Attached files
| file | filename |
|---|---|
| 8-K - FORM 8-K - INFINITY PHARMACEUTICALS, INC. | d850351d8k.htm |
| EX-99.2 - EXHIBIT 99.2 - INFINITY PHARMACEUTICALS, INC. | d850351dex992.htm |
| Exhibit 99.1
|
Building a Fully Integrated
Biopharmaceutical Company
January 12, 2015
|
|
Forward-Looking Statements
This presentation contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Such forward-looking statements include those regarding the Company’s expectations about: its plans to complete patient enrollment in DYNAMO in the first half of 2015 and in DUO in the second half of 2015; its plans to report topline data from DYNAMO in the second half of 2015; its plans to initiate additional studies of duvelisib in 2015; its expectations regarding further development of duvelisib or IPI-443 in RA; its ability to execute on its strategic plans; the potential complementary effects of inhibiting PI3K-delta and PI3K-gamma; the therapeutic potential of PI3K inhibition and duvelisib; and its fiscal year 2014 year-end cash balance and 2015 financial guidance. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from the company’s current expectations. For example, there can be no guarantee that Infinity will report data in the time frames it has estimated, that any product candidate Infinity is developing will successfully complete necessary preclinical and clinical development phases, or that development of any of Infinity’s product candidates will continue. Further, there can be no guarantee that Infinity’s strategic collaboration with AbbVie will continue or that any positive developments in Infinity’s product portfolio will result in stock price appreciation. Management’s expectations and, therefore, any forward-looking statements in this press release could also be affected by risks and uncertainties relating to a number of other factors, including the following: Infinity’s results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; a failure of Infinity and/or AbbVie to fully perform under the strategic collaboration and/or an early termination of the collaboration and license agreement; the content and timing of decisions made by the U.S. FDA and other regulatory authorities, investigational review boards at clinical trial sites and publication review bodies; Infinity’s ability to obtain and maintain requisite regulatory approvals and to enroll patients in its clinical trials; unplanned cash requirements and expenditures; development of agents by Infinity’s competitors for diseases in which Infinity is currently developing or intends to develop its product candidates; and Infinity’s ability to obtain, maintain and enforce patent and other intellectual property protection for any product candidates it is developing. These and other risks which may impact management’s expectations are described in greater detail under the caption “Risk Factors” included in Infinity’s quarterly report on Form 10-Q filed with the Securities and Exchange Commission (SEC) on November 10, 2014, and other filings filed by Infinity with the SEC. Any forward-looking statements contained in this press release speak only as of the date hereof, and Infinity expressly disclaims any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise.
Infinity’s website is http://www.infi.com. Infinity regularly uses its website to post information regarding its business, product development programs and governance. Infinity encourages investors to use www.infi.com, particularly the information in the section entitled “Investors/Media,” as a source of information about Infinity. References to www.infi.com in this presentation are not intended to, nor shall they be deemed to, incorporate information on www.infi.com into this presentation by reference.
2
|
|
|
|
|
|
|
|
|
|
|
|
|
|
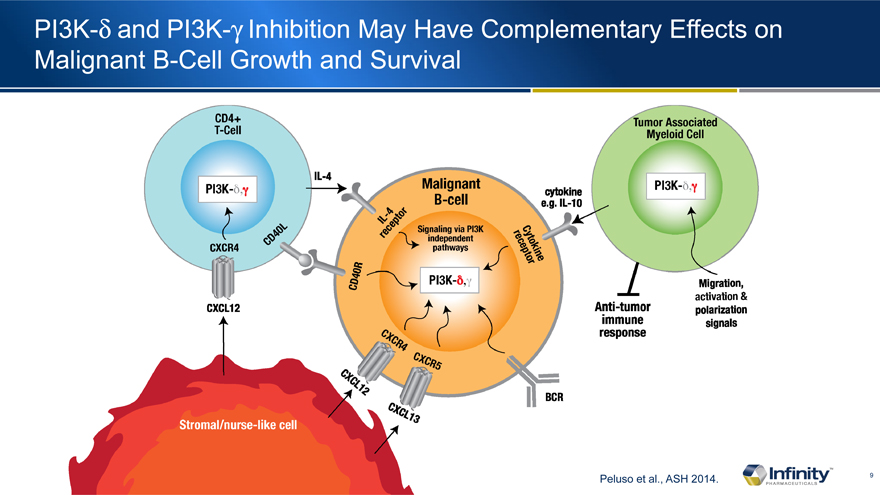
PI3K-d and PI3K-g Inhibition May Have Complementary Effects on Malignant B-Cell Growth and Survival
Peluso et al., ASH 2014.
