Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - Sorrento Therapeutics, Inc. | d850742d8k.htm |
| EX-99.1 - EX-99.1 - Sorrento Therapeutics, Inc. | d850742dex991.htm |
| Exhibit 99.2
|
Exhibit 99.2
Next-Generation Cancer Therapeutics
January 2015
|
|
Safe Harbor Statement NASDAQ: SRNE
This presentation contains “forward-looking statements” as that term is defined under the Private Securities Litigation Reform Act of 1995 (PSLRA), including statements regarding expectations, beliefs or intentions regarding our business, technologies and products strategies or prospects. Such forward-looking statements are characterized by future or conditional verbs such as “may,” “will,” “expect,” “intend,” “anticipate,” “believe,” “estimate” and “continue” or similar verbs.
Actual results may differ from those projected due to a number of risks and uncertainties, including, but not limited to, the possibility that some or all of the pending matters and transactions being considered by the Company may not proceed as contemplated, as well as risks inherent in additional financing, developing and obtaining regulatory approvals of new, commercially-viable and competitive products and product candidates, including timelines, the size of clinical trials, sufficiency of data from those trials and the requirements of the FDA for potential approval of Cynviloq™ and by all other matters described in the Company’s filings with the Securities and Exchange Commission, including the risk factors set forth therein. These statements are made based upon current expectations that are subject to risk and uncertainty and information available to the Company as of the date of this presentation. The Company does not undertake to update forward-looking statements in this presentation to reflect actual results, changes in assumptions or changes in other factors affecting such forward-looking information. Assumptions and other information that could cause results to differ from those set forth in the forward-looking information can be found in the Company’s filings with the Securities and Exchange Commission, including its most recent periodic report. We intend that all forward-looking statements be subject to the safe-harbor provisions of the PSLRA.
| 2 |
|
|
|
A Comprehensive Oncology Company
Deep and Complementary Pipeline Creates Significant Opportunities-
Novel breakthrough combination therapeutic regimens and modalities to attack cancer Significant reduction in clinical development costs and timeline Significant commercial edge in future drug pricing
Small Biologics Cell Supportive
Molecules Therapy Care
Cytotoxics Immunotherapy RTX
CYNVILOQ™ PD-1, PD-L1, CTLA-4
Bispecific Abs Intractable
Cancer Pain
Targeted Therapy Targeted Therapy Adoptive Cellular
MYC inhibitor Anti-VEGR2 ADC Immunotherapy TRAIL modulator Anti-c-MET ADC Chimeric Antigen Receptor Tumor-attacking Neukoplast® Bispecific ADC (Partnership with Conkwest)
CYNVILOQ, CAR.TNK, CAR.TNK (Chimeric Antigen Receptor Tumor-attacking Neukoplast) are trademarks owned by Sorrento Therapeutics, Inc. Neukoplast is a trademark owned by Conkwest, Inc.
| 3 |
|
|
|
Corporate Events Validate and Advance Sorrento
Pipeline Unlocking Significant Value
Exclusive global partnership with Conkwest to develop next generation anti-cancer
cellular immunotherapy with “Off-the-Shelf” CAR.TNK™ (Chimeric Antigen Receptor
Tumor-attacking NeuKoplast)
“The First joint venture with NantWorks and Abraxis BioScience Inc. founder, Dr. Patrick
Immunotherapy Soon-Shiong, to develop next generation immunotherapies for the treatment of cancer and
Antibody
Company” autoimmune disease.
Pilot PK data from TRIBECA study suggest bioequivalence (BE) between Cynviloq and
CYNVILOQ albumin-bound paclitaxel
Licensing agreement to develop and commercialize anti-PD-L1 mAb with Lee’s
Pharmaceutical for greater Chinese Market
Exclusive research and option agreement to generate and develop antibody -drug
conjugates (ADCs) with Morphotek / Eisai
| 4 |
|
|
|
Deep and Complementary Pipeline Creates
Significant Opportunities
INDICATION > TARGET PRECLINICAL PHASE 1 PHASE 2 PHASE 3
Metastatic Breast Cancer T R I B E C A
CYNVILOQ™ Non-Small Cell Lung Cancer 505(b)(2) Bioequivalence versus nab -Paclitaxel*
RTX Intractable Cancer Pain
G-MAB Immuno-oncology > PD-L1, PD1, CD47, CD137
Bi-Specific Ab PD-L1/c-MET; PD-L1/CTLA-4, PD-L1/EGFR
PD-L1.TNK, CD123.TNK, ROR1.TNK, PSMA.TNK
ADC VEGFR2, c-MET, CXCR5
MYC Inhibitor Solid tumors and hematological malignancies
*Abraxane® (paclitaxel albumin-bound particles for injectable suspension) (albumin-bound), registered trademark of and marketed by Celgene Corp.
PDL1.TNK, CD123.TNK, ROR1.TNK, PSMA.TNK are trademarks owned by Sorrento Therapeutics, Inc.
