experienced with product candidates of this type, which may make it difficult to find, hire and retain capable personnel for research, development and manufacturing positions.
Development of our Arcelis-based product candidates is subject to significant uncertainty because each product candidate is derived from source material that is inherently variable. This variability could reduce the effectiveness of our Arcelis-based product candidates, delay any FDA approval of our Arcelis-based product candidates, cause us to change our manufacturing methods and adversely affect the commercial success of any approved Arcelis-based products.
The disease samples from the patients to be treated with our Arcelis-based products vary from patient to patient. This inherent variability may adversely affect our ability to manufacture our products because each tumor or virus sample that we receive and process will yield a different product. As a result, we may not be able to consistently produce a product for every patient and we may not be able to treat all patients effectively. Such inconsistency could delay FDA or other regulatory approval of our Arcelis-based product candidates or if approved, adversely affect market acceptance and use of our Arcelis-based products. If we have to change our manufacturing methods to address any inconsistency, we may have to perform additional clinical trials, which would delay FDA or other regulatory approval of our Arcelis-based product candidates and increase the costs of development of our Arcelis-based product candidates.
The inherent variability of the disease samples from the patients to be treated with our Arcelis-based products may further adversely affect our ability to manufacture our products because variability in the source material for our product candidates, such as tumor cells or viruses, may cause variability in the composition of other cells in our product candidates. Such variability in composition or purity could adversely affect our ability to establish acceptable release specifications and the development and regulatory approval processes for our product candidates may be delayed, which would increase the costs of development of our Arcelis-based product candidates.
If we are not able to obtain, or if there are delays in obtaining, required regulatory approvals, we will not be able to commercialize our product candidates, and our ability to generate revenue will be materially impaired.
Failure to obtain regulatory approval for either of our product candidates will prevent us from commercializing the product candidate. We have not received regulatory approval to market any of our product candidates in any jurisdiction. We have only limited experience in filing and supporting the applications necessary to gain regulatory approvals and expect to rely on third party contract research organizations to assist us in this process. Securing FDA approval requires the submission of extensive preclinical and clinical data and supporting information to the FDA for each therapeutic indication to establish the product candidate’s safety and efficacy. Securing FDA approval also requires the submission of information about the product manufacturing process to, and inspection of manufacturing facilities by, the FDA. Our product candidates may not be effective, may be only moderately effective or may prove to have undesirable or unintended side effects, toxicities or other characteristics that may preclude our obtaining regulatory approval or prevent or limit commercial use.
The process of obtaining regulatory approvals, both in the United States and abroad, is expensive, may take many years if additional clinical trials are required, if approval is obtained at all, and can vary substantially based upon a variety of factors, including the type, complexity and novelty of the product candidates involved. To date, the FDA has only approved one individualized immunotherapy product. Changes in regulatory approval policies during the development period, changes in or the enactment of additional statutes or regulations, or changes in regulatory review for each submitted product application, may cause delays in the approval or rejection of an application. The FDA has substantial discretion in the approval process and may refuse to accept any application or may decide that our data is insufficient for approval and require additional preclinical, clinical or other studies. In addition, varying interpretations of the data obtained from preclinical and clinical testing could delay, limit or prevent regulatory approval of a product candidate. Any regulatory approval we ultimately obtain may be limited or subject to restrictions or post-approval commitments that render the approved product not commercially viable.
| 43 |
If we experience delays in obtaining approval or if we fail to obtain approval of our product candidates, the commercial prospects for our product candidates may be harmed and our ability to generate revenues will be materially impaired.
Failure to obtain regulatory approval in international jurisdictions would prevent our product candidates from being marketed abroad.
We are a party to arrangements with third parties, and intend to enter into additional arrangements with third parties, under which they would market our products outside the United States. In order to market and sell our products in the European Union and many other jurisdictions, we or such third parties must obtain separate regulatory approvals and comply with numerous and varying regulatory requirements. The approval procedure varies among countries and can involve additional testing. The time required to obtain approval may differ substantially from that required to obtain FDA approval. The regulatory approval process outside the United States generally includes all of the risks associated with obtaining FDA approval. In addition, in many countries outside the United States, it is required that the product be approved for reimbursement before the product can be approved for sale in that country. We or these third parties may not obtain approvals from regulatory authorities outside the United States on a timely basis, if at all. Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one regulatory authority outside the United States does not ensure approval by regulatory authorities in other countries or jurisdictions or by the FDA. We may not be able to file for regulatory approvals and may not receive necessary approvals to commercialize our products in any market.
A fast track designation by the FDA may not actually lead to a faster development, regulatory review or approval process.
If a product is intended for the treatment of a serious or life-threatening condition and the product demonstrates the potential to address unmet needs for this condition, the treatment sponsor may apply for FDA fast track designation. In April 2012, the FDA notified us that we obtained fast track designation for AGS-003 for the treatment of mRCC. Fast track designation does not ensure that we will experience a faster development, regulatory review or approval process compared to conventional FDA procedures. Additionally, the FDA may withdraw fast track designation if it believes that the designation is no longer supported by data from our clinical development program.
Risks Related to Our Financial Position and Need for Additional Capital
We have incurred significant losses since our inception. We expect to incur losses for at least the next several years and may never achieve or maintain profitability.
Since inception, we have incurred significant operating losses. Our net loss was $23.9 million for the year ended December 31, 2013, $53.3 million for the year ended December 31, 2014 and $74.8 million for the year ended December 31, 2015. As of December 31, 2015, we had an accumulated deficit of $279.0 million. To date, we have financed our operations primarily through our initial public offering of common stock, private placements of common stock, preferred stock and warrants, convertible debt financings, debt from financial institutions, government contracts, government and other third party grants and license and collaboration agreements. We have devoted substantially all of our efforts to research and development, including clinical trials. We have not completed development of any product candidates. We expect to continue to incur significant expenses and increasing operating losses for at least the next several years. We anticipate that our expenses will increase substantially if and as we:
| • | continue our ADAPT trial of AGS-003 for the treatment of mRCC; |
| • | continue to support ongoing investigator-initiated clinical trials of AGS-003 and AGS-004; |
| • | support planned investigator-initiated clinical trials of AGS-003 and AGS-004; |
| • | initiate and conduct additional trials of AGS-003 and AGS-004 for the treatment of cancers and HIV; |
| • | seek regulatory approvals for our product candidates that successfully complete clinical trials; |
| • | purchase or lease, and build out and equip, a commercial facility for the manufacture of our Arcelis-based products; |
| • | establish a sales, marketing and distribution infrastructure to commercialize products for which we may obtain regulatory approval; |
| 44 |
| • | maintain, expand and protect our intellectual property portfolio; |
| • | continue our other research and development efforts; |
| • | hire additional clinical, quality control, scientific and management personnel; and |
| • | add operational, financial and management information systems and personnel, including personnel to support our product development and planned commercialization efforts. |
To become and remain profitable, we must develop and eventually commercialize a product or products with significant market potential. This development and commercialization will require us to be successful in a range of challenging activities, including successfully completing preclinical testing and clinical trials of our product candidates, obtaining regulatory approval for these product candidates, building out and equipping a commercial manufacturing facility and manufacturing, marketing and selling those products for which we may obtain regulatory approval. We are only in the preliminary stages of some of these activities. We may never succeed in these activities and may never generate revenues that are significant or large enough to achieve profitability.
Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to become and remain profitable would decrease the value of the company and could impair our ability to raise capital, expand our business, maintain our research and development efforts or continue our operations. A decline in the value of our company could also cause you to lose all or part of your investment.
We will need substantial additional funding. If we are unable to raise capital when needed, we would be forced to delay, reduce, terminate or eliminate our product development programs, including our ADAPT trial of AGS-003, our plans to purchase, build out and equip a commercial manufacturing facility or our commercialization efforts and to take other actions to reduce out operating expenses.
We expect our expenses to increase in connection with our ongoing activities, particularly as we continue our ADAPT trial of AGS-003 for the treatment of mRCC, continue to support ongoing investigator-initiated clinical trials of AGS-003 and AGS-004, support planned investigator-initiated clinical trials of AGS-003 and AGS-004, initiate and conduct additional clinical trials of AGS-003 and AGS-004 for the treatment of cancers and HIV, seek regulatory approval for our product candidates and purchase or lease, and build out and equip, a commercial manufacturing facility. In addition, if we obtain regulatory approval of any of our product candidates, we expect to incur significant commercialization expenses for product sales, marketing, manufacturing and distribution. Furthermore, we expect to incur additional costs associated with operating as a public company. Accordingly, we will need to obtain substantial additional funding in connection with our continuing operations. If we are unable to raise capital when needed or on attractive terms, we would be forced to delay, reduce, terminate or eliminate our product development programs, our plans to purchase or lease, and build out and equip a commercial manufacturing facility or our commercialization efforts.
As of December 31, 2015, we had cash, cash equivalents and short-term investments of $7.2 million and working capital of $0.7 million. Based on our current operating plan, we expect that the $19.0 million of net proceeds from the sale of common stock and warrants at the initial closing of our March 2016 private placement, together with our existing cash, cash equivalents and short-term investments as of December 31, 2015 and the anticipated funding under our contract with the National Institutes of Health, or NIH, and the National Institute of Allergy and Infectious Diseases, or NIAID, will enable us to fund our operating expenses into the third quarter of 2016.
Our March 2016 private placement includes three tranches. The initial tranche closed in March 2016, and the second and third tranches are conditioned on the achievement of certain regulatory milestones and other closing conditions. For a description of the terms of the private placement, see “Management’s Discussion and Analysis—Liquidity and Capital Resources—Sources of Liquidity—PIPE Financing”. If the milestones and closing conditions are achieved and the second and third tranches are consummated, we expect that the estimated $59.0 million of net proceeds from all three tranches of our private placement, together with our existing cash, cash equivalents and short-term investments as of December 31, 2015 and the anticipated funding under our contract with the NIH and NIAID, will enable us to fund our operating expenses into the second quarter of 2017, when we expect final data from the ADAPT trial. We expect that these funds will be sufficient to enable us to support the ongoing and planned investigator-initiated clinical trials of AGS-003, certain of the manufacturing costs of the ongoing Phase 2 clinical trial of AGS-004 for HIV eradication in patients with early stage RCC and certain costs related to commercial manufacturing. We expect that these funds will not, however, be sufficient to enable us to complete required activities in preparation for the submission of a BLA to the FDA for AGS-003, to perform pre-commercialization activities for AGS-003 or to commercially launch AGS-003. These expectations are based on our current operating plan under which we plan to implement measures to reduce our operating expenses, including reductions in spending for activities in preparation for submission of a BLA. These reductions may include reductions in planned spending related to chemistry, manufacturing and controls, process validation, quality, supply management and commercial preparation. Under our plan, we also intend to seek to refinance our existing venture loan facility with Horizon Technology Finance Corporation, or Horizon, and Fortress Credit Co LLC, or Fortress, and to enter into collaborations for the development, marketing and distribution of AGS-003 outside North America and of our non-oncology product candidates, including AGS-004. If we are unable to refinance our venture loan facility or enter into any such collaborations, we may be required to implement additional measures to materially reduce our operating expenses, which could adversely affect our business and operations. It is also possible that our available funds will not enable us to obtain final data from the ADAPT trial because the actual costs and timing of clinical trials are difficult to predict and are subject to substantial risks and delays. We have no external sources of funds other than our contract with the NIH and NIAID for the development of AGS-004.
| 45 |
We also will need to obtain significant financing if we decide to purchase, build out and equip a new commercial manufacturing facility. Our preliminary estimate indicates that we will require approximately an additional $30.0 million prior to the commercialization of AGS-003 to purchase, build out and equip the commercial manufacturing facility. These costs do not include the costs to implement the automated manufacturing process. We are actively exploring financing arrangements in connection with the planned purchase, build out and equipping of the commercial manufacturing facility and are in discussions with developers, lenders and other potential financing sources regarding potential financial support. Unless we select an alternative plan for the commercial supply of our products, we expect to enter into such arrangements during the second quarter of 2016 and that such arrangements will likely involve material obligations and debt liabilities. We are also exploring alternatives for the commercial supply of our products, including building out and equipping our current facility for commercial manufacture, leasing and equipping an existing manufacturing space that would be built out for commercial manufacture, and engaging a contract manufacturer to supply AGS-003 on a commercial scale on our behalf. We will also need to obtain additional financing with any of these alternatives. If we are unable to obtain additional financing when needed, in the required amounts or at all, we may not be able to complete the planned purchase, build-out and equipping of the new commercial facility or be able to lease and equip an existing commercial facility, or may be delayed in doing so.
Our future capital requirements will depend on many factors, including:
| • | the progress and results of our ADAPT trial and the ongoing and planned investigator-initiated clinical trials of AGS-003 that we support or plan to support; |
| • | the progress and results of the ongoing investigator-initiated Phase 2 clinical trial of AGS-004 for HIV eradication that we support and the planned investigator-initiated clinical trial of AGS-004 that we plan to support and our ability to obtain additional funding under our NIH and NIAID contract for our AGS-004 program; |
| • | the scope, progress, results and costs of preclinical development, laboratory testing and clinical trials for our other product candidates; |
| • | the purchase price for, and the costs and timing of our planned purchase, build-out and equipping of a new commercial manufacturing facility, the costs and timing of building out and equipping a new commercial manufacturing facility at our current facility or at a new leased facility, or, the costs and timing of entering into a commercial supply arrangement with a contract manufacturer, and any costs and liabilities associated with any financing arrangements entered into to fund the costs of these activities; |
| • | the costs, timing and outcome of regulatory review of our product candidates; |
| • | the costs of commercialization activities, including product sales, marketing, manufacturing and distribution, for any of our product candidates for which we receive regulatory approval; |
| • | the potential need to repay the $8.0 million remaining outstanding under the loan under our license agreement with Medinet Co. Ltd. and its wholly-owned subsidiary, MEDcell Co., Ltd, which we refer to together as Medinet; |
| • | revenue, if any, received from commercial sales of our product candidates, should any of our product candidates be approved by the FDA or a similar regulatory authority outside the United States; |
| • | the costs of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending intellectual property-related claims; |
| • | the extent to which we acquire or invest in other businesses, products and technologies; |
| 46 |
| • | our ability to obtain government or other third party funding for the development of our product candidates; and |
| • | our ability to establish collaborations on favorable terms, if at all, particularly arrangements to develop, market and distribute AGS-003 outside North America and arrangements for the development and commercialization of our non-oncology product candidates, including AGS-004. |
Conducting preclinical testing and clinical trials is a time-consuming, expensive and uncertain process that takes years to complete, and we may never generate the necessary data or results required to obtain regulatory approval and achieve product sales. In addition, our product candidates, if approved, may not achieve commercial success. Our commercial revenues, if any, will be derived from sales of products that we do not expect to be commercially available for several years, if at all. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. Additional financing may not be available to us on acceptable terms, or at all.
Our independent registered public accounting firm has included an explanatory paragraph relating to our ability to continue as a going concern in its report on our audited financial statements included in this Annual Report on Form 10-K.
Our report from our independent registered public accounting firm for the year ended December 31, 2015 includes an explanatory paragraph stating that our losses from operations and required additional funding to finance our operations raise substantial doubt about our ability to continue as a going concern. If we are unable to obtain sufficient funding, our business, prospects, financial condition and results of operations will be materially and adversely affected and we may be unable to continue as a going concern. If we are unable to continue as a going concern, we may have to liquidate our assets and may receive less than the value at which those assets are carried on our audited financial statements, and it is likely that investors will lose all or a part of their investment. If we seek additional financing to fund our business activities in the future and there remains substantial doubt about our ability to continue as a going concern, investors or other financing sources may be unwilling to provide additional funding to us on commercially reasonable terms or at all.
Raising additional capital may cause dilution to our existing stockholders, restrict our operations or require us to relinquish rights to our technologies or product candidates.
Until such time, if ever, as we can generate substantial product revenues, we expect to finance our cash needs through a combination of equity offerings, debt financings, government contracts, government and other third party grants or other third party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. We will require substantial funding to complete the planned purchase, build-out and equipping of a new commercial manufacturing facility or to build out and equip a leased commercial manufacturing facility, complete required activities in preparation for the submission of a BLA for AGS-003, fund our commercialization efforts and fund our other operating expenses and other activities. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a stockholder. For example, in March 2016, we issued and sold in a private placement an aggregate of 3,652,430 shares of our common stock, plus warrants to purchase a total of 2,739,323 additional shares of common stock, which resulted in dilution to our existing stockholders. If we complete the second and third tranches of this private placement, this will result in further dilution to the ownership interests of our existing stockholders.
Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
We currently intend to collaborate with third parties for the manufacturing, development or commercialization of AGS-003 outside of North America. We plan to seek government or other third party funding for the continued development of AGS-004 and to collaborate with third parties for the development and commercialization of AGS-004. If we raise additional funds through government or other third party funding, marketing and distribution arrangements or other collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to us. If the loan from Medinet becomes due and we do not repay it, we have agreed to grant Medinet a non-exclusive, royalty-bearing license to make and sell Arcelis products in Japan for the treatment of cancer.
Our ability to use our net operating loss carry-forwards and tax credit carryforwards may be limited.
The utilization of the net operating loss and tax credit carryforwards may be subject to limitation under the rules regarding a change in stock ownership as determined by the Internal Revenue Code, and state and foreign tax laws. Section 382 of the Internal Revenue Code of 1986, as amended, imposes annual limitations on the utilization of net operating loss carryforwards, other tax carryforwards, and certain built-in losses upon an ownership change as defined under that section. In general terms, an ownership change may result from transactions that increase the aggregate ownership of certain of our stockholders by more than 50 percentage points over a three year testing period. If we have undergone a Section 382 ownership change, an annual limitation would be imposed on certain of our tax attributes, including NOL and capital loss carryforwards, and certain other losses, credits, deductions or tax basis. As of December 31, 2015, we have not completed a formal study to determine whether there are 382 limitations that apply However, we believe that, by engaging in the financing transactions in which we have engaged, we may have triggered an “ownership change” limitation. We may also experience ownership changes in the future as a result of subsequent shifts in our stock ownership. As a result, if we earn net taxable income, our ability to use our pre-change net operating loss carry-forwards and other tax credit carryforwards to offset U.S. federal taxable income may be subject to limitations, which potentially could result in increased future tax liability to us.
| 47 |
Risk Related to the Commercialization of our Product Candidates
We have no history of commercializing pharmaceutical products, which may make it difficult to evaluate the prospects for our future viability.
Our operations to date have been limited to financing and staffing our company, developing our technology and product candidates and establishing collaborations. We have not yet demonstrated an ability to successfully complete a pivotal clinical trial, obtain marketing approvals, manufacture a commercial scale product or conduct sales and marketing activities necessary for successful product commercialization. Consequently, predictions about our future success or viability may not be as accurate as they could be if we had a history of successfully developing and commercializing pharmaceutical products.
In addition, we may encounter unforeseen expenses, difficulties, complications, delays and other known and unknown factors. We will need to transition at some point from a company with research and development focus to a company capable of supporting commercial activities. We may not be successful in such a transition.
Even if AGS-003 or AGS-004 receives regulatory approval, it may fail to achieve the degree of market acceptance by physicians, patients, healthcare payors and others in the medical community necessary for commercial success.
We have never commercialized a product candidate. Even if AGS-003 or AGS-004 receives marketing approval, it may nonetheless fail to gain sufficient market acceptance by physicians, patients, healthcare payors and others in the medical community. Gaining market acceptance for our Arcelis-based products may be particularly difficult as, to date, the FDA has only approved one individualized immunotherapy and our Arcelis-based products are based on a novel technology. If these products do not achieve an adequate level of acceptance, we may not generate significant product revenues and we may not become profitable. The degree of market acceptance of our product candidates, if approved for commercial sale, will depend on a number of factors, including:
| • | efficacy and potential advantages compared to alternative treatments; |
| • | the ability to offer our product candidates for sale at competitive prices; |
| • | convenience and ease of administration compared to alternative treatments; |
| • | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| • | the strength of sales, marketing and distribution support; |
| • | the approval of other new products for the same indications; |
| • | the ability of our product to be combined with emerging standards of care; |
| • | availability and amount of reimbursement from government payors, managed care plans and other third party payors; |
| • | adverse publicity about the product or favorable publicity about competitive products; |
| • | clinical indications for which the product is approved; and |
| • | the prevalence and severity of any side effects. |
If any of our product candidates receives marketing approval and we, or others, later discover that the product is less effective than previously believed or causes undesirable side effects that were not previously identified, our ability to market the product could be compromised.
Clinical trials of our product candidates are conducted in carefully defined subsets of patients who have agreed to enter into clinical trials. Consequently, it is possible that our clinical trials may indicate an apparent positive effect of a product candidate that is greater than the actual positive effect, if any, in a broader patient population or alternatively fail to identify undesirable side effects. If, following approval of a product candidate, we, or others, discover that the product is less effective than previously believed or causes undesirable side effects that were not previously identified, any of the following adverse events could occur:
| 48 |
| • | regulatory authorities may withdraw their approval of the product or seize the product; |
| • | we may be required to recall the product or change the way the product is administered; |
| • | additional restrictions may be imposed on the marketing of, or the manufacturing processes for, the particular product; |
| • | regulatory authorities may require the addition of labeling statements, such as a “black box” warning or a contraindication; |
| • | we may be required to create a Medication Guide outlining the risks of the previously unidentified side effects for distribution to patients; |
| • | additional restrictions may be imposed on the distribution or use of the product via a risk evaluation and mitigation strategy, or REMS; |
| • | we could be sued and held liable for harm caused to patients; |
| • | the product may become less competitive; and |
| • | our reputation may suffer. |
Any of these events could have a material and adverse effect on our operations and business and could adversely impact our stock price.
If we are unable to establish sales and marketing capabilities or enter into agreements with third parties to sell and market our product candidates, we may not be successful in commercializing our product candidates if and when they are approved.
We do not have a sales or marketing infrastructure and have no experience in the sale, marketing or distribution of pharmaceutical products. To achieve commercial success for any approved product, we must either develop a sales and marketing organization or outsource these functions to third parties. We plan to market and sell AGS-003 in North America independently and to enter into collaborations or other arrangements with third parties for the distribution or marketing of AGS-003 in the rest of the world. We plan to enter into collaborations or other arrangements with third parties for the distribution or marketing of AGS-004 and any of our other product candidates should such candidates receive marketing approval.
There are risks involved with both establishing our own sales and marketing capabilities and entering into arrangements with third parties to perform these services. For example, recruiting and training a sales force is expensive and time consuming and could delay any product launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed or does not occur for any reason, we would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
Factors that may inhibit our efforts to commercialize our products on our own include:
| • | our inability to recruit and retain adequate numbers of effective sales and marketing personnel; |
| • | the inability of sales personnel to obtain access to or persuade adequate numbers of physicians to prescribe any future products; |
| • | the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and |
| • | unforeseen costs and expenses associated with creating an independent sales and marketing organization. |
| 49 |
If we enter into arrangements with third parties to perform sales, marketing and distribution services, our product revenues or the profitability of these product revenues to us are likely to be lower than if we were to market and sell any products that we develop ourselves. In addition, we may not be successful in entering into arrangements with third parties to sell and market our product candidates or doing so on terms that are favorable to us. We likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market our products effectively. If we do not establish sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
We face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than we do.
The development and commercialization of new drug products is highly competitive. We face competition with respect to our current product candidates, and will face competition with respect to any products that we may seek to develop or commercialize in the future, from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. There are a number of large pharmaceutical and biotechnology companies that currently market and sell products or are pursuing the development of products for the treatment of the disease indications for which we are developing our product candidates. Potential competitors also include academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
Some of these competitive products and therapies are based on scientific approaches that are the same as or similar to our approach, and others are based on entirely different approaches. Many marketed therapies for the indications that we are currently pursuing, or indications that we may in the future seek to address using our Arcelis platform, are widely accepted by physicians, patients and payors, which may make it difficult for us to replace them with any products that we successfully develop and are permitted to market.
The FDA has approved several targeted therapies as monotherapies for mRCC, including Nexavar (sorafenib), marketed by Bayer Healthcare Pharmaceuticals, Inc. and Onyx Pharmaceuticals, Inc.; Sutent (sunitinib) and Inlyta (axitinib), marketed by Pfizer, Inc.; Avastin (bevacizumab), marketed by Genentech, Inc., a member of the Roche Group; Votrient (pazopanib) and Afinitor (everolimus), marketed by Novartis Pharmaceuticals Corporation; Torisel (temsirolimus), marketed by Pfizer and most recently, Opdivo (nivolumab) for second-line RCC, marketed by Bristol-Myers Squibb. In addition, we estimate that there are numerous therapies for mRCC in clinical development by many public and private biotechnology and pharmaceutical companies targeting numerous different cancer types and stages. A number of these are in late stage development. Opdivo (nivolumab) plus Yervoy (ipilimumab) in combination for first-line mRCC is being developed by Bristol-Myers Squibb and is currently being compared in a Phase 3 trial to sunitinib. Cometriq (cabozantinib) is being developed by Exelixis has recently reported positive results from its Phase 3 trial and is expected to receive FDA approval for second-line mRCC in 2016. In addition, if a standalone therapy for mRCC were developed that demonstrated improved efficacy over currently marketed first-line therapies with a favorable safety profile and without the need for combination therapy, such a therapy might pose a significant competitive threat to AGS-003.
We are currently conducting our ADAPT trial of AGS-003 plus sunitinib / targeted therapy. We elected to study AGS-003 in clinical trials in combination with sunitinib due in part to sunitinib being the current standard of care for first-line treatment of mRCC. Although we do not expect to seek FDA approval of AGS-003 solely in combination with sunitinib and have provided that, under the protocol for the ADAPT trial, investigators may discontinue sunitinib due to disease progression or toxicity and initiate second-line treatment with other approved compatible therapies, if we obtain approval by the FDA, such FDA approval may be limited to the combination of AGS-003 and sunitinib. In such event, the commercial success of AGS-003 would be linked to the commercial success of sunitinib. As a result, if sunitinib ceases to be the standard of care for first-line treatment of mRCC or another event occurs that adversely affects sales of sunitinib, the commercial success of AGS-003 may be adversely affected.
There are also numerous FDA-approved treatments for HIV, primarily antiretroviral therapies marketed by large pharmaceutical companies. Generic competition has developed in this market as patent exclusivity periods for older drugs have expired, with more than 15 generic drugs currently on the market. The presence of these generic drugs is resulting in price pressure in the HIV therapeutics market and could affect the pricing of AGS-004. Currently, there are no approved therapies for the eradication of HIV. We expect that major pharmaceutical companies that currently market antiretroviral therapy products or other companies that are developing HIV product candidates may seek to develop products for the eradication of HIV.
Our competitors may develop products that are more effective, safer, more convenient or less costly than any that we are developing or that would render our product candidates obsolete or non-competitive. Our competitors may also obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours.
| 50 |
Many of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Mergers and acquisitions in the pharmaceutical, biotechnology and device industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Even if we are able to commercialize any product candidates, the products may become subject to unfavorable pricing regulations, third party reimbursement practices or healthcare reform initiatives, which would harm our business.
The regulations that govern marketing approvals, pricing and reimbursement for new drug products vary widely from country to country. In the United States, recently passed legislation may significantly change the approval requirements in ways that could involve additional costs and cause delays in obtaining approvals. Some countries require approval of the sale price of a drug before it can be marketed. In many countries, the pricing review period begins after marketing or product licensing approval is granted. In some foreign markets, prescription pharmaceutical pricing remains subject to continuing governmental control even after initial approval is granted. As a result, we might obtain regulatory approval for a product in a particular country, but then be subject to price regulations that delay our commercial launch of the product, possibly for lengthy time periods, and negatively impact the revenues we are able to generate from the sale of the product in that country. Adverse pricing limitations may hinder our ability to recoup our investment in one or more product candidates, even if our product candidates obtain regulatory approval.
Our ability to commercialize any products successfully also will depend in part on the extent to which reimbursement for these products and related treatments will be available from government health administration authorities, private health insurers and other organizations. Government authorities and third party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels. A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and third party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications. Increasingly, third party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. We cannot be sure that reimbursement will be available for any product that we commercialize and, if reimbursement is available, the level of reimbursement. Reimbursement may impact the demand for, or the price of, any product candidate for which we obtain marketing approval. Obtaining reimbursement for our products may be particularly difficult because of the higher prices often associated with drugs administered under the supervision of a physician. If reimbursement is not available or is available only to limited levels, we may not be able to successfully commercialize any product candidate for which we obtain marketing approval.
There may be significant delays in obtaining reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which the drug is approved by the FDA or similar regulatory authorities outside the United States. Moreover, eligibility for reimbursement does not imply that any drug will be paid for in all cases or at a rate that covers our costs, including research, development, manufacture, sale and distribution. Interim reimbursement levels for new drugs, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement rates may vary according to the use of the drug and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost drugs, and may be incorporated into existing payments for other services. Net prices for drugs may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than in the United States. Third party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement policies. Our inability to promptly obtain coverage and profitable payment rates from both government-funded and private payors for any approved products that we develop could have a material adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
Product liability lawsuits against us could cause us to incur substantial liabilities and to limit commercialization of any products that we may develop.
We face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials and will face an even greater risk if we commercially sell any products that we may develop. These risks may be even greater with respect to our Arcelis-based products which are manufactured using a novel technology. None of our product candidates has been widely used over an extended period of time, and therefore our safety data are limited. We derive the raw materials for manufacturing of our Arcelis-based product candidates from human cell sources, and therefore the manufacturing process and handling requirements are extensive and stringent, which increases the risk of quality failures and subsequent product liability claims.
If we cannot successfully defend ourselves against claims that our product candidates or products caused injuries, we will incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
| • | decreased demand for any product candidates or products that we may develop; |
| 51 |
| • | injury to our reputation and significant negative media attention; |
| • | withdrawal of clinical trial participants; |
| • | significant costs to defend the related litigation; |
| • | substantial monetary awards to trial participants or patients; |
| • | loss of revenue; and |
| • | the inability to commercialize any products that we may develop. |
We currently hold $10.0 million in product liability insurance coverage, which may not be adequate to cover all liabilities that we may incur. We will need to increase our insurance coverage when we begin commercializing our product candidates, if ever. Insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise.
We may expend our limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success.
Because we have limited financial and managerial resources, we focus on research programs and product candidates for specific indications. As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on current and future research and development programs and product candidates for specific indications may not yield any commercially viable products.
We have based our research and development efforts on our Arcelis platform. Notwithstanding our large investment to date and anticipated future expenditures in our Arcelis platform, we have not yet developed, and may never successfully develop, any marketed drugs using this approach. As a result of pursuing the development of product candidates using our Arcelis platform, we may fail to develop product candidates or address indications based on other scientific approaches that may offer greater commercial potential or for which there is a greater likelihood of success.
Our long-term business plan is to develop Arcelis-based products for the treatment of various cancers and infectious diseases. We may not be successful in our efforts to identify or discover additional product candidates that may be manufactured using our Arcelis platform. Research programs to identify new product candidates require substantial technical, financial and human resources. These research programs may initially show promise in identifying potential product candidates, yet fail to yield product candidates for clinical development.
If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product candidate.
Risks Related to Our Dependence on Third Parties
Our reliance on government funding adds uncertainty to our research and commercialization efforts and may impose requirements that increase the costs of commercialization and production of our government-funded product candidates.
Our current development of AGS-004 is primarily funded by the NIH. We are dependent upon further government funding for continued development of AGS-004. However, increased pressure on governmental budgets may reduce the availability of government funding for programs such as AGS-004. In addition, contracts and grants from the U.S. government and its agencies include provisions that give the government substantial rights and remedies, many of which are not typically found in commercial contracts, including provisions that allow the government to:
| • | terminate agreements, in whole or in part, for any reason or no reason; |
| • | reduce or modify the government’s obligations under such agreements without the consent of the other party; |
| 52 |
| • | claim rights, including intellectual property rights, in products and data developed under such agreements; |
| • | impose U.S. manufacturing requirements for products that embody inventions conceived or first reduced to practice under such agreements; |
| • | suspend or debar the contractor or grantee from doing future business with the government or a specific government agency; |
| • | pursue criminal or civil remedies under the False Claims Act, False Statements Act and similar remedy provisions specific to government agreements; and |
| • | limit the government’s financial liability to amounts appropriated by the U.S. Congress on a fiscal-year basis, thereby leaving some uncertainty about the future availability of funding for a program even after it has been funded for an initial period. |
Government agreements normally contain additional terms and conditions that may increase our costs of doing business, reduce our profits, and expose us to liability for failure to comply with these terms and conditions. These include, for example:
| • | specialized accounting systems unique to government contracts and grants; |
| • | mandatory financial audits and potential liability for price adjustments or recoupment of government funds after such funds have been spent; |
| • | public disclosures of certain contract and grant information, which may enable competitors to gain insights into our research program; and |
| • | mandatory socioeconomic compliance requirements, including labor standards, non-discrimination and affirmative action programs and environmental compliance requirements. |
We expect to depend on collaborations with third parties for the development and commercialization of our product candidates. If those collaborations are not successful, we may not be able to capitalize on the market potential of these product candidates.
We currently intend to commercialize AGS-003 independently in North America. We intend to collaborate with other third parties to manufacture, develop or commercialize AGS-003 outside North America. We have entered into an exclusive license agreement with Pharmstandard International S.A., or Pharmstandard, for the development and commercialization of AGS-003 in Russia and the other states comprising the Commonwealth of Independent States and an exclusive license agreement with Green Cross Corp., or Green Cross, for the development and commercialization of AGS-003 for the treatment of mRCC in South Korea and an exclusive license agreement with Lummy (Hong Kong) Co. Ltd., or Lummy HK, for the development, manufacture and commercialization of AGS-003 in China, Hong Kong, Taiwan and Macau. We have also entered into a license agreement with Medinet under which we granted Medinet an exclusive license to manufacture in Japan AGS-003 for the purpose of development and commercialization for the treatment of mRCC and an option to acquire a non-exclusive license to sell in Japan AGS-003 for the treatment of mRCC. We also plan to seek government or other third party funding for continued development of AGS-004 and to collaborate with third parties to develop and commercialize AGS-004. Our likely collaborators for any development, distribution, marketing, licensing or broader collaboration arrangements include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies and biotechnology companies.
Under our existing arrangements we have limited control, and under any additional arrangements we may enter into with third parties we will likely have limited control, over the amount and timing of resources that our collaborators dedicate to the development or commercialization of our product candidates. Our ability to generate revenues from these arrangements will depend on our collaborators’ abilities to successfully perform the functions assigned to them in these arrangements.
Collaborations involving our product candidates would pose the following risks to us:
| • | collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| • | collaborators may not pursue development and commercialization of our product candidates or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator’s strategic focus or available funding, or external factors such as an acquisition that diverts resources or creates competing priorities; |
| • | collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or, require a new formulation of a product candidate for clinical testing; |
| 53 |
| • | collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our products or product candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
| • | a collaborator with marketing and distribution rights to one or more products may not commit sufficient resources to the marketing and distribution of such product or products; |
| • | collaborators may have the right to conduct clinical trials of our product candidates without our consent and could conduct trials with flawed designs that result in data that adversely affect our clinical trials, our ability to obtain marketing approval for our product candidates or market acceptance of our product candidates. Pharmstandard, Green Cross, Medinet and Lummy HK each have this right under our license agreements with them; |
| • | collaborators may hold rights that could preclude us from commercializing our products in certain territories. For example, we have granted Medinet an exclusive license to manufacture in Japan AGS-003 for the treatment of mRCC and an option to acquire a non-exclusive license to sell in Japan AGS-003 for the treatment of mRCC. Even if, as expected, Medinet does not exercise the option to acquire the license to sell, we will not have the right to manufacture AGS-003 in Japan for the purposes of development and commercialization of AGS-003 for the treatment of mRCC. If we and Medinet are unable to agree to the terms of a supply agreement under these circumstances, we will not be able to sell AGS-003 in Japan unless we repurchase these rights from Medinet; |
| • | collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our proprietary information or expose us to potential litigation; |
| • | disputes may arise between the collaborators and us that result in the delay or termination of the research, development or commercialization of our products or product candidates or that result in costly litigation or arbitration that diverts management attention and resources; and |
| • | collaborations may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization of the applicable product candidates. For example, our collaboration with Kyowa Hakko Kirin Co., Ltd. with respect to AGS-003 and AGS-004 was terminated by our collaborator. |
Collaboration agreements may not lead to development or commercialization of product candidates in the most efficient manner, or at all. In addition, there have been a significant number of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators. If a present or future collaborator of ours were to be involved in a business combination, the continued pursuit and emphasis on our product development or commercialization program could be delayed, diminished or terminated.
If we are not able to establish additional collaborations, we may have to alter our development and commercialization plans.
Our drug development programs and the potential commercialization of our product candidates will require substantial additional cash to fund expenses. For some of our product candidates, we intend to collaborate with pharmaceutical and biotechnology companies for the development and commercialization of those product candidates. For example, we have entered into license agreements with third parties to develop, manufacture and/or commercialize AGS-003 in Russia and the other states comprising the Commonwealth of Independent States, South Korea, Japan, China, Hong Kong, Taiwan and Macau, and we intend to collaborate with other third parties to develop and commercialize AGS-003 in other parts of the world and to collaborate with third parties to develop and commercialize AGS-004.
We face significant competition in seeking appropriate collaborators. Whether we reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed collaboration, and the proposed collaborator’s evaluation of a number of factors. Those factors may include the design or results of clinical trials, the likelihood of approval by the FDA or similar regulatory authorities outside the United States, the potential market for the subject product candidate, the costs and complexities of manufacturing and delivering such product candidate to patients, the potential of competing products, the existence of uncertainty with respect to our ownership of technology, which can exist if there is a challenge to such ownership without regard to the merits of the challenge and industry and market conditions generally. The collaborator may also consider alternative product candidates or technologies for similar indications that may be available to collaborate on and whether such a collaboration could be more attractive than the one with us for our product candidate. We may also be restricted under existing license agreements from entering into agreements on certain terms with potential collaborators. Collaborations are complex and time-consuming to negotiate and document. We may not be able to negotiate collaborations on a timely basis, on acceptable terms, or at all.
| 54 |
If we are not able to obtain such funding or enter into collaborations for our product candidate, we may have to curtail the development of such product candidate, reduce or delay its development program or one or more of our other development programs, delay its potential commercialization or reduce the scope of any sales or marketing activities, or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development or commercialization activities on our own, we may need to obtain additional capital, which may not be available to us on acceptable terms or at all. If we do not have sufficient funds, we may not be able to further develop these product candidates or bring these product candidates to market and generate product revenue.
We rely on third parties to conduct our clinical trials, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such trials.
We do not independently conduct clinical trials of our product candidates. We rely on third parties, such as contract research organizations, clinical data management organizations, medical institutions and clinical investigators, to perform this function. Our reliance on these third parties for clinical development activities reduces our control over these activities but does not relieve us of our responsibilities. We remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols for the trial. Moreover, the FDA and other regulatory authorities require us to comply with standards, commonly referred to as Good Clinical Practice, for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. We also are required to register ongoing clinical trials and post the results of certain completed clinical trials on a government-sponsored database, ClinicalTrials.gov, within certain timeframes. Failure to do so can result in fines, adverse publicity and civil and criminal sanctions. Furthermore, these third parties may also have relationships with other entities, some of which may be our competitors. If these third parties do not successfully carry out their contractual duties, meet expected deadlines or conduct our clinical trials in accordance with regulatory requirements or our stated protocols, we will not be able to obtain, or may be delayed in obtaining, regulatory approvals for our product candidates and will not be able to, or may be delayed in our efforts to, successfully commercialize our product candidates.
For instance, we recently received a notice from Health Canada that one of the sites at which we are conducting our Phase 3 ADAPT trial in Canada had been found to be non-compliant with Good Clinical Practice in Canada and that if the issues raised in the notice were not corrected, Health Canada could suspend our authorization to conduct the ADAPT trial at all sites in Canada. We have submitted a response to Health Canada, but if we are unable to satisfy Health Canada that the site is in compliance, the Phase 3 ADAPT trial could be suspended in Canada which would adversely affect our ability to complete the ADAPT trial on the anticipated timeline.
We also rely on other third parties to store and distribute product supplies for our clinical trials. Any performance failure on the part of our existing or future distributors could delay clinical development or regulatory approval of our product candidates or commercialization of our products, producing additional losses and depriving us of potential product revenue.
Risks Related to the Manufacturing of Our Product Candidates
We have commenced the build-out of a new facility to manufacture our Arcelis-based products on a commercial scale. We do not have experience in manufacturing Arcelis-based products on a commercial scale. If, due to our lack of manufacturing experience, we cannot manufacture our Arcelis-based products on a commercial scale successfully or manufacture sufficient product to meet our expected commercial requirements, our business may be materially harmed.
We currently manufacture our Arcelis-based product candidates for research and development purposes and for clinical trials of our product candidates at our facility in Durham, North Carolina. In August 2014, in connection with our plan to build out and equip a commercial manufacturing facility, we entered into a lease agreement with the developer, TKC LXXII, LLC, or TKC. Under the lease agreement, we agreed to lease certain land and an approximately 124,000 square-foot building to be constructed in Durham County, North Carolina. This facility would house our corporate headquarters and commercial manufacturing. The shell of the new facility was constructed on a build-to-suit basis in accordance with agreed upon specifications and plans and was completed in June 2015. A portion of the build-out and equipping of the interior of the facility began in the second half of 2015.
| 55 |
Under the lease agreement, we had an option to purchase the property. In February 2015, we exercised this purchase option and entered into a Purchase and Sale Agreement with TKC. The purchase price to be paid by us is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by us, for which we have paid $1.7 million as of December 31, 2015, and the amount of any improvement allowances advanced to us by TKC prior to the closing. Under the terms of the Purchase and Sale Agreement, we had until February 16, 2016 to consummate the purchase of the property. We are currently in discussions with TKC to extend the period for purchasing the property as we will need to obtain additional funding to consummate the purchase of the property. If we purchase the property, upon the closing, the lease agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to us.
Due to our limited resources and the costs and challenges of building and equipping a new commercial facility, we are exploring alternative plans for the commercial supply of our products, including building out and equipping our current facility for commercial manufacture, leasing and equipping an existing manufacturing space that would have to be built out for commercial manufacture, and engaging a contract manufacturer to supply AGS-003 on a commercial scale on our behalf. We believe that building out and equipping our current facility would only be a temporary solution from a capacity standpoint and would only cover commercial manufacturing for a limited number of years.
We plan to initiate commercial manufacturing using our current manual manufacturing process and have decided to complete the development and implementation of our automated manufacturing process after commercial launch. Prior to implementing commercial manufacturing of AGS-003, we would be required to demonstrate that the commercial manufacturing facility is constructed and operated in accordance with current good manufacturing practice. We would also be required to show the comparability between AGS-003 that we produce using the manual processes in our current facility and AGS-003 produced using the manual process in the commercial manufacturing facility.
If we transition to automated manufacturing processes, we expect our automated manufacturing processes will be based on existing functioning prototypes of automated devices for the production of commercial quantities of our Arcelis-based product candidates. These devices can be used to perform substantially all steps required for the manufacture of our Arcelis-based product candidates.
We do not have experience in manufacturing products on a commercial scale or using automated processes. In addition, because we are aware of only one company that has manufactured an individualized immunotherapy product for commercial sale, there are limited precedents from which we can learn. We may encounter difficulties in the manufacture of our Arcelis-based products due to our limited manufacturing experience. These difficulties could delay the build-out and equipping of a commercial manufacturing facility and regulatory approval of the manufacture of our Arcelis-based products using the facility, increase our costs or cause production delays or result in us not manufacturing sufficient product to meet our expected commercial requirements, any of which could damage our reputation and hurt our profitability. If we are unable to successfully increase our manufacturing capacity to commercial scale, our business may be materially adversely affected.
We need to establish commercial manufacturing operations and augment our manufacturing personnel in advance of any regulatory submission for approval of AGS-003. If we fail to establish commercial manufacturing operations in compliance with regulatory requirements, or augment our manufacturing personnel, we may not be able to initiate commercial operations or produce sufficient product to meet our expected commercial requirements. We have delayed the implementation of our automated manufacturing process and may not be able to use such process on a timely basis or at all.
In order to meet our business plan, which contemplates manufacturing our product first using manual processes and later using automated processes for the commercial requirements of AGS-003 and any other Arcelis-based product candidates that might be approved, we plan to build out and equip a commercial manufacturing facility and add manufacturing personnel in advance of any regulatory submission for approval of AGS-003. The planned purchase, build-out and equipping of a commercial manufacturing facility or, alternatively, the build out and equipping of a leased facility, will require substantial capital expenditures and additional regulatory approvals. In addition, it will be costly and time consuming to recruit necessary additional personnel.
