Attached files
| file | filename |
|---|---|
| EX-21.1 - EXHIBIT 21.1 - Tonix Pharmaceuticals Holding Corp. | v344643_ex21-1.htm |
| EX-23.01 - EXHIBIT 23.01 - Tonix Pharmaceuticals Holding Corp. | v344643_ex23-1.htm |
As filed with the Securities and Exchange Commission on May 10, 2013
Registration No. 333-
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
WASHINGTON D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
TONIX PHARMACEUTICALS HOLDING CORP.
(Name of registrant in its charter)
| Nevada | 2834 | 26-1434750 | ||||
| (State or other Jurisdiction | (Primary Standard Industrial | (I.R.S. Employer | ||||
| of Incorporation or | Classification Code | Identification No.) | ||||
| Organization) | Number) |
509 Madison Avenue, Suite 306
New York, New York 10022
(212) 980-9155
(Address and telephone number of principal executive offices and principal place of business)
Seth Lederman, Chief Executive Officer
Tonix Pharmaceuticals Holding Corp.
509 Madison Avenue, Suite 306
New York, New York 10022
(212) 980-9155
(Name, address and telephone number of agent for service)
Copies to:
| Marc J. Ross, Esq. | John Hogoboom, Esq. |
| James M. Turner, Esq. | Lowenstein Sandler LLP |
| Sichenzia Ross Friedman Ference LLP | 1251 Avenue of the Americas |
| 61 Broadway, 32nd Flr. | New York, New York 10020 |
| New York, New York 10006 | (212) 262-6700 (phone) |
| (212) 930-9700 | (973) 597-2500 (facsimile) |
| (212) 930-9725 (fax) |
APPROXIMATE DATE OF PROPOSED SALE TO THE PUBLIC: As soon as practicable after the effective date of this registration statement.
If any securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, other than securities offered only in connection with dividend or interest reinvestment plans, check the following box: o
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a small reporting company. See definitions of “large accelerated filer,” “accelerated filed,” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer o | Accelerated filer o |
| Non-accelerated filer o | Smaller reporting company x |
| (Do not check if a smaller reporting company) |
CALCULATION OF REGISTRATION FEE
| Title of Each Class Of Securities To Be Registered | Proposed Maximum Aggregate Offering Price (1) (2) | Amount Of Registration Fee | ||||||
| Units, each unit consisting of one share of Common Stock, par value $0.001 per share, and [ * ] of a warrant to purchase one share of Common Stock | $ | 12,000,000 | $ | 1,636.80 | ||||
| Common Stock, par value $0.001 per share, included in units (3) | $ | - | - | |||||
| Warrants to purchase Common Stock, included in units (3) | $ | - | - | |||||
| Common Stock issuable upon exercise of Warrants included in units | $ | - | - | |||||
| Underwriter Warrants | $ | 480,000 | $ | 65.47 | ||||
| Common Stock issuable upon exercise of Underwriter Warrants | - | - | ||||||
| Total | $ | 12,480,000 | $ | 1,702.27 | ||||
| (1) | Estimated solely for the purpose of calculating the registration fee pursuant to Rule 457(o) under the Securities Act of 1933, as amended (the “Securities Act of 1933”). | ||
| (2) | Pursuant to Rule 416, this registration statement shall be deemed to cover additional securities that may be offered or issued to prevent dilution resulting from stock splits, stock dividends or similar transactions. | ||
| (3) | No fee is required pursuant to Rule 457(g) under the Securities Act of 1933. |
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the registration statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
The information in this prospectus is not complete and may be changed. We may not sell these securities under this prospectus until the registration statement of which it is a part and filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any state where the offer or sale is not permitted.
PRELIMINARY PROSPECTUS
SUBJECT TO COMPLETION, DATED MAY 10, 2013

[ * ] Units
Each Unit Consisting of One Share of Common Stock
and
[ * ] of a Series A Warrant, Each to Purchase One Share of Common Stock
We are offering [ * ] units, each of which consists of one share of our common stock, par value $0.001 per share, and [ * ] of a Series A Warrant, each to purchase one share of our common stock at an exercise price of [ * ] per share. The Series A Warrants will be immediately exercisable and will expire on the [ * ] anniversary of the issuance date. No units will be issued, however, and purchasers will receive only shares of common stock and Series A Warrants. The common stock and the Series A Warrants may be transferred separately immediately upon issuance.
We have applied to list our common stock on The NASDAQ Capital Market under the symbol “TNXP”. Our common stock is currently quoted on the OTCQB under the same symbol. On May 9, 2013, the last reported sale price of our common stock was $10.00 per share. We do not intend to list the Series A Warrants on any securities exchange or other trading market and we do not expect that a public trading market will develop for any of the warrants.
Investing in our common stock involves a high degree of risk. Before making any investment in our common stock, you should read and carefully consider the risks described in this prospectus under “Risk Factors ” beginning on page 9 of this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
| Per Unit | Total | |||||||
| Public offering price | $ | $ | ||||||
| Underwriting discounts and commissions | $ | $ | ||||||
| Proceeds to us, before expenses | $ | $ | ||||||
In addition to the discounts and commissions listed above, we have agreed to issue to the underwriters or their designees underwriter warrants to purchase shares of common stock equal to 4% of the total number of shares included in the units. The registration statement of which this prospectus is a part also covers the underwriter warrants and the shares of common stock issuable upon the exercise thereof. We also have agreed to reimburse the underwriters for certain of their out-of-pocket expenses. See “Underwriting” for a description of these arrangements.
The underwriters expect to deliver the units against payment on or about [ * ], 2013.
Roth Capital Partners
National Securities Corporation
This prospectus is dated , 2013
TABLE OF CONTENTS
| Page | |
| Special Note Regarding Forward-Looking Statements | 1 |
| Prospectus Summary | 2 |
| Risk Factors | 9 |
| Use of Proceeds | 33 |
| Market For Common Stock and Related Shareholder Matters | 34 |
| Dilution | 35 |
| Management’s Discussion and Analysis of Financial Condition and Results of Operations | 36 |
| Business | 44 |
| Description of Property | 67 |
| Legal Proceedings | 68 |
| Management | 69 |
| Executive Compensation | 77 |
| Certain Relationships and Related Transactions | 80 |
| Security Ownership of Certain Beneficial Owners and Management | 81 |
| Description of Capital Stock | 84 |
| Description of Securities we are Offering | 86 |
| Underwriting | 88 |
| Notice to Investors | 90 |
| Legal Matters | 91 |
| Experts | 92 |
| Additional Information | 92 |
| Index to Financial Statements | 93 |
You should rely only on the information contained in this prospectus and any free writing prospectus prepared by us or on our behalf. We have not, and the underwriters have not, authorized anyone to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely on it. We are not, and the underwriters are not, making an offer to sell these securities in any jurisdiction where the offer is not permitted. The information contained in this prospectus and any free writing prospectus that we have authorized for use in connection with this offering is accurate only as of the date of those respective documents, regardless of the time of delivery of this prospectus or any authorized free writing prospectus or the time of issuance or sale of any securities. Our business, financial condition, results of operations and prospects may have changed since those dates. You should read this prospectus and any free writing prospectus that we have authorized for use in connection with this offering in their entirety before making an investment decision. You should also read and consider the information in the documents to which we have referred you in the section of this prospectus entitled “Where You Can Find More Information.”
| i |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, or Securities Act, and Section 21E of the Securities Exchange Act of 1934, or Exchange Act. Forward-looking statements reflect the current view about future events. When used in this prospectus, the words “anticipate,” “believe,” “estimate,” “expect,” “future,” “intend,” “plan,” or the negative of these terms and similar expressions, as they relate to us or our management, identify forward-looking statements. Such statements, include, but are not limited to, statements contained in this prospectus relating to our business strategy, our future operating results and liquidity and capital resources outlook. Forward-looking statements are based on our current expectations and assumptions regarding our business, the economy and other future conditions. Because forward–looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Our actual results may differ materially from those contemplated by the forward-looking statements. They are neither statements of historical fact nor guarantees of assurance of future performance. We caution you therefore against relying on any of these forward-looking statements. Important factors that could cause actual results to differ materially from those in the forward-looking statements include, without limitation, a continued decline in general economic conditions nationally and internationally; decreased demand for our products and services; market acceptance of our products and services; our ability to protect our intellectual property rights; the impact of any infringement actions or other litigation brought against us; competition from other providers and products; our ability to develop and commercialize new and improved products and services; our ability to raise capital to fund continuing operations; changes in government regulation; our ability to complete customer transactions and capital raising transactions; and other factors (including the risks contained in the section of this prospectus entitled “Risk Factors”) relating to our industry, our operations and results of operations and any businesses that may be acquired by us. Should one or more of these risks or uncertainties materialize, or should the underlying assumptions prove incorrect, actual results may differ significantly from those anticipated, believed, estimated, expected, intended or planned.
Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We cannot guarantee future results, levels of activity, performance or achievements. Except as required by applicable law, including the securities laws of the United States, we do not intend to update any of the forward-looking statements to conform these statements to actual results.
| 1 |
PROSPECTUS SUMMARY
This summary highlights information contained throughout this prospectus and is qualified in its entirety to the more detailed information and financial statements included elsewhere in this prospectus. This summary does not contain all of the information that should be considered before investing in our common stock. Investors should read the entire prospectus carefully, including the more detailed information regarding our business, the risks of purchasing our common stock discussed in this prospectus under “Risk Factors” beginning on page 9 of this prospectus and our financial statements and the accompanying notes beginning on page F-1 of this prospectus.
Unless otherwise indicated or unless the context requires otherwise, this prospectus includes the accounts of Tonix Pharmaceuticals Holding Corp., a Nevada corporation (“Tonix”), and together with its wholly-owned subsidiaries, as follows, collectively referred to as “we”, “us” or the “Company”: Tonix Pharmaceuticals, Inc., a Delaware corporation (“Tonix Sub”), Krele LLC, a Delaware limited liability company (“Krele”) and Tonix Pharmaceuticals (Canada), Inc., a corporation incorporated under the laws of the province of New Brunswick, Canada (“Tonix Canada”). Tonix Sub is a wholly-owned subsidiary of Tonix, and Krele and Tonix Canada are wholly-owned subsidiaries of Tonix Sub.
Our Company
Corporate Structure
We were incorporated on November 16, 2007 under the laws of the State of Nevada as Tamandare Explorations Inc. From inception through October 2011, we were involved in the acquisition, exploration and development of natural resource properties in the State of Nevada. On October 7, 2011, we executed and consummated a share exchange agreement by and among Tonix Sub and the stockholders of 100% of the equity securities of Tonix Sub, including, the holders of 5,207,500 shares of common stock, 1,500,000 shares of Series A Preferred Stock and 2,275,527 shares of Series B Preferred Stock (the “Tonix Shareholders”), on the one hand, and us and David Moss, our then sole officer and director and majority shareholder, on the other hand (the “Share Exchange Agreement” and the transaction, the “Share Exchange”).
In the Share Exchange, the Tonix Shareholders exchanged their shares of Tonix Sub for newly issued shares of our common stock (“Common Stock”). As a result, upon completion of the Share Exchange, Tonix Sub became our wholly-owned subsidiary.
Upon completion of the Share Exchange, the Tonix Shareholders received an aggregate of 1,133,334 shares of our Common Stock. Mr. Moss returned 75,000 shares of Common Stock to us, which were retired, and our existing shareholders retained 200,000 shares of Common Stock. The 1,133,334 shares issued to the Tonix Shareholders constituted approximately 85% of our 1,333,334 issued and outstanding shares of Common Stock immediately after the consummation of the Share Exchange.
As a result of the Share Exchange, we acquired 100% of the capital stock of Tonix Sub and consequently, control of the business and operations of Tonix Sub and Krele. From and after the consummation of the Share Exchange, our primary operations consist of the business and operations of Tonix Sub and Krele.
On October 11, 2011, we changed our name to Tonix Pharmaceuticals Holding Corp. to reflect our new business.
On April 23, 2013, we formed Tonix Canada as a wholly-owned subsidiary of Tonix Sub. Tonix Canada is intended to perform research and development efforts in Canada. As a Canadian entity, we expect Tonix Canada will be entitled to receive certain reimbursable tax credits for research expenditures in Canada.
Corporate Background
In 1996, Seth Lederman, MD, and Donald Landry, MD, PhD, formed L & L Technologies, LLC, or L&L, to develop medications for central nervous system, or CNS, conditions. Dr. Lederman is our Chairman and Chief Executive Officer and Dr. Landry is a Director. L&L was a founder of Janus Pharmaceuticals, Inc., which later became Vela Pharmaceuticals, Inc., or Vela, which developed various therapeutics, including a very low dose, or VLD, version of cyclobenzaprine, or CBP, under an agreement with L&L. Vela decided to focus its resources on other programs and transferred the rights to VLD CBP and certain other technologies to L&L in March 2006.
| 2 |
Tonix Sub formed in June 2007 as Krele Pharmaceuticals, Inc. by L&L and Plumbline LLC, or Plumbline. Dr. Lederman is Managing Partner of Plumbline. Plumbline possessed rights to certain technology for the treatment of alcohol dependence and abuse. In connection with founding Tonix Sub, L&L and Plumbline entered into an intellectual property transfer and assignment agreement with Tonix Sub for the purpose of assigning patents and transferring intellectual property and know-how in exchange for shares of common stock of Tonix Sub. As a result of economic conditions related to the financial crisis of 2007 and 2008, Tonix Sub was not successful in raising money to fund its programs until 2009. As a result, Tonix Sub was unable to advance the development programs and had little activity except for prosecuting and maintaining patents and maintaining contracts.
In 2009, Tonix Sub contracted with the Toronto Psychiatric Research Foundation to analyze the sleep data from a Phase 2a trial of bedtime VLD CBP in fibromyalgia, or FM (the “Moldofsky Study”). The Moldofsky Study was conducted in Canada by the Toronto Psychiatric Research Foundation, and Tonix Sub obtained the data from this study from L&L. In addition, in 2009, Tonix Sub contracted with Caliper Life Sciences Inc., or Caliper, to analyze the interactions of CBP with certain receptors. In June 2010, Tonix Sub entered into consulting agreements with L&L and Lederman & Co., LLC, or Lederman & Co, and also acquired certain rights to develop isometheptene mucate as a treatment for certain types of headaches from Lederman & Co, which we are developing as TNX-201. Dr. Lederman is managing partner of Lederman & Co. Between June 2010 and October 2011, Tonix Sub was active in recruiting new officers and directors and initiating preclinical and clinical development of novel CBP formulations.
Lederman & Co predominantly provides us with clinical development expertise. L&L predominantly provided us with scientific development expertise until the termination of the consulting agreement in June 2012. Relative to traditional pharmaceutical development companies, we can be considered a virtual company, since we contract with third-party vendors to provide many functions that are core to traditional pharmaceutical companies. For example, we have contracted with PharmaNet Canada, Inc., or PharmaNet Canada, to develop methods for analyzing CBP in the blood and to conduct human clinical studies to evaluate the performance of our formulation technology. Lederman & Co is responsible for overseeing the scientific and technical aspects of PharmaNet’s contract work product.
In July 2010, Tonix Sub changed its name to Tonix Pharmaceuticals, Inc. In August 2010, Tonix Sub formed Krele.
Business Overview
We are a specialty pharmaceutical company focused on developing novel pharmaceutical products for challenging disorders of the CNS. Our drug development programs target CNS conditions that manifest with pain that originates in the brain, or central pain. Central pain results from abnormal pain processing in the CNS, rather than from dysfunction in peripheral tissues where pain is perceived. Our lead program targets fibromyalgia, or FM, a central pain syndrome. Another program targets post-traumatic stress disorder, or PTSD, in which central pain is a component. Central pain is associated with disturbed sleep, and improvements in pain or sleep can sometimes help the other symptom. Our products for FM and PTSD are administered at bedtime. We believe that part of the effect on pain in FM is due to improving sleep quality. Our potential therapeutic solutions for FM and PTSD are based on a known pharmaceutical agent that has been approved for use in another indication. We are developing proprietary products that are new dose, formulation, and delivery options tailored to the new therapeutic uses. We intend to start a Phase 2b pivotal study for FM in the third quarter of 2013, which, if successful, will be the first of two pivotal studies required for marketing approval in the U.S.
We are currently devoting the majority of our efforts to the development of our lead product candidate, TNX-102 sublingual tablet, or TNX-102 SL. TNX-102 SL is a novel dose and formulation of CBP, the active pharmaceutical ingredient of two widely prescribed muscle relaxant products, Flexeril® and Amrix®. TNX-102 SL is distinct from these products as it is being developed at a dose level significantly below the lowest marketed doses of Flexeril and Amrix. TNX-102 SL is also distinct from these products with regard to its route of administration, as it is designed to be placed under the tongue, to disintegrate, dissolve and provide sublingual absorption, whereas Flexeril and Amrix are designed to be swallowed and to provide absorption in the small intestine. TNX-102 SL is also intended for chronic use, whereas Flexeril and Amrix are marketed for two to three weeks of use.
| 3 |
As FM patients do not typically experience muscle spasm, we believe that the use of muscle relaxants in FM is off-label from a regulatory perspective and provides therapeutic effects to FM patients that are different from those in treating muscle spasm. Therefore, in FM, CBP acts as a central pain inhibitor, and not as a muscle relaxant.
Many FM patients take prescription pain and sleep medications to treat their symptoms, despite the lack of objective, factual evidence that such medications provide a prolonged therapeutic effect. An important goal in FM treatment is to reduce the dependence on opiate, benzodiazepine and selective non-benzodiazepine medications by FM patients. Since CBP has no recognized addictive potential, a CBP product that decreases pain in FM could reduce the exposure of FM patients to ineffective off-label pain and sleep medications.
Our motivation to develop a product containing CBP for FM began when a clinician noticed that a VLD of oral CBP between dinner and bedtime improved symptoms in FM patients. Following these reports, a double-blind, randomized placebo-controlled study of VLD CBP between dinner and bedtime demonstrated a significant decrease in pain and other symptoms after eight weeks of therapy, and supported a therapeutic strategy based on CBP for FM.
We are currently developing TNX-102 SL for the treatment of FM under an Investigational New Drug application, or IND, filed in the US, and under three Clinical Trial Applications, or CTAs, filed in Canada. For our FM program, we held an End-of-Phase 2/Pre-Phase 3 meeting with the FDA in February 2013. We are also developing TNX-102 SL for the treatment of PTSD, for which we held a pre-IND meeting with the FDA in October 2012. We expect that any applications we submit for FDA approval of TNX-102 SL will be submitted under Section 505(b)(2) of the Federal Food, Drug, and Cosmetic Act, or FDCA, which we believe will allow for a shorter timeline of clinical development as compared to that needed to fulfill the requirements of Section 505(b)(1), under which new chemical entities, or NCEs, are generally reviewed.
TNX-102 SL is a small, rapidly disintegrating tablet containing CBP for sublingual administration at bedtime. We designed TNX-102 SL to enable the efficient delivery of CBP to the systemic circulation across the mouth’s mucous membrane, or transmucosal absorption, and to avoid first-pass liver metabolism. We also designed TNX-102 SL to provide CBP at doses lower than those currently available. We have conducted several clinical and pre-clinical pharmacokinetic studies of TNX-102 SL which we believe support its development as a novel therapeutic product for FM and PTSD, and which demonstrate a number of potentially advantageous characteristics as compared to current CBP-containing products, none of which are approved for these indications. Based on our Phase 1 comparative study, we have observed that, as compared to oral CBP, sublingual TNX-102 SL results in faster systemic absorption and significantly higher plasma levels of CBP in the first hour following administration. TNX-102 SL was generally well-tolerated, with no serious adverse events reported in this study. Some subjects experienced transient numbness on the tongue after TNX-102 SL administration, and other side-effects reported were similar to those associated with current CBP products.
As supported by these promising results, we are advancing TNX-102 SL for the management of FM. We held an End-of-Phase 2/Pre-Phase 3 meeting with the FDA in February 2013, at which we discussed the design of the clinical program, including the acceptability of the pivotal study design and the proposed registration plan, to support the approval of TNX-102 SL for the management of FM. We believe that positive results from two adequate, well-controlled safety and efficacy studies and the completion of long-term open-label safety exposure studies would support the approval of TNX-102 SL by the FDA for the management of FM. Under the IND, we plan to initiate a potential pivotal efficacy trial (Phase 2b) in FM in the third quarter of 2013. We have entered into a non-binding letter of intent to engage Premier Research International, LLC, a contract research organization, or CRO, to perform key services for the pivotal efficacy trial.
We are also advancing TNX-102 SL for the management of PTSD. We held a pre-IND meeting with the FDA in October 2012, and we plan to file an IND for this indication in the third quarter of 2013. We then plan to begin a Phase 2 proof-of-concept trial of TNX-102 SL in PTSD in the fourth quarter of 2013.
| 4 |
CBP is the active pharmaceutical ingredient in our lead product candidate, TNX-102 SL. CBP has been approved by the FDA in the U.S. since 1977. We have utilized drug delivery technology to produce new formulations of CBP. In addition to CBP, TNX-102 SL contains excipients, which are well-characterized, are listed in the Inactive Ingredient Guide and are approved for pharmaceutical use. As a result, we anticipate seeking FDA marketing approval of TNX-102 SL through a 505(b)(2) NDA. As one of three types of new drug applications, the 505(b)(2) NDA allows drug companies to obtain FDA approval of new drug products without having to conduct the full complement of safety and efficacy trials, which is often the most time-consuming and expensive part of the drug development process. As the 505(b)(2) NDA permits the drug manufacturer to rely on the agency’s findings for a previously-approved drug, published literature, or both, it permits the FDA to make some safety and effectiveness determinations through the review of materials in the public domain or in already approved NDAs of products containing CBP. The 505(b)(2) regulatory pathway would spare us some of the burden of generating all of this data for ourselves and may allow TNX-102 SL to progress through a shorter development pathway than is typical for pharmaceutical products based on novel active ingredients. We have not filed an NDA for TNX-102 SL for any indications.
In addition to TNX-102 SL, we have developed other innovative formulations of CBP, including TNX-102 promicellar gelatin capsule, or TNX-102 gelcap. We have developed TNX-102 gelcap under an agreement with Lipocine, Inc. or Lipocine, a contract formulation developer and small-scale manufacturer. Although we had met with the FDA to discuss the TNX-102 gelcap development program in August 2011 and we have generated clinical data that support the further development of TNX-102 gelcap, we currently do not plan to advance this candidate.
We also have a pipeline of other product candidates, including TNX-201 and TNX-301. TNX-201 is based on isometheptene mucate and is under development as a treatment for certain types of headaches. TNX-201 is a purified isomer of isometheptene mucate, which has been marketed and approved only as a mixture of two isomers. TNX-301 is a fixed dose combination of two FDA-approved drugs, disulfiram and selegiline, and is under development as a treatment for alcohol abuse and dependence. For competitive reasons, we do not disclose the identities of the active ingredients or targeted indications in our pipeline until a U.S. patent has been allowed or issued. Consistent with our mission, these product candidates are or likely will be reformulations of active ingredients that have been used in humans in other products and that are designed for new CNS therapeutic indications.
In other cases, the products will be formulated to match predicate products closely enough to be considered generic copies or similarly enough to other marketed products to rely (in part) on their regulatory review and approval, as well as available published data. The predicate product may be approved by the FDA under an NDA or may have been reviewed for safety and effectiveness by the National Academy of Sciences under the Drug Efficacy Study Implementation, or DESI, program, in which case they would be considered by FDA to be “unapproved products”. For DESI products, it is our intent to develop NDA versions to meet current Good Manufacturing Practices, or cGMP, and the International Conference on Harmonisation, or ICH, requirements to seek approval under the 505(b)(2) regulatory pathway.
Because of our size and being in the development stage, we do not currently devote a significant amount of time or resources towards our other pipeline candidates. We may perform non-clinical development work on TNX-201 and possibly on TNX-301, but we do not expect to start clinical trials of either of these candidates until 2014 at the earliest.
The process to bring a new drug formulation from concept through testing to approval for a new indication by the FDA is a time-consuming, costly and high-risk process. Several different studies, varying in time, number of patients, and complexity are required prior to applying for FDA approval. These studies include a Phase 1 study, a proof-of-concept efficacy study, a Phase 2b study or a Phase 3 study (a first pivotal study) and a second pivotal “confirmatory” Phase 3 study. These studies take several years to complete, however, since we reformulate versions of approved drugs for new uses, we may need to devote less time to Phase 1 studies. Assuming our clinical trials are successful, we would submit an NDA to the FDA seeking marketing approval of TNX-102 SL for the management of FM. As a result, it typically takes more than five years to bring a new formulation of an approved drug to market for a different indication, and any delays in the process, such as lack of capital necessary to run clinical trials, unexpected results, adverse effects, or difficulty in recruiting willing subjects for trials, would result in additional time before a drug could be approved for marketing.
| 5 |
In August 2010, we formed Krele to commercialize products that are generic versions of predicate NDA products. We anticipate that when our branded products lose patent protection, Krele may market authorized generic versions of them. Krele also may develop or acquire generic products approved under FDA abbreviated new drug applications, or ANDAs, and we may market branded versions (branded generics) of such products. Krele has been issued a state license in New York.
| 6 |
The Offering
| Securities we are offering: | [ * ] units, each consisting of one share of our common stock and [ * ] of a Series A Warrant, each to purchase one share of our common stock at an exercise price of $[ * ] per share. The Series A Warrants will be immediately exercisable and will expire on the [ * ] anniversary of the issuance date. | |
| Public offering price: | $[ * ] per unit. | |
| Common stock outstanding before this offering: | 2,197,490 shares. | |
| Common stock included in the units: | [ * ] shares. | |
| Common stock to be outstanding after this offering: | [ * ] shares. | |
| Use of proceeds: | We currently intend to use the net proceeds of this offering for the clinical development of TNX-102 SL, including a Phase 2b clinical trial for the treatment of FM, and for general corporate purposes, including working capital and operational purposes, including pre-clinical development. For a more complete description of our intended use of proceeds from this offering, see “Use of Proceeds.” | |
| Market Symbol and Listing: | Our common stock is currently quoted on the OTCQB under the symbol “TNXP”. We have applied to have our common stock listed on The NASDAQ Capital Market under the same symbol. There is no established trading market for the Series A Warrants and we do not expect a market to develop. In addition, we do not intend to apply for listing of the Series A Warrants on any national securities exchange or other trading market. | |
| Risk Factors: | Investing in our securities involves substantial risks. You should carefully review and consider the “Risk Factors” section of this prospectus for a discussion of factors to consider before deciding to invest in our securities. |
The number of shares of our common stock outstanding prior to and to be outstanding immediately after this offering, as set forth in the table above, is based on 2,197,490 shares outstanding as of May 9, 2013 and excludes:
| • | 1,232,400 shares of common stock issuable upon the exercise of warrants outstanding at May 9, 2013 with a weighted average exercise price of $14.52 per share; |
| • | 376,500 shares of common stock issuable upon the exercise of options outstanding at May 9, 2013 with a weighted average exercise price of $18.09 per share; |
| • | 173,500 shares of common stock reserved for future grants, awards and issuance under our equity compensation plan as of May 9, 2013; |
| • | shares of common stock issuable upon the exercise of the Series A Warrants offered hereby; and |
| • | shares of common stock issuable upon the exercise of the underwriter warrants. |
| 7 |
Except as otherwise indicated, all information in this prospectus reflects the 1-for-20 reverse stock split of our outstanding common stock that was effected on May 1, 2013.
| 8 |
RISK FACTORS
This investment has a high degree of risk. Before you invest you should carefully consider the risks and uncertainties described below and the other information in this prospectus. If any of the following risks actually occur, our business, operating results and financial condition could be harmed and the value of our stock could go down. This means you could lose all or a part of your investment.
RISKS RELATED TO OUR BUSINESS
We have a history of operating losses and expect to incur losses for the foreseeable future. We may never generate revenues or, if we are able to generate revenues, achieve profitability.
We are focused on product development, and we have not generated any revenues to date. We have incurred losses in each year of our operations, and we expect to continue to incur operating losses for the foreseeable future. These operating losses have adversely affected and are likely to continue to adversely affect our working capital, total assets and shareholders’ equity.
The Company and its prospects should be examined in light of the risks and difficulties frequently encountered by new and early stage companies in new and rapidly evolving markets. These risks include, among other things, the speed at which we can scale up operations, our complete dependence upon development of products that currently have no market acceptance, our ability to establish and expand our brand name, our ability to expand our operations to meet the commercial demand of our clients, our development of and reliance on strategic and customer relationships and our ability to minimize fraud and other security risks.
The process of developing our products requires significant clinical, development and laboratory testing and clinical trials. In addition, commercialization of our product candidates will require that we obtain necessary regulatory approvals and establish sales, marketing and manufacturing capabilities, either through internal hiring or through contractual relationships with others. We expect to incur substantial losses for the foreseeable future as a result of anticipated increases in our research and development costs, including costs associated with conducting preclinical testing and clinical trials, and regulatory compliance activities.
Our ability to generate revenues and achieve profitability will depend on numerous factors, including success in:
| · | developing and testing product candidates; | |
| · | receiving regulatory approvals; | |
| · | commercializing our products; and | |
| · | establishing a favorable competitive position. |
Many of these factors will depend on circumstances beyond our control. We cannot assure you that we will ever have a product approved by the FDA, that we will bring any product to market or, if we are successful in doing so, that we will ever become profitable.
We expect to incur substantial additional operating expenses over the next several years as our research, development, pre-clinical testing, and clinical trial activities increase. The amount of future losses and when, if ever, we will achieve profitability are uncertain. We have no products that have generated any commercial revenue, do not expect to generate revenues from the commercial sale of products in the near future, and might never generate revenues from the sale of products. Our ability to generate revenue and achieve profitability will depend on, among other things, successful completion of the development of our product candidates; obtaining necessary regulatory approvals from the FDA; establishing manufacturing, sales, and marketing arrangements with third parties; and raising sufficient funds to finance our activities. We might not succeed at any of these undertakings. If we are unsuccessful at some or all of these undertakings, our business, prospects, and results of operations may be materially adversely affected.
| 9 |
We have a limited operating history and we expect a number of factors to cause our operating results to fluctuate on a quarterly and annual basis, which may make it difficult to predict our future performance.
We are a development stage biopharmaceutical company with a limited operating history. Our operations to date have been primarily limited to developing our technology and undertaking preclinical studies and clinical trials of our lead product candidate, TNX-102 SL. We have not yet obtained regulatory approvals for TNX-102 SL or any of our other product candidates. Consequently, any predictions made about our future success or viability may not be as accurate as they could be if we had a longer operating history or commercialized products. Our financial condition has varied significantly in the past and will continue to fluctuate from quarter-to-quarter or year-to-year due to a variety of factors, many of which are beyond our control. Factors relating to our business that may contribute to these fluctuations include other factors described elsewhere in this prospectus and also include:
| · | our ability to obtain additional funding to develop our product candidates; | |
| · | delays in the commencement, enrollment and timing of clinical trials; | |
| · | the success of our clinical trials through all phases of clinical development, including our TNX-102 SL trial; | |
| · | any delays in regulatory review and approval of product candidates in clinical development; | |
| · | our ability to obtain and maintain regulatory approval for TNX-102 SL or any of our other product candidates in the United States and foreign jurisdictions; | |
| · | potential side effects of our product candidates that could delay or prevent commercialization, limit the indications for any approved drug, require the establishment of risk evaluation and mitigation strategies, or cause an approved drug to be taken off the market; | |
| · | our dependence on third-party contract manufacturing organizations, or CMOs, to supply or manufacture our products; | |
| · | our dependence on CROs to conduct our clinical trials; | |
| · | our ability to establish or maintain collaborations, licensing or other arrangements; | |
| · | market acceptance of our product candidates; | |
| · | our ability to establish and maintain an effective sales and marketing infrastructure, either through the creation of a commercial infrastructure or through strategic collaborations; | |
| · | competition from existing products or new products that may emerge; | |
| · | the ability of patients or healthcare providers to obtain coverage of or sufficient reimbursement for our products; | |
| · | our ability to leverage our proprietary technology platform to discover and develop additional product candidates; | |
| · | our ability and our licensors’ abilities to successfully obtain, maintain, defend and enforce intellectual property rights important to our business; | |
| · | our ability to attract and retain key personnel to manage our business effectively; | |
| · | our ability to build our finance infrastructure and improve our accounting systems and controls; | |
| · | potential product liability claims; | |
| · | potential liabilities associated with hazardous materials; and | |
| · | our ability to obtain and maintain adequate insurance policies. |
Accordingly, the results of any quarterly or annual periods should not be relied upon as indications of future operating performance.
We received a report from our independent registered public accounting firm with an explanatory paragraph for the year ended December 31, 2012 with respect to our ability to continue as a going concern. The existence of such a report may adversely affect our stock price and our ability to raise capital. There is no assurance that we will not receive a similar report for our year ended December 31, 2013.
In their report dated March 8, 2013, our independent registered public accounting firm expressed substantial doubt about our ability to continue as a going concern as we have incurred losses since inception of development stage, have a negative cash flow from operations, and require additional financing to fund future operations. Our ability to continue as a going concern is subject to our ability to obtain necessary funding from outside sources, including obtaining additional funding from the sale of our securities, obtaining loans and grants from various financial institutions where possible. Our continued net operating losses increase the difficulty in meeting such goals and there can be no assurances that such methods will prove successful.
| 10 |
We have no approved products on the market and therefore do not expect to generate any revenues from product sales in the foreseeable future, if at all.
To date, we have no approved product on the market and have generated no product revenues. We have funded our operations primarily from sales of our securities. We have not received, and do not expect to receive for at least the next several years, if at all, any revenues from the commercialization of our product candidates. To obtain revenues from sales of our product candidates, we must succeed, either alone or with third parties, in developing, obtaining regulatory approval for, manufacturing and marketing drugs with commercial potential. We may never succeed in these activities, and we may not generate sufficient revenues to continue our business operations or achieve profitability.
We are largely dependent on the success of our lead product candidate, TNX-102 SL, and we cannot be certain that this product candidate will receive regulatory approval or be successfully commercialized.
We currently have no products for sale, and we cannot guarantee that we will ever have any drug products approved for sale. We and our product candidates are subject to extensive regulation by the FDA and comparable regulatory authorities in other countries governing, among other things, research, testing, clinical trials, manufacturing, labeling, promotion, selling, adverse event reporting and recordkeeping. We are not permitted to market any of our product candidates in the United States until we receive approval of an NDA for a product candidate from the FDA or the equivalent approval from a foreign regulatory authority. Obtaining FDA approval is a lengthy, expensive and uncertain process. We currently have one lead product candidate, TNX-102 SL for the treatment of FM, and the success of our business currently depends on its successful development, approval and commercialization. Any projected sales or future revenue predictions are predicated upon FDA approval and market acceptance of TNX-102 SL. If projected sales do not materialize for any reason, it would have a material adverse effect on our business and our ability to continue operations.
TNX-102 SL has not completed the clinical development process; therefore, we have not yet submitted an NDA or foreign equivalent or received marketing approval for this product candidate anywhere in the world. The clinical development program for TNX-102 SL may not lead to commercial products for a number of reasons, including if we fail to obtain necessary approvals from the FDA or foreign regulatory authorities because our clinical trials fail to demonstrate to their satisfaction that this product candidate is safe and effective or the clinical program may be put on hold due to unexpected safety issues with marketed CBP products. We may also fail to obtain the necessary approvals if we have inadequate financial or other resources to advance our product candidates through the clinical trial process. Any failure or delay in completing clinical trials or obtaining regulatory approval for TNX-102 SL in a timely manner would have a material adverse impact on our business and our stock price.
We may use our financial and human resources to pursue a particular research program or product candidate and fail to capitalize on programs or product candidates that may be more profitable or for which there is a greater likelihood of success.
Because we have limited financial and human resources, we are currently focusing on the regulatory approval of TNX-102 SL. As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on existing and future product candidates for specific indications may not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product candidate through strategic alliance, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product candidate, or we may allocate internal resources to a product candidate in a therapeutic area in which it would have been more advantageous to enter into a partnering arrangement.
| 11 |
We need additional capital. If additional capital is not available or is available at unattractive terms, we may be forced to delay, reduce the scope of or eliminate our research and development programs, reduce our commercialization efforts or curtail our operations.
In order to develop and bring our product candidates to market, we must commit substantial resources to costly and time-consuming research, preclinical and clinical trials and marketing activities. We anticipate that our existing cash and cash equivalents will enable us to maintain our current operations for at least the next six months, and we anticipate that we will require additional capital to complete the planned pivotal trial of TNX-102 SL in FM. We anticipate using our cash and cash equivalents to fund further research and development with respect to our lead product candidates. We may, however, need to raise additional funding sooner if our business or operations change in a manner that consumes available resources more rapidly than we anticipate. Our requirements for additional capital will depend on many factors, including:
| · | successful commercialization of our product candidates; | |
| · | the time and costs involved in obtaining regulatory approval for our product candidates; | |
| · | costs associated with protecting our intellectual property rights; | |
| · | development of marketing and sales capabilities; | |
| · | payments received under future collaborative agreements, if any; and | |
| · | market acceptance of our products. |
To the extent we raise additional capital through the sale of equity securities, the issuance of those securities could result in dilution to our shareholders. In addition, if we obtain debt financing, a substantial portion of our operating cash flow may be dedicated to the payment of principal and interest on such indebtedness, thus limiting funds available for our business activities. If adequate funds are not available, we may be required to delay, reduce the scope of or eliminate our research and development programs, reduce our commercialization efforts or curtail our operations. In addition, we may be required to obtain funds through arrangements with collaborative partners or others that may require us to relinquish rights to technologies, product candidates or products that we would otherwise seek to develop or commercialize ourselves or license rights to technologies, product candidates or products on terms that are less favorable to us than might otherwise be available.
We will require substantial additional funds to support our research and development activities, and the anticipated costs of preclinical studies and clinical trials, regulatory approvals and eventual commercialization. Such additional sources of financing may not be available on favorable terms, if at all. If we do not succeed in raising additional funds on acceptable terms, we may be unable to initiate clinical trials or obtain approval of any product candidates from the FDA and other regulatory authorities. In addition, we could be forced to discontinue product development, forego sales and marketing efforts and forego attractive business opportunities. Any additional sources of financing will likely involve the issuance of our equity securities, which will have a dilutive effect on our shareholders.
There is no assurance that we will be successful in raising the additional funds needed to fund our business plan. If we are not able to raise sufficient capital in the near future, our continued operations will be in jeopardy and we may be forced to cease operations and sell or otherwise transfer all or substantially all of our remaining assets.
We face intense competition in the markets targeted by our lead product candidates. Many of our competitors have substantially greater resources than we do, and we expect that all of our product candidates under development will face intense competition from existing or future drugs.
We expect that all of our product candidates under development, if approved, will face intense competition from existing and future drugs marketed by large companies. These competitors may successfully market products that compete with our products, successfully identify drug candidates or develop products earlier than we do, or develop products that are more effective, have fewer side effects or cost less than our products.
| 12 |
Additionally, if a competitor receives FDA approval before we do for a drug that is similar to one of our product candidates, FDA approval for our product candidate may be precluded or delayed due to periods of non-patent exclusivity and/or the listing with the FDA by the competitor of patents covering its newly-approved drug product. Periods of non-patent exclusivity for new versions of existing drugs such as our current product candidates can extend up to three and one-half years. See “Business—Government Regulation.”
These competitive factors could require us to conduct substantial new research and development activities to establish new product targets, which would be costly and time consuming. These activities would adversely affect our ability to commercialize products and achieve revenue and profits.
Competition and technological change may make our product candidates and technologies less attractive or obsolete.
We compete with established pharmaceutical and biotechnology companies that are pursuing other forms of treatment for the same indications we are pursuing and that have greater financial and other resources. Other companies may succeed in developing products earlier than us, obtaining FDA approval for products more rapidly, or developing products that are more effective than our product candidates. Research and development by others may render our technology or product candidates obsolete or noncompetitive, or result in treatments or cures superior to any therapy we develop. We face competition from companies that internally develop competing technology or acquire competing technology from universities and other research institutions. As these companies develop their technologies, they may develop competitive positions that may prevent, make futile, or limit our product commercialization efforts, which would result in a decrease in the revenue we would be able to derive from the sale of any products.
There can be no assurance that any of our product candidates will be accepted by the marketplace as readily as these or other competing treatments. Furthermore, if our competitors' products are approved before ours, it could be more difficult for us to obtain approval from the FDA. Even if our products are successfully developed and approved for use by all governing regulatory bodies, there can be no assurance that physicians and patients will accept our product(s) as a treatment of choice.
Furthermore, the pharmaceutical research industry is diverse, complex, and rapidly changing. By its nature, the business risks associated therewith are numerous and significant. The effects of competition, intellectual property disputes, market acceptance, and FDA regulations preclude us from forecasting revenues or income with certainty or even confidence.
If we fail to protect our intellectual property rights, our ability to pursue the development of our technologies and products would be negatively affected.
Our success will depend in part on our ability to obtain patents and maintain adequate protection of our technologies and products. If we do not adequately protect our intellectual property, competitors may be able to use our technologies to produce and market drugs in direct competition with us and erode our competitive advantage. Some foreign countries lack rules and methods for defending intellectual property rights and do not protect proprietary rights to the same extent as the United States. Many companies have had difficulty protecting their proprietary rights in these foreign countries. We may not be able to prevent misappropriation of our proprietary rights.
We have received, and are currently seeking, patent protection for numerous compounds and methods of treating diseases. However, the patent process is subject to numerous risks and uncertainties, and there can be no assurance that we will be successful in protecting our products by obtaining and defending patents. These risks and uncertainties include the following: patents that may be issued or licensed may be challenged, invalidated, or circumvented, or otherwise may not provide any competitive advantage; our competitors, many of which have substantially greater resources than us and many of which have made significant investments in competing technologies, may seek, or may already have obtained, patents that will limit, interfere with, or eliminate our ability to make, use, and sell our potential products either in the United States or in international markets; there may be significant pressure on the United States government and other international governmental bodies to limit the scope of patent protection both inside and outside the United States for treatments that prove successful as a matter of public policy regarding worldwide health concerns; countries other than the United States may have less restrictive patent laws than those upheld by United States courts, allowing foreign competitors the ability to exploit these laws to create, develop, and market competing products.
| 13 |
Moreover, any patents issued to us may not provide us with meaningful protection, or others may challenge, circumvent or narrow our patents. Third parties may also independently develop products similar to our products, duplicate our unpatented products or design around any patents on products we develop. Additionally, extensive time is required for development, testing and regulatory review of a potential product. While extensions of patent term due to regulatory delays may be available, it is possible that, before any of our product candidates can be commercialized, any related patent, even with an extension, may expire or remain in force for only a short period following commercialization, thereby reducing any advantages of the patent.
In addition, the United States Patent and Trademark Office (the “PTO”) and patent offices in other jurisdictions have often required that patent applications concerning pharmaceutical and/or biotechnology-related inventions be limited or narrowed substantially to cover only the specific innovations exemplified in the patent application, thereby limiting the scope of protection against competitive challenges. Thus, even if we or our licensors are able to obtain patents, the patents may be substantially narrower than anticipated.
Our success depends on our patents, patent applications that may be licensed exclusively to us and other patents to which we may obtain assignment or licenses. We may not be aware, however, of all patents, published applications or published literature that may affect our business either by blocking our ability to commercialize our product candidates, by preventing the patentability of our product candidates to us or our licensors, or by covering the same or similar technologies that may invalidate our patents, limit the scope of our future patent claims or adversely affect our ability to market our product candidates.
In addition to patents, we rely on a combination of trade secrets, confidentiality, nondisclosure and other contractual provisions, and security measures to protect our confidential and proprietary information. These measures may not adequately protect our trade secrets or other proprietary information. If they do not adequately protect our rights, third parties could use our technology, and we could lose any competitive advantage we may have. In addition, others may independently develop similar proprietary information or techniques or otherwise gain access to our trade secrets, which could impair any competitive advantage we may have.
Patent protection and other intellectual property protection is crucial to the success of our business and prospects, and there is a substantial risk that such protections will prove inadequate.
We may be involved in lawsuits to protect or enforce our patents, which could be expensive and time consuming.
The pharmaceutical industry has been characterized by extensive litigation regarding patents and other intellectual property rights, and companies have employed intellectual property litigation to gain a competitive advantage. We may become subject to infringement claims or litigation arising out of patents and pending applications of our competitors, or additional interference proceedings declared by the PTO to determine the priority of inventions. The defense and prosecution of intellectual property suits, PTO proceedings, and related legal and administrative proceedings are costly and time-consuming to pursue, and their outcome is uncertain. Litigation may be necessary to enforce our issued patents, to protect our trade secrets and know-how, or to determine the enforceability, scope, and validity of the proprietary rights of others. An adverse determination in litigation or interference proceedings to which we may become a party could subject us to significant liabilities, require us to obtain licenses from third parties, or restrict or prevent us from selling our products in certain markets. Although patent and intellectual property disputes might be settled through licensing or similar arrangements, the costs associated with such arrangements may be substantial and could include our paying large fixed payments and ongoing royalties. Furthermore, the necessary licenses may not be available on satisfactory terms or at all.
Competitors may infringe our patents, and we may file infringement claims to counter infringement or unauthorized use. This can be expensive, particularly for a company of our size, and time-consuming. In addition, in an infringement proceeding, a court may decide that a patent of ours is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover its technology. An adverse determination of any litigation or defense proceedings could put one or more of our patents at risk of being invalidated or interpreted narrowly.
| 14 |
Also, a third party may assert that our patents are invalid and/or unenforceable. There are no unresolved communications, allegations, complaints or threats of litigation related to the possibility that our patents are invalid or unenforceable. Any litigation or claims against us, whether or not merited, may result in substantial costs, place a significant strain on our financial resources, divert the attention of management and harm our reputation. An adverse decision in litigation could result in inadequate protection for our product candidates and/or reduce the value of any license agreements we have with third parties.
Interference proceedings brought before the U.S. Patent and Trademark Office may be necessary to determine priority of invention with respect to our patents or patent applications. During an interference proceeding, it may be determined that we do not have priority of invention for one or more aspects in our patents or patent applications and could result in the invalidation in part or whole of a patent or could put a patent application at risk of not issuing. Even if successful, an interference proceeding may result in substantial costs and distraction to our management.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation or interference proceedings, there is a risk that some of our confidential information could be compromised by disclosure. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. If investors perceive these results to be negative, the price of our common stock could be adversely affected.
If we infringe the rights of third parties we could be prevented from selling products, forced to pay damages, and defend against litigation.
If our products, methods, processes and other technologies infringe the proprietary rights of other parties, we could incur substantial costs and we may have to: obtain licenses, which may not be available on commercially reasonable terms, if at all; abandon an infringing product candidate; redesign our products or processes to avoid infringement; stop using the subject matter claimed in the patents held by others; pay damages; and/or defend litigation or administrative proceedings which may be costly whether we win or lose, and which could result in a substantial diversion of our financial and management resources.
If preclinical testing or clinical trials for our product candidates are unsuccessful or delayed, we will be unable to meet our anticipated development and commercialization timelines.
We rely and expect to continue to rely on third parties, including CROs and outside consultants, to conduct, supervise or monitor some or all aspects of preclinical testing or clinical trials involving our product candidates. We have less control over the timing and other aspects of these preclinical testing or clinical trials than if we performed the monitoring and supervision entirely on our own. Third parties may not perform their responsibilities for our preclinical testing or clinical trials on our anticipated schedule or, for clinical trials, consistent with a clinical trial protocol. Delays in preclinical and clinical testing could significantly increase our product development costs and delay product commercialization. In addition, many of the factors that may cause, or lead to, a delay in the clinical trials may also ultimately lead to denial of regulatory approval of a product candidate.
