Attached files
| file | filename |
|---|---|
| EX-31.1 - EXHIBIT 31.1 - TRIPLE-S MANAGEMENT CORP | ex31_1.htm |
| EX-32.1 - EXHIBIT 32.1 - TRIPLE-S MANAGEMENT CORP | ex32_1.htm |
| EX-32.2 - EXHIBIT 32.2 - TRIPLE-S MANAGEMENT CORP | ex32_2.htm |
| EX-31.2 - EXHIBIT 31.2 - TRIPLE-S MANAGEMENT CORP | ex31_2.htm |
| 10-K/A - TRIPLE-S MANAGEMENT CORP 10-K/A 12-31-2014 - TRIPLE-S MANAGEMENT CORP | form10ka.htm |
Exhibit 10.1
CONTRACT BETWEEN
ADMINISTRACIÓN DE SEGUROS DE SALUD DE PUERTO RICO (ASES)
and
TRIPLE-S SALUD, INC.
for
PROVISION OF PHYSICAL & BEHAVIORAL HEALTH SERVICES UNDER THE GOVERNMENT HEALTH PLAN PROGRAM
Contract No.: 2015-000087
Service Regions: Metro North &West
Page 1 of 301
TABLE OF CONTENTS
|
ARTICLE 1
|
GENERAL PROVISIONS
|
7
|
|
ARTICLE 2
|
DEFINITIONS
|
11
|
|
ARTICLE 3
|
ACRONYMS
|
29
|
|
ARTICLE 4
|
ASES RESPONSIBILITIES
|
32
|
|
ARTICLE 5
|
ELIGIBILITY AND ENROLLMENT
|
35
|
|
ARTICLE 6
|
ENROLLEE SERVICES
|
49
|
|
ARTICLE 7
|
COVERED SERVICES AND BENEFITS
|
70
|
|
ARTICLE 8
|
INTEGRATION OF PHYSICAL AND BEHAVIORAL HEALTH SERVICES
|
117
|
|
ARTICLE 9
|
PROVIDER NETWORK
|
121
|
|
ARTICLE 10
|
PROVIDER CONTRACTING
|
145
|
|
ARTICLE 11
|
UTILIZATION MANAGEMENT
|
162
|
|
ARTICLE 12
|
QUALITY IMPROVEMENT AND PERFORMANCE PROGRAM
|
168
|
|
ARTICLE 13
|
FRAUD, WASTE, AND ABUSE
|
180
|
|
ARTICLE 14
|
GRIEVANCE SYSTEM
|
188
|
|
ARTICLE 15
|
ADMINISTRATION AND MANAGEMENT
|
200
|
|
ARTICLE 16
|
PROVIDER PAYMENT MANAGEMENT
|
203
|
|
ARTICLE 17
|
INFORMATION MANAGEMENT AND SYSTEMS
|
209
|
|
ARTICLE 18
|
REPORTING
|
221
|
|
ARTICLE 19
|
ENFORCEMENT – INTERMEDIATE SANCTIONS
|
236
|
|
ARTICLE 20
|
ENFORCEMENT - LIQUIDATED DAMAGES AND OTHER REMEDIES
|
243
|
|
ARTICLE 21
|
CONTRACT TERM
|
251
|
|
ARTICLE 22
|
PAYMENT FOR SERVICES
|
252
|
|
ARTICLE 23
|
FINANCIAL MANAGEMENT
|
258
|
Page 2 of 301
|
ARTICLE 24
|
PAYMENT OF TAXES
|
270
|
|
ARTICLE 25
|
RELATIONSHIP OF PARTIES
|
271
|
|
ARTICLE 26
|
INSPECTION OF WORK
|
271
|
|
ARTICLE 27
|
GOVERNMENT PROPERTY
|
271
|
|
ARTICLE 28
|
OWNERSHIP AND USE OF DATA AND SOFTWARE
|
272
|
|
ARTICLE 29
|
CRIMINAL BACKGROUND CHECKS
|
273
|
|
ARTICLE 30
|
SUBCONTRACTS
|
274
|
|
ARTICLE 31
|
REQUIREMENT OF INSURANCE LICENSE
|
277
|
|
ARTICLE 32
|
CERTIFICATIONS
|
277
|
|
ARTICLE 33
|
RECORDS REQUIREMENTS
|
278
|
|
ARTICLE 34
|
CONFIDENTIALITY
|
280
|
|
ARTICLE 35
|
TERMINATION OF CONTRACT
|
286
|
|
ARTICLE 36
|
PHASE-OUT AND COOPERATION WITH OTHER CONTRACTORS
|
293
|
|
ARTICLE 37
|
INSURANCE
|
294
|
|
ARTICLE 38
|
COMPLIANCE WITH ALL LAWS
|
295
|
|
ARTICLE 39
|
CONFLICT OF INTEREST AND CONTRACTOR INDEPENDENCE
|
296
|
|
ARTICLE 40
|
CHOICE OF LAW OR VENUE
|
297
|
|
ARTICLE 41
|
ATTORNEY’S FEES
|
298
|
|
ARTICLE 42
|
SURVIVABILITY
|
298
|
|
ARTICLE 43
|
PROHIBITED AFFILIATIONS WITH INDIVIDUALS DEBARRED AND SUSPENDED
|
298
|
|
ARTICLE 44
|
WAIVER
|
298
|
|
ARTICLE 45
|
FORCE MAJEURE
|
298
|
|
ARTICLE 46
|
BINDING
|
299
|
|
ARTICLE 47
|
TIME IS OF THE ESSENCE
|
299
|
Page 3 of 301
|
ARTICLE 48
|
AUTHORITY
|
299
|
|
ARTICLE 49
|
ETHICS IN PUBLIC CONTRACTING
|
299
|
|
ARTICLE 50
|
CONTRACT LANGUAGE INTERPRETATION
|
299
|
|
ARTICLE 51
|
ARTICLE AND SECTION TITLES NOT CONTROLLING
|
299
|
|
ARTICLE 52
|
LIMITATION OF LIABILITY/EXCEPTIONS
|
299
|
|
ARTICLE 53
|
COOPERATION WITH AUDITS
|
300
|
|
ARTICLE 54
|
OWNERSHIP AND FINANCIAL DISCLOSURE
|
300
|
|
ARTICLE 55
|
AMENDMENT IN WRITING
|
302
|
|
ARTICLE 56
|
CONTRACT ASSIGNMENT
|
303
|
|
ARTICLE 57
|
SEVERABILITY
|
303
|
|
ARTICLE 58
|
ENTIRE AGREEMENT
|
303
|
|
ARTICLE 59
|
INDEMNIFICATION
|
303
|
|
ARTICLE 60
|
NOTICES
|
304
|
|
ARTICLE 61
|
OFFICE OF THE COMPTROLLER
|
305
|
| ATTACHMENT 1: | DESIGNATED LAWS |
| ATTACHMENT 2: | MAP OF PUERTO RICO SERVICE REGIONS |
| ATTACHMENT 3: | GHP UNIVERSAL ENROLLEE GUIDELINES HANDBOOK |
| ATTACHMENT 4: | CPTET CENTERS AND COMMUNITY-BASED ORGANIZATIONS FOR HIV/AIDS |
| ATTACHMENT 5: | MASTER FORMULARY |
| ATTACHMENT 6: | RETAIL PHARMACY REIMBURSEMENT LEVELS |
| ATTACHMENT 7: | UNIFORM GUIDE FOR SPECIAL COVERAGE |
| ATTACHMENT 8: | COST-SHARING |
| ATTACHMENT 9: | INFORMATION SYSTEMS |
| ATTACHMENT 10: | GUIDELINES FOR CO-LOCATION OF BEHAVIORAL HEALTH PROVIDERS IN PMG SETTINGS |
Page 4 of 301
| ATTACHMENT 11: | PER MEMBER PER MONTH PAYMENTS |
| ATTACHMENT 12: | INITIAL DELIVERABLE DUE DATES |
| ATTACHMENT 13: | ASES NORMATIVE LETTERS, SPECIAL NEEDS CHILDREN CODES |
| ATTACHMENT 14: | PROGRAM INTEGRITY PLAN DEVELOPMENT GUIDELINES |
| ATTACHMENT 15: | FORMULARY A-102: EVIDENCE OF LACK OF PROVIDERS AND PROVIDERS REFUSAL TO CONTRACT |
| ATTACHMENT 16: | LIST OF REQUIRED REPORTS |
| ATTACHMENT 17: | EHR ADOPTION PLAN |
| ATTACHMENT 18: | BUSINESS ASSOCIATE AGREEMENT |
| ATTACHMENT 19: | QUALITY IMPROVEMENT PROCEDURE MANUAL |
| ATTACHMENT 20: | PPN DIAGRAM |
| ATTACHMENT 21: | GUIDELINES FOR REVERSE CO-LOCATION OF PRIMARY CARE PHYSICIANS IN MENTAL HEALTH SETTINGS |
| ATTACHMENT 22: | STERILIZATION CONSENT FORM |
| ATTACHMENT 23: | POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF MEDICAID OVERPAYMENTS TO PROVIDERS |
Page 5 of 301
THIS CONTRACT, is made and entered into by and between the Puerto Rico Health Insurance Administration (Administración de Seguros de Salud de Puerto Rico, hereinafter referred to as “ASES” or “the Administration”), a public corporation in the Commonwealth of Puerto Rico (“the Commonwealth” or “Puerto Rico”), with employer identification number 66-0500678 and Triple-S Salud, Inc. (“the Contractor”), an insurance company duly organized and authorized to do business under the laws of the Commonwealth, with employer identification number 66-0555677. The Effective Date of the Contract is October 31, 2014 and the Implementation Date of the Contract is April1, 2015.
WHEREAS, pursuant to Title XIX of the Federal Social Security Act, codified as 42 USC 1396 et seq. (“the Social Security Act”), and Act No. 72 of September 7, 1993 of the Laws of the Commonwealth of Puerto Rico (“Act 72”), a comprehensive program of medical assistance for needy persons exists in the Commonwealth;
WHEREAS, ASES is responsible for health care policy, purchasing, planning, and regulation pursuant to Act 72, as amended, and other sources of law of the Commonwealth designated in Attachment 1, and pursuant to this statutory provision, ASES has established a managed care program under the medical assistance program, known as “GHP,” “GHP Program,” or “the Government Health Plan”;
WHEREAS, the Puerto Rico Health Department (“the Health Department”) is the single State agency designated to administer medical assistance in the Commonwealth under Title XIX of the Social Security Act of 1935, as amended, and is charged with ensuring the appropriate delivery of health care services under the Medicaid and the Children’s Health Insurance Program (“CHIP”) in the Commonwealth, and ASES manages these programs pursuant to a memorandum of understanding;
WHEREAS, GHP serves a mixed population including not only the Medicaid and CHIP populations, but also other eligible individuals as established in Act 72;
WHEREAS, ASES seeks to comply with Puerto Rico’s public policy objectives of creating GHP, an integrated system of physical and Behavioral Health Services, with an emphasis on preventative services and access to quality care;
WHEREAS, ASES issued a Request for Proposals (“the RFP”) for physical and Behavioral Health Services on June 25 - 27, 2014, which, except as provided in Article 58 below, are expressly incorporated as if completely restated herein;
WHEREAS, ASES has received from the Contractor a proposal in response to the RFP, “Contractor’s Proposal,” which, except as provided in Article 58 below, is expressly incorporated as if completely restated herein; and,
WHEREAS, ASES agrees to review and make appropriate changes to the co-payment requirements in the Contract in compliance with federal requirements and upon CMS approval;
WHEREAS, ASES agrees to maintain a continuous program of cost containment strategies to pursue opportunities for the identification, development of work plans and implementation of activities directed to achieve cost control objectives, in coordination and collaboration with subcontractors, PBM and PBA. The scope of the program includes, but is not limited to, pharmacy, medical and dental service utilization, provider network management and handling of exception processes. Areas of need will be identified through the analysis of ASES database, routine and special subcontractor’s reports, quarterly meetings presentations and other sources.
Page 6 of 301
WHEREAS, ASES accepts the Contractor’s Proposal to provide the services contemplated under this Contract for ASES;
NOW, THEREFORE, FOR AND IN CONSIDERATION of the mutual promises, covenants and agreements contained herein, and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, ASES and the Contractor (each individually a “Party” and collectively the “Parties”) hereby agree as follows:
ARTICLE 1 GENERAL PROVISIONS
| 1.1 | General Provisions |
| 1.1.1 | The Contractor shall assist the Commonwealth by providing and delivering services under the GHP through described tasks, obligations, and responsibilities included in this Contract. |
| 1.1.2 | The Contractor shall maintain the staff, organizational, and administrative capacity and capabilities necessary to carry out all the duties and responsibilities under this Contract. |
| 1.1.3 | The Contractor shall not make any changes to the following without explicit prior written approval from the Executive Director of ASES or his or her designee: |
| 1.1.3.1 | Its business address, telephone number, facsimile number, and e-mail address; |
| 1.1.3.2 | Its corporate status or nature; |
| 1.1.3.3 | Its business location; |
| 1.1.3.4 | Its corporate structure; |
| 1.1.3.5 | Its ownership, including but not limited to the new owner’s legal name, business address, telephone number, facsimile number, and e-mail address; and/or |
| 1.1.3.6 | Its incorporation status. |
| 1.1.4 | The Contractor shall notify ASES within five (5) Business Days of a change in the following: |
Page 7 of 301
| 1.1.4.1 | Its solvency (as a result of a non-operational event); |
| 1.1.4.2 | Its corporate officers or executive employees; or |
| 1.1.4.3 | Its Federal employee identification number or Federal tax identification number. |
| 1.1.5 | Unless otherwise specified herein, all documentation, including policies and procedures that the Contractor is required to maintain, shall be given prior written approval from ASES. All documentation, including the Deliverables listed in Attachment 12, must be submitted to ASES in English. |
| 1.1.6 | Unless otherwise specified, the Contractor shall notify ASES and/or the Puerto Rico Medicaid Program of any applicable provisions Immediately. |
| 1.2 | Background |
| 1.2.1 | From October 1, 2010 through March 30, 2015, the government health program previously referred to as La Reforma was known as MI Salud. Beginning April 1, 2015, the program will be referred to as the Government Health Plan or GHP Program. |
| 1.2.2 | The Government Health Plan (“GHP”) has the following objectives: |
| 1.2.2.1 | To transform Puerto Rico’s health system through an integrated vision of physical and Behavioral Health. |
| 1.2.2.2 | To encourage the Contractor and other selected GHP MCO(s) to work together to provide integrated physical and Behavioral Health Services in each of nine (9) Service Regions of the Commonwealth. |
| 1.2.2.3 | To establish Primary Medical Groups (“PMGs”), which shall enter into agreements with the Contractor and shall act as the gatekeepers for medical care. PMGs shall provide, manage, and direct health services, including coordination with Behavioral Health personnel and specialist services, in a timely manner. |
| 1.2.2.4 | To develop, within each of the nine (9) Service Regions, a Preferred Provider Network (“PPN”), which shall be composed of physician specialists, clinical laboratories, radiology facilities, hospitals, and Ancillary Service Providers that shall render Covered Services to persons enrolled in the GHP (“Enrollees”). |
| 1.2.2.5 | To facilitate access to quality Primary Care and specialty services within the PPN by providing all services without the requirement of a Referral, and not requiring cost-sharing for services within the PPN. |
Page 8 of 301
| 1.2.2.6 | To ensure that, other than through appropriate Utilization control measures, services to Enrollees in the GHP are not refused, restricted, or reduced, including by reason of pre-existing conditions or waiting periods. |
| 1.2.2.7 | To support the Health Department and the Puerto Rico Mental Health and Against Addiction Services Administration (Administración de Servicios de Salud Mental y Contra la Adicción, hereinafter “ASSMCA”) in health education efforts focusing on lifestyle changes, HIV/AIDS prevention, the prevention of drug and substance abuse, and maternal and child health. |
| 1.3 | Groups Eligible for Services Under the GHP |
| 1.3.1 | The Contractor will be responsible for providing services to all persons determined eligible for the GHP and enrolled in the Contractor’s MCO(s). The groups to be served under the GHP shall hereinafter be referred to collectively as “Eligible Persons.” The groups are subject to change and currently include: |
| 1.3.1.1 | Medicaid and CHIP. All Medicaid and CHIP eligibility categories covered in the Puerto Rico Medicaid and CHIP State Plans are eligible to enroll in the GHP and shall be referred to hereinafter as “Medicaid and CHIP Eligibles.” |
| 1.3.1.2 | Other Groups (Non-Medicaid and CHIP Eligibles). The following groups, which receive services under the GHP without any Federal participation, will be referred to hereinafter as “Other Eligible Persons.” |
| 1.3.1.2.1 | The “Commonwealth Population,” comprised of the following groups: |
| 1.3.1.2.1.1 | Certain persons who are between twenty-two (22) and sixty-four (64) years of age, inclusive of the age limits, and who do not qualify for either Medicaid or CHIP; |
| 1.3.1.2.1.2 | Police officers of the Commonwealth and their Dependents; |
| 1.3.1.2.1.3 | Surviving spouses of deceased police officers; |
Page 9 of 301
| 1.3.1.2.1.4 | Survivors of domestic violence referred by the Office of the Women’s Advocate; |
| 1.3.1.2.1.5 | Veterans; and |
| 1.3.1.2.1.6 | Any other group of Eligible Persons that may be added during the Contract Term as a result of a change in laws or regulations. |
| 1.3.1.2.2 | Commonwealth employees and pensioners, whose eligibility for the GHP is not based on income. |
| 1.4 | Service Regions |
| 1.4.1 | For the delivery of services under the GHP, ASES has divided the Commonwealth into nine (9) regions: eight (8) geographical Service Regions and one (1) “Virtual Region.” See Attachment 2 for a map of the geographical Service Regions. The Contractor shall perform services under this Contract in the Metro North, and West Regions. |
| 1.5 | Delegation of Authority |
| 1.5.1 | Federal law and Puerto Rico law limit the capacity of ASES to delegate decisions to the Contractor. All decisions relating to public policy and to the administration of the Medicaid, CHIP, and the Puerto Rico government health assistance program included in the GHP rest with the Puerto Rico Medicaid Program and ASES. |
| 1.6 | Availability of Funds |
| 1.6.1 | This Contract is subject to the availability of funds on the part of ASES, which in turn is subject to the transfer of Federal, Puerto Rico, and municipal funds to ASES. If available funds are insufficient to meet its contractual obligations, ASES reserves the right to terminate this Contract, pursuant to Section 35.5. |
| 1.7 | Cooperation, Assistance and Compliance with Special Projects |
| 1.7.1 | The Contractor shall provide to ASES and any other agency of the Commonwealth all necessary cooperation, assistance, and compliance with requirements in the development and implementation of any special project of ASES and any other agency of the Commonwealth or the Federal Government. The Contractor acknowledges that this is a sine qua non of this Contract and that it will comply with ASES change requests related to such projects as these are implemented due to Commonwealth or Federal mandate. |
Page 10 of 301
ARTICLE 2 DEFINITIONS
Whenever capitalized in this Contract, the following terms have the respective meaning set forth below, unless the context clearly requires otherwise.
Act 72: The law of the Commonwealth, adopted on September 7, 1993, as subsequently amended, which created ASES and empowered ASES to administer certain government health programs.
Act 408: The Puerto Rico Mental Health Code (Act No. 408 of October 2, 2000, as amended), which established the public policy and procedures regarding the delivery of Behavioral Health services in Puerto Rico.
Abandoned Call: A call initiated to a Call Center that is ended by the caller before any conversation occurs or before a caller is permitted access to a caller-selected option.
Abuse: Provider practices that are inconsistent with sound fiscal, business, or medical practices, and that result in unnecessary costs to the GHP Program, or in reimbursement for services that are not Medically Necessary or that fail to meet professionally recognized standards for the provision of health care. It also includes Enrollee practices that result in unnecessary costs to the GHP.
Access: Adequate availability of Benefits to fulfill the needs of Enrollees.
Action: The denial or limited authorization of a requested service, including the type or level of service; the reduction, suspension, or termination of a previously authorized service; the denial, in whole or part, of payment for a service (including in circumstances in which an Enrollee is forced to pay for a service; the failure to provide services in a timely manner (within the timeframes established by this Contract or otherwise established by ASES); or the failure of the Contractor to act within the timeframes provided in 42 CFR 438.408(b). For a resident of a rural area, the denial of an Enrollee's request to exercise his or her right, under 42 CFR 438.52(b)(2)(ii), to obtain services outside the network.
Actuarial Report: Actuarial reports the Contractor is required to submit in accordance with Article 18 of this Contract.
Administrative Functions: The contractual obligations of the Contractor under this Contract, other than providing Covered Services; include, without limitation, Care Management, Disease Management, Utilization Management, Credentialing Providers, Network management, Quality Improvement, Marketing, Enrollment, Enrollee services, Claims payment, Information Systems, financial management, and reporting.
Administrative Law Hearing: The Appeal process administered by the Commonwealth and as required by Federal law, available to Enrollees after they exhaust the Contractor’s Grievance System and Complaint Process.
Administrative Referral: A Referral of an Enrollee by the Contractor to a Provider or facility located outside the PPN, when the Enrollee’s PCP or other PMG physician does not provide a Referral within the required time period.
Page 11 of 301
Adult: An individual age nineteen (19) or older unless otherwise specified.
Advance Directive: A written instruction, such as a living will or durable power of attorney, granting responsibility over an individual’s health care, as defined in 42 CFR 489.100, and as recognized under Puerto Rico law under Act 160 of November 17, 2001, as amended, relating to the provision of health care when the individual is incapacitated.
ADFAN: Families and Children Administration (Administración de Familias y Niños), which is responsible for foster care children in the custody of the Commonwealth.
Affiliate: Any person, firm, corporation (including, without limitation, service corporation and processional corporation), partnership (including, without limitation, general partnership, limited partnership and limited liability partnership), limited liability company, joint venture, business trust, association or other entity or organization that now or in the future directly or indirectly controls, is controlled by, or is under common control with the Contractor.
Agent: An entity that contracts with ASES to perform Administrative Functions, including but not limited to: fiscal Agent activities; Outreach, eligibility, and Enrollment activities; and systems and technical support.
Ambulatory Services Units: Ambulatory clinics that mainly provide health services to children, families, and adults, which are staffed by an interdisciplinary team responsible for the appropriate treatment and referral processes.
Ancillary Services: Professional services, including laboratory, radiology, physical therapy, and respiratory therapy, which are provided in conjunction with other medical or hospital care.
Appeal: An Enrollee request for a review of an Action. It is a formal petition by an Enrollee, an Enrollee’s Authorized Representative, or the Enrollee’s Provider, acting on behalf of the Enrollee with the Enrollee’s written consent, to reconsider a decision in the case that the Enrollee or Provider does not agree with an Action taken.
ASES: Administración de Seguros de Salud de Puerto Rico (the Puerto Rico Health Insurance Administration), the entity in the Commonwealth responsible for oversight and administration of the GHP Program, or its Agent.
ASES Data: All Data created from Information, documents, messages (verbal or electronic), reports, or meetings involving, arising out of or otherwise in connection with this Contract.
ASES Information: All proprietary Data and/ or Information generated from any Data requested, received, created, provided, managed and stored by Contractors, -in hard copy, digital image, or electronic format - from ASES and/or Enrollees (as defined in Article 2) necessary or arising out of this Contract, except for the Contractor’s Proprietary Information.
ASSMCA: Administración de Servicios de Salud Mental y Contra la Adicción (the Puerto Rico Mental Health and Against Addiction Services Administration), the government agency responsible for the planning and establishment of mental health and substance abuse policies and procedures and for the coordination, development, and monitoring of all Behavioral Health Services rendered to Enrollees in the GHP.
Page 12 of 301
At Risk: When a Provider agrees to accept responsibility to provide, or arrange for, any service in exchange for the Per Member Per Month Payment (PMPM).
Authorized Certifier: The Contractor’s CEO, CFO, or an individual with delegated authority to sign for and who reports directly to the CEO and/or CFO.
Authorized Representative: A person given written authorization by an Enrollee to make health-related decisions on behalf of an Enrollee, including, but not limited to: Enrollment and Disenrollment decisions, filing Complaints, Grievances, and Appeals, and the choice of a PCP or PMG.
Auto-Assignment: The assignment of an Enrollee to a PMG and a PCP by the Contractor, normally at the time that ASES or the Contractor auto-enrolls the person in the GHP Program.
Auto-Enrollment: The Enrollment of a Potential Enrollee in a GHP Plan by the Contractor without any action by the Potential Enrollee, as provided in Article 5 of this Contract.
Basic Coverage: The physical and Behavioral Health Services available to all GHP Enrollees (as distinguished from Special Coverage, which is available only to Enrollees with certain diagnoses after a registration process). The GHP Covered Services are listed in Article 7 of this Contract.
Behavioral Health: The umbrella term for mental health (including psychiatric illnesses and emotional disorders) and substance use (involving addictive and chemical dependency disorders). The term also refers to preventing and treating co-occurring mental health and substance use disorders (“SUDs”).
Behavioral Health Facility: A facility for the delivery of inpatient or stabilization Behavioral Health Services, which houses at least two (2) Providers. These facilities include:
| (i) | Psychiatric hospitals (or a unit within a general hospital) |
| (ii) | Emergency or stabilization units |
| (iii) | Partial hospitalization units |
| (iv) | Intensive ambulatory services units |
| (v) | Ambulatory services units |
| (vi) | Residential units |
| (vii) | Addiction service units (detoxification, ambulatory, inpatient, and residential) |
Benefits: The services set forth in this Contract, for which the Contractor has agreed to provide, arrange, and be held fiscally responsible, including Basic Coverage, dental services, Special Coverage, and Administrative Functions.
Blocked Call: A call that cannot be connected Immediately because no circuit is available at the time the call arrives or because the telephone system is programmed to block calls from entering the queue when the queue is backed up beyond a defined threshold.
Page 13 of 301
Breach: The unauthorized acquisition, access, use, or disclosure of Personal Health Information which compromises the security or privacy of such Information.
Business Continuity and Disaster Recovery (“BC-DR”) Plan: A documented plan (process) to restore vital and critical Information/health care technology systems in the event of business interruption due to human, technical, or natural causes. The plan focuses mainly on technology systems, encompassing critical hardware, operating and application software, and tertiary elements required to support the operating environment. It must support the process requirement to restore vital business Data inside the defined business requirement, including an emergency mode operation plan as necessary. The BC-DR also provides for continuity of health care in the event of plan terminations.
Business Days: Traditional workdays, including Monday, Tuesday, Wednesday, Thursday, and Friday. Puerto Rico Holidays are excluded.
Calendar Days: All seven days of the week.
Call Center: A telephone service facility equipped to handle a large number of inbound and outbound calls. This facility must meet all requirements set forth in Section 6.8 of this Contract.
Capitation: A contractual agreement through which a Contractor or Provider agrees to provide specified health care services to Enrollees for a fixed amount per month.
Care Management: An Administrative Function comprised of a set of Enrollee-centered steps to ensure that an Enrollee with intensive needs, including catastrophic or high-risk conditions, receives the necessary services in a supportive, effective, efficient, timely, and cost-effective manner.
Care Manager: A professional with at least a Bachelor of Arts, a Bachelor of Science, or a Bachelor of Science in Nursing degree in health or Behavioral Health-related fields who is devoted to helping Enrollees access the services they need for their recuperation and for the implementation of their individual treatment plans.
Centers for Medicare & Medicaid Services (“CMS”): The agency within the US Department of Health and Human Services with responsibility for the Medicare, Medicaid, and the Children’s Health Insurance Programs (“CHIP”).
Center for the Collection of Municipal Revenues (“CRIM”): The tax collection agency of the Commonwealth.
Certification: As provided in Section 5.1.2 of this Contract, a decision by the Puerto Rico Medicaid Program that a person is eligible for services under the GHP Program because the person is Medicaid Eligible, CHIP Eligible, or a member of the Commonwealth Population. Some public employees and pensioners may enroll in GHP without first receiving a Certification.
Children’s Health Insurance Program (“CHIP”): The Commonwealth’s Children’s Health Insurance Program established pursuant to Title XXI of the Social Security Act.
Page 14 of 301
CHIP Eligible: A child eligible to enroll in the GHP Program because he or she is eligible for CHIP.
Chronic Condition: An ongoing physical, behavioral, or cognitive disorder, with a duration of at least twelve (12) months with resulting functional limitations, reliance on compensatory mechanisms (medications, special diet, assistive devices, etc.) and service use or need beyond that which is normally considered routine.
Claim: Whether submitted manually or electronically, a bill for services, a line item of services, or a bill detailing all services for one (1) Enrollee.
Clean Claim: A Claim received by the Contractor for adjudication, which can be processed without obtaining additional information from the Provider of the service or from a Third Party. It includes a Claim with errors originating in the Contractor’s Claims system. It does not include a Claim from a Provider who is under investigation for Fraud, Waste, or Abuse, or a Claim under review to determine Medical Necessity.
Cold-Call Marketing: Any unsolicited personal contact by the Contractor with a Potential Enrollee, for the purposes of Marketing.
Co-Location: An integrated care model in which Behavioral Health Services are provided in the same site as primary care.
Commonwealth Population: A group eligible for participation in the GHP as Other Eligible Persons, with no Federal participation supporting the cost of their coverage, which is comprised of low-income persons and other groups listed in Section 1.3.1.2.1.
Complaint: An expression of dissatisfaction about any matter other than an Action that is resolved at the point of contact rather than through filing a formal Grievance.
Contract: The written agreement between ASES and the Contractor; comprised of the Contract, any addenda, appendices, attachments, or amendments thereto.
Contract Term: The duration of time that this Contract is in effect, as defined in Article 21 of this Contract.
Contractor: The Managed Care Organization that is a Party of this Contract, licensed as an insurer by the Puerto Rico Commissioner of Insurance (“PRICO”), which contracts hereunder with ASES for the provision of Covered Services and Benefits to Enrollees in a designated Service Region on the basis of PMPM Payments, under the GHP program.
Co-Payment: A cost-sharing requirement which is a fixed monetary amount paid by the Enrollee to a Provider for certain Covered Services as specified by ASES.
Page 15 of 301
Corrective Action Plan: The detailed written plan required by ASES from the Contractor to correct or resolve a deficiency or event causing the assessment of a liquidated damage or sanction against the Contractor.
Cost Avoidance: A method of paying Claims in which the Provider is not reimbursed until the Provider has demonstrated that all available health insurance, and other sources of Third Party Liability, have been exhausted.
Countersignature: An authorization provided by the Enrollee’s PCP, or another Provider within the Enrollee’s PMG, for a prescription written by another Provider to be dispensed. No Countersignature shall be required if the Provider writing the prescription is within the PPN.
Covered Services: Those Medically Necessary health care services (listed in Article 7 of this Contract) provided to Enrollees by Providers, the payment or indemnification of which is covered under this Contract.
Credentialing: The Contractor’s determination as to the qualification of a specific Provider to render specific health care services.
Credible Allegation of Fraud: Any allegation of Fraud that has been verified by another State, the Commonwealth, or ASES, or otherwise has been preliminary investigated by the Contractor, as the case may be, and that has indicia of reliability that comes from any source.
Cultural Competency: A set of interpersonal skills that allow individuals to increase their understanding, appreciation, acceptance, and respect for cultural differences and similarities within, among, and between groups and the sensitivity to know how these differences influence relationships with Enrollees. This requires a willingness and ability to draw on community-based values, traditions and customs, to devise strategies to better meet culturally diverse Enrollee needs, and to work with knowledgeable persons of and from the community in developing focused interactions, communications, and other supports.
Daily Basis: Each Business Day.
Data: A series of meaningful electrical signals that may be manipulated or assigned; Data Set: demographic, health, or other Informational elements suitable for specific use.
Deductible: In the context of Medicare, the dollar amount of Covered Services that must be incurred before Medicare will pay for all or part of the remaining Covered Services.
Deliverable: A document, manual, or report submitted to ASES by the Contractor to exhibit that the Contractor has fulfilled the requirements of this Contract.
Dependent: A person who is enrolled in the GHP as the spouse or child of the principal Enrollee.
Disease Management: An Administrative Function comprised of a set of Enrollee-centered steps to provide coordinated care to Enrollees suffering from diseases listed in Section 7.8.3 of this Contract.
Disenrollment: The termination of an individual’s Enrollment in the Contractor’s Plan.
Page 16 of 301
Dual Eligible Beneficiary: An Enrollee or Potential Enrollee eligible for both Medicaid and Medicare.
Durable Medical Equipment: Equipment, including assistive technology, which: (i) can withstand repeated use; (ii) is used to service a health-related or functional purpose; (iii) is ordered by a Health Care Provider to address an illness, injury, or disability; and (iv) is appropriate for use in the home, work place, or school.
Early and Periodic Screening, Diagnostic, and Treatment (“EPSDT”) Program: A Medicaid-mandated program that covers screening and diagnostic services to determine physical and mental deficiencies in Enrollees less than twenty-one (21) years of age, and health care, prevention, treatment, and other measures to correct or ameliorate any deficiencies and Chronic Conditions discovered.
Effective Date of Contract: The day the Contract is executed by both Parties.
Effective Date of Disenrollment: The date, as defined in Section 5.3.3 of this Contract, on which an Enrollee ceases to be covered under the Contractor’s Plan.
Effective Date of Eligibility: The eligibility period specified for each population covered under the GHP as described in Section 5.1.3 of the Contract.
Effective Date of Enrollment shall have the meaning prescribed to it in Section 5.2.2. of the Contract.
Electronic Funds Transfer (“EFT”): Transfer of funds between accounts using electronic means such as a telephone or computer rather than paper-based payment methods such as cash or checks.
Electronic Health Record (“EHR") System: An electronic record of health-related information on an individual that is created, gathered, managed, and consulted upon by authorized health care clinicians and staff and certified by The Office of the National Coordinator’s Authorized Testing and Certification Bodies (“ONC-ATCBs”).
Eligible Person: A person eligible to enroll in the GHP Program, as provided in Section 1.3.1 of this Contract, by virtue of being Medicaid Eligible, CHIP Eligible, or an Other Eligible Person.
Emergency Medical Condition: A medical or Behavioral Health condition, regardless of diagnosis or symptoms, manifesting itself in acute symptoms of sufficient severity (including severe pain) that a prudent layperson, who possesses an average knowledge of health and medicine, could reasonably expect the absence of immediate medical attention to result in placing the health of the individual (or, with respect to a pregnant woman, the health of the woman or her unborn child) in serious jeopardy, serious impairments of bodily functions, serious dysfunction of any bodily organ or part, serious harm to self or other due to an alcohol or drug abuse emergency, serious injury to self or bodily harm to others, or the lack of adequate time for a pregnant women having contractions to safely reach a another hospital before delivery. The Contractor may not impose limits on what constitutes an Emergency Medical Condition.
Page 17 of 301
Emergency Services: Physical or Behavioral Health Covered Services (as described in Section 7.5.9) furnished by a qualified Provider in an emergency room that are needed to evaluate or stabilize an Emergency Medical Condition or a Psychiatric Emergency that is found to exist using the prudent layperson standard.
Encounter: A distinct set of services provided to an Enrollee in a face-to-face setting on the dates that the services were delivered, regardless of whether the Provider is paid on a Fee-for-Service or Capitated basis. Encounters with more than one (1) Provider, and multiple Encounters with the same Provider, that take place on the same day in the same location will constitute a single Encounter, except when the Enrollee, after the first Encounter, suffers an illness or injury requiring an additional diagnosis or treatment.
Encounter Data: (i) All Data captured during the course of a single Encounter that specify the diagnoses, comorbidities, procedures (therapeutic, rehabilitative, maintenance, or palliative), pharmaceuticals, medical devices, and equipment associated with the Enrollee receiving services during the Encounter; (ii) The identification of the Enrollee receiving and the Provider(s) delivering the health care services during the single Encounter; and (iii) A unique ( i.e. unduplicated) identifier for the single Encounter.
Enrollee: A person who is currently enrolled in the Contractor’s Plan, as provided in this Contract, and who, by virtue of relevant Federal and Puerto Rico laws and regulations, is an Eligible Person listed in Section 1.3.1 of this Contract.
Enrollment: The process by which an Eligible Person becomes an Enrollee of the Contractor’s Plan.
Excess Profit: The excess over 2.5 percent of the profit before income taxes as reported in the audited financial statements. Excess Profits are to be shared between the Contractor or the Subcontractors and ASES, as provided in Sections 22.1.18 and 22.1.19.
Experience of Care and Health Outcomes (“ECHO”) Survey: A survey constructed to merge the most desirable aspects of the Mental Health Statistics Program’s Consumer Survey (“MHSIP”) and the Consumer Assessment of Behavioral Health Services (“CABHS”) Instrument in order to capture as many unique aspects of mental health and substance abuse-related services while limiting redundancy. The survey is a product of nearly six (6) years of research and testing by CAHPS grantees at the Harvard Medical School, with extensive input from behavioral health care experts.
External Quality Review Organization (“EQRO”): An organization that meets the competence and independence requirements set forth in 42 CFR 438.354 and performs analyses and evaluations on the quality, timeliness, and Access to Covered Services and Benefits that the Contractor furnishes to Enrollees.
Federally Qualified Health Center (“FQHC”): An entity that provides outpatient health programs pursuant to Section 1905(l)(2)(B) of the Social Security Act.
Fee-for-Service: A method of reimbursement based on payment for specific Covered Services on a service-by-service basis rendered to an Enrollee.
Page 18 of 301
Fraud: An intentional deception or misrepresentation made by a person with the knowledge that the deception could result in some unauthorized benefit or financial gain to him/herself or some other person. It includes any act that constitutes Fraud under applicable Federal or Puerto Rico law.
General Network: The entire group of Providers under contract with the Contractor, including those that are and those that are not members of the Contractor’s Preferred Provider Network.
GHP Plan: A Managed Care Organization under contract with ASES that offers services under the Government Health Plan (“GHP”) Program.
GHP Service Line: The Enrollee support Call Center that the Contractor shall operate as described in Section 6.8 of this Contract, containing two components: the Information Service and the Medical Advice Service.
The Government Health Plan (or “the GHP”): The government health services program (formerly referred to as “La Reforma” or “MI Salud”) offered by the Commonwealth of Puerto Rico, and administered by ASES, which serves a mixed population of Medicaid Eligible, CHIP Eligible, and Other Eligible Persons, and emphasizes integrated delivery of physical and Behavioral Health Services.
Grievance: An expression of dissatisfaction about any matter other than an Action.
Grievance System: The overall system that includes Complaints, Grievances, and Appeals at the Contractor level, as well as Access to the Administrative Law Hearing process.
Health Care Acquired Conditions: A medical condition for which an individual was diagnosed that could be identified by a secondary diagnostic code described in Section 1886(d)(4)(D)(iv) of the Social Security Act.
Health Care Provider: An individual engaged in the delivery of health care services as licensed or certified by Puerto Rico in which he or she is providing services, including but not limited to physicians, podiatrists, optometrists, chiropractors, psychologists, psychiatrists, licensed Behavioral Health practitioners, dentists, physician’s assistants, physical or occupational therapists and therapists assistants, speech-language pathologists, audiologists, registered or licensed practical nurses (including nurse practitioners, clinical nurse specialist, certified registered nurse anesthetists, and certified nurse midwives), licensed certified social workers, registered respiratory therapists, and certified respiratory therapy technicians.
Health Certificate: Certificate issued by a physician after an examination that includes Venereal Disease Research Laboratory (“VRDL”) and tuberculosis (“TB”) tests if the individual suffers from a contagious disease that could incapacitate him or her or prevent him or her from doing his or her job, and does not represent a danger to public health.
Health Information Exchange (“HIE”): The secure and effective electronic transmission (push–pull) of the Personal Health Information of patients between Providers, across organizations within a region, community or hospital system, within a jurisdiction and/or between jurisdictions. HIE is also an entity that provides services to enable the electronic sharing of health Information.
Page 19 of 301
Health Information Organization (“HIO”): “An organization that oversees and governs services related to the exchange of health-related Information among organizations according to nationally recognized standards,” as defined in The National Alliance for Health Information Technology Report to the Office of the National Coordinator for Health Information Technology.
Health Information Technology for Economic and Clinical Health (“HITECH”) Act: Public Law 111-5 (2009). When referenced in this Contract, it includes all related rules, regulations, and procedures.
Healthy Child Care: The battery of screenings (listed in Section 7.5.3.1) provided to children under age two (2) who are Medicaid- or CHIP Eligible as part of Puerto Rico’s (“EPSDT”) Program.
Health Care Effectiveness Data and Information Set (“HEDIS”): A set of standardized performance measures developed by the National Committee for Quality Assurance (“NCQA”) to measure and compare MCO performance.
Health Insurance Portability and Accountability Act (“HIPAA”): A law enacted in 1996 by the US Congress. When referenced in this Contract, it includes all related rules, regulations, and procedures.
Immediately: Within twenty-four (24) hours, unless otherwise provided in this Contract.
Implementation Date of the Contract: The date on which the Contractor shall commence providing Covered Services and other Benefits under this Contract after it has passed a readiness review; the expected implementation date of this Contract is April 1, 2015
Incident: The attempted or successful unauthorized access, use, disclosure, modification, or destruction of Information or interference with system operations in an Information System.
Incurred-But-Not-Reported (“IBNR”): Estimate of unpaid Claims liability, including received but unpaid Claims.
Indian: An individual, defined in Title 25 of the U.S.C. sections 1603(c), 1603(f), 1603(f) or who has been determined eligible, as an Indian, pursuant to 42 C.F.R. 136.12 or Title V of the Indian Health Care Improvement Act, to receive health care services from Indian Health Care Providers (Indian Health Services, an Indian Tribe, Tribal Organization, or Urban Indian Organization-I/T/U) or through Referral under Contract Health Services.
Information: Data to which meaning is assigned, according to context and assumed conventions; meaningful fractal Data for decision support purposes.
Information Service: The component of the GHP Service Line, a Call Center operated by the Contractor (described in Section 6.8), intended to assist Enrollees with routine inquiries, which shall be fully staffed between the hours of 7:00 a.m. and 7:00 p.m. (Atlantic Time), Monday through Friday, excluding Puerto Rico holidays.
Page 20 of 301
Information System(s): A combination of computing and communications hardware and software that is used in: (i) the capture, storage, manipulation, movement, control, display, interchange and/or transmission of Information, i.e. structured Data (which may include digitized audio and video) and documents; and/or (ii) the processing of such Information for the purposes of enabling and/or facilitating a business process or a related transaction.
Integration Plan: The service delivery plan under the GHP Program, providing physical and Behavioral Health Services in close coordination, to ensure optimum detection, prevention, and treatment of physical and Behavioral Health conditions.
International Statistical Classification of Diseases and Related Health Problems Tenth Revision (“ICD-10”): A medical classification list created by the World Health Organization that notes various Medical Records including those used for coding diseases, signs, symptoms, abnormal findings, complaints, social circumstances, and external causes of injury or disease.
List of Excluded Individuals and Entities (“LEIE”): A database of individuals and entities excluded from Federally-funded health care programs maintained by the Department of Health and Human Services Office of the Inspector General.
MA-10: Form issued by the Puerto Rico Medicaid Program, entitled “Notice of Action Taken on Application and/or Recertification,” containing the Certification decision (whether a person was determined eligible or ineligible for Medicaid, CHIP, or the Commonwealth Population).
Managed Care Organization (“MCO”): An entity that is organized for the purpose of providing health care and is licensed as an insurer by the Puerto Rico Commissioner of Insurance (“PRICO”), which contracts with ASES for the provision of Covered Services and Benefits in designated Service Regions on the basis of PMPM Payments, under the GHP program.
Marketing: Any communication from the Contractor to any Eligible Person or Potential Enrollee that can reasonably be interpreted as intended to influence the individual to enroll in the Contractor’s Plan, or not to enroll in another plan, or to disenroll from another plan.
Marketing Materials: Materials that are produced in any medium, by or on behalf of the Contractor, that can reasonably be interpreted as intended to market to Potential Enrollees.
Medicaid: The joint Federal/state program of medical assistance established by Title XIX of the Social Security Act.
Medicaid Eligible: An individual eligible to receive services under Medicaid, who is eligible, on this basis, to enroll in the GHP Program.
Medicaid Management Information System (“MMIS”): Computerized system used for the processing, collecting, analyzing, and reporting of Information needed to support Medicaid and CHIP functions. The MMIS consists of all required subsystems as specified in the State Medicaid Manual.
Page 21 of 301
Medical Advice Service: The twenty-four (24) hour emergency medical advice toll-free phone line operated by the Contractor through its GHP Service Line service, described in Section 6.8 of this Contract.
Medical Record: The complete, comprehensive record of an Enrollee including, but not limited to, x-rays, laboratory tests, results, examinations and notes, accessible at the site of the Enrollee’s PCP, or Network Provider, that documents all health care services received by the Enrollee, including inpatient care, outpatient care, Ancillary, and Emergency Services, prepared in accordance with all applicable Federal and Puerto Rico rules and regulations, and signed by the Provider rendering the services.
Medically Necessary Services: Those services that meet the definition found in Section 7.2 of this Contract.
Medicare: The Federal program of medical assistance for persons over age sixty-five (65) and certain disabled persons under Title XVIII of the Social Security Act.
Medicare Part A: The part of the Medicare program that covers inpatient hospital stays, skilled nursing facilities, home health, and hospice care.
Medicare Part B: The part of the Medicare program that covers physician, outpatient, home health, and Preventive Services.
Medicare Part C: The part of the Medicare program that permits Medicare recipients to select coverage among various private insurance plans.
Medicare Platino: A program administered by ASES for Dual Eligible Beneficiaries, in which MCOs or other insurers under contract with ASES function as Part C plans to provide services covered by Medicare, and also to provide a “wrap-around” Benefit of Covered Services and Benefits under the GHP.
National Provider Identifier (“NPI”): The 10-digit unique-identifier numbering system for Providers created by the Centers for Medicare & Medicaid Services (CMS), through the National Plan and Provider Enumeration System.
Negative Determination or Redetermination Decision: The decision by the Puerto Rico Medicaid Program that a person is not initially eligible or no longer eligible for services under the GHP Program (because the person no longer meets the eligibility requirements for Medicaid, CHIP, or Puerto Rico’s government health assistance program).
Network Provider: A Provider that has a contract with the Contractor under the GHP Program. This term includes both Providers in the General Network and Providers in the PPN.
Non-Emergency Medical Transportation (“NEMT”): A ride, or reimbursement for a ride, provided so that an Enrollee with no other transportation resources can receive Covered Services from a Provider. NEMT does not include transportation provided on an emergency basis, such as trips to the emergency room in life threatening situations.
Page 22 of 301
Notice of Action: The written notice described in Section 14.4.3, in which the Contractor notifies both the Enrollee and the Provider of an Action.
Notice of Disposition: The notice in which the Contractor explains in writing the results and the date of resolution of a Complaint, Grievance, or Appeal to the Enrollee and the Provider.
Office of the Patient Advocate: An office of the Commonwealth created by Act 11 of April 11, 2001, which is tasked with protecting the patient rights and protections contained in the Patient’s Bill of Rights Act.
Office of the Women’s Advocate: An office of the Commonwealth which is tasked, among other responsibilities, with protecting victims of domestic violence.
Other Eligible Person: A person eligible to enroll in the GHP Program under Section 1.3.1.2 of this Contract, who is not Medicaid- or CHIP Eligible. This group is comprised of the Commonwealth Population and certain public employees and pensioners.
Outreach: Means, among other things, of educating or informing the Contractor’s Enrollees about GHP, managed care, and health issues.
Out-of-Network Provider: A Provider that does not have a contract with the Contractor under GHP; i.e., the Provider is not in either the General Network or the PPN.
Overpayment: Any funds that a person or entity receives in excess of the Medicaid allowable amount of the Contractor’s allowed amount as negotiated with the Provider. Overpayments shall not include funds that have been subject to a payment suspension or that have been identified as a Third Party Liability as set forth in Section 23.4.
Patient’s Bill of Rights Act: Law 194 of August 25, 2000, a law of the Commonwealth relating to patient rights and protection.
Patient Protection and Affordable Care Act (“PPACA”): Public Law 111-148 (2010) and the Health Care and Education Reconciliation Act of 2010 (Public Law 111-152 (2010), including any and all rules and regulations thereunder.
Payment Hold: The situation when a Provider who owes funds to Puerto Rico, such Provider cannot be paid until the amounts owed to Puerto Rico are repaid or an acceptable repayment plan is in place, as determined by ASES.
Performance Improvement Projects (PIPs): Projects consistent with 42 CFR 438.240.
Per Member Per Month (“PMPM”) Payment: The fixed monthly amount that the Contractor is paid by ASES for each Enrollee to ensure that Benefits under this Contract are provided. This payment is made regardless of whether the Enrollee receives Benefits during the period covered by the payment.
Page 23 of 301
Personal Health Information (“PHI”) Per 42 CFR 160 and 42 CFR 164, individually identifiable health Information that is transmitted by electronic media, maintained in electronic media, or transmitted or maintained in any other form or medium.
Pharmacy Benefit Manager (“PBM”): An entity under contract with ASES under the GHP Program, responsible for the administration of pharmacy Claims processing, formulary management, drug Utilization review, pharmacy network management, and Enrollee Information Services relating to pharmacy services.
Pharmacy Program Administrator (“PPA”): An entity, under contract with ASES, responsible for implementing and offering support to ASES and the contracted PBMs in the negotiation of rebates and development of the Maximum Allowable Cost (“MAC”) List.
Physician Incentive Plan: Any compensation arrangement between a Contractor and a physician or PMG that is intended to advance Utilization Management.
Plan: The Contractor’s Managed Care Plan, offering services to Enrollees under the GHP.
Post-Stabilization Services: Covered Services, relating to an Emergency Medical Condition or Psychiatric Emergency, that are provided after an Enrollee is stabilized, in order to maintain the stabilized condition or to improve or resolve the Enrollee’s condition.
Potential Enrollee: A person who has been Certified by the Puerto Rico Medicaid Program as eligible to enroll in the GHP (whether on the basis of Medicaid eligibility, CHIP eligibility, or eligibility as a member of the Commonwealth Population), but who has not yet enrolled with the Contractor.
Preferential Turns: The policy of requiring Network Providers to give priority in treating Enrollees from the island municipalities of Vieques and Culebra, so that they may be seen by a Provider within a reasonable time after arriving at the Provider’s office. This priority treatment is necessary because of the remote locations of these municipalities, and the greater travel time required for their residents to seek medical attention.
Preferred Drug List (“PDL”): A published subset of pharmaceutical products used for the treatment of physical and Behavioral Health conditions developed by the PPA from the Master Formulary after clinical and financial review.
Preferred Provider Network (“PPN”): A group of Network Providers that (i) GHP Enrollees may access without any requirement of a Referral or Prior Authorization; (ii) provides services to GHP Enrollees without imposing any Co-Payments; and (iii) meets the Network requirements described in Article 9 of this Contract.
Prevalent Non-English Language: A non-English language spoken by a significant number or percentage of Potential Enrollees and current Enrollees in Puerto Rico, as determined by the Commonwealth.
Page 24 of 301
Preventive Services: Health care services provided by a physician or other Provider within the scope of his or her practice under Puerto Rico law to detect or prevent disease, disability, Behavioral Health conditions, or other health conditions; and to promote physical and Behavioral Health and efficiency.
Primary Care: All health care services, including periodic examinations, Preventive Services and counseling, immunizations, diagnosis and treatment of illness or injury, coordination of overall medical care, record maintenance, and initiation of Referrals to specialty Providers described in this Contract and for maintaining continuity of patient care.
Primary Care Physician: A licensed medical doctor (MD) who is a Provider and who, within the scope of practice and in accordance with Puerto Rico Certification and licensure requirements, is responsible for providing all required Primary Care to Enrollees. The PCP is responsible for determining services required by Enrollees, provides continuity of care, and provides Referrals for Enrollees when Medically Necessary. A PCP may be a general practitioner, family physician, internal medicine physician, obstetrician/gynecologist, or pediatrician.
Primary Medical Group (“PMG”): A grouping of associated Primary Care Physicians and other Providers for the delivery of services to GHP Enrollees using a coordinated care model. PMGs may be organized as Provider care organizations, or as another group of Providers who have contractually agreed to offer a coordinated care model to GHP Enrollees under the terms of this Contract.
Prior Authorization: Authorization granted by the Contractor to determine whether the service is Medically Necessary. In some instances, this process is a condition for receiving the Covered Service.
Provider: Any physician, hospital, facility, or other Health Care Provider who is licensed or otherwise authorized to provide physical or Behavioral Health Services in the jurisdiction in which they are furnished.
Provider Contract: Any written contract between the Contractor and a Provider that requires the Provider to perform specific parts of the Contractor’s obligations for the provision of Covered Services under this Contract.
Psychiatric Emergency: A set of symptoms characterized by an alteration in the perception of reality, feelings, emotions, actions, or behavior, requiring immediate therapeutic intervention in order to avoid immediate damage to the patient, other persons, or property. A Psychiatric Emergency shall not be defined on the basis of lists of diagnoses or symptoms.
Puerto Rico Health Department (“the Health Department”): The Single State Agency charged with administration of the Medicaid Program of the Commonwealth, which (through the Puerto Rico Medicaid Program) is responsible for Medicaid and CHIP eligibility determinations.
Puerto Rico Insurance Commissioner’s Office (“PRICO”): The Puerto Rico Commonwealth agency responsible for regulating, monitoring, and licensing insurance business.
Puerto Rico Medicaid Program: The subdivision of the Health Department that conducts eligibility determinations under GHP for Medicaid, CHIP, and the Commonwealth Population.
Page 25 of 301
Quality Assessment and Performance Improvement Program (“QAPI”): A set of programs aimed at increasing the likelihood of desired health outcomes of Enrollees through the provision of health care services that are consistent with current professional knowledge; the QAPI Program includes incentives to comply with HEDIS standards, to provide adequate Preventive Services, and to reduce the unnecessary use of Emergency Services.
Quality Management/Quality Improvement (“QM/QI”): The process of developing and implementing strategies to ensure the delivery of available, accessible, timely, and Medically Necessary Services that meet optimal clinical standards. This includes the identification of key measures of performance, discovery and Data collection processes, identification and remediation of issues, and systems improvement activities.
Recertification: A determination by the Puerto Rico Medicaid Program that a person previously enrolled in the GHP subsequently received a Negative Redetermination Decision, is again eligible for services under the GHP Program.
Redetermination: The periodic Redetermination of eligibility of an individual for Medicaid, CHIP, or the Commonwealth Population, conducted by the Puerto Rico Medicaid Program.
Referral: A request by a PCP, Psychiatrist, Psychologist, or any other type of Provider in the PMG for an Enrollee to be evaluated and/or treated by a different Provider, usually a specialist. Referrals shall be required only for services outside the Contractor’s PPN.
Reinsurance: An agreement whereby the Contractor transfers risk or liability for losses, in whole or in part, sustained under this Contract. A Reinsurance agreement may also exist at the Provider level.
Remedy: ASES’s means to enforce the terms of the Contract through liquidated damages and other sanctions.
Request for Proposals (“RFP”): The Request for Proposals issued by the Commonwealth on June 25 -27, 2014.
Retention Fund: The amount of Withhold by ASES of the monthly PMPM Payments otherwise payable to the Contractor in order to incentivize the Contractor to meet performance targets under the Quality Incentive Program described in Section 12.5.3. This amount shall be equal to the percent of that portion of the total PMPM Payment that is determined to be attributable to the Contractor’s administration of the Quality Incentive Program described in Sections 12.5 and 22.3. Amounts withheld will be reimbursed to the Contractor in whole or in part (as set forth in Sections 12.5 and 22.3) in the event of a determination by ASES that the Contractor has complied with the quality standards and criteria established by Section 12.5.
Reverse Co-location: An integrated care model in which physical health services are available to Enrollees being treated in Behavioral Health settings.
Runoff Period: the period of time as explained in Section 35.8.2.9.1.3.
Page 26 of 301
Rural Health Clinic or Center (“RHC”): A clinic that is located in an area that has a Provider shortage. An RHC provides primary Care and related diagnostic services and may provide optometric, podiatry, chiropractic, and Behavioral Health Services. An RHC employs, contracts, or obtains volunteer services from Providers to provide services.
Service Authorization Request: An Enrollee’s request for the provision of a service.
Service Region: A geographic area comprised of those municipalities where the Contractor is responsible for providing services under the GHP Program. The GHP Program includes nine (9) Service Regions: eight (8) geographical Service Regions and one (1) Virtual Region.
Span of Control: Information Systems and telecommunications capabilities that the Contractor operates or for which it is otherwise legally responsible according to the terms and conditions of this Contract. The Contractor’s Span of Control also includes systems and telecommunications capabilities outsourced by the Contractor.
Special Coverage: A component of Covered Services provided by the Contractor, described in Section 7.7, which are more extensive than the Basic Coverage services, and for which Enrollees are eligible only by “registering.” Registration for Special Coverage is based on intensive medical needs occasioned by serious illness.
Subcontract: Any written contract between the Contractor and a Third Party, including a Provider, to perform a specified part of the Contractor’s obligations under this Contract.
Subcontractor: Any organization or person, including the Contractor’s parent, subsidiary or Affiliate, who provides any function or service for the Contractor specifically related to securing or fulfilling the Contractor’s obligations to the Commonwealth under the terms of this Contract. Subcontractors do not include Providers unless the Provider is responsible for services other than providing Covered Services pursuant to a Provider participation agreement.
Systems Unavailability: As measured within the Contractor’s Information Systems’ Span of Control, when a system user does not get the complete, correct full-screen response to an input command within three (3) minutes after pressing the “Enter” or any other function key.
Telecommunication Device for the Deaf (“TDD”): Special telephone devices with keyboard attachments for use by individuals with hearing impairments who are unable to use conventional phones.
Terminal Condition: A condition caused by injury, illness, or disease, from which, to a reasonable degree of certainty, will lead to the patient’s death in a period of, at most, six (6) months.
Termination Date of the Contract: The dated designated by ASES as the date that services under this Contract shall end, pursuant to Article 35 of this Contract.
Termination Plan: The plan referenced in Article 35.
Page 27 of 301
Third Party: Any person, institution, corporation, insurance company, public, private, or governmental entity who is or may be liable in Contract, tort, or otherwise by law or equity to pay all or part of the medical cost of injury, disease, or disability of an Enrollee.
Third Party Liability (“TPL”): Legal responsibility of any Third Party to pay for health care services.
Utilization: The rate patterns of service usage or types of service occurring within a specified time frame.
Utilization Management (“UM”): A service performed by the Contractor which seeks to ensure that Covered Services provided to Enrollees are in accordance with, and appropriate under, the standards and requirements established by the Contract, or a similar program developed, established, or administered by ASES.
Virtual Region: The Service Region for the GHP Program that is comprised of children who are in the custody of ADFAN, as well as certain survivors of domestic violence referred by the Office of the Women’s Advocate, who enroll in the GHP Program. The Virtual Region encompasses services for these Enrollees throughout Puerto Rico.
Warm Transfer: A telecommunications mechanism in which the person answering the call facilitates the transfer to a Third Party, announces the caller and issue, and remains engaged as necessary to provide assistance.
Waste: Health care spending that can be eliminated without reducing quality of care.
Week: The traditional seven-day week, Sunday through Saturday.
Withhold: A percentage of payments or set dollar amounts that ASES deducts from its payment to the Contractor, or that a Contractor deducts from its payment to a Network Provider, depending on specific predetermined factors.
ARTICLE 3 ACRONYMS
The acronyms included in this Contract stand for the following terms:
|
ACH
|
Automated Clearinghouse
|
|
ACIP
|
Advisory Committee on Immunization Practices
|
|
ADAP
|
AIDS Drug Assistance Program
|
|
ADFAN
|
Puerto Rico Administración de Familias y Niños, or Families and Children Administration
|
|
AHRQ
|
Agency for Health Care Research and Quality
|
|
AICPA
|
American Institute of Certified Public Accountants
|
|
ASES
|
Administración de Seguros de Salud, or Puerto Rico Health Insurance Administration
|
Page 28 of 301
|
ASSMCA
|
The Puerto Rico Mental Health and Against Addiction Services Administration or Administración de Servicios de Salud Mental y Contra la Adicción
|
|
ASUME
|
Minor Children Support Administration
|
|
BC-DR
|
Business Continuity and Disaster Recovery
|
|
CAHPS
|
Consumer Assessment of Health Care Providers and Systems
|
|
CEO
|
Chief Executive Officer
|
|
CFO
|
Chief Financial Officer
|
|
CFR
|
Code of Federal Regulations
|
|
CHIP
|
Children's Health Insurance Program
|
|
CLIA
|
Clinical Laboratory Improvement Amendment
|
|
CMS
|
Centers for Medicare & Medicaid Services
|
|
CPTET
|
Centro de Prevención y Tratamiento de Enfermedades Transmisibles, or Transmissible Diseases Prevention and Treatment Center
|
|
CRIM
|
Center for the Collection of Municipal Revenues
|
|
DM
|
Disease Management
|
|
DME
|
Durable Medical Equipment
|
|
DOJ
|
The Puerto Rico Department of Justice
|
|
ECHO
|
Experience of Care and Health Outcomes Survey
|
|
ECM
|
Electronic Claims Management
|
|
EDI
|
Electronic Data Interchange
|
|
EFT
|
Electronic Funds Transfer
|
|
EIN
|
Employer Identification Number
|
|
EMTALA
|
Emergency Medical Treatment and Labor Act
|
|
EPLS
|
Excluded Parties List System
|
|
EPSDT
|
Early and Periodic Screening, Diagnostic, and Treatment
|
|
EQRO
|
External Quality Review Organization
|
|
ER
|
Emergency Room
|
|
FAR
|
Federal Acquisition Regulation
|
|
FDA
|
Food and Drug Administration
|
|
FFS
|
Fee-for-Service
|
|
FQHC
|
Federally Qualified Health Center
|
|
FTP
|
File Transfer Protocol
|
|
GHP
|
Government Health Plan
|
Page 29 of 301
|
HEDIS
|
The Health Care Effectiveness Data and Information Set
|
|
HHS
|
US Department of Health & Human Services
|
|
HHS-OIG
|
US Department of Health & Human Services Office of the Inspector General
|
|
HIE
|
Health Information Exchange
|
|
HIO
|
Health Information Organization
|
|
HIPAA
|
Health Insurance Portability and Accountability Act of 1996
|
|
HITECH
|
The Health Information Technology for Economic and Clinical Health Act of 2009, 42 USC 17391 et. seq
|
|
IBNR
|
Incurred-But-Not-Reported
|
|
ICD-10
|
International Statistical Classification of Diseases and Related Health Problems (10th edition)
|
|
LEIE
|
List of Excluded Individuals and Entities
|
|
MAC
|
Maximum Allowable Cost
|
|
M-CHAT
|
Modified Checklist for Autism in Toddlers
|
|
MCO
|
Managed Care Organization
|
|
MD
|
Medical Doctor
|
|
MHSIP
|
Mental Health Statistics Improvement Program
|
|
MMIS
|
Medicaid Management Information System
|
|
NCQA
|
National Committee for Quality Assurance
|
|
NEMT
|
Non-Emergency Medical Transportation
|
|
NPI
|
National Provider Identifier
|
|
NPL
|
National Provider List
|
|
NPPES
|
National Plan and Provider Enumeration System
|
|
NQMC
|
National Quality Measures Clearinghouse
|
|
ONCHIT
|
Office of the National Coordinator for Health Information Technology
|
|
P&T
|
Pharmacy and Therapeutics
|
|
PBM
|
Pharmacy Benefit Manager
|
|
PCP
|
Primary Care Physician
|
|
PDL
|
Preferred Drug List
|
|
PHI
|
Personal Health Information
|
|
PIP
|
Performance Improvement Projects
|
|
PMG
|
Primary Medical Group
|
|
PPA
|
Pharmacy Program Administrator
|
Page 30 of 301
|
PPACA
|
Patient Protection and Affordable Care Act
|
|
PPN
|
Preferred Provider Network
|
|
PRHIEC
|
Puerto Rico Health Information Exchange Corporation
|
|
QAPI
|
Quality Assessment Performance Improvement Program
|
|
QIP
|
Quality Improvement Procedure
|
|
RFP
|
Request for Proposals
|
|
Rh
|
Rhesus
|
|
RHC
|
Rural Health Center/Clinic
|
|
SAMHSA
|
Substance Abuse and Mental Health Services Administration
|
|
SAS
|
Statements on Auditing Standards
|
|
SMI/SED
|
Serious Mental Illness/Serious Emotional Disability
|
|
SSN
|
Social Security Number
|
|
SUDs
|
Substance Use Disorders
|
|
TDD
|
Telecommunication Device for the Deaf
|
|
TPL
|
Third Party Liability
|
|
UM
|
Utilization Management
|
|
US
|
United States of America
|
|
USC
|
United States Code
|
ARTICLE 4 ASES RESPONSIBILITIES
| 4.1 | General Provision |
ASES will be responsible for administering the GHP. ASES will administer contracts, monitor MCOs’ performance, and provide oversight of all aspects of the MCOs’ operations.
| 4.2 | Legal Compliance |
ASES will comply with, and will monitor the Contractor’s compliance with, all applicable Puerto Rico and Federal laws and regulations, including but not limited to those listed in Attachment 1.
| 4.3 | Coordination with Contractor’s Key Staff |
| 4.3.1 | ASES will make diligent, good-faith efforts to facilitate effective and continuous communication and coordination with the Contractor in all areas of the GHP operations. |
Page 31 of 301
| 4.3.2 | Specifically, ASES will designate individuals within ASES who will serve as liaisons to corresponding individuals on the Contractor’s staff, including: |
| 4.3.2.1 | A program integrity staff member; |
| 4.3.2.2 | A quality oversight staff member; |
| 4.3.2.3 | A financial management staff member; |
| 4.3.2.4 | A Grievance System staff member; and |
| 4.3.2.5 | An Information Systems coordinator. |
| 4.4 | Information Systems and Reporting |
| 4.4.1 | ASES reserves the right to modify, expand, or delete the requirements contained in Article 17 with respect to the Data that Contractor is required to submit to ASES, or to issue new requirements, subject to consultation with Contractor and to cost negotiation, if necessary. Unless otherwise stipulated in the Contract or mutually agreed upon by the Parties, the Contractor shall have ninety (90) Calendar Days from the day on which ASES issues notice of a required modification, addition, or deletion, to comply with the modification, addition, or deletion. Any payment made by ASES that is based on data submitted by the Contractor is contingent upon the Contractor’s compliance with the Certification requirements contained in 42 CFR 438.606. |
| 4.4.2 | ASES will make available a secure FTP server, accessible via the Internet, for receipt of electronic files and reports from the Contractor. The Contractor shall provide a similar system for ASES to transmit files and reports deliverable by ASES to the Contractor. When such systems are not operational, ASES and the Contractor shall agree mutually on alternate methods for the exchange of files. |
| 4.4.3 | ASES will deliver to the Contractor the following information: |
| 4.4.3.1 | On a Daily Basis: |
| 4.4.3.1.1 | Certifications and Negative Redetermination Decisions; and |
| 4.4.3.1.2 | Enrollment rejections and errors; |
| 4.4.3.2 | On a Daily and monthly Basis: Eligibility Data (including Certification and Negative Redetermination Decisions); and |
| 4.4.3.3 | On a monthly Basis: PMPM Payments. |
Page 32 of 301
| 4.5 | Readiness Review |
| 4.5.1 | ASES will conduct readiness reviews of the Contractor’s operations that will include, at a minimum, one (1) on-site review, at dates and times to be determined by ASES. These reviews may include, but are not limited to, desk and on-site reviews of documents provided by the Contractor, walk-through(s) of the Contractor’s facilities, Information System demonstrations, and interviews with the Contractor’s staff. ASES will conduct the readiness review to confirm that the Contractor is capable and prepared to perform all Administrative Functions and to provide high-quality services to GHP Enrollees. |
| 4.5.2 | The Contractor shall submit policies and procedures and other Deliverables specified by ASES in accordance with Attachment 12. The Contractor shall make any changes requested by ASES to policies and procedures or other Deliverables in the timeframes specified by ASES. |
| 4.5.3 | ASES’s review will document the status of the Contractor’s compliance with the program standards set forth in this Contract. A multidisciplinary team appointed by ASES will conduct the readiness review. The scope of the readiness review will include, but not be limited to, the review and/or verification of: |
| 4.5.3.1 | Provider Network composition and Access; |
| 4.5.3.2 | Staff; |
| 4.5.3.3 | Provider Credentialing; |
| 4.5.3.4 | Call Center; |
| 4.5.3.5 | Care Management; |
| 4.5.3.6 | Marketing Materials; |
| 4.5.3.7 | Content of Provider contracts; |
| 4.5.3.8 | EPSDT plan; |
| 4.5.3.9 | Enrollee services capability; |
| 4.5.3.10 | Comprehensiveness of Quality and Utilization Management strategies; |
| 4.5.3.11 | Policies and procedures for the Grievance System; |
| 4.5.3.12 | Financial solvency; |
Page 33 of 301
| 4.5.3.13 | Contractor litigation history, current litigation, audits and other government investigations both in Puerto Rico and in other jurisdictions; |
| 4.5.3.14 | Information Systems performance, interfacing capabilities, and security management functions and capabilities; and |
| 4.5.3.15 | All other matters which ASES may deem reasonable in order to determine the Contractor’s compliance with the requirements of this Contract. |
| 4.5.4 | The readiness review may assess the Contractor’s ability to meet any requirements set forth in this Contract and the documents referenced herein. |
| 4.5.5 | Potential Enrollees may not be enrolled in a GHP Plan until ASES has determined that the Contractor is capable of meeting these standards. A Contractor’s failure to pass the readiness review may result in immediate Contract termination. If the Contract is terminated in accordance with this Section 4.5.5 of this Contract, ASES shall not make any payments to the Contractor and shall have no liability for any costs incurred by the Contractor. |
| 4.5.6 | ASES will provide the Contractor with a summary of findings from the readiness review, as well as areas requiring remedial action with the timeframes to correct the findings. |
ARTICLE 5 ELIGIBILITY AND ENROLLMENT
| 5.1 | Eligibility |
| 5.1.1 | The Commonwealth has sole authority to determine eligibility for the GHP, as provided in Federal law and Puerto Rico’s State Plan, with respect to the Medicaid and CHIP Eligibles; and, with respect to the Other Eligible Persons listed in Section 1.3.1.2, as provided in Article VI, Section 5 of Act 72 and other Puerto Rico law and Regulation 7758 – Regulation Number 138 of the Health Department. |
| 5.1.2 | The Puerto Rico Medicaid Program’s determination that a person is eligible for the GHP is contained on Form MA-10, titled “Notification of Action Taken on Application and/or Recertification.” A person who has received a MA-10 shall be referred to hereinafter as a “Potential Enrollee.” The Potential Enrollee may access Covered Services using the MA-10 as a temporary Enrollee ID Card from the first day of the eligibility period specified on the MA-10 even if the person has not received an Enrollee ID Card. Only Medicaid, CHIP, and Commonwealth Enrollees receive an MA-10 and may access Covered Services with the MA-10 as a temporary Enrollee ID Card. |
Page 34 of 301
| 5.1.3 | Effective Date of Eligibility. ASES shall provide the Effective Date of Eligibility for services under the GHP to the Contractor for all Potential Enrollees as follows: |
| 5.1.3.1 | Effective Date of Eligibility for Medicaid and CHIP Eligibles (see Section 1.3.1.1) is the eligibility period specified on the Form MA-10 which is the first day of the month in which the Potential Enrollee submits its eligibility application with the Medicaid Program Office and they shall be eligible to be enrolled as of that date. The eligibility period specified on the MA-10 may be a retroactive eligibility period which is up to ninety (90) Calendar Days before the first day of the month in which the Potential Enrollee submits its eligibility application with the Medicaid Program Office for the Medicaid and CHIP populations only during which services can be retroactively covered. |
| 5.1.3.2 | Effective date of Eligibility for the Commonwealth Population (see Section 1.3.1.2.1) is the eligibility period specified on the Form MA-10 and they shall be eligible to be enrolled as of that date. |
| 5.1.3.3 | Public employees and pensioners (see Section 1.3.1.2.2) shall be eligible to enroll in the GHP according to policies determined by the Commonwealth and their eligibility period shall be determined through such policies. The Puerto Rico Medicaid Program and ASES do not play a role in determining the eligibility for public employees and pensioners. |
| 5.1.4 | Termination of Eligibility |
| 5.1.4.1 | A Medicaid, CHIP, or Commonwealth Enrollee who is determined ineligible for the GHP after a Redetermination conducted by the Puerto Rico Medicaid Program shall remain eligible for services under the GHP until the date specified in a Negative Redetermination Decision on the MA-10 issued by the Puerto Rico Medicaid Program. |
| 5.1.4.2 | An Enrollee who is a public employee or pensioner (see Section 1.3.1.2.2) shall remain eligible until disenrolled from the GHP by the applicable Commonwealth agency. |
| 5.1.5 | ASES Notice to Contractor |
| 5.1.5.1 | ASES will receive a file with Certification and Negative Redetermination Decision Data from the Puerto Rico Medicaid Program on a Daily Basis concerning the Enrollment status of the Medicaid, CHIP, and Commonwealth Populations, and shall notify the Contractor of a Certification or Negative Redetermination Decision within one (1) Business Day of receiving notice of it via said file. ASES shall forward these Data to the Contractor in an electronic format agreed to between the Parties (the “Daily Update / Carrier Eligibility File Format”). |
Page 35 of 301
| 5.1.5.2 | The applicable Commonwealth agency will directly notify the Contractor of the Enrollment and Disenrollment status of public employees and pensioners. |
| 5.2 | Enrollment |
| 5.2.1 | The Contractor shall coordinate with ASES as necessary for all Enrollment and Disenrollment functions. |
| 5.2.1.1 | The Contractor shall accept all Potential Enrollees into its Plan without restrictions. The Contractor shall not discriminate against individuals on the basis of religion, gender, race, color, national origin, or sexual preference, and will not use any policy or practice that has the effect of discriminating on the basis of religion, gender, race, color, or national origin or on the basis of health, health status, pre-existing condition, or need for health care services. |
| 5.2.1.2 | The Contractor shall maintain adequate capacity in the Metro North & West Regions, to ensure prompt and voluntary Enrollment of all Potential Enrollees, on a Daily Basis and in the order in which they apply. |
| 5.2.1.3 | The Contractor shall provide Potential Enrollees with specific Information allowing for prompt, voluntary, and reliable Enrollment. |
| 5.2.1.4 | The Contractor guarantees the maintenance, functionality, and reliability of all systems necessary for Enrollment and Disenrollment. |
| 5.2.2 | Effective Date of Enrollment |
| 5.2.2.1 | Except as provided below, Enrollment, whether chosen or automatic, will be effective (hereinafter referred to as the “Effective Date of Enrollment”) the same date as the period of eligibility specified on the MA-10. |
| 5.2.2.2 | Effective Date of Enrollment for Newborns. A newborn shall be auto-enrolled, with an Effective Date of Enrollment as of the date of his or her birth. See Auto-Enrollment procedures for newborns in Section 5.2.6. |
| 5.2.2.3 | Re-Enrollment Policy and Effective Date of Re-Enrollment for Mothers Who are Minor Dependents. In the event that a female Enrollee who is included in a family group for coverage under the GHP as a Dependent child becomes pregnant, the Enrollee shall be referred to the Puerto Rico Medicaid Program. She will effectively establish a new family with the diagnosis of her pregnancy and will become the head of household of the new family. The eligibility period of the new family will begin on the date of the first diagnosis of the pregnancy, and the Enrollee shall be Auto-Enrolled, effective as of this date. The mother shall be Auto-Assigned to the PMG and PCP to which she was assigned before the Re-Enrollment. |
Page 36 of 301
| 5.2.2.4 | Effective Date of Re-Enrollment for Enrollees Who Lose Eligibility. If an Enrollee who is a Medicaid- or - CHIP Eligible Person or member of the Commonwealth Population loses eligibility for the GHP for a period of less than two (2) months in duration, Enrollment in the Contractor’s Plan shall be reinstated. Upon notification from ASES of the Recertification, the Contractor shall Auto-Enroll the person, with Enrollment effective as of the eligibility period specified on the MA-10. |
| 5.2.3 | Term of Enrollment. The Term of Enrollment shall be a period of twelve (12) consecutive months for all GHP Enrollees, except that in cases in which the Puerto Rico Medicaid Program has designated an eligibility period shorter than twelve (12) months for an Enrollee who is a Medicaid or CHIP Eligible or a member of the Commonwealth Population, that same period shall also be considered the Enrollee’s Term of Enrollment. Such a shortened eligibility period may apply, at the discretion of the Puerto Rico Medicaid Program, when an Enrollee is pregnant, is homeless, or anticipates a change in status (such as receipt of unemployment benefits or in family composition). Notwithstanding this Section, Section 5.3.3 controls the Effective Date of Disenrollment. |
| 5.2.3.1 | Except as otherwise provided in this Section 5.2, and notwithstanding the Term of Enrollment provided in Section 5.2.3, Enrollees shall remain enrolled in the Contractor’s Plan until the occurrence of an event listed in Section 5.3 (Disenrollment). |
| 5.2.4 | Auto-Enrollment. The Contractor shall have the policies and procedures necessary to comply with Auto-Enrollment as of the Effective Date of the Contract for the Medicaid and CHIP Eligibles and members of the Commonwealth Population which shall be prior approved in writing by ASES. |
| 5.2.4.1 | The Contractor shall Auto-Enroll each Potential Enrollee in the GHP Plan covering the Service Region where the Potential Enrollee lives. |
Page 37 of 301
| 5.2.4.2 | The Auto-Enrollment process will include Auto-Assignment of a PMG and a PCP (see Section 5.4 of this Contract). A new Enrollee who is a Dependent of a current GHP Enrollee shall be automatically assigned to the same PMG as his or her parent or spouse who is a current GHP Enrollee. |
| 5.2.5 | Enrollment Procedures for All Enrollees Except Newborns |
| 5.2.5.1 | Upon receipt of the notices in accordance with Section 5.1.5 of this Contract, the Contractor shall comply with the process of Auto-Enrollment and issue to the Enrollee a notice informing the Enrollee of the PMG and PCP they are assigned to and their rights to change the PMG or PCP during a ninety (90) Calendar Day period from the date of the communication which informed them of their initial assignment by calling or visiting the Contractor's office. |
| 5.2.5.2 | Once the Enrollee calls or visits the Contractor’s office to execute the right of changing the assigned PMG, PCP, or both, the Contractor shall request that the Enrollee select a new PMG and PCP. During the visit or call, the Contractor shall issue to the Enrollee an Enrollee ID Card, a notice of Enrollment, an Enrollee Handbook, and a Provider Directory; or, such notice of Enrollment, an ID Card, a Handbook, and a Provider Directory may be sent to the Enrollee via surface mail within five (5) Business Days of the Enrollee’s request to change the Auto-Enrollment assignments. |
| 5.2.5.3 | The notice of Enrollment that the Contractor issues pursuant to Section 5.2.5.2 will clearly state the Effective Date of Enrollment. The notice of Enrollment will explain that the Enrollee is entitled to both physical and Behavioral Health Services through the Contractor’s Plan. The notice will inform the Enrollee of his or her limited right to disenroll, per Section 5.3 of this Contract. The notice of Enrollment shall inform the Enrollee that exercising the right to disenroll from the MCO only means losing access to services under the GHP. The notice shall advise the Enrollee of the Enrollee’s right to select a different PCP or to change PMGs, as described in Section 5.4, and will encourage the Enrollee to pursue this option if he or she is dissatisfied with care or services. |
| 5.2.6 | Procedures for Auto-Enrollment of Newborns |
| 5.2.6.1 | The Contractor shall notify ASES and the Puerto Rico Medicaid Program in writing of any Enrollees who are expectant mothers Immediately at the moment of diagnosis of the pregnancy or at least sixty (60) Calendar Days before the expected date of delivery. |
Page 38 of 301
| 5.2.6.2 | The Contractor shall promptly, upon learning that an Enrollee is an expectant mother, mail a newborn Enrollment packet to the expectant mother (i) instructing her to register the newborn with the Puerto Rico Medicaid Program within ninety (90) Calendar Days of birth by providing evidence of the newborn’s birth and birth certificate; (ii) notifying her that the newborn will be auto-enrolled in the GHP; (iii) informing her that unless she visits the Contractor’s office to select a PMG and PCP, the child will be auto-assigned to the mother’s PMG and to a PCP who is a pediatrician; and (iv) informing her that she will have ninety (90) Calendar Days after the child’s birth to disenroll the child from the Plan or to change the child’s PMG and PCP, without cause. |
| 5.2.6.3 | The Contractor shall provide assistance to any expectant mother who contacts the Contractor wishing to make a PCP and PMG selection for her newborn and record that selection, per Section 5.4. |
| 5.2.6.4 | If the mother has not made a PCP and PMG selection at the time of the child’s birth, the Contractor shall, within one (1) Business Day of the birth, auto-assign the newborn to a PCP who is a pediatrician and to the mother’s PMG. |
| 5.2.6.5 | Within one (1) Business Day of acknowledging, either by concurrent review or hospital notification of the birth of a child to an Enrollee, the Contractor shall ensure the submission of a newborn notification form to ASES and to the Puerto Rico Medicaid Program; such form shall be given prior written approval by ASES and the Puerto Rico Medicaid Program. |
| 5.2.6.6 | The Contractor shall participate in any meeting, working group, or other mechanism requested by ASES in order to ensure coordination among the Contractor, ASES, and the Puerto Rico Medicaid Program in order to implement newborn Auto-Enrollment. |
| 5.2.7 | Re-Enrollment Procedures |
| 5.2.7.1 | The Contractor shall inform Enrollees who are Medicaid- and CHIP Eligibles and members of the Commonwealth Population of an impending Redetermination through written notices. Such notices shall be provided ninety (90) Calendar Days, sixty (60) Calendar Days, and thirty (30) Calendar Days before the scheduled date of the Redetermination. The notice shall inform the Enrollee that, if he or she is recertified, his or her term of Enrollment in the Plan will automatically renew; but that, effective as of the date of Recertification, he or she will have a ninety- (90) Calendar Day period in which he or she may disenroll from the Plan without cause or may change his or her PMG and/or PCP selection without cause. The notice shall advise Enrollees that Disenrollment from the MCO only will terminate the Enrollee’s access to health services. |
Page 39 of 301
| 5.2.7.2 | The Contractor shall provide Enrollees and their representatives with sixty (60) Calendar Days written notice before the start of each term of Enrollment, as specified in Section 5.2.3, of the right to disenroll or to change PMG or PCP during the first ninety (90) Calendar Days of the new term of Enrollment. |
| 5.2.7.3 | Upon the receipt of written request from ASES, the Contractor shall provide a report for a specific period of time containing documentation that the Contractor has furnished the notices required in this Section 5.2.7. |
| 5.2.7.4 | The form letters used for the notices in this Section 5.2.7 shall fall within the requirements in Section 6.2.1 that the Contractor seek advance written approval from ASES of certain documents. |
| 5.2.8 | Specific Contractor Responsibilities Regarding Dual Eligible Beneficiaries. At the time of Enrollment, the Contractor shall provide Potential Enrollees who are Medicaid-eligible and are also eligible for Medicare Part A or Medicare Part A and Part B (“Dual Eligible Beneficiaries”) with the information about their Covered Services and Co-Payments that is listed in Section 6.13. Members of the Commonwealth Population (see section 1.3.1.2.1) who are Medicare-eligible shall not be considered Dual Eligible Beneficiaries. |
| 5.3 | Disenrollment |
| 5.3.1 | Disenrollment occurs only when ASES or the Medicaid Program determines that an Enrollee is no longer eligible for the GHP; or when Disenrollment is requested by the Contractor or Enrollee, and approved by ASES, as provided in Sections 5.3.4 and 5.3.5. |
| 5.3.2 | Disenrollment will be effected by ASES, and ASES will issue notification to the Contractor. Such notice shall be delivered via file transfer to the Contractor on a Daily Basis simultaneously with Information on Potential Enrollees within five (5) Calendar Days of making a final determination on Disenrollment. |
| 5.3.2.1 | Disenrollment decisions are the responsibility of ASES; however, notice to Enrollees of Disenrollment shall be issued by the Contractor. The Contractor shall issue such notice in person or via surface mail to the Enrollee within five (5) Business Days of a final Disenrollment decision, as provided in Sections 5.3.4 and 5.3.5. |
Page 40 of 301
| 5.3.2.2 | Each notice of Disenrollment shall include information concerning: |
| 5.3.2.2.1 | The Effective Date of Disenrollment; |
| 5.3.2.2.2 | The reason for the Disenrollment; |
| 5.3.2.2.3 | The Enrollee’s Appeal rights, including the availability of the Grievance System and of ASES’s Administrative Law Hearing process, as provided by Act 72 of September 7, 1993; |
| 5.3.2.2.4 | The right to re-enroll in the GHP upon receiving a Recertification from the Puerto Rico Medicaid Program, if applicable; and |
| 5.3.2.2.5 | Disenrollment shall occur according to the following timeframes (the “Effective Date of Disenrollment”). |
| 5.3.3 | The Effective Date of Disenrollment is as follows: |
| 5.3.3.1 | Except as otherwise provided in this Section 5.3, Disenrollment will take effect as of the Effective Date of Disenrollment specified in ASES’s notice to the Contractor that an Enrollee is no longer eligible. If ASES notifies the Contractor of Disenrollment on or before the last working day of the month in which eligibility ends, the Disenrollment will be effective on the first day of the following month. |
| 5.3.3.2 | When Disenrollment is effected at the Contractor’s or the Enrollee’s request, as provided in Sections 5.3.4 and 5.3.5 of this Contract, Disenrollment shall take effect no later than the first day of the second month following the month that the Contractor or Enrollee requested the Disenrollment. If ASES fails to make a decision on the Contractor’s or Enrollee’s request before this date, the Disenrollment will be deemed granted. If the Enrollee requests reconsideration of a Disenrollment through the Contractor’s Grievance System, as provided in Article 14, the Grievance System process shall be completed in time to permit the Disenrollment (if approved) to take effect in accordance with this timeframe. |
| 5.3.3.3 | If what would otherwise be the Effective Date of Disenrollment under this Section 5.3.3 falls: |
| 5.3.3.3.1 | When the Enrollee is an inpatient at a hospital, ASES shall postpone the Effective Date of Disenrollment so that it occurs on the last day of the month in which the Enrollee is discharged from the hospital, or the last day of the month following the month in which Disenrollment would otherwise be effective, whichever occurs earlier; |
Page 41 of 301
| 5.3.3.3.2 | During a month in which the Enrollee is in the second or third trimester of pregnancy, ASES shall postpone the Effective Date of Disenrollment so that it occurs on the date of delivery; or |
| 5.3.3.3.3 | During a month in which an Enrollee is diagnosed with a Terminal Condition, ASES shall postpone the Effective Date of Disenrollment so that it occurs on the last day of the following month. |
| 5.3.3.4 | For the public employees and pensioners who are Other Eligible Persons referred to in Section 1.3.1.2.2, Disenrollment shall occur according to the timeframes set forth in a Normative Letter issued by ASES annually. |
| 5.3.4 | Disenrollment Initiated by the Contractor |
| 5.3.4.1 | The Contractor has a limited right to request that an Enrollee be disenrolled without the Enrollee’s consent. The Contractor shall notify ASES upon identification of an Enrollee who it knows or believes meets the criteria for Disenrollment. |
| 5.3.4.2 | The Contractor shall submit Disenrollment requests to ASES, and the Contractor shall honor all Disenrollment determinations made by ASES. ASES’s decision on the matter shall be final, conclusive, and not subject to appeal by the Contractor. |
| 5.3.4.3 | The following are acceptable reasons for the Contractor to request Disenrollment: |
| 5.3.4.3.1 | The Enrollee’s continued Enrollment in the Contractor’s Plan seriously impairs the ability to furnish services to either this particular Enrollee or other Enrollees; |
| 5.3.4.3.2 | The Enrollee demonstrates a pattern of disruptive or abusive behavior that could be construed as non-compliant and is not caused by a presenting illness; |
| 5.3.4.3.3 | The Enrollee’s use of services is fraudulent or abusive (for example, the Enrollee has loaned his or her Enrollee ID Card to other persons to seek services); |
| 5.3.4.3.4 | The Enrollee has moved out of the Contractor’s Service Regions; |
Page 42 of 301
| 5.3.4.3.5 | The Enrollee is placed in a long-term care nursing facility or intermediate care facility for the intellectually disabled; |
| 5.3.4.3.6 | The Enrollee’s Medicaid or CHIP eligibility category changes to a category ineligible for the GHP; or |
| 5.3.4.3.7 | The Enrollee has died, been incarcerated, or moved out of Puerto Rico, thereby making him or her ineligible for Medicaid or CHIP or otherwise ineligible for the GHP. |
| 5.3.4.4 | ASES will approve a Disenrollment request by the Contractor, in ASES’s discretion, only if ASES determines: |
| 5.3.4.4.1 | That it is impossible for the Contractor to continue to provide services to the Enrollee without endangering the Enrollee or other GHP Enrollees; and |
| 5.3.4.4.2 | That an action short of Disenrollment, such as transferring the Enrollee to a different PCP or PMG, will not resolve the problem. |
| 5.3.4.5 | The Contractor may not request Disenrollment for any discriminatory reason including, but not limited, to the following: |
| 5.3.4.5.1 | Adverse changes in an Enrollee’s health status; |
| 5.3.4.5.2 | Missed appointments; |
| 5.3.4.5.3 | Utilization of medical services; |
| 5.3.4.5.4 | Diminished mental capacity; |
| 5.3.4.5.5 | Pre-existing medical condition; |
| 5.3.4.5.6 | The Enrollee’s attempt to exercise his or her rights under the Grievance System; or |
| 5.3.4.5.7 | Uncooperative or disruptive behavior resulting from the Enrollee’s special needs. |
| 5.3.4.6 | The request of one (1) PMG to have an Enrollee assigned to a different PMG, per Section 5.4, shall not be sufficient cause for the Contractor to request that the Enrollee be disenrolled from the Plan. Rather, the Contractor shall, if possible, assign the Enrollee to a different and available PMG within the Plan. |
| 5.3.4.7 | When requesting Disenrollment of an Enrollee for reasons described in Section 5.3.4.3, the Contractor shall document at least three (3) interventions over a period of ninety (90) Calendar Days that occurred through treatment and Care Management to resolve any difficulty leading to the request. The Contractor shall also provide evidence of having given at least one (1) written warning to the Enrollee, with a certified return receipt requested, regarding implications of his or her actions. |
Page 43 of 301
| 5.3.4.8 | If the Enrollee has demonstrated abusive or threatening behavior as defined by ASES, only one (1) Contractor intervention, and a subsequent written attempt to resolve the difficulty, are required. |
| 5.3.4.9 | In the event that the Contractor seeks Disenrollment of an Enrollee, the Contractor must notify the Enrollee of the availability of the Grievance System and of ASES’s Administrative Law Hearing process, as provided by Act 72 of September 7, 1993, as amended. |
| 5.3.4.10 | The Contractor shall maintain policies and procedures to comply with the Puerto Rico Patients’ Bill of Rights Act and with the Medicaid Regulations of 42 CFR 438.100, to ensure that the Enrollee’s exercise of Grievance rights does not adversely affect the services provided to the Enrollee by the Contractor or by ASES. |
| 5.3.5 | Disenrollment Initiated by the Enrollee |
| 5.3.5.1 | An Enrollee wishing to request Disenrollment must submit an oral or written request to ASES or to the Contractor. If the request is made to the Contractor, the Contractor shall forward the request to ASES, within ten (10) Business Days of receipt of the request, with a recommendation of the action to be taken. |
| 5.3.5.2 | An Enrollee may request Disenrollment from the Contractor’s Plan without cause during the ninety (90) Calendar Days following the Effective Date of Enrollment with the Plan or the date that the Contractor sends the Enrollee notice of the Enrollment, whichever is later. An Enrollee may request Disenrollment without cause every twelve (12) months thereafter. In addition, an Enrollee may request Disenrollment without cause in the event that ASES notifies the Enrollee that ASES has imposed or intends to impose on the Contractor the intermediate sanctions set forth in 42 CFR 438.702(a)(3). |
| 5.3.5.3 | An Enrollee may request Disenrollment from the Contractor’s Plan for cause at any time. ASES shall determine whether the reason constitutes a valid cause. The following constitute cause for Disenrollment by the Enrollee: |
Page 44 of 301
| 5.3.5.3.1 | The Enrollee moves to a Service Region not covered by the Contractor, or outside of Puerto Rico; |
| 5.3.5.3.2 | The Enrollee needs related services to be performed at the same time, and not all related services are available within the network. The Enrollee’s PCP or another Provider in the Preferred Provider Network have determined that receiving services separately would subject the Enrollee to unnecessary risk; and |
| 5.3.5.3.3 | Other acceptable reasons for Disenrollment at Enrollee request, per 42 CFR 438.56(d)(2), including, but not limited to, poor quality of care, lack of Access to Covered Services, or lack of Providers experienced in dealing with the Enrollee’s health care needs. |
| 5.3.5.4 | If the Contractor fails to refer a Disenrollment request within the timeframe specified in Section 5.3.3, or if ASES fails to make a Disenrollment determination so that the Enrollee may be disenrolled by the first day of the second month following the month when the Disenrollment request was made, per Section 5.3.3, the Disenrollment shall be deemed approved at that time. |
| 5.3.5.5 | ASES shall make the final decision on Enrollees’ requests for Disenrollment. ASES may approve or disapprove the request based on the reasons specified in the Enrollee’s request, or upon any relevant Information provided to ASES by the Contractor about the Disenrollment request. |
| 5.3.5.6 | If the Enrollee’s request for Disenrollment under this Section is denied, the Contractor shall provide the Enrollee with a notice of the decision. The notice shall include the grounds for the denial and shall inform the Enrollee of his or her right to use the Grievance System as provided in Article 14, and to have Access to an Administrative Law Hearing after first exhausting the Contractor’s Grievance System. |
| 5.3.5.7 | Use of the Contractor’s Grievance System. ASES may at its option require that the Enrollee seek redress through the Contractor’s Grievance System before ASES makes a determination on the Enrollee’s request for Disenrollment. The Contractor shall Immediately inform ASES of the outcome of the Grievance process. ASES may take this Information into account in making a determination regarding the request for Disenrollment. The Grievance process must be completed in time to permit the Disenrollment (if approved) to be effective in accordance with the timeframe specified in Section 5.3.3; if the process is not completed within the specified timeframe, then the Disenrollment will be deemed approved by ASES. |
Page 45 of 301
| 5.3.6 | Disenrollment during Termination Hearing Process. If ASES notifies the Contractor of its intention to terminate the Contract as provided in Article 35, ASES may allow Enrollees to disenroll Immediately without cause. In the event of such a Termination, ASES must provide Enrollees with the notice required by 42 CFR 438.10, listing their options for receiving services following the Termination Date of the Contract. |
| 5.3.7 | ASES shall ensure, through the obligations of the Contractor under this Contract that Enrollees receive the notices contained in Section 5.2.7 (Re-Enrollment Procedures). While these notices shall be issued by the Contractor, per Section 5.2.7], ASES shall provide the Contractor with the information on Certifications and Negative Redetermination Decisions (see Section 5.1.5.1) needed for the Contractor to carry out this responsibility. |
| 5.3.8 | Enrollment Database |
| 5.3.8.1 | The Contractor shall maintain an Enrollment database that includes all Enrollees, and contains, for each Enrollee, the Information specified in the Carrier Billing File/Carrier Eligibility File format. |
| 5.3.8.2 | The Contractor shall notify the Puerto Rico Medicaid Program Immediately when the Enrollment database is updated to reflect a change in the place of residence of an Enrollee. |
| 5.3.8.3 | The Contractor shall secure any authorization required from Enrollees under the laws of Puerto Rico in order to allow the US Department of Health and Human Services, and ASES and its Agents to review Enrollee Medical Records, in order to evaluate the Information and determine quality, appropriateness, timeliness, and cost of services performed under this Contract; provided that such authorization shall be limited by the Contractor’s obligation to observe the confidentiality of Enrollees’ Personal Health Information, as provided in Article 34. |
| 5.3.9 | Notification to ASES and the PBM of New Enrollees and of Completed Disenrollments |
| 5.3.9.1 | The Contractor shall notify ASES and the PBM of new Enrollees and of completed Disenrollments on a routine Daily Basis; or at any time, if requested by ASES. Such notification will be made through electronic transmissions. |
| 5.3.9.2 | The notification will include all new Enrollees as of the Business Day before the notification is issued, and will be sent no later than the following Business Day after the Enrollment process has been completed (as signified by issuance of the Enrollee ID Card, either in person or by surface mail) or the Disenrollment process has been completed (as signified by the issuance of a Disenrollment notice). |
Page 46 of 301
| 5.3.10 | In the event that the Contractor must update information previously submitted to ASES about a new Enrollment, or that the Contractor must add a new Enrollee who was previously omitted, such update must occur the next Business Day after the information is updated or a new Enrollee is added. ASES reserves the authority not to accept any new additions or corrections to Enrollment Data after sixty (60) Calendar Days past the Effective Date of Enrollment stated in the Contractor’s notification to ASES. |
| 5.4 | Auto-Assignment and Change of a Primary Medical Group (“PMG”) and Primary Care Physician (“PCP”) |
| 5.4.1 | Change of a PMG and PCP |
| 5.4.1.1 | During the ninety (90) Calendar Days period the Enrollee can change his/her auto-assigned PMG and PCP. The Contractor can offer counseling and assistance to the enrollee in selecting a different PCP and PMG. |
| 5.4.1.2 | The Contractor shall advise certain Enrollees to choose a physician other than, or in addition to, a general practice physician as their PCP, as follows: |
| 5.4.1.2.1 | Female Enrollees will be recommended to choose an obstetrician / gynecologist as a PCP. |
| 5.4.1.2.2 | Enrollees under twenty-one (21) years of age will be recommended to choose a pediatrician as a PCP. |
| 5.4.1.2.3 | Enrollees with Chronic Conditions including heart failure, kidney failure, or diabetes will be recommended to choose an internist as a PCP. |
| 5.4.1.3 | Per Section 5.2.6, following the Contractor’s notice to an expectant mother of her child’s upcoming Auto-Enrollment in the Contractor’s Plan, the Contractor shall record any notice it receives from the mother concerning the selection of a PCP or PMG for the child. The Contractor shall ensure that such selections take effect as of the date of the child’s birth. |
Page 47 of 301
| 5.4.1.4 | Enrollee PCP and PMG changes shall take effect on the next Business Day following the Enrollee’s decision to change PCP and/or PMG. |
| 5.4.1.5 | The Contractor shall permit Enrollees to change their PMG or PCP at any time with cause. The following shall constitute cause for change of PMG or PCP: |
| 5.4.1.5.1 | The Enrollee’s religious or moral convictions conflict with the services offered by Providers in the PMG; |
| 5.4.1.5.2 | The Enrollee needs related services to be provided concurrently; not all services are available within the Preferred Provider Network associated with a PMG; and the Enrollee’s PCP or any other Provider has determined that receiving the services separately could expose the Enrollee to an unnecessary risk; or |
| 5.4.1.5.3 | Other reasons, including poor quality of care, inaccessibility to Covered Services, and inaccessibility to Providers with the experience to address the health care needs of the Enrollee. |
| 5.4.1.6 | The Contractor shall permit Enrollees to change their PMG and/or PCP for any reason, within certain timeframes: |
| 5.4.1.6.1 | During the ninety (90) Calendar Days following the Effective Date of Enrollment; |
| 5.4.1.6.2 | At least every twelve (12) months, following the ninety (90) Calendar Days after the Effective Date of Enrollment; or |
| 5.4.1.6.3 | At any time, during time periods in which the Contractor is subject to intermediate sanctions, as defined in 42 CFR 438.702(a)(3). |
| 5.4.1.7 | A Contractor may change an Enrollee’s PMG at the request of the PCP or another Provider within that PMG, in limited situations, as follows: |
| 5.4.1.7.1 | The Enrollee’s continued participation in the PMG seriously impairs the PMG’s ability to furnish services to either this particular Enrollee or other Enrollees; |
| 5.4.1.7.2 | The Enrollee demonstrates a pattern of disruptive or abusive behavior that could be construed as non-compliant and that is not caused by a presenting illness; or |
Page 48 of 301
| 5.4.1.7.3 | The Enrollee’s use of services is fraudulent or abusive (for example, the Enrollee has loaned his or her Enrollee ID Card to other persons to seek services). |
ARTICLE 6 ENROLLEE SERVICES
| 6.1 | General Provisions |
| 6.1.1 | The Contractor shall have policies and procedures, prior approved by ASES and submitted in accordance with Attachment 12, that explain how it will ensure that Enrollees: |
| 6.1.1.1 | Are aware of their rights and responsibilities; |
| 6.1.1.2 | How to obtain physical and Behavioral Health Services; |
| 6.1.1.3 | What to do in an emergency or urgent medical situation; |
| 6.1.1.4 | How to request a Grievance, Appeal, or Administrative Law Hearing; |
| 6.1.1.5 | How to report suspected Incident of Fraud, Waste, and Abuse; |
| 6.1.1.6 | Have basic information on the basic features of managed care; and |
| 6.1.1.7 | Understand the MCO’s responsibilities to coordinate Enrollee care. |
| 6.1.2 | The Contractor’s informational materials must convey to Enrollees that GHP is an integrated program that includes both physical and Behavioral Health Services, and must also explain the concepts of Primary Medical Groups and Preferred Provider Networks. |
| 6.1.3 | The information conveyed in the Contractor’s written materials shall conform with ASES’s Universal Beneficiary Guidelines, included as Attachment 3 to this Contract. |
| 6.1.4 | The Contractor shall convey Information to Enrollees and Potential Enrollees via written materials and via telephone, internet, and face-to-face communications, and shall allow Enrollees to submit questions and to receive responses from the Contractor. |
| 6.1.5 | The Contractor shall ensure that the informational materials disseminated to all GHP Enrollees accurately identify differences among the categories of Eligible Persons. |
| 6.1.6 | The Contractor shall provide Enrollees with at least thirty (30) Calendar Days written notice of any significant change in policies concerning Enrollees’ Disenrollment rights (see Section 5.3), right to change PMGs or PCPs (see Section 5.4), or any significant change to any of the items listed in Enrollee Rights and Responsibilities (section 6.5), regardless of whether ASES or the Contractor caused the change to take place. This Section 6.1.6 shall not be construed as giving the Contractor the right to change its policies and procedures without prior written approval from ASES. |
Page 49 of 301
| 6.2 | ASES Approval of All Written Materials |
| 6.2.1 | The Contractor shall submit to ASES for review and prior written approval all materials meant for distribution to Enrollees, including but not limited to, Enrollee Handbooks, Provider Directories, ID cards and, upon request, any other additional, but not required, materials and Information provided to Enrollees designed to promote health and/or educate Enrollees. |
| 6.2.2 | All materials must be submitted to ASES in paper and electronic file media, in the format prescribed by ASES. The Contractor shall submit the reading level and the methodology used to measure it concurrent with all submissions of written materials and include a plan that describes the Contractor’s intent for the use of the materials. |
| 6.2.3 | ASES reserves the right to notify the Contractor to discontinue or modify written materials after approval. |
| 6.2.4 | Except as otherwise provided below, written materials described in this Article 6 must be submitted to ASES for review at least forty-five (45) Calendar Days before their printing and distribution, as required by Act 194 of August 2000. This requirement applies to: |
| 6.2.4.1 | The materials described in this Article 6 distributed to all Enrollees, including the Enrollee Handbook; |
| 6.2.4.2 | Policy letters, coverage policy statements, or other communications about Covered Services under the GHP distributed to Enrollees; and |
| 6.2.4.3 | Standard letters and notifications, such as the notice of Enrollment required in Section 5.2.5.3, the notice of Redetermination required in Section 5.2.7.1, and the notice of Disenrollment required in Section 5.3.2. |
| 6.2.5 | The Contractor shall provide ASES with advance notice of any changes made to written materials that will be distributed to all Enrollees. Notice shall be provided to ASES at least forty-five (45) Calendar Days before the effective date of the change. Within fifteen (15) Business Days of receipt of the materials, ASES will respond to the Contractor’s submission with either an approval of the materials, recommended modifications, or a notification that more review time is required. If the Contractor receives no response from ASES within fifteen (15) Business Days of ASES’s receipt of the materials, the materials shall be deemed approved. Except as otherwise provided in this Section 6.2.5, the Contractor may distribute the revised written materials only upon written approval of the changes from ASES. |
Page 50 of 301
| 6.3 | Requirements for Written Materials |
| 6.3.1 | The Contractor shall maintain written policies and procedures governing the development and distribution of written materials including how the Contractor will meet the requirements in this Section 6.3, with such policies and procedures to be submitted in accordance with Attachment 12 for prior written approval from ASES. The Contractor shall, at a minimum, have policies and procedures regarding the process for developing/creating, proofing, approving, publishing, and mailing the (i) Enrollee ID card, (ii) Enrollee Handbook, (iii) Provider Directory, and (iv) form letters within contractual standards and timeframes. The Contractor shall include a separate set of policies and procedures for each of the items listed above (i-iv). |
| 6.3.2 | The Contractor shall make all written materials available in alternative formats and in a manner that takes into consideration the Enrollee’s special needs, including Enrollees who are visually impaired or have limited reading proficiency. The Contractor shall notify all Enrollees and Potential Enrollees that Information is available in alternative formats, and shall instruct them on how to access those formats. |
| 6.3.3 | Once an Enrollee has requested a written material in an alternative format or language, the Contractor shall (i) make a notation of the Enrollee’s preference in the Contractor’s system and (ii) provide all subsequent written materials to the Enrollee in such format unless the Enrollee requests otherwise. |
| 6.3.4 | Except as provided in Sections 1.1.5 and 6.4 (Enrollee Handbook) and subject to Section 6.3.8, the Contractor shall make all written information available in Spanish, with a language block in English, explaining that (i) Enrollees may access an English translation of the Information if needed, and (ii) the Contractor will provide oral interpretation services into any language other than Spanish or English, if needed. Such translation or interpretation shall be provided by the Contractor at no cost to the Enrollee. The language block shall comply with 42 CFR 438.10(c)(2). |
| 6.3.5 | If oral interpretation services are required in order to explain the Benefits covered under the GHP to a Potential Enrollee who does not speak either English or Spanish, the Contractor must, at its own cost, make such services available in a third language, in compliance with 42 CFR 438.10(c)(4). |
Page 51 of 301
| 6.3.6 | All written materials shall be worded such that they are understandable to a person who reads at the fourth (4th) grade level. |
| 6.3.7 | All written materials must be clearly legible with a minimum font of size twelve (12) point with the exception of Enrollee ID cards and unless otherwise approved in writing by ASES. |
| 6.3.8 | Within ninety (90) Calendar Days of a notification from ASES that ASES has identified a Prevalent Non-English Language other than Spanish or English (with “Prevalent Non-English Language” defined as a language that is the primary language of more than five percent (5%) of the population of Puerto Rico), all written materials provided to Enrollees shall be translated into and made available in such language. |
| 6.3.9 | The Contractor shall provide written notice to Enrollees of any material changes to written materials previously distributed to Enrollees at least thirty (30) Calendar Days before the effective date of the change. |
| 6.4 | Enrollee Handbook Requirements |
| 6.4.1 | The Contractor shall produce at its sole cost, and shall mail to all new Enrollees, an Enrollee Handbook including information on physical health, Behavioral Health, and all other Covered Services offered under the GHP. The Contractor shall distribute the Handbook either simultaneously with the notice of Enrollment referenced in Section 5.2.5.3 or within five (5) Calendar Days of sending the notice of Enrollment via surface mail. |
| 6.4.2 | Upon request of an Enrollee or his/her Authorized Representative for a replacement or additional copy of the Enrollee Handbook, the Contractor shall send an Enrollee Handbook within ten (10) Calendar Days. The Contractor shall give the person requesting an Enrollee Handbook the option to get the Information from the Contractor’s website or to receive a printed document. |
| 6.4.3 | The Contractor shall either: |
| 6.4.3.1 | Mail to all Enrollees an Enrollee Handbook on at least an annual basis, after the initial distribution of the Handbook at Enrollment; or |
| 6.4.3.2 | At least annually, as required by 42 CFR 438.10(i), mail to all Enrollees a Handbook supplement that includes Information on the following: |
| 6.4.3.2.1 | The Contractor’s service area; |
| 6.4.3.2.2 | Benefits covered under the GHP; |
Page 52 of 301
| 6.4.3.2.3 | Any cost-sharing imposed by the Contractor; and |
| 6.4.3.2.4 | To the extent available, quality and performance indicators, including Enrollee satisfaction. |
| 6.4.4 | The Contractor shall use the Universal Beneficiary Guide, provided by ASES and included as Attachment 3 to this Contract, as a model for its Enrollee Handbook; however, the Contractor shall ensure that its Enrollee Handbook meets all the requirements listed in this Section 6.4. |
| 6.4.5 | Pursuant to the requirements set forth in 42 CFR 438.10, the Enrollee Handbook shall include, at a minimum, the following: |
| 6.4.5.1 | A table of contents; |
| 6.4.5.2 | An explanation of the purpose of the Enrollee ID Card and a warning that transfer of the card to another person constitutes Fraud; |
| 6.4.5.3 | Information about the role of the PCP and how to choose a PCP; |
| 6.4.5.4 | Information about the PMG, how to choose a PMG, and which Benefits may be accessed through the PMG; |
| 6.4.5.5 | Information about the PPN associated with the Enrollee’s PMG, and the benefits of seeking services within the PPN; |
| 6.4.5.6 | Information about the circumstances under which Enrollees may change to a different PMG; |
| 6.4.5.7 | Information about what to do when family size changes, including the responsibility of new mothers who are Medicaid Eligible to register their newborn with the Puerto Rico Medicaid Program and to apply for the Enrollment of the newborn; |
| 6.4.5.8 | Appointment procedures; |
| 6.4.5.9 | Information on Benefits and Covered Services, including how the scope of Benefits and services differs between Medicaid- and CHIP Eligibles and Other Eligible Persons; |
| 6.4.5.10 | An explanation of how physical health and Behavioral Health Services are integrated under the GHP, and how to access Behavioral Health Services; |
| 6.4.5.11 | Information on how to access local resources for Non-Emergency Medical Transportation (“NEMT”); |
Page 53 of 301
| 6.4.5.12 | An explanation of any service limitations or exclusions from coverage; |
| 6.4.5.13 | Information on where and how Enrollees may access Benefits not available from or not covered by the Contractor’s Plan; |
| 6.4.5.14 | The Medical Necessity definition used in determining whether services will be covered (see Section 7.2); |
| 6.4.5.15 | A description of all pre-certification, Prior Authorization, or other requirements for treatments and services; |
| 6.4.5.16 | The policy on Referrals for specialty care and for other Covered Services not provided by the Enrollee’s PCP; |
| 6.4.5.17 | Information on how to obtain services when the Enrollee is outside the Contractor’s Service Region(s); |
| 6.4.5.18 | Information on how to obtain after-hours coverage; |
| 6.4.5.19 | An explanation of cost-sharing, including: |
| 6.4.5.19.1 | The differences in cost-sharing responsibilities between Medicaid- and CHIP Eligibles and Other Eligible Persons, and |
| 6.4.5.19.2 | The cost-sharing responsibilities of Dual Eligible Beneficiaries, as well as the other information for Dual Eligible Beneficiaries listed in Section 6.13; |
| 6.4.5.20 | The geographic boundaries of each Service Region; |
| 6.4.5.21 | Notice of all appropriate mailing addresses and telephone numbers to be utilized by Enrollees seeking Information or authorization, including the Contractor’s toll-free telephone line and website address; |
| 6.4.5.22 | A description of Utilization Management policies and procedures used by the Contractor; |
| 6.4.5.23 | A description of Enrollee rights and responsibilities as described in Section 6.5; |
| 6.4.5.24 | The policies and procedures for Disenrollment, including when Disenrollment may be requested without Enrollee consent by the Contractor and Information about Enrollee’s right to request Disenrollment, and including notice of the fact that the Enrollee will lose Access to services under the GHP if the Enrollee chooses to disenroll; |
Page 54 of 301
| 6.4.5.25 | Information on Advance Directives, including the right of Enrollees to file directly with ASES or with the Puerto Rico Office of the Patient Advocate, Complaints concerning Advance Directive requirements listed in Section 7.10 of this Contract; |
| 6.4.5.26 | A statement that additional Information, including the Provider Guidelines (see Section 10.2.1 of the Contract) and Information on the structure and operations of the GHP and Physician Incentive Plans, shall be made available to Enrollees and Potential Enrollees upon request; |
| 6.4.5.27 | Information on the extent to which, and how, after-hours and emergency coverage are provided, including: |
| 6.4.5.27.1 | What constitutes an Emergency Medical Condition and a Psychiatric Emergency; |
| 6.4.5.27.2 | The fact that Prior Authorization is not required for Emergency Services; |
| 6.4.5.27.3 | Notice that: |
| 6.4.5.27.3.1 | Under no circumstances will a Medicaid or CHIP Enrollee be charged a Co-Payment for the treatment of any Emergency Medical Condition or Psychiatric Emergency; |
| 6.4.5.27.3.2 | No Co-Payments shall be charged for Medicaid and CHIP children under eighteen (18) years under any circumstances. |
| 6.4.5.27.3.3 | For Medicaid or CHIP Enrollees, Co-Payments apply to emergency room services outside the Enrollee’s PPN to treat a condition determined by the attending physician at the time of visit to be non-emergency in nature, but by using the GHP Service Line service (see Section 6.8), the Enrollee may avoid a Co-Payment for such services; and |
| 6.4.5.27.3.4 | For Other Eligible Persons, Co-Payments apply to Emergency Services outside the Enrollee’s PPN, but the Enrollee may avoid a Co-Payment by using the GHP Service Line (see Section 6.8). |
| 6.4.5.27.4 | The process and procedures for obtaining Emergency Services, including the use of the 911 telephone systems or its local equivalent; |
Page 55 of 301
| 6.4.5.27.5 | The scope of Post-Stabilization Services offered under the GHP as detailed in Section 7.5.9.4; |
| 6.4.5.27.6 | The locations of emergency rooms and other locations at which Providers and hospitals furnish Emergency Services and Post-Stabilization Services covered under the GHP; and |
| 6.4.5.27.7 | The fact that an Enrollee has a right to use any hospital or other setting for Emergency Services; |
| 6.4.5.28 | An explanation of the Redetermination process, including: |
| 6.4.5.28.1 | Disenrollment as a consequence of a Negative Redetermination Decision; and |
| 6.4.5.28.2 | The Re-Enrollment period that follows a new Certification. |
| 6.4.5.29 | Information on the Contractor’s Grievance Systems policies and procedures, as described in Article 14 of this Contract. This description must include the following: |
| 6.4.5.29.1 | The right to file a Grievance and Appeal with the Contractor; |
| 6.4.5.29.2 | The requirements and timeframes for filing a Grievance or Appeal with the Contractor; |
| 6.4.5.29.3 | The availability of assistance in filing a Grievance or Appeal with the Contractor; |
| 6.4.5.29.4 | The toll-free numbers that the Enrollee can use to file a Grievance or an Appeal with the Contractor by phone; |
| 6.4.5.29.5 | The right to an Administrative Law Hearing after exhaustion of the Contractor’s Grievance System, the method for obtaining a hearing, and the rules that govern representation at the hearing; |
| 6.4.5.29.6 | Notice that if the Enrollee files an Appeal or a request for an Administrative Law Hearing and requests continuation of services, the Enrollee may be required to pay the cost of services furnished while the Appeal is pending, if the final decision is adverse to the Enrollee; |
| 6.4.5.29.7 | Any Appeal rights that ASES chooses to make available to Providers to challenge the failure of the Contractor to cover a service; |
Page 56 of 301
| 6.4.5.29.8 | Instructions on how an Enrollee can report suspected Fraud, Waste, or Abuse on the part of a Provider, and protections that are available for whistleblowers; and |
| 6.4.5.29.9 | Information on the family planning services provided by the Puerto Rico Department of Health. |
| 6.4.6 | The Enrollee Handbook in both English and Spanish shall be submitted to ASES for review and prior written approval. Submission of the Enrollee Handbook by the Contractor shall be in accordance with the timeframes specified in Attachment 12 to this Contract. |
| 6.4.7 | The Contractor shall be responsible for producing the Enrollee Handbook in both English and Spanish. |
| 6.5 | Enrollee Rights and Responsibilities |
| 6.5.1 | The Contractor shall have written policies and procedures regarding the rights of Enrollees and shall comply with any applicable Federal and Puerto Rico laws and regulations that pertain to Enrollee rights, including those set forth in 42 CFR 438.100, and in the Puerto Rico Patient’s Bill of Rights Act 194 of August 25, 2000; the Puerto Rico Mental Health Law of October 2, 2000, as amended and implemented; and Law 77 of July 24, 2013 which created the Office of the Patient Advocate. These rights shall be included in the Enrollee Handbook. At a minimum, the policies and procedures shall specify the Enrollee’s right to: |
| 6.5.1.1 | Receive information pursuant to 42 CFR 438.10; |
| 6.5.1.2 | Be treated with respect and with due consideration for the Enrollee’s dignity and privacy; |
| 6.5.1.3 | Have all records and medical and personal information remain confidential; |
| 6.5.1.4 | Receive information on available treatment options and alternatives, presented in a manner appropriate to the Enrollee’s condition and ability to understand; |
| 6.5.1.5 | Participate in decisions regarding his or her health care, including the right to refuse treatment; |
| 6.5.1.6 | Be free from any form of restraint or seclusion as a means of coercion, discipline, convenience, or retaliation, as specified in 42 CFR 482.13(e) and other Federal regulations on the use of restraints and seclusion; |
| 6.5.1.7 | Request and receive a copy of his or her Medical Records pursuant to 45 CFR Parts 160 and 164, subparts A and E, and request to amend or correct the record as specified in 45 CFR 164.524 and 164.526; |
Page 57 of 301
| 6.5.1.8 | Choose an Authorized Representative to be involved as appropriate in making care decisions; |
| 6.5.1.9 | Provide informed consent; |
| 6.5.1.10 | Be furnished with health care services in accordance with 42 CFR 438.206 through 438.210; |
| 6.5.1.11 | Freely exercise his or her rights, including those related to filing a Grievance or Appeal, and that the exercise of these rights will not adversely affect the way the Enrollee is treated; |
| 6.5.1.12 | Receive Information about Covered Services and how to access Covered Services and Network Providers; |
| 6.5.1.13 | Be free from harassment by the Contractor or its Network Providers with respect to contractual disputes between the Contractor and its Providers; |
| 6.5.1.14 | Participate in understanding physical and Behavioral Health problems and developing mutually agreed-upon treatment goals; |
| 6.5.1.15 | Not be held liable for the Contractor’s debts in the event of insolvency; not be held liable for the Covered Services provided to the Enrollee for which ASES does not pay the Contractor; not be held liable for Covered Services provided to the Enrollee for which ASES or the Contractor’s Plan does not pay the Provider that furnishes the services; and not be held liable for payments of Covered Services furnished under a contract, Referral, or other arrangement to the extent that those payments are in excess of the amount the Enrollee would owe if the Contractor provided the services directly; and |
| 6.5.1.16 | Only be responsible for cost-sharing in accordance with 42 CFR 447.50 through 42 CFR 447.60 and as permitted by the Puerto Rico Medicaid and CHIP State Plans and Puerto Rico law as applicable to the Enrollee. |
| 6.6 | Provider Directory |
| 6.6.1 | The Contractor shall develop, maintain, and mail to all new Enrollees a Provider Directory that includes Information on both physical and Behavioral Health Providers under the GHP. The Contractor shall distribute the Provider Directory by sending it via surface mail, within five (5) Calendar Days of sending the notice of Enrollment referenced in Section 5.2.5.3. |
Page 58 of 301
| 6.6.2 | The Contractor shall re-print the Provider Directory and distribute it to all Enrollees at least once per year and additionally upon Enrollee request. |
| 6.6.3 | The Contractor shall make the Provider Directory available on its website. |
| 6.6.4 | The Provider Directory shall include the names, locations, office hours, and telephone numbers of current Network Providers. This includes, at a minimum, Information sorted by Service Region on PCPs, specialists, dentists, FQHCs and RHCs, Behavioral Health Providers in each Service Region, hospitals, including locations of emergency settings and Post-Stabilization Services, with the name, location, hours of operation, and telephone number of each facility/setting. The Provider Directory shall also identify all Network Providers that are not accepting new patients. |
| 6.6.5 | The Provider Directory shall include all Network Providers in the Service Region grouped by PMG. |
| 6.6.6 | The Provider Directory must be indexed alphabetically and by specialty. |
| 6.6.7 | The Contractor shall submit the Provider Directory to ASES for review and prior written approval in the timeframe specified in Attachment 12 to this Contract. |
| 6.6.8 | The Contractor shall update and amend the Provider Directory on its website within three (3) Calendar Days of any changes as well as produce and distribute annual updates to all Enrollees. The Contractor shall maintain on its website an updated Provider Directory that includes all identified Information above and that is searchable by Provider type, distance from Enrollee’s address, and/or whether the Network Provider is accepting new patients. Information on how to access this Information shall be clearly stated in both the Enrollee and Provider sections of the website. |
| 6.6.9 | On a monthly basis, the Contractor shall submit to ASES any changes and edits to the Provider Directory. Such changes shall be submitted electronically in the format specified by ASES. |
| 6.7 | Enrollee Identification (ID) Card |
| 6.7.1 | The Contractor shall furnish to all new Enrollees an Enrollee ID Card made of durable plastic material. The card shall be mailed to the Enrollee via surface mail within five (5) Business Days of sending the notice of Enrollment referenced in Section 5.2.5.3. |
| 6.7.2 | The Enrollee ID Card must, at a minimum, include the following information: |
| 6.7.2.1 | The “GHP” logo; |
Page 59 of 301
| 6.7.2.2 | The Enrollee’s name; |
| 6.7.2.3 | The Enrollee’s date of birth; |
| 6.7.2.4 | A designation of the Enrollee as a Medicaid Eligible, a CHIP Eligible, or an Other Eligible Person; |
| 6.7.2.5 | The Enrollee’s Medicaid or CHIP identification number, if applicable; |
| 6.7.2.6 | The Enrollee’s Plan group number, when applicable; |
| 6.7.2.7 | If the Enrollee is eligible for the GHP as a Dependent, the Enrollee’s relationship to the principal Enrollee; |
| 6.7.2.8 | The Effective Date of Enrollment in the GHP; |
| 6.7.2.9 | The Master Patient Identifier; |
| 6.7.2.10 | The applicable Co-Payment levels for various services outside the Enrollee’s PPN and the assurance that no Co-Payment will be charged for a Medicaid Eligible Person and for CHIP children under eighteen (18) years under any circumstances; |
| 6.7.2.11 | The PCP’s and the PMG’s names; |
| 6.7.2.12 | The name and telephone number(s) of the Contractor; |
| 6.7.2.13 | The twenty-four (24) hour, seven (7) day a Week toll-free GHP Service Line Medical Advice Service phone number; |
| 6.7.2.14 | A notice that the Enrollee ID Card may under no circumstances be used by a person other than the identified Enrollee; and |
| 6.7.2.15 | Instructions to obtain Emergency Services. |
| 6.7.3 | The Contractor shall reissue the Enrollee ID Card in the following situations and timeframes: |
| 6.7.3.1 | Within ten (10) Calendar Days of notice if an Enrollee reports a lost, stolen, or damaged ID Card and requests a replacement; |
| 6.7.3.2 | Within ten (10) Calendar Days of notice if an Enrollee reports a name change; |
| 6.7.3.3 | Within twenty (20) Calendar Days of the effective date of a change of PMG or change or addition of a PCP, as provided in Section 5.4. |
Page 60 of 301
| 6.7.4 | The Contractor may charge a fee of five dollars ($5.00) to replace lost, damaged, or stolen Enrollee ID Cards; provided, however, that the Contractor may not charge a replacement fee because of a name change or change of PMG or PCP, and that the Contractor may not charge a replacement fee in any circumstance for Medicaid and CHIP Eligibles. |
| 6.7.5 | The Contractor shall submit a front and back sample Enrollee ID Card to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 6.7.6 | The Contractor must require an Enrollee to surrender his or her ID Card in each of the following events: |
| 6.7.6.1 | The Enrollee disenrolls from the GHP; |
| 6.7.6.2 | The Enrollee requests a change to his or her PCP or PMG, and is therefore issued a new Enrollee ID Card; or |
| 6.7.6.3 | The Enrollee requests a new ID Card because his or her existing card is damaged. |
| 6.8 | GHP Service Line (Toll Free Telephone Service) |
| 6.8.1 | The Contractor shall operate a toll-free telephone number, “GHP Service Line” equipped with caller identification and automatic call distribution equipment capable of handling the high expected volume of calls. The GHP Service Line shall have two components: |
| 6.8.1.1 | An Information Service to respond to questions, concerns, inquiries, and Complaints regarding the GHP from the Enrollee, Enrollee’s family, or Enrollee’s Authorized Representative; and |
| 6.8.1.2 | A Medical Advice Service to advise Enrollees about how to resolve non-emergency medical or Behavioral Health concerns. |
| 6.8.2 | The Contractor shall establish, operate, monitor, and support an automated call distribution system for the GHP Service Line that supports, at a minimum: |
| 6.8.2.1 | Capacity to handle the high call volume; |
| 6.8.2.2 | A daily analysis of the quantity, length, and types of calls received; |
| 6.8.2.3 | A daily analysis of the amount of time it takes to answer the call, including Blocked and Abandoned Calls; |
| 6.8.2.4 | The ability to measure average waiting time; and |
Page 61 of 301
| 6.8.2.5 | The ability to monitor calls from a remote location by a Third Party, such as ASES. |
| 6.8.3 | Hours of Operation. Each service shall be made available as: |
| 6.8.3.1 | The Information Service shall be fully staffed between the hours of 7:00 a.m. and 7:00 p.m. (Atlantic Time). Monday through Friday, excluding Puerto Rico holidays. The Contractor shall have an automated system available between the hours of 7:00 p.m. and 7:00 a.m. (Atlantic Time) Monday through Friday and during all hours on weekends and holidays. This automated system must provide callers with operating instructions on what to do in case of an emergency and shall include, at a minimum, a voice mailbox for callers to leave messages. The Contractor shall ensure that the voice mailbox has the required capacity to receive all messages. A Contractor’s representative shall reply to one hundred percent (100%) of messages by the next Business Day. |
| 6.8.3.2 | The Medical Advice Service shall be fully staffed and available to Enrollees twenty-four (24) hours per day, seven (7) days per Week. |
| 6.8.4 | Staffing |
| 6.8.4.1 | The Contractor shall be responsible for the required staffing of the GHP Service Line with individuals who are able to communicate effectively with GHP Enrollees. |
| 6.8.4.2 | The Contractor shall make key staff responsible for operating the GHP Service Line available to meet with ASES staff on a regular basis, as requested by ASES, to review reports and all other obligations under the Contract relating to GHP Service Line. |
| 6.8.4.3 | All staff shall be hired and must complete a training program at least fifteen (15) Calendar Days before the staff provides GHP Service Line services. Such training program shall include, but will not be limited to, systems, policies and procedures, and telephone scripts. |
| 6.8.4.4 | For the Information Service, the Contractor shall ensure that Call Center attendants receive the necessary training to respond to Enrollee questions, concerns, inquiries, and Complaints from the Enrollee or the Enrollee’s family relating to this Contract regarding topics, including but not limited to Covered Services (both physical and Behavioral Health), Grievances and Appeals, the Provider Network, and Enrollment and Disenrollment. |
| 6.8.4.5 | For the Medical Advice Service, the Contractor shall ensure that Call Center attendants are registered nurses with the necessary training to advise Enrollees about appropriate steps they should take to resolve a physical or Behavioral Health complaint or concern. |
Page 62 of 301
| 6.8.4.6 | The Contractor shall ensure that GHP Service Line Call Center staff is trained to identify Behavioral Health concerns and, where appropriate, to transfer Enrollee callers to the appropriate Call Center representative for assistance. |
| 6.8.4.7 | The Contractor shall ensure that GHP Service Line Call Center staff is trained to identify situations in which an Enrollee may need services that are offered through the Department of Health rather than through the GHP, and GHP Service Line staff shall provide the Enrollee with Information on where to access these services. |
| 6.8.4.8 | The Contractor shall ensure that GHP Service Line Call Center staff is trained to provide to Medicaid and CHIP Eligible Enrollees Information on how to access local NEMT resources to enable an Enrollee without available transportation to receive Medically Necessary Services. |
| 6.8.4.9 | The Contractor shall ensure that GHP Service Line Call Center staff are trained to process and fulfill requests by Enrollees and Potential Enrollees to receive, by surface mail, the Enrollee Handbook, the Provider Directory, or the Provider Guidelines. The Contractor shall fulfill such requests by mailing the requested document within five (5) Business Days of the request. |
| 6.8.5 | The Contractor may provide the Information Service and the Medical Advice Service as separate phone lines with a “Warm Transfer” capability, or as separate dialing options within one (1) phone line. |
| 6.8.6 | The Contractor shall have the capability of making out-bound calls. |
| 6.8.7 | GHP Service Line shall be equipped to handle calls in Spanish and English, as well as, through a Telecommunication Device for the Deaf (TDD) for calls from Enrollees who are hearing-impaired. For callers who speak neither English nor Spanish, the Contractor shall provide interpreter services free of charge to Enrollees. The Contractor shall not permit Enrollees’ family members, especially minor children, or friends, to provide oral interpreter services, unless specifically requested by the Enrollee. |
| 6.8.8 | All calls shall be recorded, identifying the date and time, the type of call, the reason for the call, and the resolution of the call. |
| 6.8.9 | The Contractor shall generate a call identification number for each phone call made by an Enrollee to the Medical Advice Service. Enrollees who use this service to seek advice on their health condition before visiting the emergency room will not be responsible for any Co-Payment otherwise imposed for emergency room visits (as provided under Section 7.11.4) outside the Enrollee’s PPN, provided that the Enrollee presents his or her GHP Service Line call identification number at the emergency room. Under no circumstance will a Co-Payment be imposed on a Medicaid or CHIP Eligible Enrollee for treatment of an Emergency Medical Condition or Psychiatric Emergency (regardless of whether the Enrollee uses the Medical Advice Service). The Medical Advice Service does not apply to services obtained outside of Puerto Rico; however, Enrollees should be able to access both the Medical Advice Service and the Information Service lines from the US. |
Page 63 of 301
| 6.8.10 | The Contractor shall develop GHP Service Line policies and procedures, including staffing, training, hours of operation, Access and response standards, transfers/Referrals, monitoring of calls via recording and other means, and compliance with other performance standards to be prior approved in writing by ASES. |
| 6.8.11 | The Contractor shall develop GHP Service Line quality criteria and protocols. These protocols shall, at a minimum: |
| 6.8.11.1 | Measure and monitor the accuracy of responses and phone etiquette in GHP Service Line (including through recording phone calls) and take corrective action as necessary to ensure the accuracy of responses and appropriate phone etiquette by staff; |
| 6.8.11.2 | Provide for quality calibration sessions between the Contractor’s staff and ASES; |
| 6.8.11.3 | Require that, on a monthly basis, the average speed of answer is at least eighty percent (80%) of calls answered within thirty (30) seconds; |
| 6.8.11.4 | Require that, on a monthly basis, the Blocked Call rate does not exceed three percent (3%); and |
| 6.8.11.5 | Require that, on a monthly basis, the rate of Abandoned Calls does not exceed five percent (5%). |
| 6.8.12 | The above standards serve as minimum requirements for each GHP Service Line service. The Contractor may elect to establish more rigorous performance standards. The Contractor may elect to establish different quality criteria for the Medical Advice Service than for the Information Service; provided, however, the standards governing the Medical Advice Service are stricter than the standards for the Information Service. |
Page 64 of 301
| 6.8.13 | The Contractor must develop and implement a GHP Service Line Outreach Program to educate Enrollees about the GHP Service Line service and to encourage its use. The Outreach program shall include, at a minimum, the following components: |
| 6.8.13.1 | A section on GHP Service Line in the Enrollee Handbook; |
| 6.8.13.2 | Contact information for GHP Service Line on the Enrollee ID Card and on the Contractor’s website; and |
| 6.8.13.3 | Informational flyers on the GHP Service Line to be placed in the offices of the Contractor and the Network Providers. |
| 6.8.14 | All documents and communication materials included in this Outreach program must explain that (i) by using the Medical Advice Service before visiting the emergency room, and presenting their call identification number at the emergency room, Enrollees can avoid any emergency room Co-Payments otherwise applicable under Section 7.11.4 of this Contract for services outside the PPN; and (ii) in no event will Co-Payments be imposed for services to treat an Emergency Medical Condition or Psychiatric Emergency for a Medicaid or CHIP Eligibles. All written materials included in the Outreach Program must be written at a fourth (4th) grade reading level and must be available in Spanish and English. |
| 6.8.15 | The Contractor shall prepare scripts addressing the questions expected to arise most often for both the Information Service and the Medical Advice Service. The Contractor shall submit these scripts to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. It is the responsibility of the Contractor to maintain and update these scripts and to ensure that they are developed at the fourth (4th) grade reading level. The Contractor shall submit revisions to the script to ASES for written approval prior to use. |
| 6.8.16 | The Contractor shall submit the following written materials referred to in this Section 6.8 to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract: |
| 6.8.16.1 | GHP Service Line policies and procedures; |
| 6.8.16.2 | GHP Service Line quality criteria and protocols; |
| 6.8.16.3 | GHP Service Line Outreach Program; and |
| 6.8.16.4 | Scripts and training materials for GHP Service Line Call Center employees. |
Page 65 of 301
| 6.9 | Internet Presence / Website |
| 6.9.1 | The Contractor shall provide on its website general and up-to-date information about the GHP and about the Contractor’s Plan, including the Provider Network, customer services, GHP Service Line, and its Grievance System and Complaint Process. The Enrollee Handbook and the Provider Directory shall be available on the website. All information must be written at a fourth (4th) and must be available in Spanish and English. |
| 6.9.2 | The Contractor shall maintain an Enrollee portal that allows Enrollees to access a searchable Provider Directory that shall be updated within three (3) Business Days of any change to the Provider Network. |
| 6.9.3 | The website must have the capability for Enrollees to submit questions and comments to the Contractor and receive responses. The Contractor shall reply to Enrollee questions within two (2) Business Days. |
| 6.9.4 | The website must comply with the Marketing policies and procedures and with requirements for written materials described in Sections 6.2 and 6.3 of this Contract and must be consistent with applicable Puerto Rico and Federal laws. |
| 6.9.5 | The Contractor shall submit website screenshots to ASES for review and approval of information on the website relating to the GHP Program according to the timeframe specified in Attachment 12 to this Contract. |
| 6.9.6 | The Contractor’s website shall provide secured online access to the Enrollee’s historical and current information. |
| 6.9.7 | The Contractor’s website shall prominently feature a link to the ASES website, www.ases.pr.gov. |
| 6.10 | Cultural Competency |
| 6.10.1 | In accordance with 42 CFR 438.206, the Contractor shall have a comprehensive written Cultural Competency plan describing how the Contractor will ensure that services are provided in a culturally competent manner to all Enrollees. The Cultural Competency plan must describe how the Providers, individuals, and systems within the Contractor’s Plan will effectively provide services to people of all cultures, races, ethnic backgrounds, and religions in a manner that recognizes values, affirms, and respects the worth of the individual Enrollees and protects and preserves the dignity of each individual. |
| 6.10.2 | The Contractor shall submit the Cultural Competency plan to ASES for review and approval according to the timeframe specified in Attachment 12 to this Contract. |
Page 66 of 301
| 6.10.3 | The Contractor may distribute a summary of the Cultural Competency plan, rather than the entire document, to Providers if the summary includes Information on how the Provider may access the full Cultural Competency plan on the Contractor’s website. This summary shall also detail how the Provider can request a hard copy from the Contractor at no charge to the Provider. |
| 6.11 | Interpreter Services |
| 6.11.1 | The Contractor shall provide oral interpreter services to any Enrollee or Potential Enrollee who speaks any language other than English or Spanish as his or her primary language, regardless of whether the Enrollee or Potential Enrollee speaks a language that meets the threshold of a Prevalent Non-English Language. The Contractor is required to notify its Enrollees of the availability of oral interpretation services and to inform them of how to access oral interpretation services. There shall be no charge to an Enrollee or Potential Enrollee for interpreter services. |
| 6.12 | Enrollment Outreach |
| 6.12.1 | The Contractor shall participate in any Enrollment Outreach activities as prescribed by ASES or the Puerto Rico Medicaid Program. |
| 6.13 | Special Enrollee Information Requirements for Dual Eligible Beneficiaries |
| 6.13.1 | The Contractor shall inform a Potential Enrollee who is a Dual Eligible Beneficiary: |
| 6.13.1.1 | That the Dual Eligible Beneficiary is eligible for services under the GHP with the limits stated in Section 7.12 of this Contract; |
| 6.13.1.2 | That the GHP Plan will cover Medicare Part B Deductibles and co-insurance, but not Medicare Part A Deductibles; |
| 6.13.1.3 | That the Dual Eligible Beneficiary may not be simultaneously enrolled in the GHP and in a Medicare Platino plan, for the reason that the Platino plan already includes GHP Benefits; and |
| 6.13.1.4 | That as an Enrollee in the Contractor’s Plan, the Dual Eligible Beneficiary may access Covered Services only through the PMG, not through the Medicare Provider List. |
| 6.14 | Marketing |
| 6.14.1 | Prohibited Activities. The Contractor is prohibited from engaging in the following activities: |
| 6.14.1.1 | Directly or indirectly engaging in door-to-door, telephone, or other Cold-Call Marketing activities aimed at Potential Enrollees; |
Page 67 of 301
| 6.14.1.2 | Offering any favors, inducements or gifts, promotions, or other insurance products that are designed to induce Enrollment in the Contractor’s Plan; |
| 6.14.1.3 | Distributing plans and materials that contain statements that ASES determines are inaccurate, false, or misleading. Statements considered false or misleading include, but are not limited to, any assertion or statement (whether written or oral) that the Contractor’s plan is endorsed by the Federal Government or Commonwealth, or similar entity; and |
| 6.14.1.4 | Distributing materials that, according to ASES, mislead or falsely describe the Contractor’s Provider Network, the participation or availability of Network Providers, the qualifications and skills of Network Providers (including their bilingual skills); or the hours and location of network services. |
| 6.14.1.5 | Seeking to influence Enrollment in conjunction with the sale or offering of any private insurance. |
| 6.14.2 | Allowable Activities. The Contractor shall be permitted to perform the following Marketing activities: |
| 6.14.2.1 | Distribute general information through mass media (i.e. newspapers, magazines and other periodicals, radio, television, the Internet, public transportation advertising, and other media outlets); |
| 6.14.2.2 | Make telephone calls, mailings and home visits only to Enrollees currently enrolled in the Contractor’s plan, for the sole purpose of educating them about services offered by or available through the Contractor; |
| 6.14.2.3 | Distribute brochures and display posters at Provider offices that inform patients that the Provider is part of the GHP Provider Network; and |
| 6.14.2.4 | Attend activities that benefit the entire community, such as health fairs or other health education and promotional activities. |
| 6.14.3 | If the Contractor performs an allowable activity, the Contractor must conduct that activity in one (1) or all Service Regions covered by this Contract. |
| 6.14.4 | All materials shall be in compliance with the informational requirements in 42 CFR 438.10. |
| 6.14.5 | ASES Approval of Marketing Materials |
Page 68 of 301
| 6.14.5.1 | The Contractor shall submit a detailed description of its Marketing plan and copies of all Marketing Materials (written and oral) that it or its Subcontractors plan to distribute to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. This requirement includes, but is not limited to posters, brochures, websites, and any materials that contain statements regarding the Benefit package and Provider Network-related materials. Neither the Contractor nor its Subcontractors shall distribute any Marketing Materials without prior written approval from ASES. |
| 6.14.5.2 | The Contractor shall submit any changes to previously approved Marketing Materials and receive written approval from ASES of the changes before distribution. |
| 6.14.5.3 | The Contractor must comply with ASES’ Normative Letter 13-1212 and 13-1216 as amended January 21, 2014, related to the review and approval of the Contractors Marketing Materials included in Attachment 13 of this Contract. |
| 6.14.6 | Provider Marketing Materials |
| 6.14.6.1 | The Contractor is responsible for ensuring that not only its Marketing activities, but also the Marketing activities of its Subcontractors and Providers, meet the requirements of this Section 6.14. |
| 6.14.6.2 | The Contractor shall collect from its Providers any Marketing Materials they intend to distribute and submit these to ASES for review and written approval prior to distribution. |
| 6.14.6.3 | The Contractor shall provide for equitable distribution of all Marketing Materials without bias toward or against any group. |
ARTICLE 7 COVERED SERVICES AND BENEFITS
| 7.1 | Requirement to Provide Covered Services |
| 7.1.1 | The Contractor shall at a minimum provide Medically Necessary Covered Services to Enrollees as of the Effective Date of Enrollment (including the retroactive period specified in Section 5.1.3.1) pursuant to the program requirements of the GHP, and the Puerto Rico Medicaid State Plan and CHIP Plan. The Contractor shall not impose any other exclusions, limitations, or restrictions on any Covered Service, and shall not arbitrarily deny or reduce the amount, duration, or scope of a Covered Service solely because of the diagnosis, type of illness, or condition. |
Page 69 of 301
| 7.1.1.1 | In accordance with Section 2702 of the PPACA, the Contractor must have mechanisms in place to prevent payment for the following Provider preventable conditions and must require all providers to report on such Provider preventable conditions associated with Claims for payment or Enrollee treatments for which payment would otherwise be made: |
| 7.1.1.1.1 | All hospital acquired conditions as identified by Medicare other than deep vein thrombosis (DVT)/Pulmonary Embolism (PE) following total knee replacement or hip replacement surgery in pediatric and obstetric patients for inpatient hospital services; and |
| 7.1.1.1.2 | Any incorrect surgical or other invasive procedure performed on a patient; any surgical or other invasive procedure performed on the incorrect body part; or any surgical or other invasive procedure performed on the incorrect patient for inpatient and non-institutional services. |
| 7.1.2 | The Contractor shall not deny Covered Services based on pre-existing conditions, the individual’s genetic Information, or waiting periods. |
| 7.1.3 | The Contractor shall not be required to provide a Covered Service to a person who is not an Eligible Person. |
| 7.1.4 | The Contractor shall not be required to pay for a Covered Service if: |
| 7.1.4.1 | The Enrollee paid the Provider for the service. This rule does not apply in circumstances where a Medicaid or CHIP Eligible Enrollee incurs out-of-pocket expenses for Emergency Services provided in the US. In such a case, the expenses will be reimbursed under the GHP; or |
| 7.1.4.2 | The service was provided by a person or entity that does not meet the definition of a Network Provider (with the exception of Medical Emergencies and cases where the service was Prior Authorized by the Contractor). |
| 7.2 | Medical Necessity |
| 7.2.1 | Based on generally accepted medical practices specific to the medical or Behavioral Health condition of the Enrollee at the time of treatment, Medically Necessary Services are those that relate to (i) the prevention, diagnosis, and treatment of health impairments; (ii) the ability to achieve age-appropriate growth and development; or (iii) the ability to attain, maintain, or regain functional capacity. The scope of Medically Necessary Services must not be any more restrictive than that of Puerto Rico’s Medicaid program. Additionally, Medically Necessary services must be: |
Page 70 of 301
| 7.2.1.1 | Appropriate and consistent with the diagnosis of the treating Provider and the omission of which could adversely affect the eligible Enrollee’s medical condition; |
| 7.2.1.2 | Compatible with the standards of acceptable medical practice in the community; |
| 7.2.1.3 | Provided in a safe, appropriate, and cost-effective setting given the nature of the diagnosis and the severity of the symptoms; |
| 7.2.1.4 | Not provided solely for the convenience of the Enrollee or the convenience of the Provider or hospital; and |
| 7.2.1.5 | Not primarily custodial care (for example, foster care). |
| 7.2.2 | In order for a service to be Medically Necessary, there must be no other effective and more conservative or substantially less costly treatment, service, or setting available. |
| 7.3 | Experimental or Cosmetic Procedures |
| 7.3.1 | In no instance shall the Contractor cover experimental or cosmetic procedures, except as required by the Puerto Rico Patient’s Bill of Rights Act or any other Federal or Puerto Rico law or regulation. Breast reconstruction after a mastectomy and surgical procedures that are determined to be Medically Necessary to treat morbid obesity shall not be regarded as cosmetic procedures. |
| 7.4 | Covered Services and Administrative Functions |
| 7.4.1 | Benefits under the GHP are comprised of four categories: (i) Basic and Behavioral Health Coverage, (ii) dental services, (iii) Special Coverage, and (iv) Administrative Functions. The scope of these items is covered in Sections 7.5 – 7.8, in the order listed. |
| 7.5 | Basic and Behavioral Health Coverage |
| 7.5.1 | Basic and Behavioral Health Coverage is available to all GHP Enrollees, except as provided in the table below. Basic Coverage includes the following categories: |
|
BASIC COVERAGE SERVICES
|
GHP ELIGIBILITY
GROUPS COVERED
|
|
Preventive Services
|
All
|
|
Diagnostic Test Services
|
All
|
|
Outpatient Rehabilitation Services
|
All
|
Page 71 of 301
|
BASIC COVERAGE SERVICES
|
GHP ELIGIBILITY
GROUPS COVERED
|
|
Medical and Surgical Services
|
All
|
|
Emergency Transportation Services
|
All (Services outside Puerto Rico available only for Medicaid and CHIP Eligibles)
|
|
Maternity and Pre-Natal Services
|
All
|
|
Emergency Services
|
All (Services outside Puerto Rico available only for Medicaid and CHIP Eligibles)
|
|
Hospitalization Services
|
All
|
|
Behavioral Health Services
|
All
|
|
Pharmacy Services
|
All (Note: Claims processing and adjudication Services provided by PBM; not covered under this Contract.)
|
| 7.5.2 | Exclusions from Basic Coverage |
| 7.5.2.1 | The following services are excluded from all Basic Coverage. In addition, exclusions specific to each category of Covered Services are noted in Sections 7.5.3 – 7.5.12 below. |
| 7.5.2.1.1 | Expenses for personal comfort materials or services, such as, telephone use, television, or toiletries; |
| 7.5.2.1.2 | Services rendered by close family relatives (parents, children, siblings, grandparents, grandchildren, or spouses); |
| 7.5.2.1.3 | Weight control treatment (obesity or weight gain) for aesthetic reasons. As noted, procedures determined to be Medically Necessary to address morbid obesity shall not be excluded; |
| 7.5.2.1.4 | Sports medicine, music therapy, and natural medicine; |
| 7.5.2.1.5 | Services, diagnostic testing, or treatment ordered or rendered by naturopaths, naturists, or iridologists,; |
| 7.5.2.1.6 | Health Certificates, except as provided in Section 7.5.3.2.10 (Preventive Services); |
| 7.5.2.1.7 | Epidural anesthesia services; |
| 7.5.2.1.8 | Educational tests or services; |
| 7.5.2.1.9 | Peritoneal dialysis or hemodialysis services (covered under Special Coverage, not Basic Coverage); |
Page 72 of 301
| 7.5.2.1.10 | Hospice care for Adults; |
| 7.5.2.1.11 | Services received outside the territorial limits of Puerto Rico, except as provided in Sections 7.5.7.11 (Emergency Transportation) and 7.5.9.3 (Emergency Services); |
| 7.5.2.1.12 | Expenses incurred for the treatment of conditions resulting from services not covered under the GHP (maintenance prescriptions and required clinical laboratories for the continuity of a stable health condition, as well as any emergencies which could alter the effects of the previous procedure, are covered); |
| 7.5.2.1.13 | Judicially ordered evaluations for legal purposes; |
| 7.5.2.1.14 | Travel expenses, even when ordered by the Primary Care Physician; |
| 7.5.2.1.15 | Eyeglasses, contact lenses and hearing aids for adults; |
| 7.5.2.1.16 | Acupuncture services; |
| 7.5.2.1.17 | Sex change procedures; |
| 7.5.2.1.18 | Organ and tissue transplants, except skin, bone and corneal transplants; and |
| 7.5.2.1.19 | Treatment for infertility and/or related to conception by artificial means including tuboplasty, vasovasectomy, and any other procedure to restore the ability to procreate. |
| 7.5.3 | Preventive Services |
| 7.5.3.1 | Healthy Child Care. The Contractor shall provide the following Preventive Services as Covered Services under the Healthy Child Care Program, which serves enrollees under age two (2): |
| 7.5.3.1.1 | One (1) annual comprehensive evaluation by a certified Provider, which complements other services for children and young adults provided pursuant to the periodicity scheme of the American Academy of Pediatrics and Title XIX (EPSDT); and |
| 7.5.3.1.2 | Other services, as needed, during the first two (2) years of the child’s life. |
| 7.5.3.2 | Other Preventive Services. The Contractor shall provide the following Preventive Services as Covered Services for all GHP Enrollees: |
Page 73 of 301
| 7.5.3.2.1 | Vaccines (the vaccines themselves are provided and paid for by the Health Department for the Medicaid and CHIP Eligibles. The vaccine is provided and paid for by the Contractor for the Other Eligible Persons in the GHP). The Contractor shall cover the administration of the vaccines according to the fee schedule established by the Health Department; |
| 7.5.3.2.2 | Eye exam; |
| 7.5.3.2.3 | Hearing exam, including hearing screening for newborns prior to their leaving the hospital nursery; |
| 7.5.3.2.4 | Evaluation and nutritional screening; |
| 7.5.3.2.5 | Medically Necessary laboratory exams and diagnostic tests, appropriate to the Enrollee’s age, sex, and health condition, including, but not limited to: |
| 7.5.3.2.5.1 | Prostate and gynecological cancer screening according to accepted medical practice, including Pap smears (for Enrollees over age eighteen (18)), mammograms (for Enrollees age forty (40) and over), and Prostate-Specific Antigen (PSA) tests when Medically Necessary; and |
| 7.5.3.2.5.2 | Sigmoidoscopy and colonoscopy for colon cancer detection in Adults age fifty (50) years and over, classified in risk groups according to accepted medical practice; |
| 7.5.3.2.6 | Nutritional, oral, and physical health education; |
| 7.5.3.2.7 | Reproductive health counseling and family planning. The Contractor shall cover the following family planning services: |
| 7.5.3.2.7.1 | Counseling; |
| 7.5.3.2.7.2 | Pregnancy testing; |
| 7.5.3.2.7.3 | Diagnosis and treatment of sexually transmitted diseases; |
| 7.5.3.2.7.4 | Infertility assessments; |
| 7.5.3.2.7.5 | At least one of every class of FDA approved contraceptive medication as specified in ASES’s PDL; |
Page 74 of 301
| 7.5.3.2.8 | Syringes for home medicine administration, if deemed Medically Necessary; |
| 7.5.3.2.9 | Annual physical exam and follow-up for diabetic patients according to the Diabetic Patient Treatment Guide and Health Department protocols; and |
| 7.5.3.2.10 | Health Certificates are covered under the GHP, provided that cost sharing and/or deductibles applicable for necessary procedures and laboratory testing related to generating a Health Certificate will be the Enrollee’s responsibility. Such certificates shall include: |
| 7.5.3.2.10.1 | Venereal Disease Research Laboratory (“VDRL”) tests; |
| 7.5.3.2.10.2 | Tuberculosis (“TB”) tests; and |
| 7.5.3.2.10.3 | Any Certification for GHP Enrollees related to eligibility for the Medicaid Program (provided at no charge). |
| 7.5.4 | Diagnostic Test Services |
| 7.5.4.1 | The Contractor shall provide the following diagnostic test services as Covered Services: |
| 7.5.4.1.1 | Diagnostic and testing services for Enrollees under age twenty-one (21) required by EPSDT, as defined in Section 1905(r) of the Social Security Act; |
| 7.5.4.1.2 | Clinical labs, including but not limited to, any laboratory order for disease diagnostic purposes, even if the final diagnosis is a condition or disease whose treatment is not a Covered Service; |
| 7.5.4.1.3 | Hi-tech Labs; |
| 7.5.4.1.4 | X-Rays; |
| 7.5.4.1.5 | Electrocardiograms; |
| 7.5.4.1.6 | Radiation therapy (Prior Authorization required); |
| 7.5.4.1.7 | Pathology; |
| 7.5.4.1.8 | Arterial gases and Pulmonary Function Test; |
| 7.5.4.1.9 | Electroencephalograms; |
Page 75 of 301
| 7.5.4.1.10 | Diagnostic services for Enrollees who present learning disorder symptoms; and |
| 7.5.4.1.11 | Services related to a diagnostic code included in the Diagnostic and Statistical Manual of Mental Disorders (“DSM IV or DSM V”). |
| 7.5.4.2 | The following shall not be considered diagnostic test services covered under the GHP: |
| 7.5.4.2.1 | Polysomnography studies; and |
| 7.5.4.2.2 | Clinical labs processed outside of Puerto Rico. |
| 7.5.5 | Outpatient Rehabilitation Services |
| 7.5.5.1 | The Contractor shall provide the following outpatient rehabilitation services as Covered Services: |
| 7.5.5.1.1 | Medically Necessary outpatient rehabilitation services for Enrollees under age twenty-one (21), as required by EPSDT, Section 1905(r) of the Social Security Act; |
| 7.5.5.1.2 | Physical therapy (limited to a maximum of fifteen (15) treatments per Enrollee condition per year, unless Prior Authorization of an additional fifteen (15) treatments is indicated by an orthopedist or physiatrist or chiropractor); |
| 7.5.5.1.3 | Occupational therapy, without limitations; and |
| 7.5.5.1.4 | Speech therapy, without limitations. |
| 7.5.6 | Medical and Surgical Services |
| 7.5.6.1 | The Contractor shall provide the following medical and surgical services as Covered Services: |
| 7.5.6.1.1 | Early and Periodic Screening, Diagnostic and Treatment (“EPSDT”) services, as defined in Section 1905(r) of the Social Security Act; |
| 7.5.6.1.2 | Primary Care Physician visits, including nursing services; |
| 7.5.6.1.3 | Specialist treatment, once referred by the selected PCP if outside of the Enrollee’s PPN; |
| 7.5.6.1.4 | Sub-specialist treatment, once referred by the selected PCP if outside of the Enrollee’s PPN; |
Page 76 of 301
| 7.5.6.1.5 | Physician home visits when Medically Necessary; |
| 7.5.6.1.6 | Respiratory therapy, without limitations; |
| 7.5.6.1.7 | Anesthesia services (except for epidural anesthesia); |
| 7.5.6.1.8 | Radiology services; |
| 7.5.6.1.9 | Pathology services; |
| 7.5.6.1.10 | Surgery; |
| 7.5.6.1.11 | Outpatient surgery facility services; |
| 7.5.6.1.12 | Nursing services; |
| 7.5.6.1.13 | Voluntary sterilization of men and women of legal age and sound mind, provided that they have been previously informed about the medical procedure’s implications, and that there is evidence of Enrollee’s written consent by completing the Sterilization Consent Form included as Attachment 22 of the Contract; |
| 7.5.6.1.14 | Prosthetics, including the supply of all extremities of the human body including therapeutic ocular prosthetics, segmental instrument tray, and spine fusion in scoliosis and vertebral surgery; |
| 7.5.6.1.15 | Ostomy equipment for outpatient-level ostomized patients; |
| 7.5.6.1.16 | Transfusion of blood and blood plasma services, without limitations, including the following: |
| 7.5.6.1.16.1 | Authologal and irradiated blood; |
| 7.5.6.1.16.2 | Monoclonal factor IX with the Referral of a certified hematologist; |
| 7.5.6.1.16.3 | Intermediate purity concentrated ant hemophilic factor (Factor VIII); |
| 7.5.6.1.16.4 | Monoclonal type antihemophilic factor with a certified hematologist’s authorization; and |
| 7.5.6.1.16.5 | Activated protrombine complex (Autoflex and Feiba) with a certified hematologist’s authorization; and |
Page 77 of 301
| 7.5.6.1.17 | Services to patients with Level 1 or Level 2 of chronic renal disease (Levels 3 to 5 are included in Special Coverage). |
| 7.5.6.1.17.1 | Chronic renal disease Levels 1 and 2 are defined as follows: |
| 7.5.6.1.17.1.1 | Level 1: GFR (Glomerular Filtration – ml/min. per 1.73m² per corporal area surface) over 90; slight damage when protein is present in the urine. |
| 7.5.6.1.17.1.2 | Level 2: GFR between 60 and 89, a slight decrease in kidney function. |
| 7.5.6.1.17.2 | When GFR decreases to under 60 ml/min per 1.73 m², the Enrollee must be referred to a nephrologist for proper management. The Enrollee will be registered for Special Coverage. |
| 7.5.6.2 | While cosmetic procedures shall be excluded from Covered Services, breast reconstruction after a mastectomy and surgical procedures Medically Necessary to treat morbid obesity shall not be considered to be cosmetic procedures. |
| 7.5.6.3 | DME’s are covered on a case-by-case basis with Prior Authorization as Medically Necessary. Mechanical respirators and ventilators with oxygen supplies are covered without limits as required by local law to Enrollees under age twenty-one (21).). |
| 7.5.6.4 | Abortions are covered in the following instances: (i) life of the mother would be in danger if the fetus is carried to term; (ii) when the pregnancy is a result of rape or incest; and (iii) severe and long lasting damage would be caused to the mother if the pregnancy is carried to term, as certified by a physician.. |
| 7.5.7 | Emergency Transportation Services |
| 7.5.7.1 | The Contractor shall provide Emergency Transportation Services, including but not limited to, maritime and ground transportation, in emergency situations as Covered Services. |
| 7.5.7.2 | Emergency transportation services shall be available twenty-four (24) hours a day, seven (7) days per Week throughout Puerto Rico. |
| 7.5.7.3 | Emergency transportation services do not require Prior Authorization. |
Page 78 of 301
| 7.5.7.4 | The Contractor shall ensure that adequate emergency transportation is available to transport any Enrollees experiencing an Emergency Medical Conditions or a Psychiatric Emergency, or whose conditions require emergency transportation because of their geographical location. |
| 7.5.7.5 | The Contractor may not impose limits on what constitutes an Emergency Medical Condition or a Psychiatric Emergency on the basis of lists of diagnoses or symptoms. |
| 7.5.7.6 | Aerial emergency transportation services are provided and paid for by ASES under a separate contract. The Contractor shall coordinate the provision of aerial emergency transportation on behalf of its Enrollees when Medically Necessary utilizing the Provider designated by ASES. |
| 7.5.7.7 | The Contractor shall bear the expenses of providing emergency transportation and shall adhere to Puerto Rico laws and regulations concerning emergency transportation, including applicable fees as established by the Public Service Commission of the Commonwealth of Puerto Rico (CSP for its acronym in Spanish). |
| 7.5.7.8 | The Contractor shall provide Category II and Category III Ambulance Services pursuant to Regulation No. 6737 of the Public Service Commission. |
| 7.5.7.8.1 | Category II Ambulances are Ambulances utilized for the transportation of ill, injured, hurt, and disabled patients equipped with the specifications set by the Department of Heath of Puerto Rico. Fees paid for Type III ambulances are set by Provision 57.37 of the Public Service Commission. |
| 7.5.7.8.2 | Category III Ambulances must comply with all the requirements of Category II Ambulances, have advanced stabilization equipment and are specially designed and equipped as established from time to time by the Ambulance Certification Office of the Department of Health of Puerto Rico. |
| 7.5.7.9 | The Contractor may not retroactively deny a Claim for emergency transportation services because the Enrollee’s condition, which at the time of service appeared to be an Emergency Medical Condition or a Psychiatric Emergency under the prudent layperson standard, was ultimately determined to be a non-emergency. |
| 7.5.7.10 | In any case in which an Enrollee is transported by ambulance to a facility that is not a Network Provider, and, after being stabilized, is transported by ambulance to a facility that is a Network Provider, all emergency transportation costs, provided that they are justified by prudent layperson standards, will be borne by the Contractor. |
Page 79 of 301
| 7.5.7.11 | The Contractor shall be responsible for timely payment for emergency transportation services in the US for Enrollees who are Medicaid or CHIP Eligibles, if the emergency transportation is associated with an Emergency Service in the US covered under Section 7.5.9.3.1.2 of this Contract. If, in an extenuating circumstance, a Medicaid or CHIP Eligible Enrollee incurs out-of-pocket expenses for emergency transportation services provided in the US, the Contractor shall reimburse the Enrollee for such expenses in a timely manner, and the reimbursement shall be considered a Covered Service. |
| 7.5.7.12 | Emergency transportation services will be subject to periodic reviews and/or audits by applicable governmental agencies and ASES to ensure quality of services. |
| 7.5.8 | Maternity and Pre-Natal Services |
| 7.5.8.1 | The Contractor shall provide the following maternity and pre-natal services as Covered Services: |
| 7.5.8.1.1 | Pregnancy testing; |
| 7.5.8.1.2 | Medical services, during pregnancy and post-partum; |
| 7.5.8.1.3 | Physician and nurse obstetrical services during vaginal and caesarean section deliveries, and services to address any complication that arises during the delivery; |
| 7.5.8.1.4 | Treatment of conditions attributable to the pregnancy or delivery, when medically recommended; |
| 7.5.8.1.5 | Hospitalization for a period of at least forty-eight (48) hours in cases of vaginal delivery, and at least ninety-six hours (96) in cases of caesarean section; |
| 7.5.8.1.6 | Anesthesia, excluding epidural; |
| 7.5.8.1.7 | Incubator use, without limitations; |
| 7.5.8.1.8 | Fetal monitoring services, during hospitalization only; |
| 7.5.8.1.9 | Nursery room routine care for newborns; |
| 7.5.8.1.10 | Circumcision and dilatation services for newborns; |
Page 80 of 301
| 7.5.8.1.11 | Transportation of newborns to tertiary facilities when necessary; |
| 7.5.8.1.12 | Pediatrician assistance during delivery; and |
| 7.5.8.1.13 | Delivery services provided in free-standing birth centers. |
| 7.5.8.2 | The following are excluded from maternity and pre-natal Covered Services: |
| 7.5.8.2.1 | Outpatient use of fetal monitor; |
| 7.5.8.2.2 | Treatment services for infertility and/or related to conception by artificial means; |
| 7.5.8.2.3 | Services, treatments, or hospitalizations as a result of a provoked non-therapeutic abortion or associated complications are not covered. The following are considered to be provoked abortions: |
| 7.5.8.2.3.1 | Dilatation and curettage (CPT Code 59840); |
| 7.5.8.2.3.2 | Dilatation and expulsion (CPT Code 59841); |
| 7.5.8.2.3.3 | Intra-amniotic injection (CPT Codes 59850, 59851, 59852); |
| 7.5.8.2.3.4 | One or more vaginal suppositories (e.g., Prostaglandin) with or without cervical dilatation (e.g., Laminar), including hospital admission and visits, fetus birth, and secundines (CPT Code 59855); |
| 7.5.8.2.3.5 | One or more vaginal suppositories (e.g., Prostaglandin) with dilatation and curettage/or evacuation (CPT Code 59856); and |
| 7.5.8.2.3.6 | One or more vaginal suppositories (e.g., Prostaglandin) with hysterectomy (omitted medical expulsion) (CPT Code 59857); and |
| 7.5.8.2.4 | Differential diagnostic interventions up to the confirmation of pregnancy are not covered. Any procedure after the confirmation of pregnancy will be at the Contractor’s own risk. |
| 7.5.8.3 | The Contractor shall implement a pre-natal and maternal program, aimed at preventing complications during and after pregnancy, and advancing the objective of lowering the incidence of low birth weight and premature deliveries. |
Page 81 of 301
| 7.5.8.3.1 | The program shall include, at a minimum, the following components: |
| 7.5.8.3.1.1 | A pre-natal care card, used to document services utilized; |
| 7.5.8.3.1.2 | Counseling regarding HIV testing; |
| 7.5.8.3.1.3 | Pregnancy testing; |
| 7.5.8.3.1.4 | A RhoGAM injection for all pregnant women who have a negative Rhesus (“Rh”) factor according to the established protocol; |
| 7.5.8.3.1.5 | Alcohol screening of pregnant women with the 4P-Plus instrument; |
| 7.5.8.3.1.6 | Smoking cessation counseling and treatment; |
| 7.5.8.3.1.7 | Post-partum depression screening using the Edinburgh post-natal depression scale; |
| 7.5.8.3.1.8 | Post-partum counseling and Referral to the WIC program; |
| 7.5.8.3.1.9 | Dental evaluation during the second trimester of gestation; and |
| 7.5.8.3.1.10 | Educational workshops regarding pre-natal care topics (importance of pre-natal medical visits and post-partum care), breast-feeding, stages of childbirth, oral and Behavioral Health, family planning, and newborn care, among others. |
| 7.5.8.3.2 | The Contractor shall ensure that eighty-five percent (85%) of pregnant Enrollees receive services under the Pre-Natal and Maternal Program. The Contractor shall submit its pre-natal and Maternal Program maternal wellness plan to ASES according to the timeframe specified in Attachment 12 to this Contract, and shall submit reports quarterly concerning the usage of services under this program. |
| 7.5.8.4 | The Contractor shall provide reproductive health and family planning counseling. Such services shall be provided voluntarily and confidentially, including circumstances where the Enrollee is under age eighteen (18). Family planning services will include, at a minimum, the following: |
Page 82 of 301
| 7.5.8.4.1 | Education and counseling necessary to make informed choices and understand contraceptive methods; |
| 7.5.8.4.2 | Pregnancy testing; |
| 7.5.8.4.3 | Diagnosis and treatment of sexually transmitted infections; |
| 7.5.8.4.4 | Infertility assessment; |
| 7.5.8.4.5 | -At least one of every class and category of FDA-approved contraceptive medication as specified in ASES’s PDL; and |
| 7.5.8.4.6 | At least one of every class and category of FDA-approved contraceptive method as specified by ASES. |
| 7.5.8.4.7 | Other FDA approved contraceptive medications or methods not covered by sections 7.5.8.4.5 or 7.5.8.4.6 of the Contract, when it is Medically Necessary and approved through a Prior Authorization or through an exception process and the prescribing Provider can demonstrate at least one of the following situations: |
| 7.5.8.4.7.1 | Contra-indication with drugs that are in the PDL that the Enrollee is already taking, and no other methods available in the PDL that can be use by the Enrollee. |
| 7.5.8.4.7.2 | History of adverse reaction by the Enrollee to the contraceptive methods covered as specified by ASES; or |
| 7.5.8.4.7.3 | History of adverse reaction by the Enrollee to the contraceptive medications that are on the PDL. |
| 7.5.9 | Emergency Services |
| 7.5.9.1 | The Contractor shall cover and pay for Emergency Services where necessary to treat an Emergency Medical Condition or a Psychiatric Emergency. The Contractor shall ensure that Medical and Psychiatric Emergency Services are available twenty-four (24) hours a day, seven (7) days per Week. The Contractor shall contract with emergency rooms in each Service Region in which it operates and ensure all emergency rooms have appropriate personnel to provide physical and Behavioral Health Services. No Prior Authorization will be required for Emergency Services. |
Page 83 of 301
| 7.5.9.2 | Emergency Services shall include the following without limitations: |
| 7.5.9.2.1 | Emergency room visits, including medical attention and routine and necessary services; |
| 7.5.9.2.2 | Trauma services; |
| 7.5.9.2.3 | Operating room use; |
| 7.5.9.2.4 | Respiratory therapy; |
| 7.5.9.2.5 | Specialist and sub-specialist treatment when required by the emergency room physician; |
| 7.5.9.2.6 | Anesthesia; |
| 7.5.9.2.7 | Surgical material; |
| 7.5.9.2.8 | Laboratory tests and X-Rays; |
| 7.5.9.2.9 | Post-Stabilization Services, as provided in Section 7.5.9.4 below; |
| 7.5.9.2.10 | Care as necessary in the case of a Psychiatric Emergency in an emergency room setting; |
| 7.5.9.2.11 | Drugs, medicine and intravenous solutions used in the emergency room; and |
| 7.5.9.2.12 | Transfusion of blood and blood plasma services, without limitations, including: |
| 7.5.9.2.12.1 | Authologal and irradiated blood; |
| 7.5.9.2.12.2 | Monoclonal factor IX with a certified hematologist Referral; |
| 7.5.9.2.12.3 | Intermediate purity concentrated ant hemophilic factor (Factor VIII); |
| 7.5.9.2.12.4 | Monoclonal type anti-hemophilic factor with a certified hematologist’s authorization; and |
| 7.5.9.2.12.5 | Activated protrombine complex (Autoflex and Feiba) with a certified hematologist’s authorization. |
| 7.5.9.3 | Emergency Services Within and Outside Puerto Rico |
| 7.5.9.3.1 | The Contractor shall make Emergency Services available: |
Page 84 of 301
| 7.5.9.3.1.1 | For all Enrollees, throughout Puerto Rico, including outside the Contractor’s Service Regions, and notwithstanding whether the emergency room is a Network Provider; and |
| 7.5.9.3.1.2 | For Medicaid and CHIP Eligibles, in Puerto Rico or in the US, when the services are Medically Necessary and could not be anticipated, notwithstanding that emergency rooms outside of Puerto Rico are not Network Providers. The Contractor shall be responsible for fulfilling payment for Emergency Services in the US in a timely manner. If, in an extenuating circumstance, a Medicaid or CHIP Eligible Enrollee incurs out-of-pocket expenses for Emergency Services provided in the US, the Contractor shall reimburse the Enrollee for such expenses in a timely manner, and the reimbursement shall be considered a Covered Service. |
| 7.5.9.3.2 | In covering Emergency Services provided by Puerto Rico Providers outside the Contractor’s Network, or by Providers in the US, the Contractor shall pay the Provider at least the average rate paid to Network Providers. |
| 7.5.9.4 | Post-Stabilization Services |
| 7.5.9.4.1 | The Contractor shall cover Post-Stabilization Services obtained from any Provider, regardless of whether the Provider is in the General Network or PPN, that are administered to maintain the Enrollee’s stabilized condition for one (1) hour while awaiting response on a Prior Authorization request. The attending Emergency Room physician or other treating Provider shall be responsible for determining whether the Enrollee is sufficiently stabilized for transfer or discharge. That determination will be binding for the Contractor with respect to its responsibility for coverage and payment. |
| 7.5.9.4.2 | An Enrollee who has been treated for an Emergency Medical Condition or Psychiatric Emergency shall not be held liable for any subsequent screening or treatment necessary to stabilize the Enrollee. |
| 7.5.9.4.3 | Financial Responsibility |
| 7.5.9.4.3.1 | The Contractor shall be financially responsible for Post-Stabilization Services obtained within or outside the Contractor’s General Network. These services will be subject to Prior Authorization by a Network Provider or any other Contractor representative. |
Page 85 of 301
| 7.5.9.4.3.2 | The Contractor shall be financially responsible for Post-Stabilization Services obtained within or outside the Contractor’s Network that are not given Prior Authorization by a Network Provider or other Contractor representative, but are administered to maintain, improve, or resolve the Enrollee’s stabilized condition if: |
| 7.5.9.4.3.2.1 | The Contractor does not respond to a request for Prior Authorization within one (1) hour; |
| 7.5.9.4.3.2.2 | The Contractor cannot be contacted; or |
| 7.5.9.4.3.2.3 | The Contractor and the treating physician cannot reach an agreement concerning the Enrollee’s care, and the participating Network Provider is not available for consultation. In this situation, the Contractor must give the treating physician the opportunity to consult with the participating Network Provider and the treating physician may continue with care of the patient until the participating Network Provider is reached or one of the criteria in 42 CFR 422.113(c)(3) is met. |
| 7.5.9.4.3.3 | The Contractor’s financial responsibility for Post-Stabilization Services that it has not Prior Authorized ends when: |
| 7.5.9.4.3.3.1 | A Network Provider with privileges at the treating hospital assumes responsibility for the Enrollee’s care; |
| 7.5.9.4.3.3.2 | A Network Provider assumes responsibility for the Enrollee’s care through transfer; |
Page 86 of 301
| 7.5.9.4.3.3.3 | A Contractor representative and the treating physician reach an agreement concerning the Enrollee’s care; or |
| 7.5.9.4.3.3.4 | The Enrollee is discharged. |
| 7.5.9.5 | Coverage of Services Ultimately Determined to be Non-Emergencies. The Contractor shall not retroactively deny a Claim for an emergency screening examination because the condition, which appeared to be an Emergency Medical Condition or a Psychiatric Emergency under the prudent layperson standard, turned out to be non-emergency in nature. |
| 7.5.9.6 | Enrollee Use of GHP Service Line. The Contractor shall train Emergency Services Providers concerning the GHP Service Line Medical Advice Service, and shall make Providers aware that: |
| 7.5.9.6.1 | An Enrollee who consults this service before visiting the emergency room shall not be responsible for any Co-Payment, provided that he or she presents his or her GHP Service Line call identification number when he or she arrives at the emergency room; |
| 7.5.9.6.2 | No Co-Payments shall be charged for Medicaid and CHIP children under eighteen (18) years of age under any circumstances. |
| 7.5.9.6.3 | No Co-Payments shall be imposed, or required, to an Enrollee to receive treatment for an Emergency Medical Condition or Psychiatric Emergency who is a Medicaid or CHIP Eligible; and the Contractor shall not deny payment for Emergency Services when the Enrollee seeks Emergency Services at the instruction of the Contractor or its Agent (including a GHP Service Line representative). |
| 7.5.9.7 | Coverage of All Emergency Medical Conditions and Psychiatric Emergencies. |
| 7.5.9.7.1 | The Contractor shall not deny payment for treatment of an Emergency Medical Condition or a Psychiatric Emergency, including cases in which the absence of immediate medical attention would not have resulted in the outcomes specified in the definition of Emergency Medical Condition or a Psychiatric Emergency in this Contract and in 42 CFR 438.114(a). |
Page 87 of 301
| 7.5.9.7.2 | The Contractor shall not refuse to cover an Emergency Medical Condition or a Psychiatric Emergency on the ground that the emergency room Provider, hospital, or fiscal Agent did not notify the Enrollee’s PCP or the Contractor of the Enrollee’s screening or treatment following the Enrollee’s arrival for Emergency Services. |
| 7.5.10 | Hospitalization Services |
| 7.5.10.1 | The Contractor shall provide hospitalization services, including the following: |
| 7.5.10.1.1 | Access to a nursery; |
| 7.5.10.1.2 | Access to a semi-private room (bed available twenty-four (24) hours a day, every Calendar Day of the year, including Puerto Rico holidays); |
| 7.5.10.1.3 | Access to an isolation room for physical or Behavioral Health reasons; |
| 7.5.10.1.4 | Food, including specialized nutrition services; |
| 7.5.10.1.5 | Regular nursing services; |
| 7.5.10.1.6 | Specialized room use, such as operation, surgical, recovery, treatment and maternity, without limitations; |
| 7.5.10.1.7 | Drugs, medicine, and contrast agents, without limitations; |
| 7.5.10.1.8 | Availability of materials such as bandages, gauze, plaster, or any other therapeutic or healing material; |
| 7.5.10.1.9 | Therapeutic and maintenance care services, including the use of the necessary equipment to offer the service; |
| 7.5.10.1.10 | Specialized diagnostic tests, such as electrocardiograms, electroencephalograms, arterial gases, and other specialized tests that are available at the hospital and necessary during the Enrollee's hospitalization; |
| 7.5.10.1.11 | Supply of oxygen, anesthetics, and other gases including administration; |
| 7.5.10.1.12 | Respiratory therapy, without limitations; |
| 7.5.10.1.13 | Rehabilitation services while Enrollee is hospitalized, including physical, occupational, and speech therapy; |
Page 88 of 301
| 7.5.10.1.14 | Outpatient surgery facility use; and |
| 7.5.10.1.15 | Transfusion of blood and blood plasma services, without limitations, including: |
| 7.5.10.1.15.1 | Authologal and irradiated blood; |
| 7.5.10.1.15.2 | Monoclonal factor IX with the Referral of a certified hematologist; |
| 7.5.10.1.15.3 | Intermediate purity concentrated ant hemophilic factor (Factor VIII); |
| 7.5.10.1.15.4 | Monoclonal type antihemophilic factor with a certified hematologist’s authorization; and |
| 7.5.10.1.15.5 | Activated protrombine complex (Autoflex and Feiba) with a certified hematologist’s authorization. |
| 7.5.10.2 | Hospitalization for services that would normally be considered outpatient services, or for diagnostic purposes only, is not a Covered Service under the GHP. |
| 7.5.11 | Behavioral Health Services |
| 7.5.11.1 | Covered Behavioral Health Services include the following: |
| 7.5.11.1.1 | Evaluation, screening, and treatment of individuals, couples, families and groups; |
| 7.5.11.1.2 | Outpatient services with psychiatrists, psychologists, and social workers; |
| 7.5.11.1.3 | Hospital or outpatient services for substance and alcohol abuse disorders; |
| 7.5.11.1.4 | Behavioral Health hospitalization; |
| 7.5.11.1.5 | Intensive outpatient services; |
| 7.5.11.1.6 | Immediate access to Emergency or crisis intervention Services twenty-four (24) hours a day, seven (7) days a Week (services outside of Puerto Rico available only for Medicaid and CHIP Eligibles); |
| 7.5.11.1.7 | Detoxification services for Enrollees intoxicated with illegal substances, whether as a result of substance abuse, a suicide attempt, or accidental poisoning; |
Page 89 of 301
| 7.5.11.1.8 | Long-lasting injected medicine clinics; |
| 7.5.11.1.9 | Escort/professional assistance and ambulance services when needed; |
| 7.5.11.1.10 | Prevention and secondary-education services; |
| 7.5.11.1.11 | Pharmacy coverage and access to medicine for a maximum of twenty-four (24) hours, in compliance with Act No. 408; |
| 7.5.11.1.12 | Medically Necessary clinical laboratories; |
| 7.5.11.1.13 | Treatment for Enrollees diagnosed with Attention Deficit Disorder (with or without hyperactivity). This includes, but is not limited to, neurologist visits and tests related to this diagnosis’s treatment; and |
| 7.5.11.1.14 | Substance abuse treatment. |
| 7.5.11.2 | Opiate Addiction Treatment |
| 7.5.11.2.1 | The Contractor shall provide appropriate services for Enrollees in need of Buprenorphine treatment due to a diagnosis of opiate addiction. The Contractor shall cover all services related to assessment, treatment, and monitoring of opiate addiction including: |
| 7.5.11.2.1.1 | Prescriptions for Buprenorphine or any other medically appropriate medications included on the PDL; |
| 7.5.11.2.1.2 | Comprehensive medical examination (CPT Code 99205); |
| 7.5.11.2.1.3 | Extended office visits (CPT Code 99215); |
| 7.5.11.2.1.4 | Brief office visit (CPT Code 99211); |
| 7.5.11.2.1.5 | Psychiatric Diagnostic Interview Exam – New Patient (CPT Code 90801); |
| 7.5.11.2.1.6 | Individual Therapy with Medical Evaluation and Management (CPT Code 90807); |
| 7.5.11.2.1.7 | Pharmacologic Management (CPT Code 90862); |
| 7.5.11.2.1.8 | Drug Urine Toxicology (CPT Code 80100); |
Page 90 of 301
| 7.5.11.2.1.9 | Blood Test Basic Metabolic Panel (CPT Code 80048); |
| 7.5.11.2.1.10 | Blood Test CBC (CPT Code 85025); |
| 7.5.11.2.1.11 | TB Test – Skin (CPT Code 86580), but only in conjunction with the prescription of Buprenorphine for the treatment of opiate addiction; |
| 7.5.11.2.1.12 | HIV Test (CPT Code 86703), but only in conjunction with the prescription of Buprenorphine for the treatment of opiate addiction; |
| 7.5.11.2.1.13 | Hepatitis Panel (CPT Code 80074), but only in conjunction with the prescription of Buprenorphine for the treatment of opiate addiction; |
| 7.5.11.2.1.14 | Individual Counseling (CPT Code 90806); |
| 7.5.11.2.1.15 | Group Counseling (CPT Code 90853); |
| 7.5.11.2.1.16 | Mental Health Assessment by Non-Physician Professional (CPT Code H0031); and |
| 7.5.11.2.1.17 | Alcohol and substance abuse Services, Treatment Plan Development and Modification (CPT Code T007). |
| 7.5.11.3 | The Contractor shall have Providers trained and certified by the Substance Abuse and Mental Health Services Administration (“SAMHSA”) to provide opiate addiction treatment. The training and certification of the Providers by SAMHSA may be evidenced with either (i) a copy of the letter issued by SAMHSA to the Provider certifying his/her training and certification or (ii) a copy of the Controlled Substance Registration Certification issued by the Drug Enforcement Administration with the identification number assigned to the Provider by SAMHSA. Evidence of SAMHSA certification shall be included in the Provider’s Credentialing file maintained by the Contractor. |
| 7.5.11.4 | The Contractor shall establish and strengthen relationships (if needed, through memoranda of understanding) with ASSMCA, ADFAN, the Office of the Women’s Advocate, and other government or nonprofit entities, in order to improve the delivery of Behavioral Health Services. |
| 7.5.12 | Pharmacy Services |
Page 91 of 301
| 7.5.12.1 | The Contractor shall provide pharmacy services under the GHP, including the following: |
| 7.5.12.1.1 | All costs related to prescribed medications for Enrollees, excluding the Enrollee’s Co-Payment where applicable; |
| 7.5.12.1.2 | Drugs on the Preferred Drug List (PDL); |
| 7.5.12.1.3 | Drugs included on the Master Formulary, but not in the PDL (through the exceptions process explained in Section 7.5.12.10); and |
| 7.5.12.1.4 | In some instances, through the exceptions process, drugs that are not included on either the PDL or the Master Formulary. |
| 7.5.12.2 | The Contractor may not impose restrictions on available prescription drugs beyond those stated in the PDL, Master Formulary, or any other drug formulary approved by ASES. |
| 7.5.12.3 | The following drugs are excluded from the pharmacy services Benefit: |
| 7.5.12.3.1 | Rebetron or any other medication prescribed for the treatment of Hepatitis C treatment (to be provided by the Health Department, upon Referral to the Health Department by a Network Provider. This medication is not provided through the GHP); and |
| 7.5.12.3.2 | Medications delivered directly to Enrollees by a Provider that does not have a pharmacy license, with the exception of medications that are traditionally administered in a doctor’s office, such as injections. |
| 7.5.12.4 | Prescriptions ordered under the pharmacy services Benefit are subject to the following Utilization controls: |
| 7.5.12.4.1 | Some or all prescription drugs may be subject to Prior Authorization, which shall be implemented and managed by the PBM or the Contractor, according to policies and procedures established by the ASES Pharmacy and Therapeutic (“P&T”) Committee and decided upon in consultation with the Contractor when applicable. |
| 7.5.12.4.2 | The Contractor shall ensure that Prior Authorization for pharmacy services is provided for the Enrollee in the following timeframes, including outside of normal business hours. |
Page 92 of 301
| 7.5.12.4.2.1 | The decision whether to grant a Prior Authorization of a prescription must not exceed seventy-two (72) hours from the time of the Enrollee’s Service Authorization Request for any Covered Service. An exception exists in circumstances where the Contractor or the Enrollee’s Provider determines that the Enrollee’s life or health could be endangered by a delay in accessing services. In such cases, Prior Authorization must be provided as expeditiously as the Enrollee’s health requires, and no later than within twenty-four (24) hours following the Service Authorization Request. |
| 7.5.12.4.2.2 | ASES may, in its discretion, grant an extension of the time allowed for Prior Authorization decisions, where: |
| 7.5.12.4.2.2.1 | The Enrollee, or the Provider, requests the extension; or |
| 7.5.12.4.2.2.2 | The Contractor justifies to ASES a need for the extension in order to collect additional information, such that the extension is in the Enrollee’s best interest. |
| 7.5.12.4.3 | Prescriptions written by a Provider who is outside the PPN may be filled only upon a Countersignature from the Enrollee’s PCP, or another assigned PCP from the PMG in case of absence or unavailability of the Enrollee’s PCP. A Countersignature request made to the PCP shall be acted upon within three (3) Calendar Days of the request of the prescribing Provider or, if the Enrollee’s health is in danger, within twenty-four (24) hours. |
| 7.5.12.4.4 | The Contractor shall not require a PCP Countersignature on prescriptions written by a Provider within the PPN. |
| 7.5.12.5 | The Contractor shall use bioequivalent drugs approved by the Food and Drug Administration (“FDA”), provided they are classified as “AB” and authorized by regulations, unless the Provider notes a contra-indication in the prescription. Nonetheless, the Contractor shall not refuse to cover a drug solely because the bioequivalent drug is unavailable; nor shall the Contractor impose an additional payment on the Enrollee because the bioequivalent is unavailable. |
Page 93 of 301
| 7.5.12.6 | The Contractor shall observe the following timeframe limits with respect to prescribed drugs: |
| 7.5.12.6.1 | Medication for critical conditions will be covered for a maximum of thirty (30) Calendar Days and for additional time, where Medically Necessary. |
| 7.5.12.6.2 | Medication for Chronic Conditions or severe Behavioral Health conditions will be covered for a maximum of thirty (30) Calendar Days, except at the beginning of therapy where, upon a Provider’s recommendation, a minimum of fifteen (15) Calendar Days shall be prescribed in order to reevaluate compliance and tolerance. Under a doctor’s orders, a prescription may be refilled up to five (5) times. |
| 7.5.12.6.3 | For maintenance drugs that require Prior Authorization, the Prior Authorization will be effective for six (6) months, unless there are contra-indications or side effects. |
| 7.5.12.6.4 | The prescribing Provider shall re-evaluate pharmacotherapy as to compliance, tolerance, and dosage within ninety (90) Calendar Days of having prescribed a maintenance drug. Dosage changes will not require Prior Authorization. Changes in the drug used may require Prior Authorization. |
| 7.5.12.7 | Special considerations, including cooperation with Puerto Rico governmental entities other than ASES, govern coverage of medications for the following conditions: |
| 7.5.12.7.1 | Medications for Treatment of HIV / AIDS |
| 7.5.12.7.1.1 | The following HIV/AIDS medications are excluded from the ASES PDL: Viread®, Emtriva®, Truvada®, Fuzeon®, Atripla®, Epzicom®, Selzentry®, Intelence®, Isentress®, Edurant®, Complera®, and Stribild®. |
| 7.5.12.7.1.2 | Because of an agreement between the Health Department and ASES, Enrollees diagnosed with HIV/AIDS may access the medications listed above through Health Department clinics. The Contractor is not At Risk for the coverage of these medications. |
| 7.5.12.7.1.3 | The Contractor shall inform Providers about this agreement, and shall require Providers to refer Enrollees for whom these medications are Medically Necessary to CPTET Centers (Centros de Prevención y Tratamiento de Enfermedades Transmisibles) or community-based organizations, where the Enrollee may be screened to determine whether the Enrollee is eligible for the AIDS Drug Assistance Program (“ADAP”). |
Page 94 of 301
| 7.5.12.7.1.4 | A list of CPTET Centers and community-based organizations that administer these medications is included as Attachment 4 to this Contract. |
| 7.5.12.7.2 | Medications for Chronic Conditions for Children with Special Health Needs. Directions for prescriptions for chronic use drugs for children with special health needs shall cover therapy for thirty (30) Calendar Days, and if necessary up to five (5) refills of the original prescription, according to medical opinion of a certified Provider. When Medically Necessary, additional prescriptions will be covered. |
| 7.5.12.7.3 | Medications for Enrollees with Opiate Addictions. See Section 7.5.11.2.1.1 above. |
| 7.5.12.8 | Except as provided in Section 7.5.12.3.2, all prescriptions must be dispensed by a pharmacy under contract with the PBM that is duly authorized under the laws of the Puerto Rico, and is freely selected by the Enrollee. The PBM shall maintain responsibility for ensuring that the pharmacy services network complies with the terms specified by ASES. |
| 7.5.12.9 | Prescribed drugs must be dispensed at the time and date, as established by the Puerto Rico Pharmacy Law, when the Enrollee submits the prescription for dispensation. |
| 7.5.12.10 | Use of PDL Medications. The Contractor shall ensure that drugs on the PDL are used whenever possible. |
| 7.5.12.10.1 | In the following two categories of exceptional cases, however, the Contractor shall cover drugs not included on the PDL, upon submission of acceptable written documentation of the medical justification for the drug from the Provider. |
| 7.5.12.10.1.1 | The Contractor shall cover drugs included on the Master Formulary (Attachment 5 to this Contract) in lieu of a PDL drug, only as a part of an exceptions process, upon a showing that no drug listed on the PDL is clinically effective for the Enrollee. |
Page 95 of 301
| 7.5.12.10.1.2 | The Contractor shall cover a drug that is not included on either the PDL or the Master Formulary, provided that the drug is not in an experimental stage and that the drug has been approved by the FDA for the treatment of the condition. |
| 7.5.12.10.2 | In addition to demonstrating that the drug prescribed has FDA approval and is not considered experimental, a Provider prescribing a drug not on the PDL must demonstrate that: |
| 7.5.12.10.2.1 | The drug does not have any bioequivalent on the market; and |
| 7.5.12.10.2.2 | The drug is clinically indicated because of: |
| 7.5.12.10.2.2.1 | Contra-indication with some drugs that are in the PDL that the Enrollee is already taking, and scientific literature’s indication of the possibility of serious adverse health effects related to the taking the drug; |
| 7.5.12.10.2.2.2 | History of adverse reaction by the Enrollee to some drugs that are on the PDL; |
| 7.5.12.10.2.2.3 | Therapeutic failure of all available alternatives on the PDL; or |
| 7.5.12.10.2.2.4 | Other special circumstances. |
| 7.5.12.11 | Role of Pharmacy Benefit Manager |
| 7.5.12.11.1 | Pharmacy services are administered primarily by a Pharmacy Benefit Manager (“PBM”) under contract with ASES. The Contractor shall work with the PBM as well as the Pharmacy Program Administrator (“PPA”) selected by ASES as needed, and as provided in this Section 7.5.12.11, in order to ensure the successful provision of pharmacy services. |
| 7.5.12.11.2 | The Contractor shall be obligated to accept the terms and conditions of the contract that ASES awards to a PBM. The Contractor shall use the procedures, guidelines, and other instructions implemented by ASES through the PBM. The Contractor and the PBM shall coordinate all the required efforts to achieve the integrated model of rendering all Covered Services to Enrollees under the GHP Program. |
Page 96 of 301
| 7.5.12.11.3 | Among other measures, to enhance cooperation with the PBM, the Contractor shall: |
| 7.5.12.11.3.1 | Work with the PBM to improve Information flow and to develop protocols for Information-sharing; |
| 7.5.12.11.3.2 | Establish, in consultation with the PBM, the procedures to transfer funds for the payment of Claims to the pharmacy network according to the payments cycle specified by the PBM; |
| 7.5.12.11.3.3 | Coordinate with the PBM to establish customer service protocols concerning pharmacy services; and |
| 7.5.12.11.3.4 | Collaborate with ASES to facilitate a smooth transition, since the PBM, PPA, and rebate contracts will take effect after April 1, 2015, which is the Implementation Date of this Contract. |
| 7.5.12.12 | Claims Processing and Administrative Services for Pharmacy. The Contractor shall: |
| 7.5.12.12.1 | Assume the cost of implementing and maintaining online connection with the PBM; |
| 7.5.12.12.2 | Cover all of its own costs of implementation, including but not limited to payment processes, Utilization review and approval processes, connection and line charges, and other costs incurred to implement the payment arrangements for pharmacy Claims; |
| 7.5.12.12.3 | Review Claims payments summary reports for each payment cycle and transfer funds required for payment to pharmacies; |
| 7.5.12.12.4 | Review denials and rejections of Claims; |
| 7.5.12.12.5 | Maintain a phone line to provide for the Prior Authorization of drugs, according to the established policies, the PDL, and the Master Formulary; and |
| 7.5.12.12.6 | Electronically submit a list of all Contractor’s Network Providers and a list of Enrollees to the PBM daily. |
Page 97 of 301
| 7.5.12.13 | Fraud Investigations. The Contractor shall develop tracking mechanisms for detecting Fraud, Waste, and Abuse related to pharmacy services, and shall forward Fraud, Waste, and Abuse Complaints from Enrollees related to pharmacy services to the PBM and to ASES. |
| 7.5.12.14 | Formulary Management Program |
| 7.5.12.14.1 | The Contractor shall select two (2) members of its staff to serve on a cross-functional committee, the Pharmacy Benefit Financial Committee, tasked with rebate maximization. The Committee will evaluate recommendations regarding the PDL, from the P&T Committee and the PPA, and will ultimately develop and review the PDL from time to time under the direction of ASES and the PPA. |
| 7.5.12.14.2 | The Contractor shall select a member of its staff to serve on a cross-functional subcommittee tasked with rebate maximization. The subcommittee will take recommendations on the PDL from the P&T Committee and will ultimately create and manage the PDL. |
| 7.5.12.15 | Utilization Management and Reports. The Contractor shall: |
| 7.5.12.15.1 | Perform drug Utilization reviews that meet the standards established by both ASES and Federal authorities; and |
| 7.5.12.15.2 | Develop and distribute protocols that will be subject to ASES approval, when necessary. |
| 7.5.12.16 | Communication with Providers. The Contractor shall ensure the following communications with Providers: |
| 7.5.12.16.1 | The Contractor shall advise Providers regarding the use of the PDL as a first option at the moment of prescribing and of the need to observe the exceptions process when filling a prescription for a drug not on the PDL. |
| 7.5.12.16.2 | The Contractor shall advise Providers that they may not outright deny medication because it is not included on ASES’s PDL. A medication not on the PDL may be provided through the exceptions process described in Section 7.5.12.10. |
| 7.5.12.16.3 | The Contractor shall advise Providers on the use of brand-name drugs and the availability of the bioequivalent version, if any. |
Page 98 of 301
| 7.5.12.17 | Cooperation with the Pharmacy Program Administrator (“PPA”) |
| 7.5.12.17.1 | The Contractor shall receive updates to the PDL from the PPA. The Contractor shall adhere to these updates. |
| 7.5.12.17.2 | Any rebates shall be negotiated by the PPA and retained in their entirety by ASES. The Contractor shall neither negotiate, collect, nor retain any pharmacy rebate for Enrollee Utilization of brand drugs included on ASES’s PDL. |
| 7.6 | Dental Services |
| 7.6.1 | The Contractor shall provide the following dental services as Covered Services: |
| 7.6.1.1 | All preventative and corrective services for children under age twenty-one (21) mandated by the EPSDT requirement; |
| 7.6.1.2 | Pediatric Pulp Therapy (Pulpotomy) for children under age twenty-one (21); |
| 7.6.1.3 | Stainless steel crowns for use in primary teeth following a Pediatric Pulpotomy; |
| 7.6.1.4 | Preventive dental services for Adults; |
| 7.6.1.5 | Restorative dental services for Adults; |
| 7.6.1.6 | One (1) comprehensive oral exam per year; |
| 7.6.1.7 | One (1) periodical exam every six (6) months; |
| 7.6.1.8 | One (1) defined problem-limited oral exam; |
| 7.6.1.9 | One (1) full series of intra-oral radiographies, including bite, every three (3) years; |
| 7.6.1.10 | One (1) initial periapical intra-oral radiography; |
| 7.6.1.11 | Up to five (5) additional periapical/intra-oral radiographies per year; |
| 7.6.1.12 | One (1) single film-bite radiography per year; |
| 7.6.1.13 | One (1) two-film bite radiography per year; |
| 7.6.1.14 | One (1) panoramic radiography every three (3) years; |
| 7.6.1.15 | One (1) Adult cleanse every six (6) months; |
Page 99 of 301
| 7.6.1.16 | One (1) child cleanse every six (6) months; |
| 7.6.1.17 | One (1) topical fluoride application every six (6) months for Enrollees under nineteen (19) years old; |
| 7.6.1.18 | Fissure sealants for life for Enrollees up to fourteen (14) years old (including decidual molars up to eight (8) years old when Medically Necessary because of cavity tendencies); |
| 7.6.1.19 | Amalgam restoration; |
| 7.6.1.20 | Resin restorations; |
| 7.6.1.21 | Root canal; |
| 7.6.1.22 | Palliative treatment; and |
| 7.6.1.23 | Oral surgery. |
| 7.7 | Special Coverage |
| 7.7.1 | The Special Coverage Benefit is designed to provide services for Enrollees with special health care needs caused by serious illness. |
| 7.7.2 | The Contractor shall provide ASES with the strategy implemented for the identification of populations with special health care needs in order to identify any ongoing special conditions of Enrollees that require a treatment plan and regular care monitoring by appropriate Providers. |
| 7.7.3 | The Contractor shall implement a system for screening Enrollees for Special Coverage and registering Enrollees who qualify. The Contractor shall design a form, with prior written approval from ASES, to be used by Providers in submitting a registration for Special Coverage. |
| 7.7.4 | The registration system for Special Coverage shall emphasize speedy processing of the registration that requires the Contractor, once it receives the notification from the Provider, to register the Member in Special Coverage within seventy-two (72) hours. |
| 7.7.5 | Once a Provider supplies all the required information for the Contractor to process a registration and the Contractor processes such information, Special Coverage shall take effect retroactively as of the date the Provider reaches a diagnosis, including documentation of test results, for any condition included in Special Coverage. In case Information is submitted to the Contractor after the diagnosis was reached, coverage can be made retroactive up to sixty (60) Calendar Days before the date on which Provider submitted the registration request. |
Page 100 of 301
| 7.7.6 | According to the timeframes specified in Attachment 12 to this Contract, the Contractor shall submit proposed protocols to be established for Special Coverage to ASES for prior written approval, including: |
| 7.7.6.1 | Registration procedures; |
| 7.7.6.2 | Formats established for registration forms; |
| 7.7.6.3 | Forms of notices to be issued to the Enrollee and to the Provider to inform them of the Contractor’s decision concerning Special Coverage; |
| 7.7.6.4 | Protocols for the development of a treatment plan; |
| 7.7.6.5 | Provisions for ensuring that Enrollees with Special Coverage have Immediate Access to specialists appropriate for the Enrollee’s condition and identified needs; and |
| 7.7.6.6 | A summary of the Contractor’s strategy for the identification of populations with special health care needs. |
| 7.7.7 | The protocols shall emphasize both the need for a speedy determination and the need for screening evaluations to be conducted by competent Providers with appropriate expertise. |
| 7.7.8 | The Contractor shall complete, monitor, and routinely update a treatment plan for each Enrollee who is registered for Special Coverage. |
| 7.7.8.1 | The treatment plan shall be developed by the Enrollee’s PCP, with the Enrollee’s participation, and in consultation with any specialists caring for the Enrollee. The Contractor shall require, in its Provider Contracts with PCPs, that Special Coverage registration treatment plans be submitted to the Contractor for review and approval in a timely manner. |
| 7.7.9 | Autism |
| 7.7.9.1 | The physical and Behavioral Health Services, that the autism population needs to access through specialists such as gastroenterologists, neurologists, allergists, and dentists, will be offered through Special Coverage. The Uniform Guide for Special Coverage (Attachment 7 to this Contract) includes the mandated procedures for this condition. |
| 7.7.9.2 | The Contractor shall require in its Provider Contracts with PCPs that the PCP carry out the Modified Checklist for Autism in Toddlers (“M-CHAT”) screen to detect autism in Enrollees under the age of eighteen (18) months, or in any other age range established by the Health Department. Once the PCP diagnoses autism, the PCP will refer the patient to the Behavioral Health Provider. The M-CHAT test may be accessed through the Internet, and does not entail any cost, nor does it infringe on any copyright. |
Page 101 of 301
| 7.7.9.3 | The Contractor shall also require, through its Provider Contracts, that PCPs administer the Ages and Stages Questionnaire (“ASQ”) to the parents of child Enrollees. This questionnaire must be completed when the child is nine (9), eighteen (18), and thirty (30) months old, or at any other age established by the Health Department. ASES acquired the license for the exclusive use of the questionnaire for child Enrollees in the GHP and will provide the questionnaires to the Contractor, who shall transmit the questionnaire to PCPs and train and educate them in its use. |
| 7.7.10 | Services provided under Special Coverage shall be subject to Prior Authorization by the Contractor. |
| 7.7.11 | Special Coverage shall include in its scope the following services, provided, however, that an Enrollee shall be entitled only to those services Medically Necessary to treat the condition that qualified the Enrollee for Special Coverage: |
| 7.7.11.1 | Coronary and intensive care services, without limit; |
| 7.7.11.2 | Maxillary surgery; |
| 7.7.11.3 | Neurosurgical and cardiovascular procedures, including pacemakers, valves, and any other instrument or artificial devices (Prior Authorization required); |
| 7.7.11.4 | Peritoneal dialysis, hemodialysis, and related services (Prior Authorization required); |
| 7.7.11.5 | Pathological and clinical laboratory tests that are required to be sent outside Puerto Rico for processing (Prior Authorization required); |
| 7.7.11.6 | Neonatal intensive care unit services, without limit; |
| 7.7.11.7 | Radioisotope, chemotherapy, radiotherapy, and cobalt treatments; |
| 7.7.11.8 | Treatment of gastrointestinal conditions, treatment of allergies, and nutritional services in autism patients; |
| 7.7.11.9 | The following procedures and diagnostic tests, when Medically Necessary (Prior Authorization required): |
| 7.7.11.9.1 | Computerized Tomography; |
Page 102 of 301
| 7.7.11.9.2 | Magnetic resonance test; |
| 7.7.11.9.3 | Cardiac catheters; |
| 7.7.11.9.4 | Holter test; |
| 7.7.11.9.5 | Doppler test; |
| 7.7.11.9.6 | Stress tests; |
| 7.7.11.9.7 | Lithotripsy; |
| 7.7.11.9.8 | Electromyography; |
| 7.7.11.9.9 | Single-photon Emission Computed Topography (“SPECT”) test; |
| 7.7.11.9.10 | Orthopantogram (“OPG”) test; |
| 7.7.11.9.11 | Impedance Plesthymography; |
| 7.7.11.9.12 | Other neurological, cerebrovascular, and cardiovascular procedures, invasive and noninvasive; |
| 7.7.11.9.13 | Nuclear imaging; |
| 7.7.11.9.14 | Diagnostic endoscopies; and |
| 7.7.11.9.15 | Genetic studies; |
| 7.7.11.10 | Up to fifteen (15) additional (beyond the services provided under Basic Coverage) physical therapy treatments per Enrollee condition per year when indicated by an orthopedist, physiatrist or chiropractor after Contractor Prior Authorization; |
| 7.7.11.11 | General anesthesia, including for dental treatment of special-needs children; |
| 7.7.11.12 | Hyperbaric Chamber; |
| 7.7.11.13 | Immunosuppressive medicine and clinical laboratories required for the maintenance treatment of post-surgical patients or transplant patients, to ensure the stability of the Enrollee's health, and for emergencies that may occur after said surgery; and |
| 7.7.11.14 | Treatment for the following conditions after confirmed laboratory results and established diagnosis: |
| 7.7.11.14.1 | HIV Positive factor and/or Acquired Immunodeficiency Syndrome (“AIDS”) (Outpatient and hospitalization services are included; no Referral or Prior Authorization is required for Enrollee visits and treatment at the Health Department's Regional Immunology Clinics or other qualified Providers); |
Page 103 of 301
| 7.7.11.14.2 | Tuberculosis; |
| 7.7.11.14.3 | Leprosy; |
| 7.7.11.14.4 | Lupus; |
| 7.7.11.14.5 | Cystic Fibrosis; |
| 7.7.11.14.6 | Cancer; |
| 7.7.11.14.7 | Hemophilia; |
| 7.7.11.14.8 | Special conditions of children, including the prescribed conditions in the Special Needs Children Diagnostic Manual Codes (see Attachment 13), except: |
| 7.7.11.14.8.1 | Asthma and diabetes, which are included in the Disease Management program; |
| 7.7.11.14.8.2 | Psychiatric Disorders; and |
| 7.7.11.14.8.3 | Intellectual disabilities; |
| 7.7.11.14.9 | Scleroderma; |
| 7.7.11.14.10 | Multiple Sclerosis; |
| 7.7.11.14.11 | Conditions resulting from self-inflicted damage or as a result of a felony or negligence by an Enrollee; and |
| 7.7.11.14.12 | Chronic renal disease in levels three (3), four (4) and five (5) (Levels 1 and 2 are included in the Basic Coverage); these levels of renal disease are defined as follows: |
| 7.7.11.14.12.1 | Level 3 – GFR (Glomerular Filtration – ml/min. per 1.73m² per corporal surface area) between 30 and 59, a moderate decrease in kidney function; |
| 7.7.11.14.12.2 | Level 4 - GFR between 15 and 29, a severe decrease in kidney function; and |
Page 104 of 301
| 7.7.11.14.12.3 | Level 5 – GFR under 15, renal failure that will probably require either dialysis or a kidney transplant. |
| 7.7.11.15 | Required medication for the outpatient treatment of Tuberculosis and Leprosy is included under Special Coverage. Medication for the outpatient treatment or hospitalization for AIDS-diagnosed Enrollees or HIV-positive Enrollees is also included, with the exception of Protease inhibitors which will be provided by CPTET Centers. |
| 7.7.12 | An Enrollee may register for Special Coverage based on one (1) of the conditions listed in Attachment 7 to this Contract (Uniform Guide to Special Coverage). The Contractor must seek ASES Prior Authorization for any other special condition not listed in Attachment 7, which the Enrollee, PCP, or PMG requests to be the basis of Special Coverage for an Enrollee. The request must include sufficient documentation of the Enrollee’s need for services and the cost-effectiveness of the care option. ASES will consult with the Health Department and issue a decision which will be binding between the Parties. |
| 7.7.13 | The Contractor must have a mechanism in place to allow Enrollees to directly access a specialist as appropriate for the enrollee’s condition and identified needs, in regards to all services encompassed within the scope of Special Coverage. |
| 7.7.14 | Except as expressly noted in this Section 7.7, the exclusions applied to Basic Coverage apply to Special Coverage. |
| 7.8 | Administrative Functions |
| 7.8.1 | Benefits under the GHP include the Administrative Functions of Care Management, Disease Management, and the Wellness Plan (see Section 12.5.8 of this Contract), which are intended to coordinate care for Enrollees with intense health service needs. |
| 7.8.2 | Care Management |
| 7.8.2.1 | The Contractor shall be responsible for the Care Management of Enrollees who demonstrate the greatest need, including those who have catastrophic, high-cost, or high-risk conditions and/or who require intensive assistance to ensure integration of physical and Behavioral Health needs. |
| 7.8.2.2 | Enrollees who present with the following conditions shall be offered Care Management and may elect to opt out of the program: |
Page 105 of 301
| 7.8.2.2.1 | Enrollees identified special health care needs and whom qualify for Special Coverage; |
| 7.8.2.2.2 | Enrollees diagnosed with a Serious Mental Illness or a Serious Emotional Disability (“SMI/SED”); |
| 7.8.2.2.3 | Enrollees identified as high-cost and/or high-risk; or |
| 7.8.2.2.4 | Enrollees who have accessed the emergency room seven (7) or more times within twelve (12) months. |
| 7.8.2.3 | The Contractor’s Care Management system shall emphasize prevention, continuity of care, and coordination of care. The system will advocate for, and link Enrollees to, services as necessary across Providers and settings. Care Management functions include: |
| 7.8.2.3.1 | Assignment of a specific Care Manager to each enrollee qualified for Care Management; |
| 7.8.2.3.2 | Management of Enrollee to Care Manager ratios that have been reviewed and approved by ASES; |
| 7.8.2.3.3 | Identification of Enrollees who have or may have chronic or severe Behavioral Health needs, including through use of the screening tools M-CHAT for the detection of Autism, ASQ, ASQ-SE, Conners Scale (ADHD screen), DAST-10, GAD, and PC-PTSD, and other tools available for diagnosis of Behavioral Health disorders; |
| 7.8.2.3.4 | Assessment of an Enrollee’s physical and Behavioral Health needs utilizing a standardized needs assessment within thirty (30) Calendar Days of Referral to Care Management that has been reviewed and given written approval by ASES; |
| 7.8.2.3.5 | Development of a plan of care within sixty (60) Calendar Days of the needs assessment; |
| 7.8.2.3.6 | Referrals and assistance to ensure timely Access to Providers; |
| 7.8.2.3.7 | Coordination of care actively linking the Enrollee to Providers, medical services, residential, social, and other support services where deemed necessary; |
| 7.8.2.3.8 | Monitoring of the Enrollees needs for assistance and additional services via face-to-face or telephonic contact at least quarterly (based on high- or low-risk; |
Page 106 of 301
| 7.8.2.3.9 | Continuity and transition of care; and |
| 7.8.2.3.10 | Follow-up and documentation. |
| 7.8.2.4 | The Contractor shall develop policies and procedures for Care Management that include, at a minimum, the following elements: |
| 7.8.2.4.1 | The provision of an individual needs assessment and diagnostic assessment; |
| 7.8.2.4.2 | The development of an individual treatment plan, as necessary, based on the needs assessment; |
| 7.8.2.4.3 | The establishment of treatment objectives; |
| 7.8.2.4.4 | The monitoring of outcomes; |
| 7.8.2.4.5 | A process to ensure that treatment plans are revised as necessary; |
| 7.8.2.4.6 | A strategy to ensure that all Enrollees or Authorized Representatives, as well as any specialists caring for the Enrollee, are involved in a treatment planning process coordinated by the PCP; |
| 7.8.2.4.7 | Procedures and criteria for making Referrals to specialists and subspecialists; |
| 7.8.2.4.8 | Procedures and criteria for maintaining care plans and Referral services when the Enrollee changes Providers; |
| 7.8.2.4.9 | Capacity to implement, when indicated, Care Management functions such as individual needs assessment, including establishing treatment objectives, treatment follow-up, monitoring of outcomes, or revision of the treatment plan; and |
| 7.8.2.4.10 | Process for referring Enrollees into Disease Management. |
| 7.8.2.5 | These procedures must be designed to include consultation and coordination with Enrollee’s PCP. |
| 7.8.2.6 | The Contractor shall submit its Care Management policies and procedures to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 7.8.3 | Disease Management |
Page 107 of 301
| 7.8.3.1 | The Contractor shall develop a Disease Management program for individuals with Chronic Conditions, including the following: |
| 7.8.3.1.1 | Asthma; |
| 7.8.3.1.2 | Depression; |
| 7.8.3.1.3 | Diabetes Type 1 or 2; |
| 7.8.3.1.4 | Congestive heart failure; |
| 7.8.3.1.5 | Hypertension; |
| 7.8.3.1.6 | Obesity; |
| 7.8.3.1.7 | Chronic renal disease, levels 1 and 2 (see definition at Section 7.5.6.1.17.1); and |
| 7.8.3.1.8 | Other conditions as determined necessary by ASES. |
| 7.8.3.2 | The Contractor shall identify and categorize Enrollees using clinical protocols of the Health Department and ASSMCA, and the protocols developed by the Committee for Management of Conditions established by ASES. |
| 7.8.3.3 | The Contractor shall report quarterly on the number of Enrollees diagnosed with each of these conditions. |
| 7.8.3.4 | The Contractor shall develop Disease Management policies and procedures detailing its program, including how Enrollees are identified for and referred to Disease Management, Disease Management program descriptions, and monitoring and evaluation activities. |
| 7.8.3.5 | The Contractor shall submit its Disease Management policies and procedures to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 7.8.3.6 | The Contractor shall require in its policies and procedures that an individualized treatment plan be developed for each Enrollee who receives Disease Management services. The policies and procedures shall include a strategy to ensure that all Enrollees or Authorized Representatives, as well as any specialists caring for the Enrollee, are involved in a treatment planning process coordinated by the PCP. |
Page 108 of 301
| 7.9 | Early and Periodic Screening, Diagnosis and Treatment Requirements (“EPSDT”) |
| 7.9.1 | The Contractor shall provide EPSDT services to Medicaid Eligibles and CHIP Eligible children less than twenty-one (21) years of age in compliance with all requirements found below. EPSDT services must be in compliance with Health Department guidelines and the Mothers, Children and Adolescents Program guidelines. ASES may issue additional guidelines to the Contractor in regards to the applicable EPSDT services. |
| 7.9.1.1 | The Contractor shall comply with sections 1902(a)(43), 1905(a)(4)(B), and 1905(r) of the Social Security Act, and Part 5 of the State Medicaid Manual, which require EPSDT services to include Outreach and education, screening, tracking, and diagnostic and treatment services. |
| 7.9.1.2 | The Contractor shall develop an EPSDT Plan that includes written policies and procedures for conducting Outreach and education, informing, tracking, and organizing follow-up to ensure compliance with the Healthy Child periodicity schedules. |
| 7.9.1.3 | The EPSDT Plan shall emphasize Outreach and compliance monitoring for children and adolescents (young adults), taking into account the multi-lingual, multi-cultural nature of the population, as well as other unique characteristics of this population. |
| 7.9.1.4 | The EPSDT Plan shall include procedures for follow-up of missed appointments, including missed Referral appointments for problems identified through EPSDT screens and exams. The plan shall also include procedures for Referral, tracking, and follow-up for annual dental examinations and visits. The Contractor shall submit its EPSDT Plan for review and approval according to the timeframe specified in Attachment 12 to this Contract. |
| 7.9.2 | Outreach and Education |
| 7.9.2.1 | The Contractor’s EPSDT Outreach and education process for Medicaid and CHIP Eligible children and their families shall include: |
| 7.9.2.1.1 | The importance of preventive care; |
| 7.9.2.1.2 | The periodicity schedule and the depth and breadth of services; |
| 7.9.2.1.3 | How and where to access services, including necessary transportation and scheduling services; and |
| 7.9.2.1.4 | A statement that services are provided without cost. |
Page 109 of 301
| 7.9.2.2 | The Contractor shall provide written notification to its families with EPSDT-eligible children when appropriate periodic assessments or needed services are due. The Contractor shall coordinate appointments for care. The Contractor shall follow-up with families with EPSDT-eligible children who have failed to access Healthy Child services after one hundred and twenty (120) Calendar Days of Enrollment in the GHP. |
| 7.9.2.3 | The Contractor shall inform its newly enrolled families with EPSDT-Eligible children about the EPSDT Program upon Enrollment with the Plan. This requirement includes informing pregnant women and new mothers, either before or within fourteen (14) Calendar Days after the birth of their children, that EPSDT services are available. |
| 7.9.2.4 | The Contractor shall provide each PCP, on a monthly basis, with a list of the PCP’s EPSDT-eligible children who have not had an appointment during the initial one hundred and twenty (120) Calendar Days of Enrollment, and/or are not in compliance with the EPSDT periodicity schedule. The Contractor and/or the PCP shall subsequently contact the Enrollees’ parents or guardians to schedule an appointment. |
| 7.9.2.5 | Outreach and education shall include a combination of written and oral (on the telephone, face-to-face, or films/tapes) methods, and may be done by Contractor personnel or by Providers. All Outreach and education shall be documented and shall be conducted in non-technical language at or below a fourth (4th) -grade reading level. The Contractor shall use accepted methods for informing persons who are blind or deaf, or cannot read or understand the Spanish language. |
| 7.9.2.6 | The Contractor may provide nominal, non-cash incentives of ten dollars ($10) or less to Enrollees and no more than fifty dollars ($50) in the aggregate per Enrollee, to motivate compliance with periodicity schedules if prior approved in writing by ASES. |
| 7.9.3 | Screening |
| 7.9.3.1 | The Contractor is responsible for periodic screens (“EPSDT Checkups”) in accordance with the Puerto Rico Medicaid Program’s periodicity schedule and the American Academy of Pediatrics EPSDT periodicity schedule. Such EPSDT Checkups shall include, but not be limited to, the Healthy Child checkups described in Section 7.5.3.1. |
| 7.9.3.2 | The Contractor shall provide an initial health and screening visit to all newly enrolled CHIP Eligible children within ninety (90) Calendar Days and within twenty-four (24) hours of birth to all newborns; and, after the initial Checkup, annually. |
Page 110 of 301
| 7.9.3.3 | The Contractor must advise the Enrollee child, his or her parents, or his or her legal guardian of his or her right to have an EPSDT Checkup. |
| 7.9.3.4 | EPSDT Checkups must include all of the following: |
| 7.9.3.4.1 | A comprehensive health and developmental history; |
| 7.9.3.4.2 | Developmental assessment, including mental, emotional, and Behavioral Health development; |
| 7.9.3.4.3 | Measurements (including head circumference for infants); |
| 7.9.3.4.4 | An assessment of nutritional status; |
| 7.9.3.4.5 | A comprehensive unclothed physical exam; |
| 7.9.3.4.6 | Immunizations according to the guidance issued by the Advisory Committee on Immunization Practices (ACIP) (the vaccines themselves are provided and paid for by the Health Department for the Medicaid and CHIP Eligibles. The vaccine is provided and paid for by the Contractor for the Other Eligible Persons in the GHP.) The Contractor shall cover the administration of the vaccines according to the fee schedule established by the Health Department; |
| 7.9.3.4.7 | Certain laboratory tests; |
| 7.9.3.4.8 | Anticipatory guidance and health education; |
| 7.9.3.4.9 | Vision screening; |
| 7.9.3.4.10 | Tuberculosis; |
| 7.9.3.4.11 | Hearing screening; and |
| 7.9.3.4.12 | Dental and oral health assessment. |
| 7.9.3.5 | Lead screening is a required component of an EPSDT Checkup, and the Contractor shall implement a screening program for the detection of the presence of lead toxicity. The screening program shall consist of two (2) parts: verbal risk assessment (from thirty-six (36) to seventy-two (72) months of age), and blood screening for lead. Regardless of risk, the Contractor shall provide for a blood screening testing for lead for all EPSDT-Eligible children at twelve (12) and twenty-four (24) months of age. Children between twenty-four (24) months of age and seventy-two (72) months of age should receive a blood lead screening testing for lead if there is no record of a previous test. |
Page 111 of 301
| 7.9.3.6 | The Contractor shall have procedures for Provider Referral to and follow-up with dental service Providers, including annual dental examinations and services by an oral health Provider. |
| 7.9.3.7 | The Contractor shall have procedures for Provider Referral of children for further diagnostic and/or treatment services to correct or ameliorate defects, physical and mental illnesses, and conditions discovered by the EPSDT checkup. Referral to the Provider conducting the screening or to another Provider may be made, as appropriate, as well as any follow-up appointments. |
| 7.9.3.8 | The Contractor shall ensure at a minimum fifty percent (50%) compliance during the first Contract year, sixty percent (60%) compliance during the second Contract year, and seventy-five percent (75%) compliance during the third Contract year, with the EPSDT screening requirements, including blood screening for lead and annual dental examinations and services, using the methodology prescribed by CMS to determine the screening rate. ASES may impose penalties, sanctions, and/or fines under Articles 19 and 20 if the Contractor fails to comply with the minimum requirements. |
| 7.9.4 | Tracking |
| 7.9.4.1 | The Contractor shall establish a tracking system that provides Information on compliance with EPSDT requirements. This system shall have in a place a reminder/notification system and shall track, at a minimum, the following areas: |
| 7.9.4.1.1 | Initial newborn Healthy Child hospital checkups; |
| 7.9.4.1.2 | Periodic EPSDT checkups as required by the periodicity schedule; |
| 7.9.4.1.3 | Diagnostic and treatment services, including Referrals; and |
| 7.9.4.1.4 | Immunizations, lead, tuberculosis, and dental services. |
| 7.9.4.2 | All Information generated and maintained in the tracking system shall be consistent with Encounter Data requirements as specified in Section 17.3.3 of this Contract. |
| 7.9.5 | Diagnostic and Treatment Services |
Page 112 of 301
| 7.9.5.1 | If a suspected problem is detected by a screening examination as described above, the child shall be evaluated as necessary for further diagnosis. This diagnosis is used to determine treatment needs. |
| 7.9.5.2 | EPSDT requires coverage for all follow-up diagnostic and treatment services deemed Medically Necessary to ameliorate or correct a problem discovered during an EPSDT checkup. Such Medically Necessary diagnostic and treatment services must be provided regardless of whether such services are covered by the State Medicaid Plan, as long as they are Medicaid-coverable Services as defined in Title XIX of the Social Security Act. The Contractor shall provide Medically Necessary, Medicaid-coverable diagnostic and treatment services. |
| 7.9.6 | EPSDT Reporting – See Section 18.2.4 of this Contract. |
| 7.10 | Advance Directives |
| 7.10.1 | In compliance with 42 CFR 438.6 (i), Law No. 160 of November 17, 2001, and 42 CFR 489.100, the Contractor shall maintain written policies and procedures for Advance Directives. Such Advance Directives shall be included in each Enrollee’s Medical Record. The Contractor shall provide these policies and procedures written at a fourth (4th) grade reading level in English and Spanish to all Enrollees eighteen (18) years of age and older and shall advise Enrollees of: |
| 7.10.1.1 | Their rights under the laws of Puerto Rico, including the right to accept or refuse medical or surgical treatment and the right to formulate Advance Directives; |
| 7.10.1.2 | The Contractor’s written policies respecting the implementation of those rights, including a statement of any limitation regarding the implementation of Advance Directives as a matter of conscience; and |
| 7.10.1.3 | The Enrollee’s right to file Complaints concerning noncompliance with Advance Directive requirements directly with ASES or with the Puerto Rico Office of the Patient Advocate. |
| 7.10.2 | The Information must include a description of Puerto Rico law and must reflect changes in laws as soon as possible and no later than ninety (90) Calendar Days after the effective change. |
| 7.10.3 | The Contractor shall educate its staff about its policies and procedures on Advance Directives, situations in which Advance Directives may be of benefit to Enrollees, and the staff’s responsibility to educate Enrollees about this tool and assist them in making use of it. |
Page 113 of 301
| 7.10.4 | The Contractor shall educate Enrollees about their ability to direct their care using Advance Directives and shall specifically designate which staff members or Network Providers are responsible for providing this education. |
| 7.11 | Enrollee Cost-Sharing |
| 7.11.1 | The Contractor shall ensure that Providers collect the Enrollee’s cost-sharing portion only as specified in Attachment 8 to this Contract. |
| 7.11.2 | The Contractor shall ensure that it accurately differentiates the categories of GHP Enrollees in its Marketing Materials and communications, to clarify the cost-sharing rules that are applied to each group. The Contractor shall ensure that the Enrollee’s eligibility category appears on the Enrollee ID Card, so that cost-sharing is correctly determined. |
| 7.11.3 | The Contractor shall ensure that, in keeping with the Co-Payment policies included in Attachment 8, Medicaid and CHIP Eligibles bear no cost-sharing responsibility under the GHP for services provided within the Contractor’s PPN. |
| 7.11.4 | As provided in Attachment 8 to this Contract, the Contractor shall impose Co-Payments for services provided in an emergency room outside the Enrollee’s PPN, but only in limited circumstances. |
| 7.11.4.1 | For Medicaid and CHIP Eligibles, the Contractor shall not impose any Co-Payment for the treatment of an Emergency Medical Condition or a Psychiatric Emergency. The Contractor shall, however, as provided in Attachment 8 to this Contract, impose Co-Payments for services provided in an emergency room to treat a condition that the attending physician determines, at the time of the visit, does not meet the definition of a Psychiatric Emergency or an Emergency Medical Condition and if the Enrollee does not consult the GHP Service Line Medical Advice Line before visiting the emergency room, and provide his or her call identification number at the emergency room. If the Enrollee presents the call identification number, no Co-Payment shall be imposed. |
| 7.11.4.2 | No Co-Payments shall be charged for Medicaid and CHIP children under eighteen (18) years of age under any circumstances. |
| 7.11.4.3 | For Other Eligible Persons, the Contractor shall impose a Co-Payment for any emergency room visit outside the Enrollee’s PPN, if the Enrollee does not consult the GHP Service Line Medical Advice Line before visiting the emergency room, and provide his or her call identification number at the emergency room. If the Enrollee presents the call identification number, no Co-Payment shall be imposed. |
Page 114 of 301
| 7.11.5 | As provided in 42 CFR 447.53(e), if a Medicaid or CHIP Eligible expresses his or her inability to pay the established Co-Payment at the time of service, the Contractor (through its contracted Providers) shall not deny the service. |
| 7.11.6 | Enrollees of Indian background, as defined in Article 2, are exempt from all Co-Payments. |
| 7.12 | Dual Eligible Beneficiaries |
| 7.12.1 | Dual Eligible Beneficiaries enrolled in the GHP are eligible, with the limitations provided below, for the Covered Services described in this Article 7, with the addition of some coverage of Medicare cost-sharing. |
| 7.12.1.1 | Dual Eligible Beneficiaries Who Receive Medicare Part A Only |
| 7.12.1.1.1 | The Contractor shall provide regular GHP coverage as provided in this Article 7, excluding services covered under Medicare Part A (hospitalization). However, the GHP shall cover hospitalization services after the Medicare Part A coverage limit has been reached. |
| 7.12.1.1.2 | The Contractor shall not cover the Medicare Part A premium or Deductible. |
| 7.12.1.2 | Dual Eligible Beneficiaries Who Receive Medicare Part A and Part B |
| 7.12.1.2.1 | The Contractor shall provide regular GHP coverage as detailed in this Article 7, excluding services covered under Medicare Part A or Part B. However, the GHP shall cover hospitalization services after the Medicare Part A coverage limit has been reached. |
| 7.12.1.2.2 | The Contractor shall not cover the Medicare Part A premium or Deductible. |
| 7.12.1.2.3 | The Contractor shall cover Medicare Part B Deductibles and co-insurance. |
| 7.12.1.3 | Dual Eligible Beneficiaries Enrolled in a Medicare Part C and/or Platino Plan are not eligible for services under this Contract. |
| 7.12.2 | Any GHP cost-sharing for Dual Eligible Beneficiaries shall be determined according to Section 7.11 and Attachment 8. |
Page 115 of 301
| 7.13 | Moral or Religious Objections |
| 7.13.1 | If, during the course of the Contract period, pursuant to 42 CFR 438.102, the Contractor elects not to provide, not to reimburse for, or not to provide a Referral or Prior Authorization for a service within the scope of the detailed Covered Services, because of an objection on moral or religious grounds, the Contractor shall notify: |
| 7.13.1.1 | ASES within one hundred and twenty (120) Calendar Days before adopting the policy with respect to any service; |
| 7.13.1.2 | Enrollees within ninety (90) Calendar Days after adopting the policy with respect to any service; and |
| 7.13.1.3 | Enrollees and Potential Enrollees before and during Enrollment. |
| 7.13.2 | The Contractor acknowledges that such objection will be grounds for recalculation of the rates paid to the Contractor. |
ARTICLE 8 INTEGRATION OF PHYSICAL AND BEHAVIORAL HEALTH SERVICES
| 8.1 | General Provisions |
| 8.1.1 | The Contractor shall ensure that physical and Behavioral Health Services are fully integrated, to ensure optimal detection, prevention, and treatment of physical and Behavioral Health illness. |
| 8.1.2 | The Contractor (through contracted PCPs, PMGs, and other Network Providers) shall be responsible, for identifying Enrollees’ needs and coordinating proper Access to both physical and Behavioral Health Services. |
| 8.1.3 | In implementing an integrated model of service delivery, the Contractor shall observe all the protections of the Mental Health Code (Act No. 408) and the Puerto Rico Patient’s Bill of Rights Act, as well as other applicable Federal and Puerto Rico legislation. |
| 8.2 | Co-Location of Staff |
| 8.2.1 | The Contractor shall facilitate the placement of a psychologist or other type of Behavioral Health Provider in each PMG setting. The Behavioral Health Provider shall be present and available to provide assessment, consultation, and Behavioral Health Services to Enrollees. The standard minimum criteria for weekly access will be four (4) hours per week for every five thousand (5,000) beneficiaries assigned to a PMG Setting. The Contractor must comply with the ASES Guidelines for Co-location of the Behavioral Health Provider in PMG Settings detailed in Attachment 10 of this Contract. |
Page 116 of 301
| 8.2.2 | The Contractor shall ensure that the PMG provides adequate space and resources for the Behavioral Health Provider to provide care and consultations in a confidential setting. |
| 8.2.3 | The Behavioral Health Provider housed within the PMG shall conduct screening evaluations, crisis intervention, and limited psychotherapy (between four (4) and six (6) sessions, according to the needs of the Enrollee). The Contractor shall ensure that the services provided are compliant with Act No. 408. |
| 8.2.4 | In the event that a PMG does not allow Contractor to place a psychologist or other Behavioral Health Provider in the PMG setting for the minimum time required in Section 8.2.1, the Contractor shall notify ASES and request from them instructions on how to proceed with co-location logistics with respect to said PMG. |
| 8.3 | Reverse Co-location |
| 8.3.1 | The Contractor shall establish at least two (2) Short-term Intervention Centers (Stabilization Units) per Service Region except in the Northeast Service Region. If the Contractor serves the Northeast Service Region, the Contractor must have available at least four (4) Short term Intervention Centers (Stabilization Units) in the Northeast Service Region, including one (1) in Culebra and one (1) in Vieques. Based on the needs of the population, ASES has the sole discretion of requiring the Contractor to establish additional Behavioral Health Services Facilities in any of the Service Regions. |
| 8.3.2 | The Contractor must comply with the ASES Guidelines for Reverse Co-location of the Primary Care Physicians (PCP) in Behavioral Health Facilities detailed in Attachment 21 of this Contract. |
| 8.3.3 | The Contractor shall ensure that a PCP is on site or on call as specified in the Guidelines for Reverse Collocation (see Attachment 21) to monitor the physical health of the Enrollees. |
| 8.3.4 | The Contractor shall ensure that the Behavioral Health Facility provides adequate space and resources for the PCP to provide care and consultations in a confidential setting. |
| 8.3.5 | In the event that a Behavioral Health Facility does not allow Contractor to place a PCP in the Behavioral Health Facility for the minimum time required, the Contractor shall proceed according to the Guidelines for Reverse Collocation (see Attachment 21). |
Page 117 of 301
| 8.4 | Referrals |
| 8.4.1 | GHP Enrollees with chronic or severe behavioral health conditions, which require more intensive or continuous care than can be provided within the PMG environment as set forth in Section 8.2, shall be referred to a Behavioral Health Provider for on-going services. In the same way, enrollees who require more intensive or continuous care than can be provided within the Behavioral Health Facility as set forth in Section 8.3 shall be referred to the appropriate physical provider for level of treatment. |
| 8.4.2 | An Enrollee may access Behavioral Health Services through the following means: |
| 8.4.2.1 | A Referral from the PCP or other PMG physician; |
| 8.4.2.2 | Self-referral (walk-in); |
| 8.4.2.3 | The GHP Service Line Service; |
| 8.4.2.4 | The telephone Call Center provided by ASSMCA, known as “Linea PAS”; |
| 8.4.2.5 | Hospitals; and |
| 8.4.2.6 | Emergency rooms. |
| 8.5 | Information Sharing |
| 8.5.1 | To the extent the Contractor utilizes a Subcontractor to provide Behavioral Health services, the Contractor and the Subcontractor shall share documents in their respective possession (including agreements, processes, guidelines and clinical protocols), in order to understand the other’s operations to ensure optimal cooperation and integration of physical and Behavioral Health Services. |
| 8.5.2 | The Contractor shall develop forms to facilitate electronic communication between physical health and Behavioral Health Providers, such as: |
| 8.5.2.1 | An information sheet for Enrollees on HIPAA requirements; |
| 8.5.2.2 | A Referral sheet; and |
| 8.5.2.3 | An informed consent form. |
| 8.5.3 | The Contractor shall establish a process for monitoring exchange of Information, documenting receipt of Information and following up on Information not submitted in a timely manner. |
Page 118 of 301
| 8.5.4 | The Contractor shall require PMG staff to follow-up with Behavioral Health Providers concerning the care of Enrollees referred by the PMG to a Behavioral Health Provider. |
| 8.5.5 | The Contractor shall ensure that the providers implement a certified EHR and a HIE platform with the capacity of centralizing the management of the referred EHR for all the PMG Providers including all Behavioral Health Providers. |
| 8.6 | Staff Education |
| 8.6.1 | The Contractor shall train PMG and the Behavioral Health Facility staff on the goals and operational details of the integrated model of care, and, as appropriate, the identification of Behavioral Health issues and conditions. |
| 8.6.2 | The Contractor shall require PMGs to Immediately refer Enrollees to the Behavioral Health Provider located within the PMG (or, if the Provider is not available, to the emergency room) when an Enrollee displays suicidal behavior. |
| 8.7 | Cooperation With Puerto Rico and Federal Government Agencies |
The Contractor shall ensure that governmental entities, including ASSMCA and SAMHSA, shall be consulted where appropriate and shall acknowledge that these entities participate, as appropriate, in the regulation of Behavioral Health Services under the GHP.
| 8.8 | Integration Plan |
The Contractor shall submit to ASES, for its review and approval, an Integration Plan incorporating the elements in this Article 8, according to the timeframe specified in Attachment 12 to this Contract. The plan shall cover at a minimum:
| 8.8.1 | How (1) reverse co-location and (2) co-location will be arranged, implemented, and monitored; |
| 8.8.2 | Target dates for full compliance with reverse co-location and co-location; |
| 8.8.3 | Contingency plans for PMGs and Behavioral Health Facilities who do not have appropriate space for co-location or reverse co-location or refuse to participate; |
| 8.8.4 | How Referrals are communicated, implemented, and tracked; and |
| 8.8.5 | Schedule for staff education and measurement of compliance. |
Page 119 of 301
ARTICLE 9 PROVIDER NETWORK
| 9.1 | General Provisions |
| 9.1.1 | The Contractor shall comply with the requirements specified in 42 C.F.R. §438.207(c), §438.214 and all applicable Puerto Rico requirements regarding Provider Networks. The Contractor shall have policies and procedures that reflect these requirements that are prior approved in writing by ASES in accordance with the timeframes in Attachment 12. The Contractor shall also: |
| 9.1.1.1 | Establish and maintain a comprehensive network of Providers capable of serving all Enrollees who enroll in the Contractor’s MCO; |
| 9.1.1.2 | Pursuant to Section 1932(b)(7) of the Social Security Act, not discriminate against Providers that serve high-risk populations or specialize in conditions that require costly treatment; |
| 9.1.1.3 | Not discriminate with respect to participation, reimbursement, or indemnification of any Provider acting within the scope of that Provider’s license or certification under applicable Puerto Rico law solely on the basis of the Provider’s license or certification; |
| 9.1.1.4 | Upon declining to include a Provider or group of Providers that have requested inclusion in the Contractor’s General Network, the Contractor shall give the affected Provider(s) written notice of the reason for its decision; |
| 9.1.1.5 | Be allowed to negotiate different reimbursement amounts for different specialties or for different practitioners in the same specialty; |
| 9.1.1.6 | Be allowed to establish measures that are designed to maintain quality of services and control of costs and are consistent with its responsibility to Enrollees; |
| 9.1.1.7 | Not make payment to any Provider who has been barred from participation based on existing Medicare, Medicaid or CHIP sanctions, except for Emergency Services; and |
| 9.1.1.8 | Provide Enrollees with special health care needs direct Access to a specialist, as appropriate for the Enrollee’s health care condition, as specified in 42 CFR § 438.208(c)(4). |
| 9.1.2 | The Contractor shall have an adequate network of available Providers meeting all Contract requirements in order to (i) ensure timely Access to Covered Services (including complying with all Federal and Puerto Rico requirements concerning timeliness, amount, duration, and scope of services); and (ii) provide sufficient Network Providers to satisfy the demand of Covered Services with adequate capacity and quality service delivery. |
Page 120 of 301
| 9.1.3 | When establishing and maintaining an adequate network of Providers, the Contractor shall consider and comply with each of the following criteria, in accordance with 42 CFR 438.206(b)(1): |
| 9.1.3.1 | Estimated eligible population and number of Enrollees; |
| 9.1.3.2 | Estimated use of services, considering the specific characteristics of the population and special needs for physical and Behavioral Health care; |
| 9.1.3.3 | Integration of physical health services and Behavioral Health Services using state facilities, academic medical centers, municipal health services and facilities; |
| 9.1.3.4 | Number and type of Providers required to offer services taking experience, training, and specialties into account; |
| 9.1.3.5 | Maximum number of patients per Provider; |
| 9.1.3.6 | The number of Providers in the PPN and General Network that are not accepting new patients; and |
| 9.1.3.7 | Geographic location of Providers and Enrollees, taking into account distance as permitted by law, the duration of trip, the means of transportation commonly used by Enrollees, and whether the facilities provide physical access for Enrollees with physical disabilities or special needs. |
| 9.1.4 | If the Contractor declines to include individual or groups of Providers in its network, it must give the affected Providers written notice of the reason for its decision. 42 CFR 438.12 (a) may not be construed to: |
| 9.1.4.1 | Require the Contractor to contract with Providers beyond the number necessary to meet the needs of its Enrollees; |
| 9.1.4.2 | Preclude the Contractor from using different reimbursement amounts for different specialties or for different practitioners in the same specialty; or |
| 9.1.4.3 | Preclude the Contractor from establishing measures that are designed to maintain quality of services and control costs and is consistent with its responsibilities to Enrollees. |
| 9.1.5 | The provider’s facilities must comply with Federal and Puerto Rico laws regarding the physical condition of medical facilities, the Provider’s facilities and must also comply with ASES’s requirements including, but not limited to, accessibility, cleanliness and proper hygiene. ASES reserves the right to evaluate the appropriateness of such facilities to provide the Covered Services. After receiving a written notice from ASES, the Contractor must timely notify the Provider, propose and enforce a corrective plan to be completed within ninety (90) Calendar Days to make the facilities appropriate to provide the Covered Services. |
Page 121 of 301
| 9.1.5.1 | The Contractor shall collaborate with the Providers to provide integrated GHP physical and Behavioral Health Services in order to achieve a fully integrated and holistic approach to providing Enrollee care. |
| 9.1.5.2 | The Contractor shall implement procedures in conjunction with the Providers to ensure that each GHP Enrollee has Access to both physical and Behavioral Health outpatient and inpatient services. |
| 9.1.5.3 | The Contractor shall develop policies and procedures that ensure timely Access to physical and Behavioral Health Services and integration of care. |
| 9.1.5.4 | The Contractor shall submit its policies and procedures to ASES for prior written approval according to the timeframe set forth in Attachment 12 to this Contract |
| 9.1.5.5 | If available in the Service Region, the Contractor must sub-contract ASSMCA to be a Behavioral Health Services provider. |
| 9.1.6 | The Contractor’s Network shall not include a Provider if the Provider, or any person who has an ownership or controlling interest in the Provider, or is an agent or managing employee of the Provider, is included on the List of Excluded Individuals/Entities (“LEIE”) (which is maintained by HHS-OIG), or who are on the Excluded Parties List System (“EPLS”) or on Puerto Rico’s list of excluded Providers. The Contractor shall check LEIE and EPLS upon the Provider’s Enrollment, Re-Enrollment, and on a monthly basis. Upon enrollment and Re-Enrollment the Contractor must also check the SSA Death Master File to make sure the Provider is not deceased and the National Plan and Provider Enumeration System (“NPPES”) to make sure the Provider has a NPI as required in Section 9.1.7. |
| 9.1.7 | The Contractor shall require that each Provider have a unique National Provider Identifier (“NPI”). |
| 9.1.8 | Ambulatory clinics shall have a sufficient number of Providers to efficiently and promptly provide Behavioral Health Services to Enrollees visiting such clinics. Contractor shall provide ASES with a report within the first ten (10) Business Days of each month indicating the number of Enrollees (including walk-ins) receiving Behavioral Health Services therein and the number of Providers providing these services during such period. |
Page 122 of 301
| 9.1.9 | ASES shall have the right to previously approve Contractor’s clinical protocols to render behavioral health services and substance abuse to Enrollees in ambulatory care. |
| 9.1.10 | In the event that a determined type of Provider cannot be contracted by the Contractor due to lack of such Providers in the Service Region or due to such Provider’s refusal to contract for this GHP Program, the Contractor must carry out all efforts to contract with those Providers within contiguous regions; provided that before resorting to contiguous regions the Contractor must validate and submit all supporting documents evincing the lack of Providers or refusal to contract to ASES. ASES will make a determination based on the evidence submitted if any further action is required of the Contractor. |
| 9.1.11 | The Contractor is responsible for establishing and monitoring Medical Record guidelines which include documentation of all services provided by the Primary Care Providers as well as any participating Providers in the contracted Provider Network. |
| 9.1.12 | Direct Relationship |
| 9.1.12.1 | The Contractor shall ensure that all Network Providers have knowingly and willingly agreed to participate in the Contractor’s General Network. |
| 9.1.12.2 | The Contractor shall be prohibited from acquiring established networks without contacting each individual Provider to ensure knowledge of the requirements of this Contract and to confirm the Provider’s complete understanding and agreement to fulfill all terms of the Provider Contract. |
| 9.1.12.3 | ASES reserves the right to confirm and validate, through collection of information, documentation from the Contractor and on-site visits to Network Providers, the existence of a direct relationship between the Contractor and the Network Providers. |
| 9.1.13 | Contractor Documentation of Adequate Capacity and Services |
| 9.1.13.1 | Before the Effective Date of this Contract and Immediately upon request by ASES after April 1, 2015, (Implementation Date of the Contract), the Contractor shall provide documentation demonstrating that it: |
Page 123 of 301
| 9.1.13.1.1 | Offers an appropriate range of assessment and treatment, preventive, Primary Care, and specialty services that is adequate for the anticipated number of Enrollees in each Service Region; and |
| 9.1.13.1.2 | Maintains a Provider Network that is sufficient in number, mix, and geographic distribution to meet the needs of the anticipated number of Enrollees in each Service Region. |
| 9.1.13.2 | The Contractor shall provide documentation of the Network adequacy conditions stated in this Section 9.1 Immediately, any time that there has been a significant change in the Contractor’s operations that would affect adequate capacity and services, including: |
| 9.1.13.2.1 | When there is a change in Benefits, the geographic Service Region, or payments; or |
| 9.1.13.2.2 | Upon the Enrollment of a new eligibility group in the Contractor’s Plan. |
| 9.2 | Provider Qualifications |
| 9.2.1 | The following requirements apply to Network Providers in the Contractor’s : |
|
FQHC
Federal Qualified Health Centers
|
A Federally Qualified Health Center is an entity that provides outpatient care under Section 330 of the Public Health Service Act (42 U.S.C. 254b) and complies with the standards and regulations established by the Federal government and is an eligible Provider enrolled in the Medicaid Program.
|
|
PHYSICIAN
|
A person with a license to practice medicine as an M.D. or a D.O. in Puerto Rico, whether as a PCP or in the area of specialty under which he or she will provide medical services through a contract with the Contractor; and is a Provider enrolled in the Puerto Rico Medicaid Program; and has a valid registration number from the Drug Enforcement Agency and the Certificate of Controlled Substances of Puerto Rico, if required in his or her practice.
|
Page 124 of 301
|
HOSPITAL
|
An institution licensed as a general or special hospital by the Puerto Rico Health Department under Chapter 241 of the Health and Safety Code of Private Psychiatric Hospitals under Chapter 577 of the Health and Safety Code (or who is a Provider which is a component part of the Puerto Rico or local governmental entity which does not require a license under the laws of the Commonwealth) which is enrolled as a Provider in the Puerto Rico Medicaid Program.
|
|
NON-MEDICAL PRACTICING PROVIDER
|
A person who possesses a license issued by the licensing agency of the Commonwealth enrolled in the Puerto Rico Medicaid Program or a properly trained person who practices under the direct supervision of a licensed Provider offering support in health care services.
|
|
CLINICAL LABORATORY
|
An entity that has a valid certificate issued by the Clinical Laboratory Improvement Act (“CLIA”) and which has a license issued by the licensing agency of the Commonwealth. The Contractor shall ensure that all of the clinical laboratories under contract have a CLIA registration certificate and the registration number or a waiver certificate.
|
|
RURAL HEALTH CLINIC (RHC)
|
A health facility that the Secretary of Health and Human Services has determined meets the requirements of Section 1861(a)(2) of the Social Security Act; and that has entered into an agreement with the Secretary to provide services in Rural Health Clinics or Centers under Medicare and in accordance with 42 CFR 405.2402.
|
|
LOCAL HEALTH DEPARTMENT
|
Local Health Department established under Act 81 from March 14, 1912.
|
|
NON-HOSPITAL PROVIDING FACILITY
|
A Provider which is duly licensed and credentialed to provide services and is enrolled in the Puerto Rico Medicaid Program.
|
|
SCHOOLS OF MEDICINE
|
Clinics located on the medical campus that provides Primary Care and Preventive Services to children and adolescents.
|
Page 125 of 301
|
MEDICAL PSYCHIATRIST
|
A person who possesses a license to practice medicine and a psychiatrist specialty license issued by the licensing agency of the Commonwealth and is enrolled in the Puerto Rico Medicaid Program or a properly trained person who practices psychiatry under the direct supervision of a licensed Provider.
|
|
PSYCHOLOGIST
|
A person who possesses a Doctoral or Master’s Degree and a license issued by the licensing agency of the Commonwealth and is enrolled in the Puerto Rico Medicaid Program or a properly trained person who practices psychology under the direct supervision of a licensed Provider.
|
|
SOCIAL WORKER
|
A person who possesses a social work degree and a current license issued by the licensing agency of the Commonwealth and is enrolled in the Puerto Rico Medicaid Program or a properly trained person who practices social work under the direct supervision of a licensed Provider.
|
|
DETOXIFICATION FACILITY
|
An entity or health facility that has a valid certificate and license to provide detoxification treatments issued by the licensing agency of the Commonwealth.
|
|
SHORT TERM INTERVENTION CENTER (Sala Estabilizadora)
|
An entity or health facility that has a valid certificate and license to provide Behavioral Health Services issued by the licensing agency of the Commonwealth.
|
| 9.2.2 | The Contractor shall also ensure that Network Providers meet any other qualifications as prescribed by ASES. |
| 9.2.3 | Provider Credentialing |
| 9.2.3.1 | The Contractor shall be responsible for Credentialing and Re-Credentialing its Network Providers. |
| 9.2.3.2 | The Contractor shall ensure that all Network Providers are appropriately credentialed and qualified to provide services under the terms of this Contract, all applicable Federal and Puerto Rico law, and comply with CMS Credentialing requirements included in CMS Chapter VI of the Medicare Managed Care Manual. |
| 9.2.3.3 | ASES strongly encourages Contractors to implement a Credentialing Electronic Record System. ASES reserves the right to request access to the Contractor’s Credentialing Electronic Record Systems for monitoring purposes. |
Page 126 of 301
| 9.2.3.4 | The Contractor shall contract with all available Providers that meet its Credentialing process and agree to its contractual terms, in order to ensure sufficient Network Providers to address Enrollee needs. |
| 9.2.3.5 | Credentialing is required for: |
| 9.2.3.5.1 | All physicians who provide services to the Contractor’s Enrollees, |
| 9.2.3.5.2 | All other types of Providers who provide services to the Contractor’s Enrollees, and who are permitted to practice independently under Puerto Rico law including but not limited to: hospitals, X-ray facilities, clinical laboratories, and ambulatory service Providers. |
| 9.2.3.6 | Credentialing is not required for: |
| 9.2.3.6.1 | Providers who are permitted to furnish services only under the direct supervision of another practitioner; |
| 9.2.3.6.2 | Hospital-based Providers who provide services to Enrollees Incident to hospital services, unless those Providers are separately identified in Enrollee literature as available to Enrollees; or |
| 9.2.3.6.3 | Students, residents, or fellows. |
| 9.2.3.7 | Standards for Credentialing and Re-Credentialing |
| 9.2.3.7.1 | The Contractor shall document the mechanism for Credentialing and Re-Credentialing of Network Providers or Providers it employs to treat Enrollees outside of the inpatient setting and who fall under its scope of authority and action. This documentation shall include, but not be limited to, defining the scope of Providers covered, the criteria and the primary source verification of Information used to meet the criteria, the process used to make decisions that shall not be discriminatory and the extent of delegated Credentialing and Re-Credentialing arrangements. The Contractor shall: |
| 9.2.3.7.1.1 | Have written policies and procedures for the Credentialing and Re-Credentialing process. Such process must permit providers to apply for Credentialing and Re-Credentialing online; |
| 9.2.3.7.1.2 | Meet Puerto Rico and Federal regulations for Credentialing and Re-Credentialing, including 42 C.F.R. §§ 455.104, 455.105, 455.106 and 1002.3(b); |
Page 127 of 301
| 9.2.3.7.1.3 | Use one (1) standard Credentialing form prescribed by ASES; |
| 9.2.3.7.1.4 | Designate a Credentialing committee or other peer review body to make recommendations regarding Credentialing/Re-Credentialing issues; |
| 9.2.3.7.1.5 | Complete the Credentialing process within forty-five (45) Calendar Days from receipt of completed application with all required primary source documentation; |
| 9.2.3.7.1.6 | Ensure Credentialing/Re-Credentialing forms require ownership and control disclosures, disclosure of business transactions, and criminal conviction information; |
| 9.2.3.7.1.7 | Verify that Network Providers maintain a current and valid license to practice. Verification must show that the license was in effect at the time of the Credentialing decision with a copy of a good standing; or with the Junta de Licenciamiento Médico / Junta de Profesionales de la Salud CD; |
| 9.2.3.7.1.8 | Ensure education and training records, including, but not limited to, Internship, Residency, Fellowships, Specialty Boards etc., are validated and current. As per CMS chapter VI, section 60, education verification is required only for the highest level of education or training attained; |
| 9.2.3.7.1.9 | Ensure board certification, when applicable, in each clinical specialty area for which the Provider is being credentialed; |
| 9.2.3.7.1.10 | Ensure clinical privileges are in good standing at the hospital designated by the Provider, when applicable, as the primary admitting facility. This information may be obtained by contacting the facility, obtaining a copy of the participating facility directory or attestation by the Provider; |
| 9.2.3.7.1.11 | Ensure Network Providers maintain current and adequate malpractice insurance. This information may be obtained via the malpractice carrier, a copy of the insurance face sheet or attestation by the Provider; |
Page 128 of 301
| 9.2.3.7.1.12 | Obtain Information about sanctions or limitations on licensure from the applicable Puerto Rico licensing agency or board, or from a group such as the Federation of State Medical Boards; |
| 9.2.3.7.1.13 | Ensure a valid Drug Enforcement Agency (“DEA") or Controlled Dangerous Substances (“CDS”) certificate in effect at the time of the Credentialing. This information can be obtained through confirmation with CDS, entry into the National Technical Information Service (“NTIS”) database, or by obtaining a copy of the certificate; |
| 9.2.3.7.1.14 | Review Network Provider’s history of professional liability claims that resulted in settlements or judgments paid by or on behalf of the Provider: This information can be obtained from the malpractice carrier or from the National Practitioner Data Bank; |
| 9.2.3.7.1.15 | Ensure that Behavioral Health Network Providers (as applicable) are trained and certified by the Substance Abuse and Mental Health Services Administration (“SAMHSA”) to provide the opiate addiction treatment certifications stated in Section 7.5.11.3; |
| 9.2.3.7.1.16 | Ensure Credentialing of health care facilities shall be governed by, but not limited to, Law 101 of June 26, 1965, as amended, known as "Law of Facilities of Puerto Rico;" |
| 9.2.3.7.1.17 | Screen all providers against the LEIE or Medicare Exclusion Databases monthly to ensure Providers are not employing or contracting with excluded individuals; |
| 9.2.3.7.1.18 | Have written policies and procedures, that have been prior approved in writing by ASES, to ensure and verify that providers have appropriate licenses and certifications to perform services outlined in their respective Provider agreements; and |
| 9.2.3.7.1.19 | Maintain records that verify its Credentialing and Re-Credentialing activities, including primary source verification and compliance with Credentialing/Re-Credentialing requirements. |
Page 129 of 301
| 9.2.3.7.2 | The Contractor shall perform the following functions: |
| 9.2.3.7.2.1 | Credential any Provider who contracts with the Contractor and maintaining complete Credentialing information for these Providers; |
| 9.2.3.7.2.2 | Identify potential and actual Network Providers who are enrolled with ASES as Medicaid Providers; |
| 9.2.3.7.2.3 | Require any Network Provider to be enrolled with the GHP as a managed care Provider; |
| 9.2.3.7.2.4 | Perform site visits. The organization’s site visit policy will be reviewed pursuant to CMS’ monitoring protocol. At a minimum, the organization should consider requiring initial Credentialing site visits of the offices of Primary Care practitioners, obstetrician- gynecologists, or other high-volume Providers, as defined by the organization; |
| 9.2.3.7.2.5 | Re-Credential Network Providers every three (3) years; |
| 9.2.3.7.2.6 | Ensure all required documents and licenses are current at the time of initial Credentialing or Re-Credentialing; |
| 9.2.3.7.2.7 | Maintain a Provider file for all Network Providers. The Provider file shall be updated annually and consist of, at a minimum, the following documents: annual Puerto Rico review, DEA license, malpractice insurance and ASSMCA license. |
| 9.2.3.7.2.8 | The Contractor shall ensure, and be able to demonstrate at the request of ASES, that: (i) Out-of-Network Providers have been credentialed by an authoritative entity and that (ii) the Contractor’s internal Credentialing and Re-Credentialing processes are in accordance with 42CFR 438.214 (Provider Selection). |
| 9.2.3.7.2.9 | If the Contractor determines, through the Credentialing or Re-Credentialing process, or otherwise, that a Provider could be excluded pursuant to 42 CFR 1001.1001, or if the Contractor determines that the Provider has failed to make full and accurate disclosures as required in Sections 13.5.13 above, the Contractor shall deny the Provider’s request to participate in the Provider Network, or, for a current Network Provider, as provided in Section [10.4.1.2], terminate the Provider Contract. The Contractor shall notify ASES of such a decision, and shall provide documentation of the bar on the Provider’s Network participation, within twenty (20) Business Days of communicating the decision to the Provider. The Contractor shall screen its employees, Network Providers, and Subcontractors initially and on an ongoing monthly basis to determine whether any of them have been excluded from participation in Medicare, Medicaid, CHIP, or any other Federal health care program (as defined in Section 1128B(f) of the Social Security Act). ASES or the Puerto Rico Medicaid Program shall, upon receiving notification from a plan that the plan has denied Credentialing, notify the HHS Office of the Inspector General of the denial with twenty (20) Business Days of the date it receives the Information, in conformance with 42 CFR 1002.3. |
Page 130 of 301
| 9.2.3.7.2.10 | The Contractor shall report to ASES on a monthly basis the Credentialing and Re-Credentialing status of Providers. The details of this report are described in Section 18.2.5.3 of this Contract. |
| 9.3 | Network Description |
| 9.3.1 | General Network |
| 9.3.1.1 | The General Network shall be comprised of all Providers available to Enrollees including those Providers who are designated as preferred providers and those Providers who are not associated with a PMG. |
| 9.3.1.2 | The Contractor shall ensure that its General Network of Providers is adequate to assure Access to all Covered Services, and that all Providers are appropriately credentialed, maintain current licenses, and have appropriate locations to provide the Covered Services. |
| 9.3.1.3 | The Contractor shall include and make a part of its General Network any Behavioral Health Provider who is qualified for the GHP (including meeting all Credentialing standards in this Contract) and willing to participate in the Network. |
Page 131 of 301
| 9.3.1.4 | If the Contractor’s General Network is unable to provide Medically Necessary Covered Services to an Enrollee within the timeframes set forth in Section 9.5, the Contractor shall adequately and timely (as defined in Section 9.5) cover these services using Providers outside of its Network without penalty or additional Co-Payments imposed on the Enrollee. |
| 9.3.1.5 | PCPs |
| 9.3.1.5.1 | PCPs will be responsible for providing, managing and coordinating all the services of the Enrollee, including the coordination with Behavioral Health personnel, in a timely manner, and in accordance with the guidelines, protocols, and practices generally accepted in medicine. |
| 9.3.1.5.2 | The Contractor shall offer its Enrollees freedom of choice in selecting a PCP. The Contractor shall have policies and procedures describing how Enrollees select their PCPs. The Contractor shall submit these policies and procedures to ASES for review and prior written approval according to the timeframes specified in Attachment 12 to this Contract. |
| 9.3.1.5.3 | The PCP is responsible for maintaining each Enrollee’s Medical Record, which includes documentation of all services provided by the PCP as well as any specialty services. |
| 9.3.1.5.4 | The following shall be considered PCPs for purposes of contracting with a PMG: |
| 9.3.1.5.4.1 | General practitioners; |
| 9.3.1.5.4.2 | Internists; |
| 9.3.1.5.4.3 | Family doctors; |
| 9.3.1.5.4.4 | Pediatricians (optional for minors under the age of twenty-one (21)); and |
| 9.3.1.5.4.5 | Gynecologists-Obstetricians (obligatory when the woman is pregnant or of reproductive age; this Provider will also be selected for usual gynecological visits). |
| 9.3.1.5.5 | No PCP may own any financial control or have a direct or indirect economic interest (as defined in Act 101 of July 26, 1965) in any Ancillary Services facility or any other Provider (including clinical laboratories, pharmacies, etc.) under contract with the PMG. |
Page 132 of 301
| 9.3.1.5.6 | Nurse practitioners and physician assistants may not be PCPs. |
| 9.3.1.5.7 | The Contractor shall guarantee that women who are pregnant select a gynecologist-obstetrician as their PCP. Additionally, the Contractor will permit female Enrollees to select a gynecologist-obstetrician for their routine gynecological visits at initial Enrollment. |
| 9.3.1.6 | Behavioral Health Providers |
| 9.3.1.6.1 | The Contractor shall have a sufficient number of Behavioral Health Providers within each Service Region attend to the Behavioral Health needs of the Enrollees. The Contractor shall make available all specialties specified in this Section 9.3.1.6. |
| 9.3.1.6.2 | The Contractor shall have available and under contract within each Service Region a sufficient number of the following types of Network Providers to render services to all Enrollees: |
| 9.3.1.6.2.1 | Psychiatrist; |
| 9.3.1.6.2.2 | Clinical or Counseling Psychologist; |
| 9.3.1.6.2.3 | Social Workers (“MSW”); |
| 9.3.1.6.2.4 | Care Managers; |
| 9.3.1.6.2.5 | Certified Addiction Counselors; and |
| 9.3.1.6.2.6 | Behavioral Health Facilities, as specified in Article 2. |
| 9.3.1.7 | Network Provider Types |
| 9.3.1.7.1 | For both the General Network and the PPN, the Contractor shall have available and under contract within each Service Region the following types of Network Providers: |
| 9.3.1.7.2 | Specialists: |
| 9.3.1.7.2.1 | Podiatrists, Optometrists, Ophthalmologists, Radiologists, Endocrinologists, Nephrologists, Pneumologists, Otolaryngologists (ENTs), Cardiologists, Urologists, Gastroenterologists, Rheumatologists, Dermatologists, Oncologists, Neurologists, Infectious Disease Specialists, Orthopedists, Physical & Rehabilitative Specialists(Physiatrist), General Surgeons, and Chiropractors. |
Page 133 of 301
| 9.3.1.7.2.2 | The Contractor shall offer its Enrollees freedom of choice in selecting a dentist. The Contractor shall include in its Provider Network any Provider that is qualified and willing to participate. |
| 9.3.1.7.2.3 | The Contractor shall offer its Enrollees freedom of choice in selecting Behavioral Health Providers. |
| 9.3.1.7.3 | Facilities |
| 9.3.1.7.3.1 | Clinical Laboratories; |
| 9.3.1.7.3.2 | X-Ray Facilities; |
| 9.3.1.7.3.3 | Hospitals; |
| 9.3.1.7.3.4 | Providers and facilities for Behavioral Health Services; |
| 9.3.1.7.3.5 | Specialized service Providers; |
| 9.3.1.7.3.6 | Urgent care centers and emergency rooms; and |
| 9.3.1.7.3.7 | Any other Providers or facilities needed to offer Covered Services, except pharmacies, considering the specific health needs of the Service Region. |
| 9.3.1.8 | Out-Of-Network Providers |
| 9.3.1.8.1 | If the Contractor’s General Network is unable to provide Medically Necessary Covered Services to an Enrollee, the Contractor shall adequately and timely (within the standards in Section 9.5) cover these services using Providers outside of its General Network. |
| 9.3.1.8.2 | Except as provided with respect to Emergency Services (see Section 7.5.9), if the Contractor offers the service through a Provider in the General Network but the Enrollee chooses to access the service from an Out-of-Network Provider, the Contractor is not responsible for payment. |
Page 134 of 301
| 9.3.1.8.3 | The Contractor must ensure that Out-of-Network Providers are duly credentialed and shall pay them, at a minimum, the same rates the Contractor pays its Network Providers dependent on provider type. |
| 9.3.1.9 | The Contractor shall not restrict the choice of the Provider from whom an Enrollee may receive family planning services and supplies. |
| 9.3.1.10 | The Contractor shall provide female Enrollees with direct access to a women’s health specialist within the General Network for Covered Services necessary to provide women's routine and preventive health care services in addition to the Enrollee's designated source of primary care if that source is not a woman's health specialist. |
| 9.3.2 | ASES shall ensure, in collecting Co-Payments, that in the event that a Co-Payment is imposed on Enrollees for an Out-of-Network service, the Co-Payment shall not exceed the Co-Payment that would apply if services were provided by a Provider in the General Network. |
| 9.3.3 | The Contractor shall also develop, as a subset of its General Network of Providers, a Preferred Provider Network (“PPN”). The objectives of the PPN model are to increase access to Providers and needed services, improve timely receipt of services, improve the quality of Enrollee care, enhance continuity of care, and facilitate effective exchange of Personal Health Information between Providers and the Contractor. See diagram provided as Attachment 20. |
| 9.3.3.1 | The PPN is established utilizing a PMG to deliver services to the Enrollees who select a PCP that is a member of an individual PMG. |
| 9.3.3.2 | The Contractor shall offer a PPN to all Enrollees. Each provider in the PPN shall be associated with an individual PMG whose group includes PCPs, clinical laboratories, x-ray facilities, specialists and other providers that meet network requirements described in this section. |
| 9.3.3.3 | Enrollees shall be allowed to receive services from all Providers within their PMG’s PPN without Referral or restriction. |
| 9.3.3.4 | Enrollees who receive a prescription from a Network Provider within the PPN/PMG shall be allowed to fill the prescription without the requirement of a Countersignature from their PCP. |
| 9.3.3.5 | Additional Preferred Provider Network (“PPN”) Standards |
Page 135 of 301
| 9.3.3.5.1 | The Contractor shall establish policies and procedures that, at a minimum, include: |
| 9.3.3.5.1.1 | Criteria for participating in the PPN versus the General Network; |
| 9.3.3.5.1.2 | Standards for monitoring Provider performance; |
| 9.3.3.5.1.3 | Methodologies for measuring Access to care; |
| 9.3.3.5.1.4 | Methodologies for identifying compliance issues; and |
| 9.3.3.5.1.5 | Measures to address identified compliance issues. |
| 9.3.3.5.2 | The Contractor shall submit its policies and procedures to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 9.4 | Provider Network Ratios |
| 9.4.1 | The Contractor shall comply with the following minimum Provider ratios for the General Network: |
| 9.4.1.1 | One (1) PCP per one thousand seven hundred (1,700) Enrollees in each Service Region; |
| 9.4.1.2 | One (1) Gynecologist (selected as the Enrollee’s PCP) per two thousand eight hundred 2,800 Enrollees (1:2,800) in each Service Region; |
| 9.4.1.3 | The specialist network is measured as a combined whole and must include a minimum of (1) specialist per two thousand two hundred (2,200) Enrollees in each Service Region; |
| 9.4.1.3.1 | Each Contract year, the Contractor shall submit for review and prior approval by ASES a Specialist Recruitment and Retention Plan for further ensuring the adequacy of the specialist providers considered critical. The annual plan shall include, but not be limited to, activities the Contractor will undertake to identify deficiencies, including but not limited to, Enrollee complaints regarding access, significant reductions in the number of specialist providers after transition is complete, and denied provider requests for inclusion in network. In addition the plan shall describe how the Contractor will increase the number and variety of specialists to meet the needs of the Enrollees. |
Page 136 of 301
| 9.4.1.4 | The Contractor shall provide adequate access to Enrollees at all times and are subject to the appointment requirements described in Section 9.5.1; |
| 9.4.1.5 | Critical specialist Providers include: |
| 9.4.1.5.1 | Cardiologists; |
| 9.4.1.5.2 | Gastroenterologists; |
| 9.4.1.5.3 | Pneumologists; |
| 9.4.1.5.4 | Endocrinologists; and |
| 9.4.1.5.5 | Urologists; |
| 9.4.1.6 | One (1) dentist per one thousand three hundred and fifty (1,350) Enrollees (1:1,350) in each Service Region. If there are not enough dentists in the Service Region, the Contractor must contract with dentists within contiguous Service Regions; |
| 9.4.1.7 | One (1) X-ray facility per ten thousand (10,000) Enrollees (1:10,000) in each Service Region; |
| 9.4.1.8 | One (1) clinical laboratory per five thousand (5,000) Enrollees (1:5,000) in each Service Region; |
| 9.4.1.9 | Two (2) hospitals in each Service Region; |
| 9.4.1.10 | One (1) psychiatrist per five thousand (5,000) Enrollees (1:5,000) in each Service Region; |
| 9.4.1.11 | One (1) psychologist per five thousand (5,000) Enrollees (1:5,000) in each Service Region; |
| 9.4.1.12 | Short term intervention centers (Salas Estabilizadoras) as described in Article 8. If a facility is not available in the region, the Contractor must contract with a facility in a contiguous region; |
| 9.4.1.13 | One (1) center for detoxification of alcohol and controlled substances in each Service Region. If a facility is not available in the region, the Contractor must contract with a facility in a contiguous region; and |
| 9.4.1.14 | One (1) provider duly trained and certified by SAMHSA for the treatment of opiate addiction in each Service Region. |
| 9.4.2 | The Contractor shall comply with the following minimum Provider ratios for the Preferred Provider Network (“PPN”) within each PMG: |
Page 137 of 301
| 9.4.2.1 | One (1) PCP per one thousand seven hundred (1,700) Enrollees Region (1:1,700) in each Service Region; |
| 9.4.2.2 | One (1) Gynecologist (selected as the Enrollee’s PCP) per two thousand eight hundred (2,800) Enrollees (1:2,800) in each Service Region; |
| 9.4.2.3 | The specialist network is measured as a combined whole and must include a minimum of (1) specialist per two thousand two hundred (2,200) Enrollees in each Service Region. |
| 9.4.2.4 | Each Contract year, the Contractor shall submit for review and prior approval by ASES a Specialist Recruitment and Retention Plan for further ensuring the adequacy of the specialist providers considered critical. The annual plan shall include but not be limited to, activities the Contractor will undertake to identify deficiencies including but not limited to; Enrollee complaints regarding access, significant reductions in the number of specialist providers after transition is complete, and denied provider requests for inclusion in network. In addition the plan shall describe how the Contractor will increase the number and variety of specialists to meet the needs of the Enrollees. |
| 9.4.2.5 | The Contractor shall provide adequate access to Enrollees at all times and are subject to the appointment requirements described in Section 9.5.1 |
| 9.4.2.6 | Critical specialist Providers include: |
| 9.4.2.6.1 | Cardiologists; |
| 9.4.2.6.2 | Gastroenterologists; |
| 9.4.2.6.3 | Pneumologists; |
| 9.4.2.6.4 | Endocrinologists; and |
| 9.4.2.6.5 | Urologists; |
| 9.4.2.7 | One (1) X-ray facility per ten thousand (10,000) Enrollees (1:10,000) in each Service Region; |
| 9.4.2.8 | Each PMG shall be considered a PPN based on the number of Enrollees who have selected a PCP associated with the individual PMG. The Contractor shall ensure that an individual PMG meets the ratio requirements listed in this Section 9.4.2 independently. |
| 9.4.3 | In the event that this ratio cannot be achieved by the Contractor due to lack of Providers of a determined specialty in the ratios set forth for each Service Region or due to specialists' refusal to contract as part of the PPN for the Service Region, the Contractor must carry out all efforts to contract with those specialists within contiguous Service Regions; provided that before resorting to contiguous regions Contractor must validate and submit all supporting documents evincing the lack of Providers or refusal to contract in accordance with Attachment 15 of this Contract. ASES shall approve that specialist's contract before its execution, after the Contractor has accredited such need with supporting documents. |
Page 138 of 301
| 9.4.4 | The Parties acknowledge that there are shortages of certain specialists in the Service Regions. The Contractor will work with the Provider community to address Enrollee Access to specialists to the extent possible. The Contractor will then develop policies and procedures to be prior approved in writing by ASES to ensure Enrollees have Access to specialty services as necessary. |
| 9.4.5 | If the PMG is unable to meet PPN ratio requirements, the Enrollee shall be permitted to seek services outside the PMG without penalty or co-payments. |
| 9.4.6 | Subject to Section 9.4 of this Contract, the aforementioned ratios must be maintained for Enrollees, regardless of whether the Contractor offers treatment to other private patients. |
| 9.5 | Access |
| 9.5.1 | Appointment Standards and Minimum Requirements for Access to Providers |
| 9.5.1.1 | The Contractor shall provide Access to Covered Services in accordance with the following terms: |
| 9.5.1.1.1 | Non-Urgent Conditions |
| 9.5.1.1.1.1 | Routine physical exams shall be provided for Enrollees age twenty-one (21) and over within thirty (30) Calendar Days of the Enrollee’s request for the service, taking into account both the medical and Behavioral Health need and condition. For minors less than twenty-one (21) years of age routine physical exams shall be provided within the timeframes specified in Section 7.9.3 of this Contract and in accordance with Act No. 408. |
| 9.5.1.1.1.2 | Routine evaluations for Primary Care shall be provided within thirty (30) Calendar Days, unless the Enrollee requests a later time; |
Page 139 of 301
| 9.5.1.1.1.3 | Covered Services shall be provided within fourteen (14) Calendar Days following the request for service; |
| 9.5.1.1.1.4 | Specialist Services shall be provided within thirty (30) Calendar Days of the Enrollee’s original request for service; |
| 9.5.1.1.1.5 | Dental services shall be provided within sixty (60) Calendar Days following the request, unless the Enrollee requests a later date; |
| 9.5.1.1.1.6 | Behavioral Health Services shall be provided within fourteen (14) Calendar Day following the request, unless the Enrollee requests a later date; |
| 9.5.1.1.1.7 | Diagnostic laboratory, diagnostic imaging and other testing appointments shall be provided consistent with the clinical urgency, but no more than fourteen (14) Calendar Days, unless the Enrollee requests a later time; |
| 9.5.1.1.1.8 | Diagnostic laboratory, diagnostic imaging and other testing, if a “walk-in” rather than an appointment system is used, the Enrollee wait time shall be consistent with severity of the clinical need; and |
| 9.5.1.1.1.9 | The in-person prescription fill time (ready for pickup) shall be no longer than forty (40) minutes. A prescription phoned in by a practitioner shall be filled within ninety (90) minutes. ASES highly recommends that the Providers contracted by the Contractor implement an electronic prescribing system; |
| 9.5.1.1.2 | Urgent Conditions |
| 9.5.1.1.2.1 | Emergency Services shall be provided, including Access to an appropriate level of care, within twenty-four (24) hours of the service request; |
| 9.5.1.1.2.2 | Primary medical, dental, and Behavioral Health Care outpatient appointments for urgent conditions shall be available within twenty-four (24) hours; |
| 9.5.1.1.2.3 | Urgent outpatient diagnostic laboratory, diagnostic imaging and other testing, appointment availability shall be consistent with the clinical urgency, but no longer than forty-eight (48) hours; |
Page 140 of 301
| 9.5.1.1.2.4 | Behavioral Health crisis services, face-to-face appointments shall be available within two (2) hours; and |
| 9.5.1.1.2.5 | Detoxification services shall be provided Immediately according to clinical necessity; |
| 9.5.1.1.3 | The timing of scheduled follow-up outpatient visits with practitioners shall be consistent with the clinical need; and |
| 9.5.1.1.4 | FQHC Services shall be provided in an FQHC setting. The Contractor shall adequately and timely cover these services out-of-network at no cost to Enrollees for as long as the FQHC Services are unavailable in the Contractor’s General Network. All out-of-network services require a Referral from the Enrollee’s PCP. |
| 9.5.2 | Access to Services for Enrollees with Special Health Needs |
| 9.5.2.1 | The Contractor shall require that its Network Providers evaluate any progressive condition of an Enrollee with special health needs that requires a course of regular monitored care or treatment. This evaluation will include the use of Providers for each identified case. |
| 9.5.2.2 | The Contractor shall establish a protocol to screen Enrollees for Special Coverage and for the Care Management and Disease Management Benefits, in order to facilitate direct Access to specialists. The Contractor shall submit its operational protocol to ASES for prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 9.5.3 | Hours of Service |
| 9.5.3.1 | The Contractor shall prohibit its Network Providers from having different hours and schedules for GHP Enrollees than what is offered to commercial Enrollees. |
| 9.5.3.2 | The Contractor shall prohibit its Providers from establishing specific days for the delivery of Referrals and requests for Prior Authorization for GHP Enrollees, and the Contractor shall monitor compliance with this rule and take corrective action if there is failure to comply. |
| 9.5.3.3 | The Contractor shall require Behavioral Health Facilities to have open service hours covering twelve (12) hours per day, seven (7) days per Week and shall have available one (1) nurse, one (1) social worker and one (1) psychologist/psychiatrist. |
Page 141 of 301
| 9.5.4 | Preferential Turns |
| 9.5.4.1 | The Contractor shall agree to establish a system of Preferential Turns for residents of the island municipalities of Vieques and Culebra. Preferential Turns refers to a policy of requiring Providers to give priority in treating Enrollees from these island municipalities, so that they may be seen by a physician within a reasonable time after arriving in the Provider’s office. This priority treatment is necessary because of the remote locations of these municipalities, and the greater travel time required for the residents to seek medical attention. This requirement was established in Laws No. 86 enacted on August 16, 1997 (Arts. 1 through 4) and Law No. 200 enacted on August 5, 2004 (Arts. 1 through 5). The Contractor shall include this requirement in the Provider Guidelines (see Section 10.2.1.4). |
| 9.5.5 | Extended Schedule of PMGs |
| 9.5.5.1 | PMGs shall be available to provide primary care services or consultations Monday through Saturday of each Week, from 8:00 a.m. to 6:00 p.m. The following Holidays the PMG will not have to comply with this requirement: January 1st, January 6Th, Good Friday, Thanksgiving Day and December 25th. The PMG has the sole discretion to decide whether or not to provide primary care services during the previously listed holidays. |
| 9.5.5.2 | In addition, each Provider that offers urgent care services, as well as any other qualified Provider willing to provide urgent care services, shall have sufficient personnel to offer urgent care services during extended periods Monday through Friday from 6:00 p.m. to 9:00 p.m. (Atlantic Time), in order to provide Enrollees greater Access to their PCPs and to urgent care services in each Service Region. |
| 9.5.5.3 | PMGs may collaborate with each other to establish extended office hours at one (1) or multiple facilities. |
| 9.5.5.4 | The Contractor shall submit to ASES its policies and procedures for how it will determine the adequacy and appropriateness of Providers’ available hours, approve such arrangements, and monitor their operation and take corrective action if there is failure to comply. The policies and procedures shall be submitted for prior approval according to the timeframe specified in Attachment 12 to this Contract. |
Page 142 of 301
| 9.5.6 | Provider Services Call Center |
| 9.5.6.1 | The Contractor shall operate a Provider services call center with a separate toll-free telephone line to respond to Provider questions, comments, inquiries and requests for prior authorizations. |
| 9.5.6.2 | The Contractor shall develop Provider service line policies and procedures that address staffing, training, hours of operation, access and response standards, monitoring of calls via recording or other means, and compliance with standards. Such policies and procedures shall be prior approved in writing by ASES. |
| 9.5.6.3 | The Contractor shall ensure that the Provider service line is staffed adequately to respond to Providers’ questions at a minimum from 7 a.m. to 7 p.m. Atlantic Time, Monday through Friday, excluding Puerto Rico holidays. |
| 9.5.6.4 | The Contractor shall have an automated system available during non-business hours. This automated system shall include, at a minimum, information on how to obtain after hours UM requests and a voice mailbox for callers to leave messages. The Contractor shall ensure that the automated system has adequate capacity to receive all messages. The Contractor shall return messages on the next Business Day. |
| 9.5.6.5 | The Provider service line shall also be adequately staffed to provide appropriate and timely responses regarding authorization requests as described in Article 11 of this Contract. The Contractor may meet this requirement by having a separate Utilization Management line. |
| 9.5.6.6 | The call center staff shall have access to electronic documentation from previous calls made by a Provider. |
| 9.5.6.7 | The Contractor shall adequately staff the provider service line to ensure that the line, including the Utilization Management line/queue, meets the following minimum performance standards on a monthly basis: |
| 9.5.6.7.1 | Less than five percent (5%) call abandonment rate; |
| 9.5.6.7.2 | Eighty percent (80%) of calls are answered by a live voice within thirty (30) seconds; |
| 9.5.6.7.3 | Blocked call rate does not exceed three percent (3%); and |
| 9.5.6.7.4 | One hundred percent (100%) of voicemails are returned within one (1) Business Day. |
Page 143 of 301
| 9.5.6.8 | The Contractor shall submit a Call Center Report in a format prescribed by ASES. |
ARTICLE 10 PROVIDER CONTRACTING
| 10.1 | General Provisions |
| 10.1.1 | The Contractor shall establish a care model in which the PCP, located within a PMG, manages and coordinates the Enrollee’s care in a timely manner. |
| 10.1.2 | The PCP shall provide, manage, and coordinate services to the Enrollee, including coordination with Behavioral Health personnel, in a timely manner, and in accordance with the guidelines, protocols, and practices generally accepted in medicine. |
| 10.1.3 | The Contractor and each of its Network Providers shall work to ensure that physical and Behavioral Health Services are delivered in a coordinated manner and conform to the standards as provided in Article 8. |
| 10.1.4 | The Contractor shall contract with enough PMGs to serve the Enrollees in each of its Service Regions. As a precondition to executing any Provider Contract, the Contractor shall comply with the requirements stated in Section 10.1.6.1 regarding submitting a model for each type of Provider Contract to ASES. |
| 10.1.5 | The Contractor shall not contract with any Provider without confirming and documenting that the Provider meets all of the Credentialing requirements specified in Section 9.2.3 of this Contract. Failure by the Contractor to adequately monitor the Credentialing of Providers may result in sanctions, liquidated damages, and/or fines in accordance with Articles 19 and 20 or termination of this Contract. |
| 10.1.6 | Model Provider Contracts |
| 10.1.6.1 | The Contractor shall submit a model for each type of Provider Contract to ASES for review and prior written approval, according to the timeframe specified in Attachment 12 to this Contract. The Contractor shall include in such submission, at a minimum, model contracts for PMGs, PCPs, Ancillary Service Providers, Hospitals, Emergency Rooms, and Ambulance Services. The Contractor shall provide ASES with digitized copies of each finalized Provider Contract within thirty (30) Calendar Days of the effective date of the Provider Contract. At the time of submitting the finalized Provider Contract, the Contractor shall disclose to ASES whether the Provider falls under the prohibition stated in Sections 29.1, 29.2, or 29.6 of this Contract. |
Page 144 of 301
| 10.1.6.2 | ASES shall review each executed Provider Contract against the approved models of Provider Contracts. ASES reserves the right to cancel Provider Contracts or to impose sanctions or fees against the Contractor for the omission of clauses required in the contracts with Providers. |
| 10.1.6.3 | On an ongoing basis, any modifications to models of Provider Contracts shall be submitted to ASES for review and prior written approval, before the amendment may be executed. Similarly, any amendments to Provider Contracts shall be submitted to ASES for review and prior written approval. |
| 10.1.7 | The Contractor shall not discriminate against a Provider that is acting within the scope of its license or certification under applicable Puerto Rico law, in decisions concerning contracting, solely on the basis of that license or certification. This Section shall not be construed as precluding the Contractor from using different payment amounts for different specialties, or for different Providers in the same specialty. |
| 10.1.8 | The Contractor awarded the San Juan Service Region shall make best efforts (and (i) provide proof of such efforts in accordance with Attachment 15 and (ii) demonstrate that access requirements have been met) to contract all available San Juan municipal health care facilities and Providers to provide physical and Behavioral Health Services within the Contractor’s Provider Network for the GHP in the San Juan Region including, but not limited to: |
| 10.1.8.1 | Primary Medical Groups; |
| 10.1.8.2 | Primary Care Centers (“Centros Primarios”); |
| 10.1.8.3 | Diagnostics and Ambulatory Care Centers; |
| 10.1.8.4 | Community Clinics (Dispensary); and |
| 10.1.8.5 | Hospital Municipal de San Juan. |
| 10.2 | Provider Training |
| 10.2.1 | Provider Guidelines |
| 10.2.1.1 | The Contractor shall prepare Provider Guidelines, to be distributed to all Network Providers (General Network and PPN), summarizing the GHP program. The Provider Guidelines shall, in accordance with 42 CFR 438.236, (i) be based on valid and reliable clinical evidence or a consensus of Providers in the particular field; (ii) consider the needs of the Contractor’s Enrollees; (iii) be adopted in consultation with Providers; and (iv) be reviewed and updated periodically, as appropriate. |
Page 145 of 301
| 10.2.1.2 | The Provider Guidelines shall describe the procedures to be used to comply with the Provider’s duties and obligations pursuant to this Contract, and under the Provider Contract. |
| 10.2.1.3 | The Contractor shall submit the Provider Guidelines to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 10.2.1.4 | The content of the Provider Guidelines will include, without being limited to, the following topics: the duty to verify eligibility; selection of Providers by the Enrollee; Covered Services; procedures for Access to and provision of services; Preferential Turns, as applicable; coordination of Access to Behavioral Health Services; required service schedule; Medically Necessary Services available twenty-four (24) hours ; report requirements; Utilization Management policies and procedures; Medical Record maintenance requirements; Complaint, Grievance, and Appeal procedures (see Article 14); Co-Payments; HIPAA requirements; the prohibition on denial of Medically Necessary Services; Electronic Health Records and sanctions or fines applicable in cases of non-compliance. |
| 10.2.1.5 | The Provider Guidelines shall be delivered to each Network Provider as part of the Provider contracting process, and shall be made available to Enrollees and to Potential Enrollees upon request. The selected Contractor shall maintain evidence of having delivered the Provider Guidelines to all of its Network Providers within fifteen (15) Calendar Days of award of the Provider Contract. The evidence of receipt shall include the legible name of the Network Provider, NPI, date of delivery, and signature of the Network Provider and shall be made available to ASES Immediately upon request. |
| 10.2.1.6 | The Contractor shall have policies and procedures (that have been prior approved in writing by ASES in accordance with the timeframes in Attachment 12 in place, including both updates to the Provider Guidelines and other communications) to inform its Provider Network, in a timely manner, of programmatic changes such as changes to drug formularies, Covered Services, and protocols. |
| 10.2.2 | Provider Education |
| 10.2.2.1 | The Contractor shall develop a continuing education curriculum for Providers consisting of twenty (20) hours per year divided into five (5) hours per quarter. The curriculum shall be submitted to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. Provider participation in the continuing education curriculum is part of the quality incentive program (see Section 12.5 of the Contract). The curriculum shall include a description of how the Contractor will educate Providers on Contract requirements and shall also include, at a minimum: |
Page 146 of 301
| 10.2.2.1.1 | Initial and ongoing Provider training and education regarding Medicaid with specific emphasis on EPSDT and Behavioral Health integration, the conditions of participation in the Contractor’s MCO, billing processes, and the Provider’s responsibilities to the Contractor and its Enrollees; and |
| 10.2.2.1.2 | Initial and ongoing Provider education and training to address clinical issues and improve the service delivery system, including, but not limited to, assessments, treatment plans, plans of care, discharge plans, evidence-based practices and models of care such as integrated care and trauma-informed care. |
| 10.2.2.2 | The Contractor shall coordinate topics with the PBM’s Academic Detailing Program to develop educational activities addressing: |
| 10.2.2.2.1 | Management and implications of polypharmacy; |
| 10.2.2.2.2 | Condition management; |
| 10.2.2.2.3 | Management of prescriptions; and |
| 10.2.2.2.4 | Working with patients with conditions of special concern, including autism, ADHD, depression, and diabetes among others. |
| 10.2.2.3 | The Contractor shall use various forms of delivery when providing Providers’ training sessions, including web-based sessions, group workshops, face-to-face individualized education, newsletters, communications, and office visits. |
| 10.2.2.4 | The Contractor shall make the dates and locations of sessions available to Providers, as soon as possible, but no later than five (5) Business Days prior to the event. |
| 10.2.2.5 | Training shall be offered throughout the different geographic regions of Puerto Rico and at different times of the day in order to accommodate participating Providers’ schedules. |
| 10.2.2.6 | The Contractor must have a process to document Provider participation in continuing education, and shall provide ASES with, upon request, documentation that Provider education and training requirements have been met. |
Page 147 of 301
| 10.2.2.7 | The Contractor shall provide technical assistance to Providers as determined necessary by the Contractor or by ASES. |
| 10.2.2.8 | The Contractor shall maintain a record of its training and technical assistance activities, which it shall make available to ASES upon request. |
| 10.3 | Required Provisions in Provider Contracts |
| 10.3.1 | All Provider Contracts shall be labeled with the Provider’s NPI, if applicable. In general, the Contractor’s Provider Contracts shall: |
| 10.3.1.1 | Include a section summarizing the Contractor’s obligations under this Contract, as they affect the delivery of health care services under the GHP, and describing Covered Services and populations (or, include the Provider Guidelines as an attachment); |
| 10.3.1.2 | Include a signature page that contains the Contractor and Provider names which are typed or legibly written, Provider company with titles, and dated signatures of all appropriate parties; |
| 10.3.1.3 | Specify the effective dates of the Provider Contract; |
| 10.3.1.4 | Require that the Provider work to advance the integrated model of physical and Behavioral Health Services; |
| 10.3.1.5 | Require that the Provider comply with the applicable Federal and Puerto Rico laws listed in Attachment 1 to this Contract, and with all CMS requirements; |
| 10.3.1.6 | Require that the Provider verify the Enrollee’s Eligibility before providing services or making a Referral; |
| 10.3.1.7 | Prohibit any unreasonable denial, delay, or rationing of Covered Services to Enrollees; and violation of this prohibition shall be subject to the provisions of Article VI, Section 6 of Act 72 and of 42 CFR Part 438, Subpart I (Sanctions); |
| 10.3.1.8 | Prohibit the Provider from making claims for any un-allowed administrative expenses, as listed in Section 22.1.15; |
| 10.3.1.9 | Prohibit the unauthorized sharing or transfer of ASES Data, as defined in Section 28.1; |
Page 148 of 301
| 10.3.1.10 | Notify the Provider that the terms of the contract for services under the GHP program are subject to subsequent changes in legal requirements that are outside of the control of ASES; |
| 10.3.1.11 | Require the Provider to comply with all reporting requirements contained in Article 18 of this Contract, as applicable, and particularly with the requirements to submit Encounter Data for all services provided, and to report all instances of suspected Fraud, Waste, or Abuse; |
| 10.3.1.12 | Require the Provider to acknowledge that ASES Data (as defined in Section 28.1.1) belongs exclusively to ASES, and that the Provider may not give access to, assign, or sell such Data to Third Parties, without Prior Authorization from ASES. The Contractor shall include penalty clauses in its Provider Contracts to prohibit this practice, and require that the fines be determined by and payable to ASES; |
| 10.3.1.13 | Prohibit the Provider from seeking payment from the Enrollee for any Covered Services provided to the Enrollee within the terms of the Contract, and require the Provider to look solely to the Contractor for compensation for services rendered to Enrollees, with the exception of any nominal cost-sharing, as provided in Section 7.11; |
| 10.3.1.14 | Require the Provider to cooperate with the Contractor’s quality improvement and Utilization Management activities; |
| 10.3.1.15 | Not prohibit a Provider from acting within the lawful scope of practice, from advising or advocating on behalf of an Enrollee for the Enrollee’s health status, medical care, or treatment or non-treatment options per 42 CFR 438.102(a)(1); |
| 10.3.1.16 | Not prohibit a Provider from advocating on behalf of the Enrollee in any Grievance System or Utilization Management process, or individual authorization process to obtain necessary health care services; |
| 10.3.1.17 | Require Providers to meet the timeframes for Access to services pursuant to Section 9.5 of this Contract; |
| 10.3.1.18 | Provide for continuity of treatment in the event that a Provider’s participation in the Contractor’s Network terminates during the course of an Enrollee’s treatment by that Provider; |
| 10.3.1.19 | Require Providers to monitor and as necessary and appropriate register Enrollee patients to determine whether they have a medical condition that suggests Care Management or Disease Management services are warranted; |
Page 149 of 301
| 10.3.1.20 | Prohibit Provider discrimination against high-risk populations or Enrollees requiring costly treatments; |
| 10.3.1.21 | Prohibit Providers who do not have a pharmacy license from directly dispensing medications, as required by the Puerto Rico Pharmacy Act (with the exception noted in Section 7.5.12.3.2); |
| 10.3.1.22 | Specify that the Federal Department of Health and Human Services and its sub-agencies and ASES and the Health Department and any of its sub-agencies shall have the right to inspect, evaluate, and audit any pertinent books, financial records, facilities, documents, papers, and records of any Provider involving financial transactions related to the GHP program; |
| 10.3.1.23 | Include the definition and standards for Medical Necessity, pursuant to the definition in Section 7.2.1 of this Contract; |
| 10.3.1.24 | Require that the Provider attend promptly to requests for Prior Authorizations and Referrals, when Medically Necessary, in compliance with the timeframes set forth in Section 9.5 and in 42 CFR 438.210 and the Puerto Rico Patient’s Bill of Rights; |
| 10.3.1.25 | Prohibit the Provider from establishing specific days for the delivery of Referrals or requests for Prior Authorization; |
| 10.3.1.26 | Notify the Provider that, in order to participate in the Medicare Platino Program, the Provider must accept GHP Enrollees; |
| 10.3.1.27 | Specify rates of payment, as detailed in Section 10.5, and require that Providers accept such payment as payment in full for Covered Services provided to Enrollees, less any applicable Enrollee Co-Payments pursuant to Section 7.11 of this Contract; |
| 10.3.1.28 | Specify acceptable billing and coding requirements including ICD-10; |
| 10.3.1.29 | Require that the Provider comply with the Contractor’s Cultural Competency plan; |
| 10.3.1.30 | Require that any Marketing Materials developed and distributed by the Provider be submitted to the Contractor for submission to ASES for prior written approval; |
| 10.3.1.31 | Specify that the Contractor shall be responsible for any payment owed to Providers for services rendered after the Effective Date of Enrollment, as provided in Section 5.2.2, including during the retroactive period described in Section 5.1.3.1; |
Page 150 of 301
| 10.3.1.32 | Require Providers to collect Enrollee Co-Payments as specified in Attachment 8; |
| 10.3.1.33 | Require that Providers not employ or subcontract with individuals on the Puerto Rico or Federal LEIE, or with any entity that could be excluded from the Medicaid program under 42 CFR 1001.1001 (ownership or control in sanctioned entities) and 1001.1051 (entities owned or controlled by a sanctioned person); |
| 10.3.1.34 | Require that Medically Necessary Services shall be available twenty-four (24) hours per day, seven (7) days per Week, to the extent feasible; |
| 10.3.1.35 | Prohibit the Provider from operating on a different schedule for GHP Enrollees than for other patients, and from in any other way discriminating in an adverse manner between GHP Enrollees and other patients; |
| 10.3.1.36 | Not require that Providers sign exclusive Provider Contracts with the Contractor if the Provider is an FQHC or RHC; |
| 10.3.1.37 | Provide notice that the Contractor’s negotiated rates with Providers shall be adjusted in the event that the Executive Director of ASES directs the Contractor to make such adjustments in order to reflect budgetary changes to the Medical Assistance program; |
| 10.3.1.38 | Impose fees or penalties if the Provider breaches the contract or violates Federal or Puerto Rico laws or regulations; |
| 10.3.1.39 | Require that the Provider make every effort to cost-avoid claims and identify and communicate to the Contractor available Third Party resources, as required in Section 23.4 of this Contract, and require that the Contractor cover no health care services that are the responsibility of the Medicare Program; |
| 10.3.1.40 | Provide that the Contractor shall not pay Claims for services covered under the Medicare Program, and that the Provider may not bill both the GHP and the Medicare Program for a single service to a Dual Eligible Beneficiary; |
| 10.3.1.41 | Require the Provider to sign a release giving ASES access to the Provider’s Medicare billing Data for GHP Enrollees who are Dual Eligible Beneficiaries, provided that such access is authorized by CMS and compliant with all HIPAA requirements; |
Page 151 of 301
| 10.3.1.42 | Set forth the Provider’s obligations under the Physician Incentive Programs outlined in Section 10.7 of this Contract; |
| 10.3.1.43 | Require the Provider to notify the Contractor Immediately if or whether the Provider falls within the prohibitions stated in Sections 29.1, 29.2, or 29.6 of this Contract or has been excluded from the Medicare, Medicaid, or Title XX Services Programs; |
| 10.3.1.44 | Include a penalty clause to require the return of public funds paid to a Provider that falls within the prohibitions stated in Section 29.1, 29.2 or 29.6 of this Contract; |
| 10.3.1.45 | Require that all reports submitted by the Provider to the Contractor be labeled with the Provider’s NPI, if applicable; |
| 10.3.1.46 | Require the Provider to participate in the Provider education activities described in Section 10.2.2; |
| 10.3.1.47 | Include Provider dispute process as described in Section 14; |
| 10.3.1.48 | Require the Provider to disclose information on ownership and control as specified in Section 54.2; and |
| 10.3.1.49 | Require the Provider to disclose information as listed in Section 23.7.4. |
| 10.3.2 | In addition to the required provisions in Section 10.3.1, the following requirements apply to specific categories of Provider contracts. |
| 10.3.2.1 | The Contractor’s contracts with PMGs shall: |
| 10.3.2.1.1 | Require that the PMG provide services on a regular time schedule, Monday through Saturday, from 8:00 a.m. to 6:00 p.m.; PMG will not have to comply with this requirement during the following holidays: January 1st, January 6th, Good Friday, Thanksgiving and December 25th. The PMG has sole discretion to decide whether or not to provide services during the previously listed holidays; |
| 10.3.2.1.2 | Require that the PMG employs enough personnel to offer urgent care services between 6:00 p.m. and 9:00 p.m. (Atlantic Time), Monday through Friday; |
| 10.3.2.1.3 | Require that the PMG coordinates with Behavioral Health personnel to ensure integrated physical and Behavioral Health Services, as provided in Article 8; |
Page 152 of 301
| 10.3.2.1.4 | Require the PMG to work, to the extent possible, within the Contractor’s established PPN, in directing care for Enrollees and coordinating services; |
| 10.3.2.1.5 | Authorize the Contractor to adjudicate disputes between the PMG and its Network Providers about the validity of claims by any Network Provider; and |
| 10.3.2.1.6 | Require PMGs to provide assurances that the Encounter Data submitted by the PMG to the Contractor encompass all services provided to GHP Enrollees, including clinical laboratories. |
| 10.3.2.2 | The Contractor’s contracts with PCPs shall require the PCP to inform and distribute Information to Enrollee patients about instructions on Advance Directives, and shall require the PCP to notify Enrollees of any changes in Federal or Puerto Rico law relating to Advance Directives, no more than ninety (90) Calendar Days after the effective date of such change. |
| 10.3.2.3 | The Contractor’s contracts with a Provider who is a member of the PPN shall prohibit the Provider from collecting Co-Payments from GHP Enrollees, subject only to the exceptions established in Article 9 of this Contract and the Attachment 8 to this Contract (Co-Payment Chart). |
| 10.3.2.4 | The Contractor’s contracts with Hospitals and Emergency Rooms shall prohibit the Hospital or Emergency Room from placing a lower priority on GHP Enrollees than on other patients, and from referring GHP Enrollees to other facilities for reasons of economic convenience. Such contracts must include sanctions penalizing this practice. |
| 10.4 | Termination of Provider Contracts |
| 10.4.1 | The Contractor shall comply with all Puerto Rico and Federal laws regarding Provider termination. The Provider Contracts shall: |
| 10.4.1.1 | Contain provisions allowing immediate termination of the Provider Contract by the Contractor “for cause.” Cause for termination includes gross negligence in complying with the contractual considerations or obligations; insufficiency of funds of ASES or the Contractor, which prevents them from continuing to pay for their obligations; and changes in Federal law. |
| 10.4.1.2 | Specify that in addition to any other right to terminate the Provider Contract, and notwithstanding any other provision of this Contract, ASES may demand Provider termination Immediately, or the Contractor may Immediately terminate on its own, a Provider’s participation under the Provider Contract if: |
Page 153 of 301
| 10.4.1.2.1 | The Provider fails to abide by the terms and conditions of the Provider Contract, as determined by ASES, or, in the sole discretion of ASES, if the Provider fails to come into compliance within fifteen (15) Calendar Days after a receipt of notice from the Contractor specifying such failure and requesting such Provider to abide by the terms and conditions hereof; or |
| 10.4.1.2.2 | The Contractor or ASES learns that the Provider: |
| 10.4.1.2.2.1 | Falls within the prohibition stated in Section 29.1 or 29.2, or has a criminal conviction as provided in Section 29.6; |
| 10.4.1.2.2.2 | Has been or could be excluded from participation in the Medicare, Medicaid, or CHIP Programs; |
| 10.4.1.2.2.3 | Could be excluded from the Medicaid Program under 42 CFR 1001.1001 (ownership or control in sanctioned entities) and 1001.1051 (entities owned or controlled by a sanctioned person); and/or |
| 10.4.1.2.2.4 | Fails to comply with the Provider Credentialing process and requirements. |
| 10.4.1.3 | Specify that any Provider whose participation is terminated under the Provider Contract for any reason shall utilize the applicable Appeals procedures outlined in the Provider Contract. No additional or separate right of Appeal to ASES or the Contractor is created as a result of the Contractor’s act of terminating, or decision to terminate any Provider under this Contract. Notwithstanding the termination of the Provider Contract with respect to any particular Provider, this Contract shall remain in full force and effect with respect to all other Providers. |
| 10.4.2 | The Contractor shall notify ASES at least forty-five (45) Calendar Days prior to the effective date of the suspension, termination, or withdrawal of a Provider from participation in the Contractor’s General Network. If the termination was “for cause,” the Contractor shall provide to ASES the reasons for termination immediately. |
| 10.4.3 | The Contractor shall, within fifteen (15) Calendar Days of issuance of a notice of termination to a Provider, notify Enrollees of the termination, and shall assist the Enrollee as needed in finding a new Provider. |
Page 154 of 301
| 10.5 | Provider Payment |
| 10.5.1 | General Provisions |
| 10.5.1.1 | The Contractor guarantees payment for all Medically Necessary Services rendered by Providers on a person’s Effective Date of Enrollment, including during the retroactive period described in Section 5.1.3.1. |
| 10.5.1.2 | The Contractor shall require, as a condition of payment, that the Provider accept the amount paid by the Contractor or appropriate denial made by the Contractor (or, if applicable, payment by the Contractor that is supplementary to the Enrollee’s Third Party payer) plus any applicable amount of Co-Payment responsibilities due from the Enrollee as payment in full for the service. |
| 10.5.1.3 | The Contractor shall ensure that Enrollees are held unaccountable by the Provider for the costs of Medically Necessary Services except for applicable Co-Payment amounts (described in Section 9.3 of this Contract and Attachment 8 to this contract). |
| 10.5.1.4 | The insolvency, liquidation, bankruptcy, or breach of contract of any Provider will not release the Contractor from its obligation to pay for all services rendered as authorized under this Contract. |
| 10.5.1.5 | With the exceptions noted below, the Contractor shall negotiate rates with Providers, and such rates shall be specified in the Provider Contract. Payment arrangements may take any form allowed under Federal law and the laws of Puerto Rico, including Capitation payments, Fee-for-Service payment, and salary, if any, subject to Section 10.6 concerning permitted risk arrangements. The Contractor shall inform ASES in writing when it enters any Provider payment arrangement other than Fee-for-Service. |
| 10.5.1.6 | Any Capitation payment made by the Contractor to Providers shall be based on sound actuarial methods. All Provider payments by the Contractor shall be reasonable, and the amount paid shall not jeopardize or infringe upon the quality of the services provided. |
| 10.5.1.7 | Even if the Contractor does not enter into a capitated payment arrangement with a Provider, the Provider shall nonetheless be required to submit to the Contractor detailed Encounter Data (see Section 16.8 of this Contract). |
| 10.5.1.8 | The Contractor shall be responsible for issuing to the forms required by the Department of the Treasury, in accordance with all Puerto Rico laws, regulations, and guidelines. |
Page 155 of 301
| 10.5.1.9 | The Contractor shall make timely payments to Providers in accordance with the timeliness standards outlined in Section 16.10 of this Contract. |
| 10.5.2 | Payments to FQHCs and RHCs. When the Contractor negotiates a contract with an FQHC and/or an RHC, as defined in Section 1905(a)(2)(B) and 1905(a)(2)(C) of the Social Security Act, the Contractor shall pay to the FQHC or RHC rates that are comparable to rates paid to other similar Providers providing similar services. The Contractor shall cooperate with ASES and the Department of Health in ensuring that payments to FQHCs and RHCs are consistent with Sections 1902(a)(15) and 1902(bb)(5) of the Social Security Act. |
| 10.5.3 | Requirement To Verify Eligibility. The Contractor warrants that all of its Network Providers shall verify the eligibility of Enrollees before the Provider provides Covered Services. This verification of eligibility is a condition of receiving payment from the Contractor for Covered Services. |
| 10.5.4 | Payments to Providers Owing Funds to ASES. Upon receipt of notice from ASES that ASES is owed funds by a Provider, the Contractor shall reduce payment to the Provider for all Claims submitted by that Provider by one hundred percent (100%), or such other amount as ASES may elect, until the amount owed to ASES is recovered. The Contractor shall promptly remit any such funds recovered to ASES in the manner specified by ASES. To that end, the Contractor’s Provider Contracts shall contain a provision giving notice of this obligation to the Provider, such that the Provider’s execution of the Contract shall constitute agreement with the Contractor’s obligation to ASES. |
| 10.5.5 | Payment Rates Subject to Change. The Contractor shall adjust its negotiated rates with Providers to reflect budgetary changes, as directed by the Executive Director of ASES, to the extent that such adjustments can be made within funds appropriated to ASES and available for payment to the Contractor. The Contractor’s Provider Contracts shall contain a provision giving notice of this obligation to the Provider, such that the Provider’s execution of the Provider Contract shall constitute agreement with the Contractor’s obligation to ASES. |
| 10.5.6 | Payments for Hospitalization Services or Services Extending for More than Thirty (30) Calendar Days. In the event of hospitalization or extended services that exceed thirty (30) Calendar Days, the Provider may bill and collect payments for services rendered to the Enrollee at least once per month. These services shall be paid according to the procedures discussed in this Article 10. |
| 10.5.7 | Payments for Services to Dual Eligible Beneficiaries. The Contractor shall include in its Provider Contracts a notice that the Contractor shall not pay Claims for services covered under the Medicare Program. No Provider may bill both the GHP and the Medicare Program for a single service rendered to a Dual Eligible Beneficiary. The Contractor shall include in its Provider contracts a requirement that the Provider must comply with 42 CFR 447.15 to accept Medicaid payments as payment in full. |
Page 156 of 301
| 10.5.8 | Payment for Pharmacy Services. The Contractor shall abide by and comply with the following payment process hereby established: |
| 10.5.8.1 | In covering pharmacy services, the Contractor shall adhere to the retail pharmacy reimbursement levels established in Attachment 6 to this Contract. |
| 10.5.8.2 | On a semi-monthly payment cycle to be set by the PBM, the PBM will provide the Contractor with the proposed Claims listing. The Contractor shall promptly review the payment listing. |
| 10.5.8.3 | The Contractor shall submit funds for Claims payment to the PBM’s zero-balance account. The Contractor shall provide funds or wire transfers to a bank account established for the payment of the Claims, or otherwise submit payment, within two (2) Business Days of the date that the prescription was filled. |
| 10.5.8.4 | The Contractor, ASES, and the PBM shall cooperate to identify additional savings opportunities, including special purchasing opportunities, changes in network fees, etc. |
| 10.5.9 | Payments to State Health Facilities. The Contractor shall establish a payment system, to be prior approved in writing by ASES, to improve cash flow to health care facilities administered or operated by the Central Government, State Academic Medical Centers, and certain facilities in the San Juan Municipality that participate in the General Network. The following health care facilities may participate: |
| 10.5.9.1 | Hospital Regional de Bayamon; |
| 10.5.9.2 | Hospital Universitario de Adultos; |
| 10.5.9.3 | Hospital Federico Trilla; |
| 10.5.9.4 | Hospital Pediátrico Universitario; |
| 10.5.9.5 | Centro Cardiovascular de PR y del Caribe; |
| 10.5.9.6 | Hospital Municipal de San Juan; |
| 10.5.9.7 | Administración de Servicios Médicos de PR ("ASEM”); and |
| 10.5.9.8 | ASSMCA facilities. |
Page 157 of 301
| 10.5.10 | Payments to Providers Outside the PPN. The Contractor shall provide for adequate payment in its contracts with Providers outside the PPN. |
| 10.5.11 | Payments for Emergency Services and Post-Stabilization Services |
| 10.5.11.1 | The Contractor shall not deny a Claim from a Provider for Emergency Services and shall make payment to a Provider for responding to an Enrollee’s Emergency Medical Condition or Psychiatric Emergency by performing medical screening examinations and stabilizing treatment. |
| 10.5.11.2 | Pursuant to Section 1932(b)(2)(D) of the Social Security Act, the Contractor shall limit payments to Out-of-Network Providers of Emergency Services to the amount that would have been paid if the service had been provided by a Network Provider. |
| 10.6 | Acceptable Risk Arrangements |
| 10.6.1 | The Contractor’s Provider Contracts with PMGs shall establish a financial risk arrangement agreed upon between the Contractor and the PMG which shall be clearly stated in the PMG contract with the Contractor. |
| 10.7 | Physician Incentive Programs |
| 10.7.1 | General Provisions |
| 10.7.1.1 | The Contractor may, upon prior written approval from ASES, design and implement one (1) Physician Incentive Plan, and shall incorporate the requirements of this plan into Provider Contracts. The Contractor shall submit a written request to ASES before implementing any such incentive program by providing a summary of the program for ASES review and approval at least sixty (60) Calendar Days before the projected implementation date for the program. ASES has the absolute right to approve or disapprove the Physician Incentive program, and the program may be implemented only upon receipt of prior written approval from ASES. |
| 10.7.1.2 | ASES will approve a Physician Incentive program only if it, in ASES’s discretion, meets the following requirements: |
| 10.7.1.2.1 | The program contains credible medical standards in support of the improvement of quality health services and reduces or eliminates any adverse effects on patients’ care; |
| 10.7.1.2.2 | All incentive payments to Providers are related to or made under quality initiatives supported or otherwise approved by CMS; |
Page 158 of 301
| 10.7.1.2.3 | The implementation of the program in no way reduces or otherwise limits Enrollee Access to Medically Necessary Services (including a reduction in prescription drugs, diagnostic tests or treatments, hospitalization, and other treatment available regardless of the incentives); |
| 10.7.1.2.4 | The Contractor shall employ continuous monitoring by an independent Third Party to confirm that Enrollee care is not adversely affected by the program; |
| 10.7.1.2.5 | The intent of the program is to improve the quality of the services to Enrollees. Enrollees must be informed of the existence of the Physician Incentive program, and the Provider shall be made fully responsible for the proper care to the Enrollee; and |
| 10.7.1.2.6 | Incentives are not used to penalize Providers who serve Enrollees whose treatment needs, according to the Provider’s medical judgment, do not fall within the Contractor’s fixed clinical protocols. |
| 10.7.2 | Pay for Performance for Hospitals. ASES approves the use of incentive programs targeting hospitals, provided that the incentive programs: |
| 10.7.2.1 | Encourage the use of medical standards that support quality improvement and reduce adverse effects in Enrollee care; |
| 10.7.2.2 | Advance the quality initiatives supported by CMS; |
| 10.7.2.3 | Are not geared toward, and do not have the likely effect of, reducing or limiting services that the Enrollee needs or may need (for example, reduction of diagnostic exams, hospitalization, or treatment); |
| 10.7.2.4 | Are not used solely as a mechanism for reducing payments to or recovering payments from Providers; |
| 10.7.2.5 | Contain clearly defined objectives, effectively communicated to both Providers and (upon request) Enrollees; |
| 10.7.2.6 | Aim to reduce “never events,” such as health care-associated infections and other hospital-acquired conditions (including reaction to foreign substances accidentally left in during procedure, air embolism, blood incompatibility, pressure ulcers, and falls); |
| 10.7.2.7 | Address inappropriate admissions and readmissions; and |
| 10.7.2.8 | Address over-utilization of caesarian sections. |
Page 159 of 301
ARTICLE 11 UTILIZATION MANAGEMENT
| 11.1 | General |
| 11.1.1 | The Contractor shall comply with Puerto Rico and Federal requirements for Utilization Management (“UM”) including but not limited to 42 C.F.R. Part 456. |
| 11.1.2 | The Contractor shall ensure the involvement of appropriate, knowledgeable, currently practicing Providers in the development of UM procedures. |
