Attached files
| file | filename |
|---|---|
| EX-4.1 - EXHIBIT 4.1 - CareDx, Inc. | d852478dex41.htm |
| EX-4.2 - EX-4.2 - CareDx, Inc. | d852478dex42.htm |
| EX-10.14 - EX-10.14 - CareDx, Inc. | d852478dex1014.htm |
| EXCEL - IDEA: XBRL DOCUMENT - CareDx, Inc. | Financial_Report.xls |
| EX-31.2 - EX-31.2 - CareDx, Inc. | d852478dex312.htm |
| EX-31.1 - EX-31.1 - CareDx, Inc. | d852478dex311.htm |
| EX-10.17 - EX-10.17 - CareDx, Inc. | d852478dex1017.htm |
| EX-10.19 - EX-10.19 - CareDx, Inc. | d852478dex1019.htm |
| EX-10.16 - EX-10.16 - CareDx, Inc. | d852478dex1016.htm |
| EX-10.15 - EX-10.15 - CareDx, Inc. | d852478dex1015.htm |
| EX-10.18 - EX-10.18 - CareDx, Inc. | d852478dex1018.htm |
| EX-32.1 - EX-32.1 - CareDx, Inc. | d852478dex321.htm |
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
(Mark One)
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE |
| ACT OF 1934 |
For the fiscal year ended December 31, 2014
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE |
| ACT OF 1934 |
For the transition period from to
Commission File Number 001-36536
CAREDX, INC.
(Exact Name of Registrant as Specified in its Charter)
| Delaware | 94-3316839 | |
| (State or Other Jurisdiction of Incorporation or Organization) |
(I.R.S. Employer Identification Number) |
3260 Bayshore Boulevard
Brisbane, California 94005
(Address of Principal Executive Offices, Including Zip Code)
(415) 287-2300
(Registrant’s Telephone Number, Including Area Code)
Securities Registered Pursuant to Section 12(b) of the Act:
| Title of Each Class |
Name of Each Exchange on Which Registered | |
| Common Stock, par value $0.001 per share | The NASDAQ Global Stock Market LLC |
Securities Registered Pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. x
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ¨ | Accelerated filer | ¨ | |||
| Non-accelerated filer | x (Do not check if a smaller reporting company) | Smaller reporting company | ¨ | |||
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The registrant’s common stock was not publicly traded on The NASDAQ Global Market as of June 30, 2014, the last business day of the registrant’s most recently completed second fiscal quarter. The registrant’s common stock began trading on The NASDAQ Global Market on July 16, 2014.
The aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant, based on the closing price of a share of the registrant’s common stock on December 31, 2014 as reported by The NASDAQ Global Market on such date was approximately $49,411,679. The registrant has elected to use December 31, 2014, the last day of the fiscal year covered by this report, as the calculation date because on June 30, 2014 (the last business day of the registrant’s mostly recently completed second fiscal quarter), the registrant was a privately-held company. Shares of the registrant’s common stock held by each executive officer, director and holder of 10% or more of the outstanding common stock have been excluded in that such persons may be deemed to be affiliates. This calculation does not reflect a determination that certain persons are affiliates of the registrant for any other purpose.
The number of shares of the registrant’s Common Stock outstanding as of February 27, 2015 was 11,814,825.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s Proxy Statement relating to the 2015 Annual Meeting of Stockholders to be held on June 16, 2015, are incorporated by reference into Part III of this Annual Report on Form 10-K where indicated. Such Proxy Statement will be filed with the Securities and Exchange Commission within 120 days after the end of the registrant’s fiscal year ended December 31, 2014.
Table of Contents
| Item No. |
Page No. |
|||
| PART I |
5 | |||
| 5 | ||||
| 19 | ||||
| 52 | ||||
| 52 | ||||
| 52 | ||||
| 52 | ||||
| PART II |
53 | |||
| 53 | ||||
| 56 | ||||
| Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations |
57 | |||
| Item 7A. Quantitative and Qualitative Disclosures About Market Risk |
72 | |||
| 73 | ||||
| Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
106 | |||
| 106 | ||||
| 107 | ||||
| PART III |
108 | |||
| Item 10. Directors, Executive Officers and Corporate Governance |
108 | |||
| 108 | ||||
| 108 | ||||
| Item 13. Certain Relationships and Related Transactions, and Director Independence |
108 | |||
| 108 | ||||
| PART IV |
109 | |||
| 109 | ||||
| 110 | ||||
| 111 | ||||
2
Table of Contents
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. All statements contained in this Annual Report on Form 10-K other than statements of historical fact, including statements regarding our future results of operations and financial position, our business strategy and plans, and our objectives for future operations, are forward-looking statements. The words “believe,” “may,” “will,” “potentially,” “estimate,” “continue,” “anticipate,” “intend,” “could,” “would,” “project,” “plan,” “expect” and the negative and plural forms of these words and similar expressions are intended to identify forward-looking statements.
These forward-looking statements may include, but are not limited to, statements concerning the following:
| • | our ability to generate revenue from sales of AlloMap and future solutions, if any, and our ability to increase the commercial success of AlloMap; |
| • | our plans and ability to develop and commercialize new solutions, including cell-free DNA solutions for the surveillance of heart and kidney transplant recipients; |
| • | our ability to obtain, maintain and expand reimbursement coverage for payers for AlloMap and future solutions, if any; |
| • | the outcome or success of our clinical trial collaborations and observational studies; |
| • | our dependence on certain of our suppliers and service providers |
| • | our compliance with federal, state and foreign regulatory requirements; |
| • | the favorable review of AlloMap and our future solutions, if any, in peer-reviewed publications; |
| • | our ability to protect and enforce our intellectual property rights, our strategies regarding filing additional patent applications to strengthen our intellectual property rights, and our ability to defend against intellectual property claims that may be brought against us; |
| • | our anticipated cash needs and our anticipated uses of our funds, including our estimates regarding operating expenses and capital requirements; |
| • | anticipated trends and challenges in our business and the markets in which we operate; |
| • | disruptions to our business, including disruptions at our laboratories and manufacturing facilities; |
| • | our ability to retain key members of our management; |
| • | our ability to make successful acquisitions or investments and to manage the integration of such acquisitions or investments; |
| • | our ability to expand internationally; and |
| • | our ability to comply with the requirements of being a public company. |
These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including those described in the section entitled “Risk Factors” included in Part I, Item 1A and elsewhere in this Annual Report on Form 10-K. Moreover, we operate in a very competitive and rapidly changing environment, and new risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the forward-looking events and circumstances discussed in this report may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements.
3
Table of Contents
You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward-looking statements will be achieved or occur. Moreover, neither we nor any other person assumes responsibility for the accuracy and completeness of the forward-looking statements. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this report to conform these statements to actual results or to changes in our expectations.
You should read this Annual Report on Form 10-K and the documents that we reference in this Annual Report on Form 10-K and have filed with the SEC as exhibits to this Annual Report on Form 10-K with the understanding that our actual future results, levels of activity, performance and events and circumstances may be materially different from what we expect. We qualify all forward-looking statements by these cautionary statements.
4
Table of Contents
PART I
| ITEM 1. | BUSINESS |
Company Overview
We are a commercial stage company that develops, markets and delivers a diagnostic surveillance solution for heart transplant recipients to help clinicians make personalized treatment decisions throughout the organ transplant patient’s lifetime. Our first commercialized testing solution, the AlloMap heart transplant molecular test, or AlloMap, is a blood-based test used to monitor heart transplant recipients for acute cellular rejection. Since 2008, we have sought to expand the adoption and utilization of our AlloMap solution through ongoing studies to substantiate the clinical utility and actionability of AlloMap, secure positive reimbursement decisions for AlloMap from large private and public payers, develop and enhance our relationships with key members of the transplant community, including opinion leaders at major transplant centers, and explore opportunities and technologies for the development of additional solutions for post-transplant surveillance. We believe the use of AlloMap, in conjunction with other clinical indicators, can help healthcare providers and their patients better manage long-term care following a heart transplant. In particular, we believe AlloMap can improve patient care by helping healthcare providers to avoid the use of unnecessary, invasive surveillance biopsies and to determine the appropriate dosage levels of immunosuppressants.
AlloMap is the only non-invasive method recommended in the International Society for Heart and Lung Transplantation (ISHLT) patient care guidelines for surveillance of heart transplant rejection in patients 15 years of age or older. AlloMap has received 510(k) clearance from the U.S. Food and Drug Administration (FDA), for marketing and sale as a test to aid in the identification of recipients with a low probability of moderate or severe acute cellular rejection. A 510(k) submission is a premarketing submission made to the FDA. Clearance may be granted by the FDA if it finds the device or test provides satisfactory evidence pertaining to the claimed intended uses and indications for the device or test. Additionally, we have obtained a CE mark, which indicates a product’s compliance with European Union (EU) legislation and enables the sale of such product within the EU. We have a certificate of accreditation under CLIA (Clinical Laboratory Improvement Amendments of 1988) to perform “high complexity” testing. Almost all clinical laboratories are subject to regulation under CLIA, which is designed to ensure that laboratory testing services on materials derived from the human body are accurate and reliable.
From our launch in 2005 through 2014, we have performed more than 66,000 commercial AlloMap tests, including more than 11,000 tests in 2014, in our Brisbane, California laboratory. In 2014, AlloMap was used in 110 of the approximately 129 heart transplant centers in the United States. As of December 31, 2014, significantly all of our testing revenue has come from the United States and all of our assets and operations are located in the United States, however in 2014 we began to expand our AlloMap offering through partnerships in Europe and Canada.
AlloMap has received positive coverage decisions for reimbursement from Medicare and many of the largest private payers, including Aetna, Cigna, Humana, Inc., Kaiser Foundation Health Plan, Inc. and WellPoint. We believe our success in achieving reimbursement confirms the value proposition of AlloMap to our key constituents. As of December 31, 2014, we had been reimbursed for approximately 79% of AlloMap results delivered in the twelve months ended June 30, 2014.
We have also successfully completed a number of landmark clinical trials in the transplant field demonstrating the clinical utility of AlloMap for surveillance of heart transplant recipients. We initially established the analytical and clinical validity of AlloMap on the basis of our Cardiac Transplanted Organ Rejection Gene expression Observational (Crespo-Leiro M et al., AM. J. Transplantation, 2012), or CARGO, study, which was published in the American Journal of Transplantation. A subsequent trial, Invasive Monitoring Attenuation through Gene Expression (Pham MX et al., N. Eng. J. Med., 2010), or IMAGE, published in The New England Journal of Medicine, demonstrated that clinical outcomes in recipients managed with AlloMap surveillance were
5
Table of Contents
equivalent to outcomes in recipients managed with biopsies. The results of our clinical trials have also been presented at major medical society congresses and published in peer-reviewed publications in leading medical journals.
We are also engaged in efforts to develop additional testing solutions in the heart transplant market and new testing solutions in other organ transplant markets. For instance, we are seeking to apply next generation sequencing platforms to detect and quantitate genetic differences between cell-free DNA(cfDNA) in the blood stream emanating from the donor heart and cfDNA emanating from the transplant recipient. We believe this solution may help determine rejection-specific activity manifested as cell damage in the transplanted heart.
We are organized and operate in a single segment. See “Management’s Discussion and Analysis of Financial Conditions and Results of Operations” included in Part II, Item 7 of this Annual Report on Form 10-K.
Our History
We were originally incorporated in Delaware in December 1998 under the name Hippocratic Engineering, Inc. In April 1999, we changed our name to BioCardia, Inc., in June 2002, we changed our name to Expression Diagnostics, Inc., in July 2007, we changed our name to XDx, Inc., and in March 2014, we changed our name to CareDx, Inc. Our principal executive offices are located at 3260 Bayshore Boulevard, Brisbane, California and our telephone number is (415) 287-2300.
Limitations of Existing Approaches for Surveillance of Transplant Recipients
The care of organ transplant recipients is an intense and costly effort and requires life-long surveillance and management by highly specialized clinicians and other healthcare providers. For example, heart transplant recipients often incur lifetime costs of more than $1.9 million and kidney transplant recipients often incur lifetime costs of more than $1.1 million. The historical standard for heart transplant surveillance has been the microscopic examination of heart tissue obtained through an invasive endomyocardial biopsy. In the biopsy procedure, a catheter is inserted into the right internal jugular vein via the recipient’s neck and threaded through blood vessels into the inner chamber of the heart. Four pieces of tissue are cut from the wall of the heart and sent to a laboratory for examination by a pathologist who uses a microscope to look for evidence of cellular rejection. Due to the limitations of biopsies, including pathologist evaluations which are subjective and dependent upon visual assessment and qualitative interpretation, the risk of sampling errors and the potential for complications and other health risks, these procedures are not frequently used by clinicians to tailor the use of immunosuppressants. The typical schedule of biopsy surveillance may involve eight to ten biopsies within the first six months after transplant and a total of ten to fifteen biopsies within the first year post-transplant. Because repeated biopsies incur cumulative risk and trauma to the recipient, the frequency of biopsy surveillance after one year has been low, despite the fact that recipients would benefit from continued monitoring for rejection and management of their immunosuppressive drugs for the rest of their lives. With less biopsy data collected after the first year post-transplant, clinicians have less information upon which to tailor immunosuppression treatment for their recipients.
The use of renal biopsies for surveillance of kidney transplants is also limited due to the risks associated with such biopsies. Therefore the main clinical test indicator of transplanted kidney dysfunction is an increase in serum creatinine levels, which though widely used, literature suggests may be nonspecific and only detected late, after significant and irreversible renal function loss has occurred. As the current solutions for the surveillance of organ transplant recipients provide only limited and infrequent information on the presence or absence of rejection, clinicians tend to administer relatively high levels of immunosuppression therapy to control rejection risk, which may be more than required for an individual recipient. Due in part to this long-term high level of immunosuppression therapy, illness and mortality rates among transplant recipients remain well above those of the general population.
6
Table of Contents
Immunosuppression of Heart and Kidney Transplant Recipients
The risk of rejection in heart and kidney transplant recipients is managed primarily through the use of immunosuppressive drugs. Surveillance biopsies are infrequent after the first year because of invasive procedural risks, discomfort, inconvenience, expense and the low rate of finding moderate to severe grade rejection. As a result, clinicians have limited and infrequent information about an individual recipient’s risk of rejection over the months and years following transplant. In the average recipient, the immune system gradually adapts to the organ graft, and the need for immunosuppression declines over time. However, there is meaningful variation in the level of rejection activity and need for immunosuppression among transplant recipients. Limited insight into the risk profile of the individual recipient often causes clinicians to apply a ‘one-size-fits’ all approach to immunosuppression to help protect against the severe consequences of rejection. Although typical doses of immunosuppressants result in a low rate of rejection in the transplant population as a whole, many individuals receive more immunosuppressants than they may actually need. Improved post-transplantation diagnostics are necessary to make further gains in the long-term care and health outcomes of heart, kidney and other organ transplant recipients.
The Need for a Better Surveillance Solution
More effective solutions for the surveillance and risk assessment of recipients would improve the clinician’s ability to individualize immunosuppression therapy and to reduce the use of invasive biopsies. We believe that core elements of effective surveillance solutions include:
| • | Highly accurate and quantitative results; |
| • | Non-invasive, without creating risks to the recipient; |
| • | Easy to administer; |
| • | Differentiate rejection from quiescence; |
| • | Detect rejection earlier; and |
| • | Timing and frequency of results that allow informed and effective treatment decisions. |
Our Products and Services
We develop and provide a diagnostic surveillance testing solution for heart transplant recipients. Our initial test, AlloMap, is designed to help clinicians to regularly monitor for heart transplant rejection throughout the life of the recipient, modulate the use of immunosuppression and make more personalized treatment decisions. The AlloMap test uses a sample of the patient’s blood. Blood draws are relatively painless and the process is routinely performed in laboratories around the world. AlloMap may be used instead of a surveillance heart biopsy to rule out acute cellular rejection in heart transplant recipients. AlloMap offers rapid, high quality results, and for approximately 91% of patients, we return AlloMap results to the clinician within three business days after the blood draw.
Overview
AlloMap uses gene expression technology to aid in the identification of heart transplant recipients at low risk of rejection. The test measures the molecular signatures that correlate with biological activity associated with moderate to severe acute cellular rejection. Gene expression may indicate acute cellular rejection well before the evidence of damage is visible from a tissue biopsy sample. AlloMap applies a proprietary mathematical algorithm comprised of the expression values, or RNA levels, of 20 genes of which 11 are informative and 9 are for quality control, and then yields a single AlloMap score. AlloMap may be used for heart transplant recipients 15 years of age or older 55 days post transplant.
AlloMap provides a single integer score ranging from 0 to 40 and determines the probability of moderate to severe acute cellular rejection. A key benefit of the AlloMap score is its negative predictive value, (NPV). The
7
Table of Contents
NPV of AlloMap is the likelihood that a heart transplant recipient is at low risk for rejection. The NPV for recipients with an AlloMap score below the threshold range for one or more years post-transplant can be greater than 99% depending on the actual score.
The utility of AlloMap is well established. AlloMap is the first and only non-invasive method recommended in the ISHLT patient care guidelines for surveillance of heart transplant recipients for rejection in non-infants. AlloMap has obtained 510(k) clearance from the FDA as an In Vitro Diagnostic Multivariate Index Assay (IVDMIA). In addition, the clinical utility of AlloMap is supported by numerous clinical trials sponsored by us, the results of which have been published in leading peer-reviewed medical journals.
Through December 31, 2014, we have performed more than 66,000 commercial AlloMap tests in total. We estimate that there are approximately 129 centers performing heart transplants in the United States. In 2014, AlloMap was used in 110 of these centers.
In incorporating AlloMap into their practice, clinicians may consider recipient history, a physical exam, graft function and the results of AlloMap at each post-transplant clinic visit. If the recipient’s AlloMap score is below an applicable threshold, in the absence of other clinical indicators of rejection, clinicians may elect not to conduct a surveillance biopsy at that time. Where there are signs or indications of rejection, evidence of failure or impaired function or an AlloMap score greater than the applicable threshold, a biopsy may be ordered.
Clinical Trials of AlloMap
The clinical validation and utility of AlloMap is supported by a number of major clinical trials involving more than 2,000 heart transplant recipients and published in leading peer-reviewed medical journals. Our trials have been designed to evaluate the clinical utility of our solutions and are an integral part of our business strategy, clinical development and marketing programs. In heart transplantation, two major observational trials, CARGO and CARGO II, enabled the initial development, validation and further validation of AlloMap to detect and monitor acute cellular rejection in heart transplant recipients. Blood samples and clinical data from these two trials have been preserved in a multi-year, multi-center registry which we are sponsoring. We expect these samples and data to enable further discovery and product development of new indicators of rejection activity, or biomarkers, and new diagnostic solutions. We believe these repositories, which contain over 37,000 samples, are rich sources for further new product research and development because individual recipients were followed for 10 serial visits over one year or more, on average, and in many cases associated biopsy rejection grades and other clinical outcome endpoints are available for analysis, correlative studies and validation efforts that we believe will be useful for new product development.
Additional trials, including IMAGE and EIMAGE, have demonstrated that clinical outcomes in recipients managed with AlloMap surveillance were equivalent to outcomes in recipients managed with biopsies. We have also completed two studies analyzing data from earlier trials to observe how the variability in AlloMap scores over time may be useful in predicting the risk of rejection and graft dysfunction.
Research and Development
Our research and development activities focus on developing cutting edge organ transplant surveillance solutions, and we seek to continuously explore and develop new clinically-relevant approaches to our products. Our ongoing research and development efforts include:
| • | further refinement of the AlloMap product line; |
| • | undertaking additional studies to expand the clinical utility of AlloMap and generate additional data to enhance clinical understanding of transplant rejection; |
| • | new product development in other areas of transplant surveillance, such as the use of cfDNA technology as a biomarker for rejection; and |
| • | technology platform development to increase efficiency and lower costs in our testing and laboratory operations. |
8
Table of Contents
Our research and development efforts are not limited to specific technology platforms, biomarkers or methodologies. Instead, we aim to leverage current and future innovations in biomarker identification and measurement in developing future solutions.
Our research and development expenses for the years ended December 21, 2014, 2013 and 2012 were $3.8 million, $3.2 million, and $4.8 million, respectively.
cfDNA as a Biomarker for Organ Rejection
We are currently engaged in discovery and development efforts using cfDNA to develop additional post-transplant diagnostic solutions, with a focus on a test for heart rejection followed by a test for kidney rejection. We believe donor derived cfDNA may be useful as a biomarker for the detection of rejection related organ damage in solid organ transplant recipients. cfDNA are short fragments of DNA that are released into the blood stream when cells die. cfDNA assays have transformed pre-natal testing by providing a non-invasive, accurate method to detect genetic abnormalities in a fetus, without needing an invasive amniocentesis procedure. In a transplant recipient, we believe the differences in the relative amounts of cfDNA from the donated organ and the recipient can be used to distinguish between a healthy or damaged donor organ.
Initial studies such as Heart Transplants Are Genome Transplants: Universal Noninvasive Detection of Organ Transplant Rejection (Snydev T M et al., Proceedings N. Academy Sciences, 2011) and the Highly Sensitive Non-Invasive Cardiac Transplant Rejection Monitoring Using Targeted Qualification of Donor Specific Cell Free DNA (Hidestrand M et. al., J. Am. Coll. Cardiology, 2013) indicate that cfDNA may be a universally applicable marker for rejection, not only for heart, but for kidney, liver and lung as well. Our initial studies and other outside studies have reported that the proportions of donor derived cfDNA in heart transplant recipients increase as much as five-fold during rejection episodes. Measuring the level and changes in the relative amount of donor derived cfDNA in the blood stream may be a useful new method for detecting rejection. This technique involves measuring the cfDNA released by dying cells from the donor organ into the recipient’s blood stream. The level of donor specific cfDNA from the transplanted organ can be monitored in the recipient’s blood stream over time, and changes in organ status may be detected as changes in the donor cfDNA level. The rationale for this approach arises from the observation that both acute and chronic rejection processes are associated with high levels of cell death in the transplanted organ.
cfDNA for Heart Transplants
We are pursuing development of a cfDNA-based solution for heart transplant recipients. We believe that a cfDNA-based solution for heart transplant recipients could provide additional value to clinicians, particularly in situations where a recipient’s AlloMap score does not suggest a low probability of rejection activity. Studies have reported that a higher percentage of donor derived cfDNA in the blood stream indicated the likelihood of rejection in over 90% of the cases where moderate or severe rejection was found in an associated biopsy specimen. We believe a cfDNA solution for heart would help enable clinicians to identify recipients with a higher probability of rejection and make any subsequent biopsy a more effective diagnostic tool, because the likelihood of detecting rejection in the biopsy specimen would be substantially enhanced.
We have completed internal studies to define methods to be used to test our collection of samples as well as additional samples to be acquired by us. We have established our proprietary strategy for quantification of donor specific cfDNA and we have completed initial proof of concept studies. We have further developed a research use only version of our cfDNA solution now in use with a limited number of heart transplant centers and physicians under research agreements as part of the D-OAR study.
Other steps in our development process for a cfDNA solution in heart include publication of abstracts on the results of the clinical performance of our cfDNA solution for heart based on our CARGO II sample and data repository.
9
Table of Contents
cfDNA for Kidney Transplants
We intend to apply the expertise we gain in developing our heart transplant cfDNA solution to develop a cfDNA solutions for other organ transplants, beginning with kidney transplants. We have a proprietary library of longitudinal blood and urine samples from kidney transplant recipients acquired during the course of our Kidney Transplanted Organ Rejection Gene expression Observational Study, or KARGO. In addition to reviewing our KARGO sample repository for aspects of our cfDNA biomarker research, we are also commencing an independent initiative to collect additional samples.
We may seek to acquire rights to access additional well-curated samples from other university hospitals and other sample repository consortiums in the United States with which we maintain relationships. If we are successful in developing a cfDNA-based kidney solution, we plan to move this solution into a lab compliant with the Clinical Laboratory Improvement Amendments of 1988, or CLIA, to complete the validation required to commercialize a solution for use in kidney transplant recipients. If developed, we expect to commercialize this solution as a Laboratory Developed Test, or LDT, under CLIA.
We previously applied for and obtained FDA clearance for our AlloMap solution based on draft guidance published by the FDA in September 2006. That guidance was not finalized by the FDA and, at present, we do not anticipate seeking 510(k) clearance from the FDA for our cfDNA-based kidney solution as part of our initial launch. If the FDA changes its current policy with respect to the regulation of LDTs, we may be required to seek FDA clearance or premarket approval for our cfDNA-based kidney solution. The time required to develop and validate a test for kidney transplants depends on a number of factors, including the success and timing of developing a cfDNA test for heart transplants and the time required to acquire sufficient samples.
We plan to undertake a study regarding cfDNA in Acute Rejection in Transplantation (DART), which is intended to establish the clinical validity of a cfDNA program for kidney transplant patients. We expect the DART initiative to run for a minimum of 18 months, with the goal of collecting samples from 200 patients in 10 or more centers. Once we receive relevant information from an interim analysis, we anticipate initiating a second phase for the study which will establish clinical utility of our cfDNA kidney solution.
Reimbursement
We have been successful in achieving reimbursement from many payers. The reimbursement process can take six months or more to complete depending on the payer. As of December 31, 2014, we have been reimbursed for approximately 79% of AlloMap results delivered in the twelve months ended June 30, 2014.
Reimbursement for AlloMap comes primarily from Medicare, private third party payers such as insurance companies and managed care organizations, Medicaid and hospitals. A number of payers have adopted coverage policies approving AlloMap for reimbursement. Such policies often approve reimbursement for tests performed from six-months or one year post-transplant through five years post-transplant. For tests performed outside the scope of the payer’s policy, and for tests performed where the payer has not adopted a coverage policy, we pursue reimbursement on a case-by-case basis. If a reimbursement claim is denied, we generally pursue the appeals process for the particular payer.
AlloMap has been billed since the inception of the test using an unlisted CPT code. This approach is consistent with the billing approach for many diagnostic tests. The American Medical Association also recently included AlloMap in its recommendations for a test-specific, Category 1 CPT code. We are awaiting Medicare’s decisions
10
Table of Contents
on pricing, official codes and methodologies to be implemented, which we expect to be announced in the second half of 2015.
Medicare
We are reimbursed for a substantial portion of our tests performed on recipients covered by Medicare. These represented 37%, 39% and 40% of all AlloMap tests in 2014, 2013 and 2012, respectively. Approximately 51%, 53% and 52% of all testing revenue was derived from Medicare reimbursements for the years ended December 31, 2014, 2013 and 2012, respectively. Medicare reimbursement for AlloMap began in 2006 and has continued through three successive Medicare Administrative Contractors, which are the local organizations that make most coverage decisions for Medicare.
Private Payers and Medicaid Payers
We are reimbursed for a substantial portion of the tests we perform on patients covered by private payers and Medicaid payers. Coverage policies approving AlloMap for reimbursement have been adopted by many of the largest private payers, including Aetna, Cigna, Humana, Inc., Kaiser Foundation Health Plan, Inc., WellPoint, and a number of state Medicaid programs. Many of the payers with positive coverage policies have also entered into contracts with us to formalize pricing and payment terms. With private payers and Medicaid payers that have not yet adopted positive coverage policies, we obtain reimbursement from those payers on a case-by-case basis for a significant portion of claims.
Europe and Canada
Our Canadian partner, Lifelabs Medical Laboratory Services, pays us directly for the tests we perform for them and is responsible for obtaining reimbursement from payers in their territory. In Europe, we receive revenue in two ways. First, through our sale of testing materials to our partner, Diaxonhit, and second, through royalties on Diaxonhit’s net sales of AlloMap in Europe.
Testing and Laboratory Operations
Our laboratory operations are headquartered at our Brisbane, California laboratory, which is certified under the Clinical Laboratory Improvement Amendment of 1988 (CLIA) and where we perform all testing. Through our commercial partner, we have secured a dedicated laboratory for AlloMap testing in Europe and our partner is taking steps to make the laboratory operational. We believe that our laboratory capacity will be adequate to meet demand for AlloMap for the next several years. We intend to expand our laboratory facility as we move into other areas of organ transplant surveillance.
When AlloMap is ordered by a clinician, a blood sample is drawn and processed to isolate the white blood cells, which are subsequently broken down, frozen and sent via overnight courier to our laboratory. Each of the 20 genes comprising AlloMap are tested in triplicate and results are reported to the ordering clinician by fax within three business days of receipt of the sample. Rigorous quality control testing is conducted at every phase of the test process. Test samples that fail to meet quality control criteria are immediately re-tested and the ordering clinician is notified of the need to re-test if turnaround time will be affected.
We rely solely on single suppliers to provide certain laboratory instruments and reagents that we use to perform AlloMap. These sole source suppliers include Thermo Fisher Scientific Inc., which supplies us with instruments, laboratory reagents and consumables, Becton, Dickinson, and Company which supplies us with cell preparation tubes, and Therapak Corporation, which supplies us with a proprietary buffer reagent. One of the reagents supplied to us by Therapak Corporation is, in turn, obtained by Therapak Corporation from Qiagen N.V. and is a proprietary formulation of Qiagen N.V.
11
Table of Contents
A critical test component needed to perform AlloMap is a universal master mix that is supplied by Thermo Fisher Scientific Inc., who has discontinued production of the formulation originally used. We have worked with Thermo Fisher Scientific Inc. to develop and validate an equivalent performing alternative universal master mix. We have successfully completed our validation and equivalence testing of three lots of this material, which will allow us to purchase the replacement universal master mix on a routine basis from Thermo Fisher Scientific Inc. At present, we have sufficient master mix material in house to continue delivering AlloMap through December 2015 and the ability to purchase additional material as needed to support AlloMap testing without restriction.
Sales and Marketing
Our sales organization consists of a direct sales team in the United States which interacts with all aspects of the transplantation channel, including sales, medical science, reimbursement, customer service and field laboratory/draw site support. As of December 31, 2014, our sales and marketing team consisted of 22 employees, including transplant account sales executives, reimbursement account managers, medical science liaisons and patient service center and customer service personnel. All personnel are field based except for customer service, which are based in our California headquarters.
In 2014, AlloMap was used in 110 of the approximately 129 heart transplant centers in the United States. Our marketing focuses on the clinical and economic benefits of AlloMap and the scientific validation that supports our test. Our strategy includes continued marketing to and education of clinicians and administrators at treatment centers that have used our test to increase the number of clinicians at those centers using our test, and to have centers adopt formal protocols for AlloMap use.
Internationally, we have commercial agreements in Europe and Canada that provide for exclusive rights to promote AlloMap in those territories. In Europe, Diaxonhit SA (Diaxonhit) is our commercial partner. Diaxonhit is a French, publicly traded specialty diagnostics company with activities in France, Switzerland and Belgium. Diaxonhit has agreed to commercialize AlloMap in all countries in western and central Europe directly and through sub-partners. Under the terms of our agreement, we will provide Diaxonhit with training and a license to perform AlloMap and Diaxonhit, through a third party laboratory, has agreed to perform AlloMap in Europe to facilitate the turnaround time and cost effectiveness of the test process. Diaxonhit will pay royalties to us on the net sales, as defined in the agreement, of AlloMap tests, in the mid to high teens. Diaxonhit made an upfront payment to us in cash of approximately €387,500 ($503,000) and Diaxonhit’s publicly traded common stock with a value at the time of €387,000 following execution of the agreement. The cash portion of this upfront payment will offset the royalties payable to us upon the satisfaction of certain milestones in the first three years following the first commercial sale. Diaxonhit is also obligated to pay additional royalties based on certain milestones, up to a maximum of €1,450,000, and some of the royalty payments may be made pursuant to the issuance to us of Diaxonhit’s publicly traded common stock. The first commercial sale in Europe through Diaxonhit occurred in June 2014.
In the province of Ontario, Canada, LifeLabs Medical Laboratories Services (LifeLabs), the largest Canadian reference laboratory, is our commercial partner. Under this arrangement, LifeLabs sends blood samples to our laboratory in Brisbane, California for testing. LifeLabs will pay us on a per test basis. They also made an upfront payment to us following execution of the agreement that is available to offset test fees up to the amount of the upfront payment in the first year. Under the terms of our agreement, we will provide LifeLabs with training and marketing materials. LifeLabs has an option to expand its rights to commercialize AlloMap in all other Canadian provinces. We first began performing tests under our arrangement with LifeLabs in the fourth quarter of 2013. We recognized minimal revenue from this agreement in 2014.
12
Table of Contents
Competition
We believe the principal competitive factors in our target markets include:
| • | quality and strength of clinical and analytical validation data; |
| • | confidence in diagnostic results; |
| • | the extent of reimbursement; |
| • | inclusion in practice guidelines; |
| • | cost-effectiveness; and |
| • | ease of use. |
We believe we compete favorably on the factors described above.
Existing diagnostic tests for heart transplant rejection generally involve evaluating biopsy samples to determine the presence or absence of rejection, and for kidney transplant rejection include general, non-specific clinical chemistry tests, though biopsies are also a surveillance diagnostic tool. Both of these practices have been the standard of care in the United States for many years, and we will need to continue to educate clinicians, transplant recipients and payers about the various benefits of our test in order to change clinical practice. Also, many transplant centers are located within hospitals that have their own laboratory facilities and have capacity to conduct various tests, so hospitals may choose to rely on internally developed and/or internally performed surveillance and diagnostic tests. Competition for kidney surveillance diagnostics can also come from biopsies. However, because of the risks and discomforts of the invasive kidney biopsy procedure, as well as the expense and relatively low rate of finding moderate to severe grade rejection, biopsy is not a standard practice for surveillance of transplanted kidneys. Additional competition for kidney surveillance diagnostics currently comes from general, non-specific clinical chemistry tests such as serum creatinine, urine protein, complete blood count, lipid profile and others that are widely ordered by physician offices and routinely performed in clinical reference labs and hospital labs.
We also expect the competition for post-transplant surveillance to increase as there are numerous established and early-stage companies in the process of developing novel products and services for the transplant market which may directly or indirectly compete with AlloMap or our development pipeline. In addition to companies focused on pre-transplantation such as Thermo Fisher Scientific Inc.’s One Lambda and Immucor, Inc.’s LIFECODES businesses, companies who have not historically focused on transplantation, but have knowledge of cfDNA technology, have indicated they are considering this market.
Our potential competitors may have widespread brand recognition and substantially greater financial, technical and research and development resources and selling and marketing capabilities than we do. Others may develop products with prices lower than ours that could be viewed by clinicians and payers as functionally equivalent to our solution, or offer solutions at prices designed to promote market penetration, which could force us to lower the price of our current and future solutions and affect our ability to achieve or maintain profitability.
Intellectual Property
Patents and Proprietary Technology
In order to remain competitive, we seek to develop and maintain protection on the proprietary aspects of our technologies. We rely on a combination of patents, copyrights, trademarks, material data transfer agreements and licenses to protect our intellectual property rights. We also rely upon unpatented trade secrets and improvements, unpatented know-how and continuing technological innovation to develop and maintain our competitive position. We generally protect this information with confidentiality and reasonable security measures.
Our core patent position for AlloMap is based on issued patents and patent applications disclosing identification of genes differentially expressed between activated and resting leukocytes and demonstration of correlation between gene expression patterns and specific clinical states and outcomes. Our strategy is to continue to
13
Table of Contents
broaden our intellectual property estate for AlloMap through the discovery and protection of gene expression patterns and their correlation with specific clinical states and outcomes, as well as the algorithms needed for clinical assessment.
As of December 31, 2014, we have 16 issued United States patents, one pending United States patent application, and three pending patent applications outside the United States related to transplant rejection and autoimmunity. We have five issued United States patents covering methods of diagnosing transplant rejection using 9 of the 11 informative genes measured in AlloMap. The expiration dates of these patents range from 2021 to 2024. We have six issued United States patents covering a method of diagnosing or monitoring an autoimmune or chronic inflammatory diseases, such as lupus, by detecting specific genes. While we have clinical samples and patents covering lupus diagnostics, we do not intend to actively pursue the lupus test opportunity.
In the area of cell-free DNA-based transplant diagnostics, we have filed a patent application to cover our research and development work in this field. In connection with our acquisition of ImmuMetrix, Inc., we have an exclusive license from Stanford University to an issued US patent relating to the diagnosis of rejection in organ transplant recipients using cfDNA.
AlloMap, XDx and CareDx are registered trademarks of our Company in the United States.
We have developed trade secrets and know-how since our inception. These are found particularly in technical areas such as optimized systems for making precise and reproducible quantitative PCR measurements, and in the analysis of genomic data and algorithm development.
License Agreements
We currently rely on license agreements to obtain rights under certain patents that we believe may be necessary to make, use and sell our AlloMap test and future solutions. We may in the future rely, at least in part, upon licensing agreements with third parties to obtain patent rights and transfers of technology, information and know-how that enable us to further our development of additional solutions for post-transplant surveillance.
In November 2004, we entered into a license agreement with Roche Molecular Systems, Inc. (Roche), which was amended in January 2007, July 2007, October 2008 and September 2014. The agreement grants us the right to use PCR and quantitative real time PCR for use in clinical laboratory services. This is a non-exclusive license agreement in the United States covering the claims in multiple Roche patents. Under the terms of the agreement, we are required to report and pay royalties, after adjustment due to a discount for combination services, in the mid-single digits on test revenues from products using the licensed intellectual property on a quarterly basis.
In June 2014, we entered into an amended and restated license agreement with Stanford University (Stanford) which granted us an exclusive license to a patent relating to the diagnosis of rejection in organ transplant recipients using cfDNA and a non-exclusive license to related technology provided by Stanford. Subject to various rights of extension, we are required to achieve certain development and commercialization milestones set forth in the license agreement. Under the terms of the license agreement, we are required to report and pay an annual license maintenance fee, six milestone payments and royalties in the low single digits on net sales of products incorporating the licensed technology.