| 5 |
|
|
|
Lead Oncology Product Opportunity
Cynviloq (Paclitaxel polymeric micelle)
Registration Trial
| 6 |
|
|
|
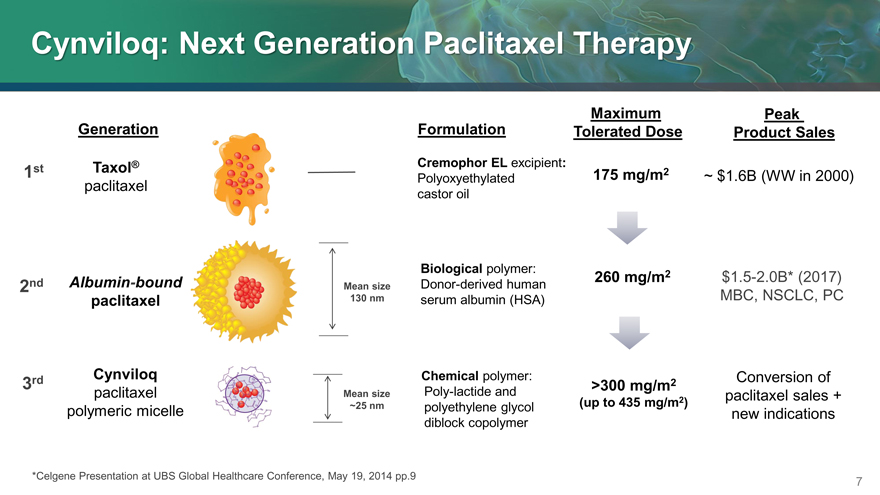
Cynviloq: Next Generation Paclitaxel Therapy
Maximum Peak
Generation Formulation Tolerated Dose Product Sales
1st Taxol® Cremophor EL excipient: 175 mg/m2 ~ $1.6B (WW in 2000)
paclitaxel Polyoxyethylated
castor oil
Biological polymer:
2nd Albumin-bound Mean size Donor-derived human 260 mg/m2 $1.5-2.0B* (2017)
paclitaxel 130 nm serum albumin (HSA) MBC, NSCLC, PC
3rd Cynviloq Chemical polymer: >300 mg/m2 Conversion of
paclitaxel Mean size Poly-lactide and paclitaxel sales +
~25 nm polyethylene glycol (up to 435 mg/m2)
polymeric micelle new indications
diblock copolymer
*Celgene Presentation at UBS Global Healthcare Conference, May 19, 2014 pp.9
| 7 |
|
|
|
Cynviloq Clinical Development Summary
Total number of patients across all trials: 1,260
Trials established MTD at >300 mg/m2—Dana Farber Cancer Inst, Russia, & S. Korea
Phase 1: (total n=80)
>300 mg/m2 (q3w) vs. 175 mg/m2 (Taxol; weekly)
Phase 2: Completed trials in MBC, NSCLC, PC, OC, BC; in US—Yale Cancer Center, Russia, S. Korea (total n=259)
Possible Phase 3 sNDA programs in these tumor types
Phase 2b*: Chemo-naïve Stage IIIb/IV NSCLC vs Taxol in S. Korea (total n=276; Cynviloq n=140)
230 mg/m2 + cis (q3w) vs. Taxol 175 mg/m2 + cis; non -inferiority established
Phase 2*: 1st line treatment of OC vs Taxol in S. Korea (total n=100; Cynviloq n=50)
260 mg/m2 + carbo (q3w) vs. Taxol 175 mg/m2 + carbo; non -inferiority established
MBC in S. Korea (total n=209; Cynviloq n=105 vs Taxol n=104)
Phase 3: GPMBC301. An Open -label, Randomized, Parallel, Phase 3 Trial to Evaluate the Efficacy and Safety of Cynviloq compared to Genexo l® (Paclitaxel
with Cremophor EL) in Subjects with Recurrent or Metastatic Breast Cancer)
PM-Safety: Completed for MBC and NSCLC (total n=502)
Efficacy and safety data supportive of 505(b)(2) submission
Data on file; * Investigator Initiated Study
| 8 |
|
|
|
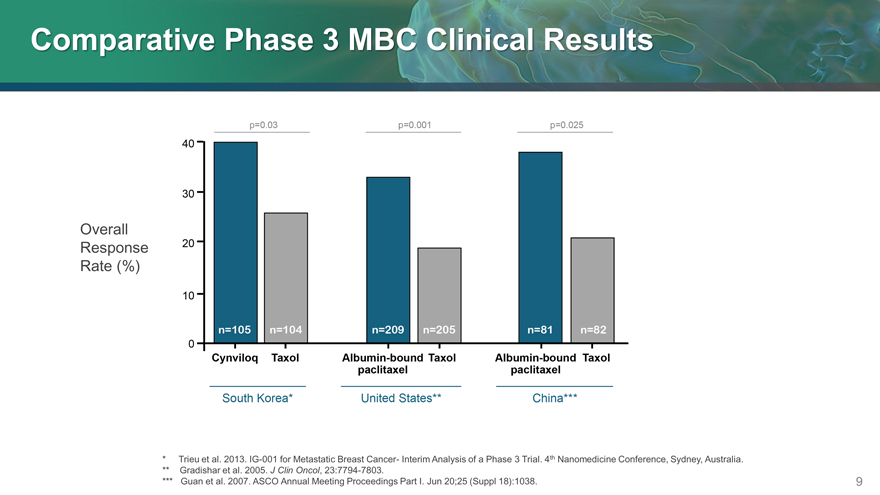
Comparative Phase 3 MBC Clinical Results
Overall
Response
Rate (%)
* Trieu et al. 2013. IG-001 for Metastatic Breast Cancer- Interim Analysis of a Phase 3 Trial. 4th Nanomedicine Conference, Sydney, Australia.