The commencement of clinical trials can be delayed for a variety of reasons, including delays in:
| · | demonstrating sufficient safety and efficacy to obtain regulatory approval to commence a clinical trial; | |
| · | reaching agreement on acceptable terms with prospective contract research organizations and trial sites; | |
| · | manufacturing sufficient quantities of a product candidate; and | |
| · | obtaining institutional review board approval to conduct a clinical trial at a prospective site. |
| 15 |
Once a clinical trial has begun, it may be delayed, suspended or terminated by us or the FDA or other regulatory authorities due to a number of factors, including:
| · | ongoing discussions with the FDA or other regulatory authorities regarding the scope or design of our clinical trials; | |
| · | failure to conduct clinical trials in accordance with regulatory requirements; | |
| · | lower than anticipated recruitment or retention rate of patients in clinical trials; | |
| · | inspection of the clinical trial operations or trial sites by the FDA or other regulatory authorities resulting in the imposition of a clinical hold; | |
| · | lack of adequate funding to continue clinical trials; | |
| · | negative results of clinical trials; or | |
| · | side-effects of CBP. |
If clinical trials are unsuccessful, and we are not able to obtain regulatory approvals for our product candidates under development, we will not be able to commercialize these products, and therefore may not be able to generate sufficient revenues to support our business.
We rely on third parties to conduct, supervise and monitor our clinical trials, and if those third parties perform in an unsatisfactory manner, it may harm our business.
We rely on CROs and clinical trial sites to ensure the proper and timely conduct of our clinical trials. While we have agreements governing their activities, we will have limited influence over their actual performance. We will control only certain aspects of our CROs’ activities. Nevertheless, we will be responsible for ensuring that our clinical trials are conducted in accordance with the applicable protocol, legal, regulatory and scientific standards and our reliance on the CROs does not relieve us of our regulatory responsibilities.
We and our CROs are required to comply with the FDA’s current good clinical practices requirements, or cGCP, for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of clinical trial participants are protected. The FDA enforces these cGCPs through periodic inspections of trial sponsors, principal investigators and clinical trial sites. If we or our CROs fail to comply with applicable cGCPs, the clinical data generated in our clinical trials may be deemed unreliable and the FDA may require us to perform additional clinical trials before approving any marketing applications. Upon inspection, the FDA may determine that our clinical trials did not comply with cGCPs. In addition, our clinical trials, including our Phase 2b trial of TNX-102 SL in FM, will require a sufficiently large number of test subjects to evaluate the safety and effectiveness of a product candidate. Accordingly, if our CROs fail to comply with these regulations or fail to recruit a sufficient number of patients, our clinical trials may be delayed or we may be required to repeat such clinical trials, which would delay the regulatory approval process.
Our CROs are not our employees, and we are not able to control whether or not they devote sufficient time and resources to our clinical trials. These CROs may also have relationships with other commercial entities, including our competitors, for whom they may also be conducting clinical trials, or other drug development activities which could harm our competitive position. If our CROs do not successfully carry out their contractual duties or obligations, fail to meet expected deadlines, or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols or regulatory requirements, or for any other reasons, our clinical trials may be extended, delayed or terminated, and we may not be able to obtain regulatory approval for, or successfully commercialize our product candidates. As a result, our financial results and the commercial prospects for such product candidates would be harmed, our costs could increase, and our ability to generate revenues could be delayed.
We also rely on other third parties to store and distribute drug products for our clinical trials. Any performance failure on the part of our distributors could delay clinical development or marketing approval of our product candidates or commercialization of our products, if approved, producing additional losses and depriving us of potential product revenue.
| 16 |
We have never conducted a Phase 3 clinical trial or submitted an NDA before, and may be unable to do so for TNX-102 SL and other product candidates we are developing.
If our Phase 2b study of TNX-102 SL is successful, then we expect to conduct a Phase 3 confirmatory study in support of product registration. The conduct of Phase 3 clinical trials and the submission of a successful NDA is a complicated process. Although members of our management team have extensive industry experience, including in the development, clinical testing and commercialization of drug candidates, our company has never conducted a Phase 3 clinical trial before, has limited experience in preparing, submitting and prosecuting regulatory filings, and has not submitted an NDA before. Consequently, we may be unable to successfully and efficiently execute and complete these planned clinical trials in a way that leads to NDA submission and approval of TNX-102 SL and other product candidates we are developing. We may require more time and incur greater costs than our competitors and may not succeed in obtaining regulatory approvals of product candidates that we develop. Failure to commence or complete, or delays in, our planned clinical trials would prevent or delay commercialization of TNX-102 SL and other product candidates we are developing.
Our product candidates may cause serious adverse events or undesirable side effects which may delay or prevent marketing approval, or, if approval is received, require them to be taken off the market, require them to include safety warnings or otherwise limit their sales.
Serious adverse events or undesirable side effects from TNX-102 SL or any of our other product candidates could arise either during clinical development or, if approved, after the approved product has been marketed. The results of future clinical trials, including TNX-102 SL, may show that our product candidates cause serious adverse events or undesirable side effects, which could interrupt, delay or halt clinical trials, resulting in delay of, or failure to obtain, marketing approval from the FDA and other regulatory authorities.
If TNX-102 SL or any of our other product candidates cause serious adverse events or undesirable side effects:
| · | regulatory authorities may impose a clinical hold which could result in substantial delays and adversely impact our ability to continue development of the product; | |
| · | regulatory authorities may require the addition of labeling statements, specific warnings, a contraindication or field alerts to physicians and pharmacies; | |
| · | we may be required to change the way the product is administered, conduct additional clinical trials or change the labeling of the product; | |
| · | we may be required to implement a risk minimization action plan, which could result in substantial cost increases and have a negative impact on our ability to commercialize the product; | |
| · | we may be required to limit the patients who can receive the product; | |
| · | we may be subject to limitations on how we promote the product; | |
| · | sales of the product may decrease significantly; | |
| · | regulatory authorities may require us to take our approved product off the market; | |
| · | we may be subject to litigation or product liability claims; and | |
| · | our reputation may suffer. |
Any of these events could prevent us from achieving or maintaining market acceptance of the affected product or could substantially increase commercialization costs and expenses, which in turn could delay or prevent us from generating significant revenues from the sale of our products.
| 17 |
If we are unable to file for approval under Section 505(b)(2) of the Federal Food, Drug and Cosmetic Act or if we are required to generate additional data related to safety and efficacy in order to obtain approval under Section 505(b)(2), we may be unable to meet our anticipated development and commercialization timelines.
Our current plans for filing NDAs for our product candidates include efforts to minimize the data we will be required to generate in order to obtain marketing approval for our product candidates and therefore possibly obtain a shortened review period for the applications. We met with the FDA in February 2013 to discuss the development of our lead product candidate, TNX-102 SL, in FM. We had held a pre-IND meeting in August 2011 to discuss initial plans for the development of TNX-102 gelcap in FM. Although these interactions with the FDA have encouraged our efforts to continue to develop TNX-102 SL for FM, there is no assurance that we will satisfy the FDA’s requirements for approval in this indication. We have not come to any agreement with the FDA as to the nature and extent of studies we may be required to conduct in order to achieve approval of TNX-102 SL in PTSD. The timeline for filing and review of our NDAs is based on our plan to submit those NDAs under Section 505(b)(2) of the Federal Food, Drug and Cosmetic Act, wherein we will rely in part on data in the public domain or elsewhere. We have not yet filed an NDA under Section 505(b)(2) for any of our lead product candidates. Depending on the data that may be required by the FDA for approval, some of the data may be related to products already approved by the FDA. If the data relied upon is related to products already approved by the FDA and covered by third-party patents we would be required to certify that we do not infringe the listed patents or that such patents are invalid or unenforceable. As a result of the certification, the third-party would have 45 days from notification of our certification to initiate an action against us. In the event that an action is brought in response to such a certification, the approval of our NDA could be subject to a stay of up to 30 months or more while we defend against such a suit. Approval of our product candidates under Section 505(b)(2) may therefore be delayed until patent exclusivity expires or until we successfully challenge the applicability of those patents to our product candidates. Alternatively, we may elect to generate sufficient additional clinical data so that we no longer rely on data which triggers a potential stay of the approval of our product candidates. Even if no exclusivity periods apply to our applications under Section 505(b)(2), the FDA has broad discretion to require us to generate additional data on the safety and efficacy of our product candidates to supplement third-party data on which we may be permitted to rely. In either event, we could be required, before obtaining marketing approval for any of our product candidates, to conduct substantial new research and development activities beyond those we currently plan to engage in order to obtain approval of our product candidates. Such additional new research and development activities would be costly and time consuming.
We may not be able to obtain shortened review of our applications, and the FDA may not agree that our products qualify for marketing approval. If CBP-containing products are withdrawn from the market by the FDA for any reason, we may not be able to reference such products to support a 505(b)(2) NDA for TNX-102 SL, and we may need to fulfill the more extensive requirements of Section 505(b)(1). If we are required to generate additional data to support approval, we may be unable to meet our anticipated development and commercialization timelines, may be unable to generate the additional data at a reasonable cost, or at all, and may be unable to obtain marketing approval of our product candidates.
We will need to expand our operations and increase the size of our company, and we may experience difficulties in managing growth.
As we advance our product candidates through preclinical studies and clinical trials and develop new product candidates, we will need to increase our product development, scientific and administrative headcount to manage these programs. In addition, to meet our obligations as a public company, we will need to increase our general and administrative capabilities. Our management, personnel and systems currently in place may not be adequate to support this future growth. Our need to effectively manage our operations, growth and various projects requires that we:
| · | successfully attract and recruit new employees with the expertise and experience we will require; | |
| · | manage our clinical programs effectively, which we anticipate being conducted at numerous clinical sites; | |
| · | develop a marketing, distribution and sales infrastructure if we seek to market our products directly; and | |
| · | continue to improve our operational, manufacturing, financial and management controls, reporting systems and procedures. |
If we are unable to successfully manage this growth and increased complexity of operations, our business may be adversely affected.
| 18 |
Our executive officers and other key personnel are critical to our business, and our future success depends on our ability to retain them.
Our success depends to a significant extent upon the continued services of Dr. Seth Lederman, our President and Chief Executive Officer. Dr. Lederman has overseen Tonix Sub since inception and provides leadership for our growth and operations strategy as well as being an inventor on many of our patents. Loss of the services of Dr. Lederman would have a material adverse effect on our growth, revenues, and prospective business. We have key-man insurance on the lives of Dr. Lederman, Dr. Leland Gershell, our Chief Financial Officer, and Dr. Bruce Daugherty, our Senior Director of Drug Development. We are also highly dependent on our directors and scientific team. We are not aware of any present intention of any of our key personnel to leave our company or to retire. However, we have no employment agreement with Dr. Lederman and while we have employment agreements with certain of our employees, all of our employees may terminate their employment at any time. The loss of any of our key personnel, or the inability to attract and retain qualified personnel, may significantly delay or prevent the achievement of our research, development or business objectives and could materially adversely affect our business, financial condition and results of operations.
Any employment agreement we enter into will not ensure the retention of the employee who is a party to the agreement. In addition, we have only limited ability to prevent former employees from competing with us. Furthermore, our future success will also depend in part on the continued service of our key scientific and management personnel and our ability to identify, hire, and retain additional personnel. We experience intense competition for qualified personnel and may be unable to attract and retain the personnel necessary for the development of our business. Moreover, our work force is located in the “Pharmaceutical Corridor” that spans New York, New Jersey and Pennsylvania, where competition for personnel with the scientific and technical skills that we seek is extremely high and is likely to remain high. Because of this competition, our compensation costs may increase significantly.
If we are unable to hire additional qualified personnel, our ability to grow our business may be harmed.
Over time we will need to hire additional qualified personnel with expertise in drug development, product registration, clinical and non-clinical research, quality compliance, government regulation, formulation and manufacturing, financial matters and sales and marketing. We compete for qualified individuals with numerous biopharmaceutical companies, universities and other research institutions. Competition for such individuals is intense, and we cannot be certain that our search for such personnel will be successful. Attracting and retaining qualified personnel will be critical to our success.
We rely on third parties to manufacture the compounds used in our trials, and we intend to rely on them for the manufacture of any approved products for commercial sale. If these third parties do not manufacture our product candidates in sufficient quantities and at an acceptable cost, clinical development and commercialization of our product candidates could be delayed, prevented or impaired.
We have no manufacturing facilities, and we have no experience in the clinical or commercial-scale manufacture of drugs or in designing drug manufacturing processes. We intend to rely on CMOs to manufacture some or all of our product candidates in clinical trials and our products that reach commercialization. Completion of our clinical trials and commercialization of our product candidates requires manufacturing of a sufficient supply of our product candidates. We have contracted with outside sources to manufacture our development compounds, including TNX-102 SL. If, for any reason, we become unable to rely on our current sources for the manufacture of our product candidates, either for clinical trials or, at some future date, for commercial quantities, then we would need to identify and contract with additional or replacement third-party manufacturers to manufacture compounds for pre-clinical, clinical, and commercial purposes. Although we are in discussions with other manufacturers we have identified as potential alternative CMOs of TNX-102 SL, we may not be successful in negotiating acceptable terms with any of them.
| 19 |
We believe that there are a variety of manufacturers that we may be able to retain to produce these products. However, once we retain a manufacturing source, if our manufacturers do not perform in a satisfactory manner, we may not be able to develop or commercialize potential products as planned. Certain specialized manufacturers are expected to provide us with modified and unmodified pharmaceutical compounds, including finished products, for use in our preclinical and clinical studies. Some of these materials are available from only one supplier or vendor. Any interruption in or termination of service by such sole source suppliers could result in a delay or interruption in manufacturing until we locate an alternative source of supply. Any delay or interruption in manufacturing operations (or failure to locate a suitable replacement for such suppliers) could materially adversely affect our business, prospects, or results of operations. We do not have any short-term or long-term manufacturing agreements with many of these manufacturers. If we fail to contract for manufacturing on acceptable terms or if third-party manufacturers do not perform as we expect, our development programs could be materially adversely affected. This may result in delays in filing for and receiving FDA approval for one or more of our products. Any such delays could cause our prospects to suffer significantly.
Failure by our third-party manufacturers to comply with the regulatory guidelines set forth by the FDA with respect to our product candidates could delay or prevent the completion of clinical trials, the approval of any product candidates or the commercialization of our products.
Such third-party manufacturers must be inspected by FDA for cGMP compliance before they can produce commercial product. We may be in competition with other companies for access to these manufacturers' facilities and may be subject to delays in manufacture if the manufacturers give other clients higher priority than they give to us. If we are unable to secure and maintain third-party manufacturing capacity, the development and sales of our products and our financial performance may be materially affected.
Manufacturers are obligated to operate in accordance with FDA-mandated requirements. A failure of any of our third-party manufacturers to establish and follow cGMP requirements and to document their adherence to such practices may lead to significant delays in the availability of material for clinical trials, may delay or prevent filing or approval of marketing applications for our products, and may cause delays or interruptions in the availability of our products for commercial distribution following FDA approval. This could result in higher costs to us or deprive us of potential product revenues.
Complying with cGMP and non-U.S. regulatory requirements will require that we expend time, money, and effort in production, recordkeeping, and quality control to assure that the product meets applicable specifications and other requirements. We, or our contracted manufacturing facility, must also pass a pre-approval inspection prior to FDA approval. Failure to pass a pre-approval inspection may significantly delay FDA approval of our products. If we fail to comply with these requirements, we would be subject to possible regulatory action and may be limited in the jurisdictions in which we are permitted to sell our products. As a result, our business, financial condition, and results of operations may be materially harmed.
Drug manufacturers are subject to ongoing periodic unannounced inspections by the FDA, the Drug Enforcement Agency and corresponding state and foreign agencies to ensure strict compliance with cGMP requirements and other requirements under Federal drug laws, other government regulations and corresponding foreign standards. If we or our third-party manufacturers fail to comply with applicable regulations, sanctions could be imposed on us, including fines, injunctions, civil penalties, failure by the government to grant marketing approval of drugs, delays, suspension or withdrawal of approvals, seizures or recalls of product, operating restrictions and criminal prosecutions.
Corporate and academic collaborators may take actions to delay, prevent, or undermine the success of our products.
Our operating and financial strategy for the development, clinical testing, manufacture, and commercialization of drug candidates is heavily dependent on our entering into collaborations with corporations, academic institutions, licensors, licensees, and other parties. Our current strategy assumes that we will successfully establish these collaborations, or similar relationships; however, there can be no assurance that we will be successful establishing such collaborations. Some of our existing collaborations are, and future collaborations may be, terminable at the sole discretion of the collaborator. Replacement collaborators might not be available on attractive terms, or at all. The activities of any collaborator will not be within our control and may not be within our power to influence. There can be no assurance that any collaborator will perform its obligations to our satisfaction or at all, that we will derive any revenue or profits from such collaborations, or that any collaborator will not compete with us. If any collaboration is not pursued, we may require substantially greater capital to undertake development and marketing of our proposed products and may not be able to develop and market such products effectively, if at all. In addition, a lack of development and marketing collaborations may lead to significant delays in introducing proposed products into certain markets and/or reduced sales of proposed products in such markets.
| 20 |
Data provided by collaborators and others upon which we rely that has not been independently verified could turn out to be false, misleading, or incomplete.
We rely on third-party vendors, scientists, and collaborators to provide us with significant data and other information related to our projects, clinical trials, and our business. If such third parties provide inaccurate, misleading, or incomplete data, our business, prospects, and results of operations could be materially adversely affected.
Our product candidates are novel and still in development.
We are a pharmaceutical company focused on the development of drug product candidates, all of which are still in development. Our drug development methods may not lead to commercially viable drugs for any of several reasons. For example, we may fail to identify appropriate targets or compounds, our drug candidates may fail to be safe and effective in clinical trials, or we may have inadequate financial or other resources to pursue development efforts for our drug candidates. Our drug candidates will require significant additional development, clinical trials, regulatory clearances and additional investment by us or our collaborators before they can be commercialized.
Successful development of our products is uncertain.
Our development of current and future product candidates is subject to the risks of failure and delay inherent in the development of new pharmaceutical products, including: delays in product development, clinical testing, or manufacturing; unplanned expenditures in product development, clinical testing, or manufacturing; failure to receive regulatory approvals; emergence of superior or equivalent products; inability to manufacture on its own, or through any others, product candidates on a commercial scale; and failure to achieve market acceptance.
Because of these risks, our research and development efforts may not result in any commercially viable products. If a significant portion of these development efforts are not successfully completed, required regulatory approvals are not obtained or any approved products are not commercially successfully, our business, financial condition, and results of operations may be materially harmed.
Clinical trials required for our product candidates are expensive and time-consuming, and their outcome is uncertain.
In order to obtain FDA approval to market a new drug product, we must demonstrate proof of safety and effectiveness in humans. To meet these requirements, we must conduct “adequate and well controlled” clinical trials. Conducting clinical trials is a lengthy, time-consuming, and expensive process. The length of time may vary substantially according to the type, complexity, novelty, and intended use of the product candidate, and often can be several years or more per trial. Delays associated with products for which we are directly conducting clinical trials may cause us to incur additional operating expenses. The commencement and rate of completion of clinical trials may be delayed by many factors, including, for example: inability to manufacture sufficient quantities of qualified materials under cGMP, for use in clinical trials; slower than expected rates of patient recruitment; failure to recruit a sufficient number of patients; modification of clinical trial protocols; changes in regulatory requirements for clinical trials; the lack of effectiveness during clinical trials; the emergence of unforeseen safety issues; delays, suspension, or termination of the clinical trials due to the institutional review board responsible for overseeing the study at a particular study site; and government or regulatory delays or “clinical holds” requiring suspension or termination of the trials.
The results from early clinical trials are not necessarily predictive of results obtained in later clinical trials. Accordingly, even if we obtain positive results from early clinical trials, we may not achieve the same success in future clinical trials. Clinical trials may not demonstrate statistically significant safety and effectiveness to obtain the requisite regulatory approvals for product candidates.
| 21 |
Our clinical trials may be conducted in patients with CNS conditions, and in some cases, our products are expected to be used in combination with approved therapies that themselves have significant adverse event profiles. During the course of treatment, these patients could suffer adverse medical events or die for reasons that may or may not be related to our products. We cannot ensure that safety issues will not arise with respect to our products in clinical development.
The failure of clinical trials to demonstrate safety and effectiveness for the desired indications could harm the development of that product candidate and other product candidates. This failure could cause us to abandon a product candidate and could delay development of other product candidates. Any delay in, or termination of, our clinical trials would delay the filing of our NDAs with the FDA and, ultimately, our ability to commercialize our product candidates and generate product revenues. Any change in, or termination of, our clinical trials could materially harm our business, financial condition, and results of operation.
We are subject to extensive and costly government regulation.
Product candidates employing our technology are subject to extensive and rigorous domestic government regulation including regulation by the FDA, the Centers for Medicare and Medicaid Services, other divisions of the United States Department of Health and Human Services, the United States Department of Justice, state and local governments, and their respective foreign equivalents. The FDA regulates the research, development, preclinical and clinical testing, manufacture, safety, effectiveness, record-keeping, reporting, labeling, storage, approval, advertising, promotion, sale, distribution, import, and export of biopharmaceutical products. The FDA regulates small molecule chemical entities as drugs, subject to a New Drug Application, or NDA, under the Federal Food, Drug, and Cosmetic Act. If products employing our technologies are marketed abroad, they will also be subject to extensive regulation by foreign governments, whether or not they have obtained FDA approval for a given product and its uses. Such foreign regulation may be equally or more demanding than corresponding United States regulation.
Government regulation substantially increases the cost and risk of researching, developing, manufacturing, and selling our products. The regulatory review and approval process, which includes preclinical testing and clinical trials of each product candidate, is lengthy, expensive, and uncertain. We or our collaborators must obtain and maintain regulatory authorization to conduct clinical trials. We or our collaborators must obtain regulatory approval for each product we intend to market, and the manufacturing facilities used for the products must be inspected and meet legal requirements. Securing regulatory approval requires the submission of extensive preclinical and clinical data and other supporting information for each proposed therapeutic indication in order to establish the product's safety and efficacy, and in the case of biologics also potency and purity, for each intended use. The development and approval process takes many years, requires substantial resources, and may never lead to the approval of a product.
Even if we are able to obtain regulatory approval for a particular product, the approval may limit the indicated medical uses for the product, may otherwise limit our ability to promote, sell, and distribute the product, may require that we conduct costly post-marketing surveillance, and/or may require that we conduct ongoing post-marketing studies. Material changes to an approved product, such as, for example, manufacturing changes or revised labeling, may require further regulatory review and approval. Once obtained, any approvals may be withdrawn, including, for example, if there is a later discovery of previously unknown problems with the product, such as a previously unknown safety issue.
If we, our collaborators, or our contract manufacturers fail to comply with applicable regulatory requirements at any stage during the regulatory process, such noncompliance could result in, among other things delays in the approval of applications or supplements to approved applications; refusal of a regulatory authority, including the FDA, to review pending market approval applications or supplements to approved applications; warning letters; fines; import and/or export restrictions; product recalls or seizures; injunctions; total or partial suspension of production; civil penalties; withdrawals of previously approved marketing applications or licenses; recommendations by the FDA or other regulatory authorities against governmental contracts; and/or criminal prosecutions.
| 22 |
We do not have, and may never obtain, the regulatory approvals we need to market our product candidates.
Following completion of clinical trials, the results are evaluated and, depending on the outcome, submitted to the FDA in the form of an NDA in order to obtain FDA approval of the product and authorization to commence commercial marketing. In responding to an NDA, the FDA may require additional testing or information, may require that the product labeling be modified, may impose post-approval study or reporting requirements or other restrictions on product distribution, or may deny the application. The FDA has established performance goals for review of NDAs - six months for priority applications and ten months for standard applications. However, the FDA is not required to complete its review within these time periods. The timing of final FDA review and action varies greatly, but can take years in some cases and may involve the input of an FDA advisory committee of outside experts. Product sales in the United States may commence only when an NDA is approved.
To date, we have not applied for or received the regulatory approvals required for the commercial sale of any of our products in the United States or in any foreign jurisdiction. None of our product candidates has been determined to be safe and effective, and we have not submitted an NDA to the FDA or an equivalent application to any foreign regulatory authorities for any of our product candidates.
It is possible that none of our product candidates will be approved for marketing. Failure to obtain regulatory approvals, or delays in obtaining regulatory approvals, may adversely affect the successful commercialization of any drugs or biologics that we or our partners develop, may impose additional costs on us or our collaborators, may diminish any competitive advantages that we or our partners may attain, and/or may adversely affect our receipt of revenues or royalties.
Even if approved, our products will be subject to extensive post-approval regulation.
Once a product is approved, numerous post-approval requirements apply. Among other things, the holder of an approved NDA is subject to periodic and other FDA monitoring and reporting obligations, including obligations to monitor and report adverse events and instances of the failure of a product to meet the specifications in the NDA. Application holders must submit new or supplemental applications and obtain FDA approval for certain changes to the approved product, product labeling, or manufacturing process. Application holders must also submit advertising and other promotional material to the FDA and report on ongoing clinical trials.
Depending on the circumstances, failure to meet these post-approval requirements can result in criminal prosecution, fines, injunctions, recall or seizure of products, total or partial suspension of production, denial or withdrawal of pre-marketing product approvals, or refusal to allow us to enter into supply contracts, including government contracts. In addition, even if we comply with FDA and other requirements, new information regarding the safety or effectiveness of a product could lead the FDA to modify or withdraw product approval.
Even if we obtain regulatory approval to market our product candidates, our product candidates may not be accepted by the market.
Even if the FDA approves one or more of our product candidates, physicians and patients may not accept it or use it. Even if physicians and patients would like to use our products, our products may not gain market acceptance among healthcare payors such as managed care formularies, insurance companies or government programs such as Medicare or Medicaid. Acceptance and use of our products will depend upon a number of factors including: perceptions by members of the health care community, including physicians, about the safety and effectiveness of our drug or device product; cost-effectiveness of our product relative to competing products; availability of reimbursement for our product from government or other healthcare payers; and effectiveness of marketing and distribution efforts by us and our licensees and distributors, if any.
The degree of market acceptance of any pharmaceutical product that we develop will depend on a number of factors, including:
| 23 |
| · | cost-effectiveness; |
| · | the safety and effectiveness of our products, including any significant potential side effects (including drowsiness and dry mouth), as compared to alternative products or treatment methods; | |
| · | the timing of market entry as compared to competitive products; | |
| · | flat or declining use of off-label muscle-relaxant products for fibromyalgia prior to the launch of TNX-102 SL; | |
| · | the rate of adoption of our products by doctors and nurses; | |
| · | product labeling or product insert required by the FDA for each of our products; | |
| · | reimbursement policies of government and third-party payors; | |
| · | effectiveness of our sales, marketing and distribution capabilities and the effectiveness of such capabilities of our collaborative partners, if any; and | |
| · | unfavorable publicity concerning our products or any similar products. |
Our product candidates, if successfully developed, will compete with a number of products manufactured and marketed by major pharmaceutical companies, biotechnology companies and manufacturers of generic drugs. Our products may also compete with new products currently under development by others. Physicians, patients, third-party payors and the medical community may not accept and utilize any of our product candidates. If our products do not achieve market acceptance, we will not be able to generate significant revenues or become profitable.
Because we expect sales of our current product candidates, if approved, to generate substantially all of our product revenues for the foreseeable future, the failure of these products to find market acceptance would harm our business and could require us to seek additional financing.
If we fail to establish marketing, sales and distribution capabilities, or fail to enter into arrangements with third parties, we will not be able to create a market for our product candidates.
Our strategy with our lead product candidates is to control, directly or through contracted third parties, all or most aspects of the product development process, including marketing, sales and distribution. Currently, we do not have any sales, marketing or distribution capabilities. In order to generate sales of any product candidates that receive regulatory approval, we must either acquire or develop an internal marketing and sales force with technical expertise and with supporting distribution capabilities or make arrangements with third parties to perform these services for us. The acquisition or development of a sales and distribution infrastructure would require substantial resources, which may divert the attention of our management and key personnel and defer our product development efforts. To the extent that we enter into marketing and sales arrangements with other companies, our revenues will depend on the efforts of others. These efforts may not be successful. If we fail to develop sales, marketing and distribution channels, or enter into arrangements with third parties, we will experience delays in product sales and incur increased costs.
Sales of pharmaceutical products largely depend on the reimbursement of patients' medical expenses by government health care programs and private health insurers. Without the financial support of the government or third-party payors, the market for our products will be limited. These third-party payors are increasingly challenging the price and examining the cost effectiveness of medical products and services. Recent proposals to change the health care system in the United States have included measures that would limit or eliminate payments for medical products and services or subject the pricing of medical treatment products to government control. Significant uncertainty exists as to the reimbursement status of newly approved health care products. Third-party payors may not reimburse sales of our products or enable our collaborators to sell them at profitable prices.
Our business strategy might involve out-licensing product candidates to or collaborating with larger firms with experience in marketing and selling pharmaceutical products. There can be no assurance that we will be able to successfully establish marketing, sales, or distribution relationships; that such relationships, if established, will be successful; or that we will be successful in gaining market acceptance for our products. To the extent that we enter into any marketing, sales, or distribution arrangements with third parties, our product revenues will be lower than if we marketed and sold our products directly, and any revenues we receive will depend upon the efforts of such third-parties. If we are unable to establish such third-party sales and marketing relationships, or choose not to do so, we will have to establish and rely on our own in-house capabilities.
| 24 |
We, as a company, have no experience in marketing or selling pharmaceutical products and currently have no sales, marketing, or distribution infrastructure. To market any of our products directly, we would need to develop a marketing, sales, and distribution force that both has technical expertise and the ability to support a distribution capability. The establishment of a marketing, sales, and distribution capability would significantly increase our costs, possibly requiring substantial additional capital. In addition, there is intense competition for proficient sales and marketing personnel, and we may not be able to attract individuals who have the qualifications necessary to market, sell, and distribute our products. There can be no assurance that we will be able to establish internal marketing, sales, or distribution capabilities. If we are unable to, or choose not to establish these capabilities, or if the capabilities we establish are not sufficient to meet our needs, we will be required to establish collaborative marketing, sales, or distribution relationships with third parties.
In the event that we are successful in bringing any products to market, our revenues may be adversely affected if we fail to obtain acceptable prices or adequate reimbursement for our products from third-party payors.
Our ability to commercialize pharmaceutical products successfully may depend in part on the availability of reimbursement for our products from:
| · | government and health administration authorities; | |
| · | private health insurers; and | |
| · | other third party payors, including Medicare. |
We cannot predict the availability of reimbursement for health care products to be approved in the future. Third-party payors, including Medicare, are challenging the prices charged for medical products and services. Government and other third-party payors increasingly are limiting both coverage and the level of reimbursement for new drugs. Third-party insurance coverage may not be available to patients for any of our products.
The continuing efforts of government and third-party payors to contain or reduce the costs of health care may limit our commercial opportunity. If government and other third-party payors do not provide adequate coverage and reimbursement for any prescription product we bring to market, doctors may not prescribe them or patients may ask to have their physicians prescribe competing drugs with more favorable reimbursement. In some foreign markets, pricing and profitability of prescription pharmaceuticals are subject to government control. In the United States, we expect that there will continue to be federal and state proposals for similar controls. In addition, we expect that increasing emphasis on managed care in the United States will continue to put pressure on the pricing of pharmaceutical products. Cost control initiatives could decrease the price that we receive for any products in the future. Further, cost control initiatives could impair our ability to commercialize our products and our ability to earn revenues from this commercialization.
If we obtain approval to commercialize any approved products outside of the United States, a variety of risks associated with international operations could materially adversely affect our business.
If TNX-102 SL or any of our other product candidates are approved for commercialization outside of the United States, we intend to enter into agreements with third parties to market them on a worldwide basis or in more limited geographical regions. We expect that we will be subject to additional risks related to entering into international business relationships, including:
| · | different regulatory requirements for drug approvals; | |
| · | reduced protection for intellectual property rights, including trade secret and patent rights; | |
| · | unexpected changes in tariffs, trade barriers and regulatory requirements; | |
| · | economic weakness, including inflation, or political instability in particular foreign economies and markets; | |
| · | compliance with tax, employment, immigration and labor laws for employees living or traveling abroad; | |
| · | foreign taxes, including withholding of payroll taxes; | |
| · | foreign currency fluctuations, which could result in increased operating expenses and reduced revenues, and other obligations incident to doing business in another country; |
| 25 |
| · | workforce uncertainty in countries where labor unrest is more common than in the United States; | |
| · | production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; | |
| · | business interruptions resulting from geopolitical actions, including war and terrorism, or natural disasters including earthquakes, hurricanes, floods and fires; and | |
| · | difficulty in importing and exporting clinical trial materials and study samples. |
We face the risk of product liability claims and may not be able to obtain insurance.
Our business exposes us to the risk of product liability claims that are inherent in the development of drugs. If the use of one or more of our or our collaborators' drugs harms people, we may be subject to costly and damaging product liability claims brought against us by clinical trial participants, consumers, health care providers, pharmaceutical companies or others selling our products. Our inability to obtain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of pharmaceutical products we develop, alone or with collaborators. While we currently carry clinical trial insurance and product liability insurance, we cannot predict all of the possible harms or side effects that may result and, therefore, the amount of insurance coverage we hold now or in the future may not be adequate to cover all liabilities we might incur. We intend to expand our insurance coverage to include the sale of commercial products if we obtain marketing approval for our drug candidates in development, but we may be unable to obtain commercially reasonable product liability insurance for any products approved for marketing. If we are unable to obtain insurance at an acceptable cost or otherwise protect against potential product liability claims, we will be exposed to significant liabilities, which may materially and adversely affect our business and financial position. If we are sued for any injury allegedly caused by our or our collaborators' products, our liability could exceed our total assets and our ability to pay the liability. A product liability claim or series of claims brought against us would decrease our cash and could cause our stock price to fall.
We use hazardous chemicals in our business. Potential claims relating to improper handling, storage or disposal of these chemicals could affect us and be time consuming and costly.
Our research and development processes and/or those of our third party contractors may involve the controlled use of hazardous materials and chemicals. These hazardous chemicals are reagents and solvents typically found in a chemistry laboratory. Our operations also produce hazardous waste products. Federal, state and local laws and regulations govern the use, manufacture, storage, handling and disposal of hazardous materials. While we attempt to comply with all environmental laws and regulations, including those relating to the outsourcing of the disposal of all hazardous chemicals and waste products, we cannot eliminate the risk of contamination from or discharge of hazardous materials and any resultant injury. In the event of such an accident, we could be held liable for any resulting damages and any liability could materially adversely affect our business, financial condition and results of operations.
Compliance with environmental laws and regulations may be expensive. Current or future environmental regulations may impair our research, development or production efforts. We might have to pay civil damages in the event of an improper or unauthorized release of, or exposure of individuals to, hazardous materials. We are not insured against these environmental risks.
If we enter into collaborations with third parties, they might also work with hazardous materials in connection with our collaborations. We may agree to indemnify our collaborators in some circumstances against damages and other liabilities arising out of development activities or products produced in connection with these collaborations.
In addition, the federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of hazardous or radioactive materials and waste products may require us to incur substantial compliance costs that could materially adversely affect our business, financial condition and results of operations.
| 26 |
Our insurance policies are expensive and protect us only from some business risks, which will leave us exposed to significant uninsured liabilities.
We carry insurance for most categories of risk that our business may encounter, however, we may not have adequate levels of coverage. We currently maintain general liability, property, workers’ compensation, products liability and directors’ and officers’ insurance, along with an umbrella policy, which collectively costs approximately $100,000 per annum. We cannot provide any assurances that we will be able to maintain existing insurance at current or adequate levels of coverage. Any significant uninsured liability may require us to pay substantial amounts, which would adversely affect our cash position and results of operations.
If we retain collaborative partners and our partners do not satisfy their obligations, we will be unable to develop our partnered product candidates.
In the event we enter into any collaborative agreements, we may not have day-to-day control over the activities of our collaborative partners with respect to any of these product candidates. Any collaborative partner may not fulfill its obligations under these agreements. If a collaborative partner fails to fulfill its obligations under an agreement with us, we may be unable to assume the development of the products covered by that agreement or enter into alternative arrangements with a third party. In addition, we may encounter delays in the commercialization of the product candidate that is the subject of the agreement. Accordingly, our ability to receive any revenue from the product candidates covered by these agreements will be dependent on the efforts of our collaborative partner. We could also become involved in disputes with a collaborative partner, which could lead to delays in or termination of our development and commercialization programs and time-consuming and expensive litigation or arbitration. In addition, any such dispute could diminish our collaborators’ commitment to us and reduce the resources they devote to developing and commercializing our products. Conflicts or disputes with our collaborators, and competition from them, could harm our relationships with our other collaborators, restrict our ability to enter future collaboration agreements and delay the research, development or commercialization of our product candidates. If any collaborative partner terminates or breaches its agreement, or otherwise fails to complete its obligations in a timely manner, our chances of successfully developing or commercializing these product candidates would be materially and adversely affected. We may not be able to enter into collaborative agreements with partners on terms favorable to us, or at all. Our inability to enter into collaborative arrangements with collaborative partners, or our failure to maintain such arrangements, would limit the number of product candidates that we could develop and ultimately, decrease our sources of any future revenues.
RISKS RELATED TO OUR STOCK
There has been a limited trading market for our Common Stock and almost no market activity to date.
Currently, our Common Stock is available for quotation on the OTCQB under the symbol “TNXP.” However, prior to February 2012, there was no trading activity in our Common Stock and limited trading has occurred to date. As of December 31, 2012, trading occurred on only 82 out of 229 possible trading days, with an average of less than 160 shares per possible trading day and less than 445 shares trades on each day when shares actually traded. It is anticipated that there will continue to be a limited trading market for the Common Stock on the OTCQB. We have applied to have our common stock listed on The NASDAQ Capital Market and trading is expected to start upon the effectiveness of this registration statement. Although we believe that this offering and the NASDAQ listing will improve the liquidity of our common stock, this offering may not improve trading volume, reduce volatility or stabilize our share price. A lack of an active market may impair your ability to sell your shares at the time you wish to sell them or at a price that you consider reasonable. The lack of an active market may also reduce the fair market value of your shares. An inactive market may also impair our ability to raise capital by selling shares of capital stock and may impair our ability to acquire other companies or technologies by using Common Stock as consideration.
Our failure to meet the continued listing requirements of The NASDAQ Capital Market could result in a de-listing of our common stock.
If after listing we fail to satisfy the continued listing requirements of The NASDAQ Capital Market, such as the corporate governance requirements or the minimum closing bid price requirement, NASDAQ may take steps to de-list our common stock. Such a de-listing would likely have a negative effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so. In the event of a de-listing, we would take actions to restore our compliance with NASDAQ’s listing requirements, but we can provide no assurance that any such action taken by us would allow our common stock to become listed again, stabilize the market price or improve the liquidity of our common stock, prevent our common stock from dropping below the NASDAQ minimum bid price requirement or prevent future non-compliance with NASDAQ’s listing requirements.
| 27 |
You may have difficulty trading and obtaining quotations for our Common Stock.
Our Common Stock may not be actively traded, and the bid and asked prices for our Common Stock on the OTCQB may fluctuate widely. As a result, investors may find it difficult to dispose of, or to obtain accurate quotations of the price of, our securities. This severely limits the liquidity of the Common Stock, and would likely reduce the market price of our Common Stock and hamper our ability to raise additional capital.
The market price for our Common Stock may be volatile, and your investment in our Common Stock could decline in value.
The stock market in general has experienced extreme price and volume fluctuations. The market prices of the securities of biotechnology and specialty pharmaceutical companies, particularly companies like ours without product revenues and earnings, have been highly volatile and may continue to be highly volatile in the future. This volatility has often been unrelated to the operating performance of particular companies. The following factors, in addition to other risk factors described in this section, may have a significant impact on the market price of our common stock:
| · | announcements of technological innovations or new products by us or our competitors; | |
| · | announcement of FDA approval or disapproval of our products or other product-related actions; | |
| · | developments involving our discovery efforts and clinical trials; | |
| · | developments or disputes concerning patents or proprietary rights, including announcements of infringement, interference or other litigation against us or our potential licensees; | |
| · | developments involving our efforts to commercialize our products, including developments impacting the timing of commercialization; | |
| · | announcements concerning our competitors, or the biotechnology, pharmaceutical or drug delivery industry in general; | |
| · | public concerns as to the safety or efficacy of our products or our competitors’ products; | |
| · | changes in government regulation of the pharmaceutical or medical industry; | |
| · | changes in the reimbursement policies of third party insurance companies or government agencies; | |
| · | actual or anticipated fluctuations in our operating results; | |
| · | changes in financial estimates or recommendations by securities analysts; | |
| · | developments involving corporate collaborators, if any; | |
| · | changes in accounting principles; and | |
| · | the loss of any of our key scientific or management personnel. |
In the past, securities class action litigation has often been brought against companies that experience volatility in the market price of their securities. Whether or not meritorious, litigation brought against us could result in substantial costs and a diversion of management’s attention and resources, which could adversely affect our business, operating results and financial condition.
Investor relations activities, nominal “float” and supply and demand factors may affect the price of our stock.
We utilize various techniques such as non-deal road shows and investor relations campaigns in order to create investor awareness for our company. These campaigns may include personal, video and telephone conferences with investors and prospective investors in which our business practices are described. We provide compensation to investor relations firms and may pay for newsletters, websites, mailings and email campaigns that are produced by third-parties based upon publicly-available information concerning our company. We will not be responsible for the content of analyst reports and other writings and communications by investor relations firms not authored by us or from publicly available information. We do not intend to review or approve the content of such analysts’ reports or other materials based upon analysts’ own research or methods. Investor relations firms should generally disclose when they are compensated for their efforts, but whether such disclosure is made or complete is not under our control. In addition, investors in our company may be willing, from time to time, to encourage investor awareness through similar activities. Investor awareness activities may also be suspended or discontinued which may impact the trading market our common stock.
| 28 |
The SEC and FINRA enforce various statutes and regulations intended to prevent manipulative or deceptive devices in connection with the purchase or sale of any security and carefully scrutinize trading patterns and company news and other communications for false or misleading information, particularly in cases where the hallmarks of “pump and dump” activities may exist, such as rapid share price increases or decreases. We, and our shareholders may be subjected to enhanced regulatory scrutiny due to the small number of holders who initially will own the registered shares of our common stock publicly available for resale, and the limited trading markets in which such shares may be offered or sold which have often been associated with improper activities concerning penny-stocks, such as the OTC Bulletin Board or the OTCQB Marketplace (Pink OTC) or pink sheets. Until such time as our restricted shares are registered or available for resale under Rule 144, there will continue to be a small percentage of shares held by a small number of investors, many of whom acquired such shares in privately negotiated purchase and sale transactions that will constitute the entire available trading market. The Supreme Court has stated that manipulative action is a term of art connoting intentional or willful conduct designed to deceive or defraud investors by controlling or artificially affecting the price of securities. Often times, manipulation is associated by regulators with forces that upset the supply and demand factors that would normally determine trading prices. Since a small percentage of the outstanding common stock of our company will initially be available for trading, held by a small number of individuals or entities, the supply of our common stock for sale will be extremely limited for an indeterminate amount of time, which could result in higher bids, asks or sales prices than would otherwise exist. Securities regulators have often cited thinly-traded markets, small numbers of holders, and awareness campaigns as components of their claims of price manipulation and other violations of law when combined with manipulative trading, such as wash sales, matched orders or other manipulative trading timed to coincide with false or touting press releases. There can be no assurance that our or third-parties’ activities, or the small number of potential sellers or small percentage of stock in the “float,” or determinations by purchasers or holders as to when or under what circumstances or at what prices they may be willing to buy or sell stock will not artificially impact (or would be claimed by regulators to have affected) the normal supply and demand factors that determine the price of the stock.
We do not anticipate paying dividends on our common stock and, accordingly, shareholders must rely on stock appreciation for any return on their investment.
We have never declared or paid cash dividends on our common stock and do not expect to do so in the foreseeable future. The declaration of dividends is subject to the discretion of our board of directors and will depend on various factors, including our operating results, financial condition, future prospects and any other factors deemed relevant by our board of directors. You should not rely on an investment in our company if you require dividend income from your investment in our company. The success of your investment will likely depend entirely upon any future appreciation of the market price of our common stock, which is uncertain and unpredictable. There is no guarantee that our common stock will appreciate in value.
We expect that our quarterly results of operations will fluctuate, and this fluctuation could cause our stock price to decline.
Our quarterly operating results are likely to fluctuate in the future. These fluctuations could cause our stock price to decline. The nature of our business involves variable factors, such as the timing of the research, development and regulatory pathways of our product candidates, which could cause our operating results to fluctuate.
Due to the possibility of fluctuations in our revenues and expenses, we believe that quarter-to-quarter comparisons of our operating results are not a good indication of our future performance.
| 29 |
A sale or perceived sale of a substantial number of shares of our common stock may cause the price of our common stock to decline.
All of our executive officers and directors and certain of our shareholders have agreed not to sell shares of our common stock for a period of 90 days following this offering, subject to extension under specified circumstances. See “Underwriting.” Common stock subject to these lock-up agreements will become eligible for sale in the public market upon expiration of these lock-up agreements, subject to limitations imposed by Rule 144 under the Securities Act of 1933, as amended. If our shareholders sell substantial amounts of our common stock in the public market, the market price of our common stock could fall. Moreover, the perceived risk of this potential dilution could cause shareholders to attempt to sell their shares and investors to short our common stock. These sales also may make it more difficult for us to sell equity or equity-related securities in the future at a time and price that we deem reasonable or appropriate. Shareholders who were issued shares of common stock pursuant to the Exchange Agreement will be able to sell their shares pursuant to Rule 144 under the Securities Act of 1933, as amended, beginning one year after the shareholders acquired their shares, subject to limitations imposed by the lock-up agreements.
Because certain of our shareholders control a significant number of shares of our common stock, they may have effective control over actions requiring shareholder approval.
Our executive officers, directors and principal shareholders, in the aggregate, beneficially own or control the votes of approximately 47.7% of our outstanding Common Stock. Following this offering, our executive officers, directors and principal shareholders, and their respective affiliates, will beneficially own approximately [ * ]% of our outstanding shares of common stock. As a result, these shareholders, acting together, will have the ability to control substantially all matters submitted to our shareholders for approval, including:
| · | removal of any of our directors; |
| · | amendment of our certificate of incorporation or bylaws; and |
| · | adoption of measures that could delay or prevent a change in control or impede a merger, takeover or other business combination involving us. |
As a result of their ownership and positions, our directors, executive officers and principal shareholders collectively are able to influence all matters requiring shareholder approval, including the election of directors and approval of significant corporate transactions. Management’s stock ownership may discourage a potential acquirer from making a tender offer or otherwise attempting to obtain control of us, which in turn could reduce our stock price or prevent our shareholders from realizing a premium over our stock price.
Our common stock is not currently traded at high volume, and you may be unable to sell at or near ask prices or at all if you need to sell or liquidate a substantial number of shares at one time.
Our common stock is currently traded, but with very low, if any, volume, based on quotations on the OTCQB, meaning that the number of persons interested in purchasing our common stock at or near bid prices at any given time may be relatively small or non-existent. This situation is attributable to a number of factors, including the fact that we are a small company which is still relatively unknown to stock analysts, stock brokers, institutional investors and others in the investment community that generate or influence sales volume, and that even if we came to the attention of such persons, they tend to be risk-averse and would be reluctant to follow an unproven company such as ours or purchase or recommend the purchase of our shares until such time as we became more seasoned and viable. As a consequence, there may be periods of several days or more when trading activity in our shares is minimal or non-existent, as compared to a seasoned issuer which has a large and steady volume of trading activity that will generally support continuous sales without an adverse effect on share price. We cannot give you any assurance that a broader or more active public trading market for our common stock will develop or be sustained, or that trading levels will be sustained.
| 30 |
Shareholders should be aware that, according to Commission Release No. 34-29093, the market for “penny stocks” has suffered in recent years from patterns of fraud and abuse. Such patterns include (1) control of the market for the security by one or a few broker-dealers that are often related to the promoter or issuer; (2) manipulation of prices through prearranged matching of purchases and sales and false and misleading press releases; (3) boiler room practices involving high-pressure sales tactics and unrealistic price projections by inexperienced sales persons; (4) excessive and undisclosed bid-ask differential and markups by selling broker-dealers; and (5) the wholesale dumping of the same securities by promoters and broker-dealers after prices have been manipulated to a desired level, along with the resulting inevitable collapse of those prices and with consequent investor losses. Our management is aware of the abuses that have occurred historically in the penny stock market. Although we do not expect to be in a position to dictate the behavior of the market or of broker-dealers who participate in the market, management will strive within the confines of practical limitations to prevent the described patterns from being established with respect to our securities. The occurrence of these patterns or practices could increase the future volatility of our share price.