Regulation
Clinical Laboratory Improvement Amendments of 1988
As a clinical laboratory, we are required to hold certain federal, state and local licenses, certifications and permits to conduct our business. Under the Clinical Laboratory Improvement Amendments of 1988, or CLIA, administered by the Centers for Medicare & Medicaid Services, or CMS, we are required to hold a certificate
14
Table of Contents
applicable to the type of work we perform and to comply with standards covering personnel, facilities administration, quality systems, proficiency testing and performance. Almost all clinical laboratories are subject to regulation under CLIA, which is designed to ensure that laboratory testing services on materials derived from the human body are accurate and reliable.
We have a certificate of accreditation under CLIA to perform “high complexity” testing. Laboratories performing high complexity testing are required to meet more stringent personnel and quality system requirements than laboratories performing less complex tests. To renew our CLIA certificate, we are subject to survey and inspection every two years to assess compliance with program standards. The standards applicable to the testing which we perform may change over time. We were inspected and recertified under CLIA in February 2014. We expect the next regular inspection under CLIA to occur in 2016.
California Laboratory Licensing
In addition to federal certification requirements of laboratories under CLIA, licensure is required and maintained for our laboratory under California law. Such laws establish standards for the day-to-day operation of a clinical laboratory, including the training and skills required of personnel and quality control. In addition, California laws mandate proficiency testing, which involves testing of specimens that have been specifically prepared for the laboratory. We are required to maintain compliance with California standards as a condition to continued operation of our laboratory.
Other States’ Laboratory Testing
Other states require out-of-state laboratories which accept specimens from those states to be licensed. We have obtained licenses in California, Florida, New York, Maryland and Pennsylvania and believe we are in compliance with applicable licensing laws.
Food and Drug Administration
The U.S. Food and Drug Administration regulates the design, testing, development, manufacture, safety, labeling, marketing, promotion, storage, sale and distribution of medical devices pursuant to its authority under the Federal Food, Drug and Cosmetic Act, or FFDCA. The FFDCA and its implementing regulations govern, among other things, the following activities relating to our medical devices: preclinical and clinical testing, design, manufacture, safety, efficacy, labeling, storage, record keeping, sales and distribution, post-market adverse event reporting, import/export, and advertising and promotion. The FDA has also asserted that it has the authority to regulate laboratory-developed tests, known as LDTs, as medical devices under the FFDCA. An LDT is a test developed by a single laboratory for use only in that laboratory, such as AlloMap.
The FDA has traditionally chosen not to exercise its authority to regulate LDTs because it regulates the primary components in most laboratory-developed tests and because it believes that laboratories certified as high complexity under CLIA, such as ours, have demonstrated expertise and ability in test procedures and analysis. However, beginning in September 2006, the FDA issued draft guidance on a subset of LDTs known as “in vitro diagnostic multivariate index assays,” or IVDMIAs. According to the draft guidance, IVDMIAs do not fall within the scope of LDTs over which FDA has exercised enforcement discretion because such tests incorporate complex and unique interpretation functions which require clinical validation. We believed that AlloMap met the definition of IVDMIA set forth in the draft guidance document. As a result, we applied for and obtained in August 2008 510(k) clearance for AlloMap for marketing and sale as a test to aid in the identification of recipients with a low probability of moderate or severe rejection.
On October 3, 2014, the FDA published two draft guidance documents that set forth the FDA’s proposed risk-based framework for regulating LDTs. The draft guidance documents provide the anticipated details through which the FDA would propose to establish an LDT oversight framework, including premarket review for higher-
15
Table of Contents
risk LDTs, such as those that have the same intended use as FDA-approved or cleared companion diagnostics currently on the market. The FDA has allotted 90 days for comment from stakeholders in order to further advance their thinking on their regulatory oversight of LDTs. In addition, the FDA convened a public meeting January 8-9, 2015 also for the purpose of stakeholders to provide input into the FDA process.
The FDA’s LDT guidance documents, if and when finalized, will significantly impact the timing, availability and reimbursement of our future tests, and will require us to modify our business model in order to maintain compliance with these new requirements. For our cfDNA test and all similar testing solutions, we may be required to conduct additional clinical trials to demonstrate clinical validity and utility of our test, and submit to the FDA a premarket approval application, or PMA, or 510(k) premarket notification application and obtain approval or clearance for the test before it can be commercialized. We cannot predict the ultimate timing or form of any FDA final guidance or regulation of LDTs, or when we must obtain regulatory approval or clearance for our LDT solutions. There can be no assurance that any of our tests or additional uses of our tests for which we seek clearance or approval in the future will be cleared or approved on a timely basis, or at all, nor can there be any assurance that labeling claims will be consistent with our current claims or adequate to support continued adoption of and reimbursement for our current and future tests. Moreover, any new FDA requirements could conflict with CLIA requirements and thereby complicate our compliance efforts.
Health Insurance Portability and Accountability Act
Under the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, the U.S. Department of Health and Human Services has issued regulations to protect the privacy and security of protected health information used or disclosed by healthcare providers, such as us. HIPAA also regulates standardization of data content, codes and formats used in healthcare transactions and standardization of identifiers for health plans and providers. Penalties for violations of HIPAA regulations include civil and criminal penalties.
We have developed policies and procedures to comply with these regulations. The requirements under these regulations may change periodically and could have an effect on our business operations if compliance becomes substantially more costly than under current requirements.
In addition to federal privacy regulations, there are a number of state laws governing confidentiality of health information that are applicable to our operations. New laws governing privacy may be adopted in the future as well. We have taken steps to comply with health information privacy requirements to which we are aware that we are subject.
Federal and State Self-referral Prohibitions
We are subject to the federal self-referral prohibitions, commonly known as the Stark Law, and to similar state restrictions such as California’s Physician Ownership and Referral Act, commonly known as PORA. Where applicable, these restrictions generally prohibit us from billing patients or certain governmental or private payers for clinical laboratory testing services when the physician ordering the test, or any member of such physician’s immediate family, has an investment interest in, or compensation arrangement with, us, unless the arrangement meets an exception to the prohibition.
Both the Stark Law and PORA contain exceptions for compensation paid to a physician for personal services rendered by the physician, provided that certain conditions are satisfied. We have compensation arrangements with a number of physicians for personal services, such as speaking engagements and specimen tissue preparation. We have structured these arrangements with terms intended to comply with the requirements of the applicable exceptions to Stark and PORA. However, we cannot be certain that regulators would find these arrangements to be in compliance with Stark, PORA or similar state laws.
16
Table of Contents
Sanctions for a violation of the Stark Law include the following:
| • | denial of Medicare payment for the services provided in violation of the prohibition; |
| • | refunds of amounts collected by an entity in violation of the Stark Law; |
| • | a civil penalty of up to $15,000 for each service arising out of the prohibited referral; |
| • | exclusion from federal healthcare programs, including the Medicare and Medicaid programs; and |
| • | a civil penalty of up to $100,000 against parties that enter into a scheme to circumvent the Stark Law’s prohibition. |
Further, a violation of PORA is a misdemeanor and could result in civil penalties and criminal fines. Finally, other states have self-referral restrictions with which we have to comply that differ from those imposed by federal and California law.
Federal and State Fraud and Abuse Laws
Because of the significant federal funding involved in Medicare and Medicaid, Congress and the states have enacted, and actively enforce, a number of laws to eliminate fraud and abuse in federal healthcare programs. Our business is subject to compliance with these laws. In March 2010, the Patient Protection and Affordable Care Act, as amended by the Healthcare and Education Affordability Reconciliation Act, which we refer to collectively as “the Affordable Care Act,” was enacted in the United States The provisions of the Affordable Care Act are effective on various dates. The Affordable Care Act expands the government’s investigative and enforcement authority and increases the penalties for fraud and abuse, including amendments to both the Anti-Kickback Statute and the False Claims Act, to make it easier to bring suit under these statutes. The Affordable Care Act also allocates additional resources and tools for the government to police healthcare fraud, with expanded subpoena power for HHS, additional funding to investigate fraud and abuse across the healthcare system and expanded use of recovery audit contractors for enforcement.
Anti-Kickback Statutes
The federal healthcare programs’ Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in exchange for or to induce either the referral of an individual, or the furnishing or arranging for a good or service, for which payment may be made under a federal healthcare program such as Medicare or Medicaid.
The definition of “remuneration” has been broadly interpreted to include anything of value, including, for example, gifts, certain discounts, the furnishing of free supplies, equipment or services, credit arrangements, payment of cash and waivers of payments. Several courts have interpreted the statute’s intent requirement to mean that if any one purpose of an arrangement involving remuneration is to induce referrals of federal healthcare covered businesses, the statute has been violated. Penalties for violations include criminal penalties and civil sanctions such as fines, imprisonment and possible exclusion from Medicare, Medicaid and other federal healthcare programs. In addition, violations of the Anti-Kickback Statute also are actionable under the Federal False Claims Act.
Many states have adopted laws similar to the Anti-Kickback Statute. Some of these state prohibitions apply to referral of recipients for healthcare items or services reimbursed by any source, not only the Medicare and Medicaid programs.
Government officials have focused their enforcement efforts on the marketing of healthcare services and products, among other activities, and recently have brought cases against companies, and certain individual sales, marketing and executive personnel, for allegedly offering unlawful inducements to potential or existing customers in an attempt to procure their business.
17
Table of Contents
Federal False Claims Act
Another development affecting the healthcare industry is the increased use of the federal False Claims Act, and in particular, action brought pursuant to the False Claims Act’s “whistleblower” or “qui tam” provisions. The False Claims Act imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal healthcare program. The qui tam provisions of the False Claims Act allow a private individual to bring actions on behalf of the federal government alleging that the defendant has violated the False Claims Act and to share in any monetary recovery. In recent years, the number of suits brought against healthcare providers by private individuals has increased dramatically. In addition, various states have enacted false claims law analogous to the False Claims Act, and many of these state laws apply where a claim is submitted to any third-party payer and not merely a federal healthcare program.
When an entity is determined to have violated the False Claims Act, it may be required to pay up to three times the actual damages sustained by the government, plus civil penalties of between $5,500 and $11,000 for each separate instance of false claim. There are many potential bases for liability under the False Claims Act. Liability arises, primarily, when an entity knowingly submits, or causes another to submit, a false claim for reimbursement to the federal government. The federal government has used the False Claims Act to assert liability on the basis of causing physicians to order excessive or unnecessary services, providing false documentation in support of claims, kickbacks, Stark Law violations and other improper referrals, and CLIA violations, in addition to the more predictable allegations as to misrepresentations with respect to the services rendered. In addition, the federal government has pursued enforcement actions under the False Claims Act in connection with off-label promotion of products. Our future activities relating to billing, compliance with CLIA and Medicare reimbursement requirements, physician and other healthcare provider financial relationships and the sale and marketing of our products may be subject to scrutiny under these laws.
Employees
As of December 31, 2014, we had a total of 68 employees, including 22 employees in sales and marketing and 17 employees in research and development. From time to time we also employ independent contractors, consultants and temporary employees to support our operations. None of our employees are subject to collective bargaining agreements. We have never experienced a work stoppage and believe that our relations with our employees are good.
Environmental Matters
Our operations require the use of hazardous materials (including biological materials) which subjects us to a variety of federal, state and local environmental and safety laws and regulations. Some of these regulations provide for strict liability, holding a party potentially liable without regard to fault or negligence. We could be held liable for damages and fines as a result of our, or others’, business operations should contamination of the environment or individual exposure to hazardous substances occur. We could also be subject to significant fines for failure to comply with applicable environmental, health and safety requirements. We cannot predict how changes in laws or new regulations will affect our business, operations or the cost of compliance.
Available Information
Our website is www.caredx.com. Information contained on, or that can be accessed through, our website is not part of this Annual Report on Form 10-K, and you should not consider information on our website to be part of this report unless specifically incorporated herein by reference. Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Securities Exchange Act of 1934, as amended, are available free of charge on our investor relations website as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. The SEC also maintains a website that contains our SEC filings. The address of the website is www.sec.gov. Further, a copy of this Annual Report on Form 10-K is located at the SEC’s Public Reference Room at 100 F Street, NE, Washington, DC 20549. Information on the operation of the Public Reference Room can be obtained by calling the SEC at 1-800-SEC-0300.
18
Table of Contents
| ITEM 1A. | RISK FACTORS |
Investing in our common stock involves a high degree of risk. You should carefully consider the risks and uncertainties described below, together with all of the other information in this Annual Report on Form 10-K, including the section titled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and related notes, before investing in our common stock. If any of the follows risks occur, our business, financial condition, results of operations and prospects could be materially harmed. In that event, the market price of our common stock could decline, and you could lose part or all of your investment.
Risks Related to Our Business
We have a history of losses, and we expect to incur net losses for the next several years.
We have incurred substantial net losses since our inception, and we expect to continue to incur additional losses for the next several years. For the year ended December 31, 2014 our net income was $0.8 million, and for the years ended 2013 and 2012, we incurred net losses of $3.5 million and $5.1 million, respectively. As of December 31, 2014, we had an accumulated deficit of $159 million. We expect to continue to incur significant operating expenses and anticipate that our expenses will increase due to costs relating to, among other things:
| • | researching, developing, validating and commercializing potential future diagnostic solutions, including our cell-free DNA (cfDNA), solutions currently in development; |
| • | developing, presenting and publishing additional clinical and economic utility data intended to increase payer coverage and clinician adoption of our current and future solutions; |
| • | expansion of our operating capabilities; |
| • | maintenance, expansion and protection of our intellectual property portfolio and trade secrets; |
| • | future clinical trials; |
| • | expansion of the size and geographic reach of our sales force and our marketing capabilities to commercialize potential future solutions; |
| • | employment of additional clinical, quality control, scientific, customer service, laboratory, billing and reimbursement and management personnel; and |
| • | employment of operational, financial, accounting and information systems personnel, consistent with expanding our operations and our status as a newly public company. |
Even if we achieve significant revenues, we may not become profitable, and even if we achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to become and remain consistently profitable could adversely affect the market price of our common stock and could significantly impair our ability to raise capital, expand our business or continue to pursue our growth strategy. For a detailed discussion of our financial condition and results of operations, see “Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
Our financial results are largely dependent on sales of one test, AlloMap, and we will need to generate sufficient revenues from this and other future solutions to grow our business.
Our ability to generate revenue is currently dependent on sales of the AlloMap heart transplant molecular test, or AlloMap, and we expect that sales of AlloMap will account for a substantial portion of our revenue for at least the next several years. Although we are working to develop a cfDNA heart transplant solution, even if we are successful in developing this new test, we expect that it would be marketed as part of AlloMap and that it would not generate additional standalone revenue for us. In addition, while we are in the process of developing a cfDNA solution for kidney transplant recipients, even if we are successful in developing this test, we do not expect this test to be commercially available for at least the next several years. If we are unable to increase sales of AlloMap or successfully develop and commercialize other solutions or enhancements, our revenues and our ability to achieve profitability would be impaired, and the market price of our common stock could decline.
19
Table of Contents
We receive a substantial portion of our revenues from Medicare, and the loss of, or a significant reduction in, reimbursement from Medicare would adversely affect our financial performance.
For the years ended December 31, 2014, 2013 and 2012, payments from Medicare for AlloMap represented approximately 51%, 53% and 52%, respectively, of testing revenue. We anticipate that Medicare will continue to be the payer for a significant portion of our claims for the foreseeable future. However, we may not be able to maintain or increase our tests reimbursed by Medicare for a variety of reasons, including changes in reimbursement practices, general policy shifts, or reductions in reimbursement amounts. We cannot predict whether Medicare reimbursements will continue at the same payment amount or with the same breadth of coverage in the future, if at all.
The development and commercialization of additional diagnostic solutions is a key to our growth strategy. New test development involves a lengthy and complex process, and we may not be successful in our efforts to develop and commercialize additional diagnostic solutions.
A key element of our strategy is to discover, develop, validate and commercialize a portfolio of new diagnostic solutions in addition to AlloMap. While we have engaged in discovery and development activity for our planned cfDNA solution for heart transplant recipients, we will be required to devote considerable additional efforts and resources to the further research and development of this test before it can be made available. Our planned new diagnostic solutions for organs other than the heart, such as our planned cfDNA solution for kidney transplant recipients, are at much earlier stages of development. cfDNA solutions are a novel technology, and to date have not been used commercially in the field of transplantation surveillance. We cannot assure you that we will be able to successfully complete development of or commercialize any of our planned future solutions, or that they will prove to be capable of reliably being used for organ surveillance in the heart or in other types of organs. Before we can successfully develop and commercialize any of our currently planned or other new diagnostic solutions, we will need to:
| • | conduct substantial research and development; |
| • | obtain the necessary testing samples and related data; |
| • | conduct clinical validation studies; |
| • | expend significant funds; |
| • | expand and scale-up our laboratory processes; |
| • | expand and train our sales force; |
| • | gain acceptance from ordering clinicians at a larger number of transplant centers; and |
| • | seek and obtain regulatory clearance or approvals of our new solutions, as required by applicable regulations. |
This process involves a high degree of risk and may take up to several years or more. Our test development and commercialization efforts may be delayed or fail for many reasons, including:
| • | failure of the test at the research or development stage; |
| • | difficulty in accessing suitable testing samples, especially testing samples with known clinical results; |
| • | lack of clinical validation data to support the effectiveness of the test; |
| • | delays resulting from the failure of third-party suppliers or contractors to meet their obligations in a timely and cost-effective manner; |
| • | failure to obtain or maintain necessary clearances or approvals to market the test; or |
| • | lack of commercial acceptance by patients, clinicians, or third-party payers. |
20
Table of Contents
Few research and development projects result in commercial products, and success in early clinical studies often is not replicated in later studies. At any point, we may abandon development of new diagnostic solutions, or we may be required to expend considerable resources repeating clinical trials, which would adversely impact the timing for generating potential revenues from those new diagnostic solutions. In addition, as we develop diagnostic solutions, we will have to make additional investments in our sales and marketing operations, which may be prematurely or unnecessarily incurred if the commercial launch of a test is abandoned or delayed. If a clinical validation study fails to demonstrate the prospectively defined endpoints of the study, we would likely abandon the development of the test or test feature that was the subject of the clinical trial, which could harm our business.
If we do not achieve our projected development goals in the time frames we announce and expect, the commercialization of additional diagnostic solutions by us may be delayed and, as a result, our business will suffer and our stock price may decline.
From time to time, we expect to estimate and publicly announce the anticipated timing of the accomplishment of various clinical and other product development goals, which we sometimes refer to as milestones. In addition, we have included a discussion of a number of anticipated milestones elsewhere in this Annual Report on Form 10-K. The actual timing of these milestones could vary dramatically compared to our estimates, in some cases for reasons beyond our control. We cannot assure you that we will meet our projected milestones and if we do not meet these milestones as publicly announced, the commercialization of our diagnostic solutions may be delayed or may not occur at all and, as a result, our business will suffer and our stock price may decline. Please see the section entitled “Business—Research and Development” for more information regarding our strategies.
The field of diagnostic testing in transplantation is evolving and is subject to rapid technological change. If we are unable to develop solutions to keep pace with rapid medical and scientific change, our operating results could be harmed.
The field of diagnostic testing in transplantation is evolving. Although there have been few advances in technology relating to organ rejection in transplant recipients, the market for medical diagnostic companies is marked by rapid and substantial technological development and innovations which could make AlloMap, and our solutions in development, outdated. We must continually innovate and expand our test offerings to address unmet needs in monitoring transplant related conditions. AlloMap and our solutions under development could become obsolete unless we continually innovate and expand our product offerings to include new clinical applications. If we are unable to demonstrate the effectiveness of AlloMap and future diagnostic solutions, if any, compared to new methodologies and technologies, then sales of our solutions could decline, which would harm our business and financial results.
If clinicians and hospital administrators do not adopt our diagnostic solutions, we will not achieve future sales growth.
Clinicians and healthcare administrators are traditionally slow to adopt new products, testing practices and clinical treatments, partly because of perceived liability risks and the uncertainty of third-party reimbursement. It is critical to the success of our sales efforts that we continue to educate clinicians and administrators about AlloMap and, subject to their development, our future solutions, and demonstrate the clinical benefits of these solutions. We believe that clinicians and transplant centers may not use our solutions unless they determine, based on published peer-reviewed journal articles and the experience of other clinicians, that our solutions provide accurate, reliable and cost-effective information that is useful in monitoring their post-transplant recipients.
We estimate that there are approximately 129 centers managing heart transplant recipients in the United States. In 2014, AlloMap was used in 110 of these centers. However, not all clinicians in these centers are currently using our test. In order for AlloMap sales to grow, we must continue to market to and educate clinicians and
21
Table of Contents
administrators at treatment centers that have used our test to increase the number of clinicians ordering our test, the number of recipients tested and the number of tests per recipient. In addition, we must actively solicit additional treatment centers to establish policies and procedures for ordering our test and to encourage clinicians at those centers to incorporate our test into their standard clinical practice. Some of the challenges that our sales team must overcome include explaining the clinical benefits of AlloMap, which is a highly technical product, and changing a 30-year patient management paradigm of using biopsy as the basis of transplant recipient monitoring. If clinicians and hospital administrators do not adopt and continue to use AlloMap or our future solutions, our business and financial results will suffer.
Our quarterly operating results may fluctuate significantly or may fall below the expectations of investors or securities analysts, each of which may cause our stock price to fluctuate or decline.
Historically, our financial results have been, and we expect that our operating results will continue to be, subject to quarterly fluctuations. Our net income (loss) and other operating results will be affected by numerous factors, including:
| • | our ability to successfully market and sell AlloMap; |
| • | our ability to commercialize new diagnostic solutions; |
| • | the amount of our research and development expenditures; |
| • | the timing of cash collections from third-party payers; |
| • | the extent to which our current test and future solutions, if any, are eligible for coverage and reimbursement from third-party payers; |
| • | changes in coverage and reimbursement or in reimbursement-related laws directly affecting our business; |
| • | any intellectual property infringement lawsuit or opposition, interference or cancellation proceeding in which we may become involved; |
| • | announcements by our competitors of new or competitive products; |
| • | regulatory developments affecting our test or competing products; |
| • | total operating expenses; and |
| • | changes in expectation as to our future financial performance, including financial estimates, publications or research reports by securities analysts. |
If our quarterly operating results fall below the expectations of investors or securities analysts, the price of our common stock could decline substantially. Furthermore, any quarterly fluctuations in our operating results may, in turn, cause the price of our stock to fluctuate substantially. We believe that quarterly comparisons of our financial results are not necessarily meaningful and should not be relied upon as an indication of our future performance.
If the utility of our current solution and solutions in development is not supported by studies published in peer-reviewed medical publications, the rate of adoption of our current and future solutions by clinicians and treatment centers and the rate of reimbursement of our current and future solutions by payers may be negatively affected.
The results of our clinical trials involving AlloMap have been presented at major medical society congresses and published in peer-reviewed publications in leading medical journals. We need to maintain a continued presence in peer-reviewed publications to promote clinician adoption and favorable reimbursement decisions. We believe that peer-reviewed journal articles that provide evidence of the utility of our current and future solutions or the technology underlying AlloMap or future solutions are very important to the commercial success of our current and any future solutions. Clinicians typically take a significant amount of time to adopt new products, testing practices and clinical treatments, partly because of perceived liability risks and the uncertainty of third-party
22
Table of Contents
reimbursement. It is critical to the success of our sales efforts that we educate a sufficient number of clinicians and administrators about AlloMap and our future solutions, and demonstrate the clinical benefits of these solutions. Clinicians may not adopt, and third-party payers may not cover or adequately reimburse for, our current and future solutions unless they determine, based on published peer-reviewed journal articles and the experience of other clinicians, that our diagnostic current and future solutions provide accurate, reliable and cost-effective information that is useful in monitoring transplant recipients and making informed and timely treatment decisions.
The administration of clinical and economic utility studies is expensive and demands significant attention from our management team. Data collected from these studies may not be positive or consistent with our existing data, or may not be statistically significant or compelling to the medical community. If the results obtained from our ongoing or future studies are inconsistent with certain results obtained from our previous studies, adoption of our current and future solutions would suffer and our business would be harmed. While we have had success in generating peer-reviewed publications regarding AlloMap, peer-reviewed publications regarding our future solutions may be limited by many factors, including delays in the completion of, poor design of, or lack of compelling data from clinical studies that would be the subject of the article. If our current and future solutions or the technology underlying AlloMap or our future solutions do not receive sufficient favorable exposure in peer-reviewed publications, the rate of clinician adoption and positive reimbursement coverage decisions could be negatively affected. The publication of clinical data in peer-reviewed journals is a crucial step in commercializing and obtaining reimbursement for diagnostic solutions such as ours, and our inability to control when, if ever, results are published may delay or limit our ability to derive sufficient revenue from any product that is the subject of a study.
Transplant centers may not adopt AlloMap or future solutions due to historical practices or due to more favorable reimbursement policies associated with other means of monitoring transplants.
Due to the historically limited monitoring options and the well-established coverage and reimbursement for biopsies, clinicians are accustomed to monitoring for acute cellular rejection in heart transplant recipients by utilizing biopsies. Many clinicians use our test in parallel with biopsies rather than as an alternative to biopsies. While we do not market AlloMap as a biopsy alternative, per se, if treatment center administrators view our test as an alternative to a biopsy and believe they would derive more revenue from the performance of biopsies, such administrators may be motivated to reduce or avoid the use of our test. We cannot provide assurance that our efforts will increase the use of our test by new or existing customers. Our failure to increase the frequency of use of our test by new and existing customers would adversely affect our growth and revenues.
If we are unable to successfully compete with larger and more established players in the clinical surveillance of transplantation field, we may be unable to increase or sustain our revenues or achieve profitability.
Our AlloMap solution for heart transplant recipients competes against existing diagnostic tests utilized by pathologists, which, in the case of heart transplant rejection, generally involve evaluating biopsy samples to determine the presence or absence of rejection. This practice has been the standard of care in the United States for many years, and we will need to continue to educate clinicians, transplant recipients and payers about the various benefits of our test in order to change clinical practice.
Competition for kidney surveillance diagnostics can also come from biopsies. However, because of the risks and discomforts of the invasive kidney biopsy procedure, as well as the expense and relatively low rate of finding moderate to severe grade rejection, biopsy is not a standard practice for surveillance of transplanted kidneys. Additional competition for kidney surveillance diagnostics currently comes from general, non-specific clinical chemistry tests such as serum creatinine, urine protein, complete blood count, lipid profile and others that are widely ordered by physician offices and routinely performed in clinical reference labs and hospital labs.
We expect the competition for post-transplant surveillance to increase as there are numerous established and startup companies in the process of developing novel products and services for the transplant market which may directly or indirectly compete with AlloMap or our development pipeline. In addition to companies focused on
23
Table of Contents
pre-transplantation such as Thermo Fisher Scientific Inc.’s One Lambda and Immucor, Inc.’s LIFECODES businesses, companies who have not historically focused on transplantation, but with existing knowledge of cfDNA technology have indicated they are considering this market.
The field of clinical surveillance of transplantation is evolving. New and well established companies are devoting substantial resources to the application of molecular diagnostics to the treatment of medical conditions. Some of these companies may elect to develop and market diagnostic solutions in the post-transplant surveillance market.
Many of our potential competitors have greater brand recognition and substantially greater financial and technical resources and development, production and marketing capabilities than we do. Others may develop lower-priced, less complex tests that could be viewed by clinicians and payers as functionally equivalent to our test, which could force us to lower the current list price of our test and impact our operating margins and our ability to achieve profitability. If we are unable to compete successfully against current or future competitors, we may be unable to increase market acceptance for and sales of AlloMap and our future solutions, which could prevent us from increasing or sustaining our revenues or achieving profitability and could cause the market price of our common stock to decline.
Our research and development efforts will be hindered if we are not able to acquire or contract with third parties for access to additional tissue and blood samples.
Our clinical development relies on our ability to secure access to tissue and blood samples, as well as recipient information including biopsy results and clinical outcomes from the same patient. Furthermore, the studies through which our future solutions are developed rely on access to multiple samples from the same recipient over a period of time as opposed to samples at a single point in time or archived samples. We will require additional samples and recipient data for future research, development and validation. Access to recipients and samples on a real-time, or non-archived, basis is limited and often on an exclusive basis, and there is no guarantee that initiatives such as the DART study will be successful in obtaining and validating additional samples. Additionally, the process of negotiating access to new and archive recipient data and samples is lengthy since it typically involves numerous parties and approval levels to resolve complex issues such as usage rights, institutional review board approval, recipient consent, privacy rights and informed consent of recipients, publication rights, intellectual property ownership and research parameters. If we are not able to acquire or negotiate access to new and archived recipient data and blood samples with source institutions, or if other laboratories or our competitors secure access to these samples before us, our ability to research, develop and commercialize future solutions will be limited or delayed.
If we cannot enter into and maintain new clinical collaborations, our efforts to commercialize AlloMap and our development of new products could be delayed.
In the past, we have entered into clinical trial collaborations with highly regarded academic institutions and leading treatment centers in the transplant field. Our success in the future may depend in part on our ability to enter into agreements with other leading institutions in the transplant field. Securing these agreements can be difficult due to internal and external constraints placed on these organizations. Some organizations may limit the number of collaborations they have with any one company so as to not be perceived as biased or conflicted. Organizations may also have insufficient administrative and related infrastructure to enable collaborations with many companies at once, which can extend the time it takes to develop, negotiate and implement a collaboration. In addition to completing clinical trial collaborations, publication of clinical data in peer-reviewed journals is a crucial step in commercializing and obtaining coverage and reimbursement for solutions such as ours. Our inability to control when, if ever, results of such studies are published may delay or limit our ability to derive sufficient revenues from any test that may result from a collaboration.
From time to time we expect to engage in discussions with potential clinical collaborators, which may or may not lead to collaborations. We cannot guarantee that any discussions will result in clinical collaborations or that any clinical studies which may result will be enrolled or completed in a reasonable time frame or with successful outcomes. Once news of discussions regarding possible collaborations become known in the medical community,
24
Table of Contents
regardless of whether the news is accurate, failure to announce a collaborative agreement or the entity’s announcement of a collaboration with an entity other than us may result in adverse speculation about us, our current and future solutions or our technology, resulting in harm to our reputation and our business.
If we are unable to successfully manage our growth and support demand for our test, our business may suffer.
As our test volume grows, we will need to continue to ramp up our testing capacity, implement increases in scale and related processing, customer service, billing and systems process improvements and expand our internal quality assurance program to support testing on a larger scale. We will also need additional certified laboratory scientists and other scientific and technical personnel to process our tests. We cannot assure you that any increases in scale, related improvements and quality assurance will be successfully implemented or that appropriate personnel will be available. As additional products are developed, we may need to bring new equipment on-line, implement new systems, technology, controls and procedures and hire personnel with different qualifications. We plan to expand our sales force to support additional products. There is significant competition for qualified, productive sales personnel with advanced sales skills and technical knowledge in our field. Our ability to achieve significant growth in revenue in the future will depend, in large part, on our success in recruiting, training, and retaining sufficient qualified sales personnel.
The value of AlloMap depends, in large part, on our ability to perform AlloMap on a timely basis and at a high quality standard, and on our reputation for such timeliness and quality. Failure to implement necessary procedures, transition to new equipment or processes or to hire new personnel could result in higher costs of processing or an inability to meet market demand in a timely manner. There can be no assurance that we will be able to perform AlloMap or our future solutions, if any, on a timely basis at a level consistent with demand, that our efforts to scale our commercial operations will not negatively affect the quality of test results or that we will be successful in responding to the growing complexity of our testing operations. If we encounter difficulty meeting market demand for our current and future solutions, our reputation could be harmed and our future prospects and our business could suffer.
In addition, our growth may place a significant strain on our management, operating and financial systems and our sales, marketing and administrative resources. As a result of our growth, our operating costs may escalate even faster than planned, and some of our internal systems may need to be enhanced or replaced. If we cannot effectively manage our expanding operations and our costs, we may not be able to grow effectively or we may grow at a slower pace, and our business could be adversely affected.
Our recent testing revenue growth rates may not be indicative of future growth, and we may not continue to grow at our recent pace, or at all.
From 2013 to 2014, our testing revenue grew from $21.7 million to $25.8 million, which represents annual growth of 19%. In the future, our revenue may not grow as rapidly as it has over the past several years. We believe that our future revenue growth will depend on, among other factors:
| • | the continued usage and acceptance of our current and future solutions; |
| • | demand for our products and services; |
| • | the introduction and acceptance of new or enhanced products or services by us or by competitors; |
| • | our ability to maintain reimbursement for AlloMap and secure reimbursement for our future solutions; |
| • | our ability to anticipate and effectively adapt to developing markets and to rapidly changing technologies; |
| • | our ability to attract, retain and motivate qualified personnel; |
| • | the initiation, renewal or expiration of significant contracts with our commercial partners; |
| • | pricing changes by us, our suppliers or our competitors; and |
| • | general economic conditions and other factors. |
25
Table of Contents
We may not be successful in our efforts to manage any of the foregoing, and any failure to be successful in these efforts could materially and adversely affect revenue growth. You should not consider our past revenue growth to be indicative of future growth.
If our sole laboratory facility becomes inoperable, we will be unable to perform AlloMap and future solutions, if any, and our business will be harmed.
We perform all of our diagnostic services in our laboratory located in Brisbane, California. We do not have redundant laboratory facilities. Brisbane is situated on or near earthquake fault lines. Our facility and the equipment we use to perform AlloMap would be costly to replace and could require substantial lead time to repair or replace, if damaged or destroyed. The facility may be harmed or rendered inoperable by natural or man-made disasters, including earthquakes, wildfires, flooding and power outages, which may render it difficult or impossible for us to perform our tests for some period of time. The inability to perform our tests may result in the loss of customers or harm our reputation, and we may be unable to regain those customers in the future. Although we possess insurance for damage to our property and the disruption of our business, this insurance may not be sufficient to cover all of our potential losses and may not continue to be available to us on acceptable terms, if at all.
In order to establish a redundant laboratory facility, we would have to spend considerable time and money securing adequate space, constructing the facility, recruiting and training employees, and establishing the additional operational and administrative infrastructure necessary to support a second facility. Additionally, any new clinical laboratory facility opened by us would be required to be certified under the Clinical Laboratory Improvement Amendments of 1988, or CLIA, a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for the diagnosis, prevention or treatment of disease. We would also be required to secure and maintain state licenses required by several states, including California, Florida, Maryland, New York, and Pennsylvania, which can take a significant amount of time and result in delays in our ability to begin operations at that facility. If we failed to secure any such licenses, we would not be able to process samples from recipients in such states. We also expect that it would be difficult, time-consuming and costly to train, equip and use a third-party to perform tests on our behalf. We could only use another facility with the established state licensures and CLIA certification necessary to perform AlloMap or future solutions following validation and other required procedures. We cannot assure you that we would be able to find another CLIA-certified facility willing or able to adopt AlloMap or future solutions and comply with the required procedures, or that this laboratory would be willing or able to perform the tests for us on commercially reasonable terms.
Our commercial partner in Europe will rely on a third party laboratory to perform AlloMap. Training and other preparation is required before the laboratory is operational, and our partner may encounter unanticipated obstacles. We do not have access to redundant facilities in Europe and our exclusive arrangement precludes the engagement by us of another collaboration partner whose laboratories we could use in the event that our primary facility is harmed or rendered inoperable. Without immediate access to an alternative facility, any disruption to our European partner’s laboratory may result in delays in the delivery of test results, patient claims, loss of customers or harm to our reputation.
Performance issues, service interruptions or price increases by our shipping carriers could adversely affect our business and harm our reputation and ability to provide our services on a timely basis.
Expedited, reliable shipping is essential to our operations. We rely heavily on providers of transport services for reliable and secure point-to-point transport of recipient samples to our laboratory and enhanced tracking of these recipient samples. Should a carrier encounter delivery performance issues such as loss, damage or destruction of a sample, it may be difficult to replace our recipient samples in a timely manner and such occurrences may damage our reputation and lead to decreased demand for our services and increased cost and expense to our business. In addition, any significant increase in shipping rates could adversely affect our operating margins and results of operations. Similarly, strikes, severe weather, natural disasters or other service interruptions affecting delivery services we use would adversely affect our ability to receive and process recipient samples on a timely basis.
26
Table of Contents
Our ability to commercialize the diagnostic solutions that we develop is dependent on our relationships with laboratory services providers and their willingness to support our current and future solutions.
We rely on third-party laboratory services providers to draw the recipient blood samples that are analyzed in our Brisbane, California laboratory. The Company’s business will suffer if these service providers do not support AlloMap or the other solutions that we may develop. For example, these laboratories may deem the effort to process the samples for our solutions to require too much additional effort. Additionally, if transplant facilities have relationships with large reference laboratories that will not process and send out our specimens, the clinicians at these facilities may deem ordering our tests outside of these relationships too inconvenient for their patients. A lack of acceptance of our current and future solutions by these service providers could result in lower test volume.
If we are unable to raise additional capital on acceptable terms in the future, it may limit our ability to develop and commercialize new diagnostic solutions and technologies, and we may have to curtail or cease operations.
We expect capital outlays and operating expenditures to increase over the next several years as we expand our infrastructure, commercial operations and research and development activities. Specifically, we may need to raise additional capital to, among other things:
| • | complete development of our proposed cfDNA test for heart and kidney or to develop other solutions for clinical surveillance in transplantation; |
| • | increase our selling and marketing efforts to drive market adoption and address competitive developments; |
| • | expand our clinical laboratory operations; |
| • | fund our clinical validation study activities; |
| • | expand our research and development activities; |
| • | sustain or achieve broader commercialization of AlloMap or enhancements to that test; |
| • | acquire or license products or technologies; and |
| • | finance our capital expenditures and general and administrative expenses. |
Our present and future funding requirements will depend on many factors, including:
| • | the level of research and development investment required to develop our cfDNA test for heart transplant recipients and additional solutions for the surveillance of transplantation of other organs; |
| • | costs of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; |
| • | our need or decision to acquire or license complementary technologies or acquire complementary businesses; |
| • | changes in test development plans needed to address any difficulties in commercialization; |
| • | competing technological and market developments; |
| • | whether our diagnostic solutions become subject to additional U.S. Food and Drug Administration, or FDA, or other regulation; and |
| • | changes in regulatory policies or laws that affect our operations. |
Additional capital, if needed, may not be available on satisfactory terms, or at all. Furthermore, if we raise additional funds by issuing equity securities, dilution to our existing stockholders could result. Any equity securities issued also may provide for rights, preferences or privileges senior to those of holders of our common stock. If we raise additional funds by issuing debt securities, these debt securities would have rights, preferences and privileges senior to those of holders of our common stock, and the terms of the debt securities issued could impose significant restrictions on our operations. If we raise additional funds through collaborations and
27
Table of Contents
licensing arrangements, we might be required to relinquish significant rights to our technologies, AlloMap or our solutions under development, or grant licenses on terms that are not favorable to us, which could lower the economic value of those programs to our company. If adequate funds are not available, we may have to scale back our operations or limit our research and development activities, which may cause us to grow at a slower pace, or not at all, and our business could be adversely affected.