** Gradishar et al. 2005. J Clin Oncol, 23:7794-7803.
*** Guan et al. 2007. ASCO Annual Meeting Proceedings Part I. Jun 20;25 (Suppl 18):1038.
9
|
|
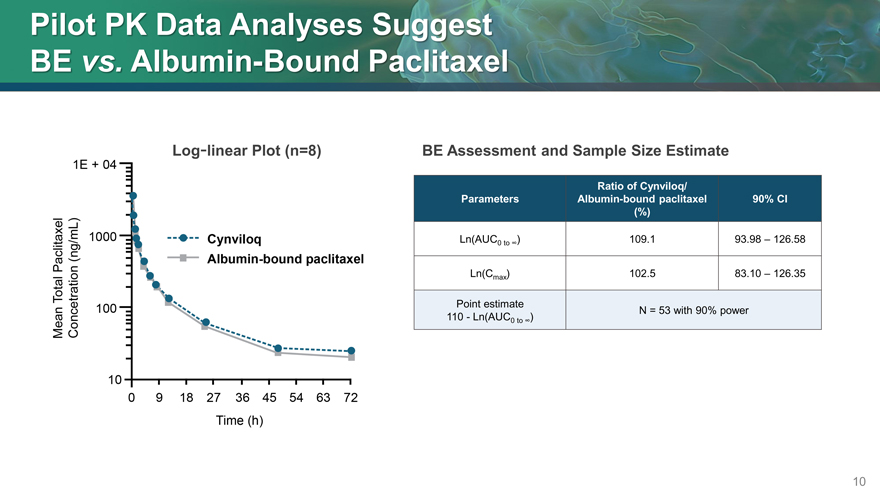
Pilot PK Data Analyses Suggest
BE vs. Albumin-Bound Paclitaxel
Log-linear Plot (n=8)
BE Assessment and Sample Size Estimate
Ratio of Cynviloq/
Parameters Albumin-bound paclitaxel 90% CI
(%)
Ln(AUC0 to ) 109.1 93.98 – 126.58
Ln(Cmax ) 102.5 83.10 – 126.35
Point estimate N = 53 with 90% power
110—Ln(AUC0 to )
10
|
|
Bioequivalence = Accelerated Pathway to Market
T R I B E C A™
(TRIal establishing BioEquivalence between Cynviloq™ and Albumin-bound paclitaxel)
- Patients with MBC
Key Parameters:
Albumin-bound Cynviloq
Cycle 1 paclitaxel Dose: 260 mg/m2
(n = 27)*
(n = 27)* Infusion time: 30 min
Duration: 3 weeks +
Albumin-bound crossover for 3 weeks
Cycle 2 Cynviloq
paclitaxel Endpoints: AUC and Cmax
(90% CI)
Note: Previous trial size estimate of 100 patients was based on PK simulation of albumin-bound paclitaxel and
Cynviloq historical data with both drugs given at different doses and infusion rates. Based on the recent positive initial PK data and subject to FDA guidance, 53 patients may be sufficient to establish BE.
T R I B E C A™ (TRIal establishing BioEquivalence between Cynviloq™ and Albumin-bound paclitaxel) is a trademark owned by Sorrento Therapeutics, Inc.
11
|
|
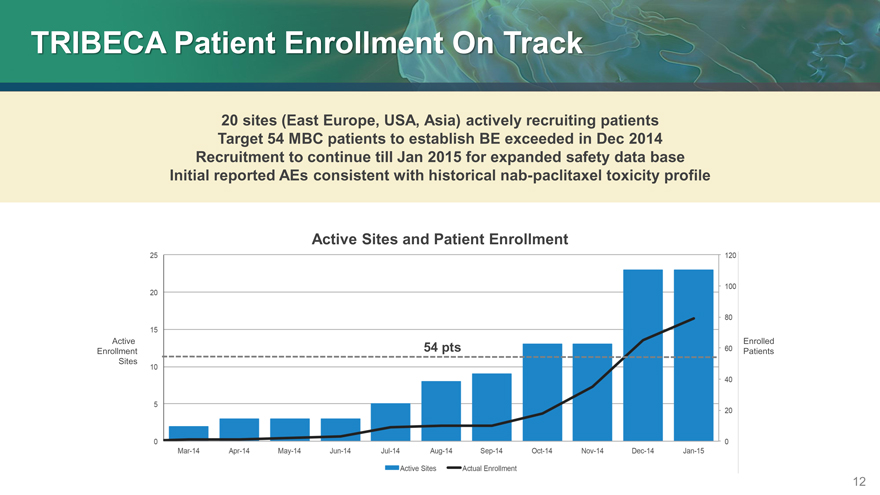
TRIBECA Patient Enrollment On Track
20 sites (East Europe, USA, Asia) actively recruiting patients Target 54 MBC patients to establish BE exceeded in Dec 2014 Recruitment to continue till Jan 2015 for expanded safety data base Initial reported AEs consistent with historical nab-paclitaxel toxicity profile
Active Sites and Patient Enrollment
Active Enrolled
Enrollment 54 pts Patients
Sites
12
|
|
Estimated Timeline and Next Steps*
First patient dosed: March 31, 2014 Last patient in: January 2015
NDA filing: Q3 2015
Product launch (MBC and NSCLC): 2016
LAUNCH
2016
FDA
Approval
2016
NDA
BE Filing 2016
Study
2015
2014
*Estimates, subject to discussions with the FDA.
13
|
|
Resiniferatoxin (RTX): A Novel, Non-opiate Analgesic
Intractable
RTX Cancer
Pain
14
|
|
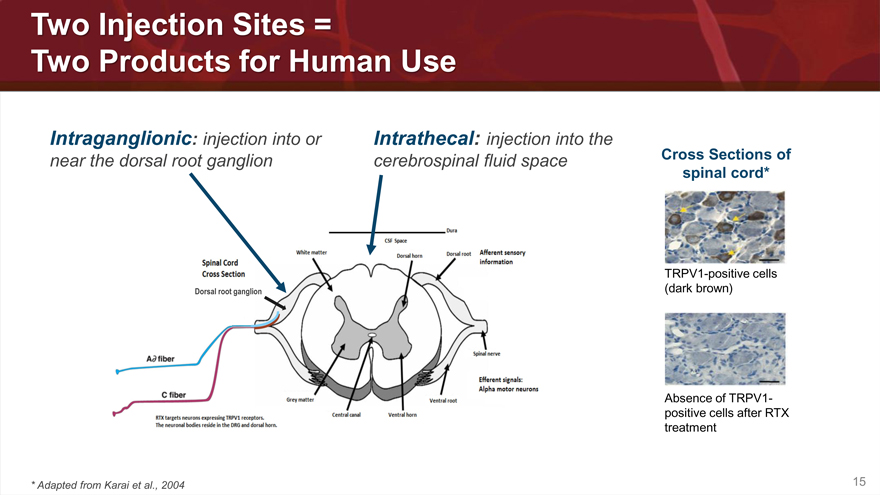
Two Injection Sites =
Two Products for Human Use
Intraganglionic: injection into or Intrathecal: injection into the
near the dorsal root ganglion cerebrospinal fluid space
Dorsal root ganglion
Cross Sections of
spinal cord*
TRPV1-positive cells
(dark brown)
Absence of TRPV1-
positive cells after RTX
treatment
15
|
|
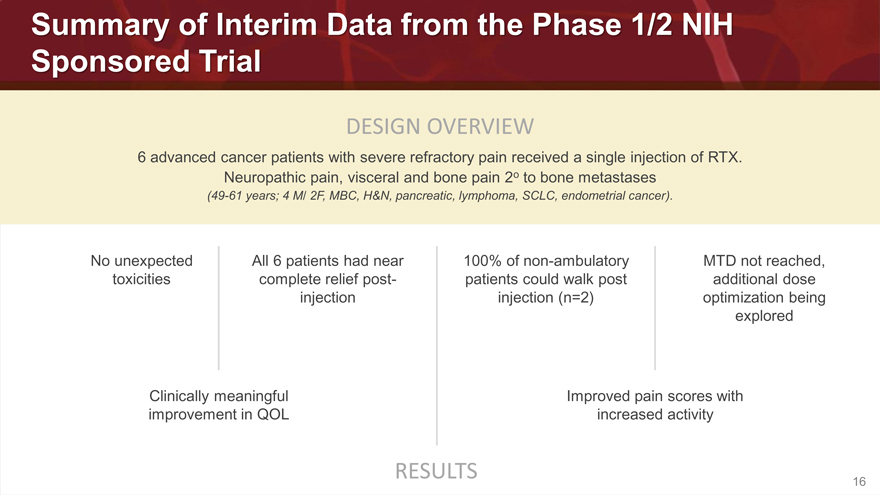
Summary of Interim Data from the Phase 1/2 NIH
Sponsored Trial
DESIGN OVERVIEW
6 advanced cancer patients with severe refractory pain received a single injection of RTX. Neuropathic pain, visceral and bone pain 2o to bone metastases
(49-61 years; 4 M/ 2F, MBC, H&N, pancreatic, lymphoma, SCLC, endometrial cancer).