Efforts to comply with recently enacted changes in securities laws and regulations will increase our costs and require additional management resources, and we still may fail to comply.
As directed by Section 404 of the Sarbanes-Oxley Act of 2002, the SEC adopted rules requiring public companies to include a report of management on their internal controls over financial reporting in their annual reports on Form 10-K. In addition, in the event we are no longer a smaller reporting company, the independent registered public accounting firm auditing our financial statements would be required to attest to the effectiveness of our internal controls over financial reporting. Such attestation requirement by our independent registered public accounting firm would not be applicable to us until the report for the year ended December 31, 2013 at the earliest, if at all. If we are unable to conclude that we have effective internal controls over financial reporting or if our independent registered public accounting firm is required to, but is unable to provide us with a report as to the effectiveness of our internal controls over financial reporting, investors could lose confidence in the reliability of our financial statements, which could result in a decrease in the value of our securities.
We have implemented a reverse stock split, which has reduced our trading volume and may result in a decrease in our market capitalization.
Effective May 1, 2013, we implemented a one-for-twenty reverse stock split. This reverse stock split was implemented because one of the listing requirements for The NASDAQ Stock Market is for our common stock to trade at or above $4.00 per share for at least 30 of the 60 trading days prior to listing. At our annual meeting of shareholders held on April 19, 2013, the shareholders authorized a reverse stock split. We cannot guarantee that the price increase of our common stock price resulting from the reverse split will:
| · | be proportionate to the reverse split ratio; | |
| · | last in the marketplace for any length of time; | |
| · | remain at a price sufficient to meet the listing requirements of The NASDAQ Stock Market; or | |
| · | be sufficient to facilitate raising capital. |
If our common stock becomes subject to the SEC’s penny stock rules, broker-dealers may experience difficulty in completing customer transactions and trading activity in our securities may be adversely affected.
If at any time our securities are no longer listed on a national securities exchange, including The NASDAQ Stock Market or we have net tangible assets of $5,000,000 or less and our common stock has a market price per share of less than $5.00, transactions in our common stock will be subject to the SEC’s “penny stock” rules. If our common stock becomes subject to the “penny stock” rules promulgated under the Securities Exchange Act of 1934, broker-dealers may find it difficult to effectuate customer transactions and trading activity in our securities may be adversely affected. For any transaction involving a penny stock, unless exempt, the rules require:
| · | that a broker or dealer approve a person’s account for transactions in penny stocks; and | |
| · | the broker or dealer receive from the investor a written agreement to the transaction, setting forth the identity and quantity of the penny stock to be purchased. |
| 31 |
In order to approve a person’s account for transactions in penny stocks, the broker or dealer must:
| · | obtain financial information and investment experience objectives of the person; and | |
| · | make a reasonable determination that the transactions in penny stocks are suitable for that person and the person has sufficient knowledge and experience in financial matters to be capable of evaluating the risks of transactions in penny stocks. |
The broker or dealer must also deliver, prior to any transaction in a penny stock, a disclosure schedule prescribed by the SEC relating to the penny stock market, which, in highlight form:
| · | sets forth the basis on which the broker or dealer made the suitability determination; and | |
| · | that the broker or dealer received a signed, written agreement from the investor prior to the transaction. |
Generally, brokers may be less willing to execute transactions in securities subject to the “penny stock” rules. This may make it more difficult for investors to dispose of our common stock and cause a decline in the market value of our stock.
Disclosure also has to be made about the risks of investing in penny stocks in both public offerings and in secondary trading and about the commissions payable to both the broker-dealer and the registered representative, current quotations for the securities and the rights and remedies available to an investor in cases of fraud in penny stock transactions. Finally, monthly statements have to be sent disclosing recent price information for the penny stock held in the account and information on the limited market in penny stocks.
RISKS RELATING TO THIS OFFERING
Our management will have broad discretion in allocating the net proceeds of this offering, and may use the proceeds in ways in which you disagree.
Our management has significant flexibility in applying the net proceeds we expect to receive in this offering. Because the net proceeds are not required to be allocated to any specific investment or transaction, and therefore you cannot determine at this time the value or propriety of our application of those proceeds, you and other shareholders may not agree with our decisions. In addition, our use of the proceeds from this offering may not yield a significant return or any return at all for our shareholders. The failure by our management to apply these funds effectively could have a material adverse effect on our business, results of operations or financial condition. See “Use of Proceeds” for a further description of how management intends to apply the proceeds from this offering.
You will experience immediate dilution in the book value per share of the common stock you purchase.
Because the public offering price per unit is expected to be substantially higher than the book value per share of our common stock, you will suffer substantial dilution in the net tangible book value of the common stock you purchase in this offering. After giving effect to assumed sale of [ * ] units in this offering at an assumed public offering price of $[ * ] per unit (the closing bid price of our common stock on May [ * ], 2013), and after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us, and attributing no value to the Series A Warrants, if you purchase units in this offering, you will suffer immediate and substantial dilution of approximately $[ * ] per share in the net tangible book value of the common stock you acquire. In the event that you exercise your Series A Warrants, you will experience additional dilution to the extent that the exercise price of those warrants is higher than the book value per share of our common stock. See “Dilution” below for a more detailed discussion of the dilution you will incur if you purchase securities in this offering.
Future sales and issuances of our common stock or rights to purchase common stock, including pursuant to our equity incentive plan, could result in additional dilution of the percentage ownership of our shareholders and could cause our stock price to fall.
We expect that significant additional capital will be needed in the future to continue our planned operations. To the extent we raise additional capital by issuing equity securities, our shareholders may experience substantial dilution. We may sell common stock, convertible securities or other equity securities. If we sell common stock, convertible securities or other equity securities, your investment in our common stock will be diluted. These sales may also result in material dilution to our existing shareholders, and new investors could gain rights superior to our existing shareholders.
| 32 |
There is no public market for the Series A Warrants being sold in this offering.
There is no established public trading market for the Series A Warrants being offered in this offering, and we do not expect a market to develop. We do not intend to apply for listing of any such warrants on any securities exchange or other trading market. Without an active market, the liquidity of the warrants will be limited.
Holders of our Series A Warrants will have no rights as a common shareholder until they acquire our common stock.
Until you acquire shares of our common stock upon exercise of your Series A Warrants, you will have no rights with respect to our common stock. Upon exercise of your warrants, you will be entitled to exercise the rights of a common shareholder only as to matters for which the record date occurs after the exercise date.
USE OF PROCEEDS
We estimate that the net proceeds to us from the sale of the units offered by this prospectus will be approximately $[ * ] million, assuming the sale by us of [ * ] units at an assumed public offering price of $[ * ] per unit (the closing bid price of our common stock on May [ * ], 2013) and after deducting the estimated underwriting discounts and commissions and estimated offering expenses payable by us. This amount does not include the proceeds which we may receive in connection with the exercise of the warrants. We cannot predict when or if the warrants will be exercised, and it is possible that the warrants may expire and never be exercised. We currently intend to use the net proceeds of this offering for the clinical development of TNX-102 SL, for which a Phase 2/3 clinical trial for the treatment of FM is in process to begin in the third quarter of 2013, and for general corporate purposes, including working capital and operational purposes, including pre-clinical development. Pending use of the net proceeds, we intend to invest these net proceeds in interest-bearing, investment-grade securities.
If the net proceeds of this offering are less than $6,000,000, we will be unable to achieve our planned operations for the next 12 months. If that occurs, we will prioritize our clinical trial and development programs, and select programs which our management believes can be adequately funded given the amount of proceeds and that provide the best opportunity for return to shareholders, consistent with the program interests and preferences of investors in this offering.
An $[ * ] increase (decrease) in the assumed public offering price of $[ * ] per unit would increase (decrease) the expected net cash proceeds of the offering to us by approximately $[ * ]. An increase (decrease) in the assumed number of units sold in this offering would increase (decrease) the expected net cash proceeds of the offering to us by approximately $[ * ].
The net proceeds from this offering will not be sufficient to complete clinical trials and other studies required for the approval of any product by the FDA, and we will need significant additional funds in the future. See the sections entitled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operation.”
| 33 |
MARKET FOR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS
Our common stock is currently traded on the OTCQB under the symbol “TNXP.” Prior to July 23, 2012, our common stock was quoted on the Over-the-Counter Bulletin Board under the symbol “TNXP.OB.” Prior to October 19, 2011, our common stock was quoted on the Over-the-Counter Bulletin Board under the symbol “TAEI.OB.” Prior to February 2012, no public trades occurred in our common stock. The following table sets forth, for the periods indicated, the high and low sales prices per share of our common stock as reported by the OTCQB or the Over-the-Counter Bulletin Board, as applicable.
| Fiscal Year 2012 | ||||||||
| High | Low | |||||||
| First Quarter | $ | 41.20 | $ | 40.00 | ||||
| Second Quarter | $ | 40.00 | $ | 16.60 | ||||
| Third Quarter | $ | 20.00 | $ | 14.80 | ||||
| Fourth Quarter | $ | 16.40 | $ | 5.00 | ||||
| Fiscal Year 2013 | ||||||||
| High | Low | |||||||
| First Quarter | $ | 14.60 | $ | 4.80 | ||||
| Second Quarter (1) | $ | 15.00 | $ | 6.00 | ||||
| (1) | As of May 9, 2013. |
Holders
As of May 9, 2013, we had approximately 186 holders of our common stock. The number of record holders was determined from the records of our transfer agent and does not include beneficial owners of common stock whose shares are held in the names of various security brokers, dealers, and registered clearing agencies. The transfer agent of our common stock is vStock Transfer, LLC, 77 Spruce Street, Suite 201, Cedarhurst, NY 11516.
Dividends
We have not declared or paid any cash dividends on our common stock and we do not anticipate paying any cash dividends to shareholders in the foreseeable future. Any future determination to pay cash dividends will be at the discretion of the Board of Directors and will be dependent upon our financial condition, results of operations, capital requirements, and such other factors as the Board of Directors deem relevant.
| 34 |
DILUTION
If you purchase any of the units offered by this prospectus, you will experience dilution to the extent of the difference between the offering price per unit you pay in this offering and the net tangible book value per share of our common stock immediately after this offering, assuming no value is attributed to the Series A Warrants included in the units. Our net tangible book value as of December 31, 2012 was $978,418, or approximately $0.45 per share of common stock. Net tangible book value per share is equal to our total tangible assets minus total liabilities, divided by the number of shares of common stock outstanding.
After giving effect to the assumed sale by us of [ * ] units in this offering at an assumed public offering price of $[ * ] per unit (the closing bid price of our common stock on May [ * ], 2013), assuming no value is attributed to the Series A Warrants included in the units, and after deducting estimated underwriting discounts and commissions and expenses payable by us, our as adjusted net tangible book value as of December 31, 2012 would have been approximately $[ * ] million, or approximately $[ * ] per share of common stock. This represents an immediate increase in net tangible book value of approximately $[ * ] per share to existing shareholders and an immediate dilution of approximately $[ * ] per share to new investors. The following table illustrates this per share dilution:
| Assumed offering price per unit | $ | [ ] | ||||||
| Net tangible book value per share as of December 31, 2012 | $ | 0.45 | ||||||
| Increase per share attributable to new investors | $ | [ ] | ||||||
| As adjusted net tangible book value per share after this offering | $ | [ ] | ||||||
| Dilution per share to new investors | $ | [ ] |
Investors that acquire additional shares of common stock through the exercise of the Series A Warrants offered hereby may experience additional dilution depending on our net tangible book value at the time of exercise.
The information in the table above is based on 2,159,156 shares of our common stock outstanding on December 31, 2012, and does not include:
| • | 1,259,934 shares of common stock issuable upon the exercise of warrants outstanding at December 31, 2012 with a weighted average exercise price of $14.34 per share; |
| • | 150,000 shares of common stock issuable upon the exercise of options outstanding at December 31, 2012 with a weighted average exercise price of $30.00 per share; |
| • | 50,000 shares of common stock reserved for future grants and awards under our equity incentive plan as of December 31, 2012; |
| • | shares of common stock issuable upon the exercise of the Series A Warrants offered hereby; and |
| • | shares of common stock issuable upon the exercise of the underwriter warrants. |
A $[ * ] increase (decrease) in the assumed public offering price of $[ * ] per unit would increase (decrease) our pro forma as adjusted net tangible book value by approximately $[ * ], our as adjusted net tangible book value per share by approximately $[ * ] and dilution per share to new investors by approximately $[ * ], assuming that the number of units offered by us, remains the same. A [ * ] increase (decrease) in the number of units offered by us would increase (decrease) our pro forma as adjusted net tangible book value by approximately $[ * ], our as adjusted net tangible book value per share by approximately $[ * ] and dilution per share to new investors by approximately $[ * ].
| 35 |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Some of the information in this Form S-1 contains forward-looking statements that involve substantial risks and uncertainties. You can identify these statements by forward-looking words such as “may,” “will,” “expect,” “anticipate,” “believe,” “estimate” and “continue,” or similar words. You should read statements that contain these words carefully because they:
| · | discuss our future expectations; |
| · | contain projections of our future results of operations or of our financial condition; and |
| · | state other “forward-looking” information. |
We believe it is important to communicate our expectations. However, there may be events in the future that we are not able to accurately predict or over which we have no control. Our actual results and the timing of certain events could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including those set forth under “Risk Factors,” “Business” and elsewhere in this prospectus. See “Risk Factors.”
Business Overview
We are a specialty pharmaceutical company focused on developing novel pharmaceutical products for challenging disorders of the CNS. Our drug development programs target CNS conditions that manifest as central pain. Central pain results from abnormal pain processing in the CNS, rather than from dysfunction in peripheral tissues where pain is perceived. Our lead program targets FM, a central pain syndrome. Another program targets PTSD, in which central pain is a component. Central pain is associated with disturbed sleep, and improvements in pain or sleep can sometimes help the other symptom. Our products for FM and PTSD are administered at bedtime. We believe that the pain associated with FM is partly related to sleep quality. Our potential therapeutic solutions for FM and PTSD are based on a known pharmaceutical agent that has been approved for use in another indication. We are developing proprietary products that are new dose, formulation, and delivery options tailored to such new potential therapeutic uses. We intend to start a Phase 2b pivotal study for FM in the third quarter of 2013, which, if successful, will be the first of two pivotal studies required for marketing approval in the U.S.
We are currently devoting the majority of our efforts to the development of our lead product candidate, TNX-102 sublingual tablet, or TNX-102 SL. TNX-102 SL is a novel dose and formulation of CBP, the active pharmaceutical ingredient of two widely prescribed muscle relaxant products, Flexeril and Amrix. TNX-102 SL is distinct from these products as it is being developed at a dose level significantly below the lowest marketed doses of Flexeril and Amrix. TNX-102 SL is also distinct from these products with regard to its route of administration, as it is designed to be placed under the tongue, to disintegrate, dissolve and provide sublingual absorption, whereas Flexeril and Amrix are designed to be swallowed and to provide absorption in the small intestine. TNX-102 SL is also intended for chronic use, whereas Flexeril and Amrix are marketed for two to three weeks of use. We are currently developing TNX-102 SL for the treatment of FM under an IND and under three CTAs filed in Canada. We are also developing TNX-102 SL for the treatment of PTSD for which we held a pre-IND meeting in October 2012. We expect that any applications we submit for FDA approval of TNX-102 SL will be submitted under Section 505(b)(2) of the FDCA, which we believe will allow for a shorter timeline of clinical development as compared to that needed to fulfill the requirements of Section 505(b)(1), under which NCEs are generally reviewed.
TNX-102 SL is a small, rapidly disintegrating tablet containing CBP for sublingual administration at bedtime. We designed TNX-102 SL to enable the efficient delivery of CBP to the systemic circulation via sublingual transmucosal absorption and to avoid first-pass liver metabolism. We also designed TNX-102 SL to provide CBP at doses lower than those currently available. We have conducted several clinical and pre-clinical pharmacokinetic studies of TNX-102 SL which we believe support its development as a novel therapeutic product for FM and PTSD, and which demonstrate a number of potentially advantageous characteristics as compared to current CBP-containing products, none of which are approved for these indications. Based on our Phase 1 comparative study, we have observed that, as compared to oral CBP tablets, TNX-102 SL results in faster systemic absorption and significantly higher plasma levels of CBP in the first hour following administration. TNX-102 SL was generally well-tolerated, with no serious adverse events reported in this study. Some subjects experienced transient numbness on the tongue after TNX-102 SL administration, and other side-effects reported were similar to those associated with current CBP products.
| 36 |
We also have a pipeline of other product candidates, including TNX-201 and TNX-301. TNX-201 is based on isometheptene mucate and is under development as a treatment for certain types of headaches. TNX-201 is a purified isomer of isometheptene mucate, which has been marketed and approved as a mixture of two isomers. TNX-301 is a fixed dose combination of two FDA-approved drugs, disulfiram and selegiline and is under development as a treatment for alcohol abuse and dependence. For competitive reasons, we do not disclose the identities of the active ingredients or targeted indications in our pipeline until a U.S. patent has been allowed or issued. Consistent with our mission, these product candidates are or likely will be reformulations of active ingredients that have been used in humans in other products and that are designed for new CNS therapeutic indications.
In other cases, the products will be formulated to match predicate products closely enough to be considered generic copies or similarly enough to other marketed products to rely (in part) on their regulatory review and approval, as well as available published data. The predicate product may be approved by the FDA under an NDA or may have been reviewed for safety and effectiveness by the National Academy of Sciences under the DESI program, in which case they would be considered by FDA to be “unapproved products”. For DESI products, it is our intent to develop NDA versions to meet cGMP and the ICH requirements to seek approval under the 505(b)(2) regulatory pathway.
Because of our size and being in the development stage, we do not currently devote a significant amount of time or resources towards our other pipeline candidates. We anticipate that sometime in 2013 we will begin developing formulations for TNX-201 and possibly TNX-301, but do not expect to start clinical trials until 2014 at the earliest.
On October 7, 2011, we executed and consummated the Share Exchange Agreement with Tonix Sub. Pursuant to the Share Exchange, each share of Tonix Sub’s common stock was exchanged for 0.045 shares of our common stock, and each share of Tonix Sub’s Series A and B preferred stock was exchanged for 0.24 shares of our common stock. Upon completion of the Share Exchange, the Tonix Sub shareholders, including holders of 1,396,982 restricted shares, which were subject to accelerated vesting, received in exchange for all of their shares, an aggregate of 1,133,334 shares of our common stock and our existing shareholders retained 200,000 shares of common stock. The 1,133,334 shares issued to the Tonix Sub shareholders constituted approximately 85% of our 1,333,334 shares of common stock issued and outstanding after the Share Exchange. Upon completion of the Share Exchange, Tonix Sub became our wholly-owned subsidiary. For accounting purposes, the acquisition has been treated as a recapitalization of Tonix Sub, accompanied by the issuance of our common stock for the outstanding common stock of Toxic Sub, which was recorded at a nominal value. The historical financial statements are those of Tonix Sub. The accompanying financial statements give retroactive effect to the recapitalization as if it had occurred on June 7, 2007 (inception date). Also, professional services expenses were allocated to research and development and general and administrative expenses in the 2010 and cumulative from inception through December 31, 2011 statement of operations to be consistent with the current period’s presentation.
Current Operating Trends
Our current research and development efforts are focused on developing our lead product, TNX-102 SL, but we also expend some effort on our earlier pipeline programs. Our research and development expenses consist of manufacturing work and the cost of drug ingredients used in such work, fees paid to providers for conducting various clinical studies as well as for the analysis of the results of such studies, and for other medical research addressing the potential efficacy of our drugs. We believe that significant investment in product development is a competitive necessity, and we plan to continue these investments in order to be in a position to realize the potential of our product candidates and proprietary technologies.
| 37 |
We plan to start the next phase of clinical development for TNX-102 SL over the next six months, subject to raising necessary funds. Clinical trials can be very expensive. If these and additional necessary clinical trials are successful, we plan to prepare and submit applications to the FDA for marketing approval for our drug candidates. This process entails significant costs. As a result of these and other factors, we expect our research and development expenses to increase significantly over the next 12 to 24 months.
We expect that a larger percentage of our research and development expenses in the future will be incurred in support of our current and future preclinical and clinical development programs rather than technology development. These expenditures are subject to numerous uncertainties relating to timing and cost to completion. We test compounds in numerous preclinical studies for safety, toxicology and efficacy. At the appropriate time, subject to the approval of regulatory authorities, we expect to conduct early-stage clinical trials for each drug candidate. We anticipate funding these trials ourselves, and possibly with the assistance of federal grants. As we obtain results from trials, we may elect to discontinue or delay clinical trials for certain products in order to focus our resources on more promising products. Completion of clinical trials may take several years, and the length of time generally varies substantially according to the type, complexity, novelty and intended use of a product candidate.
The commencement and completion of clinical trials for our products may be delayed by many factors, including lack of efficacy during clinical trials, unforeseen safety issues, slower than expected patient recruitment, or government delays. In addition, we may encounter regulatory delays or rejections as a result of many factors, including results that do not support the intended safety or efficacy of our product candidates, perceived defects in the design of clinical trials and changes in regulatory policy during the period of product development. As a result of these risks and uncertainties, we are unable to accurately estimate the specific timing and costs of our clinical development programs or the timing of material cash inflows, if any, from our product candidates. Our business, financial condition and results of operations may be materially adversely affected by any delays in, or termination of, our clinical trials or a determination by the FDA that the results of our trials are inadequate to justify regulatory approval, insofar as cash in-flows from the relevant drug or program would be delayed or would not occur.
Results of Operations
We anticipate that our results of operations will fluctuate for the foreseeable future due to several factors, such as the progress of our research and development efforts and the timing and outcome of regulatory submissions. Due to these uncertainties, accurate predictions of future operations are difficult or impossible to make.
Fiscal year Ended December 31, 2012 Compared to Fiscal year Ended December 31, 2011
Revenues and Cost of Goods Sold. We had no revenues or cost of goods sold during the fiscal years ended December 31, 2012 and 2011.
Research and Development Expenses. Research and development expenses for the fiscal year ended December 31, 2012 were $2,583,308, an increase of $1,425,141, or 123%, from $1,158,167 for the fiscal year ended December 31, 2011. This increase is primarily due to increased development work related to TNX-102 SL, including formulation development, manufacturing, human and animal pharmacokinetic studies, and market research. In 2012, we incurred $552,953, $836,278 and $468,509 in manufacturing cost, clinical activities and cost, non-clinical activities cost, respectively, as compared to $0, $318,616 and $342,398 in 2011, respectively.
General and Administrative Expenses. General and administrative expenses for the fiscal year ended December 31, 2012 were $4,078,102, an increase of $1,857,741, or 84%, from $2,220,361 incurred in the fiscal year ended December 31, 2011. This increase is primarily due to payroll related expenses and professional services.
Payroll related expenses increased to $1,820,877 in the current year from $731,285 for the fiscal year ended December 31, 2011, an increase of $1,089,592, or 149%. We incurred $865,157 in stock based compensation in connection with the vesting of stock options issued to board members, officers and employees in 2012 as compared to $159,596 in stock based compensation in 2011 relating to the acceleration of vesting in conjunction with our reverse merger in 2011 of restricted stock previously issued to our employees. The increase in cash payroll related costs of $384,032 was a result of the hiring of new employees, cash bonuses to employees, and severance payments to a former employee.
| 38 |
Professional services for the fiscal year ended December 31, 2012 totaled $1,444,455, an increase of $322,908, or 29%, over the $1,121,547 recognized for the fiscal year ended December 31, 2011. Of professional services, legal fees totaled $465,523 for the fiscal year ended December 31, 2012, an increase of $92,448, or 25%, from $373,075 incurred for the fiscal year ended December 31, 2011. Consulting fees totaled $734,520 for the fiscal year ended December 31, 2012, an increase of $435,376 or 146%, from $299,144 for the fiscal year ended December 31, 2011. The increase was primarily a result of $451,619 in public and investor relations costs in the fiscal year ended December 31, 2012 compared to $100,378 in 2011. Accounting fees incurred in the fiscal year ended December 31, 2012 amounted to $244,164, an increase of $1,161, or 0%, from $243,003 incurred in fiscal 2011.
Travel, meals and entertainment costs for the fiscal year ended December 31, 2012 were $108,248, an increase of $38,980, or 56%, from $69,268 incurred in the fiscal year ended December 31, 2011. Travel, meals and entertainment costs include travel related to medical and life sciences conferences, which accounted for the primary increase from 2011. Rent for the fiscal year ended December 31, 2012 totaled $116,732, a decrease of $11,496, or 9%, from $128,228 incurred in fiscal 2011, due primarily to the opening of new office space in New York in late 2011. Market research and analysis for the fiscal year ended December 31, 2012 was $229,546, an increase of $169,757 or 284% from $59,789 incurred in the fiscal year ended December 31, 2011. We continue to research and analyze the potential market for our products. Depreciation expense in fiscal 2012 totaled $14,329, an increase of $5,029, or 54%, over the expense of $9,300 incurred in fiscal 2011, as a result of the purchase of new office computers.
Change in fair value of warrant liability. In connection with our January and March 2012 financing, we issued warrants that contained certain reset provisions. As such, we were required to record the fair value as a liability and mark to market each reporting period. In June 2012, upon the effectiveness of our registration statement, these reset provisions expired. Therefore we adjusted the fair value of the warrants from their initial issuance in January and March 2012, charged operations for the increase in fair value of $1,177,026 and reclassified the fair value of warrants to equity.
Interest Expense. Interest expense for the fiscal year ended December 31, 2012 totaled $1,613,039, an increase of $1,521,454, or 1,661%, from $91,585 incurred during the fiscal year ended December 31, 2011. In the fiscal year ended December 31, 2012, our interest costs were comprised primarily of a beneficial conversion feature related to our issuance of convertible debentures in December 2012 charged to interest of $710,000, $196,166 of deferred financing costs related to the issuance of our secured convertible debentures in October 2011 and December 2012, allocated offering costs of $270,743 charged to interest as part of a financing, and the fair value of $426,152, net with prior period accrual, of common stock and warrants issued to convertible debentures holders in connection with the conversion to a financing. In addition, we incurred interest expense related to our convertible debentures. In 2011, our interest costs were comprised primarily of amortization of deferred financing costs in conjunction with the issuance of our secured convertible debentures in October 2011. We incurred an aggregate of $249,543 in deferred financing costs, of which we amortized $53,377 as interest expense for the fiscal year ended December 31, 2011. In addition, we incurred interest expense related to $500,000 of notes payable and our secured convertible debentures.
Net Loss. As a result of the foregoing, net loss for the year ended December 31, 2012 was $9,449,600, compared to a net loss of $3,470,113 for the year ended December 31, 2011.
Three Months Ended March 31, 2013 Compared to Three Months Ended March 31, 2012
Revenues and Cost of Goods Sold. We had no revenues or cost of goods sold during the three month periods ended March 31, 2013 and 2012.
Research and Development Expenses. Research and development expenses for the three months ended March 31, 2013 were $740,629, an increase of $343,001, or 86%, from $397,628 for the three months ended March 31, 2012. The increase in clinical and non-clinical cost and activities is primarily due to increased development work related to TNX-102 SL, including formulation development, manufacturing, regulatory, clinical development and market research.
| 39 |
General and Administrative Expenses. General and administrative expenses for the three months ended March 31, 2013 were $1,260,596, an increase of $497,859, or 65%, from $762,737 incurred in the three months ended March 31, 2012. This increase is primarily due to an increase in payroll-related expenses, along with increases in investor relations fees, legal professional fees, travel, meals and entertainment expense, and marketing expenses, offset by a decrease in accounting expense.
Payroll-related expenses increased to $561,490 in the current period from $361,739 for the three months ended March 31, 2012, an increase of $199,751, or 55%, primarily related to stock-based compensation, offset by a decrease in cash compensation due to one-time bonuses and severance paid in the three months ended March 31, 2012. Payroll-related expenses include non-cash compensation associated with options granted in 2012 and 2013 of $392,323 for the three months ended March 31, 2013, as compared to $-0- for the same period last year.
Professional services for the three months ended March 31, 2013 totaled $497,698, an increase of $238,582, or 92%, over the $259,116 incurred for the three month period ended March 31, 2012. The increase was primarily a result of $262,978 in investor and public relations in the three months ended March 31, 2013, an increase of $210,349, or 400%, compared to $52,629 in 2012. Accounting and auditing fees incurred in the three months ended March 31, 2013 amounted to $67,769, a slight decrease of $3,780, or 5%, from $71,549 incurred in the three months ended March 31, 2012. Legal fees totaled $138,607 for the three months ended March 31, 2013, an increase of $21,164, or 18%, from $117,443 incurred for the three months ended March 31, 2012. The increase in legal fees is due to legal expenses incurred relating to our patent filing costs. Other professional fees totaled $28,344 for the three months ended March 31, 2013, an increase of $10,849 or 62%, from $17,495 for the three months ended March 31, 2012.
Travel, meals and entertainment costs for three months ended March 31, 2013 were $45,096, an increase of $29,542, or 190%, from $15,554 incurred in the three months ended March 31, 2012. Travel, meals and entertainment costs primarily include travel to contractors and consultants engaged in research and development activities related to TNX-102 as well as travel related to investor relations activities.
Rent for three months ended March 31, 2013 totaled $28,595, a decrease of $1,765, or 6%, from $30,360 incurred in the three months ended March 31, 2012. Depreciation expense in the three months ended March 31, 2013 totaled $4,138, an increase of $1,784, or 76%, over the expense of $2,354 incurred in the three months ended March 31, 2012, as a result of the purchase of new office computers.
Change in fair value of warrant liability. In connection with a financing conducted in the first quarter of 2012, we issued warrants that contained certain reset provisions. As such, we were required to record the fair value as a liability and mark to market each reporting period. On March 31, 2012, we adjusted the fair value of the warrants from their initial issuance in January and March 2012 and credited operations for $47,023 for the decrease in fair value of the issued warrants. In June 2012, upon the effectiveness of our registration statement, these reset provisions expired. Therefore we adjusted the fair value of the warrants from their initial issuance in January and March 2012 and reclassified the fair value of warrants to equity.
Interest and Other Financing Costs. Interest income for the three months ended March 31, 2013 totaled $22, as compared to interest expense of $901,646 incurred during the three months ended March 31, 2012. In 2012, our interest costs were comprised primarily of amortization and write-off of deferred financing costs related to the issuance of our secured convertible debentures in October 2011 of $196,166, allocated offering costs charged to interest as part of our current period financing of $270,743 and the fair value of common stock and warrants issued to convertible debentures holders in connection with the conversion to current period financing of $426,153, net with prior period accrual. In addition, we incurred interest expense related to our convertible debentures during the three months ended March 31, 2012.
Net Loss. As a result of the foregoing, net loss for the three months ended March 31, 2013 was $2,001,203, compared to a net loss of $2,014,988 for the three months ended March 31, 2012, a decrease of $13,785, or 1%.
| 40 |
Liquidity and Capital Resources
As of March 31, 2013, we had a working capital deficit of $725,358, comprised primarily of cash of $423,739 and prepaid expenses of $67,691, which was offset by $870,096 of accounts payable and $346,692 of accrued expenses. A significant portion of the accounts payable and accrued expenses are due to work performed in relation to our anticipated Phase 2b clinical trial of TNX-102 SL in FM, but we do not expect a number of these expenses to be due or payable for several months. For the three months ended March 31, 2013 and 2012, we used $1,361,629 and $1,777,614 of cash in operating activities, respectively, which represent cash outlays for research and development and general and administrative expenses in such periods. Increases in cash outlays principally resulted from manufacturing, pre-clinical and clinical cost and activities, regulatory cost, and payroll. For the three months ended March 31, 2013, we did not have any financing activities. In the comparable 2012 period, $4,387,895 was raised through the sale of shares of common stock and warrants, net with $150,000 repayment of convertible debentures. At March 31, 2013, we had cash of $423,739 compared to $1,785,390 at December 31, 2012. Our cash is held in bank deposit accounts.
Cash used in investing activities for the three months ended March 31, 2013 and 2012 was $22. Both periods reflect interest earned in restricted cash accounts.
Cash used in operations for the year ended December 31, 2012 and 2011 was $5,712,864 and $2,637,578, respectively, which represent cash outlays for research and development and general and administrative expenses in such periods. Increase in cash outlays principally resulted from manufacturing, pre-clinical, and clinical cost and activities, regulatory cost, payroll and rent.
Cash used in investing activities for the year ended December 31, 2012 was $35,763 compared to cash provided by investing activities of $302 in the year ended December 31, 2011. In the year ended December 31, 2012 and 2011, we purchased office furniture and computer equipment of $35,673 and $2,764, respectively.
In their report dated March 8, 2013, our independent registered public accounting firm stated at December 31, 2012, there is substantial doubt about our ability to continue as a going concern. Our ability to continue as a going concern is an issue raised due to our net losses and negative cash flows from operations since inception and our expectation that these conditions will continue for the foreseeable future. In addition, we will require additional financing to fund future operations. Further, we do not have any commercial products available for sale and have not generated revenues and there is no assurance that if approval of our products is received that we will be able to generate cash flow to fund operations. In addition, there can be no assurance that our research and development will be successfully completed or that any product will be approved or commercially viable. Our ability to continue as a going concern is subject to our ability to obtain necessary funding from outside sources, including obtaining additional funding from the sale of our securities, obtaining loans from various financial institutions or being awarded grants from government agencies, where possible. Our continued net operating losses increase the difficulty in meeting such goals and there can be no assurances that such methods will prove successful.
We expect to incur losses from operations for the near future. We expect to incur increasing research and development expenses, including expenses related to additional clinical trials. We expect that our general and administrative expenses will increase in the future as we expand our business development, add infrastructure and incur additional costs related to being a public company, including incremental audit fees, investor relations programs and increased professional services.
Our future capital requirements will depend on a number of factors, including the progress of our research and development of product candidates, the timing and outcome of regulatory approvals, the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims and other intellectual property rights, the status of competitive products, the availability of financing and our success in developing markets for our product candidates. We believe our existing cash will be sufficient to fund our operating expenses and capital equipment requirements for the next three months. We anticipate we will need approximately $2,500,000 to fund our operating expenses and capital equipment requirements for the next 12 months. We will have to raise additional funds to continue our operations and, while we have been successful in doing so in the past, there can be no assurance that we will be able to do so in the future. Our continuation as a going concern is dependent upon our ability to obtain necessary additional funds to continue operations and the attainment of profitable operations.
| 41 |
We presently do not have any available credit, bank financing or other external sources of liquidity. Due to our history and historical operating losses, our operations have not been a source of liquidity. We will need to obtain additional capital in order to expand operations and fund research and development activities. Future financing may include the issuance of equity or debt securities, obtaining credit facilities, or other financing mechanisms. Even if we are able to raise the funds required, it is possible that we could incur unexpected costs and expenses, fail to collect significant amounts owed to us, or experience unexpected cash requirements that would force us to seek alternative financing. Furthermore, if we issue additional equity or debt securities, stockholders may experience additional dilution or the new equity securities may have rights, preferences or privileges senior to those of existing holders of our common stock.
If additional financing is not available or is not available on acceptable terms, we may be required to delay, reduce the scope of or eliminate our research and development programs, reduce our commercialization efforts or obtain funds through arrangements with collaborative partners or others that may require us to relinquish rights to certain product candidates that we might otherwise seek to develop or commercialize independently.
Transactions with Related Parties
We have entered into an agreement with Lederman & Co., LLC (“Lederman & Co”), a company under the control of Dr. Seth Lederman, our Chief Executive Officer and Chairman of the Board. Effective February 1, 2012, Lederman & Co receives $250,000 per annum for its consulting services. The agreement renews automatically for subsequent terms of one year at $250,000 per annum.
Stock Compensation
In February 2012, we approved the 2012 Incentive Stock Options Plan, which was amended and restated in February 2013 (“2012 Plan”). The 2012 Plan provides for the issuance of options to purchase up to 550,000 shares of our common stock to officers, directors, employees and consultants. Under the terms of the 2012 Plan, we may issue Incentive Stock Options, as defined by the Internal Revenue Code, and nonstatutory options. The Board of Directors determines the exercise price, vesting and expiration period of the options granted under the 2012 Plan. However, the exercise price of an Incentive Stock Option must be at least 100% of fair value of the common stock at the date of the grant (or 110% for any stockholder that owns 10% or more of our common stock). The fair market value of the common stock determined based on quoted market price or in absence of such quoted market price, by the Board of Directors in a good faith. Additionally, the vesting period of the grants under the 2012 Plan should not be more than five years and expiration period not more than ten years. We reserved 550,000 shares of our common stock for future issuance under the terms of the 2012 Plan.
In May 2012, we issued options to purchase 175,000 shares of common stock pursuant to the 2012 Plan, with such options vesting 1/3rd on May 9, 2013 and 1/36th on the 9th of each month thereafter for 24 months, having an exercise price of $30.00 and expiring 10 years from date of issuance. In February 2013, we issued options to purchase 226,500 shares of common stock pursuant to the 2012 Plan, with such options vesting 1/3rd on February 12, 2014 and 1/36th on the 12th of each month thereafter for 24 months, having an exercise price of $10.20 and expiring 10 years from date of issuance.
Lease Commitments
In September 2010, we entered into a five-year lease for office space in New York City, with monthly payments escalating from approximately $10,000 in the first year to approximately $11,000 in the fifth year. The Company received a rent credit of $9,420 in each of the months of November 2010, December 2010 and January 2011. We issued a letter of credit in the amount of approximately $60,000 for the benefit of the landlord, which is collateralized by a money market account. Our future minimum lease payments under the operating lease are as follows:
| 42 |
| Year Ending December 31, | ||||
| 2013 | 96,141 | |||
| 2014 | 131,513 | |||
| 2015 | 100,719 | |||
| $ | 328,373 | |||
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations are based on our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America. The preparation of these financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities and expenses. We evaluate our estimates and judgments on an ongoing basis. We base our estimates on historical experience and on assumptions that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates.
We believe the following critical accounting policies affect our more significant judgments and estimates used in the preparation of our consolidated financial statements.
Research and Development. Tonix outsources its research and development efforts and expenses related costs as incurred, including the cost of manufacturing product for testing, licensing fees and costs associated with planning and conducting clinical trials. The value ascribed to patents and other intellectual property acquired was expensed as research and development costs, as it related to particular research and development projects and had no alternative future uses.
Stock Based Compensation. All stock-based payments to employees and to nonemployee directors for their services as directors consisted of grants of restricted stock and stock options, which are measured at fair value on the grant date and recognized in the consolidated statements of operations as compensation expense over the relevant vesting period. Restricted stock payments to nonemployees are recognized as an expense over the period of performance. Such payments are measured at fair value at the earlier of the date a performance commitment is reached or the date performance is completed. In addition, for awards that vest immediately and are nonforfeitable, the measurement date is the date the award is issued.
Income Taxes. Deferred income tax assets and liabilities are determined based on the estimated future tax effects of net operating loss and credit carryforwards and temporary differences between the tax basis of assets and liabilities and their respective financial reporting amounts measured at the current enacted tax rates. The Company records an estimated valuation allowance on its deferred income tax assets if it is not more likely than not that these deferred income tax assets will be realized. The Company recognizes a tax benefit from an uncertain tax position only if it is more likely than not that the tax position will be sustained on examination by taxing authorities, based on the technical merits of the position. The tax benefits recognized in the consolidated financial statements from such a position are measured based on the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement.
Recent Accounting Pronouncements
There were various updates recently issued, most of which represented technical corrections to the accounting literature or application to specific industries and are not expected to a have a material impact on the Company's consolidated financial position, results of operations or cash flows.
| 43 |
BUSINESS
Corporate Structure
We were incorporated on November 16, 2007 under the laws of the State of Nevada as Tamandare Explorations Inc. From inception through October 2011, we were involved in the acquisition, exploration and development of natural resource properties in the State of Nevada. On October 7, 2011, we executed and consummated the Share Exchange.
In the Share Exchange, the Tonix Shareholders exchanged their shares of Tonix Sub for newly issued shares of Common Stock. As a result, upon completion of the Share Exchange, Tonix Sub became our wholly-owned subsidiary.
Upon completion of the Share Exchange, the Tonix Shareholders received an aggregate of 1,133,334 shares of our Common Stock. David Moss returned 75,000 shares of Common Stock to us, which were retired, and our existing shareholders retained 200,000 shares of Common Stock. The 1,133,334 shares issued to the Tonix Shareholders constituted approximately 85% of our 1,333,334 issued and outstanding shares of Common Stock immediately after the consummation of the Share Exchange.
As a result of the Share Exchange, we acquired 100% of the capital stock of Tonix Sub and consequently, control of the business and operations of Tonix Sub and Krele. From and after the consummation of the Share Exchange, our primary operations consist of the business and operations of Tonix Sub and Krele.
On October 11, 2011, we changed our name to Tonix Pharmaceuticals Holding Corp. to reflect our new business.
Corporate Background
In 1996, Seth Lederman, MD, and Donald Landry, MD, PhD, formed L&L to develop medications for CNS conditions. Dr. Lederman is our Chairman and Chief Executive Officer and Dr. Landry is a Director. L&L was a founder of Janus Pharmaceuticals, Inc., which later became Vela, which developed various therapeutics, including a VLD version of CBP under an agreement with L&L. Vela decided to focus its resources on other programs and transferred the rights to VLD CBP and certain other technologies to L&L in March 2006.
Tonix Sub formed in June 2007 as Krele Pharmaceuticals, Inc. by L&L and Plumbline. Dr. Lederman is Managing Partner of Plumbline. Plumbline possessed rights to certain technology for the treatment of alcohol dependence and abuse. In connection with founding Tonix Sub, L&L and Plumbline entered into an intellectual property transfer and assignment agreement with Tonix Sub for the purpose of assigning patents and transferring intellectual property and know-how in exchange for shares of common stock of Tonix Sub. As a result of economic conditions related to the financial crisis of 2007 and 2008, Tonix Sub was not successful in raising money to fund its programs until 2009. As a result, Tonix Sub was unable to advance the development programs and had little activity except for prosecuting and maintaining patents and maintaining contracts.
In 2009, Tonix Sub contracted with the Toronto Psychiatric Research Foundation to perform the Moldofsky Study. The Moldofsky Study was conducted in Canada by the Toronto Psychiatric Research Foundation, and Tonix Sub obtained the data from this study from L&L. In addition, in 2009, Tonix Sub contracted with Caliper to analyze the interactions of CBP with certain receptors. In June 2010, Tonix Sub entered into consulting agreements with L&L and Lederman & Co and also acquired certain rights to develop isometheptene mucate as a treatment for certain types of headaches from Lederman & Co, which we are developing as TNX-201. Dr. Lederman is managing partner of Lederman & Co. Between June 2010 and October 2011, Tonix Sub was active in recruiting new officers and directors and initiating preclinical and clinical development of novel CBP formulations.
| 44 |
Lederman & Co predominantly provides us with clinical development expertise. L&L predominantly provided us with scientific development expertise until the termination of the consulting agreement in June 2012. Relative to traditional pharmaceutical development companies, we can be considered a virtual company, since we contract with third-party vendors to provide many functions that are core to traditional pharmaceutical companies. For example, we have contracted with PharmaNet Canada to develop methods for analyzing CBP in the blood and to conduct human clinical studies to evaluate the performance of our formulation technology. Lederman & Co is responsible for overseeing the scientific and technical aspects of PharmaNet’s contract work product.
In July 2010, Tonix Sub changed its name to Tonix Pharmaceuticals, Inc. In August 2010, we formed Krele to commercialize products that are generic versions of predicate NDA products. We anticipate that when our branded products lose patent protection, Krele may market authorized generic versions of them. Krele also may develop or acquire generic products approved under ANDAs and we may market branded versions (branded generics) of such products. Krele has been issued a state license in New York.
On April 23, 2013, we formed Tonix Canada. Tonix Canada is intended to perform research and development efforts in Canada. As a Canadian entity, we expect Tonix Canada will be entitled to receive certain reimbursable tax credits for research expenditures in Canada.
Business Overview
We are a specialty pharmaceutical company focused on developing novel pharmaceutical products for challenging disorders of the CNS. Our drug development programs target CNS conditions that manifest as central pain. Central pain results from abnormal pain processing in the CNS, rather than from dysfunction in peripheral tissues where pain is perceived. Our lead program targets FM, a central pain syndrome. Another program targets PTSD, in which central pain is a component. Central pain is associated with disturbed sleep, and improvements in pain or sleep can sometimes help the other symptom. Our products for FM and PTSD are administered at bedtime. We believe that part of the effect on pain in FM is due to improving sleep quality. Our potential therapeutic solutions for FM and PTSD are based on a known pharmaceutical agent that has been approved for use in another indication. We are developing proprietary products that are new dose, formulation, and delivery options tailored to the new therapeutic uses. We intend to start a Phase 2b pivotal study for FM in the third quarter of 2013, which, if successful, will be the first of two pivotal studies required for marketing approval in the U.S.
A number of important CNS drugs have been identified by physicians who observe unexpected improvements in their patients’ CNS conditions despite being prescribed for a different purpose. One of our goals is to establish formal clinical study programs to determine if such anecdotal observations are, in fact, reflections of a compound’s ability to treat a particular CNS condition. While some new applications can use the commercially-available form of a given drug, in other cases, reformulating the active ingredient may improve the active ingredient’s safety or effectiveness in treating the condition. If we demonstrate success in our formal development programs, we will seek marketing approval from the FDA.
We are currently devoting the majority of our efforts to the development of our lead product candidate, TNX-102 SL. TNX-102 SL is a novel dose and formulation of CBP, the active pharmaceutical ingredient of two widely prescribed muscle relaxant products, Flexeril and Amrix. TNX-102 SL is distinct from these products as it is being developed at a dose level significantly below the lowest marketed doses of Flexeril and Amrix. TNX-102 SL is also distinct from these products with regard to its route of administration, as it is designed to be placed under the tongue, to disintegrate, dissolve and provide sublingual absorption, whereas Flexeril and Amrix are designed to be swallowed and to provide absorption in the small intestine. TNX-102 SL is also intended for chronic use, whereas Flexeril and Amrix are marketed for two to three weeks of use. We are currently developing TNX-102 SL for the treatment of FM under an IND filed in the US, and under three CTAs filed in Canada. We are also developing TNX-102 SL for the treatment of PTSD, for which we held a pre-IND meeting with the FDA in October 2012. We expect that any applications we submit for FDA approval of TNX-102 SL will be submitted under Section 505(b)(2) of the FDCA, which we believe will allow for a shorter timeline of clinical development as compared to that needed to fulfill the requirements of Section 505(b)(1), under which NCEs are generally reviewed.
| 45 |
TNX-102 SL is a small, rapidly disintegrating tablet containing CBP for sublingual administration at bedtime. We designed TNX-102 SL to enable the efficient delivery of CBP to the systemic circulation via sublingual transmucosal absorption and to avoid first-pass liver metabolism. We also designed TNX-102 SL to provide CBP at doses lower than those currently available. We have conducted several clinical and pre-clinical pharmacokinetic studies of TNX-102 SL which we believe support its development as a novel therapeutic product for FM and PTSD, and which demonstrate a number of potentially advantageous characteristics as compared to current CBP-containing products, none of which are approved for these indications. Based on our Phase 1 comparative study, we have observed that, as compared to oral CBP tablets, TNX-102 SL results in faster systemic absorption and significantly higher plasma levels of CBP in the first hour following administration. TNX-102 SL was generally well-tolerated, with no serious adverse events reported in this study. Some subjects experienced transient numbness on the tongue after TNX-102 SL administration, and other side-effects reported were similar to those associated with current CBP products.