| 11.1.3 | The Contractor shall manage the use of a limited set of resources and maximize the effectiveness of care by evaluating clinical appropriateness, and authorizing the type and volume of services through fair, consistent, and Culturally Competent decision-making processes while ensuring equitable Access to care and a successful link between care and outcomes. |
| 11.1.4 | The Contractor shall submit to ASES on an annual basis existing UM edits in the Contractor’s Claims processing system that control Utilization and prevent payment for Claims that are duplicates, unbundled when they should be bundled, already covered under another charge, etc. |
| 11.1.5 | ASES reserves the right require the Contractor to submit any Utilization Management report. |
| 11.2 | Utilization Management Policies and Procedures |
| 11.2.1 | The Contractor shall provide assistance to Enrollees and Providers to ensure the appropriate Utilization of resources. The Contractor shall have written Utilization Management policies and procedures included in the Provider Guidelines (see Section 10.2.1.4) that: |
| 11.2.1.1 | Include protocols and criteria for evaluating Medical Necessity, authorizing services, and detecting and addressing over, under, and inappropriate Utilization. Such protocols and criteria shall comply with Federal and Puerto Rico laws and regulations. |
| 11.2.1.2 | Address which services require PCP Referral, which services require Prior Authorization, and how requests for initial and continuing services are processed, and which services will be subject to concurrent, retrospective, or prospective review. |
| 11.2.1.3 | Describe mechanisms in place that ensure consistent application of review criteria for Prior Authorization decisions and consult with the requesting Provider when appropriate. |
Page 160 of 301
| 11.2.1.4 | Require that all Medical Necessity determinations be made in accordance with ASES’s Medical Necessity definition as stated in Section 7.2. |
| 11.2.1.5 | Facilitate the delivery of high quality, low cost, efficient, and effective care. |
| 11.2.1.6 | Ensure that services are based on the history of the problem or illness, its context, and desired outcomes. |
| 11.2.1.7 | Emphasize relapse and crisis prevention, not just crisis intervention. |
| 11.2.1.8 | Detect over, under, and inappropriate Utilization of services to assess quality and appropriateness of services and to assess quality and appropriateness of care furnished to Enrollees with special health care needs. |
| 11.2.1.9 | Ensure that any decision to deny a Service Authorization Request or to authorize a service in an amount, duration, or scope that is less than requested, be made by a Provider who has appropriate clinical expertise to understand the treatment of the Enrollee’s condition or disease, such as the Contractor’s medical director. |
| 11.2.2 | The Contractor shall submit its Utilization Management policies and procedures to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 11.2.3 | The Contractor’s Utilization Management policies and procedures shall define when a conflict of interest for a Provider involved in Utilization Management activities in its Service Region may exist and shall describe the remedy for such conflict. |
| 11.2.4 | The Contractor, and any delegated Utilization Management agent, shall not permit or provide compensation or anything of value to its employees, Agents, or contractors based on: |
| 11.2.4.1 | Either a percentage of the amount by which a Claim is reduced for payment or the number of Claims or the cost of services for which the person has denied authorization or payment; or |
| 11.2.4.2 | Any other method that encourages a decision to deny, limit, or discontinue a Medically Necessary Covered Service to any Enrollee, as set forth by 42 CFR 438.210(e). |
Page 161 of 301
| 11.3 | Utilization Management Guidance to Enrollees. |
| 11.3.1 | As provided in Section 6.4.5.22, the Contractor shall provide clear guidance in its Enrollee Handbook on Utilization Management policies. Upon request, the Contractor shall provide Utilization Management decision criteria to Providers, Enrollees, their families, and the public. |
| 11.4 | Prior Authorization and Referral Policies |
| 11.4.1 | Referrals |
| 11.4.1.1 | The Contractor shall not require a Referral from a PCP when an Enrollee seeks care from a Provider in the Contractor’s PPN. |
| 11.4.1.2 | A written Referral from the PCP shall be required: |
| 11.4.1.2.1 | For the Enrollee to access specialty care and services within the Contractor’s General Network but outside the PPN; and |
| 11.4.1.2.2 | For the Enrollee to access an Out-of-Network Provider (with the exception of Emergency Services). |
| 11.4.1.3 | A Referral for either the General Network or out-of-network services will be provided during the same visit with the PCP but no later than twenty-four (24) hours of the Enrollee’s request. |
| 11.4.1.4 | When a Provider does not make the Referral in the required timeframe specified, or refuses to make a Referral, the Contractor shall issue an Administrative Referral. |
| 11.4.1.5 | Neither the Contractor nor any Provider may impose a requirement that Referrals be submitted for the approval of committees, boards, Medical Directors, etc. The Contractor shall strictly enforce this directive and shall issue Administrative Referrals (see Section 11.4.1.4) whenever it deems medically necessary. |
| 11.4.1.6 | If the Provider Access requirements of Section 9.5 of this Contract cannot be met within the PPN within thirty (30) Calendar Days of the Enrollee’s request for the Covered Service, the PMG shall refer the Enrollee to a specialist within the General Network, without the imposition of Co-Payments. However, the Enrollee shall return to the PPN specialist once the PPN specialist is available to treat the Enrollee. |
| 11.4.1.7 | The Contractor shall ensure that PMGs comply with the rules stated in this Section concerning Referrals, so that Enrollees are not forced to change PMGs in order to obtain needed Referrals. |
Page 162 of 301
| 11.4.1.8 | If the Referral system that is developed by the Contractor requires the use of electronic media, such equipment shall be installed in Network Providers’ offices at the Contractor’s expense. |
| 11.4.2 | Timeliness of Prior Authorization |
| 11.4.2.1 | The Contractor shall ensure that Prior Authorization is provided for the Enrollee in the following timeframes, including on holidays and outside of normal business hours. |
| 11.4.2.1.1 | The decision to grant or deny a Prior Authorization must not exceed seventy-two (72) hours from the time of the Enrollee’s Service Authorization Request for all Covered Services; except that, where the Contractor or the Enrollee’s Provider determines that the Enrollee’s life or health could be endangered by a delay in accessing services, the Prior Authorization must be provided as expeditiously as the Enrollee’s health requires, and no later than twenty-four (24) hours from the Service Authorization Request. |
| 11.4.2.1.2 | ASES may, in its discretion, grant an extension of the time allowed for Prior Authorization decisions for up to fourteen (14) Calendar Days, where: |
| 11.4.2.1.2.1 | The Enrollee, or the Provider, requests the extension; or |
| 11.4.2.1.2.2 | The Contractor justifies to ASES a need for the extension in order to collect additional Information, such that the extension is in the Enrollee’s best interest. |
| 11.4.2.1.3 | If ASES extends the timeframe, the Contractor must give the Enrollee written notice of the reason behind granting the extension and inform the Enrollee of the right to file a Grievance if he or she disagrees with that decision. The notice of the determination must be sent as expeditiously as the Enrollee’s health condition requires and no later than the expiration date of the extension. |
| 11.4.2.2 | For services that require Prior Authorization by the Contractor, the Service Authorization Request shall be submitted promptly by the Provider for the Contractor’s approval, so that Prior Authorization may be provided within the timeframe set forth in this Section 11.4.2. |
Page 163 of 301
| 11.4.3 | The Contractor shall submit to ASES Utilization Management clinical criteria to be used for services requiring Prior Authorization. ASES shall prior approve in writing such Utilization Management clinical criteria. |
| 11.4.4 | Prohibited Actions |
| 11.4.4.1 | Any denial, unreasonable delay, or rationing of Medically Necessary Services to Enrollees is expressly prohibited. The Contractor shall ensure compliance with this prohibition from Network Providers or any other entity related to the provision of Behavioral Health services to GHP Enrollees. Should the Contractor violate this prohibition, the Contractor shall be subject to the provisions of Article VI, Section 6 of Act 72 and 42 CFR Subpart I (Sanctions). |
| 11.4.5 | The Contractor shall employ appropriately licensed professionals to supervise all Prior Authorization decisions and shall specify the type of personnel responsible for each type of Prior Authorization in its policies and procedures. Any decision to deny a Service Authorization Request or to authorize a service in an amount, duration, or scope that is less than requested shall be made by a Provider who possesses the appropriate clinical expertise for treating the Enrollee’s condition. For Service Authorization Requests for dental services, only licensed dentists are authorized to make such decisions. |
| 11.4.6 | Emergency Services |
| 11.4.6.1 | Neither a Referral nor Prior Authorization shall be required for any Emergency Service, no matter whether the Provider is within the PPN, and notwithstanding whether there is ultimately a determination that the condition for which the Enrollee sought treatment in the emergency room was not an Emergency Medical Condition or Psychiatric Emergency. |
| 11.4.7 | Dental Services |
| 11.4.7.1 | The Contractor shall not require a Prior Authorization or a Referral for dental services except for maxillofacial surgery which requires Prior Authorization from a PCP. |
| 11.4.8 | Pharmacy Services |
| 11.4.8.1 | The Contractor shall require Prior Authorization for filling a drug prescription for certain drugs specified on the PDL, as provided in Section 7.5.12.10. |
Page 164 of 301
| 11.4.8.2 | The Contractor shall require a Countersignature from the Enrollee’s PCP in order to fill a prescription written by a Provider who is not in the PPN. |
| 11.4.8.3 | Any required Prior Authorization or Countersignature for pharmacy services shall be conducted within the timeframes provided in Sections 7.5.12.4. |
| 11.4.8.4 | The Contractor shall comply with the Utilization Management policies and procedures in Section 7.5.12 of this Contract for pharmacy services. |
| 11.4.9 | Special Coverage |
| 11.4.9.1 | In order to obtain services under Special Coverage, an Enrollee must be registered in the program, as provided in Section 7.7. Registration is a form of Utilization control, to determine whether the Enrollee’s health condition warrants Access to the expanded services included in Special Coverage. |
| 11.4.9.2 | In addition, as noted in Section 7.7.12, some individual Special Coverage services require Prior Authorization even for Enrollees who have registered under Special Coverage. |
| 11.4.10 | Behavioral Health Services. The Contractor shall not require a Prior Authorization or a Referral for Behavioral Health services. |
| 11.5 | Use of Technology to Promote Utilization Management |
| 11.5.1 | ASES strongly encourages the Contractor to develop electronic, web-based Referral processes and systems. In the event that a Referral is made via the telephone, the Contractor shall ensure that Referral Data are maintained in a Data file that can be accessed electronically by the Contractor, the Provider, and ASES. |
| 11.5.2 | In conjunction with its other Utilization Management policies, the Contractor shall submit the Referral processes to ASES for review and prior written approval in accordance with Attachment 12. |
| 11.6 | Court-Ordered Evaluations and Services |
| 11.6.1 | In the event that an Enrollee requires Medicaid-covered services ordered by a court, the Contractor shall fully comply with all court orders while maintaining appropriate Utilization Management practices. |
| 11.7 | Second Opinions |
| 11.7.1 | The Contractor shall provide a second opinion in any situation when there is a question concerning a diagnosis, the options for surgery, or alternative treatments of a health condition when requested by any Enrollee, or by a parent, guardian, or other person exercising a custodial responsibility over the Enrollee. |
Page 165 of 301
| 11.7.2 | The second opinion must be provided by a qualified Network Provider, or, if a Network Provider is unavailable, the Contractor shall arrange for the Enrollee to obtain a second opinion from an Out-of-Network Provider. |
| 11.7.3 | The second opinion shall be provided at no cost to the Enrollee. |
ARTICLE 12 QUALITY IMPROVEMENT AND PERFORMANCE PROGRAM
| 12.1 | General Provisions |
| 12.1.1 | The Contractor shall provide for the delivery of quality care to all Enrollees with the primary goal of improving health status or, in instances where the Enrollee’s health is not amenable to improvement, maintaining the Enrollee’s current health status by implementing measures to prevent any further deterioration of his or her health status. |
| 12.1.2 | The Contractor shall seek input from, and work with, Enrollees, Providers, community resources, and agencies to actively improve the quality of care provided to Enrollees. |
| 12.1.3 | The Contractor shall ensure that its Quality Assessment and Performance Improvement Program effectively monitors the program elements listed in 42 CFR 438.66. |
| 12.1.4 | ASES, in strict compliance with 42 CFR 438.204 and other Federal and Puerto Rico regulations, shall evaluate the delivery of health care by the Contractor. Such quality monitoring shall include monitoring of all the Contractor’s Quality Management/Quality Improvement (“QM/QI”) programs described in this Article 12 of this Contract. |
| 12.1.5 | The Contractor shall cooperate with any Puerto Rico or Federal monitoring of its performance under this Contract, which may include but is not limited to external quality reviews, operational reviews, performance audits and evaluations. |
| 12.1.6 | The Contractor shall identify, collect and provide any Data, Medical Records or other Information requested by ASES or its authorized representative or the Federal agency or its authorized representative in the format specified by ASES/Federal agency or its authorized representative. The Contractor shall ensure that the requested Data, Medical Records, and other Information is provided at no charge to ASES, all Federal agencies, or their authorized representative. |
| 12.1.7 | If requested, the Contractor shall provide workspace at the Contractor’s local offices for ASES, any Federal agencies, or their authorized representative to review requested Data, Medical Records, or other Information. |
Page 166 of 301
| 12.1.8 | Advisory Board |
| 12.1.8.1 | The Contractor shall convene and facilitate an advisory board. Advisory board members shall serve to advise the Contractor on issues concerning service delivery and quality of all Covered Services (e.g., Behavioral Health, physical health), Enrollee rights and responsibilities, resolution of Enrollee Grievances and Appeals and the needs of groups represented by advisory board members as they pertain to Medicaid. |
| 12.1.8.2 | The advisory board shall consist of representatives from all GHP populations, family members, and Providers. The Contractor shall have an equitable representation of its representatives in terms of race, gender, special populations, and Puerto Rico’s geographic areas in the Contractor’s Service Region(s). |
| 12.1.8.3 | The Contractor’s advisory board shall keep a written record of all attempts to invite and include its representatives in its meetings. The Advisory Board roster and minutes shall be made available to ASES ten (10) Calendar Days following the meeting date. See Article 18 of this Contract for additional reporting requirements. |
| 12.1.8.4 | The Contractor shall hold quarterly, centrally located advisory board meetings throughout the Contract Term. The Contractor shall advise ASES ten (10) Calendar Days in advance of meetings to be held. At least two (2) of the quarterly meetings shall focus on Enrollee issues to help ensure that Enrollee issues and concerns are heard and addressed. Attendance rosters and minutes for these two (2) meetings shall be made available to ASES within ten (10) Calendar Days following the meeting date. |
| 12.1.8.5 | The Contractor shall ensure that all advisory board representatives actively participate in deliberations and that no one board representative dominates proceedings in order to foster an inclusive meeting environment. |
| 12.2 | Quality Assessment Performance Improvement (“QAPI”) Program |
| 12.2.1 | The Contractor shall comply with Puerto Rico and Federal standards for Quality Management/Quality Improvement (“QM/QI”). |
| 12.2.1.1 | The Contractor shall establish QAPI that specifies the Contractor’s quality measurement and performance improvement activities using clinically sound, nationally developed and accepted criteria. |
Page 167 of 301
| 12.2.2 | For Medicaid and CHIP Eligibles, the QAPI program shall be in compliance with Federal requirements specified at 42 CFR 438.240. |
| 12.2.3 | The Contractor’s QAPI program shall be based on the latest available research in the area of quality assurance and at a minimum shall include: |
| 12.2.3.1 | A method of monitoring, analyzing, evaluating, and improving the delivery, quality and appropriateness of health care furnished to all Enrollees (including over, under, and inappropriate Utilization of services) and including those with special health care needs; |
| 12.2.3.2 | Written policies and procedures for quality assessment, Utilization Management, and continuous quality improvement that are periodically assessed for efficacy and reflect Enrollee and Network Provider input; |
| 12.2.3.3 | Include an Information System sufficient to support the collection, integration, tracking, analysis, and reporting of Data, in compliance with 42 CFR 438.242; |
| 12.2.3.4 | Designated staff with expertise in quality assessment, Utilization Management, and continuous quality improvement; |
| 12.2.3.5 | A review of outcome Data at least quarterly for performance improvement recommendations and interventions; |
| 12.2.3.6 | A mechanism to detect over, under, and inappropriate Utilization of services; |
| 12.2.3.7 | Reports that have been evaluated, indicated recommendations that are implemented, and provided feedback to Providers and Enrollees; |
| 12.2.3.8 | A methodology and process for conducting Provider Credentialing and Re-Credentialing; |
| 12.2.3.9 | Procedures for validating completeness and quality of Encounter Data; |
| 12.2.3.10 | Annual PIPs as specified by ASES; |
| 12.2.3.11 | Development of an emergency room (ER) quality initiative program (see Section 12.4); |
| 12.2.3.12 | Development of a quality incentive program (see Section 12.5); |
| 12.2.3.13 | Reporting on specified performance measures, including specified performance measures (see Section 12.5.4.1); |
Page 168 of 301
| 12.2.3.14 | Conducting Provider and Enrollee satisfaction surveys (see Section 12.6); |
| 12.2.3.15 | Quarterly reports on program results, conclusions, recommendations, and implemented system changes, as specified by ASES; and |
| 12.2.3.16 | Process for evaluating the impact and effectiveness of the Contractor’s QAPI program at least annually. |
| 12.2.4 | The Contractor’s annual QAPI program shall be submitted to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract and the annual reporting requirements outlined in Article 18. |
| 12.2.5 | The Contractor shall submit any changes to its QAPI program to ASES for review and prior written approval sixty (60) Calendar Days prior to implementation of the change. |
| 12.2.6 | Upon the request of ASES, the Contractor shall provide any Information and documents related to the implementation of the QAPI program. |
| 12.3 | Performance Improvement Projects |
| 12.3.1 | At a minimum, the Contractor shall have a PIPs work plan and activities that are consistent with Federal and Puerto Rico statues, regulations, and Quality Assessment and Performance Improvement Program requirements for pursuant to 42 C.F.R. 438.240.For more detailed information refer to the “EQR Managed Care Organization Protocol” available at http://www.medicaid.gov/Medicaid-CHIP- Program-Information/By-Topics/Quality-of-Care/Quality-of-Care-External- Quality-Review.html. |
| 12.3.2 | PIPs must be designed to achieve, through ongoing measurements and intervention, significant improvement, sustained over time, in clinical care and administrative areas that are expected to have a favorable effect on health outcomes and Enrollee satisfaction. |
| 12.3.3 | The Contractor shall implement PIPs in the following areas: |
| 12.3.3.1 | One (1) clinical care project in the area of increasing fistula use for Enrollees at-risk for dialysis; |
| 12.3.3.2 | One (1) clinical care project in the area of Behavioral Health; |
| 12.3.3.3 | One (1) administrative project in the area of EPSDT screening; |
| 12.3.3.4 | One (1) administrative project in the area of reverse co-location and co-location of physical and Behavioral Health and their integration; and |
Page 169 of 301
| 12.3.3.5 | The Contractor shall conduct additional PIPs as specified by ASES during the Contract Term. |
| 12.3.4 | In designing its PIPs, the Contractor shall: |
| 12.3.4.1 | Show that the selected area of study is based on a demonstration of need and is expected to achieve measurable benefit to Enrollee (rationale); |
| 12.3.4.2 | Establish clear, defined and measurable goals and objectives that the Contractor shall achieve in each year of the project; |
| 12.3.4.3 | Measure performance using quality indicators that are objective, measurable, clearly defined and that allow tracking of performance and improvement over time; |
| 12.3.4.4 | Implement interventions designed to achieve quality improvements; |
| 12.3.4.5 | Evaluate the effectiveness of the interventions; |
| 12.3.4.6 | Establish standardized performance measures (such as HEDIS or another similarly standardized product); |
| 12.3.4.7 | Plan and initiate activities for increasing or sustaining improvement; and |
| 12.3.4.8 | Document the Data collection methodology used (including sources) and steps taken to assure Data is valid and reliable. |
| 12.3.5 | The Contractor shall submit all descriptions of PIPs and program details to ASES annually as part of the QAPI program. |
| 12.3.6 | Each PIP shall be evaluated by the EQRO. The Contractor shall provide information to the EQRO on the status and outcomes of the PIP upon request. |
| 12.3.7 | When requested, the Contractor shall submit Data to ASES or the EQRO for standardized PIPs. The Contractor shall collect valid and reliable Data, using qualified staff and personnel to collect the Data. Failure of the Contractor to follow Data collection and submission requirements may result in sanctions. |
| 12.4 | ER Quality Initiative Program |
| 12.4.1 | The Contractor shall develop an emergency room (ER) quality initiative program, implementing efficient and timely monitoring of Enrollees’ use of the emergency room, including whether such use was justified by a legitimate Emergency Medical Condition or Psychiatric Emergency. |
Page 170 of 301
| 12.4.2 | The ER quality initiative program shall be designed to identify high users of Emergency Services for non-emergency situations and to allow for early interventions in order to ensure appropriate Utilization of services and resources. |
| 12.4.3 | The ER quality initiative program shall specify all strategies to be used by the Contractor to address high users of inappropriate Emergency Services and include, at a minimum, the following components: |
| 12.4.3.1 | Description of system(s) for tracking, monitoring, and reporting high users of ER services for non-emergency situations; |
| 12.4.3.2 | Criteria for defining non-emergency situations; |
| 12.4.3.3 | Educational component to inform (i) Enrollees about the proper use of ER services and how to access ER services and (ii) PCPs about identifying high users or potential high users of ER services and reporting to the Contractor; |
| 12.4.3.4 | Protocols for identifying high users of inappropriate ER services and referring them to Care Management for needs assessment and identification of other more appropriate services and resources; |
| 12.4.3.5 | Process for ensuring the provision of physical and Behavioral Health Services in an appropriate setting upon identification of the need. |
| 12.4.3.6 | Quarterly reporting on ER services Utilization; and |
| 12.4.3.7 | Process for monitoring and evaluating program effectiveness, identifying issues, and modifying the ER quality initiative program as necessary to improve service Utilization. |
| 12.4.4 | The Contractor shall submit its ER quality initiative program to ASES as part of its QAPI program. |
| 12.5 | Quality Incentive Program |
| 12.5.1 | The Contractor shall establish and implement a quality incentive program as a mechanism to improve the quality of services provided to Enrollees. |
| 12.5.2 | The Quality Incentive Program shall consist of three (3) categories of performance indicators: performance measures, preventive clinical program measures, and ER Utilization measures. |
| 12.5.3 | ASES shall establish a Retention Fund, whereby, per Section 22.3, ASES shall withhold a portion of annual PMPM Payments otherwise payable to the Contractor in order to incent the Contractor to meet performance targets under the quality incentive program described in the Quality Improvement Procedure Manual (the “QIP Manual”). The QIP Manual is subject to review and revision on an annual basis. The QIP Manual will be provided to the Contractor prior to the Effective Date of this Contract. The Retention Fund shall be reimbursed to the Contractor when a determination is made by ASES that the Contractor has complied with the quality standards and criteria established by ASES in accordance with Section 22.3 of this Contract. |
Page 171 of 301
| 12.5.4 | The following is a description of each of the three (3) categories of performance indicators and the associated reimbursement level for each. |
| 12.5.4.1 | Performance Measures |
| 12.5.4.1.1 | The Contractor shall demonstrate an improvement in performance, as described in the QIP Manual each year, using the previous year Data as the baseline for each region, in the following performance measures for effectiveness of medical care and Access: |
| 12.5.4.1.1.1 | Breast cancer screening; |
| 12.5.4.1.1.2 | Cervical cancer screening; |
| 12.5.4.1.1.3 | Cholesterol management; |
| 12.5.4.1.1.4 | Diabetes care management; |
| 12.5.4.1.1.5 | Access to preventive care visits; |
| 12.5.4.1.1.6 | Access to dental preventive care visits; |
| 12.5.4.1.1.7 | Timeliness in pre-natal care; |
| 12.5.4.1.1.8 | Asthma management; |
| 12.5.4.1.1.9 | Antidepressant medication management |
| 12.5.4.1.1.9.1 | Follow-up care for children with prescribed ADHD medication; |
| 12.5.4.1.1.9.2 | Follow-up after hospitalization for mental illness and engagement of alcohol and other drug dependence treatment; |
| 12.5.4.1.1.9.3 | Identification of alcohol and other drug treatment services; and |
| 12.5.4.1.1.9.4 | Behavioral Health Utilization. |
Page 172 of 301
| 12.5.4.1.2 | ASES shall reimburse the Contractor, in accordance with Section 22.3, and the QIP Manual QIP for successful compliance with the above performance measures based upon annual evaluation of this criterion. |
| 12.5.4.2 | Preventive Clinical Programs |
| 12.5.4.2.1 | The Contractor shall comply with the objectives of each of the following preventive clinical programs as described throughout this Contract: |
| 12.5.4.2.1.1 | Care Management; |
| 12.5.4.2.1.2 | Disease Management; |
| 12.5.4.2.1.3 | Wellness Program |
| 12.5.4.2.1.4 | Pre-natal and Maternal Program; |
| 12.5.4.2.1.5 | Provider continuing education curriculum program (see Section 10.2.2 of the Contract);); and |
| 12.5.4.2.1.6 | Physician Incentive Plan. |
| 12.5.4.2.2 | ASES shall reimburse the Contractor, in accordance with Section 22.3, and the QIP Manual for successful compliance with the above performance measures based upon annual evaluation of this criterion. |
| 12.5.4.3 | Emergency Room Use Indicators |
| 12.5.4.3.1 | As described in Section 12.4 above, the Contractor shall develop an ER Quality Incentive Program to reduce the inappropriate use of ER services for non-emergency situations. The Contractor shall be evaluated based on the effectiveness of its program. The benchmark to be applied for each Service Region shall be provided to the Contractor in the QIP Manual (Attachment 19). |
| 12.5.4.3.2 | ASES shall reimburse the Contractor, in accordance with Section 22.3, and the QIP Manual for successful compliance with the above performance measures based upon annual evaluation of this criterion. |
| 12.5.4.4 | The Contractor shall submit its quality incentive program as part of its QAPI program. The program description shall include, at a minimum: |
Page 173 of 301
| 12.5.4.4.1 | How the Contractor will educate Providers regarding the program requirements; and |
| 12.5.4.4.2 | Strategies for ensuring and monitoring program compliance. |
| 12.5.5 | During the Contract Term, ASES may issue from time to time normative or policy letters setting forth the terms and conditions it may deem necessary for the purpose of implementing the Quality Incentive Program described in this Article 12. |
| 12.5.6 | The Contractor shall contract with a certified HEDIS auditor to validate the processes of the Contractor. For Medicaid and CHIP Eligibles, the validation procedures shall be consistent with Federal requirements specified at 42 CFR 438.358(b)(2). |
| 12.5.7 | When requested, the Contractor shall submit Data to ASES for standardized performance measures, within specified timelines and according to the established procedures Data collection and reporting. The Contractor shall collect valid and reliable Data, using qualified staff and personnel to collect the Data. Failure of the Contractor to follow Data collection and reporting requirements may result in sanctions. |
| 12.5.8 | Wellness Plan |
| 12.5.8.1 | In order to advance the goals of strengthening Preventive Services, providing integrated physical, Behavioral Health, and dental services to all Eligible Persons, and educating Enrollees on health and wellness, the Contractor shall develop a Wellness Plan. |
| 12.5.8.2 | The Wellness Plan shall include a strategy for coordination with government agencies of Puerto Rico integral to disease prevention efforts and education efforts, including the Health Department, the Department of the Family, and the Department of Education. The Wellness Plan shall incorporate strategies to reach all Enrollees including those living in remote areas of the Contractor’s Service Regions. |
| 12.5.8.3 | The Wellness Plan shall present strategies for encouraging Enrollees to: |
| 12.5.8.3.1 | Seek an annual health checkup; |
| 12.5.8.3.2 | Appropriately use the services of the GHP, including GHP Service Line; |
| 12.5.8.3.3 | Seek women’s health screenings including mammograms, pap smears, cervical screenings, and tests for sexually transmitted infections; |
Page 174 of 301
| 12.5.8.3.4 | Maintain a healthy body weight, through good nutrition and exercise; |
| 12.5.8.3.5 | Seek an annual dental exam; |
| 12.5.8.3.6 | Seek Behavioral Health screening; |
| 12.5.8.3.7 | Attend to the medical and developmental needs of children and adolescents, including vaccinations; and |
| 12.5.8.3.8 | Receive education regarding the diagnosis and treatment of high-risk diagnoses including: |
| 12.5.8.3.8.1 | Depression; |
| 12.5.8.3.8.2 | Schizophrenia; |
| 12.5.8.3.8.3 | Bipolar disorders; |
| 12.5.8.3.8.4 | Attention Deficit Disorder and Attention Deficit Hyperactivity Disorder; |
| 12.5.8.3.8.5 | Substance abuse; and |
| 12.5.8.3.8.6 | Anxiety disorders. |
| 12.5.8.3.9 | The Contractor shall ensure that its Wellness Plan reaches, at a minimum, eighty-five percent (85%) of GHP Enrollees. To achieve the eighty-five (85%) goal, the Contractor shall, in compliance with the requirements of HIPAA and the rules and regulations thereunder, utilize wellness advertisements, campaigns and/or seminars, including without limitation, health fairs, educational activities, visits to enrollees, and others. |
| 12.5.8.4 | The Contractor shall, according to the timeframe specified in Attachment 12 to this Contract, present its Wellness Plan to ASES for review and prior written approval. |
| 12.6 | Provider and Enrollee Satisfaction Surveys |
| 12.6.1 | The Contractor shall perform an annual satisfaction survey for Providers and Enrollees. The survey for Enrollees shall be the Consumer Assessment of Health Care Providers and Systems (“CAHPS”) and the Experience of Care and Health Outcomes (“ECHO”) survey instruments. |
| 12.6.2 | The sample size for both surveys shall equal the number of respondents needed for a statistical confidence level of ninety-five percent (95%) with a margin of error not more than five percent (5%) and shall not have a response rate less than fifty percent (50%). |
Page 175 of 301
| 12.6.3 | The results of the surveys shall be submitted to ASES and to the Puerto Rico Medicaid Program. |
| 12.6.4 | The Contractor shall have a process for notifying Providers and Enrollees about the availability of survey findings and making survey findings available upon request. |
| 12.6.5 | The Contractor shall have a process for utilizing the results of the Provider and Enrollee surveys for monitoring service delivery and quality of services and for making program enhancements. |
| 12.7 | External Quality Review |
| 12.7.1 | In compliance with Federal requirements at 42 CFR 438.358(b)(3), ASES will contract with an External Quality Review Organization (“EQRO”) to conduct annual, external, independent reviews of the quality outcomes, timeliness of, and Access to, the services covered in this Contract. The Contractor shall collaborate with ASES’s EQRO to develop studies, surveys, and other analytic activities to assess the quality of care and services provided to Enrollees and to identify opportunities for program improvement. To facilitate this process the Contractor shall supply Data, including but not limited to Claims Data and Medical Records, to the EQRO. Upon the request of ASES, the Contractor shall provide its protocols for providing Information, participating in review activities, and using the results of the reviews to improve the quality of the services and programs provided to Enrollees. |
| 12.7.2 | The EQRO shall also audit the Contractor’s Performance Improvement Projects (“PIPs”), performance measure program, and the Contractor’s performance against quality standards based on CMS criteria. The Contractor shall cooperate fully with the EQRO. |
| 12.7.3 | The Contractor shall participate with the EQRO in various other tasks and projects identified by ASES to gauge performance in a variety of areas, including the integration of physical and Behavioral Health, care coordination, and treatment of special populations. |
| 12.7.4 | The EQRO retained by ASES shall not be a competitor of the Contractor and shall comply with 42 C.F.R. § 438.354. |
| 12.7.5 | Disease Management |
| 12.7.5.1 | The Contractor shall provide Disease Management (“DM”) strategies to Enrollees with identified Chronic Conditions as part of its wellness programs and activities. The Contractor’s DM strategies may include population identification/stratification, collaborative practice models, patient self-management education, evidence-based practice guidelines, process and outcomes measurements, and internal quality improvement processes. |
Page 176 of 301
| 12.7.5.2 | The Contractor shall improve its ability to manage Chronic illnesses/diseases/Conditions through DM protocols. The Contractor shall: |
| 12.7.5.2.1 | Participate in DM projects annually; |
| 12.7.5.2.2 | Provide comprehensive DM and a Care Manager for the following conditions: |
| 12.7.5.2.2.1 | Asthma in Adults and children; |
| 12.7.5.2.2.2 | Diabetes Type 1 and Type 2; |
| 12.7.5.2.2.3 | Congestive heart failure; |
| 12.7.5.2.2.4 | Hypertension; |
| 12.7.5.2.2.5 | Obesity; |
| 12.7.5.2.2.6 | Chronic Renal Disease Stages 1 and 2; and |
| 12.7.5.2.2.7 | Depression; |
| 12.7.5.2.3 | The DM program shall utilize strategies consistent with nationally recognized DM guidelines, such as those available through the Agency of Health Care Research and Quality’s (“AHRQ”), NQMC website, or the Care Continuum Alliance (formerly the Disease Management Association of America); |
| 12.7.5.2.4 | Submit cumulative Data-driven measurements with written analysis describing the effectiveness of its DM interventions as well as any modifications implemented by the Contractor to improve its DM performance; |
| 12.7.5.2.5 | Submit to ASES the Contractor’s DM plan, which shall include a description of the strategies and interventions, the overall and measurable objectives, and targeted interventions. The Contractor shall also submit to ASES its methodology for identifying other diseases/conditions for potential DM strategies and interventions; and |
Page 177 of 301
| 12.7.5.2.6 | Submit to ASES a quantitative and qualitative evaluation of the efficacy of the prior year’s DM strategies; document how well goals were addressed, such as identification, Enrollment, targeted interventions, and outcomes. |
ARTICLE 13 FRAUD, WASTE, AND ABUSE
| 13.1 | General Provisions |
| 13.1.1 | The Contractor shall have and implement a comprehensive internal administrative and management controls, policies, and procedures in place designed to prevent, detect, report, investigate, correct, and resolve potential or confirmed cases of Fraud, Waste, and Abuse in the administration and delivery of services detailed in this Contract. |
| 13.1.2 | For Medicaid and CHIP Eligibles, the Contractor’s internal controls, policies, and procedures shall comply with all Federal requirements regarding Fraud, Waste, and Abuse and program integrity, including but not limited to Sections 1128, 1156, and 1902(a)(68) of the Social Security Act, Section 6402(h) of PPACA, 42 CFR 438.608, the CMS Medicaid Integrity program, and the Deficit Reduction Act of 2005. The Contractor shall exercise diligent efforts to ensure that no payments are made to any person or entity that has been excluded from participation in Federal health care programs. (See State Medicaid Director Letter #09-001, January 16, 2009.) |
| 13.1.3 | The Contractor shall have surveillance and Utilization control programs and procedures (see 42 CFR 456.3, 42 CFR 456.4, 42 CFR 456.23) to safeguard against under-utilization, unnecessary or inappropriate use of Covered Services and against excess payments for Covered Services. |
| 13.1.4 | The Contractor shall have adequate staffing and resources to identify and investigate unusual incidents and develop and implement Corrective Action plans to assist the Contractor in preventing and detecting potential Fraud, Waste, and Abuse. |
| 13.1.5 | The Contractor shall establish effective lines of communication between the Contractor’s compliance officer and the Contractor’s employees to facilitate the oversight of systems that monitor service Utilization and Encounters for Fraud, Waste, and Abuse. |
| 13.1.6 | The Contractor shall submit its Fraud, Waste, and Abuse policies and procedures, its proposed compliance plan, and its program integrity plan to ASES for prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 13.1.7 | Any changes to the Contractor’s Fraud, Waste, and Abuse policies and procedures must be submitted to ASES for approval within fifteen (15) Calendar Days of the date the Contractor plans to implement the changes and the changes shall not go into effect until ASES provides prior written approval. |
Page 178 of 301
| 13.1.8 | The Contractor shall comply with all program integrity provisions of the PPACA including: |
| 13.1.8.1 | Enhanced Provider screening and enrollment, Section 6401; |
| 13.1.8.2 | Termination of Provider participation, Section 6501; |
| 13.1.8.3 | Provider discloser of current or previous affiliation with excluded Provider(s), Section 6401; and |
| 13.1.8.4 | Provider screening and enrollment, 42 CFR Part 455, Subpart E. |
| 13.1.9 | The Contractor shall inform ASES in writing Immediately upon becoming aware of a compliance breach related to the Contractor’s MCO and/or Network Provider. |
| 13.1.10 | The Contractor shall inform ASES of any meetings it holds with any other GHP MCOs related to compliance and program integrity issues at least forty-eight (48) hours prior to the meeting. The Contractor shall provide a copy of the meeting minutes as well as the results of any follow-up investigations to ASES in writing Immediately. |
| 13.1.11 | The Contractor shall have policies and procedures prior approved in writing by ASES to address (i) Immediately notifying ASES of pending Network Provider investigations, suspensions and debarment and (ii) transitioning Enrollees from suspended and debarred Network Providers. |
| 13.2 | Compliance Plan |
| 13.2.1 | The Contractor shall have a written Fraud, Waste, and Abuse compliance plan with stated program goals and objectives, program scope, and methodology to evaluate program performance. A paper and electronic copy of the compliance plan shall be provided to ASES annually for prior written approval. ASES shall provide notice of approval, denial, or modification to the Contractor within thirty (30) Calendar Days of receipt. The Contractor shall make any necessary changes required by ASES within an additional thirty (30) Calendar Days of the request. |
| 13.2.2 | At a minimum, the Contractor’s Fraud, Waste, and Abuse compliance plan shall, in accordance with 42 CFR 438.608: |
| 13.2.2.1 | Ensure that all of its officers, directors, managers and employees know and understand the provisions of the Contractor’s Fraud, Waste, and Abuse compliance plan; |
Page 179 of 301
| 13.2.2.2 | Require the designation of a compliance officer and a compliance committee that are accountable to the Contractor’s senior management; |
| 13.2.2.3 | Ensure and describe effective training and education for the compliance officer and the Contractor’s employees; |
| 13.2.2.4 | Ensure that Providers and Enrollees are educated about Fraud, Waste, and Abuse identification and reporting in the materials provided to them; |
| 13.2.2.5 | Ensure effective lines of communication between the Contractor’s compliance officer and the Contractor’s employees to ensure that employees understand and comply with the Contractor’s Fraud, Waste, and Abuse program; |
| 13.2.2.6 | Ensure enforcement of standards of conduct through well-publicized disciplinary guidelines; |
| 13.2.2.7 | Ensure internal monitoring and auditing with provisions for prompt response to potential offenses, and for the development of corrective action initiatives relating to the Contractor’s Fraud, Waste, and Abuse efforts; |
| 13.2.2.8 | Describe standards of conduct that articulate the Contractor’s commitment to comply with all applicable Puerto Rico and Federal requirements and standards; |
| 13.2.2.9 | Ensure that no individual who reports Provider violations or suspected cases of Fraud, Waste, and Abuse is retaliated against; and |
| 13.2.2.10 | Include a monitoring program that is designed to prevent and detect potential or suspected Fraud, Waste, and Abuse. This monitoring program shall include but not be limited to: |
| 13.2.2.10.1 | Monitoring the billings of its Providers to ensure Enrollees receive services for which the Contractor is billed; |
| 13.2.2.10.2 | Requiring the investigation of all reports of suspected cases of Fraud and over-billings; |
| 13.2.2.10.3 | Reviewing Providers for over, under and inappropriate Utilization; |
| 13.2.2.10.4 | Verifying with Enrollees the delivery of services as claimed; and |
Page 180 of 301
| 13.2.2.10.5 | Reviewing and trending Enrollee Complaints regarding Providers. |
| 13.2.3 | The Contractor shall include in all employee handbooks a specific discussion of its Fraud, Waste, and Abuse policies and procedures, the rights of whistleblowers, and the Contractor’s procedures for detecting and preventing Fraud, Waste, and Abuse. |
| 13.2.4 | The Contractor shall include in the Enrollee Handbook instructions on how to report Fraud, Waste, and Abuse and the protections for whistleblowers. |
| 13.3 | Program Integrity Plan |
| 13.3.1 | The Contractor shall develop a program integrity plan that at a minimum: |
| 13.3.1.1 | Defines Fraud, Waste, and Abuse; |
| 13.3.1.2 | Specifies methods to detect Fraud, Waste, and Abuse; |
| 13.3.1.3 | Describes a process to perform investigations on each suspected case of Fraud, Waste, and Abuse; |
| 13.3.1.4 | Describes the Contractor’s staff responsible for conducting the investigations and reporting of potential Fraud, Waste, or Abuse, including an organizational chart documenting roles and responsibilities; |
| 13.3.1.5 | Includes a variety of methods for identifying, investigating, and referring suspected cases to appropriate entities; |
| 13.3.1.6 | Includes a systematic approach to Data analysis; |
| 13.3.1.7 | Defines mechanisms to monitor frequency of Encounters and services rendered to Enrollees billed by Providers; |
| 13.3.1.8 | Identifies requirements to complete the preliminary investigation of Providers and Enrollees; |
| 13.3.1.9 | Include provisions regarding prompt terminations of inactive Providers due to inactivity in the past twelve (12) months; |
| 13.3.1.10 | Include a risk assessment of the Contractor’s various Fraud, Waste, and Abuse processes. The risk assessment shall include a listing of the Contractor’s top three (3) vulnerable areas and outline action plans to mitigate risks; |
Page 181 of 301
| 13.3.1.11 | Include procedures for the confidential reporting of potential Fraud, Waste, and Abuse, including potential Contractor violations; and |
| 13.3.1.12 | Include procedures to ensure that there is no retaliation against an individual who reports Contractor violations or other potential Fraud, Waste, or Abuse to the Contractor or an external entity. |
| 13.3.2 | The Contractor’s program integrity plan shall comply in all respects with the ASES Guidelines for the development of a program integrity plan, included as Attachment 14 to this Contract. Upon review of the Contractor’s Program Integrity Plan (see Section 13.3), ASES will promptly (within twenty (20) Business Days) notify the Contractor of any needed revisions in order for the program integrity plan to comply with the guidelines and with Federal law. The Contractor, in turn, shall promptly (within twenty (20) Business Days of receipt of the ASES comments) re-submit its Plan for ASES review and prior written approval. |
| 13.3.3 | The Contractor shall notify ASES within twenty (20) Business Days of any initiated investigation of a suspected case of Fraud, Waste, or Abuse. The Contractor shall subsequently report preliminary results of such investigations activities to ASES and other appropriate Puerto Rico and Federal entities. ASES will provide the Contractor with guidance during the pendency of the investigation and will refer the matter to the US Department of Justice. |
| 13.4 | Prohibited Affiliations with Individuals Debarred by Federal Agencies |
| 13.4.1 | The Contractor shall not knowingly have a relationship with the following: |
| 13.4.1.1 | An individual who is debarred, suspended, or otherwise excluded from participating in procurement activities under the Federal Acquisition Regulation or from participating in non-procurement activities under Executive Order No. 12549 or under any guidelines implementing the Executive Order. |
| 13.4.1.2 | An individual who is an Affiliate, as defined in the Federal Acquisition Regulation, of a person described in Section 13.4.1.1. The relationship is defined as follows: |
| 13.4.1.2.1 | A director, officer, or partner of the Contractor; |
| 13.4.1.2.2 | A person with beneficial ownership of five percent (5%) or more of the Contractor’s equity; or |
| 13.4.1.2.3 | A person with an employment, consulting, or other arrangement with the Contractor for the provision of items or services that are significant and material the Contractor’s obligations under this Contract. |
Page 182 of 301
| 13.5 | Reporting and Investigations |
| 13.5.1 | The Contractor shall cooperate with all duly authorized Federal and Puerto Rico agencies and representatives in reporting, investigating and prosecuting Fraud, Waste, and Abuse. |
| 13.5.2 | The Contractor shall have methods for identifying, investigating, and referring suspected Fraud, Waste, and Abuse pursuant to 42 CFR 455.1, 42 CFR 455.13, 42 CFR 455.14 and 42 CFR 455.21 and Immediately notifying ASES. |
| 13.5.3 | The Contractor shall Immediately report to ASES the identity of any Provider or other person who is debarred, suspended, or otherwise prohibited from participating in procurement activities. ASES shall promptly notify the Secretary of Health and Human Services of the noncompliance, as required by 42 CFR 438.610(c). |
| 13.5.4 | The Contractor shall conclude its preliminary investigation within ten (10) Business Days of identifying the potential Fraud, Waste, or Abuse and shall provide the findings of its preliminary investigation in writing to ASES within two (2) Business Days of completing the preliminary investigation. |
| 13.5.5 | If directed by ASES, the Contractor shall conduct a full investigation. The Contractor shall provide the results of its full investigations in writing to Puerto Rico and ASES within two (2) Business Days of completing the investigation. This report shall include any referrals made and actions taken by the Contractor or any external entity. |
| 13.5.6 | The Contractor and all Subcontractors shall cooperate fully with Federal and Puerto Rico agencies in Fraud, Waste, and Abuse investigations and subsequent legal actions, whether administrative, civil, or criminal. Such cooperation shall include actively participating in meetings, providing requested Information, access to records, and access to interviews with employees and consultants, including but not limited to those with expertise in the administration of the program and/or medical or pharmaceutical matters or in any matter related to an investigation or prosecution. Such cooperation shall also include providing personnel to testify at any hearings, trials, or other legal proceedings on an as-needed basis. |
| 13.5.7 | In accordance with Section 6402 of the PPACA and 42 CFR 455.23, the Contractor must have a mechanism in place to identify and suspend payments to any Provider or other Subcontractor when there is a pending investigation of a Credible Allegation of Fraud under the Medicaid program. |
Page 183 of 301
| 13.5.8 | If a Provider is suspended or terminated from participation in the Puerto Rico Medicaid Program by Puerto Rico, the Contractor shall also suspend or terminate the Provider. |
| 13.5.9 | If a Provider is terminated from Medicare or another state’s Medicaid or State Children’s Health Insurance Program, the Contractor shall terminate its Provider participation agreement with that Provider (see Section 1902(a)(39) of the Social Security Act and 42 CFR 455.416). |
| 13.5.10 | The Contractor shall notify ASES within two (2) Business Days of taking any action against a Provider for program integrity reasons, including, but not limited to, denial of a Provider Credentialing/Re-Credentialing application, corrective action or limiting the ability of a Provider to participate in the program (e.g., suspending or terminating a Provider). The notification shall include but not be limited to identification of the Provider and a description of the action, the reason for the action, and documentation to support the reason. The Contractor shall provide additional Information upon ASES’s request. |
| 13.5.11 | The Contractor shall submit a risk assessment on an “as needed” basis and Immediately after a program integrity-related action against a Provider. The Contractor shall inform ASES of such action and provide details of such financial action. |
| 13.5.12 | The Contractor shall immediately disclose to ASES any and all criminal convictions of its managing employees (see 42 CFR 455.106). |
| 13.5.13 | Regarding Provider disclosers, the Contractor shall: |
| 13.5.13.1 | Not make payment to a Provider unless the Provider has submitted completed disclosures required by Federal law either to ASES or the Contractor. This includes but is not limited to disclosure regarding ownership and control, business transactions, and criminal convictions (see 42 CFR Part 455, Subpart B). |
| 13.5.13.2 | Track information received from ASES identifying Providers from whom ASES has received completed disclosures. |
| 13.5.13.3 | For participating Providers for whom ASES has not received completed disclosures, as reported to the Contractor, collect and retain completed Provider disclosures as part of initial Credentialing and then annually, using a disclosure form prior approved by ASES in writing. |
Page 184 of 301
| 13.5.13.4 | In accordance with 42 CFR 455.106, Immediately report any criminal conviction disclosures to ASES and explain what action it will take (e.g., terminate the Provider). |
| 13.5.13.5 | In accordance with Section 1866(j)(5) of the Social Security Act and implementing regulations, as part of Credentialing and Re-Credentialing, collect disclosures from Out-of-Network Providers regarding any current or previous affiliations with a Provider or supplier that has uncollected debt, has been or is subject to a payment suspension under a Federal health care program (as defined in Section 1128B(f)), has been excluded from participation under Medicare, Medicaid, CHIP, or has had its billing privileges denied or revoked. The Contractor shall notify ASES if the Contractor determines that such affiliation poses an undue risk of Fraud, Waste, or Abuse and denies the application. |
| 13.6 | Service Verification with Enrollees |
| 13.6.1 | In accordance with 42 CFR 455.20, the Contractor shall implement a process for verifying with Enrollees whether services billed by Providers were received. |
| 13.6.2 | The Contractor must employ a methodology and sampling process prior approved by ASES to verify with its Enrollees on a monthly whether services billed to the Contractor by Providers were actually received. The methodology and sampling process must include criteria for identifying “high-risk” services and Provider types. |
| 13.6.3 | Stark Law Compliance. The Contractor must have mechanisms in place to ensure that payments are not made in violation of Section 1903(s) of the Social Security Act with respect to certain physician Referrals as defined in Section 1877 of the Social Security Act. The Contractor shall ensure that disclosing Parties provide a financial analysis that includes the total amount actually or potentially due and owed as a result of the disclosed violation, a description of the methodology used to determine the amount due and owing, the total amount of remuneration involved physicians (or an immediate family member of such physicians) received as a result of an actual or potential violation, and a summary of audit activity and documents used in the audit. In accordance with Section 6409 of the PPACA, the Contractor will encourage provider use of the self-referral disclosure protocols, under which providers of services and suppliers may self-disclose actual or potential violations of the physicians’ self-referral statute (Section 1877 of the Social Security Act). |
Page 185 of 301
ARTICLE 14 GRIEVANCE SYSTEM
| 14.1 | General Requirements |
| 14.1.1 | In accordance with 42 CFR Part 438, Subpart F, the Contractor shall establish an internal Grievance System under which Enrollees, or Providers acting on their behalf, may challenge the denial of coverage of, or payment for, Covered Services. |
| 14.1.2 | The Contractor’s Grievance System shall include (i) a Complaint process, (ii) Grievance process, (iii) Appeal process, and (iv) access to the Administrative Law Hearing process. |
| 14.1.3 | The Contractor shall designate, in writing, an officer who shall have primary responsibility for ensuring that Complaints, Grievances, and Appeals are resolved pursuant to this Contract and for signing all Notices of Action. For such purposes, an officer shall mean a president, vice president, secretary, treasurer, chairperson of the board of directors of the Contractor’s organization, the sole proprietor, the managing general partner of a partnership, or a person having similar executive authority in the organization. |
| 14.1.4 | The Contractor shall develop a written Grievance System and the policies and procedures that detail the operation of the Grievance System. The Grievance System policies and procedures shall be submitted to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 14.1.5 | At a minimum, the Contractor’s Grievance System policies and procedures shall include the following: |
| 14.1.5.1 | Process for filing a Complaint, Grievance, or Appeal, or seeking an Administrative Law Hearing; |
| 14.1.5.2 | Process for receiving, recording, tracking, reviewing, reporting, and resolving Grievances filed verbally, in writing, or in-person; |
| 14.1.5.3 | Process for receiving, recording, tracking, reviewing, reporting, and resolving Appeals filed verbally or in writing; |
| 14.1.5.4 | Process for requesting an expedited review of an Appeal; |
| 14.1.5.5 | Process and timeframe for a Provider to file a Complaint, Grievance or Appeal on behalf of an Enrollee; |
| 14.1.5.6 | Process for notifying Enrollees of their right to file a Complaint, Grievance, or Appeal with the Patient Advocate Office and how to contact the Patient Advocate Office; |
Page 186 of 301
| 14.1.5.7 | Procedures for the exchange of Information with Providers, ASES, and the Enrollees regarding Complaints, Grievances, and Appeals; |
| 14.1.5.8 | Process and timeframes for notifying Enrollees in writing regarding receipt of Complaints, Grievances, Appeals, resolution, action, delay of review, and denial of request for expedited review. |
| 14.1.6 | The Contractor’s Grievance System shall fully comply with the Patient’s Bill of Rights Act and with Act No. 11 of April 11, 2001 (known as the Organic Law of the Office of the Patient Advocate), to the extent that such provisions do not conflict with, or pose an obstacle to Federal regulations. |
| 14.1.7 | The Contractor shall process each Complaint, Grievance, or Appeal in accordance with applicable Puerto Rico and Federal statutory and regulatory requirements, this Contract, and the Contractor’s written policies and procedures. Pertinent facts from all Parties must be collected during the process. |
| 14.1.8 | The Contractor shall include educational information in the Enrollee Handbook regarding the Contractor’s Grievance System which at a minimum includes: |
| 14.1.8.1 | A description of the Contractor’s Grievance System; |
| 14.1.8.2 | Instructions on how to file Complaints, Grievances and Appeals including the timeframes for filing; |
| 14.1.8.3 | The Contractor’s toll-free telephone number and office hours; |
| 14.1.8.4 | Information regarding an Enrollee’s right to file a Complaint, Grievance, or Appeal with the Patient Advocate Office and how to file a Complaint, Grievance, or Appeal with the Patient Advocate Office; |
| 14.1.8.5 | Information describing the Administrative Law Hearing process and governing rules, including that the Enrollee must first exhaust the MCO’s Grievance System before accessing the Administrative Law Hearing process; and |
| 14.1.8.6 | Timelines and limitations associated with filing Grievances or Appeals. |
| 14.1.9 | The Contractor shall give Enrollees reasonable assistance in completing forms and taking other procedural steps for Complaints, Grievances and Appeals. This includes, but is not limited to, providing interpreter services and toll-free numbers that have adequate TDD and interpreter capability. |
| 14.1.10 | The Contractor shall include information regarding the Grievance System in the Provider Guidelines and upon joining the Contractor’s Network, all Providers shall receive training and education regarding the Contractor’s Grievance System, which includes but is not limited to: |
Page 187 of 301
| 14.1.10.1 | The Enrollee’s right to file Complaints, Grievances and, Appeals and the requirements and timeframes for filing; |
| 14.1.10.2 | The Enrollee’s right to file a Complaint, Grievance, or Appeal with the Patient Advocate Office; |
| 14.1.10.3 | The Enrollee’s right to an Administrative Law Hearing, how to obtain an Administrative Law Hearing, and representation rules at an Administrative Law Hearing; |
| 14.1.10.4 | The availability of assistance in filing a Complaint, Grievance, or Appeal; |
| 14.1.10.5 | The toll-free numbers to file oral Complaints, Grievances, and Appeals; |
| 14.1.10.6 | The Enrollee’s right to request continuation of Benefits during an Appeal, or an Administrative Law Hearing filing, and that if the Contractor’s Action is upheld in an Administrative Law Hearing, the Enrollee may be liable for the cost of any continued Benefits; and |
| 14.1.10.7 | Any Puerto Rico-determined Provider Appeal rights to challenge the failure of the Contractor to cover a service. |
| 14.1.11 | The Contractor shall have procedures in place to notify all Enrollees in their primary language of Complaint, Grievance, and Appeal dispositions. |
| 14.1.12 | The Contractor shall develop Grievance System forms to be submitted for prior written approval by ASES according to the timeframe specified in Attachment 12 to this Contract. The approved forms shall be made available to all Enrollees, shall meet all requirements listed in Sections 6.2 and 6.3 for written materials, and shall, at a minimum: |
| 14.1.12.1 | Instruct the Enrollee or Enrollee’s Authorized Representative that documentary evidence should be included, if available; and |
| 14.1.12.2 | Include instructions for completion and submission. |
| 14.1.13 | All ASES prior approved Complaints, Grievances, and Appeals files and forms shall be made available to ASES for auditing. All Complaint, Grievance, and Appeal documents and related information shall be considered as containing protected health information and shall be treated in accordance with HIPAA regulations and other applicable laws of Puerto Rico. |
Page 188 of 301
| 14.1.14 | The Contractor shall ensure that the individuals who make decisions on Grievances and Appeals are individuals: |
| 14.1.14.1 | Who were not involved in any previous level of review or decision-making; and |
| 14.1.14.2 | Who, if deciding any of the following, are Providers who have the appropriate clinical expertise, as determined by ASES, in treating the Enrollee’s condition or disease if deciding any of the following: |
| 14.1.14.3 | An Appeal of a denial that is based on lack of Medical Necessity; |
| 14.1.14.4 | A Grievance regarding denial of expedited resolutions of Appeal; and |
| 14.1.14.5 | Any Grievance or Appeal that involves clinical issues. |
| 14.1.15 | The Contractor shall ensure that punitive action is not taken against a Provider who requests a Grievance, Appeal or an Administrative Law Hearing or supports an Enrollee’s Grievance, Appeal or Administrative Law Hearing. |
| 14.1.16 | The Contractor shall have a system in place to collect, analyze, and integrate Data regarding Complaints, Grievances, and Appeals. At a minimum, the following information shall be recorded: |
| 14.1.16.1 | Date Complaint, Grievance, or Appeal was filed; |
| 14.1.16.2 | Enrollee’s name; |
| 14.1.16.3 | Enrollee’s Medicaid ID number, if applicable; |
| 14.1.16.4 | Name of the individual filing the Complaint, Grievance, or Appeal on behalf of the Enrollee; |
| 14.1.16.5 | Date of acknowledgement that receipt of Grievance or Appeal was mailed to the Enrollee; |
| 14.1.16.6 | Summary of Complaint, Grievance, or Appeal; |
| 14.1.16.7 | Date Notice of Disposition or Notice of Adverse Action was mailed to the Enrollee; |
| 14.1.16.8 | Corrective Action required; and |
| 14.1.16.9 | Date of resolution. |
Page 189 of 301
| 14.2 | Complaint |
| 14.2.1 | The Complaint process is the procedure for addressing Enrollee Complaints, defined as expressions of dissatisfaction about any matter other than an Action that are resolved at the point of contact rather than through filing a formal Grievance. |
| 14.2.2 | An Enrollee or Enrollee’s Authorized Representative may file a Complaint either orally or in writing. The Enrollee or Enrollee’s Authorized Representative may follow-up an oral request with a written request. However, the timeframe for resolution begins with the date the Contractor receives the oral request. |
| 14.2.3 | An Enrollee or Enrollee’s Authorized Representative shall file a Complaint within fifteen (15) Calendar Days after the date of occurrence that initiated the Complaint. |
| 14.2.4 | The Contractor shall have procedures in place to provide Notice of Dispositions of Complaints to all Enrollees in their primary language. |
| 14.2.5 | The Contractor shall resolve each Complaint within seventy-two (72) hours of the time the Contractor received the initial Complaint, whether orally or in writing. If the Complaint is not resolved within this timeframe, the Complaint shall be treated as a Grievance. |
| 14.2.6 | The Notice of Disposition shall include the results and date of the resolution of the Complaint and shall include notice of the right to file a Grievance or Appeal and information necessary to allow the Enrollee to request an Administrative Law Hearing, if appropriate, including contact information necessary to pursue an Administrative Law Hearing. |
| 14.3 | Grievance Process |
| 14.3.1 | An Enrollee or Enrollee’s Authorized Representative may file a Grievance with the Contractor or with the Office of the Patient’s Advocate of Puerto Rico either orally or in writing. A Provider cannot file a Grievance on behalf of an Enrollee unless written consent is granted by the Enrollee. |
| 14.3.2 | The timeframe for filing a Grievance shall not exceed ninety (90) Calendar Days from the date of the occurrence. |
| 14.3.3 | The Contractor shall acknowledge receipt of each Grievance in writing to the Enrollee (and the Provider, if the Provider filed the Grievance on the Enrollee’s behalf) within ten (10) Business Days of receipt. |
| 14.3.4 | The Contractor shall provide written notice of the disposition of the Grievance as expeditiously as the Enrollee’s health condition requires, but in any event, within ninety (90) Calendar Days from the day the Contractor receives the Grievance. |
Page 190 of 301
| 14.3.5 | The Notice of Disposition shall include the following: |
| 14.3.5.1 | The resolution of the Grievance, |
| 14.3.5.2 | The basis for the resolution, and |
| 14.3.5.3 | The date of the resolution. |
| 14.3.6 | The Contractor may extend the timeframe to provide a written notice of disposition of a Grievance for up to fourteen (14) Calendar Days if the Enrollee requests the extension or the Contractor demonstrates (to the satisfaction of ASES, upon its request) that there is a need for additional Information and how the delay is in the Enrollee’s interest. If the Contractor extends the timeframe, it shall, for any extension not requested by the Enrollee, give the Enrollee written notice of the reason for the delay prior to the delay. |
| 14.4 | Notice of Action |
| 14.4.1 | Pursuant to 42 CFR 438.210(c), the Contractor shall provide written notice to the requesting Provider and the Enrollee of any decision by the Contractor to deny a Service Authorization Request, or to authorize a service in an amount, duration, or scope that is less than requested. The Contractor’s notices shall meet the requirements of 42 CFR 438.404. |
| 14.4.2 | The Contractor’s written Notice of Action to Enrollees must meet the language and format requirements in Section 6.2 and 6.3 and be set in accordance with the timeframes described in Section 14.4.4. |
| 14.4.3 | The Notice of Action shall contain the following: |
| 14.4.3.1 | The Action the Contractor has taken or intends to take; |
| 14.4.3.2 | The reasons for the Action; |
| 14.4.3.3 | The Enrollee’s right to file an Appeal through the Contractor’s internal Grievance System and the procedure for filing an Appeal; |
| 14.4.3.4 | The Enrollee’s right to request an Administrative Law Hearing after exhaustion of the Contractor’s Grievance System; |
| 14.4.3.5 | The Enrollee’s right to allow a Provider to file an Appeal or an Administrative Law Hearing on behalf of the Enrollee, upon written consent; |
| 14.4.3.6 | The circumstances under which expedited review is available and how to request it; and |
Page 191 of 301
| 14.4.3.7 | The Enrollee’s right to have Benefits continue pending resolution of the Appeal with the Contractor or during the Administrative Law Hearing, how to request that Benefits be continued, and the circumstances under which the Enrollee may be required to pay for the costs of these services. |
| 14.4.4 | The Contractor shall mail the Notice of Action within the following timeframes: |
| 14.4.4.1 | For termination, suspension, or reduction of previously authorized Covered Services, at least ten (10) Calendar Days before the date of Action or no later than the date of Action except in the event of one of the following exceptions: |
| 14.4.4.1.1 | The Contractor has factual Information confirming the death of an Enrollee. |
| 14.4.4.1.2 | The Contractor receives a clear written statement signed by the Enrollee that he or she no longer wishes to receive services or gives Information that requires termination or reduction of services and indicates that he or she understands that this must be the result of supplying that Information. |
| 14.4.4.1.3 | The Enrollee’s whereabouts are unknown and the post office returns the Contractor’s mail directed to the Enrollee indicating no forwarding address (refer to 42 CFR 431.231(d) for procedures if the Enrollee’s whereabouts become known). |
| 14.4.4.1.4 | The Enrollee’s Provider prescribes a change in the level of medical care. |
| 14.4.4.1.5 | The date of Action will occur in less than ten (10) Calendar Days in accordance with 42 CFR 483.12(a)(5)(ii). |
| 14.4.4.1.6 | The Contractor may shorten the period of advance notice to five (5) Calendar Days before the date of Action if the Contractor has facts indicating that Action should be taken because of probable Enrollee Fraud and the facts have been verified, if possible, through secondary sources. |
| 14.4.4.2 | For denial of payment, at the time of any Action affecting the Claim. |
| 14.4.4.3 | For standard authorization decisions that deny or limit Covered Services within the timeframes required in Section 11.4. |
Page 192 of 301
| 14.4.4.4 | If the Contractor extends the timeframe for the authorization decision and issuance of Notice of Action according to Section 14.4.3, the Contractor shall give the Enrollee written notice of the reasons for the decision to extend if he or she did not request the extension. The Contractor shall issue and carry out its determination as expeditiously as the Enrollee’s health requires and no later than the date the extension expires. |
| 14.4.4.5 | For authorization decisions not reached within the timeframes required in Section 11.4 for either standard or expedited authorizations, the Notice of Action shall be mailed on the date the timeframe expires, as this constitutes a denial and is thus an Action. |
| 14.5 | Appeal Process |
| 14.5.1 | The Enrollee, the Enrollee’s Authorized Representative, or the Provider may file an Appeal either orally or in writing. |
| 14.5.2 | Oral inquiries seeking to appeal an Action are treated as Appeals (to establish the earliest possible filing date for the Appeal), but Enrollees must confirm oral requests for Appeals in writing within ten (10) Calendar Days of the oral filing, unless the Enrollee requests expedited resolution, then no additional follow-up is required. |
| 14.5.3 | The requirements of the Appeal process shall be binding for all types of Appeals, including expedited Appeals, unless otherwise established for expedited Appeals. |
| 14.5.4 | The Enrollee, the Enrollee’s Authorized Representative, or the Provider acting on behalf of the Enrollee with the Enrollee’s written consent, may file an Appeal to the Contractor within sixty (60) Calendar Days from the date on the Contractor’s Notice of Action. |
| 14.5.5 | Appeals shall be filed directly with the Contractor, or its delegated representatives. The Contractor may delegate this authority to an Appeal committee, but the delegation shall be in writing. |
| 14.5.6 | The Appeals process shall provide the Enrollee, the Enrollee’s Authorized Representative, or the Provider acting on behalf of the Enrollee with the Enrollee’s written consent, a reasonable opportunity to present evidence and allegations of fact or law, in person, as well as in writing. The Contractor shall inform the Enrollee of the limited time available to provide this in case of expedited review. |
| 14.5.7 | The Appeals process shall provide the Enrollee, the Enrollee’s Authorized Representative, or the Provider acting on behalf of the Enrollee with the Enrollee’s written consent, opportunity, before and during the Appeals process, to examine the Enrollee’s case file, including Medical Records, and any other documents and records considered during the Appeals process and provide copies of documents contained therein without charge. |
Page 193 of 301
| 14.5.8 | The Appeals process shall include as Parties to the Appeal the Enrollee, the Enrollee’s Authorized Representative, the Provider acting on behalf of the Enrollee with the Enrollee’s written consent, or the legal representative of a deceased Enrollee’s estate. |
| 14.5.9 | The Contractor shall resolve each standard Appeal and provide written notice of the disposition, as expeditiously as the Enrollee’s health condition requires but no more than forty-five (45) Calendar Days from the date the Contractor receives the Appeal. |
| 14.5.10 | The Contractor shall establish and maintain an expedited review process for Appeals, subject to prior written approval by ASES, when the Contractor determines (based on a request from the Enrollee) or the Provider indicates (in making the request on the Enrollee’s behalf) that taking the time for a standard resolution could seriously jeopardize the Enrollee’s life or health or ability to attain, maintain, or regain maximum function. The Enrollee, the Enrollee’s Authorized Representative, or the Provider acting on behalf of the Enrollee with the Enrollee’s written consent, may file an expedited Appeal either orally or in writing. |
| 14.5.11 | The Contractor shall resolve each expedited Appeal and provide a written Notice of Disposition, as expeditiously as the Enrollee’s health condition requires, but no longer than three (3) Business Days after the Contractor receives the Appeal and make reasonable efforts to provide oral notice. |
| 14.5.12 | If the Contractor denies an Enrollee’s request for expedited review, it shall utilize the timeframe for standard Appeals specified herein and shall make reasonable efforts to give the Enrollee prompt oral notice of the denial, and follow-up within two (2) Calendar Days with a written notice. If the Enrollee disagrees with the decision to extend the prescribed timeframe, he or she has the right to file a Grievance and the Grievance shall be resolved within twenty-four (24) hours. The Contractor shall also make reasonable efforts to provide oral notice for resolution of an expedited review of an Appeal. |
| 14.5.13 | The Contractor may extend the timeframe for standard or expedited resolution of the Appeal by up to fourteen (14) Calendar Days if the Enrollee, Enrollee’s Authorized Representative, or the Provider acting on behalf of the Enrollee with the Enrollee’s written consent, requests the extension or the Contractor demonstrates (to the satisfaction of ASES, upon its request) that there is need for additional information and how the delay is in the Enrollee’s interest. If the Contractor extends the timeframe, it shall, for any extension not requested by the Enrollee, give the Enrollee written notice of the reason for the delay. The Contractor shall inform the Enrollee of the right to file a Grievance if the Enrollee disagrees with the decision to extend the timeframe. |
Page 194 of 301
| 14.5.14 | The Contractor shall provide written Notice of Disposition of an Appeal to the Enrollee (and the Provider, if the Provider filed the Appeal on the Enrollee’s behalf) as well as a copy to ASES within two (2) Business Days of the resolution. |
| 14.5.15 | The written notice of Disposition shall include: |
| 14.5.15.1 | The results and date of the Appeal resolution; and |
| 14.5.15.2 | For decisions not wholly in the Enrollee’s favor: |
| 14.5.15.3 | The right to request an Administrative Law Hearing; |
| 14.5.15.4 | How to request an Administrative Law Hearing; |
| 14.5.15.5 | The right to continue to receive Benefits pending an Administrative Law Hearing; |
| 14.5.15.6 | How to request the continuation of Benefits; and |
| 14.5.15.7 | Notification that if the Contractor’s Action is upheld in a hearing, the Enrollee may liable for the cost of any continued Benefits. |
| 14.6 | Administrative Law Hearing |
| 14.6.1 | The Contractor is responsible for explaining the Enrollee’s right to and the procedures for an Administrative Law Hearing, including that the Enrollee must exhaust the Contractor’s Grievance, Complaints, and Appeals process before requesting an Administrative Law Hearing. |
| 14.6.2 | The parties to the Administrative Law Hearing include the Contractor as well as the Enrollee or his or her Authorized Representative, or the representative of a deceased Enrollee’s estate. |
| 14.6.3 | If the Contractor takes an Action, the Enrollee appeals the Action and the resolution of the Appeal is not in the Enrollee’s favor, and the Enrollee requests an Administrative Law Hearing, ASES shall grant the Enrollee such hearing. The right to such Administrative Law Hearing, how to obtain it, and the rules concerning who may represent the Enrollee at such hearing shall be explained to the Enrollee and by the Contractor. |
| 14.6.4 | ASES shall permit the Enrollee to request an Administrative Law Hearing within thirty (30) Calendar Days of the Notice of Resolution of the Appeal. |
Page 195 of 301
| 14.6.5 | Before the Administrative Law Hearing, the Enrollee and the Enrollee’s Authorized Representative, if applicable, can ask to look at and copy the documents and records the Contractor will use at the Administrative Law Hearing or that the Enrollee may otherwise need to prepare his/her case for the hearing. The Contractor shall provide such documents and records at no charge to the Enrollee. |
| 14.6.6 | The Administrative Law Hearing resolution shall be: |
| 14.6.6.1 | For standard resolution: within ninety (90) Calendar Days of the date the Enrollee filed the appeal with the Contractor (excluding the days the Enrollee took to subsequently file for an Administrative Law Hearing). |
| 14.6.6.2 | For an expedited resolution: within three (3) Business Days from agency receipt of an Administrative Law Hearing request for a denial of a service. |
| 14.6.7 | The Contractor shall comply with all determinations rendered as a result of Administrative Law Hearings. Nothing in this Section 14.6 shall limit the remedies available to ASES or the Federal government relating to any non-compliance by the Contractor with an Administrative Law Hearing determination or by the Contractor’s refusal to provide disputed services. |
| 14.6.8 | The decision issued as a result of the Administrative Law Hearing is subject to review before the Court of Appeals of the Commonwealth. |
| 14.6.9 | The Contractor shall comply with all determinations rendered as a result of Administrative Law Hearings. Nothing in this Section 14.6 shall limit the remedies available to the Commonwealth or the Federal government relating to any non-compliance by the Contractor with an Administrative Law Hearing determination or by the Contractor’s refusal to provide disputed services. |
| 14.7 | Continuation of Benefits while the Appeal and Administrative Law Hearing are Pending |
| 14.7.1 | As used in this Section, “timely” filing means filing on or before the later of the following: |
| 14.7.1.1 | Within ten (10) Calendar Days of the Contractor mailing the Notice of Action; or |
| 14.7.1.2 | The intended effective date of the Contractor’s proposed Action. |
| 14.7.2 | The Contractor shall continue the Enrollee’s Benefits if the Enrollee or the Enrollee’s Authorized Representative files the Appeal in a timely manner; the Appeal involves the termination, suspension, or reduction of a previously authorized course of treatment; the services were ordered by an authorized Provider; the period covered by the original authorization has not expired; and the Enrollee requests extension of the Benefits. |
Page 196 of 301
| 14.7.3 | If, at the Enrollee’s request, the Contractor continues or reinstates the Enrollee’s Benefits while the Appeal or Administrative Law Hearing is pending, the Benefits shall be continued until one of the following occurs: |
| 14.7.3.1 | The Enrollee withdraws the Appeal or request for the Administrative Law Hearing. |
| 14.7.3.2 | Ten (10) Calendar Day pass after the Contractor mails the Notice of Action, unless the Enrollee, within the ten (10) Calendar Day timeframe, has requested an Administrative Law Hearing with continuation of Benefits until an Administrative Law Hearing decision is reached. |
| 14.7.3.3 | An administrative law judge issues an Administrative Law Hearing decision adverse to the Enrollee. |
| 14.7.3.4 | The time period or service limits of a previously authorized service has been met. |
| 14.7.4 | If the final resolution of Appeal or Administrative Law Hearing is adverse to the Enrollee, that is, upholds the Contractor’s Action, the Contractor may recover from the Enrollee the cost of the services furnished to the Enrollee while the Appeal / Administrative Law Hearing was pending, to the extent that they were furnished solely because of the requirements of this Section. |
| 14.7.5 | If the Contractor or ASES reverses a decision to deny, limit, or delay services that were not furnished while the Appeal / Administrative Law Hearing was pending, the Contractor shall authorize or provide the disputed services promptly and as expeditiously as the Enrollee’s health condition requires. |
| 14.7.6 | If the Contractor or ASES reverses a decision to deny authorization of services, and the Enrollee received the disputed services while the Appeal / Administrative Law Hearing was pending, the Contractor shall pay for those services. The Contractor shall submit evidence of compliance. |
| 14.8 | Reporting Requirements |
| 14.8.1 | The Contractor shall log and track all Complaints, Grievances, Notices of Action, Appeals, and Administrative Law Hearing requests (see Section 14.1.16 for details regarding Information collected). |
| 14.8.2 | ASES may publicly disclose summary Information regarding the nature of Complaints, Grievances, and Appeals and related dispositions or resolutions in consumer Information materials. |
Page 197 of 301
| 14.8.3 | The Contractor shall submit quarterly Grievance System reports to ASES using a format prescribed by ASES and incorporate the findings of these reports into its Quality Strategy. |
| 14.9 | Remedy for Contractor Non-Compliance with Advance Directive Requirements. |
In addition to the Complaint, Grievance, and Appeal rights described in this Article, an Enrollee may lodge with ASES a Complaint concerning the Contractor’s non-compliance with the Advance Directive requirements stated in Section 7.10 of this Contract.
ARTICLE 15 ADMINISTRATION AND MANAGEMENT
| 15.1 | General Provisions |
| 15.1.1 | The Contractor shall be responsible for the administration and management of all requirements of this Contract, and consistent with the Medicaid Managed Care regulations of 42 CFR Part 438. |
| 15.1.2 | All costs and expenses related to the administration and management of this Contract shall be the responsibility of the Contractor. |
| 15.2 | Place of Business and Hours of Operation |
| 15.2.1 | Given that Enrollment occurs chiefly on site in the Contractor’s administrative offices, the Contractor shall ensure that its administrative offices are physically accessible to all Enrollees and fully equipped to perform all functions related to carrying out this Contract. |
| 15.2.2 | The Contractor shall maintain administrative offices in each Service Region. |
| 15.2.3 | The Contractor shall accommodate any request by ASES to visit the Contractor’s administrative offices to ensure that the offices are compliant with the Americans with Disabilities Act’s (“ADA”) requirements for public buildings, and with all other applicable Federal and Puerto Rico rules and regulations. |
| 15.2.4 | The Contractor must maintain one (1) central administrative office and an additional administrative office in each Service Region covered under this Contract. |
| 15.2.5 | The Contractor’s office shall be centrally located and in a location accessible by foot and by vehicle traffic. The Contractor may establish more than one (1) administrative office within each of its Service Regions, but must designate one (1) of the offices as the central administrative office. |
Page 198 of 301
| 15.2.6 | All of the Contractor’s written communications to Enrollees must contain the address of the location identified as the legal, duly-licensed, central administrative office. This administrative office must be open at least between the hours of 9:00 a.m. and 5:00 p.m. (Atlantic Time), Monday through Friday; In addition, pursuant to the Contractor’s Enrollment Outreach plan (see Section 6.12), the Contractor’s administrative office must have extended open hours (until 7:00 p.m. (Atlantic Time) at least one (1) Business Day per Week; and must be open (to the extent necessary to permit Enrollment activities) one Saturday per month, from 9:00 a.m. to 5:00 p.m. (Atlantic Time)). |
| 15.2.7 | The Contractor shall ensure that the office(s) are adequately staffed, throughout the Contract Term, to ensure that Potential Enrollees may visit the office to enroll at any time during Contractor’s hours of operation. This provision will ensure that Enrollees and Providers receive prompt and accurate responses to inquiries. |
| 15.2.8 | The Contractor shall provide access to Information to Enrollees through GHP Service Line, during the hours provided in Section 6.8.3 of this Contract. |
| 15.2.9 | The Contractor shall provide access twenty-four (24) hours a day, seven (7) days per Week to its website. |
| 15.3 | Training and Staffing |
| 15.3.1 | The Contractor shall conduct ongoing training for all of its staff, in all departments, to ensure appropriate functioning in all areas and to ensure that staff: |
| 15.3.1.1 | Understand the GHP program and the Medicaid Managed Care requirements; |
| 15.3.1.2 | Are aware of all programmatic changes; and |
| 15.3.1.3 | Are trained in the Contractor’s Cultural Competency plan. |
| 15.3.2 | The Contractor shall submit a staff training plan and a current organizational chart to ASES for review and prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 15.4 | Data Certification |
| 15.4.1 | The Contractor shall certify all Data pursuant to 42 CFR 438.606. The Data that must be certified include, but are not limited to, Enrollment Information, Encounter Data, and other Information required by ASES and contained in Contracts, the Contractor’s Proposal, and related documents. The Data must be certified by one of the following: the Contractor’s Chief Executive Officer (“CEO”), the Contractor’s Chief Financial Officer (“CFO”), or an individual who has delegated authority to sign for, and who reports directly to the Contractor’s CEO or CFO. The certification must attest, based on best knowledge, Information, and belief, as follows: |
Page 199 of 301
| 15.4.1.1 | To the accuracy, completeness and truthfulness of the Data; and |
| 15.4.1.2 | To the accuracy, completeness, and truthfulness of the documents specified by ASES. |
| 15.4.2 | The Contractor shall submit the certification concurrently with the certified Data. |
| 15.5 | Implementation Plan and Submission of Initial Deliverables |
| 15.5.1 | The Contractor shall develop an Implementation Plan that verifies that the Contractor will submit the Deliverables listed in the chart in Attachment 12 to this Contract, and that details any additional procedures and activities that will be accomplished during the period between the Effective Date of this Contract and April 1, 2015, which is the Implementation Date of this Contract. The Implementation Plan shall include coordination and cooperation with ASES and its representatives during all phases. |
| 15.5.2 | The Contractor shall submit its implementation plan to ASES for ASES’s review and written approval according to the timeframe specified in Attachment 12 to this Contract. Implementation of the Contract shall not commence prior to ASES written approval. |
| 15.5.3 | The Contractor will not receive any additional payment to cover start up or implementation costs. |
ARTICLE 16 PROVIDER PAYMENT MANAGEMENT
| 16.1 | General Provisions |
| 16.1.1 | The Contractor shall administer an effective, accurate and efficient Provider payment management function that (i) under this Contract’s risk arrangement adjudicates and settles Provider Claims for Covered Services that are filed within the timeframes specified by this Article 16 and in compliance with all applicable Puerto Rico and Federal laws, rules, and regulations; (ii) processes PMPM Payments to applicable Providers within the timeframes specified by this Article; and (iii) performs Claims payment administrative functions for all Providers as specified by this Article 16. |
| 16.1.2 | The Contractor shall maintain a Claims management system that can accurately identify the date of receipt (the date the Contractor receives the Claim as indicated by the date-stamp), real-time-accurate history of actions taken on each Provider Claim (i.e. paid, denied, suspended, appealed, etc.), and the date of payment (the date of the check or other form of payment). |
Page 200 of 301
| 16.1.3 | To the extent feasible, the Contractor shall implement an Automated Clearinghouse (“ACH”) mechanism that allows Providers to request and receive Electronic Funds Transfer (“EFT”) of Claims payments. The Contractor shall encourage its Providers, as an alternative to the filing of paper-based Claims, to submit and receive Claims Information through Electronic Data Interchange (“EDI”), i.e., electronic Claims. Electronic Claims must be processed in adherence to Information exchange and Data management requirements specified in Article 17. As part of this electronic Claims management (“ECM”) function, the Contractor shall also provide on-line and phone-based capabilities to obtain Claims processing status Information. |
| 16.1.4 | If the Contractor does not receive Claims through an EDI system, the Contractor shall either provide a central address to which Providers must submit Claims; or provide to each Network Provider a complete list, including names, addresses, electronic mail and phone number, of entities to which the Providers must submit Claims. |
| 16.1.5 | The Contractor shall notify Network Providers in writing of any changes in the policies and procedures, subject to prior written approval of ASES, for filing Claims at least thirty (30) Calendar Days before the effective date of the change. If the Contractor is unable to provide thirty (30) Calendar Days of notice, it must give Providers a thirty (30) Calendar Day extension on their Claims filing deadline to ensure Claims are routed to the correct processing center. |
Page 201 of 301
| 16.2 | To be processed, all Claims submitted for payment shall comply with the Clean Claim standards as established by Federal regulation (42 CFR 447.46), and with the standards described in Section 16.10.2 of this Contract. |
| 16.3 | The Contractor shall generate explanations of benefits and remittance advices in accordance with ASES standards for formatting, content, and timeliness. |
| 16.4 | The Contractor shall not pay any Claim submitted by a Provider who is excluded or suspended from the Medicare, Medicaid or CHIP programs for Fraud, Waste, or Abuse or otherwise included on the Department of Health and Human Services Office of the Inspector General exclusions list, or employs someone on this list. The Contractor shall not pay any Claim submitted by a Provider that is on Payment Hold. |
| 16.5 | The Contractor is prohibited from paying for an item or service with respect to any amount expended for which funds may not be used under the Assisted Suicide Funding Restriction Act of 1997. |
| 16.6 | The Contractor is prohibited from making payment on any amount expended for roads, bridges, stadiums, or other item or service not covered under the Medicaid State Plan. |
| 16.7 | Payment Schedule |
| 16.7.1 | At a minimum, the Contractor shall run one (1) Provider payment cycle per Week, on the same day each Week, as determined by the Contractor. The Contractor shall develop a payment schedule to be submitted to ASES for review and its prior written approval according to the timeframe specified in Attachment 12 to this Contract. |
| 16.7.2 | Other than for cause explicitly stated in the Provider Contract, payment to Providers made in the form of a Capitation payment shall be issued not later than the fifteenth (15th) Calendar Day of the month. Any Provider Capitation payment retained by the Contractor past the 15th Calendar Day of each month shall accrue interest at the prevailing highest legal interest rate for personal loans as such rate is determined by the Board of the Office of the Commissioner of Financial Institutions, and interest shall be paid along with the Capitation payment to the Provider for that month. The Contractor shall make such payment regardless of receiving the PMPM Payment under Section 22.1.1 of the Contract. |
| 16.8 | Required Claims Processing Reports |
| 16.8.1 | The Contractor shall submit to ASES a monthly report not later than the fifth (5th) Calendar Day after the last day of the month listing all paid, pending, and denied Claims during that month. The report shall be made available in an electronic format and shall detail all paid, pending, and denied Claims for all Providers. |
Page 202 of 301
| 16.8.2 | The report shall list, by Provider, Claims paid from the preceding month, and those that are pending payment and the reason for the payment delay or the reason for the Contractor’s decision to deny the Claim. |
| 16.8.3 | In the event that Providers associated with a PMG consent to the disbursement of payment directly to the PMG, the Contractor shall so specify in its report. |
| 16.9 | Submission of Encounter Data |
| 16.9.1 | Providers shall furnish Encounter Data to the Contractor per Section 17.3.3 of the Contract on a monthly basis. The Data shall be submitted regardless of the payment arrangement, capitated or otherwise, agreed upon between the Contractor and the Provider. |
| 16.10 | Relationship With Pharmacy Benefit Manager (PBM) |
| 16.10.1 | The Contractor shall work with the PBM engaged by ASES to facilitate the processing of pharmacy services Claims submitted by the PBM, as provided in Section 7.5.12.11. |
| 16.10.2 | To facilitate Claims processing, the Contractor shall send to the PBM, on a Daily Basis, the Enrollee Data described in Section 5.3.8. |
| 16.11 | Timely Payment of Claims |
| 16.11.1 | The Contractor shall comply with the timely processing of Claims standards contained in Section 1902(a)(37) of the Social Security Act and in implementing Federal Medicaid regulations at 42 CFR 447.46. |
| 16.11.2 | Provider Contracts shall include the following provisions for timely payment of Clean Claims. |
| 16.11.2.1 | A Clean Claim under 42 CFR 447.46(b), as defined in 42 CFR 447.45(b), is a Claim received by the Contractor for adjudication, which can be processed without obtaining additional Information from the Provider of the service or from a Third Party. It includes a Claim with errors originating in the Contractor’s Claims system. It does not include a Claim from a Provider who is under investigation for Fraud, Waste, or Abuse, or a Claim under review for Medical Necessity. |
| 16.11.2.2 | Provider Contracts shall provide that ninety-five percent (95%) of all Clean Claims must be paid by the Contractor not later than thirty (30) Calendar Days from the date of receipt of the Claim (including Claims billed by paper and electronically), and one hundred percent (100%) of all Clean Claims must be paid by the Contractor not later than fifty (50) Calendar Days from the date of receipt of the Claim. |
Page 203 of 301
| 16.11.2.3 | Any Clean Claims not paid within thirty (30) Calendar Days shall bear interest in favor of the Provider on the total unpaid amount of such Claim, according to the prevailing highest legal interest rate fixed by the Puerto Rico Commissioner of Financial Institutions. Such interest shall be considered payable on the day following the terms of this Section 16.10, and interest shall be paid together with the claim. |
| 16.11.3 | An Unclean Claim is any Claim that falls outside the definition of Clean Claim in Section 16.10.2.1. The Contractor shall include the following provisions in its Provider Contracts for timely resolution of Unclean Claims. |
| 16.11.3.1 | Ninety percent (90%) of Unclean Claims must be resolved and processed with payment by the Contractor, if applicable, not later than ninety (90) Calendar Days from the date of initial receipt of the Claim. This includes Claims billed on paper or electronically. |
| 16.11.3.2 | Of the remaining ten percent (10%) of total Unclean Claims that may remain outstanding after ninety (90) Calendar Days, |
| 16.11.3.2.1 | Nine percent (9%) of the Unclean Claims must be resolved and processed with payment by the Contractor, if applicable, not later than six (6) calendar months from the date of initial receipt (including Claims billed on paper and those billed electronically); and |
| 16.11.3.2.2 | One percent (1%) of the Unclean Claims must be resolved and processed with payment by the Contractor, if applicable, not later than one year (twelve (12) months) from the date of initial receipt of the Claim (including Claims billed on paper and those billed electronically). |
| 16.11.4 | The Contractor shall not establish any administrative procedures, such as administrative audits, authorization number, or other formalities under the control of the Contractor, which could prevent the Provider from submitting a Clean Claim. |
| 16.11.5 | The foregoing timely payment standards are more stringent than those required in the Federal regulations, at 42 CFR 447.46. The Contractor shall include the foregoing standards in each Provider Contract and, per 42 CFR 447.46(c). |
| 16.11.6 | The Contractor shall deliver to Providers, within fifteen (15) Calendar Days of award of the Provider Contract (along with the Provider Guidelines described in Section 10.2.1), Claims coding and processing guidelines for the applicable Provider type, and the definition of a Clean Claim, as requested in this Article 16, to be applied. |
Page 204 of 301
| 16.11.7 | The Contractor shall give Providers ninety (90) Calendar Days’ notice in advance of the effective date of any change in Claims coding and processing deadlines. |
| 16.12 | Contractor Denial of Claims and Resolution of Contractual and Claims Disputes |
| 16.12.1 | Not later than the fifth (5th) Business Day after the receipt of a Provider Claim that the Contractor has deemed not to meet the Clean Claim requirements, the Contractor shall suspend the Claim and request in writing (notification via e-mail, the Contractor’s website, or an interim remittance advice satisfies this requirement) all outstanding Information such that the Claim can be deemed clean. Upon receipt of all the requested Information from the Provider, the Contractor shall complete processing of the Claim in accordance with the standards outlined in Section 16.10. |
| 16.12.2 | Claims suspended for additional Information must be closed (paid or denied) such that compliance with the timely payment rules outlined in Section 16.10 is achieved. |
| 16.12.3 | The Contractor must process, and finalize, all appealed Claims to a paid or denied status within thirty (30) Calendar Days of receipt of the appealed Claim; for Claims for which the Contractor has requested further information, per Section 16.11.1, the Contractor shall pay or deny the Claim within thirty (30) Calendar Days of receipt of the requested Information. |
| 16.12.4 | The Contractor shall send Providers written notice (notification via e-mail, surface mail, the Contractor’s website, or a remittance advice satisfies this requirement) for each Claim that is denied, including the reason(s) for the denial, the date the Contractor received the Claim, and a reiteration of the outstanding Information required from the Provider to adjudicate the Claim. |
| 16.12.5 | In situations in which the Contractor denies a Provider’s Claim for services, and the Provider disputes the denial, as provided in Section 16.11.6, the Contractor shall not withhold payment pending final resolution of the dispute, but instead shall pay the Claim within thirty (30) Calendar Days of the Contractor’s receipt of the Provider’s written complaint (see Section 16.11.6). The Contractor shall seek recoupment of the paid Claim only in the event that the dispute is resolved, at the level of the dispute resolution described in Section 16.11.6, in the Contractor’s favor. |
| 16.12.6 | Provider Dispute Resolution System |
| 16.12.6.1 | The Contractor shall establish and use a procedure to resolve billing, payment, and other administrative disputes between Providers and the Contractor arising under Provider Contracts including a Provider Complaint resolution process implemented by the Contractor to address, among others, lost or incomplete Claims forms or electronic submissions; Contractor requests for additional explanation as to services or treatment rendered by a Provider; and inappropriate or unapproved Referrals issued by Providers. This dispute resolution system shall exclude Grievances filed by Providers on behalf of Enrollees pursuant to Section 14.3 of this Contract. |
Page 205 of 301
| 16.12.6.2 | For any dispute between the Provider and Contractor arising under the Provider Contract, the Contractor shall implement an internal dispute resolution system, which shall include the opportunity for an aggrieved Provider to submit a timely written complaint to the Contractor. The Contractor shall issue a written decision on the Provider’s complaint within fifteen (15) Calendar Days of receipt of the Provider’s written complaint. A Contractor’s written decision that is in any way adverse to the Provider shall include an explanation of the grounds for the decision and a notice of the Provider’s right to and procedures for an Administrative Law Hearing within ASES. |
| 16.12.6.3 | If the Provider is not satisfied with the decision on its complaint within the Contractor’s dispute resolution system, the Provider may pursue an Administrative Law Hearing. The parties to the Administrative Law Hearing shall be the Contractor and the Provider. ASES shall grant a Provider request for an Administrative Law Hearing, provided that the Provider submits a written appeal, accompanied by supporting documentation, not more than thirty (30) Calendar Days following the Provider’s receipt of the Contractor’s written decision. |
| 16.12.6.4 | Judicial Review. A decision issued as a result of the Administrative Law Hearing provided for in Section 16.11.6.3 shall be subject to review before the Court of Appeals of the Commonwealth. |
| 16.13 | Contractor Recovery from Providers |
| 16.13.1 | When the Contractor determines after the fact that it has paid a Claim incorrectly, or when the Contractor, per Section 16.11.5, is entitled to seek recoupment, the Contractor may request applicable reimbursement from the Provider through written notice, stating the basis for the request. The notice shall list the Claims and the amounts to be recovered. |
| 16.13.2 | The Provider will have a period of ninety (90) Calendar Days to make the requested payment, to agree to Contractor retention of said payment, or to dispute the recovery Action following the process described in Section 16.11.6. |
Page 206 of 301
| 16.14 | ASES Review of Contractor, Subcontractor, and Provider Use of Puerto Rico and Federal Funds |
| 16.14.1 | The Contractor shall cooperate fully and diligently with ASES and/or its auditors in their review of the use of Puerto Rico and Federal funds provided to the Contractor under the GHP Program. The Contractor, its Subcontractors, and Network Providers shall, upon request, make available to ASES and/or its auditors any and all administrative, financial, and Medical Records relating to the administration of and the delivery of items or services for which Puerto Rico and Federal monies are expended. In addition, the Contractor and its Subcontractors including Network Providers shall provide ASES and/or its auditors with access during normal business hours to its respective place of business and records. |
| 16.15 | ASES Recovery From Contractor |
| 16.15.1 | ASES and the Contractor shall diligently work in good faith together to resolve any audit findings identified through audits by ASES. All audit findings shall be resolved or a Corrective Action Plan shall be implemented within ninety (90) Calendar Days of issuance of a final audit report. Any Overpayment remittance due to ASES from the Contractor will be offset from future payments to the Contractor. |
ARTICLE 17 INFORMATION MANAGEMENT AND SYSTEMS
| 17.1 | General Provisions |
| 17.1.1 | The Contractor shall have Information management processes and Information Systems (hereafter referred to as Systems) that enable it to meet GHP requirements, ASES and Federal reporting requirements, all other Contract requirements, and any other applicable Puerto Rico and Federal laws, rules and regulations including but not limited to the standards and operating rules in Section 1104 of the PPACA and associated regulations, the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), Health Information Technology for Economic and Clinical Health Act (HITECH) and associated regulations and 42 CFR 438.242. |
| 17.1.2 | The Contractor shall file a statement of certification with the U.S. Department of Health and Human Services (HHS) no later than the April 1, 2015, the Implementation Date of the Contract, certifying that the Contractor’s Data and Systems are in compliance with the standards and operating rules for EFT, eligibility, Claim status and health care payment/remittance advice transactions, in accordance with Section 1104 of the PPACA and associated regulations. |
Page 207 of 301
| 17.1.3 | The Contractor’s Systems shall possess capacity sufficient to handle the workload projected for the start of the program and will be scalable and flexible so they can be adapted as needed, within negotiated timeframes, in response to program or Enrollment changes. |
| 17.1.4 | The Contractor’s Systems shall have the capability of adapting to any future changes necessary as a result of modifications to the service delivery system and its requirements, including Data collection, records and reporting based upon unique Enrollee and Provider identifiers to track services and expenditures across funding streams. The Systems shall be scalable and flexible so they can be adapted as needed, within negotiated timeframes, in response to changes in Contract requirements, increases in Enrollment estimates, etc. The System architecture shall facilitate rapid application of the more common changes that can occur in the Contractor’s operation, including but not limited to: |
| 17.1.4.1 | Changes in pricing methodology; |
| 17.1.4.2 | Rate changes; |
| 17.1.4.3 | Eligibility criteria changes; |
| 17.1.4.4 | Changes in Utilization Management criteria; |
| 17.1.4.5 | Additions and deletions of Provider types; and |
| 17.1.4.6 | Additions and deletions of procedure, diagnosis and other service codes. |
| 17.1.4.7 | Changes in the Enrollment methodology. |
| 17.1.5 | The Contractor shall provide secure, online access to select system functionality to at least three (3) ASES personnel to facilitate resolution of Enrollee inquiries and to research Enrollee-related issues as needed. |
| 17.1.6 | The Contractor shall participate in systems work groups organized by ASES. The Systems work groups will meet on a designated schedule as agreed to by ASES and the GHP MCOs. |
| 17.1.7 | The Contractor shall provide a continuously available electronic mail communication link (E-mail system) with ASES. This system shall be: |
| 17.1.7.1 | Available from the workstations of the designated Contractor contacts; and |
| 17.1.7.2 | Capable of attaching and sending documents created using software products other than Contractor systems, including the Commonwealth’s currently installed version of Microsoft Office and any subsequent upgrades as adopted. |
Page 208 of 301
| 17.2 | Global System Architecture and Design Requirements |
| 17.2.1 | The Contractor shall comply with Federal and Puerto Rico policies, standards and regulations in the design, development and/or modification of the Systems it will employ to meet the aforementioned requirements and in the management of information contained in those Systems. Additionally, the Contractor shall adhere to ASES and Puerto Rico-specific system and Data architecture standards and/or guidelines. |
| 17.2.2 | The Contractor’s Systems shall meet Federal and industry standards of architecture, including but not limited to the following requirements: |
| 17.2.2.1 | Conform to HIPAA standards for Data and document management; |
| 17.2.2.2 | Contain controls to maintain information integrity. These controls shall be in place at all appropriate points of processing. The controls shall be tested in periodic and spot audits following a methodology to be developed jointly by and mutually agreed upon by the Contractor and ASES; and |
| 17.2.2.3 | Partner with ASES in the development of transaction/event code set, Data exchange and reporting standards not specific to HIPAA or other Federal efforts and will conform to such standards as stipulated in the plan to implement the standards. |
| 17.2.3 | Where web services are used in the engineering of applications, the Contractor’s Systems shall conform to World Wide Web Consortium (W3C) standards such as XML, UDDI, WSDL and SOAP so as to facilitate integration of these Systems with ASES and other Commonwealth systems that adhere to a service-oriented architecture. |
| 17.2.4 | Audit trails shall be incorporated into all Systems to allow information on source Data files and documents to be traced through the processing stages to the point where the information is finally recorded. The audit trails shall: |
| 17.2.4.1 | Contain a unique log-on or terminal ID, the date, and time of any create/modify/delete action and, if applicable, the ID of the system job that effected the action; |
| 17.2.4.2 | Have the date and identification “stamp” displayed on any on-line inquiry; |
| 17.2.4.3 | Have the ability to trace Data from the final place of recording back to its source Data file and/or document shall also exist; |
| 17.2.4.4 | Be supported by listings, transaction reports, update reports, transaction logs, or error logs; |
Page 209 of 301
| 17.2.4.5 | Facilitate auditing of individual Claim records as well as batch audits; and |
| 17.2.4.6 | Be maintained for seven (7) years in either live and/or archival systems. The duration of the retention period may be extended at the discretion of and as indicated to the Contractor by ASES as needed for ongoing audits or other purposes. |
| 17.2.5 | The Contractor shall house indexed images of documents used by Enrollees and Providers to transact with the Contractor in the appropriate database(s) and document management systems so as to maintain the logical relationships between certain documents and certain Data. The Contractor shall follow all applicable requirements for the management of Data in the management of documents. |
| 17.2.6 | The Contractor shall institute processes to insure the validity and completeness of the Data it submits to ASES. At its discretion, ASES will conduct general Data validity and completeness audits using industry-accepted statistical sampling methods. Data elements that will be audited include but are not limited to: Enrollee ID, date of service, Provider ID, category and sub category (if applicable) of service, diagnosis codes, procedure codes, revenue codes, date of Claim processing, and date of Claim payment. |
| 17.2.7 | Where a System is herein required to, or otherwise supports, the applicable batch or on-line transaction type, the system shall comply with HIPAA-standard transaction code sets. |
| 17.2.8 | The Contractor shall assure that all Contractor staff is trained in all HIPAA requirements, as applicable. |
| 17.2.9 | The layout and other applicable characteristics of the pages of Contractor websites shall be compliant with Federal “Section 508 standards” and Web Content Accessibility Guidelines developed and published by the Web Accessibility Initiative. |
| 17.3 | System and Data Integration Requirements |
| 17.3.1 | The Contractor’s applications shall be able to interface with ASES’s systems for purposes of Data exchange and will conform to standards and specifications set by ASES. These standards and specifications are subject to change. Current standards and specifications are detailed in Attachment 9. |
| 17.3.2 | The Contractor’s System(s) shall be able to transmit and receive transaction Data to and from ASES’s systems as required for the appropriate processing of Claims. |
Page 210 of 301
| 17.3.2.1 | The Contractor will be required to perform any necessary changes to update interfaces to ASES’s systems, including those required by the expected implementation of a new Medicaid Management Information System (MMIS) as well as new Eligibility and Enrollment processes. This interface changes may require changes in the Contractors core systems. |
| 17.3.3 | Each month the Contractor shall generate Encounter Data files from its Claims management system(s) and/or other sources. The files will contain settled Claims and Claim adjustments and Encounter Data from Providers for the most recent month for which all such transactions were completed. The Contractor shall provide these files electronically to ASES and/or its Agent in adherence to the procedure, content standards and format indicated in Attachment 9. The Contractor shall make changes or corrections to any systems, processes or Data transmission formats as needed to comply with Encounter Data quality standards as originally defined or subsequently amended. |
| 17.3.4 | The Contractor’s System(s) shall be capable of generating files in the prescribed formats for upload into ASES Systems used specifically for program integrity and compliance purposes. |
| 17.3.5 | The Contractor’s System(s) shall possess mailing address standardization functionality in accordance with US Postal Service conventions. |
| 17.4 | System Access Management and Information Accessibility Requirements |
| 17.4.1 | The Contractor’s System shall employ an access management function that restricts access to varying hierarchical levels of system functionality and Information. The access management function shall: |
| 17.4.1.1 | Restrict access to information on a "need-to-know" basis, e.g. users permitted inquiry privileges only will not be permitted to modify information; |
| 17.4.1.2 | Restrict access to specific System functions and Information based on an individual user profile, including inquiry only capabilities; global access to all functions will be restricted to specified staff jointly agreed to by ASES and the Contractor; and |
| 17.4.1.3 | Restrict attempts to access system functions to three (3), with a system function that automatically prevents further access attempts and records these occurrences. |
| 17.4.2 | The Contractor shall make System information available to duly Authorized Representatives of ASES and other Puerto Rico and Federal agencies to evaluate, through inspections or other means, the quality, appropriateness and timeliness of services performed. |
Page 211 of 301
| 17.4.3 | The Contractor shall have procedures to provide for prompt transfer of System Information upon request to other Network or Out-of-Network Providers for the medical management of the Enrollee in adherence to HIPAA and other applicable requirements. |
| 17.4.4 | All Information, whether Data or documents, and reports that contain or make references to said Information, involving or arising out of this Contract, are owned by ASES. The Contractor is expressly prohibited from sharing or publishing ASES Information and reports without the prior written consent of ASES. In the event of a dispute regarding the sharing or publishing of Information and reports, ASES’s decision on this matter shall be final and not subject to appeal. |
| 17.5 | Systems Availability and Performance Requirements |
| 17.5.1 | The Contractor shall ensure that critical systems, including but not limited to the Enrollee and Provider portal and/or phone-based functions and information, such as confirmation of Contractor Enrollment (“CCE”) and electronic Claims management (ECM), Enrollee services and Provider services, are available to the applicable System users twenty-four (24) hours a day, seven (7) Calendar Days a Week, except during periods of scheduled System Unavailability agreed upon by ASES and the Contractor. Unavailability caused by events outside of a Contractor’s Span of Control is outside of the scope of this requirement. |
| 17.5.2 | The Contractor shall ensure that at a minimum all non-critical system functions and information are available to the applicable system users between the hours of 7:00 a.m. and 7:00 p.m. Monday through Friday (Atlantic Time). |
| 17.5.3 | The Contractor shall develop an automated method of monitoring critical systems on at least a thirty (30) minute basis twenty-four (24) hours a day, seven (7) days per Week. |
| 17.5.4 | Upon discovery of any problem within its Span of Control that may jeopardize System availability and performance as defined in this Section of the Contract, the Contractor shall notify the applicable ASES staff in person, via phone, and/or electronic mail. The Contractor shall deliver notification as soon as possible but no later than 7:00 pm (Atlantic Time) if the problem occurs during the Business Day and no later than 9:00 am (Atlantic Time) the following Business Day if the problem occurs after 7:00 pm (Atlantic Time). |
| 17.5.5 | Where the operational problem results in delays in report distribution or problems in on-line access during the Business Day, the Contractor shall notify the applicable ASES staff within fifteen (15) minutes of discovery of the problem, in order for the applicable work activities to be rescheduled or be handled based on System Unavailability protocols. |
Page 212 of 301
| 17.5.6 | The Contractor shall provide to appropriate ASES staff information on System Unavailability events, as well as status updates on problem resolution. These up-dates shall be provided on an hourly basis and made available via electronic mail, telephone and, if applicable, the Contractor’s website. |
| 17.5.7 | The following rules govern unscheduled System Unavailability. |
| 17.5.7.1 | CCE Functions |
| 17.5.7.1.1 | Unscheduled System Unavailability of CCE functions caused by the failure of systems and telecommunications technologies within the Contractor’s Span of Control will be resolved, and the restoration of services implemented, within thirty (30) minutes of the official declaration of System Unavailability. |
| 17.5.7.1.2 | Throughout the Contract Term, the Contractor shall have in place a method to validate eligibility manually twenty-four (24) hours per day, seven (7) days a Week as a contingency to any unscheduled Systems Unavailability for CCE functions. |
| 17.5.7.2 | ECM Functions. Unscheduled System Unavailability of ECM functions caused by the failure of systems and technologies within the Contractor’s Span of Control will be resolved, and the restoration of services implemented, within sixty (60) minutes of the official declaration of System Unavailability, if unavailability occurs during normal business hours; or within sixty (60) minutes of the start of the next Business Day, if unavailability occurs outside business hours. |
| 17.5.7.3 | All Other Contractor System Functions. Unscheduled System Unavailability of all other Contractor System functions caused by systems and telecommunications technologies within the Contractor’s Span of Control shall be resolved, and the restoration of services implemented: |
| 17.5.7.3.1 | Within four (4) hours of the official declaration of Unscheduled System Unavailability, when unavailability occurs during business hours, and |
| 17.5.7.3.2 | Within two (2) hours of the start of the next Business Day, when unavailability occurs during non-business hours. |
| 17.5.8 | Cumulative System Unavailability caused by systems and telecommunications technologies within the Contractor’s Span of Control shall not exceed one (1) hour during any continuous five (5) Calendar Day period for functions that affect GHP Enrollees and services. For functions that do not affect GHP Enrollees, cumulative System Unavailability caused by systems and telecommunications technologies within the Contractor’s Span of Control shall not exceed four (4) hours during any continuous five (5) Business Day periods. |
Page 213 of 301
| 17.5.9 | The Contractor shall not be responsible for the availability and performance of systems and telecommunications technologies outside of the Contractor’s Span of Control. |
| 17.5.10 | For any System outage that is not corrected within the required time limits, the Contractor shall provide full written documentation that includes a Corrective Action Plan, describing how the problem will be prevented from occurring again, within five (5) Business Days of the problem’s occurrence. |
| 17.5.11 | Regardless of the architecture of its Systems, the Contractor shall develop and be continually ready to invoke a Business Continuity and Disaster Recovery (“BC-DR”) plan that at a minimum addresses the following scenarios: (i) the central computer installation and resident software are destroyed or damaged; (ii) System interruption or failure resulting from network, operating hardware, software, or operational errors that compromises the integrity of transactions that are active in a live system at the time of the outage; (iii) System interruption or failure resulting from network, operating hardware, software or operational errors that compromises the integrity of Data maintained in a live or archival system; and (iv) System interruption or failure resulting from network, operating hardware, software or operational errors that does not compromise the integrity of transactions or Data maintained in a live or archival system but does prevent access to the System, i.e. causes unscheduled System Unavailability. This BC-DR plan must be prior approved by ASES. |
| 17.5.12 | The Contractor shall on a quarterly basis test its BC-DR plan through simulated disasters and lower level failures in order to demonstrate to ASES that it can restore System functions per the standards outlined elsewhere in this Section 17.5 of the Contract. The results of these tests shall be reported to ASES within thirty (30) Calendar Days of completion of said tests. |
| 17.5.13 | In the event that the Contractor fails to demonstrate in the tests of its BC-DR plan that it can restore system functions per the standards outlined in this Contract, the Contractor shall be required to submit to ASES a Corrective Action Plan that describes how the failure will be resolved. The Corrective Action Plan will be delivered within five (5) Business Days of the conclusion of the test. |
Page 214 of 301
| 17.5.14 | The Contractor shall submit a monthly Systems Availability and Performance Report to ASES as further described in Section18.2.8 of this Contract. |
| 17.6 | System Testing and Change Management Requirements |
| 17.6.1 | The Contractor shall absorb the cost of routine maintenance, inclusive of defect correction, System changes required to effect changes in Puerto Rico and Federal statute and regulations, and production control activities, of all Systems within its Span of Control. |
| 17.6.2 | The Contractor shall respond to ASES reports of System problems not resulting in System Unavailability according to the following timeframes: |
| 17.6.2.1 | Within five (5) Calendar Days of receipt, the Contractor shall respond in writing to notices of System problems. |
| 17.6.2.2 | Within fifteen (15) Calendar Days, the correction will be made or a requirements analysis and specifications document will be due. |
| 17.6.3 | The Contractor shall correct the deficiency by an effective date to be determined by ASES. |
| 17.6.4 | The Contractor’s Systems will have a system-inherent mechanism for recording any change to a software module or subsystem. |
| 17.6.5 | The Contractor shall put in place procedures and measures for safeguarding ASES from unauthorized modifications to the Contractor’s Systems. |
| 17.6.6 | Unless otherwise agreed to in advance by ASES, scheduled System Unavailability to perform System maintenance, repair and/or upgrade activities to Contractor’s CCE systems shall take place between 11 p.m. on a Saturday and 6 a.m. on the following Sunday (Atlantic Time). |
| 17.6.7 | The Contractor shall work with ASES pertaining to any testing initiative as required by ASES. |
| 17.6.8 | The Contractor shall provide sufficient System access to allow verification of System functionality, availability and performance by ASES during the times required by ASES prior to April 1, 2015 which is the Implementation Date of the Contract, and as subsequently required during the Contract Term. |
| 17.7 | System Security and Information Confidentiality and Privacy Requirements |
| 17.7.1 | The Contractor shall provide for the physical safeguarding of its Data processing facilities and the Systems and Information housed therein. The Contractor shall provide ASES with access to Data facilities upon ASES’s request. The physical security provisions shall be in effect for the life of this Contract. |
Page 215 of 301
| 17.7.2 | The Contractor shall restrict perimeter access to equipment sites, processing areas, and storage areas through a card key or other comparable system, as well as provide accountability control to record access attempts, including attempts of unauthorized access. |
| 17.7.3 | The Contractor shall include physical security features designed to safeguard processor site(s) through required provision of fire retardant capabilities, as well as smoke and electrical alarms, monitored by security personnel. |
| 17.7.4 | The Contractor shall ensure that the operation of all of its Systems is performed in accordance with Puerto Rico and Federal regulations and guidelines related to security and confidentiality of the protected information managed by the Contractor, and shall strictly comply with HIPAA Privacy and Security Rules, as amended, and with the Breach Notification Rules under the HITECH Act. |
| 17.7.5 | The Contractor will put in place procedures, measures and technical security to prohibit unauthorized access to the regions of the Data communications network inside of a Contractor’s Span of Control. |
| 17.7.6 | The Contractor shall ensure compliance with: |
| 17.7.6.1 | 42 CFR Part 431 Subpart F (confidentiality of information concerning applicants and enrollees of public medical assistance programs); |
| 17.7.6.2 | 42 CFR Part 2 (confidentiality of alcohol and drug abuse records); and |
| 17.7.6.3 | Special confidentiality provisions in Puerto Rico or Federal law related to people with HIV/AIDS and mental illness. |
| 17.7.7 | The Contractor shall provide its Enrollees with a privacy notice as required by HIPAA. The Contractor shall provide ASES with a copy of its Privacy Notice for its filing. |
| 17.8 | Information Management Process and Information Systems Documentation Requirements |
| 17.8.1 | The Contractor shall ensure that written System Process and Procedure Manuals document and describe all manual and automated system procedures for its information management processes and Information Systems. These manuals shall be provided to ASES Immediately upon request. |
Page 216 of 301
| 17.8.2 | The System User Manuals shall contain information about, and instructions for, using applicable System functions and accessing applicable system Data. |
| 17.8.3 | When a System change that would alter the conditions and services agreed upon in this Contract is subject to ASES sign off, the Contractor shall draft revisions to the appropriate manuals prior to ASES sign off of the change. |
| 17.8.4 | Updates to the electronic version of these manuals shall occur in real time; updates to the printed version of these manuals shall occur within ten (10) Business Days of the update taking effect. |
| 17.8.5 | ASES reserves the right to audit the Contractor’s policies and procedures manuals and protocols compliance related to its Information Systems. |
| 17.9 | Reporting Functionality Requirements |
| 17.9.1 | The Contractor’s Systems shall have the capability of producing a wide variety of reports that support program management, policymaking, quality improvement, program evaluation, analysis of fund sources and uses, funding decisions and assessment of compliance with Federal and Puerto Rico requirements. |
| 17.9.2 | The Contractor shall support a mechanism for obtaining service and expenditure reports by funding source, Provider, Provider type or other characteristic; and Enrollee, Enrollee group/category or other characteristic. |
| 17.9.3 | The Contractor shall extend access to this mechanism to select ASES personnel in a secure manner to access Data, including program and fiscal information regarding Enrollees served, services rendered, etc. and the ability for said personnel to develop and/or retrieve reports. This requirement could be met by the provision of access to a decision support system/Data warehouse. The Contractor shall provide training in and documentation on the use of this mechanism. |
| 17.9.4 | Within five (5) Calendar Days upon ASES’s request, the Contractor will deliver a copy of the then current ASES’s System information to ASES in a mutually acceptable form and format. |
| 17.10 | Disaster Recovery, Disaster Declaration, Data Content Delivery to ASES |
| 17.10.1 | Contractor shall maintain a disaster recovery and business recovery plan in effect throughout the term of the Contract. The disaster recovery plan shall be subject to ASES review upon reasonable notice to Contractor. Contractor shall maintain reasonable safeguards against the destruction, loss, intrusion and unauthorized alteration of printed materials and data in its possession. At a minimum, Contractor shall perform (i) incremental daily back-ups, (ii) weekly full backups, and (iii) such additional back-ups as the Contractor may determine to be necessary to maintain such reasonable safeguards. |
Page 217 of 301
| 17.10.2 | Both Parties recognize that a failure by the Contractor’s Network may adversely impact ASES business and operations, as the responsible party for the GHP. Therefore, in the event that the Contractor’s Network designed to deliver the services herein contemplated becomes unable, or is anticipated to become unable, to deliver such services on a timely basis, Contractor shall Immediately notify ASES by telephone, and shall work closely with ASES to fix the problem. In the event that Contractor fails to provide such required notice to ASES and such delay in the notification has a material and adverse effect upon ASES and/or Enrollees, ASES may terminate this Contract for cause as provided in Article 35 of this Contract. |
| 17.10.3 | Within five (5) Calendar Days upon ASES’s request, Contractor will deliver a copy of the then current ASES’s Data Content to ASES in a mutually acceptable form and format which is useable and readable and understandable by ASES. |
| 17.11 | Health Information Organization and Health Information Exchange (HIE) Requirements |
| 17.11.1 | The Contractor shall initiate the active participation in any Health Information Organization that offers Health Information Exchange services, in order to integrate the Enrollees’ Personal Health Information, facilitate access to and retrieval of their clinical Data to provide safer and more timely, efficient, effective, and equitable patient-centered care. The HIO participation is also required to support the analysis of the health of the population. As required by ASES, the Contractor shall be active in a HIO and cooperate with this effort. |
| 17.11.2 | ASES shall retain the right to request from the Contractor the active participation in the Puerto Rico Health Information Exchange Corporation (PRHIEC), the Puerto Rico HIO State Designated Entity, in order to achieve the effective alignment of activities across Medicaid and Commonwealth public health programs, to avoid duplicate efforts and to ensure integration and support of a unified approach to information exchange for the GHP Program. |
| 17.11.3 | The Contractor shall verify that the HIO complies with all Information System standards and requirements for interoperability and security capabilities dictated by ONCHIT, and other Federal and Puerto Rico regulations. |
| 17.11.4 | The Contractor shall work with Network Providers and staff to encourage |
Page 218 of 301
ARTICLE 18 REPORTING
| 18.1 | General Requirements |
| 18.1.1 | ASES may, at its discretion, require the Contractor to submit additional reports both ad hoc and recurring. If ASES requests any revisions to the reports already submitted, the Contractor shall make the changes and re-submit the reports, according to the time period and format specified by ASES. |
| 18.1.2 | The Contractor shall submit all reports to ASES in the manner and format prescribed by ASES. |
| 18.1.3 | The Contractor shall submit all reports, including but not limited to those required by Law 72, Article 7, Section 2 in a manner and format prescribed by ASES. |
| 18.1.4 | All reports submitted to ASES containing information about a Provider must include the Provider’s National Provider Identifier (NPI), if applicable. |
| 18.1.5 | All quantitative reports shall include a summary table that presents Data over time including monthly, quarterly and/or year-to-date summaries as directed by ASES. |
| 18.1.6 | ASES’s requirements regarding reports, report content, and frequency of submission are subject to change at any time during the term of the Agreement upon no less than forty-five (45) Calendar Days prior written notice to the Contractor. A list of required reports is provided in Attachment 16. The Contractor shall comply with all changes specified in writing by ASES, after ASES has discussed such changes with the Contractor. ASES shall notify the Contractor, in writing, of changes to existing required report content, format or schedule at least fourteen (14) Calendar Days prior to implementing the reporting change. ASES shall notify the Contractor, in writing, of new reports at least forty-five (45) Calendar Days prior to implementing the new report. The Contractor shall be held harmless if ASES fails to meet this requirement for any changes for existing reports. However, the Contractor is not otherwise relieved of any responsibility for the submission of late, inaccurate or otherwise incomplete reports. The first submission of a report revised by ASES to include a change in Data requirements or definition will not be subject to penalty for accuracy. |
| 18.1.7 | The Contractor shall submit reports timely and in proper format. The submission of late, inaccurate, or otherwise incomplete reports constitutes failure to report. “Timely submission” shall mean that the report was submitted on or before the date it was due. “Accuracy” shall mean the report was prepared according to the specific written guidance, including report template, provided by ASES to the Contractor. All elements must be met for each required report submission. Therefore, the report must be timely, accurate and contain an analysis. If any portion of the report element is not met, the report is deemed in “error” and the Contractor will be considered to not be in compliance with the Contract and will be subject to intermediate sanctions and or liquidated damages and/or fines in accordance with Articles 19 and 20 of this Contract. The Contractor shall not be penalized if an error in a previously submitted report is identified by the Contractor and reported to ASES prior to ASES’s identification of the error. Corrected reports in this type of situation will be submitted to ASES in a timeframe determined by ASES after consulting with the Contractor. Failure to comply with the agreed upon timeframes for correction and resubmission shall be subject to intermediate sanctions and or liquidated damages and/or fines in accordance with Articles 19 and 20 of this Contract. |
Page 219 of 301
| 18.1.8 | Each report must include an analysis, which shall include, at a minimum: (i) identification of any changes compared to previous reporting periods as well as trending over time; (ii) an explanation of said changes (positive or negative); (iii) an action plan or performance improvement activities addressing any negative changes; and (iv) any other additional information pertinent to the reporting period. ASES may assess intermediate sanctions, liquidated damages and/or fines in accordance with Articles 19 and 20 of this Contract for failure to address any of these requirements. The above Data requirements may be represented in charts, graphs, tables and any other Data illustrations to demonstrate findings. |
| 18.1.9 | The Contractor shall review, as part of its continuous improvement activities, timeliness and accuracy of reports submitted to ASES to identify instances and patterns of non-compliance. The Contractor shall perform an analysis identifying any patterns or issues of non-compliance and shall implement quality improvement activities to improve overall performance and compliance. |
| 18.1.10 | The Contractor shall submit all reports to ASES, unless indicated otherwise in this Contract, according to the schedule below. Failure to report timely may result in intermediate sanctions, liquidated damages and/or fines in accordance with Articles 19 and 20. Reports or other required Data shall be received on or before scheduled due dates. |
| 18.1.11 | The Contractor shall submit all reports to ASES, unless indicated otherwise in this Contract, according to the schedule below: |
Page 220 of 301
|
DELIVERABLES
|
DUE DATE
|
|
|
Weekly Reports
|
Friday of the following Week
|
|
|
Monthly Reports
|
Fifth (5th) Calendar Day of the following month
|
|
|
Quarterly Reports
|
Thirtieth (30th) Calendar Day of the following month
|
|
|
Semi-Annual Reports
|
January 31 and July 31 of the Contract year
|
|
|
Annual Reports
|
Ninety (90) Calendar Days after the end of the fiscal year
|
| 18.1.12 | If a report due date falls on a weekend or a Commonwealth holiday, receipt of the report the next Business Day is acceptable. |
| 18.1.13 | Extensions to report submission dates will be considered by ASES after the Contractor has contacted the ASES designated point of contact via email at least twenty-four (24) hours in advance of the report due date. Extension for submission of reports should be under rare and unusual circumstances. If ASES grants an extension, and the report is submitted before the extended deadline, the report(s) will be considered timely and not subject to penalty for timeliness. Not requesting an extension within at least twenty-four (24) hours of the report due date is considered failure to report timely. |
| 18.1.14 | Anytime a report is rejected for any reason, the Contractor shall resubmit the report within ten (10) Business Days from notification of the rejection or as directed by ASES. |
| 18.1.15 | The Contractor shall submit all reports electronically to ASES’s FTP site unless directed otherwise by ASES. ASES shall provide the Contractor with access to the FTP site. The email generated by the FTP upload will be used as the time stamp for the submission of the report(s). |
| 18.1.16 | ASES shall provide feedback to the Contractor regarding format and timeliness of reports within forty-five (45) Calendar Days from the due date of the report. |
| 18.1.17 | All reports in the reporting templates provided to the Contract require Contractor certification. The Authorized Certifier or an equivalent position as delegated by the Contractor and approved by ASES, shall review the accuracy of language, analysis, and Data in each report prior to submitting the report to ASES. The Authorized Certifier shall include a signed attestation each time the report is submitted. The attestation must include a certification, based on best knowledge, information, and belief, as to the accuracy, completeness and truthfulness of the Data in the report. Reports will be deemed incomplete if an attestation is not included. |
Page 221 of 301
| 18.1.18 | The Contractor Data transfers shall occur in standard format as prescribed by ASES and will be compliant with HIPAA and Federal regulations. The Contractor shall submit in formats as prescribed by ASES so long as ASES’s direction does not conflict with any Federal law. |
| 18.2 | Specific Requirements |
| 18.2.1 | The following section provides an overview and description of all reports required by this Contract. The details and requirements of the reports are subject to change at the discretion of ASES. |
| 18.2.2 | Administrative Reports |
| 18.2.2.1 | The Contractor shall submit a monthly Call Center Report that provides information about the Enrollee services, Provider services, and nurse advice lines. The report shall, at a minimum, include by language queue: (i) number of calls received; (ii) number of calls answered; (iii) abandonment rate; (iv) number of calls answered within thirty (30) seconds; and (v) call topics. |
| 18.2.2.2 | The Contractor shall submit a quarterly Enrollee Enrollment Materials Report regarding the mailing of initial and replacement Enrollee Enrollment materials including Enrollee ID cards, Enrollee handbooks, and Provider directories. The Data in the report shall be reported separately for initial mailings to new Enrollees and requests for replacement materials for current Enrollees. The report shall include, at a minimum, the following: (i) number of ID cards, handbooks and Provider directories mailed during the month regardless of whether the request was made by phone, online or in person; (ii) number of ID cards, handbooks and Provider directories mailed within Contract standards; and (iii) number of ID cards, handbooks and Provider directories not mailed within Contract standards. |
| 18.2.2.3 | The Contractor shall submit a quarterly Fraud, Waste, and Abuse Report that provides information regarding suspicious activity, Fraud, Waste, and Abuse cases, recoupments, Cost Avoidance, Referrals, and other information as directed by ASES. At a minimum, the report shall include: (i) Enrollee name and ID number; (ii) Provider name, Provider type and NPI; (iii) source and date of Complaint; (iv) nature of Complaint (including alleged persons or entities involved, category of services, factual explanation of the allegation and dates of contact); (v) all communications between the Contractor and the Provider about the Complaint; (vi) approximate dollars involved or amount paid to the Provider during past three (3) years (whichever is greater); (vii) disciplinary measures imposed, if any; and (viii) legal disposition of the case. The Contractor shall also include in the report as a qualitative analysis; information regarding investigative activities, corrective actions, prevention efforts and the results of prevention efforts. |
Page 222 of 301
| 18.2.2.4 | The Contractor shall submit a quarterly Employee and Contractor Suspensions/Debarment Report that captures information pertaining to employees and Contractors that have been suspended or debarred from participating in the program. |
| 18.2.2.5 | The Contractor shall submit an annual Compliance Plan that meets the requirements outlined in Section 13.2 of the Contract. |
| 18.2.2.6 | The Contractor shall submit an annual Program Integrity Plan that meets the requirements outlined in Section 13.3 of the Contract. |
| 18.2.2.7 | The Contractor shall submit a report regarding Activities of the Advisory Board ten (10) Calendar Days following the meeting date. The report shall, at a minimum, include: (i) a summary of the Contractor’s approach to inviting Enrollees that represent all eligibility groups/populations; (ii) meeting agenda; (iii) a list of meeting attendees; (iv) meeting minutes; and (v) the date, time, and location of the next meeting. |
| 18.2.2.8 | The Contractor shall submit a monthly Privacy and Confidentiality Report. The report shall provide information on any Incidents that involve the loss, theft or unauthorized use or access of Enrollee PHI. The report shall include, at a minimum: (i) the date of the Incident; (ii) the date of notification to ASES; (iii) the nature and scope of the Incident; (iv) the Contractor’s response to the Incident; and (v) any mitigating measures taken by the Contractor to prevent similar Incidents. |
| 18.2.2.9 | The Contractor shall submit an annual Systems Incident Report. The report shall provide information on any Incidents that involve unauthorized access to the Contractor’s systems, databases or servers. This report shall be provided at least annually, but the Contractor shall provide the report ten (10) Business Days following an Incident. The report shall include, at a minimum, the date of the Incident, the date of notification to ASES, the nature and scope of the Incident, the Contractor’s response to the Incident, and the mitigating measures taken by the Contractor to prevent similar Incidents in the future. “Port scans” or other unsuccessful queries to the Contractor’s Information System shall not be considered a privacy/security Incident for purposes of this report. |
| 18.2.3 | Claims |
Page 223 of 301
| 18.2.3.1 | The Contractor shall submit a monthly Claims Activity Report. At a minimum, this report shall identify: (i) the number of Claims received; (ii) number of Claims denied (by reason); (iii) number of Claims paid; (iv) number of Claims pending (by reason); (v) and the total amount paid for all Providers (by Provider category) specified by ASES in accordance with Section 16.7 of this Contract. |
| 18.2.3.2 | The Contractor shall submit Encounter Data in a standardized format as specified by ASES (see Section 16.8 of this Contract) and transmitted electronically to ASES on a monthly basis. The Contractor shall provide any information and/or Data requested in a format to be specified by ASES as required to support the validation, testing or auditing of the completeness and accuracy of Encounter Data submitted by the Contractor. |
| 18.2.4 | Covered Services |
| 18.2.4.1 | The Contractor shall submit a quarterly Care Management Report to assess the Contractor’s performance and timeliness associated with the care management process. The report shall present Data separately for Enrollees new to the care management process and those Enrollees who are receiving ongoing care management. The report shall include, at a minimum information regarding: (i) number of initial assessments completed; (ii) number of Enrollees receiving intensive one-on-one counseling interventions from Care Managers; (iii) number of Prior Authorizations and denials of Prior Authorizations by the Contractor for conditions included in Special Coverage; (iv) number of Adults screened for depression using the PHQ-9; (vi) number of children screened using the ASQ; (vi) number of children screened for depression using the ASQ-SE; (vii) number of Enrollees unable to be reached for initial assessments; and (viii) number of Enrollees with Chronic Behavioral Health Conditions. |
| 18.2.4.2 | The Contractor shall submit a Disease Management Report that includes Information on Utilization of physical and Behavioral Health Services by Enrollees in Disease Management (DM) as described in Section 7.8.3. The report shall, at a minimum, include the following Data: (i) physical health services received by Enrollees in Disease Management; (ii) Behavioral Health services received by Enrollees in Disease Management; and (iii) the number of Enrollees in Disease Management with the following conditions: asthma, depression, diabetes (type 1 and type 2), congestive heart failure, hypertension, obesity and chronic renal disease (Level 1 and Level 2). |
Page 224 of 301
| 18.2.4.3 | The Contractor shall submit an annual Maternal and Pre-Natal Plan as described in Section 7.5.8 of this Contract. The plan shall include, at a minimum: (i) description of the program; (ii) maternal and pre-natal services offered through the program; and (iii) number of Enrollees in the program. |
| 18.2.4.4 | The Contractor shall submit an annual Wellness Plan that at a minimum, describes the Contractor’s plans to promote Enrollee wellness in accordance with Section 12.5.8 of the Contract. |
| 18.2.4.5 | The Contractor shall submit an annual EPSDT Plan as described in Section 7.9 of this Contract. |
| 18.2.4.6 | The Contractor shall submit an annual CMS 416 Report that measures and documents EPSDT screening and participation rates. In addition to the requirements in the CMS 416 Report, the Contractor shall report on any additional Data that ASES determines is necessary for monitoring and compliance purposes. |
| 18.2.4.7 | The Contractor shall submit a quarterly Executive Director Report that provides information on selected GHP populations and providers. The report shall include, at a minimum, information regarding: (i) GHP Enrollees, (ii) Enrollees in special programs (including Enrollees with Special Coverage), (iii) PPN and Network Providers, (iv) services for children, (v) dental services and (vi) hospitalizations. |
| 18.2.5 | Provider Reports |
| 18.2.5.1 | The Contractor shall submit a monthly National Provider List (NPL) Report that provides information on the number of Providers with and without assigned lives in the Contractor’s General and PPN network. At a minimum, the report shall include information on the: (i) Network Provider’s name; (ii) Network Provider’s specialty; (iii) Network Provider’s NPI; (iv) Network Provider’s specialty code; (v) Network Provider license number; (vi) Network Provider’s primary office location; (vii) Network Provider’s office hours; (viii) Network Provider’s Credentialing status; (ix) Network Provider PMG affiliation; (x) ratio of Network Provider to Enrollees (including PCPs, Behavioral Health Providers); and (xi) the number of assigned lives (if applicable) to Network Providers. For facilities the report shall include: (i) EIN; (ii) name of the entity; (iii) municipality code; (iv) Provider type code; and (v) the Provider’s NPI. |
| 18.2.5.2 | The Contractor shall submit quarterly Geographical Access reports using geographic Information Systems software that allows ASES to analyze, at a minimum, the following: (i) description of geographic systems software utilized to generate geographic Access report; (ii) description of monitoring activities to ensure Access standards are met and that Enrollees have Access to services; (iii) description of gaps in geographic Access and methodologies used to identify them; (iv) Data on all service locations for PCP and all specialty Providers; and (v) number of Enrollees that are currently assigned to the Network Provider (PCPs only) by Service Region. |
Page 225 of 301
| 18.2.5.3 | The Contractor shall submit a quarterly Provider Credentialing and Re-Credentialing Report that lists all Providers credentialed or re-credentialed during the reporting period. At a minimum, the report shall include: (i) each Network Provider’s name; (ii) the Network Provider’s specialty; (iii) the Network Provider’s NPI; (iv) the Network Provider’s primary office location; and (v) the date of Credentialing/Re-Credentialing. |
| 18.2.5.4 | The Contractor shall submit a quarterly Provider Suspensions and Terminations Report that lists by name all Network Provider suspensions or terminations. This report shall include information on all Network Providers. At a minimum, the report shall include: (i) each Network Provider’s name; (ii) the Network Provider’s specialty; (iii) the Network Provider’s NPI; (iv) the Network Provider’s primary city; (v) reason(s) for the action taken; and (vi) the effective date of the suspension or termination. If the Contractor has taken no action against Providers during the quarter this should be documented in the report. |
| 18.2.5.5 | The Contractor shall submit an annual Provider Training and Outreach Plan/Evaluation Report describing the Contractor’s plans to educate Providers and an Evaluation Report to evaluate the initiatives in the plan and present findings of lessons learned. Both the plan and the evaluation report shall be submitted in narrative format. The Provider Training and Outreach Plan shall describe Provider training initiatives including, but not limited to, the following: (i) Prior Authorizations; (ii) Claims/Encounter Data submissions; (iii) how to access Ancillary Service Providers; (iv) Enrollee rights and responsibilities; (v) quality improvement program/ initiatives; (vi) Provider and Enrollee Appeals and Grievances; (vii) recoupment of funds processes and procedures; and (viii) EPSDT benefit requirements, including Preventive Services guidelines. The Evaluation Report shall specify the training topic (s), the targeted Providers, the content of the training, the training schedule (including dates/times and locations), and training methods. The Contractor shall, upon request, provide information regarding Provider training and Outreach initiatives including, but not limited to, the following: (i) target audiences; (ii) location of training/event; (iii) date of training/event; (iv) topics; (v) funds expended; and (vi) number and types of attendees. |
Page 226 of 301
| 18.2.5.6 | The Contractor shall submit an annual Provider Satisfaction Survey Report that encompasses physical and Behavioral Health Network Providers. The report shall include but not be limited to, a summary of the Provider survey methods and findings for physical and Behavioral Health Network Providers separately and an analysis of opportunities for improvement. See Section 12.6 of this Contract for additional information regarding Provider Satisfaction Surveys. |
| 18.2.6 | Quality |
| 18.2.6.1 | The Contractor shall submit a quarterly Grievances and Appeals Report. The Contractor shall submit reports of all Provider and Enrollee Grievances (informal and formal), Appeals, Notices of Actions and Administrative Law Hearings utilizing the ASES-provided reporting templates and codes. The report will also capture Enrollee comments and inquiries made through the Contractor’s website. |
| 18.2.6.2 | The Contractor shall submit a quarterly Quality Improvement Performance (QIP) Report. The Contractor shall use measurements and performance guidelines outlined in the Quality Improvement Procedure Manual. The report shall, at a minimum, include Data on: (i) preventive clinical programs; (ii) performance measures; and (iii) ER quality program. |
| 18.2.6.3 | The Contractor shall submit an annual QAPI Program Description as described in Section 12.2 of this Contract. The description shall, at a minimum, include the following: (i) program overview, methodology, performance measures and analysis on the MCO’s ER Quality Incentive Program; (ii) program overview, methodology, performance measures and analysis of the MCO’s HEDIS Quality Incentive Program; (iii) program overview, methodology, performance measures and analysis of the Contractor’s preventive clinical programs; and (iv) program overview, methodology, performance measures and analysis on the MCO’s Performance Improvement Projects. |
| 18.2.6.4 | The Contractor shall submit an annual Enrollee Satisfaction Survey Report that includes, but is not limited to, a summary of the Enrollee survey methods, findings, analysis and evaluation. The report shall present information separately for CAHPS and ECHO. The survey and findings shall be presented by populations as determined by ASES (e.g., Adults, children, Behavioral Health and Chronic Conditions). The report must provide an action plan addressing areas for improvement of the Contractor as identified in the survey results. Refer to Section 12.6 of this Contract for additional information regarding the survey. |
Page 227 of 301
| 18.2.6.5 | The Contractor shall submit an annual Audited HEDIS Results Report. The Contractor shall use only NCQA published HEDIS standardized measures that specify how MCOs collect, audit, calculate and report performance information. |
| 18.2.6.5.1 | Each HEDIS submission must require the following information: |
| 18.2.6.5.1.1 | A signed attestation that will provided by ASES; |
| 18.2.6.5.1.2 | Quantitative Data and qualitative Data collected according to HEDIS technical specifications. This Data shall be reported to ASES in an excel workbook and as a searchable .PDF document; and |
| 18.2.6.5.1.3 | A final HEDIS Compliance Audit Report and supporting documentation according to HEDIS Compliance Audit standards, policies and procedures. |
| 18.2.6.5.2 | As specified in Section 12.3.4.6 of this Contract. The Contractor will submit the following standardized HEDIS measures in a format specified by ASES: |
| 18.2.6.5.2.1 | Effectiveness of Care: Prevention and Screening Measures |
| 18.2.6.5.2.1.1 | Childhood immunization; |
| 18.2.6.5.2.1.2 | Breast cancer screening; |
| 18.2.6.5.2.1.3 | Cervical cancer screening; |
| 18.2.6.5.2.1.4 | Chlamydia screening; |
| 18.2.6.5.2.1.5 | Adult Body Mass Index (“BMI”) assessment; and |
| 18.2.6.5.2.1.6 | Weight assessment and counseling for nutrition and physical activities for children and adolescents. |
Page 228 of 301
| 18.2.6.5.2.2 | Effectiveness of Care: Respiratory Condition Measures |
| 18.2.6.5.2.2.1 | Use of appropriate medication for people with asthma; and |
| 18.2.6.5.2.2.2 | Appropriate treatment for children with upper respiratory conditions. |
| 18.2.6.5.2.3 | Effectiveness of Care: Cardiovascular Conditions |
| 18.2.6.5.2.3.1 | Cholesterol management for people with cardiovascular conditions; and |
| 18.2.6.5.2.3.2 | Controlling high blood pressure. |
| 18.2.6.5.2.4 | Access/Availability of Care Measures |
| 18.2.6.5.2.4.1 | Comprehensive diabetes care (with all its components); |
| 18.2.6.5.2.4.2 | Adult Access to preventive/outpatient health services; |
| 18.2.6.5.2.4.3 | Annual dentist visit; |
| 18.2.6.5.2.4.4 | Children and adolescent Access to PCPs; |
| 18.2.6.5.2.4.5 | Prenatal and postpartum care; |
| 18.2.6.5.2.4.6 | Frequency of ongoing prenatal care; |
| 18.2.6.5.2.4.7 | Healthy Child Care visits in the first fifteen (15) months of life; and |
| 18.2.6.5.2.4.8 | Adolescent well care visits. |
| 18.2.6.5.2.5 | Behavioral Health Measures: Effectiveness of Medical Care and Access |
| 18.2.6.5.2.5.1 | Antidepressant Medication Management; |
| 18.2.6.5.2.5.2 | Follow up care for children with prescribed ADHD medication; |
Page 229 of 301
| 18.2.6.5.2.5.3 | Follow up after hospitalization for mental illness and engagement of alcohol and other drug dependence treatment; |
| 18.2.6.5.2.5.4 | Identification of alcohol and other drug treatment services; and |
| 18.2.6.5.2.5.5 | Behavioral Health Utilization. |
| 18.2.6.5.3 | ASES may add, change, or remove HEDIS reporting requirements with sixty (60) Calendar Days’ notice in advance of the effective date of the addition, change, or removal. |
| 18.2.6.5.4 | When requested, the Contractor shall submit Data to ASES for standardized performance measures, within specified timelines and according to the established procedures Data collection and reporting. The Contractor shall collect valid and reliable Data, using qualified staff and personnel to collect the Data. Failure of the Contractor to follow Data collection and reporting requirements may result in sanctions, liquidated damages and/or other fines in accordance with Articles 19 and 20 of this Contract. |
| 18.2.7 | Utilization Management |
| 18.2.7.1 | The Contractor shall submit a quarterly Utilization Management Report that includes Information on Utilization of physical and Behavioral Health Services. The report shall, at a minimum, include the following Data: (i) physical health services received by Enrollees; (ii) Behavioral Health Services received by Enrollees; (iii) services received by Enrollees with specific Chronic Conditions; (iv) EPSDT Utilization, screening and list of EPSDT-eligible children who had not had an appointment as described in Section 7.9.2.4; (v) register of Enrollees in Special Coverage; (vi) register of Enrollees receiving pre-natal services; (vii) number of Enrollees in autism Special Coverage program; (viii) number of co-occurring diagnosis (substance abuse including alcohol and Behavioral Health); and (ix) statistical Data on (a) the twenty (20) most prevalent Behavioral Health diagnosis; (b) the twenty (20) most prevalent substance abuse diagnosis; (c) the twenty (20) most prevalent diagnosis leading to emergency room Utilization. |
| 18.2.7.2 | The Contractor shall submit a quarterly Admissions and Readmissions Report that provides information by region(s) regarding the number of Enrollees who are readmitted to a facility such as, an RHC, FQHC, detoxification facility, short term intervention center or hospital within thirty (30) Calendar Days of a previous discharge by Service Region. The report shall provide Data by procedure codes and populations as specified by ASES. |
Page 230 of 301
| 18.2.7.3 | The Contractor shall submit a quarterly Prior Authorization Report that includes Prior Authorization Information by service. The report shall, at a minimum, include the following Data: (i) the service for which a Prior Authorization is being requested; (ii) the number of initial and continued requests for each service; (iii) the number of requests approved, denied (administrative and clinical), pended for each service (initial and continued); and (iv) the number of terminations and reductions in service. |
| 18.2.7.4 | The Contractor shall submit a quarterly Integration Report. The report shall, at a minimum, include the following Data: (i) number of Enrollees receiving care management for physical health and behavioral needs; (ii) number of Referrals to physical health Providers by Behavioral Health Providers; (iii) number of short-term counseling appointments provided and fulfilled; (iv) number of Referrals to care management; (v) number of Behavioral Health assessments performed and the number of Referrals to Behavioral Health Providers made; and (vi) the ratio of Enrollees receiving Behavioral Health Services per each PMG. |
| 18.2.7.5 | The Contractor shall submit an annual UM Program Description/Work Plan Report. The program description shall include a description of the structure and accountability mechanisms. At a minimum, the description shall include: (i) scope of the UM program, (ii) goals and objective of the UM program, (iii) program structure including organizational structure, authority and accountability and committee structure; (iv) description of UM networking and support; and (v) a description of the following UM processes: pre-service review, concurrent review, post service review, discharge planning and emergency department services. The Work Plan shall include: (i) planned UM improvement activities that will address quality of service delivery; (ii) Disease Management; (iii) specific mechanism for periodic Data tracking and trending of UM performance indicators; and (iv) periodic evaluations of the effectiveness of UM interventions. |
| 18.2.8 | Systems |
| 18.2.8.1 | The Contractor shall submit a monthly Systems Availability and Performance Report that provides information on availability and unavailability by major system as well as response times for the Contractor’s confirmation of Contractor’s Enrollment and electronic Claims management functions, as measured within the Contractor’s Span of Control. The report shall meet the requirements of Section 17.5 of this Contract. |
Page 231 of 301
| 18.2.8.2 | The Contractor shall submit a quarterly Business Continuity and Disaster Recovery (“BC-DR”) Test Report for review and written approval as specified by ASES in accordance with Section 17.5 of this Contract. The Contractor shall conduct quarterly tests of the BC-DR system and report the findings of the test results with the system generated log report within thirty (30) Calendar Days of the date of the test. |
| 18.2.8.3 | The Contractor shall submit an annual BC-DR Plan in accordance with Section 17.5 of this Contract. |
| 18.2.9 | Financial Management |
| 18.2.9.1 | The Contractor shall submit a monthly Per Member Per Month Payment Disbursement Report. The report shall present the distribution of the Capitation or other service payments to Providers, Claim expenses by coverage, reserves, and administrative expenses. |
| 18.2.9.2 | The Contractor shall submit a monthly Actuarial Data Report in a format specified by ASES. |
| 18.2.9.3 | The Contractor shall submit a monthly Enrollee TPL Health Insurance Report as described in Section 23.4.7.1 of this Contract. The report is due the fifth (5th) Calendar Day after the close of the month during which the Contractor learns that an Enrollee has new health insurance coverage, or casualty insurance coverage, or of any change in an Enrollee’s health insurance coverage. The Contractor shall impose a corresponding requirement on its Providers to notify the Contractor of any newly discovered coverage. |
| 18.2.9.4 | The Contractor shall submit a quarterly Retention Fund Report. The report shall include outcomes information on the Quality Incentive Program as described in Section 12.5. The report shall contain, at a minimum: (i) Data on the amount of PMPM withheld for current reporting period; and (ii) Data on the amount of PMPM withheld in the previous reporting period(s). |
| 18.2.9.5 | The Contractor shall submit a quarterly Unaudited Financial Statement Report. The Contractor shall submit (i) a separate accounting of activities relating to each Service Region, and (ii) a consolidated section accounting for all GHP program activities. |
Page 232 of 301
| 18.2.9.6 | The Contractor shall submit an annual Physician Incentive Plan Report that provides adequate information about the Contractor’s monitoring activities for the Physician Incentive Plan as described in Section 23.6. The Contractor shall submit, at a minimum: (i) description of the Physician Incentive Plan; (ii) description of incentive arrangements; (iii) description and Data on percentage of Withhold or bonus attached to the plan; and (iv) the number of Providers participating in the plan and the number of Enrollees affected. |
| 18.2.9.7 | The Contractor shall submit an annual Report on Controls Placed in Operation and Tests of Operating Effectiveness. The report must meet all standards and requirements of the AICPA’s SSA E 16, for the Contractor’s operations performed for ASES under this Contract. |
| 18.2.9.8 | The Contractor shall submit annual Audited Financial Statements. The Contractor shall provide ASES with copies of its audited financial statements following Generally Accepted Accounting Principles (“GAAP”) in the US, at its own cost and charge, for the duration of the Contract, and as of the end of each fiscal year during the Contract Term, regarding the financial operations related to the GHP Program. The statements shall provide (i) a separate accounting of activities relating to each Service Region, and (ii) a consolidated section accounting for all GHP Program activities. These reports shall be submitted to ASES no later than ninety (90) Calendar Days after the close of the fiscal year. |
| 18.2.9.9 | The Contractor shall submit a quarterly Cost Avoidance Report. The report shall describe as specified by ASES the Contractor’s findings regarding routine audits of Network Providers to evaluate cost-avoidance performance. |
| 18.2.9.10 | The Contractor shall submit an annual Disclosure of Information on Annual Business Transactions as described in Section 23.7.4 of this Contract. |
| 18.2.9.11 | The Contractor shall submit an annual Report to Puerto Rico Insurance Commissioner’s Office in the format agreed upon by the National Association of Insurance Commissioners (NAIC). |
| 18.2.9.12 | The Contractor shall submit an Annual Corporate Report at the close of the MCO’s fiscal/calendar year. |
Page 233 of 301
ARTICLE 19 ENFORCEMENT – INTERMEDIATE SANCTIONS
| 19.1 | General Provisions |
| 19.1.1 | In monitoring Contractor’s compliance with the terms of the Contract, ASES may impose intermediate sanctions, and/or liquidated damages, and/or fines pursuant to Puerto Rico Act No. 134, for Contractor’s failure to comply with the terms and conditions of this Contract (as further specified in Articles 19 and 20 of the Contract). |
| 19.1.2 | In the event the Contractor incurs any proscribed conduct or otherwise is in default as to any applicable term, condition, or requirement of this Contract, and in accordance with any applicable provision of 42 CFR 438.700 and Section 4707 of the Balanced Budget Act of 1997, at any time following the Effective Date of the Contract, the Contractor agrees that, in addition to the terms of Section 35.1.1 of this Contract, ASES may impose intermediate sanctions against the Contractor for any such default in accordance with this Article 19. ASES may not impose intermediate sanctions with respect to a specific event of default of the Contractor for which liquidated damages sought to be imposed or are imposed against the Contractor in accordance with Article 20 of this Contract. ASES may impose both intermediate sanctions and fines pursuant to Puerto Rico Act No. 134. The assessment of intermediate sanctions under this Contract cannot and will not limit the power or authority of ASES to impose any other fines, civil money penalties, sanctions, or other remedies recognized by the Commonwealth or Federal laws or regulations. |
| 19.1.3 | Notwithstanding any intermediate sanctions imposed upon the Contractor under this Article 19, other than Contract termination, the Contractor shall continue to provide all Covered Services and other Benefits under this Contract. |
| 19.1.4 | ASES shall have the right impose the following intermediate sanctions: |
| 19.1.4.1 | Civil Money Penalty – ASES may impose a civil money penalty for the following categories of events. |
| 19.1.4.1.1 | Category 1 - A civil money penalty in accordance with any applicable provision of 42 CFR 438.700 up to one-hundred thousand dollars ($100,000) per determination shall be imposed for this category. The following constitute Category 1 events: |
| 19.1.4.1.1.1 | Acts that discriminate among Enrollees on the basis of their health status or need for health care services. This includes termination of Enrollment or refusal to reenroll a Potential Enrollee, except as permitted under the Medicaid program, or any practice that would reasonably be expected to discourage Enrollment by beneficiaries whose medical or Behavioral Health condition or history indicates probable need for substantial future medical or Behavioral Health Services. Notwithstanding the foregoing, ASES may impose a civil money penalty in the amount of fifteen thousand dollars ($15,000) per each (i) Potential Enrollee that was not enrolled because of discriminatory practices as described above and/or (ii) discriminatory practices imposed on Enrollees, subject to the overall limit of one-hundred thousand dollars ($100,000) per each determination. |
Page 234 of 301
| 19.1.4.1.1.2 | The misrepresentation or falsification of information submitted to ASES and/or CMS. |
| 19.1.4.1.2 | Category 2 - A civil money penalty in accordance with any applicable provision of 42 CFR 438.700 up to twenty-five thousand dollars ($25,000) per determination shall be imposed for this category. The following constitute Category 2 events: |
| 19.1.4.1.2.1 | Failure by the Contractor to substantially provide Medically Necessary Services that the Contractor is required to provide, under applicable law or under this Contract, to an Enrollee under this Contract. |
| 19.1.4.1.2.2 | Misrepresentation or falsification by the Contractor of information that it furnishes to an Enrollee, Potential Enrollee, or Provider. |
| 19.1.4.1.2.3 | Failure by the Contractor to comply with the requirements for Physician Incentive Plans, as set forth in 42 CFR 422.208 and 422.210. |
| 19.1.4.1.2.4 | The distribution by the Contractor, directly or indirectly through any Agent or independent contractor, of Marketing Materials that have not been prior approved by ASES or that contain false or materially misleading information. |
| 19.1.4.1.3 | Category 3 – Pursuant to 42 CFR 438.704 (c), ASES may impose a civil money penalty for the Contractor’s imposition of premiums or charges in excess of the amounts permitted under the Medicaid program. The maximum amount of the penalty is the greater of twenty-five thousand dollars ($25,000) or double the amount of the excess charges. ASES will deduct from the penalty the amount of overcharge and return it to the affected Enrollees. |
Page 235 of 301
| 19.1.4.2 | Temporary Management - ASES may appoint temporary management for the Contractor’s GHP operations, as provided in 42 C.F.R. 438.702 and 42 C.F.R. 438.706 as a result of Contractor’s: |
| 19.1.4.2.1 | Continued egregious behavior, including but not limited to behavior described in Categories 1 through 3 of this Article 19; |
| 19.1.4.2.2 | Behavior that is contrary to, or is non-compliant with, Sections 1903(m) or 1932 of the Social Security Act, as amended, found at 42 U.S.C. §§ 1396b (m) and 1396u-2; |
| 19.1.4.2.3 | Actions which have caused substantial risk to an Enrollee’s health; and/or |
| 19.1.4.2.4 | Behavior which has led ASES to determine that temporary management is necessary to ensure the health of Contractor’s Enrollees while improvements to remedy Category 1 through 3 violations are being made, or until the Contractor’s orderly termination or reorganization. |
| 19.1.4.2.5 | If temporary management is appointed for any reason specified in Sections 19.1.4.2 above, such temporary management will cease once ASES has, in its discretion, determined that the sanctioned behavior will not re-occur. |
| 19.1.4.3 | Enrollment Termination – ASES may grant Enrollees the right to terminate Enrollment without cause, and notify the affected Enrollees of their right to disenroll when: |
| 19.1.4.3.1 | The Contractor has engaged in continued egregious behavior, including but not limited to behavior described in Categories 1 through 3 of this Article 19; |
| 19.1.4.3.2 | The Contractor has engaged in behavior that is contrary to, or is non-compliant with, Sections 1903(m) or 1932 of the Social Security Act, as amended, found at 42 U.S.C. §§ 1396b (m) and 1396u-2; |
| 19.1.4.3.3 | The Contractor has taken Actions that have caused substantial risk to Enrollees’ health; |
Page 236 of 301
| 19.1.4.3.4 | ASES determines that temporary management is necessary or convenient to ensure the health of the Contractor’s Enrollees; or |
| 19.1.4.3.5 | ASES determines that such Enrollment termination is necessary or appropriate to remedy Category 1 through 3 violations. |
| 19.1.4.4 | Enrollment Suspension – ASES may suspend all new Enrollments, including default Enrollment, after the effective date of the intermediate sanction and until the intermediate sanction is no longer in effect. |
| 19.1.4.5 | Payment Suspension – ASES may suspend payment of the PMPM Payment for Enrollees enrolled after the effective date of the intermediate sanction and until CMS or ASES is satisfied that the reason for imposition of the intermediate sanction no longer exists and is not likely to re-occur or upon the Termination Date of the Contract. |
| 19.1.4.6 | Mandatory Imposition of Certain Intermediate Sanctions – ASES shall impose the temporary management and Enrollment suspension intermediate sanctions described in Sections 19.1.4.2 and 19.1.4.3 above, if ASES finds that the Contractor has repeatedly failed to meet substantive requirements in Sections 1903(m) or 1932 of the Social Security Act, as amended, found at 42 U.S.C. §§ 1396b (m) and 1396u-2. |
| 19.1.4.7 | Subject to Article 35 of this Contract, in lieu of imposing a sanction allowed under this Article 19, ASES may terminate this Contract, without any liability whatsoever (but subject to making any payments due under this Contract through any such date of termination), if the terms of a Corrective Action Plan implemented pursuant to this Article 19 to address a failure specified in Category 1 or Category 2 of this Article 19 are not implemented to ASES’s approval or if such failure continues or is not corrected, to ASES’s satisfaction. |
| 19.2 | Notice of Intention to Impose Intermediate Sanctions |
| 19.2.1 | In the event that ASES performs an investigation or acknowledges facts regarding a possible contractual non-compliance, according to Section 19, ASES shall issue a Notice of Intention to Impose Intermediate Sanctions, delivered thorough US Postal Service Certified Mail, to the Contractor that includes the following: |
| 19.2.1.1 | A brief description of the facts; |
Page 237 of 301
| 19.2.1.2 | Applicable Puerto Rico and Federal laws and regulations, or Contract provisions; |
| 19.2.1.3 | The Contractor’s non-compliance with Puerto Rico and Federal laws and regulations as referenced in the Contract; |
| 19.2.1.4 | The Contractor’s breach of applicable intermediate sanction Contract provisions; |
| 19.2.1.5 | ASES’s authority to determine and impose intermediate sanctions under this Article 19; |
| 19.2.1.6 | The amount of potential, or the Contractor’s exposure to, intermediate sanctions, and how they were computed; and |
| 19.2.1.7 | At ASES discretion, a statement describing the Contractor’s right to submit a Corrective Action Plan within fifteen (15) Calendar Days of receipt of the notice of administrative inquiry under this Article 19. |
| 19.2.2 | At ASES discretion, the Contractor shall have the right to submit a Corrective Action Plan within fifteen (15) Calendar Days of receipt of the Notice of Intention to Impose Intermediate Sanctions. If the Contractor timely submits a Corrective Action Plan acceptable and approved by ASES, ASES shall not impose intermediate sanctions on the facts described in its Notice of Intention to Impose Intermediate Sanctions pursuant to this Article 19. The Contractor shall not have the right to submit a Corrective Action Plan for an incident similar to a previous incident for which the Contractor had previously been under a Corrective Action Plan approved by ASES. |
| 19.2.3 | Nevertheless, if after the investigation is finished or the information at hand reveals a contractual non-compliance, or violation to the Puerto Rico and Federal laws and regulations administered by ASES, and at ASES’s discretion it cannot be corrected through a Corrective Action Plan, ASES will not issue a Notice of Intention to Impose Intermediate Sanctions, and instead shall notify the imposition of Intermediate Sanctions. |
| 19.2.4 | If the Contractor fails to comply with any material provision under a Corrective Action Plan submitted to ASES pursuant to Section 19.2.2 above, ASES may impose: |
| 19.2.4.1 | A daily five-thousand dollar ($5,000) civil money penalty, up to a maximum total of one-hundred thousand dollars ($100,000), for Contractor’s ongoing failure to comply with any provision deemed material by ASES under the Corrective Action Plan; or |
Page 238 of 301
| 19.2.4.2 | The applicable intermediate sanction for any or all behavior that resulted in the Contractor’s submission of the Corrective Action Plan pursuant to Section 19.1 above. |
| 19.3 | Notice of Imposition of Intermediate Sanctions |
| 19.3.1 | Prior to the imposition of intermediate sanctions, ASES will issue a notification, delivered thorough US Postal Service Certified Mail, to the Contractor that includes the following: |
| 19.3.1.1 | A brief description of the facts; |
| 19.3.1.2 | Applicable Puerto Rico and Federal laws and regulations, or Contract provision; |
| 19.3.1.3 | ASES’s determination to impose intermediate sanctions; |
| 19.3.1.4 | Intermediate sanctions imposed and their effective date; |
| 19.3.1.5 | Methodology for the civil money penalty calculation or determination of the intermediate sanctions; and |
| 19.3.1.6 | At ASES’s discretion, a statement describing the Contractor’s option to submit a Corrective Action Plan within thirty (30) Calendar Days following receipt of the notice of imposition of intermediate sanctions or in lieu thereof to seek administrative review of the imposed intermediate sanctions pursuant to Section 19.4. |
| 19.3.2 | At ASES’s discretion, the Contractor may submit a Corrective Action Plan to ASES within thirty (30) Calendar Days of receipt of the notice of intermediate sanctions. If the Contractor submits a Corrective Action Plan under this section, ASES may only recover ten percent (10 %) of the civil money penalty, if any, imposed under the notice of intermediate sanctions, and/or discontinue the imposition of the intermediate sanction. Alternatively, the Contractor may seek administrative review of the imposition of intermediate sanctions pursuant to Section 19.4. |
| 19.3.3 | Contractor’s right to seek administrative review of ASES’s Actions by Puerto Rico’s Court of Appeals, San Juan Panel, within thirty (30) Calendar Days of the Contractor’s receipt of the notice of intermediate sanctions. |
| 19.3.4 | ASES shall notify CMS in writing of the imposition of intermediate sanctions within thirty (30) Calendar Days of imposing the intermediate sanctions and concurrently provide the Contractor with a copy of such notice. |
Page 239 of 301
| 19.4 | Administrative Review – The Contractor has the right to seek administrative review of the imposition of intermediate sanctions, including but not limited to civil money penalties, by ASES, pursuant to the following procedure: |
| 19.4.1 | The Contractor has the right within thirty (30) Calendar Days following receipt of the notice of imposition of intermediate sanctions to seek administrative review in writing of ASES’s determination and any such intermediate sanctions, pursuant to Act 72 or under any other applicable law or regulation. |
| 19.4.2 | As part of the administrative review, the Parties shall cooperate with the examining officer, and follow all applicable procedures for the administrative review. |
| 19.4.3 | Upon completion of the administrative review, the examining officer may recommend: |
| 19.4.3.1 | Confirm the intermediate sanctions; |
| 19.4.3.2 | Modify or amend the intermediate sanctions pursuant to applicable law or regulation; or |
| 19.4.3.3 | Eliminate the imposed intermediate sanctions. |
| 19.4.4 | Once the sanction becomes final ASES shall withhold the amount of the sanction from the PMPM Payment. |
| 19.4.5 | In addition to the actions described under Section 19.4.3, the examining officer may recommend the delivery and implementation of a Corrective Action Plan with respect to Contractor’s failure to comply with the terms of this Contract as set forth in ASES’ notice of intermediate sanctions. |
| 19.4.6 | ASES shall notify CMS in writing of any modification in the imposition of intermediate sanctions through the administrative review process within thirty (30) Calendar Days of receipt of the examining officer’s determination, and concurrently provide the Contractor with a copy of such notice. |
Page 240 of 301
| 19.5 | Judicial Review – To the extent administrative review is sought by the Contractor pursuant to Section 19.4, the Contractor has the right to seek judicial review of ASES’s actions by the Puerto Rico Court of Appeals, San Juan Panel, within thirty (30) Calendar Days of the notice of final determination issued by ASES. |
| 19.6 | Federal Sanctions - Payments provided under this Contract will be denied for new Enrollees when, and for so long as, payment for those Enrollees is denied by CMS in accordance with the requirements in 42 C.F.R. 438.730. |
ARTICLE 20 ENFORCEMENT - LIQUIDATED DAMAGES AND OTHER REMEDIES
| 20.1 | General Provisions |
| 20.1.1 | ASES may impose intermediate sanctions, liquidated damages, and/or fines pursuant to Puerto Rico Act No. 134 (as indicated in Articles 19 and 20 of this Contract). |
| 20.1.2 | In the event the Contractor is in default as to any applicable term, condition, or requirement of this Contract, and in accordance with any applicable provision of 42 CFR 438.700 and Section 4707 of the Balanced Budget Act of 1997, at any time following the Effective Date of this Contract, the Contractor agrees that, in addition to the terms of Section [35.1.1] of this Contract, ASES may assess liquidated damages against the Contractor for any such default, in accordance with this Article 20. ASES may not impose liquidated damages with respect to a specific event of default of Contractor for which intermediate sanctions, including but not limited to civil monetary penalties, sought to be imposed or are imposed against the Contractor under Article 19. The Parties further acknowledge and agree that the specified liquidated damages are reasonable and the result of a good faith effort by the Parties to estimate the anticipated or actual harm caused by the Contractor’s breach and are in lieu of any other financial remedies to which ASES may otherwise have been entitled. The assessment of liquidated damages under the Contract cannot and will not limit the power or authority of ASES to impose fines, civil money penalties, sanctions, or other remedies under Article 19 of this Contract or otherwise under by Commonwealth or Federal laws or regulations, including fines pursuant to Puerto Rico Act No. 134. |
| 20.1.3 | Notwithstanding any sanction, including liquidated damages, imposed upon the Contractor, other than Contract termination, the Contractor shall continue to provide all Covered Services and other Benefits under this Contract. |
| 20.1.4 | The Parties have determined that the Contractor’s breach or failure to comply with the terms and conditions of this Contract for which liquated damages may be assessed under this Article 20 shall be divided into four (4) categories of events. |
Page 241 of 301
| 20.2 | Category 1 |
| 20.2.1 | Liquidated damages in accordance with any applicable provision of this Contract of up to one-hundred thousand dollars ($100,000) per violation, Incident or occurrence may be imposed for Category 1 events. The following constitute Category 1 events: |
| 20.2.1.1 | Material non-compliance with an ASES or CMS directive, determination or notice to cease and desist not otherwise described in Article 19 or other provision of this Article 20, provided that the Contractor has received prior written notice with respect to such specific material non-compliance, and afforded an opportunity to cure within a reasonable period to be determined by ASES in its sole discretion. |
| 20.3 | Category 2 |
| 20.3.1 | Liquidated damages in accordance with any applicable provision of this Contract of up to twenty-five thousand dollars ($25,000) per violation, Incident, or occurrence may be imposed for Category 2 events. The following constitute Category 2 events: |
| 20.3.1.1 | Subject to ASES compliance with its obligations under Article 22 of this Contract, repeated noncompliance by the Contractor with any material obligation that adversely affects the services that the Contractor is required to provide under Article 7 of this Contract; |
| 20.3.1.2 | Failure of the Contractor to assume its duties and obligations under this Contract in accordance with the transition timeframes specified herein; |
| 20.3.1.3 | Failure of the Contractor to terminate a Provider that imposes Co-Payments or other cost-sharing on Enrollees that are in excess of the fees permitted by ASES, as listed on Attachment 8 (ASES will deduct the amount of the overcharge and return it to the affected Enrollees); |
| 20.3.1.4 | Failure of the Contractor to address Enrollees’ Complaints, Appeals, and Grievances, and Provider disputes, within the timeframes specified in this Contract; |
| 20.3.1.5 | Failure of the Contractor to comply with the confidentiality provisions in accordance with 45 CFR 160 and 164; and |
| 20.3.1.6 | Failure of the Contractor to comply with a subcontracting requirement in the Contract. |
Page 242 of 301
| 20.4 | Category 3 |
| 20.4.1 | Liquidated damages in accordance with any applicable provision this Contract of five-thousand dollars ($5,000) per day may be imposed for Category 3 events. The following constitute Category 3 events: |
| 20.4.1.1 | Failure to submit required reports in the timeframes prescribed in Article 18; |
| 20.4.1.2 | Submission of incorrect or deficient Deliverables or reports in accordance with Article 18 of this Contract; |
| 20.4.1.3 | Failure to comply with the Claims processing standards as follows: |
| 20.4.1.3.1 | Failure to process and finalize to a paid or denied status ninety-five percent (95%) of all Clean Claims within thirty (30) Calendar Days of receipt; |
| 20.4.1.3.2 | Failure to process and finalize to a paid or denied status one hundred percent (100%) of all Clean Claims within fifty (50) Calendar Days of receipt; and |
| 20.4.1.3.3 | Failure to process Unclean Claims as specified in Section 16.10.3 of this Contract; |
| 20.4.1.4 | Failure to pay Providers interest at the rate identified in and otherwise in accordance with Section 16.10.2 of this Contract when a Clean Claim is not adjudicated within the Claims processing deadlines; |
| 20.4.1.5 | Failure to comply with the quarterly submission of EPSDT reports to ASES according to the guidelines to be issued by ASES under Section 7.9.1; |
| 20.4.1.6 | Failure to notify PCPs of the gaps in care analysis in accordance with the EPSDT guidelines to be issued by ASES under Section 7.9.1; |
| 20.4.1.7 | Reserved; |
| 20.4.1.8 | Failure to seek, collect and/or report Third Party Liability information as provided in Section 23.4 of this Contract; and |
| 20.4.1.9 | Failure of Contractor to issue written notice to Enrollees upon Provider’s termination of a Provider as described in Section 10.4.3 of this Contract. |
Page 243 of 301
| 20.5 | Category 4 |
| 20.5.1 | Liquidated damages as specified below may be imposed for Category 4 events. The following constitute Category 4 events: |
| 20.5.1.1 | Failure to implement the BC-DR plan as follows: |
| 20.5.1.1.1 | Implementation of the (BC-DR) plan exceeds the proposed time by two (2) or less Calendar Days: five thousand dollars ($5,000) per day up to day 2; |
| 20.5.1.1.2 | Implementation of the (BC-DR) plan exceeds the proposed time by more than two (2) and up to five (5) Calendar Days: ten thousand dollars ($10,000) per each day beginning with day 3 and up to day 5; |
| 20.5.1.1.3 | Implementation of the (BC-DR) plan exceeds the proposed time by more than five (5) and up to ten (10) Calendar Days, twenty-five thousand dollars ($25,000) per day beginning with day 6 and up to day 10; |
| 20.5.1.1.4 | Implementation of the (BC-DR) plan exceeds the proposed time by more than ten (10) Calendar Days: fifty thousand dollars ($50,000) per each day beginning with day 11; |
| 20.5.1.2 | Unscheduled System Unavailability in violation of Article 17, in ASES’s discretion, two hundred fifty dollars ($250) for each thirty (30) minute period or portions thereof; |
| 20.5.1.3 | Failure to make available to ASES or its Agent, valid extracts of Encounter Information for a specific month within fifteen (15) Calendar Days of the close of the month: five hundred dollars ($500) per day. After thirty (30) Calendar Days of the close of the month: two thousand dollars ($2,000) per Calendar Day; |
| 20.5.1.4 | Failure to correct a system problem not resulting in System Unavailability within the allowed timeframe, where failure to complete was not due to the action or inaction on the part of ASES as documented in writing by the Contractor: |
| 20.5.1.4.1 | One (1) to fifteen (15) Calendar Days late: two hundred and fifty dollars ($250) per Calendar Day for days 1 through 15; |
| 20.5.1.4.2 | Sixteen (16) to thirty (30) Calendar Days late: five hundred dollars ($500) per Calendar Day for days 16 through 30; and |
Page 244 of 301
| 20.5.1.4.3 | More than thirty (30) Calendar Days late: one thousand dollars ($1,000) per Calendar Day for days 31 and beyond; and |
| 20.5.1.5 | Failure to meet the GHP Service Line performance standards: |
| 20.5.1.5.1 | One-thousand dollars ($1,000) for each percentage point that is below the target answer rate of eighty percent (80%) in thirty (30) seconds; |
| 20.5.1.5.2 | One-thousand dollars ($1,000) for each percentage point that is above the target of a three percent (3%) Blocked Call rate; and |
| 20.5.1.5.3 | One-thousand dollars ($1,000) for each percentage point that is above the target of a five percent (5%) Abandoned Call rate. |
| 20.6 | Other Remedies |
| 20.6.1 | Subject to Article 35 of this Contract, in lieu of imposing a Remedy allowed under this Article 20, ASES may elect to terminate this Contract, without any liability whatsoever (but subject to making any payments due, if any, under this Contract through any such date of termination), if the terms of a Corrective Action Plan implemented pursuant to this Article 20 to address a failure specified in Category 1 or Category 2 of this Article 20 are not implemented to ASES’s satisfaction or if such failure continues or is not corrected, to ASES’s sole satisfaction. |
| 20.6.2 | In the event of non-compliance by the Contractor with Article 18 of this Contract, ASES shall have the right to Withhold, with respect to Article 18, a sum not to exceed ten percent (10%) of the PMPM Payment for the following month and for continuous consecutive months thereafter until such noncompliance is cured and corrected to ASES’ satisfaction in lieu of imposing any liquidated damages, penalties or sanctions against the Contractor hereunder. ASES shall release the Withhold of the PMPM Payment to the Contractor within two (2) Business Days after the corresponding event of noncompliance is cured to ASES’s sole satisfaction. |
| 20.7 | Notice of Intention to Impose Liquidated Damages and/or Other Article 20 Remedies |
| 20.7.1 | In the event that ASES performs an investigation or acknowledges facts regarding a possible contractual non-compliance, according to Section 20, ASES shall issue a Notice of Intention to Impose Liquidated Damages and/or Other Article 20 remedies, delivered thorough US Postal Service Certified Mail, to the Contractor that includes the following: |
Page 245 of 301
| 20.7.1.1 | A brief description of the facts; |
| 20.7.1.2 | Applicable Puerto Rico and Federal laws and regulations, or Contract provision; |
| 20.7.1.3 | The Contractor’s non-compliance with Puerto Rico and Federal laws and regulations; |
| 20.7.1.4 | The Contractor’s breach of applicable Contract provisions and event categories that could result in remedies or liquidated damages under this Article 20; |
| 20.7.1.5 | ASES’s authority to determine and seek liquidated damages or other remedies against the Contractor under this Article 20; |
| 20.7.1.6 | The amount of potential, or Contractor’s exposure to liquidated damages, or other Article 20 remedies, and how they were computed; and |
| 20.7.1.7 | At ASES’s discretion, a statement describing the Contractor’s right to submit a Corrective Action Plan within fifteen (15) Calendar Days of receipt of the notice of intent of imposition of liquidated damages or other article 20 remedies, under this Article 20. |
| 20.7.2 | At ASES’s discretion, the Contractor shall have the right to submit a Corrective Action Plan within fifteen (15) Calendar Days of receipt of the Notice of Intention to Impose Liquidated Damages or Other Article 20 Remedies issued pursuant to this Article 20. If the Contractor submits a Corrective Action Plan to ASES on a timely basis, ASES shall not impose damages or other remedies under this Article 20 based on the facts described in its Notice of Intention to Impose Liquidated Damages or other article 20 remedies, if the terms contained in the Corrective Action Plan are acceptable to ASES in its sole discretion. |
| 20.7.3 | A Notice of Intention to Impose Liquidated Damages or Other Article 20 Remedies, shall not constitute ASES’s final or partial determination of liquidated damages. Thus, any administrative inquiries made are not subject to administrative review under Section 20.8.2 and would be construed to be premature rendering any administrative examiner without jurisdiction to review the matter. |
| 20.7.4 | If the Contractor fails to comply with any material provision under a Corrective Action Plan submitted to ASES pursuant to Section 20.7.2 above, ASES may impose: |
| 20.7.4.1 | A daily amount of five-thousand dollars ($5,000) in liquidated damages, up to a maximum total amount of one-hundred thousand dollars ($100,000), for the Contractor’s failure to comply with any material provision part or condition of the Corrective Action Plan; and/or |
Page 246 of 301
| 20.7.4.2 | The applicable Article 20 Remedy for any or all behavior that resulted in the submission of Corrective Action Plan pursuant to Section 20.7.1 above. |
| 20.8 | Notice of Imposition of Liquidated Damages and/or Other Remedies – |
| 20.8.1 | Prior to the imposition of liquidated damages and/or any other remedies under this Article 20, ASES will issue a notification, delivered thorough US Postal Service Certified Mail, to the Contractor that includes the following: |
| 20.8.1.1.1 | A brief description of the facts; |
| 20.8.1.1.2 | Applicable Puerto Rico and Federal laws and regulations, or Contract provision; |
| 20.8.1.1.3 | ASES’s determination to assess and impose liquidated damages and/or any other Article 20 Remedy; |
| 20.8.1.1.4 | Liquidated damages and/or any other Article 20 Remedy imposed and their effective date; |
| 20.8.1.1.5 | Methodology for the liquidated damages and/or any other Article 20 Remedy calculation; and |
| 20.8.1.1.6 | In ASES’s discretion, a statement describing the Contractor’s option to submit a Corrective Action Plan within thirty (30) Calendar Days of receipt of a notice of liquidated damages or other remedies pursuant to this Article 20 or in lieu thereof seek administrative review of the imposed liquidated damages and/or any other Article 20 Remedy pursuant to Section 20.8.2. |
| 20.8.1.2 | At ASES’s discretion the Contractor may submit a Corrective Action Plan to ASES within thirty (30) Calendar Days of receipt of a notice of liquidated damages or other remedies pursuant to this Article 20. If the Contractor submits a Corrective Action Plan under this section, under terms and conditions acceptable to ASES, ASES may only recover ten percent (10%) of the liquidated damages or any other Remedy imposed under such notice of liquidated damages and/or may discontinue the imposition of the liquidated damages. Alternatively, the Contractor may seek administrative review of the imposition of remedies pursuant to Article 20. |
Page 247 of 301
| 20.8.1.3 | The Contractor’s right to seek administrative review of ASES’s actions by Puerto Rico’s Court of Appeals, San Juan Panel, within thirty (30) Calendar Days of the Contractor’s receipt of the notice of liquidated damages. |
| 20.8.2 | Administrative Review – The Contractor has the right to seek administrative review of the imposition of liquidated damages and/or any other Remedy under this Article 20.8.2, pursuant to the following procedure: |
| 20.8.2.1 | The Contractor has the right within thirty (30) Calendar Days following receipt of the notice of liquidated damages and/or any other Remedy under this Article 20 to seek administrative review in writing of ASES’s determination and any such remedies, pursuant to Act 72 or under any other applicable law or regulation. |
| 20.8.2.2 | As part of the administrative review, the Parties shall cooperate with the examining officer, and follow all applicable procedures for the administrative review. |
| 20.8.2.3 | Upon the completion of the administrative review, the examining officer may recommend to: |
| 20.8.2.3.1 | Confirm the liquidated damages and/or any other Remedy; |
| 20.8.2.3.2 | Modify or amend the liquidated damages and/or any other Remedy; or |
| 20.8.2.3.3 | Eliminate the imposed liquidated damages and/or any other Remedy. |
| 20.8.2.4 | Once the sanction becomes final ASES shall withhold the amount of the sanction from the PM/PM payment. |
| 20.8.2.5 | In addition to the actions described under Section 20.8.2.3, the examining officer shall have the right to recommend the institution of a Corrective Action Plan with respect to the Contractor’s alleged noncompliance described in ASES’s notice of liquidated damages. |
| 20.9 | Judicial Review – To the extent administrative review is sought by the Contractor pursuant to Section 20.8.2, the Contractor has the right to seek judicial review of ASES’s actions by the Puerto Rico Court of Appeals, San Juan Panel, within thirty (30) Calendar Days of the Contractor’s receipt of the notice of final determination issued by ASES. |
ARTICLE 21 CONTRACT TERM
| 21.1 | Subject to and upon the terms and conditions herein, this Contract shall be in full force and effect on October 31, 2014 and shall terminate on June 30, 2017. The Contractor shall begin providing Covered Services to Enrollees on April 1, 2015 which shall be deemed to be the Implementation Date of the Contract. The foregoing notwithstanding, ASES, subject to Article 35 reserves the right, prior written notice of ninety (90) Calendar Days, to amend or partially terminate the Contract at any time to implement a demonstrative plan to incorporate the new public health policies and/or strategies of the Commonwealth in any Service Region or portion thereof. Upon written notice of amendment or partial termination of this Contract pursuant to this Article 21, ASES will evaluate in good faith a renegotiation of Per Member Per Month fees payable under this Contract. |
Page 248 of 301
| 21.2 | The Contract Term shall begin at 12:01 a.m., Puerto Rico Time, Effective Date of the Contract and shall continue until 11:59 p.m., Puerto Rico time, on June 30, 2017. |
| 21.3 | The provision of Covered Services and Benefits to Enrollees by the Contractor under this Contract shall begin on April 1, 2015 which is the Implementation Date of the Contract. |
| 21.4 | The PMPMPMPM Payment rate shall be negotiated for every fiscal year covered by the Contract (namely from April 1, 2015 to June 30, 2016, and from July 1, 2016 to June 30, 2017. Any increase in the PMPM Payment shall be subject to ASES’s determination that the proposed new amount is actuarially sound. |
| 21.5 | The Contract shall expire at the close of the Contract Term unless earlier terminated under Article 35. |
| 21.6 | ASES is hereby granted the option to renew this Contract for an additional term of up to one (1) fiscal year, which shall begin on July 1, 2017 and end at midnight on June 30, 2018. The terms of the renewal shall be negotiated, but any increase in PMPM Payment shall be subject to ASES’s determination that the proposed new amount is actuarially sound. The option to renew the Contract shall be exercisable solely and exclusively by ASES. As to each term, the Contract shall be terminated absolutely at the close of the then current Commonwealth fiscal year without further obligation by ASES. |
ARTICLE 22 PAYMENT FOR SERVICES
| 22.1 | General Provisions |
| 22.1.1 | The actual PMPM Payment will be equal to the number of Enrollees as of the last day of the month preceding the month in which payment is made, multiplied by the negotiated PMPM Payment agreed to between the Contractor and ASES for each Service Region covered by the Contract. The rate is specified in Attachment 11. The due date for the PMPM Payment to the Contractor shall be the fifth (5th) day of each month. However, ASES shall have the right to make partial payments throughout the month, provided that payment in full will be made on or before the last day of each month. The PMPM Payment made based upon the number of Enrollees as of the last day of the preceding month will be reconciled to the actual number of Enrollees for that month when that information is available and appropriate PMPM Payment adjustments will be made. |
Page 249 of 301
| 22.1.2 | ASES shall provide PMPM Payments only for those Enrollees for whom ASES has received adequate notification of Enrollment from the Contractor as of the date specified by ASES, per Section 5.2.2. ASES will work with the Contractor to establish the amount of any PMPM Payments that are due to the Contractor for any Enrollee that has retroactive coverage per Section 5.1.3.1 |
| 22.1.3 | ASES will have the discretion to recoup payments made to the Contractor for the following: |
| 22.1.3.1 | Enrollees incorrectly enrolled with more than one Contractor; |
| 22.1.3.2 | Enrollees who die prior to the Enrollment month for which the payment was made; or |
| 22.1.3.3 | Enrollees whom ASES later determines were not eligible for Medicaid during the Enrollment month for which payment was made. |
| 22.1.4 | Any such payments due to ASES from the Contractor will be offset from future payments to the Contractor. |
| 22.1.5 | The Contractor shall have the right to recoup from Providers or other persons to whom the Contractor has made payment for any payments made for which ASES has recouped the PMPM Payment. |
| 22.1.6 | The PMPM Payment for Enrollees not enrolled for the full month shall be determined on a pro rata basis by dividing the monthly Capitation amount by the number of days in the month and multiplying the result by the number of days including and following the Effective Date of Enrollment. The Contractor is entitled to a PMPM Payment for each Enrollee as of the Effective Date of Enrollment, including the period referred to in Section 5.2.2. |
| 22.1.7 | Payment for services under this Contract will not commence before Implementation Date of the Contract. |
| 22.1.8 | Payments for the first month of program operations under this Contract will be made only upon a determination by ASES that the Contractor has complied with all of its obligations for the implementation of this Contract, including a finding by ASES that the Contractor has satisfied the readiness review, and the Contractor’s submission of initial Deliverables as specified in Attachment 12 to this Contract. |
Page 250 of 301
| 22.1.9 | In order to receive payments from ASES, the Contractor shall provide to ASES, and keep current, its tax identification number, billing address, and other contact information, as required by ASES. |
| 22.1.10 | The Contractor acknowledges that the payments agreed to under the terms of this Contract in addition to any applicable cost-sharing as provided in Attachment 8 constitute full payment for Covered Services and Benefits under GHP. ASES will have no responsibility for payment for Covered Services and Benefits beyond that amount unless the Contractor has obtained prior written approval, in the form of a Contract amendment, authorizing an increase in the total payment. |
| 22.1.11 | Fee-for-Service amounts paid by the Contractor for Claims, or Capitation payments made by the Contractor derived or otherwise based on Encounter Data submitted by Providers, resulting from services determined not to be Medically Necessary by the Contractor, will not be considered in the Contract's experience for purposes of prospective rate adjustments. |
| 22.1.12 | Pursuant to the terms of this Contract, should ASES assess liquidated damages or other Remedies for the Contractor’s noncompliance or deficiency with the terms of this Contract, such amount shall be withheld from the PMPM Payment for the following month, and for continuous consecutive months thereafter until such noncompliance or deficiency is corrected at ASES’s satisfaction |
| 22.1.13 | The Contractor shall maintain all the Utilization and financial Data related to this Contract duly segregated from its regular accounting system including, but not limited to, the general ledger. In addition, the Contractor shall maintain separate Utilization and financial Data for each Service Region covered under this Contract. |
| 22.1.14 | Administrative expenses to be included in determining the experience of the program are those directly related to this Contract. Separate allocations of expenses from the Contractor’s insurance plans, other than GHP, from the Contractor’s related companies, from the Contractor’s parent company, or from other entities will be reflected or made a part of the financial Data described in the preceding section. Any pooling of operating expenses with other of the Contractor's groups, cost-shifting, financial consolidation or the implementation of other combined financial measures is expressly forbidden. |
| 22.1.15 | The following administrative expenses are unallowable for purposes of reporting program expenditures and prospective rate setting: |
| 22.1.15.1 | Costs of entertainment, festivities and other activities for the recreation of the personnel of the insurer, including employees, managers, directors, officers or Third Parties, such as: expenses for parties, dinners, food, alcoholic beverages, gifts, etc.; |
Page 251 of 301
| 22.1.15.2 | Costs of advertising, public relations and marketing, except as provided in Section 6.14 of this Contract; |
| 22.1.15.3 | Costs of recruiting office, managerial and executive personnel; |
| 22.1.15.4 | Payroll costs related to corporate officers and employees exceeding the equivalent time dedicated to work related to the GHP program if these same officers and employees also perform duties in support of other lines of business. Payroll expenses to be charged to GHP shall be reasonable according to industry standards and the only time that may be charged is when they perform work specific to the GHP program; |
| 22.1.15.5 | Any payment related to the liquidation of payroll or marginal benefits due to termination (severance) and restructuring of the company (downsizing), including “parachute” clauses, for board of directors, corporate officers or executives of the Contractor; |
| 22.1.15.6 | The Contractor’s employer contributions to savings plans for employees, directors, officers or executives of the Contractor; |
| 22.1.15.7 | Costs related to the awarding and exercise of stock options of employees, directors, officers or executives of the Contractor; |
| 22.1.15.8 | Payment of productivity bonuses, or bonuses of another nature, to directors, officers, executives and employees, excluding the Christmas bonus as required by the law; |
| 22.1.15.9 | Costs of trips to the US or to foreign countries, whether for business, continued education or pleasure; |
| 22.1.15.10 | Expenses or payments related to vacations, including, but not limited to, stay expenses, hotel, air, land or sea transportation, food, gratuity, etc.; |
| 22.1.15.11 | First class fees for air tickets, and travel expenses including charter flights or in commercial lines, within or outside of Puerto Rico; |
| 22.1.15.12 | Payments related to attendance and stay at conventions, seminars, workshops, or continued education, for executives, directors, officers or employees of the Contractor, whether within or outside of Puerto Rico; |
| 22.1.15.13 | Payments related to educational expenses such as: training, retraining, studies, scholarships, memberships, dues, employee licenses, etc.; |
Page 252 of 301
| 22.1.15.14 | Payments related to automobile expenses, including rent, lease, purchase and depreciation, car allowance, maintenance expenses, gasoline, repairs, etc.; |
| 22.1.15.15 | Costs of transportation, including taxi service, airplanes, charters, urban train, automobiles, and gasoline or diesel for motor vehicles; |
| 22.1.15.16 | Payment of cellular phone expenses, including Internet access; |
| 22.1.15.17 | Monies used for gifts, gratuity, contests, prizes, donations, charity, etc.; |
| 22.1.15.18 | Any commissions, management fees or similar charges from related parties without express approval from ASES; |
| 22.1.15.19 | Categorizing expenses under a general category such as overhead, other, miscellaneous, is expressly forbidden; and |
| 22.1.15.20 | Any other expense not allowed by ASES. |
| 22.1.16 | The Contractor shall provide ASES every month with a PMPM Payment Disbursement Report. This document shall present the distribution of the Capitation or other service payments to Providers, Claim expenses by coverage, reserves, and administrative expenses. Failure to comply with the requirements contained herein may be cause for the imposition of liquidated damages as outlined in Section 20 of this Contract. |
| 22.1.17 | The Contractor shall provide to ASES, on a monthly basis, actuarial Data in a format specified in the Actuarial Report provided by ASES. Failure to comply with the requirements contained herein may be cause for the imposition of liquidated damages as outlined in Section 20 of this Contract. |
| 22.1.18 | The profit of the Contractor and Subcontractors for each fiscal year of the Contract Term shall not exceed two point five percent (2.5 %) of the PMPM Payment (Excess Profit). In the event that the profit exceeds this amount as a result of the positive impact the high quality services provided by the Contractor and Sub-Contractors had on the Enrollees Health, the Parties shall share the Excess Profit in proportions of fifty percent (50%) for the Contractor and Subcontractors, and fifty percent (50%) for ASES. For the purpose of this section high quality services will be measured on the Contractor’s compliance with eighty-five percent (85%) of the quality metrics as established by ASES under this Contract. In the event ASES discovers the existence of Excess Profit by means of an audit during the Control and Supervision Plan or the Contractor does not meet the high quality services standard mentioned in this section, ASES is entitled to one hundred percent (100%) of the Excess Profit. |
Page 253 of 301
| 22.1.19 | ASES will determine the Contractor’s Excess Profit based on the Contractor’s and Subcontractors’ audited financial statements submitted annually to ASES pursuant to Sections 23.1.3 and 18.2.9.8 of this Contract, and the validation of the IBNR reserve by ASES’s actuary. The Excess Profit calculation will include the entire fiscal year (total aggregated earned premium for all Service Regions). ASES will determine Excess Profit using the actual medical expenses and the contracted administrative fee portion of the PMPM. ASES shall notify the Contractor of ASES’s determination of the Contractor’s Excess Profit within fifteen (15) Calendar Days of receipt by ASES of the Contractor’s audited financial statement. The Contractor shall remit the portion of Excess Profit payable to ASES within fifteen (15) Calendar Days of receiving the notice of Excess Profit determination from ASES. The same regulations shall apply to any and all Subcontractors. |
| 22.1.20 | To comply with 42 CFR 433.312, the Contractor shall refund (i) the share of the Overpayment due to ASES within eleven (11) months of the discovery and (ii) the share of an Overpayment due to ASES within fifteen (15) Calendar Days from a final judgment on a Fraud, Waste, or Abuse Action. |
| 22.2 | Contractor Objections to Payment |
| 22.2.1 | If the Contractor wishes to contest the amount of payments made by ASES in accordance with the terms outlined in Section 22.1 for services provided under the terms of this Contract, the Contractor shall submit to ASES all relevant documentation supporting the Contractor’s objection no later than thirty (30) Calendar Days after payment is made. Once this term has ended, the Contractor forfeits its right to claim any additional amounts. |
| 22.2.2 | After the Contractor’s submission of all relevant information, the Contractor and ASES will meet to discuss the matter. If after discussing the matter and analyzing all relevant Data it is subsequently determined that an error in payment was made, the Contractor and ASES will develop a plan to remedy the situation, which would include a timeframe for resolution agreed to by both Parties, within a time period mutually agreed upon by both Parties. |
| 22.3 | Retention Fund for Quality Incentive Program |
| 22.3.1 | ASES shall maintain a Retention Fund of the PMPM Payment each month as part of the Quality Incentive Program described in Section 12.5 according to the following table: |
Page 254 of 301
|
Time Period (Relative from Effective Date of Contract Term)
|
Retention Fund Percentage
|
|
4/1/2015 through 12/31/2015
|
0 % (9-month)
|
|
1/1/2016 through 6/30/2016
|
1.0% (until FY16 end)
|
|
7/1/2016 through 6/30/2017
|
2.0% (until FY17 end)
|
A portion of the retained amount shall be associated with each of the Quality Incentive initiatives as follows:
| 22.3.1.1 | Performance measures (see Section 12.5.4.1, two percent (2%); |
| 22.3.1.2 | Preventive clinical programs (see Section 12.5.4.2, one percent (1%); and |
| 22.3.1.3 | Emergency room use indicators (see Section 12.5.4.3, two percent (2%). |
| 22.3.2 | With respect to each Quality Incentive initiative, ASES, as indicated herein, shall upon expiration of each quarter during the Contract Term conduct a review to determine if the Contractor has met the applicable performance targets for that period according to the following process: |
| 22.3.2.1 | The Contractor shall submit a quarterly report no later than thirty (30) Calendar Days after the end of each quarter regarding each of the performance indicators to be evaluated by ASES (listed in Section 12.5); |
| 22.3.2.2 | No later than thirty (30) Calendar Days after receipt of the Contractor’s quarterly reports, ASES shall determine if the Contractor has met the applicable performance objectives for each measure for that period; |
| 22.3.2.3 | If the Contractor is in full compliance with the applicable performance targets for said period, ASES shall disburse to the Contractor, no later than thirty (30) Calendar Days after ASES determines compliance with the performance objectives, the portion of the PMPM Payment associated with each initiative for such period. |
ARTICLE 23 FINANCIAL MANAGEMENT
| 23.1 | General Provisions |
| 23.1.1 | The Contractor shall be responsible for the sound financial management of Puerto Rico and Federal funds provided to the Contractor under the GHP Program. |
Page 255 of 301
| 23.1.2 | The Contractor shall notify ASES in writing of any loans or other special financial arrangements made between the Contractor and any PMG or any other Provider. Any such loans shall strictly conform to the legal requirements of Federal and Puerto Rico anti-Fraud and anti-kickback laws and regulations. |
| 23.1.3 | The Contractor shall provide ASES with copies of its audited financial statements following Generally Accepted Accounting Principles (“GAAP”) in the US, at its own cost and expenses, for the duration of the Contract, and as of the end of each fiscal year during the Contract Term, regarding the financial operations related to the GHP. The statements shall provide (1) a separate accounting of activities relating to each Service Region, and (2) a consolidated section accounting for all GHP activities. These reports shall be submitted to ASES no later than ninety (90) Calendar Days after the close of the fiscal year of ASES. |
| 23.1.4 | The Contractor shall provide to ASES a copy of its Annual Report required to be filed with the Office of the Insurance Commissioner, as applicable, in the format agreed upon by the National Association of Insurance Commissioners (NAIC), for the year ended on December 31, 2014, and subsequently thereafter, during the Contract Term and any renewals, not later than March 31 of each year. |
| 23.1.5 | The Contractor shall provide to ASES unaudited financial statements for each quarter during the Contract Term, not later thirty (30) Calendar Days after the close of each quarter. The Contractor shall submit (1) a separate accounting of activities relating to each Service Region, and (2) a consolidated section accounting for all GHP activities. |
| 23.1.6 | The Contractor shall provide to ASES a copy of the annual corporate report of its parent company at the close of the calendar year. |
| 23.1.7 | The Contractor shall maintain adequate procedures and controls to ensure that any payments pursuant to this Contract are properly made. In establishing and maintaining such procedures, the Contractor shall provide for separation of the functions of certification and disbursement. |
| 23.1.8 | The Contractor acknowledges, and shall incorporate in contracts with Subcontractors, that the GHP is a government-funded program. As such, the administrative costs that are deemed allowable shall be in accordance with cost principles permissible, and with Federal and Puerto Rico applicable guidelines, including Office of Management and Budget Circulars, primarily recognizing that: (1) a cost shall be reasonable if it is of the type generally recognized as ordinary and necessary, and if in its nature and amount, and taking into consideration the purpose for which it was disbursed, it does not exceed that which would be incurred by a prudent person in the ordinary course of business under the circumstances prevailing at the time the decision was made to incur the cost; and (2) a cost shall be reasonable if it is allocable to or related to the cost objective that compels cost association. The Contractor will not allow administrative costs as specified in Section 22.1.15 above. |
Page 256 of 301
| 23.1.9 | The Contractor shall maintain an accounting system for GHP separate from the rest of its commercial activities. This system will only include GHP Data. The Data will be segregated by Service Region. |
| 23.1.10 | The Contractor shall provide, throughout the Contract Term, any other necessary and related information that is deemed necessary by ASES in order to evaluate the Contractor’s financial capacity and stability. |
| 23.2 | Solvency and Financial Requirements |
| 23.2.1 | The Contractor shall establish and maintain adequate net worth, working capital, and financial reserves to carry out its obligations under this Contract. An indemnity agreement containing terms and conditions acceptable to ASES between the Contractor and its parent company shall satisfy the requirements set forth in Sections 23.2.2 and 23.2.3. |
| 23.2.2 | The Contractor shall maintain at all times during the Contract Term a minimum two hundred percent (200) of risk-based capital. ASES reserves the right to require additional capital guarantees as ASES deems reasonably necessary. The Contractor shall comply, as applicable, with Article 3.151 and Article 19.140 of the Puerto Rico Insurance Code relating to insolvency protection. |
| 23.2.3 | The Contractor shall provide assurances to ASES that its provision against the risk of insolvency is adequate, in compliance with the Federal standards set forth in 42 CFR 438.116. In particular, the Contractor shall, according to the timeframe specified in Attachment 12 to this Contract, furnish documentation, certified by a Certified Public Accountant, of: |
| 23.2.3.1 | The relationship between PMPM Payments and capital, with the optimal relationship being 10:1, in order to prove capacity to assume risk; |
| 23.2.3.2 | A debt level of less than seventy-five percent (75%).and |
| 23.2.3.3 | Relationship of current assets to total liabilities shall be, at least, 80% |
| 23.2.4 | As part of its accounting and budgeting function, and in accordance with the Insurance Code of Puerto Rico, the Contractor shall establish an actuarially sound process for estimating and tracking potential liability associated with IBNR Claims. As part of its reserving process the Contractor shall conduct annual reviews to assess its IBNR reserving methodology and make adjustments as necessary. |
Page 257 of 301
| 23.2.5 | The Contractor shall establish a reserve fund for IBNR Claims that under no circumstances may exceed ten percent (10%) of PMPM Payments to PMGs. The reserve shall be reconciled and adjusted every ninety (90) Calendar Days and, if necessary, any excess will be liquidated. Once the PMG has the reserve necessary as determined by the Contractor, the monthly retention may not exceed three percent (3%). Any increase must be justified in information from the PMG file. One hundred and eighty (180) Calendar Days after the end of the Contract Term, the Contractor shall reconcile the IBNR reserve. Any remainder of the IBNR funds shall be returned to the PMGs within sixty (60) Calendar Days from the date that the Contractor conducts the reconciliation. This period may not be extended. |
| 23.2.6 | The Contractor agrees to provide any additional guarantees that ASES may require as a result of the periodical evaluation performed by the Office of the Commissioner of Insurance of the financial health of the Contractor. |
| 23.3 | Reinsurance and Stop Loss |
| 23.3.1 | ASES will not administer a Reinsurance program. |
| 23.3.2 | The Contractor shall have and maintain a minimum of one million dollars ($1,000,000.00) in Reinsurance protection against financial loss due to outlier (catastrophic) cases or maintain self-insurance acceptable to ASES. The Contractor shall submit to ASES such documentation as is necessary to prove the existence of this protection, which may include policies and procedures of Reinsurance. The Contractor may request that ASES remove this requirement by providing sufficient documentation to ASES that the Contractor has adequate protection against financial loss due to outlier (catastrophic) cases. ASES shall review such documentation and, at its discretion, deem this requirement to be met. |
| 23.3.3 | The Contractor shall establish a stop-loss limit of ten thousand dollars ($10,000) per Enrollee per fiscal year for PMGs. Stop-loss coverage shall comply with the limits specified in 42 CFR 422.208(f). The limit shall be activated when the expense of providing Covered Services to an Enrollee, including all outpatient and inpatient expenses, reaches this sum. The Contractor shall have mechanisms in place to identify the stop loss once it is reached for an Enrollee, and shall establish monthly reports to inform PMGs of Enrollees who have reached the stop-loss limit. The Contractor shall assume all losses exceeding the limit. |
| 23.3.4 | The Contractor’s stop-loss responsibility shall not be transferred to a PMG unless the PMG and the Contractor expressly agree in writing to the PMG’s assuming this risk and the associated risk distribution arrangement has been previously approved in writing by ASES. |
Page 258 of 301
| 23.4 | Third Party Liability and Cost Avoidance |
| 23.4.1 | General Provisions |
| 23.4.1.1 | The GHP shall be the payer of last resort for all Covered Services rendered on behalf of Medicaid and CHIP Enrollees in accordance with Federal regulations at 42 CFR 433 Subpart D; ASES will enforce this rule with respect to all GHP Enrollees. |
| 23.4.1.2 | The Contractor shall exercise full assignment rights as applicable and shall be responsible for making every reasonable effort to determine the legal liability of Third Parties to pay for services rendered to Enrollees under this Contract and to cost avoid or recover any such liability from the Third Party. “Third Party,” for purposes of this Section, shall mean any person or entity that is or may be liable to pay for the care and services rendered to a GHP Enrollee. Examples of a Third Party include, but are not limited to, an Enrollee’s health insurer, casualty insurer, a managed care organization, and Medicare. |
| 23.4.1.3 | The Contractor, and by extension its Providers and Subcontractors, hereby agree to utilize for Claims Cost Avoidance purposes, within thirty (30) Calendar Days of learning of such sources, other available public or private sources of payment for services rendered to Enrollees in the Contractor’s Plan. If Third Party Liability (TPL) exists for part or all of the services provided directly by the Contractor to an Enrollee, the Contractor shall make reasonable efforts to recover from TPL sources the value of services rendered. If TPL exists for part or all of the services provided to an Enrollee by a Subcontractor or a Provider, and the Third Party will make payment within a reasonable time, the Contractor may pay the Subcontractor or Provider only the amount, if any, by which the Subcontractor’s or Provider’s allowable Claim exceeds the amount of TPL. |
| 23.4.1.4 | The Contractor shall deny payment on a Claim that has been denied by a Third Party payer when the reason for denial is the Provider’s failure to follow prescribed procedures, including, but not limited to, failure to obtain Prior Authorization, failure to file Claims timely, etc. |
| 23.4.1.5 | The Contractor shall, within five (5) Business Days of issuing a denial of any Claim based on TPL, provide TPL Data to the Provider. |
| 23.4.1.6 | The Contractor shall treat funds recovered from Third Parties as offsets to Claims payments. The Contractor shall report all Cost Avoidance values to ASES in accordance with Federal guidelines and as provided for in this Section. |
Page 259 of 301
| 23.4.1.7 | The Contractor shall post all Third Party payments or recoveries to Claim-level detail by Enrollee. |
| 23.4.1.8 | If the Contractor operates or administers a non-GHP program or other lines of business, the Contractor shall access the resources of those entities to assist ASES with the identification of Enrollees with access to other insurance or sources of payment. |
| 23.4.1.9 | The Contractor shall audit and review its Providers’ Claims, using monthly the reports submitted pursuant to Section 16.7 of this Contract or other pertinent Data, to ensure that Providers are not receiving duplicate payment for services billable to Third Parties. The Contractor shall report to ASES on a quarterly basis its findings regarding Claims, invoices, or duplicate or inappropriate payments. According to the timeframe specified in Attachment 12 to this Contract, the Contractor shall submit to ASES for its review and prior written approval a plan for such routine audits. |
| 23.4.1.10 | The Contractor shall demonstrate, upon request, to ASES that reasonable effort has been made to seek, including through collaboration with Providers, to collect and report Third Party recoveries. ASES shall have the sole responsibility for determining whether or not reasonable efforts have been demonstrated. Said determination shall take into account reasonable industry standards and practices. |
| 23.4.1.11 | The Contractor shall comply with 42 CFR 433 Subpart D – Third Party Liability and 42 CFR 447.20 Provider Restrictions: State Plan Requirements, and work cooperatively with ASES to assure compliance with the requirements therein, as it relates to the Medicaid and CHIP populations served by the Contractor’s plan and its Third Party Liability and Cost Avoidance responsibilities. |
| 23.4.2 | Legal Causes of Action for Damages. ASES or its designee will have the sole and exclusive right to pursue and collect payments made by the Contractor when a legal cause of action for damages is instituted on behalf of a GHP Enrollee against a Third Party, or when ASES receives notices that legal counsel has been retained by or on behalf of any Enrollee. The Contractor shall cooperate with ASES in all collection efforts, and shall also direct its Providers to cooperate with ASES in these efforts. |
| 23.4.3 | Estate Recoveries. ASES (or another agency of the Commonwealth) will have the sole and exclusive right to pursue and recover correctly paid benefits from the estate of a deceased GHP Enrollee who was Medicaid Eligible in accordance with Federal and Puerto Rico law. Such recoveries will be retained by ASES. |
Page 260 of 301
| 23.4.4 | Subrogation |
| 23.4.4.1 | Third Party resources shall include subrogation recoveries. The Contractor shall be required to seek subrogation amounts regardless of the amount believed to be available as required by Federal Medicaid guidelines and Puerto Rico law. |
| 23.4.4.2 | The amount of any subrogation recoveries collected by the Contractor outside of the Claims processing system shall be treated by the Contractor as offsets to medical expenses for the purposes of reporting. |
| 23.4.4.3 | The Contractor shall conduct diagnosis and trauma code editing to identify potential subrogation Claims. This editing should, at minimum, identify Claims with a diagnosis of 900.00 through 999.99 (excluding 994.6) or Claims submitted with an accident trauma indicator of ‘Y.’ |
| 23.4.5 | Cost Avoidance |
| 23.4.5.1 | When the Contractor is aware of health or casualty insurance coverage before paying for a Covered Service, the Contractor shall avoid payment by promptly (within fifteen (15) Business Days of receipt) rejecting the Provider’s Claim and directing that the Claim be submitted first to the appropriate Third Party. |
| 23.4.5.2 | Exceptions to the Cost Avoidance Rule. In the following situations, the Contractor shall first pay its Providers and then coordinate with the liable Third Party, unless prior approval to take other action is obtained from ASES: |
| 23.4.5.2.1 | The coverage is derived from a parent whose obligation to pay support is being enforced by a government agency. |
| 23.4.5.2.2 | The Claim is for maternal and prenatal services to a pregnant woman or for EPSDT services that are covered by the Medicaid program. |
| 23.4.5.2.3 | The Claim is for labor, delivery, and post-partum care and does not involve hospital costs associated with an inpatient stay. |
| 23.4.5.2.4 | The Claim is for a child who is in the custody of ADFAN. |
| 23.4.5.2.5 | The Claim involves coverage or services mentioned in this Section in combination with another service. |
Page 261 of 301
| 23.4.5.3 | If the Contractor knows that the Third Party will neither pay for nor provide the Covered Service, and the service is Medically Necessary, the Contractor shall neither deny payment for the service nor require a written denial from the Third Party. |
| 23.4.5.4 | If the Contractor does not know whether a particular service is covered by the Third Party, and the service is Medically Necessary, the Contractor shall promptly (within ten (10) Business Days of receipt of the Claim) contact the Third Party and determine whether or not such service is covered rather than requiring the Enrollee to do so. Further, the Contractor shall require the Provider to bill the Third Party if coverage is available. |
| 23.4.6 | Sharing of TPL Information by ASES |
| 23.4.6.1 | By the fifth (5th) Calendar Day after the close of the month during which ASES learns of such information, ASES will provide the Contractor with a list of all known health insurance information on Enrollees for the purpose of updating the Contractor’s files. |
| 23.4.6.2 | Additionally, by the fifteenth (15th) Calendar Day after the close of the calendar quarter, ASES will provide to the Contractor a copy of a report containing all of the health insurers licensed by the Commonwealth as of the close of the previous quarter, and any other related information that is needed to file TPL Claims. |
| 23.4.7 | Sharing of TPL Information by the Contractor |
| 23.4.7.1 | The Contractor shall submit a monthly report to ASES (following ASES file content, format and transmission specifications) by the fifth (5th) Calendar Day after the close of the month during which the Contractor learns that an Enrollee has new health insurance coverage, or casualty insurance coverage, or of any change in an Enrollee’s health insurance coverage. The Contractor shall impose a corresponding requirement on its Providers to notify the Contractor of any newly discovered coverage. |
| 23.4.7.2 | When the Contractor becomes aware that an Enrollee has retained counsel, who either may institute or has instituted a legal cause of action for damages against a Third Party, the Contractor shall notify ASES in writing, including the Enrollee’s name and GHP Enrollee identification number, the date of the accident/incident, the nature of the injury, the name and address of the Enrollee’s legal representative, copies of the pleadings, and any other documents related to the action in the Contractor’s possession or control. This shall include, but not be limited to, the name of the Provider, the Enrollee’s diagnosis, the Covered Service provided to the Enrollee, and the amount paid to the Provider for each service. |
Page 262 of 301
| 23.4.7.3 | The Contractor shall notify ASES within thirty (30) Calendar Days of the date it becomes aware of the death of one of its Medicaid Eligible Enrollees age fifty-five (55) or older, giving the Enrollee’s full name, Social Security number, and date of death. ASES will then determine whether it can recover correctly paid Medicaid benefits from the Enrollee’s estate. |
| 23.4.7.4 | The Contractor agrees to share with ASES instances of Enrollee non-cooperation with the Contractor’s and with Network Providers’ efforts to determine sources of Third Party Liability. |
| 23.4.7.5 | The Contractor agrees to cooperate with ASES in its oversight and monitoring reviews of all Third Party Liability activities. |
| 23.4.8 | Historic Cost Avoidance due to the existence of liable Third Parties is embedded in the cost of health services delivery and is reflected in the rates upon which ASES will base PMPM Payments to the Contractor. The PMPM Payment does not include any reductions due to tort recoveries. |
| 23.5 | GHP as Secondary Payer to Medicare |
| 23.5.1 | In general, as provided in Section 7.12, except for services offered by Medicare Platino plans which operate independently of this Contract, the GHP does not duplicate coverage provided by Medicare to Dual Eligible Beneficiaries and the Contractor shall not be a secondary payer for services for which Medicare is liable. |
| 23.5.1.1 | However, in a situation in which a Covered Service is covered in whole or part by both Medicare and GHP (for example, hospitalization services for a Dual Eligible Beneficiary who is enrolled in Medicare Part A only and whose hospitalization costs exceed the Medicare limit, per Section 7.12. of this Contract), the Contractor shall determine liability as a secondary payer as follows: |
| 23.5.1.1.1 | If the total amount of Medicare’s established liability for the services (Medicare paid amount) is equal to or greater than the negotiated contract rate between the Contractor and the Provider for the services, minus any GHP cost-sharing requirements, then the Provider is not entitled to, and the Contractor shall not pay, any additional amounts for the services. |
| 23.5.1.1.2 | If the total amount of Medicare’s established liability (Medicare paid amount) is less than the negotiated contract rate between the Contractor and the Provider for the services, minus any GHP cost-sharing requirements, the Provider is entitled to, and the Contractor shall pay, the lesser of: |
Page 263 of 301
| 23.5.1.1.2.1 | The Medicaid cost-sharing (Deductibles and coinsurance) payment amount for which the Dual Eligible Beneficiary is responsible under Medicare, and |
| 23.5.1.1.2.2 | An amount which represents the difference between (1) the negotiated contract rate between the Contractor and the Provider for the service minus any GHP cost-sharing requirements, and (2) the established Medicare liability for the services. |
| 23.5.2 | Enrollment Exclusions and Contractor Liability for the Cost of Care. Any Dual Eligible Beneficiary who is already enrolled in a Medicare Platino Plan may not be enrolled by the Contractor. However, if the Contractor operates its own Medicare Platino plan, the Contractor may enroll a Dual Eligible Beneficiary in the Platino plan, which furnishes GHP Benefits, per separate contract with ASES. |
| 23.5.3 | Protections for Medicaid Enrollees |
| 23.5.3.1 | The Contractor shall neither impose, nor allow Providers to impose, any cost-sharing charges of any kind upon Medicaid Eligibles enrolled in GHP, other than as authorized in this Contract. |
| 23.5.3.2 | Unless otherwise permitted by Federal or Puerto Rico law, Covered Services may not be denied to a Medicaid Enrollee because of a Third Party’s potential liability to pay for the services, and the Contractor shall ensure that its Cost Avoidance efforts do not prevent Enrollees from receiving Medically Necessary Services. |
| 23.6 | Physician Incentive Plans |
| 23.6.1 | Any Physician Incentive Plans established by the Contractor shall comply with Federal and Puerto Rico regulations, including 42 CFR 422.208 and 422.210, and 42 CFR 438.6(h), and with the requirements in Section 10.7 of this Contract. |
| 23.6.2 | The Contractor shall obtain prior written approval from ASES before implementing any Physician Incentive Plan arrangements, as required in Section 10.7, and shall provide information about such arrangements to Enrollees upon request, as required in Section 6.4.5 of the Contract. Such disclosure shall include: |
Page 264 of 301
| 23.6.2.1 | Whether services not furnished by the Provider or PMG are covered by the incentive plan; |
| 23.6.2.2 | The type of incentive arrangement; |
| 23.6.2.3 | The percentage of Withhold or bonus; |
| 23.6.2.4 | The panel size and if patients are pooled, the method used; and |
| 23.6.2.5 | If the Provider or PMG is at substantial financial risk proof that the Provider or PMG has adequate stop loss coverage, including amount and type of stop loss. |
| 23.6.3 | Annually, the Contractor shall report the information specified by the regulations to ASES in order that ASES can adequately monitor the Contractor’s plan, under the criteria set forth in 42 CFR 422.208 and 422.210. |
| 23.6.4 | Such Physician Incentive Plans may not provide for payment, either directly or indirectly, to a Provider or PMG as an inducement to reduce or limit Medically Necessary Services furnished to an Enrollee. |
| 23.7 | Financial Reporting Requirements |
| 23.7.1 | The Contractor shall submit to ASES all of the reports as indicated in Section 22.1. |
| 23.7.2 | Failure to submit the reports within the established timeframes, or failure to submit complete, accurate reports, may result in the imposition of liquidated damages and/or fines as outlined in Article 20 of this Contract. |
| 23.7.3 | The Contractor, at its sole expense, shall submit by May 15 (or a later date if approved by ASES) of each year a “Report on Controls Placed in Operation and Tests of Operating Effectiveness,” meeting all standards and requirements of the AICPA’s SAS 70, for the Contractor’s operations performed for ASES under the GHP Contract. |
| 23.7.3.1 | The audit shall be conducted by an independent auditing firm, which has prior SAS 70 audit experience. The auditor shall meet all AICPA standards for independence. The selection of, and contract with the independent auditor shall be subject to the prior written approval of ASES. ASES reserves the right to, at the Contractor’s expense, designate other auditors or reviewers to examine the Contractor’s operations and records for monitoring and/or stewardship purposes. |
| 23.7.3.2 | The independent auditing firm shall simultaneously deliver identical reports of its findings and recommendations to the Contractor and ASES within forty-five (45) Calendar Days after the close of each review period. The audit shall be conducted and the report shall be prepared in accordance with generally accepted auditing standards for such audits as defined in the publications of the AICPA, entitled “Statements on Auditing Standards” (SAS). In particular, both the “Statements on Auditing Standards Number 70-Reports on the Processing of Transactions by Service Organizations” and the AICPA Audit Guide, “Audit Guide of Service-Center-Produced Records” are to be used. |
Page 265 of 301
| 23.7.3.3 | The Contractor shall respond to the audit findings and recommendations within thirty (30) Calendar Days of receipt of the final audit report. Also the Contractor must submit a Corrective Action Plan to ASES which will be subject to ASES’ prior review and written approval within twenty (20) Calendars Days of the notification of the audit. The Contractor must implement the Corrective Action Plan, as a maximum, within fifteen (15) Calendar Days of its approval by ASES. The entity should request an extension by formal written request addressed to the Office of Compliance of ASES who will evaluate the request and provide the specific timeframe for the extension. |
| 23.7.4 | The Contractor shall submit to ASES a “Disclosure of Information on Annual Business Transactions.” This report must include: |
| 23.7.4.1 | Definition of A Party in Interest – As defined in Section 1318(b) of the Public Health Service Act, a party in interest is: |
| 23.7.4.1.1 | Any director, officer, partner, or employee responsible for management or administration of the Contractor; any person who is directly or indirectly the beneficial owner of more than five percent (5%) of the equity of the Contractor; any person who is the beneficial owner of a mortgage, deed of trust, note, or other interest secured by, and valuing more than five percent (5%) of the Contractor; or, in the case of a Contractor organized as a nonprofit corporation, an incorporator or enrollee of such corporation under applicable Commonwealth corporation law; |
| 23.7.4.1.2 | Any organization in which a person described in Section 23.7.4.1.1 is director, officer or partner; has directly or indirectly a beneficial interest of more than five percent (5%) of the equity of the Contractor; or has a mortgage, deed of trust, note, or other interest valuing more than five percent (5%) of the assets of the Contractor; |
| 23.7.4.1.3 | Any person directly or indirectly controlling, controlled by, or under common control with the Contractor; or |
Page 266 of 301
| 23.7.4.1.4 | Any spouse, child, or parent of an individual described in Sections 23.7.4.1.1-23.7.4.1.3. |
| 23.7.4.2 | Types of Transactions Which Must Be Disclosed. Business transactions which must be disclosed include: |
| 23.7.4.2.1 | Any sale, exchange or lease of any property between the Contractor and a party in interest; |
| 23.7.4.2.2 | Any lending of money or other extension of credit between the Contractor and a party in interest; and |
| 23.7.4.2.3 | Any furnishing for consideration of goods, services (including management services) or facilities between the Contractor and the party in interest. This does not include salaries paid to employees for services provided in the normal course of their employment. |
| 23.7.4.3 | The information which must be disclosed in the transactions listed in this Section 23.7.4 between the Contractor and a party of interest includes: |
| 23.7.4.3.1 | The name of the party in interest for each transaction; |
| 23.7.4.3.2 | A description of each transaction and the quantity or units involved; |
| 23.7.4.3.3 | The accrued dollar value of each transaction during the fiscal year; and |
| 23.7.4.3.4 | Justification of the reasonableness of each transaction. |
| 23.7.4.4 | As per 42 CFR § 455.105 the Contractor, within thirty-five (35) Calendar Days of the date of request by the HHS Secretary or the Commonwealth Medicaid agency, shall report full and complete information about: |
| 23.7.4.4.1 | The ownership of any subcontractor with whom the provider has had business transactions totaling more than $25,000 during the 12-month period ending on the day of the request; and |
| 23.7.4.4.2 | Any significant business transactions between the provider and any wholly owned supplier, or between the provider and any subcontractor, during the five (5)-year period ending on the date of the request. |
Page 267 of 301
ARTICLE 24 PAYMENT OF TAXES
| 24.1 | The Contractor certifies and guarantees that at the time of execution of this Contract: |
| 24.1.1 | It is an entity duly authorized to conduct business in Puerto Rico and has filed income tax returns for the previous five (5) years; |
| 24.1.2 | It complied with and paid unemployment insurance tax, disability insurance tax (Law 139), social security for drivers (“seguro social choferil”), if applicable; |
| 24.1.3 | It filed State Department reports for the five (5) previous years; and |
| 24.1.4 | It does not owe any kind of taxes to the Commonwealth. |
| 24.2 | The Contractor will forthwith pay all taxes lawfully imposed upon it with respect to this Contract or any product delivered in accordance herewith. ASES makes no representation whatsoever as to the liability or exemption from liability of Contractor to any tax imposed by any governmental entity. |
| 24.3 | Notwithstanding the above, if, as a result of the enactment of any state, local or municipal legal provision, administrative regulation, or government directive, the Contractor is burdened with a requirement to pay a fee, tax, imposition, levy, or duty with regards to any of the proceeds of this Contract, including but not limited to the imposition of any fees pertaining to the existence of any government contracts, or any added value tax (IVU, for its Spanish acronym), ASES will evaluate, in good faith, an adjustment to the PMPM Payment under this Contract. |
ARTICLE 25 RELATIONSHIP OF PARTIES
| 25.1 | Neither Party is an Agent, employee, or servant of the other. It is expressly agreed that the Contractor and any Subcontractors and Agents, officers, and employees of the Contractor or any Subcontractor in the performance of this Contract shall act as independent contractors and not as officers or employees of ASES. The Parties acknowledge, and agree, that the Contractor, its Agent, employees, and servants shall in no way hold themselves out as Agent, employees, or servants of ASES. It is further expressly agreed that this Contract shall not be construed as a partnership or joint venture between the Contractor or any Subcontractor and ASES. |
ARTICLE 26 INSPECTION OF WORK
| 26.1 | ASES, the Puerto Rico Medicaid Program, other agencies of the Commonwealth, the US Department of Health and Human Services, the General Accounting Office, the US Comptroller General, the Comptroller General of the Commonwealth, if applicable, or their Authorized Representatives, shall have the right to enter into the premises of the Contractor or all Subcontractors, or such other places where duties under this Contract are being performed for ASES, to inspect, monitor or otherwise evaluate the services or any work performed pursuant to this Contract. All inspections and evaluations of work being performed shall be conducted with prior notice and during normal business hours. All inspections and evaluations shall be performed in such a manner that will not unduly delay work. |
Page 268 of 301
ARTICLE 27 GOVERNMENT PROPERTY
| 27.1 | The Contractor agrees that any papers, materials and other documents that are produced or that result, directly or indirectly, from, under or in connection with the Contractor’s provision of the services under this Contract shall be the property of ASES upon creation of such documents, for whatever use that ASES deems appropriate, and the Contractor further agrees to prepare any and all documents, including the Deliverables listed in Attachment 12 to this Contract, or to take any additional actions that may be necessary in the future to effectuate this provision fully. In particular, if the work product or services include the taking of photographs or videotapes of individuals, the Contractor shall obtain the consent from such individuals authorizing the use by ASES of such photographs, videotapes, and names in conjunction with such use. The Contractor shall also obtain necessary releases from such individuals, releasing ASES from any and all claims or demands arising from such use. |
| 27.2 | The Contractor shall be responsible for the proper custody and care of any ASES-owned property furnished for the Contractor’s use in connection with the performance of this Contract. The Contractor will reimburse ASES for its loss or damage, normal wear and tear excepted, while such property is in the Contractor’s custody or use. |
ARTICLE 28 OWNERSHIP AND USE OF DATA AND SOFTWARE
| 28.1 | Ownership and Use of Data |
| 28.1.1 | All Information created from Data, documents, messages (verbal or electronic), reports, or meetings involving or arising out of or in connection with this Contract is owned by ASES (the information will be hereinafter referred to as “ASES Data and Information”). The Contractor shall make all Data and Information available to ASES, which will also provide the Data to CMS or other pertinent government agencies and authorities upon request. The Contractor is expressly prohibited from sharing, distributing, disseminating, or publishing ASES Data and Information without the express prior written consent of ASES. In the event of a dispute regarding what is or is not ASES Data and Information, ASES’s decision on this matter shall be final and not subject to appeal. |
| 28.1.2 | ASES acknowledges that before executing this Contract and in contemplation of the same, the Contractor has developed and designed certain programs and systems such as standard operating procedures, programs, business plans, policies and procedures, which ASES acknowledges are the exclusive property of the Contractor. Nevertheless, in case of default by the Contractor, ASES is hereby authorized to use to the extent allowable by any applicable commercial software and hardware licensing that exists at that moment or with which agreement can be reached at that moment with the vendor to modify such licensing to permit its use by ASES, at no cost to ASES, such properties for a period of one hundred and twenty (120) Calendar Days to effect an orderly transition to any new Contractor or service provider. In any cases where the use of such systems from an operational perspective would also impact other lines of the Contractor’s business or where licensing restrictions cannot be remedied, the Contractor shall operate such systems on behalf of ASES. Such operation by the Contractor on behalf of ASES can occur at ASES’ discretion under the full supervision of their employees or appointed third party personnel. Under such a scenario, ASES’ access to Data will be restricted through the most efficient means possible to the Contractor’s Data segment. If the Contractor fails to operate such systems on ASES’ behalf in a timely manner per normal previous operating schedule, ASES may claim ownership of such systems and operate them for its own purposes. |
Page 269 of 301
| 28.1.3 | The Contractor shall not deny access to ASES’s Data under any case or circumstances, nor retain ASES’s Data while controversies between ASES and the Contractor are resolved and finally adjudicated |
| 28.2 | Responsibility for Information Technology Investments |
The Parties understand and agree that the cost of any newly acquired or developed software programs or upgrades or enhancements to existing software programs, hardware, or other related information technology equipment or infrastructure component, made in order to comply with the requirements of this Contract shall be borne in its entirety by the Contractor.
ARTICLE 29 CRIMINAL BACKGROUND CHECKS
| 29.1 | ASES is prohibited by law from entering into contracts with any person or entity that has been, or whose affiliated subsidiary companies, or any of its shareholders, partners, officers, principals, managing employees, subsidiaries, parent companies, officers, directors, board members, or ruling bodies have been, under investigation for, accused of, convicted of, or sentenced to imprisonment, in Puerto Rico, the US, or any other jurisdiction, for any crime involving corruption, fraud, embezzlement, or unlawful appropriation of public funds, pursuant to Act 458, as amended, and Act 84 of 2002. |
| 29.2 | Before the Effective Date of this Contract, and in order for the Contract to take effect, the Contractor shall provide to ASES a certification that neither the Contractor nor the affiliated persons/entities listed in Section 29.1 falls under the prohibition stated in Section 29.1. In addition, the Contractor shall provide to ASES a certification as to whether, to the best of its knowledge after inquiry, any Network Provider, or any shareholder, partner, officer, principal, managing employee, subsidiary, parent company, officer, director, board member, or ruling body of a Network Provider, falls under the prohibition stated in [Section 29.1]. |
Page 270 of 301
| 29.3 | ASES may terminate this Contract if ASES determines that the Contractor, or any of the natural persons listed in Section 29.1, falls within the prohibition stated in Section 29.1, or failed to provide an accurate certification as required in Section 29.2. In addition, the Contractor shall terminate a Provider Contract if it determines that a Provider, or any of the natural persons listed in Section 29.1, falls within the prohibition stated in Section 29.1. |
| 29.4 | During the Contract Term, the Contractor shall promptly (within twenty (20) Business Days of the date it receives the information) report any significant fact or event related to the rule stated in this Article. |
| 29.5 | In cases in which none of the events listed in Section 29.1 has occurred, but statements or admissions of crimes have been made by or against the Contractor or one of its shareholders, partners, officers, principals, subsidiaries, or parent companies, ASES shall provide all pertinent information about the matter, within twenty (20) Business Days from the date it receives the information, to the Secretary of Justice of Puerto Rico, who will make the pertinent findings and recommendations concerning the Contract. |
| 29.6 | In addition, as provided in 42 CFR 455.106(c), ASES may refuse to enter into or renew an agreement with any entity if any person who has an ownership or control interest in the entity, or is an Agent or managing employee of the entity, has ever been convicted of a criminal offense related to the person’s involvement in any program established under Medicare, Medicaid, or the Title XX services programs. Before the Effective Date of this Contract, pursuant to 42 CFR 455.106(a), the Contractor shall disclose to ASES the identity of any person who has ever been convicted of a criminal offense related to the Medicare, Medicaid, or Title XX services programs. The Contractor shall collect the same information on criminal conviction for Providers during the Credentialing process, as provided in Section 9.2.3 and shall, immediately upon receipt of such information relating to a Provider, disclose the information to ASES. ASES will notify the HHS Inspector General of any disclosures related to criminal convictions within twenty (20) Business Days from the date that ASES receives the information, as required by 42 CFR 455.106. |
ARTICLE 30 SUBCONTRACTS
| 30.1 | Use of Subcontractors |
| 30.1.1 | In carrying out the terms of this Contract, the Contractor, with the prior written approval of ASES, may enter into written Subcontract(s) with other entities for the provision of administrative services or a combination of Covered Services and administrative services, under terms and conditions acceptable to ASES in its sole discretion. |
| 30.1.2 | The Contractor shall assume sole responsibility for all functions performed by a Subcontractor(s), as well as any payments to a Subcontractor(s) for services related to this Contract. In the event that a Subcontractor is incapable of performing the service contracted for by the Contractor, the Contractor shall (i) notify ASES within two (2) Business Days and (ii) assume responsibility for providing the services that the Subcontractor is incapable of performing. The Contractor shall remain obligated to provide any services that the Subcontractor is incapable of performing. |
Page 271 of 301
| 30.1.3 | If the Contractor becomes aware of a Subcontractor’s failure to comply with this Contract, the Contractor shall correct the failure within thirty (30) Calendar Days of becoming aware of the failure. |
| 30.1.4 | All contracts between the Contractor and Subcontractors must be in writing and must specify the activities and responsibilities delegated to the Subcontractor containing terms and conditions consistent with this Contract. The contracts must also include provisions for revoking delegation or imposing other sanctions if the Subcontractor’s performance is inadequate. The Contractor and the Subcontractors must also make reference to a business associates agreement between the Parties. |
| 30.1.5 | All contracts between the Contractor and Subcontractors must ensure that the Contractor evaluates the prospective Subcontractor’s ability to perform the activities to be delegated; monitors the Subcontractor’s performance on an ongoing basis and subjects it to formal review according to a periodic schedule established by ASES and consistent with industry standards or Puerto Rico laws and regulations; and identifies deficiencies or areas for improvement, ensuring that corrective action is taken as appropriate or required. |
| 30.1.6 | The Contractor shall not subcontract or permit anyone other than Contractor personnel to perform any of the work, services, or other performances required of the Contractor under this Contract relating to functions associated with the provision of Benefits to Enrollees or assign any of its rights or obligations hereunder, without the prior written consent of ASES. Prior to hiring or entering into an agreement with any Subcontractor, any and all Subcontractors shall be previously approved in writing by ASES. ASES reserves the right to review all Subcontract agreements at any time during the Contract Term. Upon request from ASES, the Contractor shall provide in writing the names of all proposed or actual Subcontractors. |
| 30.1.7 | The Contractor shall not engage nor contract with a person or entity that is debarred or suspended or otherwise excluded from participating in procurement activities under the Federal Acquisition Regulation (FAR) or from participating in non-procurement activities under regulations issued under Executive Order No. 12549 or under guidelines implementing Executive Order No. 12549, or a person or entity that is an Affiliate, as defined in FAR, of a such a person or entity (see 42 CFR 438.610). |
Page 272 of 301
| 30.1.8 | ASES shall have the right to review all financial or business transactions between the Contractor and a Subcontractor upon request. ASES shall also retain the right to review all criminal background checks for all employees of the Subcontractor, as referenced in Article 29, as well as any past exclusions from Federal programs. |
| 30.1.9 | The Contractor shall provide ASES Immediate notice by certified mail, of any action or suit filed and of any claim made against the Contractor by the Subcontractor or against a Subcontractor(s) that, in the opinion of the Contractor, may result in litigation related in any way to this Contract. The Contractor shall provide notification in writing as to how this action or suit may affect the overall provision of services to Enrollees and the Contractor’s plan to mitigate such affect. |
| 30.1.10 | When a Subcontract related to the provision of Covered Services or that includes Claims processing services is being terminated other than for cause, the Contractor shall give at least one hundred twenty (120) Calendar Days prior written notice of the termination to ASES. If the termination is for cause, the Contractor shall Immediately notify ASES. |
| 30.1.11 | The Contractor shall give ASES Immediate notice in writing by registered mail or certified mail of any action or suit filed by any Subcontractor and prompt notice of any claim made against the Contractor by any Subcontractor or vendor that, in the opinion of Contractor, may result in litigation related in any way to this Contract. |
| 30.1.12 | All Subcontractors must fulfill the requirements of 42 CFR 438.6 as appropriate. |
| 30.1.13 | All Provider Contracts shall be in compliance with the requirements and provisions as set forth in Section 10.3 of this Contract. |
| 30.2 | Cost or Pricing by Subcontractors |
| 30.2.1 | The Contractor shall submit, and shall require any Subcontractors hereunder to submit, cost or pricing Data for any subcontract to this Contract prior to award. The Contractor shall also certify that the information submitted by the Subcontractor is, to the best of the Contractor’s knowledge and belief, accurate, complete and current as of the date of agreement, or the date of the negotiated price of the Subcontract or amendment to the Contract. The Contractor shall insert the substance of this Section in each Subcontract hereunder. |
| 30.2.2 | If ASES determines that any price, including profit or fee negotiated in connection with this Contract, or any cost reimbursable under this Contract was increased by any significant sum because of the inaccurate cost or pricing Data, then such price and cost shall be reduced accordingly and this Contract and the Subcontract shall be modified in writing to reflect such reduction. |
Page 273 of 301
ARTICLE 31 REQUIREMENT OF INSURANCE LICENSE
| 31.1 | In order for this Contract to take effect, the Contractor must be licensed to underwrite health insurance by the Puerto Rico Insurance Commissioner. The Contractor must submit a copy of its insurance license according to the timeframe specified in Attachment 12 to this Contract. |
| 31.2 | The Contractor shall renew the license as required, and shall submit evidence of the renewal to ASES within thirty (30) Calendar Days of the expiration date of the license. |
ARTICLE 32 CERTIFICATIONS
| 32.1 | The Contractor shall provide to ASES within fifteen (15) Calendar Days of the Effective Date of this Contract, and thereafter by January 10 of each calendar year during the Contract Term, the certifications and other documents set forth below, according to the timeframe specified below. If any certification, document, acknowledgment, or other representation or assurance on the Contractor’s part under this Article, or elsewhere in this Contract, is determined to be false or misleading, ASES shall have cause for termination of this Contract. In the event that the Contract is terminated based upon this Article, the Contractor shall reimburse ASES all sums of monies received under the Contract; provided, however, that the amount reimbursed shall not exceed the amount of outstanding debt, less any payments made by the Contractor in satisfaction of such debt. |
| 32.2 | The Contractor shall submit the following certifications: |
| 32.2.1 | Certification issued by the Treasury Department of Puerto Rico (Model SC-2888) with evidence that that the Contractor has filed income tax returns in the past five (5) years or has non-profit status; |
| 32.2.2 | Certification from the Treasury Department of Puerto Rico that Contractor has no outstanding debt with the Department or, if such a debt exists, it is subject to a payment plan or pending administrative review under applicable law or regulation (Model SC-3537); |
| 32.2.3 | Certification from the Center for the Collection of Municipal Revenues certifying that there is no outstanding debt or, if a debt exists, that such debt is subject to payment plan or pending administrative review under applicable law or regulations; |
| 32.2.4 | Certification from the Department of Labor and Human Resources certifying compliance with unemployment insurance, temporary disability insurance and/or chauffeur’s social security, if applicable; |
| 32.2.5 | Evidence of Incorporation and of Good Standing issued by the Department of State of Puerto Rico; |
Page 274 of 301
| 32.2.6 | Certification of current municipal license tax (“Patentes Municipales”), if applicable; |
| 32.2.7 | Certification issued by the Minor Children Support Administration (“ASUME”, by its Spanish acronym) of no outstanding alimony or child support debts, if applicable; |
| 32.2.8 | A sworn statement certifying that it has no debt with the government of the Commonwealth, or with any state agencies, corporations or instrumentalities that provide or are related to the provision of health services; and |
| 32.2.9 | Certification from the Puerto Rico Administration of Medical Services (“ASEM”, its Spanish acronym) certifying that there is no outstanding debt or, if a debt exists, that such debt is subject to a payment plan or pending administrative review under applicable law or regulations. |
| 32.3 | The Contractor shall, in addition, provide the following documents: |
| 32.3.1 | A list of all contracts the Contractor has with government agencies, public corporations or municipalities, including those contracts in the process of being executed; |
| 32.3.2 | A letter indicating if any of its directors serves as member of any governmental board of directors or commission; |
| 32.3.3 | A certificate of the Corporate Resolution authorizing the person signing this Contract to appear on behalf of the Contractor; |
| 32.3.4 | Evidence of compliance with the Compensation System for Work-Related Accidents Act (“Fondo del Seguro del Estado de Puerto Rico”); and |
| 32.3.5 | A copy of the Insurance Coverage Certificate as required in Article 37. |
| 32.4 | If the Contractor fails to meet the obligations of Sections 32.2 and 32.3 within the required timeframe, ASES shall cease payment to the Contractor until the documents have been delivered to the ASES’s satisfaction, or adequate evidence is provided to ASES that reasonable efforts have been made to obtain the documents. |
ARTICLE 33 RECORDS REQUIREMENTS
| 33.1 | General Provisions |
| 33.1.1 | The Contractor and its Subcontractors, if any, shall preserve and make available all of its records pertaining to the performance under this Contract for inspection or audit, as provided below, throughout the Contract Term, for a period of seven (7) years from the date of final payment under this Contract, and for such period, if any, as is required by applicable statute or by any other section of this Contract. If the Contract is completely or partially terminated, the records relating to the work terminated shall be preserved and made available for period of seven (7) years from the Termination Date of the Contract or of any resulting final settlement. The Contractor is responsible to preserve all records pertaining to its performance under this Contract, and to have them available and accessible in a timely manner, and in a reasonable format that assures their integrity. Records that relate to Appeals, litigation, or the settlements of Claims arising out of the performance of this Contract, or costs and expenses of any such agreements as to which exception has been taken by the Contractor or any of its duly Authorized Representatives, shall be retained by Contractor until such Appeals, litigation, Claims or exceptions have been disposed of. |
Page 275 of 301
| 33.2 | Records Retention and Audit Requirements |
| 33.2.1 | Since funds from the Puerto Rico Plans under Title XIX and Title XXI of the Social Security Act Medical Assistance Programs (Medicaid and CHIP) are used to finance this project in part, the Contractor shall agree to comply with the requirements and conditions of the Centers for Medicare and Medicaid Services (CMS), the US Comptroller General, the Comptroller of Puerto Rico and ASES, as to the maintenance of records related to this Contract. |
| 33.2.2 | Puerto Rico and Federal standards for audits of ASES Agents, contractors, and programs are applicable to this section and are incorporated by reference into this Contract as though fully set out herein. |
| 33.2.3 | Pursuant to the requirements of 42 CFR 434.6(a)(5) and 42 CFR 434.38, the Contractor shall make all of its books, documents, papers, Provider records, Medical Records, financial records, Data, surveys and computer databases available for examination and audit by ASES, the Department of Health and Human Services and its sub-agencies, the Comptroller of Puerto Rico, the US Comptroller General and/or their Authorized Representatives. Any records requested hereunder shall be produced Immediately for on-site review or sent to the requesting authority by mail within fourteen (14) Calendar Days following a request. All records shall be provided at the sole cost and expense of the Contractor. ASES shall have unlimited rights to use, disclose, and duplicate all Information and Data in any way relating to this Contract in accordance with applicable Puerto Rico and Federal laws and regulations. |
| 33.2.4 | In certain circumstances, as follows, the authorities listed in Section 33.2.3 shall have the right to inspect and audit records in a timeframe that exceeds the timeframe set forth in Section 33.1.1. |
| 33.2.4.1 | ASES determines that there is a special need to retain a particular record or group of records for a longer period and notifies the Contractor at least thirty (30) Calendar Days before the expiration of the timeframe set forth in Section 33.1.1. |
Page 276 of 301
| 33.2.4.2 | There has been a Contract termination, dispute, fraud, or similar fault by the Contractor, resulting in a final judgment or settlement against the Contractor, in which case the retention may be extended to three (3) years from the date of the final judgment or settlement. |
| 33.2.4.3 | ASES determines that there is a reasonable possibility of Fraud, and gives the Contractor notice, before the expiration of the timeframe set forth in Section 33.1.1, that it wishes to extend the time period for retention of records. |
| 33.2.4.4 | There has been, during the time period set forth in Section 33.1.1, an audit initiated by CMS, the Comptroller of Puerto Rico, the US Comptroller General, and/or ASES, in which case the timeframe for retention of records shall extend until the conclusion of the audit and publication of the final report. |
| 33.2.5 | All records retention requirements set forth in this Article or in any other Article shall be subject at all times and to the extent mandated by law and regulation, to the HIPAA regulations described elsewhere in this Contract. |
| 33.3 | Medical Record Requests |
| 33.3.1 | The Contractor shall ensure that a copy of each Enrollee’s Medical Record is made available, without charge, upon the written request of the Enrollee or Authorized Representative within fourteen (14) Calendar Days of the receipt of the written request. |
| 33.3.2 | The Contractor shall ensure that Medical Records are furnished at no cost to a Provider, upon the Enrollee’s request, no later than fourteen (14) Calendar Days following the written request. |
ARTICLE 34 CONFIDENTIALITY
| 34.1 | General Confidentiality Requirements |
| 34.1.1 | The Contractor shall protect all information, records, and Data collected in connection with the Contract from unauthorized disclosures. In addition, the Contractor shall agree to guard the confidentiality of Enrollee information. Access to all individually identifiable information relating to Medicaid Enrollees that is obtained by the Contractor shall be limited by the Contractor to Subcontractors, consultants, advisors or agencies that require the information in order to perform their duties in accordance with this Contract, and to such others as may be authorized by ASES in accordance with applicable law. |
Page 277 of 301
| 34.1.2 | The Contractor is responsible for understanding the degree to which information obtained through the performance of this Contract is confidential under Puerto Rico and Federal law, rules, and regulations. |
| 34.1.3 | Any other party shall be granted access to confidential Information only after complying with the requirements of Puerto Rico and Federal law pertaining to such access. ASES shall have absolute authority to determine if and when any other party has properly obtained the right to have access to this confidential information. Nothing herein shall prohibit the disclosure of information in summary, statistical, or other form that does not identify particular individuals. The Contractor shall retain the right to use information for its quality and Utilization Management and research purposes subject to the Data ownership and publicity requirements defined within the Contract. |
| 34.1.4 | The Contractor, its employees, Agents, Subcontractors, consultants or advisors must treat all information that is obtained through Providers’ performance of the services under this Contract, including, but not limited to, information relating to Enrollees, Potential Enrollees, as confidential Information to the extent that confidential treatment is provided under Puerto Rico and Federal law, rules, and regulations. |
| 34.1.5 | Any disclosure or transfer of confidential information by the Contractor, including information required by ASES, will be in accordance with applicable law. If the Contractor receives a request for information deemed confidential under this Contract, the Contractor will Immediately notify ASES of such request, and will make reasonable efforts to protect the information from public disclosure. |
| 34.1.6 | In accordance with the timeframes outlined in Attachment 12, the Contractor shall develop and provide to ASES for review and approval written policies and procedures for the protection of all records and all other documents deemed confidential under this Contract including Medical Records/Enrollee information and adolescent/sexually transmitted disease appointment records. All Enrollee information, Medical Records, Data and Data elements collected, maintained, or used in the administration of this Contract shall be protected by the Contractor from unauthorized disclosure per the HIPAA Privacy and Security standards codified at 45 CFR Part 160 and 45 CFR Part 164, Subparts A, C and E. The Contractor must provide safeguards that restrict the use or disclosure of protected health information (PHI) concerning Enrollees to purposes directly connected with the administration of this Contract. |
| 34.1.7 | The Contractor must comply with HIPAA notification requirements, including those set forth in HITECH. The Contractor must notify ASES of all Breaches or potential Breaches of unspecified PHI, as defined by HITECH, without unreasonable delay and in no event later than thirty (30) Calendar Days after discovery of the Breach or potential Breach. If, in ASES’s determination, the Contractor has not provided notice in the manner or format prescribed by HITECH, then ASES may require the Contractor to provide such notice. |
Page 278 of 301
| 34.1.8 | Assurance of Confidentiality |
| 34.1.8.1 | The Contractor shall take reasonable steps to ensure the physical security of Data under its control, including, but not limited to: fire protection; protection against smoke and water damage; alarm systems; locked files, guards, or other devices reasonably expected to prevent loss or unauthorized removal of manually held Data; passwords, access logs, badges, or other methods reasonably expected to prevent loss or unauthorized access to electronically or mechanically held Data; limited terminal access; limited access to input documents and output documents; and design provisions to limit use of Enrollee names. |
| 34.1.8.2 | The Contractor shall inform and provide quarterly trainings to each of its employees having any involvement with personal Data or other confidential information, whether with regard to design, development, operation, or maintenance, of the Puerto Rico and Federal law relating to confidentiality. |
| 34.1.9 | Return of Confidential Data |
| 34.1.9.1 | The Contractor shall return all Personal Health Information Data furnished pursuant to this Contract promptly at the request of ASES in whatever form it is maintained by the Contractor. Upon the termination or completion of the Contract, the Contractor may not use any such Data or any material derived from the Data for any purpose not permitted by Puerto Rico or Federal law or regulation and where so instructed by ASES shall destroy such Data or material if permitted and required by Puerto Rico or Federal law or regulation. |
| 34.1.10 | Publicizing Safeguarding Requirements |
| 34.1.10.1 | The Contractor shall comply with 42 CFR 431.304. The Contractor agrees to publicize provisions governing the confidential nature of information about Enrollees, including the legal sanctions imposed for improper disclosure and use. The Contractor must include these provisions in the Enrollee handbook and provide copies of these provisions to Enrollees and to other persons and agencies to which information is disclosed. |
| 34.1.10.2 | In addition to the requirements expressly stated in this Article, the Contractor must comply with any policy, rule, or reasonable requirement of ASES that relates to the safeguarding or disclosure of information relating to Enrollees, the Contractor’s operations, or the Contractor’s performance of this Contract. |
Page 279 of 301
| 34.1.10.3 | In the event of the expiration of this Contract or termination thereof for any reason, all confidential information disclosed to and all copies thereof made by the Contractor must be returned to ASES or, at ASES’s option, erased or destroyed. The Contractor must provide ASES certificates evidencing such destruction. |
| 34.1.10.4 | The Contractor’s contracts with practitioners and other Providers shall explicitly state expectations about the confidentiality of ASES’s confidential information and Enrollee records. |
| 34.1.10.5 | The Contractor shall afford Enrollees and/or their Authorized Representatives the opportunity to approve or deny the release of identifiable personal information by the Contractor to a person or entity outside of the Contractor, except to duly authorized Subcontractors, Providers or review organizations, or when such release is required by law, regulation, or quality standards. |
| 34.1.10.6 | This Article 34 does not restrict the Contractor from making any disclosure pursuant to any applicable law, or under any court or government agency, provided that the Contractor provides immediate notice to ASES of such order. |
| 34.1.11 | Disclosure of ASES’s Confidential Information |
| 34.1.11.1 | The Contractor shall Immediately report to ASES any and all unauthorized disclosures or uses of confidential information of which it or its Subcontractors, consultants, or Agents is aware or has knowledge. The Contractor acknowledges that any publication or disclosure of confidential information to others may cause immediate and irreparable harm to ASES and may constitute a violation of Puerto Rico or Federal statutes. If the Contractor, its Subcontractors, consultants, or Agents should publish or disclose Confidential Information to others without authorization, ASES will immediately be entitled to injunctive relief or any other remedies to which it is entitled under law or equity. ASES will have the right to recover from the Contractor all damages and liabilities caused by or arising from the Contractor’s, its Subcontractors’, Network Providers’, representatives’, consultants’, or Agents’ failure to protect confidential Information. The Contractor will defend with counsel approved by ASES, indemnify and hold harmless ASES from all damages, costs, liabilities, and expenses caused by or arising from the Contractor’s, or its Subcontractors’, Providers’, representatives’, consultants’ or Agents’ failure to protect confidential Information. ASES will not unreasonably withhold approval of counsel selected by the Contractor. |
Page 280 of 301
| 34.1.12 | The Contractor shall remove any person from performance of services hereunder upon notice that ASES reasonably believes that such person has failed to comply with the confidentiality obligations of this Contract. The Contractor shall replace such removed personnel in accordance with the staffing requirements of this Contract. |
| 34.1.13 | ASES, the Commonwealth, Federal officials as authorized by Federal law or regulations, or the Authorized Representatives of these Parties shall have access to all confidential information in accordance with the requirements of Puerto Rico and Federal laws and regulations. |
| 34.1.14 | The confidentiality provisions contained in this Contract survive the termination of this contract and shall bind the Contractor, and its PMGs and Network Providers, so long as they maintain any “protected health information” relating to Enrollees, as such term is defined by 45 CFR Parts 160 and 164. |
| 34.2 | HIPAA Compliance |
| 34.2.1 | The Contractor shall assist ASES in its efforts to comply with the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”) and its amendments, rules, procedures, and regulations. To that end, the Contractor shall cooperate with and abide by any requirements mandated by HIPAA or any other applicable laws. The Contractor acknowledges that HIPAA requires the Contractor and ASES to sign documents for compliance purposes, including but not limited to a business associate agreement. A standard business associate agreement is included as Attachment 18 to this Contract. The Contractor shall cooperate with ASES on these matters and sign whatever documents may be required for HIPAA compliance and abide by their terms and conditions. |
| 34.3 | Privacy of Information in Enrollment Database |
| 34.3.1 | Any individually identifiable health information held in the Enrollment Database described in Section 5.3.8 of this Contract shall be kept confidential and shall be used and disclosed by the Contractor or its Network Providers only for purposes directly connected with performance of all obligations contained in this Contract, and in strict compliance with HIPAA’s privacy and security requirements and any applicable laws of Puerto Rico. |
| 34.4 | Data Breach |
| 34.4.1 | The Contractor shall report to ASES, as required in § 13402 of the HITECH Act, of any event where ASES’s Data could be exposed in a non-authorized or illegal circumstance, and/or when any Data Breach occurs. The Contractor must take all reasonable steps to mitigate the Breach. |
Page 281 of 301
| 34.4.2 | The Contractor agrees that without unreasonable delay, but no later than twenty-four (24) hours after it suspects or has determined that a Data Breach occurred, the Contractor shall notify ASES of such Breach. The notification shall include sufficient information for ASES to understand the nature of the Breach. For instance, such notification must include, to the extent available at the time of the notification, the following information: |
| 34.4.2.1 | One or two sentence description of the event; |
| 34.4.2.2 | Description of the roles of the people involved in the Breach (e.g., employees, participant users, service Providers, unauthorized persons, etc.) |
| 34.4.2.3 | The type of Data / Information as well as Personal Health Information that was breached; |
| 34.4.2.4 | Enrollees likely impacted by the Breach; |
| 34.4.2.5 | Number of individuals or records impacted/estimated to be impacted by the Breach; |
| 34.4.2.6 | Actions taken by the Contractor to mitigate the Breach; |
| 34.4.2.7 | Current status of the Breach (under investigation or resolved); |
| 34.4.2.8 | Corrective action taken and steps planned to be taken to prevent a similar Breach. |
| 34.4.3 | The Contractor shall have a duty to supplement the information contained in the notification as it becomes available and to cooperate with ASES. The notification required by this Section shall not include any PHI. |
ARTICLE 35 TERMINATION OF CONTRACT
| 35.1 | General Procedures |
| 35.1.1 | In addition to any other non-financial remedy set forth in this Contract or available by law, or in lieu of any financial Remedy contained in Articles 19 and 20 of this Contract or available by law, and subject to compliance with the termination procedures set forth in Section 35.8 below, ASES may terminate this Contract for any or all of the following reasons: |
| 35.1.1.1 | Default by the Contractor, upon thirty (30) Calendar Days’ notice, unless ASES, in its reasonable discretion, determines that the Contractor has cured the default to ASES’s satisfaction within the notice period; |
Page 282 of 301
| 35.1.1.2 | Immediately, in the event of insolvency or declaration of bankruptcy by the Contractor; |
| 35.1.1.3 | Immediately, when sufficient appropriated funds no longer exist for the payment of ASES's obligation under this Contract; or |
| 35.1.1.4 | In the event that the Contractor or any of its shareholders, director, officers, or employees fall under the prohibition stated in Section 29.1 or 29.6 of this Contract. |
| 35.1.2 | A decision by ASES not to renew this Contract, per Article 21, shall not be considered a Termination of the Contract. |
| 35.1.3 | The Contractor shall have a limited right of termination of this Contract only in the events described in Section 35.10 of this Contract. |
| 35.1.4 | Each Party shall have the opportunity to cure any default alleged in a termination notice sent pursuant to this Article 35, upon receiving a written termination notice the other Party. With respect to termination by ASES, the Contractor shall have the right to submit to ASES a written Corrective Action Plan containing terms and conditions acceptable to ASES in its sole discretion to cure such default or an explanation of non-default in the thirty (30) Calendar Day period from the date of receipt of ASES’ written termination notice and such plan or explanation of non-default is accepted by ASES, in ASES’ sole discretion, which acceptance shall not be unreasonably withheld, conditioned or delayed. . |
| 35.1.5 | Notwithstanding the termination of this Contract pursuant to this Article 35 for any reason, the Contractor shall remain obligated to provide the Administrative Functions as described in Article 36, including but not limited to the payment of Claims for Covered Services provided to Enrollees prior to the Termination Date and as specified in the Patient’s Bill of Rights Act through the Runoff Period. |
| 35.1.6 | Continuing Obligations of ASES. Notwithstanding the termination of this Contract for pursuant to this Article 35 for any reason, ASES shall remain obligated to pay to the Contractor the PMPM through the Termination Date (inclusive of the Transition Period). |
| 35.1.7 | Termination Procedures to be Strictly Followed. No termination of this Contract shall be effective unless the termination procedures under Section 35 of this Contract have been strictly followed or waived by the Parties. |
Page 283 of 301
| 35.2 | Termination by Default |
| 35.2.1 | In the event ASES determines that the Contractor has defaulted by failing to carry out the terms or conditions of this Contract or by failing to meet the applicable requirements in sections 1932 and 1903(m) of the Social Security Act, or in the event that ASES determines that the Contractor falls within the prohibitions stated in Section 29.1 or 29.6, ASES may terminate the Contract in addition to or in lieu of any other remedies set out in this Contract or available by law. |
| 35.2.2 | Before terminating this Contract, ASES will: |
| 35.2.2.1 | Provide written notice of the intent to terminate at least thirty (30) Calendar Days prior to the Termination Date, stating the reason for the termination and the time and place of a hearing, to take place at least fifteen (15) Calendar Days after the date of mailing of the notice of intent to terminate, to give the Contractor an opportunity to appeal the determination or cure the default; |
| 35.2.2.2 | Provide written notice of the decision affirming or reversing the proposed termination of the Contract, and for an affirming decision, the effective date of the termination; and |
| 35.2.2.3 | For an affirming decision, give Enrollees of the Contractor notice of the termination and information consistent with 42 CFR 438.10 on their options for receiving services following the Termination Date of the Contract. |
| 35.3 | Termination for Convenience |
| 35.3.1 | ASES may terminate this Contract for convenience and without cause upon thirty (30) Calendar Days written notice. Termination for convenience shall not be a breach of the Contract by ASES. The Contractor shall be entitled to receive, and shall be limited to just and equitable compensation for any satisfactory authorized work performed as of the Termination Date of the Contract. |
| 35.4 | Termination for Insolvency or Bankruptcy |
| 35.4.1 | The Contractor’s insolvency, or the Contractor’s filing of a petition in bankruptcy, shall constitute grounds for termination for cause. In the event of the filing of a petition in bankruptcy, the Contractor shall immediately advise ASES. If ASES reasonably determines that the Contractor's financial condition is not sufficient to allow the Contractor to provide the services as described herein in the manner required by ASES, ASES may terminate this Contract in whole or in part, Immediately or in stages. The Contractor's financial condition shall be presumed not sufficient to allow the Contractor to provide the services described herein, in the manner required by ASES if the Contractor cannot demonstrate to ASES's satisfaction that the Contractor has risk reserves and a minimum net worth sufficient to meet the statutory standards for licensed health care plans, as required under this Contract. The Contractor shall cover continuation of services to Enrollees for the duration of period for which payment has been made, as well as for inpatient admissions up to discharge. |
Page 284 of 301
| 35.4.2 | In the event that this Contract is terminated because of the Contractor’s insolvency, the Contractor shall guarantee that Enrollees shall not be liable for: |
| 35.4.2.1 | The Contractor’s debts; |
| 35.4.2.2 | The Covered Services provided to the Enrollee, for which ASES does not pay the Contractor or its Network Providers; |
| 35.4.2.3 | The Covered Services provided to the Enrollee, for which ASES or the Contractor does not pay a Provider who furnishes the services under a contractual, Referral, or other arrangement; or |
| 35.4.2.4 | Payment for Covered Services furnished under a contractual, Referral, or other arrangement, to the extent that those payments are in excess of the amount that the Enrollee would owe if the Contractor provided the services directly. |
| 35.4.3 | The Contractor shall cover continuation of services to Enrollees for the duration of the period for which payment has been made by ASES, as well as for inpatient admissions up to discharge. |
| 35.5 | Termination for Insufficient Funding |
| 35.5.1 | In the event that Federal and/or Puerto Rico funds to finance this Contract become unavailable or insufficient, ASES may terminate the Contract in writing, unless both Parties agree, through a written amendment, to a modification of the obligations under this Contract. |
| 35.5.2 | The Termination Date of the Contract when the Contract is terminated due to insufficient funding shall be ninety (90) Calendar Days after ASES delivers written notice to the Contractor, unless available funds are insufficient to continue payments in full during the ninety (90) Calendar Day period, in which case ASES shall give the Contractor written notice of an earlier date at which the Contract shall terminate. |
| 35.5.3 | Upon termination, the Contractor shall comply with the phase-out obligations established in Article 36 of this Contract. |
| 35.5.4 | In the event of termination for insufficient funding, the Contractor shall be entitled to receive, and shall be limited to, just and equitable compensation for any satisfactory authorized work performed as of the Termination Date of the Contract. |
Page 285 of 301
| 35.5.5 | Availability of funds shall be determined solely by ASES. |
| 35.6 | Termination Under Section 29.3 |
| 35.6.1 | If any of the events specified in Section 29.3 of this Contract occur, ASES may terminate this Contract as required under Act 458 and Act 84. |
| 35.6.2 | Upon Termination, the Contractor shall comply with the phase-out obligations established in Article 36 of this Contract. |
| 35.7 | ASES may terminate this Contract for any other just reason upon thirty (30) Calendar Days written notice. |
| 35.8 | Termination Procedures |
| 35.8.1 | ASES will issue a written notice of termination to the Contractor by certified mail, return receipt requested, or in person with evidence of delivery. The notice of termination shall cite the provision of this Contract giving the right to terminate, the circumstances giving rise to termination, and the Termination Date of the Contract. Termination shall be effective at 11:59 p.m. EST on the Termination Date of the Contract. |
| 35.8.2 | Upon receipt of notice of termination or on the date specified in the notice of termination and as directed by ASES, the Contractor shall: |
| 35.8.2.1 | Stop work under the Contract on the date and to the extent specified in the notice of termination; |
| 35.8.2.2 | Place no further orders or subcontract for materials, services, or facilities, except as may be necessary for completion of such portion of the work under the Contract as is not terminated; |
| 35.8.2.3 | Terminate all orders and subcontracts to the extent that they relate to the performance of work terminated by the notice of termination; |
| 35.8.2.4 | Assign to ASES, in the manner and to the extent directed by ASES, all of the right, title, and interest of Contractor under the orders or subcontracts so terminated, in which case ASES will have the right, at its discretion, to settle or pay any or all Claims arising out of the termination of such orders and subcontracts; |
| 35.8.2.5 | With the prior written approval of ASES, settle all outstanding liabilities and all Claims arising out of such termination or orders and subcontracts, the cost of which would be reimbursable in whole or in part, in accordance with the provisions of this Contract; |
Page 286 of 301
| 35.8.2.6 | Complete the performance of such part of the work that was not terminated by the notice of termination; |
| 35.8.2.7 | Take such action as may be necessary, or as ASES may direct, for the protection and preservation of any and all property or information related to the Contract that is in the possession of the Contractor and in which ASES has or may acquire an interest; |
| 35.8.2.8 | Promptly make available to ASES, or to another MCO acting on behalf of ASES, any and all records, whether medical or financial, related to the Contractor's activities undertaken pursuant to this Contract. Such records shall be provided at no expense to ASES; |
| 35.8.2.9 | Promptly supply all information necessary to ASES, or another ASES plan acting on behalf of ASES, for reimbursement of any outstanding Claims at the time of termination; and |
| 35.8.2.10 | Submit a termination/transition plan to ASES for review and prior written approval that includes commitments to carry out at minimum the following obligations: |
| 35.8.2.10.1 | Provide Enrollees continuation of all the Covered Services and Benefits during a defined transition period, such transition period to be determined by ASES; |
| 35.8.2.10.2 | Comply with all duties and/or obligations incurred prior to the actual Termination Date of the Contract, including but not limited to, the Grievance and Appeal process as described in Article 14; |
| 35.8.2.10.3 | Maintain Claims processing functions as necessary for ten (10) consecutive months from the Termination Date of the Contract in order to complete adjudication of all Claims; |
| 35.8.2.10.4 | Create a task force to reconcile and certify any pending and outstanding balances in connection with services rendered by the Contractor under the Contract and previous contracts between ASES and the Contractor. |
| 35.8.2.10.5 | File all reports concerning the Contractor’s operations during the term of the Contract in the manner described in this Contract; |
| 35.8.2.10.6 | Assist ASES in making all necessary notices to Enrollees and Providers at least thirty (30) Calendar Days prior to the effective date of change and as may be required under the Contract, or otherwise required under applicable law, regarding notices to Enrollees; |
Page 287 of 301
| 35.8.2.10.7 | Ensure the efficient and orderly transition of Enrollees from coverage under this Contract to coverage under any new arrangement developed or agreed to by ASES, including cooperation with another contractor, as provided in Article 35; |
| 35.8.2.10.8 | Ensure the proper identification of the Enrollees requiring the authorization for either prescription medications or DME to avoid any interruptions in services by providing such Data to ASES as contemplated in the transition plan; |
| 35.8.2.10.9 | Submit to ASES all scripts used at Call Centers to communicate with Enrollees during the transition period; |
| 35.8.2.10.10 | Maintain the financial requirements and insurance set forth in this Contract until ASES provides the Contractor written notice that all continuing obligations of this Contract have been fulfilled; |
| 35.8.2.10.11 | Submit reports to ASES as directed but no less frequently than every thirty (30) Calendar Days, detailing the Contractor’s progress in completing its continuing obligations under this Contract, until completion; and |
| 35.8.2.10.12 | Meet with ASES personnel, as requested, to ensure satisfactory completion of all obligations under the Termination Plan. |
| 35.8.3 | This Termination Plan shall be subject to review and approval by CMS. |
| 35.8.4 | Upon completion of these continuing obligations, the Contractor shall submit a final report to ASES describing how the Contractor has completed its continuing obligations. ASES will advise, within twenty (20) Calendar Days of receipt of this report, if all of the Contractor’s obligations are discharged. If ASES finds that the final report does not evidence that the Contractor has fulfilled its continuing obligations, then ASES will require the Contractor to submit a revised final report to ASES for approval, and take any other action necessary to discharge all of its duties under this Contract, as directed by ASES. |
| 35.8.5 | Except as provided in this Article 35, a notification that ASES intends to terminate this Contract shall not release the Contractor from its obligations to pay for Covered Services rendered or otherwise to perform under this Contract. |
Page 288 of 301
| 35.9 | Termination Claims |
| 35.9.1 | After receipt of a notice of termination, the Contractor shall submit to ASES any termination claim in the form, and with the certification prescribed by, ASES. Such claim shall be submitted promptly but in no event later than ten (10) months from the Termination Date of the Contract. Upon failure of the Contractor to submit its termination claim within the time allowed, ASES may determine, on the basis of information available, the amount, if any, due to the Contractor by reason of the termination and shall thereupon cause to be paid to the Contractor the amount so determined. |
| 35.9.2 | Upon receipt of notice of termination, the Contractor shall have no entitlement to receive any amount for lost revenues or anticipated profits or for expenditures associated with this Contract or any other contract. Upon termination the Contractor shall be paid in accordance with the following: |
| 35.9.2.1 | At the Contract price(s) for services delivered to and accepted by ASES; and/or |
| 35.9.2.2 | At a price mutually agreed upon by the Contractor and ASES for partially completed services. |
| 35.9.3 | In the event the Contractor and ASES fail to agree in whole or in part as to the amounts with respect to costs to be paid to the Contractor in connection with the total or partial termination of work pursuant to this article, ASES will determine, on the basis of information available, the amount, if any, due to the Contractor by reason of termination and shall pay to the Contractor the amount so determined. |
| 35.10 | Limited Right of Termination by the Contractor |
| 35.10.1 | Subject to compliance with the termination procedures set forth in Section 35.8, the Contractor may terminate this Contract under the following circumstances: |
| 35.10.1.1 | Termination Due to ASES’s Financial Breach. Upon fifteen (15) Calendar Days written notice, in the event ASES defaults in making payment of three (3) consecutive monthly PMPM Payments and fails to cure such breach within the notice period. For purposes of this Section, a default in making payment does not include instances where ASES has made any Withhold payments pursuant to the terms of this Contract, provided that ASES has given the Contractor advance written notice of any such Withhold. |
| 35.10.1.2 | Termination Due to Insufficient Funding. Immediately, upon receipt from ASES of a written notice pursuant to Section 35.5 that appropriated federal and/or Puerto Rico funds become unavailable or that such funds will be insufficient for the payment of ASES's obligation under this Contract when due, unless both Parties agree, through a written amendment, to a modification of the obligations under this Contract. |
Page 289 of 301
| 35.10.1.3 | If forty-five (45) Calendar Days before the last day of each fiscal year covered under the Contract, the Contractor and ASES have not (as provided in Section 21.4 agreed to PMPM for the succeeding fiscal year, the Contractor may exercise an option to terminate the Contract by giving ASES written notice of the Contractor’s intent not to continue to provide services under the Contract no later than forty-five (45) Calendar Days prior to the termination of the corresponding fiscal year. Once the Contractor has given ASES such written notice, the Contractor shall fully discharge the termination phase-out obligations listed in Section 35.8. At any time before the end of the fiscal year, the Contractor may rescind its notice of termination, if the Parties reach an agreement on rates for the following fiscal year. |
ARTICLE 36 PHASE-OUT AND COOPERATION WITH OTHER CONTRACTORS
| 36.1 | If, in the best interest of Enrollees of GHP, ASES terminates any GHP contract, the Contractor shall, upon the request of ASES, assume responsibility for the geographic areas (municipalities or Service Regions) previously managed by any MCO or other contractor whose contractual arrangement with ASES was terminated, in accordance with the contracted PMPM Payment, pursuant to the written amendment of the Contract, if required. |
| 36.2 | If in the best interest of Enrollees of GHP, ASES develops and implements new projects that impact the scope of services, the Contractor shall assist in the transition process, after receiving at least ninety (90) Calendar Days written notice from ASES of such change, and pursuant to written amendment of the Contract, if required. PMPM Payments shall be adjusted accordingly. |
| 36.3 | In the event that ASES has entered into, or enters into, agreements with other contractors for additional work related to the Benefits rendered hereunder, the Contractor agrees to cooperate fully with such other contractors. The Contractor shall not commit any act or omission that will interfere with the performance of work by any other contractor, or actions taken by ASES to facilitate the work. |
| 36.4 | If ASES chooses not to renew this Contract, pursuant to Article 21, the Contractor agrees that it will not engage in any behavior or inaction that prevents or hinders the work of another contractor or ASES, as the case may be. Upon receiving ASES’s notice that it does not intend to renew the Contract, the Contractor agrees to submit a written termination/transition plan to ASES within thirty (30) Calendar Days of receiving the notice. The turn-over plan shall include all the elements listed in Section 35.8.2.9.1. The Parties agree that the Contractor has not successfully met this obligation until ASES accepts its turn-over plan and/or transition plan, required under this Article 36 |
Page 290 of 301
ARTICLE 37 INSURANCE
| 37.1 | The Contractor shall, at a minimum, prior to the commencement of work, procure the insurance policies identified below at the Contractor’s own cost and expense and shall furnish ASES with proof of coverage at least in the amounts indicated. It shall be the responsibility of the Contractor to require any Subcontractor to secure the same insurance coverage as prescribed herein for the Contractor, and to obtain a certificate evidencing that such insurance is in effect. In the event that any such insurance is proposed to be reduced, terminated or cancelled for any reason, the Contractor shall provide to ASES at least thirty (30) Calendar Days prior written notice. Prior to the reduction, expiration and/or cancellation of any insurance policy required hereunder, the Contractor shall secure replacement coverage upon the same terms and provisions to ensure no lapse in coverage, and shall furnish, at the request of ASES, a certificate of insurance indicating the required coverage. The Contractor shall maintain insurance coverage sufficient to insure against claims arising at any time during the term of the Contract. The provisions of this Section shall survive the expiration or termination of this Contract for any reason. In addition, the Contractor shall indemnify and hold harmless ASES and the Commonwealth from any liability arising out of the Contractor’s or its Subcontractor’s untimely failure in securing adequate insurance coverage as prescribed herein: |
| 37.1.1 | Workers’ Compensation Insurance, the policy(ies) to insure the statutory limits established by law of the Commonwealth. The Workers’ Compensation Policy must include Coverage B – Employer’s Liability Limits of: |
| 37.1.1.1 | Bodily injury by accident: five hundred thousand dollars ($500,000) each accident; |
| 37.1.1.2 | Bodily injury by disease: five hundred thousand dollars ($500,000) each employee; and |
| 37.1.1.3 | One million dollars ($1,000,000) policy limits. |
| 37.1.2 | The Contractor shall require all Subcontractors performing work under this Contract to obtain an insurance certificate showing proof of Worker’s Compensation Coverage. |
| 37.1.3 | The Contractor shall have commercial general liability policy(ies) as follows: |
| 37.1.3.1 | Combined single limits of one million dollars ($1,000,000) per person and three million dollars ($3,000,000) per occurrence; |
| 37.1.3.2 | On an “occurrence” basis; and |
Page 291 of 301
| 37.1.3.3 | Liability for property damage in the amount of three million dollars ($3,000,000) including contents coverage for all records maintained pursuant to this Contract. |
ARTICLE 38 COMPLIANCE WITH ALL LAWS
| 38.1 | Nondiscrimination |
| 38.1.1 | The Contractor shall comply with applicable Federal and Puerto Rico laws, rules, and regulations, and the Puerto Rico policy relative to nondiscrimination in employment practices because of political affiliation, religion, race, color, sex, physical handicap, age, or national origin. Applicable Federal nondiscrimination law includes, but is not limited to, Title VI of the Civil Rights Act of 1964, as amended; Title IX of the Education Amendments of 1972, as amended; the Age Discrimination Act of 1975, as amended; Equal Employment Opportunity and its implementing regulations (45 CFR 74 Appendix A (1), Executive Order 11246 and 11375); the Rehabilitation Act of 1973; and the Americans with Disabilities Act of 1993 and its implementing regulations (including but not limited to 28 CFR § 35.100 et seq.). Nondiscrimination in employment practices is applicable to employees for employment, promotions, dismissal and other elements affecting employment. |
| 38.1.2 | The Contractor shall comply with all provisions of the Puerto Rico Patient’s Bill of Rights and it’s implementing regulation, which prohibit discrimination against any patient. |
| 38.2 | Compliance with All Laws in the Delivery of Service |
| 38.2.1 | The Contractor agrees that all work done as part of this Contract will comply fully with and abide by all applicable Federal and Puerto Rico laws, rules, regulations, statutes, policies, or procedures that may govern the Contract, including but not limited to those listed in Attachment 1. |
| 38.2.2 | All applicable Puerto Rico and Federal laws, rules, and regulations, consent decrees, court orders, policy letters and normative letters, and policies and procedures, including but not limited to those described in Attachment 1, are hereby incorporated by reference into this Contract. Any change in those applicable laws and requirements, including any new law, regulations, policy guidance, or normative letter, shall be automatically incorporated into this Contract by reference as soon as it becomes effective. |
| 38.2.3 | To the extent that applicable laws, rules, regulations, statutes, policies, or procedures require the Contractor to take action or inaction, any costs, expenses, or fees associated with that action or inaction shall be borne and paid by the Contractor solely. Such compliance-associated costs include, but are not limited to, attorneys’ fees, accounting fees, research costs, or consultant costs, where these costs are related to, arise from, or are caused by compliance with any and all laws. In the event of a disagreement on this matter, ASES’s determination on this matter shall be conclusive and not subject to appeal. |
Page 292 of 301
| 38.2.4 | The Contractor shall include notice of grantor agency requirements and regulations pertaining to reporting and patient rights under any contracts involving research, developmental, experimental or demonstration work with respect to any discovery or invention which arises or is developed in the course of or under such contract, and of grantor agency requirements and regulations pertaining to copyrights and rights in Data. |
| 38.2.5 | The Contractor certifies and warrants to ASES that at the time of execution of this Contract: (i) it is a corporation duly authorized to conduct business in Puerto Rico, and has filed all the required income tax returns for the preceding five years; and (ii) it filed its report due with the Office of the Commissioner of Insurance during the five (5) years preceding the Execution Date of this Contract. |
ARTICLE 39 CONFLICT OF INTEREST AND CONTRACTOR INDEPENDENCE
| 39.1 | The duty to provide information about interests and conflicting relations is continuous and extends throughout the Contract Term. |
| 39.2 | The Contractor covenants that it presently has no interest and shall not acquire any interest, direct or indirect, that would conflict in any material manner or degree with, or have a material adverse effect on the performance of its services hereunder. The Contractor further covenants that in the performance of the Contract no person having any such interest shall be employed. The Contractor shall submit a conflict of interest form, attesting to these same facts, by January 10 of each calendar year; and at any time, within fifteen (15) Calendar Days of request by ASES. |
| 39.3 | It shall be the responsibility of the Contractor to maintain independence and to establish necessary policies and procedures to assist the Contractor in determining if the actual individuals performing work under this Contract have any impairment to their independence. |
| 39.4 | The Contractor further agrees to take all necessary actions to eliminate threats to impartiality and independence, including but not limited to reassigning, removing, or terminating Providers or Subcontractors. |
ARTICLE 40 CHOICE OF LAW OR VENUE
| 40.1 | This Contract shall be governed in all respects by the laws of Puerto Rico. Any lawsuit or other action brought against ASES or the Commonwealth based upon or arising from this Contract shall be brought in a court or other forum of competent jurisdiction in Puerto Rico. |
Page 293 of 301
ARTICLE 41 ATTORNEY’S FEES
| 41.1 | In the event that either Party deems it necessary to take legal action to enforce any provision of this Contract, and in the event ASES prevails, the Contractor agrees to pay all expenses of such an action including reasonable attorney’s fees and costs at all stages of litigation as awarded by the court, a lawful tribunal, a hearing officer, or an administrative law judge. The term legal action shall be deemed to include administrative proceedings of all kinds, as well as all actions regarding the law or equity. |
ARTICLE 42 SURVIVABILITY
| 42.1 | The terms, provisions, representations, and warranties contained in this Contract shall survive the delivery or provision of all services hereunder. |
ARTICLE 43 PROHIBITED AFFILIATIONS WITH INDIVIDUALS DEBARRED AND SUSPENDED
| 43.1 | The Contractor certifies that it is not presently debarred, suspended, proposed for debarment, or declared ineligible for award of contracts by any Federal or Puerto Rico agency, as provided in Section 13.4. In addition, the Contractor certifies that it does not employ or subcontract with any person or entity that could be excluded from participation in the Medicaid Program under 42 CFR 1001.1001 (exclusion of entities owned or controlled by a sanctioned person) or 1001.1051 (exclusion of individuals with ownership or control interest in sanctioned entities). Any violation of this Article shall be grounds for termination of the Contract. |
ARTICLE 44 WAIVER
| 44.1 | No covenant, condition, duty, obligation, or undertaking contained in or made a part of the Contract shall be waived except by the written agreement of the Parties. Forbearance or indulgence in any form or manner by either Party in any regard whatsoever shall not constitute a waiver of the covenant, conditions, duties, obligations, and undertakings to be kept, performed, or discharged by the Party to which the same may apply. Notwithstanding any such forbearance or indulgence, the other Party shall have the right to invoke any Remedy available under law or equity until complete performance or satisfaction of all such covenants, conditions, duties, obligations, and undertakings. |
| 44.2 | The waiver by ASES of any breach of any provision contained in this Contract shall not be deemed to be a waiver of such provision or any subsequent breach of the same or any other provision contained in this Contract and shall not establish a course of performance between the Parties contradictory to the terms hereof. No term or condition of the Contract shall be held to be waived, modified, or deleted except by an instrument, in writing, signed by the Parties thereto. |
ARTICLE 45 FORCE MAJEURE
| 45.1 | Neither Party of this Contract shall be held responsible for delays or failures in performance resulting from acts beyond the control of each Party. Such acts shall include, but not be limited to, acts of God, strikes, riots, lockouts, acts of war, epidemics, fire, earthquakes, or other disasters. |
Page 294 of 301
ARTICLE 46 BINDING
| 46.1 | This Contract and all of its terms, conditions, requirements, and amendments shall be binding on ASES and the Contractor and for their respective successors and permitted assigns. |
ARTICLE 47 TIME IS OF THE ESSENCE
| 47.1 | Time is of the essence in this Contract. Any reference to “days” shall be deemed Calendar Days unless otherwise specifically stated. |
ARTICLE 48 AUTHORITY
| 48.1 | ASES has full power and authority to enter into this Contract as does the person acting on behalf of and signing for the Contractor. Additionally, the person signing on behalf of the Contractor has been properly authorized and empowered to enter into this Contract on behalf of the Contractor and to bind the Contractor to the terms of this Contract. Each Party further acknowledges that it has had the opportunity to consult with and/or retain legal counsel of its choice and read this Contract. Each party acknowledges that it understands this Contract and agrees to be bound by it. |
ARTICLE 49 ETHICS IN PUBLIC CONTRACTING
| 49.1 | The Contractor understands, states, and certifies that it made its Proposal without collusion or Fraud and that it did not offer or receive any kickbacks or other inducements from any other Contractor, supplier, manufacturer, or Subcontractor in connection with its Proposal. |
ARTICLE 50 CONTRACT LANGUAGE INTERPRETATION
| 50.1 | The Contractor and ASES agree that in the event of a disagreement regarding, arising out of, or related to, Contract language interpretation, ASES’s interpretation of the Contract language in dispute shall control and govern. |
ARTICLE 51 ARTICLE AND SECTION TITLES NOT CONTROLLING
| 51.1 | The Article and Section titles used in this Contract are for reference purposes only and shall not be deemed to be a part of this Contract. |
ARTICLE 52 LIMITATION OF LIABILITY/EXCEPTIONS
| 52.1 | Nothing in this Contract shall limit the Contractor’s indemnification liability or civil liability arising from, based on, or related to claims brought by ASES or any Third Party or any claims brought against ASES or the Commonwealth by a Third Party or the Contractor. |
Page 295 of 301
ARTICLE 53 COOPERATION WITH AUDITS
| 53.1 | The Contractor shall assist and cooperate with ASES in any and all matters and activities related to or arising out of any audit or review, whether Federal, private, or internal in nature, at no cost to ASES. |
| 53.2 | The Parties also agree that the Contractor shall be solely responsible for any costs it incurs for any audit related inquiries or matters. Moreover, the Contractor may not charge or collect any fees or compensation from ASES for any matter, activity, or inquiry related to, arising out of, or based on an audit or review. |
| 53.3 | ASES reserves the right to audit the Contractor and/or its Subcontractors at any time during the term of the Contract. The Contractor and/or its Subcontractors shall be solely responsible for the cost of such audits. |
ARTICLE 54 OWNERSHIP AND FINANCIAL DISCLOSURE
| 54.1 | The Contractor and Subcontractors shall disclose financial statements for each person or corporation with an ownership or control interest of five percent (5%) or more of its entity. For the purposes of this Section, a person or corporation with an ownership or control interest shall mean a person or corporation: |
| 54.1.1 | That owns directly or indirectly five percent (5%) or more of the Contractor’s/Subcontractor’s capital or stock or received five percent (5%) or more of its profits; |
| 54.1.2 | That has an interest in any mortgage, deed of trust, note, or other obligation secured in whole or in part by the Contractor/Subcontractor or by its property or assets, and that interest is equal to or exceeds five percent (5%) of the total property and assets of the Contractor/Subcontractor; and |
| 54.1.3 | That is an officer or director of the Contractor/Subcontractor (if it is organized as a corporation) or is a partner in the Contractor’s/Subcontractor’s organization (if it is organized as a partnership). |
| 54.2 | As per 42 CFR §455.104, disclosure by the Contractor will include the following information on ownership and control: |
| 54.2.1 | The name and address of any person (individual or corporation) with an ownership or control interest in the disclosing entity, fiscal agent, or managed care entity. The address for corporate entities must include as applicable primary business address, every business location, and P.O. Box address. |
Page 296 of 301
| 54.2.2 | Date of birth and Social Security Number (in the case of an individual). |
| 54.2.3 | Other tax identification number (in the case of a corporation) with an ownership or control interest in the disclosing entity (or fiscal agent or managed care entity) or in any Subcontractor in which the disclosing entity (or fiscal agent or managed care entity) has a five percent (5%) or more interest. |
| 54.2.4 | Whether the person (individual or corporation) with an ownership or control interest in the disclosing entity (or fiscal agent or managed care entity) is related to another person with ownership or control interest in the disclosing entity as a spouse, parent, child, or sibling; or whether the person (individual or corporation) with an ownership or control interest in any Subcontractor in which the disclosing entity (or fiscal agent or managed care entity) has a five percent (5%) or more interest is related to another person with ownership or control interest in the disclosing entity as a spouse, parent, child, or sibling. |
| 54.2.5 | The name of any other disclosing entity (or fiscal agent or managed care entity) in which an owner of the disclosing entity (or fiscal agent or managed care entity) has an ownership or control interest. |
| 54.2.6 | The name, address, date of birth, and Social Security Number of any managing employee of the disclosing entity (or fiscal agent or managed care entity). |
| 54.2.7 | Disclosures from providers or disclosing entities. Providers or disclosing entities shall comply with the information disclosure required by Section 54.2. Disclosure from any provider or disclosing entity is due at any of the following times: |
| 54.2.7.1 | Upon the provider or disclosing entity submitting the provider application. |
| 54.2.7.2 | Upon the provider or disclosing entity executing the provider agreement. |
| 54.2.7.3 | Upon request of the Medicaid agency during the re-validation of enrollment process under 42 § 455.414. |
| 54.2.7.4 | Within 35 days after any change in ownership of the disclosing entity. |
| 54.2.8 | Disclosures from fiscal agents. Fiscal agents shall comply with the information disclosure required by Section 54.2 Disclosures from fiscal agents are due at any of the following times: |
| 54.2.8.1 | Upon the fiscal agent submitting the proposal in accordance with the Commonwealth's procurement process. |
Page 297 of 301
| 54.2.8.2 | Upon the fiscal agent executing the contract with the Commonwealth. |
| 54.2.8.3 | Upon renewal or extension of the contract. |
| 54.2.8.4 | Within thirty-five (35) Calendar Days after any change in ownership of the fiscal agent. |
| 54.2.9 | Disclosures from managed care entities. Managed care entities shall comply with the information disclosure required by Section 54.2. Disclosures from managed care entities (MCOs, PIHPs, PAHPs, and HIOs), are due at any of the following times: |
| 54.2.9.1 | Upon the managed care entity submitting the proposal in accordance with the Commonwealth's procurement process. |
| 54.2.9.2 | Upon the managed care entity executing the contract with the Commonwealth. |
| 54.2.9.3 | Upon renewal or extension of the contract. |
| 54.2.10 | Within thirty-five (35) Calendar Days after any change in ownership of the managed care entity. |
ARTICLE 55 AMENDMENT IN WRITING
| 55.1 | No amendment, waiver, termination, or discharge of this Contract, or any of the terms or provisions hereof, shall be binding upon either Party unless confirmed in writing by ASES and any other appropriate governmental agency. Additionally, CMS approval shall be required before any such amendment is effective. Any agreement of the Parties to amend, modify, eliminate, or otherwise change any part of this Contract shall not affect any other part of this Contract, and the remainder of this Contract shall continue to be in full force and effect as set out herein. |
| 55.2 | ASES reserves the authority to seek an amendment to this Contract at any time if such an amendment is necessary in order for the terms of this Contract to comply with Federal law. The Contractor shall consent to any such amendment. |
ARTICLE 56 CONTRACT ASSIGNMENT
| 56.1 | The Contractor shall not assign this Contract, in whole or in part, without the prior written consent of ASES, and any attempted assignment not in accordance herewith shall be null and void and of no force or effect. |
ARTICLE 57 SEVERABILITY
| 57.1 | If any Article, Section, paragraph, term, condition, provision, or other part of this Contract (including items incorporated by reference) is judged, held, declared, or found to be voidable, illegal, unenforceable, invalid or void, then both ASES and the Contractor shall be relieved of all obligations arising under such provision. However, if the remainder of the Contract is capable of being performed, it shall not be affected by such declaration or finding and those duties and tasks shall be fully performed. To this end, the provisions of the Contract are declared to be severable. |
Page 298 of 301
ARTICLE 58 ENTIRE AGREEMENT
| 58.1 | This Contract constitutes the entire agreement between the Parties with respect to the subject matter herein and supersedes all prior negotiations, representations, or contracts. No written or oral agreements, representatives, statements, negotiations, understandings, or discussions that are not set out, referenced, or specifically incorporated in this Contract shall in any way be binding or of effect between the Parties. |
| 58.2 | The terms of the Request for Proposals and of the Contractor’s Proposal are incorporated by reference, except as otherwise provided in this Contract. However, in the event of a conflict between the terms of this Contract and the terms of the Request for Proposals or the terms of the Contractor’s Proposal, the terms of this Contract shall prevail. |
| 58.3 | All applicable laws are incorporated by reference into this Contract, as provided in Article 38. |
| 58.4 | Subject to Section 55, the Contractor acknowledges that it may be necessary or convenient during the Contract Term to clarify or supplement certain terms and conditions of this Contract so that it conforms to the terms of the Request for Proposals or otherwise in order to incorporate CMS requirements. In any of these events, the Contractor agrees that ASES shall have the right to issue from time to time normative letters which shall be then incorporated into the Contract. Such normative letters are advisory in nature, and shall not, absent an amendment to the Contract, effect a change in the Contractor’s substantive obligations under this Contract. |
ARTICLE 59 INDEMNIFICATION
| 59.1 | The Contractor hereby releases and agrees to indemnify and hold ASES, the Commonwealth, and its departments, agencies, and instrumentalities harmless from and against any and all claims, demands, liabilities, losses, costs or expenses, and attorneys' fees, caused by, growing out of, or arising from this Contract, due to any act or omission on the part of the Contractor, its Agents, employees, customers, invitees, licensees, or others working at the direction of the Contractor or on its behalf, or due to any breach of this Contract by the Contractor, or due to the application or violation of any pertinent Federal, Puerto Rico or local law, rule or regulation. This indemnification extends to the successors and assigns of the Contractor and survives the termination of the Contract and the dissolution or, to the extent allowed by the law, the bankruptcy of the Contractor. |
ARTICLE 60 NOTICES
| 60.1 | All notices, consents, approvals, and requests required or permitted shall be given in writing and shall be effective for all purposes if hand delivered or sent by (i) personal delivery, (ii) expedited prepaid delivery service, either commercial or US Postal Service, with proof of attempted delivery, (iii) telecopies, or (iv) electronic mail. In each case of (c) and (d), with answer back acknowledged, addressed as follows: |
Page 299 of 301
| 60.1.1 | If to ASES at: |
|
Mailing Address:
Administración de Seguros de Salud
P.O. Box 195661
San Juan, PR 00919-5661
|
Physical Address:
Administración de Seguros de Salud
Urb. Caribe 1552
Ave. Ponce de León, Sec. El Cinco
San Juan, PR 00926-2706
|
Attention: Executive Director
| 60.1.2 | If to Contractor at: |
|
Mailing Address:
PO Box 363628
San Juan, PR 00919-1580
|
Physical Address:
Triple-S Salud, Inc.
1441 Ave. Roosevelt, 6th Floor
San Juan, PR 00920
|
Attention: President
| 60.1.3 | All notices, elections, requests, and demands under this Contract shall be effective and deemed received upon the earliest of (i) the actual receipt of the item by personal delivery or otherwise, (ii) two (2) Business Days after being deposited with a nationally recognized overnight courier service as required above, (iii) three (3) Business Days after being deposited in the US mail as required above or (iv) on the day sent if sent by facsimile with voice confirmation on or before 4:00 p.m. Atlantic Time on any Business Day or on the next Business Day if so delivered after 4:00 p.m. Atlantic Time or on any day other than a Business Day. Rejection or other refusal to accept or the inability to deliver because of changed address of which no notice was given as herein required shall be deemed to be receipt of the notice, election, request, or demand sent. |
ARTICLE 61 OFFICE OF THE COMPTROLLER
| 61.1 | ASES will file this Contract in the Office of the Comptroller of Puerto Rico within fifteen (15) Calendar Days from the Effective Date of the Contract. |
(Signatures on following page)
Page 300 of 301
SIGNATURE PAGE
IN WITNESS WHEREOF, the Parties state and affirm that they are duly authorized to bind the respected entities designated below as of the day and year indicated.
ADMINISTRACIÓN DE SEGUROS DE SALUD DE PUERTO RICO (ASES)
|
Ricardo Rivera, Executive Director
|
Date
|
TRIPLE-S SALUD, INC.
|
Pablo Almodóvar-Scalley, President
|
Date
|
Page 301 of 301
ATTACHMENT 1
Relevant Puerto Rico and Federal Laws and Regulations
Applicable Puerto Rico laws and regulations:
| • | Act 72 of September 7, 1993, as amended, known as “Puerto Rico Health Insurance Administration Law”. |
| • | Puerto Rico Insurance Code and its applicable regulations. |
| • | Act 81 of May 14, 1912; known as “Organic Law for the Puerto Rico Health Department” |
| • | Act 194 of August 25, 2000, as amended, known as “The Declaration of Patient’s Rights and Responsibilities” |
| • | Act 408 of October 2, 2000, as amended, known as “Puerto Rico Pharmacy Law” |
| • | Act 11 of April 11, 2011, as amended, known as “Organic Law of the Office of Patient Advocate” |
| • | Act 247 of September 3, 2004, as amended, known as the “Puerto Rico Pharmacy Law” |
| • | Act 139 of August 1, 2008, as amended, known as “Law for the Medical Licensing and Discipline Board” |
| • | Act 109 of June 28, 1974, as amended, known as “Law for the Puerto Rico Public Services Commission” |
| • | Act 225 of July 23, 1974, as amended, known as “Law for Ambulance Services” |
| • | The Public Services Commission’s Regulations for ambulance services in Puerto Rico, Regulation Num. 6737 of December 1, 2003. |
| • | Act 86 of August 16, 1997, known as “Law for Residents of Culebra and Vieques” |
| • | Act 227 of August 12, 1999, known as “Law for the Implementation of the Public Policy on Suicide Prevention” |
| • | Act 243 of November 10, 2006, known as “Law to establish the public policy concerning the use of the Social Security Number for identification and the protection of its confidentiality” |
| • | Act 84 of June 18, 2002, known as “Code of Ethics for Contractors, Suppliers and Applicants for Economic Incentives from the Executive Agencies of the Commonwealth” |
| • | Act 12 of July 24, 1985, as amended, known as the “Government Ethics Law” |
| • | Act 458 of December 29, 2000, as amended, known as “Law to Prohibit the Adjudication of Auctions to convicts of Fraud, Embezzlement or Illegal Misappropriation of Public Funds” |
| • | Act 70 of August 12, 1988, as amended, known as the “Puerto Rico Uniform Administrative Proceedings Law” |
| • | Act 111 of September 7, 2005, as amended, known as the “Law to Inform Citizens of the Security of Data Banks” |
| • | Act 126 of October 31, 2013, known as the “Law for the Protocol of Interagency Services for the Erderly Population Living in Infrahuman Conditions” |
Applicable federal laws and regulations:
| • | Puerto Rico Health Department’s State Plan (“Medicaid State Plan” and “CHIPS State Plan” |
| • | Title XIX of the Medical Assistance Program (“Grants to States for Medical Assistance Programs”) |
| • | Title XXI of the Social Security Act, Children’s Health Insurance Program (“CHIP”) |
| • | Federal rules and Regulations as established by the Center for Medicare & Medicaid Services (“CMS”) and the Checklist for Managed Care Contract Approval including, but not limited to: 42 CFR 422.208 and 210 (Physician incentive plans); 422.560-422.626; 42 CFR 438 (managed care) including subsections 56, 60, 66, 206(b), 214, 242; 42 CFR 431 (fair hearings and appeals); 42 CFR 455 (fraud and abuse reporting); 42 CFR 447 (timely claims payment); 45 CFR 74.53 (retention requirements for records); 42 CFR 433 Subpart D, 42 CFR 447.20 and 42 CFR 434 (third party liability); 42 CFR 435.911 and 435.914; 42 CFR 431.52-53 (ambulance services); 42 CFR 405.2402; 42 CFR Part 455.104; 42 CFR Part 455.106; 42 CFR 447.20 and 42 CFR 434.6(a)(9) |
| • | Davis-Bacon Act, 40 U.S.C. 276a, et seq.; |
| • | The Social Security Act, including Titles VI, VII, XIX and XXI |
| • | Copeland Anti-Kickback Act, 40 U.S.C 276c |
| • | Fair Labor Standards Act of 1938, 29 U.S.C 201 et seq. |
| • | Clean Air Act, 42 U.S.C. 7401 et seq. |
| • | Federal Water Pollution Control Act as Amended, 33 U.S.C. 1251 et seq. |
| • | Federal Rehabilitation Act of 1973 |
| • | Byrd Anti-Lobbying Amendment, 31 U.S.C. 1352 |
| • | The Clinical Laboratory Improvement Amendments of 1988; |
| • | The Health Insurance Portability and Accountability Act of 1996 (HIPAA); |
| • | Omnibus Budget Reconciliation Act of 1981, P.L. 97-38; |
| • | Debarment and Suspensions, 45 CFR 74 Appendix A(8) and Executive Orders 12549 and 12689 |
| • | Americans with Disabilities Act, 42 USC 12101 et seq.; |
| • | Medicare Modernization Act of 2003, P.L. 108-173 |
| • | Mental Health Parity and Addiction Equity Act of 2008, P.L. 110-343 |
| • | Patient Protection and Affordable Care Act, P.L. 111-148 |
Medicaid Laws, regulations and requirements pertain only to the Medicaid population.