If we are unable to successfully build out and equip a commercial manufacturing facility in compliance with regulatory requirements or hire and train additional necessary manufacturing personnel appropriately, our filing for regulatory approval of AGS-003 may be delayed or denied.
We plan to delay the implementation of our automated manufacturing process until we obtain additional funding, and thus plan to seek approval of AGS-003 and, if approved, to initially commercially supply AGS-003 using manual manufacturing processes. Prior to implementing commercial manufacturing of AGS-003, we will be required to demonstrate that the commercial manufacturing facility is constructed and operated in accordance with current Good Manufacturing Practice, or cGMP. We will also be required to show the comparability between AGS-003 that we produce using the manual processes in our current facility and AGS-003 produced using the manual process in the new facility.
| 56 |
Our implementation of automated processes could take longer, particularly if we are unable to achieve any of the required tasks on a timely basis, or at all. We are collaborating with Invetech Pty Ltd., or Invetech, and Saint-Gobain Performance Plastics Corporation, or Saint-Gobain, to develop the equipment and disposables necessary to implement the automated manufacturing processes for Arcelis-based products. If Invetech or Saint-Gobain do not perform as expected under the agreements or the projects with Invetech or Saint-Gobain are unsuccessful for any other reason, our timelines for the implementation of our automated manufacturing processes could be further delayed and our business could be adversely affected.
Prior to implementing the automated manufacturing processes for Arcelis-based products, we will be required to:
| • |
demonstrate that the disposable components and sterilization and packaging methods used in the manufacturing process are suitable for use in manufacturing in accordance with current good manufacturing practice, or cGMP, and current Good Tissue Practices, or cGTP;
|
| • |
build and validate processing equipment that complies with cGMP and cGTP;
|
| • |
equip a commercial manufacturing facility to accommodate the automated manufacturing process;
|
| • |
perform process testing with final equipment, disposable components and reagents to demonstrate that the methods are suitable for use in cGMP and cGTP manufacturing;
|
| • |
demonstrate consistency and repeatability of the automated manufacturing processes in the production of AGS-003 in our new facility to fully validate the manufacturing and control process using the actual automated cGMP processing equipment; and
|
| • | demonstrate comparability between AGS-003 that we produce using our manual processes and AGS-003 produced using the automated processes. |
We will need regulatory approval to use the automated manufacturing processes for commercial purposes. If the FDA requires us to conduct a bridging study to demonstrate comparability between AGS-003 that we produce manually and AGS-003 produced using the automated processes, the implementation of the automated manufacturing processes and the filing for such approval will likely be delayed.
If we are unable to successfully implement the automated processes required and demonstrate comparability between the AGS-003 that we produce manually and the AGS-003 produced using the automated processes, our filing for regulatory approval of the commercial use of our automated manufacturing processes may be delayed or denied and we may not be able to initiate commercial
manufacturing using our automated manufacturing processes. In such event, our commercial manufacturing costs will be higher than anticipated and we may not be able to manufacture sufficient product to meet our expected commercial requirements.
We may in the future be dependent on contract manufacturers for the manufacture of AGS-003 or any of our other product candidates. If we experience problems with any such contract manufacturers, the manufacturing of AGS-003 or any of our other product candidates could be delayed.
We may choose to engage a contract manufacturer to supply AGS-003 on a commercial scale on our behalf. Reliance on such third-party contractors entails risks, including:
| • | manufacturing delays if our third-party contractors give greater priority to the supply of other products over our product candidates or otherwise do not satisfactorily perform according to the terms of the agreements between us and them; |
| • | the possible termination or nonrenewal of agreements by our third-party contractors at a time that is costly or inconvenient for us; |
| • | the possible breach by the third-party contractors of our agreements with them; |
| • | the failure of third-party contractors to comply with applicable regulatory requirements; |
| • | the possible mislabeling of clinical supplies, potentially resulting in the wrong dose amounts being supplied or active drug or placebo not being properly identified; |
| 57 |
• the possibility of clinical supplies not being delivered to clinical sites on time, leading to clinical trial interruptions, or of drug supplies not being distributed to commercial vendors in a timely manner, resulting in lost sales; and
• the possible misappropriation of our proprietary AGS-003 or any of our other product candidates.
Any loss of a contract manufacturer could be disruptive to our operations, delay our clinical trials and, if our products are approved for sale, result in lost sales.
Lack of coordination internally among our employees and externally with physicians, hospitals and third- party suppliers and carriers, could cause manufacturing difficulties, disruptions or delays and cause us to not have sufficient product to meet our expected clinical trial requirements or potential commercial requirements.
Manufacturing our Arcelis-based product candidates requires coordination internally among our employees and externally with physicians, hospitals and third party suppliers and carriers. For example, a patient’s physician or clinical site will need to coordinate with us for the shipping of a patient’s disease sample and leukapheresis product to our manufacturing facility, and we will need to coordinate with them for the shipping of the manufactured product to them. Such coordination involves a number of risks that may lead to failures or delays in manufacturing our Arcelis-based product candidates, including:
| • | failure to obtain a sufficient supply of key raw materials of suitable quality; |
| • | difficulties in manufacturing our product candidates for multiple patients simultaneously; |
| • | difficulties in obtaining adequate patient-specific material, such as tumor samples, virus samples or leukapheresis product, from physicians; |
| • | difficulties in completing the development and validation of the specialized assays required to ensure the consistency of our product candidates; |
| • | failure to ensure adequate quality control and assurances in the manufacturing process as we increase production quantities; |
| • | difficulties in the timely shipping of patient-specific materials to us or in the shipping of our product candidates to the treating physicians due to errors by third party carriers, transportation restrictions or other reasons; |
| • | destruction of, or damage to, patient-specific materials or our product candidates during the shipping process due to improper handling by third party carriers, hospitals, physicians or us; |
| • | destruction of, or damage to, patient-specific materials or our product candidates during storage at our facilities; and |
| • | destruction of, or damage to, patient-specific materials or our product candidates stored at clinical and future commercial sites due to improper handling or holding by clinicians, hospitals or physicians. |
If we are unable to coordinate appropriately, we may encounter delays or additional costs in achieving our clinical and commercialization objectives, including in obtaining regulatory approvals of our product candidates and supplying product, which could materially damage our business and financial position.
If our existing manufacturing facility or any commercial manufacturing facility that we are using is damaged or destroyed, or production at one of these facilities is otherwise interrupted, our business and prospects would be negatively affected.
We currently have a single manufacturing facility and expect that the new commercial manufacturing facility that we build out and equip will be our only commercial manufacturing facility in North America. If our existing manufacturing facility or the new commercial manufacturing facility that we plan to build out and equip, or the equipment in either of these facilities, is damaged or destroyed, we likely would not be able to quickly or inexpensively replace our manufacturing capacity and possibly would not be able to replace it at all. Any new facility needed to replace either our existing manufacturing facility or a new commercial manufacturing facility would need to comply with the necessary regulatory requirements, need to be tailored to our specialized automated manufacturing requirements and require specialized equipment. We would need FDA approval before selling any products manufactured at a new facility. Such an event could delay our clinical trials or, if any of our product candidates are approved by the FDA, reduce or eliminate our product sales.
| 58 |
We maintain insurance coverage to cover damage to our property and equipment and to cover business interruption and research and development restoration expenses. If we have underestimated our insurance needs with respect to an interruption in our clinical manufacturing of our product candidates, we may not be able to adequately cover our losses.
Risks Related to Our Intellectual Property
If we fail to comply with our obligations under our intellectual property licenses with third parties, we could lose license rights that are important to our business.
We are a party to a number of intellectual property license agreements with third parties, including with respect to each of AGS-003 and AGS-004, and we expect to enter into additional license agreements in the future. Our existing license agreements impose, and we expect that future license agreements will impose, various diligence, milestone payment, royalty, insurance and other obligations on us. For example, under our license agreement with Duke University which relates to patents and patent applications directed towards the composition of matter of Arcelis-based products, dendritic cells loaded with RNA from tumors or pathogens, methods of manufacture of these products and methods of using these products to treat tumors, we are required to use commercially reasonable efforts to research, develop and market license products and to satisfy other specified obligations. If we fail to comply with our obligations under these licenses, our licensors may have the right to terminate these license agreements, in which event we might not be able to market any product that is covered by these agreements, or to convert the license to a non-exclusive license, which could materially adversely affect the value of the product candidate being developed under the license agreement. Termination of these license agreements or reduction or elimination of our licensed rights may result in our having to negotiate new or reinstated licenses with less favorable terms.
| 59 |
If we are unable to obtain and maintain patent protection for our technology and products, or if our licensors are unable to obtain and maintain patent protection for the technology or products that we license from them, or if the scope of the patent protection obtained is not sufficiently broad, our competitors could develop and commercialize technology and products similar or identical to ours, and our ability to successfully commercialize our technology and products may be adversely affected.
Our success depends in large part on our and our licensors’ ability to obtain and maintain patent protection in the United States and other countries with respect to our proprietary technology and products. We and our licensors have sought to protect our proprietary position by filing patent applications in the United States and abroad related to our novel technologies and products that are important to our business. This process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our research and development efforts before it is too late to obtain patent protection. Moreover, in some circumstances, we do not have the right to control the preparation, filing and prosecution of patent applications, or to maintain the patents, covering technology or products that we license from third parties and are reliant on our licensors. Therefore, we cannot be certain that these patents and applications will be prosecuted and enforced in a manner consistent with the best interests of our business. If such licensors fail to maintain such patents, or lose rights to those patents, the rights we have licensed may be reduced or eliminated.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions and has in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial value of our and our licensors’ patent rights are highly uncertain. Our and our licensors’ pending and future patent applications may not result in patents being issued which protect our technology or products or which effectively prevent others from commercializing competitive technologies and products. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of our patents or narrow the scope of our patent protection.
Third parties could practice our inventions in territories where we do not have patent protection. Furthermore, the laws of foreign countries may not protect our rights to the same extent as the laws of the United States. For example, we own or exclusively license patents relating to our process of manufacturing an individualized drug product. A U.S. patent may be infringed by anyone who, without authorization, practices the patented process in the United States or imports a product made by a process covered by the U.S. patent. In foreign countries, however, importation of a product made by a process patented in that country may not constitute an infringing activity, which would limit our ability to enforce our process patents against importers in that country. Furthermore, the legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection. This could make it difficult for us to stop the infringement of our patents or the misappropriation of our other intellectual property rights. If competitors are able to use our technologies, our ability to compete effectively could be harmed.
Furthermore, publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. Therefore we cannot be certain that we or our licensors were the first to make the inventions claimed in our owned or licensed patents or pending patent applications, or that we or our licensors were the first to file for patent protection of such inventions.
Assuming the other requirements for patentability are met, in the United States, the first to invent the claimed invention is entitled to the patent, while outside the United States, the first to file a patent application is generally entitled to the patent. Under the America Invents Act, or AIA, enacted in September 2011, the United States moved to a first inventor to file system in March 2013. The United States Patent and Trademark Office only recently finalized the rules relating to these changes and courts have yet to address the new provisions. These changes could increase the costs and uncertainties surrounding the prosecution of our patent applications and the enforcement or defense of our patent rights. Furthermore, we may become involved in interference proceedings, opposition proceedings, or other post-grant proceedings, such as reissue, reexamination or inter partes review proceedings, which may challenge our patent rights or the patent rights of others. For example, we have filed an application for reissue of one of our U.S. patents directed towards methods of manufacture of dendritic cells from monocytes stored for more than six hours and up to four days without freezing. An adverse determination in any such proceeding or litigation could reduce the scope of, or invalidate, our patent rights, allow third parties to commercialize our technology or products and compete directly with us, without payment to us, or result in our inability to manufacture or commercialize products without infringing third party patent rights.
Even if our owned and licensed patent applications issue as patents, they may not issue in a form that will provide us with any meaningful protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage. Our competitors may be able to circumvent our owned or licensed patents by developing similar or alternative technologies or products in a non-infringing manner. The issuance of a patent is not conclusive as to its scope, validity or enforceability, and our owned and licensed patents may be challenged in the courts or patent offices in the United States and abroad. Such challenges may result in patent claims being narrowed, invalidated or held unenforceable, which could limit our ability to or stop or prevent us from stopping others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection of our technology and products. Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after such candidates are commercialized. For example, certain of the U.S. patents we exclusively license from Duke University expire as early as 2016 and the European and Japanese patents exclusively licensed from Duke University expire in 2017. As a result, our owned and licensed patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
| 60 |
We may become involved in lawsuits to protect or enforce our patents, which could be expensive, time consuming and unsuccessful.
Competitors may infringe our patents. To counter such infringement or unauthorized use, we may be required to file infringement claims against third parties, which can be expensive and time consuming. In addition, during an infringement proceeding, a court may decide that the patent rights we are asserting are invalid or unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated or interpreted narrowly. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, our licensors may have rights to file and prosecute such claims and we are reliant on them.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain and could have a material adverse effect on the success of our business.
Our commercial success depends upon our ability and the ability of our collaborators to develop, manufacture, market and sell our product candidates and use our proprietary technologies without infringing the proprietary rights of third parties. We cannot ensure that third parties do not have, or will not in the future obtain, intellectual property rights such as granted patents that could block our ability to operate as we would like. There may be patents in the United States or abroad owned by third parties that, if valid, may block our ability to make, use or sell our products in the United States or certain countries outside the United States, or block our ability to import our products into the United States or into certain countries outside the United States.
We may become party to, or threatened with, future adversarial proceedings or litigation regarding intellectual property rights with respect to our products and technology. For example, third parties may assert infringement claims against us based on existing patents or patents that may be granted in the future. If we are found to infringe a third party’s intellectual property rights, we could be required to obtain a license from such third party to continue developing and marketing our products and technology. However, we may be unable to obtain any required license on commercially reasonable terms or even obtain a license at all. Even if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed to us. We could be forced, including by court order, to cease commercializing the infringing technology or product. In addition, we could be found liable for monetary damages. A finding of infringement could prevent us from commercializing our product candidates or force us to cease some of our business operations, which could materially harm our business. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on our business.
We have research licenses to certain reagents and their use in the development of our product candidates. We would need commercial licenses to these reagents for any of our product candidates that receive approval for sale in the United States. We believe that commercial licenses to these reagents will be available. However, if we are unable to obtain any such commercial licenses, we may be unable to commercialize our product candidates without infringing the patent rights of third parties. If we did seek to commercialize our product candidates without a license, these third parties could initiate legal proceedings against us.
We may be subject to claims that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
Many of our employees were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that we or these employees have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employer. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management.
| 61 |
Intellectual property litigation could cause us to spend substantial resources and distract our personnel from their normal responsibilities.
Even if resolved in our favor, litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant expenses, and could distract our technical and management personnel from their normal responsibilities. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of our common stock. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities. We may not have sufficient financial or other resources to adequately conduct such litigation or proceedings. Some of our competitors may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In addition to seeking patents for some of our technology and products, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. The types of protections available for trade secrets are particularly important with respect to our Arcelis platform’s manufacturing capabilities, which involve significant unpatented know-how. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside scientific collaborators, sponsored researchers, contract manufacturers, consultants, advisors and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees and consultants. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor, our competitive position would be harmed.
Risks Related to Legal Compliance Matters
Any product candidate for which we obtain marketing approval could be subject to restrictions or withdrawal from the market and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unanticipated problems with our products, when and if any of them are approved.
Any product candidate for which we obtain marketing approval, along with the manufacturing processes, post-approval clinical data, labeling, advertising and promotional activities for such product, will be subject to continual requirements of and review by the FDA and other regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, registration and listing requirements, cGMP requirements relating to quality control, quality assurance and corresponding maintenance of records and documents, cGTP requirements, requirements regarding the distribution of samples to physicians and recordkeeping. Even if regulatory approval of a product candidate is granted, the approval may be subject to limitations on the indicated uses for which the product may be marketed or to the conditions of approval, or contain requirements for costly post-marketing testing and surveillance to monitor the safety or efficacy of the product. The FDA closely regulates the post-approval marketing and promotion of drugs to ensure drugs are marketed only for the approved indications and in accordance with the provisions of the approved label. The FDA imposes stringent restrictions on manufacturers’ communications regarding off-label use and if we do not market our products for their approved indications, we may be subject to enforcement action for off-label marketing. In addition, later discovery of previously unknown problems with our products, manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may yield various results, including:
| • | restrictions on such products, manufacturers or manufacturing processes; |
| • | restrictions on the marketing of a product; |
| • | restrictions on product distribution; |
| • | requirements to conduct post-marketing clinical trials; |
| • | warning or untitled letters; |
| 62 |
| • | withdrawal of the products from the market; |
| • | refusal to approve pending applications or supplements to approved applications that we submit; |
| • | recall of products; |
| • | fines, restitution or disgorgement of profits or revenue; |
| • | suspension or withdrawal of regulatory approvals; |
| • | refusal to permit the import or export of our products; |
| • | product seizure; or |
| • | injunctions or the imposition of civil or criminal penalties. |
Our relationships with customers and third party payors will be subject to applicable anti-kickback, fraud and abuse and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties, program exclusion, contractual damages, reputational harm and diminished profits and future earnings.
Healthcare providers, physicians and third party payors play a primary role in the recommendation and prescription of any product candidates for which we obtain marketing approval. Our future arrangements with third party payors and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we market, sell and distribute our products for which we obtain marketing approval. Restrictions under applicable federal and state healthcare laws and regulations, include the following:
| • | the federal healthcare anti-kickback statute prohibits, among other things, persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made under federal and state healthcare programs such as Medicare and Medicaid; |
| • | the federal False Claims Act imposes civil penalties, including civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government; |
| • | the federal Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act, imposes criminal and civil liability for executing a scheme to defraud any healthcare benefit program and also imposes obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information; |
| • | the federal false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement in connection with the delivery of or payment for healthcare benefits, items or services; |
| • | the federal transparency requirements under the Health Care Reform Law will require manufacturers of drugs, devices, biologics and medical supplies to report to the Department of Health and Human Services information related to physician payments and other transfers of value and physician ownership and investment interests; and |
| • | analogous state laws and regulations, such as state anti-kickback and false claims laws, may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third party payors, including private insurers, and some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government in addition to requiring drug manufacturers to report information related to payments to physicians and other health care providers or marketing expenditures. |
Some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government and may require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures. State and foreign laws also govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, thus complicating compliance efforts.
| 63 |
Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, exclusion from government funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations. If any of the physicians or other providers or entities with whom we expect to do business are found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs.
Recently enacted and future legislation may increase the difficulty and cost for us to obtain marketing approval of and commercialize our product candidates and affect the prices we may obtain.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of our product candidates, restrict or regulate post-approval activities and affect our ability to profitably sell any product candidates for which we obtain marketing approval.
In the United States, the Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or Medicare Modernization Act, changed the way Medicare covers and pays for pharmaceutical products. The legislation expanded Medicare coverage for drug purchases by the elderly and introduced a new reimbursement methodology based on average sales prices for physician administered drugs. In addition, this legislation provided authority for limiting the number of drugs that will be covered in any therapeutic class in certain cases. Cost reduction initiatives and other provisions of this and other more recent legislation could decrease the coverage and reimbursement that is provided for any approved products. While the Medicare Modernization Act applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own reimbursement rates. Therefore, any reduction in reimbursement that results from the Medicare Modernization Act or other more recent legislation may result in a similar reduction in payments from private payors.
In March 2010, President Obama signed into law the Health Care Reform Law, a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for health care and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. Effective October 1, 2010, the Health Care Reform Law revises the definition of “average manufacturer price” for reporting purposes, which could increase the amount of Medicaid drug rebates to states. Further, the new law imposes a significant annual fee on companies that manufacture or import branded prescription drug products. Substantial new provisions affecting compliance have also been enacted, which may affect our business practices with health care practitioners. We will not know the full effects of the Health Care Reform Law until applicable federal and state agencies issue regulations or guidance under the new law. Although it is too early to determine the effect of the Health Care Reform Law, the new law appears likely to continue the pressure on pharmaceutical pricing, especially under the Medicare program, and may also increase our regulatory burdens and operating costs.
Legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. We cannot be sure whether additional legislative changes will be enacted, or whether the FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing approvals of our product candidates, if any, may be. In addition, increased scrutiny by the U.S. Congress of the FDA’s approval process may significantly delay or prevent marketing approval, as well as subject us to more stringent product labeling and post-marketing testing and other requirements.
With the enactment of the Biologics Price Competition and Innovation Act of 2009, or BPCIA, as part of the Health Care Reform Law, an abbreviated pathway for the approval of biosimilar and interchangeable biological products was created. The new abbreviated regulatory pathway establishes legal authority for the FDA to review and approve biosimilar biologics, including the possible designation of a biosimilar as “interchangeable” based on its similarity to an existing brand product. Under the BPCIA, an application for a biosimilar product cannot be submitted to the FDA until four years, or approved by the FDA until 12 years, after the original brand product identified as the reference product was approved under a BLA. The new law is complex and is only beginning to be interpreted and implemented by the FDA. As a result, its ultimate impact, implementation and meaning is subject to uncertainty. While it is uncertain when any such processes may be fully adopted by the FDA, any such processes could have a material adverse effect on the future commercial prospects for our biological products.
| 64 |
We believe that if any of our product candidates were to be approved as biological products under a BLA, such approved products should qualify for the four-year and 12-year periods of exclusivity. However, there is a risk that the U.S. Congress could amend the BPCIA to significantly shorten these exclusivity periods as proposed by President Obama, or that the FDA will not consider our product candidates to be reference products for competing products, potentially creating the opportunity for generic competition sooner than anticipated. Moreover, the extent to which a biosimilar, once approved, will be substituted for any one of our reference products in a way that is similar to traditional generic substitution for non-biological products is not yet clear, and will depend on a number of marketplace and regulatory factors that are still developing.
If we fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of our business.
We are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Our operations involve the use of hazardous and flammable materials, including chemicals and radioactive and biological materials. Our operations also produce hazardous waste products. We generally contract with third parties for the disposal of these materials and wastes. We cannot eliminate the risk of contamination or injury from these materials. In the event of contamination or injury resulting from our use of hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts. Failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions.
Risks Related to Employee Matters and Managing Growth
Our future success depends on our ability to retain our chief executive officer and other key executives and to attract, retain and motivate qualified personnel.
We are highly dependent on Jeffrey Abbey, our President and Chief Executive Officer, Charles Nicolette, our Vice President of Research and Development and Chief Scientific Officer, Lee F. Allen, our Chief Medical Officer, and Fred Miesowicz, our Vice President of Manufacturing and Chief Operating Officer, as well as the other principal members of our management and scientific teams. Although we have formal employment agreements with each of our executive officers, these agreements do not prevent our executives from terminating their employment with us at any time. We do not maintain “key person” insurance for any of our executives or other employees. The loss of the services of any of these persons could impede the achievement of our research, development and commercialization objectives.
Recruiting and retaining qualified scientific, clinical, manufacturing and sales and marketing personnel will also be critical to our success. We may not be able to attract and retain these personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel from universities and research institutions. In addition, we rely on consultants and advisors, including scientific and clinical advisors, to assist us in formulating our research and development and commercialization strategy. Our consultants and advisors may be employed by employers other than us and may have commitments under consulting or advisory contracts with other entities that may limit their availability to us.
We expect to expand our development, regulatory, manufacturing and sales and marketing capabilities, and as a result, we may encounter difficulties in managing our growth, which could disrupt our operations.
We expect to experience significant growth in the number of our employees and the scope of our operations, particularly in the areas of drug development, regulatory affairs, manufacturing and sales and marketing. To manage our anticipated future growth, we must continue to implement and improve our managerial, operational and financial systems, expand our facilities and continue to recruit and train additional qualified personnel. Due to our limited financial resources and the limited experience of our management team in managing a company with such anticipated growth, we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel. The physical expansion of our operations may lead to significant costs and may divert our management and business development resources. Any inability to manage growth could delay the execution of our business plans or disrupt our operations.
| 65 |
Risks Related to Our Common Stock
Our executive officers, directors and affiliates of all officers and directors who own our outstanding common stock maintain the ability to control all matters submitted to stockholders for approval.
Our executive officers, directors and affiliates of all officers and directors who own our outstanding common stock beneficially own, in the aggregate, shares representing approximately 61.6% of our outstanding common stock as of March 23, 2016. If we consummate the second and third tranches of the March 2016 private placement, and assuming that the shares in the third tranche are sold at a price of $5.35 per share, our executive officers, directors and affiliates of all officers and directors who own our outstanding common stock would beneficially own, in the aggregate, shares representing approximately 79.1% of our outstanding common stock, based on the number of shares of common stock outstanding as of March 23, 2016. As a result, if these stockholders were to choose to act together, they would be able to control all matters submitted to our stockholders for approval, as well as our management and affairs. For example, these persons, if they choose to act together, would control the election of directors and approval of any merger, consolidation or sale of all or substantially all of our assets. This concentration of voting power could delay or prevent an acquisition of our company on terms that other stockholders may desire.
Our largest stockholder, Pharmstandard, could exert significant influence over us and could limit your ability to influence the outcome of key transactions, including any change of control.
Our largest stockholder, Pharmstandard, beneficially owns, in the aggregate, shares representing approximately 34.2% of our outstanding common stock as of March 23, 2016. If we consummate the second and third tranches of the March 2016 private placement, and assuming that the shares in the third tranche are sold at a price of $5.35 per share, Pharmstandard would beneficially own, in the aggregate, shares representing approximately 43.1% of our outstanding common stock. In addition, two members of our board of directors are affiliates of Pharmstandard. As a result, although Pharmstandard is subject to contractual limitations on its voting rights, we expect that Pharmstandard will be able to exert significant influence over our business. Pharmstandard may have interests that differ from your interests, and it may vote in a way with which you disagree and that may be adverse to your interests. The concentration of ownership of our capital stock may have the effect of delaying, preventing or deterring a change of control of our company, could deprive our stockholders of an opportunity to receive a premium for their common stock as part of a sale of our company and may adversely affect the market price of our common stock.
Provisions in our corporate charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our corporate charter and our bylaws may discourage, delay or prevent a merger, acquisition or other change in control of us that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your shares. These provisions could also limit the price that investors might be willing to pay in the future for shares of our common stock, thereby depressing the market price of our common stock. In addition, because our board of directors is responsible for appointing the members of our management team, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors. Among other things, these provisions:
| • | establish a classified board of directors such that not all members of the board are elected at one time; |
| • | allow the authorized number of our directors to be changed only by resolution of our board of directors; |
| • | limit the manner in which stockholders can remove directors from the board; |
| • | establish advance notice requirements for stockholder proposals that can be acted on at stockholder meetings and nominations to our board of directors; |
| • | require that stockholder actions must be effected at a duly called stockholder meeting and prohibit actions by our stockholders by written consent; |
| • | limit who may call stockholder meetings; |
| 66 |
| • | authorize our board of directors to issue preferred stock without stockholder approval, which could be used to institute a “poison pill” that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by our board of directors; and |
| • | require the approval of the holders of at least 75% of the votes that all our stockholders would be entitled to cast to amend or repeal certain provisions of our charter or bylaws. |
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner.
An active trading market for our common stock may not be sustained.
Although our common stock is listed on The NASDAQ Global Market, an active trading market for our shares may not be sustained. If an active market for our common stock is not sustained, it may be difficult for you to sell your shares without depressing the market price for the shares or sell your shares at all. Any inactive trading market for our common stock may also impair our ability to raise capital to continue to fund our operations by selling shares and may impair our ability to acquire other companies or technologies by using our shares as consideration.
If our stock price is volatile, purchasers of our common stock could incur substantial losses.
Our stock price is likely to be volatile. For example, our stock has traded in a range from a low of $1.61 and high of $13.74 during the period of February 7, 2014 through March 23, 2016.The stock market in general and the market for biotechnology companies in particular have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. The market price for our common stock may be influenced by many factors, including:
| • | our cash resources; |
| • | results of clinical trials of our product candidates or those of our competitors; |
| • | the success of competitive products or technologies; |
| • | potential approvals of our product candidates for marketing by the FDA or equivalent foreign regulatory authorities or our failure to obtain such approvals; |
| • | regulatory or legal developments in the United States and other countries; |
| • | the results of our efforts to commercialize our product candidates; |
| • | developments or disputes concerning patents or other proprietary rights; |
| • | the recruitment or departure of key personnel; |
| • | the level of expenses related to any of our product candidates or clinical development programs; |
| • | the results of our efforts to discover, develop, acquire or in-license additional product candidates or products; |
| • | actual or anticipated changes in estimates as to financial results, development timelines or recommendations by securities analysts; |
| • | variations in our financial results or those of companies that are perceived to be similar to us; |
| • | changes in the structure of healthcare payment systems; |
| • | market conditions in the pharmaceutical and biotechnology sectors and issuance of new or changed securities analysts’ reports or recommendations; |
| • | general economic, industry and market conditions; and |
| 67 |
| • | the other factors described in this “Risk Factors” section. |
We will continue to incur increased costs as a result of operating as a public company, and our management will be required to devote substantial time to new compliance initiatives and corporate governance practices.
Pursuant to Section 404 of the Sarbanes-Oxley Act of 2002, we are required to furnish a report by our management on our internal control over financial reporting. However, while we remain an emerging growth company, we will not be required to include an attestation report on internal control over financial reporting issued by our independent registered public accounting firm. To achieve compliance with Section 404 of the Sarbanes-Oxley Act of 2002 within the prescribed period, we will be engaged in a process to document and evaluate our internal control over financial reporting, which is both costly and challenging. In this regard, we will need to continue to dedicate internal resources, potentially engage outside consultants and adopt a detailed work plan to assess and document the adequacy of internal control over financial reporting, continue steps to improve control processes as appropriate, validate through testing that controls are functioning as documented and implement a continuous reporting and improvement process for internal control over financial reporting. If we identify one or more material weaknesses, it could result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our financial statements.
We are an “emerging growth company,” and the reduced disclosure requirements applicable to emerging growth companies may make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act, and may remain an emerging growth company for up to five years. For so long as we remain an emerging growth company, we are permitted and plan to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, reduced disclosure obligations regarding executive compensation and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. In our 2015 Annual Report on Form 10-K, we did not include all of the executive compensation related information that would be required if we were not an emerging growth company. We cannot predict whether investors will find our common stock less attractive if we rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
Because we do not anticipate paying any cash dividends on our capital stock in the foreseeable future, capital appreciation, if any, will be your sole source of gain.
We have never declared or paid cash dividends on our capital stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. In September 2014, we entered into a venture loan and security agreement with Horizon Technology Finance Corporation and Fortress Credit Co LLC. The terms of this agreement preclude us from paying dividends. As a result, capital appreciation, if any, of our common stock will be your sole source of gain for the foreseeable future.
A significant portion of our total outstanding shares is restricted from immediate resale but may be sold into the market in the near future, which could cause the market price of our common stock to decline significantly, even if our business is doing well.
Sales of a substantial number of shares of our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares of common stock intend to sell shares, could reduce the market price of our common stock. As of March 23, 2016, we had 25,330,229 shares of common stock outstanding. Of these shares, the 3,652,430 shares of our common stock issued in our March 2016 private placement are restricted and are subject to limitations on sale under federal securities laws for a period of up to six months or earlier if we file a registration statement covering such shares as we agreed under the registration rights agreement entered into in connection with the private placement, and the additional 12,589,229 shares held by our directors, officers and affiliates are subject to applicable volume limitations on sales under federal securities laws. The balance of our outstanding shares of common stock may be freely sold in the public market at any time.
| 68 |
In addition, as of March 23, 2016, there were 3,908,549 shares subject to outstanding options under our equity incentive plans, all of which shares we have registered under the Securities Act of 1933, as amended, which we refer to as the Securities Act, on a registration statement on Form S-8. These shares, once vested and issued upon exercise, will be able to be freely sold in the public market, subject to volume limits applicable to affiliates. Furthermore, following the initial closing of the private placement, there were 3,560,165 shares subject to outstanding warrants. The shares issuable upon exercise of these warrants will become eligible for sale in the public market to the extent such warrants are exercised and to the extent covered by a registration statement or permitted by Rule 144 under the Securities Act.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our share price and trading volume could decline.
The trading market for our common stock will depend on the research and reports that securities or industry analysts publish about us or our business. We do not have any control over these analysts. There can be no assurance that analysts will cover us, or provide favorable coverage. If one or more analysts downgrade our stock or change their opinion of our stock to be less favorable, our share price would likely decline. In addition, if one or more analysts cease coverage of our company or fail to regularly publish reports on us, we could lose visibility in the financial markets, which could cause our share price or trading volume to decline.
Item 1B. Unresolved Staff Comments
None.
Our only facility is located in Durham, North Carolina, where we occupy approximately 20,000 square feet of office, laboratory and manufacturing space. Our lease expires in November 2016.
In August 2014, we entered into a lease agreement, or the Lease Agreement, with TKC LXXII, LLC, a North Carolina limited liability company, or TKC. Under the Lease Agreement, we agreed to lease certain land and an approximately 124,000 square-foot building to be constructed on approximately 11 acres in Durham County, North Carolina. This facility is being built to house our corporate headquarters and primary manufacturing facility. The shell of the new facility was constructed on a build-to-suit basis by TKC in accordance with agreed upon specifications and plans as set forth in the Lease Agreement, at the expense of TKC, other than those costs resulting from changes requested by us for which we have paid $1.7 million as of December 31, 2015, as further described below.
The term of the Lease Agreement is 10 years from the commencement date of July 1, 2015. We have an option to extend the Lease Agreement by six five-year renewal terms. Initial rent is $46,917 per month, subject to certain fixed increases over the course of the term as set forth in the Lease Agreement and to adjustment based on our use of certain amounts allocated for upfitting the interior of the facility.
Under the Lease Agreement, we had an option to purchase the property. In February 2015, we exercised this purchase option and entered into a Purchase and Sale Agreement with TKC. The purchase price to be paid by us is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by us, for which we have paid $1.7 million as of December 31, 2015, and the amount of any improvement allowances advanced to us by TKC prior to the closing. Under the terms of the Purchase and Sale Agreement, we had until February 16, 2016 to consummate the purchase of the property. We are currently in discussions with TKC to extend the period for purchasing the property as we will need to obtain additional funding to consummate the purchase of the property. If we purchase the property, upon the closing, the Lease Agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to us.
From time to time, we have been and may again become involved in legal proceedings arising in the ordinary course of our business. We are not presently a party to any material litigation.
Item 4. Mine Safety Disclosures
Not Applicable.
| 69 |
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Our common stock is listed on the NASDAQ Global Market under the symbol “ARGS” and began trading on February 7, 2014. Prior to that, there was no public trading market for our common stock. As of March 23, 2016, there were 25,330,229 outstanding shares and 57 stockholders of record. This number does not include beneficial owners whose shares were held in street name. This number of holders of record also does not include stockholders whose shares may be held in trust by other entities.
The following table sets forth for the periods indicated the high and low closing sale prices for our common stock as reported on the NASDAQ Global Market:
| 2015 | ||||||||
| High | Low | |||||||
| First quarter | $ | 10.56 | $ | 6.36 | ||||
| Second quarter | $ | 9.64 | $ | 6.51 | ||||
| Third quarter | $ | 6.98 | $ | 4.11 | ||||
| Fourth quarter | $ | 6.35 | $ | 1.61 | ||||
| 2014 | ||||||||
| High | Low | |||||||
| First quarter (February 7, 2014 to March 31, 2014) | $ | 13.74 | $ | 7.97 | ||||
| Second quarter | $ | 10.55 | $ | 6.21 | ||||
| Third quarter | $ | 10.80 | $ | 5.61 | ||||
| Fourth quarter | $ | 10.28 | $ | 7.80 | ||||
Stock Performance Graph
The graph set forth below compares the cumulative total stockholder return on an initial investment of $100 in our common stock between February 7, 2014, the date on which our common stock began trading on the NASDAQ Global Market, and December 31, 2015, with the comparative cumulative total return of such amount on (i) the NASDAQ Composite Index and (ii) the NASDAQ Biotechnology Index over the same period. We have not paid any cash dividends and, therefore, the cumulative total return calculation for us is based solely upon our stock price appreciation or depreciation and does not include any reinvestment of cash dividends. The graph assumes our closing sales price on February 7, 2014 of $8.00 per share as the initial value of our common stock.
| 70 |
The comparisons shown in the graph below are based upon historical data. We caution that the stock price performance shown in the graph below is not necessarily indicative of, nor is it intended to forecast, the potential future performance of our common stock.
The information presented above in the stock performance graph shall not be deemed to be "soliciting material" or to be "filed" with the SEC or subject to Regulation 14A or 14C, except to the extent that we subsequently specifically request that such information be treated as soliciting material or specifically incorporate it by reference into a filing under the Securities Act of 1933, as amended, or a filing under the Securities Exchange Act of 1934, as amended.
Dividends
We have never declared or paid cash dividends on our capital stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. In September 2014, we entered into a venture loan and security agreement with Horizon Technology Finance Corporation and Fortress Credit Co LLC. The terms of this agreement preclude us from paying dividends.
Recent Sales of Unregistered Securities
We did not sell any unregistered equity securities during the period covered by this Annual Report on Form 10-K that have not already been reported in a Quarterly Report on Form 10-Q or in a Current Report on Form 8-K.
Purchase of Equity Securities
We did not purchase any of our equity securities during the fourth quarter of the period covered by this Annual Report on Form 10-K.
| 71 |
Item 6. Selected Financial Data
You should read the following selected consolidated financial data together with our consolidated financial statements and the related notes in “Item 8. Financial Statements and Supplementary Data” and “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” in this Annual Report on Form 10-K. We have derived the consolidated statements of operations data for the years ended December 31, 2013, 2014 and 2015, and the consolidated balance sheet data as of December 31, 2014 and 2015 from our audited consolidated financial statements included in “Item 8. Financial Statements and Supplementary Data.” The consolidated statements of operations data for the year ended December 31, 2011 and 2012 and the consolidated balance sheet data as of December 31, 2011, 2012 and 2013 was derived from our audited financial statements which are not included in this Annual Report on Form 10-K. Our historical results for any prior period are not necessarily indicative of results to be expected in any future period. The consolidated financial information reflects a one-for-six reverse stock split of our common stock effected on January 17, 2014, which has been retrospectively applied for all periods presented other than the year ended December 31, 2015.
Consolidated Statements of Operations Data:
| Year Ended December 31, | ||||||||||||||||||||
| 2011 | 2012 | 2013 | 2014 | 2015 | ||||||||||||||||
| Revenue | $ | 7,642,695 | $ | 7,039,010 | $ | 4,421,689 | $ | 1,974,019 | $ | 518,329 | ||||||||||
| Operating expenses: | ||||||||||||||||||||
| Research and development | 12,668,025 | 17,616,892 | 23,991,151 | 45,498,916 | 62,054,823 | |||||||||||||||
| General and administrative | 3,703,813 | 6,135,581 | 4,662,317 | 8,599,359 | 11,011,011 | |||||||||||||||
| Operating loss | (8,729,143 | ) | (16,713,463 | ) | (24,231,779 | ) | (52,124,256 | ) | (72,547,505 | ) | ||||||||||
| Other income (expense): | ||||||||||||||||||||
| Interest income | 1,259 | 4,604 | 7,184 | 66,580 | 25,382 | |||||||||||||||
| Interest expense | (6,656,366 | ) | (292,496 | ) | (4,705 | ) | (1,123,579 | ) | (2,263,599 | ) | ||||||||||
| Change in fair value of warrant liability | (4,661,811 | ) | 4,916,785 | 355,352 | — | — | ||||||||||||||
| Derivative (expense) income | (94,668 | ) | 1,036,403 | — | — | — | ||||||||||||||
| Investment tax credits | — | 694,331 | — | 140,556 | — | |||||||||||||||
| Other expense | — | (117,494 | ) | (47,615 | ) | (265,239 | ) | (2,799 | ) | |||||||||||
| Other (expense) income, net | (11,411,586 | ) | 6,242,133 | 310,216 | (1,181,682 | ) | (2,241,016 | ) | ||||||||||||
| Net loss | (20,140,729 | ) | (10,471,330 | ) | (23,921,563 | ) | (53,305,938 | ) | (74,788,521 | ) | ||||||||||
| Net loss attributable to noncontrolling interest | (63,047 | ) | — | — | — | — | ||||||||||||||
| Net loss attributable to Argos Therapeutics, Inc. | (20,077,682 | ) | (10,471,330 | ) | (23,921,563 | ) | (53,305,938 | ) | (74,788,521 | ) | ||||||||||
| Accretion of redeemable convertible preferred stock | (926,542 | ) | (351,371 | ) | 4,772,991 | (863,226 | ) | — | ||||||||||||
| Less: Preferred stock dividend due to exchanges of preferred shares | — | — | (14,726,088 | ) | — | — | ||||||||||||||
| Net loss attributable to common stockholders | $ | (21,004,224 | ) | $ | (10,822,701 | ) | $ | (33,874,660 | ) | $ | (54,169,164 | ) | $ | (74,788,521 | ) | |||||
| Basic and diluted net loss attributable to common stockholders per share | $ | (197.29 | ) | $ | (54.58 | ) | $ | (147.37 | ) | $ | (3.12 | ) | $ | (3.66 | ) | |||||
| Basic and diluted weighted average shares outstanding | 106,466 | 198,306 | 229,865 | 17,367,632 | 20,457,245 | |||||||||||||||
| 72 |
Consolidated Balance Sheet Data:
| As of December 31, | ||||||||||||||||||||
| 2011 | 2012 | 2013 | 2014 | 2015 | ||||||||||||||||
| Cash, cash equivalents and short-term investments | $ | 2,002,814 | $ | 12,363,736 | $ | 46,957,782 | $ | 56,239,937 | $ | 7,166,304 | ||||||||||
| Total assets | 5,973,958 | 15,396,973 | 51,131,295 | 64,366,878 | 31,131,202 | |||||||||||||||
| Total long-term liabilities | — | 48,428 | 10,080,106 | 29,718,320 | 51,169,824 | |||||||||||||||
| Redeemable convertible preferred stock | 77,722,306 | 75,800,882 | 113,664,469 | — | — | |||||||||||||||
| Total stockholders’ (deficit) equity | (96,355,106 | ) | (68,567,710 | ) | (75,776,593 | ) | 31,351,804 | (28,201,435 | ) | |||||||||||
| 73 |
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes appearing in “Item 8. Financial Statements and Supplementary Data” in this Annual Report on Form 10-K. In addition to historical information, some of the information contained in this discussion and analysis or set forth elsewhere in this Annual Report on Form 10-K, including information with respect to our plans and strategy for our business, future financial performance, expense levels and liquidity sources, includes forward-looking statements that involve risks and uncertainties. You should read “Item 1A. Risk Factors” in this Annual Report on Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
Overview
We are an immuno-oncology company focused on the development and commercialization of fully individualized immunotherapies for the treatment of cancer and infectious diseases based on our proprietary technology platform called Arcelis.
Our most advanced product candidate is AGS-003, which we are developing for the treatment of metastatic renal cell carcinoma, or mRCC, and other cancers. We are currently conducting a pivotal Phase 3 clinical trial of AGS-003 plus sunitinib or another targeted therapy for the treatment of newly diagnosed mRCC under a special protocol assessment, or SPA, with the Food and Drug Administration, or FDA. We refer to this trial as the ADAPT trial. We initiated the ADAPT trial in January 2013, dosed the first patient in May 2013 and completed enrollment of the trial in July 2015. Based upon the actual rate of enrollment and projected event rate as defined in the protocol, we anticipate having a sufficient number of events to permit the primary analysis and assessment of overall survival to occur in the first half of 2017. We are also supporting investigator-initiated Phase 2 trials in patients with early stage RCC and non-small cell lung cancer and plan to support investigator-initiated trials of AGS-003 in mRCC and muscle invasive bladder cancer.
We are developing AGS-004, our second most advanced Arcelis-based product candidate, for the treatment of HIV. We have completed Phase 1 and Phase 2 trials funded by government grants and a Phase 2b trial that was funded in full by the National Institutes of Health, or NIH, and the National Institute of Allergy and Infectious Diseases, or NIAID, under a $39.8 million agreement. We are currently supporting an ongoing investigator-initiated Phase 2 clinical trial of AGS-004 in adult HIV patients evaluating the use of AGS-004 in combination with a latency reversing drug for HIV eradication, and plan to support a second investigator-initiated Phase 2 clinical trial of AGS-004 evaluating AGS-004 for long-term viral control in pediatric patients.