CBP is the active pharmaceutical ingredient in our lead product candidate, TNX-102 SL. CBP has been approved by the FDA in the U.S. since 1977. We have utilized drug delivery technology to produce new formulations of CBP. In addition to CBP, TNX-102 SL contains excipients, which are well-characterized, are listed in the Inactive Ingredient Guide and are approved for pharmaceutical use. As a result, we anticipate seeking FDA marketing approval of TNX-102 SL through a 505(b)(2) NDA. As one of three types of drug applications, the 505(b)(2) NDA allows drug companies to obtain FDA approval of new drug products without having to conduct the full complement of safety and efficacy trials, which is often the most time-consuming and expensive part of the drug development process. As the 505(b)(2) NDA permits the drug manufacturer to rely on the agency’s findings for a previously-approved drug, published literature, or both, it permits the FDA to make some safety and effectiveness determinations through the review of materials in the public domain or in already approved NDAs of products containing CBP. The 505(b)(2) regulatory pathway would spare us some of the burden of generating all of this data for ourselves and may allow TNX-102 SL to progress through a shorter development pathway than is typical for pharmaceutical products based on novel active ingredients. We have not filed an NDA for TNX-102 SL for any indications.
TNX-102 SL for FM
We are advancing TNX-102 SL for the management of FM. We held an End-of-Phase 2/Pre-Phase 3 meeting with the FDA in February 2013, at which we discussed the design of the clinical program, including the acceptability of the pivotal study design and the proposed registration plan, to support the approval of TNX-102 SL for the management of FM. We believe that positive results from two adequate, well-controlled safety and efficacy studies and the completion of long-term open-label safety exposure studies would support the approval of TNX-102 SL for the management of FM. Under the IND, we plan to initiate a potential pivotal efficacy trial (Phase 2b) in FM (TNX-CY-F202) in the third quarter of 2013. We have entered into a letter of intent to engage Premier Research International, LLC, a CRO to perform key services for the pivotal efficacy trial.
To make sure we have selected the best sublingual formulation for pharmaceutical product development, two prototype sublingual formulations (same amount of active with minor changes in excipients) have been manufactured to compare with the Phase 2b formulation. A formulation comparison PK study (TNX-CY-F104) will be initiated in Canada prior to the initiation of the Phase 2b study (TNX-CY-F202). The selected formulation will be used for the open-label extension safety study (TNX-CY-F203) and the remaining clinical studies supporting the NDA submission. We are planning to conduct a comparative multi-dose PK study (TNX-CY-F105) with the first pivotal study (Phase 2b) drug product and the second pivotal study drug product to establish bioequivalence. Results from this “bridging” study will show that the drug product from the commercial manufacturer is comparable to the Phase 2b drug product manufactured by the clinical manufacturer. It can also support a formulation change if a different formulation (same active with a minor change in excipients) is selected.
During the End-of-Phase 2/Pre-Phase 3 meeting with the FDA, the FDA indicated that it is receptive about presenting results from a pharmacodynamic study in the product labeling. To achieve a more competitive labeling, we plan to conduct a comparative PK/PD study (TNX-CY-F10X) to measure the effect of TNX-102 SL on measures like somnolence at different times.
| 46 |
The process to bring a new drug formulation from concept through testing to approval for a new indication by the FDA is a time-consuming, costly and high-risk process. Once a drug formulation has been tested in laboratories, we need to conduct clinical trials of the product candidate to test its uptake into the blood stream, elimination, effectiveness and safety. Neither laboratory nor animal studies predict the properties of drugs in humans, so designing new formulations can result in a large number of unexpected outcomes. The Phase 1 studies are performed by administering new drug formulations to a limited number of healthy human volunteers and are designed to test the initial concept of the drug formulation and to determine the correct dosage to be tested subsequently on patients affected with the target disorder. The initial Phase 1 studies can take up to a year or more to complete, however, since we reformulate versions of approved drugs for new uses, we may need to devote less time to Phase 1 studies since our testing is informed by significant prior human research that we believe allows us to reduce the possible safety-related outcomes. The next step in the process is to conduct a proof-of-concept efficacy study to identify the effective dose(s). A small Phase 2a efficacy study in the representative patient population will use either a pilot formulation or the formulation selected for further development. A larger study in which the selected formulation has been optimized for the target indication can be referred to as a first pivotal study, a Phase 2b study or a Phase 3 study. If the results of this study are positive and are accepted by the FDA as fulfilling the requirements of a registrational study, then this study may be considered to be one of the two pivotal studies typically required for drug approval. The first pivotal study for a condition like FM typically takes a year to complete and two to three months for data analysis. If the first pivotal study suggests the drug is safe and effective, then a second pivotal “confirmatory” Phase 3 study is conducted. The second pivotal study in FM typically takes 18 months to complete including data analysis. To meet the ICH long-term safety exposure requirement, we plan to conduct one or more long-term safety exposure studies of TNX-102 SL to support the chronic use of TNX-102 SL in FM. Assuming our clinical development of TNX-102 SL in FM meets with success, we would submit an NDA to the FDA seeking marketing approval of TNX-102 SL for the management of FM. We believe it would take approximately six months to prepare and file the NDA and another 14 months to obtain final FDA approval. The drug could be marketed shortly after FDA approval. Therefore, it typically takes more than five years to bring a new formulation of an approved drug to market for a different indication, and any delays in the process, such as lack of capital necessary to run clinical trials, unexpected results, adverse effects, or difficulty in recruiting willing subjects for trials, would result in additional time before a drug could be approved for marketing.
TNX-102 SL for PTSD
We are also advancing TNX-102 SL for the management of PTSD. We held a pre-IND meeting with the FDA in October 2012, and we plan to file an IND to initiate a proof-of concept study in the third quarter of 2013. We plan to begin this trial using 2.8 mg TNX-102 SL tablets in the fourth quarter of 2013. Based on the pre-IND discussion with the FDA, the clinical data necessary to support the PTSD NDA approval will be comprised of positive results from two adequate, well-controlled efficacy and safety studies and the completion of long-term open-label safety exposure studies to meet the FDA/ICH long term safety exposure requirement for new pharmaceutical drug products intended for chronic use.
Pipeline of Additional Product Candidates
In addition to TNX-102 SL, we have developed other innovative formulations of CBP, including TNX-102 gelcap. We have developed TNX-102 gelcap under an agreement with Lipocine, a contract formulation developer and small-scale manufacturer. Although we had met with the FDA to discuss the TNX-102 gelcap development program in August 2011 and we have generated clinical data that support the further development of TNX-102 gelcap, we currently do not plan to advance this candidate.
We also have a pipeline of other product candidates, including TNX-201 and TNX-301. TNX-201 is based on isometheptene mucate and is under development as a treatment for certain types of headaches. Headache is a type of central pain syndrome. TNX-201 is a purified isomer of isometheptene mucate, which has been marketed and approved only as a mixture of two isomers. TNX-301 is a fixed dose combination of two FDA-approved drugs, disulfiram and selegiline, and is under development as a treatment for alcohol abuse and dependence. Central pain is commonly experienced by alcoholics in the period after detoxification. For competitive reasons, we do not disclose the identities of the active ingredients or targeted indications in our pipeline until a U.S. patent has been allowed or issued. Consistent with our mission, these product candidates are or likely will be reformulations of active ingredients that have been used in humans in other products and that are designed for new CNS therapeutic indications.
In other cases, the products will be formulated to match predicate products closely enough to be considered generic copies or similarly enough to other marketed products to rely (in part) on their regulatory review and approval, as well as available published data. The predicate product may be approved by the FDA under an NDA or may have been reviewed for safety and effectiveness by the National Academy of Sciences under the DESI, program, in which case they would be considered by FDA to be “unapproved products”. For DESI products, it is our intent to develop NDA versions to meet cGMP and ICH requirements to seek approval under the 505(b)(2) regulatory pathway.
| 47 |
Because of our size and being in the development stage, we do not currently devote a significant amount of time or resources towards our other pipeline candidates. We may perform non-clinical development work on TNX-201 and possibly on TNX-301, but we do not expect to start clinical trials of either of these candidates until 2014 at the earliest.
Our Strategy
Our objective is to develop and commercialize our product candidates to treat CNS conditions, including FM and PTSD. The principal components of our strategy to achieve this objective are to:
| · | pursue development and regulatory approval pathways by reformulating versions of approved drugs for new uses and by using the Section 505(b)(2) regulatory pathway for NDA approval; | |
| · | adopt a multi-pronged patent strategy to protect our products, including patents which protect methods of use for the active ingredients in our products, the formulation technology employed in our products, and the performance characteristics of our products in the human body; | |
| · | provide clear value propositions to third-party payers, such as managed care companies or government programs like Medicare, to merit reimbursement for our product candidates; and | |
| · | enter into collaborations with other pharmaceutical companies with respect to, among others, our FM and PTSD product candidates and other products that will benefit from development or marketing resources beyond those in our Company. |
Pursue development and regulatory approval pathways. We believe our lead product candidates may be approvable under pathways that are potentially shorter than those typically available for drug products based on novel active ingredients (Section 505(b)(1)). By focusing on developing new formulations of approved drugs for new uses, we believe that we will be able to use the Section 505(b)(2) regulatory pathway for NDA approval. This pathway can reduce the time and expense required for our development programs by allowing our use of previously-generated safety and efficacy information regarding the active pharmaceutical ingredients in our lead product candidates to support the filing and approval of our NDA application. Our use of this information may help reduce the size and scope of our preclinical and clinical trials. The FDA has strict requirements on the marketing of drugs, and FDA approves each drug for specific uses which are called the label indications. The use of a drug product for the treatment of a condition other than one of its approved label indications is called off-label use. The development of an existing FDA-approved drug for the treatment of a condition other than one of its approved label indications is considered a “new use”. For companies involved in the ethical development and marketing of prescription drugs in the US, FDA approval of a new use or new label indication is the only legal basis of marketing claims for that use or indication. Off-label use is not recognized by the FDA or FDA-regulated companies as a new use.
Adopt a multi-pronged patent strategy. We are pursuing a multi-pronged patent strategy by seeking intellectual property protection on several aspects of our products. Aspects we seek to protect include, among others, methods of use for certain known active pharmaceutical ingredients, formulation technologies incorporated into our products, and performance characteristics of our products in the human body. With respect to methods of use patents, we believe the therapeutic uses we target are new uses for these active ingredients and we have been issued patents directed to certain aspects of our new uses. For example, the invention of bedtime VLD CBP as a treatment for FM was novel and unexpected when our patents were filed in 2000. We are seeking additional patents to cover other new uses. For example, we filed a patent application seeking to protect the use of CBP in PTSD. With respect to formulation patents, we believe our products will be protected by patents that describe inventions of technology for making new formulations, which may include novel routes of delivery for the active ingredients. With respect to patents related to the performance characteristics of our products in the human body, we believe our products will be protected by patents that describe novel pharmacokinetic properties of the active ingredient, as well as of its active metabolites, at certain times after administration. For example, we filed a patent application seeking to protect novel pharmacokinetic properties of CBP as enabled by TNX-102 SL.
| 48 |
Provide clear value propositions to third-party payors to merit reimbursement for our product candidates. We are designing our clinical development programs to demonstrate compelling competitive advantages to patients and prescribers and also to demonstrate value propositions to third-party payors. We believe TNX-102 SL might help in the management of FM by reducing pain and other symptoms, such as fatigue. In addition, primarily as a result of its lower dosage, we believe that bedtime treatment with TNX-102 SL will have fewer day time side-effects than off-label bedtime treatment with immediate-release CBP, or CBP IR. For FM, we believe an FDA-approved product would capture some of the off-label use of generic CBP. Because FDA approvals are based on objective data, we believe that third-party payors will provide reimbursement for an FDA approved product, even at a premium price relative to other drugs that are used off-label, such as CBP IR, tizanidine, baclofen, carisoprodol or metaxalone. For example, third-party payors reimburse the use of Lyrica® and Cymbalta® for FM despite the availability of off-label generic versions of drugs with similar mechanisms of action, for example, Neurontin® (gabapentin) and generic anti-depressants, respectively.
Enter into collaborations to maximize the value of our technology. We believe certain of our drug development candidates, including TNX-102 SL, can be developed and marketed more effectively by companies that already have significant drug development and marketing capabilities. We will seek to enter into collaborations with pharmaceutical or biotechnology companies for the commercialization of these product candidates at the times we believe most effective.
Our Lead Product Candidates
Our lead product candidate is TNX-102 SL, for the treatment of FM and PTSD. TNX-102 SL consists of CBP in a mixture of inactive ingredients that are called “excipients”, which we believe will improve the absorption rate of CBP in ways that will optimize the product for bedtime treatment. The excipients used in TNX-102 SL are approved by the FDA for pharmaceutical uses.
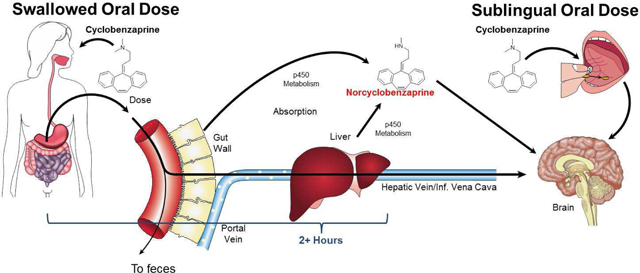
Figure 1: Absorption of oral and sublingual cyclobenzaprine and effect of liver metabolism
Cyclobenzaprine
CBP was first synthesized in 1961 by Merck, and the 10 mg Flexeril immediate-release, or IR, dose form was FDA approved in 1977 for the relief of muscle spasm associated with acute, painful musculoskeletal conditions as an adjunct to rest and physical therapy.
| 49 |
Although a number of clinical studies have addressed the potential use and benefit of CBP in treating symptoms of FM, to our knowledge these studies have not motivated a sponsor to pursue FDA approval.
Based on CBP’s safety and efficacy for treating muscle spasm, in the 1990s, Merck conducted studies to support an application to market a 5 mg Flexeril tablet (low dose) for the over-the-counter, or OTC, market, whereby patients can purchase medicine without a physician’s prescription. Although Merck’s studies re-affirmed the safety and demonstrated efficacy of 5 mg Flexeril in several large trials, the OTC division of the FDA rejected the application for use without a prescription, apparently, we believe, because muscle spasm was deemed a condition that required a physician to diagnose and supervise treatment.
Merck divested the Flexeril franchise to Alza Pharmaceuticals, or Alza. Alza subsequently was acquired by Johnson and Johnson and Flexeril is part of their McNeil Specialty Pharmaceuticals division. Based largely on the Merck studies, McNeil won approval of Flexeril 5 mg tablets as a prescription medicine to treat muscle spasm. McNeil promoted Flexeril 5 mg tablets for the three year period of market exclusivity based on The Drug Price Competition and Patent Term Restoration Act of 1984, generally referred to as the Hatch-Waxman Act. Following this exclusivity period, several generics entered the market and took market share from Flexeril. McNeil has discontinued the manufacture of Flexeril.
Despite the approved uses of CBP in treating muscle spasm, we believe current marketed formulations of CBP are limited for treating FM by slow and unpredictable absorption. Following the ingestion of CBP IR, it takes more than one hour for clinically-meaningful blood levels to be achieved. As described in the Flexeril package insert, the amount of CBP absorbed into the bloodstream varies between 33-55% of the dose ingested. The variability in absorption may be due to several factors, including effects of the stomach pH (acidity or base) on the dissolution of the tablets, as well as the context of either an empty stomach or a recent meal. Food in the stomach and small intestine from a recent meal contributes to variability in absorbing other drugs. The uncertainties in absorption rates make it challenging for a physician contemplating a bedtime treatment for FM to ensure the intended therapeutic effect is achieved without risking side effects like next-day drowsiness, which could result if the patient has too much CBP remaining in the bloodstream the next day.
If a product could provide rapid and consistent absorption of CBP, patients would be more likely to receive a drug exposure profile that is aligned with the intended period of exposure and less likely to receive too little drug to receive a therapeutic effect. Conversely, patients would be less likely to receive too much drug, which might lead to potential side effects, including next-day drowsiness. An optimal bedtime VLD CBP product could have faster absorption, lower blood levels in the morning and more predictable effects than the IR tablet format. We have tested a number of technologies to optimize the properties of VLD CBP as a bedtime therapy for FM and PTSD. Our lead product, TNX-102 SL is a novel sublingual tablet form of VLD CBP that we have tested in pre-clinical and clinical studies. We intend to enter TNX-102 SL into a potential pivotal clinical trial program in FM in the third quarter of 2013, and begin a Phase 2 proof-of-concept trial in PTSD in the fourth quarter of 2013. We believe the unique properties of TNX-102 SL, as demonstrated by the results of our studies, support its development in both FM and PTSD. We have developed other innovative formulations of CBP, including TNX-102 gelcap. Although we had met with the FDA to discuss the TNX-102 gelcap development program in August 2011 and we have generated clinical data that support the further development of TNX-102 gelcap, we currently do not plan to advance this candidate.
TNX-102 SL in Fibromyalgia Syndrome
TNX-102 SL, our most advanced product candidate, is a rapidly disintegrating tablet containing VLD CBP that is designed to be placed under the tongue at bedtime. The development of TNX-102 SL in FM is supported by the results of the Moldofsky Study, which evaluated oral administration of CBP at doses below the lowest marketed dose in the evening, as well as by preclinical and comparative clinical pharmacokinetic studies.
In the Moldofsky Study, which was a randomized, double-blind, placebo-controlled, Phase 2a trial, it was demonstrated that VLD CBP in a capsule swallowed between dinner and bedtime resulted in significant decreases in next-day pain and other core FM symptoms, as well as in a significant improvement in sleep quality. We believe that CBP exerts its benefit in FM via its ability to improve the restorative quality of sleep, which has been shown to be frequently impaired in patients with FM or PTSD. Current CBP products are believed to be widely used off-label by FM patients.
| 50 |
FM is diagnosed by groups of symptoms that have been defined by committees of the American College of Rheumatology, or ACR, and a committee of experts from the organization Outcome Measures in Rheumatology. In 2007, Pfizer’s Lyrica (pregabalin) became the first medicine approved by the FDA for the management of FM. In 2008, Eli Lilly’s Cymbalta (duloxetine) became the second medicine approved by the FDA for the management of FM. In 2009, Savella® (milnacipran) was the third medicine approved by the FDA for the management of FM. Savella is marketed by Forest Laboratories.
Product Development Plan
Phase 2a Data of VLD CBP in FM Patients
Our motivation to focus our efforts on developing TNX-102 SL for FM stems from the results of the Moldofsky Study, the related rights to which we acquired from L&L. Specifically, this study was a randomized, double-blind, placebo-controlled, dose-escalating eight week trial conducted at two study centers. The study subjects met ACR criteria for FM.
Patients received VLD CBP IR 1 mg capsules or corresponding placebo capsules to ingest after dinner and before bedtime. Initially, patients took one capsule each evening, but over the course of the study, they were allowed to increase the number of capsules taken in increments of one capsule per week. The maximum number of capsules allowed was four per evening, which would be a total dose of 4 mg CBP IR.
Patients treated with VLD CBP demonstrated significant improvements in pain, fatigue and tenderness at week 8 relative to baseline, whereas placebo-treated patients did not improve (Figure 1). Although this study excluded patients who met formal criteria for major depressive disorder or any anxiety disorder, there is a high degree of co-existing symptoms of depression and anxiety associated with FM. VLD CBP treatment resulted in significant reductions in total Hospital Anxiety and Depression Scale, or HAD, which measures symptoms of anxiety and depression, and the HAD depression subscale which measures depressive symptoms (Figure 1).

Figure 2: Results of a Phase 2 Study of VLD CBP in FM Patients as administered between dinner and bedtime
| 51 |
This study showed treatment with VLD CBP:
| · | provided benefit in core symptoms of FM, including pain and fatigue; | |
| · | improved mood, by demonstrating a significant decrease in HAD scores; and | |
| · | was well tolerated, with no serious adverse events, or SAEs, or discontinuations due to adverse events, or AEs. |
This study also showed that VLD CBP taken between dinner and bedtime resulted in a significant improvement in sleep quality. We believe that CBP exerts its benefit in FM via its ability to improve the restorative quality of sleep, which has been shown to be frequently impaired in patients with FM.
This research was published in the Journal of Rheumatology, in an article entitled “Effects of Bedtime Very Low Dose (VLD) Cyclobenzaprine (CBP) on Symptoms and Sleep Physiology in Patients with Fibromyalgia Syndrome (FM): A Double-blind, Randomized, Placebo-controlled Study.” The citation is: Moldofsky H, Harris H, Kwong T, Archambault WT and Lederman S. J Rheum 2011 Dec;38(12):2653-63.
Pharmacokinetic and Bioequivalence Studies
We have conducted two preclinical and two clinical studies of our sublingual formulations of CBP, which have evaluated the pharmacokinetics of these formulations as well as their comparative bioavailability to oral CBP.
Our preclinical animal studies demonstrated that our sublingual formulations provide faster delivery and more efficient systemic absorption of CBP as compared to current oral forms of the drug.
Our first clinical study of sublingual CBP evaluated a solution formulation in which certain key ingredients of TNX-102 SL were delivered under the tongue in a small volume of water. This single-dose study was conducted in Canada. The trial enrolled 23 healthy volunteers, and subjects received one of: a sublingual solution containing 2.4 mg of CBP and sublingual absorption-enabling ingredients of TNX-102 SL (Arm 1), a sublingual solution that was designed to simulate crushed CBP IR tablets, i.e., without the sublingual absorption-enabling ingredients (2.4 mg) (Arm 2), an oral CBP IR tablet (5 mg) (Arm 3), or intravenous CBP (2.4 mg) (Arm 4). The study measured circulating blood levels of CBP at pre-defined time-points over six days after receiving study medication. Patients receiving sublingual formulations were instructed to spit and rinse 90 seconds following administration. The results demonstrated that the solution formulation of TNX-102 SL (Arm 1) delivered CBP to the systemic circulation more efficiently than the sublingual solution of a simulated crushed tablet (Arm 2) and faster than the ingested tablet (Arm 3). In the study, all of the CBP formulations were well-tolerated, and there were no unexpected adverse events.
Our second clinical study of sublingual CBP evaluated TNX-102 SL, the sublingual tablet formulation we expect to advance into further development. This study was conducted in Canada. This study enrolled 24 healthy volunteers and evaluated a single dose of one 2.4 mg tablet or two tablets (4.8 mg) of TNX-102 SL or the currently-marketed 5 mg CBP tablet. In comparison to oral administration of the 5 mg CBP tablet, both sublingual doses of TNX-102 SL demonstrated faster systemic absorption. After administration of TNX-102 SL, blood levels of CBP were significantly higher at 20, 30, 45 and 60 minutes relative to administration of the 5 mg CBP tablet. In the study, TNX-102 SL was generally well tolerated. There were no unexpected adverse events, with the exception of a mild, temporary numbness at the tongue experienced by less than one-third of the subjects that received TNX-102 SL tablets.
Prospective Phase 2b Study
We expect to advance the clinical development of TNX-102 SL, containing 2.8 mg CBP, for the management of FM by conducting a Phase 2b study. In this multicenter, randomized, double-blind, placebo-controlled clinical trial, FM patients will be administered either TNX-102 SL or placebo at bedtime nightly for 12 weeks. We expect to enroll 100-200 patients into this study. We expect that our proposed Phase 2b study, if successful and accepted by the FDA, will be one of the two pivotal studies required to support the NDA approval.
| 52 |
The primary efficacy measure in this study will be the change in pain severity at week 12 with TNX-102 SL as compared to placebo, as assessed by the Numeric Rating Scale, or NRS. This endpoint is similar to that utilized in clinical trials of drug products currently approved for use in FM. We will also collect information on other outcome measures, including NRS scores at other time points, the revised Fibromyalgia Impact Questionnaire, and the Patient Global Impression of Change. We have entered into a non-binding letter of intent to engage Premier Research International, LLC, a CRO, to perform key services for the pivotal efficacy trial. Subsequent to receiving FDA concurrence with our proposed protocol, including the methodology for primary endpoint analysis, the study will begin enrollment in the third quarter of 2013 and we expect it will be completed in the second half of 2014. We have contracted with a CMO to manufacture and perform stability testing on TNX-102 SL tablets for this Phase 2b study.
Prospective Multi-dose Pharmacokinetic Study
Since CBP will be used chronically in TNX-102 SL, we will study TNX-102 SL in comparison to CBP IR in a multiple-day dosing (once daily) study. Subjects will receive TNX-102 SL or CBP IR for four or more consecutive days. Peak and trough blood levels of CBP will be measured. The results of this study will provide information regarding blood levels of CBP resulting from use of the marketed IR tablet and our sublingual TNX-102 SL tablet when taken in a multiple day regimen. We expect the data from this study to serve as a ‘bridge’, in that they will allow us to use the CBP IR tablet as the reference product in our submission of a Section 505(b)(2) NDA for TNX-102 SL.
Prospective Study Comparing Safety and Tolerability of TNX-102 SL with CBP IR
We plan to conduct a small study designed to evaluate next morning drowsiness and other cognitive measures following the bedtime use of TNX-102 SL and the bedtime use of CBP IR. The goal of this study is to determine the potential benefit of TNX-102 SL compared with CBP IR on next morning drowsiness and on other cognitive functions.
Prospective Phase 3 Study
If our Phase 2b study of TNX-102 SL is successful, then we expect to conduct a Phase 3 confirmatory study in support of product registration. At that time, we plan to conduct a randomized, double-blind, placebo-controlled Phase 3 study in which patients with FM will receive TNX-102 SL or placebo at bedtime nightly for 12 weeks. It is likely that the primary efficacy measure in this study will be the change in pain severity with TNX-102 SL as compared to placebo at week 12, assessed by the NRS, similar to the primary efficacy measure of the Phase 2b study. Secondary outcome measures will be carefully considered to best support desired label claims and to optimize the marketing message for product differentiation. We expect approximately 300 FM patients will be enrolled in this trial.
Safety Exposure Study
To evaluate the safety of TNX-102 SL for chronic use, we expect to conduct one or more long-term open-label safety exposure studies. The FDA agreed that the safety database needed to support a 505(b)(2) NDA submission for TNX-102 SL would contain a total exposure of at least 300 FM patients, with at least 100 patients receiving TNX-102 SL for six months and at least 50 patients for one year. We plan to conduct open-label extension studies in which patients may be eligible to enroll following their completion of our Phase 2b and Phase 3 safety and efficacy trials in FM.
Nonclinical
In addition to the clinical data necessary to support the TNX-102 SL 505(b)(2) NDA filing for the fibromyalgia indication, the FDA also clarified the nonclinical studies required for the NDA filing since the information from the reference product is either unavailable for reference or failed to meet the current regulatory standard. We have initiated discussions with several Good Laboratory Practices-compliant laboratories to conduct a six month repeated-dose toxicology study in rats, a nine month repeated-dose toxicology study in dogs and a peri- and post-natal Segment III study required for the NDA filing. We plan to submit these draft toxicology protocols for FDA review and seek agreement on the doses and facilities chosen for these studies. These studies will be performed concurrently with the pivotal clinical trial program and will be completed for the NDA submission. Based on the Flexeril labeling, and post-marketing surveillance information, there is no evidence of abuse for CBP. As a result, TNX-102 SL will not have to assess the abuse potential of CBP for the NDA submission.
| 53 |
Manufacturing
The TNX-102 drug product that has been manufactured for our first pivotal study (Phase 2b) was manufactured in a small-scale GMP facility that is licensed to manufacture clinical trial materials, but not equipped for large-scale commercial production. For the second pivotal study and for the commercial product, we will engage a commercial GMP facility that is capable of manufacturing the registration batches to support the NDA. The product’s comparability will be supported by the bioequivalence results from the “bridging” study, TNX-CY-F105.
Other NDA Requirements
We have submitted an initial Pediatric Study Plan, or PSP, which contains a full waiver of the requirement to submit pediatric assessments per Section 505B(a)(4)(A)(i) of the FDCA, since pediatric studies are impossible or highly impractical because the number of patients is so small (Section 505B(a)(4)(A)(i) of the FDCA) and TNX-102 SL tablets do not represent a meaningful therapeutic benefit over existing therapies for pediatric patients, and is not likely to be used in a substantial number of pediatric patients (Section 505B(a)(4)(A)(iii) of the FDCA). Furthermore, TNX-102 SL will be submitted as a 505(b) NDA, referencing the approved CBP products (Flexeril and Amrix) for safety. Based on the approved Flexeril and Amrix labeling, the safety and effectiveness of CBP has not been studied in pediatric patients.
Based our discussions with the FDA and the FDA formal meeting minutes, we will not have to conduct special populations (geriatric and renal/hepatic impaired), drug-drug interaction or a cardiovascular safety study to support the NDA filing. Due to the well-established safety profile of CBP at much higher doses than we proposed for FM, the FDA requests no risk management plan or medication guide for this product.
Regulatory Strategy
The FDA approvals of Lyrica, Cymbalta and Savella establish a regulatory approval standard for the management of FM. However, given the heterogeneity of patients with this disease, it may not prove to be the only pathway or approval requirement. We hope to register TNX-102 SL with the FDA through the provisions of Section 505(b)(2). This regulatory pathway may help to accelerate product development and reduce overall business risk. The 505(b)(2)-based product development plan for TNX-102 SL is designed to leverage the safety data that have been generated by other manufacturers for CBP-containing products and accepted by the FDA in support of their product registrations, in addition to the safety data we generate. TNX-102 SL contains significantly less active CBP than other marketed products. We believe that the safety data package from these products and the CBP prescriptions utilization database analyzed by IMS Health Incorporated will provide adequate safety margin to support TNX-102 SL development. We held an End-of-Phase 2/Pre-Phase 3 meeting with the FDA in February 2013, at which we discussed the nature and extent of the Phase 2b and Phase 3 clinical trials we need to conduct to so as to receive regulatory acceptance of our proposed NDA plan for a differentiated product for the management of FM.
If NDA approval of TNX-102 SL is granted, in addition to the three-year marketing exclusivity granted by law, we expect this product to be protected by patents that extend through at least 2021, during which time it should not be subject to generic substitution. We plan to continue to support the TNX-102 SL program with new patent applications as we obtain data from the clinical evaluation of our new formulation in healthy human subjects and in FM patients. For example, we have recently filed patent applications on TNX-102 SL which, if issued, would be expected to provide protection from generic substitution until 2033.
| 54 |
TNX-102 SL in Post-Traumatic Stress Disorder
We are also developing TNX-102 SL for the management of PTSD, a psychiatric disorder that begins in the aftermath of traumatic experiences. We held a pre-IND meeting with the FDA in October 2012, at which our clinical program for PTSD was discussed. We have not yet conducted any clinical trials on PTSD patients.
Parallels Between FM and PTSD
A number of parallels have been noted between FM and PTSD. In addition, symptom overlap may exist between patients diagnosed with FM or PTSD. In a survey of males with PTSD or major depression (Amital et al, Posttraumatic stress disorder, tenderness, and fibromyalgia syndrome: are they different entities? J. Psychosom. Res. 2006, 61(5):663-9), 49% of PTSD patients met the ACR criteria for FM compared to 5% of major depression patients. Conversely, in a different survey of FM patients (Cohen et al., Prevalence of post-traumatic stress disorder in fibromyalgia patients: overlapping syndromes or post-traumatic fibromyalgia syndrome? Semin. Arthritis Rheum. 2002, 32(1):38-50), 57% of the sample had symptoms associated with PTSD.
A core feature of PTSD is sleep disturbance, including insomnia and nightmares. Sleep disturbances are believed to exacerbate daytime symptoms of PTSD, including irritability, poor concentration, and diminished interest in significant activities. We believe the sleep disturbances of PTSD bear similarity to those associated with FM.
Emerging Market Opportunity
The selective serotonin reuptake inhibitors Paxil® (paroxetine) and Zoloft® (sertraline) are FDA approved for PTSD, but are not satisfactory treatments for many patients. Other drugs that show promise for the treatment of PTSD, but are not FDA approved, include antidepressants such as nefazodone, mirtazapine and trazodone; the antihistamine cyproheptadine; certain atypical antipsychotics such as olanzapine and risperidone; and an adrenergic alpha-1 receptor blocker, prazosin. Prazosin may decrease nightmares and insomnia and has been associated with improvements in daytime PTSD symptoms, depression, and quality of life.
Our rationale for studying the effects of CBP in PTSD derives from the following:
| · | our clinical studies that VLD CBP improves FM symptoms, a disorder having significant overlap with PTSD; | |
| · | our clinical studies that VLD CBP can improve sleep quality, which is impaired in PTSD; and | |
| · | in studies conducted by Caliper, CBP interacts with a receptor on brain cells called the serotonin type 2a receptor. Based on numerous peer-reviewed scientific publications, we have identified a number of compounds that bind this receptor that have been shown to have effects in treating PTSD. Therefore, it is our belief that CBP, because it binds to the serotonin type 2a receptor, will have a therapeutic effect in treating PTSD. |
As very little information was available on the biochemical effects of CBP and its primary metabolite, norcyclobenzaprine, or nCBP, in the central nervous system, we have engaged several CROs to better understand the interactions of these agents with certain receptors in the brain. CROs we have engaged in this effort include Caliper, Cerep, Millipore, and DiscoveRx. Results from a series of binding and functional studies show that both of these molecules are potent antagonists of the serotonin type 2a and the histamine H1 receptors, which known to have effects on sleep and sleep maintenance. The results also show that CBP and nCBP antagonize the adrenergic alpha 1A and 1B receptors, which may have effects on autonomic dysfunction. The results of some of these studies were presented at a poster session during the 2012 American College of Rheumatology Annual Meeting (Daugherty et al, “Cyclobenzaprine (CBP) and its Major Metabolite Norcyclobenzaprine (nCBP) are Potent Antagonists of Human Serotonin Receptor 2a (5-HT2a), Histamine Receptor H1 and Alpha-Adrenergic Receptors: Mechanistic and Safety Implications for Treating Fibromyalgia Syndrome by Improving Sleep Quality”, Abstract #960).
| 55 |
Product Development Path
We anticipate that the dose of TNX-102 SL sufficient to treat PTSD symptoms may be higher than that sufficient to treat FM. We plan to utilize the data obtained from our pharmacokinetic studies of TNX-102 SL to inform the design of efficacy trials in PTSD.
Based on the recommendations and guidance received at our October 2012 pre-IND meeting with the FDA, we plan to file an IND application for TNX-102 SL in the PTSD indication in the third quarter of 2013, and to begin a Phase 2 proof-of-concept trial in the fourth quarter of 2013. We expect to be able to use TNX-102 SL tablets manufactured for the FM studies in the initial PTSD clinical trials.
Prospective Proof-of-Concept Phase 2 Study
We plan to use the IND to support a small clinical study to ascertain the potential efficacy of TNX-102 SL in this disorder. This will be a randomized, double-blind, placebo-controlled, crossover study in subjects with PTSD. TNX-102 SL and placebo will be administered once daily at bedtime. The primary efficacy measure will be the change in the Clinician-Administered PTSD Scale from baseline to week six. Secondary outcome variables may include the PTSD Dream Rating Scale, the PTSD Checklist, the Clinical Global Impression of Improvement, the Pittsburgh Sleep Quality Index and the Beck Depression Inventory. In addition, polysomnograms may be obtained at baseline and at specified times during the trial.
Prospective Phase 3 Studies
If our Phase 2 trial of TNX-102 SL in PTSD is successful, we intend to conduct two multicenter, double-blind, placebo-controlled, Phase 3 studies designed to evaluate the efficacy, safety, and tolerability of TNX-102 SL in patients with PTSD. We expect both of these Phase 3 studies to be of 12 weeks’ duration and of crossover design. We expect the results of the Phase 2 trial to determine dose levels in these Phase 3 trials, but like the Phase 2 trial, TNX-102 SL may be dosed flexibly. The primary endpoints for both Phase 3 studies are anticipated to be similar to those proposed to be featured in the Phase 2 study, and as with the Phase 2 study, in addition to standardized measures of PTSD symptomatology and severity, polysomnograms may be obtained.
Regulatory Strategy
The approvals by the FDA of Paxil (paroxetine) and Zoloft (sertraline) for treating PTSD establish a regulatory approval pathway for symptom reduction in PTSD. We believe our clinical development program of TNX-102 SL and the chronic safety data generated from the TNX-102 SL FM NDA program will result in a differentiated product suitable for chronic use for the treatment of PTSD. We believe that our planned clinical trials in PTSD, if successful, will provide sufficient evidence of clinical efficacy and safety to support a 505(b)(2) NDA for TNX-102 SL for the management of PTSD.
We held a pre-IND meeting with the FDA in October 2012, and we plan to file an IND to initiate a proof-of concept study in the third quarter of 2013. We plan to begin this trial using 2.8 mg TNX-102 SL tablets in the fourth quarter of 2013. Based on the pre-IND discussion with the FDA, the clinical data necessary to support the PTSD NDA approval will be comprised of positive results from two adequate, well-controlled efficacy and safety studies and the completion of long-term open-label safety exposure studies to meet the FDA/ICH long term safety exposure requirement for new pharmaceutical drug products intended for chronic use. Any change in formulation after the completion of the pivotal study will be supported by a “bridging” study demonstrating bioequivalence of the clinical and commercial formulations. Justified by the approved Flexeril labeling and supported by post-marketing surveillance data, we do not anticipate the need to conduct abuse potential assessment study for the PTSD NDA filing. Our initial Pediatric Study Plan is to request a partial waiver for age 4-11 years old and a deferral for the age 12-17 years old until efficacy and safety have been demonstrated in adult PTSD patients. Similar to the FM NDA, we do not anticipate the need to conduct special populations (geriatric and renal/hepatic impaired), drug-drug interaction and thorough cardiovascular safety assessment studies to support the PTSD NDA filing. Since the PTSD patient population is different from the FM patient population, we plan to discuss with the FDA at the End-of-Phase 2 meeting the need for a risk management program for this product.
| 56 |
Drug Delivery Technology
TNX-102 SL
TNX-102 SL is a small tablet that rapidly disintegrates in saliva and transmucosally delivers CBP into the systemic circulation. TNX-102 SL contains sublingual absorption-enabling ingredients that promote a local oral environment that facilitates oromucosal absorption of CBP. We own all rights to TNX-102 SL in all geographies, and we bear no obligations to third-parties for any future development or commercialization.
TNX-102 Gelcap
In June 2007, we entered into a Feasibility and Option Agreement with Lipocine, which was amended in October 2010 (the “Feasibility Agreement”). Pursuant to the Feasibility Agreement, we identified and obtained an exclusive worldwide option on technology from Lipocine that employs mixtures of different types of lipids to envelop CBP molecules in the small intestine and facilitate absorption into the bloodstream. We believe this approach has potential for more consistent absorption and decreased variability in blood levels.
The Feasibility Agreement provided for two stages of work, stated as Stage I and Stage II. The Stage I work involved developing methods and testing compatibility between Lipocine’s technology and our drug formulation. The Stage II work involved supporting us in our efforts to conduct a clinical trial study, based on the Stage I work, and is expected to conclude upon the completion of a final report on the results of the clinical study (the “Final Report”). Upon completion of the Final Report, we have the right, within 30 days after the Final Report, to exercise an exclusive worldwide license to the Lipocine technology.
Under the Feasibility Agreement, Lipocine completed the Stage I work, which involved studying a number of combinations of lipids for their ability to form micelles that solubilize the free base of CBP and which might serve as inactive ingredients in a gelatin capsule formulation. We selected a candidate formulation, TNX-102 gelcap, based on properties that included the dispersion of the active ingredient in simulated gastric or small-intestinal fluids and the stability of the formulation over time prior to testing. Lipocine was also engaged to manufacture gelatin capsules of TNX-102 gelcap for use in a pharmacokinetic trial.
In August 2011, we provided notice to Lipocine that we intended to move forward with the Stage II work. The clinical phase of the Stage II trial was completed during the fourth quarter of 2011. Some of the data has been collected and some data is still awaiting the development and validation of assays. We are working to analyze the data and write the Final Report, which is anticipated to be completed in 2013. After completion of the Final Report, we will have 30 days to decide whether to exercise the option to license Lipocine’s US patent 6,294,192 “Triglyceride-free compositions and methods for improved delivery of hydrophobic therapeutic agents” and US Patent 6,451,339 “Compositions and methods for improved delivery of hydrophobic agents”. These patents expire on September 24, 2021 and September 16, 2022, respectively.
If we elect to exercise the option, we will execute a license agreement with Lipocine. If we exercise the option to license these patents, we will be obligated to pay Lipocine low single-digit percentage royalties based on net sales or mid-teen sublicense fees based on the consideration that we receive from a licensee. The maximum amount of milestone payments we could be required to pay to Lipocine pursuant to the Feasibility Agreement is $3,000,000. We currently do not plan to exercise the option with Lipocine.
Market Dynamics
We believe the U.S. market for products that treat CNS conditions has several characteristics that make it an attractive market for pharmaceuticals, including that the customer base is driven by physicians who are involved in long-term care of patients with chronic disorders. Patients with CNS disorders sometimes carry disease burdens that require long-term treatment.
| 57 |
We believe the market for FDA-approved FM treatments is underserved and that there is a constant need for new treatment options, since many prescription drugs provide relief only to some of the affected patients, only to some of some patients’ symptoms, or provide relief only for limited periods of time.
In 2007, Lyrica (pregabalin) became the first medicine approved by the FDA for the management of FM. Lyrica previously had been approved and marketed to treat pain in other conditions as well as epilepsy. In 2008, Cymbalta (duloxetine) became the second medicine approved by the FDA for the management of FM. Cymbalta previously had been approved and marketed to treat depression. FM shares a number of symptoms with depression, and a number of FM patients are believed to experience depression as a co-existing condition. Savella (milnacipran) was the third medicine approved by the FDA for the management of FM. Savella’s active ingredient, milnacipran, is approved for the treatment of depression in Europe.
As many products used for the treatment of FM are approved and marketed for other conditions, sales of these products related specifically to FM can only be estimated. According to a study conducted by Frost & Sullivan on behalf of Tonix relating to the FM market, U.S. sales of prescription drugs specifically for the treatment of FM totaled $1.2 billion in 2010, which had grown at a compounded annual growth rate of 18.4% since 2007. This figure includes sales of Lyrica, Cymbalta, and Savella of $478 million, $342 million, and $68 million, respectively. Despite the availability of FDA approved products, we believe the current treatment options for FM continue to leave many patients dissatisfied.
Prior to 2007, the landscape of prescription drugs used to treat FM was characterized by off-label use of generically-available therapies. Drugs that had been prescribed as the primary treatments for FM were approved for other indications, with analgesics, antidepressants, and muscle relaxants among the categories receiving the greatest use by the FM population. Despite the significant FM-related sales growth of the three products approved for FM following their approvals for this indication, according to Frost and Sullivan, the unit volume of medications prescribed to specifically treat FM had been nearly flat between 2007 and 2010, implying that the sales growth of the approved products was mainly driven by patients switching from off-label, generic medications to on-label, branded medications. In particular, these market dynamics are consistent with the interpretation that Lyrica’s growth in FM was driven by switching from off-label analgesics, and Cymbalta’s and Savella’s growth in FM was driven by switching from off-label anti-depressants. Increasingly, Cymbalta, Savella and Lyrica are recognized as central pain inhibitors and not just treatments for their original indications.
Despite the wide use of muscle relaxants by FM patients, this category lacks a product approved for FM. Demand continues to be satisfied by off-label medicines such as CBP, tizanidine, baclofen, carisoprodol and metaxalone. These muscle relaxants have generic and branded versions. According to Frost and Sullivan, 48 million doses of the Flexeril brand and its associated CBP IR generic products were prescribed off-label for FM in 2010 and accounted for approximately 35% of the daily doses of muscle relaxants prescribed for FM that year. These figures indicate that muscle relaxants in general, and CBP in particular, have been widely adopted in FM despite the lack of an approval for this disorder. As FM patients do not typically experience muscle spasm, we believe that the use of muscle relaxants in FM is off-label from a regulatory perspective and provides therapeutic effects to FM patients that are different from those in treating muscle spasm. Therefore, in FM, CBP acts as a central pain inhibitor and not as a muscle relaxant.
Many FM patients take prescription pain and sleep medications to try and treat the pain, despite the lack of objective, factual evidence that such medications provide a prolonged therapeutic effect. An important goal in FM treatment is to reduce the dependence on opiate, benzodiazepine and selective non-benzodiazepine medications by FM patients. Since CBP has no recognized addictive potential, a VLD bedtime CBP product that decreases pain in FM could reduce the exposure of FM patients to ineffective off-label pain and sleep medications.
Challenges in the Market for CNS Therapies
Developers of pharmaceutical treatments for syndromes and disorders that affect the CNS face special challenges. In many cases, the causes and exacerbating factors of CNS conditions remain unknown. Frequently, key symptoms are known only by patient reports and cannot be objectively validated or measured. Symptoms like pain, fatigue, disturbed sleep or altered mood are characteristics of more than one condition. Often, physicians may not agree that a particular patient is affected by one or another condition or by more than one co-existing conditions.
| 58 |
CNS conditions are typically defined by committees of expert professionals who set criteria based on the presence of several symptoms or groups of symptoms. Sometimes groups of subjective symptoms are insufficient to describe CNS disorders and further refinement of diagnostic categories can be achieved by patient demographics, such as gender, age or concurrent medical processes, such as menopause or adolescence. Many CNS conditions, including syndromes and disorders, have not yet been characterized by laboratory tests, such as blood tests or x-ray imaging. However, laboratory tests are often important to exclude other conditions, such as inflammatory or infectious processes. Consequently, a CNS condition is sometimes called a diagnosis of exclusion because inflammation and infection should typically be ruled out by laboratory tests before applying the criteria of groups of symptoms to diagnose it.
Once a CNS condition is diagnosed, physicians may select from among treatment options based on a patient’s symptoms and history. Some medications improve or relieve only one or another symptom in a condition. Consequently, physicians may prescribe several different medications concurrently to treat individual symptoms or groups of symptoms. A desirable quality for CNS medications is the ability to relieve more than one symptom of a CNS condition. Another desirable quality for CNS medications is safety, particularly if a medicine is safe enough to be used with other medicines concurrently or at different times of the day.
Opportunity for New Treatments of FM
We believe the market for the treatment of FM is underserved, which we believe fuels a need for new therapeutic options. Due to the market acceptance of FM treatments (such as Lyrica, Cymbalta and Savella), we believe there will be a growing interest in alternative drug treatment options.
We believe that if TNX-102 SL won FDA approval, it would be an appealing option because it is believed to act by an entirely different mechanism of action from the currently approved products and we expect TNX-102 SL will be recommended for use at bedtime. Lyrica is recommended for twice or three-times daily dosing. Cymbalta was found effective at once-daily or twice-daily dosing and is generally restricted to daytime use and not recommended for bedtime use. Cymbalta and Savella act on the CNS in ways that are believed to interfere with sleep, while data support the view that CBP, the active ingredient in TNX-102 SL, improves sleep quality.
Competition
Our industry is highly competitive and subject to rapid and significant technological change. Our potential competitors include large pharmaceutical and biotechnology companies, specialty pharmaceutical and generic drug companies, academic institutions, government agencies and research institutions. We believe that key competitive factors that will affect the development and commercial success of our product candidates are efficacy, safety, tolerability, reliability, price and reimbursement level. Many of our potential competitors, including many of the organizations named below, have substantially greater financial, technical and human resources than we do and significantly greater experience in the discovery and development of product candidates, obtaining FDA and other regulatory approvals of products and the commercialization of those products. Accordingly, our competitors may be more successful than we may be in obtaining FDA approval for drugs and achieving widespread market acceptance. Our competitors’ drugs may be more effective, or more effectively marketed and sold, than any drug we may commercialize and may render our product candidates obsolete or non-competitive before we can recover the expenses of developing and commercializing any of our product candidates. We anticipate that we will face intense and increasing competition as new drugs enter the market and advanced technologies become available. Further, the development of new treatment methods for the conditions we are targeting could render our drugs non-competitive or obsolete.