___________, 2015
Dear Enrollee:
Greetings and welcome to the Government Health Plan of Puerto Rico!
The Health Insurance Administration (ASES, its Spanish acronym) has developed this Uniform Guide for you to be informed on the use of the benefits provided by the Government Health Plan (GHP). This way you can have available the information you need regardless of the company that provides your healthcare.
The GHP offers the broadest benefit coverage through a coordinated care model. Under the GHP model you will be able to move freely within the preferred network and visit your specialists, sub-specialists, laboratories, x-rays and other health care providers without the need for referral and without co-pays. Your Primary Medical Group (PMG) and the Health Plan will inform you who are the providers that compose the preferred network.
You have the opportunity to choose a Primary Medical Group (PMG) and a Primary Care Physician (PCP) and if you do not choose you will be assigned a PMG and PCP in your area. They will keep a complete clinical record on your health conditions, allergies, medications. All of the services offered will comply with the strictest quality and cost-effective standards required by the health industry and federal and Commonwealth regulations.
We ask that you keep your address and personal information updated by contacting the Medicaid Program Office in which you submitted your eligibility application. You must also attend your re-certification appointments so that you do not lose your health care benefits.
Visit your PCP for the necessary tests for your cholesterol, sugar, and blood pressure. Visiting your PCP can also help to early detect diseases such as cardiovascular diseases, diabetes and cancer.
We invite you to make good use of this benefit offered by the Government of Puerto Rico, whose aim is to safeguard your health.
Cordially,
Ricardo A. Rivera Cardona
Director Ejecutivo
Table of Contents
|
Table of Contents
|
2
|
|
CONTACT US
|
4
|
|
LANGUAGE
|
4
|
|
DEFINITIONS
|
4
|
|
HIPAA
|
7
|
|
IMPORTANT INFORMATION ON YOUR HEALTH PLAN
|
8
|
|
The Government Health Plan
|
8
|
|
Who is eligible to enjoy the services and benefits of the Government Health Plan?
|
8
|
|
Coordination of Benefits
|
9
|
|
AUTO-ENROLLMENT
|
9
|
|
Auto-Assignment
|
9
|
|
THIS IS YOUR ID CARD OF THE GOVERNMENT HEALTH PLAN
|
10
|
|
DISENROLLMENT
|
11 |
|
RE-ENROLLMENT
|
12
|
|
FRAUD, WASTE AND ABUSE
|
12
|
|
What is Fraud?
|
12
|
|
What is Abuse?
|
13
|
|
What is Waste?
|
13
|
|
What can I do to avoid Fraud, Waste and Abuse?
|
13
|
|
How can I report situations on Fraud, Waste and/or Abuse?
|
14
|
|
PRIMARY MEDICAL GROUP AND PRIMARY CARE PHYSICIAN
|
14
|
|
Can I change my Primary Medical Group or the Primary Care Physician?
|
14
|
|
Choosing the Primary Medical Group and the Primary Care Physician
|
15
|
|
RECERTIFICATION OF ELIGIBILITY
|
16
|
|
PUBLIC EMPLOYEE
|
16 |
|
What can I do if my eligibility in GHP is cancelled?
|
17 |
|
How can I enroll in another of the plans contracted for government employees?
|
17 |
|
Can the members of the Police Department of Puerto Rico enroll in the Government Health Plan?
|
18 |
|
WHAT IS COORDINATED CARE?
|
18 |
|
YOUR PRIMARY MEDICAL GROUP AND YOUR PRIMARY CARE PHYSICIAN
|
18 |
|
What is a Primary Medical Group?
|
18
|
|
What is a Preferred Providers Network?
|
19
|
|
Are all my Specialists within the Preferred Network of my Primary Medical Group?
|
20
|
|
What is your Health Plan’s General Network?
|
21
|
|
Will I need the Countersignature on the Prescriptions of Medications?
|
22
|
|
KNOW THE RESPONSIBILITY OF YOUR PRIMARY CARE PHYSICIAN
|
22
|
|
HOW TO OBTAIN INFORMATION ABOUT PARTICIPATING PHYSICIANS
|
23
|
|
THESE ARE YOUR RIGHTS
|
23
|
|
THESE ARE YOUR RESPONSIBILITIES
|
25
|
|
QUALITY AND PERFORMANCE INDICATORS
|
25
|
|
UTILIZATION MANAGEMENT POLICIES AND PROCEDURES
|
26
|
|
EMERGENCIES AND URGENCIES
|
27
|
|
How do I know when it is an emergency?
|
27
|
|
When can I receive emergency services?
|
27
|
2
|
And then, what is an Urgency?
|
27
|
|
How can I receive Urgency services?
|
28 |
|
How can I receive services outside business hours from my Primary Care Physician, the Primary Medical Group or the Preferred Network of Providers?
|
28
|
|
WHAT IS AN ADVANCE DIRECTIVE?
|
28
|
|
COMPLAINTS, GRIEVANCES AND APPEALS
|
28
|
|
What is a Complaint?
|
28
|
|
How can you file a Complaint?
|
29
|
|
What is a Grievance?
|
29
|
|
How can you file a Grievance?
|
29
|
|
What is an Action?
|
30
|
|
What is a Notice of Action?
|
30
|
|
What can I do if I do not agree with the Notice of Action?
|
31
|
|
What is an appeal?
|
31
|
|
Who will hear your Appeal?
|
32
|
|
How much time will they take to make a determination on my Appeal?
|
32
|
|
What is an Administrative Law Hearing?
|
32
|
|
How can you request an Administrative Law Hearing?
|
32
|
|
TIME TO SOLVE COMPLAINTS, GRIEVANCES AND APPEALS
|
33 |
|
DENTAL SERVICES
|
33 |
|
MENTAL HEALTH SERVICES
|
34 |
|
How can I receive mental health services or services against drug dependence?
|
34 |
|
PREVENTIVE SERVICES
|
34 |
|
What are preventive services?
|
34 |
|
HIV-AIDS
|
35 |
|
HEPATITIS-C
|
37 |
|
This is your Benefits Coverage
|
37 |
|
Preventive Services
|
38 |
|
Dental Services
|
39 |
|
Diagnostic Testing Services
|
40 |
|
Ambulatory Rehabilitation Services
|
40 |
|
Medical and Surgical Services
|
40 |
|
Ambulance Services
|
41 |
|
Non-Emergency Transportation Services (NEMT)
|
41 |
|
Maternity and Prenatal Services
|
42 |
|
Emergency Room Services
|
42 |
|
Post-Stabilization Services
|
43 |
|
Hospitalization Services
|
43 |
|
Mental Health Services
|
44 |
|
Mental Health Hospitalization Services
|
44 |
|
Pharmacy Services
|
44 |
|
Services Excluded from the Basic Coverage
|
45 |
|
Special Coverage Services
|
47 |
|
Services excluded from the Special Coverage
|
49 |
|
Medicare Coverage Services
|
50 |
|
DISEASE MANAGEMENT
|
50 |
|
Chronic Disease Management
|
50 |
|
Case Management
|
50 |
|
THESE ARE YOUR COPAYMENTS AND COINSURANCES
|
51 |
|
HEALTH REGIONS MAP
|
52 |
3
CONTACT US