No unexpected All 6 patients had near 100% of non-ambulatory MTD not reached,
toxicities complete relief post- patients could walk post additional dose
injection injection (n=2) optimization being
explored
Clinically meaningful Improved pain scores with
improvement in QOL increased activity
RESULTS
16
|
|
Next Steps for RTX Development
Complete intractable cancer pain clinical Phase 1/2
trial (intrathecal injection ) under Sorrento IND;
n=45-60 patients; optimization of dosing study
OBJECTIVES End of Phase 2 meeting with FDA (for intrathecal injection)
for
2015 and 2016 Initiate Phase 3 (intrathecal injection)
Phase 1/2 trial(s) (intraganglionic injection)
End of Phase 2 meeting with FDA (for intraganglionic injection)
~3 years for clinical development
17
|
|
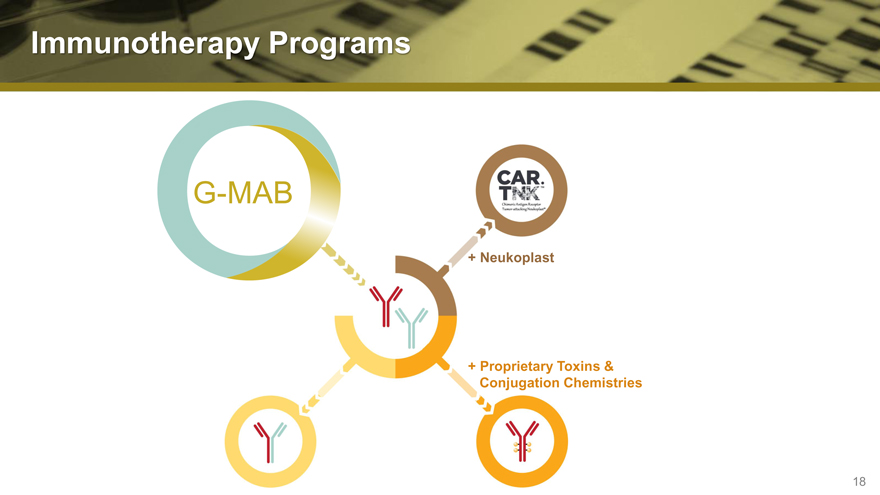
Immunotherapy Programs
G-MAB
+ Neukoplast
+ Proprietary Toxins &
Conjugation Chemistries
18
|
|
G-MAB: Library of Therapeutic Antibodies
Very high library diversity:
Proprietary technology: 2.1 x 1016 distinct antibodies
RNA amplification used for library Fully human antibodies
generation
Freedom-To-Operate High successful screening hit rate
(over 70 targets screened)
No stacking royalties Ideal for CAR-Generation
Difficult Targets:
Small Peptides
High Value Oncology Targets:
Immune modulation: PD-1, PD-L1, CD47 Antibody Drug Conjugates: VEGFR2, c-Met
Most Difficult Targets:
G Protein-Coupled Receptors (GPCRs)
Size of Target Antigen
19
|
|
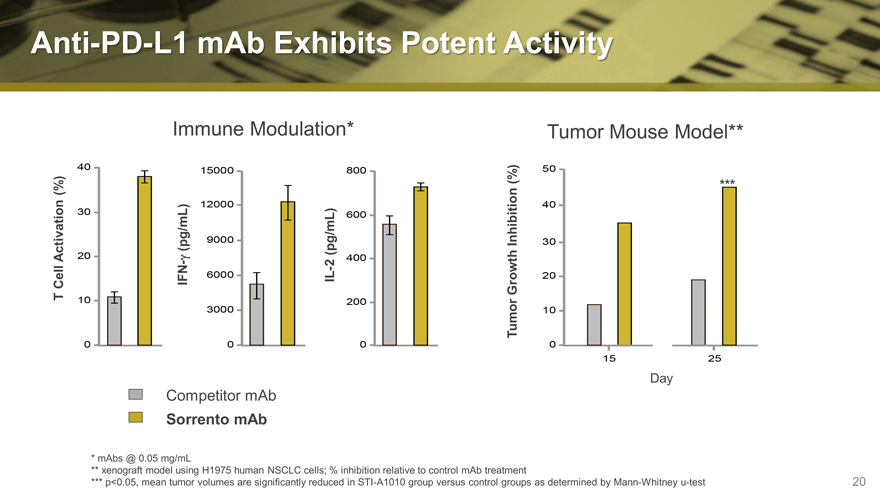
Anti-PD-L1 mAb Exhibits Potent Activity
T Cell Activation (%)
Sorrento Competitor IFN-g (pg/mL) Immune
mAb mAb
IL-2 (pg/mL) Modulation*
Tumor Growth Inhibition (%)
Tumor
Day Mouse
*** Model**
mAbs @ 0.05 mg/mL
xenograft model using H1975 human NSCLC cells; % inhibition relative to control mAb treatment
p<0.05, mean tumor volumes are significantly reduced in STI -A1010 group versus control groups as determined by Mann-Whitney u-test
20
|
|