We have devoted substantially all of our resources to our drug development efforts, including advancing our Arcelis platform, conducting clinical trials of our product candidates, protecting our intellectual property and providing general and administrative support for these operations. We have not generated any revenue from product sales and, to date, have funded our operations primarily through our initial public offering, a venture loan, private placements of common stock, preferred stock and warrants, convertible debt financings, government contracts, government and other third party grants and license and collaboration agreements. From inception in May 1997 through December 31, 2015, we have raised a total of $388.3 million in cash, including:
| • | $225.8 million from the sale of our common stock, convertible debt, warrants and preferred stock; |
| • | $32.9 million from the licensing of our technology; |
| • | $104.6 million from government contracts, grants and license and collaboration agreements; and |
| • | $25.0 million from our venture loan and security agreement, or the Loan Agreement, with Horizon Technology Finance Corporation and Fortress Credit Co LLC, or the Lenders. |
In February 2014, we issued and sold 6,228,725 shares of our common stock, including 603,725 shares of common stock sold pursuant to the underwriters’ exercise of their option to purchase additional shares, in our initial public offering, at a public offering price of $8.00 per share, for aggregate gross proceeds of $49.8 million. The net offering proceeds to us, after deducting underwriting discounts and commissions of approximately $3.5 million and offering expenses of approximately $2.9 million, were approximately $43.4 million.
In September 2014, we entered into the Loan Agreement with the Lenders under which we could borrow up to $25.0 million in two tranches of $12.5 million each. We borrowed the first tranche of $12.5 million upon the closing of the transaction in September 2014 and borrowed the second tranche of $12.5 million in August 2015, following completion of enrollment in the ADAPT trial.
| 74 |
In April 2015, we entered into stock purchase agreements with Tianyi Lummy International Holdings Group, Ltd., or Tianyi Lummy, and China BioPharma Capital I, L.P., or China BioPharma. Pursuant to the purchase agreements, the purchasers purchased an aggregate of 1,000,000 shares of our common stock at a per share price of $10.11, or approximately $10.1 million, which represented a premium to the then fair market value price of $8.57 per share. The $10.1 million of proceeds was allocated $8.6 million to equity and $1.5 million to deferred revenue. The purchasers also agreed to purchase approximately $10.0 million in additional shares of our common stock, for a total aggregate investment of approximately $20 million, within 31 days of and subject to reaching full enrollment of our ADAPT trial, receiving a recommendation of the review board for the continuation of our ADAPT trial following 50% of events and receiving positive feedback from the FDA on a qualified protocol to demonstrate comparability of our automated manufacturing process for AGS-003 to the manufacturing process used by us in our ADAPT trial. However, in March 2016, in connection with the agreement by Tianyi Lummy and China BioPharma to purchase approximately $10 million of shares of our common stock and warrants in our private placement, we agreed they would have no further obligation to purchase shares pursuant to the purchase agreements.
In March 2016, we entered into a securities purchase agreement with certain investors pursuant to which we agreed to issue and sell an aggregate of up to $60 million of our common stock and warrants to purchase shares of common stock in a private placement financing. The financing will take place in up to three tranches. At the closing of the initial tranche in March 2016, we sold and the investors purchased, for a total purchase price of approximately $19.9 million, a total of 3,652,430 shares of common stock and warrants to purchase a total of 2,739,323 shares of common stock (0.75 shares of common stock for each share of common stock purchased), based on a purchase price per share of common stock and accompanying warrant equal to $5.44375. At the second closing, we have agreed to sell and the investors have agreed to purchase, for an additional purchase price of approximately $29.8 million, a total of 5,478,672 shares of common stock and warrants to purchase a total of 4,109,005 shares of common stock at the same price and on the same terms as the first tranche. The second closing is conditioned on the independent data monitoring committee for the ADAPT trial at or following the independent data monitoring committee’s next regular meeting (currently scheduled for June 2016) recommending that we continue the ADAPT trial or discontinue the ADAPT trial based on favorable efficacy data. The warrants to be issued in each closing will have an exercise price of $5.35 per share and expire five years from the date of issuance. Our stockholder, Pharmstandard International S.A., or Pharmstandard, has also agreed pursuant to the securities purchase agreement that, at our option following the satisfaction of certain conditions, including the independent data monitoring committee having made a recommendation at or following its next regular meeting after the June 2016 independent data monitoring committee meeting (currently anticipated to be held in November or December 2016), that we continue the ADAPT trial or discontinue the ADAPT trial based on favorable efficacy data, and our cash position at such time, Pharmstandard shall purchase at the third closing up to approximately $10.3 million of shares of common stock (without warrants) at a price per share to be determined pursuant to an agreed upon formulation. The dollar amount committed to be purchased by Pharmstandard at the third closing is subject to reduction on a dollar-for-dollar basis for certain cash amounts raised by us after the initial closing through equity or debt financings or collaborations. All three closings are subject to the satisfaction of certain customary closing conditions.
Commercial Facility. In August 2014, we entered into a lease agreement, or the Lease Agreement, with TKC LXXII, LLC, a North Carolina limited liability company, or TKC. Under the Lease Agreement, we agreed to lease certain land and an approximately 124,000 square-foot building to be constructed on approximately 11 acres in Durham County, North Carolina. This facility is being built to house our corporate headquarters and primary manufacturing facility. The lease for the current facility expires in November 2016. The shell of the new facility was constructed on a build-to-suit basis by TKC in accordance with agreed upon specifications and plans as set forth in the Lease Agreement, at the expense of TKC, other than those costs resulting from changes requested by us for which we have paid $1.7 million as of December 31, 2015, as further described below.
The term of the Lease Agreement is 10 years from the commencement date of July 1, 2015. We have an option to extend the Lease Agreement by six five-year renewal terms. Initial rent is $46,917 per month, subject to certain fixed increases over the course of the term as set forth in the Lease Agreement and to adjustment based on our use of certain amounts allocated for upfitting the interior of the facility.
Under the Lease Agreement, we had an option to purchase the property. In February 2015, we exercised this purchase option and entered into a Purchase and Sale Agreement with TKC. The purchase price to be paid by us is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by us, for which we have paid $1.7 million as of December 31, 2015, and the amount of any improvement allowances advanced to us by TKC prior to the closing. Under the terms of the Purchase and Sale Agreement, we had until February 16, 2016 to consummate the purchase of the property. We are currently in discussions with TKC to extend the period for purchasing the property as we will need to obtain additional funding to consummate the purchase of the property. If we purchase the property, upon the closing, the Lease Agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to us.
| 75 |
We have incurred losses in each year since our inception in May 1997. Our net loss was $23.9 million for the year ended December 31, 2013, $53.3 million for the year ended December 31, 2014 and $74.8 million for the year ended December 31, 2015. As of December 31, 2015, we had an accumulated deficit of $279.0 million. Substantially all of our operating losses have resulted from costs incurred in connection with our development programs and from general and administrative costs associated with our operations.
We expect to continue to incur significant expenses and increasing operating losses for at least the next several years. We anticipate that our expenses will increase substantially if and as we:
| • | continue our ongoing ADAPT trial of AGS-003 for the treatment of mRCC; |
| • | continue to support ongoing investigator-initiated clinical trials of AGS-003 and AGS-004; |
| • | support planned investigator-initiated clinical trials of AGS-003 and AGS-004; |
| • |
initiate and conduct additional clinical trials of AGS-003 and AGS-004 for the treatment of cancers and HIV; |
| • | seek regulatory approvals for our product candidates that successfully complete clinical trials; |
| • | purchase or lease, and build out and equip, a commercial facility for the manufacture of our Arcelis-based products; |
| • | establish a sales, marketing and distribution infrastructure to commercialize products for which we may obtain regulatory approval; |
| • | maintain, expand and protect our intellectual property portfolio; |
| • | continue our other research and development efforts; |
| • | hire additional clinical, quality control, scientific and management personnel; and |
| • | add operational, financial and management information systems and personnel, including personnel to support our product development and planned commercialization efforts. |
We do not expect to generate significant funds or product revenue unless and until we successfully complete development, obtain marketing approval and commercialize our product candidates, either alone or in collaboration with third parties, which we expect will take a number of years and is subject to significant uncertainty. Accordingly, we will need to raise additional capital prior to the commercialization of AGS-003, AGS-004 or any of our other product candidates. Until such time, if ever, as we can generate substantial product revenues, we expect to finance our operating activities through a combination of equity offerings, debt financings, government contracts, government and other third party grants or other third party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. However, we may be unable to raise additional funds through these means when needed, on favorable terms or at all.
NIH Funding
In September 2006, we entered into a multi-year research contract with the NIH and NIAID to design, develop and clinically test an autologous HIV immunotherapy capable of eliciting therapeutic immune responses. We have used funds from this contract to develop AGS-004, including to fund in full our Phase 2b clinical trial of AGS-004. We have agreed to a statement of work under the contract, and are obligated to furnish all the services, qualified personnel, material, equipment, and facilities not otherwise provided by the U.S. government needed to perform the statement of work.
Under this contract, as amended, the NIH and NIAID have committed to fund up to a total of $39.8 million, including reimbursement of direct expenses and allocated overhead and general and administrative expenses of up to $38.4 million and payment of other specified amounts totaling up to $1.4 million upon our achievement of specified development milestones. This amount includes a September 2014 modification of the contract under which the NIH and NIAID agreed to fund up to an additional $500,000 to cover a portion of the manufacturing costs of the planned Phase 2 clinical trial of AGS-004 for long-term viral control in pediatric patients. The NIH’s commitment under the contract extends to July 2016. Since September 2010, we have received reimbursement of our allocated overhead and general and administrative expenses at provisional indirect cost rates equal to negotiated provisional indirect cost rates agreed to with the NIH and NIAID in September 2010. These provisional indirect cost rates are subject to adjustment based on our actual costs pursuant to the agreement with the NIH and NIAID and may result in additional payments to us from the NIH and NIAID to reflect our actual costs since September 2010.
| 76 |
We have recorded revenue of $37.3 million through December 31, 2015 under the NIH and NIAID contract. This contract is the only arrangement under which we have generated substantial revenue. As of December 31, 2015, there was up to $2.5 million of potential revenue remaining to be earned under the agreement with the NIH and NIAID.
Collaborations
Pharmstandard
In August 2013, in connection with the purchase of shares of our series E preferred stock by Pharmstandard, we entered into an exclusive royalty-bearing license agreement with Pharmstandard. Under this license agreement, we granted Pharmstandard and its affiliates a license, with the right to sublicense, to develop, manufacture and commercialize AGS-003 and other products for the treatment of human diseases which are developed by Pharmstandard using our individualized immunotherapy platform in the Russian Federation, Armenia, Azerbaijan, Belarus, Georgia, Kazakhstan, Kyrgyzstan, Moldova, Tajikistan, Turkmenistan, Ukraine and Uzbekistan, which we refer to as the Pharmstandard Territory. We also provided Pharmstandard with a right of first negotiation for development and commercialization rights in the Pharmstandard Territory to specified additional products we may develop.
Pharmstandard agreed to pay us royalties on net sales of all licensed products in the low single digits until it has generated a specified amount of aggregate net sales. Once the net sales threshold is achieved, Pharmstandard will pay us royalties on net sales of specified licensed products, including AGS-003, in the low double digits below 20%. After the net sales threshold is achieved, Pharmstandard has the right to offset a portion of the royalties Pharmstandard pays to third parties for licenses to necessary third party intellectual property against the royalties that Pharmstandard pays to us. For further information regarding this collaboration, see “Business — Development and Commercialization Agreements — Pharmstandard.”
In November 2013, we entered into an agreement with Pharmstandard under which Pharmstandard purchased additional shares of our series E preferred stock. Under this agreement, we agreed to enter into a manufacturing rights agreement for the European market with Pharmstandard and that the manufacturing rights agreement would provide for the issuance of warrants to Pharmstandard to purchase 499,788 shares of our common stock at an exercise price of $5.82 per share. As of March 23, 2016, we had not entered into this manufacturing rights agreement or issued the warrants. The shares of series E preferred stock converted to common stock on a 6-for-1 basis in connection with our initial public offering.
Green Cross
In July 2013, in connection with the purchase of shares of our series E preferred stock by Green Cross Corp., or Green Cross, we entered into an exclusive royalty-bearing license agreement with Green Cross. Under this agreement we granted Green Cross a license to develop, manufacture and commercialize AGS-003 for mRCC in South Korea. We also provided Green Cross with a right of first negotiation for development and commercialization rights in South Korea to specified additional products we may develop.
Under the terms of the license, Green Cross has agreed to pay us $0.5 million upon the initial submission of an application for regulatory approval of a licensed product in South Korea, $0.5 million upon the initial regulatory approval of a licensed product in South Korea and royalties ranging from the mid-single digits to low double digits below 20% on net sales until the fifteenth anniversary of the first commercial sale in South Korea. For further information regarding this collaboration, see “Business — Development and Commercialization Agreements — Green Cross.” The shares of series E preferred stock converted to common stock on a 6-for-1 basis in connection with our initial public offering.
Medinet
In December 2013, we entered into a license agreement with Medinet. Under this agreement, we granted Medinet an exclusive, royalty-free license to manufacture in Japan AGS-003 and other products using our Arcelis technology solely for the purpose of the development and commercialization of AGS-003 and these other products for the treatment of mRCC. We refer to this license as the manufacturing license. In addition, under this agreement, we granted Medinet an option to acquire a nonexclusive, royalty-bearing license under our Arcelis technology to sell in Japan AGS-003 and other products for the treatment of mRCC. We refer to the option as the sale option and the license as the sale license. We expect the Medinet option to expire without having been exercised on March 31, 2016.
| 77 |
In consideration for the manufacturing license, Medinet paid us $1.0 million. Medinet also loaned us $9.0 million in connection with us entering into the agreement. We have agreed to use these funds in the development and manufacturing of AGS-003 and the other products. Medinet also agreed to pay us milestone payments of up to a total of $9.0 million upon the achievement of developmental and regulatory milestones and $5.0 million upon the achievement of a sales milestone related to AGS-003 and these products. If Medinet exercises the sale option, it will pay us $1.0 million, as well as royalties on net sales at a rate to be negotiated until the later of the expiration of the licensed patent rights in Japan and the twelfth anniversary of the first commercial sale in Japan. If we cannot agree on the royalty rate, we have agreed to submit the matter to arbitration.
We borrowed the $9.0 million pursuant to an unsecured promissory note that bears interest at a rate of 3.0% per annum. The principal and interest under the note are due and payable on December 31, 2018. Under the terms of the note and the license agreement, any milestone payments related to the developmental and regulatory milestones that become due will be applied first to the repayment of the loan. We have the right to prepay the loan at any time. If we have not repaid the loan by December 31, 2018, then we have agreed to grant to Medinet a non-exclusive, royalty-bearing license to make and sell Arcelis products in Japan for the treatment of cancer. In such event, the amounts owing under the loan as of December 31, 2018 will constitute pre-paid royalties under the license and will not be otherwise due and payable. Royalties under this license would be paid until the expiration of the licensed patent rights in Japan at a rate to be negotiated. If we cannot agree on the royalty rate, we have agreed to submit the matter to arbitration.
We recorded the $1.0 million payment from Medinet as a deferred liability. In addition, because the $9.0 million promissory note was issued at a below market interest rate, we allocated the proceeds of the loan between the manufacturing license and the debt at the time of issuance. As a result, we recorded $6.9 million as notes payable, based upon an effective interest rate of 8%, and $2.1 million as a deferred liability. The total deferred liability recorded related to the manufacturing license was $3.1 million as of December 31, 2014.
During the year ended December 31, 2015, we recognized a $1.0 million milestone payment as deferred revenue under this license agreement and reduced the related note payable by $0.8 million and the deferred liability by $0.2 million. As of December 31, 2015, the amount of the note payable was $7.5 million, including $1.3 million accrued interest. As of December 31, 2015, the total deferred liability associated with the Medinet note was $3.8 million.
For further information regarding this collaboration, see “Business—Development and Commercialization Agreements—Medinet.”
Lummy
On April 7, 2015, we and Lummy HK entered into a license agreement pursuant to which we granted to Lummy HK an exclusive license under the Arcelis technology, including patents, know-how and improvements to manufacture, develop and commercialize products for the treatment of cancer in China, Hong Kong, Taiwan and Macau. Lummy HK also has a right of first negotiation with respect to a license under the Arcelis technology for the treatment of infectious diseases in China, Hong Kong, Taiwan and Macau.
Under the terms of the license agreement, the parties will share relevant data, and we will have a right to reference Lummy HK data for purposes of its development programs under the Arcelis technology. In addition, Lummy HK has granted to us an exclusive, royalty-free license under and to any and all Lummy HK improvements to the Arcelis technology conceived or reduced to practice by Lummy HK and Lummy HK data to develop and/or commercialize products outside China, Hong Kong, Taiwan and Macau, an exclusive, royalty-free license under and to any and all INDs and other regulatory approvals and Lummy HK trademarks used for an Arcelis-Based Product to develop and/or commercialize an Arcelis-Based Product outside China, Hong Kong, Taiwan and Macau and a non-exclusive, worldwide, royalty-free license under any Lummy HK improvements and Lummy HK data to manufacture Arcelis-Based Products anywhere in the world. Lummy HK has the right to reference our data, INDs and other regulatory filings and submissions for the purpose of developing and obtaining regulatory approval of licensed products in China, Hong Kong, Taiwan and Macau.
Pursuant to the license agreement, Lummy HK will pay us royalties on net sales and up to an aggregate of $20.0 million upon the achievement of manufacturing, regulatory and commercial milestones. The license agreement will terminate upon expiration of the last to expire royalty term for all Arcelis-Based Products, with each royalty term being the longer of the expiration of the last valid patent claim covering the applicable Arcelis-Based Product and 10 years from the first commercial sale of such Arcelis-Based Product. Either party may terminate the license agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy. We may terminate the license agreement if Lummy HK challenges or assists a third party in challenging specified patent rights of ours. If Lummy HK terminates the license agreement upon our material breach or bankruptcy, Lummy HK is entitled to terminate the licenses it granted to us and retain its licenses from us with respect to Arcelis-Based Products then in development or being commercialized, subject to Lummy HK’s continued obligation to pay royalties and milestones with respect to such Arcelis-Based Products.
| 78 |
Financial Overview
Revenue
To date, we have not generated revenue from the sale of any products. During the years ended December 31, 2013, 2014 and 2015, substantially all of our revenue has been derived from our NIH and NIAID contract. We may generate revenue in the future from government contracts and grants, payments from future license or collaboration agreements and product sales. We expect that any revenue we generate will fluctuate from quarter to quarter.
Research and Development Expenses
Since our inception in 1997, we have focused our resources on our research and development activities, including conducting preclinical studies and clinical trials, manufacturing development efforts and activities related to regulatory filings for our product candidates. We recognize our research and development expenses as they are incurred. Our research and development expenses consist primarily of:
| • | salaries and related expenses for personnel in research and development functions; |
| • | fees paid to consultants and clinical research organizations, or CROs, including in connection with our clinical trials, and other related clinical trial fees, such as for investigator grants, patient screening, laboratory work and statistical compilation and analysis; |
| • | commercial manufacturing development consisting of costs incurred under our development agreement with Invetech Pty Ltd, or Invetech, under which Invetech has agreed to develop and provide prototypes of the automated production system to be used for the manufacture of our Arcelis-based products; |
| • | allocation of facility lease and maintenance costs; |
| • | costs incurred under our development agreement with Saint-Gobain to develop a range of disposables for use in the automated production system; |
| • | depreciation of leasehold improvements, laboratory equipment and computers; |
| • | costs related to production of product candidates for clinical trials; |
| • | costs related to compliance with regulatory requirements; |
| • | consulting fees paid to third parties related to non-clinical research and development; |
| • | costs related to stock options or other stock-based compensation granted to personnel in research and development functions; and |
| • | acquisition fees, license fees and milestone payments related to acquired and in-licensed technologies. |
Our direct research and development expenses consist principally of external costs, such as fees paid to investigators, consultants, central laboratories and CROs, including in connection with our clinical trials, and related clinical trial fees. Commercial manufacturing development costs consist primarily of costs incurred under our development agreement with Invetech to develop and provide prototypes of the automated production system to be used for the manufacture of our Arcelis-based products. We have been developing AGS-003 and AGS-004 in parallel, and typically use our employee and infrastructure resources across multiple research and development programs. We do not allocate salaries, stock-based compensation, employee benefit or other indirect costs related to our research and development function to specific product candidates.
The successful development of our clinical and preclinical product candidates is highly uncertain. At this time, we cannot reasonably estimate the nature, timing or costs of the efforts that will be necessary to complete the remainder of the development of any of our clinical or preclinical product candidates or the period, if any, in which material net cash inflows from these product candidates may commence. This is due to the numerous risks and uncertainties associated with developing drugs, including the uncertainty of:
| • | the scope, rate of progress, expense and results of our ongoing clinical trials; |
| • | the scope, rate of progress, expense and results of additional clinical trials that we may conduct; |
| 79 |
| • | the scope, rate of progress, expense and results of our commercial manufacturing development efforts; |
| • | other research and development activities; and |
| • | the timing of regulatory approvals. |
A change in the outcome of any of these variables with respect to the development of a product candidate could mean a significant change in the costs and timing associated with the development of that product candidate. For example, if the FDA or another regulatory authority were to require us to conduct clinical trials beyond those which we currently anticipate will be required for the completion of clinical development of a product candidate or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development.
AGS-003. We are developing AGS-003 for the treatment of mRCC and other cancers. We are currently conducting the ADAPT trial of AGS-003 plus sunitinib / targeted therapy for the treatment of newly diagnosed mRCC under an SPA with the FDA. We initiated the ADAPT trial in January 2013 and dosed the first patient in May 2013. In July 2015 we completed enrollment in the trial, enrolling 462 patients with the goal of generating 290 events for the primary endpoint of overall survival. We enrolled these patients at approximately 130 clinical sites in North America and Europe. Based upon the actual rate of enrollment and projected event rate as defined in the protocol, we anticipate having a sufficient number of events to permit the primary analysis and assessment of overall survival to occur in the first half of 2017.
In addition, in mRCC we plan to support an investigator-initiated clinical trial of AGS-003 in combination with a PD-1 inhibitor that we expect will begin in the second half of 2016.
We are also supporting an ongoing investigator-initiated Phase 2 clinical trial designed to evaluate treatment with AGS-003 in patients with early stage RCC prior to nephrectomy. This trial was open for enrollment in late 2014 and four patients were enrolled as of March 15, 2016. We expect that a total of 10 patients will be enrolled in this trial and that initial data will be available in the fourth quarter of 2016. In the trial, patients undergo a core needle biopsy of their renal tumor prior to nephrectomy in order to provide material to extract patient-specific tumor RNA for the manufacture of AGS-003. We expect the investigator to have preliminary data from this trial later this year with respect to the safety, feasibility and initial immunologic effects of AGS-003 relative to the induction of memory T cells. In addition, since the tumor is harvested post-AGS-003 treatment, this trial should enable a direct assessment of whether the immune cells in the peripheral blood end up infiltrating the primary tumor.
We believe that AGS-003 may be capable of treating a wide range of cancers and are planning to evaluate AGS-003 in clinical trials in additional cancer indications. We are supporting an investigator-initiated Phase 2 clinical trial of AGS-003 in patients with non-small cell lung cancer patients, or NSCLC, that was initiated in the first quarter of 2016. In the trial, the safety, efficacy and immunologic effects of AGS-003 when combined with platinum-based chemotherapy and radiation will be evaluated in approximately 20 stage 3 NSCLC patients. We also plan to support an additional investigator-initiated Phase 2 clinical trial of AGS-003 in muscle invasive bladder cancer, which we expect to begin mid-2016.
AGS-004. We are developing AGS-004 for the treatment of HIV and are focusing this program on the use of AGS-004 in combination with other therapies for the eradication of HIV. We believe that by combining AGS-004 with therapies that are being developed to expose the virus in latently infected cells to the immune system, we can potentially eradicate the virus. We are supporting an investigator-initiated Phase 2 clinical trial of AGS-004 in up to 12 adult HIV patients to evaluate the use of AGS-004 in combination with one of these latency reversing therapies for this purpose at the University of North Carolina. The patient clinical costs from the first stage of this trial are being funded by Collaboratory of AIDS Researchers for Eradication. The NIH Division of AIDS has approved $6.6 million in funding for the second stage of this trial.
We also plan to explore the use of AGS-004 monotherapy to provide long-term control of HIV viral load in otherwise immunologically healthy patients and eliminate their need for antiretroviral therapy, or ART. Accordingly, we plan to support an investigator-initiated Phase 2 clinical trial of AGS-004 monotherapy in pediatric patients infected with HIV who have otherwise healthy immune systems and have been treated with ART since birth or shortly thereafter and, as a result, are lacking the antiviral memory T-cells to combat the virus. The commencement of this trial is subject to supportive data obtained from the adult eradication trial and approval of the protocol by the principal investigator(s), institutional review boards, the IMPAACT Network leadership and the FDA and to the agreement by the NIH to fund the trial costs not related to AGS-004 manufacturing. Assuming the supportive data and the necessary approvals are obtained, we expect this trial to initiate in 2017.
Commercial Manufacturing Development. Commercial manufacturing development costs consist primarily of costs incurred under our development agreement with Invetech to develop and provide prototypes of the automated production system to be used for the manufacture of our Arcelis-based products and our development agreement with Saint-Gobain to develop a range of disposables for use in the automated production system.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries and related costs for employees in executive, operational and finance, information technology and human resources functions. Other significant general and administrative expenses include allocation of facilities costs, professional fees for accounting and legal services and expenses associated with obtaining and maintaining patents.
| 80 |
Interest Income and Interest Expense
Interest income consists of interest earned on our cash, cash equivalents and short-term investments.
Interest expense consists primarily of cash and non-cash interest costs related to our debt. During the years ended December 31, 2015 and 2014, interest expense primarily resulted from accrued interest on our note payable to Medinet, which was issued in December 2013 and interest from the Loan Agreement entered into in September 2014. During the year ended December 31, 2013, interest expense was not significant.
Venture Loan and Security Agreement. In September 2014, we entered into the Loan Agreement with the Lenders under which we could borrow up to $25.0 million in two tranches of $12.5 million each. We borrowed the first tranche of $12.5 million upon the closing of the transaction in September 2014 and borrowed the second tranche of $12.5 million on August 7, 2015, following completion of enrollment of the ADAPT trial. The per annum interest rate for each tranche is a floating rate equal to 9.25% plus the amount by which the one-month London Interbank Offered Rate, or LIBOR, exceeds 0.50% (effectively a floating rate equal to 8.75% plus the one-month LIBOR Rate). The total per annum interest rate shall not exceed 10.75%.
Medinet. In December 2013, in connection with the license agreement with Medinet, as described in Note 12 to our consolidated financial statements appearing in “Item 8. Financial Statements and Supplementary Data,” we borrowed $9.0 million pursuant to an unsecured promissory note that bears interest at a rate of 3.0% per annum. The principal and interest under the note are due and payable on December 31, 2018. Under the terms of the note and the license agreement, any milestone payments related to the developmental and regulatory milestones that become due will be applied first to the repayment of the loan. We have the right to prepay the loan at any time. If we have not repaid the loan by December 31, 2018, then we have agreed to grant to Medinet a non-exclusive, royalty-bearing license to make and sell Arcelis products in Japan for the treatment of cancer. In such event, the amounts owing under the loan as of December 18, 2018 may constitute pre-paid royalties under the license or would be due and payable. Royalties under this license would be paid until the expiration of the licensed patent rights in Japan at a rate to be negotiated. If we and Medinet cannot agree on the royalty rate, Medinet has agreed to submit the matter to arbitration. Because the $9.0 million promissory note was issued at a below market interest rate, we allocated the proceeds of the loan between the license agreement and the debt at the time of issuance. Accordingly, as of December 31, 2013, we recorded $6.9 million to notes payable, based upon an effective interest rate of 8.0%, and $2.1 million as a deferred liability. As of December 31, 2014, we recorded $7.6 million to notes payable, including $0.7 million accrued interest recorded during the year ended December 31, 2014. During the year ended December 31, 2015, we recognized a $1.0 million milestone payment as deferred revenue under this license agreement and reduced the related note payable by $0.8 million and the deferred liability by $0.2 million. As of December 31, 2015, we recorded $7.5 million to notes payable, including $1.3 million of accrued interest.
Accretion of Preferred Stock
Our preferred stock was reflected on our balance sheet at its cost, less associated issuance costs. The amount reflected on the balance sheet for our preferred stock was increased by periodic accretions of the issuance costs so that the original amount reflected on the balance sheet will equal the aggregate redemption price.
On February 12, 2014, all outstanding shares of our preferred stock converted into an aggregate of 13,188,251 shares of our common stock upon the closing of our initial public offering.
Critical Accounting Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which we have prepared in accordance with generally accepted accounting principles in the United States, or U.S. GAAP. The preparation of these consolidated financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the reported revenues and expenses during the reporting periods. We evaluate these estimates and judgments on an ongoing basis. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Our actual results may differ from these estimates under different assumptions or conditions.
| 81 |
While our significant accounting policies are more fully described in Note 1 to our consolidated financial statements appearing in “Item 8. Financial Statements and Supplementary Data,” we believe that the following accounting policies are the most critical to aid you in fully understanding and evaluating our financial condition and results of operations.
Revenue Recognition
We recognize revenue in accordance with the Financial Accounting Standards Board, or FASB, Accounting Standards Codification 605, Revenue Recognition, or ASC 605. We recognize revenue when the following criteria are met: persuasive evidence of an arrangement exists, services have been rendered, the price is fixed or determinable and collectability is reasonably assured.
We have previously entered into license agreements with collaborators. The terms of these agreements have included nonrefundable signing and licensing fees, as well as milestone payments and royalties on any future product sales developed by the collaborators under these licenses. We assess these multiple elements in accordance with ASC 605, in order to determine whether particular components of the arrangement represent separate units of accounting.
These collaboration agreements will be accounted for in accordance with Accounting Standards Update, or ASU, No. 2009-13, Topic 605—Multiple-Deliverable Revenue Arrangements, or ASU 2009-13. This guidance requires the application of the “relative selling price” method when allocating revenue in a multiple deliverable arrangement. The selling price for each deliverable shall be determined using vendor specific objective evidence of selling price, if it exists; otherwise, third-party evidence of selling price shall be used. If neither exists for a deliverable, the vendor shall use its best estimate of the selling price for that deliverable. We recognize upfront license payments as revenue upon delivery of the license only if the license has standalone value and the fair value of the undelivered performance obligations can be determined. If the fair value of the undelivered performance obligations can be determined, such obligations are accounted for separately as the obligations are fulfilled. If the license is considered to either not have stand-alone value or have stand-alone value but the fair value of any of the undelivered performance obligations cannot be determined, the arrangement is accounted for as a single unit of accounting and the license payments and payments for performance obligations are recognized as revenue over the estimated period of when the performance obligations are performed.
Whenever we determine that an arrangement should be accounted for as a single unit of accounting, we must determine the period over which the performance obligations will be performed and revenue will be recognized. If we cannot reasonably estimate the timing and the level of effort to complete our performance obligations under the arrangement, then we recognize revenue under the arrangement on a straight-line basis over the period that we expect to complete our performance obligations.
Our license agreements with Pharmstandard, Green Cross, Medinet and Lummy HK provide for, and any future license agreements we may enter into may provide for, milestone payments. Revenues from milestones, if they are non-refundable and considered substantive, are recognized upon successful accomplishment of the milestones. If not considered substantive, milestones are initially deferred and recognized over the remaining performance obligation.
Our current license agreements with Pharmstandard, Green Cross, Medinet and Lummy HK provide for and any future license agreements we may enter into may provide for royalty payments. To date, we have not received any royalty payments and accordingly have not recognized any related revenue. We will recognize royalty revenue upon the sale of the related products, provided we have no remaining performance obligations under the arrangements.
We record deferred revenue when payments are received in advance of the culmination of the earnings process. This revenue is recognized in future periods when the applicable revenue recognition criteria have been met.
Under our NIH and NIAID contract, we receive reimbursement of our direct expenses and allocated overhead and general and administrative expenses, as well as payment of other specified amounts totaling up to $1.4 million upon our achievement of specified development milestones. We recognize revenue from reimbursements earned in connection with the NIH and NIAID contract as reimbursable costs are incurred. We recognize revenues from the achievement of milestones under the NIH and NIAID contract upon the accomplishment of any such milestone.
Accrued Expenses
As part of the process of preparing financial statements, we are required to estimate accrued expenses. This process involves reviewing open contracts and purchase orders, communicating with applicable vendor personnel to identify services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of actual cost. The majority of our service providers invoice us monthly in arrears for services performed. We make estimates of our accrued expenses as of each balance sheet date in our financial statements based on facts and circumstances known to us. We periodically confirm the accuracy of our estimates with the service providers and make adjustments if necessary. Examples of estimated accrued expenses include:
| 82 |
| • | fees paid to CROs in connection with clinical trials; |
| • | fees paid to investigative sites in connection with clinical trials; |
| • | professional service fees; and |
| • | unpaid salaries, wages and benefits. |
We accrue our expenses related to clinical trials based on our estimates of the services received and efforts expended pursuant to contracts with multiple research institutions and CROs that conduct and manage clinical trials on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. Payments under some of these contracts depend on factors such as the successful enrollment of patients and the completion of clinical trial milestones. In accruing service fees, we estimate the time period over which services will be performed and the level of effort to be expended in each period. If the actual timing of the performance of services or the level of effort varies from our estimate, we will adjust the accrual accordingly. If we do not identify costs that we have begun to incur or if we underestimate or overestimate the level of services performed or the costs of these services, our actual expenses could differ from our estimates. We do not anticipate the future settlement of existing accruals to differ materially from our estimates.
Stock-Based Compensation
In accordance with ASC 718, Stock Compensation, we record the fair value of stock options, restricted stock awards and other stock-based compensation issued to employees as of the grant date as compensation expense. We recognize expense over the requisite service period, which is typically the vesting period. For non-employees, we also record stock options, restricted stock awards and other stock-based compensation issued to these non-employees at their fair value as of the grant date. We then periodically remeasure the awards to reflect the current fair value at each reporting period and recognize expense over the related service period.
We calculate the fair value of stock-based compensation awards using the Black-Scholes option-pricing model. The Black-Scholes option-pricing model requires the use of subjective assumptions, including stock price volatility, the expected life of stock options, risk free interest rate and the fair value of the underlying common stock on the date of grant.
| • | We do not have sufficient history to estimate the volatility of our common stock price. We calculate expected volatility based on reported data for selected reasonably similar publicly traded companies for which the historical information is available. For the purpose of identifying peer companies, we consider characteristics such as industry, length of trading history, similar vesting terms and in-the-money option status. We plan to continue to use the guideline peer group volatility information until the historical volatility of our common stock is relevant to measure expected volatility for future option grants. |
| • | The assumed dividend yield is based on our expectation of not paying dividends in the foreseeable future. |
| • | The expected term represents the period that the stock-based awards are expected to be outstanding. Our historical share option exercise experience does not provide a reasonable basis upon which to estimate an expected term because of a lack of sufficient data. Therefore we estimate the expected term by using the simplified method provided by the SEC. The simplified method calculates the expected term as the average of the time-to-vesting and the contractual life of the options. |
| • | We determine the risk-free interest rate by reference to implied yields available from U.S. Treasury securities with a remaining term equal to the expected life assumed at the date of grant. |
| • | We estimate forfeitures based on our historical analysis of actual stock option forfeitures. |
The assumptions that we used in the Black-Scholes option-pricing model for the years ended December 31, 2013, 2014 and 2015, are set forth below:
| 2013 | 2014 | 2015 | ||||||||||
| Risk-free interest rate | 2.12 | % | 2.26 | % | 2.05 | % | ||||||
| Dividend yield | 0 | % | 0 | % | 0 | % | ||||||
| Expected option term (in years) | 7 | 7 | 7 | |||||||||
| Volatility | 94 | % | 96 | % | 87 | % | ||||||
| 83 |
Prior to February 7, 2014, the date our common stock began publicly trading following our initial public offering, the fair value of our common stock for purposes of determining the exercise price for stock option grants was determined by our board of directors, with the assistance and upon the recommendation of management, in good faith based on a number of objective and subjective factors consistent with the methodologies outlined in the American Institute of Certified Public Accountants Practice Aid, Valuation of Privately-Held-Company Equity Securities Issued as Compensation, or the Practice Aid. Since our initial public offering, the exercise price per share of all option grants has been set at the closing price of our common stock on The NASDAQ Global Market on the applicable date of grant, which our board of directors believes represents the fair value of our common stock.
Results of Operations – Year-Over-Year Comparisons
The following table summarizes the results of our operations for each of the years ended December 31, 2013, 2014 and 2015, together with the changes in those items in dollars and as a percentage:
| Year Ended December 31, | $ | % | Year Ended December 31, | $ | % | |||||||||||||||||||||||||||
| 2014 | 2015 | Change | Change | 2013 | 2014 | Change | Change | |||||||||||||||||||||||||
| (in thousands) | ||||||||||||||||||||||||||||||||
| Revenue | $ | 1,974 | $ | 518 | $ | (1,456 | ) | (73.8 | )% | $ | 4,422 | $ | 1,974 | $ | (2,448 | ) | (55.4 | )% | ||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||||||
| Research and development | 45,499 | 62,055 | 16,556 | 36.4 | % | 23,991 | 45,499 | 21,508 | 89.7 | % | ||||||||||||||||||||||
| General and administrative | 8,599 | 11,011 | 2,412 | 28.1 | % | 4,662 | 8,599 | 3,937 | 84.4 | % | ||||||||||||||||||||||
| Total operating expenses | 54,098 | 73,066 | 18,968 | 35.1 | % | 28,653 | 54,098 | 25,445 | 88.8 | % | ||||||||||||||||||||||
| Loss from operations | (52,124 | ) | (72,548 | ) | (20,424 | ) | 39.2 | % | (24,231 | ) | (52,124 | ) | (27,893 | ) | 115.1 | % | ||||||||||||||||
| Interest income | 67 | 25 | (42 | ) | (62.7 | )% | 7 | 67 | 60 | * | ||||||||||||||||||||||
| Interest expense | (1,124 | ) | (2,264 | ) | (1,140 | ) | 101.4 | % | (5 | ) | (1,124 | ) | (1,119 | ) | * | |||||||||||||||||
| Change in fair value of warrant liability | — | — | — | * | 355 | — | (355 | ) | * | |||||||||||||||||||||||
| Investment tax credits | 141 | — | (141 | ) | * | — | 141 | 141 | * | |||||||||||||||||||||||
| Other expense | (266 | ) | (2 | ) | 264 | * | (48 | ) | (266 | ) | (218 | ) | * | |||||||||||||||||||
| Net loss | $ | (53,306 | ) | $ | (74,789 | ) | $ | (21,483 | ) | 40.3 | % | $ | (23,922 | ) | $ | (53,306 | ) | $ | (29,384 | ) | 122.8 | % | ||||||||||
_______________
| * | Not meaningful |
Revenue
To date, we have not generated revenue from the sale of any products. Substantially all of our revenue has been derived from our NIH and NIAID contract. We may generate revenue in the future from government contracts and grants, payments from future license or collaboration agreements and product sales. We expect that any revenue we generate will fluctuate from quarter to quarter.
Revenue was $2.0 million for the year ended December 31, 2014, compared with $0.5 million for the year ended December 31, 2015, a decrease of approximately $1.5 million, or 73.8%. The $1.5 million decrease for the year ended December 31, 2015 resulted from lower reimbursement under our NIH and NIAID contract in 2015 as there was decreased activity with respect to our Phase 2b clinical trial of AGS-004 in the year ended December 31, 2015.
Revenue was $4.4 million for the year ended December 31, 2013, compared with $2.0 million for the year ended December 31, 2014, a decrease of $2.4 million, or 55.4%. The $2.4 million decrease for the year ended December 31, 2014 was due to a $2.2 million decline in reimbursement under our NIH and NIAID contract associated with decreased activity in the year ended December 31, 2014 with respect to our Phase 2b clinical trial of AGS-004 as the number of patients receiving treatment in the trial declined. This decrease in revenue was partially offset by $0.2 million in revenue recognized in connection with technology transfers with Medinet during the year ended December 31, 2014.
| 84 |
Research and Development Expenses
The table below summarizes our direct research and development expenses by program for the periods indicated. Our direct research and development expenses consist principally of external costs, such as fees paid to investigators, consultants, central laboratories and CROs, including in connection with our clinical trials, and related clinical trial fees. Research and development expenses also include commercial manufacturing development costs consisting primarily of costs incurred under our development agreements with Invetech and Saint-Gobain Performance Plastics Corporation, or Saint-Gobain, to develop and provide prototypes of the automated production system to be used for the manufacture of our Arcelis-based products. We have been developing AGS-003 and AGS-004, in parallel, and typically use our employee and infrastructure resources across multiple research and development programs. We do not allocate salaries, stock-based compensation, employee benefit or other indirect costs related to our research and development function to specific product candidates. Those expenses are included in “Indirect research and development expense” in the table below.
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| (in thousands) | ||||||||||||
| Direct research and development expense by program: | ||||||||||||
| AGS-003 | $ | 11,301 | $ | 16,940 | $ | 22,503 | ||||||
| AGS-004 | 1,602 | 903 | 289 | |||||||||
| Other | 52 | 131 | 40 | |||||||||
| Total direct research and development program expense | 12,955 | 17,974 | 22,832 | |||||||||
| Commercial manufacturing development | — | 11,588 | 17,926 | |||||||||
| Indirect research and development expense | 11,036 | 15,937 | 21,297 | |||||||||
| Total research and development expense | $ | 23,991 | $ | 45,499 | $ | 62,055 | ||||||
Research and development expenses were $45.5 million for the year ended December 31, 2014, compared with $62.1 million for the year ended December 31, 2015, an increase of approximately $16.6 million, or 36.4%. The increase in research and development expense reflects a $4.9 million increase in direct research and development expense, a $6.3 million increase in commercial manufacturing development expense and a $5.4 million increase in indirect research and development expense. The increase in direct research and development expenses resulted primarily from the following:
| • | Direct research and development expense for AGS-003 increased from $16.9 million in the year ended December 31, 2014 to $22.5 million in the year ended December 31, 2015. This increase was primarily due to increased patient enrollment in the ADAPT trial in the year ended December 31, 2015 as compared with the year ended December 31, 2014 and a payment during the year ended December 31, 2015 of $2.1 million in common stock for activities related to our master process development and supply agreement with Cellscript LLC; and |
| • | Direct research and development expense with respect to AGS-004 decreased from $0.9 million in the year ended December 31, 2014 to $0.3 million in the year ended December 31, 2015 primarily due to the decreased activity around our Phase 2b clinical trial of AGS-004. |
We also incurred $11.6 million and $17.9 million of research and development expense related to our commercial manufacturing development efforts during the years ended December 31, 2014 and 2015, respectively. We commenced our commercial manufacturing development efforts during the three months ended March 31, 2014 and such activity increased progressively during 2014 and 2015.
The increase in indirect research and development expense was primarily due to higher personnel costs, as we had 97 employees engaged in research and development activities as of December 31, 2014 compared with 118 employees as of December 31, 2015.
Research and development expenses were $24.0 million for the year ended December 31, 2013, compared with $45.5 million for the year ended December 31, 2014, an increase of $21.5 million, or 89.7%. The increase in research and development expense primarily reflects a $5.0 million increase in direct research and development expense, a $11.6 million increase in commercial manufacturing development expense and a $4.9 million increase in indirect research and development expense. The increase in direct research and development expenses resulted primarily from the following:
| 85 |
| • | Direct research and development expense for AGS-003 increased from $11.3 million for the year ended December 31, 2013 to $16.9 million in the year ended December 31, 2014. This increase primarily reflects increased patient enrollment in the ADAPT trial in the year ended December 31, 2014 as compared with the year ended December 30, 2013; and |
| • | Direct research and development expense with respect to AGS-004 decreased from $1.6 million in the year ended December 31, 2013 to $0.9 million in the year ended December 31, 2014 primarily due to the decreased activity in our Phase 2b clinical trial of AGS-004 as the number of patients receiving treatment in the trial declined, which decrease was partially offset by $0.2 million in costs to support stage 1 of an investigator-initiated Phase 2 clinical trial of AGS-004 in adult HIV patients aimed at eradication of the virus that began in May 2014. |
We also incurred $11.6 million of research and development expense related to the initiation of our commercial manufacturing development efforts during the year ended December 31, 2014. The increase in indirect research and development expense was primarily due to higher personnel costs, as we had 80 employees engaged in research and development activities as of December 31, 2013 compared with 97 employees as of December 31, 2014.
General and Administrative Expenses
General and administrative expenses were $8.6 million for the year ended December 31, 2014, compared with $11.0 million for the year ended December 31, 2015, an increase of $2.4 million or 28.1%. This increase was primarily due to an additional $1.7 million in personnel costs, including salaries, benefits and stock-based compensation, additional consulting costs resulting from the implementation of a new enterprise resource planning, or ERP system, and an increase of $0.2 million in expenses relating to our status as a public company, including liability and directors’ and officers’ insurance and registration and service fees.