|
Health Advocate Office
Toll-free 1-800-981-0031
Metro Area 787-977-1100
|
 |
Puerto Rico Health Insurance Administration
Toll-free 1-800981-2737
|
4
LANGUAGE
This Guide is provided in Spanish and English for your benefit. If any member of your family is enrolled in the Government Health Plan and the person has problems reading or has a disability such as blindness and needs special services to be able to receive the information provided in this Guide, the person may request help from your Health Plan. Your Health Plan must have different formats for the information to make them available to the Enrollees.
If the information provided in this Guide is confusing or if you need to clarify any questions, you may contact your Health Plan for assistance. Information is a vital component of the commitment of the Government Health Plan with you, our Enrollees. You may contact your Health Plan at the telephone numbers found on the back of your Government Health Plan ID card.
DEFINITIONS
Abuse: The excessive and improper use of a product, service or benefit, which results in unnecessary or excessive costs for the health care system.
Access: The guarantee that the Enrollee will be able to receive all the medically necessary services included in the Government Health Plan coverage without any barriers.
Administrative Referral: Written Authorization issued by your Health Plan for the Enrollee to receive the required service, if medically necessary, when the PCP or other PMG physician does not provide a Referral within the required time period.
Advance Directives: Written or verbal instructions, such as wills or powers-of attorney related to decisions about services and health care expressed by the person in advance in case an event occurs and he/she may be unable to make such decisions.
Ancillary Services: All those supplementary services provided to the Patient to assist in the diagnosis and Treatment of illness or injury. Examples of these services include laboratory, radiology, therapies, etc.
Authorization: A written document through which a person freely and voluntarily authorizes another person or provider to represent, him/her, apply, use and disclose health information for medical or Treatment purposes or to initiate an action such as a Grievance. It may also be used to annul a previous authorization.
Auto Enrollment: Automatic enrollment in the Health Plan of a Medicaid certified eligible person once the Health Plan is notified of such eligibility.
CHIP: Children Health Insurance Program, a federal program that provides medical Service Coverage to low-income children under age 18 through health plans qualified to offer coverage under this program.
Coinsurance: A percentage of the cost of a health service which the Enrollee must pay after receiving the service.
Commonwealth Population: The Commonwealth Population is comprised of the following: (i) Certain persons who are between twenty-two (22) and sixty-four (64) years of age, inclusive of the age limits, and who do not qualify for either Medicaid or CHIP; (ii) Police officers of the Commonwealth and their Dependents; (iii) Surviving spouses of deceased police officers;(iv) Survivors of domestic violence referred by the Office of the Women’s Advocate; and (v) Veterans.
5
Complaint: An informal claim on the quality of care, customer service or Treatment received by providers, personnel of your Health Plan, or PMG. It does not include disputes involving medical services, coverage or payment for services.
Consultation: An opinion a health professional requests to another health professional on a matter related to the health condition of a Patient.
Coordinated Care: Is the service provided to Enrollees by doctors who are part of the preferred network of providers in your Primary Medical Group. The Primary Care Physician is the leading provider of services and is responsible to periodically evaluate your health and coordinate all medical services you need.
Coordination of Benefits: The order in which health services are paid when the person has more than one medical plan. One of the plans is considered the primary plan and the other the secondary plan or secondary payer.
Copayment: An established fixed amount that is the Enrollee’s contribution to the expense for a medical service he/she receives.
Covered Services: Those services and benefits included in the Government Health Plan coverage.
Deductible: A fixed amount pre-determined by ASES, which the Enrollee must pay when he/she receives, health services.
ELA Puro: An option available to public employees so they can maintain medical coverage when they lose eligibility in the Medicaid Program and the enrollment for other health plans contracted under Law 95 has ended. This coverage is the same as the coverage of the Government Health Plan.
Enrollee: A person who after being certified as eligible under the Medicaid Program has completed the enrollment process with the Health Plan and for whom the Health Plan has issued the ID card that identifies the person as a Government Health Plan Enrollee.
Federal Population: CHIP and Medicaid eligible individuals.
Good Cause: Refers to situations that allow Enrollees to change his/her PCP or Primary Medical Group. These are: 1) The Enrollee moved outside the Region, 2) For reasons of moral or religious nature, the Health Plan does not perform the services the Enrollee needs, 3) The Enrollee need services that can be provided at the same time and not all services are available; failure to receive all the services as ordered may expose the Enrollee to unnecessary risk, 4) Other acceptable reasons include, but are not limited to, poor quality of care, lack of Access to services covered or lack of providers with experience to provide the health care the Enrollee needs. ASES will determine if the reason constitutes a Good Cause.
6
Grievance: A formal claim made by the Enrollee in writing, by telephone or by visiting your Health Plan or the Health Advocate Office, requesting a solution be granted when a service has been denied or allowed on a limited basis. A service; reduction, suspension or termination of a previously authorized service; total or partial denial of payment for a service; not having received services in a timely manner; when your Health Plan has not acted on a situation according to the established terms, refusal of your Health Plan let the Enrollee exercise his/her right to receive services outside the network
Health Plan: The managed care organization that is providing services in the GHP program. There is one health plan per region.
HIPAA (Health Insurance Portability and Accountability Act): The law that includes regulations for establishing safe electronic health records that will protect the privacy of a person’s medical information and prevent the misuse of this information.
Hospital: A facility that provides medical-surgical services to hospitalized Patients.
Identification (ID) Card: A card your Health Plan delivers to you once the Auto Enrollment is completed or you complete the subscription process, which identifies the Enrollee by name and contract number, and includes information on coverage, Copayments, customer service and health advice telephone numbers.
Medical Record: Detailed collection of data and information on the Treatment and care the Patient receives from a health professional.
Medicare Beneficiary: Persons aged 65 or more, who are disabled or have renal disease, who have Medicare Parts A coverage for Hospital services or Parts A and B for Hospital, ambulatory and medical services.
Medicaid: Program that provides health insurance for people with low or no income and limited resources, according to federal regulations.
Primary Care Physician (PCP): A licensed medical doctor (MD) who is a provider and who, within the scope of practice and in accordance with Puerto Rico Certification and licensure requirements, is responsible for providing all required primary care to Enrollees. The PCP is responsible for determining services required by Enrollees, provides continuity of care, and provides Referrals for Enrollees when Medically Necessary. A PCP may be a general practitioner, family physician, internal medicine physician, obstetrician/gynecologist, or pediatrician.
Patient: Person receiving Treatment for his mental and physical health.
Post-Stabilization Services: Covered Services, relating to an Emergency Medical Condition or Psychiatric Emergency, that are provided after an Enrollee is stabilized, in order to maintain the stabilized condition or to improve or resolve the Enrollee’s condition.
Preauthorization: Permission your Health Plan grants in writing to you, at the request of the PCP, Specialist or sub-specialist, to obtain a specialized service.
Prescription: Original written order issued by a duly licensed health professional, ordering the dispensing of a product, drug or formula.
7
Preferred Provider Network: Health Professionals duly licensed to practice medicine in Puerto Rico contracted by your Health Plan for the Enrollee to use as the first option. Enrollees can access these providers without Referral or co-payments if they belong to their Primary Medical Group.
Primary Medical Group: Health Professionals grouped to contract with your Health Plan to provide health services under a Coordinated Care model.
Referral: Written authorization a PCP issues to an Enrollee to receive services from a Specialist, sub-specialist or facility outside the preferred network of the Primary Medical Group.
Semi-Private Room: Hospital room with two beds.
Service Coverage: All the services offered to the Government Health Plan Enrollees under the Basic, Special, Mental, Dental and Pharmacy Coverages.
Special Coverage Registry: A form your Health Plan fills out at the request of the PCP when the Enrollee is diagnosed with one or more of the conditions that are part of the Special Coverage, for the Patient to receive Treatment and services directly from Specialists or sub-specialists without the need of a Referral.
Specialist: A health professional licensed to practice medicine and surgery in Puerto Rico that provides specialized medical and complementary services to the primary physicians. This category includes: cardiologists, endocrinologists, neurologists, surgeons, radiologists, psychiatrists, ophthalmologists, nephrologists, urologists, physiatrists, orthopedists, and other physicians not included in the definition of PCP.
Second Opinion: Additional Consultation the Enrollee makes to another physician with the same medical specialty to receive or confirm that the initially recommended medical procedure is the Treatment indicated for his condition.
Treatment: To provide, coordinate or manage health care and related services offered by health care providers.
Urgency: A medical condition that poses no risk of imminent death that can be treated in the doctor's office or in the facilities with extended hours and not in emergency rooms. An Urgency can become an emergency if not properly dealt with at the right time.
Waste: Is the overutilization of services, misuse of resources or other practices that, directly or indirectly, result in unnecessary costs.
HIPAA
The Health Insurance Administration (ASES) and the Health Plans are committed to maintain the confidentiality of your information. We may use and share information related to your Treatment, payment for medical services and everything related to health care within the strictest standards of confidentiality. With your written authorization we may provide your information to others for any purpose.
If you are interested in more information about the privacy practices or have questions or concerns, contact the Health Plan of the Region to which you belong.
8
|
IMPORTANT:
As a member of the Government Health Plan, you authorize the Federal Government, ASES, and the Health Advocate Office, the Health Plans or their representatives, to see your medical records to assess the quality, convenience, cost and promptness of services you receive.
|
IMPORTANT INFORMATION ON YOUR HEALTH PLAN
The Government Health Plan
Now, the new Government Health Plan of Puerto Rico offers more services and benefits. It also offers a Preferred Provider Network within the Primary Medical Group of your choice, which you can visit freely without the need for Referrals or paying Copayments.
Under the Government Health Plan you do not need the countersignature of the Primary Care Physician on the Prescriptions ordered by Specialists or sub-specialists within the Preferred Provider Network of your Primary Medical Group. You can freely choose dentists and pharmacies of your choice, among those contracted by the Government Health Plan.
In addition, you can receive mental health services within the same facility of Primary Medical Group. The Government Health Plan offers physical and mental health integrated services, so you can receive these services in one place.
Additional information including provider guidelines and information on the structure and operations of the GHP and physician incentive plans is available to you upon request. Please contact your Health Plan if you would like additional information.
Who is eligible to enjoy the services and benefits of the Government Health Plan?
The persons eligible under Law 72 of September 7, 1993, are:
| · | American citizens. |
| · | Persons with low or no income. |
| · | Population of Federal Medicaid Program: persons over age 65, persons that are blind or disabled and pregnant women. |
| · | Children under the CHIP Program. |
| · | Government employees, retirees and their dependents whose payroll is processed by the Treasury Department. |
| · | Members of the Police Department of Puerto Rico, their widows, widowers and children that survive them. |
| · | Veterans. |
9
| · | Children under State custody through the Family and Children Administration (ADFAN, for its acronym in Spanish). |
| · | Survivors of domestic violence through the Women’s Advocate Office. |
The Medicaid program will determine whether you are eligible for the Government Health Plan of Puerto Rico. Once you are certified eligible for Medicaid, they will give you form MA-10 entitled "Notice of Action Taken on Application and/or Re-Assessment" which indicates that you have been certified eligible. The MA-10 form includes the dates of your eligibility period. The Medicaid Program will also give the welcome letter to the Government Health Plan, from the Health Plan in your region.
You will receive your ID card by mail within five (5) business days after being certified eligible for Medicaid. If you do not receive the card during that period and you need medical services, you can show the MA-10 form to the contracted service provider with a contract with the Government Health Plan to show that your name is on the MA-10, that it is signed and you are authorized to receiving services.
Coordination of Benefits
As establish on Law 72 September 7, 1993, the Puerto Rico Government Health Plan, became the secondary payer to other healthcare plans that members are enrolled in. If you are currently enrolled in other healthcare plans, your responsibility is to provide ID Cards for each of your healthcare plans. Through this process, you agree to coordinate services and it will be your responsibility for the payment of the Deductible of the Government Health Plan.
AUTO-ENROLLMENT
As of July 1, 2011, every new beneficiary, who is eligible to the Government Health Plan of Puerto Rico, will be automatically enrolled and insured. This means you no longer have to visit your Health Plan to select your Primary Medical Group or your primary physician.
Auto-Assignment
Your Health Plan will send you the ID cards and information regarding the Primary Medical Group and Primary Care Physician assigned to you in order for you to access medical services immediately. You must receive your ID card by mail within 5 business days from the date you were certified as eligible. If you do not receive your card within this period, you must contact ASES Customer Service at 1-800-091-2737 or your Health Plan’s Customer Service at [xxx], If you do not agree with the assigned Primary Medical Group and/or Primary Care Physician, you have the right to request a change within 90 days from the date you received your ID card.
10
THIS IS YOUR ID CARD OF THE GOVERNMENT HEALTH PLAN