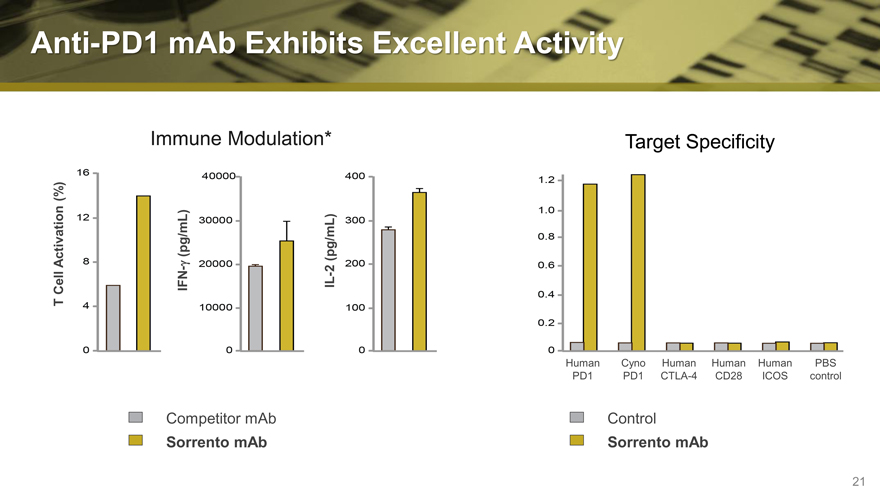
Anti-PD1 mAb Exhibits Excellent Activity
Immune Modulation* Target Specificity
(%)
Activation g (pg/mL) (pg/mL)
— 2
—
Cell IFN IL
T
Human Cyno Human Human Human PBS
PD1 PD1 CTLA-4 CD28 ICOS control
Competitor mAb Control
Sorrento mAb Sorrento mAb
21
|
|
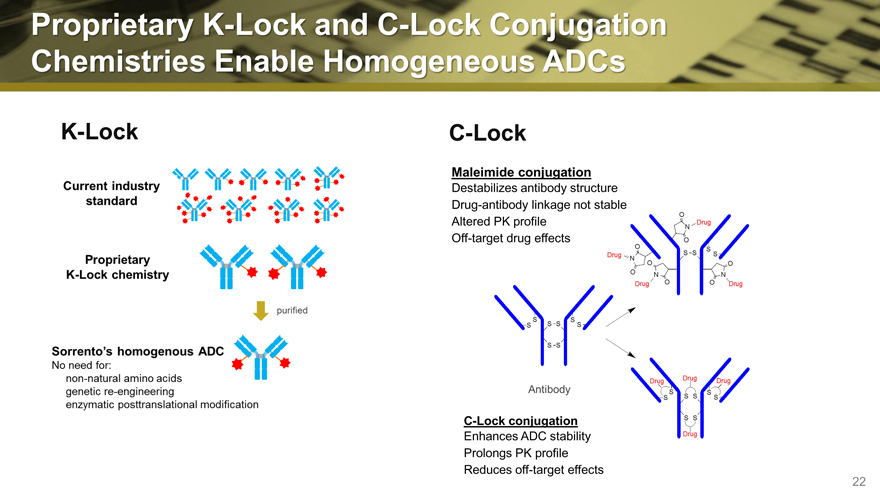
Proprietary K-Lock and C-Lock Conjugation
Chemistries Enable Homogeneous ADCs
K-Lock
C-Lock
Maleimide conjugation
Destabilizes antibody structure Drug-antibody linkage not stable Altered PK profile Off-target drug effects
C-Lock conjugation
Enhances ADC stability Prolongs PK profile Reduces off-target effects
22
|
|
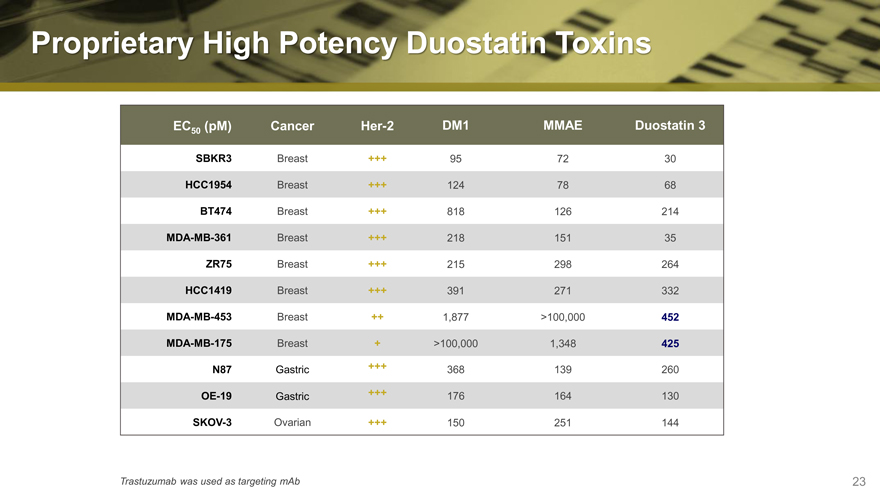
Proprietary High Potency Duostatin Toxins
EC50 (pM) Cancer Her-2 DM1 MMAE Duostatin 3
SBKR3 Breast +++ 95 72 30
HCC1954 Breast +++ 124 78 68
BT474 Breast +++ 818 126 214
MDA-MB-361 Breast +++ 218 151 35
ZR75 Breast +++ 215 298 264
HCC1419 Breast +++ 391 271 332
MDA-MB-453 Breast ++ 1,877 >100,000 452
MDA-MB-175 Breast + >100,000 1,348 425
N87 Gastric +++ 368 139 260
OE-19 Gastric +++ 176 164 130
SKOV-3 Ovarian +++ 150 251 144
Trastuzumab was used as targeting mAb
23
|
|
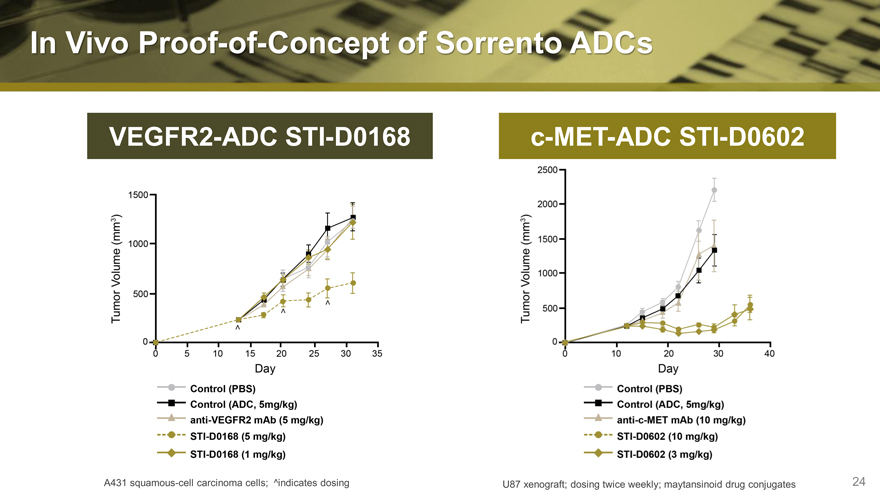
In Vivo Proof-of-Concept of Sorrento ADCs
VEGFR2-ADC STI-D0168
c-MET-ADC STI-D0602
A431 squamous-cell carcinoma cells; ^indicates dosing
U87 xenograft; dosing twice weekly; maytansinoid drug conjugates
24
|
|
“The Immunotherapy Antibody JV Company”
Independent company focused on advancing next generation immunotherapies against cancer and auto-immune diseases.
Both companies will contribute to its pipeline of clinical and preclinical assets of novel and proprietary immunotherapies, ADCs, and bispecific antibodies.
Joint venture will draw from NantWorks’ proteomic and genomic capabilities and Sorrento’s industry-leading, highly diverse G-MAB library.
25
|
|
AN EXCLUSIVE JOINT PARTNERSHIP