General and administrative expenses were $4.7 million for the year ended December 31, 2013, compared with $8.6 million for the year ended December 31, 2014, an increase of $3.9 million or 84.4%. This increase was primarily due to an additional $1.4 million in personnel costs, including salaries, benefits and stock-based compensation; an increase of $1.3 million in outside services including legal, patent, and other consulting services; and an increase of $1.2 million in expenses relating to our status as a public company, including liability and directors’ and officers’ insurance, board of directors’ fees and expenses, franchise taxes and registration and service fees.
We expect that our general and administrative expenses will increase with the continued development and potential commercialization of our product candidates.
Interest Expense
Interest expense was $1.1 million for the year ended December 31, 2014, compared with $2.3 million for the year ended December 31, 2015. The increase in interest expense primarily resulted from higher outstanding indebtedness of $12.5 million borrowed under the Loan Agreement during the year ended December 31, 2015. We expect that interest expense will increase as a result of interest incurred under the Loan Agreement.
Interest expense was $5,000 for the year ended December 31, 2013, compared with $1.1 million for the year ended December 31, 2014. During the year ended December 31, 2014, interest expense primarily resulted from accrued interest on our note payable to Medinet, which was issued in December 2013, and interest from the Loan Agreement with the Lenders entered into in September 2014.
Change in Fair Value of Warrant Liability
Income from the change in fair value of the warrant liability was $0.4 million for the year ended December 31, 2013, compared with $0 for the years ended December 31, 2014 and 2015. The 2013 amount represented the decrease in the fair value of our warrant liability during the year ended December 31, 2013. In July 2013, in connection with an exchange in which the holders of shares of our series D-1 preferred stock exchanged all shares of our series D-1 preferred stock held by such holders for shares of our series D preferred stock, all of our outstanding warrants to purchase shares of our preferred stock were cancelled. As a result, there were no warrants to purchase preferred stock outstanding as of December 31, 2013 or during the years ended December 31, 2014 or 2015.
| 86 |
Investment Tax Credits
Other income of $140,556 was recognized during the year ended December 31, 2014 for scientific research and experimental development, or SR&ED, investment tax credits in Canada. Under Canadian and Ontario law, the Company’s Canadian subsidiary is entitled to SR&ED. Because these credits are subject to a claims review, the Company recognizes such credits when received. No such credits were received during the years ended December 31, 2013 or 2015.
Other Expense
Other expense totaled $48,000, $266,000 and $2,799 for the years ended December 31, 2013, 2014 and 2015, respectively. Under a previous loan and security agreement to which we were a party, we had agreed to pay a success fee of $200,000 upon consummation of a liquidity event, including an initial public offering. Our initial public offering closed on February 12, 2014. Accordingly, this fee was paid in March 2014 and was recorded in Other expense on the consolidated statement of operations during the year ended December 31, 2014.
Liquidity and Capital Resources
Sources of Liquidity
As of December 31, 2015, we had cash, cash equivalents and short-term investments of approximately $7.2 million.
Since our inception in May 1997 through December 31, 2015, we have funded our operations principally with $225.8 million from the sale of common stock, convertible debt, warrants and preferred stock, $32.9 million from the licensing of our technology, $104.6 million from government contracts, grants and license and collaboration agreements, and $25.0 million from the Loan Agreement.
The gross proceeds we received from the issuance and sale of our preferred stock were as follows:
| Issue | Year | Gross Proceeds | ||||||
| (in thousands) | ||||||||
| Series A Preferred | 2000 | $ | 1,594 | |||||
| Series B Preferred | 2001 | $ | 39,382 | |||||
| Series B-1 Preferred | 2004 | $ | 5,000 | |||||
| Series C Preferred | 2008 | $ | 33,462 | |||||
| Series D Preferred | 2012 | $ | 9,022 | |||||
| Series D-1 Preferred | 2012 | $ | 15,978 | |||||
| Series E Preferred | 2013 | $ | 48,000 | |||||
Upon the closing of our initial public offering in February 2014, all of the then-outstanding shares of our redeemable convertible preferred stock automatically converted into 13,188,251 shares of common stock.
In February 2014, we issued and sold 6,228,725 shares of our common stock, including 603,725 shares of common stock sold pursuant to the underwriters’ exercise of their option to purchase additional shares, in our initial public offering at a public offering price of $8.00 per share, for aggregate gross proceeds of $49.8 million. The net offering proceeds to us, after deducting underwriting discounts and commissions of approximately $3.5 million and offering expenses of approximately $2.9 million, were approximately $43.4 million.
Venture Loan and Security Agreement. In September 2014, we entered into the Loan Agreement with the Lenders, under which we could borrow up to $25.0 million in two tranches of $12.5 million each.
We borrowed the first tranche of $12.5 million upon the closing of the transaction in September 2014 and borrowed the second tranche of $12.5 million in August 2015 following completion of enrollment of the ADAPT trial. The per annum interest rate for each tranche is a floating rate equal to 9.25% plus the amount by which the one-month LIBOR exceeds 0.50% (effectively a floating rate equal to 8.75% plus the one-month LIBOR Rate). The total per annum interest rate shall not exceed 10.75%.
| 87 |
We have agreed to repay the first tranche of $12.5 million on an interest-only basis monthly until September 30, 2016, followed by monthly payments of principal and accrued interest through the scheduled maturity date for the first tranche loan on September 30, 2018. In addition, a final payment for the first tranche loan equal to $625,000 will be due on September 30, 2018, or such earlier date specified in the Loan Agreement. We have agreed to repay the second tranche of $12.5 million in 18 monthly payments of interest only until February 7, 2017, followed by 24 monthly payments of principal and accrued interest through the scheduled maturity date for the second tranche loan on February 7, 2019. In addition, a final payment of $625,000 will be due on February 7, 2019, or such earlier date specified in the Loan Agreement. In addition, if we repay all or a portion of the loan prior to the applicable maturity date, we will pay the Lenders a prepayment penalty fee, based on a percentage of the then outstanding principal balance, equal to 3% if the prepayment occurs on or before 24 months after the funding date thereof, 2% if the prepayment occurs more than 24 months after, but on or before 36 months after, the funding date thereof, or 1% if the prepayment occurs more than 36 months after the funding date thereof.
Our obligations under the Loan Agreement are secured by a first priority security interest in substantially all of our assets other than our intellectual property. We also have agreed not to pledge or otherwise encumber our intellectual property assets, subject to certain exceptions.
The Loan Agreement includes customary affirmative and restrictive covenants, but does not include any covenants to attain or maintain certain financial metrics, and also includes customary events of default, including payment defaults, breaches of covenants, change of control and a material adverse change default. Upon the occurrence of an event of default and following any applicable cure periods, a default interest rate of an additional 5% may be applied to the outstanding loan balances, and the Lenders may declare all outstanding obligations immediately due and payable and take such other actions as set forth in the Loan Agreement.
In connection with the Loan Agreement, we issued to the Lenders and their affiliates warrants to purchase a total of 82,780 shares of our Common Stock, or the Warrants, at a per share exercise price of $9.06. Upon our satisfaction of the conditions precedent to the making of the second tranche loan, the Warrants became exercisable in full. The Warrants will terminate on September 29, 2021 or such earlier date as specified in the Warrants.
Commercial Facility. In August 2014, we entered into the Lease Agreement with TKC. Under the Lease Agreement, we agreed to lease certain land and an approximately 124,000 square-foot building to be constructed on approximately 11 acres in Durham County, North Carolina. This facility is being built to house our corporate headquarters and primary manufacturing facility and to replace our existing facility in Durham, North Carolina. The lease for the current facility expires in November 2016. The shell of the new facility was constructed on a build-to-suit basis by TKC in accordance with agreed upon specifications and plans as set forth in the Lease Agreement at the expense of TKC, other than those costs resulting from changes requested by us for which we have paid $1.7 million as of December 31, 2015.
The term of the Lease Agreement is 10 years from the commencement date of July 1, 2015. We have the option to extend the Lease Agreement by six five-year renewal terms. Initial rent is $46,917 per month, subject to certain fixed increases over the course of the term as set forth in the Lease Agreement and to adjustment based on our use of certain amounts allocated for upfitting the interior of the facility.
Under the lease agreement, we had an option to purchase the property. In February 2015, we exercised this purchase option and entered into a Purchase and Sale Agreement with TKC. The purchase price to be paid by us is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by us, for which we have paid $1.7 million as of December 31, 2015, and the amount of any improvement allowances advanced to us by TKC prior to the closing. Under the terms of the Purchase and Sale Agreement, we had until February 16, 2016 to consummate the purchase of the property. We are currently in discussions with TKC to extend the period for purchasing the property as we will need to obtain additional funding to consummate the purchase of the property. If we purchase the property, upon the closing, the lease agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to us.
Lummy License Agreement. On April 7, 2015, we and Lummy HK entered into a license agreement, or the License Agreement, whereby we granted to Lummy HK an exclusive license to our Arcelis technology, including patents, know-how and improvements to manufacture, develop and commercialize products for the treatment of cancer in China, Hong Kong, Taiwan and Macau.
In connection with the License Agreement, we entered into stock purchase agreements with Tianyi Lummy and China BioPharma of which Lummy HK’s parent company is an affiliate and limited partner, respectively. Pursuant to the purchase agreements, the purchasers purchased an aggregate of 1,000,000 shares of our common stock at a per share price of $10.11, or approximately $10.1 million. The purchasers also agreed to purchase approximately $10.0 million in additional shares of our common stock, for a total aggregate investment of approximately $20 million, within 31 days of and subject to reaching full enrollment of our ADAPT trial of AGS-003 for the treatment of mRCC, receiving a recommendation of the review board for the continuation of our ADAPT trial following 50% of events and receiving positive feedback from the FDA on a qualified protocol to demonstrate comparability of our automated manufacturing process for AGS-003 to the manufacturing process used by us in our ADAPT trial. However, in March 2016, in connection with the agreement by Tianyi Lummy and China BioPharma to purchase approximately $10 million of shares of our common stock and warrants in our private placement, we agreed they would have no further obligation to purchase shares pursuant to the purchase agreements.
| 88 |
Shelf Registration Statement; ATM. On May 8, 2015, we filed a shelf registration statement on Form S-3, or the 2015 Shelf, with the SEC, which covers the offering, issuance and sale of up to $125,000,000 of our common stock, preferred stock, debt securities, depositary shares, purchase contracts, purchase units and warrants. We simultaneously entered into a Sales Agreement with Cowen and Company LLC to provide for the offering, issuance and sale of up to $30,000,000 of our common stock from time to time in “at-the-market” offerings under the 2015 Shelf. The 2015 Shelf was declared effective by the SEC on May 14, 2015. We have not sold any shares under the Sales Agreement.
PIPE Financing. On March 4, 2016, we entered into a securities purchase agreement with certain investors pursuant to which we agreed to issue and sell an aggregate of up to $60 million of our common stock and warrants to purchase shares of common stock in a private placement financing. The financing will take place in up to three tranches. At the closing of the initial tranche in March 2016, we sold and the investors purchased, for a total purchase price of approximately $19.9 million, a total of 3,652,430 shares of common stock and warrants to purchase a total of 2,739,323 shares of common stock (0.75 shares of common stock for each share of common stock purchased), based on a purchase price per share of common stock and accompanying warrant equal to $5.44375. At the second closing, we have agreed to sell and the investors have agreed to purchase, for an additional purchase price of approximately $29.8 million, a total of 5,478,672 shares of common stock and warrants to purchase a total of 4,109,005 shares of common stock at the same price and on the same terms as the first tranche. The second closing is conditioned on the independent data monitoring committee for the ADAPT trial at or following the independent data monitoring committee’s next regular meeting (currently scheduled for June 2016) recommending that we continue the ADAPT trial or discontinue the ADAPT trial based on favorable efficacy data. The warrants to be issued in each closing will have an exercise price of $5.35 per share and expire five years from the date of issuance. Our stockholder, Pharmstandard International S.A., or Pharmstandard, has also agreed pursuant to the securities purchase agreement that, at our option following the satisfaction of certain conditions, including the independent data monitoring committee having made a recommendation at or following its next regular meeting after the June 2016 independent data monitoring committee meeting (currently anticipated to be held in November or December 2016), that we continue the ADAPT trial or discontinue the ADAPT trial based on favorable efficacy data, and our cash position at such time, Pharmstandard shall purchase at the third closing up to approximately $10.3 million of shares of common stock (without warrants) at a price per share to be determined pursuant to an agreed upon formulation. The dollar amount committed to be purchased by Pharmstandard at the third closing is subject to reduction on a dollar-for-dollar basis for certain cash amounts raised by us after the initial closing through equity or debt financings or collaborations. All three closings are subject to the satisfaction of certain customary closing conditions. In connection with entering into the securities purchase agreement, we entered into a registration rights agreement with the investors pursuant to which we agreed to register for resale the shares issued in the financing and the shares issuable upon exercise of the warrants issued in the financing.
Cash Flows
The following table sets forth the major sources and uses of cash for the periods set forth below:
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| (in thousands) | ||||||||||||
| Net cash (used in) provided by: | ||||||||||||
| Operating activities | $ | (18,256 | ) | $ | (45,241 | ) | $ | (61,021 | ) | |||
| Investing activities | (10,057 | ) | (7,789 | ) | 8,923 | |||||||
| Financing activities | 53,404 | 56,966 | 21,062 | |||||||||
| Effect of exchange rate changes on cash | (8 | ) | (10 | ) | (24 | ) | ||||||
| Net increase (decrease) in cash and cash equivalents | $ | 25,083 | $ | 3,926 | $ | (31,060 | ) | |||||
Operating Activities. Net cash used in operating activities of $18.3 million during the year ended December 31, 2013 was primarily a result of our $23.9 million net loss, partially offset by changes in operating assets and non-cash items of $1.3 million and liabilities of $4.3 million. These non-cash items primarily consisted of depreciation of $0.6 million and compensation expense related to stock options of $1.1 million, partially offset by the gain recorded due to the decrease in fair value of our warrant liability of $0.4 million.
Net cash used in operating activities of $45.2 million during the year ended December 31, 2014 was primarily a result of our $53.3 million net loss, partially offset by non-cash items of $4.4 million and changes in operating assets and liabilities of $3.7 million. These non-cash items primarily consisted of depreciation and amortization of $0.6 million, compensation expense related to stock options of $3.0 million and interest accrued on long-term debt of $0.7 million. The long-term portion of accrued manufacturing research and development expenses increased by $3.7 million.
| 89 |
Net cash used in operating activities of $61.0 million during the year ended December 31, 2015 was primarily a result of our $74.8 million net loss, partially offset by non-cash items of $7.2 million and changes in operating assets and liabilities of $6.6 million. These non-cash items primarily consisted of depreciation and amortization expense of $0.7 million, compensation expense related to stock options of $4.0 million, payment for research and development services by issuing common stock of $2.1 million and amortization of debt issuance costs and debt discount of $0.3 million. In addition, accrued expenses increased by $0.8 million, prepaid expenses and other receivables decreased by $0.2 million, long-term deferred liabilities increased by $1.5 million and the long-term portion of our manufacturing research and development obligation increased by $4.3 million, which were partially offset by a decrease in accounts payable of $0.1 million.
Investing Activities. Net cash (used in) provided by investing activities amounted to ($10.1) million, ($7.8) million and $8.9 million for the years ended December 31, 2013, 2014 and 2015, respectively. Cash used in and provided by investing activities during each of these periods primarily reflected our purchases of property and equipment and purchases and maturities of short-term investments. Cash paid for purchases of property and equipment totaled $0.6 million, $1.1 million and $9.7 million during the years ended December 31, 2013, 2014 and 2015, respectively. Cash used in investment activities during the year ended December 31, 2014 also included $25.6 million of purchases of short-term investments with funds received in our initial public offering and the $1.3 million payment to a restricted cash account securing a letter of credit, partially offset by $20.2 million in proceeds from maturities of short-term investments. Cash provided by investing activities during the year ended December 31, 2015 included $20.7 million in proceeds from maturities of short-term investments and the $0.6 million reduction and receipt from the restricted cash account securing a letter of credit, partially offset by $2.7 million of purchases of short-term investments.
Financing Activities. Net cash provided by financing activities amounted to $53.4 million, $57.0 million and $21.1 million for the years ended December 31, 2013, 2014 and 2015, respectively. Cash provided by financing activities for the year ended December 31, 2013 primarily consisted of proceeds of $48.0 million from the sale of our series E preferred stock and attached common stock warrants and $6.9 million from the note payable to Medinet, partially offset by stock issuance costs of $1.5 million. Cash provided by financing activities for the year ended December 31, 2014 consisted primarily of proceeds of $49.8 million from the sale of common stock in our initial public offering, which closed on February 12, 2014, $12.5 million of loan proceeds from our Loan Agreement, which closed on September 29, 2014, partially offset by stock and debt issuance costs totaling $5.3 million, and payments on other notes payable of $51,481. Cash provided by financing activities for the year ended December 31, 2015 consisted of $12.5 million of loan proceeds from our Loan Agreement, proceeds of $8.6 million from the sale of common stock and $0.4 million of proceeds from the exercise of stock options and from our employee stock purchase plan, partially offset by $35,480 of payments on notes payable.
Significant Changes in Consolidated Balance Sheet as of December 31, 2015 Compared with December 31, 2014
Property and equipment, net, increased by $16.8 million during the year ended December 31, 2015 primarily due to a $3.9 million increase in the asset recognized related to the Lease Agreement for our new manufacturing facility and $12.5 million from construction-in-progress. The facility lease obligation increased by $3.9 million during the year ended December 31, 2015 due to activity under the lease to build the facility. Accounts payable increased by $1.0 million and accrued expenses increased by $2.3 million during the year ended December 31, 2015 primarily due to an increase in activity under our manufacturing development contracts and purchases of property and equipment. Long-term deferred liabilities increased by $2.3 million, primarily due to deferred revenue recognized under the license agreements with Lummy HK and Medinet.
Funding Requirements
To date, we have not generated any product revenue from our development stage product candidates. We do not know when, or if, we will generate any product revenue. We do not expect to generate significant product revenue unless or until we obtain marketing approval of, and commercialize AGS-003 or AGS-004. At the same time, we expect our expenses to increase in connection with our ongoing activities, particularly as we continue our ADAPT trial of AGS-003, initiate additional clinical trials of AGS-003 and AGS-004, seek regulatory approval for our product candidates and purchase or lease, and build out and equip a new commercial manufacturing facility. In addition, if we obtain regulatory approval of any of our product candidates, we expect to incur significant commercialization expenses for product sales, marketing, manufacturing and distribution. Furthermore, we expect to incur additional costs associated with operating as a public company. We will need substantial additional funding in connection with our continuing operations.
| 90 |
Based on our current operating plan, we expect that our cash, cash equivalents and short-term investments as of December 31, 2015, the $19.0 million of net proceeds from the sale of common stock and warrants at the initial closing of our March 2016 private placement and the anticipated funding under our NIH and NIAID contract, will enable us to fund our operating expenses into the third quarter of 2016. If the second and third tranches of our March 2016 private placement financing occur, we expect that our cash, cash equivalents and short-term investments as of December 31, 2015, the estimated $59.0 million of net proceeds from the financing and the anticipated funding under our contract with the NIH and NIAID, will enable us to fund our operating expenses into the second quarter of 2017, when we expect final data from the ADAPT trial.
We expect that these funds will be sufficient to enable us to support the ongoing and planned investigator-initiated clinical trials of AGS-003, certain of the manufacturing costs of the ongoing phase 2 clinical trial of AGS-004 for HIV eradication in patients with early stage RCC and certain costs related to commercial manufacturing. We expect that these funds will not, however, be sufficient to enable us to complete required activities in preparation for the submission of a BLA to the FDA for AGS-003, to perform pre-commercialization activities for AGS-003 or to commercially launch AGS-003. These expectations are based on our current operating plan under which we plan to implement measures to reduce our operating expenses, including reductions in spending for activities in preparation for submission of a BLA. These reductions may include reductions in planned spending related to chemistry, manufacturing and controls, process validation, quality, supply management and commercial preparation. Under our plan, we also intend to seek to refinance our existing venture loan facility with Horizon Technology Finance Corporation, or Horizon, and Fortress Credit Co LLC, or Fortress, and to enter into collaborations for the development, marketing and distribution of AGS-003 outside North America and of our non-oncology product candidates, including AGS-004. If we are unable to refinance our venture loan facility or enter into any such collaborations, we may be required to implement additional measures to materially reduce our operating expenses, which could adversely affect our business and operations. It is also possible that our available funds will not enable us to obtain final data from the ADAPT trial because the actual costs and timing of clinical trials are difficult to predict and are subject to substantial risks and delays. We have no external sources of funds other than our contract with the NIH and NIAID for the development of AGS-004.
We also will need to obtain significant financing if we decide to purchase, build out and equip a new commercial manufacturing facility. Our preliminary estimate indicates that we will require approximately an additional $30.0 million prior to the commercialization of AGS-003 to purchase, build out and equip the commercial manufacturing facility. These costs do not include the costs to implement the automated manufacturing process. We are actively exploring financing arrangements in connection with the planned purchase, build out and equipping of the commercial manufacturing facility and are in discussions with developers, lenders and other potential financing sources regarding potential financial support. Unless we select an alternative plan for the commercial supply of our products, we expect to enter into such arrangements during the second quarter of 2016 and that such arrangements will likely involve material obligations and debt liabilities. We are also exploring alternatives for the commercial supply of our products, including building out and equipping our current facility for commercial manufacture, leasing and equipping an existing manufacturing space that would be built out for commercial manufacture, and engaging a contract manufacturer to supply AGS-003 on a commercial scale on our behalf. We will also need to obtain additional financing with any of these alternatives. If we are unable to obtain additional financing when needed, in the required amounts or at all, we may not be able to complete the planned purchase, build-out and equipping of the new commercial facility or be able to lease and equip an existing commercial facility, or may be delayed in doing so.
We have based our estimates on assumptions that may prove to be wrong, and we may use our available capital resources sooner than we currently expect. Because of the numerous risks and uncertainties associated with the development and commercialization of our product candidates, we are unable to estimate the amounts of increased capital outlays and operating expenditures necessary to complete the development of our product candidates.
Our future capital requirements will depend on many factors, including:
| • | the progress and results of our ADAPT trial and the ongoing and planned investigator-initiated clinical trials of AGS-003 that we support or plan to support; |
| • | the progress and results of the ongoing investigator-initiated Phase 2 clinical trial of AGS-004 for HIV eradication and the planned investigator-initiated clinical trials of AGS-004 that we support and our ability to obtain additional funding under our NIH and NIAID contract for our AGS-004 program; |
| 91 |
| • | the scope, progress, results and costs of preclinical development, laboratory testing and clinical trials for our other product candidates; |
| • | the purchase price for, and the costs and timing of our planned purchase, build-out and equipping of a new commercial manufacturing facility, the costs and timing of building out and equipping a new commercial manufacturing facility at our current facility or at a new leased facility, or, the costs and timing of entering into a commercial supply arrangement with a contract manufacturer, and any costs and liabilities associated with any financing arrangements entered into to fund the costs of these activities; |
| • | the costs, timing and outcome of regulatory review of our product candidates; |
| • | the costs of commercialization activities, including product sales, marketing, manufacturing and distribution, for any of our product candidates for which we receive regulatory approval; |
| • | the potential need to repay the $8.0 million remaining outstanding under the loan under our license agreement with Medinet Co. Ltd. and its wholly-owned subsidiary, MEDcell Co., Ltd, which we refer to together as Medinet; |
| • | revenue, if any, received from commercial sales of our product candidates, should any of our product candidates be approved by the FDA or a similar regulatory authority outside the United States; |
| • | the costs of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending intellectual property-related claims; |
| • | the extent to which we acquire or invest in other businesses, products and technologies; |
| • | our ability to obtain government or other third party funding for the development of our product candidates; and |
| • | our ability to establish collaborations on favorable terms, if at all, particularly arrangements to develop, market and distribute AGS-003 outside North America and arrangements for the development and commercialization of our non-oncology product candidates, including AGS-004. |
Until such time, if ever, as we can generate substantial product revenue, we expect to finance our cash needs through a combination of equity offerings, debt financings, government contracts, government and other third party grants or other third party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. To the extent that we raise additional capital through the sale of equity or convertible debt securities, stockholder ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect stockholder rights. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through government or other third party funding, marketing and distribution arrangements or other collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to us.
We are seeking government or other third party funding for the continued development of AGS-004. In January 2014, CARE agreed that it would fund all patient clinical costs of Stage 1 of our Phase 2 adult eradication clinical trial of AGS-004, except for the associated manufacturing costs for which we will be responsible. NIAID’s Division of AIDS has approved $6.6 million in funding for Stage 2 of this Phase 2 clinical trial to be provided directly to the University of North Carolina. If we are unable to raise additional government or other third party funding when needed, we may be required to delay, limit, reduce or terminate our development of AGS-004 or to grant rights to develop and market AGS-004 that we would otherwise prefer to keep for ourselves.
| 92 |
Contractual Obligations and Commitments
The following table summarizes our significant contractual obligations and commercial commitments as of December 31, 2015 and the effects such obligations are expected to have on our liquidity and cash flows in future periods (in thousands):
| Payments Due by Period | ||||||||||||||||||||
| Total | Less Than 1 Year | 1-3 Years | 3-5 Years | More Than 5 Years | ||||||||||||||||
| Operating leases for existing facilities and equipment | $ | 515 | $ | 435 | $ | 80 | $ | — | $ | — | ||||||||||
| Facility lease obligation for new facility | 5,954 | 569 | 1,178 | 1,231 | 2,976 | |||||||||||||||
| Notes payable to Horizon Technology and Fortress Credit | 25,000 | 1,563 | 22,396 | 1,041 | — | |||||||||||||||
| Interest on notes payable to Horizon Technology and Fortress Credit | 4,691 | 2,339 | 2,340 | 12 | — | |||||||||||||||
| Final payment to Horizon Technology and Fortress Credit | 1,250 | — | 625 | 625 | — | |||||||||||||||
| Note payable to Medinet, including interest | 9,249 | — | 9,249 | — | — | |||||||||||||||
| Other notes payable, including interest | 52 | 17 | 35 | — | — | |||||||||||||||
| Amount due under development agreement with Invetech | 7,777 | — | 7,777 | — | — | |||||||||||||||
| Interest on development agreement with Invetech | 930 | — | 930 | — | — | |||||||||||||||
| Purchase obligation with Invetech, including interest | 25,000 | — | 25,000 | — | — | |||||||||||||||
| Purchase obligation with Saint-Gobain | 4,700 | 4,700 | — | — | — | |||||||||||||||
| Total | $ | 85,118 | $ | 9,623 | $ | 69,610 | $ | 2,909 | $ | 2,976 | ||||||||||
Under the Lease Agreement with TKC, we agreed to lease certain land and an approximately 124,000 square-foot building to be constructed on approximately 11 acres in Durham County, North Carolina. This facility is being built to house our corporate headquarters and primary manufacturing facility. The shell of the new facility was constructed on a build-to-suit basis by TKC in accordance with agreed upon specifications and plans as set forth in the Lease Agreement, at the expense of TKC, other than those costs resulting from changes requested by us for which we have paid $1.7 million as of December 31, 2015, as further described below.
The term of the Lease Agreement is 10 years from the commencement date of July 1, 2015. We have an option to extend the Lease Agreement by six five-year renewal terms. Initial rent is $46,917 per month, subject to certain fixed increases over the course of the term as set forth in the Lease Agreement and to adjustment based on our use of certain amounts allocated for upfitting the interior of the facility.
In February 2015, we entered into a Purchase and Sale Agreement, or the Purchase Agreement, with TKC which represented our exercise of the purchase option under the Lease Agreement. The purchase price to be paid by us is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by us and the amount of any improvement allowances advanced to us by TKC prior to the closing. Under the terms of the Purchase and Sale Agreement, we had until February 16, 2016 to consummate the purchase of the property. We are currently in discussions with TKC to extend the period for purchasing the property as we will need to obtain additional funding to consummate the purchase of the property. If we purchase the property, upon the closing, the lease agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to us. Our payment of the purchase price and the termination of the Lease Agreement are not reflected in the table above.
In September 2014, we entered into the Loan Agreement with the Lenders under which we could borrow up to $25.0 million in two tranches of $12.5 million each. We borrowed the first tranche of $12.5 million upon the closing of the transaction in September 2014 and the second tranche of $12.5 million in August 2015. See “Liquidity and Capital Resources – Sources of Liquidity” for additional information regarding the Loan Agreement.
| 93 |
In October 2014, we entered into a development agreement with Invetech. The development agreement supersedes and replaces the development agreement entered into by the parties in July 2005. Under the development agreement, Invetech agreed to continue to develop and provide prototypes of the automated production system to be used for the manufacture of our Arcelis-based products, or the Production Systems. Development services will be performed on a proposal by proposal basis. Invetech has agreed to defer 30% of its fees, but such deferral may not exceed $5,000,000. Deferred fees (plus interest of 7% per annum) would become payable either, at our option, in a lump sum within 90 days of the “Sunset Date Trigger Event” or pursuant to an installment plan (either in four installments payable within the first year or eight installments payable within the first two years after the “Sunset Date Trigger Event”). The “Sunset Date Trigger Event” is December 31, 2016 or June 30, 2016 if, prior to such date, our ADAPT trial is closed early following the interim review at 50% or 75% of events, due to a positive efficacy outcome in favor of the active treatment arm that contains AGS-003. Invetech is entitled to a 10% bonus payment if this clinical trial is closed early indicating positive efficacy and Invetech has timely completed all activities up to the time of such early closure. We are planning to negotiate the terms of the development agreement related to the deferred fees and bonus payment.
The development agreement requires the parties to discuss in good faith Invetech’s supply of Production Systems for use in manufacturing commercial product. We have an obligation to purchase $25.0 million worth of Production Systems, components, subsystems and spare parts for commercial use. Once that obligation has been satisfied, we have the right to have a third party supply Production Systems for use in manufacturing commercial product, provided that Invetech has a right of first refusal with respect to any offer by a third party and we may not accept an offer from a third party unless that offer is at a price that is less than that offered by Invetech and otherwise under substantially the same or better terms. We will own all intellectual property arising from the development services (with the exception of existing Invetech intellectual property incorporated therein-under which we will have a license). The term of the development agreement will continue until the completion of the development of the Production Systems. The development agreement can be terminated early by either party because of a technical failure or by us without cause.
In January 2015, we entered into a development agreement with Saint-Gobain. Under the agreement, Saint-Gobain will develop a range of disposables for use in our automated production systems to be used for the manufacture of our Arcelis-based products, which we refer to as the Disposables. We do not expect the fees and expenses incurred under the Saint-Gobain Agreement to exceed $6,000,000. We made a payment of $400,000, and Saint-Gobain has agreed to defer one half of the fees and expenses incurred until the earlier of (i) the date upon which we have raised an additional $60.0 million in capital, and (ii) September 30, 2016. The Saint-Gobain agreement required the parties to execute a commercial supply agreement under which Saint-Gobain would become the exclusive supplier of Disposables for the manufacture of our products treating solid tumors for no less than fifteen years by December 31, 2015. The parties are in discussions to extend this date and to finalize the commercial supply agreement. The Saint-Gobain agreement will continue until December 31, 2016, but can be terminated earlier by written agreement of the parties because of a material default, including the failure to execute the commercial supply agreement, or a failure to achieve a performance milestone.
We are a party to license agreements with universities and other third parties, as well as patent assignment agreements, under which we have obtained rights to patents, patent applications and know-how. Under these agreements, we have agreed to pay the other parties milestone payments upon the achievement of specified clinical, regulatory and commercialization events and royalties based on future sales of products. We have not included these payments in the table as we cannot estimate if, when or in what amounts such payments will become due under these agreements.
Net Operating Losses
As of December 31, 2015, we had U.S. federal and state, and Canadian federal and provincial net operating loss carryforwards of approximately $199,443,200, $244,608,100, $5,479,100, and $5,479,100, respectively. These net operating loss carryforwards begin to expire in 2018, 2017, 2016 and 2016, respectively. As of December 31, 2015, we had U.S. federal and state tax credit carryforwards of approximately $9,319,400 and $340,400, respectively. These credit carryforwards begin to expire in 2020 and 2024, respectively. As of December 31, 2015, we had Canadian investment tax credit carryforwards of approximately $29,400 that begin to expire in 2024. The utilization of the net operating loss and tax credit carryforwards may be subject to limitation under the rules regarding a change in stock ownership as determined by the Internal Revenue Code, and state and foreign tax laws. Section 382 of the Internal Revenue Code of 1986, as amended, imposes annual limitations on the utilization of net operating loss carryforwards, other tax carryforwards, and certain built-in losses upon an ownership change as defined under that section. In general terms, an ownership change may result from transactions that increase the aggregate ownership of certain of our stockholders by more than 50 percentage points over a three year testing period. If we have undergone a Section 382 ownership change, an annual limitation would be imposed on certain of our tax attributes, including NOL and capital loss carryforwards, and certain other losses, credits, deductions or tax basis. As of December 31, 2015, we have not completed a formal study to determine whether there are 382 limitations that apply.
As of December 31, 2015, we have received $2.9 million in refunds through scientific research and experimental development tax credits through our consolidated subsidiary in Canada.
Off-Balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements as defined under Securities and Exchange Commission, or SEC, rules.
| 94 |
Item 7A. Quantitative and Qualitative Disclosures about Market Risk
Our primary exposure to market risk is limited to our cash, cash equivalents and short-term investments, all of which have maturities of one year or less. The related interest income sensitivity is affected by changes in the general level of short-term U.S. interest rates. We primarily invest in high quality, short-term marketable debt securities issued by high quality financial and industrial companies.
Due to the short-term duration and low risk profile of our cash, cash equivalents and short-term investments, an immediate 10.0% change in interest rates would not have a material effect on the fair value of our portfolio. Accordingly, we would not expect our operating results or cash flows to be affected to any significant degree by the effect of a sudden change in market interest rates on our cash, cash equivalents and short-term investments.
We do not believe that our cash, cash equivalents and short-term investments have significant risk of default or illiquidity. While we believe our cash, cash equivalents and short-term investments do not contain excessive risk, we cannot provide absolute assurance that in the future our investments will not be subject to adverse changes in fair value. In addition, we maintain significant amounts of cash and cash equivalents at one or more financial institutions that are in excess of federally insured limits.
We are also exposed to market risk from the potential increase in short-term U.S. interest rates under our Loan Agreement with Horizon and Fortress. The per annum interest rate for each tranche is a floating rate equal to 9.25% plus the amount by which the one-month LIBOR exceeds 0.50% (effectively a floating rate equal to 8.75% plus the one-month LIBOR Rate). The total per annum interest rate shall not exceed 10.75%. Since the one month LIBOR Rate has been less than 0.50% since the Loan Agreement was executed in September 2014, our interest rate has been and is currently 9.25%. If the one month LIBOR Rate increases to 2.0% or greater, the interest expense we will incur under the Loan Agreement will increase by $375,000 per year.
All of our other debt instruments and liabilities that incur interest charges do so at fixed-rates. We incur interest expense at fixed rates under the unsecured promissory note payable to Medinet (3% per annum), the manufacturing research and development obligations payable to Invetech (7% per annum), and other notes payable (8.31% per annum).
Item 8. Financial Statements and Supplementary Data.
Our financial statements and the financial statement schedule required by this item, together with the report of our independent registered public accounting firm and the notes to our financial statements, appear on pages F-1 through F-32 of this Annual Report on Form 10-K and are incorporated herein by reference.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
There has been no change of accountants nor any disagreements with accountants on any matter of accounting principles or practices or financial disclosure required to be reported under this Item.
| 95 |
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Under the supervision of and with the participation of our management, including our chief executive officer, who is our principal executive officer, and our vice president of finance, who is our principal financial officer, we conducted an evaluation of the effectiveness of our disclosure controls and procedures as of December 31, 2015, the end of the period covered by this Annual Report. The term "disclosure controls and procedures," as set forth in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934, as amended, or the Exchange Act, means controls and other procedures of a company that are designed to provide reasonable assurance that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the rules and forms promulgated by the Securities and Exchange Commission (the “SEC”). Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to the company's management, including its principal executive and principal financial officers, as appropriate to allow timely decisions regarding required disclosure. Management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives, and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on the evaluation of our disclosure controls and procedures as of December 31, 2015, our chief executive officer and chief financial officer concluded that, as of such date, our disclosure controls and procedures were effective at the reasonable assurance level.
Changes in Internal Control over Financial Reporting
There was no change in our internal control over financial reporting identified in connection with the evaluation required by Rule 13a-15(d) and 15d-15(d) of the Exchange Act that occurred during the quarter ended December 31, 2015 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
Management’s Report on Internal Control Over Financial Reporting and Attestation Report of the Registered Public Accounting Firm
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on our evaluation under this framework, our management concluded that our internal control over financial reporting was effective as of December 31, 2015.
This Annual Report on Form 10-K does not include an attestation report of our independent registered public accounting firm due to a transition period established by the rules of the SEC for newly public companies.
None.
| 96 |
Item 10. Directors, Executive Officers and Corporate Governance
MANAGEMENT
The following table sets forth the name, age and position of each of our executive officers and directors as of March 23, 2016.
|
Name |
Age |
Position |
| Jeffrey D. Abbey | 54 | President, Chief Executive Officer and Director |
| Charles A. Nicolette, Ph.D. | 53 | Chief Scientific Officer and Vice President of Research and Development |
| Frederick M. Miesowicz, Ph.D. | 67 | Chief Operating Officer and Vice President of Manufacturing |
| Lee F. Allen, M.D., Ph.D. | 64 | Chief Medical Officer |
| Lori R. Harrelson | 46 | Vice President of Finance |
| Joan C. Winterbottom | 59 | Chief Human Resources Officer |
| Hubert Birner, Ph.D.(1)(3) | 49 | Chairman of the Board of Directors |
| Robert F. Carey(1) | 57 | Director |
| Andrei Petrov(2)(3) | 42 | Director |
| Sander van Deventer M.D., Ph.D. | 61 | Director |
| Philippe Van Holle(1)(2) | 61 | Director |
| Alexey Vinogradov, Ph.D. | 42 | Director |
______________
(1) Member of the Audit Committee
(2) Member of the Compensation Committee
(3) Member of the Nominating and Corporate Governance Committee
Jeffrey D. Abbey has served as our President and Chief Executive Officer and a member of our board of directors since February 2010. Mr. Abbey served in various other positions at our company from September 2002 to February 2010, including as our Vice President of Business Development from February 2004 to January 2009 and as our Chief Business Officer from January 2009 to February 2010. Prior to joining us, Mr. Abbey served as Vice President of Business Development and Finance at Internet Appliance Network, an information technology company, from 1999 to 2001. Mr. Abbey was a partner at Eilenberg and Krause, LLP, a corporate law firm, from 1994 to 1999. Mr. Abbey received an A.B. in mathematical economics from Brown University and an M.B.A. and J.D. from the University of Virginia. We believe that Mr. Abbey is qualified to serve on our board of directors due to his extensive knowledge of our company and our industry.
Charles A. Nicolette, Ph.D. has served as our Chief Scientific Officer since December 2007 and as our Vice President of Research and Development since December 2004. Dr. Nicolette served as our Vice President of Research from July 2003 to December 2004. Prior to joining us, Dr. Nicolette served in various positions at Genzyme Molecular Oncology, Inc., a biotechnology company, from 1997 to 2003, most recently as Director of Antigen Discovery. Dr. Nicolette received a B.S. from the State University of New York at Stony Brook and a Ph.D. in biochemistry and cellular and developmental biology from the State University of New York at Stony Brook, completing his doctoral dissertation and post-doctoral fellowship at Cold Spring Harbor Laboratory.
Frederick M. Miesowicz, Ph.D. has served as our Chief Operating Officer and Vice President of Manufacturing since February 2005. Dr. Miesowicz served as our Vice President of Manufacturing from May 2003 to February 2005. Prior to joining us, Dr. Miesowicz served as Vice President of U.S. Operations for Gamida-Cell Ltd., a stem cell company, from 2000 to 2003; Senior Vice President and General Manager at Hybridon Specialty Products, a manufacturing division of a biotechnology company, from 1998 to 2000; and Vice President and General Manager at Cellcor, a subsidiary of Cytogen Corporation, a biopharmaceutical company, from 1995 to 1998. Dr. Miesowicz received a B.S. in chemistry from Siena College and a Ph.D. in chemistry from Harvard University.
Lee F. Allen, M.D., Ph.D. has served as our Chief Medical Officer since January 2016. Prior to joining us, Dr. Allen served as Chief Medical Officer for Spectrum Pharmaceuticals from April 2013 to January 2016. Dr. Allen also served as Chief Medical Officer at AMAG Pharmaceuticals from August 2007 to March 2013, and was Executive Vice President of Medical Development from 2009 to 2013 and Senior Vice President of Medical Development from 2007 to 2009. Dr. Allen has also served in clinical leadership roles in the Division of Hematology/Oncology at the University of Utah Health Sciences’ Huntsman Cancer Institute and Duke University Medical Center. He earned a Ph.D. in pathology and an M.D. from the University of Medicine and Dentistry of New Jersey, and trained in Internal Medicine and Hematology/Oncology at the Duke University Medical Center.
| 97 |
Lori R. Harrelson has served as our Vice President of Finance since July 2011. Ms. Harrelson served as our Director of Finance and Accounting from January 2007 to July 2011 and as our Director of Accounting and Financial Reporting from September 2004 to January 2007. Prior to joining us, Ms. Harrelson served as Finance Manager at LipoScience, Inc., a diagnostic company, from 2001 to 2004 and a senior auditor at Ernst & Young, from 1997 to 2001. Ms. Harrelson received a B.S. in finance from East Carolina University and is a C.P.A.
Joan C. Winterbottom has served as our Chief of Human Resources Officer since February 2015. From August 2012 to February 2014, she served as Senior Vice President, Human Resources at Amicus Therapeutics, Inc. From August 2011 to August 2012, she served as Senior Vice President, Human Resources at Savient Pharmaceuticals, Inc. Prior to joining Savient, Ms. Winterbottom held a variety of human resources leadership roles at Johnson & Johnson from 2001 to 2011, most recently as Vice President of Human Resources for the Global Over-the-Counter/Nutritionals/Wellness and Prevention global business unit. During her time at Johnson & Johnson, she also served as Vice President of Human Resources for McNeil Consumer Healthcare and World Wide Director, Head of Human Resources for Biologics, Immunology, and Oncology Research & Development. Earlier in her career, Ms. Winterbottom held human resources positions of increasing responsibility in various financial services companies. Ms. Winterbottom earned a B.S. in business and economics from Lehigh University, and a graduate certification in organization development from Saint Joseph's University.
Hubert Birner, Ph.D. has served as chairman of our board of directors since 2005 and a member of our board of directors since 2001. Dr. Birner joined the Munich office of TVM Capital, a venture capital firm and affiliate of Argos, as an investment manager in 2000 and currently serves as the Managing Partner of the firm. From 1998 to 2000, Dr. Birner served as head of European business development and director of marketing for Germany at Zeneca Agrochemicals, a biopharmaceutical company. Prior to joining Zeneca Agrochemicals, Dr. Birner served as a management consultant in McKinsey & Company’s European healthcare and pharmaceutical practice. Dr. Birner currently serves on the board of directors of Proteon Therapeutics, Inc. and SpePharm Holdings BV. Dr. Birner previously served on the board of directors of Horizon Pharma, Inc., Bioxell SA, Evotec AG, Probiodrug AG and Jerini AG. Dr. Birner received an M.B.A. from Harvard Business School and a doctorate in biochemistry from Ludwig-Maximilians University in Munich, Germany. His doctoral thesis was honored with the Hoffmann-La Roche prize for outstanding basic research in metabolic diseases. We believe that Dr. Birner is qualified to serve as chairman of our board of directors due to his extensive experience with biopharmaceutical companies and his years of experience providing strategic and advisory services to pharmaceutical and biotechnology companies as a lead director and investor.
Robert F. Carey has served as a member of our board of directors since September 2015. Mr. Carey has been executive vice president, chief business officer for Horizon Pharma plc since March 2014. Prior to that, Mr. Carey served as managing director and head of the healthcare investment banking group at JMP Securities LLC, a full-service investment bank from March 2003 to March 2014. Prior to JMP, Mr. Carey was a managing director in the healthcare groups at Dresdner Kleinwort Wasserstein and Vector Securities International, Inc.. Mr. Carey also has held roles at Red Hen Bread, InStadium, Shearson Lehman Hutton and Ernst & Whinney. Mr. Carey received his B.S. in accounting from the University of Notre Dame. We believe that Mr. Carey is qualified to serve on our board of directors due to his valuable and relevant healthcare investment banking experience with financings, mergers, acquisitions and global expansion and other strategic transactions as well as his role as a CPA supporting the audits of public and private corporations, which we expect will assist Mr. Carey in fulfilling his duties as chair of our audit committee.