On the front of the card, you will find the following information:
| · | Your name and both last names; |
| · | Your contract number; |
| · | The group to which you belong; |
| · | Your coverages; and |
| · | Your Copayments and Coinsurances. |
Be sure that:
| · | You take your ID card with you when you visit your physicians, request laboratory or X- rays services or need health services. |
| · | They give you your ID card back after you receive medical services. |
| · | Each insured person in your family even if he/she is a baby, has his/her own ID card. |
| · | You keep your card in a safe place to avoid losing and having to wait for a new card. |
On the back of your ID card you will find the toll-free numbers for the call center of the Government Health Plan, Customer Service and the Mental Health Crisis helpline.
If you lose your card, you may request a duplicate by visiting your Health Plan’s Service Centers or by calling Customer Service at the number that appears on the back of your card.
11
|
IMPORTANT:
No Hospital can refuse emergency services for not having the Government Health Plan card. Under EMTALA you have the right to receive adequate emergency services, including evaluation and Treatment of an emergency condition or delivery in Hospital Emergency Rooms.
|
DISENROLLMENT
You may request Disenrollment from your Health Plan without cause during the ninety (90) calendar days following the Effective Date of Enrollment with the Health Plan or the date that the Health Plan sends you notice of the Enrollment, whichever is later. You may request Disenrollment without cause every twelve (12) months thereafter.
You may request Disenrollment from the Government Health Plan for cause at any time. The following constitute cause for Disenrollment by the Enrollee:
| 1. | The Enrollee moves to a Service Region not administered by the Health Plan, or outside of Puerto Rico; |
| 2. | The Enrollee needs related services to be performed at the same time, and not all related services are available within the General Network. The Enrollee’s PCP or another Provider in the Preferred Provider Network have determined that receiving service separately would subject the Enrollee to unnecessary risk; |
| 3. | Poor quality of care; or |
| 4. | Lack of Access to Covered Services, or lack of Providers experienced in dealing with the Enrollee’s health care needs. |
ASES shall make the final decision on Enrollee requests for Disenrollment. An Enrollee wishing to request Disenrollment must submit an oral or written request to ASES or to the Health Plan. If the request is made to the Health Plan, the Health Plan shall forward the request to ASES, within ten (10) Business Days of receipt of the request, with a recommendation of the action to be taken.
The following are acceptable reasons for the Health Plan to request Disenrollment:
| 1. | The Enrollee’s continued enrollment in the Government Health Plan seriously impairs the ability to provide services to either this particular Enrollee or other Enrollees; |
| 2. | The Enrollee demonstrates a pattern of disruptive or abusive behavior that is not caused by a presenting illness; |
| 3. | The Enrollee’s use of services constitutes Fraud, Waste or Abuse (for example, the Enrollee has loaned his or her Enrollee ID Card to other persons to seek services); |
| 4. | The Enrollee has moved out of Puerto Rico or out of the Health Plan’s Service Regions; |
12
| 5. | The Enrollee is placed in a long-term care nursing facility or intermediate care facility for the developmentally disabled; |
| 6. | The Enrollee’s Medicaid or CHIP eligibility category changes to a category ineligible for the Government Health Plan; or |
| 7. | The Enrollee has died or has been incarcerated, thereby making him or her ineligible for Medicaid or CHIP or otherwise ineligible for the Government Health Plan. |
If you are disenrolled from your Health Plan, you will lose access to services under the Government Health Plan.
RE-ENROLLMENT
If you are a Medicaid or CHIP member or member of the Commonwealth Population and you lose eligibility for the GHP for a period of less than two (2) months, you will be re-enrolled in your Health Plan.
FRAUD, WASTE AND ABUSE
What is Fraud?
Fraud affects adversely insured beneficiaries, health plans and professionals and entities that render health services. Fraud refers to any intentional and deliberate act to deprive another of property or money through deception or any other unfair action. It is done with the purpose of deceiving or making false misrepresentation with the purpose of obtaining a personal benefit or to benefit another person.
You have the responsibility of reporting any situation you understand may involve fraud against the Medicaid Program. Some examples of fraud are:
| · | Billing for medical services or procedures not actually performed. |
| · | Billing for supplies or medications not dispensed. |
| · | Lending an ID card to someone who is not entitled to it (misrepresentation) to obtain clinical services or medications. |
| · | Billing for a more costly payment than the one actually performed to obtain a higher payment. |
| · | Submitting false documents to obtain reimbursements. |
| · | Billing for the same service more than once. |
| · | Providing false information in a health enrollment form. |
| · | Billing for the dispensing of full Prescription when the Prescription was actually filled partially. |
| · | Receiving services rendered by a provider that has been excluded from the Medicaid Program. |
| · | Receiving reimbursement for services that are not medically necessary or that do not comply with the health care professional standards. |
13
It is important that any illegal or fraudulent action be reported immediately to your Health Plan’s Complaint Unit, the Patient’s Advocate Office or to ASES at the telephone numbers or email addresses included in page [5] of this Handbook.
What is Abuse?
Abuse is the excessive and improper use of a product, service or benefit, which results in unnecessary or excessive costs for the health care system.
Some examples are:
| · | Overuse of services that are not medically necessary, such as the constantly using the emergency room instead of going to the Primary Care Physician. |
| · | Excess in the orders for diagnostic tests that do not have a medical justification. |
| · | Waiving health plan Copayments or Coinsurances to attract customers. |
What is Waste?
Waste is the overutilization of services, misuse of resources or other practices that, directly or indirectly, result in unnecessary costs.
Some examples are:
| · | Prescribing high cost medications instead of similar generic or lower cost medication. |
| · | Billing errors due to inefficient billing systems. |
| · | Inflated prices on services or devices. |
What can I do to avoid Fraud, Waste and Abuse?
| · | Protect your ID card information: never provide information on your Health Plan to strangers or to callers by phone. |
| · | Learn the terms of your coverage and keep a copy of the medical studies to avoid duplicating services. If you visit a doctor, keep a copy of your laboratory results and other tests performed and have on hand a list of the medications you are taking. In this way you will not have to repeat tests that will consume time and money. |
| · | Verify the information before signing any insurance enrollment form or health service form. |
| · | Request and review the quarterly summary of the services you receive. You may request the summary of services directly to the Health Plan that provides you the Government Health Plan Services. |
14
How can I report situations on Fraud, Waste and/or Abuse?
If you have information or suspicion that you have been a victim of health plan fraud, you may contact your Health Plan through the call center of the Government Health Plan at the numbers that appear on the back of your ID card. You may also contact the Health Advocate Office at 787-977-0909 or, ASES at 1-800-981-2737 or by visiting your Health Plan, the Patient’s Advocate Office, ASES Offices or Customer Service Centers.
For more guidance on this matter you can access the orientation on Fraud, Waste and Abuse section on the ASES website at www.ases.pr.org. From this section of the website you can download the incident referral document which you can use to report any situation on Fraud, Waste and/or Abuse. Additionally, this section contains the contact information of all the agencies you can call to report any situation on Fraud, Waste and/or Abuse. Upon completing ASES’s incident referral document you have the option of indicating that you do not want to be contacted and/or remain anonymous. Similarly, you can also access your Health Plan’s website which also contains a Fraud, Waste and Abuse orientation section with all the previously described information.
Your call or written communication will be handled confidentially and your Government Health Plan Coverage will not be affected by this referral. If the investigation shows that Fraud, Waste or Abuse was committed, the case will be referred to the corresponding authorities.
PRIMARY MEDICAL GROUP AND PRIMARY CARE PHYSICIAN
Can I change my Primary Medical Group or the Primary Care Physician?
Yes, you may change your Primary Medical Group or your Primary Care Physician either by visiting your Health Plan’s Service Center or by calling your Health Plan’s Customer Service Line at [xxx-xxx-xxxx].
Changes to the Primary Medical Group and Primary Care Physician – You will only be able to change within the first 90 days following the date in which you received your Government Health Plan ID card. After this 90-day period, you may only change your Primary Medical Group and your Primary Care Provider once a year. If there is a Good Cause, you may change your Primary Medical Group or your Primary Care Physician at any time.
The following events are considered a Good Cause for a change:
| 1. | You move out of the region; |
| 2. | For moral or religious reasons, the provider does not render the services you need; |
| 3. | You need services that must be rendered at the same time and the services are unavailable. Not receiving all the services as ordered may put you at risk unnecessarily. |
| 4. | Other acceptable reasons include, but are not limited to: |
| a. | Bad quality of services; |
| b. | Lack of Access to Covered Services; and |
| c. | Lack of providers with experience to take care of your health care needs. |
ASES will determine if the reason is a Good Cause.
15
Changing the Primary Care Physician and the Primary Medical Group, (referred in previous paragraph) must be made during the first 5 days of the month, so the change becomes effective the next month (e.g. If you make the change on January 5, the change will be effective on February 1). However, if you change after the first 5 days of the month, the change will be effective on the subsequent month. (e.g. If you make the change on January 6, it will be effective on March 1).
To change the Primary Care Physician within the same Primary Medical Group, you only have to choose the new Primary Care Physician within same Primary Medical Group you have now and the change will be effective on the following month.
|
IMPORTANT:
The Medicaid Program is the only office authorized to make changes on your personal information and your residential address. You must notify the Medicaid Program of any changes such as changes in address, family group, marital status, your income, corrections to names, and dates of birth, among others.
|
Your Health Plan must keep you informed when a Primary Care Physician. Specialist or Sub-specialist is no longer available to be your medical service provider, so you can choose a new Primary Care Physician, Specialist or Sub-Specialist. You must receive the notice sent by your Health Plan within 15 days from the date your Health Plan was informed that the provider will not continue providing services. The notice the Health Plan provides to you will give you the instructions for you to be able to choose a new physician among those in your Primary Medical Group.
Choosing the Primary Medical Group and the Primary Care Physician
Remember that you have the freedom to choose the Primary Medical Group and the Primary Care Physician you want. If you do not agree with the assigned PMG and/or PCP made by your Health Plan, you can change. The Primary Medical Group and the Primary Care Physician you choose must render services with the region to which you belong.
You must choose a Primary Care Physician for each insured member in your family. The Primary Care Physicians you use for you and your dependents may be different, but they must belong to the same Primary Medical Group.
If you are a woman, you may choose a gynecologist/obstetrician in addition to any other Primary Care Physician. If you are pregnant, your Primary Care Physician will be your gynecologist/obstetrician during your pregnancy. When your pregnancy ends you will go back to receive care from the Primary Care Physician you chose: a Generalist, Internist, or Family Practitioner. Your gynecologist will still be your other Primary Care Physician to meet your gynecological situations. You may choose a pediatrician for your baby or one will be assigned to you.
|
IMPORTANT:
Remember, you must register your baby in the Medicaid Program before he is 90 days of age. You must bring with you the birth certificate.
|
16
RECERTIFICATION OF ELIGIBILITY
Once the Medicaid Program (PAM, for its acronym in Spanish) of the Health Department certifies you as eligible, you must attend all the appointments to all reevaluation appoints, so you don’t lose your eligibility. If you lose eligibility you will lose the benefits of the Government Health Plan, because you will not have the benefits of your Health Plan. Your Health Plan will send you a letter at 90 days, 60 days and 30 days before your eligibility ends as a reminder that you must visit the Medicaid Office in your hometown to recertify your eligibility.
If you miss your recertification appointment, you must immediately call the Medicaid Program Call Center at the toll-free number 1-885-400-4224 or visit your Medicaid Office located in your hometown to request a new appointment.
You must notify the Medicaid Program of any changes in address, income level, dependents, corrections to your address or name, or changes in marital status (married, divorced widower, etc.).
If you are pregnant, when you have your baby, you must visit the Medicaid Program Office and submit the birth certificate to enroll the baby in the Government Health Plan. If you do not comply with this requirement, the baby will lose the right to receive services under the Government Health Plan of Puerto Rico. It is possible that with the arrival of this new baby you can obtain more benefits if your level of poverty changes.
|
IMPORTANT:
Remember, it is your responsibility to keep appointments and update your information and mailing address with the Medicaid office in order to receive communications related to your recertification. If you do not receive the notification from your Health Plan, it is your responsibility to request the reevaluation appointment.
|
PUBLIC EMPLOYEE
If you are a public employee or a retiree from the Government of Puerto Rico and your payroll is process by the Treasury Department, you may enroll in the Government Health Plan during the open enrollment period to choose public employees health insurance plans. If you choose the Government Health Plan, the employer contribution will go to ASES and you will pay the difference, if any.
You can also visit the Medicaid Program for them to evaluate your case and, if found eligible and medically indigent, you will not have to pay the difference, if any, between the premium and the employer contribution as it will be paid with government funds.
17
Medical indigence is granted for a period of 12 months. Your plan will send you a letter 90 days, 60 days, and 30 days prior to the end of your eligibility period, reminding you that your eligibility is about to end and that you must visit your Medicaid Program Office located in your town of residence and request the reevaluation of your case.
In case of public employees that are married, they may enroll in the Government Health Plan combining both employer contributions (known as joint enrollment) for your eligibility. Your employer will provide the contributions to ASES, while you remain active and eligible under the Medicaid Program.
If after the evaluation, it is determined that you are no longer eligible to the Government Health Plan as medically indigent, you can enroll in the Government Health Plan as ELA Puro until the new health plan open enrollment period for public employees or you may enroll in any other health insurance plans contracted for public employees. It is your choice!
|
IMPORTANT:
Remember to attend your eligibility reevaluations on time, so you do not lose your Government Health Plan benefits.
|
What can I do if my eligibility in GHP is cancelled?
If the Medicaid Program determined that you are no longer eligible to the Government Health Plan, and you are an employee or retiree of the Government of Puerto Rico, you have the right to enroll in the Government Health Plan under ELA Puro within the 30 days following the date in which you lost your eligibility. In this way, you will not lose your medical coverage until the new government employee open enrollment period and you can choose any of the health plans contracted, including enrolling in the Government Health Plan.
If you are not an employee or retiree of the Government of Puerto Rico and you lose your eligibility, you may enroll in a Pago Directo Plan by submitting an application with your Health Plan. You must complete the formalities within 30 days from the date your eligibility to the Government Health Plan was cancelled.
How can I enroll in another of the plans contracted for government employees?
If you decide to join another plan from among the plans contracted for government employees according to Law 95, which is not the Government Health Plan, before you enroll in the new plan you will have to go to the Medicaid Program Office in your hometown to cancel your eligibility. The cancellation of your Government Health Plan coverage will be effective on the first day of the month following the date in which you requested your cancellation under the Medicaid Program.
If you do not cancel your eligibility to the Medicaid Program, ASES will continue receiving you employer contribution and you will have to pay the total premium of the Private Plan you chose.
|
IMPORTANT:
Remember that for you to be able to enroll in another plan, you must have lost your eligibility and may only enroll in another plan during the open health insurance enrollment period for the employees of the Government of Puerto Rico established by ASES.
|
18
Can the members of the Police Department of Puerto Rico enroll in the Government Health Plan?
The members of the Police Department of Puerto Rico, their spouses and children may also enroll in the Government Health Plan of Puerto Rico and the Police Department of Puerto Rico will transfer to ASES their employer contribution.
You must visit the Medicaid Program Office located in your town of residence to be certified under the Medicaid Program. This benefit will remain valid even if the member of the Police Department dies under any circumstance and as long as the widow does not re-marry and the children are under age 26 and are not married.
WHAT IS COORDINATED CARE?
The Government Health Plan uses a Coordinated Care model in which your health is under the care of a Primary Care Physician, who will be responsible to evaluate the beneficiary periodically and coordinate all the health services the person may need. Under this model your Primary Care Physician will keep an updated record of all the services you receive.
YOUR PRIMARY MEDICAL GROUP AND YOUR PRIMARY CARE PHYSICIAN
What is a Primary Medical Group?
Primary Medical Group (PMG) – is composed of several physicians who have joined to provide the services you need to keep you healthy. What was known as IPA, now it is known as PMG. Within this Group, there are physicians with different specialization which have been classified as Primary Care Physicians, among which there are:
| · | General Practitioners |
| · | Family Physicians |
| · | Pediatricians |
| · | Gynecologists/Obstetricians |
| · | Internists |
Besides these five categories of Primary Care Physicians, under the new model of the Government Health Plan you will also have Specialists, sub-specialists, laboratories, X-rays facilities and Hospitals, among others, to form what we call the Preferred Providers Network of the Primary Medical Group. You have the freedom to visit the physicians and providers that are part of the Preferred Network without the need of a Referral or Copayment.
19
|
IMPORTANT:
Routine physical exams shall be provided for Enrollees age twenty-one (21) and over within thirty (30) calendar days of the Enrollee’s request for the service.
Routine physical exams for minors less than twenty-one (21) years of age - an initial health and screening visit shall be provided to all newly enrolled Medicaid and CHIP Eligible children within ninety (90) calendar days and within twenty-four (24) hours of birth to all newborns; and, after the initial checkup, annually.
Routine evaluations for Primary Care shall be provided within thirty (30) calendar days, unless the Enrollee requests a later time.
Covered Services shall be provided within fourteen (14) calendar days following the request for service.
Specialist Services shall be provided within thirty (30) calendar days of the Enrollee’s original request for service.
Dental services shall be provided within sixty (60) calendar days following the request, unless the Enrollee requests a later date.
Behavioral Health Services shall be provided within fourteen (14) calendar days following the request, unless the Enrollee requests a later date.
Diagnostic laboratory, diagnostic imaging and other testing appointments shall be provided consistent with the clinical Urgency, but no more than fourteen (14) calendar days, unless the Enrollee requests a later time.
Appointment for urgent situations – as long there is not a risk of death or damage to the body or body organs, they must be obtained within a period of 24 hours.
These conditions must be treated at the medical office or offices with extended business hours, not at emergency rooms.
|
What is a Preferred Providers Network?
They are a group of Specialists, sub-specialists and health service facilities with a contract with your Health Plan to provide services under your Primary Medical Group. As long as you visit your Primary Medical Group Preferred Network, you will not have to wait for a Referral or pay Copayments.
The information below tells about some physicians and providers, without limiting to these specializations that may belong to the Primary Medical Group of your choice:
| · | Specialists and sub-specialists (including but not limited to Cardiologists, Orthopedists, Rheumatologists, Endocrinologists, Urologists, Gastroenterologist, Oncologists, Physiatrists). |
20
| · | Ancillary Service providers: physical therapists, nutritionists, speech pathologists, among others. |
| · | Clinical laboratories. |
| · | Specialized diagnostic tests. |
| · | Imaging centers. |
| · | Cardiovascular surgery and catheterism centers. |
| · | Hospitals. |
| · | Urgency rooms. |
| · | Emergency rooms. |
Another benefit you will now have under the Government Health Plan is that you will no longer need the countersignature of your Primary Care Physician on the Prescriptions ordered by any other physician that is not your Primary Care Physician, as long as the physician ordering the Prescription is part of the Preferred Network of your Primary Medical Group.
For laboratory and X-rays services you will need an order from the prescribing physician, but not the Authorization of your Primary Care Physician, as long as you receive the services at a laboratory or X-rays that belongs to the Preferred Provider Network of your Primary Medical Group.
The preferred networks will guarantee Access, quality and availability of the health services to be rendered to beneficiaries.
Are all my Specialists within the Preferred Network of my Primary Medical Group?
In case that the Specialist or sub-specialist that you need is not part of the Preferred Network of your Primary Medical Group, your Primary Care Physician must give you a Referral so you can visit the Specialists or sub-specialists outside the Preferred Network of your Primary Medical Group and you will have to pay corresponding Copayments. Your Primary Care Physician will be the one to coordinate the visits to physician and providers of medical services outside the Preferred Network of Providers of your Primary Medical Group.
You may visit Specialists or sub-specialists from your Health Plan’s General Network of Providers as long as your Primary Care Physician gives you the corresponding Referral and coordinates the visit, which will be subject to the applicable Copayments.
The Health Plan must cover FQHC services out-of-network at no cost to you for as long as FQHC services are unavailable in the Health Plan’s Preferred and General Network of providers. The out-of-network FQHC services require a Referral from your PCP and there is no applicable Copayment. If you wish to visit a Specialist or sub-specialist that does not belong to the Preferred Network of your Primary Medical Group, when there is a physician with the same specialty in the Preferred Network of the Primary Medical Group, you will also need a Referral from your Primary Care Physician and you will be responsible of paying the corresponding Copayment.
21
Referrals to visit a Specialist or other health provider, for either the general network or out of network services, must be provided during the same visit with the PCP but no later than 24 hours of the Enrollee's request.
Authorizations or Preauthorizations for services must be provided within 72 hours. If life or health could be endangered by a delay in Accessing services, Prior Authorizations must be provided as expeditiously as the Enrollee’s health requires, and no later than twenty-four (24) hours from the Service Authorization Request.
Non-compliance with these terms will be a reason to submit a Complaint. Nevertheless, if you are in the Special Coverage Registry you will not need Referrals from your Primary Care Physician, as long as the Treatment you are going to receive corresponds to your Special Coverage diagnosis.
|
IMPORTANT:
Your Primary Care Physician is the only authorized provider to give you the referrals you need for your health condition. The Administrator, the Medical Director or the Board of the Primary Medical Group cannot issue or authorize the referral.
If your Primary Care Physician does not provide you with the referral, you can request an Administrative Referral from your Health Plan by submitting a Complaint. Your Health Plan evaluates the Complaint or Grievance before proceeding with the final determination.
|
Your Health Plan will mail to you the Directory of the Providers of the Primary Medical Group and General Network. This information is also available on your Health Plan’s website.
What is your Health Plan’s General Network?
They are the Specialists, sub-specialists and health services facilities your Health Plan has contracted to provide support to the Primary Medical Groups. This General Network of your Health Plan will be available to provide those services you cannot obtain through the Preferred Network of your Primary Medical Group, as long as your Primary Care Physician gives you a Referral.
To be able to receive services from your Health Plan’s General Network, you must obtain a Referral from your Primary Care Physician and pay the corresponding Copayments. Prescription drugs or other service orders issued by your Health Plan’s General Network will need the countersignature or Authorization of your Primary Care Physician. That is, you will always have to go back to your Primary Care Physician for him/her to authorize the service ordered (laboratory, x-rays) and to countersign the Prescription of the medications for the pharmacy to be able to dispense them.
22
Will I need the Countersignature on the Prescriptions of Medications?
No participating pharmacy of the Government Health Plan can request the countersignature of the Primary Care Physician on Prescriptions ordered by Specialists or sub-specialists that belong to the Preferred Network of the Primary Medical Group.
If the Prescription of medications is from a Specialist or sub-specialist that belongs to your Health Plan’s General Network or the Preferred Network of another Primary Medical Group that is not the Primary Medical Group you chose, you will need the countersignature of Primary Care Physician for the Prescription to be dispensed.
Remember, you must visit the Specialists and sub-specialists within the Preferred Network of your Primary Medical Group, so you will not need the countersignature of your Primary Care Physician.
|
IMPORTANT:
Remember to use the Specialists and sub-specialists within the Preferred Network of your Primary Medical Group, so you do not need the countersignature of your Primary Care Physician on your Prescriptions.
|
KNOW THE RESPONSIBILITY OF YOUR PRIMARY CARE PHYSICIAN
Your Primary Care Physician is responsible to:
| · | Perform medical assessments relevant to your health. |
| · | Provide, coordinate and manage all health services and Treatments that you and your family need. |
| · | Provide preventive health services to keep you healthy. |
| · | Provide care when you feel or are sick. |
| · | Tell you when he believes it is necessary that you visit a Specialist or sub-specialist. |
| · | Provide Referrals when necessary, if you should visit a Specialist or sub-specialist outside of the Preferred Network of Primary Medical Group or when you want a Second Opinion. |
| · | Coordinate visits to Specialists or sub-specialists outside the Preferred Network of the Primary Medical Group. |
| · | Provide the Prescriptions for your medications or the orders for your Treatments. |
| · | Keep your Medical Record updated with all the information on your health conditions, medications, Treatments, etc. |
| · | Consult with other health professionals about your diagnosis and Treatment. |
Call or visit your Primary Care Physician every time you need medical services.
23
HOW TO OBTAIN INFORMATION ABOUT PARTICIPATING PHYSICIANS
Your Health Plan will mail you the Directory of Participating Physicians and Providers that are part of the Preferred Network of your Primary Medical Group, which also includes the Medical Groups that belong to the Region. You will also receive the Directory of your Health Plan’s General Network Physicians and Providers. These Directories will also be available in the Primary Medical Groups and at your Health Plan’s Service Centers. The directories provide the following information about the physicians:
| · | Medical Specialty |
| · | Name |
| · | Address |
| · | Telephone numbers |
| · | Office days and business hours |
You can contact your Health Plan to receive information on the providers available in your Region at the telephone numbers that appear on the back of your ID card, calling the Government Health Plan call center, going to your Health Plan’s office or through your Health Plan’s website. You may also contact your Primary Medical Group, which will provide information on the providers that belong to your Primary Medical Group.
When you contact your Health Plan, you can request additional information on your providers such as, where the physician studies, what did he studied, certifications of specialties the physician has, as well as all the information required to practice medicine.
THESE ARE YOUR RIGHTS
| · | You have the right to demand to be kept informed and receive information about: |
| o | your Health Plan, |
| o | health care facilities, |
| o | health care professionals, |
| o | health services covered, and |
| o | Access to contracted services; |
| · | The right to be treated with respect and with due regard for your dignity and privacy; |
| · | Select freely your Primary Medical Group, your Primary Care Physician, laboratory, X-rays, Hospital, Specialist and sub-specialists available within the Preferred Network of Primary Medical Group; |
| · | Contact your Primary Care Physician or Specialist, freely and under strict confidentiality; |
| · | Be free to receive emergency services 24 hours a day, 7 days a week; |
24
| · |
Receive information about Treatment alternatives and options available and, that these alternatives and options be presented to you in a manner appropriate to your condition and ability to understand;
|
| · | Participate in decisions regarding your health care, including the right to refuse Treatment; |
| · | Request a Second Opinion if you are interested in confirming a diagnosis or Treatment plan; |
| · | Express with Advance Directives, either verbally or in writing, your wish as to what Treatment and services you want to be provided or do not want to be provided if you become unable to make such decisions; |
| · | Be free from any form of restraint or seclusion used as a means of limitation, discipline, convenience or retaliation; |
| · | Receive copies of your Medical Records; |
| · | Receive high quality services; |
| · | Continuity of health care; |
| · | Access to adequate health services; |
| · | Filing Complaints and appeals, when you understand that your rights have been violated by denial of, limitation of or, improper collection for services; |
| · | Not to be discriminated against for any reason; |
| · | Have the freedom to choose the pharmacy or dentist of your preference among those contracted by your Health Plan; |
| · | Choose an Authorized Representative to be involved as appropriate in making care decisions; |
| · | Provide informed consent; |
| · | Be free from harassment by your Health Plan or its Network Providers with respect to contractual disputes between the Health Plan and its Providers; |
| · | Participate in understanding physical and behavioral health problems and developing mutually agreed upon Treatment goals; |
| · | Not be held liable for: |
| o | The Health Plan's debt in the event of insolvency; |
| o | Covered Services provided to you for which ASES does not pay the Health Plan; |
| o | Covered Services provided to you for which ASES or the Health Plan does not pay the provider that furnished the services; |
| o | Payments of Covered Services under a contract, Referral or other arrangement to the extent that those payments are in excess of the amount you would owe if the Health Plan provided the services directly; and |
| · | Only be responsible for cost-sharing or co-pays as permitted by the Puerto Rico which are applicable to you. |
25
THESE ARE YOUR RESPONSIBILITIES
| · | Inform yourself about the Government Health Plan Coverage, its limits and exclusions. |
| · | Give your physician all your health-related information. |
| · | Inform your doctor of any changes in your health. |
| · | Follow the medical Treatment as recommended by your Primary Care Physician, Specialist or sub-specialist. |
| · | Inform your physician when you do not understand an instruction or does not clearly understand what you are being inform. |
| · | Inform your physician when there is a reason why you cannot comply with the recommended Treatment. |
| · | Recognize when you need to make changes to your lifestyle to benefit your health. |
| · | Participate in any decision regarding your health. |
| · | Communicating either verbally or in writing any Advance Directive you want to be fulfilled regarding your decision on medical Treatment for the extension of your life. |
| · | Maintain appropriate behavior, so your behavior does not affect or does not allow other Patients to receive necessary medical care. |
| · | Maintain an appropriate behavior, so your behavior does not affect the operation of your Health Plan’s Service Centers or prevent other beneficiaries from receiving the services provided at the Service Centers. |
| · | Provide all the information on other health insurance plans you may have. |
| · | Inform ASES of any fraud or improper action related to the services, providers and health facilities. |
QUALITY AND PERFORMANCE INDICATORS
Puerto Rico Health Insurance Plan developed a series of quality and performance indicators as part of its quality improvement process. The quality and performance indicators are part of its Clinical and Mental Health Quality Program. The focus areas in the Clinical Quality Program are:
| · | Prenatal care services provided by your doctors; |
| · | Health education on wellness and prevention programs; |
| · | Care management support on severe acute medical condition; |
| · | Disease Management support on chronic medical condition such as diabetes or hypertension; |
| · | Education and support to your doctors to provide a better medical care; and |
| · | Scorecard on preventive benefits level achieved by your Health Plan. |
Some of the focus areas in the Mental Health Program are:
| · | Prenatal care services provided by your provider, such as: |
26
| o | Screening for alcohol and tobacco for pregnant women |
| o | Smoking cessation counseling and Treatment |
| · | Screening of postpartum depression; |
| · | Care Management for Enrollees diagnosed with serious mental illness or serious emotional disability; and |
| · | Education and support to your provider to provide a better mental health care. |
In addition, a series of service satisfaction indicators are measured on a yearly basis. You may be asked to participate on a survey that helps your Health Plan improve the quality of care and services delivered to you. Among the quality of service measures reported by your Health Plan are the following:
| · | How easy is it for you to receive the medical care that you need? |
| · | How quickly did you receive the medical care? |
| · | How is the customer service provided by your Health Plan? |
| · | How easy is it for you to receive your Prescription drugs? |
| · | How easy do you get the information you need about your medication drugs? |
| · | How well does your doctor communicate with you? |
For more information regarding Quality and Performance indicators please contact your Health Plan customer service to request it.
UTILIZATION MANAGEMENT POLICIES AND PROCEDURES
Utilization management is an evaluation of medical information to see if the service requested or recommended by your provider is necessary according to certain rules, known as clinical criteria or guidelines. The utilization management decisions are made by trained health professionals.
Your Health Plan offers clinical support through specialized programs that facilitate the Access and adequacy of services with the guarantee that services offered meet the highest standards of quality. These programs are: Disease Management, Preauthorization of Services and Case Management. Your Primary Care Physician (PCP) may decide to refer you to these programs if he/she believes you may benefit from them. For Covered Services that need Preauthorization, your Health Plan evaluates and issues a precertification of service once the care needs for the service is confirmed. If you have a chronic condition, such as Asthma, Diabetes, Hypertension or Congestive Heart Failure, you may benefit from participating in the Health Plan’s Disease Management Program. When your health conditions require Treatment and management considered complex, or Treatments needs to be provided in the home setting, your Primary Care Physician may refer you to the Case Management Program for assistance and coordination of your care needs. For more information please contact your Health Plan’s Customer Service.
27
The Health Plan also has a staff of trained mental health professionals to make decisions concerning utilization management. Some of the services that require an evaluation (Preauthorization) by the utilization management department staff before you get them are:
| · | Hospitalizations at a psychiatric Hospital; |
| · | Partial hospitalizations; |
| · | Some medications; and |
| · | Some specialized Treatments (e. g. electroconvulsive therapy). |
For more information please contact your Health Plan’s Customer Service.
EMERGENCIES AND URGENCIES
How do I know when it is an emergency?
“It is a medical or Behavioral Health condition that manifests itself by acute symptoms of sufficient severity (including severe pain) that a prudent layperson who has average knowledge of medicine and health would reasonably expect the absence of immediate medical attention to result in placing a person’s health in serious jeopardy, serious impairment of bodily functions or serious dysfunction of any bodily organ or part, serious harm to self or other due to an alcohol or drug abuse emergency, serious injury to self or bodily harm to others. In case of a pregnant woman that has contractions it may be that there is not enough time to transfer her to any facility before delivery or, that transferring her to a facility, may seriously jeopardize her health or the health of the unborn child.
When can I receive emergency services?
You just have to arrive at any emergency room throughout Puerto Rico. You do not need Referrals or Preauthorization for emergency services.
You can also call the Government Health Plan at the toll-free number listed on the back of your Government Health Plan ID card. When you contact the call center of the Government Health Plan for information and medical advice, you will be provided a code, so you do not have to pay Copayments if you had to go to an ER. No Co-Payment will be imposed on a Medicaid or CHIP Eligible Enrollee for Treatment of an Emergency Medical Condition or Psychiatric Emergency (regardless of whether the Enrollee uses the Medical Advice Service and gets the code or not).
And then, what is an Urgency?
A medical condition that poses no risk of imminent death that can be treated in the doctor's office or in the facilities with extended hours and not in emergency rooms. An Urgency can become an emergency if not properly dealt with at the right time.
28
How can I receive Urgency services?
Visit or call your Primary Care Physician. If you have an Urgency or a question about your health, you may call toll-free to the Government Health Plan hotline for medical information and advice. The telephone to this hotline, which is available 24 hours a day, 7 days a week, appears on the back of your Government Health Plan ID card.
How can I receive services outside business hours from my Primary Care Physician, the Primary Medical Group or the Preferred Network of Providers?
You must consult the Directory of Providers your Health Plan provided you, to learn about the business hours of your physicians. In addition, the Directory gives you the number for the Government Health Plan call center, so you can receive information and advice regarding your health condition as well as how to obtain services on extended hours.
If you understand that it is necessary to go to an emergency room, nobody can stop that right. When you use the Government Health Plan call center for information and medical advice, they will provide you a code, so you do not have to pay Copayments if you need to go to an emergency room. They will have to give you the code, regardless of your condition. No Co-Payment will be imposed on a Medicaid or CHIP Eligible Enrollee for Treatment of an Emergency Medical Condition or Psychiatric Emergency (regardless of whether the Enrollee uses the Medical Advice Service and gets the code or not).
WHAT IS AN ADVANCE DIRECTIVE?
An Advance Directive is a written legal document which allows you to instruct your attending physician on your Treatment preferences in case there is a moment that you lose your capacity to approve the Treatment. The written document that states the Advance Directive is known as a living will.
The instructions regarding your Treatment may be stated before a lawyer, who will prepare a legal document with your instructions or before your attending physician with two witnesses, of legal age and legal capacity, who are not relatives.
Your physician can provide you information on how you can exercise your right to Advance Directives. In case you are confined in a Hospital, the staff from the Hospital Administration Office can provide you the necessary information and the forms you must you fill out to validate your Advance Directives. You may also contact the Senior Citizens Advocate Office at 787-721-6121, who provides information booklets on this topic.
For any Complaints concerning Advance Directives, you can file directly them with ASES or with the Puerto Rico Office of the Patient Advocate.
COMPLAINTS, GRIEVANCES AND APPEALS
What is a Complaint?
A Complaint is an expression of dissatisfaction about any matter that is resolved at the point of contact rather than through filing a formal Grievance (see below for what is a Grievance). Complaints are not Actions; please see below for what is an Action.
29
For example, you can make a Complaint for incidents related to, but not limited to:
| · | Problems getting an appointment, or having to wait a long time for an appointment; or |
| · | Disrespectful or rude behavior by doctors, nurses or other Health Plan clinic or Hospital staff. |
How can you file a Complaint?
You can call, write or visit your Health Plan’s Service Centers for them to take your Complaint. Your physician, a relative or a person authorized by you, can file the Complaint on your behalf. Your Health Plan’s staff can provide help to you to file your Complaint.
You or your authorized representative must file a Complaint within fifteen (15) calendar days after the date of occurrence that initiated the Complaint. The Health Plan shall resolve your Complaint within seventy-two (72) hours of receipt of the initial Complaint, whether orally or in writing. If the Complaint is not resolved within this timeframe, the Complaint shall be treated as a Grievance.
What is a Grievance?
A Grievance is a formal expression of dissatisfaction about any matter other than an Action that is documented and investigated by the plan. Grievances are not Actions; please see below for what is an Action.
The Grievance can be presented in writing, by telephone or by visiting any of your Health Plan’s Service Centers or the Health Advocate Office (OPS, for its acronym in Spanish). For example, you can file a Grievance for incidents related to, but not limited to:
| · | The quality of care or services provided |
| · | Access to care or services |
| · | Aspects of interpersonal relationships such as rudeness of a provider or employee, |
| · | Misinformation provided by the Health Plan or providers |
| · | Failure to respect the Enrollee's rights |
| · | Preauthorization requests |
| · | Network provider changes |
| · | Referrals |
| · | Hazardous environment conditions |
How can you file a Grievance?
You can call, write or visit your Health Plan’s Service Centers to file a Grievance. Your physician, a relative or a person authorized by you, can file the Grievance on your behalf.
30
You have up to ninety (90) calendar days from the date of the occurrence to file a Grievance with the Health Plan. The Health Plan shall acknowledge receipt of your Grievance in writing to you (and the provider, if the provider filed the Grievance on your behalf) within ten (10) business days of receipt.
The Health Plan shall provide written notice of how the Grievance is resolved as promptly as your health condition requires, but in any event, within ninety (90) calendar days from the day the Health Plan receives the Grievance.
What is an Action?
An Action is a decision that your Health Plan makes that may affect the services you receive, specifically, an Action is:
| · | The denial or limited Authorization of a requested service, including the type or level of service; |
| · | The reduction, suspension, or termination of a previously authorized service; |
| · | The denial, in whole or in part, of payment for a service; or |
| · | The failure to provide services in a timely manner. |
What is a Notice of Action?
A Notice of Action is a written notice provided by the Health Plan to you notifying you of an Action (as defined above). The Notice of Action must contain the following information:
| · | The Action the Health Plan has taken or intends to take; |
| · | The reasons for the Action; |
| · | Your right to file an Appeal through the Health Plan’s internal Grievance System and the procedure for filing an Appeal; |
| · | Your right to request an Administrative Law Hearing after exhaustion of the Health Plan’s Grievance System; |
| · | Your right to allow a Provider to file an Appeal or an Administrative Law Hearing on behalf of you, upon written consent; |
| · | The circumstances under which expedited review is available and how to request it; |
| · | Your right to continue receiving Benefits and Covered Services pending resolution of the Appeal with the Health Plan or during the Administrative Law Hearing; and |
| · | How you can request that Benefits be continued and the circumstances under which you may be required to pay the costs of these services. |
31
The Health Plan shall mail the Notice of Action within the following timeframes:
| · | For termination, suspension, or reduction of previously authorized Covered Services, at least ten (10) calendar days before the date of Action or no later than the date of Action except in the event of one of the following exceptions: |
| 1. | The Health Plan has factual information confirming the death of an Enrollee. |
| 2. | The Health Plan receives a clear written statement signed by the Enrollee that he or she no longer wishes to receive services or gives information that requires termination or reduction of services and indicates that he or she understands that this must be the result of supplying that information. |
| 3. | The Enrollee’s whereabouts are unknown and the post office returns the Health Plan’s mail directed to the Enrollee indicating no forwarding address. |
| 4. | The Enrollee’s Provider prescribes a change in the level of medical care. |
| 5. | The Health Plan may shorten the period of advance notice to five (5) calendar days before the date of Action if the Health Plan has facts indicating that Action should be taken because of probable Enrollee Fraud and the facts have been verified, if possible, through secondary sources. |
| · | For denial of payment, at the time of any Action affecting the Claim. |
| · | If the Health Plan extends the timeframe for the Authorization decision and issuance of Notice of Action the Health Plan shall give you written notice of the reasons for the decision to extend if you did not request the extension. The Health Plan shall issue and carry out its determination as expeditiously as your health requires and no later than the date the extension expires. |
What can I do if I do not agree with the Notice of Action?
If you do not agree with your Health Plan’s determination included in the Notice of Action, you have the right to appeal the determination before your Health Plan or the Health Advocate Office (OPS, for its acronym in Spanish) within sixty (60) calendar days from the date of the Notice of Action.
What is an appeal?
An appeal is a formal request that you file with your Health Plan or the Health Advocate Office when you do not agree with the determination (Notice of Action) or with the Health Plan’s denial of a service, procedure, study, collection or payment. Once you receive the Notice of Action from your Health Plan, you have a period of sixty (60) Calendar days to file your appeal with your Health Plan.
32
Who will hear your Appeal?
Your Appeal will be evaluated by a team of experts (specific for your health condition) that did not take part in the determination or in the Notice of Action. This is to ensure the appeal process is fair, transparent and dependable.
How much time will they take to make a determination on my Appeal?
If the Appeal does not adversely affect your health and/or does not put your life at risk, you must receive the determination of your appeal within a period that does not exceed forty-five (45) calendar days. However, if your health condition requires an expedited determination; you will receive an answer within a period of three (3) business days or less.
Your Health Plan can request a 14-day extension to send its determination, as long as this extension request benefits the beneficiary (you) or because you need more time to find evidence or data that may benefit your case.
Remember that if during the appeal process you request a continuation of services, you may be required to pay the cost of services furnished while the Appeal is still pending. This would be the case if the final decision is adverse to you.
What is an Administrative Law Hearing?
An Administrative Law Hearing is an appeal process that is available to you after you exhaust your Health Plan’s Complaint, Grievance and Appeals processes described above for an Action that your plan takes.
How can you request an Administrative Law Hearing?
If you are not satisfied with the outcome after you have gone through your Health Plan’s Complaint, Grievance and Appeal procedures for an Action, you can request an Administrative Law Hearing to ASES or the Health Advocate Office, or both, in a period not to exceed thirty (30) calendar days from the date of the notice of disposition of the Appeal that your Health Plan sends you.
The Administrative Law Hearing resolution will be ninety (90) calendar days of the date you file an Appeal with your Health Plan (not including the days it took you to file for an Administrative Law Hearing) for standard resolutions. For expedited resolution, the Administrative Law Hearing resolution will be within three (3) business days from ASES’s receipt of a request for a hearing for a denial of service.
Before the Administrative Law Hearing, you and your authorized representative, or a representative of a deceased Enrollee, if applicable, can ask to look at and copy the documents and records your Health Plan will use at the Administrative Law Hearing or that you may otherwise need to prepare your case for the hearing. Your Health Plan shall provide such documents and records at no charge to you.
If you receive an unfavorable decision at the Administrative Law Hearing, you may appeal the decision to the Court of Appeals of Puerto Rico.
33
TIME TO SOLVE COMPLAINTS, GRIEVANCES AND APPEALS
Below are timeframes your Health Plan has to address and resolve Complaints, Grievances, Appeals and ASES has to resolve issues brought before an Administrative Law Hearing:
| · | The Health Plan must resolve Complaints within 72 hours of receipt whether orally or in writing. If it is not resolved in 72 hours, it becomes a Grievance. |
| · | The Health Plan must send a written notice to Enrollees within ten (10) business days or receiving a Grievance. |
| · | The Health Plan must provide a written notice on the resolution of the Grievance as quickly as your health requires but no later than ninety (90) calendar days from the day the Health Plan receives the Grievance. |
| · | If the Health Plan denies a service Authorization request, it must provide a written Notice of Action to you and your provider. |
| · | You or your provider on your behalf can appeal the Notice of Action no later than sixty (60) calendar days after receiving the Notice of Action. |
| · | The determination on standard Appeals must be sent to the affected parties within a period that does not exceed 45 days. Your Health Plan may request a 14-day extension, as long as it is for your benefit. |
| · | Determination on expedited Appeals will always depend on your health condition and may not exceed three (3) business days. The Health Plan may request a 14-day extension as long as it is for your benefit. |
| · | You may request an Administrative Law Hearing before ASES thirty (30) days from the date you received notification of determination on the appeal. |
| · | The Administrative Law Hearing resolution will be ninety (90) calendar days of the date you file an Appeal with your Health Plan (not including the days it took you to file for an Administrative Law Hearing) for standard resolutions. For expedited resolution, the Administrative Law Hearing resolution will be within three (3) business days from ASES’s receipt of a request for a hearing for a denial of service. |
DENTAL SERVICES
Dental services are free choice services and do not need Referrals, that is, you can visit the dentist whenever you need dental services. You can visit your dentist as you have always done, as long as they are participating dentists of the Government Health Plan.
The information on participating dentists is included in the Directory of Contracted Providers which your Health Plan will mail to you and which is available at your Health Plan’s website. Dentists are not part of the Preferred Networks.
34
MENTAL HEALTH SERVICES
How can I receive mental health services or services against drug dependence?
Mental health services and services against substance abuse will be provided through your Health Plan. To receive these services you do not need a Referral from your Primary Care Physician, you may request these services for yourself when you feel it is necessary.
The Government Health Plan offers integrated mental health and physical health services. Under the Government Health Plan you can receive mental health services at the same facility where you visit your Primary Care Physician; and primary care health services at the same facility where you visit your mental health provider.
This means that when either your Primary Care Physician or your mental health provider detects that you need mental or physical health services, he/she does not have to send you to another office to receive the services. The psychologist and/or social worker will be physically at your PMG at least sixteen (16) hours per week during regular business hours.
In the same way, a PCP will be physically present at your mental health clinic at least sixteen (16) hours per week at Ambulatory Clinics and twelve (12) hours per week at Addiction Services Units. Psychiatric Hospitals will have at least a PCP on call on a daily basis. Hospitalization Units will have at least one collocated PCP two (2) days per week for four (4) hours and Stabilization Units will have one PCP for Consultation (on call) on a daily basis.
When you fill out your enrollment form at your Health Plan’s Service Centers, among the materials you will receive there will be detailed information about the mental health services providers for your Region and how to obtain them when you need them. In addition, the Directory of Providers provided by your Health Plan will indicate the address and telephone numbers of the providers that render mental health services in your Region. For additional information regarding the services and benefits, you may refer to the Mental Health Coverage Section this Guide offers.
You may also contact the Government Health Plan helpline if you do not know where to go. The Government Health Plan helpline will provide you all the information you need or you may contact your Health Plan at the numbers that appear on the back of your card.
PREVENTIVE SERVICES
Your Government of Puerto Rico Health Plan offers you a variety of services under preventive services.
What are preventive services?
They are health care services offered to help you keep your health in optimal condition. If you have any condition, preventive services will help you have better knowledge of your condition, so you can keep it under control and will help prevent your health from getting worse. These services not only will help you understand your condition, but also will tell you what to do to keep yourself healthy. Refer to the Preventive Service Coverage found in this Guide, so you find out all the services covered under the Government Health Plan.
35
To keep your health in optimal conditions you should:
| · | Maintain healthy nutrition. |
| · |
Exercise, such as walking, at least 30 minutes 4 to 5 days a week.
|
| · | Avoid being overweight. |
| · | Be calm and in peace. |
| · | Take a few minutes daily to relax. This will help you reduce stress. |
| · | Get enough rest. |
| · | Do not smoke. |
| · | Do not use drugs or alcohol. |
| · | Visit or consult your doctor whenever you feel sick. |
Your Health Plan will provide the preventive services and additional services as required by the Government Health Plan. The Health Plan will provide you information in booklets that will be a part of these guidelines.
HIV-AIDS
If you are diagnosed with the Acquired Immunodeficiency Syndrome or the Human Immunodeficiency Virus (HIV), your Primary Care Physician must request that you be included in the Special Coverage Registry. Once the Health Plan includes you in the Special Coverage Registry, they will mail you a letter authorizing you to receive services under the Special Coverage. This letter will include information on the effective date and the expiration of this coverage.
This letter will allow you to Access all the services and Treatments for your condition without Referrals, countersignatures on your Prescriptions or Service orders for laboratory, X-rays services, among others, from your Primary Care Physician.
There are certain medications for your HIV/AIDS condition that will be provided by the Health Department, which may be acquired through the following Immunology Centers and Pharmacies:
36
Centers for the Prevention and Treatment of Communicable
Diseases (CPTET, for its acronym in Spanish)
Updated as of June 2014
|
REGION
|
MEDICAL DIRECTOR
|
TELEPHONE/FAX
|
ADDRESS
|
|
ARECIBO
|
Dr. Evelyn Reyes García
Director
Internist
|
(787) 878-7895
Fax. (787) 881-5773
Fax. Medical Director Office
Fax. (787) 878-8288
Tel. (787) 879-3168
|
Antiguo Hosp. Distrito
Carretera 129 hacia Lares
#627 Ave. San Luis
Arecibo, PR 00612-3666
|
|
BAYAMON
|
Dr. Odette García Viña
Director
Dr. Fco. Bellaflores
Internist
|
(787) 787-5151
Ext. 2224, 2475
(787) 787-5154 (d)
Fax. (787) 778-1209
(787) 787-4211
|
Antigua Casa de Salud
Hosp. Regional Bayamón
Dr. Ramón Ruíz Arnau,
Ave. Laurel Santa Juanita
Bayamón, PR 00956
|
|
CAGUAS
CLINICA SATELITE
HUMACAO
|
Dr. Gloria Morales
Director
General Medicine
|
(787) 653-0550
Ext. 1142, 1150
Fax (787) 746-2898
(787) 744-8645
(787) 640-0980
(787) 852-0665
|
Hosp. San Juan Bautista
PO Box 8548
Caguas, PR 00726-8548
Centro Comercial Humacao
Ave. Font Martelo 100
Humacao, PR
|
|
CAROLINA
|
Dr. Milton Garland
Director
Internist
|
(787)757-1800
Ext. 454, 459
Diirecto y Fax. (787)257-3615
|
Hospital UPR Dr. Federico Trilla
P. O. Box 6021
Carolina, PR 00984-6021
Carretera 3, Km. 8.3
|
|
CLETS
|
Dr. Hermes García
Director
|
(787)754-8118 ©
(787)754-8128 (directo)
(787)754-8127
Fax. (787)754-8199
|
P. O. Box 70184
San Juan, PR 00936-8523
Calle José Celso Barbosa
Centro Médico de PR
Bo. Monacillos, San Juan
|
|
FAJARDO
|
Dr. Arturo Hernández
Director
General Medicine
|
(787)801-1992
(787)801-1995
Fax. (787)863-5437
|
Calle San Rafael # 55
Fajardo, PR
Urb. Monte Brisas I
Suite 69 Calle E
Fajardo, PR 00738
|
|
MAYAGUEZ
|
Dr. Ramón Ramírez Ronda
Director
Infectologist
|
(787)834-2115, 2118
Ext. 4634
Fax Regional Director
(787)806-3440
|
Centro Médico de Mayagüez
Hospital Ramón Emeterio Betances Suite 6
Ave. Hostos 410
Antigua Casa de Salud
Mayagüez, PR 00680
|
|
PONCE
|
Dr. Gladys Sepúlveda
Director
Infectologist
|
(787)259-4731
(787)259-4046, (787)842-8626
Fax (787)259-3998
Fax pharmacy (787)843-2188
|
Departamento de Salud
Región Ponce
Antiguo Hosp. Distrito Ponce
Dr. José Gándara
Carretara Estatal 14
Bo. Machuelo
Ponce, PR 00731
|
|
NIVEL CENTRAL
|
Dr. Greduvel Durán
Executive Director
Services Director
OCASET
|
(787)765-2929
Ext. 4026, 4027
Fax (787)274-5523
|
P.O. Box 70184
San Juan, PR 00936
Ant. Hosp. Psiquiatría
Pabellón 1, primer piso, 4ta. Puerta - Terrenos de Centro Médico, Río Piedras
|
37
|
IMPORTANT:
The Special Coverage is authorized for a specific time period. When this period expires, your physician must justify any extension that is medically necessary for your condition and will have to request your registration in the Special Coverage again.
|
HEPATITIS-C
If you are diagnosed with Hepatitis C, once laboratory tests are performed confirming that you have been infected with this disease, all the services and Treatments will be provided through the Health Department. Your Primary Care Physician must inform your diagnosis to your Health Plan’s Disease Management Program, for your Health Plan to provide you information and coordinate your enrollment in the Hepatitis C Program of the Health Department.
This is your Benefits Coverage
The Government Health Plan offers a broad Service Coverage with minimum exclusions. Your services will not be reduced, limited or will be excluded because you had a preexisting conditions before enrolling in the Government Health Plan. You will not have to comply with a waiting period to receive any of the Covered Services. Services will be covered from the moment Medicaid grants your eligibility. Services will be provided if medically necessary. Medically necessary means:
Services related to (i) the prevention, diagnosis, and Treatment of health impairments; (ii) the ability to achieve age-appropriate growth and development; or (iii) the ability to attain, maintain, or regain functional capacity. Additionally, Medically Necessary services must be:
| · | Appropriate and consistent with the diagnosis of the treating provider and the omission of which could adversely affect your medical condition; |
| · | Compatible with the standards of acceptable medical practice in the community; |
| · | Provided in a safe, appropriate, and cost-effective setting given the nature of the diagnosis and the severity of the symptoms; |
| · | Not provided solely for your convenience or the convenience of the Provider or Hospital; and |
| · | Not primarily custodial care (for example, foster care). |
In order for a service to be Medically Necessary, there must be no other effective and more conservative or substantially less costly Treatment, service, or setting available.
The information that follows details all the services covered.
38
Preventive Services
| · | Vaccines – Provided by the Health Department. The Government Health Plan will cover the administration of the vaccines following the dates established in the schedule provided by the Health Department. |
| · | Healthy Child Care - during the child’s first 2 years of life. |
| · | Healthy Child Care - One comprehensive annual assessment performed by a certified health professional. This annual assessment supplements the services for children and young adults is provided during the period established in the schedule of the American Academy of Pediatrics and Title XIX (EPSDT). |
| · | Vision test. |
| · | Hearing exam, including the newborn hearing screening before they are released from the Hospital nursery. |
| · | Nutritional evaluations and tests. |
| · | Laboratory tests and all the diagnostic and screening tests according to the beneficiary’s age, sex and health condition. |
| · | Prostate and gynecologic cancer screening according to the accepted medical practices, including Papanicolau, mammography and PSA tests when medically necessary and according to the age of the beneficiary. |
| · | Puerto Rico public policy sets the age of 40 years as a starting point for mammograms and breast cancer screening. |
| · | Sigmoidoscopy and colonoscopy to detect colon cancer in adults aged 50 or more, classified by risk group, according to the accepted medical practices. |
| · | Education on physical, nutritional and oral health. |
| · | Reproductive Health Counseling (Family Planning). Such services shall be provided voluntarily and confidentially, including circumstances where the Enrollee is under age eighteen (18). Family planning services will include, at a minimum, the following: |
| o | Education and counseling necessary to make informed choices and understand contraceptive methods; |
| o | Pregnancy testing; |
| o | Diagnosis and treatment of sexually transmitted infections; |
| o | Infertility assessment; |
| o | At least one of every class and category of FDA-approved contraceptive medication as specified in ASES’s preferred drug list (PDL); and |
| o | At least one of every class and category of FDA-approved contraceptive method as specified by ASES. |
| o | Other FDA approved contraceptive medications or methods when it is Medically Necessary and approved through a Preauthorization or through an exception process and the prescribing provider can demonstrate at least one of the following situations: |
39
| § | Contra-indication with drugs that are in the PDL that the Enrollee is already taking, and no other methods available in the preferred drug list that can be used by the Enrollee. |
| § | History of adverse reaction by the Enrollee to the contraceptive methods covered as specified by ASES; or |
| § | History of adverse reaction by the Enrollee to the contraceptive medications that are on the preferred drug list. |
| · | Syringes for the administration of medications at home. |
| · | Health certificates covered under the Government Health Plan (any other health certificates are excluded). |
| · | Health Certificates that include tests for sexually transmitted diseases (VDRL) and tuberculin tests. The certificate must have the seal of the Health Department with a Copayment that will not exceed $5.00. The PR Department of Health charges a nominal administrative fee of $5.00 for the certificate. This is not a co-payment to receive the service or the results. |
| · | Any certification for the Government Health Plan beneficiaries related to the Medicaid and CHIP Program eligibility (e.g. Medications History) will be provided to the beneficiary free of charge. |
| · | Any Copayment that applies to necessary procedures and laboratory tests for the issuance of a Health Certificate will the responsibility of the beneficiary. |
| · | Annual physical exam and follow-up to diabetic Patients according to Treatment guidelines for the Treatment of diabetic Patients and the protocols of the Health Department. |
Dental Services
You may visit the dentist of your choice that accepts the Government Health Plan. Covered dental services will be identified using the codes published by the American Dental Association (ADA) for the procedures established by ASES. The services that follow are covered under the Government Health Plan:
| · | Preventive services for children. |
| · | Preventive services for adults. |
| · | Restorative services. |
| · | A comprehensive oral exam. |
| · | A periodic oral evaluation every 6 months. |
| · | Limited oral evaluation- problem focused. |
| · | Intraoral X-rays complete series, including bitewings, every 3 years. |
| · | One intraoral/periapical first film. |
| · | Up to a maximum of 5 additional intraoral/periapical X-rays a year. |
| · | Bitewing single film a year. |
| · | One Bitewings double film a year. |
| · | One set of panoramic film every 3 years. |
| · | Prophylaxis – adult, every 6 months. |
40
| · | Prophylaxis – children, every 6 months. |
| · | Topical fluoride application for children under age 19, every 6 months. |
| · | Topical application of sealant, per tooth, on posterior teeth for beneficiaries up to 14 years old. Includes deciduous molars up to 8 years of age when it is medically necessary because of a tendency to cavities. This service is limited to one lifetime Treatment. |
| · | Resin composite restorations. |
| · | Amalgam restoration. |
| · | Pediatric therapeutic pulpotomy. |
| · | Stainless steel crowns for primary teeth followed by a pediatric therapeutic pulpotomy. |
| · | Root canals. |
| · | Palliative Treatment. |
| · | Oral surgery. |
Diagnostic Testing Services
| · | High tech laboratories. |
| · | Clinical laboratories including, but not limiting to, any laboratory order with the purpose of diagnosing the disease, even if the diagnosis is an excluded condition or disease. |
| · | X-rays. |
| · | Radiotherapy. |
| · | Electrocardiograms. |
| · | Pathology. |
| · | Arterial blood gases. |
| · | Electroencephalograms. |
Ambulatory Rehabilitation Services
| · | Physical therapy – a minimum of 15 physical therapy Treatments a year per condition, per beneficiary, when prescribed by an orthopedist or a physiatrist; unless Preauthorization of an additional fifteen (15) Treatments is indicated by an orthopedist, physiatrist or chiropractor. |
| · | Occupational therapy – unlimited. |
| · | Speech therapy – unlimited. |
Medical and Surgical Services
| · | Visits to primary care providers, including Primary Care Physicians and nursing services. |
| · | Treatments by Specialists and sub-specialists, without Referral, if they belong to the Preferred Provider Network of your Primary Medical Group. |
41
| · | Treatments by Specialists and sub-specialists outside the Preferred Provider Network of the Primary Medical Group with a Referral of the Primary Care Physician you chose. |
| · | Physician home visits when it is medically necessary. |
| · | Respiratory therapy, without limits. |
| · | Anesthesia services, except epidural anesthesia. |
| · | Radiological services. |
| · | Pathology services. |
| · | Surgery. |
| · | Use of ambulatory surgery facilities. |
| · | Diagnostic services for cases of learning disabilities. |
| · | Practical nurse services. |
| · | Voluntary sterilization for men and women of appropriate age after being previously informed on the consequences of the medical procedure. The physician must have the written consent of the Patient. |
| · | Prosthesis: includes the supply of all body extremities including therapeutic ocular prosthesis, segmented instrument tray and spinal fusion in scoliosis and vertebral surgery. |
| · | Ostomy equipment for Patients ostomized ambulatorily. |
| · | Blood, plasma and their derivatives. |
| · | Services to Patients with chronic kidney disease in the first two levels (levels 3 to 5 are included in the Special Coverage). |
| · | Breast reconstruction surgery after a mastectomy because of cancer. |
| · | Treatments and surgery in cases of morbid obesity. |
| · | Abortions are covered in the following instances: (i) life of the mother would be in danger if the fetus is carried to term; (ii) when the pregnancy is a result of rape or incest; and (iii) severe and long lasting damage would be caused to the mother if the pregnancy is carried to term, as certified by a physician. |
| · | Durable medical equipment (DME) is covered on a case-by-case basis with Preauthorization as Medically Necessary. Mechanical respirators and ventilators with oxygen supplies are covered without limits as required by local law to Enrollees under age twenty-one (21).) |
Ambulance Services
| · | Sea, air and land transportation will be covered within Puerto Rican territory limits in cases of emergency. These services do not require Preauthorization or precertification. |
Non-Emergency Transportation Services (NEMT)
| · | Each Municipality in Puerto Rico has a variety of free transportation services available to assist you in getting to your medical appointments. You can access the service by contacting your local Municipal office or your Health Plan and asking about how to obtain transportation services the transportation. |
42
| · | The Health Plans and some providers do offer transportation for members with certain conditions through case management. If you need the help of a case manager and you do not have one, call your Health Plan. |
Maternity and Prenatal Services
| · | Women have the freedom to choose a gynecologist/obstetrician among the providers of the Primary Medical Group or from your Health Plan’s General Network or any gynecologist/obstetrician, subject to the final coordination with the provider. The different interventions until the confirmation of the pregnancy are not part of this coverage. |
| · | Pregnancy tests. |
| · | Pre-natal services. |
| · | Services of the physician and an obstetric nurse during a normal delivery, c-section and in any other complication that may arise. |
| · | Maternity hospitalization or for pregnancy secondary conditions, when medically recommended. |
| · | Hospitalization of at least 48 hours for the mother and the newborn in case of a vaginal delivery and of 96 hours in case of c-section. |
| · | Anesthesia, except epidural anesthesia. |
| · | Use of incubator, unlimited. |
| · | Nursery room care for the newborn. |
| · | Circumcision and dilatation services for the newborn. |
| · | Transportation of the newborn to tertiary facilities. |
| · | Assistance of a Pediatrician during a c-section or high risk delivery. |
Emergency Room Services
You do not need a Preauthorization or a precertification to receive these services.
| · | Visits, medical attention, routine emergency room necessary services. |
| · | Services for trauma. |
| · | Use of emergency room and surgery. |
| · | Necessary and routine emergency room services. |
| · | Respiratory services, without limitations. |
| · | Treatment by a Specialist or a sub-specialist when requested by the emergency room physician. |
| · | Anesthesia, excluding epidural anesthesia. |
| · | Surgical supplies. |
| · | Clinical laboratory tests. |
43
| · | X-rays. |
| · | Drugs, medications and intravenous solutions to be used in the emergency room. |
| · | Blood, plasma and their derivatives, without limitations. |
Emergency services outside Puerto Rico will be covered only for the Federal Population according to non-participating providers’ fees in Puerto Rico.
Post-Stabilization Services
| · | Post-Stabilization Services are services that are provided after the Enrollee is stabilized to maintain or improve the Enrollee’s condition after experience an emergency medical condition or psychiatric emergency for one hour while awaiting responses on a Preauthorization request. |
| · | The attending Emergency Room physician or other treating Provider shall be responsible for determining whether the Enrollee is sufficiently stabilized for transfer or discharge. That determination will be binding for the Health Plan with respect to its responsibility for coverage and payment. |
| · | An Enrollee who has been treated for an Emergency Medical Condition or Psychiatric Emergency shall not be held liable for any subsequent screening or Treatment necessary to stabilize the Enrollee. |
Hospitalization Services
| · | Semi-Private Room, available 24 hours a day, year round. |
| · | Isolation room for medical reasons. |
| · | Nursery. |
| · | Meals, including specialized nutrition services. |
| · | Regular nursing services. |
| · | Use of specialized rooms such as surgery room, recovery room, Treatment and delivery room, without limitations. |
| · | Drugs, medications and contrast agents, without limitations. |
| · | Materials such as bandages, gauze, plaster bandages or any other therapeutic dressing materials. |
| · | Therapeutic and maintenance care services, including the use of the necessary equipment to render the service. |
| · | Specialized diagnostic tests such as electrocardiograms, electroencephalograms, arterial blood gases, and other specialized test available at the Hospital and necessary during the beneficiary’s hospitalization. |
| · | Supply of oxygen, anesthesia and other gases, including their administration. |
| · | Respiratory therapy, without limitations. |
| · | Rehabilitation services while the Patient is confined in the Hospital, including physical, occupational and speech therapy. |
| · | Blood, plasma and their derivatives, without limitations. |
44
Mental Health Services
| · | Evaluation, screening and Treatment to individuals, couples, families and groups. |
| · | Ambulatory services rendered by psychiatrists, psychologists and social workers. |
| · | Hospital and ambulatory services for substance abuse and alcoholism. |
| · | Intensive ambulatory services. |
| · | Emergency and crisis intervention services available 24 hours a day, 7 days a week. |
| · | Detoxification services for beneficiaries that use illegal drugs, have had suicidal attempts or accidental poisoning. |
| · | Administration of and Treatment with Buprenorphine (requires Preauthorization). |
| · | Clinics for injectable extended-release medications. |
| · | Escort, professional assistance and ambulance services when the services are necessary. |
| · | Prevention services and secondary education. |
| · | Pharmacy coverage and Access to medications within 24 hours. |
| · | Laboratory tests that are medically necessary. |
| · | Treatment for Patients diagnosed with Attention Deficit Disorder (ADD) with or without hyperactivity (ADHD). This includes, but is not limited to, visits to neurologists and tests related to the Treatment of this diagnosis. |
| · | Consultations and coordination with other Agencies. |
| · | Substance abuse Treatment. |
Mental Health Hospitalization Services
| · | Partial hospitalization services for cases referred by a psychiatrist for primary phase diagnosis and Treatment, according to the parity provisions of Law 408 of October 2, 2000. |
| · | Hospitalization that presents a mental pathology that is not drug abuse when referred by a psychiatrist for primary phase diagnosis and Treatment, according to the parity provisions of Law 408 of October 2, 2000. |
Pharmacy Services
| · | The GHP has Prescription drug coverage for the Physical and Mental Health needs of beneficiaries established in the Preferred Drug List (PDL). |
| · | The pharmacy benefit coverage is generic-bioequivalent mandatory as general rule. |
| · | Copayments are required for prescribed medication covered by the GHP. |
| · | No co-payments will be charged to Medicaid and CHIP children under eighteen (18) years of age, and pregnant women. . |
| · | Medications included in the Master Formulary are covered through the exception processes. |
45
| · | Pharmacy Management Program: Program of 90 days dispensing for Patients with chronic conditions: Providers can prescribe a 90-day supply for certain medications. This program allows the beneficiary to pay one (1) co-payment for a 90-day supply of medications instead of paying three (3) co-payments (1 co-payment per month). |
Services Excluded from the Basic Coverage
The following services are excluded from Basic Coverage; if you have any questions about the list or regarding your coverage please call your Health Plan.
| · | Services to Patients not eligible to the Government Health Plan. |
| · | Services for non-covered illnesses or trauma. |
| · | Services for automobile accidents covered by the Administration of Compensation for Automobile Accidents (ACAA, for its acronym in Spanish). |
| · | Accidents on the job that are covered by the State Insurance Fund Corporation. |
| · | Services covered by another insurance or entity with primary responsibility (third party liability). |
| · | Specialized nursing services for the comfort of the Patient when they are not medically necessary. |
| · | Hospitalizations for services that can be rendered on an outpatient basis. |
| · | Hospitalization of a Patient for diagnostic services only. |
| · | Expenses for services or materials for the Patient’s comfort such as telephone, television, admission kits, etc. |
| · | Services rendered by Patient’s relative (parents, children, siblings, grandparents, grandchildren, spouse, etc.). |
| · | Organ and tissue transplants, except skin, bone and corneal transplants. |
| · | Weight control Treatments (obesity or weight increase for aesthetic reasons). |
| · | Sports medicine, music therapy and natural medicine. |
| · | Cosmetic surgery to correct physical appearance defects. |
| · | Services, diagnostic tests ordered or provided by naturopaths, and iridologists. |
| · | Health Certificates except for (i) venereal disease research laboratory tests, (ii) tuberculosis tests and (iii) any certification related to the eligibility for the Medicaid program. |
| · | Mammoplasty or plastic reconstruction of breast for aesthetic purposes only. |
| · | Outpatient use of fetal monitor. |
| · | Services, Treatment or hospitalization as a result of induced, non-therapeutic abortions or their complications. The following are considered induced abortions (code and description): |
| ✓ | 59840 – Induced abortion – dilation and curettage; |
| ✓ | 59841 – Induced abortion – dilation and expulsion; |
| ✓ | 59850 – Induced abortion – intra-amniotic injection; |
| ✓ | 59851 – Induced abortion – intra-amniotic injection; |
46
| ✓ | 59852 - Induced abortion – intra-amniotic injection; |
| ✓ | 59855 - Induced abortion – by one or more vaginal suppositories (e.g. prostaglandin) with or without cervical dilation (e.g. laminate) including admission and visits, expulsion of the fetus and afterbirth; |
| ✓ | 59856 - Induced abortion – by one or more vaginal suppositories (e.g. prostaglandin) with dilation and curettage or evacuation; and |
| ✓ | 59857 - Induced abortion – by one or more vaginal suppositories (e.g. prostaglandin) with hysterectomy (failed medical evaluation). |
| · | Rebetron or any other prescribed medication for Hepatitis C Treatment, both Treatment and medications are excluded from the Health Plan coverage. The medications as well as the Treatment will be provided by the Hepatitis Program of the Health Department. For additional information refer to the Hepatitis Section previously mentioned in this Handbook. |
| · | Medications delivered by a provider that does not have a pharmacy license, with the exception of medications that are traditionally administered in a doctor’s office such as an injection. |
| · | Epidural anesthesia services. |
| · | Services that are not reasonable or necessary according to the regulations accepted in the practice of medicine. Services rendered in excess to those normally required for diagnostics, prevention, diseases, Treatment, injury or organ system dysfunction or pregnancy condition. |
| · | Mental health services that are not reasonable or necessary according to the accepted regulations for the practice of medical Psychiatry or the services rendered in excess to those usually required for the diagnostic, prevention and Treatment of a mental illness. |
| · | Educational tests, educational services. |
| · | Peritoneal dialysis or hemodialysis services (Covered under the Special Coverage). |
| · | New or experimental procedures not approved by ASES to be included in the Basic Coverage. |
| · | Custody, rest and convalescence once the disease is under control or in irreversible terminal cases (hospice care for Members under 21 is part of basic coverage). |
| · | Services covered under the Special Coverage. |
| · | Services received outside the territorial limit of the Commonwealth of Puerto Rico, except for emergency services for Medicaid or CHIP beneficiaries. |
| · | Judicial order for evaluations for legal purposes. |
| · | Travel expenses, even when ordered by the Primary Care Physician are excluded. |
| · | Eyeglasses, contact lenses and hearing aids (for members over age 21). |
| · | Acupuncture services. |
| · | Procedures for sex changes, including hospitalizations and complications. |
| · | Treatment for infertility and/or related to conception by artificial means including tuboplasty, vasovasectomy, and any other procedure to restore the ability to procreate. |
47
| · | Expenses incurred for the Treatment of conditions resulting from services not covered under the GHP (maintenance Prescriptions and required clinical laboratories for the continuity of a stable health condition, as well as any emergencies which could alter the effects of the previous procedure, are covered). |
Special Coverage Services
Enrollees with special health care needs caused by serious illnesses may be enrolled into Special Coverage Registry to receive Special Coverage services.
Your Primary Care Physician, the personnel designated by the Primary Medical Group or the case coordinator of the Primary Medical Group can instruct you on the conditions that qualify for the special coverage. Any of them can help you to be included in the Special Coverage by sending all the necessary information on your medical condition your Health Plan.
Once enrolled in special coverage, Enrollees have the freedom to choose the providers for these services among the providers in the Preferred Provider Network of the Primary Medical Group or your Health Plan’s General Network, differential diagnostic interventions up to the verification of the final diagnosis are not part of the Special Coverage.
Medications, laboratory test, diagnostic test and other related procedures specified in this coverage as necessary for ambulatory Treatment or convalescence are part of this coverage and do not require the Preauthorization of the Primary Care Physician or of your Health Plan. Your Health Plan must identify the Enrollees included under this coverage to facilitate Access to the contracted services. The Government Health Plan Special Coverage will be activated when the Enrollee reaches the limit of any other Special Coverage the Enrollee may have under any other plan.
The purpose of this coverage is to facilitate the effective management of beneficiaries with special health condition that require specialized medical attention. This coverage will become effective when the diagnosis is confirmed through the results of tests or procedures performed.
The benefits under this coverage are:
| · | Coronary disease services and intensive care, without limitations. |
| · | Maxillary surgery, with a Referral. |
| · | Neurosurgical and cardiovascular procedures, including pacemakers, valves and any other instrument or artificial device (requires Preauthorization). |
| · | Peritoneal dialysis, hemodialysis and related services (requires Preauthorization). |
| · | Clinical and pathological laboratory test that must be sent outside Puerto Rico for their processing (requires Preauthorization). |
| · | Neonatal intensive care unit services, without limitations. |
48
| · | Treatment with radioisotopes, chemotherapy, radiotherapy and cobalt. |
| · | Gastrointestinal conditions, allergies and nutritional evaluation for autistic Patients. |
| · | The following procedures and diagnostic tests, when medically necessary (require Preauthorization): |
| ✓ | Computerized tomography; |
| ✓ | Magnetic resonance tests; |
| ✓ | Cardiac catheterisms; |
| ✓ | Holter Test; |
| ✓ | Doppler Test; |
| ✓ | Stress Test; |
| ✓ | Lithotripsy; |
| ✓ | Electromyography; |
| ✓ | Tomography test (SPECT); |
| ✓ | Ocular Pletismography test (OPG); |
| ✓ | Impedance Pletismography (IPG); |
| ✓ | Other neurological cerebral-vascular and cardiovascular tests, invasive or non-invasive; |
| ✓ | Nuclear Medicine tests; |
| ✓ | Diagnostic Endoscopies; and |
| ✓ | Genetic Studies. |
| · | Physical therapy – up to 15 additional Treatments per condition per beneficiary a year, when ordered by an Orthopedist, Physiatrist or Chiropractor (requires Preauthorization from your Health Plan). |
| · | General Anesthesia. |
| ✓ | General anesthesia for dental Treatment to children with special needs. |
| · | Hyperbaric chamber. |
| · | Immunosuppressive drugs and laboratory tests required for the maintenance Treatment of Patients who have been operated to receive any transplant, which assure the stability of the beneficiary’s health and the emergencies that may arise after this surgery. |
| · | Treatment for the following conditions after being confirmed by the results of laboratory tests and the diagnosis has been established: |
| ✓ | Positive HIV Factor and Acquired Immunodeficiency Syndrome (AIDS) – Ambulatory and hospitalization services are included. You do not need a Referral or Preauthorization from your Health Plan or the Primary Care Physician for the visits and Treatment at the Immunology Regional Clinics of the Health Department; |
| ✓ | Tuberculosis; |
| ✓ | Leprosy; |
49
| ✓ | Lupus; |
| ✓ | Cystic fibrosis; |
| ✓ | Cancer; |
| ✓ | Hemophilia; |
| ✓ | Aplastics Anemia; |
| ✓ | Reumatoid Artritis ; |
| ✓ | Autism; |
| ✓ | OBG Obstetricians; |
| ✓ | Post Organ Transplantation; and |
| ✓ | Children with special needs, including the conditions described in the Manual of Diagnosis for Children with Special Needs of the Health Department, Office of Health Protection and Promotion, Habilitation Division (the manual) which is part of this part of this document, except: |
| o | Asthma and diabetes, which are included in the Disease Management Program, |
| o | Psychiatric disorders, and |
| o | Intellectual disabilities, behavior manifestations will be managed by the mental health providers under the basic coverage, with the exception of a catastrophic disease. |
| · | Scleroderma. |
| · | Multiple Sclerosis and Amiotrofic Sclerosis Lateral (ALS). |
| · | Services for the Treatment of conditions resulting from self-inflicted damage or as a result of a felony committed by a beneficiary or negligence. |
| · | Chronic renal disease in levels 3, 4 and 5. (Levels 1 and 2 are included in the Basic Coverage). The following is a description of the stages of chronic renal disease: |
| o | Level 3 - FG (glomerular filtration - ml / min. bu 1.73 m ² per unit of body area) between 30 and 59, a moderate decrease in kidney function |
| o | Level 4 - TFG between 15 and 29, a serious decrease in kidney function |
| o | Level 5 - TFG under 15, renal failure with probability of dialysis or kidney transplantation. |
| · | The medications required for the ambulatory Treatment of Tuberculosis and Leprosy are included under the Special Coverage. Medications required for the ambulatory Treatment or hospitalization for beneficiaries diagnosed with AIDS or that are HIV positive are covered under the Special Coverage, except protease inhibitors, which will be provided by the Clinics for the Prevention and Treatment of Sexually Transmitted Diseases (CPTEST, for its acronym in Spanish). |
Services excluded from the Special Coverage
Exclusions and limitations under the Basic Coverage are not covered under the Special unless expressly included in the Special Coverage.
50
Medicare Coverage Services
For Medicare Parts A and B Beneficiaries, the following factors will be considered to determine the coverage to be offered:
| · | Beneficiaries eligible to Part A: |
| ✓ | They will be offered the regular Government Health Plan coverage, excluding the benefits covered by Part A until they reach their limit. In other words, once you reach the benefit limit of Medicare Part A coverage, the Government Health Plan will be activated. |
| ✓ | Part A Deductibles are not included. |
| ✓ | The payment of Deductibles for the regular coverage will be according to the payment capacity table provided to all the Government Health Plan beneficiaries. |
| · | Beneficiaries eligible to Parts A/B: |
| ✓ | They are offered the regular the Government Health Plan pharmacy and dental coverage. |
| ✓ | Part A Deductibles are not included. |
| ✓ | Part B Deductibles and Copayments will be included. |
| · | Dual eligible (Medicare and Medicaid eligible) may not be simultaneously enrolled in the Government Health Plan and in a Medicare Platino plan, for the reason that the Platino plan already included GHP benefits. In addition, as an Enrollee in the plan, the dual eligible may access Covered Services only through the PMG, not through the Medicare Provider List. |
DISEASE MANAGEMENT
Chronic Disease Management
Your Health Plan has programs that will help you control your chronic diseases, such as Diabetes Mellitus, Hypertension, and Congestive Heart Failure (CHF), Obesity, Kidney Failure and Bronchial Asthma. To benefit from these programs you may call your Health Plan. Your Health Plan has a nursing and nutritionist staff available to manage your condition in coordination with the Primary Care Physician.
Case Management
Your Health Plan has a Case Management Program, which is designed to help you with the coordination of medically necessary services for high cost conditions or catastrophic diseases. This program has a staff of nurses, social workers and nutritionists to assist you. You physician, the Hospital staff, your family or you may seek help through this program by calling your Health Plan.
51
THESE ARE YOUR COPAYMENTS AND COINSURANCES
|
CO-PAYS & CO-INSURANCE - Effective on July 1st, 2013
|
||||||||
|
Services
|
Federal
|
CHIPS
|
Población Estatal
|
ELA*
|
||||
|
100
|
110
|
230
|
300
|
310
|
320
|
330
|
400
|
|
|
HOSPITAL
|
HOSPITAL
|
HOSPITAL
|
HOSPITAL
|
HOSPITAL
|
||||
|
Admissions
|
$0
|
$3
|
$0
|
$3
|
$5
|
$6
|
$20
|
$50
|
|
Nursery
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
||||
|
Emergency Room (ER) Visit
|
$0
|
$0
|
$0
|
$1
|
$5
|
$10
|
$15
|
$20
|
|
Non-emergency visit to a hospital emergency room.
|
$3.80
|
$3.80
|
$0
|
$15
|
$15
|
$15
|
$15
|
$20
|
|
Trauma
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
||||
|
Primary Care Physician (PCP)
|
$0
|
$1
|
$0
|
$0
|
$1
|
$2
|
$2
|
$3
|
|
Specialist
|
$0
|
$1
|
$0
|
$1
|
$1
|
$3
|
$4
|
$7
|
|
Sub-Specialist
|
$0
|
$1
|
$0
|
$1
|
$1
|
$3
|
$5
|
$10
|
|
Pre-natal services
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
OTHER SERVICES
|
OTHER SERVICES
|
OTHER SERVICES
|
OTHER SERVICES
|
OTHER SERVICES
|
||||
|
High-Tech Laboratories**
|
$0
|
50¢
|
$0
|
$1
|
$1
|
$2
|
$3
|
20%
|
|
Clinical Laboratories**
|
$0
|
50¢
|
$0
|
$1
|
$1
|
$2
|
$3
|
20%
|
|
X-Rays**
|
$0
|
50¢
|
$0
|
$1
|
$1
|
$2
|
$3
|
20%
|
|
Special diagnostic Tests**
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$6
|
40%
|
|
Therapy – Physical
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$3
|
$5
|
|
Therapy – Respiratory
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$3
|
$5
|
|
Therapy – Occupational
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$3
|
$5
|
|
Vaccines
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
Healthy Child Care
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
DENTAL
|
DENTAL
|
DENTAL
|
DENTAL
|
DENTAL
|
||||
|
Preventive (Child)
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
Preventive (Adult)
|
$0
|
$1
|
$0
|
$0
|
$1
|
$2
|
$3
|
$3
|
|
Restorative
|
$0
|
$1
|
$0
|
$0
|
$1
|
$5
|
$6
|
$10
|
|
PHARMACY***
|
PHARMACY***
|
PHARMACY***
|
PHARMACY***
|
PHARMACY***
|
||||
|
Generic (Children 0-18)
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$5
|
|
Generic (Adult)****
|
$1
|
$1
|
N/A
|
$1
|
$2
|
$3
|
$5
|
$5
|
|
Brand (Children 0-18)
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$10
|
|
Brand (Adult)****
|
$3
|
$3
|
N/A
|
$3
|
$4
|
$5
|
$7
|
$10
|
|
Services
|
Federal
|
CHIPS
|
Población Estatal
|
ELA*
|
||||
|
100
|
110
|
230
|
300
|
310
|
320
|
330
|
400
|
|
*Code 400 in ELA column refers to the population that subscribes as public employees of the Puerto Rico Government.
** Apply to diagnostic tests only. Copays do not applied to tests required as part of a preventive service.
***Copays apply to each drug included in the same prescription pad. Pharmacy exception (children 0- 18) does not apply to 400 ELA employees.
****Co-pays for children 0-18 years of age are not applicable for Medicaid, Commonwealth medically indigent eligible, and for children 0-18 enrolled in the CHIP Program in group ages 0-18.
Co-pays may apply to children ages over 18 years old as well as to adults.
52
HEALTH REGIONS MAP
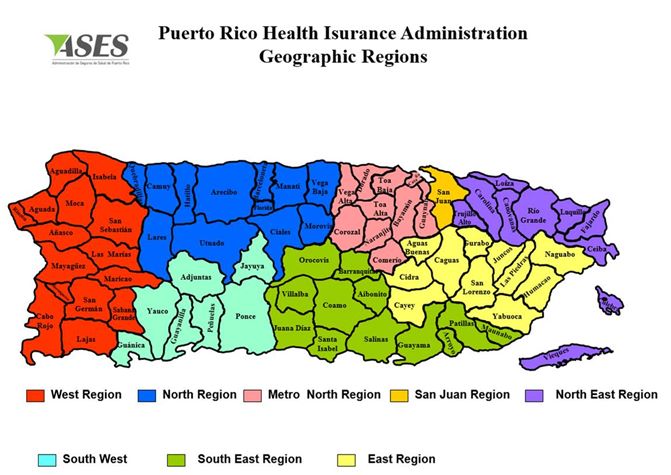
53
Commonwealth of Puerto Rico
Health Department
Assistant Secretary for Family Health, Integrated Services and Health Promotion
Central Office for AIDS Affairs and Transmissible Diseases
Division of HIV/AIDS Services: Preventive Care and Health
Ryan White Part B Program
|
ARECIBO REGION
|
|
|
Clinic
|
Pharmacy
|
|
CPTET Arecibo / Clinic de Immunology
Tel. (787) 817-2677 / 878-7895
Fax. (787) 881-5773
Physical Address: Antiguo Hospital de Distrito, Rd.129 Hacia Lares
Postal Address: PO Box 397 Arecibo,PR 00613
Medical Director: Dra. Evelyn Reyes
MC: Lourdes Castro
|
Pharmacy García
Tel. (787) 898-3975 / 820-5158
Fax. (787) 820-9048
Physical Address: St. Vidal Félix #121
Hatillo, P.R. 00659
Postal Address: PO Box 67 Hatillo PR 00659
Pharmacist: Daniel Mahíques Nieves
|
|
Centro de Salud de Lares, Inc.
Ryan White Part C
Tel. (787) 897-2727 / 2155 /1720 /1730
Fax. (787) 897-2155 / 2725
Physical Address: Rd.111 KM 1.9 Ave. Los Patriotas
Postal Address: PO Box 379
Lares, P.R. 00669
Director: Sr. Gonzálo Maldonado R.N.
MC: Elsie Camacho
MC: Zulma Román
|
In House Pharmacy
Tel. (787) 897-3610 / 897-3023
Tel. (787) 897-2727 / 2155 / 1720
Cel. (787) 414-1304
Fax: (787) 897-2725
Physical Address: Rd.111 KM 1.9
Ave. Los Patriotas
Postal Address: PO Box 379
Lares, P.R. 00669
Pharmacist: Lic. Mayra Vélez
E mail: rw_csl@hotmail.com
Contact: Domingo Carrero
|
|
*Branches attached to Lares Health Center, Inc.
|
|
|
*Centro de Salud de Quebradillas
Tel. (787) 895-2660 / 2670
Physical Address: St. Muñoz Rivera Esq. San Justo #114 Quebradillas, PR 00678
Postal Address: PO Box 1551 Quebradillas, PR 00678
Administrador: Sr. Daniel González Rivera
MC: Marilyn Acevedo
E mail: cdtquebradillas@hotmail.com
|
In House Pharmacy
Tel. (787) 895-2512
Fax. (787) 895-2512
Physical Address: St. Muñoz Rivera Esq. San Justo #114 Quebradillas, PR 00678
Postal Address: PO Box 1551 Quebradillas, PR 00678
Contact: Vanessa Pérez
Pharmacist: Hilda Torres
|
|
*Camuy Health Services, Inc.
Tel. (787) 898-2290 / 262-6603
Fax. (787) 262-1210 / 3789
Physical Address: Ave. Muñoz Rivera #63, Camuy PR 00627
Postal Address: PO Box 660 Camuy, PR 00627-0660
Director: Lcdo. Eddie Pérez
MC: Zaida González
|
In House Pharmacy
Tel.(787)898-2660 Ext.227 Fax.(787)262-4822
Physical Address: Ave. Muñoz Rivera #63, Camuy PR 00627
Postal Address: PO Box 660 Camuy, PR 00627-0660
Pharmacist: Lcda. Luz del Alba Ramírez
E mail: camuy660@coqui.net
|
|
ARECIBO REGION
|
|
|
Clinic
|
Pharmacy
|
|
*Corporación de Servicios Médicos
Primarios y Prevención de Hatillo
Tel. (787) 898-3935 / 4190
Fax. (787) 262-3984
Physical Address: Ave. Dr. Susoni #121 Hatillo
Postal Address: PO Box 907 Hatillo, PR 00659
Director: Armando Legarreta
MC: Wanda León
|
In House Pharmacy
Tel. (787) 898-5764
Fax. (787) 262-3984
Physical Address: Ave. Dr. Susoni #121 Hatillo
Postal Address: PO Box 907 Hatillo, PR 00659
Contact: Leticia Reyes
Pharmacist: Carmelo Nistal
E mail: hatipa19@libertypr.net
|
|
*Ciales Primary Health Care Services
Tel. (787) 871-0601 / 0602 / 0603
Fax. (787) 871-3960
Physical Address: Rd.149 k.m. 12.3 Ciales
Postal Address: PO Box 1427 Ciales, PR 00638
Medical Director: Gladys Rivera Estela
MC: Iraida Marero
Contact: Carmen Sandoval Santiago
|
In House Pharmacy
Tel. (787) 871-0601 Ext. 210
Fax: (787) 871-3960
Physical Address: Rd.149 k.m. 12.3 Ciales PR 00638
Postal Address: PO Box 1427 Ciales, PR 00638
Contact: Janet Maldonado Villalobos
Pharmacist: Aixa Bou
E mail: cphcsinc@yahoo.com
|
|
*Hospital General de Castañer
Tel. (787) 829-5010 / 2055 /7500 /5600
Fax. (787) 829-2913 / 2166 / 4668
Physical Address: Rd.135 k.m. 64.2 Castañer
Postal Address: PO Box 1003 Castañer, PR 00631
Contact: Doris Bengoechea
|
In House Pharmacy
Tel. (787) 829-5010 Ext. 233 / 289
Fax. (787) 829-1479
Physical Address: Rd.135 k.m. 64.2 Castañer
Postal Address: PO Box 1003 Castañer, PR 00631
Pharmacist: Yivet Aquino
E mail: hospitalcastaner@hotmail.com
|
|
BAYAMÓN REGION
|
|
|
Clinic
|
Pharmacy
|
|
Salud Integral en la Montaña, Inc. (SIM) Central Office
Tel. (787) 869-5900 / 5960 / 5950 x. 225, 251
Fax: 1 (787) 869-6120
Physical Address: Rd.152 Naranjito a Barranquitas
Postal Address: PO Box 515 Naranjito, PR 00719
Director: Sandra V. García
Gerente Servicios Clínicos: Nelly Vargas
MC: Maritza Rolón
Madeline Figueroa
E mail: mfigueroa@sim.pr.com
|
Central Office
Tel. (787) 869-5900 Ext. 252
Contact: Sra. Lourdes Chevere * Pharmacy
Director, including 5 clinics
Pharmacist: Sandra V. García
E mail: sgarcia@sim.pr.com
|
|
*Centro de Salud Integral de Naranjito
Tel. (787) 869-1290 Ext. 2203, 2237, 2204
Fax: (787) 869-1800
Physical Address: 164 Rd.Sector el Desvío, Barrio Achiote, Naranjito PR 00719
Postal Address: PO Box 525 Naranjito PR 00719
Director: Maritza Ortíz Berríos
MC: Maritza Rolón Nieves
MC: Judith Rosa
|
In House Pharmacy
Tel. (787) 869-1290 Ext. 2208, 2238
Fax: (787) 869-1800
Physical Address: Rd.164 Sector el Desvío,
Barrio Achiote, Naranjito PR 00719
Postal Address: PO Box 525 Naranjito PR 00719
Pharmacist: Marienilda La Santa
|
|
*Centro de Salud Integral de Barranquitas
Tel. (787) 857-2688
Fax: (787) 857-1730 / 3440
Physical Address: St. Barceló #3, Barranquitas, PR 00794
Postal Address: PO Box 728 Barranquitas, PR 00794
Director: Lourdes
MC: Carmen Carro
|
In House Pharmacy
Tel. (787) 857-2688 Ext. 225
Fax. (787) 857-1730
Physical Address: St. Barceló #3, Barranquitas, PR 00794
Postal Address: PO Box 728
Barranquitas, PR 00794
Pharmacist: Lcda. Lourdes Chéverez
|
|
*Centro de Salud Integral de Comerío
Tel. (787) 875-3375 / 2750
Fax: (787) 875-4230 / 2769
Physical Address: St. Georgetti, Rd.167
Postal Address: PO Box 418 Comerío PR 00782
Director: Angel robles
Medical Director: Fernando Roura
Contact: Jannette Gailé
|
In House Pharmacy
Tel. (787) 875-3375
Fax. (787) 875-4230
Physical Address: Georgetti St., Rd.167
Postal Address: PO Box 418 Comerío PR 00782
Pharmacist: Helen Feshold
|
|
BAYAMÓN REGION
|
|
|
Clinic
|
Pharmacy
|
|
*Centro de Salud Integral de Corozal
Tel. (787) 859-2560 / 2470
Fax: (787) 859-5390
Physical Address: Rd.159 Sector El Desvio, Corozal PR 00783
Postal Address: PO Box 739 Corozal PR 00783
Medical Director: Michael Fusile Nelson
MC: Daysi Alvino Martínez
MC: Leidy Rivera Santiago
|
In House Pharmacy
Tel. (787) 859-2560 / 215
Fax. (787) 859-5390
Physical Address: Rd.159 Sector El Desvio, Corozal PR 00783
Postal Address: PO Box 739 Corozal PR 00783
Contact: Hairilys Vázquez
|
|
*Centro de Salud Integral de Orocovis
Tel. (787) 867-6010
Fax. (787) 867-5210
Physical Address: Ave. Luis Muñoz Marín, Rd.155, Orocovis PR 00720
Postal Address: PO Box 2105 Orocovis PR 00720
Director: Maritza Rolón Nieves
Medical Director: Ada L. Santos Santos
MC: Elba Miranda
|
In House Pharmacy
Tel. (787) 867-6010
Fax. (787) 867-6008
Physical Address: Ave. Luis Muñoz Marín, Rd.155, Orocovis PR 00720
Postal Address: PO Box 2105 Orocovis PR 00720
Pharmacist: Lcda. Brenda Ortíz
|
|
CPTET Bayamón
Immunology Clinic
Tel. (787) 798-1580 / 5154
Tel. (787) 780-6690
Hospital
Switchboard
Tel. (787) 787-5151
Ext. 2112,2224,2534,2510
Fax. (787) 269-7740
ETS. (787) 786-4211
Pediatric: (787) 786-6940
Contact:(787) 313-2573 Zuley Huguet
E mail: zhuguet@salud.gov.pr
Physical Address: Hospital Universitario Dr. Ramón Ruiz Arnau, Ave. Laurel, Santa Juanita Bayamón, P.R. 00956
Postal Address: University Hospital Dr. Ramón Ruiz Arnau, Ave. Laurel, Santa Juanita Bayamón, P.R. 00956
Coordinator: Dr. Calos León Valiente
|
Caridad Pharmacy
Tel.(787) 785-3055 / 269-3140
Fax: (787) 740-5445
Fax. (787) 269-0022
Physical Address: Barrio Hato Tejas Carr.862 Km 1.9 Bayamón, PR 00954
Postal Address: PO Box 4218
Bayamón, PR 00954
Pharmacist: Linnette Rivera
E mail: lynnettepr@hotmail.com
|
|
BAYAMÓN REGION
|
|
|
Clinic
|
Pharmacy
|
|
Centro de Epidemiología de Bayamón
Tel. (787) 787-9831
Fax. (787) 269-5230 / (787) 785-2387
Physical Address: St. Isabel 2da Esq. Degetau Sotano, Antiguo CDT Bay. Pueblo
Postal Address: PO Box 1588 BAyamón, P.R. 00961
Director: Deborath Medina, Marisel Cruz
MC: Alma Ortíz, Rocío Román, David Ayala
|
Plaza III Pharmacy
Tel. (787) 785-0000
Fax: (787) 785-2387
Physical Address: St. Barbosa # 57 Bayamón PR 00961
Postal Address: St. Barbosa # 57 Bayamón PR 00961
Contact: Lcda. Mayda Rodríguez
|
|
Casa Joven del Caribe
Tel. (787) 870-1911 Cel. (787) 630-3571
Fax. (787) 796-2832 870-1911
Physical Address: Rd.# 820 Bo. Marzán Sector Río Lajas, Toa Alta
Postal Address: PO Box 694, Dorado, P.R. 00646
Director: Rev. Samuel Agosto López
MC: Felicita Santiago, Betzaida Rivera
|
Plaza III Pharmacy
Tel. (787) 785-0000
Fax: (787) 785-2387
Physical Address: St. Barbosa # 57 Bayamón PR 00961
Postal Address: St. Barbosa #57 Bayamón, PR 00961
Contact: Lcda. Mayda Rodríguez
|
|
CAGUAS REGION
|
|
|
Clinics
|
Pharmacy
|
|
CPTET Caguas
Immunology Clinic
Tel. (787) 744-3141 Ext.1142
ETS. (787) 744-8645
Fax. (787) 746-2898
Postal Address: San Juan Bautista Medical Center
PO Box 5729 Caguas, P.R. 00726
Contacts: Norma Sánchez, Nancy del Valle
Coordinator: Dr. Milton Garland
Pediatric
Tel. (787) 744-3141 Ext.1158 / 1563
Tel. (787) 282-6300 / 8509
Tel. (787) 649-3930
Case Manager: Ivette Peña
Nurse: Juanita Gómez
Ext. 1153
Infectious: Armando Torres Nieves
|
Arleen Pharmacy
Tel. (787) 746-5952 / 745-2838
Fax. (787) 744-3397
Physical Address: Rd. 172 3rd. Section Villa del Rey Caguas, PR 00725
Postal Address: PO Box 5986 Caguas, PR 00726
Contact: Ms. Adria
Pharmacist: Roberto Peirats
Cel. (787) 379-0116
Pharmacist: Arleen Hernández
E-mail: Pharmacyarleen@gmail.com
|
|
Humacao Satellite Clinic
Tel. (787) 640-0980
Physical Address: Humacao Shopping Center Ave. Font Martelo # 100 Humacao, P.R. 00792
|
Central Pharmacy
Tel. (787) 852-0520
Fax. (787) 850-5500
Physical Address Noya Hernández St. # 12 East, Humacao PR 00791
Postal Address: PO Box 669 Humacao 00792
Contact: Julio Garriga
E-mail: julioegarriga@hotmail.com
|
|
Gurabo Community Health Center/Gurabo Family Medicine Center
SIVIF Program
Tel. (787) 737-1131/ 630-5564
Fax.(787) 737-2365
Tel.(787) 737-2377 SIVIF Fax. (787) 737-2377
Physical Address: Ramal 941 Oscar Dávila Section, Gurabo PR
Postal Address: PO Box 1277 Gurabo, PR 00778
Contact: Denisse Figueroa
E mail: dfigueroa@sivif.com
|
In House Pharmacy
Tel. (787) 737-4449
Fax: (787) 737-1242
Physical Address: Rd. 941 Exit Barrio Jaguas Gurabo, PR
Postal Address: PO Box 1277 Gurabo, PR 00778
Pharmacist: Noemí Rivera
|
|
CAGUAS REGION
|
|
|
Clinic
|
Pharmacy
|
|
Project CIS
Ryder Memorial Hospital
Ryan White Part C
Tel. (787) 852-0768
Ext. 4716, 4717, 4609, 4276
Fax. (787) 656-0735 850-1444
Physical Address: Font Martello Ave. #355 (Humacao Exit to Las Piedras)
Postal Address: PO Box 859 Humacao, P.R. 00792
Contacts:
Carmelo Rivera, Awilda, Felicita de Jesús
|
In House Pharmacy
Tel. (787) 852-0768
Ext. 4730, 4724, 4466, 4467, 4718
Fax: (787) 850-1444
Physical Address: Ave. Font Martello #355 (Humacao Exit to Las Piedras)
Postal Address: PO Box 859 Humacao, P.R. 00792
Contacts:
Carmen Ortíz, Cristina Marrero
Lilliam Sepúlveda
María Carradero
|
|
*Corporación de Servicios de Salud y Medicina Avanzada (COSSMA)
Humacao
Tel. (787) 852-2551 / 2595
Fax: (787) 850-1218
Physical Address: Ulises Martínez St. # 50 Humacao, PR 00791
Contact: Bárbara Rodríguez
|
In House Pharmacy
Tel. (787) 852-2551
Fax. (787) 937-0062
Physical Address: Ulises Martínez St. # 50 Humacao, PR 00791
Postal Address: PO Box 1330, Cidra, P.R. 00739
Pharmacist: María de L. García
|
|
*Corporación de Servicios de Salud y Medicina Avanzada (COSSMA)
San Lorenzo
Tel. (787) 736-3655 / 3646
Fax: (787) 937-0059
Physical Address: St. Muñoz Rivera #186, San Lorenzo, P.R. 00754
Postal Address: St. Muñoz Rivera #186, San Lorenzo, P.R. 00754
Contact: Wanda Nieves
|
In House Pharmacy
Tel. (787) 736-3655 Ext.1115
Fax: (787) 937-0064
Physical Address: St. Muñoz Rivera # 186, San Lorenzo, P.R. 00754
Postal Address: PO Box 1330, Cidra, P.R. 00739
Pharmacist: Dra. Ilia Huertas
|
|
*Corporación de Servicios de Salud y Medicina Avanzada (COSSMA)
Cidra
Tel. (787) 739-8182 / 8183
Physical Address: Industrial El Jíbaro Ave. Lote #2 Rd.172 Km. 13.5
Postal Address: PO Box 1330 Cidra, PR 00739
Contact: Yesenia Velázquez
E mail: cossma@cossmapr.org
Internet page: www.cossmapr.org
Sra. Isolina Miranda: imiranda@cossmapr.com
|
In House Pharmacy
Tel. (787) 739-8182 Ext. 1228
Fax: (787) 714-1444
Physical Address: Industrial El Jíbaro Ave. Lote #2 Rd.172 Km. 13.5
Postal Address: PO Box 1330 Cidra, PR 00739
Pharmacist: Lidian Rosario
|
|
*Corporación de Servicios de Salud y Medicina Avanzada (COSSMA)
Yabucoa
Tel. (787) 893-3060 / 3055
Fax: (787) 266-6292
Physical Address: Muñoz Rivera St. #15 Yabucoa, PR
Contact: Diana Mulero
|
Pharmacy
Not applicable
|
|
FAJARDO REGION
|
|
|
Clinic
|
Pharmacy
|
|
CPTET Fajardo
Immunology Clinic
Tel. (787) 801-1992 / 1995
Tel. (787) 863-5437
Fax. (787)801-6767
Physical Address: Urb. Monte Brisa St. Rafael #55 Fajardo, P.R. 00738
Coordinator: Dr. Arturo Hernández Estrella
Contact: Rafaela Díaz
|
Denirka Pharmacy
Tel. (787) 863-7788 / 860-7788
Fax. (787) 863-1422
Physical Address: Avenida General Valero # 305 Fajardo, PR 00738
Postal Address: PO Box 850 Fajardo, PR 00738
Contact: Vanessa Hernández
Contact: Gil Nieves
E-mail: gilnieves@hotmail.com
|
|
MAYAGÜEZ REGION
|
|
|
Clinic
|
Pharmacy
|
|
CPTET Mayagüez
Immunology Clinic
Tel.(787) 834-2115 / 2118
Fax. (787) 265-2850 / 834-2370
Physical Address: Medical Center Next to Mental Health, Rd.#2
Postal Address: PO Box 400
Mayagüez, P.R. 00680
Contact: Damariz Ruiz, Marianita Torres
Coordinator: Ramón Ramírez Ronda
|
In House Pharmacy
Tel. (787) 834-2116 / 2115 / 2118
Cel. (787) 546-5527 Sonia
Cel. (787) 644-3277 Janette Torres
Cel. (787) 233-8080 Sandra
Physical Address: Medical Center Next to Mental Health, Rd.#2
Postal Address: PO Box 400
Mayagüez, P.R. 00680
Pharmacist: Janette Torres
Contacts: Sonia Vargas, Sandra Rivera
|
|
Centro de Salud de Migrantes
West REGION
SSIMA Program Serv. Salud Integrado
Ryan White Part C
Tel.(787) 805-2900 / 805-2920 Ext. 294
Fax.(787) 805-4750
Clinic:(787) 834-1470
ADM: (787) 834-1924
Fax.(787) 805-4750
Physical Address: St. Ramón E. Betánces #392 South Mayagüez, PR 00680
Postal Address: PO Box 7128
Mayagüez, PR 00681-7128
Contact: Wanda Acosta
Director: Reynaldo Serrano
|
In House Pharmacy
Tel.(787) 805-2900 / 920 Ext. 237
Fax:(787) 265-4245
Physical Address: Ramón E. Betances St. #392 South Mayagüez, PR 00680
Postal Address: PO Box 190 Mayagüez PR 00681-7128
Contact: Juliana Torres
Contact: Lilliam Torres
Pharmacy
San Sebastián Satellite Clinic
Tel.(787) 896-1665
Tel. (787) 896-6975
Fax. (787) 896-4570
Physical Address: Rd.119 k.m. 35.2 Bo. Piedras Blancas San Sebastián PR
Contact: Grisel A. Cabá
Pharmacy
Guánica Satellite Clinic
Tel.(787) 821-4511 / 821-3377 / 821-2144
Fax.(787) 821-4511
Physical Address: Montalva St. #23 Ensenada Guánica, PR 00647
Postal Address: Montalva St. #23 Ensenada Guánica, PR 00647
Pharmacist: Lcda. Wanda Torres
|
|
PONCE REGION
|
|
|
Clinic
|
Pharmacy
|
|
Ararat Center
Tel. (787) 284-5884 / 5887 Fax. (787) 284-5874
Physical Address: 8169 San Vicente Condominium, Suite 204, Ponce, P.R.
Postal Address: PO Box 7793 Ponce, P.R. 00732
Contact: Juan Rivera, Madeline Torres
E mail: jrivera@centroararat.org
Administrator: Iván Meléndez Rivera
|
El Apotecario Pharmacy
Tel. (787) 844-2135 / 290-4654
Fax. (787) 844-2135
Physical Address: Tito Castro Ave #625 Rd.14 Front to Pharmacy El Amal La Rambla, Ponce
Postal Address: PMB 381 Ave. Tito Castro #609, Ponce, PR 00716
Contact: Ricardo Cintrón
|
|
Centro de Salud Fam. Dr. Julio Palmieri
Ryan White Part C Program
Tel. (787) 271-3779 / (787) 839-4150
Fax. (787) 271-3779
MC: Ilka Santiago
Physical Address: Valentina Corner Morse St., Arroyo
Postal Address: PO Box 450 Arroyo, P.R. 00714
Contact: Dra. Antonia Márquez
|
In House Pharmacy
Tel. (787) 839-4150 Ext. 234
Fax: (787) 839-3989
Physical Address: Valentina Corner –Morse St. , Arroyo, PR 00714
Postal Address: PO Box 450 Arroyo, P.R. 00714
Pharmacist: Doris Porrata
|
|
Consejo de Salud de la Comunidad
CDT Playa de Ponce
Ryan White Part C Program
Tel. (787) 843-9393 Ext. 258, 262
Fax. (787) 843-0899 / 841-0077
Physical Address: Hostos Ave. 1034 Ponce, P.R. 00716
Postal Address: PO Box 220 Ponce, P.R. 00715 - 0220
Contact: Dra. Awilda García
|
In House Pharmacy
Tel. (787) 843-9370
Fax. (787) 843-9395
Physical Address: Hostos Ave. 1034 Ponce, P.R. 00716
Postal Address: PO Box 220 Ponce, P.R. 00715 - 0220
Contact: Hilda Torres
|
|
CPTET Ponce
Immunology Clinic
Tel. (787) 259-4046 / 259-4731
ETS. (787) 848-2000
Fax. (787) 259-3998 / 842-1943
Physical Address: Antiguo Hospital de Distrito de Ponce Carretera Estatal Bo. Machuelo #14 Ponce, P.R. 00717
Coordinatora: Dra. Gladys Sepúlveda
|
In House Pharmacy
Tel. (787) 843-2188
Fax. (787) 842-1943
Physical Address: Antiguo Hospital de Distrito de Ponce Carretera Estatal Bo. Machuelo #14 Ponce, P.R. 00717
Pharmacist: Jorge López Vega
|
|
PONCE REGION
|
|
|
Clinic
|
Pharmacy
|
|
Especial de la Salud Clinic
Tel. (787) 260-9446
Cuadro: (787) 837-2185 Ext. 2296, 2297, 2298
Fax. (787) 260-2943
Physical Address: St. Hostos #23 Juana Díaz
Postal Address: PO Box 1409 Juana Díaz, P.R. 00795
Director: Carlos Ortíz Torres
|
Sonia Pharmacy
Tel. (787) 837-2666
Fax: (787) 837-4602
Physical Address: Comercio St. #61 Juana Díaz, PR 00795
Postal Address: Comercio St. #61 Juana Díaz, PR 00795
Pharmacist: Sonia Muñoz De Toro
|
|
Centro de Ambulantes Cristo Pobre Inc.
Tel. (787) 841-7149
Fax. (787) 844-5656
Physical Address: Guadalupe St. Esq. Unión #100 Ponce, P.R.
Postal Address: PO Box 334651 Ponce, P.R. 00733-4651
Contact: Irma Valentín
Administrador: Juan de Dios Videau
|
El Apotecario Pharmacy
Tel. (787) 844-2135 / 290-4654
Fax. (787) 844-2135
Physical Address: Ave.Tito Castro #625 Rd.14 In front of El Amal Pharmacy La Rambla, Ponce
Postal Address: PMB 381 Ave. Tito Castro #609, Ponce, PR 00716
Contact: Ricardo Cintrón
|
|
Centro de Servicios de Salud de Patillas
Tel. (787) 839-4320 / 839-4360
Fax. (787) 271-0004
Physical Address: St. Riefhkol # 99
Patillas, PR 00723
Postal Address: PO Box 697
Patillas, PR 00723
Contact: Sra. Carmen Carro
|
In House Pharmacy
Tel. (787) 839-4320 Ext. 241 / 247
Fax. (787) 839-4337
Physical Address: St. Riefhkol # 99
Patillas, PR 00723
Postal Address:PO Box 697
Contact: Anette Alvarado
E mail: pphsc_inc.com
|
|
Amor que Sana, Inc. Project
Administrative Offices
Tel. (787) 844-8081
Fax. (787) 844-8117
Tel./Fax. (787) 259-2882 Manejo de Caso
Physical Address: Traditional Plaza 7033 Méndez Vigo y Aurora Ponce, PR 00717-1250
Postal Address: Traditional Plaza 7033 Méndez Vigo y Aurora Ponce, PR 00717-1250
Contact: Héctor Torres
Director: Juan A. Panelli
|
El Apotecario Pharmacy
Tel. (787) 844-2135 / 290-4654
Fax. (787) 844-8117
Physical Address: Ave. Tito Castro #625 Rd.14 (Front of El Amal Pharmacy) La Rambla, Ponce, PR 00716
Postal Address: PMB 381 Tito Castro Ave. #609 Ponce, PR 00716
Contact: Ricardo Cintrón
|
|
SAN JUAN REGION
|
|
|
Clinic
|
Pharmacy
|
|
Centro Latinoamericano de Enfermedades Transmisibles (CLETS)
Tel. (787) 754-8123 / 8118
Fax. (787) 754-8127
Physical Address: Barrio Monacillos Paseo Celso Barbosa Centro Médico San Juan, P.R. 00936
Postal Address: PO Box 70184 San Juan PR 00936
Director: Dr. Hermes García
|
In House Pharmacy
Tel. (787) 754-8118
Fax: (787) 754-8127
Physical Address: Barrio Monacillos Paseo Celso Barbosa Centro Médico San Juan, P.R. 00936
Postal Address: PO Box 70184 San Juan PR 00936
Pharmacist: Lourdes Fornés
|
|
Concilio de Salud Integral de Loíza
Tel. (787) 876-3130 / 2042 Ext. 261
Fax. (787) 256-1900
Physical Address: Rd.188 Intersección # 187 Loíza
Postal Address: PO Box 509, Loíza P.R. 00772
Contact: Juan C. Castellanos
|
In House Pharmacy
Tel. (787) 876-2005
Fax. (787) 876-6613 / 1120 / 1900
Physical Address: Rd.188 Intersección # 187 Loíza
Postal Address: PO Box 509, Loíza P.R. 00772
Contact: Irma Alvárez
|
|
CPTET Carolina
Immunology Clinic
Tel. (787) 757-1800
Fax. (787) 257-3615
Physical Address: 65 Infantería Ave. KM 8.4 next to Plaza Carolina
Postal Address: Hospital Universitario de Carolina, PO Box 6021, Carolina P.R. 00984-6021
Coordinatora: Dra. Adiana Zayas
|
Hospital UPR Pharmacy
Tel. (787) 757-1800 Ext. 227
Fax: (787) 750-4214
Physical Address: 65 Infantería Ave. KM 8.4 next to Plaza Carolina
Postal Address: Hospital Universitario de Carolina, PO Box 6021, Carolina P.R. 00984-6021
Pharmacist: Juan Villegas
|
|
Community Initiative
Tel. (787) 250-8629 / 8680 / 8690
Fax. (787) 753-4454
Physical Address: Quisqueya St. #61 Esq. Chile, Hato Rey P.R. 00918
Postal Address: PO Box 366535 San Juan, P.R. 00936-366535
Contact: Jonathan Fernández
Director: Dr. José Vargas Vidot
E mail: iniciator@hotmail.com
|
San Rafael Pharmacy
Tel. (787) 724-3307
Fax: (787) 721-4165
Physical Address: 851 Lafayette Esq. San Rafael, Urb. Hipódromo Pda. 20 Santurce PR
Postal Address: 851 Lafayette Esq. San Rafael, Urb. Hipódromo Pda. 20 Santurce PR 00909
Contact: Rafael Díaz
|
|
SAN JUAN REGION
|
|
|
Clinic
|
Pharmacy
|
|
San Juan Municipality
Más Salud Program: Sida con Salud
Tel. (787) 723-2424 Ext. 239 / 243 /294
Fax. (787) 724-5104
Physical Address: Fernández Juncos Ave. 1306 Pda. 19 Santurce, P.R. 00908
Postal Address: PO Box 13694 San Juan, P.R. 00908
Contact: Dr. Luis Martínez
Manager: Sonia Collazo
|
In House Pharmacy
Tel. (787) 723-2424 Ext. 4071
Fax: (787) 721-7596
Physical Address: Fernández Juncos Ave. 1306 Pda. 19 Santurce, P.R. 00908
Postal Address: PO Box 13694 San Juan, P.R. 00908
Contact: Laura Del Carmen Rivera
|
|
New Puerto Rico CoNCRA
Ryan White Part C Program
Tel. (787) 753-9443 / 9463 Ext. 32
Fax. (787) 753-2894 / 9463
Physical Address; Urb. García Ubarry
Brunbaugh St. #162 San Juan, PR 00925
Postal Address: PO Box 20850 San Juan, P.R. 00928-9463
Contact: Manuel Quiñones, Magaly Nieves
Director: Rosaura López
Medical Director: Ext. 11
|
García Pharmacy
Tel. (787) 898-3975 / 820-5158
Fax. (787) 820-9048
Physical Address: Vidal Félix St. #121
Hatillo, P.R. 00659
Postal Address: PO Box 67 Hatillo PR 00659
Pharmacist: Daniel Mahíques Nieves
|
|
CEMI Project
Tel. (787) 766-0025 / 0030
Tel. (787) 771-4740 / 753-5913
Pediatric AIDS Tel. (787) 274-5762
Physical Address: Medical Sciences Campus
Biomedical II Building Floor I, Río Piedras, PR 00935
Contact: Janice Pérez
|
|
|
ACTU Project
Tel. (787) 767-9192 /9193 /9194 /9195
Fax. (787) 754-8002
Physical Address: UPR Medical Sciences Campus
Biomedical II Bldg. next to nuclear medicine , Río Piedras, PR 00935
Contact:Silvia Dávila / sdavila@rcm.upr.edu
|
|
|
SAN JUAN REGION
|
|
|
Clinic
|
Pharmacy
|
|
GAMMA Project
Tel. (787) 759-9595
Fax: (787) 767-4798
Physical Address: ,Centro Cardiovascular de PR Project GAMMA, 8th Floor Suite 814 San Juan
Postal Address: University of Puerto Rico RCM Project GAMMA, PO Box 365067
San Juan PR 00936-5067
Contact: Dra. Irma L. Febo,
Contact: Iraida Salabarria
|
|
Attachment 5
List of Specialty Drugs
Administration of Health Insurance
of the Commonwealth of Puerto Rico (ASES)
Mi Salud Specialty Drug List (contracted)
2013-2014
|
I.
|
Antibiotic
|
Manufacturers
|
||
|
Linezolid tab.
|
Zyvox
|
PA, P
|
Pfizer
|
|
|
II.
|
Antineoplasic Agents
|
|||
|
Ixabepilone
|
Ixempra
|
PA, P
|
BMS
|
|
|
III.
|
Anticonvulsant Agents
|
|||
|
Lacosamide tab. sol.
|
Vimpat
|
PA, P
|
UCB
|
|
|
IV.
|
Anticoagulant Agents
|
|||
|
Rivaroxaban tab.
|
Xarelto
|
PA, P
|
Janssen
|
|
|
V.
|
Osteoporosis
|
|||
|
Teriparadide inj.
|
Forteo
|
PA, P
|
Lilly
|
|
|
VI.
|
Immune Modulators
|
|||
|
Abatacept inj. (SQ & IV)
|
Orencia
|
PA, P
|
BMS
|
|
|
Certolizumab inj.
|
Cimzia
|
PA, P
|
UCB
|
|
|
Infliximab inj.
|
Remicade
|
PA, P
|
Janssen
|
|
|
Golimumab inj.
|
Simponi
|
PA, P
|
Janssen
|
|
|
Ustekinumab inj.
|
Stelara
|
PA, P
|
Janssen
|
|
|
VII.
|
Multiple Sclerosis
|
|||
|
Natalizumab inj.
|
Tysabri
|
PA, P
|
Elan
|
|
|
Dalframpridine tab.
|
Ampyra
|
PA, P
|
Acorda
|
|
|
Fingolimod
|
Gilenda
|
PA, P
|
Novartys
|
|
|
Dymenthyl Fumarate cap.
|
Tecfidera
|
PA, P
|
Biogen
|
|
|
VIII.
|
Antipsychotics
|
|||
|
Paliperidone inj.
|
Invega Sustenna
|
PA, P
|
Janssen
|
|
|
IX.
|
Secondary Hiperparathyroidism
|
|||
|
Paracalcitol
|
Zemplar
|
PA,P
|
Abbvie
|
|
|
PA:
|
Requires preauthorization
|
|||
|
P:
|
Contracted brand name product (rebate)
|
|||
Access to the List of Contracted Specialized Medications:
In the situation in which a provider of medical services prescribes a specialized medication from the List of Contracted Specialized Medications, the MCO, MBHO, TPA or the organization contracted by ASES, shall evaluate the specialized medication requested by means of the exception mechanism. The evaluation shall take into consideration the specific conditions of each case, which may include, but that is not limited to the following: 1) therapeutical failure with regard to all the alternatives in ASES’s PDL, 2) the lack of availability of therapeutic alternatives in ASES’s PDL for the services or conditions covered, 3) history record of adverse reactions to the medications included in ASES’s PDL, 4) contraindications of use in the medications included in ASES’s PDL.
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
ADHD/ANTI-NARCOLEPSY/ANTI- OBESITY/ANOREXIANTS
|
AT LEAST ONE OF THE FOLLOWING
|
61100025100120
|
Lisdexamfetamine Dimesylate Cap 20 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
61100025100130
|
Lisdexamfetamine Dimesylate Cap 30 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
61100025100140
|
Lisdexamfetamine Dimesylate Cap 40 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
61100025100150
|
Lisdexamfetamine Dimesylate Cap 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
61100025100160
|
Lisdexamfetamine Dimesylate Cap 60 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
61100025100170
|
Lisdexamfetamine Dimesylate Cap 70 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
61100030100305
|
Methamphetamine HCl Tab 5 MG
|
|||||
|
61400016100320
|
Dexmethylphenidate HCl Tab 2.5 MG
|
LE > 6
|
|||||
|
61400016100330
|
Dexmethylphenidate HCl Tab 5 MG
|
LE > 6
|
|||||
|
61400016100340
|
Dexmethylphenidate HCl Tab 10 MG
|
LE > 6
|
|||||
|
61400020100305
|
Methylphenidate HCl Tab 5 MG
|
LE > 6
|
|||||
|
61400020100310
|
Methylphenidate HCl Tab 10 MG
|
LE > 6
|
|||||
|
61400020100315
|
Methylphenidate HCl Tab 20 MG
|
LE > 6
|
|||||
|
61400020107010
|
Methylphenidate HCl Cap SR 24HR 10 MG
|
LE > 6, P
|
|||||
|
61400020107020
|
Methylphenidate HCl Cap SR 24HR 20 MG
|
LE > 6, P
|
|||||
|
61400020107030
|
Methylphenidate HCl Cap SR 24HR 30 MG
|
LE > 6, P
|
|||||
|
61400020107040
|
Methylphenidate HCl Cap SR 24HR 40 MG
|
LE > 6, P
|
|||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107020
|
Dexmethylphenidate HCl Cap SR 24 HR 5 MG
|
LE > 6, ST
|
1
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107030
|
Dexmethylphenidate HCl Cap SR 24 HR 10 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107035
|
Dexmethylphenidate HCl Cap SR 24 HR 15 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107040
|
Dexmethylphenidate HCl Cap SR 24 HR 20 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107045
|
Dexmethylphenidate HCl Cap SR 24 HR 25 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107050
|
Dexmethylphenidate HCl Cap SR 24 HR 30 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107055
|
Dexmethylphenidate HCl Cap SR 24 HR 35 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400016107060
|
Dexmethylphenidate HCl Cap SR 24 HR 40 MG
|
LE > 6, ST
|
2
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100210
|
Methylphenidate HCl Cap CR 10 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100220
|
Methylphenidate HCl Cap CR 20 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100230
|
Methylphenidate HCl Cap CR 30 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100240
|
Methylphenidate HCl Cap CR 40 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100250
|
Methylphenidate HCl Cap CR 50 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100260
|
Methylphenidate HCl Cap CR 60 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100460
|
Methylphenidate HCl Tab SA OSM 18 MG
|
LE > 6, ST
|
3
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100465
|
Methylphenidate HCl Tab SA OSM 27 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100470
|
Methylphenidate HCl Tab SA OSM 36 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020100480
|
Methylphenidate HCl Tab SA OSM 54 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020107010
|
Methylphenidate HCl Cap SR 24HR 10 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020107020
|
Methylphenidate HCl Cap SR 24HR 20 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020107030
|
Methylphenidate HCl Cap SR 24HR 30 MG
|
LE > 6, ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING MODIFIED RELEASE (DRUG DELIVERY SYSTEM)
|
61400020107040
|
Methylphenidate HCl Cap SR 24HR 40 MG
|
LE > 6, ST
|
||||
|
61100020100303
|
Dextroamphetamine Sulfate Tab 2.5 MG
|
LE > 4
|
|||||
|
61100020100305
|
Dextroamphetamine Sulfate Tab 5 MG
|
LE > 4
|
4
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
61100020100308
|
Dextroamphetamine Sulfate Tab 7.5 MG
|
LE > 4
|
|||||
|
61100020100310
|
Dextroamphetamine Sulfate Tab 10 MG
|
LE > 4
|
|||||
|
61100020100315
|
Dextroamphetamine Sulfate Tab 15 MG
|
LE > 4
|
|||||
|
61100020100330
|
Dextroamphetamine Sulfate Tab 20 MG
|
LE > 4
|
|||||
|
61100020100350
|
Dextroamphetamine Sulfate Tab 30 MG
|
LE > 4
|
|||||
|
61100020107005
|
Dextroamphetamine Sulfate Cap SR 24HR 5 MG
|
LE > 4
|
|||||
|
61100020107010
|
Dextroamphetamine Sulfate Cap SR 24HR 10 MG
|
LE > 4
|
|||||
|
61100020107015
|
Dextroamphetamine Sulfate Cap SR 24HR 15 MG
|
LE > 4
|
|||||
|
61109902100305
|
Amphetamine-Dextroamphetamine Tab 5 MG
|
LE > 4
|
|||||
|
61109902100307
|
Amphetamine-Dextroamphetamine Tab 7.5 MG
|
LE > 4
|
|||||
|
61109902100310
|
Amphetamine-Dextroamphetamine Tab 10 MG
|
LE > 4
|
|||||
|
61109902100312
|
Amphetamine-Dextroamphetamine Tab 12.5 MG
|
LE > 4
|
|||||
|
61109902100315
|
Amphetamine-Dextroamphetamine Tab 15 MG
|
LE > 4
|
|||||
|
61109902100320
|
Amphetamine-Dextroamphetamine Tab 20 MG
|
LE > 4
|
|||||
|
61109902100330
|
Amphetamine-Dextroamphetamine Tab 30 MG
|
LE > 4
|
|||||
|
61354015100110
|
Atomoxetine HCl Cap 10 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
61354015100118
|
Atomoxetine HCl Cap 18 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
61354015100125
|
Atomoxetine HCl Cap 25 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
61354015100140
|
Atomoxetine HCl Cap 40 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
61354015100160
|
Atomoxetine HCl Cap 60 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
61354015100170
|
Atomoxetine HCl Cap 80 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
61354015100180
|
Atomoxetine HCl Cap 100 MG (Base Equiv)
|
LE > 6, PA
|
|||||
|
AMEBICIDES
|
14000030000310
|
Iodoquinol Tab 210 MG
|
|||||
|
14000030000320
|
Iodoquinol Tab 650 MG
|
||||||
|
AMINOGLYCOSIDES
|
07000060102105
|
Streptomycin Sulfate For Inj 1 GM
|
|||||
|
07000070000120
|
Tobramycin Inhal Cap 28 MG
|
PA
|
|||||
|
07000070002520
|
Tobramycin Nebu Soln 300 MG/5ML
|
PA
|
|||||
|
07000070002530
|
Tobramycin Nebu Soln 300 MG/4ML
|
PA
|
|||||
|
ANALGESICS - ANTI-INFLAMMATORY
|
AT LEAST TWO OF THE FOLLOWING
|
66270015006410
|
Adalimumab Inj Kit 20 MG/0.4ML
|
PA
|
5
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST TWO OF THE FOLLOWING
|
66270015006420
|
Adalimumab Inj Kit 40 MG/0.8ML (50 MG/ML)
|
PA
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
66290030002020
|
Etanercept Subcutaneous Inj 50 MG/ML
|
PA
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
66290030002025
|
Etanercept Subcutaneous Inj 25 MG/0.5ML
|
PA
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
66290030006420
|
Etanercept For Subcutaneous Inj Kit 25 MG
|
PA
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
52505040002120
|
Infliximab For IV Inj 100 MG
|
PA
|
||||
|
66270040002040
|
Golimumab Subcutaneous Inj 100 MG/ML
|
PA
|
|||||
|
66100020000320
|
Ibuprofen Tab 400 MG
|
||||||
|
66100020000330
|
Ibuprofen Tab 600 MG
|
||||||
|
66100020000340
|
Ibuprofen Tab 800 MG
|
||||||
|
66100030000105
|
Indomethacin Cap 25 MG
|
||||||
|
66100030000110
|
Indomethacin Cap 50 MG
|
||||||
|
66100030000205
|
Indomethacin Cap CR 75 MG
|
||||||
|
66100030001805
|
Indomethacin Susp Oral 25 MG/5ML
|
||||||
|
66100055000320
|
Nabumetone Tab 500 MG
|
||||||
|
66100055000330
|
Nabumetone Tab 750 MG
|
||||||
|
66100060000305
|
Naproxen Tab 250 MG
|
||||||
|
66100060000310
|
Naproxen Tab 375 MG
|
||||||
|
66100060000315
|
Naproxen Tab 500 MG
|
||||||
|
66100060000610
|
Naproxen Tab EC 375 MG
|
||||||
|
66100060000615
|
Naproxen Tab EC 500 MG
|
||||||
|
66100060001805
|
Naproxen Susp 125 MG/5ML
|
||||||
|
66100080000305
|
Sulindac Tab 150 MG
|
||||||
|
66100080000310
|
Sulindac Tab 200 MG
|
||||||
|
66100525000110
|
Celecoxib Cap 50 MG
|
ST
|
|||||
|
66100525000120
|
Celecoxib Cap 100 MG
|
ST
|
6
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
66100525000130
|
Celecoxib Cap 200 MG
|
ST
|
|||||
|
66100525000140
|
Celecoxib Cap 400 MG
|
ST
|
|||||
|
66200010000105
|
Auranofin Cap 3 MG
|
PA
|
|||||
|
66250050100320
|
Methotrexate Sodium Tab 2.5 MG (Antirheumatic)
|
||||||
|
ANALGESICS - NONNARCOTIC
|
AT LEAST ONE OF THE FOLLOWING
|
64100050000310
|
Diflunisal Tab 500 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
64100055000320
|
Magnesium Salicylate Tab 600 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
64100075000305
|
Salsalate Tab 500 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
64100075000310
|
Salsalate Tab 750 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
64109902200315
|
Choline & Magnesium Salicylates Tab 1000 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
64109902200910
|
Choline & Magnesium Salicylates Liq 500 MG/5ML
|
|||||
|
64100010000307
|
ASPIRIN TAB 81 MG
|
Suplido de 90 dias en cada receta
|
|||||
|
64100010000315
|
ASPIRIN TAB 325 MG
|
Suplido de 90 dias en cada receta
|
|||||
|
64991003100108
|
Butalbital-Acetaminophen-Caffeine Cap 50-300-40 MG
|
||||||
|
64991003100110
|
Butalbital-Acetaminophen-Caffeine Cap 50-325-40 MG
|
||||||
|
64991003100310
|
Butalbital-Acetaminophen-Caffeine Tab 50-325-40 MG
|
||||||
|
64991003102020
|
Butalbital-Acetaminophen-Caffeine Soln 50-325-40 MG/15ML
|
7
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
ANALGESICS - OPIOID
|
65100020200305
|
Codeine Sulfate Tab 15 MG
|
|||||
|
65100020200310
|
Codeine Sulfate Tab 30 MG
|
||||||
|
65100020200315
|
Codeine Sulfate Tab 60 MG
|
||||||
|
65100020202020
|
Codeine Sulfate Oral Soln 30 MG/5ML
|
||||||
|
65100025008610
|
Fentanyl TD Patch 72HR 12 MCG/HR
|
||||||
|
65100025008620
|
Fentanyl TD Patch 72HR 25 MCG/HR
|
||||||
|
65100025008630
|
Fentanyl TD Patch 72HR 50 MCG/HR
|
||||||
|
65100025008640
|
Fentanyl TD Patch 72HR 75 MCG/HR
|
||||||
|
65100025008650
|
Fentanyl TD Patch 72HR 100 MCG/HR
|
||||||
|
65100035100310
|
Hydromorphone HCl Tab 2 MG
|
||||||
|
65100035100320
|
Hydromorphone HCl Tab 4 MG
|
||||||
|
65100035100330
|
Hydromorphone HCl Tab 8 MG
|
||||||
|
65100035100920
|
Hydromorphone HCl Liqd 1 MG/ML
|
||||||
|
65100035102007
|
Hydromorphone HCl Preservative Free (PF) Inj 1 MG/ML
|
||||||
|
65100035102010
|
Hydromorphone HCl Inj 2 MG/ML
|
||||||
|
65100035102012
|
Hydromorphone HCl Preservative Free (PF) Inj 2 MG/ML
|
||||||
|
65100035102022
|
Hydromorphone HCl Preservative Free (PF) Inj 4 MG/ML
|
||||||
|
65100035102027
|
Hydromorphone HCl Preservative Free (PF) Inj 10 MG/ML
|
||||||
|
65100035102120
|
Hydromorphone HCl For Inj 250 MG
|
||||||
|
65100045102007
|
Meperidine HCl Inj 10 MG/ML
|
||||||
|
65100045102010
|
Meperidine HCl Inj 25 MG/ML
|
||||||
|
65100045102013
|
Meperidine HCl Inj 25 MG/0.5ML (50 MG/ML)
|
||||||
|
65100045102015
|
Meperidine HCl Inj 50 MG/ML
|
||||||
|
65100045102016
|
Meperidine HCl Inj 75 MG/1.5ML (50 MG/ML)
|
||||||
|
65100045102018
|
Meperidine HCl Inj 100 MG/2ML (50 MG/ML)
|
||||||
|
65100045102020
|
Meperidine HCl Inj 75 MG/ML
|
||||||
|
65100045102030
|
Meperidine HCl Inj 100 MG/ML
|
8
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
65100055100310
|
Morphine Sulfate Tab 15 MG
|
||||||
|
65100055100315
|
Morphine Sulfate Tab 30 MG
|
||||||
|
65100055102065
|
Morphine Sulfate Oral Soln 10 MG/5ML
|
||||||
|
65100055102070
|
Morphine Sulfate Oral Soln 20 MG/5ML
|
||||||
|
65100055102090
|
Morphine Sulfate (Concentrate) Oral Soln 20 MG/ML
|
||||||
|
65100055105205
|
Morphine Sulfate Suppos 5 MG
|
||||||
|
65100055105210
|
Morphine Sulfate Suppos 10 MG
|
||||||
|
65100055105215
|
Morphine Sulfate Suppos 20 MG
|
||||||
|
65100055105220
|
Morphine Sulfate Suppos 30 MG
|
||||||
|
65100075100110
|
Oxycodone HCl Cap 5 MG
|
||||||
|
65100075100310
|
Oxycodone HCl Tab 5 MG
|
||||||
|
65100075100320
|
Oxycodone HCl Tab 10 MG
|
||||||
|
65100075100325
|
Oxycodone HCl Tab 15 MG
|
||||||
|
65100075100330
|
Oxycodone HCl Tab 20 MG
|
||||||
|
65100075100340
|
Oxycodone HCl Tab 30 MG
|
||||||
|
65100075101320
|
Oxycodone HCl Conc 100 MG/5ML (20 MG/ML)
|
||||||
|
65100075102005
|
Oxycodone HCl Soln 5 MG/5ML
|
||||||
|
65100095100320
|
Tramadol HCl Tab 50 MG
|
||||||
|
65200010200715
|
Buprenorphine HCl-Naloxone HCl SL Tab 1.4-0.36 MG (Base Eq)
|
PA
|
|||||
|
65200010200720
|
Buprenorphine HCl-Naloxone HCl SL Tab 2-0.5 MG (Base Equiv)
|
PA
|
|||||
|
65200010200732
|
Buprenorphine HCl-Naloxone HCl SL Tab 5.7-1.4 MG (Base Eq)
|
PA
|
|||||
|
65200010200740
|
Buprenorphine HCl-Naloxone HCl SL Tab 8-2 MG (Base Equiv)
|
PA
|
|||||
|
65200010208220
|
Buprenorphine HCl-Naloxone HCl SL Film 2-0.5 MG (Base Equiv)
|
PA
|
|||||
|
65200010208230
|
Buprenorphine HCl-Naloxone HCl SL Film 4-1 MG (Base Equiv)
|
PA
|
9
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
65200010208240
|
Buprenorphine HCl-Naloxone HCl SL Film 8-2 MG (Base Equiv)
|
PA
|
|||||
|
65200010208250
|
Buprenorphine HCl-Naloxone HCl SL Film 12-3 MG (Base Equiv)
|
PA
|
|||||
|
65990002200305
|
Oxycodone w/ Acetaminophen Tab 2.5-325 MG
|
||||||
|
65990002200308
|
Oxycodone w/ Acetaminophen Tab 5-300 MG
|
||||||
|
65990002200310
|
Oxycodone w/ Acetaminophen Tab 5-325 MG
|
||||||
|
65990002200325
|
Oxycodone w/ Acetaminophen Tab 7.5-300 MG
|
||||||
|
65990002200327
|
Oxycodone w/ Acetaminophen Tab 7.5-325 MG
|
||||||
|
65990002200333
|
Oxycodone w/ Acetaminophen Tab 10-300 MG
|
||||||
|
65990002200335
|
Oxycodone w/ Acetaminophen Tab 10-325 MG
|
||||||
|
65990002200430
|
Oxycodone w/ Acetaminophen Tab CR 7.5-325 MG
|
||||||
|
65990002202005
|
Oxycodone w/ Acetaminophen Soln 5-325 MG/5ML
|
||||||
|
65991002050310
|
Acetaminophen w/ Codeine Tab 300-15 MG
|
||||||
|
65991002050315
|
Acetaminophen w/ Codeine Tab 300-30 MG
|
||||||
|
65991002050320
|
Acetaminophen w/ Codeine Tab 300-60 MG
|
||||||
|
65991002051805
|
Acetaminophen w/ Codeine Susp 120-12 MG/5ML
|
||||||
|
65991002052020
|
Acetaminophen w/ Codeine Soln 120-12 MG/5ML
|
||||||
|
65991702100302
|
Hydrocodone-Acetaminophen Tab 2.5-325 MG
|
||||||
|
65991702100305
|
Hydrocodone-Acetaminophen Tab 10-325 MG
|
||||||
|
65991702100309
|
Hydrocodone-Acetaminophen Tab 5-300 MG
|
||||||
|
65991702100322
|
Hydrocodone-Acetaminophen Tab 7.5-300 MG
|
||||||
|
65991702100356
|
Hydrocodone-Acetaminophen Tab 5-325 MG
|
||||||
|
65991702100358
|
Hydrocodone-Acetaminophen Tab 7.5-325 MG
|
||||||
|
65991702100375
|
Hydrocodone-Acetaminophen Tab 10-300 MG
|
||||||
|
65991702102015
|
Hydrocodone-Acetaminophen Soln 7.5-325 MG/15ML
|
||||||
|
65991702102024
|
Hydrocodone-Acetaminophen Soln 10-300 MG/15ML
|
||||||
|
65991702102025
|
Hydrocodone-Acetaminophen Soln 10-325 MG/15ML
|
||||||
|
ANDROGENS-ANABOLIC
|
AT LEAST ONE OF THE FOLLOWING
|
23100030101710
|
Testosterone Cypionate IM in Oil 100 MG/ML
|
10
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
23100030101715
|
Testosterone Cypionate IM in Oil 200 MG/ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
23100030201710
|
Testosterone Enanthate IM in Oil 200 MG/ML
|
|||||
|
23100020000105
|
Methyltestosterone Cap 10 MG
|
||||||
|
23100020000310
|
Methyltestosterone Oral Tab 10 MG
|
||||||
|
ANORECTAL AGENTS
|
89150010005110
|
Hydrocortisone Enema 100 MG/60ML
|
|||||
|
89991002313710
|
Hydrocortisone Acetate w/ Pramoxine Rectal Cream 1- 1%
|
||||||
|
89991002313714
|
Hydrocortisone Acetate w/ Pramoxine Rectal Cream 1.85-1.15%
|
||||||
|
89991002313720
|
Hydrocortisone Acetate w/ Pramoxine Rectal Cream 2.5- 1%
|
||||||
|
89991002313910
|
Hydrocortisone Acetate w/ Pramoxine Rectal Foam 1- 1%
|
||||||
|
ANTHELMINTICS
|
15000002000320
|
Albendazole Tab 200 MG
|
|||||
|
15000007000310
|
Ivermectin Tab 3 MG
|
||||||
|
ANTIANGINAL AGENTS
|
32100020000305
|
Isosorbide Dinitrate Tab 5 MG
|
|||||
|
32100020000310
|
Isosorbide Dinitrate Tab 10 MG
|
||||||
|
32100020000315
|
Isosorbide Dinitrate Tab 20 MG
|
||||||
|
32100020000320
|
Isosorbide Dinitrate Tab 30 MG
|
||||||
|
32100020000325
|
Isosorbide Dinitrate Tab 40 MG
|
||||||
|
32100025000310
|
Isosorbide Mononitrate Tab 10 MG
|
||||||
|
32100025000320
|
Isosorbide Mononitrate Tab 20 MG
|
||||||
|
32100030000710
|
Nitroglycerin SL Tab 0.3 MG
|
||||||
|
32100030000715
|
Nitroglycerin SL Tab 0.4 MG
|
||||||
|
32100030000720
|
Nitroglycerin SL Tab 0.6 MG
|
||||||
|
32100030008510
|
Nitroglycerin TD Patch 24HR 0.1 MG/HR
|
||||||
|
32100030008520
|
Nitroglycerin TD Patch 24HR 0.2 MG/HR
|
||||||
|
32100030008530
|
Nitroglycerin TD Patch 24HR 0.3 MG/HR
|
11
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
32100030008540
|
Nitroglycerin TD Patch 24HR 0.4 MG/HR
|
||||||
|
32100030008550
|
Nitroglycerin TD Patch 24HR 0.6 MG/HR
|
||||||
|
32100030008560
|
Nitroglycerin TD Patch 24HR 0.8 MG/HR
|
||||||
|
ANTIANXIETY AGENTS
|
57100010000305
|
Alprazolam Tab 0.25 MG
|
|||||
|
57100010000310
|
Alprazolam Tab 0.5 MG
|
||||||
|
57100010000315
|
Alprazolam Tab 1 MG
|
||||||
|
57100010001310
|
Alprazolam Conc 1 MG/ML
|
||||||
|
57100010007505
|
Alprazolam Tab SR 24HR 0.5 MG
|
||||||
|
57100010007510
|
Alprazolam Tab SR 24HR 1 MG
|
||||||
|
57100010007520
|
Alprazolam Tab SR 24HR 2 MG
|
||||||
|
57100010007530
|
Alprazolam Tab SR 24HR 3 MG
|
||||||
|
57100020100105
|
Chlordiazepoxide HCl Cap 5 MG
|
||||||
|
57100020100110
|
Chlordiazepoxide HCl Cap 10 MG
|
||||||
|
57100020100115
|
Chlordiazepoxide HCl Cap 25 MG
|
||||||
|
57100030100305
|
Clorazepate Dipotassium Tab 3.75 MG
|
||||||
|
57100030100310
|
Clorazepate Dipotassium Tab 7.5 MG
|
||||||
|
57100030100320
|
Clorazepate Dipotassium Tab 15 MG
|
||||||
|
57100040000305
|
Diazepam Tab 2 MG
|
||||||
|
57100040000310
|
Diazepam Tab 5 MG
|
||||||
|
57100040000315
|
Diazepam Tab 10 MG
|
||||||
|
57100040001310
|
Diazepam Conc 5 MG/ML
|
||||||
|
Diazepam Conc Oral 5 MG/ML
|
|||||||
|
57100040002001
|
Diazepam Soln 1 MG/ML
|
||||||
|
Diazepam Soln Oral 1 MG/ML
|
|||||||
|
57100060000305
|
Lorazepam Tab 0.5 MG
|
||||||
|
57100060000310
|
Lorazepam Tab 1 MG
|
||||||
|
57100060000315
|
Lorazepam Tab 2 MG
|
||||||
|
57100060001320
|
Lorazepam Conc 2 MG/ML
|
||||||
|
57100070000105
|
Oxazepam Cap 10 MG
|
||||||
|
57100070000110
|
Oxazepam Cap 15 MG
|
||||||
|
57100070000115
|
Oxazepam Cap 30 MG
|
12
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
57200005100310
|
Buspirone HCl Tab 5 MG
|
||||||
|
57200005100315
|
Buspirone HCl Tab 7.5 MG
|
||||||
|
57200005100320
|
Buspirone HCl Tab 10 MG
|
||||||
|
57200005100330
|
Buspirone HCl Tab 15 MG
|
||||||
|
57200005100340
|
Buspirone HCl Tab 30 MG
|
||||||
|
57200040100305
|
Hydroxyzine HCl Tab 10 MG
|
||||||
|
57200040100310
|
Hydroxyzine HCl Tab 25 MG
|
||||||
|
57200040100315
|
Hydroxyzine HCl Tab 50 MG
|
||||||
|
57200040101210
|
Hydroxyzine HCl Syrup 10 MG/5ML
|
||||||
|
57200040200105
|
Hydroxyzine Pamoate Cap 25 MG
|
||||||
|
57200040200110
|
Hydroxyzine Pamoate Cap 50 MG
|
||||||
|
57200040200115
|
Hydroxyzine Pamoate Cap 100 MG
|
||||||
|
ANTIARRHYTHMICS
|
35100030300310
|
Quinidine Sulfate Tab 200 MG
|
|||||
|
35100030300315
|
Quinidine Sulfate Tab 300 MG
|
||||||
|
35200025100105
|
Mexiletine HCl Cap 150 MG
|
||||||
|
35200025100110
|
Mexiletine HCl Cap 200 MG
|
||||||
|
35300010100303
|
Flecainide Acetate Tab 50 MG
|
||||||
|
35300010100305
|
Flecainide Acetate Tab 100 MG
|
||||||
|
35300050000320
|
Propafenone HCl Tab 150 MG
|
||||||
|
35300050000325
|
Propafenone HCl Tab 225 MG
|
||||||
|
35300050000330
|
Propafenone HCl Tab 300 MG
|
||||||
|
35400005000303
|
Amiodarone HCl Tab 100 MG
|
||||||
|
35400005000305
|
Amiodarone HCl Tab 200 MG
|
||||||
|
35400005000320
|
Amiodarone HCl Tab 400 MG
|
||||||
|
ANTIASTHMATIC AND BRONCHODILATOR AGENTS
|
AT LEAST ONE OF THE FOLLOWING
|
44201010103410
|
Albuterol Sulfate Inhal Aero 108 MCG/ACT (90MCG Base Equiv)
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44201045503220
|
Levalbuterol Tartrate Inhal Aerosol 45 MCG/ACT (Base Equiv)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
44201027100120
|
Formoterol Fumarate Inhal Cap 12 MCG
|
13
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
44201027102520
|
Formoterol Fumarate Soln Nebu 20 MCG/2ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
44201058108020
|
Salmeterol Xinafoate Aer Pow BA 50 MCG/DOSE (Base Equiv)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902413220
|
Budesonide-Formoterol Fumarate Dihyd Aerosol 80-4.5 MCG/ACT
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902413240
|
Budesonide-Formoterol Fumarate Dihyd Aerosol 160- 4.5 MCG/ACT
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902703250
|
Fluticasone-Salmeterol Inhal Aerosol 45-21 MCG/ACT
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902703260
|
Fluticasone-Salmeterol Inhal Aerosol 115-21 MCG/ACT
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902703270
|
Fluticasone-Salmeterol Inhal Aerosol 230-21 MCG/ACT
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902708020
|
Fluticasone-Salmeterol Aer Powder BA 100-50 MCG/DOSE
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902708030
|
Fluticasone-Salmeterol Aer Powder BA 250-50 MCG/DOSE
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
44209902708040
|
Fluticasone-Salmeterol Aer Powder BA 500-50 MCG/DOSE
|
ST
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400010103408
|
Beclomethasone Dipropionate Inhal Aero Soln 40 MCG/ACT
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400010103428
|
Beclomethasone Dipropionate Inhal Aero Soln 80 MCG/ACT
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400015001830
|
Budesonide Inhalation Susp 0.25 MG/2ML
|
LE < 12
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400015001840
|
Budesonide Inhalation Susp 0.5 MG/2ML
|
LE < 12
|
14
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING
|
44400015001850
|
Budesonide Inhalation Susp 1 MG/2ML
|
LE < 12
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400015008009
|
Budesonide Inhal Aero Powd 90 MCG/ACT (Breath Activated)
|
LE < 12
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400015008018
|
Budesonide Inhal Aero Powd 180 MCG/ACT (Breath Activated)
|
LE < 12
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400033208010
|
Fluticasone Propionate Aer Pow BA 50 MCG/BLISTER
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400033208020
|
Fluticasone Propionate Aer Pow BA 100 MCG/BLISTER
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400033208030
|
Fluticasone Propionate Aer Pow BA 250 MCG/BLISTER
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400033223220
|
Fluticasone Propionate HFA Inhal Aero 44 MCG/ACT (50/Valve)
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400033223230
|
Fluticasone Propionate HFA Inhal Aer 110 MCG/ACT (125/Valve)
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
44400033223240
|
Fluticasone Propionate HFA Inhal Aer 220 MCG/ACT (250/Valve)
|
|||||
|
44100030102020
|
Ipratropium Bromide Inhal Soln 0.02%
|
||||||
|
44100030123420
|
Ipratropium Bromide HFA Inhal Aerosol 17 MCG/ACT
|
||||||
|
44100080100120
|
Tiotropium Bromide Monohydrate Inhal Cap 18 MCG (Base Equiv)
|
PA
|
|||||
|
44150010102505
|
Cromolyn Sodium Soln Nebu 20 MG/2ML
|
||||||
|
44201010100305
|
Albuterol Sulfate Tab 2 MG
|
||||||
|
44201010100310
|
Albuterol Sulfate Tab 4 MG
|
||||||
|
44201010101205
|
Albuterol Sulfate Syrup 2 MG/5ML
|
||||||
|
44201010102515
|
Albuterol Sulfate Soln Nebu 0.083% (2.5 MG/3ML)
|
||||||
|
44201010102520
|
Albuterol Sulfate Soln Nebu 0.5% (5 MG/ML)
|
||||||
|
44201010102555
|
Albuterol Sulfate Soln Nebu 0.63 MG/3ML (Base Equiv)
|
15
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
44201010102560
|
Albuterol Sulfate Soln Nebu 1.25 MG/3ML (Base Equiv)
|
||||||
|
44201060200305
|
Terbutaline Sulfate Tab 2.5 MG
|
||||||
|
44201060200310
|
Terbutaline Sulfate Tab 5 MG
|
||||||
|
44209902013420
|
Ipratropium-Albuterol Inhal Aerosol Soln 20-100 MCG/ACT
|
ST
|
|||||
|
44300040001010
|
Theophylline Elixir 80 MG/15ML
|
||||||
|
44300040002010
|
Theophylline Soln 80 MG/15ML
|
||||||
|
44300040007020
|
Theophylline Cap SR 24HR 100 MG
|
||||||
|
44300040007030
|
Theophylline Cap SR 24HR 200 MG
|
||||||
|
44300040007040
|
Theophylline Cap SR 24HR 300 MG
|
||||||
|
44300040007050
|
Theophylline Cap SR 24HR 400 MG
|
||||||
|
44300040007420
|
Theophylline Tab SR 12HR 100 MG
|
||||||
|
44300040007430
|
Theophylline Tab SR 12HR 200 MG
|
||||||
|
44300040007440
|
Theophylline Tab SR 12HR 300 MG
|
||||||
|
44300040007455
|
Theophylline Tab SR 12HR 450 MG
|
||||||
|
44300040007540
|
Theophylline Tab SR 24HR 400 MG
|
||||||
|
44300040007560
|
Theophylline Tab SR 24HR 600 MG
|
||||||
|
44505050100330
|
Montelukast Sodium Tab 10 MG (Base Equiv)
|
||||||
|
44505050100516
|
Montelukast Sodium Chew Tab 4 MG (Base Equiv)
|
||||||
|
44505050100520
|
Montelukast Sodium Chew Tab 5 MG (Base Equiv)
|
||||||
|
44603060002120
|
Omalizumab inj 150 MG
|
PA
|
|||||
|
ANTICOAGULANTS
|
83100020202015
|
Heparin Sodium (Porcine) Inj 1000 Unit/ML
|
|||||
|
83100020202018
|
Heparin Sodium (Porcine) Inj 2000 Unit/ML
|
||||||
|
83100020202020
|
Heparin Sodium (Porcine) Inj 2500 Unit/ML
|
||||||
|
83100020202025
|
Heparin Sodium (Porcine) Inj 5000 Unit/ML
|
||||||
|
83100020202034
|
Heparin Sodium (Porcine) PF Inj 5000 Unit/0.5ML
|
||||||
|
83100020202035
|
Heparin Sodium (Porcine) Inj 10000 Unit/ML
|
||||||
|
83100020202045
|
Heparin Sodium (Porcine) Inj 20000 Unit/ML
|
||||||
|
83101010102015
|
Dalteparin Sodium Inj 10000 Unit/ML
|
PA
|
|||||
|
83101010102020
|
Dalteparin Sodium Inj 2500 Unit/0.2ML
|
PA
|
16
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
83101010102040
|
Dalteparin Sodium Inj 5000 Unit/0.2ML
|
PA
|
|||||
|
83101010102045
|
Dalteparin Sodium Inj 7500 Unit/0.3ML
|
PA
|
|||||
|
83101010102053
|
Dalteparin Sodium Inj 12500 Unit/0.5ML
|
PA
|
|||||
|
83101010102056
|
Dalteparin Sodium Inj 15000 Unit/0.6ML
|
PA
|
|||||
|
83101010102060
|
Dalteparin Sodium Inj 18000 Unit/0.72ML
|
PA
|
|||||
|
83101010102065
|
Dalteparin Sodium Inj 25000 Unit/ML
|
PA
|
|||||
|
83101020102012
|
Enoxaparin Sodium Inj 30 MG/0.3ML
|
PA
|
|||||
|
83101020102013
|
Enoxaparin Sodium Inj 40 MG/0.4ML
|
PA
|
|||||
|
83101020102014
|
Enoxaparin Sodium Inj 60 MG/0.6ML
|
PA
|
|||||
|
83101020102015
|
Enoxaparin Sodium Inj 80 MG/0.8ML
|
PA
|
|||||
|
83101020102016
|
Enoxaparin Sodium Inj 100 MG/ML
|
PA
|
|||||
|
83101020102018
|
Enoxaparin Sodium Inj 120 MG/0.8ML
|
PA
|
|||||
|
83101020102020
|
Enoxaparin Sodium Inj 150 MG/ML
|
PA
|
|||||
|
83101020102050
|
Enoxaparin Sodium Inj 300 MG/3ML
|
PA
|
|||||
|
83103030102020
|
Fondaparinux Sodium Inj 2.5 MG/0.5ML
|
PA
|
|||||
|
83103030102035
|
Fondaparinux Sodium Inj 5 MG/0.4ML
|
PA
|
|||||
|
83103030102040
|
Fondaparinux Sodium Inj 7.5 MG/0.6ML
|
PA
|
|||||
|
83103030102045
|
Fondaparinux Sodium Inj 10 MG/0.8ML
|
PA
|
|||||
|
83200030200303
|
Warfarin Sodium Tab 1 MG
|
||||||
|
83200030200305
|
Warfarin Sodium Tab 2 MG
|
||||||
|
83200030200310
|
Warfarin Sodium Tab 2.5 MG
|
||||||
|
83200030200311
|
Warfarin Sodium Tab 3 MG
|
||||||
|
83200030200313
|
Warfarin Sodium Tab 4 MG
|
||||||
|
83200030200315
|
Warfarin Sodium Tab 5 MG
|
||||||
|
83200030200317
|
Warfarin Sodium Tab 6 MG
|
||||||
|
83200030200320
|
Warfarin Sodium Tab 7.5 MG
|
||||||
|
83200030200325
|
Warfarin Sodium Tab 10 MG
|
||||||
|
83370060000320
|
Rivaroxaban Tab 10 MG
|
PA
|
|||||
|
83370060000330
|
Rivaroxaban Tab 15 MG
|
PA
|
|||||
|
83370060000340
|
Rivaroxaban Tab 20 MG
|
PA
|
|||||
|
ANTICONVULSANTS
|
72100010000305
|
Clonazepam Tab 0.5 MG
|
17
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
72100010000310
|
Clonazepam Tab 1 MG
|
||||||
|
72100010000315
|
Clonazepam Tab 2 MG
|
||||||
|
72170070100302
|
Tiagabine HCl Tab 2 MG
|
||||||
|
72170070100305
|
Tiagabine HCl Tab 4 MG
|
||||||
|
72170070100315
|
Tiagabine HCl Tab 12 MG
|
||||||
|
72170070100320
|
Tiagabine HCl Tab 16 MG
|
||||||
|
72200030000505
|
Phenytoin Chew Tab 50 MG
|
||||||
|
72200030001810
|
Phenytoin Susp 125 MG/5ML
|
||||||
|
72200030200105
|
Phenytoin Sodium Extended Cap 30 MG
|
||||||
|
72200030200110
|
Phenytoin Sodium Extended Cap 100 MG
|
||||||
|
72200030200120
|
Phenytoin Sodium Extended Cap 200 MG
|
||||||
|
72200030200130
|
Phenytoin Sodium Extended Cap 300 MG
|
||||||
|
72400010000105
|
Ethosuximide Cap 250 MG
|
||||||
|
72400010002005
|
Ethosuximide Soln 250 MG/5ML
|
||||||
|
72500010100605
|
Divalproex Sodium Tab Delayed Release 125 MG
|
||||||
|
72500010100610
|
Divalproex Sodium Tab Delayed Release 250 MG
|
||||||
|
72500010100615
|
Divalproex Sodium Tab Delayed Release 500 MG
|
||||||
|
72500010107520
|
Divalproex Sodium Tab SR 24 HR 250 MG
|
||||||
|
72500010107530
|
Divalproex Sodium Tab SR 24 HR 500 MG
|
||||||
|
72500030000105
|
Valproic Acid Cap 250 MG
|
||||||
|
72600020000305
|
Carbamazepine Tab 200 MG
|
||||||
|
72600020000505
|
Carbamazepine Chew Tab 100 MG
|
||||||
|
72600020007410
|
Carbamazepine Tab SR 12HR 100 MG
|
||||||
|
72600020007420
|
Carbamazepine Tab SR 12HR 200 MG
|
||||||
|
72600020007440
|
Carbamazepine Tab SR 12HR 400 MG
|
||||||
|
72600030000110
|
Gabapentin Cap 100 MG
|
||||||
|
72600030000130
|
Gabapentin Cap 300 MG
|
||||||
|
72600030000140
|
Gabapentin Cap 400 MG
|
||||||
|
72600030000330
|
Gabapentin Tab 600 MG
|
||||||
|
72600030000340
|
Gabapentin Tab 800 MG
|
||||||
|
72600030002020
|
Gabapentin Oral Soln 250 MG/5ML
|
18
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
72600036000320
|
Lacosamide Tab 50 MG
|
PA
|
|||||
|
72600036000330
|
Lacosamide Tab 100 MG
|
PA
|
|||||
|
72600036000340
|
Lacosamide Tab 150 MG
|
PA
|
|||||
|
72600036000350
|
Lacosamide Tab 200 MG
|
PA
|
|||||
|
72600036002020
|
Lacosamide IV Inj 200 MG/20ML (10 MG/ML)
|
PA
|
|||||
|
72600036002060
|
Lacosamide Oral Solution 10 MG/ML
|
PA
|
|||||
|
72600040000310
|
Lamotrigine Tab 25 MG
|
||||||
|
72600040000330
|
Lamotrigine Tab 100 MG
|
||||||
|
72600040000335
|
Lamotrigine Tab 150 MG
|
||||||
|
72600040000340
|
Lamotrigine Tab 200 MG
|
||||||
|
72600040007225
|
Lamotrigine Orally Disintegrating Tab 25 MG
|
||||||
|
72600040007230
|
Lamotrigine Orally Disintegrating Tab 50 MG
|
||||||
|
72600040007240
|
Lamotrigine Orally Disintegrating Tab 100 MG
|
||||||
|
72600040007250
|
Lamotrigine Orally Disintegrating Tab 200 MG
|
||||||
|
72600043000320
|
Levetiracetam Tab 250 MG
|
||||||
|
72600043000330
|
Levetiracetam Tab 500 MG
|
||||||
|
72600043000340
|
Levetiracetam Tab 750 MG
|
||||||
|
72600043000350
|
Levetiracetam Tab 1000 MG
|
||||||
|
72600043002020
|
Levetiracetam Oral Soln 100 MG/ML
|
||||||
|
72600046000310
|
Oxcarbazepine Tab 150 MG
|
||||||
|
72600046000320
|
Oxcarbazepine Tab 300 MG
|
||||||
|
72600046000340
|
Oxcarbazepine Tab 600 MG
|
||||||
|
72600046001820
|
Oxcarbazepine Susp 300 MG/5ML (60 MG/ML)
|
||||||
|
72600057000110
|
Pregabalin Cap 25 MG
|
PA
|
|||||
|
72600057000115
|
Pregabalin Cap 50 MG
|
PA
|
|||||
|
72600057000120
|
Pregabalin Cap 75 MG
|
PA
|
|||||
|
72600057000125
|
Pregabalin Cap 100 MG
|
PA
|
|||||
|
72600057000135
|
Pregabalin Cap 150 MG
|
PA
|
|||||
|
72600057000145
|
Pregabalin Cap 200 MG
|
PA
|
|||||
|
72600057000150
|
Pregabalin Cap 225 MG
|
PA
|
|||||
|
72600057000160
|
Pregabalin Cap 300 MG
|
PA
|
19
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
72600057002020
|
Pregabalin Soln 20 MG/ML
|
PA
|
|||||
|
72600060000305
|
Primidone Tab 50 MG
|
||||||
|
72600060000310
|
Primidone Tab 250 MG
|
||||||
|
72600075000310
|
Topiramate Tab 25 MG
|
||||||
|
72600075000320
|
Topiramate Tab 50 MG
|
||||||
|
72600075000330
|
Topiramate Tab 100 MG
|
||||||
|
72600075000340
|
Topiramate Tab 200 MG
|
||||||
|
72600090000105
|
Zonisamide Cap 25 MG
|
||||||
|
72600090000110
|
Zonisamide Cap 50 MG
|
||||||
|
72600090000120
|
Zonisamide Cap 100 MG
|
||||||
|
ANTIDEPRESSANTS
|
AT LEAST TWO OF THE FOLLOWING
|
58160034100310
|
Escitalopram Oxalate Tab 5 MG (Base Equiv)
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160034100320
|
Escitalopram Oxalate Tab 10 MG (Base Equiv)
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160034100330
|
Escitalopram Oxalate Tab 20 MG (Base Equiv)
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160034102020
|
Escitalopram Oxalate Soln 5 MG/5ML (Base Equiv)
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160060007520
|
Paroxetine HCl Tab SR 24HR 12.5 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160060007530
|
Paroxetine HCl Tab SR 24HR 25 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160060007540
|
Paroxetine HCl Tab SR 24HR 37.5 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160070100305
|
Sertraline HCl Tab 25 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160070100310
|
Sertraline HCl Tab 50 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
58160070100320
|
Sertraline HCl Tab 100 MG
|
20
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST TWO OF THE FOLLOWING
|
58160070101320
|
Sertraline HCl Oral Conc 20 MG/ML
|
|||||
|
58030050000308
|
Mirtazapine Tab 7.5 MG
|
||||||
|
58030050000315
|
Mirtazapine Tab 15 MG
|
||||||
|
58030050000330
|
Mirtazapine Tab 30 MG
|
||||||
|
58030050000345
|
Mirtazapine Tab 45 MG
|
||||||
|
58120080100305
|
Trazodone HCl Tab 50 MG
|
||||||
|
58120080100310
|
Trazodone HCl Tab 100 MG
|
||||||
|
58120080100315
|
Trazodone HCl Tab 150 MG
|
||||||
|
58160020100310
|
Citalopram Hydrobromide Tab 10 MG (Base Equiv)
|
||||||
|
58160020100320
|
Citalopram Hydrobromide Tab 20 MG (Base Equiv)
|
||||||
|
58160020100340
|
Citalopram Hydrobromide Tab 40 MG (Base Equiv)
|
||||||
|
58160020102020
|
Citalopram Hydrobromide Oral Soln 10 MG/5ML
|
||||||
|
58160040000110
|
Fluoxetine HCl Cap 10 MG
|
||||||
|
58160040000120
|
Fluoxetine HCl Cap 20 MG
|
||||||
|
58160040000140
|
Fluoxetine HCl Cap 40 MG
|
||||||
|
58160040000310
|
Fluoxetine HCl Tab 10 MG
|
||||||
|
58160040000320
|
Fluoxetine HCl Tab 20 MG
|
||||||
|
58160040000360
|
Fluoxetine HCl Tab 60 MG
|
||||||
|
58160060000320
|
Paroxetine HCl Tab 20 MG
|
||||||
|
58160060000330
|
Paroxetine HCl Tab 30 MG
|
||||||
|
58160060000340
|
Paroxetine HCl Tab 40 MG
|
||||||
|
58180025106720
|
Duloxetine HCl Enteric Coated Pellets Cap 20 MG
|
PA (SALUD
MENTAL)
|
|||||
|
58180025106730
|
Duloxetine HCl Enteric Coated Pellets Cap 30 MG
|
PA (SALUD MENTAL)
|
|||||
|
58180025106750
|
Duloxetine HCl Enteric Coated Pellets Cap 60 MG
|
PA (SALUD MENTAL)
|
|||||
|
58180090100320
|
Venlafaxine HCl Tab 25 MG
|
||||||
|
58180090100340
|
Venlafaxine HCl Tab 37.5 MG
|
21
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
58180090100350
|
Venlafaxine HCl Tab 50 MG
|
||||||
|
58180090100360
|
Venlafaxine HCl Tab 75 MG
|
||||||
|
58180090100370
|
Venlafaxine HCl Tab 100 MG
|
||||||
|
58180090107020
|
Venlafaxine HCl Cap SR 24HR 37.5 MG (Base Equivalent)
|
||||||
|
58180090107030
|
Venlafaxine HCl Cap SR 24HR 75 MG (Base Equivalent)
|
||||||
|
58180090107050
|
Venlafaxine HCl Cap SR 24HR 150 MG (Base Equivalent)
|
||||||
|
58180090107510
|
Venlafaxine HCl Tab SR 24HR 37.5 MG (Base Equivalent)
|
||||||
|
58180090107520
|
Venlafaxine HCl Tab SR 24HR 75 MG (Base Equivalent)
|
||||||
|
58180090107530
|
Venlafaxine HCl Tab SR 24HR 150 MG (Base Equivalent)
|
||||||
|
58180090107540
|
Venlafaxine HCl Tab SR 24HR 225 MG (Base Equivalent)
|
||||||
|
58200010100305
|
Amitriptyline HCl Tab 10 MG
|
||||||
|
58200010100310
|
Amitriptyline HCl Tab 25 MG
|
||||||
|
58200010100315
|
Amitriptyline HCl Tab 50 MG
|
||||||
|
58200010100320
|
Amitriptyline HCl Tab 75 MG
|
||||||
|
58200010100325
|
Amitriptyline HCl Tab 100 MG
|
||||||
|
58200010100330
|
Amitriptyline HCl Tab 150 MG
|
||||||
|
58200025100120
|
Clomipramine HCl Cap 25 MG
|
||||||
|
58200025100130
|
Clomipramine HCl Cap 50 MG
|
||||||
|
58200025100140
|
Clomipramine HCl Cap 75 MG
|
||||||
|
58200030100305
|
Desipramine HCl Tab 10 MG
|
||||||
|
58200030100310
|
Desipramine HCl Tab 25 MG
|
||||||
|
58200030100315
|
Desipramine HCl Tab 50 MG
|
||||||
|
58200030100320
|
Desipramine HCl Tab 75 MG
|
||||||
|
58200030100325
|
Desipramine HCl Tab 100 MG
|
22
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
58200030100330
|
Desipramine HCl Tab 150 MG
|
||||||
|
58200040100105
|
Doxepin HCl Cap 10 MG
|
||||||
|
58200040100110
|
Doxepin HCl Cap 25 MG
|
||||||
|
58200040100115
|
Doxepin HCl Cap 50 MG
|
||||||
|
58200040100120
|
Doxepin HCl Cap 75 MG
|
||||||
|
58200040100125
|
Doxepin HCl Cap 100 MG
|
||||||
|
58200040100130
|
Doxepin HCl Cap 150 MG
|
||||||
|
58200040101305
|
Doxepin HCl Conc 10 MG/ML
|
||||||
|
58200050100305
|
Imipramine HCl Tab 10 MG
|
||||||
|
58200050100310
|
Imipramine HCl Tab 25 MG
|
||||||
|
58200050100315
|
Imipramine HCl Tab 50 MG
|
||||||
|
58200060100105
|
Nortriptyline HCl Cap 10 MG
|
||||||
|
58200060100110
|
Nortriptyline HCl Cap 25 MG
|
||||||
|
58200060100115
|
Nortriptyline HCl Cap 50 MG
|
||||||
|
58200060100120
|
Nortriptyline HCl Cap 75 MG
|
||||||
|
58200060102005
|
Nortriptyline HCl Soln 10 MG/5ML
|
||||||
|
58300040100305
|
Bupropion HCl Tab 75 MG
|
||||||
|
58300040107420
|
Bupropion HCl Tab SR 12HR 100 MG
|
||||||
|
58300040107430
|
Bupropion HCl Tab SR 12HR 150 MG
|
||||||
|
58300040107440
|
Bupropion HCl Tab SR 12HR 200 MG
|
||||||
|
58999002206320
|
*Bupropion HCl Tab 75 MG & Dietary Management Cap Pack***
|
||||||
|
ANTIDIABETICS
|
AT LEAST ONE OF THE FOLLOWING
|
27104003002020
|
Insulin Glargine Inj 100 Unit/ML
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27104006002020
|
Insulin Detemir Inj 100 Unit/ML
|
|||||
|
27607050100320
|
Pioglitazone HCl Tab 15 MG (Base Equiv)
|
||||||
|
27607050100330
|
Pioglitazone HCl Tab 30 MG (Base Equiv)
|
||||||
|
27607050100340
|
Pioglitazone HCl Tab 45 MG (Base Equiv)
|
||||||
|
27997802400320
|
Pioglitazone HCl-Glimepiride Tab 30-2 MG
|
23
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
27997802400340
|
Pioglitazone HCl-Glimepiride Tab 30-4 MG
|
||||||
|
27998002400320
|
Pioglitazone HCl-Metformin HCl Tab 15-500 MG
|
||||||
|
27998002400340
|
Pioglitazone HCl-Metformin HCl Tab 15-850 MG
|
||||||
|
AT LEAST ONE OF THE FOLLOWING
|
27500010000310
|
Acarbose Tab 25 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
27500010000320
|
Acarbose Tab 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
27500010000340
|
Acarbose Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
27500050000310
|
Miglitol Tab 25 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
27500050000320
|
Miglitol Tab 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
27500050000340
|
Miglitol Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550010100310
|
Alogliptin Benzoate Tab 6.25 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550010100320
|
Alogliptin Benzoate Tab 12.5 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550010100330
|
Alogliptin Benzoate Tab 25 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550050000320
|
Linagliptin Tab 5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550065100320
|
Saxagliptin HCl Tab 2.5 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550065100330
|
Saxagliptin HCl Tab 5 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550070100320
|
Sitagliptin Phosphate Tab 25 MG (Base Equiv)
|
ST
|
24
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
27550070100330
|
Sitagliptin Phosphate Tab 50 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27550070100340
|
Sitagliptin Phosphate Tab 100 MG (Base Equiv)
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502100320
|
Alogliptin-Metformin HCl Tab 12.5-500 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502100330
|
Alogliptin-Metformin HCl Tab 12.5-1000 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502400320
|
Linagliptin-Metformin HCl Tab 2.5-500 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502400330
|
Linagliptin-Metformin HCl Tab 2.5-850 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502400340
|
Linagliptin-Metformin HCl Tab 2.5-1000 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502607520
|
Saxagliptin-Metformin HCl Tab SR 24HR 2.5-1000 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502607530
|
Saxagliptin-Metformin HCl Tab SR 24HR 5-500 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502607540
|
Saxagliptin-Metformin HCl Tab SR 24HR 5-1000 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502700320
|
Sitagliptin-Metformin HCl Tab 50-500 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502700340
|
Sitagliptin-Metformin HCl Tab 50-1000 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502707520
|
Sitagliptin-Metformin HCl Tab SR 24HR 50-500 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502707530
|
Sitagliptin-Metformin HCl Tab SR 24HR 50-1000 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27992502707540
|
Sitagliptin-Metformin HCl Tab SR 24HR 100-1000 MG
|
ST
|
25
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
27994002100320
|
Alogliptin-Pioglitazone Tab 12.5-15 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27994002100325
|
Alogliptin-Pioglitazone Tab 12.5-30 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27994002100330
|
Alogliptin-Pioglitazone Tab 12.5-45 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27994002100340
|
Alogliptin-Pioglitazone Tab 25-15 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27994002100345
|
Alogliptin-Pioglitazone Tab 25-30 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
27994002100350
|
Alogliptin-Pioglitazone Tab 25-45 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING ULTRA SHORT ACTING AGENTS
|
27104002002020
|
Insulin Aspart Inj 100 Unit/ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING ULTRA SHORT ACTING AGENTS
|
27104004002022
|
Insulin Glulisine Inj 100 Unit/ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING ULTRA SHORT ACTING AGENTS
|
27104005002020
|
Insulin Lispro (Human) Inj 100 Unit/ML
|
|||||
|
27104010002005
|
INSULIN REGULAR (HUMAN) INJ 100 UNIT/ML
|
||||||
|
27104020001805
|
INSULIN ISOPHANE (HUMAN) INJ 100 UNIT/ML
|
||||||
|
27104090001810
|
INSULIN ISOPHANE & REGULAR (HUMAN) INJ 100 UNIT/ML (70-30)
|
||||||
|
27200027000310
|
Glimepiride Tab 1 MG
|
||||||
|
27200027000320
|
Glimepiride Tab 2 MG
|
||||||
|
27200027000340
|
Glimepiride Tab 4 MG
|
||||||
|
27200030000305
|
Glipizide Tab 5 MG
|
26
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
27200030000310
|
Glipizide Tab 10 MG
|
||||||
|
27200030007505
|
Glipizide Tab SR 24HR 2.5 MG
|
||||||
|
27200030007510
|
Glipizide Tab SR 24HR 5 MG
|
||||||
|
27200030007520
|
Glipizide Tab SR 24HR 10 MG
|
||||||
|
27200040000305
|
Glyburide Tab 1.25 MG
|
||||||
|
27200040000310
|
Glyburide Tab 2.5 MG
|
||||||
|
27200040000315
|
Glyburide Tab 5 MG
|
||||||
|
27250050000320
|
Metformin HCl Tab 500 MG
|
||||||
|
27250050000340
|
Metformin HCl Tab 850 MG
|
||||||
|
27250050000350
|
Metformin HCl Tab 1000 MG
|
||||||
|
27250050002020
|
Metformin HCl Oral Soln 500 MG/5ML
|
||||||
|
27250050007520
|
Metformin HCl Tab SR 24HR 500 MG
|
||||||
|
27250050007530
|
Metformin HCl Tab SR 24HR 750 MG
|
||||||
|
27300010106410
|
Glucagon (rDNA) For Inj Kit 1 MG
|
||||||
|
27300010152110
|
Glucagon HCl (rDNA) For Inj 1 MG (Base Equiv)
|
||||||
|
27574020100320
|
Bromocriptine Mesylate Tab 0.8 MG (Base Equivalent)
|
||||||
|
ANTIDIARRHEALS
|
47100010100310
|
Diphenoxylate w/ Atropine Tab 2.5-0.025 MG
|
|||||
|
47100010100910
|
Diphenoxylate w/ Atropine Liq 2.5-0.025 MG/5ML
|
||||||
|
47100020100105
|
Loperamide HCl Cap 2 MG
|
||||||
|
ANTIDOTES
|
93400030001920
|
Naltrexone For IM Extended Release Susp 380 MG
|
PA
|
||||
|
93400030100305
|
Naltrexone HCl Tab 50 MG
|
PA
|
|||||
|
ANTIEMETICS
|
AT LEAST ONE OF THE FOLLOWING
|
50250025200320
|
Dolasetron Mesylate Tab 50 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
50250025200330
|
Dolasetron Mesylate Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
50250035100310
|
Granisetron HCl Tab 1 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
50250065007220
|
Ondansetron Orally Disintegrating Tab 4 MG
|
PA
|
27
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
50250065007240
|
Ondansetron Orally Disintegrating Tab 8 MG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
50250065050310
|
Ondansetron HCl Tab 4 MG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
50250065050320
|
Ondansetron HCl Tab 8 MG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
50250065050340
|
Ondansetron HCl Tab 24 MG
|
PA
|
||||
|
50200070100120
|
Trimethobenzamide HCl Cap 300 MG
|
||||||
|
50200070102005
|
Trimethobenzamide HCl Inj 100 MG/ML
|
||||||
|
50280020000110
|
Aprepitant Capsule 40 MG
|
PA
|
|||||
|
50280020000120
|
Aprepitant Capsule 80 MG
|
PA
|
|||||
|
50280020000130
|
Aprepitant Capsule 125 MG
|
PA
|
|||||
|
ANTIFUNGALS
|
11000020000105
|
Flucytosine Cap 250 MG
|
|||||
|
11000020000110
|
Flucytosine Cap 500 MG
|
||||||
|
11000030100315
|
Griseofulvin Microsize Tab 500 MG
|
||||||
|
11000030101805
|
Griseofulvin Microsize Susp 125 MG/5ML
|
||||||
|
11000030200305
|
Griseofulvin Ultramicrosize Tab 125 MG
|
||||||
|
11000030200315
|
Griseofulvin Ultramicrosize Tab 250 MG
|
||||||
|
11000080100310
|
Terbinafine HCl Tab 250 MG
|
||||||
|
11000080103020
|
Terbinafine HCl Oral Granules Packet 125 MG
|
||||||
|
11000080103030
|
Terbinafine HCl Oral Granules Packet 187.5 MG
|
||||||
|
11404040000310
|
Ketoconazole Tab 200 MG
|
||||||
|
11407015000310
|
Fluconazole Tab 50 MG
|
||||||
|
11407015000320
|
Fluconazole Tab 100 MG
|
||||||
|
11407015000325
|
Fluconazole Tab 150 MG
|
||||||
|
11407015000330
|
Fluconazole Tab 200 MG
|
||||||
|
11407015001910
|
Fluconazole For Susp 10 MG/ML
|
||||||
|
11407015001940
|
Fluconazole For Susp 40 MG/ML
|
||||||
|
11407035000120
|
Itraconazole Cap 100 MG
|
VIH/SIDA
|
28
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
11407035000330
|
Itraconazole Tab 200 MG
|
VIH/SIDA
|
|||||
|
11407035002020
|
Itraconazole Oral Soln 10 MG/ML
|
VIH/SIDA
|
|||||
|
11407080000320
|
Voriconazole Tab 50 MG
|
PA
|
|||||
|
11407080000340
|
Voriconazole Tab 200 MG
|
PA
|
|||||
|
11407080001920
|
Voriconazole For Susp 40 MG/ML
|
PA
|
|||||
|
ANTIHISTAMINES
|
41200030102010
|
Diphenhydramine HCl Inj 50 MG/ML
|
|||||
|
41400020100305
|
Promethazine HCl Tab 12.5 MG
|
||||||
|
41400020100310
|
Promethazine HCl Tab 25 MG
|
||||||
|
41400020100315
|
Promethazine HCl Tab 50 MG
|
||||||
|
41400020101210
|
Promethazine HCl Syrup 6.25 MG/5ML
|
||||||
|
41400020102005
|
Promethazine HCl Inj 25 MG/ML
|
||||||
|
41400020102010
|
Promethazine HCl Inj 50 MG/ML
|
||||||
|
41400020105205
|
Promethazine HCl Suppos 12.5 MG
|
||||||
|
41400020105210
|
Promethazine HCl Suppos 25 MG
|
||||||
|
41400020105215
|
Promethazine HCl Suppos 50 MG
|
||||||
|
41550020102010
|
Cetirizine HCl Oral Soln 1 MG/ML (5 MG/5ML)
|
AL 6 MO-2 Y/O
|
|||||
|
41550024100310
|
FEXOFENADINE HCL TAB 30 MG
|
||||||
|
41550024100320
|
FEXOFENADINE HCL TAB 60 MG
|
||||||
|
41550024100350
|
FEXOFENADINE HCL TAB 180 MG
|
||||||
|
41550030000320
|
LORATADINE TAB 10 MG
|
||||||
|
41550030000520
|
LORATADINE CHEW TAB 5 MG
|
||||||
|
41550030001220
|
LORATADINE SYRUP 5 MG/5ML
|
||||||
|
41550030007210
|
LORATADINE ORALLY DISINTEGRATING TAB 5 MG
|
||||||
|
41550030007220
|
LORATADINE RAPIDLY-DISINTEGRATING TAB 10 MG
|
||||||
|
ANTIHYPERLIPIDEMICS
|
AT LEAST ONE OF THE FOLLOWING
|
39100010002905
|
Cholestyramine Powder 4 GM/DOSE
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
39100010003005
|
Cholestyramine Powder Packets 4 GM
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39100010102905
|
Cholestyramine Light Powder 4 GM/DOSE
|
29
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
39100010103005
|
Cholestyramine Light Powder Packets 4 GM
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39100020100320
|
Colestipol HCl Tab 1 GM
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39100020102705
|
Colestipol HCl Granules 5 GM
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39100020103010
|
Colestipol HCl Granule Packets 5 GM
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400010100310
|
Atorvastatin Calcium Tab 10 MG (Base Equivalent)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400010100320
|
Atorvastatin Calcium Tab 20 MG (Base Equivalent)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400010100330
|
Atorvastatin Calcium Tab 40 MG (Base Equivalent)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400010100350
|
Atorvastatin Calcium Tab 80 MG (Base Equivalent)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400060100305
|
Rosuvastatin Calcium Tab 5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400060100310
|
Rosuvastatin Calcium Tab 10 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400060100320
|
Rosuvastatin Calcium Tab 20 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400060100340
|
Rosuvastatin Calcium Tab 40 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400075000310
|
Simvastatin Tab 5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400075000320
|
Simvastatin Tab 10 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
39400075000330
|
Simvastatin Tab 20 MG
|
30
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
39400075000340
|
Simvastatin Tab 40 MG
|
|||||
|
39200025100104
|
Fenofibrate Micronized Cap 43 MG
|
||||||
|
39200025100107
|
Fenofibrate Micronized Cap 67 MG
|
||||||
|
39200025100114
|
Fenofibrate Micronized Cap 130 MG
|
||||||
|
39200025100115
|
Fenofibrate Micronized Cap 134 MG
|
||||||
|
39200025100130
|
Fenofibrate Micronized Cap 200 MG
|
||||||
|
39200030000310
|
Gemfibrozil Tab 600 MG
|
||||||
|
39300030000320
|
Ezetimibe Tab 10 MG
|
ST
|
|||||
|
39400065100320
|
Pravastatin Sodium Tab 10 MG
|
PA
|
|||||
|
39400065100330
|
Pravastatin Sodium Tab 20 MG
|
PA
|
|||||
|
39400065100340
|
Pravastatin Sodium Tab 40 MG
|
PA
|
|||||
|
39400065100360
|
Pravastatin Sodium Tab 80 MG
|
PA
|
|||||
|
39400075000360
|
Simvastatin Tab 80 MG
|
ST
|
|||||
|
ANTIHYPERTENSIVES
|
AT LEAST ONE OF THE FOLLOWING
|
36150020100310
|
Candesartan Cilexetil Tab 4 MG
|
ST
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
36150020100320
|
Candesartan Cilexetil Tab 8 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150020100330
|
Candesartan Cilexetil Tab 16 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150020100340
|
Candesartan Cilexetil Tab 32 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150030000310
|
Irbesartan Tab 75 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150030000320
|
Irbesartan Tab 150 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150030000340
|
Irbesartan Tab 300 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150040200320
|
Losartan Potassium Tab 25 MG
|
31
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
36150040200330
|
Losartan Potassium Tab 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150040200340
|
Losartan Potassium Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150070000310
|
Telmisartan Tab 20 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150070000320
|
Telmisartan Tab 40 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150070000340
|
Telmisartan Tab 80 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150080000310
|
Valsartan Tab 40 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150080000320
|
Valsartan Tab 80 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150080000330
|
Valsartan Tab 160 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36150080000340
|
Valsartan Tab 320 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002200320
|
Candesartan Cilexetil-Hydrochlorothiazide Tab 16-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002200340
|
Candesartan Cilexetil-Hydrochlorothiazide Tab 32-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002200350
|
Candesartan Cilexetil-Hydrochlorothiazide Tab 32-25 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002300320
|
Irbesartan-Hydrochlorothiazide Tab 150-12.5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002300340
|
Irbesartan-Hydrochlorothiazide Tab 300-12.5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002450320
|
Losartan Potassium & Hydrochlorothiazide Tab 50-12.5 MG
|
32
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002450325
|
Losartan Potassium & Hydrochlorothiazide Tab 100-12.5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002450340
|
Losartan Potassium & Hydrochlorothiazide Tab 100-25 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002600320
|
Telmisartan-Hydrochlorothiazide Tab 40-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002600340
|
Telmisartan-Hydrochlorothiazide Tab 80-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002600345
|
Telmisartan-Hydrochlorothiazide Tab 80-25 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002700320
|
Valsartan-Hydrochlorothiazide Tab 80-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002700340
|
Valsartan-Hydrochlorothiazide Tab 160-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002700350
|
Valsartan-Hydrochlorothiazide Tab 160-25 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002700360
|
Valsartan-Hydrochlorothiazide Tab 320-12.5 MG
|
ST
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
36994002700370
|
Valsartan-Hydrochlorothiazide Tab 320-25 MG
|
ST
|
||||
|
36100010000305
|
Captopril Tab 12.5 MG
|
||||||
|
36100010000310
|
Captopril Tab 25 MG
|
||||||
|
36100010000315
|
Captopril Tab 50 MG
|
||||||
|
36100010000320
|
Captopril Tab 100 MG
|
||||||
|
36100020100303
|
Enalapril Maleate Tab 2.5 MG
|
||||||
|
36100020100305
|
Enalapril Maleate Tab 5 MG
|
||||||
|
36100020100310
|
Enalapril Maleate Tab 10 MG
|
||||||
|
36100020100315
|
Enalapril Maleate Tab 20 MG
|
||||||
|
36100027100310
|
Fosinopril Sodium Tab 10 MG
|
||||||
|
36100027100320
|
Fosinopril Sodium Tab 20 MG
|
33
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
36100027100340
|
Fosinopril Sodium Tab 40 MG
|
||||||
|
36100030000303
|
Lisinopril Tab 2.5 MG
|
||||||
|
36100030000305
|
Lisinopril Tab 5 MG
|
||||||
|
36100030000310
|
Lisinopril Tab 10 MG
|
||||||
|
36100030000315
|
Lisinopril Tab 20 MG
|
||||||
|
36100030000324
|
Lisinopril Tab 30 MG
|
||||||
|
36100030000330
|
Lisinopril Tab 40 MG
|
||||||
|
36201010100305
|
Clonidine HCl Tab 0.1 MG
|
||||||
|
36201010100310
|
Clonidine HCl Tab 0.2 MG
|
||||||
|
36201010100315
|
Clonidine HCl Tab 0.3 MG
|
||||||
|
36201010108810
|
Clonidine HCl TD Patch Weekly 0.1 MG/24HR
|
||||||
|
36201010108820
|
Clonidine HCl TD Patch Weekly 0.2 MG/24HR
|
||||||
|
36201010108830
|
Clonidine HCl TD Patch Weekly 0.3 MG/24HR
|
||||||
|
36201030000310
|
Methyldopa Tab 250 MG
|
||||||
|
36201030000315
|
Methyldopa Tab 500 MG
|
||||||
|
36202005100310
|
Doxazosin Mesylate Tab 1 MG
|
||||||
|
36202005100320
|
Doxazosin Mesylate Tab 2 MG
|
||||||
|
36202005100330
|
Doxazosin Mesylate Tab 4 MG
|
||||||
|
36202005100340
|
Doxazosin Mesylate Tab 8 MG
|
||||||
|
36202040100105
|
Terazosin HCl Cap 1 MG
|
||||||
|
36202040100110
|
Terazosin HCl Cap 2 MG
|
||||||
|
36202040100115
|
Terazosin HCl Cap 5 MG
|
||||||
|
36202040100120
|
Terazosin HCl Cap 10 MG
|
||||||
|
36400010100305
|
Hydralazine HCl Tab 10 MG
|
||||||
|
36400010100310
|
Hydralazine HCl Tab 25 MG
|
||||||
|
36400010100315
|
Hydralazine HCl Tab 50 MG
|
||||||
|
36400010100320
|
Hydralazine HCl Tab 100 MG
|
||||||
|
36400020000305
|
Minoxidil Tab 2.5 MG
|
||||||
|
36400020000310
|
Minoxidil Tab 10 MG
|
||||||
|
36991802250310
|
Captopril & Hydrochlorothiazide Tab 25-15 MG
|
||||||
|
36991802250320
|
Captopril & Hydrochlorothiazide Tab 25-25 MG
|
34
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
36991802250330
|
Captopril & Hydrochlorothiazide Tab 50-15 MG
|
||||||
|
36991802250340
|
Captopril & Hydrochlorothiazide Tab 50-25 MG
|
||||||
|
36991802350305
|
Enalapril Maleate & Hydrochlorothiazide Tab 5-12.5 MG
|
||||||
|
36991802350310
|
Enalapril Maleate & Hydrochlorothiazide Tab 10-25 MG
|
||||||
|
36991802550305
|
Lisinopril & Hydrochlorothiazide Tab 10-12.5 MG
|
||||||
|
36991802550310
|
Lisinopril & Hydrochlorothiazide Tab 20-12.5 MG
|
||||||
|
36991802550320
|
Lisinopril & Hydrochlorothiazide Tab 20-25 MG
|
||||||
|
36992002100310
|
Atenolol & Chlorthalidone Tab 50-25 MG
|
||||||
|
36992002100320
|
Atenolol & Chlorthalidone Tab 100-25 MG
|
||||||
|
36992002200310
|
Metoprolol & Hydrochlorothiazide Tab 50-25 MG
|
||||||
|
36992002200320
|
Metoprolol & Hydrochlorothiazide Tab 100-25 MG
|
||||||
|
36992002200325
|
Metoprolol & Hydrochlorothiazide Tab 100-50 MG
|
||||||
|
36992002207520
|
Metoprolol & Hydrochlorothiazide Tab SR 24HR 25-12.5 MG
|
||||||
|
36992002207530
|
Metoprolol & Hydrochlorothiazide Tab SR 24HR 50-12.5 MG
|
||||||
|
36992002207540
|
Metoprolol & Hydrochlorothiazide Tab SR 24HR 100- 12.5 MG
|
||||||
|
36992002400310
|
Propranolol & Hydrochlorothiazide Tab 40-25 MG
|
||||||
|
36992002400320
|
Propranolol & Hydrochlorothiazide Tab 80-25 MG
|
||||||
|
ANTI-INFECTIVE AGENTS - MISC.
|
16000035000107
|
Metronidazole Cap 375 MG
|
|||||
|
16000035000305
|
Metronidazole Tab 250 MG
|
||||||
|
16000035000310
|
Metronidazole Tab 500 MG
|
||||||
|
16000045002170
|
Pentamidine Isethionate For Nebulization Soln 300 MG
|
||||||
|
16000060100110
|
Vancomycin HCl Cap 125 MG
|
||||||
|
16000060100120
|
Vancomycin HCl Cap 250 MG
|
||||||
|
16220020100105
|
Clindamycin HCl Cap 75 MG
|
||||||
|
16220020100110
|
Clindamycin HCl Cap 150 MG
|
35
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
16220020100120
|
Clindamycin HCl Cap 300 MG
|
||||||
|
16220020222120
|
Clindamycin Palmitate HCl For Soln 75 MG/5ML (Base Equiv)
|
||||||
|
16300010000310
|
Dapsone Tab 25 MG
|
||||||
|
16300010000320
|
Dapsone Tab 100 MG
|
||||||
|
16400020001820
|
Atovaquone Susp 750 MG/5ML
|
||||||
|
16400060000330
|
Nitazoxanide Tab 500 MG
|
PA
|
|||||
|
16400060001920
|
Nitazoxanide For Susp 100 MG/5ML
|
PA
|
|||||
|
16990002300310
|
Sulfamethoxazole-Trimethoprim Tab 400-80 MG
|
||||||
|
16990002300320
|
Sulfamethoxazole-Trimethoprim Tab 800-160 MG
|
||||||
|
16990002301810
|
Sulfamethoxazole-Trimethoprim Susp 200-40 MG/5ML
|
||||||
|
ANTIMALARIALS
|
13000010200305
|
Chloroquine Phosphate Tab 250 MG
|
|||||
|
13000010200310
|
Chloroquine Phosphate Tab 500 MG
|
||||||
|
13000020100305
|
Hydroxychloroquine Sulfate Tab 200 MG
|
||||||
|
13000025100310
|
Mefloquine HCl Tab 250 MG
|
||||||
|
13000030100310
|
Primaquine Phosphate Tab 26.3 MG
|
||||||
|
13000040000310
|
Pyrimethamine Tab 25 MG
|
||||||
|
ANTIMYASTHENIC/CHOLINERGIC AGENTS
|
76000040100305
|
NEOSTIGMINE BROMIDE TAB 15 MG
|
|||||
|
76000050100305
|
Pyridostigmine Bromide Tab 60 MG
|
||||||
|
76000050100405
|
Pyridostigmine Bromide Tab CR 180 MG
|
||||||
|
76000050101205
|
Pyridostigmine Bromide Syrup 60 MG/5ML
|
||||||
|
ANTIMYCOBACTERIAL AGENTS
|
09000020102105
|
Capreomycin Sulfate For Inj 1 GM
|
|||||
|
09000030000105
|
Cycloserine Cap 250 MG
|
||||||
|
09000040100305
|
Ethambutol HCl Tab 100 MG
|
||||||
|
09000040100310
|
Ethambutol HCl Tab 400 MG
|
||||||
|
09000050000310
|
Ethionamide Tab 250 MG
|
||||||
|
09000060000305
|
Isoniazid Tab 100 MG
|
||||||
|
09000060000310
|
Isoniazid Tab 300 MG
|
||||||
|
09000060001210
|
Isoniazid Syrup 50 MG/5ML
|
||||||
|
09000070000310
|
Pyrazinamide Tab 500 MG
|
36
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
09000075000120
|
Rifabutin Cap 150 MG
|
||||||
|
09000080000105
|
Rifampin Cap 150 MG
|
||||||
|
09000080000110
|
Rifampin Cap 300 MG
|
||||||
|
09990002100110
|
Isoniazid & Rifampin Cap 150-300 MG
|
||||||
|
ANTINEOPLASTICS AND ADJUNCTIVE THERAPIES
|
AT LEAST ONE OF THE FOLLOWING
|
21402420000320
|
Bicalutamide Tab 50 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
21402440000110
|
Flutamide Cap 125 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
21402810000310
|
Anastrozole Tab 1 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
21402835000320
|
Exemestane Tab 25 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
21402860000320
|
Letrozole Tab 2.5 MG
|
|||||
|
21100010000305
|
Busulfan Tab 2 MG
|
||||||
|
21100010002020
|
Busulfan Inj 6 MG/ML
|
||||||
|
21100015002030
|
Carboplatin IV Soln 50 MG/5ML
|
||||||
|
21100015002035
|
Carboplatin IV Soln 150 MG/15ML
|
||||||
|
21100015002040
|
Carboplatin IV Soln 450 MG/45ML
|
||||||
|
21100015002045
|
Carboplatin IV Soln 600 MG/60ML
|
||||||
|
21100015002120
|
Carboplatin IV For Inj 150 MG
|
||||||
|
21100020002020
|
Cisplatin Inj 50 MG/50ML (1 MG/ML)
|
||||||
|
21100020002025
|
Cisplatin Inj 100 MG/100ML (1 MG/ML)
|
||||||
|
21100020002030
|
Cisplatin Inj 200 MG/200ML (1 MG/ML)
|
||||||
|
21100028002025
|
Oxaliplatin IV Soln 50 MG/10ML
|
PA
|
|||||
|
21100028002030
|
Oxaliplatin IV Soln 100 MG/20ML
|
PA
|
|||||
|
21100028002035
|
Oxaliplatin IV Soln 200 MG/40ML
|
PA
|
|||||
|
21100028002120
|
Oxaliplatin For IV Inj 50 MG
|
PA
|
|||||
|
21100028002130
|
Oxaliplatin For IV Inj 100 MG
|
PA
|
37
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21101010000305
|
Chlorambucil Tab 2 MG
|
||||||
|
21101020000105
|
Cyclophosphamide Cap 25 MG
|
||||||
|
21101020000110
|
Cyclophosphamide Cap 50 MG
|
||||||
|
21101020000305
|
Cyclophosphamide Tab 25 MG
|
||||||
|
21101020000310
|
Cyclophosphamide Tab 50 MG
|
||||||
|
21101020002120
|
Cyclophosphamide For Inj 500 MG
|
||||||
|
21101020002125
|
Cyclophosphamide For Inj 1 GM
|
||||||
|
21101020002130
|
Cyclophosphamide For Inj 2 GM
|
||||||
|
21101025002110
|
Ifosfamide For Inj 1 GM
|
||||||
|
21101025002130
|
Ifosfamide For Inj 3 GM
|
||||||
|
21101030102105
|
Mechlorethamine HCl For Inj 10 MG
|
||||||
|
21101040000305
|
Melphalan Tab 2 MG
|
||||||
|
21101040102110
|
Melphalan HCl For Inj 50 MG (Base Equiv)
|
||||||
|
21102010002105
|
Carmustine For Inj 100 MG
|
||||||
|
21102020000110
|
Lomustine Cap 10 MG
|
||||||
|
21102020000115
|
Lomustine Cap 40 MG
|
||||||
|
21102020000120
|
Lomustine Cap 100 MG
|
||||||
|
21102030002105
|
Streptozocin For Inj 1 GM
|
||||||
|
21104070000110
|
Temozolomide Cap 5 MG
|
PA
|
|||||
|
21104070000120
|
Temozolomide Cap 20 MG
|
PA
|
|||||
|
21104070000140
|
Temozolomide Cap 100 MG
|
PA
|
|||||
|
21104070000143
|
Temozolomide Cap 140 MG
|
PA
|
|||||
|
21104070000147
|
Temozolomide Cap 180 MG
|
PA
|
|||||
|
21104070000150
|
Temozolomide Cap 250 MG
|
PA
|
|||||
|
21104070002120
|
Temozolomide For IV Soln 100 MG
|
PA
|
|||||
|
21200010102105
|
Bleomycin Sulfate For Inj 15 Unit
|
||||||
|
21200010102115
|
Bleomycin Sulfate For Inj 30 Unit
|
||||||
|
21200020002105
|
Dactinomycin For Inj 0.5 MG
|
||||||
|
21200030102210
|
Daunorubicin HCl Inj 5 MG/ML (Base Equiv)
|
||||||
|
21200040102010
|
Doxorubicin HCl Inj 2 MG/ML
|
||||||
|
21200040102105
|
Doxorubicin HCl For Inj 10 MG
|
38
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21200040102110
|
Doxorubicin HCl For Inj 20 MG
|
||||||
|
21200040102115
|
Doxorubicin HCl For Inj 50 MG
|
||||||
|
21200045102025
|
Idarubicin HCl IV Inj 5 MG/5ML (1 MG/ML)
|
||||||
|
21200045102030
|
Idarubicin HCl IV Inj 10 MG/10ML (1 MG/ML)
|
||||||
|
21200045102035
|
Idarubicin HCl IV Inj 20 MG/20ML (1 MG/ML)
|
||||||
|
21200050002105
|
Mitomycin For Inj 5 MG
|
||||||
|
21200050002110
|
Mitomycin For Inj 20 MG
|
||||||
|
21200050002120
|
Mitomycin For Inj 40 MG
|
||||||
|
21200055001320
|
Mitoxantrone HCl Inj Conc 20 MG/10ML (2 MG/ML)
|
PA
|
|||||
|
21200055001325
|
Mitoxantrone HCl Inj Conc 25 MG/12.5ML (2 MG/ML)
|
PA
|
|||||
|
21200055001330
|
Mitoxantrone HCl Inj Conc 30 MG/15ML (2 MG/ML)
|
PA
|
|||||
|
21250010402120
|
Asparaginase Erwinia Chrysanthemi For IM Inj 10000 Unit
|
||||||
|
21300005000320
|
Capecitabine Tab 150 MG
|
PA
|
|||||
|
21300005000350
|
Capecitabine Tab 500 MG
|
PA
|
|||||
|
21300007002010
|
Cladribine Inj 1 MG/ML
|
||||||
|
21300010002010
|
Cytarabine Inj 20 MG/ML
|
||||||
|
21300010002011
|
Cytarabine Inj PF 20 MG/ML
|
||||||
|
21300010002040
|
Cytarabine Inj PF 100 MG/ML
|
||||||
|
21300010002105
|
Cytarabine For Inj 100 MG
|
||||||
|
21300010002110
|
Cytarabine For Inj 500 MG
|
||||||
|
21300010002115
|
Cytarabine For Inj 1 GM
|
||||||
|
21300025102020
|
Fludarabine Phosphate Inj 25 MG/ML
|
||||||
|
21300025102120
|
Fludarabine Phosphate For Inj 50 MG
|
||||||
|
21300030002020
|
Fluorouracil Inj 500 MG/10ML (50 MG/ML)
|
||||||
|
21300030002025
|
Fluorouracil Inj 1 GM/20ML (50 MG/ML)
|
||||||
|
21300030002030
|
Fluorouracil Inj 2.5 GM/50ML (50 MG/ML)
|
||||||
|
21300030002035
|
Fluorouracil Inj 5 GM/100ML (50 MG/ML)
|
||||||
|
21300034102020
|
Gemcitabine HCl Inj 200 MG/5.26ML (38 MG/ML) (Base Equiv)
|
39
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21300034102040
|
Gemcitabine HCl Inj 1 GM/26.3ML (38 MG/ML) (Base Equiv)
|
||||||
|
21300034102060
|
Gemcitabine HCl Inj 2 GM/52.6ML (38 MG/ML) (Base Equiv)
|
||||||
|
21300034102110
|
Gemcitabine HCl For Inj 200 MG
|
||||||
|
21300034102140
|
Gemcitabine HCl For Inj 1 GM
|
||||||
|
21300034102160
|
Gemcitabine HCl For Inj 2 GM
|
||||||
|
21300040000305
|
Mercaptopurine Tab 50 MG
|
||||||
|
21300040001830
|
Mercaptopurine Susp 2000 MG/100ML (20 MG/ML)
|
||||||
|
21300050100310
|
Methotrexate Sodium Tab 2.5 MG (Base Equiv)
|
||||||
|
21300050100320
|
Methotrexate Sodium Tab 5 MG (Base Equiv)
|
||||||
|
21300050100330
|
Methotrexate Sodium Tab 7.5 MG (Base Equiv)
|
||||||
|
21300050100340
|
Methotrexate Sodium Tab 10 MG (Base Equiv)
|
||||||
|
21300050100350
|
Methotrexate Sodium Tab 15 MG (Base Equiv)
|
||||||
|
21300050102030
|
Methotrexate Sodium Inj 25 MG/ML
|
||||||
|
21300050102031
|
Methotrexate Sodium Inj PF 25 MG/ML
|
||||||
|
21300050102150
|
Methotrexate Sodium For Inj 1 GM
|
||||||
|
21300060000305
|
Thioguanine Tab 40 MG
|
||||||
|
21335020002025
|
Bevacizumab IV Soln 100 MG/4ML (For Infusion)
|
||||||
|
21335020002030
|
Bevacizumab IV Soln 400 MG/16ML (For Infusion)
|
||||||
|
21353025002020
|
Cetuximab IV Soln 100 MG/50ML (2 MG/ML)
|
PA
|
|||||
|
21353025002025
|
Cetuximab IV Soln 200 MG/100ML (2 MG/ML)
|
PA
|
|||||
|
21353070002120
|
Trastuzumab For IV Soln 440 MG
|
||||||
|
21402250000320
|
Mitotane Tab 500 MG
|
||||||
|
21402680100310
|
Tamoxifen Citrate Tab 10 MG (Base Equivalent)
|
||||||
|
21402680100320
|
Tamoxifen Citrate Tab 20 MG (Base Equivalent)
|
||||||
|
21402680102020
|
Tamoxifen Citrate Oral Soln 10 MG/5ML (Base Equivalent)
|
||||||
|
21403020100105
|
Estramustine Phosphate Sodium Cap 140 MG
|
||||||
|
21404010101840
|
Medroxyprogesterone Acetate IM Susp 400 MG/ML
|
PA
|
|||||
|
21404020100305
|
Megestrol Acetate Tab 20 MG
|
40
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21404020100310
|
Megestrol Acetate Tab 40 MG
|
||||||
|
21404020101810
|
Megestrol Acetate Susp 40 MG/ML
|
||||||
|
21405005102310
|
Goserelin Acetate Implant 3.6 MG
|
PA
|
|||||
|
21405005102330
|
Goserelin Acetate Implant 10.8 MG
|
PA
|
|||||
|
21405010106405
|
Leuprolide Acetate For Inj Kit 3.75 MG
|
PA
|
|||||
|
21405010106407
|
Leuprolide Acetate Inj Kit 5 MG/ML
|
PA
|
|||||
|
21405010106410
|
Leuprolide Acetate For Inj Kit 7.5 MG
|
PA
|
|||||
|
21405010106415
|
Leuprolide Acetate For Subcutaneous Inj Kit 7.5 MG
|
PA
|
|||||
|
21405010156420
|
Leuprolide Acetate (3 Month) For Inj Kit 11.25 MG
|
PA
|
|||||
|
21405010156430
|
Leuprolide Acetate (3 Month) For Inj Kit 22.5 MG
|
PA
|
|||||
|
21405010156432
|
Leuprolide Acetate (3 Month) For Subcutaneous Inj Kit 22.5MG
|
PA
|
|||||
|
21405010206430
|
Leuprolide Acetate (4 Month) For Inj Kit 30 MG
|
PA
|
|||||
|
21405010206435
|
Leuprolide Acetate (4 Month) For Subcutaneous Inj Kit 30 MG
|
PA
|
|||||
|
21405010256445
|
Leuprolide Acetate (6 Month) For Subcutaneous Inj Kit 45 MG
|
PA
|
|||||
|
21405010256450
|
Leuprolide Acetate (6 Month) For Inj Kit 45 MG
|
PA
|
|||||
|
21500005001310
|
Docetaxel For Inj Conc 20 MG/ML
|
||||||
|
21500005001315
|
Docetaxel For Inj Conc 80 MG/4ML (20 MG/ML)
|
||||||
|
21500005001316
|
Docetaxel For Inj Conc 140 MG/7ML (20 MG/ML)
|
||||||
|
21500005001317
|
Docetaxel For Inj Conc 160 MG/8ML (20 MG/ML)
|
||||||
|
21500005001320
|
Docetaxel For Inj Conc 20 MG/0.5ML (40 MG/ML)
|
||||||
|
21500005001325
|
Docetaxel For Inj Conc 80 MG/2ML (40 MG/ML)
|
||||||
|
21500005002030
|
Docetaxel Soln for IV Infusion 20 MG/2ML
|
||||||
|
21500005002040
|
Docetaxel Soln for IV Infusion 80 MG/8ML
|
||||||
|
21500005002050
|
Docetaxel Soln for IV Infusion 160 MG/16ML
|
||||||
|
21500005002120
|
Docetaxel For Inj 20 MG
|
||||||
|
21500005002140
|
Docetaxel For Inj 80 MG
|
||||||
|
21500010000120
|
Etoposide Cap 50 MG
|
||||||
|
21500010002025
|
Etoposide Inj 100MG/5ML (20 MG/ML)
|
41
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21500010002030
|
Etoposide Inj 500MG/25ML (20 MG/ML)
|
||||||
|
21500010002040
|
Etoposide Inj 1 GM/50ML (20 MG/ML)
|
||||||
|
21500010602120
|
Etoposide Phosphate IV For Inj 100 MG
|
||||||
|
21500012001325
|
Paclitaxel IV Conc 30 MG/5ML (6 MG/ML)
|
||||||
|
21500012001335
|
Paclitaxel IV Conc 100 MG/16.7ML (6 MG/ML)
|
||||||
|
21500012001340
|
Paclitaxel IV Conc 150 MG/25ML (6 MG/ML)
|
||||||
|
21500012001350
|
Paclitaxel IV Conc 300 MG/50ML (6 MG/ML)
|
||||||
|
21500015002020
|
Teniposide IV Soln 10 MG/ML
|
||||||
|
21500020102005
|
Vincristine Sulfate IV Soln 1 MG/ML
|
||||||
|
21500020201820
|
Vincristine Sulfate Liposome IV Susp 5 MG/31ML (0.16 MG/ML)
|
||||||
|
21500030102020
|
Vinblastine Sulfate Inj 1 MG/ML
|
||||||
|
21500030102105
|
Vinblastine Sulfate For Inj 10 MG
|
||||||
|
21500050802020
|
Vinorelbine Tartrate Inj 10 MG/ML
|
||||||
|
21500050802025
|
Vinorelbine Tartrate Inj 50 MG/5ML (10 MG/ML)
|
||||||
|
21532025100120
|
Dabrafenib Mesylate Cap 50 MG (Base Equivalent)
|
PA
|
|||||
|
21532025100130
|
Dabrafenib Mesylate Cap 75 MG (Base Equivalent)
|
PA
|
|||||
|
21532080000320
|
Vemurafenib Tab 240 MG (Base Equivalent)
|
PA
|
|||||
|
21532530000310
|
Everolimus Tab 2.5 MG
|
PA
|
|||||
|
21532530000320
|
Everolimus Tab 5 MG
|
PA
|
|||||
|
21532530000325
|
Everolimus Tab 7.5 MG
|
PA
|
|||||
|
21532530000330
|
Everolimus Tab 10 MG
|
PA
|
|||||
|
21532530007310
|
Everolimus Tab for Oral Susp 2 MG
|
PA
|
|||||
|
21532530007320
|
Everolimus Tab for Oral Susp 3 MG
|
PA
|
|||||
|
21532530007340
|
Everolimus Tab for Oral Susp 5 MG
|
PA
|
|||||
|
21532570002020
|
Temsirolimus Soln For IV Infusion 25 MG/ML
|
||||||
|
21533050000320
|
Regorafenib Tab 40 MG
|
PA
|
|||||
|
21533060400320
|
Sorafenib Tosylate Tab 200 MG (Base Equivalent)
|
PA
|
|||||
|
21533070300120
|
Sunitinib Malate Cap 12.5 MG (Base Equivalent)
|
PA
|
|||||
|
21533070300130
|
Sunitinib Malate Cap 25 MG (Base Equivalent)
|
PA
|
|||||
|
21533070300135
|
Sunitinib Malate Cap 37.5 MG (Base Equivalent)
|
PA
|
42
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21533070300140
|
Sunitinib Malate Cap 50 MG (Base Equivalent)
|
PA
|
|||||
|
21533570100310
|
Trametinib Dimethyl Sulfoxide Tab 0.5 MG (Base Equivalent)
|
PA
|
|||||
|
21533570100330
|
Trametinib Dimethyl Sulfoxide Tab 2 MG (Base Equivalent)
|
PA
|
|||||
|
21534012000320
|
Bosutinib Tab 100 MG
|
PA
|
|||||
|
21534012000340
|
Bosutinib Tab 500 MG
|
PA
|
|||||
|
21534020000320
|
Dasatinib Tab 20 MG
|
PA
|
|||||
|
21534020000340
|
Dasatinib Tab 50 MG
|
PA
|
|||||
|
21534020000350
|
Dasatinib Tab 70 MG
|
PA
|
|||||
|
21534020000354
|
Dasatinib Tab 80 MG
|
PA
|
|||||
|
21534020000360
|
Dasatinib Tab 100 MG
|
PA
|
|||||
|
21534020000380
|
Dasatinib Tab 140 MG
|
PA
|
|||||
|
21534025000320
|
Erlotinib Tab 25 MG
|
PA
|
|||||
|
21534025000340
|
Erlotinib Tab 100 MG
|
PA
|
|||||
|
21534025000360
|
Erlotinib Tab 150 MG
|
PA
|
|||||
|
21534035100320
|
Imatinib Mesylate Tab 100 MG (Base Equivalent)
|
PA
|
|||||
|
21534035100340
|
Imatinib Mesylate Tab 400 MG (Base Equivalent)
|
PA
|
|||||
|
21534050100320
|
Lapatinib Ditosylate Tab 250 MG (Base Equiv)
|
||||||
|
21534060000115
|
Nilotinib Cap 150 MG
|
PA
|
|||||
|
21534060000120
|
Nilotinib Cap 200 MG
|
PA
|
|||||
|
21534070100320
|
Pazopanib HCl Tab 200 MG (Base Equiv)
|
PA
|
|||||
|
21536015002120
|
Bortezomib For Inj 3.5 MG
|
||||||
|
21550040102025
|
Irinotecan HCl Inj 40 MG/2ML (20 MG/ML)
|
||||||
|
21550040102030
|
Irinotecan HCl Inj 100 MG/5ML (20 MG/ML)
|
||||||
|
21550040102035
|
Irinotecan HCl Inj 300 MG/15ML (20 MG/ML)
|
||||||
|
21550040102040
|
Irinotecan HCl Inj 500 MG/25ML (20 MG/ML)
|
||||||
|
21700013001930
|
BCG Live Intravesical For Susp 50 MG
|
||||||
|
21700013001940
|
BCG Live Intravesical For Susp 81 MG/VIAL
|
||||||
|
21700020002105
|
Dacarbazine For Inj 100 MG
|
||||||
|
21700020002110
|
Dacarbazine For Inj 200 MG
|
43
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
21700030000105
|
Hydroxyurea Cap 500 MG
|
||||||
|
21700050100105
|
Procarbazine HCl Cap 50 MG
|
||||||
|
21700060202022
|
Interferon Alfa-2B Inj 6000000 Unit/ML
|
PA
|
|||||
|
21700060202030
|
Interferon Alfa-2B Inj 10000000 Unit/ML
|
PA
|
|||||
|
21700060202130
|
Interferon Alfa-2B For Inj 10000000 Unit
|
PA
|
|||||
|
21700060202135
|
Interferon Alfa-2B For Inj 18000000 Unit
|
PA
|
|||||
|
21700060202160
|
Interferon Alfa-2B For Inj 50000000 Unit
|
PA
|
|||||
|
21700060702020
|
Interferon Gamma-1B Inj 100 MCG/0.5ML (2000000 Unit/0.5ML)
|
PA
|
|||||
|
21754040002120
|
Dexrazoxane For Inj 250 MG
|
||||||
|
21754040002140
|
Dexrazoxane For Inj 500 MG
|
||||||
|
21755040100310
|
Leucovorin Calcium Tab 5 MG
|
||||||
|
21755040100325
|
Leucovorin Calcium Tab 10 MG
|
||||||
|
21755040100335
|
Leucovorin Calcium Tab 15 MG
|
||||||
|
21755040100345
|
Leucovorin Calcium Tab 25 MG
|
||||||
|
21755040102050
|
Leucovorin Calcium Inj 300 MG/30ML (10 MG/ML)
|
||||||
|
21755040102120
|
Leucovorin Calcium For Inj 50 MG
|
||||||
|
21755040102130
|
Leucovorin Calcium For Inj 100 MG
|
||||||
|
21755040102150
|
Leucovorin Calcium For Inj 200 MG
|
||||||
|
21755040102160
|
Leucovorin Calcium For Inj 350 MG
|
||||||
|
21755040102170
|
Leucovorin Calcium For Inj 500 MG
|
||||||
|
21990002406440
|
Ifosfamide & Mesna Inj Kit 1000-1000 MG
|
||||||
|
ANTIPARKINSON AGENTS
|
AT LEAST ONE OF THE FOLLOWING
|
73203060100305
|
Pramipexole Dihydrochloride Tab 0.125 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203060100310
|
Pramipexole Dihydrochloride Tab 0.25 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203060100315
|
Pramipexole Dihydrochloride Tab 0.5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203060100317
|
Pramipexole Dihydrochloride Tab 0.75 MG
|
44
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
73203060100320
|
Pramipexole Dihydrochloride Tab 1 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203060100330
|
Pramipexole Dihydrochloride Tab 1.5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100310
|
Ropinirole Hydrochloride Tab 0.25 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100315
|
Ropinirole Hydrochloride Tab 0.5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100320
|
Ropinirole Hydrochloride Tab 1 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100330
|
Ropinirole Hydrochloride Tab 2 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100337
|
Ropinirole Hydrochloride Tab 3 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100344
|
Ropinirole Hydrochloride Tab 4 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
73203070100350
|
Ropinirole Hydrochloride Tab 5 MG
|
|||||
|
73100010100305
|
Benztropine Mesylate Tab 0.5 MG
|
||||||
|
73100010100310
|
Benztropine Mesylate Tab 1 MG
|
||||||
|
73100010100315
|
Benztropine Mesylate Tab 2 MG
|
||||||
|
73100070100310
|
Trihexyphenidyl HCl Tab 2 MG
|
||||||
|
73100070100320
|
Trihexyphenidyl HCl Tab 5 MG
|
||||||
|
73100070101005
|
Trihexyphenidyl HCl Elixir 0.4 MG/ML
|
||||||
|
73200010100105
|
Amantadine HCl Cap 100 MG
|
||||||
|
73200010100310
|
Amantadine HCl Tab 100 MG
|
||||||
|
73200010101205
|
Amantadine HCl Syrup 50 MG/5ML
|
||||||
|
73200020100105
|
Bromocriptine Mesylate Cap 5 MG
|
||||||
|
73200020100305
|
Bromocriptine Mesylate Tab 2.5 MG
|
||||||
|
73209902100310
|
Carbidopa & Levodopa Tab 10-100 MG
|
45
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
73209902100320
|
Carbidopa & Levodopa Tab 25-100 MG
|
||||||
|
73209902100330
|
Carbidopa & Levodopa Tab 25-250 MG
|
||||||
|
73209902100410
|
Carbidopa & Levodopa Tab CR 25-100 MG
|
||||||
|
73209902100420
|
Carbidopa & Levodopa Tab CR 50-200 MG
|
||||||
|
73209903300320
|
Carbidopa-Levodopa-Entacapone Tabs 12.5-50-200 MG
|
||||||
|
73209903300325
|
Carbidopa-Levodopa-Entacapone Tabs 18.75-75-200 MG
|
||||||
|
73209903300330
|
Carbidopa-Levodopa-Entacapone Tabs 25-100-200 MG
|
||||||
|
73209903300335
|
Carbidopa-Levodopa-Entacapone Tabs 31.25-125-200 MG
|
||||||
|
73209903300340
|
Carbidopa-Levodopa-Entacapone Tabs 37.5-150-200 MG
|
||||||
|
73209903300350
|
Carbidopa-Levodopa-Entacapone Tabs 50-200-200 MG
|
||||||
|
73300030100320
|
Selegiline HCl Tab 5 MG
|
||||||
|
ANTIPSYCHOTICS/ANTIMANIC AGENTS
|
AT LEAST THREE OF THE FOLLOWING
|
59070070000303
|
Risperidone Tab 0.25 MG
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59070070000306
|
Risperidone Tab 0.5 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59070070000310
|
Risperidone Tab 1 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59070070000320
|
Risperidone Tab 2 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59070070000330
|
Risperidone Tab 3 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59070070000340
|
Risperidone Tab 4 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59070070002010
|
Risperidone Soln 1 MG/ML
|
46
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070100310
|
Quetiapine Fumarate Tab 25 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070100314
|
Quetiapine Fumarate Tab 50 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070100320
|
Quetiapine Fumarate Tab 100 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070100330
|
Quetiapine Fumarate Tab 200 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070100340
|
Quetiapine Fumarate Tab 300 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070100350
|
Quetiapine Fumarate Tab 400 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070107505
|
Quetiapine Fumarate Tab SR 24HR 50 MG
|
PA, LC= 3 Days
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070107515
|
Quetiapine Fumarate Tab SR 24HR 150 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070107520
|
Quetiapine Fumarate Tab SR 24HR 200 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070107530
|
Quetiapine Fumarate Tab SR 24HR 300 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59153070107540
|
Quetiapine Fumarate Tab SR 24HR 400 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060000305
|
Olanzapine Tab 2.5 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060000310
|
Olanzapine Tab 5 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060000315
|
Olanzapine Tab 7.5 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060000320
|
Olanzapine Tab 10 MG
|
47
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060000330
|
Olanzapine Tab 15 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060000340
|
Olanzapine Tab 20 MG
|
|||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060007210
|
Olanzapine Orally Disintegrating Tab 5 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060007220
|
Olanzapine Orally Disintegrating Tab 10 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060007230
|
Olanzapine Orally Disintegrating Tab 15 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59157060007240
|
Olanzapine Orally Disintegrating Tab 20 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015000310
|
Aripiprazole Tab 5 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015000320
|
Aripiprazole Tab 10 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015000330
|
Aripiprazole Tab 15 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015000340
|
Aripiprazole Tab 20 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015000350
|
Aripiprazole Tab 30 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015002020
|
Aripiprazole Oral Solution 1 MG/ML
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015007220
|
Aripiprazole Orally Disintegrating Tab 10 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59250015007230
|
Aripiprazole Orally Disintegrating Tab 15 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400023100310
|
Lurasidone HCl Tab 20 MG
|
PA
|
48
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST THREE OF THE FOLLOWING
|
59400023100320
|
Lurasidone HCl Tab 40 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400023100330
|
Lurasidone HCl Tab 60 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400023100340
|
Lurasidone HCl Tab 80 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400023100350
|
Lurasidone HCl Tab 120 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400085100120
|
Ziprasidone HCl Cap 20 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400085100130
|
Ziprasidone HCl Cap 40 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400085100140
|
Ziprasidone HCl Cap 60 MG
|
PA
|
||||
|
AT LEAST THREE OF THE FOLLOWING
|
59400085100150
|
Ziprasidone HCl Cap 80 MG
|
PA
|
||||
|
59100010100305
|
Haloperidol Tab 0.5 MG
|
||||||
|
59100010100310
|
Haloperidol Tab 1 MG
|
||||||
|
59100010100315
|
Haloperidol Tab 2 MG
|
||||||
|
59100010100320
|
Haloperidol Tab 5 MG
|
||||||
|
59100010100325
|
Haloperidol Tab 10 MG
|
||||||
|
59100010100330
|
Haloperidol Tab 20 MG
|
||||||
|
59100010201305
|
Haloperidol Lactate Oral Conc 2 MG/ML
|
||||||
|
59100010302010
|
Haloperidol Decanoate IM Soln 50 MG/ML
|
||||||
|
59100010302020
|
Haloperidol Decanoate IM Soln 100 MG/ML
|
||||||
|
59152020000320
|
Clozapine Tab 25 MG
|
PA
|
|||||
|
59152020000330
|
Clozapine Tab 100 MG
|
PA
|
|||||
|
59200015100305
|
Chlorpromazine HCl Tab 10 MG
|
||||||
|
59200015100310
|
Chlorpromazine HCl Tab 25 MG
|
||||||
|
59200015100315
|
Chlorpromazine HCl Tab 50 MG
|
49
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
59200015100320
|
Chlorpromazine HCl Tab 100 MG
|
||||||
|
59200015100325
|
Chlorpromazine HCl Tab 200 MG
|
||||||
|
59200025100305
|
Fluphenazine HCl Tab 1 MG
|
||||||
|
59200025100310
|
Fluphenazine HCl Tab 2.5 MG
|
||||||
|
59200025100315
|
Fluphenazine HCl Tab 5 MG
|
||||||
|
59200025100320
|
Fluphenazine HCl Tab 10 MG
|
||||||
|
59200025101005
|
Fluphenazine HCl Elixir 2.5 MG/5ML
|
||||||
|
59200025101320
|
Fluphenazine HCl Oral Conc 5 MG/ML
|
||||||
|
59200025302005
|
Fluphenazine Decanoate Inj 25 MG/ML
|
||||||
|
59200045000305
|
Perphenazine Tab 2 MG
|
||||||
|
59200045000310
|
Perphenazine Tab 4 MG
|
||||||
|
59200045000315
|
Perphenazine Tab 8 MG
|
||||||
|
59200045000320
|
Perphenazine Tab 16 MG
|
||||||
|
59200055005215
|
Prochlorperazine Suppos 25 MG
|
||||||
|
59200055100305
|
Prochlorperazine Maleate Tab 5 MG
|
||||||
|
59200055100310
|
Prochlorperazine Maleate Tab 10 MG
|
||||||
|
59200055202005
|
Prochlorperazine Edisylate Inj 5 MG/ML
|
||||||
|
59200080100305
|
Thioridazine HCl Tab 10 MG
|
||||||
|
59200080100315
|
Thioridazine HCl Tab 25 MG
|
||||||
|
59200080100320
|
Thioridazine HCl Tab 50 MG
|
||||||
|
59200080100325
|
Thioridazine HCl Tab 100 MG
|
||||||
|
59200085100305
|
Trifluoperazine HCl Tab 1 MG
|
||||||
|
59200085100310
|
Trifluoperazine HCl Tab 2 MG
|
||||||
|
59200085100315
|
Trifluoperazine HCl Tab 5 MG
|
||||||
|
59200085100320
|
Trifluoperazine HCl Tab 10 MG
|
||||||
|
59300020100105
|
Thiothixene Cap 1 MG
|
||||||
|
59300020100110
|
Thiothixene Cap 2 MG
|
||||||
|
59300020100115
|
Thiothixene Cap 5 MG
|
||||||
|
59300020100120
|
Thiothixene Cap 10 MG
|
||||||
|
59500010100103
|
Lithium Carbonate Cap 150 MG
|
||||||
|
59500010100105
|
Lithium Carbonate Cap 300 MG
|
50
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
59500010100110
|
Lithium Carbonate Cap 600 MG
|
||||||
|
59500010100305
|
Lithium Carbonate Tab 300 MG
|
||||||
|
59500010202010
|
Lithium Citrate Oral Soln 8 mEq/5ML
|
||||||
|
ANTISEPTICS & DISINFECTANTS
|
92100030102060
|
Chlorhexidine Gluconate Soln 20%
|
|||||
|
ANTIVIRALS
|
THE FOLLOWING PROTEASE INHIBITORS ARE COVERED ONLY THROUGH OCASET (PUERTO RICO DEPARTMENT OF HEALTH)
|
12104515200130
|
Atazanavir Sulfate Cap 150 MG (Base Equiv)
|
||||
|
12104515200140
|
Atazanavir Sulfate Cap 200 MG (Base Equiv)
|
||||||
|
12104515200150
|
Atazanavir Sulfate Cap 300 MG (Base Equiv)
|
||||||
|
12104520100310
|
Darunavir Ethanolate Tab 75 MG (Base Equiv)
|
||||||
|
12104520100315
|
Darunavir Ethanolate Tab 150 MG (Base Equiv)
|
||||||
|
12104520100340
|
Darunavir Ethanolate Tab 600 MG (Base Equiv)
|
||||||
|
12104520100350
|
Darunavir Ethanolate Tab 800 MG (Base Equiv)
|
||||||
|
12104520101820
|
Darunavir Ethanolate Susp 100 MG/ML (Base Equiv)
|
||||||
|
12104525100330
|
Fosamprenavir Calcium Tab 700 MG (Base Equiv)
|
||||||
|
12104525101820
|
Fosamprenavir Calcium Susp 50 MG/ML (Base Equiv)
|
||||||
|
12104530200120
|
Indinavir Sulfate Cap 200 MG
|
||||||
|
12104530200140
|
Indinavir Sulfate Cap 400 MG
|
||||||
|
12104545200320
|
Nelfinavir Mesylate Tab 250 MG
|
||||||
|
12104545200340
|
Nelfinavir Mesylate Tab 625 MG
|
||||||
|
12104560000120
|
Ritonavir Cap 100 MG
|
||||||
|
12104560000320
|
Ritonavir Tab 100 MG
|
||||||
|
12104560002020
|
Ritonavir Oral Soln 80 MG/ML
|
||||||
|
12104580200120
|
Saquinavir Mesylate Cap 200 MG
|
||||||
|
12104580200320
|
Saquinavir Mesylate Tab 500 MG
|
||||||
|
12104585000120
|
Tipranavir Cap 250 MG
|
||||||
|
12104585002020
|
Tipranavir Oral Soln 100 MG/ML
|
51
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
12109902550310
|
Lopinavir-Ritonavir Tab 100-25 MG
|
||||||
|
12109902550320
|
Lopinavir-Ritonavir Tab 200-50 MG
|
||||||
|
12109902552020
|
Lopinavir-Ritonavir Soln 400-100 MG/5ML (80-20 MG/ML)
|
||||||
|
12102060000320
|
Maraviroc Tab 150 MG
|
PA
|
|||||
|
12102060000330
|
Maraviroc Tab 300 MG
|
PA
|
|||||
|
12102530002120
|
Enfuvirtide For Inj 90 MG
|
PA
|
|||||
|
12103015100320
|
Dolutegravir Sodium Tab 50 MG (Base Equiv)
|
||||||
|
12103060100320
|
Raltegravir Potassium Tab 400 MG (Base Equiv)
|
||||||
|
12103060100510
|
Raltegravir Potassium Chew Tab 25 MG (Base Equiv)
|
||||||
|
12103060100540
|
Raltegravir Potassium Chew Tab 100 MG (Base Equiv)
|
||||||
|
12103060103020
|
Raltegravir Potassium Packet For Susp 100 MG (Base Equiv)
|
||||||
|
12105005100320
|
Abacavir Sulfate Tab 300 MG (Base Equiv)
|
||||||
|
12105005102020
|
Abacavir Sulfate Soln 20 MG/ML (Base Equiv)
|
||||||
|
12105015002120
|
Didanosine For Oral Soln 2 GM
|
||||||
|
12105015002140
|
Didanosine For Oral Soln 4 GM
|
||||||
|
12106030000120
|
Emtricitabine Caps 200 MG
|
||||||
|
12106030002010
|
Emtricitabine Soln 10 MG/ML
|
||||||
|
12106060000315
|
Lamivudine Tab 100 MG
|
PA
|
|||||
|
12106060000320
|
Lamivudine Tab 150 MG
|
PA
|
|||||
|
12106060000330
|
Lamivudine Tab 300 MG
|
PA
|
|||||
|
12106060002010
|
Lamivudine Oral Soln 5 MG/ML
|
PA
|
|||||
|
12106060002020
|
Lamivudine Oral Soln 10 MG/ML
|
PA
|
|||||
|
12108070000115
|
Stavudine Cap 15 MG
|
||||||
|
12108070000120
|
Stavudine Cap 20 MG
|
||||||
|
12108070000130
|
Stavudine Cap 30 MG
|
||||||
|
12108070000140
|
Stavudine Cap 40 MG
|
||||||
|
12108070002120
|
Stavudine For Oral Soln 1 MG/ML
|
||||||
|
12108085000110
|
Zidovudine Cap 100 MG
|
||||||
|
12108085000330
|
Zidovudine Tab 300 MG
|
52
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
12108085001210
|
Zidovudine Syrup 10 MG/ML
|
||||||
|
12108085002020
|
Zidovudine IV Soln 10 MG/ML
|
||||||
|
12108570100305
|
Tenofovir Disoproxil Fumarate Tab 150 MG
|
||||||
|
12108570100310
|
Tenofovir Disoproxil Fumarate Tab 200 MG
|
||||||
|
12108570100315
|
Tenofovir Disoproxil Fumarate Tab 250 MG
|
||||||
|
12108570100320
|
Tenofovir Disoproxil Fumarate Tab 300 MG
|
||||||
|
12108570102920
|
Tenofovir Disoproxil Fumarate Oral Powder 40 MG/GM
|
||||||
|
12109020200320
|
Delavirdine Mesylate Tab 100 MG
|
||||||
|
12109020200330
|
Delavirdine Mesylate Tab 200 MG
|
||||||
|
12109030000110
|
Efavirenz Cap 50 MG
|
||||||
|
12109030000140
|
Efavirenz Cap 200 MG
|
||||||
|
12109030000330
|
Efavirenz Tab 600 MG
|
||||||
|
12109035000310
|
Etravirine Tab 25 MG
|
PA
|
|||||
|
12109035000320
|
Etravirine Tab 100 MG
|
PA
|
|||||
|
12109035000340
|
Etravirine Tab 200 MG
|
PA
|
|||||
|
12109050000320
|
Nevirapine Tab 200 MG
|
||||||
|
12109050001820
|
Nevirapine Susp 50 MG/5ML
|
||||||
|
12109050007510
|
Nevirapine Tab SR 24HR 100 MG
|
||||||
|
12109050007520
|
Nevirapine Tab SR 24HR 400 MG
|
||||||
|
12109080100320
|
Rilpivirine HCl Tab 25 MG (Base Equivalent)
|
||||||
|
12109902200340
|
Abacavir Sulfate-Lamivudine Tab 600-300 MG
|
||||||
|
12109902300320
|
Emtricitabine-Tenofovir Disoproxil Fumarate Tab 200- 300 MG
|
||||||
|
12109902500320
|
Lamivudine-Zidovudine Tab 150-300 MG
|
||||||
|
12109903200320
|
Abacavir Sulfate-Lamivudine-Zidovudine Tab 300-150- 300 MG
|
||||||
|
12109903300320
|
Efavirenz-Emtricitabine-Tenofovir DF Tab 600-200-300 MG
|
||||||
|
12109903400320
|
Emtricitabine-Rilpivirine-Tenofovir DF Tab 200-25-300 MG
|
53
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
12109904300320
|
Elvitegrav-Cobicis-Emtricitab-Tenofov Tab 150-150-200- 300 MG
|
||||||
|
12200066100320
|
Valganciclovir HCl Tab 450 MG
|
||||||
|
12200066102120
|
Valganciclovir HCl For Oral Soln 50 MG/ML (Base Equiv)
|
||||||
|
12352030000320
|
Entecavir Tab 0.5 MG
|
PA
|
|||||
|
12352030000330
|
Entecavir Tab 1 MG
|
PA
|
|||||
|
12352030002020
|
Entecavir Oral Soln 0.05 MG/ML
|
PA
|
|||||
|
12405010000110
|
Acyclovir Cap 200 MG
|
||||||
|
12405010000320
|
Acyclovir Tab 400 MG
|
||||||
|
12405010000330
|
Acyclovir Tab 800 MG
|
||||||
|
12405010000365
|
Acyclovir Buccal Tab 50 MG
|
||||||
|
12405010001810
|
Acyclovir Susp 200 MG/5ML
|
||||||
|
12500070100320
|
Rimantadine Hydrochloride Tab 100 MG
|
||||||
|
12504060200110
|
Oseltamivir Phosphate Cap 30 MG (Base Equiv)
|
PA
|
|||||
|
12504060200115
|
Oseltamivir Phosphate Cap 45 MG (Base Equiv)
|
PA
|
|||||
|
12504060200120
|
Oseltamivir Phosphate Cap 75 MG (Base Equiv)
|
PA
|
|||||
|
12504060201910
|
Oseltamivir Phosphate For Susp 6 MG/ML (Base Equiv)
|
PA
|
|||||
|
ASSORTED CLASSES
|
99200030000110
|
Penicillamine Cap 250 MG
|
PA
|
||||
|
99200030000305
|
Penicillamine Tab 250 MG
|
PA
|
|||||
|
99402020000110
|
Cyclosporine Cap 25 MG
|
PA
|
|||||
|
99402020000140
|
Cyclosporine Cap 100 MG
|
PA
|
|||||
|
99402020002010
|
Cyclosporine Oral Soln 100 MG/ML
|
PA
|
|||||
|
99402020300120
|
Cyclosporine Modified Cap 25 MG
|
PA
|
|||||
|
99402020300130
|
Cyclosporine Modified Cap 50 MG
|
PA
|
|||||
|
99402020300150
|
Cyclosporine Modified Cap 100 MG
|
PA
|
|||||
|
99402020302020
|
Cyclosporine Modified Oral Soln 100 MG/ML
|
PA
|
|||||
|
99403030100120
|
Mycophenolate Mofetil Cap 250 MG
|
PA
|
|||||
|
99403030100330
|
Mycophenolate Mofetil Tab 500 MG
|
PA
|
|||||
|
99403030101920
|
Mycophenolate Mofetil For Oral Susp 200 MG/ML
|
PA
|
54
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
99403030300620
|
Mycophenolate Sodium Tab DR 180 MG (Mycophenolic Acid Equiv)
|
PA
|
|||||
|
99403030300630
|
Mycophenolate Sodium Tab DR 360 MG (Mycophenolic Acid Equiv)
|
PA
|
|||||
|
99404035000320
|
Everolimus Tab 0.25 MG
|
PA
|
|||||
|
99404035000325
|
Everolimus Tab 0.5 MG
|
PA
|
|||||
|
99404035000330
|
Everolimus Tab 0.75 MG
|
PA
|
|||||
|
99404070000310
|
Sirolimus Tab 0.5 MG
|
PA
|
|||||
|
99404070000320
|
Sirolimus Tab 1 MG
|
PA
|
|||||
|
99404070000330
|
Sirolimus Tab 2 MG
|
PA
|
|||||
|
99404070002020
|
Sirolimus Oral Soln 1 MG/ML
|
PA
|
|||||
|
99404080000105
|
Tacrolimus Cap 0.5 MG
|
PA
|
|||||
|
99404080000110
|
Tacrolimus Cap 1 MG
|
PA
|
|||||
|
99404080000120
|
Tacrolimus Cap 5 MG
|
PA
|
|||||
|
99406010000305
|
Azathioprine Tab 50 MG
|
PA
|
|||||
|
99406010000315
|
Azathioprine Tab 75 MG
|
PA
|
|||||
|
99406010000325
|
Azathioprine Tab 100 MG
|
PA
|
|||||
|
99450010001840
|
Sodium Polystyrene Sulfonate Oral Susp 15 GM/60ML
|
||||||
|
99450010001870
|
Sodium Polystyrene Sulfonate Rectal Susp 30 GM/120ML
|
||||||
|
99450010002900
|
*Sodium Polystyrene Sulfonate Powder**
|
||||||
|
BETA BLOCKERS
|
33100030000305
|
Pindolol Tab 5 MG
|
|||||
|
33100030000310
|
Pindolol Tab 10 MG
|
||||||
|
33100040100305
|
Propranolol HCl Tab 10 MG
|
||||||
|
33100040100310
|
Propranolol HCl Tab 20 MG
|
||||||
|
33100040100315
|
Propranolol HCl Tab 40 MG
|
||||||
|
33100040100320
|
Propranolol HCl Tab 60 MG
|
||||||
|
33100040100325
|
Propranolol HCl Tab 80 MG
|
||||||
|
33100040102050
|
Propranolol HCl Oral Soln 20 MG/5ML
|
||||||
|
33100040102060
|
Propranolol HCl Oral Soln 40 MG/5ML
|
55
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
33100045100310
|
Sotalol HCl Tab 80 MG
|
||||||
|
33100045100315
|
Sotalol HCl Tab 120 MG
|
||||||
|
33100045100320
|
Sotalol HCl Tab 160 MG
|
||||||
|
33100045100330
|
Sotalol HCl Tab 240 MG
|
||||||
|
33200020000305
|
Atenolol Tab 50 MG
|
||||||
|
33200020000310
|
Atenolol Tab 100 MG
|
||||||
|
33200030100305
|
Metoprolol Tartrate Tab 25 MG
|
||||||
|
33200030100310
|
Metoprolol Tartrate Tab 50 MG
|
||||||
|
33200030100315
|
Metoprolol Tartrate Tab 100 MG
|
||||||
|
33300007000305
|
Carvedilol Tab 3.125 MG
|
||||||
|
33300007000310
|
Carvedilol Tab 6.25 MG
|
||||||
|
33300007000320
|
Carvedilol Tab 12.5 MG
|
||||||
|
33300007000330
|
Carvedilol Tab 25 MG
|
||||||
|
33300010100305
|
Labetalol HCl Tab 100 MG
|
||||||
|
33300010100310
|
Labetalol HCl Tab 200 MG
|
||||||
|
33300010100315
|
Labetalol HCl Tab 300 MG
|
||||||
|
CALCIUM CHANNEL BLOCKERS
|
34000003100320
|
Amlodipine Besylate Tab 2.5 MG
|
|||||
|
34000003100330
|
Amlodipine Besylate Tab 5 MG
|
||||||
|
34000003100340
|
Amlodipine Besylate Tab 10 MG
|
||||||
|
34000010100305
|
Diltiazem HCl Tab 30 MG
|
||||||
|
34000010100310
|
Diltiazem HCl Tab 60 MG
|
||||||
|
34000010100315
|
Diltiazem HCl Tab 90 MG
|
||||||
|
34000010100320
|
Diltiazem HCl Tab 120 MG
|
||||||
|
34000010106910
|
Diltiazem HCl Cap SR 12HR 60 MG
|
||||||
|
34000010106915
|
Diltiazem HCl Cap SR 12HR 90 MG
|
||||||
|
34000010106920
|
Diltiazem HCl Cap SR 12HR 120 MG
|
||||||
|
34000010107020
|
Diltiazem HCl Cap SR 24HR 120 MG
|
||||||
|
34000010107030
|
Diltiazem HCl Cap SR 24HR 180 MG
|
||||||
|
34000010107040
|
Diltiazem HCl Cap SR 24HR 240 MG
|
||||||
|
34000010117020
|
Diltiazem HCl Extended Release Beads Cap SR 24HR 120 MG
|
56
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost Containment
|
|||
|
34000010117030
|
Diltiazem HCl Extended Release Beads Cap SR 24HR 180 MG
|
||||||
|
34000010117040
|
Diltiazem HCl Extended Release Beads Cap SR 24HR 240 MG
|
||||||
|
34000010117050
|
Diltiazem HCl Extended Release Beads Cap SR 24HR 300 MG
|
||||||
|
34000010117060
|
Diltiazem HCl Extended Release Beads Cap SR 24HR 360 MG
|
||||||
|
34000010117070
|
Diltiazem HCl Extended Release Beads Cap SR 24HR 420 MG
|
||||||
|
34000010127020
|
Diltiazem HCl Coated Beads Cap SR 24HR 120 MG
|
||||||
|
34000010127030
|
Diltiazem HCl Coated Beads Cap SR 24HR 180 MG
|
||||||
|
34000010127040
|
Diltiazem HCl Coated Beads Cap SR 24HR 240 MG
|
||||||
|
34000010127050
|
Diltiazem HCl Coated Beads Cap SR 24HR 300 MG
|
||||||
|
34000010127060
|
Diltiazem HCl Coated Beads Cap SR 24HR 360 MG
|
||||||
|
34000010127520
|
Diltiazem HCl Coated Beads Tab SR 24HR 120 MG
|
||||||
|
34000010127530
|
Diltiazem HCl Coated Beads Tab SR 24HR 180 MG
|
||||||
|
34000010127540
|
Diltiazem HCl Coated Beads Tab SR 24HR 240 MG
|
||||||
|
34000010127550
|
Diltiazem HCl Coated Beads Tab SR 24HR 300 MG
|
||||||
|
34000010127560
|
Diltiazem HCl Coated Beads Tab SR 24HR 360 MG
|
||||||
|
34000010127570
|
Diltiazem HCl Coated Beads Tab SR 24HR 420 MG
|
||||||
|
34000020007530
|
Nifedipine Tab SR 24HR 30 MG
|
||||||
|
34000020007540
|
Nifedipine Tab SR 24HR 60 MG
|
||||||
|
34000020007550
|
Nifedipine Tab SR 24HR 90 MG
|
||||||
|
34000030100303
|
Verapamil HCl Tab 40 MG
|
||||||
|
34000030100305
|
Verapamil HCl Tab 80 MG
|
||||||
|
34000030100310
|
Verapamil HCl Tab 120 MG
|
||||||
|
34000030100410
|
Verapamil HCl Tab CR 120 MG
|
||||||
|
34000030100415
|
Verapamil HCl Tab CR 180 MG
|
||||||
|
34000030100420
|
Verapamil HCl Tab CR 240 MG
|
||||||
|
34000030107015
|
Verapamil HCl Cap SR 24HR 100 MG
|
57
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
34000030107020
|
Verapamil HCl Cap SR 24HR 120 MG
|
||||||
|
34000030107025
|
Verapamil HCl Cap SR 24HR 180 MG
|
||||||
|
34000030107030
|
Verapamil HCl Cap SR 24HR 200 MG
|
||||||
|
34000030107035
|
Verapamil HCl Cap SR 24HR 240 MG
|
||||||
|
34000030107040
|
Verapamil HCl Cap SR 24HR 300 MG
|
||||||
|
34000030107045
|
Verapamil HCl Cap SR 24HR 360 MG
|
||||||
|
CARDIOTONICS
|
31200010000303
|
Digoxin Tab 62.5 MCG (0.0625 MG)
|
|||||
|
31200010000305
|
Digoxin Tab 125 MCG (0.125 MG)
|
||||||
|
31200010000307
|
Digoxin Tab 187.5 MCG (0.1875 MG)
|
||||||
|
31200010000310
|
Digoxin Tab 250 MCG (0.25 MG)
|
||||||
|
CARDIOVASCULAR AGENTS - MISC.
|
40143060100320
|
Sildenafil Citrate Tab 20 MG
|
PA
|
||||
|
40160015000320
|
Bosentan Tab 62.5 MG
|
PA
|
|||||
|
40160015000330
|
Bosentan Tab 125 MG
|
PA
|
|||||
|
40160050000320
|
Macitentan tab 10 mg
|
PA
|
|||||
|
40134050000310
|
Riociguat tab 0.5 mg
|
PA
|
|||||
|
40134050000320
|
Riociguat tab 1 mg
|
PA
|
|||||
|
40134050000330
|
Riociguat tab 1.5 mg
|
PA
|
|||||
|
40134050000340
|
Riociguat tab 2 mg
|
PA
|
|||||
|
40134050000350
|
Riociguat tab 2.5 mg
|
PA
|
|||||
|
CEPHALOSPORINS
|
02100010001910
|
Cefadroxil For Susp 250 MG/5ML
|
AL < 12 Y/O
|
||||
|
02100010001915
|
Cefadroxil For Susp 500 MG/5ML
|
AL < 12 Y/O
|
|||||
|
02100020000105
|
Cephalexin Cap 250 MG
|
||||||
|
02100020000110
|
Cephalexin Cap 500 MG
|
||||||
|
02100020000120
|
Cephalexin Cap 750 MG
|
||||||
|
02100020000310
|
Cephalexin Tab 250 MG
|
||||||
|
02100020000315
|
Cephalexin Tab 500 MG
|
||||||
|
02100020001910
|
Cephalexin For Susp 125 MG/5ML
|
||||||
|
02100020001915
|
Cephalexin For Susp 250 MG/5ML
|
||||||
|
02200040000105
|
Cefaclor Cap 250 MG
|
||||||
|
02200040000110
|
Cefaclor Cap 500 MG
|
||||||
|
02200040001905
|
Cefaclor For Susp 125 MG/5ML
|
58
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
02200040001910
|
Cefaclor For Susp 250 MG/5ML
|
||||||
|
02200040001915
|
Cefaclor For Susp 375 MG/5ML
|
||||||
|
02200062000320
|
Cefprozil Tab 250 MG
|
||||||
|
02200062000330
|
Cefprozil Tab 500 MG
|
||||||
|
02200062001910
|
Cefprozil For Susp 125 MG/5ML
|
||||||
|
02200062001920
|
Cefprozil For Susp 250 MG/5ML
|
||||||
|
02300040000120
|
Cefdinir Cap 300 MG
|
||||||
|
02300040001920
|
Cefdinir For Susp 125 MG/5ML
|
||||||
|
02300040001930
|
Cefdinir For Susp 250 MG/5ML
|
||||||
|
CONTRACEPTIVES
|
CONTRACEPTIVES, SYSTEMIC (FOR THERAPEUTIC USE ONLY)
|
25990002100320
|
Desogestrel & Ethinyl Estradiol Tab 0.15 MG-30 MCG
|
PA
|
|||
|
25990002500320
|
Norethindrone & Ethinyl Estradiol Tab 1 MG-35 MCG
|
PA
|
|||||
|
25990002700310
|
Norethindrone & Mestranol Tab 1 MG-50 MCG
|
PA
|
|||||
|
25990002900310
|
Norgestrel & Ethinyl Estradiol Tab 0.3 MG-30 MCG
|
PA
|
|||||
|
25992002200310
|
Norethindrone-Eth Estradiol Tab 0.5-35/0.75-35/1-35 MG-MCG
|
PA
|
|||||
|
25150035101820
|
Medroxyprogesterone Acetate IM Susp 150 MG/ML
|
PA
|
|||||
|
CORTICOSTEROIDS
|
22100012007020
|
Budesonide Cap SR 24HR 3 MG
|
PA
|
||||
|
22100015100310
|
Cortisone Acetate Tab 25 MG
|
||||||
|
22100020000315
|
Dexamethasone Tab 0.5 MG
|
||||||
|
22100020000320
|
Dexamethasone Tab 0.75 MG
|
||||||
|
22100020000325
|
Dexamethasone Tab 1 MG
|
||||||
|
22100020000330
|
Dexamethasone Tab 1.5 MG
|
||||||
|
22100020000335
|
Dexamethasone Tab 2 MG
|
||||||
|
22100020000340
|
Dexamethasone Tab 4 MG
|
||||||
|
22100020000345
|
Dexamethasone Tab 6 MG
|
||||||
|
22100020002005
|
Dexamethasone Soln 0.5 MG/5ML
|
||||||
|
22100020202005
|
Dexamethasone Sodium Phosphate Inj 4 MG/ML
|
OB-GYN
|
|||||
|
22100025000303
|
Hydrocortisone Tab 5 MG
|
59
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
22100025000305
|
Hydrocortisone Tab 10 MG
|
||||||
|
22100025000310
|
Hydrocortisone Tab 20 MG
|
||||||
|
22100030000305
|
Methylprednisolone Tab 2 MG
|
||||||
|
22100030000310
|
Methylprednisolone Tab 4 MG
|
||||||
|
22100030000315
|
Methylprednisolone Tab 8 MG
|
||||||
|
22100030000320
|
Methylprednisolone Tab 16 MG
|
||||||
|
22100030000330
|
Methylprednisolone Tab 32 MG
|
||||||
|
22100030006405
|
Methylprednisolone Tab 4 MG Dose Pack
|
||||||
|
22100030101805
|
Methylprednisolone Acetate Inj Susp 20 MG/ML
|
||||||
|
22100030101810
|
Methylprednisolone Acetate Inj Susp 40 MG/ML
|
||||||
|
22100030101815
|
Methylprednisolone Acetate Inj Susp 80 MG/ML
|
||||||
|
22100040000305
|
Prednisolone Tab 5 MG
|
||||||
|
22100040001205
|
Prednisolone Syrup 15 MG/5ML (USP Solution Equivalent)
|
||||||
|
22100045000305
|
Prednisone Tab 1 MG
|
||||||
|
22100045000310
|
Prednisone Tab 2.5 MG
|
||||||
|
22100045000315
|
Prednisone Tab 5 MG
|
||||||
|
22100045000320
|
Prednisone Tab 10 MG
|
||||||
|
22100045000325
|
Prednisone Tab 20 MG
|
||||||
|
22100045000335
|
Prednisone Tab 50 MG
|
||||||
|
22100050101805
|
Triamcinolone Acetonide Inj Susp 10 MG/ML
|
||||||
|
22100050101810
|
Triamcinolone Acetonide Inj Susp 40 MG/ML
|
||||||
|
22100050301805
|
Triamcinolone Hexacetonide Inj Susp 5 MG/ML
|
||||||
|
22100050301810
|
Triamcinolone Hexacetonide Inj Susp 20 MG/ML
|
||||||
|
22109902101810
|
Betamethasone Sod Phosphate & Acetate Inj Susp 6 (3- 3) MG/ML
|
OB-GYN
|
|||||
|
22200030100305
|
Fludrocortisone Acetate Tab 0.1 MG
|
||||||
|
COUGH/COLD/ALLERGY
|
43993002597430
|
LORATADINE & PSEUDOEPHEDRINE TAB SR 12HR 5-120 MG
|
|||||
|
43993002597520
|
LORATADINE & PSEUDOEPHEDRINE TAB SR 24HR 10-240 MG
|
60
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
COUGH/COLD/ALLERGY*
|
43997002281210
|
CODEINE-GUAIFENESIN SYRUP 10-100 MG/5ML
|
|||||
|
DERMATOLOGICALS
|
AT LEAST ONE OF THE FOLLOWING
|
90784060003720
|
Pimecrolimus Cream 1%
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
90784075004210
|
Tacrolimus Oint 0.03%
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
90784075004230
|
Tacrolimus Oint 0.1%
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550020003705
|
Betamethasone Dipropionate Cream 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550020004105
|
Betamethasone Dipropionate Lotion 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550020004205
|
Betamethasone Dipropionate Oint 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550060002005
|
Fluocinonide Soln 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550060003705
|
Fluocinonide Cream 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550060004005
|
Fluocinonide Gel 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550060004205
|
Fluocinonide Oint 0.05%
|
61
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550085103720
|
Triamcinolone Acetonide Cream 0.5%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGH POTENCY AGENTS
|
90550085104215
|
Triamcinolone Acetonide Oint 0.5%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGHEST POTENCY AGENTS
|
90550020054205
|
Betamethasone Dipropionate Augmented Oint 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGHEST POTENCY AGENTS
|
90550025102005
|
Clobetasol Propionate Soln 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGHEST POTENCY AGENTS
|
90550025103705
|
Clobetasol Propionate Cream 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGHEST POTENCY AGENTS
|
90550025104010
|
Clobetasol Propionate Gel 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING HIGHEST POTENCY AGENTS
|
90550025104205
|
Clobetasol Propionate Oint 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550020103710
|
Betamethasone Valerate Cream 0.1%
|
|||||
|
AT LEAST ONE OF THE
FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550020104105
|
Betamethasone Valerate Lotion 0.1%
|
62
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550020104205
|
Betamethasone Valerate Oint 0.1%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550040004005
|
Desoximetasone Gel 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550055103710
|
Fluocinolone Acetonide Cream 0.025%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550055104205
|
Fluocinolone Acetonide Oint 0.025%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550082104210
|
Mometasone Furoate Oint 0.1%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550085103710
|
Triamcinolone Acetonide Cream 0.1%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550085104110
|
Triamcinolone Acetonide Lotion 0.1%
|
63
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING INTERMEDIATE POTENCY AGENTS
|
90550085104210
|
Triamcinolone Acetonide Oint 0.1%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550035003705
|
Desonide Cream 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550035004105
|
Desonide Lotion 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550035004205
|
Desonide Oint 0.05%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550055102005
|
Fluocinolone Acetonide Soln 0.01%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550055103705
|
Fluocinolone Acetonide Cream 0.01%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550075003725
|
Hydrocortisone Cream 2.5%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550075004118
|
Hydrocortisone Lotion 2%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550075004120
|
Hydrocortisone Lotion 2.5%
|
64
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING LOW POTENCY AGENTS
|
90550075004215
|
Hydrocortisone Oint 2.5%
|
|||||
|
ONLY LEGEND DOSAGE FORMS ARE COVERED
|
90154020002005
|
Clotrimazole Soln 1%
|
|||||
|
90154020003705
|
Clotrimazole Cream 1%
|
||||||
|
90050013000110
|
Isotretinoin Cap 10 MG
|
||||||
|
90050013000120
|
Isotretinoin Cap 20 MG
|
||||||
|
90050013000130
|
Isotretinoin Cap 30 MG
|
||||||
|
90050013000140
|
Isotretinoin Cap 40 MG
|
||||||
|
90050030003703
|
Tretinoin Cream 0.025%
|
AL 21 < Y/O
|
|||||
|
90050030003705
|
Tretinoin Cream 0.05%
|
AL 21 < Y/O
|
|||||
|
90050030003710
|
Tretinoin Cream 0.1%
|
AL 21 < Y/O
|
|||||
|
90050030004005
|
Tretinoin Gel 0.01%
|
AL 21 < Y/O
|
|||||
|
90050030004010
|
Tretinoin Gel 0.025%
|
AL 21 < Y/O
|
|||||
|
90051010102005
|
Clindamycin Phosphate Soln 1%
|
||||||
|
90051020002010
|
Erythromycin Soln 2%
|
||||||
|
90051020004010
|
Erythromycin Gel 2%
|
||||||
|
90059903201620
|
Sulfacetamide Sodium w/ Sulfur Emulsion 10-5%
|
||||||
|
90059903201820
|
Sulfacetamide Sodium w/ Sulfur Susp 10-5%
|
||||||
|
90059903203720
|
Sulfacetamide Sodium w/ Sulfur Cream 10-5%
|
||||||
|
90059903204110
|
Sulfacetamide Sodium w/ Sulfur Lotion 10-5%
|
||||||
|
90060010004020
|
Azelaic Acid Gel 15%
|
AL > 21 Y/O
|
|||||
|
90060040003710
|
Metronidazole Cream 0.75%
|
||||||
|
90060040004010
|
Metronidazole Gel 0.75%
|
||||||
|
90060040004110
|
Metronidazole Lotion 0.75%
|
||||||
|
90100050103705
|
Gentamicin Sulfate Cream 0.1%
|
||||||
|
90100065104210
|
Mupirocin Oint 2%
|
||||||
|
90150080004215
|
Nystatin Oint 100000 Unit/GM
|
||||||
|
90154045003710
|
Ketoconazole Cream 2%
|
65
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
90154045004510
|
Ketoconazole Shampoo 2%
|
||||||
|
90154050102910
|
MICONAZOLE NITRATE POWDER 2%
|
OTC
|
|||||
|
90250020003725
|
Anthralin Cream 1%
|
||||||
|
90250025003710
|
Calcipotriene Cream 0.005%
|
||||||
|
90250025004210
|
Calcipotriene Oint 0.005%
|
||||||
|
90250070003720
|
Tazarotene Cream 0.05%
|
||||||
|
90250070003730
|
Tazarotene Cream 0.1%
|
||||||
|
90250070004020
|
Tazarotene Gel 0.05%
|
||||||
|
90250070004030
|
Tazarotene Gel 0.1%
|
||||||
|
90250510000110
|
Acitretin Cap 10 MG
|
||||||
|
90250510000115
|
Acitretin Cap 17.5 MG
|
||||||
|
90250510000125
|
Acitretin Cap 25 MG
|
||||||
|
90250560000110
|
Methoxsalen Cap 10 MG
|
||||||
|
90300050004120
|
Selenium Sulfide Lotion 2.5%
|
||||||
|
90372030002020
|
Fluorouracil Soln 2%
|
||||||
|
90372030002050
|
Fluorouracil Soln 5%
|
||||||
|
90450030003710
|
Silver Sulfadiazine Cream 1%
|
||||||
|
90559802404220
|
Pramoxine-HC Oint 1-1%
|
||||||
|
90559802404230
|
Pramoxine-HC Oint 1-2.5%
|
||||||
|
90736030000310
|
Finasteride Tab 1 MG
|
||||||
|
90773040003720
|
Imiquimod Cream 5%
|
||||||
|
90900017004120
|
Ivermectin Lotion 0.5%
|
||||||
|
90900020004110
|
Lindane Lotion 1%
|
STEP THERAPY;
LC = 60ML
|
|||||
|
90900020004510
|
Lindane Shampoo 1%
|
STEP THERAPY;
LC = 60ML
|
|||||
|
90900035003720
|
Permethrin Cream 5%
|
LC = 60MG
|
|||||
|
DIGESTIVE AIDS
|
51200024000330
|
Pancrelipase (Lip-Prot-Amyl) Tab 10440-39150-39150 Unit
|
|||||
|
51200024000360
|
Pancrelipase (Lip-Prot-Amyl) Tab 20880-78300-78300 Unit
|
66
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
51200024006705
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 3000-9500-15000 Unit
|
||||||
|
51200024006706
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 3000-10000-16000 Unit
|
||||||
|
51200024006710
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 4200-10000-17500 Unit
|
||||||
|
51200024006715
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 5000-17000-27000 Unit
|
||||||
|
51200024006720
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 6000-19000-30000 Unit
|
||||||
|
51200024006725
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 8000-28750-30250 Unit
|
||||||
|
51200024006730
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 10000-34000- 55000 Unit
|
||||||
|
51200024006734
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 10500-25000- 43750 Unit
|
||||||
|
51200024006740
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 12000-38000- 60000 Unit
|
||||||
|
51200024006744
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 13800-27600- 27600 Unit
|
||||||
|
51200024006748
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 15000-51000- 82000 Unit
|
||||||
|
51200024006749
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 16000-57500- 60500 Unit
|
||||||
|
51200024006750
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 16800-40000- 70000 Unit
|
||||||
|
51200024006752
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 20000-68000- 109000 Unit
|
||||||
|
51200024006753
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 20700-41400- 41400 Unit
|
67
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
51200024006754
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 21000-37000- 61000 Unit
|
||||||
|
51200024006758
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 23000-46000- 46000 Unit
|
||||||
|
51200024006760
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 24000-76000- 120000 Unit
|
||||||
|
51200024006765
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 25000-85000- 136000 Unit
|
||||||
|
51200024006780
|
Pancrelipase (Lip-Prot-Amyl) DR Cap 36000-114000- 180000 Unit
|
||||||
|
DIURETICS
|
37100010000305
|
Acetazolamide Tab 125 MG
|
|||||
|
37100010000310
|
Acetazolamide Tab 250 MG
|
||||||
|
37200010000305
|
Bumetanide Tab 0.5 MG
|
||||||
|
37200010000310
|
Bumetanide Tab 1 MG
|
||||||
|
37200010000315
|
Bumetanide Tab 2 MG
|
||||||
|
37200030000305
|
Furosemide Tab 20 MG
|
||||||
|
37200030000310
|
Furosemide Tab 40 MG
|
||||||
|
37200030000315
|
Furosemide Tab 80 MG
|
||||||
|
37200030002045
|
Furosemide Oral Soln 8 MG/ML
|
||||||
|
37200030002050
|
Furosemide Oral Soln 10 MG/ML
|
||||||
|
37500020000305
|
Spironolactone Tab 25 MG
|
||||||
|
37500020000310
|
Spironolactone Tab 50 MG
|
||||||
|
37500020000315
|
Spironolactone Tab 100 MG
|
||||||
|
37600020000305
|
Chlorothiazide Tab 250 MG
|
||||||
|
37600020000310
|
Chlorothiazide Tab 500 MG
|
||||||
|
37600020001805
|
Chlorothiazide Susp 250 MG/5ML
|
||||||
|
37600025000305
|
Chlorthalidone Tab 25 MG
|
||||||
|
37600025000310
|
Chlorthalidone Tab 50 MG
|
||||||
|
37600025000315
|
Chlorthalidone Tab 100 MG
|
||||||
|
37600040000110
|
Hydrochlorothiazide Cap 12.5 MG
|
||||||
|
37600040000303
|
Hydrochlorothiazide Tab 12.5 MG
|
68
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost Containment
|
|||
|
37600040000305
|
Hydrochlorothiazide Tab 25 MG
|
||||||
|
37600040000310
|
Hydrochlorothiazide Tab 50 MG
|
||||||
|
37600050000303
|
Indapamide Tab 1.25 MG
|
||||||
|
37600050000305
|
Indapamide Tab 2.5 MG
|
||||||
|
37600060000305
|
Metolazone Tab 2.5 MG
|
||||||
|
37600060000310
|
Metolazone Tab 5 MG
|
||||||
|
37600060000315
|
Metolazone Tab 10 MG
|
||||||
|
37990002300105
|
Triamterene & Hydrochlorothiazide Cap 37.5-25 MG
|
||||||
|
37990002300110
|
Triamterene & Hydrochlorothiazide Cap 50-25 MG
|
||||||
|
37990002300315
|
Triamterene & Hydrochlorothiazide Tab 37.5-25 MG
|
||||||
|
37990002300330
|
Triamterene & Hydrochlorothiazide Tab 75-50 MG
|
||||||
|
ENDOCRINE AND METABOLIC AGENTS - MISC.
|
AT LEAST ONE OF THE FOLLOWING
|
30042010100305
|
Alendronate Sodium Tab 5 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042010100310
|
Alendronate Sodium Tab 10 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042010100335
|
Alendronate Sodium Tab 35 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042010100340
|
Alendronate Sodium Tab 40 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042010100370
|
Alendronate Sodium Tab 70 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042065100305
|
Risedronate Sodium Tab 5 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042065100320
|
Risedronate Sodium Tab 30 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042065100330
|
Risedronate Sodium Tab 35 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
30042065100380
|
Risedronate Sodium Tab 150 MG
|
|||||
|
OMNITROPE
|
30100020002050
|
Somatropin Inj 5 MG/1.5ML
|
PA
|
69
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
30100020002056
|
Somatropin Inj 10 MG/1.5ML
|
PA
|
|||||
|
30100020002123
|
Somatropin For Inj 5.8 MG
|
PA
|
|||||
|
30042040100305
|
Etidronate Disodium Tab 200 MG
|
||||||
|
30042040100310
|
Etidronate Disodium Tab 400 MG
|
||||||
|
30044070002020
|
Teriparatide (Recombinant) Inj 600 MCG/2.4ML
|
PA
|
|||||
|
30080050106420
|
Leuprolide Acetate For Inj Pediatric Kit 7.5 MG
|
PA
|
|||||
|
30080050106430
|
Leuprolide Acetate For Inj Pediatric Kit 11.25 MG
|
PA
|
|||||
|
30080050106440
|
Leuprolide Acetate For Inj Pediatric Kit 15 MG
|
PA
|
|||||
|
30080050156420
|
Leuprolide Acetate (3 Month) For Inj Pediatric Kit 11.25 MG
|
PA
|
|||||
|
30080050156440
|
Leuprolide Acetate (3 Month) For Inj Pediatric Kit 30 MG
|
||||||
|
30089902506420
|
Leuprolide (1 Mon) Inj 3.75 MG & Norethindrone Tab 5 MG Kit
|
PA
|
|||||
|
30089902506440
|
Leuprolide (3 Mon) Inj 11.25 MG & Norethindrone Tab 5 MG Kit
|
PA
|
|||||
|
30170070102005
|
Octreotide Acetate Inj 50 MCG/ML (0.05 MG/ML)
|
PA
|
|||||
|
30170070102010
|
Octreotide Acetate Inj 100 MCG/ML (0.1 MG/ML)
|
PA
|
|||||
|
30170070102015
|
Octreotide Acetate Inj 200 MCG/ML (0.2 MG/ML)
|
PA
|
|||||
|
30170070102020
|
Octreotide Acetate Inj 500 MCG/ML (0.5 MG/ML)
|
PA
|
|||||
|
30170070102030
|
Octreotide Acetate Inj 1000 MCG/ML (1 MG/ML)
|
PA
|
|||||
|
30170070106410
|
Octreotide Acetate For IM Inj Kit 10 MG
|
PA
|
|||||
|
30170070106420
|
Octreotide Acetate For IM Inj Kit 20 MG
|
PA
|
|||||
|
30170070106430
|
Octreotide Acetate For IM Inj Kit 30 MG
|
PA
|
|||||
|
30201010100310
|
Desmopressin Acetate Tab 0.1 MG
|
||||||
|
30201010100320
|
Desmopressin Acetate Tab 0.2 MG
|
||||||
|
30201010102015
|
Desmopressin Acetate Nasal Soln 1.5 MG/ML
|
||||||
|
30201010102030
|
Desmopressin Acetate Inj 4 MCG/ML
|
||||||
|
30402020000320
|
Cabergoline Tab 0.5 MG
|
||||||
|
30903045100330
|
Levocarnitine Tab 330 MG
|
PA
|
|||||
|
30903045102010
|
Levocarnitine Oral Soln 1 GM/10ML (10%)
|
PA
|
70
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost Containment
|
|||
|
30905030000105
|
Calcitriol Cap 0.25 MCG
|
||||||
|
30905030000110
|
Calcitriol Cap 0.5 MCG
|
||||||
|
30905030002050
|
Calcitriol Oral Soln 1 MCG/ML
|
||||||
|
30905225100320
|
Cinacalcet HCl Tab 30 MG (Base Equiv)
|
PA
|
|||||
|
30905225100330
|
Cinacalcet HCl Tab 60 MG (Base Equiv)
|
PA
|
|||||
|
30905225100340
|
Cinacalcet HCl Tab 90 MG (Base Equiv)
|
PA
|
|||||
|
ERYTHROMYCIN-SULFISOXAZOLE
|
16990002101910
|
ERYTHROMYCIN-SULFISOXAZOLE FOR SUSP 200-600 MG/5ML
|
|||||
|
FLUOROQUINOLONES
|
AT LEAST ONE OF THE FOLLOWING
|
05000034000320
|
Levofloxacin Tab 250 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
05000034000330
|
Levofloxacin Tab 500 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
05000034000340
|
Levofloxacin Tab 750 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
05000034002020
|
Levofloxacin IV Soln 25 MG/ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
05000034002050
|
Levofloxacin Oral Soln 25 MG/ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
05000037100320
|
Moxifloxacin HCl Tab 400 MG (Base Equiv)
|
|||||
|
05000020001920
|
Ciprofloxacin For Oral Susp 250 MG/5ML (5%) (5 GM/100ML)
|
||||||
|
05000020001930
|
Ciprofloxacin For Oral Susp 500 MG/5ML (10%) (10 GM/100ML)
|
||||||
|
05000020100305
|
Ciprofloxacin HCl Tab 100 MG (Base Equiv)
|
||||||
|
05000020100310
|
Ciprofloxacin HCl Tab 250 MG (Base Equiv)
|
||||||
|
05000020100315
|
Ciprofloxacin HCl Tab 500 MG (Base Equiv)
|
||||||
|
05000020100320
|
Ciprofloxacin HCl Tab 750 MG (Base Equiv)
|
||||||
|
GASTROINTESTINAL AGENTS - MISC.
|
AT LEAST ONE OF THE FOLLOWING
|
52800045200540
|
Lanthanum Carbonate Chew Tab 500 MG
|
PA
|
71
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
52800045200550
|
Lanthanum Carbonate Chew Tab 750 MG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
52800045200560
|
Lanthanum Carbonate Chew Tab 1000 MG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
52800070050340
|
Sevelamer Carbonate Tab 800 MG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
52800070053020
|
Sevelamer Carbonate Packet 0.8 GM
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
52800070053040
|
Sevelamer Carbonate Packet 2.4 GM
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
52800080100520
|
Sucroferric oxyhydroxide chew tab 500 mg
|
PA
|
||||
|
52100040000120
|
Ursodiol Cap 300 MG
|
||||||
|
52100040000325
|
Ursodiol Tab 250 MG
|
||||||
|
52100040000350
|
Ursodiol Tab 500 MG
|
||||||
|
52160015101320
|
Cromolyn Sodium Oral Conc 100 MG/5ML
|
||||||
|
52300020100303
|
Metoclopramide HCl Tab 5 MG
|
||||||
|
52300020100305
|
Metoclopramide HCl Tab 10 MG
|
||||||
|
52300020102005
|
Metoclopramide HCl Inj 5 MG/ML
|
||||||
|
52300020102013
|
Metoclopramide HCl Soln 5 MG/5ML (10 MG/10ML)
|
||||||
|
52500030000210
|
Mesalamine Cap CR 250 MG
|
||||||
|
52500030000220
|
Mesalamine Cap CR 500 MG
|
||||||
|
52500030000650
|
Mesalamine Tab Delayed Release 800 MG
|
||||||
|
52500030000670
|
Mesalamine Tab Delayed Release 1.2 GM
|
||||||
|
52500030005105
|
Mesalamine Enema 4 GM
|
||||||
|
52500030005110
|
Mesalamine Sulfite-Free (SF) Enema 4 GM/60ML
|
||||||
|
52500030005240
|
Mesalamine Suppos 1000 MG
|
||||||
|
52500040100120
|
Olsalazine Sodium Cap 250 MG
|
||||||
|
52500060000310
|
Sulfasalazine Tab 500 MG
|
||||||
|
52500060000610
|
Sulfasalazine Tab Delayed Release 500 MG
|
72
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost Containment
|
|||
|
52580050102015
|
Methylnaltrexone Bromide Inj 8 MG/0.4ML (20 MG/ML)
|
||||||
|
52580050102020
|
Methylnaltrexone Bromide Inj 12 MG/0.6ML (20 MG/ML)
|
||||||
|
52580050106420
|
Methylnaltrexone Bromide Inj Kit 12 MG/0.6ML
|
||||||
|
52800020100120
|
Calcium Acetate (Phosphate Binder) Cap 667 MG (169 MG Ca)
|
||||||
|
52800020100320
|
Calcium Acetate (Phosphate Binder) Tab 667 MG
|
||||||
|
52800020102020
|
Calcium Acetate (Phosphate Binder) Oral Soln 667 MG/5ML
|
||||||
|
GENITOURINARY AGENTS - MISCELLANEOUS
|
56300010100305
|
Phenazopyridine HCl Tab 100 MG
|
LC
|
||||
|
56300010100310
|
Phenazopyridine HCl Tab 200 MG
|
LC
|
|||||
|
56851030000320
|
Finasteride Tab 5 MG
|
||||||
|
GOUT AGENTS
|
68000010000305
|
Allopurinol Tab 100 MG
|
|||||
|
68000010000310
|
Allopurinol Tab 300 MG
|
||||||
|
68000020000310
|
Colchicine Tab 0.6 MG
|
||||||
|
68100010000310
|
Probenecid Tab 500 MG
|
||||||
|
68990002100310
|
Colchicine w/ Probenecid Tab 0.5-500 MG
|
||||||
|
HEMATOLOGICAL AGENTS - MISC.
|
85100010002109
|
Antihemophilic Factor (Human) For Inj 220-400 Unit
|
PA
|
||||
|
85100010002110
|
Antihemophilic Factor (Human) For Inj 250 Unit
|
PA
|
|||||
|
85100010002125
|
Antihemophilic Factor (Human) For Inj 401-800 Unit
|
PA
|
|||||
|
85100010002130
|
Antihemophilic Factor (Human) For Inj 500 Unit
|
PA
|
|||||
|
85100010002140
|
Antihemophilic Factor (Human) For Inj 1000 Unit
|
PA
|
|||||
|
85100010002146
|
Antihemophilic Factor (Human) For Inj 1700 Unit
|
PA
|
|||||
|
85100010006410
|
Antihemophilic Factor (Human) For Inj Kit 250 Unit
|
PA
|
|||||
|
85100010006430
|
Antihemophilic Factor (Human) For Inj Kit 500 Unit
|
PA
|
|||||
|
85100010202115
|
Antihemophilic Factor (Recombinant) For Inj 220-400 Unit
|
PA
|
|||||
|
85100010202125
|
Antihemophilic Factor (Recombinant) For Inj 401-800 Unit
|
PA
|
73
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
85100010202135
|
Antihemophilic Factor (Recombinant) For Inj 801-1240 Unit
|
PA
|
|||||
|
85100010206420
|
Antihemophilic Factor (Recombinant) For Inj Kit 250 Unit
|
PA
|
|||||
|
85100010206430
|
Antihemophilic Factor (Recombinant) For Inj Kit 500 Unit
|
PA
|
|||||
|
85100010206440
|
Antihemophilic Factor (Recombinant) For Inj Kit 1000 Unit
|
PA
|
|||||
|
85100010206450
|
Antihemophilic Factor (Recombinant) For Inj Kit 2000 Unit
|
PA
|
|||||
|
85100010252120
|
Antihemophilic Factor rAHF-PFM For Inj 250 Unit
|
PA
|
|||||
|
85100010252130
|
Antihemophilic Factor rAHF-PFM For Inj 500 Unit
|
PA
|
|||||
|
85100010252140
|
Antihemophilic Factor rAHF-PFM For Inj 1000 Unit
|
PA
|
|||||
|
85100010252150
|
Antihemophilic Factor rAHF-PFM For Inj 1500 Unit
|
PA
|
|||||
|
85100010252170
|
Antihemophilic Factor rAHF-PFM For Inj 2000 Unit
|
PA
|
|||||
|
85100010252180
|
Antihemophilic Factor rAHF-PFM For Inj 3000 Unit
|
PA
|
|||||
|
85100010266420
|
Antihemophilic Factor Recombinant PAF For Inj Kit 250 Unit
|
PA
|
|||||
|
85100010266430
|
Antihemophilic Factor Recombinant PAF For Inj Kit 500 Unit
|
PA
|
|||||
|
85100010266440
|
Antihemophilic Factor Recombinant PAF For Inj Kit 1000 Unit
|
PA
|
|||||
|
85100010266460
|
Antihemophilic Factor Recombinant PAF For Inj Kit 2000 Unit
|
PA
|
|||||
|
85100010266470
|
Antihemophilic Factor Recombinant PAF For Inj Kit 3000 Unit
|
PA
|
|||||
|
85100020002100
|
*Antiinhibitor Coagulant Complex For Inj**
|
PA
|
|||||
|
85100026202130
|
COAGULATION FACTOR VIIA (RECOMB) FOR INJ 2.4 MG (2400 MCG)
|
PA
|
|||||
|
85100028002160
|
Coagulation Factor IX For Inj 250 Unit
|
PA
|
|||||
|
85100028002170
|
Coagulation Factor IX For Inj 500 Unit
|
PA
|
74
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
85100028002180
|
Coagulation Factor IX For Inj 1000 Unit
|
PA
|
|||||
|
85100028002185
|
Coagulation Factor IX For Inj 1500 Unit
|
PA
|
|||||
|
85100028202120
|
Coagulation Factor IX (Recombinant) For Inj 250 Unit
|
PA
|
|||||
|
85100028202130
|
Coagulation Factor IX (Recombinant) For Inj 500 Unit
|
PA
|
|||||
|
85100028202140
|
Coagulation Factor IX (Recombinant) For Inj 1000 Unit
|
PA
|
|||||
|
85100030002105
|
Factor IX Complex For Inj 500 Unit
|
PA
|
|||||
|
85100030002150
|
Factor IX Complex For Inj 200-1200 Unit
|
PA
|
|||||
|
85155516000320
|
Cilostazol Tab 50 MG
|
||||||
|
85155516000330
|
Cilostazol Tab 100 MG
|
||||||
|
85158020100320
|
Clopidogrel Bisulfate Tab 75 MG (Base Equiv)
|
||||||
|
85158020100340
|
Clopidogrel Bisulfate Tab 300 MG (Base Equiv)
|
||||||
|
85158060100320
|
Prasugrel HCl Tab 5 MG (Base Equiv)
|
PA
|
|||||
|
85158060100330
|
Prasugrel HCl Tab 10 MG (Base Equiv)
|
PA
|
|||||
|
85159902206920
|
Aspirin-Dipyridamole Cap SR 12HR 25-200 MG
|
||||||
|
85200010000410
|
Pentoxifylline Tab CR 400 MG
|
||||||
|
HEMATOPOIETIC AGENTS
|
82100010002015
|
Cyanocobalamin Inj 1000 MCG/ML
|
|||||
|
82200010000315
|
Folic Acid Tab 1 MG
|
||||||
|
82300010000332
|
FERROUS SULFATE TAB 325 MG (65 MG ELEMENTAL FE)
|
OTC
|
|||||
|
82300040002010
|
Iron Dextran Inj 50 MG/ML (Elemental Iron)
|
||||||
|
82401015112010
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 25 MCG/ML
|
PA
|
|||||
|
82401015112014
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 25 MCG/0.42ML
|
PA
|
|||||
|
82401015112020
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 40 MCG/ML
|
PA
|
|||||
|
82401015112024
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 40 MCG/0.4ML
|
PA
|
|||||
|
82401015112030
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 60 MCG/ML
|
PA
|
|||||
|
82401015112034
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 60 MCG/0.3ML
|
PA
|
75
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
82401015112040
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 100 MCG/ML
|
PA
|
|||||
|
82401015112043
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 100 MCG/0.5ML
|
PA
|
|||||
|
82401015112046
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 150 MCG/0.75ML
|
PA
|
|||||
|
82401015112048
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 150 MCG/0.3ML
|
PA
|
|||||
|
82401015112050
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 200 MCG/ML
|
PA
|
|||||
|
82401015112054
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 200 MCG/0.4ML
|
PA
|
|||||
|
82401015112060
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 300 MCG/ML
|
PA
|
|||||
|
82401015112064
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 300 MCG/0.6ML
|
PA
|
|||||
|
82401015112075
|
Darbepoetin Alfa-Polysorbate 80 Soln Inj 500 MCG/ML
|
PA
|
|||||
|
82401020002010
|
Epoetin Alfa Inj 2000 Unit/ML
|
PA
|
|||||
|
82401020002015
|
Epoetin Alfa Inj 3000 Unit/ML
|
PA
|
|||||
|
82401020002020
|
Epoetin Alfa Inj 4000 Unit/ML
|
PA
|
|||||
|
82401020002040
|
Epoetin Alfa Inj 10000 Unit/ML
|
PA
|
|||||
|
82401020002050
|
Epoetin Alfa Inj 20000 Unit/ML
|
PA
|
|||||
|
82401020002060
|
Epoetin Alfa Inj 40000 Unit/ML
|
PA
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
82401520002010
|
Filgrastim Inj 300 MCG/ML
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82401520002012
|
Filgrastim Inj 480 MCG/1.6ML (300 MCG/ML)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82401520002016
|
Filgrastim Inj 300 MCG/0.5ML (600 MCG/ML)
|
PA
|
76
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
82401520002018
|
Filgrastim Inj 480 MCG/0.8ML (600 MCG/ML)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82401570002020
|
Pegfilgrastim Inj 6 MG/0.6ML
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
8240152070E530
|
Tbo-filgrastim soln prefilled syringe 300 mcg/0.5ml
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
8240152070E540
|
Tbo-filgrastim soln prefilled syringe 480 mcg/0.8ml
|
PA
|
||||
|
82402050002120
|
Sargramostim Lyophilized For Inj 250 MCG
|
PA
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
82405060002120
|
Romiplostim For Inj 250 MCG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82405060002130
|
Romiplostim For Inj 500 MCG
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82405030100310
|
Eltrombopag olamine tab 12.5 mg (base equiv)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82405030100320
|
Eltrombopag olamine tab 25 mg (base equiv)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82405030100330
|
Eltrombopag olamine tab 50 mg (base equiv)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
82405030100340
|
Eltrombopag olamine tab 75 mg (base equiv)
|
PA
|
||||
|
82803030000120
|
Hydroxyurea Cap 200 MG
|
||||||
|
82803030000130
|
Hydroxyurea Cap 300 MG
|
||||||
|
82803030000140
|
Hydroxyurea Cap 400 MG
|
||||||
|
HYPNOTICS
|
60100060000305
|
Phenobarbital Tab 15 MG
|
|||||
|
60100060000308
|
Phenobarbital Tab 16.2 MG
|
||||||
|
60100060000315
|
Phenobarbital Tab 30 MG
|
||||||
|
60100060000317
|
Phenobarbital Tab 32.4 MG
|
||||||
|
60100060000320
|
Phenobarbital Tab 60 MG
|
77
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
60100060000322
|
Phenobarbital Tab 64.8 MG
|
||||||
|
60100060000324
|
Phenobarbital Tab 97.2 MG
|
||||||
|
60100060000325
|
Phenobarbital Tab 100 MG
|
||||||
|
60100060001010
|
Phenobarbital Elixir 20 MG/5ML
|
||||||
|
60201005000310
|
Estazolam Tab 1 MG
|
||||||
|
60201005000320
|
Estazolam Tab 2 MG
|
||||||
|
60201010100105
|
Flurazepam HCl Cap 15 MG
|
||||||
|
60201010100110
|
Flurazepam HCl Cap 30 MG
|
||||||
|
60201025102002
|
Midazolam HCl Inj 2 MG/2ML (Base Equivalent)
|
LC
|
|||||
|
60201025102003
|
Midazolam HCl Inj 5 MG/5ML (Base Equivalent)
|
LC
|
|||||
|
60201025102004
|
Midazolam HCl Inj 10 MG/10ML (Base Equivalent)
|
LC
|
|||||
|
60201025102005
|
Midazolam HCl Inj 5 MG/ML (Base Equivalent)
|
LC
|
|||||
|
60201025102010
|
Midazolam HCl Inj 10 MG/2ML (Base Equivalent)
|
LC
|
|||||
|
60201025102025
|
Midazolam HCl Inj 25 MG/5ML (Base Equivalent)
|
LC
|
|||||
|
60201025102050
|
Midazolam HCl Inj 50 MG/10ML (Base Equivalent)
|
LC
|
|||||
|
60201030000103
|
Temazepam Cap 7.5 MG
|
||||||
|
60201030000105
|
Temazepam Cap 15 MG
|
||||||
|
60201030000108
|
Temazepam Cap 22.5 MG
|
||||||
|
60201030000110
|
Temazepam Cap 30 MG
|
||||||
|
60204070000120
|
Zaleplon Cap 5 MG
|
LC=30 days
|
|||||
|
60204070000130
|
Zaleplon Cap 10 MG
|
LC=30 days
|
|||||
|
60204080100310
|
Zolpidem Tartrate Tab 5 MG
|
||||||
|
60204080100315
|
Zolpidem Tartrate Tab 10 MG
|
||||||
|
MACROLIDES
|
03100005000305
|
Erythromycin Tab 250 MG
|
|||||
|
03100005000310
|
Erythromycin Tab 500 MG
|
||||||
|
03100006000605
|
Erythromycin w/ Enteric Coated Particles Tab 333 MG
|
||||||
|
03100006000610
|
Erythromycin w/ Enteric Coated Particles Tab 500 MG
|
||||||
|
03100030300305
|
Erythromycin Ethylsuccinate Tab 400 MG
|
||||||
|
03100030301910
|
Erythromycin Ethylsuccinate For Susp 200 MG/5ML
|
||||||
|
03100030301915
|
Erythromycin Ethylsuccinate For Susp 400 MG/5ML
|
||||||
|
03400010000320
|
Azithromycin Tab 250 MG
|
78
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
03400010000334
|
Azithromycin Tab 500 MG
|
||||||
|
03400010000340
|
Azithromycin Tab 600 MG
|
||||||
|
03400010001920
|
Azithromycin For Susp 100 MG/5ML
|
||||||
|
03400010001930
|
Azithromycin For Susp 200 MG/5ML
|
||||||
|
03500010000310
|
Clarithromycin Tab 250 MG
|
||||||
|
03500010000320
|
Clarithromycin Tab 500 MG
|
||||||
|
03500010001910
|
Clarithromycin For Susp 125 MG/5ML
|
||||||
|
03500010001920
|
Clarithromycin For Susp 250 MG/5ML
|
||||||
|
MEDICAL DEVICES
|
97051030056320
|
Insulin Syringe (Disp) U-100 1 ML
|
|||||
|
97051030906305
|
Insulin Syringe/Needle U-100 0.3 ML 29 x 1/2"
|
||||||
|
97051030906307
|
Insulin Syringe/Needle U-100 0.3 ML 30 x 5/16"
|
||||||
|
97051030906320
|
Insulin Syringe/Needle U-100 1/2 ML 28 x 1/2"
|
||||||
|
97051030906327
|
Insulin Syringe/Needle U-100 1/2 ML 29 x 1/2"
|
||||||
|
97051030906328
|
Insulin Syringe/Needle U-100 1/2 ML 30 x 5/16"
|
||||||
|
97051030906350
|
Insulin Syringe/Needle U-100 1 ML 27 x 1/2"
|
||||||
|
97051030906370
|
Insulin Syringe/Needle U-100 1 ML 28 x 1/2"
|
||||||
|
97051030906380
|
Insulin Syringe/Needle U-100 1 ML 29 x 1/2"
|
||||||
|
97051030906384
|
Insulin Syringe/Needle U-100 1 ML 30 x 5/16"
|
||||||
|
MIGRAINE PRODUCTS
|
AT LEAST ONE OF THE FOLLOWING
|
67406010100320
|
Almotriptan Malate Tab 6.25 MG
|
LC
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
67406010100330
|
Almotriptan Malate Tab 12.5 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406025100320
|
Eletriptan Hydrobromide Tab 20 MG (Base Equivalent)
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406025100340
|
Eletriptan Hydrobromide Tab 40 MG (Base Equivalent)
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406060100310
|
Rizatriptan Benzoate Tab 5 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406060100320
|
Rizatriptan Benzoate Tab 10 MG
|
LC
|
79
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
67406060107220
|
Rizatriptan Benzoate Orally Disintegrating Tab 5 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406060107230
|
Rizatriptan Benzoate Orally Disintegrating Tab 10 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406070002010
|
Sumatriptan Nasal Spray 5 MG/ACT
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406070002040
|
Sumatriptan Nasal Spray 20 MG/ACT
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406070100305
|
Sumatriptan Succinate Tab 25 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406070100310
|
Sumatriptan Succinate Tab 50 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406070100320
|
Sumatriptan Succinate Tab 100 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406080000320
|
Zolmitriptan Tab 2.5 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406080000330
|
Zolmitriptan Tab 5 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406080002010
|
Zolmitriptan Nasal Spray 2.5 MG/Spray Unit
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406080002020
|
Zolmitriptan Nasal Spray 5 MG/Spray Unit
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406080007220
|
Zolmitriptan Orally Disintegrating Tab 2.5 MG
|
LC
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
67406080007230
|
Zolmitriptan Orally Disintegrating Tab 5 MG
|
LC
|
||||
|
67991002100310
|
Ergotamine w/ Caffeine Tab 1-100 MG
|
LC
|
|||||
|
MINERALS & ELECTROLYTES
|
79700030000205
|
Potassium Chloride Cap CR 8 mEq
|
|||||
|
79700030000210
|
Potassium Chloride Cap CR 10 mEq
|
||||||
|
79700030000420
|
Potassium Chloride Tab CR 8 mEq (600 MG)
|
80
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
79700030000430
|
Potassium Chloride Tab CR 10 mEq
|
||||||
|
79700030000445
|
Potassium Chloride Tab CR 20 mEq (1500 MG)
|
||||||
|
79700030000910
|
Potassium Chloride Oral Liq 10% (20 MEQ/15ML)
|
||||||
|
79700030000920
|
Potassium Chloride Oral Liq 20% (40 MEQ/15ML)
|
||||||
|
MOUTH/THROAT/DENTAL AGENTS
|
88100010001805
|
Nystatin Susp 100000 Unit/ML
|
|||||
|
88100020004805
|
Clotrimazole Troche 10 MG
|
OTC
|
|||||
|
88150020102012
|
Chlorhexidine Gluconate Soln 0.12%
|
||||||
|
88350065102050
|
Lidocaine HCl Viscous Soln 2%
|
||||||
|
MULTIVITAMINS
|
WITH AT LEAST 1MG FA AND 30 MG ELEMENTAL IRON
|
78512000000115
|
*Prenatal Multivitamins & Minerals w/ Iron & FA Cap 1 MG***
|
OB/GYN ONLY
|
|||
|
78512010000352
|
*Prenatal Vit w/ Iron Carbonyl-FA Tab 50-1.25 MG***
|
OB/GYN ONLY
|
|||||
|
78512010000365
|
*Prenatal Vit w/ Iron Carbonyl-FA Tab 65-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512014000350
|
*Prenatal Vit w/ Fe Cbn-Fe Asp Glyc-FA Tab 30-20-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512015000360
|
*Prenatal Vit w/ Fe Fumarate-FA Tab 60-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512015000366
|
*Prenatal Vit w/ Fe Fumarate-FA Tab 65-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512015000385
|
*Prenatal Vit w/ Fe Fumarate-FA Tab 75-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512050000162
|
*Prenatal w/o A Vit w/ Fe Fumarate-FA Cap 106.5-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512050000330
|
*Prenatal w/o A Vit w/ Fe Fumarate-FA Tab 30-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512050000540
|
*Prenatal w/o A Vit w/ Fe Fum-FA Tab Chew 40-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512058000150
|
*Prenatal w/o A w/Fe Fum-Fe Poly-FA Cap 130-92.4-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512058000160
|
*Prenatal w/o A w/Fe Fum-Fe Poly-FA Cap 162.115.2-1MG***
|
OB/GYN ONLY
|
|||||
|
78512064000360
|
*Prenatal Vit w/ Docusate-Iron-FA Tab 65-1 MG***
|
OB/GYN ONLY
|
81
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
78512065000375
|
*Prenatal Vit w/ DSS-Iron Carbonyl-FA Tab 90-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512066000340
|
*Prenatal Vit w/o Vit A w/ Fe Bisglycinate-FA Tab 32-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512067006340
|
*Prenat w/o A w/ Fe Bisglyc-FA Tab 32-1 MG & Omega Cap Pack*
|
OB/GYN ONLY
|
|||||
|
78512068000160
|
*Prenatal w/o Vit A w/ Fe Polysac Cmplx-FA Cap 60-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512070000475
|
*Prenatal Vit w/ DSS-Fe Fumarate-FA Tab CR 90-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512072000130
|
*Prenatal w/o A w/FeCbn-Fe Asp Glyc-FA-Fish Cap 40-10- 1 MG*
|
OB/GYN ONLY
|
|||||
|
78512072000135
|
*Prenatal w/o A w/FeCbn-Fe Asp Glyc-FA-Fish Cap 50-1- 476 MG*
|
OB/GYN ONLY
|
|||||
|
78512073000140
|
*Prenat w/o A w/FeCbn-FeAspGlyc-FA-Omega Cap 35-5- 1-200 MG**
|
OB/GYN ONLY
|
|||||
|
78512079000230
|
*Prenat w/oA w/FeFum-Na Fered-FA-DHA Cap CR 30-1.4 200 MG***
|
OB/GYN ONLY
|
|||||
|
78512091000135
|
*Prenatal w/Fe Fum-Fe Poly -FA-Omega 3 Cap 53.5-38-1 MG***
|
OB/GYN ONLY
|
|||||
|
78512095000130
|
*Prenat w/ Fe Cbn-Fe Bisglyc-FA-Fish Oil Cap 35-5-1.2 MG**
|
OB/GYN ONLY
|
|||||
|
78512096006330
|
*Prenatal w/ Sod Feredetate-FA Tab 30-1 & Omega 3 Cap DR***
|
OB/GYN ONLY
|
|||||
|
78516020006330
|
*Prenatal MV w/Fe Fum-FA Tab 65-1 MG & DHA Cap 250 MG Pack *
|
OB/GYN ONLY
|
|||||
|
78516035000135
|
*Prenatal w/o Vit A w/ Fe Cbn-DSS-FA-DHA Cap 30-1- 260 MG***
|
OB/GYN ONLY
|
|||||
|
78516037000140
|
*Prenatal w/o Vit A w/ Fe Fum-DSS-FA-DHA Cap 30-1.2- 265 MG**
|
OB/GYN ONLY
|
82
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
78516037000142
|
*Prenatal w/o Vit A w/Fe Fum-DSS-FA-DHA Cap 30-1.24- 265 MG**
|
OB/GYN ONLY
|
|||||
|
78516060000145
|
*Prenat w/o A w/FeCbn-Bisg-Methylf-DSS-DHA Cap 38-1- 225 MG**
|
OB/GYN ONLY
|
|||||
|
78133000000130
|
*B-Complex w/ C & Folic Acid Cap 1 MG***
|
NEPHROLOGY
|
|||||
|
78133000000330
|
*B-COMPLEX W/ C & FOLIC ACID TAB 1 MG***
|
NEPHROLOGY
|
|||||
|
MUSCULOSKELETAL THERAPY AGENTS
|
75100010000305
|
Baclofen Tab 10 MG
|
|||||
|
75100010000310
|
Baclofen Tab 20 MG
|
||||||
|
75100050100303
|
Cyclobenzaprine HCl Tab 5 MG
|
||||||
|
75100050100304
|
Cyclobenzaprine HCl Tab 7.5 MG
|
||||||
|
75100050100305
|
Cyclobenzaprine HCl Tab 10 MG
|
||||||
|
75200010100105
|
Dantrolene Sodium Cap 25 MG
|
||||||
|
75200010100110
|
Dantrolene Sodium Cap 50 MG
|
||||||
|
75200010100115
|
Dantrolene Sodium Cap 100 MG
|
||||||
|
NASAL AGENTS - SYSTEMIC AND TOPICAL
|
42200032301810
|
Fluticasone Propionate Nasal Susp 50 MCG/ACT
|
|||||
|
OPHTHALMIC AGENTS
|
AT LEAST ONE OF THE FOLLOWING
|
86101023102010
|
Ciprofloxacin HCl Ophth Soln 0.3%
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
86101023104210
|
Ciprofloxacin HCl Ophth Oint 0.3%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
86101047002020
|
Ofloxacin Ophth Soln 0.3%
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
86103007004020
|
Ganciclovir Ophth Gel 0.15%
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
86103020002005
|
Trifluridine Ophth Soln 1%
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
86805010102010
|
Diclofenac Sodium Ophth Soln 0.1%
|
MDL (30 DAYS IN 365 DAYS)
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
86805035102015
|
Ketorolac Tromethamine Ophth Soln 0.4%
|
MDL (30 DAYS IN 365 DAYS)
|
83
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
86805035102020
|
Ketorolac Tromethamine Ophth Soln 0.5%
|
MDL (30 DAYS IN
365 DAYS)
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
86330015002010
|
Bimatoprost Ophth Soln 0.01%
|
ST
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
86330050002020
|
Latanoprost Ophth Soln 0.005%
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
86330070002020
|
Travoprost Ophth Soln 0.004%
|
ST
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
86330070002025
|
Travoprost Ophth Soln 0.004% (Benzalkonium Free) (BAK Free)
|
ST
|
||||
|
86101005004205
|
Bacitracin Ophth Oint 500 Unit/GM
|
||||||
|
86101025004210
|
Erythromycin Ophth Oint 5 MG/GM
|
||||||
|
86101030002005
|
Gentamicin Sulfate Ophth Soln 0.3%
|
||||||
|
86101030004205
|
Gentamicin Sulfate Ophth Oint 0.3%
|
||||||
|
86101070002005
|
Tobramycin Ophth Soln 0.3%
|
||||||
|
86101070004205
|
Tobramycin Ophth Oint 0.3%
|
||||||
|
86102010102010
|
Sulfacetamide Sodium Ophth Soln 10%
|
||||||
|
86109902602020
|
Polymyxin B-Trimethoprim Ophth Soln 10000 Unit/ML- 0.1%
|
||||||
|
86250010101810
|
Betaxolol HCl Ophth Susp 0.25%
|
||||||
|
86250010102005
|
Betaxolol HCl Ophth Soln 0.5%
|
||||||
|
86250020102003
|
Levobunolol HCl Ophth Soln 0.25%
|
||||||
|
86250020102005
|
Levobunolol HCl Ophth Soln 0.5%
|
||||||
|
86250030002020
|
Timolol Ophth Soln 0.25%
|
||||||
|
86250030002030
|
Timolol Ophth Soln 0.5%
|
||||||
|
86250030107620
|
Timolol Maleate Ophth Gel Forming Soln 0.25%
|
||||||
|
86250030107630
|
Timolol Maleate Ophth Gel Forming Soln 0.5%
|
||||||
|
86300020001810
|
Fluorometholone Ophth Susp 0.1%
|
||||||
|
86300020001820
|
Fluorometholone Ophth Susp 0.25%
|
84
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
86300020101810
|
Fluorometholone Acetate Ophth Susp 0.1%
|
||||||
|
86300050101809
|
Prednisolone Acetate Ophth Susp 0.12%
|
||||||
|
86300050101815
|
Prednisolone Acetate Ophth Susp 1%
|
||||||
|
86300050202015
|
Prednisolone Sodium Phosphate Ophth Soln 1%
|
||||||
|
86309902801810
|
Tobramycin-Dexamethasone Ophth Susp 0.3-0.05%
|
||||||
|
86309902801820
|
Tobramycin-Dexamethasone Ophth Susp 0.3-0.1%
|
||||||
|
86350010102010
|
Atropine Sulfate Ophth Soln 1%
|
||||||
|
86350010104210
|
Atropine Sulfate Ophth Oint 1%
|
||||||
|
86501030102015
|
Pilocarpine HCl Ophth Soln 1%
|
||||||
|
86501030102020
|
Pilocarpine HCl Ophth Soln 2%
|
||||||
|
86501030102030
|
Pilocarpine HCl Ophth Soln 4%
|
||||||
|
86602010102010
|
Apraclonidine HCl Ophth Soln 0.5% (Base Equivalent)
|
||||||
|
86602010102020
|
Apraclonidine HCl Ophth Soln 1% (Base Equivalent)
|
||||||
|
86602020102010
|
Brimonidine Tartrate Ophth Soln 0.2%
|
||||||
|
86802340102020
|
Dorzolamide HCl Ophth Soln 2%
|
||||||
|
OTIC AGENTS
|
87100060002010
|
Ofloxacin Otic Soln 0.3%
|
|||||
|
87300020102000
|
Hydrocortisone w/ Acetic Acid Otic Soln 1-2%
|
||||||
|
87400010102010
|
Acetic Acid Otic Soln 2%
|
||||||
|
87400025002010
|
Acetic Acid 2% in Aluminum Acetate Otic Soln
|
||||||
|
87991003101807
|
Neomycin-Polymyxin-HC Otic Susp 3.5 MG/ML-10000 Unit/ML-1%
|
||||||
|
87991003102010
|
Neomycin-Polymyxin-HC Otic Soln 1%
|
||||||
|
PASSIVE IMMUNIZING AGENTS
|
19502060002015
|
Palivizumab IM Soln 50 MG/0.5ML
|
PA
|
||||
|
19502060002020
|
Palivizumab IM Soln 100 MG/ML
|
PA
|
|||||
|
PENICILLINS
|
01100020001810
|
Penicillin G Benzathine Intramuscular Susp 600000 Unit/ML
|
|||||
|
01100020001815
|
Penicillin G Benzathine Intramuscular Susp 1200000 Unit/2ML
|
||||||
|
01100020001820
|
Penicillin G Benzathine Intramuscular Susp 2400000 Unit/4ML
|
85
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
01100030001820
|
Penicillin G Procaine Intramuscular Susp 600000 Unit/ML
|
||||||
|
01100040100310
|
Penicillin V Potassium Tab 250 MG
|
||||||
|
01100040100315
|
Penicillin V Potassium Tab 500 MG
|
||||||
|
01100040102105
|
Penicillin V Potassium For Soln 125 MG/5ML
|
||||||
|
01100040102110
|
Penicillin V Potassium For Soln 250 MG/5ML
|
||||||
|
01200010100105
|
Amoxicillin (Trihydrate) Cap 250 MG
|
||||||
|
01200010100110
|
Amoxicillin (Trihydrate) Cap 500 MG
|
||||||
|
01200010100303
|
Amoxicillin (Trihydrate) Tab 500 MG
|
||||||
|
01200010100315
|
Amoxicillin (Trihydrate) Tab 875 MG
|
||||||
|
01200010100505
|
Amoxicillin (Trihydrate) Chew Tab 125 MG
|
||||||
|
01200010100510
|
Amoxicillin (Trihydrate) Chew Tab 250 MG
|
||||||
|
01200010101910
|
Amoxicillin (Trihydrate) For Susp 125 MG/5ML
|
||||||
|
01200010101913
|
Amoxicillin (Trihydrate) For Susp 200 MG/5ML
|
||||||
|
01200010101915
|
Amoxicillin (Trihydrate) For Susp 250 MG/5ML
|
||||||
|
01200010101924
|
Amoxicillin (Trihydrate) For Susp 400 MG/5ML
|
||||||
|
01200020200105
|
Ampicillin Cap 250 MG
|
||||||
|
01200020200110
|
Ampicillin Cap 500 MG
|
||||||
|
01200020201910
|
Ampicillin For Susp 125 MG/5ML
|
||||||
|
01200020201915
|
Ampicillin For Susp 250 MG/5ML
|
||||||
|
01990002200310
|
Amoxicillin & K Clavulanate Tab 250-125 MG
|
||||||
|
01990002200320
|
Amoxicillin & K Clavulanate Tab 500-125 MG
|
||||||
|
01990002200340
|
Amoxicillin & K Clavulanate Tab 875-125 MG
|
||||||
|
01990002200515
|
Amoxicillin & K Clavulanate Chew Tab 200-28.5 MG
|
||||||
|
01990002200535
|
Amoxicillin & K Clavulanate Chew Tab 400-57 MG
|
||||||
|
01990002201910
|
Amoxicillin & K Clavulanate For Susp 125-31.25 MG/5ML
|
||||||
|
01990002201915
|
Amoxicillin & K Clavulanate For Susp 200-28.5 MG/5ML
|
||||||
|
01990002201920
|
Amoxicillin & K Clavulanate For Susp 250-62.5 MG/5ML
|
86
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
01990002201935
|
Amoxicillin & K Clavulanate For Susp 400-57 MG/5ML
|
||||||
|
01990002201960
|
Amoxicillin & K Clavulanate For Susp 600-42.9 MG/5ML
|
||||||
|
01990002207420
|
Amoxicillin & K Clavulanate Tab SR 12HR 1000-62.5 MG
|
||||||
|
PROGESTINS
|
26000020200305
|
Medroxyprogesterone Acetate Tab 2.5 MG
|
|||||
|
26000020200310
|
Medroxyprogesterone Acetate Tab 5 MG
|
||||||
|
26000020200315
|
Medroxyprogesterone Acetate Tab 10 MG
|
||||||
|
26000023201840
|
Megestrol Acetate Susp 625 MG/5ML
|
||||||
|
PSYCHOTHERAPEUTIC AND NEUROLOGICAL AGENTS - MISC.
|
AT LEAST ONE OF THE FOLLOWING
|
62403060452020
|
Interferon Beta-1a Inj 22 MCG/0.5ML (12MU/ML) (44 MCG/ML)
|
PA
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
62403060452040
|
Interferon Beta-1a Inj 44 MCG/0.5ML (24MU/ML) (88 MCG/ML)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
62403060452060
|
Interferon Beta-1a Inj 6 X 8.8 MCG/0.2ML & 6 X 22 MCG/0.5ML
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
62403060456420
|
Interferon Beta-1a For IM Inj Kit 30MCG (33MCG(6.6 MU)/Vial)
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
62403060456430
|
Interferon Beta-1a IM Inj Kit 30 MCG/0.5ML
|
PA
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
62403060506420
|
Interferon Beta-1b For Inj Kit 0.3 MG
|
PA
|
||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051025100310
|
Donepezil Hydrochloride Tab 5 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051025100320
|
Donepezil Hydrochloride Tab 10 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051025100330
|
Donepezil Hydrochloride Tab 23 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051030107020
|
Galantamine Hydrobromide Cap SR 24HR 8 MG
|
87
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost Containment
|
|||
|
AT LEAST TWO OF THE FOLLOWING
|
62051030107030
|
Galantamine Hydrobromide Cap SR 24HR 16 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051030107040
|
Galantamine Hydrobromide Cap SR 24HR 24 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040008520
|
Rivastigmine TD Patch 24HR 4.6 MG/24HR
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040008530
|
Rivastigmine TD Patch 24HR 9.5 MG/24HR
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040008540
|
Rivastigmine TD Patch 24HR 13.3 MG/24HR
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040200110
|
Rivastigmine Tartrate Cap 1.5 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040200120
|
Rivastigmine Tartrate Cap 3 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040200130
|
Rivastigmine Tartrate Cap 4.5 MG
|
|||||
|
AT LEAST TWO OF THE FOLLOWING
|
62051040200140
|
Rivastigmine Tartrate Cap 6 MG
|
|||||
|
62000010000310
|
Ergoloid Mesylates Tab 1 MG
|
||||||
|
62053550100320
|
Memantine HCl Tab 5 MG
|
ST
|
|||||
|
62053550100330
|
Memantine HCl Tab 10 MG
|
ST
|
|||||
|
62053550102020
|
Memantine HCl Oral Solution 2 MG/ML
|
ST
|
|||||
|
62400030106420
|
Glatiramer Acetate Inj Kit 20 MG/ML
|
PA
|
|||||
|
6240003010E540
|
Glatiramer Acetate Soln Prefilled Syringe 40 MG/ML
|
PA
|
|||||
|
62404070000320
|
Teriflunomide Tab 7 MG
|
PA
|
|||||
|
62404070000330
|
Teriflunomide Tab 14 MG
|
PA
|
|||||
|
62405050001320
|
Natalizumab for IV Inj Conc 300 MG/15ML
|
PA
|
|||||
|
62405525006320
|
Dimethyl Fumarate Capsule DR Starter Pack 120 MG & 240 MG
|
PA
|
|||||
|
62405525006520
|
Dimethyl Fumarate Capsule Delayed Release 120 MG
|
PA
|
88
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
62405525006540
|
Dimethyl Fumarate Capsule Delayed Release 240 MG
|
PA
|
|||||
|
62406030007420
|
Dalfampridine Tab SR 12HR 10 MG
|
PA
|
|||||
|
62407025100120
|
Fingolimod HCl Cap 0.5 MG (Base Equiv)
|
PA
|
|||||
|
62802010200620
|
Acamprosate Calcium Tab Delayed Release 333 MG
|
||||||
|
62802040000325
|
Disulfiram Tab 250 MG
|
||||||
|
62802040000350
|
Disulfiram Tab 500 MG
|
||||||
|
SULFONAMIDES
|
08000020000305
|
Sulfadiazine Tab 500 MG
|
|||||
|
TETRACYCLINES
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000105
|
Doxycycline Monohydrate Cap 50 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000107
|
Doxycycline Monohydrate Cap 75 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000110
|
Doxycycline Monohydrate Cap 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000115
|
Doxycycline Monohydrate Cap 150 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000305
|
Doxycycline Monohydrate Tab 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000307
|
Doxycycline Monohydrate Tab 75 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000310
|
Doxycycline Monohydrate Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020000315
|
Doxycycline Monohydrate Tab 150 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020001905
|
Doxycycline Monohydrate For Susp 25 MG/5ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020100105
|
Doxycycline Hyclate Cap 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020100110
|
Doxycycline Hyclate Cap 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020100302
|
Doxycycline Hyclate Tab 20 MG
|
89
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
04000020100310
|
Doxycycline Hyclate Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000040100105
|
Minocycline HCl Cap 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000040100107
|
Minocycline HCl Cap 75 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000040100110
|
Minocycline HCl Cap 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000040100305
|
Minocycline HCl Tab 50 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000040100307
|
Minocycline HCl Tab 75 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000040100310
|
Minocycline HCl Tab 100 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000060100105
|
Tetracycline HCl Cap 250 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
04000060100110
|
Tetracycline HCl Cap 500 MG
|
|||||
|
04000010100305
|
Demeclocycline HCl Tab 150 MG
|
||||||
|
04000010100310
|
Demeclocycline HCl Tab 300 MG
|
||||||
|
THYROID AGENTS
|
28100010100105
|
Levothyroxine Sodium Cap 13 MCG
|
|||||
|
28100010100110
|
Levothyroxine Sodium Cap 25 MCG
|
||||||
|
28100010100115
|
Levothyroxine Sodium Cap 50 MCG
|
||||||
|
28100010100120
|
Levothyroxine Sodium Cap 75 MCG
|
||||||
|
28100010100125
|
Levothyroxine Sodium Cap 88 MCG
|
||||||
|
28100010100130
|
Levothyroxine Sodium Cap 100 MCG
|
||||||
|
28100010100135
|
Levothyroxine Sodium Cap 112 MCG
|
||||||
|
28100010100140
|
Levothyroxine Sodium Cap 125 MCG
|
||||||
|
28100010100145
|
Levothyroxine Sodium Cap 137 MCG
|
||||||
|
28100010100150
|
Levothyroxine Sodium Cap 150 MCG
|
90
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
28100010100305
|
Levothyroxine Sodium Tab 25 MCG
|
||||||
|
28100010100310
|
Levothyroxine Sodium Tab 50 MCG
|
||||||
|
28100010100315
|
Levothyroxine Sodium Tab 75 MCG
|
||||||
|
28100010100317
|
Levothyroxine Sodium Tab 88 MCG
|
||||||
|
28100010100320
|
Levothyroxine Sodium Tab 100 MCG
|
||||||
|
28100010100322
|
Levothyroxine Sodium Tab 112 MCG
|
||||||
|
28100010100325
|
Levothyroxine Sodium Tab 125 MCG
|
||||||
|
28100010100327
|
Levothyroxine Sodium Tab 137 MCG
|
||||||
|
28100010100330
|
Levothyroxine Sodium Tab 150 MCG
|
||||||
|
28100010100335
|
Levothyroxine Sodium Tab 175 MCG
|
||||||
|
28100010100340
|
Levothyroxine Sodium Tab 200 MCG
|
||||||
|
28100010100345
|
Levothyroxine Sodium Tab 300 MCG
|
||||||
|
28300010000305
|
Methimazole Tab 5 MG
|
||||||
|
28300010000310
|
Methimazole Tab 10 MG
|
||||||
|
28300020000310
|
Propylthiouracil Tab 50 MG
|
||||||
|
ULCER DRUGS
|
AT LEAST ONE OF THE FOLLOWING
|
49200010000305
|
Cimetidine Tab 200 MG
|
||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200010000310
|
Cimetidine Tab 300 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200010000315
|
Cimetidine Tab 400 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200010000320
|
Cimetidine Tab 800 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200010102050
|
Cimetidine HCl Soln 300 MG/5ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200020100105
|
Ranitidine HCl Cap 150 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200020100110
|
Ranitidine HCl Cap 300 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200020100305
|
Ranitidine HCl Tab 150 MG
|
91
|
ASES MASTER FORMULARY Updated as of:
|
|
September 11, 2014 (Physical Health)
|
|
February 20,2014 (Mental Health)
|
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
AT LEAST ONE OF THE FOLLOWING
|
49200020100310
|
Ranitidine HCl Tab 300 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200020101210
|
Ranitidine HCl Syrup 15 MG/ML (75 MG/5ML)
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200030000320
|
Famotidine Tab 20 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200030000340
|
Famotidine Tab 40 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200030001920
|
Famotidine For Susp 40 MG/5ML
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200040000110
|
Nizatidine Cap 150 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200040000120
|
Nizatidine Cap 300 MG
|
|||||
|
AT LEAST ONE OF THE FOLLOWING
|
49200040002050
|
Nizatidine Oral Soln 15 MG/ML
|
|||||
|
49102070100310
|
Propantheline Bromide Tab 15 MG
|
||||||
|
49103010100105
|
Dicyclomine HCl Cap 10 MG
|
||||||
|
49103010100305
|
Dicyclomine HCl Tab 20 MG
|
||||||
|
49103010102050
|
Dicyclomine HCl Oral Soln 10 MG/5ML
|
||||||
|
49250030000310
|
Misoprostol Tab 100 MCG
|
||||||
|
49250030000320
|
Misoprostol Tab 200 MCG
|
||||||
|
49270040006510
|
Lansoprazole Cap Delayed Release 15 MG
|
LC
|
|||||
|
49270040006520
|
Lansoprazole Cap Delayed Release 30 MG
|
LC
|
|||||
|
49270060006510
|
Omeprazole Cap Delayed Release 10 MG
|
||||||
|
49270060006520
|
Omeprazole Cap Delayed Release 20 MG
|
||||||
|
49270060006530
|
Omeprazole Cap Delayed Release 40 MG
|
||||||
|
49300010000305
|
Sucralfate Tab 1 GM
|
||||||
|
49300010001820
|
Sucralfate Susp 1 GM/10ML
|
||||||
|
URINARY ANTI-INFECTIVES
|
53000050100110
|
Nitrofurantoin Macrocrystalline Cap 25 MG
|
92
|
ASES MASTER FORMULARY Updated as of:
|
|||||||
|
September 11, 2014 (Physical Health)
|
|||||||
|
February 20,2014 (Mental Health)
|
|||||||
|
Therapeutic Class
|
COMMENTS
|
GPI
|
Gpi Name
|
Cost
Containment
|
|||
|
53000050100115
|
Nitrofurantoin Macrocrystalline Cap 50 MG
|
||||||
|
53000050100120
|
Nitrofurantoin Macrocrystalline Cap 100 MG
|
||||||
|
53000050150120
|
Nitrofurantoin Monohydrate Macrocrystalline Cap 100 MG
|
||||||
|
53992005200128
|
*Methenamine-Hyosc-Meth Blue-Sod Phos-Phen Sal Cap 118 MG***
|
||||||
|
53992005200130
|
*Methenamine-Hyosc-Meth Blue-Sod Phos-Phen Sal Cap 120 MG***
|
||||||
|
53992005200320
|
*Methenamine-Hyosc-Meth Blue-Sod Phos-Phen Sal Tab 81 MG***
|
||||||
|
53992005200322
|
*Methenamine-Hyos-Meth Blue-Sod Phos-Phen Sal Tab81.6 MG***
|
||||||
|
53992005200330
|
*Methenamine-Hyosc-Meth Blue-Sod Phos-Phen Sal Tab 120 MG***
|
||||||
|
URINARY ANTISPASMODICS
|
54100045200330
|
Oxybutynin Chloride Tab 5 MG
|
|||||
|
54100045201220
|
Oxybutynin Chloride Syrup 5 MG/5ML
|
||||||
|
VACCINES
|
17200010002200
|
BCG Vaccine Inj
|
|||||
|
VAGINAL PRODUCTS
|
55100018103720
|
Clindamycin Phosphate Vaginal Cream 2%
|
|||||
|
55100035004020
|
Metronidazole Vaginal Gel 0.75%
|
||||||
|
55104070003710
|
Terconazole Vaginal Cream 0.4%
|
||||||
|
55104070003720
|
Terconazole Vaginal Cream 0.8%
|
||||||
|
55104070005210
|
Terconazole Vaginal Suppos 80 MG
|
||||||
|
55350020003705
|
Estradiol Vaginal Cream 0.1 MG/GM
|
||||||
|
VITAMINS
|
77204030000305
|
Phytonadione Tab 5 MG
|
|||||
93
ATTACHMENT 5C
PREFERRED DRUG LIST (PDL) FOR PHYSICAL HEALTH
ASES
HEALTH INSURANCE ADMINISTRATION
Commonwealth of Puerto Rico
December 19, 2012
Dear provider:
I am pleased to present to you the list of Preferred Drugs (PDL) for Physical Health from the Health Plan of the Commonwealth of Puerto Rico (“Mi Salud”), effective since January 1, 2013.
The medications included herein have been evaluated and approved by a Pharmacy and Therapeutics Committee comprised by primary physicians and clinical pharmacists.
This committee meets periodically to evaluate the therapeutical classifications and issue recommendations based on clinical aspects. Therefore, this PDL and the specialized PDLs that are included in this guide, may suffer changes, which are notified by means of Normative Letters, in conformity to how they arise.
I exhort you to conserve this Guide for your reference as long as it is necessary. The same is also available in our electronic page www.asespr.org.
Cordially,
(signed)
Frank R. Díaz Ginés, MHSA
Executive Director
|
PO Box 195661, San Juan, Puerto Rico 00919-5661
|
Tel. (787) 474-3300
|
Fax (787) 474-3346
|
GENERAL ASPECTS OF THE PHARMACY COVERAGE
FROM THE HEALTH PLAN OF THE GOVERNMENT OF PUERTO RICO
| 1. | Listing of Drugs |
The Administration of Health Insurance (ASES) is the agency responsible for establishing and reviewing the Preferred Drug List (PDL, on the basis of its initials in English) for Physical Health and Mental Health of “Mi Salud” (My Health). For this purpose, there has been established a Pharmacy and Therapeutics Committee for Physical Health, and another one for Mental Health, both comprised by different health professionals. These committees meet periodically to evaluate the different therapeutical classifications and issue their recommendations to ASES about the drug lists, based on scientific evidence and clinical aspects.
The Preferred Drug List (PDL) shall serve as a guide in the supplying of drugs under the pharmacy coverage. The PDL has the purpose of improving, putting up to date and attaining the effective cost use of drugs within the coverage of Mi Salud.
In addition, ASES maintains the Uniform Core List (FMU), which is utilized as the official listing from which the PDL is derived. The drugs in this list which do not appear in the PDL may be utilized the insurance companies, mental health organizations and service providers only as exceptions in the cases in which the drugs in the PDL were not the most clinically effective for the patient in particular.
In like manner, a List of Specialized Drugs (contracted) was created. In the situation where one provider of medical services prescribes a specialized drug from this list, it should be evaluated by means of the exception process by the MCO, MBHO, TPA or the organization contracted by ASES.
In exceptional cases drugs outside of the aforementioned lists may be utilized by means of an exception procedure.
| 2. | Exception Process |
In the case of patients who need a drug that does not appear in the PDL or in the FMU, the insurance companies, mental health organizations and health service providers must utilize the process described herein for the approval of the drugs. This procedure shall consider the particular merits of each case, which may include:
| 1. | Contraindication to the drug(s) that appear in the PDL. |
| 2. | History record of adverse reaction to the drug(s) that appear in the PDL. |
| 3. | Therapeutical failure with regard to all the alternatives available in the PDL. |
| 4. | Non-existence of a therapeutical alternative in the PDL. |
This exception process requires the official documentation from the service provider regarding the clinical reasons that justify the use of drugs outside the PDL.
| 3. | Other aspects of the pharmacy coverage |
| A. | The pharmacy coverage of Mi Salud establishes as mandatory the use of bioequivalent products, as long as it is not contraindicated and it is so clinically justified by the provider. |
| B. | The insurance companies, mental health organizations and health service providers shall process their pharmacy claims through the Pharmacy Benefit Managers (PBMs) contracted by the Administration. |
| C. | ASES has an active process to continuously review the drugs that it is required to include in the pharmacy coverage to the insurance companies or TPA, mental health organizations and health service providers. In addition, it shall evaluate any new drug for inclusion in the same or remove drugs from said listing. Due to the dynamic nature of this process, ASES may require the inclusion or exclusion of drugs pursuant to changes or advances in standards of practice within an illness or area of treatment. |
| D. | No entity contracted by ASES or the entities contracted by these ( Medical groups, IPAs, independent providers, specialists, etc.) may be ruled by a listing different from the PDL and the Core List, nor can they create an internal drug list that is different from the ones established by ASES. Neither can they, in any way, limit in a manner contrary to what is established in the contract between ASES and the insurance companies and in this coverage, the drugs that are included in said lists. The insurance companies shall be responsible for keeping tabs in their providers and employees with regard to the compliance with these provisions. |
| E. | The maximum of dispatch for severe conditions shall be to cover a thirty (30) day therapy. When medically necessary, additional prescriptions shall be covered. |
| F. | The maximum of dispatch for chronic conditions (maintenance drugs) shall be to cover a thirty (30) day therapy, except at the beginning of the therapy when, on the basis of medical criterion, a minimum of fifteen (15) days may be prescribed for the purpose of reevaluating compliance and tolerance. On the basis of a recommendation on the part of the physician, the dispatch of each prescription may be refilled up to five (5) times (original prescription plus five (5) refills). The drugs that require pre-authorization shall be in effect for six months unless there are contraindications or secondary effects. On or before the ninety (90) days after having prescribed said maintenance drug, the physician must reevaluate the pharmacotherapy for the purpose of compliance, tolerance and classification. Changes in the dosage will not require pre-authorization. Changes in the drugs utilize may require pre-authorization. |
| G. | The indications in the prescriptions for chronic use drugs, in favor of Children with Special Health Needs, must indicate clearly that they are covering a thirty (30) day therapy and that they may be refilled up to five (5) times (original prescription plus five (5) refills), according to medical criterion. When it is medically necessary, additional prescriptions shall be refilled. |
| H. | Coordination with the Department of Health is required for the supplying of birth control methods for family planning. The contraceptives for the treatment of menstrual dysfunction and for other menstrual conditions shall be covered through your primary physician from Physical Health. |
| I. | The use of bioequivalents approved by the Food and Drug Administration (FDA), classified as AB, is required, as well as authorized by the local regulations as long as it is not contraindicated and it is so consigned by the provider in the medical prescription. |
| J. | The lack of existence in the inventory of bioequivalent drugs does not exempt the dispatch of the prescribed drug nor does it imply any additional payment whatsoever on the part of the beneficiary. As a general rule, bioequivalent drugs should be dispatched as long as there exists a bioequivalent for the drug of the corresponding brand name unless, in spite of the existence of a particular bioequivalent, ASES decides to cover the brand name drug or to cover both. |
| K. | All the prescriptions must be dispatched by a pharmacy contracted by the PBM, duly authorized under the laws of the Commonwealth of Puerto Rico and freely selected by the beneficiary. |
| L. | The right to free selection requires the availability of a determinate number of pharmacies in every municipality to be able to so exercise it. The PBM is the entity in charge of contracting the network of pharmacies, pursuant to the terms specified by ASES. |
| M. | The prescribed drugs must be delivered concurrently on the date and time when the beneficiary receives the prescription and requires the dispatch of the drug. |
For any doubt regarding the pharmacy coverage you may get in touch with the insurance company contracted by ASES in your service region.
PART I - DESIGN OF THE PREFERRED DRUG LIST, PRESENTATION OF THE PREFERRED DRUG LIST (“PDL”) AND REFERENCE GUIDES
In the example that follows we can see the information that is provided for the drugs included in the PDL.
|
Indicator of Relative Cost
|
Generic Name (in bold if the drug is available in generic)
|
Brand Name
|
Reference Guides
|
|
$ sign
|
Nystatin susp.
|
Mycostatin
|
P, PA
|
For every drug included in the PDL, there appears an Indicator of Cost (Relative Cost), the Generic Name, the Brand Name and Reference Guides as applicable. In the cases in which the generic drug is available, the same appears in bold. Those generic drugs that have an asterisk (*) indicates that not all of the product's presentations have a generic available. As long as the bioequivalent generic of drug exists, the same shall be dispatched. The brand name is mentioned only for reference.
We exhort you to utilize the PDL as reference when you are going to prescribe drugs to the beneficiaries of the Health Plan of the Commonwealth of Puerto Rico.
INDICATOR OF RELATIVE COST
The indicator of relative cost is included in the PDL to offer an estimated value of the cost of a drug therapy including any discount for utilization and comparing the specific product with the other alternatives available in that classification, or to treat the specific disease or condition. In the majority of the cases, the cost per therapy for fifteen or thirty days is compared depending on whether the drug is of acute or of maintenance use, respectively. The comparison of costs and assignment of dollar signs is made on the basis of all the products included within a therapeutical classification, (for example, cardiovascular drugs, gastrointestinal drugs). The dollar signs next to a drug identify its relative cost and must be construed in the following manner:
|
$
|
Less Costly
|
|
$$
|
|
|
$$$
|
|
|
$$$$
|
|
|
$$$$$
|
|
|
$$$$$!
|
|
|
$$$$$!!
|
Most Costly
|
When several drugs within the same therapeutical classification have the same number of dollar signs, the drug that is mentioned first must be considered as the least costly one.
Where there exists an alternative for a number of adequate preparations to treat a particular disease or condition, the indicator of relative cost may be utilized to make a selection on the basis of the cost.
The designation of the relative costs is effective at the moment of the publication of this edition of the PDL. The cost of the drugs is subject to constant changes.
GENERIC DRUGS
The bioequivalent generic drugs are identified in bold. Certain bioequivalent generics have a Maximum Allowable Cost (or MAC List) for the payment of the same. This price typically covers the cost of the purchase of the generic products, but not of the brand name ones. The selection of products to be included in the MAC List are those that are prescribed commonly and have been approved by the Food and Drug Administration (FDA, on the basis of its initials in English) to be marketed.
REFERENCE GUIDES
The drugs which appear in the PDL are those preferred drugs in the coverage of the Health Plan of the Commonwealth of Puerto Rico. These drugs are selected on the basis of their safety, efficacy, high quality, existence of bioequivalents and cost. It is suggested to the physicians that they prescribe and to the pharmacists that they dispense only the drugs which are in the PDL. Ail the drugs included in this document are covered drugs unless they are designated as Unlisted (NF; for example, forms of dosage with prolonged action that are not included in the PDL).
KEY FOR THE SYMBOLS AND ABBREVIATIONS
IN THE LIST
|
$ up to $$$$$!
|
Represents the relative cost of the drug. The smaller the number of dollar signs, the lower is the cost of the drug. The nigher the number of dollar signs, the higher is the cost.
|
|
PA
|
Requires that the pharmacy endeavor a Preauthorization.
|
|
NF
|
Unlisted
|
|
P
|
Contracted brand name product “Rebates”
|
|
Bold
|
Identifies that the drug has generic bioequivalent available in all the presentations.
|
|
Bold*
|
Identifies those drugs for which not all the presentations or forms are available in generics; for example, tablets, liquids, injections, etc.
|
|
LC
|
Identifies those drugs for which there exists some limitation in the amount that the pharmacy can dispatch
|
|
AL
|
Identifies those drugs for which there exists some limitation as to the age for the dispatch of the medication.
|
|
Cap.
|
Capsule
|
|
Tab.
|
Tablet
|
|
Chew tab.
|
Chewable tablet
|
|
Disp. tab.
|
In English, dispersible tab.
|
|
Inj.
|
Injectable
|
|
Susp.
|
Suspension
|
|
ER, SR, CR
|
Prolonged action (extended release, sustained release, controlled release)
|
|
SL
|
Sublingual
|
|
Cr.
|
Creme
|
|
Oint.
|
Ointment
|
|
Sol.
|
Solution
|
|
Syr.
|
Syrup
|
|
Lot.
|
Lotion
|
|
Ophth.
|
Ophtalmic
|
|
Inh.
|
Inhaler
|
|
SNC
|
Central Nervous System
|
|
Supp.
|
Suppository
|
|
TDS
|
Transdermal Patch (transdermal release system)
|
|
TTS
|
Transdermal Patch (transdermal topic system)
|
|
OTC
|
Over the counter
|
|
OTC NF
|
Over the Counter Unlisted
|
PHYSICIAL HEALTH
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LISTING (PDL)
PHYSICAL HEALTH
2013-2014
PART II – MEDIATIONS BY THERAPEUTICAL CLASSIFICATION
|
1.0
|
ANTIINFECTIOUS
|
|||
|
1.1
|
CEPHALOSPORINS
|
|||
|
1.1.1
|
FIRST GENERATION
|
|||
|
$
|
Cephalexin cap.
|
Keflex
|
||
|
$
|
Cephalexin susp.
|
Keflex
|
||
|
$$$
|
Cefadroxil* susp.
|
Duricef
|
AL < 12 years
|
|
|
1.1.2
|
SECOND GENERATION
|
|||
|
$
|
Cefaclor cap.
|
Ceclor
|
||
|
Ceclor CD
|
NF
|
|||
|
$$$
|
Cefprozil tab., susp.
|
Cefzil
|
||
|
1.1.3
|
THIRD GENERATION
|
|||
|
$$$
|
Cefdinir cap., susp.
|
Omnicef
|
||
|
1.2
|
MACROLIDS
|
|||
|
$
|
EES/Sulfi soxazole susp.
|
Pediazole
|
||
|
$
|
Erythromycin cap.
|
|||
|
$
|
Erythromycin stearate tab.
|
Erythrocin
|
||
|
$
|
Erythromycin tab.
|
|||
|
$
|
Erythromycin EC* tab.
|
E-Mycin,
|
||
|
EryTab
|
||||
|
$
|
Erythromycin ethylsuccinate*
|
E.E.S., Eryped
|
||
|
tab., susp.
|
||||
|
$$
|
Azithromycin tab.,susp.,
|
Zithromax
|
||
|
powder pack for susp. (1 gm)
|
Zithromax Tri-Pack
|
NF
|
||
|
$$$
|
Clarithromycin tab., susp.
|
Biaxin
|
||
|
Biaxin XL
|
NF
|
|||
|
1.3
|
PENICILLINS | |||
|
$
|
Ampicillin cap., susp.
|
Principen
|
||
|
$
|
Penicillin VK tab., sol.
|
Veetids,
|
||
|
Pen-Vee K
|
||||
|
$
|
Amoxicillin* cap., tab.,
|
Trimox, Amoxil,
|
||
|
chew tab., susp.
|
Wymox
|
|||
|
$
|
Penicillin G Procaine inj.
|
|||
|
$$
|
Penicillin G Benzathine inj.
|
Bicillin LA
|
||
|
$$$
|
Amoxicillin / Clavulanic
|
Augmentin
|
||
|
acid tab., chew tab., susp.
|
Augmentin ES
|
NF
|
||
|
Augmentin XR
|
NF
|
|||
|
1.4
|
SULFONAMIDES
|
|||
|
$
|
Trimethoprim /
|
Bactrim, Bactrim DS,
|
||
|
Sulfamethoxazole tab., susp.
|
Septra, Septra DS
|
|||
|
$$$$
|
Sulfadiazine tab.
|
|||
|
1.5
|
TETRACYCLINES
|
|||
|
$
|
Doxycycline hyclate tab., cap.
|
Vibratab, Vibramycin
|
||
|
$
|
Tetracycline cap.
|
Achromycin
|
||
|
$
|
Minocycline cap.
|
Minocin
|
||
|
$$$$
|
Doxycycline syr., susp.
|
Vibramycin
|
||
|
$$$$$!
|
Demeclocycline tab.
|
Declomycin
|
||
|
1.6
|
QUINOLONES
|
|||
|
$
|
Ciprofloxacin tab. (250mg,
|
Cipro
|
||
|
500mg, 750mg)
|
||||
|
$
|
Levofloxacin tab.
|
Levaquin
|
||
|
$$$
|
Moxifloxacin tab.
|
Avelox
|
P
|
|
|
$$$$
|
Ciprofloxacin susp.
|
Cipro
|
||
|
1.7
|
ANTIVIRALS | |||
|
1.7.1
|
INFLUENZA | |||
|
$
|
Amantadine cap., syr.
|
Symmetrel
|
||
|
1.1.2
|
HERPETIC INFECTIONS | |||
|
$
|
Acyclovir cap.
|
Zovirax
|
||
|
$$
|
Acyclovir susp.
|
Zovirax
|
||
|
1.7.3
|
HIV-AIDS THERAPY & HEPATITIS
|
|||
|
1.7.3.1
|
NON-NUCLEOSIDES ANALOGOUS TO TRANSCRIPTASE
|
|||
|
$
|
Nevirapine tab., susp.
|
Viramune
|
||
|
$$$$$
|
Delavirdine tab.
|
Rescriptor
|
||
|
$$$$$!
|
Efavirenz tab., cap.
|
Sustiva
|
P
|
|
|
$$$$$!
|
Nevirapine ER tab.
|
Viramune
|
||
|
1.7.3.2
|
NUCLEOSIDES ANALOGOUS TO TRANSCRIPTASE
|
|||
|
$$
|
Stavudine cap., sol. *
|
Zerit
|
||
|
$$$
|
Zidovudine* tab., cap.,
|
Retrovir
|
||
|
syr., inj.
|
||||
|
$$$$
|
Abacavir tab., sol.
|
Ziagen
|
||
|
$$$$
|
Didanosine delayed
|
Videx EC
|
||
|
release cap.
|
||||
|
$$$$
|
Didanosine sol.
|
Videx
|
||
|
$$$$
|
Lamivudine tab., sol.
|
Epivir
|
||
|
1.7.3.3
|
NUCLEOSIDES ANALOGOUS TO TRANSCRIPTASE IN COMBINATION
|
|||
|
$$$$
|
Lamivudine / Zidovudine tab.
|
Combivir
|
||
|
$$$$$!!
|
Abacavir/Lamivudine/
|
|||
|
Zidovudine tab
|
Trizivir
|
P
|
||
|
1.7.3.4
|
PROTEASE INHIBITORS
|
|||
|
Covered via the Transmissible Diseases Prevention and Treatment Centers (CPTETs) (Inmunology Clinics)
|
||||
|
1.7.3.5
|
ORAL AGENTS FOR HEPATITIS B
|
|||
|
$$$$$!!
|
Lamivudine tab., sol.
|
Epivir HBV
|
PA
|
|
|
1.7.4
|
MISCELANEOUS ANTIVIRALS
|
|||
|
$$
|
Rimantadine tab.
|
Flumadine
|
||
|
$$$$$!!
|
Palivizumab inj.
|
Synagis
|
PA, P
|
|
|
$$$$$!!
|
Ganciclovir cap.
|
Cytovene
|
||
|
$$$$$!!
|
Valganciclovir tab.
|
Valcyte
|
||
|
1.8
|
ANTIMYCOTICS
|
|||
|
$
|
Terbinafine tab.
|
Lamisil
|
LC= 84 tab.
|
|
|
$
|
Ketoconazole tab.
|
Nizoral
|
||
|
$
|
Nystatin susp.
|
Mycostatin, Nystat
|
||
|
$
|
Fluconazole tab., susp.
|
Diflucan
|
||
|
$
|
Griseofulvin microsize tab.
|
Grifulvin V
|
||
|
$$$
|
Griseofulvin
|
Gris-PEG
|
||
|
ultramicrosize * tab.
|
||||
|
$$$
|
Clotrimazole troche
|
Mycelex
|
||
|
$$$
|
Itraconazole* cap., sol.
|
Sporanox
|
VIH-SIDA
|
|
|
$$$$$
|
Flucytosine cap. Ancobon
|
|||
|
1.9
|
ANTITUBERCULOUS
|
|||
|
$
|
Isoniazid tab.
|
|||
|
$$
|
Isoniazid syr.
|
|||
|
$$$$
|
Ethambutol tab.
|
Myambutol
|
||
|
$$$$
|
Pyrazinamide tab.
|
|||
|
$$$$
|
Rifampin cap.
|
Rifadin
|
||
|
$$$$
|
Isoniazid/Rifampin cap.
|
Rifamate
|
||
|
$$$$$
|
Ethionamide tab.
|
Trecator
|
||
|
$$$$$!
|
Rifabutin cap.
|
Mycobutin
|
||
|
$$$$$!
|
Cycloserine cap.
|
Seromycin
|
||
|
$$$$$!
|
Capreomycin inj.
|
Capastat
|
||
|
1.10
|
ANTIPARASITICS
|
|||
|
$$$
|
Iodoquinol tab.
|
Yodoxin
|
||
|
$$$
|
Albendazole tab.
|
Albenza
|
||
|
1.11
|
ANTIMALARIALS
|
|||
|
$
|
Pyrimethamine tab.
|
Daraprim
|
||
|
$
|
Hydroxychloroquine tab.
|
Plaquenil
|
||
|
$
|
Quinine sulfate* tab., cap.
|
|||
|
$
|
Primaquine phosphate tab.
|
Primaquine
|
||
|
$
|
Chloroquine phosphate tab.
|
Aralen
|
||
|
$$$
|
Mefl oquine tab.
|
Lariam
|
||
|
1.12
|
MISCELANEOUS ANTIINFECTIOUS
|
|||
|
$
|
Metronidazole tab.
|
Flagyl
|
||
|
Flagyl ER
|
NF
|
|||
|
$
|
Metronidazole Extemporaneous
|
Giardia lamblia
|
||
|
|
Preparation | |||
|
$
|
Dapsone tab.
|
Dapsone
|
||
|
$
|
Nitrofurantoin monohydrate
|
Macrobid
|
||
|
macrocrystalline cap.
|
||||
|
$
|
Clindamycin cap.
|
Cleocin
|
||
|
(150mg, 300mg)
|
||||
|
$$
|
Nitrofurantoin
|
Macrodantin
|
||
|
macrocrystals* cap.
|
||||
|
$$$
|
Clindamycin cap. (75mg), sol.
|
Cleocin
|
||
|
$$$
|
Pentamidine inh.
|
NebuPent
|
||
|
$$$$
|
Streptomycin inj.
|
|||
|
$$$$$!
|
Atovaquone susp.
|
Mepron
|
||
|
$$$$$!!
|
Vancomycin cap., oral sol.
|
Vancocin
|
||
|
$$$$$!!
|
Tobramycin inh.
|
Tobi
|
PA
|
|
|
2.0
|
CARDIOVASCULAR AGENTS FOR HYPERTENSION AND LIPIDS
|
|||
|
2.1
|
GLYCOSIDES
|
|||
|
$
|
Digoxin* tab.
|
Lanoxin
|
||
|
$$
|
Digoxin elixir
|
Lanoxin
|
||
|
2.2
|
ANTIHYPERTENSIVE THERAPY
|
|||
|
2.2.1
|
DIURETICS
|
|||
|
2.2.1.1
|
THIAZYDES
|
|||
|
$
|
Indapamide tab.
|
Lozol
|
||
|
$
|
Hydrochlorothiazide* tab.
|
Microzide
|
||
|
$
|
Chlorothiazide* tab., susp.
|
Diuril
|
||
|
$
|
Chlorthalidone tab.
|
Hygroton
|
||
|
$$
|
Metolazone tab.
|
Zaroxolyn
|
||
|
2.2.1.2
|
ANHIDRASE INHIBITORS
|
|||
|
$
|
Acetazolamide tab.
|
Diamox
|
||
|
Diamox sequels
|
NF
|
|||
|
2.2.1.3
|
LOOP DIURETICS
|
|||
|
$
|
Furosemide tab., sol.
|
Lasix
|
||
|
$
|
Bumetanide tab.
|
Bumex
|
||
|
2.2.1.4
|
MISCELLANEUOS DIURETICS
|
|||
|
$
|
Triamterene/HCTZ tab.
|
Maxzide
|
||
|
$
|
Triamterene/HCTZ cap.
|
Dyazide
|
||
|
$$
|
Spironolactone tab.
|
Aldactone
|
||
|
2.2.2
|
BETA BLOCKERS
|
|||
|
$
|
Atenolol tab.
|
Tenormin
|
||
|
$
|
Atenolol/ Chlortalidone tab.
|
Tenoretic
|
||
|
$
|
Metoprolol tartrate tab.
|
Lopressor
|
||
|
$
|
Propranolol tab.
|
Inderal
|
||
|
$
|
Propranolol/ HCTZ tab.
|
Inderide
|
||
|
$
|
Labetalol tab.
|
Normodyne
|
||
|
$
|
Pindolol tab.
|
Visken
|
||
|
$$
|
Metoprolol tartrate / HCTZ tab.
|
Lopressor HCT
|
||
|
$$
|
Propranolol sol., conc.
|
|||
|
$$
|
Metoprolol succinate SR* tab.
|
Toprol XL
|
PA
|
|
|
$$
|
Carvedilol tab.
|
Coreg
|
||
|
2.2.3
|
CALCIUM CHANNEL ANTAGONISTS | |||
|
$
|
Amlodipine tab.
|
Norvasc
|
||
|
$
|
Verapamil tab.
|
Isoptin,
|
||
|
Calan
|
||||
|
$
|
Diltiazem tab.
|
Cardizem
|
||
|
Cardizem LA
|
NF
|
|||
|
$
|
Verapamil ER tab.
|
Calan SR,
|
||
|
Isoptin SR
|
||||
|
Verelan
|
NF
|
|||
|
Verelan PM
|
NF
|
|||
|
Covera HS
|
NF
|
|||
|
$
|
Diltiazem SR 24hr cap.
|
Dilacor XR
|
||
|
$$
|
Diltiazem HCl Extended
|
Tiazac
|
||
|
Release Beads SR 24hr
|
||||
|
cap. (120mg, 180mg,
|
||||
|
240mg, 300mg, 360mg)
|
||||
|
$$
|
Diltiazem SR 12hr cap.
|
Cardizem SR
|
||
|
$$$
|
Nifedipine SR tab., CR tab.
|
Generic only
|
||
|
Adalat CC
|
NF
|
|||
|
Procardia XL
|
NF
|
|||
|
$$$
|
Diltiazem HCl coated
|
|||
|
beads SR 24hr cap.
|
Generic only
|
|||
|
2.2.4
|
ANGIOTENSINE INHIBITORS (ACE) | |||
|
$
|
Captopril tab.
|
Capoten
|
||
|
$
|
Enalapril tab.
|
Vasotec
|
||
|
$
|
Lisinopril tab.
|
Privinil, Zestril
|
||
|
$
|
Lisinopril/HCTZ tab.
|
Prinzide, Zestoretic
|
||
|
$
|
Enalapril/HCTZ tab.
|
Vaseretic
|
||
|
$
|
Captopril/HCTZ tab.
|
Capozide
|
||
|
$$
|
Fosinopril tab.
|
Monopril
|
||
|
2.2.5
|
ANGIOTENSINE RECEPTOR BLOCKERS (ARB) | |||
|
$
|
Losartan tab.
|
Cozaar
|
||
|
$
|
Losartan HCT tab.
|
Hyzaar
|
||
|
$
|
Irbesartan tab.
|
Avapro
|
||
|
$
|
Irbesartan/ HCTZ tab.
|
Avalide
|
||
|
2.2.6
|
ANTIHYPERTENSIVES WITH CENTRAL ACTION | |||
|
$
|
Clonidine tab.
|
Catapress
|
||
|
$
|
Methyldopa tab.
|
Aldomet
|
||
|
$$$$
|
Clonidine TTS
|
Catapress TTS
|
||
|
2.2.7
|
VASODILATORS | |||
|
$
|
Isosorbide dinitrate SL tab.
|
Isordil
|
||
|
$
|
Isosorbide mononitrate ER tab.
|
Imdur
|
||
|
$
|
Nitroglycerin SL tab.
|
Nitrostat
|
||
|
$
|
Isosorbide dinitrate* tab.
|
Isordil
|
||
|
$
|
Isosorbide mononitrate tab.
|
Ismo, Monoket
|
||
|
$
|
Hydralazine tab.
|
Apresoline
|
||
|
$$
|
Minoxidil tab.
|
Loniten
|
||
|
$$
|
Isosorbide dinitrate* ER
|
|||
|
tab., cap.
|
||||
|
$$$
|
Nitroglycerin Film ER TD
|
Generic only
|
||
|
NitroDur
|
NF
|
|||
|
Minitran
|
NF
|
|||
|
2.2.8
|
ALPHA RECEPTOR BLOCKERS
|
|||
|
$
|
Doxazosin tab.
|
Cardura
|
||
|
$
|
Terazosin cap.
|
Hytrin
|
||
|
2.3
|
ANTIARRHYTHMICS
|
|||
|
$$
|
Quinidine sulfate tab.
|
|||
|
$$$
|
Quinidine gluconate tab.
|
Quinaglute
|
||
|
$$$
|
Mexiletine cap.
|
Mexitil
|
||
|
$$$
|
Sotalol tab.
|
Betapace,
|
||
|
Betapace AF
|
||||
|
$$$
|
Quinidine sulfate CR tab.
|
|||
|
$$$
|
Flecainide tab.
|
Tambocor
|
||
|
$$$$
|
Amiodarone tab. (200mg,
|
Cordarone
|
||
|
400mg)
|
||||
|
$$$$
|
Propafenone tab.
|
Rythmol
|
||
|
2.4
|
CHOLESTEROL AND LIPID REDUCING AGENTS
|
|||
|
$
|
Simvastatin tab.
|
Zocor
|
||
|
$
|
Atorvastatin tab.
|
Lipitor
|
||
|
$
|
Gemfi brozil tab.
|
Lopid tab.
|
||
|
$
|
Pravastatin
|
Pravachol
|
||
|
$$
|
Cholestyramine powder packs
|
Questran
|
||
|
$$
|
Niacin CR tab.
|
Niaspan
|
P
|
|
|
3.0
|
AUTONOMIC MEDICATIONS AND FOR THE CENTRAL NERVOUS SYSTEM, NEUROLOGY AND PHYCHIATRY
|
|||
|
3.1
|
ANALGESICS, NARCOTICS AND AGENTS IN COMBINATION
|
|||
|
$
|
Meperidine inj. Demerol
|
|||
|
$
|
APAP/Codeine* tab., cap.,
|
|||
|
elixir, sol., susp.
|
||||
|
$
|
Tramadol tab.
|
Ultram
|
||
|
$
|
Methadone* tab., disp. tab., sol., conc.
|
ASSMCA
|
||
|
$
|
Morphine sulfate tab., sol.
|
|||
|
$$
|
APAP/Hydrocodone* tab.,
|
|||
|
cap., elixir
|
||||
|
$$
|
Oxycodone tab., cap., sol.
|
Roxicodone
|
||
|
$$
|
APAP/Butalbital/Caffeine
|
|||
|
tab., cap.
|
Fioricet
|
|||
|
$$
|
Hydromorphone tab., liq., inj.
|
Dilaudid
|
||
|
$$
|
Codeine sulfate tab.
|
|||
|
$$
|
Oxycodone/APAP* tab.,
|
|||
|
cap., sol.
|
||||
|
$$$$
|
Morphine sulfate SR tab., supp.
|
|||
|
$$$$
|
Fentanyl TDS
|
Duragesic
|
||
|
$$$$$
|
Hydromorphone supp.
|
Dilaudid
|
||
|
3.2
|
MIGRAINE | |||
|
$
|
Divalproex sodium ER
|
Depakote ER
|
||
|
tab. (500 mg)
|
||||
|
$
|
Ergotamine tartrate/
|
Cafergot
|
||
|
Caffeine tab.
|
||||
|
$
|
Sumatriptan tab.
|
Imitrex
|
LC=6 tab.
|
|
|
$$
|
APAP/Butalbital/Caffeine
|
|||
|
tab., cap.
|
Fioricet
|
|||
|
$$$
|
Divalproex sodium ER
|
Depakote ER
|
P
|
|
|
tab. (500 mg)
|
||||
|
$$$
|
Ergotamine tartrate/
|
Cafergot
|
||
|
Caffeine supp.
|
||||
|
$$$$
|
Sumatriptan Nasal Spray
|
Imitrex Nasal
|
LC=6
|
|
|
Spray
|
inhalers
|
|||
|
3.3
|
ANTICONVULSIVES
|
|||
|
$
|
Phenobarbital tab., elixir
|
|||
|
$
|
Clonazepam tab.
|
Klonopin
|
||
|
$
|
Carbamazepine tab.
|
Tegretol
|
||
|
$
|
Carbamazepine chew tab.
|
Tegretol
|
||
|
$
|
Phenytoin* chew tab.,
|
Dilantin
|
||
|
cap, susp.
|
||||
|
$
|
Valproic acid cap., syr.
|
Depakene
|
||
|
$
|
Divalproex sodium EC tab.,
|
|||
|
sprinkle cap.
|
Depakote
|
|||
|
$
|
Divalproex sodium ER
|
Depakote ER
|
||
|
tab. (500 mg)
|
||||
|
$$
|
Primidone tab.
|
Mysoline
|
||
|
$$
|
Carbamazepine susp.
|
Tegretol
|
||
|
$$
|
Divalproex sodium EC tab.,
|
|||
|
cap. sprinkle cap.
|
Depakote
|
P
|
||
|
$$
|
Zonisamide cap.
|
Zonegran
|
||
|
$$
|
Carbamazepine ER tab.*
|
Tegretol XR
|
||
|
$$
|
Gabapentin cap., tab.
|
Neurontin
|
||
|
$$
|
Ethosuximide cap., syr.
|
Zarontin
|
||
|
$$
|
Oxcarbazepine tab., susp.
|
Trileptal
|
||
|
$$
|
Lamotrigine tab.,
|
Lamictal, Lamictal CD
|
||
|
chew disp. tab.
|
||||
|
$$
|
Topiramate tab., cap.
|
Topamax
|
||
|
$$
|
Levetiracetam tab., sol.
|
Keppra
|
||
|
$$$
|
Divalproex sodium ER
|
Depakote ER
|
P
|
|
|
tab. (500 mg)
|
||||
|
$$$
|
Gabapentin sol. Neurontin
|
|||
|
$$$$
|
Tiagabine tab. Gabitril
|
|||
|
3.4
|
ANTIVERTIGO AND ANTIEMETICS
|
|||
|
$
|
Promethazine inj.
|
Phenergan
|
||
|
$
|
Metoclopramide syr., inj.
|
Reglan
|
||
|
$
|
Trimethobenzamide inj.
|
Tigan
|
||
|
$
|
Promethazine syr.
|
Phenergan
|
||
|
$
|
Prochlorperazine tab.
|
Compazine
|
||
|
$
|
Prochlorperazine inj.
|
Compazine inj.
|
||
|
$
|
Metoclopramide tab.
|
Reglan
|
||
|
$
|
Promethazine* tab.
|
Phenergan
|
||
|
$
|
Ondansetron tab., ODT
|
Zofran
|
||
|
tab.
|
||||
|
$$
|
Trimethobenzamide cap.,
|
|||
|
supp.
|
Tigan
|
|||
|
$$
|
Promethazine supp.
|
Phenergan
|
||
|
$$$
|
Prochlorperazine* supp.
|
Compazine
|
||
|
3.5
|
ANTIPARKINSON
|
|||
|
3.5.1
|
ANTICHOLINERGIC
|
|||
|
$
|
Benztropine tab.
|
Cogentin
|
||
|
$
|
Trihexyphenidyl HCl tab.
|
Artane
|
||
|
$
|
Trihexyphenidyl HCl elixir
|
Artane
|
||
|
3.5.2
|
DOPAMINERGICS
|
|||
|
$
|
Selegiline tab.
|
Carbex
|
||
|
$
|
Carbidopa/Levodopa tab.
|
Sinemet
|
||
|
$
|
Carbidopa/Levodopa ER tab.
|
Sinemet CR
|
||
|
$$$
|
Bromocriptine tab., cap.
|
Parlodel
|
||
|
3.5.3
|
DOPAMINE RECEPTOR AGONISTS
|
|||
|
$
|
Pramipexole tab.
|
Mirapex
|
||
|
$
|
Ropinirole tab.
|
Requip
|
||
|
3.5.4
|
MISCELLANEOUS AGENTS
|
|||
|
$
|
Amantadine cap., syr.
|
Symmetrel
|
||
|
$$$
|
Carbidopa/ Levodopa/
|
Stalevo
|
P
|
|
|
Entacapone tab.
|
||||
|
3.6
|
PSYCHOTROPIC AGENTS | |||
|
3.6.1
|
ANTIDEPRESSIVES
|
|||
|
3.6.1.1
|
TRICYCLICS
|
|||
|
$
|
Amitriptyline tab.
|
Elavil
|
||
|
$
|
Nortriptyline cap., sol.
|
Pamelor, Aventyl
|
||
|
$
|
Doxepin cap., conc.
|
Sinequan
|
||
|
$
|
Clomipramine cap.
|
Anafranil
|
||
|
$
|
Imipramine HCl tab.
|
Tofranil
|
||
|
$
|
Desipramine tab.
|
Norpramin
|
||
|
3.7
|
ANTIANXIETY/HYPNOTICS
|
|||
|
3.7.1
|
BENZODIAZEPINES
|
|||
|
$
|
Clonazepam tab.
|
Klonopin
|
||
|
$
|
Flurazepam cap.
|
Dalmane
|
||
|
$
|
Temazepam* cap.
|
Restoril
|
||
|
$
|
Chlordiazepoxide cap.
|
Librium
|
||
|
$
|
Estazolam tab.
|
ProSom
|
||
|
$
|
Clorazepate tab. T
|
ranxene
|
||
|
$
|
Lorazepam tab.
|
Ativan
|
||
|
$
|
Alprazolam tab.
|
Xanax
|
|||
|
$
|
Diazepam tab.
|
Valium
|
|||
|
$$
|
Oxazepam cap.
|
Serax
|
|||
|
$$
|
Midazolam inj.
|
Versed
|
LC=5mg/30días
|
||
|
3.8
|
SNC AGENTS AND MISCELLANEOUS
|
||||
|
$
|
Ergoloid mesylate cap.
|
Hydergine LC
|
|||
|
$$
|
Disulfi ram tab.
|
Antabuse
|
PA
|
||
|
$$$
|
Ergoloid mesylate liq.
|
Hydergine
|
|||
|
$$$
|
Pyridostigmine tab.
|
Mestinon
|
|||
|
$$$
|
Pyridostigmine CR tab.
|
Mestinon Timespan
|
|||
|
$$$
|
Ergoloid mesylate tab.
|
Hydergine
|
|||
|
$$$
|
Ergoloid mesylate SL tab.
|
Hydergine SL
|
|||
|
$$$$
|
Neostigmine tab.
|
Prostigmin
|
|||
|
$$$$
|
Pyridostigmine syr.
|
Mestinon
|
|||
|
3.9
|
ALZHEIMER
|
||||
|
$$
|
Rivastigmine cap., sol.
|
Exelon
|
|||
|
$$$
|
Donepezil tab., ODT
|
Aricept
|
|||
|
$$$$
|
Memantine tab., sol.
|
Namenda
|
ST
|
||
|
$$$$
|
Rivastigmine patch
|
Exelon patch
|
P
|
||
|
4.0
|
DERMATOLOGICAL AGENTS
|
||||
|
4.1
|
TOPICAL CORTICOSTEROIDS
|
||||
|
4.1.1
|
TOPICAL CORTICOSTEROIDS OF VERY HIGH POTENCY
|
||||
|
$
|
Clobetasol cr., oint. (0.05%)
|
Temovate
|
|||
|
$
|
Augmented betamethasone
|
Diprolene
|
|||
|
dipropionate oint. (0.05%)
|
|||||
|
$
|
Clobetasol gel (0.05%)
|
Temovate
|
|||
|
$$
|
Clobetasol sol. (0.05%)
|
Temovate
|
|||
|
4.1.2
|
TOPICAL CORTICOSTEROIDS OF HIGH POTENCY
|
||||
|
$
|
Betamethasone dipropionate
|
Diprosone
|
|||
|
cr., oint., lot. (0.05%)
|
|||||
|
$
|
Triamcinolone acetonide
|
Kenalog
|
|||
|
cr., oint. (0.5%)
|
|||||
|
$
|
Fluocinonide cr., oint., gel,
|
Lidex
|
|||
|
sol. (0.05%)
|
|||||
|
4.1.3
|
TOPICAL CORTICOSTEROIDS OF MEDIUM POTENCY
|
||||
|
$
|
Triamcinolone acetonide
|
Kenalog
|
|||
|
cr., oint. (0.1%)
|
|||||
|
$
|
Fluocinolone acetonide
|
Synalar
|
|||
|
cr., oint. (0.025%)
|
|||||
|
$
|
Betamethasone valerate
|
Valisone
|
|||
|
cr., oint., lot. (0.1%)
|
|||||
|
$
|
Mometasone furoate oint.(0.1%)
|
Elocon
|
||
|
$
|
Desoximetasone gel (0.05%)
|
Topicort
|
||
|
$$
|
Triamcinolone acetonide lot. (0.1%)
|
Kenalog
|
||
|
4.1.4
|
TOPICAL CORTICOSTEROIDS OF LOW POTENCY
|
|||
|
$
|
Desonide cr., oint, lot. (0.05%)
|
Desowen
|
||
|
$
|
Hydrocortisone cr., oint. (≥ 2%)
|
|||
|
$
|
Fluocinolone acetonide
|
Synalar
|
||
|
cr., sol. (0.01%)
|
||||
|
$$
|
Hydrocortisone lot. (≥ 2%)
|
|||
|
4.2
|
THERAPY FOR ACNE
|
|||
|
$
|
Erythromycin topical sol. (2%)
|
|||
|
$
|
Clindamycin sol. (1%)
|
Cleocin T sol.
|
||
|
$
|
Erythromycin gel (2%)
|
EryGel
|
||
|
$$
|
Tretinoin gel (0.01%, 0.025%)
|
Retin A
|
AL<21 años
|
|
|
$$
|
Tretinoin cr. (0.025%, 0.05%, 0.1%)
|
Retin A
|
AL<21 años
|
|
|
$$$
|
Isotretinoin cap. (10mg, 20mg,
|
Accutane, Sotret,
|
||
|
40mg)
|
Claravis, Amnesteem
|
|||
|
4.3
|
TOPICAL ANTIPSORIATICS
|
|||
|
$$$$
|
Tazarotene cr., gel
|
Tazorac
|
P
|
|
|
4.4
|
TOPICAL ANTIMYCOTICS
|
|||
|
$
|
Nystatin oint.
|
Mycostatin
|
||
|
$
|
Clotrimazole cr., sol. (1%)
|
Various
|
OTC
|
|
|
$
|
Miconazole nitrate powder,
|
Various
|
OTC
|
|
|
tincture (2%)
|
||||
|
$
|
Ketoconazole cr.,
|
Nizoral
|
||
|
shampoo (2%)
|
||||
|
4.5
|
TOPICAL ANTIBACTERIALS
|
|||
|
$
|
Gentamicin cr., oint.
|
Garamycin
|
||
|
$
|
Silver sulfadiazine cr.
|
Silvadene
|
||
|
$$
|
Mupirocin oint.
|
Bactroban
|
||
|
4.6
|
SCABIES DRUGS
|
|||
|
$
|
Permethrin cr. (5%)
|
Elimite
|
LC = 60gm
|
|
|
Acticin
|
NF
|
|||
|
$$$$
|
Lindane lot. (1%)
|
ST, LC=60cc
|
||
|
4.7
|
MISC. TOPICAL AGENTS
|
|||
|
$
|
Selenium sulfide
|
Selsun
|
||
|
shampoo 2.5%
|
||||
|
$$$
|
Anthralin cr. (0.5%)
|
Drithocream
|
||
|
$$$
|
Fluorouracil sol. (2%, 5%)
|
Efudex
|
||
|
$$$$
|
Fluorouracil cr. (5%)
|
Efudex
|
||
|
$$$$
|
Calcipotriene cr. (0.005%)
|
Dovonex
|
||
|
4.8
|
THERAPY FOR ROSACEA
|
|||
|
$$
|
Sulfacetamide/Sulfur cr.,
|
|||
|
lot., emulsion
|
Sulfacet – R
|
|||
|
$$$
|
Metronidazole cr., gel, lot.
|
MetroCream, MetroGel,
|
||
|
(0.75%)
|
MetroLotion
|
|||
|
$$$
|
Sulfacetamide/Sulfur susp.
|
Sulfacet-R
|
||
|
4.9
|
ORAL DERMATOLOGICAL AGENTS
|
|||
|
$$$$
|
Methoxsalen cap.
|
Oxsoralen
|
||
|
$$$$$
|
Acitretin cap.
|
Soriatane
|
P
|
|
|
4.10
|
MISCELLANEOUS ANTIPSORIATICS
|
|||
|
4.10.1
|
TNF ANTAGONISTS
|
|||
|
$$$$$!
|
Adalimumab inj.
|
Humira,
|
PA, P
|
|
|
Humira Pen
|
PA, P
|
|||
|
$$$$$!
|
Etanercept inj.
|
Enbrel
|
PA, P
|
|
|
5.0
|
MEDICATIONS FOR THE EAR, NOSE AND THROAT
|
|||
|
5.1
|
OTIC PREPARATIONS
|
|||
|
$
|
Hydrocortisone/Neomycin/
|
|||
|
Polymixin B otic sol., susp.
|
Cortisporin
|
|||
|
$
|
Burrow’s (Acetic acid 2%/
|
|||
|
Aluminum acetate) otic sol.
|
Domeboro
|
|||
|
$$
|
Ofloxacin otic sol.
|
Floxin Otic
|
||
|
Floxin Otic Singles
|
NF
|
|||
|
$$
|
Acetic acid otic sol.
|
Vosol
|
||
|
$$
|
Acetic acid/
|
Vosol-HC
|
||
|
Hydrocortisone otic sol.
|
||||
|
5.2
|
AGENTS FOR THE MOUTH AND THROAT | |||
|
$
|
Chlorhexidine gluconate sol. (0.12%)
|
Peridex
|
||
|
$
|
Lidocaine viscous sol.
|
Xylocaine
|
||
|
$
|
Hydrocortisone acetate
|
Orabase
|
||
|
dental paste
|
||||
|
$$
|
Clotrimazole troche
|
Mycelex
|
||
|
6.0
|
GASTROENTEROLOGY
|
|||
|
6.1
|
AGENTS FOR ULCERS
|
|||
|
6.1.1
|
H2 ANTAGONISTS
|
|||
|
$
|
Ranitidine tab. (300mg)
|
Zantac
|
||
|
$
|
Cimetidine tab., sol.
|
Tagamet
|
||
|
$$
|
Ranitidine syr.
|
Zantac
|
||
|
6.1.2
|
PROTON PUMP INHIBITORS | |||
|
$
|
Omeprazole cap. (10mg, 20mg)
|
Prilosec
|
||
|
6.1.3
|
MISCELLANEUOUS ULCER AGENTS | |||
|
$
|
Sucralfate tab.
|
Carafate
|
||
|
$$$
|
Sucralfate susp.
|
Carafate
|
||
|
$$$$
|
Misoprostol tab.
|
Cytotec
|
||
|
6.2
|
ANTIDIARRHEA DRUGS | |||
|
$
|
Diphenoxylate/ Atropine
|
Lomotil
|
||
|
tab., liq.
|
||||
|
6.3
|
ANTIPASMODICS | |||
|
$
|
Dicyclomine tab., cap.
|
Bentyl
|
||
|
$
|
Dicyclomine syr.
|
Bentyl
|
||
|
6.4
|
MISCELLANEOUS GASTROINTESTINAL AGENTS | |||
|
6.4.1
|
GASTROINTESTINAL AGENTS | |||
|
$
|
Sulfasalazine tab.
|
Azulfi dine
|
||
|
$$
|
Sulfasalazine EC tab.
|
Azulfi dine EN
|
||
|
$$$$
|
HC retention enema
|
Colocort, Hydrocort
|
||
|
$$$$
|
Mesalamine EC tab.
|
Asacol, Asacol HD
|
P
|
|
|
$$$$
|
Olsalazine cap.
|
Dipentum
|
||
|
$$$$$
|
Mesalamine supp.
|
Canasa
|
||
|
$$$$$
|
Mesalamine CR cap.
|
Pentasa
|
P
|
|
|
$$$$$
|
Budesonide cap.
|
Entocort EC
|
PA
|
|
|
$$$$$
|
Mesalamine enema
|
Rowasa
|
||
|
6.4.2
|
BILIARY ACIDS | |||
|
$$$
|
Ursodiol cap. (300 mg)
|
Actigall
|
||
|
6.4.3
|
DIGESTIVES | |||
|
$$
|
Pancreatic enzymes
|
Creon
|
P
|
|
|
$$
|
Pancreatic enzymes*
|
|||
|
6.4.4
|
OTHERS | |||
|
$
|
Hydrocortisone rectal cr.
|
Anusol-HC
|
||
|
(2.5%)
|
||||
|
$$
|
Hydrocortisone acetate/
|
Analpram-HC
|
|||
|
Pramoxine rectal cr.
|
|||||
|
$$$
|
Hydrocortisone acetate/
|
Proctofoam HC
|
|||
|
Pramoxine rectal foam
|
|||||
|
$$$$$
|
Cromolyn conc.
|
Gastrocrom
|
|||
|
7.0
|
ENDOCRINOLOGY AGENTS
|
||||
|
7.1
|
DIABETES THERAPY
|
||||
|
7.1.1
|
HYPOGLYCEMIC AGENTS
|
||||
|
7.1.1.1
|
SULPHONILUREA
|
||||
|
$
|
Glyburide micronized tab.
|
Generic Only
|
|||
|
$
|
Glipizide tab.
|
Glucotrol
|
|||
|
$
|
Glipizide XL tab.
|
Glucotrol XL
|
|||
|
$
|
Glyburide tab.
|
Generic Only
|
|||
|
$
|
Glimepiride tab.
|
Amaryl
|
|||
|
7.1.1.2
|
ALFA GLUCOSIDASE INHIBITORS
|
||||
|
$$$
|
Acarbose tab.
|
Precose
|
|||
|
7.1.1.3
|
TIAZOLIDINEDIONAS
|
||||
|
$$$
|
Pioglitazone tab.
|
Actos
|
|||
|
7.1.1.4
|
DPP IV INHIBITOR
|
||||
|
$$$
|
Saxagliptin tab.
|
Onglyza
|
ST, P
|
||
|
$$$
|
Saxagliptin/Metformin tab.
|
Kombiglyze
|
ST, P
|
||
|
7.1.1.5
|
INSULIN
|
||||
|
$
|
Human insulin (regular, NPH,
|
Humulin
|
P
|
||
|
70/30, lente) vial
|
Humulin Pen
|
NF
|
|||
|
$$$
|
Insulin lispro vial
|
HumaLog
|
P
|
||
|
HumaLog Mix
|
NF
|
||||
|
HumaLog Pen
|
NF
|
||||
|
$$$
|
Insulin glargine vial
|
Lantus
|
P
|
||
|
Lantus Solostar
|
P
|
||||
|
7.1.1.6
|
OTHERS
|
||||
|
$
|
Insulin syringe & needle
|
Several
|
|||
|
$
|
Metformin tab.
|
Glucophage
|
|||
|
$
|
Metformin tab.
|
Glucophage XR
|
|||
|
7.1.2
|
HIPERGLYCEMIC AGENTS
|
||||
|
$$
|
Glucagon inj.
|
||||
|
7.2
|
THYROID
|
|||
|
7.2.1
|
ANTITHYROID AGENTS
|
|||
|
$
|
Propylthiouracil tab.
|
|||
|
$$
|
Methimazole tab.
|
Tapazole
|
||
|
7.2.2
|
THYROID HORMONES
|
|||
|
$
|
Levo-thyroxine tab.
|
Several
|
||
|
$
|
Levo-thyroxine tab.
|
Synthroid
|
P
|
|
|
$
|
Levo-thyroxine tab.
|
Levoxyl
|
P
|
|
|
7.3
|
CORTICOSTEROIDS
|
|||
|
$
|
Dexamethasone sodium phosphate inj.
|
OB-GYN
|
||
|
$
|
Prednisone tab.
|
Deltasone
|
||
|
$
|
Dexamethasone tab.
|
Decadron
|
||
|
$
|
Methylprednisolone* tab.
|
Medrol
|
||
|
$
|
Prednisolone syrup
|
Prelone
|
||
|
$
|
Triamcinolone acetonide inj.
|
Kenalog,
|
||
|
Aristocort Forte
|
||||
|
Aristospan
|
NF
|
|||
|
$
|
Hydrocortisone* tab., sol.
|
Cortef
|
||
|
$
|
Cortisone acetate tab.
|
|||
|
$
|
Fludrocortisone acetate tab.
|
Florinef
|
||
|
$
|
Dexamethasone elixir,
|
Decadron
|
||
|
syrup, sol.
|
||||
|
$$
|
Betamethasone acetate &
|
Celestone
|
OB-GYN
|
|
|
sodium phosphate inj.
|
Soluspan
|
|||
|
7.4
|
GROWTH HORMONES
|
|||
|
$$$$$
|
Somatropin inj.
|
Omnitrope
|
PA, P
|
|
|
7.5
|
MISCELLANEOUS ENDOCRINOLOGICAL AGENTS
|
|||
|
7.5.1
|
ANDROGENS
|
|||
|
$$$.
|
Testosterone cypionate* in oil inj
|
Depo-Testosterone
|
||
|
7.5.2
|
ANTIHYPERPROLACTINEMIA AGENTS
|
|||
|
$$$$$
|
Cabergoline tab.
|
Dostinex
|
||
|
7.5.3
|
OTHERS
|
|||
|
$$$
|
Desmopressin acetate
|
DDAVP
|
||
|
nasal sol. (0.01%)
|
||||
|
$$$
|
Etidronate disodium tab.
|
Didronel
|
PA
|
|
|
$$$$
|
Desmopressin acetate
|
DDAVP
|
||
|
nasal spray sol. (0.01%)
|
||||
|
$$$$
|
Desmopressin acetate tab.
|
DDAVP
|
||
|
$$$$$
|
Desmopressin acetate inj.
|
DDAVP
|
||
|
(4mcg/ml vial, small vial)
|
||||
|
$$$$$!
|
Desmopressin acetate nasal
|
Stimate
|
||
|
spray sol. 1.5 mg/ml
|
||||
|
$$$$$!!
|
Octreotide acetate* inj.
|
Sandostatin
|
PA
|
|
|
Sandostatin LAR
|
PA
|
|||
|
8.0
|
MUSCULOSKELETAL SYSTEM AND RHEUMATOLOGY
|
|||
|
8.1
|
NONSTEROID ANTIINFLAMATORIES
|
|||
|
$
|
Ibuprofen tab. (≥400mg)
|
Motrin (≥400mg)
|
OTC are NF
|
|
|
$
|
Naproxen tab.
|
Naprosyn
|
||
|
$
|
Indomethacin cap.
|
Indocin
|
||
|
$
|
Salsalate tab.
|
Disalcid
|
||
|
$
|
Sulindac tab.
|
Clinoril
|
||
|
$$
|
Naproxen EC tab.
|
EC-Naprosyn
|
||
|
$$
|
Naproxen sodium tab.
|
Anaprox, Anaprox DS
|
||
|
Naprelan
|
NF
|
|||
|
$$
|
Nabumetone tab.
|
Relafen
|
||
|
$$$$
|
Indomethacin ER cap.
|
Indocin SR
|
||
|
$$$$
|
Celecoxib cap.
|
Celebrex
|
ST, P
|
|
|
8.2
|
GOUT
|
|||
|
$
|
Colchicine tab.
|
|||
|
$
|
Allopurinol tab.
|
Zyloprim
|
||
|
$
|
Probenecid tab.
|
Benemid
|
||
|
8.3
|
MUSCLE RELAXANTS
|
|||
|
$
|
Cyclobenzaprine tab. (10mg)
|
Flexeril
|
||
|
$
|
Diazepam* tab., sol., conc.
|
Valium
|
||
|
$
|
Baclofen tab.
|
Lioresal
|
||
|
$$
|
Dantrolene cap.
|
Dantrium
|
||
|
8.4
|
TNF ANTAGONISTS
|
|||
|
$$$$$!
|
Adalimumab inj.
|
Humira,
|
PA, P
|
|
|
Humira Pen
|
PA, P
|
|||
|
$$$$$!
|
Etanercept inj.
|
Enbrel
|
PA, P
|
|
|
8.5
|
MISC. RHEUMATOLOGICAL AGENTS
|
|||
|
$
|
Methotrexate* tab.
|
Rheumatrex
|
||
|
$
|
Penicillamine cap.
|
Cuprimine
|
||
|
$$$
|
Azathioprine tab.
|
Imuran
|
||
|
$$$$
|
Aurothioglucose inj.
|
Solganal
|
PA
|
|
|
$$$$$
|
Auranofin cap.
|
Ridaura
|
PA
|
|
|
9.0
|
OBSTETRICS AND GYNECOLOGY
|
|||
|
9.1
|
PRENATAL VITAMINS
|
|||
|
$
|
Prental Vitamins with iron and
|
Generic only
|
OB-GYN
|
|
|
folic acid
|
||||
|
9.2
|
BIOLOGICAL AGENTS
|
|||
|
Rho Gam- Second dose (post partum).
|
||||
|
(The Department of Health covers the first dose at selected Pharmacies – Mothers and Children Program.)
|
||||
|
9.3
|
ESTROGEN AND PROGESTIN
|
|||
|
9.3.1
|
ESTROGEN
|
|||
|
$
|
Estradiol tab.
|
|||
|
$
|
Estropipate tab.
|
|||
|
$$
|
Conjugated estrogens tab.
|
Premarin
|
P
|
|
|
(0.3mg, 0.625mg, 0.9mg,1.25mg, 2.5mg)
|
||||
|
9.3.2
|
ESTROGENS IN COMBINATION
|
|||
|
$$
|
Conjugated estrogen/
|
PremPro
|
P
|
|
|
Medroxyprogesterone tab.
|
PremPro Low Dose
|
NF
|
||
|
(0.625/2.5mg; 0.625/5mg)
|
||||
|
$$$
|
Estradiol/Norethindrone
|
Activella
|
||
|
acetate tab.
|
||||
|
9.3.3
|
PROGESTERONE
|
|||
|
$
|
Medroxyprogesterone
|
Provera
|
||
|
acetate tab.
|
||||
|
9.3.4
|
BIRTH CONTROL PILLS
|
|||
|
Available through the Department of Health
|
PA-AUB
|
|||
|
9.4
|
TOPICAL AGENTS
|
|||
|
9.4.1
|
VAGINAL ESTROGENS
|
|||
|
$$
|
Conjugated estrogen vaginal cr.
|
Premarin
|
P
|
|
|
Vaginal cream
|
||||
|
$$
|
Estradiol vaginal tab.
|
Vagifem
|
||
|
9.4.2
|
VAGINAL ANTIINFECTIOUS
|
|||
|
$$
|
Terconazole* vaginal cr., supp.
|
Terazol - 3,
|
||
|
Terazol - 7
|
||||
|
$$$
|
Clindamycin phosphate
|
|||
|
vaginal* cr., supp.
|
Cleocin
|
|||
|
$$$
|
Metronidazole vaginal gel
|
Vandazole
|
||
|
(0.75%)
|
||||
|
9.5
|
MISCELANEOUS AGENTS
|
|||
|
9.5.1
|
OSTEOPOROSIS
|
|||
|
$$
|
Alendronate tab. (includes
|
Fosamax
|
||
|
weekly dose)
|
||||
|
$$$
|
Risedronate tab.
|
Actonel
|
P
|
|
|
10.0
|
NUTRIENTS, VITAMINS AND CLOTTING THERAPY
|
|||
|
10.1
|
NUTRIENTS AND VITAMINS
|
|||
|
10.1.1
|
NUTRIENTS
|
|||
|
$
|
Potassium chloride CR tab.
|
Klor-Con
|
||
|
$
|
Potassium chloride* CR cap.
|
Kay-Ciel/ Kaon Cl
|
||
|
$
|
Potassium chloride* packs
|
Klor-Con
|
||
|
$$
|
Potassium chloride* sol.
|
Kay-Ciel/ Kaon Cl
|
||
|
$$
|
Levocarnitine tab. (330mg), sol.
|
Carnitor
|
||
|
$$
|
Iron dextran inj.
|
Infed
|
||
|
10.1.2
|
VITAMINS
|
|||
|
$
|
Phytonadione tab.
|
Mephytoin
|
||
|
$
|
Vitamin D tab., cap.
|
OTC
|
||
|
$
|
Ferrous sulfate tab. (325 mg)
|
Several
|
OTC
|
|
|
$
|
Acido fólico tab. (1 mg)
|
|||
|
$
|
Cyanocobalamin inj.
|
Vit B-12 inj.
|
||
|
$
|
Leucovorin inj.
|
|||
|
$$
|
Ergocalciferol inj.
|
Calciferol
|
||
|
$$
|
Calcitriol cap.
|
Rocaltrol
|
||
|
$$
|
Leucovorin tab.
|
|||
|
10.2
|
CLOTTING THERAPY
|
|||
|
10.2.1
|
ANTICOAGULANTS
|
|||
|
$
|
Heparin* inj.
|
|||
|
$
|
Warfarin tab.
|
Coumadin
|
||
|
$$$
|
Prasugrel tab.
|
Effient
|
PA, P
|
|
|
$$$$
|
Enoxaparin inj.
|
Lovenox
|
PA
|
|
|
10.2.2
|
ANTIPLATELETS
|
|||
|
$
|
Aspirin tab., EC tab.
|
Several
|
OTC
|
|
|
(81mg, 325mg)
|
(90 days’ supply per prescription)
|
|||
|
$$$$
|
Clopidogrel tab.
|
Plavix
|
||
|
$$$$
|
Dipyridamole/ ASA cap.
|
Aggrenox
|
||
|
10.2.3
|
ANTIHEMOPHILIC AGENTS
|
|||
|
$$$$$!!
|
Factor IX
|
Complex for inj.
|
PA
|
|
|
$$$$$!!
|
Antihemophilic factor VIII
|
Hemofil M
|
PA, P
|
|
|
(human) for inj.
|
||||
|
$$$$$!!
|
Antihemophilic factor VIII
|
Kogenate
|
PA, P
|
|
|
recombinant inj.
|
Recombinate
|
PA, P
|
||
|
Advate
|
PA, P
|
|||
|
Xyntha
|
PA, P
|
|||
|
$$$$$!!
|
Coagulation factor IX inj. PA
|
|||
|
$$$$$!!
|
Antihemophilic factor VWF PA
|
|||
|
(human) inj.
|
||||
|
$$$$$!!
|
Coagulation factor IX
|
Benefix
|
PA, P
|
|
|
recombinant inj.
|
||||
|
$$$$$!!
|
Coagulant factor VIIA
|
PA | ||
|
recombinant inj.
|
|
|||
|
$$$$$!!
|
Antiinhibitor coagulant
|
PA
|
||
|
complex inj.
|
||||
|
10.2.4
|
AGENTS FOR INTERMITENT CLAUDICATION
|
|||
|
$
|
Pentoxifylline tab.
|
Trental
|
||
|
$
|
Cilostazol tab.
|
Pletal
|
||
|
11.0
|
ANTIDOTES
|
|||
|
$$
|
Sodium polystyrene/
|
Kayexalate
|
||
|
sulfonate powder, susp.
|
Kionex
|
|||
|
$$
|
Calcium acetate cap.
|
Phoslo
|
||
|
$$$$
|
Sevelamer carbonate tab.,
|
Renvela
|
PA, P
|
|
|
powder
|
||||
|
$$$$
|
Cinacalcet tab.
|
Sensipar
|
PA, P
|
|
|
12.0
|
RESPIRATORY AGENTS
|
|||
|
12.1
|
ANTIHISTAMINES
|
|||
|
12.1.1
|
ANTIHISTAMINES
|
|||
|
$
|
Diphenhydramine cap. (50mg)
|
Benadryl
|
OTC are NF
|
|
|
$
|
Hydroxyzine pamoate cap.
|
Vistaril
|
||
|
$
|
Hydroxyzine* HCl tab., syr.
|
Atarax
|
||
|
12.1.2
|
NON-SEDATIVE ANTIHISTAMINES
|
|||
|
$
|
Loratadine OTC tab., syr.
|
Claritin
|
OTC
|
|
|
12.2
|
INTRANASAL STEROIDS
|
|||
|
$$
|
Fluticasone nasal susp.
|
Flonase
|
||
|
12.3
|
OTHERS
|
|||
|
$
|
Cromolyn nasal sol.
|
Nasalcrom
|
OTC
|
|
|
12.4
|
ASTHMA AGENTS
|
|||
|
12.4.1
|
BRONCHIODILATORS
|
|||
|
12.4.1.1
|
XANTINES
|
|||
|
$
|
Theophylline sol.
|
|||
|
$
|
Theophylline SR 12hr tab.
|
Theo-Dur
|
||
|
$
|
Theophylline elixir
|
Aerolate,
|
||
|
Elixophylline
|
||||
|
$
|
Theophylline* SR cap., CR cap.
|
Slo-BID Gyro,
|
||
|
Theo – 24, Theocap,
|
||||
|
Theo-Dur SPR
|
||||
|
$$
|
Theophylline* SR 24hr tab.
|
Uniphyl
|
||
|
$$$
|
Theophylline tab.
|
Theolair
|
||
|
12.4.1.2
|
ORAL BETA AGONISTS
|
|||
|
$
|
Albuterol tab., syr.
|
Ventolin, Proventil
|
||
|
Albuterol CR tab.
|
Volmax
|
NF
|
||
|
Proventil Repetabs
|
NF
|
|||
|
$
|
Terbutaline tab.
|
Brethine
|
||
|
12.4.1.3
|
INHALED BETA AGONISTS
|
|||
|
$
|
Albuterol inh. sol.
|
Generic Only
|
||
|
$$
|
Albuterol HFA inh.
|
ProAir HFA
|
P
|
|
|
Ventolin HFA
|
P
|
|||
|
Proventil HFA
|
NF
|
|||
|
$$
|
Levalbuterol inh.
|
Xopenex HFA
|
P
|
|
|
$$$
|
Formoterol inh.
|
Foradil
|
P
|
|
|
$$$$
|
Salmeterol inh., diskus
|
Serevent
|
||
|
12.4.1.4
|
ANTICHOLINERGICS
|
|||
|
$
|
Ipratropium Br inh. sol.
|
Atrovent
|
||
|
$$$
|
Ipratropium Br inh.
|
Atrovent HFA
|
||
|
$$$
|
Tiotropium inhalation powder
|
Spiriva
|
PA
|
|
|
12.4.1.5
|
INHALED CORTICOSTEROIDS
|
|||
|
$$
|
Beclomethasone inh.
|
Qvar
|
P
|
|
|
$$$
|
Fluticasone inhalation powder
|
Flovent HFA
|
P
|
|
|
Flovent Diskus
|
P
|
|||
|
$$$$
|
Budesonide inh. susp.
|
Pulmicort
|
||
|
(respules)
|
||||
|
12.4.1.6
|
AGENTS IN COMBINATION
|
|||
|
$$$
|
Albuterol / Ipratropium
|
Combivent
|
ST
|
|
|
bromide inh.
|
||||
|
$$$$
|
Fluticasone / Salmeterol powder
|
Advair Diskus
|
ST, P
|
|
|
Advair HFA
|
ST, P
|
|||
|
12.4.1.7
|
MISCELLANEOUS AGENTS
|
|||
|
$
|
Montelukast tab., chew tab.
|
Singulair
|
||
|
$
|
Cromolyn sodium inh. sol.
|
Intal
|
||
|
$$$
|
Cromolyn inh.
|
Intal inh.
|
||
|
12.5
|
ANTITUSSIVES AND EXPECTORANTS
|
|||
|
$
|
Codeine / Guaifenesin
|
Generic Only
|
||
|
liq., syr.
|
||||
|
13.0
|
OPHTALMIC AGENTS
|
|||
|
13.1
|
OPHTALMIC ANTIBIOTICS
|
|||
|
$
|
Gentamicin ophth. sol.
|
Garamycin
|
||
|
$
|
Erythromycin ophth. oint.
|
|||
|
$
|
Tobramycin ophth. sol.
|
Tobrex
|
||
|
$
|
Bacitracin ophth. oint.
|
Bacitracin
|
||
|
$
|
Sodium sulfacetamide
|
Bleph 10
|
||
|
ophth. sol. (10%)
|
||||
|
$
|
Trimethoprim/Polymyxin B
|
Polytrim
|
||
|
ophth. sol.
|
||||
|
$
|
Sodium sulfacetamide ophth.
|
|||
|
oint. (10%)
|
||||
|
$
|
Ofloxacin ophth. sol.
|
Ocuflox
|
||
|
$
|
Gentamicin ophth. oint.
|
Garamycin
|
||
|
$
|
Ciprofloxacin ophth. sol.
|
Ciloxan
|
||
|
$$
|
Tobramycin ophth. oint.
|
Tobrex
|
||
|
$$
|
Ciprofloxacin ophth. oint.
|
Ciloxan
|
||
|
$$$
|
Trifluridine ophth. sol.
|
Viroptic
|
PA
|
|
|
13.2
|
OPHTALMIC ANTIINFLAMATORIES
|
|||
|
13.2.1
|
CORTICOSTEROIDS
|
|||
|
$
|
Prednisolone acetate ophth.
|
|||
|
susp. (1%)
|
Pred Forte
|
|||
|
$
|
Fluorometholone ophth.
|
|||
|
susp. (0.1%) FML
|
Liquifi lm
|
|||
|
$$
|
Prednisolone phosphate
|
|||
|
ophth. sol. (1%)
|
Inflamase
|
|||
|
$$
|
Fluorometholone acetate
|
Efl one
|
||
|
ophth. susp. (0.1%)
|
Flarex
|
NF
|
||
|
13.2.2
|
NON STEROIDAL ANTIINFLAMMATORIES
|
|||
|
$$$
|
Ketorolac ophth. sol.
|
Acular
|
||
|
(max 30 days in 365 days)
|
||||
|
Acular LS
|
||||
|
Acular PF
|
NF
|
|||
|
$$$
|
Diclofenac ophth. sol.
|
Voltaren
|
||
|
(max 30 days in 365 days)
|
||||
|
13.3
|
PRODUCTS IN COMBINATION
|
|||
|
$$$
|
Tobramycin/Dexamethasone
|
Tobradex
|
||
|
ophth. susp.
|
Tobradex oint.
|
NF
|
||
|
13.4
|
AGENTS FOR GLAUCOMA
|
|||
|
13.4.1
|
MYOTICS
|
|||
|
$
|
Pilocarpine ophth. sol.
|
|||
|
13.4.2
|
SELECTIVE ADRENERGIC AGONISTS
|
|||
|
$
|
Brimonidine ophth. sol. (0.2%)
|
Alphagan
|
||
|
Alphagan P
|
NF
|
|||
|
13.4.3
|
BETA BLOCKERS
|
|||
|
$
|
Timolol* ophth. sol.
|
Timoptic
|
||
|
$
|
Levobunolol ophth. sol.
|
Betagan
|
||
|
$
|
Betaxolol* ophth. sol. (0.5%)
|
Betoptic
|
||
|
Betopic S
|
NF
|
|||
|
$
|
Timolol XE ophth. gel
|
Timoptic-XE
|
||
|
13.4.4
|
ANHIDRASE INHIBITORS
|
|||
|
$
|
Dorzolamide ophth. sol.
|
Trusopt Plus
|
||
|
$
|
Brinzolamide ophth. susp.
|
Azopt
|
P
|
|
|
13.4.5
|
PROSTAGLANDINES
|
|||
|
$
|
Latanoprost ophth. sol.
|
Xalatan
|
||
|
$$
|
Bimatoprost ophth. sol.
|
Lumigan
|
ST, P
|
|
|
$$
|
Travaprost ophth. sol.
|
Travatan Z
|
ST, P
|
|
|
13.5
|
MISCELLANEOUS OPHTALMIC AGENTS
|
|||
|
$
|
Atropine ophth. sol., oint.
|
Iso-Atropine
|
||
|
14.0
|
UROLOGY
|
|||
|
14.1
|
ANTISPASMODICS
|
|||
|
$
|
Oxybutinin tab.
|
Ditropan
|
||
|
Ditropan XL
|
NF
|
|||
|
$$
|
Oxybutynin syr.
|
Ditropan
|
||
|
14.2
|
ANESTHESICS
|
|||
|
$
|
Phenazopyridine tab.
|
Pyridium
|
LC= 6 tab.
|
|
|
(100 mg, 200 mg)
|
||||
|
14.3
|
MISCELLANEOUS UROLOGICAL AGENTS
|
|||
|
$
|
Methenamine-hyosciaminemethylene
|
|||
|
blue-sod biphosphenyl
|
Urin D/S,
|
|||
|
salicilate tab. 81.6 mg
|
Uretron D/S
|
|||
|
$$
|
Finasteride tab. (5 mg)
|
Proscar
|
||
|
15.0
|
CANCER
|
|||
|
There shall be covered under the pharmacy coverage only the oral presentations of the cancer products which are detailed as follows. Other presentations shall be covered through the ambulatory chemotherapy clinics.
|
||||
|
15.1
|
ANTIMETABOLITS
|
|||
|
$$
|
Methotrexate tab.
|
|||
|
$$$
|
Mercaptopurine tab.
|
Purinethol
|
||
|
$$$
|
Thioguanine tab.
|
|||
|
$$$$$!
|
Capecitabine tab.
|
Xeloda
|
PA
|
|
|
15.2
|
ALKALATING AGENTS
|
|||
|
$$
|
Lomustine cap.
|
CEENU
|
||
|
$$$$
|
Busulfan tab.
|
Myleran
|
||
|
$$$$$
|
Melphalan tab.
|
Alkeran
|
||
|
$$$$$!
|
Cyclophosphamide tab.
|
Cytoxan
|
||
|
$$$$$!
|
Chlorambucil tab.
|
Leukeran
|
||
|
$$$$$!!
|
Temozolamide cap.
|
Temodar
|
PA
|
|
|
$$$$$!!
|
Procarbazine cap.
|
Matulane
|
||
|
15.3
|
ANDROGENS, ESTROGENS, PROGESTINS
|
|||
|
15.3.1
|
PROGESTINS
|
|||
|
$$$
|
Megestrol acetate tab., susp.
|
Megace
|
||
|
$$$$
|
Medroxyprogesterone acetate
|
Depo-Provera
|
PA
|
|
|
inj. (400 mg)
|
||||
|
15.3.2
|
ANTIANDROGENS
|
|||
|
$$
|
Bicalutamide tab.
|
Casodex
|
PA
|
|
|
$$
|
Flutamide cap.
|
Eulexin
|
PA
|
|
|
15.3.3
|
BREAST CANCER
|
|||
|
15.3.3.1
|
ANTISTROGENS
|
|||
|
$
|
Tamoxifen tab.
|
|||
|
15.3.3.2
|
AROMATASE INHIBITORS
|
|||
|
$
|
Letrozole tab.
|
|||
|
$
|
Anastrozole tab.
|
|||
|
$
|
Exemestane tab.
|
|||
|
15.3.4
|
HORMONAS
|
|||
|
$$$$$!!
|
Estramustine cap.
|
Emcyt
|
||
|
15.4
|
MISCELANEOUS CANCER AGENTS
|
|||
|
$$$
|
Hydroxyurea cap.
|
Hydrea
|
||
|
$$$$
|
Leuprolide inj.
|
Eligard
|
PA, P
|
|
|
(all the presentations)
|
||||
|
$$$$$
|
Leuprolide inj.
|
Lupron Depot
|
PA, P
|
|
|
(all the presentations)
|
||||
|
$$$$$!
|
Etoposide cap.
|
Vepesid
|
||
|
$$$$$!
|
Dasatinib tab.
|
Sprycel
|
PA, P
|
|
|
$$$$$!
|
Sunitinib cap.
|
Sutent
|
PA, P
|
|
|
$$$$$!
|
Sorafenib tab.
|
Nexavar
|
PA, P
|
|
|
$$$$$!
|
Nilotinib
|
Tasigna
|
PA, P
|
|
|
$$$$$!
|
Everolimus tab. Afinitor PA, P
|
|||
|
$$$$$!!
|
Goserelin implant
|
Zoladex
|
PA
|
|
|
$$$$$!!
|
Mitotane tab.
|
Lysodren
|
||
|
$$$$$!!
|
Imatinib tab., cap.
|
Gleevec
|
PA
|
|
|
15.5
|
INMUNOSUPRESSORS
|
|||
|
$$$$
|
Azathioprine tab.
|
Imuran
|
||
|
$$$$
|
Cyclosporine modifi ed cap., sol.
|
Neoral
|
PA, P
|
|
|
$$$$
|
Cyclosporine modifi ed
|
Generic
|
PA
|
|
|
cap., sol.
|
only
|
|||
|
Gengraf
|
NF
|
|||
|
$$$$$
|
Cyclosporine* cap., sol.
|
Sandimmune
|
PA
|
|
|
$$$$$
|
Cyclosporine cap., sol.
|
Sandimmune
|
PA, P
|
|
|
$$$$$!
|
Sirolimus tab., sol.
|
Rapamune
|
PA
|
|
|
$$$$$!
|
Mycophenolate sodium tab.
|
Myfortic
|
PA, P
|
|
|
$$$$$!
|
Mycophenolate mofetil tab.,
|
CellCept
|
PA
|
|
|
cap., liq.
|
||||
|
$$$$$!
|
Tacrolimus cap.
|
Prograf
|
PA
|
|
|
16.0
|
BIOTECHNOLOGY
|
|||
|
16.1
|
MULTIPLE SCLEROSIS
|
|||
|
$$$$$!!
|
Glatiramer acetate inj.
|
Copaxone
|
PA, P
|
|
|
$$$$$!!
|
Interferon beta-1A inj.
|
Avonex
|
PA, P
|
|
|
$$$$$!!
|
Interferon beta-1B inj.
|
Extavia
|
PA, P
|
|
|
$$$$$!!
|
Mitoxantrone inj.
|
Novantrone
|
PA
|
|
|
16.2
|
ERYTHROID STIMULANTS
|
|||
|
$$$$$
|
Darbepoetin alfa inj.
|
Aranesp
|
PA, P
|
|
|
$$$$$
|
Epoetin alfa inj.
|
Procrit
|
PA, P
|
|
|
16.3
|
MYELOID STIMULANTS
|
|||
|
$$$$$!
|
Sargramostim inj.
|
Leukine
|
PA, P
|
|
|
$$$$$!!
|
Filgrastim inj.
|
Neupogen
|
PA, P
|
|
|
$$$$$!!
|
Pegfi lgrastim inj.
|
Neulasta
|
PA, P
|
|
|
16.4
|
INTERFERONS
|
|||
|
$$$$$!!
|
Interferon alfa-2B inj.
|
Intron A
|
PA
|
|
|
$$$$$!!
|
Interferon Gamma-1B inj.
|
Actimmune
|
PA
|
|
Revised 12/31/2013
NF Unlisted
PA Requires preauthorization
P Contracted brand name product (rebate)
Bold Generic bioequivalent available in all the presentations
Bold* Some presentations of the medications are not available in generic
LC Limit as to the amount to be dispatched
ST: Step therapy, Clinical protocol for its use
AL Age limit
OB-GYN Only in Obstetrics-Gynecology listing
VIH-AIDS Only in HIV-AIDS listing
OTC-Over the Counter
OTC-Over the Counter-Unlisted
PART III - APPENDIX I
PRODUCTS WITH A LIMITATION
AS TO THE AMOUNT TO BE DISPATCHED
|
1.
|
Antimycotics
|
|
Product
|
Generic
Name
|
Limitation
|
|
Lamisil
|
Terbinafine tab.
|
Maximum 12 weeks
|
| 2. | Triptanes (Agents for migraine) |
|
Product
|
Generic Name
|
Limitation
|
|
Imitrex
|
Sumatriptan nasal spray
|
Maximum amount within a 30 day period = 6 inhalers
|
|
Imitrex
|
Sumatriptan tab.
|
Maximum amount within a 30 day
period = 6 tablets
|
| 3. | Antianxiety/Hypnotic |
|
Product
|
Generic Name
|
Limitation
|
|
Versed
|
Midazolam inj.
|
Maximum amount within a 30 day period = 5 mg.
|
| 4. | Scabicides |
|
Product
|
Generic Name
|
Limitation
|
|
Elimite
|
Permethrin cr. (5%)
|
Maximum amount within a 30 day period = 60 gm.
|
|
Lindane
|
Lindane lot. (1%)
|
Maximum amount within a 30 day period = 60 cc
|
| 5. | Anesthesics |
|
Product
|
Generic Name
|
Limitation
|
|
Pyridium
|
Phenazopyridine tab.
|
Maximum treatment for three days = 6 tablets
|
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST DENTAL 2013-2014
|
1.0
|
ANTIINFECTIOUS
|
|||
|
1.1
|
CEPHALOSPORINES
|
|||
|
1.1.1
|
FIRST GENERATION
|
|||
|
$
|
Cephalexin cap.
|
Keflex
|
||
|
$
|
Cephalexin susp.
|
Keflex
|
||
|
$$$
|
Cefadroxil* susp.
|
Duricef
|
AL < 12 years
|
|
|
1.2
|
MACROLIDS
|
|||
|
$
|
Erythromycin cap.
|
|||
|
$
|
Erythromycin stearate tab.
|
Erythrocin
|
||
|
$
|
Erythromycin tab.
|
|||
|
$
|
Erythromycin EC* tab.
|
E-Mycin,
|
||
|
EryTab
|
||||
|
$
|
Erythromycin ethylsuccinate* tab., chew tab, susp.
|
E.E.S.,
|
||
|
1.3
|
PENICILLINS
|
|||
|
$
|
Ampicillin cap., susp.
|
Principen
|
||
|
$
|
Penicillin VK tab., sol.
|
Veetids,
|
||
|
Pen-Vee K
|
||||
|
$
|
Amoxicillin* cap., tab., chew tab., susp.
|
Trimox,
Amoxil,
|
||
|
Wymox
|
||||
|
1.4
|
ANTIINFECTIOUS MISCELLANEOUS
|
|||
|
$
|
Clindamycin cap.
|
|||
| Cleocin | ||||
|
$$$
|
(150mg, 300mg)
Clindamycin cap. (75mg), sol.
|
Cleocin
|
||
|
2.0
|
NARCOTIC ANALGESICS AND AGENTS IN COMBINATION
|
|||
|
$
|
APAP/ Codeine* tab., cap.,
|
|||
|
elixir, sol., susp.
|
||||
|
$
|
APAP/ Hydrocodone tab.,
|
|||
|
cap., elixir
|
||||
|
3.0
|
NON-STEROID ANTIINFLAMMATORIES
|
|||
|
$
|
Ibuprofen tab. (>400mg)
|
Motrin (>400mg)
|
OTC are NF
|
|
|
$
|
Naproxen tab.
|
Naprosyn
|
||
|
$$
|
Naproxen EC tab.
|
EC-Naprosyn
|
||
|
$$
|
Naproxen sodium tab.
|
Anaprox, Anaprox DS
|
||
|
Naprelan
|
NF
|
|||
|
Revided 12/31/13
|
||||
|
Erype
|
||||
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST
EMERGENCY ROOMS
2013-2014
|
1.0
|
ANTIINFECTIOUS | |||
|
1.1.1
|
CHEPHALOSPORINES | |||
|
1.1.1
|
FIRST GENERATION | |||
|
$
|
Cephalexin cap.
|
Keflex
|
||
|
$
|
Cephalexin susp.
|
Keflex
|
||
|
$$$
|
Cefadroxil* susp.
|
Duricef
|
AL « 12 years
|
|
|
1.1.2
|
SECOND GENERATION | |||
|
$$
|
Cefaclor cap.
|
Ceclor
|
||
|
Ceclor CD
|
NF
|
|||
|
$$$
|
Cefprozil tab., susp.
|
Cefzil
|
||
|
1.1.3
|
THIRD GENERATION
|
|||
|
$$$
|
Cefdinir cap., susp.
|
Omnicef
|
||
|
1.1.2
|
MACROLIDS
|
|||
|
$
|
EES/Sulfisoxasole susp.
|
Pediazole
|
||
|
$
|
Erythromycin cap.
|
|||
|
$
|
Erythroycim stearate tab.
|
Erythrocin
|
||
|
$
|
Erythromycin tab.
|
|||
|
$
|
Erythromycin EC*tab.
|
E-Mycin,
|
||
|
Ery Tab
|
||||
|
$
|
Erythromycin ethylsuccinate*
|
E.E.S.,
|
||
|
Tab., susp.
|
Eryped
|
|||
|
$$$
|
Azithromycin tab.,susp.,
powder pack for susp. (1 gm)
|
Zithromax
Zithromax Tri-Pack
|
||
|
$$$
|
Clarithromycin tab., susp.
|
Biaxin
|
NF
|
|
|
1.3
|
PENICILINS
|
|||
|
$
|
Ampicillin cap., susp.
|
Principen
|
||
|
$
|
Penicillin VK tab., sol.
|
Veetids,
|
||
|
Pen-Vee K
|
||||
|
$
|
Amoxicillin* cap., tab.,
|
Trimox,
|
||
|
Chew tab., susp.,
|
Amoxil
|
|||
|
Wymox
|
||||
|
$$$
|
Amoxicillin / Clavulanic
|
Augmentin
|
||
|
acid tab., chew tab., susp.
|
Augmentin ES
|
NF
|
||
|
Augmentin XR
|
NF
|
|||
|
1.4
|
SULFONAMIDES
|
|||
|
$
|
Trimethoprim /
|
Bactrim, Bactrim DS,
|
||
|
Sulfamethoxazole tab., susp.
|
Septra, Septra DS
|
|||
|
1.5
|
TETRACYICLINES
|
|||
|
$
|
Doxycycline hyclate tab., cap.
|
Vibratab,
Vibramycin
|
||
|
$
|
Tetracycline cap.
|
Achromycin
|
||
|
$$$$
|
Doxycycline syr., susp.
|
Vibramycin
|
||
|
1.6
|
ANTIMYCOTICS
|
|||
|
$
|
Nystatin susp.
|
Mycostatin, Nystat
|
||
|
1.7
|
ANTIVIRALS
|
|||
|
$
|
Acyclovir tab., cap.
|
Zovirax
|
||
|
$$
|
Acyclovir susp.
|
Zovirax
|
||
|
1.8.
|
QUNINOLONES
|
|||
|
$
|
Ciprofloxacin tab. (250mg, 500mg, 750mg)
|
Cipro
|
||
|
$
|
Levofloxacin tab.
|
Levaquin
|
||
|
$$$$
|
Ciprofloxacin susp.
|
Cipro
|
||
|
1.9
|
MISCELLANEOUS ANTIINFECTIOUS
|
|||
|
$
|
Metronidazole tab.
|
Flagyl
|
||
|
Flagyl ER
|
NF | |||
|
$
|
Nitrofurantoin/monohydrate Macrobid
|
|||
|
Macrocrystals* cap.
|
||||
|
$
|
Clindamycin cap.
(150mg, 300mg)
|
Cleocin
|
||
|
$$
|
Nitrofurantoin macrocrystals* cap.
|
Macrodantin
|
||
|
$$$
|
Clindamycin cap. (75mg), susp.
|
Cleocin
|
||
|
2.0
|
CARDIOVASCULAR AGENTS FOR HYPERTENSION AND LIPDS
|
|||
|
2.1
|
GLYCOSIDES
|
|||
|
$
|
Digoxin* tab.
|
Lanoxin
|
||
|
$$
|
Digoxin elixir
|
Lanoxin
|
||
|
2.2
|
ANTIHYPERTENSIVE THERAPY
|
|||
|
2.2.1
|
DIURETICS
|
|||
|
2.2.1.1
|
THIAZIDES
|
|||
|
$
|
Hydrochlorothiazide* tab.
|
Microzide
|
||
|
2.2.1.2
|
LOOP DIURETICS
|
|||
|
$
|
Furosemide tab., sol.
|
Lasix
|
||
|
2.2.2
|
BETA BLOCKERS
|
|||
|
$
|
Atenolol tab.
|
Tenormin
|
||
|
$
|
Atenolol/ HCTZ
|
Tenoretic
|
||
|
$
|
Metoprolol tartrate tab.
|
Lopressor
|
||
|
$$
|
Metroprolol/HCTZ
|
Lopressor HCT
|
||
|
2.2.3
|
CALCIUM CHANNEL ANTAGONISTS
|
||
|
$
|
Verapamil tab.
|
Isoptin, Calan
|
|
|
2.2.4
|
ANGIOTENSINE INHIBITORS (ACE)
|
||
|
$
|
Captopril tab.
|
Capoten
|
|
|
$
|
Captopril/HCTZ
|
Capozide
|
|
|
$
|
Enalapril tab.
|
Vasotec
|
|
|
$
|
Enalapril/HCTZ
|
Vaseretic
|
|
|
2.2.5
|
ANGIOTESINE RECEPTOR BLOCKER (ARB)
|
||
|
$
|
Losartan tab.
|
Cozaar
|
|
|
2.2.6
|
ANTIHYPTERTENSIVES WITH CENTRAL ACTION
|
||
|
$
|
Clonidine tab.
|
Catapress
|
|
|
2.2.7
|
VASODILATORS
|
||
|
$
|
Nitroglycerin SL tab.
|
Nitrostat
|
|
|
3.0
|
AUTONOMIC DRUGS FOR THE CENTRAL NERVOUS SYSTEM, NEUROLOGY AND PSYCHIATRY
|
||
|
3.1
|
ANALGESICS, NARCOTICS AND AGENTS IN COMBINATION
|
||
|
$
|
APAP/Codeine* tab., elixir, sol., susp.
|
||
|
$
|
Tramadol tab.
|
Ultram
|
|
|
$$
|
APAP/Butalbital/Caffeine tab., cap.
|
Fioricet
|
|
|
3.2
|
ANTICONVULSIVES
|
||
|
$
|
Phenobarbital tab., elixir
|
||
|
$
|
Phenytoin* chew tab., cap, susp.
|
Dilantin
|
|
|
$$
|
Levetiracetam tab., sol. Keppra
|
||
|
3.3
|
ANTIVERTIGO AND ANTIEMETICS
|
||
|
$
|
Promethazine Syr.
|
Phenergan
|
|
|
$
|
Prochlorperazine tab.
|
Compazine
|
|
|
$
|
Metoclopramide tab., syr., inj.
|
Reglan
|
|
|
$
|
Promethazine* tab.
|
Phenergan
|
|
|
$$
|
Trimethobenzamide cap., supp.
|
Tigan
|
|
|
$$
|
Promethazine supp.
|
Phenergan
|
|
|
$$$
|
Prochlorperazine* supp.
|
Compazine
|
|
|
4.0
|
DERMATOLOGICAL AGENTS/ TOPICAL THERAPY
|
|||
|
4.1
|
TOPICAL ANTIBATERIAL
|
|||
|
$
|
Gentamicin cr., oint.
|
Garamycin
|
||
|
$
|
Silver sulfadiazine cr.
|
Silvadene
|
||
|
$$
|
Mupirocin oint.
|
Bactroban
|
||
|
4.2
|
ESCABIDES
|
|||
|
$$$$
|
Lindane lot. (1%)
|
ST, LC = 60cc
|
||
|
5.0
|
DRUGS FOR THE EARS AND THROAT
|
|||
|
5.1
|
OTIC PREPARATIONS
|
|||
|
$
|
Burrow’s (Acetic acid 2%/
|
|||
|
Aluminum acetate) otic sol.
|
Domeboro
|
|||
|
$
|
Hydrocortisone/Neomycin/
|
|||
|
Polymixin B otic sol., susp.
|
Cortisporin
|
|||
|
$
|
Ofloxacin otic sol.
|
Floxin Otic
|
||
|
$$
|
Acetic acid otic sol.
|
Vosol
|
||
|
$$
|
Acetic acid/ Hydrocortisone otic sol.
|
Vosol-HC
|
||
|
$$$$
|
Ciprofloxacin/Hydrocortisone
|
Cipro HC
|
||
|
5.2
|
AGENTS FOR THE MOUTH AND THROAT | |||
|
$
|
Lidocaine viscous sol.
|
Xylocaine
|
||
|
$$$
|
Clotrimazole troche
|
Mycelex
|
||
|
6.0
|
GASTROENTEROLOGY
|
|||
|
6.1
|
AGENTS FOR ULCERS
|
|||
|
6.1.1
|
H2 ANTAGONISTS
|
|||
|
$
|
Ranitidine tab. (300mg)
|
Zantac
|
||
|
$
|
Cimetidine tab., sol.
|
Tagamet
|
||
|
$$
|
Ranitidine syr.
|
Zantac
|
||
|
6.1.2
|
PROTON PUMP INHIBITOR
|
|||
|
$
|
Omeprazole cap. Prilosec (10mg, 20mg)
|
|||
|
6.2
|
ANTIDIARRHEICS
|
|||
|
$
|
Diphenoxylate/ Atropine Tab., liq.
|
Lomotil
|
||
|
7.0
|
ENDOCRINOLOGICAL AGENTS | |||
|
7.1
|
DIABETES THERAPY
|
|||
|
7.1.1.
|
HIPOGLYCEMIC AGENTS
|
|||
|
7.1.1.1
|
SULFONILUREANS
|
|||
|
$
|
Glipizide tab.
|
Glucotrol
|
||
|
$
|
Glipizide XL tab.
|
Glucotrol XL
|
||
|
$
|
Glimepiride tab.
|
Amaryl
|
||
|
7.1.1.2
|
INSULIN
|
|||
|
$
|
Human insulin (regular) vial
|
Humulin R P
|
||
|
7.1.1.3.
|
OTHERS
|
|||
|
$
|
Insulin syringe & needle
|
Varios
|
||
|
$
|
Metformin tab.
|
Glucophage
|
||
|
7.2
|
CORTICOSTEROIDS
|
|||
|
$
|
Prednisone tab.
|
Deltasone
|
||
|
$
|
Dexamethasone tab.
|
Decadron
|
||
|
$
|
Dexamethasone elixir, syrup, sol.
|
Decadron
|
||
|
$
|
Methylprednisolone* tab.
|
Medrol
|
||
|
$
|
Prednisolone syrup
|
Prelone
|
||
|
8.0
|
MUSCULOSKELETAL SYSTEM AND RHEUMATOLOGY
|
|||
|
8.1
|
NON-STEROIDAL ANIINFLAMMATORIES
|
|||
|
$
|
Ibuprofen tab. (≥400mg)
|
Motrin (≥400mg) OTC are NF
|
||
|
$
|
Naproxen tab.
|
Naprosyn
|
||
|
$
|
Indomethacin cap.
|
Indocin
|
||
|
$
|
Salsalate tab.
|
Disalcid
|
||
|
$$
|
Naproxen sodium tab.
|
Anaprox,
|
||
|
Anaprox DS
|
||||
|
Naprelan NF
|
||||
|
$$
|
Nabumetone tab.
|
Relafen
|
||
|
8.2
|
GOUT
|
|||
|
$
|
Colchicine tab.
|
Colcrys
|
||
|
8.3
|
MUSCLE RELAXANTS
|
|||
|
$
|
Cyclobenzaprine tab. (10mg)
|
Flexeril
|
||
|
9.0
|
COAGULATION THERAPHY
|
|||
|
9.1
|
ANTICOAGULANTS
|
|||
|
$
|
Warfarin tab.
|
Coumadin
|
||
|
9.2
|
ANTIPLATELETS
|
|||
|
$
|
Clopidogrel tab.
|
Plavix
|
||
|
10.0
|
RESPIRATORY AGENTS
|
|||
|
10.1
|
ANTIHISTAMINES
|
|||
|
$
|
Diphenhydramine cap. (50mg)
|
Benadryl cap.
|
OTC are NF
|
|
|
$
|
Hydroxyzine pamoate cap.
|
Vistaril
|
||
|
$
|
Hydroxyzine* HCl tab., syr.
|
Atarax
|
||
|
10.2
|
ASTHMA AGENTS
|
|||
|
10.2.1
|
BRONCODILATORS
|
|||
|
10.2.1.1
|
BETA ORAL AGONISTS
|
|||
|
$
|
Albuterol tabl, syr.
|
Ventolin
|
||
|
Albuterol CR tab.
|
Volmax
|
NF
|
||
|
Proventil Repetab
|
NF
|
|||
|
$$
|
Terbutaline tab.
|
Brethine
|
||
|
10.2.1.2
|
INHALED BETA AGONISTS
|
|||
|
$
|
Albuterol inh. sol.
|
Ventolin HFA
|
Generic only
|
|
|
$$
|
Albuterol HFA
|
LC = 1 pump / 30 days
|
P
|
|
|
Proventil HFA
|
NF
|
|||
|
ProAir HFA
|
NF
|
|||
|
10.2.1.3
|
ANTICHOLINERGICS
|
|||
|
10.3
|
ANTITUSSIVES AND EXPECTORANTS
|
|||
|
$
|
Codeine / Guaifenesin liq.
|
Generic only
|
||
|
11.0
|
OPHTHALMIC AGENTS
|
|||
|
11.1
|
OPHTHALMIC ANTIBIOTICS
|
|||
|
$
|
Gentamicin ophth. sol.
|
Garamycin
|
||
|
$
|
Gentamicin ophth. oint.
|
Garamycin
|
||
|
$
|
Trimethoprim/Polymyxin B ophth. sol.
|
Polytrim
|
||
|
$
|
Tobramycin ophth. sol.
|
Tobrex
|
||
|
11.2
|
OPHTHALMIC ANTIINFLAMMATORIES
|
|||
|
$
|
Prednisolone acetate ophth. susp. (1%)
|
Pred Forte
|
||
|
12.0
|
UROLOGY
|
|||
|
12.1
|
ANESTHESICS
|
|||
|
$
|
Phenazopyridine tab.
|
Pyridium
|
LC=6 tab.
|
|
|
(100mg, 200mg)
|
||||
|
12.2
|
MISCELLANEOUS UROLOGICAL AGENTS
|
|||
|
$
|
Methenamine-hyosciaminemethylene
|
|||
|
blue-sod biphosphenyl
|
Urin D/S,
|
|||
|
salicilate tab. 81.6 mg
|
Uretron D/S
|
|||
|
Revised 12/31/2013
|
||||
NEPHROLOGY
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST
NEPHROLOGY
2013-2014
|
1.0
|
ANTIINFECTIOUS
|
|||
|
1.1
|
CEPHALOSPORINES
|
|||
|
1.1.1
|
FIRST GENERATION
|
|||
|
$
|
Cephalexin cap.
|
Keflex
|
||
|
$
|
Cephalexin susp.
|
Keflex
|
||
|
$$$
|
Cefadroxil* susp.
|
Duricef
|
AL less than 12 years
|
|
|
1.1.2
|
SECOND GENERATION
|
|||
|
$
|
Cefaclor cap.
|
Ceclor
|
||
|
Ceclor CD
|
NF
|
|||
|
$$$
|
Cefprozil tab., susp.
|
Cefzil
|
||
|
1.1.3
|
THIRD GENERATION
|
|||
|
$$$
|
Cefdinir cap., susp.
|
Omnicef
|
||
|
1.2
|
MACROLIDS
|
|||
|
$
|
EES/Sulfisoxazole susp.
|
Pediazole
|
||
|
$
|
Erythromycin cap.
|
|||
|
$
|
Erythromycin stearate tab.
|
Erythrocin
|
||
|
$
|
Erythromycin tab.
|
|||
|
$
|
Erythromycin EC* tab.
|
E-Mycin,
|
||
|
EryTab
|
||||
|
$
|
Erythromycin
ethylsuccinate* tab., susp.
|
E.E.S., Eryped
|
||
|
$$
|
Azithromycin tab., susp.,
|
Zithromax
|
||
|
powder pack for susp. (1 gm)
|
Zithromax Tri-Pack
|
NF
|
||
|
$$$
|
Clarithromycin tab., susp.
|
Biaxin
|
||
|
Biaxin XL
|
NF
|
|||
|
1.3
|
PENICILLINS
|
||||
|
$
|
Ampicillin cap., susp.
|
Principen
|
|||
|
$
|
Penicillin VK tab., sol.
|
Veetids,
|
|||
|
Pen-Vee K
|
|||||
|
$
|
Amoxicillin* cap., tab.
|
Trimox,
|
|||
|
chew tab., susp.
|
Amoxil,
|
||||
|
Wymox
|
|||||
|
$
|
Penicillin G Procaine inj.
|
||||
|
$$
|
Penicillin G Benzathine inj.
|
Bicillin LA
|
|||
|
$$$
|
Amoxicillin/Clavulanic
|
Augmentin
|
|||
|
acid tab., susp.
|
Augmentin ES
|
NF
|
|||
|
Augmentin XR
|
NF
|
||||
|
1.4
|
SULFONAMIDES
|
||||
|
$
|
Trimethoprim/
|
Bactrim, Bactrim DS,
|
|||
|
Sulfamethoxazole tab., susp.
|
Septra, Septra DS
|
||||
|
1.5
|
QUINOLONES
|
||||
|
$
|
Ciprofloxacin tab. (250 mg,
|
Cipro
|
|||
|
500 mg, 750 mg)
|
|||||
|
$
|
Levofloxacin tab. Levaquin
|
||||
|
$$$
|
Moxifloxacin tab.
|
Avelox
|
P
|
||
|
$$$$
|
Ciprofloxacin susp.
|
Cipro
|
|||
|
2.0
|
CARDIOVASCULAR AGENTS FOR HYPERTENSION AND LIPIDS
|
|||
|
2.1
|
ANTIHYPERTENSIVE THERAPY
|
|||
|
2.1.1
|
DIURETICS
|
|||
|
2.1.1.1
|
THIAZIDES
|
|||
|
$
|
Hydrochlorothiazide* tab.
|
Microzide
|
||
|
$
|
Chlorothiazide* tab., susp.
|
Diuril
|
||
|
$
|
Chlorthalidone tab.
|
Hygroton
|
||
|
$$
|
Metolazone tab.
|
Zaroxolyn
|
||
|
2.1.1.2 LOOP DIURETICS
|
||||
|
$
|
Furosemide tab., sol.
|
Lasix
|
||
|
$
|
Bumetanide tab.
|
Bumex
|
||
|
2.1.2
|
BETA BLOCKERS
|
|||
|
$
|
Atenolol tab.
|
Tenormin
|
||
|
$
|
Atenolol/Chlortalidone tab.
|
Tenoretic
|
||
|
$
|
Metoprolol tartrate tab.
|
Lopressor
|
||
|
$
|
Propranolol tab.
|
Inderal
|
||
|
$
|
Propranolol/HCTZ tab.
|
Inderide
|
||
|
$
|
Labetalol tab.
|
Normodyne
|
||
|
$$
|
Metoprolol tartrate/HCTZ tab.
|
Lopressor HCT
|
||
|
$$
|
Propranolol sol., conc.
|
|||
|
$$
|
Metoprolol succinate SR* tab.
|
Toprol XL
|
||
|
$$
|
Carvedilol tab.
|
Coreg
|
||
|
2.1.3
|
CALCIUM CHANNEL ANTAGONISTS
|
|||
|
$
|
Amlodipine tab.
|
Norvasc
|
||
|
$
|
Verapamil tab.
|
Isoptin,
|
||
|
Calan
|
||||
|
$
|
Ditiazem tab.
|
Cardizem
|
||
|
Cardizem LA
|
NF
|
|||
|
$
|
Verapamil ER tab.
|
Calan SR,
|
||
|
Isoptin SR
|
||||
|
Verelan
|
||||
|
Verelan PM
|
NF
|
|||
|
Covera HS
|
NF
|
|||
|
|
$
|
Diltiazem SR 24 hr cap.
|
Dilacor XR
|
|
|
$$
|
Diltiazem HCL Extended
|
Tiazac
|
||
|
Release Beads SR 24 hr
|
||||
|
cap. (120mg, 180mg,
|
||||
|
240mg, 300mg, 360mg)
|
||||
|
$$
|
Diltiazem SR 12 hr cap.
|
Cardizem SR
|
||
|
$$$
|
Nifedipine SR tab., CR tab.
|
Generic only
|
||
|
Adalat CC
|
NF
|
|||
|
Procardia XL
|
NF
|
|||
|
$$$
|
Diltiazem HCI coated
|
Generic only
|
||
|
beads SR 24hr cap.
|
||||
|
2.1.4
|
ANGIOTENSINE INHIBITORS (ACE)
|
||||
|
$
|
Captopril tab.
|
Capoten
|
|||
|
$
|
Enalapril tab.
|
Vasotec
|
|||
|
$
|
Lisinopril tab.
|
Privinil,
|
|||
|
Zestril
|
|||||
|
$
|
Captopril HCTZ
|
Capozide
|
|||
|
$
|
Enalapril HCTZ
|
Vaseretic
|
|||
|
$
|
Lisinopril HCTZ
|
Prinzide,
|
|||
|
Zestoretic
|
|||||
|
$$
|
Fosinopril tab.
|
Monopril
|
|||
|
2.1.5
|
ANGIOTENSINE RECEPTOR BLOCKERS (ARB)
|
||||
|
$
|
Losartan tab.
|
Cozaar
|
|||
|
$
|
Losartan HCT tab.
|
Hyzaar
|
|||
|
$
|
Irbesartan tab.
|
Avapro
|
ST, P
|
||
|
$
|
Irbesartan/HCTZ tab.
|
Avalide
|
ST, P
|
||
|
2.1.6
|
VASODILATORS
|
||||
|
2.1.6.1
|
ALPHA RECEPTOR BLOCKERS
|
||||
|
$
|
Doxazosin tab.
|
Cardura
|
|||
|
$
|
Terazosin cap.
|
Hytrin
|
|||
|
2.2
|
CHOLESTEROL AND LIPID REDUCING AGENTS
|
||||
|
$
|
Simvastatin tab.
|
Zocor
|
|||
|
$
|
Atorvastatin tab.
|
Lipitor
|
|||
|
$
|
Gemfibrozil tab.
|
Lopid tab.
|
|||
|
Lopid cap.
|
|||||
|
$
|
Pravastatin
|
Pravachol
|
|||
|
$$$
|
Cholestyramine powder packs
|
Questran
|
|||
|
$$$
|
Niacin CR tab.
|
Niaspan
|
P
|
||
|
3.0
|
ANTIVERTIGO AND ANTIEMETICS
|
||||
|
$
|
Metoclopramide syr., inj.
|
Reglan
|
|||
|
$
|
Metoclopramide tab.
|
Reglan
|
|||
|
4.0
|
GASTROENTEROLOGY
|
||||
|
4.1
|
AGENTS FOR ULCERS
|
||||
|
4.1.1
|
H2 ANTAGONISTS
|
||||
|
$
|
Ranitidine tab. (300 mg)
|
Zantac
|
|||
|
$
|
Cimetidine tab., sol.
|
Tagamet
|
|||
|
$
|
Ranitidine syr.
|
Zantac
|
|||
|
4.2
|
ANTIDIARRHEA
|
||||
|
$
|
Diphenoxylate/
|
Lomotil
|
|||
|
Atropine tab., liq.
|
|||||
|
5.0
|
ENDOCRINOLOGIC AGENTS
|
|||
|
5.1
|
DIABETES THERAPY
|
|||
|
5.1.1
|
HYPOGLYCEMIC AGENTS
|
|||
|
5.1.1.1.1 SULFONILUREAS
|
||||
|
$
|
Glyburide micronized tab.
|
Generic Only
|
||
|
$
|
Glipizide tab.
|
Glucotrol
|
||
|
$
|
Glipizide XL tab. G
|
lucotrol XL
|
||
|
$
|
Glyburide tab.
|
Generic Only
|
||
|
$
|
Glimepiride
|
Amaryl
|
||
|
5.1.1.2 ALPHA GLUCOSIDASE INHIBITORS
|
||||
|
$$$
|
Acarbose tab.
|
Precose
|
||
|
5.1.1.3 THIAZOLIDINEDIONES
|
||||
|
$$$$
|
Pioglitazone tab.
|
Actos
|
||
|
5.1.1.4 DPP IV INHIBITOR
|
||||
|
$$$ Saxagliptin tab.
|
Onglyza
|
ST, P | ||
|
$$$ Saxagliptin/Metformin tab.
|
Kombiglyze
|
ST, P | ||
|
5.1.1.5 INSULIN
|
||||
|
$
|
Human insulin (regular, NPH,
|
Humulin
|
P
|
|
|
70/30, lens) vial
|
Humulin Pen
|
NF
|
||
|
$$$
|
Insulin lispro vial
|
Humalog
|
P
|
|
|
Humalog Mix
|
NF
|
|||
|
Humalog Pen
|
NF
|
|||
|
$$$
|
Insulin glargine vial
|
Lantus
|
P
|
|
|
Lantus Solostar
|
P
|
|||
|
5.1.1.6 OTHERS
|
||||
|
$
|
Insulin syringe & needles
|
Several
|
||
|
$
|
Metformin tab.
|
Glucophage
|
||
|
$
|
Metformin XR tab
|
Glucophage XR
|
||
|
5.2
|
CORTICOSTEROIDS
|
|||
|
$
|
Prednisone tab.
|
Deltasone
|
||
|
$
|
Dexamethasone tab.
|
Decadron
|
||
|
$
|
Methylprednisolone* tab.
|
Medrol
|
||
|
$
|
Prednisolone syrup
|
Prelone
|
||
|
$
|
Hydrocortisone* tab., sol.
|
Cortef
|
||
|
$
|
Fludrocortisone acetate tab.
|
Florinef
|
||
|
$
|
Dexamethasone elixir,
|
Decadron
|
||
|
syrup, sol.
|
||||
|
5.3
|
MISCELLANEOUS ENDOCRINOLOGIC AGENTS
|
|||
|
$$$
|
Desmopressin acetate
|
DDAVP
|
||
|
nasal sol. (0.01%)
|
||||
|
$$$$
|
Desmopressin acetate
|
DDAVP
|
||
|
nasal spray sol. (0.01%)
|
||||
|
$$$$$!
|
Desmopressin acetate nasal
|
Stimate
|
||
|
spray sol. 1.5 mg/ml
|
||||
|
6.0
|
NUTRIENTS AND VITAMINS
|
|||
|
6.1
|
NUTRIENTS
|
|||
|
$
|
Potassium chloride CR tab.
|
Klor-Con
|
||
|
$
|
Potassium chloride* CR cap.
|
Kay-Ciel, Kaon Cl
|
||
|
$
|
Potassium chloride* packs
|
Klor-Con
|
||
|
$$
|
Potassium chloride* sol.
|
Kay-Ciel, Kaon CI
|
||
|
$$
|
Iron dextran inj.
|
INFed
|
||
|
6.2
|
VITAMINS
|
|||
|
$
|
Vitamin D tab., cap.
|
OTC
|
||
|
$
|
Ferrous sulfate tab. (325 mg)
|
Several
|
OTC
|
|
|
$
|
Folic acid tab. (1 mg)
|
|||
|
$
|
Cyanocobalamin inj.
|
Vit. B-12 inj.
|
||
|
$$
|
Ergocalciferol inj.
|
Calciferol
|
||
|
$$
|
Calcitriol cap.
|
Rocaltrol
|
||
|
7.0
|
ANTIDOTES
|
|||
|
$$
|
Sodium polystyrene/
|
Kayexalate
|
||
|
sulfonate powder, susp.
|
Kionex
|
|||
|
$$
|
Calcium acetate cap.
|
Phoslo
|
||
|
$$$$
|
Sevelamer carbonate tab.
|
Renvela
|
PA, P
|
|
|
$$$$
|
Cinacalcet tab.
|
Sensipar
|
PA, P
|
|
|
8.0
|
IMMUNOSUPPRESSORS
|
|||
|
$$$$
|
Azathioprine tab.
|
Imuran
|
||
|
$$$$
|
Cyclosporine modified cap., sol.
|
Neoral
|
PA, P
|
|
|
$$$$
|
Cyclosporine modified
|
Generic only
|
PA
|
|
|
cap., sol.
|
||||
|
Gengraf
|
NF
|
|||
|
$$$$$
|
Cyclosporine* cap., sol.
|
Sandimmune
|
PA
|
|
|
$$$$$
|
Cyclosporine cap., sol.
|
Sandimmune
|
PA, P
|
|
|
$$$$$!
|
Sirolimus tab., sol.
|
Rapamune
|
PA
|
|
|
$$$$$!
|
Mycophenolate sodium tab.
|
Myfortic
|
PA, P
|
|
|
$$$$$!!
|
Mycophenolate mofetil tab.,
|
CellCept
|
PA
|
|
|
cap., liq.
|
PA
|
|||
|
$$$$$!!
|
Tacrolimus cap.
|
Prograf
|
||
|
9.0
|
BIOTECHNOLOGY
|
|||
|
9.1
|
ERYTHROID STIMULANTS
|
|||
|
$$$$$
|
Darbepoetin alpha inj.
|
Aranesp
|
PA, P
|
|
|
$$$$$
|
Epoetin alpha inj.
|
Procrit
|
PA, P
|
|
|
10.0
|
MISCELLANEOUS AGENTS
|
|||
|
$
|
Indomethacin cap.
|
Indocin
|
||
|
$$$
|
Megestrol acetate tab., susp.
|
Megace
|
||
|
$$$
|
Indomethacin ER cap.
|
Indocin
|
||
Revised 12/31/2013
Code (for all the pages):
NF - Unlisted
P - Contracted brand name product (rebate)
Bold - Bioequivalent generic available in all presentations
Bold* - Some presentations of the drugs are not available in generic
ST - Step Therapy, Clinical protocol for its use
AL - Age Limitation
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST
OBSTETRICS-GYNECOLOGY
2013-2014
|
1.0
|
ANTIINFECTIOUS
|
|||
|
1.1
|
CEPHALOSPORINES
|
|||
|
1.1.1
|
FIRST GENERATION
|
|||
|
$
|
Cephalexin cap.
|
Keflex
|
||
|
$
|
Cephalexin susp.
|
Keflex
|
||
|
1.1.2
|
SECOND GENERATION
|
|||
|
$
|
Cefaclor cap.
|
Ceclor
|
||
|
Ceclor CD
|
NF
|
|||
|
$$$
|
Cefprozil tab., susp.
|
Cefzil
|
||
|
1.1.3
|
THIRD GENERATION
|
|||
|
$$$
|
Cefdinir cap., susp.
|
Omnicef
|
||
|
1.2
|
MACROLIDS
|
|||
|
$
|
Erythromycin cap.
|
|||
|
$
|
Erythromycin stearate tab.
|
Erythromycin
|
||
|
$
|
Erythromycin tab.
|
|||
|
$
|
Erythromycin EC* tab.
|
E-Mycin
|
||
|
EryTab
|
||||
|
$
|
Erythromycin
|
|||
|
ethylsuccinate* tab susp.
|
E.E.S., Eryped
|
|||
|
$$
|
Azithromycin susp., powder
|
Zithromax
|
||
|
pack for susp. (1 gm)
|
Zithromax
|
|||
|
Zithromax Tri-Pack
|
NF
|
|||
|
1.3
|
PENICILLINS
|
|||
|
$
|
Ampicillin cap., susp.
|
Principen
|
||
|
$
|
Penicillin VK tab., sol.
|
Veetids,
|
||
|
Pen-Vee K
|
||||
|
$
|
Amoxicillin* cap., tab.
|
Trimox,
|
||
|
chew tab., susp.
|
Amoxil,
|
|||
|
Wymox
|
||||
|
$
|
Penicillin G Procaine inj.
|
|||
|
$$
|
Penicillin G Benzathine inj.
|
Bicillin LA
|
||
|
$$$
|
Amoxicillin, Clavulanic
|
Augmentin
|
||
|
acid tab., susp.
|
Augmentin ES
|
NF
|
||
|
Augmentin XR
|
NF
|
|||
|
1.4
|
SULFONAMIDES
|
|||
|
$
|
Trimethoprim/
|
Bactrim, Bactrim DS
|
||
|
Sulfamethoxazole tab.
|
Septra, Septra DS
|
|||
|
susp.
|
||||
|
1.5
|
ANTIVIRALS
|
|||
|
1.5.1
|
HERPETIC INFECTIONS
|
|||
|
Acyclovir tab., cap.
|
Zovirax
|
|||
|
$
|
Acyclovir susp.
|
Zovirax
|
||
|
$$
|
||||
|
1.5.2
|
HIV-AIDS THERAPY
|
||||
|
1.5.2.1
|
ANALOGOUS NUCLEOSIDES OF TRANSCRIPTASE
|
||||
|
$$$$$
|
Zidovudine* tab., cap.,
|
Retrovir
|
|||
|
syr., inj.
|
|||||
|
1.5.2.2
|
PROTEASE INHIBITORS
|
||||
|
Covered through the Prevention and Treatment Centers for Transmissible Diseases (CPTETs) (Immunology Clinics)
|
|||||
|
1.6
|
ANTITUBERCULOUS
|
||||
|
$
|
Isoniazid tab.
|
||||
|
$$
|
Isoniazid syr.
|
||||
|
$$$$
|
Ethambutol tab.
|
Myambutol
|
|||
|
$$$$
|
Rifampin cap.
|
Rifadin
|
|||
|
$$$$
|
Isoniazid/Rifampin cap.
|
Rifamate
|
|||
|
$$$$$
|
Ethionamide tab.
|
Trecator
|
|||
|
$$$$$!
|
Rifabutin cap.
|
Mycobutin
|
|||
|
1.7
|
MISCELLANEOUS ANTIINFECTIOUS
|
||||
|
$
|
Metronidazole tab.
|
Flagyl
|
|||
|
Flagyl ER
|
NF
|
||||
|
$
|
Dapsone tab.
|
Dapsone
|
|||
|
$
|
Clindamycin cap.
|
Cleocin
|
|||
|
(150mg, 300mg)
|
|||||
|
$$
|
Nitrofurantoin
|
Macrodantin
|
|||
|
macrocystals* cap.
|
|||||
|
$$$
|
Clindamycin cap. (75mg)
|
Cleocin
|
|||
|
$$$
|
Pentamidine inh.
|
NebuPent
|
|||
|
2.0
|
ANTIHYPERTENSIVE THERAPY
|
||||
|
2.1
|
BETA BLOCKERS
|
||||
|
$
|
Atenolol tab.
|
Tenormin
|
|||
|
$
|
Atenolol/HCTZ
|
Tenoretic
|
|||
|
$
|
Metoprolol tartrate tab.
|
Lopressor
|
|||
|
$
|
Propranolol tab.
|
Inderal
|
|||
|
$
|
Propranolol/HCTZ
|
Inderide
|
|||
|
$
|
Labetalol tab.
|
Normodyne
|
|||
|
$
|
Metoprolol/HCTZ
|
Lopressor HCT
|
|||
|
$$
|
Metoprolol succinate SR*
|
Toprol XL
|
|||
|
tab.
|
|||||
|
$$
|
Carvedilol tab.
|
Coreg
|
ST | ||
|
2.2
|
CALCIUM CHANNEL ANTAGONISTS
|
||||
|
$$$
|
Nifedipine SR tab., CR tab
|
Generic only
|
|||
|
Adalat CC
|
NF
|
||||
|
Procardia XL
|
NF
|
||||
|
3.3
|
ANTIHYPERTENSIVES WITH CENTRAL ACTION
|
||||
|
$
|
Methyldopa tab.
|
Aldomet
|
|||
|
3.4
|
VASODILATORS
|
||||
|
$
|
Hydralazine tab.
|
Apresoline
|
|||
|
3.0
|
ANALGESICS, NARCOTICS AND AGENTS IN COMBINATION
|
||||
|
$
|
Oxycodone/APAP cap.
|
||||
|
$
|
Oxycodone/APAP sol.
|
||||
|
$$$
|
Oxycodone/APAP tab.
|
||||
|
4.0
|
ANTIVERTIGO AND ANTIEMETICS
|
||||
|
$
|
Metoclopramide syr., inj.
|
Reglan
|
|||
|
$
|
Trimethobenzamide inj.
|
Tigan
|
|||
|
$
|
Promethazine syr.
|
Phenergan
|
|||
|
$
|
Prochlorperazine tab.
|
Compazine
|
|||
|
$
|
Prochlorperazine inj.
|
Compazine inj.
|
|||
|
$
|
Metoclopramide tab.
|
Reglan
|
|||
|
$
|
Promethazine* tab.
|
Phenergan
|
|||
|
$$
|
Trimethobenzamide cap., supp.Tigan
|
||||
|
$$
|
Promethazine supp.
|
Phenergan
|
|||
|
$$$
|
Prochlorperazine* supp.
|
Compazine
|
|||
|
5.0
|
DEMALOTOGIC AGENTS/TOPICAL THERAPY
|
||||
|
5.1
|
TOPICAL ANTIMYCOTICS
|
||||
|
$
|
Nystain oint.
|
Mycostatin
|
|||
|
$
|
Ketoconazole cr.,
|
Nizoral
|
|||
|
shampoo (2%)
|
|||||
|
5.2
|
TOPICAL ANTIBACTERIALS
|
||||
|
$
|
Gentamicin cr., oint.
|
Garamycin
|
|||
|
6.0
|
GASTROENTEROLOGY
|
||||
|
6.1
|
H2 ANTAGONISTS
|
||||
|
$
|
Ranitidine tab. (300mg)
|
Zantac
|
|||
|
$
|
Cimetidine tab., sol.
|
Tagamet
|
|||
|
$$
|
Ranitidine syr.
|
Zantac
|
|||
|
6.2
|
MISCELLANEOUS ANTIULCER AGENTS
|
||||
|
$
|
Sucralfate tab.
|
Carafate
|
|||
|
$$$
|
Sucralfate susp.
|
Carafate
|
|||
|
6.3
|
MISC. GASTROINTESTINAL AGENTS
|
||||
|
$
|
Hydrocortisone rectal cr.
|
Anusol-HC
|
|||
|
(2.5%)
|
|||||
|
$$
|
Hydrocortisone acetate/
|
Analpram-HC
|
|||
|
Pramoxine rectal cr.
|
|||||
|
$$$
|
Hydrocortisone acetate/
|
Proctofoam HC
|
|||
|
Pramoxine rectal foam
|
||||
|
7.0
|
ENDOCRINOLOGICAL AGENTS
|
|||
|
7.1
|
DIABETES THERAPY
|
|||
|
7.1.1
|
HYPOGLYCEMIC AGENTS
|
|||
|
7.1.1.1
|
INSULIN
|
|||
|
$
|
Human insulin (regular,
|
Humulin
|
P
|
|
|
NPH, 70/30, lens)vial
|
Humulin Pen
|
NF
|
||
|
$$$
|
Insulin lispro vial
|
Humalog
|
P
|
|
|
Humalog Mix
|
NF
|
|||
|
Humalog Pen
|
NF
|
|||
|
$$$
|
Insulin glargine vial
|
Lantus
|
P
|
|
|
Lantus Solostar
|
P
|
|||
|
7.1.1.2 OTHERS
|
||||
|
$
|
Insulin syringe & needles
|
Several
|
||
|
7.2
|
THYROID
|
|||
|
7.2.1
|
THYROID HORMONES
|
|||
|
$
|
Levo-thyroxine tab.
|
Several
|
||
|
$
|
Levo-thyroxine tab.
|
Synthroid
|
P
|
|
|
7.3
|
CORTICOSTEROIDS
|
|||
|
$
|
Dexmethasone sodium
|
OB-GYN
|
||
|
phosphate inj.
|
||||
|
$
|
Prednisone tab.
|
Deltasone
|
||
|
$
|
Dexamethasone tab.
|
Decadron
|
||
|
$
|
Methylprednisolone* tab.
|
Medrol
|
||
|
$
|
Prednisolone tab., syrup
|
Prelone
|
||
|
$
|
Hydrocortisone* tab., sol.
|
Cortef
|
||
|
$
|
Fludrocortisone acetate tab.
|
Florinef
|
||
|
$
|
Dexamethasone elixir,
|
Decadron
|
||
|
syrup, sol.
|
||||
|
$$
|
Betamethasone acetate &
|
Celestone
|
OB-GYN
|
|
|
sodium phosphate inj.
|
Soluspan
|
|||
|
8.0
|
OBSTETRICS AND GYNECOLOGY
|
|||
|
8.1
|
PRENATAL VITAMINS
|
|||
|
$
|
Prenatal vitamins with
|
Generic only
|
OB-GYN
|
|
|
iron and folic acid
|
||||
|
8.2
|
BIOLOGICAL AGENTS
|
|||
Rho Gam- Second dose (post-partum)
(The Department of Health covers the first dose at selected pharmacies - Mothers and Children Program.)
|
8.3
|
ESTROGEN AND PROGESTIN
|
|||
|
8.3.1
|
ESTROGEN
|
|||
|
$
|
Estradiol tab.
|
|||
|
$
|
Estropipate tab.
|
|||
|
$$
|
Conjugated estrogens tab.
|
Premarin
|
P
|
|
|
(0.3mg, 0.625mg, 0.9mg,
|
||||
|
1.25mg, 2.5mg)
|
||||
|
8.3.2
|
ESTROGENS IN COMBINATION
|
|||
|
$$
|
Conjugated estrogen/
|
PremPro
|
P
|
|
|
Medroxyprogesterone tab.
|
PremPro Low Dose
|
NF
|
||
|
(O.625/2.5mg; 0.625/5mg)
|
||||
|
$$$
|
Estradiol/Norethindrone
|
Activella
|
||
|
acetate tab.
|
||||
|
8.3.3
|
PROGESTERONE
|
|||
|
$
|
Medroxyprogesterone
|
Provera
|
||
|
acetate tab.
|
||||
|
8.3.4
|
BIRTH CONTROL PILLS
|
|||
|
Available through the Department of Health
|
PA-AUB
|
|||
|
8.4
|
TOPICAL AGENTS
|
|||
|
8.4.1
|
VAGINAL ESTROGENS
|
|||
|
$$
|
Conjugated estrogen vaginal cr.
|
Premarin
|
P
|
|
|
Vaginal
|
||||
|
$$
|
Estradiol vaginal tab.
|
Vagifem
|
||
|
8.4.2
|
VAGINAL ANTIINFECTIVES
|
|||
|
$$
|
Terconazole* vaginal cr., supp.
|
Terazol - 3,
|
||
|
Terazol - 7
|
||||
|
$$$
|
Clindamycin phosphate
|
Cleocin
|
||
|
vaginal* cr., supp.
|
||||
|
$$$
|
Metronidazole vaginal gel
|
Vandazole
|
||
|
(0.75%)
|
||||
|
8.5
|
MISCELLANEOUS AGENTS
|
|||
|
8.5.1
|
OSTEOPOROSIS
|
|||
|
$$
|
Alendronate tab. (includes
|
Fosamax
|
||
|
weekly dosage)
|
||||
|
$$$
|
Risedronate tab.
|
Actonel
|
P
|
|
|
9.0
|
NUTRIENTS AND VITAMINS
|
|||
|
9.1
|
NUTRIENTS
|
|||
|
$$
|
Iron dextran inj.
|
Infed
|
||
|
9.2
|
VITAMINS
|
|||
|
$
|
Ferrous sulfate tab. (325 mg)
|
Several
|
OTC
|
|
|
$
|
Folic acid tab. (1 mg)
|
|||
|
$
|
Cyanocobalamin inj.
|
Vit.B-12 inj.
|
||
|
$
|
Leucovorin inj.
|
|||
|
$$$$$
|
Leucovorin tab.
|
|||
|
10.0
|
ANTICLOTTING
|
|||
|
$
|
Heparin* inj.
|
|||
|
11.0
|
RESPIRATORY AGENTS
|
|||
|
11.1
|
LOW SEDATION ANTIHISTAMINES
|
|||
|
$
|
Diphenhydramine cap. (50 mg)
|
Benadryl
|
OTC are NF
|
|
|
$
|
Hydroxyzine pamoate cap.
|
Vistaril
|
||
|
$$
|
Hydroxyzine* HCI tab., syr.
|
Atarax
|
||
|
11.2
|
NON-SEDATING ANTIHISTAMINES
|
|||
|
$
|
Loratadine OTC tab., syr.
|
Claritin
|
OTC
|
|
|
11.3
|
ASTHMA AGENTS
|
|||
|
11.3.1 ORAL BETA AGONISTS
|
||||
|
$$
|
Terbutaline tab.
|
Brethine
|
||
|
11.3.2 INHALED BETA AGONISTS
|
||||
|
$
|
Albuterol inh., inh. sol.
|
Generic only
|
||
|
$$
|
Albuterol HFA inh.
|
Ventolin HFA
|
P
|
|
|
LC = 1 pump /30 days
|
||||
|
Proventil HFA
|
NF
|
|||
|
Proair HFA
|
NF
|
|||
|
11.3.3
|
ANTICHOLINERGICS
|
|||
|
$
|
Ipratropium Br inh. sol.
|
Atrovent
|
||
|
$$$
|
Tiotropium inh.
|
Spiriva
|
PA
|
|
|
LC = 1 pump /30 days
|
||||
|
11.3.4 INHALED CORTICOSTEROIDS
|
||||
|
$$$
|
Fluticasone inh., powder
|
Flovent HFA
|
P
|
|
|
LC = 1 pump /30 days
|
||||
|
Flovent Diskus
|
P
|
|||
|
LC = 1 pump /30 days
|
||||
|
$$$$
|
Budesonide inh. Susp.
|
Pulmicort
|
||
|
(respules)
|
||||
|
11.3.5 AGENTS IN COMBINATION
|
||||
|
$$$$
|
Fluticasone/Salmeterol powder
|
Advair Diskus
|
ST, P
|
|
|
LC = 1 pump /30 days
|
||||
|
Advair HFA
|
ST, P
|
|||
|
LC = 1 pump /30 days
|
||||
|
12.0
|
CANCER
|
|||
|
12.1
|
PROGESTINS
|
||
|
$$$
|
Megestrol acetate tab., susp.
|
Megace
|
|
|
$$$$
|
Medroxyprogesterone acetate
|
Depo-Provera
|
|
|
inj. (400 mg)
|
|||
Revised 12/31/2013
Code (for all the pages):
NF - Unlisted
P - Contracted brand name product
Bold - Bioequivalent generic available in all the presentations
Bold* -Some presentations of the drugs are not available in generic
ST - Step Therapy, Clinical protocol for use
OB-GYN - Only in OB-GYN list
ATTACHMENT 5D
PREFERRED DRUG LIST (PDL) FOR MENTAL HEALTH
ASES
HEALTH INSURANCE ADMINISTRATION
Commonwealth of Puerto Rico
December 19, 2012
Dear provider:
I am pleased to present to you the list of Preferred Drugs (PDL) for Mental Health from the Health Plan of the Commonwealth of Puerto Rico (“Mi Salud”), effective since January 1, 2013.
The medications included herein have been evaluated and approved by a Pharmacy and Therapeutics Committee comprised by primary physicians and clinical pharmacists.
This committee meets periodically to evaluate the therapeutical classifications and issue recommendations based on clinical aspects. Therefore, this PDL and the specialized PDLs that are included in this guide, may suffer changes, which are notified by means of Normative Letters, in conformity to how they arise.
I exhort you to conserve this Guide for your reference as long as it is necessary. The same is also available in our electronic page www.asespr.org.
Cordially,
(signed)
Frank R. Díaz Ginés, MHSA
Executive Director
|
PO Box 195661, San Juan, Puerto Rico 00919-5661
|
Tel. (787) 474-3300
|
Fax (787) 474-3346
|
GENERAL ASPECTS OF THE PHARMACY COVERAGE
FROM THE HEALTH PLAN OF THE GOVERNMENT OF PUERTO RICO
|
1.
|
Listing of Drugs
|
The Administration of Health Insurance (ASES) is the agency responsible for establishing and reviewing the Preferred Drug List (PDL, on the basis of its initials in English) for Physical Health and Mental Health of “Mi Salud” (My Health). For this purpose, there has been established a Pharmacy and Therapeutics Committee for Physical Health, and another one for Mental Health, both comprised by different health professionals. These committees meet periodically to evaluate the different therapeutical classifications and issue their recommendations to ASES about the drug lists, based on scientific evidence and clinical aspects.
The Preferred Drug List (PDL) shall serve as a guide in the supplying of drugs under the pharmacy coverage. The PDL has the purpose of improving, putting up to date and attaining the effective cost use of drugs within the coverage of Mi Salud.
In addition, ASES maintains the Uniform Core List (FMU), which is utilized as the official listing from which the PDL is derived. The drugs in this list which do not appear in the PDL may be utilized the insurance companies, mental health organizations and service providers only as exceptions in the cases in which the drugs in the PDL were not the most clinically effective for the patient in particular.
In like manner, a List of Specialized Drugs (contracted) was created. In the situation where one provider of medical services prescribes a specialized drug from this list, it should be evaluated by means of the exception process by the MCO, MBHO, TPA or the organization contracted by ASES.
In exceptional cases drugs outside of the aforementioned lists may be utilized by means of an exception procedure.
|
2.
|
Exception Process
|
In the case of patients who need a drug that does not appear in the PDL or in the FMU, the insurance companies, mental health organizations and health service providers must utilize the process described herein for the approval of the drugs. This procedure shall consider the particular merits of each case, which may include:
|
1.
|
Contraindication to the drug(s) that appear in the PDL.
|
|
2.
|
History record of adverse reaction to the drug(s) that appear in the PDL.
|
|
3.
|
Therapeutical failure with regard to all the alternatives available in the PDL.
|
|
4.
|
Non-existence of a therapeutical alternative in the PDL.
|
This exception process requires the official documentation from the service provider regarding the clinical reasons that justify the use of drugs outside the PDL.
|
3.
|
Other aspects of the pharmacy coverage
|
|
A.
|
The pharmacy coverage of Mi Salud establishes as mandatory the use of bioequivalent products, as long as it is not contraindicated and it is so clinically justified by the provider.
|
|
B.
|
The insurance companies, mental health organizations and health service providers shall process their pharmacy claims through the Pharmacy Benefit Managers (PBMs) contracted by the Administration.
|
|
C.
|
ASES has an active process to continuously review the drugs that it is required to include in the pharmacy coverage to the insurance companies or TPA, mental health organizations and health service providers. In addition, it shall evaluate any new drug for inclusion in the same or remove drugs from said listing. Due to the dynamic nature of this process, ASES may require the inclusion or exclusion of drugs pursuant to changes or advances in standards of practice within an illness or area of treatment.
|
|
D.
|
No entity contracted by ASES or the entities contracted by these ( Medical groups, IPAs, independent providers, specialists, etc.) may be ruled by a listing different from the PDL and the Core List, nor can they create an internal drug list that is different from the ones established by ASES. Neither can they, in any way, limit in a manner contrary to what is established in the contract between ASES and the insurance companies and in this coverage, the drugs that are included in said lists. The insurance companies shall be responsible for keeping tabs in their providers and employees with regard to the compliance with these provisions.
|
|
E.
|
The maximum of dispatch for severe conditions shall be to cover a thirty (30) day therapy. When medically necessary, additional prescriptions shall be covered.
|
| F. | The maximum of dispatch for chronic conditions (maintenance drugs) shall be to cover a thirty (30) day therapy, except at the beginning of the therapy when, on the basis of medical criterion, a minimum of fifteen (15) days may be prescribed for the purpose of reevaluating compliance and tolerance. On the basis of a recommendation on the part of the physician, the dispatch of each prescription may be refilled up to five (5) times (original prescription plus five (5) refills). The drugs that require pre-authorization shall be in effect for six months unless there are contraindications or secondary effects. On or before the ninety (90) days after having prescribed said maintenance drug, the physician must reevaluate the pharmacotherapy for the purpose of compliance, tolerance and classification. Changes in the dosage will not require pre-authorization. Changes in the drugs utilize may require pre-authorization. |
| G. | The indications in the prescriptions for chronic use drugs, in favor of Children with Special Health Needs, must indicate clearly that they are covering a thirty (30) day therapy and that they may be refilled up to five (5) times (original prescription plus five (5) refills), according to medical criterion. When it is medically necessary, additional prescriptions shall be refilled. |
| H. | Coordination with the Department of Health is required for the supplying of birth control methods for family planning. The contraceptives for the treatment of menstrual dysfunction and for other menstrual conditions shall be covered through your primary physician from Physical Health. |
| I. | The use of bioequivalents approved by the Food and Drug Administration (FDA), classified as AB, is required, as well as authorized by the local regulations as long as it is not contraindicated and it is so consigned by the provider in the medical prescription. |
| J. | The lack of existence in the inventory of bioequivalent drugs does not exempt the dispatch of the prescribed drug nor does it imply any additional payment whatsoever on the part of the beneficiary. As a general rule, bioequivalent drugs should be dispatched as long as there exists a bioequivalent for the drug of the corresponding brand name unless, in spite of the existence of a particular bioequivalent, ASES decides to cover the brand name drug or to cover both. |
| K. | All the prescriptions must be dispatched by a pharmacy contracted by the PBM, duly authorized under the laws of the Commonwealth of Puerto Rico and freely selected by the beneficiary. |
| L. | The right to free selection requires the availability of a determinate number of pharmacies in every municipality to be able to so exercise it. The PBM is the entity in charge of contracting the network of pharmacies, pursuant to the terms specified by ASES. |
| M. | The prescribed drugs must be delivered concurrently on the date and time when the beneficiary receives the prescription and requires the dispatch of the drug. |
For any doubt regarding the pharmacy coverage you may get in touch with the insurance company contracted by ASES in your service region.
HEATLH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST (PDL)
MENTAL HEALTH 2013-2014
TABLE OF CONTENTS
|
PART I - DESIGN OF THE PREFERRED DRUG LIST FOR MENTAL HEALTH
|
iii
|
|
| PRESENTATION OF THE PREFERRED DRUG LIST (“PDL”) | ||
| FOR MENTAL HEALTH AND REFERENCE GUIDES |
iii
|
|
| RELATIVE COST INDICATOR |
iii
|
|
| GENERIC DRUGS |
iv
|
|
| REFERENCE GUIDES |
iv
|
|
| LEGEND FOR SYMBOLS AND ABBREVIATIONS | ||
| IN THE FORM | ||
|
PART II – DRUGS BY THERAPEUTIC CLASSIFICATION
|
1
|
|
|
PSYCHOTROPIC AGENTS
|
1
|
|
|
1.0
|
ANTIDEPRESSANTS
|
1
|
|
1.1
|
TRICYCLICS
|
1
|
|
1.2
|
SEROTONIN REUPTAKE INHIBITORS
|
1
|
|
1.3
|
SEROTONIN REUPTAKE INHIBITORS & NOREPINEPHRINE
|
2
|
|
1.4
|
MISCELLANEOUS ANTIDEPRESSANTS
|
2
|
|
2.0
|
ANTIPSYCHOTICS
|
2
|
|
2.1
|
PHENOTHIAZINES
|
2
|
|
2.2
|
BUTYROPHENONES
|
3
|
|
2.3
|
MISCELLANEOUS ANTIPSYCHOTICS
|
3
|
|
2.4
|
NON TYPICAL ANTIPSYCHOTICS
|
3
|
|
3.0
|
MOOD STABILIZERS
|
4
|
|
4.1
|
ANXIOLYTICS / HYPNOTICS
|
4
|
|
4.2
|
BENZODIAZEPINES
|
4
|
|
4.3
|
SEDATIVE ANTIHISTAMINES
|
5
|
|
4.4
|
HYPNOTICS
|
5
|
|
5.0
|
PSYCHO-STIMULANTS
|
5
|
|
5.1
|
SYMPATHOMIMETICS AMINES
|
5
|
|
5.2
|
SELECTIVE REUPTAKE INHIBITORS OF NOREPINEPHRINE
|
6
|
|
6.0
|
ANTICOLINERGICS
|
6
|
|
7.0
|
DETOX TREATMENT
|
6
|
|
8.0
|
ADICTION TREATMENT DRUGS
|
7
|
|
PART III - APPENDIX I - PRODUCTS WITH A DISPATCH QUANTITY LIMIT
|
8
|
|
|
PART IV- INDEX.
|
9
|
|
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST (PDL)
MENTAL HEALTH 2013-2014
PART I - DESIGN OF THE PREFERRED DRUG LIST FOR MENTAL HEALTH
PRESENTATION OF THE PREFERRED DRUG LIST FOR MENTAL HEALTH (“PDL”) AND REFERENCE GUIDES
In the following example, we illustrate the information that is provided for the drugs included in the “PDL” for Mental Health.
|
Relative Cost Indicator
|
Generic Name (in bold if the
drug is available as a generic)
|
Brand
Name
|
Reference Guides
|
|
Dollar Sign ($)
|
Trazodone tab
|
Desyrel
|
PA
|
For each drug included in the Preferred Drug List (“PDL”) for Mental Health, there is a Cost Indicator (Relative Cost), the Generic Name, the Brand Name and Reference Guides, as applicable. In the cases in which the generic drug is available, it will appear in bold lettering. Those generic drugs that have an asterisk (*) indicate that not all the presentations of the product have a generic available. As long as the generic bioequivalent of a drug exists, it will be dispatched as long as it is more cost effective. The brand name is only mentioned for reference.
We encourage you to use the “PDL” for Mental Health as a reference when you prescribe drugs to the beneficiaries of the Health Plan of the Commonwealth of Puerto Rico.
RELATIVE COST INDICATOR
The relative cost indicator is included in the “PDL” of Mental Health to offer an estimated value of the cost of a drug therapy including any discount for utilization and comparing the specific product with the other available alternatives in that class, or to treat the disease or specific condition. In the majority of cases, it compares the cost for therapy for fifteen (15) or thirty (30) days depending if the drug is for acute use or maintenance, respectively.
The comparison of costs and assignment of dollar signs ($) is based on all the products included within a therapeutics class, (for example, tricyclic antidepressants, serotonin reuptake inhibitors). The dollar signs on the side of a drug identify its relative cost and must be interpreted in the following manner:
|
$
|
Less Costly
|
|
$$
|
|
|
$$$
|
|
|
$$$$
|
|
|
$$$$$
|
|
|
$$$$$!
|
|
|
$$$$$!!
|
More Costly
|
When several drugs within the same therapeutics class have the same number of dollar signs, the drug that is mentioned first must be considered as the least costly.
When there is an alternative of a number of adequate preparations to treat a particular disease or condition, the relative cost indicator can be used to make a selection based in the cost.
The designation of the relative costs is in force at the time of the publication of this edition of the Mental Health “PDL". The cost of the drugs changes continuously.
GENERIC DRUGS
The generics / bioequivalent drugs are identified in bold. Some generics / bioequivalent have a Maximum Allowable Cost or MAC for their payment. This price typically covers the cost of acquisition of the generic products, but not of the brand product. The selection of products to be included in the MAC List are those that are commonly prescribed and that have been approved by the Food and Drug Administration (FDA) to be marketed.
REFERENCE GUIDES
The drugs that are in the Mental Health “PDL” are those preferred drugs in the coverage of the Health Plan of the Commonwealth of Puerto Rico. These drugs are selected based on their safety, effectiveness, high quality, existence of bioequivalents and cost. We suggest to physicians to prescribe and to the pharmacists to dispatch only those drugs that are in the “PDL”. All the drugs included in this document are medically covered unless they are designated as Non-Form (NF).
LEGEND FOR THE SYMBOLS AND ABBREVIATIONS IN THE FORM
|
$ to
$$$$$!!
|
Represents the relative cost of the drug. The fewer number of dollar signs ($), the lesser the cost of the drug. The higher number of dollar signs, the higher the cost.
|
|
PA
|
Requires the physician to obtain a Pre-authorization.
|
|
NF
|
No-Form- It is not covered.
|
|
P
|
Contracted Brand Name Product / “Rebates”
|
|
Bold
|
Identifies that the drug has a generics / bioequivalent available in all of its presentations
|
|
(Bold) *
|
Identifies those drugs for which not all presentations or forms are available in generics; for example, tablets, liquids, injections, etc.
|
|
LC
|
Identifies those drugs for which there is a limit in the quantity that the pharmacy can dispatch.
|
|
AL
|
Identifies those drugs for which there is some limit in the age for the dispatch of the drug.
|
|
ST
|
Step Therapy, Clinical Protocol for its use
|
|
Cap.
|
Capsule
|
|
Tab.
|
Tablet
|
|
Susp.
|
Suspension
|
|
ER, SR, CR
|
Prolonged Action (extended release, sustained release, controlled release)
|
|
SL
|
Sublingual
|
|
Cr.
|
Cream
|
|
Oint.
|
Ointment
|
|
Sol.
|
Solution
|
|
Syr.
|
Syrup
|
|
Lot.
|
Lotion
|
|
Ophth.
|
Ophthalmic
|
|
Inh.
|
Inhaler
|
|
SNC
|
Central Nervous System
|
|
Liq.
|
Liquid
|
|
Conc.
|
Concentrated
|
|
Inj.
|
Injectable
|
|
Supp.
|
Suppository
|
|
Disp. Tab.
|
Dispersible tab.
|
HEALTH PLAN OF THE COMMONWEALTH OF PUERTO RICO
PREFERRED DRUG LIST (PDL)
MENTAL HEALTH 2013-2014
PART II – DRUGS BY THERAPEUTIC CLASSIFICATION
|
PSYCHOTROPIC AGENTS
|
|||
|
1.0
|
ANTIDEPRESSANTS
|
||
|
1.1
|
TRICYCLICS
|
||
|
$
|
Amitriptyline tab.
|
Elavil
|
|
|
$
|
Nortriptyline cap., sol.
|
Pamelor, Aventyl
|
|
|
$
|
Doxepin cap., conc.
|
Sinequan
|
|
|
$
|
Clomipramine cap.
|
Anafranil
|
|
|
$
|
Imipramine HCL tab.
|
Tofranil
|
|
|
$
|
Desipramine tab.
|
Norpramin
|
|
| 1.2 | SEROTONIN REUPTAKE INHIBITORS (SSRI) | ||
|
$
|
Fluoxetine cap. (10mg, 20mg)
|
Prozac
|
|
|
$
|
Citalopram tab.
|
Celexa
|
|
|
$
|
Sertraline tab.
|
Zoloft
|
|
|
$
|
Escitalopram tab.
|
Lexapro
|
|
|
$
|
Paroxetine tab. (20mg, 30mg, 40mg)
|
Paxil
|
|
|
$$
|
Escitalopram sol.
|
Lexapro
|
|
| 1.3 | SEROTONINE REUPTAKE INHIBITORS & NOREPINEPHRINE (SNRI) | |||
|
$$
|
Venlafaxine tab.
|
Effexor
|
||
|
$$
|
Venlafaxine XR 24hr cap.
|
Effexor XR
|
||
|
$$$
|
Duloxetine cap.
|
Cymbalta
|
PA, P
|
|
|
1.4
|
MISCELLANEOUS ANTIDEPRESSANTS
|
||
|
|
|
|
|
|
|
$
|
Mirtazapine tab.
|
Remeron
|
|
|
$
|
Bupropion tab.
|
Wellbutrin
|
|
|
$
|
Trazodone tab.
(50mg, 100mg, 150mg)
|
Desyrel
|
|
$$$
|
Bupropion SR 12hr tab.
|
Wellbutrin SR
|
|
|
2.1
|
ANTIPSYCHOTICS
|
||
|
|
|
|
|
|
2.2
|
PHENOTHIAZINES
|
||
|
|
|
|
|
|
|
$
|
Fluphenazine decanoate inj.
|
Prolixin Decanoate
|
|
|
$
|
Fluphenazine* tab., elixir, conc.
|
Prolixin
|
|
|
$
|
Chlorpromazine* tab., CR cap., syrup, conc., supp.
|
Thorazine
|
|
$
|
Thiothixene* cap., conc.
|
Navane
|
|
|
$
|
Thioridazine* tab., susp., conc.
|
Mellaril
|
|
|
|
|||
| 2.2 | BUTYROPHENONES | ||
|
$$
|
Haloperidol* tab., conc.
|
Haldol
|
|
|
$$$
|
Haloperidol decanoate inj.
|
Haldol Decanoate
|
|
| 2.3 | MISCELLANEOUS ANTIPSYCHOTICS | ||
|
$$$
|
Loxapine cap.
|
Loxitane
|
|
| 2.4 | NON TYPICAL ANTIPSYCHOTICS | ||
|
$
|
Clozapine tab.
|
Clozaril PA
|
|
|
$
|
Risperidone tab.
|
Risperdal
|
|
|
$
|
Quetiapine tab.
|
Seroquel PA
|
|
| (25mg, 50mg) |
LC= 10 days
|
||
|
$
|
Olanzapine tab.
|
Zyprexa PA
|
|
|
$
|
Olanzapine tab.
|
Zyprexa PA
|
|
|
$$
|
Quetiapine tab. (except 25mg, 50mg)
|
Seroquel PA
|
|
|
$$$$
|
Ziprasidone cap.
|
Geodon PA
|
|
|
$$$$
|
Quetiapine ER tab.
|
Seroquel XR PA, P
|
|
|
$$$$
|
Quetiapine ER tab. 50mg
|
Seroquel XR PA, P
|
|
|
LC= 3 days
|
|||
|
$$$$$
|
Aripiprazole tab.
|
Abilify PA, P
|
|
|
3.0
|
MOOD STABILIZERS
|
||
|
|
|
|
|
|
|
$
|
Lithium carbonate* cap., tab.,
CR tab. (300mg, 450mg)
|
Eskalith,
Lithotabs,
Lithobid,
Lithonate,
Eskalith CR
|
|
|
$
|
Valproate sodium syr.
|
Depakene
|
|
|
$$
|
Lithium citrate syrup
|
Lithium
|
|
|
$$
|
Valproic acid cap.
|
Depakene
|
|
|
$$
|
Divalproex sodium* EC tab., sprinkle cap.
|
Depakote
|
|
$$
|
Lamotrigine chew disp. tab.
|
Lamictal CD
|
|
|
$$
|
Lamotrigine tab.
|
Lamictal
|
|
|
$$
|
Divalproex sodium extended release tab. |
Depakote ER
|
|
| 4.0 | ANXIOLYTICS / HYPNOTICS | ||
| 4.1 | BENZODIAZEPINES | ||
|
$
|
Clonazepam tab.
|
Klonopin
|
|
|
$
|
Flurazepam cap.
|
Dalmane
|
|
|
$
|
Temazepam* cap.
|
Restoril
|
|
|
$
|
Chlordiazepoxide cap.
|
Librium
|
|
|
$
|
Estazolam tab.
|
Prosom
|
|
|
$
|
Clorazepate tab.
|
Tranxene
|
|
|
$
|
Lorazepam* tab., conc.
|
Ativan
|
|
|
$
|
Alprazolam* tab., conc. (except 2mg tab.)
|
Xanax
|
|
|
$
|
Diazepam* tab., sol., conc.
|
Valium
|
|
|
$$
|
Oxazepam cap.
|
Serax | |
|
4.2
|
SEDATIVE ANTIHISTAMINES
|
||
|
$
|
Hydroxyzine pamoate cap.
|
Vistaril
|
|
|
$
|
Diphenhydramine cap. (50mg)
|
Benadryl
|
|
|
$$$
|
Hydroxyzine pamoate susp.
|
Vistaril
|
|
|
4.3
|
HYPNOTICS
|
|||
|
$
|
Zolpidem cap.
|
Ambien
|
||
|
5.0
|
PSYCHO-STIMULANTS
|
|||
|
5.1
|
SYMPATHOMIMETIC AMINES
|
|||
|
$
|
Dextroamphetamine tab.
|
Dexedrine
|
AL (4-18)
|
|
|
$
|
Methylphenidate tab.
|
Ritalin
|
AL (6-18)
|
|
|
$$
|
Methylphenidate CR tab.
|
Metadate ER Ritalin SR
|
AL (6-18)
AL (6-18)
|
|
|
$$
|
Amphetamine/ Dextroamphetamine tab.
|
Adderall
|
AL (4-18)
|
|
|
$$
|
Dextroamphetamine SR 24hr cap.
|
Dexedrine
|
AL (4-18)
|
|
|
$$
|
Dexmethylphenidate regular release tab.
|
Focalin
|
AL (6-18)
|
|
|
$$$
|
Dexmethylphenydate cap.
|
Focalin XR
|
ST, AL (6-18)
|
|
|
$$$
|
Methylphenidate CR cap. (Modified-release)
|
Metadate CD
|
ST, AL (6-18)
|
|
|
5.2
|
SELECTIVE REUPTAKE INHIBITOR OF NOREPINEPHRINE
|
|||
|
$$$$
|
Atomoxetine cap.
|
Strattera
|
PA, P, AL (6-18)
|
|
|
6.0
|
ANTICOLINERGICS
|
|||
|
$
|
Benztropine tab.
|
Cogentin
|
||
|
7.0
|
DETOX TREATMENT
|
|||
|
$
|
Thiamine tab. (100mg)
|
LC= 7 days
|
||
|
$
|
Ibuprofen tab. (800mg)
|
Motrin
|
LC= 7 days
|
|
|
$
|
Folic Acid tab. (1mg)
|
LC= 7 days
|
||
|
$
|
Loperamide cap. (2mg)
|
Imodium
|
LC= 7 days
|
|
|
$
|
Clonidine tab. (0.1 mg)
|
Catapress
|
LC= 7 days
|
|
|
8.1
|
DRUGS FOR THE TREATMENT RELATED TO THE ABUSE AND DEPENDENCE OF SUBSTANCES
|
This drug will be covered through the Administración de Servicios de Salud Mental y Contra la Adicción (ASSMCA - Mental Health Services and Against Addiction Administration), according to its availability.
|
$
|
Methadone tab., disp. tab., sol., conc.
|
ASSMCA
|
||
|
$$
|
Buprenorphine sl. tab.
|
Subutex
|
PA
|
|
|
$$$
|
Buprenorphine/ naloxone sl. film
|
Suboxone
|
PA, P
|
|
Bold
|
Generics Bioequivalent alternative available
|
|
Bold*
|
Some presentations of the drug are not available in generics
|
|
PA
|
Requires Pre-Authorization
|
|
P
|
Brand Name Drug Contracted
|
|
LC
|
Limit in the Quantity to dispatch
|
|
AL
|
Limit in Age
|
|
ST
|
Step therapy, Clinical Protocol for its use
|
Revised 12/21/2012
PRODUCTS WITH LIMITS IN THE QUANTITY TO DISPATCH
|
1.
|
Antipsychotics
|
|
Product
|
Generic Name
|
Limit
|
|
Seroquel 25mg, Seroquel 50mg
Seroquel XR 50mg
|
Quetiapine tab.
Quetiapine ER tab.
|
Maximum 10 days
Maximum 3 days
|
| 2. | Hypnotics |
|
Product
|
Generic Name
|
Limit
|
|
Ambien
|
Zolpidem tab.
|
Maximum 30 days
|
|
2.
|
Detox Treatment
|
|
Product
|
Generic Name
|
Limit
|
|
Thiamine tab. (100mg)
|
Maximum 7 days of therapy
|
|
|
Motrin
|
Ibuprofen tab. (800mg)
|
Maximum 7 days of therapy
|
|
Folic Acid tab. (1mg)
|
Maximum 7 days of therapy
|
|
|
Imodium
|
Loperamide cap. (2mg)
|
Maximum 7 days of therapy
|
|
Catapress
|
Clonidine tab. (0.1mg)
|
Maximum 7 days of therapy
|
|
A
|
D
|
|||
|
Abilify
|
3
|
Dalmane
|
4
|
|
|
Adderall
|
6
|
Decanoate
|
2
|
|
|
Alprazolam
|
5
|
Depakene
|
4
|
|
|
Amitriptyline
|
1
|
Depakote
|
4
|
|
|
Amphetamine/Dextroamphetamine
|
6
|
Depakote ER
|
4
|
|
|
Anafranil
|
1
|
Desipramine
|
1
|
|
|
Aripiprazole
|
3
|
Desyrel
|
2
|
|
|
Ativan
|
5
|
Dexedrine
|
5,6
|
|
|
Atomoxetine
|
6
|
Dexmethylphenidate.
|
6
|
|
|
Dexmethylphenidate regular release
|
6
|
|||
|
Aventyl
|
1
|
Dextroamphetamine
|
5
|
|
|
Dextroamphetamine SR
|
6
|
|||
|
B
|
Diazepam
|
5
|
||
|
Benadryl
|
5
|
Diphenhydramine
|
5
|
|
|
Benztropine
|
6
|
Divalproex sodium
|
4
|
|
|
Buprenorphine
|
7
|
Divalproex sodium EC
|
4
|
|
|
Buprenorphine/naloxone
|
7
|
Doxepin
|
1
|
|
|
Bupropion
|
2
|
Duloxetine
|
2
|
|
|
Bupropion SR
|
2
|
|||
|
E
|
||||
|
C
|
Elavil
|
1
|
||
|
Catapress
|
6
|
Effexor
|
2
|
|
|
Celexa
|
1
|
Effexor XR
|
2
|
|
|
Chlordiazepoxide
|
4
|
Escitalopram
|
1
|
|
|
Chlorpromazine
|
2
|
Eskalith
|
4
|
|
|
Citalopram
|
1
|
Eskalith CR
|
4
|
|
|
Clomipramine
|
1
|
Estazolam
|
4
|
|
|
Clonazepam
|
4
|
|||
|
Clonidine
|
6
|
F
|
||
|
Clorazepate
|
5
|
Fluoxetine
|
1
|
|
|
Clozapine
|
3
|
Fluphenazine
|
2
|
|
|
Clozaril
|
3
|
Fluphenazine decanoate
|
2
|
|
|
Cogentin
|
6
|
Flurazepam
|
4
|
|
|
Concerta
|
5
|
Focalin
|
6
|
|
|
Cymbalta
|
2
|
INDEX
|
Focalin XR
|
6
|
Loxitane
|
3
|
|
|
Folic Acid
|
6
|
|||
|
M
|
||||
|
G
|
Mellaril
|
2
|
||
|
Geodon
|
3
|
Metadate CD
|
6
|
|
|
Metadate ER
|
5
|
|||
|
H
|
Methadone
|
7
|
||
|
Haldol
|
3
|
Methylphenidate
|
5
|
|
|
Haldol decanoate
|
3
|
Methyphenidate CR
|
5
|
|
|
Haloperidol
|
3
|
Methyphenidate CR cap Modified-Release)
|
5,6
|
|
|
Haloperidol decanoate
|
3
|
|||
|
Hydroxyzine pamoate
|
5
|
|||
|
Mirtazapine
|
2
|
|||
|
I
|
Motrin
|
6
|
||
|
Ibuprofen
|
6
|
|||
|
N
|
||||
|
Imipramine
|
1
|
Navane
|
2
|
|
|
Imodium
|
6
|
Nortriptyline
|
1
|
|
|
Norpramin
|
1
|
|||
|
K
|
||||
|
Klonopin
|
4
|
O
|
||
|
Olanzapine
|
3
|
|||
| L |
Oxazepam
|
5 | ||
|
Lamictal
|
4
|
|||
|
Lamictal CD
|
4
|
P
|
||
|
Lamotrigine
|
4
|
Pamelor
|
1
|
|
|
Lamotrigine chew disp
|
4
|
Paroxetine
|
1
|
|
|
Lexapro
|
1
|
Paxil
|
1
|
|
|
Librium
|
4
|
Perphenazine
|
3
|
|
|
Lithium
|
4
|
Prolixin
|
2
|
|
|
Lithium carbonate
|
4
|
Prolixin decanoate
|
2
|
|
|
Lithium citrate
|
4
|
Prosom
|
4
|
|
|
Lithobid
|
4
|
Prozac
|
1
|
|
|
Lithonate
|
4
|
|||
|
Lithotabs
|
4
|
Q
|
||
|
Loperamide
|
6
|
Quetiapine
|
3
|
|
|
Lorazepam
|
5
|
Quetiapine ER
|
3
|
|
|
Loxapine
|
3
|
|
R
|
||||
|
Remeron
|
2
|
Venlafaxine
|
2
|
|
|
Restoril
|
4
|
Venlafaxine XR
|
2
|
|
|
Risperidone
|
3
|
Vistaril
|
5
|
|
|
Risperdal
|
3
|
|||
|
Ritalin
|
5
|
W
|
||
|
Ritalin SR
|
5
|
Wellbutrin
|
2
|
|
|
Wellbutrin SR
|
2
|
|||
|
S
|
||||
|
Serax
|
5
|
X
|
||
|
Seroquel
|
3
|
Xanax
|
5
|
|
|
Seroquel XR
|
3
|
|||
|
Sertraline
|
1
|
Z
|
||
|
Sertraline conc
|
2
|
Ziprasidone
|
3
|
|
|
Sinequan
|
1
|
Zoloft
|
2
|
|
|
Stelazine
|
3
|
Zolpidem
|
5
|
|
|
Strattera
|
6
|
Zyprexa
|
3
|
|
|
Subutex
|
7
|
|||
|
Suboxone
|
7
|
|||
|
T
|
||||
|
Temazepam
|
4
|
|||
|
Thiamine
|
6
|
|||
|
Thiothixene
|
2
|
|||
|
Thioridazine
|
2
|
|||
|
Thorazine
|
2
|
|||
|
Tofranil
|
1
|
|||
|
Tranxene
|
5
|
|||
|
Trazodone
|
2
|
|||
|
Trifluoperazine
|
3
|
|||
|
Trilafon
|
3
|
|||
|
V
|
||||
|
Valium
|
5
|
|||
|
Valproate sodium
|
4
|
|||
|
Valproic acid
|
4
|
ATTACHMENT 6
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
Retail Pharmacy Reimbursement Level
Effective date: November 1, 2011
|
|
Pharmacy Type
|
Ingredient Cost
(AWP Discounts)
|
Dispensive Fee
|
||||||
|
Independent:
|
||||||||
|
Brand
|
11
|
%
|
$
|
2.50
|
||||
|
Bioequivalent Generics
|
ASES' MAC List
|
$
|
2.50
|
|||||
|
Non *MAC Generics
|
11
|
%
|
$
|
2.50
|
||||
|
Local Pharmacy Chains:
|
||||||||
|
Brand
|
11
|
%
|
$
|
2.50
|
||||
|
Bioequivalent Generics
|
ASES' MAC List
|
$
|
2.50
|
|||||
|
Non MAC Generics
|
11
|
%
|
$
|
2.50
|
||||
|
National Pharmacy Chain:
|
||||||||
|
Brand
|
15
|
%
|
$
|
1.75
|
||||
|
Generics
|
ASES' MAC List
|
$
|
2.50
|
|||||
|
Non-MAC Generics
|
15
|
%
|
$
|
2.00
|
||||
|
*Walgreens
|
Not Contracted
|
|||||||
|
Diagnostic and Treatment Centers
|
||||||||
|
Brand
|
12
|
%
|
$
|
2.50
|
||||
|
Generics
|
ASES' MAC List
|
$
|
2.50
|
|||||
|
Non-MAC Generics
|
12
|
%
|
$
|
2.50
|
||||
|
*MAC=Maximum Allowable Cost
|
Rev./06.2013
|
|||||||
Uniform and Mandatory Protocol for the Conditions Included in the Special Coverage
Inception:
Every physician specialist or primary care physician that has evaluated the patient can perform the registration request as long as he or she has all the required documentation for the request. The insurance company is obliged to perform a determination of approval or denial of the registration and will inform this decision through a letter to the insured and the physician who requests the registration. In the event that the physician who requests the registration is not the insured’s primary care physician, the insurance company will send a copy of the determination to the primary care physician. The insurance company will determine the result of the request for a special condition in a period of 72 hours, after receiving complete documentation as required in this protocol for each condition.
Once a Provider supplies all the required information for the Contractor to process a registration and the Contractor processes such information, Special Coverage shall take effect retroactively as of the date the Provider reaches a diagnosis, including documentation of test results, for any condition included in Special Coverage. In case Information is submitted to the Contractor after the diagnosis was reached, coverage can be made retroactive up to sixty (60) Calendar Days before the date on which Provider submitted the registration request. (Contract Section 7.7.5)
Reactivation:
Every insured that has lost eligibility to the PSG for a period greater than a year will be required to have a new certification from the primary care physician that shows evidence of the treatment plan that he/she receives at the present to be reactivated in the special coverage. Every insured that loses his/her eligibility for a period less than 12 months will be registered again without the need of additional documents or certifications, unless another limit is established for that specific registration.
*Risk Assignment: The risk distribution of the special coverage between the insurance company and primary care physician groups is defined in the following table. It can be modified by request of the insurance company subject to previous revision and approval from ASES.
1
|
Special Condition
|
Definitive Diagnostic Criteria for Inclusion in Coverage
|
Effectiveness and Duration of Special Coverage
|
Services Included in the Special Coverage
|
Risk Assignment *
|
|
Aplastic Anemia
|
I- Hematological Evaluation:
a- Absolute Count of Neutrophil <500/mm3
b- Platelets <20,000/mm3
c- Reticulocyte <1%
d- Bone Marrow Aspiration and/or biopsy Results
e- Diagnostic Certification by an
Hematologist /Oncologist
|
Effectiveness= From the date of the diagnostic certification by the Hematologist / Oncologist or the date when the biopsy was performed if the reading establishes a definitive diagnostic.
Duration = The Special Coverage will begin from the date in which the definitive diagnostic is established. The Special Coverage will be in place as long as the insured is eligible in the PSG.
|
1. All medical services in hospitals, emergency rooms or by a medical specialist rendered with a main diagnostic of Aplastic Anemia.
2- All medical services rendered or ordered by the Hematologist / Oncologist.
3- Prescribed medications by the oncologist / hematologist and specific to treat the condition.
|
Insurance Company –
Services included for the condition in the special coverage or any other service contemplated under the special coverage regardless of this diagnostic category.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
|
Rheumatoid Arthritis
|
1-Diagnostic Certification by the rheumatologist with evidence of at least 4 of the 7 criteria established by the American College of Rheumatology;
a- Periarticular Morning numbness (more than an hour of duration) for more than 6 weeks
b- Swelling of soft tissues in three or more articulations, for more than 6 weeks.
c- Swelling of articulations in proximal phalanges or metacarpo-falangeal, for more than 6 weeks.
d- Symmetrical Arthritis for at least 6 weeks.
e- Presence of subcutaneous nodules
f- Positive Test of Rheumatoid Factor
(The insurance company will provide a sheet with the criteria and treatment plan to be filled out by the specialist.)
2-Laboratory Tests; ESR, ANA Test, CRP, RA Factor.
3- Evidence of treatment with the DMARD medication
|
Effectiveness = From the date of certification of a diagnostic by a rheumatologist. The evidence of the following laboratory tests: ESR, CRP, ANA test and pertinent x-rays confirming the diagnostic, will be required with the certification of a rheumatologist.
Duration = As long as the insured maintains the eligibility in the PSG.
|
1. All hospital services, emergency room or by a medical specialist rendered with a main diagnostic of Rheumatoid Arthritis.
2. All medical services rendered or ordered by the rheumatologist.
3. Medications prescribed by the rheumatologist including the DMARD.
|
Insurance Company –
Services included for the condition in the special coverage or any other service contemplated under the special coverage regardless of this diagnostic category.
GMP/PCP – Receives monthly capitation corresponding to the insured
|
2
|
Autism
|
1-Diagnostic Certification of the condition by a neurologist and psychiatrist. Both certifications will be required to perform the registration.
2-Evidence of the M-CHAT and “Ages and Stages” Tests.
|
Effectiveness = From the date of the diagnostic certification of the neurologist and the psychiatrist. The earliest certification date will be the effective date.
The special coverage will be in place as long as the insured maintains his/her eligibility to the PSG in place until he/she reaches 21years of age. After 21 years of age there will be a requirement for a certification by a neurologist or psychiatrist that establishes the need for management and handling for the condition as an adult.
|
1. Medical services rendered or ordered by the psychiatrist and psychologist will not require the referral of a primary care physician, as well as the medications prescribed by the psychiatrist.
Medical services rendered by the neurologist or any other specialist recommended for the management of his condition will not require the referral of the primary care physician.
Any medication prescribed by the neurologist or any other specialist and specific to treat the condition is included.
|
Insurance Company -
All the medical services provided by neurologists and psychiatrists including medications prescribed by them.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
|
Cancer
|
1-Diagnostic Certification with stage, by Hematologist / oncologist or medical specialist in charge of the management of the condition, with treatment plan with estimated dates of beginning and end.
The insurance company will provide a specific document for the specialist to document the Certification and Request for Cancer Registration.
2-Evidence of diagnostic in the biopsy results.
3-In cases in which the diagnostic cannot be confirmed through pathology, it will be taken in consideration the evidence of diagnostic tests of CT, MRI, PET Scan, Sonograms, that support the diagnostic or stage.
|
Effectiveness= From the date of the diagnostic certification by the Hematologist / Oncologist or date when the biopsy was performed if in this reading it establishes a definitive diagnostic.
Duration = Until the active treatment of the condition with radiotherapy or chemotherapy finishes. Every insured will receive a certification with the period of validity in the Registration, until the date that the insured complies with his/her surgical treatment, chemotherapy or radiotherapy. The insured will have the benefit of the coverage for visits to his hematologist oncologist up to a maximum of one year. After a year, the hematologist oncologist will perform an extension request in which he/she will document the condition’s stage and the treatment plan for the next year. The insurance company will grant a temporary registration up to a maximum of 30 days to receive the documentation in the Cancer Registration Extension sheet, provided by the insurance company. If the process is not complied with, the insured will automatically lose his/her registration coverage.
|
1. All hospital medical services, emergency room or medical specialist services rendered with a main diagnostic of cancer.
2-All medical services rendered or ordered by the hematologist / oncologist.
3- Medications prescribed by the oncologist / hematologist and specific to treat the condition.
|
Insurance Company - Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
3
|
In the cases of prostate cancer, the treatment with cancer agents such as Lupron, Eligard and Trelstar will be considered chemotherapy and they will continue active in the registration. Their visits to the urologist and the medical orders and treatment ordered by this specialist (urologist) will be covered.
|
||||
|
In the cases of breast cancer, once the active treatment with radiotherapy and chemotherapy is finished, the insured will no longer be in the registration. However, treatment with anti-estrogen (Tamoxifen) and Aromatase Inhibitors (Arimidex. Aromasim and Femara) will continue being considered a service under the cancer special coverage.
|
||||
|
Skin Cancer
Carcinoma IN SITU
|
- Positive Biopsy
|
Effectiveness: The results of skin cancer and carcinoma in situ will only be given special coverage at the date of the surgery.
Duration: The day or days of the procedure of surgical removal and all services during that day and any other radiotherapy code applied at any moment.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
4
|
Skin Cancer as Invasive Melanoma or those of the Squamous Cells with Evidence of Metastasis
|
- Pathology or positive Biopsy
- Special Studies CT Scan, MRI, Sonogram
- Certification of Registration filled out by a dermatologist or oncologist / hematologist
|
Effectiveness: The Special Coverage will begin from the date in which the diagnostic is established.
Duration = Until the active treatment of the condition with radiotherapy or chemotherapy finishes.
Every insured will receive a certification with the period of validity in the Registration,
for a maximum of a year. After a year, the hematologist / oncologist will perform a request for extension in which he/she will document the condition’s stage and the treatment plan for the next year. The insurance company will grant a temporary registration for up to a maximum of 30 days to receive the documentation in the Cancer Registration Extension sheet, provided by the insurance company. If the process is not complied with, the insured will automatically lose the registration coverage.
|
1. All hospital medical services, emergency room or medical specialist services rendered with a main diagnostic of cancer.
2-All medical services provided or ordered by the hematologist / oncologist.
3-Medications prescribed by the oncologist / hematologist and specific to treat the condition.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
5
|
Chronic Renal Disease
Level 1 and 2
Level 3 and 4
|
The glomerular filtration rate is used. Another requirement is evidence of recent creatinine in blood results, age, sex and race of the insured.
Level 1: GFR higher than 90,
ICD-9-CM 585.1
Level 2: GFR between 60-89,
ICD-9-CM 585.2
Level 3: GFR between 30-59,
ICD-9-CM 585.3
Level 4: GFR between 15-29,
ICD-9-CM 585.4
Level 5: GFR lower than 15,
ICD-9-CM 585.5 y
585.6 (ESRD)
|
Level 1 and 2: Are not registered.
Level 3 and 4: Require registration.
Duration = As long as he/she maintains eligibility in the PSG.
Duration = As long as the insured maintains eligibility in the PSG.
|
GMP/PCP: Level 1 and 2 are total risk of the GMP.
Level 3 and 4- The insurance company assumes the nephrologist’s visits (without the need of a referral), renal laboratories and diagnostic studies ordered by this specialist. Periferovascular studies to document the access to hemodialysis and the medications ordered by the nephrologist and related to the condition and limited to: immunosuppressive, erythrocytes stimulants, Megace, renal antidotes and systemic corticosteroids.
Level 5- All types of services as long as the insured is active in the registration.
|
GMP/PCP: Level 1 and 2 are total risk of the GMP.
Level 3 and 4: Insurance Company: All medical services rendered or ordered by a nephrologist from the effective date of the coverage.
Including also:
- Catheter insertion for dialysis
- Surgeries to establish arterio-venous fistulas.
- Administration of hematopoietic agents
- Transfusions
GMP/PCP Level 3 and 4; Primary care physician receives capitation.
Level 5: Once the Registration is authorized for Chronic Renal Condition, the insured receives a notification by mail, indicating the changes in the coverage or change of GMP to one of the Renal GMP. (Dialysis Center). The change of GMP will be effective in the month in which the request for change is performed. From this moment forward, the GMP ceases to receive the per capita payment corresponding to this insured. The risk of the services received by the insured previous to the change in GMP or registration of the insured will be the risk of the GMP, except those directly related with the dialysis. Ambulatory services, not emergency, that are rendered to these insured in the Renal GMP, have to be coordinated through a referral of the nephrologist, who will become the primary care physician for these insured.
GMP/PCP – Level 5 - Does not receive monthly capitation corresponding to the insured.
|
6
|
Scleroderma
|
1. Evidence of the positive ANA Test result > or equal to 1:80 dil
2. Result of a positive skin biopsy
3. Diagnostic Certification by the rheumatologist confirming the condition.
For the specialist’s certification, it will be required that it establishes that the diagnostic complies with at least one (1) major criteria or two (2) minor criteria (reference to the American College of Rheumatologists)
MAJOR CRITERIA: (1)
- Proximal Scleroderma
- Loss of skin elasticity
- Skin Hyperpigmentation and Hypopigmentation (salt &
pepper).
or
MINOR CRITERIA: (2)
- Sclerodactilia
- Loss of substance in the finger pad
- Pulmonary fibrosis in both bases
The insurance company or TPA will design a sheet for the registration of this condition to be filled out by the specialist that certifies the condition with the required elements to validate diagnostic and treatment plan.
|
Effectiveness = From the date of the diagnostic certification by the rheumatologist who establishes the definitive diagnostic.
Duration = As long as the insured maintains eligibility in the PSG.
|
1- All hospital medical services, emergency room or medical specialist services rendered with a main diagnostic of Scleroderma.
2- All medical services provided or ordered by the rheumatologist.
3-Medications prescribed by the rheumatologist and specific to treat the condition.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
7
|
Multiple Sclerosis and Amyotrophic Lateral Sclerosis
|
Revised criteria by McDonald:
The diagnostic is confirmed when a combination of the following are met:
1. Two (2) different episodes
Of verifiable neurological symptoms by a neurologist
2. Symptoms that indicate damage or lesion in more than one region of the Central Nervous System, MRI and laboratory tests with abnormal findings and consistent with MS
3. Absence of another disease or condition that could be causing the symptomatology or the laboratory findings.
1. Result of Brain MRI
2. Result of lumbar puncture
3. Diagnostic Certification by a neurologist confirming the condition and treatment.
|
The Special Coverage will begin from the date in which the definitive diagnostic is established.
Effectiveness = From the date of the diagnostic certification by a neurologist that establishes the definitive diagnostic and treatment.
Duration = As long as the insured maintains eligibility in the PSG.
|
1- All hospital medical services, emergency room, or medical specialist services rendered with a main diagnostic of Multiple Sclerosis and/or Amyotrophic Lateral Sclerosis.
2- All medical services rendered or ordered by a neurologist.
3-Medications prescribed by the neurologist and specific to treat the condition.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
|
Cystic Fibrosis
|
1. Sweat test.
2. Evidence of treatments
3. Diagnostic Certification by a pneumologist confirming the condition.
|
The Special Coverage will begin from the date in which the definitive diagnostic is established.
Effectiveness = From the date of the diagnostic certification by a pneumologist who establishes the definitive diagnostic.
Duration = As long as the insured maintains eligibility in the PSG.
|
All types of services as long as the insured is active in the registration.
|
Insurance Company- All medical services and medications as defined in the special coverage in this document.
GMP/PCP – Does not receive monthly capitation corresponding to the insured.
|
8
|
Hemophilia –
ICD-9 286-286.59
|
I- Evaluation of the Hematologist:
a- Severe: Level of Factor VIII <1%
b- Moderate: Level of Factor VIII<1-5%
c- Mild: Level of Factor VIII 5-25% with severe bleeding manifestations.
II- Levels of Coagulation Factors:
a- Patients with severe Hemophilia A and B.
b- Patients with severe Hemophilia A and B with presence of inhibitors.
c- Moderate Hemophilia A and B with presence of inhibitors.
1. Results of coagulation levels.
2. Diagnostic Certification by a hematologist or Hemophilia Clinics confirming the condition.
|
The Special Coverage will begin from the date in which the definitive diagnostic is established.
Effectiveness = From the date of the diagnostic certification by a hematologist who establishes the definitive diagnostic.
Duration = As long as the insured maintains eligibility in the PSG.
|
1- All hospital medical services, emergency room or medical specialist services rendered with a diagnostic of hemophilia.
ICD-9 286-286.59
2- All medical services provided by the hematologist.
3-Medications prescribed by the hematologist specific to treat the condition and all coagulation factors administered to the insured (anti-hemophilic).
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
|
Leprosy –
ICD-9 030-030.9
|
1. Evidence of skin biopsy result
2.Positive infection cultures
3. Diagnostic Certification by infectious diseases physician specialist, confirming the condition.
|
Effectiveness = It begins from the date of the certification which establishes the definitive diagnostic by an infectious diseases physician specialist and ends when the treatment is completed.
|
1-All hospital medical services, emergency room or medical specialist services, cultures and follow up biopsies with a diagnostic of leprosy, ICD-9 030-030.9.
2-All medical services rendered by the infectious diseases physician specialist.
3-Medications prescribed by the infectious diseases physician specialist.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
|
Systemic Lupus Erythematous
|
1-Diagnostic Certification by the rheumatologist with evidence of the following laboratory tests; ANA Test, DS-DNA, Anti Sm and Anti Phospholipids.
|
Effectiveness = From the date of the diagnostic certification by the rheumatologist who establishes the definitive diagnostic.
Duration = As long as the insured maintains eligibility in the PSG.
|
1- All hospital medical services, emergency room or medical specialist services rendered with a main diagnostic of Systemic Lupus Erythematous.
2- All medical services rendered or ordered by the rheumatologist.
3-Medications prescribed by the rheumatologist that is directed to treat the SLE condition.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
9
|
Children with Special Health Needs
|
To complete the Form of Registration of Children with Special Health Needs by the primary care physician with evidence of the medical condition, according to the list of diagnostics in the attachment included by ASES in the current contract titled “Conditions to Include Patients in the Registry of Children with Special Health Needs”.
The medical evidence will be composed of tests or pertinent laboratory tests, evidence of current treatment, diagnostic certifications by specialist physicians consulted and others.
|
Effectiveness = From the date of the diagnostic certification.
Duration = It will depend if the condition is permanent or temporary. The case manager will determine, based in the protocol established by the insurance company, the duration of the registration, limited to the fact that the insured is not older than 21 years of age.
|
All types of services while the insured is active in the registry.
|
Refer to the list of diagnostic codes of conditions for the Registry of Children with Special Needs.
|
|
Obstetrics
|
Registration Sheet for Obstetric Certification of pregnancy by the obstetrician gynecologist.
|
Inception: Once the case is registered, the insurance company will send a special coverage certification to the insured through the mail.
Duration: The registration will be effective on the estimated day of conception according to the obstetrician’s certification and will be effective until 56 days after the date of delivery, as long as this happens after week 20.
If the pregnancy ends in an abortion, before week 20, only 30 days will be granted after the abortion occurred.
|
All medical covered services.
As long as the insured is in the obstetrics Registry, the medications outside the obstetric formulary must be pre-certified, completing the request form and sending it through the fax determined by the insurance company.
Sterilizations:
The sterilization performed in a separate admission, after the delivery or cesarean section, will be the responsibility of the primary care physician group, for which they will require the referral of the primary care physician.
Newborns:
Newborn babies will have the coverage as long as they have their mothers’ contract and until the Obstetric registration ends (56 days after delivery) under the risk of the insurance company.
Under this premise, the assistance of the pediatrician during the cesarean section or high-risk labor and the routine care for the newborn in the hospital (nursery room) are part of the special coverage of obstetrics.
|
Insurance Company-
Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Does not receive monthly capitation corresponding to the insured.
Newborn: The per capita payment for the newborn will be paid once the mother leaves the registry or the newborn is certified by the mother, whatever occurs first.
|
10
|
Tuberculosis
|
Pneumologist Certification with a treatment plan and evidence of:
1- Tuberculin Test Result
2- Thorax X-Rays (lung infiltrates, cavities, consolidation, hiliar lymph nodes. disseminated nodes, miliar)
3- Samples of sputum for AFB and culture for M. tuberculosis or bronchial lavage when they cannot expectorate.
4- Biopsies of the affected area, if applicable.
5- HIV Test Result
|
Effectiveness: The Special Coverage will begin from the date in which the definitive diagnostic is established.
Duration: The coverage will be variable, depending on the duration of the treatment, which could vary from six (6) months to one (1) year, depending on the treatment plan certified by the pneumologist. After the first year, if the patient requires to continue treatment a reevaluation of the case will be requested by the pneumologist and according to the new treatment plan, the coverage will be extended.
|
The medical services related to the condition, follow up, complications, and/or complications of the diagnostic procedure and/or the treatment will be the risk of the Insurance Company from the effective date of the Special Coverage.
It will include any medication indicated to treat or control the special condition or conditions that could arise as part of the diagnostic studies performed or complications of the disease.
. Follow up Chest X-Ray Films until the treatment is completed is a risk of the insurance company.
The Department of Health covers:
- Tuberculin
- Cultures
- Bronchial Lavage
- Medical Treatment
|
Insurance Company- Medical services and medications as defined in the special coverage in this document.
GMP/PCP – Receives monthly capitation corresponding to the insured.
|
11
|
HIV-AIDS
|
Registration Certification by primary care physician or HIV clinic physician with evidence of:
1- Western Blot positive (IFA) Inmunofluorescent Assay positive
2- CD 4 Test
3- Evidence of Opportunistic Diseases:
- Candidiasis
- Uterine cervix Cancer (invasive)
- Coccidioidomicosis, criptococosis, cryptosporidiosis
- Cytomegalovirus Disease
- Encephalopathy (related with HIV)
- Simple Herpes (severe infection)
- Histoplasmosis
- Isosporiasis
- Kaposi Sarcoma
- Lymphoma (some types)
- Mycobacterium avium complex
- Pneumonia by pneumocystis carinii/jiroveci
- Pneumonia (recurring)
- Progressive Multifocal Leukoencephalopathy (PML)
- Septicemia by salmonella (recurring)
- Brain Toxoplasmosis
- Tuberculosis
- Emaciation Syndrome
|
Effectiveness:
The effectiveness of the Coverage will begin from the date in which the definitive diagnostic is established.
Duration:
The special coverage will be active as long as the insured maintains eligibility in the PSG.
|
All types of services as long as the insured is active in the Registry.
|
Insurance Company-
All medical services and medications as defined in the special coverage in this document.
GMP/PCP – Does not receive monthly capitation corresponding to the insured.
|
12
Attachment 8
Cost-Sharing
|
CO-PAYS & CO-INSURANCE - Effective on July 1st, 2013
|
||||||||
|
Services
|
Federal
|
CHIPS
|
Población Estatal
|
ELA*
|
||||
|
100
|
110
|
230
|
300
|
310
|
320
|
330
|
400
|
|
|
HOSPITAL
|
HOSPITAL
|
HOSPITAL
|
HOSPITAL
|
HOSPITAL
|
||||
|
Admissions
|
$0
|
$3
|
$0
|
$3
|
$5
|
$6
|
$20
|
$50
|
|
Nursery
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
EMERGENCY ROOM (ER)
|
||||
|
Emergency Room (ER) Visit
|
$0
|
$0
|
$0
|
$1
|
$5
|
$10
|
$15
|
$20
|
|
Non-emergency visit to a hospital emergency room.
|
$3.80
|
$3.80
|
$0
|
$15
|
$15
|
$15
|
$15
|
$20
|
|
Trauma
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
AMBULATORY VISITS TO
|
||||
|
Primary Care Physician (PCP)
|
$0
|
$1
|
$0
|
$0
|
$1
|
$2
|
$2
|
$3
|
|
Specialist
|
$0
|
$1
|
$0
|
$1
|
$1
|
$3
|
$4
|
$7
|
|
Sub-Specialist
|
$0
|
$1
|
$0
|
$1
|
$1
|
$3
|
$5
|
$10
|
|
Pre-natal services
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
OTHER SERVICES
|
OTHER SERVICES
|
OTHER SERVICES
|
OTHER SERVICES
|
OTHER SERVICES
|
||||
|
High-Tech Laboratories**
|
$0
|
50¢
|
$0
|
$1
|
$1
|
$2
|
$3
|
20%
|
|
Clinical Laboratories**
|
$0
|
50¢
|
$0
|
$1
|
$1
|
$2
|
$3
|
20%
|
|
X-Rays**
|
$0
|
50¢
|
$0
|
$1
|
$1
|
$2
|
$3
|
20%
|
|
Special diagnostic Tests**
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$6
|
40%
|
|
Therapy – Physical
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$3
|
$5
|
|
Therapy – Respiratory
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$3
|
$5
|
|
Therapy – Occupational
|
$0
|
$1
|
$0
|
$1
|
$2
|
$2
|
$3
|
$5
|
|
Vaccines
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
Healthy Child Care
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
DENTAL
|
DENTAL
|
DENTAL
|
DENTAL
|
DENTAL
|
||||
|
Preventive (Child)
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
|
Preventive (Adult)
|
$0
|
$1
|
$0
|
$0
|
$1
|
$2
|
$3
|
$3
|
|
Restorative
|
$0
|
$1
|
$0
|
$0
|
$1
|
$5
|
$6
|
$10
|
|
PHARMACY***
|
PHARMACY***
|
PHARMACY***
|
PHARMACY***
|
PHARMACY***
|
||||
|
Generic (Children 0-18)
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$5
|
|
Generic (Adult)****
|
$1
|
$1
|
N/A
|
$1
|
$2
|
$3
|
$5
|
$5
|
|
Brand (Children 0-18)
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$0
|
$10
|
|
Brand (Adult)****
|
$3
|
$3
|
N/A
|
$3
|
$4
|
$5
|
$7
|
$10
|
|
Services
|
Federal
|
CHIPS
|
Población Estatal
|
ELA*
|
||||
|
100
|
110
|
230
|
300
|
310
|
320
|
330
|
400
|
|
*Code 400 in ELA column refers to the population that subscribes as public employees of the Puerto Rico Government.
** Apply to diagnostic tests only. Copays do not applied to tests required as part of a preventive service.
***Copays apply to each drug included in the same prescription pad. Pharmacy exception (children 0- 18) does not apply to 400 ELA employees.
****Co-pays for children 0-18 years of age are not applicable for Medicaid, Commonwealth medically indigent eligible, and for children 0-18 enrolled in the CHIP Program in group ages 0-18.
Co-pays may apply to children ages over 18 years old as well as to adults.
Co-pays may apply to children ages over eighteen (18) as well as to adults.
As established in 42 CFR 44.7.53(b) the following exceptions will be applicable for federal population under code 110:
(b) Exclusions from cost sharing. The plan may not provide for imposition of a deductible, coinsurance, copayment, or similar charge upon categorically or medically needy individuals for the following:
(1) Children. Services furnished to individuals under 18 years of age (and, at the option of the State, individuals under 21, 20, or 19 years of age, or any reasonably category of individuals 18 years of age or over but under 21) are excluded from cost sharing.
(2) Pregnant women. Services furnished to pregnant women if such services related to the pregnancy, or to any other medical condition which may complicate the pregnancy are excluded from cost sharing obligations. These services include routine prenatal care, labor and delivery, routine post-partum care, family planning services, complications of pregnancy or delivery likely to affect the pregnancy, such as hypertension, diabetes, urinary tract infection, and services furnished during the postpartum period for conditions or complications related to the pregnancy. The postpartum period is the immediate postpartum period which begins on the last day of pregnancy and extends through the end of the month in which the 60-day period following termination of pregnancy tests. States may further exclude from cost sharing all services furnished to pregnant women if that desire.
(3) Institutional individuals. Services furnished to any individual who is an inpatient in a hospital, long-term care facility, or other medical institution if the individual is required (pursuant to §435.725, §435.733, §435.832, or §436.832)
(4) Emergency services. Services as defined at section 1932(b)(2) of the Act and §438.114(a)
(5) Family planning. Family planning services and supplies furnished to individuals of child-bearing age are excluded from cost sharing.
(6) American Indians. Items and services furnished to an American Indian directly by an American Indian health care provider or through referral under contract health services.
Pharmacy Management Program
aaa