The loss of key members of our senior management team or our inability to attract and retain highly skilled scientists, clinicians and laboratory and field personnel could adversely affect our business.
Our success depends largely on the skills, experience and performance of key members of our executive management team. The efforts of each of these persons will be critical to us as we continue to develop our technologies and testing processes and as we attempt to transition to a company with more than one commercialized test. If we were to lose one or more of these key employees, we may experience difficulties in competing effectively, developing our technologies and implementing our business strategies.
Our research and development programs and commercial laboratory operations depend on our ability to attract and retain highly skilled scientists and technicians, including geneticists, biostatisticians, engineers, licensed laboratory technicians and chemists. We may not be able to attract or retain qualified scientists and technicians in the future due to the intense competition for qualified personnel among life science businesses, particularly in the San Francisco Bay Area. We also face competition from universities, public and private research institutions and other organizations in recruiting and retaining highly qualified scientific personnel.
In addition, our success depends on our ability to attract and retain laboratory and field personnel with extensive experience in post-transplant recipient care and surveillance and close relationships with clinicians, pathologists and other hospital personnel. We may have difficulties locating, recruiting or retaining qualified salespeople, which could cause a delay or decline in the rate of adoption of AlloMap or our future solutions, if any. If we are not able to attract and retain the necessary personnel to accomplish our business objectives, we may experience constraints that will adversely affect our ability to support our discovery, development, verification and commercialization programs.
Recent and future acquisitions and investments could disrupt our business and harm our financial condition and operating results.
Our success will depend, in part, on our ability to expand our existing know-how, expertise and intellectual property in other fields, including for the development of other commercial tests. In some circumstances, we may decide to do so through the acquisition of complementary businesses and technologies rather than through internal development, including, for example, our 2014 acquisition of ImmuMetrix, Inc., a privately held development-stage company working on cfDNA-based solutions in transplantation and other fields. The identification of suitable acquisition candidates can be difficult, time-consuming and costly, and we may not successfully complete acquisitions that we target in the future. The risks we face in connection with acquisitions, including our acquisition of ImmuMetrix, Inc., include:
| • | diversion of management time and focus from operating our business to addressing acquisition integration challenges; |
| • | reduction of available cash reserves, assumption of debt or dilutive issuances of equity securities due to payment of consideration; |
| • | coordination of research and development and sales and marketing functions; |
| • | integration of product and service offerings; |
| • | acquired technology or research and development expectations prove unsuccessful; |
| • | retention of key personnel from the acquired company; |
28
Table of Contents
| • | financial reporting, revenue recognition or other financial control deficiencies of the acquired company that we do not adequately address and that cause our reported results to be incorrect; |
| • | liability for activities of the acquired company before the acquisition, including intellectual property infringement claims, violations of laws, commercial disputes, tax liabilities and other known and unknown liabilities; and |
| • | litigation or other claims in connection with the acquired company, including claims from terminated employees, customers, former stockholders or other third parties. |
Our failure to address these risks or other problems encountered in connection with our past or future acquisitions and investments could cause us to fail to realize the anticipated benefits of these acquisitions or investments, cause us to incur unanticipated liabilities, and harm our business generally. For example, we completed our acquisition of ImmuMetrix, Inc. in June 2014, and some risks remain, including the risks that the intellectual property we acquired in this acquisition may not lead to a successful product, risks associated with milestone payments due under the merger agreement and the probability of achieving them, and the risk that Stanford University could terminate our patent license relating to the diagnosis of rejection in organ transplant recipients using cfDNA if we do not meet certain performance and commercialization conditions. There is also a risk that future acquisitions will result in the incurrence of debt, contingent liabilities, amortization expenses, incremental operating expenses or the write-off of goodwill, any of which could harm our business and results of operations.
We may acquire other businesses or assets or form joint ventures that could harm our operating results, dilute your ownership of us, increase our debt or cause us to incur significant expense.
As part of our business strategy, we may pursue acquisitions of complementary businesses and assets, as well as technology licensing arrangements. We also may pursue strategic alliances that leverage our core technology and industry experience to expand our test offerings or distribution. We have limited experience with respect to acquiring other companies and limited experience with respect to the acquisition of strategic assets or the formation of collaborations, strategic alliances and joint ventures. If we make any acquisitions, we may not be able to integrate these acquisitions successfully into our existing business, and we could assume unknown or contingent liabilities. Any future acquisitions by us also could result in significant write-offs or the incurrence of debt and contingent liabilities, any of which could harm our operating results. Integration of an acquired company, product or technology also may require management resources that otherwise would be available for ongoing development of our existing business. We may not identify or complete these transactions in a timely manner, on a cost-effective basis, or at all, and we may not realize the anticipated benefits of any acquisition, technology license, strategic alliance or joint venture.
To finance any acquisitions, we may choose to issue shares of our common stock as consideration, which would dilute your interest in us. If the price of our common stock is low or volatile, we may not be able to acquire other companies using our stock as consideration. Alternatively, it may be necessary for us to raise additional funds for acquisitions through public or private financings. Additional funds may not be available on terms that are favorable to us, or at all.
Defects in AlloMap or other solutions we develop could result in substantial product liabilities or professional liabilities that exceed our resources.
The marketing, sale and use of AlloMap and future solutions could lead to the filing of product liability claims if someone were to allege that our test failed to perform as it was designed. For example, a defect in one of our diagnostic solutions could lead to a false positive or false negative result, affecting the eventual diagnosis. Any incomplete or inaccurate analysis on the part of our technicians could also affect the reliability of the test results. A product liability or professional liability claim could result in substantial damages and be costly and time-consuming for us to defend. Although we maintain product and professional liability insurance, our
29
Table of Contents
insurance may not fully protect us from the financial impact of defending against product liability or professional liability claims or any judgments, fines or settlement costs arising out of any such claims. Any product liability or professional liability claim brought against us, with or without merit, could increase our insurance rates or prevent us from securing insurance coverage in the future. Additionally, any product liability lawsuit could cause injury to our reputation, result in the suspension of our testing pending an investigation into the cause of the alleged failure, or cause current collaborators to terminate existing agreements and potential collaborators to seek other partners, any of which could impact our results of operations.
We rely extensively on third party service providers. Failure of these parties to perform as expected, or interruptions in the Company’s relationship with these providers or their provision of services to the Company, could interfere with the Company’s ability to provide test results.
The Company’s relationship with any of its third party service providers may impair the Company’s ability to perform its services. The failure of any of the Company’s third party service providers to adequately perform their service obligations may reduce the Company’s revenues and increase its expenses or prevent the Company from providing its services in a timely manner or if at all. In addition, the Company’s reputation, business and financial performance could be materially harmed if it is unable to, or is perceived as unable to, perform reliable services.
We rely solely on certain suppliers to supply some of the laboratory instruments and key reagents that we use to perform AlloMap. These sole source suppliers include Thermo Fisher Scientific Inc., which supplies us with instruments, laboratory reagents and consumables, Becton, Dickinson and Company, which supplies us with cell preparation tubes, or CPTs, and Therapak Corporation, which supplies us with a proprietary buffer reagent. One of the reagents supplied to us by Therapak Corporation is, in turn, obtained by Therapak Corporation from Qiagen N.V. and is a proprietary formulation of Qiagen N.V. We have no relationship with or control over, Qiagen N.V. We do not have guaranteed supply agreements with Thermo Fisher Scientific Inc., Becton, Dickinson and Company, Therapak Corporation or Qiagen N.V., which exposes us to the risk that these suppliers may choose to discontinue doing business with us at any time. We periodically forecast our needs to these sole source suppliers and enter into standard purchase orders based on these forecasts.
A critical test component needed to perform AlloMap is a universal master mix that is supplied by Thermo Fisher Scientific Inc., who has discontinued production of the formulation originally used. We have worked with Thermo Fisher Scientific Inc. to develop and validate an equivalent performing alternative universal master mix. We have successfully completed our validation and equivalence testing for this material, which will allow us to purchase the replacement universal master mix on a routine basis from Thermo Fisher Scientific. At present, we have sufficient master mix material in house to continue delivering AlloMap through December 2015 and the ability to purchase additional material as needed to support AlloMap testing without restriction.
In addition, our ABI 7900 Thermocycler, a real time PCR instrument used in AlloMap, is no longer in production. Thermo Fisher Scientific Inc. has committed to provide service and support of this instrument through 2017. We believe we have secured sufficient instrument inventory to last for the next three to five years and are in the process of validating an alternative instrument. We believe that there are relatively few suppliers other than Thermo Fisher Scientific Inc., Becton, Dickinson and Company and Qiagen N.V. that are currently capable of supplying the instruments, reagents and other supplies necessary for AlloMap. Even if we were to identify secondary suppliers, there can be no assurance that we will be able to enter into agreements with such suppliers on a timely basis on acceptable terms, if at all. If we should encounter delays or difficulties in securing from Thermo Fisher Scientific Inc., Becton, Dickinson and Company or Therapak Corporation, or Therapak Corporation encounters delays or difficulties with Qiagen N.V., the quality and quantity of reagents, or supplies, or instruments that we require for AlloMap or other solutions we develop, we may need to reconfigure our test processes, which would result in delays in commercialization or an interruption in sales. Clinicians who order AlloMap rely on the continued availability of our test and have an expectation that results will be reported within two to three business days. If we are unable to provide results within a timely manner, clinicians may elect not to use our test in the future and our business and operating results could be harmed.
30
Table of Contents
Security breaches, loss of data and other disruptions could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of our business, we and our third-party billing and collections provider collect and store sensitive data, including legally-protected health information, credit card information and personally identifiable information about our customers, payers, recipients and collaboration partners. We also store sensitive intellectual property and other proprietary business information, including that of our customers, payers and collaboration partners. We manage and maintain our applications and data utilizing a combination of on-site systems, managed data center systems and cloud-based data center systems. These applications and data encompass a wide variety of business critical information, including research and development information, commercial information and business and financial information.
We face four primary risks relative to protecting this critical information: loss of access risk, inappropriate disclosure risk, inappropriate modification risk and the risk of our being unable to identify and audit our controls over the first three risks.
We are highly dependent on information technology networks and systems, including the Internet, to securely process, transmit and store this critical information. Security breaches of this infrastructure, including physical or electronic break-ins, computer viruses, attacks by hackers and similar breaches, can create system disruptions, shutdowns or unauthorized disclosure or modification of confidential information. The secure processing, storage, maintenance and transmission of this critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive information from unauthorized access or disclosure, our information technology and infrastructure, and that of our third-party billing and collections provider, may be vulnerable to attacks by hackers or viruses or breached due to employee error, malfeasance or other disruptions.
A security breach or privacy violation that leads to disclosure or modification of or prevents access to consumer information (including personally identifiable information or protected health information) could harm our reputation, compel us to comply with disparate state breach notification laws, require us to verify the correctness of database contents and otherwise subject us to liability under laws that protect personal data, resulting in increased costs or loss of revenue. If we are unable to prevent such security breaches or privacy violations or implement satisfactory remedial measures, our operations could be disrupted, and we may suffer loss of reputation, financial loss and other regulatory penalties because of lost or misappropriated information, including sensitive consumer data. In addition, these breaches and other inappropriate access can be difficult to detect, and any delay in identifying them may lead to increased harm of the type described above.
Any such breach or interruption could compromise our networks or those of our third-party billing and collections provider, and the information stored there could be inaccessible or could be accessed by unauthorized parties, publicly disclosed, lost or stolen. Any such interruption in access, improper access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, such as the Health Insurance Portability and Accountability Act of 1996, or HIPAA, and regulatory penalties. Unauthorized access, loss or dissemination could also disrupt our operations, including our ability to perform tests, provide test results, bill payers or patients, process claims and appeals, provide customer assistance services, conduct research and development activities, collect, process and prepare company financial information, provide information about our current and future solutions and other patient and clinician education and outreach efforts through our website, and manage the administrative aspects of our business and damage our reputation, any of which could adversely affect our business. Any such breach could also result in the compromise of our trade secrets and other proprietary information, which could adversely affect our competitive position.
In addition, the interpretation and application of consumer, health-related, privacy and data protection laws in the U.S., Europe and elsewhere are often uncertain, contradictory and in flux. It is possible that these laws may be
31
Table of Contents
interpreted and applied in a manner that is inconsistent with our practices. If so, this could result in government-imposed fines or orders requiring that we change our practices, which could adversely affect our business. Complying with these various laws could cause us to incur substantial costs or require us to change our business practices and compliance procedures in a manner adverse to our business.
International expansion of our business exposes us to business, regulatory, political, operational, financial and economic risks associated with doing business outside of the United States.
As part of our longer-term growth strategy, we intend to target select international markets to grow our presence outside of the U.S. We currently have commercial agreements for the promotion of AlloMap in Europe and Canada with Diaxonhit and LifeLabs Medical Laboratories Services, respectively. To promote the growth of our business internationally, we will need to attract additional partners to expand into new markets. Relying on partners for our sales and marketing subjects us to various risks, including:
| • | our partners may fail to commit the necessary resources to develop a market for our products, may spend the majority of their time selling products unrelated to ours, or may be unsuccessful in marketing our products for other reasons; |
| • | under certain agreements, our partners’ obligations, including their required level of promotional activities, may be conditioned upon our ability to achieve or maintain a specified level of reimbursement coverage; |
| • | agreements with our partners may terminate prematurely due to disagreements or may result in disputes or litigation with our partners; |
| • | we may not be able to renew existing partner agreements, or enter into new agreements, on acceptable terms; |
| • | our existing relationships with partners may preclude us from entering into additional future arrangements; |
| • | our partners may violate local laws or regulations, potentially causing reputational or monetary damage to our business; |
| • | our partners may engage in sales practices that are locally acceptable but do not comply with standards required under U.S. laws that apply to us; and |
| • | our partners in Europe may be negatively affected by the financial instability of, and austerity measures implemented by, several countries in Europe. |
If our present or future partners do not perform adequately, or we are unable to enter into agreements in new markets, we may be unable to achieve revenue growth or market acceptance in jurisdictions in which we depend on partners.
In addition, conducting international operations subjects us to new risks that, generally, we have not faced in the U.S., including:
| • | uncertain or changing regulatory registration and approval processes associated with AlloMap and other potential diagnostic solutions; |
| • | failure by us to obtain regulatory approvals or adequate reimbursement for the use of our current and future solutions in various countries; |
| • | competition from companies located in the countries in which we offer our products may put us at a competitive disadvantage; |
| • | financial risks, such as longer accounts receivable payment cycles and difficulties in collecting accounts receivable; |
| • | logistics and regulations associated with shipping recipient samples, including infrastructure conditions and transportation delays; |
32
Table of Contents
| • | limits in our ability to penetrate international markets if we are not able to process solutions locally; |
| • | difficulties in managing and staffing international operations and assuring compliance with foreign corrupt practices laws; |
| • | potentially adverse tax consequences, including the complexities of foreign value added tax systems, tax inefficiencies related to our corporate structure and restrictions on the repatriation of earnings; |
| • | increased financial accounting and reporting burdens and complexities; |
| • | multiple, conflicting and changing laws and regulations such as healthcare regulatory requirements and other governmental approvals, permits and licenses; |
| • | the imposition of trade barriers such as tariffs, quotas, preferential bidding or import or export licensing requirements; |
| • | political and economic instability, including wars, terrorism, and political unrest, general security concerns, outbreak of disease, boycotts, curtailment of trade and other business restrictions; |
| • | fluctuations in currency exchange rates; |
| • | regulatory and compliance risks that relate to maintaining accurate information and control over activities that may fall within the purview of the Foreign Corrupt Practices Act of 1977, its books and records provisions or its anti-bribery provisions, as well as risks associated with other anti-bribery and anti-corruption laws; and |
| • | reduced or varied protection for intellectual property rights in some countries. |
The occurrence of any one of the above could harm our business and, consequently, our revenues and results of operations. Our expanding international operations could be affected by changes in laws, trade regulations, labor and employment regulations, and procedures and actions affecting approval, production, pricing, reimbursement and marketing of our current and future solutions, as well as by inter-governmental disputes. Any of these changes could adversely affect our business. Additionally, operating internationally requires significant management attention and financial resources. We cannot be certain that the investment and additional resources required in establishing operations in other countries will produce desired levels of revenue or profitability.
In addition, any failure to comply with applicable legal and regulatory obligations could impact us in a variety of ways that include, but are not limited to, significant criminal, civil and administrative penalties, including imprisonment of individuals, fines and penalties, denial of export privileges, seizure of shipments, and restrictions on certain business activities. Also, the failure to comply with applicable legal and regulatory obligations could result in the disruption of our distribution and sales activities.
Our success expanding internationally will depend, in part, on our ability to develop and implement policies and strategies that are effective in anticipating and managing these and other risks in the countries in which we do business. Failure to manage these and other risks may have a material adverse effect on our operations in any particular country and on our business as a whole.
Our insurance policies are expensive and protect us only from some business risks, which will leave us exposed to significant uninsured liabilities.
We do not carry insurance for all categories of risk that our business may encounter. For example, we do not carry earthquake insurance. In the event of a major earthquake in our region, our business could suffer significant and uninsured damage and loss. Some of the policies we currently maintain include general liability, foreign liability, employee benefits liability, property, automobile, umbrella, workers’ compensation, products liability and directors’ and officers’ insurance. We do not know, however, if we will be able to maintain existing insurance with adequate levels of coverage. Any significant uninsured liability may require us to pay substantial amounts, which would adversely affect our cash position and results of operations.
33
Table of Contents
If we use hazardous materials in a manner that causes injury, we could be liable for damages.
Our activities currently require the use of hazardous chemicals. We cannot eliminate the risk of accidental contamination or injury to employees or third parties from the use, storage, handling or disposal of these materials. In the event of contamination or injury, we could be held liable for any resulting damages, and any liability could exceed our resources or any applicable insurance coverage we may have. Additionally, we are subject on an ongoing basis to federal, state and local laws and regulations governing the use, storage, handling and disposal of these materials and specified waste products.
We may use third party collaborators to help us develop, validate or commercialize any new diagnostic solutions, and our ability to commercialize such solutions could be impaired or delayed if these collaborations are unsuccessful.
We may in the future selectively pursue strategic collaborations for the development, validation and commercialization of any new diagnostic solutions we may develop. In any future third party collaboration, we may be dependent upon the success of the collaborators in performing their responsibilities and their continued cooperation. Our collaborators may not cooperate with us or perform their obligations under our agreements with them. We cannot control the amount and timing of our collaborators’ resources that will be devoted to performing their responsibilities under our agreements with them. Our collaborators may choose to pursue alternative technologies in preference to those being developed in collaboration with us. The development, validation and commercialization of our potential solutions may be delayed if collaborators fail to fulfill their responsibilities in a timely manner or in accordance with applicable regulatory requirements or if they breach or terminate their collaboration agreements with us. AlloMap testing in Europe and Canada will be conducted through exclusive distribution agreements with a sole collaborator in each region. Any issues arising from these arrangements will affect our ability to serve the entire region, and our reputation may suffer even if we subsequently locate new partners, which may permanently affect our business. Disputes with our collaborators could also impair our reputation or result in development delays, decreased revenues and litigation expenses.
Changes in, or interpretations of, accounting rules and regulations could result in unfavorable accounting changes or require us to change our compensation policies.
Accounting methods and policies for diagnostic companies, including policies governing revenue recognition, research and development and related expenses and accounting for stock-based compensation, are subject to further review, interpretation and guidance from relevant accounting authorities, including the SEC. Changes to, or interpretations of, accounting methods or policies may require us to reclassify, restate or otherwise change or revise our financial statements, including those contained in this Annual Report on Form 10-K.
Risks Related to Billing and Reimbursement
Health insurers and other third-party payers may decide to revoke coverage of our existing test, decide not to cover our future solutions or may provide inadequate reimbursement, which could jeopardize our commercial prospects.
Successful commercialization of AlloMap depends, in large part, on the availability of coverage and adequate reimbursement from government and private payers. Favorable third-party payer coverage and reimbursement are essential to meeting our immediate objectives and long-term commercial goals. We do not recognize revenue for test results delivered without a contract for reimbursement, or an established coverage policy and a history of payment. Revenue for AlloMap is recognized on a cash basis if the conditions for recognizing revenue on an accrual basis are not met. We delivered approximately 11,900 AlloMap results in 2014 and recognized revenue for approximately 9,700 tests; approximately 1,100 of which were for test results delivered prior to 2014.
For new diagnostic solutions, each private and government payer decides whether to cover the test, the amount it will reimburse for a covered test and the specific conditions for reimbursement. Clinicians and recipients may be likely not to order a diagnostic test unless third-party payers pay a substantial portion of the test price. Therefore,
34
Table of Contents
coverage determinations and reimbursement levels and conditions are critical to the commercial success of a diagnostic product, and if we are not able to secure positive coverage determinations and reimbursement levels, our business will be materially adversely affected.
Coverage and reimbursement by a commercial payer may depend on a number of factors, including a payer’s determination that our current and future solutions are:
| • | not experimental or investigational; |
| • | medically necessary; |
| • | appropriate for the specific recipient; |
| • | cost-saving or cost-effective; and |
| • | supported by peer-reviewed publications. |
In addition, several payers and other entities conduct technology assessments of new medical tests and devices and provide the results of their assessments for informational purposes to other parties. These assessments may be used by third-party payers and healthcare providers as grounds to deny coverage for or refuse to use a test or procedure. We believe we have received a negative technology assessment from at least one of these entities and could receive more.
If third-party payers decide not to cover our diagnostic solutions or if they offer inadequate payment amounts, our ability to generate revenue from AlloMap and future solutions could be limited. Payment for diagnostic tests furnished to Medicare beneficiaries is typically made based on a fee schedule set by the Centers for Medicare & Medicaid Services, or CMS. In recent years, payments under these fee schedules have decreased and may decrease further. Any third-party payer may stop or lower payment at any time, which could substantially reduce our revenue.
Since each payer makes its own decision as to whether to establish a policy to reimburse for a test, seeking payer coverage and other approvals is a time-consuming and costly process. We cannot assure you that adequate coverage and reimbursement for AlloMap or future solutions will be provided in the future by any third-party payer.
Reimbursement for AlloMap comes primarily from Medicare, private third party payers such as insurance companies and managed care organizations, Medicaid and hospitals. The reimbursement process can take six months or more to complete depending on the payer. As of December 31, 2014, we had been reimbursed for approximately 79% of AlloMap results delivered in the twelve months ended June 30, 2014. Coverage policies approving AlloMap have been adopted by many of the largest private payers, including Aetna, Cigna, Humana, Inc., Kaiser Foundation Health Plan, Inc., WellPoint, and a number of state Medicaid programs. Many of the payers with positive coverage policies have also entered into contracts with us to formalize pricing and payment terms. We continue to work with third-party payers to seek such coverage and to appeal denial decisions based on existing and ongoing studies, peer reviewed publications, support from physician and patient groups and the growing number of AlloMap tests that have been reimbursed by public and private payers. There are no assurances that the current policies will not be modified in the future. If our test is considered on a policy-wide level by major third-party payers, whether at our request or on their own initiative, and our test is determined to be ineligible for coverage and reimbursement by such payers, our collection efforts and potential for revenue growth could be adversely impacted.
Our Medicare Part B coverage for AlloMap is included in a formal local coverage decision for molecular diagnostics; however, any change in this coverage decision or other future adverse coverage decisions by the Centers for Medicare & Medicaid Services, or CMS, including with respect to coding, could substantially reduce our revenue.
Medicare reimbursements currently comprise a significant portion of our revenue. Our current Medicare Part B reimbursement was not set pursuant to a national coverage determination by CMS. Although we believe that
35
Table of Contents
coverage is available under Medicare Part B even without such a determination, we currently lack the national coverage certainty afforded by a formal coverage determination by CMS. This means that Medicare contractors, including our California Medicare contractor, currently may continue to develop their own coverage and reimbursement policies with respect to our technology.
Decisions by CMS with respect to coding could also affect our revenue. For example, on September 25, 2013, CMS released the preliminary payment determinations for the Clinical Laboratory Fee Schedule, or CLFS, for 2014. CMS proposed to not recognize certain Current Procedural Terminology codes, or CPT codes, called Multianalyte Assays with Algorithmic Analyses codes, or MAAA codes, as valid for Medicare purposes under the CLFS because it determined that an algorithm is not a clinical diagnostic test. This preliminary determination would have reversed a CMS final determination released on November 6, 2012 for 2013 that withdrew a proposal to not cover algorithmic analysis and stated that laboratories performing MAAA tests for Medicare beneficiaries should continue to bill for these tests in 2013 as they were then billed under the CLFS. When the final payment determination for 2014 was issued, CMS stated instead that it will continue to consider each test classified by the CPT as a MAAA on its own merits, and payment amounts would be determined using a gapfilling methodology if the Medicare contractor determines the code is payable.
AlloMap has been billed since the inception of the test using an unlisted CPT code. The test also has been granted a second code through a Medicare program for molecular diagnostics, which is included on all Medicare claims. If AlloMap is assigned a different MAAA CPT code in the future, a determination not to pay for such MAAA CPT code could be harmful to our business, and could have negative spillover implications that prevent or limit coverage by other third-party payers that might mirror aspects of Medicare payment criteria. Reimbursement for AlloMap under an MAAA code could also be lower than that currently received when AlloMap is billed under a miscellaneous CPT code.
We receive a substantial portion of our revenues from Medicare, and the loss of, or a significant reduction in, reimbursement from Medicare would adversely affect our financial performance
To date, we have received a substantial portion of our revenues from Medicare, which has been paid by our California Medicare Administrative Contractor. Payments from Medicare for AlloMap represented approximately 51% of testing revenue for the year ended December 31, 2014 and approximately 53% of testing revenue for the year ended December 31, 2013. We anticipate that Medicare will continue to be the payer for a significant portion of our claims for the foreseeable future. However, we may not be able to maintain or increase our rate of reimbursement by Medicare for a variety of reasons, including:
| • | changes in the local Medicare Administrative Contractor, or MAC, servicing our jurisdiction, which may result in a change in reimbursement practices for Medicare claims submitted by us or others in California and other states affected by the change; |
| • | any policy level review of our test by the CMS contractors could result in a reduction or denial of coverage and payment for our test; and |
| • | the assignment of a specific billing code to our test by CMS may result in reductions in the per test amount reimbursed for our current and future solutions by Medicare. |
On a five-year rotational basis, Medicare requests bids for its regional MAC services. Medicare reimbursement for AlloMap began in 2006 and has continued through three successive MACs. The MAC for California is currently Noridian Healthcare Solutions. Our current Medicare coverage through Nordian provides for reimbursement for tests performed for qualifying Medicare patients throughout the U.S. so long as the tests are performed in our California laboratory. We cannot predict whether Noridian or any future MAC will continue to provide reimbursement for AlloMap at the same payment amount or with the same breadth of coverage in the future, if at all. Additional changes in the MAC processing Medicare claims for AlloMap could impact the coverage or payment amount for our test and our ability to obtain Medicare coverage for any products we may launch in the future.
36
Table of Contents
Any decision by CMS or its local contractors to reduce or deny coverage for our test could have a significant adverse effect on our revenue and results of operations. Any such decision could also cause affected clinicians treating Medicare covered patients to reduce or discontinue the use of our test.
The assignment of a CPT code to AlloMap could adversely affect future payments for clinical laboratory testing services, including AlloMap and our future solutions.
Currently, AlloMap is paid under a non-specific billing code, which means there is no specific CPT code for our test and therefore, no established payment for the test under the Clinical Laboratory Fee Schedule. The local Medicare contractor processing our claims determines the amount of payment for the tests we bill. If the test is classified under a specific billing code, the payment amount established under the Clinical Laboratory Fee Schedule would be the basis for Medicare payment for AlloMap. We may in the near future apply for a unique CPT code for AlloMap, which would likely take one or more years to be considered and, if granted, would likely result in a change in our reimbursed amount. At this time, we cannot predict whether the classification of AlloMap under a billing code subject to the fee schedule would result in a lower payment amount.
In addition, it is possible that competitive bidding will be applied more broadly to clinical laboratory services under Medicare at some point in the future, which would impact payment for AlloMap. If a competitive bidding program is implemented and includes AlloMap, and if comparable solutions are identified, we may experience a decrease in our reimbursement rates for our clinical laboratory solutions.
Billing complexities associated with obtaining payment or reimbursement for our current and future solutions may negatively affect our revenue, cash flow and profitability.
Billing for clinical laboratory testing services is complex. In cases where we do not have a contract in place requiring the payment of a fixed fee per test, we perform tests in advance of payment and without certainty as to the outcome of the billing process. In cases where we do receive a fixed fee per test, we may still have disputes over pricing and billing. We receive payment from individual recipients and from a variety of payers, such as commercial insurance carriers and governmental programs, primarily Medicare. Each payer typically has different billing requirements. Among the factors complicating our billing of third-party payers are:
| • | disputes among payers regarding which party is responsible for payment; |
| • | disparity in coverage among various payers; |
| • | different process, information and billing requirements among payers; and |
| • | incorrect or missing billing information, which is required to be provided by the prescribing clinician. |
Additionally, from time to time, payers change processes that may affect timely payment. These changes may result in uneven cash flow or impact the timing of revenue recognized with these payers. With respect to payments received from governmental programs, factors such as a prolonged government shutdown could cause significant regulatory delays or could result in attempts to reduce payments made to us by federal government healthcare programs. These billing complexities, and the related uncertainty in obtaining payment for AlloMap and future solutions, could negatively affect our revenue, cash flow and profitability.
Healthcare reform measures could hinder or prevent the commercial success of AlloMap.
The pricing and reimbursement environment may change in the future and become more challenging as a result of any of several possible regulatory developments, including policies advanced by the U.S. government, new healthcare legislation or fiscal challenges faced by government health administration authorities. Specifically, there have been a number of legislative and regulatory proposals and initiatives to change the healthcare system in ways that could affect our ability to profitably sell any diagnostic products we may develop and commercialize. Some of these proposed and implemented reforms could result in reduced reimbursement rates for our diagnostic products from governmental agencies or other third-party payers, which would adversely
37
Table of Contents
affect our business strategy, operations and financial results. For example, as a result of the Patient Protection and Affordable Care Act of 2010 (as amended by the Health Care and Education Reconciliation Act of 2010), or the Affordable Care Act, substantial changes could be made to the current system for paying for healthcare in the U.S., including changes made in order to extend medical benefits to those who currently lack insurance coverage. Beginning in 2013, each medical device manufacturer must pay an excise tax in an amount equal to 2.3% of the price for which such manufacturer sells its medical devices that are listed with the FDA. While we do not believe that we are subject to this excise tax, the FDA has contended that clinical laboratory tests that are developed and validated by a laboratory for its own use, or LDTs, such as our proprietary tests, are medical devices. AlloMap is currently listed with the FDA. We cannot assure you that the tax will not be extended to services such as ours in the future. The Affordable Care Act also provides that payments under the Medicare Clinical Laboratory Fee Schedule are to receive a negative 1.75% annual adjustment through 2015. Although we have not been subject to such adjustment in the past, we cannot assure you that the claims administrators will not attempt to apply this adjustment in the future.
Among other things, the Affordable Care Act includes payment reductions to Medicare Advantage plans. These cuts have been mitigated in part by a CMS demonstration program set to expire in 2015. We cannot be assured that future cuts would be mitigated by CMS. Any reductions in payment to Medicare Advantage plans could materially impact coverage and reimbursement for AlloMap.
In addition to the Affordable Care Act, various healthcare reform proposals have also emerged from federal and state governments. For example, in February 2012, Congress passed the “Middle Class Tax Relief and Job Creation Act of 2012” which in part reduced the potential future cost-based increases to the Medicare Clinical Laboratory Fee Schedule, or CLFS, by 2%. The Protecting Access to Medicare Act of 2014 introduced a multi-year pricing program for services paid under the CLFS. Under the program, beginning in 2016 laboratories will report to CMS the payment rates paid to the laboratories by commercial third-party payors including Medicare and Medicaid managed care plans, for each test and the volume of each test performed. CMS will use the reported data to set new payment rates under the CLFS in the future. For newly developed tests that are considered to be “advanced diagnostic lab tests,” the Medicare payment rate will be the actual list price offered to third-party payors for the first three quarters that the tests are offered, subject to later adjustment. CMS will establish subsequent payment rates using the commercial third-party payor data reported for those tests.
Regardless of the impact of the Affordable Care Act on us, the government has shown significant interest in pursuing healthcare reform and reducing healthcare costs. Any government-adopted reform measures could decrease the amount of reimbursement available from governmental and other third-party payers. Additionally, annual federal budget negotiations frequently include healthcare payment reform measures impacting clinical laboratory payments. On April 1, 2013, cuts to the federal budget resulting from sequestration were implemented, requiring a 2% cut in Medicare payment for all services, including AlloMap. Federal budgetary limitations and changes in healthcare policy, such as the creation of broad limits for diagnostic products or requirements that Medicare patients pay for portions of clinical laboratory tests or services received, could substantially diminish the sale, or inhibit the utilization, of AlloMap and our future diagnostic solutions, increase costs, divert management’s attention and adversely affect our ability to generate revenue and achieve profitability.
38
Table of Contents
Healthcare Regulatory Risks
In order to operate our laboratory, we have to comply with the Clinical Laboratory Improvement Amendments of 1988, or CLIA, and state laws governing clinical laboratories.
We are subject to CLIA, a federal law that regulates clinical laboratories that perform testing on specimens taken from humans for the purpose of providing information for the diagnosis, prevention or treatment of disease. If our laboratory is out of compliance with CLIA requirements, we may be subject to sanctions such as suspension, limitation or revocation of our CLIA certificate, as well as a direct plan of correction, state on-site monitoring, civil money penalties, civil injunctive suit or criminal penalties. We must maintain CLIA compliance and certification to be eligible to bill for services provided to Medicare beneficiaries. If we were to be found to be out of compliance with CLIA program requirements and subjected to sanction, our business could be materially harmed.
In addition to federal certification requirements of laboratories under CLIA, licensure is required and maintained for our laboratory under California law. We are required to maintain a license to conduct testing in California. California laws establish standards for day-to-day operation of our clinical laboratory, including the training and skills required of personnel and quality control. Moreover, several states, including New York, require that we hold licenses to test specimens from patients residing in those states. Other states have similar requirements or may adopt similar requirements in the future. In addition to our California certifications, we currently hold licenses in Florida, Maryland, New York, and Pennsylvania. The loss of any of these state certifications would impact our ability to provide services in those states, which could negatively affect our business. Finally, we may be subject to regulation in foreign jurisdictions where we offer our test. Failure to maintain certification in those states or countries where it is required could prevent us from testing samples from those states or countries, could lead to the suspension or loss of licenses, certificates or authorizations, and could have an adverse effect on the Company’s business.
We were inspected and recertified under CLIA in February 2014 and we expect the next regular inspection under CLIA to occur in 2016. If we were to lose our CLIA accreditation or California license, whether as a result of a revocation, suspension or limitation, we would no longer be able to perform AlloMap, which would limit our revenues and materially harm our business. If we were to lose our license in other states where we are required to hold licenses, we would not be able to test specimens from those states, which could also have a material adverse effect on our business.
If the FDA’s recently published draft guidance setting forth a comprehensive regulatory scheme for LDTs becomes final, we would incur substantial costs and delays associated with trying to obtain premarket clearance or approval for those solutions.
Clinical laboratory tests that are developed and validated by a laboratory for its own use are called laboratory-developed tests, or LDTs. The laws and regulations governing the marketing of diagnostic products for use as LDTs are extremely complex, and in many instances, there are no significant regulatory or judicial interpretations of these laws. For instance, while the FDA maintains that LDTs are subject to the FDA’s authority as diagnostic medical devices under the Federal Food, Drug, and Cosmetic Act, or FFDCA, the FDA has in the past generally exercised enforcement discretion with respect to most LDTs performed by CLIA-certified laboratories.
The FDA has traditionally chosen not to exercise its authority to regulate LDTs because it regulates the primary components in most laboratory-developed tests and because it believes that laboratories certified as high complexity under CLIA, such as ours, have demonstrated expertise and ability in test procedures and analysis. However, beginning in September 2006, the FDA issued draft guidance on a subset of LDTs known as “in vitro diagnostic multivariate index assays,” or IVDMIAs. According to the draft guidance, IVDMIAs do not fall within the scope of LDTs over which FDA has exercised enforcement discretion because such tests incorporate complex and unique interpretation functions which require clinical validation. We believed that AlloMap met the definition of IVDMIA set forth in the draft guidance document. As a result, we applied for and obtained, in
39
Table of Contents
August 2008, 510(k) clearance for AlloMap for marketing and sale as a test to aid in the identification of recipients with a low probability of moderate or severe rejection. A 501(k) submission is a premarketing submission made to the FDA. Clearance may be granted by the FDA if it finds the device or test provides satisfactory evidence pertaining to the claimed intended uses and indications for the device or test.