Andrei Petrov, Ph.D. has served as a member of our board of directors since August 2013. Dr. Petrov has been the Chief Scientific Officer of International Biotechnology at Center Generium, a private scientific research and drug development company, since 2011, and the Chief Executive Officer of CJSC ‘Kollectsiya,’ a venture investment company, since 2013. From 2008 to 2011, Dr. Petrov served as Senior Scientist at CJSC Masterclone, a drug discovery and development company. Dr. Petrov has also served as a member on the board of directors of Affitech A/S since 2010, as a member on the board of directors of co.don AG from 2012 to 2015 and as a member on the board of directors of EnGene Inc. since 2015. We believe that Dr. Petrov is qualified to serve on our board of directors due to his extensive experience in drug discovery and development, international collaboration and co-development as well his business development skills in mergers, acquisitions and licensing deals.
Sander van Deventer, M.D., Ph.D. has served as a member of our board of directors since 2001. Dr. van Deventer has been a General Partner of Forbion Capital Partners (formerly ABN AMRO Capital), an affiliate of Argos, since 2006. From 2008 to 2009, he served as the Chief Executive Officer of Amsterdam Molecular Therapeutics, or AMT, a gene therapy company that he co-founded in 1998. He has also served as a member of AMT’s board of directors since 2007 and as a member of the board of directors of UniQure since February 2014. Dr. van Deventer has also served as a Professor of Translational Gastroenterology at Leiden University since 2008. He received an M.D. and Ph.D. from the University of Amsterdam. We believe that Dr. van Deventer is qualified to serve on our board of directors due to his experience as a founder of a biopharmaceutical company and his expertise in clinical development.
| 98 |
Philippe Van Holle has served as a member of our board of directors since November 2014. Mr. Van Holle held the position of Senior VP Global Human Resources at Celgene Corporate HQ in Summit, New Jersey from January 2013 to October 2014. From January 2012 until December 2012, he held the position of Chairman International in Celgene. He was President EMEA since November 2008, after building the European organization as Head of Europe since January 2006. Mr. Van Holle served as Vice President Northern Europe for the Genzyme Corporation from September 2001 to December 2005. Previously, he had been Vice President Global Marketing at Baxter International, for the Renal Division, which he joined in 1998. He was Vice President Marketing, Amgen Europe from 1989 to 1997. He received his undergraduate degree in Econometrics at the Antwerp University St. Ignatius, Belgium in 1976 and a M.B.A from the Cornell University. We believe that Mr. Van Holle is qualified to serve on our board of directors due to his experience in the biopharmaceutical industry and as a senior executive.
Alexey Vinogradov, Ph.D. has served as a member of our board of directors since February 2014. Dr. Vinogradov has served as a Managing Partner of CJSC Kollektsiya, or Inbio Ventures, a management company for Pharmstandard International, S.A., an affiliate of Argos, since 2012. Prior to joining Inbio Ventures, from 2009 to 2012, Dr. Vinogradov served as Investment Manager at Bioprocess Capital Partners, Russia’s first venture capital fund, specializing in life sciences and drug discovery. From 2004 to 2009, Dr. Vinogradov was employed by the International Science and Technology Center, where he provided consulting and investment support to early-stage biotechnology companies. From 2002 to 2004, Dr. Vinogradov was employed by Core-Biotech, a private company specialized in industrial biotechnology. Dr. Vinogradov received a Ph.D. in biochemistry from Moscow State University and completed a post-doctoral fellowship at Wageningen University (the Netherlands). We believe that Dr. Vinogradov is qualified to serve on our board of directors due to his experience in the venture capital and biopharmaceutical industries and his scientific background.
Board Composition and Election of Directors
Our board of directors is currently authorized to have up to seven members. In accordance with the terms of our certificate of incorporation and bylaws, our board of directors is divided into three classes, class I, class II and class III, with members of each class serving staggered three-year terms. The members of the classes are divided as follows:
| • | the class I directors are Sander van Deventer, M.D., Ph.D. and Alexey Vinogradov, Ph.D., and their term expires at our annual meeting of stockholders to be held in 2018; |
| • | the class II directors are Hubert Birner, Ph.D. and Robert F. Carey, and their term expires at our annual meeting of stockholders to be held in 2016; and |
| • | the class III directors are Jeffrey D. Abbey, Andrei Petrov and Philippe Van Holle, and their term expires at our annual meeting of stockholders to be held in 2017. |
Upon the expiration of the term of a class of directors, directors in that class will be eligible to be elected for a new three-year term at the annual meeting of stockholders in the year in which their term expires. In accordance with the terms of our certificate of incorporation and bylaws, our directors are only be able to be removed for cause by the affirmative vote of the holders of 75% or more of our voting stock.
Applicable NASDAQ rules require a majority of a listed company’s board of directors to be comprised of independent directors. In addition, NASDAQ rules require that, subject to specified exceptions, each member of a listed company’s audit, compensation and nominating and corporate governance committees be independent and that audit committee members also satisfy independence criteria set forth in Rule 10A-3 under the Securities Exchange Act of 1934, as amended, or the Exchange Act, and that compensation committee members satisfy independence criteria set forth in Rule 10C-1 under the Exchange Act. Under applicable NASDAQ rules, a director only qualifies as an “independent director” if, in the opinion of our board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. In order to be considered independent for purposes of Rule 10A-3, a member of an audit committee of a listed company may not, other than in his or her capacity as a member of the audit committee, the board of directors, or any other board committee, accept, directly or indirectly, any consulting, advisory, or other compensatory fee from the listed company or any of its subsidiaries or otherwise be an affiliated person of the listed company or any of its subsidiaries. In addition, in affirmatively determining the independence of any director who will serve on a listed company’s compensation committee, Rule 10C-1 under the Exchange Act requires that a company’s board of directors must consider all factors specifically relevant to determining whether a director has a relationship to such company which is material to that director’s ability to be independent from management in connection with the duties of a compensation committee member, including: the source of compensation to the director, including any consulting, advisory or other compensatory fee paid by such company to the director, and whether the director is affiliated with the company or any of its subsidiaries or affiliates.
Our board of directors has determined that all of our directors, other than Mr. Abbey, are independent directors as defined by applicable NASDAQ rules. In making such determinations, our board of directors considered the relationships that each such non-employee director and director nominee has with our company and all other facts and circumstances that our board of directors deems relevant in determining their independence, including the beneficial ownership of our capital stock by each non-employee director and director nominee.
| 99 |
There are no family relationships among any of our directors or executive officers.
Board Leadership Structure
The positions of chairman of the board of directors and chief executive officer are presently separated and have generally been separated at our company. The duties of the chairman of the board include the following:
| • | chairing meetings of our board and of the independent directors in executive session; |
| • | meeting with any director who is not adequately performing his or her duties as a member of our board or any committees; |
| • | facilitating communications between other members of our board and the chief executive officer; |
| • | determining the frequency and length of board meetings and recommending when special meetings of our board should be held; |
| • | preparing or approving the agenda for each board meeting; and |
| • | reviewing and, if appropriate, recommending action to be taken with respect to written communications from stockholders submitted to our board. |
Our board of directors decided to separate the roles of chairman and chief executive officer because it believes that a bifurcated leadership structure offers the following benefits:
| • | increasing the independent oversight of our company and enhancing our board’s objective evaluation of our chief executive officer; |
| • | freeing the chief executive officer to focus on company operations instead of board administration; |
| • | providing the chief executive officer with an experienced sounding board; |
| • | providing greater opportunities for communication between stockholders and our board; |
| • | enhancing the independent and objective assessment of risk by our board; and |
| • | providing an independent spokesman for our company. |
Board Committees
Our board of directors has established an audit committee, a compensation committee and a nominating and corporate governance committee. Each of these committees operates under a charter that has been approved by our board.
Audit Committee
The current members of our audit committee are Hubert Birner, Ph.D., Robert F. Carey and Philippe Van Holle. Mr. Carey chairs our audit committee. Jean Lamarre served as a member of our audit committee in 2015 until September 2015 at which time he ceased to serve as a member of our board of directors.
Our audit committee’s responsibilities include:
| • | appointing, approving the compensation of, and assessing the independence of our registered public accounting firm; |
| • | overseeing the work of our independent registered public accounting firm, including through the receipt and consideration of reports from such firm; |
| • | reviewing and discussing with management and our independent registered public accounting firm our annual and quarterly financial statements and related disclosures; |
| • | monitoring our internal control over financial reporting, disclosure controls and procedures and code of business conduct and ethics; |
| • | overseeing our internal audit function, if any; |
| 100 |
| • | overseeing our risk assessment and risk management policies; |
| • | establishing policies regarding hiring employees from our independent registered public accounting firm and procedures for the receipt and retention of accounting related complaints and concerns; |
| • | meeting independently with our internal auditing staff, our independent registered public accounting firm and management; |
| • | reviewing and approving or ratifying any related person transactions; and |
| • | preparing the audit committee report required by SEC rules. |
All audit and non-audit services, other than de minimis non-audit services, to be provided to us by our independent registered public accounting firm must be approved in advance by our audit committee.
Our board of directors has determined that Mr. Carey is an “audit committee financial expert” as defined in applicable SEC rules and qualifies as independent for audit committee members as defined under applicable NASDAQ rules.
Compensation Committee
The current members of our compensation committee are Andrei Petrov and Philippe Van Holle. Brian J. Underdown served as chair of our compensation committee in 2015 and 2016 until March 14, 2016 at which time he ceased to serve as a member of our board of directors.
Our compensation committee’s responsibilities include:
| • | reviewing and approving, or making recommendations to our board with respect to, the compensation of our chief executive officer and our other executive officers; |
| • | overseeing an evaluation of our senior executives; |
| • | overseeing and administering our cash and equity incentive plans; |
| • | reviewing and making recommendations to our board with respect to director compensation; |
| • | reviewing and discussing annually with management our compensation disclosure required by SEC rules; and |
| • | preparing the annual compensation committee report required by SEC rules. |
Nominating and Corporate Governance Committee
The members of our nominating and corporate governance committee are Hubert Birner, Ph.D. and Andrei Petrov, Ph.D. Dr. Birner chairs our nominating and corporate governance committee. Brian J. Underdown served as chair of our nominating and governance committee in 2015 and 2016 until March 14, 2016 at which time he ceased to serve as a member of our board of directors. Our nominating and corporate governance committee’s responsibilities include:
| • | identifying individuals qualified to become members of our board; |
| • | recommending to our board the persons to be nominated for election as directors and to each of our board’s committees; |
| • | reviewing and making recommendations to our board with respect to our board leadership structure; |
| • | reviewing and making recommendations to our board with respect to management succession planning; |
| • | developing and recommending to our board corporate governance principles; and |
| • | overseeing a periodic evaluation of our board. |
Code of Ethics and Code of Conduct
On December 20, 2013, we adopted a written code of business conduct and ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, or persons performing similar functions. The code became effective on February 6, 2014. We have posted a current copy of the code on our website, www.argostherapeutics.com. In addition, we intend to post on our website all disclosures that are required by law or NASDAQ stock market listing standards concerning any amendments to, or waivers from, any provision of the code.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Exchange Act requires our directors, executive officers and holders of more than 10% of a registered class of our equity securities to file with the SEC initial reports of ownership of our equity securities on a Form 3 and reports of changes in such ownership on a Form 4 or Form 5. Based solely on our review of copies of such filings by our directors, executive officers, and 10% shareholders, or written representations from certain of those persons, we believe that all filings required to be made by those persons during fiscal 2015 were timely made, except for late filings by each of Messrs. Abbey and Nicolette and Mrs. Winterbottom. Each of these officers inadvertently filed a Form 4 reporting acquisitions of common stock after the deadline due to administrative errors.
| 101 |
Item 11. Executive Compensation
This section describes the material elements of compensation awarded to, earned by or paid to each of our named executive officers in 2015. This section also provides qualitative information regarding the manner and context in which compensation is awarded to and earned by our named executive officers and is intended to place into perspective the data presented in the tables and narrative that follow. Our “named executive officers” for 2015 were:
| • | Jeffrey D. Abbey, our president and chief executive officer; |
| • | Charles A. Nicolette, Ph.D., our vice president of research and development and chief scientific officer; and |
| • | Joan C. Winterbottom, our chief of human resources officer. |
Summary Compensation Table
The following table sets forth information regarding compensation awarded to, earned by or paid to our named executive officers during the years ended December 31, 2015 and 2014.
Name and Principal Position | Year | Salary | Bonus | Option | All
Other | Total ($) | ||||||||||||||||||
| Jeffrey D. Abbey(4) | 2015 | 450,000 | 202,500 | 714,502 | 6,911 | 1,373,913 | ||||||||||||||||||
| President and Chief Executive Officer | 2014 | 379,788 | 163,800 | 482,991 | 16,632 | 1,043,211 | ||||||||||||||||||
| Charles A Nicolette, Ph.D. | 2015 | 325,000 | 108,875 | 446,564 | 9,267 | 889,706 | ||||||||||||||||||
| Vice President of Research and Development and Chief Scientific Officer | 2014 | 294,327 | 88,200 | 160,902 | 8,117 | 551,546 | ||||||||||||||||||
| Joan C. Winterbottom(5) | 2015 | 247,244 | 73,270 | 561,589 | 27,070 | 909,173 | ||||||||||||||||||
| Chief of Human Resources Officer | 2014 | — | — | — | — | — | ||||||||||||||||||
____________________
| (1) | In lieu of paying annual cash bonuses for 2015, in January 2016 we granted restricted stock awards to our executive officers, including 90,401 shares to Mr. Abbey, 48,604 shares to Dr. Nicolette and 32,709 shares to Ms. Winterbottom. The number of shares granted to each named executive officer was calculated by dividing the amount of the 2015 annual cash bonus that would otherwise have been paid by the closing price of our common stock on January 8, 2016. Each of the restricted stock awards is subject to a lapsing right of repurchase in our favor, which right will lapse with respect to 100% of the underlying shares of each award on November 20, 2016, assuming such executive is still providing services to us on such date. |
| (2) | The amounts reported in the “Option Awards” column reflect the aggregate fair value of share-based compensation awarded during the year computed in accordance with the provisions of Financial Accounting Standards Board Accounting Standard Codification, or ASC, Topic 718. See Notes 2 and 12 to our consolidated financial statements appearing in “Item 8. Financial Statements and Supplementary Data” regarding assumptions underlying the valuation of equity awards. |
| (3) | The amounts reported in the “All Other Compensation” column reflect, for each named executive officer, the sum of the incremental cost to us of all perquisites and other personal benefits and are comprised of post-tax insurance earnings. The amount reported in this column for Ms. Winterbottom for 2015 also includes $24,154 in temporary living expenses paid on behalf of Ms. Winterbottom. |
| (4) | Mr. Abbey serves as a member of our board of directors but does not receive any additional compensation for his service as a director. |
| (5) | Ms. Winterbottom was appointed as our chief of human resources officer in February 2015. |
Narrative Disclosure to Summary Compensation Table
The primary elements of our executive compensation program are:
| • | base salary; |
| • | annual cash bonuses; and |
| • | equity incentive awards. |
| 102 |
We strive to achieve an appropriate mix between the various elements of our compensation program to meet our compensation objectives and philosophy; however, we have not adopted any formal policies or guidelines for allocating compensation among these elements.
Base Salary. We use base salaries to recognize the experience, skills, knowledge and responsibilities required of all our employees, including our named executive officers. None of our named executive officers is currently party to an employment agreement or other agreement or arrangement that provides for automatic or scheduled increases in base salary. In 2015, we paid an annual base salary of $450,000 to Mr. Abbey, $325,000 to Dr. Nicolette and $290,000 to Ms. Winterbottom. These annual base salaries have not been increased for 2016.
Annual Bonus. In addition to base salaries, our executive officers are eligible to receive annual discretionary cash bonuses based on the achievement of corporate objectives and individual performance. Bonuses are typically prorated on a monthly basis, as applicable, for executive officers who commence employment after the beginning of the year. Our executive officers’ annual bonus opportunities are generally set as a specified percentage of annual base salary. The 2015 annual target bonus amount for each of our current executive officers was 60% of base salary for Mr. Abbey, 40% of base salary for Dr. Nicolette and 35% of base salary for Ms. Winterbottom. In determining annual bonuses, we have generally attributed 85% of the target bonus to the achievement of specified corporate objectives and 15% to the individual’s effectiveness in helping us achieve our corporate objectives or other individual performance criteria. The annual corporate objectives are recommended by our chief executive officer and approved by the compensation committee and the board of directors. Historically, annual bonuses have been determined by the compensation committee and ratified by the non-employee directors in December of each year and paid by the end of December of the year in which they were determined. Due to the limitations on our cash resources, in lieu of annual cash bonuses to our named executive officers for 2015, we granted to each of our named executive officers restricted stock awards under our 2014 stock incentive plan having a value based on the closing market price on January 8, 2016 equal to $202,500 (90,401 shares) to Mr. Abbey, $108,875 (48,604 shares) to Dr. Nicolette and $73,270 (32,709 shares) to Ms. Winterbottom. Each of the restricted stock awards is subject to a lapsing right of repurchase in our favor, which will lapse with respect to 100% of the underlying shares on November 20, 2016, assuming such executive is still providing services to us on such date.
Equity Incentive Awards. Our equity award program is the primary vehicle for offering long-term incentives to our executives. We believe that equity awards provide our executives with a strong link to our long-term performance, create an ownership culture and help to align the interests of our executives and our stockholders. To date, we have used stock option grants for this purpose because we believe they are an effective means by which to align the long-term interests of our executive officers with those of our stockholders. The use of options also can provide tax and other advantages to our executive officers relative to other forms of equity compensation. We believe that our equity awards are an important retention tool for our executive officers, as well as for our other employees.
We award stock options broadly to our employees, including to our non-executive employees. Grants to our executives and other employees are made at the discretion of our board of directors and are not made at any specific time period during a fiscal year. All of our named executive officers have received stock option grants under our 2008 stock incentive plan or our 2014 stock incentive plan, each of which is described below. No further options may be granted under the 2008 plan.
Initial option grants to our executive officers are generally set forth in their employment agreements. These initial grants are the product of negotiation with the executive officer, but we generally seek to establish equity ownership levels that we believe are commensurate with the equity stakes held by executive officers serving in similar roles at comparable biopharmaceutical companies. In addition, from time to time in connection with corporate finance transactions and at other times as our compensation committee and board of directors deem appropriate, we provide subsequent option grants to those executive officers determined to be performing well.
Initial option grants to our executive officers are generally set forth in their employment agreements. These initial grants are the product of negotiation with the executive officer, but we generally seek to establish equity ownership levels that we believe are commensurate with the equity stakes held by executive officers serving in similar roles at comparable biopharmaceutical companies. In addition, from time to time in connection with corporate finance transactions and at other times as our compensation committee and board of directors deem appropriate, we provide subsequent option grants to those executive officers determined to be performing well.
The majority of the stock option grants we have made to our executive officers vest over four years. However, from time to time, our board of directors has approved grants with different and sometimes shorter vesting provisions. Our historical practice has been to provide for 100% acceleration of vesting of outstanding stock options in the event of a change of control. Additional information regarding the effect of accelerated vesting upon a change in control with respect to our named executive officers is discussed below under “— Agreements with our Named Executive Officers.”
| 103 |
Outstanding Equity Awards at 2015 Fiscal Year End
The following table provides information about outstanding stock options held by each of our named executive officers at December 31, 2015. All of the listed options were granted under our 2014 and 2008 stock incentive plans.
| Name | Number of Securities Underlying Unexercised Options (#) Exercisable | Number of Securities Underlying Unexercised Options (#) Unexercisable | Option Exercise Price ($) | Option Expiration Date | ||||||||||
| Jeffrey D. Abbey | 14,175 | (1) | — | 4.20 | (8) | 7/2/18 | ||||||||
| 5,706 | (2) | — | 4.20 | (8) | 12/5/18 | |||||||||
| 50,425 | (3) | — | 4.20 | (8) | 12/10/20 | |||||||||
| 63,452 | (4) | — | 4.20 | (8) | 4/10/22 | |||||||||
| 45,602 | (5) | — | 4.20 | (8) | 12/11/22 | |||||||||
| 201,102 | (6) | 185,016 | (6) | 5.82 | 11/1/23 | |||||||||
| 33,376 | (7) | 30,706 | (7) | 5.82 | 11/11/23 | |||||||||
| 20,683 | (9) | 37,717 | (9) | 6.09 | 7/27/24 | |||||||||
| — | (10) | 87,600 | (10) | 6.09 | 7/27/24 | |||||||||
| — | (11) | 120,000 | (11) | 7.80 | 6/17/25 | |||||||||
| Charles A. Nicolette, Ph.D. | 14,640 | (1) | — | 4.20 | (8) | 7/2/18 | ||||||||
| 5,893 | (2) | — | 4.20 | (8) | 12/5/18 | |||||||||
| 14,619 | (3) | — | 4.20 | (8) | 12/10/20 | |||||||||
| 31,726 | (4) | — | 4.20 | (8) | 4/10/22 | |||||||||
| 22,801 | (5) | — | 4.20 | (8) | 12/11/22 | |||||||||
| 84,189 | (6) | 77,454 | (6) | 5.82 | 11/1/23 | |||||||||
| 14,833 | (7) | 13,648 | (7) | 5.82 | 11/11/23 | |||||||||
| 6,375 | (9) | 11,625 | (9) | 6.09 | 7/27/24 | |||||||||
| — | 27,000 | (10) | 6.09 | 7/27/24 | ||||||||||
| — | 75,000 | (11) | 7.80 | 6/17/25 | ||||||||||
| Joan C. Winterbottom | — | 65,000 | (12) | 9.01 | 2/25/25 | |||||||||
| — | 20,000 | (13) | 6.21 | 9/18/25 | ||||||||||
| 104 |
___________________
| (1) | This option was granted on July 2, 2008 and vested as to 50% of the shares on the date of grant, and as to the remaining 50% of the shares in equal amounts monthly over the two year period commencing on April 1, 2008. |
| (2) | This option was granted on December 5, 2008 and vested in specified increments over a two-year period ending on April 1, 2010. |
| (3) | This option was granted on December 10, 2010. This option vested in equal monthly installments over a four year period, with the first installment vesting on February 24, 2010, provided that recipient continued to provide services to us over such period. |
| (4) | This option was granted on April 10, 2012 and vested as to 1/3 of the shares on the date of grant, and as to the remaining 2/3 of the shares vesting in equal amounts monthly over the three year period commencing on April 10, 2012, provided that the recipient continued to provide services to us over such period. |
| (5) | This option was granted on December 11, 2012 and vested as to 50% of the shares on the date of grant, and as to the remaining 50% of the shares vesting in equal amounts monthly over the two year period commencing on October 31, 2012, provided that the recipient continued to provide services to us over such period. |
| (6) | This option was granted on November 1, 2013 and vested as to 25% on November 1, 2014, with the remaining 75% of the shares vesting in equal amounts monthly over the three year period commencing on November 1, 2014, provided that the recipient continues to provide services to us over such period. |
| (7) | This option was granted on November 11, 2013 and vested as to 25% on November 1, 2014, with the remaining 75% of the shares vesting in equal amounts monthly over the three year period commencing on November 1, 2014, provided that the recipient continues to provide services to us over such period. |
| (8) | In April 2012, our board of directors approved the repricing of stock options that had exercise prices between $10.86 and $36.66 per share, including this option, to the then estimated fair value of our common stock, determined to be an exercise price of $4.20 per share. |
| (9) | This option was granted on July 28, 2014 and vested as to 25% of the shares on July 1, 2015, with the remaining 75% of the shares vesting in equal amounts monthly over the three year period commencing on July 1, 2015, provided that the recipient continues to provide services to us over such period. |
| (10) | These options were granted on July 28, 2014 and vest based on the achievement of various individual performance and market targets at various dates through December 31, 2017, provided that the recipient continues to provide services to us through the individual target measurement date. |
| (11) | These options were granted on June 17, 2015 and will vest as to 25% of the shares on June 1, 2016, with the remaining 75% of the shares vesting in equal amounts monthly over the three year period commencing on June 1, 2016, provided that the recipient continues to provide services to us over such period. |
| (12) | This option was granted on February 25, 2015 and vested as to 25% of the shares on February 1, 2016, with the remaining 75% of the shares vesting in equal amounts monthly over the three year period commencing on February 1, 2016, provided that the recipient continues to provide services to us over such period. |
| (13) | This option was granted on September 18, 2015 and vests as to 25% of the shares on September 1, 2016, with the remaining 75% of the shares vesting in equal amounts monthly over the three year period commencing on September 1, 2016, provided that the recipient continues to provide services to us over such period. |
Agreements with our Named Executive Officers
We have entered into written employment agreements with each of our named executive officers. The agreements set forth the terms of the named executive officer’s compensation, including base salary, severance and an annual cash bonus opportunity. In addition, the agreements provide that the named executive officers are eligible to participate in company-sponsored benefit programs that are available generally to all of our employees. The agreements also subject our named executive officers to certain non-competition and non-solicitation restrictions. In connection with the commencement of their employment with us, our named executive officers executed our standard confidential information and invention assignment agreements.
Each named executive officer is eligible to receive an annual performance cash bonus under his or her employment agreement based on the achievement of corporate objectives and the named executive officer’s individual performance, which is determined by our board of directors in its sole discretion. The bonus opportunity is calculated as a percentage of the named executive officer’s then annual base salary. For the year ended December 31, 2015, the target annual bonus for each named executive officer was 60% for Mr. Abbey, 40% for Dr. Nicolette and 35% for Ms. Winterbottom. These target bonuses are also in effect for the year ending December 31, 2016. Each named executive officer must be employed on the date the bonus is paid in order to be eligible for and receive his or her annual bonus.
| 105 |
Potential Payments upon Termination or Change in Control
Upon execution and effectiveness of a separation agreement and release of all claims, each named executive officer is entitled to severance payments if his or her employment is terminated under specified circumstances pursuant to the terms of his or her employment agreement.
If we terminate Mr. Abbey’s or Dr. Nicolette’s employment without cause or if each such named executive officer terminates his employment with us for good reason in accordance with the terms of his employment agreement, he is entitled to receive from us an amount equal to nine months of his then annual base salary, payable in nine equal monthly installments in accordance with our payroll practices, and standard health insurance coverage for a period of nine months, subject to such benefits being available to non-employees. If we terminate Ms. Winterbottom’s employment without cause or if she terminates her employment with us for good reason in accordance with the terms of her employment agreement, she is entitled to receive from us an amount equal to six months of her then annual base salary, payable in six equal monthly installments in accordance with our payroll practices, and standard health insurance coverage for a period of six months, subject to such benefits being available to non-employees. If his or her standard health insurance coverage is not available to non-employees under our company sponsored plan, we will reimburse the named executive officer in an amount equal to the cost of the premium for coverage under a medical plan at the same level and on the same terms and conditions in place immediately before his or her termination.
If we terminate the named executive officer’s employment without cause or if the named executive officer terminates his or her employment with us for good reason in accordance with the terms of his or her employment agreement, in either case within 90 days before or six months after a “change in control event” as defined in our 2008 stock incentive plan, and such event also constitutes a “change in control event” within the meaning of the regulations promulgated under Section 409A of the Internal Revenue Code, as amended, or the Code, Mr. Abbey and Dr. Nicolette will be entitled to receive the payments and benefits specified above for a period of 15 months rather than nine months and Mrs. Winterbottom will be entitled to receive the payments and benefits for a period of six months. Additionally, in such circumstances, Mr. Abbey and Dr. Nicolette will each be entitled to receive an amount equal to 15 months of his target bonus for the year in which his employment terminates, payable in 15 equal monthly installments in accordance with our payroll practices and Mrs. Winterbottom will be entitled to six months of her target bonus payable in six monthly installments.
Under Mr. Abbey’s and Dr. Nicolette’s employment agreements, effective upon the closing of our IPO in February 2014 and for a period of four years from such date, to the extent that any payment, benefit or distribution, or combination thereof, by us or any of our affiliates to the executive officer pursuant to his employment agreement or any other agreement, plan or arrangement would constitute a “parachute payment” within the meaning of Section 280G of the Code and would be subject to the excise tax imposed under Section 4999 of the Code, Mr. Abbey and Dr. Nicolette, as applicable, will be entitled to receive a “gross-up” payment equal to the sum of such excise tax and related interest or penalties, plus the amount necessary to put him in the same after-tax position that he would have been in had he not incurred any tax liability under Section 4999 of the Code. After such four-year period, the applicable executive officer will not be entitled to any such “gross up” payment associated with any “parachute payment” or excise tax.
If required by Section 409A of the Code, the payments we are required to make to each named executive officer in the first six months following the termination of such named executive officer’s employment under such employment agreement will be made as a lump sum on the date that is six months and one day following such termination.
Under the terms of the stock options granted to our named executive officers prior to 2013 under our 2008 stock incentive plan, upon a “change of control event” as defined in our 2008 stock incentive plan, all unvested portions of any outstanding options held by them will vest in full. Under the terms of the stock options granted to our named executive officers in 2013 under our 2008 stock incentive plan, upon a “change of control event” all unvested portions of any outstanding options held by them will vest in full if we terminate the named executive officer’s employment without cause or if the named executive officer terminates his or her employment with us for good reason, in each case within ninety days before or six months after the “change in control event.”
2014 Stock Incentive Plan
In January 2014, our board of directors adopted and our stockholders approved the 2014 stock incentive plan, which became effective immediately prior to the closing of our IPO, which occurred in February 2014. The 2014 stock incentive plan provides for the grant of incentive stock options, non-statutory stock options, stock appreciation rights, restricted stock awards, restricted stock units and other stock-based awards. Upon effectiveness of the plan, the number of shares of our common stock that were reserved for issuance under the 2014 stock incentive plan was the sum of 1,951,182 shares, plus such number of shares of our common stock (up to 357,841 shares) as was equal to the sum of the number of shares of common stock reserved for issuance under the 2008 stock incentive plan that remained available for grant under the 2008 stock incentive plan immediately prior to the closing of our IPO and the number of shares of common stock subject to outstanding awards under the 2008 stock incentive plan that expire, terminate or are otherwise surrendered, cancelled, forfeited or repurchased by us at their original issuance price pursuant to a contractual repurchase right, plus an annual increase, to be added on the first day of the 2015 fiscal year and each subsequent anniversary through January 1, 2024, equal to the lowest of 2,309,023 shares of our common stock, 4% of the number of our outstanding shares on the first day of each such fiscal year and an amount determined by our board of directors. On January 1, 2015, an additional 589,722 shares of common stock were authorized for issuance under the 2014 stock incentive plan, and on January 1, 2016, an additional 865,660 of common stock were authorized for issuance under the 2014 stock incentive plan.
| 106 |
Our employees, officers, directors, consultants and advisors will be eligible to receive awards under the 2014 stock incentive plan. However, incentive stock options may only be granted to our employees.
Pursuant to the terms of the 2014 stock incentive plan, our board of directors will administer the plan and, subject to any limitations in the plan, select the recipients of awards and determines:
| • | the number of shares of our common stock covered by options and the dates upon which the options become exercisable; |
| • | the type of options to be granted; |
| • | the duration of options, which may not be in excess of ten years; |
| • | the exercise price of options, which may not be less than the fair market value of our common stock on the date of grant of the options; and |
| • | the number of shares of our common stock subject to any stock appreciation rights, restricted stock awards, restricted stock units or other stock-based awards and the terms and conditions of such awards, including conditions for repurchase, issue price and repurchase price. |
As of January 1, 2016, options to purchase 1,627,153 shares of our common stock, at a weighted average exercise price per share of $7.05 were outstanding under the 2014 stock incentive plan.
Our board of directors has delegated authority to an executive officer to grant awards under the 2014 stock incentive plan to all of our employees, except employees at or above the director level. Our board of directors has fixed the terms of the awards to be granted by such executive officer, including the exercise price of such awards, and the maximum number of shares subject to awards that such executive officer may make.
Upon a merger or other reorganization event, our board of directors may, in its sole discretion, take any one or more of the following actions pursuant to the 2014 stock incentive plan as to some or all outstanding awards other than restricted stock:
| • | provide that all outstanding awards shall be assumed, or substantially equivalent awards shall be substituted, by the acquiring or successor corporation (or an affiliate thereof); |
| • | upon written notice to a participant, provide that all of the participant’s unexercised awards will terminate immediately prior to the consummation of such reorganization event unless exercised by the participant; |
| • | provide that outstanding awards shall become exercisable, realizable or deliverable, or restrictions applicable to an award shall lapse, in whole or in part, prior to or upon such reorganization event; |
| • | in the event of a reorganization event pursuant to which holders of shares of our common stock will receive a cash payment for each share surrendered in the reorganization event, make or provide for a cash payment to the participants with respect to each award held by a participant equal to (1) the number of shares of common stock subject to the vested portion of the award (after giving effect to any acceleration of vesting that occurs upon or immediately prior to such reorganization event) multiplied by (2) the excess, if any, of the cash payment for each share surrendered in the reorganization event over the exercise, measurement or purchase price of such award and any applicable tax withholdings, in exchange for the termination of such award; and |
| • | provide that, in connection with a liquidation or dissolution, awards shall convert into the right to receive liquidation proceeds. |
In the case of certain restricted stock units, no assumption or substitution is permitted, and the restricted stock units will instead be settled in accordance with the terms of the applicable restricted stock unit agreement.
Upon the occurrence of a reorganization event other than a liquidation or dissolution, the repurchase and other rights with respect to outstanding restricted stock will continue for the benefit of the successor company and will, unless the board of directors may otherwise determine, apply to the cash, securities or other property into which shares of our common stock are converted or exchanged pursuant to the reorganization event. Upon the occurrence of a reorganization event involving a liquidation or dissolution, all restrictions and conditions on each outstanding restricted stock award will automatically be deemed terminated or satisfied, unless otherwise provided in the agreement evidencing the restricted stock award.
| 107 |
At any time, our board of directors may, in its sole discretion, provide that any award under the 2014 stock incentive plan will become immediately exercisable in full or in part, free of some or all restrictions or conditions, or otherwise realizable in full or in part.
No award may be granted under the 2014 stock incentive plan on or after January 17, 2024. Our board of directors may amend, suspend or terminate the 2014 stock incentive plan at any time, except that stockholder approval will be required to comply with applicable law or stock market requirements.
2008 Stock Incentive Plan
In February 2008, our board of directors adopted our 2008 stock incentive plan. Our stockholders approved our 2008 stock incentive plan in March 2008. Upon the completion of our IPO, our board of directors agreed not to grant any further awards under the 2008 stock incentive plan but all outstanding awards will continue to be governed by their existing terms.
Types of Awards. The 2008 stock incentive plan provides for the grant of incentive stock options within the meaning of Section 422 of the Internal Revenue Code, nonstatutory stock options, restricted stock awards, consisting of restricted stock and restricted stock units, and other forms of stock-based awards. Awards under the plan may be granted to our employees, directors and individual consultants and advisors. Only our employees are eligible to receive incentive stock options.
Share Reserve. When initially adopted, an aggregate of 201,278 shares were reserved for issuance under the 2008 stock incentive plan. The 2008 stock incentive plan was subsequently amended to increase the total number of shares which were available for issuance under the plan to 2,325,898.
As of January 1, 2016, options to purchase 1,691,388 shares of our common stock, at a weighted average exercise price per share of $5.45, were outstanding under the 2008 stock incentive plan.
Administration. Our board of directors, or a duly authorized committee thereof, is authorized to administer our 2008 stock incentive plan. Our board of directors has delegated certain authority to administer the 2008 stock incentive plan to our compensation committee; however, our general practice was that awards were approved by the board of directors. In addition, our compensation committee delegated to Mr. Abbey the authority to award stock options to purchase 251,324 shares of our common stock to non-executive employees. Our board of directors or its authorized committee has the authority under the plan to interpret and adopt rules and procedures relating to the 2008 stock incentive plan, as well as to determine the terms of any award or amend the terms of any award made under the plan. No amendment to any award made under the plan may materially and adversely affect the rights of a participant under any outstanding award without the participant’s consent.
Stock Options. Each stock option awarded under the 2008 stock incentive plan was granted pursuant to a notice of stock option and stock option agreement. The board of directors determined the exercise price for a stock option, within the terms and conditions of the 2008 stock incentive plan, provided that the exercise price of a stock option generally could not be less than 100% of the fair market value of our common stock on the date of grant. The vesting and other terms of each grant under the 2008 stock incentive plan were determined by the board of directors in its discretion; however, shares subject to stock options granted under the 2008 stock incentive plan generally vest in installments over a specified period of service, typically four years.
The board of directors determined the term of stock options granted under the 2008 stock incentive plan, subject to limitations in the case of some incentive stock options, as described below. In general, if an optionee’s service relationship with us, or any of our affiliates, ceases for any reason other than disability, death or cause, the optionee may generally exercise the vested portion of any option for a period of three months following the cessation of service. If an optionee’s service relationship with us, or any of our affiliates, ceases due to disability or death or if an optionee dies within a specified period following cessation of service, the optionee or a beneficiary generally may exercise the vested portion of any option for a period of 12 months following the death or disability. If an optionee’s services are terminated for cause, options generally terminate immediately upon such termination. In no event may an option be exercised beyond the expiration of its term.
Stock purchased upon the exercise of a stock option may, depending on the terms of the particular option agreement, be paid for using any of the following: (1) cash or check, (2) a broker-assisted cashless exercise, (3) so long as our common stock is registered under the Securities Exchange Act of 1934, the tender of common stock previously owned by the optionee, (4) delivery of a promissory note, (5) payment of other lawful consideration as determined by the plan administrator, or (6) any combination of the above.
| 108 |
Tax Limitations on Incentive Stock Options. Incentive stock options are subject to certain restrictions contained in the Internal Revenue Code. Among such restrictions, incentive stock options may be granted only to our employees. The maximum term of an incentive stock option is ten years from the date of grant. Any incentive stock option granted to any person who, at the time of the grant, owns or is deemed to own stock possessing more than 10% of our total combined voting power or that of any of our affiliates must have an exercise price equal at least to 110% of the fair market value of the stock subject to the option on the date of grant, and the term of the incentive stock option may not exceed five years from the date of grant. The aggregate fair market value, determined at the time of grant, of shares of our common stock with respect to incentive stock options that are exercisable for the first time by an optionee during any calendar year under all of our stock plans may not exceed $100,000.
Restricted Stock Awards. Each restricted stock award granted under the 2008 stock incentive plan was granted pursuant to a summary of restricted stock purchase and a restricted stock purchase agreement. An award of restricted stock entitles a participant to acquire shares of our common stock that are subject to specified restrictions, which may include a repurchase right or forfeiture right, if the shares are issued at no cost, in our favor that lapses in accordance with a vesting schedule or as conditions specified in the award are satisfied. The board of directors determined the terms and conditions of restricted stock awards, including the conditions for repurchase or forfeiture and the purchase price, if any. Unless the board of directors determined otherwise, participants holding shares of restricted stock are entitled to all ordinary cash dividends paid with respect to such shares.
Amendment. The board of directors may amend, suspend or terminate the plan at any time, subject to approval of the stockholders in certain circumstances if required by the Internal Revenue Code to ensure that incentive stock options are tax-qualified and to a participant’s consent to the extent that any amendment to the plan may materially and adversely affect the rights of a participant under any outstanding award.
Effect of Certain Corporate Transactions. Unless otherwise provided in an individual award document, in the event of specified changes of control of our company, our board of directors may take any one or more actions as to any outstanding equity award, or as to a portion of any outstanding equity award, including:
| • | providing that such awards will be assumed, or substantially equivalent awards substituted, by the acquiring or succeeding corporation or an affiliate thereof; |
| • | providing, upon notice to the participant, that all unexercised awards will terminate immediately prior to the consummation of such transaction unless exercised within a specified period of time; |
| • | providing that all or any outstanding awards will become vested or exercisable, or restrictions applicable to such awards will lapse, in full or in part, at or immediately prior to such event; |
| • | in the event of a consolidation, merger, combination, reorganization or similar transaction under the terms of which holders of our common stock will receive a cash payment per share surrendered in the transaction, making or providing for an equivalent cash payment in exchange for the termination of such equity awards; or |
| • | providing that in the event of a liquidation or dissolution awards will convert into the right to receive liquidation proceeds. |
| • | The majority of the awards granted under the 2008 stock incentive plan provide that the unvested portion of such award would become fully vested upon specified changes of control of our company. |
Transferability. Awards made under the 2008 stock incentive plan are not transferable except by will or by the laws of descent or distribution or, other than in the case of an incentive stock option, pursuant to a domestic relations order.
2014 Employee Stock Purchase Plan
In January 2014, our board of directors adopted and our stockholders approved the 2014 Employee Stock Purchase Plan, or the 2014 ESPP, which became effective immediately prior to the closing of our IPO. Under the 2014 ESPP, as of January 1, 2016, an aggregate of 312,998 shares of the Company’s common stock are reserved for issuance. Our compensation committee administers the 2014 ESPP.
The 2014 ESPP provides for six month offering periods during which eligible employees may elect to have wages or salary withheld through payroll deductions for the purpose of purchasing shares at the end of the period. All of our employees or employees of any designated subsidiary, as defined in the 2014 ESPP, are eligible to participate in the 2014 ESPP, provided that:
| • | such person is customarily employed by us or a designated subsidiary for more than 20 hours a week and for more than five months in a calendar year; |
| • | such person has been employed by us or a designated subsidiary for at least three months prior to enrolling in the 2014 ESPP; and |
| 109 |
| • | such person was our employee or an employee of a designated subsidiary on the first day of the applicable offering period under the 2014 ESPP. |
No employee is eligible to receive an option to purchase shares of our common stock that would result in the employee owning 5% or more of the total combined voting power or value of our stock immediately after the grant of such option.
We intend to make multiple offerings to our employees to purchase stock under the 2014 ESPP. Purchase plan periods under the 2014 ESPP will commence at such time or times as our board of directors determines. The first purchase plan period commenced on September 1, 2014 and ended on February 28, 2015. Our board of directors has currently authorized additional offering periods of six months each, beginning on March 1 and September 1 of each year until amended, suspended or terminated by the board. Payroll deductions made during each purchase plan period will be held for the purchase of our common stock at the end of each purchase plan period.
On the offering commencement date of each purchase plan period, we will grant to each eligible employee who is then a participant in the 2014 ESPP an option to purchase shares of our common stock. The employee may authorize up to a maximum of 10% of his or her base pay to be deducted by us during the purchase plan period. Each employee who continues to be a participant in the 2014 ESPP on the last business day of the purchase plan period is deemed to have exercised the option, to the extent of accumulated payroll deductions within the 2014 ESPP ownership limits. Under the terms of the 2014 ESPP, the option exercise price shall be determined by our board of directors for each purchase plan period and the option exercise price will be at least 85% of the applicable closing price. If our board of directors does not make a determination of the option exercise price, the option exercise price will be 85% of the lesser of the closing price of our common stock on either the first business day of the purchase plan period or the last business day of the purchase plan period. In no event may an employee purchase in any one purchase plan period a number of shares that exceeds the number of shares determined by dividing (1) the product of $2,083 and the number of full months in the purchase plan period by (2) the closing price of a share of our common stock on the commencement date of the purchase plan period. Our board of directors may, in its discretion, choose a different purchase plan period of twelve months or less for each offering.
An employee who is not a participant on the last day of the offering period is not entitled to exercise any option, and the employee’s accumulated payroll deductions will be refunded. An employee’s rights under the purchase plan terminate upon voluntary withdrawal from the purchase plan at any time, or when the employee ceases employment for any reason.
We will be required to make equitable adjustments in connection with the 2014 ESPP and any outstanding awards to reflect stock splits, reverse stock splits, stock dividends, recapitalizations, combination of shares, reclassification of shares, spin-offs and other similar changes in capitalization.