9
|
|
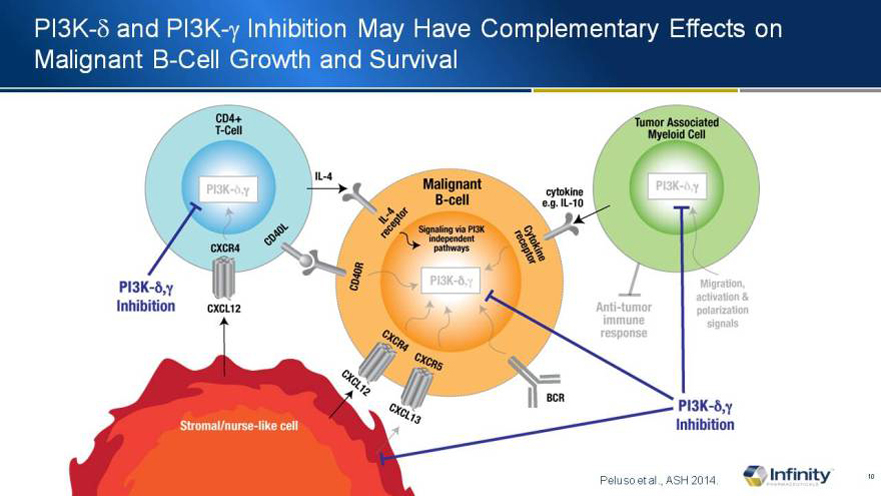
PI3K-d and PI3K-g Inhibition May Have Complementary Effects on Malignant B-Cell Growth and Survival
Peluso et al., ASH 2014.
10
|
|
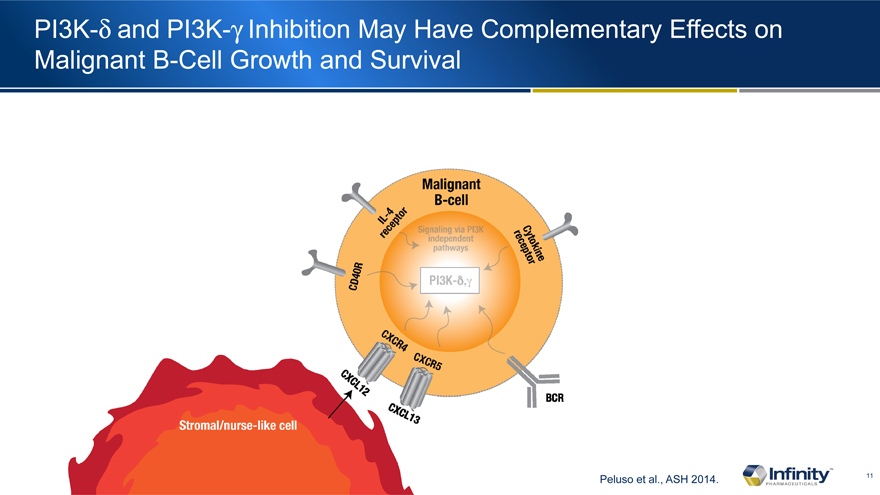
PI3K-d and PI3K-g Inhibition May Have Complementary Effects on Malignant B-Cell Growth and Survival
Peluso et al., ASH 2014.
11
|
|
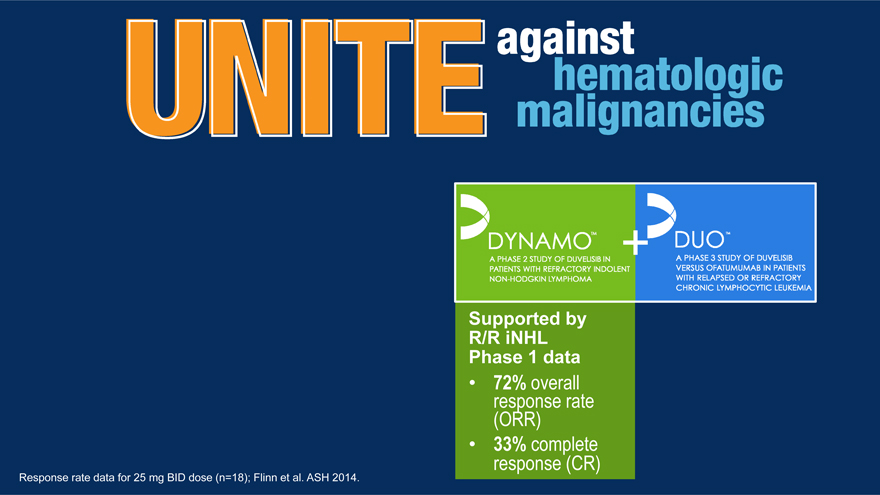
Supported by R/R iNHL Phase 1 data
72% response overall rate (ORR) response 33% complete (CR)
Response rate data for 25 mg BID dose (n=18); Flinn et al. ASH 2014.