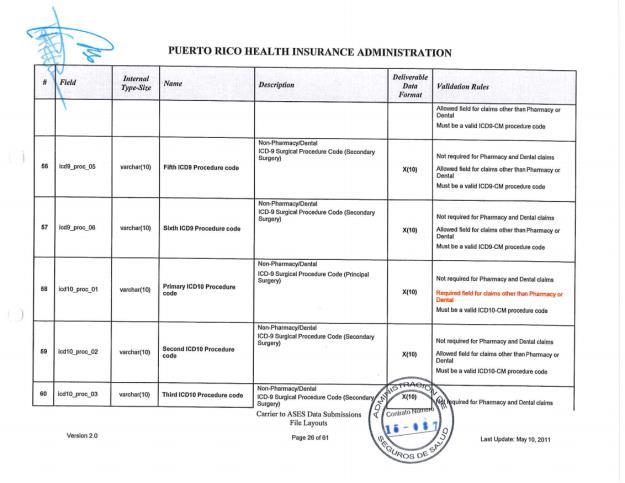
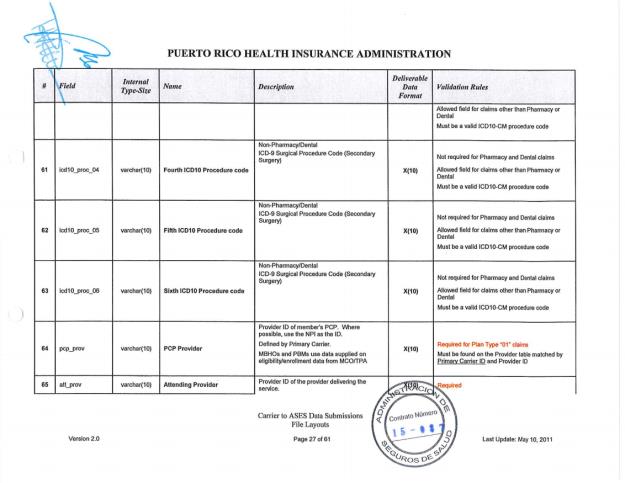
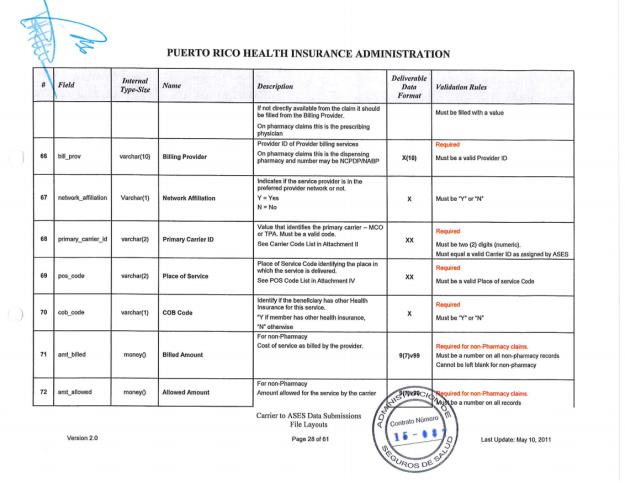