Upon the occurrence of a reorganization event, as defined in the 2014 ESPP, our board of directors is authorized to take any one or more of the following actions as to outstanding options under the 2014 ESPP:
| • | provide that options will be assumed, or substantially equivalent options will be substituted, by the acquiring or succeeding corporation (or an affiliate thereof); |
| • | upon written notice to employees, provide that all outstanding options will be terminated immediately prior to the consummation of such reorganization event and that all such outstanding options will become exercisable to the extent of accumulated payroll deductions as of a date specified by our board of directors; |
| • | upon written notice to employees, provide that all outstanding options will be cancelled as of a date prior to the effective date of the reorganization event and that all accumulated payroll deductions will be returned to participating employees on such date; |
| • | upon the occurrence of a reorganization event in which holders of our common stock will receive a cash payment for each share surrendered in the reorganization event, change the last day of the purchase plan period to be the date of the consummation of the reorganization event and provide that participants will receive a cash payment equal to the acquisition price times the number of shares of common stock that the participant’s accumulated payroll deductions as of immediately prior to the reorganization event could purchase at the option price minus the result of multiplying such number of shares by such option price; and |
| • | provide that, in connection with a liquidation or dissolution of our company, options will convert into the right to receive liquidation proceeds (net of the option price). |
| 110 |
Our board of directors may at any time, and from time to time, amend or suspend the 2014 ESPP. We will obtain stockholder approval for any amendment if such approval is required by Section 423 of the Code. Further, our board of directors may not make any amendment that would cause the 2014 ESPP to fail to comply with Section 423 of the Code. Our board of directors may terminate the 2014 ESPP at any time. Upon termination, we will refund all amounts in the accounts of participating employees.
Director Compensation
The form and amount of director compensation is reviewed and assessed from time to time by the Compensation Committee with changes, if any, recommended to the Board for action. Director compensation may take the form of cash, equity, and other benefits ordinarily available to directors.
Our non-employee director compensation policy is designed to provide a total compensation package that enables us to attract and retain qualified and experienced individuals to serve as directors and to align our directors’ interests with those of our stockholders. It provides for non-employee directors to receive an option grant of 11,000 shares upon election to the board, which will vest in equal quarterly installments over a term of three years so long as such person continues to serve as a director; an annual option grant of 5,500 shares upon the annual meeting of stockholders, which will vest in equal quarterly installments over a term of one year so long as such person continues to serve as a director; an annual retainer of $35,000; and a supplemental retainer of $25,000 in the event such director is the chairman or lead director. If the non-employee director is a member of our audit or compensation committee, he or she would receive an additional $5,000 retainer, which would be increased to $10,000 if such director was serving as the chair of such committee. If the non-employee director is a member of our governance and nominating committee, he or she would receive an additional $2,500 retainer, which would be increased to $5,000 if such director was serving as the chair of such committee.
We reimburse each non-employee director for reasonable travel expenses incurred and in connection with attendance at Board and committee meetings on our behalf, and for expenses such as supplies.
The form and amount of director compensation is reviewed and assessed from time to time by our Compensation Committee with changes, if any, recommended to our board of directors for action. Director compensation may take the form of cash, equity, and other benefits ordinarily available to directors.
| 111 |
Fiscal 2015 Compensation of Non-Employee Directors
Our non-employee directors received the following aggregate amounts of compensation in respect of the year ended December 31, 2015.
| Name | Fees Earned or Paid in Cash | Option Awards (1) | Total | |||||||||
| ($) | ($) | ($) | ||||||||||
| Hubert Birner, Ph.D. | 70,000 | 32,748 | 102,748 | |||||||||
| Robert F. Carey | 12,717 | 51,247 | 63,964 | |||||||||
| Jean Lamarre (2) | 31,249 | 32,748 | 63,997 | |||||||||
| Andrei Petrov | 42,500 | 32,748 | 75,248 | |||||||||
| Brian J. Underdown, Ph.D. | 44,999 | 32,748 | 77,747 | |||||||||
| Sander van Deventer, M.D., Ph.D. | 35,000 | 32,748 | 67,748 | |||||||||
| Philippe Van Holle | 45,000 | 32,748 | 77,748 | |||||||||
| Alexey Vinogradov, Ph.D. | 35,000 | 32,748 | 67,748 | |||||||||
___________________
| (1) | The amounts shown in this column reflect the aggregate grant date fair value of the stock awards and option awards granted to our non-employee directors computed in accordance with FASB ASC Topic 718. The assumptions made in determining the fair values of our stock awards and option awards are set forth in Notes 2 and 12 to our 2015 Consolidated Financial Statements in Part II, Item 8 of this Annual Report on Form 10-K. |
| (2) | Mr. Lamarre resigned from our board of directors in September 2015. |
| (3) | Dr. Underdown resigned from our board of directors in March 2016. |
As of December 31, 2015, our non-employee directors as of such date held the following numbers of stock options, all of which were granted under the 2014 Plan:
| Name | Option Awards | |||
| Hubert Birner, Ph.D. | 16,500 | |||
| Robert F. Carey | 11,000 | |||
| Andrei Petrov | 16,500 | |||
| Brian J. Underdown, Ph.D. | 16,500 | |||
| Sander van Deventer, M.D., Ph.D. | 16,500 | |||
| Philippe Van Holle | 16,500 | |||
| Alexey Vinogradov, Ph.D. | 16,500 | |||
| 112 |
Compensation Committee Interlocks and Insider Participation
None of our executive officers serves as a member of the board of directors or compensation committee, or other committee serving an equivalent function, of any other entity that has one or more of its executive officers serving as a member of our board of directors or our compensation committee. None of the members of our compensation committee is, or has ever been, an officer or employee of our company. The current members of our compensation committee are Andrei Petrov and Philippe Van Holle. Brian Underdown served as a member and the chair of our compensation committee until March 14, 2016, at which time he ceased to serve as a member of our board of directors.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
Securities Authorized for Issuance under Equity Compensation Plan
The following table shows information relating to our equity compensation plans as of December 31, 2015.
| Equity Compensation Plan Information | ||||||||||||
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights | Weighted average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans (excluding securities in first column) (1) | |||||||||
| Equity compensation plans approved by security holders | 3,401,322 | $ | 6.31 | 1,975,150 | ||||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 3,401,322 | $ | 6.31 | 1,975,150 | ||||||||
| (1) | Reflects the total number of shares of our common stock available for issuance under the 2014 Plan and the 2014 ESPP as of December 31, 2015. Our 2014 Plan contains an “evergreen” provision, which allows for an annual increase in the number of shares of our common stock available for issuance under the plan on the first day of each fiscal year starting in the 2015 fiscal year and on each subsequent anniversary through January 1, 2024, equal to the lowest of 2,309,023 shares of our common stock, 4% of the number of our outstanding shares of common stock on the first day of each such fiscal year or an amount determined by our board of directors. On January 1, 2016, 865,660 additional shares of our common stock were authorized for issuance under the 2014 stock incentive plan. |
| 113 |
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth information with respect to the beneficial ownership of our common stock, as of March 23, 2016 by:
| • | each of our directors; |
| • | each of our named executive officers; |
| • | all of our directors and executive officers as a group; and |
| • | each person, or group of affiliated persons, who is known by us to beneficially own more than 5% of our common stock. |
Beneficial ownership is determined in accordance with the rules and regulations of the SEC and includes voting or investment power with respect to our common stock. Shares of our common stock subject to options or warrants that are currently exercisable or exercisable within 60 days of March 23, 2016 are considered outstanding and beneficially owned by the person holding the options or warrants for the purpose of calculating the percentage ownership of that person but not for the purpose of calculating the percentage ownership of any other person. Except as otherwise noted, the persons and entities in this table have sole voting and investing power with respect to all of the shares of our common stock beneficially owned by them, subject to community property laws, where applicable.
Except as otherwise set forth in the footnotes below, the address of the beneficial owner is c/o Argos Therapeutics, Inc., 4233 Technology Drive, Durham, North Carolina 27704. Beneficial ownership representing less than one percent of our outstanding common stock is denoted with an “*.”
| Name and Address of Beneficial Owner | Number of Shares Beneficially Owned | Percentage of Shares Beneficially Owned | ||||||
| 5% Stockholders: | ||||||||
| Pharmstandard International S.A.(1) | 9,108,234 | 34.15 | % | |||||
| Entities affiliated with Forbion(2) | 3,993,198 | 15.36 | % | |||||
| Wasatch Advisors, Inc.(3) | 2,407,011 | 9.48 | % | |||||
| TVM V Life Science Ventures GmbH & Co. KG(4) | 1,792,561 | 7.04 | % | |||||
| Directors and Named Executive Officers: | ||||||||
| Andrei Petrov(5) | 17,375 | * | ||||||
| Alexey Vinogradov, Ph.D.(6) | 14,375 | * | ||||||
| Sander van Deventer, M.D., Ph.D.(7) | 4,005,573 | 15.40 | % | |||||
| Hubert Birner, Ph.D.(8) | 1,804,936 | 7.08 | % | |||||
| Philippe Van Holle(9) | 54,725 | * | ||||||
| Robert Carey(10) | 1,833 | * | ||||||
| Jeffrey D. Abbey(11) | 597,696 | 2.32 | % | |||||
| Joan C. Winterbottom(12) | 53,417 | * | ||||||
| Charles A. Nicolette, Ph.D.(13) | 281,873 | 1.10 | % | |||||
| All executive officers and directors as a group (12 persons)(14) | 16,241,659 | 61.64 | % | |||||
__________________
| (1) | The address of Pharmstandard International S.A. is 65, Boulevard Grande Duchesse Charlotte, L-1331 Luxembourg, Grand Duchy of Luxembourg. Consists of (i) 7,769,083 shares of common stock and (ii) warrants to purchase 1,339,151 shares of common stock. Pharmstandard International S.A. is a wholly owned subsidiary of Public Joint Stock Company “Pharmstandard.” As the parent entity, Public Joint Stock Company “Pharmstandard” has voting and investment control over the shares of the Company held by Pharmstandard International S.A. |
| (2) | The address of Forbion is Gooimeer 2-35 1411 DC Naarden, the Netherlands. Consists of (i) 1,254,388 shares of common stock held by Coöperatieve AAC LS U.A., (ii) 1,195,756 shares of common stock held by Forbion Co-Investment II Coöperatief U.A., (iii) 881,745 shares of common stock held by ForArgos B.V. and (iv) warrants to purchase 661,309 shares of common stock held by ForArgos B.V. Forbion 1 Management B.V., the director of Cooperatieve AAC LS U.A. and ForArgos B.V., has voting and investment power over the shares and warrants held by Cooperatieve AAC LS U.A. and ForArgos B.V., which are exercised through Forbion 1 Management B.V.‘s investment committee, consisting of L.P.A. Bergstein, H. A. Slootweg, M. A. van Osch, G. J. Mulder and Sander van Deventer. None of the members of the investment committee has individual voting and investment power with respect to such shares, and the members disclaim beneficial ownership of such shares except to the extent of their pecuniary interests therein. Forbion 1 Co- II Management B.V., the director of Forbion Co-Investment II Cooperatief U.A., has voting and investment power over the shares held by Forbion Co-Investment II Cooperatief U.A., which are exercised through Forbion 1 Co II Management B.V.‘s investment committee, consisting of L.P.A. Bergstein, H. A. Slootweg, M. A. van Osch, G. J. Mulder and Sander van Deventer. None of the members of the investment committee has individual voting and investment power with respect to such shares, and the members disclaim beneficial ownership of such shares except to the extent of their pecuniary interests therein. |
| 114 |
| (3) | Reference is hereby made to the Schedule 13G/A filed by Wasatch Advisors, Inc. on February 16, 2016 for information about the number of shares held by such reporting person and the nature of its beneficial ownership. Consists of (i) 2,350,949 shares of common stock and (ii) warrants to purchase 56,062 shares of common stock. Wasatch Advisors, Inc.'s beneficial ownership percentage was calculated using the total number of shares of common stock outstanding as of February 28, 2015. |
| (4) | The address of TVM V Life Science Ventures GmbH &Co. KG is Ottostr. 4, 80333 Munich, Germany. Consists of (i) 1,654,788 shares of common stock and (ii) warrants to purchase 137,773 shares of common stock. The shares and warrants represented here are directly held by TVM V Life Science Ventures GmbH & Co. KG (“TVM V”), the managing limited partner of which is TVM V Life Science Ventures Management GmbH & Co. KG (“TVM V Management”), for which Hubert Birner, Stefan Fischer, Alexandra Goll and Alex Polack, each a member of the investment committee of TVM V Management, share voting and investment authority over the shares held by TVM V. Each of TVM V Management, Hubert Birner, Stefan Fischer, Alexandra Goll and Alex Polack disclaims beneficial ownership of these shares, except to the extent of their pecuniary interest therein, if any. |
| (5) | Consists of (i) 5,000 shares of common stock owned directly and (ii) 12,375 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (6) | Consists of (i) 2,000 shares of common stock owned directly and (ii) 12,375 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (7) | Consists of (i) 3,993,198 shares of common stock beneficially owned by Forbion as described in footnote (2) above and (ii) 12,375 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (8) | Consists of (i) 1,792,561 shares of common stock beneficially owned by TVM V as described in footnote (4) above and (ii) 12,375 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (9) | Consists of (i) 45,100 shares of common stock and (ii) 9,625 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (10) | Consists of 1,833 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (11) | Consists of (i) 110,196 shares of common stock and (ii) 487,500 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (12) | Consists of (i) 34,459 shares of common stock and (ii) 18,958 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (13) | Consists of (i) 65,119 shares of common stock and (ii) 216,754 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| (14) | Includes (i) 15,224,717 shares of common stock and (ii) 1,035,900 shares of common stock issuable upon exercise of options that are exercisable as of March 23, 2016 or will become exercisable within 60 days after such date. |
| 115 |
Item 13. Certain Relationships and Related Transactions, and Director Independence
Since January 1, 2015, we engaged in the following transactions, in which the amount involved in the transaction exceeds $120,000 with our directors, executive officers and holders of more than 5% of our voting securities, and affiliates or immediate family members of our directors, executive officers and holders of more than 5% of our voting securities. We believe that all of these transactions were on terms as favorable as we could have obtained from unrelated third parties. Compensation arrangements for our directors and named executive officers are described in “Item 11. Executive Compensation.”
Participation in our PIPE Financing
PIPE Financing. On March 4, 2016, we entered into a securities purchase agreement with certain investors pursuant to which we agreed to issue and sell an aggregate of up to $60 million of our common stock and warrants to purchase shares of common stock in a private placement financing. The financing will take place in up to three tranches. At the closing of the initial tranche in March 2016, we sold and the investors purchased, for a total purchase price of approximately $19.9 million, a total of 3,652,430 shares of common stock and warrants to purchase a total of 2,739,323 shares of common stock (0.75 shares of common stock for each share of common stock purchased), based on a purchase price per share of common stock and accompanying warrant equal to $5.44375. At the second closing, we have agreed to sell and the investors have agreed to purchase, for an additional purchase price of approximately $29.8 million, a total of 5,478,672 shares of common stock and warrants to purchase a total of 4,109,005 shares of common stock at the same price and on the same terms as the first tranche. The second closing is conditioned on the independent data monitoring committee for the ADAPT trial at or following the independent data monitoring committee’s next regular meeting (currently scheduled for June 2016) recommending that we continue the ADAPT trial or discontinue the ADAPT trial based on favorable efficacy data. The warrants to be issued in each closing will have an exercise price of $5.35 per share and expire five years from the date of issuance. Our stockholder, Pharmstandard International S.A., or Pharmstandard, has also agreed pursuant to the securities purchase agreement that, at our option following the satisfaction of certain conditions, including the independent data monitoring committee having made a recommendation at or following its next regular meeting after the June 2016 independent data monitoring committee meeting (currently anticipated to be held in November or December 2016), that we continue the ADAPT trial or discontinue the ADAPT trial based on favorable efficacy data, and our cash position at such time, Pharmstandard shall purchase at the third closing up to approximately $10.3 million of shares of common stock (without warrants) at a price per share to be determined pursuant to an agreed upon formulation. The dollar amount committed to be purchased by Pharmstandard at the third closing is subject to reduction on a dollar-for-dollar basis for certain cash amounts raised by us after the initial closing through equity or debt financings or collaborations. All three closings are subject to the satisfaction of certain customary closing conditions.
The number of shares and warrants that each of our principal stockholders purchased and the aggregate purchase price paid for such shares and warrants is set forth in the table below.
| Name | Shares Purchased in the Initial Closing | Shares of Common Stock Underlying Warrants Purchased in the Initial Closing | Purchase Price | |||||||||
| Pharmstandard International S.A. | 1,785,534 | 1,339,151 | $ | 9,720,000 | ||||||||
| Entities affiliated with Forbion | 881,745 | 661,309 | $ | 4,799,999 | ||||||||
| Entities affiliated with Lummy (Hong Kong) Co. Ltd. | 726,704 | 545,028 | $ | 3,955,995 | ||||||||
| TVM V Life Science Ventures GmbH & Co. KG | 183,697 | 137,773 | $ | 1,000,001 | ||||||||
| Entities affiliated with Wasatch Advisors, Inc. | 74,750 | 56,062 | $ | 406,920 | ||||||||
Indemnification Agreements
Our certificate of incorporation provides that we will indemnify our directors and officers to the fullest extent permitted by Delaware law. In addition, we have entered into indemnification agreements with all of our directors and executive officers. These agreements may require us, among other things, to indemnify each such director for some expenses, including attorneys’ fees, judgments, fines and settlement amounts incurred by him in any action or proceeding arising out of his service as one of our directors.
Policies and Procedures for Related Person Transactions
Our board of directors has adopted written policies and procedures for the review of any transaction, arrangement or relationship in which we are a participant, the amount involved exceeds $120,000, and one of our executive officers, directors, director nominees or 5% stockholders, or their immediate family members, each of whom we refer to as a “related person,” has a direct or indirect material interest.
If a related person proposes to enter into such a transaction, arrangement or relationship, which we refer to as a “related person transaction,” the related person must report the proposed related person transaction to our chief executive officer. The policy calls for the proposed related person transaction to be reviewed and, if deemed appropriate, approved by our audit committee. Whenever practicable, the reporting, review and approval will occur prior to entry into the transaction. If advance review and approval is not practicable, the committee will review, and, in its discretion, may ratify the related person transaction. The policy also permits the chairman of the committee to review and, if deemed appropriate, approve proposed related person transactions that arise between committee meetings, subject to ratification by the committee at its next meeting. Any related person transactions that are ongoing in nature will be reviewed annually.
A related person transaction reviewed under the policy will be considered approved or ratified if it is authorized by the committee after full disclosure of the related person’s interest in the transaction. As appropriate for the circumstances, the committee will review and consider:
| • | the related person’s interest in the related person transaction; |
| 116 |
| • | the approximate dollar value of the amount involved in the related person transaction; |
| • | the approximate dollar value of the amount of the related person’s interest in the transaction without regard to the amount of any profit or loss; |
| • | whether the transaction was undertaken in the ordinary course of our business; |
| • | whether the terms of the transaction are no less favorable to us than terms that could have been reached with an unrelated third party; |
| • | the purpose of, and the potential benefits to us of, the transaction; and |
| • | any other information regarding the related person transaction or the related person in the context of the proposed transaction that would be material to investors in light of the circumstances of the particular transaction. |
The committee may approve or ratify the transaction only if the committee determines that, under all of the circumstances, the transaction is in, or is not inconsistent with, our best interests. The committee may impose any conditions on the related person transaction that it deems appropriate.
In addition to the transactions that are excluded by the instructions to the SEC’s related person transaction disclosure rule, our board of directors has determined that the following transactions do not create a material direct or indirect interest on behalf of related persons and, therefore, are not related person transactions for purposes of this policy:
| • | interests arising solely from the related person’s position as an executive officer of another entity (whether or not the person is also a director of such entity), that is a participant in the transaction, where (a) the related person and all other related persons own in the aggregate less than a 10% equity interest in such entity, (b) the related person and his or her immediate family members are not involved in the negotiation of the terms of the transaction and do not receive any special benefits as a result of the transaction, and (c) the amount involved in the transaction equals less than the greater of $200,000 dollars or 5% of the annual gross revenues of the other entity that is a party to the transaction; and |
| • | a transaction that is specifically contemplated by provisions of our charter or bylaws. |
The policy provides that transactions involving compensation of executive officers shall be reviewed and approved by the compensation committee in the manner specified in its charter.
Director Independence
Our board of directors has determined that all of the members of the audit committee, the compensation committee and the nominating and corporate governance committee are independent, as defined under the NASDAQ rules, including, in the case of all the members of our audit committee, the independence requirements contemplated by Rule 10A-3 under the Exchange Act. In making such determination, our board of directors considered the relationships that each such director has with our company and all other facts and circumstances that our board of directors deemed relevant in determining independence, including the beneficial ownership of our capital stock by each non-employee director.
Item 14. Principal Accountant Fees and Services
PricewaterhouseCoopers LLP has been approved by our Audit Committee to act as our independent registered public accounting firm for the year ending December 31, 2016.
Audit and other fees billed to us by PricewaterhouseCoopers LLP for the years ended December 31, 2015 and 2014 are as follows:
| 2015 | 2014 | |||||||
| Audit Fees (1) | $ | 314,906 | $ | 379,974 | ||||
| Audit-Related Fees (2) | — | — | ||||||
| Tax Fees (3) | 3,167 | — | ||||||
| All Other Fees (4) | — | — | ||||||
| Total Fees for Services Provided | $ | 318,073 | $ | 379,974 | ||||
| (1) | Audit fees include fees associated with the annual audit, reviews of interim financial statements included in SEC registration statements, accounting and reporting consultations and audits conducted under OMB Circular A-133. |
| 117 |
| (2) | There were no audit-related fees for the years ended December 31, 2015 or 2014. |
| (3) | Consisted of tax fees related to our Canadian subsidiary, DC Bio for the year ended December 31, 2015. There were no tax fees for the year ended December 31, 2014. |
| (4) | Other fees include fees billed for other services rendered not included within Audit Fees, Audit Related Fees or Tax Fees. There were no other fees for the years ended December 31, 2015 or 2014. |
PricewaterhouseCoopers LLP did not perform any professional services related to financial information systems design and implementation for us in the year ended December 31, 2015 or 2014.
The Audit Committee has determined in its business judgment that the provision of non-audit services described above is compatible with maintaining PricewaterhouseCoopers LLP’s independence.
In 2014, the Audit Committee adopted a formal policy concerning approval of audit and non-audit services to be provided to the Company by its independent registered public accounting firm, PricewaterhouseCoopers LLP. The policy requires that all services to be provided by PricewaterhouseCoopers LLP, including audit services and permitted audit-related and non-audit services, must be preapproved by the Audit Committee, provided that de minimis non-audit services may instead be approved in accordance with applicable SEC rules. The Board of Directors preapproved all audit and non-audit services provided by PricewaterhouseCoopers LLP during years ended December 31, 2015 and 2014.
PART IV
Item 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
(a) The following documents are included in this Annual Report on Form 10-K:
1. The following Report and Consolidated Financial Statements of the Company are included in this Annual Report:
Report of Independent Registered Public Accounting Firm
Consolidated Balance Sheets
Consolidated Statements of Operations
Consolidated Statements of Comprehensive Loss
Consolidated Statements of Changes in Stockholders’ (Deficit) Equity
Consolidated Statements of Cash Flows
Notes to Consolidated Financial Statements
Financial Statement Schedule:
Schedule II – Valuation and Qualifying Accounts
2. All other schedules are omitted as they are inapplicable or the required information is furnished in the Consolidated Financial Statements or notes thereto.
3. The exhibits filed as part of this Annual Report on Form 10-K are listed in the Exhibit Index immediately following the signature page of this Annual Report on Form 10-K and are incorporated herein.
| 118 |
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| ARGOS THERAPEUTICS, INC. | ||
| By: | /s/ Jeffrey D. Abbey | |
| Name: Jeffrey D. Abbey | ||
| Title: President and Chief Executive Officer | ||
Date: March 30, 2016
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant on March 30, 2016 in the capacities indicated.
|
Signature |
Title |
Date |
|
/s/ Jeffrey D. Abbey Jeffrey D. Abbey |
President, Chief Executive Officer and Director (Principal Executive Officer) |
March 30, 2016 |
|
/s/ Lori R. Harrelson Lori R. Harrelson |
Vice President of Finance (Principal Financial Officer and Principal Accounting Officer) |
March 30, 2016 |
|
/s/ Hubert Birner, Ph.D. Hubert Birner, Ph.D. |
Director | March 30, 2016 |
|
/s/ Robert F. Carey Robert F. Carey |
Director | March 30, 2016 |
|
/s/ Andrei Petrov Andrei Petrov |
Director | March 30, 2016 |
|
/s/ Sander van Deventer, M.D., Ph.D. Sander van Deventer, M.D., Ph.D. |
Director | March 30, 2016 |
|
/s/ Philippe Van Holle Philippe Van Holle |
Director | March 30, 2016 |
|
/s/ Alexey Vinogradov, Ph.D. Alexey Vinogradov, Ph.D. |
Director | March 30, 2016 |
| 119 |
EXHIBIT INDEX
|
Exhibit |
Description of Exhibit |
| 3.1 | Restated Certificate of Incorporation of the Registrant (filed as Exhibit 3.1 to the Registrant’s Current Report on Form 8-K (File No. 001-35443) on February 18, 2014 and incorporated herein by reference) |
| 3.2 | Amended and Restated Bylaws of the Registrant (filed as Exhibit 3.2 to the Registrant’s Current Report on Form 8-K (File No. 001-35443) on February 18, 2014 and incorporated herein by reference) |
| 4.1 | Specimen Stock Certificate evidencing the shares of common stock (filed as Exhibit 4.1 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 4.2 | Fifth Amended and Restated Registration Rights Agreement, dated as of August 9, 2013 (filed as Exhibit 4.2 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.1+ | 2008 Stock Incentive Plan, as amended (filed as Exhibit 10.2 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.2+ | Form of Incentive Stock Option Agreement under 2008 Stock Incentive Plan (filed as Exhibit 10.3 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.3+ | Form of Nonstatutory Stock Option Agreement under 2008 Stock Incentive Plan (filed as Exhibit 10.4 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.4+ | 2014 Stock Incentive Plan (filed as Exhibit 10.5 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 10.5+ | Form of Incentive Stock Option Agreement under 2014 Stock Incentive Plan (filed as Exhibit 10.6 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 10.6+ | Form of Nonstatutory Stock Option Agreement under 2014 Stock Incentive Plan (filed as Exhibit 10.7 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 10.7 | Lease Agreement, dated as of January 16, 2001, between the Registrant and HCP MOP, as amended (filed as Exhibit 10.8 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.8+ | Employment Agreement between the Registrant and Jeffrey D. Abbey, dated December 9, 2013 (filed as Exhibit 10.9 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.9+ | Employment Agreement between the Registrant and Charles A. Nicolette, dated December 9, 2013 (filed as Exhibit 10.10 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.10+ | Employment Agreement between the Registrant and Frederick M. Miesowicz, dated December 9, 2013 (filed as Exhibit 10.11 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 120 |
Exhibit |
Description of Exhibit |
| 10.11+ | Employment Agreement between the Registrant and Lori R. Harrelson, dated December 9, 2013 (filed as Exhibit 10.12 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.12 | Form of Indemnification Agreement between the Registrant and each director and executive officer (filed as Exhibit 10.14 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.13† | Contract No. HHSN266200600019C, dated September 30, 2006, by and among the Registrant, the National Institutes of Health and the National Institutes of Allergy and Infectious Diseases, as amended (filed as Exhibit 10.15 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.14† | License Agreement, dated August 9, 2013, by and between the Registrant and Pharmstandard S.A. (filed as Exhibit 10.16 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.15† | License Agreement, dated July 31, 2013, by and between the Registrant and Green Cross Corp. (filed as Exhibit 10.17 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 10.16† | License Agreement, dated July 28, 2011, by and between the Registrant and Celldex Therapeutics, Inc. (filed as Exhibit 10.18 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.17† | License Agreement, dated January 10, 2000, by and between the Registrant and Duke University, as amended (filed as Exhibit 10.19 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on December 30, 2013 and incorporated herein by reference) |
| 10.18 | Acknowledgement Agreement, dated November 4, 2013, by and between the Registrant and Pharmstandard International S.A. (filed as Exhibit 10.20 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 10.19+ | 2014 Employee Stock Purchase Plan (filed as Exhibit 10.21 to Amendment No. 1 to the Registrant’s Registration Statement on Form S-1 (File No. 333-193137) on January 21, 2014 and incorporated herein by reference) |
| 10.20 | Lease Agreement, dated August 18, 2014, by and between by and between the Registrant and TKC LXXII, LLC (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K on August 22, 2014 and incorporated herein by reference) |
| 10.21 |
Venture Loan and Security Agreement, dated September 29, 2014, by and between the Registrant and Horizon Technology Finance Corporation and Fortress Credit Co LLC (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K on September 30, 2014 and incorporated herein by reference)
|
| 10.22 |
Form of Warrant to Purchase Common Stock, issued to Horizon Technology Finance Corporation on September 29, 2014 (filed as Exhibit 10.2 to the Registrant’s Current Report on Form 8-K on September 30, 2014 and incorporated herein by reference)
|
| 10.23 | Form of Warrant to Purchase Common Stock, issued to Drawbridge Special Opportunities Fund LP on September 29, 2014 (filed as Exhibit 10.3 to the Registrant’s Current Report on Form 8-K on September 30, 2014 and incorporated herein by reference) |
| 10.24 | Development Agreement, dated October 29, 2014, by and between the Registrant and Invetech Lty Ltd (filed as Exhibit 10.5 to the Registrant’s Quarterly Report on Form 10-Q on November 14, 2014 and incorporated herein by reference) |
| 10.25 | Development Agreement, dated January 5, 2015, by and between the Registrant and Saint-Gobain Performance Plastics Corporation (filed as Exhibit 10.27 to the Registrant’s Annual Report on Form 10-K on March 31, 2015 and incorporated herein by reference) |
| 121 |
Exhibit |
Description of Exhibit |
| 10.26 | Purchase and Sale Agreement, dated February 16, 2015, by and between the Registrant and TKC LXXII, LLC (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K on February 20, 2015 and incorporated by reference) |
| 10.27 | Novated, Amended and Restated License Agreement effective as of October 1, 2014, by and between the Registrant and MEDcell Co., Ltd., as amended on December 28, 2015 and January 28, 2016 (filed as Exhibit 10.29 to the Registrant’s Annual Report on Form 10-K on March 31, 2015 and incorporated herein by reference) |
| 10.28 | Modification No. 11, effective September 18, 2014, to Contract No. HHSN266200600019C dated September 30, 2006, by and among the Registrant, the National Institutes of Health and the National Institutes of Allergy and Infectious Diseases, as amended (filed as Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q on November 16, 2015 and incorporated herein by reference) |
| 10.29† | License Agreement, dated April 7, 2015, by and between the Registrant and Lummy (Hong Kong) Co., Ltd. (filed as Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q on May 15, 2015 and incorporated herein by reference) |
| 10.30†* |
Master Process Development and Supply Agreement, dated December 22, 2015, by and between the Registrant and Cellscript, LLC |
| 10.31* |
Employment Agreement, dated February 3, 2015, by and between the Registrant and Joan Winterbottom |
| 10.32 | Securities Purchase Agreement, dated March 4, 2016, by and between the Registrant and the investors named therein (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K on March 7, 2016 and incorporated herein by reference) |
| 10.33 | Form of Common Stock Warrant (filed as Exhibit 10.2 to the Registrant’s Current Report on Form 8-K on March 7, 2016 and incorporated herein by reference) |
| 10.34 |
Registration Rights Agreement, dated March 4, 2016, by and between the Registrant and the investors named therein (filed as Exhibit 10.3 to the Registrant’s Current Report on Form 8-K on March 7, 2016 and incorporated herein by reference) |
| 21.1 | Subsidiaries of the Registrant (filed as Exhibit 21.1 to the Registrant’s Registration Statement on Form S-1 on December 30, 2013 and incorporated herein by reference) |
| 23.1* | Consent of PriceWaterhouseCoopers, an independent registered public accounting firm |
| 31.1* | Certification of principal executive officer pursuant to Rules 13a-14(a) or 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
| 31.2* | Certification of principal financial officer pursuant to Rules 13a-14(a) or 15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 |
| 32.1* | Certifications pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of The Sarbanes-Oxley Act of 2002, by the Registrant’s principal executive officer and principal financial officer |
|
101.INS |
XBRL Instance Document |
|
101.SCH |
XBRL Taxonomy Extension Schema Document |
|
101.CAL |
XBRL Taxonomy Extension Calculation Linkbase Document |
| 122 |
Exhibit |
Description of Exhibit |
|
101.DEF
|
XBRL Taxonomy Extension Definition Linkbase Document
|
|
101.LAB
|
XBRL Taxonomy Extension Label Linkbase Document
|
|
101.PRE |
XBRL Taxonomy Extension Presentation Linkbase Document |
| † | Confidential treatment requested as to portions of the exhibit. Confidential materials omitted and filed separately with the Securities and Exchange Commission. |
| + | Management contract or compensatory plan or arrangement required to be filed as exhibits hereto pursuant to Item 15(a) of Form 10-K. |
| * | Filed herewith. |
| 123 |
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| F-1 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of
Argos Therapeutics, Inc.
In our opinion, the consolidated financial statements listed in the index appearing under Item 15(a)(1) present fairly, in all material respects, the financial position of Argos Therapeutics, Inc. and its subsidiaries at December 31, 2015 and 2014, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2015 in conformity with accounting principles generally accepted in the United States of America. In addition, in our opinion, the financial statement schedule listed in the index appearing under Item 15(a)(1) presents fairly, in all material respects, the information set forth therein when read in conjunction with the related consolidated financial statements. These financial statements and financial statement schedule are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements and financial statement schedule based on our audits. We conducted our audits of these statements in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has suffered recurring losses from operations and has an accumulated deficit that raise substantial doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
/s/ PricewaterhouseCoopers LLP
Raleigh, North Carolina
March 30, 2016
| F-2 |
CONSOLIDATED BALANCE SHEETS
| December 31, | ||||||||
| 2014 | 2015 | |||||||
| Assets | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | $ | 37,223,590 | $ | 6,163,144 | ||||
| Short-term investments | 19,016,347 | 1,003,160 | ||||||
| Restricted cash | — | 740,000 | ||||||
| Prepaid expenses | 838,420 | 740,173 | ||||||
| Deferred financing costs | 309,927 | 93,503 | ||||||
| Other receivables | 129,019 | 73,818 | ||||||
| Total current assets | 57,517,303 | 8,813,798 | ||||||
| Property and equipment, net | 5,513,555 | 22,306,384 | ||||||
| Restricted cash | 1,325,000 | — | ||||||
| Other assets | 11,020 | 11,020 | ||||||
| Total assets | $ | 64,366,878 | $ | 31,131,202 | ||||
| Liabilities and Stockholders’ Equity (Deficit) | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 1,860,491 | $ | 2,907,683 | ||||
| Other accrued expenses | 1,405,378 | 3,676,846 | ||||||
| Current portion of notes payable | 30,885 | 1,578,284 | ||||||
| Total current liabilities | 3,296,754 | 8,162,813 | ||||||
| Long-term portion of notes payable | 19,796,545 | 30,821,761 | ||||||
| Long-term portion of manufacturing research and development obligation | 3,475,552 | 7,777,436 | ||||||
| Long-term portion of facility lease obligation | 3,380,223 | 7,249,627 | ||||||
| Long-term deferred liabilities | 3,066,000 | 5,321,000 | ||||||
| Commitments | — | — | ||||||
| Stockholders’ equity (deficit) | ||||||||
| Preferred stock $0.001 par value; 5,000,000 shares authorized as of December 31, 2014 and 2015; 0 shares issued and outstanding as of December 31, 2014 and 2015 | — | — | ||||||
| Common stock $0.001 par value; 200,000,000 shares authorized as of December 31, 2014 and 2015; 19,657,412 and 21,641,509 shares issued and outstanding as of December 31, 2014 and 2015 | 19,657 | 21,642 | ||||||
| Accumulated other comprehensive loss | (124,841 | ) | (138,245 | ) | ||||
| Additional paid-in capital | 235,627,174 | 250,873,875 | ||||||
| Accumulated deficit | (204,170,186 | ) | (278,958,707 | ) | ||||
| Total stockholders’ equity (deficit) | 31,351,804 | (28,201,435 | ) | |||||
| Total liabilities and stockholders’ equity (deficit) | $ | 64,366,878 | $ | 31,131,202 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-3 |
CONSOLIDATED STATEMENTS OF OPERATIONS
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| Revenue | $ | 4,421,689 | $ | 1,974,019 | $ | 518,329 | ||||||
| Operating expenses | ||||||||||||
| Research and development | 23,991,151 | 45,498,916 | 62,054,823 | |||||||||
| General and administrative | 4,662,317 | 8,599,359 | 11,011,011 | |||||||||
| Total operating expenses | 28,653,468 | 54,098,275 | 73,065,834 | |||||||||
| Operating loss | (24,231,779 | ) | (52,124,256 | ) | (72,547,505 | ) | ||||||
| Other income (expense) | ||||||||||||
| Interest income | 7,184 | 66,580 | 25,382 | |||||||||
| Interest expense | (4,705 | ) | (1,123,579 | ) | (2,263,599 | ) | ||||||
| Change in fair value of warrant liability | 355,352 | — | — | |||||||||
| Investment tax credits | — | 140,556 | — | |||||||||
| Other expense | (47,615 | ) | (265,239 | ) | (2,799 | ) | ||||||
| Other income (expense), net | 310,216 | (1,181,682 | ) | (2,241,016 | ) | |||||||
| Net loss | (23,921,563 | ) | (53,305,938 | ) | (74,788,521 | ) | ||||||
| Accretion of redeemable convertible preferred stock (See Note 15) | 4,772,991 | (863,226 | ) | — | ||||||||
| Less: Preferred stock dividend due to exchanges of preferred shares (See Note 15) | (14,726,088 | ) | — | — | ||||||||
| Net loss attributable to common stockholders | $ | (33,874,660 | ) | $ | (54,169,164 | ) | $ | (74,788,521 | ) | |||
| Net loss attributable to common stockholders per share, basic and diluted | $ | (147.37 | ) | $ | (3.12 | ) | $ | (3.66 | ) | |||
| Weighted average shares outstanding, basic and diluted | 229,865 | 17,367,665 | 20,457,245 | |||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-4 |
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| Net loss | $ | (23,921,563 | ) | $ | (53,305,938 | ) | $ | (74,788,521 | ) | |||
| Other comprehensive loss: | ||||||||||||
| Foreign currency translation (loss) | (8,264 | ) | (10,382 | ) | (25,061 | ) | ||||||
| Unrealized (loss) gain on short-term investments | — | (11,928 | ) | 11,657 | ||||||||
| Total comprehensive loss | $ | (23,929,827 | ) | $ | (53,328,248 | ) | $ | (74,801,925 | ) | |||
The accompanying notes are an integral part of these consolidated financial statements.
| F-5 |
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT)
| Common Stock Shares | Common Stock Amount | Additional Paid-in Capital | Accumulated Other Comprehensive Income (Loss) | Accumulated Deficit | Total Stockholders’ Equity (Deficit) | |||||||||||||||||||
| Balance as of December 31, 2012 | 226,839 | $ | 227 | $ | 58,469,015 | $ | (94,267 | ) | $ | (126,942,685 | ) | $ | (68,567,710 | ) | ||||||||||
| Exercise of common stock options | 1,379 | 2 | 5,791 | — | — | 5,793 | ||||||||||||||||||
| Issuance of restricted stock | 7,571 | 7 | 16,493 | — | — | 16,500 | ||||||||||||||||||
| Issuance of common warrants | — | — | 618,155 | — | — | 618,155 | ||||||||||||||||||
| Distribution of shares to affiliates | (7 | ) | — | — | — | — | — | |||||||||||||||||
| Surrender of shares | (75 | ) | — | — | — | — | — | |||||||||||||||||
| Stock-based compensation | — | — | 1,053,163 | — | — | 1,053,163 | ||||||||||||||||||
| Reversal of prior accretion | — | — | 5,657,638 | — | — | 5,657,638 | ||||||||||||||||||
| Accretion of preferred stock | — | — | (884,647 | ) | — | — | (884,647 | ) | ||||||||||||||||
| Exchange of preferred shares | — | — | (14,726,088 | ) | — | — | (14,726,088 | ) | ||||||||||||||||
| Reduction of liquidation value | — | — | 24,980,430 | — | — | 24,980,430 | ||||||||||||||||||
| Cumulative translation adjustment | — | — | — | (8,264 | ) | — | (8,264 | ) | ||||||||||||||||
| Net loss | — | — | — | — | (23,921,563 | ) | (23,921,563 | ) | ||||||||||||||||
| Balance as of December 31, 2013 | 235,707 | $ | 236 | $ | 75,189,950 | $ | (102,531 | ) | $ | (150,864,248 | ) | $ | (75,776,593 | ) | ||||||||||
| Issuance of common stock | 6,228,725 | 6,229 | 49,823,571 | — | — | 49,829,800 | ||||||||||||||||||
| Common stock issuance costs | — | — | (6,391,588 | ) | — | — | (6,391,588 | ) | ||||||||||||||||
| Exercise of common stock options | 3,050 | 3 | 12,807 | — | — | 12,810 | ||||||||||||||||||
| Conversion of warrants into common stock | 1,679 | 1 | (1 | ) | — | — | — | |||||||||||||||||
| Stock-based compensation | — | — | 3,013,284 | — | — | 3,013,284 | ||||||||||||||||||
| Accretion of preferred stock | — | — | (863,226 | ) | — | — | (863,226 | ) | ||||||||||||||||
| Conversion of preferred stock into common stock | 13,188,251 | 13,188 | 114,514,267 | — | — | 114,527,455 | ||||||||||||||||||
| Issuance of warrants | — | — | 328,110 | — | — | 328,110 | ||||||||||||||||||
| Cumulative translation adjustment | — | — | — | (10,382 | ) | — | (10,382 | ) | ||||||||||||||||
| Unrealized loss on short-term investments | — | — | — | (11,928 | ) | — | (11,928 | ) | ||||||||||||||||
| Net loss | — | — | — | — | (53,305,938 | ) | (53,305,938 | ) | ||||||||||||||||
| Balance as of December 31, 2014 | 19,657,412 | $ | 19,657 | $ | 235,627,174 | $ | (124,841 | ) | $ | (204,170,186 | ) | $ | 31,351,804 | |||||||||||
| Issuance of common stock | 1,906,194 | 1,906 | 10,679,526 | — | — | 10,681,432 | ||||||||||||||||||
| Exercise of common stock options | 44,548 | 46 | 201,042 | — | — | 201,088 | ||||||||||||||||||
| Stock-based compensation | — | — | 4,014,938 | — | — | 4,014,938 | ||||||||||||||||||
| Issuance of common stock under employee stock purchase plan | 33,355 | 33 | 207,183 | — | — | 207,216 | ||||||||||||||||||
| Issuance of warrants | — | — | 144,012 | — | — | 144,012 | ||||||||||||||||||
| Cumulative translation adjustment | — | — | — | (25,061 | ) | — | (25,061 | ) | ||||||||||||||||
| Unrealized gain on short-term investments | — | — | — | 11,657 | — | 11,657 | ||||||||||||||||||
| Net loss | — | — | — | — | (74,788,521 | ) | (74,788,521 | ) | ||||||||||||||||
| Balance as of December 31, 2015 | 21,641,509 | $ | 21,642 | $ | 250,873,875 | $ | (138,245 | ) | $ | (278,958,707 | ) | $ | (28,201,435 | ) | ||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-6 |
CONSOLIDATED STATEMENTS OF CASH FLOWS
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| Cash flows from operating activities | ||||||||||||
| Net loss | $ | (23,921,563 | ) | $ | (53,305,938 | ) | $ | (74,788,521 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 611,421 | 566,117 | 680,444 | |||||||||
| Share-based compensation expense | 1,081,186 | 3,013,283 | 4,014,938 | |||||||||
| Amortization of debt issuance costs | — | 69,308 | 216,424 | |||||||||
| Amortization of debt discount | — | 24,917 | 116,042 | |||||||||
| Interest accrued on long-term debt | — | 693,114 | 25,732 | |||||||||
| Noncash interest expense | 2,465 | — | — | |||||||||
| Decrease in fair value of warrant liability | (355,352 | ) | — | — | ||||||||
| Issuance of common stock as payment for research and development services | — | — | 2,111,432 | |||||||||
| Issuance of restricted stock recorded as consulting expense | 16,500 | — | — | |||||||||
| (Gain) loss on disposal of equipment | (50,835 | ) | (1,710 | ) | 2,799 | |||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Prepaid expenses and other receivables | 438,867 | 86,997 | 153,449 | |||||||||
| Deferred financing costs | (571,327 | ) | — | — | ||||||||
| Other assets | — | (10,471 | ) | — | ||||||||
| Accounts payable | 181,965 | 543,419 | (136,981 | ) | ||||||||
| Accrued expenses | 1,244,940 | (395,416 | ) | 796,684 | ||||||||
| Long-term portion of manufacturing research and development obligation | — | 3,475,552 | 4,301,884 | |||||||||
| Long-term deferred liabilities | 3,066,000 | — | 1,485,000 | |||||||||
| Net cash used in operating activities | (18,255,733 | ) | (45,240,828 | ) | (61,020,674 | ) | ||||||
| Cash flows from investing activities | ||||||||||||
| Purchase of property and equipment | (596,455 | ) | (1,097,798 | ) | (9,686,643 | ) | ||||||
| Proceeds from sale of property and equipment | 50,835 | 2,000 | — | |||||||||
| Purchases of short-term investments | (13,600,941 | ) | (25,593,464 | ) | (2,677,155 | ) | ||||||
| (Payment to) receipt from restricted cash account securing letter of credit | — | (1,325,000 | ) | 585,000 | ||||||||
| Proceeds from maturity of short-term investments | 4,090,000 | 20,225,000 | 20,702,000 | |||||||||
| Net cash (used in) provided by investing activities | (10,056,561 | ) | (7,789,262 | ) | 8,923,202 | |||||||
| Cash flows from financing activities | ||||||||||||
| Proceeds from sale of common stock | — | 49,829,800 | 8,570,000 | |||||||||
| Proceeds from sale of redeemable convertible preferred stock | 47,409,867 | — | — | |||||||||
| Proceeds from issuance of common stock warrants | 590,132 | — | — | |||||||||
| Stock issuance costs | (1,501,344 | ) | (4,875,404 | ) | — | |||||||
| Proceeds from issuance of notes payable with detachable common stock warrants | 6,934,000 | 12,500,000 | 12,500,000 | |||||||||
| Debt issuance costs | — | (449,796 | ) | — | ||||||||
| Payment on long-term portion of facility lease obligation | — | — | (381,033 | ) | ||||||||
| Payments on notes payable | (34,934 | ) | (51,481 | ) | (35,480 | ) | ||||||
| Proceeds from exercise of employee stock purchase plan rights | — | — | 207,233 | |||||||||
| Proceeds from exercise of common stock options | 5,793 | 12,810 | 201,067 | |||||||||
| Net cash provided by financing activities | 53,403,514 | 56,965,929 | 21,061,787 | |||||||||
| Effect of exchange rates changes on cash | (8,115 | ) | (10,219 | ) | (24,761 | ) | ||||||
| Net increase (decrease) in cash and cash equivalents | 25,083,105 | 3,925,620 | (31,060,446 | ) | ||||||||
| Cash and cash equivalents | ||||||||||||
| Beginning of period | 8,214,865 | 33,297,970 | 37,223,590 | |||||||||
| End of period | $ | 33,297,970 | $ | 37,223,590 | $ | 6,163,144 | ||||||
| Supplemental disclosure of cash flow information | ||||||||||||
| Cash paid for interest | $ | 512 | $ | 307,944 | $ | 1,648,707 | ||||||
| Supplemental disclosure of noncash investing and financing activities | ||||||||||||
| Issuance of common stock for research and development services | $ | — | $ | — | $ | 2,111,432 | ||||||
| Conversion of preferred stock into common stock | $ | — | $ | 114,527,695 | $ | — | ||||||
| Preferred stock accretion | $ | (4,772,991 | ) | $ | 863,226 | $ | — | |||||
| Interest capitalized on construction-in-progress | $ | — | $ | — | $ | 880,334 | ||||||
| Purchases of property and equipment included in accounts payable and accrued expenses | $ | — | $ | — | $ | 2,658,958 | ||||||
| Recognition of asset and facility lease obligation related to construction of new property | $ | — | $ | 3,380,223 | $ | 4,250,437 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-7 |
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Organization and Accounting Policies
Argos Therapeutics, Inc. (the “Company”), was incorporated in the State of Delaware on May 8, 1997. The Company is an immuno-oncology company focused on the development and commercialization of fully individualized immunotherapies for the treatment of cancer using its Arcelis technology platform. The Company’s most advanced product candidate, AGS-003, is being evaluated in the pivotal ADAPT Phase 3 clinical trial (the “ADAPT trial”) for the treatment of metastatic renal cell carcinoma (“mRCC”). The Company is also developing a separate Arcelis-based product candidate, AGS-004, for the treatment of HIV, which is currently being evaluated in a Phase 2 clinical trial aimed at HIV eradication in adult patients.