CAR.TNK is a trademark owned by Sorrento Therapeutics, Inc. Neukoplast is a trademark owned by Conkwest, Inc.
26
|
|
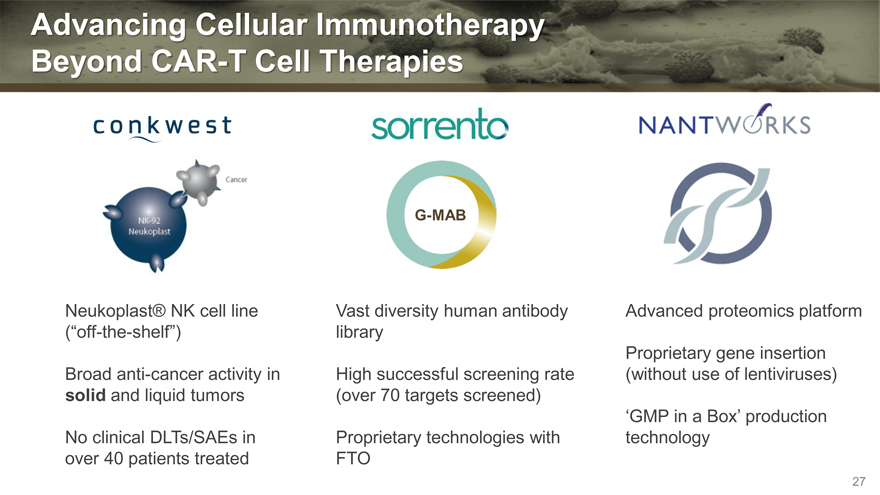
Advancing Cellular Immunotherapy
Beyond CAR-T Cell Therapies
Neukoplast® NK cell line Vast diversity human antibody Advanced proteomics platform
(“off-the-shelf”) library
Proprietary gene insertion
Broad anti-cancer activity in High successful screening rate (without use of lentiviruses)
solid and liquid tumors (over 70 targets screened)
‘GMP in a Box’ production
No clinical DLTs/SAEs in Proprietary technologies with technology
over 40 patients treated FTO
27
|
|
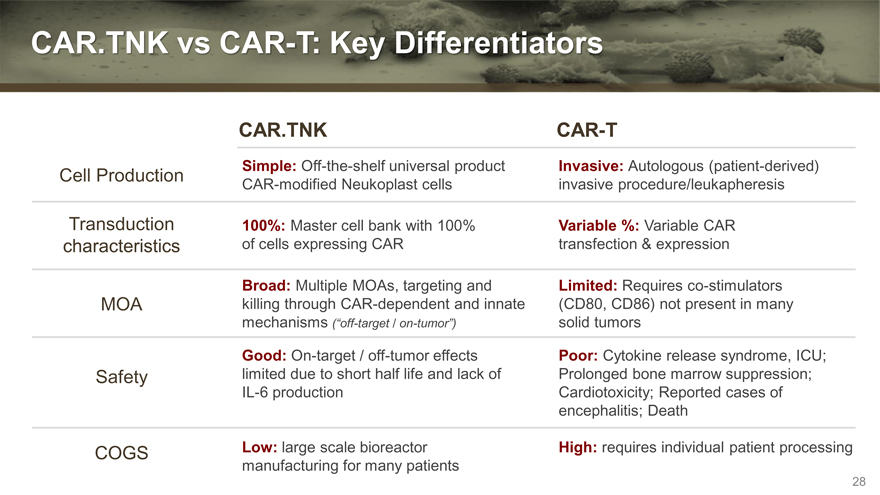
CAR.TNK vs CAR-T: Key Differentiators
CAR.TNK CAR-T
Simple: Off-the-shelf universal product Invasive: Autologous (patient-derived)
Cell Production CAR-modified Neukoplast cells invasive procedure/leukapheresis
Transduction 100%: Master cell bank with 100% Variable %: Variable CAR
characteristics of cells expressing CAR transfection & expression
Broad: Multiple MOAs, targeting and Limited: Requires co-stimulators
MOA killing through CAR-dependent and innate (CD80, CD86) not present in many
mechanisms (“off-target / on-tumor”) solid tumors
Good: On-target / off-tumor effects Poor: Cytokine release syndrome, ICU;
Safety limited due to short half life and lack of Prolonged bone marrow suppression;
IL-6 production Cardiotoxicity; Reported cases of
encephalitis; Death
COGS Low: large scale bioreactor High: requires individual patient processing
manufacturing for many patients
28
|
|
Unmodified Neukoplast Clinically Validated In Several
Phase 1 Studies
More than 40 patients treated
Advanced metastatic disease refractory to chemo, biologics, cytokines, radiation, and surgery
Many patients received multiple dosing regimens (up to 6 months)
Promising activity against different cancer types, including acute myelogenous leukemia (AML), lymphoma (NHL, HL), melanoma, renal cell cancer (RCC), and lung cancers (SCLC, NSCLC)