|
|
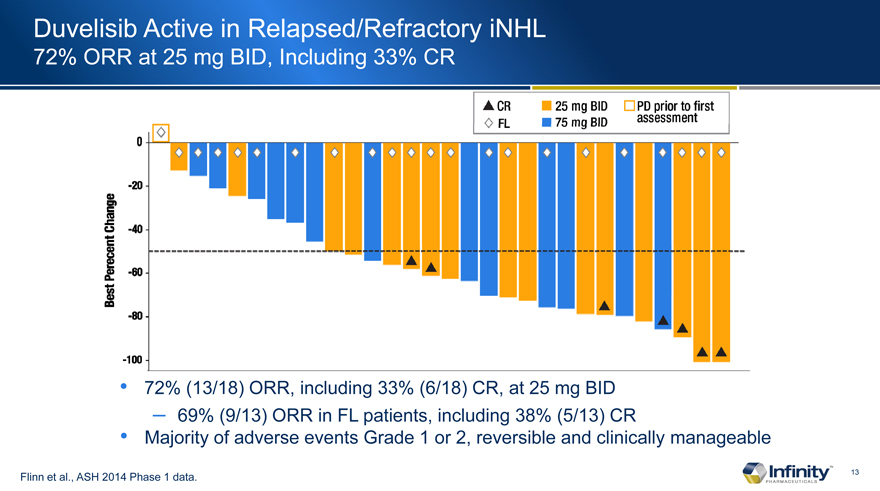
Duvelisib Active in Relapsed/Refractory iNHL
72% ORR at 25 mg BID, Including 33% CR
72% (13/18) ORR, including 33% (6/18) CR, at 25 mg BID
– 69% (9/13) ORR in FL patients, including 38% (5/13) CR
Majority of adverse events Grade 1 or 2, reversible and clinically manageable
Flinn et al., ASH 2014 Phase 1 data.
13
|
|
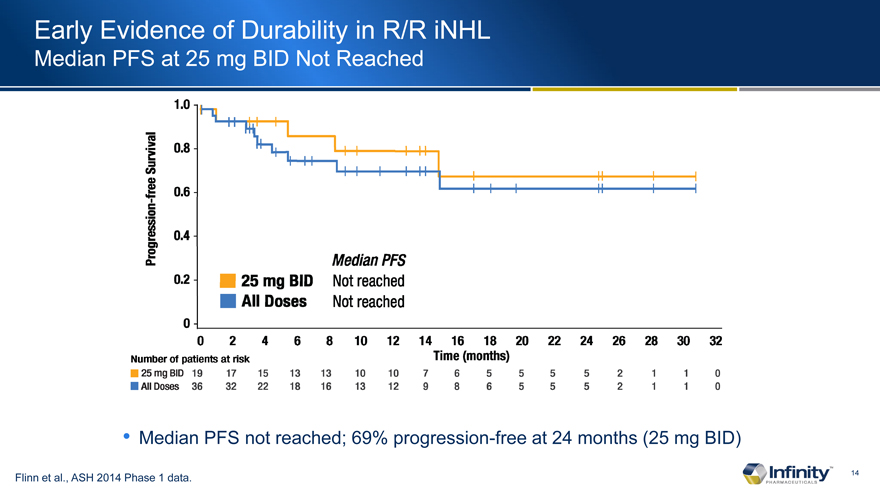
Early Evidence of Durability in R/R iNHL
Median PFS at 25 mg BID Not Reached
Median PFS not reached; 69% progression-free at 24 months (25 mg BID)
Flinn et al., ASH 2014 Phase 1 data.
14
|
|
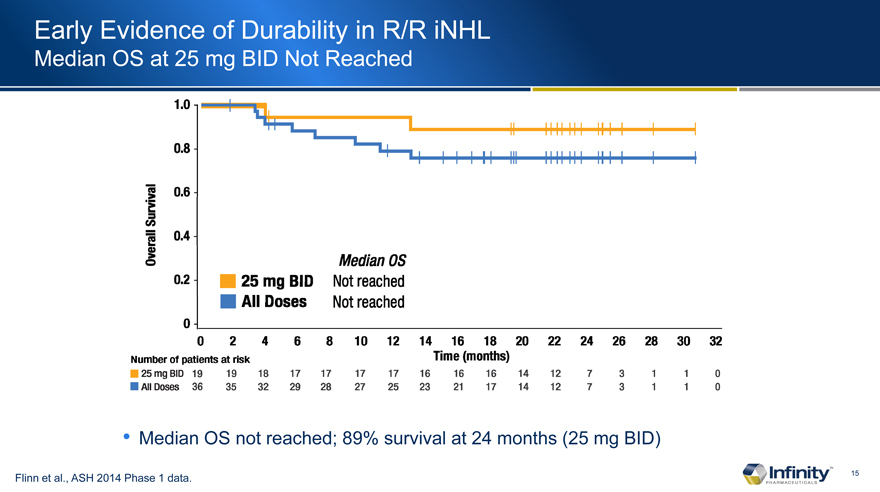
Early Evidence of Durability in R/R iNHL
Median OS at 25 mg BID Not Reached
Median OS not reached; 89% survival at 24 months (25 mg BID)
Flinn et al., ASH 2014 Phase 1 data.