On July 31, 2014, the FDA notified Congress (as required by the Food and Drug Administration Safety and Innovation Act of 2012) of its intent to publish a proposed and comprehensive risk-based framework for the regulation of LDTs. The notice to Congress provides the anticipated details and proposed timing of the implementation of the draft guidance and regulatory framework, including the requirement for premarket review and approval for higher-risk LDTs, such as our planned cell-free DNA solutions for heart, kidney and other organs. Such guidance, if and when finalized, will significantly impact the timing, availability and reimbursement of our future products, and will require us to modify our business model in order to maintain compliance with these new laws. For our cell-free DNA test and all similar testing solutions, we will be required to conduct additional clinical trials to clinically validate our test, and submit to the FDA a pre-market approval application, (PMA), or 510(k) clearance application and obtain approval or clearance for the test. We cannot predict the ultimate timing or form of any FDA final guidance or regulation of LDTs, or when we must obtain regulatory approval or clearance for our LDT solutions. There can be no assurance that any of our solutions or additional uses of solutions for which we will seek clearance or approval in the future will be cleared or approved on a timely basis, or at all, nor can there be any assurance that labeling claims will be consistent with our current claims or adequate to support continued adoption of and reimbursement for our current and future solutions. Moreover, any new FDA requirements could conflict with CLIA requirements and thereby complicate our compliance efforts.
While we believe that we are currently in material compliance with applicable laws and regulations relating to LDTs, we cannot assure you that the FDA or other regulatory agencies would agree with our determination. A determination that we have violated these laws, or a public announcement that we are being investigated for possible violation of these laws, could hurt our business and our reputation.
If we were required to conduct additional clinical trials prior to marketing our solutions under development, those trials could lead to delays or a failure to obtain necessary regulatory approvals and harm our ability to be profitable.
If the FDA decides to regulate our solutions under development as medical devices, it could require extensive premarket clinical testing prior to submitting a regulatory application for commercial sales. Conducting clinical trials generally entails a long, expensive and uncertain process that is subject to delays and failure at any stage. If we are required to conduct premarket clinical trials, whether using prospectively acquired samples or archival samples, delays in the commencement or completion of clinical testing could significantly increase our development costs and delay test commercialization. Many of the factors that may cause or lead to a delay in the commencement or completion of clinical trials may also ultimately lead to delay or denial of regulatory clearance or approval. The commencement of clinical trials may be delayed due to insufficient blood or tissue samples or insufficient data regarding the associated clinical outcomes. We may find it necessary to engage contract research organizations to perform data collection and analysis and other aspects of our clinical trials, which might increase the cost and complexity of our trials and reduce our control over such activities. If these parties do not successfully carry out their contractual duties or obligations or meet expected deadlines, or if the quality, completeness or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, applicable regulatory requirements, or for other reasons, our clinical trials may have to be extended, delayed or terminated. Our reliance on third parties that we do not control would not relieve us of any applicable requirement to ensure compliance with various procedures required under good clinical practices. We may not be able to enter into replacement arrangements without undue delays or considerable expenditures. If there are delays in testing or approvals as a result of the failure to perform by third parties, our research and development costs would increase, and we may not be able to obtain regulatory clearance or approval for our solutions under development. In addition, we may not be able to establish or maintain relationships with these parties on favorable terms, if at all. Each of these outcomes would harm our ability to market our solutions under development and our ability to be profitable.
40
Table of Contents
Any test for which we obtain regulatory clearance will be subject to extensive ongoing regulatory requirements, and we may be subject to penalties if we or our contractors or commercial partners fail to comply with regulatory requirements or if we experience unanticipated problems with our products.
AlloMap and our solutions under development, along with the manufacturing processes, packaging, labeling, distribution, import, export, and advertising and promotional activities for such solutions or devices, are or will be subject to continual requirements of, and review by, CMS, state licensing agencies, the FDA and comparable regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, registration and listing requirements, requirements relating to quality control, quality assurance and corresponding maintenance of records and documents, requirements relating to product labeling, advertising and promotion and recordkeeping. Regulatory clearance of a test or device may be subject to limitations by the regulatory body as to the indicated uses for which the product may be marketed or to other conditions of approval. For example, we are exploring utilization of AlloMap in areas that could be considered outside the scope of our current labeling. Broader uses would require FDA approval as well as changes to the labeling. In addition, approval may contain requirements for costly post-marketing testing and surveillance to monitor the safety or efficacy of the test or device. Discovery of previously-unknown problems with our current or future solutions, or failure to comply with regulatory requirements, may result in actions such as:
| • | restrictions on operations of our laboratory; |
| • | restrictions on manufacturing processes; |
| • | restrictions on marketing of a test; |
| • | warning or untitled letters; |
| • | withdrawal of the test from the market; |
| • | refusal to approve applications or supplements to approved applications that we may submit; |
| • | fines, restitution or disgorgement of profits or revenue; |
| • | suspension, limitation or withdrawal of regulatory clearances; |
| • | exclusion from participation in U.S. federal or state healthcare programs, such as Medicare and Medicaid; |
| • | refusal to permit the import or export of our products; |
| • | product seizure; |
| • | injunctions; and |
| • | imposition of civil or criminal penalties. |
We are subject to numerous fraud and abuse and other laws and regulations pertaining to our business, the violation of any one of which could harm our business.
The clinical laboratory testing industry is highly regulated, and there can be no assurance that the regulatory environment in which we operate will not change significantly and adversely in the future. Our arrangements with customers may expose us to broadly applicable fraud and abuse and other laws and regulations that may restrict the financial arrangements and relationships through which we market, sell and distribute our products. Our employees, consultants, principal investigators and commercial partners may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements. In addition to CLIA regulation, other federal and state healthcare laws and regulations that may affect our ability to conduct business, include, without limitation:
| • | federal and state laws and regulations regarding billing and claims payment applicable to clinical laboratories and/or regulatory agencies enforcing those laws and regulations; |
| • | federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented to the |
41
Table of Contents
| government, claims for payment from Medicare, Medicaid or other third-party payers that are false or fraudulent, or making a false statement material to a false or fraudulent claim; |
| • | the federal anti-kickback statute, which constrains our marketing practices, educational programs, pricing policies, and relationships with healthcare providers or other entities, by prohibiting, among other things, knowingly and willfully soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce or reward, or in return for, either the referral of an individual or the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
| • | the federal physician self-referral law, commonly known as the Stark Law, which prohibits a physician from making a referral to an entity for certain designated health services, including clinical laboratory services, reimbursed by Medicare if the physician (or a member of the physician’s family) has a financial relationship with the entity, and which also prohibits the submission of any claims for reimbursement for designated health services furnished pursuant to a prohibited referral; |
| • | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, and its implementing regulations, which imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information; HIPAA also created criminal liability for knowingly and willfully falsifying or concealing a material fact or making a materially false statement in connection with the delivery of or payment for healthcare benefits, items or services; |
| • | state laws regarding prohibitions on fee-splitting; |
| • | the federal healthcare program exclusion statute; and |
| • | state and foreign law equivalents of each of the above federal laws and regulations, such as anti-kickback, false claims, and self-referral laws, which may apply to items or services reimbursed by any third-party payer, including commercial insurers, and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
Because of the breadth of these laws and the narrowness of available statutory and regulatory exemptions, it is possible that some of our business activities could be subject to challenge under one or more of such laws. Any action brought against us for violation of these laws or regulations, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. We may be subject to private “qui tam” actions brought by individual whistleblowers on behalf of the federal or state governments, with potential liability under the federal False Claims Act including mandatory treble damages and significant per-claim penalties, currently set at $5,500 to $11,000 per false claim. If our operations are found to be in violation of any of the federal, state and foreign laws described above or any other current or future fraud and abuse or other healthcare laws and regulations that apply to us, we may be subject to penalties, including significant criminal, civil, and administrative penalties, damages, fines, imprisonment for individuals, exclusion from participation in government programs, such as Medicare and Medicaid, injunctions, recall or seizure of products, total or partial suspension of production, denial or withdrawal of pre-marketing product approvals, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations. Any of the foregoing consequences could seriously harm our business and our financial results.
Foreign governments may impose reimbursement standards, which may adversely affect our future profitability.
When we market AlloMap and our solutions under development in foreign jurisdictions, we are subject to rules and regulations in those jurisdictions relating to our testing. In some foreign countries, including countries in the European Union, the reimbursement of our current and future solutions is subject to governmental control. In these countries, reimbursement negotiations with governmental authorities can take considerable time after the
42
Table of Contents
receipt of marketing approval for a test candidate. If reimbursement of our future solutions in any jurisdiction is unavailable or limited in scope or amount, or if reimbursement rates are set at unsatisfactory levels, we may be unable to, or decide not to, market our test in that jurisdiction.
Changes in healthcare policy could increase our costs and subject us to additional regulatory requirements that may interrupt commercialization of our current and future solutions.
Changes in healthcare policy could increase our costs, decrease our revenues and impact sales of and reimbursement for our current and future solutions. In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, or the Affordable Care Act, became law. This law substantially changes the way healthcare is financed by both governmental and private insurers, and significantly impacts our industry. The Affordable Care Act contains a number of provisions that are expected to impact our business and operations, some of which in ways we cannot currently predict, including those governing enrollment in federal healthcare programs, reimbursement changes and fraud and abuse, which will impact existing government healthcare programs and will result in the development of new programs.
In addition to the Affordable Care Act, there will continue to be proposals by legislators at both the federal and state levels, regulators and third-party payors to reduce costs while expanding individual healthcare benefits. Certain of these changes could impose additional limitations on the prices we will be able to charge for our current and future solutions or the amounts of reimbursement available for our current and future solutions from governmental agencies or third-party payors. While in general it is too early to predict specifically what effect the Affordable Care Act and its implementation or any future healthcare reform legislation or policies will have on our business, current and future healthcare reform legislation and policies could have a material adverse effect on our business and financial condition.
Risks Relating to Our Intellectual Property
Our competitive position depends on maintaining intellectual property protection.
Our ability to compete and to achieve and maintain profitability depends on our ability to protect our proprietary discoveries and technologies. We currently rely on a combination of patents, copyrights, trademarks, trade secrets, confidentiality agreements and license agreements to protect our intellectual property rights.
Our patent position for AlloMap is based on issued patents and patent applications disclosing identification of genes differentially expressed between activated and resting leukocytes and demonstration of correlation between gene expression patterns and specific clinical states and outcomes. Our strategy is to continue to broaden our intellectual property estate for AlloMap through the discovery and protection of gene expression patterns and their correlation with specific clinical states and outcomes, as well as the algorithms needed for clinical assessment.
As of December 31, 2014, we had 16 issued U.S. patents, one pending U.S. patent application, and three pending patent applications outside the United States related to autoimmunity and transplant rejection. We have five issued U.S. patents covering methods of diagnosing transplant rejection using 9 of the 11 informative genes measured in AlloMap. The expiration dates of these patents range from 2021 to 2024. In connection with our June 2014 acquisition of ImmuMetrix, we obtained an exclusive license from Stanford University to a patent relating to the diagnosis of rejection in organ transplant recipients using cfDNA. This patent has an expiration date of November 5, 2030. We have six issued U.S. patents covering a method of diagnosing or monitoring an autoimmune or chronic inflammatory disease, such as lupus, by detecting specific genes. While we have clinical samples and patents covering lupus diagnostics, we do not intend to actively pursue the lupus test opportunity. In cfDNA-based transplant diagnostics, we have submitted a provisional patent application to cover some of our initial research and development work in this field. There is no guarantee that the U.S. Patent and Trademark
43
Table of Contents
Office, or PTO, will approve this provisional application. We do not know what claims, if any, will be granted in our existing and future applications. Our patents and patents that we exclusively license from others address fields that are rapidly evolving, and, particularly with respect to cfDNA-based transplant diagnostics, it is possible that other patents have and will be granted to others that affect our ability to develop and commercialize our current and future solutions. If the reviewers of our patent applications at the PTO refuse our claims, we may not be able to sufficiently protect our intellectual property. Further, recent and future changes in the patent laws and regulations of the United States and other jurisdictions may require us to modify our patent strategy and could restrict our ability to obtain additional patents for our technology.
Our patents and the patents we exclusively license from others may be successfully challenged by third parties as being invalid or unenforceable. Third parties may independently develop similar or competing technology that avoids the patents we own or exclusively license. We cannot be certain that the steps we have taken will prevent the misappropriation and use of our intellectual property, particularly in foreign countries where the laws may not protect our proprietary rights as fully as in the United States.
The extent to which the patent rights of life sciences companies effectively protect their products and technologies is often highly uncertain and involves complex legal and factual questions for which important legal principles remain unresolved. No consistent policy regarding the proper scope of allowable claims of patents held by such companies has emerged to date in the United States. Various courts, including the United States Supreme Court, have rendered decisions that impact the scope of patentability of certain inventions or discoveries relating to diagnostic solutions or genomic diagnostics. These decisions generally stand for the proposition that inventions that recite laws of nature are not themselves patentable unless they have sufficient additional features that provide practical assurance that the processes are genuine inventive applications of those laws rather than patent drafting efforts designed to monopolize a law of nature itself. What constitutes a “sufficient” additional feature for this purpose is uncertain. This evolving case law in the United States may adversely impact our ability to obtain new patents and may facilitate third-party challenges to our existing owned and exclusively licensed patents.
Changes in either the patent laws or in interpretations of patent laws in the United States or other countries may diminish the value of our intellectual property rights. In particular, in September 2011, the United States Congress passed the Leahy-Smith America Invents Act, or the AIA, which became effective in March 2013. The AIA reforms United States patent law in part by changing the standard for patent approval for certain patents from a “first to invent” standard to a “first to file” standard and developing a post-grant review system. It is too early to determine what the effect or impact the AIA will have on the operation of our business and the protection and enforcement of our intellectual property. However, the AIA and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition. Patent applications in the United States and many foreign jurisdictions are not published until at least eighteen months after filing, and it is possible for a patent application filed in the United States to be maintained in secrecy until a patent is issued on the application. In addition, publications in the scientific literature often lag behind actual discoveries. We therefore cannot be certain that others have not filed patent applications that cover inventions that are the subject of pending applications that we own or exclusively license or that we or our licensors, as applicable, were the first to invent the technology (pre-AIA) or first to file (post-AIA). Our competitors may have filed, and may in the future file, patent applications covering technology that is similar to or the same as our technology. Any such patent application may have priority over patent applications that we own or exclusively license and, if a patent issues on such patent application, we could be required to obtain a license to such patent in order to carry on our business. If another party has filed a United States patent application covering an invention this is similar to, or the same as, an invention that we own or license, we or our licensors may have to participate in an interference or other proceeding in the PTO or a court to determine priority of invention in the United States, for pre-AIA applications and patents. For post-AIA applications and patents, we or our licensors may have to participate in a derivation proceeding to resolve disputes relating to inventorship. The costs of these proceedings could be substantial, and it is possible that such efforts would be
44
Table of Contents
unsuccessful, resulting in our inability to obtain or retain any United States patent rights with respect to such invention.
We may face intellectual property infringement claims that could be time-consuming and costly to defend and could result in our loss of significant rights and the assessment of treble damages.
We may in the future receive offers to license patents or notices of claims of infringement, misappropriation or misuse of other parties’ proprietary rights. We may also initiate claims to defend our intellectual property. Intellectual property litigation, regardless of outcome, is unpredictable, expensive and time-consuming, could divert management’s attention from our business and have a material negative effect on our business, operating results or financial condition. If there is a successful claim of infringement against us, we may be required to pay substantial damages (including treble damages if we were to be found to have willfully infringed a third party’s patent) to the party claiming infringement, develop non-infringing technology, stop selling our test or using technology that contains the allegedly infringing intellectual property or enter into royalty or license agreements that may not be available on acceptable or commercially practical terms, if at all. Our failure to develop non-infringing technologies or license the proprietary rights on a timely basis could harm our business. In addition, revising our current or future solutions to exclude any infringing technologies would require us to re-validate the test, which would be costly and time consuming. Also, we may be unaware of pending patent applications that relate to our current or future solutions. Parties making infringement claims on future issued patents may be able to obtain an injunction that would prevent us from selling our current or future solutions or using technology that contains the allegedly infringing intellectual property, which could harm our business.
If we are unable to protect or enforce our intellectual property rights effectively in all major markets, our business would be harmed.
Filing, prosecuting, defending and enforcing patents on all of our technologies and solutions throughout the world would be prohibitively expensive. As a result, we seek to protect our proprietary position by filing patent applications in the U.S. and in select foreign jurisdictions and cannot guarantee that we will obtain the patent protection necessary to protect our competitive position in all major markets. Competitors may use our technologies or solutions in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export infringing products to territories where we have patent protection but where enforcement is not as strong as that in the U.S. These products may compete with our current and future products in jurisdictions where we do not have any issued patents, and our patent claims or other intellectual property rights may not be effective or sufficient to prevent them from so competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, which could make it difficult for us to stop the infringement of our patents or the marketing of competing products in violation of our proprietary rights generally. The legal systems of certain countries make it difficult or impossible to obtain patent protection for diagnostic solutions. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and could divert our efforts and attention from other aspects of our business.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In addition to seeking patents for some of our technologies and solutions, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside scientific collaborators, contract manufacturers, consultants, advisors and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees and consultants that obligate them to assign to us
45
Table of Contents
any inventions developed in the course of their work for us. However, we cannot guarantee that we have executed these agreements with each party that may have or have had access to our trade secrets or that the agreements we have executed will provide adequate protection. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Monitoring unauthorized disclosure is difficult and we do not know whether the procedures we have followed to prevent such disclosure are, or will be adequate. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the U.S. may be less willing or unwilling to protect trade secrets. If any of the technology or information that we protect as trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to, or independently developed by, a competitor, our competitive position would be harmed.
If our trademarks and trade names are not adequately protected, we may not be able to build name recognition in our markets of interest, and our business may be adversely affected.
AlloMap and CareDx are registered trademarks of our company in the United States. Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, declared generic or determined to be infringing on other marks. As a means to enforce our trademark rights and prevent infringement, we may be required to file trademark claims against third parties or initiate trademark opposition proceedings. This can be expensive, particularly for a company of our size, and time-consuming. In addition, in an infringement proceeding, a court may decide that a trademark of ours is not valid or is unenforceable, or may refuse to stop the other party from using the trademark at issue. We may not be able to protect our rights to these and other trademarks and trade names which we need to build name recognition by potential partners or customers in our markets of interest. Over the long-term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected.
We may be subject to claims by third parties that we or our employees have wrongfully used or disclosed alleged trade secrets or misappropriated intellectual property, or claiming ownership of what we view as our own intellectual property.
As is commonplace in our industry, we employ individuals who were previously employed at other diagnostics, medical device, life sciences or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees do not use the proprietary information of others in the course of their work for us and no claims against us are currently pending, we may be subject to claims that these employees have inadvertently or otherwise used or disclosed trade secrets or other proprietary information of their former employers. Litigation may be necessary to defend against these claims. We may also be forced to bring claims against third parties or defend against third-party claims in order to determine the ownership of our intellectual property. An adverse result in the prosecution or defense of any such claims could require us to pay substantial monetary damages and could result in the loss of valuable intellectual property rights or personnel. Even if we are successful in prosecuting or defending against these claims, litigation could result in substantial costs and be a distraction to management.
Our business is dependent on licenses from third parties.
We license from third parties technology necessary to develop and commercialize our products. Our most significant license covers PCR technology used in AlloMap and may be required for future solutions we develop. We license this technology from Roche Molecular Systems, Inc. In connection with our acquisition of ImmuMetrix, Inc., we obtained an exclusive license from Stanford University to a patent relating to the diagnosis of rejection in organ transplant recipients using cfDNA. Our rights to use these and other licensed technologies, data and materials and to employ the inventions claimed in licensed patents are subject to the continuation of and
46
Table of Contents
our compliance with the terms of the applicable licenses. We are obligated under these licenses to, among other things, pay certain royalties upon commercial sales of our products. These licenses generally last until the expiration of the last to expire of the patents included within the licenses that cover our use within our products, but the licenses may be terminated earlier in certain circumstances. Termination of any of these licenses could prevent us from producing or selling some or all of our products, and a failure of the licensors to abide by the terms of the licenses or to prevent infringement by third parties could harm our business and negatively impact our market position. Failure of a licensor to abide by the terms of a license or to prevent infringement by third parties could also harm our business and negatively impact our market position.
Risks Related to Our Common Stock
Our operating results may fluctuate, which could cause our stock price to decrease.
Fluctuations in our operating results may lead to fluctuations, including declines, in our share price. Our operating results and our share price may fluctuate from period to period due to a variety of factors, including:
| • | demand by clinicians and recipients for our current and future solutions, if any; |
| • | coverage and reimbursement decisions by third-party payers and announcements of those decisions; |
| • | clinical trial results and publication of results in peer-reviewed journals or the presentation at medical conferences; |
| • | the inclusion or exclusion of our current and future solutions in large clinical trials conducted by others; |
| • | new or less expensive tests and services or new technology introduced or offered by our competitors or us; |
| • | the level of our development activity conducted for new solutions, and our success in commercializing these developments; |
| • | the level of our spending on test commercialization efforts, licensing and acquisition initiatives, clinical trials, and internal research and development; |
| • | changes in the regulatory environment, including any announcement from the FDA regarding its decisions in regulating our activities; |
| • | changes in recommendations of securities analysts or lack of analyst coverage; |
| • | failure to meet analyst expectations regarding our operating results; |
| • | additions or departures of key personnel; and |
| • | general market conditions. |
Variations in the timing of our future revenues and expenses could also cause significant fluctuations in our operating results from period to period and may result in unanticipated earning shortfalls or losses. In addition, national stock exchanges in general, and the market for life science companies in particular, have experienced significant price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of those companies. In addition, we may be subject to additional securities class action litigation as a result of volatility in the price of our common stock, which could result in substantial costs and diversion of management’s attention and resources and could harm our stock price, business, prospects, results of operations and financial condition.
47
Table of Contents
The market price of our common stock has been and will likely continue to be volatile, and you could lose all or part of your investment.
Prior to our initial public offering in July 2014, there had been no public market for our shares of common stock. Our stock is currently traded on the NASDAQ Global Market, but we can provide no assurances that there will be active trading on that market or on any other market in the future. If there is no active market or if the volume of trading is limited, holders of our common stock may have difficulty selling their shares. The market price of our common stock has been and may continue to be subject to wide fluctuations in response to various factors, some of which are beyond our control. In addition to the factors discussed in this “Risk Factors” section and elsewhere in the Annual Report on Form 10-K, factors that could cause fluctuations in the market price of our common stock include the following:
| • | price and volume fluctuations in the overall stock market from time to time; |
| • | volatility in the market prices and trading volumes of life sciences stocks; |
| • | changes in operating performance and stock market valuations of other life sciences companies generally, or those in our industry in particular; |
| • | sales of shares of our common stock by us or our stockholders; |
| • | failure of securities analysts to maintain coverage of us, changes in financial estimates by securities analysts who follow our company, or our failure to meet these estimates or the expectations of investors; |
| • | the financial projections we may provide to the public, any changes in those projections or failure to meet those projections; |
| • | announcements by us or our competitors of new products or services; |
| • | the public’s reaction to our press releases, other public announcements and filings with the SEC; |
| • | rumors and market speculation involving us or other companies in our industry; |
| • | actual or anticipated changes in our operating results or fluctuations in our operating results; |
| • | actual or anticipated developments in our business, our competitor’s businesses or the competitive landscape generally; |
| • | litigation involving us, our industry or both, or investigations by regulators into our operations or those of our competitors; |
| • | developments or disputes concerning our intellectual property or other proprietary rights; |
| • | announced or completed acquisitions of businesses or technologies by us or our competitors; |
| • | new laws or regulations or new interpretations of existing laws or regulations applicable to our business; |
| • | changes in accounting standards, policies, guidelines, interpretations or principles; |
| • | any significant change in our management; and |
| • | general economic conditions and slow or negative growth of our markets. |
If our principal stockholders, executive officers and directors choose to act together, they may be able to control our management and operations, which may prevent us from taking actions that may be favorable to you.
Our executive officers, directors and principal stockholders, and entities affiliated with them, beneficially own in the aggregate approximately 42.3% of our common stock as of December 31, 2014. This significant concentration of share ownership may adversely affect the trading price of our common stock because investors often perceive disadvantages in owning stock in companies with controlling stockholders. These stockholders, acting together, will have the ability to exert substantial influence over all matters requiring approval by our stockholders, including the
48
Table of Contents
election and removal of directors and any proposed merger, consolidation or sale of all or substantially all of our assets. In addition, they could dictate the management of our business and affairs. This concentration of ownership could have the effect of delaying, deferring or preventing a change in control of us or impeding a merger or consolidation, takeover or other business combination that could be favorable to you.
Sales of substantial amounts of our common stock in the public markets, or sales of our common stock by our executive officers and directors under Rule 10b5-1 plans, could adversely affect the market price of our common stock.
Sales of a substantial number of shares of our common stock in the public market, or the perception that such sales could occur, could adversely affect the market price of our common stock and may make it more difficult for you to sell your common stock at a time and price that you deem appropriate. In addition, our executive officers and directors may adopt written plans, known as “Rule 10b5-1 Plans,” under which they will contract with a broker to sell shares of our common stock on a periodic basis to diversify their assets and investments. Sales made by our executive officers and directors pursuant to Rule 10b5-1, regardless of the amount of such sales, could adversely affect the market price of our common stock.
We incur costs and demands upon management as a result of complying with the laws and regulations affecting public companies in the U.S., which may adversely affect our operating results.
As a public company listed in the U.S., we incur significant additional legal, accounting and other expenses. In addition, changing laws, regulations and standards relating to corporate governance and public disclosure, including regulations implemented by the SEC and the NASDAQ Global Market exchange may increase legal and financial compliance costs and make some activities more time-consuming. These laws, regulations and standards are subject to varying interpretations, and as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. We invest resources to comply with evolving laws, regulations and standards, and this investment may result in increased general and administrative expenses and a diversion of management’s time and attention from revenue-generating activities to compliance activities. If, notwithstanding our efforts to comply with new laws, regulations and standards, we fail to comply, regulatory authorities may initiate legal proceedings against us, and our business may be harmed.
Further, failure to comply with these laws, regulations and standards might also make it more difficult for us to obtain certain types of insurance, including director and officer liability insurance, and we might be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these events could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, on committees of our board of directors or as members of senior management.
If equity research analysts do not publish research or reports about our business, or if they issue unfavorable commentary or downgrade our common stock, the price of our common stock could decline.
The trading market for our common stock relies in part on the research and reports that equity research analysts publish about us and our business. We do not control these analysts or the content and opinions included in their reports. Securities analysts may elect not to provide research coverage of our common stock and such a lack of research coverage may adversely affect the market price of our common stock. The price of our stock could decline if one or more equity research analysts downgrade our stock or if those analysts issue other unfavorable commentary or cease publishing reports about us or our business. If one or more equity research analysts ceases coverage of our company, we could lose visibility in the market, which in turn could cause our stock price to decline.
49
Table of Contents
We do not expect to pay dividends in the foreseeable future. As a result, you must rely on stock appreciation for any return on your investment.
We do not anticipate paying cash dividends on our common stock in the foreseeable future. Any payment of cash dividends will also depend on our financial condition, results of operations, capital requirements and other factors and will be at the discretion of our board of directors. Accordingly, you will have to rely on capital appreciation, if any, to earn a return on your investment in our common stock. Furthermore, we may in the future become subject to contractual restrictions on, or prohibitions against, the payment of dividends.
If we are unable to substantially utilize our net operating loss carryforwards, our financial results could be harmed.
As of December 31, 2014, our net operating loss, or NOL, carryforward amounts for U.S. federal income and California tax purposes were $164 million and $126 million, respectively. Under Section 382 of the Internal Revenue Code, a corporation that undergoes an “ownership change” may be subject to limitations on its ability to utilize its pre-change NOLs to offset future taxable income. Ownership changes beyond these limits, as determined by the IRS, may limit our ability to utilize our NOLs in the future. Limitations imposed on our ability to utilize NOLs could cause U.S. federal and state income taxes to be paid earlier than would be paid if such limitations were not in effect and could cause such NOLs to expire unused, in each case reducing or eliminating the benefit of such NOLs. Furthermore, we may not be able to generate sufficient taxable income to utilize our NOLs before they expire. If any of these events occur, we may not derive some or all of the expected benefits from our NOLs.
Our financial controls and procedures may not be sufficient to ensure timely and reliable reporting of financial information, which, as a public company, could materially harm our stock price and exchange listing.
As a public reporting company, we will be required to comply with the Sarbanes-Oxley Act of 2002 and the related rules and regulations of the Securities and Exchange Commission, including expanded disclosures and accelerated reporting requirements and more complex accounting rules. Compliance with Section 404 of the Sarbanes-Oxley Act and other requirements will increase our costs and require additional management resources. We recently have been upgrading our finance and accounting systems, procedures and controls and will need to continue to implement additional finance and accounting systems, procedures and controls as we grow our business and organization and to satisfy new reporting requirements. If we are unable to complete the required Section 404 assessment as to the adequacy of our internal control over financial reporting, if we fail to maintain or implement adequate controls, or if our independent registered public accounting firm is unable to provide us with an unqualified report as to the effectiveness of our internal control over financial reporting as of the date of our first Form 10-K for which compliance is required, our ability to obtain additional financing could be impaired.
We previously identified material weaknesses in our internal control over financial reporting related to an entity acquired in 2014. We determined that we did not design and maintain effective controls related to certain process-level activities of the newly acquired entity that contributed to material weaknesses related to our proposed purchase accounting that required adjustments to valuation of in-process technology, deferred income tax liability related to acquire in-process technology, goodwill, share-based compensation and recording of transaction cost. Subsequently, we implemented numerous measures designed to improve our internal control over financial reporting. Although, we believe the material weaknesses above have been remediated, these new controls may become inadequate because of changes in conditions or the degree of compliance with these policies or procedures may deteriorate. In addition, investors could lose confidence in the reliability of our internal control over financial reporting and in the accuracy of our periodic reports filed under the Securities Exchange Act. A lack of investor confidence in the reliability and accuracy of our public reporting could cause our stock price to decline.
As a public company, we will require greater financial resources than we have had as a private company. We cannot provide you with assurance that our finance department has or will maintain adequate resources to ensure
50
Table of Contents
that we will not have any future material weaknesses in our system of internal controls, including with respect to business combinations processes. The effectiveness of our controls and procedures may in the future be limited by a variety of factors, including:
| • | faulty human judgment and simple errors, omissions or mistakes; |
| • | fraudulent action of an individual or collusion of two or more people; |
| • | inappropriate management override of procedures; and |
| • | the possibility that any enhancements to controls and procedures may still not be adequate to assure timely and accurate financial information. |
If we fail to have effective controls and procedures for financial reporting in place, we could be unable to provide timely and accurate financial information and may be subject to NASDAQ Global Market delisting, SEC investigation and civil or criminal sanctions.
Our organizational documents and Delaware law make a takeover of our company more difficult, which may prevent certain changes in control and limit the market price of our common stock.
Our certificate of incorporation and bylaws and Section 203 of the Delaware General Corporation Law contain provisions that may have the effect of deterring or delaying attempts by our stockholders to remove or replace management, engage in proxy contests and effect changes in control. These provisions include:
| • | our board of directors will be authorized, without prior stockholder approval, to create and issue preferred stock which could be used to implement anti-takeover devices; |
| • | advance notice will be required for director nominations or for proposals that can be acted upon at stockholder meetings; |
| • | our board of directors will be classified such that not all members of our board are elected at one time, which may make it more difficult for a person who acquires control of a majority of our outstanding voting stock to replace all or a majority of our directors; |
| • | stockholder action by written consent will be prohibited; |
| • | special meetings of the stockholders will be permitted to be called only by the chairman of our board of directors, a majority of our board of directors or by our chief executive officer or president (if at such time we have no chief executive officer); |
| • | stockholders will not be permitted to cumulate their votes for the election of directors; and |
| • | stockholders will be permitted to amend our bylaws and certain provisions of our certificate of incorporation only upon receiving at least 66 2/3% of the votes entitled to be cast by holders of all outstanding shares then entitled to vote generally in the election of directors, voting together as a single class. |
In addition, as a Delaware corporation, we are subject to Delaware law, including Section 203 of the Delaware General Corporation Law. In general, Section 203 prohibits a Delaware corporation from engaging in any business combination with any interested stockholder for a period of three years following the date that the stockholder became an interested stockholder unless certain specific requirements are met as set forth in Section 203. These provisions, alone or together, could have the effect of deterring or delaying changes in incumbent management, proxy contests or changes in control.
These provisions also could discourage proxy contests and make it more difficult for you and other stockholders to elect directors and take other corporate actions. The existence of these provisions could limit the price that investors might be willing to pay in the future for shares of our common stock. Some provisions in our certificate
51
Table of Contents
of incorporation and bylaws may deter third parties from acquiring us, which may limit the market price of our common stock.
We are an “emerging growth company,” and, if we decide to comply only with reduced disclosure requirements applicable to emerging growth companies, our common stock could be less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act, or JOBS Act, enacted in April 2012, and for as long as we continue to be an “emerging growth company,” we may choose to take advantage of exemptions from various reporting requirements applicable to other public companies but not to “emerging growth companies,” including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We will continue to be an “emerging growth company” until the earlier of (1) the last day of the fiscal year (a) following the fifth anniversary of the completion of our initial public offering, (b) in which we have total annual gross revenue of at least $1.0 billion, or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeds $700 million as of the prior June 30th, and (2) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period. We cannot predict if investors will find our common stock less attractive if we choose to rely on these exemptions. If some investors find our common stock less attractive as a result of any choices to reduce future disclosure, there may be a less active trading market for our common stock, and our stock price may be more volatile.
Under the JOBS Act, emerging growth companies that become public can delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards, and therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
Our headquarters in Brisbane, California comprise approximately 46,000 square feet of leased space, which includes office space, our clinical laboratory and our research and development laboratories. The lease agreement for the Brisbane facility expires on December 31, 2020. We do not own any real property. We believe that our leased facilities are adequate to meet our current needs and that additional facilities are available for lease to meet future needs.
| ITEM 3. | LEGAL PROCEEDINGS |
We are not currently a party to any material legal proceedings that we believe are material to our business, financial condition or results of operations. We may from time to time become involved in legal proceedings arising in the ordinary course of business.
| ITEM 4. | MINE SAFETY DISCLOSURES |
Not applicable.
52
Table of Contents
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our common stock is traded on The NASDAQ Global Market under the symbol “CDNA” since July 22, 2014. Prior to that date, there was no public trading market for our common stock.
Holders of Record
As of February 27, 2015, there were approximately 202 holders of record of our common stock. Because many of our shares of common stock are held by brokers and other institutions on behalf of stockholders, we are unable to estimate the total number of stockholders represented by these record holders.
Price Range of Our Common Stock
The following table sets forth the high and low sales price per share of our common stock as reported on The NASDAQ Global Market for the period indicated:
| Year Ended December 31, 2014 |
High | Low | ||||||
| Fourth Quarter |
$ | 10.89 | $ | 5.40 | ||||
Stock Performance Graph
The following information is not deemed to be “soliciting material” or to be “filed” with the Securities and Exchange Commission for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (Exchange Act) or otherwise subject to the liabilities under that Section , and will not be deemed to be incorporated by reference into any filing under the Securities Act of 1933, as amended, or the Exchange Act, except to the extent we specifically incorporate it by reference into such a filing.
The graph and table below shows the cumulative total stockholder return on our common stock (change in stock price plus reinvested dividends) relative to the total returns of the NASDAQ Composite Index, and the NASDAQ Biotechnology Index. An investment of $100 is assumed to have been made in our common stock and in each index on July 22, 2014 (the date our common stock began trading following our initial public offering) and its relative performance is tracked through December 31, 2014. The comparison is based on historical results and is not intended to forecast or be indicative of future performance of our common stock.
53
Table of Contents
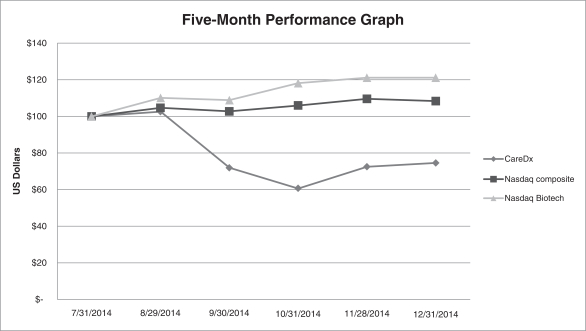
| Trade Date | CareDx, Inc. | Nasdaq Composite | Nasdaq Biotech | |||||||||
| 12/31/2014 |
$ | 74.67 | $ | 108.38 | $ | 121.12 | ||||||
| 11/28/2014 |
$ | 72.61 | $ | 109.65 | $ | 121.20 | ||||||
| 10/31/2014 |
$ | 60.56 | $ | 105.97 | $ | 118.27 | ||||||
| 9/30/2014 |
$ | 72.09 | $ | 102.83 | $ | 108.98 | ||||||
| 8/29/2014 |
$ | 102.68 | $ | 104.82 | $ | 110.26 | ||||||
| 7/31/2014 |
$ | 100.00 | $ | 100.00 | $ | 100.00 | ||||||
Dividend Policy
We have never declared or paid any cash dividend on our capital stock. We currently intend to retain any future earnings and do not expect to pay any dividends in the foreseeable future. Our loan and security agreement restricts our ability to pay cash dividends on our common stock, and we may also enter into credit agreements or other borrowing arrangements in the future that will further restrict our ability to declare or pay cash dividends on our common stock. Any determination to declare or pay dividends in the future will be at the discretion of our board of directors and will depend on a number of factors, including our financial condition, operating results, capital requirements, contractual restrictions, general business conditions and other factors that our board of directors may deem relevant.
Sales of Unregistered Securities
From January 1, 2014 through July 18, 2014 (the date of the filing of our registration statement on Form S-8, File No. 333-197493), we granted stock options to our directors, officers, employees, consultants and other service providers under our 2008 Equity Incentive Plan to purchase 458,215 shares of our common stock at an exercise price of $12.3985 per share, and issued an aggregate of 2,613 shares of common stock pursuant to the exercise of stock options with aggregate proceeds of approximately $4,784.
On April 17, 2014, we issued and sold a subordinated convertible promissory note in the aggregate principal amount of $5,000,000 to Illumina, Inc. In connection with the closing of our initial public offering, this subordinated convertible promissory note converted into 510,777 shares of common stock at the initial public offering price per share of $10.00.