The Company’s consolidated financial statements have been prepared assuming that the Company will continue as a going concern, which contemplates the realization of assets and the settlement of liabilities and commitments in the normal course of business. The Company has incurred losses in each year since inception and as of December 31, 2015, had an accumulated deficit of $279.0 million. Also, as of December 31, 2015, the Company’s current assets totaled $8.8 million compared with current liabilities of $8.2 million, and the Company had cash, cash equivalents and short-term investments of $7.2 million. These factors raise substantial doubt about its ability to continue as a going concern. The financial statements for the year ended December 31, 2015 do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from uncertainty related to the Company’s ability to continue as a going concern.
Until such time, if ever, as the Company can generate substantial product revenues, it expects to seek to raise additional funds through a combination of equity offerings, debt financings, government contracts, government and other third party grants or other third party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. There can be no assurance that the Company will be able to generate funds in these manners, on terms acceptable to the Company, on a timely basis or at all. The failure of the Company to obtain sufficient funds on acceptable terms when needed could have a material adverse effect on the Company’s business, results of operations and financial condition and the Company could be forced to delay, reduce, terminate or eliminate its product development programs.
In connection with the Company’s initial public offering in February 2014, the Company effected a one–for-six reverse split of its common stock. All references to shares of common stock outstanding, average number of shares outstanding and per share amounts in these consolidated financial statements and notes to consolidated financial statements have been restated to reflect the reverse split on a retroactive basis.
The Company prepares consolidated financial statements in conformity with accounting principles generally accepted in the United States (“GAAP”). The Company also applies the following accounting policies when preparing its consolidated financial statements:
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities as of the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Principles of Consolidation
The condensed consolidated financial statements include the accounts of the Company and DC Bio Corp., the Company’s Canadian wholly-owned subsidiary, an unlimited liability corporation incorporated in the Province of Nova Scotia. Significant intercompany transactions and accounts have been eliminated.
Net Loss per Share
Basic net loss per share is calculated by dividing net loss attributable to common stockholders by the weighted average shares outstanding during the period, without consideration of common stock equivalents. Diluted net loss per share is calculated by adjusting weighted average shares outstanding for the dilutive effect of common stock equivalents outstanding for the period, determined using the treasury-stock method. For purposes of the diluted net loss per share calculation, preferred stock, stock options and warrants are considered to be common stock equivalents but are excluded from the calculation of diluted net loss per share because their effect would be anti-dilutive and, therefore, basic and diluted net loss per share were the same for all periods presented.
| F-8 |
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original maturity of three months or less as of the date of purchase to be cash equivalents. Cash deposits are all in financial institutions in the United States of America, Canada and the European Union. The Company maintains cash in accounts which are in excess of federally insured limits. As of December 31, 2014 and 2015, $36,973,590 and $5,913,144, respectively, in cash and cash equivalents was uninsured.
Short-Term Investments
All investments with original maturities less than one year from the balance sheet date are considered short-term investments. All short-term investments are classified as available-for-sale and therefore carried at fair value. Generally, the fair value of short-term investments approximates amortized cost. The Company primarily invests in high-quality marketable debt securities issued by high quality financial and industrial companies.
Credit Risk
Financial instruments which potentially subject the Company to concentrations of credit risk consist principally of cash and cash equivalents and short-term investments.
Property and Equipment
Property and equipment are recorded at cost and depreciated over their estimated useful lives using the straight-line method. Property and equipment held under capital leases and leasehold improvements are amortized over the shorter of the lease term or the estimated useful life of the related asset. Upon retirement or sale, the cost of assets disposed of and the related accumulated depreciation are removed from the accounts and any resulting gain or loss is credited or charged to income. Repairs and maintenance costs are expensed as incurred.
Impairment of Long-Lived Assets
The Company periodically assesses the impairment of long lived assets. When indicators of impairment are present, the Company evaluates the carrying value of these assets in relation to the operating performance of the business and future undiscounted cash flows expected to result from the use of these assets. No such impairments have been recognized during the years ended December 31, 2013, 2014 or 2015.
Revenue Recognition
The Company recognizes revenue in accordance with the Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 605, Revenue Recognition, or ASC 605. The Company recognizes revenue when the following criteria are met: persuasive evidence of an arrangement exists, services have been rendered, the price is fixed or determinable, and collectability is reasonably assured.
The Company has entered into license agreements with collaborators. The terms of these agreements have included nonrefundable signing and licensing fees, as well as milestone payments and royalties on any future product sales developed by the collaborators under such licenses. The Company assesses these multiple elements in accordance with ASC 605, to determine whether particular components of the arrangement represent separate units of accounting.
These collaboration agreements will be accounted for in accordance with Accounting Standards Update (“ASU”) No. 2009-13, Topic 605 – Multiple-Deliverable Revenue Arrangements, or ASU 2009-13. This guidance requires the application of the “relative selling price” method when allocating revenue in a multiple deliverable arrangement. The selling price for each deliverable shall be determined using vendor specific objective evidence of selling price, if it exists; otherwise, third-party evidence of selling price shall be used. If neither exists for a deliverable, the vendor shall use its best estimate of the selling price for that deliverable. The Company recognizes upfront license payments as revenue upon delivery of the license only if the license has standalone value and the fair value of the undelivered performance obligations can be determined. If the fair value of the undelivered performance obligations can be determined, such obligations are accounted for separately as the obligations are fulfilled. If the license is considered to either not have stand-alone value or have stand-alone value but the fair value of any of the undelivered performance obligations cannot be determined, the arrangement is accounted for as a single unit of accounting and the license payments and payments for performance obligations are recognized as revenue over the estimated period of when the performance obligations are performed.
| F-9 |
When the Company determines that an arrangement should be accounted for as a single unit of accounting, the Company must determine the period over which the performance obligations will be performed and revenue will be recognized, to the extent this is determinable. If the timing and the level of effort to complete performance obligations under the arrangement is not estimable, then the Company recognizes revenue under the arrangement on a straight-line basis over the period that the Company expects to complete such performance obligations.
The Company’s license agreements with Pharmstandard International S.A. (“Pharmstandard”), Medinet Co., Ltd. (“Medinet”), Green Cross Corp. (“Green Cross”) and Lummy (Hong Kong) Co. Ltd. (“Lummy HK”) contain, and any future license agreements it enters into may also contain, milestone payments. Revenues from milestones, if they are non-refundable and considered substantive, are recognized upon successful accomplishment of the milestones. If not considered substantive, milestones are initially deferred and recognized over the remaining performance obligation. Pharmstandard is considered a related party based on Pharmstandard’s ownership of stock of the Company.
The Company’s current license agreements with Pharmstandard, Medinet, Green Cross and Lummy HK provide for, and any future license agreements the Company may enter into may provide for royalty payments. Royalty revenue is recognized upon the sale of the related products, provided there are no remaining performance obligations under the arrangement. To date, the Company has not received any royalty payments.
In September 2006, the Company entered into a multi-year research contract with the National Institutes of Health (“NIH”) and the National Institute of Allergy and Infectious Diseases (“NIAID”) to design, develop and clinically test an autologous HIV immunotherapy capable of eliciting therapeutic immune responses. The Company is using funds from this contract to develop AGS-004. Under this contract, as amended, the NIH and NIAID have committed to fund up to a total of $39.8 million, including reimbursement of direct expenses and allocated overhead and general and administrative expenses of up to $38.4 million and payment of other specified amounts totaling up to $1.4 million upon the Company’s achievement of specified development milestones. Since September 2010, the Company has received reimbursement of its allocated overhead and general and administrative expenses at provisional indirect cost rates equal to negotiated provisional indirect cost rates agreed to with the NIH and NIAID in September 2010. These provisional indirect cost rates are subject to adjustment based on the Company’s actual costs pursuant to the agreement with the NIH and NIAID. This commitment originally extended until May 2013. The Company agreed to an additional modification of the Company’s contract with the NIH and NIAID under which the NIH and NIAID agreed to increase their funding commitment to the Company by an additional $5.4 million in connection with the extension of the contract from May 2013 to September 2015. Additionally, a contract modification for a $0.5 million increase was agreed to by the NIH on September 18, 2014 to cover a portion of the manufacturing costs of the planned Phase 2 clinical trial of AGS-004 for long-term viral control in pediatric patients. This modification extended the NIH and NIAID’s commitment under the contract to July 2016. The Company has agreed to a statement of work under the contract, and is obligated to furnish all the services, qualified personnel, material, equipment, and facilities, not otherwise provided by the U.S. government, needed to perform the statement of work.
The Company recognizes revenue from reimbursements earned in connection with the contract as reimbursable costs are incurred and revenues from the achievement of milestones under the NIH and NIAID contract upon the accomplishment of any such milestone.
For the years ended December 31, 2013, 2014 and 2015, the Company recorded revenue under this agreement of $4,421,689, $1,797,054 and $448,273, respectively. The Company has recorded total revenue of $37.3 million through December 31, 2015 under the NIH and NIAID agreement. As of December 31, 2015, there was up to $2.5 million of potential revenue remaining to be earned under the agreement with the NIH and NIAID. As of December 31, 2014 and 2015, the Company recorded a receivable from the NIH and NIAID of $129,019 and $73,818, respectively. The concentration of credit risk is equal to the outstanding accounts receivable and such risk is subject to the credit worthiness of the NIH and NIAID. There have been no credit losses under this arrangement.
Income Taxes
The Company provides for income taxes using the asset and liability method. Under this method, deferred tax assets and liabilities are determined based on differences between financial reporting and tax bases of assets and liabilities, and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. Valuation allowances are provided if, based upon the weight of available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
| F-10 |
Segment and Geographic Information
Operating segments are defined as components of an enterprise engaging in business activities from which it may earn revenues and incur expenses, for which discrete financial information is available and whose operating results are regularly reviewed by the chief operating decision maker in deciding how to allocate resources and in assessing performance. The Company views its operations and manages its business in one operating segment and all of the Company operations are in North America.
Research and Development
Research and development costs include all direct costs related to the development of the Company’s technology, including salaries and related benefits of research and development (“R&D”) personnel, depreciation of laboratory equipment, fees paid to consultants and contract research organizations, stock-based compensation for R&D personnel, sponsored research payments and license fees. R&D costs are expensed as incurred.
Redeemable Convertible Preferred Stock
The carrying value of redeemable convertible preferred stock was increased by periodic accretions so that the carrying amount would equal the redemption amount as of the redemption date. These increases were recorded through charges against additional paid-in capital, to the extent it was available, or the accumulated deficit.
Stock-Based Compensation
The Company estimates the grant date fair value of its share-based awards and amortizes this fair value to compensation expense over the requisite service period or vesting term (see Note 10).
Investment Tax Credits
Other income of $0, $140,556 and $0 was recognized during the years ended December 31, 2013, 2014 and 2015, respectively, for scientific research and experimental development (“SR&ED”) investment tax credits in Canada. Under Canadian and Ontario law, the Company’s Canadian subsidiary is entitled to SR&ED. Because these credits are subject to a claims review, the Company recognizes such credits when received.
Comprehensive Income (Loss)
ASC 220, Comprehensive Income, establishes standards for reporting and display of comprehensive income and its components in a full set of financial statements. The Company’s other comprehensive income (loss) is related to foreign currency translation adjustments and unrealized gain (loss) on short-term investments.
Foreign Currency Translation
Gains and losses from foreign currency transactions are reflected in income currently.
The Company has identified the functional currency of its subsidiaries with foreign operations as the applicable local currency. The translation from the applicable local currency to United States dollars is performed using the exchange rate in effect as of the balance sheet date. Revenue and expense accounts are translated using the average exchange rate experienced during the period. Adjustments resulting from the translation of the Company’s subsidiaries’ financial statements from its functional currency to the United States dollar are not included in determining net loss, but are reported as accumulated other comprehensive gain (loss), a separate component of stockholders’ equity (deficit).
Interest Expense
During the years ended December 31, 2014 and 2015, interest expense primarily resulted from accrued interest on our note payable to Medinet, which was issued in December 2013, and interest from a venture loan and security agreement entered into in September 2014 with two financial institutions (see Note 5).
| F-11 |
Recently Issued Accounting Pronouncements
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842) ("ASU 2016-02"). The provisions of ASU 2016-02 set out the principles for the recognition, measurement, presentation and disclosure of leases for both parties to a contract (i.e. lessees and lessors). The new standard requires lessees to apply a dual approach, classifying leases as either finance or operating leases based on the principle of whether or not the lease is effectively a financed purchase by the lessee. This classification will determine whether lease expense is recognized based on an effective interest method or on a straight line basis over the term of the lease, respectively. A lessee is also required to record a right-of-use asset and a lease liability for all leases with a term of greater than 12 months regardless of their classification. Leases with a term of 12 month or less will be accounted for similar to existing guidance for operating leases today. Topic 842 supersedes the previous leave standard, Topic 840 Leases. The guidance is effective for annual periods and interim periods within those annual periods beginning after December 15, 2018, and is effective for the Company for the year ending December 31, 2019. The Company is currently evaluating the impact that the implementation of this standard will have on the Company's consolidated financial statements.
In November 2015, the FASB issued ASU No. 2015-17, Income Taxes (Topic 740): Balance Sheet Classification of Deferred Taxes. The new standard requires that deferred tax assets and liabilities be classified as noncurrent in a classified statement of financial position. The guidance is effective for financial statements issued for annual periods beginning after December 15, 2016, and interim periods within those annual periods. Earlier application is permitted for all entities as of the beginning of an interim or annual report period. The amendments in this ASU may be applied either prospectively to all deferred tax assets and liabilities or retrospectively to all periods presented. We adopted this standard as of December 31, 2015 with prospective application. As a result, we reclassified our deferred tax assets classified as current to noncurrent and our deferred tax liabilities classified as current to noncurrent in our December 31, 2015 consolidated balance sheet. Prior balance sheets were not retrospectively adjusted.
In May 2015, the FASB issued a new standard which removes the requirement to categorize the investments for which fair value is measured using net asset value per share as a practical expedient for fair value within the fair value hierarchy. This standard is effective for the Company for reporting periods beginning after December 15, 2015 and is to be applied retrospectively; early adoption is permitted. The adoption of this standard is not expected to have a material impact on the consolidated financial statements.
In April 2015, the FASB issued a new standard update which requires debt issuance costs to be presented in the balance sheet as a direct deduction from the associated debt liability. This standard is effective for the Company beginning January 1, 2016. The new guidance will be applied on a retrospective basis. The adoption of this standard is not expected to have a material impact on the consolidated financial statements.
In August 2014, the FASB issued a new standard update that specifies the responsibility that an entity’s management has to evaluate whether there is substantial doubt about the entity’s ability to continue as a going concern. The standard is effective for interim and annual periods beginning after December 15, 2016, and is not expected to have an effect on the Company’s consolidated financial statements.
In May 2014, the FASB issued a new accounting standard update pertaining to accounting for revenue from contracts with customers. The new guidance clarifies the principles for recognizing revenue and develops a common revenue standard for U.S. GAAP. The standard outlines a single comprehensive model for entities to use in accounting for revenue arising from contracts with customers and supersedes the most current revenue recognition guidance. In July 2015, the FASB finalized a one year delay in the effective date of this standard, which will now be effective for the Company beginning January 1, 2018, however early adoption is permitted any time after the original effective date, which for the Company is January 1, 2017. The Company is currently evaluating the impact that the implementation of this standard will have on the Company’s consolidated financial statements.
2. Fair Value of Financial Instruments
The estimated fair values of all of the Company’s financial instruments, excluding long-term debt, approximate their carrying amounts in the consolidated balance sheets as of December 31, 2014 and 2015.
As of December 31, 2014 and 2015, the Company held certain assets that are required to be measured at fair value on a recurring basis. These assets include money market funds included in cash equivalents and short-term investments in corporate debt securities. The Company’s short-term investments are classified as available-for sale investments. As of December 31, 2015, the Company’s only short-term investment in a corporate debt security had a contractual maturity of January 25, 2016. The valuation of these financial instruments uses a three tiered approach, which requires that fair value measurements be classified and disclosed in one of three tiers. These tiers are: Level 1, defined as quoted prices in active markets for identical assets or liabilities; Level 2, defined as valuations based on observable inputs other than those included in Level 1, such as quoted prices for similar assets and liabilities in active markets, or other inputs that are observable or can be corroborated by observable input data; and Level 3, defined as valuations based on unobservable inputs reflecting the Company’s own assumptions, consistent with reasonably available assumptions made by other market participants.
| F-12 |
The Company’s Level 1 assets consist of money-market funds and restricted cash in a deposit account at a bank. The method used to estimate the fair value of the Level 1 assets is based on observable market data, as these money-market funds are publicly-traded. The Company’s Level 2 assets consist of short-term debt instruments in corporate debt securities valued using independent pricing services which normally derive security prices from recently reported trades for identical or similar securities, making adjustments based on significant observable transactions. As of each balance sheet date, observable market inputs may include trade information, broker or dealer quotes, bids, offers or a combination of these data sources. During the years ended December 31, 2014 and 2015, there were no transfers between Levels 1 and 2 assets.
As of December 31, 2014 and 2015, these financial instruments and respective fair values have been classified as follows:
| Quoted Prices in Active Markets for Identical Assets (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Balance as of December 31, 2014 | |||||||||||||
| Assets | ||||||||||||||||
| Money-market funds | $ | 35,541,595 | $ | — | $ | — | $ | 35,541,595 | ||||||||
| Corporate debt securities – short-term | — | 20,266,243 | — | 20,266,243 | ||||||||||||
| Restricted cash – long-term | 1,325,000 | 1,325,000 | ||||||||||||||
| Total assets at fair value | $ | 36,866,595 | $ | 20,266,243 | $ | — | $ | 57,132,838 | ||||||||
| Quoted Prices in Active Markets for Identical Assets (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | Balance as of December 31, 2015 | |||||||||||||
| Assets | ||||||||||||||||
| Money-market funds | $ | 5,589,684 | $ | — | $ | — | $ | 5,589,684 | ||||||||
| Corporate debt securities – short-term | — | 1,003,160 | — | 1,003,160 | ||||||||||||
| Restricted cash – long-term | 740,000 | 740,000 | ||||||||||||||
| Total assets at fair value | $ | 6,329,684 | $ | 1,003,160 | $ | — | $ | 7,332,844 | ||||||||
The amortized cost, gross unrealized holding gains, gross unrealized holding losses, and estimated fair value of money-market funds included in cash and cash and equivalents, available-for-sale corporate debt securities included in short-term investments and cash and cash equivalents, and restricted cash as of December 31, 2014 and 2015 were as follows:
| As of December 31, 2014 | ||||||||||||||||
| Amortized Cost Basis | Gross Unrealized Holding Gains | Gross Unrealized Holding Losses | Aggregate Fair Value | |||||||||||||
| Money-market funds | $ | 35,541,595 | $ | — | $ | — | $ | 35,541,595 | ||||||||
| Corporate debt securities – short term | 20,278,171 | 67 | (11,995 | ) | 20,266,243 | |||||||||||
| Restricted cash – long-term | 1,325,000 | — | — | 1,325,000 | ||||||||||||
| $ | 57,144,766 | $ | 67 | $ | (11,995 | ) | $ | 57,132,838 | ||||||||
| F-13 |
| As of December 31, 2015 | ||||||||||||||||
| Amortized Cost Basis | Gross Unrealized Holding Gains | Gross Unrealized Holding Losses | Aggregate Fair Value | |||||||||||||
| Money-market funds | $ | 5,589,684 | $ | — | $ | — | $ | 5,589,684 | ||||||||
| Corporate debt securities – short term | 1,003,431 | — | (271 | ) | 1,003,160 | |||||||||||
| Restricted cash – short-term | 740,000 | — | — | 740,000 | ||||||||||||
| $ | 7,333,115 | $ | — | $ | (271 | ) | $ | 7,332,844 | ||||||||
The fair value of the Company’s long-term debt was derived by evaluating the nature and terms of each note, considering the prevailing economic and market conditions as of each balance sheet date and based on the Level 2 valuation hierarchy of the fair value measurements standard using a present value methodology. The fair value of the Company’s long-term debt as of December 31, 2014 was approximately $19.4 million compared with its carrying value of $19.8 million. The fair value of the Company’s debt as of December 31, 2015 was approximately $32.0 million compared with its carrying value of $32.4 million.
3. Property and Equipment
Property and equipment consist of the following as of December 31, 2014 and 2015:
| Useful Life (Years) | 2014 | 2015 | ||||||||||
| Office furniture and equipment | 7 | $ | 458,380 | $ | 681,954 | |||||||
| Computer equipment | 3 | 788,563 | 970,689 | |||||||||
| Computer software | 3 | 629,948 | 629,948 | |||||||||
| Laboratory equipment | 7 | 5,345,786 | 5,916,539 | |||||||||
| Leasehold improvements | 5 | 2,646,891 | 2,664,669 | |||||||||
| Assets related to facility lease obligation | 3,380,223 | 7,630,660 | ||||||||||
| Construction-in-progress | 500,093 | 12,656,260 | ||||||||||
| Total property and equipment, net | 13,749,884 | 31,150,719 | ||||||||||
| Less: Accumulated depreciation and amortization | (8,236,329 | ) | (8,844,335 | ) | ||||||||
| Property and equipment, net | $ | 5,513,555 | $ | 22,306,384 | ||||||||
Assets related to the Company’s facility lease obligation and construction-in-progress were recognized primarily due to the Company being deemed to be the accounting owner of the facility being built to be the Company’s headquarters and primary manufacturing facility during its construction period under build-to-suite lease accounting (see Note 7). As of December 31, 2015, construction-in-progress included $880,334 of capitalized interest.
Depreciation and amortization expense was as follows:
| Year ended December 31, 2013 | $ | 611,421 | ||
| Year ended December 31, 2014 | 566,117 | |||
| Year ended December 31, 2015 | 680,444 |
4. Income Taxes
No provision for U.S. federal, state or foreign income taxes has been recorded as the Company has incurred net operating losses since its inception in 1997.
| F-14 |
Significant components of the Company’s deferred tax assets and liabilities consist of the following:
| December 31, | ||||||||
| 2014 | 2015 | |||||||
| U.S. federal and state net operating loss carryforwards | $ | 59,689,264 | $ | 74,268,811 | ||||
| Foreign net operating loss carryforwards | 1,743,603 | 1,451,968 | ||||||
| Contribution carryforwards | 4,245 | 4,245 | ||||||
| Research and development credits | 5,253,055 | 6,748,213 | ||||||
| Investment tax credits | 35,108 | 29,433 | ||||||
| Stock-based compensation | 462,796 | 847,475 | ||||||
| Patents and other intangibles | 611 | — | ||||||
| Other accruals | 125,614 | 710,248 | ||||||
| Deferred revenue | 372,040 | 365,896 | ||||||
| Property and equipment | 510,466 | 476,297 | ||||||
| Total deferred tax assets | 68,196,802 | 84,902,586 | ||||||
| Valuation allowance for deferred assets | (68,196,802 | ) | (84,902,586 | ) | ||||
| Net deferred tax assets | $ | — | $ | — | ||||
As of December 31, 2014 and 2015, the Company provided a full valuation allowance against its net deferred tax assets since as of that time, the Company could not assert that it was more likely than not that these deferred tax assets would be realized. There was an increase in the valuation allowance in the year ended December 31, 2015 of $16,705,784, all of which was allocable to current operating activities.
As of December 31, 2015, the Company had U.S. federal and state, and Canadian federal and provincial net operating loss carryforwards of approximately $199,443,200, $244,608,100, $5,479,100, and $5,479,100, respectively. These net operating loss carryforwards begin to expire in 2018, 2017, 2016 and 2016, respectively. As of December 31, 2015, the Company had U.S. federal and state tax credit carryforwards of approximately $9,319,400 and $340,400, respectively. These credit carryforwards begin to expire in 2020 and 2024, respectively. As of December 31, 2015, the Company had Canadian investment tax credit carryforwards of approximately $29,400 that begin to expire in 2024. The utilization of the net operating loss and tax credit carryforwards may be subject to limitation under the rules regarding a change in stock ownership as determined by the Internal Revenue Code, and state and foreign tax laws. Section 382 of the Internal Revenue Code of 1986, as amended, imposes annual limitations on the utilization of net operating loss (“NOL”) carryforwards, other tax carryforwards, and certain built-in losses upon an ownership change as defined under that section. In general terms, an ownership change may result from transactions that increase the aggregate ownership of certain stockholders in the Company’s stock by more than 50 percentage points over a three year testing period. If the Company has undergone a Section 382 ownership change, an annual limitation would be imposed on certain of the Company’s tax attributes, including NOL and capital loss carryforwards, and certain other losses, credits, deductions or tax basis. As of December 31, 2015, the Company has not completed a formal study to determine whether there are 382 limitations that apply.
As of December 31, 2015, the Company had no foreign unremitted earnings from DC Bio, its Canadian subsidiary.
| F-15 |
Taxes computed at the statutory U.S. federal income tax rate of 34.0% are reconciled to the provision for income taxes for the years ended December 31, 2013, 2014 and 2015 as follows:
| 2013 | 2014 | 2015 | ||||||||||||||||||||||
| Amount | Percent of Pretax Earnings | Amount | Percent of Pretax Earnings | Amount | Percent of Pretax Earnings | |||||||||||||||||||
| U.S. federal tax statutory rate | $ | (8,133,331 | ) | 34.0 | % | $ | (18,124,019 | ) | 34.0 | % | $ | (25,428,097 | ) | 34.0 | % | |||||||||
| State taxes (net of federal benefit) | (789,412 | ) | 3.3 | % | (1,707,912 | ) | 3.2 | % | (1,936,719 | ) | 2.6 | % | ||||||||||||
| U.S. federal research and development tax credits | (1,262,562 | ) | 5.3 | % | (1,701,727 | ) | 3.2 | % | (2,135,940 | ) | 2.9 | % | ||||||||||||
| Nondeductible interest expense | (132,546 | ) | 0.6 | % | — | 0.0 | % | — | 0.0 | % | ||||||||||||||
| Other nondeductible expenses | 419,934 | (1.8 | %) | 658,740 | (1.1 | %) | 1,032,480 | (1.4 | %) | |||||||||||||||
| Expiration of capital loss carryforward | — | (0.0 | %) | — | 0.0 | % | — | 0.0 | % | |||||||||||||||
| Increase in unrecognized tax benefits | 378,769 | (1.6 | %) | 510,516 | (1.0 | %) | 640,782 | (0.9 | %) | |||||||||||||||
| Change in effective state tax rate | 1,349,062 | (5.6 | %) | 1,685 | (0.0 | %) | 1,190,519 | (1.6 | %) | |||||||||||||||
| Expiration of NOL & contribution carryforwards | 6,630 | (0.0 | %) | 77,391 | (0.1 | %) | 9,590,101 | (12.8 | %) | |||||||||||||||
| Change in valuation reserves | 8,811,127 | (36.8 | %) | 20,252,063 | (38.1 | %) | 16,705,784 | (22.3 | %) | |||||||||||||||
| Deferred tax asset true-ups | (647,671 | ) | 2.6 | % | 33,263 | (0.1 | %) | 341,090 | (0.5 | %) | ||||||||||||||
| Provision for income taxes | $ | — | 0.0 | % | $ | — | 0.0 | % | $ | — | 0.0 | % | ||||||||||||
On July 23, 2013, North Carolina enacted House Bill 998, which reduced the corporate income tax rate from 6.9% in 2013 to 6% in 2014 and to 5% in 2015. As a result of the new enacted tax rate, the Company adjusted its deferred tax assets in 2013 by applying the lower rate, which resulted in a decrease to the deferred tax assets and a corresponding decrease to the valuation allowance of $1,349,062. On September 18, 2015, North Carolina enacted House Bill 97, which reduced the corporate income tax rate from 5% to 4% in 2016. As a result of the new enacted tax rate, the Company adjusted its deferred tax assets in 2015 by applying the lower rate, which resulted in a decrease in the deferred tax assets and a corresponding decrease to the valuation allowance of $1,190,519.
The Company had gross unrecognized tax benefits of approximately $2,155,000 as of January 1, 2015. As of December 31, 2015, the total gross unrecognized tax benefits were approximately $2,795,800 and of this total, none would affect the Company’s effective tax rate if recognized. The Company does not anticipate a significant change in total unrecognized tax benefits or the Company’s effective tax rate due to the settlement of audits or the expiration of statutes of limitations within the next 12 months.
The Company’s policy is to recognize interest and penalties related to uncertain tax positions in the provision for income taxes. As of December 31, 2014 and 2015, the Company had no accrued interest or penalties related to uncertain tax positions.
The Company has analyzed its filing positions in all significant federal and state jurisdictions where it is required to file income tax returns, as well as open tax years in these jurisdictions. With few exceptions, the Company is no longer subject to United States federal, state, and local tax examinations by tax authorities for years before 2012 although carryforward attributes that were generated prior to 2012 may still be adjusted upon examination by the Internal Revenue Service if they either have been or will be used in a future period. No income tax returns are currently under examination by taxing authorities.
| F-16 |
The following is a tabular reconciliation of the Company’s change in gross unrecognized tax positions during the years ended December 31, 2013, 2014 and 2015:
| 2013 | 2014 | 2015 | ||||||||||
| Beginning balance | $ | 1,265,700 | $ | 1,644,500 | $ | 2,155,000 | ||||||
| Gross increase for tax positions related to current periods | 378,800 | 474,300 | 640,800 | |||||||||
| Gross (decrease) increase for tax positions related to prior periods | — | 36,200 | — | |||||||||
| Ending balance | $ | 1,644,500 | $ | 2,155,000 | $ | 2,795,800 | ||||||
5. Notes Payable
Notes payable consist of the following as of December 31, 2014 and 2015:
| 2014 | 2015 | |||||||
| Notes payable under the venture loan and security agreement, including accrued interest | $ | 12,500,000 | $ | 25,265,451 | ||||
| Less related debt discount | (373,756 | ) | (401,726 | ) | ||||
| Notes payable under the venture loan and security agreement, net of debt discount | 12,126,244 | 24,863,725 | ||||||
| Promissory note payable to Medinet, including accrued interest | 7,623,546 | 7,489,565 | ||||||
| Other notes payable | 77,640 | 46,755 | ||||||
| Total notes payable | 19,827,430 | 32,400,045 | ||||||
| Less current portion | (30,885 | ) | (1,578,284 | ) | ||||
| Long-term portion of notes payable | $ | 19,796,545 | $ | 30,821,761 | ||||
Venture Loan Facility. On September 29, 2014, the Company entered into a venture loan and security agreement (the “Loan Agreement”) with Horizon Technology Finance Corporation and Fortress Credit Co LLC (together, the “Lenders”) under which the Company could borrow up to $25.0 million in two tranches of $12.5 million each (the “Loan Facility”).
The Company borrowed the first tranche of $12.5 million upon the closing of the transaction in September 2014 and borrowed the second tranche of $12.5 million on August 7, 2015. The per annum interest rate for each tranche is a floating rate equal to 9.25% plus the amount by which the one-month London Interbank Offered Rate (“LIBOR”) exceeds 0.50% (effectively a floating rate equal to 8.75% plus the one-month LIBOR Rate). The total per annum interest rate shall not exceed 10.75%.
The Company incurred $449,796 in costs in connection with the closing of the Loan Agreement. These costs were capitalized as deferred financing costs and amortized to interest expense over the terms of the related debt.
The Company has agreed to repay the first tranche of $12.5 million on an interest only basis monthly until September 30, 2016, followed by monthly payments of principal and accrued interest through the scheduled maturity date for the first tranche loan on September 30, 2018. In addition, a final payment for the first tranche loan equal to $625,000 will be due on September 30, 2018, or such earlier date specified in the Loan Agreement. The Company is recognizing the final payment of $625,000 as accrued interest over the expected life of the first tranche loan. The Company has agreed to repay the second tranche loan of $12.5 million in 18 monthly payments of interest only until February 7, 2017, followed by 24 monthly payments of principal and accrued interest through the scheduled maturity date for the second tranche loan on February 7, 2019. In addition, a final payment of $625,000 will be due on February 7, 2019, or such earlier date specified in the Loan Agreement. The Company is recognizing the final payment of $625,000 as accrued interest over the expected life of the second tranche loan. In addition, if the Company repays all or a portion of the loan prior to the applicable maturity date, it will pay the Lenders a prepayment penalty fee, based on a percentage of the then outstanding principal balance, equal to 3% if the prepayment occurs on or before 24 months after the funding date, 2% if the prepayment occurs more than 24 months after, but on or before 36 months after, the funding date thereof, or 1% if the prepayment occurs more than 36 months after the funding date thereof.
| F-17 |
The Company’s obligations under the Loan Agreement are secured by a first priority security interest in substantially all of its assets other than its intellectual property. The Company also has agreed not to pledge or otherwise encumber its intellectual property assets, subject to certain exceptions.
The Loan Agreement includes customary affirmative and restrictive covenants, but does not include any covenants to attain or maintain certain financial metrics, and also includes customary events of default, including payment defaults, breaches of covenants, change of control and a material adverse change default. Upon the occurrence of an event of default and following any applicable cure periods, a default interest rate of an additional 5% may be applied to the outstanding loan balances, and the Lenders may declare all outstanding obligations immediately due and payable and take such other actions as set forth in the Loan Agreement.
In connection with the Loan Agreement, the Company issued to the Lenders and their affiliates warrants to purchase a total of 82,780 shares of Common Stock at a per share exercise price of $9.06 (the “Warrants”). Upon the Company’s satisfaction of the conditions precedent to the making of the second tranche loan, the Warrants became exercisable in full. The Warrants will terminate on September 29, 2021 or such earlier date as specified in the Warrants. The Company has recorded a debt discount of $338,673 equal to the value of these Warrants. This debt discount is offset against the note payable balance and included in additional paid-in capital on the Company's balance sheet.
Medinet Loan. In December 2013, in connection with a license agreement currently with Medinet Co., Ltd and its wholly-owned subsidiary, MEDcell Co., Ltd. (together "Medinet"), as described in Note 12, the Company borrowed $9.0 million pursuant to an unsecured promissory note that bears interest at a rate of 3.0% per annum. The principal and interest under the note are due and payable on December 31, 2018. Under the terms of the note and the license agreement, any milestone payments related to the developmental and regulatory milestones that become due will be applied first to the repayment of the loan. The Company has the right to prepay the loan at any time. If the Company has not repaid the loan by December 31, 2018, then the Company has agreed to grant to Medinet a non-exclusive, royalty-bearing license to make and sell Arcelis products in Japan for the treatment of cancer. In such event, the amounts owing under the loan as of December 18, 2018 may constitute pre-paid royalties under the license or would be due and payable. Royalties under this license would be paid until the expiration of the licensed patent rights in Japan at a rate to be negotiated. If the Company and Medinet cannot agree on the royalty rate, they have agreed to submit the matter to arbitration. Because the $9.0 million promissory note was issued at a below market interest rate, the Company allocated the proceeds of the loan between the license agreement and the debt at the time of issuance. Accordingly, as of the borrowing date, December 31, 2013, the Company recorded $6.9 million to notes payable, based upon an effective interest rate of 8.0%, and $2.1 million as a deferred liability.
During the year ended December 31, 2015, the Company recognized a $1.0 million milestone payment as deferred revenue under the Medinet license agreement and reduced the related note payable by $0.8 million and the deferred liability by $0.2 million. As of December 31, 2015, the amount of the note payable was $7.5 million, including $1.3 million of accrued interest. As of December 31, 2015, the total deferred liability associated with the Medinet note was $3.8 million (see Note 12).
Other Notes. The Company entered into a Master Lease Agreement in July 2012 with a lending institution, which provided for the Company to borrow funds up to $100,000 to finance computer equipment. The Company borrowed a total of $95,756 under this agreement, of which $16,356 and $0 was outstanding as of December 31, 2014 and 2015, respectively. The loans under this agreement were repaid in full in July 2015. Borrowings were collateralized by substantially all of the computer equipment financed under the agreement, with interest at a rate of 0.98% per annum and were repaid in 36 equal monthly installments commencing on the date of borrowing.
During November 2013, the Company borrowed $77,832 from a lending institution to finance the purchase of additional computer equipment, of which $61,284 and $46,754 in principal was outstanding as of December 31, 2014 and 2015, respectively. Borrowings are collateralized by substantially all of the computer equipment financed under the agreement, bear interest at a rate of 8.31% per annum and are to be repaid in 60 equal monthly installments commencing on the date of borrowing.
| F-18 |
6. Manufacturing Research and Development Obligation
In October 2014, the Company entered into a development agreement (the “Invetech Development Agreement”) with Invetech Pty Ltd (“Invetech”). The Invetech Development Agreement supersedes and replaces the development agreement entered into by the parties in July 2005. Under the Invetech Development Agreement, Invetech will continue to develop and provide prototypes of the automated production system to be used for the manufacture of the Company’s Arcelis-based products, or the Production Systems. Development services will be performed on a proposal by proposal basis.
Invetech has agreed to defer 30% of its fees, but such deferral will not exceed $5,000,000. Deferred fees (plus interest of 7% per annum) would become payable either, at the Company’s option, in a lump sum within 90 days of the “Sunset Date Trigger Event” or pursuant to an installment plan (either in four installments payable within the first year or eight installments payable within the first two years after the “Sunset Date Trigger Event”). The “Sunset Date Trigger Event” is June 30, 2016 if the ADAPT trial is closed early indicating positive efficacy, or is otherwise December 31, 2016. Invetech is entitled to a 10% bonus payment if the ADAPT trial is closed early indicating positive efficacy or if the ADAPT trial meets the primary endpoint of overall survival and the 100% of events analysis indicating positive efficacy, and Invetech has timely completed all activities up to the time the ADAPT trial is stopped.
As of December 31, 2015, the Company recorded the long-term portion of this manufacturing research and development obligation on its consolidated balance sheet at $7.8 million, representing $5.2 million in deferred fees, $2.3 million in estimated bonus payments and $0.3 million in accrued interest. As of December 31, 2014, the Company recorded a long-term portion of this manufacturing research and development obligation on its consolidated balance sheet totaling $3.5 million representing $2.5 million in deferred fees, $0.9 million in estimated bonus payments and $34,330 in accrued interest.
The Invetech Development Agreement requires the parties to discuss in good faith Invetech’s supply of Production Systems for use in manufacturing commercial product. The Company has an obligation to purchase $25.0 million worth of Production Systems, components, subsystems and spare parts for commercial use. Once that obligation has been satisfied, the Company has the right to have a third party supply Production Systems for use in manufacturing commercial product provided that Invetech has a right of first refusal with respect to any offer by a third party and the Company may not accept an offer from a third party unless that offer is at a price that is less than that offered by Invetech and otherwise under substantially the same or better terms. The Company will own all intellectual property arising from the development services (with the exception of existing Invetech intellectual property incorporated therein-under which the Company will have a license). The Invetech Development Agreement will continue until the completion of the development of the Production Systems. The Invetech Development Agreement can be terminated early by either party because of a technical failure or by the Company without cause.
7. Facility Lease Obligation
In August 2014, the Company entered into a Lease Agreement (the “Lease Agreement”) with TKC LXXII, LLC, a North Carolina limited liability company (“TKC”).
Under the Lease Agreement, the Company agreed to lease certain land and an approximately 124,000 square-foot building to be constructed on approximately 11 acres in Durham County, North Carolina. This facility is being built to potentially house the Company’s corporate headquarters and primary manufacturing facility. The lease for the current facility expires in November 2016. The shell of the new facility was constructed on a build-to-suit basis by TKC in accordance with agreed upon specifications and plans as set forth in the Lease Agreement at the expense of TKC, other than those costs resulting from changes requested by the Company for which the Company has paid $1.7 million as of December 31, 2015.
The term of the Lease Agreement is 10 years from the commencement date of July 1, 2015. The Company has an option to extend the Lease Agreement by six five-year renewal terms. Initial rent is $46,917 per month, subject to certain fixed increases over the course of the term as set forth in the Lease Agreement and to adjustment based on the Company’s use of certain amounts allocated for upfitting the interior of the facility. Rent expense was $292,095 during the year ended December 31, 2015.
The Lease Agreement required the Company to provide the landlord with a letter of credit. The Company provided the bank that issued the letter of credit on its behalf a security deposit of $1,325,000 to guarantee the letter of credit. In accordance with the Lease Agreement, this deposit was reduced to $740,000 as of December 31, 2015 due to the Company’s intention to acquire the facility under a purchase and sale agreement with the landlord. The deposit is recorded as restricted cash as of December 31, 2014 and 2015 on the Company’s consolidated balance sheets.
| F-19 |
Under the Lease Agreement, the Company is involved in the construction of the building. To the extent the Company is involved with the structural improvements of the construction project or takes construction risk prior to the commencement of a lease, ASC 840-40-05-5 requires for accounting purposes that the Company be considered the owner of this project during the construction period. Therefore, the Company recorded an asset in property and equipment, net on the consolidated balance sheets, for the cost of the Company’s portion of the building plus the amount of estimated structural construction costs incurred by the landlord and the Company as of the balance sheet date. The Company recorded a corresponding facility lease obligation on its consolidated balance sheets representing the amounts paid by TKC.