15
|
|
DYNAMO™: Phase 2 Monotherapy Study in Refractory iNHL
Completion of Enrollment Expected in 1H’15
Open-label, single-arm monotherapy study under way
Primary endpoint: Objective response rate
Regulatory support for potential accelerated approval
*Includes follicular lymphoma (FL), marginal zone lymphoma (MZL), small lymphocytic lymphoma (SLL) Duvelisib dosed at 25 mg BID; Clinicaltrials.gov NCT01882803.
16
|
|
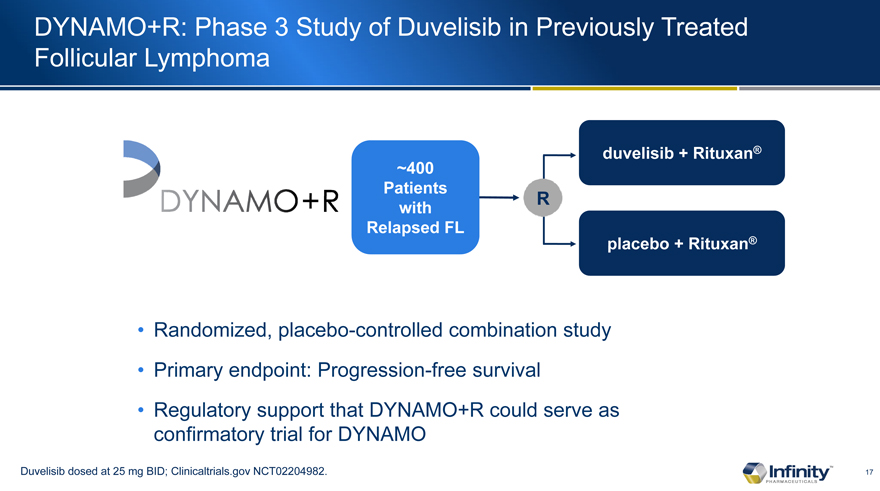
DYNAMO+R: Phase 3 Study of Duvelisib in Previously Treated Follicular Lymphoma
~400 Patients with Relapsed FL
Randomized, placebo-controlled combination study
Primary endpoint: Progression-free survival
Regulatory support that DYNAMO+R could serve as confirmatory trial for DYNAMO
Duvelisib dosed at 25 mg BID; Clinicaltrials.gov NCT02204982.
duvelisib + Rituxan®
placebo + Rituxan®
17
|
|
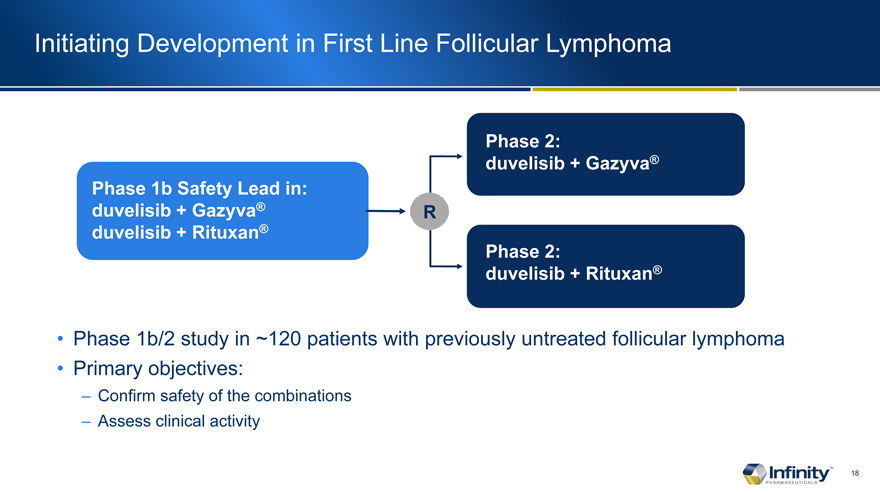
Initiating Development in First Line Follicular Lymphoma
Phase 1b Safety Lead in: duvelisib + Gazyva® duvelisib + Rituxan®
Phase 1b/2 study in ~120 patients with previously untreated follicular lymphoma
Primary objectives:
– Confirm safety of the combinations
– Assess clinical activity
Phase 2: duvelisib + Gazyva®
Phase 2: duvelisib + Rituxan®
18
|
|
Supported by R/R CLL Phase 1 data
response 83% nodal
57% (IWCLL), ORR including 1 CR
Response rate data for 25 mg BID dose (n=30); O’Brien et al. ASH 2014.