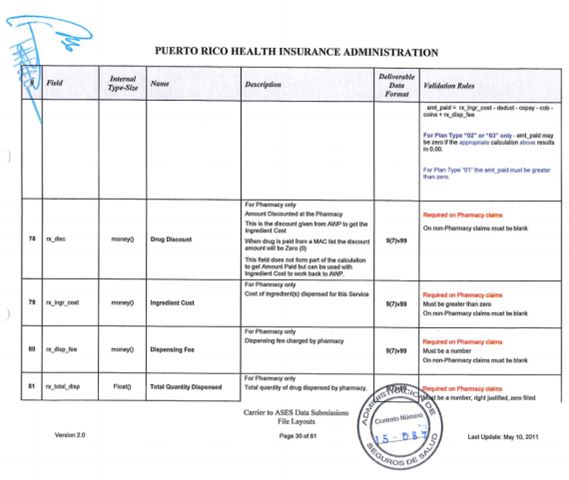

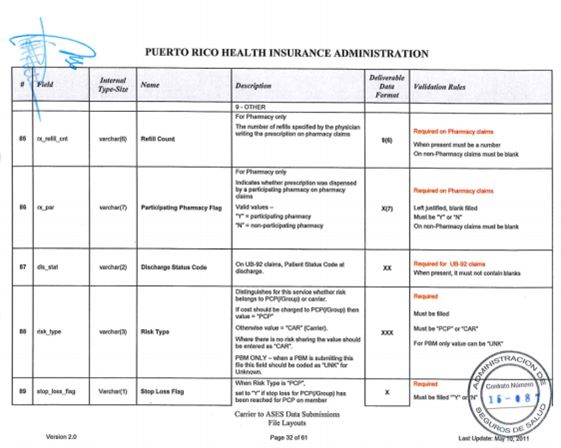











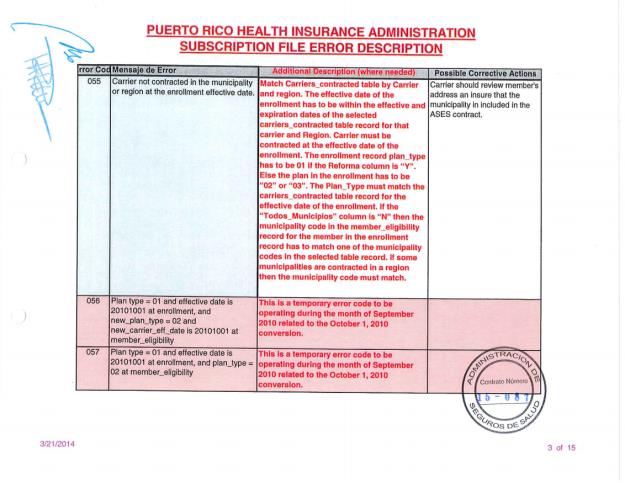

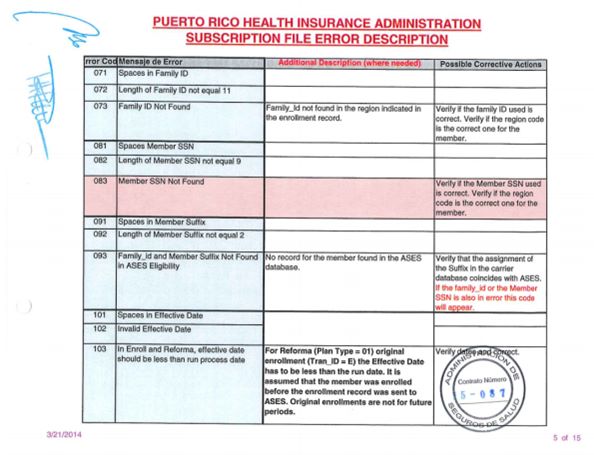
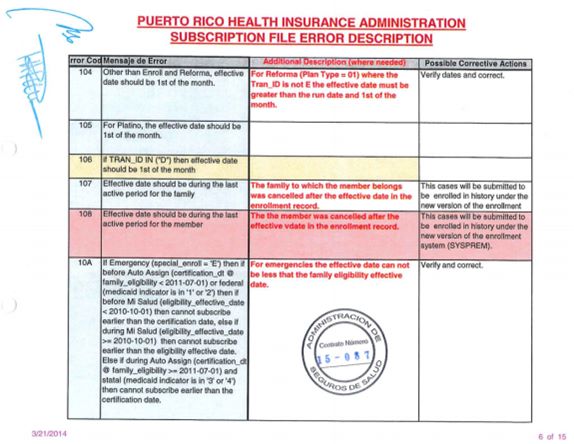






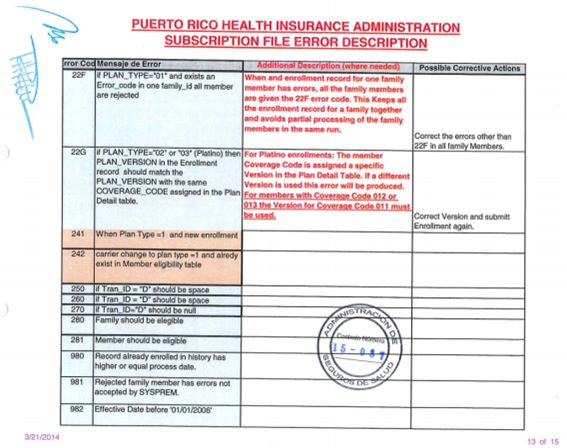




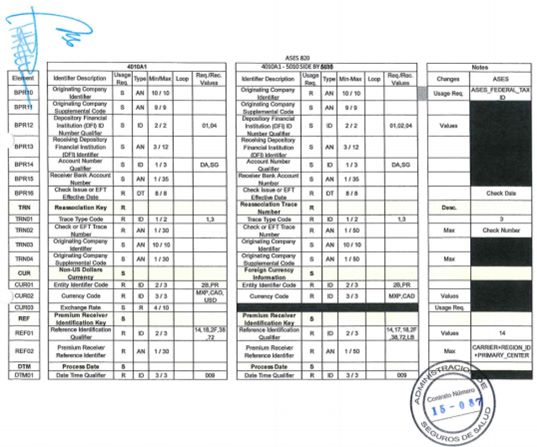
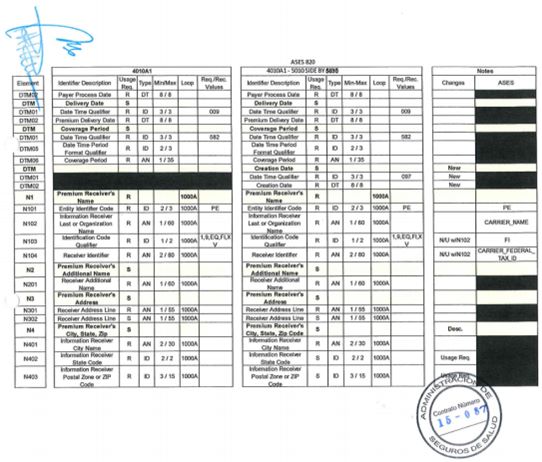






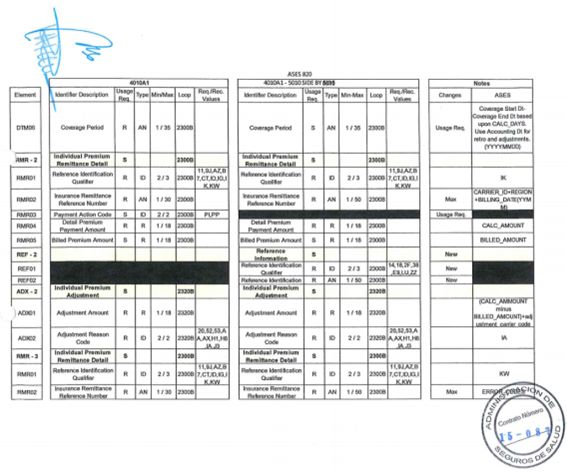
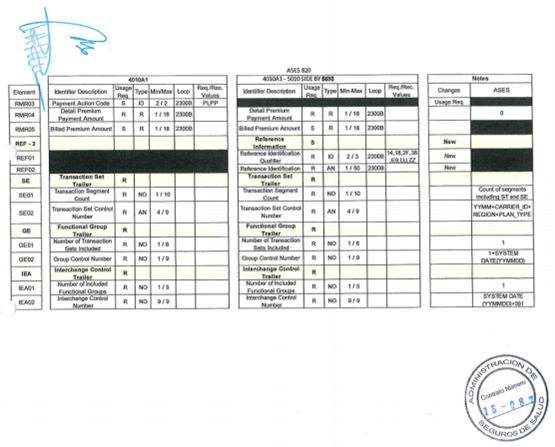
Attachment 9
ASES
Enrollment Manual
June 2011
Table of Contents #
|
I. INTRODUCTION
|
|||
|
A.
|
BACKGROUND
|
||
|
B.
|
BASIC ELIGIBILITY CONCEPTS
|
||
|
C.
|
GENERAL ENROLLMENT CONCEPTS
|
||
|
II. ENROLLMENT PROCESS
|
|||
|
A.
|
DATA FLOW
|
||
|
i. Mi Salud
|
|||
|
ii. Platino
|
|||
|
B.
|
ENROLLMENT RECORD
|
||
|
i. Data definition
|
|||
|
ii. Uses
|
|||
|
iii. Edit & Update Process
|
|||
|
C.
|
CARRIER RESPONSIBILITIES
|
||
|
D.
|
ENROLLMENT RECORD REJECTIONS
|
||
|
i. Reject Process
|
|||
|
ii. Error Codes
|
|||
|
III. PREMIUM PAYMENT
|
|||
|
A.
|
CONCEPTS
|
||
|
B.
|
RELATION TO ENROLLMENT
|
||
|
C.
|
TYPES OF PAYMENT CALCULATIONS
|
||
|
IV. SYSPREM – ENROLLMENT IN HISTORY
|
|||
|
A.
|
ENROLLMENT CONCEPTS
|
||
|
B.
|
SYSPREM FUNCTIONALITY
|
||
|
C.
|
PREMIUM PAYMENT FOR SYSPREM ENROLLMENTS
|
||
|
V. ADDENDUMS
|
|||
|
A.
|
ENROLLMENT RECORD LAYOUT
|
||
|
B.
|
Consistency Hierarchy Table
|
||
|
C.
|
ERROR CODE TABLE
|
||
|
E.
|
FLOW DIAGRAM
|
||
I. Introduction This document is the reference manual to guide Insurance Companies and Medicare Advantage Organizations contracted by ASES in enrolling their contracted beneficiaries.
|
a.
|
Background
|
|
|
Previous to January 2006 Mi Salud beneficiaries were assigned to MCO’s or TPA’s by region. (MCO’s, TPA’s and MAO’s will be referred to as “carriers” in this document). Enrollment, which is the process by which the carrier sends an electronic record to ASES notifying of the subscription of a member, was done at the family level. With one record the carrier would enroll all the members of a family. At the most there could be two carriers in a region, one MCO and one TPA so conflicts were minimal. The establishment of the Medicare Platino Plans by ASES starting on January 2006 increased the complexity of identifying in the ASES database which member is covered by which organization. Once Platino was implemented the enrollment had to be done at the member level since a family could have members subscribed by different carriers. The complexity was also affected by having MAO’s providing services to all the ASES regions. Therefore Platino beneficiaries had a wide choice of options which included the capacity to change carriers on a monthly basis.
|
|
b.
|
Basic Eligibility Concepts
|
|
i.
|
Eligibility for Mi Salud beneficiaries is determined by Medicaid Offices. Typically the beneficiaries are given eligibility for a year after which they must recertify.
|
|
ii.
|
Those beneficiaries which do not recertify are cancelled at the eligibility expiration date. This occurs at the end of each month.
|
|
iii.
|
Data for eligible beneficiaries is sent by Medicaid Offices to ASES and updated in the ASES database on a daily basis.
|
|
iv.
|
ASES sends any updates, cancellations or additions to the carriers on a daily basis.
|
|
1) Mi Salud carriers receive data for all the members in their contracted regions.
|
|
2) Platino carriers receive data for all their members enrolled in each contracted region.
|
|
v.
|
Mi Salud eligible members are those which appear as eligible in the ASES database.
|
|
vi.
|
Platino eligible members are those Mi Salud eligible members which also have Medicare A&B coverage.
|
|
1) Medicare A&B coverage is determined by the Platino carriers by querying CMS.
|
|
2) Platino carriers also have to query ASES to determine Mi Salud eligibility.
|
|
c.
|
General Enrollment Concepts
|
|
The enrollment record (see attached) used by the carriers to notify ASES of the subscription of a member contains a series of data elements for verification of correctness and to inform ASES the particulars of the enrollment. A member can be enrolled in one of three different Plan Types :
|
|
i. 01 = Mi Salud
|
|
ii. 02 = Platino MA-SNP (Special Needs Plan)
|
|
iii. 03 = Platino MA-PD (Medicare Advantage Prescription Drugs)
|
|
A particular carrier can offer different products under a Plan Type. These products are identified by their Plan Version number. ASES assigns a Plan Version number for each Platino product contracted. For Mi Salud enrollments the Plan Version field must equal the coverage code assigned to the particular beneficiaries. Some of the Plans contracted with ASES may require the assignment of Primary Centers (IPAs) and /or PCP s to the beneficiaries. The enrollment record includes those fields as well as the Plan Type and Version. The record also informs of the date the member was processed by the carrier and the effective date of the enrollment. (For more detail se section II.b below.)
|
II. Enrollment Process
|
a.
|
Data Flow
|
|
The data flow for Mi Salud and Platino enrollments is similar with the principal exception of the queries that are needed in the Platino process. (see flow diagram attached)
|
|
i.
|
Mi Salud – The process starts with the receipt of the eligibility data by the carriers. The carriers update their database and communicate with the beneficiaries. The beneficiaries visit the carriers’ premises and sign up in the Mi Salud Plan. The carrier then produces the electronic enrollment record and sends it to ASES. These transmissions occur on a daily basis. In ASES the records are passed through an edit program. The records that pass the edits are updated to the ASES database and the beneficiaries are deemed enrolled. Those record found with error are returned to the carriers for correction. Until the records are submitted correctly the member is not enrolled in ASES.
|
|
ii.
|
Platino – Before a Platino Plan can enroll a member it must verify Medicare coverage by querying CMS. They must also query ASES to verify if the member is eligible for Mi Salud. Once those requirements are met then the enrollment is submitted to ASES. In ASES the record follows the same process as described above for Mi Salud.
|
|
b.
|
Enrollment Record
|
|
i.
|
Data Definition – The enrollment record contains the following data elements to be complimented by the carrier:
|
1) RECORD_TYPE – This is always an “E” it identifies the record as an enrollment file record.
2) TRAN_ID – This is the field which identifies to the ASES system which action to take based on the data contained in the record. It can have one of several values:
|
a)
|
E = means that the record is a new enrollment for a member which has not been previously enrolled.
|
|
b)
|
C = Change Carrier. Used when the member has selected a different carrier than the one in which he/she is presently enrolled. It is also used for initial enrollment in Platino Plans.
|
|
c)
|
P = Changes the Plan Type. It is used when a member enrolled under a particular carrier chooses to change the product the carrier offers to one which is identified under a different Plan Type under the same carrier. Example: changing from an MA-PD Plan (Type 03) to a SNP Plan (Type 02) under the same carrier.
|
|
d)
|
V = Type Version change. It is used when a member enrolled under a particular carrier and Plan Type chooses to change the product the carrier offers to one which is identified under the same Plan Type but with a different version number under the same carrier. Example: changing from a SNP Plan (Type 02 Version 001) to a SNP Plan (Type 02 Version 002) under the same carrier. The version change value in the Tran_id is also used when a Mi Salud member changes coverage code. In this case the carrier must reissue an ID Card with the new benefits and submit a version change enrollment record to ASES where the Version number is equal to the coverage code.
|
|
e)
|
I = IPA (Primary Center) Change. Used to record in ASES a change in the beneficiaries’ selected IPA under the same carrier, Plan Type and Version.
|
|
f)
|
1 = PCP1 change. Used to record in ASES a change in the beneficiaries’ selected PCP1 under the same carrier, Plan Type, Version and IPA.
|
|
g)
|
2 = PCP2 change. Used to record in ASES a change in the beneficiaries’ selected PCP2 under the same carrier, Plan Type, Version and IPA.
|
|
h)
|
3 = PCP1 and PCP2 change. Used to record in ASES a change in the beneficiaries’ selected PCP1 and PCP2 under the same carrier, Plan Type, Version and IPA.
|
|
i)
|
X = delete incorrect enrollment 10)
|
|
j)
|
O = Contract number change only 11)
|
|
k)
|
D = Disenroll. For future use.
|
3) PROCESS_DATE – Sign up date. Date the member contracted with the carrier. Relationship with effective date:
a) Platino – Process date must be less than effective date.
b) Mi Salud – process date must be equal or less than effective date.
4) REGION – Contains ASES region code. Must be the region in which the member is located in the ASES database. Platino carriers obtain this code from the ASES query response.
5) CARRIER - Two digit carrier code assigned by ASES.
6) MEMBER_PRIMARY_CENTER – Up to four digits assigned by carrier to identify their Primary centers (IPAs). Not required for some Plan Types/Versions.
7) ODSI_FAMILY_ID – Eleven digit family ID assigned by MEDICAID OFFICES (ODSI). This is the first part of the key for the beneficiaries in the ASES database. Platino carriers obtain this code from the ASES query response.
8) MEMBER_SSN– Social Security number of the member. It is required that this number matches with the one for the member in the ASES database.
9) MEMBER_SUFFIX – Two digit number which identifies a member within a family. Second part of the key in the ASES database.
10) EFFECTIVE_DATE – Date in which the carriers starts coverage for the member under the enrolled Plan or effective date of the change for which the
11) PLAN_TYPE – Plan Type code under which the member is enrolled.
12) PLAN_VERSION – Plan version under which the member is enrolled.
13) MPI – Master Patient Index. Unique number which identifies a Member in ASES and MEDICAID OFFICESs databases.
14) PCP1 – Fifteen digit number assigned by carriers. Use to identify the PCP1 selected by the beneficiaries.
15) PCP1_EFFECTIVE_DATE – Date in which the PCP1 assignment was effective.
16) PCP2 – Fifteen digit number assigned by carriers. Use to identify the PCP2 selected by the beneficiaries.
17) PCP2_EFFECTIVE_DATE – Date in which the PCP2 assignment was effective.
18) FAMILY_PRIMARY_CENTER – IPA assigned to all Mi Salud family members.
19) FAM_PRIMARY_CENTER_EFF_DATE –Datein which the assignment of the family IPA was effective.
20) IPA_PCP_CHANGE_REASON – Not in use.
21) MEDICARE INDICATOR – Required for Platino enrollments. (1=A&B, 3=A, 9=B)
22) HIC NUMBER – Medicare Health Insurance Claim Number. Required for Platino enrollment.
23) IPA_ESPECIAL – A “1” indicates that the member is assigned to a special IPA which is not the family IPA. Used for Mi Salud.
24) Contract Number – Contract number assigned by the carrier. It should be the number by which the member is identified in the carriers ID card and internally in their database.
25) Special Enroll – Used to identify that the enrollment is for a newborn (N) or an emergency (E) case submitted by MEDICAID OFFICES or ASES. When this field is used then if the values is:
a) N – The system allows enrollment as of the date of birth.
b) E – The system allows enrollment as of the certification date.
c) This mechanism can be utilized in cases where the date of birth or certification is on or after January 1, 2006.
26) Other data elements complimented by ASES – When the record is edited the ASES system enters the following data in the enrollment record:
a) Reject Identifier - As a result of the edits the record could be accepted or rejected. This field contains the codes that specify that result. ( "A" = Accepted; "M" = Accepted Retroactive; "R" = Rejected; "X" = Deleted)
b) Record Key – Internal number assigned by the ASES system.
c) Error Codes one to ten – record up to ten possible error codes.
d) Update Date – Date to which the edit run belongs. Correspond to the date of the daily cycle the edit run was a part of.
e) Update User – ASES internal user code.
ii. Uses
1. The enrollment record can be used to trigger several actions in the ASES database. The content of the TRAN_ID field determines which action. An “E” for a Mi Salud carrier will perform the original enrollment of a member. A “C” will transfer a member from one carrier to the one submitting the enrollment or perform the original enrollment for a Platino carrier. Codes P, V, I, 1, 2, and 3 will inform the ASES system that the carrier has changed a beneficiaries Plan, Version, IPA or PCP. An “X” will delete a previously submitted record and an “O” will change a beneficiaries Contract number. In the future a “D” will produce the disenrollment of a member from its existing carrier.
iii. Edit and update process – Carriers can transmit enrollment files to ASES on a daily basis. They must follow the naming convention for those files which is as follows:
1. CCYYMMDD.SUS
1. CC = Carrier Code
2. YY = Year
3. MM = Month
4. DD = Day
5. .SUS = File extension identifies enrollment file.
The enrollment file can contain records pertaining to any of the regions contracted by the carrier. The files received by 9:00am are entered in the ASES daily cycle. If a file is received after 9:00am it will be entered in the following day’s cycle. In the cycle there are several steps which handle the enrollment records:
2) Enrollment Merge – joins the enrollment files from all carriers into a single file.
3) Enrollment Region Split – Separates the merged file into different files (one per region) based on the region code in the enrollment records. If the record sent does not have a valid region code it will go into a special error file and will not continue processing.
4) Edits - ASES run a separate edit and update cycle for each region. The enrollments are passed though the edit programs and are identified as valid or rejected.
5) Update - Valid enrollments will be used to update the beneficiaries’ record in the ASES database. In this process the data in the enrollment record is entered into the beneficiaries’ record. There are to types of Valid enrollments:
a) Reject identifier = A – Identifies an accepted enrollment which is to be applied at a future effective date. The update process moves the enrollment fields (carrier, Plan, Version, Ipa and PCP) to the fields destined for new enrollments in the member’s record. Until the new effective date is reached the member stays under the present enrollment condition (same carrier, Plan, Version, Ipa and PCP). At the month end cycle previous to the effective date the new field are moved to the actual fields and the enrollment becomes effective.
b) Reject identifier = M – Indicates a retroactive enrollment. In these cases the enrollment data (carrier, Plan, Version, Ipa and PCP) is updated directly to the actual enrollment field in the member’s record.
6) Carrier eligibility file extract – When the member’s information is updated because of an enrollment being processed, a record is sent to the carrier affected in the Carrier eligibility file which is produced in every daily cycle.
c. Carrier Responsibilities - In order to process enrollment transactions correctly the carriers need to maintain in their particular systems the updated member eligibility data received from ASES. Such data is sent by ASES in the following files:
i. Carrier Eligibility File (Daily & Month End) – Produced by the ASES daily cycle. Contains all the data pertaining to the beneficiaries that have been added, updated or cancelled in the daily cycle. This includes updates caused by enrollment records being processed in that cycle. The carrier’s system must identify the following situations based on the data received in these files:
1) When a member is added.
a) Mi Salud carriers must start the enrollment process with the member.
2) When a member changes carrier:
a) The carrier which lost the member must identify the loss of business.
3) When any of the enrollment data changes. This includes Plan Type, Version, IPA, PCPs.
a) The carrier system must be updated accordingly, If not this could cause the rejection of future enrollment record submissions.
4) When a Member’s demographics Changes:
a) The carrier needs to update the new data in their database.
5) When a member is cancelled:
a) All carriers must cancel effective at the end of the month
b) Carriers should follow up with member in case the cancellation is caused by expiration of certification.
6) When a member has a change in coverage code:
a) Carriers must evaluate if the new coverage code requires that the member be enrolled in a different Plan_Version and send a Version change enrollment record to ASES before the end of the month.
b) Members where the Plan_Version does not agree
with the coverage code will be disenrolled by ASES during the month end cycle. (For valid members, the carrier must then re-enroll the member under a new Plan_Version that agrees with the new coverage code.)
ii. Enrollment Reject File – Produced by the ASES daily cycle. It contains the enrollment records rejected by the validation program. The carrier must examine the rejected records and take action to correct the cause based on the error codes included. See details below about the specific error codes. The carriers system must have the capability of identifying the errors and provide the mechanisms for correction and submittal to ASES for reprocessing.
d. Enrollment Record Rejections
i. Reject Process - Rejected enrollments are sent daily on a file which includes the error codes for the edit that failed the validation process. The carriers must correct the errors found and submit the corrected records to ASES in the next enrollment file. The file name for the reject file is:
1. CCYYMMDD.rjc
a. CC = Carrier Code
b. YY = Year
c. MM = Month
d. DD = Day
e. .rjc = File extension identifies reject file.
ii. Error Codes – The attached (Subscription Error table) table contains the error codes produced by the Validation Program. Additional descriptions and possible corrective actions have been included to assist in the correction process.
III. Premium Payment
a. Concepts - The new Premium Payment System works under the concept that premiums are calculated and paid for only those beneficiaries that are enrolled by the first day of the payment month. The carriers do not need to submit billing documents or files. There is one payment run per month per ASES region in which the payment for all carriers in the region is calculated.
b. Relation to Enrollment - Enrolled beneficiaries are those which are eligible and assigned to a particular carrier as the result of an enrollment transaction. For a particular month’s run the system will consider enrolled beneficiaries in the ASES database with an enrollment date (update date in ASES) previous to the 1 st day of that month. Beneficiaries enrolled after that date will be considered for payment in the next payment run after the enrollment date.
c. Types of payment calculations - The payment system computes several categories of payments:
a. Monthly payments – For all beneficiaries enrolled at the beginning of the month for which the system is run ( Payment Month ).
b. Prorate Payments – Prorate payments are calculated for Mi Salud beneficiaries that were enrolled during the previous month to the payment month. A prorated daily premium is calculated based on effective date of the enrollment.
c. Retroactive Payments – Is calculated when the effective date of the enrollment is previous to the payment month. In Platino this calculation may include the previous month since no prorate is paid and because the enrollment always starts at the beginning of a month. In Mi Salud retroactive payments are always for periods two month or more before the payment month.
d. Retroactive prorate payments - Retroactive prorate payments are calculated when the effective date of the enrollment falls within the first month considered for a retroactive payment
e. Adjustments – Adjustments are calculated when a member changes Carrier retroactively after ASES had paid the first carrier in a previous payment run. The adjustment takes away the premium amount paid the first carrier.
IV. SYSPREM – Enrollment in History
a. Enrollment concepts
i. Enrollments are applied to the current eligibility data.
ii. Enrollments are allowed only in a member’s current eligibility period. The current eligibility period is the:
|
1)
|
eligibility period after a cancellation period (for a member that has been cancelled and then re-certified)
|
| 2) | the current period since the initial update in ASES (as eligible) and the present time when the member has not been cancelled and remains eligible |
| iii. | When an enrollment is not sent in time by the carrier (or a rejected record is not corrected) the eligibility data for the member will remain un-enrolled. |
|
iv.
|
Premiums will not be paid for un-enrolled beneficiaries when the premium payment system is run.
|
| v. | If the member is then cancelled or enrolled in a second carrier the first carrier is prevented (by the system edits) to enroll the member in a period previous to the cancellation or the enrollment. |
b. SYSPREM Functionality. The SYSPREM sub-system will permit the enrollment of beneficiaries to be recorded in historic data. The main functions are:
i. Identification of enrollment records that are candidates for processing against the history database. Rejected with error codes:
1) 107- Effective date before current eligibility period for family
2) 108- Effective date before current eligibility period for member
3) 280- Family must be eligible in current eligibility period
4) 281- Member must be eligible in current eligibility period
5) 177- Enrolled in another carrier at or after effective date
ii. Limitations:
1) Member must be active on effective date
2) Member must not have family members with errors not acceptable by SYSPREM in the same Mi Salud enrollment batch
3) Enroll record must not have Effective Date before 01/01/2006***
iii. New Error Codes (Reject File) for accepted history enrollments:
1) 996 – SYSPREM record inserted in history. No action by the carrier is required.
iv. New Error Codes (Reject File) for rejected history enrollments:
1) 980 - Process date in enroll record must be greater than process date of the previously enrolled Member record
2) 981 – Member must not have family members with errors not acceptable by SYSPREM in the same enrollment batch (for Mi Salud).
3) 982 – Enroll record must not have Effective Date before 01/01/2006***
v. Carrier Eligibility File – The daily carrier eligibility file will include the data for the members updated in history by the SYSPREM sub-system. The TRAN_ID field will contain an “H” to identify history data. The carriers must modify their systems so that the SYSPREM data is not included as actual data when processing the eligibility file.
c. Premium Payment for SYSPREM enrollments
i. Monthly Premium Payment run will include all SYSPREM records processed during the previous month.
ii. Payment will be calculated for months from the effective date of the SYSPREM enrollment up to:
1) The month in which the member is enrolled in a different carrier
2) The month in which the Member is cancelled
3) Actual Billing date
d. SYSPREM in summary
i. SYSPREM will enroll beneficiaries in history for cases where the enrollment cannot be applied to actual data.
ii. Some members will not be enrolled in history because they are:
1) Not eligible at the effective date
2) Enrolled in a different carrier
iii. Carriers need to evaluate cases rejected by SYSPREM in order to determine:
1) Errors in the effective date assigned
2) Correctness of the beneficiaries' data included in the enrollment record
V. Addendums
e. Enrollment Record Layout
f. Consistency Hierarchy Table
g. Error Code Table
h. Flow Diagram
ENROLLMENT AND CARRIER IPA/PCP CHANGE FILE
|
November 1, 2007
|
|
This file is received by ASES from the insurance companies and TPO’s on a daily basis. It contains data pertinent to new enrollment and families which have selected to change their enrollment to the organization sending the file. Modified for Medicare Plan Enrollment on September 2005. Concept change from one record per family enrolled to one record per member. Modify to include special enroll field on novembre 2007. Modified on April 2013 to include Trailer record for the Migration Project.
|
|
Member Record
|
|||
|
Record Fields
|
Position
|
Size
|
Notes
|
|
RECORD_TYPE
|
1
|
1
|
"E" for Enrollment Record (Constant)
|
|
TRAN_ID
|
2
|
1
|
E=new enrollment, P=Plan Type change, C=Carrier change, V= Version change, I=IPA change, 1=PCP1 change, 2=PCP2 change, 3=PCP1 and PCP2 change, X= Delete incorrect enrollment, O=Contract Number Change only
|
|
PROCESS_DATE
|
3
|
8
|
MMDDYYYY - Date Enrolled by Carrier
|
|
REGION
|
11
|
1
|
Region code
|
|
CARRIER
|
12
|
2
|
Carrier code
|
|
MEMBER_PRIMARY_CENTER
|
14
|
4
|
IPA or PHO code
|
|
ODSI_FAMILY_ID
|
18
|
11
|
|
|
MEMBER_SSN
|
29
|
9
|
|
|
MEMBER_SUFFIX
|
38
|
2
|
|
|
EFFECTIVE_DATE
|
40
|
8
|
MMDDYYYY- Card issue date for new Reforma enrollment (Trans_ID= E) or Effective date (1st day of month) for other Trans_ID's
|
|
PLAN_TYPE
|
48
|
2
|
See Plan Type Table
|
|
PLAN_VERSION
|
50
|
3
|
Used to identify version of Plan within PLAN_TYPE (if needed)
|
|
MPI
|
53
|
13
|
Alpha-numeric ej.-"0080012345678"
|
|
PCP1
|
66
|
15
|
Text
|
|
PCP1_EFFECTIVE_DATE
|
81
|
8
|
MMDDYYYY
|
|
PCP2
|
89
|
15
|
Text
|
|
PCP2_EFFECTIVE_DATE
|
104
|
8
|
MMDDYYYY
|
|
FAMILY_PRIMARY_CENTER
|
112
|
4
|
IPA or PHO code
|
|
FAM_PRIMARY_CENTER_EFF_DATE
|
116
|
8
|
MMDDYYYY
|
|
IPA_PCP_CHANGE_REASON
|
124
|
2
|
Code Table to be supplied
|
|
MEDICARE INDICATOR
|
126
|
2
|
1=A&B, 3=A, 9=B
|
|
HIC NUMBER
|
127
|
12
|
|
|
Reject Identifier
|
139
|
1
|
"A" = Accepted; "M" = MA Retroactive; "R" = R ejected; "X" = Deleted
|
|
Record Key
|
140
|
14
|
YYYYMMDD999999
|
|
Error Code 1
|
154
|
3
|
Indicates error (see error code table)
|
|
Error Code 2
|
157
|
3
|
Indicates error (see error code table)
|
|
Error Code 3
|
160
|
3
|
Indicates error (see error code table)
|
|
Error Code 4
|
163
|
3
|
Indicates error (see error code table)
|
|
Error Code 5
|
166
|
3
|
Indicates error (see error code table)
|
|
Error Code 6
|
169
|
3
|
Indicates error (see error code table)
|
|
Error Code 7
|
172
|
3
|
Indicates error (see error code table)
|
|
Error Code 8
|
175
|
3
|
Indicates error (see error code table)
|
|
Error Code 9
|
178
|
3
|
Indicates error (see error code table)
|
|
Error Code 10
|
181
|
3
|
Indicates error (see error code table)
|
|
Update Date
|
184
|
8
|
YYYYMMDD
|
|
Update User
|
192
|
8
|
"SYSTUPD "
|
|
IPA_ESPECIAL
|
200
|
1
|
1 = IPA Especial
|
|
Contract Number
|
201
|
13
|
Character left justified
|
|
Special Enroll
|
214
|
1
|
E = Emergency N = New Born
|
|
Filler
|
215
|
15
|
|
|
230
|
|
TRAILER Record
|
|||
|
Record Fields
|
Position
|
Size
|
Notes
|
|
RECORD_TYPE
|
1
|
7
|
“TRAILER” for Record (Constant)
|
|
FILLER
|
8
|
10
|
SPACES
|
|
NUMBER OF RECORDS
|
18
|
8
|
99999999 Numeric – right justified – zero filled
|
|
Filler
|
26
|
10
|
SPACES
|
|
Filler
|
36
|
3
|
“230” (Numeric Constant)
|
|
Filler
|
39
|
191
|
SPACES
|
|
230
|
*** NUMBER OF RECORDS FILED CONTAINS THE SUM OF THE NUMBER OF RECORDS IN THE FILE NOR INCLUDING THE TRAILER.

Carrier to ASES Data Submissions
New File Layouts
Version 2
October 30, 2013
John Rullan: jrullan@asespr.org
Angie Rivera: aavila@asespr.org
David Cusick: david.cusick@milliman.com
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
Carrier to ASES Data Submissions
File Layouts
TABLE OF CONTENTS
|
Version Changes
|
4
|
|
NOTES
|
6
|
|
Changes and Additions in Data File Layouts
|
11
|
|
File Naming Convention
|
13
|
|
CLAIMSERVICE INPUT FILE LAYOUT
|
15
|
|
PROVIDERS INPUT FILE LAYOUT
|
35
|
|
IPA INPUT FILE LAYOUT
|
38
|
|
CAPITATION INPUT FILE LAYOUT
|
40
|
|
ATTACHMENT I – Municipality Codes
|
43
|
|
ATTACHMENT II – Carrier Codes
|
47
|
|
ATTACHMENT III – Specialty Codes
|
49
|
|
ATTACHMENT IV – Place of Service Codes
|
55
|
|
ATTACHMENT V – Provider Type Codes
|
61
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
Carrier to ASES Data Submissions
File Layouts
Version Changes
Version 2.0
CLAIMS Input File Layout - Removed
Removed
SERVICES Input File Layout - Removed
Removed
CLAIMSERVICE Input File Layout – Added
New layout that combines claims and services into one file.
New fields added to layout.
Filename Change
Filenames for all data files have been changed to change 1 digit year to 2 digit year and to remove the region form the filename.
Data Validation Change
File will no longer be tested individual field validations. Data submitted must reconcile to control totals provided by carriers and will undergo a data audit to test for data quality issues. Monthly files that do not pass the reconciliation process and the data audit process will be rejected.
Version 2.0
Last Update February: 7, 2013
NOTES
Changes and Additions in Data File Layouts
ASES new file layouts for submission by Carriers for data generated from April, 2013 forward.
CLAIMS AND SERVICES FILES -
In the previous version of the layouts, a CLAIMS file and a detailed SERVICE file were used to represent claims. In version 2.0 of the layouts, only one file exists for both the header and detail portions of the claim, now referred to as the CLAIMSERVICES file. Records in the CLAlMSERVICES file have both the header and detail portions of the claim.
In addition to the merging of CLAIMS and SERVICES, a number of changes to the claims data layout have occurred, including the transaction processing methodology and the addition of several fields. Read carefully the section below on transaction handling for claims data.
CLAIMS TRANSACTION HANDLING -
The previous method of handling claim adjustments and reversals used a transaction code to completely replace a claim with a new claim. This method has been changed in this version of the layouts to more closely match existing claims processing systems and allow for adjustments to occur on different dates. All adjustments of an adjudicated claim line are now accepted in the CLAIMSERVICES file. Do not send claims that are in an open status, such as pended claims, held, rejected, or pre-adjudicated claims. Claims reversals and adjustments happen as follows:
Paid or Denied FFS Claims
Individual service lines are adjusted at the line level with additional adjustment services marked with a claim line status code of 'A', while the original claim has a status code of 'P' for paid, 'D' for denied claims, or 'E' for encounter claims. The adjustment service may have the same claim ID and line number, may have the same claim ID and a different line number, or may be provided with a different claim ID and different line number.
Encounter Claims
Claims representing encounters have no allowed or paid amounts and are therefore not able to be adjusted monetarily. If an encounter needs to be updated to change any of the fields of the encounter, the adjusting claim must have a claim status code of 'E' and the claim ID and service line number must be the same as the encounter being adjusted. Our process will remove the original encounter so that duplicate encounters will not be counted in the data.
PROVIDER IDS -
ASES is requesting that provider NPIs are populated in the NPI field on provider records to assist in provider attribution and rep01ting across all Carriers. ASES will still accept the carrier's own provider id as the provider ID for medical claims, but the additional field NPI should be populated.
For pharmacy claims only
For pharmacy providers, only the NCPDP/NABP number is accepted. Carriers must include pharmacy providers in their provider files sent to ASES and the IDs must be consistent within the carriers claims. In other words, a pharmacy should have one NCPDP/NABP number and that number should as the billing provider be used for all claims from that pharmacy. When claims are validated, the Billing Provider on the claim record will be validated against the Provider file and will be matched even if the provider is unique for the carrier.
PROVIDER TELEPHONE NUMBERS -
Provider Telephone remains a required field on the Provider Input Layout. In the event, and as an exception, if the carrier does not have the actual provider' s telephone number they should insert their own (Carrier's) telephone number. This also applies to the IPA Work Phone field in the same way.
Note that all telephone number fields must be filled using only numbers. No spaces or 0- characters should be included. For example, the telephone number (939) 123-4567 will be coded in the data field as 9391234567
CAPITATION AMOUNT -
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
The amount to be reported on capitation records must be a net amount that represents any costs associated with providing services which are not repo1ted in claims and encounters. This may come from fonnal contracts with providers such as HCO/PCPs, or any other financial arrangement or allocation of costs.
The number should represent a calculation which includes the earned capitation for the period less claims paid amounts, if any, chargeable against the provider risk. Other types of deductions which may be taken out of the provider's payment such as repayment of advances, retentions for reserves should not be included in the calculation.
CAPITATION ADJUSTMENTS -
There may be circumstances in which capitation payments which have already been reported, need in a later month to be adjusted or even reversed . To accomplish this, the Capitation records will behave differently than Claims and Services. The carrier will send a new record for the provider I member I experience date with an amount to be added or subtracted from the previously reported amount. Ifa capitation of $10.00 is to be reversed then the new record should contain the same information as the original but with a new Capitation Date and a Capitation Amount of -$10.00. Inside Medlnsight the capitation for that Provider I Member for that particular date will be the aggregate of all the records and this example will result in $0.00.
Note that, as Capitation net amounts for any particular record may be negative, a reversal in such a case would be a positive amount.
DATA VALIDATION PROCESS -
All files will pass through a validation process. Validation will check the basic structure of the file and its records and may result in a file being rejected . Such rejections may be caused for example, by file names which fail to follow the naming convention, a file containing wrong length records or other basic tests.
All files which are rejected will be notified to the carrier with an explanation of why the file is rejected. No records from such a file will be retained in the system and the carrier will be required to re-submit the rejected file in its entirety before the next months files become due. Such re-submitted files must be carefully named using the sequence number part of the naming convention to ensure the
name is distinct from the rejected file and is named in the correct order.
CLAIMS AND CAPITATION LAG REPORTS -
Carriers are required to submit claims and capitation payment reports, called lag reports, on a monthly basis. These reports will be used to reconcile the data submitted. Data that does not match the lag reports on paid amount within a reasonable percentage will be deemed invalid and must be corrected. The lag reports submitted by the carrier will be considered to be financially accurate and may be used for other purposes, including negotiations or other financial analyses. Therefore, it is in the carrier's best interests to produce lag reports that are either from another source that the actual files that are submitted, or to verify that the lag reports tie to financial reports.
The required claims lag reports need to be an excel file with the following characteristics:
1. Claims paid amounts by region, incurred month, paid month, and claim type (medical, pharmacy and dental) for claims.
2. The report must include at least all paid and incurred months going back 2 full years prior to the month the report is run.
An example of how the claims lag report data should look for claims is as follows:
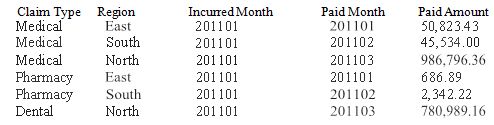
The required capitation lag reports need to be an excel file with the following characteristics:
1. Capitation paid amounts by region, capitation experience month, paid month
2. The report must include at least all paid and experience months going back 2 full years prior to the month the report is run.
An example of how the capitation lag report data should look for claims is as follows:
|
Region
|
Incurred Month
|
Paid Month
|
Capitation Paid Amount
|
|||||||||
|
East
|
201101
|
201101
|
5,023.43
|
|||||||||
|
South
|
201101
|
201102
|
4,534.00
|
|||||||||
|
North
|
201101
|
201103
|
98,796.36
|
|||||||||
|
201101
|
201101
|
66.89
|
||||||||||
|
201101
|
201102
|
242.22
|
||||||||||
|
201101
|
201103
|
70,989.16
|
||||||||||
PRIMARY CARRIER ID -
A field for the Primary Carrier ID has been added to the Claims Input Layout to recognize the MCO or TPA which enrolls the member and assigns IPA and PCP Provider IDs. The Carrier ID filed will can)' the ID of the carrier generating the Claims Input File. These files will contain the same value when the reporting carrier is an MCO or TPA. When the reporting carrier is an MBHO or PBM the CaITier ID will contain the code of the MBHO or PBM and the Primary Carrier ID will contain the code of the MCO or TPA of the member.
IPA CODES AND PROVIDER CODES -
The Primary Carrier ID field has been added to be able to distinguish the validation of IPA and Provider codes by carrier. The Primary Carrier ID will carry the code of the MCO or TPA which contracts the members IPA and PCP Provider. In Claims records the codes for IPA and PCP Provider will be those created by the MCO/TPA and delivered to the MBHOs and PBMs in eligibility/enrollment data exchanges.
ATIENDING PROVIDER -
The validation rules for Attending Provider have been changes to remove the requirement that the value match a valid provider (i.e. a provider code reported by the carrier in its Provider file. The field is still required.
MUNICIPALITY SERVICE -
Recognizing that claims may be processed for services outside of Puerto Rico, code 0666 is included in the list of Municipality Codes. This value is valid only for use in the field Municipality Service on the Claims Input File. This value should be used only when services are paid for outside of Puerto Rico.
General Notes on data layout requirements
Date Fields - All date fields in the following data layout are defined to the same size and format as YYYYMMDD. An 8 byte field where YYYY = 4 digit year, MM = 2 digit month and DD = 2 digit day. 1 digit month and day values must always have the leading zero (0). Date fields must contain a valid date with months between 01 and 12 and days between 01 and maximum day in month. July
1, 2006 will be coded as 20060701.
Amount Fields - All amount fields representing money must be numeric and are defined as 9 bytes in the format 9(7)v99 where v represents and implied decimal point. This allows a maximum of 7 digits for dollars plus the last two digits for cents. These numbers are always right justified and zero filled to the left. As examples:
$1.23 will be coded as 000000123
$100.00 will be coded as 000010000
All amount fields are positive and follow the above definition unless clearly specified otherwise. End of Record Filler -All file layouts have been designed to end with a filler field of 1 byte which must always be coded as an "*" character. This is done to avoid issues between different systems when generating and transferring ASCII files in which ending field may be empty. The fixed End of Record Filler guarantees that all records in a file can be constructed to the fixed length format as defined in the layouts.
Justification and filling of Fields -The layouts have all been specified to provide fixed length fields and fixed length records. While other methods can be used, it is felt that this provides the best common ground for working with multiple entities each of which uses varying systems. To be sure everyone understands the same about the comments on justification and filling the following examples are given to help keep this concept clear.
All numeric fields must be filled completely with numeric digits. If there are exceptions these are clearly spelled out in the documentation of the layouts. Typically numeric field are right justified and to keep them numeric must be zero filled. In a field specified as numeric such a 9(7)v99 where v represents an implied decimal the following examples illustrate how data will look in the field:
|
Value
|
Field
|
|||
|
12.50
|
000001250
|
|||
|
101
|
000010100
|
|||
|
1,234 .56
|
000123456
|
|||
|
1,000,000
|
100000000
|
|||
All alphanumeric fields must be filled completely. If the value of data in the field is less than the width of the field then care must be taken to ensure that the field is filled with blanks. Allowing "NULLS" or other special characters through may cause unexpected results and make reading, loading and validation of the data difficult. Typically alphanumeric field are left justified and filled to the right with blanks to complete the field. In a field specified as alphanumeric such a X(20) the following examples illustrate how data will look in the field where the [] characters represent the start and end of the field -
|
Value
|
Field
|
|
|
P.R.
|
[P.R.
|
] |
|
Jose Rivera
|
[José Rivera
|
] |
|
Blanks
|
[
|
] |
|
(Metro-North Region)
|
[(Metro-No rth Region)
|
] |
References to CMS 1500 and UB-92 - All references to CMS 1500 or UB-92 in this document are for convenience and correspond equally to equivalent electronic formats and will apply equally to the next version of CMS 1500 or the UB-04 when implemented.
MPI Number fields - In all files in which MPI Number is required, carriers should code all 9s if the MPI is unknown. This should not be true for any current beneficiary. This exception will continue until such time as ASES determines that the issue of MPI being unavailable has disappeared from historical data. For Government Employee MPI should be filled with Contract Number.
File Naming Convention
All files to be delivered to ASES by the carriers must follow the naming conventions below. Files which do not fit the naming convention will be ignored and the canier deemed to have failed in delivery of such a file.
File names must adhere strictly to this naming convention as the structure includes information for identification of the carrier, dates and file type. Ifnot named correctly the file cannot be processed properly.
The general format of file names will be -

Examples of completing this naming conversion are =
For imaginary carrier 96 in the files for ClaimServices and payment in April 2013 will be named as follows:

When the Capitation file is rejected, the corrected file will be re-submitted as 09612041.CAP



























PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENT I - MUNICIPALITY CODES
|
Alphabetical by Municipality
|
|
Ordered By Code
|
||||
|
MUNICIPALITY
|
REGION
|
CODE
|
CODE
|
MUNICIPALITY
|
REGION
|
|
|
Adjuntas
|
S
|
0004
|
|
0004
|
Adjuntas
|
S
|
|
Aguada
|
Z
|
0008
|
|
0008
|
Aguada
|
Z
|
|
Aguadilla
|
Z
|
0012
|
|
0012
|
Aguadilla
|
Z
|
|
Aguas Buenas
|
E
|
0016
|
|
0016
|
Aguas Buenas
|
E
|
|
Aibonito
|
G
|
0020
|
|
0020
|
Aibonito
|
G
|
|
Anasco
|
Z
|
0024
|
|
0024
|
Anasco
|
Z
|
|
Arecibo
|
A
|
0028
|
|
0028
|
Arecibo
|
A
|
|
Arroyo
|
G
|
0032
|
|
0032
|
Arroyo
|
G
|
|
Barceloneta
|
A
|
0036
|
|
0036
|
Barceloneta
|
A
|
|
Barranquitas
|
G
|
0040
|
|
0040
|
Barranquitas
|
G
|
|
Bayamon
|
B
|
0044
|
|
0044
|
Bayamon
|
B
|
|
Cabo Rojo
|
Z
|
0048
|
|
0048
|
Cabo Rojo
|
Z
|
|
Caguas
|
E
|
0052
|
|
0052
|
Caguas
|
E
|
|
Camuy
|
A
|
0056
|
|
0056
|
Camuy
|
A
|
|
Canovanas
|
F
|
0060
|
|
0060
|
Canovanas
|
F
|
|
Carolina
|
F
|
0064
|
|
0064
|
Carolina
|
F
|
|
Catano
|
B
|
0068
|
|
0068
|
Catano
|
B
|
|
Cayey
|
E
|
0072
|
|
0072
|
Cayey
|
E
|
|
Ceiba
|
F
|
0076
|
|
0076
|
Ceiba
|
F
|
|
Ciales
|
A
|
0080
|
|
0080
|
Ciales
|
A
|
|
Cidra
|
E
|
0084
|
|
0084
|
Cidra
|
E
|
|
Coamo
|
G
|
0088
|
|
0088
|
Coamo
|
G
|
|
Comerio
|
B
|
0092
|
|
0092
|
Comerio
|
B
|
|
Corozal
|
B
|
0096
|
0096
|
Corozal
|
B
|
|
|
Culebra
|
F
|
0100
|
0100
|
Culebra
|
F
|
|
|
Dorado
|
B
|
0104
|
0104
|
Dorado
|
B
|
|
|
Fajardo
|
F
|
0108
|
0108
|
Fajardo
|
F
|
|
|
Florida
|
A
|
0112
|
0112
|
Florida
|
A
|
|
|
Guanica
|
S
|
0116
|
0116
|
Guanica
|
S
|
|
|
Guayama
|
G
|
0120
|
0120
|
Guayama
|
G
|
|
|
Guayanilla
|
S
|
0124
|
0124
|
Guayanilla
|
S
|
|
|
Guaynabo
|
B
|
0128
|
0128
|
Guaynabo
|
B
|
|
|
Gurabo
|
E
|
0132
|
0132
|
Gurabo
|
E
|
|
|
Hatillo
|
A
|
0136
|
0136
|
Hatillo
|
A
|
|
|
Hormigueros
|
Z
|
0140
|
0140
|
Hormigueros
|
Z
|
|
|
Humacao
|
E
|
0144
|
0144
|
Humacao
|
E
|
|
|
Isabela
|
Z
|
0148
|
0148
|
Isabela
|
Z
|
|
|
Jayuya
|
S
|
0152
|
0152
|
Jayuya
|
S
|
|
|
Juana Diaz
|
G
|
0156
|
0156
|
Juana Diaz
|
G
|
|
|
Juncos
|
E
|
0160
|
0160
|
Juncos
|
E
|
|
|
Lajas
|
Z
|
0164
|
0164
|
Lajas
|
Z
|
|
|
Lares
|
A
|
0168
|
0168
|
Lares
|
A
|
|
|
Las Marias
|
Z
|
0172
|
0172
|
Las Marias
|
Z
|
|
|
Las Piedras
|
E
|
0176
|
0176
|
Las Piedras
|
E
|
|
|
Loiza
|
F
|
0180
|
0180
|
Loiza
|
F
|
|
|
Luquillo
|
F
|
0184
|
0184
|
Luquillo
|
F
|
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTI - MUNICIPALITY CODES
|
Alphabetical by Municipality
|
|
Ordered By Code
|
||||
|
MUNICIPALITY
|
REGION
|
CODE
|
|
CODE
|
MUNICIPALITY
|
REGION
|
|
Manati
|
A
|
0188
|
|
0188
|
Manati
|
A
|
|
Maricao
|
Z
|
0192
|
|
0192
|
Maricao
|
Z
|
|
Maunabo
|
G
|
0196
|
|
0196
|
Maunabo
|
G
|
|
Mayaguez
|
Z
|
0200
|
|
0200
|
Mayaguez
|
Z
|
|
Moca
|
Z
|
0204
|
|
0204
|
Moca
|
Z
|
|
Morovisl
|
A
|
0208
|
|
0208
|
Morovis
|
A
|
|
Naguabo
|
E
|
0212
|
|
0212
|
Naguabo
|
E
|
|
Naranjito
|
B
|
0216
|
|
0216
|
Naranjito
|
B
|
|
Orocovis
|
G
|
0220
|
|
0220
|
Orocovis
|
G
|
|
Patillas
|
G
|
0224
|
|
0224
|
Patillas
|
G
|
|
Penuelas
|
S
|
0228
|
|
0228
|
Penuelas
|
S
|
|
Ponce
|
S
|
0232
|
|
0232
|
Ponce
|
S
|
|
Puerta de Tierra
|
J
|
0264
|
|
0236
|
Quebradillas
|
A
|
|
Puerto Nuevo
|
J
|
0270
|
|
0240
|
Rincon
|
Z
|
|
Quebradillas
|
A
|
0236
|
|
0244
|
Rio Grande
|
F
|
|
Rincon
|
Z
|
0240
|
|
0248
|
Sabana Grande
|
Z
|
|
Rio Grande
|
F
|
0244
|
|
0252
|
Salinas
|
G
|
|
Rio Piedras
|
J
|
0272
|
|
0256
|
San German
|
Z
|
|
Sabana Grande
|
Z
|
0248
|
|
0264
|
Puerta de Tierra
|
|
|
Salinas
|
G
|
0252
|
|
0266
|
San Juan
|
|
|
San German
|
Z
|
0256
|
|
0270
|
Puerto Nuevo
|
|
|
San Jose
|
J
|
0274
|
|
0272
|
Rio Piedras
|
|
|
San Juan
|
J
|
0266
|
|
0274
|
San Jose
|
|
|
San Lorenzo
|
E
|
0276
|
|
0276
|
San Lorenzo
|
E
|
|
San Sebastian
|
Z
|
0280
|
|
0280
|
San Sebastian
|
Z
|
|
Santa Isabel
|
G
|
0284
|
|
0284
|
Santa Isabel
|
G
|
|
Toa Alta
|
B
|
0288
|
|
0288
|
Toa Alta
|
B
|
|
Toa Baja
|
B
|
0292
|
|
0292
|
Toa Baja
|
B
|
|
Trujillo Alto
|
F
|
0296
|
|
0296
|
Trujillo Alto
|
F
|
|
Utuado
|
A
|
0300
|
|
0300
|
Utuado
|
A
|
|
Vega Alta
|
B
|
0304
|
|
0304
|
Vega Alta
|
B
|
|
Vega Baja
|
A
|
0308
|
|
0308
|
Vega Baja
|
A
|
|
Vieques
|
F
|
0312
|
|
0312
|
Vieques
|
F
|
|
Villalba
|
G
|
0316
|
|
0316
|
Villalba
|
G
|
|
Yabucoa
|
E
|
0320
|
|
0320
|
Yabucoa
|
E
|
|
Yauco
|
S
|
0324
|
|
0324
|
Yauco
|
S
|
|
Outside Puerto Rico
|
--
|
0666
|
*
|
0666
|
Outside Puerto Rico
|
--
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTII - CARRIER CODES
|
CODE
|
Carrier
|
Type
|
|
01
|
Triple S
|
MCO
|
|
03
|
(discontinued)
|
MCO
|
|
02
|
Humana
|
MCO
|
|
17
|
MCS
|
MCO
|
|
25
|
(discontinued)
|
MCO
|
|
27
|
MCS Life
|
Medicare Platino
|
|
28
|
Red Medica
|
Medicare Platino
|
|
29
|
Medicare y Mucho Mas
|
Medicare Platino
|
|
31
|
Triple S
|
Medicare Platino
|
|
33
|
Preferred Medicare Choice
|
Medicare Platino
|
|
34
|
MCS Advantage
|
Medicare Platino
|
|
35
|
COSVIMed
|
Medicare Platino
|
|
37
|
Salud Dorada con Medicare
|
Medicare Platino
|
|
39
|
MAPFRE
|
Medicare Platino
|
|
41
|
Health Medicare Ultra
|
Medicare Platino
|
|
42
|
Humana
|
Medicare Platino
|
|
44
|
Auxilio Platino
|
Medicare Platino
|
|
47
|
American Health
|
Medicare Platino
|
|
49
|
FirstPlus
|
Medicare Platino
|
|
51
|
Triple S
|
TPA - Direct Contract
|
|
52
|
Humana
|
TPA - Direct Contract
|
|
53
|
MCS
|
TPA - Direct Contract
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENT II - CARRIER CODES
|
CODE
|
Carrier
|
Type
|
|
54
|
Triple S
|
TPA - Direct Contract
|
|
55
|
COSVI
|
TPA - Direct Contract
|
|
60
|
Caremark
|
PBM
|
|
64
|
MC-21
|
PBM
|
|
70
|
ASSMCA
|
Mental Health Pilot
|
|
71
|
Plan de Salud Hospital Menonita
|
Government Employee
|
|
72
|
MMM Healthcare,INC
|
Government Employee
|
|
73
|
National Life Insurance Company
|
Government Employee
|
|
74
|
Ryder Health Plan, Inc.
|
Government Employee
|
|
75
|
Triple-S Salud Inc.
|
Government Employee
|
|
76
|
(discontinued)
|
MBHO
|
|
77
|
Humana Health Plan of Puerto Rico, Inc.
|
Government Employee
|
|
78
|
Humana Insurance of Puerto Rico,Inc.
|
Government Employee
|
|
79
|
MCS Advantage,Inc.
|
Government Employee
|
|
80
|
MCS Life Insurance Company
|
Government Employee
|
|
81
|
Asociacion de Maestros de Puerto Rico
|
Government Employee
|
|
82
|
First Medical Health Plan, Inc.
|
Government Employee
|
|
83
|
APS
|
MBHO
|
|
95
|
FHC
|
MBHO
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIII - SPECIALTY CODES
|
CODE
|
Specialty
|
|
Codes included in this table are designed for completeness and in no way imply coverage of services under the Government Health Insurance Plan
|
|
|
01
|
General Practice
|
|
02
|
General Surgery
|
|
03
|
Allergy/Immunology
|
|
04
|
Otolaryngology
|
|
05
|
Anesthesiology
|
|
06
|
Cardiology
|
|
07
|
Dermatology
|
|
08
|
Family Practice
|
|
09
|
Interventional Pain Management
|
|
10
|
Gastroenterology
|
|
11
|
Internal Medicine
|
|
12
|
Osteopathic Manipulative Therapy
|
|
13
|
Neurology
|
|
14
|
Neurosurgery
|
|
16
|
Obstetrics / Gynecology
|
|
18
|
Ophthalmology
|
|
19
|
Oral Surgery
|
|
20
|
Orthopedic Surgery
|
|
22
|
Pathology
|
|
24
|
Plastic and Reconstructive Surgery
|
|
25
|
Physical Medicine / Rehabilitation
|
|
26
|
Psychiatry
|
|
28
|
Colorectal Surgery (Formerly Proctology)
|
|
29
|
Pulmonary Diseases
|
|
30
|
Diagnostic Radiology
|
|
32
|
Anesthesiologist Assistant
|
|
33
|
Thoracic Surgery
|
|
34
|
Urology
|
|
35
|
Chiropractic
|
|
36
|
Nuclear Medicine
|
|
37
|
Pediatric Medicine
|
|
38
|
Geriatric Medicine
|
|
39
|
Nephrology
|
|
40
|
Hand Surgery
|
|
41
|
Optometry
|
|
42
|
Certified Nurse Midwife
|
|
43
|
Certified Registered Nurse Assistant (CRNA)
|
|
44
|
Infectious Disease
|
|
45
|
Mammography Screening Center
|
|
46
|
Endocrinology
|
|
47
|
Independent Diagnostics Testing Facility
|
|
48
|
Podiatry
|
|
49
|
Ambulatory Surgical Center
|
|
50
|
Nurse Practitioner
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENT III - SPECIALTY CODES
|
CODE
|
Specialty
|
|
51
|
Medical Supply Company with Orthotist
|
|
52
|
Medical Supply Company with Prosthetist
|
|
53
|
Medical Supply Company with Orthotist-Prosthetist
|
|
54
|
Other Medical Supply Company
|
|
55
|
Individual Certified Orthotist
|
|
56
|
Individual Certified Prosthetist
|
|
57
|
Individual Certified Orthotist-Prosthetist
|
|
58
|
Medical Supply Company with pharmacist
|
|
59
|
Ambulance Service Provider
|
|
60
|
Public Health and Welfare Agency
|
|
61
|
Voluntary Health or Charitable Agency
|
|
62
|
Psychologist
|
|
63
|
Portable X-ray Supplier
|
|
64
|
Audiologist
|
|
65
|
Physical Therapist
|
|
66
|
Rheumatology
|
|
67
|
Occupational Therapy
|
|
68
|
Clinical Psychologist
|
|
69
|
Clinical Laboratory
|
|
70
|
Multi-Specialty Clinic or Group Practice
|
|
71
|
Registered Dietician / Nutritional Professional
|
|
72
|
Pain Management
|
|
73
|
Mass Immunization Roster Billers
|
|
74
|
Radiation Therapy Center
|
|
75
|
Slide Preparation Facilities
|
|
76
|
Peripheral Vascular Disease
|
|
77
|
Vascular Surgery
|
|
78
|
Cardiac Surgery
|
|
79
|
Addiction Medicine
|
|
80
|
Licensed Clinical Social Worker
|
|
81
|
Critical Care (Intensivists)
|
|
82
|
Hematology
|
|
83
|
Hematology / Oncology
|
|
84
|
Preventive Medicine
|
|
85
|
Maxillofacial Surgery
|
|
86
|
Neuropsychiatry
|
|
87
|
All Other Suppliers
|
|
88
|
Unknown Supplier / Provider Specialty
|
|
89
|
Certified Clinical Nurse Specialist
|
|
90
|
Medical Oncology
|
|
91
|
Surgical Oncology
|
|
92
|
Radiation Oncology
|
|
93
|
Emergency Medicine
|
|
94
|
Intervention Radiology
|
|
96
|
Optician
|
|
97
|
Physician Assistant
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENT III - SPECIALTY CODES
|
CODE
|
Specialty
|
|
98
|
Gynecological Oncology
|
|
99
|
Unknown Physician Specialty
|
|
A1
|
Skilled Nursing Facility
|
|
A2
|
Intermediate Care Nursing Facility
|
|
A3
|
Other Nursing Facility
|
|
A4
|
Home Health Agency
|
|
A5
|
Pharmacy
|
|
A6
|
Medical Supply Company with Respiratory Therapist
|
|
A7
|
Department Store
|
|
A8
|
Grocery Store
|
|
DD
|
Dentist
|
|
EN
|
Endodontist
|
|
HE
|
Health Educator
|
|
HN
|
Home Health Nurse
|
|
PE
|
Periodontist
|
|
RT
|
Respiratory Therapist
|
|
ST
|
Speech Therapist
|
|
BB
|
Blood Bank
|
|
CV
|
Cardiac Catheterization Facility
|
|
DF
|
Dialysis Facility
|
|
EC
|
Emergency Care Facility
|
|
HV
|
HIV Ambulatory Antibiotic Facility
|
|
HO
|
Hospice
|
|
IC
|
Intensive Care Unit
|
|
IT
|
Infusion Therapy
|
|
LI
|
Lithotripsy
|
|
NI
|
Neonatal ICU
|
|
OP
|
Optical
|
|
PC
|
Clinic - Primary Level
|
|
PH
|
Private Hospital
|
|
PP
|
Private Psychiatric Hospital
|
|
PS
|
Psychiatric Partial Hospital
|
|
SH
|
State Hospital
|
|
SP
|
State Psychiatric Hospital
|
|
XR
|
X-ray Facility
|
|
Z4
|
Cardiovascular Surgery Program
|
|
O1
|
Occupational Medicine
|
|
P1
|
Perinatology
|
|
N1
|
Neonatolgy
|
|
G1
|
Geneticist
|
|
P2
|
Pediatric Surgery
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIV - PLACE OF SERVICE CODES
|
CODE
|
Name
|
Description
|
|
Codes included in this table are designed for completeness and in no way imply coverage of services under the Government Health Insurance Plan
|
||
|
01
|
Pharmacy
|
A facility or location where drugs and other medically related items and services are sold, dispensed, or otherwise provided directly to patients.
|
|
02
|
Unassigned
|
N/A
|
|
03
|
School
|
A facility whose primary purpose is education.
|
|
04
|
Homeless Shelter
|
A facility or location whose primary purpose is to provide temporary housing to homeless individuals.
|
|
05
|
Indian Health Service Free-standing Facility
|
A facility or location, owned and operated by the Indian Health Service, which provides diagnostic, therapeutic (surgical and non-surgical), and rehabilitation services to American Indians and Alaska Natives who do not require hospitalization
|
|
06
|
Indian Health Service Provider-based Facility
|
A facility or location, owned and operated by the Indian Health Service, which provides diagnostic, therapeutic (surgical and non-surgical), and rehabilitation services rendered by, or under the supervision of, physicians to American Indians and Alaska Natives admitted as inpatients or outpatients.
|
|
07
|
Tribal 638 Free-standing Facility
|
A facility or location owned and operated by a federally recognized American Indian or Alaska Native tribe or tribal organization under a 638 agreement, which provides diagnostic, therapeutic (surgical and nonsurgical), and rehabilitation services to tribal members who do not require hospitalization.
|
|
08
|
Tribal 638 Provider-based Facility
|
A facility or location owned and operated by a federally recognized American Indian or Alaska Native tribe or tribal organization under a 638 agreement, which provides diagnostic, therapeutic (surgical and nonsurgical), and rehabilitation services to tribal members admitted as inpatients or outpatients.
|
|
09-10
|
Unassigned
|
N/A
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIV - PLACE OF SERVICE CODES
|
CODE
|
Name
|
Description
|
|
11
|
Office
|
Location, other than a hospital, Skilled Nursing Facility (SNF), military treatment facility, community health center, State or local public health clinic, or Intermediate Care Facility (ICF), where the health professional routinely provides health examinations, diagnosis, and treatment of illness or injury on an ambulatory basis.
|
|
12
|
Home
|
Location, other than a hospital or other facility, where the patient receives care in a private residence.
|
|
13
|
Assisted Living Facility
|
Congregate residential facility with self-contained living units providing assessment of each resident's needs and on-site support 24 hours a day, 7 days a week, with the capacity to deliver or arrange for services including some health care and other services.
|
|
14
|
Group Home
|
A residence, with shared living areas, where clients receive supervision and other services such as social and/or behavioral services, custodial service, and minimal services.
|
|
15
|
Mobile Unit
|
A facility/unit that moves from place-to-place equipped to provide preventive, screening, diagnostic, and/or treatment services.
|
|
16-19
|
Unassigned
|
N/A
|
|
20
|
Urgent Care Facility
|
Location, distinct from a hospital emergency room, an office, or a clinic, whose purpose is to diagnose and treat illness or injury for unscheduled ambulatory patients seeking immediate medical attention.
|
|
21
|
Inpatient Hospital
|
A facility, other than psychiatric, which primarily provides diagnostic, therapeutic (both surgical and nonsurgical), and rehabilitation services by, or under, the supervision of physicians to patients admitted for a variety of medical conditions.
|
|
22
|
Outpatient Hospital
|
A portion of a hospital, which provides diagnostic, therapeutic (both surgical and nonsurgical), and rehabilitation services to sick or injured persons who do not require hospitalization or institutionalization.
|
|
23
|
Emergency Room - Hospital
|
A portion of a hospital where emergency diagnosis and treatment of illness or injury is provided.
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIV - PLACE OF SERVICE CODES
|
CODE
|
Name
|
Description
|
|
24
|
Ambulatory Surgical Center
|
A freestanding facility, other than a physician's office, where surgical and diagnostic services are provided on an ambulatory basis.
|
|
25
|
Birthing Center
|
A facility, other than a hospital's maternity facilities or a physician's office, which provides a setting for labor, delivery, and immediate post-partum care as well as immediate care of newborn infants.
|
|
26
|
Military Treatment Facility
|
A medical facility operated by one or more of the Uniformed Services. Military Treatment Facility (MTF) also refers to certain former U.S. Public Health Service (USPHS) facilities now designated as Uniformed Service Treatment Facilities (USTF).
|
|
27-30
|
Unassigned
|
N/A
|
|
31
|
Skilled Nursing Facility
|
A facility, which primarily provides inpatient skilled nursing care and related services to patients who require medical, nursing, or rehabilitative services but does not provide the level of care or treatment available in a hospital.
|
|
32
|
Nursing Facility
|
A facility which primarily provides to residents skilled nursing care and related services for the rehabilitation of injured, disabled, or sick persons, or, on a regular basis, health-related care services above the level of custodial care to other than mentally retarded individuals.
|
|
33
|
Custodial Care Facility
|
A facility which provides room, board and other personal assistance services, generally on a long-term basis, and which does not include a medical component.
|
|
34
|
Hospice
|
A facility, other than a patient's home, in which palliative and supportive care for terminally ill patients and their families are provided.
|
|
35-40
|
Unassigned
|
N/A
|
|
41
|
Ambulance - Land
|
A land vehicle specifically designed, equipped and staffed for lifesaving and transporting the sick or injured.
|
|
42
|
Ambulance - Air or Water
|
An air or water vehicle specifically designed, equipped and staffed for lifesaving and transporting the sick or injured.
|
|
43-48
|
Unassigned
|
N/A
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIV - PLACE OF SERVICE CODES
|
CODE
|
Name
|
Description
|
|
49
|
Independent Clinic
|
A location, not part of a hospital and not described by any other Place of Service code, that is organized and operated to provide preventive, diagnostic, therapeutic, rehabilitative, or palliative services to outpatients only.
|
|
50
|
Federally Qualified Health Center
|
A facility located in a medically underserved area that provides Medicare beneficiaries preventive primary medical care under the general direction of a physician.
|
|
51
|
Inpatient Psychiatric Facility
|
A facility that provides inpatient psychiatric services for the diagnosis and treatment of mental illness on a 24-hour basis, by or under the supervision of a physician.
|
|
52
|
Psychiatric Facility Partial Hospitalization
|
A facility for the diagnosis and treatment of mental illness that provides a planned therapeutic program for patients who do not require full time hospitalization, but who need broader programs than are possible from outpatient visits to a hospital-based or hospital-affiliated facility.
|
|
53
|
Community Mental Health Center
|
A facility that provides the following services:
• Outpatient services, including specialized outpatient services for children, the elderly, individuals who are chronically ill, and residents of the CMHC's mental health services area who have been discharged from inpatient treatment at a mental health facility.
• 24 hour a day emergency cares services.
• Day treatment, other partial hospitalization services, or psychosocial rehabilitation services.
• Screening for patients being considered for admission to State mental health facilities to determine the appropriateness of such admission.
• Consultation and education services.
|
|
54
|
Intermediate Care Facility/Mentally Retarded
|
A facility which primarily provides health-related care and services above the level of custodial care to mentally retarded individuals but does not provide the level of care or treatment available in a hospital or SNF.
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIV - PLACE OF SERVICE CODES
|
CODE
|
Name
|
Description
|
|
55
|
Residential Substance Abuse Treatment Facility
|
A facility, which provides treatment for substance (alcohol and drug) abuse to live-in residents who, does not require acute medical care. Services include individual and group therapy and counseling, family counseling, laboratory tests, drugs and supplies, psychological testing, and room and board.
|
|
56
|
Psychiatric Residential Treatment Center
|
A facility or distinct part of a facility for psychiatric care, which provides a total 24-hour therapeutically, planned and professionally staffed group living and learning environment.
|
|
57
|
Non-residential Substance Abuse Treatment Facility
|
A location which provides treatment for substance (alcohol and drug) abuse on an ambulatory basis. Services include individual and group therapy and counseling, family counseling, laboratory tests, drugs and supplies, and psychological testing.
|
|
58-59
|
Unassigned
|
N/A
|
|
60
|
Mass Immunization Center
|
A location where providers administer pneumococcal pneumonia and influenza virus vaccinations and submit these services as electronic media claims, paper claims, or using the roster billing method. This generally takes place in a mass immunization setting, such as, a public health center, pharmacy, or mall but may include a physician office setting.
|
|
61
|
Comprehensive Inpatient Rehabilitation Facility
|
A facility that provides comprehensive rehabilitation services under the supervision of a physician to inpatients with physical disabilities. Services include physical therapy, occupational therapy, speech pathology, social or psychological services, and orthotics and prosthetics services.
|
|
62
|
Comprehensive Outpatient Rehabilitation Facility
|
A facility that provides comprehensive rehabilitation services under the supervision of a physician to outpatients with physical disabilities. Services include physical therapy, occupational therapy, and speech pathology services.
|
|
63-64
|
Unassigned
|
N/A
|
|
65
|
End-Stage Renal Disease Treatment Facility
|
A facility other than a hospital, which provides dialysis treatment, maintenance, and/or training to patients or caregivers on an ambulatory or home-care basis.
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
ATTACHMENTIV - PLACE OF SERVICE CODES
|
CODE
|
Name
|
Description
|
|
66-70
|
Unassigned
|
N/A
|
|
71
|
State or Local Public Health Clinic
|
A facility maintained by either State or local health departments that provide ambulatory primary medical care under the general direction of a physician.
|
|
72
|
Rural Health Clinic
|
A certified facility, which is located in a rural medically, underserved area that provides ambulatory primary medical care under the general direction of a physician.
|
|
73-80
|
Unassigned
|
N/A
|
|
81
|
Independent Laboratory
|
A laboratory certified to perform diagnostic and/or clinical tests independent of an institution or a physician's office.
|
|
82-98
|
Unassigned
|
N/A
|
|
99
|
Other Place of Service
|
Other service facilities not specified above.
|
Attachment IV – Provider Type Codes
|
CODE
|
Description
|
|
Codes included in this table are designed for completeness and in no way imply coverage of services under the Government Health Insurance Plan
|
|
|
AM
|
Ambulance
|
|
AS
|
Ambulatory Surgical Center
|
|
BB
|
Blood Bank
|
|
CL
|
Clinical Facility
|
|
DE
|
Dentist
|
|
DM
|
Durable Medical Equipment (DME)
|
|
EM
|
Emergency Facility
|
|
HH
|
Home Health Agency
|
|
HO
|
Hospital
|
|
HS
|
Hospice
|
|
LA
|
Laboratory
|
|
MD
|
Medical Doctor (Physician)
|
|
RX
|
Pharmacy
|
|
SN
|
Skilled Nursing Facility (SNF)
|
|
UF
|
Urgent Care facility
|
|
XR
|
Radiology Facility
|
|
ZZ
|
Other
|
Relationship Requirements
CARRIER ELIGIBILITY OUTPUT FILE – Insurance Record
This file is created by the HCRE export program and contains the demographic and eligibility information sent to ASES from the Department of Health and verified by ASES as eligible for Health Reform. This Insurance Record is added for the Meditis Implementaion on Februar 2011.
|
# Field
|
Record Fields
|
Position
|
Size
|
Notes
|
|
1
|
RECORD-TYPE
|
1
|
1
|
"I" for Insurance
|
|
2
|
TRAN-ID
|
2
|
1
|
E=eligible
|
|
3
|
PROCESS-DATE
|
3
|
8
|
MMDDYYYY
|
|
4
|
ODSI-FAMILY-ID
|
11
|
11
|
|
|
5
|
Member Suffix
|
22
|
2
|
|
|
6
|
Health Insurer Code
|
24
|
3
|
Code identifies Insurance Company
|
|
7
|
Policy Number
|
27
|
20
|
|
|
8
|
Policy-EXPIRATION DATE
|
47
|
8
|
|
|
9
|
Covered Services
|
55
|
40
|
20 coverage code filed (2 characters each)
|
|
10
|
FILLER
|
95
|
445
|
|
|
540
|
*** All are text fields
CARRIER ELIGIBILITY FILE - Medicare
MEMBERS RECORD
CARRIER ELIGIBLITY OUTPUT FILE
This file is created by the HCRE export program and contains the demographic and eligibility information sent to ASES from the Department of Health and verified by ASES as eligible for Health Reform. Modified on May 2003 for the direct contracting pilot project. Modified on March 2004 for Smartcard project. See entries in bold and highlighted. Modified on Sept. 2005 for Medicare Project. Modified August 2006 to add Coverage Fiels for new PSG contrating. Modified on January 2008 to add tran_id = H for sysprem records. Modify for Mediti on January 2011.
|
# Field
|
Record Fields
|
Position
|
Size
|
Notes
|
|
|
1
|
RECORD-TYPE
|
1
|
1
|
"M" for member
|
|
|
2
|
TRAN-ID
|
2
|
1
|
E=eligible, I=ineligible, R=reject, H= SYSPREM (history)
|
|
|
3
|
PROCESS-DATE
|
3
|
8
|
MMDDYYYY
|
|
|
4
|
FAMILY-SSN
|
11
|
9
|
SSN of Head-of-Household
|
|
|
5
|
FAMILY-SUFFIX
|
20
|
2
|
Zero fill, right justify.
|
|
|
6
|
FILLER
|
22
|
1
|
||
|
7
|
MEMBER-SSN
|
23
|
9
|
||
|
8
|
MEMBER-SUFFIX
|
32
|
2
|
||
|
9
|
FILLER
|
34
|
14
|
||
|
10
|
1ST-LAST-NAME
|
48
|
15
|
||
|
11
|
2ND-LAST-NAME
|
63
|
15
|
||
|
12
|
FIRST-NAME
|
78
|
20
|
||
|
13
|
MIDDLE-INITIAL
|
98
|
1
|
||
|
14
|
RELATIONSHIP
|
99
|
1
|
||
|
15
|
DATE-OF-BIRTH
|
100
|
8
|
MMDDYYYY
|
|
|
16
|
PLACE-OF-BIRTH
|
108
|
1
|
||
|
17
|
SEX
|
109
|
1
|
||
|
18
|
CATEGORY
|
110
|
1
|
||
|
19
|
CATEGORY-2
|
111
|
1
|
||
|
20
|
CONDITION
|
112
|
1
|
||
|
21
|
SOURCE-CODE
|
113
|
1
|
||
|
22
|
RECEIVE-SS
|
114
|
1
|
||
|
23
|
MED-INS-CODE
|
115
|
1
|
Zero fill, right justify.
|
|
|
24
|
POLICY
|
116
|
2
|
||
|
25
|
CLASS
|
118
|
1
|
||
|
26
|
CLASS-2
|
119
|
1
|
||
|
27
|
DENIAL-CAT
|
120
|
1
|
||
|
28
|
DENIAL-CAT-2
|
121
|
1
|
||
|
29
|
MARITAL-STATUS
|
122
|
1
|
||
|
30
|
SSN
|
123
|
9
|
||
|
31
|
PREG-IND
|
132
|
1
|
||
|
32
|
ABSENT-PARENT
|
133
|
1
|
||
|
33
|
HICN
|
134
|
11
|
||
|
34
|
PILOT-CAT
|
145
|
1
|
||
|
35
|
PILOT-CLASS
|
146
|
1
|
||
|
36
|
PILOT-DENIAL
|
147
|
1
|
||
|
37
|
HCRE-ELIGIBILITY-IND
|
148
|
1
|
||
|
38
|
HCRE-DENIAL-CODE
|
149
|
2
|
Zero fill, right justify.
|
|
|
39
|
OTHER-INSURER1
|
151
|
2
|
Insurance co. code NOT USED
|
|
|
40
|
OTH_POLICY1
|
153
|
20
|
Policy number NOT USED
|
|
|
41
|
OTHER-INSURER2
|
173
|
2
|
Insurance co. code NOT USED
|
|
|
42
|
OTH_POLICY2
|
175
|
20
|
Policy number NOT USED
|
|
|
43
|
OTHER-INSURER3
|
195
|
2
|
Insurance co. code NOT USED
|
|
|
44
|
OTH_POLICY3
|
197
|
20
|
Policy number NOT USED
|
|
|
45
|
GROUP-IDENT
|
217
|
2
|
"06" - ELA, "02" - Veteran, "22" - Small Bus. Zero fill, right justify.
|
|
|
46
|
ODSI-FAMILY-NO
|
219
|
11
|
"Gx"+HOH SSN for ELA (x=0,1,2 … by subscription period)
|
|
47
|
ELA-ERRORS
|
230
|
10
|
5 2-digit error codes for ELA-SB-Vet
|
|
|
48
|
AGENCY
|
240
|
5
|
Agency # for ELA / Group Num for SB. Zero fill, right justify.
|
|
|
49
|
MASTER PATIENT INDEX (MPI)
|
245
|
13
|
||
|
50
|
MEMBER CERTIFICATION DATE
|
258
|
8
|
MMDDYYYY
|
|
|
51
|
CONTRACT NUMBER
|
266
|
13
|
Include Suffix.
|
|
|
52
|
MEMBER PRIMARY CENTER
|
279
|
4
|
||
|
53
|
MEMBER PRIMARY CENTER EFFECTIVE DATE
|
283
|
8
|
MMDDYYYY
|
|
|
54
|
MEMBER NEW PRIMARY CENTER
|
291
|
4
|
||
|
55
|
MEMBER NEW PRIMARY CENTER EFFECTIVE DATE
|
295
|
8
|
MMDDYYYY
|
|
|
56
|
PCP1
|
303
|
15
|
||
|
57
|
PCP1 EFFECTIVE DATE
|
318
|
8
|
MMDDYYYY
|
|
|
58
|
PCP2
|
326
|
15
|
||
|
59
|
PCP2 EFFECTIVE DATE
|
341
|
8
|
MMDDYYYY
|
|
|
60
|
NEW PCP1
|
349
|
15
|
||
|
61
|
NEW PCP1 EFFECTIVE DATE
|
364
|
8
|
MMDDYYYY
|
|
|
62
|
NEW PCP2
|
372
|
15
|
||
|
63
|
NEW PCP2 EFFECTIVE DATE
|
387
|
8
|
MMDDYYYY
|
|
|
64
|
CARD ID NUMBER
|
395
|
15
|
||
|
65
|
CARD ID DATE
|
410
|
8
|
MMDDYYYY
|
|
|
66
|
ELA INDICATOR
|
418
|
1
|
1=NO PREMIUM
2=PREMIUM
Spaces when not ELA.
|
|
|
67
|
PRIMARY CENTER PCP CHANGE REASON
|
419
|
2
|
Based on the Reason of Code table.
|
|
|
68
|
MEDICAID INDICATOR
|
421
|
1
|
1=Medicaid Federal, 2=SCHIPS 3=Estatal4= Estatal other
|
|
|
69
|
MEDICARE INDICATOR
|
422
|
1
|
1=A&B, 3=A, 9=B
|
|
|
70
|
CARRIER
|
423
|
2
|
||
|
71
|
CARRIER_EFF_DATE
|
425
|
8
|
MMDDYYYY
|
|
|
72
|
NEW_CARRIER
|
433
|
2
|
||
|
73
|
NEW_CARRIER_EFF_DATE
|
435
|
8
|
MMDDYYYY
|
|
|
74
|
PLAN_TYPE
|
443
|
2
|
"bb"=eligible not enrolled, See Plan Type table
|
|
|
75
|
PLAN_TYPE_EFF_DATE
|
445
|
8
|
MMDDYYYY
|
|
|
76
|
PLAN_VERSION
|
453
|
3
|
Version of MA plan enrolled
|
|
|
77
|
PLAN_VERSION_EFF_DATE
|
456
|
8
|
MMDDYYYY
|
|
|
78
|
NEW_PLAN_TYPE
|
464
|
2
|
||
|
79
|
NEW_PLAN_TYPE_EFF_DATE
|
466
|
8
|
MMDDYYYY
|
|
|
80
|
NEW_PLAN_VERSION
|
474
|
3
|
||
|
81
|
NEW_PLAN_VERSION_EFF_DATE
|
477
|
8
|
MMDDYYYY
|
|
|
82
|
INSTITUTIONAL_STATUS
|
485
|
1
|
Y or N
|
|
|
83
|
HIC NUMBER MA
|
486
|
12
|
||
|
84
|
AUTO_ENROLL_INDICATOR
|
498
|
1
|
0 = Not Auto; >0 = Auto Enroll
|
|
|
85
|
AUTO_ENROLL_DATE
|
499
|
8
|
MMDDYYYY
|
|
|
86
|
IPA_ESPECIAL
|
507
|
1
|
1 = IPA Special
|
|
|
87
|
CMS_Cert_Status
|
508
|
2
|
Status of certification in CMS
|
|
|
88
|
Coverage_Code
|
510
|
3
|
||
|
89
|
New Contract Number
|
513
|
13
|
||
|
Special_Enroll
|
526
|
1
|
E = Emergency N = New Born
|
||
|
90
|
FILLER
|
527
|
13
|
||
|
540
|
|||||
*** All are Text Fields
CARRIER ELIGIBILITY FILE - Medicare
FAMILY RECORD
CARRIER ELIGIBLITY OUTPUT FILE
This file is created by the DAILY export program and contains the demographic and eligibility information sent to ASES from the Department of Health and verified by ASES as eligible for Health Reform. (Modified on May 2003 for the direct contracting pilot project. See entries in bold. Modified on March 2004 for Smartcard project. See entries in bold and highlighted. Modified on July 2005 for Medicare Project. Modified on January 2008 to add tran_id = H for sysprem records.) Modified for Mediti on January 2011.
|
# Field
|
Record Fields
|
Position
|
Size
|
Notes
|
|
1
|
RECORD-TYPE
|
1
|
1
|
"F" for family
|
|
2
|
TRAN-ID
|
2
|
1
|
E=eligible, I=ineligible, R=reject, H= SYSPREM (history)
|
|
3
|
PROCESS-DATE
|
3
|
8
|
MMDDYYYY
|
|
4
|
FAMILY-SSN
|
11
|
9
|
SSN of Head-of-Household(HOH)
|
|
5
|
FAMILY-SUFFIX
|
20
|
2
|
"00"
|
|
6
|
FILLER
|
22
|
14
|
|
|
7
|
ODSI-FAMILY-ID
|
36
|
11
|
"Gx"+HOH SSN for ELA (x=0,1,2 … by subscription period)
|
|
8
|
HOH-1ST-LAST-NAME
|
47
|
15
|
|
|
9
|
HOH-2ND-LAST-NAME
|
62
|
15
|
|
|
10
|
HOH-FIRST-NAME
|
77
|
20
|
|
|
11
|
REGION
|
97
|
1
|
|
|
12
|
MUNICIPALITY
|
98
|
4
|
Zero fill, right justify.
|
|
13
|
FACILITY
|
102
|
4
|
|
|
14
|
INVESTIGATION-IND
|
106
|
1
|
|
|
15
|
TRANSACTION-TYPE
|
107
|
1
|
|
|
16
|
EFFECTIVE-DATE
|
108
|
8
|
Start date of eligibility MMDDYYYY
|
|
17
|
FINANCIAL-RESP-PCT
|
116
|
1
|
|
|
18
|
CERTIFIER-NUMBER
|
117
|
2
|
|
|
19
|
EXPIRATION-DATE
|
119
|
8
|
End date of eligibility MMDDYYYY
|
|
20
|
COND-ELIG-IND
|
127
|
1
|
|
|
21
|
MAILING-ADDRESS1
|
128
|
25
|
|
|
22
|
MAILING-ADDRESS2
|
153
|
25
|
|
|
23
|
MAILING-CITY
|
178
|
16
|
|
|
24
|
MAILING-ZIP
|
194
|
5
|
|
|
25
|
MAILING-ZIP4
|
199
|
4
|
|
|
26
|
RESIDENCE-ADDRESS1
|
203
|
25
|
|
|
27
|
RESIDENCE-ADDRESS2
|
228
|
25
|
|
|
28
|
RESIDENCE-CITY
|
253
|
16
|
|
|
29
|
RESIDENCE-ZIP
|
269
|
5
|
|
|
30
|
RESIDENCE-ZIP4
|
274
|
4
|
|
|
31
|
PHONE
|
278
|
7
|
|
|
32
|
OTHER-INSURER1
|
285
|
2
|
Insurance co. code Not USED
|
|
33
|
OTH-POLICY1
|
287
|
20
|
Policy number NOT USED
|
|
34
|
OTHER-INSURER2
|
307
|
2
|
Insurance co. code NOT USED
|
|
35
|
OTH-POLICY2
|
309
|
20
|
Policy number NOT USED
|
|
36
|
OTHER-INSURER3
|
329
|
2
|
Insurance co. code NOT USED
|
|
37
|
OTH-POLICY3
|
331
|
20
|
Policy number NOT USED
|
|
38
|
MEMBERS
|
351
|
2
|
# members in family
|
|
39
|
ODSI-MEMBERS-ELIGIBLE
|
353
|
2
|
# members eligible ODSI / optionals ELA-SB-Vet
|
|
40
|
USER-CODE
|
355
|
6
|
|
|
41
|
ENTRY-DATE
|
361
|
8
|
MMDDYYYY
|
|
42
|
PCT-OF-POVERTY-LEVEL
|
369
|
3
|
|
|
43
|
DEDUCTIBLE-LEVEL-CODE
|
372
|
1
|
|
|
44
|
HCRE-MEMBERS-ELIGIBLE
|
373
|
2
|
# members eligible by ASES. Zero fill, right justify.
|
|
45
|
HCRE-DENIAL-CODE
|
375
|
2
|
Zero fill, right justify.
|
|
46
|
CARRIER-CODE
|
377
|
2
|
|
|
47
|
EFFECTIVE-CARRIER-DATE
|
379
|
8
|
For Family Carrier . MMDDYYYY
|
|
48
|
ELA-ERRORS
|
387
|
10
|
5 2-digit error codes for ELA-SB-Vet
|
|
49
|
MANCOMUNADO
|
397
|
1
|
Y / N (ELA Only)
|
|
50
|
FILLER
|
398
|
3
|
|
|
51
|
Family-PRIMARY-CENTER
|
401
|
4
|
IPA or PHO
|
|
52
|
NEW-CARRIER
|
405
|
2
|
New carrier code
|
|
53
|
NEW-Family-PRIMARY-CENTER
|
407
|
4
|
new IPA or PHO for families changing carrier
|
|
54
|
NEW-Family-PRIMARY CENTER EFFECTIVE DATE
|
411
|
8
|
MMDDYYYY - effective date of IPA/PHO change
|
|
55
|
CONTRACT NUMBER
|
419
|
13
|
Common part of Contract
|
|
56
|
REGION ASES
|
432
|
1
|
|
|
58
|
NEW CARRIER EFFECTIVE DATE
|
433
|
8
|
New Carrier MMDDYYYY
|
|
59
|
FAMILY PRIMARY CENTER EFFECTIVE DATE
|
441
|
8
|
MMDDYYYY
|
|
60
|
CERTIFICATION DATE
|
449
|
8
|
MMDDYYYY
|
|
61
|
PRIMARY CENTER PCP CHANGE REASON
|
457
|
2
|
Base on Reason Code table.
|
|
62
|
AUTO_ENROLL_INDICATOR
|
459
|
1
|
0 = Not Auto; >0 = Auto Enroll
|
|
63
|
AUTO_ENROLL_DATE
|
460
|
8
|
MMDDYYYY
|
|
64
|
PAM NEW FAMILY ID
|
468
|
11
|
New Family_Id assigned by PAM for Meditis. Use as a reference only.
|
|
65
|
FILLER
|
479
|
61
|
|
|
540
|
||||
*** All are Text Fields
|
QUERY RESPONSE FILE LAYOUT
|
||||
|
October 20, 2008
|
||||
|
This file is sent by ASES to Carriers as a response to query records. The Response Record informs if a Beneficiary is elegible for GHIP (Reform) coverage. It provides the key data elements which the Carrier will use to notify enrollment to ASES once approved by CMS.
|
||||
|
Query Response Record
|
||||
|
# Field
|
Record Fields
|
Position
|
Size
|
Notes
|
|
1
|
RECORD_TYPE
|
1
|
1
|
"R" for Response
|
|
2
|
CARRIER_PROCESS_DATE
|
2
|
8
|
YYYYMMDD
|
|
3
|
BENEFICARY SSN
|
10
|
9
|
|
|
4
|
CARRIER_1ST_LAST_NAME
|
19
|
15
|
|
|
5
|
CARRIER_2ND_LAST_NAME
|
34
|
15
|
|
|
6
|
CARRIER_FIRST_NAME
|
49
|
20
|
|
|
7
|
CARRIER_SEX
|
69
|
1
|
1 = Male, 2 = Female
|
|
8
|
CARRIER_DATE OF BIRTH
|
70
|
8
|
YYYYMMDD
|
|
9
|
CARRIER_REGION
|
78
|
1
|
|
|
10
|
CARRIER
|
79
|
2
|
Carrier Code
|
|
11
|
ASES_1ST_LAST_NAME
|
81
|
15
|
|
|
12
|
ASES_2ND_LAST_NAME
|
96
|
15
|
|
|
13
|
ASES_FIRST_NAME
|
111
|
20
|
|
|
14
|
ASES_SEX
|
131
|
1
|
1 = Male, 2 = Female
|
|
15
|
ASES_DATE OF BIRTH
|
132
|
8
|
YYYYMMDD
|
|
16
|
ASES_REGION
|
140
|
1
|
|
|
17
|
ELEGIBILITY_INDICATOR
|
141
|
1
|
Y or N
|
|
18
|
ODSI FAMILY ID
|
142
|
11
|
|
|
19
|
MEMBER SUFFIX
|
153
|
2
|
|
|
20
|
MPI
|
155
|
13
|
Alpha-numeric ej.-"0080012345678"
|
|
21
|
MEDICAID INDICATOR
|
168
|
1
|
1 = Federal Medicaid
|
|
22
|
ELEGIBILITY_EFFECTIVE_DATE
|
169
|
8
|
YYYYMMDD
|
|
23
|
ELEGIBILITY_EXPIRATION_DATE
|
177
|
8
|
YYYYMMDD
|
|
24
|
ASES_PROCESS_DATE
|
185
|
8
|
YYYYMMDD
|
|
25
|
MESSAGE_CODE
|
193
|
6
|
Spaces= no errors, 01=SSN no match, 02=Sex no match, 03=DOB no match, 04=Region no match, 05=Miembro de municipio no contratado por Carrier, 06=Empleado ELA, 07=SSN no match (history records)
|
|
26
|
ASES_DEDUCTIBLE_LEVEL
|
199
|
1
|
|
|
27
|
MUNICIPIO
|
200
|
4
|
Codigo Municipio en ASES
|
|
28
|
FECHA DE EFECTIVIDAD
|
204
|
8
|
Para uso en queries historicos. Formato YYYYMMDD.
|
|
29
|
CODIGO DE CUBIERTA
|
212
|
3
|
Codigo de Cubierta (Coverage Code)
|
|
30
|
FILLER
|
215
|
5
|
|
|
220
|
||||
ASES QUERY FILE
|
ELIGIBILITY QUERY FILE LAYOUT
|
||||
|
August 1, 2008
|
||||
|
This file is produced by MA Carriers and sent to ASES to verify the elegibility of Medicare Beneficiaries in the GHIP (Reforma).
|
||||
|
Query Record
|
||||
|
# Field
|
Record Fields
|
Position
|
Size
|
Notes
|
|
1
|
RECORD TYPE
|
1
|
1
|
"Q" for Query
|
|
2
|
PROCESS DATE
|
2
|
8
|
YYYYMMDD
|
|
3
|
BENEFICARY SSN
|
10
|
9
|
|
|
4
|
1ST LAST NAME
|
19
|
15
|
|
|
5
|
2ND LAST NAME
|
34
|
15
|
|
|
6
|
FIRST NAME
|
49
|
20
|
|
|
7
|
SEX
|
69
|
1
|
1 = Male, 2 = Female
|
|
8
|
DATE OF BIRTH
|
70
|
8
|
YYYYMMDD
|
|
9
|
REGION
|
78
|
1
|
|
|
10
|
CARRIER
|
79
|
2
|
Carrier Code
|
|
11
|
EFFECTIVE DATE
|
81
|
8
|
For historical queries. Enter the enrollment date for the enrollee. YYYYMMDD Fortmat. Day must be the first of the month. If the query is not hystorical, leave in blank.
|
|
12
|
FILLER
|
89
|
11
|
|
|
100
|
||||
*** All are Text Fields
Suscription File Error Description















ASES QUERY FILE
|
ELIGIBILITY QUERY FILE LAYOUT
|
||||
|
August 1, 2008
|
||||
|
This file is produced by MA Carriers and sent to ASES to verify the elegibility of Medicare Beneficiaries in the GHIP (Reforma).
|
||||
|
Query Record
|
||||
|
# Field
|
Record Fields
|
Position
|
Size
|
Notes
|
|
1
|
RECORD TYPE
|
1
|
1
|
"Q" for Query
|
|
2
|
PROCESS DATE
|
2
|
8
|
YYYYMMDD
|
|
3
|
BENEFICARY SSN
|
10
|
9
|
|
|
4
|
1ST LAST NAME
|
19
|
15
|
|
|
5
|
2ND LAST NAME
|
34
|
15
|
|
|
6
|
FIRST NAME
|
49
|
20
|
|
|
7
|
SEX
|
69
|
1
|
1 = Male, 2 = Female
|
|
8
|
DATE OF BIRTH
|
70
|
8
|
YYYYMMDD
|
|
9
|
REGION
|
78
|
1
|
|
|
10
|
CARRIER
|
79
|
2
|
Carrier Code
|
|
11
|
EFFECTIVE DATE
|
81
|
8
|
For historical queries. Enter the enrollment date for the enrollee. YYYYMMDD Fortmat. Day must be the first of the month. If the query is not hystorical, leave in blank.
|
|
12
|
FILLER
|
89
|
11
|
|
|
100
|
||||
*** All are Text Fields
ASES 820 (HIPPA COMPLIANCE PREMIUM OR FEE PAYMENT FILE)