The initial recording of these assets and liabilities is classified as non-cash investing and financing items, respectively, for purposes of the consolidated statements of cash flows. The Company recorded an asset related to the facility lease obligation included in property and equipment of $3.4 million and $7.6 million as of December 31, 2014 and 2015, respectively. The facility lease obligation on the Company’s consolidated balance sheet is $3.4 million and $7.2 million as of December 31, 2014 and 2015, respectively.
Under the Lease Agreement, the Company had an option to purchase the property. In February 2015, the Company exercised this purchase option and entered into a Purchase and Sale Agreement (the “Purchase Agreement”) with TKC. The purchase price to be paid by the Company is $7.6 million plus the amount of any additional costs incurred by TKC as a result of changes requested by the Company, for which the Company has paid $1.7 million as of December 31, 2015, and the amount of any improvement allowances advanced to the Company by TKC prior to the closing. Under the terms of the Purchase Agreement, the Company had until February 16, 2016 to consummate the purchase of the property. The Company is currently in discussions with TKC to extend the period for purchasing the property as the Company will need to obtain additional funding to consummate the purchase of the property. If the Company purchases the property, upon the closing, the lease agreement will terminate. If TKC is unwilling to extend the period, TKC will have no further obligation to sell the property to the Company.
If the purchase of the property is not consummated, future minimum payments due under the Lease Agreement are as follows as of December 31, 2015:
| Year ending December 31: | ||||
| 2016 | $ | 569,334 | ||
| 2017 | 582,144 | |||
| 2018 | 595,242 | |||
| 2019 | 608,635 | |||
| 2020 | 622,329 | |||
| Thereafter | 2,976,440 | |||
| Total future minimum lease payments | $ | 5,954,124 | ||
8. Common Stock, Preferred Stock and Redeemable Convertible Preferred Stock
Issuance of Common Stock in 2015
In connection with the Company’s entry into the Lummy License Agreement (see Note 12), on April 7, 2015, the Company entered into stock purchase agreements with Tianyi Lummy International Holdings Group, Ltd. and China BioPharma Capital I, L.P. (the “Lummy Entities”), of which Lummy (Hong Kong) Co. Ltd.’s parent company is an affiliate and limited partner, respectively. Pursuant to the purchase agreements, the Lummy Entities purchased an aggregate of 1,000,000 shares of the Company’s common stock at a per share price of $10.11. The closing price of the Company’s common stock on April 7, 2015 was $8.57 per share, or approximately 18% lower than the $10.11 purchase price per share. The cash proceeds received of $10,110,000 from the issuance of the Company’s common stock were allocated $8,570,000 to common stock and additional paid-in capital and $1,540,000 representing the premium to fair market value paid by the Lummy Entities to deferred revenue attributable to the Lummy License Agreement.
| F-20 |
The Lummy Entities have also agreed to purchase approximately $10.0 million in additional shares of the Company’s common stock, for a total aggregate investment of approximately $20.0 million, within 31 days of and subject to the Company reaching full enrollment of the ADAPT trial of AGS-003 for mRCC, receiving a recommendation of the review board for the continuation of the ADAPT trial following 50% of events and receiving positive feedback from the FDA on a qualified protocol to demonstrate comparability of the Company’s automated manufacturing process for AGS-003 to the manufacturing process used by Company in its ADAPT trial. However, on March 4, 2016, the Company entered into a letter agreement with each of the Lummy Entities pursuant to which the Company agreed that upon their purchase of shares and warrants in the PIPE Financing (see Note 17) they would have no further obligation to purchase shares pursuant to the purchase agreements.
On December 22, 2015, the Company entered into a Master Process Development and Supply Agreement with Cellscript, LLC (“Cellscript”). Under the agreement, Cellscript has agreed to develop cGMP processes for the manufacture and production of CD40L RNA, a ribonucleic acid used in the production of the Company’s Arcelis-based products, and to manufacture and produce CD40L RNA for the Company, in each case in accordance with the agreement and a project work agreement previously agreed to by the Company and Cellscript.
In consideration for these development and production services, the Company has agreed to pay Cellscript total fees of $4,600,000. Upon the execution of the agreement and in exchange for research and development services, the Company made a payment to Cellscript of $2,111,432 through the issuance to Cellscript of 906,194 shares of the Company’s common stock. The balance of the owed fees are payable to Cellscript, at the Company’s option, in cash, common stock or a combination of cash and common stock upon the achievement of development milestones. Any shares of common stock issued pursuant to the Cellscript agreement are subject to a lock-up period of 180 days from the date of issuance of such shares to Cellscript.
Initial Public Offering and Issuance of Common Stock in 2014
In February 2014, the Company issued and sold 6,228,725 shares of its common stock, including 603,725 shares of common stock sold pursuant to the underwriters’ exercise of their option to purchase additional shares, in the Company’s initial public offering, at a public offering price of $8.00 per share, for aggregate gross proceeds of $49.8 million. The net offering proceeds to the Company, after deducting underwriting discounts and commissions of approximately $3.5 million and offering expenses of approximately $2.9 million, were approximately $43.4 million. Upon the closing of the initial public offering, all of the then-outstanding shares of the Company’s redeemable convertible preferred stock automatically converted into 13,188,251 shares of common stock. Accordingly, as of December 31, 2014 and 2015, the Company had no redeemable convertible preferred stock outstanding and no shares of preferred stock have been issued since their authorization in 2014.
Redeemable Convertible Preferred Stock
In August 2013, the Company sold 16,898,436 shares of series E preferred stock for an aggregate purchase price of $22,007,404.
In October 2013 and November 2013, the Company sold 921,423 shares of its series E preferred stock for an aggregate purchase price of $1,200,000 and 19,037,063 shares of its series E preferred stock for an aggregate purchase price of $24,792,595, respectively.
9. Warrants
In connection with the issuance and sale of series E preferred stock, in December 2013, the Company issued a warrant to purchase 9,598 shares of its common stock, at an exercise price of $6.60 per share, to a placement agent. During the year ended December 31, 2014, warrants to purchase 9,598 shares of the Company’s common stock at $6.60 per share were settled in a cashless exercise for 1,679 shares of common stock in conjunction with the closing of the Company’s initial public offering in February 2014.
As discussed in Note 6 regarding the Company’s notes payable, in connection with the Loan Agreement, the Company issued to the Lenders and their affiliates the Warrants to purchase a total of 82,780 shares of Common Stock at a per share exercise price of $9.06. Upon the Company’s satisfaction of the conditions precedent to the making of the second tranche loan, the Warrants became exercisable in full. The Warrants will terminate on September 29, 2021 or such earlier date as specified in the Warrants.
In conjunction with entering into a loan agreement with a bank in December 2000, the Company issued warrants to purchase shares of its common stock, all of which expired as of December 31, 2012, except for one warrant. This warrant to purchase one share of the Company’s common stock, at an exercise price of $23,894.34 per share, remained outstanding as of December 31, 2014 and 2015.
| F-21 |
Outstanding warrants to purchase the Company’s common stock as of December 31, 2014 and 2015 were as follows.
| Type of Warrant | Number of Shares | Exercise Price | Expiration Date(s) | |||||||
| Common stock | 82,780 | $ | 9.06 | 9/29/21 | ||||||
| Common stock | 1 | $ | 23,894.34 | 7/13/16 | ||||||
In November 2013, the Company entered into an agreement with Pharmstandard under which Pharmstandard purchased additional shares of the Company’s series E preferred stock. Upon the closing of the Company’s initial public offering, all of the outstanding shares of redeemable convertible preferred stock automatically converted into 13,188,251 shares of the Company’s common stock. Under this agreement, the Company agreed to enter into a manufacturing rights agreement for the European market with Pharmstandard and that the manufacturing rights agreement would provide for the issuance of warrants to Pharmstandard to purchase 499,788 shares of the Company’s common stock at an exercise price of $5.82 per share. As of March 30, 2016, the Company had not entered into this manufacturing rights agreement or issued the warrants.
10. Stock Options and Employee Stock Purchase Plan
2014 Stock Incentive Plan and 2014 Employee Stock Purchase Plan
In January 2014, the Company’s board of directors and stockholders approved, effective upon the closing of the Company’s initial public offering, the 2014 Stock Incentive Plan (the “2014 Plan”). Under the 2014 Plan, the Company may grant incentive stock options, nonstatutory stock options, stock appreciation rights, restricted stock, restricted stock units and other stock-based awards for the purchase of that number of shares of Common Stock equal to the sum of 1,951,182 shares, plus such number of shares, up to 357,841 shares, as is equal to the sum of the number of shares reserved for issuance under the Company’s 2008 Stock Incentive Plan (the “2008 Plan”) that remained available for grant under the 2008 Plan immediately prior to the closing of the Company’s initial public offering on February 12, 2014 (381,250 shares) and the number of shares subject to outstanding awards under the 2008 Plan that expire, terminate or are otherwise surrendered, cancelled, forfeited or repurchased by the Company at their original issuance price pursuant to a contractual repurchase right, plus an annual increase, to be added on the first day of each fiscal year from January 1, 2015 through January 1, 2024, equal to the lowest of 2,309,023 shares of Common Stock, 4% of the number of the Company’s outstanding shares on the first day of each such fiscal year and an amount determined by the Company’s board of directors.
Also in January 2014, the Company’s board of directors and stockholders approved, effective upon the closing of the Company’s initial public offering, a 2014 Employee Stock Purchase Plan (the “2014 ESPP”). Under the 2014 ESPP, on the offering commencement date of each plan period (the “Purchase Plan Period”), the Company will grant to each eligible employee who is then a participant in the 2014 ESPP an option to purchase shares of Common Stock. The employee may authorize up to a maximum of 10% of his or her base pay to be deducted by the Company during the Purchase Plan Period. Each employee who continues to be a participant in the 2014 ESPP on the last business day of the Purchase Plan Period is deemed to have exercised the option, to the extent of accumulated payroll deductions within the 2014 ESPP ownership limits.
Under the terms of the 2014 ESPP, the option exercise price shall be determined by the Company’s board of directors for each Purchase Plan Period and the option exercise price will be at least 85% of the applicable closing price of the Common Stock. The option exercise price will be 85% of the lower of the Company’s closing stock price on the first and last business day of each Purchase Plan Period. The Company’s first Purchase Plan Period commenced on September 2, 2014 and ended on February 27, 2015. For the first Purchase Plan Period, 13,054 shares were purchased with employee withholdings at an option exercise price based upon 85% of the lower of the closing price at the beginning of the first Purchase Plan Period of $9.83 and the closing price on February 27, 2015 of $9.02, resulting in the recognition of a stock-based compensation expense of $54,508. The Company’s second Purchase Plan Period commenced on March 2, 2015 and ended on August 31, 2015. For the second Purchase Plan Period, 20,301 shares were purchased with employee withholdings at an option exercise price based upon 85% of the lower of the closing price at the beginning of the second Purchase Plan Period of $9.02 and the closing price on August 31, 2015 of $6.21, resulting in the recognition of a stock-based compensation expense of $72,800. The Company’s third Purchase Plan Period commenced on September 1, 2015 and will end on February 29, 2016. Based upon 85% of the lower of the closing price at the beginning of the third Purchase Plan Period of $6.24 and the closing price of $2.31 on December 31, 2015, stock-based compensation expense of $209,931 was recognized.
| F-22 |
Upon the exercise of stock options, vesting of other awards and purchase of shares through the 2014 ESPP or under the 2014 Plan, the Company issues new shares of common stock to the Company’s employees. All awards granted under the 2014 Plan that are canceled prior to vesting or expire unexercised are returned to the approved pool of reserved shares under the 2014 Plan and made available for future grants. As of December 31, 2015, there were 4,005,611 shares of common stock remaining available for future issuance under the 2014 Plan and 312,998 shares of common stock remaining available for future issuance under the 2014 ESPP.
The Company recorded the following stock-based compensation expense:
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| Research and development | $ | 524,495 | $ | 1,604,215 | $ | 2,104,401 | ||||||
| General and administrative | 528,668 | 1,409,068 | 1,910,537 | |||||||||
| Total stock-based compensation expense | $ | 1,053,163 | $ | 3,013,283 | $ | 4,014,938 | ||||||
Allocations to research and development and general and administrative expense are based upon the department to which the associated employee reported. No related tax benefits of the stock-based compensation expense have been recognized. Stock-based payments issued to nonemployees are recorded at their fair values, and are periodically revalued as the equity instruments vest and are recognized as expense over the related service period.
Valuation Assumptions for Stock Option Plans
The employee stock-based compensation expense recognized was determined using the Black-Scholes option valuation model. Option valuation models require the input of subjective assumptions and these assumptions can vary over time. The weighted average assumptions used were as follows:
| 2013 | 2014 | 2015 | ||||||||||
| Risk-free interest rate | 2.12 | % | 2.26 | % | 2.05 | % | ||||||
| Expected dividend yield | 0 | % | 0 | % | 0 | % | ||||||
| Expected option term (in years) | 7 | 7 | 7 | |||||||||
| Expected volatility | 94 | % | 96 | % | 87 | % | ||||||
The risk-free interest rate is based on the U.S. Treasury yield curve in effect on the date of grant. The dividend yield percentage is zero because the Company neither currently pays dividends nor intends to do so during the expected option term. The Company’s historical share option exercise experience does not provide a reasonable basis upon which to estimate an expected term because of a lack of sufficient data. Therefore the Company estimates the expected term by using the simplified method allowed by the SEC. Expected stock price volatility is based on an average of several peer public companies. For purposes of identifying peer companies, the Company considered characteristics such as industry, length of trading history, similar vesting terms and in the-money option status. Upon the adoption of ASC 718, the Company was also required to estimate the level of forfeitures expected to occur and record compensation expense only for those awards that ultimately expect to vest. The Company performed a historical analysis of option awards that were forfeited prior to vesting and recorded total stock option expense that reflected this estimated forfeiture rate. The weighted average grant date fair value of stock options was $4.77, $5.54 and $5.76 in the years ended December 31, 2013, 2014 and 2015, respectively.
| F-23 |
The following table summarizes the Company’s stock option activity during the year ended December 31, 2015:
| Number of Shares | Weighted Average Exercise Price | Weighted Average Contractual Term (in years) | Aggregate Intrinsic Value | |||||||||||||
| Outstanding as of December 31, 2014 | 2,847,097 | $ | 5.89 | |||||||||||||
| Granted | 755,950 | $ | 7.50 | |||||||||||||
| Exercised | (44,548 | ) | $ | 4.53 | ||||||||||||
| Cancelled | (239,958 | ) | $ | 6.40 | ||||||||||||
| Outstanding as of December 31, 2015 | 3,318,541 | $ | 6.24 | 8.03 | $ | — | ||||||||||
| Exercisable as of December 31, 2015 | 1,453,656 | $ | 5.60 | 7.23 | $ | — | ||||||||||
| Vested and expected to vest as of December 31, 2015 | 3,182,369 | $ | 6.21 | 8.00 | $ | — | ||||||||||
The aggregate intrinsic value of stock options in the table above represents the difference between the $2.31 closing price of the Company’s common stock as of December 31, 2015 and the exercise price of outstanding, exercisable, and vested and expected to vest in-the-money stock options.
Included in amounts in the table above, the Company granted performance-based options to four executives to purchase a total of 145,200 shares of the Company’s common stock at an exercise price of $6.09 per share in July 2014. These options vest based on the successful completion of various performance requirements of each of the four executives at various times through December 31, 2018.
The following table summarizes information about the Company’s stock options as of December 31, 2015:
| Exercise Price or Range of Exercise Price | Options Outstanding | Weighted Average Contractual Life (Years) | Options Exercisable | |||||||||||
| $3.88 | to | $4.27 | 545,572 | 5.88 | 509,183 | |||||||||
| $5.35 | to | $6.68 | 1,999,512 | 8.21 | 813,210 | |||||||||
| $7.05 | to | $7.98 | 602,858 | 9.17 | 96,093 | |||||||||
| $8.25 | to | $11.09 | 169,328 | 8.88 | 33,899 | |||||||||
| $35.30 | to | $36.66 | 1,271 | 5.70 | 1,271 | |||||||||
| 3,318,541 | 8.03 | 1,453,656 | ||||||||||||
Stock options with a fair value of $0.8 million, $2.3 million and $3.4 million completed vesting in the years ended December 31, 2013, 2014 and 2015, respectively. As of December 31, 2015, the Company had a total of $8,719,350 in unrecognized compensation expense from unvested stock option awards, of which $3,679,987 is expected to be recognized in 2016, $3,198,816 in 2017, $1,469,680 in 2018, and $370,867 in 2019.
11. License Agreement
In July 2011, the Company entered into an agreement with Celldex Therapeutics, Inc. (“Celldex”), pursuant to which Celldex granted the Company a nonexclusive license to specified patents and patent applications regarding actions necessary or helpful for processing dendritic cells. Upon the execution of the agreement, the Company paid Celldex $50,000 of a $100,000 up front license fee. The Company paid the balance of this fee on January 31, 2012. Under this agreement, the Company is required to pay:
| • | a $75,000 annual license fee; |
| • | a specified milestone payment based on the achievement of a specified regulatory milestone; and |
| • | a specified dollar amount per dose of AGS-003 the Company sells. |
| F-24 |
The agreement will terminate on a country-by-country basis upon the expiration of the last to expire of the patent rights licensed under the agreement in a country. The latest date of expiration of the licensed Celldex patents is April 2016.
12. Collaboration Agreements
Pharmstandard License Agreement
In August 2013, Pharmstandard International S.A. ("Pharmstandard"), purchased shares of the Company’s series E preferred stock. Concurrently with such purchase, the Company entered into an exclusive royalty-bearing license agreement with Pharmstandard. Under this license agreement, the Company granted Pharmstandard and its affiliates a license, with the right to sublicense, develop, manufacture and commercialize AGS-003 and other products for the treatment of human diseases, which are developed by Pharmstandard using the Company’s individualized immunotherapy platform, in the Russian Federation, Armenia, Azerbaijan, Belarus, Georgia, Kazakhstan, Kyrgyzstan, Moldova, Tajikistan, Turkmenistan, Ukraine and Uzbekistan, which the Company refers to as the Pharmstandard Territory. The Company also provided Pharmstandard with a right of first negotiation for development and commercialization rights in the Pharmstandard Territory to specified additional products the Company may develop.
Under the terms of the license agreement, Pharmstandard licensed the Company rights to clinical data generated by Pharmstandard under the agreement and granted the Company an option to obtain an exclusive license outside of the Pharmstandard Territory to develop and commercialize improvements to the Company’s Arcelis technology generated by Pharmstandard under the agreement, a non-exclusive worldwide royalty-free license to Pharmstandard improvements to manufacture products using the Company’s Arcelis technology and a license to specified follow-on licensed products generated by Pharmstandard outside of the Pharmstandard Territory, each on terms to be negotiated upon the Company’s request for a license. In addition, Pharmstandard agreed to pay the Company pass-through royalties on net sales of all licensed products in the low single digits until it has generated a specified amount of aggregate net sales. Once the net sales threshold is achieved, Pharmstandard will pay the Company royalties on net sales of specified licensed products, including AGS-003, in the low double digits below 20%. These royalty obligations last until the later of the expiration of specified licensed patent rights in a country or the twelfth anniversary of the first commercial sale in such country on a country by country basis and no further royalties on specified other licensed products. After the net sales threshold is achieved, Pharmstandard has the right to offset a portion of the royalties Pharmstandard pays to third parties for licenses to necessary third party intellectual property against the royalties that Pharmstandard pays to the Company.
The agreement will terminate upon expiration of the royalty term, upon which all licenses will become fully paid-up perpetual exclusive licenses. Either party may terminate the agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy and the Company may terminate the agreement if Pharmstandard challenges or assists a third party in challenging specified patent rights of ours. If Pharmstandard terminates the agreement upon the Company’s material breach or bankruptcy, Pharmstandard is entitled to terminate the Company’s licenses to improvements generated by Pharmstandard, upon which the Company may come to rely for the development and commercialization of AGS-003 and other licensed products outside of the Pharmstandard Territory, and to retain its licenses from the Company and to pay the Company substantially reduced royalty payments following such termination.
In November 2013, the Company entered into an agreement with Pharmstandard under which Pharmstandard purchased additional shares of the Company’s series E preferred stock. Under this agreement, the Company agreed to enter into a manufacturing rights agreement for the European market with Pharmstandard and that the manufacturing rights agreement would provide for the issuance of warrants to Pharmstandard to purchase 499,788 shares of the Company’s common stock at an exercise price of $5.82 per share. The Company has not entered into this manufacturing rights agreement or issued the warrants. All outstanding shares of the Company’s preferred stock converted into shares of the Company’s common stock upon the closing of its initial public offering in February 2014.
Green Cross License Agreement
In July 2013, the Company entered into an exclusive royalty-bearing license agreement with Green Cross Corp. ("Green Cross"). Under this agreement, the Company granted Green Cross a license to develop, manufacture and commercialize AGS-003 for mRCC in South Korea. The Company also provided Green Cross with a right of first negotiation for development and commercialization rights in South Korea to specified additional products the Company may develop.
| F-25 |
Under the terms of the license, Green Cross has agreed to pay the Company $500,000 upon the initial submission of an application for regulatory approval of a licensed product in South Korea, $500,000 upon the initial regulatory approval of a licensed product in South Korea and royalties ranging from the mid-single digits to low double digits below 20% on net sales until the fifteenth anniversary of the first commercial sale in South Korea. In addition, Green Cross has granted the Company an exclusive royalty free license to develop and commercialize all Green Cross improvements to the Company’s licensed intellectual property in the rest of the world, excluding South Korea, except that, as to such improvements for which Green Cross makes a significant financial investment and that generate significant commercial benefit in the rest of the world, the Company is required to negotiate in good faith a reasonable royalty that the Company will be obligated to pay to Green Cross for such license. Under the terms of the agreement, the Company is required to continue to develop and to use commercially reasonable efforts to obtain regulatory approval for AGS-003 in the United States.
The agreement will terminate upon expiration of the royalty term, which is 15 years from the first commercial sale, upon which all licenses will become fully paid up perpetual non-exclusive licenses. Either party may terminate the agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy and the Company may terminate the agreement if Green Cross challenges or assists a third party in challenging specified patent rights of ours. If Green Cross terminates the agreement upon the Company’s material breach or bankruptcy, Green Cross is entitled to terminate the Company’s licenses to improvements and retain its licenses from the Company and to pay the Company substantially reduced milestone and royalty payments following such termination.
Medinet License Agreement
In December 2013, the Company entered into a license agreement with Medinet Co., Ltd. This agreement was subsequently novated, amended and restated between the Company, Medinet Co., Ltd. and MEDcell Co., Ltd. in October 2014. Pursuant to the novation, Medinet Co., Ltd. assigned and transferred all of its rights and obligations under the original license agreement, including the rights to receive payments under the $9.0 million Medinet note, to MEDcell Co., Ltd. without any substantive change in the underlying rights or obligations. Medinet Co., Ltd. and MEDcell Co., Ltd. together are referred to herein as “Medinet.” Under this agreement, the Company granted Medinet an exclusive, royalty-free license to manufacture in Japan AGS-003 and other products using the Company’s Arcelis technology solely for the purpose of the development and commercialization of AGS-003 and these other products for the treatment of mRCC. The Company refers to this license as the manufacturing license. In addition, under this agreement, the Company granted Medinet an option to acquire a nonexclusive, royalty-bearing license under the Company’s Arcelis technology to sell in Japan AGS-003 and other products for the treatment of mRCC. The Company refers to the option as the sale option and the license as the sale license.
Under the manufacturing license, if Medinet does not exercise the sale option, Medinet may only manufacture AGS-003 and these other products for the Company or its designee. If Medinet does not exercise the sale option, the Company and Medinet have agreed to negotiate in good faith a supply agreement under which Medinet would supply the Company or its designee with AGS-003 and these other products for development and sale for the treatment of mRCC in Japan. If Medinet exercises the sale option, it may only manufacture AGS-003 and these other products for itself, its related parties and its sublicensees. During the term of the manufacturing license, the Company may not manufacture AGS-003 or these other products for the Company or any designee for development or sale for the treatment of mRCC in Japan. The Medinet option expires on March 31, 2016.
In consideration for the manufacturing license, Medinet paid the Company $1.0 million. Medinet also loaned the Company $9.0 million in connection with the Company entering into the agreement. The Company has agreed to use these funds in the development and manufacturing of AGS-003 and the other products. Medinet also agreed to pay the Company milestone payments of up to a total of $9.0 million upon the achievement of developmental and regulatory milestones and $5.0 million upon the achievement of a sales milestone related to AGS-003 and these products. The first milestone with a $1.0 million payment was achieved in July 2015 (see Notes 5 and 10). If Medinet exercises the sale option, it will pay the Company $1.0 million, as well as royalties on net sales at a rate to be negotiated until the later of the expiration of the licensed patent rights in Japan and the twelfth anniversary of the first commercial sale in Japan. If the Company and Medinet cannot agree on the royalty rate, the Company and Medinet have agreed to submit the matter to arbitration.
In December 2013, in connection with the manufacturing license agreement with Medinet, the Company borrowed $9.0 million pursuant to an unsecured promissory note that bears interest at a rate of 3.0% per annum. The principal and interest under the note are due and payable on December 31, 2018. Under the terms of the note and the manufacturing license agreement, any milestone payments related to the developmental and regulatory milestones that become due will be applied first to the repayment of the loan. The Company has the right to prepay the loan at any time. If the Company has not repaid the loan by December 31, 2018, then the Company has agreed to grant to Medinet a non-exclusive, royalty-bearing license to make and sell Arcelis products in Japan for the treatment of cancer. In such event, the amounts owing under the loan as of December 18, 2018 may constitute pre-paid royalties under the license or would be due and payable. Royalties under this license would be paid until the expiration of the licensed patent rights in Japan at a rate to be negotiated. If the Company and Medinet cannot agree on the royalty rate, the Company and Medinet have agreed to submit the matter to arbitration.
| F-26 |
The Company recorded the $1.0 million payment from Medinet as a deferred liability. In addition, because the $9.0 million promissory note was issued at a below market interest rate, the Company allocated the proceeds of the loan between the manufacturing license agreement and the debt at the time of issuance. Accordingly, as of December 31, 2013, the date of borrowing, the Company recorded $6.9 million to notes payable, based upon an effective interest rate of 8.0%, and $2.1 million as a deferred liability. During the year ended December 31, 2015, the Company recognized a $1.0 million milestone payment as deferred revenue under this license agreement and reduced the related note payable by $0.8 million and the deferred liability by $0.2 million. As of December 31, 2015, the amount of the note payable was $7.5 million, including $1.3 million accrued interest. As of December 31, 2015, the total deferred liability associated with the Medinet note was $3.8 million.
The agreement will terminate upon expiration of the royalty term, upon which all licenses will become fully paid up, perpetual non-exclusive licenses. Either party may terminate the agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy, and the Company may terminate the agreement if Medinet challenges or assists a third party in challenging specified patent rights of the Company. If Medinet terminates the agreement upon the Company’s material breach or bankruptcy, Medinet is entitled to terminate the Company’s licenses to improvements and retain its royalty-bearing licenses from the Company.
Lummy License Agreement
On April 7, 2015, the Company and Lummy HK, a wholly owned subsidiary of Chongqing Lummy Pharmaceutical Co. Ltd., entered into a license agreement (the “License Agreement”) whereby the Company granted to Lummy HK an exclusive license under the Arcelis technology, including patents, know-how and improvements to manufacture, develop and commercialize products for the treatment of cancer (“Licensed Product”) in China, Hong Kong, Taiwan and Macau (the “Territory”). Lummy HK also has a right of first negotiation with respect to a license under the Arcelis technology for the treatment of infectious diseases in the Territory.
Under the terms of the License Agreement, the parties will share relevant data, and the Company will have a right to reference Lummy HK data for purposes of its development programs under the Arcelis technology. In addition, Lummy HK has granted to the Company an exclusive, royalty-free license under and to any and all Lummy HK improvements to the Arcelis technology conceived or reduced to practice by Lummy HK (“Lummy HK Improvements”) and Lummy HK data to develop and/or commercialize products (“Arcelis-Based Products”) outside the Territory, an exclusive, royalty-free license under and to any and all investigational new drug applications ("INDs") and other regulatory approvals and Lummy HK trademarks used for an Arcelis-Based Product to develop and/or commercialize an Arcelis-Based Product outside the Territory and a non-exclusive, worldwide, royalty-free license under any Lummy HK Improvements and Lummy HK data to manufacture Arcelis-Based Products anywhere in the world. Lummy HK has the right to reference Company’s data, INDs and other regulatory filings and submissions for the purpose of developing and obtaining regulatory approval of Licensed Products in the Territory.
Pursuant to the License Agreement, Lummy HK will pay the Company royalties on net sales and up to an aggregate of $20.0 million upon the achievement of manufacturing, regulatory and commercial milestones. The License Agreement will terminate upon expiration of the last to expire royalty term for all Arcelis-Based Products, with each royalty term being the longer of the expiration of the last valid patent claim covering the applicable Arcelis-Based Product and 10 years from the first commercial sale of such Arcelis-Based Product. Either party may terminate the License Agreement for the other party’s uncured material breach or if specified conditions occur relating to the other party’s insolvency or bankruptcy. The Company may terminate the License Agreement if Lummy HK challenges or assists a third party in challenging specified patent rights of the Company. If Lummy HK terminates the License Agreement upon the Company’s material breach or bankruptcy, Lummy HK is entitled to terminate the licenses it granted to the Company and retain its licenses from the Company with respect to Arcelis-Based Products then in development or being commercialized, subject to Lummy HK’s continued obligation to pay royalties and milestones with respect to such Arcelis-Based Products.
See Note 8 for a description of issuances by the Company of the Company’s common stock in connection with the License Agreement.
| F-27 |
13. Commitments
The Company rents laboratory and office space and equipment under operating leases that expire in various years through 2017. Future minimum lease payments under noncancelable operating leases as of December 31, 2015 are as follows:
| 2016 | $ | 435,359 | ||
| 2017 | 79,689 | |||
| Total minimum lease payments | $ | 515,048 |
Rent expense related to operating leases for the years ended December 31, 2013, 2014 and 2015 was $348,191, $447,918 and $536,932, respectively.
The Company has entered into various licensing agreements with universities and other research institutions under which the Company receives substantially all rights of the inventors or co-assignee to produce and market technology protected by certain patents and patent applications. The Company also entered into various assignment agreements with a scientist under which the Company receives exclusive rights to produce and market technology protected by certain patents and patent applications.
The Company is generally required to make royalty payments ranging from 1% to 4% of future sales of products employing the technology or falling under claims of a patent. If future sales require the use of technology licensed from multiple different sources, the total royalty rates could be higher. As royalty payments are directly related to future sales volume, future commitments cannot be determined. No accrual for future payments under these agreements has been recorded, as the Company cannot estimate if, when or in what amount payments may become due.
14. Employee Benefit Plan
The Company provides a retirement plan qualified under section 401(k) of the Internal Revenue Code of 1986, as amended (“IRC”). Participants may elect to contribute a portion of their annual compensation to the plan, after complying with certain limitations set by the IRC. All employees are eligible to participate in the plan after attaining the age of 21. The Company matched 25% of the first 6% contributed by eligible participants in the plan during the years ended December 31, 2013, 2014 and 2015, or $101,700, $121,399 and $334,487, respectively.
15. Net Loss Per Share
The following table presents the computation of basic and diluted net loss per share of common stock:
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| Net loss | $ | (23,921,563 | ) | $ | (53,305,938 | ) | $ | (74,788,521 | ) | |||
| Accretion of redeemable convertible preferred stock | (884,647 | ) | (863,226 | ) | — | |||||||
| Reverse prior accretion on redeemable preferred stock due to reduction in liquidation value of Series A, B, and C | 5,657,638 | — | — | |||||||||
| Preferred stock dividend due to exchanges of preferred shares | (14,726,088 | ) | — | — | ||||||||
| Net loss attributable to common stockholders | $ | (33,874,660 | ) | $ | (54,169,164 | ) | $ | (74,788,521 | ) | |||
| Weighted average common shares outstanding, basic and diluted | 229,865 | 17,367,665 | 20,457,245 | |||||||||
| Net loss per share attributable to common stockholders, basic and diluted | $ | (147.37 | ) | $ | (3.12 | ) | $ | (3.66 | ) | |||
| F-28 |
The following potentially dilutive securities have been excluded from the computation of diluted weighted average shares outstanding, as they would be antidilutive:
| Year Ended December 31, | ||||||||||||
| 2013 | 2014 | 2015 | ||||||||||
| Redeemable convertible preferred stock | 6,322,747 | — | — | |||||||||
| Stock options outstanding | 767,510 | 2,847,097 | 3,149,010 | |||||||||
| Warrants outstanding | 424,961 | 82,781 | 82,781 | |||||||||
16. Selected Quarterly Data (unaudited)
| Quarter Ended | ||||||||||||||||
| March 31, 2015 | June 30, 2015 | September 30, 2015 | December 31, 2015 | |||||||||||||
| Revenue | $ | 178,771 | $ | 107,391 | $ | 158,349 | $ | 73,818 | ||||||||
| Operating expenses: | ||||||||||||||||
| Research and development | 14,766,982 | 16,084,834 | 17,227,405 | 13,975,602 | ||||||||||||
| General and administrative | 2,371,201 | 2,909,358 | 2,689,398 | 3,041,054 | ||||||||||||
| Operating loss | (16,959,412 | ) | (18,886,801 | ) | (19,758,454 | ) | (16,942,838 | ) | ||||||||
| Other income (expense), net | (586,803 | ) | (718,017 | ) | (320,114 | ) | (616,082 | ) | ||||||||
| Net loss attributable to common stockholders | $ | (17,546,215 | ) | $ | (19,604,818 | ) | $ | (20,078,568 | ) | $ | (17,558,920 | ) | ||||
| Net loss attributable to common stockholders per share, basic and diluted | $ | (0.89 | ) | $ | (0.95 | ) | $ | (0.97 | ) | $ | (0.84 | ) | ||||
| Weighted average shares outstanding, basic and diluted | 19,674,245 | 20,622,326 | 20,704,163 | 20,823,456 | ||||||||||||
| Quarter Ended | ||||||||||||||||
| March 31, 2014 | June 30, 2014 | September 30, 2014 | December 31, 2014 | |||||||||||||
| Revenue | $ | 798,788 | $ | 473,163 | $ | 398,615 | $ | 303,453 | ||||||||
| Operating expenses: | ||||||||||||||||
| Research and development | 8,472,195 | 10,569,134 | 12,998,409 | 13,459,178 | ||||||||||||
| General and administrative | 1,933,476 | 1,865,822 | 2,320,036 | 2,480,025 | ||||||||||||
| Operating loss | (9,606,883 | ) | (11,961,793 | ) | (14,919,830 | ) | (15,635,750 | ) | ||||||||
| Other income (expense), net | (394,097 | ) | (21,738 | ) | (181,187 | ) | (584,660 | ) | ||||||||
| Net loss | (10,000,980 | ) | (11,983,531 | ) | (15,101,017 | ) | (16,220,410 | ) | ||||||||
| Accretion of redeemable convertible preferred stock | (863,226 | ) | — | — | — | |||||||||||
| Net loss attributable to common stockholders | $ | (10,864,206 | ) | $ | (11,983,531 | ) | $ | (15,101,017 | ) | $ | (16,220,410 | ) | ||||
| Net loss attributable to common stockholders per share, basic and diluted | $ | (1.05 | ) | $ | (0.61 | ) | $ | (0.77 | ) | $ | (0.83 | ) | ||||
| Weighted average shares outstanding, basic and diluted | 10,376,561 | 19,655,187 | 19,655,561 | 19,656,209 | ||||||||||||
| F-29 |
17. Subsequent Events
PIPE Financing
On March 4, 2016, the Company entered into a securities purchase agreement (the “Securities Purchase Agreement”) with certain investors (the “Investors”), pursuant to which the Company will issue and sell an aggregate of up to $60 million of the Company’s common stock, par value $0.001 per share (the “Shares”) and warrants to purchase shares of common stock (the “Warrants”) in a private placement (the “Financing”). Investors participating in the Financing include Pharmstandard, Forargos B.V., Tianyi Lummy International Holdings Group Ltd. (“Tianyi Lummy”), China BioPharma Capital I, L.P. (“China BioPharma”), TVM V Life Science Ventures GmbH & Co. KG and Wasatch Funds Trust. Alexey Vinogradov, Andrei Petrov, Hubert Birner and Sander van Deventer (collectively, the “Investor Directors”), who are members of the Company’s board of directors, are affiliated with certain of the Investors.
The Financing will take place in up to three tranches. Under the Securities Purchase Agreement, at the initial closing, which occurred on March 14, 2016, the Company sold and the Investors purchased for a total purchase price of $19,882,915 a total of 3,652,430 Shares and Warrants to purchase a total of 2,739,323 shares of common stock (0.75 shares of common stock for each Share purchased), based on a purchase price per Share and accompanying Warrant equal to $5.44375 (the “Purchase Price”). At the second closing, which must occur no later than September 30, 2016, the Company has agreed to sell and the Investors have agreed to purchase for an additional purchase price of $29,824,520 a total of 5,478,672 Shares and Warrants to purchase a total of 4,109,005 shares of common stock (0.75 shares of common stock for each Share purchased), based on a purchase price per Share and accompanying Warrant equal to the Purchase Price. The second closing is subject to a recommendation by the Independent Data Monitoring Committee (the “IDMC”) for the Company’s ongoing pivotal Phase 3 clinical trial of AGS-003 (the “ADAPT Study”) at or following the IDMC’s next regular meeting following the initial closing (currently scheduled for June 2016) (the “First IDMC Meeting”) that the Company (a) continue the ADAPT Study or (b) discontinue the ADAPT Study based on favorable efficacy data. The Warrants will have an exercise price of $5.35 per share, expire on the fifth anniversary of the date of issuance and have other terms described below under “Warrants.”
Under the Securities Purchase Agreement, Pharmstandard has agreed that, at the Company’s option following the satisfaction of certain conditions, but no later than February 28, 2017, it shall purchase at the third closing up to $10,292,563 of Shares (without Warrants) at a price per Share equal to the lesser of (i) $5.35 and (ii) the fair market value of the Company’s common stock at the time the Company exercises its option (but not lower than $1.50 per Share). If the price per Share is less than $5.35, the Company may elect to proceed with the third closing only if it has received stockholder approval to proceed with the third closing or stockholder approval is not otherwise required under Nasdaq rules. Pharmstandard’s obligation to purchase shares at the third closing is subject to the IDMC having held its next regular meeting after the First IDMC Meeting (currently anticipated to be held in November or December 2016) (the “Second IDMC Meeting”) and having made a recommendation at or following the Second IDMC Meeting for the ADAPT Study that the Company (a) continue the ADAPT Study or (b) discontinue the ADAPT Study based on favorable efficacy data, and the Company’s cash position at such time. The dollar amount of Shares to be purchased by Pharmstandard at the third closing is subject to reduction on a dollar-for-dollar basis for certain cash amounts raised by the Company after the initial closing through equity or debt financings or collaborations.
Under the Securities Purchase Agreement, Pharmstandard has also agreed that:
| • | if the Company determines to seek stockholder approval in connection with the sale and issuance of the Shares to be sold in the third closing, Pharmstandard will vote, and cause its affiliates to vote, all shares of the Company’s voting securities held by Pharmstandard or its affiliates in favor of such proposal, subject to certain limitations; and |
| • | to the extent that and for so long as the total voting power of voting securities of the Company beneficially owned by Pharmstandard and its affiliates and any other Persons whose beneficial ownership of the Company’s common stock would be aggregated with Pharmstandard for purposes of Section 13(d) of the Securities Exchange Act of 1934, as amended, exceeds 33% of the voting power of the total number of voting securities of the Company then outstanding, in any election of directors and in any other matter submitted to a general vote of the stockholders of the Company (whether taken at an annual or special meeting of stockholders or by written action) Pharmstandard shall and shall cause its affiliates and such other persons whose beneficial ownership of the Company’s common stock would be aggregated with Pharmstandard for purposes of Section 13(d) of the Securities Exchange Act of 1934, as amended, to vote such number of voting securities of the Company which exceed 33% of the voting power of the total number of voting securities of the Company then outstanding in the same manner as and in the same proportion to the votes cast on the matter by the other holders of voting securities of the Company. |
| F-30 |
Under the Securities Purchase Agreement, the Company has agreed it will use the net proceeds from the Financing in accordance with a budget approved by the Company’s board of directors, as it may be modified from time to time by the Company’s board of directors, including a majority of the Investor Directors.
The Company has agreed to pay at each closing the legal expenses of the Investors and the legal expenses of the Investors incurred in connection with the resale registration obligations of the Company set forth in the Registration Rights Agreement; provided, however, that the Company shall have no obligation to pay more than a total of $140,000 with respect to such expenses.
The second and third closings will be subject to the satisfaction of certain customary closing conditions. The Company has granted the Investors, and the Investors have granted the Company, indemnification rights with respect to its or their representations, warranties, covenants and agreements under the Securities Purchase Agreement.
Warrants
The Warrants issued at the first closing and to be issued at the second closing, are and will be exercisable for shares of the Company’s common stock at an exercise price of $5.35 per share on or prior to the fifth anniversary of the date of issuance, will be immediately exercisable, and will be exercisable for cash or by cashless exercise in certain limited circumstances. The number of shares that may be exercised by certain Investors may be limited to ensure that, following such exercise, the number of shares of the Company’s common stock beneficially owned by such Investor does not exceed 4.999% of the number of shares of the Company’s common stock then issued and outstanding, which limitation is subject to modification by such Investors in accordance with requirements specified in the Warrants.
Registration Rights Agreement
On March 4, 2016, in connection with entering into the Securities Purchase Agreement, the Company entered into a registration rights agreement (the “Registration Rights Agreement”) with the Investors, pursuant to which the Company has agreed to register for resale the Shares and the shares of the Company’s common stock issued upon exercise of the Warrants (the “Warrant Shares,” and together with the Shares, the “Registrable Securities”). Under the Registration Rights Agreement, the Company has agreed to use its best efforts to file a registration statement after each closing covering the Shares and the Warrant Shares underlying the Warrants sold at such closing, as applicable, and to use its best efforts to keep such registration statement effective until the date the Shares and Warrant Shares covered by such registration statement have been sold or may be sold pursuant to Rule 144 without restriction.
In the event a registration statement has not been filed by within 60 days of the initial closing and within 30 days of each of the second and third closings (in each case subject to extension under certain circumstances), then the Company has agreed to make pro rata payments to each Investor that participated in such closing as liquidated damages in an amount equal to 1% of the aggregate amount invested by each such Investor in such closing per 30-day period or pro rata for any portion thereof during which no such registration statement is filed with respect to the applicable Registrable Securities. Moreover, in the event (i) a registration statement covering Registrable Securities issued at a certain closing is not declared effective by the Securities and Exchange Commission (the “SEC”) by the 120th day following the initial closing and by the 90th day following each of the second and third closings, or (ii) after a registration statement has been declared effective by the SEC, such registration statement is not available to cover any sales of Registrable Securities registered by such registration statement, then the Company has agreed to make pro rata payments to each Investor that participated in such closing as liquidated damages in an amount equal to 1% of the aggregate amount invested by each such Investor in such closing per 30-day period or pro rata for any portion thereof following the date by which such Registration Statement should have been effective, subject to specified exceptions.
The Company has granted the Investors, and the Investors have granted to the Company, customary indemnification rights in connection with the registration statements.
| F-31 |
Schedule II—Valuation and Qualifying Accounts
Deferred Tax Asset Valuation Allowance
Information presented below is in thousands:
| Additions | ||||||||||||||||||||
| Balance at Beginning of Year | Charged to Expenses (a) | Charged to Other Accounts | Increases | Balance at End of Year | ||||||||||||||||
| Year Ended December 31, 2015 | $ | 68,197 | $ | 16,706 | $ | — | $ | — | $ | 84,903 | ||||||||||
| Year Ended December 31, 2014 | $ | 47,945 | $ | 20,252 | $ | — | $ | — | $ | 68,197 | ||||||||||
| Year Ended December 31, 2013 | $ | 39,134 | $ | 8,811 | $ | — | $ | — | $ | 47,945 | ||||||||||
| (a) | – Impact of providing full valuation allowance against all deferred tax assets since the Company could not assert that it was more likely than not that these deferred tax assets would be realized. |
F-32