No DLTs; only 1 “grade 4 SAE” (hypoglycemia likely related to tumor lysis)
29
|
|
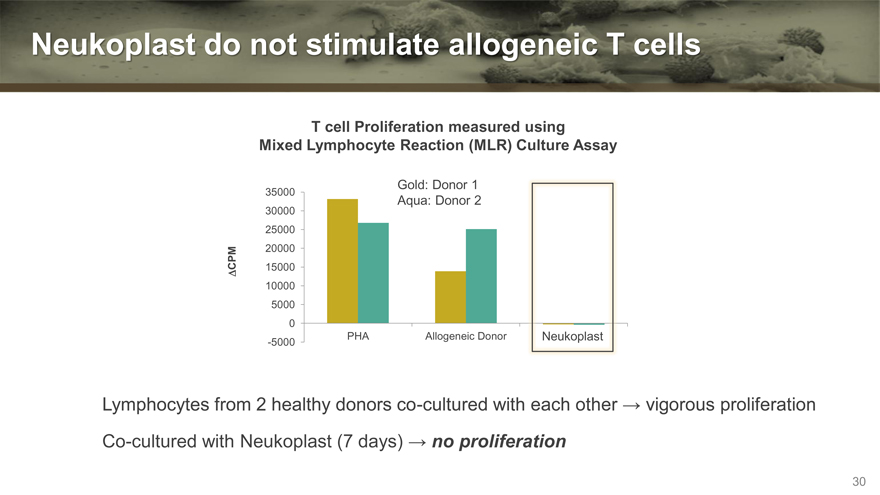
Neukoplast do not stimulate allogeneic T cells
T cell Proliferation measured using
Mixed Lymphocyte Reaction (MLR) Culture Assay
Gold: Donor 1
35000 Aqua: Donor 2
30000
25000
20000
CPM 15000
D
10000
5000
0
-5000 PHA Allogeneic Donor Neukoplast
Lymphocytes from 2 healthy donors co-cultured with each other vigorous proliferation
Co-cultured with Neukoplast (7 days) no proliferation
30
|
|
CAR.TNK: CAR-modified Neukoplast
Clonal cell lines expressing one or more CARs to establish a range of distinct products
Multiple killing mechanisms—CAR-targeted as well as broad intrinsic anti-cancer activity of Neukoplast (“off-target / on-tumor”)
Engages the adaptive immune system through cytokine secretion and immune cell recruitment
Titratable: repeat dosing option; controllable dose exposure to manage safety risk
31
|
|
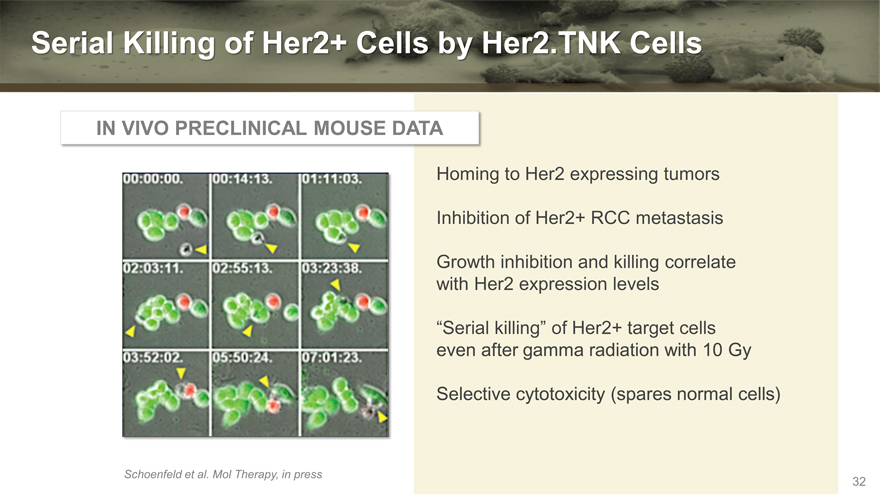
Serial Killing of Her2+ Cells by Her2.TNK Cells
IN VIVO PRECLINICAL MOUSE DATA
Homing to Her2 expressing tumors
Inhibition of Her2+ RCC metastasis
Growth inhibition and killing correlate with Her2 expression levels
“Serial killing” of Her2+ target cells even after gamma radiation with 10 Gy
Selective cytotoxicity (spares normal cells)
Schoenfeld et al. Mol Therapy, in press
32
|
|
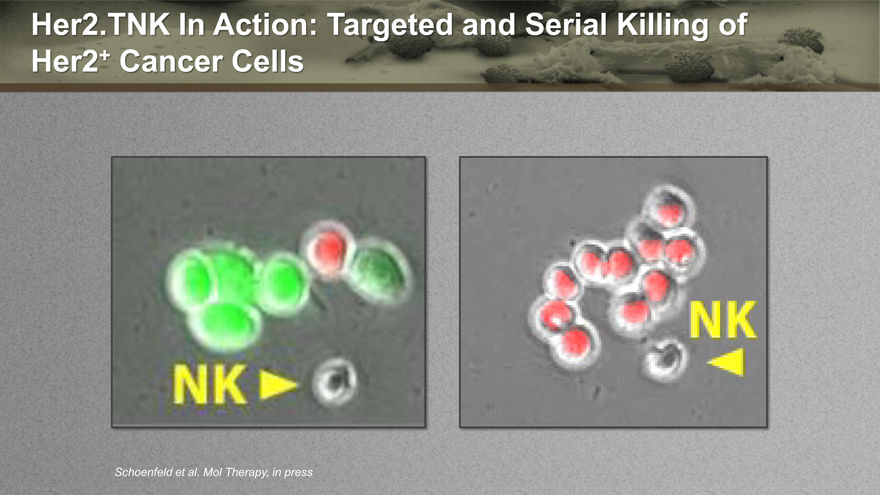
Her2.TNK In Action: Targeted and Serial Killing of
Her2+ Cancer Cells
Schoenfeld et al. Mol Therapy, in press
33
|
|
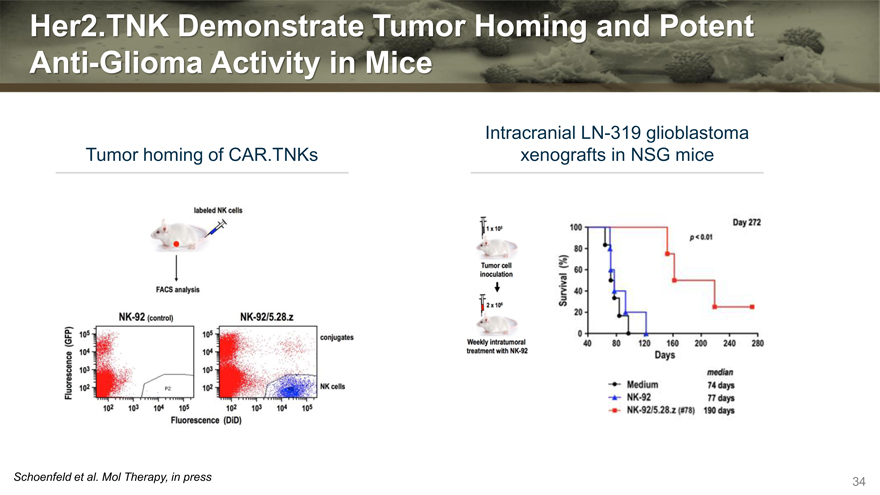
Her2.TNK Demonstrate Tumor Homing and Potent Anti-Glioma Activity in Mice
Tumor homing of CAR.TNKs
Intracranial LN-319 glioblastoma
xenografts in NSG mice
Schoenfeld et al. Mol Therapy, in press
34
|
|
Prospective CAR.TNKs for Development
(Initial List)
Target Potential Indication(s)
EGFRviii.TNK Glioma
EphA3.TNK Glioma, AML
L1CAM.TNK Gastric, pancreatic, NSCLC