54
Table of Contents
On June 10, 2014, in connection with our acquisition of ImmuMetrix, Inc. we issued 888,135 shares of our Series G preferred stock to the former stockholders of ImmuMetrix, Inc. The shares converted into common stock upon the closing of our initial public offering on July 22, 2014. On June 10, 2014, in connection with our acquisition of ImmuMetrix, Inc., we assumed the ImmuMetrix, Inc. 2013 Equity Incentive Plan and all outstanding options thereunder, and converted them into options to purchase 23,229 shares of our common stock.
None of the above transactions involved any underwriters, underwriting discounts or commissions, or any public offering. We believe that the offers, sales and issuances of the above securities were exempt from registration under the Securities Act by virtue of Section 4(2) of the Securities Act (or Regulation D promulgated thereunder) as transactions not involving a public offering, or in reliance on Rule 701 promulgated under the Securities Act as transactions pursuant to compensatory benefit plans or contracts relating to compensation as provided under such rule. The recipients of the securities in each of these transactions represented their intentions to acquire the securities for investment purposes only and not with a view to or for sale in connection with any distribution thereof, and appropriate legends were placed upon the stock certificates or book-entry positions representing the shares issued in these transactions. All recipients had adequate access, through their relationships with us, to information about us. The sale of these securities were made without any general solicitation or advertising.
Use of Proceeds
On July 22, 2014, we completed our initial public offering (IPO) in which we sold 4,000,000 shares of our common stock at a price to the public of $10.00 per share. On August 13, 2014, the underwriters partially exercised their overallotment option, at which time we sold an additional 220,000 shares. We received net cash proceeds of $35.5 million from the IPO, including additional shares sold, after deducting underwriting discounts and commissions of $3.0 million and offering expenses of $3.8 million. The underwriters of that offering were Piper Jaffray, Leerink Partners, Raymond James and Mizuho Securities. No payments were made by us to directors, officers or other persons owning ten percent or more of our common stock or to their associates, or to our affiliates, other than payments in the ordinary course of business to officers for salaries.
We registered the shares under the Securities Act on a Registration Statement on Form S-1 (Registration No. 333-196494), which was declared effective on July 16, 2014.
There has been no material change in our planned use of proceeds from or IPO as described in the final prospectus filed with the SEC on July 18, 2014, pursuant to Rule 424(b).
Securities Authorized for Issuance Under Equity Compensation Plans
See Item 12 of Part III of this Annual Report on Form 10-K regarding information about securities authorized for issuance under our equity compensation plans.
Issuer Purchases of Equity Securities
There were no repurchases of equity securities by us during the fourth quarter of 2014.
55
Table of Contents
ITEM 6. SELECTED FINANCIAL DATA
The following selected historical financial data should be read in conjunction with “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our financial statements and related notes included elsewhere in this Annual Report on Form 10-K. The selected balance sheet data at December 31, 2014 and 2013 and the selected statements of operations data for each of the years ended December 31, 2014, 2013 and 2012 have been derived from our audited financial statements that are included elsewhere in this Annual Report on Form 10-K. The financial data included in this report are historical and are not necessarily indicative of results to be expected in any future period.
Statements of Operations Data:
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| (In thousands, except share and per share data) | ||||||||||||
| Revenue: |
||||||||||||
| Testing revenue |
$ | 25,842 | $ | 21,672 | $ | 19,730 | ||||||
| Collaboration and license revenue |
1,464 | 426 | 721 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total revenue |
27,306 | 22,098 | 20,451 | |||||||||
| Operating expenses: |
||||||||||||
| Cost of testing |
8,541 | 9,078 | 7,930 | |||||||||
| Research and development |
3,846 | 3,176 | 4,752 | |||||||||
| Sales and marketing |
6,472 | 5,892 | 5,417 | |||||||||
| General and administrative |
8,436 | 4,809 | 4,694 | |||||||||
| Change in estimated fair value of contingent consideration |
(1,239 | ) | — | — | ||||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
26,056 | 22,955 | 22,793 | |||||||||
|
|
|
|
|
|
|
|||||||
| Income (loss) from operations |
1,250 | (857 | ) | (2,342 | ) | |||||||
| Interest expense, net |
(2,116 | ) | (2,149 | ) | (2,703 | ) | ||||||
| Other income (expense), net |
147 | (536 | ) | (14 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Loss before income taxes |
(719 | ) | (3,542 | ) | (5,059 | ) | ||||||
| Income tax benefit |
1,500 | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) |
$ | 781 | $ | (3,542 | ) | $ | (5,059 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) per share: |
||||||||||||
| Basic |
$ | 0.13 | $ | (3.50 | ) | $ | (5.01 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Diluted |
$ | 0.10 | $ | (3.50 | ) | $ | (5.01 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Shares used to compute net income (loss) per share: |
||||||||||||
| Basic |
5,815,928 | 1,010,795 | 1,009,236 | |||||||||
|
|
|
|
|
|
|
|||||||
| Diluted |
9,283,001 | 1,010,795 | 1,009,236 | |||||||||
|
|
|
|
|
|
|
|||||||
Balance Sheet Data:
| As of December 31, | ||||||||
| 2014 | 2013 | |||||||
| (In thousands) | ||||||||
| Cash and cash equivalents |
$ | 36,431 | $ | 5,128 | ||||
| Working capital |
29,211 | 578 | ||||||
| Total assets |
61,141 | 9,873 | ||||||
| Total debt |
11,412 | 15,375 | ||||||
| Convertible preferred stock |
— | 135,202 | ||||||
| Accumulated deficit |
(159,375 | ) | (160,156 | ) | ||||
| Total stockholders’ equity (deficit) |
41,297 | (150,673 | ) | |||||
56
Table of Contents
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion of our financial condition and results of operations should be read in conjunction with our financial statements and related notes included elsewhere in this Annual Report on Form 10-K. This discussion contains certain forward-looking statements that involve risk and uncertainties. Our actual results may differ materially from those discussed below. Factors that could cause or contribute to such differences include, but are not limited to, those identified below and those set forth under the Section entitled “Risk Factors” in Item 1A, and other documents we file with the Securities and Exchange Commission. Historical results are not necessarily indicative of future results.
Business Overview
We are a commercial stage company that develops, markets and delivers a diagnostic surveillance solution for heart transplant recipients to help clinicians make personalized treatment decisions throughout a patient’s lifetime. Our commercialized testing solution, the AlloMap heart transplant molecular test, is a blood-based test used to monitor heart transplant recipients for moderate or acute cellular rejection. We believe the use of AlloMap, in conjunction with other clinical indicators, can help healthcare providers and their patients better manage long-term care following a heart transplant. In particular, we believe AlloMap can improve patient care by helping healthcare providers to avoid the use of unnecessary, invasive surveillance biopsies and to determine the appropriate dosage levels of immunosuppressants. We believe that there is a significant unmet need for post-transplant surveillance solutions and are applying our expertise in transplantation towards the development of additional solutions for other organ transplant recipients, including recipients of kidney transplants.
Since the launch of AlloMap in January 2005 we have performed more than 66,000 commercial AlloMap tests, including more than 11,000 tests in 2014, in our Brisbane, California laboratory. In 2014, the test was used in 110 of the approximately 129 heart transplant management centers in the U.S. We believe that there is an opportunity for AlloMap outside of the U.S. and through recent partnerships we have expanded the AlloMap offering to Europe and Canada. We believe that we are not currently capacity constrained and that our current facility can support a substantial increase in AlloMap testing volume.
Reimbursement for AlloMap tests comes primarily from Medicare, private third party payers such as insurance companies and managed care organizations, hospitals and state Medicaid programs. Tests performed on patients covered by Medicare represented 37%, 39% and 40% of all AlloMap tests in 2014, 2013 and 2012, respectively. A number of payers have adopted coverage policies approving AlloMap tests for reimbursement. Such policies often approve reimbursement for tests performed from six-months or one year post-transplant through five years post-transplant. For tests performed outside the scope of the payer’s policy, and for tests performed where the payer has not adopted a coverage policy, we pursue reimbursement on a case-by-case basis. If a reimbursement claim is denied, we generally pursue the appeals process for the particular payer.
Since our inception, we have generated significant net losses. As of December 31, 2014, we had an accumulated deficit of $159 million. For the year ended December 31, 2014, we recognized net income of $0.8 million, and for the years ended December 31, 2013 and 2012, we incurred net losses of $3.5 million and $5.1 million,
57
Table of Contents
respectively. Together with our cash and cash equivalents and cash receipts from our AlloMap testing and from our collaboration and license agreements, we expect to be able to continue to accelerate the development of new transplant surveillance solutions, such as our planned cell-free DNA, or cfDNA, solution for heart and kidney, using both our proprietary and third party libraries of blood samples from multiple organs.
Financial Overview
Testing Revenue
Our testing revenue is derived from AlloMap tests which represented 95%, 98% and 97% of our total revenue in 2014, 2013 and 2012, respectively. Our testing revenue depends on a number of factors, including (i) the number of tests performed; (ii) establishment of coverage policies by third-party insurers and government payers; (iii) our ability to collect from payers with whom we do not have positive coverage determination, which often requires that we pursue a case-by-case appeals process; (iv) our ability to recognize revenues on tests billed prior to the establishment of reimbursement policies, contracts or payment histories; (v) our ability to expand into markets outside of the United States; and (vi) how quickly we can successfully commercialize new product offerings.
We currently market AlloMap to healthcare providers through our direct sales force that targets transplant centers and their physicians, coordinators and nurse practitioners. The healthcare providers that order the tests and on whose behalf we provide our testing services are generally not responsible for the payment of these services. As of December 31, 2014, the list price of AlloMap was $3,600 per test. However, amounts actually received by us vary from payer to payer based on each payer’s internal coverage practices and policies. We generally bill third-party payers upon delivery of an AlloMap score report to the ordering physician. As such, we take the assignment of benefits and the risk of collection from the third-party payer and individual patients.
As of December 31, 2014 and 2013, the number of tests for which results were delivered and billed, but for which the associated revenue had not been recognized because our revenue recognition criteria were not met, and taking into account claim status and possibility of collection, was approximately 3,500 and 3,900, respectively. We cannot provide any assurance as to when, if ever, or to what extent any of these amounts will be collected.
Collaboration and License Revenue
Revenue from our collaboration and license agreements was not more than 6% of total revenue for each period presented. Collaboration and license agreements may include non-refundable upfront payments, partial or complete reimbursement of research and development costs, contingent payments based on the occurrence of specified events under the agreements, license fees and royalties on sales of products or product candidates if they are successfully commercialized. Note 6 to our audited financial statements included elsewhere in this Annual Report includes descriptions of these agreements. Our performance obligations under the collaboration and license agreements may include the transfer of intellectual property rights in the form of licenses, obligations to provide research and development services and obligations to participate on certain development committees with the collaboration partners. We make judgments that affect the periods over which we recognize revenue. We periodically review our estimated periods of performance based on the progress under each arrangement and account for the impact of any change in estimated periods of performance on a prospective basis.
Cost of Testing
Cost of testing reflects the aggregate costs incurred in delivering our AlloMap test results to clinicians. The components of our cost of testing are materials and service costs, direct labor costs, including stock-based compensation, equipment and infrastructure expenses associated with testing samples, shipping, logistics and specimen processing charges to collect and transport samples and allocated overhead including rent, information technology, equipment depreciation and utilities and royalties. Costs associated with performing tests (except royalties) are recorded as the test is processed regardless of whether and when revenue is recognized
58
Table of Contents
with respect to that test. Royalties incurred for licensed technology, calculated as a percentage of test revenues, are recorded as license fees in cost of testing at the time the test revenues are recognized.
Royalties included in cost of testing are associated with a license from Roche Molecular Systems, Inc., or Roche. In February 2014, we received a demand for arbitration from Roche regarding our claim that the royalty rate being assessed under the Roche license should be reduced. In September 2014, we entered into a settlement and mutual release agreement with Roche whereby (i) for the period beginning July 1, 2011 through June 30, 2014, we agreed to pay the amount of $2.8 million in settlement of past royalties due; (ii) for the period beginning July 1, 2014 through September 30, 2014, we agreed to pay royalties based on the same combination services percentage used to determine the past royalties due; (iii) for the period beginning October 1, 2014 through September 30, 2017, we agreed to a downward adjustment of the combination services percentage used to determine the portion of the AlloMap service that is royalty bearing under the terms of the license; (iv) we agreed to report and pay quarterly royalties within 45 days of the end of each calendar quarter; (v) Roche agreed that, subject to our timely payment of all applicable royalties through such date, no further royalties will be payable by us for periods after September 30, 2017; and (v) mutual releases by the Company and Roche of all claims under the license agreement through the settlement date.
We expect cost of testing to increase, in absolute dollars, as the number of tests we perform increases. However, due to the fixed nature of expenses associated with direct labor, equipment and infrastructure, we expect the cost per test will decrease over time as volume increases.
Research and Development Expenses
Research and development expenses represent costs incurred to develop new surveillance solutions as well as continued efforts related to our AlloMap test. These expenses include payroll and related expenses, consulting expenses, laboratory supplies, and certain allocated expenses as well as amounts incurred under certain collaborative agreements. Research and development costs are expensed as incurred. We record accruals for estimated study costs comprised of work performed by contract research organizations under contract terms. We expect our research and development expenses will increase in absolute dollars in future periods as we invest in research and discovery work to develop new surveillance solutions, as well as clinical outcomes studies for AlloMap.
Sales and Marketing Expenses
Sales and marketing expenses represent costs incurred to sell, promote and increase awareness of our AlloMap test to both clinicians and payers, including education of patients, clinicians and payers. Sales and marketing expenses include payroll and related expenses, educational and promotional expenses, and infrastructure expenses, including allocated facility and overhead costs. Compensation related to sales and marketing includes annual salaries and eligibility for quarterly or semi-annual commissions or bonuses based on the achievement of predetermined sales goals or other management objectives.
General and Administrative Expenses
General and administrative expenses include costs for our executive, finance, accounting and human resources functions. Costs consist primarily of payroll and related expenses, professional service fees related to billing and collection, accounting, legal and other contract and administrative services and related infrastructure expenses, including allocated facility and overhead costs. We expect to incur additional expenses as a result of operating as a public company, including expenses related to compliance with the rules and regulations of the Securities and Exchange Commission and The NASDAQ Global Market, additional insurance expenses, investor relations activities and other administrative and professional services. We also expect our general and administrative expenses will increase in absolute dollars related to anticipated testing volume and collections growth.
59
Table of Contents
Interest Expense, Net
Interest expense, net is associated with borrowings under our loan agreements.
Other Income (Expense), Net
Other income (expense), net is primarily associated with the remeasurement of the estimated fair value of warrants to purchase shares of our convertible preferred stock and changes in the estimated fair value of a derivative associated with our subordinated convertible debt. Convertible preferred warrants and the subordinated convertible debt were converted to common stock warrants and common stock, respectively, upon the closing of our initial public offering on July 22, 2014.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of our financial condition and results of operation is based on our financial statements, which have been prepared in accordance with United States generally accepted accounting principles or U.S. GAAP. The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. Items subject to estimates based on judgments include, but are not limited to: revenue recognition, the valuation of warrants to purchase convertible preferred stock, the determination of the valuation allowance associated with deferred tax assets, the determination of the accruals for clinical studies, the determination of estimated refunds to be requested from third-party payers, any impairment of long-lived assets and legal contingencies. Actual results could differ from these estimates and such differences could affect the results of operations in future periods.
Our significant accounting policies are described in Note 2 to our audited financial statements included elsewhere in this Annual Report. Some of these accounting policies require us to make difficult and subjective judgments, often as a result of the need to make estimates of matters that are inherently uncertain. We believe that the following critical accounting policies reflect the more significant estimates and assumptions used in the preparation of our financial statements.
Revenue Recognition
Testing Revenue
We recognize revenues for tests delivered when the following criteria are met: (i) persuasive evidence that an arrangement exists, which may include a contract or a coverage policy; (ii) delivery has occurred or services rendered; (iii) the fee is fixed or determinable; and (iv) collectability is reasonably assured.
The first criterion is satisfied when a third-party payer makes a coverage decision or enters into a contractual arrangement with us for the test. The second criterion is satisfied when we perform the test and deliver the test result to the ordering physician. The third criterion is satisfied if the third-party payer’s coverage decision or reimbursement contract specifies a price for the test. The fourth criterion is satisfied based on management’s judgments regarding the collectability of the fees charged under the arrangement.
Such judgments include review of past payment history. AlloMap testing may be considered investigational by some payers and not covered under their reimbursement policies. Others may cover the test, but not pay a set or determinable amount. As a result, in the absence of a reimbursement agreement or sufficient payment history, collectability cannot reasonably be assured so revenue is not recognized at the time the test is delivered.
If all criteria set forth above are met, revenue is recognized. When the first, third or fourth criteria are not met but third-party payers make a payment to us for tests performed, we recognize revenue on a cash basis in the period in which the payment is received.
60
Table of Contents
Revenue is recognized on an accrual basis, net of adjustments for differences between amounts billed and the estimated receipts from payers. The amount we expect to collect may be lower than the agreed upon amount due to several factors, such as the amount of patient co-payments, the existence of secondary payers and claim denials. Estimated receipts are based upon historical payment practices of payers. Differences between estimated and actual cash receipts are recorded as an adjustment to revenue, which have been immaterial to date.
For tests performed where an agreed upon reimbursement rate and a predictable history of collection exists, such as in the case of Medicare, we recognize revenue upon delivery of a score report to the ordering physician based on the established billing rate less contractual and other adjustments to arrive at the amount that we expect to collect. We determine the amount we expect to collect based on a per payer, per contract or agreement basis, after analyzing historical payment trends. The expected amount is typically lower than the agreed upon reimbursement amount due to several factors, such as the amount of patient co-payments and claim denials. In all other situations, where we do not have sufficient history of collection and are unable to determine a predictable pattern of payment, we recognize revenue upon the receipt of cash. In 2014, 2013 and 2012, approximately 64%, 64% and 56%, respectively, of our testing revenue was recognized on the accrual basis.
Occasionally, we may receive requests from third-party payers for refunds for previously paid-for tests. We maintain a liability for actual overpayments and estimated future refund claims based on historical experience. Accruals for overpayments and refunds are recorded as a reduction of revenue. The approximate number of delivered AlloMap tests and AlloMap tests for which we recognized revenue in accordance with our revenue recognition policies discussed above, were as follows:
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| AlloMap tests delivered |
11,930 | 10,100 | 8,300 | |||||||||
| AlloMap tests for which revenue was recognized |
9,786 | 8,400 | 7,500 | |||||||||
| AlloMap tests for which revenue was recognized, delivered prior to the period presented |
1,172 | 1,100 | 1,800 | |||||||||
We did not recognize revenue for the remaining tests because either there was no contract, no coverage policy in place, insufficient payment history or we had not received payment for those tests from a payer. We will continue to make requests for payment from payers and patients and/or appeal payment decisions made by third-party payers. As a result, we may receive payment for a portion of these tests. However, a portion of our requests for payments could be denied or only partially satisfied. If third-party payers agree to pay us for these tests in the future, we will recognize revenue for such tests in the period in which all of our revenue recognition criteria are met. This will continue to affect the comparability of our revenues from period to period. We regularly review to determine if payers meet our revenue recognition criteria and account for the impact of any change on a prospective basis.
The process for determining the appropriate amount expected to be collected involves judgment, and considers such factors as, historical payment trends, current economic conditions and regulatory changes. The ultimate amounts of collections could be different from the amounts we estimate.
Collaboration and License Revenue
Revenue from our collaboration and license agreements was not more than 5% of total revenue for each period presented. Collaboration and license agreements may include non-refundable upfront payments, partial or complete reimbursement of research and development costs, contingent payments based on the occurrence of specified events under the agreements, license fees and royalties on sales of products or product candidates if they are successfully commercialized.
We recognize collaboration and license revenue based upon the relative-selling–price method which is used to allocate arrangement consideration to all of the units of accounting in an arrangement. We evaluate our collaboration and license agreements to identify the deliverables, determine if the deliverables have stand-alone
61
Table of Contents
value, to identify the units of accounting and to allocate arrangement consideration to each unit of accounting based on relative best estimate selling price.
Business Combinations
In accordance with ASC 805, Business Combinations, the Company determines and allocates the purchase price of an acquired business to the tangible and identifiable intangible assets acquired and liabilities assumed based on their estimated fair values as of the business combination date, including identifiable intangible assets which either arise from a contractual or legal right or are separable from goodwill. The Company bases the estimated fair value of identifiable intangible assets acquired in a business combination on independent valuations that use information and assumptions provided by management, which consider management’s best estimates of inputs and assumptions that a market participant would use.
The Company allocates any excess purchase price over the estimated fair value assigned to the net tangible and identifiable intangible assets acquired and liabilities assumed to goodwill. The use of alternative valuation assumptions, including estimated revenue projections, growth rates, royalty rates, cash flows, discount rates, estimated useful lives and probabilities surrounding the achievement of contingent milestones, could result in different purchase price allocations and amortization expense in current and future periods.
In those circumstances where an acquisition involves a contingent consideration arrangement that meets the definition of a liability under ASC 480, Distinguishing Liabilities from Equity, the Company recognizes a liability equal to the fair value of the contingent payments the Company expects to make as of the acquisition date. The Company remeasures this liability each reporting period and records changes in the fair value as a component of operating expenses.
Transaction costs associated with acquisitions are expensed as incurred in general and administrative expenses. Results of operations and cash flows of acquired companies are included in the Company’s operating results from the date of acquisition.
Warrant Liability
We have freestanding warrants enabling counterparties to purchase shares of our common stock, which prior to conversion upon our IPO were warrants to purchase shares of our convertible preferred stock. In accordance with the accounting guidance regarding distinguishing liabilities from equity, freestanding warrants for convertible preferred stock that are contingently redeemable are classified as liabilities on the balance sheets and recorded at their estimated fair value. Through the date of our IPO, these warrants were remeasured at each balance sheet date, and any change in estimated fair value was recognized in other income or expense on the statements of operations. We adjusted the liability for changes in estimated fair value until the completion of our IPO, at which time the remaining preferred stock warrants were converted into warrants to purchase common stock, and, accordingly, the liability was reclassified to equity. Upon the IPO, the then-current aggregate fair value of the warrants was reclassified from liabilities to additional paid-in capital, a component of stockholders’ equity, and we then ceased to record any further related periodic fair value adjustments.
62
Table of Contents
The estimated fair value of the convertible preferred stock warrant liability was determined using the Black-Scholes option pricing model using the following assumptions:
| As of December 31, 2013 |
||||
| Estimated fair value of common stock |
$ | 8.97 | ||
| Risk-free interest rate |
0.8% - 2.1% | |||
| Expected volatility |
40% - 45% | |||
| Estimated term equal to remaining contractual term |
3.3 - 5.6 years | |||
| Expected dividend yield |
— | |||
We recorded a benefit of $14,000 for the year ended December 31, 2014, and expense of $525,000 for the year ended December 31, 2013 to other income (expense), net on the statements of operations, to reflect changes in the estimated fair value of the preferred stock warrants.
Stock–Based Compensation
We recognize stock-based compensation cost for only those shares underlying stock options that we expect to vest on a straight-line basis over the requisite service period of the award. We estimate the fair value of stock options using a Black-Scholes valuation model, which requires the input of highly subjective assumptions, including the option’s expected term and stock price volatility. In addition, judgment is also required in estimating the number of stock-based awards that are expected to be forfeited. Forfeitures are estimated based on historical experience at the time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates. The assumptions used in calculating the fair value of share-based payment awards represent management’s best estimates, but these estimates involve inherent uncertainties and the application of management’s judgment. As a result, if factors change and we use different assumptions, our stock-based compensation expense could be materially different in the future.
Factors Affecting Our Performance
The Number of AlloMap Tests We Receive and Report
The growth of our business is tied to the number of AlloMap tests we receive and report. Historically, less than two percent of tests received are not reported due to improper sampling or damage in transit or other causes. We incur costs of collecting and shipping all samples and a portion of the costs where we cannot ultimately issue a score report. As a result, the number of samples received largely directly correlates to the number of score reports.
How We Recognize Revenue
Medicare and certain other payers with agreed upon reimbursement rates and a predictable history of collections allows us to recognize the related revenue on an accrual basis. In 2014, 2013 and 2012, 36%, 36% and 44%, respectively, of our revenue was recognized when cash was received. Until we achieve our revenue recognition criteria for a larger number of payers, we will continue to recognize a large portion of our revenue when cash is received. Because we often need to appeal prior to being paid for certain tests, it can take over a year for a test to result in revenue being recorded, and for a portion of our tests, we may never realize revenue.
Additionally, as we commercialize new products, we will need to achieve our revenue recognition criteria for each payer for each new product prior to being able to recognize the related revenue on an accrual basis. Because the timing and amount of cash payments received from payers is difficult to predict, we expect our revenue may fluctuate significantly in any given quarter. In addition, even if we begin to accrue larger amounts of revenue
63
Table of Contents
related to AlloMap, when we introduce new products, we do not expect we will be able to recognize revenue from new products on an accrual basis for some period of time.
Continued Adoption of and Reimbursement for AlloMap
Our reimbursement rate has steadily increased over time since the launch of AlloMap, as payers adopt coverage policies and fewer payers consider AlloMap as experimental and investigational. The rate at which our tests are covered and reimbursed has, and is expected to continue to vary by payer. As of December 31, 2014, we had been reimbursed for approximately 79% of AlloMap results delivered in the twelve months ended June 30, 2014. Reimbursement performance is reviewed using a lagging metric of six months as any period less than this is considered not to be reflective of future performance, as the reimbursement process can take six months or more to complete depending on the payer. Revenue growth depends on our ability to achieve broader reimbursement from third party payers, to expand the number of tests per patient and the base of ordering physicians.
Development of Additional Products
We rely on sales of AlloMap to generate the majority of our revenue. Our product development pipeline includes other surveillance solutions for organ transplant recipients to help clinicians make personalized treatment decisions throughout a transplant patient’s lifetime. Accordingly, we expect to invest in research and development in order to develop additional products. Our success in developing new products will be important in our efforts to grow our business by expanding the potential market for our products and diversifying our sources of revenue.
Timing of Research and Development Expenses
Our spending on experiments may vary substantially from quarter to quarter. We also spend to secure clinical samples that can be used in discovery, product development, clinical validation, utility and outcome studies. The timing of these research and development activities is difficult to predict. If a substantial number of clinical samples are acquired in a given quarter or if a high-cost experiment is conducted in one quarter versus the next, the timing of these expenses can affect our financial results. We conduct clinical studies to validate our new products as well as on-going clinical and outcome studies to further the published evidence to support our commercialized AlloMap test. Spending on research and development for both experiments and studies, may vary significantly by quarter depending on the timing of these various expenses.
64
Table of Contents
Results of Operations
Comparison of the Years Ended December 31, 2014 and 2013
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | Change | ||||||||||
| (In thousands) | ||||||||||||
| Revenue |
||||||||||||
| Testing revenue |
$ | 25,842 | $ | 21,672 | $ | 4,170 | ||||||
| Collaboration and license revenue |
1,464 | 426 | 1,038 | |||||||||
|
|
|
|
|
|
|
|||||||
| 27,306 | 22,098 | 5,208 | ||||||||||
| Operating expenses: |
||||||||||||
| Cost of testing |
8,541 | 9,078 | (537 | ) | ||||||||
| Research and development |
3,846 | 3,176 | 670 | |||||||||
| Selling and marketing |
6,472 | 5,892 | 580 | |||||||||
| General and administrative |
8,436 | 4,809 | 3,627 | |||||||||
| Change in estimated fair value of contingent consideration |
(1,239 | ) | — | (1,239 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
26,056 | 22,955 | 3,101 | |||||||||
|
|
|
|
|
|
|
|||||||
| Income (loss) from operations |
1,250 | (857 | ) | 2,107 | ||||||||
| Interest expense, net |
(2,116 | ) | (2,149 | ) | 33 | |||||||
| Other income (expense), net |
147 | (536 | ) | 683 | ||||||||
|
|
|
|
|
|
|
|||||||
| Loss before income taxes |
(719 | ) | (3,542 | ) | 2,823 | |||||||
| Income tax benefit |
1,500 | — | 1,500 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) |
$ | 781 | $ | (3,542 | ) | $ | 4,323 | |||||
|
|
|
|
|
|
|
|||||||
Testing Revenue
AlloMap test results delivered increased by approximately 1,800 or 18% in 2014 compared to 2013. Testing revenue increased by $4.2 million or 19% in 2014 compared to 2013 primarily due to increased test volume and secondarily to increased number of payers for whom we recognize revenue on an accrual basis of approximately $2.6 million, of which Medicare accounted for $1.3 million of the increase, and additional cash collections of approximately $1.6 million from payers from which we recognize revenue on a cash basis.
Collaboration and License Revenue
Collaboration and license revenue increased by approximately $1.0 million, or 244% in 2014 compared to 2013 primarily due to our recognition of the termination fee of $0.5 million paid to us in connection with the termination of our Collaboration and License Agreement with Laboratory Corporation of America Holdings, dated April 2, 2012, as amended on September 13, 2012 (LabCorp Agreement), and amortization of the remaining deferred revenue balance under that agreement of $0.6 million.
Cost of Testing
Cost of testing decreased by approximately $0.5 million, or 6% in 2014 compared to 2013 primarily due to our royalty settlement with Roche, which resulted in a one-time reduction to royalty expense in 2014 of approximately $0.5 million.
65
Table of Contents
Research and Development
Research and development expenses increased $0.7 million or 21% in 2014 compared to 2013 primarily due to increased expenditure of $0.3 million in the area of cell-free DNA technology, an increase in headcount related expenses of $0.2 million, and an increase in expenditure of $0.2 million on AlloMap.
Sales and Marketing
Sales and marketing expenses increased by approximately $0.6 million, or 10% in 2014 compared to 2013. The increase primarily reflects increased salaries of $0.3 million, and increased travel and conference expenses of $0.3 million as we increased our commercialization efforts.
General and Administrative
General and administrative expenses increased approximately $3.6 million, or 75% in 2014 compared to 2013 due to increased employee costs of $1.1 million driven by additional hiring in preparation for our IPO, increased tax, audit and professional fees of $0.8 million as a result of our acquisition of ImmuMetrix and increased compliance costs of being a public company, $0.4 million of increased consulting fees, increased legal costs of $0.8 million largely as a result of our acquisition of ImmuMetrix and arbitration with Roche, $0.3 million associated with recruiting expenses of key employees and $0.2 million in general expenses. We anticipate our general and administrative expenses will increase as we operate as a public company.
Interest Expense, Net
Interest expense, net was flat from 2013 to 2014. There was an increase of $0.4 million associated with the $5.0 million Illumina subordinated convertible note issued in April 2014 with an interest rate of 8% which converted to common stock upon our IPO in July 2014, which was offset by a one-time reduction in interest expense of approximately $0.1 million due to the Roche settlement and a $0.3 million decrease in interest under our debt obligations due to principal payments.
Other Income (Expense), Net
Other income, net was $0.1 million in 2014 compared to $0.5 million in other expense, net in 2013. Other income, net for 2014 was primarily due to the remeasurement of the derivative associated with the Illumina subordinated convertible note. The subordinated convertible note was converted to common stock in July 2014 in connection with our IPO. Other expense, net for 2013 was primarily due to the remeasurement of the estimated fair value of the warrants to purchase shares of our convertible preferred stock. The warrants to purchase our convertible preferred stock were converted to warrants to purchase common stock in July 2014 in connection with our IPO and were reclassified to additional paid-in-capital on the IPO date at which point we ceased to record any further related periodic fair value adjustments.
Income Tax Benefit
In conjunction with the acquisition of ImmuMetrix, a tax benefit of $1.5 million was recognized in 2014. This benefit resulted from the expectation that amortization of the in-process technology acquired, when completed and placed in service, is not expected to be deductible for tax purposes, as the transaction was structured as a tax-free reorganization. Accordingly, a deferred tax liability was recorded at the acquisition date for the difference between the financial reporting and tax basis of the acquired in-process technology. While the in-process technology is considered an indefinite lived intangible asset, this asset is expected to be amortized or impaired prior to the expiration of net operating loss carryforwards available to us.
66
Table of Contents
Comparison of the Years Ended December 31, 2013 and 2012
| Year Ended December 31, | ||||||||||||
| 2013 | 2012 | Change | ||||||||||
| (In thousands) | ||||||||||||
| Revenue |
||||||||||||
| Testing revenue |
$ | 21,672 | $ | 19,730 | $ | 1,942 | ||||||
| Collaboration and license revenue |
426 | 721 | (295 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Operating expenses: |
22,098 | 20,451 | 1,647 | |||||||||
| Cost of testing |
9,078 | 7,930 | 1,148 | |||||||||
| Research and development |
3,176 | 4,752 | (1,576 | ) | ||||||||
| Selling and marketing |
5,892 | 5,417 | 475 | |||||||||
| General and administrative |
4,809 | 4,694 | 115 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
22,955 | 22,793 | 162 | |||||||||
|
|
|
|
|
|
|
|||||||
| Loss from operations |
(857 | ) | (2,342 | ) | 1,485 | |||||||
| Interest expense, net |
(2,149 | ) | (2,703 | ) | 554 | |||||||
| Other expense, net |
(536 | ) | (14 | ) | (522 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net loss |
$ | (3,542 | ) | $ | (5,059 | ) | $ | 1,517 | ||||
|
|
|
|
|
|
|
|||||||
Testing Revenue
Testing revenue increased by $1.9 million or 10%, in 2013 compared to 2012 primarily due to additional volume and to a lesser extent, an increase in payers meeting revenue recognition criteria. There was no material change year over year to our average revenue per test. Revenue recognized in 2013 as a result of payers meeting accrual criteria rather than remaining on the cash basis was approximately $0.3 million. Testing volume increased approximately 21% in 2013, as compared to 2012. The percentage increase in testing revenue was less than the percentage increase in testing volume due to the timing of the tests performed and our ability to recognize related revenue until the revenue recognition criteria were met.
Collaboration and License Revenue
Collaboration and license revenue decreased by $0.3 million or 41% in 2013 compared to 2012 primarily due to lower revenues from the LabCorp Agreement. Under the LabCorp Agreement, in 2012, we provided certain samples to the collaboration partner for $250,000; no such samples were provided under the agreement in 2013.
Cost of Testing
Cost of testing increased $1.1 million, or 14% in 2013 compared to 2012 reflecting our 21% testing volume growth in 2013. These increases included payroll and related expenses of $1.0 million, specimen processing of $0.2 million, licensing fees of $0.1 million and expired reagents of $0.2 million, partially offset by decreased depreciation of $0.4 million as certain lab equipment and software became fully depreciated. Royalty expense, included in cost of testing, was $1.1 million in 2012 and $1.2 million in 2013.
Research and Development
Research and development expenses decreased by $1.6 million, or 33%, in 2013 compared with 2012. The decrease reflects lower payroll and related costs of $1.0 million due to reduced headcount primarily in our informatics, clinical and regulatory groups. In addition, there was a reduction of approximately $0.5 million in
67
Table of Contents
depreciation and facilities-related expenses. During 2013, we focused our efforts on stabilizing and enhancing our current AlloMap business, choosing not to spend our limited resources on new product development. We expect our research and development expenses will increase in absolute dollars in future periods as we invest in research and discovery work to develop new surveillance solutions, as well as clinical outcomes studies for AlloMap and new tests, when developed.
Sales and Marketing
Sales and marketing increased by $0.5 million, or 9% in 2013 compared with 2012 primarily as a result of increased commissions of $0.3 million and increased marketing activities such as physician and nurse advisory boards and speaker programs of $0.2 million.
General and Administrative
General and administrative expenses increased $0.1 million, or 2%, in 2013 compared with 2012 primarily due to 2013 expenses of $0.3 million in connection with the evaluation of strategic alternatives, increased payroll and related costs of $0.2 million, partially offset by a reduction in costs associated with reporting capabilities for executive management of $0.3 million, and decreased outside services of $0.1 million.
Interest Expense, Net
Interest expense, net decreased by $0.6 million, or 20%, in 2013 compared with 2012. In April 2012, we converted all of our convertible subordinated promissory notes of $12.4 million principal and interest which had been issued in 2010, into preferred stock and preferred stock warrants. Interest expense on these notes recorded in 2012 was $0.4 million. In August 2012, we entered into a $15.0 million loan, and repaid at that time an existing loan with a principal balance of $10.3 million. Prepayment penalties and writeoff of the remaining unamortized costs associated with the paid off loan resulted in a charge to interest expense in 2012 of $0.6 million. The reduction in interest expense in 2013 compared to 2012 was partially offset by a higher effective interest rate on the $15.0 million loan compared to the previous $10.3 million loan.
Other Expense, Net
We recorded other expense of $0.5 million, for the year ended December 31, 2013 compared to a negligible amount for the year ended December 31, 2012. This change is due to the remeasurement of the estimated fair value of the warrants to purchase shares of our convertible preferred stock.
Liquidity and Capital Resources
Since our inception, substantially all of our operations have been financed through the issuance of our convertible preferred stock, the issuance of stock in our July 2014 initial public offering, the incurrence of debt, and cash received from AlloMap testing revenues. Through December 31, 2014, we have received net proceeds of $151 million from the issuances of preferred stock, including preferred stock issued on conversion of promissory notes, $35.5 million from our initial public offering, $15.0 million in proceeds from a venture debt loan, $5.0 million from a subordinated convertible note and approximately $131 million from AlloMap testing revenues. As of December, 2014, we had cash and cash equivalents of $36.4 million and $11.4 million of debt outstanding under our debt and capital lease obligations.
We have been using and plan to continue to use the $35.5 million of net proceeds from our initial public offering for research and development, including research aimed at expanding the clinical utility of AlloMap and the development of new solutions for the surveillance of heart and kidney transplant patients such as our cell-fee DNA technology, sales and marketing activities, general and administrative expenses and for working capital and other general corporate purposes. A portion of the net proceeds may also be used to acquire or invest in complementary businesses, technologies, services or products. We have no current agreements or commitments with respect to any such acquisition or investment.