The markets for medicines to treat FM, PTSD and other CNS conditions are well developed and populated with established drugs marketed by large and small pharmaceutical, biotechnology and generic drug companies. Pfizer (Lyrica), Eli Lilly (Cymbalta) and Forest Laboratories/Cyprus Biosciences (Savella) market FDA approved drugs for FM. Pfizer (Zoloft) and GlaxoSmithKline (Paxil) market FDA approved drugs for PTSD.
| 59 |
As of February 2013, we are aware of several companies developing prescription medications for FM, including Chelsea Therapeutics, Johnson and Johnson, Meda, Merck, Pfizer, RiboCor, Teva, and Theravance. Clinical trials in the U.S. are registered with the FDA and reported on the website www.clinicaltrials.gov.
A number of companies are specifically engaged in developing prescription medications for PTSD, including AstraZeneca, Biotie, Forest, GlaxoSmithKline, Marinus Pharmaceuticals, Merck, Nanotherapeutics, Johnson and Johnson, Pfizer, and UCB. Medications that may be used for the treatment of PTSD include: anti-depressants, such as nefazodone and trazodone; the antihistamine cyproheptadine; and certain atypical antipsychotics, such as olanzapine and risperidone. Several of these products are supported by companies such as AstraZeneca, GlaxoSmithKline and Pfizer.
Intellectual Property
Proprietary protection for our product candidates, technology and processes are important to our business and we seek patent protection in the U.S. and internationally when we deem appropriate. We also rely on trade secrets, know-how and continuing technological advances to protect various aspects of our core technology. We require our employees, consultants and scientific collaborators to execute confidentiality and invention assignment agreements with us.
We own numerous patents and have patent applications pending in the United States and abroad. In addition, we have one trademark application pending.
Our commercial success will depend in part on obtaining and maintaining patent protection and trade secret protection of our current and future product candidates and the methods used to manufacture them, as well as successfully defending these patents against third-party challenges. Our ability to stop third parties from making, using, selling, offering to sell or importing our products depends on the extent to which we have rights under valid and enforceable patents or trade secrets that cover these activities. We cannot assure you that our pending patent applications will result in issued patents.
Approved Patents
Our current patents owned are as follows:
Number |
Name |
Jurisdiction |
Expiration Date | |||
| 6,541,523 | “Methods For Treating Or Preventing Fibromyalgia Using Very Low Doses Of Cyclobenzaprine” | U.S.A. | August 11, 2020 | |||
| 6,395,788 | “Methods And Compositions For Treating Or Preventing Sleep Disturbances And Associated Illnesses Using Very Low Doses Of Cyclobenzaprine” | U.S.A. | August 11, 2020 | |||
| 6,358,944 | “Methods And Compositions For Treating Generalized Anxiety Disorder” | U.S.A. | August 11, 2020 | |||
| EP 1202722 | “Uses of Compositions for Treating or Preventing Sleep Disturbances Using Very Low Doses of Cyclobenzaprine” | European Patent Office, Belgium, France, Ireland, Luxembourg, Monaco, Portugal, Switzerland and United Kingdom | August 11, 2020 | |||
| AT 299369 | “Uses of Compositions for Treating or Preventing Sleep Disturbances Using Very Low Doses of Cyclobenzaprine” | Austria | August 11, 2020 | |||
| DE 60021266 | “Uses of Compositions for Treating or Preventing Sleep Disturbances Using Very Low Doses of Cyclobenzaprine” | Germany | August 11, 2020 | |||
| NZ 516749 | “Uses of Compositions for Treating or Preventing Sleep Disturbances Using Very Low Doses of Cyclobenzaprine” | New Zealand | August 11, 2020 | |||
| ES 2245944 | “Uses of Compositions for Treating or Preventing Sleep Disturbances Using Very Low Doses of Cyclobenzaprine” | Spain | August 11, 2020 | |||
| HK 1047691 | “Uses of Compositions for Treating or Preventing Sleep Disturbances Using Very Low Doses of Cyclobenzaprine” | Hong Kong | August 11, 2020 |
| 60 |
| 8,093,300 | “Compositions and Methods for Increasing Compliance with Therapies using Aldehyde Dehydrogenase Inhibitors and Treating Alcoholism” | U.S.A. | May 25, 2023 | |||
| AU 2002354017 | “Compositions and Methods for Increasing Compliance with Therapies using Aldehyde Dehydrogenase Inhibitors and Treating Alcoholism” | Australia | November 4, 2022 | |||
| CA 2463987 | “Compositions and Methods for Increasing Compliance with Therapies using Aldehyde Dehydrogenase Inhibitors and Treating Alcoholism” | Canada | November 4, 2022 | |||
| EP 1441708 | “Compositions and Methods for Increasing Compliance with Therapies using Aldehyde Dehydrogenase Inhibitors and Treating Alcoholism” | European Patent Office, Austria, Belgium, Switzerland, Denmark, Luxembourg, Monaco, Germany, France, Portugal and United Kingdom |
November 4, 2022 | |||
| NZ 532583 | “Compositions and Methods for Increasing Compliance with Therapies using Aldehyde Dehydrogenase Inhibitors and Treating Alcoholism” | New Zealand | November 4, 2022 |
Patent Applications
Our current patent applications that are pending are as follows:
| Number | Name | Jurisdiction | ||
| 61/814,664 | “Isometheptene Isomer” | U.S.A. | ||
| 61/754,281 | “Isometheptene Isomer” | U.S.A. | ||
| 61/660,593 | “Compositions and Methods for Transmucosal Absorption” | U.S.A. | ||
| 61/667,774 | “Compositions and Methods for Transmucosal Absorption” | U.S.A. | ||
| 61/725,402 | “Compositions and Methods for Transmucosal Absorption” | U.S.A. | ||
| 61/281,661 | “Methods and Compositions for Treating Symptoms Associated with Post-Traumatic Stress Disorder Using Cyclobenzaprine” | U.S.A. | ||
| 12/948,828 | “Methods And Compositions For Treating Symptoms Associated With Post-Traumatic Stress Disorder Using Cyclobenzaprine” | |||
| 10831895.7 | “Methods And Compositions For Treating Symptoms Associated With Post-Traumatic Stress Disorder Using Cyclobenzaprine” | European Patent Office | ||
| 61/449,838 | “Methods and Compositions for Treating Depression Using Cyclobenzaprine” | U.S.A. | ||
| 13/157,270 | “Method for Improving Fatigue Using Low Dose Cyclobenzaprine” | U.S.A. | ||
| PCT/US 10/02979 | “Methods And Compositions For Treating Symptoms Associated With Post-Traumatic Stress Disorder Using Cyclobenzaprine” | PCT | ||
| PCT/US 11/01529 | “Method for Treating Cocaine Addiction” | PCT | ||
| 12/151,200 | “Method For Treating Neurodegenerative Dysfunction” | U.S.A. | ||
| CA 2723688 | “Method For Treating Neurodegenerative Dysfunction” | Canada | ||
| EP 2299822 | “Method For Treating Neurodegenerative Dysfunction” | European Patent Office | ||
| 61/792,757 | “Eutectic Formulations of Cyclobenzaprine Hydrochloride and Amitriptyline Hydrochloride” | U.S.A. | ||
| 61/792,819 | “Compositions and Methods for Transmucosal Absorption” | U.S.A. | ||
| 61/792,900 | “Compositions and Methods for Transmucosal Absorption” | U.S.A. |
Trademark Application
We have one trademark application that is pending as follows:
| Number | Name | Jurisdiction | ||
| 85088881 | Tonix Pharmaceuticals | U.S.A. |
| 61 |
Research and Development
We have one employee dedicated to research and development. We anticipate that our research and development expenditures will increase several fold as we advance TNX-102 SL into late-stage clinical development and advance other candidates in our pipeline. We need to raise additional capital to fund our development plans and there is no certainty that we will be successful in continuing to attract new investments. Our research and development operations are located in New York, NY. We have used, and expect to continue to use, third parties to conduct our preclinical and clinical studies.
Manufacturing
We have contracted with third parties for the manufacture of TNX-102 SL for investigational purposes, including preclinical and clinical testing, as follows:
| CMO | Purpose | |
| Lipocine Inc. | TNX-102 gelcap used in our completed pharmacokinetic study on this candidate | |
| KABS Laboratories, Inc. (Quebec, Canada) | TNX-102 intravenous and sublingual solutions | |
| Laboratorio Farmacologico Milanese S.r.l. (Milan, Italy) | TNX-102 SL tablets used in our completed pharmacokinetic studies | |
| Pharmatek Laboratories | TNX-102 SL tablets to be used in our planned Phase 2b FM study and/or PTSD Phase 2 POC study |
All of our compounds are small molecules, synthesized using industry standard processes, and our drug products are formulated using commercially available raw materials.
Government Regulation
The FDA and other federal, state, local and foreign regulatory agencies impose substantial requirements upon the clinical development, approval, labeling, manufacture, marketing and distribution of drug products. These agencies regulate, among other things, research and development activities and the testing, approval, manufacture, quality control, safety, effectiveness, labeling, storage, record keeping, advertising and promotion of our product candidates. The regulatory approval process is generally lengthy and expensive, with no guarantee of a positive result. Moreover, failure to comply with applicable FDA or other requirements may result in civil or criminal penalties, recall or seizure of products, injunctive relief including partial or total suspension of production, or withdrawal of a product from the market.
The FDA regulates, among other things, the research, manufacture, promotion and distribution of drugs in the United States under the FDCA and other statutes and implementing regulations. The process required by the FDA before prescription drug product candidates may be marketed in the United States generally involves the following:
| · | completion of extensive nonclinical laboratory tests, animal studies and formulation studies, all performed in accordance with the FDA’s Good Laboratory Practice regulations; | |
| · | submission to the FDA of an IND, which must become effective before human clinical trials may begin; | |
| · | for some products, performance of adequate and well-controlled human clinical trials in accordance with the FDA’s regulations, including Good Clinical Practices, to establish the safety and efficacy of the product candidate for each proposed indication; | |
| · | submission to the FDA of an NDA; | |
| · | satisfactory completion of an FDA preapproval inspection of the manufacturing facilities at which the product is produced to assess compliance with cGMP regulations; and | |
| · | FDA review and approval of the NDA prior to any commercial marketing, sale or shipment of the drug. |
| 62 |
The testing and approval process requires substantial time, effort and financial resources, and we cannot be certain that any approvals for our product candidates will be granted on a timely basis, if at all.
Nonclinical tests include laboratory evaluations of product chemistry, formulation and stability, as well as studies to evaluate toxicity in animals and other animal studies. The results of nonclinical tests, together with manufacturing information and analytical data, are submitted as part of an IND to the FDA. Some nonclinical testing may continue even after an IND is submitted. The IND also includes one or more protocols for the initial clinical trial or trials and an investigator’s brochure. An IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, raises concerns or questions relating to the proposed clinical trials as outlined in the IND and places the clinical trial on a clinical hold. In such cases, the IND sponsor and the FDA must resolve any outstanding concerns or questions before any clinical trials can begin. Clinical trial holds also may be imposed at any time before or during studies due to safety concerns or non-compliance with regulatory requirements. An independent institutional review board, or IRB, at each of the clinical centers proposing to conduct the clinical trial must review and approve the plan for any clinical trial before it commences at that center. An IRB considers, among other things, whether the risks to individuals participating in the trials are minimized and are reasonable in relation to anticipated benefits. The IRB also approves the consent form signed by the trial participants and must monitor the study until completed.
Clinical Trials
Clinical trials involve the administration of the product candidate to human subjects under the supervision of qualified medical investigators according to approved protocols that detail the objectives of the study, dosing procedures, subject selection and exclusion criteria, and the parameters to be used to monitor participant safety. Each protocol for a U.S. study is submitted to the FDA as part of the IND.
Human clinical trials are typically conducted in three sequential phases, but the phases may overlap, or be combined.
| · | Phase 1 clinical trials typically involve the initial introduction of the product candidate into healthy human volunteers. In Phase 1 clinical trials, the product candidate is typically tested for safety, dosage tolerance, absorption, metabolism, distribution, excretion and pharmacodynamics. | |
| · | Phase 2 clinical trials are generally conducted in a limited patient population to gather evidence about the efficacy of the product candidate for specific, targeted indications; to determine dosage tolerance and optimal dosage; and to identify possible adverse effects and safety risks. Phase 2 clinical trials, in particular Phase 2b trials, can be undertaken to evaluate clinical efficacy and to test for safety in an expanded patient population at geographically dispersed clinical trial sites. | |
| · | Phase 3 clinical trials are undertaken to evaluate clinical efficacy and to test for safety in an expanded patient population at geographically dispersed clinical trial sites. The size of Phase 3 clinical trials depends upon clinical and statistical considerations for the product candidate and disease, but sometimes can include several thousand patients. Phase 3 clinical trials are intended to establish the overall risk-benefit ratio of the product candidate and provide an adequate basis for product labeling. |
Clinical testing must satisfy extensive FDA regulations. Reports detailing the results of the clinical trials must be submitted at least annually to the FDA and safety reports must be submitted for serious and unexpected adverse events. Success in early stage clinical trials does not assure success in later stage clinical trials. The FDA, an IRB or we may suspend a clinical trial at any time on various grounds, including a finding that the research subjects or patients are being exposed to an unacceptable health risk.
| 63 |
New Drug Applications
Assuming successful completion of the required clinical trials, the results of product development, nonclinical studies and clinical trials are submitted to the FDA as part of an NDA. An NDA also must contain extensive manufacturing information, as well as proposed labeling for the finished product. An NDA applicant must develop information about the chemistry and physical characteristics of the drug and finalize a process for manufacturing the product in accordance with cGMP. The manufacturing process must be capable of consistently producing quality product within specifications approved by the FDA. The manufacturer must develop methods for testing the quality, purity and potency of the final product. In addition, appropriate packaging must be selected and tested and stability studies must be conducted to demonstrate that the product does not undergo unacceptable deterioration over its shelf life. Prior to approval, the FDA will conduct an inspection of the manufacturing facilities to assess compliance with cGMP.
The FDA reviews all NDAs submitted before it accepts them for filing. The FDA may request additional information rather than accept an NDA for filing. In this event, the NDA must be resubmitted with the additional information and is subject to review before the FDA accepts it for filing. After an application is filed, the FDA may refer the NDA to an advisory committee for review, evaluation and recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers them carefully when making decisions. The FDA may deny approval of an NDA if the applicable regulatory criteria are not satisfied. Data obtained from clinical trials are not always conclusive and the FDA may interpret data differently than we interpret the same data. The FDA may issue a complete response letter, which may require additional clinical or other data or impose other conditions that must be met in order to secure final approval of the NDA. If a product receives regulatory approval, the approval may be significantly limited to specific diseases and dosages or the indications for use may otherwise be limited, which could restrict the commercial value of the product. In addition, the FDA may require us to conduct Phase 4 testing which involves clinical trials designed to further assess a drug’s safety and effectiveness after NDA approval, and may require surveillance programs to monitor the safety of approved products which have been commercialized. Once issued, the FDA may withdraw product approval if ongoing regulatory requirements are not met or if safety or efficacy questions are raised after the product reaches the market.
Section 505(b)(2) NDAs
There are two types of NDAs: the Section 505(b)(1) NDA, or full NDA, and the Section 505(b)(2) NDA. When possible, we intend to file Section 505(b)(2) NDAs that might, if accepted by the FDA, save time and expense in the development and testing of our product candidates. A full NDA is submitted under Section 505(b)(1) of the FDCA, and must contain full reports of investigations conducted by the applicant to demonstrate the safety and effectiveness of the drug. A Section 505(b)(2) NDA may be submitted for a drug for which one or more of the investigations relied upon by the applicant was not conducted by or for the applicant and for which the applicant has no right of reference from the person by or for whom the investigations were conducted. A Section 505(b)(2) NDA may be submitted based in whole or in part on published literature or on the FDA’s finding of safety and efficacy of one or more previously approved drugs, which are known as reference drugs. Thus, the filing of a Section 505(b)(2) NDA may result in approval of a drug based on fewer clinical or nonclinical studies than would be required under a full NDA. The number and size of studies that need to be conducted by the sponsor depends on the amount and quality of data pertaining to the reference drug that are publicly available, and on the similarity of and differences between the applicant’s drug and the reference drug. In some cases, extensive, time-consuming, and costly clinical and nonclinical studies may still be required for approval of a Section 505(b)(2) NDA.
Because we are developing new formulations of previously approved chemical entities, such as CBP, our drug approval strategy is to submit Section 505(b)(2) NDAs to the FDA. The FDA may not agree that our product candidates are approvable as Section 505(b)(2) NDAs. If the FDA determines that Section 505(b)(2) NDAs are not appropriate and that full NDAs are required for our product candidates, the time and financial resources required to obtain FDA approval for our product candidates could substantially and materially increase, and our products might be less likely to be approved. If the FDA requires full NDAs for our product candidates, or requires more extensive testing and development for some other reason, our ability to compete with alternative products that arrive on the market more quickly than our product candidates would be adversely impacted. If CBP-containing products are withdrawn from the market by the FDA for any reason, we may not be able to reference such products to support our anticipated TNX-102 SL 505(b)(2) NDA, and we may be required to follow the requirements of Section 505(b)(1).
| 64 |
Based on our intent to file under Section 505(b)(2) with respect to our lead product candidate, we believe it is unlikely the development process for this product candidate will follow the ordinary course of Phase 1, Phase 2 and Phase 3 studies. Our human pharmacokinetic studies of reformulated CBP dosage forms represented the first use of TNX-102 SL and TNX-102 gelcap, or collectively, TNX-102, in humans and could therefore be described as “Phase 1.” However, because these studies compared TNX-102 to existing approved formulations of CBP and specified the comparable ability to deliver effective levels of CBP to the bloodstream of FM patients, these studies provide a reference to the therapeutic effects previously observed in our dose-ranging clinical study of CBP IR capsules in FM patients. For these reasons, rather than always identifying clinical trials by Phase, we find it more illustrative to describe in a narrative form the purpose of the studies and the nature and potential significance of the results. Because our double-blind, randomized, placebo-controlled, dose-ranging study on bedtime CBP was performed in Canada, we did not meet with the FDA’s Center for Drug Evaluation and Research to discuss our approach and plans until August 2011. In February 2013, we held an End-of-Phase 2/Pre-Phase 3 meeting with the FDA to discuss the clinical and nonclinical requirements to register TNX-102 SL for the management of FM based on the 505(b)(2) regulatory pathway.
Patent Protections
An applicant submitting a Section 505(b)(2) NDA must certify to the FDA with respect to the patent status of the reference drug upon which the applicant relies in support of approval of its drug. With respect to every patent listed in the FDA’s Orange Book, which is the FDA’s list of approved drug products, as claiming the reference drug or an approved method of use of the reference drug, the Section 505(b)(2) applicant must certify that: (1) there is no patent information listed by the FDA for the reference drug; (2) the listed patent has expired; (3) the listed patent has not expired, but will expire on a particular date; (4) the listed patent is invalid, unenforceable, or will not be infringed by the manufacture, use, or sale of the product in the Section 505(b)(2) NDA; or (5) if the patent is a use patent, that the applicant does not seek approval for a use claimed by the patent. If the applicant files a certification to the effect of clause (1), (2) or (5), FDA approval of the Section 505(b)(2) NDA may be made effective immediately upon successful FDA review of the application, in the absence of marketing exclusivity delays, which are discussed below. If the applicant files a certification to the effect of clause (3), the Section 505(b)(2) NDA approval may not be made effective until the expiration of the relevant patent and the expiration of any marketing exclusivity delays.
If the Section 505(b)(2) NDA applicant provides a certification to the effect of clause (4), referred to as a paragraph IV certification, the applicant also must send notice of the certification to the patent owner and the holder of the NDA for the reference drug. The filing of a patent infringement lawsuit within 45 days of the receipt of the notification may prevent the FDA from approving the Section 505(b)(2) NDA for 30 months from the date of the receipt of the notification unless the court determines that a longer or shorter period is appropriate because either party to the action failed to reasonably cooperate in expediting the action. However, the FDA may approve the Section 505(b)(2) NDA before the 30 months have expired if a court decides that the patent is invalid, unenforceable, or not infringed, or if a court enters a settlement order or consent decree stating the patent is invalid or not infringed.
Notwithstanding the approval of many products by the FDA pursuant to Section 505(b)(2), over the last few years certain brand-name pharmaceutical companies and others have objected to the FDA’s interpretation of Section 505(b)(2). If the FDA’s interpretation of Section 505(b)(2) is successfully challenged in court, the FDA may be required to change its interpretation of Section 505(b)(2) which could delay or even prevent the FDA from approving any Section 505(b)(2) NDA that we submit. The pharmaceutical industry is highly competitive, and it is not uncommon for a manufacturer of an approved product to file a citizen petition with the FDA seeking to delay approval of, or impose additional approval requirements for, pending competing products. If successful, such petitions can significantly delay, or even prevent, the approval of the new product. Moreover, even if the FDA ultimately denies such a petition, the FDA may substantially delay approval while it considers and responds to the petition.
| 65 |
Marketing Exclusivity
Market exclusivity provisions under the FDCA can delay the submission or the approval of Section 505(b)(2) NDAs, thereby delaying a Section 505(b)(2) product from entering the market. The FDCA provides five-year marketing exclusivity to the first applicant to gain approval of an NDA for a new chemical entity, or NCE, meaning that the FDA has not previously approved any other drug containing the same active moiety. This exclusivity prohibits the submission of a Section 505(b)(2) NDA for any drug product containing the active ingredient during the five-year exclusivity period. However, submission of a Section 505(b)(2) NDA that certifies that a listed patent is invalid, unenforceable, or will not be infringed, as discussed above, is permitted after four years, but if a patent infringement lawsuit is brought within 45 days after such certification, FDA approval of the Section 505(b)(2) NDA may automatically be stayed until 7 1/2 years after the NCE approval date. The FDCA also provides three years of marketing exclusivity for the approval of new and supplemental NDAs for product changes, including, among other things, new indications, dosage forms, routes of administration or strengths of an existing drug, or for a new use, if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by FDA to be essential to the approval of the application. Five-year and three-year exclusivity will not delay the submission or approval of another full NDA; however, as discussed above, an applicant submitting a full NDA under Section 505(b)(1) would be required to conduct or obtain a right of reference to all of the preclinical and adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
Other types of exclusivity in the United States include orphan drug exclusivity and pediatric exclusivity. The FDA may grant orphan drug designation to a drug intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making available in the United States a drug for this type of disease or condition will be recovered from sales in the United States for that drug. Seven-year orphan drug exclusivity is available to a product that has orphan drug designation and that receives the first FDA approval for the indication for which the drug has such designation. Orphan drug exclusivity prevents approval of another application for the same drug for the same orphan indication, for a period of seven years, regardless of whether the application is a full NDA or a Section 505(b)(2) NDA, except in limited circumstances, such as a showing of clinical superiority to the product with orphan exclusivity. Pediatric exclusivity, if granted, provides an additional six months to an existing exclusivity or statutory delay in approval resulting from a patent certification. This six-month exclusivity, which runs from the end of other exclusivity protection or patent delay, may be granted based on the voluntary completion of a pediatric study in accordance with an FDA-issued “Written Request” for such a study.
Section 505(b)(2) NDAs are similar to full NDAs filed under Section 505(b)(1) in that they are entitled to any of these forms of exclusivity if they meet the qualifying criteria. They also are entitled to the patent protections described above, based on patents that are listed in the FDA’s Orange Book in the same manner as patents claiming drugs and uses approved for NDAs submitted as full NDAs.
Other Regulatory Requirements
Maintaining substantial compliance with appropriate federal, state and local statutes and regulations requires the expenditure of substantial time and financial resources. Drug manufacturers are required to register their establishments with the FDA and certain state agencies, and after approval, the FDA and these state agencies conduct periodic unannounced inspections to ensure continued compliance with ongoing regulatory requirements, including cGMPs. In addition, after approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further FDA review and approval. The FDA may require post-approval testing and surveillance programs to monitor safety and the effectiveness of approved products that have been commercialized. Any drug products manufactured or distributed by us pursuant to FDA approvals are subject to continuing regulation by the FDA, including:
| · | record-keeping requirements; |
| · | reporting of adverse experiences with the drug; |
| · | providing the FDA with updated safety and efficacy information; |
| · | reporting on advertisements and promotional labeling; |
| · | drug sampling and distribution requirements; and |
| · | complying with electronic record and signature requirements. |
| 66 |
In addition, the FDA strictly regulates labeling, advertising, promotion and other types of information on products that are placed on the market. There are numerous regulations and policies that govern various means for disseminating information to health-care professionals as well as consumers, including to industry sponsored scientific and educational activities, information provided to the media and information provided over the Internet. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label.
The FDA has very broad enforcement authority and the failure to comply with applicable regulatory requirements can result in administrative or judicial sanctions being imposed on us or on the manufacturers and distributors of our approved products, including warning letters, refusals of government contracts, clinical holds, civil penalties, injunctions, restitution and disgorgement of profits, recall or seizure of products, total or partial suspension of production or distribution, withdrawal of approvals, refusal to approve pending applications, and criminal prosecution resulting in fines and incarceration. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability. In addition, even after regulatory approval is obtained, later discovery of previously unknown problems with a product may result in restrictions on the product or even complete withdrawal of the product from the market.
Food and Drug Administration Amendments Act of 2007
In September 2007, the Food and Drug Administration Amendments Act of 2007, or FDAAA, became law. This legislation grants significant new powers to the FDA, many of which are aimed at improving drug safety and assuring the safety of drug products after approval. In particular, the new law authorizes the FDA to, among other things, require post-approval studies and clinical trials, mandate changes to drug labeling to reflect new safety information, and require risk evaluation and mitigation strategies for certain drugs, including certain currently approved drugs. In addition, the new law significantly expands the federal government’s clinical trial registry and results databank and creates new restrictions on the advertising and promotion of drug products. Under the FDAAA, companies that violate these and other provisions of the new law are subject to substantial civil monetary penalties.
The FDA has not yet implemented many of the provisions of the FDAAA, so we cannot predict the impact of the new legislation on the pharmaceutical industry or our business. However, the requirements and changes imposed by the FDAAA may make it more difficult, and more costly, to obtain and maintain approval for new pharmaceutical products, or to produce, market and distribute existing products. In addition, the FDA’s regulations, policies and guidance are often revised or reinterpreted by the agency or the courts in ways that may significantly affect our business and our products. It is impossible to predict whether additional legislative changes will be enacted, or FDA regulations, guidance or interpretations changed, or what the impact of such changes, if any, may be.
Employees
As of May 9, 2013, we had two full-time employees, Leland Gershell, our Chief Financial Officer, and Bruce Daugherty, our Senior Director of Drug Development and Controller, as well as one part-time senior director of research.
In addition, we rely on consultants instead of employees for critical activities, including Seth Lederman who serves as our Chief Executive Officer and as President of Tonix Sub pursuant to a consulting agreement with Lederman & Co. None of our employees are represented by a labor union, and we believe that our relations with our employees are good.
DESCRIPTION OF PROPERTIES
We maintain our principal office at 509 Madison Avenue, Suite 306, New York, New York 10022. Our telephone number at that office is (212) 980-9155 and our fax number is (212) 923-5700. Our current office space consists of approximately 2,355 square feet. The lease expires in September 2015. The base rent is as follows:
| Lease Period | Amount Per Annum | |||
| October 1, 2010 – September 30, 2011 | $ | 120,105.00 | ||
| October 1, 2011 – September 30, 2012 | $ | 123.496.20 | ||
| October 1, 2012 – September 30, 2013 | $ | 126,989.14 | ||
| October 1, 2013 – September 30, 2014 | $ | 130,586.86 | ||
| October 1, 2014 – September 30, 2015 | $ | 134,292.52 | ||
We believe that our existing facilities are suitable and adequate to meet our current business requirements. We maintain websites at www.tonixpharma.com and www.krele.com and the information contained on those websites is not deemed to be a part of this prospectus.
| 67 |
LEGAL PROCEEDINGS
From time to time, we may become involved in various lawsuits and legal proceedings which arise in the ordinary course of business. However, litigation is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may harm our business. We are currently not aware of any such legal proceedings or claims that we believe will have, individually or in the aggregate, a material adverse effect on our business, financial condition or operating results.
| 68 |
MANAGEMENT
The names of our executive officers and directors and their age, title, and biography as of May 9, 2013 are set forth below:
| Name | Age | Title | ||
| Seth Lederman | 55 | President, CEO and Chairman of the Board of Directors | ||
| Leland Gershell | 41 | Chief Financial Officer and Treasurer | ||
| Bruce Daugherty | 56 | Senior Director of Drug Development, Controller and Secretary | ||
| Stuart Davidson | 56 | Director | ||
| Patrick Grace | 57 | Director | ||
| Donald W. Landry | 58 | Director | ||
| Ernest Mario | 74 | Director | ||
| Charles E. Mather IV | 53 | Director | ||
| John Rhodes | 56 | Director | ||
| Samuel Saks | 58 | Director |
Directors are elected annually and hold office until the next annual meeting of the shareholders of the Company and until their successors are elected. Officers are elected annually and serve at the discretion of the Board of Directors.
Seth Lederman, MD became our President, Chief Executive Officer, Chairman of the Board and a Director in October 2011. Dr. Lederman founded Tonix Sub in June of 2007 and has acted as its Chairman of the Board of Directors since inception and as President since June 2010. Dr. Lederman has been the Chairman of Krele since its inception in August 2010. Dr. Lederman has also been the President and a director of Tonix Pharmaceuticals (Canada), Inc. since its inception in April 2013. Since 1996, Dr. Lederman has been an Associate Professor at Columbia University. As an Assistant Professor at Columbia, Dr. Lederman discovered and characterized the CD40-ligand and invented therapeutic candidates to treat autoimmune diseases and transplant rejection. Dr. Lederman has been a Manager of L&L since 1996. In addition, Dr. Lederman has been the Managing Member of Seth Lederman Co, LLC since January 2007 and the Managing Member of Lederman & Co since 2002, both of which are biopharmaceutical consulting and investing companies. Dr. Lederman has also been the Managing Member of Targent since 2000, and Managing Member of Plumbline since 2002. Targent was a founder of Targent Pharmaceuticals Inc. on which Board of Directors Dr. Lederman served from inception in 2001 until the sale of its assets to Spectrum Pharmaceuticals Inc. in 2006. Between January 2007 and November 2008, Dr. Lederman was a Managing Partner of Konanda Pharma Partners, LLC, a Director of Konanda Pharma Fund I, LP, and a Managing Partner of Konanda General Partner, LLC, which were related private growth equity fund entities. As well, between January 2007 and November 2008, Dr. Lederman was Chairman of Validus Pharmaceuticals, Inc. and Fontus Pharmaceuticals, Inc., which were portfolio companies of the Konanda private growth equity fund. Since December 2011, Dr. Lederman has served as CEO and Chairman of Leder Laboratories Inc. and Starling Pharmaceuticals Inc, which are biopharmaceutical development companies. Since March 2013, Dr. Lederman has been the chairman of Leder Laboratories, Ltd., a wholly-owned subsidiary of Leder Laboratories Inc. Between 2006 and 2011, Dr. Lederman was a director of Research Corporation, a New York-based non-profit organization. Dr. Lederman received his BA degree in Chemistry from Princeton University in 1979 and his MD from Columbia University in 1983. Dr. Lederman has been a New York State licensed physician since 1985. Dr. Lederman’s significant experience with our patent portfolio and his experience as an entrepreneur, seed capital investor, fund manager, and director of start-up biopharmaceutical companies were instrumental in his selection as a member of the board of directors.
| 69 |
Leland Gershell, MD PhD became our Chief Financial Officer in April 2012 and our Treasurer in November 2012. From May 2011 to December 2011, Dr. Gershell was Managing Director and Senior Analyst at Madison Williams and Company, where he was responsible for equity research coverage of specialty pharmaceutical and biotechnology companies. From April 2010 to October 2010, Dr. Gershell was Senior Analyst at Favus Institutional Research, where he was responsible for issuing research reports on a variety of healthcare companies to institutional investors. From October 2008 to October 2009, Dr. Gershell was Senior Analyst at Apothecary Capital, a healthcare investment firm. From November 2004 to September 2008, Dr. Gershell was an equity research analyst at Cowen and Company, most recently as Vice President, where he was responsible for the equity research coverage of small and middle capitalization biotechnology companies. Dr. Gershell earned his M.D. and Ph.D. in Organic Chemistry from Columbia University and his B.A. magna cum laude in Chemistry and Asian Studies from Dartmouth College. Dr. Gershell is an inventor on Columbia’s patents for SAHA/vorinostat, which is marketed by Merck as Zolinza® and is the first histone deacetylase (HDAC) inhibitor to receive FDA approval.
Bruce Daugherty, PhD became our Senior Director of Drug Development and Controller in April 2012 and our Secretary in November 2012. Dr. Daugherty has also been the Secretary and a director of Tonix Pharmaceuticals (Canada), Inc. since its inception in April 2013. Since January 2009, Dr. Daugherty has worked as a consultant to academia and biotechnology companies in drug discovery/development and licensing through his consulting company, LeClair Pharma Consulting, LLC. Dr. Daugherty was a consultant to our company between November 2011 and March 2012. In 2009, Dr. Daugherty was employed at Assumption College in Mendham, New Jersey, where he was a lecturer in Biology for freshman students. From 1987 to 2008, Dr. Daugherty was employed at Merck & Co., where he was a scientist in drug discovery and development. Dr. Daugherty earned his MBA from Emory University’s Goizueta Business School, his PhD in Molecular Genetics and Microbiology from UMDNJ-Robert Wood Johnson Medical School, his MS in Zoology from Rutgers University and his BA in Biology from Washington University in St. Louis.
Stuart Davidson became a Director in October 2011. Between July 2010 and October 2011, Mr. Davidson served as a director of Tonix Sub. Since 2011, Mr. Davidson has been a Managing Director of Sonen Capital. Since 1994, Mr. Davidson has been a Managing Partner of Labrador Ventures. Prior to Labrador, Mr. Davidson founded and served as CEO of Combion, Inc., which was acquired by Incyte. He also served as President of Alkermes, Inc., a biotechnology company focused on drug delivery. Mr. Davidson received his Bachelor’s Degree from Harvard College in 1978 and his MBA from Harvard Business School in 1984. Mr. Davidson’s prior experience as a venture capital investor, entrepreneur, and biotechnology industry executive experience leading pharmaceutical companies was instrumental in his selection as a member of our board of directors.
Patrick Grace became a Director in October 2011. Between June 2007 and October 2011, Mr. Grace served as a director of Tonix Sub. Mr. Grace was the co-founder of and has served as the Managing Partner of Apollo Philanthropy Partners, L.L.C. since October 2008. He has also been President of MLP Capital, Inc., New York, New York, an investment holding company, since 1996. Mr. Grace served in various senior management roles with W. R. Grace & Co. from 1977-1995, and was last President and CEO of Grace Logistics Services, Inc. From January 2002 to August 2002, Mr. Grace was also President and Chief Executive Officer of Kingdom Group, LLC (“Kingdom”), New York, New York (a provider of turnkey compressed natural gas fueling systems), and he was Executive Vice President of Kingdom from August 1999 to December 2000. Since 1996, he has been a director of Chemed Corporation. Mr. Grace was a liberal arts major at the University of Notre Dame and earned a MBA in finance from Columbia University. Mr. Grace’s extensive executive experience, along with his membership on the board of directors of a public company was instrumental in his selection as a member of our board of directors.
Donald W. Landry, MD, PhD became a Director in October 2011. Between June 2007 and October 2011, Dr. Landry served as a director of Tonix Sub. Dr. Landry has been a member of the faculty of Columbia University since 1986, and has served as the Samuel Bard Professor of Medicine, Chair of the Department of Medicine and Physician-in-Chief at New York Presbyterian Hospital/Columbia University since 2008. Dr. Landry was a co-founder and has been a member of L&L since 1996. Dr. Landry received his BS degree in Chemistry from Lafayette College in 1975, his PhD in Organic Chemistry from Harvard University in 1979 and his M.D. from Columbia University in 1983. Dr. Landry has been a New York State licensed physician since 1985. In 2008, Dr. Landry was awarded the Presidential Citizens Medal, the second-highest award that the President can confer upon a civilian. Dr. Landry’s significant medical and scientific background was instrumental in his selection as a member of the board of directors.
| 70 |
Ernest Mario, PhD became a Director in October 2011. Between September 2010 and October 2011, Dr. Mario served as a director of Tonix Sub. Dr. Mario is a former Deputy Chairman and Chief Executive of Glaxo Holdings plc and a former Chairman and Chief Executive Officer of ALZA Corporation. Since August 2007, Dr. Mario has served as the Chief Executive Officer and Chairman of Capnia, Inc., a privately held specialty pharmaceutical company in Palo Alto, CA. From 2003 to 2007, he was Chairman and Chief Executive of Reliant Pharmaceuticals, Inc. Dr. Mario is currently a Director of Boston Scientific Corp. (since 2001), Celgene Corp. (since 2007), Chimerix, Inc. (since February 2013), Maxygen Inc. (since 2001), VIVUS Inc. (since 2012) and XenoPort Inc. (since 2012). Dr. Mario is also Chairman of Chimerix. He is Chairman of the American Foundation for Pharmaceutical Education and serves as an advisor to The Ernest Mario School of Pharmacy at Rutgers University. In 2007, Dr. Mario was awarded the Remington Medal by the American Pharmacists’ Association, pharmacy’s highest honor. Dr. Mario received a PhD and an MS in physical sciences from the University of Rhode Island and a BS in pharmacy from Rutgers University. Dr. Mario brings to his service as a director his significant executive leadership experience, including his experience leading several pharmaceutical companies, as well as his membership on public company boards and foundations. He also has extensive experience in financial and operations management, risk oversight, and quality and business strategy.
Charles E. Mather IV became a Director in October 2011. Between April and October 2011, Mr. Mather served as a director of Tonix Sub. Mr. Mather has been the Head of Private and Alternative Capital and Co-Head of ECM at Janney Montgomery Scott since December 2009. Between October 2008 and December 2009, Mr. Mather served as an independent consultant to various securities firms. Between May 2007 and September 2008, Mr. Mather was the head of the Structured Equity Group at Jefferies Group Inc. Prior to that, Mr. Mather held various senior investment banking positions at Cowen and Company, including as Co-Head of the Private Equity Group. Mr. Mather received a BA in History from Brown University and an MBA in Finance from The Wharton School, University of Pennsylvania. Mr. Mather’s extensive experience as an investment banker was instrumental in his selection as a member of our board of directors.
John Rhodes became a Director in October 2011. Mr. Rhodes has served as director of the Center for Market Innovation at Natural Resources Defense Council since January 2012. Between October 2010 and October 2011, Mr. Rhodes served as a director of Tonix Sub. Mr. Rhodes has been a director of Dewey Electronics Company, a manufacturer of electronic and electromechanical systems for the military and commercial markets, since 2005. Between April 2007 and June 2010, Mr. Rhodes was a Senior Advisor to Good Energies, Inc., a renewable energy company. Mr. Rhodes is a former Vice President of Booz Allen Hamilton, Inc. Mr. Rhodes is a graduate of Princeton University and the Yale School of Management. Mr. Rhodes’ extensive business and consulting experience, along with his membership on the board of directors of a public company was instrumental in his selection as a member of our board of directors.
Samuel Saks, MD became a Director in May 2012. Between 2003 and April 2009, Dr. Saks was the chief executive officer and a director of Jazz Pharmaceuticals, Inc., a publicly-held biopharmaceutical company, which he co-founded in 2003. From April 2011 until February 2012, Dr. Saks served as interim Chief Medical Officer of Threshold Pharmaceuticals, a publicly-held biopharmaceutical company. From 2001 until 2003, Dr. Saks was company group chairman of ALZA Corporation and a member of the Johnson & Johnson Pharmaceuticals Operating Committee. From 1992 until 2001, Dr. Saks held various positions at ALZA, including Chief Medical Officer and Group Vice President, where he was responsible for clinical, regulatory and commercial activities. Previously, Dr. Saks held clinical research and development management positions with Schering-Plough, Xoma and Genentech. Dr. Saks formerly served as a scientific advisor to ArQule Pharmaceuticals, CMEA Ventures and ProQuest Investments. Dr. Saks is currently a Director of Auspex Pharmaceuticals, Inc. (since 2009), Depomed (since 2012), Bullet Biotechnology, Inc. (since 2012) and Velocity Pharmaceutical Development LLC (since 2011). From September 2005 until October 2010, Dr. Saks served on the board of directors of Trubion Pharmaceuticals, a publicly-held biopharmaceutical company. Between September 2007 and July 2009, Dr. Saks served on the board of directors of Cougar Biotechnology, a publicly-held biopharmaceutical company. Dr. Saks has also served on the board of directors of Corixa, Coulter and Ribozyme. Dr. Saks is board certified in oncology and received a B.S. and an M.D. from the University of Illinois. Mr. Saks’ extensive scientific and medical expertise and experience in formulating partnering and business development strategies, including those involving larger pharmaceutical companies, was instrumental in his selection as a member of our board of directors.
| 71 |
Scientific and Medical Advisory Network
We have an established advisory team and network of well-recognized scientists, clinicians and opinion leaders in the fields of FM and PTSD. Depending on their expertise, these advisors provide assistance in trial design, conduct clinical trials, keep us apprised of the latest scientific advances and help us identify and evaluate business development opportunities. A number of the members of this network form our Scientific Advisory Board. We consult with members of our Scientific Advisory Board on an as-needed basis to identify pipeline opportunities, review in vitro data and the design of preclinical and clinical studies. The members of our Scientific Advisory Board are:
Daniel Goodman, MD
Dr. Goodman is a psychiatrist and scientist entrepreneur. Dr. Goodman was co-founder and CEO of PsychoGenics, a leading preclinical neuropharmacology company that combines in vivo behavioral expertise with robotics, computer vision, and bio/cheminformatics to evaluate drug candidates for utility across the spectrum of neuropsychiatric disease indications. Using its unique technology platform, PsychoGenics identifies new therapeutic potentials for novel, discontinued, and early stage compounds. Dr. Goodman was also co-founder and President of Resolvyx Pharmaceuticals, a biopharmaceutical company dedicated to the discovery, development, and commercialization of resolvins, a novel class of therapies to treat inflammatory diseases. Dr. Goodman’s ongoing research interests span a broad spectrum, from the design and development of novel small molecule compounds for neuropsychiatric and inflammatory diseases to the repurposing of existing drugs for new neuropsychiatric indications. In addition to his scientific interests, Dr. Goodman is a practicing physician with a private psychiatry and psychopharmacology practice in New York and Greenwich, CT, where he treats patients with cognitive, mood, and anxiety disorders. Dr. Goodman also maintains an academic appointment at Cornell University Medical College. He is a graduate of Yale College, Cambridge University, which he attended as a Churchill Fellow, Harvard Medical School, and Columbia Business School.
Herbert Harris, MD, PhD
Since August 2012, Dr. Harris has served as Medical Director at Rho, Inc., a Chapel Hill, North Carolina based clinical contract research organization. Prior to Rho, Dr. Harris served as Medical Director of Welcare Counseling Services in North Carolina and as a consultant to pharmaceutical companies and venture capital firms. Dr. Harris served as Chief Medical Officer (CMO) at Tonix Sub from July 2009 until joining Welcare in September 2010. Prior to Tonix Sub, Dr. Harris served as CMO of Validus Pharmaceuticals where he was responsible for drug safety, clinical research and development, regulatory affairs, medical affairs, and compliance for several marketed products. Dr. Harris has more than a decade of experience in drug development at Jazz Pharmaceuticals, GSK, Vela Pharmaceuticals, Cephalon and Merck. Dr. Harris served as CMO of Vela. His experience includes several international phase 3 programs in schizophrenia, depression, and fibromyalgia as well as phase 1, phase 2, and preclinical research in various CNS-related therapeutic areas. Prior to joining the industry, Dr. Harris served as Chief of the Geriatric Psychopharmacology Program at the National Institute of Mental Health (NIMH). While at NIMH, he contributed to several major public health initiatives in mental health research including the Surgeon General’s Report on Mental Health, DSM-IV-TR, and the STAR*D study of treatment-resistant depression. He also served as scientific liaison to the Neuropharmacology Division of the FDA. Dr. Harris received his MD and PhD from the University of Pittsburgh, and his psychiatric training at Yale University. After completing his clinical training, he served as Senior Staff Fellow at the National Institute on Aging, where his research focused on the molecular biology of Alzheimer’s disease.
Iredell W. Iglehart, III, MD
Dr. Iglehart is an internist and rheumatologist in solo private practice in Baltimore, MD and Assistant Professor, part-time in rheumatology at Johns Hopkins University School of Medicine. Dr. Iglehart is the inventor of our patented technology using VLD CBP at bedtime to treat FM. Dr. Iglehart is former President of the Johns Hopkins Alumni Association and in that capacity served on the Johns Hopkins University Board of Trustees under Chairman Michael Bloomberg for four years. In private practice for over 22 years, he spends more time with fewer patients, and still makes neighborhood house-calls. Dr. Iglehart’s discovery of VLD CBP’s effects was motivated by his appreciation of the importance of sleep quality in both his general medical and rheumatologic patients. Dr. Iglehart graduated Princeton University cum laude in 1979 and earned his M.D. at the Johns Hopkins University School of Medicine in 1983. He completed his residency in internal medicine and fellowship in rheumatology at Johns Hopkins.
| 72 |
Harvey Moldofsky, M.D., Dip. Psych., F.R.C.P.(C)
Dr. Moldofsky is the Director of Research of the Centre for Sleep and Chronobiology Inc. and President of the Toronto Psychiatric Research Foundation. He is the past President and Medical Director of the Sleep Disorders Clinics of the Center for Sleep and Chronobiology, an honorary member of the University Health Network Department of Psychiatry and consults for several Toronto hospitals and government agencies. Dr. Moldofsky has devoted his career to studying sleep physiology and biologic rhythms. He is renowned for work in FM and particularly the relationships between chronic musculoskeletal pain, fatigue, and non-restorative sleep. In 2001, Dr. Moldofsky was principal investigator of a study that tested the effect of bedtime administration of VLD CBP on symptoms in FM patients, which is the proof-of-principle study for TNX-102. Dr. Moldofsky was formerly Professor of Psychiatry and Medicine and is currently Professor Emeritus of the Dept. of Psychiatry, Faculty of Medicine and Member Emeritus, Institute of Medical Science, School of Graduate Studies at the University of Toronto. From 1993-2000 he served as founding Director of the University of Toronto’s Center for Sleep and Chronobiology, and from 1998-2003 he was a medical assessor for the Workplace Safety and Insurance Appeals Tribunal of Ontario. Dr. Moldofsky has been the recipient of many local, national and international awards, including the regional award of the Royal College of Physicians and Surgeons of Canada for his contributions as a medical educator. In 1989, The Dr. Harvey Moldofsky Scholarship for Psychiatric/Neuroscience Research was formed in his honor. It is awarded annually to medical students at the University of Toronto. Dr. Moldofsky earned his MD from the University of Toronto in 1959 and was engaged in postgraduate training in Vancouver, Toronto, London and San Francisco. He is a Distinguished Life Fellow of the American Psychiatric Association.
Gregory M. Sullivan, MD
Dr. Sullivan is a physician and scientist specializing in somatic treatments of psychiatric disorders. He is an Assistant Professor of Clinical Psychiatry in the Department of Psychiatry at Columbia University (CU). Dr. Sullivan’s research focuses on the neurobiology and treatment of affective and anxiety disorders, and includes work elucidating dysfunctional neurocircuitry in PTSD. Much of his work employs fully quantitative neuroreceptor mapping using positron emission tomography brain imaging technology as well as functional magnetic resonance imaging. His work during an NIMH Mentored Clinical Scientist Development Award has led to several important findings on the role of the serotonin-1A receptor in both mood and anxiety disorders. Dr. Sullivan is also a Research Scientist of the New York State Psychiatric Institute (NYSPI), and he serves as a member of the IRB of NYSPI and the CU Department of Psychiatry. Dr. Sullivan graduated from the University of California, Berkeley where he majored in biological sciences. He received his MD from CU’s College of Physicians & Surgeons, and he also attended CU for residency training in psychiatry. After residency, he continued at CU in an NIMH-sponsored Research Training Fellowship in Affective and Anxiety Disorders. As part of fellowship training, he spent substantial time in the laboratory of Dr. Joseph LeDoux at New York University, developing translational animal models of anxiety disorders for preclinical testing of novel anxiolytic therapies.