CSPG4.TNK H&N, breast, mesothelioma
BCMA.TNK Myeloma
ROR1.TNK CLL, ALL, MCL, breast, lung, pancreas
PSMA or
PSCA.TNK Prostate
PDL1.TNK Myeloma, RCC, NSCLC, TNBC
CS1.TNK Myeloma
CD123.TNK AML
CD19.TNK CLL, ALL
CD22.TNK CLL, ALL
CAR targets jointly selected by the Steering Committee
Lead company will be responsible for all pre-clinical and clinical development, regulatory filings, and commercialization
Profit sharing on all CAR.TNKs revenues proportional to contribution
EGFRviii.TNK, EphA3.TNK, L1CAM.TNK, CSPG4.TNK, BCMA.TNK, ROR1.TNK, PSMA.TNK, PCMA.TNK, PDL1.TNK, CS1.TNK, CD123.TNK, CD19.TNK, CD22.TNK are trademarks owned by Sorrento Therapeutics, Inc.
35
|
|
Next Steps for CAR.TNK Development
H1 2015 Generation of CARs
H2 2015 Generation and evaluation of stable CAR.TNK cell lines
2016 IND-enabling studies, IND submission, and initiation of Phase 1 studies
36
|
|
A Comprehensive Oncology Company
Deep and Complementary Pipeline Creates Significant Opportunities-
Novel breakthrough combination therapeutic regimens and modalities to attack cancer Significant reduction in clinical development costs and timeline Significant commercial edge in future drug pricing
Small Biologics Cell Supportive
Molecules Therapy Care
Cytotoxics Immunotherapy RTX
CYNVILOQ™ PD1, PD-L1, CTLA-4
Bispecific Abs Intractable
Cancer Pain
Targeted Therapy Targeted Therapy Adoptive Cellular
MYC inhibitor Anti-VEGR2 ADC Immunotherapy
TRAIL modulator Anti-CMET ADC Chimeric Antigen Receptor
Bispecific ADC Tumor-attacking Neukoplast®
(Partnership with Conkwest)
CYNVILOQ, CAR.TNK, CAR.TNK (Chimeric Antigen Receptor Tumor-attacking Neukoplast) are trademarks owned by Sorrento Therapeutics, Inc. Neukoplast is a trademark owned by Conkwest, Inc.
37
|
|
Next-Generation
Cancer Therapeutics
CONTACT:
George Uy Henry Ji, Ph.D.
Executive Vice President and CCO President and CEO
guy@sorrentotherapeutics.com hji@sorrentotherapeutics.com
(661) 607-4057 (858) 668-6923