|
|
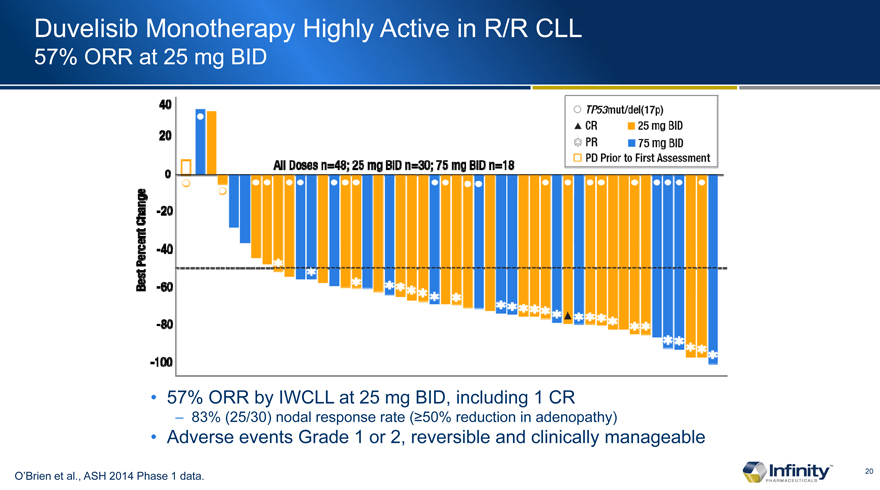
Duvelisib Monotherapy Highly Active in R/R CLL
57% ORR at 25 mg BID
57% ORR by IWCLL at 25 mg BID, including 1 CR
– 83% (25/30) nodal response rate (³50% reduction in adenopathy)
Adverse events Grade 1 or 2, reversible and clinically manageable
O’Brien et al., ASH 2014 Phase 1 data.
20
|
|
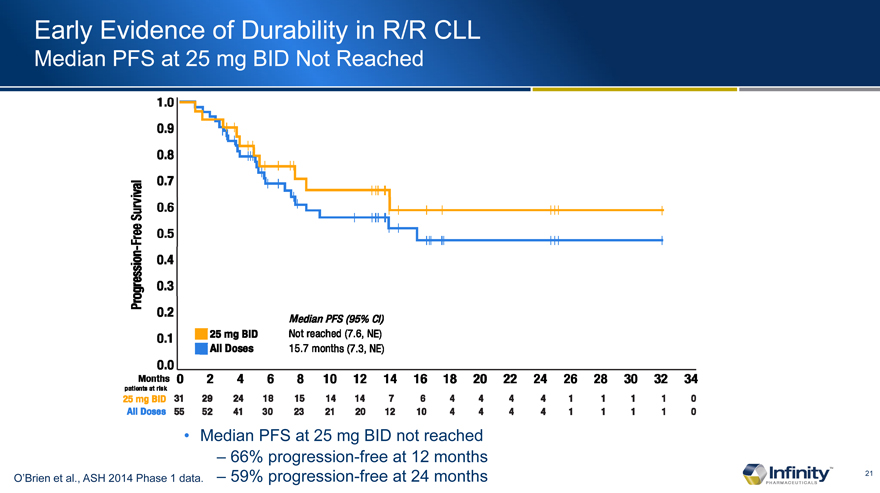
Early Evidence of Durability in R/R CLL
Median PFS at 25 mg BID Not Reached
Median PFS at 25 mg BID not reached
– 66% progression-free at 12 months
O’Brien et al., ASH 2014 Phase 1 data. – 59% progression-free at 24 months
21
|
|
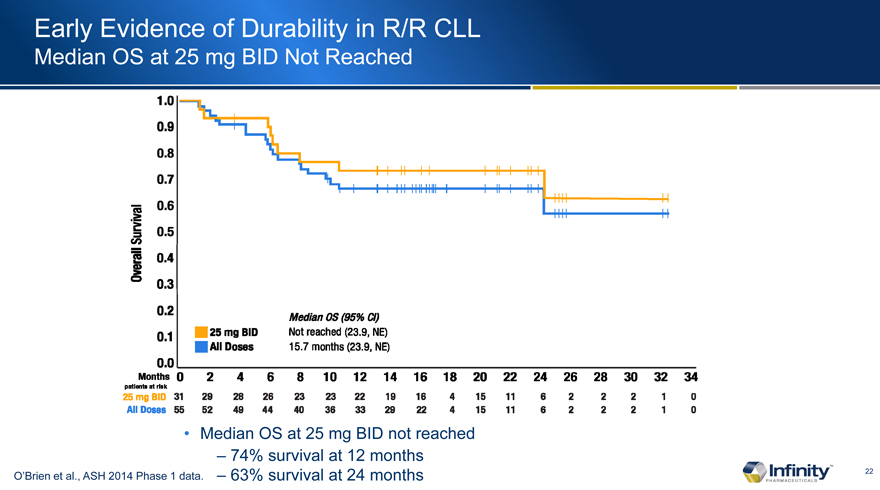
Early Evidence of Durability in R/R CLL
Median OS at 25 mg BID Not Reached
Median OS at 25 mg BID not reached
– 74% survival at 12 months