Significant Consultants
In addition to our SAB, from time to time we engage consultants to provide additional, specialized advice and guidance. Two of these significant consultants are:
Daniel Clauw, MD
Dr. Clauw is serving as the Principal Investigator of Tonix’s Phase 2b pivotal clinical trial of TNX-102 in FM. Dr. Clauw is a world renowned expert on FM, having authored over 200 scientific papers as well as a number of book chapters. Dr. Clauw is a Professor of Anesthesiology and Medicine (in the Division of Rheumatology) at the University of Michigan. He serves as Director of the Chronic Pain and Fatigue Research Center. Until 2009, he also served as the Director of the Michigan Institute for Clinical and Health Research and Associate Dean for Clinical and Translational Research within the University of Michigan Medical School.
| 73 |
Since moving to the University of Michigan in 2001, Dr. Clauw has continued his commitment to the clinical care and research into FM and overlapping conditions such as chronic fatigue syndrome, interstitial cystitis and Gulf War illnesses. He leads a multidisciplinary team of collaborators who are studying the mechanisms and most effective treatments of these illnesses.
Dr. Clauw served as Chairman of the Scientific Advisory Board of Cypress Bioscience Inc. from 2001 to 2009. Cypress developed milnacipran for FM and it is marketed by Forest Laboratories as Savella. Since 2006, he has served on the Pain/Fibromyalgia Scientific Advisory Boards, of Pfizer, Lilly, Wyeth, Forest Pharmaceuticals, and UCB. Dr. Clauw joined the faculty at Georgetown University in 1990, and while there, served as the Scientific Director of the Georgetown Chronic Pain and Fatigue Research Center, the Division Chief of Rheumatology, Immunology and Allergy, and a Vice Chair of the Department of Medicine. Dr. Clauw attended the University of Michigan for both undergraduate and medical school studies, before completing his Internal Medicine residency and Rheumatology Fellowship at Georgetown University.
R. Michael Gendreau, MD, PhD
Dr. Gendreau is Managing Director of Gendreau Consulting, LLC and provides consultation to Tonix on clinical trial design and conduct as well as the selection of clinical trial sites and investigators. Before Gendreau Consulting, Dr. Gendreau served as Chief Medical Officer of Cypress Bioscience Inc. from 1995 to 2011. In that capacity, Dr. Gendreau led the development of milnacipran for FM, starting with IND development, design and implementation of the Phase 2 proof of concept trial, design of the Phase 3 program, co-managing multiple Phase 3 trials and NDA preparation along with commercial partner Forest Laboratories. Savella received first round regulatory approval for FM in 2009. In that program, Dr. Gendreau pioneered use of electronic diaries to characterize FM pain in the home setting, defined the regulatory course for FM approval, and won approval for a special protocol assessment for the milnacipran Phase 3 program. Prior to Cypress, Dr. Gendreau served as Chief Medical Officer of Microprobe Corporation (1991-94) and as executive Vice President for Research and Development at Source Scientific Systems (1988-91) until it acquired Microprobe. Dr. Gendreau served in a variety of positions at Battelle Memorial Institute of Columbus, OH (1977-88).
Dr. Gendreau serves as an officer of Cerephex Corporation, a private neurostimulation technology company. Dr. Gendreau received his MD and PhD degrees from Ohio State University and his undergraduate BS from Ohio University.
Family Relationships
None.
Board Independence
We are not required to have any independent members of the Board of Directors. The board of directors has determined that (i) Seth Lederman, has a relationship which, in the opinion of the board of directors, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director and is not an “independent director” as defined in the Marketplace Rules of The NASDAQ Stock Market and (ii) Stuart Davidson, Patrick Grace, Donald Landry, Ernest Mario, Charles Mather, John Rhodes and Samuel Saks are each an independent director as defined in the Marketplace Rules of The NASDAQ Stock Market.
Meetings and Committees of the Board of Directors
During the fiscal year ended December 31, 2012, our board of directors held five meetings and approved certain actions by unanimous written consent. We expect our directors to attend all board and committee meetings and to spend the time needed and meet as frequently as necessary to properly discharge their responsibilities.
Audit Committee
Our Audit Committee consists of Patrick Grace, Charles Mather and John Rhodes, with Mr. Grace elected as Chairman of the Committee. Our Board of Directors has determined that each of Messrs. Grace, Mather and Rhodes are “independent” as that term is defined under applicable SEC rules and under the current listing standards of the NASDAQ Stock Market. Mr. Grace is our audit committee financial expert.
| 74 |
Our Audit Committee’s responsibilities include: (i) reviewing the independence, qualifications, services, fees, and performance of the independent auditors, (ii) appointing, replacing and discharging the independent auditor, (iii) pre-approving the professional services provided by the independent auditor, (iv) reviewing the scope of the annual audit and reports and recommendations submitted by the independent auditor, and (v) reviewing our financial reporting and accounting policies, including any significant changes, with management and the independent auditor. The Audit Committee has reviewed and discussed with management the Company’s audited financial statements for the year ended December 31, 2012.
Compensation Committee
Our Compensation Committee consists of Stuart Davidson, Ernest Mario and Samuel Saks, with Mr. Davidson elected as Chairman of the Committee. Our Board of Directors has determined that all of the members are “independent” under the current listing standards of the NASDAQ Stock Market. Our Board of Directors has adopted a written charter setting forth the authority and responsibilities of the Compensation Committee.
Our Compensation Committee has responsibility for assisting the Board of Directors in, among other things, evaluating and making recommendations regarding the compensation of our executive officers and directors, assuring that the executive officers are compensated effectively in a manner consistent with our stated compensation strategy, producing an annual report on executive compensation in accordance with the rules and regulations promulgated by the SEC, periodically evaluating the terms and administration of our incentive plans and benefit programs and monitoring of compliance with the legal prohibition on loans to our directors and executive officers.
Governance and Nominating Committee
Our Governance and Nominating Committee consists of Donald Landry, Charles Mather and John Rhodes, with Mr. Rhodes elected as Chairman of the Committee. The Board of Directors has determined that all of the members are “independent” under the current listing standards of the NASDAQ Stock Market.
Our Governance and Nominating Committee has responsibility for assisting the Board in, among other things, effecting the organization, membership and function of the Board and its committees. The Governance and Nominating Committee shall identify and evaluate the qualifications of all candidates for nomination for election as directors. In addition, the Governance and Nominating Committee is responsible for developing, recommending and evaluating corporate governance standards and a code of business conduct and ethics.
Involvement in Certain Legal Proceedings
Our Directors and Executive Officers have not been involved in any of the following events during the past ten years:
| 1. | any bankruptcy petition filed by or against such person or any business of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time; |
| 2. | any conviction in a criminal proceeding or being subject to a pending criminal proceeding (excluding traffic violations and other minor offenses); |
| 3. | being subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining him from or otherwise limiting his involvement in any type of business, securities or banking activities or to be associated with any person practicing in banking or securities activities; |
| 75 |
| 4. | being found by a court of competent jurisdiction in a civil action, the Securities and Exchange Commission or the Commodity Futures Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated; |
| 5. | being subject of, or a party to, any federal or state judicial or administrative order, judgment decree, or finding, not subsequently reversed, suspended or vacated, relating to an alleged violation of any federal or state securities or commodities law or regulation, any law or regulation respecting financial institutions or insurance companies, or any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or |
| 6. | being subject of or party to any sanction or order, not subsequently reversed, suspended, or vacated, of any self-regulatory organization, any registered entity or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member. |
| 76 |
EXECUTIVE COMPENSATION
The following table provides certain summary information concerning compensation awarded to, earned by or paid to our Chief Executive Officer, the two highest paid executive officers and up to two other highest paid individuals whose total annual salary and bonus exceeded $100,000 for fiscal years 2012 and 2011.
| Name & Principal
Position | Year | Salary ($) | Bonus ($) | Stock Awards ($) | Option Awards ($) | Non-Equity
Incentive Plan Compensation ($) | Change in
Pension Value and Non- Qualified Deferred Compensation Earnings ($) | All Other
Compensation ($) | Total ($) | |||||||||||||||||||||||||
| Seth Lederman (1) | 2012 | - | - | - | 822,715 | - | - | 279,750 | (2) | 1,102,465 | ||||||||||||||||||||||||
| Chief Executive Officer | 2011 | - | - | - | - | - | - | 300,750 | (2) | 300,750 | ||||||||||||||||||||||||
| Leland Gershell (3) | 2012 | 138,542 | - | - | 587,654 | - | - | - | 726,196 | |||||||||||||||||||||||||
| Chief Financial Officer | ||||||||||||||||||||||||||||||||||
| Bruce Daugherty (4) | 2012 | 110,833 | - | - | 470,123 | - | - | - | 580,956 | |||||||||||||||||||||||||
| Senior Director of Drug Development | ||||||||||||||||||||||||||||||||||
| Benjamin Selzer (5) | 2012 | 192,708 | - | - | - | - | - | - | 192,708 | |||||||||||||||||||||||||
| Chief Operating Officer | ||||||||||||||||||||||||||||||||||
| David J. Moss (6) | 2011 | - | - | - | - | - | - | - | - | |||||||||||||||||||||||||
| Chief Executive Officer | ||||||||||||||||||||||||||||||||||
| Rhonda Rosen (7) | 160,104 | 160,104 | ||||||||||||||||||||||||||||||||
| Chief Financial Officer | 2011 | 140,463 | - | - | - | - | - | - | 140,463 | |||||||||||||||||||||||||
| Susan Oliver (8) | 2011 | 113,249 | - | - | - | - | - | - | 113,249 | |||||||||||||||||||||||||
| Secretary | ||||||||||||||||||||||||||||||||||
| (1) | Dr. Lederman became our President and Chief Executive Officer on October 7, 2011. His compensation reflects payments made to him either through Tonix or Tonix Sub. |
| (2) | Represents $40,000 and $96,000 of consulting fees paid to L&L, $239,750 and $198,750 of consulting fees paid to Lederman & Co and $0 and $6,000 of director fees paid for the years ended December 31, 2012 and 2011, respectively. |
| (3) | Dr. Gershell became our Chief Financial Officer on April 1, 2012 and our Treasurer in November 2012. |
| (4) | Dr. Daugherty became our Senior Director of Drug Development and Controller on April 1, 2012 and our Secretary in November 2012 |
| (5) | Mr. Selzer became our Chief Operating Officer in October 2011 and our interim Chief Financial Officer, Secretary and Treasurer in February 2012. Mr. Selzer resigned as our interim Chief Financial Officer on April 1, 2012. Mr. Selzer was terminated effective October 26, 2012. |
| (6) | Mr. Moss become our Chief Executive Officer on November 22, 2010 and resigned effective October 7, 2011. |
| (7) | Ms. Rosen become our Chief Financial Officer on October 7, 2011. Her compensation reflects payments made to her either through Tonix or Tonix Sub. Ms. Rosen was terminated effective February 16, 2012. |
| (8) | Ms. Oliver was terminated effective October 20, 2011. |
| 77 |
Option/SAR Grants in Fiscal Year Ended December 31, 2012
| Name | Grant Date | All Other Option Awards: Number of Securities Underlying Options (#) | Exercise or Base Price of Option Awards ($/Share) | Grant Date Fair Value of Stock and Option Awards ($) | ||||||||||
| Seth Lederman | 5/9/2012 | 35,000 | $ | 30.00 | $ | 822,715 | ||||||||
| Leland Gershell | 5/9/2012 | 25,000 | $ | 30.00 | $ | 587,654 | ||||||||
| Bruce Daugherty | 5/9/2012 | 20,000 | $ | 30.00 | $ | 470,123 | ||||||||
Outstanding Equity Awards at Fiscal Year-End Table
The following table sets forth information for the named executive officers regarding the number of shares subject to both exercisable and unexercisable stock options, as well as the exercise prices and expiration dates thereof, as of December 31, 2012.
| Name | Number of Securities underlying Unexercised Options (#) Exercisable | Number of Securities underlying Unexercised Options (#) Unexercisable | Option Exercise Price ($/Sh) | Option Expiration Date | ||||||||||
| Seth Lederman | - | 35,000 | $ | 30.00 | 5/9/2022 | |||||||||
| Leland Gershell | - | 25,000 | $ | 30.00 | 5/9/2022 | |||||||||
| Bruce Daugherty | - | 20,000 | $ | 30.00 | 5/9/2022 | |||||||||
Equity Compensation Plan Information
| Plan category | Number of securities to be issued upon exercise of outstanding options (a) | Weighted- average exercise price of outstanding options (b) | Securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) | |||||||||
| Equity compensation plans approved by security holders | 150,000 | $ | 30.00 | 50,000 | ||||||||
| Equity compensation plans not approved by security holders | - | - | - | |||||||||
| Total | 150,000 | $ | 30.00 | 50,000 | ||||||||
| 78 |
Employment Contracts and Termination of Employment and Change-In-Control Arrangements
Employment Agreement with Leland Gershell
Effective April 1, 2012, we entered into an employment agreement (the “Gershell Agreement”) with Dr. Gershell to serve as Chief Financial Officer. The base salary under the Gershell Agreement is $175,000 per annum, which shall increase to $325,000 per annum upon our consummation of an equity sale of securities in excess of $20 million (the “Gershell Threshold”). The Gershell Agreement provides for at-will employment and can be terminated at any time by either party, provided, however, that if we terminate Dr. Gershell for any reason other than cause (as defined in the Gershell Agreement), then Dr. Gershell shall be entitled to six weeks of severance, which severance payment shall increase to six months if such termination occurs after the Gershell Threshold. In addition, Dr. Gershell is entitled to participate in any and all benefit plans, from time to time, in effect for our employees, along with vacation, sick and holiday pay in accordance with its policies established and in effect from time to time.
Employment Agreement with Bruce Daugherty
Effective April 1, 2012, we entered into an employment agreement (the “Daugherty Agreement”) with Dr. Daugherty to serve as Senior Director of Drug Development. The base salary under the Daugherty Agreement is $140,000 per annum, which shall increase to $220,000 per annum upon our consummation of an equity sale of securities in excess of $20 million (the “Daugherty Threshold”). The Daugherty Agreement provides for at-will employment and can be terminated at any time by either party, provided, however, that if we terminate Dr. Daugherty for any reason other than cause (as defined in the Daugherty Agreement), then Dr. Daugherty shall be entitled to six weeks of severance, which severance payment shall increase to six months if such termination occurs after the Daugherty Threshold. In addition, Dr. Daugherty is entitled to participate in any and all benefit plans, from time to time, in effect for our employees, along with vacation, sick and holiday pay in accordance with its policies established and in effect from time to time.
Director Compensation
The following table sets forth summary information concerning the total compensation paid to our non-employee directors in 2012 for services to our company.
| Name | Fees Earned or Paid in Cash ($) | Option Awards ($) | Total ($) | |||||||||
| Stuart Davidson | - | 235,062 | 235,062 | |||||||||
| Patrick Grace | - | 235,062 | 235,062 | |||||||||
| Donald Landry | - | 235,062 | 235,062 | |||||||||
| Ernest Mario | - | 235,062 | 235,062 | |||||||||
| Charles Mather IV | - | 235,062 | 235,062 | |||||||||
| John Rhodes | - | 235,062 | 235,062 | |||||||||
| Samuel Saks | - | 235,062 | 235,062 | |||||||||
| Total: | - | 1,645,434 | 1,645,434 | |||||||||
| 79 |
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Other than as disclosed below, during the last two fiscal years, there have been no transactions, or proposed transactions, which have materially affected or will materially affect us in which any director, executive officer or beneficial holder of more than 5% of the outstanding common, or any of their respective relatives, spouses, associates or affiliates, has had or will have any direct or material indirect interest. We have no policy regarding entering into transactions with affiliated parties.
On June 4, 2010, Tonix Sub entered into a consulting agreement with Lederman & Co, of which our Chairman, CEO and President Seth Lederman is the Managing Member. Pursuant to this agreement, Lederman & Co shall provide clinical development, strategic, management and operational consulting services. In exchange for its services, Tonix Sub shall pay Lederman & Co compensation of $250,000 per annum and issued to Lederman & Co 13,090 shares of its common stock, 20% of which vested on the date of the agreement and the remainder vesting in equal amounts on each of the first, second, third and fourth anniversaries of the date of the agreement. On August 1, 2011, the cash compensation was reduced to $127,000 per annum. On February 1, 2012, the cash compensation was increased to $250,000 per annum. Immediately prior to the Share Exchange, the unvested shares of common stock vested.
On June 4, 2010, Tonix Sub entered into a technology transfer and assignment agreement with Lederman & Co. Pursuant to this agreement, Lederman & Co transferred intellectual property rights related to isometheptene mucate to Tonix Sub. In exchange for the assignment of the intellectual property rights, Tonix Sub issued to Lederman & Co 65,447 shares of its common stock.
On June 4, 2010, Tonix Sub entered into a consulting agreement with L&L, of which our Chairman, CEO and President Seth Lederman is the Manager. Pursuant to this agreement, L&L shall provide scientific and medical consulting services. In exchange for its services, Tonix Sub shall pay L&L compensation of $96,000 per annum, or such greater amount as the Board may designate from time to time, and issued to L&L 51,310 shares of its common stock, 25% of which vested on the date of the agreement and the remainder vesting in equal amounts on each of the first, second and third anniversaries of the date of the agreement. Immediately prior to the Share Exchange, the unvested shares of common stock vested.
| 80 |
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information regarding beneficial ownership of our common stock as of May 9, 2013:
| • | by each person who is known by us to beneficially own more than 5% of our common stock; | |
| • | by each of our officers and directors; and | |
| • | by all of our officers and directors as a group. |
Unless otherwise indicated in the footnotes to the following table, each person named in the table has sole voting and investment power and that person’s address is c/o Tonix Pharmaceuticals Holding Corp., 509 Madison Avenue, Suite 306, New York New York 10022.
| NAME OF OWNER | TITLE OF CLASS | NUMBER OF SHARES OWNED (1) | PERCENTAGE OF COMMON STOCK (2) | |||||||
| Seth Lederman | Common Stock | 488,091 | (3) | 20.65 | % | |||||
| Leland Gershell | Common Stock | 17,153 | (4) | * | ||||||
| Bruce Daugherty | Common Stock | 32,224 | (5) | 1.45 | % | |||||
| Stuart Davidson | Common Stock | 95,528 | (6) | 4.32 | % | |||||
| Patrick Grace | Common Stock | 10,158 | (7) | * | ||||||
| Donald Landry | Common Stock | 100,290 | (8) | 4.50 | % | |||||
| Ernest Mario | Common Stock | 87,100 | (9) | 3.94 | % | |||||
| Charles Mather IV | Common Stock | 16,641 | (10) | * | ||||||
| John Rhodes | Common Stock | 86,210 | (11) | 3.88 | % | |||||
| Samuel Saks | Common Stock | 28,613 | (12) | 1.30 | % | |||||
| Officers and Directors as a Group (10 persons) | Common Stock | 860,259 | (13) | 34.95 | % | |||||
| Lederman & Co., LLC (14) | Common Stock | 289,128 | (15) | 12.56 | % | |||||
| Eli Lederman (16) | Common Stock | 136,391 | (17) | 6.16 | % | |||||
| Technology Partners Fund VIII, LP (18) | Common Stock | 230,020 | (19) | 9.99 | % | |||||
* Denotes less than 1%
(1) Beneficial Ownership is determined in accordance with the rules of the SEC and generally includes voting or investment power with respect to securities. Shares of common stock subject to options or warrants currently exercisable or convertible, or exercisable or convertible within 60 days of May 9, 2013 are deemed outstanding for computing the percentage of the person holding such option or warrant but are not deemed outstanding for computing the percentage of any other person.
(2) Percentage based upon 2,197,490 shares of common stock issued and outstanding as of May 9, 2013.
(3) Includes 12,639 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days, 184,628 shares of common stock and 104,500 shares of common stock underlying warrants owned by Lederman & Co, 32,457 shares of common stock and 24,334 shares of common stock underlying warrants owned by L&L, 58,972 shares of common stock and 8,250 shares of common stock underlying warrants owned by Targent, 4,167 shares of common stock and 8,334 shares of common stock underlying warrants owned by Leder Laboratories, Inc. and 4,167 shares of common stock and 8,334 shares of common stock underlying warrants owned by Starling Pharmaceuticals, Inc. Seth Lederman, as the Managing Member of Lederman & Co and Targent, the Manager of L&L and the Chairman of Leder Laboratories, Inc. and Starling Pharmaceuticals, Inc., has investment and voting control over the shares held by these entities.
| 81 |
(4) Includes 9,028 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days and 3,125 shares of common stock underlying warrants.
(5) Includes 7,223 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days and 14,792 shares of common stock underlying warrants.
(6) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days, 74,536 shares of common stock and 10,834 shares of common stock underlying warrants owned by Lysander, LLC and 6,546 shares owned by Oystercatcher Trust. Stuart Davidson, as the Member of Lysander, LLC and Trustee of Oystercatcher Trust, has investment and voting control over the shares held by these entities.
(7) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days.
(8) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days, 32,457 shares of common stock and 24,334 shares of common stock underlying warrants owned by L&L. Donald Landry, as a Member of L&L, has investment and voting control over the shares held by this entity.
(9) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days and 10,834 shares of common stock underlying warrants.
(10) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days and 4,250 shares of common stock underlying warrants.
(11) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days and 19,167 shares of common stock underlying warrants.
(12) Includes 3,612 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days and 8,334 shares of common stock underlying warrants.
(13) Includes 54,174 shares of common stock underlying options which are currently exercisable or become exercisable within 60 days, 184,628 shares of common stock and 104,500 shares of common stock underlying warrants owned by Lederman & Co, 32,457 shares of common stock and 24,334 shares of common stock underlying warrants owned by L&L, 58,972 shares of common stock and 8,250 shares of common stock underlying warrants owned by Targent, 4,167 shares of common stock and 8,334 shares of common stock underlying warrants owned by Leder Laboratories, Inc., 4,167 shares of common stock and 8,334 shares of common stock underlying warrants owned by Starling Pharmaceuticals, Inc., 74,536 shares of common stock and 10,834 shares of common stock underlying warrants owned by Lysander, LLC, 6,546 shares owned by Oystercatcher Trust and 61,752 shares of common stock underlying warrants owned directly by the executive officers and directors.
(14) Seth Lederman, our President and Chief Executive Officer, has investment and voting control over the shares held by this entity. The mailing address for this entity is 245 E. 93 rd St. 14E, New York, New York 10128.
(15) Includes 104,500 shares of common stock underlying warrants.
(16) The mailing address for this beneficial owner is Malt House Cottage, Hurley, Berkshire, SL6 5LT, United Kingdom.
(17) Includes 15,000 shares of common stock underlying warrants.
(18) The mailing address for this beneficial owner is 100 Shoreline Highway, Suite 282-B, Mill Valley, California 94941. Sheila Mutter and Roger Quy are the managing members of TP Management VIII, LLC, the general partner of Technology Partners Fund VIII, LP and have voting and investment power over the securities owned by it.
| 82 |
(19) Based upon a Schedule 13G filed with the SEC on February 19, 2013. Includes 105,020 shares of common stock underlying warrants and represents the maximum beneficial ownership percentage pursuant to exercise limitations contained within warrants owned by this beneficial owner.
| 83 |
DESCRIPTION OF CAPITAL STOCK
The following is a summary of all material characteristics of our capital stock as set forth in our articles of incorporation and bylaws. The summary does not purport to be complete and is qualified in its entirety by reference to our articles of incorporation and bylaws, and to the provisions of the Nevada Business Corporation Act of the State of Nevada, as amended.
Common Stock
We are authorized to issue up to 150,000,000 shares of our common stock, par value $0.001 per share. As of May 9, 2013, there were 2,197,490 shares of our common stock issued and outstanding. The outstanding shares of our common stock are validly issued, fully paid and nonassessable.
Holders of our common stock are entitled to one vote for each share on all matters submitted to a shareholder vote. Holders of our common stock do not have cumulative voting rights. Therefore, holders of a majority of the shares of our common stock voting for the election of directors can elect all of the directors. Holders of our common stock representing a majority of the voting power of our capital stock issued, outstanding and entitled to vote, represented in person or by proxy, are necessary to constitute a quorum at any meeting of shareholders. A vote by the holders of a majority of our outstanding shares is required to effectuate certain fundamental corporate changes such as liquidation, merger or an amendment to our articles of incorporation.
Holders of our common stock are entitled to share in all dividends that our Board of Directors, in its discretion, declares from legally available funds. In the event of a liquidation, dissolution or winding up, each outstanding share entitles its holder to participate pro rata in all assets that remain after payment of liabilities and after providing for each class of stock, if any, having preference over our common stock. Our common stock has no pre-emptive, subscription or conversion rights and there are no redemption provisions applicable to our common stock.
Preferred Stock
We are authorized to issue up to 5,000,000 shares of preferred stock, par value $0.001 per share, none of which are currently outstanding. The shares of preferred stock may be issued in series, and shall have such voting powers, full or limited, or no voting powers, and such designations, preferences and relative participating, optional or other special rights, and qualifications, limitations or restrictions thereof, as shall be stated and expressed in the resolution or resolutions providing for the issuance of such stock adopted from time to time by the board of directors. The board of directors is expressly vested with the authority to determine and fix in the resolution or resolutions providing for the issuances of preferred stock the voting powers, designations, preferences and rights, and the qualifications, limitations or restrictions thereof, of each such series to the full extent now or hereafter permitted by the laws of the State of Nevada.
Options
As of May 9, 2013, there are an aggregate of 376,500 options to purchase shares of our common stock issued and outstanding. 150,000 of the options are exercisable at $30.00 per share, expire on May 9, 2022 and vest 1/3rd on May 9, 2013 and 1/36th on the 9th of each month thereafter for 24 months. 226,500 of the options are exercisable at $10.20 per share, expire on February 12, 2023 and vest 1/3rd on February 12, 2014 and 1/36th on the 12th of each month thereafter for 24 months
Warrants
In connection with the 2011 Financing, we issued Conversion Warrants to purchase 13,750 shares of Common Stock. In addition, we issued 2011 Agent Warrants to the placement agents to purchase an aggregate of 1,538 shares of Common Stock. The Conversion Warrants have a three year term and $20.00 exercise price. The 2011 Agent Warrants have a $20.00 exercise price and 750 have a two year term and the remaining 788 have a three year term. The Conversion Warrants may be exercised on a cashless basis.
| 84 |
In connection with the March 2012 Financing, we issued March 2012 Class A Warrants to purchase 330,889 shares of Common Stock. In addition, we issued 2012 Agent Warrants to the placement agent to purchase 23,339 shares of Common Stock. The March 2012 Class A Warrants have a five year term and $25.00 exercise price. The 2012 Agent Warrants have a seven year term and $25.00 exercise price. The March 2012 Class A Warrants and 2012 Agent Warrants may be exercised on a cashless basis and contain customary anti-dilution protection.
In connection with the December 2012 Financing, we issued Class A Warrants to purchase 445,209 shares of Common Stock and Class B Warrants to purchase 445,209 shares of Common Stock. The Class A Warrants have a five year term and $12.00 exercise price and the Class B Warrants have a one year term and $8.00 exercise price. The Class A Warrants may be exercised on a cashless basis under certain conditions. As of May 9, 2013, all of the Class A Warrants and 406,875 of the Class B Warrants remain outstanding.
On January 1, 2013, we issued warrants to non-employees to purchase 10,800 shares of our common stock at an exercise price of $12.00 per share expiring five years from the date of issuance vesting ratably over twelve months beginning January 1, 2013 in connection with services.
Convertible Securities
None.
| 85 |
DESCRIPTION OF SECURITIES WE ARE OFFERING
We are offering units, each unit consisting of one share of our common stock and one Series A Warrant to purchase one share of our common stock.
The units will not be issued or certificated. The shares of common stock and the Series A Warrants that we are issuing are immediately separable and will be issued separately. We are not registering the shares of common stock issuable from time to time upon exercise of the Series A Warrants offered hereby.
Common Stock
The material terms and provisions of our common stock and each other class of our securities which qualifies or limits our common stock are described under the caption “Description of Capital Stock” in this prospectus.
Series A Warrants
The following summary of certain terms and provisions of Series A Warrants that are being offered hereby is not complete and is subject to, and qualified in its entirety by the provisions of the Series A Warrant, the form of which has been filed as an exhibit to the registration statement of which this prospectus is a part. Prospective investors should carefully review the terms and provisions of the form of Series A Warrant for a complete description of the terms and conditions of the Series A Warrants.
Duration and Exercise Price. The Series A Warrants offered hereby will entitle the holders thereof to purchase up to an aggregate of [ * ] shares of our common stock at an initial exercise price per share of common stock of [ * ]. The Series A Warrants will be immediately exercisable and will expire on the [ * ] anniversary of the date of issuance. The Series A Warrants will be issued separately from the common stock included in the units, and may be transferred separately immediately thereafter. Series A Warrants will be issued in certificated form only.
Exercisability. The Series A Warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise as discussed below). A holder (together with its affiliates) may not exercise any portion of the warrant to the extent that the holder would own more than 4.9% of the outstanding common stock after exercise, except that upon at least 61 days’ prior notice from the holder to us, the holder may increase the amount of ownership of outstanding stock after exercising the holder’s warrants up to 9.9% of the number of shares of our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the Series A Warrants.
Cashless Exercise. If, at the time a holder exercises its Series A Warrant, there is no effective registration statement registering, or the prospectus contained therein is not available for an issuance of the shares underlying the Series A Warrant to the holder, then in lieu of making the cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the holder may elect instead to receive upon such exercise (either in whole or in part) the net number of shares of common stock determined according to a formula set forth in the Series A Warrant. If we deliver the opinion of counsel referred to above, the Series A Warrants will only be exercisable through a cashless exercise.
Transferability. Subject to applicable laws and the restriction on transfer set forth in the Series A Warrant, the Series A Warrant may be transferred at the option of the holder upon surrender of the Series A Warrant to us together with the appropriate instruments of transfer.
Exchange Listing. We do not intend to list the Series A Warrants on any securities exchange or other trading market.
| 86 |
Right as a Shareholder. Except as otherwise provided in the Series A Warrants or by virtue of such holder’s ownership of shares of our common stock, the holders of the Series A Warrants do not have the rights or privileges of holders of our common stock, including any voting rights, until they exercise their Series A Warrants.
Waivers and Amendments. Subject to certain exceptions, any term of the Series A Warrants may be amended or waived with our written consent and the written consent of the holders of at least 66 2/3% of the then-outstanding Series A Warrants and, in certain instances, with the prior written consent of the underwriter.
| 87 |
UNDERWRITING
We have entered into an underwriting agreement with Roth Capital Partners, LLC and National Securities Corporation with respect to the units subject to this offering. Subject to certain conditions, we have agreed to sell to the underwriters, and the underwriters have severally agreed to purchase, the number of units provided below opposite their respective names.
| Underwriters | Number of Units | |||
| Roth Capital Partners, LLC | ||||
| National Securities Corporation | ||||
| Total | ||||
The underwriters are offering the units subject to their acceptance of the units from us and subject to prior sale. The underwriting agreement provides that the obligations of the several underwriters to pay for and accept delivery of the units offered by this prospectus are subject to the approval of certain legal matters by their counsel and to certain other conditions. The underwriters are obligated to take and pay for all of the units if any such units are taken.
Discounts, Commissions and Expenses
The underwriters have advised us that they propose to offer the units to the public at the initial public offering price set forth on the cover page of this prospectus and to certain dealers at that price less a concession not in excess of $[ * ] per unit. The underwriters may allow, and certain dealers may reallow, a discount from the concession not in excess of $[ * ] per unit to certain brokers and dealers. After this offering, the initial public offering price, concession and reallowance to dealers may be changed by the underwriters. No such change shall change the amount of proceeds to be received by us as set forth on the cover page of this prospectus. The units are offered by the underwriters as stated herein, subject to receipt and acceptance by them and subject to their right to reject any order in whole or in part. The underwriters have informed us that they do not intend to confirm sales to any accounts over which they exercise discretionary authority.
The following table shows the underwriting discounts and commissions payable to the underwriters by us in connection with this offering.
| Per unit1 | Total | |||||||
| Public offering price | $ | $ | ||||||
| Underwriting discounts and commissions payable by us | $ | $ | ||||||
__________________________
1 Does not include the warrants to purchase shares of common stock equal to 4.0% of the number of shares included in the units sold in the offering to be issued to the underwriters at the closing.
We estimate that expenses payable by us in connection with this offering, other than the underwriting discounts and commissions referred to above, will be approximately $[ * ]. We have agreed to reimburse the underwriters for certain out-of-pocket expenses provided that expenses exceeding $100,000 will require our prior approvals, such approval not to be unreasonably withheld.
| 88 |
Underwriters’ Warrants
We have also agreed to issue to the underwriters’ warrants to purchase a number of our shares of common stock equal to an aggregate of 4.0% of the shares of common stock included in the units sold in this offering. The underwriters’ warrants will have an exercise price equal to the public offering price of the units set forth on the cover of this prospectus and may be exercised on a cashless basis. The underwriters’ warrants are not redeemable by us. This prospectus also covers the sale of the underwriters’ warrants and the shares of common stock issuable upon the exercise of the underwriters’ warrants. Except as described above or as summarized below, the underwriters’ warrants will be in substantially the same form as the Series A Warrants included in the units. The underwriters’ warrants and the underlying shares of common stock have been deemed compensation by the Financial Institutions Regulatory Authority, Inc., or FINRA, and are therefore subject to FINRA Rule 5110(g)(1). In accordance with FINRA Rule 5110(g)(1), neither the underwriters’ warrants nor any shares of our common stock issued upon exercise of the underwriters’ warrants may be sold, transferred, assigned, pledged, or hypothecated, or be the subject of any hedging, short sale, derivative, put, or call transaction that would result in the effective economic disposition of such securities by any person for a period of 180 days immediately following the date of effectiveness or commencement of sales of the offering pursuant to which the underwriter warrants are being issued, except the transfer of any security:
| · | by operation of law or by reason of reorganization of our company; |
| · | to any FINRA member firm participating in this offering and the officers or partners thereof, if all securities so transferred remain subject to the lock-up restriction described above for the remainder of the time period; |
| · | if the aggregate amount of our securities held by either an underwriter or a related person do not exceed 1% of the securities being offered; |
| · | that is beneficially owned on a pro-rata basis by all equity owners of an investment fund, provided that no participating member manages or otherwise directs investments by the fund, and participating members in the aggregate do not own more than 10% of the equity in the fund; or |
| · | the exercise or conversion of any security, if all securities received remain subject to the lock-up restriction set forth above for the remainder of the time period. |
In addition, in accordance with FINRA Rule 5110(f)(2)(H), the underwriter warrants may not contain certain terms.
Indemnification
We have agreed to indemnify the underwriters against certain liabilities, including liabilities under the Securities Act of 1933, as amended, or the Securities Act, and liabilities arising from breaches of representations and warranties contained in the underwriting agreement, or to contribute to payments that the underwriters may be required to make in respect of those liabilities.
Lock-up Agreements
We, our officers, directors and certain of our shareholders have agreed, subject to limited exceptions, for a period of 90 days after the date of the underwriting agreement, not to offer, sell, contract to sell, pledge, grant any option to purchase, make any short sale or otherwise dispose of, directly or indirectly any shares of common stock or any securities convertible into or exchangeable for our common stock either owned as of the date of the underwriting agreement or thereafter acquired without the prior written consent of the underwriters. This 90-day period may be extended if (1) during the last 17 days of the 90-day period, we issue an earnings release or material news or a material event regarding us occurs or (2) prior to the expiration of the 90-day period, we announce that we will release earnings results during the 16-day period beginning on the last day of the 90-day period, then the period of such extension will be 18 days, beginning on the issuance of the earnings release or the occurrence of the material news or material event. If after any announcement described in clause (2) of the preceding sentence, we announce that we will not release earnings results during the 16-day period, the lock-up period shall expire the later of the expiration of the 90-day period and the end of any extension of such period made pursuant to clause (1) of the preceding sentence. The underwriters may, in their sole discretion and at any time or from time to time before the termination of the lock-up period, without notice, release all or any portion of the securities subject to lock-up agreements.
| 89 |
Price Stabilization, Short Positions and Penalty Bids
The underwriters have advised us that they do not intend to conduct any stabilization or over-allotment activities in connection with this offering.
Electronic Distribution
This prospectus in electronic format may be made available on websites or through other online services maintained by one or more of the underwriters, or by their affiliates. Other than this prospectus in electronic format, the information on any underwriter’s website and any information contained in any other website maintained by an underwriter is not part of this prospectus or the registration statement of which this prospectus forms a part, has not been approved and/or endorsed by us or any underwriter in its capacity as underwriter, and should not be relied upon by investors.
Other
From time to time, certain of the underwriters and/or their affiliates have provided, and may in the future provide, various investment banking and other financial services for us for which services they have received and, may in the future receive, customary fees. Except for services provided in connection with this offering, no underwriter has provided any investment banking or other financial services during the 180-day period preceding the date of this prospectus and we do not expect to retain any underwriter to perform any investment banking or other financial services for at least 90 days after the date of this prospectus.
NOTICE TO INVESTORS
Notice to Investors in the United Kingdom
In relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (each, a “Relevant Member State”) an offer to the public of any securities which are the subject of the offering contemplated by this prospectus may not be made in that Relevant Member State except that an offer to the public in that Relevant Member State of any such securities may be made at any time under the following exemptions under the Prospectus Directive, if they have been implemented in that Relevant Member State:
(a) to legal entities which are authorized or regulated to operate in the financial markets or, if not so authorized or regulated, whose corporate purpose is solely to invest in securities;
(b) to any legal entity which has two or more of (1) an average of at least 250 employees during the last financial year; (2) a total balance sheet of more than €43,000,000 and (3) an annual net turnover of more than €50,000,000, as shown in its last annual or consolidated accounts;
(c) by an underwriter to fewer than 100 natural or legal persons (other than qualified investors as defined in the Prospectus Directive); or
(d) in any other circumstances falling within Article 3(2) of the Prospectus Directive, provided that no such offer of these securities shall result in a requirement for the publication by the issuer or the underwriter of a prospectus pursuant to Article 3 of the Prospectus Directive.
| 90 |
For the purposes of this provision, the expression an “offer to the public” in relation to any of the securities in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and any such securities to be offered so as to enable an investor to decide to purchase any such securities, as the same may be varied in that Member State by any measure implementing the Prospectus Directive in that Member State and the expression “Prospectus Directive” means Directive 2003/71/EC and includes any relevant implementing measure in each Relevant Member State.
Each underwriter has represented, warranted and agreed that:
(a) it has only communicated or caused to be communicated and will only communicate or cause to be communicated any invitation or inducement to engage in investment activity (within the meaning of section 21 of the Financial Services and Markets Act 2000 (the FSMA)) received by it in connection with the issue or sale of any of the securities in circumstances in which section 21(1) of the FSMA does not apply to the issuer; and
(b) it has complied with and will comply with all applicable provisions of the FSMA with respect to anything done by it in relation to the securities in, from or otherwise involving the United Kingdom.
European Economic Area
In particular, this document does not constitute an approved prospectus in accordance with European Commission’s Regulation on Prospectuses no. 809/2004 and no such prospectus is to be prepared and approved in connection with this offering. Accordingly, in relation to each Member State of the European Economic Area which has implemented the Prospectus Directive (being the Directive of the European Parliament and of the Council 2003/71/EC and including any relevant implementing measure in each Relevant Member State) (each, a Relevant Member State), with effect from and including the date on which the Prospectus Directive is implemented in that Relevant Member State (the Relevant Implementation Date) an offer of securities to the public may not be made in that Relevant Member State prior to the publication of a prospectus in relation to such securities which has been approved by the competent authority in that Relevant Member State or, where appropriate, approved in another Relevant Member State and notified to the competent authority in that Relevant Member State, all in accordance with the Prospectus Directive, except that it may, with effect from and including the Relevant Implementation Date, make an offer of securities to the public in that Relevant Member State at any time:
| · | to legal entities which are authorized or regulated to operate in the financial markets or, if not so authorized or regulated, whose corporate purpose is solely to invest in securities; |
| · | to any legal entity which has two or more of (1) an average of at least 250 employees during the last financial year; (2) a total balance sheet of more than €43,000,000; and (3) an annual net turnover of more than €50,000,000, as shown in the last annual or consolidated accounts; or |
| · | in any other circumstances which do not require the publication by the Issuer of a prospectus pursuant to Article 3 of the Prospectus Directive. |
For the purposes of this provision, the expression an “offer of securities to the public” in relation to any of the securities in any Relevant Member State means the communication in any form and by any means of sufficient information on the terms of the offer and the securities to be offered so as to enable an investor to decide to purchase or subscribe for the securities, as the same may be varied in that Member State by any measure implementing the Prospectus Directive in that Member State. For these purposes the units offered hereby are “securities.”
LEGAL MATTERS
The legality of the securities offered hereby has been passed on for us by Sichenzia Ross Friedman Ference LLP, New York, New York. Lowenstein Sandler LLP, New York, New York, is acting as counsel for the underwriters in connection with this offering.
| 91 |
EXPERTS
EisnerAmper LLP, an independent registered public accounting firm, has audited, as set forth in its report thereon appearing elsewhere herein, our financial statements at December 31, 2012 and 2011 and for the years then ended that appear in the prospectus. The financial statements referred to above are included in this prospectus in reliance upon the independent registered public accounting firm’s report given on their authority as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We have filed a registration statement on Form S-1 under the Securities Act of 1933, as amended, relating to the shares of common stock and warrants being offered by this prospectus, and reference is made to such registration statement. This prospectus constitutes the prospectus of Tonix Pharmaceuticals Holding Corp., filed as part of the registration statement, and it does not contain all information in the registration statement, as certain portions have been omitted in accordance with the rules and regulations of the Securities and Exchange Commission. Whenever we make reference in this prospectus to any of our contracts, agreements or other documents, the references are not necessarily complete, and you should refer to the exhibits attached to the registration statement for copies of the actual contract, agreement or other document.
We file annual, quarterly and special reports and other information with the SEC (Commission File Number 000-54879). These filings contain important information that does not appear in this prospectus. For further information about us, you may read and copy any reports, statements and other information filed by us at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549-0102. You may obtain further information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. Our SEC filings are also available on the SEC Internet site at http://www.sec.gov, which contains periodic reports and other information regarding issuers that file electronically. You can find information about Tonix on our website at http://www.tonixpharma.com. Information found on our website is not part of this prospectus. You may also request a copy of any of our periodic reports filed with the SEC by writing or telephoning us at the following address:
Leland Gershell
Chief Financial Officer
Tonix Pharmaceuticals Holding Corp.
509 Madison Avenue, Suite 306
New York, New York 10022
Telephone (212) 980-9155
| 92 |
INDEX TO FINANCIAL STATEMENTS
TONIX PHARMACEUTICALS HOLDING CORP.
INDEX TO FINANCIAL STATEMENTS
| For the Years Ended December 31, 2012 and 2011 | ||
| Report of Independent Registered Public Accounting Firm | F-1 | |
| Consolidated balance sheets as of December 31, 2012 and 2011 | F-2 | |
| Consolidated statements of operations for the years ended December 31, 2012 and 2011 and for the period from June 7, 2007 (date of inception) through December 31, 2012 | F-3 | |
| Consolidated statements of stockholders’ equity (deficiency) for the years ended December 31, 2012, 2011, 2010, 2009, 2008 and for the period from June 7, 2007 (date of inception) through December 31, 2007 | F-4 – F-6 | |
| Consolidated statements of cash flows for the years ended December 31, 2012 and 2011 and for the period from June 7, 2007 (date of inception) through December 31, 2012 | F-7 | |
| Notes to consolidated financial statements | F-8 – F-19 | |
| For the Three Months Ended March 31, 2013 and 2012 | ||
| Condensed consolidated balance sheets as of March 31, 2013 (unaudited) and December 31, 2012 | F-20 | |
| Condensed consolidated statements of operations for the three months ended March 31, 2013 and 2012 and for the period from June 7, 2007 (date of inception) through March 31, 2013 (unaudited) | F-21 | |
| Condensed consolidated statement of stockholders’ (deficiency) equity for the three months ended March 31, 2013 (unaudited) | F-22 | |
| Condensed consolidated statements of cash flows for the three months ended March 31, 2013 and 2012 and for the period from June 7, 2007 (date of inception) through March 31, 2013 (unaudited) | F-23 | |
| Notes to condensed consolidated financial statements (unaudited) | F-24 – F-28 |
| 93 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders
Tonix Pharmaceuticals Holding Corp.
We have audited the accompanying consolidated balance sheets of Tonix Pharmaceuticals Holding Corp. (a development stage company) (the "Company") as of December 31, 2012 and 2011, and the related consolidated statements of operations and cash flows for the years then ended and for the period from June 7, 2007 (inception) through December 31, 2012 and the consolidated statements of stockholders' (deficiency) equity for each of the five years in the period ended December 31, 2012 and for the period from June 7, 2007 (inception) through December 31, 2007. These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of Tonix Pharmaceuticals Holding Corp. as of December 31, 2012 and 2011, the consolidated results of its operations and its cash flows for the years then ended and for the period from June 7, 2007 (inception) through December 31, 2012 and consolidated changes in stockholders' (deficiency) equity for each of the five years in the period ended December 31, 2012 and for the period from June 7, 2007 (inception) through December 31, 2007, in conformity with accounting principles generally accepted in the United States of America.