68
Table of Contents
In April 2014, we issued a $5.0 million subordinated convertible note to Illumina, Inc., which provided for interest at an annual rate of 8.0%. Conversion was mandatory in the event of a qualified initial public offering, and the note converted into 510,777 shares of our common stock upon our IPO in July 2014 at a conversion price of $10.00 per share.
We currently anticipate that our cash and cash equivalents and projected cash receipts from AlloMap sales to customers will be sufficient to fund our operations for at least the next 18 months. We cannot be certain that any of our development of new transplant surveillance solutions will be successful or that we will be able to raise sufficient additional funds, if necessary, to see these programs through to a successful result. Our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement and involves risk and uncertainties, and actual results could vary as a result of a number of factors. We have based this estimate on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect.
The following table summarizes our cash flows for the years ended December 31, 2014, 2013 and 2012:
| Year Ended December 31, | ||||||||||||
| (in thousands) | 2014 | 2013 | 2012 | |||||||||
| Net cash (used in) provided by: |
||||||||||||
| Operating activities |
$ | (3,350 | ) | $ | (546 | ) | $ | (1,776 | ) | |||
| Investing activities |
(1,333 | ) | (98 | ) | 642 | |||||||
| Financing activities |
35,986 | (58 | ) | 4,607 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net increase (decrease) in cash and cash equivalents |
$ | 31,303 | $ | (702 | ) | $ | 3,473 | |||||
|
|
|
|
|
|
|
|||||||
Cash Flows from Operating Activities
Net cash provided by or used in operating activities consists of net income or loss, adjusted for certain non-cash items in the statements of operations and changes in operating assets and liabilities.
Net cash used in operating activities for the year ended December 31, 2014 was $3.4 million. The net income of $0.8 million reflects a non-cash income tax benefit of $1.5 million related to our acquisition of ImmuMetrix, the recognition of $0.7 million in collaboration revenue primarily from the termination of the LabCorp agreement, a $1.5 million decrease in the fair value of a contingent consideration related to the ImmuMetrix acquisition and an embedded derivative as a result of mark-to-market adjustments, partially offset by an increase in debt discount accretion and other non-cash interest expense of $0.8 million, depreciation and amortization of $0.5 million and stock-based compensation expense of $0.5 million, and an increase in net operating assets of $2.3 million. The increase in net operating assets primarily comprised of a decrease in accrued royalties of $2.6 million, an increase in accounts receivable of $0.4 million and an increase in inventory of $0.1 million as a result of an increase in our testing revenue, an increase in prepaid expenses and other assets of $0.3 million primarily due to our purchase of a directors and officers insurance policy when we became a public company in July 2014, partially offset by an increase in accounts payable of $0.5 million and an increase in accrued and other liabilities of $0.3 million as our expenditures increased in line with the growth of our core business, an increase in payroll-related liabilities of $0.3 million due to an increase in headcount. The decrease in accrued royalties was the result of a $2.8 million payment of past due royalties to Roche in September 2014 upon settlement of our dispute with Roche (for more information, see Note 6 of consolidated financial statements elsewhere in this Annual Report).
Net cash used in operating activities for the year ended December 31, 2013 was $0.5 million and reflected (i) net loss of $3.5 million, (ii) net non-cash items of $1.6 million, consisting primarily of depreciation and amortization of $0.7 million, amortization of debt discount and non-cash interest expense of $0.5 million and revaluation of warrants to estimated fair value of $0.5 million, and (iii) a net cash inflow from changes in balances of operating assets and liabilities of $1.4 million. The significant items comprising the changes in balances of operating assets
69
Table of Contents
and liabilities were a higher balance of accrued royalties of $1.3 million and a higher deferred revenue balance of $1.1 million, partially offset by increased accounts receivable balance of $1.3 million. The increased accounts receivable balance was due to increased volume of approximately $0.7 million, the change in our Medicare contractor effective October 2013, resulting in slower payments for Medicare tests of approximately $0.3 million, and more payers meeting our revenue recognition criteria of approximately $0.3 million. Our experience with Medicare contractor changes in the past has shown initially slower payments, which accelerate after the new contractor is in place for some period.
Net cash used in operating activities for the year ended December 31, 2012 was $1.8 million and reflected the net loss of $5.1 million, net non-cash items of $1.6 million, consisting primarily of depreciation and amortization of $1.1 million and amortization of debt discount and non-cash interest expense of $0.6 million, and a net cash inflow from changes in balances of operating assets and liabilities of $1.7 million. The significant items comprising the changes in balances of operating assets and liabilities were a higher balance of accrued royalties of $0.8 million and a higher deferred revenue balance of $1.0 million.
Cash flow from operations in 2013 and 2012 was aided by our suspension of royalty payments under our license agreement with Roche. We reached an agreement with Roche in September 2014 regarding the past-due royalties, and paid in full the agreed-to amount of $2.8 million.
Cash Flows from Investing Activities
For the year ended December 31, 2014 net cash used in investing activities was $1.3 million consisting $0.4 million in cash used to purchase ImmuMetrix and $0.7 million in purchases of property and equipment.
For the year ended December 31, 2013, net cash used in investing activities was $0.1 million and consisted of purchases of property and equipment.
For the year ended December 31, 2012, net cash provided by investing activities was $0.6 million and consisted of net maturities of short-term investments of $0.8 million, partially offset by $0.2 million of purchases of property and equipment.
We expect capital expenditures to increase modestly in 2015 and beyond as we expand our research and discovery work to develop new transplant surveillance solutions. We believe that our testing facility is not currently capacity constrained and that our current facility can support a substantial increase in testing volume and support new surveillance solutions currently being developed.
Cash Flows from Financing Activities
Net cash provided by financing activities for the year ended December 31, 2014 of $35.9 million was primarily from the receipt of $35.5 million in net proceeds from our July 2014 initial public offering, net of underwriters’ discounts and issuance costs, the receipt of $5.0 million in net proceeds from borrowing in connection with our April 2014 subordinated convertible note, partially offset by net payments of $4.6 million on our existing debt.
Net cash used in financing activities for the year ended December 31, 2013 was $0.1 million and primarily consisted of principal payments on capital lease obligations.
Net cash provided by financing activities for the year ended December 31, 2012 was $4.6 million and primarily consisted of net proceeds from the issuance of debt for $14.7 million, proceeds of $2.9 million from the sale of our convertible preferred stock, partially offset by principal payments on debt of $13.0 million.
70
Table of Contents
Contractual Obligations
The following table summarizes certain contractual obligations as of December 31, 2014:
| Payments Due by Period | ||||||||||||||||||||
| Total | Less Than 1 year |
1 to 3 Years |
3 to 5 Years |
More Than 5 Years |
||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Debt obligations |
11,394 | 6,037 | 5,357 | — | — | |||||||||||||||
| Operating lease obligations |
8,086 | 1,235 | 2,639 | 2,780 | 1,432 | |||||||||||||||
| Capital lease obligations |
197 | 80 | 105 | 12 | — | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
| Total |
19,677 | 7,352 | 8,101 | 2,792 | 1,432 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||
In August 2012, we entered into a $15.0 million loan and security agreement maturing in August 2016. As of December 31, 2014, we had an aggregate principal amount of $11.4 million outstanding under this loan. The loan provided for interest-only payments through February 2014. Beginning March 2014, the loan provided for 30 equal monthly principal and interest payments of $566,822 at a stated annual interest rate of 9.95%. In addition, a final payment of $1,275,000 was due at the end of the loan term. The loan was collateralized by a security interest in all of our assets except intellectual property on which there is a negative pledge, and the loan agreement contained covenants, including a revenue covenant, and restrictions on our ability to pay cash dividends. At December 31, 2014, we believe we were in compliance with all loan covenants.
Our cancelable operating lease obligations consist of the lease for our laboratory and office facility in Brisbane, California expiring in December 2020. Our capital lease obligations consist of equipment financing arrangements with vendors. The contractual obligations table above includes two capital leases entered into in April 2014.
Subsequent Event
On January 30, 2015, the Company entered into a Loan and Security Agreement (the “Loan Agreement”) which provides a secured term loan facility in an aggregate principal amount of up to $20.0 million. The Company borrowed the first advance of $16.0 million (“Draw A”) on January 30, 2015. Under the terms of the Loan Agreement, following a six month period from the closing date and until any time before December 31, 2015, the Company may, at its option, borrow from the lender a second advance of $4.0 million (“Draw B”), subject to the Company’s satisfaction of certain conditions described in the Loan Agreement. Draw A was used to pay-off the Company’s existing term debt of $11.3 million. Draw A and Draw B each bear interest at a floating rate equal to 2.00%, plus the greater of (i) 3.25% or (ii) the prime rate published by the lender.
In connection with the Loan Agreement, the Company agreed to issue to the lender warrants to purchase shares of the Company’s common stock upon the drawdown of each advance in an amount equal to 1.5% of the amount drawn, divided by the exercise price per share for that tranche. As a result of Draw A, the Company issued to the lender a warrant to purchase an aggregate of 34,483 shares of the Company’s common stock, at an exercise price equal to $6.96 per share.
Off-balance Sheet Arrangements
We have not entered into any off-balance sheet arrangements.
JOBS Act Accounting Election
We are an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. Under the JOBS Act, emerging growth companies can delay adopting new or revised accounting
71
Table of Contents
standards issued subsequent to the enactment of the JOBS Act until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
Recent Accounting Pronouncement
In May 2014, the Financial Accounting Standards Board issued Accounting Standards Update No. 2014-09, Revenue from Contracts with Customers (Topic 606) (“ASU 2014-09”), which amends the existing accounting standards for revenue recognition. ASU 2014-09 is based on principles that govern the recognition of revenue at an amount an entity expects to be entitled when products are transferred to customers. ASU 2014-09 will be effective for us beginning in the first quarter of 2017. Early adoption is not permitted. The new revenue standard may be applied retrospectively to each prior period presented or retrospectively with the cumulative effect recognized as of the date of adoption. We are currently evaluating the impact of adopting the new revenue standard on our financial statements.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK.
Foreign Currency Risk
Since 2014, our AlloMap test has been offered in Europe through our agreement with Diaxonhit and in Canada through our agreement with LifeLabs Medical Laboratory Services. Payments to us under these agreements are denominated in U.S. dollars. Expenses we incur in currencies other than U.S. dollars are not material. We do not believe that our foreign currency exchange rate fluctuation risk is significant and we do not believe that a hypothetical 10% change in foreign currency exchange rates would have a significant effect on our financial condition, operating results or cash flows.
Interest Rate Risk
We are exposed to market risks in the ordinary course of our business. These risks primarily relate to interest rates. We had cash and cash equivalents of $36.4 million and $5.1 million as of December 31, 2014 and December 31, 2013, respectively, which consist of bank deposits and money market funds. Such interest-bearing instruments carry a degree of risk; however, we have not been exposed to, nor do we anticipate being exposed to, material risks due to changes in interest rates. A hypothetical 10% change in interest rates during any of the periods presented would not have had a material impact on our audited financial statements.
72
Table of Contents
| Page No. | ||||
| 74 | ||||
| 75 | ||||
| 76 | ||||
| Statements of Convertible Preferred Stock and Stockholders’ (Deficit) Equity |
77 | |||
| 78 | ||||
| 79 | ||||
73
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders of
CareDx, Inc.
We have audited the accompanying balance sheets of CareDx, Inc. as of December 31, 2014 and 2013, and the related statements of operations, convertible preferred stock and stockholders’ (deficit) equity, and cash flows for each of the three years in the period ended December 31, 2014. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. We were not engaged to perform an audit of the Company’s internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of CareDx, Inc. at December 31, 2014 and 2013, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2014, in conformity with U.S. generally accepted accounting principles.
/s/ Ernst & Young LLP
Redwood City, California
March 31, 2015
74
Table of Contents
Balance Sheets
(In thousands, except share and per share data)
| As of December 31, | ||||||||
| 2014 | 2013 | |||||||
| Assets |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 36,431 | $ | 5,128 | ||||
| Accounts receivable |
2,687 | 2,270 | ||||||
| Inventory |
686 | 518 | ||||||
| Prepaid and other assets |
542 | 255 | ||||||
|
|
|
|
|
|||||
| Total current assets |
40,346 | 8,171 | ||||||
| Property and equipment, net |
1,968 | 1,553 | ||||||
| Intangible assets, net |
6,650 | — | ||||||
| Goodwill |
12,005 | — | ||||||
| Restricted cash |
147 | 147 | ||||||
| Other noncurrent assets |
25 | 2 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 61,141 | $ | 9,873 | ||||
|
|
|
|
|
|||||
| Liabilities, convertible preferred stock, and stockholders’ equity (deficit) |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 1,128 | $ | 618 | ||||
| Accrued payroll liabilities |
1,684 | 1,386 | ||||||
| Accrued and other liabilities |
1,616 | 1,048 | ||||||
| Accrued royalties |
241 | — | ||||||
| Deferred revenue |
505 | 80 | ||||||
| Current portion of long-term debt |
5,961 | 4,461 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
11,135 | 7,593 | ||||||
| Accrued royalties |
— | 2,804 | ||||||
| Deferred rent, net of current portion |
1,684 | 1,885 | ||||||
| Deferred revenue, net of current portion |
471 | 1,623 | ||||||
| Long-term portion of debt and other, net of current portion |
5,451 | 10,914 | ||||||
| Convertible preferred stock warrant liability |
— | 525 | ||||||
| Contingent consideration |
1,074 | — | ||||||
| Other liabilities |
28 | — | ||||||
|
|
|
|
|
|||||
| Total liabilities |
19,843 | 25,344 | ||||||
| Commitments and contingencies (Note 7) |
||||||||
| Convertible preferred stock: $0.001 par value; 0 and 6,417,954 shares authorized at December 31, 2014 and 2013, respectively; 0 and 5,155,673 shares issued and outstanding at December 31, 2014 and 2013, respectively; liquidation value of $0 and $137,221 at December 31, 2014 and 2013, respectively |
— | 135,202 | ||||||
| Stockholders’ equity (deficit): |
||||||||
| Preferred stock: $0.001 par value; 10,000,000 and 0 shares authorized at December 31, 2014 and December 31, 2013, respectively; 0 and 0 shares issued and outstanding at December 31, 2014 and December 31, 2013, respectively |
— | — | ||||||
| Common stock: $0.001 par value; 100,000,000 and 7,737,226 shares authorized at December 31, 2014 and December 31, 2013, respectively; 11,803,970 and 1,010,711 shares issued and outstanding at December 31, 2014 and December 31, 2013, respectively |
12 | 1 | ||||||
| Additional paid-in capital |
200,661 | 9,482 | ||||||
| Accumulated deficit |
(159,375 | ) | (160,156 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders’ equity (deficit) |
41,298 | (150,673 | ) | |||||
|
|
|
|
|
|||||
| Total liabilities, convertible preferred stock and stockholders’ equity (deficit) |
$ | 61,141 | $ | 9,873 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these financial statements.
75
Table of Contents
Statements of Operations
(In thousands, except share and per share data)
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Revenue: |
||||||||||||
| Testing revenue |
$ | 25,842 | $ | 21,672 | $ | 19,730 | ||||||
| Collaboration and license revenue |
1,464 | 426 | 721 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total revenue |
27,306 | 22,098 | 20,451 | |||||||||
| Operating expenses: |
||||||||||||
| Cost of testing |
8,541 | 9,078 | 7,930 | |||||||||
| Research and development |
3,846 | 3,176 | 4,752 | |||||||||
| Sales and marketing |
6,472 | 5,892 | 5,417 | |||||||||
| General and administrative |
8,436 | 4,809 | 4,694 | |||||||||
| Change in estimated fair value of contingent consideration |
(1,239 | ) | — | — | ||||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
26,056 | 22,955 | 22,793 | |||||||||
|
|
|
|
|
|
|
|||||||
| Income (loss) from operations |
1,250 | (857 | ) | (2,342 | ) | |||||||
| Interest expense, net |
(2,116 | ) | (2,149 | ) | (2,703 | ) | ||||||
| Other income (expense), net |
147 | (536 | ) | (14 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Loss before income taxes |
(719 | ) | (3,542 | ) | (5,059 | ) | ||||||
| Income tax benefit |
1,500 | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) |
$ | 781 | $ | (3,542 | ) | $ | (5,059 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) per share (Note 3): |
||||||||||||
| Basic |
$ | 0.13 | $ | (3.50 | ) | $ | (5.01 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Diluted |
$ | 0.10 | $ | (3.50 | ) | $ | (5.01 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Shares used to compute net income (loss) per share: |
||||||||||||
| Basic |
5,815,928 | 1,010,795 | 1,009,236 | |||||||||
|
|
|
|
|
|
|
|||||||
| Diluted |
9,283,001 | 1,010,795 | 1,009,236 | |||||||||
|
|
|
|
|
|
|
|||||||
The accompanying notes are an integral part of these financial statements.
76
Table of Contents
Statements of Convertible Preferred Stock and Stockholders’ (Deficit) Equity
(In Thousands, Except Share Data)
| Convertible Preferred Stock |
Common Stock | Additional Paid-In Capital |
Accumulated Deficit |
Total Stockholders’ (Deficit) Equity |
||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||
| Balance at December 31, 2011 |
4,447,657 | $ | 119,837 | 1,007,575 | $ | 1 | $ | 9,331 | $ | (151,555 | ) | $ | (142,223 | ) | ||||||||||||||
| Issuance of Series G convertible preferred stock in April 2012 for cash, net of issuance costs |
137,722 | 2,941 | — | — | — | — | — | |||||||||||||||||||||
| Conversion of notes payable and interest in April 2012 to Series G convertible preferred stock, net of issuance costs |
570,294 | 12,424 | — | — | — | — | — | |||||||||||||||||||||
| Issuance of common stock for cash upon exercise of stock options |
— | — | 2,924 | — | 10 | — | 10 | |||||||||||||||||||||
| Employee share-based compensation expense |
— | — | — | — | 69 | — | 69 | |||||||||||||||||||||
| Net loss |
— | — | — | — | — | (5,059 | ) | (5,059 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance at December 31, 2012 |
5,155,673 | 135,202 | 1,010,499 | 1 | 9,410 | (156,614 | ) | (147,203 | ) | |||||||||||||||||||
| Issuance of common stock for cash upon exercise of stock options |
— | — | 212 | — | — | — | — | |||||||||||||||||||||
| Employee share-based compensation expense |
— | — | — | — | 72 | — | 72 | |||||||||||||||||||||
| Net loss |
— | — | — | — | — | (3,542 | ) | (3,542 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance at December 31, 2013 |
5,155,673 | 135,202 | 1,010,711 | 1 | 9,482 | (160,156 | ) | (150,673 | ) | |||||||||||||||||||
| Convertible preferred stock Series G issued for the acquisition of ImmuMetrix, Inc. |
888,135 | 14,242 | — | — | — | — | — | |||||||||||||||||||||
| Conversion of convertible preferred stock to common stock upon initial public offering |
(6,043,808 | ) | (149,444 | ) | 6,048,220 | 6 | 149,438 | — | 149,444 | |||||||||||||||||||
| Conversion of subordinated convertible note to common stock upon initial public offering |
— | — | 510,777 | 1 | 5,107 | — | 5,108 | |||||||||||||||||||||
| Conversion of convertible preferred stock warrants to common stock warrants upon initial public offering |
— | — | — | — | 539 | — | 539 | |||||||||||||||||||||
| Issuance of common stock upon initial public offering, net of offering costs |
— | — | 4,220,000 | 4 | 35,507 | — | 35,511 | |||||||||||||||||||||
| Issuance of common stock for services |
— | — | 4,899 | — | 34 | — | 34 | |||||||||||||||||||||
| Issuance of common stock for cash upon exercise of stock options |
— | — | 9,363 | — | 19 | — | 19 | |||||||||||||||||||||
| Employee and non-employee share-based compensation expense |
— | — | — | — | 535 | — | 535 | |||||||||||||||||||||
| Net income |
— | — | — | — | — | 781 | 781 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance at December 31, 2014 |
— | $ | — | 11,803,970 | $ | 12 | $ | 200,661 | $ | (159,375 | ) | $ | 41,298 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
The accompanying notes are an integral part of these financial statements.
77
Table of Contents
Statements of Cash Flows
(In thousands)
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Operating activities: |
||||||||||||
| Net income (loss) |
$ | 781 | $ | (3,542 | ) | $ | (5,059 | ) | ||||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: |
||||||||||||
| Depreciation and amortization |
512 | 663 | 1,072 | |||||||||
| Stock-based compensation |
535 | 72 | 69 | |||||||||
| Amortization of deferred revenue |
(727 | ) | (193 | ) | (187 | ) | ||||||
| Amortization of debt discount and noncash interest expense |
799 | 553 | 634 | |||||||||
| Revaluation of warrants and derivatives to estimated fair value |
(1,464 | ) | 525 | (2 | ) | |||||||
| Non-cash income tax benefit in connection with business combinations |
(1,500 | ) | — | — | ||||||||
| Changes in operating assets and liabilities: |
||||||||||||
| Accounts receivable |
(417 | ) | (1,318 | ) | (150 | ) | ||||||
| Inventory |
(168 | ) | 58 | (152 | ) | |||||||
| Prepaid and other assets |
(310 | ) | (4 | ) | 146 | |||||||
| Accounts payable |
510 | (19 | ) | (113 | ) | |||||||
| Accrued payroll liabilities |
298 | 408 | (76 | ) | ||||||||
| Accrued royalties |
(2,563 | ) | 1,259 | 843 | ||||||||
| Deferred revenue |
— | 1,083 | 1,000 | |||||||||
| Accrued and other liabilities |
364 | (91 | ) | 199 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net cash used in operating activities |
(3,350 | ) | (546 | ) | (1,776 | ) | ||||||
| Investing activities: |
||||||||||||
| Purchase of investments |
— | — | (1,623 | ) | ||||||||
| Sales of investments |
— | — | 383 | |||||||||
| Maturities of investments |
— | — | 2,023 | |||||||||
| Purchase of property and equipment |
(733 | ) | (98 | ) | (141 | ) | ||||||
| Payment for acquisitions, net of cash acquired |
(600 | ) | — | — | ||||||||
|
|
|
|
|
|
|
|||||||
| Net cash (used in) provided by investing activities |
(1,333 | ) | (98 | ) | 642 | |||||||
| Financing activities: |
||||||||||||
| Proceeds from initial public offering, net of underwriters discount |
39,246 | — | — | |||||||||
| Payment of initial public offering costs |
(3,733 | ) | — | — | ||||||||
| Proceeds from subordinated convertible debt, net of issuance costs |
4,982 | — | — | |||||||||
| Proceeds from issuance of convertible preferred stock, net of issuance costs |
— | — | 2,941 | |||||||||
| Proceeds from debt, net of issuance costs |
— | — | 14,650 | |||||||||
| Proceeds from exercise of stock options |
19 | — | 10 | |||||||||
| Principal payments on debt |
(4,528 | ) | (58 | ) | (12,994 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by (used in) financing activities |
35,986 | (58 | ) | 4,607 | ||||||||
|
|
|
|
|
|
|
|||||||
| Net increase (decrease) in cash and cash equivalents |
31,303 | (702 | ) | 3,473 | ||||||||
| Cash and cash equivalents at beginning of period |
5,128 | 5,830 | 2,357 | |||||||||
|
|
|
|
|
|
|
|||||||
| Cash and cash equivalents at end of period |
$ | 36,431 | $ | 5,128 | $ | 5,830 | ||||||
|
|
|
|
|
|
|
|||||||
| Supplemental disclosures of cash information |
||||||||||||
| Cash paid for interest |
$ | 1,207 | $ | 1,506 | $ | 1,966 | ||||||
|
|
|
|
|
|
|
|||||||
| Supplemental disclosures of noncash investing and financing activities |
||||||||||||
| Property and equipment purchased under capital leases |
$ | 193 | $ | — | $ | 43 | ||||||
| Common stock issued for acquisition |
14,242 | — | — | |||||||||
| Conversion of convertible preferred stock to common stock upon initial public offering |
149,444 | — | — | |||||||||
| Conversion of convertible preferred stock warrants to common stock warrants upon initial public offering |
539 | — | — | |||||||||
| Conversion of subordinated convertible note to common stock upon initial public offering |
5,108 | — | — | |||||||||
| Common stock issued for services |
34 | — | — | |||||||||
The accompanying notes are an integral part of these financial statements.
78
Table of Contents
NOTES TO FINANCIAL STATEMENTS
1. ORGANIZATION AND DESCRIPTION OF BUSINESS
CareDx, Inc., (“CareDx” or the “Company”) is a commercial stage company that develops, markets and delivers a diagnostic surveillance solution for heart transplant recipients to help clinicians make personalized treatment decisions throughout a transplant patient’s lifetime. The Company’s commercialized testing solution, the AlloMap heart transplant molecular test (“AlloMap”), an FDA-cleared test, is a blood-based test used to monitor for acute cellular rejection in heart transplant recipients. The Company was incorporated in Delaware in December 1998, as Hippocratic Engineering, Inc. In April 1999, the Company changed its name to BioCardia, Inc., in June 2002 to Expression Diagnostics, Inc., in July 2007 to XDx, Inc. and in March 2014 to CareDx, Inc. The Company’s operations are based in Brisbane, California and it operates in one segment.
Reverse Stock Split, and Increase in Authorized Shares
On July 1, 2014, the Company’s Board of Directors approved an amendment to the Company’s Certificate of Incorporation to reflect a 1 for 6.85 reverse stock split (the “Reverse Stock Split”) of the Company’s outstanding common stock and convertible preferred stock and increase the authorized common stock to 10,000,000 shares, after giving effect to the Reverse Stock Split. The Reverse Stock Split became effective July 14, 2014. The par value per share was not adjusted as a result of the Reverse Stock Split. Effective July 22, 2014, the Company’s certificate of incorporation was amended and restated to provide for 100,000,000 authorized shares of common stock with a par value of $0.001 per share, and 10,000,000 authorized shares of preferred stock with a par value of $0.001 per share. All authorized, issued and outstanding shares of common stock, convertible preferred stock, options and warrants to purchase common or preferred stock and related per share amounts contained in the financial statements have been retroactively adjusted to reflect the Reverse Stock Split for all periods presented.
Initial Public Offering
On July 22, 2014, the Company closed its initial public offering (“IPO”) of 4,000,000 shares of its common stock, and issued an additional 220,000 shares of common stock on August 13, 2014 pursuant to the exercise of the over-allotment option granted to its underwriters. The public offering price of the shares sold in the offering was $10.00 per share. The total proceeds from the offering to the Company, net of underwriting discounts and commissions of $3.0 million, were $39.2 million. After deducting offering expenses payable by the Company of $3.7 million, net proceeds to the Company were $35.5 million. Upon the closing of the IPO, all shares of convertible preferred stock then outstanding converted into 6,048,220 shares of common stock, and a subordinated convertible note previously issued by the Company in the principal amount of $5.0 million converted into 510,777 shares of common stock. In addition, all of our convertible preferred stock warrants were converted into warrants to purchase common stock.
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The Company’s financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“U.S. GAAP”). In September 2014, the Company’s wholly-owned subsidiary, ImmuMetrix, Inc., was merged into CareDx, Inc., and as a result, at December 31, 2014, the financial statements of the former ImmuMetrix, Inc. are included in the financial statements of CareDx, Inc.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities and the reported amounts of revenues and expenses in the financial statements and accompanying
79
Table of Contents
notes. On an ongoing basis, management evaluates its estimates, including those related to (i) revenue recognition, (ii) the differences between amounts billed and estimated receipts from payers, (iii) the determination of the accruals for clinical studies, (iv) the determination of refunds to be requested by third-party payers, (v) the fair value of assets and liabilities, (vi) the valuation of warrants to purchase convertible preferred stock, (vii) the determination of fair value of the Company’s common stock, (viii) the contingent consideration in a business acquisition, (ix) the fair value of the embedded derivative associated with the subordinated convertible note, (x) the determination of the valuation allowance and estimated tax benefit associated with deferred tax assets and net deferred tax liability, (xi) any impairment of long-lived assets including in-process technology and goodwill and (xii) legal contingencies. Actual results could differ from those estimates.
Concentrations of Credit Risk and Other Risks and Uncertainties
Financial instruments that potentially subject the Company to credit risk consist of cash and cash equivalents and accounts receivable. The Company’s policy is to invest its cash and cash equivalents and short-term investments in money market funds, obligations of U.S. government agencies and government-sponsored entities, commercial paper, and various bank deposit accounts. These financial instruments were held in Company accounts at two financial institutions. The counterparties to the agreements relating to the Company’s investments consist of financial institutions of high credit standing. The Company is exposed to credit risk in the event of default by the financial institutions to the extent of amounts recorded on the balance sheets which may be in excess of insured limits.
The Company is also subject to credit risk from its accounts receivable which are derived from revenue earned from AlloMap tests provided for patients located in the U.S. and billed to various third-party payers. For the years ended December 31, 2014, 2013 and 2012, approximately 51%, 53% and 52%, respectively, of testing revenue was derived from Medicare. No other payers represented more than 10% of testing revenue for these periods. At December 31, 2014 and 2013, approximately 78% and 72%, respectively, of accounts receivable was from Medicare. No other payer represented more than 10% of accounts receivable at December 31, 2014 or 2013.
Cash Equivalents
Cash equivalents consist of short-term, highly liquid investments with original maturities of three months or less from the date of purchase. Cash equivalents consist primarily of amounts invested in money market funds.
Restricted Cash
Under lease agreements for certain facilities and an agreement with the State of Florida Medicaid, the Company must maintain letters of credit, minimum collateral requirements, and a surety bond. These agreements are collateralized by cash. The cash which will continue long-term to support these arrangements is classified as restricted cash on the accompanying balance sheets.
Inventory
Inventory is finished goods and raw materials which consist of AlloMap reagent plates, laboratory supplies and reagents. Inventories are used in connection with tests performed and may also be used for research and product development efforts. Laboratory supplies subsequently designated for research and product development use are expensed. Obsolete or damaged inventories are written off and excluded from the physical inventory. Inventories are stated at the lower of actual purchased cost determined on a first-in, first-out basis, or net realizable value.
80
Table of Contents
Property and Equipment, net
Property and equipment are stated at cost, less accumulated depreciation. Property and equipment are depreciated using the straight-line method over the estimated useful lives of the assets, generally three years for laboratory, computer, and office equipment, and generally seven years for furniture and fixtures. Leasehold improvements are amortized over the shorter of their estimated useful lives or the remaining lease term.
Assets held under capital leases are recorded at the lower of the net present value of the minimum lease payments or the fair market value of the leased asset at the inception of the lease. Amortization expense is computed using the straight-line method over the shorter of the estimated useful lives of the assets or the period of the related lease.
The Company capitalizes certain costs incurred for software developed or obtained for internal use. These costs include software licenses, consulting services, and direct materials, as well as employee payroll and payroll-related costs. Capitalized internal-use software costs are depreciated over three years.
Purchased Intangible Assets
Acquired intangible assets with indefinite useful lives are related to purchased in-process research and development (IPR&D) projects and are measured at their respective fair values as of the acquisition date. We do not amortize goodwill and intangible assets with indefinite useful lives. Intangible assets related to IPR&D projects are considered to be indefinite-lived until the completion or abandonment of the associated R&D efforts. If and when development is complete, which generally occurs if and when regulatory approval to market a product is obtained, the associated assets would be deemed finite-lived and would then be amortized based on their respective estimated useful lives at that point in time.
We test IPR&D for impairment on an annual basis and in between annual tests if we become aware of any events or changes that would indicate that it is more likely than not that the fair value of the assets are below their carrying amounts. If the fair value exceeds the carrying value, then there is no impairment. Impairment losses on indefinite-lived intangible assets are recognized based solely on a comparison of the fair value of the asset to its carrying value, without consideration of any recoverability test. The Company has not identified any such impairment losses to date.
Impairment of Long-lived Assets
The Company evaluates its long-lived assets for indicators of possible impairment when events or changes in circumstances indicate the carrying amount of an asset may not be recoverable. The Company then compares the carrying amounts of the assets with the future net undiscounted cash flows expected to be generated by such asset. Should an impairment exist, the impairment loss would be measured based on the excess carrying value of the asset over the asset’s fair value determined using discounted estimates of future cash flows. The Company has not identified any such impairment losses to date.
Fair Value of Financial Instruments
Fair value is defined as the price that would be received from selling an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining fair value, the Company considers the principal or most advantageous market in which the Company would transact, and it takes into consideration the assumptions that market participants would use when pricing the asset or liability. The Company’s assessment of the significance of a particular input to the fair value measurement of an asset or liability requires management to make judgments and to consider specific characteristics of that asset or liability.
81
Table of Contents
The carrying amounts of certain of the Company’s financial instruments, including cash equivalents, accounts receivable, accounts payable, and accrued liabilities, approximate fair value due to their short maturities. The carrying amount of the convertible preferred stock warrant liability and contingent consideration liability also represents its fair value.
Warrants
The Company had freestanding warrants enabling counterparties to purchase shares of its convertible preferred stock as of December 31, 2013, which were converted to warrants to purchase common stock on the Company’s IPO date.
In accordance with the accounting guidance regarding distinguishing liabilities from equity, freestanding warrants for convertible preferred stock that are contingently redeemable are classified as liabilities on the balance sheet and recorded at their estimated fair value. These warrants are remeasured at each balance sheet date and any change in estimated fair value is recognized in other expense, net on the statements of operations.
Upon the completion of the Company’s IPO in July 2014, preferred stock warrants were converted into warrants to purchase common stock, and, accordingly, the liability was reclassified to equity and became no longer subject to remeasurement.
Testing Revenue
The Company recognizes revenues for tests delivered when the following criteria are met: (i) persuasive evidence that an arrangement exists; (ii) delivery has occurred or services rendered; (iii) the fee is fixed or determinable; and (iv) collectability is reasonably assured.
The first criterion is satisfied when a third-party payer makes a coverage decision or enters into a contractual arrangement with the Company for the test. The second criterion is satisfied when the Company performs the test and delivers the test result to the ordering physician. The third criterion is satisfied if the third-party payer’s coverage decision or reimbursement contract specifies a price for the test. The fourth criterion is satisfied based on management’s judgments regarding the collectability of the fees charged under the arrangement. Such judgments include review of past payment history. AlloMap testing may be considered investigational by some payers and not covered under their reimbursement policies. Others may cover the test, but not pay a set or determinable amount. As a result, in the absence of a reimbursement agreement or sufficient payment history, collectability cannot reasonably be assured so revenue is not recognized at the time the test is delivered.
If all criteria set forth above are met, revenue is recognized. When the first, third or fourth criteria are not met but third-party payers make a payment to the Company for tests performed, the Company recognizes revenue on the cash basis in the period in which the payment is received.
Revenue is recognized on the accrual basis net of adjustments for differences between amounts billed and the estimated receipts from payers. The amount the Company expects to collect may be lower than the agreed upon amount due to several factors, such as the amount of patient co-payments, the existence of secondary payers and claim denials. Estimated receipts are based upon historical payment practices of payers. Differences between estimated and actual cash receipts are recorded as an adjustment to revenue, which have been immaterial to date.
Taxes assessed by governmental authorities on revenue, including sales and value added taxes, are excluded from revenue in the statements of operations.
Collaboration and License Revenue
The Company generates revenue from collaboration and license agreements. Collaboration and license agreements may include non-refundable upfront payments, partial or complete reimbursement of research and
82
Table of Contents
development costs, contingent payments based on the occurrence of specified events under the agreements, license fees and royalties on sales of products or product candidates if they are successfully commercialized. The Company’s performance obligations under the collaborations may include the transfer of intellectual property rights in the form of licenses, obligations to provide research and development services and obligations to participate on certain development committees with the collaboration partners. The Company makes judgments that affect the periods over which it recognizes revenue. The Company periodically reviews its estimated periods of performance based on the progress under each arrangement and accounts for the impact of any change in estimated periods of performance on a prospective basis.
The Company recognizes contingent consideration received from the achievement of a substantive milestone in its entirety in the period in which the milestone is achieved, which the Company believes is more consistent with the substance of its performance under its various license and collaboration agreements. The Company did not recognize any milestones during the years ended December 31, 2014, 2013 or 2012.
Cost of Testing
Cost of testing reflects the aggregate costs incurred in delivering the Company’s AlloMap test results to clinicians. The components of cost of testing are materials and service costs, direct labor costs, including stock-based compensation, equipment and infrastructure expenses associated with testing samples, shipping, logistics and specimen processing charges to collect and transport samples and allocated overhead including rent, information technology, equipment depreciation and utilities and royalties. Costs associated with performing tests (except royalties) are recorded as the test is processed regardless of whether and when revenue is recognized with respect to that test. As a result, our cost of testing as a percentage of revenue may vary significantly from period to period because we do not recognize all revenue in the period in which the associated costs are incurred. Royalties for licensed technology, calculated as a percentage of test revenues, are recorded as license fees in cost of testing at the time the test revenues are recognized.
Business Combinations
In accordance with ASC 805, Business Combinations, the Company determines and allocates the purchase price of an acquired business to the tangible and identifiable intangible assets acquired and liabilities assumed based on their estimated fair values as of the business combination date, including identifiable intangible assets which either arise from a contractual or legal right or are separable from goodwill. The Company bases the estimated fair value of identifiable intangible assets acquired in a business combination on independent valuations that use information and assumptions provided by management, which consider management’s best estimates of inputs and assumptions that a market participant would use. The Company allocates any excess purchase price over the estimated fair value assigned to the net tangible and identifiable intangible assets acquired and liabilities assumed to goodwill. The use of alternative valuation assumptions, including estimated revenue projections, growth rates, royalty rates, cash flows, discount rates, estimated useful lives and probabilities surrounding the achievement of contingent milestones could result in different purchase price allocations and amortization expense in current and future periods.
In those circumstances where an acquisition involves a contingent consideration arrangement that meets the definition of a liability under ASC 480, Distinguishing Liabilities from Equity, the Company recognizes a liability equal to the fair value of the contingent payments the Company expects to make as of the acquisition date. The Company remeasures this liability each reporting period and records changes in the fair value as a component of operating expenses.