O’Brien et al., ASH 2014 Phase 1 data. – 63% survival at 24 months
22
|
|
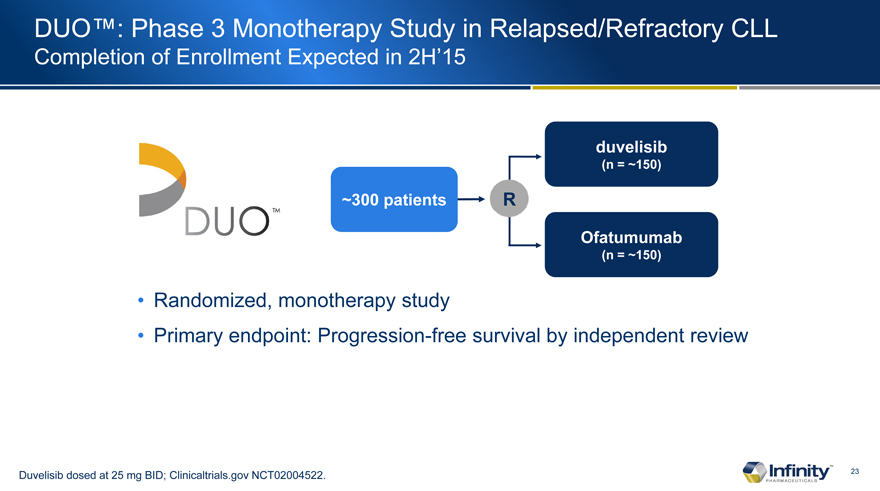
DUO™: Phase 3 Monotherapy Study in Relapsed/Refractory CLL
Completion of Enrollment Expected in 2H’15
~300 patients
duvelisib
(n = ~150)
Ofatumumab
(n = ~150)
Randomized, monotherapy study
Primary endpoint: Progression-free survival by independent review
Duvelisib dosed at 25 mg BID; Clinicaltrials.gov NCT02004522.
23
|
|
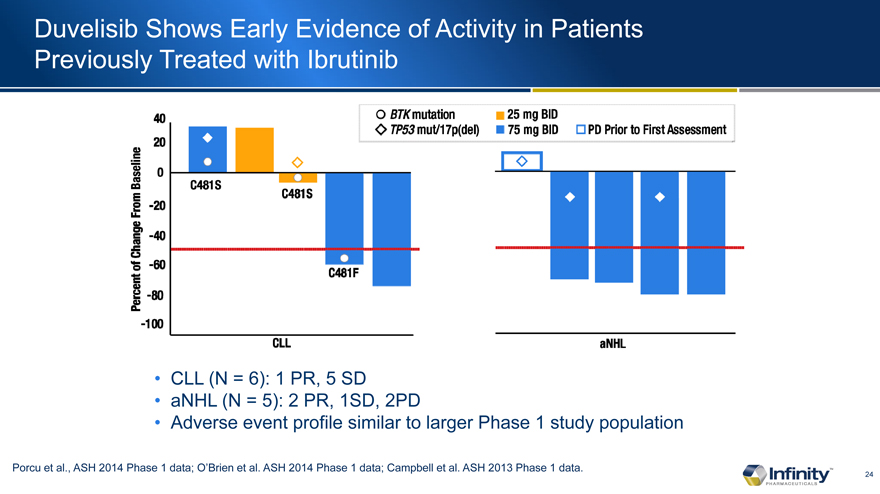
Duvelisib Shows Early Evidence of Activity in Patients Previously Treated with Ibrutinib
CLL (N = 6): 1 PR, 5 SD
aNHL (N = 5): 2 PR, 1SD, 2PD
Adverse event profile similar to larger Phase 1 study population
Porcu et al., ASH 2014 Phase 1 data; O’Brien et al. ASH 2014 Phase 1 data; Campbell et al. ASH 2013 Phase 1 data.