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the financial statements, the Company has incurred recurring net losses and negative cash flows from operations and requires additional financing to fund future operations. These events and conditions, among others referred to in Note 2, raise substantial doubt about the Company's ability to continue as a going concern. Management's plans concerning these matters are also described in Note 2. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
/s/ EisnerAmper LLP
EisnerAmper LLP
New York, New York
March 8, 2013, except for the third paragraph of Note 15, as to which the date is May 1, 2013
| F-1 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
CONSOLIDATED BALANCE SHEETS
DECEMBER 31, 2012 AND 2011
| 2012 | 2011 | |||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash | $ | 1,785,390 | $ | 41,123 | ||||
| Prepaid expenses and other | 224,659 | 102,430 | ||||||
| Total current assets | 2,010,049 | 143,553 | ||||||
| Furniture and equipment, net | 46,894 | 25,550 | ||||||
| Deferred financing costs, net | - | 196,166 | ||||||
| Restricted cash | 60,267 | 60,177 | ||||||
| Total assets | $ | 2,117,210 | $ | 425,446 | ||||
| LIABILITIES AND STOCKHOLDERS' EQUITY (DEFICIENCY) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable, including $6,809 and $27,483 to related parties as of December 31, 2012 and 2011, respectively | $ | 825,837 | $ | 695,198 | ||||
| Accrued expenses | 309,800 | 10,229 | ||||||
| Accrued interest, including $3,155 and $5,006 to related parties as of December 31, 2012 and 2011, respectively | 3,155 | 38,306 | ||||||
| Liability to placement agent | - | 31,543 | ||||||
| Convertible debentures | - | 150,000 | ||||||
| Total current liabilities | 1,138,792 | 925,276 | ||||||
| Convertible debentures, including $265,000 to related parties | - | 1,925,000 | ||||||
| Deferred rent payable | 19,710 | 29,083 | ||||||
| Total liabilities | 1,158,502 | 2,879,359 | ||||||
| Commitments | - | - | ||||||
| Stockholders' equity (deficiency): | ||||||||
| Preferred stock, $0.001 par value; 5,000,000 and -0- shares authorized as of December 31, 2012 and 2011, respectively; none issued or outstanding | - | - | ||||||
| Common stock, $0.001 par value; 150,000,000 and 75,000,000 shares authorized as of December 31, 2012 and 2011, respectively; 2,159,156 and 1,353,350 shares issued and outstanding as of December 31, 2012 and 2011, respectively | 2,159 | 1,353 | ||||||
| Additional paid in capital | 16,800,829 | 3,939,414 | ||||||
| Deficit accumulated during development stage | (15,844,280 | ) | (6,394,680 | ) | ||||
| Total stockholders' equity (deficiency) | 958,708 | (2,453,913 | ) | |||||
| Total liabilities and stockholders' equity (deficiency) | $ | 2,117,210 | $ | 425,446 | ||||
See the accompanying notes to consolidated financial statements
| F-2 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
CONSOLIDATED STATEMENTS OF OPERATIONS
| From June 7, 2007 | ||||||||||||
| (date of inception) | ||||||||||||
| Year ended December 31, | Through | |||||||||||
| 2012 | 2011 | December 31, 2012 | ||||||||||
| COSTS AND EXPENSES: | ||||||||||||
| Research and development | $ | 2,583,308 | $ | 1,158,167 | $ | 4,535,262 | ||||||
| General and administrative | 4,078,102 | 2,220,361 | 8,333,349 | |||||||||
| 6,661,410 | 3,378,528 | 12,868,611 | ||||||||||
| Operating Loss | (6,661,410 | ) | (3,378,528 | ) | (12,868,611 | ) | ||||||
| Gain on extinguishment of debt | - | - | 7,908 | |||||||||
| Other income | 1,875 | - | 1,875 | |||||||||
| Change in fair value of warrants liability | (1,177,026 | ) | (1,177,026 | ) | ||||||||
| Interest and other financing costs, net | (1,613,039 | ) | (91,585 | ) | (1,808,426 | ) | ||||||
| NET LOSS | $ | (9,449,600 | ) | $ | (3,470,113 | ) | $ | (15,844,280 | ) | |||
| Net loss per common share, basic and diluted | $ | (5.58 | ) | $ | (3.24 | ) | ||||||
| Weighted average common shares outstanding, basic and diluted | 1,693,436 | 1,071,295 | ||||||||||
See the accompanying notes to consolidated financial statements
| F-3 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
CONSOLIDATED STATEMENT OF STOCKHOLDERS' EQUITY (DEFICIENCY)
| Deficit | ||||||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||||||
| Additional | During | |||||||||||||||||||||||||||
| Preferred stock | Common stock | Paid in | Development | |||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Stage | Total | ||||||||||||||||||||||
| Shares issued to founders for intellectual property in June 2007 ($3.00 per share) | - | $ | - | 29,451 | $ | 29 | $ | 87,721 | $ | - | $ | 87,750 | ||||||||||||||||
| Shares issued to bankers for services in June 2007 ($3.00 per share) | - | - | 3,272 | 3 | 9,747 | - | 9,750 | |||||||||||||||||||||
| Compensation related to restricted share awards issued to directors in November 2007 | - | - | - | - | 24,187 | - | 24,187 | |||||||||||||||||||||
| Net loss | - | - | - | - | - | (537,001 | ) | (537,001 | ) | |||||||||||||||||||
| Balance at December 31, 2007 | - | - | 32,723 | 32 | 121,655 | (537,001 | ) | (415,314 | ) | |||||||||||||||||||
| Compensation related to cancelled restricted share awards in December 2008 | - | - | - | - | 72,563 | - | 72,563 | |||||||||||||||||||||
| Net loss | - | - | - | - | - | (202,262 | ) | (202,262 | ) | |||||||||||||||||||
| Balance at December 31, 2008 | - | - | 32,723 | 32 | 194,218 | (739,263 | ) | (545,013 | ) | |||||||||||||||||||
| Conversion of senior convertible notes into Preferred stock in June 2009 ($2.60 per share) | - | - | 360,004 | 360 | 199,640 | - | 200,000 | |||||||||||||||||||||
| Shares issued to directors in July 2009 ($3.00 per share) | - | - | 1,571 | 2 | 4,678 | - | 4,680 | |||||||||||||||||||||
| Capital contribution in June 2009 | - | - | - | - | 23,725 | - | 23,725 | |||||||||||||||||||||
| Net loss | - | - | - | - | - | (220,834 | ) | (220,834 | ) | |||||||||||||||||||
| Balance at December 31, 2009 | - | $ | - | 394,298 | $ | 394 | $ | 422,261 | $ | (960,097 | ) | $ | (537,442 | ) | ||||||||||||||
| F-4 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
CONSOLIDATED STATEMENT OF STOCKHOLDERS' EQUITY (DEFICIENCY)
| Deficit | ||||||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||||||
| Additional | During | |||||||||||||||||||||||||||
| Preferred stock | Common stock | Paid in | Development | |||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Stage | Total | ||||||||||||||||||||||
| Conversion of demand notes into capital stock in July 2010 ($4.60 per share) | - | $ | - | 104,729 | $ | 105 | $ | 479,895 | $ | - | $ | 480,000 | ||||||||||||||||
| Conversion of accrued interest on demand notes into capital stock in July 2010 ($4.60 per share) | - | - | 15,072 | 15 | 69,063 | - | 69,078 | |||||||||||||||||||||
| Issuance of capital stock in August to December 2010 ($4.60 per share) | - | - | 292,804 | 293 | 1,341,708 | - | 1,342,001 | |||||||||||||||||||||
| Shares issued to founders for intellectual property in June 2010 ($4.52 per share) | - | - | 65,447 | 66 | 295,434 | - | 295,500 | |||||||||||||||||||||
| Issuance of restricted shares to directors, employees and consultants in June to November 2010 ($4.76 per share) | - | - | 29,386 | 29 | 139,853 | - | 139,882 | |||||||||||||||||||||
| Net loss | - | - | - | - | - | (1,964,470 | ) | (1,964,470 | ) | |||||||||||||||||||
| Balance at December 31, 2010 | - | - | 901,735 | 902 | 2,748,214 | (2,924,567 | ) | (175,451 | ) | |||||||||||||||||||
| Vesting and issuance of capital stock in January to September 2011 ($4.60 per share) | - | - | 133,530 | 133 | 611,867 | - | 612,000 | |||||||||||||||||||||
| Vesting and issuance of restricted shares to directors, employees and consultants in February to April 2011 and vesting of restricted shares pursuant to share exchange agreement in October 2011 ($4.60 per share) | - | - | 98,084 | 98 | 435,553 | - | 435,651 | |||||||||||||||||||||
| Common stock issued in connection with the share exchange transaction in October 2011 | - | - | 200,000 | 200 | (200 | ) | - | - | ||||||||||||||||||||
| Common stock issued in October 2011 in exchange for services rendered ($7.20 per share) | - | - | 20,000 | 20 | 143,980 | - | 144,000 | |||||||||||||||||||||
| Net loss | - | - | - | - | - | (3,470,113 | ) | (3,470,113 | ) | |||||||||||||||||||
| Balance at December 31, 2011 | - | $ | - | 1,353,350 | $ | 1,353 | $ | 3,939,414 | $ | (6,394,680 | ) | $ | (2,453,913 | ) | ||||||||||||||
| F-5 |
| TONIX PHARMACEUTICALS HOLDING CORP. |
| (a development stage company) |
| CONSOLIDATED STATEMENT OF STOCKHOLDERS' EQUITY (DEFICIENCY) |
| Deficit | ||||||||||||||||||||||||||||
| Accumulated | ||||||||||||||||||||||||||||
| Additional | During | |||||||||||||||||||||||||||
| Preferred stock | Common stock | Paid in | Development | |||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Stage | Total | ||||||||||||||||||||||
| Issuance of common stock in January 2012 to holders of convertible debentures ($12.40 per share) | - | $ | - | 29,700 | $ | 30 | $ | 368,250 | $ | - | $ | 368,280 | ||||||||||||||||
| Issuance of common stock in January and March 2012 ($12.40 per share) net of transaction expenses of $435,713 | - | - | 330,892 | 331 | 3,631,980 | - | 3,632,311 | |||||||||||||||||||||
| Warrants issued in January 2012 to holders of convertible debentures | - | - | - | - | 83,289 | - | 83,289 | |||||||||||||||||||||
| Warrants issued to placement agent in January 2012 | - | - | - | - | 6,126 | - | 6,126 | |||||||||||||||||||||
| Warrants reclassified to equity upon expiry of reset provisions | - | - | - | - | 3,938,946 | - | 3,938,946 | |||||||||||||||||||||
| Issuance of common stock and warrants in December 2012 to holders of convertible debentures ($6.00) per share | - | - | 118,335 | 118 | 709,882 | 710,000 | ||||||||||||||||||||||
| Issuance of common stock and warrants in December 2012 ($8.00 per share) net of transaction expenses of $70,000 | - | - | 326,879 | 327 | 2,544,673 | - | 2,545,000 | |||||||||||||||||||||
| Beneficial conversion feature in connection with convertible debentures | - | - | - | - | 710,000 | - | 710,000 | |||||||||||||||||||||
| Capital contribution of accrued interest | - | - | - | - | 3,111 | - | 3,111 | |||||||||||||||||||||
| Stock based compensation | - | - | - | - | 865,158 | - | 865,158 | |||||||||||||||||||||
| Net loss | - | - | - | - | - | (9,449,600 | ) | (9,449,600 | ) | |||||||||||||||||||
| Balance at December 31, 2012 | - | $ | - | 2,159,156 | $ | 2,159 | $ | 16,800,829 | $ | (15,844,280 | ) | $ | 958,708 | |||||||||||||||
See the accompanying notes to consolidated financial statements
| F-6 |
| TONIX PHARMACEUTICALS HOLDING CORP. |
| (a development stage company) |
| CONSOLIDATED STATEMENTS OF CASH FLOWS |
| From June 7, 2007 | ||||||||||||
| (date of inception) | ||||||||||||
| Year ended December 31, | Through | |||||||||||
| 2012 | 2011 | December 31, 2012 | ||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||||||
| Net loss | $ | (9,449,600 | ) | $ | (3,470,113 | ) | $ | (15,844,280 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation | 14,329 | 9,300 | 31,641 | |||||||||
| Amortization and write off of deferred financing costs | 196,166 | 53,377 | 249,543 | |||||||||
| Non cash interest, consisting of beneficial conversion feature in connection with convertible debentures | 710,000 | - | 710,000 | |||||||||
| Non cash interest, consisting of common stock and warrants issued in connection with convertible debentures | 426,152 | - | 426,152 | |||||||||
| Non-cash financing costs related to January and March 2012 financing | 81,337 | - | 81,337 | |||||||||
| Stock based compensation | 865,158 | 435,651 | 1,551,871 | |||||||||
| Change in fair value of warrant liability | 1,177,026 | - | 1,177,026 | |||||||||
| Common stock issued in exchange for intellectual property | - | - | 383,250 | |||||||||
| Gain on extinguishment of debt | - | - | (7,908 | ) | ||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Prepaid expenses | (122,229 | ) | (79,117 | ) | (224,659 | ) | ||||||
| Accounts payable | 130,639 | 377,453 | 825,837 | |||||||||
| Accrued interest | (32,040 | ) | 38,306 | 6,266 | ||||||||
| Accrued expenses | 293,125 | (12,304 | ) | 404,065 | ||||||||
| Deferred rent payable | (2,927 | ) | 9,909 | 26,156 | ||||||||
| Net cash used in operating activities | (5,712,864 | ) | (2,637,538 | ) | (10,203,703 | ) | ||||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||||||
| Purchase of furniture and fixtures | (35,673 | ) | (2,764 | ) | (78,535 | ) | ||||||
| Proceeds from security deposit | - | 3,156 | - | |||||||||
| Payment of restricted cash and interest earned on restricted cash | (90 | ) | (90 | ) | (60,267 | ) | ||||||
| Net cash (used in) provided by investing activities | (35,763 | ) | 302 | (138,802 | ) | |||||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||||||
| Proceeds from demand notes | - | - | 480,000 | |||||||||
| Proceeds from other notes payable | 320,000 | 500,000 | 1,020,000 | |||||||||
| Proceeds, net of expenses of $24,000 as of December 31, 2011, from Convertible Debentures | 390,000 | 1,501,000 | 1,891,000 | |||||||||
| Repayment of Convertible Debentures | (150,000 | ) | - | (150,000 | ) | |||||||
| Proceeds, net of expenses of $374,870 from sale of units consisting of common stock and warrants | 6,932,894 | - | 6,932,894 | |||||||||
| Proceeds from the sale of capital stock | - | 612,000 | 1,954,001 | |||||||||
| Net cash provided by financing activities | 7,492,894 | 2,613,000 | 12,127,895 | |||||||||
| Net increase (decrease) in cash | 1,744,267 | (24,236 | ) | 1,785,390 | ||||||||
| Cash, beginning of the period | 41,123 | 65,359 | - | |||||||||
| Cash, end of period | $ | 1,785,390 | $ | 41,123 | $ | 1,785,390 | ||||||
| Supplemental disclosures of cash flow information: | ||||||||||||
| Interest paid | $ | 35,195 | $ | - | $ | - | ||||||
| Non cash investing and financing activities: | ||||||||||||
| Senior convertible notes exchanged for preferred shares | $ | - | $ | - | $ | 200,000 | ||||||
| Capital contribution of accrued interest | $ | 3,111 | $ | - | $ | 26,836 | ||||||
| Demand notes together with accrued interest converted into capital stock | $ | - | $ | - | $ | 549,078 | ||||||
| Common stock issued for deferred financing costs | $ | - | $ | 144,000 | $ | 144,000 | ||||||
| Exchange of Notes Payable for Convertible Debenture | $ | 320,000 | $ | - | $ | 820,000 | ||||||
| Warrants Liability reclassified to Stockholders' Equity | $ | 3,938,946 | $ | - | $ | 3,938,946 | ||||||
| Exchange of Convertible Debenture for Units consisting of common stock and warrants | $ | 2,635,000 | $ | - | $ | 2,635,000 | ||||||
See the accompanying notes to consolidated financial statements
| F-7 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
NOTE 1 –BUSINESS AND RECAPITALIZATION
Tonix Pharmaceuticals Holding Corp. through its wholly owned subsidiary Tonix Pharmaceuticals, Inc. is attempting to develop safer and more effective versions of widely prescribed central nervous system ("CNS") drugs. While some new applications can use the commercially available form of the drug, in other cases reformulating the active ingredient improves its safety or effectiveness in treating the CNS condition. When formal development programs have proven successful in clinical tests, Tonix Pharmaceuticals, Inc. intends to seek marketing approval from the Food and Drug Administration ("FDA").
On August 16, 2010, Tonix Pharmaceuticals, Inc. formed Krele LLC ("Krele") in the state of Delaware. Krele is a limited liability corporation whose sole member is Tonix Pharmaceuticals Inc. Krele was established to commercialize products that are generic versions of predicate new drug application products or versions of drug efficacy study implementation products. The Company expects that its relationship to Krele will be similar to that of several other pharmaceutical companies and their subsidiaries that market generic versions of the parent's branded products at different periods in their product life-cycle.
On October 7, 2011, Tonix Pharmaceuticals, Inc. (formerly Krele Pharmaceuticals, Inc. incorporated on June 7, 2007 in the State of Delaware) and a publicly traded non-operating shell company Tamandare Explorations Inc. (“Tamandare”), incorporated under the laws of the State of Nevada, along with certain other parties executed and consummated a share exchange agreement (the “Share Exchange”). Pursuant to the Share Exchange, each share of Tonix Pharmaceuticals Inc.’s common stock was exchanged for 0.9 shares of Tamandare’s common stock and each share of Tonix Pharmaceuticals, Inc.’s Series A and B preferred stock was exchanged for 4.8 shares of Tamandare’s common stock. Upon completion of the Share Exchange, the Tonix Pharmaceuticals, Inc. shareholders, including holders of restricted shares, which were subject to accelerated vesting, received in exchange for all of their shares, an aggregate of 1,133,334 shares of Tamandare’s common stock and Tamandare’s existing stockholders retained 200,000 shares of common stock. The 1,133,334 shares issued to the Tonix Pharmaceuticals, Inc. shareholders constituted approximately 85% of Tamandare’s 1,333,334 issued and outstanding shares of common stock after the Share Exchange. Upon completion of the Share Exchange, Tonix Pharmaceuticals, Inc. became Tamandare’s wholly-owned subsidiary and in October 2011 Tamandare was renamed Tonix Pharmaceuticals Holding Corp. As the owners and management of Tonix Pharmaceuticals, Inc. obtained voting and operating control of Tamandare after the Share Exchange and Tamandare was non-operating, had no assets or liabilities and did not meet the definition of a business, the transaction has been accounted for as a recapitalization of Tonix Pharmaceuticals, Inc., accompanied by the issuance of its common stock for outstanding common stock of Tamandare, which was recorded at a nominal value. The accompanying financial statements and related notes give retroactive effect to the recapitalization as if it had occurred on June 7, 2007 (inception date) and accordingly all share and per share amounts have been adjusted.
NOTE 2 - SIGNIFICANT ACCOUNTING POLICIES
Basis of presentation:
The consolidated financial statements include the accounts of Tonix Pharmaceuticals Holding Corp. and its wholly owned subsidiaries, Tonix Pharmaceuticals, Inc. and Krele LLC (hereafter referred to as the “Company” or “Tonix”). All significant intercompany balances and transactions have been eliminated in consolidation.
As the Company is devoting substantially all of its efforts to establishing a new business, and while planned principal operations have commenced, there has been no revenue generated from sales, license fees or royalties, the Company is considered a development stage enterprise. Accordingly, the Company's consolidated financial statements are presented in accordance with authoritative accounting guidance related to a development stage enterprise. Financial position, results of operations and cash flows of a development stage enterprise are presented in conformity with generally accepted accounting principles that apply to established operating enterprises.
As a development stage enterprise, the Company's primary efforts are devoted to conducting research and development for the treatment of CNS diseases. The Company has experienced net losses and negative cash flows from operations since inception and expects these conditions to continue for the foreseeable future. In addition, although the Company has approximately $900,000 of working capital at December 31, 2012, the Company will require additional financing to fund future operations as it is expected that cash to be used in operations will increase significantly over the next several years. Further, the Company does not have any commercial products available for sale and has not generated revenues and there is no assurance that if approval of their products is received that the Company will be able to generate cash flow to fund operations. In addition, there can be no assurance that the Company's research and development will be successfully completed or that any product will be approved or commercially viable.
| F-8 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
The Company intends to raise additional capital to complete the development and commercialization of its current product candidates through equity or debt financing; however the Company does not have any commitments or definitive or binding arrangements for such funds. There can be no assurance that such funds, if available at all, can be obtained on terms reasonable to the Company. If the Company is unsuccessful in raising additional capital it will need to reduce costs and operations substantially.
The above factors raise substantial doubt as to the Company's ability to continue as a going concern. The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern and do not include any adjustments that may result from the outcome of this uncertainty.
Use of estimates
The preparation of financial statements in accordance with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities at the date of the financial statements, and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates. Significant estimates include assumptions used in the fair value of stock-based compensation and the fair value of other equity instruments.
Research and Development costs
The Company outsources its research and development efforts and expenses related costs as incurred, including the cost of manufacturing product for testing, licensing fees and costs associated with planning and conducting clinical trials. The value ascribed to patents and other intellectual property acquired was expensed in 2007 and 2010 as research and development costs, as it related to particular research and development projects and had no alternative future uses.
Furniture and equipment
Furniture and equipment are stated at cost, less accumulated depreciation. Depreciation is calculated using the straight-line method over the asset's estimated useful life, which is three years for computer assets and five years for furniture and all other equipment. Expenditures for maintenance and repairs are expensed as incurred.
Income taxes
Deferred income tax assets and liabilities are determined based on the estimated future tax effects of net operating loss and credit carryforwards and temporary differences between the tax basis of assets and liabilities and their respective financial reporting amounts measured at the current enacted tax rates. The Company records an estimated valuation allowance on its deferred income tax assets if it is not more likely than not that these deferred income tax assets will be realized.
The Company recognizes a tax benefit from an uncertain tax position only if it is more likely than not that the tax position will be sustained on examination by taxing authorities, based on the technical merits of the position. The tax benefits recognized in the consolidated financial statements from such a position are measured based on the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement. As of December 31, 2012 and 2011, the Company has not recorded any unrecognized tax benefits.
Stock-based compensation
All stock-based payments to employees and to nonemployee directors for their services as directors, including grants of restricted stock and stock options, are measured at fair value on the grant date and recognized in the consolidated statements of operations as compensation or other expense over the relevant service period. Stock-based payments to nonemployees are recognized as an expense over the period of performance. Such payments are measured at fair value at the earlier of the date a performance commitment is reached or the date performance is completed. In addition, for awards that vest immediately and are non forfeitable the measurement date is the date the award is issued.
| F-9 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
Per share data:
Basic and diluted net loss per common share is calculated by dividing net loss, by the weighted average number of outstanding shares of common stock, adjusted to give effect to the exchange ratio in the Share Exchange in October 2011, which was accounted for as a recapitalization of the Company (see Note 1), and to the 1-for-20 reverse stock split, which was effected on May 1, 2013 (see Note 15).
In October 2011, upon completion of the share exchange referred to above, the Company issued Convertible Debentures in the amount of $2,075,000 which, as of December 31, 2011, were convertible into approximately 199,250 common shares. In January 2012, the debentures were exchanged for units or repaid (see Note 5). In computing diluted net loss per share for 2011, no effect has been given to such shares as their effect would be anti-dilutive.
During the year ended December 31, 2012, upon completion of the various financings, the Company issued warrants to purchase an aggregate of 1,259,932 shares of the Company’s common stock (see Note 11). In addition, in May 2012, the Company issued to employees options to acquire an aggregate of 175,000 shares of the Company’s common stock of which 150,000 were outstanding at December 31, 2012 (see Note 10). In computing diluted net loss per share for 2012, no effect has been given to such options and warrants as their effect would be anti-dilutive.
NOTE 3 – FURNITURE AND EQUIPMENT
Furniture and equipment as of December 31, 2012 and 2011 is summarized as follows:
| 2012 | 2011 | |||||||
| Office furniture and equipment | $ | 78,535 | $ | 42,862 | ||||
| Less: accumulated depreciation | (31,641 | ) | (17,312 | ) | ||||
| $ | 46,894 | $ | 25,550 | |||||
Depreciation expense for the years ended December 31, 2012 and 2011 was $14,329 and $9,300, respectively.
NOTE 4 - RESTRICTED CASH
Restricted cash at December 31, 2012 and 2011 collateralizes a letter of credit in the amount of approximately $60,000 issued in connection with the lease of office space in New York City (see Note 12).
NOTE 5 – 2011 CONVERTIBLE DEBENTURES
On October 7, 2011, concurrently with the Share Exchange, the Company issued secured Convertible Debentures (“Convertible Debentures”) in the amount of $1,625,000 of which $1,125,000 were sold to certain investors for aggregate cash proceeds of $1,065,000, net of selling commissions to a placement agent of $40,000 and $20,000 of legal fees, and $500,000 were exchanged for 8% Notes Payable (“Notes Payable”) issued on September 9, 2011. In addition, 20,000 shares of common stock with the fair market value of $144,000 were issued to a second placement agent. On November 16, the Company issued Convertible Debentures in the amount of $450,000 for aggregate cash proceeds of $436,000, net of selling commissions to a third placement agent of $14,000.
The Convertible Debentures matured on the earlier of (i) one year from the date of issuance or (ii) the date of closing of a private placement of equity, equity equivalent, convertible debt or debt financing in which the Company receives gross proceeds, in one or more transactions, of at least $3,425,000 (a “Subsequent Financing”), which took place on January 20, 2012 (“January 2012 Financing”) (see Note 6). The Convertible Debentures bore interest at 8% per annum and were convertible at the holder’s option into a Subsequent Financing. In the event that a Subsequent Financing has not occurred within 12 months from the date of issuance of the Convertible Debentures, the holder had the option to convert into a number of shares of the Company's common stock equal to 1% of the Company's shares of common stock on a fully diluted basis for every $125,000 of Convertible Debentures (the “Conversion Shares”) or an aggregate of approximately 199,250 shares based on the outstanding shares of the Company common stock as of December 31, 2011.
| F-10 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
Upon the January 2012 Financing (See Note 6), $1,925,000 of debentures were exchanged for Units and the remaining $150,000 of debentures were repaid. As a result of the exchange, $1,925,000 principal amount of debentures are classified as a non-current liability in the accompanying balance sheet at December 31, 2011.
Upon conversion or repayment of the Convertible Debenture, the holder was entitled to receive, at the holder’s option, either (i) a warrant (the “Debenture Warrant”), which has a three year term and is exercisable at the offering price in a Subsequent Financing, to purchase such number of shares of the Company's common stock equal to the principal amount of the Convertible Debenture divided by the offering price in a Subsequent Financing, (the “Warrant Shares”) or (ii) shares of the Company's common stock equal to 33% of the principal amount of the Convertible Debenture divided by the offering price in a Subsequent Financing (the “Incentive Shares”). The Conversion Shares, Warrant Shares and Incentive Shares are entitled to piggyback registration rights. Upon the January 2012 Financing, the holders of the Convertible Debenture elected to receive 13,750 Debenture Warrants exercisable at $20.00 per share with a fair value of $83,289 and 29,700 Incentive Shares valued at $368,280. The value of the Debenture Warrants and Incentive Shares was charged to operations as interest expense in the first quarter of 2012.
In addition to selling commissions paid to the placement agents on the sale of certain Convertible Debentures, the placement agents received warrants that expire in January 2014 and 2015 (“Agents Warrants”), respectively, and are exercisable at the offering price in a Subsequent Financing to purchase shares of the Company's common stock equal to 3% and 9%, respectively, of the gross proceeds delivered by purchasers introduced by such placement agents divided by the purchase price per share in the Subsequent Financing. In the event that the Subsequent Financing has not occurred within 12 months from the date of issuance of the Convertible Debentures, the placement agents were entitled to receive, in lieu of the warrants, shares of common stock equal to 3% and 9%, respectively, of the number of shares of the Company's common stock such purchasers were entitled to receive upon conversion of their Convertible Debentures or an aggregate of approximately 4,400 shares based on the outstanding shares of the Company's common stock as of December 31, 2011.
The Company recognized a liability to placement agents to issue shares of its common stock based on their fair value of approximately $32,000 as of December 31, 2011. Upon the January 2012 Financing, the placement agents become entitled to receive 1,538 warrants exercisable at $20.00 per share with a fair value $6,126, which was charged to operations as interest expense in the first quarter of 2012. Additionally the liability to placement agent of $32,000 was credited to interest expense in the first quarter of 2012.
The fair value of the Debenture and Agents Warrants was determined using the Black Scholes option pricing model with the following assumptions: fair value of the Company’s common stock $12.40 per share determined based on January and March 2012 proceeds; dividends yield 0%; expected terms 2 to 3 years; risk free interest rate: 0.91%; and expected volatility: 73 to 94%.
The following expenses in connection with the issuance of Convertible Debentures were recorded as deferred financing costs: fair value of 20,000 shares of the Company’s common stock issued to the placement agent valued at $144,000, cash payments to the placement agents of $54,000, legal expenses of $20,000 and fair value of the liability to placement agent to issue the Company’s shares of common stock in the amount of $32,000. The deferred financing costs were amortized using the effective interest method over the twelve month term of the Convertible Debentures. During the year ended December 31, 2011, amortization of deferred financing costs amounted to approximately $53,000 and was charged to interest expense in the statement of operations and the remaining balance of $196,166 was charged to operations in connection with the extinguishment of the debentures resulting from their exchange and repayment in 2012.
Pursuant to a Pledge and Security Agreement and Subsidiary Guaranty, the Company granted the Debenture holders a first priority lien on all its assets.
NOTE 6 – JANUARY AND MARCH 2012 FINANCING
On January 20, 2012, the Company issued an aggregate of 172.118 units (“Units”) to certain investors (the “Purchasers”) for aggregate cash proceeds of $2,377,950 and $1,925,000 in previously issued Convertible Debentures of the Company that were exchanged for Units (“January 2012 Financing”). On March 1, 2012, the Company issued an aggregate of 92.5926 Units to certain investors for aggregate cash proceeds of $2,314,815 (“March 2012 Financing”).
| F-11 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
Each Unit had a purchase price of $25,000 per Unit and consisted of one thousand two hundred and fifty (1,250) shares of the Company’s common stock, a Class A Warrant to purchase one thousand two hundred and fifty (1,250) shares of Common Stock (the “Class A Warrants”), and a Class B Warrant to purchase up to one thousand two hundred and fifty (1,250) shares of Common Stock (the “Class B Warrants” and together with the Class A Warrants, the “Warrants”).
The Class A Warrants have an exercise price of $25.00 per share of common stock and will be exercisable for a period of five years from the date of issuance. The warrants had certain anti-dilutive provisions that were set to expire the earlier of i) one year or ii) upon effectiveness of a registration of all shares covered by Class A Warrants, which took place on June 6, 2012. The Company determined the fair value of the Class A Warrants and the Agent Warrants, described below, to be $2,549,684 and $212,235 on the issuance dates and initially classified them as a liability due to transactions which cause an adjustment to the conversion rate (reset provisions) contained in the warrant agreements. On June 6, 2012, upon the Company's registration statement being declared effective by the Securities and Exchange Commission, the reset provisions expired and the Company reclassified $3,938,946, the fair value of the Class A Warrants and Agent Warrants as of that date to equity. The increase of $1,177,026 in fair value of warrants liability was included in results of operations for the year ended December 31, 2012.
The following assumptions were used in the Binomial Lattice model to determine fair value of the Class A Warrants and the Agent Warrants:
| Issuance date January 20 and March 1, 2012 | Expiration date June 6, 2012 | |||||||
| Price of the Company’s common stock | $ | 12.40 | $ | 17.00 | ||||
| Dividend yield | 0 | % | 0 | % | ||||
| Expected terms | 5 – 7 years | 4.6 - 6.7 years | ||||||
| Risk free interest rate | 0.89 - 1.47 | % | 0.73 - 1.11 | % | ||||
| Expected volatility | 96.68 - 96.69 | % | 95.73 | % | ||||
| Expected price at which holders are likely to exercise their warrants | $ | 1.25 | $ | 1.25 | ||||
The Class B Warrants were exercisable automatically on their expiration date by cashless exercise or expire without exercise. In the event that the average of the Company’s daily volume weighted average price was below $15.00 during the 10 trading days after the Announcement Date (as hereinafter defined) (the “Measuring Period”), then the holder was entitled to receive additional shares of the Company’s Common Stock upon the exercise of the Class B Warrants on the expiration date, which is the 12th trading day after the Announcement Date. In the event that the Company’s average daily volume weighted average price was at or above $15.00 during the Measuring Period, the Class B Warrants were to expire unexercised. The Announcement Date was the earlier of (1) the date on which the Company announces via press release the results of the pharmacokinetic study of its TNX-102 drug formulation; or (2) June 1, 2012. On April 5, 2012 the Company issued a press release announcing the results of the pharmacokinetic study of its TNX-102 drug formulation, which is defined as an Announcement Date for the purpose of the Class B Warrants. Based on the Company’s average daily volume weighted average price, which was $34.60 per share, during the Measuring Period, the Class B Warrants expired unexercised.
In connection with the January and March 2012 Financing, the Company paid a placement agent (the “Agent”) an aggregate cash payment of $466,777, which represented an 8% commission and a 2% non-accountable expense allowance of the gross proceeds delivered by Purchasers in the January and March 2012 Financing. In addition, the Agent earned an aggregate of 23,339 warrants to purchase shares of common stock equal to 10% of the gross proceeds delivered by Purchasers in the January and March 2012 Financing (the “Agent Warrants”), which have an exercise price of $25.00 per share of common stock, exercisable for a period of seven years, contained anti-dilution protection and are entitled to piggy-back registration rights. Total expenses related to the financing, including cash and the fair value of warrants given to the Agent, amounted to $706,511, of which $435,713 was charged to additional paid-in capital and $270,798, deemed initially allocable to the warrant liability, was charged to current and other financing costs.
| F-12 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
In connection with the financings, the Company entered into a Registration Rights Agreement with Purchasers. The Company is required to file a registration statement registering for resale the common stock included in the Units and the common stock underlying the Class A Warrants and the Agent Warrants to be filed no later than 60 days from the date of termination of the financings on March 1, 2012 and must be declared effective no later than 120 days from the date of termination of the Financing (June 29, 2012). On April 26, 2012, the Company filed the registration statement, which was declared effective on June 6, 2012. The Company is required to maintain the effectiveness of the registration statement from its effective date unless all securities registered under the registration statement have been sold or are otherwise able to be sold. If the Company failed to comply with the registration statement filing or effective date requirements, the Company was required to pay the investors a fee equal to 1.0% of the Purchaser’s investment, for each 30-day period of delay, subject to a maximum payment of 10% to each Purchaser.
NOTE 7 – 2012 CONVERTIBLE DEBENTURES
On November 14, 2012, the Company sold to accredited investors for aggregate cash proceeds of $390,000, convertible debentures (“Debentures”) in the principal face amount of $390,000 and the Company exchanged $320,000 in previously issued promissory notes of the Company for Debentures in the principal face amount of $320,000.
The previously issued promissory notes were issued between October and November 2012 in the amount of $320,000 in exchange for $320,000 borrowed from six affiliated investors. The Notes bore no interest and were payable on demand.
The Debentures mature on the earlier of (i) November 14, 2013 or (ii) the date of closing of a private placement of equity, equity equivalent, convertible debt or debt financing in which we receive gross proceeds, in one or more transactions, of at least $100,000 (a “Subsequent Financing”). The Debentures bear interest at 8% per annum and are convertible at the holder’s option into either (i) a Subsequent Financing at a price equal to a 25% discount to the price of securities sold in the Subsequent Financing or (ii) shares of the Company’s common stock at a conversion price per share equal to $20.00.
On December 4, 2012, upon completion of a Subsequent Financing, the $710,000 of Debentures were converted into Units at a price of $6.00 per Unit representing a 25% discount to the price ($8.00) of securities sold (the “Financing”). Accordingly, the Company recorded a beneficial conversion feature in connection with the Debentures at the date of conversion of $710,000 as a charge to interest expense and a credit to additional paid in capital.
The beneficial conversion feature, which was contingent on a Subsequent Financing, was computed based on the excess of the number of shares received upon conversion based on the adjusted conversion price ($6.00) over the number of shares that would have been received based on the original conversion price ($20.00) multiplied by the stock price ($10.20) on November 14, 2012, the date the Debentures were issued, limited to the amount of proceeds allocated to the Debentures, or $710,000.
NOTE 8 – DECEMBER 2012 FINANCING
On December 4, 2012, the Company issued an aggregate of 6,404,167 units (“Units”) to certain accredited investors for aggregate cash proceeds of $1,615,000, at a price per Unit of $0.40, and the exchange of $710,000 in previously issued convertible debentures of the Company that were converted into Units at a price of $0.30 per Unit. On December 21, 2012, the Company issued 2,500,000 Units to a single accredited investor for cash proceeds of $1,000,000, at a price per Unit of $0.40. In connection with the Financing, the Company paid an agent a cash payment of $70,000, which represented a 7% commission of the gross proceeds delivered by the investor in the financing.
Each Unit consisted of .05 share of the Company’s common stock, $0.001 par value, a Class A Warrant to purchase .05 share of Common Stock (the “Class A Warrants”), and a Class B Warrant to purchase .05 share of Common Stock (the “Class B Warrants” and together with the Class A Warrants, the “Warrants”).The Class A Warrants have an exercise price of $12.00 per share of Common Stock and will be exercisable for a period of five years from the date of issuance. The Class A Warrants may be exercised on a cashless basis under certain circumstances. The Class B Warrants have an exercise price of $8.00 per share of Common Stock and will be exercisable for a period of one year from the date of issuance.
In connection with the Financing, the Company granted each Purchaser registration rights. The Company is obligated to use its best efforts to cause a registration statement registering for resale the common stock included in the Units and the common stock underlying the Class A Warrants to be filed no later than 60 days (as amended) from the date of termination of the Financing and must be declared effective no later than 120 days from the date of termination of the Financing. Moreover, the Company will maintain the effectiveness of the registration statement from its effective date unless all securities registered under the registration statement have been sold or are otherwise able to be sold pursuant to Rule 144 of the Securities Act of 1933, as amended (the “Securities Act”). If the Company fails to comply with the registration statement filing or effective date requirements, the Company is required to pay the investors a fee equal to 1.0% of the Purchaser’s investment, for each 30-day period of delay, subject to a maximum payment of 10% to each Purchaser. On January 25, 2013, the Company filed the required registration statement.
| F-13 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
NOTE 9 – STOCKHOLDERS' EQUITY
On May 2, 2012, the Company filed amended and restated Articles of Incorporation. Among other changes, the Company increased the number of authorized shares of common stock, $0.001 par value to 150,000,000. Additionally, the Company is now authorized to issue 5,000,000 shares of preferred stock, $0.001 par value with such designations, preferences and participating, optional or other special rights and qualifications, limitations or restrictions thereof as shall be determined by the Company’s Board of Directors.
NOTE 10 – SHARE BASED COMPENSATION
2010 Stock Plan
In June and August 2010, respectively, the Board of Directors and stockholders of Tonix Pharmaceuticals, Inc. approved, and in December 2010 and February 2011, the Board of Directors amended, the terms and provisions of the 2010 Stock Plan (the "2010 Plan") whereby the Company reserved 228,232 shares of its Common Stock for issuance pursuant to the 2010 Plan. The 2010 Plan allowed for grants of options to purchase shares of Common Stock and awards of restricted Common Stock to employees, officers, directors, consultants and advisors of the Company.
No options were granted under the 2010 Plan. Following is a summary of activity for the year ended December 31, 2011, with respect to restricted stock granted under the 2010 Plan:
| Weighted | ||||||||
| Number of | Average | |||||||
| Restricted | Grant-Date | |||||||
| Nonvested Restricted Stock | Shares | Fair Value | ||||||
| Nonvested at December 31, 2010 | 84,893 | $ | 4.60 | |||||
| Granted | 18,436 | $ | 4.60 | |||||
| Vested prior to Share Exchange | (28,243 | ) | $ | 4.60 | ||||
| Vested pursuant to Share Exchange | (69,849 | ) | $ | 4.60 | ||||
| Forfeited | (5,237 | ) | $ | 4.60 | ||||
| Nonvested at December 31, 2011 | 0 | $ | 0 | |||||
Restricted stock is not considered to be issued until the stock vests.
The Company recognized share-based compensation expense of $139,063 prior to the Share Exchange and remaining expense of $296,588 was recognized on October 7, 2011, the date of the Share Exchange, upon which all non vested restricted shares vested and the 2010 Plan ceased to exist.
2012 Incentive Stock Option Plan
On February 12, 2012, the Company’s Board of Directors approved the 2012 Incentive Stock Option Plan (the “2012 Plan”). The 2012 Plan provides for the issuance of options to purchase up to 200,000 shares of the Company’s common stock to officers, directors, employees and consultants of the Company. Under the terms of the 2012 Plan, the Company may issue Incentive Stock Options as defined by the Internal Revenue Code to employees of the Company only and nonstatutory options. The Board of Directors of the Company determines the exercise price, vesting and expiration period of the grants under the 2012 Plan. However, the exercise price of an Incentive Stock Option should not be less than 110% of fair value of the common stock at the date of the grant for a 10% or more stockholder and 100% of fair value for a grantee who is not 10% stockholder. The fair value of the common stock is determined based on quoted market price or in absence of such quoted market price, by the Board of Directors in good faith. Additionally, the vesting period of the grants under the 2012 Plan should not be more than five years and expiration period not more than ten years. The Company reserved 200,000 shares of its common stock for future issuance under the terms of the 2012 Plan. On May 9, 2012, 175,000 options had been granted under the 2012 Plan (of which 25,000 were subsequently canceled and 150,000 are outstanding at December 31, 2012) with an exercise price of $30.00, a 10 year life and fair value of $23.50. The options vest 1/3rd on May 9, 2013 and 1/36th on the 9th of each month thereafter for 24 months.
| F-14 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
The Company measures the fair value of stock options on the date of grant, based on a Binomial option pricing model using certain assumptions discussed in the following paragraph, and the closing market price of the Company's common stock on the date of the grant. Stock options granted vest over a three year period and expire ten years from the date of grant. Share-based compensation expense related to awards is amortized over the applicable vesting periods using the straight-line method. Share-based compensation expense of $865,158 was recognized for the year ended December 31, 2012.
The assumptions used in the valuation of stock options granted during the year ended December 31, 2012 were as follows:
| Risk-free interest rate | 1.87 | % | ||
| Expected term of option | 6.5 years | |||
| Expected stock price volatility | 95.89 | % | ||
| Expected dividend yield | $ | 0.0 |
The risk-free rate of return is based on the yield of Daily U.S. Treasury Yield Curve Rates with terms equal to the expected term of the options as of the grant date. The expected term of options is determined using the simplified method and the expected stock price volatility is based on comparable companies’ historical stock price volatility since the Company does not have sufficient historical exercise data because its equity shares have been publicly traded for only a limited period of time.
As of December 31, 2012, the Company had approximately $2,742,000 of total unrecognized compensation cost related to non-vested awards granted under the Company’s 2012 Plan, which the Company expects to recognize over approximately a three-year period.
A summary of the stock options activity and related information for the 2012 Incentive Stock Option Plan for the year ended December 31, 2012 is as follows:
| Shares | Weighted-Average Exercise Price | Weighted-Average Remaining Contractual Term | Aggregate Intrinsic Value | |||||||||||||
| Outstanding at January 1, 2012 | - | |||||||||||||||
| Grants | 175,000 | $ | 30.00 | 10.00 | $ | - | ||||||||||
| Exercised | - | |||||||||||||||
| Forfeitures or expirations | (25,000 | ) | 30.00 | |||||||||||||
| Outstanding at December 31, 2012 | 150,000 | $ | 30.00 | 9.35 | $ | - | ||||||||||
| Vested and expected to vest at December 31, 2012 | 150,000 | $ | 30.00 | 9.35 | $ | - | ||||||||||
| Exercisable at December 31, 2012 | - | $ | - | - | $ | - | ||||||||||
The aggregate intrinsic value in the preceding tables represents the total pretax intrinsic value, based on options with an exercise price less than the Company’s closing stock price of $11.00 as of December 31, 2012, which would have been received by the option holders had those option holders exercised their options as of that date.
NOTE 11 – STOCK WARRANTS
The following table summarizes information with respect to outstanding warrants to purchase common stock of the Company, all of which were exercisable, at December 31, 2012:
| Exercise | Number | Expiration | ||||||
| Price | Outstanding | Date | ||||||
| $ | 8.00 | 445,209 | December 2013 | |||||
| 12.00 | 445,209 | December 2017 | ||||||
| $ | 20.00 | 15,288 | January 2014 to January 2015 | |||||
| 25.00 | 354,228 | January 2017 to March 2019 | ||||||
| 1,259,934 | ||||||||
| F-15 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
On January 20, 2012, the Company issued an aggregate of 13,750 and 1,538 warrants to purchase the Company's common stock at an exercise price of $20.00 per share expiring five and seven years from the date of issuance to convertible debenture holders and debenture placement agents, respectively (see Note 5).
In connection with the January and March 2012 Financing, the Company issued to investors an aggregate of 215,148 and 115,741 warrants, respectively, to purchase the Company's common stock at an exercise price of $25.00 per share expiring five years from the date of issuance. In addition, the Company issued an aggregate of 11,765 and 11,574 warrants to purchase the Company's common stock at an exercise price of $25.00 per share expiring seven years from the date of issuance to placement agents. These warrants contained certain anti-dilutive provisions and are covered under a registration rights agreement (see Note 6).
In connection with the December 2012 Financing, the Company issued to investors of 445,209 and 445,209 Class A warrants and Class B warrants, respectively to purchase the Company's common stock. The Class A warrant is exercisable at $12.00 per share expiring five years from the date of issuance and may be exercised on a cashless basis under certain circumstances. The Class B warrant is exercisable at $8.00 per share expiring one year from the date of issuance. These warrants are covered under a registration rights agreement (see Note 8).
NOTE 12 - COMMITMENTS
Operating leases
On September 28, 2010, the Company entered into a five-year lease for office space in New York City, with monthly payments escalating from approximately $10,000 in first year to approximately $11,000 in fifth year. The Company received a rent credit of $9,420 in each of the months of November 2010, December 2010 and January 2011. The Company has posted a letter of credit in the amount of approximately $60,000 for the benefit of the landlord which is collateralized by a money market account (see Note 4 - Restricted Cash).
Future minimum lease payments under the operating lease are as follows:
| Year Ending December 31, | ||||
| 2013 | $ | 127,889 | ||
| 2014 | 131,513 | |||
| 2015 | 100,719 | |||
| $ | 360,121 | |||
Rent expense charged to operations, which differs from rent paid due to the rent credits and to increasing amounts of base rent, is calculated by allocating total rental payments on a straight-line basis over the term of the lease. During the years ended December 31, 2012 and 2011, rent expense was $116,732 and $128,228, respectively and as of December 31, 2012 and 2011 deferred rent payable was $26,156 and $29,083, respectively.
Consulting agreements
In June 2010, the Company entered into a two-year consulting agreement with L & L Technologies, LLC (“L&L”), an entity controlled by a member of the Company’s Board of Directors, for scientific and medical consulting services. In consideration for such services, L&L received $96,000 per annum and 51,310 shares of restricted common stock which were granted at the inception of the agreement. The restricted shares vest as follows: 25% on the grant date (June 4, 2010) and 25% on each of the first and second annual anniversaries of the grant date and, if the consulting agreement is renewed, 25% on the third anniversary of the grant date. Vesting of the unvested 25,655 restricted shares accelerated on October 7, 2011, the date of the Share Exchange. The consulting agreement expired in June 2012.
| F-16 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
In June 2010, the Company entered into a two-year consulting agreement with Lederman & Co., LLC (“Lederman & Co”), an entity controlled by a member of the Company’s Board of Directors, for clinical development, strategic, management and operational consulting services. In consideration for such services, Lederman & Co will receive $250,000 per annum and 13,090 shares of restricted common stock which were granted at the inception of the agreement. The consulting agreement renews automatically for subsequent terms of one year at $250,000 per annum. The restricted shares vest as follows: 20% on the grant date (June 4, 2010) and 20% on each of the first and second anniversaries of the grant date and, if the consulting agreement is renewed, 20% on each of the third and fourth anniversaries of the grant date. Vesting of the unvested 7,855 restricted shares accelerated on October 7, 2011, the date of the Share Exchange. On August 1, 2011, the cash compensation was reduced to $127,000 per annum. On February 1, 2012, the cash compensation was increased to $250,000 per annum.
During 2011 and 2012, the Company entered into contracts with various contract research organizations for which there are outstanding commitments aggregating approximately $650,000 at December 31, 2012 for future work to be performed.
Employment agreements
In October 2011, the position of Vice President of Strategy was eliminated and in conjunction with this event, the Company paid $37,500 in December 2011 in exchange for the release from future obligations. In February 2012, the Company terminated its employment agreement with its Chief Financial Officer and in accordance with the agreement paid such officer approximately $88,000.
Effective April 1, 2012, the Company entered into an employment agreement (the “Gershell Agreement”) with Dr. Gershell to serve as Chief Financial Officer. The base salary under the Gershell Agreement is $175,000 per annum, which shall increase to $325,000 per annum upon our consummation of an equity sale of securities in excess of $20 million (the “Gershell Threshold”). The Gershell Agreement provides for at-will employment and can be terminated at any time by either party, provided, however, that if we terminate Dr. Gershell for any reason other than cause (as defined in the Gershell Agreement), then Dr. Gershell shall be entitled to six weeks of severance, which severance payment shall increase to six months if such termination occurs after the Gershell Threshold. In addition, Dr. Gershell is entitled to participate in any and all benefit plans, from time to time, in effect for our employees, along with vacation, sick and holiday pay in accordance with its policies established and in effect from time to time.
Effective April 1, 2012, the Company entered into an employment agreement (the “Daugherty Agreement”) with Dr. Daugherty to serve as Senior Director of Drug Development. The base salary under the Daugherty Agreement is $140,000 per annum, which shall increase to $220,000 per annum upon our consummation of an equity sale of securities in excess of $20 million (the “Daugherty Threshold”). The Daugherty Agreement provides for at-will employment and can be terminated at any time by either party, provided, however, that if we terminate Dr. Daugherty for any reason other than cause (as defined in the Daugherty Agreement), then Dr. Daugherty shall be entitled to six weeks of severance, which severance payment shall increase to six months if such termination occurs after the Daugherty Threshold. In addition, Dr. Daugherty is entitled to participate in any and all benefit plans, from time to time, in effect for our employees, along with vacation, sick and holiday pay in accordance with its policies established and in effect from time to time.