Transaction costs associated with these acquisitions are expensed as incurred in general and administrative expenses. Results of operations and cash flows of acquired companies are included in the Company’s operating results from the date of acquisition.
83
Table of Contents
Research and Development Expenses
Research and development expenses represent costs incurred to develop new surveillance solutions as well as continued efforts related to the Company’s AlloMap test. These expenses include payroll and related expenses, consulting expenses, laboratory supplies, and certain allocated expenses as well as amounts incurred under certain collaboration and license agreements. Research and development costs are expensed as incurred. The Company records accruals for estimated study costs comprised of work performed by contract research organizations under contract terms.
Stock-based Compensation
The Company uses the Black-Scholes valuation model, which requires the use of estimates such as stock price volatility and expected option lives, to value employee stock options. The Company estimates the expected option lives using historical data, volatility using data of similar companies in the diagnostics industry, and risk-free rates based on the implied yield currently available in the U.S. Treasury zero-coupon issues with a remaining term equal to the expected option lives, and dividend yield based on the Company’s historical data.
The Company uses the straight-line attribution method for recognizing compensation expense. Compensation expense is recognized on awards ultimately expected to vest and reduced for forfeitures that are estimated at the time of grant and revised, if necessary, in subsequent periods if actual forfeitures differ from those estimates. Forfeitures are estimated based on the Company’s historical experience.
Compensation expense for stock options issued to nonemployees is calculated using the Black-Scholes option pricing model and is recorded over the service performance period. Options subject to vesting are required to be periodically remeasured over their service performance period, which is generally the same as the vesting period.
Income Taxes
The Company accounts for income taxes under the liability method. Under this method, deferred tax assets and liabilities are determined based on the difference between the financial statement and tax bases of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to affect taxable income. Valuation allowances are established when necessary to reduce deferred tax assets to the amounts expected to be realized.
The Company assesses all material positions taken in any income tax return, including all significant uncertain positions, in all tax years that are still subject to assessment or challenge by relevant taxing authorities. The Company’s assessment of an uncertain tax position begins with the initial determination of the position’s sustainability and is measured at the largest amount of benefit that is greater than 50 percent likely of being realized upon ultimate settlement. As of each balance sheet date, unresolved uncertain tax positions must be reassessed, and the Company will determine whether (i) the factors underlying the sustainability assertion have changed and (ii) the amount of the recognized tax benefit is still appropriate. The recognition and measurement of tax benefits requires significant judgment. Judgments concerning the recognition and measurement of a tax benefit may change as new information becomes available.
Comprehensive Income (Loss)
Net income (loss) and comprehensive income (loss) are the same for all periods presented.
Recent Accounting Pronouncement
In May 2014, the Financial Accounting Standards Board issued Accounting Standards Update No. 2014-09, Revenue from Contracts with Customers (Topic 606) (“ASU 2014-09”), which amends the existing accounting standards for revenue recognition. ASU 2014-09 is based on principles that govern the recognition of revenue at an amount an entity expects to be entitled to when products are transferred to customers. ASU 2014-09 will be effective for the Company beginning in its first quarter of 2017. Early adoption is not permitted. The new
84
Table of Contents
revenue standard may be applied retrospectively to each prior period presented or retrospectively with the cumulative effect recognized as of the date of adoption. The Company is currently evaluating the impact of adopting the new revenue standard on its financial statements.
3. NET INCOME (LOSS) PER SHARE
Basic net income (loss) per share has been computed by dividing the net income (loss) by the weighted-average number of common shares outstanding during the period, without consideration for common share equivalents.
Diluted net income (loss) per share has been computed by dividing the net income (loss) by the sum of the weighted-average number of common shares and common share equivalents outstanding during the period, to the extent that such common share equivalents are dilutive.
For the year ended December 31, 2014, certain common share equivalents have been included in diluted net income per share, as their effect is dilutive. For the year ended December 31, 2014, common share equivalents include: (i) options and warrants to purchase common stock; (ii) options and warrants to purchase convertible preferred stock prior to their conversion into options and warrants to purchase common stock upon the IPO on July 22, 2014; and (iii) convertible preferred stock and the subordinated convertible note prior to their conversion into common stock upon the IPO. Common share equivalents for convertible preferred stock and the subordinated convertible note are determined using the if-converted method. Common share equivalents for options and warrants are determined using the treasury-stock method.
For the years ended December 31, 2013 and 2012, all common share equivalents have been excluded from the calculation of diluted net loss per share, as the effect would be antidilutive.
The following tables set forth the computation of the Company’s basic and diluted net income (loss) per share (in thousands, except shares and per share data):
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Numerator: |
||||||||||||
| Net income (loss) |
$ | 781 | $ | (3,542 | ) | $ | (5,059 | ) | ||||
| Add: interest expense related to subordinated convertible note |
364 | — | — | |||||||||
| Less: gain on change in fair value of derivative related to subordinated convertible note |
(118 | ) | — | — | ||||||||
| Less: gain on extinguishment of derivative related to subordinated convertible note |
(120 | ) | — | — | ||||||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) attributable to common stockholders |
$ | 907 | $ | (3,542 | ) | $ | (5,059 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Denominator: |
||||||||||||
| Weighted-average shares used to compute basic net income (loss) per share attributable to common stockholders |
5,815,928 | 1,010,795 | 1,009,236 | |||||||||
| Effect of potentially dilutive securities: |
||||||||||||
| Shares of common stock subject to conversion from preferred stock |
2,972,051 | — | — | |||||||||
| Shares of common stock subject to conversion from subordinated convertible note |
134,341 | — | — | |||||||||
| Shares of common stock subject to outstanding options |
360,681 | — | — | |||||||||
|
|
|
|
|
|
|
|||||||
| Weighted-average shares used to compute diluted net income (loss) per share attributable to common stockholders |
9,283,001 | 1,010,795 | 1,009,236 | |||||||||
|
|
|
|
|
|
|
|||||||
| Net income (loss) per share attributable to common stockholders: |
||||||||||||
| Basic |
$ | 0.13 | $ | (3.50 | ) | $ | (5.01 | ) | ||||
|
|
|
|
|
|
|
|||||||
| Diluted |
$ | 0.10 | $ | (3.50 | ) | $ | (5.01 | ) | ||||
|
|
|
|
|
|
|
|||||||
85
Table of Contents
The following potentially dilutive securities have been excluded from diluted net income (loss) per share, because their effect would be antidilutive:
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Shares of common stock subject to outstanding options |
539,645 | 466,965 | 619,906 | |||||||||
| Shares of common stock subject to outstanding warrants |
213,677 | 82,190 | 82,190 | |||||||||
| Shares of common stock subject to conversion from preferred stock |
— | 5,160,085 | 5,160,085 | |||||||||
| Shares of common stock subject to conversion from preferred stock warrants |
— | 541,613 | 541,613 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total common stock equivalents |
753,322 | 6,250,853 | 6,403,794 | |||||||||
|
|
|
|
|
|
|
|||||||
4. FAIR VALUE MEASUREMENTS
The Company records its financial assets and liabilities at fair value except for its debt, which is recorded at amortized cost. The carrying amounts of certain financial instruments of the Company, including cash and cash equivalents, prepaid expenses and other current assets, accounts payable and accrued liabilities, approximate fair value due to their relatively short maturities. The accounting guidance for fair value provides a framework for measuring fair value, clarifies the definition of fair value, and expands disclosures regarding fair value measurements. Fair value is defined as the price that would be received to sell an asset or paid to transfer a liability (an exit price) in an orderly transaction between market participants at the reporting date. The accounting guidance establishes a three-tiered hierarchy, which prioritizes the inputs used in the valuation methodologies in measuring fair value as follows:
| • | Level 1: Inputs which include quoted prices in active markets for identical assets and liabilities. |
| • | Level 2: Inputs other than Level I that are observable, either directly or indirectly, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities. |
| • | Level 3: Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities. |
The following table sets forth the fair value of the Company’s financial assets and liabilities measured on a recurring basis, as of December 31, 2014 and 2013 (in thousands):
| December 31, 2014 | ||||||||||||||||
| Fair Value Measured Using | Total Balance |
|||||||||||||||
| (Level 1) | (Level 2) | (Level 3) | ||||||||||||||
| Assets |
||||||||||||||||
| Money market funds |
$ | 36,779 | $ | — | $ | — | $ | 36,779 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Liabilities |
||||||||||||||||
| Contingent consideration |
$ | — | $ | — | $ | 1,074 | $ | 1,074 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total liabilities |
$ | — | $ | — | $ | 1,074 | $ | 1,074 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| December 31, 2013 | ||||||||||||||||
| Fair Value Measured Using | Total Balance |
|||||||||||||||
| (Level 1) | (Level 2) | (Level 3) | ||||||||||||||
| Assets |
||||||||||||||||
| Money market funds |
$ | 5,204 | $ | — | $ | — | $ | 5,204 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Liabilities |
||||||||||||||||
| Warrants to purchase convertible preferred stock |
$ | — | $ | — | $ | 525 | $ | 525 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total liabilities |
$ | — | $ | — | $ | 525 | $ | 525 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
86
Table of Contents
The following table presents the issuances, changes in fair value and reclassifications of the Company’s Level 3 financial instruments that are measured at fair value on a recurring basis (in thousands):
| Level 3 | ||||||||||||||||
| Contingent Consideration Liability |
Warrants to Purchase Convertible Preferred Stock |
Derivative Liability Related to Subordinated Convertible Note |
Total | |||||||||||||
| Balance as of January 1, 2013 |
$ | — | $ | — | $ | — | $ | — | ||||||||
| Change in estimated fair value |
— | 525 | — | 525 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Balance as of December 31, 2013 |
— | 525 | — | 525 | ||||||||||||
| Issuance of financial instruments |
2,313 | — | 239 | 2,552 | ||||||||||||
| Change in estimated fair value |
(1,239 | ) | 14 | (239 | ) | (1,464 | ) | |||||||||
| Reclassification to stockholders’ equity |
— | (539 | ) | (539 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Balance as of December 31, 2014 |
$ | 1,074 | $ | — | $ | — | $ | 1,074 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
The Company recognizes transfers between levels of the fair value hierarchy as of the end of the reporting period. There were no transfers between Level 1, Level 2 and Level 3 categories during the periods presented.
In determining fair value, the Company uses various valuation approaches within the fair value measurement framework. The valuation methodologies used for the Company’s instruments measured at fair value and their classification in the valuation hierarchy are summarized below:
| • | Money market funds—Investments in money market funds are classified within Level 1. At December 31, 2014 and December 31, 2013, money market funds were included on the balance sheets in cash and cash equivalents. |
| • | Contingent consideration—As of December 31, 2014, the Company had a contingent obligation to issue 227,845 shares of the Company’s common stock to the former owners of ImmuMetrix, Inc. in conjunction with the Company’s acquisition of ImmuMetrix, Inc. (see Note 15). The issuance will occur if the Company completes 2,500 commercial tests involving the measurement of cfDNA in organ transplant recipients in the United States by June 10, 2020. The Company recorded its estimate of the fair value of the contingent consideration based on its evaluation of the probability of the achievement of the contractual conditions that would result in the payment of the contingent consideration. The fair value of the contingent consideration was estimated using the fair value of the shares to be paid if the contingency is met multiplied by management’s 65% estimate at December 31, 2014 of the probability of success. The significant input in the Level 3 measurement not supported by market activity is the Company’s probability assessment of the milestone being met. The value of the liability is subsequently remeasured to fair value each reporting date, and the change in estimated fair value is recorded to the operating expense item captioned “change in estimated fair value of contingent consideration” until the milestone contingency is paid, expires or is no longer achievable. Increases (decreases) in the estimation of the probability percentage result in a directionally similar impact to the fair value measurement of the contingent consideration liability. |
| • | Warrants to purchase convertible preferred stock—At December 31, 2013, the Company’s warrants to purchase convertible preferred stock were classified as Level 3 because they were valued based on unobservable inputs and management’s judgment due to the absence of quoted market prices, inherent lack of liquidity and the long-term nature of such financial instruments. These assumptions are inherently subjective and involve significant management judgment. The significant unobservable input used in the fair value measurement of the warrant liability was the fair value of the underlying convertible preferred stock at the valuation remeasurement date. Generally, increases (decreases) in the fair value of the underlying stock would result in a directionally similar impact to the fair value |
87
Table of Contents
| measurement of the preferred stock warrants. Any change in estimated fair value is recognized in other income or expense on the statements of operations. Upon the Company’s IPO in July 2014, certain warrants to purchase convertible preferred stock were converted into warrants to purchase common stock and were reclassified to equity, while other warrants to purchase convertible preferred stock expired pursuant to their terms. |
| • | Derivative liability related to subordinated convertible note—On April 17, 2014, the Company issued a $5.0 million subordinated convertible promissory note to Illumina, Inc. that had some features that constituted embedded derivatives. The Company determined that the optional conversion or repayment upon a change in control is an equity call option with a potentially variable value to be received and meets the definition of a derivative which would be required to be bifurcated. The estimated fair value of this embedded derivative was affected by the estimated probability assigned to the various scenarios for the host instrument. As of April 17, 2014, management estimated repayment upon a change in control within the loan term at a 10% probability. As of June 30, 2014 management estimated repayment upon a change in control within the loan term at a 5% probability. The $239,000 original estimated fair value of the embedded derivative liability was included in accrued and other liabilities. At June 30, 2014, the fair value of the derivative was remeasured to $120,000, resulting in a gain of $119,000, which was recorded in other income in the statements of operations for the three months ended June 30, 2014. Upon the Company’s IPO in July 2014, subordinated convertible note was converted into common stock, and so the embedded conversion option was extinguished. Accordingly, the fair value of the derivative became $0, and a gain of $120,000 was recorded in other income. The significant unobservable input used in the fair value measurement of the derivative liability was the probability assigned to the various scenarios. Generally, increases (decreases) in the probability of the factors primarily impacting the valuation would result in a directionally similar impact to the fair value measurement of the derivative liability. Changes in estimated fair value were recognized in other income (expense) on the statements of operations. |
The Company’s liabilities classified as Level 3 were valued based on unobservable inputs and management’s judgment due to the absence of quoted market prices, inherent lack of liquidity and the long-term nature of the financial instruments.
5. BALANCE SHEET COMPONENTS
Inventory
Inventory consisted of the following (in thousands):
| December 31, | ||||||||
| 2014 | 2013 | |||||||
| Finished Goods |
$ | 277 | $ | 230 | ||||
| Raw Materials |
409 | 288 | ||||||
|
|
|
|
|
|||||
| Total Inventory |
$ | 686 | $ | 518 | ||||
|
|
|
|
|
|||||
88
Table of Contents
Property and Equipment, Net
Property and equipment consisted of the following (in thousands):
| December 31, | ||||||||
| 2014 | 2013 | |||||||
| Laboratory equipment |
$ | 4,392 | $ | 3,798 | ||||
| Leasehold improvements |
4,040 | 4,336 | ||||||
| Furniture and fixtures |
825 | 838 | ||||||
| Computer equipment and office equipment |
3,775 | 3,444 | ||||||
| Construction-in-process |
28 | 39 | ||||||
|
|
|
|
|
|||||
| 13,060 | 12,455 | |||||||
| Less: Accumulated depreciation and amortization |
(11,092 | ) | (10,902 | ) | ||||
|
|
|
|
|
|||||
| Property and equipment, net |
$ | 1,968 | $ | 1,553 | ||||
|
|
|
|
|
|||||
Depreciation and amortization expense was $512,000, $663,000 and $1,072,000 for the years ended December 31, 2014, 2013 and 2012, respectively.
Assets purchased under capital leases, included above in laboratory equipment and computer and office equipment, were $1,631,000 and $1,439,000 at December 31, 2014 and 2013, respectively. Accumulated amortization was $1,473,000 and $1,414,000 at December 31, 2014 and 2013, respectively. Related amortization expense, included in depreciation and amortization expense, was $59,000, $26,000 and $70,000 for the years ended December 31, 2014, 2013 and 2012, respectively.
Accrued and Other Liabilities
Accrued and other liabilities consisted of the following (in thousands):
| December 31, | ||||||||
| 2014 | 2013 | |||||||
| Professional fees |
$ | 273 | $ | 175 | ||||
| Test sample processing fees |
318 | 195 | ||||||
| Accrued overpayments and refunds |
146 | 215 | ||||||
| Clinical Studies |
144 | 84 | ||||||
| Deferred rent – current portion |
202 | 145 | ||||||
| Capital leases – current portion |
70 | 43 | ||||||
| Other accrued expenses |
463 | 191 | ||||||
|
|
|
|
|
|||||
| Total accrued and other liabilities |
$ | 1,616 | $ | 1,048 | ||||
|
|
|
|
|
|||||
6. COMMITMENTS AND CONTINGENCIES
Leases
The Company leases its laboratory and office facility in Brisbane, California, under a non-cancelable operating lease agreement expiring in December 2020. The terms of the facility lease provide for rental payments on a graduated scale. The Company recognizes rent expense on a straight-line basis over the lease period and has accrued for rent expense incurred but not paid. In addition, incentives were granted, including allowances to fund leasehold improvements and rent holidays. As such, these allowances have been recorded as deferred rent, and these items are being recognized as reductions to rental expense on a straight-line basis over the term of the lease.
89
Table of Contents
During 2013, the Company had leases for blood draw centers under non-cancelable operating leases. Although the Company had subleased some of the blood draw center properties, it remained obligated under the original operating leases. The final lease payment related to the blood draw centers was made in August 2013.
Rent expense under the non-cancelable operating leases was $1,007,000, $1,020,000 and $1,016,000 for the years ended December 31, 2014, 2013 and 2012, respectively. Future minimum lease commitments under these operating and capital leases at December 31, 2014, are as follows (in thousands):
| Years ending December 31, | Capital Leases | Operating leases | ||||||
| 2015 |
$ | 81 | $ | 1,235 | ||||
| 2016 |
62 | 1,291 | ||||||
| 2017 |
42 | 1,348 | ||||||
| 2018 |
7 | 1,376 | ||||||
| 2019 and thereafter |
5 | 2,836 | ||||||
|
|
|
|
|
|||||
| Total minimum lease payments |
197 | $ | 8,086 | |||||
|
|
|
|
|
|||||
| Less: amounts representing interest |
(21 | ) | ||||||
|
|
|
|||||||
| Present value of minimum lease payments |
176 | |||||||
| Less: current portion of obligations under capital leases |
(70 | ) | ||||||
|
|
|
|||||||
| Long-term portion of obligations under capital leases |
$ | 106 | ||||||
|
|
|
|||||||
The current portion of obligations under capital leases is included in accrued and other liabilities on the balance sheets. The long-term portion is included in long-term debt on the balance sheets.
See Note 8 for the aggregate annual payment schedule for the Company’s outstanding venture debt.
Royalty Commitments
In November 2004, the Company entered into a license agreement with Roche Molecular Systems, Inc., or Roche, that grants the Company the right to use certain Roche technology relating to polymerase chain reaction, or PCR, and quantitative real-time PCR, in clinical laboratory services, including in connection with AlloMap. This is a non-exclusive license agreement in the United States covering claims in multiple Roche patents. The Company had disputed the combination services percentage Roche sought to apply under the agreement. The combination service percentage is a multiplier used to calculate royalties where licensed services are sold in combination with other services. From July 2011 through September 2014, the Company withheld payment of such royalties pending resolution of the matter. On February 11, 2014, Roche filed a demand for arbitration with the American Arbitration Association seeking a declaration that the Company had materially breached the Roche license agreement by failing to report and pay royalties owing to Roche in respect of licensed services performed by the Company after July 1, 2011. Since July 1, 2011, the Company fully accrued the unpaid royalties on the balance sheets, and the amount of the unpaid royalties has been reflected as an expense in the Company’s income statements in the periods to which the royalties relate.
In September 2014, the Company entered into a settlement and mutual release agreement with Roche whereby: (i) for the period beginning July 1, 2011 through June 30, 2014, the Company agreed to pay the amount of $2,827,220 in settlement of past royalties due; (ii) for the period beginning July 1, 2014 through September 30, 2014, the Company agreed to pay royalties based on the same combination services percentage used to determine the past royalties due; (iii) for the period beginning October 1, 2014 through September 30, 2017, Roche and the Company agreed to a downward adjustment of the combination services percentage used to determine the portion of the AlloMap testing revenue that is royalty bearing under the terms of the license; (iv) the Company agreed to report and pay quarterly royalties within 45 days of the end of each calendar quarter; (v) Roche agreed that, subject to the Company’s timely payment of all applicable royalties through such date, no further royalties will
90
Table of Contents
be payable by the Company for periods after September 30, 2017; (vi) the Company and Roche agreed to mutually release all claims under the license agreement through the settlement date; and (vii) Roche agreed to dismiss the arbitration claims. For all time periods, the contractual royalty rate in the license agreement was or will be applied to the applicable combination services percentage to determine the royalties payable for the AlloMap service.
Under the license agreement, the Company incurs royalty expenses as a percentage of combination services revenue and classifies those expenses as a component of cost of testing in the statements of operations. As a result of the Company’s September 2014 settlement and payment to Roche of $2.8 million as payment in full of all royalties under the license agreement from July 1, 2011 through June 30, 2014, the Company recorded a reduction of $566,000 to cost of testing and $132,000 to interest expense in the statements of operations for the year ended December 31, 2014. Of the $2.8 million paid by the Company under the terms of the settlement agreement, $0.4 million represented royalties on AlloMap revenue for the six months ended December 31, 2011, $0.9 million represented royalties on AlloMap revenue for the year ended December 31, 2012, $1.0 million represented royalties on AlloMap revenue for the year ended December 31, 2013, and $0.6 million represented royalties on AlloMap revenue for the year ended December 31, 2014.
For the years ended December 31, 2014, 2013 and 2012, royalty expenses in connection with the Roche agreement were $650,000, $1.2 million and $1.1 million, respectively.
Contingencies
The Company is subject to claims and assessments from time to time in the ordinary course of business. The Company’s management does not believe that any such matters, individually or in the aggregate, will have a material adverse effect on the Company’s business, financial condition, or results of operations.
7. COLLABORATION AND LICENSING AGREEMENTS
Laboratory Corporation of America Holdings (“LabCorp”)
In April 2012, CareDx and LabCorp entered into a collaboration and license agreement (“2012 Agreement”) to develop a lupus flare predictor test. The agreement provided for CareDx to license technology to LabCorp. Of the total arrangement consideration, the fair value of the license was assessed to be $1.0 million. The license term in the 2012 Agreement was the later of 10 years from the date of the agreement or the expiration of the last-to-expire patents and patent applications included in the CareDx technology licensed to LabCorp, unless the license were terminated by mutual agreement. The agreement provided that CareDx and LabCorp would share equally the costs of developing the lupus flare predictor test; however LabCorp’s share of the development cost was subject to certain limits at each stage of the arrangement.
Under the agreement, in 2012 LabCorp paid the Company a nonrefundable and non-creditable upfront license fee payment of $1,000,000, and a nonrefundable and non-creditable payment of $250,000 for certain lupus samples. The Company was to receive royalties in the high single digits from LabCorp on net sales of the commercialized flare predictor test or other tests developed using the samples sold.
Phase 1 of the project was completed in the first quarter of 2014.
On September 18, 2014, CareDx and LabCorp terminated the 2012 agreement. The termination agreement provides that:
| • | CareDx transfer and assign to LabCorp, 300 “SAGE I” clinical samples and related clinical data and documentation that CareDx obtained from patients during the discovery phase of the collaboration; |
| • | CareDx grant a perpetual, non-exclusive worldwide, fully paid, sublicensable, royalty-free license to use any collaboration intellectual property and data for any and all purposes; and |
91
Table of Contents
| • | LabCorp pay $500,000 to CareDx within 30 days of CareDx’s delivery of the clinical samples and clinical data and documentation. No further royalties, milestone fees or other fees will be payable by LabCorp after the termination date. |
During the three months ended December 31, 2014, the Company delivered the clinical samples and the related clinical data and documentation to LabCorp, and accordingly recognized the $500,000 termination fee and the remaining $611,000 previously unrecognized license fee.
During the years ended December 31, 2014, 2013 and 2012, the Company recognized $1.1 million, $328,000 and $437,000, respectively, in revenue under this arrangement, which consisted of amortization of the upfront license fee of $626,000, $187,000 and 187,000, respectively, reimbursement of research and development expenses of $16,000, $141,000 and $250,000, respectively, and a collaboration termination fee of $500,000, $0 and $0, respectively. Such revenues are included in collaboration and license revenue on the statements of operations.
Included in research and development expenses were $32,000, $282,000 and $499,000 for the years ended December 31, 2014, 2013 and 2012, respectively, for development costs associated with the 2012 Agreement.
Diaxonhit (“DHT”)
In June 2013, the Company entered into an exclusive Distribution and Licensing Agreement with DHT, a French public company, whereby DHT will have the AlloMap test performed in a European laboratory and commercialize the test in the European Economic Area (“EEA”). The agreement will expire at the later of the last-to-expire patent in the EEA or ten years from the first commercial sale of the test in the EEA, which is expected to occur in late 2014 or early 2015.
Consideration under the agreement includes an upfront cash payment of approximately €387,500 ($503,000) that is designated to offset royalties earned by the Company in the first three years following the first commercial sale. The Company is entitled to receive royalties from DHT as a percent of net sales, as defined in the agreement, of AlloMap tests in the mid to high teens. Approximately €250,000 ($344,000) of the upfront payments is refundable under certain circumstances. Upon confirmation that the CE mark was in place, the Company also received an equity payment of DHT common stock with a value of €387,500 ($503,000). The CE mark is a mandatory conformity marking for certain products sold within the EEA. These shares were promptly sold by the Company in July 2013 for total consideration of $467,000.
Other consideration that may be earned by the Company includes agreed-upon per unit pricing for the supply of AlloMap products, and additional royalties that are payable upon the achievement of various sales milestones by DHT. In this arrangement, there is one combined unit of accounting.
Commercial sales began in the EEA in June 2014. Total revenue recognized from this arrangement for the year ended December 31, 2014 was $36,000.
CardioDx, Inc. (“CDX”)
In 2005, the Company entered into a services agreement with what at the time was a related party, CDX, whereby the Company provided CDX with biological samples and related data and performed laboratory services on behalf of CDX. Each company granted the other a worldwide license under certain of its intellectual property rights. Pursuant to this agreement, CDX pays royalties to the Company of a low single-digit percentage of the cash collected from sales of CDX licensed products. In 2009, CDX terminated the services portion of this agreement, however, the royalty obligation from CDX continues until the tenth anniversary of the first commercial sale of a CDX licensed product. The first commercial sale of such product by CDX occurred in 2009, therefore the royalty obligation to the Company continues until 2019. Two board members of CDX serve
92
Table of Contents
on the Company’s board of directors and are affiliated with stockholders of the Company. Royalty revenues, recorded when earned, were $221,000, $94,000 and $34,000 for the years ended December 31, 2014, 2013 and 2012, respectively, and are included in collaboration and license revenue on the statements of operations. The Company had receivable balances from CDX of $54,000 and $37,000 at December 31, 2014 and December 31, 2013, respectively.
8. DEBT
Venture Debt
In August 2012, the Company entered into a $15,000,000 loan and security agreement (“the 2012 Loan”), and repaid a loan entered into in 2010 (the “2010 Loan”) including principal of $10,333,833, a termination fee of $875,000, and other costs associated with the payoff. Prepayment penalties and write-off of the remaining unamortized costs associated with the 2010 Loan resulted in a charge to interest expense of approximately $628,000 during the year ended December 31, 2012. These transactions generated net cash proceeds to the Company of $3,432,260.
In August 2013, the Company amended the 2012 Loan to defer the beginning of repaying principal for six months, to March 1, 2014. To obtain this deferral, there was an additional fee of $150,000 due at the end of the loan term. The 2012 Loan, as amended, provides for interest-only payments for 18 months followed by 30 equal monthly principal and interest payments of $566,822 at an annual interest rate of 9.95%. In addition, a final payment of $1,275,000 will be due at the end of the loan term. The 2012 Loan also included a facility fee of $75,000.
In connection with the 2012 Loan, the Company issued to the lenders warrants to purchase 167,182 shares of Series G convertible preferred stock or Next Round Stock at $21.78 per share. The warrants are exercisable until 2019. The estimated fair value of warrants on the date of issuance was negligible. The estimated fair value of the warrants at December 31, 2013, including the methodology and input assumptions used in the valuation, is discussed in Note 10. On July 22, 2014, upon the completion of the Company’s IPO, the warrants converted into warrants to purchase common stock.
The 2012 Loan is collateralized by a security interest in all of the Company’s assets except intellectual property on which there is a negative pledge, and the loan agreement contains covenants, including a revenue covenant, and restrictions on the Company’s ability to pay cash dividends. At December 31, 2014, the Company was in compliance with all loan covenants.
In connection with the 2010 Loan, the Company issued to the lenders warrants to purchase 17,215 shares of Series G convertible preferred stock at $21.78 per share. The warrants were exercisable until 2017. The estimated fair value of the warrants on the date of issuance of $8,000 was recorded as a debt discount liability which was being amortized to interest expense over the term of the loan. For the year ended December 31, 2012, amortization of $4,400 was included in interest expense. The estimated fair value of the warrants at December 31, 2013, including the methodology and input assumptions used in the valuation, is discussed in Note 9. On July 22, 2014, upon the completion of the Company’s IPO, the warrants converted into warrants to purchase common stock.
93
Table of Contents
Aggregate annual payments at December 31, 2014 due on the 2012 Loan, as amended, are as follows (in thousands):
| Year ending December 31: | Annual Payments |
|||
| 2015 |
$ | 6,802 | ||
| 2016 |
5,809 | |||
|
|
|
|||
| 12,611 | ||||
| Less: amounts representing interest |
(2,204 | ) | ||
|
|
|
|||
| 10,407 | ||||
| Plus: unamortized premium, net |
899 | |||
|
|
|
|||
| 11,306 | ||||
| Less: current portion |
(5,961 | ) | ||
|
|
|
|||
| Long-term portion |
$ | 5,345 | ||
|
|
|
|||
Convertible Notes
At December 31, 2011, the Company had a liability for $11,365,000 principal amount of convertible subordinated promissory notes, which had been issued at various times from June 2010 to October 2011 (“the 2010 Notes”). The 2010 Notes had an annual interest rate of 7%, compounded annually and payable at maturity. Amortization of $39,000 for an embedded debt discount related to the 2010 Notes is included in interest expense during the year ended December 31, 2012.
In April 2012, the Company converted all the 2010 Notes including principal and interest of $12,423,000, into 570,294 shares of Series G preferred stock at $21.78 per share, and issued 357,216 Series G preferred stock warrants exercisable at $21.78 per share. The estimated fair value of the warrants on the date of issuance was negligible.
Subordinated Convertible Note
On April 17, 2014, the Company issued a $5.0 million Subordinated Convertible Promissory Note to Illumina, Inc. (the “the 2014 Note”) which provided for interest at an annual rate of 8.0%. The 2014 Note was to mature one year following its issuance with principal and unpaid interest due at that time unless the Note were converted into equity prior to the maturity date. As described below, conversion was mandatory in the event of a Qualified Initial Public Offering (as defined in the 2014 Note). Upon the closing of the IPO on July 22, 2014, the 2014 Note converted into 510,777 shares of common stock in accordance with its terms.
The original estimated fair value of the embedded derivative was accounted for as a debt discount to the subordinated convertible note payable on the consolidated condensed balance sheet. The estimated fair value of the embedded derivative liability was included in accrued and other liabilities on the condensed balance sheets. Amortization of the debt discount was $256,000 for the period from April 17, 2014 to July 22, 2014, when the 2014 Note was converted into common stock. Extinguishment of the embedded derivative liability at July 22, 2014 resulted in other income of $120,000.
94
Table of Contents
9. WARRANTS
At December 31, 2013, outstanding convertible preferred stock warrants consisted of:
| Issue Date |
Original Term |
Convertible Preferred Stock |
Exercise Price |
Number of Shares of Outstanding Underlying Warrant |
Estimated Fair Value as of December 31, 2013 (In thousands) |
|||||||||||||||
| December 2010 (a) |
7 years | Series G | $ | 21.78 | 17,215 | $ | 14 | |||||||||||||
| April 2012 (b) |
5 years | Series G | $ | 21.78 | 357,216 | 218 | ||||||||||||||
| August 2012 (c) |
7 years | Series G | $ | 21.78 | 167,182 | 293 | ||||||||||||||
|
|
|
|
|
|||||||||||||||||
| 541,613 | $ | 525 | ||||||||||||||||||
|
|
|
|
|
|||||||||||||||||
| (a)—Issued | to lenders in connection with the 2010 Loan (see Note 8). |
| (b)—Issued | to note holders upon the conversion of all of the 2010 Notes (see Note 8). |
| (c)—Issued | to lenders in connection with the 2012 Loan (see Note 8). |
The warrants were exercisable upon issuance. At December 31, 2013, the warrants were classified as liabilities on the balance sheet. Upon the completion of the Company’s IPO in July 2014, all outstanding convertible preferred stock warrants were converted into warrants to purchase common stock and accordingly the liability was reclassified to stockholders’ equity. Prior to the IPO, the Company adjusted the liability for changes in fair value at each balance sheet date.
The estimated fair value of the outstanding convertible preferred stock warrants was measured at December 31, 2013, using the Black-Scholes option pricing model with the following assumptions: expected volatility ranging from 40% to 45%, a contractual term equal to the remaining contractual term, risk-free interest rate ranging from 0.8% to 2.1%, underlying common stock price of $8.97, and dividend yield of 0%.
In October 2011, a number of preferred stock holders did not purchase their pro rata share of the 2010 Notes, and as a result, 82,190 warrants to purchase Series E, F, and G preferred stock converted to warrants to purchase common stock. The strike price of the warrants remained the same. At December 31, 2013, outstanding common stock warrants consisted of:
| Original Term |
Exercise Price |
Number of Shares Outstanding Underlying Warrants |
||||||||||
| Original issue date: |
||||||||||||
| July 2006 |
10 years | $ | 31.72 | 17,656 | ||||||||
| November 2006 |
10 years | $ | 31.72 | 1,576 | ||||||||
| February 2008 |
10 years | $ | 35.07 | 22,792 | ||||||||
| August 2009 |
10 years | $ | 21.78 | 33,472 | ||||||||
| July 2010 |
9 years | $ | 21.78 | 6,694 | ||||||||
|
|
|
|||||||||||
| 82,190 | ||||||||||||
|
|
|
|||||||||||
10. CONVERTIBLE PREFERRED STOCK
Upon the closing of the Company’s IPO on July 22, 2014, all shares of convertible preferred stock then outstanding converted into 6,048,220 shares of common stock, and a subordinated convertible note previously issued by the Company in the principal amount of $5.0 million converted into 510,777 shares of common stock.
95
Table of Contents
Convertible preferred stock as of December 31, 2013 consisted of the following:
| Shares | ||||||||||||||||
| Series |
Authorized | Outstanding | Carrying Value | Liquidation Value | ||||||||||||
| A |
79,708 | 16,930 | $ | 267,000 | $ | 290,000 | ||||||||||
| B |
145,221 | 16,065 | 826,143 | 853,143 | ||||||||||||
| C |
850,899 | 829,209 | 14,139,373 | 14,200,373 | ||||||||||||
| D |
965,672 | 877,880 | 18,459,000 | 20,024,985 | ||||||||||||
| E |
857,323 | 835,547 | 26,423,000 | 26,499,984 | ||||||||||||
| F |
1,463,859 | 1,441,053 | 50,443,000 | 50,541,097 | ||||||||||||
| G |
2,055,272 | 1,138,989 | 24,644,515 | 24,811,238 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total |
6,417,954 | 5,155,673 | $ | 135,202,031 | $ | 137,220,820 | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
At December 31, 2013, the convertible preferred stock was classified as temporary equity on the Company’s balance sheets. These shares were contingently redeemable upon events that were outside of the control of the Company, including a liquidation, sale or transfer of control. Prior to the automatic conversion of the convertible preferred stock to common stock upon the IPO in July 2014, the Company did not adjust the carrying values of its convertible preferred stock to their redemption values since it was uncertain whether or when a redemption event would occur.
Dividends
Prior to conversion of all convertible preferred stock into common stock upon the Company’s IPO, holders of Series A, Series B, Series C, Series D, Series E, Series F, and Series G convertible preferred stock were entitled to noncumulative dividends of $1.199, $3.720, $1.199, $1.596, $2.219, $2.459, and $1.528 per share, respectively, if and when declared by the Board of Directors. These dividends were to be paid in advance of any distributions to common stockholders. No dividends were declared prior to the conversion of the convertible preferred stock into common stock upon the IPO.
Conversion
At December 31, 2013, each share of Series A, Series C, Series D, Series E, Series F, and Series G convertible preferred stock was convertible, at the option of the holder, into one share of common stock. Each share of Series B convertible preferred stock was convertible at the option of the holder into 0.18611 shares of common stock. The initial conversion prices for shares of Series A, Series B, Series C, Series D, Series E, Series F, and Series G convertible preferred stock were $17.12, $13.43, $17.12, $21.78, $21.78, $21.78, and $21.78 per share, respectively. Upon the Company’s IPO, all shares of convertible preferred stock were converted into shares of common stock.
Liquidation Preference
Prior to conversion of all convertible preferred stock into common stock upon the Company’s IPO, in the event of the liquidation or dissolution of the Company, the Series A, Series B, Series C, Series D, Series E, Series F, and Series G convertible preferred stock carried liquidation preferences of $17.12, $53.09, $17.12, $22.81, $31.72, $35.07, and $21.78 per share, respectively, plus all declared but unpaid dividends. Holders of Series D and Series E convertible preferred stock had liquidation preference subsequent to the holders of Series F and Series G convertible stock. Holders of Series F convertible preferred stock had liquidation preference subsequent to holders of Series G convertible stock. Holders of Series A, Series B, and Series C convertible preferred stock had liquidation preference subsequent to the holders of Series D, Series E, Series F, and Series G convertible preferred stock. After liquidation preferences to all convertible preferred stockholders have been paid, the remaining assets of the Company shall be distributed among the holders of common stock.