24
|
|
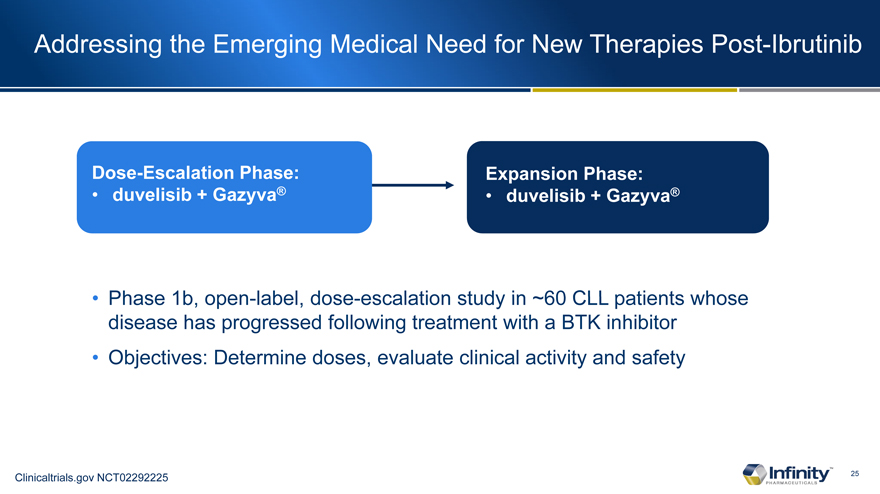
Addressing the Emerging Medical Need for New Therapies Post-Ibrutinib
Dose-Escalation Phase: • duvelisib + Gazyva®
Expansion Phase: • duvelisib + Gazyva®
Phase 1b, open-label, dose-escalation study in ~60 CLL patients whose disease has progressed following treatment with a BTK inhibitor
Objectives: Determine doses, evaluate clinical activity and safety
Clinicaltrials.gov NCT02292225
25
|
|
|
|
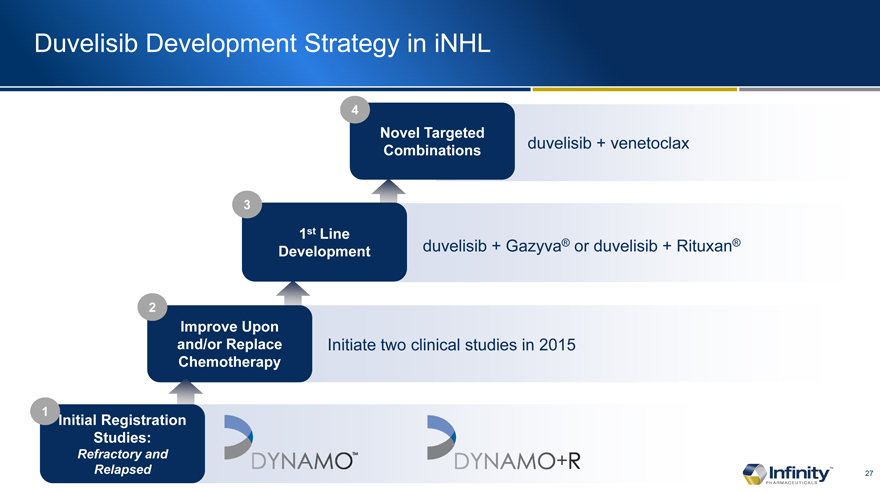
Duvelisib Development Strategy in iNHL
duvelisib + Gazyva® or duvelisib + Rituxan®
duvelisib + venetoclax
Initiate two clinical studies in 2015
27
|
|
Novel Ta Combinations
1st Line Development
Improve Upon and/or Replace Chemotherapy
Initial Registration Studies:
Refractory and Relapsed
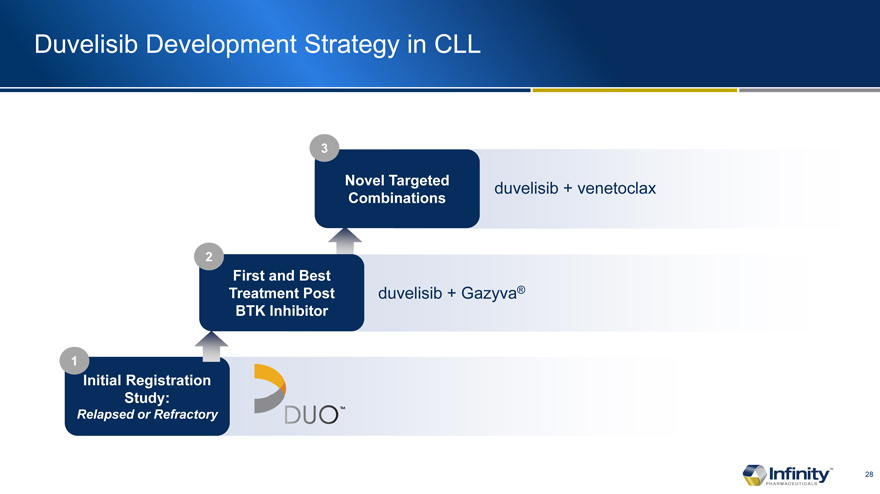
Duvelisib Development Strategy in CLL
Novel Targeted Combinations
First and Treatment BTK
Initial Registration Study:
Relapsed or Refractory
duvelisib + venetoclax
28
|
|
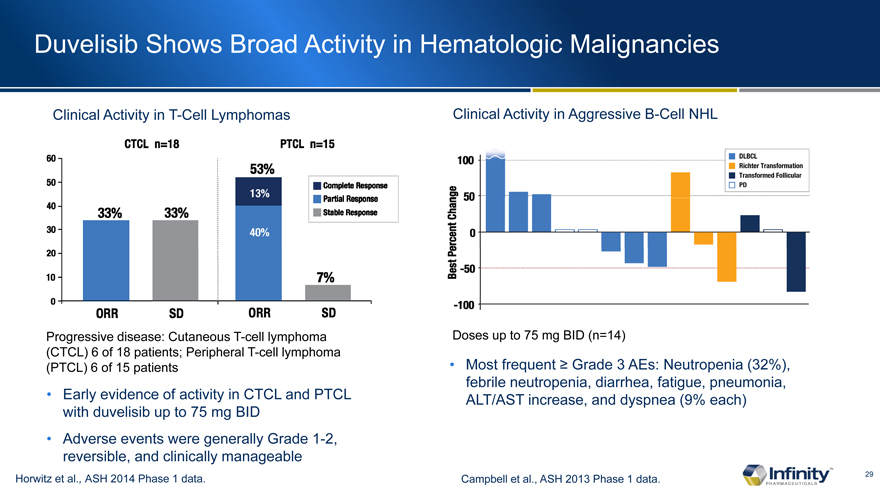
Duvelisib Shows Broad Activity in Hematologic Malignancies
Clinical Activity in T-Cell Lymphomas
Progressive disease: Cutaneous T-cell lymphoma (CTCL) 6 of 18 patients; Peripheral T-cell lymphoma (PTCL) 6 of 15 patients
Early evidence of activity in CTCL and PTCL with duvelisib up to 75 mg BID
Adverse events were generally Grade 1-2, reversible, and clinically manageable
Horwitz et al., ASH 2014 Phase 1 data.
Clinical Activity in Aggressive B-Cell NHL
Doses up to 75 mg BID (n=14)
Most frequent Grade 3 AEs: Neutropenia (32%), febrile neutropenia, diarrhea, fatigue, pneumonia, ALT/AST increase, and dyspnea (9% each)