ATTACHMENT 10
ASES Guidelines for Co-Location of Behavioral Health Provider in PMG Settings*
In accordance with the provisions of the Puerto Rico Mental Health Code, Law No. 408 of October 2, 2000, as amended, and the Puerto Rico Patient’s Bill of Rights and Responsibilities, the Government Health Plan (GHP) is committed to promoting mental and physical health integration, in order to improve program effectiveness and quality of life for enrollees.
Historically, physical and behavioral health services have had limited information and communication interchange, which suggests that patients were not being treated in a holistic approach. Our goal is to achieve better access to care and cost containment, while considering people’s health as a whole. The GHP health care coordination integration strategy for physical and behavioral health services, specifically through its Co-Location Integration Model, provides a mean to open communication channels so that better access, more targeted services and cost containment is achieved.
A Primary Medical Group (PMG) can actually operate out of one or multiple service locations such, for example, as medical offices or clinics. These locations can offer different kinds or levels of services and attend different volumes of beneficiaries. Some PMGs actually operate one or more full service clinics, with a complete array of multidisciplinary services such as primary care services, physician specialists’ services, laboratory, pharmacy and others. Other PMGs have a central clinic or office and then several smaller offices or clinics that offer different specific services. And there are still other PMGs, often referred to as “virtual PMGs,” that do not have a central office and have multiple providers in separate, stand-alone offices, operating as the pure concept of an independent practice association or IPA.
Accordingly, it is necessary to clarify which PMG service locations will be considered as PMG Settings for purposes of the Co-Location requirements. Specifically, it is necessary to provide guidance as to which PMG service locations must include the placement of a behavioral health provider and the amount of time per week that the provider must be available at each covered PMG service location.
The following guidelines are presented in order to clarify and adequately monitor compliance with the Co-Location requirements. These guidelines seek to ensure access to services and adequate communication between professionals without affecting the financial stability of the Co-Location Integrated Model.
Definition of PMG Setting: The following key elements are considered when defining “PMG Setting”:
| 1. | Volume: A PMG setting is the physical service location (clinic or office) where the population accesses most of the services within the PMG. These service locations can vary in size, kinds of services offered and number of beneficiaries attended. ASES has determined to define the term “PMG Setting” on the basis of the volume of beneficiaries served. ASES will consider as a “PMG Setting” any PMG service location that serves at least 5,000 beneficiaries. This setting must have available a behavioral health provider in the weekly timeframes detailed in these guidelines. Any PMG service location that services less than 5,000 beneficiaries must follow the “Virtual Co-Location Model” also set forth in these guidelines. |
| 2. | Comprehensiveness: The PMG setting where multidisciplinary services are rendered, among these, primary care services, physician specialists services, laboratory, pharmacy, behavioral provider, etc. |
1
| 3. | Substitution: Location with the capability of keeping services in case that the PCP is not available. |
Required Co-Location of Staff per PMG Setting. In view of the different kinds of PMG Settings and particularly, the different number of beneficiaries served, ASES has decided that the most reasonable course is to establish a table with the required weekly access to behavioral health providers according to the number of beneficiaries assigned to each PMG Setting. The standard minimum criteria for weekly access will be 4 hours per week for every 5,000 beneficiaries assigned to a PMG Setting. The following table details the minimum required weekly hours of mental health professional availability according to the number of beneficiaries served in each PMG Setting.
|
Covered Beneficiaries per PMG Setting
|
Minimum Behavioral
Health Colocation
Hours Required
|
|
5,000 – 9,999
|
4 hrs.
|
|
10,000 – 14,999
|
8 hrs.
|
|
15,000 – 19,999
|
12 hrs.
|
|
20,000 – 24,999
|
16 hrs.
|
|
25,000 – 29,999
|
20 hrs.
|
|
30,000 – 34,999
|
24 hrs.
|
|
35,000 – 39,999
|
28 hrs.
|
|
40,000 – 44,999
|
32 hrs.
|
|
45 000 – 49,999
|
36 hrs.
|
|
50,000 – 54,999
|
40 hrs.
|
Virtual Co-Location. In the case of those PMG service locations that have less than 5,000 beneficiaries assigned, such PMG service locations shall not be required to have a behavioral health provider available on site. In these instances, the PMG service location may refer the beneficiary to another service location within the same PMG that actually has a behavioral health provider available or consult with this behavioral health provider. This model aims to provide mental health treatment and coordinate the levels of services needed by patients referred by the PMG service location in question. Behavioral health providers will be available to address consults and discussions of cases.
A corrective action plan (“CAP”) will be required of every PMG Setting that does not comply with the required co-location level. The PMG must present the CAP to the corresponding Entity within seven (7) calendar days from the receipt of the notice of the need for corrective action. The Entity will evaluate and approve or deny the CAP within seven (7) calendar days from the day such CAP is received. All PMGs with an approved CAP must comply with the terms of the CAP and achieve the required co-location within the timeframes established in the CAP.
2
Penalty Matrix. In the event that a PMG does not comply with the required co-location levels in any of its PMG settings, the PMG may be subjected to penalties according to the following matrix:
|
Sanction Level
|
Sanction Type
|
Timeframe to cure
|
Comments
|
|
0
|
Notice of Non Compliance with Colocation Level
|
30 days
(Day 1-30)
|
A Corrective Action Plan is required
|
|
1
|
New members subscription Hold
|
30 days
(Day 31-60)
|
If within the first 30 day period, the PMG continues non-compliant.
|
|
2
|
PM/PM payment withhold and new member subscription
|
30 days
(Day 61-90)
|
A Standard $1.50 PM/PM payment withhold (in addition to sanction 1) if after the previous two 30day periods, the GMP is still non-compliant.
|
|
3
|
Fine
|
15 days
(Day 91-105)
|
Fines to be defined in accordance to contract
|
|
4
|
PMG Contract Cancelation
|
Day 106
|
*This document is under review and pending approval of CMS
ATTACHMENT 11
This exhibit was not available as of the filing date.
ATTACHMENT 12 - DELIVERABLES
All deliverables and documents submitted in accordance with this Attachment 12 must be submitted to ASES in English. Deliverables included in this list (as well as other documents that are subject to ASES review in accordance with this Contract) will be due to ASES in accordance with the deadlines established in the request for information and the readiness schedule established by ASES.
|
Deliverable Name
|
Contract Cite
|
|
|
1.
|
Newborn Notification Form
|
5.2.6.5
|
|
2.
|
Enrollee Handbook
|
6.2.1
|
|
3.
|
Provider Directory
|
6.2.1
|
|
4.
|
Enrollee ID Card
|
6.2.1
|
|
5.
|
Notice of Enrollment
|
6.2.4.3
|
|
6.
|
Notice of Redetermination
|
6.2.4.3
|
|
7.
|
Notice of Disenrollment
|
6.2.4.3
|
|
8.
|
Development and Distribution of Written Materials Policies and Procedures
|
6.3.1
|
|
9.
|
Tele GHP Policies and Procedures
|
6.8.10
|
|
10.
|
Tele GHP Quality Criteria and Protocols
|
6.8.16.2
|
|
11.
|
Tele GHP Outreach Program
|
6.8.16.3
|
|
12.
|
Scripts and Training Materials for Tele GHP Call Center Employees
|
6.8.16.4
|
|
13.
|
FAQs for Information Service and Medical Advice Service
|
6.8.15
|
|
14.
|
Website Screenshots
|
6.9.5
|
|
15.
|
Cultural Competency Plan
|
6.10.2
|
|
16.
|
Marketing Plan and copies of all Marketing Materials
|
6.14.5.1
|
|
17.
|
Special Coverage Registration form
|
7.7.3
|
|
18.
|
Special Coverage Registration Procedures
|
7.7.6.1
|
|
19.
|
Special Coverage Notification Forms
|
7.7.6.2
|
|
20.
|
Protocols for the Development of a Treatment Plan
|
7.7.6.4
|
|
21.
|
Special Coverage Provisions for Immediate Access to Specialists
|
7.7.6.5
|
|
22.
|
Strategy for Identification of Populations with Special Health Care Needs
|
7.7.6.6
|
|
23.
|
Needs Assessment Tool
|
7.8.2.3.4
|
|
24.
|
Care Management Policies and Procedures
|
7.8.2.6
|
|
25.
|
Disease Management Policies and Procedures
|
7.8.3.5
|
|
26.
|
EPSDT Outreach and Education Plan
|
7.9.1.4
|
|
27.
|
Integration Plan
|
8.7
|
|
28.
|
Timely Access to Behavioral Health Services Policies and Procedures
|
9.1.5.4
|
|
29.
|
Provider Licensing and Certification Policies and Procedures
|
9.2.3.6.1.18
|
|
30.
|
Enrollee Selection of PCP
|
9.3.1.5.2
|
|
31.
|
PPN Participation Policies and Procedures
|
9.3.3.5.1
|
|
32.
|
Enrollee Access to Specialists Policies and Procedures
|
9.4.5
|
|
33.
|
Protocol to Screen Enrollees for Special Coverage
|
9.5.2.2
|
ATTACHMENT 12 - DELIVERABLES
Page 2
|
34.
|
Provider Hours and Operational Monitoring Policies and Procedures
|
9.5.5
|
|
35.
|
Model Provider Contracts
|
10.1.6.1
|
|
36.
|
Provider Guidelines
|
10.2.1.3
|
|
37.
|
Programmatic Changes Policies and Procedures
|
10.2.1.6
|
|
38.
|
Provider Continuing Education Curriculum
|
10.2.2.1
|
|
39.
|
Payment System to State Health Facilities
|
10.5.9
|
|
40.
|
Utilization Management Policies and Procedures (including referrals)
|
11.2.2
|
|
41.
|
Utilization Management Clinical Criteria
|
11.4.3
|
|
42.
|
QAPI Program (including ER quality)
|
12.2.4
|
|
43.
|
Wellness Plan
|
12.5.8.4
|
|
44.
|
Fraud, Waste, and Abuse Policies and Procedures
|
13.1.6
|
|
45.
|
Fraud, Waste and Abuse Compliance Plan
|
13.2.1
|
|
46.
|
Network Provider Investigations, Suspensions and Debarment Policies and Procedures
|
13.1.11
|
|
47.
|
Provider Disclosure Form
|
13.5.13.3
|
|
48.
|
Grievance System Policies and Procedures
|
14.1.4
|
|
49.
|
Grievance System forms
|
14.1.2
|
|
50.
|
Staff Training Plan
|
15.3.2
|
|
51.
|
Current MCO Organization Chart
|
15.3.2
|
|
52.
|
Implementation Plan
|
15.5.2
|
|
53.
|
Provider Payment Schedule
|
16.5.1
|
|
54.
|
Business Continuity and Disaster Recovery (BC-DR)
|
18.2.8.3
|
|
55.
|
Protection of Enrollee Health Records Policies and Procedures
|
34.1.6
|
ATTACHMENT 13
NL AUTO ENROLLMENT
October 22, 2014
REGULATORY LETTER #14-10-22
TO ALL CONTRACTED ENTITITES AND PROVIDERS WHO OFFER HEALTH SERVICES
UNDER THE “MI SALUD” HEALTH PLAN
Re: To Repeal Regulatory Letter #11-06-29 about Auto-Enrollment
This Regulatory Letter replaces Regulatory Letter #11-06-29 from June 29, 2011. This New Regulatory Letter establishes that every person who is eligible to the “Mi Salud” Health Plan from the Government of Puerto Rico will be automatically insured and subscribed to the health Plan according to the Region of his/her residence. His/her card from the Government’s Health Plan will be sent through mail in a period not greater than five (5) working days after being eligible.
The insured will be able to begin receiving health plan services the same day in which the Medicaid Office, from the Puerto Rico Health Department, delivers the Notification of the Action Taken through Form MA-10. The date to determine since when the person is insured, is the date indicated in the section titled “Certification Date” in Form MA-10 (upper right section). From that date forward, you can access medical services through your Medical Group and Primary Care Physician, even if you have not received your Health Plan’s identification card. If you have not received your card, you can present Form MA-10 to receive the medically necessary services.
Due to the fact that the Medicaid Program does not issue cards at the moment when beneficiaries are eligible, we require all the collaboration from the insurance company to issue the cards in a period not greater than two (2) working days. Providers cannot deny services because the patient does not have the card, as long as the patient presents the MA-10 Form and an identification with photo.
It is the provider’s responsibility to communicate with the insurance company to ensure that the beneficiary is duly subscribed in the Health Plan and that the insured has a Medical Group and Primary Care Physician assigned. Even if the insured does not have a primary care physician, the provider can render the physical, mental or dental health services, as applicable. The health entities contracted by ASES will have the responsibility to pay for the rendered services according to the terms and conditions of the contract.
To facilitate billing and payment of the services rendered by the provider, Form MA-10 includes a section that indicates "MPI/SS" which provides the insured’s identification number. The provider must accompany, with the invoice, a copy of Form MA-10, to send to the insurance company. Form MA-10 indicates the period in which the beneficiary is eligible.
The information in this Regulatory Letter does not apply to beneficiaries subscribed to a Platino Medicare Health Plan.
ASES requires the contracted entities that in a non-extendable term of five
(5) calendar days, they send an exact copy of this Regulatory Letter to each of their participating providers in the “Mi Salud” Health Plan. The entities must send an Affidavit to the attention of the Executive Director of ASES, signed by its Corporate President stating that they have duly complied with what is required in this letter.
We request faithful compliance with this regulatory letter to continue rendering excellent and quality services according to the “Mi Salud” Health Plan from the Government of Puerto Rico.
As always, we are sure that we will count with the support of all our providers.
Cordially,
(signed)
Ricardo A. Rivera Cardona
Executive Director
# 1552. Ave Ponce de León
Sector El Cinco, San Juan, Puerto Rico
PO Box 195661
San Juan, PR 00919-5661
Tel: 787-474-3300 Fax. 787-474-3345
ATTACHMENT 13
NL REFERRALS
October 22, 2014
REGULATORY LETTER # 14-10-22-A
TO ALL CONTRACTED ENTITIES BY THE HEALTH INSURANCE ADMINISTRATION (ASES) AND HEALTH CARE PROVIDERS OF THE GOVERNMENT’S HEALTH PLAN
Re: REFERRALS
This Regulatory Letter is published to repeal Regulatory Letter #11-0501 from May 1, 2011 and Letter #2013-11-14 from November 14, 2013, according to the legislation and applicable regulation.
The Health Program of the Government of the Commonwealth of Puerto Rico ("the plan") is an integrated health model adopted to render services to the beneficiaries covered in accordance with Law Num. 72 from 1993, known as the Law for the Administration of Health Insurance, and the people who are medically indigent covered by the Medicaid Federal program. Law Num. 72 defines all health care providers as the “primary care physicians, support physicians, primary services, primary providers and health services organizations”. Law Num. 72 defines the “primary care physician” as the "professional participating provider who evaluates and gives initial treatment to the beneficiaries" and who is responsible to determine the services that the beneficiary needs, to provide continuity and to refer beneficiaries to specialized services". "We consider as primary care physicians, the general practitioners, internists, family doctors, pediatricians, gynecologists and obstetricians”. To guarantee the quality and free selection of beneficiaries to the health services covered by the Plan, Law Num. 72 also states that the beneficiaries of the Plan will have the right to choose, and also change, their primary care physician, right to free access to the medical services, and that they are not denied services under the coverage.
According to Law Num. 72, the health plan beneficiaries subscribe to a Primary Care Physician Group (Grupo Medico Primario or GMP, initials in Spanish), that is the custodian of the integrated health management of the beneficiaries subscribed to the plan and assigned to that GMP. Within each GMP there are a number of primary care physicians and specialists necessary for the health care of the population under their attention, as required in the contract between ASES and the insurance company selected, the medical conditions that prevail and the demographics of this population. These GMPs, as a general norm, must include general practitioners, family doctors, internists, pediatricians, gynecologists and gynecologists/obstetricians.
The GMP is responsible to watch and guarantee that beneficiaries under its attention have access to the specialized services covered and necessary for their health conditions. To facilitate free access to the specialized services, the health plan model provides that each GMP creates its own network of preferred health providers. These preferred networks guarantee to the beneficiaries, direct access to the coverage of specialized services without a referral or authorization from the primary care physician.
The health plan’s integrated model also gives beneficiaries access to specialists who are not part of the GMP and its preferred network, through the general network of the insurance company contracted by ASES. This general network complements the preferred network of the GMP and offers other options to the beneficiary in case that the alternatives available within the preferred network are not viable or if there is a need of other providers. However, for the GMP to be able to offer its patients a health care that is administered in an orderly, coordinated and responsible way, in compliance with Law Num. 72, it is precise that before they use a specialist outside the GMP and its preferred network, the beneficiary obtains a referral from the primary care physician and pays, if applicable, the corresponding co-payment when he/she visits the specialist of the general network.
The issuance of referrals is the exclusive responsibility of the primary care physician selected by the insured. The insured, under no circumstances, can be directed to another facility or medical group to obtain a referral or authorization for services. No Board, Committee, Medical Group Administrator, etc. can issue referrals. And they cannot pass judgment over the primary care physician’s determination when issuing the referral.
In the particular case of access to gynecologists and gynecologists/obstetricians, it is precise to recognize that Law Num. 194 from August 25, 2000, as amended, better known as “Bill of Beneficiary Rights and Responsibilities", also applicable to the Health Plan of the Government of the Commonwealth of Puerto Rico, establishes that in relation to the selection of health care plans and providers of health care medical and hospital services, every patient, user or consumer of those plans and services in Puerto Rico has the right "that the individual or group health care plans cover services of gynecology and obstetrics with direct access, without the need of a referral or previous authorization of the plan, as long as the physician is part of the provider network of the health care plan".
In consideration with everything previously presented and in conformity with Law Num. 72 from ASES and according to the Bill of Patients Rights and Responsibilities, the model of the Government’s Health Plan recognizes the rights of its female beneficiaries to select and directly access the gynecologist or gynecologist-obstetrician of their preference through their GMP and preferred network without the need of a referral from their primary care physician. In the case in which the female beneficiaries need the services of a gynecologist or gynecologist-obstetrician in the general network and outside the preferred network, they must obtain the corresponding referral from the primary care physician and pay, if applicable, the co-payment that corresponds in the visit to the specialist.
We remind you that the referrals must be provided in the same visit to the primary care physician (PCP) or in a term no greater than twenty-four (24) hours after that visit. In those cases in which the specialist to be referred is not a part of the preferred network of the GMP, the primary care physician will have the responsibility to coordinate the appointment and make sure that the service is provided in a term no greater than thirty (30) days, always taking into consideration the patient’s health status. In this case, the applicable co-payments for specialists outside the network will not be charged.
In terms of the virtual region, there is a free selection model for the insured that belong to this region. As you should know, this region is composed of children under the custody of the Family Department (ADFAN) and women who are survivors of domestic violence (Women’s Advocate Office). For this region the selection of group or primary care physician does not apply, as this is a floating population. For this reason it is not necessary to issue referrals for this population to access health care services.
To conclude, we reiterate, that under the plan’s preferred networks, it is not required to have referrals to visit specialists, or countersignatures from the primary care physician for services of physical coverage. The insured will not have to pay copayments when using the preferred networks within the Primary Care Physician Group of his/her choice and the insurance company cannot deny the invoices for these services if they don’t have the referral of the primary care physician. Besides, a referral is not required to access dental services, with the exception of the maxillofacial surgeries in which the referral of the primary care physician is required for the maxillofacial surgeon.
We request faithful compliance with this regulation to continue rendering excellent and quality services in conformity with the Health Program of the Government of the Commonwealth of Puerto Rico.
Cordially,
(signed)
Ricardo A. Rivera Cardona
Executive Director
# 1552. Ave Ponce de León Sector El Cinco
San Juan, Puerto Rico
PO Box 195661
San Juan, PR 00919-5661
Tel: 787-474-3300 Fax. 787-474-3345
ATTACHMENT 13
NL ER CLAIMS PAYMENT
October 22, 2014
REGULATORY LETTER #14-10-22-B
CLAIMS PAYMENT IN EMERGENCY ROOMS FOR PHYSICAL AND MENTAL HEALTH SERVICES IN MEDICAL-SURGERY HOSPITALS
Re: To Repeal Regulatory Letter 04-01-30 about the separation of the rendering and billing of physical and mental health services.
From April 1, 2015*, the services model of the Health Plan of the Government establishes that each insurance company, responsible to administer the health services for a determined region, will also be responsible to pay for the physical and mental health services under the same billing system.
The integration model ensures the integration of the data and the payment responsibility under one entity. According to this policy, each insurance company will establish the contracts with every emergency room service and will be the Single Payer regardless if the service is a diagnostic of physical or mental health. As this service and financial risk model is an integrated one, Regulatory Letter Num. 04-01- 30 sent on February 13, 2004, that established the separation of rendering and billing physical and mental health services, ceases to have effect.
If there is a service model in which mental health entities act as subcontracted entities to give mental health services, the arrangement needs to be adjusted so that only the entity responsible for the financial risk is the one that processes the payment for the claims of such services. It will be the responsibility of the entity contracted by ASES, to contract the facilities and providers of physical and mental health. It will also be responsible for the configuration and credentialing of both types of providers in its claims adjudication system, with the fees agreed for the services according the benefits coverage.
From April 1, 2015 * Regulatory Letter Num. 04-01-30 ceases to be in effect and it will be replaced by this Regulatory Letter.
(signed)
Ricardo A. Rivera Cardona
Executive Director
# 1552. Ave Ponce de León Sector El Cinco
San Juan, Puerto Rico
PO Box 195661
San Juan, PR 00919-5661
Tel: 787-474-3300 Fax. 787-474-3345
*Final date will depend on the effectiveness of the contract.
ATTACHMENT 13
NL ER CLAIMS PAYMENT
January 21, 2014
Regulatory Letters 13-1212 and 13-1216 Amended
Companies contracted by ASES to provide physical and mental health services and pharmacy
MARKETING MATERIALS
Receive warm greetings from the team that works at the Health Insurance Administration (ASES). As a result of the evaluation of the promotional materials and written communications, effective January 1, 2014, the following is mandatory:
1. The content of the lectures, presentations, sponsorships, health fairs, educational materials, efforts in mass media, printed advertisements, radio and television, media plan, posters and banners, etc. that include information of benefits and services of the Government’s Health Plan and that is financed totally or partially with funds from ASES, must be approved by ASES. It is a requirement to include a detailed estimate of the costs for the event, along with the materials to be evaluated by ASES. (Continues on next page).
2. All presentations, public expositions and/or participation of any employee of the company, in any publicity media, press and/or radio with the objective to explain benefits and services of the Government’s Health Plan to all types of audiences such as beneficiaries, providers, municipal and state officers, and the community, must be approved by ASES.
3. The content of all marketing materials must comply with the following guidelines:
| • | The ASES logo to be used is the color green and gray. (We include copy of the logo and a guide on how to handle the logo). |
| • | The ASES logo will be placed in the heading and the cover of all pieces. |
| • | The logo of the company will be placed in the lower or posterior part of each piece, as applicable, in a smaller size than the ASES logo. |
| • | Mi Salud will be referred as the Government’s Health Plan |
| • | All headings must have: Government’s Health Plan, the ASES logo and the title of the piece. |
| • | All pieces will include in the lower or posterior part, as applicable, the seal with the ASES customer service number (image is included). |
| • | Banners and tents must include the name of the Government’s Health Plan and the ASES logo in the foreground. The logo of the company can be placed in the background and in a smaller size than the ASES logo. |
4. All marketing materials must be sent to the ASES Compliance Department for approval. An approval number will be assigned which must be placed in the inferior or posterior part of each promotional material in a size no less than 8. Without this approval number, the marketing material cannot be published. The time for revision for each marketing material will be of 15 working days.
Contract 14-050 allows ASES to impose sanctions and monetary penalties among others, when the company does not comply with the instructions that ASES has notified. For this reason the direct or indirect distribution of any marketing material without duly approval from ASES, will constitute an event subject to monetary sanctions and the immediate cease and desist of the distribution of this marketing. For any doubts or questions regarding this subject, you can contact Mr. Jorge Mas or Mr. William Ruiz at 787-474-3300 ext. 2308 or 2220 respectively or through electronic mail at: jmas@asespr.org or wruiz@asespr .org.
(signed)
Ricardo A. Rivera Cardona
Executive Director
| cc. | William Ruiz |
Jorge L. Mas
# 1552. Ave Ponce de León Sector El Cinco
San Juan, Puerto Rico
PO Box 195661
San Juan, PR 00919-5661
Tel: 787-474-3300 Fax. 787-474-3345
ATTACHMENT 13
SPECIAL NEEDS CHILDREN DIAGNOSTIC CODE
Commonwealth of Puerto Rico
Department of Health
December 23, 2008
Minerva Rivera, Esq.
Executive Director of ASES
(Signed)
Johnny V. Rullán, MD, FACPM
Secretary of Health
LISTING OF DIAGNOSES OF CHILDREN WITH SPECIAL HEALTH NEEDS (NNES)
The Listing of Diagnoses of Children with Special Health Needs was revised in response to your request.
It’s necessary to point out that at the beginning this listing was provided to ASES as a guide; therefore, it’s important that a child who does presents some condition which is in the listing not be excluded from the benefits of the coverage of the Health Card of the Government of Puerto Rico. If the child fulfills the definition of Children with Special Health Needs of the Bureau for the Child-Maternal Health, he/she must receive the services even before a diagnosis is established.
In addition, we submit our recommendations for the identification, diagnosis and treatment of the children and youth with special health needs to as assure access to the services which this population needs. These have the purpose of ensuring some uniform needs for all the children with special health needs regardless of the insurer.
Thank you for your attention to this matter.
NNES Diagnosis Listing
d/several 2008-06
CHILDREN WITH SPECIAL HEALTH NEEDS
DEFINITION:
Children who have or are at a greater risk of developing a chronic physical, conduct, emotional or developmental condition, who also need health services and other related services of a type or in an amount that goes beyond what children in general need.
STANDARD OF MEDICAL NEED SPECIFIC TO CHILDREN
• Medically necessary services are those necessary for the prevention and maintenance of health or for the diagnosis and treatment of a physical or mental condition, or if they were necessary to prevent the deterioration of that condition or to promote the development or the maintenance of appropriate functioning for the age.
NNES SPECIAL COVERAGE
In the “Special Coverage”, the Insurance Companies, with whom ASES contracts the services, assume the risks of the services for the conditions classified with Diagnoses of Conditions of Children with Special Needs. (See list of ASES diagnoses).
In this list of conditions there are included the most frequent diagnoses, but it is not limited to or excludes other conditions that fulfill the definition. With this purpose there should be utilized a screener to determine its applicability.
It is the responsibility of the primary physician to request the coverage, and register the insured person utilizing the corresponding form for Children with Special Health Needs that is found in the Manual of the Provider. The certification process may also be initiated by one of the Pediatric Centers of the Department of Health.
To be able to evaluate and certify these cases it is necessary to include, together with the form, the necessary information: Ex:
| • | Summary of the case: Up-to-date history record and physical examination |
| • | Evaluations and consultations from specialists |
| • | Results of diagnostic procedures and tests |
| • | Results of diagnostic laboratory tests |
| • | Necessary follow-up plan |
| • | Treatment plan |
This information and the registration form must be sent to the Case Handling Program (PMC) of the Insurer. The PMC will evaluate the application for certification and the documentary information included. Each case is evaluated individually by the Handler of cases and consulting the Program’s advisory team. This depend on the coverage negotiated. The agreement with the insurance companies must be uniform and that it obligates all the companies equally.
The family and the primary physician are notified directly by letter as to whether the application for inclusion of his/her patient in the NNE registry has been accepted or denied; or if there is information missing for the consideration of the case. The physician and/or the family may appeal in writing any denial decision, with the necessary additional information.
Index by Diagnosis and Condition
ICD 9
Metabolic Disorders
| 270 | Disorders of the metabolism of aromatic amino acids |
| 270.0 | Disorders of the transport of amino acids |
| 270.0 | Cystinosis |
| 270.0 | Cystinuria |
| 270.0 | Fanconi |
| 270.0 | Hartnup’s |
| 270.0 | Lowe’s |
| 270.1 | Phenylketonuria (PKU) |
| 270.2 | Disorders of metabolism of tyrosine |
| 270.2 | Alcaptonuria |
| 270.2 | Hyperthyrosinemia |
| 270.2 | Ocronosis |
| 270.2 | Tyrosinosis |
| 270.2 | Tyrosinuria |
| 270.2 | Albinism |
| 270.3 | Maple-Syrup disease |
| 270.3 | Other metabolic disorders of chained amino acids |
| 270.3 | Hyperleukina-isoleukinemia |
| 270.3 | Hypervalinemia |
| 270.3 | Isovaleric acidemia |
| 270.3 | Methylmalonic acidemia |
| 270.3 | Propionic acidemia |
| 270.3 | Metabolic disorders with amino acids with sulfide |
| 270.4 | Homocystinuria |
| 270.4 | Deficiency of sulfite oxidase |
| 270.4 | Homocystine cystathionine |
| 270.5 | Other metabolic disorders of aromatic amino acids |
| 270.5 | Disorder of: |
| 270.5 | Histidine metabolism |
| 270.5 | Tryptophan metabolism |
| 270.5 | Metabolic disorders of chain amino acids and fatty acids |
| 270.6 | Metabolic disorders of the citrulinemia urea cycle |
| 270.6 | Hyperammonemia |
| 270.6 | Argininosuccinic acid |
| 270.7 | Metabolic disorders of lysine and hydroxilisine |
| 270.7 | Glutaric aciduria |
| 270.7 | Hydroxilisinemia |
| 270.7 | Hyperlisinemia |
| 270.7 | Metabolic disorders of glycine |
| 270.7 | Non-ketosic hyperglysinemia |
| 270.8 | Deomitine metabolic disorders |
| 270.8 | Omitinemia type I, II |
| 270.8 | Hyperhydroxyprolinemia |
| 270.8 | Hyperprolynemia types I, II |
| 270.8 | Sarcosinemia |
| 270.8 | Other specific amino acid metabolic disorders |
| 270.9 | Other non-specific metabolic and amino acid transport disorders |
| 271 | Carbohydrate transport and metabolism disorders |
| 271.0 | Glycogenosis |
| 271.0 | Amylopectinosis |
| 271.0 | Deficiency of glucose-6-phosphatase |
| ICD9 | Index by Diagnosis and Condition |
| 271.0 | Cardiac glycogenosis |
| 271.0 | Disease: |
| 271.0 | Andersen |
| 271.0 | Cori |
| 271.0 | Forbes |
| 271.0 | Hers |
| 271.0 | McArdle |
| 271.0 | Pompe |
| 271.0 | Tauri |
| 271.0 | Von Gierke |
| 271.0 | Deficiency of hepatic phospholirase |
| 271.1 | Metabolic disorder of Galactosemia galactose |
| 271.2 | Metabolic disorder of fructose, Fructosemia |
| 271.3 | Intolerance to lactose |
| 271.3 | Other disorders of intestinal absorption of carbohydrates |
| 271.4 | Other specific metabolic disorders of carbohydrates Pentosuria, renal Glycosuria |
| 271.8 | Metabolic disorders of pyruvate and gluconeogenesis |
| 271.8 | Defects in degradation of glycoprotein |
| 271.9 | Non-specific disorder of the transport and metabolism of carbohydrates |
| 272 | Metabolic disorder of lipids |
| 272.0 | Hypercholesterolemia |
| 272 | Gangliosidosis |
| 272.0 | Hypercholesterolemia |
| 272.1 | Hyperglycerinemia |
| 272.4 | Other non-specific hyperlipidemias |
| 272.7 | Other gangliosidosis |
| 272.7 | Lipidosis |
| 272.7 | Anderson’s |
| 272.7 | Fabry’s |
| 272.7 | Gaucher’s |
| 272.7 | Krabbe |
| 272.7 | Neimman-Pick |
| 272.7 | Faber’s |
| 272.7 | Metachromatic leukodystrophia |
| 272.7 | Mucopolysaccaridosis, type I |
| 272.7 | Hurler’s |
| 272.7 | Hurler-Scheie |
| 272.7 | Scheie |
| 272.7 | Mucopolysaccaridosis, type II |
| 272.7 | Hunter’s |
| 272.7 | Other mucopolysaccaridosis |
| 272.7 | Maroteaux-Lamy |
| 272.7 | Morquio’s |
| 272.7 | Sanfilippo |
| 273 | Metabolic disorders of plasma protein |
| 274.9 | Unspecific gout |
| 275 | Metabolic disorder of minerals |
| 275.0 | Metabolic disorders of iron |
| 275.1 | Metabolic disorders of copper |
| 275.1 | Wilson’s |
| 275.2 | Metabolic disorders of magnesium |
| 275.3 | Metabolic disorders of phosphorus |
| ICD9 | Index by Diagnosis and Condition |
| 275.4 | Metabolic disorders of calcium |
| 275.9 | Other metabolic disorders of minerals |
| 276.2 | Lactic Acidosis |
| 277 | Other metabolic disorders |
| 277.00 | Cystic Fibrosis |
| 277.1 | Metabolic disorders of purine and pyrimidine |
| 277.1 | Hereditary eritropoietic porphyria |
| 277.2 | Other metabolic disorders of purine and pyrimidine |
| 277.2 | Lesch-Nyhan |
| 277.2 | Hereditary Xantinuria |
| 277.3 | Amyloidosis |
| 277.4 | Gilbert’s |
| 277.4 | Crigler-Najjar |
| 277.4 | Other metabolic disorders of bilirubin |
| 277.4 | Dubin-Johnson |
| 277.4 | Rotor’s |
| 277.6 | Antitrypsin alpha-1 deficiency |
| 277.8 | Other specific metabolic disorders |
| 277.81 | Primary carnitine deficiency |
| 277.82 | Carnitine deficiency |
| 277.85 | Disorders of the oxidation of fatty acids |
| 277.85 | CPT1, CPT2, LCHAD, VLHAD, MCAD, SCAD |
| 277.87 | Mitochondrial metabolic disorders |
| 277.89 | Other specific disorders of the metabolism |
| 277.89 | Hans Schuler Christian, Hystiocitosis, Hystiocitosis |
| 277.9 | Other non-specific metabolic disorders |
Hereditary and degenerative diseases of the Nervous System
| 330 | Cerebral degeneration |
| 330.0 | Sphingolipidosis (Leukodystrophia) |
| 330.1 | Cerebral Lipidosis |
| 330.8 | Other cerebral degenerations |
| 330.8 | Alper’s |
| 330.8 | Leigh’s |
| 330.8 | Sub-acute necrotizing encelopathy |
| 331.4 | Obstructive hydrocephalia, acquired |
| 333.1 | Essential shakes |
| 333.2 | Myoclonus |
| 333.4 | Huntington’s chorea |
| 334.0 | Spinocerebral disease |
| 334.0 | ereditary ataxia |
| 334.0 | Friedreich’s ataxia |
| 334.1 | Hereditary spastic paraplegia |
| 334.2 | Primary cerebellar degeneration |
| 334.2 | Marie’s |
| 334.2 | Sanger’s-Brown |
| 334.8 | Telangiectasia-ataxia |
| 335 | Spinal muscular atrophy and kindred syndromes |
| 335.0 | Infantile spinal muscular atrophy, type I (Werdnig-Hoffman) |
| 335.1 | Other hereditary spinal muscular atrophies |
| ICD9 | Index by Diagnosis and Condition |
| 335.10 | Spinal muscular atrophy: |
| 335.10 | Infantile, type II |
| 335.11 | Juvenile, type II (Kugelberg-Welander) |
| 340 | Multiple sclerosis |
| 341.0 | Other demyelinant diseases of the central nervous system |
| 341.1 | Diffuse sclerosis |
| 341.1 | Periaxial encephalitis |
| 341.1 | Schiller’s disease |
| 341.8 | Other demyelinant diseases of the central nervous system |
| 341.8 | Central demyelination of the corpus callosum |
| 341.8 | Pontine central myelinosis |
| 341.8 | Acute transverse myelitis in demyelinant disease of the central nervous system |
| 341.8 | Subacute necrotizing myelitis |
| 341.9 | Non-specific demyelinant diseases of the central nervous system |
| 345 | Epilepsy |
| 345.1 | Generalizes epilepsy without convulsions |
| 345.1 | Generalized epilepsy with convulsions |
| 345.1 | · clonic |
| 345.1 | · myoclonic |
| 345.1 | · tonic |
| 345.1 | · tonic-clonic |
| 345.1 | Lennox-Gastaut syndrome |
| 345.2 | Epileptic petit mal state |
| 345.3 | Epileptic grand mal state |
| 345.3 | Tonic-clonic epileptic state |
| 345.4 | Partial epilepsy, with loss of consciousness |
| 345.4 | Epileptic absence state |
| 345.4 | Complex partial epileptic mal state |
| 345.5 | Partial epilepsy, without loss of consciousness |
| 345.6 | Salaam attacks |
| 345.6 | Infantile spasms |
| 345.7 | Continuous partial epilepsy (Kozhevnikof) |
| 345.8 | Other epileptic states |
| 345.9 | Non-specified type epileptic mal state |
| 342.0 | Flaccid hemiplegia |
| 342.1 | Spastic hemiplegia |
| 342.3 | Infantile monoplegia |
| 342.9 | Non-specified hemiplegia |
| 343 | Infantile cerebral paralysis |
| 343.0 | Spastic diplegia |
| 343.1 | Congenital hemiplegia |
| 343.2 | Non-specified, quadriplegia |
| 343.4 | Infantile hemiplegia |
| 343.8 | Congenital spastic paralysis (cerebral) |
| 343.9 | Non-specific infantile cerebral paralysis |
| 344 | Other infantile spastic paralysis syndromes, non-congenital |
| 356 | Motor and sensory hereditary neuropathy |
| 356.0 | Idiopathic hereditary neuropathies |
| 356.0 | Dejerine-Sottas disease |
| 356.1 | Peroneal muscular atrophy, Charcot-Marie-Tooth disease |
| 356.2 | Sensory hereditary neuropathy, types I-IV |
| ICD9 | Index by Diagnosis and Condition |
| 356.8 | Roussy Levy syndrome |
| 348 | Other conditions of the brain |
| 348.0 | Cerebral cyst |
| 348.30 | Unspecified encephalopathy |
| 356.3 | Resfsum disease |
| 356.3 | Neuropathy associated with hereditary ataxia |
| 356.4 | Idiopathic progressive neuropathy |
| 356.6 | Other hereditary and idiopathic neuropathies |
| 356.9 | Hereditary and idiopathic neuropathy, without another specification |
| 357 | Inflammatory polyneuropathy |
| 357.0 | Guillain-Barre syndrome |
| 357.0 | Acute infectious polyneuritis (post) |
| 359 | Muscular dystrophy and other neuropathies |
| 359.0 | Hereditary congenital muscular dystrophy |
| 359.1 | Progressive hereditary muscular dystrophy |
| 359.1 | * autosomic recessive, infantile type, similar to Duchenne or Becker |
| 359.1 | · benign (Becker) |
| 359.1 | · waist-pelvic |
| 359.1 | · distal |
| 359.1 | · scapuloperoneal |
| 359.1 | · benign scapuloperoneal with precocious contractures [Emery-Dreituss] |
| 359.1 | · fascioscapulohumeral |
| 359.1 | · gravis [Duchenne] |
| 359.1 | · ocular |
| 359.1 | · oculopharyngea |
| 359.2 | Motonic disorders |
| 359.2 | Myotonic disorders [Steiner] |
| 359.2 | Congenital myotonia: |
| 359.2 | · dominant [Thomsen] |
| 359.2 | · recessive [Becker] |
| 359.9 | Myopathies, without specifying |
Musculo-skeletal disorders
| 723.5 | Torticollis, non-specific |
| 732.1 | Juvenile osteochondritis of the pelvis and hip |
| 732.1 | Plana coxa |
| 732.1 | Legg-Calve-Perthes |
| 732.1 | Scheuermann disease |
| 732.4 | Juvenile osteochondritis of the tibia and peroneus |
| 732.4 | Proximal of the tibia (Blount) |
| 732.4 | Tuberosity of the tibia (Osgood-Schlatter) |
| 732.4 | Vara tibia |
| 736.7 | Other acquired deformities of the limbs |
| 736.71 | Acquired equinovarus deformity |
| 736.79 | Other equine deformities of the foot, acquired |
| 737 | Curvature of the spine |
| 737.1 | Acquired cifosis |
| 737.2 | Acquired lordosis |
| 737.3 | Idiopathic scoliosis |
| 754.1 | Torticollis of the sternocleidomastoid muscle |
| ICD9 | Index by Diagnosis and Condition |
Congenital Anomalies
Congenital Anomalies of the nervous system
| 740.0 | Anencephaly |
| 740.1 | Craniorachischisis |
| 740.2 | Iniencephaly |
| 741 | Spina bifida |
| 741.00 | Spina bifida with hydrocephalia, non-specific region |
| 741.01 | Spina bifida with hydrocephalia, cervical region |
| 741.02 | Spina bifida with hydrocephalia, dorsal (thoracic) |
| 741.03 | Spina bifida with hydrocephalia, lumbar region |
| 741.9 | Spina bifida, non-specified |
| 741.91 | Spina bifida without mentioning hydrocephalia, cervical region |
| 741.92 | Spina bifida without hydrocephalia, dorsal region (thoracic) |
| 741.93 | Spina bifida without hydrocephalia, lumbar region |
| 742.0 | Encephalocele |
| 742.1 | Microcephalia |
| 742.2 | Congenital malformations of the corpus callosum |
| 742.2 | Agenesia of the corpus callosum |
| 742.2 | Arrinencephaly |
| 742.2 | Holoprosencephaly |
| 742.2 | Other hypoplastic anomalies of the encephalus: agenesia, hypoplasia, lisencephaly... |
| 742.3 | Congenital hydrocephalia |
| 742.3 | Malformations of the cerebral aqueduct (“Silvio”): Anomaly, stenosis, obstruction |
| 742.4 | Other congenital malformations of the encephalus |
| 742.4 | Megaencephalia |
| 742.4 | Congenital cerebral cysts: |
| 742.4 | Schizencephaly |
| 742.4 | Porencephaly |
| 742.4 | Macrogiria |
| 742.51 | Diastematomyelia |
| 742.53 | Hydromyelia |
| 742.59 | Other congenital anomalies of the spinal cord |
| 742.8 | Other congenital anomalies of the spinal cord, specific |
| 742.8 | Other congenital anomalies of the nervous system |
| 742.8 | Arnold-Chiari syndrome |
| 742.9 | Congenital anomalies of the brain, spinal cord and nervous system non-specific |
| 743 | Congenital malformations of the eye, of the ear, of the face and the neck |
| 743 | Anophthalmia, microphthalmia and macrophthalmia |
| 743.03 | Cystic ocular globe |
| 743.1 | Microphthalmia |
| 743.2 | Buphthalmos, congenital glaucoma |
| 743.2 | Congenital glaucoma |
| 743.3 | Congenital malformations of the crystalline |
| 743.3 | Congenital cataract |
| 743.35 | Congenital aphaquia |
| 743.36 | Other congenital malformations of the crystalline |
| 743.37 | Congenital displacement of the crystalline |
| 743.39 | Coloboma of the crystalline |
| 743.4 | Congenital malformations of the anterior segment of the eye |
| 743.41 | Anomaly of the size and shape of the cornea |
| 743.42 | Congenital corneal opacity |
| 743.43 | Other congenital malformations of the cornea |
| 743.44 | Other congenital malformations of the anterior segment of the eye |
| ICD9 | Index by Diagnosis and Condition |
| 743.44 | Rieger’s anomaly |
| 743.45 | Absence of iris, Aniridia |
| 743.46 | Coloboma of the iris |
| 743.46 | Other congenital malformations of the iris |
| 743.47 | Blue sclerotia |
| 743.48 | Congenital malformation of the anterior segment of the eye, non-specified |
| 743.51 | Congenital malformations of the posterior segment of the eye |
| 743.51 | Congenital malformation of the vitreous humor |
| 743.52 | Other congenital malformations of the posterior segment of the eye |
| 743.52 | Coloboma of the bottom of the eye |
| 743.53 | Congenital malformation of the choroid |
| 743.56 | Congenital malformation of the retina |
| 743.57 | Congenital malformation of the optic disk |
| 743.57 | Coloboma of the optic disk |
| 743.59 | Congenital malformation of the posterior segment of the eye, non-specified |
| 743.6 | Congenital malformations of the eyelids, of the tear sac and of the orbit |
| 743.61 | Congenital ectropion |
| 743.62 | Congenital entropion |
| 743.62 | Other congenital malformations of the eyelids |
| 743.64 | Absence and agenesia of the lacrimal sac |
| 743.65 | Congenital stenosis and narrowing of the lacrimal conduit |
| 743.65 | Other congenital malformations of the lacrimal sac |
| 743.66 | Congenital malformation of the orbit |
| 743.8 | Other congenital malformations of the eye, specified |
| 743.9 | Congenital malformations of the eye, not specified |
| 744 | Congenital malformations of the ear that alter audition |
| 744.01 | Congenital absence of the pavilion of the ear |
| 744.02 | Congenital absence, atresia or narrowing of the external auditive conduit |
| 744.03 | Other congenital malformations of the middle ear |
| 744.04 | Congenital malformation of the small bones of the ear |
| 744.04 | Fusion of the small bones of the ear |
| 744.05 | Congenital malformation of the internal ear |
| 744.09 | Congenital absence of the ear SAI |
| 744.09 | Congenital absence of the auricular lobule |
| 744.1 | Accessory auricle |
| 744.2 | Other congenital malformations of the ear |
| 744.21 | Other congenital malformations of the ear, specified |
| 744.22 | Macrotia |
| 744.23 | Microtia |
| 744.24 | Absence of the Eustachian tube |
| 744.3 | Congenital malformation of the ear, not specified |
| 744.4 | Sinus, fistula or cyst of the branchial cleft |
| 744.43 | Cervical ear |
| 744.47 | Sinus and preauricular cyst, fistula: |
| 744.49 | Other malformations of the branchial clefts |
| 744.5 | Pterigion of the neck |
| 744.8 | Other specified congenital malformations of the face and neck |
| 744.81 | Macrocheilia |
| 744.82 | Microcheilia |
| 744.83 | Macrostomy |
| 744.84 | Microstomy |
| 744.9 | Congenital malformation of the face and neck, not specified |
| ICD9 | Index by Diagnosis and Condition |
| 745 | Congenital malformations of the circulatory system |
| 745 | Congenital malformations of the chambers of the heart and its connections |
| 745.0 | Common truncus arteriosus |
| 745.0 | Persistence of the truncus arteriosus |
| 745.10 | Transposition (complete) of the large vessels |
| 745.11 | Transposition of the large vessels in right ventricle |
| 745.11 | Taussig-Bing syndrome |
| 745.11 | Transposition of the large vessels in left ventricle |
| 745.12 | Corrected transposition |
| 745.2 | Fallot’s tetralogy |
| 745.3 | Common ventricle |
| 745.3 | Sole ventricle |
| 745.4 | Defect of the ventricular septal |
| 745.4 | Eisenmenger syndrome |
| 745.5 | Defect of the auricular septal |
| 745.5 | Oval hole |
| 745.5 | Ostium secundum (type II) |
| 745.6 | Defect of the aurioventricular septal |
| 745.6 | Defect of the endocardial pillow |
| 745.6f | Defect of the auricular septal ostium primum (type I) |
| 745.69 | Common auriculoventricular channel |
| 745.7 | Biauricular trilocular heart |
| 745.8 | Other congenital malformations of the cardiac septals |
| 745.9 | Congenital malformation of the cardiac septal, unspecified |
| 746 | Congenital malformations of the pulmonary and tricuspid valves |
| 746.00 | Anomaly of the pulmonary valve, unspecified |
| 746.01 | Atresia of the pulmonary valve |
| 746.02 | Congenital stenosis of the pulmonary valve |
| 746.09 | Congenital insufficiency of the pulmonary valve |
| 746.1 | Stenosis, congenital atresia of the tricuspid valve |
| 746.2 | Ebstein’s anomaly |
| 746.3 | Congenital stenosis of the aortic valve |
| 746.4 | Congenital insufficiency of the aortic valve |
| 746.5 | Congenital mitral stenosis |
| 746.6 | Congenital mitral insufficiency |
| 746.7 | Syndrome of left hypoplasia of the heart |
| 746.7 | Syndrome of left hypoplasia of the heart |
| 746.81 | Congenital subaortic stenosis |
| 746.82 | Triauricular heart |
| 746.83 | Stenosis of the pulmonary infundible |
| 746.84 | Other congenital malformations of the heart, specified |
| 746.85 | Malformation of the coronary vessels |
| 746.86 | Congenital heart block |
| 746.87 | Other congenital malformations of the heart |
| 746.87 | Dextrocardia |
| 746.87 | Levocardia |
| 746.89 | Congenital diverticule of the left ventricle |
| 746.9 | Congenital malformation of the heart, unspecified |
| 747 | Congenital malformations of the large arteries |
| 747.0 | Permeable arterius ductus |
| 747.0 | Open Botalli conduit (hole) |
| 747.0 | Persistence of the arterius ductus |
| 747.1 | Coarctation of the aorta |
| ICD9 | Index by Diagnosis and Condition |
| 747.2 | Other anomalies of the aorta |
| 747.21 | Anomaly of the arch of the aorta |
| 747.22 | Atresia and stenosis of the aorta |
| 747.22 | Absence of the aorta |
| 747.22 | Aplasia of the aorta |
| 747.29 | Other congenital malformations of the aorta |
| 747.29 | Aneurism of the Vaisaiva sinus (with rupture) |
| 747.29 | Congenital aunerism |
| 747.3 | Anomalies of the pulmonary artery |
| 747.40 | Congenital malformations of the large veins |
| 747.41 | Total anomalous connection of the pulmonary veins |
| 747.42 | Partial anomalous connection of the pulmonary veins |
| 747.49 | Anomalous connection of the pulmonary veins, without other specification |
| 747.5 | Congenital absence and hypoplasia of the umbilical artery |
| 747.5 | Sole umbilical artery |
| 747.60 | Other congenital malformations of the peripheral vascular system |
| 747.6 | Peripheral arteriovenous malformation |
| 747.62 | Congenital stenosis of the renal artery |
| 747.62 | Other congenital malformations of the renal artery |
| 747.8 | Other congenital malformations of the vascular system, specified |
| 747.81 | Anomalies of the cerebrovascular system |
| 747.82 | Spinal vascular anomaly |
| 747.83 | Persistent fetal circulation |
| 747.9 | Congenital malformation of the vascular system, unspecified |
| 748 | Congenital malformations of the respiratory system |
| 748.0 | Atresia of the choanas |
| 748.1 | Agenesia or hypoplasia and other malformations of the nose |
| 748.2 | Pterygium of the larynx |
| 748.3 | Congenital malformations of the larynx, trachea and bronchii |
| 748.3 | Congenital bronchomalacia |
| 748.4 | Congenital malformations of the lung |
| 748.4 | Congenital pulmonary cyst |
| 748.5 | Agenesia, hypoplasia and dysplasia of the lung |
| 748.5 | Sequestration of the lung |
| 748.6 | Other congenital malformations of the lung |
| 748.61 | Congenital bronchioectasia |
| 748.8 | Other specific anomalies of the respiratory system |
| 749 | Cleft lip and palate |
| 749.00 | Cleft palate |
| 749.01 | Cleft palate, unilateral complete |
| 749.02 | Unilateral cleft palate, incomplete |
| 749.03 | Bilateral cleft palate, complete |
| 749.04 | Bilateral cleft palate, incomplete |
| 749.10 | Cleft lip |
| 749.11 | Cleft lip, unilateral complete |
| 749.12 | Cleft lip, unilateral incomplete |
| 749.13 | Cleft lip, bilateral complete |
| 749.14 | Cleft lip, bilateral incomplete |
| 749.20 | Cleft palate with cleft lip |
| 749.21 | Cleft of the hard palate with cleft lip, unilateral |
| 749.21 | Cleft of the hard palate and of the soft palate with lip |
| 749.22 | Cleft of the soft palate with cleft lip, unilateral |
| 749.23 | Cleft of the hard palate with cleft lip, bilateral |
| ICD9 | Index by Diagnosis and Condition |
| 749.23 | Cleft of the hard palate and of the soft palate with cleft lip, bilateral |
| 749.24 | Cleft of the soft palate with cleft lip, bilateral |
| 749.25 | Cleft of the palate with cleft lip, without other specification |
| 750 | Other congenital malformations of the digestive system |
| 750.0 | Anquiloglosia, short lingual fraenum |
| 750.1 | Other congenital malformations of the tongue |
| 750.15 | Macroglosia |
| 750.2 | Other congenital malformations of the mouth and the pharynx |
| 750.2 | Congenital malformations of the salivary glands and ducts |
| 750.26 | Other congenital malformations of the mouth |
| 750.27 | Pharyngeal diverticula |
| 750.29 | Other congenital malformations of the pharynx |
| 750.3 | Atresia of the esophagus without mention of fistula |
| 750.3 | Atresia of the esophagus with tracheoesophagic fistula |
| 750.3 | Congenital tracheoesophagic fistula without mention of atresia |
| 750.3 | Congenital narrowness or stenosis of the esophagus |
| 750.4 | Congenital malformations of the esophagus |
| 750.4 | Pterigion of the esophagus, congenitally dilated esophagus, diverticula, duplication |
| 750.5 | Congenital hypertrophic pyloric stenosis |
| 750.6 | Congenital hiatal hernia |
| 750.7 | Other congenital malformations of the stomach, specified |
| 750.8 | Other congenital malformations of the top part of the digestive tube |
| 751.0 | Meckel diverticula, persistence of the duct |
| 751.1 | Congenital absence, atresia and stenosis of the small intestine |
| 751.1 | Congenital absence, atresia and stenosis of the duodenum |
| 751.1 | Congenital absence, atresia and stenosis of the jejunum |
| 751.2 | Congenital absence, atresia and stenosis of the large intestine, unspecified part |
| 751.2 | Congenital absence, atresia and stenosis of the rectum and anus |
| 751.3 | Hirschsprung disease, Aganglionosis, congenital megacolon (aganglionar) |
| 751.5 | Other congenital malformations of the intestine |
| 751.6 | Congenital malformations of the gallbladder, of the biliar ducts and the liver |
| 751.61 | Agenesia, aplasia and hypoplasia of the gallbladder |
| 751.61 | Atresia of the biliar ducts |
| 751.62 | Cystic disease of the liver |
| 751.69 | Cyst of the choledocal |
| 751.7 | Agenesia, aplasia and hypoplasia of the pancreas |
| 751.7 | Anular pancreas |
| 751.7 | Congenital cyst of the pancreas |
| 751.9 | Other congenital malformations of the digestive system |
| 752 | Congenital malformations of the genital organs |
| 752.0 | Anomalies and congenital absence of ovary |
| 752.1 | Congenital malformations of the Eustachian tubes and of the broad ligaments |
| 752.2 | Congenital malformations of the uterus |
| 752.2 | Duplication of the uterus with duplication of the uterine neck and of the vagina |
| 752.3 | Agenesia and aplasia of the uterus and Other anomalies of the uterus |
| 752.3 | Other congenital malformations of the uterus |
| 752.40 | Anomalies of the uterine neck, vagina and external feminine genitalia |
| 752.41 | Embryonic cyst of the uterine neck |
| 752.42 | Unperforated hymen |
| 752.49 | Agenesia and aplasia of the uterine neck |
| 752.49 | Other congenital malformations of the feminine genital organs |
| 752.49 | Congenital absence of the vagina |
| 752.51 | Cryptordchidism |
| 752.6 | Hypospadias, epispadias and other anomalies of the penis |
| ICD9 | Index by Diagnosis and Condition |
| 752.64 | Aplasia and congenital absence of the penis |
| 752.69 | Other congenital malformations of the penis |
| 752.7 | Indeterminate sex and pseudohermaphroditism |
| 752.7 | Indeterminate sex, without other specification, ambiguous genitals |
| 752.8 | Other congenital malformations of the masculine genital organs |
| 752.8 | Other congenital malformations of the deferent ducts, of epididymis |
| 753 | Congenital malformations of the urinary system |
| 753.0 | Renal agenesia and other hypoplastic malformations of the kidney |
| 753.0 | Renal agenesia, unilateral |
| 753.0 | Renal agenesia, bilateral |
| 753.0 | Renal agenesia, without other specification |
| 753.0 | Renal hypoplasia, unilateral |
| 753.0 | Renal hypoplasia, bilateral |
| 753.0 | Renal hypoplasia, not specified |
| 753.0 | Potter syndrome |
| 753.1 | Polycystic kidney, infantile type |
| 753.11 | Solitary renal cyst, congenital |
| 753.12 | Polycystic kidney, unspecified type |
| 753.15 | Renal displasia |
| 753.16 | Medular cystic kidney |
| 753.17 | SAI spongioid kidney |
| 753.19 | Other cystic renal diseases |
| 753.2 | Congenital obstructive defects of the pelvis, renal and congenital malformations of the ureter |
| 753.23 | Other obstructive defects of the renal pelvis and the ureter |
| 753.23 | Congenital ureterocele |
| 753.29 | Congenital hydronephrosis |
| 753.29 | Atresia and stenosis of the ureter |
| 753.29 | Congenital megaloureter |
| 753.29 | Agenesia of the ureter |
| 753.29 | Duplication of the ureter |
| 753.29 | Bad position of the ureter |
| 753.29 | Congenital vesico-ureteral-renal reflux |
| 753.3 | Other congenital malformations of the kidney |
| 753.3 | Supernumerary kidney |
| 753.3 | Lobulated, fused and horseshoe kidney |
| 753.3 | Ectopic kidney |
| 753.3 | Renal hyperplasia and giant kidney |
| 753.4 | Other specific anomalies of the ureter |
| 753.5 | Bladder exstrophy |
| 753.6 | Congenital posterior urethral valves |
| 753.6 | Other atresias and stenosis of the urethra and bladder neck |
| 753.7 | Anomalies of the urachus |
| 753.8 | Congenital absence of the bladder and of the urethra |
| 753.8 | Congenital diverticula of the bladder |
| 753.8 | Other congenital malformations of the bladder and the urethra |
| 754 | Congenital malformations and deformities of the osteomuscular system |
| 754.0 | Congenital osteomuscular deformities of the head, of the face |
| 754.0 | Facial asymmetry |
| 754.0 | Compressed fancies |
| ICD9 | Index by Diagnosis and Condition |
| 754.0 | Dolicocephalia |
| 754.0 | Plagiocephalia |
| 754.0 | Other congenital deformities of the cranium, of the face and of the jaw |
| 754.0 | Congenital flattening of the nose |
| 754.0 | Hemifacial atrophy or hypertrophy |
| 754.0 | Depressions in the cranium |
| 754.0 | Congenital deviation of the nasal septum |
| 754.10 | Congenial torticollis |
| 754.2 | Congenital deformity of the vertebral column |
| 754.2 | Congenital scoliosis: |
| 754.3 | Congenital deformities of the hip |
| 754.30 | Congenital luxation of the hip, unilateral |
| 754.3 | Congenital acetabular displasia |
| 754.31 | Congenital luxation of the hip, bilateral |
| 754.32 | Congenital subluxation of the hip, unilateral |
| 754.33 | Congenital subluxation of the hip, bilateral |
| 754.35 | Unstable hip |
| 754.4 | Congenital deformity of the knee |
| 754.4 | Congenital recurvatum genu |
| 754.41 | Congenital luxation of the knee |
| 754.42 | Congenital curvature of the femur |
| 754.43 | Congenital curvature of the tibia and the perone |
| 754.44 | Congenital curvature of the long bones de the lower limb, without other specification |
| 754.5 | Congenital deformity of the feet |
| 754.51 | Talipes equinovarus |
| 754.53 | Metatarsus varus |
| 754.59 | Other congenital varus deformities of the feet |
| 754.61 | Congenital piano foot |
| 754.62 | Calcaneovalgus talipes |
| 754.69 | Valgus metatarsus |
| 754.71 | Cavus foot |
| 754.79 | Calcaneovarus talipes |
| 754.79 | Congenital varus hallux |
| 754.79 | Other congenital deformities of the feet |
| 754.81 | Pectus excavatum |
| 754.81 | Pectus curvatum |
| 754.82 | Pectus carinatum (shaped like the keel of a boat) |
| 754.82 | Pectus carinatum (pigeon chest) |
| 754.89 | Other congenital deformities of the extremities |
| 754.89 | Congenital arthrogriposis multiple |
| 754.89 | Congenital deformed finger |
| 754.89 | Hand on shovel (congenital) |
| 755.0 | Polydactyly |
| 755.02 | Supernumerary toe(s) of the foot |
| 755.1 | Sindactyly |
| 755.13 | Interdigital membrane of the foot |
| 755.14 | Fusion of the toes of the foot |
| 755.2 | Defects due to reduction of the superior extremity |
| 755.21 | Complete congenital absence of the limb(s) |
| ICD9 | Index by Diagnosis and Condition |
| 755.23 | Congenital absence of the forearm and the hand |
| 755.26 | Defect due to longitudinal reduction of the radius |
| 755. 27 | Defect due to longitudinal reduction of the cubit |
| 755.29 | Congenital absence of the hand and the finger(s) |
| 755.3 | Defects due to reduction of the lower limb(s) |
| 755.3 | Other defects due to reduction of the lower limb(s) |
| 755.31 | Congenital complete absence of the lower limb(s) |
| 755.34 | Defect due to longitudinal reduction of the femur |
| 755.35 | Defect due to longitudinal reduction of the tibia |
| 755.37 | Defect due to longitudinal reduction of the peroné |
| 755.4 | Other defects due to reduction of the superior limb(s) |
| 755.4 | Complete absence of the non-specified limb(s) |
| 755.4 | Phocomelia, non-specified limb(s) |
| 755.5 | Other congenital malformations of the superior limb(s), including the shoulder girdle |
| 755.54 | Deformity of: |
| 755.56 | Supernumerary bones of the carpus |
| 755.57 | Macrodactyly (fingers of the hand) |
| 755.58 | Lobster claw hand |
| 755.59 | Cleidocranial dysostosis |
| 755.59 | Triphalangic thumb |
| 755.6 | Other congenital malformations of the inferior limb(s), including the pelvis girdle |
| 755.64 | Congenital malformation of the knee |
| 756.0 | Congenital malformations of the bones of the cranium and of the face |
| 756.0 | Craniosynostosis |
| 756.0 | Acrocephalia |
| 756.0 | Imperfect fusion of the cranium |
| 756.0 | Oxycephaly |
| 756.0 | Trigonocephaly |
| 756.0 | Craniofacial dysostosis |
| 756.0 | Crouzon disease |
| 756.0 | Hypertelorism |
| 756.0 | Macrocephaly |
| 756.0 | Maxillofacial dysostosis |
| 756.0 | Oculomaxillar dysostosis |
| 756.0 | Absence of bone(s) of the cranium, congenital |
| 756.0 | Congenital deformity of the forehead |
| 756.0 | Platybasia |
| 756.1 | Congenital malformations of the vertebral column and the bony thorax |
| 756.10 | Anomalies of the vertebral column, without specifying |
| 756.11 | Spondylolysis, L-S |
| 756.12 | Congenital spondylolystesis |
| 756.14 | Hemivertebra, congenital lordosis |
| 756.15 | Cervical fusion syndrome |
| 756.16 | Klippel-Feil syndrome |
| 756.17 | Spina bifida occulta |
| 756.2 | Cervical rib |
| 756.3 | Congenital malformation of the sternum |
| 756.4 | Osteochondrodysplasia with growth defect |
| 756.4 | Acondrogenesis |
| 756.4 | Tanatophoric dwarfism |
| 756.4 | Achondroplasia |
| 756.51 | Osteopetrosis |
| 756.52 | Other specified ostechondrodiyplasias; Osteopoichylosis |
| 756.54 | Fibrous polyostotic dysplasia |
| ICD9 | Index by Diagnosis and Condition |
| 756.55 | Chondroectodermic dysplasia, Ellis-van Creveld syndrome |
| 756.56 | Progressive diafisaria displasia |
| 756.56 | Metafissary dysplasia |
| 756.59 | Other osteochondrodysplasias |
| 756.59 | Albright syndrome (-McCune)(-Sternberg) |
| 756.6 | Congenital malformations of the diaphragm |
| 756.6 | Absence |
| 756.6 | Eventration |
| 756.71 | Fructose malabsorption |
| 756.79 | Exomphalos |
| 756.79 | Omphalocele |
| 756.79 | Gastroschisis |
| 756.79 | Other congenital malformations of the abdominal wall |
| 756.83 | Ehlers-Danlos syndrome |
| 757 | Congenital malformations of the skin, hair and nails |
| 757.0 | Hereditary Lymphedema |
| 757.1 | Congenital ichthyosis |
| 757.1 | Vulgar ichthyosis |
| 757.1 | Ichthyosis linked to chromosome X |
| 757.1 | Lamellar ichthyosis |
| 757.1 | Colloidon baby |
| 757.1 | Congenital vesicular ichthyoiform eritrodermia |
| 757.1 | Harlequin fetus |
| 757.2 | Other congenital malformations of the skin, specified |
| 757.31 | Ectodermic dysplasia (anhydrotic) |
| 757.32 | Vascular hamartomas, non-neoplasic nevus, congenital |
| 757.33 | Other congenital malformations of the skin |
| 757.33 | Pigmentous congenital anomalies, pigmentous xeroderma |
| 757.33 | Mastocytosis, pigmentous urticary |
| 757.39 | Epidermolysis bullosa |
| 757.39 | Supernumerary cutaneous appendices |
| 757.4 | Congenital alopecia, other congenital malformations of the hair |
| 757.5 | Anonychia, other congenital malformations of the nails |
| 757.6 | Congenital malformations of the mamma |
| 759 | Other non-specific congenital anomalies |
| 759.0 | Congenital malformations of the spleen |
| 759.0 | Asplenia (congenital) |
| 759.0 | Congenital splenomegaly |
| 759.1 | Congenital malformations of the adrenal glands |
| 759.2 | Congenital malformations of the other endocrine glands |
| 759.2 | Persistent thyroglosal duct |
| 759.2 | Congenital malformation of thyroid or parathyroid gland |
| 759.2 | Thyroglosal cyst |
| 759.3 | Situs inversus |
| 759.3 | Dextrocardia with situs inversus |
| 759.3 | Auricular disposition in mirror image with situs inversus |
| 759.3 | Situs inversus or transversus |
| 759.3 | Transposition of bowels |
| 759.4 | Siamese twins |
| 759.5 | Tuberous sclerosis |
| 759.6 | Other congenital hamartosis, without classifying |
| 759.6 | Peutz-Jeghers |
| 759.6 | Sturge-Weber |
| ICD9 | Index by Diagnosis and Condition |
| 759.7 | Multiple congenital anomalies, as described |
| 759.81 | Prader Willi syndrome |
| 759.82 | Marfan’s syndrome |
| 759.83 | Fragile X syndrome |
| 759.89 | Other syndromes of congenital malformations |
| 758.89 | Russell-Silver syndrome |
| 759.89 | Alport syndrome |
| 759.89 | Lawrence-Moon-(Bardet)- Biedl syndrome |
| 759.89 | Zeilweger syndrome |
| 759.89 | Carpenter’s syndrome |
| 759.89 | Angleman’s syndrome |
| 759.89 | Jarcho-Levin syndrome |
| 758 | Chromosomic anomalies; unclassified in another part |
| 758.0 | Down syndrome |
| 758.0 | Trisomy 21, due to lack of meioitic disjunction |
| 758.0 | Trisomy 21, mosaic (due to lack of mitotic disjunction) |
| 758.0 | Trisomy 21, due to translocation |
| 758.0 | Down syndrome, unspecified |
| 758.1 | Edwards syndrome |
| 758.1 | Trisomy 18, due to lack of meioitic disjunction |
| 758.1 | Trisomy 18, mosaic (due to lack of mitotic disjunction) |
| 758.1 | Trisomy 18, due to translocation |
| 758.2 | Patau syndrome |
| 758.2 | Trisomy 13, due to lack of meioitic disjunction |
| 758.2 | Trisomy 13, mosaic (due to lack of mitotic disjunction) |
| 758.2 | Trisomy 13, due to translocation |
| 758.3 | Other suppressions on the part of an autosomic chromosome |
| 758.3 | Suppression of the short arm of chromosome 4 |
| 758.3 | Wolff-Hirschorn syndrome |
| 758.31 | Suppression of the short arm of chromosome 5 |
| 758.31 | Criduchat syndrome |
| 758.32 | Velo-cardio-facial syndrome |
| 758.5 | Other conditions due to anomalies in autosomic chromosomes |
| 758.5 | Suppression of the autosomes, unspecified |
| 758.6 | Turner syndrome |
| 758.7 | Klinefelter’s syndrome |
| 758.6 | Caryotype 45, X |
| 758.81 | Other conditions resulting from anomalies in sexual chromosomes |
| 758.89 | Other conditions resulting from anomalies in non-specific chromosomes |
| 760 | Conditions in the perinatal period |
| 760.71 | Fetal syndrome due to consumption of alcohol |
| 765.00 | Prematurity |
| 767.6 | Injury of the brachial plexus |
| 768.9 | Perinatal hypoxia, asphyxia or anoxia |
| 772.1 | Intraventricular hemorrhage (Grade III-IV) |
| 774.7 | Kernicterus |
| 779.7 | Cystic periventricular leukomalacia |
| 779.7 | Cystic periventricular leukomalacia |
Disorders of the Sensory organs
| 360 | Disorders of the eye and attachments |
| 362.2 | Retinopathy of the Premature |
| 369 | Blindness and loss of vision |
| ICD9 | Index by Diagnosis and Condition |
| 369.2 | Moderate to severe blindness, both eyes |
| 369.4 | Legal blindness |
| 369.6 | Blindness in one eye (the other one is normal) |
| 378.0 | Strabism (alternating, congenital, non-paralytic) |
| 378.00 | Esotropia, non-specific |
| 378.10 | Exotropia |
| 378.6 | Mechanic |
| 378.60 | Paralytic |
| 378.71 | NCOP specified (Duane’s syndrome) |
| 389 | Loss of hearing |
| 389.00 | Bilateral conductive loss of hearing |
| 389.10 | Neurosensorial loss of hearing, non-specific |
| 389.2 | Conductive and neurosensorial loss of hearing, mixed |
| 478.4 | Polyps in the vocal chords |
| 784.4 | Alterations in the voice |
| 784.41 | Aphonia |
| 748.49 | Dysphonia |
Burns and traumas
| 709.2 | Scars and fibrosis of the skin |
| 709.2 | Disfiguring scar |
| 709.2 | Disabling scar |
| 906.9 | Delayed defects of burns |
| 949.0 | Burns and corrosions |
| 952.9 | Damage to the spinal cord |
Lack of normal physiological development
| 783.4 | Delays in physiological development, not specific |
| 783.41 | Failure to thrive, failure in gaining weight |
| 783.42 | Delay in general development (non-specific area) |
| 783.43 | Short height, failure in growth |
Bronchial asthma
| 493.00 | Asthma |
| 493.0 | Extrinsec predominantly allergic asthma |
| 493.1 | Non-allergic asthma |
| 493.9 | Asthma, non-specified |
Mental and Conduct disorders
| 295.00 | Schizophrenia |
| 296.0 | Depression |
| 298.9 | Psychosis |
| 299.0 | Autism |
| 300.9 | Neurosis |
| 300.9 | Self-damaging conduct (suicidal conduct) |
| 312.00 | Conduct disorders (conduct disorders in children and adolescents) |
| 313.81 | Oppositional defiant disorder |
| 314.00 | Activity and attention disorder (ADD) |
| 314.01 | Attention deficit with hyperactivity |
| 315 | Developmental delays and disorders |
| 315.3 | Disorder in language development |
| 315.4 | Delay in motor development and coordination |
| ICD9 | Index by Diagnosis and Condition |
| 315.5 | Delay in development, mixed |
| 315.9 | Delay in development, not specific |
Mental Retardation
| 317.00 | Slight, intellectual coefficient of 50 to 70 |
| 318.0 | Moderate, intellectual coefficient of 35 to 49 |
| 318.1 | Severe, intellectual coefficient of 20 to 34 |
| 318.2 | Deep, intellectual coefficient under 20 |
| 319.0 | Mental retardation, not specified |
| 319.0 | William syndrome |
Endocrine and nutritional disorders
| 243.0 | Congenital hypothyroidism 224.0 244.9 Acquired hypothyroidism |
| 246.8 | Other specific disorders of the thyroid gland |
| 250.01 | Insulin Dependent Diabetes Mellitus, Type I, Juvenile |
| 250.02 | Juvenile non-insulin dependent Diabetes Mellitus |
| 252.0-252.08 | Hyperparathyroidism 252.1 Hypoparathyroidism |
| 252.8 | Other specific disorders of the parathyroid gland |
| 253.0 | Acromegalia and giantism |
| 253.3 | Dwarfism due to deficiency of the growth hormone |
| 255.2 | Adrenal congenital hyperplasia |
| 255.8 | Other specific disorders of the adrenal glands |
| 259.1 | Precocious sexual development, precocious puberty |
| 259.4 | Dwarfism, NOS |
| 278.01 | Morbose obesity |
Immunological and hematological disorders
| 42 | Disease of the Human Immunodeficiency Virus (HIV) |
| 279.0 | Deficiency of humoral immunity |
| 279.1 | Deficiency of cellular immunity |
| 279.3 | Deficiency of non-specific immunity |
| 279.4 | Autoimmune disorder, not classified |
| 282.4 | Major Thalassemia |
| 282.6 | Sickle cell anemia |
| 283.9 | Hemolitic anemia |
| 284.9 | Aplastic anemia |
| 286.0 | Hemophilia |
| 279.2 | Combined immunity deficiency |
| 710 | Diseases of the connective tissue and collagen |
| 710 | Systemic eritematous lupus |
| 710.1 | Sclerosis, scleroderma |
| 710.2 | Sicca Syndrome |
| 710.3 | Dermatomyositis |
| 710.4 | Poliomyositis |
| 714 | Juvenile rheumatoid arthritis |
Cancer and Tumors
| 140-239 | Neoplasms |
Malignant tumors
Invasive tumors
| 208.9 | Leukemia |
ATTACHMENT 14
Guidelines for the Development of Program Integrity Plan
2014 -2015
(This document is to be used by all contracted companies participating in the Government Health Plan of the Commonwealth of Puerto Rico. The purpose of sharing information with contracted companies is to provide them guidelines with minimum requirements to formulate their own Plan Integrity Program for the Health Care Delivery System sponsored by the Commonwealth of Puerto Rico)
The Insurer shall comply with the following Medicaid Integrity requirements:
| A. | 60 days after the dated of the agreement the Company must submit to ASES Compliance Office copy of the policies and procedures for identifying and tracking potential provider fraud cases, for conducting preliminary and full investigation and for referring cases of suspected fraud to an appropriate law enforcement agency. The Compliance Plan should be developed in accordance with 42 CFR 438.608. |
| B. | Each company must submit to the Administration's Compliance Office on a quarterly basis a report with the following information: preliminary and full investigations, audits performed, administrative actions against providers, overpayments identified and providers referred to the Department of Justice (if not submit a certification signed by the Compliance Director and the President or CEO). |
| C. | Each company must submit to the Compliance Office on a quarterly basis a report with the following information: fraud investigations pending, fraud investigations in process, fraud investigations finished and referrals to the Department of Justice or U.S. Attorney's Field Office (if there were no investigations, submit a certifications signed by the Compliance Director and the President or CEO). |
| D. | Each Company has five (5) days to notify ASES about the referrals made to the US Attorney's Field Office and HHS-OIG. |
| E. | Each company must submit to the Compliance Office a certification signed by the Compliance Director and the President or CEO indicating that all full investigations were made in accordance with 42 CFR 455.15. |
| F. | Each Company has five (5) days to notify ASES about any adverse or negative action that the MCO has taken on provider application (upon initial application or application renewal) or actions which limit the ability of providers to participate in the program. |
| G. | Each Company must review the credentialing forms of all providers and any fiscal agents they may use to ensure that they are in accordance with federal regulation 42 CFR 455.104. |
| H. | Each Company must require providers to fill out a complete ownership and control disclosures form. The Company is responsible to ensure compliance with regulation. |
| I. | Each Company must review providers agreement to incorporate appropriate business transaction language to ensure accordance with federal regulation 42 CFR 455.105. |
| J. | Each Company must request providers to fulfill a business transactions form and verify compliance with regulation. |
| K. | Each Company must establish a method to capture criminal conviction information on owners, persons with control interest, agents, and managing employees of providers to ensure that is in accordance with federal regulation 42 CFR 455.106. |
| L. | Each Company must review the enrollment packages for all provider types to request criminal conviction information as stated before. |
| M. | Each Company should develop and implement procedures to report to HHS-OIG and ASES within 20 working days any criminal conviction disclosures made during the MCO credentialing process. Copy of the policies should be submitted to ASES Compliance Office. |
| N. | Each Company must submit to the Compliance Office a certification signed by the Compliance Director and the President or CEO stating compliance with 42 CFR 455.106. |
| O. | Each Company must comply with requirement in 42 CFR 455.20 and must document in a quarterly report compliance with regulation. |
| P. | Each Company must comply with requirement in 42 CFR 455.101. |
| Q. | Each Company must review the enrollment form and credentialing packages for all provider types to capture the identity of agents and managing employees. |
TABLE OF CONTENTS
|
Integrity Program Basis and Scope
|
|
Definitions
|
|
Other applicable regulations
|
|
Guidelines for Sub-Parts A, B
|
|
Sub-Part A: Fraud Detection and Investigation Program
|
|
PI A001: State plan requirement. § 455.12
|
|
PI A002: Methods for identification, investigation, and referral. § 455.13
|
|
PI A003: Preliminary investigation. § 455.14
|
|
PI A004: Full investigation. § 455.15
|
|
PI A005: Resolution of full investigation. § 455.16
|
|
PI A006: Reporting requirements. § 455.17
|
|
PI A007: Provider's statements on claims forms. § 455.18
|
|
PI A008: Provider's statement on check. § 455.19
|
|
PI A009: Recipient verification procedure. § 455.20
|
|
PI AO 10: Cooperation with State Medicaid fraud control units. § 455.21
|
|
PI A011: Withholding of payments in cases of fraud or willful misrepresentation
|
|
(§ 455.23)
|
|
Sub-Part B: Disclosure of Information by Providers and Fiscal Agents
|
|
PI B001: Purpose § 455.100
|
|
PI B002; Definitions.§ 455.101
|
|
PI B003: Determination of ownership or control percentages.§ 455.102
|
| PI B004: State Plan requirements § 455.103 |
|
PI B005: Disclosure by providers and agents: Information on ownership and control. § 455.104
|
|
PI B006: Disclosure by providers: Information related to business transactions. § 455.105
|
|
PI B007: Disclosure by providers: Information on persons convicted of crimes. § 455.106
|
|
Sub-Part E:
|
|
PI E001: Termination or denial of enrollment § 455.416
|
|
PI E002: Reactivation of provider enrollment § 455.420
|
|
PI E003: Appeal rights. § 455.422
|
|
PI E004: Criminal background checks. §455.434
|
|
Other
|
|
State Medicaid Directors Letter (SMDL) #09-001
|
Introduction
Under the authority of Sec. 1102 of the Social Security Act (42 U.S.C. 1302); as detailed in the 43 FR 45262, Sept. 29, 1978, the Medicaid Program must have a program to detect and investigate fraud, waste and abuse.
The Commonwealth of Puerto Rico Department of Health and its Office for the Medically Indigent, acting as the single state agency are responsible for the management of the Medicaid and SCHIP grant funds. These funds are transferred to the Puerto Rico Health Insurance Administration (ASES), to be combined with state funds to provide health benefit coverage to the medically indigent population under a managed care fully capitated health plan. Acting as a sub-grantee to the Office for the Medically Indigent Medicaid program, ASES establishes contracts with insurance companies and other organizations to facilitate the beneficiaries' access to the benefit coverage throughout their provider's networks.
Integrity Program Basis and Scope
This document sets forth guidelines with minimum criteria for the compliance with Program Integrity Policies and Procedures that each organization (grantee, sub-grantee, insurance companies) must have for the administration of the Commonwealth of Puerto Rico's Medicaid and State Health Plans. This document includes guidelines for the elaboration of the 3 main sections in the organizations Program Integrity Plan (PIP):
| 1. | Fraud Detection and Investigation |
| 2. | Providers and Fiscal Agents Disclosure of Information on Ownership and Control |
| 3. | Integrity Program |
Regulation Citation
Sections 1902(a)(4) [42 USC 1396(a)(4)l, (61)2, (64)3); 1903(i)(2) [42 USC 1396(b)(i)(2)]4 1936[42 USC 1396u-6]5) and regulations at 42 CFR Parts 438,455,1001 and 1002
Overall Requirement
All providers/contractors are required to comply with the CMS Medicaid Integrity Group State Medicaid Director Letters #08-003 and #09-001, which explain what all states and contractors should do in terms of checking for excluded parties. The letters provide guidance on where to check for excluded individuals as well as the consequences of contracting with individuals and entities that have been excluded from participating in federally funded programs.
Companies are also required to notify to the Department of Health and Human Services- Office of Inspector General (HHS-OIG) of any action it takes to limit the ability of an individual or entity to participate in its program as stated in 42 CFR 1002.3.
Each contracted company must report actions it takes when it denies a provider enrollment based on program integrity concerns. Companies should report on each provider whom it has disenrolled, suspended, terminated or otherwise restricted from participation in the Medicaid program based on program integrity concerns. Companies are required to report affected providers directly to HHS-OIG while copying ASES.
Definitions
Abuse means provider practices that are inconsistent with sound fiscal, business, or medical practices, and result in an unnecessary cost to the Medicaid program, or in reimbursement for services that are not medically necessary or that fail to meet professionally recognized standards for health care. It also includes recipient practices that result in unnecessary cost to the Medicaid program.
Agent means any person who has been delegated the authority to obligate or act on behalf of a provider
Conviction or Convicted means that a judgment of conviction has been entered by a Federal, State, or local court, regardless of whether an appeal from that judgment is pending.
Disclosing Entity means a Medicaid provider (other than an individual practitioner or group of practitioners) or a fiscal agent
Exclusion means that items or services furnished by a specific provider who has defrauded or abused the Medicaid program will not be reimbursed under Medicaid.
Fiscal agent means a contractor that processes or pays vendor claims on behalf of the Medicaid agency.
Fraud means an intentional deception or misrepresentation made by a person with the knowledge that the deception could result in some unauthorized benefit for him/her or some other person. It includes any act that constitutes fraud under applicable Federal or State law.
Furnished refers to items and services provided directly by, or under the direct supervision of, or ordered by, a practitioner or other individual (either as an employee ^ or in his or her own capacity), a provider, or other supplier of services. (For purposes of denial of reimbursement within this part, it does not refer to services ordered by one party but billed for and provided by or under the supervision of another.)
Group of practitioners means two or more health care practitioners who practice their profession at a common location (whether or not they share common facilities, common supporting staff, or common equipment).
Health insuring organization (HIO) has the meaning specified in §438.2.
Indirect ownership interest means an ownership interest in an entity that has an ownership interest in the disclosing entity. This term includes an ownership interest in any entity that has an indirect ownership interest in the disclosing entity.
Managing employee means a general manager, business manager, administrator, director, or other individual who exercises operational or managerial control over, or who directly or indirectly conducts the day-to-day operation of an institution, organization, or agency.
Other disclosing entity means any other Medicaid disclosing entity and any entity that does not participate in Medicaid, but is required to disclose certain ownership and control information because of participation in any of the programs established under title V, XVIII, or XX of the Act. This includes:
| (a) | Any hospital, skilled nursing facility, home health agency, independent clinical laboratory, renal disease facility, rural health clinic, or health maintenance organization that participates in Medicare (title XVIII); |
| (b) | Any Medicare intermediary or carrier; and |
| (c) | Any entity (other than an individual practitioner or group of practitioners) that furnishes, or arranges for the furnishing of, health-related services for which it claims payment under any plan or program established under title V or title XX of the Act. |
Person with an ownership or control interest means a person or corporation that—
| (a) | Has an ownership interest totaling 5 percent or more in a disclosing entity; |
| (b) | Has an indirect ownership interest equal to 5 percent or more in a disclosing entity; |
| (c) | Has a combination of direct and indirect ownership interests equal to 5 percent or more in a disclosing entity; |
| (d) | Owns an interest of 5 percent or more in any mortgage, deed of trust, note, or other obligation secured by the disclosing entity if that interest equals at least 5 percent of the value of the property or assets of the disclosing entity; |
| (e) | Is an officer or director of a disclosing entity that is organized as a corporation; or |
| (f) | Is a partner in a disclosing entity that is organized as a partnership. |
Practitioner means a physician or other individual licensed under State law to practice his or her profession.
Program Integrity Plan (PIP) means the program, process or policy that each contracted company has to comply with integrity requirements. The plan should be developed in accordance with federal regulation.
Significant business transaction means any business transaction or series of transactions that, during any one fiscal year, exceed the lesser of $25,000 and 5 percent of a provider's total operating expenses.
Subcontractor means-
| (a) | An individual, agency, or organization to which a disclosing entity has contracted or delegated some of its management functions or responsibilities of providing medical care to its patients; or |
| (b) | An individual, agency, or organization with which a fiscal agent has entered into a contract, agreement, purchase order, or lease (or leases of real property) to obtain space, supplies, equipment, or services provided under the Medicaid agreement. |
Supplier means an individual, agency, or organization from which a provider purchases goods and services used in carrying out its responsibilities under Medicaid (e.g., a commercial laundry, a manufacturer of hospital beds, or a pharmaceutical firm).
Stakeholder means the single state agency, the sub-grantee and all organizations contracted to provide health care management and services to Medicaid beneficiaries
Suspension means that items or services furnished by a specified provider who has been convicted of a program-related offense in a Federal, State, or local court will not be reimbursed under Medicaid.
Termination means—
|
(1)
|
For a—
|
| (i) | Medicaid or CHIP provider, a State Medicaid program or CHIP has taken an action to revoke the provider's billing privileges, and the provider has exhausted all applicable appeal rights or the timeline for appeal has expired; and |
| (ii) | Medicare provider, supplier or eligible professional, the Medicare program has revoked the provider or supplier's billing privileges, and the provider has exhausted all applicable appeal rights or the timeline for appeal has expired. |
| (2) | (i) In all three programs, there is no expectation on the part of the provider or supplier or the State or Medicare program that the revocation is temporary. |
(ii) The provider, supplier, or eligible professional will be required to reenroll with the applicable program if they wish billing privileges to be reinstated.
(3) The requirement for termination applies in cases where providers, suppliers, or eligible professionals were terminated or had their billing privileges revoked for cause which may include, but is not limited to—
(i) Fraud;
(ii) Integrity; or
(iii) Quality.
Wholly owned supplier means a supplier whose total ownership interest is held by a provider or by a person, persons, or other entity with an ownership or control interest in a provider
Section A
Fraud Detection and Investigation sub part represents each one of the elements that must be included as part of the integrity program activities, although they are not necessarily the only elements that come into play.
All contracted plans must have an integrity program with their own structure, policies and procedures. Among other areas, they should have written policies and procedures on methods for the identification, investigation and referral of suspected cases; procedure to perform preliminary investigations as well as full investigations; procedures to address resolution of full investigations; procedures to comply with reporting requirements; provider's statements on claims form (if applicable); provider's statement on checks; cooperation with the Commonwealth of Puerto Rico Office for the Medically Indigent fraud control unit and procedure to withhold payments in case of fraud or willful misrepresentation. Contracted companies are required to submit to ASES Compliance Office copy of their integrity programs for evaluation. The plan should be developed in accordance with 42 CFR 438.608.
Each one of the Guidelines under section A includes the name or title of the guideline, scope, purpose, process and general information to identify the creation date, creator, and revisions or updates. This document will be attached to the contract each organization holds with the Puerto Rico Insurance Administration; while each one of the contracted organization should have at least a minimum set of policies and procedures to address the guidelines included.
The Program Integrity Plan (PIP) of each organization is to be monitored by the sub- grantee on periodic basis. An annual report will be issued reporting data and findings.
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA1.1
|
State Plan Requirements
|
|||
|
Scope
|
Applies to Single State Agency and Sub-Grantee
|
|||
|
Purpose
|
This guideline describes the commitment of the single state agency and the sub-grantee in adhering to the statue rules and regulations and the implementation of a Program Integrity Plan for the Medicaid Program
|
|||
|
General
|
The grantee and the sub-grantee will abide bye the following guidelines on how to manage the integrity program activities in the whole service delivery system.
|
|||
|
Guidelines
|
The PIP must include an explicit definition of methods to perform identification of cases suspected of fraud, waste and abuse
|
|||
|
1.
|
The single state agency and sub-grantee acknowledge the need to adhere to a Medicaid Integrity Program as defined in the state plan.
|
|||
|
2.
|
The grantee and sub-grantee agree to establish a structure to manage Program Integrity Plan (PIP) activities.
|
|||
|
3.
|
The organization structure to perform above mentioned activities is furnished with a Program Integrity Plan (PIP) of members representing the single state agency, the sub-grantee and each contracted organization.
|
|||
|
4.
|
The PIP leads the efforts toward achieving compliance with state plan requirements regulation by establishing the minimum criteria of required PI program policies and procedures.
|
|||
|
5.
|
The PIP monitors contracted companies plan compliance on regular basis.
|
|||
|
6.
|
The PIP chairman develops the meeting calendar each year, develops the committee agenda, and keeps minutes of all meetings and call for meetings.
|
|||
|
7.
|
Sub-grantee facilitates the development and update of the Program Integrity Plan guidelines, reports and notification to guarantees its distribution and final acceptance among contracted companies and regulatory agencies.
|
|||
|
8.
|
Sub-grantee review performance of each organization, level of adherence to policies and recommend corrective action plan development for areas that must be improved.
|
|||
|
9.
|
Sub-grantee develops an annual report that is to be submitted to the Medicaid Program Integrity Group and to the CMS region 2. The report will include the areas and companies reviewed during the period and the findings of each company, if any.
|
|||
|
10.
|
The PIP provides guidance and guarantees that each contracted companies develop and implement policies and procedures in their organizations.
|
|||
|
11.
|
The PIP guidelines are integrated into each contracted organization Program Integrity Plan Policies and Procedures; and are assumed as a standard operating procedure to prevent fraud, waste and abuse
|
|||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA02.1
|
Methods for identification, investigation, and referral
|
|||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
|||
|
Purpose
|
This guide describes what the organization must include in their PIP to guarantee the use of methods for the identification, investigation, and referral of suspected fraud and abuse cases.
|
|||
|
General
|
The organization must establish methods for the identification, investigation and referral of suspected cases, that guarantees the use of a consistent and objective approach to address fraud, waste and abuse when performing PIP activities.
|
|||
|
Guidelines
|
The PIP must include an explicit definition of methods to perform identification of cases suspected of fraud, waste and abuse
|
|||
|
a.
|
what is fraud, waste and abuse
|
|||
|
b.
|
how is detected fraud, waste and abuse
|
|||
|
c.
|
who performs the identification
|
|||
|
d.
|
when preliminary, full investigation and resolutions are done
|
|||
|
The PIP must have a detailed process to perform investigations on each suspected case guaranteeing objective methods to identify potential cases and perform investigations
|
||||
|
a.
|
open and documents the case
|
|||
|
b.
|
initiate data gathering process
|
|||
|
c.
|
follow a protocol to verify information
|
|||
|
d.
|
issue a report of findings
|
|||
|
e.
|
refer case to next level
|
|||
|
f.
|
close the case
|
|||
|
The PIP must include a variety of methods for the identification, investigation and referral of suspected cases, accepted in the industry and without infringing provider or beneficiary rights. Methods might include
|
||||
|
a.
|
electronic data exchanges
|
|||
|
b.
|
data mining
|
|||
|
c.
|
claims registries / reconciliation
|
|||
|
d.
|
targeted procedures
|
|||
|
e.
|
profiling
|
|||
|
The PIP must include a systematic approach of data analysis by:
|
||||
|
a.
|
flagging the case
|
|||
|
b.
|
identifying cause for flagging (i.e. over-under payment)
|
|||
|
c.
|
establishing actions and sanctions
|
|||
|
The PIP must have procedures in placed for referring suspect fraud cases to law enforcement officials, at a minimum:
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Guidelines
|
a.
|
an organizational structure to address the reports.
|
||
|
b.
|
a due process that includes but is not limited to: case identification, complete record with supporting materials, notification letter to suspect, notification letter to single state agency, documentation of entrance and exit interviews, and if necessary copy of referral letters and case resolution letter to and from legal authorities.
|
|||
|
c.
|
a flowchart to work in cooperation with the grantee and sub-grantee as well as with the state legal authorities such as: Organization's Legal Affairs Department, ASES, Single State Agency - Department of Health Legal Department, State Department of Justice, and the Office of Inspector General.
|
|||
|
d.
|
a follow up process to work with legal authorities each case of fraud, waste and abuse suspicion until final disposition and notification to the single state agency.
|
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA03
|
Preliminary Investigations
|
|||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
|||
|
Purpose
|
To provide guidance on how to perform a preliminary investigation when the agency receives a complaint of fraud or abuse from any source or identifies any questionable practices.
|
|||
|
General
|
The organization must conduct a preliminary investigation to determine whether there is sufficient basis to warrant a full investigation.
|
|||
|
Guidelines
|
The PIP defines a standard operating procedure to complete a preliminary investigation of all suspect cases of fraud, waste and abuse.
|
|||
|
The PIP identifies the requirements to complete the preliminary investigation when evaluating providers and beneficiaries. It should include at least:
|
||||
|
a.
|
Source of information
|
|||
|
b.
|
Identification method (how the case is detected)
|
|||
|
c.
|
Cause for investigation
|
|||
|
d.
|
Case documentation
|
|||
|
e.
|
Analysis of Data and documents
|
|||
|
f.
|
Report of Findings
|
|||
|
g.
|
Action Taken (Recommended Action)
|
|||
|
The PIP includes a mechanism to keep tracking of all preliminary investigations and results.
|
||||
|
The PIP establishes a mechanism to report preliminary investigations activity to the sub-grantee (ASES) which will be in charge of reporting activity to the single state agency (Office for the Medically Indigent).
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA04
|
Full Investigations
|
|||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
|||
|
Purpose
|
To provide guidance and minimum set of elements in the PIP to perform full investigations on incidents of fraud and abuse.
|
|||
|
General
|
If the findings of a preliminary investigation give the agency reason to believe that an incident of fraud or abuse has occur in the Medicaid program, the organization must take the appropriate actions.
|
|||
|
Guidelines
|
The PIP must define the process to conduct a full investigation and specify when a case requires the full investigation. Full investigations must be done in accordance with federal regulation and based in the company written policy. The company must submit copy of the written policies to ASES for review and approval.
|
|||
|
The PIP must define the process to refer the cases to the companies fraud liaison (i.e. companies compliance office), the appropriate law enforcement agency / sub-grantee when there is a reason:
|
||||
|
a.
|
to suspect a provider has engaged in fraud or abuse of the program.
|
|||
|
b.
|
to suspect a recipient is defrauding the program.
|
|||
|
c.
|
to suspect a recipient has abused the Medicaid program.
|
|||
|
The PIP must have a mechanism to keep tracking of all full investigations performed in progress and closed.
|
||||
|
The PIP must have a mechanism to report the sub-grantee (ASES) informed full investigations in progress, conducted and results.
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA05
|
Resolution of full investigation
|
|||||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
|||||
|
Purpose
|
To provide guidance on minimum actions that must be taken in order to complete the process of a full investigation.
|
|||||
|
General
|
The full investigations must continue until the cases are referred, solved or closed.
|
|||||
|
Guidelines
|
The PIP must include the process to guarantee that a full investigation must continue until:
|
|||||
|
a.
|
appropriate legal action is initiated.
|
|||||
|
b.
|
the case is closed or dropped because of insufficient evidence to support the allegations of fraud or abuse.
|
|||||
|
c.
|
the matter is resolved between the organization and the provider or recipient
|
|||||
|
✓
|
the resolution may include but is not limited to:
|
|||||
|
1)
|
Sending a warning letter to the provider or recipient, giving notice that continuation of the activity in question will result in further action;
|
|||||
|
2)
|
Suspending or terminating the provider from participation in the Medicaid program;
|
|||||
|
3)
|
Seeking recovery of payments made to the provider; or
|
|||||
|
4)
|
Imposing other sanctions provided under the organization PIP plan.
|
|||||
|
The PIP must have a mechanism to report the sub-grantee (ASES) informed full investigations in progress, conducted and results.
|
||||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 – 2015
|
Title SA06
|
Reporting Requirements
|
||||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
||||
|
Purpose
|
To provide guidance on how to adhere to a minimum set of elements that must be included in the process to report fraud and abuse information to the appropriate organizations officials.
|
||||
|
General
|
The organization must submit a progress report the fraud and abuse information and statistics to the appropriate department / grantee / sub- grantee on quarterly basis.
|
||||
|
Guidelines
|
The PIP must describe the mechanism to report fraud and abuse data to the appropriate fraud liaison, organization structure, sub-grantee (ASES) and grantee (Office for the Medically Indigent).
|
||||
|
The PIP progress report must include at least the following information:
|
|||||
|
a.
|
# of complaints on fraud and abuse received.
|
||||
|
b.
|
.# of complaints that warrant preliminary investigation.
|
||||
|
c.
|
Detailed information for each case of suspected provider fraud and abuse that warrants a full investigation:
|
||||
|
✓
|
Provider's name and id number
|
||||
|
✓
|
Source of the complaint
|
||||
|
ü
|
Type of the provider
|
||||
|
✓
|
Nature of the complaint
|
||||
|
ü
|
Estimate amount of money involved
|
||||
|
ü
|
Legal and administrative disposition of the case and actions taken by the law enforcement officials to whom the case has been referred.
|
||||
|
Suspected fraud cases must be reported immediately in a written format to ASES Compliance Office.
|
|||||
|
The PIP reports must be submitted in electronic format to facilitate its inclusion in the Commonwealth of Puerto Rico Medicaid Program PI Annual Report.
|
|||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA07
|
Provider's statements on claims forms
|
|||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
|||
|
Purpose
|
To provide guidance on how to comply with regulation on provider's statements on claims forms.
|
|||
|
General
|
The organization may print that all provider claims forms be imprinted in boldface type with the following statement, or with alternate wording that is approved by the Regional CMS Administration.
|
|||
|
Guidelines
|
The PIP must include that providers are required to attest in the claim forms that they agree with the following statement:
|
|||
|
✓
|
"This is to certify that the foregoing information is true accurate and complete".
|
|||
|
✓
|
"I understand that payment of this claim will be from federal and state funds and that any falsification or concealment of a material fact maybe prosecutes under federal and state laws".
|
|||
|
For electronic claims, providers must attest that they agree with the following statements:
|
||||
|
✓
|
"This is to certify the truthfulness of the foregoing information and certify that is true, accurate, complete and that the service was provided".
|
|||
|
The statements may be printed above the claimant's signature or, if they are printed on the revenue of the form, a reference to the statements must appear immediately preceding the claimant's signature.
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA08
|
Provider's statements on check
|
|||
|
Scope
|
Grantee, Sub-grantee and Contracted Organizations
|
|||
|
Purpose
|
To provide guidance on how to comply with regulation on provider's statements on check.
|
|||
|
General
|
The organization may print the following wording above the claimant's endorsement on the reverse of checks or warrants payable to each provider.
|
|||
|
Guidelines
|
The PIP must include that providers are required to attest (in addition to the statements required in providers claims form) that they agree with the following statement either by having it written on checks or temporarily in a legal document as an affidavit:
|
|||
|
✓
|
"I understand in endorsing or depositing this check that payment will be from Federal and State funds and that any falsification, or concealment of a material fact, may be prosecuted under Federal and State laws".
|
|||
|
The above attestation must be included in electronic and checks payment.
|
||||
|
The PIP must indicate frequency and responsible for conducting spot checks to guarantee the organization complies with the provider's statements and / or the provider signature appears on a legal document attesting compliance.
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA09
|
Recipient verification procedure
|
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|
Purpose
|
To verify that the services listed on claims forms have been rendered.
|
|
General
|
The organization must have a method for verifying with recipients whether services billed by providers were received.
|
|
Guidelines
|
The PIP must include a description of how the organization performs claims matches with medical records to guarantee adequacy of billing.
The PIP must define the mechanism to monitor frequency of encounters and services rendered to patients billed by providers.
The PIP will provide periodic updates on reconciliation findings report to the sub-grantee and grantee.
The sub-grantee will select a sample to perform independent reviews to verify that recipient's services billed by providers (as well as encounters under capitated environment) were indeed rendered. This review will be performed through confirmations to beneficiaries.
|
Note: All contracted companies are required to comply with Law 114 which require that the beneficiaries must receive an Evidence of Medical Benefits with a detailed of the services and expenses incurred during a quarter. ASES compliance office will review the compliance with the Law.
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA10
|
Cooperation with Medicaid Fraud Control Units
|
|||
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|||
|
Purpose
|
To provide guidance on how to communicate findings and to cooperate with any Puerto Rico or federal law enforcement agency. To request that all contracted companies must communicate preliminary findings to ASES.
|
|||
|
General
|
The organization must have a mechanism to provide information to the regulatory and legal authorities on cases, investigations, schemes and any other activity where intention to commit fraud, abuse and waste of services occur.
|
|||
|
Guidelines
|
The PIP must demonstrate it has an effective mechanism to cooperate with the Medicaid anti-fraud unit as well as with other program divisions in charge of preventing and prosecuting cases related to fraud, waste and abuse of services under the Medicaid program.
|
|||
|
The PIP must establish a process to guarantee the organization complies with the following:
|
||||
|
✓
|
All cases of suspected provider fraud are referred to the anti-fraud / integrity organization's unit.
|
|||
|
✓
|
If the anti-fraud / integrity unit determines that it may be useful in carrying out the unit's responsibilities, promptly comply with a request from the unit for -
|
|||
|
i.
|
Access to, and free copies of, any records or information kept by the organization or its contractors;
|
|||
|
ii.
|
Computerized data stored by the organization or its contractors. These data must be supplied without charge and in the form requested by the unit;
|
|||
|
iii.
|
Access to any information kept by providers to which the organization is authorized access. In using this information, the unit must protect the privacy rights of recipients;
|
|||
|
✓
|
Communicate to ASES preliminary findings; and
|
|||
|
✓
|
On referral from the unit, coordinate with ASES or appropriate law enforcement agency before initiating any available administrative or judicial action to recover improper payments to a provider.
|
|||
|
The PIP must recommend the organization to have in the provider's contract a disclaimer that as a contracted provider any data related to services or payments provided must be available for review of the integrity staff.
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA11
|
Suspension of payments in cases of fraud
|
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|
Purpose
|
To provide guidance on elements to be considered when suspending payments
to providers who committed fraud.
|
|
General
|
The organization must suspend payments to providers as a mechanism to prevent wrong disbursement of payments when there is a credible allegation of fraud for which an investigation is pending unless the agency have a good cause to not suspend payments or to suspend payment only in part.
|
|
Guidelines
|
Th ePIP will establish a mechanism and adhere to the following recommendations when considering suspension of payments:
|
|
(a)
|
Basis for suspension. The Organization must suspend all Medicaid payments to a provider after the agency determines there is a credible allegation of fraud for which an investigation is pending under the Medicaid program against an individual or entity unless the agency has good cause to not suspend payments or to suspend payment only in part. The Organization may suspend payments without first notifying the provider of its intention to suspend such payments. A provider may request, and must be granted, administrative review where State law so requires.
|
|
(b)
|
Notice of suspension . The Organization must send notice of its suspension of program payments within:
|
|
•
|
5 days of taking such action unless requested in writing by a law enforcement agency to temporarily withhold such notice.
|
|
•
|
30 days if requested by law enforcement in writing to delay sending such notice, which request for delay may be renew in writing up to twice and in no event may exceed 90 days.
|
|
•
|
The notice must include or address all of the following:
|
- State that payments are being suspended in accordance with this provision (CFR 42 CFR 455.23);
- Set forth the general allegations as to the nature of the suspension action, but need not disclose any specific information concerning an ongoing investigation.
- Specify, when applicable, to which type or types of Medicaid claims (capitation or claims) or business units of a provider suspension is effective.
- Inform the provider of the right to submit written evidence for consideration by the agency.
- Set forth the applicable administrative appeals process and corresponding citations to State law.
|
(c)
|
Duration of suspension
|
|
1)
|
All suspension of payment actions under this section will be temporary and will not continue after either of the following:
|
- The agency or the prosecuting authorities determine that there is insufficient evidence of fraud by the provider.
- Legal proceedings related to the provider's alleged fraud are completed.
|
2)
|
It must be documented in writing the termination of a suspension including, where applicable and appropriate, any appeal rights available to a provider.
|
|
(d)
|
Referrals to the ASES, Medicaid and OIG.
|
|
(1)
|
Whenever the Organization investigation leads to the initiation of a payment suspension in whole or part, the Organization must make a fraud referral to ASES who will notify the OIG and the Medicaid Office.
|
|
(2)
|
The fraud referral must meet all of the following requirements:
|
|
•
|
Be made in writing and provided to ASES not later than the next business day after the suspension is enacted.
|
|
•
|
Conform to fraud referral performance standards issued by the Secretary .
|
|
(3)(i)
|
If the Medicaid fraud control unit or other law enforcement agency accepts the fraud referral for investigation, the payment suspension may be continued until such time as the investigation and any associated enforcement proceedings are completed.
|
(ii) On a quarterly basis, the Organization must request a certification from the Medicaid fraud control unit or other law enforcement agency that any matter accepted on the basis of a referral continues to be under investigation thus warranting continuation of the suspension.
|
(4)
|
If the Medicaid fraud control unit or other law enforcement agency declines to accept the fraud referral for investigation the payment suspension must be discontinued unless the State Medicaid agency or ASES has alternative Federal or State authority by which it may impose a suspension or makes a fraud referral to another law enforcement agency. In that situation, the provisions of paragraph (d)(3) of this section apply equally to that referral as well.
|
|
(5)
|
A decision to exercise the good cause exceptions in paragraphs
|
|
(e)
|
or (f) of this section not to suspend payments or to suspend payments only in part does not relieve the Organization of the obligation to refer any credible allegation of fraud as provided in paragraph (d)(l) of this section.
|
|
(e)
|
Good cause not to suspend payments. The Organization may find that good cause exists not to suspend payments, or not to continue a payment suspension previously imposed, to an individual entity against which there is an investigation of a credible allegation of fraud if any of the following are applicable:
|
|
(1)
|
Law enforcement officials have specifically requested that a payment suspension not be imposed because such a payment suspension may compromise or jeopardize an investigation.
|
|
(2)
|
Other available remedies implemented by the Organization more effectively or quickly protect Medicaid funds.
|
|
(3)
|
The Organization determines, based upon the submission of written evidence by the individual or entity that is the subject of the payment suspension, that the suspension should be removed.
|
|
(4)
|
Beneficiary access to items or services would be jeopardized by a payment suspension because of either of the following:
|
|
(i)
|
An individual or entity is the sole community physician or the sole source of essential specialized services in a community.
|
|
(ii)
|
The individual or entity serves a large number of recipients within a HRSA-designated medically underserved area.
|
|
(5)
|
Law enforcement declines to certify that a matter continues to be under investigation per the requirements of paragraph (d)(3) of this section.
|
|
(6)
|
The State determines that payment suspension is not in the best interests of the Medicaid program.
|
|
(f)
|
Good cause to suspend payment only in part. The Organization may find that good cause exists to suspend payments in part, or to convert a payment suspension previously imposed in whole to one only in part, to an individual or entity against which there is an investigation of a credible allegation of fraud if any of the following are applicable :
|
|
(1)
|
Recipient access to items or services would be jeopardized by a payment suspension in whole or part because of either of the following:
|
|
(i)
|
An individual or entity is the sole community physician or the sole source of essential specialized services in a community.
|
|
(ii)
|
The individual or entity serves a large number of recipients within a HRSA-designated medically underserved area.
|
|
(2)
|
The Organization determines, based upon the submission of written evidence by the individual or entity, that is the subject of a whole payment suspension, that such suspension should be imposed only in part.
|
|
(3)(i)
|
The credible allegation focuses solely and definitively on only a specific type of claim or arises from only a specific business unit of a provider; and
|
(ii) The Organization determines and documents in writing that a payment suspension in part would effectively ensure that potentially fraudulent claims were not continuing to be paid.
|
(4)
|
Law enforcement declines to certify that a matter continues to be under investigation per the requirements of paragraph (d)(3) of this section.
|
|
(5)
|
The State determines that payment suspension only in part is in the best interests of the Medicaid program.
|
|
(g)
|
Documentation and record retention. The Organization must meet the following requirements:
|
|
(1)
|
Maintain for a minimum of 5 years from the date of issuance all materials documenting the life cycle of a payment suspension that was imposed in whole or part, includingthe following:
|
|
(i)
|
All notices of suspension of payment in whole or part.
|
|
(ii)
|
All fraud referrals to the Medicaid fraud control unit or other law enforcement agency.
|
|
(iii)
|
All quarterly certifications of continuing investigation status by law enforcement.
|
|
(iv)
|
All notices documenting the termination of a suspension. (2)(i) Maintain for a minimum of 5 years from the date of issuance all materials documenting each instance where a payment suspension was not imposed, imposed only in part, or discontinued for good cause.
|
|
(ii)
|
This type of documentation must include, at a minimum, detailed information on the basis for the existence of the good cause not to suspend payments, to suspend payments only in part,or to discontinue a payment suspension and, where applicable, must specify how long the Organization anticipates such good cause will exist.
|
|
(3)
|
Annually report to the Secretary and ASES summary information on each of following:
|
|
(i)
|
Suspension of payment, including the nature of the suspected fraud, the basis for suspension, and the outcome of the suspension.
|
|
(ii)
|
Situation in which the Organization determined good cause existed to not suspend payments, to suspend payments only in part, or to discontinue a payment suspension as described in this section, including describing the nature of the suspected fraud and the nature of the good cause.
|
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA12
|
Disclosure of Information and Screen for Identity and Exclusions by Providers and Fiscal Agents
|
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|
Purpose
|
To provide definition of concepts in order to fully adhere to the regulation on providers control and ownership of facilities and verification of employees for
exclusions
|
|
General
|
The organization must adhere to standard definitions when dealing with disclosure of information by providers and fiscal agents when establishing
mechanism to regulate providers control and ownership of facilities and verification of employees for identity and exclusions
|
|
Guidelines
|
The PIP will adhere to the following definitions of concepts to keep consistency with federal regulation and application of law:
|
|
The PIP will adhere to the following definitions of concepts to keep consistency with federal regulation and application of law:
Agent means any person who has been delegated the authority to obligate or act on behalf of a provider.
Disclosing entity means a Medicaid provider (other than an individual practitioner or group of practitioners), or a fiscal agent.
Other disclosing entity means any other Medicaid disclosing entity and any entity that does not participate in Medicaid, but is required to disclose certain ownership and control information because of participation in any of the federal programs (Medicaid, SCHIP, FQHC's). This includes:
(a) Any hospital, skilled nursing facility, home health agency, independent clinical laboratory, renal disease facility, rural health clinic, or health maintenance organization that participates in Medicare (title XVIII);
(b) Any Medicare intermediary or carrier; and
(c) Any entity (other than an individual practitioner or group of practitioners) that furnishes, or arranges for the furnishing of, health related services for which it claims payment under any plan or program established under title V or title XX of the Act.
Fiscal agent means a contractor that processes or pays vendor claims on behalf of the Medicaid agency .
Group of practitioners means two or more health care practitioners who practice their profession at a common location (whether or not they share
common facilities, common supporting stff, or common equipment).
Indirect ownership interest means an ownership interest in an entity that has an ownership interest in the disclosing entity. This term includes an ownership interest in any entity that has an indirect ownership interest in the disclosing entity.
Managing employee means a general manager, business manager, administrator, director, or other individual who exercises operational or managerial control over, or who directly or indirectly conducts the day-to-day operation of an institution, organization, or agency.
Ownership interest means the possession of equity in the capital, the stock, or the profits of the disclosing entity.
Person with an ownership or control interest means a person or corporation that -
|
|
|
(a) Has an ownership interest totaling 5 percent or more in a disclosing entity;
(b) Has an indirect ownership interest equal to 5 percent or more in a disclosing entity;
(c) Has a combination of direct and indirect ownership interests equal to 5 percent or more in a disclosing entity;
(d) Owns an interest of 5 percent or more in any mortgage, deed of trust, note, or other obligation secured bye the disclosing entity if that interest equals at least 5 percent of the value of the property or assets of the disclosing entity;
(e) Is an officer or director of a disclosing entity that is organized as a corporation; or
(f) Is a partner in a disclosing entity that is organized as a partnership.
Significant business transaction means any business transaction or series of transactions that, during any one fiscal year, exceed the lesser of $25,000 and 5 percent of a provider's total operating expenses.
Subcontractor means -
(a) An individual, agency or organization to which a disclosing entity has contracted or delegated some of its management functions or responsibilities of providing medical care to its patients; or
(b) An individual, agency, or organization with which a fiscal agent has entered into a contract, agreement, purchase order, or lease (or leases of real property) to obtain space, supplies, equipment, or services provided under the Medicaid agreement.
Supplier means an individual, agency or organization from which a provider purchases goods and services used in carrying out its responsibilities under Medicaid (e.g., a commercial laundry, a manufacturer of hospital beds, or a pharmaceutical firm).
Wholly owned supplier means a supplier whose total ownership interest is held by a provider or by a person, persons, or other entity with an ownership or control interest in a provider.
The PIP must also ensure providers compliance with screening employees for identity and exclusions. To further protect against payments for items and services furnished or ordered by excluded parties, all current providers and providers applying to participate in the plan must be advised to take the following steps to confirm identities and to determine whether their employees and contractors are excluded individuals or entities:
- Providers have the obligation to screen all employees and contractors to confirm the identity and determine the exclusion status through routine checks of Federal databases. The Organization should communicate this obligation to providers upon enrollment and reenrollment.
- Providers should explicitly be required to agree to comply with this obligation as a condition of enrollment.
- Providers should be informed that they can search the Social Security Administration's Death Master File, the National Plan and Provider Enumeration System (NPPES), the List of Excluded Individuals/Entities (LEIE), the Excluded Parties List System (EPLS), and any such other databases as the Secretary may prescribe.
- Providers should be required to search the LEIE and EPLS no less frequently than monthly to capture exclusions and reinstatements that have occurred since the last search.
- Providers should be required to immediately report to them any exclusion information discovered.
This line of defense in combating fraud and abuse must be conducted accurately, thoroughly, and routinely. The Organization must notify ASES and the HHS-OIG promptly of any administrative action taken against a provider for failure to comply with these screening and reporting obligations. See 42 CFR section 1002.3(b)(3). The Organization can satisfy this obligation by communicating the relevant information to ASES and the appropriate Regional Office of the OIG Office of Investigations.
The Organizations also should inform providers that civil monetary penalties may be imposed against Medicaid providers and entities contracted by ASES who employ or enter into contracts with excluded individuals or entities to provide items or services to Medicaid recipients. (Section 1128A(a)(6) of the Act; and 42 CFR section 1003.102(a)(2)).
|
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA13
|
Disclosure by disclosing entities: Information on ownership and control.
|
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|
Purpose
|
To provide guidelines on what information must be disclosed by entities that have ownership and control over facilities.
|
|
General
|
The organization must have a mechanism to monitor on a timely manner the providers and fiscal agents that owns or control facilities where Medicaid beneficiaries receive services.
|
|
Guidelines
|
The Organization must require each disclosing entity to disclose the following information in a timely manner:
(a) Type of Information that must be disclosed.
(1) (i) The name and address of any person (individual or corporation) with an ownership or control interest in the disclosing entity, fiscal agent, or the entity contracted by ASES. The address for corporate entities must include as applicable primary business address, every business location, and P.O. Box address.
(ii) Date of birth and Social Security Number (in the case of an individual).
(iii) Other tax identification number (in the case of a corporation) with an ownership or control interest in the disclosing entity (or fiscal agent or the entity contracted by ASES) or in any subcontractor in which the disclosing entity (or fiscal agent or the entity contracted by ASES) has a 5 percent or more interest.
(2) Whether the person (individual or corporation) with an ownership or control interest in the disclosing entity (or fiscal agent or the entity contracted by ASES) is related to another person with ownership or control interest in the disclosing entity as a spouse, parent, child, or sibling; or whether the person (individual or corporation) with an ownership or control interest in any subcontractor in which the disclosing entity (or fiscal agent or the entity contracted by ASES} has a 5 percent or more interest is related to another person with ownership or control interest in the disclosing entity as a spouse, parent, child, or sibling.
(3) The name of any other disclosing entity (or fiscal agent or the entity contracted by ASES} in which an owner of the disclosing entity (or fiscal agent or the entity contracted by ASES) has an ownership or control interest.
(4) The name, address, date of birth, and Social Security Number of any managing employee of the disclosing entity (or fiscal agent or the entity contracted by ASES).
(b) When the disclosures must be provided.
(l) Disclosures from providers or disclosing entities. Disclosure from any provider or disclosing entity is due at any of the following times:
(i) Upon the provider or disclosing entity submitting the provider application.
(ii) Upon the provider or disclosing entity executing the provider agreement.
(iii) Upon request of the organization during the re-validation of enrollment process under § 455.414.
(iv) Within 35 days after any change in ownership of the disclosing entity.
(2) Disclosures from fiscal agents or managed care entities - .Disclosures from fiscal agents are due at any of the following times:
(i) Upon the fiscal agent submitting the proposal in accordance with the State's procurement process.
(ii) Upon the fiscal agent executing the contract with the State. (iii) Upon renewal or extension of the contract.
(iv) Within 35 days after any change in ownership of the fiscal agent.
Updated information must be furnished to the Secretary or the State survey or the Organization at intervals between recertification or contract renewals, within 35 days of a written request.
(c) Consequences for failure to provide required disclosures.
- Federal financial participation (FFP) is not available in payments made to a disclosing entity that fails to disclose ownership or control information as required by this section.
- The Organization shall not approve a provider agreement or a contract with a fiscal agent, and must terminate an existing agreement or contract, if the provider or fiscal agent fails to disclose ownership or control information as required by this section.
The PIP will include the process to provide an annual report to the grantee and sub-grantee on above information and data.
|
Commonwealth of Puerto Rico
Program Integrity Plan 2013 - 2014
|
Title SA14
|
Disclosure by providers: Information related to business transactions.
|
|||
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|||
|
Purpose
|
The organization must establish a mechanism to facilitate the providers disclose information related to their business transactions when own or control facilities where Medicaid beneficiaries received services.
|
|||
|
Guidelines
|
The PIP must describe the mechanism to allow providers owning or controlling facilities disclose information related to business transactions.
|
|||
|
The PIP must attest the organization abide by the following regulation:
|
||||
|
(a)
|
Provider agreements. The organization must enter into an agreement with each provider or provider group under which the provider agrees to furnish to it or to the grantee / sub-grantee on request, information related to business transactions.
|
|||
|
(b)
|
Information that must be submitted. A provider must submit, within 35 days of the date on a request by the organization full and complete information about-
|
|||
|
✓
|
The ownership of any subcontractor with whom the provider has had business transactions totaling more than $25,000 during the 12-month period ending on the date of the request; and
|
|||
|
✓
|
Any significant business transactions between the provider and any wholly owned supplier, or between the provider and any subcontractor, during the 5-year period ending on the date of the request.
|
|||
|
The PIP must include withholding of payment processes and procedures to enforce above guideline.
|
||||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 - 2015
|
Title SA15
|
Disclosure by providers: Information on persons convicted of crimes
|
|||
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|||
|
Purpose
|
To provide guidance on type of information providers must report in compliance with integrity program.
|
|||
|
General
|
The organization is obliged to request providers to report any conviction of crimes or any other in the program integrity regulation.
|
|||
|
Guidelines
|
The PIP must include a mechanism to confirm information included below is considered as part of the integrity activities.
|
|||
|
(a)
|
Information that must be disclosed. Before the organization enters into or renews a provider agreement, or at any time upon written request by the organization, the provider must disclose to the organization the identity of any person who:
|
|||
|
(1)
|
Has ownership or control interest in the provider, or is an agent or managing employee of the provider; and
|
|||
|
(2)
|
Has been convicted of a criminal offense related to that person's involvement in any program under Medicare, Medicaid, or the title XX services program since the inception of those programs.
|
|||
|
(b)
|
Notification to Inspector General.
|
|||
|
(1)
|
The organization must notify the Inspector General of the Department of any disclosures made under paragraph (a) of this section within 20 working days from the date it receives the information.
|
|||
|
(2)
|
The organization must also promptly notify the Inspector General of the Department of any action it takes on the provider's application for participation in the program.
|
|||
|
(c)
|
Denial or termination of provider participation.
|
|||
|
(1)
|
The organization may refuse to enter into or renew an agreement with a provider if any person who has an ownership or control interest in the provider, or who is an agent or managing employee of the provider, has been convicted of a criminal offense related to that person's involvement in any program established under Medicare, Medicaid or the title XX Services Program.
|
|||
|
(2)
|
The organization may refuse to enter into or may terminate a provider agreement if it determines that the provider did not fully and accurately make any disclosure required under paragraph (a) of this section.
|
|||
Commonwealth of Puerto Rico
Program Integrity Plan 2014 – 2015
|
Title SA16
|
Provider Screening and Enrollment
|
|
Scope
|
Grantee, Sub-Grantee and Contracted Organizations
|
|
Purpose
|
To provide guidance on termination or denial of enrollment and criminal background checks.
|
|
General
|
The organization is obliged to establish procedures for termination or denial of enrollment and to obtain providers consent to criminal background checks.
|
|
Guidelines
|
The PIP must include a process to confirm that the requirements included below are considered as part of the integrity activities.
1. The Organization must:
(a) terminate the enrollment of any provider where any person with a 5 percent or greater direct or indirect ownership interest in the provider did not submit timely and accurate information and cooperate with any screening methods required under this subpart.
(b) deny enrollment or terminate the enrollment of any provider where any person with a 5 percent or greater direct or indirect ownership interest in the provider has been convicted of a criminal offense related to that person's involvement with the Medicare, Medicaid,or title XXI program in the last 10 years, unless ASES determines that denial or termination of enrollment is not in the best interests of the Medicaid program and the State Medicaid agency documents that determination in writing.
(c) deny enrollment or terminate the enrollment of any provider that is terminated on or after January 1, 2011, under title XVIII of the Act or under the Medicaid program or CHIP of any other State.
(d) terminate the provider's enrollment or deny enrollment of the provider if the provider or a person with an ownership or control interest or who is an agent or managing employee of the provider fails to submit timely or accurate information, unless ASES determines that termination or denial of enrollment is not in the best interests of the Medicaid program and the State Medicaid agency documents that determination in writing.
(e) terminate or deny enrollment if the provider, or any person with a percent or greater direct or indirect ownership interest in the provider, fails to submit sets of fingerprints in a form and manner to be determined by ASES within 30 days of a CMS or a ASES request, unless ASES determines that termination or denial of enrollment is not in the best interests of the Medicaid program and ASES documents that determination in writing.
(f) terminate or deny enrollment if the provider fails to permit access to provider locations for any site visits under § 455.432, unless ASES determines that termination or denial of enrollment is not in the best interests of the Medicaid program and the State Medicaid agency documents that determination in writing.
(g) May terminate or deny the provider's enrollment if CMS, ASES or the State Medicaid agency-
(1) Determines that the provider has falsified any information provided on the application; or
(2) Cannot verify the identity of any provider applicant. 1. Reactivation of provider enrollment
After deactivation of a provider enrollment number for any reason, before the provider's enrollment may be reactivated, the Organization must re-screen the provider.
2. Appeal rights
The State Medicaid agency must give providers terminated or denied under §455.416 any appeal rights available under procedures established by State law or regulations.
3. Criminal background checks
As a condition of enrollment, the organization must require providers to consent to criminal background checks including finger printing when required by law enforcement agencies or State law.
|
Attachment 15
Formulary A-102 - Evidence of Lack of Providers and Providers Refusal to Contract
Pursuant to section 9.4.3 of the Contract, the Contractor must use this Formulary to evidence the lack of providers in its Service Region or refusal to contract as part of the General Network or the PPN of its Service Region. The Contractor must carry out all efforts to contract with those specialists within contiguous Service Regions; provided that before recurring to contiguous regions Contractor must validate and submit all supporting documents evidencing the lack of Providers or refusal to contract required in this Formulary.
|
Provider Name
|
|
|
|
Specialty
|
|
|
|
Service Region
|
|
|
List of MCO's Recruitment Activities and dates of such activities:
|
|
|
|
|
|
|
|
|
|
Provide the Dates and Outcomes of Meetings with provider:
|
|
|
|
|
|
|
|
|
|
Explain the issues causing concerns or barriers to Contracting
|
|
|
|
|
|
|
|
|
|
Provide description of contract offers to the provider including fees and any other incentives:
|
|
|
|
|
|
|
|
|
|
Provide reasons why the provider refused the contract:
|
|
|
|
|
|
|
|
|
|
Describe provider counter offers:
|
|
|
|
|
|
|
|
|
|
Describe remedies offered by MCO to address provider's concerns in order to come to terms with the contract
|
|
|
|
|
|
|
|
|
|
Please attach to this Formulary any relevant document in support of your responses.
|
|
Attachment 16
List of Required Reports
|
Program Area
|
ASES Dept.
|
Report
Number
|
Report
Reference
|
Frequency
|
Report Title
|
|
Administrative
|
Customer Service
|
1
|
6.8
|
Monthly
|
Call Center Report
|
|
Administrative
|
Systems and Compliance
|
2
|
17.7
|
Monthly
|
Privacy and Confidentiality Report
|
|
Administrative
|
Compliance
|
3
|
13.5
|
Quarterly
|
Fraud Waste Abuse Report
|
|
Administrative
|
Customer Service
|
4
|
6.2
|
Quarterly
|
Enrollee Enrollment Materials Report
|
|
Administrative
|
Compliance
|
5
|
13.1
|
Quarterly
|
Employee and Contractor Suspensions/Debarment Report
|
|
Administrative
|
Compliance
|
6
|
13.2
|
Annually
|
Compliance Plan
|
|
Administrative
|
Compliance
|
7
|
13.3
|
Annually
|
Program Integrity Plan
|
|
Administrative
|
Systems and Compliance
|
8
|
17.7
|
Annually or 10 Business Days following incident
|
Systems Incident Report
|
|
Administrative
|
Executive
|
9
|
12.2.7
|
10 days following each meeting
|
Activities of the Advisory Board
|
|
Claims
|
Finance
|
10
|
16.7
|
Monthly
|
Claims Activity Report
|
|
Claims
|
Finance
|
11
|
7.9
|
Monthly
|
Encounter Data
|
|
Covered Services
|
Quality
|
12
|
7.8.2
|
Quarterly
|
Care Management Report
|
|
Covered Services
|
Quality
|
13
|
7.8.3
|
Quarterly
|
Disease Management Report
|
|
Program Area
|
ASES Dept.
|
Report
Number
|
Report
Reference
|
Frequency
|
Report Title
|
|
Covered Services
|
Quality
|
14
|
7.5
|
Annually
|
Wellness Plan
|
|
Covered Services
|
Quality
|
15
|
7.5.8
|
Annually
|
Maternal and Pre-Natal Plan
|
|
Covered Services
|
Quality
|
16
|
7.9
|
Annually
|
EPSDT Plan
|
|
Covered Services
|
Quality
|
17
|
7.9
|
Annually
|
CMS-416 Report
|
|
Financial Management
|
Finance
|
18
|
22.1
|
Monthly
|
Per Member Per Month Disbursement Report
|
|
Financial Management
|
Systems
|
19
|
22.1
|
Monthly
|
Actuarial Data
|
|
Financial Management
|
Finance
|
20
|
23.4
|
Monthly
|
Enrollee TPL Health Insurance Report
|
|
Financial Management
|
Finance
|
21
|
23.4
|
Quarterly
|
Retention Fund Report
|
|
Financial Management
|
Finance
|
22
|
23.1
|
Quarterly
|
Unaudited Financial Statement
|
|
Financial Management
|
Finance
|
23
|
23.4
|
Quarterly
|
Cost Avoidance Report
|
|
Financial Management
|
Compliance
|
24
|
23.7.4
|
Annually
|
Disclosure of Information on Annual Business Transactions
|
|
Financial Management
|
Finance
|
25
|
23.1
|
Annually
|
Report to Puerto Rico Insurance Commissioner's Office
|
|
Financial Management
|
Finance
|
26
|
23.1
|
Annually
|
Annual Corporate Report
|
|
Financial Management
|
Quality
|
27
|
23.6
|
Annually
|
Physician Incentive Plan Report
|
|
Program Area
|
ASES Dept.
|
Report
Number
|
Report
Reference
|
Frequency
|
Report Title
|
|
Financial Management
|
Finance
|
28
|
23.1
|
Annually
|
Audited Financial Statements
|
|
Financial Management
|
Finance
|
29
|
23.7.3
|
Annually
|
Report on Controls Placed in Operation and Tests of Operating Effectiveness
|
|
Provider
|
Compliance
|
30
|
9.2.3
|
Monthly
|
Provider Credentialing and Re- Credentialing Report
|
|
Provider
|
Quality
|
31
|
9.1
|
Monthly
|
National Provider List
|
|
Provider
|
Compliance
|
32
|
9.1
|
Quarterly
|
Provider Suspensions and Terminations Report
|
|
Provider
|
Quality
|
33
|
9.1
|
Quarterly
|
Geographic Access Report
|
|
Provider
|
Customer Service
|
34
|
12.7
|
Annually
|
Provider Satisfaction Survey Report
|
|
Provider
|
Quality
|
35
|
9.1
|
Annually
|
Provider Training and Outreach Plan/Evaluation Report
|
|
Quality
|
Customer Service
|
36
|
14.8
|
Quarterly
|
Grievances and Appeals Report
|
|
Quality
|
Quality
|
37
|
12.6
|
Quarterly
|
Quality Improvement Performance Report
|
|
Quality
|
Quality
|
38
|
12.6
|
Annually
|
Audited HEDIS Results Report
|
|
Quality
|
Customer Service
|
39
|
12.7
|
Annually
|
Enrollee Satisfaction Survey Report
|
|
Quality
|
Customer Service
|
40
|
12.3-12.5
|
Annually
|
QAPI Program Description
|
|
Quality
|
Quality
|
41
|
17.5
|
Monthly
|
Systems Availability and Performance Report
|
|
Program Area
|
ASES Dept.
|
Report Number
|
Report Reference
|
Frequency
|
Report Title
|
|
Quality
|
Systems
|
42
|
17.5
|
Quarterly
|
Business Continuity and Disaster Recovery (BC-DR) Test Report
|
|
Systems
|
Systems
|
43
|
17.5
|
Annually
|
Business Continuity and Disaster Recovery (BC-DR) Plan
|
|
Utilization Management
|
Quality
|
44
|
10.7
|
Quarterly
|
Admissions and Readmissions
|
|
Utilization Management
|
Clinical Affairs
|
45
|
8.7
|
Quarterly
|
Integration Report
|
|
Utilization Management
|
Clinical Affairs
|
46
|
7.5
|
Quarterly
|
Prior Authorization Report
|
|
Utilization Management
|
Clinical Affairs
|
47
|
11.1.3
|
Quarterly
|
Utilization Management Report
|
|
Utilization Management
|
Clinical Affairs
|
48
|
11.1.2
|
Annually
|
UM Program Description/Work Plan
|

PLAN FOR THE ADOPTION OF ELECTRONIC HEALTH RECORDS
BY THE GOVERNMENT HEALTH PLAN PROVIDER NETWORK
According to the public policy established by the Health Information Technology for Economic and Clinical Health (HITECH) Act, enacted as part of the American Recovery and Reinvestment Act of 2009, that promotes the adoption and meaningful use of health information technology and the Act 40 of 2012, enacted by the Commonwealth of Puerto Rico, the Administración de Seguros de Salud de Puerto Rico [ASES] as the agency responsible for the implementing the government health plan (GHP) established a Plan for the adoption of electronic health records (EHRs) by the GHP health care provider network.
ASES recognizes that physicians are the gateway to organized and integrated healthcare delivery systems. The implementation of this Strategic Plan will accomplish the integration of all the health care providers’ network, as an organized health care system, allowing ASES to plan for, provide/purchase, and coordinate all core services along the continuum of health care services for the population served by the GHP. The progressive adoption of electronic health records and the necessary secure and effective exchange of the patient health information constitute the backbone of an organized integrated health system.
The proper implementation of the Plan in a structured and progressive way will allow the achievement of the following objectives:
| · | Focus on meeting the GHP population health needs; |
| · | Efficient information systems that enhance communication and information flow across the continuum of care; |
| · | Coordinate and integrate health care across the continuum; |
| · | Able to obtain and manage information on quality outcomes and costs; |
| · | Patient access to care continuum with multiple points of access; ensuring the patient receives the “right care at the right place at the right time”; |
| · | Population-based needs assessment; focused on defined population as needed; |
| · | Maximize patient accessibility and minimize duplication of services ; |
| · | Encourage and facilitate prudent use of resources and eliminate wasteful practices; |
| · | Align service funding to ensure equitable funding distribution for different services or levels of services; |
| · | Provider-developed, evidence-based care guidelines and protocols to enforce one standard of care regardless of where patients are treated; |
| · | Cooperation between health care providers and organizations - medicine management partnerships; and |
| · | Facilitate prevention and health promotion. |
ASES, according to the authority conferred by the law, has required the MCO to promote and request the adoption and implementation of the EHR by their health care provider network and an active participation in the PRHIN (State HIE) to enable the health information exchange between the health care providers.
The adoption of electronic health records and the meaningful use by the GHP health care provider network will allow ASES to establish mechanisms that guarantee, directly and indirectly, the accessibility, quality improvement, and cost and utilization controls of health care services provided and funded by federal and state governments, as well as the protection of patients’ rights.
Strategies to Achieve ASES Goals and Objectives
ASES understands that achieving its goals and objectives will require it to work together with the contractors, to ensure that all health care providers move forward in a concerted and consistent manner in support and compliance with this Plan. The following are critical to achieving ASES’ goals and objectives.
| 1. | Promote and require the GHP health care provider networks to adopt the meaningful use of a certified health records and an active exchange of patient health information through the State health information organization, Puerto Rico Health Information Network (PRHIN). |
ASES will request the MCOs to perform a region-specific survey within their GHP health care provider networks to obtain the following information:
- Number of health care service providers/ organizations using a certified electronic health record;
- Number of health care service providers/ organizations that are active participants of a Health Information Exchange;
- Number of health care service providers/ organizations in the process of adopting and implementing a certified electronic health record system;
- Number of health care service providers/ organizations that do not have a certified electronic health record system and the reasons for that (ex. technical issues, financial issues, lack of knowledge, etc.);
Using the results of the survey, ASES and the contractors will develop and present a series of educational initiatives to advance and support, the adoption and implementation of meaningful use of the certified electronic health record by the provider networks.
Other related educational initiatives/programs will be developed and offered to assure the adequate use of the electronic health records to include the following;
- the health information exchange between providers and between providers and the contractors for the benefit of the patient care;
- the privacy and security (Privacy Framework) of the electronic management of patient health information in compliance with the federal and state regulations; and
- the patients insured by GHP are informed about the benefits of the electronic health record and the health information exchange between their health care providers.
| 2. | Ensure the Health Care Provider Networks Comply with Meaningful Use Care Goals |
-In order to comply with the Federal Government’s guidelines of what constitutes a “Meaningful Use” performance, ASES envisions that their provider networks will achieve meaningful use within the CMS program requirements. ASES and the contractors will work together to monitor the provider’s engagement in a Health Information Organization and participate in the health information exchange platform.
| 3. | Monitoring EHR Adoption and PRHIN (STATE HIE) Engagement |
The MCO will develop a milestone and auditing program to be shared with the provider networks to measure EHR adoption and implementation. By measuring the progress, the MCO will be able to identify areas where EHR adoption and/ or PRHIN (STATE HIE) engagement are successful and where more effort is needed to help certain providers so that ongoing progress towards meeting the CMS deadlines is maintained. As a result, the MCO must report ASES the milestones achieved and the findings results from the audits performed.
ASES, as the agency responsible for the implementing the GHP, will start the monitoring program using CMS requirements, as included in the contract ASES will work on a systematic measurement program that will produce reporting to demonstrate and/or validate the GHP provider networks performance. The monitoring program will include:
| - | Monthly periodic reporting of EHR adoption and PRHIN (STATE HIE) engagement; |
| - | Reporting requirements aligned with CMS EHR Meaningful Use criteria, CMS quality reporting and/ other data fields required by ASES; |
HIT ADOPTION AND PRHIN (STATE HIE) ENGAGEMENT EXPECTED TIMELINE
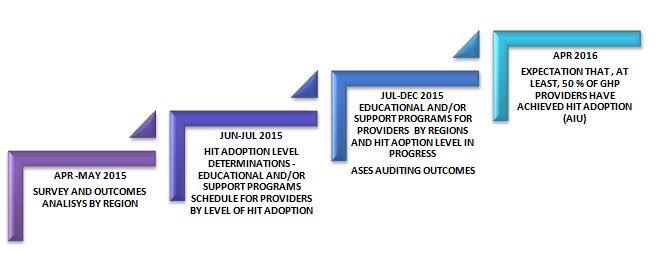
HIT ADOPTION AND PRHIN (STATE HIE) ENGAGEMENT OPERATIONAL PLAN
|
GOAL I.
Promote and require the GHP health care provider networks to adopt meaningful use of a certified electronic health record (EHR) and an active exchange of patient health information through a health information exchange PRHIN (STATE HIE)
|
||
|
OBJECTIVES
|
STRATEGIES
|
DATE
|
|
I.A. To obtain real time data on the GHP health care provider networks and the current status of their adoption and implementation of an EHR, Meaningful Use compliance, and their active participation in the PRHIN (STATE HIE).
|
I.A.1 Develop and submit to ASES for approval a survey tool related to the adoption and implementation of a certified EHR by the GHP healthcare providers and their participation in the PRHIN (STATE HIE). Preferably, the survey tool should be on-line.
I.A.2 Submit the EHR Adoption Survey to the providers. EHR Adoption Survey MUST be completed by May 8, 2015.
I.A.3 Collection and analysis of the EHR Adoption Survey results the contractors. Determine providers’ EHR adoption levels by Region. Preferably, the survey tool should be on-line.
I.A.4 Develop the EHR Adoption Communication/Education Plan for GHP health care provider networks in compliance with federal and state requirements. The EHR Adoption Communication/Education Plan will specify those GHP network providers that require additional targeted educational initiatives to be provided in order to accelerate adoption and effective use of EHRs within the GHP provider networks. Submit the EHR Adoption Communication/Education Plan for the GHP Health care provider networks to ASES for approval.
I.A.5 The MCO will be responsible to discuss GHP Insured Population/ Patient Education Plan with providers; encourage health care providers for the incorporation of privacy and security policies and procedures; and provide monitoring results to ASES.
|
April 6-17, 2015
April 20-May 8, 2015
May 11-29, 2015
June 1-19, 2015
June 22-July 10, 2015
|
|
I.BDevelop and schedule the educational initiatives to be offered to GHP health care providers
|
I.B.1 Educational initiatives begin targeting providers by EHR Adoption levels.
Educational programs must include:
· EHR adoption policy – federal and state overview
· EHR Medicaid Incentive Program
· Federal and State legal framework
· Level of meaningful use compliance
· Privacy and Security Frameworks
· Health Information Exchange Platform – Active Participation Requirements
|
July 13 – September 4, 2015
|
|
· Patients’ Rights
· Quality Improvement Programs/Measures Requirements
I.B.2 MCO will schedule the continuing education program for the GHP network providers along with the communication and engagement process for the health care providers.
I.B.3 MCO will conduct follow up surveys to audit the health care provider networks progress in increasing their EHR Adoption level and must provide findings to ASES.
|
September 7 - 25, 2015
September 28 – November 27, 2015
|
|
GOAL 2.
Ensure that health care provider networks to comply with Meaningful Use Goals
|
||
|
OBJECTIVES
|
STRATEGIES
|
DATE
|
|
2.A Monitor the Medicaid Meaningful Use certification process and compare with the data obtained under the educational program -follow up surveys
|
2.A.1 MCO will compare the results obtained from the follow up surveys from health care provider networks related to their progress in EHR Adoption level and the Meaningful Use Incentive Program
|
November 30 – December 18, 2015
|
|
GOAL 3.
Monitoring EHR Adoption and PRHIN (STATE HIE) Engagement
|
||
|
OBJECTIVES
|
STRATEGIES
|
DATE
|
|
3.A Report and analyze progress on EHR educational program
3.B Integrate a Quality Improvement Culture into GHP Provider Network
|
3.A.1 MCO will implement policies that require EHR and engagement with PRHIN (STATE HIE) the standard business practice for GHP Network Providers.
3.B.1 MCO will align EHR standards for quality measurements and improvements across GHP/Medicaid and Medicare programs.
3.B.2 MCO will accelerate alignment and implementation of electronic clinical quality measures and electronic reporting
3.B.3 ASES will develop standards and policies to enable electronic management of patient consent forms and PRHIN (STATE HIE) among GHP Network Providers with sensitive health data such as mental and behavioral health conditions.
3.B.4 ASES and the contractors will conduct follow up surveys to audit the health care provider networks progress in their HIT adoption, PRHIN (STATE HIE) participation, and quality measurement programs progress
|
January 11-29, 2016
January 11-29, 2016
February 1- April 1, 2016
April 4 –June 3, 2016
June 6 – September 30, 2016
|
ATTACHMENT 18
Business Associate Agreement
2015-000087
Business Associate Agreement
THIS AGREEMENT is made by and between Triple-S Salud, Inc., having its principal offices located at San Juan, PR ("Business Associate "), represented by Pablo Almodovar Scalley; and Ricardo A. Rivera Cardona, acting for and on behalf of The Puerto Rico Health Insurance Administration (PRHIA) ("Covered Entity") and with its principal offices at San Juan, Puerto Rico. Covered Entity and Business Associate, collectively, may hereinafter be referred to as the "Parties," as in the parties to this Agreement.
WHEREAS, Covered Entity and Business Associate are parties to one or more agreements and/or may in the future become parties to additional agreements (collectively, the "Underlying Agreements "), pursuant to which Business Associate provides certain services to Covered Entity and, in connection with such services, creates, receives, uses or discloses for or on behalf of Covered Entity certain individually identifiable Protected Health Information relating to patients and/or insured members of Covered Entity ("PHI") that is subject to protection under the Health Insurance Portability and Accountability Act of 1996 as amended by the Health Information Technology for Economic and Clinical Health Act Title XIII of Division A of the American Recovery and Reinvestment Act, 2009 (HITECH Act) and regulations promulgated there under, as such law and regulations may be amended from time to time (collectively, "HIPAA"); and
WHEREAS, Covered Entity and Business Associate wish to comply in respects with the requirements of HIPAA, including requirements applicable to the relationship between a Covered Entity and its Business Associates;
Section 1. Definitions.
| a. | "Breach " shall have same meaning given to such term as defined in 45 CFR § 160.102 |
| b. | “Business Associate" shall have the same meaning given to such term as defined in 45 CFR § 160.103. |
| c | “Covered Entity" shall have the same meaning given to such term as defined in 45 CFR § 160.103. |
| d. | "Designated Record Set" shall have the same meaning given to such term as defined in 45 CFR § 164.501. |
| e. | "Disclosure" shall have the same meaning given to such term as defined in 45 CFR §160.103. |
| f. | "Electronic Protected Health Information" or "e-PHI" shall have the same meaning given to such term as defined in 45 CFR §160.103 limited to the information transmitted or maintained by the Business Associate in electronic form format or media. |
| g. | "Individual" shall have the same meaning given to such term as defined in 45 CFR § 160.103 and shall include a person who qualifies as a personal representative in accordance with 45 CFR § 164.502(g). |
| h. | "Privacy Rule" shall mean the Standards for Privacy of Individually Identifiable Health Information at 45 CFR part 160 and part 164, subparts A and E respectively. |
| i | "Protected Health Information" or "PHI" shall have the same meaning given to such term as defined in 45 CFR §160.103, limited to the information created or received by Business Associate from or on behalf of Covered Entity. |
| j. | "Required By Law" shall have the same meaning given to such term as defined in 45 CFR§ 164.103 and The Health Information Technology for Economic and Clinical Health Act (HITECH) Division A: Title XIII, Subtitle D. |
| k. | "Security" or "Security Measures" encompass all of the administrative, physical, and technical safeguards in an information system specified in subpart C of 45, CFR § 164. |
| 1. | "Security Rule" shall mean the Standards for Security of Electronic Protected Health Information as specified in subparts A and C in 45 C.F.R. Parts 160 and 164, respectively. |
| m. | "Secretary" shall mean the Secretary of the Department of Health and Human Services or his/her designee. |
Section 2. Obligations and Activities of Business Associate.
2.1 1 Business Associate may not use or disclose Protected Health Information other than as permitted or required by the Underlying Agreement or as Required by Law.
2.2 Business Associate agrees to use appropriate safeguards, including without limitation, administrative, physical and technical safeguards, to prevent use or Disclosure of the Protected Health Information other than as provided for by this Agreement and to reasonably and appropriately employ the same standards as Required by Law to, protect the confidentiality, integrity and availability of any Electronic Protected Health Information (e-PHI) that it may receive, maintain or transmit on behalf of the Covered Entity.
2.3 Business Associate agrees to mitigate, to the extent practicable, any harmful effect that is known to Business Associate of a use or Disclosure of Protected Health Information by Business Associate in violation of the requirements of this Agreement.
2.4 Business Associate agrees to report to Covered Entity any use or Disclosure of the Protected Health Information not provided for by this Agreement or any Security incident resulting in an unauthorized access or acquisition of e-PHI, of which it becomes aware, involving Protected Health Information of the Covered Entity.
2.5 Business Associate must in accordance with 45 CFR 164.502(e)(l)(ii) and 164.308(b)(2), if applicable, ensure that any subcontractors, agents or affiliates of the Business Associate, that create, receive, maintain, or transmit PHI on behalf of the Business Associate agree to the same restrictions, conditions, and requirements that apply to the Business Associate with respect to such information. Business Associate must obtain satisfactory assurances in the form of a written agreement or memorandum of understanding directly from subcontractors stipulating that the subcontractor agrees to comply with the terms and conditions of the Business Associate Agreement. Business Associate must ensure that any agent or subcontractor to whom the Business Associate provides PHI, not export PHI beyond the borders of the Commonwealth of Puerto Rico without express written consent of the Covered Entity.
2.6. Business Associate agrees to provide access, at the written request of Covered Entity, and in the time and manner designated by Covered Entity, to Protected Health Information in a Designated Record Set, to Covered Entity in order to meet the requirements under 45 CFR §164.524.
2.7 Business Associate agrees to make any amendment(s) to Protected Health Information in a Designated Record Set that the Covered Entity directs or agrees to pursuant to 45 CFR §164.526 at the written request of Covered Entity or an Individ al, and in the time and manner designated by Covered Entity.
2.8 Business Associate agrees to make available internal practices, books, and records relating to the use and Disclosure of Protected Health Information received from, or created or received by Business Associate on behalf of, Covered Entity, or at the request of the Covered Entity to the Secretary, in a time and manner designated by the Covered Entity or the Secretary, for purposes of the Secretary determining Covered Entity's compliance with the Privacy and Security Rules.
2.9 Business Associate agrees to document such Disclosures of Protected Health Information and information related to such Disclosures as would be required for Covered Entity to respond to a request by an Individual for an accounting of Disclosures of Protected Health Information in accordance with 45 CFR §164.528.
2.10 Business Associate agrees to provide to Covered Entity or an Individual, in time and manner designated by Covered Entity, information collected in accordance with Section (l)(i) of this Agreement, to permit Covered Entity to respond to a request by an Individual for an accounting of Disclosures of Protected Health Information in accordance with 45 CFR §164.528.
2.11 Business Associate understands and agrees that it will not access or use any Protected Health Information of any Individual except for those Individuals whose PHI has been disclosed to Business Associate and it will further limit access to that Protected Health Information that is necessary to the activities undertaken by Business Associate on behalf of Covered Entity.
2.13 Business Associate will, pursuant to the HITECH Act and its implementing regulations, comply with the requirements of the Privacy Rule, including those contained in 45 CFR §§ 164.502(e) and 164.504(e)(l)(ii), at such time as the requirements are applicable to Business Associate. Business Associate will not directly or indirectly receive remuneration in exchange for any Protected Health Information, subject to the exceptions contained in the HITECH Act, without a valid authorization from the applicable Individual. Business Associate will not engage in any communication which might be deemed to be "Marketing" under the HITECH Act. In addition, Business Associate will, pursuant to the HITECH Act and its implementing regulations, comply with all applicable requirements of the Security Rule, contained in 45 CFR § 164.308, 164.310, 164.312 and 163.316 at such time as the requirements are applicable to Business Associate.
Section 3. Permitted Uses and Disclosures by Business Associate.
3.1 In case Business Associate obtains or creates Protected Health Information, Business Associate may use or disclose Protected Health Information, or any information derived from that Protected Health Information, only as explicitly permitted in the underlying agreement, and only if such use or Disclosure, respectively, is in compliance with each applicable requirement of 45 CFR § 164.504(e). It means that:
3.1.1 Except as otherwise limited in this Agreement, Business Associate may use Protected Health Information for the proper management and administration of the Business Associate or to carry out the legal responsibilities of the Business Associate.
3.1.2 Except as otherwise limited in this Agreement, Business Associate may disclose Protected Health Information for the proper management and administration of the Business Associate, provided that Disclosures are Required By Law, or Business Associate obtains reasonable assurances from the person/organi zation to whom the information is disclosed that it will remain confidential and used or further disclosed only as Required By Law or for the purpose for which it was disclosed to the person/organization , and the person/organization notifies the Business Associate of any instances of which it is aware in which the confidentiality of the information has been Breached.
3.1.3 In accordance with 45 CPR 164.502(e)(l)(ii) and 164.308(b)(2), if applicable, ensure that any subcontractors that create, receive, maintain, or transmit Protected Health Information on behalf of the Business Associate agree to the same restrictions, conditions, and requirements that apply to the Business Associate with respect to such information .
3.2 Business Associate understands and agrees that its access to Protected Health Information stored in databases and information systems at the Covered Entity is subject to review and audit by the Covered Entity or agents of the HHS and OCR at any time, that remote audits of such access may occur at any time, that on-site audits of such access will be conducted during regular business hours, and that any review or audit may occur with or without prior notice by the Covered Entity.
Section 4. Application of Security and Privacy Provisions to Business Associate.
4.1 Security Measures: 45 CFR §164.308, 164.310, 164.312 and 164.316, dealing with the administrative, physical and technical safeguards as well as policies, procedures and documentation requirements that apply to Covered Entity shall in the same manner apply to Business Associate as Required By Law. Any additional Security requirements contained in Division A Title XIII Health Information Technology of the American Recovery and Reinvestment Act that apply to Covered Entity shall also apply to Business Associate as of February 17, 2010. Business Associates that require access to Covered Entity electronic patient information systems, electronic health record (EHR) and electronic infrastructure systems (either on site or remote) will supply the necessary information of employees to uniquely identify such employees, as employees with a need to access systems and will supply to Covered Entity Information Security Officer a valid state or federal issued photo ID for such employees to receive a unique user name and password to access the system(s).
4.2 Application of Civil and Criminal Penalties- If Business Associate violates any Security provision as Required By Law specified in Section 4.1 above, sections 1176 and 1177 of the Social Security Act 42 U.S.C. §1320d-5, 1320d-6 shall apply to Business Associate with respect to such violation in the same manner that such sections apply to Covered Entity if it violates such Security provision.
Section 5. Information Breach Notification Requirements.
5.1 Business Associate expressly recognizes that Covered Entity has certain reporting and Disclosure obligations to the Secretary of the Department of Health and Human Services and the Individual in case of a Security Breach of unsecured Protected Health Information (as defined in 45 CFR §164.402).
5.2 Where Business Associate accesses, maintains, retains, modifies, records, stores, destroys, or otherwise holds, uses, or discloses unsecured Protected Health Information, Business Associate without unreasonable delay and in no case later than thirty (30) days following the discovery of a Breach of such information, shall notify Covered Entity of such Breach. Such notice shall include the identification of each Individual whose unsecured Protected Health Information has been, or is reasonably believed by the Business Associate to have been, accessed, acquired or disclosed during the Breach.
5.3 Business Associate shall be liable for the costs associated with such Breach if caused by the Business Associate's negligent or willful acts or omissions, or the negligent or willful acts or omissions of Business Associate's agents, officers, employees or subcontractors.
Section 6. Insurance and Indemnification
6.1 Indemnification. The Business Associate agrees to indemnify, defend and hold harmless Covered Entity and Covered Entity's employees, directors, officers, subcontractors, agents or other members of its workforce from any costs, damages, expenses, judgments , losses, and attorney 's fees arising from any breach of this Agreement by Business Associate, or arising from any negligent or wrongful acts or omissions of Business Associate, including failure to perform its obligations under the Privacy Rule. The Business Associate's indemnification obligation shall survive the expiration or termination of this Agreement for any reason.
Section 7. Terms and Termination.
7.l Term. The Term of this Agreement shall commence as of the Effective Date (as defined below), and shall terminate on the termination date of the underlying agreement or on the date Covered Entity terminates this agreement for cause as authorized on paragraph (7.2) of this section, whichever is sooner.
7.2 Termination for Cause. The parties acknowledge that in the event the Covered Entity learns of a pattern or activity or practice of the Business Associate that constitutes violation of a material term of this Agreement , then the parties promptly shall take reasonable steps to cure the violation. If such steps are, in the judgment of the Covered Entity, unsuccessful, ineffective or not feasible, then the Covered Entity may terminate, in its sole discretion, any or all of the Underlying Agreements upon written notice to the Business Associate, if feasible, and if not feasible, shall report the violation to the Secretary of the Department of Health and Human Services.
7.3 Effect of Termination. Except as provided in paragraph (2) of this section, upon termination of this Agreement or the Underlying Agreement(s) for any reason, Business Associate shall return or destroy all Protected Health Information pursuant to 45 C.F.R. § 164.504(e)(2)(I) received from Covered Entity, or created or received by Business Associate on behalf of Covered Entity. This provision shall apply to Protected Health Information that is in the possession of subcontractors or agents of Business Associate. Business Associate shall retain no copies of the Protected Health Information.
In the event that Business Associate determines that returning or destroying the Protected Health Information is infeasible, Business Associate shall provide to Covered Entity notification, in writing, of the conditions that make return or destruction infeasible. Said notification shall include: (i) a statement that the Business Associate has determined that it is not feasible to return or destroy the Protected Health Information in its possession , and (ii) the specific reasons for such determination. The Covered Entity may disagree with the Business Associate' s determination. Upon mutual agreement of the Parties that return or destruction of Protected Health Information is infeasible, Business Associate shall extend the protections of this Agreement to such Protected Health Information and limit further uses and Disclosures of such Protected Health Information to those purposes that make the return or destruction infeasible, for as long as Business Associate maintains such Protected Health Information. If it is infeasible for the Business Associates to obtain, from a subcontractor or agent, any Protected Health Infonnation in the possession of the subcontractor or agent, the Business Associate must provide a written explanation to Covered Entity and require the subcontractors and agents to agree to extend any and all protections, limitations, and restrictions contained in this Agreement to the subcontractors; and/or agents' use and/or Disclosure of any Protected Health Information retained after the termination of this Agreement , and to limit any further uses and/or Disclosures to the purposes that make the return or destruction of Protected Health Information infeasible.
7.4 Automatic Termination. This agreement will automatically terminate without any further action of the Parties upon termination or expiration of the Underlying Agreement.
7.5 Effective Date. The effective date of this Agreement (the “Effective Date”) shall be the date of the last signature below.
Section 8. Miscellaneous.
8.1 Regulatory References. A referent in this Agreement to a section in the Privacy and Security Rules means the section as in effect or as amended, and for which compliance is required.
8.2 Agreement. The Parties agree to take such action as is necessary to amend the Underlying Agreement from time to time as is necessary for Covered Entity to comply with the requirements of the HIPAA; provided.
8.3 Amendments; Waiver. This agreement may not be modified, nor shall any provision hereof be waived or amended, except in a writing duly signed by authorized representatives of the Parties. A waiver with respect to one event shall not be construed as continuing, or as a bar to a waiver of any right or remedy as to subsequent events. The Parties agree to take such action as is necessary to amend this agreement from time to time as is necessary for compliance with the requirements of the HIPAA rules and any other applicable law.
8.4 Survival. The respective rights and obligations of Business Associate under this Agreement and Covered Entity under this Agreement shall survive the termination of this Agreement and/or the Underlying Agreements, as shall the rights of access and inspection of Covered Entity.
8.5 No Third Party Beneficiaries. Nothing express or implied in this Agreement is intended to confer, nor shall anything herein confer, upon any person other than the parties and the respective successors or assigns of the Parties, any rights, remedies, obligations, or liabilities whatsoever.
8.6 Interpretation. Any ambiguity in this Agreement shall be resolved in favor of a meaning that permits Covered Entity to comply with the HIPAA Privacy and Security Rules.
|
[Signed]
Pablo Almodóvar Scalley
President/CEO
Triple-S Salud, Inc.
SS# [redacted]
|
|
[Signed]
Ricardo A. Rivera Cardona
Executive Director
PR Health Insurance Administration
SS# [redacted]
|
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
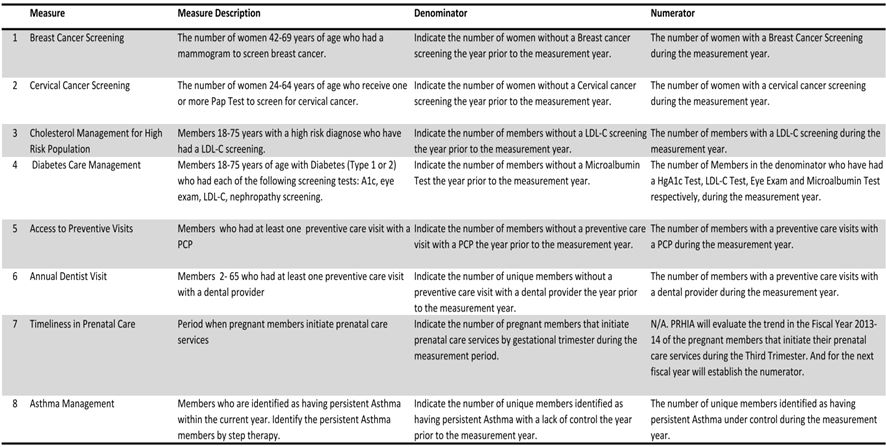


COMMONWEALTH OF PUERTO RICO
PLANNING AND QUALITY AFFAIRS OFFICE
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1
Version 2.1, Reviewed: July 2014
M. Espada, Y. Berríos & N. Ortiz
|
I.
|
Table of Contents
|
|
II.
|
INTRODUCTION
|
3
|
|
III.
|
RETENTION FUND
|
3
|
|
IV.
|
DEFINITIONS
|
4
|
|
V.
|
PREVENTIVE CLINICAL PROGRAMS
|
6
|
|
VI.
|
PERFORMANCE MEASURES
|
12
|
|
VII.
|
ER QUALITY PROGRAM
|
17
|
|
VIII.
|
CONCLUSION
|
18
|
Year 2014
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 2 of 18
|
|
II.
|
INTRODUCTION
|
This Quality Improvement Procedure Manual has the sole purpose of providing the necessary guidelines for attaining the required performance indicators for each of the categories measured under the Quality Incentive Program (QIP), as described in Article 12 of the contract executed between the Contractor and the Puerto Rico Health Insurance Administration (ASES, by its acronym in Spanish). ASES shall maintain a Retention Fund of the Per Member Per Month (PMPM) each month as part of the Quality Incentive Program described in Section 12.5.3 A portion of the retained amount shall be associated with each of the Quality Incentive initiatives outlined below:
|
●
|
Performance measures (Section 12.5.4.1)
|
|
●
|
Preventive Clinical Programs (Section 12.5.4.2)
|
|
●
|
Emergency Room Use Indicators (Section 12.5.4.3)
|
ASES will reimburse the Contractor according to compliance with each of the categories of performance indicators in section 12.5. The Planning and Quality Affairs Office will audit the results of the data in the timeframes stated in Section 12.5 of the Contract for the performance indicators in the following categories: Performance measures, Preventive clinical program measures, and ER Utilization measures. This Manual describes in detail the requirements and the specific metrics for each category of the Quality Incentive Program. The Quality Improvement Procedure Manual will enter in effect the Effective Date of the Contract and will be revised every contract year unless required in another timeframe by law or regulation, at the discretion of ASES or by mutual agreement during the term of the contract year.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 3 of 18
|
|
III.
|
RETENTION FUND
|
ASES will withhold a portion of annual PMPM otherwise payable to the Contractor in order to incent the Contractor to meet performance targets under the Quality Incentive Program. The retention fund will be reimbursed to the Contractor when a determination is made by ASES that the Contractor has complied with the quality standards and criteria established by ASES in accordance with 22.3 of the contract. On a quarterly basis the Contractor will submit a quarterly Retention Fund Report in accordance to 18.2.9.4 of the contract.
On a monthly basis, ASES will maintain a retention fund according to the following timeframes:
|
Time Period
(Relative Effective Data of Contract Term)
|
Retention Fund
Percentage
|
|
4/1/2015 through 12/31/2015
|
0 % (9 month baseline)
|
|
1/1/2016 through 6/30/2016
|
1% (until end of FY16)
|
|
7/1/2016 through 6/30/2017
|
2% (until end of FY17)
|
A portion of the retained amount will be associated with each of the Quality Incentive initiatives outlined below for each of the specified timeframes:
|
QIP Initiative
|
Retention Fund Breakdown
|
|||||||||||
|
Year
|
CY 15 (0%)1
|
FY 16 (1%)
|
FY 17 (2%)
|
|||||||||
|
Performance Measures
|
0
|
%
|
.40
|
%
|
.80
|
%
|
||||||
|
Preventive Clinical Programs
|
0
|
%
|
.20
|
%
|
.40
|
%
|
||||||
|
Emergency Room Use Indicators
|
0
|
%
|
.40
|
%
|
.80
|
%
|
||||||
|
IV.
|
DEFINITIONS
|
The following definitions apply to measures of the Quality Improvement Manual:
|
1.
|
Care Management: An Administrative Function comprised of a set of Enrollee-centered steps to ensure that an Enrollee with intensive needs, including catastrophic or high-risk conditions (described in Attachment 7 of the Contract), receives needed services in a supportive, effective, efficient, timely, and cost-effective manner.
|
1 The first 9 months from the date of an executed contract ASES will not withhold a retention fund. The first 9 month time period will be used a grace period to determine baseline data for each QIP initiative. The period will end on 12/31/2015. At that time a 1% Retention Fund withholds will be activated.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 4 of 18
|
|
2.
|
Disease Management: An Administrative Function comprised of a set of Enrollee-centered steps to provide coordinated care to Enrollees suffering from diseases listed in Section [7.8.3] of this Contract.
|
|
3.
|
Hot Spotting: The ability to identify in a timely manner heavy users of the systems and their patterns of utilization to provide targeted interventions and care through mapping data.
|
|
4.
|
Incurred date: Is the date in which the service took place.
|
|
5.
|
Intervention: activities targeted at the achievement of client stability, wellness, and autonomy through advocacy, assessment, planning, communication, education, resource management, care coordination, collaboration, and service facilitation.
|
|
6.
|
Performance measures: regular measurement of outcomes and results, which generates reliable data on the effectiveness and efficiency of programs.
|
|
7.
|
Per member per month payment (PMPM): The fixed monthly amount that the Contractor is paid by ASES for each Enrollee to ensure that Benefits under this Contract are provided. This payment is made regardless of whether the Enrollee receives Benefits during the period covered by the payment.
|
|
8.
|
Preventive Services: Health care services provided by a physician or other Provider within the scope of his or her practice under Puerto Rico law to detect or prevent disease, disability, Behavioral Health conditions, or other health conditions; and to promote physical and Behavioral Health and efficiency.
|
|
9.
|
Primary Care Physician: A licensed medical doctor (MD) who is a Provider and who, within the scope of practice and in accordance with Puerto Rico Certification and licensure requirements, is responsible for providing all required Primary Care to Enrollees. The PCP is responsible for determining services required by Enrollees, provides continuity of care, and provides Referrals for Enrollees when Medically Necessary. A PCP may be a general practitioner, family physician, internal medicine physician, obstetrician/gynecologist, or pediatrician.
|
|
10.
|
Retention Fund: The amount of Withhold by ASES of the monthly Per Member per Month Payments otherwise payable to the Contractor in order to incentivize the Contractor to meet performance targets under the Quality Incentive Program described in Section [12.5.3]. This amount shall be equal to the percent of that portion of the total Per Member per Month Payment that is determined to be attributable to the Contractor’s administration of the Quality Incentive Program described in Sections [12.5 and 22.3]. Amounts withheld will be reimbursed to the Contractor in whole or in part (as set forth in Sections [12.5 and 22.3]) in the event of a determination by ASES that the Contractor has complied with the quality standards and criteria established by Section [12.5].
|
|
11.
|
Special Coverage: A component of Covered Services provided by the Contractor, described in Section [7.7], which are more extensive than the Basic Coverage services, and for which Enrollees are eligible only by “registering.” Registration for Special Coverage is based on intensive medical needs occasioned by serious illness.
|
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 5 of 18
|
|
12.
|
Quality Incentive Program: mechanism to improve the quality of services provided to Enrollees. The program shall consist of three (3) categories of performance indicators: performance measures, preventive clinical program measures and ER Utilization measures.
|
|
V.
|
PREVENTIVE CLINICAL PROGRAMS
|
The Contractor shall comply with the objectives of each of the following Preventive Clinical Programs as stated in the GHP Contract in section 12.5.4.2. The Preventive Clinical Programs are:
1. Pre-Natal and Maternal Program as described in 7.5.8.3 of the Contract.
2. Wellness Plan as described in section 12.5.8 of the Contract.
3. Care Management as described in section 7.8.2 of the Contract.
4. Disease Management as described in 7.8.3 of the Contract.
5. Provider Education as described in section 10.2.2 of the Contract.
6. Physician Incentive Programs as described in section 10.7 of the Contract.
ASES shall release to the Contractor, in accordance with Section 22.3, the applicable percent (see table below) of the Retention fund for compliance with the objectives for each of the Preventive Clinical Programs.
|
QIP Initiative
|
Retention Fund Breakdown
|
|||||||||||
|
Year
|
CY 15 (0%)2
|
FY 16 (1%)
|
FY 17 (2%)
|
|||||||||
|
Preventive Clinical Programs
|
0
|
%
|
.20
|
%
|
.40
|
%
|
||||||
2 The first 9 months from the date of an executed contract ASES will not withhold a retention fund. The first 9 month time period will be used a grace period to determine baseline data for each QIP initiative. The period will end on 12/31/2015. At that time a 1% Retention Fund withholds will be activated.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 6 of 18
|
|
1.
|
Prenatal and Maternal Plan
|
Goal Statement: Increase the number of pregnant women who receive early prenatal care.
REPORTING:For each region, report the following metrics for enrollees receiving prenatal and maternal services:
|
o
|
Number of pregnant women enrolled in GHP by trimester and age;
|
|
o
|
Number of pregnant women enrolled in GHP by trimester and age who received HIV tests;
|
|
o
|
Number of pregnant women screened for substance abuse with the 4P Plus screening tool;
|
|
o
|
Number of pregnant women in postpartum care screened for depression with the Edinburgh screening tool;
|
|
o
|
Number of pregnant women who received educational interventions.
|
OUTCOME(S): Increase annually by 3% the number of pregnant women with HIV tests in the First and Third Trimester as established by public policy of the Department of Health.
|
o
|
In quarters 1-3 the Contractor will report the number of Providers (OB-GYN) and pregnant enrollees with educational interventions.
|
|
o
|
In the 4th quarter the Contractor will report the 3% increase in HIV tests among pregnant enrollees using as comparison the trend of HIV testing in births from August 1, 2013 through April 30, 2014.
|
|
●
|
Screen 50% of pregnant women registered by quarter for alcohol and tobacco use with 4P Plus screening tool.
|
|
o
|
Report the number of cases referred to the behavioral health provider for smoking cessation counseling and treatment.
|
|
●
|
Screen 50% of women in postpartum period during the measurement quarter for Depression using Edinburgh screening tool.
|
|
o
|
Report the number of cases referred to the behavioral health provider with an Edinburgh score of 10 or above.
|
|
●
|
Reach 70% of pregnant women in registry with educational interventions regarding Prenatal care in the following topics :
|
1. Importance of Prenatal and Postpartum visits.
2. Breastfeeding
3. Stages of birth
4. Oral Health
5. Family Planning
6. Behavioral Health topics in the areas of Domestic Violence, Post partum Depression, Tobacco Cessation, Alcohol Use/Abstinence and Substance Abuse, Parenting, HIV Screening and prevention and socio emotional screening in children.
7. Newborn Care
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 7 of 18
|
|
2.
|
Wellness Program:
|
Goal Statement: Increase the number of members who receive preventive health information and services.
OUTCOME(S):
|
●
|
Develop 5 educational campaigns on the following topics to be applied during the measurement year:
|
a. Nutrition and Exercise; Knowing your BMI
b. Importance of preventive dental exam
c. Awareness of HPV vaccination
d. Preventive Cancer Screening (PAPS, Mammography, Oral cancer examination).
e. Stress Management
|
o
|
Minimum 1 educational campaign by quarter.
|
The Contractor shall submit a detailed description of the Educational Campaign and copies of all materials (written and oral) that it or its Subcontractors plan to distribute to ASES for review and approval. This requirement includes, but is not limited to posters, brochures, Web sites, and any other related materials. Neither the Contractor nor its Subcontractors shall distribute any materials without prior written approval from ASES.
|
●
|
EPSDT:
|
|
o
|
Quarterly educational outreach for PCPs providing a list of EPSDT eligible children who are not in compliance with periodicity schedule.
|
|
o
|
The Contractor shall provide to each PCP, at least four times per year (April, July, November and January), a list of the PCP’s EPSDT Eligible Children who are not in compliance with the EPSDT periodicity Schedule.
|
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 8 of 18
|
|
3.
|
Care Management Program
|
Goal Statement: Achievement of optimal health, integration of Physical Health and Behavioral Health services, access to care and appropriate utilization of resources, balanced with the patient's right to self-determination.
OUTCOME(s):
|
●
|
New cases
|
|
o
|
Report on the number of new enrollees in Care Management in the following categories:
|
|
●
|
Enrollees with special health care needs who qualify for Special Coverage
|
|
●
|
Enrollees diagnosed with a Serious Mental Illness or a Serious Emotional Disability (“SMI/SED”)
|
|
●
|
Enrollees participating in the Buprenorphine program
|
|
●
|
Enrollees who have accessed the emergency room seven (7) or more times within twelve (12) months
|
|
●
|
Enrollees who are pregnant and have a behavioral health diagnosis.
|
|
o
|
Report on Prior Authorizations (PA) and Prior Authorization denials on each condition on special coverage registry and SMI/SED including Buprenorphine program)
|
|
●
|
Screen at least 50% of adult members registered in Special Coverage for depression using PHQ-93 screening tool.
|
|
o
|
Report the number of cases referred to the behavioral health provider with a PHQ-9 score of 10 or above.
|
|
4.
|
Disease Management Program
|
Goal Statement: Enhance the treatment and prevention of diseases that contribute most heavily to the causes of death.
OUTCOME(s):
3 PHQ-9: Patient Health Questionnaire – 9 (Screening for depression)
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 9 of 18
|
|
●
|
Submit a hot spotting report by Region, PMG and municipality of residence of enrollees stratified by the following conditions:
|
|
o
|
Asthma
|
|
o
|
Diabetes (Type 1 or 2)
|
|
o
|
Congestive Heart Failure
|
|
o
|
Hypertension
|
|
o
|
Chronic renal disease (Stages 1 and 2)
|
|
o
|
Obesity
|
|
o
|
Mental health disorders
|
|
o
|
Alcohol abuse or dependence
|
|
o
|
Substance use disorders
|
The report must include: number of severe cases identified, percent of severe cases among PMG population, number of active cases, number of health cases referred to mental health treatment, and the number of interventions (educational and care coordination) performed for the population identified.
Minimum per quarter: one intervention by member.
|
5.
|
Provider Education
|
Goal Statement: The Provider Education Program is aimed to promote compliance with clinical quality guidelines and standards among all primary care physicians, and to keep them up to date regarding the best practices in the managed care model.
OUTCOME(s):
Provide educational activities to PCP and BHPCPs in coordination with the PBM providers for the following topics:
|
o
|
Primary Care Integration Model
|
|
o
|
Poly-pharmacy
|
|
o
|
EHR Poly-pharmacy
|
|
o
|
Electronic Health Records/e-prescribing
|
|
o
|
Diabetes Care Management
|
|
o
|
Renal Clinical Guidelines
|
|
o
|
Quality Incentive Program Guidelines
|
|
o
|
Mental health conditions
|
|
o
|
Working with patients with conditions of special concern, including autism, ADHD, depression, diabetes, alcohol and substance abuse, tobacco cessation, among others.
|
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 10 of 18
|
The Contractor must provide a minimum of 5 hours per quarter for a total of 20 hours per year. A report on topics, contact hours, PBM and providers attending the activities will be provided each quarter.
|
●
|
Reach 70% of PCPs (with 25 or more pediatric assigned lives) with technical assistance4 in the administration of MCHAT and Ages and Stages Questionnaire (ASQ) in their practices, with a minimum per quarter of 17.5%.
|
|
6.
|
Physician Incentive Plan
|
Goal Statement: Ensure the participation and commitment of the PCPs to Preventive Services and improve the quality of the services to all members.
OUTCOME(s):
|
●
|
Evaluate 100% of the PCPs through Medical Record Review:
|
|
o
|
The Contractor will submit in the first three quarters reports on the number of PCP evaluated and the score obtained by region and Integrated PMG.
|
|
o
|
By April 2014 the Contractor will provide a list by Integrated PMG and region of the certified PCP and BHP eligible for the financial incentive.
|
4 Technical Assistance: to assist providers to attain and maintain regulatory standards.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 11 of 18
|
|
VI.
|
PERFORMANCE MEASURES
|
The reporting templates for each of the performance measures mentioned below will be provided to the Contractor through the ASES FTP site. Each reporting template will be in Excel format. ASES shall reimburse the Contractor the percent applicable of the Retention fund as shown in the following table in accordance with Section 22.3 of the contract for successful compliance with the performance measures below based upon annual evaluation of this criterion. The Contractor shall demonstrate a three percent (3%) increase in the measurement year (.75 quarterly),for the following performance measures:
|
QIP Initiative
|
Retention Fund Breakdown
|
|||||||||||
|
Year
|
CY 15 (0%)1
|
FY 16 (1%)
|
FY 17 (2%)
|
|||||||||
|
Performance Measures
|
0
|
%
|
.40
|
%
|
.80
|
%
|
||||||
|
●
|
Breast Cancer Screening |
|
●
|
Cervical Cancer Screening |
|
●
|
Cholesterol Management |
|
●
|
Diabetes Care Management |
|
●
|
Access to Preventive Care Visits |
|
●
|
Access to Dental Preventive Care Visits |
|
●
|
Timeliness in Prenatal Care |
|
●
|
Asthma Management |
|
●
|
Follow-up care for children prescribed ADHD medication |
|
●
|
Antidepressant medication management |
|
●
|
Initiation of drug or alcohol abuse treatment |
|
●
|
Follow up after hospitalization for mental health |
The Performance Measures reports are based on claims incurred in the measurement period for each region. The Contractor shall provide data for each region in a separate tab and a summary tab that combines data for all regions.
For each report submission, the Contractor shall use the same template that was submitted in previous quarter(s). The Contractor may not update data submitted for previous reporting periods when new claims data is available. Then, with the 4th quarter submission, the Contractor may update data submitted for previous reporting periods (“year to date”) as applicable.
5 The first 9 months from the date of an executed contract ASES will not withhold a retention fund. The first 9 month time period will be used a grace period to determine baseline data for each QIP initiative. The period will end on 12/31/2015. At that time a 1% Retention Fund withholds will be activated.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 12 of 18
|
The Contractor shall submit the report with the following file name structure: Contractor Name_PM##_ Date Report Due (e.g. Contractor A_ PM01_ 20140150). The Contractor shall ensure that all data is captured in the workbook prior to submitting the report.
The 1st submission of the reports, excluding the Timeliness in Prenatal Care, will establish the baseline for each measure. Regarding the Timeliness in Prenatal Care, the Contractor will report the trend of the year prior the measurement year. The submissions dates for this reports will be provided by ASES through a normative letter with the established due dates.
SPECIAL AGREEMENTS
| 1. | The Contractor shall demonstrate a sustained improvement by performance measure on a quarterly basis. |
| 2. | The number of members with a LDL-C screening during the measurement year will be evaluated in the Cholesterol Management for High Risk Population Performance Measure. The Contractor will include the results under the Diabetes Care Management for information purposes only. |
| 3. | Asthma Management for Contract Year 2015-2016 |
| i. | For this measure, PRHIA will evaluate, as the results of the Contractor interventions the population outreached for this purposes during the first to second quarter. At the end of the 3rd quarter onwards the Contractor will report the 3% increase of unique members identified as having persistent Asthma under control during the measurement year (Steps 2-5). |
| ii. | Definitions of Treatment Steps are based on The Global Strategy for Asthma Management and Prevention (www.ginasthma.org). |
| 4. | Timeliness in Prenatal Care |
| i. | PRHIA will evaluate the trend in the Contract Year 2015-16 of the pregnant members that initiate their prenatal care services during the Third Trimester. And for the next contract year will establish the numerator. |
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 13 of 18
|
REQUIREMENTS BY PERFORMANCE MEASURE
Physical Health

|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 14 of 18
|
Behavioral Health

|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 15 of 18
|

|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 16 of 18
|
|
VII.
|
ER QUALITY PROGRAM
|
Goal Statement: Develop an ER Quality Initiative Program focusing on reducing the inappropriate use of ER services.
The ER Quality Initiative Program shall be designed to identify high users of Emergency Services (including behavioral health) for non-emergency situations and to allow for early interventions in order to ensure appropriate utilization of services and resources. The program design required by ASES for the ER Quality Initiative will be based on the “Hot Spotting Model of the Camden Coalition of Health Providers”. The activities for the work plan shall include, but not limited to, the following:
|
●
|
Hot spotting report by Region, PMG and municipality of residence of enrollees by severity level. |
|
●
|
Establish outreach activities and care coordination for High ER utilizers. |
Member identification will be as follows:
|
Severity Criteria
|
|
|
Level 1: Mild
|
3-6 visits a year
|
|
Level 2: Moderate
|
7-11 visits a year
|
|
Level 3: Severe
|
12 or more visits a year
|
ASES will reimburse to the Contractor, in accordance with Section 22.3 the percent applicable of the Retention fund as shown in the table below for compliance with the above program based upon a quarterly review of the approved work plan.
|
QIP Initiative
|
Retention Fund Breakdown
|
|||||||||||
|
Year
|
CY 15 (0%)1
|
FY 16 (1%)
|
FY 17 (2%)
|
|||||||||
|
Emergency Room Use Indicators
|
0
|
%
|
.40
|
%
|
.80
|
%
|
||||||
6 The first 9 months from the date of an executed contract ASES will not withhold a retention fund. The first 9 month time period will be used a grace period to determine baseline data for each QIP initiative. The period will end on 12/31/2015. At that time a 1% Retention Fund withholds will be activated.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 17 of 18
|
|
VIII.
|
CONCLUSION
|
The compliance with the quality categories established in this Manual will be measured and shall be accomplished with by the Contractor on a quarterly basis. Contractor shall comply with the required quarterly metrics in order to receive the reimbursement of the amount retained by ASES for each quarter as defined in Section 22.3 of the Contract.
|
QUALITY IMPROVEMENT PROCEDURE MANUAL Version 2.1 / July 2014
|
Page 18 of 18
|
ATTACHMENT 20
PREFERRED PROVIDER NETWORK DIAGRAM

ATTACHMENT 21
Guidelines for Reverse Collocation of the
primary care physicians in mental health facilities
In accordance with the provisions of the Puerto Rico Mental Health Code, Law No. 408 of October 2, 2000, as amended, and the Puerto Rico Patient’s Bill of Rights and Responsibilities, the Government Health Plan (GHP) is committed to promoting mental and physical health integration, in order to improve program effectiveness and quality of life for enrollees.
Reverse collocation is an integrated care model in which medical services are available to members being treated in behavioral health facilities. It has been known that patients with co‐morbid conditions that include chronic or acute medical conditions and behavioral health diagnoses are at higher risk for increased utilization and costs in health care. Persons with serious mental illness have high levels of medical co-morbidity compared to the general population, as well as increased risk for diabetes, obesity, and high cholesterol due to the use of some second-generation antipsychotic medications (Milbank Memorial Fund, 2010)
In the reverse collocation model, a primary care physician is located part or full time in a behavioral health facility to monitor the physical health of patients.
Collocated Primary Care Physicians (PCPs) are independently sub-contracted and supervised by the contractor. They use the Behavioral Health Facility (BHF) records, and coordinate follow up with the member’s PMG as necessary. The Collocated PCP can make the same primary interventions and referrals as any PCP in the PMG.
Behavioral Health Facilities (BHF)
The following BHF will be considered for purposes of the Reverse Collocation requirements.
| 1. | Psychiatric Hospitals ( or a unit within a general hospital) |
| 2. | Emergency or Stabilization Units |
| 3. | Partial Hospitalization Units |
| 4. | Intensive Ambulatory Services Units |
| 5. | Ambulatory Services Units |
| 6. | Addiction Services Unit ( detoxification, ambulatory, inpatient) |
Required Reverse Collocation Staff per BHF.
| 1. | Ambulatory Services Units must have at least one collocated PCP 4 days per week for 4 hours. |
| 2. | Addiction Services Units must have at least one collocated PCP 3 days per week for 4 hours. |
| 3. | Psychiatric Hospitals are required to have at least a PCP on call on a daily basis. |
1
| 4. | Partial Hospitalization Units must have at least one collocated PCP 2 days per week for 4 hours. |
| 5. | Stabilization units must have one PCP for consultation (on call) on a daily basis. |
In the event that a BHF does not comply with the required collocation level, may be subjected to penalties according to the following matrix:
|
Sanction Level
|
Sanction Type
|
Timeframe to cure
|
Comments
|
|
0
|
Notice of Non Compliance with Reverse Collocation Level and CAP
|
60 days
(Day 1-60)
|
A corrective action plan (“CAP”) will be required of every BHF that does not comply with the required Reverse Collocation level. All BHF with an approved CAP must comply with the terms of the CAP and achieve the required collocation within the timeframes established in the CAP.
|
|
1
|
Fine
|
30 days
(Day 61-90)
|
Fines to be defined in accordance to contract.
|
|
2
|
Contract Cancelation
|
(Day 91)
|
2


Form Approved: OMB No. 0937-0166 Expiration date: 10/31/2015 CONSENT FOR STERILIZATION NOTICE: YOUR DECISION AT ANY TIME NOT TO BE STERILIZED WILL NOT RESULT IN THE WITHDRAWAL OR WITHHOLDING OF ANY BENEFITS PROVIDED BY PROGRAMS OR PROJECTS RECEIVING FEDERAL FUNDS.
• CONSENT TO STERILIZATION • I have asked for and received information about sterilization from __________________________________________. When I first asked Doctor or Clinic for the information, I was told that the decision to be sterilized is completely up to me. I was told that I could decide not to be sterilized. If I decide not to be sterilized, my decision will not affect my right to future care or treatment. I will not lose any help or benefits from programs receiving Federal funds, such as Temporary Assistance for Needy Families (TANF) or Medicaid that I am now getting or for which I may become eligible. I UNDERSTAND THAT THE STERILIZATION MUST BE CONSIDERED PERMANENT AND NOT REVERSIBLE. I HAVE DECIDED THAT I DO NOT WANT TO BECOME PREGNANT, BEAR CHILDREN OR FATHER CHILDREN. I was told about those temporary methods of birth control that are available and could be provided to me which will allow me to bear or father a child in the future. I have rejected these alternatives and chosen to be sterilized. I understand that I will be sterilized by an operation known as a _. The discomforts, risks Specify Tipe of Operation and benefits associated with the operation have been explained to me. All my questions have been answered to my satisfaction. I understand that the operation will not be done until at least 30 days after I sign this form. I understand that I can change my mind at any time and that my decision at any time not to be sterilized will not result in the withholding of any benefits or medical services provided by federally funded programs. I am at least 21 years of age and was born on: __ Date I, hereby consent of my own free will to be sterilized by Doctor or Clinic by a method called __. My Specify Type of Operation consent expires 180 days from the date of my signature below. I also consent to the release of this form and other medical records about the operation to: Representatives of the Department of Health and Human Services, or Employees of programs or projects funded by the Department but only for determining if Federal laws were observed. I have received a copy of this form. Signature Date You are requested to supply the following information, but it is not required: (Ethnicity and Race Designation) (please check) Ethnicity: Race (mark one or more): Hispanic or Latino American Indian or Alaska Native Not Hispanic or Latino Asian Black or African American Native Hawaiian or Other Pacific Islander White • INTERPRETER'S STATEMENT • If an interpreter is provided to assist the individual to be sterilized: have translated the information and advice presented orally to the individual to be sterilized by the person obtaining this consent. I have also read him/her the consent form in language and explained its contents to him/her. To the best of my knowledge and belief he/she understood this explanation. Interpreter’s Signature Date • STATEMENT OF PERSON OBTAINING CONSENT • Before ________signed the Name of Individual consent form, I explained to him/her the nature of sterilization operation _______, the fact that it is Specify Type of Operation intended to be a final and irreversible procedure and the discomforts, risks and benefits associated with it. I counseled the individual to be sterilized that alternative methods of birth control are available which are temporary. I explained that sterilization is different because it is permanent. I informed the individual to be sterilized that his/her consent can be withdrawn at any time and that he/she will not lose any health services or any benefits provided by Federal funds. To the best of my knowledge and belief the individual to be sterilized is at least 21 years old and appears mentally competent. He/She knowingly and voluntarily requested to be sterilized and appears to understand the nature and consequences of the procedure. Signature of Person Obtaining Consent Date _Facility _Address • PHYSICIAN'S STATEMENT • Shortly before I performed a sterilization operation upon _ on Name of Individual Date of Sterilization I explained to him/her the nature of the sterilization operation ___, the fact that it is Specify Type of Operation intended to be a final and irreversible procedure and the discomforts, risks and benefits associated with it. I counseled the individual to be sterilized that alternative methods of birth control are available which are temporary. I explained that sterilization is different because it is permanent. I informed the individual to be sterilized that his/her consent can be withdrawn at any time and that he/she will not lose any health services or benefits provided by Federal funds. To the best of my knowledge and belief the individual to be sterilized is at least 21 years old and appears mentally competent. He/She knowingly and voluntarily requested to be sterilized and appeared to understand the nature and consequences of the procedure. (Instructions for use of alternative final paragraph: Use the first paragraph below except in the case of premature delivery or emergency abdominal surgery where the sterilization is performed less than 30 days after the date of the individual's signature on the consent form. In those cases, the second paragraph below must be used. Cross out the paragraph which is not used.) (1) At least 30 days have passed between the date of the individual's signature on this consent form and the date the sterilization was performed. (2) This sterilization was performed less than 30 days but more than 72 hours after the date of the individual's signature on this consent form because of the following circumstances (check applicable box and fill in Premature delivery
Individual's expected date of delivery: _ Emergency abdominal surgery (describe circumstances): Physician’s Signature Date
PAPERWORK REDUCTION ACT STATEMENT
A Federal agency may not conduct or sponsor, and a person is not required to respond to, a collection of information unless it displays the currently valid OMB control number. Public reporting burden for this collection of information will vary; however, we estimate an average of one hour per response, including for reviewing instructions, gathering and maintaining the necessary data, and disclosing the information. Send any comment regarding the burden estimate or any other aspect of this collection of information to the OS Reports Clearance Officer, ASBTF/Budget Room 503 HHH Building, 200 Independence Avenue, SW., Washington, DC 20201.
Respondents should be informed that the collection of information requested on this form is authorized by 42 CFR part 50, subpart B, relating to the sterilization of persons in federally assisted public health programs. The purpose of requesting this information is to ensure that individuals requesting sterilization receive information regarding the risks, benefits and consequences, and to assure the voluntary and informed consent of all persons undergoing sterilization procedures in federally assisted public health programs. Although not required, respondents are requested to supply information on their race and ethnicity. Failure to provide the other information requested on this consent form, and to sign this consent form, may result in an inability to receive sterilization procedures funded through federally assisted public health programs.
All information as to personal facts and circumstances obtained through this form will be held confidential, and not disclosed without the individual's consent, pursuant to any applicable confidentiality regulations. [43 FR 52165, Nov. 8, 1978, as amended at 58 FR 33343, June 17, 1993; 68 FR 12308, Mar. 14, 2003]
HHS-687 (10/12)
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
|
A.
|
OBJECTIVES (42 CFR 433.300)
|
To establish the policies and procedures of the Puerto Rico Health Insurance Administration (ASES for its anachronism in Spanish) to recoup overpayments made to its providers in accordance with 42 CFR 433, Subpart F—Refunding of Federal Share of Medicaid Overpayments to Providers and to implement:
| 1) | Section 1903(d)(2)(A) of the Social Security Act (the “Act”), which directs that quarterly Federal payments to the States under title XIX (Medicaid) of the Act are to be reduced or increased to make adjustment for prior overpayments or underpayments that the Secretary determines have been made. |
| 2) | Section 1903(d)(2) (C) and (D) of the Act, which provides that a State has 1-year from discovery of an overpayment for Medicaid services to recover or attempt to recover the overpayment from the provider before adjustment in the Federal Medicaid payment to the State is made; and that adjustment will be made at the end of the 1-year period, whether or not recovery is made, unless the State is unable to recover from a provider because the overpayment is a debt that has been discharged in bankruptcy or is otherwise uncollectable. |
| 3) | Section 1903(d)(3) of the Act, which provides that the Secretary will consider the pro rata Federal share of the net amount recovered by a State during any quarter to be an overpayment. |
|
B.
|
DEFINITIONS (42 CFR 433.304)
|
| 1) | Abuse (in accordance with 42 CFR 455.2) - provider practices that are inconsistent with sound fiscal, business, or medical practices, and result in an unnecessary cost to the Medicaid program, or in reimbursement for services that are not medically necessary or that fail to meet professionally recognized standards for health care. It also includes beneficiary practices that result in unnecessary cost to the Medicaid program. |
| 2) | Discovery (or discovered) - identification by any ASES, the Federal Government, or the provider of an overpayment, and the communication of that overpayment finding or the initiation of a formal recoupment action without notice as described in 42 CFR 433.316. |
| 3) | Fraud (in accordance with 42 CFR 455.2) - an intentional deception or misrepresentation made by a person with the knowledge that the deception could result in some unauthorized benefit to himself or some other person. It includes any act that constitutes fraud under applicable Federal or State law. |
| 4) | Overpayment - the amount paid by a Medicaid agency to a provider which is in excess of the amount that is allowable for services furnished under section 1902 of the Act and which is required to be refunded under section 1903 of the Act. |
1 of 7
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
| 5) | Provider (in accordance with 42 CFR 400.203) - any individual or entity furnishing Medicaid services under a provider agreement with the Medicaid agency. |
| 6) | Recoupment - any formal action by ASES to initiate recovery of an overpayment without advance official notice by reducing future payments to a provider. |
| 7) | Third party (in accordance with 42 CFR 433.136) - an individual, entity, or program that is or may be liable to pay for all or part of the expenditures for medical assistance furnished under a State plan. |
|
C.
|
APPLICABILITY (42 CFR 433.310)
|
The provisions of these policies and procedures apply to:
| 1) | Overpayments made to providers that are discovered by ASES; |
| 2) | Overpayments made to providers that are initially discovered by the provider and made known to ASES; and |
| 3) | Overpayments that are discovered through Federal reviews. |
|
D.
|
BASIC REQUIREMENTS FOR REFUNDS (42 CFR 433.312)
|
| 1) | ASES has 1-year from the date of discovery of an overpayment to a provider to recover or seek to recover the overpayment before the Federal share must be refunded to CMS. |
| 2) | ASES must refund the Federal share of overpayments at the end of 1-year period following discovery, whether or not ASES has recovered the overpayment from the provider. Notwithstanding, ASES is not required to refund the Federal share of an overpayment made to a provider when ASES is unable to recover the overpayment amount because the provider has been determined bankrupt or out of business in accordance with 42 CFR 433.318. |
| 3) | The date upon which an overpayment occurs is the date upon which ASES, using its normal method of reimbursement for a particular class of provider (e.g., check, interfund transfer), makes the payment involving unallowable costs to a provider. |
|
E.
|
WHEN DISCOVERY OF OVERPAYMENT OCCURS AND ITS SIGNIFICANCE. (42 CFR 433.316)
|
| 1) | The date on which an overpayment is discovered is the beginning date of the 1-year period allowed for a State to recover or seek to recover an overpayment before a refund of the Federal share of an overpayment must be made to CMS. |
2 of 7
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
| 2) | Unless ASES chooses to initiate a formal recoupment action against a provider without first giving written notification of its intent, ASES must notify the provider in writing of any overpayment it discovers in accordance with ASES’ policies and procedures and must take reasonable actions to attempt to recover the overpayment in accordance with State law and procedures. |
| 3) | An overpayment resulting from a situation other than fraud is discovered on the earliest of: |
| a) | The date on which ASES first notifies a provider in writing of an overpayment and specifies a dollar amount that is subject to recovery; |
| b) | The date on which a provider initially acknowledges a specific overpaid amount in writing to ASES; or |
| c) | The date on which ASES initiates a formal action to recoup a specific overpaid amount from a provider without having first notified the provider in writing. |
| 4) | Overpayments resulting from fraud. An overpayment resulting from fraud is discovered on the date of the final written notice (as defined in 42 CFR 433.304) of the overpayment determination that ASES sends to the provider. |
| 5) | If a Federal review at any time indicates that ASES has failed to identify an overpayment or that ASES has identified an overpayment but has failed to either send written notice of the overpayment to the provider that specified a dollar amount subject to recovery or initiate a formal recoupment from the provider without having first notified the provider in writing, CMS will consider the overpayment as discovered on the date that the Federal official first notifies ASES in writing of the overpayment and specifies a dollar amount subject to recovery. |
| 6) | Any adjustment in the amount of an overpayment during the 1-year period following discovery (made in accordance with the approved State plan, Federal law and regulations governing Medicaid, and the appeals resolution process specified in ASES’ administrative policies and procedures) has the following effect on the 1-year recovery period: |
| a) | A downward adjustment in the amount of an overpayment subject to recovery that occurs after discovery does not change the original 1-year recovery period for the outstanding balance. |
| b) | An upward adjustment in the amount of an overpayment subject to recovery that occurs during the 1-year period following discovery does not change the 1-year recovery period for the original overpayment amount. A new 1-year period begins for the incremental amount only, beginning with the date of ASES’ written notification to the provider regarding the upward adjustment. |
3 of 7
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
| 7) | A partial collection of an overpayment amount by ASES from a provider during the 1-year period following discovery does not change the 1-year recovery period for the original overpayment amount due to CMS. |
| 8) | Any appeal rights extended to a provider do not extend the date of discovery. |
|
F.
|
OVERPAYMENTS INVOLVING PROVIDERS WHO ARE BANKRUPT OR OUT OF BUSINESS (42 CFR 433.318)
|
| 1) | ASES is not required to refund the Federal share of an overpayment made to a provider as required by 42 CFR 433.312(a) to the extent that ASES is unable to recover the overpayment because the provider has been determined bankrupt or out of business in accordance with the provisions of this section. ASES must notify the provider that an overpayment exists in any case involving a bankrupt or out-of-business provider and, if the debt has not been determined uncollectable, take reasonable actions to recover the overpayment during the 1-year recovery period in accordance with policies prescribed by applicable State law and administrative procedures. |
| 2) | Overpayments are considered debts that ASES is unable to recover within the 1-year period following discovery if the following criteria are met: |
| a) | The provider has filed for bankruptcy, as specified in paragraph (c) of this section; or |
| b) | The provider has gone out of business and the State is unable to locate the provider and its assets, as specified in paragraph (4) of this section. |
| 3) | ASES is not required to refund to CMS the Federal share of an overpayment at the end of the 1-year period following discovery, if: |
| a) | The provider has filed for bankruptcy in Federal court at the time of discovery of the overpayment or the provider files a bankruptcy petition in Federal court before the end of the 1-year period following discovery; and |
| b) | ASES is on record with the court as a creditor of the petitioner in the amount of the Medicaid overpayment. |
| 4) | ASES is not required to refund to CMS the Federal share of an overpayment at the end of the 1-year period following discovery if the provider is out of business on the date of discovery of the overpayment or if the provider goes out of business before the end of the 1-year period following discovery. A provider is considered to be out of business on the effective date of a determination to that effect under State law. ASES must: |
4 of 7
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
| a) | Document its efforts to locate the party and its assets. These efforts must be consistent with applicable State policies and procedures; and |
| b) | Make available an affidavit or certification from the appropriate State legal authority establishing that the provider is out of business and that the overpayment cannot be collected under State law and procedures and citing the effective date of that determination under State law. |
A provider is not out of business when ownership is transferred within the State unless State law and procedures deem a provider that has transferred ownership to be out of business and preclude collection of the overpayment from the provider.
| 5) | If the 1-year recovery period has expired before an overpayment is found to be uncollectable under the provisions of this section, if ASES recovers an overpayment amount under a court-approved discharge of bankruptcy, or if a bankruptcy petition is denied, ASES must refund the Federal share of the overpayment in accordance with the procedures specified in 42 CFR 433.320. |
|
G.
|
PROCEDURES FOR REFUNDS TO CMS (42 CFR 433.320)
|
| 1) | In accordance with section 1903(d) of the Social Security Act, ASES shall reduce its claims of reimbursement to the extent of any overpayment in the expense reports submitted to CMS, and on the corresponding quarter with respect to the adjustment. |
| 2) | ASES must refund the Federal share of overpayments that are subject to recovery to CMS through credit on the Quarterly Statement of Expenditures (Form CMS–64). Accordingly, the Federal share of overpayments subject to recovery must be credited on the Form CMS–64 report submitted for the quarter in which the 1-year period following discovery, established in accordance with 42 CFR 433.316, ends. |
| 3) | A credit on the Form CMS–64 must be made whether or not the overpayment has been recovered by ASES from the provider. |
| 4) | Effect of reporting collections and submitting reduced expenditure claims. (1) The State is not required to refund the Federal share of an overpayment at the end of the 1-year period if the State has already reported a collection or submitted an expenditure claim reduced by a discrete amount to recover the overpayment prior to the end of the 1-year period following discovery. |
| 5) | If ASES does not refund the Federal share of such overpayment, the State will be liable for interest on the amount equal to the Federal share of the non-recovered, non-refunded overpayment amount. Interest during this period will be at the current Value of Funds Rate (CVFR), and will accrue beginning on the day after the end of the 1-year period following discovery until the last day of the quarter for which the State submits a CMS-64 report refunding the Federal share of overpayment. |
5 of 7
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
| 6) | ASES is not required to report on the Form CMS–64 any collections made on overpayment amounts for which the Federal share has been refunded previously. Furthermore, if ASES has refunded the Federal share of an overpayment and subsequently makes recovery by reducing future provider payments by a discrete amount, ASES need not reflect that reduction in its claim for Federal financial participation. |
| 7) | If the amount of an overpayment is adjusted downward after the agency has credited CMS with the Federal share, ASES may reclaim the amount of the downward adjustment on the Form CMS–64. Under this provision: |
| a) | Downward adjustment to an overpayment amount previously credited to CMS is allowed only if it is properly based on the approved State plan, Federal law and regulations governing Medicaid, and the appeals resolution processes specified in ASES’ administrative policies and procedures. |
| b) | The 2-year filing limit for retroactive claims for Medicaid expenditures does not apply. A downward adjustment is not considered a retroactive claim but rather a reclaiming of costs previously claimed. |
| 8) | If an overpayment has not been determined uncollectable in accordance with the requirements of 42 CFR 433.318 at the end of the 1-year period following discovery of the overpayment, ASES must refund the Federal share of the overpayment to CMS in accordance with the procedures specified above. |
| 9) | If ASES recovers any portion of an overpayment under a court-approved discharge of bankruptcy, ASES must refund to CMS the Federal share of the overpayment amount collected on the next quarterly expenditure report that is due to CMS for the period that includes the date on which the collection occurs. |
| 10) | If a provider's petition for bankruptcy is denied in Federal court, ASES must credit CMS with the Federal share of the overpayment on the later of: |
| a) | The Form CMS–64 submission due to CMS immediately following the date of the decision of the court; or |
| b) | The Form CMS–64 submission for the quarter in which the 1-year period following discovery of the overpayment ends. |
| 11) | If a provider is determined bankrupt or out of business under this section after the 1-year period following discovery of the overpayment ends and ASES has not been able to make complete recovery, ASES may reclaim the amount of the Federal share of any unrecovered overpayment amount previously refunded to CMS. CMS allows the reclaim of a refund if ASES submits to CMS documentation that it has made reasonable efforts to obtain recovery. If ASES reclaims a refund of the Federal share of an overpayment: |
6 of 7
PUERTO RICO HEALTH INSURANCE ADMINISTRATION
POLICIES AND PROCEDURES FOR REFUNDING OF FEDERAL SHARE OF
MEDICAID OVERPAYMENTS TO PROVIDERS
| b) | In bankruptcy cases, ASES must submit to CMS a statement of its efforts to recover the overpayment during the period before the petition for bankruptcy was filed; and |
| c) | In out-of-business cases, ASES must submit to CMS a statement of its efforts to locate the provider and its assets and to recover the overpayment during any period before the provider is found to be out of business in accordance with 42 CFR 433.318. |
| 12) | ASES must report the following information to support each Quarterly Statement of Expenditures Form CMS–64: |
| a) | Amounts of overpayments not collected during the quarter but refunded because of the expiration of the 1-year period following discovery; |
| b) | Upward and downward adjustments to amounts credited in previous quarters; |
| c) | Amounts of overpayments collected under court-approved discharges of bankruptcy; |
| d) | Amounts of previously reported overpayments to providers certified as bankrupt or out of business during the quarter; and |
| e) | Amounts of overpayments previously credited and reclaimed by ASES. |
|
H.
|
MAINTENANCE OF RECORDS (42 CFR 433.322)
|
ASES must maintain a separate record of all overpayment activities for each provider in a manner that satisfies the retention and access requirements of 45 CFR 92.42.
7 of 7