96
Table of Contents
Voting Rights
Prior to conversion of all convertible preferred stock into common stock upon the Company’s IPO, each holder of shares of Series A, Series B, Series C, Series D, Series E, Series F, and Series G convertible preferred stock was entitled to voting rights equivalent to the number of shares of common stock into which their respective shares were convertible. So long as 14,599 shares of Series C convertible preferred stock remained outstanding, the holders of Series C convertible preferred stock were entitled to elect three members to the Company’s Board of Directors. So long as 14,599 shares of Series D and Series E convertible preferred stock remained outstanding, the holders of Series D and Series E convertible preferred stock were entitled to elect one member to the Board of Directors. So long as 14,599 shares of Series F and Series G convertible preferred stock remained outstanding, the holders of Series F and Series G convertible preferred stock were entitled to elect one member to the Company’s Board of Directors. The holders of Series A and B convertible preferred stock and the holders of common stock, voting together as a single class, were entitled to elect three members to the Board of Directors.
11. STOCK INCENTIVE PLANS
Stock Option Plans
Prior to its IPO in July 2014, the Company had one active stock option plan, the 2008 Equity Incentive Plan (“2008 Plan”), one assumed stock option plan (the ImmuMetrix 2013 Equity Incentive Plan) and one terminated stock option plan, the 1998 Stock Plan.
Upon its IPO, the Company reserved 838,695 shares of common stock for issuance under a new 2014 Equity Incentive Plan (“2014 Plan”). The shares reserved for issuance under the 2014 Plan also include shares returned to the 2008 Plan as the result of expiration or termination of options, provided that the maximum number of shares that may be added to the 2014 Plan thereby is limited to a maximum of 865,252 shares. The number of shares available for issuance under the 2014 Plan will also include an annual increase on the first day of each year beginning in 2014, equal to the least of:
| • | 357,075 shares; |
| • | 4.0% of the outstanding shares of common stock as of the last day of the immediately preceding year; or |
| • | such other number of shares as the Company’s board of directors may determine. |
97
Table of Contents
The following table summarizes option activity under the plans, and related information:
| Shares Available for Grant |
Stock Options Outstanding |
Weighted- average Exercise Price |
||||||||||
| Balance—December 31, 2011 |
306,188 | 496,908 | $ | 3.70 | ||||||||
| Granted |
(211,056 | ) | 211,056 | 0.55 | ||||||||
| Exercised |
— | (2,924 | ) | 3.36 | ||||||||
| Forfeited |
32,585 | (32,585 | ) | 3.43 | ||||||||
| Expired |
52,549 | (52,549 | ) | 3.49 | ||||||||
|
|
|
|
|
|||||||||
| Balance—December 31, 2012 |
180,266 | 619,906 | 2.67 | |||||||||
| Granted |
(4,081 | ) | 4,081 | 0.48 | ||||||||
| Exercised |
— | (212 | ) | 0.55 | ||||||||
| Forfeited |
7,247 | (7,247 | ) | 2.33 | ||||||||
| Expired |
149,563 | (149,563 | ) | 4.73 | ||||||||
|
|
|
|
|
|||||||||
| Balance—December 31, 2013 |
332,995 | 466,965 | 1.99 | |||||||||
| Additional options authorized |
940,884 | — | — | |||||||||
| Granted |
(585,345 | ) | 585,345 | 11.76 | ||||||||
| Assumed in business combination |
— | 23,229 | 2.06 | |||||||||
| Exercised |
— | (9,363 | ) | 2.14 | ||||||||
| Forfeited |
20,591 | (20,591 | ) | 10.36 | ||||||||
| Expired |
13,781 | (13,781 | ) | 2.74 | ||||||||
|
|
|
|
|
|||||||||
| Balance—December 31, 2014 |
722,906 | 1,031,804 | 7.36 | |||||||||
|
|
|
|
|
|||||||||
Outstanding, exercisable and vested stock options as of December 31, 2014 are summarized as follows:
| Options Outstanding & Exercisable at December 31, 2014 | Options Vested at December 31, 2014 | |||||||||||||||||||
| Range of Exercise Prices |
Number of Options Outstanding |
Weighted Average Remaining Contractual Life (Years) |
Weighted Average Exercise Price |
Number of Options |
Weighted Average Exercise Price |
|||||||||||||||
| $0.27 - 0.55 |
200,002 | 7.80 | $ | 0.55 | 136,560 | $ | 0.55 | |||||||||||||
| $2.05 - 3.01 |
153,469 | 6.36 | 2.78 | 147,641 | 2.77 | |||||||||||||||
| $3.36 - 3.97 |
110,188 | 3.55 | 3.47 | 110,188 | 3.47 | |||||||||||||||
| $7.03 - 12.40 |
568,145 | 9.38 | 11.75 | 21,012 | 11.82 | |||||||||||||||
|
|
|
|
|
|||||||||||||||||
| 1,031,804 | 8.00 | 7.36 | 415,401 | 2.68 | ||||||||||||||||
|
|
|
|
|
|||||||||||||||||
Options outstanding that have vested and are expected to vest at December 31, 2014 are as follows:
| Number of Shares |
Weighted Average Exercise Price |
Weighted Average Remaining Contractual Life (Years) |
Aggregate Intrinsic Value (In thousands) |
|||||||||||||
| Vested |
415,401 | $ | 2.68 | 6.17 | $ | 1,994 | ||||||||||
| Expected to Vest |
616,403 | 10.51 | 9.19 | $ | 457 | |||||||||||
|
|
|
|
|
|||||||||||||
| Total |
1,031,804 | 7.36 | 7.98 | $ | 2,451 | |||||||||||
|
|
|
|
|
|||||||||||||
98
Table of Contents
The aggregate intrinsic value is calculated as the difference between the exercise price of the underlying stock options and the fair value of the Company’s common stock at December 31, 2014 for stock options that were in-the-money. The fair market value of the Company’s common stock as of December 31, 2014 was $7.25 per share.
The weighted average grant-date fair value of options to purchase common stock granted for the years ended December 31, 2014, 2013 and 2012 using the Black-Sholes valuation model was $4.81, $0.21 and $0.27, respectively.
The Company uses the grant date fair value of its common stock to value both employee and non-employee options when granted. The Company revalues non-employee options each reporting period using the fair market value of the Company’s common stock as of the last day of each reporting period.
Valuation Assumptions
The Company’s board of directors determines the estimated fair value of its common stock based on assistance from an independent third party valuation. The fair value of stock-based awards was estimated using the Black-Scholes option-pricing model using the following weighted-average assumptions.
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Expected term (in years) |
5.1 | 6.0 | 6.0 | |||||||||
| Expected volatility |
42.18 | % | 45.25 | % | 46.55 | % | ||||||
| Risk-free interest rate |
1.69 | % | 1.21 | % | 1.01 | % | ||||||
| Expected dividend yield |
— | % | — | % | — | % | ||||||
Risk-free Interest Rate: The Company based the risk-free interest rate over the expected term of the options based on the constant maturity rate of U. S. Treasury securities with similar maturities as of the date of grant.
Volatility: The Company used an average historical stock price volatility of comparable public companies that were deemed to be representative of future stock price trends as the Company does not have any trading history for its common stock.
Expected Term: The expected term represents the period for which the Company’s stock-based awards are expected to be outstanding and is based on analyzing the vesting and contractual terms of the options and the holders’ historical exercise patterns and termination behavior.
Expected Dividends: The Company has not paid and does not anticipate paying any dividends in the near future.
99
Table of Contents
The following table summarizes stock-based compensation expense related to stock options for the years ended December 31, 2013, 2013 and 2012, included in the statements of operations and comprehensive loss as follows (in thousands):
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Cost of testing |
$ | 28 | $ | 3 | $ | 2 | ||||||
| Research and development |
88 | 7 | 13 | |||||||||
| Sales and marketing |
29 | 3 | 5 | |||||||||
| General and administrative |
390 | 59 | 49 | |||||||||
|
|
|
|
|
|
|
|||||||
| $ | 535 | $ | 72 | $ | 69 | |||||||
|
|
|
|
|
|
|
|||||||
If all of the remaining non-vested and outstanding stock option awards that have been granted vested, the Company would recognize approximately $2.2 million in compensation expense, net of estimated forfeitures, over a weighted average remaining period of 3.1 years. No compensation expense will be recognized for any stock options that do not vest.
No tax benefit was recognized related to share-based compensation expense since the Company has never reported taxable income and has established a full valuation allowance to offset all of the potential tax benefits associated with its deferred tax assets. In addition, no amounts of share-based compensation costs were capitalized for the periods presented.
Non-Employee Director Equity-based Compensation
For the year ended December 31, 2014, the Company paid a portion of its non-employee directors’ compensation through the award of common shares. The stock awards are classified as equity-based compensation expense. Expense associated with the awards was $94,000, which was included in general and administrative expense in the statement of operations. The shares issued and associated expense were as follows:
| Service Period Three Months Ended |
Number of Shares Issued |
Fair Market Value per Share at Date of Issuance |
Fair Value of Shares Issued |
|||||||||
| September 30, 2014 |
4,899 | $ | 7.03 | $ | 34,440 | |||||||
| December 31, 2014 |
8,277 | 7.25 | 60,008 | |||||||||
|
|
|
|
|
|||||||||
| Total |
13,176 | $ | 94,448 | |||||||||
|
|
|
|
|
|||||||||
The shares issued at each date were for services performed during the three-month period that ended one day prior to the date of issuance.
100
Table of Contents
12. INCOME TAXES
The Company generated pretax net losses of $719,000, $3.5 million and $5.1 million for the years ended December 31, 2014, 2013 and 2012, respectively. The Company recorded a federal and state deferred tax benefit of $1.5 million and $0 for 2014, respectively, and no benefit for income taxes for 2013 and 2012. A reconciliation of the difference between the benefit for income taxes and income taxes at the statutory U.S. federal income tax rate is as follows:
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Federal tax at statutory rate |
34.0 | % | 34.0 | % | 34.0 | % | ||||||
| Stock based compensation |
-9.5 | % | -11.4 | % | 0.0 | % | ||||||
| Change in valuation allowance |
190.6 | % | -8.6 | % | -30.9 | % | ||||||
| Change in unrecognized tax benefits |
0.0 | % | -8.4 | % | 0.0 | % | ||||||
| Preferred stock warrant revaluation |
-0.7 | % | -5.0 | % | 0.0 | % | ||||||
| Interest expense |
-5.8 | % | 0.0 | % | -2.3 | % | ||||||
| Contingent liability for IMX acquisition |
38.2 | % | 0.0 | % | 0.0 | % | ||||||
| IMX acquisition costs |
-36.7 | % | 0.0 | % | 0.0 | % | ||||||
| Other |
-1.3 | % | -0.6 | % | -0.8 | % | ||||||
|
|
|
|
|
|
|
|||||||
| Effective income tax rate |
208.8 | % | 0.0 | % | 0.0 | % | ||||||
|
|
|
|
|
|
|
|||||||
In connection with the Company’s June 2014 acquisition of ImmuMetrix, Inc., a tax benefit of $1.5 million was recognized during the year ended December 31, 2014. This benefit resulted from the expectation that amortization of the in-process technology acquired, when completed and placed in service, is not expected to be deductible for tax purposes, as the transaction was structured as a tax-free reorganization. Accordingly, a deferred tax liability was recorded at the acquisition date for the difference between the financial reporting and tax basis of the acquired in-process technology. While the in-process technology is considered an indefinite lived intangible asset, this asset is expected to be amortized or impaired prior to the expiration of net operating loss carryforwards available to the Company.
There were significant differences in several of the items affecting the rate reconciliation above. The difference for 2014 primarily relates to the release of a valuation allowance, and for 2013 and 2012 the differences primarily related to the IMX acquisition and to the cancellation of stock options for which deferred taxes were previously provided and an increase in unrecognized tax benefits which were netted against the respective deferred tax assets. These items are separately disclosed and not included with the change in the valuation allowance.
101
Table of Contents
The tax effects of temporary differences and carryforwards that give rise to significant deferred tax assets and liabilities are as follows (in thousands):
| As of December 31, | ||||||||
| 2014 | 2013 | |||||||
| Deferred tax assets: |
||||||||
| Net operating loss carryforwards |
$ | 63,116 | $ | 62,863 | ||||
| Tax credit carryforwards |
4,065 | 3,973 | ||||||
| Accruals |
942 | 1,097 | ||||||
| Property and equipment |
— | 147 | ||||||
| Other |
551 | 97 | ||||||
|
|
|
|
|
|||||
| Gross deferred tax assets |
68,674 | 68,177 | ||||||
|
|
|
|
|
|||||
| Deferred tax liabilities: |
||||||||
| Property and equipment |
(8 | ) | — | |||||
| Purchased Intangible |
(2,349 | ) | — | |||||
|
|
|
|
|
|||||
| Total deferred tax liabilities |
(2,357 | ) | — | |||||
|
|
|
|
|
|||||
| Valuation allowance |
(66,317 | ) | (68,177 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax assets |
$ | — | $ | — | ||||
|
|
|
|
|
|||||
The Company has recorded pretax net losses from operations since its inception. The Company believes that based on the history of such losses and other factors, the weight of available evidence indicates that it is more likely than not that it will not be able to realize its net deferred tax assets. Accordingly, the net deferred tax assets have been offset by a full valuation allowance. The valuation allowance decreased by $1.9 million and $0.5 million during the years ended December 31, 2014 and 2013, respectively.
As of December 31, 2014, the Company had net operating loss carryforwards of approximately $164 million and $127 million available to reduce future taxable income, if any, for federal and state income tax purposes, respectively. The U.S. federal net operating loss carryforwards will begin to expire in 2018 while for state purposes, the net operating losses will begin to expire in 2015.
As of December 31, 2014, the Company had credit carryforwards of approximately $3.1 million and $3.9 million available to reduce future taxable income, if any, for federal and California state income tax purposes, respectively. The Federal credit carryforwards begin to expire in 2021. California credits have no expiration date.
Utilization of the Company’s net operating loss carryforwards and credits may be subject to a substantial annual limitation due to the ownership change limitations provided by the Tax Reform Act of 1986, as amended and similar state provisions. The annual limitation may result in the expiration of net operating losses and credits before utilization. Based on a preliminary review of our equity transactions since inception, the Company believes a portion of its net operating loss carryforwards may be limited due to equity financings which occurred in 2000, 2004, 2007 and 2014.
A reconciliation of the Company’s unrecognized tax benefits is as follows (in thousands):
| Year Ended December 31, | ||||||||||||
| 2014 | 2013 | 2012 | ||||||||||
| Balance at beginning of year |
$ | 2,196 | $ | 1,159 | $ | 1,130 | ||||||
| Additions based on tax positions related to current year |
83 | 177 | 53 | |||||||||
| Additions (reductions) based on tax positions related to prior years |
(225 | ) | 860 | (24 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Balance at end of year |
$ | 2,054 | $ | 2,196 | $ | 1,159 | ||||||
|
|
|
|
|
|
|
|||||||
102
Table of Contents
The unrecognized tax benefits, if recognized and in the absence of a full valuation allowance, would impact the income tax provision by $1.6 million and $1.5 million as of December 31, 2014 and 2013, respectively. Given the Company’s valuation allowance, the uncertain tax positions would not impact the effective tax rate.
The Company has elected to include interest and penalties as a component of tax expense. During the years ended December 31, 2014, 2013 and 2012, the Company did not recognize accrued interest and penalties related to unrecognized tax benefits. The Company does not anticipate a significant change in the unrecognized tax benefits over the next twelve months.
Because the Company has not utilized any of its net operating loss carryforwards, its federal and state income tax returns are subject to tax authority examination from inception.
13. 401(K) PLAN
The Company sponsors a 401(k) defined contribution plan covering all employees. To date, there have been no employer contributions to the plan.
14. BUSINESS COMBINATION
On June 10, 2014, in accordance with an agreement and plan of merger, the Company acquired ImmuMetrix, Inc. (“IMX”), a privately held development stage company working in new technologies using cell-free donor DNA (“cfDNA”) technology for the diagnosis, treatment and management of transplant rejection, immune disorders and diseases, including the development of a new, non-invasive test designed to detect the early stages of solid organ transplant rejection. The Company acquired all IMX assets associated with transplant diagnostics, including related immune repertoire and infectious diseases. An IMX successor company retained the limited assets not associated with transplant diagnostics. The acquisition was structured as a tax-free reorganization.
The Company acquired all of the issued and outstanding capital stock of IMX for the total estimated purchase price of $17.2 million consisting of $600,000 in cash; 911,364 shares of the Company’s Series G convertible preferred stock with an estimated fair value of $14.2 million, including 23,229 shares of the Company’s Series G convertible preferred stock with an estimated fair value of $369,000 as a result of the Company’s assumption of IMX outstanding stock options; and an additional payment of 227,845 shares of CareDx Series G convertible preferred stock if a future milestone is achieved. The Agreement provides that the milestone will be achieved if the Company completes 2,500 commercial tests involving the measurement of cfDNA in organ transplant recipients in the United States no later than six years after the closing date of the acquisition. All shares of Series G Preferred Stock and options to acquire Series G Preferred Stock converted into common stock and options to acquire common stock immediately prior to the closing of the Company’s initial public offering. The additional shares to be paid for the achievement of the milestone will also be issued in common stock. The fair value of this contingent consideration was $2.3 million at the acquisition date and $1.1 million at December 31, 2014.
The intellectual property acquired includes an exclusive license from Stanford University to a patent relating to the diagnosis of rejection in organ transplant recipients using cfDNA. The license provides for the Company to pay royalties to Stanford University on sales of the Company’s cfDNA tests.
Assets acquired in the business combination consist of In-Process Technology, for which estimated fair value was $6.7 million at the date of acquisition, and goodwill, for which estimated fair value was $12.0 million at the date of acquisition.
The in-process technology is recorded as an indefinite-life intangible asset until it reaches technological feasibility and will be tested for impairment in accordance with ASC 350, Intangibles-Goodwill and Other. Amortization into earnings will begin once the research and development activities are complete and the technology is proven to work, at which time technological feasibility will have been achieved. The Company
103
Table of Contents
expects that will occur at approximately the time when revenue is generated in the marketplace, currently estimated to be during 2017. Amortization will be based on the estimated remaining useful life of the patent when the product is proven feasible, estimated to be 15 years. Amortization will be recorded using the straight line method. Accordingly, at December 31, 2014, accumulated amortization of the in-process technology intangible asset was $0. Given that amortization has not yet begun and technological feasibility has not yet occurred, we cannot currently estimate amortization of the in-process technology asset during each of the next five years.
The goodwill recorded from the acquisition of ImmuMetrix is primarily related to expected synergies. Substantially all of the goodwill recognized is not deductible for tax purposes.
IMX’s post-acquisition results of operations for the period from June 11, 2014 through December 31, 2014 are included in the Company’s statements of operations.
Pro Forma Impact of the Acquisition of IMX
The following table presents pro forma results of operations and gives effect to the IMX transaction as if the transaction had been consummated on January 1, 2013. The unaudited pro forma results of operations have been prepared for comparative purposes only and are not necessarily indicative of what would have occurred had the business combination been completed at the beginning of the period or of the results that may occur in the future. Furthermore, the pro forma financial information does not reflect the impact of any reorganization or operating efficiencies resulting from combining the two companies.
| Year Ended December 31, | ||||||||
| (in thousands, except per share data). | 2014 | 2013 | ||||||
| Net revenue |
$ | 27,306 | $ | 22,098 | ||||
|
|
|
|
|
|||||
| Net loss |
$ | (1,080 | ) | $ | (3,768 | ) | ||
|
|
|
|
|
|||||
| Net loss per common share—basic |
$ | (0.19 | ) | $ | (3.73 | ) | ||
|
|
|
|
|
|||||
| Net loss per common share—diluted |
$ | (0.19 | ) | $ | (3.73 | ) | ||
|
|
|
|
|
|||||
The unaudited pro forma consolidated financial information was prepared using the acquisition method of accounting and is based on the historical financial information of the Company and IMX, reflecting the Company’s and IMX’s results of operations for the years ended December 31, 2014 and 2013. The historical financial information has been adjusted to give effect to the pro forma events that are: (i) directly attributable to the acquisition, (ii) factually supportable and (iii) expected to have a continuing impact on the combined results. The unaudited pro forma consolidated financial information reflects: (a) the removal of acquisition-related costs of $1.7 million incurred by both CareDx and IMX for the year ended December 31, 2014 including the removal of $0.2 million of IMX stock-based compensation expense that resulted from modifications to options in anticipation of the acquisition; (b) the removal of a $1.5 million tax benefit for the year ended December 31, 2014 that resulted from the acquisition; (c) the addition of salaries, benefits and fees for IMX employees and consultants retained after the acquisition; and (d) the addition of the $1.5 million acquisition-related tax benefit for the year ended December 31, 2013, as if the acquisition had occurred on January 1, 2013 and the benefit had been recognized during the year ended December 31, 2013. Acquisition related expenses are primarily included in general and administrative expenses.
Finalization of preliminary purchase price allocation
Under U.S. GAAP, the period that is allowed for finalizing the identification and measurement of the fair value of the assets acquired and the liabilities assumed in a business combination ends when the acquiring entity is no longer waiting for information that it has arranged to obtain and that is known to be available or obtainable. At
104
Table of Contents
September 30, 2014, the Company determined that its measurement and recognition of assets acquired and liabilities assumed in the IMX acquisition was recorded on a final basis.
15. SELECTED QUARTERLY FINANCIAL DATA (UNAUDITED)
The following table presents selected unaudited consolidated financial data for each of the eight quarters in the two-year period ended December 31, 2014. The Company believes this information reflects all recurring adjustments necessary to fairly present this information when read in conjunction with the Company’s financial statements and the related notes. Net income (loss) per share, basic and diluted, for the four quarters of each fiscal year may not sum to the total for the fiscal year because of the different number of shares outstanding during each period. The results of operations for any quarter are not necessarily indicative of the results to be expected for any future period.
| Quarter Ended: | March 31 | June 30 | September 30 | December 31 | ||||||||||||
| (In thousands, except share and per share data) | ||||||||||||||||
| 2014 |
||||||||||||||||
| Total revenue |
$ | 5,924 | $ | 6,776 | $ | 6,654 | $ | 7,952 | ||||||||
| Net income (loss) |
$ | (1,304 | ) | $ | 877 | $ | 1,213 | $ | (5 | ) | ||||||
| Net income (loss) per common share, basic |
$ | (1.29 | ) | $ | 0.87 | $ | 0.13 | $ | — | |||||||
| Net income (loss) per common share, diluted |
$ | (1.29 | ) | $ | 0.13 | $ | 0.12 | $ | — | |||||||
| Shares used in calculation of net income (loss) per share, basic |
1,011,980 | 1,013,128 | 9,279,649 | 11,802,241 | ||||||||||||
| Shares used in calculation of net income (loss) per share, diluted |
1,011,980 | 6,939,568 | 11,219,377 | 11,802,241 | ||||||||||||
| 2013 |
||||||||||||||||
| Total revenue |
$ | 4,981 | $ | 5,457 | $ | 5,805 | $ | 5,855 | ||||||||
| Net loss |
$ | (1,348 | ) | $ | (802 | ) | $ | (830 | ) | $ | (562 | ) | ||||
| Net loss per common share, basic and diluted |
$ | (1.33 | ) | $ | (0.79 | ) | $ | (0.82 | ) | $ | (0.56 | ) | ||||
| Shares used in calculation of net loss per share, basic and diluted |
1,010,684 | 1,011,123 | 1,011,136 | 1,011,136 | ||||||||||||
16. SUBSEQUENT EVENT
On January 30, 2015, the Company entered into a Loan and Security Agreement (the “Loan Agreement”) which provides a secured term loan facility in an aggregate principal amount of up to $20.0 million. The Company borrowed the first advance of $16.0 million (“Draw A”) on January 30, 2015. Under the terms of the Loan Agreement, following a six month period from the closing date and until any time before December 31, 2015, the Company may, at its option, borrow from the lender a second advance of $4.0 million (“Draw B”), subject to the Company’s satisfaction of certain conditions described in the Loan Agreement. Draw A was used to pay-off the Company’s existing term debt of $11.3 million. Draw A and Draw B each bear interest at a floating rate equal to 2.00%, plus the greater of (i) 3.25% or (ii) the prime rate published by the lender.
In connection with the Loan Agreement, the Company agreed to issue to the lender warrants to purchase shares of the Company’s common stock upon the drawdown of each advance in an amount equal to 1.5% of the amount drawn, divided by the exercise price per share for that tranche. As a result of Draw A, the Company issued to the lender a warrant to purchase an aggregate of 34,483 shares of the Company’s common stock, at an exercise price equal to $6.96 per share.
105
Table of Contents
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
| ITEM 9A. | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures as of December 31, 2014. The term “disclosure controls and procedures,” as defined in Rule 13a-15(e) under the Securities Exchange Act of 1934, as amended (Exchange Act) means controls and other procedures of a company that are designed to ensure that information required to be disclosed by us in reports that we file or submit under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in Securities and Exchange Commission rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company in the reports that it files or submits under the Exchange Act is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating our disclosure controls and procedures, management recognized that disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the disclosure controls and procedures are met. Our disclosure controls and procedures have been designed to meet reasonable assurance standards. Additionally, in designing disclosure controls and procedures, our management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible disclosure controls and procedures. The design of any disclosure controls and procedures also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Management and our independent registered public accounting firm had previously identified a material weakness in our internal control over financial reporting with our preliminary purchase accounting and supporting analyses related to our acquisition of ImmuMetrix, Inc. A “material weakness” is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented or detected and corrected on a timely basis. The material weakness related to our internal control over financial reporting pertaining to business combinations processes that were not adequately designed and therefore not operating effectively. Specifically, the material weaknesses involved aspects of our proposed purchase accounting for our ImmuMetrix, Inc. acquisition that required adjustment, including adjustments to valuation of in-process technology, deferred income tax liability related to acquired in-process technology, goodwill, share-based compensation and recording of transaction costs.
In response to this material weakness, we implemented numerous measures designed to improve our internal control over financial reporting. These measures include:
| • | Continued training on accounting processes and procedures, including those relating to business combinations; |
| • | The recent hiring of a corporate controller with over 10 years of experience at a “Big 4” public accounting firm, with experience with business combinations processes and in preparing periodic reports filed under the Securities Exchange Act; |
| • | Increased rigor in management review procedures and additional consultation with subject matter experts in areas of accounting complexity such as business combinations; |
| • | Continued experience with purchase accounting for the Immumetrix, Inc. acquisition in subsequent quarterly periods; |
| • | Use of third party advisors with appropriate expertise, as necessary; and |
| • | The hiring of additional general accounting staff. |
106
Table of Contents
We believe that our remediation efforts have been effective and that the previously identified material weakness in our internal controls has been remediated.
Based on their evaluation as of the end of the period covered by this Annual Report on Form 10-K, our Chief Executive Officer and Chief Financial Officer have concluded that, as of such date, our disclosure controls and procedures were effective at the reasonable assurance level.
Changes in Internal Control over Financial Reporting
Except for the remediation efforts described above, there were no other changes to our internal control over financial reporting identified in connection with the evaluation required by rule 13a-15(d) and 15d-15(d) of the Exchange Act that occurred during the quarter ended December 31, 2014 that has materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Management’s Report on Internal Control over Financial Reporting
This Annual Report on Form 10-K does not include a report of management’s assessment regarding internal control over financial reporting or an attestation report of our registered public accounting firm due to a transition period established by rules of the Securities and Exchange Commission for newly public companies.
| ITEM 9B. | OTHER INFORMATION |
None.
107
Table of Contents
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The information required by this item is incorporated by reference to our Proxy Statement for our 2015 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days after the end of the fiscal year ended December 31, 2014.
We have adopted a Code of Business Conduct and Ethics that applies to all of our officers and employees (including our principal executive officer, principal financial officer, principal accounting officer or controller and other employees who perform financial or accounting functions), agents and representatives, including our independent directors and consultants, who are not employees of ours, with regard to their CareDx-related activities. Our code of business conduct and ethics is available on our website at www.caredx.com under the heading “Compliance” under the section titled “Company”. We will post on this section of our website any amendment to our code of business conduct and ethics, as well as any waivers of our code of business conduct and ethics, that are required to be disclosed by the rules of the SEC or the NASDAQ Stock Market.
ITEM 11. EXECUTIVE COMPENSATION
The information required by this item is incorporated by reference to our Proxy Statement for our 2015 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days after the end of the fiscal year ended December 31, 2014.
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The information required by this item is incorporated by reference to our Proxy Statement for our 2015 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days after the end of the fiscal year ended December 31, 2014.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE
The information required by this item is incorporated by reference to our Proxy Statement for our 2015 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days after the end of the fiscal year ended December 31, 2014.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The information required by this item is incorporated by reference to our Proxy Statement for our 2015 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission within 120 days after the end of the fiscal year ended December 31, 2014.
108
Table of Contents
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
Documents filed as part of this report are as follows:
| 1. | Consolidated Financial Statements: |
Our Consolidated Financial Statements are listed in the “Index to Financial Statements” of CareDx, Inc. in Part II, Item 8 of this Annual Report on Form 10-K.
| 2. | Financial Statement Schedules |
All financial statement schedules have been omitted because they are not required, not applicable, or the required information is included in the financial statements or notes thereto included in this Annual Report on Form 10-K.
| 3. | Exhibits |
The documents listed in the Exhibit Index of this Annual Report on Form 10-K are incorporated by reference or are filed with this report, in each case as indicated therein (numbered in accordance with Item 601 of Regulation S-K).
109
Table of Contents
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| CAREDX, INC. | ||
| By: | /s/ PETER MAAG | |
| Peter Maag President and Chief Executive Officer | ||
Date: March 31, 2015
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Peter Maag and Ken Ludlum, and each of them, his true and lawful attorneys-in-fact, each with full power of substitution, for him or her in any and all capacities, to sign any amendments to this Annual Report on Form 10-K and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, hereby ratifying and confirming all that each of said attorneys-in-fact or their substitute or substitutes may do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons, on behalf of the registrant on the dates and the capacities indicated.
| Signature |
Title |
Date | ||
| /s/ PETER MAAG Peter Maag |
Chief Executive Officer, President and Director (Principal Executive Officer) |
March 31, 2015 | ||
| /s/ KEN LUDLUM Ken Ludlum |
Chief Financial Officer (Principal Financial and Accounting Officer) |
March 31, 2015 | ||
| /s/ MICHAEL GOLDBERG Michael Goldberg |
Director | March 31, 2015 | ||
| /s/ GEORGE W. BICKERSTAFF George W. Bickerstaff |
Director | March 31, 2015 | ||
| /s/ BROOK BYERS Brook Byers |
Director | March 31, 2015 | ||
| /s/ FRED E. COHEN Fred E. Cohen |
Director | March 31, 2015 | ||
| /s/ RALPH SNYDERMAN Ralph Snyderman |
Director | March 31, 2015 | ||
| /s/ WILLIAM HAGSTROM William Hagstrom |
Director | March 31, 2015 | ||
110
Table of Contents
| Exhibit | Incorporated by Reference | |||||||||
| Number |
Description |
Form |
File No. |
Exhibit |
Filing Date | |||||
| 2.1† | Agreement and Plan of Merger, dated May 17, 2014, by and between Registrant, Monitor Acquisition Corporation, ImmuMetrix, Inc. and Mattias Westman, as Holders’ Agent. | S-1/A | 333-196494 | 2.1 | 07/15/2015 | |||||
| 2.2 | Amendment No. 1 to Agreement and Plan of Merger, dated June 9, 2014, by and between the Registrant, Monitor Acquisition Corporation, ImmuMetrix, Inc. and Mattias Westman, as Holders’ Agent. | S-1/A | 333-196494 | 2.2 | 06/25/2014 | |||||
| 3.1 | Amended and Restated Certificate of Incorporation of the Registrant. | 10-Q | 001-36536 | 3.1 | 08/28/2014 | |||||
| 3.2 | Amended and Restated Bylaws of the Registrant. | 10-Q | 001-36536 | 3.4 | 08/28/2014 | |||||
| 4.1* | Form of Registrant’s common stock certificate. | |||||||||
| 4.2* | Sixth Amended and Restated Investors Rights Agreement, dated July 1, 2009, as amended on March 29, 2012, June 10, 2014, and July 14, 2014, between the Registrant and certain holders of the Registrant’s capital stock named therein. | |||||||||
| 4.3# | 1998 Equity Incentive Plan and forms of agreements thereunder. | S-1 | 333-196494 | 10.2 | 06/03/2014 | |||||
| 4.4# | 2008 Equity Incentive Plan and forms of agreements thereunder. | S-1 | 333-196494 | 10.3 | 06/03/2014 | |||||
| 4.5# | 2014 Equity Incentive Plan and forms of agreements thereunder. | S-8 | 333-197493 | 4.4 | 07/18/2014 | |||||
| 4.6# | 2014 Employee Stock Purchase Plan and forms of agreements thereunder. | S-8 | 333-197493 | 4.5 | 07/18/2014 | |||||
| 4.7# | ImmuMetrix, Inc. 2013 Equity Incentive Plan | S-1 | 333-196494 | 10.19 | 06/03/2014 | |||||
| 10.1# | Chief Executive Employment Agreement, dated September 19, 2012, by and between the Registrant and Peter Maag. | S-1 | 333-196494 | 10.6 | 06/03/2014 | |||||
| 10.2# | Offer Letter, dated July 31, 2006, by and between the Registrant and James Yee. | S-1 | 333-196494 | 10.7 | 06/03/2014 | |||||
| 10.3# | Offer Letter, dated July 19, 2010, by and between the Registrant and Matthew Meyer. | S-1 | 333-196494 | 10.8 | 06/03/2014 | |||||
| 10.4# | Offer Letter, dated March 18, 2014, by and between the Registrant and Ken Ludlum. | S-1 | 333-196494 | 10.9 | 06/03/2014 | |||||
| 10.5# | Offer Letter, dated November 21, 2006, by and between the Registrant and Mitchell Nelles. | S-1 | 333-196494 | 10.10 | 06/03/2014 | |||||
| 10.6# | Form of Change of Control and Severance Agreement between the Registrant and each of its executive officers. | S-1 | 333-196494 | 10.11 | 06/03/2014 | |||||
Table of Contents
| Exhibit | Incorporated by Reference | |||||||||||
| Number |
Description |
Form |
File No. |
Exhibit |
Filing Date |
|||||||
| 10.7# | Form of Indemnification Agreement between the Registrant and each of its directors and executive officers. | S-1 | 333-196494 | 10.1 | 06/03/2014 | |||||||
| 10.8 | Lease, dated April 27, 2006, as amended on November 10, 2010, by and between the Registrant and BMR-Bayshore Boulevard LLC, for office and laboratory space located at 3260 Bayshore Boulevard, Brisbane, California 94005. | S-1 | 333-196494 | 10.12 | 06/03/2014 | |||||||
| 10.9† | PCR Patent License Agreement, dated November 16, 2004, by and between the Registrant and Roche Molecular Systems, Inc., and amendments thereto. | S-1 | 333-196494 | 10.14 | 06/03/2014 | |||||||
| 10.10† | Distribution and Licensing Agreement, dated June 20, 2013, by and between the Registrant and Diaxonhit SA. | S-1/A | 333-196494 | 10.15 | 06/25/2014 | |||||||
| 10.11† | Amended and Restated Exclusive Agreement, dated January 27, 2014, by and between the Board of Trustees of the Leland Stanford Junior University and ImmuMetrix, Inc. | S-1/A | 333-196494 | 10.17 | 07/15/2014 | |||||||
| 10.12† | Settlement Agreement and Mutual Release, dated September 11, 2014, by and between the Registrant and Roche Molecular Systems, Inc. | 10-Q | 001-36536 | 10.14.1 | 11/14/2014 | |||||||
| 10.13 | Loan and Security Agreement, dated as of January 30, 2015, by and between CareDx, Inc. and East West Bank. | 8-K | 001-36536 | 10.1 | 02/04/2015 | |||||||
| 10.14*# | Offer Letter, dated April 8, 2014, by and between the Registrant and George Bickerstaff. | |||||||||||
| 10.15*# | Offer Letter, dated October 18, 2011, by and between the Registrant and Michael Goldberg. | |||||||||||
| 10.16*# | Offer Letter, dated December 3, 2014, by and between the Registrant and John Sninsky. | |||||||||||
| 10.17*# | Offer Letter, dated March 11, 2015 by and between the Registrant and Josh DeFonzo. | |||||||||||
| 10.18*# | Outside Director Compensation Policy. | |||||||||||
| 10.19*# | Executive Incentive Compensation Plan. | |||||||||||
| 24.1* | Power of Attorney (see page 110 of this Annual Report on Form 10-K). | |||||||||||
| 31.1* | Principal Executive Officer’s Certifications Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |||||||||||
| 31.2* | Principal Financial Officer’s Certifications Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002. | |||||||||||
| 32.1** | Certification Pursuant to 18 U.S.C. § 1350 (Section 906 of Sarbanes-Oxley Act of 2002). | |||||||||||
Table of Contents
| Exhibit | Incorporated by Reference | |||||||||
| Number |
Description |
Form |
File No. |
Exhibit |
Filing Date | |||||
| 101.INS | XBRL Instance Document | |||||||||
| 101.SCH | XBRL Taxonomy Extension Schema | |||||||||
| 101.CAL | XBRL Taxonomy Extension Calculation Linkbase | |||||||||
| 101.DEF | XBRL Taxonomy Extension Definition Linkbase | |||||||||
| 101.LAB | XBRL Taxonomy Extension Label Linkbase | |||||||||
| 101.PRE | XBRL Taxonomy Extension Presentation Linkbase | |||||||||
| † | Confidential treatment has been granted with respect to certain portions of this Exhibit. |
| # | Indicates management contract or compensatory plan or arrangement. |
| * | Filed herewith. |
| ** | Furnished herewith. |