Campbell et al., ASH 2013 Phase 1 data.
29
|
|
|
|
Strategic Partnership with AbbVie for Duvelisib in Oncology
Partnership Terms:
Financial
Development
Commercial
$275M up-front payment
$530M in potential development and commercial milestones
Up to $405 million for the achievement of milestones through first commercial sale
Infinity will fund the trials it conducts, up to $667 million, after which costs will be shared equally
Infinity and AbbVie will equally share funding for trials conducted by AbbVie
Co-promotion and 50/50 profit share in U.S.
Tiered royalty payments ex-U.S., ranging from 23.5% to 30.5% of net sales
31
|
|
2015 Financial Guidance (as of January 12, 2015)
Cash and investments at 12/31/14 (unaudited):
Revenue:
Net loss:
Year-end cash and investments:
2014 year-end cash balance and 2015 financial guidance provided as of January 12, 2015.
2015 financial revenue guidance assumes receipt of a $130 million milestone payment from AbbVie Inc. associated with the anticipated completion of enrollment in DYNAMO or DUO; financial guidance is based on current operating plan and exclusive of any business development activities.
~$330M
$105M-$125M $190M-$210M $145M-$165M
32
|
|
Anticipated 2015 Milestones
Novel Duvelisib Combinations
Initiate first clinical study of duvelisib in combination with venetoclax*
Duvelisib in iNHL
Complete enrollment in DYNAMOTM in 1H’15 Report topline data from DYNAMO in 2H’15 Initiate 2 additional clinical studies
Duvelisib in CLL
Complete enrollment in DUOTM in 2H’15
*This study will be initiated by AbbVie.
33
|
|
Building a Fully Integrated Biopharmaceutical Company
www.infi.com