On October 26, 2012, the Company elected to voluntarily terminate Benjamin Selzer as Chief Operating Officer, Secretary and Treasurer, effective immediately and under the terms of his employment agreement, no severance was paid. In conjunction with the termination, 25,000 unvested options previously issued to Mr. Selzer were cancelled.
NOTE 13 - INCOME TAXES
There is no provision for federal or state income taxes for the years ended December 31, 2012 and 2011 since the Company has established a valuation allowance equal to the total deferred tax asset related to losses incurred during such periods.
| F-17 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
Deferred tax assets and liabilities and related valuation allowance as of December 31, 2012 and 2011 are as follows:
| December 31, | ||||||||
| 2012 | 2011 | |||||||
| Deferred tax assets: | ||||||||
| Organization costs | $ | - | $ | 733 | ||||
| Research and development credit carryforward (1) | 6,188 | 6,188 | ||||||
| Net operating loss carryforwards | 5,207,759 | 2,329,829 | ||||||
| Other | 147,003 | 132,482 | ||||||
| Total deferred tax assets | 5,360,950 | 2,469,232 | ||||||
| Valuation allowance | (5,360,950 | ) | (2,469,232 | ) | ||||
| Net deferred tax assets | $ | 0 | $ | 0 | ||||
| (1) | The Company has incurred research and development (“R&D”) expenses, a portion of which may qualify for tax credits. The Company has not conducted an R&D credit study to quantify the amount of credits and has not claimed an R&D credit on its federal tax returns filed except for $6,188 in 2007. The Company may conduct the study in future years and may establish the R&D credit carryforward for prior years. In such event, the net operating loss carryforward will be correspondingly reduced by the amount of the credit. |
Based on the Company's historical losses and its expectation of continuation of losses for the foreseeable future, the Company has determined that it is not more likely than not that the deferred tax assets will be realized and accordingly, has provided a valuation allowance. The increase in the valuation allowance for the years ended December 31, 2012 and 2011 was $2,891,718 and $1,380,642, respectively.
At December 31, 2012, the Company has available unused net operating loss carryforwards of approximately $12.3 million that expire from 2027 to 2032 for federal tax purposes and the same amount for New Jersey state tax purposes, which expire from 2014 to 2019. The Company also has approximately $11.6 million of net operating loss carryforwards for New York state and New York City purposes expiring from 2030 to 2032. At December 31, 2012, the Company has a research and development carryforward of $6,188 for federal tax purposes that expires in 2027. These net operating loss and research and development credit carryforwards may be subject to annual limitations in their use in accordance with IRC Section 382. Accordingly, the extent to which such carryforwards can be used to offset future taxable income may be limited.
The Company's federal and state tax returns remain open and subject to examination by the tax authorities for the tax years 2009 and after.
A reconciliation of the effect of applying the federal statutory rate and the effective income tax rate used to calculate the Company's income tax provision is as follows:
| Year Ended December 31, | ||||||||
| 2012 | 2011 | |||||||
| Statutory federal income tax | (34.0 | )% | (34.0 | )% | ||||
| State income tax, net of federal tax effect | (10.5 | )% | (5.9 | )% | ||||
| Permanent difference | 13.9 | % | 5.0 | % | ||||
| Increase in valuation allowance | 30.6 | % | 34.9 | % | ||||
| Income tax provision | 0 | % | 0 | % | ||||
NOTE 14 - RELATED PARTY TRANSACTIONS
Dr. Seth Lederman, our Chief Executive Officer and Chairman of the Board, and Dr. Donald Landry, one of our directors, are the primary founders of the Company. We have entered into various transactions with several companies under their control, including L&L, Plumbline, Targent Pharmaceuticals, LLC and Lederman & Co (see Note 12 – Consulting Agreements). Total expenses paid under these agreements were $300,583 and $294,750 during the years ended December 31, 2012 and 2011, respectively.
On September 9, 2011, the Company sold $500,000 principal amount of 8% convertible notes (the “Notes”) to members of the board of directors and their related parties. The Notes were due one year from the date of issuance, and were exchangeable for a future financing (the “New Financing”) at the option of the holders. Interest is payable on either the maturity date or on the date the Notes are exchanged into the New Financing, or such accrued interest can be converted into the New Financing. On October 7, 2011, the Notes were exchanged into debentures issued by the Company concurrently with the Share Exchange (see Note 5). In January 2012, the related party companies received interest on the convertible notes in the aggregate amount of $6,183.
| F-18 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2012 AND 2011
Between October and November 2012, the Company issued promissory notes in the amount of $320,000 in exchange for $320,000 borrowed from six affiliated investors. The Notes bear no interest and were payable on demand.
On November 14, 2012, the Company sold to officers, members of the board of directors and their related parties for aggregate cash proceeds of $390,000, debentures (the “Debentures”) in the principal face amount of $390,000 and the exchange of the promissory notes described above for Debentures in the principal face amount of $320,000. In December 2012, the Debentures were exchanged for the December 2012 Units at a conversion price of $6.00 per share. Interest expense on the Debentures for the year ended December 31, 2012 was $3,155 (See Note 8).
NOTE 15 - SUBSEQUENT EVENTS
On February 12, 2013, the Company’s Board of Directors approved the Amended and Restated 2012 Incentive Stock Option Plan (the “Amended and Restated 2012 Plan”), subject to stockholder approval. The Amended and Restated 2012 Plan includes amendments which: 1) authorize 550,000 shares of the Company’s common stock for issuance; and 2) prohibit the issuance of any options with terms or features that would cause the options to be nonqualified deferred compensation that fails to comply with, or be exempt from, Section 409A of the Internal Revenue Code of 1986, as amended.
On February 12, 2013, 226,500 options were granted under the Amended and Restated 2012 Plan, with an exercise price of $10.20 and a 10 year life. The exercise price is equal to the volume weighted average price of the Company’s common stock during the immediate prior 30 calendar day period. The options vest 1/3rd on February 12, 2014 and 1/36th on the 12th of each month thereafter for 24 months.
On May 1, 2013, the Company filed an amendment to its Articles of Incorporation and effected a 1-for-20 reverse stock split of its issued and outstanding shares of common stock, $0.001 par value, whereby 43,182,599 outstanding shares of the Company’s common stock were exchanged for 2,159,156 newly issued shares of the Company's common stock. Under the terms of the reverse stock split, fractional shares issuable to stockholders were rounded up to the nearest whole share, resulting in a reverse split slightly less than 1-for-20 in the aggregate. All per share amounts and number of shares (other than authorized shares) in the consolidated financial statements and related notes have been retroactively restated to reflect the reverse stock split resulting in the transfer of $41,024 from common stock to additional paid in capital at December 31, 2012.
| F-19 |
| TONIX PHARMACEUTICALS HOLDING CORP. |
| (a development stage company) |
| CONDENSED CONSOLIDATED BALANCE SHEETS |
| March 31, | December 31, | |||||||
| 2013 | 2012 | |||||||
| (unaudited) | ||||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash | $ | 423,739 | $ | 1,785,390 | ||||
| Prepaid expenses and other | 67,691 | 224,659 | ||||||
| Total current assets | 491,430 | 2,010,049 | ||||||
| Furniture and equipment, net | 42,756 | 46,894 | ||||||
| Restricted cash | 60,289 | 60,267 | ||||||
| Total assets | $ | 594,475 | $ | 2,117,210 | ||||
| LIABILITIES AND STOCKHOLDERS' (DEFICIENCY) EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable, including $26,757 and $6,809 to related parties as of March 31, 2013 and December 31, 2012, respectively | $ | 870,096 | $ | 825,837 | ||||
| Accrued expenses | 346,692 | 309,800 | ||||||
| Accrued interest, related party | - | 3,155 | ||||||
| Total current liabilities | 1,216,788 | 1,138,792 | ||||||
| Deferred rent payable | 17,424 | 19,710 | ||||||
| Total liabilities | 1,234,212 | 1,158,502 | ||||||
| Stockholders' (deficiency) equity: | ||||||||
| Preferred stock, $0.001 par value; 5,000,000 authorized; none issued or outstanding | - | - | ||||||
| Common stock, $0.001 par value; 150,000,000 authorized; 2,159,156 shares issued and outstanding as of March 31, 2013 and December 31, 2012 | 2,159 | 2,159 | ||||||
| Additional paid in capital | 17,203,587 | 16,800,829 | ||||||
| Deficit accumulated during development stage | (17,845,483 | ) | (15,844,280 | ) | ||||
| Total stockholders' (deficiency) equity | (639,737 | ) | 958,708 | |||||
| Total liabilities and stockholders' (deficiency) equity | $ | 594,475 | $ | 2,117,210 | ||||
See the accompanying notes to condensed consolidated financial statements
| F-20 |
| TONIX PHARMACEUTICALS HOLDING CORP. |
| (a development stage company) |
| CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS |
| (unaudited) |
| From June 7, 2007 | ||||||||||||
| (date of inception) | ||||||||||||
| Three months ended March 31, | through | |||||||||||
| 2013 | 2012 | March 31, 2013 | ||||||||||
| COSTS AND EXPENSES: | ||||||||||||
| Research and development | $ | 740,629 | $ | 397,628 | $ | 5,275,891 | ||||||
| General and administrative | 1,260,596 | 762,737 | 9,593,945 | |||||||||
| 2,001,225 | 1,160,365 | 14,869,836 | ||||||||||
| Operating Loss | (2,001,225 | ) | (1,160,365 | ) | (14,869,836 | ) | ||||||
| Gain on extinguishment of debt | - | - | 7,908 | |||||||||
| Other income | - | - | 1,875 | |||||||||
| Change in fair value of warrant liability | - | 47,023 | (1,177,026 | ) | ||||||||
| Interest and other financing costs, net | 22 | (901,646 | ) | (1,808,404 | ) | |||||||
| NET LOSS | $ | (2,001,203 | ) | $ | (2,014,988 | ) | $ | (17,845,483 | ) | |||
| Net loss per common share, basic and diluted | $ | (0.93 | ) | $ | (1.27 | ) | ||||||
| Weighted average common shares outstanding, basic and diluted | 2,159,156 | 1,582,544 | ||||||||||
See the accompanying notes to condensed consolidated financial statements
| F-21 |
| TONIX PHARMACEUTICALS HOLDING CORP. |
| (a development stage company) |
| CONDENSED CONSOLIDATED STATEMENT OF STOCKHOLDERS' EQUITY (DEFICIENCY) |
| For the Three Months Ended March 31, 2013 |
| (unaudited) |
| Deficit | ||||||||||||||||||||
| Accumulated | ||||||||||||||||||||
| Additional | During | |||||||||||||||||||
| Common stock | Paid in | Development | ||||||||||||||||||
| Shares | Amount | Capital | Stage | Total | ||||||||||||||||
| Balance at December 31, 2012 | 2,159,156 | $ | 2,159 | $ | 16,800,829 | $ | (15,844,280 | ) | $ | 958,708 | ||||||||||
| Stock based compensation | - | - | 392,323 | - | 392,323 | |||||||||||||||
| Warrants issued for services rendered | - | - | 10,435 | - | 10,435 | |||||||||||||||
| Net loss | - | - | - | (2,001,203 | ) | (2,001,203 | ) | |||||||||||||
| Balance at March 31, 2013 | 2,159,156 | $ | 2,159 | $ | 17,203,587 | $ | (17,845,483 | ) | $ | (639,737 | ) | |||||||||
See the accompanying notes to the condensed consolidated financial statements
| F-22 |
| TONIX PHARMACEUTICALS HOLDING CORP. |
| (a development stage company) |
| CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS |
| (unaudited) |
| From June 7, 2007 | ||||||||||||
| (date of inception) | ||||||||||||
| Three months ended March 31, | Through | |||||||||||
| 2013 | 2012 | March 31, 2013 | ||||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES: | ||||||||||||
| Net loss | $ | (2,001,203 | ) | $ | (2,014,988 | ) | $ | (17,845,483 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation | 4,138 | 2,354 | 35,779 | |||||||||
| Amortization and write off of deferred financing costs | - | 196,166 | 249,543 | |||||||||
| Non cash interest, consisting of beneficial conversion feature in connection with convertible debentures | - | 426,152 | 710,000 | |||||||||
| Non cash interest, consisting of common stock and warrants issued in connection with convertible debentures | - | 81,337 | 426,152 | |||||||||
| Non-cash financing costs related to January and March 2012 financing | - | - | 81,337 | |||||||||
| Warrants issued for services rendered | 10,435 | - | 10,435 | |||||||||
| Stock based compensation | 392,323 | - | 1,944,194 | |||||||||
| Change in fair value of warrant liability | - | (47,023 | ) | 1,177,026 | ||||||||
| Common stock issued in exchange for intellectual property | - | - | 383,250 | |||||||||
| Gain on extinguishment of debt | - | - | (7,908 | ) | ||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Prepaid expenses | 156,968 | 38,383 | (67,691 | ) | ||||||||
| Accounts payable | 44,259 | (512,790 | ) | 870,096 | ||||||||
| Accrued interest | (3,155 | ) | (35,195 | ) | 3,111 | |||||||
| Accrued expenses | 35,993 | 88,503 | 440,058 | |||||||||
| Deferred rent payable | (1,387 | ) | (513 | ) | 24,769 | |||||||
| Net cash used in operating activities | (1,361,629 | ) | (1,777,614 | ) | (11,565,332 | ) | ||||||
| CASH FLOWS FROM INVESTING ACTIVITIES: | ||||||||||||
| Purchase of furniture and fixtures | - | - | (78,535 | ) | ||||||||
| Payment of restricted cash and interest earned on restricted cash | (22 | ) | (22 | ) | (60,289 | ) | ||||||
| Net cash used in investing activities | (22 | ) | (22 | ) | (138,824 | ) | ||||||
| CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||||||
| Proceeds from demand notes | - | - | 480,000 | |||||||||
| Proceeds from other notes payable | - | - | 1,020,000 | |||||||||
| Proceeds, net of expenses of $24,000 from Convertible Debentures | - | - | 1,891,000 | |||||||||
| Repayment of Convertible Debentures | - | (150,000 | ) | (150,000 | ) | |||||||
| Proceeds, net of expenses of $374,870, from sale of units consisting of common stock and warrants | - | 4,387,895 | 6,932,894 | |||||||||
| Proceeds from the sale of capital stock | - | - | 1,954,001 | |||||||||
| Net cash provided by financing activities | - | 4,237,895 | 12,127,895 | |||||||||
| Net (decrease) increase in cash | (1,361,651 | ) | 2,460,259 | 423,739 | ||||||||
| Cash, beginning of the period | 1,785,390 | 41,123 | - | |||||||||
| Cash, end of period | $ | 423,739 | $ | 2,501,382 | $ | 423,739 | ||||||
| Supplemental disclosures of cash flow information: | ||||||||||||
| Interest paid | $ | 3,155 | $ | 35,195 | $ | 38,350 | ||||||
| Non cash investing and financing activities: | ||||||||||||
| Senior convertible notes exchanged for preferred shares | $ | - | $ | - | $ | 200,000 | ||||||
| Capital contribution of accrued interest | $ | - | $ | - | $ | 26,836 | ||||||
| Demand notes together with accrued interest converted into capital stock | $ | - | $ | - | $ | 549,078 | ||||||
| Common stock issued for deferred financing costs | $ | - | $ | - | $ | 144,000 | ||||||
| Exchange of Notes Payable for Convertible Debenture | $ | - | $ | - | $ | 820,000 | ||||||
| Warrants Liability reclassified to Stockholders' Equity | $ | - | $ | - | $ | 3,938,946 | ||||||
| Exchange of Convertible Debenture for Units consisting of common stock and warrants | $ | - | $ | - | $ | 2,635,000 | ||||||
See the accompanying notes to condensed consolidated financial statements
| F-23 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2013 AND 2012 (UNAUDITED)
NOTE 1 – BUSINESS AND RECAPITALIZATION
Tonix Pharmaceuticals Holding Corp., through its wholly owned subsidiary Tonix Pharmaceuticals, Inc., or Tonix Sub, is attempting to develop safer and more effective versions of widely prescribed central nervous system ("CNS") drugs. While some new applications can use the commercially available form of the drug, in other cases, reformulating the active ingredient improves its safety or effectiveness in treating the CNS condition. When formal development programs have proven successful in clinical tests, Tonix Sub intends to seek marketing approval from the Food and Drug Administration ("FDA").
On August 16, 2010, Tonix Sub formed Krele LLC ("Krele") in the state of Delaware. Krele is a limited liability corporation whose sole member is Tonix Sub. Krele was established to commercialize products that are generic versions of predicate new drug application products or versions of drug efficacy study implementation products. The Company expects that its relationship to Krele will be similar to that of several other pharmaceutical companies and their subsidiaries that market generic versions of the parent's branded products at different periods in their product life-cycle.
On October 7, 2011, Tonix Sub (formerly Krele Pharmaceuticals, Inc. incorporated on June 7, 2007 in the State of Delaware) and a publicly traded non-operating shell company Tamandare Explorations Inc. (“Tamandare”), incorporated under the laws of the State of Nevada, along with certain other parties executed and consummated a share exchange agreement (the “Share Exchange”). Pursuant to the Share Exchange, each share of Tonix Sub’s common stock was exchanged for 0.045 shares of Tamandare’s common stock and each share of Tonix Sub’s Series A and B preferred stock was exchanged for 0.24 shares of Tamandare’s common stock. Upon completion of the Share Exchange, the Tonix Sub shareholders, including holders of restricted shares, which were subject to accelerated vesting, received in exchange for all of their shares, an aggregate of 1,133,334 shares of Tamandare’s common stock and Tamandare’s existing stockholders retained 200,000 shares of common stock. The 1,133,334 shares issued to the Tonix Sub shareholders constituted approximately 85% of Tamandare’s 1,333,334 issued and outstanding shares of common stock after the Share Exchange. Upon completion of the Share Exchange, Tonix Sub became Tamandare’s wholly-owned subsidiary and in October 2011 Tamandare was renamed Tonix Pharmaceuticals Holding Corp. As the owners and management of Tonix Sub obtained voting and operating control of Tamandare after the Share Exchange and Tamandare was non-operating, had no assets or liabilities and did not meet the definition of a business, the transaction has been accounted for as a recapitalization of Tonix Sub, accompanied by the issuance of its common stock for outstanding common stock of Tamandare, which was recorded at a nominal value. The accompanying financial statements and related notes give retroactive effect to the recapitalization as if it had occurred on June 7, 2007 (inception date) and accordingly all share and per share amounts have been adjusted.
Tonix Pharmaceutical Holding Corp. and its subsidiaries are hereafter referred to as the “Company”.
NOTE 2 – SIGNIFICANT ACCOUNTING POLICIES
Interim Financial Statements
The unaudited condensed consolidated interim financial statements of the Company have been prepared in accordance with accounting principles generally accepted in the United States (“GAAP”) for interim financial information and the instructions to Form 10-Q and Rule 8-03 of Regulation S-X. Accordingly, they do not include all of the information and footnotes required by GAAP for complete financial statements. In the opinion of management, all adjustments (consisting of normal recurring accruals) considered necessary for a fair presentation have been included.
The condensed consolidated balance sheet as of December 31, 2012 contained herein has been derived from audited financial statements.
Operating results for the three months ended March 31, 2013 are not necessarily indicative of results that may be expected for the year ending December 31, 2013. These condensed consolidated financial statements should be read in conjunction with the audited consolidated financial statements and notes thereto for the year ended December 31, 2012 included in the Company’s Annual Report on Form 10-K, filed with the Securities and Exchange Commission (“SEC”) on March 11, 2013.
| F-24 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2013 AND 2012 (UNAUDITED)
Basis of presentation
As the Company is devoting substantially all of its efforts to establishing a new business, and while planned principal operations have commenced, there has been no revenue generated from sales, license fees or royalties; the Company is considered a development stage enterprise. Accordingly, the Company’s consolidated financial statements are presented in accordance with authoritative accounting guidance related to a development stage enterprise. Financial position, results of operations and cash flows of a development stage enterprise are presented in conformity with generally accepted accounting principles that apply to established operating enterprises.
As a development stage enterprise, the Company's primary efforts are devoted to conducting research and development for the treatment of CNS diseases. The Company has experienced net losses and negative cash flows from operations since inception and expects these conditions to continue for the foreseeable future operations. In addition, the Company has working capital and stockholders' deficiencies as of March 31, 2013. The Company requires additional financing, for which there are no existing commitments, to fund its working capital deficiency and future operations. Further, the Company does not have any commercial products available for sale and there is no assurance that if approval of its products is received that the Company will be able to generate cash flow to fund operations. In addition, there can be no assurance that the Company's research and development will be successfully completed or that any product will be approved or commercially viable.
The above factors raise substantial doubt as to the Company's ability to continue as a going concern. The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern and do not include any adjustments that may result from the outcome of this uncertainty.
The Company expects that cash used in operations will increase significantly over the next several years and it is the Company's intent to raise additional capital to complete the development and commercialization of its current product candidates through equity or debt financing. There can be no assurance that such funds, if available at all, can be obtained on terms reasonable to the Company. If the Company is unsuccessful in raising additional capital it will need to reduce costs and may be required to reduce or cease operations.
Use of estimates
The preparation of financial statements in accordance with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities and expenses and disclosures of contingent assets and liabilities at the date of the financial statements. Actual results could differ from those estimates. Significant estimates include the useful life of fixed assets and assumptions used in the fair value of stock-based compensation.
Research and development costs
The Company outsources its research and development efforts and expenses these costs as incurred, including the cost of manufacturing products for testing, licensing fees and costs associated with planning and conducting clinical trials. The value ascribed to patents and other intellectual property acquired was expensed in 2007 and 2010 as research and development costs, as it related to particular research and development projects and had no alternative future uses.
Income taxes
Income tax provisions or benefits for interim periods are computed based on the Company’s estimated annual effective tax rate. Based on the Company's historical losses and its expectation of continuation of losses for the foreseeable future, the Company has determined that it is more likely than not that deferred tax assets will not be realized and, accordingly, has provided a full valuation allowance. As the Company anticipates or anticipated that its net deferred tax assets at December 31, 2013 and 2012 would be fully offset by a valuation allowance, there is no federal or state income tax benefit for the periods ended March 31, 2013 and 2012 related to losses incurred during such periods.
Per share data
Basic and diluted net loss per common share is calculated by dividing net loss, by the weighted average number of outstanding shares of common stock, adjusted to give effect to a 1-for-20 reverse stock split (see Note 3).
As of March 31, 2013, there are outstanding warrants to purchase an aggregate of 1,270,732 shares of the Company’s common stock (see Note 5). In addition, the Company has issued to employees, options to acquire shares of the Company’s common stock of which 376,500 were outstanding at March 31, 2013 (see Note 4). In computing diluted net loss per share for the three months ended March 31, 2013 and 2012, no effect has been given to such options and warrants as their effect would be anti-dilutive.
| F-25 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2013 AND 2012 (UNAUDITED)
NOTE 3 – REVERSE STOCK SPLIT
On May 1, 2013, the Company filed an amendment to its Articles of Incorporation and effected a 1-for-20 reverse stock split of its issued and outstanding shares of common stock, $0.001 par value, whereby 43,182,599 outstanding shares of the Company’s common stock were exchanged for 2,159,156 newly issued shares of the Company's common stock. Under the terms of the reverse stock split, fractional shares issuable to stockholders were rounded up to the nearest whole share, resulting in a reverse split slightly less than 1-for-20 in the aggregate. All per share amounts and number of shares (other than authorized shares) in the consolidated financial statements and related notes have been retroactively restated to reflect the reverse stock split resulting in the transfer of $41,024 from common stock to additional paid in capital at March 31, 2013 and December 31, 2012.
NOTE 4 – STOCK OPTIONS
2012 Incentive Stock Option Plan
On February 12, 2012, the Company’s Board of Directors approved the 2012 Incentive Stock Option Plan (the “2012 Plan”). The 2012 Plan provides for the issuance of options to purchase up to 200,000 shares of the Company’s common stock to officers, directors, employees and consultants of the Company. Under the terms of the 2012 Plan, the Company may issue Incentive Stock Options as defined by the Internal Revenue Code to employees of the Company and may also issue nonstatutory options to employees and others. The Board of Directors of the Company determines the exercise price, vesting and expiration period of the grants under the 2012 Plan. However, the exercise price of an Incentive Stock Option may not be less than 110% of fair value of the common stock at the date of the grant for a 10% or more stockholder and 100% of fair value for a grantee who is not a 10% stockholder. The fair value of the common stock is determined based on quoted market price or in absence of such quoted market price, by the Board of Directors in good faith. Additionally, the vesting period of the grants under the 2012 Plan may not be more than five years and expiration period not more than ten years. The Company reserved 200,000 shares of its common stock for future issuance under the terms of the 2012 Plan. On May 9, 2012, 175,000 options had been granted under the 2012 Plan (of which 150,000 were outstanding at March 31, 2013) with an exercise price of $30.00, a 10 year life and fair value of $23.50. The options vest 1/3rd on May 9, 2013 and 1/36th on the 9th of each month thereafter for 24 months.
On February 12, 2013, the Company’s board of directors approved the Amended and Restated 2012 Incentive Stock Option Plan (the “Amended and Restated 2012 Plan”) to increase the number of shares reserved under the plan to 550,000. On February 12, 2013, 226,500 options were granted under the Amended and Restated 2012 Plan (all of which were outstanding at March 31, 2013) with an exercise price of $10.20, a 10 year life and fair value of $7.83. The options vest 1/3rd on February 12, 2014 and 1/36th on the 12th of each month thereafter for 24 months.
The Company measures the fair value of stock options on the date of grant, based on a Binomial option pricing model using certain assumptions discussed in the following paragraph, and the closing market price of the Company's common stock on the date of the grant. Stock options granted vest over a three year period and expire ten years from the date of grant. Share-based compensation expense related to awards is amortized over the applicable vesting period using the straight-line method.
The assumptions used in the valuation of stock options granted during the three months ended March 31, 2013 were as follows:
| Risk-free interest rate | 2.02 | % | ||
| Expected term of option | 6.0 years | |||
| Expected stock price volatility | 99.96 | % | ||
| Expected dividend yield | $ | 0.0 |
The risk-free rate of return is based on the yield of Daily U.S. Treasury Yield Curve Rates with terms equal to the expected life of the options as of the grant date. The expected term of options are determined using the simplified method and the expected stock price volatility is based on comparable companies’ historical stock price volatility since the Company does not have sufficient historical exercise data because its equity shares have been publicly traded for only a limited period of time.
| F-26 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2013 AND 2012 (UNAUDITED)
Share-based compensation expense of $392,323 was recognized for the three month period ended March 31, 2013.
As of March 31, 2013, the Company had approximately $4,122,995 of total unrecognized compensation cost related to non-vested awards granted under the Company’s option plan, which the Company expects to recognize over approximately a three-year period.
NOTE 5 – STOCK WARRANTS
The following table summarizes information with respect to outstanding warrants to purchase common stock of the Company at March 31, 2013:
| Exercise | Number | Number | Expiration | |||||||||
| Price | Outstanding | Vested | Date | |||||||||
| $ | 8.00 | 445,209 | 445,209 | December 2013 | ||||||||
| 12.00 | 456,008 | 447,908 | December 2017 to January 2018 | |||||||||
| 20.00 | 15,288 | 15,288 | January 2014 to January 2015 | |||||||||
| 25.00 | 354,227 | 354,227 | January 2017 to March 2019 | |||||||||
| 1,270,732 | 1,262,632 | |||||||||||
On January 1, 2013, the Company issued warrants to non-employees to purchase 10,800 shares of the Company's common stock at an exercise price of $12.00 per share expiring five years from the date of issuance vesting ratably over twelve months beginning January 1, 2013 in connection with services.
The Company measures the fair value of the vested portion of the issued warrants based on a Binomial option pricing model using certain assumptions discussed in the following paragraph, and the closing market price of the Company's common stock on the date of the fair value determination.
The assumptions used in the valuation of warrants which vested during the three months ended March 31, 2013 were as follows:
| Risk-free interest rate | 0.77 | % | ||
| Life of warrant | 5 years | |||
| Expected stock price volatility | 102.46 | % | ||
| Expected dividend yield | $ | 0.0 |
The risk-free rate of return is based on the yield of Daily U.S. Treasury Yield Curve Rates with terms equal to the life of the warrants as of the grant date. The expected stock price volatility is based on comparable companies’ historical stock price volatility since the Company does not have sufficient historical exercise data because its equity shares have been publicly traded for only a limited period of time.
Compensation of $10,435 related to vested warrants was recognized for the three month period ended March 31, 2013.
As of March 31, 2013, unrecognized compensation related to unvested warrants based on the market price of the Company’s common stock on such date was $31,306.
NOTE 6 – RELATED PARTY TRANSACTIONS
The Company has entered into an agreement with Lederman & Co., LLC (“Lederman & Co”), a company under the control of Dr. Seth Lederman, the Company’s Chief Executive Officer and Chairman of the Board. Effective February 1, 2012, Lederman & Co receives $250,000 per annum for its consulting services. The agreement renews automatically for subsequent terms of one year at $250,000 per annum. Total expenses paid under this agreement were $62,500 and $76,250 during the three months ended March 31, 2013 and 2012, respectively.
| F-27 |
TONIX PHARMACEUTICALS HOLDING CORP.
(a development stage company)
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
MARCH 31, 2013 AND 2012 (UNAUDITED)
NOTE 7 – SUBSEQUENT EVENTS
On April 26, 2013, the Company issued an aggregate of 38,334 shares of common stock in exchange for $306,667 upon exercise of warrants.
| F-28 |
Tonix Pharmaceuticals Holding Corp.

[ * ] Units
Each Unit Consisting of One Share of Common Stock
and
[ * ] of a Warrant, Each to Purchase One Share of Common Stock
PROSPECTUS
Roth Capital Partners
National Securities Corporation
__________________ , 2013
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
ITEM 13. OTHER EXPENSES OF ISSUANCE AND DISTRIBUTION.
The following table sets forth the various expenses to be incurred in connection with the registration of the securities being registered hereby, all of which will be borne by the registrant. All amounts shown are estimates except the SEC registration fee, the FINRA filing fee and the NASDAQ listing fee.
| SEC registration fee | $ | 1,703 | ||
| FINRA filing fee | $ | * | ||
| NASDAQ listing fee | $ | 50,000 | ||
| Legal fees and expenses | $ | * | ||
| Accounting fees and expenses | $ | * | ||
| Transfer agent fees and expenses | $ | * | ||
| Printing and engraving expenses | $ | * | ||
| Miscellaneous expenses | $ | * | ||
| Total | $ | * |
| * | To be completed by amendment. |
ITEM 14. INDEMNIFICATION OF DIRECTORS AND OFFICERS.
Our bylaws, as amended, provide to the fullest extent permitted by Nevada law, our directors or officers shall not be personally liable to us or our shareholders for damages for breach of such director's or officer's fiduciary duty. The effect of this provision of our bylaws, as amended, is to eliminate our right and our shareholders (through shareholders' derivative suits on behalf of our company) to recover damages against a director or officer for breach of the fiduciary duty of care as a director or officer (including breaches resulting from negligent or grossly negligent behavior), except under certain situations defined by statute. We believe that the indemnification provisions in our bylaws, as amended, are necessary to attract and retain qualified persons as directors and officers.
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
ITEM 15. RECENT SALES OF UNREGISTERED SECURITIES.
During the past three years, the registrant has sold the following securities which were not registered under the Securities Act of 1933, as amended.
On October 7, 2011, we issued 1,133,334 shares of our common stock to the shareholders of Tonix Sub in exchange for 100% of the issued and outstanding shares of common stock of Tonix Sub. The shares were issued to accredited investors pursuant to Rule 506 of Regulation D or non-U.S. Persons pursuant to Rule 903 of Regulation S of the Securities Act of 1933, as amended.
| II-1 |
On October 7, 2011, we issued 20,000 shares of our common stock to a placement agent in connection with an amendment to a placement agent agreement. The shares were issued to an accredited investor pursuant to Rule 506 of Regulation D or Section 4(2) of the Securities Act of 1933, as amended.
Between October and November 2011, we sold to certain investors (the “Purchasers”) for aggregate cash proceeds of $1,575,000, secured convertible debentures (the “Debentures”) in the principal face amount of $1,575,000 and the exchange of $500,000 in previously issued notes of Tonix Sub that were converted into Debentures in the principal face amount of $500,000 (the “2011 Financing”). The Debentures were sold to accredited investors pursuant to Rule 506 of Regulation D or non-U.S. Persons pursuant to Rule 903 of Regulation S of the Securities Act of 1933, as amended.
The Debentures mature on the earlier of (i) one year from the date of issuance or (ii) the date of closing of a private placement of equity, equity equivalent, convertible debt or debt financing in which we receive gross proceeds, in one or more transactions, of at least $3,425,000 (a “Subsequent Financing”). The Debentures bear interest at 8% per annum and are convertible at the holder’s option into a Subsequent Financing. In the event that a Subsequent Financing has not occurred within 12 months from the date of issuance of the Debenture, the holder has the option to convert the Debenture into a number of shares of our common stock equal to 1% of our shares of common stock on a fully diluted basis for every $125,000 of Debentures (the “Conversion Shares”).
In addition, upon conversion or repayment of the Debenture, the holder is entitled to receive, at the holder’s option, either (i) a warrant (the “Warrant”) to purchase such number of shares of common stock equal to the principal amount of the Debenture divided by the offering price in a Subsequent Financing (the “Warrant Shares”) or (ii) shares of our common stock equal to 33% of the principal amount of the Debenture divided by the offering price in a Subsequent Financing (the “Incentive Shares”).
In connection with the 2011 Financing, placement agents earned warrants to purchase shares of our common stock equal to 3% or 9% of the gross proceeds delivered by Purchasers introduced by such placement agents in the 2011 Financing divided by the purchase price per share in the Subsequent Financing (collectively, the “2011 Agent Warrants”). In the event that the Subsequent Financing has not occurred within 12 months from the date of issuance of the Debentures, the placement agents will receive, in lieu of the 2011 Agent Warrants, shares of common stock equal to 3% or 9% of the number of shares of our common stock such Purchasers introduced by such placement agent in the 2011 Financing are entitled to receive upon conversion of their Debentures.
Between January and March, 2012, we consummated the 2012 Financing pursuant to which we issued an aggregate of 264.7106 Units to certain investors for aggregate cash proceeds of $4,692,765 and the exchange of $1,925,000 in previously issued debentures that were converted into Units.
Each Unit had a purchase price of $25,000 per Unit and consisted of 1,250 shares of our Common Stock, 1,250 Class A Warrants and 1,250 Class B Warrants.
The Class A Warrants have an exercise price of $25.00 per share of Common Stock and will be exercisable for a period of five years from the date of issuance. The Class B Warrants were not exercisable by the Purchasers and would be exercised automatically on their expiration date by cashless exercise or expire without exercise. Effective April 24, 2012, the Class B Warrants expired unexercised.
In connection with the Financing, we issued Dawson James 23,339 2012 Agent Warrants.
Between October and November 2012, we issued promissory notes in the amount of $320,000 (the “Notes”) in exchange for $320,000 borrowed from six affiliated investors. The Notes bear no interest and were payable on demand.
On November 14, 2012, we sold to accredited investors for aggregate cash proceeds of $390,000, convertible debentures (the “Debentures”) in the principal face amount of $390,000 and the exchange of the Notes for Debentures in the principal face amount of $320,000.
| II-2 |
The Debentures mature on the earlier of (i) November 14, 2013 or (ii) the date of closing of a private placement of equity, equity equivalent, convertible debt or debt financing in which we receive gross proceeds, in one or more transactions, of at least $100,000 (a “Subsequent Financing”). The Debentures bear interest at 8% per annum and are convertible at the holder’s option into either (i) a Subsequent Financing at a price equal to a 25% discount to the price of securities sold in the Subsequent Financing or (ii) shares of our common stock at a conversion price per share equal to $20.00.
In December 2012, the Company issued an aggregate of 8,904,167 units to certain accredited investors for aggregate cash proceeds of $2,615,000, at a price per Unit of $0.40, and the exchange of $710,000 in previously issued convertible debentures (the “Prior Debentures”) of the Company that were converted into Units at a price of $0.30 per Unit.
Each Unit consisted of 0.05 share of the Common Stock, a Class A Warrant to purchase 0.05 share of Common Stock (the “Class A Warrants”), and a Class B Warrant to purchase 0.05 share of Common Stock (the “Class B Warrants” and together with the Class A Warrants, the “Warrants”).The Class A Warrants have an exercise price of $12.00 per share of Common Stock and will be exercisable for a period of five years from the date of issuance. The Class A Warrants may be exercised on a cashless basis under certain circumstances. The Class B Warrants have an exercise price of $8.00 per share of Common Stock and will be exercisable for a period of one year from the date of issuance.
On April 26, 2013, we issued an aggregate of 38,334 shares of common stock in exchange for $306,667 upon exercise of warrants.
ITEM 16. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES.
The following exhibits are included as part of this Form S-1. References to “the Company” in this Exhibit List mean Tonix Pharmaceuticals Holding Corp., a Nevada corporation.
| 1.01* | Form of Underwriting Agreement. |
| 2.01 | Share Exchange Agreement, dated as of October 7, 2011 by and among Tamandare Explorations Inc., David J. Moss, Tonix Pharmaceuticals, Inc. and the shareholders of Tonix Pharmaceuticals, Inc. filed as an exhibit to the Current Report on Form 8-K, filed with the Securities and Exchange Commission (the “Commission”) on October 14, 2011 and incorporated herein by reference. |
| 3.01 | Articles of Incorporation, filed as an exhibit to the Registration Statement on Form S-1, filed with the Commission on April 9, 2008 and incorporated herein by reference. |
| 3.02 | Articles of Merger between Tamandare Explorations Inc. and Tonix Pharmaceuticals Holding Corp., effective October 11, 2011, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 17, 2011 and incorporated herein by reference. |
| 3.03 | Amended and Restated Bylaws, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on February 23, 2012 and incorporated herein by reference. |
| 4.01* | Form of Series A Warrant. |
| 5.01* | Opinion of Sichenzia Ross Friedman Ference LLP. |
| 10.01 | Feasibility and Option Agreement, dated as of June 20, 2007, by and between Krele Pharmaceuticals, Inc. (now, Tonix Pharmaceuticals, Inc.) and Lipocine, Inc., filed as an exhibit to the amended Current Report on Form 8-K/A, filed with the Commission on April 3, 2012 and incorporated herein by reference. † |
| II-3 |
| 10.02 | Consulting Agreement, dated as of June 4, 2010, by and between Krele Pharmaceuticals, Inc. (now, Tonix Pharmaceuticals, Inc.) and Lederman & Co., LLC, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.03 | Technology Transfer and Assignment Agreement, dated as of June 4, 2010, by and between Krele Pharmaceuticals, Inc. (now, Tonix Pharmaceuticals, Inc.) and Lederman & Co., LLC, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.04 | Lease Agreement, dated as of September 28, 2010, by and between 509 Madison Avenue Associates, L.P. and Tonix Pharmaceuticals, Inc., filed as an exhibit to the amended Current Report on Form 8-K/A, filed with the Commission on February 3, 2012 and incorporated herein by reference. |
| 10.05 | Amendment to Feasibility and Option Agreement, dated as of October 4, 2010, by and between Tonix Pharmaceuticals, Inc. and Lipocine, Inc., filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. † |
| 10.06 | Engagement Agreement, dated as of October 6, 2010, by and between Tonix Pharmaceuticals, Inc. and Frost and Sullivan, filed as an exhibit to the amended Current Report on Form 8-K/A, filed with the Commission on April 3, 2012 and incorporated herein by reference. |
| 10.07 | Amendment to Consulting Agreement, dated as of December 9, 2010, by and between Tonix Pharmaceuticals, Inc. and Lederman & Co., LLC, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.08 | Employment Agreement, dated as of April 1, 2011, by and between Tonix Pharmaceuticals, Inc. and Rhonda Rosen, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.09 | Employment Agreement, dated as of April 1, 2011, by and between Tonix Pharmaceuticals, Inc. and Benjamin A. Selzer, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.10 | Employment Agreement, dated as of April 1, 2011, by and between Tonix Pharmaceuticals, Inc. and Susan Oliver (now, Susan Kerridge), filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.11 | API Supply and Development Agreement, dated as of April 7, 2011, by and between Tonix Pharmaceuticals, Inc. and JFC Technologies, Inc., filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.12 | Consulting Agreement, dated as of June 2, 2011, by and between Tonix Pharmaceuticals, Inc. and Pharmanet Canada, Inc., filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.13 | Amendment to Employment Agreement, dated as of July 27, 2011, by and between Tonix Pharmaceuticals, Inc. and Rhonda Rosen, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.14 | Amendment to Employment Agreement, dated as of July 27, 2011, by and between Tonix Pharmaceuticals, Inc. and Benjamin A. Selzer, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.15 | Amendment to Employment Agreement, dated as of July 27, 2011, by and between Tonix Pharmaceuticals, Inc. and Susan Oliver (now, Susan Kerridge), filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| II-4 |
| 10.16 | Financial Public Relations Agreement, dated as of August 1, 2011, by and between Tonix Pharmaceuticals, Inc. and Porter, LeVay & Rose, Inc., filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.17 | Form of 8% Secured Convertible Debenture, issued October 7, 2011, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.18 | Form of Subscription Agreement, dated October 7, 2011, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.19 | Form of Pledge and Security Agreement, dated as of October 7, 2011, by and among Tamandare Explorations Inc., Tonix Pharmaceuticals, Inc., Krele LLC and the investors, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.20 | Form of Subsidiary Guaranty, dated as of October 7, 2011, by and among Tonix Pharmaceuticals, Inc., Krele LLC and Sandor Capital Master Fund L.P., on behalf of the investors, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 10.21 | Form of Subscription Agreement, dated January 20, 2012, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on January 23, 2012 and incorporated herein by reference. |
| 10.22 | Form of Class A Warrant, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on January 23, 2012 and incorporated herein by reference. |
| 10.23 | Form of Class B Warrant, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on January 23, 2012 and incorporated herein by reference. |
| 10.24 | Form of Registration Rights Agreement, dated January 20, 2012, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on January 23, 2012 and incorporated herein by reference. |
| 10.25 | Amendment to Consulting Agreement, dated as of March 30, 2012 but effective as of July 27, 2011, by and between Tonix Pharmaceuticals, Inc. and Lederman & Co., LLC, filed as an exhibit to the Annual Report on Form 10-K filed with the Commission on March 30, 2012 and incorporated herein by reference. |
| 10.26 | Employment Agreement, between Tonix Pharmaceuticals Holding Corp. and Leland Gershell, dated April 1, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on April 5, 2012 and incorporated herein by reference. |
| 10.27 | Employment Agreement, between Tonix Pharmaceuticals Holding Corp. and Benjamin Selzer, dated April 2, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on April 5, 2012 and incorporated herein by reference. |
| 10.28 | Amendment to Employment Agreement, between Tonix Pharmaceuticals Holding Corp. and Benjamin Selzer, dated October 5, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on October 10, 2012 and incorporated herein by reference. |
| 10.29 | Form of Subscription Agreement, dated November 13, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on November 14, 2012 and incorporated herein by reference. |
| II-5 |
| 10.30 | Form of Convertible Debenture, dated November 13, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on November 14, 2012 and incorporated herein by reference. |
| 10.31 | Form of Subscription Agreement, dated December 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 5, 2012 and incorporated herein by reference. |
| 10.32 | Form of Class A Warrant, dated December 4, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 5, 2012 and incorporated herein by reference. |
| 10.33 | Form of Class B Warrant, dated December 4, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 5, 2012 and incorporated herein by reference. |
| 10.34 | Form of Registration Rights Agreement, dated December 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 5, 2012 and incorporated herein by reference. |
| 10.35 | Form of Class A Warrant, dated December 21, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 27, 2012 and incorporated herein by reference. |
| 10.36 | Form of Class B Warrant, dated December 21, 2012, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 27, 2012 and incorporated herein by reference. |
| 10.37 | Form of Amendment No. 1 to the Purchase Agreement, Registration Rights Agreement and Escrow Agreement, filed as an exhibit to the Current Report on Form 8-K filed with the Commission on December 27, 2012 and incorporated herein by reference. |
| 21.01 | List of Subsidiaries. |
| 23.01 | Consent of EisnerAmper LLP. |
| 23.02 | Consent of Sichenzia Ross Friedman Ference LLP (included in Exhibit 5.01). |
| 24.01 | Power of Attorney (included on signature page to the registration statement). |
| 99.01 | Frost & Sullivan Fibromyalgia Market Study, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
| 99.02 | Lipocine Cyclobenzaprine Study Results, filed as an exhibit to the Current Report on Form 8-K, filed with the Commission on October 14, 2011 and incorporated herein by reference. |
* To be filed by amendment.
† Confidential treatment granted for certain confidential portions of this exhibit pursuant to Rule 24b-2 under the Exchange Act. In accordance with Rule 24b-2, these confidential portions have been omitted from this exhibit and filed separately with the Commission.
ITEM 17. UNDERTAKINGS.
The undersigned registrant hereby undertakes that:
(1) For purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective.
| II-6 |
(2) For the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
| II-7 |
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the registrant has duly caused this Registration Statement on Form S-1 to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of New York, State of New York, on this 10th day of May, 2013.
| TONIX PHARMACEUTICALS HOLDING CORP. | ||
| Date: May 10, 2013 | By: | /s/ SETH LEDERMAN |
| Seth Lederman | ||
| Chief Executive Officer (Principal Executive Officer) | ||
| Date: May 10, 2013 | By: | /s/ LELAND GERSHELL |
| Leland Gershell | ||
| Chief Financial Officer (Principal Accounting Officer) | ||
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS:
That the undersigned officers and directors of Tonix Pharmaceuticals Holding Corp., a Nevada corporation, do hereby constitute and appoint Seth Lederman and Leland Gershell and each of them his or her true and lawful attorney-in-fact and agent with full power and authority to do any and all acts and things and to execute any and all instruments which said attorney and agent, determine may be necessary or advisable or required to enable said corporation to comply with the Securities Act of 1933, as amended, and any rules or regulations or requirements of the Securities and Exchange Commission in connection with this Registration Statement. Without limiting the generality of the foregoing power and authority, the powers granted include the power and authority to sign the names of the undersigned officers and directors in the capacities indicated below to this Registration Statement, and to any and all instruments or documents filed as part of or in conjunction with this Registration Statement or amendments or supplements thereof, including post-effective amendments, to this Registration Statement or any registration statement relating to this offering to be effective upon filing pursuant to Rule 462(b) under the Securities Act of 1933, and each of the undersigned hereby ratifies and confirms that said attorney and agent, shall do or cause to be done by virtue thereof. This Power of Attorney may be signed in several counterparts.
IN WITNESS WHEREOF, each of the undersigned has executed this Power of Attorney. In accordance with the requirements of the Securities Act of 1933, as amended, this registration statement was signed by the following persons in the capacities and on the dates stated:
| Signature | Title | Date | ||
| /s/ SETH LEDERMAN | Chief Executive Officer (Principal Executive Officer) and Director | May 10, 2013 | ||
| Seth Lederman | ||||
| /s/ LELAND GERSHELL | Chief Financial Officer (Principal Financial Officer and Principal Accounting Officer) | May 10, 2013 | ||
| Leland Gershell | ||||
| /s/ STUART DAVIDSON | Director | May 10, 2013 | ||
| Stuart Davidson |
| II-8 |
| /s/ PATRICK GRACE | Director | May 10, 2013 | ||
| Patrick Grace | ||||
| /s/ DONALD W. LANDRY | Director | May 10, 2013 | ||
| Donald W. Landry | ||||
| /s/ ERNEST MARIO | Director | May 10, 2013 | ||
| Ernest Mario | ||||
| /s/ CHARLES MATHER IV | Director | May 10, 2013 | ||
| Charles Mather IV | ||||
| /s/ JOHN RHODES | Director | May 10, 2013 | ||
| John Rhodes | ||||
| /s/ SAMUEL SAKS | Director | May 10, 2013 | ||
| Samuel Saks |
| II-9 |
